User login
When’s the right time to use dementia as a diagnosis?
Is dementia a diagnosis?
I use it myself, although I find that some neurologists consider this blasphemy.
The problem is that there aren’t many terms to cover cognitive disorders beyond mild cognitive impairment (MCI). Phrases like “cortical degeneration” and “frontotemporal disorder” are difficult for families and patients. They aren’t medically trained and want something easy to write down.
“Alzheimer’s,” or – as one patient’s family member says, “the A-word” – is often more accurate, but has stigma attached to it that many don’t want, especially at a first visit. It also immediately conjures up feared images of nursing homes, wheelchairs, and bed-bound people.
So I use a diagnosis of dementia with many families, at least initially. Since, with occasional exceptions, we tend to perform a work-up of all cognitive disorders the same way, I don’t have a problem with using a more generic blanket term. As I sometimes try to simplify things, I’ll say, “It’s like squares and rectangles. Alzheimer’s disease is a dementia, but not all dementias are Alzheimer’s disease.”
I don’t do this to avoid confrontation, be dishonest, mislead patients and families, or avoid telling the truth. I still make it very clear that this is a progressive neurologic illness that will cause worsening cognitive problems over time. But many times families aren’t ready for “the A-word” early on, or there’s a concern the patient will harm themselves while they still have that capacity. Sometimes, it’s better to use a different phrase.
It may all be semantics, but on a personal level, a word can make a huge difference.
So I say dementia. In spite of some editorials I’ve seen saying we should retire the phrase, I argue that in many circumstances it’s still valid and useful.
It may not be a final, or even specific, diagnosis, but it is often the best and most socially acceptable one at the beginning of the doctor-patient-family relationship. When you’re trying to build rapport with them, that’s equally critical when you know what’s to come down the road.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Is dementia a diagnosis?
I use it myself, although I find that some neurologists consider this blasphemy.
The problem is that there aren’t many terms to cover cognitive disorders beyond mild cognitive impairment (MCI). Phrases like “cortical degeneration” and “frontotemporal disorder” are difficult for families and patients. They aren’t medically trained and want something easy to write down.
“Alzheimer’s,” or – as one patient’s family member says, “the A-word” – is often more accurate, but has stigma attached to it that many don’t want, especially at a first visit. It also immediately conjures up feared images of nursing homes, wheelchairs, and bed-bound people.
So I use a diagnosis of dementia with many families, at least initially. Since, with occasional exceptions, we tend to perform a work-up of all cognitive disorders the same way, I don’t have a problem with using a more generic blanket term. As I sometimes try to simplify things, I’ll say, “It’s like squares and rectangles. Alzheimer’s disease is a dementia, but not all dementias are Alzheimer’s disease.”
I don’t do this to avoid confrontation, be dishonest, mislead patients and families, or avoid telling the truth. I still make it very clear that this is a progressive neurologic illness that will cause worsening cognitive problems over time. But many times families aren’t ready for “the A-word” early on, or there’s a concern the patient will harm themselves while they still have that capacity. Sometimes, it’s better to use a different phrase.
It may all be semantics, but on a personal level, a word can make a huge difference.
So I say dementia. In spite of some editorials I’ve seen saying we should retire the phrase, I argue that in many circumstances it’s still valid and useful.
It may not be a final, or even specific, diagnosis, but it is often the best and most socially acceptable one at the beginning of the doctor-patient-family relationship. When you’re trying to build rapport with them, that’s equally critical when you know what’s to come down the road.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Is dementia a diagnosis?
I use it myself, although I find that some neurologists consider this blasphemy.
The problem is that there aren’t many terms to cover cognitive disorders beyond mild cognitive impairment (MCI). Phrases like “cortical degeneration” and “frontotemporal disorder” are difficult for families and patients. They aren’t medically trained and want something easy to write down.
“Alzheimer’s,” or – as one patient’s family member says, “the A-word” – is often more accurate, but has stigma attached to it that many don’t want, especially at a first visit. It also immediately conjures up feared images of nursing homes, wheelchairs, and bed-bound people.
So I use a diagnosis of dementia with many families, at least initially. Since, with occasional exceptions, we tend to perform a work-up of all cognitive disorders the same way, I don’t have a problem with using a more generic blanket term. As I sometimes try to simplify things, I’ll say, “It’s like squares and rectangles. Alzheimer’s disease is a dementia, but not all dementias are Alzheimer’s disease.”
I don’t do this to avoid confrontation, be dishonest, mislead patients and families, or avoid telling the truth. I still make it very clear that this is a progressive neurologic illness that will cause worsening cognitive problems over time. But many times families aren’t ready for “the A-word” early on, or there’s a concern the patient will harm themselves while they still have that capacity. Sometimes, it’s better to use a different phrase.
It may all be semantics, but on a personal level, a word can make a huge difference.
So I say dementia. In spite of some editorials I’ve seen saying we should retire the phrase, I argue that in many circumstances it’s still valid and useful.
It may not be a final, or even specific, diagnosis, but it is often the best and most socially acceptable one at the beginning of the doctor-patient-family relationship. When you’re trying to build rapport with them, that’s equally critical when you know what’s to come down the road.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Transdermal estradiol may modulate the relationship between sleep, cognition
LOS ANGELES – Estrogen therapy may have scored another goal in its comeback game, as a 7-year prospective study shows that a transdermal formulation preserves some measures of cognitive function and brain architecture in postmenopausal women.
In addition to performing better on subjective tests of memory, women using the estrogen patch experienced less cortical atrophy and were less likely to show amyloid on brain imaging. The observations were moderately associated with the improved sleep these women reported, Burcu Zeydan, MD, said at the Alzheimer’s Association International Conference.
“By 7 years, among the cognitive domains studied ... [less brain and cognitive change] correlated with lower global sleep score, meaning better sleep quality in the estradiol group,” said Dr. Zeydan, assistant professor of radiology at the Mayo Clinic in Rochester, Minn. “We previously found that preservation of dorsolateral prefrontal cortex over 7 years was associated with lower cortical beta-amyloid deposition on PET only in the estradiol group, pointing out the potential role of estrogen receptors in modulating this relationship.”
Dysregulated sleep is more common among women than men, particularly as menopause approaches and estrogen levels fluctuate, then decline, Dr. Zeydan said.
Dr. Zeydan reported the sleep substudy of KEEPS (the Kronos Early Estrogen Prevention Study), a randomized, double-blind, placebo-controlled, multisite trial that compared oral conjugated equine estrogen with transdermal estradiol. A control group received oral placebo and a placebo patch.*
Brain architecture was similar between the placebo and transdermal groups, but it was actually worse in some measures in the oral-estrogen group, compared with the placebo group. Women taking oral estrogen had more white matter hyperintensities, greater ventricle enlargement, and more cortical thinning. Those differences resolved after they stopped taking the oral formulation, bringing them into line with the transdermal and placebo groups.
The investigation also found that the transdermal group showed lower cerebral amyloid binding on PET scans relative to both placebo and oral estrogen.
“The relative preservation of dorsolateral prefrontal cortical volume in the [transdermal estradiol] group over 7 years indicates that hormone therapy may have long-term effects on the brain,” the team concluded. They noted that the original KEEPS study didn’t find any cognitive correlation with these changes.
The subanalysis looked at 69 women of the KEEPS cohort who had been followed for the full 7 years (4 years on treatment and 3 years off treatment). They were randomized to oral placebo and a placebo patch,* oral conjugated equine estrogen (0.45 mg/day), or transdermal estradiol (50 mcg/day). Participants in the active treatment groups received oral micronized progesterone 12 days each month. All had complete data on cognitive testing and brain imaging. Sleep quality was measured by the Pittsburgh Sleep Quality Index. Dr. Zeydan compared cognition and brain architecture findings in relation to the sleep score; lower scores mean better sleep.
The women were aged 42-58 years at baseline, and within 36 months from menopause. They had no history of menopausal hormone therapy or cardiovascular disease.
The investigators were particularly interested in how estrogen might have modulated the disturbed sleep patterns that often accompany perimenopause and early menopause, and whether the observed brain and cognitive changes tracked with sleep quality.
“During this time, 40% to 60% of women report problems sleeping, and estrogen decline seems to play an important role in sleep disturbances during this phase,” Dr. Zeydan said. “Although poor sleep quality is common in recently menopausal women, sleep quality improves with hormone therapy, as was previously demonstrated in KEEPS hormone therapy trial in recently menopausal women.”
By year 7, the cohort’s mean age was 61 years. The majority had at least some college education. The percentage who carried an apolipoprotein E epsilon-4 allele varied by group, with 15% positivity in the oral group, 48% in the transdermal group, and 16% in the placebo group.
Cognitive function was estimated with a global cognitive measure and four cognitive domain scores: verbal learning and memory, auditory attention and working memory, visual attention and executive function, and mental flexibility.
Higher attention and executive function scores were moderately correlated with a lower sleep score in the transdermal group (r = –0.54, a significant difference compared with the oral formulation). Lower sleep scores also showed a moderate correlation with preserved cortical volume of the dorsolateral prefrontal region (r = –0.47, also significantly different from the oral group).
Lower brain amyloid also positively correlated with better sleep. The correlation between sleep and global amyloid burden in the transdermal group was also moderate (r = 0.45), while the correlation in the oral group was significantly weaker (r = 0.18).
“We can say that sleep quality and transdermal estradiol during early postmenopausal years somehow interact to influence beta-amyloid deposition, preservation of dorsolateral prefrontal cortex volume, and attention and executive function,” Dr. Zeydan said.
Dr. Zeydan had no financial disclosures.
*Correction, 8/7/2019: An earlier version of this story did not make clear that participants in the control group received oral placebo and a placebo patch.
LOS ANGELES – Estrogen therapy may have scored another goal in its comeback game, as a 7-year prospective study shows that a transdermal formulation preserves some measures of cognitive function and brain architecture in postmenopausal women.
In addition to performing better on subjective tests of memory, women using the estrogen patch experienced less cortical atrophy and were less likely to show amyloid on brain imaging. The observations were moderately associated with the improved sleep these women reported, Burcu Zeydan, MD, said at the Alzheimer’s Association International Conference.
“By 7 years, among the cognitive domains studied ... [less brain and cognitive change] correlated with lower global sleep score, meaning better sleep quality in the estradiol group,” said Dr. Zeydan, assistant professor of radiology at the Mayo Clinic in Rochester, Minn. “We previously found that preservation of dorsolateral prefrontal cortex over 7 years was associated with lower cortical beta-amyloid deposition on PET only in the estradiol group, pointing out the potential role of estrogen receptors in modulating this relationship.”
Dysregulated sleep is more common among women than men, particularly as menopause approaches and estrogen levels fluctuate, then decline, Dr. Zeydan said.
Dr. Zeydan reported the sleep substudy of KEEPS (the Kronos Early Estrogen Prevention Study), a randomized, double-blind, placebo-controlled, multisite trial that compared oral conjugated equine estrogen with transdermal estradiol. A control group received oral placebo and a placebo patch.*
Brain architecture was similar between the placebo and transdermal groups, but it was actually worse in some measures in the oral-estrogen group, compared with the placebo group. Women taking oral estrogen had more white matter hyperintensities, greater ventricle enlargement, and more cortical thinning. Those differences resolved after they stopped taking the oral formulation, bringing them into line with the transdermal and placebo groups.
The investigation also found that the transdermal group showed lower cerebral amyloid binding on PET scans relative to both placebo and oral estrogen.
“The relative preservation of dorsolateral prefrontal cortical volume in the [transdermal estradiol] group over 7 years indicates that hormone therapy may have long-term effects on the brain,” the team concluded. They noted that the original KEEPS study didn’t find any cognitive correlation with these changes.
The subanalysis looked at 69 women of the KEEPS cohort who had been followed for the full 7 years (4 years on treatment and 3 years off treatment). They were randomized to oral placebo and a placebo patch,* oral conjugated equine estrogen (0.45 mg/day), or transdermal estradiol (50 mcg/day). Participants in the active treatment groups received oral micronized progesterone 12 days each month. All had complete data on cognitive testing and brain imaging. Sleep quality was measured by the Pittsburgh Sleep Quality Index. Dr. Zeydan compared cognition and brain architecture findings in relation to the sleep score; lower scores mean better sleep.
The women were aged 42-58 years at baseline, and within 36 months from menopause. They had no history of menopausal hormone therapy or cardiovascular disease.
The investigators were particularly interested in how estrogen might have modulated the disturbed sleep patterns that often accompany perimenopause and early menopause, and whether the observed brain and cognitive changes tracked with sleep quality.
“During this time, 40% to 60% of women report problems sleeping, and estrogen decline seems to play an important role in sleep disturbances during this phase,” Dr. Zeydan said. “Although poor sleep quality is common in recently menopausal women, sleep quality improves with hormone therapy, as was previously demonstrated in KEEPS hormone therapy trial in recently menopausal women.”
By year 7, the cohort’s mean age was 61 years. The majority had at least some college education. The percentage who carried an apolipoprotein E epsilon-4 allele varied by group, with 15% positivity in the oral group, 48% in the transdermal group, and 16% in the placebo group.
Cognitive function was estimated with a global cognitive measure and four cognitive domain scores: verbal learning and memory, auditory attention and working memory, visual attention and executive function, and mental flexibility.
Higher attention and executive function scores were moderately correlated with a lower sleep score in the transdermal group (r = –0.54, a significant difference compared with the oral formulation). Lower sleep scores also showed a moderate correlation with preserved cortical volume of the dorsolateral prefrontal region (r = –0.47, also significantly different from the oral group).
Lower brain amyloid also positively correlated with better sleep. The correlation between sleep and global amyloid burden in the transdermal group was also moderate (r = 0.45), while the correlation in the oral group was significantly weaker (r = 0.18).
“We can say that sleep quality and transdermal estradiol during early postmenopausal years somehow interact to influence beta-amyloid deposition, preservation of dorsolateral prefrontal cortex volume, and attention and executive function,” Dr. Zeydan said.
Dr. Zeydan had no financial disclosures.
*Correction, 8/7/2019: An earlier version of this story did not make clear that participants in the control group received oral placebo and a placebo patch.
LOS ANGELES – Estrogen therapy may have scored another goal in its comeback game, as a 7-year prospective study shows that a transdermal formulation preserves some measures of cognitive function and brain architecture in postmenopausal women.
In addition to performing better on subjective tests of memory, women using the estrogen patch experienced less cortical atrophy and were less likely to show amyloid on brain imaging. The observations were moderately associated with the improved sleep these women reported, Burcu Zeydan, MD, said at the Alzheimer’s Association International Conference.
“By 7 years, among the cognitive domains studied ... [less brain and cognitive change] correlated with lower global sleep score, meaning better sleep quality in the estradiol group,” said Dr. Zeydan, assistant professor of radiology at the Mayo Clinic in Rochester, Minn. “We previously found that preservation of dorsolateral prefrontal cortex over 7 years was associated with lower cortical beta-amyloid deposition on PET only in the estradiol group, pointing out the potential role of estrogen receptors in modulating this relationship.”
Dysregulated sleep is more common among women than men, particularly as menopause approaches and estrogen levels fluctuate, then decline, Dr. Zeydan said.
Dr. Zeydan reported the sleep substudy of KEEPS (the Kronos Early Estrogen Prevention Study), a randomized, double-blind, placebo-controlled, multisite trial that compared oral conjugated equine estrogen with transdermal estradiol. A control group received oral placebo and a placebo patch.*
Brain architecture was similar between the placebo and transdermal groups, but it was actually worse in some measures in the oral-estrogen group, compared with the placebo group. Women taking oral estrogen had more white matter hyperintensities, greater ventricle enlargement, and more cortical thinning. Those differences resolved after they stopped taking the oral formulation, bringing them into line with the transdermal and placebo groups.
The investigation also found that the transdermal group showed lower cerebral amyloid binding on PET scans relative to both placebo and oral estrogen.
“The relative preservation of dorsolateral prefrontal cortical volume in the [transdermal estradiol] group over 7 years indicates that hormone therapy may have long-term effects on the brain,” the team concluded. They noted that the original KEEPS study didn’t find any cognitive correlation with these changes.
The subanalysis looked at 69 women of the KEEPS cohort who had been followed for the full 7 years (4 years on treatment and 3 years off treatment). They were randomized to oral placebo and a placebo patch,* oral conjugated equine estrogen (0.45 mg/day), or transdermal estradiol (50 mcg/day). Participants in the active treatment groups received oral micronized progesterone 12 days each month. All had complete data on cognitive testing and brain imaging. Sleep quality was measured by the Pittsburgh Sleep Quality Index. Dr. Zeydan compared cognition and brain architecture findings in relation to the sleep score; lower scores mean better sleep.
The women were aged 42-58 years at baseline, and within 36 months from menopause. They had no history of menopausal hormone therapy or cardiovascular disease.
The investigators were particularly interested in how estrogen might have modulated the disturbed sleep patterns that often accompany perimenopause and early menopause, and whether the observed brain and cognitive changes tracked with sleep quality.
“During this time, 40% to 60% of women report problems sleeping, and estrogen decline seems to play an important role in sleep disturbances during this phase,” Dr. Zeydan said. “Although poor sleep quality is common in recently menopausal women, sleep quality improves with hormone therapy, as was previously demonstrated in KEEPS hormone therapy trial in recently menopausal women.”
By year 7, the cohort’s mean age was 61 years. The majority had at least some college education. The percentage who carried an apolipoprotein E epsilon-4 allele varied by group, with 15% positivity in the oral group, 48% in the transdermal group, and 16% in the placebo group.
Cognitive function was estimated with a global cognitive measure and four cognitive domain scores: verbal learning and memory, auditory attention and working memory, visual attention and executive function, and mental flexibility.
Higher attention and executive function scores were moderately correlated with a lower sleep score in the transdermal group (r = –0.54, a significant difference compared with the oral formulation). Lower sleep scores also showed a moderate correlation with preserved cortical volume of the dorsolateral prefrontal region (r = –0.47, also significantly different from the oral group).
Lower brain amyloid also positively correlated with better sleep. The correlation between sleep and global amyloid burden in the transdermal group was also moderate (r = 0.45), while the correlation in the oral group was significantly weaker (r = 0.18).
“We can say that sleep quality and transdermal estradiol during early postmenopausal years somehow interact to influence beta-amyloid deposition, preservation of dorsolateral prefrontal cortex volume, and attention and executive function,” Dr. Zeydan said.
Dr. Zeydan had no financial disclosures.
*Correction, 8/7/2019: An earlier version of this story did not make clear that participants in the control group received oral placebo and a placebo patch.
REPORTING FROM AAIC 2019
Gum bacteria and Alzheimer’s: A hypothesis inches forward
LOS ANGELES – Scientists testing an unusual hypothesis about the pathogenesis of Alzheimer’s disease say that use of an experimental antimicrobial drug to target a common oral infection was associated with biomarker improvements in people with the disease.
At the Alzheimer’s Association International Conference, Michael Detke, MD, PhD, of Cortexyme in South San Francisco, Calif., presented findings from a small cohort (n = 9) of people with mild to moderate Alzheimer’s disease (AD).
Six patients, mean age 72, were randomized to 4 weeks’ treatment with an agent called COR388, which inhibits toxic proteases produced by Porphyromonas gingivalis, a bacterium that colonizes the mouth and gums and has been found in the brains and cerebrospinal fluid of people with AD more than in non-AD patients. Another three subjects were randomized to placebo.
At 28 days, the CSF levels of an Alzheimer’s-associated apolipoprotein E (ApoE) fragment and serum levels of RANTES, a chemokine associated with chronic inflammation, were reduced from baseline by about one-third in treated subjects, compared with placebo subjects (P less than .05).
“In AD, ApoE is known to be fragmented and the fragments are known to be neurotoxic,” Dr. Detke, the lead study author, said in an interview. “We hypothesized that these P. gingivalis proteases may be acting on ApoE cleavage – so if you bind the proteases you should reduce the fragments. We saw in this study that ApoE fragment was reduced by 30%, or back to about normal levels.”
It is difficult to eradicate P. gingivalis infection using conventional antibiotics. The experimental agent COR388 “does not kill the bacteria but rather neutralizes it by binding to the proteases it produces, making the bacteria benign,” Dr. Detke said.
While hypotheses involving infectious causes of Alzheimer’s remain on the periphery of dementia research, Dr. Detke and his colleagues will soon be able to test theirs in a more meaningful way. At the conference, Dr. Detke presented detailed plans for a phase 2/3, placebo-controlled, clinical trial of COR388 in 570 patients aged 55-80 with mild to moderate AD.
Patients in the study are currently being randomized to 48 weeks of treatment with one of two doses, or placebo, with cognitive and biomarker endpoints planned, including for amyloid-beta, tau, and serum, plasma, and CSF markers of neuroinflammation.
Dr. Detke is an employee of Cortexyme, as are another 8 of the study’s 11 authors.
LOS ANGELES – Scientists testing an unusual hypothesis about the pathogenesis of Alzheimer’s disease say that use of an experimental antimicrobial drug to target a common oral infection was associated with biomarker improvements in people with the disease.
At the Alzheimer’s Association International Conference, Michael Detke, MD, PhD, of Cortexyme in South San Francisco, Calif., presented findings from a small cohort (n = 9) of people with mild to moderate Alzheimer’s disease (AD).
Six patients, mean age 72, were randomized to 4 weeks’ treatment with an agent called COR388, which inhibits toxic proteases produced by Porphyromonas gingivalis, a bacterium that colonizes the mouth and gums and has been found in the brains and cerebrospinal fluid of people with AD more than in non-AD patients. Another three subjects were randomized to placebo.
At 28 days, the CSF levels of an Alzheimer’s-associated apolipoprotein E (ApoE) fragment and serum levels of RANTES, a chemokine associated with chronic inflammation, were reduced from baseline by about one-third in treated subjects, compared with placebo subjects (P less than .05).
“In AD, ApoE is known to be fragmented and the fragments are known to be neurotoxic,” Dr. Detke, the lead study author, said in an interview. “We hypothesized that these P. gingivalis proteases may be acting on ApoE cleavage – so if you bind the proteases you should reduce the fragments. We saw in this study that ApoE fragment was reduced by 30%, or back to about normal levels.”
It is difficult to eradicate P. gingivalis infection using conventional antibiotics. The experimental agent COR388 “does not kill the bacteria but rather neutralizes it by binding to the proteases it produces, making the bacteria benign,” Dr. Detke said.
While hypotheses involving infectious causes of Alzheimer’s remain on the periphery of dementia research, Dr. Detke and his colleagues will soon be able to test theirs in a more meaningful way. At the conference, Dr. Detke presented detailed plans for a phase 2/3, placebo-controlled, clinical trial of COR388 in 570 patients aged 55-80 with mild to moderate AD.
Patients in the study are currently being randomized to 48 weeks of treatment with one of two doses, or placebo, with cognitive and biomarker endpoints planned, including for amyloid-beta, tau, and serum, plasma, and CSF markers of neuroinflammation.
Dr. Detke is an employee of Cortexyme, as are another 8 of the study’s 11 authors.
LOS ANGELES – Scientists testing an unusual hypothesis about the pathogenesis of Alzheimer’s disease say that use of an experimental antimicrobial drug to target a common oral infection was associated with biomarker improvements in people with the disease.
At the Alzheimer’s Association International Conference, Michael Detke, MD, PhD, of Cortexyme in South San Francisco, Calif., presented findings from a small cohort (n = 9) of people with mild to moderate Alzheimer’s disease (AD).
Six patients, mean age 72, were randomized to 4 weeks’ treatment with an agent called COR388, which inhibits toxic proteases produced by Porphyromonas gingivalis, a bacterium that colonizes the mouth and gums and has been found in the brains and cerebrospinal fluid of people with AD more than in non-AD patients. Another three subjects were randomized to placebo.
At 28 days, the CSF levels of an Alzheimer’s-associated apolipoprotein E (ApoE) fragment and serum levels of RANTES, a chemokine associated with chronic inflammation, were reduced from baseline by about one-third in treated subjects, compared with placebo subjects (P less than .05).
“In AD, ApoE is known to be fragmented and the fragments are known to be neurotoxic,” Dr. Detke, the lead study author, said in an interview. “We hypothesized that these P. gingivalis proteases may be acting on ApoE cleavage – so if you bind the proteases you should reduce the fragments. We saw in this study that ApoE fragment was reduced by 30%, or back to about normal levels.”
It is difficult to eradicate P. gingivalis infection using conventional antibiotics. The experimental agent COR388 “does not kill the bacteria but rather neutralizes it by binding to the proteases it produces, making the bacteria benign,” Dr. Detke said.
While hypotheses involving infectious causes of Alzheimer’s remain on the periphery of dementia research, Dr. Detke and his colleagues will soon be able to test theirs in a more meaningful way. At the conference, Dr. Detke presented detailed plans for a phase 2/3, placebo-controlled, clinical trial of COR388 in 570 patients aged 55-80 with mild to moderate AD.
Patients in the study are currently being randomized to 48 weeks of treatment with one of two doses, or placebo, with cognitive and biomarker endpoints planned, including for amyloid-beta, tau, and serum, plasma, and CSF markers of neuroinflammation.
Dr. Detke is an employee of Cortexyme, as are another 8 of the study’s 11 authors.
FROM AAIC 2019
Hypertension, white matter hyperintensities, and dementia: What’s the link?
LOS ANGELES – While uncontrolled hypertension is an established risk factor for dementias including Alzheimer’s disease, the pathways by which it might lead to dementia remain poorly understood.
In research presented at the Alzheimer’s Association International Conference, Jérémie Lespinasse, PharmD, an investigator with the University of Bordeaux and INSERM U1219 in Bordeaux, France, presented data showing that hypertension-linked increases in the brain’s load of white matter hyperintensities – an imaging biomarker linked to small-vessel disease – was associated with cognitive decline independent of amyloid cascade biomarkers.
“We know that hypertension is related to cognitive decline and dementia, including Alzheimer’s dementia,” Carole Dufouil, PhD, the study’s last author and the research director at INSERM U1219, said in an interview. “But we didn’t know the pathway. It could go through what’s more typical of vascular pathology or it could go through what’s more typical of AD – and that’s what we wanted to test.”
The researchers found that the impact of hypertension on cognition doesn’t “go through amyloid,” Dr. Dufouil said, but rather a typically vascular pathway of white matter hyperintensities and neurodegeneration. “This is a big difference from other brain markers for which you know they exist, but you don’t know how to treat them,” she said. “This one is treatable.”
For their research, Dr. Lespinasse, Dr. Dufouil, and colleagues used a cross-sectional sample of data from the MEMENTO study, a 5-year observational cohort of 2,323 patients recruited at 26 memory centers in France between 2011 and 2014. Of the patients in MEMENTO, 62% were women, and the mean age was 71. All patients were deemed free of dementia and had isolated cognitive complaints or mild cognitive impairment at baseline. A total of 60% had hypertension, and 17% had uncontrolled hypertension defined as above 140/90 mm Hg despite treatment. Cognitive testing and MRI was conducted on all patients, while 60% also had 18F-fluorodeoxyglucose PET scanning and a minority, 18%, had cerebrospinal fluid samples.
The investigators found in using a structural equation model that the uncontrolled hypertension subjects had significantly lower cognition when compared against those without (P = .001). About half of the harmful effect of uncontrolled hypertension on brain functions was mediated by white matter hyperintensities load (P = .021) and neurodegeneration (P = .024) but not by cerebrospinal fluid biomarkers for amyloid-beta 42/40 ratio or tau.
The study’s main limitation was its use of cross-sectional data, the investigators said, while its strength was in a multifactorial model that allowed for a more integrative look at the relationships among hypertension, Alzheimer’s biomarkers, white matter hyperintensities, and cognition.
The investigators stressed the importance of controlling hypertension generally – and for all clinicians to be more aware of its cognitive impacts. Dr. Dufouil said that memory clinics should make blood pressure monitoring a key part of their workups, and should ensure that people are well controlled. “Up until recently it hasn’t been obvious” that control of hypertension has a key role in dementia prevention.
“It’s now obvious,” she said.
Dr. Lespinasse and Dr. Dufouil disclosed no industry relationships.
LOS ANGELES – While uncontrolled hypertension is an established risk factor for dementias including Alzheimer’s disease, the pathways by which it might lead to dementia remain poorly understood.
In research presented at the Alzheimer’s Association International Conference, Jérémie Lespinasse, PharmD, an investigator with the University of Bordeaux and INSERM U1219 in Bordeaux, France, presented data showing that hypertension-linked increases in the brain’s load of white matter hyperintensities – an imaging biomarker linked to small-vessel disease – was associated with cognitive decline independent of amyloid cascade biomarkers.
“We know that hypertension is related to cognitive decline and dementia, including Alzheimer’s dementia,” Carole Dufouil, PhD, the study’s last author and the research director at INSERM U1219, said in an interview. “But we didn’t know the pathway. It could go through what’s more typical of vascular pathology or it could go through what’s more typical of AD – and that’s what we wanted to test.”
The researchers found that the impact of hypertension on cognition doesn’t “go through amyloid,” Dr. Dufouil said, but rather a typically vascular pathway of white matter hyperintensities and neurodegeneration. “This is a big difference from other brain markers for which you know they exist, but you don’t know how to treat them,” she said. “This one is treatable.”
For their research, Dr. Lespinasse, Dr. Dufouil, and colleagues used a cross-sectional sample of data from the MEMENTO study, a 5-year observational cohort of 2,323 patients recruited at 26 memory centers in France between 2011 and 2014. Of the patients in MEMENTO, 62% were women, and the mean age was 71. All patients were deemed free of dementia and had isolated cognitive complaints or mild cognitive impairment at baseline. A total of 60% had hypertension, and 17% had uncontrolled hypertension defined as above 140/90 mm Hg despite treatment. Cognitive testing and MRI was conducted on all patients, while 60% also had 18F-fluorodeoxyglucose PET scanning and a minority, 18%, had cerebrospinal fluid samples.
The investigators found in using a structural equation model that the uncontrolled hypertension subjects had significantly lower cognition when compared against those without (P = .001). About half of the harmful effect of uncontrolled hypertension on brain functions was mediated by white matter hyperintensities load (P = .021) and neurodegeneration (P = .024) but not by cerebrospinal fluid biomarkers for amyloid-beta 42/40 ratio or tau.
The study’s main limitation was its use of cross-sectional data, the investigators said, while its strength was in a multifactorial model that allowed for a more integrative look at the relationships among hypertension, Alzheimer’s biomarkers, white matter hyperintensities, and cognition.
The investigators stressed the importance of controlling hypertension generally – and for all clinicians to be more aware of its cognitive impacts. Dr. Dufouil said that memory clinics should make blood pressure monitoring a key part of their workups, and should ensure that people are well controlled. “Up until recently it hasn’t been obvious” that control of hypertension has a key role in dementia prevention.
“It’s now obvious,” she said.
Dr. Lespinasse and Dr. Dufouil disclosed no industry relationships.
LOS ANGELES – While uncontrolled hypertension is an established risk factor for dementias including Alzheimer’s disease, the pathways by which it might lead to dementia remain poorly understood.
In research presented at the Alzheimer’s Association International Conference, Jérémie Lespinasse, PharmD, an investigator with the University of Bordeaux and INSERM U1219 in Bordeaux, France, presented data showing that hypertension-linked increases in the brain’s load of white matter hyperintensities – an imaging biomarker linked to small-vessel disease – was associated with cognitive decline independent of amyloid cascade biomarkers.
“We know that hypertension is related to cognitive decline and dementia, including Alzheimer’s dementia,” Carole Dufouil, PhD, the study’s last author and the research director at INSERM U1219, said in an interview. “But we didn’t know the pathway. It could go through what’s more typical of vascular pathology or it could go through what’s more typical of AD – and that’s what we wanted to test.”
The researchers found that the impact of hypertension on cognition doesn’t “go through amyloid,” Dr. Dufouil said, but rather a typically vascular pathway of white matter hyperintensities and neurodegeneration. “This is a big difference from other brain markers for which you know they exist, but you don’t know how to treat them,” she said. “This one is treatable.”
For their research, Dr. Lespinasse, Dr. Dufouil, and colleagues used a cross-sectional sample of data from the MEMENTO study, a 5-year observational cohort of 2,323 patients recruited at 26 memory centers in France between 2011 and 2014. Of the patients in MEMENTO, 62% were women, and the mean age was 71. All patients were deemed free of dementia and had isolated cognitive complaints or mild cognitive impairment at baseline. A total of 60% had hypertension, and 17% had uncontrolled hypertension defined as above 140/90 mm Hg despite treatment. Cognitive testing and MRI was conducted on all patients, while 60% also had 18F-fluorodeoxyglucose PET scanning and a minority, 18%, had cerebrospinal fluid samples.
The investigators found in using a structural equation model that the uncontrolled hypertension subjects had significantly lower cognition when compared against those without (P = .001). About half of the harmful effect of uncontrolled hypertension on brain functions was mediated by white matter hyperintensities load (P = .021) and neurodegeneration (P = .024) but not by cerebrospinal fluid biomarkers for amyloid-beta 42/40 ratio or tau.
The study’s main limitation was its use of cross-sectional data, the investigators said, while its strength was in a multifactorial model that allowed for a more integrative look at the relationships among hypertension, Alzheimer’s biomarkers, white matter hyperintensities, and cognition.
The investigators stressed the importance of controlling hypertension generally – and for all clinicians to be more aware of its cognitive impacts. Dr. Dufouil said that memory clinics should make blood pressure monitoring a key part of their workups, and should ensure that people are well controlled. “Up until recently it hasn’t been obvious” that control of hypertension has a key role in dementia prevention.
“It’s now obvious,” she said.
Dr. Lespinasse and Dr. Dufouil disclosed no industry relationships.
REPORTING FROM AAIC 2019
Alzheimer’s disease raises risk for recurrent seizures
LOS ANGELES – Seizures are not uncommon among patients with Alzheimer’s disease – particularly as patients live longer with the disease – and are often associated with worse cognitive and functional performance, according to research findings presented at the Alzheimer’s Association International Conference.
Jonathan Vöglein, MD, of the German Center for Neurodegenerative Diseases and Ludwig-Maximilian University in Munich presented results from a cohort of 9,127 patients with Alzheimer’s disease (AD), of whom 287 had experienced a seizure, and more than 10,000 non-AD control subjects recruited at clinics during 2005-2016.
Dr. Vöglein and colleagues found that seizure risk increased with duration of disease, from 1.5% of patients at 4.8 years with the disease to 5.4% at 11 years, with likelihood of a seizure increasing steadily over time.
Moreover, 70% of AD patients who experienced a seizure had a second one within 7.5 months. People who had seizures fared worse on cognitive and functional tests: a mean 16.6 on the Mini Mental State Examination, compared with 19.6 for patients without seizures. On a severity rating scale, the Clinical Dementia Rating Sum of Boxes, patients with seizures also fared worse, with scores of 9.3, compared with 6.8 for patients without seizures (P less than .0001 for all, with results adjusted for age and disease duration).
“The data of our study show that there’s an association of seizures with worse cognitive and functional performance,” Dr. Vöglein said in an interview.
“It’s important for clinicians to know that Alzheimer’s patients are at an increased risk for seizures,” Dr. Vöglein said. “In my clinical care experience, seizures are rarely the main complaint of patients with Alzheimer’s disease.” Detailed interviews with the patient and a proxy are important, he added, because patients with Alzheimer’s disease may not always remember events that could be a seizure.
Dr. Vöglein noted that, to his knowledge, there are no reliable data showing that treating seizures with antiepileptic drugs slows cognitive decline. “The results of our study suggest that an antiepileptic treatment after a first seizure in patients with Alzheimer’s dementia may be considered,” he said.
Also at the conference, researcher Ruby Castilla-Puentes, MD, DrPH, of Janssen Pharmaceuticals in Hopewell, N.J., along with Miguel Habeych, MD, MPH, of the University of Cincinnati presented findings on dementia and seizure risk from a large U.S. national managed care database of nearly 3 million people aged 60 years and older, of whom 56% were women.
The researchers analyzed this cohort during 2005-2014 and identified 80,000 people (2.8% of the cohort) as having any dementia diagnosis. The overall incidence of new-onset seizures in patients with dementia was 12.3% per year. In general, all subtypes of seizures and epileptic disorders (partial, generalized, or undifferentiated) occurred more frequently in patients with dementia, compared against patients without dementia (P less than .0001).
People with dementia had more than six times greater risk for experiencing recurring epileptic seizures than did people without dementia (95% confidence interval, 4.4-9.5). They were at six times higher risk for partial seizures (95% CI, 5.5-6.6); fivefold higher risk for generalized (95% CI, 4.9-5.5) and undifferentiated epilepsy (95% CI, 4.8-5.2); and 4.75 times higher risk for generalized seizures (95% CI, 4.5-5.0) and partial epilepsy (95% CI, 4.4-5.1).
“Although there are limitations with the use of administrative claims databases to calculate incidence rates, this analysis suggests that patients of 60 years of age or older have higher risks of new-onset seizures associated with a dementia diagnosis,” Dr. Castilla-Puentes commented.
The findings, she said, reinforce the need for clinicians to monitor for seizures to ensure that patients with dementia receive appropriate treatment.
Dr. Vöglein disclosed no financial conflicts of interest. Dr. Castilla-Puentes disclosed being an employee of Janssen, which funded her study.
LOS ANGELES – Seizures are not uncommon among patients with Alzheimer’s disease – particularly as patients live longer with the disease – and are often associated with worse cognitive and functional performance, according to research findings presented at the Alzheimer’s Association International Conference.
Jonathan Vöglein, MD, of the German Center for Neurodegenerative Diseases and Ludwig-Maximilian University in Munich presented results from a cohort of 9,127 patients with Alzheimer’s disease (AD), of whom 287 had experienced a seizure, and more than 10,000 non-AD control subjects recruited at clinics during 2005-2016.
Dr. Vöglein and colleagues found that seizure risk increased with duration of disease, from 1.5% of patients at 4.8 years with the disease to 5.4% at 11 years, with likelihood of a seizure increasing steadily over time.
Moreover, 70% of AD patients who experienced a seizure had a second one within 7.5 months. People who had seizures fared worse on cognitive and functional tests: a mean 16.6 on the Mini Mental State Examination, compared with 19.6 for patients without seizures. On a severity rating scale, the Clinical Dementia Rating Sum of Boxes, patients with seizures also fared worse, with scores of 9.3, compared with 6.8 for patients without seizures (P less than .0001 for all, with results adjusted for age and disease duration).
“The data of our study show that there’s an association of seizures with worse cognitive and functional performance,” Dr. Vöglein said in an interview.
“It’s important for clinicians to know that Alzheimer’s patients are at an increased risk for seizures,” Dr. Vöglein said. “In my clinical care experience, seizures are rarely the main complaint of patients with Alzheimer’s disease.” Detailed interviews with the patient and a proxy are important, he added, because patients with Alzheimer’s disease may not always remember events that could be a seizure.
Dr. Vöglein noted that, to his knowledge, there are no reliable data showing that treating seizures with antiepileptic drugs slows cognitive decline. “The results of our study suggest that an antiepileptic treatment after a first seizure in patients with Alzheimer’s dementia may be considered,” he said.
Also at the conference, researcher Ruby Castilla-Puentes, MD, DrPH, of Janssen Pharmaceuticals in Hopewell, N.J., along with Miguel Habeych, MD, MPH, of the University of Cincinnati presented findings on dementia and seizure risk from a large U.S. national managed care database of nearly 3 million people aged 60 years and older, of whom 56% were women.
The researchers analyzed this cohort during 2005-2014 and identified 80,000 people (2.8% of the cohort) as having any dementia diagnosis. The overall incidence of new-onset seizures in patients with dementia was 12.3% per year. In general, all subtypes of seizures and epileptic disorders (partial, generalized, or undifferentiated) occurred more frequently in patients with dementia, compared against patients without dementia (P less than .0001).
People with dementia had more than six times greater risk for experiencing recurring epileptic seizures than did people without dementia (95% confidence interval, 4.4-9.5). They were at six times higher risk for partial seizures (95% CI, 5.5-6.6); fivefold higher risk for generalized (95% CI, 4.9-5.5) and undifferentiated epilepsy (95% CI, 4.8-5.2); and 4.75 times higher risk for generalized seizures (95% CI, 4.5-5.0) and partial epilepsy (95% CI, 4.4-5.1).
“Although there are limitations with the use of administrative claims databases to calculate incidence rates, this analysis suggests that patients of 60 years of age or older have higher risks of new-onset seizures associated with a dementia diagnosis,” Dr. Castilla-Puentes commented.
The findings, she said, reinforce the need for clinicians to monitor for seizures to ensure that patients with dementia receive appropriate treatment.
Dr. Vöglein disclosed no financial conflicts of interest. Dr. Castilla-Puentes disclosed being an employee of Janssen, which funded her study.
LOS ANGELES – Seizures are not uncommon among patients with Alzheimer’s disease – particularly as patients live longer with the disease – and are often associated with worse cognitive and functional performance, according to research findings presented at the Alzheimer’s Association International Conference.
Jonathan Vöglein, MD, of the German Center for Neurodegenerative Diseases and Ludwig-Maximilian University in Munich presented results from a cohort of 9,127 patients with Alzheimer’s disease (AD), of whom 287 had experienced a seizure, and more than 10,000 non-AD control subjects recruited at clinics during 2005-2016.
Dr. Vöglein and colleagues found that seizure risk increased with duration of disease, from 1.5% of patients at 4.8 years with the disease to 5.4% at 11 years, with likelihood of a seizure increasing steadily over time.
Moreover, 70% of AD patients who experienced a seizure had a second one within 7.5 months. People who had seizures fared worse on cognitive and functional tests: a mean 16.6 on the Mini Mental State Examination, compared with 19.6 for patients without seizures. On a severity rating scale, the Clinical Dementia Rating Sum of Boxes, patients with seizures also fared worse, with scores of 9.3, compared with 6.8 for patients without seizures (P less than .0001 for all, with results adjusted for age and disease duration).
“The data of our study show that there’s an association of seizures with worse cognitive and functional performance,” Dr. Vöglein said in an interview.
“It’s important for clinicians to know that Alzheimer’s patients are at an increased risk for seizures,” Dr. Vöglein said. “In my clinical care experience, seizures are rarely the main complaint of patients with Alzheimer’s disease.” Detailed interviews with the patient and a proxy are important, he added, because patients with Alzheimer’s disease may not always remember events that could be a seizure.
Dr. Vöglein noted that, to his knowledge, there are no reliable data showing that treating seizures with antiepileptic drugs slows cognitive decline. “The results of our study suggest that an antiepileptic treatment after a first seizure in patients with Alzheimer’s dementia may be considered,” he said.
Also at the conference, researcher Ruby Castilla-Puentes, MD, DrPH, of Janssen Pharmaceuticals in Hopewell, N.J., along with Miguel Habeych, MD, MPH, of the University of Cincinnati presented findings on dementia and seizure risk from a large U.S. national managed care database of nearly 3 million people aged 60 years and older, of whom 56% were women.
The researchers analyzed this cohort during 2005-2014 and identified 80,000 people (2.8% of the cohort) as having any dementia diagnosis. The overall incidence of new-onset seizures in patients with dementia was 12.3% per year. In general, all subtypes of seizures and epileptic disorders (partial, generalized, or undifferentiated) occurred more frequently in patients with dementia, compared against patients without dementia (P less than .0001).
People with dementia had more than six times greater risk for experiencing recurring epileptic seizures than did people without dementia (95% confidence interval, 4.4-9.5). They were at six times higher risk for partial seizures (95% CI, 5.5-6.6); fivefold higher risk for generalized (95% CI, 4.9-5.5) and undifferentiated epilepsy (95% CI, 4.8-5.2); and 4.75 times higher risk for generalized seizures (95% CI, 4.5-5.0) and partial epilepsy (95% CI, 4.4-5.1).
“Although there are limitations with the use of administrative claims databases to calculate incidence rates, this analysis suggests that patients of 60 years of age or older have higher risks of new-onset seizures associated with a dementia diagnosis,” Dr. Castilla-Puentes commented.
The findings, she said, reinforce the need for clinicians to monitor for seizures to ensure that patients with dementia receive appropriate treatment.
Dr. Vöglein disclosed no financial conflicts of interest. Dr. Castilla-Puentes disclosed being an employee of Janssen, which funded her study.
REPORTING FROM AAIC 2019
Changes in sleep-wake timing accompany cerebral glucose hypometabolism and cognitive function
LOS ANGELES – Dysregulated sleep-wake cycles may be linked to cerebral glucose hypometabolism and subtle cognitive changes, both of which are early signs of Alzheimer’s disease–like neurodegeneration, according to a 2-year study of older Korean adults.
The association was particularly strong in subjects who experienced delayed acrophase, the peak of the normal sleep-wake cycle, So-Yeon Jeon, MD, said at the Alzheimer’s Association International Conference. It’s not yet clear whether the changes are a risk factor for dementia or a prodromal sign of neurodegeneration, but even without full elucidation, the findings could have value as a signal of impending neurodegeneration, said Dr. Jeon of Seoul (South Korea) National University.
“Our findings suggest that delayed acrophase may be used as a predictor for the progression of Alzheimer’s-type neurodegeneration and cognitive decline in the near future in old individuals with diverse cognitive status,” she said. “But the relationship between circadian phases and neurodegeneration is complex and not yet well understood.”
The 24-month study comprised 215 elderly adults enrolled in the Korean Brain Aging Study for the Early Diagnosis and Prediction of Alzheimer’s Disease (KBASE). They were a mean of 70 years old at baseline; 143 were cognitively normal, 40 had mild cognitive impairment, and 32 had Alzheimer’s dementia. Both at baseline and 2 years, everyone underwent a comprehensive neuropsychological assessment, amyloid PET brain imaging with Pittsburgh compound B, and an [18F]-fluorodeoxyglucose PET scan to determine brain glucose metabolic rate.
Before each assessment, the investigators measured sleep and circadian rhythms with 8 days of actigraphy. This assessed sleep variables (total sleep time, sleep latency, sleep efficiency, and wakefulness after sleep); rest-activity rhythm variables (midline estimated statistic of rhythm, amplitude, and acrophase), and some nonparametric values including interdaily stability, intradaily variability, and relative amplitude of sleep cycles. Subjects also completed sleep diaries during these periods.
The study’s main outcomes were 2-year changes in the Mini Mental State Exam (MMSE) score and in Alzheimer’s imaging biomarkers, including glucose metabolism and amyloid deposition. All analyses controlled for age, sex, Clinical Dementia Rating score, apolipoprotein E allele status, and baseline cognition.
At baseline, lower total sleep time was significantly associated with hypometabolism in areas associated with Alzheimer’s pathology as well as lower mean MMSE scores. Circadian variables showed no significant associations with these characteristics. However, the relative amplitude of circadian rhythm was significantly associated with hypometabolism and with lower MMSE score. There were no associations with brain amyloid load.
At 2 years, acrophase was associated with declines in cerebral glucose metabolism and further changes in the MMSE, even after the researchers controlled for the potential confounders. Delayed acrophase, although not associated with either metabolic rate or cognition at baseline, did significantly influence both at 2 years, suggesting a rapidly eroding clinical picture.
“Neurodegeneration over 2 years means the disease is progressing rapidly and subjects are likely to have tauopathies or other proteinopathy,” Dr. Jeon said. “These pathologies may either be resulting in delayed acrophase followed by neurodegeneration, or they may be prodromal symptoms of impending neurodegeneration. Whether they are early symptoms or early risk factors is not currently known, however. Two years is too short of a follow-up to determine these questions.”
Dr. Jeon had no financial declarations.
SOURCE: Jeon SY et al. AAIC 2019, abstract 33543.
LOS ANGELES – Dysregulated sleep-wake cycles may be linked to cerebral glucose hypometabolism and subtle cognitive changes, both of which are early signs of Alzheimer’s disease–like neurodegeneration, according to a 2-year study of older Korean adults.
The association was particularly strong in subjects who experienced delayed acrophase, the peak of the normal sleep-wake cycle, So-Yeon Jeon, MD, said at the Alzheimer’s Association International Conference. It’s not yet clear whether the changes are a risk factor for dementia or a prodromal sign of neurodegeneration, but even without full elucidation, the findings could have value as a signal of impending neurodegeneration, said Dr. Jeon of Seoul (South Korea) National University.
“Our findings suggest that delayed acrophase may be used as a predictor for the progression of Alzheimer’s-type neurodegeneration and cognitive decline in the near future in old individuals with diverse cognitive status,” she said. “But the relationship between circadian phases and neurodegeneration is complex and not yet well understood.”
The 24-month study comprised 215 elderly adults enrolled in the Korean Brain Aging Study for the Early Diagnosis and Prediction of Alzheimer’s Disease (KBASE). They were a mean of 70 years old at baseline; 143 were cognitively normal, 40 had mild cognitive impairment, and 32 had Alzheimer’s dementia. Both at baseline and 2 years, everyone underwent a comprehensive neuropsychological assessment, amyloid PET brain imaging with Pittsburgh compound B, and an [18F]-fluorodeoxyglucose PET scan to determine brain glucose metabolic rate.
Before each assessment, the investigators measured sleep and circadian rhythms with 8 days of actigraphy. This assessed sleep variables (total sleep time, sleep latency, sleep efficiency, and wakefulness after sleep); rest-activity rhythm variables (midline estimated statistic of rhythm, amplitude, and acrophase), and some nonparametric values including interdaily stability, intradaily variability, and relative amplitude of sleep cycles. Subjects also completed sleep diaries during these periods.
The study’s main outcomes were 2-year changes in the Mini Mental State Exam (MMSE) score and in Alzheimer’s imaging biomarkers, including glucose metabolism and amyloid deposition. All analyses controlled for age, sex, Clinical Dementia Rating score, apolipoprotein E allele status, and baseline cognition.
At baseline, lower total sleep time was significantly associated with hypometabolism in areas associated with Alzheimer’s pathology as well as lower mean MMSE scores. Circadian variables showed no significant associations with these characteristics. However, the relative amplitude of circadian rhythm was significantly associated with hypometabolism and with lower MMSE score. There were no associations with brain amyloid load.
At 2 years, acrophase was associated with declines in cerebral glucose metabolism and further changes in the MMSE, even after the researchers controlled for the potential confounders. Delayed acrophase, although not associated with either metabolic rate or cognition at baseline, did significantly influence both at 2 years, suggesting a rapidly eroding clinical picture.
“Neurodegeneration over 2 years means the disease is progressing rapidly and subjects are likely to have tauopathies or other proteinopathy,” Dr. Jeon said. “These pathologies may either be resulting in delayed acrophase followed by neurodegeneration, or they may be prodromal symptoms of impending neurodegeneration. Whether they are early symptoms or early risk factors is not currently known, however. Two years is too short of a follow-up to determine these questions.”
Dr. Jeon had no financial declarations.
SOURCE: Jeon SY et al. AAIC 2019, abstract 33543.
LOS ANGELES – Dysregulated sleep-wake cycles may be linked to cerebral glucose hypometabolism and subtle cognitive changes, both of which are early signs of Alzheimer’s disease–like neurodegeneration, according to a 2-year study of older Korean adults.
The association was particularly strong in subjects who experienced delayed acrophase, the peak of the normal sleep-wake cycle, So-Yeon Jeon, MD, said at the Alzheimer’s Association International Conference. It’s not yet clear whether the changes are a risk factor for dementia or a prodromal sign of neurodegeneration, but even without full elucidation, the findings could have value as a signal of impending neurodegeneration, said Dr. Jeon of Seoul (South Korea) National University.
“Our findings suggest that delayed acrophase may be used as a predictor for the progression of Alzheimer’s-type neurodegeneration and cognitive decline in the near future in old individuals with diverse cognitive status,” she said. “But the relationship between circadian phases and neurodegeneration is complex and not yet well understood.”
The 24-month study comprised 215 elderly adults enrolled in the Korean Brain Aging Study for the Early Diagnosis and Prediction of Alzheimer’s Disease (KBASE). They were a mean of 70 years old at baseline; 143 were cognitively normal, 40 had mild cognitive impairment, and 32 had Alzheimer’s dementia. Both at baseline and 2 years, everyone underwent a comprehensive neuropsychological assessment, amyloid PET brain imaging with Pittsburgh compound B, and an [18F]-fluorodeoxyglucose PET scan to determine brain glucose metabolic rate.
Before each assessment, the investigators measured sleep and circadian rhythms with 8 days of actigraphy. This assessed sleep variables (total sleep time, sleep latency, sleep efficiency, and wakefulness after sleep); rest-activity rhythm variables (midline estimated statistic of rhythm, amplitude, and acrophase), and some nonparametric values including interdaily stability, intradaily variability, and relative amplitude of sleep cycles. Subjects also completed sleep diaries during these periods.
The study’s main outcomes were 2-year changes in the Mini Mental State Exam (MMSE) score and in Alzheimer’s imaging biomarkers, including glucose metabolism and amyloid deposition. All analyses controlled for age, sex, Clinical Dementia Rating score, apolipoprotein E allele status, and baseline cognition.
At baseline, lower total sleep time was significantly associated with hypometabolism in areas associated with Alzheimer’s pathology as well as lower mean MMSE scores. Circadian variables showed no significant associations with these characteristics. However, the relative amplitude of circadian rhythm was significantly associated with hypometabolism and with lower MMSE score. There were no associations with brain amyloid load.
At 2 years, acrophase was associated with declines in cerebral glucose metabolism and further changes in the MMSE, even after the researchers controlled for the potential confounders. Delayed acrophase, although not associated with either metabolic rate or cognition at baseline, did significantly influence both at 2 years, suggesting a rapidly eroding clinical picture.
“Neurodegeneration over 2 years means the disease is progressing rapidly and subjects are likely to have tauopathies or other proteinopathy,” Dr. Jeon said. “These pathologies may either be resulting in delayed acrophase followed by neurodegeneration, or they may be prodromal symptoms of impending neurodegeneration. Whether they are early symptoms or early risk factors is not currently known, however. Two years is too short of a follow-up to determine these questions.”
Dr. Jeon had no financial declarations.
SOURCE: Jeon SY et al. AAIC 2019, abstract 33543.
REPORTING FROM AAIC 2019
Metformin linked to lower dementia risk in black patients
Black individuals who develop type 2 diabetes are more likely than their white counterparts to develop dementia. Now, findings from a new study point to a possible preventive strategy: Putting older patients on metformin when they are diagnosed could reduce their risk for dementia by as much as 40%, whereas sulfonylureas do not seem to have such an effect.
The researchers did not examine cause and effect, so their findings are not conclusive, and very few women were included in the study. Still, the authors said that their data showing a 29% lower risk of dementia associated with metformin use in black patients aged 65-74 years, and a 40% lower risk in those aged 50-64 years, suggested that “this inexpensive, widely available treatment could be broadly prescribed to substantially reduce the risk of dementia in younger [black] patients with [type 2 diabetes]” (Ann Fam Med. 2019;17:352-62).
Previous findings have suggested that black patients with type 2 diabetes face a 10%-18% higher risk of dementia, compared with white patients (Diabetes Care. 2014; 37[4]:1009-15). Another study linked type 2 diabetes in middle-aged black patients to a 41% decrease in cognition per test results over 14 years. There was no such decrease in white patients (Neuroepidemiology. 2014;43[3-4]: 220-7).
For the new study, researchers led by Jeffrey F. Scherrer, PhD, of Saint Louis University tracked 73,761 patients aged 50 years or older from 2000-2001 (when they were free of dementia and not taking diabetes) to 2015. Among the patients, 86% were white and 14% were black. In the white and black groups, 97% and 95% were men, respectively, and 61% and 55% were obese, respectively.
All participants began metformin (76%) or sulfonylurea (24%) monotherapy after the baseline period. Guidelines recommend metformin as a first-line treatment for type 2 diabetes, whereas sulfonylureas are considered second-line drugs that should be added to metformin.
After adjustment for confounders such as socioeconomic status and other medical conditions, the researchers found a significantly lower risk of dementia in black patients who took metformin, compared with those taking a sulfonylurea (hazard ratio, 0.73; 95% confidence interval, 0.6-0.89). There was no difference between the drugs among white patients (HR, 0.96; 95% CI, 0.9-1.03, both P = .008)
The results were not statistically significant among age groups, but there were trends. In black patients, the dementia-lowering benefit was largest among those aged 50-64 years (HR, 0.6; 95% CI, 0.45-0.81), followed by those aged 65-74 years (HR, 0.71; 95% CI, 0.53-0.94), and there was no benefit among those aged at least 75 (HR, 1.17; 95% CI, 0.73-1.85) all P = .055. There was a slight benefit among white patients in one of the age groups – 65-74 years (HR, 0.9; 95% CI, 0.82-0.99; P = .315).
The authors suggested that the findings could have been the result of an effect of metformin to reduce vascular disease and chronic inflammation in black patients.
They also noted that further research is needed to identify the demographic and clinical subgroups in which metformin is most strongly associated with a reduction in the risk of dementia. In addition, they emphasized that clinical trials are needed to confirm the study findings.
The National Institutes of Health funded the study. The authors report no relevant disclosures.
SOURCE: Scherrer JF et al. Ann Fam Med. 2019;17:352-62.
Black individuals who develop type 2 diabetes are more likely than their white counterparts to develop dementia. Now, findings from a new study point to a possible preventive strategy: Putting older patients on metformin when they are diagnosed could reduce their risk for dementia by as much as 40%, whereas sulfonylureas do not seem to have such an effect.
The researchers did not examine cause and effect, so their findings are not conclusive, and very few women were included in the study. Still, the authors said that their data showing a 29% lower risk of dementia associated with metformin use in black patients aged 65-74 years, and a 40% lower risk in those aged 50-64 years, suggested that “this inexpensive, widely available treatment could be broadly prescribed to substantially reduce the risk of dementia in younger [black] patients with [type 2 diabetes]” (Ann Fam Med. 2019;17:352-62).
Previous findings have suggested that black patients with type 2 diabetes face a 10%-18% higher risk of dementia, compared with white patients (Diabetes Care. 2014; 37[4]:1009-15). Another study linked type 2 diabetes in middle-aged black patients to a 41% decrease in cognition per test results over 14 years. There was no such decrease in white patients (Neuroepidemiology. 2014;43[3-4]: 220-7).
For the new study, researchers led by Jeffrey F. Scherrer, PhD, of Saint Louis University tracked 73,761 patients aged 50 years or older from 2000-2001 (when they were free of dementia and not taking diabetes) to 2015. Among the patients, 86% were white and 14% were black. In the white and black groups, 97% and 95% were men, respectively, and 61% and 55% were obese, respectively.
All participants began metformin (76%) or sulfonylurea (24%) monotherapy after the baseline period. Guidelines recommend metformin as a first-line treatment for type 2 diabetes, whereas sulfonylureas are considered second-line drugs that should be added to metformin.
After adjustment for confounders such as socioeconomic status and other medical conditions, the researchers found a significantly lower risk of dementia in black patients who took metformin, compared with those taking a sulfonylurea (hazard ratio, 0.73; 95% confidence interval, 0.6-0.89). There was no difference between the drugs among white patients (HR, 0.96; 95% CI, 0.9-1.03, both P = .008)
The results were not statistically significant among age groups, but there were trends. In black patients, the dementia-lowering benefit was largest among those aged 50-64 years (HR, 0.6; 95% CI, 0.45-0.81), followed by those aged 65-74 years (HR, 0.71; 95% CI, 0.53-0.94), and there was no benefit among those aged at least 75 (HR, 1.17; 95% CI, 0.73-1.85) all P = .055. There was a slight benefit among white patients in one of the age groups – 65-74 years (HR, 0.9; 95% CI, 0.82-0.99; P = .315).
The authors suggested that the findings could have been the result of an effect of metformin to reduce vascular disease and chronic inflammation in black patients.
They also noted that further research is needed to identify the demographic and clinical subgroups in which metformin is most strongly associated with a reduction in the risk of dementia. In addition, they emphasized that clinical trials are needed to confirm the study findings.
The National Institutes of Health funded the study. The authors report no relevant disclosures.
SOURCE: Scherrer JF et al. Ann Fam Med. 2019;17:352-62.
Black individuals who develop type 2 diabetes are more likely than their white counterparts to develop dementia. Now, findings from a new study point to a possible preventive strategy: Putting older patients on metformin when they are diagnosed could reduce their risk for dementia by as much as 40%, whereas sulfonylureas do not seem to have such an effect.
The researchers did not examine cause and effect, so their findings are not conclusive, and very few women were included in the study. Still, the authors said that their data showing a 29% lower risk of dementia associated with metformin use in black patients aged 65-74 years, and a 40% lower risk in those aged 50-64 years, suggested that “this inexpensive, widely available treatment could be broadly prescribed to substantially reduce the risk of dementia in younger [black] patients with [type 2 diabetes]” (Ann Fam Med. 2019;17:352-62).
Previous findings have suggested that black patients with type 2 diabetes face a 10%-18% higher risk of dementia, compared with white patients (Diabetes Care. 2014; 37[4]:1009-15). Another study linked type 2 diabetes in middle-aged black patients to a 41% decrease in cognition per test results over 14 years. There was no such decrease in white patients (Neuroepidemiology. 2014;43[3-4]: 220-7).
For the new study, researchers led by Jeffrey F. Scherrer, PhD, of Saint Louis University tracked 73,761 patients aged 50 years or older from 2000-2001 (when they were free of dementia and not taking diabetes) to 2015. Among the patients, 86% were white and 14% were black. In the white and black groups, 97% and 95% were men, respectively, and 61% and 55% were obese, respectively.
All participants began metformin (76%) or sulfonylurea (24%) monotherapy after the baseline period. Guidelines recommend metformin as a first-line treatment for type 2 diabetes, whereas sulfonylureas are considered second-line drugs that should be added to metformin.
After adjustment for confounders such as socioeconomic status and other medical conditions, the researchers found a significantly lower risk of dementia in black patients who took metformin, compared with those taking a sulfonylurea (hazard ratio, 0.73; 95% confidence interval, 0.6-0.89). There was no difference between the drugs among white patients (HR, 0.96; 95% CI, 0.9-1.03, both P = .008)
The results were not statistically significant among age groups, but there were trends. In black patients, the dementia-lowering benefit was largest among those aged 50-64 years (HR, 0.6; 95% CI, 0.45-0.81), followed by those aged 65-74 years (HR, 0.71; 95% CI, 0.53-0.94), and there was no benefit among those aged at least 75 (HR, 1.17; 95% CI, 0.73-1.85) all P = .055. There was a slight benefit among white patients in one of the age groups – 65-74 years (HR, 0.9; 95% CI, 0.82-0.99; P = .315).
The authors suggested that the findings could have been the result of an effect of metformin to reduce vascular disease and chronic inflammation in black patients.
They also noted that further research is needed to identify the demographic and clinical subgroups in which metformin is most strongly associated with a reduction in the risk of dementia. In addition, they emphasized that clinical trials are needed to confirm the study findings.
The National Institutes of Health funded the study. The authors report no relevant disclosures.
SOURCE: Scherrer JF et al. Ann Fam Med. 2019;17:352-62.
FROM ANNALS OF FAMILY MEDICINE
Key clinical point:
Major finding: Metformin monotherapy, compared with sulfonylurea monotherapy, was linked to a significantly lower risk for dementia in black patients (HR, 0.73; 95% CI, 0.6-0.89), but not in white patients (HR, 0.96; 95% CI, 0.9-1.03; P = .008).
Study details: Retrospective analysis of 73,761 patients aged 50 years or older in the Veterans Health Administration system who were tracked from 2000-2001 to 2015 and began metformin or sulfonylurea monotherapy after baseline.
Disclosures: The National Institutes of Health funded the study. The authors report no relevant disclosures.
Source: Scherrer JF et al. Ann Fam Med. 2019;17:352-62.
Treatment of delirium: A review of 3 studies
Delirium is defined as a disturbance in attention, awareness, and cognition that develops over hours to days as a direct physiological consequence of an underlying medical condition and is not better explained by another neurocognitive disorder.1 This condition is found in up to 31% of general medical patients and up to 87% of critically ill medical patients. Delirium is commonly seen in patients who have undergone surgery, those who are in palliative care, and patients with cancer.2 It is associated with increased morbidity and mortality. Compared with those who do not develop delirium, patients who are hospitalized who develop delirium have a higher risk of longer hospital stays, post-hospitalization nursing facility placement, persistent cognitive dysfunction, and death.3
Thus far, the management and treatment of delirium have been complicated by an incomplete understanding of the pathophysiology of this condition. However, prevailing theories suggest a dysregulation of neurotransmitter synthesis, function, or availability.2 Recent literature reflects this theory; researchers have investigated agents that target dopamine or acetylcholine. Below we review some of this recent literature on treating delirium; these studies are summarized in the Table.4-6

1. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalized non-ICU patients. Cochrane Database Syst Rev. 2018;6:CD005594.
An extensive literature review identified randomized or quasi-randomized trials on the treatment of delirium among non-critically ill hospitalized patients in which antipsychotics were compared with nonantipsychotic medications or placebo, or in which a first-generation antipsychotic (FGA) was compared with a second-generation antipsychotic (SGA).4
Study design
- Researchers conducted a literature review of 9 trials that included 727 hospitalized but not critically ill patients (ie, they were not in an ICU) who developed delirium.
- Four trials compared an antipsychotic with a medication from another drug class or with placebo.
- Seven trials compared a FGA with an SGA.
Outcomes
- Although the intended primary outcome was the duration of delirium, none of the included studies reported on duration of delirium. Secondary outcomes were delirium severity and resolution, mortality, hospital length of stay, discharge disposition, health-related quality of life, and adverse effects.
- Among the secondary outcomes, no statistical difference was observed between delirium severity, delirium resolution, or mortality.
- None of the included studies reported on hospital length of stay, discharge disposition, or health-related quality of life.
- Evidence related to adverse effects was determined to be very low quality due to potential bias, inconsistency, and imprecision.
Conclusion
- A review of 9 randomized trials did not find any evidence supporting the use of antipsychotics for treating delirium. However, most of the studies included were of lower quality because they were single-center trials with insufficient sample sizes, heterogeneous study populations, and risk of bias.
Continue to: 2...
2. Girard TD, Exline MC, Carson SS, et al; MIND-USA Investigators. Haloperidol and ziprasidone for treatment of delirium in critical illness. N Engl J Med. 2018;379(26):2506-2516.
Study design
- Researchers used the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU) to assess 1,183 patients with acute respiratory failure or shock in 16 medical centers in the United States.5
- Overall, 566 patients developed delirium and were randomized in a double-blind fashion to receive IV haloperidol, ziprasidone, or placebo.
- Haloperidol was started at 2.5 mg (age <70) or 1.25 mg (age ≥70) every 12 hours and titrated to a maximum dose of 20 mg/d as tolerated.
- Ziprasidone was started at 5 mg (age <70) or 2.5 mg (age ≥70) every 12 hours and titrated to a maximum dose of 40 mg/d as tolerated.
Outcomes
- The primary endpoint was days alive without delirium or coma. Secondary endpoints included duration of delirium, time to freedom from mechanical ventilation, time to final successful ICU discharge, time to ICU readmission, time to successful hospital discharge, 30-day survival, and 90-day survival.
- Neither ziprasidone nor haloperidol had an impact on number of days alive without delirium or coma.
- There was also no statistically significant difference in 30-day survival, 90-day survival, time to freedom from mechanical ventilation, ICU discharge, ICU readmission, or hospital discharge.
Conclusion
- This study found no evidence supporting haloperidol or ziprasidone for the treatment of delirium. Because all patients in this study were critically ill, it is unclear if these results would be generalizable to other hospitalized patient populations.
3. Yu A, Wu S, Zhang Z, et al. Cholinesterase inhibitors for the treatment of delirium in non-ICU settings. Cochrane Database Syst Rev. 2018;6:CD012494.
Study design
- A literature review identified published and unpublished randomized controlled trials in English and Chinese in which cholinesterase inhibitors were compared with placebo or another drug for treating delirium in non-critically ill patients.6
- Only one study met the criteria to be included in the review. It included 15 participants treated with rivastigmine or placebo.
Outcomes
- The intended primary outcomes were severity of delirium and duration of delirium. However, the included study did not report on the severity of delirium. It also lacked statistical power to determine a difference in duration of delirium between the rivastigmine and placebo groups.
- Secondary outcomes included use of a rescue medication, persistent cognitive impairment, length of hospitalization, institutionalization, mortality, cost of intervention, early departure from the study, and quality of life.
- There was no clear difference between the rivastigmine group and the placebo group in terms of the use of rescue medications, mortality, or early departure from the study. The included study did not report on persistent cognitive impairment, length of hospitalization, institutionalization, cost of intervention, or quality of life.
Conclusion
- This literature review did not find any evidence to support the use of cholinesterase inhibitors for treating delirium. However, because this review included only a single small study, limited conclusions can be drawn from this research.
In summary, delirium is common, especially among patients who are acutely medically ill, and it is associated with poor physical and cognitive clinical outcomes. Because of these poor outcomes, it is important to identify delirium early and intervene aggressively. Clearly, there is a need for further research into short- and long-term treatments for delirium.
1. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Maldonado JR. Acute brain failure: pathophysiology, diagnosis, management, and sequelae of delirium. Crit Care Clin. 2017;33(3):461-519.
3. Marcantonio ER. Delirium in hospitalized older adults. N Engl J Med. 2017;377(15):1456-1466.
4. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalized non-ICU patients. Cochrane Database Syst Rev. 2018;6:CD005594. doi: 10.1002/14651858.CD005594.pub3.
5. Girard TD, Exline MC, Carson SS, et al; MIND-USA Investigators. Haloperidol and ziprasidone for treatment of delirium in critical illness. N Engl J Med. 2018;379(26):2506-2516.
6. Yu A, Wu S, Zhang Z, et al. Cholinesterase inhibitors for the treatment of delirium in non-ICU settings. Cochrane Database Syst Rev. 2018;6:CD012494.
Delirium is defined as a disturbance in attention, awareness, and cognition that develops over hours to days as a direct physiological consequence of an underlying medical condition and is not better explained by another neurocognitive disorder.1 This condition is found in up to 31% of general medical patients and up to 87% of critically ill medical patients. Delirium is commonly seen in patients who have undergone surgery, those who are in palliative care, and patients with cancer.2 It is associated with increased morbidity and mortality. Compared with those who do not develop delirium, patients who are hospitalized who develop delirium have a higher risk of longer hospital stays, post-hospitalization nursing facility placement, persistent cognitive dysfunction, and death.3
Thus far, the management and treatment of delirium have been complicated by an incomplete understanding of the pathophysiology of this condition. However, prevailing theories suggest a dysregulation of neurotransmitter synthesis, function, or availability.2 Recent literature reflects this theory; researchers have investigated agents that target dopamine or acetylcholine. Below we review some of this recent literature on treating delirium; these studies are summarized in the Table.4-6

1. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalized non-ICU patients. Cochrane Database Syst Rev. 2018;6:CD005594.
An extensive literature review identified randomized or quasi-randomized trials on the treatment of delirium among non-critically ill hospitalized patients in which antipsychotics were compared with nonantipsychotic medications or placebo, or in which a first-generation antipsychotic (FGA) was compared with a second-generation antipsychotic (SGA).4
Study design
- Researchers conducted a literature review of 9 trials that included 727 hospitalized but not critically ill patients (ie, they were not in an ICU) who developed delirium.
- Four trials compared an antipsychotic with a medication from another drug class or with placebo.
- Seven trials compared a FGA with an SGA.
Outcomes
- Although the intended primary outcome was the duration of delirium, none of the included studies reported on duration of delirium. Secondary outcomes were delirium severity and resolution, mortality, hospital length of stay, discharge disposition, health-related quality of life, and adverse effects.
- Among the secondary outcomes, no statistical difference was observed between delirium severity, delirium resolution, or mortality.
- None of the included studies reported on hospital length of stay, discharge disposition, or health-related quality of life.
- Evidence related to adverse effects was determined to be very low quality due to potential bias, inconsistency, and imprecision.
Conclusion
- A review of 9 randomized trials did not find any evidence supporting the use of antipsychotics for treating delirium. However, most of the studies included were of lower quality because they were single-center trials with insufficient sample sizes, heterogeneous study populations, and risk of bias.
Continue to: 2...
2. Girard TD, Exline MC, Carson SS, et al; MIND-USA Investigators. Haloperidol and ziprasidone for treatment of delirium in critical illness. N Engl J Med. 2018;379(26):2506-2516.
Study design
- Researchers used the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU) to assess 1,183 patients with acute respiratory failure or shock in 16 medical centers in the United States.5
- Overall, 566 patients developed delirium and were randomized in a double-blind fashion to receive IV haloperidol, ziprasidone, or placebo.
- Haloperidol was started at 2.5 mg (age <70) or 1.25 mg (age ≥70) every 12 hours and titrated to a maximum dose of 20 mg/d as tolerated.
- Ziprasidone was started at 5 mg (age <70) or 2.5 mg (age ≥70) every 12 hours and titrated to a maximum dose of 40 mg/d as tolerated.
Outcomes
- The primary endpoint was days alive without delirium or coma. Secondary endpoints included duration of delirium, time to freedom from mechanical ventilation, time to final successful ICU discharge, time to ICU readmission, time to successful hospital discharge, 30-day survival, and 90-day survival.
- Neither ziprasidone nor haloperidol had an impact on number of days alive without delirium or coma.
- There was also no statistically significant difference in 30-day survival, 90-day survival, time to freedom from mechanical ventilation, ICU discharge, ICU readmission, or hospital discharge.
Conclusion
- This study found no evidence supporting haloperidol or ziprasidone for the treatment of delirium. Because all patients in this study were critically ill, it is unclear if these results would be generalizable to other hospitalized patient populations.
3. Yu A, Wu S, Zhang Z, et al. Cholinesterase inhibitors for the treatment of delirium in non-ICU settings. Cochrane Database Syst Rev. 2018;6:CD012494.
Study design
- A literature review identified published and unpublished randomized controlled trials in English and Chinese in which cholinesterase inhibitors were compared with placebo or another drug for treating delirium in non-critically ill patients.6
- Only one study met the criteria to be included in the review. It included 15 participants treated with rivastigmine or placebo.
Outcomes
- The intended primary outcomes were severity of delirium and duration of delirium. However, the included study did not report on the severity of delirium. It also lacked statistical power to determine a difference in duration of delirium between the rivastigmine and placebo groups.
- Secondary outcomes included use of a rescue medication, persistent cognitive impairment, length of hospitalization, institutionalization, mortality, cost of intervention, early departure from the study, and quality of life.
- There was no clear difference between the rivastigmine group and the placebo group in terms of the use of rescue medications, mortality, or early departure from the study. The included study did not report on persistent cognitive impairment, length of hospitalization, institutionalization, cost of intervention, or quality of life.
Conclusion
- This literature review did not find any evidence to support the use of cholinesterase inhibitors for treating delirium. However, because this review included only a single small study, limited conclusions can be drawn from this research.
In summary, delirium is common, especially among patients who are acutely medically ill, and it is associated with poor physical and cognitive clinical outcomes. Because of these poor outcomes, it is important to identify delirium early and intervene aggressively. Clearly, there is a need for further research into short- and long-term treatments for delirium.
Delirium is defined as a disturbance in attention, awareness, and cognition that develops over hours to days as a direct physiological consequence of an underlying medical condition and is not better explained by another neurocognitive disorder.1 This condition is found in up to 31% of general medical patients and up to 87% of critically ill medical patients. Delirium is commonly seen in patients who have undergone surgery, those who are in palliative care, and patients with cancer.2 It is associated with increased morbidity and mortality. Compared with those who do not develop delirium, patients who are hospitalized who develop delirium have a higher risk of longer hospital stays, post-hospitalization nursing facility placement, persistent cognitive dysfunction, and death.3
Thus far, the management and treatment of delirium have been complicated by an incomplete understanding of the pathophysiology of this condition. However, prevailing theories suggest a dysregulation of neurotransmitter synthesis, function, or availability.2 Recent literature reflects this theory; researchers have investigated agents that target dopamine or acetylcholine. Below we review some of this recent literature on treating delirium; these studies are summarized in the Table.4-6

1. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalized non-ICU patients. Cochrane Database Syst Rev. 2018;6:CD005594.
An extensive literature review identified randomized or quasi-randomized trials on the treatment of delirium among non-critically ill hospitalized patients in which antipsychotics were compared with nonantipsychotic medications or placebo, or in which a first-generation antipsychotic (FGA) was compared with a second-generation antipsychotic (SGA).4
Study design
- Researchers conducted a literature review of 9 trials that included 727 hospitalized but not critically ill patients (ie, they were not in an ICU) who developed delirium.
- Four trials compared an antipsychotic with a medication from another drug class or with placebo.
- Seven trials compared a FGA with an SGA.
Outcomes
- Although the intended primary outcome was the duration of delirium, none of the included studies reported on duration of delirium. Secondary outcomes were delirium severity and resolution, mortality, hospital length of stay, discharge disposition, health-related quality of life, and adverse effects.
- Among the secondary outcomes, no statistical difference was observed between delirium severity, delirium resolution, or mortality.
- None of the included studies reported on hospital length of stay, discharge disposition, or health-related quality of life.
- Evidence related to adverse effects was determined to be very low quality due to potential bias, inconsistency, and imprecision.
Conclusion
- A review of 9 randomized trials did not find any evidence supporting the use of antipsychotics for treating delirium. However, most of the studies included were of lower quality because they were single-center trials with insufficient sample sizes, heterogeneous study populations, and risk of bias.
Continue to: 2...
2. Girard TD, Exline MC, Carson SS, et al; MIND-USA Investigators. Haloperidol and ziprasidone for treatment of delirium in critical illness. N Engl J Med. 2018;379(26):2506-2516.
Study design
- Researchers used the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU) to assess 1,183 patients with acute respiratory failure or shock in 16 medical centers in the United States.5
- Overall, 566 patients developed delirium and were randomized in a double-blind fashion to receive IV haloperidol, ziprasidone, or placebo.
- Haloperidol was started at 2.5 mg (age <70) or 1.25 mg (age ≥70) every 12 hours and titrated to a maximum dose of 20 mg/d as tolerated.
- Ziprasidone was started at 5 mg (age <70) or 2.5 mg (age ≥70) every 12 hours and titrated to a maximum dose of 40 mg/d as tolerated.
Outcomes
- The primary endpoint was days alive without delirium or coma. Secondary endpoints included duration of delirium, time to freedom from mechanical ventilation, time to final successful ICU discharge, time to ICU readmission, time to successful hospital discharge, 30-day survival, and 90-day survival.
- Neither ziprasidone nor haloperidol had an impact on number of days alive without delirium or coma.
- There was also no statistically significant difference in 30-day survival, 90-day survival, time to freedom from mechanical ventilation, ICU discharge, ICU readmission, or hospital discharge.
Conclusion
- This study found no evidence supporting haloperidol or ziprasidone for the treatment of delirium. Because all patients in this study were critically ill, it is unclear if these results would be generalizable to other hospitalized patient populations.
3. Yu A, Wu S, Zhang Z, et al. Cholinesterase inhibitors for the treatment of delirium in non-ICU settings. Cochrane Database Syst Rev. 2018;6:CD012494.
Study design
- A literature review identified published and unpublished randomized controlled trials in English and Chinese in which cholinesterase inhibitors were compared with placebo or another drug for treating delirium in non-critically ill patients.6
- Only one study met the criteria to be included in the review. It included 15 participants treated with rivastigmine or placebo.
Outcomes
- The intended primary outcomes were severity of delirium and duration of delirium. However, the included study did not report on the severity of delirium. It also lacked statistical power to determine a difference in duration of delirium between the rivastigmine and placebo groups.
- Secondary outcomes included use of a rescue medication, persistent cognitive impairment, length of hospitalization, institutionalization, mortality, cost of intervention, early departure from the study, and quality of life.
- There was no clear difference between the rivastigmine group and the placebo group in terms of the use of rescue medications, mortality, or early departure from the study. The included study did not report on persistent cognitive impairment, length of hospitalization, institutionalization, cost of intervention, or quality of life.
Conclusion
- This literature review did not find any evidence to support the use of cholinesterase inhibitors for treating delirium. However, because this review included only a single small study, limited conclusions can be drawn from this research.
In summary, delirium is common, especially among patients who are acutely medically ill, and it is associated with poor physical and cognitive clinical outcomes. Because of these poor outcomes, it is important to identify delirium early and intervene aggressively. Clearly, there is a need for further research into short- and long-term treatments for delirium.
1. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Maldonado JR. Acute brain failure: pathophysiology, diagnosis, management, and sequelae of delirium. Crit Care Clin. 2017;33(3):461-519.
3. Marcantonio ER. Delirium in hospitalized older adults. N Engl J Med. 2017;377(15):1456-1466.
4. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalized non-ICU patients. Cochrane Database Syst Rev. 2018;6:CD005594. doi: 10.1002/14651858.CD005594.pub3.
5. Girard TD, Exline MC, Carson SS, et al; MIND-USA Investigators. Haloperidol and ziprasidone for treatment of delirium in critical illness. N Engl J Med. 2018;379(26):2506-2516.
6. Yu A, Wu S, Zhang Z, et al. Cholinesterase inhibitors for the treatment of delirium in non-ICU settings. Cochrane Database Syst Rev. 2018;6:CD012494.
1. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Maldonado JR. Acute brain failure: pathophysiology, diagnosis, management, and sequelae of delirium. Crit Care Clin. 2017;33(3):461-519.
3. Marcantonio ER. Delirium in hospitalized older adults. N Engl J Med. 2017;377(15):1456-1466.
4. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalized non-ICU patients. Cochrane Database Syst Rev. 2018;6:CD005594. doi: 10.1002/14651858.CD005594.pub3.
5. Girard TD, Exline MC, Carson SS, et al; MIND-USA Investigators. Haloperidol and ziprasidone for treatment of delirium in critical illness. N Engl J Med. 2018;379(26):2506-2516.
6. Yu A, Wu S, Zhang Z, et al. Cholinesterase inhibitors for the treatment of delirium in non-ICU settings. Cochrane Database Syst Rev. 2018;6:CD012494.
Stigma in dementia: It’s time to talk about it
Dementia is a family of disorders characterized by a decline in multiple cognitive abilities that significantly interferes with an individual’s functioning. An estimated 50 million people are living with a dementia worldwide.1 Alzheimer’s disease (AD) is the leading cause of dementia, accounting for approximately two-thirds of dementia cases.1 These numbers are expected to increase dramatically in the upcoming decades.
Sociologist Erving Goffman defined stigma as “an attribute, behaviour, or reputation which is socially discrediting in a particular way: it causes an individual to be mentally classified by others in an undesirable, rejected stereotype rather than in an accepted, normal one.”2 Goffman2 defined 3 broad categories of stigma: public, self, and courtesy (Table 12).

Considerable evidence shows that the combined impact of having dementia and the negative response to the diagnosis significantly undermines an individual’s psychosocial well-being and quality of life.3 Persons with dementia (PwD) commonly report a loss of identity and self-worth, and stigma appears to deepen this distress.3 Stigma also negatively affects individuals associated with PwD, including family members and professionals. In this article, we discuss the impact of dementia-related stigma, and steps you can take to address it, including implementing person-centered clinical practices, promoting anti-stigma messaging campaigns, and advocating for public policy action to improve the lives of PwD and their families.
A pervasive problem
Although the Alzheimer’s Society International and the World Health Organization acknowledge that stigma has a central role in defining the experience of AD, how stigma may present, how clinicians and researchers can recognize and measure stigma, and how to best combat it have been understudied.3-5 A recent systematic literature review examined worldwide evidence on dementia-related stigma over the past decade.6 Hermann et al6 found that health care providers and the general public may hold stigmatizing attitudes toward PwD, and that stigma may be particularly harsh among racial and ethnic minorities, although the literature is scarce in this area. Cultural factors may also worsen stigma, and stigma may be associated with reduced awareness of dementia services and reduced help-seeking among minority groups.7,8 Studies show that stigmatizing attitudes are more pronounced in people with limited knowledge of dementia, in those with little contact with PwD, in men, in younger individuals, and in the context of cultural interpretations of dementia.6 Health care providers can also sometimes contribute to the perpetuation of stigma.6
In terms of standardized scales or instruments for evaluating dementia-related stigma, there is no uniformly accepted “gold standard” measure, which makes it difficult to compare studies.6 In order to effectively study efforts to reduce stigma, researchers need to identify and establish a consensus on rating scales for evaluating stigma among PwD, caregivers, and the general public. Three instruments that may be used for this purpose are the Family Stigma in Alzheimer’s Disease Scale (FS-ADS),9 the Stigma Scale for Chronic Illness (SSCI),10 and the Perceptions Regarding Investigational Screening for Memory in Primary Care (PRISM-PC).11
The detrimental effects of stigma
Burgener et al12 reported that personal stigma impacted functioning and quality of life in PwD. Higher levels of stigma were associated with higher anxiety, depression, and behavioral symptoms and lower self-esteem, social support, participation in activities, personal control, and physical health.12 Personal characteristics that may affect stigma include gender, location (rural vs urban), ethnicity, education level, and living arrangements (alone vs with family).12
In a subset of PwD with early-stage memory loss (n = 22), Burgener and Buckwalter13 found that 42% of participants were reluctant to reveal their diagnosis to others, with some fearing they would no longer be allowed to live alone and would be “sent to a facility.” In addition, 46% indicated they did not want “to be talked about like they were not there.” More than 50% of participants reported changes in their social network after receiving the diagnosis, including reducing activities and limiting types of contacts (ie, telephone only) or interacting only when “people come to me.” Participants were most comfortable with good friends “who understand” and persons within their faith communities. When asked about how they were treated by family members, >50% of participants described being treated differently, including loss of financial independence, more limited contact, and being “treated like a baby” by their children, who in general were uncomfortable talking about the diagnosis.
Continue to: In a recent study...
In a recent study by Harper et al,14 stigma was prevalent in the experience of PwD. One participant disclosed:
“I think there is [are] people I know who don’t ask me to go places or do things ’cause I have a dementia…I think lots of people don’t know what dementia is and I think it scares them ’cause they think of it as crazy. It hurts…”
Another participant said:
“I have had friends for over thirty years. They have turned their backs on me…we used to go for walks and they would phone me and go for coffee. Now I don’t hear from any of them…those aren’t true friends…true friends will stand behind you, not in front of you. That’s why I am not happy.”
Overall, quantitative and qualitative findings indicate multiple, detrimental effects of personal stigma on PwD. These effects fit well with measures of self-stigma, including social rejection (eg, being treated differently, participating in fewer activities, and having fewer friends), internalized shame (eg, being treated like a child, having fewer responsibilities, others acting as if dementia is “contagious”), and social isolation (eg, being less outgoing, feeling more comfortable in small groups, having limited social contacts).15
Continue to: Receiving a diagnosis of dementia...
Receiving a diagnosis of dementia presents patients and their families with psychological and social challenges.16 Many of these challenges are the consequence of stigma. A broad range of efforts are underway worldwide to reduce dementia-related stigma. These efforts include programs to promote public awareness and education, campaigns to develop inclusive social policies, and skills-based training initiatives to promote delivery of patient-centered care by clinicians and educators.3,17,18 Many of these efforts share a common focus on promoting the “dignity” and “personhood” of PwD in order to disrupt stereotypes or fixed, oversimplified beliefs associated with dementia.
Implementing person-centered clinical care
In clinical practice, direct discussion that encourages reflection and the use of effective and sensitive communication can help to limit passing on stigmatizing beliefs and to reduce negative stereotypes associated with the disease. Health care communications that call attention to stereotypes may allow PwD to identify stereotypes as well as inaccuracies in those stereotypes. Interventions that validate the value of diversity can help PwD accept the ways in which they may not conform to social norms. This could include language such as “There is no one way to have Alzheimer’s disease. A person’s experience can differ from what others might experience or expect, and that’s okay.” In addition, the use of language that is accurate, respectful, inclusive, and empowering can support PwD and their caregivers.19,20 For example, referring to PwD as “individuals living with dementia” rather than “those who are demented” conveys respect and appreciation for personhood. Other clinicians have provided additional practical suggestions.21
Anti-stigma messaging campaigns
The mass media is a common source of stereotypes about AD and other dementias. They typically present a “worst-case” scenario that promotes ageism, gerontophobia, and negative emotions, which may worsen stigma and discrimination towards PwD and the people who care for them. However, public messaging campaigns are emerging to counter negative messages and stereotypes in the mass media. Projects such as Typical Day, People with Dementia, and other online anti-stigma messaging campaigns allow a broad audience to gain a more nuanced understanding of the lives of PwD and their caregivers. These projects are rich resources that offer education and personal stories that can counter common stereotypes about dementia.
Typical Day is a photography project developed and maintained by clinicians and researchers at the University of Pennsylvania. Since early 2017, the project has provided a forum for individuals with mild cognitive impairment or dementia to document their lives and show what it means to them to live with dementia. Participants in the project photo-document the people, places, and objects that define their daily lives. They review and explain these photos with researchers at Penn Memory Center, who help them tell their stories. The participants’ stories, the photos they capture, and their portraits are available at www.mytypicalday.org.
People of Dementia. Storytelling is a powerful way to raise awareness of and reduce the stigma associated with dementia. For PwD, telling their stories can be an effective and therapeutic way to communicate their emotions and deliver an important message. In the blog People of Dementia (www.peopleofdementia.com),22,23 PwD highlight who they were before the disease and how things have changed, with family members highlighting the challenges of caring for a person with dementia.
Continue to: The common thread is...
The common thread is the enduring “person” behind the exterior that is obscured by dementia. By allowing the audience to form a connection with who the individual was prior to the disease, and understanding the changes that have come as a result of dementia to both PwD and their support network, readers gain a greater appreciation of those affected by dementia. Between May 1, 2017 and May 31, 2019, the blog had more than 3,860 visitors. In an accompanying online survey (N = 57), 79% of respondents agreed/strongly agreed that after visiting the People of Dementia blog, they had a better understanding of the changes that occur as a result of cognitive impairment/dementia (Figure 1). Almost two-thirds of respondents (65%) agreed/strongly agreed that they felt more comfortable interacting with PwD (Figure 2). Additionally, 60% of respondents agreed/strongly agreed that they were more encouraged to work with PwD, and 90% agreed/strongly agreed that they had a greater appreciation of the challenges of being a caregiver for PwD. Overall, these findings suggest that the People of Dementia blog is useful for engaging the public and promoting a better understanding of dementia.
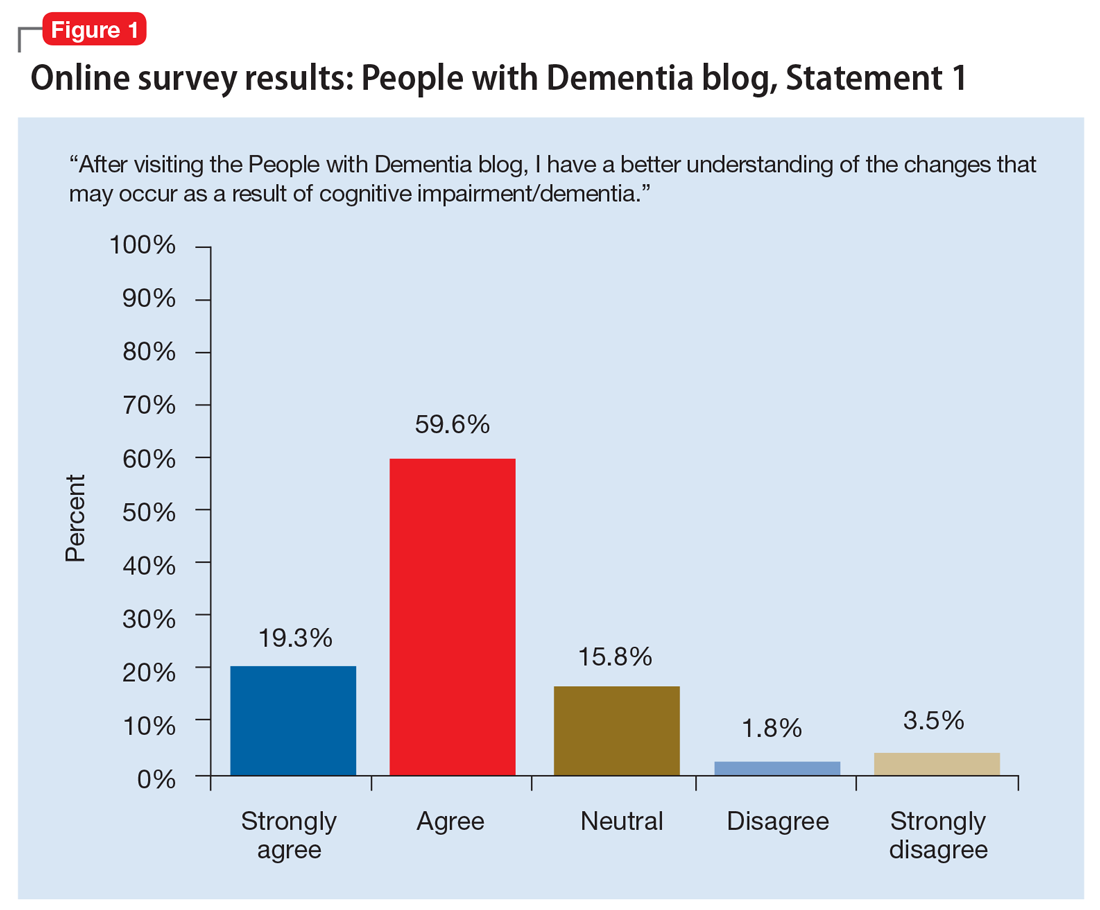
Work for policy changes
Clinicians can support public policy through education and advocacy both in the delivery of care and as spokespersons and stakeholders in their local communities. Public policies are important for providing access to medical and social services to meet the needs of PwD and their caregivers. The absence—real or perceived—of sufficient resources exacerbates dementia-related stigma. In addition to facilitating access to resources, national dementia strategies or legal frameworks, such as the National Alzheimer’s Project Act in the United States, include policy initiatives to identify and promote communication approaches that are effective and sensitive with respect to people living with dementia and their caregivers.
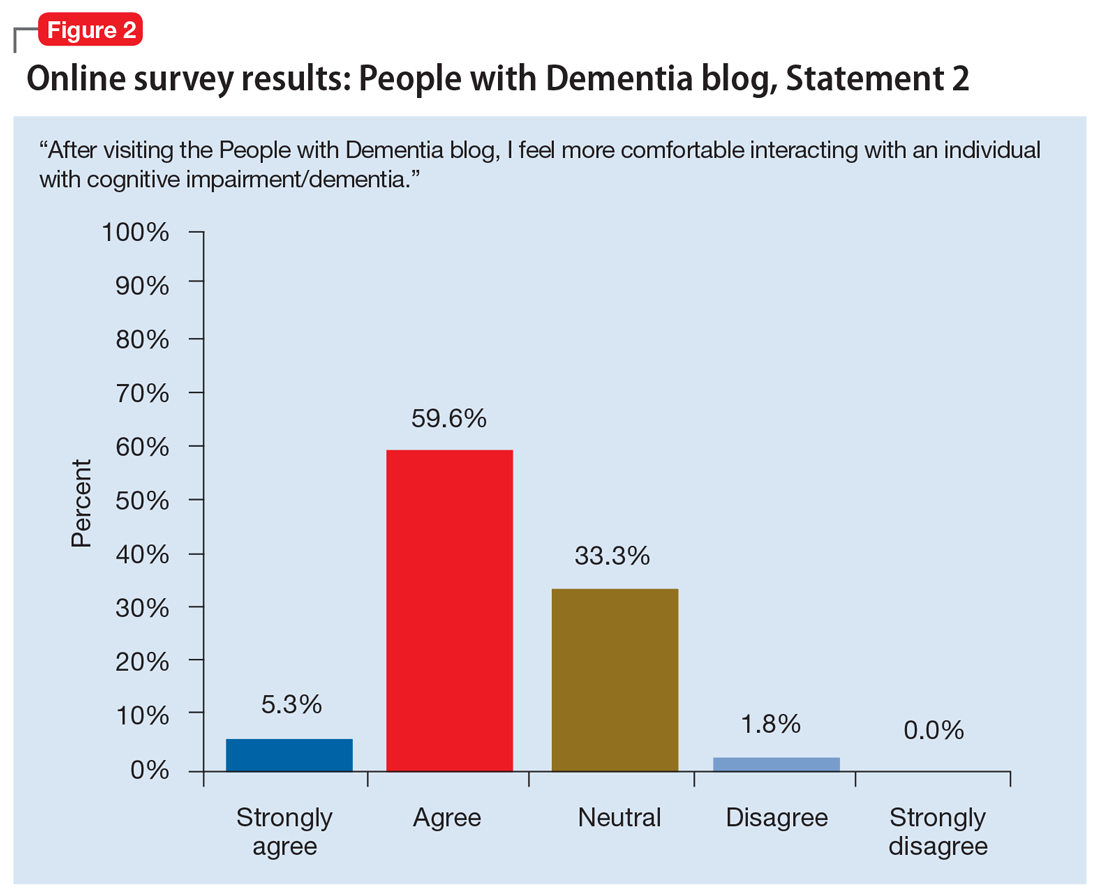
State and local legislators and patient advocates are leading policy efforts to reduce dementia-related stigma. For example, Colorado recently changed statutory references from being specific to diseases that cause dementia to the broader, more inclusive phrase “dementia diseases and related disabilities.”18 In addition to making funds available to support caregiving services for PwD, this legislative change added training for first responders to better meet the needs of missing PwD, and shifted the terminology used to diagnose and communicate about diseases causing dementia. The shift in language added new terminology that was chosen for being more person-centered to replace prior references to “senior senility,” “senility,” and other terms with pejorative meanings.
In Canada, a National Dementia Strategy will commit the Canadian government to action with definitive timelines, targets, reporting structures, and measurable outcomes.24
Table 2 summarizes approaches to a

Continue to: An open discussion
An open discussion
Larger studies and testing of diverse approaches are needed to better understand whether intergenerational initiatives or other approaches can genuinely modify stigmatizing attitudes in various dementia populations, especially considering language, health literacy, cultural preferences, and other needs. The identified effects on physical and mental health, quality of life, self-esteem, and behavioral symptoms further support the extensive, negative effects of self-stigma on PwD, and emphasize the need to develop and test interventions to ameliorate these effects.
We presented at a Stigma Symposium at the 2018 Gerontological Society of America Annual Scientific Meeting in Boston, Massachusetts.25 Attendees of this conference shared our concerns about the detrimental effects of stigma. The main question we were asked was “What can we do to reduce stigma?” Perhaps the most immediate response is that in order to move the stigma dial, clinicians need to recognize that stigma has multiple, broad-reaching, and negative effects on PwD and their families.6 Bringing the discussion into the open and targeting stigma at multiple levels needs to be addressed by clinicians, researchers, administrators, and society at large.
Bottom Line
Stigma has multiple, broad-reaching, and negative effects on persons with dementia and their families. In clinical practice, direct discussion that encourages reflection and the use of effective and sensitive communication can help to limit passing on stigmatizing beliefs and to reduce negative stereotypes associated with the disease. Anti-stigma messaging campaigns and public policy changes also can be used to address societal and social inequities of patients with dementia and their caregivers.
Related Resources
- Khoury R, Shach R, Nair A, et al. Can lifestyle modifications delay or prevent Alzheimer’s disease? Current Psychiatry. 2019;18(1):29-36,38.
- Burke AD, Burke WJ. Antipsychotics for patients with dementia: The road less traveled. Current Psychiatry. 2018;17(10):26-32,35-37.
1. World Health Organization. Towards a dementia plan: a WHO guide. https://www.who.int/mental_health/neurology/dementia/policy_guidance/en/. Published 2018. Accessed May 28, 2019.
2. Goffman E. Stigma. New York, NY: Prentice-Hall; 1963:1-123.
3. Alzheimer’s Disease International. World Alzheimer Report 2012: overcoming the stigma of dementia. https://www.alz.co.uk/research/WorldAlzheimerReport2012.pdf. Published 2012. Accessed May 28, 2019.
4. Blay SL, Peluso ETP. Public stigma: the community’s tolerance of Alzheimer disease. Am J Geriatr Psychiatry. 2010;18(2):163-171.
5. Piver LC, Nubukpo P, Faure A, et al. Describing perceived stigma against Alzheimer’s disease in a general population in France: the STIG-MA survey. Int J Geriatr Psychiatry. 2013;28(9):933-938.
6. Herrmann LK, Welter E, Leverenz J, et al. A systematic review of dementia-related stigma research: can we move the stigma dial? Am J Geriatr Psychiatry. 2018;26(3):316-331.
7. Eng KJ, Woo BKP. Knowledge of dementia community resources and stigma among Chinese American immigrants. Gen Hosp Psychiatry. 2015;37(1):e3-e4. doi:10.1016/j.genhosppsych.2014.11.003.
8. Jang Y, Kim G, Chiriboga D. Knowledge of Alzheimer’s disease, feelings of shame, and awareness of services among Korean American elders. J Aging Health. 2010;22(4):419-433.
9. Werner P, Goldstein D, Heinik J. Development and validity of the Family Stigma in Alzheimer’s disease scale (FS-ADS). Alzheimer Disease & Associated Disorders. 2011;25(1):42-48.
10. Rao D, Choi SW, Victorson D, et al. Measuring stigma across neurological conditions: the development of the stigma scale for chronic illness (SSCI). Qual Life Res. 2009;18(5):585-595.
11. Boustani M, Perkins AJ, Monahan P, et al. Measuring primary care patients’ attitudes about dementia screening. Int J Geriatr Psychiatry. 2008;23(8):812-820.
12. Burgener SC, Buckwalter K, Perkounkova Y, et al. Perceived stigma in persons with early-stage dementia: longitudinal findings: Part 2. Dementia. 2015;14(5):609-632.
13. Burgener SC, Buckwalter K. The effects of perceived stigma on persons with dementia and their family caregivers. In: Symposium on Stigma: It’s time to talk about it. Boston, MA: Gerontological Society of America 2018 Annual Scientific Meeting; 2018. Session 2805.
14. Harper L, Dobbs B, Royan H, et al. The experience of stigma in care partners of people with dementia – results from an exploratory study. In Symposium on stigma: it’s time to talk about it. Boston, MA: Gerontological Society of America 2018 Annual Scientific Meeting; 2018. Session 2805.
15. Burgener S, Berger B. Measuring perceived stigma in persons with progressive neurological disease: Alzheimer’s dementia and Parkinson disease. Dementia. 2008;7(1):31-53.
16. Stites SD, Milne R, Karlawish J. Advances in Alzheimer’s imaging are changing the experience of Alzheimer’s disease. Alzheimer’s & Dementia. 2018;10;285-300.
17. Anderson LA, Egge R. Expanding efforts to address Alzheimer’s disease: the Healthy Brain Initiative. Alzheimer’s Dement. 2014;10(50):S453-S456.
18. Alzheimer’s Association National Plan Milestone Workgroup. Report on the milestones for the US National plan to address Alzheimer’s disease. Alzheimer’s Dementia. 2014;10(Suppl 5);S430-S452. doi:10.1016/j/jalz.2014.08.103.
19. Kirkman AM. Dementia in the news: the media coverage of Alzheimer’s disease. Australasian Journal on Ageing. 2006;25(2):74-79.
20. Swaffer, K. Dementia: stigma, language, and dementia-friendly. Dementia. 2014;13(6):709-716.
21. Stites SD, Karlawish J. Stigma of Alzheimer’s disease dementia: considerations for practice. Practical Neurology. https://practicalneurology.com/articles/2018-june/stigma-of-alzheimers-disease-dementia. Published June 2018. Accessed May 28, 2019.
22. Jamieson J, Dobbs B, Charles L, et al. Forgetful, but not forgotten people of dementia: a novel, technology focused project with a humanistic touch. Geriatric Grand Rounds; October 10, 2017. Edmonton, Alberta, Canada.
23. Dobbs B, Charles L, Chan K, et al. People of Dementia. CGS 37th Annual Scientific Meeting: Integrating Care, Making an Impact. Can Geriatr J. 2017;20(3):220.
24. Government of Canada. Conference report: National Dementia Conference. https://www.canada.ca/en/services/health/publications/diseases-conditions/national-dementia-conference-report.html. Government of Canada. Published August 2018. Accessed May 28, 2019.
25. The Gerontological Society of America. Program Abstracts from the GSA 2018 Annual Scientific Meeting “The Purposes of Longer Lives.” Innovation in Aging. 2018;2(Suppl 1):143.
Dementia is a family of disorders characterized by a decline in multiple cognitive abilities that significantly interferes with an individual’s functioning. An estimated 50 million people are living with a dementia worldwide.1 Alzheimer’s disease (AD) is the leading cause of dementia, accounting for approximately two-thirds of dementia cases.1 These numbers are expected to increase dramatically in the upcoming decades.
Sociologist Erving Goffman defined stigma as “an attribute, behaviour, or reputation which is socially discrediting in a particular way: it causes an individual to be mentally classified by others in an undesirable, rejected stereotype rather than in an accepted, normal one.”2 Goffman2 defined 3 broad categories of stigma: public, self, and courtesy (Table 12).

Considerable evidence shows that the combined impact of having dementia and the negative response to the diagnosis significantly undermines an individual’s psychosocial well-being and quality of life.3 Persons with dementia (PwD) commonly report a loss of identity and self-worth, and stigma appears to deepen this distress.3 Stigma also negatively affects individuals associated with PwD, including family members and professionals. In this article, we discuss the impact of dementia-related stigma, and steps you can take to address it, including implementing person-centered clinical practices, promoting anti-stigma messaging campaigns, and advocating for public policy action to improve the lives of PwD and their families.
A pervasive problem
Although the Alzheimer’s Society International and the World Health Organization acknowledge that stigma has a central role in defining the experience of AD, how stigma may present, how clinicians and researchers can recognize and measure stigma, and how to best combat it have been understudied.3-5 A recent systematic literature review examined worldwide evidence on dementia-related stigma over the past decade.6 Hermann et al6 found that health care providers and the general public may hold stigmatizing attitudes toward PwD, and that stigma may be particularly harsh among racial and ethnic minorities, although the literature is scarce in this area. Cultural factors may also worsen stigma, and stigma may be associated with reduced awareness of dementia services and reduced help-seeking among minority groups.7,8 Studies show that stigmatizing attitudes are more pronounced in people with limited knowledge of dementia, in those with little contact with PwD, in men, in younger individuals, and in the context of cultural interpretations of dementia.6 Health care providers can also sometimes contribute to the perpetuation of stigma.6
In terms of standardized scales or instruments for evaluating dementia-related stigma, there is no uniformly accepted “gold standard” measure, which makes it difficult to compare studies.6 In order to effectively study efforts to reduce stigma, researchers need to identify and establish a consensus on rating scales for evaluating stigma among PwD, caregivers, and the general public. Three instruments that may be used for this purpose are the Family Stigma in Alzheimer’s Disease Scale (FS-ADS),9 the Stigma Scale for Chronic Illness (SSCI),10 and the Perceptions Regarding Investigational Screening for Memory in Primary Care (PRISM-PC).11
The detrimental effects of stigma
Burgener et al12 reported that personal stigma impacted functioning and quality of life in PwD. Higher levels of stigma were associated with higher anxiety, depression, and behavioral symptoms and lower self-esteem, social support, participation in activities, personal control, and physical health.12 Personal characteristics that may affect stigma include gender, location (rural vs urban), ethnicity, education level, and living arrangements (alone vs with family).12
In a subset of PwD with early-stage memory loss (n = 22), Burgener and Buckwalter13 found that 42% of participants were reluctant to reveal their diagnosis to others, with some fearing they would no longer be allowed to live alone and would be “sent to a facility.” In addition, 46% indicated they did not want “to be talked about like they were not there.” More than 50% of participants reported changes in their social network after receiving the diagnosis, including reducing activities and limiting types of contacts (ie, telephone only) or interacting only when “people come to me.” Participants were most comfortable with good friends “who understand” and persons within their faith communities. When asked about how they were treated by family members, >50% of participants described being treated differently, including loss of financial independence, more limited contact, and being “treated like a baby” by their children, who in general were uncomfortable talking about the diagnosis.
Continue to: In a recent study...
In a recent study by Harper et al,14 stigma was prevalent in the experience of PwD. One participant disclosed:
“I think there is [are] people I know who don’t ask me to go places or do things ’cause I have a dementia…I think lots of people don’t know what dementia is and I think it scares them ’cause they think of it as crazy. It hurts…”
Another participant said:
“I have had friends for over thirty years. They have turned their backs on me…we used to go for walks and they would phone me and go for coffee. Now I don’t hear from any of them…those aren’t true friends…true friends will stand behind you, not in front of you. That’s why I am not happy.”
Overall, quantitative and qualitative findings indicate multiple, detrimental effects of personal stigma on PwD. These effects fit well with measures of self-stigma, including social rejection (eg, being treated differently, participating in fewer activities, and having fewer friends), internalized shame (eg, being treated like a child, having fewer responsibilities, others acting as if dementia is “contagious”), and social isolation (eg, being less outgoing, feeling more comfortable in small groups, having limited social contacts).15
Continue to: Receiving a diagnosis of dementia...
Receiving a diagnosis of dementia presents patients and their families with psychological and social challenges.16 Many of these challenges are the consequence of stigma. A broad range of efforts are underway worldwide to reduce dementia-related stigma. These efforts include programs to promote public awareness and education, campaigns to develop inclusive social policies, and skills-based training initiatives to promote delivery of patient-centered care by clinicians and educators.3,17,18 Many of these efforts share a common focus on promoting the “dignity” and “personhood” of PwD in order to disrupt stereotypes or fixed, oversimplified beliefs associated with dementia.
Implementing person-centered clinical care
In clinical practice, direct discussion that encourages reflection and the use of effective and sensitive communication can help to limit passing on stigmatizing beliefs and to reduce negative stereotypes associated with the disease. Health care communications that call attention to stereotypes may allow PwD to identify stereotypes as well as inaccuracies in those stereotypes. Interventions that validate the value of diversity can help PwD accept the ways in which they may not conform to social norms. This could include language such as “There is no one way to have Alzheimer’s disease. A person’s experience can differ from what others might experience or expect, and that’s okay.” In addition, the use of language that is accurate, respectful, inclusive, and empowering can support PwD and their caregivers.19,20 For example, referring to PwD as “individuals living with dementia” rather than “those who are demented” conveys respect and appreciation for personhood. Other clinicians have provided additional practical suggestions.21
Anti-stigma messaging campaigns
The mass media is a common source of stereotypes about AD and other dementias. They typically present a “worst-case” scenario that promotes ageism, gerontophobia, and negative emotions, which may worsen stigma and discrimination towards PwD and the people who care for them. However, public messaging campaigns are emerging to counter negative messages and stereotypes in the mass media. Projects such as Typical Day, People with Dementia, and other online anti-stigma messaging campaigns allow a broad audience to gain a more nuanced understanding of the lives of PwD and their caregivers. These projects are rich resources that offer education and personal stories that can counter common stereotypes about dementia.
Typical Day is a photography project developed and maintained by clinicians and researchers at the University of Pennsylvania. Since early 2017, the project has provided a forum for individuals with mild cognitive impairment or dementia to document their lives and show what it means to them to live with dementia. Participants in the project photo-document the people, places, and objects that define their daily lives. They review and explain these photos with researchers at Penn Memory Center, who help them tell their stories. The participants’ stories, the photos they capture, and their portraits are available at www.mytypicalday.org.
People of Dementia. Storytelling is a powerful way to raise awareness of and reduce the stigma associated with dementia. For PwD, telling their stories can be an effective and therapeutic way to communicate their emotions and deliver an important message. In the blog People of Dementia (www.peopleofdementia.com),22,23 PwD highlight who they were before the disease and how things have changed, with family members highlighting the challenges of caring for a person with dementia.
Continue to: The common thread is...
The common thread is the enduring “person” behind the exterior that is obscured by dementia. By allowing the audience to form a connection with who the individual was prior to the disease, and understanding the changes that have come as a result of dementia to both PwD and their support network, readers gain a greater appreciation of those affected by dementia. Between May 1, 2017 and May 31, 2019, the blog had more than 3,860 visitors. In an accompanying online survey (N = 57), 79% of respondents agreed/strongly agreed that after visiting the People of Dementia blog, they had a better understanding of the changes that occur as a result of cognitive impairment/dementia (Figure 1). Almost two-thirds of respondents (65%) agreed/strongly agreed that they felt more comfortable interacting with PwD (Figure 2). Additionally, 60% of respondents agreed/strongly agreed that they were more encouraged to work with PwD, and 90% agreed/strongly agreed that they had a greater appreciation of the challenges of being a caregiver for PwD. Overall, these findings suggest that the People of Dementia blog is useful for engaging the public and promoting a better understanding of dementia.

Work for policy changes
Clinicians can support public policy through education and advocacy both in the delivery of care and as spokespersons and stakeholders in their local communities. Public policies are important for providing access to medical and social services to meet the needs of PwD and their caregivers. The absence—real or perceived—of sufficient resources exacerbates dementia-related stigma. In addition to facilitating access to resources, national dementia strategies or legal frameworks, such as the National Alzheimer’s Project Act in the United States, include policy initiatives to identify and promote communication approaches that are effective and sensitive with respect to people living with dementia and their caregivers.

State and local legislators and patient advocates are leading policy efforts to reduce dementia-related stigma. For example, Colorado recently changed statutory references from being specific to diseases that cause dementia to the broader, more inclusive phrase “dementia diseases and related disabilities.”18 In addition to making funds available to support caregiving services for PwD, this legislative change added training for first responders to better meet the needs of missing PwD, and shifted the terminology used to diagnose and communicate about diseases causing dementia. The shift in language added new terminology that was chosen for being more person-centered to replace prior references to “senior senility,” “senility,” and other terms with pejorative meanings.
In Canada, a National Dementia Strategy will commit the Canadian government to action with definitive timelines, targets, reporting structures, and measurable outcomes.24
Table 2 summarizes approaches to a

Continue to: An open discussion
An open discussion
Larger studies and testing of diverse approaches are needed to better understand whether intergenerational initiatives or other approaches can genuinely modify stigmatizing attitudes in various dementia populations, especially considering language, health literacy, cultural preferences, and other needs. The identified effects on physical and mental health, quality of life, self-esteem, and behavioral symptoms further support the extensive, negative effects of self-stigma on PwD, and emphasize the need to develop and test interventions to ameliorate these effects.
We presented at a Stigma Symposium at the 2018 Gerontological Society of America Annual Scientific Meeting in Boston, Massachusetts.25 Attendees of this conference shared our concerns about the detrimental effects of stigma. The main question we were asked was “What can we do to reduce stigma?” Perhaps the most immediate response is that in order to move the stigma dial, clinicians need to recognize that stigma has multiple, broad-reaching, and negative effects on PwD and their families.6 Bringing the discussion into the open and targeting stigma at multiple levels needs to be addressed by clinicians, researchers, administrators, and society at large.
Bottom Line
Stigma has multiple, broad-reaching, and negative effects on persons with dementia and their families. In clinical practice, direct discussion that encourages reflection and the use of effective and sensitive communication can help to limit passing on stigmatizing beliefs and to reduce negative stereotypes associated with the disease. Anti-stigma messaging campaigns and public policy changes also can be used to address societal and social inequities of patients with dementia and their caregivers.
Related Resources
- Khoury R, Shach R, Nair A, et al. Can lifestyle modifications delay or prevent Alzheimer’s disease? Current Psychiatry. 2019;18(1):29-36,38.
- Burke AD, Burke WJ. Antipsychotics for patients with dementia: The road less traveled. Current Psychiatry. 2018;17(10):26-32,35-37.
Dementia is a family of disorders characterized by a decline in multiple cognitive abilities that significantly interferes with an individual’s functioning. An estimated 50 million people are living with a dementia worldwide.1 Alzheimer’s disease (AD) is the leading cause of dementia, accounting for approximately two-thirds of dementia cases.1 These numbers are expected to increase dramatically in the upcoming decades.
Sociologist Erving Goffman defined stigma as “an attribute, behaviour, or reputation which is socially discrediting in a particular way: it causes an individual to be mentally classified by others in an undesirable, rejected stereotype rather than in an accepted, normal one.”2 Goffman2 defined 3 broad categories of stigma: public, self, and courtesy (Table 12).

Considerable evidence shows that the combined impact of having dementia and the negative response to the diagnosis significantly undermines an individual’s psychosocial well-being and quality of life.3 Persons with dementia (PwD) commonly report a loss of identity and self-worth, and stigma appears to deepen this distress.3 Stigma also negatively affects individuals associated with PwD, including family members and professionals. In this article, we discuss the impact of dementia-related stigma, and steps you can take to address it, including implementing person-centered clinical practices, promoting anti-stigma messaging campaigns, and advocating for public policy action to improve the lives of PwD and their families.
A pervasive problem
Although the Alzheimer’s Society International and the World Health Organization acknowledge that stigma has a central role in defining the experience of AD, how stigma may present, how clinicians and researchers can recognize and measure stigma, and how to best combat it have been understudied.3-5 A recent systematic literature review examined worldwide evidence on dementia-related stigma over the past decade.6 Hermann et al6 found that health care providers and the general public may hold stigmatizing attitudes toward PwD, and that stigma may be particularly harsh among racial and ethnic minorities, although the literature is scarce in this area. Cultural factors may also worsen stigma, and stigma may be associated with reduced awareness of dementia services and reduced help-seeking among minority groups.7,8 Studies show that stigmatizing attitudes are more pronounced in people with limited knowledge of dementia, in those with little contact with PwD, in men, in younger individuals, and in the context of cultural interpretations of dementia.6 Health care providers can also sometimes contribute to the perpetuation of stigma.6
In terms of standardized scales or instruments for evaluating dementia-related stigma, there is no uniformly accepted “gold standard” measure, which makes it difficult to compare studies.6 In order to effectively study efforts to reduce stigma, researchers need to identify and establish a consensus on rating scales for evaluating stigma among PwD, caregivers, and the general public. Three instruments that may be used for this purpose are the Family Stigma in Alzheimer’s Disease Scale (FS-ADS),9 the Stigma Scale for Chronic Illness (SSCI),10 and the Perceptions Regarding Investigational Screening for Memory in Primary Care (PRISM-PC).11
The detrimental effects of stigma
Burgener et al12 reported that personal stigma impacted functioning and quality of life in PwD. Higher levels of stigma were associated with higher anxiety, depression, and behavioral symptoms and lower self-esteem, social support, participation in activities, personal control, and physical health.12 Personal characteristics that may affect stigma include gender, location (rural vs urban), ethnicity, education level, and living arrangements (alone vs with family).12
In a subset of PwD with early-stage memory loss (n = 22), Burgener and Buckwalter13 found that 42% of participants were reluctant to reveal their diagnosis to others, with some fearing they would no longer be allowed to live alone and would be “sent to a facility.” In addition, 46% indicated they did not want “to be talked about like they were not there.” More than 50% of participants reported changes in their social network after receiving the diagnosis, including reducing activities and limiting types of contacts (ie, telephone only) or interacting only when “people come to me.” Participants were most comfortable with good friends “who understand” and persons within their faith communities. When asked about how they were treated by family members, >50% of participants described being treated differently, including loss of financial independence, more limited contact, and being “treated like a baby” by their children, who in general were uncomfortable talking about the diagnosis.
Continue to: In a recent study...
In a recent study by Harper et al,14 stigma was prevalent in the experience of PwD. One participant disclosed:
“I think there is [are] people I know who don’t ask me to go places or do things ’cause I have a dementia…I think lots of people don’t know what dementia is and I think it scares them ’cause they think of it as crazy. It hurts…”
Another participant said:
“I have had friends for over thirty years. They have turned their backs on me…we used to go for walks and they would phone me and go for coffee. Now I don’t hear from any of them…those aren’t true friends…true friends will stand behind you, not in front of you. That’s why I am not happy.”
Overall, quantitative and qualitative findings indicate multiple, detrimental effects of personal stigma on PwD. These effects fit well with measures of self-stigma, including social rejection (eg, being treated differently, participating in fewer activities, and having fewer friends), internalized shame (eg, being treated like a child, having fewer responsibilities, others acting as if dementia is “contagious”), and social isolation (eg, being less outgoing, feeling more comfortable in small groups, having limited social contacts).15
Continue to: Receiving a diagnosis of dementia...
Receiving a diagnosis of dementia presents patients and their families with psychological and social challenges.16 Many of these challenges are the consequence of stigma. A broad range of efforts are underway worldwide to reduce dementia-related stigma. These efforts include programs to promote public awareness and education, campaigns to develop inclusive social policies, and skills-based training initiatives to promote delivery of patient-centered care by clinicians and educators.3,17,18 Many of these efforts share a common focus on promoting the “dignity” and “personhood” of PwD in order to disrupt stereotypes or fixed, oversimplified beliefs associated with dementia.
Implementing person-centered clinical care
In clinical practice, direct discussion that encourages reflection and the use of effective and sensitive communication can help to limit passing on stigmatizing beliefs and to reduce negative stereotypes associated with the disease. Health care communications that call attention to stereotypes may allow PwD to identify stereotypes as well as inaccuracies in those stereotypes. Interventions that validate the value of diversity can help PwD accept the ways in which they may not conform to social norms. This could include language such as “There is no one way to have Alzheimer’s disease. A person’s experience can differ from what others might experience or expect, and that’s okay.” In addition, the use of language that is accurate, respectful, inclusive, and empowering can support PwD and their caregivers.19,20 For example, referring to PwD as “individuals living with dementia” rather than “those who are demented” conveys respect and appreciation for personhood. Other clinicians have provided additional practical suggestions.21
Anti-stigma messaging campaigns
The mass media is a common source of stereotypes about AD and other dementias. They typically present a “worst-case” scenario that promotes ageism, gerontophobia, and negative emotions, which may worsen stigma and discrimination towards PwD and the people who care for them. However, public messaging campaigns are emerging to counter negative messages and stereotypes in the mass media. Projects such as Typical Day, People with Dementia, and other online anti-stigma messaging campaigns allow a broad audience to gain a more nuanced understanding of the lives of PwD and their caregivers. These projects are rich resources that offer education and personal stories that can counter common stereotypes about dementia.
Typical Day is a photography project developed and maintained by clinicians and researchers at the University of Pennsylvania. Since early 2017, the project has provided a forum for individuals with mild cognitive impairment or dementia to document their lives and show what it means to them to live with dementia. Participants in the project photo-document the people, places, and objects that define their daily lives. They review and explain these photos with researchers at Penn Memory Center, who help them tell their stories. The participants’ stories, the photos they capture, and their portraits are available at www.mytypicalday.org.
People of Dementia. Storytelling is a powerful way to raise awareness of and reduce the stigma associated with dementia. For PwD, telling their stories can be an effective and therapeutic way to communicate their emotions and deliver an important message. In the blog People of Dementia (www.peopleofdementia.com),22,23 PwD highlight who they were before the disease and how things have changed, with family members highlighting the challenges of caring for a person with dementia.
Continue to: The common thread is...
The common thread is the enduring “person” behind the exterior that is obscured by dementia. By allowing the audience to form a connection with who the individual was prior to the disease, and understanding the changes that have come as a result of dementia to both PwD and their support network, readers gain a greater appreciation of those affected by dementia. Between May 1, 2017 and May 31, 2019, the blog had more than 3,860 visitors. In an accompanying online survey (N = 57), 79% of respondents agreed/strongly agreed that after visiting the People of Dementia blog, they had a better understanding of the changes that occur as a result of cognitive impairment/dementia (Figure 1). Almost two-thirds of respondents (65%) agreed/strongly agreed that they felt more comfortable interacting with PwD (Figure 2). Additionally, 60% of respondents agreed/strongly agreed that they were more encouraged to work with PwD, and 90% agreed/strongly agreed that they had a greater appreciation of the challenges of being a caregiver for PwD. Overall, these findings suggest that the People of Dementia blog is useful for engaging the public and promoting a better understanding of dementia.

Work for policy changes
Clinicians can support public policy through education and advocacy both in the delivery of care and as spokespersons and stakeholders in their local communities. Public policies are important for providing access to medical and social services to meet the needs of PwD and their caregivers. The absence—real or perceived—of sufficient resources exacerbates dementia-related stigma. In addition to facilitating access to resources, national dementia strategies or legal frameworks, such as the National Alzheimer’s Project Act in the United States, include policy initiatives to identify and promote communication approaches that are effective and sensitive with respect to people living with dementia and their caregivers.

State and local legislators and patient advocates are leading policy efforts to reduce dementia-related stigma. For example, Colorado recently changed statutory references from being specific to diseases that cause dementia to the broader, more inclusive phrase “dementia diseases and related disabilities.”18 In addition to making funds available to support caregiving services for PwD, this legislative change added training for first responders to better meet the needs of missing PwD, and shifted the terminology used to diagnose and communicate about diseases causing dementia. The shift in language added new terminology that was chosen for being more person-centered to replace prior references to “senior senility,” “senility,” and other terms with pejorative meanings.
In Canada, a National Dementia Strategy will commit the Canadian government to action with definitive timelines, targets, reporting structures, and measurable outcomes.24
Table 2 summarizes approaches to a

Continue to: An open discussion
An open discussion
Larger studies and testing of diverse approaches are needed to better understand whether intergenerational initiatives or other approaches can genuinely modify stigmatizing attitudes in various dementia populations, especially considering language, health literacy, cultural preferences, and other needs. The identified effects on physical and mental health, quality of life, self-esteem, and behavioral symptoms further support the extensive, negative effects of self-stigma on PwD, and emphasize the need to develop and test interventions to ameliorate these effects.
We presented at a Stigma Symposium at the 2018 Gerontological Society of America Annual Scientific Meeting in Boston, Massachusetts.25 Attendees of this conference shared our concerns about the detrimental effects of stigma. The main question we were asked was “What can we do to reduce stigma?” Perhaps the most immediate response is that in order to move the stigma dial, clinicians need to recognize that stigma has multiple, broad-reaching, and negative effects on PwD and their families.6 Bringing the discussion into the open and targeting stigma at multiple levels needs to be addressed by clinicians, researchers, administrators, and society at large.
Bottom Line
Stigma has multiple, broad-reaching, and negative effects on persons with dementia and their families. In clinical practice, direct discussion that encourages reflection and the use of effective and sensitive communication can help to limit passing on stigmatizing beliefs and to reduce negative stereotypes associated with the disease. Anti-stigma messaging campaigns and public policy changes also can be used to address societal and social inequities of patients with dementia and their caregivers.
Related Resources
- Khoury R, Shach R, Nair A, et al. Can lifestyle modifications delay or prevent Alzheimer’s disease? Current Psychiatry. 2019;18(1):29-36,38.
- Burke AD, Burke WJ. Antipsychotics for patients with dementia: The road less traveled. Current Psychiatry. 2018;17(10):26-32,35-37.
1. World Health Organization. Towards a dementia plan: a WHO guide. https://www.who.int/mental_health/neurology/dementia/policy_guidance/en/. Published 2018. Accessed May 28, 2019.
2. Goffman E. Stigma. New York, NY: Prentice-Hall; 1963:1-123.
3. Alzheimer’s Disease International. World Alzheimer Report 2012: overcoming the stigma of dementia. https://www.alz.co.uk/research/WorldAlzheimerReport2012.pdf. Published 2012. Accessed May 28, 2019.
4. Blay SL, Peluso ETP. Public stigma: the community’s tolerance of Alzheimer disease. Am J Geriatr Psychiatry. 2010;18(2):163-171.
5. Piver LC, Nubukpo P, Faure A, et al. Describing perceived stigma against Alzheimer’s disease in a general population in France: the STIG-MA survey. Int J Geriatr Psychiatry. 2013;28(9):933-938.
6. Herrmann LK, Welter E, Leverenz J, et al. A systematic review of dementia-related stigma research: can we move the stigma dial? Am J Geriatr Psychiatry. 2018;26(3):316-331.
7. Eng KJ, Woo BKP. Knowledge of dementia community resources and stigma among Chinese American immigrants. Gen Hosp Psychiatry. 2015;37(1):e3-e4. doi:10.1016/j.genhosppsych.2014.11.003.
8. Jang Y, Kim G, Chiriboga D. Knowledge of Alzheimer’s disease, feelings of shame, and awareness of services among Korean American elders. J Aging Health. 2010;22(4):419-433.
9. Werner P, Goldstein D, Heinik J. Development and validity of the Family Stigma in Alzheimer’s disease scale (FS-ADS). Alzheimer Disease & Associated Disorders. 2011;25(1):42-48.
10. Rao D, Choi SW, Victorson D, et al. Measuring stigma across neurological conditions: the development of the stigma scale for chronic illness (SSCI). Qual Life Res. 2009;18(5):585-595.
11. Boustani M, Perkins AJ, Monahan P, et al. Measuring primary care patients’ attitudes about dementia screening. Int J Geriatr Psychiatry. 2008;23(8):812-820.
12. Burgener SC, Buckwalter K, Perkounkova Y, et al. Perceived stigma in persons with early-stage dementia: longitudinal findings: Part 2. Dementia. 2015;14(5):609-632.
13. Burgener SC, Buckwalter K. The effects of perceived stigma on persons with dementia and their family caregivers. In: Symposium on Stigma: It’s time to talk about it. Boston, MA: Gerontological Society of America 2018 Annual Scientific Meeting; 2018. Session 2805.
14. Harper L, Dobbs B, Royan H, et al. The experience of stigma in care partners of people with dementia – results from an exploratory study. In Symposium on stigma: it’s time to talk about it. Boston, MA: Gerontological Society of America 2018 Annual Scientific Meeting; 2018. Session 2805.
15. Burgener S, Berger B. Measuring perceived stigma in persons with progressive neurological disease: Alzheimer’s dementia and Parkinson disease. Dementia. 2008;7(1):31-53.
16. Stites SD, Milne R, Karlawish J. Advances in Alzheimer’s imaging are changing the experience of Alzheimer’s disease. Alzheimer’s & Dementia. 2018;10;285-300.
17. Anderson LA, Egge R. Expanding efforts to address Alzheimer’s disease: the Healthy Brain Initiative. Alzheimer’s Dement. 2014;10(50):S453-S456.
18. Alzheimer’s Association National Plan Milestone Workgroup. Report on the milestones for the US National plan to address Alzheimer’s disease. Alzheimer’s Dementia. 2014;10(Suppl 5);S430-S452. doi:10.1016/j/jalz.2014.08.103.
19. Kirkman AM. Dementia in the news: the media coverage of Alzheimer’s disease. Australasian Journal on Ageing. 2006;25(2):74-79.
20. Swaffer, K. Dementia: stigma, language, and dementia-friendly. Dementia. 2014;13(6):709-716.
21. Stites SD, Karlawish J. Stigma of Alzheimer’s disease dementia: considerations for practice. Practical Neurology. https://practicalneurology.com/articles/2018-june/stigma-of-alzheimers-disease-dementia. Published June 2018. Accessed May 28, 2019.
22. Jamieson J, Dobbs B, Charles L, et al. Forgetful, but not forgotten people of dementia: a novel, technology focused project with a humanistic touch. Geriatric Grand Rounds; October 10, 2017. Edmonton, Alberta, Canada.
23. Dobbs B, Charles L, Chan K, et al. People of Dementia. CGS 37th Annual Scientific Meeting: Integrating Care, Making an Impact. Can Geriatr J. 2017;20(3):220.
24. Government of Canada. Conference report: National Dementia Conference. https://www.canada.ca/en/services/health/publications/diseases-conditions/national-dementia-conference-report.html. Government of Canada. Published August 2018. Accessed May 28, 2019.
25. The Gerontological Society of America. Program Abstracts from the GSA 2018 Annual Scientific Meeting “The Purposes of Longer Lives.” Innovation in Aging. 2018;2(Suppl 1):143.
1. World Health Organization. Towards a dementia plan: a WHO guide. https://www.who.int/mental_health/neurology/dementia/policy_guidance/en/. Published 2018. Accessed May 28, 2019.
2. Goffman E. Stigma. New York, NY: Prentice-Hall; 1963:1-123.
3. Alzheimer’s Disease International. World Alzheimer Report 2012: overcoming the stigma of dementia. https://www.alz.co.uk/research/WorldAlzheimerReport2012.pdf. Published 2012. Accessed May 28, 2019.
4. Blay SL, Peluso ETP. Public stigma: the community’s tolerance of Alzheimer disease. Am J Geriatr Psychiatry. 2010;18(2):163-171.
5. Piver LC, Nubukpo P, Faure A, et al. Describing perceived stigma against Alzheimer’s disease in a general population in France: the STIG-MA survey. Int J Geriatr Psychiatry. 2013;28(9):933-938.
6. Herrmann LK, Welter E, Leverenz J, et al. A systematic review of dementia-related stigma research: can we move the stigma dial? Am J Geriatr Psychiatry. 2018;26(3):316-331.
7. Eng KJ, Woo BKP. Knowledge of dementia community resources and stigma among Chinese American immigrants. Gen Hosp Psychiatry. 2015;37(1):e3-e4. doi:10.1016/j.genhosppsych.2014.11.003.
8. Jang Y, Kim G, Chiriboga D. Knowledge of Alzheimer’s disease, feelings of shame, and awareness of services among Korean American elders. J Aging Health. 2010;22(4):419-433.
9. Werner P, Goldstein D, Heinik J. Development and validity of the Family Stigma in Alzheimer’s disease scale (FS-ADS). Alzheimer Disease & Associated Disorders. 2011;25(1):42-48.
10. Rao D, Choi SW, Victorson D, et al. Measuring stigma across neurological conditions: the development of the stigma scale for chronic illness (SSCI). Qual Life Res. 2009;18(5):585-595.
11. Boustani M, Perkins AJ, Monahan P, et al. Measuring primary care patients’ attitudes about dementia screening. Int J Geriatr Psychiatry. 2008;23(8):812-820.
12. Burgener SC, Buckwalter K, Perkounkova Y, et al. Perceived stigma in persons with early-stage dementia: longitudinal findings: Part 2. Dementia. 2015;14(5):609-632.
13. Burgener SC, Buckwalter K. The effects of perceived stigma on persons with dementia and their family caregivers. In: Symposium on Stigma: It’s time to talk about it. Boston, MA: Gerontological Society of America 2018 Annual Scientific Meeting; 2018. Session 2805.
14. Harper L, Dobbs B, Royan H, et al. The experience of stigma in care partners of people with dementia – results from an exploratory study. In Symposium on stigma: it’s time to talk about it. Boston, MA: Gerontological Society of America 2018 Annual Scientific Meeting; 2018. Session 2805.
15. Burgener S, Berger B. Measuring perceived stigma in persons with progressive neurological disease: Alzheimer’s dementia and Parkinson disease. Dementia. 2008;7(1):31-53.
16. Stites SD, Milne R, Karlawish J. Advances in Alzheimer’s imaging are changing the experience of Alzheimer’s disease. Alzheimer’s & Dementia. 2018;10;285-300.
17. Anderson LA, Egge R. Expanding efforts to address Alzheimer’s disease: the Healthy Brain Initiative. Alzheimer’s Dement. 2014;10(50):S453-S456.
18. Alzheimer’s Association National Plan Milestone Workgroup. Report on the milestones for the US National plan to address Alzheimer’s disease. Alzheimer’s Dementia. 2014;10(Suppl 5);S430-S452. doi:10.1016/j/jalz.2014.08.103.
19. Kirkman AM. Dementia in the news: the media coverage of Alzheimer’s disease. Australasian Journal on Ageing. 2006;25(2):74-79.
20. Swaffer, K. Dementia: stigma, language, and dementia-friendly. Dementia. 2014;13(6):709-716.
21. Stites SD, Karlawish J. Stigma of Alzheimer’s disease dementia: considerations for practice. Practical Neurology. https://practicalneurology.com/articles/2018-june/stigma-of-alzheimers-disease-dementia. Published June 2018. Accessed May 28, 2019.
22. Jamieson J, Dobbs B, Charles L, et al. Forgetful, but not forgotten people of dementia: a novel, technology focused project with a humanistic touch. Geriatric Grand Rounds; October 10, 2017. Edmonton, Alberta, Canada.
23. Dobbs B, Charles L, Chan K, et al. People of Dementia. CGS 37th Annual Scientific Meeting: Integrating Care, Making an Impact. Can Geriatr J. 2017;20(3):220.
24. Government of Canada. Conference report: National Dementia Conference. https://www.canada.ca/en/services/health/publications/diseases-conditions/national-dementia-conference-report.html. Government of Canada. Published August 2018. Accessed May 28, 2019.
25. The Gerontological Society of America. Program Abstracts from the GSA 2018 Annual Scientific Meeting “The Purposes of Longer Lives.” Innovation in Aging. 2018;2(Suppl 1):143.
Automated measurements of plasma predict amyloid status
, according to research published online ahead of print June 24 in JAMA Neurology. Analyzing APOE genotype in addition to these biomarkers increases the accuracy of the prediction. This blood test thus could allow neurologists to identify patients at risk of amyloid-beta positivity who should undergo further assessment, said the authors. It also could be used to enroll amyloid-beta–positive participants in clinical trials.
In vivo PET imaging and analysis of cerebrospinal fluid (CSF) can detect amyloid-beta, but these procedures are expensive, and their availability is limited. Clinicians need readily available methods for detecting amyloid-beta, and research has indicated that blood-based biomarkers correlate with those in CSF. Fully automated immunoassays, such as the Elecsys test developed by Roche Diagnostics, have recently demonstrated high reliability and precision for CSF amyloid-beta. Using the Elecsys assay, Sebastian Palmqvist, MD, PhD, a neurologist at Skåne University Hospital in Malmö, Sweden, and colleagues sought to examine the accuracy of plasma amyloid-beta and tau, together with other blood-based biomarkers, at detecting cerebral amyloid-beta.
Testing the immunoassay in two cohorts
Dr. Palmqvist and colleagues examined participants in the prospective Swedish BioFINDER Study, which enrolled patients between July 6, 2009, and February 11, 2015. This cohort included 513 cognitively unimpaired (CU) participants, 265 participants with mild cognitive impairment (MCI), and 64 participants with Alzheimer’s disease dementia. Investigators collected blood and CSF samples at the same time from all participants. Participants’ amyloid-beta status was ascertained using the Elecsys CSF amyloid-beta 42/amyloid-beta 40 ratio. The researchers defined amyloid-beta positivity with an unbiased cutoff of less than 0.059.
Dr. Palmqvist and colleagues also examined a validation cohort that included 237 participants who had been enrolled between January 29, 2000, and October 11, 2006, in Ulm and Hannover, Germany. This group included 34 CU participants, 109 participants with MCI, and 94 participants with mild Alzheimer’s disease dementia. The investigators applied the same cutoff of CSF amyloid-beta 42/amyloid-beta 40 to define amyloid-beta positivity in this cohort as they applied to the BioFINDER cohort.
Automated immunoassay had high predictive accuracy
The mean age of the BioFINDER cohort was 72 years, and 52.5% of participants were female. Overall, 44% of this cohort was amyloid-beta positive, including 29% of CU participants, 60% of participants with MCI, and 100% of participants with Alzheimer’s dementia. The investigators found statistically significant positive correlations between all plasma and corresponding CSF biomarkers in this cohort.
Plasma amyloid-beta 42 and amyloid-beta 40 levels predicted amyloid-beta status with an area under the receiver operating characteristic curve (AUC) of 0.80. When the researchers added APOE to the model, the AUC increased significantly to 0.85. Accuracy improved slightly when the researchers added plasma tau (AUC, 0.86) or tau and neurofilament light (AUC, 0.87) to amyloid-beta 42, amyloid-beta 40, and APOE. The results were similar in CU and cognitively impaired participants, and in younger and older participants.
In the validation cohort, the mean age was 66 years, and 50.6% of participants were female. When Dr. Palmqvist and colleagues applied the plasma amyloid-beta 42 and amyloid-beta 40 model from the BioFINDER cohort to this population, they obtained a slightly higher AUC (0.86), but plasma tau did not increase predictive accuracy.
The investigators performed a cost-benefit analysis using a scenario in which 1,000 amyloid-positive participants are included in a trial and given a cost of $4,000 per participant for amyloid PET. Using plasma amyloid-beta 42, amyloid-beta 40, and APOE in this scenario reduced PET costs by as much as 30%-50%, depending on the cutoff.
Validation cohort was small
Dr. Palmqvist and colleagues acknowledged that a lack of data about APOE was a limitation of their validation analysis. Other limitations that they acknowledged were the small population size, which precluded subpopulation analysis, and the lack of improvement in predictive ability when they replicated the model that included plasma tau.
“Overall, the accuracies of the amyloid-beta 42 and amyloid-beta 40 assays are not sufficient to be used on their own as a clinical test of amyloid-beta positivity,” said Dr. Palmqvist and colleagues. “Additional assay development is needed before this can be recommended, possibly together with other blood biomarkers and screening tools in diagnostic algorithms.”
Even though additional validation studies are necessary, the present findings indicate “the potential usefulness blood assays might have, especially considering the ongoing great need to recruit large cohorts for Alzheimer’s disease drug trials in preclinical and prodromal stages,” the authors concluded.
This investigation was funded by foundations including the European Research Council, the Swedish Research Council, and the Knut and Alice Wallenberg foundation. Several authors are employees of the Roche Group. One author served on a scientific advisory board for Roche Diagnostics, and another received institutional research support from that company.
SOURCE: Palmqvist S et al. JAMA Neurol. 2019 Jun 24. doi: 10.1001/jamaneurol.2019.1632.
The investigation by Palmqvist et al. “makes several significant advancements in the field,” said Sid E. O’Bryant, PhD, professor of pharmacology and neuroscience at the University of North Texas Health Science Center in Fort Worth, in an accompanying editorial. The study’s protocol design clears the ground for a context of use of a blood screen for amyloid positivity. Also, the fully automated immunoassay “yields performance measurements that are superior to [those of] many earlier nonautomated procedures,” said Dr. O’Bryant. When Dr. Palmqvist and colleagues applied their discovery findings from a training cohort directly to a test cohort, it produced strong results. “This study suggests that the field is one step closer to the actual application of blood-based biomarkers with specific contexts of use in Alzheimer’s disease.”
The main concern about the plasma biomarkers, however, is the scalability of the methods used to measure them. “If primary care physicians are to use such a technology, the technology must have the capacity to conduct hundreds of millions of assays annually around the globe,” said Dr. O’Bryant. “A blood test for primary care must fit into the existing protocols and parameters in clinical laboratory settings. The blood collection and processing procedures are not applicable to standard clinical lab practice and will cause substantial barriers to clinical application.”
In addition, the study authors emphasize the utility of the immunoassay for primary care, but the study was designed to test for amyloid positivity, which is more appropriate for clinical trials. “No currently available drugs for patient use target amyloid,” said Dr. O’Bryant. “Therefore, this specific context of use is geared more toward clinical trial application than primary care physicians who currently need a test for the presence or absence of Alzheimer’s disease so currently available treatments and support can be put in place for patients and family members.”
Nevertheless, Dr. Palmqvist and associates have presented promising data, Dr. O’Bryant continued. The question in the field is ceasing to be whether blood biomarkers can be used in Alzheimer’s disease, and becoming how they can be used.
The investigation by Palmqvist et al. “makes several significant advancements in the field,” said Sid E. O’Bryant, PhD, professor of pharmacology and neuroscience at the University of North Texas Health Science Center in Fort Worth, in an accompanying editorial. The study’s protocol design clears the ground for a context of use of a blood screen for amyloid positivity. Also, the fully automated immunoassay “yields performance measurements that are superior to [those of] many earlier nonautomated procedures,” said Dr. O’Bryant. When Dr. Palmqvist and colleagues applied their discovery findings from a training cohort directly to a test cohort, it produced strong results. “This study suggests that the field is one step closer to the actual application of blood-based biomarkers with specific contexts of use in Alzheimer’s disease.”
The main concern about the plasma biomarkers, however, is the scalability of the methods used to measure them. “If primary care physicians are to use such a technology, the technology must have the capacity to conduct hundreds of millions of assays annually around the globe,” said Dr. O’Bryant. “A blood test for primary care must fit into the existing protocols and parameters in clinical laboratory settings. The blood collection and processing procedures are not applicable to standard clinical lab practice and will cause substantial barriers to clinical application.”
In addition, the study authors emphasize the utility of the immunoassay for primary care, but the study was designed to test for amyloid positivity, which is more appropriate for clinical trials. “No currently available drugs for patient use target amyloid,” said Dr. O’Bryant. “Therefore, this specific context of use is geared more toward clinical trial application than primary care physicians who currently need a test for the presence or absence of Alzheimer’s disease so currently available treatments and support can be put in place for patients and family members.”
Nevertheless, Dr. Palmqvist and associates have presented promising data, Dr. O’Bryant continued. The question in the field is ceasing to be whether blood biomarkers can be used in Alzheimer’s disease, and becoming how they can be used.
The investigation by Palmqvist et al. “makes several significant advancements in the field,” said Sid E. O’Bryant, PhD, professor of pharmacology and neuroscience at the University of North Texas Health Science Center in Fort Worth, in an accompanying editorial. The study’s protocol design clears the ground for a context of use of a blood screen for amyloid positivity. Also, the fully automated immunoassay “yields performance measurements that are superior to [those of] many earlier nonautomated procedures,” said Dr. O’Bryant. When Dr. Palmqvist and colleagues applied their discovery findings from a training cohort directly to a test cohort, it produced strong results. “This study suggests that the field is one step closer to the actual application of blood-based biomarkers with specific contexts of use in Alzheimer’s disease.”
The main concern about the plasma biomarkers, however, is the scalability of the methods used to measure them. “If primary care physicians are to use such a technology, the technology must have the capacity to conduct hundreds of millions of assays annually around the globe,” said Dr. O’Bryant. “A blood test for primary care must fit into the existing protocols and parameters in clinical laboratory settings. The blood collection and processing procedures are not applicable to standard clinical lab practice and will cause substantial barriers to clinical application.”
In addition, the study authors emphasize the utility of the immunoassay for primary care, but the study was designed to test for amyloid positivity, which is more appropriate for clinical trials. “No currently available drugs for patient use target amyloid,” said Dr. O’Bryant. “Therefore, this specific context of use is geared more toward clinical trial application than primary care physicians who currently need a test for the presence or absence of Alzheimer’s disease so currently available treatments and support can be put in place for patients and family members.”
Nevertheless, Dr. Palmqvist and associates have presented promising data, Dr. O’Bryant continued. The question in the field is ceasing to be whether blood biomarkers can be used in Alzheimer’s disease, and becoming how they can be used.
, according to research published online ahead of print June 24 in JAMA Neurology. Analyzing APOE genotype in addition to these biomarkers increases the accuracy of the prediction. This blood test thus could allow neurologists to identify patients at risk of amyloid-beta positivity who should undergo further assessment, said the authors. It also could be used to enroll amyloid-beta–positive participants in clinical trials.
In vivo PET imaging and analysis of cerebrospinal fluid (CSF) can detect amyloid-beta, but these procedures are expensive, and their availability is limited. Clinicians need readily available methods for detecting amyloid-beta, and research has indicated that blood-based biomarkers correlate with those in CSF. Fully automated immunoassays, such as the Elecsys test developed by Roche Diagnostics, have recently demonstrated high reliability and precision for CSF amyloid-beta. Using the Elecsys assay, Sebastian Palmqvist, MD, PhD, a neurologist at Skåne University Hospital in Malmö, Sweden, and colleagues sought to examine the accuracy of plasma amyloid-beta and tau, together with other blood-based biomarkers, at detecting cerebral amyloid-beta.
Testing the immunoassay in two cohorts
Dr. Palmqvist and colleagues examined participants in the prospective Swedish BioFINDER Study, which enrolled patients between July 6, 2009, and February 11, 2015. This cohort included 513 cognitively unimpaired (CU) participants, 265 participants with mild cognitive impairment (MCI), and 64 participants with Alzheimer’s disease dementia. Investigators collected blood and CSF samples at the same time from all participants. Participants’ amyloid-beta status was ascertained using the Elecsys CSF amyloid-beta 42/amyloid-beta 40 ratio. The researchers defined amyloid-beta positivity with an unbiased cutoff of less than 0.059.
Dr. Palmqvist and colleagues also examined a validation cohort that included 237 participants who had been enrolled between January 29, 2000, and October 11, 2006, in Ulm and Hannover, Germany. This group included 34 CU participants, 109 participants with MCI, and 94 participants with mild Alzheimer’s disease dementia. The investigators applied the same cutoff of CSF amyloid-beta 42/amyloid-beta 40 to define amyloid-beta positivity in this cohort as they applied to the BioFINDER cohort.
Automated immunoassay had high predictive accuracy
The mean age of the BioFINDER cohort was 72 years, and 52.5% of participants were female. Overall, 44% of this cohort was amyloid-beta positive, including 29% of CU participants, 60% of participants with MCI, and 100% of participants with Alzheimer’s dementia. The investigators found statistically significant positive correlations between all plasma and corresponding CSF biomarkers in this cohort.
Plasma amyloid-beta 42 and amyloid-beta 40 levels predicted amyloid-beta status with an area under the receiver operating characteristic curve (AUC) of 0.80. When the researchers added APOE to the model, the AUC increased significantly to 0.85. Accuracy improved slightly when the researchers added plasma tau (AUC, 0.86) or tau and neurofilament light (AUC, 0.87) to amyloid-beta 42, amyloid-beta 40, and APOE. The results were similar in CU and cognitively impaired participants, and in younger and older participants.
In the validation cohort, the mean age was 66 years, and 50.6% of participants were female. When Dr. Palmqvist and colleagues applied the plasma amyloid-beta 42 and amyloid-beta 40 model from the BioFINDER cohort to this population, they obtained a slightly higher AUC (0.86), but plasma tau did not increase predictive accuracy.
The investigators performed a cost-benefit analysis using a scenario in which 1,000 amyloid-positive participants are included in a trial and given a cost of $4,000 per participant for amyloid PET. Using plasma amyloid-beta 42, amyloid-beta 40, and APOE in this scenario reduced PET costs by as much as 30%-50%, depending on the cutoff.
Validation cohort was small
Dr. Palmqvist and colleagues acknowledged that a lack of data about APOE was a limitation of their validation analysis. Other limitations that they acknowledged were the small population size, which precluded subpopulation analysis, and the lack of improvement in predictive ability when they replicated the model that included plasma tau.
“Overall, the accuracies of the amyloid-beta 42 and amyloid-beta 40 assays are not sufficient to be used on their own as a clinical test of amyloid-beta positivity,” said Dr. Palmqvist and colleagues. “Additional assay development is needed before this can be recommended, possibly together with other blood biomarkers and screening tools in diagnostic algorithms.”
Even though additional validation studies are necessary, the present findings indicate “the potential usefulness blood assays might have, especially considering the ongoing great need to recruit large cohorts for Alzheimer’s disease drug trials in preclinical and prodromal stages,” the authors concluded.
This investigation was funded by foundations including the European Research Council, the Swedish Research Council, and the Knut and Alice Wallenberg foundation. Several authors are employees of the Roche Group. One author served on a scientific advisory board for Roche Diagnostics, and another received institutional research support from that company.
SOURCE: Palmqvist S et al. JAMA Neurol. 2019 Jun 24. doi: 10.1001/jamaneurol.2019.1632.
, according to research published online ahead of print June 24 in JAMA Neurology. Analyzing APOE genotype in addition to these biomarkers increases the accuracy of the prediction. This blood test thus could allow neurologists to identify patients at risk of amyloid-beta positivity who should undergo further assessment, said the authors. It also could be used to enroll amyloid-beta–positive participants in clinical trials.
In vivo PET imaging and analysis of cerebrospinal fluid (CSF) can detect amyloid-beta, but these procedures are expensive, and their availability is limited. Clinicians need readily available methods for detecting amyloid-beta, and research has indicated that blood-based biomarkers correlate with those in CSF. Fully automated immunoassays, such as the Elecsys test developed by Roche Diagnostics, have recently demonstrated high reliability and precision for CSF amyloid-beta. Using the Elecsys assay, Sebastian Palmqvist, MD, PhD, a neurologist at Skåne University Hospital in Malmö, Sweden, and colleagues sought to examine the accuracy of plasma amyloid-beta and tau, together with other blood-based biomarkers, at detecting cerebral amyloid-beta.
Testing the immunoassay in two cohorts
Dr. Palmqvist and colleagues examined participants in the prospective Swedish BioFINDER Study, which enrolled patients between July 6, 2009, and February 11, 2015. This cohort included 513 cognitively unimpaired (CU) participants, 265 participants with mild cognitive impairment (MCI), and 64 participants with Alzheimer’s disease dementia. Investigators collected blood and CSF samples at the same time from all participants. Participants’ amyloid-beta status was ascertained using the Elecsys CSF amyloid-beta 42/amyloid-beta 40 ratio. The researchers defined amyloid-beta positivity with an unbiased cutoff of less than 0.059.
Dr. Palmqvist and colleagues also examined a validation cohort that included 237 participants who had been enrolled between January 29, 2000, and October 11, 2006, in Ulm and Hannover, Germany. This group included 34 CU participants, 109 participants with MCI, and 94 participants with mild Alzheimer’s disease dementia. The investigators applied the same cutoff of CSF amyloid-beta 42/amyloid-beta 40 to define amyloid-beta positivity in this cohort as they applied to the BioFINDER cohort.
Automated immunoassay had high predictive accuracy
The mean age of the BioFINDER cohort was 72 years, and 52.5% of participants were female. Overall, 44% of this cohort was amyloid-beta positive, including 29% of CU participants, 60% of participants with MCI, and 100% of participants with Alzheimer’s dementia. The investigators found statistically significant positive correlations between all plasma and corresponding CSF biomarkers in this cohort.
Plasma amyloid-beta 42 and amyloid-beta 40 levels predicted amyloid-beta status with an area under the receiver operating characteristic curve (AUC) of 0.80. When the researchers added APOE to the model, the AUC increased significantly to 0.85. Accuracy improved slightly when the researchers added plasma tau (AUC, 0.86) or tau and neurofilament light (AUC, 0.87) to amyloid-beta 42, amyloid-beta 40, and APOE. The results were similar in CU and cognitively impaired participants, and in younger and older participants.
In the validation cohort, the mean age was 66 years, and 50.6% of participants were female. When Dr. Palmqvist and colleagues applied the plasma amyloid-beta 42 and amyloid-beta 40 model from the BioFINDER cohort to this population, they obtained a slightly higher AUC (0.86), but plasma tau did not increase predictive accuracy.
The investigators performed a cost-benefit analysis using a scenario in which 1,000 amyloid-positive participants are included in a trial and given a cost of $4,000 per participant for amyloid PET. Using plasma amyloid-beta 42, amyloid-beta 40, and APOE in this scenario reduced PET costs by as much as 30%-50%, depending on the cutoff.
Validation cohort was small
Dr. Palmqvist and colleagues acknowledged that a lack of data about APOE was a limitation of their validation analysis. Other limitations that they acknowledged were the small population size, which precluded subpopulation analysis, and the lack of improvement in predictive ability when they replicated the model that included plasma tau.
“Overall, the accuracies of the amyloid-beta 42 and amyloid-beta 40 assays are not sufficient to be used on their own as a clinical test of amyloid-beta positivity,” said Dr. Palmqvist and colleagues. “Additional assay development is needed before this can be recommended, possibly together with other blood biomarkers and screening tools in diagnostic algorithms.”
Even though additional validation studies are necessary, the present findings indicate “the potential usefulness blood assays might have, especially considering the ongoing great need to recruit large cohorts for Alzheimer’s disease drug trials in preclinical and prodromal stages,” the authors concluded.
This investigation was funded by foundations including the European Research Council, the Swedish Research Council, and the Knut and Alice Wallenberg foundation. Several authors are employees of the Roche Group. One author served on a scientific advisory board for Roche Diagnostics, and another received institutional research support from that company.
SOURCE: Palmqvist S et al. JAMA Neurol. 2019 Jun 24. doi: 10.1001/jamaneurol.2019.1632.
FROM JAMA NEUROLOGY












