User login
WHO’s psychosocial guidelines for Zika are a useful tool
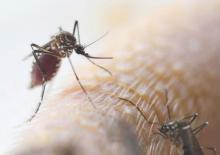
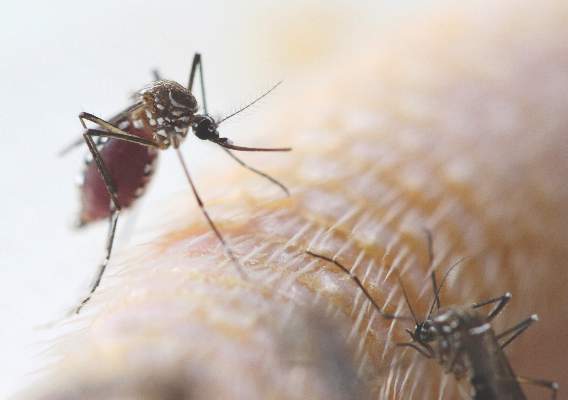
As the Zika virus spreads across 31 countries in the Americas, bringing with it the threat of microcephaly seen in Brazil, local and international public health experts are scrambling to assess the extent of the threat. Systems for epidemiologic surveillance are emerging, as are guidelines for pregnant women and those of childbearing age.
Amid all of this is the World Health Organization’s recently released guidelines, “Psychosocial Support for Pregnant Women and for Families With Microcephaly and Other Neurological Complications in the Context of Zika Virus” (http://who.int/csr/resources/publications/zika/psychosocial-support/en). These guidelines, an adaptation of previous interventions used in disasters, are a helpful resource for physicians.
The guidelines emphasize eight areas: having accurate information, what information is conveyed, how that information is conveyed, understanding common distress reactions, providing basic support, strengthening social support, teaching stress reduction, and educating mothers about parenting children with microcephaly. Readers familiar with psychological first aid (PFA), used in disaster response to provide basic psychological support, will recognize these elements as distillations of PFA specifically for Zika.
PFA focuses on addressing peoples’ basic physiologic, safety, and social needs as a means of addressing their overall psychological needs. It has been promulgated as a best practice by the National Institute of Mental Health since the Sept. 11, 2001, terrorist attacks.
Another useful aspect of the guidelines is that they are aimed at health professionals in general, rather than mental health professionals in particular. This approach makes sense, because women concerned about potential infection with Zika, of course, are not going to go to a mental health professional to address their anxiety but to an internist, family physician, ob.gyn., or possibly a pediatrician. They are understandably focused on the distressing problem rather than on the distress itself.
Health care professionals now on the frontlines of the Zika public health response may naturally be following many of the principles in the psychosocial guidelines. Nevertheless, they probably would benefit from reviewing them in order to lend some more structure to the psychosocial soundness of their practice. In addition, becoming aware of the guidelines might help those health professionals deal with their own risk for burnout.
In discussing common distress reactions, the guidelines note that severely distressed individuals should be referred for “specialized care,” which means a mental health professional when psychosocial care is discussed in low-resource settings around the world. The overwhelming majority of countries in the Americas where Zika transmission has been reported are low- and middle-income countries, according to the World Bank’s ranking system. As such, they are surely the places where governments are most likely to devote the least amount of their health care budgets to mental health services.
And, even in high-income countries among the list of Zika-affected countries, it is not clear from consulting the World Health Organization’s 2014 Mental Health Atlas that even those countries fund anything more than inpatient psychiatric care (for example, information from Barbados lists only inpatient psychiatric resources). My point? The ranks of mental health professionals in Zika-affected countries who might benefit from the WHO’s Zika guidelines probably are few. Those who are there probably are overwhelmed tending to the preexisting (mostly inpatient) psychiatric needs of their countries.
Fortunately, the WHO’s Mental Health Atlas shows Brazil to be an exception with a comparatively robust outpatient as well as inpatient public mental health system. This is fortuitous given that Brazil for the moment is the center of the surge in microcephaly. Hopefully, the Northeastern region of Brazil, where that surge is highest, has a proportionate share of Brazil’s mental health resources. For mental health professionals there and potentially elsewhere in Zika-affected countries, the guidelines for psychosocial support can prove to be an essential tool.
To the extent that mental health professionals can and should provide support to their Zika-affected communities, these guidelines will help them to “stand down” from a traditional psychiatric model of care to a more normalizing one, where diagnoses and treatment are not the focus of attention. This was certainly the case in the comparable setting of post-Ebola Liberia, where mental health clinicians trained by the Carter Center found PFA central to what they could offer their devastated communities and to gaining unprecedented acceptance from those communities (unpublished observations).
As of this writing, the Zika virus has not spared the continental United States. Florida has been hit the hardest, followed by Texas, according to data from the Centers for Disease Control and Prevention. Those of us looking to help patients deal with the possibility of coming in contact with the Zika virus should remember the WHO psychosocial guidelines. They can help health professionals integrate mental health into their practices, and help mental health professionals transfer their skills and knowledge to their communities.
Dr. Katz is associate clinical professor of psychiatry and medical education, and director of the program in global mental health, at the Icahn School of Medicine at Mount Sinai, New York.
As the Zika virus spreads across 31 countries in the Americas, bringing with it the threat of microcephaly seen in Brazil, local and international public health experts are scrambling to assess the extent of the threat. Systems for epidemiologic surveillance are emerging, as are guidelines for pregnant women and those of childbearing age.
Amid all of this is the World Health Organization’s recently released guidelines, “Psychosocial Support for Pregnant Women and for Families With Microcephaly and Other Neurological Complications in the Context of Zika Virus” (http://who.int/csr/resources/publications/zika/psychosocial-support/en). These guidelines, an adaptation of previous interventions used in disasters, are a helpful resource for physicians.
The guidelines emphasize eight areas: having accurate information, what information is conveyed, how that information is conveyed, understanding common distress reactions, providing basic support, strengthening social support, teaching stress reduction, and educating mothers about parenting children with microcephaly. Readers familiar with psychological first aid (PFA), used in disaster response to provide basic psychological support, will recognize these elements as distillations of PFA specifically for Zika.
PFA focuses on addressing peoples’ basic physiologic, safety, and social needs as a means of addressing their overall psychological needs. It has been promulgated as a best practice by the National Institute of Mental Health since the Sept. 11, 2001, terrorist attacks.
Another useful aspect of the guidelines is that they are aimed at health professionals in general, rather than mental health professionals in particular. This approach makes sense, because women concerned about potential infection with Zika, of course, are not going to go to a mental health professional to address their anxiety but to an internist, family physician, ob.gyn., or possibly a pediatrician. They are understandably focused on the distressing problem rather than on the distress itself.
Health care professionals now on the frontlines of the Zika public health response may naturally be following many of the principles in the psychosocial guidelines. Nevertheless, they probably would benefit from reviewing them in order to lend some more structure to the psychosocial soundness of their practice. In addition, becoming aware of the guidelines might help those health professionals deal with their own risk for burnout.
In discussing common distress reactions, the guidelines note that severely distressed individuals should be referred for “specialized care,” which means a mental health professional when psychosocial care is discussed in low-resource settings around the world. The overwhelming majority of countries in the Americas where Zika transmission has been reported are low- and middle-income countries, according to the World Bank’s ranking system. As such, they are surely the places where governments are most likely to devote the least amount of their health care budgets to mental health services.
And, even in high-income countries among the list of Zika-affected countries, it is not clear from consulting the World Health Organization’s 2014 Mental Health Atlas that even those countries fund anything more than inpatient psychiatric care (for example, information from Barbados lists only inpatient psychiatric resources). My point? The ranks of mental health professionals in Zika-affected countries who might benefit from the WHO’s Zika guidelines probably are few. Those who are there probably are overwhelmed tending to the preexisting (mostly inpatient) psychiatric needs of their countries.
Fortunately, the WHO’s Mental Health Atlas shows Brazil to be an exception with a comparatively robust outpatient as well as inpatient public mental health system. This is fortuitous given that Brazil for the moment is the center of the surge in microcephaly. Hopefully, the Northeastern region of Brazil, where that surge is highest, has a proportionate share of Brazil’s mental health resources. For mental health professionals there and potentially elsewhere in Zika-affected countries, the guidelines for psychosocial support can prove to be an essential tool.
To the extent that mental health professionals can and should provide support to their Zika-affected communities, these guidelines will help them to “stand down” from a traditional psychiatric model of care to a more normalizing one, where diagnoses and treatment are not the focus of attention. This was certainly the case in the comparable setting of post-Ebola Liberia, where mental health clinicians trained by the Carter Center found PFA central to what they could offer their devastated communities and to gaining unprecedented acceptance from those communities (unpublished observations).
As of this writing, the Zika virus has not spared the continental United States. Florida has been hit the hardest, followed by Texas, according to data from the Centers for Disease Control and Prevention. Those of us looking to help patients deal with the possibility of coming in contact with the Zika virus should remember the WHO psychosocial guidelines. They can help health professionals integrate mental health into their practices, and help mental health professionals transfer their skills and knowledge to their communities.
Dr. Katz is associate clinical professor of psychiatry and medical education, and director of the program in global mental health, at the Icahn School of Medicine at Mount Sinai, New York.
As the Zika virus spreads across 31 countries in the Americas, bringing with it the threat of microcephaly seen in Brazil, local and international public health experts are scrambling to assess the extent of the threat. Systems for epidemiologic surveillance are emerging, as are guidelines for pregnant women and those of childbearing age.
Amid all of this is the World Health Organization’s recently released guidelines, “Psychosocial Support for Pregnant Women and for Families With Microcephaly and Other Neurological Complications in the Context of Zika Virus” (http://who.int/csr/resources/publications/zika/psychosocial-support/en). These guidelines, an adaptation of previous interventions used in disasters, are a helpful resource for physicians.
The guidelines emphasize eight areas: having accurate information, what information is conveyed, how that information is conveyed, understanding common distress reactions, providing basic support, strengthening social support, teaching stress reduction, and educating mothers about parenting children with microcephaly. Readers familiar with psychological first aid (PFA), used in disaster response to provide basic psychological support, will recognize these elements as distillations of PFA specifically for Zika.
PFA focuses on addressing peoples’ basic physiologic, safety, and social needs as a means of addressing their overall psychological needs. It has been promulgated as a best practice by the National Institute of Mental Health since the Sept. 11, 2001, terrorist attacks.
Another useful aspect of the guidelines is that they are aimed at health professionals in general, rather than mental health professionals in particular. This approach makes sense, because women concerned about potential infection with Zika, of course, are not going to go to a mental health professional to address their anxiety but to an internist, family physician, ob.gyn., or possibly a pediatrician. They are understandably focused on the distressing problem rather than on the distress itself.
Health care professionals now on the frontlines of the Zika public health response may naturally be following many of the principles in the psychosocial guidelines. Nevertheless, they probably would benefit from reviewing them in order to lend some more structure to the psychosocial soundness of their practice. In addition, becoming aware of the guidelines might help those health professionals deal with their own risk for burnout.
In discussing common distress reactions, the guidelines note that severely distressed individuals should be referred for “specialized care,” which means a mental health professional when psychosocial care is discussed in low-resource settings around the world. The overwhelming majority of countries in the Americas where Zika transmission has been reported are low- and middle-income countries, according to the World Bank’s ranking system. As such, they are surely the places where governments are most likely to devote the least amount of their health care budgets to mental health services.
And, even in high-income countries among the list of Zika-affected countries, it is not clear from consulting the World Health Organization’s 2014 Mental Health Atlas that even those countries fund anything more than inpatient psychiatric care (for example, information from Barbados lists only inpatient psychiatric resources). My point? The ranks of mental health professionals in Zika-affected countries who might benefit from the WHO’s Zika guidelines probably are few. Those who are there probably are overwhelmed tending to the preexisting (mostly inpatient) psychiatric needs of their countries.
Fortunately, the WHO’s Mental Health Atlas shows Brazil to be an exception with a comparatively robust outpatient as well as inpatient public mental health system. This is fortuitous given that Brazil for the moment is the center of the surge in microcephaly. Hopefully, the Northeastern region of Brazil, where that surge is highest, has a proportionate share of Brazil’s mental health resources. For mental health professionals there and potentially elsewhere in Zika-affected countries, the guidelines for psychosocial support can prove to be an essential tool.
To the extent that mental health professionals can and should provide support to their Zika-affected communities, these guidelines will help them to “stand down” from a traditional psychiatric model of care to a more normalizing one, where diagnoses and treatment are not the focus of attention. This was certainly the case in the comparable setting of post-Ebola Liberia, where mental health clinicians trained by the Carter Center found PFA central to what they could offer their devastated communities and to gaining unprecedented acceptance from those communities (unpublished observations).
As of this writing, the Zika virus has not spared the continental United States. Florida has been hit the hardest, followed by Texas, according to data from the Centers for Disease Control and Prevention. Those of us looking to help patients deal with the possibility of coming in contact with the Zika virus should remember the WHO psychosocial guidelines. They can help health professionals integrate mental health into their practices, and help mental health professionals transfer their skills and knowledge to their communities.
Dr. Katz is associate clinical professor of psychiatry and medical education, and director of the program in global mental health, at the Icahn School of Medicine at Mount Sinai, New York.
Zika more complex public health challenge than Ebola
The vector-borne and often asymptomatic nature of Zika virus infection makes it a more complex public health challenge than Ebola, researchers say.
The Zika virus was first isolated from a macaque in Uganda in 1947, and has historically been restricted to Africa and Asia. However since its introduction to Brazil in 2014 or early 2015, possibly via Polynesia, it has spread rapidly and estimates now point to between 440,000 and 1,300,000 cases of Zika virus infection in Brazil during 2015.
In the past, Zika virus infection in adults has presented with non-life-threatening symptoms including mild fever, maculopapular rash, arthralgia, myalgia, headache, retro-orbital pain and vomiting.
Writing in the March 1 online edition of PLoS Neglected Tropical Diseases, researchers say the viral variant currently associated with the outbreak in Brazil is presenting a new and more challenging public health problem.
“What makes this outbreak a high priority global public health concern is the association with incidence of birth defects involving the central nervous system and the apparent increased incidence of Guillain-Barré syndrome,” wrote Dr. Robert W. Malone, from RW Malone MD, and his coauthors (PLoS Negl Trop Dis. 2016 Mar 1. doi: 10.1371/journal.pntd.0004530).
They cited one retrospective study in French Polynesia that suggested there was a ratio of one case of Zika-associated Guillain-Barré syndrome for every 208 suspected cases of Zika virus infection.
The outbreak has also been linked to an unusually high incidence of the otherwise rare microcephaly, with Brazil recording a 20-fold increase in incidence during 2015. The connection with Zika virus is supported by a case study in which large numbers of viral particles were found in the central nervous system tissue of a microcephalic Zika-infected fetus.
Based on estimates of the overall incidence of Zika virus infection, researchers have calculated that Brazilian mothers infected with the virus are 3,700-11,000 times more likely to deliver infants with primary microcephaly, compared with those who are not infected.
There are still some key uncertainties around the transmission of Zika virus, the authors said.
“The degree to which humans, nonhuman primates, or other animals can amplify and transmit the virus to insect vectors is poorly understood,” they wrote. “The typical range and types of insect vectors observed in the past may not be predictive for the virus now circulating in the Americas [and] infectivity of the circulating strain, viremia levels, duration, and risk of occult persistence are not yet understood.”
The virus is transmitted primarily by mosquito vectors such as Aedes aegypti and Aedes albopictus, with primates – including humans – the best documented animal reservoir.
“Recent reports indicate the potential for both human blood-borne and sexual transmission of Zika virus, including prolonged presence of virus in semen,” the authors wrote.
The virus has also been found in the saliva of infected individuals, and viral sequences have been identified in breast milk.Commenting on possible medical countermeasures to combat the spread and impact of the Zika virus, the paper’s authors noted that due to the absence of an existing vaccine and long potential development times for candidate vaccines, other prophylactics and therapeutics need to be explored.
In particular, they called for development and deployment of Zika diagnostics to regional clinical health laboratories, discussions between obstetricians and patients about the risks to ongoing or planned pregnancies, and resources for neurologists dealing with “unprecedented” Guillain-Barré syndrome outbreaks.
“Perhaps the biggest challenge with Zika will be to recognize it for what it is: a new disease which does not fit the epidemiology or response paradigm of Ebola or dengue and which will demand effort, resources, unparalleled collaboration, and above all, open mindedness in formulating responses.”
Two authors declared employment with and equity holdings in RW Malone MD, and two authors declared employment with – and one of these also declared equity holdings in – Nanotherapeutics.
The vector-borne and often asymptomatic nature of Zika virus infection makes it a more complex public health challenge than Ebola, researchers say.
The Zika virus was first isolated from a macaque in Uganda in 1947, and has historically been restricted to Africa and Asia. However since its introduction to Brazil in 2014 or early 2015, possibly via Polynesia, it has spread rapidly and estimates now point to between 440,000 and 1,300,000 cases of Zika virus infection in Brazil during 2015.
In the past, Zika virus infection in adults has presented with non-life-threatening symptoms including mild fever, maculopapular rash, arthralgia, myalgia, headache, retro-orbital pain and vomiting.
Writing in the March 1 online edition of PLoS Neglected Tropical Diseases, researchers say the viral variant currently associated with the outbreak in Brazil is presenting a new and more challenging public health problem.
“What makes this outbreak a high priority global public health concern is the association with incidence of birth defects involving the central nervous system and the apparent increased incidence of Guillain-Barré syndrome,” wrote Dr. Robert W. Malone, from RW Malone MD, and his coauthors (PLoS Negl Trop Dis. 2016 Mar 1. doi: 10.1371/journal.pntd.0004530).
They cited one retrospective study in French Polynesia that suggested there was a ratio of one case of Zika-associated Guillain-Barré syndrome for every 208 suspected cases of Zika virus infection.
The outbreak has also been linked to an unusually high incidence of the otherwise rare microcephaly, with Brazil recording a 20-fold increase in incidence during 2015. The connection with Zika virus is supported by a case study in which large numbers of viral particles were found in the central nervous system tissue of a microcephalic Zika-infected fetus.
Based on estimates of the overall incidence of Zika virus infection, researchers have calculated that Brazilian mothers infected with the virus are 3,700-11,000 times more likely to deliver infants with primary microcephaly, compared with those who are not infected.
There are still some key uncertainties around the transmission of Zika virus, the authors said.
“The degree to which humans, nonhuman primates, or other animals can amplify and transmit the virus to insect vectors is poorly understood,” they wrote. “The typical range and types of insect vectors observed in the past may not be predictive for the virus now circulating in the Americas [and] infectivity of the circulating strain, viremia levels, duration, and risk of occult persistence are not yet understood.”
The virus is transmitted primarily by mosquito vectors such as Aedes aegypti and Aedes albopictus, with primates – including humans – the best documented animal reservoir.
“Recent reports indicate the potential for both human blood-borne and sexual transmission of Zika virus, including prolonged presence of virus in semen,” the authors wrote.
The virus has also been found in the saliva of infected individuals, and viral sequences have been identified in breast milk.Commenting on possible medical countermeasures to combat the spread and impact of the Zika virus, the paper’s authors noted that due to the absence of an existing vaccine and long potential development times for candidate vaccines, other prophylactics and therapeutics need to be explored.
In particular, they called for development and deployment of Zika diagnostics to regional clinical health laboratories, discussions between obstetricians and patients about the risks to ongoing or planned pregnancies, and resources for neurologists dealing with “unprecedented” Guillain-Barré syndrome outbreaks.
“Perhaps the biggest challenge with Zika will be to recognize it for what it is: a new disease which does not fit the epidemiology or response paradigm of Ebola or dengue and which will demand effort, resources, unparalleled collaboration, and above all, open mindedness in formulating responses.”
Two authors declared employment with and equity holdings in RW Malone MD, and two authors declared employment with – and one of these also declared equity holdings in – Nanotherapeutics.
The vector-borne and often asymptomatic nature of Zika virus infection makes it a more complex public health challenge than Ebola, researchers say.
The Zika virus was first isolated from a macaque in Uganda in 1947, and has historically been restricted to Africa and Asia. However since its introduction to Brazil in 2014 or early 2015, possibly via Polynesia, it has spread rapidly and estimates now point to between 440,000 and 1,300,000 cases of Zika virus infection in Brazil during 2015.
In the past, Zika virus infection in adults has presented with non-life-threatening symptoms including mild fever, maculopapular rash, arthralgia, myalgia, headache, retro-orbital pain and vomiting.
Writing in the March 1 online edition of PLoS Neglected Tropical Diseases, researchers say the viral variant currently associated with the outbreak in Brazil is presenting a new and more challenging public health problem.
“What makes this outbreak a high priority global public health concern is the association with incidence of birth defects involving the central nervous system and the apparent increased incidence of Guillain-Barré syndrome,” wrote Dr. Robert W. Malone, from RW Malone MD, and his coauthors (PLoS Negl Trop Dis. 2016 Mar 1. doi: 10.1371/journal.pntd.0004530).
They cited one retrospective study in French Polynesia that suggested there was a ratio of one case of Zika-associated Guillain-Barré syndrome for every 208 suspected cases of Zika virus infection.
The outbreak has also been linked to an unusually high incidence of the otherwise rare microcephaly, with Brazil recording a 20-fold increase in incidence during 2015. The connection with Zika virus is supported by a case study in which large numbers of viral particles were found in the central nervous system tissue of a microcephalic Zika-infected fetus.
Based on estimates of the overall incidence of Zika virus infection, researchers have calculated that Brazilian mothers infected with the virus are 3,700-11,000 times more likely to deliver infants with primary microcephaly, compared with those who are not infected.
There are still some key uncertainties around the transmission of Zika virus, the authors said.
“The degree to which humans, nonhuman primates, or other animals can amplify and transmit the virus to insect vectors is poorly understood,” they wrote. “The typical range and types of insect vectors observed in the past may not be predictive for the virus now circulating in the Americas [and] infectivity of the circulating strain, viremia levels, duration, and risk of occult persistence are not yet understood.”
The virus is transmitted primarily by mosquito vectors such as Aedes aegypti and Aedes albopictus, with primates – including humans – the best documented animal reservoir.
“Recent reports indicate the potential for both human blood-borne and sexual transmission of Zika virus, including prolonged presence of virus in semen,” the authors wrote.
The virus has also been found in the saliva of infected individuals, and viral sequences have been identified in breast milk.Commenting on possible medical countermeasures to combat the spread and impact of the Zika virus, the paper’s authors noted that due to the absence of an existing vaccine and long potential development times for candidate vaccines, other prophylactics and therapeutics need to be explored.
In particular, they called for development and deployment of Zika diagnostics to regional clinical health laboratories, discussions between obstetricians and patients about the risks to ongoing or planned pregnancies, and resources for neurologists dealing with “unprecedented” Guillain-Barré syndrome outbreaks.
“Perhaps the biggest challenge with Zika will be to recognize it for what it is: a new disease which does not fit the epidemiology or response paradigm of Ebola or dengue and which will demand effort, resources, unparalleled collaboration, and above all, open mindedness in formulating responses.”
Two authors declared employment with and equity holdings in RW Malone MD, and two authors declared employment with – and one of these also declared equity holdings in – Nanotherapeutics.
FROM PLOS NEGLECTED TROPICAL DISEASES
Stronger evidence links Zika to Guillain-Barré syndrome
Serological evidence from French Polynesia links an outbreak of Zika virus to a spike in cases of Guillain-Barré syndrome seen there in 2013-2014.
The research, published online Feb. 29 in The Lancet, is the first to use a case-control design to demonstrate that Zika, a mosquito-borne flavivirus, is associated with Guillain-Barré syndrome (Lancet. 2016 Feb 29. doi: 10.1016/S0140-6736(16)00562-6).
Guillain-Barré syndrome (GBS) is an immune-mediated flaccid paralysis that can follow viral or bacterial infections. Most patients with GBS recover with intensive care in hospitals, although the syndrome can be permanently debilitating or, in rare cases, fatal.
As a large outbreak of Zika continues in Central and South America, hospitals should be prepared for excess GBS cases, the authors of the study say, and assure adequate intensive-care capacity to treat them. Based on the 66% attack rate of Zika during the French Polynesia outbreak, investigators estimated the incidence of GBS at 0.24 per 1,000 Zika infections, but noted that it could be different in the current outbreak.
Dr. Van-Mai Cao-Lormeau of the Unit of Emerging Infectious Diseases at Institut Louis Malardé in Papeete, French Polynesia, alongside colleagues in France and French Polynesia, used a case-control design to compare serological samples from 42 patients (74% male) diagnosed at a Tahiti hospital with GBS with samples from age-and sex-matched patients who presented at the same hospital, also during the time of the outbreak, with a nonfebrile illness (n = 98) or with acute Zika disease without neurological symptoms (n = 70).
The investigators found that all but one patient with GBS had Zika virus antibodies, and all of them had neutralizing antibodies to Zika virus. By comparison, only 56% (n = 54) of the control group admitted with nonfebrile illness had neutralizing antibodies (P less than .0001).
Also, 93% of the GBS patients had Zika virus immunoglobulin M (IgM) and 88% reported symptoms consistent with Zika infection a mean of 6 days before onset of neurological symptoms. Acute Zika infection is usually characterized by rash, fever, and conjunctivitis.
Past dengue virus infection, which had been considered a possible risk factor for Zika-mediated GBS, did not differ significantly between patients in the control groups and those with GBS.
The investigators were also able to subtype the clinical characteristics of the GBS cases as consistent with acute motor axonal neuropathy, or AMAN, phenotype. However, the antibodies typically seen associated with AMAN were not seen in these patients, leading investigators to suspect that a different biological pathway was responsible.
More than a third of the GBS patients in the study required intensive care, most of these also with respiratory support, though none died.
The government of France, the European Union, and the Wellcome Trust funded the study. The researchers declared that they had no competing interests.
Zika virus can be added to our list of viruses that can cause Guillain-Barré syndrome, and investigation of these cases should include tests for Zika when there is a possibility of infection by that virus. Whether Zika will be proven to pose a greater threat in causing Guillain-Barré syndrome than its various flavivirus cousins remains to be determined. A little caution should be taken because the data are still scarce and we do not know whether the current Zika virus is identical to that in previous outbreaks, whether it will behave exactly the same in a different population with a different genetic and immunity background, or whether a cofactor or co-infection is responsible. Reassuringly, the investigators did not find any evidence that previous dengue infection enhanced the severity of the disease, which could substantially have increased the threat in areas of regular activity.
Dr. David W. Smith is a clinical professor of pathology and laboratory medicine at the University of Western Australia in Perth. John Mackenzie, Ph.D., is a professor of tropical and infectious diseases at Curtin University in Bentley, Australia. They had no competing interests to disclose.
Zika virus can be added to our list of viruses that can cause Guillain-Barré syndrome, and investigation of these cases should include tests for Zika when there is a possibility of infection by that virus. Whether Zika will be proven to pose a greater threat in causing Guillain-Barré syndrome than its various flavivirus cousins remains to be determined. A little caution should be taken because the data are still scarce and we do not know whether the current Zika virus is identical to that in previous outbreaks, whether it will behave exactly the same in a different population with a different genetic and immunity background, or whether a cofactor or co-infection is responsible. Reassuringly, the investigators did not find any evidence that previous dengue infection enhanced the severity of the disease, which could substantially have increased the threat in areas of regular activity.
Dr. David W. Smith is a clinical professor of pathology and laboratory medicine at the University of Western Australia in Perth. John Mackenzie, Ph.D., is a professor of tropical and infectious diseases at Curtin University in Bentley, Australia. They had no competing interests to disclose.
Zika virus can be added to our list of viruses that can cause Guillain-Barré syndrome, and investigation of these cases should include tests for Zika when there is a possibility of infection by that virus. Whether Zika will be proven to pose a greater threat in causing Guillain-Barré syndrome than its various flavivirus cousins remains to be determined. A little caution should be taken because the data are still scarce and we do not know whether the current Zika virus is identical to that in previous outbreaks, whether it will behave exactly the same in a different population with a different genetic and immunity background, or whether a cofactor or co-infection is responsible. Reassuringly, the investigators did not find any evidence that previous dengue infection enhanced the severity of the disease, which could substantially have increased the threat in areas of regular activity.
Dr. David W. Smith is a clinical professor of pathology and laboratory medicine at the University of Western Australia in Perth. John Mackenzie, Ph.D., is a professor of tropical and infectious diseases at Curtin University in Bentley, Australia. They had no competing interests to disclose.
Serological evidence from French Polynesia links an outbreak of Zika virus to a spike in cases of Guillain-Barré syndrome seen there in 2013-2014.
The research, published online Feb. 29 in The Lancet, is the first to use a case-control design to demonstrate that Zika, a mosquito-borne flavivirus, is associated with Guillain-Barré syndrome (Lancet. 2016 Feb 29. doi: 10.1016/S0140-6736(16)00562-6).
Guillain-Barré syndrome (GBS) is an immune-mediated flaccid paralysis that can follow viral or bacterial infections. Most patients with GBS recover with intensive care in hospitals, although the syndrome can be permanently debilitating or, in rare cases, fatal.
As a large outbreak of Zika continues in Central and South America, hospitals should be prepared for excess GBS cases, the authors of the study say, and assure adequate intensive-care capacity to treat them. Based on the 66% attack rate of Zika during the French Polynesia outbreak, investigators estimated the incidence of GBS at 0.24 per 1,000 Zika infections, but noted that it could be different in the current outbreak.
Dr. Van-Mai Cao-Lormeau of the Unit of Emerging Infectious Diseases at Institut Louis Malardé in Papeete, French Polynesia, alongside colleagues in France and French Polynesia, used a case-control design to compare serological samples from 42 patients (74% male) diagnosed at a Tahiti hospital with GBS with samples from age-and sex-matched patients who presented at the same hospital, also during the time of the outbreak, with a nonfebrile illness (n = 98) or with acute Zika disease without neurological symptoms (n = 70).
The investigators found that all but one patient with GBS had Zika virus antibodies, and all of them had neutralizing antibodies to Zika virus. By comparison, only 56% (n = 54) of the control group admitted with nonfebrile illness had neutralizing antibodies (P less than .0001).
Also, 93% of the GBS patients had Zika virus immunoglobulin M (IgM) and 88% reported symptoms consistent with Zika infection a mean of 6 days before onset of neurological symptoms. Acute Zika infection is usually characterized by rash, fever, and conjunctivitis.
Past dengue virus infection, which had been considered a possible risk factor for Zika-mediated GBS, did not differ significantly between patients in the control groups and those with GBS.
The investigators were also able to subtype the clinical characteristics of the GBS cases as consistent with acute motor axonal neuropathy, or AMAN, phenotype. However, the antibodies typically seen associated with AMAN were not seen in these patients, leading investigators to suspect that a different biological pathway was responsible.
More than a third of the GBS patients in the study required intensive care, most of these also with respiratory support, though none died.
The government of France, the European Union, and the Wellcome Trust funded the study. The researchers declared that they had no competing interests.
Serological evidence from French Polynesia links an outbreak of Zika virus to a spike in cases of Guillain-Barré syndrome seen there in 2013-2014.
The research, published online Feb. 29 in The Lancet, is the first to use a case-control design to demonstrate that Zika, a mosquito-borne flavivirus, is associated with Guillain-Barré syndrome (Lancet. 2016 Feb 29. doi: 10.1016/S0140-6736(16)00562-6).
Guillain-Barré syndrome (GBS) is an immune-mediated flaccid paralysis that can follow viral or bacterial infections. Most patients with GBS recover with intensive care in hospitals, although the syndrome can be permanently debilitating or, in rare cases, fatal.
As a large outbreak of Zika continues in Central and South America, hospitals should be prepared for excess GBS cases, the authors of the study say, and assure adequate intensive-care capacity to treat them. Based on the 66% attack rate of Zika during the French Polynesia outbreak, investigators estimated the incidence of GBS at 0.24 per 1,000 Zika infections, but noted that it could be different in the current outbreak.
Dr. Van-Mai Cao-Lormeau of the Unit of Emerging Infectious Diseases at Institut Louis Malardé in Papeete, French Polynesia, alongside colleagues in France and French Polynesia, used a case-control design to compare serological samples from 42 patients (74% male) diagnosed at a Tahiti hospital with GBS with samples from age-and sex-matched patients who presented at the same hospital, also during the time of the outbreak, with a nonfebrile illness (n = 98) or with acute Zika disease without neurological symptoms (n = 70).
The investigators found that all but one patient with GBS had Zika virus antibodies, and all of them had neutralizing antibodies to Zika virus. By comparison, only 56% (n = 54) of the control group admitted with nonfebrile illness had neutralizing antibodies (P less than .0001).
Also, 93% of the GBS patients had Zika virus immunoglobulin M (IgM) and 88% reported symptoms consistent with Zika infection a mean of 6 days before onset of neurological symptoms. Acute Zika infection is usually characterized by rash, fever, and conjunctivitis.
Past dengue virus infection, which had been considered a possible risk factor for Zika-mediated GBS, did not differ significantly between patients in the control groups and those with GBS.
The investigators were also able to subtype the clinical characteristics of the GBS cases as consistent with acute motor axonal neuropathy, or AMAN, phenotype. However, the antibodies typically seen associated with AMAN were not seen in these patients, leading investigators to suspect that a different biological pathway was responsible.
More than a third of the GBS patients in the study required intensive care, most of these also with respiratory support, though none died.
The government of France, the European Union, and the Wellcome Trust funded the study. The researchers declared that they had no competing interests.
FROM THE LANCET
Key clinical point: Acute infection with Zika virus in French Polynesia was associated with Guillain-Barré syndrome.
Major finding: Among GBS patients admitted to hospitals during an 2013-2014 outbreak of Zika virus, nearly all had antibodies or neutralizing antibodies to Zika, vs. 56% of age and sex-matched controls (P less than .0001).
Data source: A case-cohort study comparing blood results from 42 GBS cases and two cohorts of controls, one with acute Zika infection without GBS (n = 70) and another admitted during the outbreak for other illnesses (n = 98).
Disclosures: The French government, the European Union, and the Wellcome Trust sponsored the study. Investigators disclosed no conflicts of interest.
Zika diagnostic test available from CDC to certified labs
As concern about Zika virus mounts, the Food and Drug Administration on Feb. 26 authorized use of an investigational test from the Centers for Disease Control and Prevention to detect antibodies in sera and cerebrospinal fluid.
Known as the Zika Immunoglobulin M Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA), the test is available under an FDA Emergency Use Authorization to labs certified by CDC for high-complexity tests.
If infection is suspected, “the Zika MAC-ELISA may be ordered. Please contact your state or local health department to facilitate testing,” FDA said in a fact sheet for health care providers.
“At this time, there are no FDA approved/cleared tests available that can detect Zika virus in clinical specimens in the United States. Therefore, CDC has developed this test to detect evidence of Zika virus infections.” Positive and inconclusive results must be confirmed by CDC or authorized laboratories, the FDA noted.
Testing is appropriate in patients with signs and symptoms of Zika virus infection who recently traveled to areas with active transmissions. Anti-Zika IgM can usually be detected after about 4 days of symptoms, and remains detectable for about 3 months.
Most who are infected with the Zika virus don’t know they have it. For others, symptoms set in after a few days, tend to be mild, last about a week, and can include fever, rash, joint pain, and conjunctivitis. The virus has, however, been associated in Brazil with Guillain-Barre syndrome and microcephaly, although detection in pregnant women does not necessarily mean the fetus has been harmed.
So far, there have been more than 90 confirmed cases of Zika infection in the United States, most, but not all, in people who had recently traveled to endemic areas.
The FDA did not report sensitivity and specificity figures, but it’s clear from what it did say that reliability is an issue. Closely-related flavivirus infections, such as dengue fever, can trigger false positives, and in patients vaccinated against yellow fever or Japanese encephalitis, cross-reactive antibodies can also “make it difficult to identify which flavivirus is causing the patient’s current illness,” the fact sheet noted.
False negatives should be considered when “recent exposures or clinical presentation are consistent with Zika virus infection and diagnostic tests for other causes of illness are negative. Conversely, a negative result in an asymptomatic patient with a lower likelihood of exposure (e.g., a short term traveler to an affected area) may suggest the patient is not infected,” the FDA noted.
Zika MAC-ELISA labeling, fact sheets for patients, and other materials are available on the FDA’s Emergency Use Authorizations webpage. The CDC has a Zika virus site for health care providers, as well.
As concern about Zika virus mounts, the Food and Drug Administration on Feb. 26 authorized use of an investigational test from the Centers for Disease Control and Prevention to detect antibodies in sera and cerebrospinal fluid.
Known as the Zika Immunoglobulin M Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA), the test is available under an FDA Emergency Use Authorization to labs certified by CDC for high-complexity tests.
If infection is suspected, “the Zika MAC-ELISA may be ordered. Please contact your state or local health department to facilitate testing,” FDA said in a fact sheet for health care providers.
“At this time, there are no FDA approved/cleared tests available that can detect Zika virus in clinical specimens in the United States. Therefore, CDC has developed this test to detect evidence of Zika virus infections.” Positive and inconclusive results must be confirmed by CDC or authorized laboratories, the FDA noted.
Testing is appropriate in patients with signs and symptoms of Zika virus infection who recently traveled to areas with active transmissions. Anti-Zika IgM can usually be detected after about 4 days of symptoms, and remains detectable for about 3 months.
Most who are infected with the Zika virus don’t know they have it. For others, symptoms set in after a few days, tend to be mild, last about a week, and can include fever, rash, joint pain, and conjunctivitis. The virus has, however, been associated in Brazil with Guillain-Barre syndrome and microcephaly, although detection in pregnant women does not necessarily mean the fetus has been harmed.
So far, there have been more than 90 confirmed cases of Zika infection in the United States, most, but not all, in people who had recently traveled to endemic areas.
The FDA did not report sensitivity and specificity figures, but it’s clear from what it did say that reliability is an issue. Closely-related flavivirus infections, such as dengue fever, can trigger false positives, and in patients vaccinated against yellow fever or Japanese encephalitis, cross-reactive antibodies can also “make it difficult to identify which flavivirus is causing the patient’s current illness,” the fact sheet noted.
False negatives should be considered when “recent exposures or clinical presentation are consistent with Zika virus infection and diagnostic tests for other causes of illness are negative. Conversely, a negative result in an asymptomatic patient with a lower likelihood of exposure (e.g., a short term traveler to an affected area) may suggest the patient is not infected,” the FDA noted.
Zika MAC-ELISA labeling, fact sheets for patients, and other materials are available on the FDA’s Emergency Use Authorizations webpage. The CDC has a Zika virus site for health care providers, as well.
As concern about Zika virus mounts, the Food and Drug Administration on Feb. 26 authorized use of an investigational test from the Centers for Disease Control and Prevention to detect antibodies in sera and cerebrospinal fluid.
Known as the Zika Immunoglobulin M Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA), the test is available under an FDA Emergency Use Authorization to labs certified by CDC for high-complexity tests.
If infection is suspected, “the Zika MAC-ELISA may be ordered. Please contact your state or local health department to facilitate testing,” FDA said in a fact sheet for health care providers.
“At this time, there are no FDA approved/cleared tests available that can detect Zika virus in clinical specimens in the United States. Therefore, CDC has developed this test to detect evidence of Zika virus infections.” Positive and inconclusive results must be confirmed by CDC or authorized laboratories, the FDA noted.
Testing is appropriate in patients with signs and symptoms of Zika virus infection who recently traveled to areas with active transmissions. Anti-Zika IgM can usually be detected after about 4 days of symptoms, and remains detectable for about 3 months.
Most who are infected with the Zika virus don’t know they have it. For others, symptoms set in after a few days, tend to be mild, last about a week, and can include fever, rash, joint pain, and conjunctivitis. The virus has, however, been associated in Brazil with Guillain-Barre syndrome and microcephaly, although detection in pregnant women does not necessarily mean the fetus has been harmed.
So far, there have been more than 90 confirmed cases of Zika infection in the United States, most, but not all, in people who had recently traveled to endemic areas.
The FDA did not report sensitivity and specificity figures, but it’s clear from what it did say that reliability is an issue. Closely-related flavivirus infections, such as dengue fever, can trigger false positives, and in patients vaccinated against yellow fever or Japanese encephalitis, cross-reactive antibodies can also “make it difficult to identify which flavivirus is causing the patient’s current illness,” the fact sheet noted.
False negatives should be considered when “recent exposures or clinical presentation are consistent with Zika virus infection and diagnostic tests for other causes of illness are negative. Conversely, a negative result in an asymptomatic patient with a lower likelihood of exposure (e.g., a short term traveler to an affected area) may suggest the patient is not infected,” the FDA noted.
Zika MAC-ELISA labeling, fact sheets for patients, and other materials are available on the FDA’s Emergency Use Authorizations webpage. The CDC has a Zika virus site for health care providers, as well.
CDC reports nine U.S. Zika cases among pregnant women
Officials at the Centers for Disease Control and Prevention reported that they are aware of at least nine cases of laboratory-confirmed Zika virus infection in pregnant travelers, and that in four of these cases, fetuses were either spontaneously lost or aborted.
Confirmed cases of Zika virus infection were reported among women who had traveled to one or more of the following nine areas with ongoing local transmission of Zika virus: American Samoa, Brazil, El Salvador, Guatemala, Haiti, Honduras, Mexico, Puerto Rico, and Samoa, according to the CDC’s latest Morbidity and Mortality Weekly Report (2016 Feb 26. doi: http://dx.doi.org/10.15585/mmwr.mm6508e1er).
The agency is also investigating reports of 10 other cases of pregnant women with possible Zika virus infection.
Of the nine confirmed cases, six women were infected with Zika virus in their first trimester. In two cases, the pregnancies were terminated and two of the pregnancies resulted in stillbirths. Another woman gave birth to an infant with microcephaly, and one woman is still pregnant and has not experienced any complications so far.
Two of the nine women became infected with Zika virus during their second trimesters, one of whom has delivered a healthy infant and the other of whom is still pregnant with no known complications thus far. The last of the nine women, who became infected during her third trimester, gave birth to a healthy infant with no known complications.
There were no Zika virus–related hospitalizations or deaths among the nine women with Zika virus.
“To better understand the effects of Zika virus infection during pregnancy, CDC has established the U.S. Pregnancy Registry for Zika Virus Infection,” announced Dr. Denise J. Jamieson, colead of the Pregnancy and Birth Defects Team at the CDC. “This registry will provide information about the effects of Zika virus on pregnant women and their children.”
Participation in the registry is voluntary and information will be available on the CDC website soon, Dr. Jamieson added. Until then, the CDC maintains a 24/7 consultation hotline for both pregnant women and health care providers concerned about Zika virus infections, at 1-800-CDC-INFO. Patients and providers can also email [email protected].
Additionally, CDC officials stated that they have received reports of at least 14 instances in which Zika virus may have been transmitted between individuals through sexual contact. Two of these cases are confirmed to have been transmitted to women from men who visited a Zika-endemic area, while another four are “probable cases” of sexual transmission, and another six are under investigation. Two reported cases were excluded after receiving additional information.
“Men who reside in or have traveled to an area of ongoing Zika virus transmission and have a pregnant partner should abstain from sexual activity or consistently and correctly use condoms during sex with their pregnant partner for the duration of the pregnancy,” said the CDC in a statement (MMWR. 2016 Feb 26. doi: http://dx.doi.org/10.15585/mmwr.mm6508e2er).
The World Health Organization also released a new situation report on Zika virus, microcephaly, and Guillain-Barré syndrome, saying that although Zika virus has been spreading to more geographic areas since the beginning of the crisis, cases of microcephaly and neonatal malformations are increasing only in Brazil and French Polynesia.
Additionally, the WHO released interim guidelines on psychosocial support for pregnant women who may be infected with Zika virus and families dealing with an infant born with either microcephaly or another neurologic disorder.
At a press briefing on Feb. 26, CDC Director Dr. Tom Frieden, noted that it has been 6 weeks since the CDC issued its first travel warning regarding Zika virus, and that the agency is “learning more about Zika everyday.” He added that the current state of the Zika virus outbreak is an “unprecedented situation.”
But Dr. Frieden also stressed that the exact link between Zika virus infection and microcephaly is still unknown, and it is not clear at what stage in the pregnancy Zika virus affects the fetus.
It’s also unknown whether infants born to mothers with Zika virus infection who don’t develop microcephaly will have any other health problems in the future. “Unfortunately, this is something we may not know for many years,” Dr. Frieden said.
Officials at the Centers for Disease Control and Prevention reported that they are aware of at least nine cases of laboratory-confirmed Zika virus infection in pregnant travelers, and that in four of these cases, fetuses were either spontaneously lost or aborted.
Confirmed cases of Zika virus infection were reported among women who had traveled to one or more of the following nine areas with ongoing local transmission of Zika virus: American Samoa, Brazil, El Salvador, Guatemala, Haiti, Honduras, Mexico, Puerto Rico, and Samoa, according to the CDC’s latest Morbidity and Mortality Weekly Report (2016 Feb 26. doi: http://dx.doi.org/10.15585/mmwr.mm6508e1er).
The agency is also investigating reports of 10 other cases of pregnant women with possible Zika virus infection.
Of the nine confirmed cases, six women were infected with Zika virus in their first trimester. In two cases, the pregnancies were terminated and two of the pregnancies resulted in stillbirths. Another woman gave birth to an infant with microcephaly, and one woman is still pregnant and has not experienced any complications so far.
Two of the nine women became infected with Zika virus during their second trimesters, one of whom has delivered a healthy infant and the other of whom is still pregnant with no known complications thus far. The last of the nine women, who became infected during her third trimester, gave birth to a healthy infant with no known complications.
There were no Zika virus–related hospitalizations or deaths among the nine women with Zika virus.
“To better understand the effects of Zika virus infection during pregnancy, CDC has established the U.S. Pregnancy Registry for Zika Virus Infection,” announced Dr. Denise J. Jamieson, colead of the Pregnancy and Birth Defects Team at the CDC. “This registry will provide information about the effects of Zika virus on pregnant women and their children.”
Participation in the registry is voluntary and information will be available on the CDC website soon, Dr. Jamieson added. Until then, the CDC maintains a 24/7 consultation hotline for both pregnant women and health care providers concerned about Zika virus infections, at 1-800-CDC-INFO. Patients and providers can also email [email protected].
Additionally, CDC officials stated that they have received reports of at least 14 instances in which Zika virus may have been transmitted between individuals through sexual contact. Two of these cases are confirmed to have been transmitted to women from men who visited a Zika-endemic area, while another four are “probable cases” of sexual transmission, and another six are under investigation. Two reported cases were excluded after receiving additional information.
“Men who reside in or have traveled to an area of ongoing Zika virus transmission and have a pregnant partner should abstain from sexual activity or consistently and correctly use condoms during sex with their pregnant partner for the duration of the pregnancy,” said the CDC in a statement (MMWR. 2016 Feb 26. doi: http://dx.doi.org/10.15585/mmwr.mm6508e2er).
The World Health Organization also released a new situation report on Zika virus, microcephaly, and Guillain-Barré syndrome, saying that although Zika virus has been spreading to more geographic areas since the beginning of the crisis, cases of microcephaly and neonatal malformations are increasing only in Brazil and French Polynesia.
Additionally, the WHO released interim guidelines on psychosocial support for pregnant women who may be infected with Zika virus and families dealing with an infant born with either microcephaly or another neurologic disorder.
At a press briefing on Feb. 26, CDC Director Dr. Tom Frieden, noted that it has been 6 weeks since the CDC issued its first travel warning regarding Zika virus, and that the agency is “learning more about Zika everyday.” He added that the current state of the Zika virus outbreak is an “unprecedented situation.”
But Dr. Frieden also stressed that the exact link between Zika virus infection and microcephaly is still unknown, and it is not clear at what stage in the pregnancy Zika virus affects the fetus.
It’s also unknown whether infants born to mothers with Zika virus infection who don’t develop microcephaly will have any other health problems in the future. “Unfortunately, this is something we may not know for many years,” Dr. Frieden said.
Officials at the Centers for Disease Control and Prevention reported that they are aware of at least nine cases of laboratory-confirmed Zika virus infection in pregnant travelers, and that in four of these cases, fetuses were either spontaneously lost or aborted.
Confirmed cases of Zika virus infection were reported among women who had traveled to one or more of the following nine areas with ongoing local transmission of Zika virus: American Samoa, Brazil, El Salvador, Guatemala, Haiti, Honduras, Mexico, Puerto Rico, and Samoa, according to the CDC’s latest Morbidity and Mortality Weekly Report (2016 Feb 26. doi: http://dx.doi.org/10.15585/mmwr.mm6508e1er).
The agency is also investigating reports of 10 other cases of pregnant women with possible Zika virus infection.
Of the nine confirmed cases, six women were infected with Zika virus in their first trimester. In two cases, the pregnancies were terminated and two of the pregnancies resulted in stillbirths. Another woman gave birth to an infant with microcephaly, and one woman is still pregnant and has not experienced any complications so far.
Two of the nine women became infected with Zika virus during their second trimesters, one of whom has delivered a healthy infant and the other of whom is still pregnant with no known complications thus far. The last of the nine women, who became infected during her third trimester, gave birth to a healthy infant with no known complications.
There were no Zika virus–related hospitalizations or deaths among the nine women with Zika virus.
“To better understand the effects of Zika virus infection during pregnancy, CDC has established the U.S. Pregnancy Registry for Zika Virus Infection,” announced Dr. Denise J. Jamieson, colead of the Pregnancy and Birth Defects Team at the CDC. “This registry will provide information about the effects of Zika virus on pregnant women and their children.”
Participation in the registry is voluntary and information will be available on the CDC website soon, Dr. Jamieson added. Until then, the CDC maintains a 24/7 consultation hotline for both pregnant women and health care providers concerned about Zika virus infections, at 1-800-CDC-INFO. Patients and providers can also email [email protected].
Additionally, CDC officials stated that they have received reports of at least 14 instances in which Zika virus may have been transmitted between individuals through sexual contact. Two of these cases are confirmed to have been transmitted to women from men who visited a Zika-endemic area, while another four are “probable cases” of sexual transmission, and another six are under investigation. Two reported cases were excluded after receiving additional information.
“Men who reside in or have traveled to an area of ongoing Zika virus transmission and have a pregnant partner should abstain from sexual activity or consistently and correctly use condoms during sex with their pregnant partner for the duration of the pregnancy,” said the CDC in a statement (MMWR. 2016 Feb 26. doi: http://dx.doi.org/10.15585/mmwr.mm6508e2er).
The World Health Organization also released a new situation report on Zika virus, microcephaly, and Guillain-Barré syndrome, saying that although Zika virus has been spreading to more geographic areas since the beginning of the crisis, cases of microcephaly and neonatal malformations are increasing only in Brazil and French Polynesia.
Additionally, the WHO released interim guidelines on psychosocial support for pregnant women who may be infected with Zika virus and families dealing with an infant born with either microcephaly or another neurologic disorder.
At a press briefing on Feb. 26, CDC Director Dr. Tom Frieden, noted that it has been 6 weeks since the CDC issued its first travel warning regarding Zika virus, and that the agency is “learning more about Zika everyday.” He added that the current state of the Zika virus outbreak is an “unprecedented situation.”
But Dr. Frieden also stressed that the exact link between Zika virus infection and microcephaly is still unknown, and it is not clear at what stage in the pregnancy Zika virus affects the fetus.
It’s also unknown whether infants born to mothers with Zika virus infection who don’t develop microcephaly will have any other health problems in the future. “Unfortunately, this is something we may not know for many years,” Dr. Frieden said.
FROM MMWR
Zika virus in pregnancy linked to hydrops fetalis
Zika virus infection in pregnant women may be linked to hydrops fetalis and fetal demise, according to a case report published online Feb. 25 in PLOS Neglected Tropical Diseases.
A 20-year-old pregnant woman without history or signs of Zika virus infection was referred to the Hospital Geral Roberto Santos in Salvador, Brazil, in the 18th week of gestation because of low fetal weight, but by week 26 and 30, ultrasound examinations showed microcephaly, hydranencephaly, intracranial calcifications, destructive lesions of posterior fossa, and evidence of hydrothorax, ascites, and subcutaneous edema.
Ultrasound examination at week 32 showed fetal demise, and after delivery, researchers found evidence of Zika virus in the brain and in the cerebrospinal and amniotic fluid, but not in the heart, lung, liver, eye, or placenta (PLoS Negl Trop Dis. 2016 Feb 25. doi: 10.1371/journal.pntd.0004517).
“This case report of a fetus provides additional evidence for the link between ZIKV [Zika virus] infection and microcephaly,” wrote Dr. Manoel Sarno of Hospital Geral Roberto Santos, and his coauthors. “Furthermore, it serves as an alert to clinicians that in addition to central nervous system and ophthalmological manifestations, congenital ZIKV infection may cause hydrops fetalis and fetal demise.”
The researchers reported having no financial disclosures.
Zika virus infection in pregnant women may be linked to hydrops fetalis and fetal demise, according to a case report published online Feb. 25 in PLOS Neglected Tropical Diseases.
A 20-year-old pregnant woman without history or signs of Zika virus infection was referred to the Hospital Geral Roberto Santos in Salvador, Brazil, in the 18th week of gestation because of low fetal weight, but by week 26 and 30, ultrasound examinations showed microcephaly, hydranencephaly, intracranial calcifications, destructive lesions of posterior fossa, and evidence of hydrothorax, ascites, and subcutaneous edema.
Ultrasound examination at week 32 showed fetal demise, and after delivery, researchers found evidence of Zika virus in the brain and in the cerebrospinal and amniotic fluid, but not in the heart, lung, liver, eye, or placenta (PLoS Negl Trop Dis. 2016 Feb 25. doi: 10.1371/journal.pntd.0004517).
“This case report of a fetus provides additional evidence for the link between ZIKV [Zika virus] infection and microcephaly,” wrote Dr. Manoel Sarno of Hospital Geral Roberto Santos, and his coauthors. “Furthermore, it serves as an alert to clinicians that in addition to central nervous system and ophthalmological manifestations, congenital ZIKV infection may cause hydrops fetalis and fetal demise.”
The researchers reported having no financial disclosures.
Zika virus infection in pregnant women may be linked to hydrops fetalis and fetal demise, according to a case report published online Feb. 25 in PLOS Neglected Tropical Diseases.
A 20-year-old pregnant woman without history or signs of Zika virus infection was referred to the Hospital Geral Roberto Santos in Salvador, Brazil, in the 18th week of gestation because of low fetal weight, but by week 26 and 30, ultrasound examinations showed microcephaly, hydranencephaly, intracranial calcifications, destructive lesions of posterior fossa, and evidence of hydrothorax, ascites, and subcutaneous edema.
Ultrasound examination at week 32 showed fetal demise, and after delivery, researchers found evidence of Zika virus in the brain and in the cerebrospinal and amniotic fluid, but not in the heart, lung, liver, eye, or placenta (PLoS Negl Trop Dis. 2016 Feb 25. doi: 10.1371/journal.pntd.0004517).
“This case report of a fetus provides additional evidence for the link between ZIKV [Zika virus] infection and microcephaly,” wrote Dr. Manoel Sarno of Hospital Geral Roberto Santos, and his coauthors. “Furthermore, it serves as an alert to clinicians that in addition to central nervous system and ophthalmological manifestations, congenital ZIKV infection may cause hydrops fetalis and fetal demise.”
The researchers reported having no financial disclosures.
FROM PLOS NEGLECTED TROPICAL DISEASES
Key clinical point: Zika virus infection in pregnant women may be associated with hydrops fetalis and fetal demise.
Major finding: Congenital Zika virus infection was associated with microcephaly, hydranencephaly, and fetal demise in a single case.
Data source: Case report of a fetus with congenital Zika virus infection.
Disclosures: The researchers reported having no financial disclosures.
Ebola’s effects linger long after disease abates
BOSTON – A majority of Ebola virus survivors continue to have significant physical, psychological, and sociological consequences for an extended period after discharge, investigators report.
Among 417 Ebola virus survivors enrolled in a follow-up study in Guinea, about 80% had residual clinical signs up to 9 months after discharge, reported Dr. Eric Delaporte of the French National Institute of Health and Medical Research (INSERM) and Montpellier (France) University.
Results of a second, small study, also from Guinea, suggest that up to 10% of men infected with Ebola have semen that remains positive for the virus for nearly a year after disease onset, said Dr. Daouda Sissoko of INSERM in Bordeaux, France.
“The results describe a post-Ebola syndrome with frequent clinical symptoms long after discharge, the gravity of ocular complications, long-term RNA positivity in semen, and the frequency of psychological consequences of the disease,” Dr. Delaporte said at a media briefing prior to presentation of the data in an oral session at the 2016 Conference on Retroviruses and Opportunistic Infections.
Dr. Delaporte is a coinvestigator in the Postebogui Cohort Study looking at Ebola survivors in Conakry and Macenta, Guinea. The goals of the study are to provide follow-up care for survivors and to describe the clinical, biologic, virologic, immunologic, and psychosocial consequences of infection with Ebola virus.
At CROI 2016, investigators reported on the first 417 patients enrolled, including 60 children. The median age was 28 years.
The investigators found that 46% of patients reported joint pains or other rheumatologic signs, 32% had neuropsychiatric signs, 29% reported chronic headache, 22% reported fatigue, and 16% had ophthalmologic signs,
Ophthalmic slit-lamp examinations in 160 patients showed 24 cases of uveitis, 4 cases of episcleritis, 2 cases of keratitis, and 2 cases of blindness in children due to inflammatory cataracts.
Of 160 semen samples tested, 28% were positive for Ebola RNA up to 9 months after disease onset. By 1 year, however, all semen samples tested negative, Dr. Delaporte said.
The investigators also found that among 131 patients discharged from an Ebola treatment center in the Guinean capital of Conakry and followed for a mean of 5 months, 20% had a score on the Center for Epidemiologic Studies depression scale (CES-D) that indicated the patients could benefit from psychological or psychiatric interventions.
Viral reservoir in semen?
In the second study, Dr. Sissoko and colleagues enrolled 26 men who had been discharged from three Ebola treatment units in coastal regions of Guinea from February through June, 2015. The investigators obtained semen specimens every 3-6 weeks until two consecutive samples were negative for Ebola virus on reverse transcriptase polymerase chain reaction testing (RT-PCR).
In all, 19 of the men had semen positive for Ebola RNA at baseline. At a median of 250 days of follow-up 25% of patients continued to have semen positive for Ebola.
The investigators used linear mixed modeling to estimate that at 11 months, approximately 10% of patients will continue to shed Ebola virus in semen, Dr. Sissoko said.
The investigators recommend testing semen samples from each man who has recovered from an Ebola infection until the semen test negative on two consecutive samples.
Both studies were supported by INSERM. Dr. Delaporte and Dr Sissoko reported having no conflicts of interest.
BOSTON – A majority of Ebola virus survivors continue to have significant physical, psychological, and sociological consequences for an extended period after discharge, investigators report.
Among 417 Ebola virus survivors enrolled in a follow-up study in Guinea, about 80% had residual clinical signs up to 9 months after discharge, reported Dr. Eric Delaporte of the French National Institute of Health and Medical Research (INSERM) and Montpellier (France) University.
Results of a second, small study, also from Guinea, suggest that up to 10% of men infected with Ebola have semen that remains positive for the virus for nearly a year after disease onset, said Dr. Daouda Sissoko of INSERM in Bordeaux, France.
“The results describe a post-Ebola syndrome with frequent clinical symptoms long after discharge, the gravity of ocular complications, long-term RNA positivity in semen, and the frequency of psychological consequences of the disease,” Dr. Delaporte said at a media briefing prior to presentation of the data in an oral session at the 2016 Conference on Retroviruses and Opportunistic Infections.
Dr. Delaporte is a coinvestigator in the Postebogui Cohort Study looking at Ebola survivors in Conakry and Macenta, Guinea. The goals of the study are to provide follow-up care for survivors and to describe the clinical, biologic, virologic, immunologic, and psychosocial consequences of infection with Ebola virus.
At CROI 2016, investigators reported on the first 417 patients enrolled, including 60 children. The median age was 28 years.
The investigators found that 46% of patients reported joint pains or other rheumatologic signs, 32% had neuropsychiatric signs, 29% reported chronic headache, 22% reported fatigue, and 16% had ophthalmologic signs,
Ophthalmic slit-lamp examinations in 160 patients showed 24 cases of uveitis, 4 cases of episcleritis, 2 cases of keratitis, and 2 cases of blindness in children due to inflammatory cataracts.
Of 160 semen samples tested, 28% were positive for Ebola RNA up to 9 months after disease onset. By 1 year, however, all semen samples tested negative, Dr. Delaporte said.
The investigators also found that among 131 patients discharged from an Ebola treatment center in the Guinean capital of Conakry and followed for a mean of 5 months, 20% had a score on the Center for Epidemiologic Studies depression scale (CES-D) that indicated the patients could benefit from psychological or psychiatric interventions.
Viral reservoir in semen?
In the second study, Dr. Sissoko and colleagues enrolled 26 men who had been discharged from three Ebola treatment units in coastal regions of Guinea from February through June, 2015. The investigators obtained semen specimens every 3-6 weeks until two consecutive samples were negative for Ebola virus on reverse transcriptase polymerase chain reaction testing (RT-PCR).
In all, 19 of the men had semen positive for Ebola RNA at baseline. At a median of 250 days of follow-up 25% of patients continued to have semen positive for Ebola.
The investigators used linear mixed modeling to estimate that at 11 months, approximately 10% of patients will continue to shed Ebola virus in semen, Dr. Sissoko said.
The investigators recommend testing semen samples from each man who has recovered from an Ebola infection until the semen test negative on two consecutive samples.
Both studies were supported by INSERM. Dr. Delaporte and Dr Sissoko reported having no conflicts of interest.
BOSTON – A majority of Ebola virus survivors continue to have significant physical, psychological, and sociological consequences for an extended period after discharge, investigators report.
Among 417 Ebola virus survivors enrolled in a follow-up study in Guinea, about 80% had residual clinical signs up to 9 months after discharge, reported Dr. Eric Delaporte of the French National Institute of Health and Medical Research (INSERM) and Montpellier (France) University.
Results of a second, small study, also from Guinea, suggest that up to 10% of men infected with Ebola have semen that remains positive for the virus for nearly a year after disease onset, said Dr. Daouda Sissoko of INSERM in Bordeaux, France.
“The results describe a post-Ebola syndrome with frequent clinical symptoms long after discharge, the gravity of ocular complications, long-term RNA positivity in semen, and the frequency of psychological consequences of the disease,” Dr. Delaporte said at a media briefing prior to presentation of the data in an oral session at the 2016 Conference on Retroviruses and Opportunistic Infections.
Dr. Delaporte is a coinvestigator in the Postebogui Cohort Study looking at Ebola survivors in Conakry and Macenta, Guinea. The goals of the study are to provide follow-up care for survivors and to describe the clinical, biologic, virologic, immunologic, and psychosocial consequences of infection with Ebola virus.
At CROI 2016, investigators reported on the first 417 patients enrolled, including 60 children. The median age was 28 years.
The investigators found that 46% of patients reported joint pains or other rheumatologic signs, 32% had neuropsychiatric signs, 29% reported chronic headache, 22% reported fatigue, and 16% had ophthalmologic signs,
Ophthalmic slit-lamp examinations in 160 patients showed 24 cases of uveitis, 4 cases of episcleritis, 2 cases of keratitis, and 2 cases of blindness in children due to inflammatory cataracts.
Of 160 semen samples tested, 28% were positive for Ebola RNA up to 9 months after disease onset. By 1 year, however, all semen samples tested negative, Dr. Delaporte said.
The investigators also found that among 131 patients discharged from an Ebola treatment center in the Guinean capital of Conakry and followed for a mean of 5 months, 20% had a score on the Center for Epidemiologic Studies depression scale (CES-D) that indicated the patients could benefit from psychological or psychiatric interventions.
Viral reservoir in semen?
In the second study, Dr. Sissoko and colleagues enrolled 26 men who had been discharged from three Ebola treatment units in coastal regions of Guinea from February through June, 2015. The investigators obtained semen specimens every 3-6 weeks until two consecutive samples were negative for Ebola virus on reverse transcriptase polymerase chain reaction testing (RT-PCR).
In all, 19 of the men had semen positive for Ebola RNA at baseline. At a median of 250 days of follow-up 25% of patients continued to have semen positive for Ebola.
The investigators used linear mixed modeling to estimate that at 11 months, approximately 10% of patients will continue to shed Ebola virus in semen, Dr. Sissoko said.
The investigators recommend testing semen samples from each man who has recovered from an Ebola infection until the semen test negative on two consecutive samples.
Both studies were supported by INSERM. Dr. Delaporte and Dr Sissoko reported having no conflicts of interest.
AT CROI 2016
Key clinical point: Ebola virus infection has significant physical and psychological sequelae that may require long-term follow-up and patient support.
Major finding: Ebola virus can be shed in semen of survivors for at least 11 months following disease onset.
Data source: Two cohort studies of Ebola virus survivors in Guinea.
Disclosures: Both studies were supported by INSERM. Dr. Delaporte and Dr. Sissoko reported having no conflicts of interest.
CDC investigating 14 suspected Zika virus cases
Fourteen new reports of possible sexual transmission of Zika virus are being investigated by health officials in the United States, including several that involve pregnant women.
According to a media statement released by the Centers for Disease Control and Prevention on Feb. 23, in 2 of the 14 suspected cases, Zika virus infection “has been confirmed in women whose only known risk factor was sexual contact with an ill male partner who had recently traveled to an area with local Zika virus transmission; testing for the male partners is still pending.”
Confirmatory tests are pending for four additional suspected sexual transmission events, and the investigation is ongoing for an additional eight suspected cases.
“Like previously reported cases of sexual transmission, these cases involve possible transmission of the virus from men to their sex partners,” the statement reads. “At this time, there is no evidence that women can transmit Zika virus to their sex partners; however, more research is needed to understand this issue.”
In a separate announcement on Feb. 23, the CDC added Trinidad and Tobago and the Marshall Islands to the Zika virus travel notices. A full list of affected countries regions can be found at http://wwwnc.cdc.gov/travel/page/zika-information.
Fourteen new reports of possible sexual transmission of Zika virus are being investigated by health officials in the United States, including several that involve pregnant women.
According to a media statement released by the Centers for Disease Control and Prevention on Feb. 23, in 2 of the 14 suspected cases, Zika virus infection “has been confirmed in women whose only known risk factor was sexual contact with an ill male partner who had recently traveled to an area with local Zika virus transmission; testing for the male partners is still pending.”
Confirmatory tests are pending for four additional suspected sexual transmission events, and the investigation is ongoing for an additional eight suspected cases.
“Like previously reported cases of sexual transmission, these cases involve possible transmission of the virus from men to their sex partners,” the statement reads. “At this time, there is no evidence that women can transmit Zika virus to their sex partners; however, more research is needed to understand this issue.”
In a separate announcement on Feb. 23, the CDC added Trinidad and Tobago and the Marshall Islands to the Zika virus travel notices. A full list of affected countries regions can be found at http://wwwnc.cdc.gov/travel/page/zika-information.
Fourteen new reports of possible sexual transmission of Zika virus are being investigated by health officials in the United States, including several that involve pregnant women.
According to a media statement released by the Centers for Disease Control and Prevention on Feb. 23, in 2 of the 14 suspected cases, Zika virus infection “has been confirmed in women whose only known risk factor was sexual contact with an ill male partner who had recently traveled to an area with local Zika virus transmission; testing for the male partners is still pending.”
Confirmatory tests are pending for four additional suspected sexual transmission events, and the investigation is ongoing for an additional eight suspected cases.
“Like previously reported cases of sexual transmission, these cases involve possible transmission of the virus from men to their sex partners,” the statement reads. “At this time, there is no evidence that women can transmit Zika virus to their sex partners; however, more research is needed to understand this issue.”
In a separate announcement on Feb. 23, the CDC added Trinidad and Tobago and the Marshall Islands to the Zika virus travel notices. A full list of affected countries regions can be found at http://wwwnc.cdc.gov/travel/page/zika-information.
Zika virus a great unknown, but let’s stick to the facts
Much has rightly been written about the Zika virus in the last few months, yet still we see a number of misunderstandings, rumors, and fears about potential risk, being propagated widely through the media.
Each day brings new developments and expert opinion on the subject, but also, more speculation and subterfuge, with prophylactic advice varying from travel bans, mosquito repellents, contraception, and even abstention from pregnancy.
A crucial factor amplifying this trend, and perhaps the elephant in the room, is the impending 2016 Rio Summer Olympic games. With the world’s eye soon to be focused on Brazil, there is clearly a demand from governments, companies, and other interested parties to ensure they safeguard their respective populations as best they can. Unfortunately, this also means we have seen a number of them act too hastily in their assessment of the risks presented by the Zika virus.
So, what are the risks? Well, we know for sure that there is no vaccine, and it is unlikely we will see one developed this year. But the risk to adults and healthy individuals is still relatively small, as symptoms of Zika virus infection are comparable with the common cold – and certainly not life threatening or even highly infectious in the way prior tropical diseases from Africa, like Ebola, have been. In fact, Zika remained an “unsexy” and largely forgotten virus until we saw the recent probable linkages with microcephaly.
The greatest risk currently recognized is clearly to women who are pregnant or are planning to be pregnant in the near future, and at this point, we believe the risk is probably greatest in the first trimester. This is obviously a major challenge for the domestic populations of Latin America, especially when it is at present unclear whether there is any risk in asymptomatic cases of Zika. From the information we have, and until a fuller understanding of the virus risk is known, it appears that the strongest link of microcephaly presents in cases in which patients have shown symptoms; however, 25% of the mothers of babies with microcephaly remain without any symptoms. And this is, of course, a situation made even more complex by the religious elements of society in South America, where contraception remains enshrouded in cultural stigma and abortion is illegal in many countries – even in those with cases of known prebirth microcephaly.
With no vaccine on the horizon and no cure or effective treatment once infected, clearly, the only viable preventive technique is to cut the spread of the virus. In this case, we must look to reduce the vectors of this disease – namely the Aedes aegypti mosquito. This particular type of Aedes mosquito is prominent throughout tropical regions – namely much of Latin America, South Asia, around the Gulf coast, and in isolated pockets near the Black Sea in Europe. In terms of the virus and the regions in which it has been found, most cases appear in the Latin American mainland, the Caribbean, and Cape Verde – although it may be just a matter of time before it spreads into the Aedes aegypti mosquito’s other habitats within Africa and even Asia. And, of course, it is essential we continue to investigate how viable a vector other species of the Aedes mosquito may be.
Despite these concerns, the risk in terms of the 2016 Olympics, especially for traveling populations, remains low – with the notable exception of pregnant women. In fact, the U.S. Centers for Disease Control and Prevention recommends that any travelers who are pregnant (at any stage/trimester) or planning to become pregnant, should avoid traveling to areas with Zika virus outbreaks. If they cannot avoid travel or if people live in areas where Zika virus transmission is known to occur, meticulous efforts to avoid mosquito bites during both daytime and nighttime hours must be adhered to. While it may sound a rather low-tech solution for modern health care, pregnant women in Zika-affected areas should wear protective clothing, apply a U.S. Environmental Protection Agency–approved insect repellent, and sleep in a screened room or under a mosquito net. However, it is also important to note that the Aedes aegypti mosquitoes predominantly bite during the day, especially around dawn and dusk, and therefore the correct timing and use of mosquito repellents and other personal protection measures are essential.
One final important point to emphasize is that contraception for travelers during the Rio Olympics, and when they return home, is another area of vigilance. By this, we mean both men and women, as evidence suggests Zika may be able to survive in semen for up to 1 month after infection – some reports even suggest cases of Zika remaining in semen for several months. However, the advice remains the same, if you were symptomless, then 1 month of condom use after your return will be enough to mitigate the risk of infection to a sexual partner, and 6 months after return for those who have symptoms.
The obvious unknown is the symptomless cases: Can the virus remain transmissible after the return of an athlete or visitor to the games? This is a crucial point and helps explain where there are still a number of misunderstandings. While in Brazil’s population there are some symptomless cases, people from abroad with no prior exposure to Zika virus (and therefore no resistance), would certainly have some symptoms. This remains true for both North Americans and Europeans, and so they are at no risk of further spreading the virus 1 month after their return from Brazil – should they not present any symptoms.
In one recent case of overkill, Kenya threatened to boycott the Summer Games entirely on safety grounds, a move clearly based on no identifiable scientific evidence, as the risk for athletes alone remains very low and fundamentally manageable. Conversely, the other controversial advice I have recently seen stems from the World Health Organization itself – which suggested women in Latin America should not put off pregnancy for fear of Zika. But until more information is known, and the RNA is properly analyzed or a vaccine becomes available, this is not a position I can yet support. We need to fully understand the risks of Zika virus infection, and there is still a long way to go.
Prof. Dr. Eskild Petersen is a member of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) study group for Infections in Travellers and Migrants (ESGITM) as well as a professor of tropical medicine at Aarhus University Hospital, Denmark, and senior consultant of infectious diseases at the Royal Hospital, Muscat, Oman. He has undertaken work on infectious diseases, clinical immunology, and tropical and travel medicine. His recent research focuses on the rapid spread of Zika virus in the Americas and the implications for the 2016 Rio Olympic Games.
Much has rightly been written about the Zika virus in the last few months, yet still we see a number of misunderstandings, rumors, and fears about potential risk, being propagated widely through the media.
Each day brings new developments and expert opinion on the subject, but also, more speculation and subterfuge, with prophylactic advice varying from travel bans, mosquito repellents, contraception, and even abstention from pregnancy.
A crucial factor amplifying this trend, and perhaps the elephant in the room, is the impending 2016 Rio Summer Olympic games. With the world’s eye soon to be focused on Brazil, there is clearly a demand from governments, companies, and other interested parties to ensure they safeguard their respective populations as best they can. Unfortunately, this also means we have seen a number of them act too hastily in their assessment of the risks presented by the Zika virus.
So, what are the risks? Well, we know for sure that there is no vaccine, and it is unlikely we will see one developed this year. But the risk to adults and healthy individuals is still relatively small, as symptoms of Zika virus infection are comparable with the common cold – and certainly not life threatening or even highly infectious in the way prior tropical diseases from Africa, like Ebola, have been. In fact, Zika remained an “unsexy” and largely forgotten virus until we saw the recent probable linkages with microcephaly.
The greatest risk currently recognized is clearly to women who are pregnant or are planning to be pregnant in the near future, and at this point, we believe the risk is probably greatest in the first trimester. This is obviously a major challenge for the domestic populations of Latin America, especially when it is at present unclear whether there is any risk in asymptomatic cases of Zika. From the information we have, and until a fuller understanding of the virus risk is known, it appears that the strongest link of microcephaly presents in cases in which patients have shown symptoms; however, 25% of the mothers of babies with microcephaly remain without any symptoms. And this is, of course, a situation made even more complex by the religious elements of society in South America, where contraception remains enshrouded in cultural stigma and abortion is illegal in many countries – even in those with cases of known prebirth microcephaly.
With no vaccine on the horizon and no cure or effective treatment once infected, clearly, the only viable preventive technique is to cut the spread of the virus. In this case, we must look to reduce the vectors of this disease – namely the Aedes aegypti mosquito. This particular type of Aedes mosquito is prominent throughout tropical regions – namely much of Latin America, South Asia, around the Gulf coast, and in isolated pockets near the Black Sea in Europe. In terms of the virus and the regions in which it has been found, most cases appear in the Latin American mainland, the Caribbean, and Cape Verde – although it may be just a matter of time before it spreads into the Aedes aegypti mosquito’s other habitats within Africa and even Asia. And, of course, it is essential we continue to investigate how viable a vector other species of the Aedes mosquito may be.
Despite these concerns, the risk in terms of the 2016 Olympics, especially for traveling populations, remains low – with the notable exception of pregnant women. In fact, the U.S. Centers for Disease Control and Prevention recommends that any travelers who are pregnant (at any stage/trimester) or planning to become pregnant, should avoid traveling to areas with Zika virus outbreaks. If they cannot avoid travel or if people live in areas where Zika virus transmission is known to occur, meticulous efforts to avoid mosquito bites during both daytime and nighttime hours must be adhered to. While it may sound a rather low-tech solution for modern health care, pregnant women in Zika-affected areas should wear protective clothing, apply a U.S. Environmental Protection Agency–approved insect repellent, and sleep in a screened room or under a mosquito net. However, it is also important to note that the Aedes aegypti mosquitoes predominantly bite during the day, especially around dawn and dusk, and therefore the correct timing and use of mosquito repellents and other personal protection measures are essential.
One final important point to emphasize is that contraception for travelers during the Rio Olympics, and when they return home, is another area of vigilance. By this, we mean both men and women, as evidence suggests Zika may be able to survive in semen for up to 1 month after infection – some reports even suggest cases of Zika remaining in semen for several months. However, the advice remains the same, if you were symptomless, then 1 month of condom use after your return will be enough to mitigate the risk of infection to a sexual partner, and 6 months after return for those who have symptoms.
The obvious unknown is the symptomless cases: Can the virus remain transmissible after the return of an athlete or visitor to the games? This is a crucial point and helps explain where there are still a number of misunderstandings. While in Brazil’s population there are some symptomless cases, people from abroad with no prior exposure to Zika virus (and therefore no resistance), would certainly have some symptoms. This remains true for both North Americans and Europeans, and so they are at no risk of further spreading the virus 1 month after their return from Brazil – should they not present any symptoms.
In one recent case of overkill, Kenya threatened to boycott the Summer Games entirely on safety grounds, a move clearly based on no identifiable scientific evidence, as the risk for athletes alone remains very low and fundamentally manageable. Conversely, the other controversial advice I have recently seen stems from the World Health Organization itself – which suggested women in Latin America should not put off pregnancy for fear of Zika. But until more information is known, and the RNA is properly analyzed or a vaccine becomes available, this is not a position I can yet support. We need to fully understand the risks of Zika virus infection, and there is still a long way to go.
Prof. Dr. Eskild Petersen is a member of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) study group for Infections in Travellers and Migrants (ESGITM) as well as a professor of tropical medicine at Aarhus University Hospital, Denmark, and senior consultant of infectious diseases at the Royal Hospital, Muscat, Oman. He has undertaken work on infectious diseases, clinical immunology, and tropical and travel medicine. His recent research focuses on the rapid spread of Zika virus in the Americas and the implications for the 2016 Rio Olympic Games.
Much has rightly been written about the Zika virus in the last few months, yet still we see a number of misunderstandings, rumors, and fears about potential risk, being propagated widely through the media.
Each day brings new developments and expert opinion on the subject, but also, more speculation and subterfuge, with prophylactic advice varying from travel bans, mosquito repellents, contraception, and even abstention from pregnancy.
A crucial factor amplifying this trend, and perhaps the elephant in the room, is the impending 2016 Rio Summer Olympic games. With the world’s eye soon to be focused on Brazil, there is clearly a demand from governments, companies, and other interested parties to ensure they safeguard their respective populations as best they can. Unfortunately, this also means we have seen a number of them act too hastily in their assessment of the risks presented by the Zika virus.
So, what are the risks? Well, we know for sure that there is no vaccine, and it is unlikely we will see one developed this year. But the risk to adults and healthy individuals is still relatively small, as symptoms of Zika virus infection are comparable with the common cold – and certainly not life threatening or even highly infectious in the way prior tropical diseases from Africa, like Ebola, have been. In fact, Zika remained an “unsexy” and largely forgotten virus until we saw the recent probable linkages with microcephaly.
The greatest risk currently recognized is clearly to women who are pregnant or are planning to be pregnant in the near future, and at this point, we believe the risk is probably greatest in the first trimester. This is obviously a major challenge for the domestic populations of Latin America, especially when it is at present unclear whether there is any risk in asymptomatic cases of Zika. From the information we have, and until a fuller understanding of the virus risk is known, it appears that the strongest link of microcephaly presents in cases in which patients have shown symptoms; however, 25% of the mothers of babies with microcephaly remain without any symptoms. And this is, of course, a situation made even more complex by the religious elements of society in South America, where contraception remains enshrouded in cultural stigma and abortion is illegal in many countries – even in those with cases of known prebirth microcephaly.
With no vaccine on the horizon and no cure or effective treatment once infected, clearly, the only viable preventive technique is to cut the spread of the virus. In this case, we must look to reduce the vectors of this disease – namely the Aedes aegypti mosquito. This particular type of Aedes mosquito is prominent throughout tropical regions – namely much of Latin America, South Asia, around the Gulf coast, and in isolated pockets near the Black Sea in Europe. In terms of the virus and the regions in which it has been found, most cases appear in the Latin American mainland, the Caribbean, and Cape Verde – although it may be just a matter of time before it spreads into the Aedes aegypti mosquito’s other habitats within Africa and even Asia. And, of course, it is essential we continue to investigate how viable a vector other species of the Aedes mosquito may be.
Despite these concerns, the risk in terms of the 2016 Olympics, especially for traveling populations, remains low – with the notable exception of pregnant women. In fact, the U.S. Centers for Disease Control and Prevention recommends that any travelers who are pregnant (at any stage/trimester) or planning to become pregnant, should avoid traveling to areas with Zika virus outbreaks. If they cannot avoid travel or if people live in areas where Zika virus transmission is known to occur, meticulous efforts to avoid mosquito bites during both daytime and nighttime hours must be adhered to. While it may sound a rather low-tech solution for modern health care, pregnant women in Zika-affected areas should wear protective clothing, apply a U.S. Environmental Protection Agency–approved insect repellent, and sleep in a screened room or under a mosquito net. However, it is also important to note that the Aedes aegypti mosquitoes predominantly bite during the day, especially around dawn and dusk, and therefore the correct timing and use of mosquito repellents and other personal protection measures are essential.
One final important point to emphasize is that contraception for travelers during the Rio Olympics, and when they return home, is another area of vigilance. By this, we mean both men and women, as evidence suggests Zika may be able to survive in semen for up to 1 month after infection – some reports even suggest cases of Zika remaining in semen for several months. However, the advice remains the same, if you were symptomless, then 1 month of condom use after your return will be enough to mitigate the risk of infection to a sexual partner, and 6 months after return for those who have symptoms.
The obvious unknown is the symptomless cases: Can the virus remain transmissible after the return of an athlete or visitor to the games? This is a crucial point and helps explain where there are still a number of misunderstandings. While in Brazil’s population there are some symptomless cases, people from abroad with no prior exposure to Zika virus (and therefore no resistance), would certainly have some symptoms. This remains true for both North Americans and Europeans, and so they are at no risk of further spreading the virus 1 month after their return from Brazil – should they not present any symptoms.
In one recent case of overkill, Kenya threatened to boycott the Summer Games entirely on safety grounds, a move clearly based on no identifiable scientific evidence, as the risk for athletes alone remains very low and fundamentally manageable. Conversely, the other controversial advice I have recently seen stems from the World Health Organization itself – which suggested women in Latin America should not put off pregnancy for fear of Zika. But until more information is known, and the RNA is properly analyzed or a vaccine becomes available, this is not a position I can yet support. We need to fully understand the risks of Zika virus infection, and there is still a long way to go.
Prof. Dr. Eskild Petersen is a member of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) study group for Infections in Travellers and Migrants (ESGITM) as well as a professor of tropical medicine at Aarhus University Hospital, Denmark, and senior consultant of infectious diseases at the Royal Hospital, Muscat, Oman. He has undertaken work on infectious diseases, clinical immunology, and tropical and travel medicine. His recent research focuses on the rapid spread of Zika virus in the Americas and the implications for the 2016 Rio Olympic Games.
CDC updates Zika treatment guidelines for infants, children
The Centers for Disease Control and Prevention has updated its interim guidelines on treatment of infants born to mothers who may have been exposed to Zika virus during pregnancy, adding new protocols and expanding the scope of its guidance to include all children under age 18 years.
The updated guidelines, which were released Feb. 19, call for “routine care” for infants whose mothers traveled to or resided in areas with Zika virus transmission during pregnancy but did not receive Zika virus testing, provided that the infant has a normal head circumference, normal prenatal and postnatal ultrasounds, and a normal physical examination (Morb Mortal Wkly Rep. 2016;65[early release]:1-6).
If an infant’s mother has traveled to a Zika-endemic area, the first step is screening the infant for microcephaly or intracranial calcifications, either prenatally or at birth. If such defects are found, a complete physical examination and Zika virus tests should be performed on the infant.
If tests are positive or inconclusive for Zika virus infection, an additional clinical evaluation is warranted, as well as an assessment for possible long-term sequelae.
Even if no microcephaly or intracranial calcification is detected in the infant, the guidelines state that the mother should still undergo testing for Zika virus infection. If test results are negative, then “routine care” of the infant can proceed; however, positive or inconclusive results should be followed by a physical examination of the infant.
For laboratory testing of congenital Zika virus infection, infant serum should be tested for Zika virus RNA, immunoglobulin M (IgM), dengue virus IgM, and related neutralizing antibodies. If testing on a placenta or umbilical cord sample, use Zika virus immunohistochemical staining, while reverse transcription–polymerase chain reaction (RT-PCR) should be used on fixed or frozen tissue.
Acute Zika virus infections should be tested via RT-PCR for Zika virus RNA of either the serum or cerebrospinal fluid in children who have been symptomatic for less than 7 days. If symptoms have been present for 4 or more days, but Zika virus RNA has not been detected, then the serum or cerebrospinal fluid should be tested for Zika virus IgM and dengue virus IgM, along with related neutralizing antibodies.
All children under age 18 years should be considered likely for Zika infection if they have traveled to or resided in a Zika-endemic area within the previous 2 weeks and have at least two of the following known symptoms of Zika virus infection: fever, rash, conjunctivitis, and arthralgia. The same criteria apply to infants during the first 2 weeks of life if the mother has been to an area where Zika is endemic and if the infant exhibits any two symptoms of the virus.
“Arthralgia can be difficult to detect in infants and young children, and can manifest as irritability, walking with a limp (for ambulatory children), difficulty moving or refusing to move an extremity, pain on palpation, or pain with active or passive movement of the affected joint,” according to the guidelines.
NSAIDs should not be used to treat potential Zika virus infections until dengue virus infection has been definitively ruled out as the cause of illness, because of “the potential for hemorrhagic complications of dengue fever.” Further, no child under age 6 months should be prescribed NSAIDs, and no children of any age who present with acute viral illness should be given aspirin, because of associations with Reye syndrome.
Although Zika virus RNA has been isolated in breast milk, there have been no reported cases of the virus being transmitted via breastfeeding. The CDC considers breastfeeding by mothers with Zika virus infections to be safe, saying that “the benefits of breastfeeding outweigh the theoretical risks of Zika virus transmission through breast milk.”
For infants and children, the best way to avoid Zika virus infection is to avoid getting bitten by mosquitoes. The most effective ways to prevent mosquito bites are by “using air conditioning or window and door screens when indoors, wearing long-sleeved shirts and long pants, using permethrin-treated clothing and gear, and using insect repellents.” The CDC also warns against using oil of lemon eucalyptus on children under age 3 years.
“Persons with Zika virus infection should take steps to prevent mosquito bites for at least the first week of illness to decrease the risk for human-to-mosquito-to-human transmission,” the updated guidelines state.
Information on areas where Zika virus is currently prevalent, and what precautions to take when traveling to these areas, can be found on the CDC website.
The Centers for Disease Control and Prevention has updated its interim guidelines on treatment of infants born to mothers who may have been exposed to Zika virus during pregnancy, adding new protocols and expanding the scope of its guidance to include all children under age 18 years.
The updated guidelines, which were released Feb. 19, call for “routine care” for infants whose mothers traveled to or resided in areas with Zika virus transmission during pregnancy but did not receive Zika virus testing, provided that the infant has a normal head circumference, normal prenatal and postnatal ultrasounds, and a normal physical examination (Morb Mortal Wkly Rep. 2016;65[early release]:1-6).
If an infant’s mother has traveled to a Zika-endemic area, the first step is screening the infant for microcephaly or intracranial calcifications, either prenatally or at birth. If such defects are found, a complete physical examination and Zika virus tests should be performed on the infant.
If tests are positive or inconclusive for Zika virus infection, an additional clinical evaluation is warranted, as well as an assessment for possible long-term sequelae.
Even if no microcephaly or intracranial calcification is detected in the infant, the guidelines state that the mother should still undergo testing for Zika virus infection. If test results are negative, then “routine care” of the infant can proceed; however, positive or inconclusive results should be followed by a physical examination of the infant.
For laboratory testing of congenital Zika virus infection, infant serum should be tested for Zika virus RNA, immunoglobulin M (IgM), dengue virus IgM, and related neutralizing antibodies. If testing on a placenta or umbilical cord sample, use Zika virus immunohistochemical staining, while reverse transcription–polymerase chain reaction (RT-PCR) should be used on fixed or frozen tissue.
Acute Zika virus infections should be tested via RT-PCR for Zika virus RNA of either the serum or cerebrospinal fluid in children who have been symptomatic for less than 7 days. If symptoms have been present for 4 or more days, but Zika virus RNA has not been detected, then the serum or cerebrospinal fluid should be tested for Zika virus IgM and dengue virus IgM, along with related neutralizing antibodies.
All children under age 18 years should be considered likely for Zika infection if they have traveled to or resided in a Zika-endemic area within the previous 2 weeks and have at least two of the following known symptoms of Zika virus infection: fever, rash, conjunctivitis, and arthralgia. The same criteria apply to infants during the first 2 weeks of life if the mother has been to an area where Zika is endemic and if the infant exhibits any two symptoms of the virus.
“Arthralgia can be difficult to detect in infants and young children, and can manifest as irritability, walking with a limp (for ambulatory children), difficulty moving or refusing to move an extremity, pain on palpation, or pain with active or passive movement of the affected joint,” according to the guidelines.
NSAIDs should not be used to treat potential Zika virus infections until dengue virus infection has been definitively ruled out as the cause of illness, because of “the potential for hemorrhagic complications of dengue fever.” Further, no child under age 6 months should be prescribed NSAIDs, and no children of any age who present with acute viral illness should be given aspirin, because of associations with Reye syndrome.
Although Zika virus RNA has been isolated in breast milk, there have been no reported cases of the virus being transmitted via breastfeeding. The CDC considers breastfeeding by mothers with Zika virus infections to be safe, saying that “the benefits of breastfeeding outweigh the theoretical risks of Zika virus transmission through breast milk.”
For infants and children, the best way to avoid Zika virus infection is to avoid getting bitten by mosquitoes. The most effective ways to prevent mosquito bites are by “using air conditioning or window and door screens when indoors, wearing long-sleeved shirts and long pants, using permethrin-treated clothing and gear, and using insect repellents.” The CDC also warns against using oil of lemon eucalyptus on children under age 3 years.
“Persons with Zika virus infection should take steps to prevent mosquito bites for at least the first week of illness to decrease the risk for human-to-mosquito-to-human transmission,” the updated guidelines state.
Information on areas where Zika virus is currently prevalent, and what precautions to take when traveling to these areas, can be found on the CDC website.
The Centers for Disease Control and Prevention has updated its interim guidelines on treatment of infants born to mothers who may have been exposed to Zika virus during pregnancy, adding new protocols and expanding the scope of its guidance to include all children under age 18 years.
The updated guidelines, which were released Feb. 19, call for “routine care” for infants whose mothers traveled to or resided in areas with Zika virus transmission during pregnancy but did not receive Zika virus testing, provided that the infant has a normal head circumference, normal prenatal and postnatal ultrasounds, and a normal physical examination (Morb Mortal Wkly Rep. 2016;65[early release]:1-6).
If an infant’s mother has traveled to a Zika-endemic area, the first step is screening the infant for microcephaly or intracranial calcifications, either prenatally or at birth. If such defects are found, a complete physical examination and Zika virus tests should be performed on the infant.
If tests are positive or inconclusive for Zika virus infection, an additional clinical evaluation is warranted, as well as an assessment for possible long-term sequelae.
Even if no microcephaly or intracranial calcification is detected in the infant, the guidelines state that the mother should still undergo testing for Zika virus infection. If test results are negative, then “routine care” of the infant can proceed; however, positive or inconclusive results should be followed by a physical examination of the infant.
For laboratory testing of congenital Zika virus infection, infant serum should be tested for Zika virus RNA, immunoglobulin M (IgM), dengue virus IgM, and related neutralizing antibodies. If testing on a placenta or umbilical cord sample, use Zika virus immunohistochemical staining, while reverse transcription–polymerase chain reaction (RT-PCR) should be used on fixed or frozen tissue.
Acute Zika virus infections should be tested via RT-PCR for Zika virus RNA of either the serum or cerebrospinal fluid in children who have been symptomatic for less than 7 days. If symptoms have been present for 4 or more days, but Zika virus RNA has not been detected, then the serum or cerebrospinal fluid should be tested for Zika virus IgM and dengue virus IgM, along with related neutralizing antibodies.
All children under age 18 years should be considered likely for Zika infection if they have traveled to or resided in a Zika-endemic area within the previous 2 weeks and have at least two of the following known symptoms of Zika virus infection: fever, rash, conjunctivitis, and arthralgia. The same criteria apply to infants during the first 2 weeks of life if the mother has been to an area where Zika is endemic and if the infant exhibits any two symptoms of the virus.
“Arthralgia can be difficult to detect in infants and young children, and can manifest as irritability, walking with a limp (for ambulatory children), difficulty moving or refusing to move an extremity, pain on palpation, or pain with active or passive movement of the affected joint,” according to the guidelines.
NSAIDs should not be used to treat potential Zika virus infections until dengue virus infection has been definitively ruled out as the cause of illness, because of “the potential for hemorrhagic complications of dengue fever.” Further, no child under age 6 months should be prescribed NSAIDs, and no children of any age who present with acute viral illness should be given aspirin, because of associations with Reye syndrome.
Although Zika virus RNA has been isolated in breast milk, there have been no reported cases of the virus being transmitted via breastfeeding. The CDC considers breastfeeding by mothers with Zika virus infections to be safe, saying that “the benefits of breastfeeding outweigh the theoretical risks of Zika virus transmission through breast milk.”
For infants and children, the best way to avoid Zika virus infection is to avoid getting bitten by mosquitoes. The most effective ways to prevent mosquito bites are by “using air conditioning or window and door screens when indoors, wearing long-sleeved shirts and long pants, using permethrin-treated clothing and gear, and using insect repellents.” The CDC also warns against using oil of lemon eucalyptus on children under age 3 years.
“Persons with Zika virus infection should take steps to prevent mosquito bites for at least the first week of illness to decrease the risk for human-to-mosquito-to-human transmission,” the updated guidelines state.
Information on areas where Zika virus is currently prevalent, and what precautions to take when traveling to these areas, can be found on the CDC website.
FROM MMWR