User login
Even light drinking ups CV risk; harm rises along with intake
Even very light alcohol intake is associated with an increased risk for cardiovascular disease, compared with not drinking at all, and the risk increases exponentially as alcohol intake rises, even at moderate levels, a new study shows.
“Our findings suggest that the observed benefit in individuals with light to moderate alcohol intake, which is consistently shown in epidemiological studies, is likely due to other positive lifestyle factors that are common in these individuals who drink lightly,” senior author Krishna Aragam, MD, Massachusetts General Hospital, Boston, told this news organization.
“Our results also showed that while all levels of alcohol were linked to increased risk of cardiovascular disease, the association was not linear. Rather, light alcohol intake was associated with rather modest risk increases, but there were exponential increases in cardiovascular risk with increasing amounts of alcohol consumption,” he said.
As the risk gradient appeared to increase quite sharply even between 1 and 2 drinks per day, Dr. Aragam suggested that what might be regarded as safe levels of drinking may trend downward in the future.
The study was published online March 25 in JAMA Network Open.
The cohort study used data from the UK Biobank, collected between 2006 and 2010 with follow-up until 2016, to assess the relationship between various levels of alcohol consumption and risk for cardiovascular disease.
Data were analyzed from 371,463 participants (mean age, 57 years; 46% men) who consumed an average of 9.2 standard drinks per week. Of these participants, 33% had hypertension and 7.5% had coronary artery disease.
“Use of the UK biobank database gives the advantage of a large, well-phenotyped population with a lot of information on various lifestyle factors that could be potential confounders,” Dr. Aragam noted.
Results showed that well-established J- or U-shaped curves were seen for the association between alcohol consumption and both the prevalence and hazards of hypertension, coronary artery disease, myocardial infarction, stroke, heart failure, and atrial fibrillation.
However, individuals in the light and moderate consumption group had healthier lifestyle behaviors than abstainers, self-reporting better overall health and exhibiting lower rates of smoking, lower body mass index, higher physical activity, and higher vegetable intake.
Adjustment for these lifestyle factors attenuated the cardioprotective associations with modest alcohol intake. For example, in baseline models, moderate intake was associated with significantly lower risk of hypertension and coronary artery disease, but adjustment for just six lifestyle factors rendered these results insignificant.
“Adjustments for yet unmeasured or unknown factors may further attenuate, if not eliminate, the residual, cardioprotective associations observed among light drinkers,” the researchers suggest.
They also conducted genetic analyses to examine the effect of alcohol and cardiovascular disease.
Dr. Aragam explained that previous work has shown good evidence, in individuals who choose to drink, that several relevant genetic variants predict levels of alcohol consumption quite accurately.
“Mendelian randomization using these gene variants allows for stronger inferences about potential causality than do observational studies, as they are less affected by confounding factors,” he noted.
Newer techniques in Mendelian randomization in which data on several gene variants linked to alcohol consumption are combined into a score allow for a greater understanding of the risk linked to different amount of alcohol intake, he added.
In these Mendelian randomization analyses, a 1-standard deviation increase in genetically predicted alcohol consumption was associated with 1.3-fold higher risk of hypertension (P < .001) and 1.4-fold higher risk of coronary artery disease (P = .006).
Further analyses suggested nonlinear associations between alcohol consumption and both hypertension and coronary artery disease; light alcohol intake was associated with minimal increases in cardiovascular risk, whereas heavier consumption was associated with exponential increases in risk of both clinical and subclinical cardiovascular disease.
These results were replicated in a second database of 30,716 individuals from the Mass General Brigham Biobank.
“The findings of this study suggest that the observed cardioprotective effects of light to moderate alcohol intake may be largely mediated by confounding lifestyle factors,” the researchers conclude. “Genetic analyses suggest causal associations between alcohol intake and cardiovascular disease but with unequal and exponential increases in risk at greater levels of intake, which should be accounted for in health recommendations around the habitual consumption of alcohol.”
What is an acceptable level?
“Specifically, our results suggest that consuming as many as 7 drinks per week is associated with relatively modest increases in cardiovascular risk,” they write.
But they point out that there are unequal increases in cardiovascular risk when progressing from 0 to 7 versus 7 to 14 drinks per week in both men and women.
“Although risk thresholds are inherently somewhat subjective, these findings again bring into question whether an average consumption of 2 drinks per day (14 drinks per week) should be designated a low-risk behavior,” they say.
“Furthermore, as several-fold increases in risk were observed for those consuming 21 or more drinks per week, our results emphasize the importance of aggressive efforts to reduce alcohol intake among heavy drinkers,” they add.
Dr. Aragam elaborated: “Our data suggest that reducing alcohol intake will reduce cardiovascular risk in all individuals, but the extent of the relative risk reduction is quite different depending on the current levels of consumption. For the same absolute reduction in alcohol intake, the gains in terms of reduction in cardiovascular risk will be more pronounced in those who drink heavily and will be more modest in those who drink at a light level.”
The results also suggest that while all levels of alcohol intake increase cardiovascular risk, there are low levels of alcohol consumption that do not carry major elevations in risk, but these are probably lower than those currently recommended, Dr. Aragam pointed out.
“This doesn’t mean that everyone has to give up drinking alcohol completely, just that you shouldn’t consume with the goal of improving cardiovascular health. In fact, our analyses suggest that in an otherwise healthy person, up to 1 drink per day may not pose outsized risks,” he said. “And, even in a less healthy person who might be smoking, eating poorly, and drinking up to 1 drink per day, it may be a higher priority to focus on smoking cessation and diet than cutting back further on alcohol.”
“Beyond that amount, though, the jury is still out. Our models suggested marked increases in risk even between 1 and 2 drinks per day, and of course even greater risk increases beyond that. So, it’s probably worth revisiting what one might consider a ‘safe’ amount within the moderate drinking categories. The conservative move for now might be to advise a limit of 1 drink per day,” he said.
Dr. Aragam is supported by grants from the National Institutes of Health and the American Heart Association. He reports receiving speaking fees from the Novartis Institute for Biomedical Research.
A version of this article first appeared on Medscape.com.
Even very light alcohol intake is associated with an increased risk for cardiovascular disease, compared with not drinking at all, and the risk increases exponentially as alcohol intake rises, even at moderate levels, a new study shows.
“Our findings suggest that the observed benefit in individuals with light to moderate alcohol intake, which is consistently shown in epidemiological studies, is likely due to other positive lifestyle factors that are common in these individuals who drink lightly,” senior author Krishna Aragam, MD, Massachusetts General Hospital, Boston, told this news organization.
“Our results also showed that while all levels of alcohol were linked to increased risk of cardiovascular disease, the association was not linear. Rather, light alcohol intake was associated with rather modest risk increases, but there were exponential increases in cardiovascular risk with increasing amounts of alcohol consumption,” he said.
As the risk gradient appeared to increase quite sharply even between 1 and 2 drinks per day, Dr. Aragam suggested that what might be regarded as safe levels of drinking may trend downward in the future.
The study was published online March 25 in JAMA Network Open.
The cohort study used data from the UK Biobank, collected between 2006 and 2010 with follow-up until 2016, to assess the relationship between various levels of alcohol consumption and risk for cardiovascular disease.
Data were analyzed from 371,463 participants (mean age, 57 years; 46% men) who consumed an average of 9.2 standard drinks per week. Of these participants, 33% had hypertension and 7.5% had coronary artery disease.
“Use of the UK biobank database gives the advantage of a large, well-phenotyped population with a lot of information on various lifestyle factors that could be potential confounders,” Dr. Aragam noted.
Results showed that well-established J- or U-shaped curves were seen for the association between alcohol consumption and both the prevalence and hazards of hypertension, coronary artery disease, myocardial infarction, stroke, heart failure, and atrial fibrillation.
However, individuals in the light and moderate consumption group had healthier lifestyle behaviors than abstainers, self-reporting better overall health and exhibiting lower rates of smoking, lower body mass index, higher physical activity, and higher vegetable intake.
Adjustment for these lifestyle factors attenuated the cardioprotective associations with modest alcohol intake. For example, in baseline models, moderate intake was associated with significantly lower risk of hypertension and coronary artery disease, but adjustment for just six lifestyle factors rendered these results insignificant.
“Adjustments for yet unmeasured or unknown factors may further attenuate, if not eliminate, the residual, cardioprotective associations observed among light drinkers,” the researchers suggest.
They also conducted genetic analyses to examine the effect of alcohol and cardiovascular disease.
Dr. Aragam explained that previous work has shown good evidence, in individuals who choose to drink, that several relevant genetic variants predict levels of alcohol consumption quite accurately.
“Mendelian randomization using these gene variants allows for stronger inferences about potential causality than do observational studies, as they are less affected by confounding factors,” he noted.
Newer techniques in Mendelian randomization in which data on several gene variants linked to alcohol consumption are combined into a score allow for a greater understanding of the risk linked to different amount of alcohol intake, he added.
In these Mendelian randomization analyses, a 1-standard deviation increase in genetically predicted alcohol consumption was associated with 1.3-fold higher risk of hypertension (P < .001) and 1.4-fold higher risk of coronary artery disease (P = .006).
Further analyses suggested nonlinear associations between alcohol consumption and both hypertension and coronary artery disease; light alcohol intake was associated with minimal increases in cardiovascular risk, whereas heavier consumption was associated with exponential increases in risk of both clinical and subclinical cardiovascular disease.
These results were replicated in a second database of 30,716 individuals from the Mass General Brigham Biobank.
“The findings of this study suggest that the observed cardioprotective effects of light to moderate alcohol intake may be largely mediated by confounding lifestyle factors,” the researchers conclude. “Genetic analyses suggest causal associations between alcohol intake and cardiovascular disease but with unequal and exponential increases in risk at greater levels of intake, which should be accounted for in health recommendations around the habitual consumption of alcohol.”
What is an acceptable level?
“Specifically, our results suggest that consuming as many as 7 drinks per week is associated with relatively modest increases in cardiovascular risk,” they write.
But they point out that there are unequal increases in cardiovascular risk when progressing from 0 to 7 versus 7 to 14 drinks per week in both men and women.
“Although risk thresholds are inherently somewhat subjective, these findings again bring into question whether an average consumption of 2 drinks per day (14 drinks per week) should be designated a low-risk behavior,” they say.
“Furthermore, as several-fold increases in risk were observed for those consuming 21 or more drinks per week, our results emphasize the importance of aggressive efforts to reduce alcohol intake among heavy drinkers,” they add.
Dr. Aragam elaborated: “Our data suggest that reducing alcohol intake will reduce cardiovascular risk in all individuals, but the extent of the relative risk reduction is quite different depending on the current levels of consumption. For the same absolute reduction in alcohol intake, the gains in terms of reduction in cardiovascular risk will be more pronounced in those who drink heavily and will be more modest in those who drink at a light level.”
The results also suggest that while all levels of alcohol intake increase cardiovascular risk, there are low levels of alcohol consumption that do not carry major elevations in risk, but these are probably lower than those currently recommended, Dr. Aragam pointed out.
“This doesn’t mean that everyone has to give up drinking alcohol completely, just that you shouldn’t consume with the goal of improving cardiovascular health. In fact, our analyses suggest that in an otherwise healthy person, up to 1 drink per day may not pose outsized risks,” he said. “And, even in a less healthy person who might be smoking, eating poorly, and drinking up to 1 drink per day, it may be a higher priority to focus on smoking cessation and diet than cutting back further on alcohol.”
“Beyond that amount, though, the jury is still out. Our models suggested marked increases in risk even between 1 and 2 drinks per day, and of course even greater risk increases beyond that. So, it’s probably worth revisiting what one might consider a ‘safe’ amount within the moderate drinking categories. The conservative move for now might be to advise a limit of 1 drink per day,” he said.
Dr. Aragam is supported by grants from the National Institutes of Health and the American Heart Association. He reports receiving speaking fees from the Novartis Institute for Biomedical Research.
A version of this article first appeared on Medscape.com.
Even very light alcohol intake is associated with an increased risk for cardiovascular disease, compared with not drinking at all, and the risk increases exponentially as alcohol intake rises, even at moderate levels, a new study shows.
“Our findings suggest that the observed benefit in individuals with light to moderate alcohol intake, which is consistently shown in epidemiological studies, is likely due to other positive lifestyle factors that are common in these individuals who drink lightly,” senior author Krishna Aragam, MD, Massachusetts General Hospital, Boston, told this news organization.
“Our results also showed that while all levels of alcohol were linked to increased risk of cardiovascular disease, the association was not linear. Rather, light alcohol intake was associated with rather modest risk increases, but there were exponential increases in cardiovascular risk with increasing amounts of alcohol consumption,” he said.
As the risk gradient appeared to increase quite sharply even between 1 and 2 drinks per day, Dr. Aragam suggested that what might be regarded as safe levels of drinking may trend downward in the future.
The study was published online March 25 in JAMA Network Open.
The cohort study used data from the UK Biobank, collected between 2006 and 2010 with follow-up until 2016, to assess the relationship between various levels of alcohol consumption and risk for cardiovascular disease.
Data were analyzed from 371,463 participants (mean age, 57 years; 46% men) who consumed an average of 9.2 standard drinks per week. Of these participants, 33% had hypertension and 7.5% had coronary artery disease.
“Use of the UK biobank database gives the advantage of a large, well-phenotyped population with a lot of information on various lifestyle factors that could be potential confounders,” Dr. Aragam noted.
Results showed that well-established J- or U-shaped curves were seen for the association between alcohol consumption and both the prevalence and hazards of hypertension, coronary artery disease, myocardial infarction, stroke, heart failure, and atrial fibrillation.
However, individuals in the light and moderate consumption group had healthier lifestyle behaviors than abstainers, self-reporting better overall health and exhibiting lower rates of smoking, lower body mass index, higher physical activity, and higher vegetable intake.
Adjustment for these lifestyle factors attenuated the cardioprotective associations with modest alcohol intake. For example, in baseline models, moderate intake was associated with significantly lower risk of hypertension and coronary artery disease, but adjustment for just six lifestyle factors rendered these results insignificant.
“Adjustments for yet unmeasured or unknown factors may further attenuate, if not eliminate, the residual, cardioprotective associations observed among light drinkers,” the researchers suggest.
They also conducted genetic analyses to examine the effect of alcohol and cardiovascular disease.
Dr. Aragam explained that previous work has shown good evidence, in individuals who choose to drink, that several relevant genetic variants predict levels of alcohol consumption quite accurately.
“Mendelian randomization using these gene variants allows for stronger inferences about potential causality than do observational studies, as they are less affected by confounding factors,” he noted.
Newer techniques in Mendelian randomization in which data on several gene variants linked to alcohol consumption are combined into a score allow for a greater understanding of the risk linked to different amount of alcohol intake, he added.
In these Mendelian randomization analyses, a 1-standard deviation increase in genetically predicted alcohol consumption was associated with 1.3-fold higher risk of hypertension (P < .001) and 1.4-fold higher risk of coronary artery disease (P = .006).
Further analyses suggested nonlinear associations between alcohol consumption and both hypertension and coronary artery disease; light alcohol intake was associated with minimal increases in cardiovascular risk, whereas heavier consumption was associated with exponential increases in risk of both clinical and subclinical cardiovascular disease.
These results were replicated in a second database of 30,716 individuals from the Mass General Brigham Biobank.
“The findings of this study suggest that the observed cardioprotective effects of light to moderate alcohol intake may be largely mediated by confounding lifestyle factors,” the researchers conclude. “Genetic analyses suggest causal associations between alcohol intake and cardiovascular disease but with unequal and exponential increases in risk at greater levels of intake, which should be accounted for in health recommendations around the habitual consumption of alcohol.”
What is an acceptable level?
“Specifically, our results suggest that consuming as many as 7 drinks per week is associated with relatively modest increases in cardiovascular risk,” they write.
But they point out that there are unequal increases in cardiovascular risk when progressing from 0 to 7 versus 7 to 14 drinks per week in both men and women.
“Although risk thresholds are inherently somewhat subjective, these findings again bring into question whether an average consumption of 2 drinks per day (14 drinks per week) should be designated a low-risk behavior,” they say.
“Furthermore, as several-fold increases in risk were observed for those consuming 21 or more drinks per week, our results emphasize the importance of aggressive efforts to reduce alcohol intake among heavy drinkers,” they add.
Dr. Aragam elaborated: “Our data suggest that reducing alcohol intake will reduce cardiovascular risk in all individuals, but the extent of the relative risk reduction is quite different depending on the current levels of consumption. For the same absolute reduction in alcohol intake, the gains in terms of reduction in cardiovascular risk will be more pronounced in those who drink heavily and will be more modest in those who drink at a light level.”
The results also suggest that while all levels of alcohol intake increase cardiovascular risk, there are low levels of alcohol consumption that do not carry major elevations in risk, but these are probably lower than those currently recommended, Dr. Aragam pointed out.
“This doesn’t mean that everyone has to give up drinking alcohol completely, just that you shouldn’t consume with the goal of improving cardiovascular health. In fact, our analyses suggest that in an otherwise healthy person, up to 1 drink per day may not pose outsized risks,” he said. “And, even in a less healthy person who might be smoking, eating poorly, and drinking up to 1 drink per day, it may be a higher priority to focus on smoking cessation and diet than cutting back further on alcohol.”
“Beyond that amount, though, the jury is still out. Our models suggested marked increases in risk even between 1 and 2 drinks per day, and of course even greater risk increases beyond that. So, it’s probably worth revisiting what one might consider a ‘safe’ amount within the moderate drinking categories. The conservative move for now might be to advise a limit of 1 drink per day,” he said.
Dr. Aragam is supported by grants from the National Institutes of Health and the American Heart Association. He reports receiving speaking fees from the Novartis Institute for Biomedical Research.
A version of this article first appeared on Medscape.com.
Hybrid ACC 2022 resurrects the live scientific session
Regardless of the pandemic’s sometimes mercurial behavior, the cardiology community appears set to reclaim valued traditions perhaps taken for granted in the pre-COVID era.
They include the bustling scientific congress and its myriad educational and networking prospects, along with pleiotropic effects like unplanned reunions with colleagues and catching up face-to-face with old friends.
That seems evident in the growing number of registrants for live attendance at at the annual scientific sessions of the American College of Cardiology, set for this Saturday through Monday in Washington as well as virtually, for a global reach that was unattainable in the pre-COVID era.
Registrations had hit the 11,000 mark and were picking up speed in recent weeks, ACC 2022 cochair Pamela B. Morris, MD, Medical University of South Carolina, Charleston, said at a mid-March presentation to the media.
They had reached about 12,880 and were still climbing a week before the conference, the ACC confirmed to this news organization. By then the professional registration had surpassed 9,900, of whom more than two-thirds reported plans to attend in person.
Dr. Morris said there had been 117 international submissions for what turned out to be 39 coveted spots on the meeting’s Late-Breaking Clinical Trial (LBCT) and Featured Clinical Research agenda spread across eight separate sessions.
On-site participants at the Walter E. Washington Convention Center should head for the Main Tent in Hall D for all LBCT presentations; venues for the Featured Clinical Research sessions are as noted below. Their real-time virtual equivalents will reside on the online platform’s Hot Topics channel. All noted session times are Eastern Daylight Time.
Saturday, April 2, 9:30 a.m.–10:30 a.m. Joint American College of Cardiology/Journal of the American College of Cardiology LBCT (I)
Leading off the conference’s first LBCT session, the randomized VALOR-HCM trial explored whether 16 weeks of mavacamten (MyoKardia) could help patients with severe obstructive hypertrophic cardiomyopathy (HCM) avoid septal reduction therapy, either surgical or by alcohol ablation.
The 22-center VALOR-HCM trial with an estimated enrollment of 100 follows EXPLORER-HCM, which in 2020 suggested the novel myosin-inhibiting agent could improve symptoms, exercise capacity, cardiac remodeling, and quality of life in such patients.
Simply advising people with heart failure (HF) to consume less salt is one thing, but it’s another to show them clinical trial evidence that it might help keep them out of the hospital. The SODIUM-HF (Study of Dietary Intervention Under 100 mmol in Heart Failure) study, conducted at 27 sites in six countries, sought to provide that evidence.
The trial randomly assigned 1,000 patients with NYHA class 2-3 HF to consume no more than 1,500 mg/day in sodium or to receive standard advice to limit sodium intake, and followed them for a year for the endpoint of death from any cause, cardiovascular (CV) hospitalization, or CV emergency department visit.
SODIUM-HF “may provide a rigorous evidence base for sodium restriction in patients with heart failure and may truly change our practice and how we recommend dietary modification,” ACC 2022 vice chair Douglas E. Drachman, MD, Massachusetts General Hospital, Boston, said at the media presentation.
In the same session, the CHAP (Chronic Hypertension and Pregnancy) study explored whether blood pressure (BP) control in pregnant women with new or untreated chronic hypertension could help avert preeclampsia, poor fetal outcomes, and other adverse events.
CHAP assigned about 2,400 women to receive either stepwise antihypertensive therapy to a BP goal of 140/90 mm Hg or lower or no such meds unless their BP reached or exceeded 160/105 mm Hg. Stepwise therapy featured either labetalol or extended-release nifedipine to start, the other agent added as necessary.
The LBCT block also includes the POISE-3 (Perioperative Ischemic Evaluation-3) comparison of the hemostatic agent tranexamic acid vs. placebo in nearly 10,000 patients undergoing noncardiac surgery. A separate randomization of the same cohort, to be reported at a Monday LBCT session, compared pre- and perioperative BP-control strategies.
Saturday, April 2, 12:00 p.m.–1:15 p.m. Featured Clinical Research I. Room 143A
This session features a subgroup analysis by age from the REVERSE-IT trial, which had previously showcased the monoclonal antibody bentracimab (PhaseBio Pharmaceuticals) for its ability to reverse the antiplatelet effects of ticagrelor.
REVERSE-IT is accompanied on the schedule by several secondary-endpoint presentations from trials whose primary outcomes have already been presented at meetings or in the journals.
They include the SCORED trial of sotagliflozin in patients with diabetes and chronic kidney disease (CKD); COMPLETE, which explored complete revascularization of multivessel coronary disease at primary stenting; and the FAME-3 comparison of coronary bypass surgery (CABG) vs. percutaneous coronary intervention (PCI) guided by fractional flow reserve (FFR) readings.
The session is to conclude with EDIT-CMD, which was a small, randomized assessment of diltiazem for improving microvascular dysfunction in patients with chronic angina despite nonobstructive coronary disease.
Sunday, April 3, 8:00 a.m.–9:15 a.m. Joint American College of Cardiology/Journal of the American Medical Association LBCT (II)
The SuperWIN (Supermarket Web Intervention) study tested an innovative strategy for community-based promotion of healthy lifestyle choices: point-of-purchase dietary education for grocery shoppers with an online instructional component, and follow-up to determine whether it influenced future food choices.
“Dietary interventions are notoriously difficult for us to implement, let alone to study scientifically,” Dr. Drachman observed. “So we think that there may be opportunity for dietary interventions to be best implemented at grocery stores where people are doing their shopping for food.”
SuperWIN compared supermarket shoppers with at least one CV risk factor who participated in the education intervention to a nonintervention control group for any changes in their DASH scores. The scores reflected consistency with the venerable DASH diet based on participants’ food purchases over 3 months.
In the same session, the MITIGATE trial explored whether daily administration of icosapent ethyl (Vascepa) might cut the risk of upper respiratory infection (especially from SARS-CoV-2 or seasonal influenza virus) in persons 50 or older with a history of clinical coronary, neurovascular, or peripheral vascular disease or revascularization. The trial has an estimated enrollment of 39,600.
Accompanying SuperWIN and MITIGATE are studies of several dyslipidemia drugs, including the discontinued antisense agent vupanorsen (Pfizer), as tested in TRANSLATE-TIMI 70; the PCSK9 inhibitor alirocumab (Praluent), explored for its effects on coronary plaque volume and composition in the PACMAN-AMI trial; and the APOLLO trial, a phase 1 evaluation of SLN360 (Silence Therapeutics), a short interfering ribonucleic acid (siRNA) that suppresses the molecular machinery in the liver that produces lipoprotein(a), or Lp(a).
The 32-patient APOLLO trial’s recently released top-line results suggested that SLN360 at varying dosages reduced Lp(a) levels by about one-half to more than 90%. Although elevated Lp(a) is known to track with CV risk, it remains to be shown whether dropping Lp(a) levels pharmacologically is protective.
Sunday, April 3, 9:45 a.m.–11:00 a.m. Joint American College of Cardiology/New England Journal of Medicine LBCT (III)
The meeting’s all-HF late-breaker session includes the METEORIC-HF trial, which compared the myotropic agent omecamtiv mecarbil (Cytokinetics) against placebo for effects on exercise performance over 20 weeks. The trial entered 276 patients with HF with reduced ejection fraction (HFrEF) and reduced peak VO2.
The GALACTIC-HF trial had previously suggested that the drug improved the risk of HF-related events or CV death in more than 8000 patients with HFrEF, those with the lowest ejection fractions benefiting the most.
This block of trials also features DIAMOND, the latest trial with a gemologic name to look at the potassium sequestrant patiromer (Veltassa) for any protection against hyperkalemia, a familiar side effect of renin-angiotensin-aldosterone inhibitors. DIAMOND tested patiromer in 878 patients with HFrEF who were on beta-blockers and other HF-appropriate medications and had a history of drug-associated hyperkalemia.
Previously, the AMBER trial of patients with CKD or refractory hypertension on spironolactone had suggested the drug might be protective enough against hyperkalemia to allow higher and more consistent dosing of BP-lowering agents.
Also in the session: the randomized IVVE (Influenza Vaccine to Prevent Adverse Vascular Events) trial, with an estimated 5,000 patients with HF in Africa, Asia, and the Middle East; PROMPT-HF, with a projected 1,310 HF patients and billed as a cluster-randomized pragmatic trial of a strategy for improving guideline-directed outpatient medical therapy; and MAVA-LTE, the long-term extension study of an estimated 310 patients who were in the MAVERICK-HCM and EXPLORER-HCM mavacamten trials.
Sunday, April 3, 12:15–1:30 p.m. Featured Clinical Research II. Main Tent, Hall D
The arrhythmia-centric session includes PARTITA, with its estimated 590 patients with primary- or secondary-prevention implantable cardioverter-defibrillators (ICDs). The trial followed them initially for burden of untreated nonsustained ventricular tachycardia (VT) or events treated with anti-tachycardia pacing. Then it randomly assigned those who experienced a first appropriate ICD shock to either immediate VT ablation or standard care. The latter included ablation on next occurrence of arrhythmic storm.
Investigational oral factor XIa inhibitors, viewed by many as potentially safer as anticoagulants than contemporary oral inhibitors of factor Xa, are now on the scene and include milvexian (Bristol-Myers Squibb/Janssen) and, lately, asundexian (BAY 2433334; Bayer). The latter agent was compared to the factor Xa inhibitor apixaban (Eliquis) in 753 patients with AF in the phase 2 PACIFIC-AF trial, which looked at the newer drug’s safety and optimal dosing.
Also on the bill: a long-term follow-up of the mAFA-2 (Mobile AF Application 2) extension study, which explored the value of a smartphone-based atrial fibrillation (AF) screening app for improving risk of AF-related events; a presentation billed as “Residual Leaks Post Left Atrial Appendage Occlusion”; and one that declares “low rates of guideline-directed care” to be “associated with higher mortality” in patients with pacemakers or ICDs.
Monday, April 4, 8:30 a.m.–9:45 a.m. LBCT IV
This session is to open with the PROTECT trial, which sought to determine whether perioperative “aggressive warming” may be cardioprotective in patients with CV risk factors undergoing noncardiac surgery. Its estimated 5,100 patients were randomly assigned to a procedure that achieves normothermia, that is 37° C (98.6° F), vs. standard care in which patients’ core temperature may decline to no further than 35.5° C (95.9° F).
Next on the list are a second POISE-3 comparison of BP-control strategies comparing hypotension avoidance vs. hypertension avoidance in patients undergoing noncardiac surgery; the pivotal CLASP 2 TR trial of patients with symptomatic tricuspid regurgitation on optimal medical therapy with vs. without treatment with the Edwards PASCAL Transcatheter Repair System; and one said to provide “insights from the Corevalve US Pivotal and SURTAVI trials” on 5-year incidence, timing, and predictors of hemodynamic valve deterioration transcatheter and surgical aortic bioprostheses.”
Rounding out the block of presentations: the ADAPT-TAVR comparison of the factor Xa inhibitor edoxaban (Lixiana) to dual-antiplatelet therapy for prevention of leaflet thrombosis after successful transcatheter aortic valve replacement (TAVR). The 235-patient trial was conducted at five centers in South Korea, Hong Kong, and Taiwan.
Monday, April 4, 11:00–12:15 p.m. LBCT V
This session includes the FLAVOUR randomized comparison of PCI guided by either FFR or intravascular ultrasound (IVUS) in 1,700 patients with 40%-70% stenoses. The patients from centers in China and South Korea were followed for death from any cause, MI, or any repeat revascularization at 24 months.
Also scheduled: the 2-year report on 4,000 patients with ST-segment elevation MI (STEMI) in the ACC-sponsored quality improvement program GHATI (Global Heart Attack Treatment Initiative); the GIPS-4 myocardial protection study of an estimated 380 patients with STEMI assigned to receive pre- and post-PCI infusions of sodium thiosulfate or placebo, with infarct size at 4 months as the primary endpoint; and a randomized test of an arrhythmia-monitoring implant for influence on clinical outcomes in 802 patients with a history of MI but no pacemaker or ICD indication, called BIO-GUARD-MI,
Last in the session: the Chocolate Touch Study of peripheral-artery angioplasty using a drug-coated balloon (DCB) with a confectionery name that treats lesions not with theobromine, but the antiproliferative mainstay paclitaxel.
The randomized comparison of the Chocolate Touch DCB (TriReme Medical) and the more established Lutonix DCB (Bard) assigned a projected 585 patients with symptomatic peripheral vascular disease to treatment of superficial femoral or popliteal artery lesions with one of the two paclitaxel-coated balloon catheters.
Monday, April 4, 12:45–2 p.m. Featured Clinical Research III. Room 143A
The final session features five subgroup analyses or other updates from trials that have already reported their primary outcomes. Among them is the SPYRAL HTN-ON MED trial, which helped to revitalize hopes for renal denervation therapy as a catheter-based treatment for drug-resistant hypertension by showing significant effects on both systolic and diastolic blood pressure. The new data follow the trial’s more than 400 patients out to 3 years.
There is also a symptom and quality-of-life analysis from the 530-patient EMPULSE trial of 530 patients with stabilized acute HF assigned in-hospital to start on empagliflozin (Jardiance) or placebo. The trial made a splash last year when it reported a significant improvement in risk for death or HF rehospitalization for its patients put on the SGLT2 inhibitor.
A secondary analysis from CANTOS is also featured; the trial had randomly assigned more than 10,000 patients with recent acute MI and elevated C-reactive protein (CRP) levels to receive or not receive the anti-inflammatory canakinumab (Ilaris). Those assigned to active therapy showed benefits for a range of outcomes, including CV mortality and stroke, but no decreases in cholesterol levels. Billing for the new CANTOS analysis promises insights on the “differential impact of residual inflammatory risk and residual cholesterol risk among atherosclerosis patients with and without chronic kidney disease.”
The session also features “trends and final results” from the NACMI (North American COVID-19 Myocardial Infarction) registry, which had shown excellent primary-PCI results without compromise of door-to-balloon times in patients with confirmed SARS-CoV-2 infection; and a FIDELITY analysis of cardiorenal endpoints by history of CV disease in the study’s more than 13,000 patients with diabetes and CKD assigned to placebo or finerenone (Kerendia), a mineralocorticoid receptor antagonist.
A version of this article first appeared on Medscape.com.
Regardless of the pandemic’s sometimes mercurial behavior, the cardiology community appears set to reclaim valued traditions perhaps taken for granted in the pre-COVID era.
They include the bustling scientific congress and its myriad educational and networking prospects, along with pleiotropic effects like unplanned reunions with colleagues and catching up face-to-face with old friends.
That seems evident in the growing number of registrants for live attendance at at the annual scientific sessions of the American College of Cardiology, set for this Saturday through Monday in Washington as well as virtually, for a global reach that was unattainable in the pre-COVID era.
Registrations had hit the 11,000 mark and were picking up speed in recent weeks, ACC 2022 cochair Pamela B. Morris, MD, Medical University of South Carolina, Charleston, said at a mid-March presentation to the media.
They had reached about 12,880 and were still climbing a week before the conference, the ACC confirmed to this news organization. By then the professional registration had surpassed 9,900, of whom more than two-thirds reported plans to attend in person.
Dr. Morris said there had been 117 international submissions for what turned out to be 39 coveted spots on the meeting’s Late-Breaking Clinical Trial (LBCT) and Featured Clinical Research agenda spread across eight separate sessions.
On-site participants at the Walter E. Washington Convention Center should head for the Main Tent in Hall D for all LBCT presentations; venues for the Featured Clinical Research sessions are as noted below. Their real-time virtual equivalents will reside on the online platform’s Hot Topics channel. All noted session times are Eastern Daylight Time.
Saturday, April 2, 9:30 a.m.–10:30 a.m. Joint American College of Cardiology/Journal of the American College of Cardiology LBCT (I)
Leading off the conference’s first LBCT session, the randomized VALOR-HCM trial explored whether 16 weeks of mavacamten (MyoKardia) could help patients with severe obstructive hypertrophic cardiomyopathy (HCM) avoid septal reduction therapy, either surgical or by alcohol ablation.
The 22-center VALOR-HCM trial with an estimated enrollment of 100 follows EXPLORER-HCM, which in 2020 suggested the novel myosin-inhibiting agent could improve symptoms, exercise capacity, cardiac remodeling, and quality of life in such patients.
Simply advising people with heart failure (HF) to consume less salt is one thing, but it’s another to show them clinical trial evidence that it might help keep them out of the hospital. The SODIUM-HF (Study of Dietary Intervention Under 100 mmol in Heart Failure) study, conducted at 27 sites in six countries, sought to provide that evidence.
The trial randomly assigned 1,000 patients with NYHA class 2-3 HF to consume no more than 1,500 mg/day in sodium or to receive standard advice to limit sodium intake, and followed them for a year for the endpoint of death from any cause, cardiovascular (CV) hospitalization, or CV emergency department visit.
SODIUM-HF “may provide a rigorous evidence base for sodium restriction in patients with heart failure and may truly change our practice and how we recommend dietary modification,” ACC 2022 vice chair Douglas E. Drachman, MD, Massachusetts General Hospital, Boston, said at the media presentation.
In the same session, the CHAP (Chronic Hypertension and Pregnancy) study explored whether blood pressure (BP) control in pregnant women with new or untreated chronic hypertension could help avert preeclampsia, poor fetal outcomes, and other adverse events.
CHAP assigned about 2,400 women to receive either stepwise antihypertensive therapy to a BP goal of 140/90 mm Hg or lower or no such meds unless their BP reached or exceeded 160/105 mm Hg. Stepwise therapy featured either labetalol or extended-release nifedipine to start, the other agent added as necessary.
The LBCT block also includes the POISE-3 (Perioperative Ischemic Evaluation-3) comparison of the hemostatic agent tranexamic acid vs. placebo in nearly 10,000 patients undergoing noncardiac surgery. A separate randomization of the same cohort, to be reported at a Monday LBCT session, compared pre- and perioperative BP-control strategies.
Saturday, April 2, 12:00 p.m.–1:15 p.m. Featured Clinical Research I. Room 143A
This session features a subgroup analysis by age from the REVERSE-IT trial, which had previously showcased the monoclonal antibody bentracimab (PhaseBio Pharmaceuticals) for its ability to reverse the antiplatelet effects of ticagrelor.
REVERSE-IT is accompanied on the schedule by several secondary-endpoint presentations from trials whose primary outcomes have already been presented at meetings or in the journals.
They include the SCORED trial of sotagliflozin in patients with diabetes and chronic kidney disease (CKD); COMPLETE, which explored complete revascularization of multivessel coronary disease at primary stenting; and the FAME-3 comparison of coronary bypass surgery (CABG) vs. percutaneous coronary intervention (PCI) guided by fractional flow reserve (FFR) readings.
The session is to conclude with EDIT-CMD, which was a small, randomized assessment of diltiazem for improving microvascular dysfunction in patients with chronic angina despite nonobstructive coronary disease.
Sunday, April 3, 8:00 a.m.–9:15 a.m. Joint American College of Cardiology/Journal of the American Medical Association LBCT (II)
The SuperWIN (Supermarket Web Intervention) study tested an innovative strategy for community-based promotion of healthy lifestyle choices: point-of-purchase dietary education for grocery shoppers with an online instructional component, and follow-up to determine whether it influenced future food choices.
“Dietary interventions are notoriously difficult for us to implement, let alone to study scientifically,” Dr. Drachman observed. “So we think that there may be opportunity for dietary interventions to be best implemented at grocery stores where people are doing their shopping for food.”
SuperWIN compared supermarket shoppers with at least one CV risk factor who participated in the education intervention to a nonintervention control group for any changes in their DASH scores. The scores reflected consistency with the venerable DASH diet based on participants’ food purchases over 3 months.
In the same session, the MITIGATE trial explored whether daily administration of icosapent ethyl (Vascepa) might cut the risk of upper respiratory infection (especially from SARS-CoV-2 or seasonal influenza virus) in persons 50 or older with a history of clinical coronary, neurovascular, or peripheral vascular disease or revascularization. The trial has an estimated enrollment of 39,600.
Accompanying SuperWIN and MITIGATE are studies of several dyslipidemia drugs, including the discontinued antisense agent vupanorsen (Pfizer), as tested in TRANSLATE-TIMI 70; the PCSK9 inhibitor alirocumab (Praluent), explored for its effects on coronary plaque volume and composition in the PACMAN-AMI trial; and the APOLLO trial, a phase 1 evaluation of SLN360 (Silence Therapeutics), a short interfering ribonucleic acid (siRNA) that suppresses the molecular machinery in the liver that produces lipoprotein(a), or Lp(a).
The 32-patient APOLLO trial’s recently released top-line results suggested that SLN360 at varying dosages reduced Lp(a) levels by about one-half to more than 90%. Although elevated Lp(a) is known to track with CV risk, it remains to be shown whether dropping Lp(a) levels pharmacologically is protective.
Sunday, April 3, 9:45 a.m.–11:00 a.m. Joint American College of Cardiology/New England Journal of Medicine LBCT (III)
The meeting’s all-HF late-breaker session includes the METEORIC-HF trial, which compared the myotropic agent omecamtiv mecarbil (Cytokinetics) against placebo for effects on exercise performance over 20 weeks. The trial entered 276 patients with HF with reduced ejection fraction (HFrEF) and reduced peak VO2.
The GALACTIC-HF trial had previously suggested that the drug improved the risk of HF-related events or CV death in more than 8000 patients with HFrEF, those with the lowest ejection fractions benefiting the most.
This block of trials also features DIAMOND, the latest trial with a gemologic name to look at the potassium sequestrant patiromer (Veltassa) for any protection against hyperkalemia, a familiar side effect of renin-angiotensin-aldosterone inhibitors. DIAMOND tested patiromer in 878 patients with HFrEF who were on beta-blockers and other HF-appropriate medications and had a history of drug-associated hyperkalemia.
Previously, the AMBER trial of patients with CKD or refractory hypertension on spironolactone had suggested the drug might be protective enough against hyperkalemia to allow higher and more consistent dosing of BP-lowering agents.
Also in the session: the randomized IVVE (Influenza Vaccine to Prevent Adverse Vascular Events) trial, with an estimated 5,000 patients with HF in Africa, Asia, and the Middle East; PROMPT-HF, with a projected 1,310 HF patients and billed as a cluster-randomized pragmatic trial of a strategy for improving guideline-directed outpatient medical therapy; and MAVA-LTE, the long-term extension study of an estimated 310 patients who were in the MAVERICK-HCM and EXPLORER-HCM mavacamten trials.
Sunday, April 3, 12:15–1:30 p.m. Featured Clinical Research II. Main Tent, Hall D
The arrhythmia-centric session includes PARTITA, with its estimated 590 patients with primary- or secondary-prevention implantable cardioverter-defibrillators (ICDs). The trial followed them initially for burden of untreated nonsustained ventricular tachycardia (VT) or events treated with anti-tachycardia pacing. Then it randomly assigned those who experienced a first appropriate ICD shock to either immediate VT ablation or standard care. The latter included ablation on next occurrence of arrhythmic storm.
Investigational oral factor XIa inhibitors, viewed by many as potentially safer as anticoagulants than contemporary oral inhibitors of factor Xa, are now on the scene and include milvexian (Bristol-Myers Squibb/Janssen) and, lately, asundexian (BAY 2433334; Bayer). The latter agent was compared to the factor Xa inhibitor apixaban (Eliquis) in 753 patients with AF in the phase 2 PACIFIC-AF trial, which looked at the newer drug’s safety and optimal dosing.
Also on the bill: a long-term follow-up of the mAFA-2 (Mobile AF Application 2) extension study, which explored the value of a smartphone-based atrial fibrillation (AF) screening app for improving risk of AF-related events; a presentation billed as “Residual Leaks Post Left Atrial Appendage Occlusion”; and one that declares “low rates of guideline-directed care” to be “associated with higher mortality” in patients with pacemakers or ICDs.
Monday, April 4, 8:30 a.m.–9:45 a.m. LBCT IV
This session is to open with the PROTECT trial, which sought to determine whether perioperative “aggressive warming” may be cardioprotective in patients with CV risk factors undergoing noncardiac surgery. Its estimated 5,100 patients were randomly assigned to a procedure that achieves normothermia, that is 37° C (98.6° F), vs. standard care in which patients’ core temperature may decline to no further than 35.5° C (95.9° F).
Next on the list are a second POISE-3 comparison of BP-control strategies comparing hypotension avoidance vs. hypertension avoidance in patients undergoing noncardiac surgery; the pivotal CLASP 2 TR trial of patients with symptomatic tricuspid regurgitation on optimal medical therapy with vs. without treatment with the Edwards PASCAL Transcatheter Repair System; and one said to provide “insights from the Corevalve US Pivotal and SURTAVI trials” on 5-year incidence, timing, and predictors of hemodynamic valve deterioration transcatheter and surgical aortic bioprostheses.”
Rounding out the block of presentations: the ADAPT-TAVR comparison of the factor Xa inhibitor edoxaban (Lixiana) to dual-antiplatelet therapy for prevention of leaflet thrombosis after successful transcatheter aortic valve replacement (TAVR). The 235-patient trial was conducted at five centers in South Korea, Hong Kong, and Taiwan.
Monday, April 4, 11:00–12:15 p.m. LBCT V
This session includes the FLAVOUR randomized comparison of PCI guided by either FFR or intravascular ultrasound (IVUS) in 1,700 patients with 40%-70% stenoses. The patients from centers in China and South Korea were followed for death from any cause, MI, or any repeat revascularization at 24 months.
Also scheduled: the 2-year report on 4,000 patients with ST-segment elevation MI (STEMI) in the ACC-sponsored quality improvement program GHATI (Global Heart Attack Treatment Initiative); the GIPS-4 myocardial protection study of an estimated 380 patients with STEMI assigned to receive pre- and post-PCI infusions of sodium thiosulfate or placebo, with infarct size at 4 months as the primary endpoint; and a randomized test of an arrhythmia-monitoring implant for influence on clinical outcomes in 802 patients with a history of MI but no pacemaker or ICD indication, called BIO-GUARD-MI,
Last in the session: the Chocolate Touch Study of peripheral-artery angioplasty using a drug-coated balloon (DCB) with a confectionery name that treats lesions not with theobromine, but the antiproliferative mainstay paclitaxel.
The randomized comparison of the Chocolate Touch DCB (TriReme Medical) and the more established Lutonix DCB (Bard) assigned a projected 585 patients with symptomatic peripheral vascular disease to treatment of superficial femoral or popliteal artery lesions with one of the two paclitaxel-coated balloon catheters.
Monday, April 4, 12:45–2 p.m. Featured Clinical Research III. Room 143A
The final session features five subgroup analyses or other updates from trials that have already reported their primary outcomes. Among them is the SPYRAL HTN-ON MED trial, which helped to revitalize hopes for renal denervation therapy as a catheter-based treatment for drug-resistant hypertension by showing significant effects on both systolic and diastolic blood pressure. The new data follow the trial’s more than 400 patients out to 3 years.
There is also a symptom and quality-of-life analysis from the 530-patient EMPULSE trial of 530 patients with stabilized acute HF assigned in-hospital to start on empagliflozin (Jardiance) or placebo. The trial made a splash last year when it reported a significant improvement in risk for death or HF rehospitalization for its patients put on the SGLT2 inhibitor.
A secondary analysis from CANTOS is also featured; the trial had randomly assigned more than 10,000 patients with recent acute MI and elevated C-reactive protein (CRP) levels to receive or not receive the anti-inflammatory canakinumab (Ilaris). Those assigned to active therapy showed benefits for a range of outcomes, including CV mortality and stroke, but no decreases in cholesterol levels. Billing for the new CANTOS analysis promises insights on the “differential impact of residual inflammatory risk and residual cholesterol risk among atherosclerosis patients with and without chronic kidney disease.”
The session also features “trends and final results” from the NACMI (North American COVID-19 Myocardial Infarction) registry, which had shown excellent primary-PCI results without compromise of door-to-balloon times in patients with confirmed SARS-CoV-2 infection; and a FIDELITY analysis of cardiorenal endpoints by history of CV disease in the study’s more than 13,000 patients with diabetes and CKD assigned to placebo or finerenone (Kerendia), a mineralocorticoid receptor antagonist.
A version of this article first appeared on Medscape.com.
Regardless of the pandemic’s sometimes mercurial behavior, the cardiology community appears set to reclaim valued traditions perhaps taken for granted in the pre-COVID era.
They include the bustling scientific congress and its myriad educational and networking prospects, along with pleiotropic effects like unplanned reunions with colleagues and catching up face-to-face with old friends.
That seems evident in the growing number of registrants for live attendance at at the annual scientific sessions of the American College of Cardiology, set for this Saturday through Monday in Washington as well as virtually, for a global reach that was unattainable in the pre-COVID era.
Registrations had hit the 11,000 mark and were picking up speed in recent weeks, ACC 2022 cochair Pamela B. Morris, MD, Medical University of South Carolina, Charleston, said at a mid-March presentation to the media.
They had reached about 12,880 and were still climbing a week before the conference, the ACC confirmed to this news organization. By then the professional registration had surpassed 9,900, of whom more than two-thirds reported plans to attend in person.
Dr. Morris said there had been 117 international submissions for what turned out to be 39 coveted spots on the meeting’s Late-Breaking Clinical Trial (LBCT) and Featured Clinical Research agenda spread across eight separate sessions.
On-site participants at the Walter E. Washington Convention Center should head for the Main Tent in Hall D for all LBCT presentations; venues for the Featured Clinical Research sessions are as noted below. Their real-time virtual equivalents will reside on the online platform’s Hot Topics channel. All noted session times are Eastern Daylight Time.
Saturday, April 2, 9:30 a.m.–10:30 a.m. Joint American College of Cardiology/Journal of the American College of Cardiology LBCT (I)
Leading off the conference’s first LBCT session, the randomized VALOR-HCM trial explored whether 16 weeks of mavacamten (MyoKardia) could help patients with severe obstructive hypertrophic cardiomyopathy (HCM) avoid septal reduction therapy, either surgical or by alcohol ablation.
The 22-center VALOR-HCM trial with an estimated enrollment of 100 follows EXPLORER-HCM, which in 2020 suggested the novel myosin-inhibiting agent could improve symptoms, exercise capacity, cardiac remodeling, and quality of life in such patients.
Simply advising people with heart failure (HF) to consume less salt is one thing, but it’s another to show them clinical trial evidence that it might help keep them out of the hospital. The SODIUM-HF (Study of Dietary Intervention Under 100 mmol in Heart Failure) study, conducted at 27 sites in six countries, sought to provide that evidence.
The trial randomly assigned 1,000 patients with NYHA class 2-3 HF to consume no more than 1,500 mg/day in sodium or to receive standard advice to limit sodium intake, and followed them for a year for the endpoint of death from any cause, cardiovascular (CV) hospitalization, or CV emergency department visit.
SODIUM-HF “may provide a rigorous evidence base for sodium restriction in patients with heart failure and may truly change our practice and how we recommend dietary modification,” ACC 2022 vice chair Douglas E. Drachman, MD, Massachusetts General Hospital, Boston, said at the media presentation.
In the same session, the CHAP (Chronic Hypertension and Pregnancy) study explored whether blood pressure (BP) control in pregnant women with new or untreated chronic hypertension could help avert preeclampsia, poor fetal outcomes, and other adverse events.
CHAP assigned about 2,400 women to receive either stepwise antihypertensive therapy to a BP goal of 140/90 mm Hg or lower or no such meds unless their BP reached or exceeded 160/105 mm Hg. Stepwise therapy featured either labetalol or extended-release nifedipine to start, the other agent added as necessary.
The LBCT block also includes the POISE-3 (Perioperative Ischemic Evaluation-3) comparison of the hemostatic agent tranexamic acid vs. placebo in nearly 10,000 patients undergoing noncardiac surgery. A separate randomization of the same cohort, to be reported at a Monday LBCT session, compared pre- and perioperative BP-control strategies.
Saturday, April 2, 12:00 p.m.–1:15 p.m. Featured Clinical Research I. Room 143A
This session features a subgroup analysis by age from the REVERSE-IT trial, which had previously showcased the monoclonal antibody bentracimab (PhaseBio Pharmaceuticals) for its ability to reverse the antiplatelet effects of ticagrelor.
REVERSE-IT is accompanied on the schedule by several secondary-endpoint presentations from trials whose primary outcomes have already been presented at meetings or in the journals.
They include the SCORED trial of sotagliflozin in patients with diabetes and chronic kidney disease (CKD); COMPLETE, which explored complete revascularization of multivessel coronary disease at primary stenting; and the FAME-3 comparison of coronary bypass surgery (CABG) vs. percutaneous coronary intervention (PCI) guided by fractional flow reserve (FFR) readings.
The session is to conclude with EDIT-CMD, which was a small, randomized assessment of diltiazem for improving microvascular dysfunction in patients with chronic angina despite nonobstructive coronary disease.
Sunday, April 3, 8:00 a.m.–9:15 a.m. Joint American College of Cardiology/Journal of the American Medical Association LBCT (II)
The SuperWIN (Supermarket Web Intervention) study tested an innovative strategy for community-based promotion of healthy lifestyle choices: point-of-purchase dietary education for grocery shoppers with an online instructional component, and follow-up to determine whether it influenced future food choices.
“Dietary interventions are notoriously difficult for us to implement, let alone to study scientifically,” Dr. Drachman observed. “So we think that there may be opportunity for dietary interventions to be best implemented at grocery stores where people are doing their shopping for food.”
SuperWIN compared supermarket shoppers with at least one CV risk factor who participated in the education intervention to a nonintervention control group for any changes in their DASH scores. The scores reflected consistency with the venerable DASH diet based on participants’ food purchases over 3 months.
In the same session, the MITIGATE trial explored whether daily administration of icosapent ethyl (Vascepa) might cut the risk of upper respiratory infection (especially from SARS-CoV-2 or seasonal influenza virus) in persons 50 or older with a history of clinical coronary, neurovascular, or peripheral vascular disease or revascularization. The trial has an estimated enrollment of 39,600.
Accompanying SuperWIN and MITIGATE are studies of several dyslipidemia drugs, including the discontinued antisense agent vupanorsen (Pfizer), as tested in TRANSLATE-TIMI 70; the PCSK9 inhibitor alirocumab (Praluent), explored for its effects on coronary plaque volume and composition in the PACMAN-AMI trial; and the APOLLO trial, a phase 1 evaluation of SLN360 (Silence Therapeutics), a short interfering ribonucleic acid (siRNA) that suppresses the molecular machinery in the liver that produces lipoprotein(a), or Lp(a).
The 32-patient APOLLO trial’s recently released top-line results suggested that SLN360 at varying dosages reduced Lp(a) levels by about one-half to more than 90%. Although elevated Lp(a) is known to track with CV risk, it remains to be shown whether dropping Lp(a) levels pharmacologically is protective.
Sunday, April 3, 9:45 a.m.–11:00 a.m. Joint American College of Cardiology/New England Journal of Medicine LBCT (III)
The meeting’s all-HF late-breaker session includes the METEORIC-HF trial, which compared the myotropic agent omecamtiv mecarbil (Cytokinetics) against placebo for effects on exercise performance over 20 weeks. The trial entered 276 patients with HF with reduced ejection fraction (HFrEF) and reduced peak VO2.
The GALACTIC-HF trial had previously suggested that the drug improved the risk of HF-related events or CV death in more than 8000 patients with HFrEF, those with the lowest ejection fractions benefiting the most.
This block of trials also features DIAMOND, the latest trial with a gemologic name to look at the potassium sequestrant patiromer (Veltassa) for any protection against hyperkalemia, a familiar side effect of renin-angiotensin-aldosterone inhibitors. DIAMOND tested patiromer in 878 patients with HFrEF who were on beta-blockers and other HF-appropriate medications and had a history of drug-associated hyperkalemia.
Previously, the AMBER trial of patients with CKD or refractory hypertension on spironolactone had suggested the drug might be protective enough against hyperkalemia to allow higher and more consistent dosing of BP-lowering agents.
Also in the session: the randomized IVVE (Influenza Vaccine to Prevent Adverse Vascular Events) trial, with an estimated 5,000 patients with HF in Africa, Asia, and the Middle East; PROMPT-HF, with a projected 1,310 HF patients and billed as a cluster-randomized pragmatic trial of a strategy for improving guideline-directed outpatient medical therapy; and MAVA-LTE, the long-term extension study of an estimated 310 patients who were in the MAVERICK-HCM and EXPLORER-HCM mavacamten trials.
Sunday, April 3, 12:15–1:30 p.m. Featured Clinical Research II. Main Tent, Hall D
The arrhythmia-centric session includes PARTITA, with its estimated 590 patients with primary- or secondary-prevention implantable cardioverter-defibrillators (ICDs). The trial followed them initially for burden of untreated nonsustained ventricular tachycardia (VT) or events treated with anti-tachycardia pacing. Then it randomly assigned those who experienced a first appropriate ICD shock to either immediate VT ablation or standard care. The latter included ablation on next occurrence of arrhythmic storm.
Investigational oral factor XIa inhibitors, viewed by many as potentially safer as anticoagulants than contemporary oral inhibitors of factor Xa, are now on the scene and include milvexian (Bristol-Myers Squibb/Janssen) and, lately, asundexian (BAY 2433334; Bayer). The latter agent was compared to the factor Xa inhibitor apixaban (Eliquis) in 753 patients with AF in the phase 2 PACIFIC-AF trial, which looked at the newer drug’s safety and optimal dosing.
Also on the bill: a long-term follow-up of the mAFA-2 (Mobile AF Application 2) extension study, which explored the value of a smartphone-based atrial fibrillation (AF) screening app for improving risk of AF-related events; a presentation billed as “Residual Leaks Post Left Atrial Appendage Occlusion”; and one that declares “low rates of guideline-directed care” to be “associated with higher mortality” in patients with pacemakers or ICDs.
Monday, April 4, 8:30 a.m.–9:45 a.m. LBCT IV
This session is to open with the PROTECT trial, which sought to determine whether perioperative “aggressive warming” may be cardioprotective in patients with CV risk factors undergoing noncardiac surgery. Its estimated 5,100 patients were randomly assigned to a procedure that achieves normothermia, that is 37° C (98.6° F), vs. standard care in which patients’ core temperature may decline to no further than 35.5° C (95.9° F).
Next on the list are a second POISE-3 comparison of BP-control strategies comparing hypotension avoidance vs. hypertension avoidance in patients undergoing noncardiac surgery; the pivotal CLASP 2 TR trial of patients with symptomatic tricuspid regurgitation on optimal medical therapy with vs. without treatment with the Edwards PASCAL Transcatheter Repair System; and one said to provide “insights from the Corevalve US Pivotal and SURTAVI trials” on 5-year incidence, timing, and predictors of hemodynamic valve deterioration transcatheter and surgical aortic bioprostheses.”
Rounding out the block of presentations: the ADAPT-TAVR comparison of the factor Xa inhibitor edoxaban (Lixiana) to dual-antiplatelet therapy for prevention of leaflet thrombosis after successful transcatheter aortic valve replacement (TAVR). The 235-patient trial was conducted at five centers in South Korea, Hong Kong, and Taiwan.
Monday, April 4, 11:00–12:15 p.m. LBCT V
This session includes the FLAVOUR randomized comparison of PCI guided by either FFR or intravascular ultrasound (IVUS) in 1,700 patients with 40%-70% stenoses. The patients from centers in China and South Korea were followed for death from any cause, MI, or any repeat revascularization at 24 months.
Also scheduled: the 2-year report on 4,000 patients with ST-segment elevation MI (STEMI) in the ACC-sponsored quality improvement program GHATI (Global Heart Attack Treatment Initiative); the GIPS-4 myocardial protection study of an estimated 380 patients with STEMI assigned to receive pre- and post-PCI infusions of sodium thiosulfate or placebo, with infarct size at 4 months as the primary endpoint; and a randomized test of an arrhythmia-monitoring implant for influence on clinical outcomes in 802 patients with a history of MI but no pacemaker or ICD indication, called BIO-GUARD-MI,
Last in the session: the Chocolate Touch Study of peripheral-artery angioplasty using a drug-coated balloon (DCB) with a confectionery name that treats lesions not with theobromine, but the antiproliferative mainstay paclitaxel.
The randomized comparison of the Chocolate Touch DCB (TriReme Medical) and the more established Lutonix DCB (Bard) assigned a projected 585 patients with symptomatic peripheral vascular disease to treatment of superficial femoral or popliteal artery lesions with one of the two paclitaxel-coated balloon catheters.
Monday, April 4, 12:45–2 p.m. Featured Clinical Research III. Room 143A
The final session features five subgroup analyses or other updates from trials that have already reported their primary outcomes. Among them is the SPYRAL HTN-ON MED trial, which helped to revitalize hopes for renal denervation therapy as a catheter-based treatment for drug-resistant hypertension by showing significant effects on both systolic and diastolic blood pressure. The new data follow the trial’s more than 400 patients out to 3 years.
There is also a symptom and quality-of-life analysis from the 530-patient EMPULSE trial of 530 patients with stabilized acute HF assigned in-hospital to start on empagliflozin (Jardiance) or placebo. The trial made a splash last year when it reported a significant improvement in risk for death or HF rehospitalization for its patients put on the SGLT2 inhibitor.
A secondary analysis from CANTOS is also featured; the trial had randomly assigned more than 10,000 patients with recent acute MI and elevated C-reactive protein (CRP) levels to receive or not receive the anti-inflammatory canakinumab (Ilaris). Those assigned to active therapy showed benefits for a range of outcomes, including CV mortality and stroke, but no decreases in cholesterol levels. Billing for the new CANTOS analysis promises insights on the “differential impact of residual inflammatory risk and residual cholesterol risk among atherosclerosis patients with and without chronic kidney disease.”
The session also features “trends and final results” from the NACMI (North American COVID-19 Myocardial Infarction) registry, which had shown excellent primary-PCI results without compromise of door-to-balloon times in patients with confirmed SARS-CoV-2 infection; and a FIDELITY analysis of cardiorenal endpoints by history of CV disease in the study’s more than 13,000 patients with diabetes and CKD assigned to placebo or finerenone (Kerendia), a mineralocorticoid receptor antagonist.
A version of this article first appeared on Medscape.com.
Sit-to-stand BP spike tied to CV events in young adults
A sudden drop in blood pressure when standing is a common and concerning problem in elderly hypertensive people. Now, research suggests a large BP swing in the opposite direction on standing may be equally concerning in younger hypertensive people.
Young and middle-aged adults with a systolic BP response to standing greater than 6.5 mm Hg had almost double the risk of major adverse cardiovascular events (MACE) during follow-up, compared with other participants.
An exaggerated BP response remained an independent predictor of MACE, even after adjusting for traditional risk factors, including 24-hour BP (hazard ratio, 1.94; 95% confidence interval, 1.10 to 3.44), the study showed.
“The clinical implication is important, because now doctors measure blood pressure in young people in the upright posture, but what we say is it must be measured also while standing,” said Paolo Palatini, MD, a professor of internal medicine at the University of Padova, Italy, who led the study.
Previous studies have found that an exaggerated BP response to standing is a predictor of future hypertension, CV events, and mortality, particularly in older patients, but few prognostic data exist in those who are young to middle age, he noted.
The study, published in Hypertension, included 1,207 participants ages 18-45 years with untreated stage 1 hypertension (systolic BP 140-159 mm Hg or diastolic BP 90-100 mm Hg) in the prospective multicenter HARVEST study that began in Italy in 1990. The average age at enrollment was 33 years.
BP was measured at two visits 2 weeks apart, with each visit including three supine measurements taken after the patient had lain down for a minimum of 5 minutes, followed by three standing measurements taken 1 minute apart.
Based on the average of standing-lying BP differences during the two visits, participants were then classified as having a normal or exaggerated (top decile, lower limit > 6.5 mm Hg) systolic BP response to standing.
The 120 participants classified as “hyper-reactors” averaged an 11.4 mm Hg systolic BP increase upon standing, whereas the rest of the participants averaged a 3.8 mm Hg fall in systolic BP upon standing.
At their initial visit, hyper-reactors were more likely to be smokers (32.1% vs. 19.9%) and coffee drinkers (81.7% vs. 73%) and to have ambulatory hypertension (90.8% vs. 76.4%).
They were, however, no more likely to have a family history of cardiovascular events and had a lower supine systolic BP (140.5 mm Hg vs. 146.0 mm Hg), lower total cholesterol (4.93 mmol/L vs. 5.13 mmol/L), and higher HDL cholesterol (1.42 mmol/L vs. 1.35 mmol/L).
Age, sex, and body mass index were similar between the two groups, as was BP variability, nocturnal BP dip, and the frequency of extreme dippers. Participants with a normal systolic BP response were more likely to be treated for hypertension during follow-up (81.7% vs. 69.7%; P = .003).
In 630 participants who had catecholamines measured from 24-hour urine samples, the epinephrine/creatinine ratio was higher in hyper-reactors than normal responders (118.4 nmol/mol vs. 77.0 nmol/mol; P = .005).
During a median follow-up of 17.3 years, there were 105 major cardiovascular events, broadly defined to include acute coronary syndromes (48), any stroke (13), heart failure requiring hospitalization (3), aortic aneurysms (3), peripheral vascular disease (6), chronic kidney disease (12), and permanent atrial fibrillation (20).
The near doubling of MACE risk among hyper-reactors remained when atrial fibrillation was excluded and when 24-hour ambulatory systolic BP was included in the model, the author reported.
The results are in line with previous studies, indicating that hyper-reactors to standing have normal sympathetic activity at rest but an increased sympathetic response to stressors, observed Dr. Palatini and colleagues. This neurohumoral overshoot seems to be peculiar to young adults, whereas vascular stiffness seems to be the driving mechanism of orthostatic hypertension in older adults.
If a young person’s BP spikes upon standing, “then you have to treat them according to the average of the lying and the standing pressure,” Dr. Palatini said. “In these people, blood pressure should be treated earlier than in the past.”
“The study is important because it identified a new marker for hypertension that is easily evaluated in clinical practice,” Nieca Goldberg, MD, medical director of the Atria Institute, New York, and an associate professor of medicine at New York University Grossman School of Medicine, commented via email.
She noted that standing blood pressures are usually not taken as part of a medical visit and, in fact, seated blood pressures are often taken incorrectly while the patient is seated on the exam table rather than with their feet on the floor and using the proper cuff size.
“By incorporating standing BP, we will improve our diagnosis for hypertension, and with interventions such as diet and exercise, salt reduction, and medication when indicated, lower risk for heart attack, stroke, heart failure, [and] kidney and eye disease,” said Dr. Goldberg, who is also a spokesperson for the American Heart Association.
“The biggest barrier is that office visits are limited to 15 minutes, and not enough time is spent on the vital signs,” she noted. “We need changes to the health care system that value our ability to diagnose BP and take the time to counsel patients and explain treatment options.”
Limitations of the present study are that 72.7% of participants were men and all were White, Dr. Palatini said. Future work is also needed to create a uniform definition of BP hyper-reactivity to standing, possibly based on risk estimates, for inclusion in future hypertension guidelines.
The study was funded by the Association 18 Maggio 1370 in Italy. The authors have disclosed no relevant financial relationships. Dr. Goldberg reported being a spokesperson for the American Heart Association.
A version of this article first appeared on Medscape.com.
A sudden drop in blood pressure when standing is a common and concerning problem in elderly hypertensive people. Now, research suggests a large BP swing in the opposite direction on standing may be equally concerning in younger hypertensive people.
Young and middle-aged adults with a systolic BP response to standing greater than 6.5 mm Hg had almost double the risk of major adverse cardiovascular events (MACE) during follow-up, compared with other participants.
An exaggerated BP response remained an independent predictor of MACE, even after adjusting for traditional risk factors, including 24-hour BP (hazard ratio, 1.94; 95% confidence interval, 1.10 to 3.44), the study showed.
“The clinical implication is important, because now doctors measure blood pressure in young people in the upright posture, but what we say is it must be measured also while standing,” said Paolo Palatini, MD, a professor of internal medicine at the University of Padova, Italy, who led the study.
Previous studies have found that an exaggerated BP response to standing is a predictor of future hypertension, CV events, and mortality, particularly in older patients, but few prognostic data exist in those who are young to middle age, he noted.
The study, published in Hypertension, included 1,207 participants ages 18-45 years with untreated stage 1 hypertension (systolic BP 140-159 mm Hg or diastolic BP 90-100 mm Hg) in the prospective multicenter HARVEST study that began in Italy in 1990. The average age at enrollment was 33 years.
BP was measured at two visits 2 weeks apart, with each visit including three supine measurements taken after the patient had lain down for a minimum of 5 minutes, followed by three standing measurements taken 1 minute apart.
Based on the average of standing-lying BP differences during the two visits, participants were then classified as having a normal or exaggerated (top decile, lower limit > 6.5 mm Hg) systolic BP response to standing.
The 120 participants classified as “hyper-reactors” averaged an 11.4 mm Hg systolic BP increase upon standing, whereas the rest of the participants averaged a 3.8 mm Hg fall in systolic BP upon standing.
At their initial visit, hyper-reactors were more likely to be smokers (32.1% vs. 19.9%) and coffee drinkers (81.7% vs. 73%) and to have ambulatory hypertension (90.8% vs. 76.4%).
They were, however, no more likely to have a family history of cardiovascular events and had a lower supine systolic BP (140.5 mm Hg vs. 146.0 mm Hg), lower total cholesterol (4.93 mmol/L vs. 5.13 mmol/L), and higher HDL cholesterol (1.42 mmol/L vs. 1.35 mmol/L).
Age, sex, and body mass index were similar between the two groups, as was BP variability, nocturnal BP dip, and the frequency of extreme dippers. Participants with a normal systolic BP response were more likely to be treated for hypertension during follow-up (81.7% vs. 69.7%; P = .003).
In 630 participants who had catecholamines measured from 24-hour urine samples, the epinephrine/creatinine ratio was higher in hyper-reactors than normal responders (118.4 nmol/mol vs. 77.0 nmol/mol; P = .005).
During a median follow-up of 17.3 years, there were 105 major cardiovascular events, broadly defined to include acute coronary syndromes (48), any stroke (13), heart failure requiring hospitalization (3), aortic aneurysms (3), peripheral vascular disease (6), chronic kidney disease (12), and permanent atrial fibrillation (20).
The near doubling of MACE risk among hyper-reactors remained when atrial fibrillation was excluded and when 24-hour ambulatory systolic BP was included in the model, the author reported.
The results are in line with previous studies, indicating that hyper-reactors to standing have normal sympathetic activity at rest but an increased sympathetic response to stressors, observed Dr. Palatini and colleagues. This neurohumoral overshoot seems to be peculiar to young adults, whereas vascular stiffness seems to be the driving mechanism of orthostatic hypertension in older adults.
If a young person’s BP spikes upon standing, “then you have to treat them according to the average of the lying and the standing pressure,” Dr. Palatini said. “In these people, blood pressure should be treated earlier than in the past.”
“The study is important because it identified a new marker for hypertension that is easily evaluated in clinical practice,” Nieca Goldberg, MD, medical director of the Atria Institute, New York, and an associate professor of medicine at New York University Grossman School of Medicine, commented via email.
She noted that standing blood pressures are usually not taken as part of a medical visit and, in fact, seated blood pressures are often taken incorrectly while the patient is seated on the exam table rather than with their feet on the floor and using the proper cuff size.
“By incorporating standing BP, we will improve our diagnosis for hypertension, and with interventions such as diet and exercise, salt reduction, and medication when indicated, lower risk for heart attack, stroke, heart failure, [and] kidney and eye disease,” said Dr. Goldberg, who is also a spokesperson for the American Heart Association.
“The biggest barrier is that office visits are limited to 15 minutes, and not enough time is spent on the vital signs,” she noted. “We need changes to the health care system that value our ability to diagnose BP and take the time to counsel patients and explain treatment options.”
Limitations of the present study are that 72.7% of participants were men and all were White, Dr. Palatini said. Future work is also needed to create a uniform definition of BP hyper-reactivity to standing, possibly based on risk estimates, for inclusion in future hypertension guidelines.
The study was funded by the Association 18 Maggio 1370 in Italy. The authors have disclosed no relevant financial relationships. Dr. Goldberg reported being a spokesperson for the American Heart Association.
A version of this article first appeared on Medscape.com.
A sudden drop in blood pressure when standing is a common and concerning problem in elderly hypertensive people. Now, research suggests a large BP swing in the opposite direction on standing may be equally concerning in younger hypertensive people.
Young and middle-aged adults with a systolic BP response to standing greater than 6.5 mm Hg had almost double the risk of major adverse cardiovascular events (MACE) during follow-up, compared with other participants.
An exaggerated BP response remained an independent predictor of MACE, even after adjusting for traditional risk factors, including 24-hour BP (hazard ratio, 1.94; 95% confidence interval, 1.10 to 3.44), the study showed.
“The clinical implication is important, because now doctors measure blood pressure in young people in the upright posture, but what we say is it must be measured also while standing,” said Paolo Palatini, MD, a professor of internal medicine at the University of Padova, Italy, who led the study.
Previous studies have found that an exaggerated BP response to standing is a predictor of future hypertension, CV events, and mortality, particularly in older patients, but few prognostic data exist in those who are young to middle age, he noted.
The study, published in Hypertension, included 1,207 participants ages 18-45 years with untreated stage 1 hypertension (systolic BP 140-159 mm Hg or diastolic BP 90-100 mm Hg) in the prospective multicenter HARVEST study that began in Italy in 1990. The average age at enrollment was 33 years.
BP was measured at two visits 2 weeks apart, with each visit including three supine measurements taken after the patient had lain down for a minimum of 5 minutes, followed by three standing measurements taken 1 minute apart.
Based on the average of standing-lying BP differences during the two visits, participants were then classified as having a normal or exaggerated (top decile, lower limit > 6.5 mm Hg) systolic BP response to standing.
The 120 participants classified as “hyper-reactors” averaged an 11.4 mm Hg systolic BP increase upon standing, whereas the rest of the participants averaged a 3.8 mm Hg fall in systolic BP upon standing.
At their initial visit, hyper-reactors were more likely to be smokers (32.1% vs. 19.9%) and coffee drinkers (81.7% vs. 73%) and to have ambulatory hypertension (90.8% vs. 76.4%).
They were, however, no more likely to have a family history of cardiovascular events and had a lower supine systolic BP (140.5 mm Hg vs. 146.0 mm Hg), lower total cholesterol (4.93 mmol/L vs. 5.13 mmol/L), and higher HDL cholesterol (1.42 mmol/L vs. 1.35 mmol/L).
Age, sex, and body mass index were similar between the two groups, as was BP variability, nocturnal BP dip, and the frequency of extreme dippers. Participants with a normal systolic BP response were more likely to be treated for hypertension during follow-up (81.7% vs. 69.7%; P = .003).
In 630 participants who had catecholamines measured from 24-hour urine samples, the epinephrine/creatinine ratio was higher in hyper-reactors than normal responders (118.4 nmol/mol vs. 77.0 nmol/mol; P = .005).
During a median follow-up of 17.3 years, there were 105 major cardiovascular events, broadly defined to include acute coronary syndromes (48), any stroke (13), heart failure requiring hospitalization (3), aortic aneurysms (3), peripheral vascular disease (6), chronic kidney disease (12), and permanent atrial fibrillation (20).
The near doubling of MACE risk among hyper-reactors remained when atrial fibrillation was excluded and when 24-hour ambulatory systolic BP was included in the model, the author reported.
The results are in line with previous studies, indicating that hyper-reactors to standing have normal sympathetic activity at rest but an increased sympathetic response to stressors, observed Dr. Palatini and colleagues. This neurohumoral overshoot seems to be peculiar to young adults, whereas vascular stiffness seems to be the driving mechanism of orthostatic hypertension in older adults.
If a young person’s BP spikes upon standing, “then you have to treat them according to the average of the lying and the standing pressure,” Dr. Palatini said. “In these people, blood pressure should be treated earlier than in the past.”
“The study is important because it identified a new marker for hypertension that is easily evaluated in clinical practice,” Nieca Goldberg, MD, medical director of the Atria Institute, New York, and an associate professor of medicine at New York University Grossman School of Medicine, commented via email.
She noted that standing blood pressures are usually not taken as part of a medical visit and, in fact, seated blood pressures are often taken incorrectly while the patient is seated on the exam table rather than with their feet on the floor and using the proper cuff size.
“By incorporating standing BP, we will improve our diagnosis for hypertension, and with interventions such as diet and exercise, salt reduction, and medication when indicated, lower risk for heart attack, stroke, heart failure, [and] kidney and eye disease,” said Dr. Goldberg, who is also a spokesperson for the American Heart Association.
“The biggest barrier is that office visits are limited to 15 minutes, and not enough time is spent on the vital signs,” she noted. “We need changes to the health care system that value our ability to diagnose BP and take the time to counsel patients and explain treatment options.”
Limitations of the present study are that 72.7% of participants were men and all were White, Dr. Palatini said. Future work is also needed to create a uniform definition of BP hyper-reactivity to standing, possibly based on risk estimates, for inclusion in future hypertension guidelines.
The study was funded by the Association 18 Maggio 1370 in Italy. The authors have disclosed no relevant financial relationships. Dr. Goldberg reported being a spokesperson for the American Heart Association.
A version of this article first appeared on Medscape.com.
New test for Lp(a) allows more accurate LDL-cholesterol results
A new study has drawn attention to inaccurate measurement of LDL-cholesterol levels in some patients with current assays, which could lead to incorrect therapeutic approaches.
The patient groups most affected are those with high levels of the lipoprotein Lp(a), in whom LDL-cholesterol levels are being overestimated in current laboratory tests, the authors say.
“Current laboratory assays all have the limitation that they cannot measure or report LDL cholesterol accurately. They are actually measuring the combination of LDL and Lp(a),” senior study author Sotirios Tsimikas, MD, University of California, San Diego, explained to this news organization.
“While this may not matter much in individuals with normal Lp(a) levels, in those with elevated Lp(a), the Lp(a) cholesterol may constitute a substantial proportion of the reported LDL cholesterol, and the actual LDL-cholesterol levels could be much lower that the value the lab is telling us,” he said.
Dr. Tsimikas gave the example of a patient with an LDL-cholesterol lab measurement of 75 mg/dL. “If that patient has an Lp(a) level of zero, then they do actually have an LDL level of 75. But as the Lp(a) increases, then the proportion of the result accounted for by LDL cholesterol decreases. So, if a patient with a measured LDL cholesterol of 75 has an Lp(a)-cholesterol level of 20, then their actual LDL level is 55.”
Dr. Tsimikas said it is important to know levels of both lipoproteins individually, so the correct therapeutic approach is used in situations where the Lp(a) cholesterol might be elevated.
“By understanding the actual values of LDL cholesterol and Lp(a) cholesterol, this will allow us to personalize the use of cholesterol-lowering medications and decide where to focus treatment. In the patient with a high level of Lp(a), their residual risk could be coming from Lp(a) cholesterol and less so from LDL cholesterol,” he added. “As we develop drugs to lower Lp(a), this patient might be better off on one of these rather than increasing efforts to lower LDL cholesterol, which might already be at goal.”
The study was published in the March 22 issue of the Journal of the American College of Cardiology.
Dr. Tsimikas noted that Lp(a) is now accepted as a genetic, independent, causal risk factor for cardiovascular disease, but current LDL-lowering drugs do not have much effect on Lp(a).
“Lp(a) can be lowered a little with niacin and PCSK9 inhibitors, but both have a quite a weak effect, and statins increase Lp(a). However, there are now multiple RNA-based therapeutics specifically targeting Lp(a) in clinical development,” he said.
At present, Lp(a) cholesterol has to be mathematically estimated, most commonly with the Dahlén formula, because of the lack of a validated, quantitative method to measure Lp(a) cholesterol, Dr. Tsimikas says.
For the current study, the researchers used a novel, quantitative, sensitive method to directly measure Lp(a) cholesterol, then applied this method to data from a recent study with the one of the new Lp(a)-lowering drugs in development – pelacarsen – which was conducted in patients with elevated Lp(a) levels.
Results showed that direct Lp(a)-cholesterol assessment, and subtracting this value from the laboratory LDL-cholesterol value, provides a more accurate reflection of the baseline and change in LDL cholesterol, the authors report. In the current study, corrected LDL cholesterol was 13 to 16 mg/dL lower than laboratory-reported LDL cholesterol.
Using the corrected LDL-cholesterol results, the study showed that pelacarsen significantly decreases Lp(a) cholesterol, with neutral to modest effects on LDL.
The study also suggests that the current method of calculating Lp(a) cholesterol, and then deriving a corrected LDL cholesterol – the Dahlén formula – is not accurate.
“The Dahlén formula relies on the assumption that Lp(a) cholesterol is universally a fixed 30% of Lp(a) mass, but this usually isn’t the case. The Dahlén formula needs to be discontinued. It can be highly inaccurate,” Dr. Tsimikas said.
Important implications
In an accompanying editorial, Guillaume Paré, MD, Michael Chong, PhD student, and Pedrum Mohammadi-Shemirani, BSc, all of McMaster University, Hamilton, Ont., say the current findings have three important clinical implications.
“First, they provide further proof that in individuals with elevated Lp(a), the contribution of Lp(a)-cholesterol to LDL-cholesterol is non-negligible using standard assays, with 13-16 mg/dL lower LDL-cholesterol post-correction.”
Secondly, the editorialists point out that these new findings confirm that the effect of Lp(a) inhibitors is likely to be mostly confined to Lp(a), “as would be expected.”
Finally, “and perhaps more importantly, the authors highlight the need to improve clinical reporting of lipid fractions to properly treat LDL-cholesterol and Lp(a) in high-risk patients,” they note.
“The report paves the way for future studies investigating the clinical utility of these additional measurements to initiate and monitor lipid-lowering therapy,” they conclude.
The clinical trial was funded by Ionis Pharmaceuticals, and the direct Lp(a)-cholesterol measurements were funded by Novartis through a research grant to the University of California, San Diego. Dr. Tsimikas is an employee of Ionis Pharmaceuticals and of the University of California, San Diego, and he is a cofounder of Covicept Therapeutics. He is also a coinventor and receives royalties from patents owned by UCSD on oxidation-specific antibodies and on biomarkers related to oxidized lipoproteins, as well as a cofounder and has equity interest in Oxitope and Kleanthi Diagnostics.
A version of this article first appeared on Medscape.com.
A new study has drawn attention to inaccurate measurement of LDL-cholesterol levels in some patients with current assays, which could lead to incorrect therapeutic approaches.
The patient groups most affected are those with high levels of the lipoprotein Lp(a), in whom LDL-cholesterol levels are being overestimated in current laboratory tests, the authors say.
“Current laboratory assays all have the limitation that they cannot measure or report LDL cholesterol accurately. They are actually measuring the combination of LDL and Lp(a),” senior study author Sotirios Tsimikas, MD, University of California, San Diego, explained to this news organization.
“While this may not matter much in individuals with normal Lp(a) levels, in those with elevated Lp(a), the Lp(a) cholesterol may constitute a substantial proportion of the reported LDL cholesterol, and the actual LDL-cholesterol levels could be much lower that the value the lab is telling us,” he said.
Dr. Tsimikas gave the example of a patient with an LDL-cholesterol lab measurement of 75 mg/dL. “If that patient has an Lp(a) level of zero, then they do actually have an LDL level of 75. But as the Lp(a) increases, then the proportion of the result accounted for by LDL cholesterol decreases. So, if a patient with a measured LDL cholesterol of 75 has an Lp(a)-cholesterol level of 20, then their actual LDL level is 55.”
Dr. Tsimikas said it is important to know levels of both lipoproteins individually, so the correct therapeutic approach is used in situations where the Lp(a) cholesterol might be elevated.
“By understanding the actual values of LDL cholesterol and Lp(a) cholesterol, this will allow us to personalize the use of cholesterol-lowering medications and decide where to focus treatment. In the patient with a high level of Lp(a), their residual risk could be coming from Lp(a) cholesterol and less so from LDL cholesterol,” he added. “As we develop drugs to lower Lp(a), this patient might be better off on one of these rather than increasing efforts to lower LDL cholesterol, which might already be at goal.”
The study was published in the March 22 issue of the Journal of the American College of Cardiology.
Dr. Tsimikas noted that Lp(a) is now accepted as a genetic, independent, causal risk factor for cardiovascular disease, but current LDL-lowering drugs do not have much effect on Lp(a).
“Lp(a) can be lowered a little with niacin and PCSK9 inhibitors, but both have a quite a weak effect, and statins increase Lp(a). However, there are now multiple RNA-based therapeutics specifically targeting Lp(a) in clinical development,” he said.
At present, Lp(a) cholesterol has to be mathematically estimated, most commonly with the Dahlén formula, because of the lack of a validated, quantitative method to measure Lp(a) cholesterol, Dr. Tsimikas says.
For the current study, the researchers used a novel, quantitative, sensitive method to directly measure Lp(a) cholesterol, then applied this method to data from a recent study with the one of the new Lp(a)-lowering drugs in development – pelacarsen – which was conducted in patients with elevated Lp(a) levels.
Results showed that direct Lp(a)-cholesterol assessment, and subtracting this value from the laboratory LDL-cholesterol value, provides a more accurate reflection of the baseline and change in LDL cholesterol, the authors report. In the current study, corrected LDL cholesterol was 13 to 16 mg/dL lower than laboratory-reported LDL cholesterol.
Using the corrected LDL-cholesterol results, the study showed that pelacarsen significantly decreases Lp(a) cholesterol, with neutral to modest effects on LDL.
The study also suggests that the current method of calculating Lp(a) cholesterol, and then deriving a corrected LDL cholesterol – the Dahlén formula – is not accurate.
“The Dahlén formula relies on the assumption that Lp(a) cholesterol is universally a fixed 30% of Lp(a) mass, but this usually isn’t the case. The Dahlén formula needs to be discontinued. It can be highly inaccurate,” Dr. Tsimikas said.
Important implications
In an accompanying editorial, Guillaume Paré, MD, Michael Chong, PhD student, and Pedrum Mohammadi-Shemirani, BSc, all of McMaster University, Hamilton, Ont., say the current findings have three important clinical implications.
“First, they provide further proof that in individuals with elevated Lp(a), the contribution of Lp(a)-cholesterol to LDL-cholesterol is non-negligible using standard assays, with 13-16 mg/dL lower LDL-cholesterol post-correction.”
Secondly, the editorialists point out that these new findings confirm that the effect of Lp(a) inhibitors is likely to be mostly confined to Lp(a), “as would be expected.”
Finally, “and perhaps more importantly, the authors highlight the need to improve clinical reporting of lipid fractions to properly treat LDL-cholesterol and Lp(a) in high-risk patients,” they note.
“The report paves the way for future studies investigating the clinical utility of these additional measurements to initiate and monitor lipid-lowering therapy,” they conclude.
The clinical trial was funded by Ionis Pharmaceuticals, and the direct Lp(a)-cholesterol measurements were funded by Novartis through a research grant to the University of California, San Diego. Dr. Tsimikas is an employee of Ionis Pharmaceuticals and of the University of California, San Diego, and he is a cofounder of Covicept Therapeutics. He is also a coinventor and receives royalties from patents owned by UCSD on oxidation-specific antibodies and on biomarkers related to oxidized lipoproteins, as well as a cofounder and has equity interest in Oxitope and Kleanthi Diagnostics.
A version of this article first appeared on Medscape.com.
A new study has drawn attention to inaccurate measurement of LDL-cholesterol levels in some patients with current assays, which could lead to incorrect therapeutic approaches.
The patient groups most affected are those with high levels of the lipoprotein Lp(a), in whom LDL-cholesterol levels are being overestimated in current laboratory tests, the authors say.
“Current laboratory assays all have the limitation that they cannot measure or report LDL cholesterol accurately. They are actually measuring the combination of LDL and Lp(a),” senior study author Sotirios Tsimikas, MD, University of California, San Diego, explained to this news organization.
“While this may not matter much in individuals with normal Lp(a) levels, in those with elevated Lp(a), the Lp(a) cholesterol may constitute a substantial proportion of the reported LDL cholesterol, and the actual LDL-cholesterol levels could be much lower that the value the lab is telling us,” he said.
Dr. Tsimikas gave the example of a patient with an LDL-cholesterol lab measurement of 75 mg/dL. “If that patient has an Lp(a) level of zero, then they do actually have an LDL level of 75. But as the Lp(a) increases, then the proportion of the result accounted for by LDL cholesterol decreases. So, if a patient with a measured LDL cholesterol of 75 has an Lp(a)-cholesterol level of 20, then their actual LDL level is 55.”
Dr. Tsimikas said it is important to know levels of both lipoproteins individually, so the correct therapeutic approach is used in situations where the Lp(a) cholesterol might be elevated.
“By understanding the actual values of LDL cholesterol and Lp(a) cholesterol, this will allow us to personalize the use of cholesterol-lowering medications and decide where to focus treatment. In the patient with a high level of Lp(a), their residual risk could be coming from Lp(a) cholesterol and less so from LDL cholesterol,” he added. “As we develop drugs to lower Lp(a), this patient might be better off on one of these rather than increasing efforts to lower LDL cholesterol, which might already be at goal.”
The study was published in the March 22 issue of the Journal of the American College of Cardiology.
Dr. Tsimikas noted that Lp(a) is now accepted as a genetic, independent, causal risk factor for cardiovascular disease, but current LDL-lowering drugs do not have much effect on Lp(a).
“Lp(a) can be lowered a little with niacin and PCSK9 inhibitors, but both have a quite a weak effect, and statins increase Lp(a). However, there are now multiple RNA-based therapeutics specifically targeting Lp(a) in clinical development,” he said.
At present, Lp(a) cholesterol has to be mathematically estimated, most commonly with the Dahlén formula, because of the lack of a validated, quantitative method to measure Lp(a) cholesterol, Dr. Tsimikas says.
For the current study, the researchers used a novel, quantitative, sensitive method to directly measure Lp(a) cholesterol, then applied this method to data from a recent study with the one of the new Lp(a)-lowering drugs in development – pelacarsen – which was conducted in patients with elevated Lp(a) levels.
Results showed that direct Lp(a)-cholesterol assessment, and subtracting this value from the laboratory LDL-cholesterol value, provides a more accurate reflection of the baseline and change in LDL cholesterol, the authors report. In the current study, corrected LDL cholesterol was 13 to 16 mg/dL lower than laboratory-reported LDL cholesterol.
Using the corrected LDL-cholesterol results, the study showed that pelacarsen significantly decreases Lp(a) cholesterol, with neutral to modest effects on LDL.
The study also suggests that the current method of calculating Lp(a) cholesterol, and then deriving a corrected LDL cholesterol – the Dahlén formula – is not accurate.
“The Dahlén formula relies on the assumption that Lp(a) cholesterol is universally a fixed 30% of Lp(a) mass, but this usually isn’t the case. The Dahlén formula needs to be discontinued. It can be highly inaccurate,” Dr. Tsimikas said.
Important implications
In an accompanying editorial, Guillaume Paré, MD, Michael Chong, PhD student, and Pedrum Mohammadi-Shemirani, BSc, all of McMaster University, Hamilton, Ont., say the current findings have three important clinical implications.
“First, they provide further proof that in individuals with elevated Lp(a), the contribution of Lp(a)-cholesterol to LDL-cholesterol is non-negligible using standard assays, with 13-16 mg/dL lower LDL-cholesterol post-correction.”
Secondly, the editorialists point out that these new findings confirm that the effect of Lp(a) inhibitors is likely to be mostly confined to Lp(a), “as would be expected.”
Finally, “and perhaps more importantly, the authors highlight the need to improve clinical reporting of lipid fractions to properly treat LDL-cholesterol and Lp(a) in high-risk patients,” they note.
“The report paves the way for future studies investigating the clinical utility of these additional measurements to initiate and monitor lipid-lowering therapy,” they conclude.
The clinical trial was funded by Ionis Pharmaceuticals, and the direct Lp(a)-cholesterol measurements were funded by Novartis through a research grant to the University of California, San Diego. Dr. Tsimikas is an employee of Ionis Pharmaceuticals and of the University of California, San Diego, and he is a cofounder of Covicept Therapeutics. He is also a coinventor and receives royalties from patents owned by UCSD on oxidation-specific antibodies and on biomarkers related to oxidized lipoproteins, as well as a cofounder and has equity interest in Oxitope and Kleanthi Diagnostics.
A version of this article first appeared on Medscape.com.
Death of pig heart transplant patient is more a beginning than an end
The genetically altered pig’s heart “worked like a rock star, beautifully functioning,” the surgeon who performed the pioneering Jan. 7 xenotransplant procedure said in a press statement on the death of the patient, David Bennett Sr.
“He wasn’t able to overcome what turned out to be devastating – the debilitation from his previous period of heart failure, which was extreme,” said Bartley P. Griffith, MD, clinical director of the cardiac xenotransplantation program at the University of Maryland, Baltimore.
Representatives of the institution aren’t offering many details on the cause of Mr. Bennett’s death on March 8, 60 days after his operation, but said they will elaborate when their findings are formally published. But their comments seem to downplay the unique nature of the implanted heart itself as a culprit and instead implicate the patient’s diminished overall clinical condition and what grew into an ongoing battle with infections.
The 57-year-old Bennett, bedridden with end-stage heart failure, judged a poor candidate for a ventricular assist device, and on extracorporeal membrane oxygenation (ECMO), reportedly was offered the extraordinary surgery after being turned down for a conventional transplant at several major centers.
“Until day 45 or 50, he was doing very well,” Muhammad M. Mohiuddin, MD, the xenotransplantation program’s scientific director, observed in the statement. But infections soon took advantage of his hobbled immune system.
Given his “preexisting condition and how frail his body was,” Dr. Mohiuddin said, “we were having difficulty maintaining a balance between his immunosuppression and controlling his infection.” Mr. Bennett went into multiple organ failure and “I think that resulted in his passing away.”
Beyond wildest dreams
The surgeons confidently framed Mr. Bennett’s experience as a milestone for heart xenotransplantation. “The demonstration that it was possible, beyond the wildest dreams of most people in the field, even, at this point – that we were able to take a genetically engineered organ and watch it function flawlessly for 9 weeks – is pretty positive in terms of the potential of this therapy,” Dr. Griffith said.
But enough questions linger that others were more circumspect, even as they praised the accomplishment. “There’s no question that this is a historic event,” Mandeep R. Mehra, MD, of Harvard Medical School, and director of the Center for Advanced Heart Disease at Brigham and Women’s Hospital, both in Boston, said in an interview.
Still, “I don’t think we should just conclude that it was the patient’s frailty or death from infection,” Dr. Mehra said. With so few details available, “I would be very careful in prematurely concluding that the problem did not reside with the heart but with the patient. We cannot be sure.”
For example, he noted, “6 to 8 weeks is right around the time when some cardiac complications, like accelerated forms of vasculopathy, could become evident.” Immune-mediated cardiac allograft vasculopathy is a common cause of heart transplant failure.
Or, “it could as easily have been the fact that immunosuppression was modified at 6 to 7 weeks in response to potential infection, which could have led to a cardiac compromise,” Dr. Mehra said. “We just don’t know.”
“It’s really important that this be reported in a scientifically accurate way, because we will all learn from this,” Lori J. West, MD, DPhil, said in an interview.
Little seems to be known for sure about the actual cause of death, “but the fact there was not hyperacute rejection is itself a big step forward. And we know, at least from the limited information we have, that it did not occur,” observed Dr. West, who directs the Alberta Transplant Institute, Edmonton, and the Canadian Donation and Transplantation Research Program. She is a professor of pediatrics with adjunct positions in the departments of surgery and microbiology/immunology.
Dr. West also sees Mr. Bennett’s struggle with infections and adjustments to his unique immunosuppressive regimen, at least as characterized by his care team, as in line with the experience of many heart transplant recipients facing the same threat.
“We already walk this tightrope with every transplant patient,” she said. Typically, they’re put on a somewhat standardized immunosuppressant regimen, “and then we modify it a bit, either increasing or decreasing it, depending on the posttransplant course.” The regimen can become especially intense in response to new signs of rejection, “and you know that that’s going to have an impact on susceptibility to all kinds of infections.”
Full circle
The porcine heart was protected along two fronts against assault from Mr. Bennett’s immune system and other inhospitable aspects of his physiology, either of which could also have been obstacles to success: Genetic modification (Revivicor) of the pig that provided the heart, and a singularly aggressive antirejection drug regimen for the patient.
The knockout of three genes targeting specific porcine cell-surface carbohydrates that provoke a strong human antibody response reportedly averted a hyperacute rejection response that would have caused the graft to fail almost immediately.
Other genetic manipulations, some using CRISPR technology, silenced genes encoded for porcine endogenous retroviruses. Others were aimed at controlling myocardial growth and stemming graft microangiopathy.
Mr. Bennett himself was treated with powerful immunosuppressants, including an investigational anti-CD40 monoclonal antibody (KPL-404, Kiniksa Pharmaceuticals) that, according to UMSOM, inhibits a well-recognized pathway critical to B-cell proliferation, T-cell activation, and antibody production.
“I suspect the patient may not have had rejection, but unfortunately, that intense immunosuppression really set him up – even if he had been half that age – for a very difficult time,” David A. Baran, MD, a cardiologist from Sentara Advanced Heart Failure Center, Norfolk, Va., who studies transplant immunology, said in an interview.
“This is in some ways like the original heart transplant in 1967, when the ability to do the surgery evolved before understanding of the immunosuppression needed. Four or 5 years later, heart transplantation almost died out, before the development of better immunosuppressants like cyclosporine and later tacrolimus,” Dr. Baran said.
“The current age, when we use less immunosuppression than ever, is based on 30 years of progressive success,” he noted. This landmark xenotransplantation “basically turns back the clock to a time when the intensity of immunosuppression by definition had to be extremely high, because we really didn’t know what to expect.”
Emerging role of xeno-organs
Xenotransplantation has been touted as potential strategy for expanding the pool of organs available for transplantation. Mr. Bennett’s “breakthrough surgery” takes the world “one step closer to solving the organ shortage crisis,” his surgeon, Dr. Griffith, announced soon after the procedure. “There are simply not enough donor human hearts available to meet the long list of potential recipients.”
But it’s not the only proposed approach. Measures could be taken, for example, to make more efficient use of the human organs that become available, partly by opening the field to additional less-than-ideal hearts and loosening regulatory mandates for projected graft survival.
“Every year, more than two-thirds of donor organs in the United States are discarded. So it’s not actually that we don’t have enough organs, it’s that we don’t have enough organs that people are willing to take,” Dr. Baran said. Still, it’s important to pursue all promising avenues, and “the genetic manipulation pathway is remarkable.”
But “honestly, organs such as kidneys probably make the most sense” for early study of xenotransplantation from pigs, he said. “The waiting list for kidneys is also very long, but if the kidney graft were to fail, the patient wouldn’t die. It would allow us to work out the immunosuppression without putting patients’ lives at risk.”
Often overlooked in assessments of organ demand, Dr. West said, is that “a lot of patients who could benefit from a transplant will never even be listed for a transplant.” It’s not clear why; perhaps they have multiple comorbidities, live too far from a transplant center, “or they’re too big or too small. Even if there were unlimited organs, you could never meet the needs of people who could benefit from transplantation.”
So even if more available donor organs were used, she said, there would still be a gap that xenotransplantation could help fill. “I’m very much in favor of research that allows us to continue to try to find a pathway to xenotransplantation. I think it’s critically important.”
Unquestionably, “we now need to have a dialogue to entertain how a technology like this, using modern medicine with gene editing, is really going to be utilized,” Dr. Mehra said. The Bennett case “does open up the field, but it also raises caution.” There should be broad participation to move the field forward, “coordinated through either societies or nationally allocated advisory committees that oversee the movement of this technology, to the next step.”
Ideally, that next step “would be to do a safety clinical trial in the right patient,” he said. “And the right patient, by definition, would be one who does not have a life-prolonging option, either mechanical circulatory support or allograft transplantation. That would be the goal.”
Dr. Mehra has reported receiving payments to his institution from Abbott for consulting; consulting fees from Janssen, Mesoblast, Broadview Ventures, Natera, Paragonix, Moderna, and the Baim Institute for Clinical Research; and serving on a scientific advisory board NuPulseCV, Leviticus, and FineHeart. Dr. Baran disclosed consulting for Getinge and LivaNova; speaking for Pfizer; and serving on trial steering committees for CareDx and Procyrion, all unrelated to xenotransplantation. Dr. West has declared no relevant conflicts.
A version of this article first appeared on Medscape.com.
The genetically altered pig’s heart “worked like a rock star, beautifully functioning,” the surgeon who performed the pioneering Jan. 7 xenotransplant procedure said in a press statement on the death of the patient, David Bennett Sr.
“He wasn’t able to overcome what turned out to be devastating – the debilitation from his previous period of heart failure, which was extreme,” said Bartley P. Griffith, MD, clinical director of the cardiac xenotransplantation program at the University of Maryland, Baltimore.
Representatives of the institution aren’t offering many details on the cause of Mr. Bennett’s death on March 8, 60 days after his operation, but said they will elaborate when their findings are formally published. But their comments seem to downplay the unique nature of the implanted heart itself as a culprit and instead implicate the patient’s diminished overall clinical condition and what grew into an ongoing battle with infections.
The 57-year-old Bennett, bedridden with end-stage heart failure, judged a poor candidate for a ventricular assist device, and on extracorporeal membrane oxygenation (ECMO), reportedly was offered the extraordinary surgery after being turned down for a conventional transplant at several major centers.
“Until day 45 or 50, he was doing very well,” Muhammad M. Mohiuddin, MD, the xenotransplantation program’s scientific director, observed in the statement. But infections soon took advantage of his hobbled immune system.
Given his “preexisting condition and how frail his body was,” Dr. Mohiuddin said, “we were having difficulty maintaining a balance between his immunosuppression and controlling his infection.” Mr. Bennett went into multiple organ failure and “I think that resulted in his passing away.”
Beyond wildest dreams
The surgeons confidently framed Mr. Bennett’s experience as a milestone for heart xenotransplantation. “The demonstration that it was possible, beyond the wildest dreams of most people in the field, even, at this point – that we were able to take a genetically engineered organ and watch it function flawlessly for 9 weeks – is pretty positive in terms of the potential of this therapy,” Dr. Griffith said.
But enough questions linger that others were more circumspect, even as they praised the accomplishment. “There’s no question that this is a historic event,” Mandeep R. Mehra, MD, of Harvard Medical School, and director of the Center for Advanced Heart Disease at Brigham and Women’s Hospital, both in Boston, said in an interview.
Still, “I don’t think we should just conclude that it was the patient’s frailty or death from infection,” Dr. Mehra said. With so few details available, “I would be very careful in prematurely concluding that the problem did not reside with the heart but with the patient. We cannot be sure.”
For example, he noted, “6 to 8 weeks is right around the time when some cardiac complications, like accelerated forms of vasculopathy, could become evident.” Immune-mediated cardiac allograft vasculopathy is a common cause of heart transplant failure.
Or, “it could as easily have been the fact that immunosuppression was modified at 6 to 7 weeks in response to potential infection, which could have led to a cardiac compromise,” Dr. Mehra said. “We just don’t know.”
“It’s really important that this be reported in a scientifically accurate way, because we will all learn from this,” Lori J. West, MD, DPhil, said in an interview.
Little seems to be known for sure about the actual cause of death, “but the fact there was not hyperacute rejection is itself a big step forward. And we know, at least from the limited information we have, that it did not occur,” observed Dr. West, who directs the Alberta Transplant Institute, Edmonton, and the Canadian Donation and Transplantation Research Program. She is a professor of pediatrics with adjunct positions in the departments of surgery and microbiology/immunology.
Dr. West also sees Mr. Bennett’s struggle with infections and adjustments to his unique immunosuppressive regimen, at least as characterized by his care team, as in line with the experience of many heart transplant recipients facing the same threat.
“We already walk this tightrope with every transplant patient,” she said. Typically, they’re put on a somewhat standardized immunosuppressant regimen, “and then we modify it a bit, either increasing or decreasing it, depending on the posttransplant course.” The regimen can become especially intense in response to new signs of rejection, “and you know that that’s going to have an impact on susceptibility to all kinds of infections.”
Full circle
The porcine heart was protected along two fronts against assault from Mr. Bennett’s immune system and other inhospitable aspects of his physiology, either of which could also have been obstacles to success: Genetic modification (Revivicor) of the pig that provided the heart, and a singularly aggressive antirejection drug regimen for the patient.
The knockout of three genes targeting specific porcine cell-surface carbohydrates that provoke a strong human antibody response reportedly averted a hyperacute rejection response that would have caused the graft to fail almost immediately.
Other genetic manipulations, some using CRISPR technology, silenced genes encoded for porcine endogenous retroviruses. Others were aimed at controlling myocardial growth and stemming graft microangiopathy.
Mr. Bennett himself was treated with powerful immunosuppressants, including an investigational anti-CD40 monoclonal antibody (KPL-404, Kiniksa Pharmaceuticals) that, according to UMSOM, inhibits a well-recognized pathway critical to B-cell proliferation, T-cell activation, and antibody production.
“I suspect the patient may not have had rejection, but unfortunately, that intense immunosuppression really set him up – even if he had been half that age – for a very difficult time,” David A. Baran, MD, a cardiologist from Sentara Advanced Heart Failure Center, Norfolk, Va., who studies transplant immunology, said in an interview.
“This is in some ways like the original heart transplant in 1967, when the ability to do the surgery evolved before understanding of the immunosuppression needed. Four or 5 years later, heart transplantation almost died out, before the development of better immunosuppressants like cyclosporine and later tacrolimus,” Dr. Baran said.
“The current age, when we use less immunosuppression than ever, is based on 30 years of progressive success,” he noted. This landmark xenotransplantation “basically turns back the clock to a time when the intensity of immunosuppression by definition had to be extremely high, because we really didn’t know what to expect.”
Emerging role of xeno-organs
Xenotransplantation has been touted as potential strategy for expanding the pool of organs available for transplantation. Mr. Bennett’s “breakthrough surgery” takes the world “one step closer to solving the organ shortage crisis,” his surgeon, Dr. Griffith, announced soon after the procedure. “There are simply not enough donor human hearts available to meet the long list of potential recipients.”
But it’s not the only proposed approach. Measures could be taken, for example, to make more efficient use of the human organs that become available, partly by opening the field to additional less-than-ideal hearts and loosening regulatory mandates for projected graft survival.
“Every year, more than two-thirds of donor organs in the United States are discarded. So it’s not actually that we don’t have enough organs, it’s that we don’t have enough organs that people are willing to take,” Dr. Baran said. Still, it’s important to pursue all promising avenues, and “the genetic manipulation pathway is remarkable.”
But “honestly, organs such as kidneys probably make the most sense” for early study of xenotransplantation from pigs, he said. “The waiting list for kidneys is also very long, but if the kidney graft were to fail, the patient wouldn’t die. It would allow us to work out the immunosuppression without putting patients’ lives at risk.”
Often overlooked in assessments of organ demand, Dr. West said, is that “a lot of patients who could benefit from a transplant will never even be listed for a transplant.” It’s not clear why; perhaps they have multiple comorbidities, live too far from a transplant center, “or they’re too big or too small. Even if there were unlimited organs, you could never meet the needs of people who could benefit from transplantation.”
So even if more available donor organs were used, she said, there would still be a gap that xenotransplantation could help fill. “I’m very much in favor of research that allows us to continue to try to find a pathway to xenotransplantation. I think it’s critically important.”
Unquestionably, “we now need to have a dialogue to entertain how a technology like this, using modern medicine with gene editing, is really going to be utilized,” Dr. Mehra said. The Bennett case “does open up the field, but it also raises caution.” There should be broad participation to move the field forward, “coordinated through either societies or nationally allocated advisory committees that oversee the movement of this technology, to the next step.”
Ideally, that next step “would be to do a safety clinical trial in the right patient,” he said. “And the right patient, by definition, would be one who does not have a life-prolonging option, either mechanical circulatory support or allograft transplantation. That would be the goal.”
Dr. Mehra has reported receiving payments to his institution from Abbott for consulting; consulting fees from Janssen, Mesoblast, Broadview Ventures, Natera, Paragonix, Moderna, and the Baim Institute for Clinical Research; and serving on a scientific advisory board NuPulseCV, Leviticus, and FineHeart. Dr. Baran disclosed consulting for Getinge and LivaNova; speaking for Pfizer; and serving on trial steering committees for CareDx and Procyrion, all unrelated to xenotransplantation. Dr. West has declared no relevant conflicts.
A version of this article first appeared on Medscape.com.
The genetically altered pig’s heart “worked like a rock star, beautifully functioning,” the surgeon who performed the pioneering Jan. 7 xenotransplant procedure said in a press statement on the death of the patient, David Bennett Sr.
“He wasn’t able to overcome what turned out to be devastating – the debilitation from his previous period of heart failure, which was extreme,” said Bartley P. Griffith, MD, clinical director of the cardiac xenotransplantation program at the University of Maryland, Baltimore.
Representatives of the institution aren’t offering many details on the cause of Mr. Bennett’s death on March 8, 60 days after his operation, but said they will elaborate when their findings are formally published. But their comments seem to downplay the unique nature of the implanted heart itself as a culprit and instead implicate the patient’s diminished overall clinical condition and what grew into an ongoing battle with infections.
The 57-year-old Bennett, bedridden with end-stage heart failure, judged a poor candidate for a ventricular assist device, and on extracorporeal membrane oxygenation (ECMO), reportedly was offered the extraordinary surgery after being turned down for a conventional transplant at several major centers.
“Until day 45 or 50, he was doing very well,” Muhammad M. Mohiuddin, MD, the xenotransplantation program’s scientific director, observed in the statement. But infections soon took advantage of his hobbled immune system.
Given his “preexisting condition and how frail his body was,” Dr. Mohiuddin said, “we were having difficulty maintaining a balance between his immunosuppression and controlling his infection.” Mr. Bennett went into multiple organ failure and “I think that resulted in his passing away.”
Beyond wildest dreams
The surgeons confidently framed Mr. Bennett’s experience as a milestone for heart xenotransplantation. “The demonstration that it was possible, beyond the wildest dreams of most people in the field, even, at this point – that we were able to take a genetically engineered organ and watch it function flawlessly for 9 weeks – is pretty positive in terms of the potential of this therapy,” Dr. Griffith said.
But enough questions linger that others were more circumspect, even as they praised the accomplishment. “There’s no question that this is a historic event,” Mandeep R. Mehra, MD, of Harvard Medical School, and director of the Center for Advanced Heart Disease at Brigham and Women’s Hospital, both in Boston, said in an interview.
Still, “I don’t think we should just conclude that it was the patient’s frailty or death from infection,” Dr. Mehra said. With so few details available, “I would be very careful in prematurely concluding that the problem did not reside with the heart but with the patient. We cannot be sure.”
For example, he noted, “6 to 8 weeks is right around the time when some cardiac complications, like accelerated forms of vasculopathy, could become evident.” Immune-mediated cardiac allograft vasculopathy is a common cause of heart transplant failure.
Or, “it could as easily have been the fact that immunosuppression was modified at 6 to 7 weeks in response to potential infection, which could have led to a cardiac compromise,” Dr. Mehra said. “We just don’t know.”
“It’s really important that this be reported in a scientifically accurate way, because we will all learn from this,” Lori J. West, MD, DPhil, said in an interview.
Little seems to be known for sure about the actual cause of death, “but the fact there was not hyperacute rejection is itself a big step forward. And we know, at least from the limited information we have, that it did not occur,” observed Dr. West, who directs the Alberta Transplant Institute, Edmonton, and the Canadian Donation and Transplantation Research Program. She is a professor of pediatrics with adjunct positions in the departments of surgery and microbiology/immunology.
Dr. West also sees Mr. Bennett’s struggle with infections and adjustments to his unique immunosuppressive regimen, at least as characterized by his care team, as in line with the experience of many heart transplant recipients facing the same threat.
“We already walk this tightrope with every transplant patient,” she said. Typically, they’re put on a somewhat standardized immunosuppressant regimen, “and then we modify it a bit, either increasing or decreasing it, depending on the posttransplant course.” The regimen can become especially intense in response to new signs of rejection, “and you know that that’s going to have an impact on susceptibility to all kinds of infections.”
Full circle
The porcine heart was protected along two fronts against assault from Mr. Bennett’s immune system and other inhospitable aspects of his physiology, either of which could also have been obstacles to success: Genetic modification (Revivicor) of the pig that provided the heart, and a singularly aggressive antirejection drug regimen for the patient.
The knockout of three genes targeting specific porcine cell-surface carbohydrates that provoke a strong human antibody response reportedly averted a hyperacute rejection response that would have caused the graft to fail almost immediately.
Other genetic manipulations, some using CRISPR technology, silenced genes encoded for porcine endogenous retroviruses. Others were aimed at controlling myocardial growth and stemming graft microangiopathy.
Mr. Bennett himself was treated with powerful immunosuppressants, including an investigational anti-CD40 monoclonal antibody (KPL-404, Kiniksa Pharmaceuticals) that, according to UMSOM, inhibits a well-recognized pathway critical to B-cell proliferation, T-cell activation, and antibody production.
“I suspect the patient may not have had rejection, but unfortunately, that intense immunosuppression really set him up – even if he had been half that age – for a very difficult time,” David A. Baran, MD, a cardiologist from Sentara Advanced Heart Failure Center, Norfolk, Va., who studies transplant immunology, said in an interview.
“This is in some ways like the original heart transplant in 1967, when the ability to do the surgery evolved before understanding of the immunosuppression needed. Four or 5 years later, heart transplantation almost died out, before the development of better immunosuppressants like cyclosporine and later tacrolimus,” Dr. Baran said.
“The current age, when we use less immunosuppression than ever, is based on 30 years of progressive success,” he noted. This landmark xenotransplantation “basically turns back the clock to a time when the intensity of immunosuppression by definition had to be extremely high, because we really didn’t know what to expect.”
Emerging role of xeno-organs
Xenotransplantation has been touted as potential strategy for expanding the pool of organs available for transplantation. Mr. Bennett’s “breakthrough surgery” takes the world “one step closer to solving the organ shortage crisis,” his surgeon, Dr. Griffith, announced soon after the procedure. “There are simply not enough donor human hearts available to meet the long list of potential recipients.”
But it’s not the only proposed approach. Measures could be taken, for example, to make more efficient use of the human organs that become available, partly by opening the field to additional less-than-ideal hearts and loosening regulatory mandates for projected graft survival.
“Every year, more than two-thirds of donor organs in the United States are discarded. So it’s not actually that we don’t have enough organs, it’s that we don’t have enough organs that people are willing to take,” Dr. Baran said. Still, it’s important to pursue all promising avenues, and “the genetic manipulation pathway is remarkable.”
But “honestly, organs such as kidneys probably make the most sense” for early study of xenotransplantation from pigs, he said. “The waiting list for kidneys is also very long, but if the kidney graft were to fail, the patient wouldn’t die. It would allow us to work out the immunosuppression without putting patients’ lives at risk.”
Often overlooked in assessments of organ demand, Dr. West said, is that “a lot of patients who could benefit from a transplant will never even be listed for a transplant.” It’s not clear why; perhaps they have multiple comorbidities, live too far from a transplant center, “or they’re too big or too small. Even if there were unlimited organs, you could never meet the needs of people who could benefit from transplantation.”
So even if more available donor organs were used, she said, there would still be a gap that xenotransplantation could help fill. “I’m very much in favor of research that allows us to continue to try to find a pathway to xenotransplantation. I think it’s critically important.”
Unquestionably, “we now need to have a dialogue to entertain how a technology like this, using modern medicine with gene editing, is really going to be utilized,” Dr. Mehra said. The Bennett case “does open up the field, but it also raises caution.” There should be broad participation to move the field forward, “coordinated through either societies or nationally allocated advisory committees that oversee the movement of this technology, to the next step.”
Ideally, that next step “would be to do a safety clinical trial in the right patient,” he said. “And the right patient, by definition, would be one who does not have a life-prolonging option, either mechanical circulatory support or allograft transplantation. That would be the goal.”
Dr. Mehra has reported receiving payments to his institution from Abbott for consulting; consulting fees from Janssen, Mesoblast, Broadview Ventures, Natera, Paragonix, Moderna, and the Baim Institute for Clinical Research; and serving on a scientific advisory board NuPulseCV, Leviticus, and FineHeart. Dr. Baran disclosed consulting for Getinge and LivaNova; speaking for Pfizer; and serving on trial steering committees for CareDx and Procyrion, all unrelated to xenotransplantation. Dr. West has declared no relevant conflicts.
A version of this article first appeared on Medscape.com.
Early treatment may delay atherosclerosis in familial hypercholesterolemia
Patients with familial hypercholesterolemia (FH) who start lipid-lowering treatment earlier in life may reduce their cardiovascular risk, compared with those who don’t begin treatment early, according to results of a recent meta-analysis.
They showed a difference in the carotid intima-media thickness (IMT) between patients with and without FH that increased with age, but there was also a difference in IMT seen among patients with FH who started treatment early, compared with untreated patients with FH, Kika van Bergen en Henegouwen, of the departments of pediatrics and epidemiology and data science at Amsterdam University Medical Center, and colleagues wrote in their report, published in the Journal of Clinical Lipidology.
“The fact that the difference in IMT increases with age between FH patients and unaffected controls, and is more pronounced in studies with untreated FH patients than in studies with treated patients, suggests that starting treatment already at a young age in patients with FH is preferred,” the researchers wrote. “However, despite treatment, IMT in treated FH patients is still thicker in comparison to subjects without FH.”
The researchers identified 42 studies with among patients with FH and healthy control groups across the MEDLINE, EMBASE and Trials.gov databases up to a cutoff date of April 2020, with 39 studies specifically examining carotid IMT, 2 studies evaluating carotid and femoral IMT, and 1 study evaluating femoral IMT alone. Overall, the researchers examined IMT measurements in 3,796 patients with FH and 2,363 control group participants.
Although data on age and gender for FH and control groups were not available in 6 studies, the mean age ranged from 9 to 57 years for patients with FH and from 8 to 61 years in the control group. Men comprised just under half of both the FH and control groups.
The mean between-group difference in carotid IMT in 34 studies was 0.11 mm (95% confidence interval, 0.06-0.15 mm; P < .001) for patients with FH, compared with the control group, while the mean difference in femoral IMT in three studies was 0.47 mm (95% CI, 0.19-0.74 mm; P < .001) between FH and control groups.
In 13 studies in which data on differences between partly treated and untreated FH were available, there was a significant between-group difference in carotid IMT with partly treated patients with FH, compared with the control group (0.05 mm; 95% CI, 0.03-0.08 mm; P < .001), but a larger mean between-group difference in carotid IMT among untreated patients with FH, compared with a control group (0.12 mm; 95% CI, 0.03-0.21 mm; P = .009).
The researchers also analyzed how age impacts carotid IMT, and they found patients with FH had a mean increase of 0.0018 mm (95% CI, –0.0007 to 0.0042 mm) over a control group in 34 studies. For patients with partly treated FH, compared with patients with untreated FH, the mean between-group increase per year was smaller (0.0023 mm; 95% CI, 0.0021-0.0025 mm), compared with the control group (0.0104 mm; 95% CI, 0.0100-0.0108 mm).
“This sign of residual risk might suggest that more robust cholesterol-lowering treatment and achieving treatment targets, or earlier treatment initiation, is needed to reduce IMT progression to non-FH conditions,” the researchers said. “Therefore, we must find and diagnose these patients, and treat them according to current guidelines.”
Limitations of the authors’ meta-analyses include heterogeneity among studies, differences in IMT measurement protocols, and inclusion of studies with an open-label design. Although randomized clinical trials would be preferable to compare treatment effect, “since statin therapy is indicated in FH patients to reduce [cardiovascular disease], it would be unethical to have a placebo group,” they said.
The authors reported no relevant financial disclosures.
Patients with familial hypercholesterolemia (FH) who start lipid-lowering treatment earlier in life may reduce their cardiovascular risk, compared with those who don’t begin treatment early, according to results of a recent meta-analysis.
They showed a difference in the carotid intima-media thickness (IMT) between patients with and without FH that increased with age, but there was also a difference in IMT seen among patients with FH who started treatment early, compared with untreated patients with FH, Kika van Bergen en Henegouwen, of the departments of pediatrics and epidemiology and data science at Amsterdam University Medical Center, and colleagues wrote in their report, published in the Journal of Clinical Lipidology.
“The fact that the difference in IMT increases with age between FH patients and unaffected controls, and is more pronounced in studies with untreated FH patients than in studies with treated patients, suggests that starting treatment already at a young age in patients with FH is preferred,” the researchers wrote. “However, despite treatment, IMT in treated FH patients is still thicker in comparison to subjects without FH.”
The researchers identified 42 studies with among patients with FH and healthy control groups across the MEDLINE, EMBASE and Trials.gov databases up to a cutoff date of April 2020, with 39 studies specifically examining carotid IMT, 2 studies evaluating carotid and femoral IMT, and 1 study evaluating femoral IMT alone. Overall, the researchers examined IMT measurements in 3,796 patients with FH and 2,363 control group participants.
Although data on age and gender for FH and control groups were not available in 6 studies, the mean age ranged from 9 to 57 years for patients with FH and from 8 to 61 years in the control group. Men comprised just under half of both the FH and control groups.
The mean between-group difference in carotid IMT in 34 studies was 0.11 mm (95% confidence interval, 0.06-0.15 mm; P < .001) for patients with FH, compared with the control group, while the mean difference in femoral IMT in three studies was 0.47 mm (95% CI, 0.19-0.74 mm; P < .001) between FH and control groups.
In 13 studies in which data on differences between partly treated and untreated FH were available, there was a significant between-group difference in carotid IMT with partly treated patients with FH, compared with the control group (0.05 mm; 95% CI, 0.03-0.08 mm; P < .001), but a larger mean between-group difference in carotid IMT among untreated patients with FH, compared with a control group (0.12 mm; 95% CI, 0.03-0.21 mm; P = .009).
The researchers also analyzed how age impacts carotid IMT, and they found patients with FH had a mean increase of 0.0018 mm (95% CI, –0.0007 to 0.0042 mm) over a control group in 34 studies. For patients with partly treated FH, compared with patients with untreated FH, the mean between-group increase per year was smaller (0.0023 mm; 95% CI, 0.0021-0.0025 mm), compared with the control group (0.0104 mm; 95% CI, 0.0100-0.0108 mm).
“This sign of residual risk might suggest that more robust cholesterol-lowering treatment and achieving treatment targets, or earlier treatment initiation, is needed to reduce IMT progression to non-FH conditions,” the researchers said. “Therefore, we must find and diagnose these patients, and treat them according to current guidelines.”
Limitations of the authors’ meta-analyses include heterogeneity among studies, differences in IMT measurement protocols, and inclusion of studies with an open-label design. Although randomized clinical trials would be preferable to compare treatment effect, “since statin therapy is indicated in FH patients to reduce [cardiovascular disease], it would be unethical to have a placebo group,” they said.
The authors reported no relevant financial disclosures.
Patients with familial hypercholesterolemia (FH) who start lipid-lowering treatment earlier in life may reduce their cardiovascular risk, compared with those who don’t begin treatment early, according to results of a recent meta-analysis.
They showed a difference in the carotid intima-media thickness (IMT) between patients with and without FH that increased with age, but there was also a difference in IMT seen among patients with FH who started treatment early, compared with untreated patients with FH, Kika van Bergen en Henegouwen, of the departments of pediatrics and epidemiology and data science at Amsterdam University Medical Center, and colleagues wrote in their report, published in the Journal of Clinical Lipidology.
“The fact that the difference in IMT increases with age between FH patients and unaffected controls, and is more pronounced in studies with untreated FH patients than in studies with treated patients, suggests that starting treatment already at a young age in patients with FH is preferred,” the researchers wrote. “However, despite treatment, IMT in treated FH patients is still thicker in comparison to subjects without FH.”
The researchers identified 42 studies with among patients with FH and healthy control groups across the MEDLINE, EMBASE and Trials.gov databases up to a cutoff date of April 2020, with 39 studies specifically examining carotid IMT, 2 studies evaluating carotid and femoral IMT, and 1 study evaluating femoral IMT alone. Overall, the researchers examined IMT measurements in 3,796 patients with FH and 2,363 control group participants.
Although data on age and gender for FH and control groups were not available in 6 studies, the mean age ranged from 9 to 57 years for patients with FH and from 8 to 61 years in the control group. Men comprised just under half of both the FH and control groups.
The mean between-group difference in carotid IMT in 34 studies was 0.11 mm (95% confidence interval, 0.06-0.15 mm; P < .001) for patients with FH, compared with the control group, while the mean difference in femoral IMT in three studies was 0.47 mm (95% CI, 0.19-0.74 mm; P < .001) between FH and control groups.
In 13 studies in which data on differences between partly treated and untreated FH were available, there was a significant between-group difference in carotid IMT with partly treated patients with FH, compared with the control group (0.05 mm; 95% CI, 0.03-0.08 mm; P < .001), but a larger mean between-group difference in carotid IMT among untreated patients with FH, compared with a control group (0.12 mm; 95% CI, 0.03-0.21 mm; P = .009).
The researchers also analyzed how age impacts carotid IMT, and they found patients with FH had a mean increase of 0.0018 mm (95% CI, –0.0007 to 0.0042 mm) over a control group in 34 studies. For patients with partly treated FH, compared with patients with untreated FH, the mean between-group increase per year was smaller (0.0023 mm; 95% CI, 0.0021-0.0025 mm), compared with the control group (0.0104 mm; 95% CI, 0.0100-0.0108 mm).
“This sign of residual risk might suggest that more robust cholesterol-lowering treatment and achieving treatment targets, or earlier treatment initiation, is needed to reduce IMT progression to non-FH conditions,” the researchers said. “Therefore, we must find and diagnose these patients, and treat them according to current guidelines.”
Limitations of the authors’ meta-analyses include heterogeneity among studies, differences in IMT measurement protocols, and inclusion of studies with an open-label design. Although randomized clinical trials would be preferable to compare treatment effect, “since statin therapy is indicated in FH patients to reduce [cardiovascular disease], it would be unethical to have a placebo group,” they said.
The authors reported no relevant financial disclosures.
FROM THE JOURNAL OF CLINICAL LIPIDOLOGY
Cardiac arrest survival lower in COVID-19 inpatients
Survival after in-hospital cardiac arrest was roughly one-third lower in patients with COVID-19 infections compared to uninfected patients, based on data from nearly 25,000 individuals.
Survival rates of less than 3% were reported in the United States and China for patients who suffered in-hospital cardiac arrest (IHCA) while infected with COVID-19 early in the pandemic, but the data came from small, single-center studies in overwhelmed hospitals, wrote Saket Girotra, MD, of the University of Iowa, Iowa City, and fellow American Heart Association Get With the Guidelines–Resuscitation Investigators. Whether these early reports reflect the broader experience of patients with COVID-19 in hospitals in the United States remains unknown.
In a study published as a research letter in JAMA Network Open, the researchers reviewed data from the American Heart Association Get With the Guidelines–Resuscitation registry. The registry collects detailed information on patients aged 18 years and older who experience cardiac arrest at participating hospitals in the United States. The study population included 24,915 patients aged 18 years and older from 286 hospitals who experienced IHCA during March–December 2020. The mean age of the patients was 64.7 years; 61.1% were White, 24.8% were Black, 3.8% were of other race or ethnicity, and 10.3% were of unknown race or ethnicity.
The primary outcomes were survival to discharge and return of spontaneous circulation (ROSC) for at least 20 minutes.
A total of 5,916 patients (23.7%) had suspected or confirmed COVID-19 infections, and infected patients were more likely to be younger, male, and Black. Patients with COVID-19 infections also were significantly more likely than noninfected patients to have nonshockable rhythm, pneumonia, respiratory insufficiency, or sepsis, and to be on mechanical ventilation or vasopressors when the IHCA occurred, the researchers noted.
Survival rates to hospital discharge were 11.9% for COVID-19 patients, compared with 23.5% for noninfected patients (adjusted relative risk, 0.65; P < .001). ROSC was 53.7% and 63.6%, for infected and noninfected patients, respectively (aRR, 0.86; P < .001).
COVID-19 patients also were more likely than noninfected patients to receive delayed defibrillation, the researchers said. “Although delays in resuscitation, especially defibrillation, may have contributed to lower survival, the negative association of COVID-19 with survival in this study was consistent across subgroups, including patients who received timely treatment with defibrillation and epinephrine.”
The extremely low survival rate in early pandemic studies likely reflected the overwhelming burden on health systems at the time, the researchers said in their discussion.
The study findings were limited by several factors, including potential confounding from unmeasured variables, the use of a quality improvement registry that may not reflect nonparticipating hospitals, and potential false-positive COVID-19 cases. However, the result support findings from recent studies of multiple centers and extend clinical knowledge by comparing infected and noninfected patients from a larger group of hospitals than previously studied, the researchers said.
“We believe that these data will be relevant to health care providers and hospital administrators as the COVID-19 pandemic continues,” they concluded.
Think beyond COVID-19 for cardiac care
“Early during the pandemic, questions were raised whether COVID-19 patients should be treated with CPR,” Dr. Girotra said in an interview. “This was because initial studies had found a dismal survival of 0%-3% in COVID patients treated with CPR. The potential of transmitting the virus to health care professionals during CPR further heightened these concerns. We wanted to know whether the poor survival reported in these initial studies were broadly representative.”
Dr. Girotra said that some of the study findings were surprising. “We found that of all patients with IHCA in 2020 in our study, one in four were suspected or confirmed to have COVID-19 infection. We were surprised by the magnitude of COVID’s impact on the cardiac arrest incidence.”
The implications for clinical decision-making are to think outside of COVID-19 infection, said Dr. Girotra. In the current study, “Although overall survival of cardiac arrest in COVID-positive patients was 30% lower, compared to non-COVID patients, it was not as poor as previously reported. COVID-19 infection alone should not be considered the sole factor for making decisions regarding CPR.
“Over the past 2 decades, we have experienced large gains in survival for in-hospital cardiac arrest. However, the COVID-19 pandemic has eroded these gains,” said Dr. Girotra. “Future studies are needed to monitor the impact of any new variants on cardiac arrest care,” as well as studies “to see whether we return to the prepandemic levels of IHCA survival once the pandemic recedes.”
Dr. Girotra has no relevant financial disclosures.
Survival after in-hospital cardiac arrest was roughly one-third lower in patients with COVID-19 infections compared to uninfected patients, based on data from nearly 25,000 individuals.
Survival rates of less than 3% were reported in the United States and China for patients who suffered in-hospital cardiac arrest (IHCA) while infected with COVID-19 early in the pandemic, but the data came from small, single-center studies in overwhelmed hospitals, wrote Saket Girotra, MD, of the University of Iowa, Iowa City, and fellow American Heart Association Get With the Guidelines–Resuscitation Investigators. Whether these early reports reflect the broader experience of patients with COVID-19 in hospitals in the United States remains unknown.
In a study published as a research letter in JAMA Network Open, the researchers reviewed data from the American Heart Association Get With the Guidelines–Resuscitation registry. The registry collects detailed information on patients aged 18 years and older who experience cardiac arrest at participating hospitals in the United States. The study population included 24,915 patients aged 18 years and older from 286 hospitals who experienced IHCA during March–December 2020. The mean age of the patients was 64.7 years; 61.1% were White, 24.8% were Black, 3.8% were of other race or ethnicity, and 10.3% were of unknown race or ethnicity.
The primary outcomes were survival to discharge and return of spontaneous circulation (ROSC) for at least 20 minutes.
A total of 5,916 patients (23.7%) had suspected or confirmed COVID-19 infections, and infected patients were more likely to be younger, male, and Black. Patients with COVID-19 infections also were significantly more likely than noninfected patients to have nonshockable rhythm, pneumonia, respiratory insufficiency, or sepsis, and to be on mechanical ventilation or vasopressors when the IHCA occurred, the researchers noted.
Survival rates to hospital discharge were 11.9% for COVID-19 patients, compared with 23.5% for noninfected patients (adjusted relative risk, 0.65; P < .001). ROSC was 53.7% and 63.6%, for infected and noninfected patients, respectively (aRR, 0.86; P < .001).
COVID-19 patients also were more likely than noninfected patients to receive delayed defibrillation, the researchers said. “Although delays in resuscitation, especially defibrillation, may have contributed to lower survival, the negative association of COVID-19 with survival in this study was consistent across subgroups, including patients who received timely treatment with defibrillation and epinephrine.”
The extremely low survival rate in early pandemic studies likely reflected the overwhelming burden on health systems at the time, the researchers said in their discussion.
The study findings were limited by several factors, including potential confounding from unmeasured variables, the use of a quality improvement registry that may not reflect nonparticipating hospitals, and potential false-positive COVID-19 cases. However, the result support findings from recent studies of multiple centers and extend clinical knowledge by comparing infected and noninfected patients from a larger group of hospitals than previously studied, the researchers said.
“We believe that these data will be relevant to health care providers and hospital administrators as the COVID-19 pandemic continues,” they concluded.
Think beyond COVID-19 for cardiac care
“Early during the pandemic, questions were raised whether COVID-19 patients should be treated with CPR,” Dr. Girotra said in an interview. “This was because initial studies had found a dismal survival of 0%-3% in COVID patients treated with CPR. The potential of transmitting the virus to health care professionals during CPR further heightened these concerns. We wanted to know whether the poor survival reported in these initial studies were broadly representative.”
Dr. Girotra said that some of the study findings were surprising. “We found that of all patients with IHCA in 2020 in our study, one in four were suspected or confirmed to have COVID-19 infection. We were surprised by the magnitude of COVID’s impact on the cardiac arrest incidence.”
The implications for clinical decision-making are to think outside of COVID-19 infection, said Dr. Girotra. In the current study, “Although overall survival of cardiac arrest in COVID-positive patients was 30% lower, compared to non-COVID patients, it was not as poor as previously reported. COVID-19 infection alone should not be considered the sole factor for making decisions regarding CPR.
“Over the past 2 decades, we have experienced large gains in survival for in-hospital cardiac arrest. However, the COVID-19 pandemic has eroded these gains,” said Dr. Girotra. “Future studies are needed to monitor the impact of any new variants on cardiac arrest care,” as well as studies “to see whether we return to the prepandemic levels of IHCA survival once the pandemic recedes.”
Dr. Girotra has no relevant financial disclosures.
Survival after in-hospital cardiac arrest was roughly one-third lower in patients with COVID-19 infections compared to uninfected patients, based on data from nearly 25,000 individuals.
Survival rates of less than 3% were reported in the United States and China for patients who suffered in-hospital cardiac arrest (IHCA) while infected with COVID-19 early in the pandemic, but the data came from small, single-center studies in overwhelmed hospitals, wrote Saket Girotra, MD, of the University of Iowa, Iowa City, and fellow American Heart Association Get With the Guidelines–Resuscitation Investigators. Whether these early reports reflect the broader experience of patients with COVID-19 in hospitals in the United States remains unknown.
In a study published as a research letter in JAMA Network Open, the researchers reviewed data from the American Heart Association Get With the Guidelines–Resuscitation registry. The registry collects detailed information on patients aged 18 years and older who experience cardiac arrest at participating hospitals in the United States. The study population included 24,915 patients aged 18 years and older from 286 hospitals who experienced IHCA during March–December 2020. The mean age of the patients was 64.7 years; 61.1% were White, 24.8% were Black, 3.8% were of other race or ethnicity, and 10.3% were of unknown race or ethnicity.
The primary outcomes were survival to discharge and return of spontaneous circulation (ROSC) for at least 20 minutes.
A total of 5,916 patients (23.7%) had suspected or confirmed COVID-19 infections, and infected patients were more likely to be younger, male, and Black. Patients with COVID-19 infections also were significantly more likely than noninfected patients to have nonshockable rhythm, pneumonia, respiratory insufficiency, or sepsis, and to be on mechanical ventilation or vasopressors when the IHCA occurred, the researchers noted.
Survival rates to hospital discharge were 11.9% for COVID-19 patients, compared with 23.5% for noninfected patients (adjusted relative risk, 0.65; P < .001). ROSC was 53.7% and 63.6%, for infected and noninfected patients, respectively (aRR, 0.86; P < .001).
COVID-19 patients also were more likely than noninfected patients to receive delayed defibrillation, the researchers said. “Although delays in resuscitation, especially defibrillation, may have contributed to lower survival, the negative association of COVID-19 with survival in this study was consistent across subgroups, including patients who received timely treatment with defibrillation and epinephrine.”
The extremely low survival rate in early pandemic studies likely reflected the overwhelming burden on health systems at the time, the researchers said in their discussion.
The study findings were limited by several factors, including potential confounding from unmeasured variables, the use of a quality improvement registry that may not reflect nonparticipating hospitals, and potential false-positive COVID-19 cases. However, the result support findings from recent studies of multiple centers and extend clinical knowledge by comparing infected and noninfected patients from a larger group of hospitals than previously studied, the researchers said.
“We believe that these data will be relevant to health care providers and hospital administrators as the COVID-19 pandemic continues,” they concluded.
Think beyond COVID-19 for cardiac care
“Early during the pandemic, questions were raised whether COVID-19 patients should be treated with CPR,” Dr. Girotra said in an interview. “This was because initial studies had found a dismal survival of 0%-3% in COVID patients treated with CPR. The potential of transmitting the virus to health care professionals during CPR further heightened these concerns. We wanted to know whether the poor survival reported in these initial studies were broadly representative.”
Dr. Girotra said that some of the study findings were surprising. “We found that of all patients with IHCA in 2020 in our study, one in four were suspected or confirmed to have COVID-19 infection. We were surprised by the magnitude of COVID’s impact on the cardiac arrest incidence.”
The implications for clinical decision-making are to think outside of COVID-19 infection, said Dr. Girotra. In the current study, “Although overall survival of cardiac arrest in COVID-positive patients was 30% lower, compared to non-COVID patients, it was not as poor as previously reported. COVID-19 infection alone should not be considered the sole factor for making decisions regarding CPR.
“Over the past 2 decades, we have experienced large gains in survival for in-hospital cardiac arrest. However, the COVID-19 pandemic has eroded these gains,” said Dr. Girotra. “Future studies are needed to monitor the impact of any new variants on cardiac arrest care,” as well as studies “to see whether we return to the prepandemic levels of IHCA survival once the pandemic recedes.”
Dr. Girotra has no relevant financial disclosures.
FROM JAMA NETWORK OPEN
How Lp(a) can help improve ASCVD risk assessment
A look back at a pair of large cohort studies suggests a telling relation between two distinct predictors of atherosclerotic cardiovascular disease (ASCVD) risk and may offer guidance on how to interpret them together.
Elevated levels of lipoprotein(a), or Lp(a), and high coronary artery calcium (CAC) scores were both predictive of ASCVD risk over 10 years, but independent of each other and a host of more traditional cardiovascular risk factors, for example, in the analysis of data from the MESA (Multi-Ethnic Study of Atherosclerosis) and DHS (Dallas Heart Study) longitudinal cohorts.
Notably, the risk when both Lp(a) and CAC scores were high far exceeded that associated with either marker alone. But when CAC scores were less than 100 Agatston units, predicted ASCVD risk wasn’t influenced by levels of Lp(a). Indeed, a CAC score of 0 predicted the lowest levels of ASCVD risk, even with elevated Lp(a).
That is, the findings suggest, the addition of Lp(a) makes a difference to the risk assessment only when CAC scores are high, at least 100 units, and elevated Lp(a) doesn’t mean increased ASCVD risk in the absence of coronary calcium.
“Our novel findings indicate that elevated Lp(a) drives ASCVD risk independent of the subclinical coronary atherosclerosis burden captured by CAC score,” concluded a report on the analysis, published in the Journal of the American College of Cardiology, with lead author Anurag Mehta, MD, Emory University, Atlanta.
There are no formal recommendations on how to interpret Lp(a) and CAC scores together, but the current findings “provide impetus for measuring Lp(a) in more individuals as part of the shared decision-making process,” the authors contended.
“Really, the calcium score carries the majority of the information in terms of risk, except in the highest CAC score group. That is, if you have a high Lp(a) and a high burden of calcium, your risk is significantly higher than if you just have the high calcium score and the normal Lp(a),” senior author Parag H. Joshi, MD, MHS, said in an interview.
“We thought we would see that the group with higher Lp(a) would have more events over 10 years, even among those who didn’t have coronary calcium,” said Dr. Joshi, of the University of Texas Southwestern Medical Center, Dallas. “But we really don’t see that, at least in a statistically significant way.”
A CAC score of 0 would at least support a more conservative approach in a patient with elevated Lp(a) “who is hesitant to be on a statin or to be more aggressive managing their risk,” Dr. Joshi said.
“This study should be very reassuring for a patient like that,” Ron Blankstein, MD, director of cardiac computed tomography at Brigham and Women’s Hospital, Boston, said in an interview.
“If you have a high Lp(a) and you’re concerned, I think this study really supports the role of calcium scoring for further risk assessment,” said Dr. Blankstein, who is not associated with the new report. “We often check Lp(a) in individuals who perhaps have a family history or who come to see us in a preventive cardiology clinic. If it is high and there is concern, a calcium score can be very helpful. If it’s zero, that really means a very low risk of events. And if it’s elevated, I think we’re going to be more concerned about that patient.”
The current analysis suggests “that, when a patient without clinical cardiovascular disease is identified with either CAC ≥100 or Lp(a) >50 mg/dL, the next step in the risk evaluation should be to measure either Lp(a) or CAC, respectively – if not already performed – to identify the patients at highest risk,” Sotirios Tsimikas, MD, director of vascular medicine at University of California, San Diego, wrote in an accompanying editorial.
“Both Lp(a) and CAC should be more broadly applied in clinical care settings in patients without prior ASCVD to identify those that most likely will benefit from more aggressive therapy and, in the future, from Lp(a)-lowering therapies,” he wrote.
The analyses were conducted separately on data from 4,512 initially asymptomatic patients in MESA and 2,078 from the DHS cohort, who were followed for ASCVD events an average of 13 years and 11 years, respectively. Such events included coronary heart disease–related death, nonfatal MI, and fatal or nonfatal stroke.
In the MESA cohort – 52% women, 36.8% White, 29.3% Black, 22.2% Hispanic, and 11.7% Chinese – elevated Lp(a) (quintile 5 vs. quintiles 1-4) and CAC scores of 1-99 and above 100 (both compared with 0) were each independently associated with increased risk for ASCVD events. The hazard ratio was 1.29 (P = .02) for elevated Lp(a), 1.68 (P < .01) for a CAC score of 1-99, and 2.66 (P < .01) for a CAC score of at least 100.
The corresponding HRs in the DHS cohort were 1.54 (P = .07) for Lp(a), 3.32 (P < .01) for a CAC score of 1-99, and 5.21 (P < .01) for a CAC score of at least 100.
Of note, the authors wrote, ASCVD risk among MESA participants with a CAC score of 0 was not significantly different in those with normal and elevated Lp(a).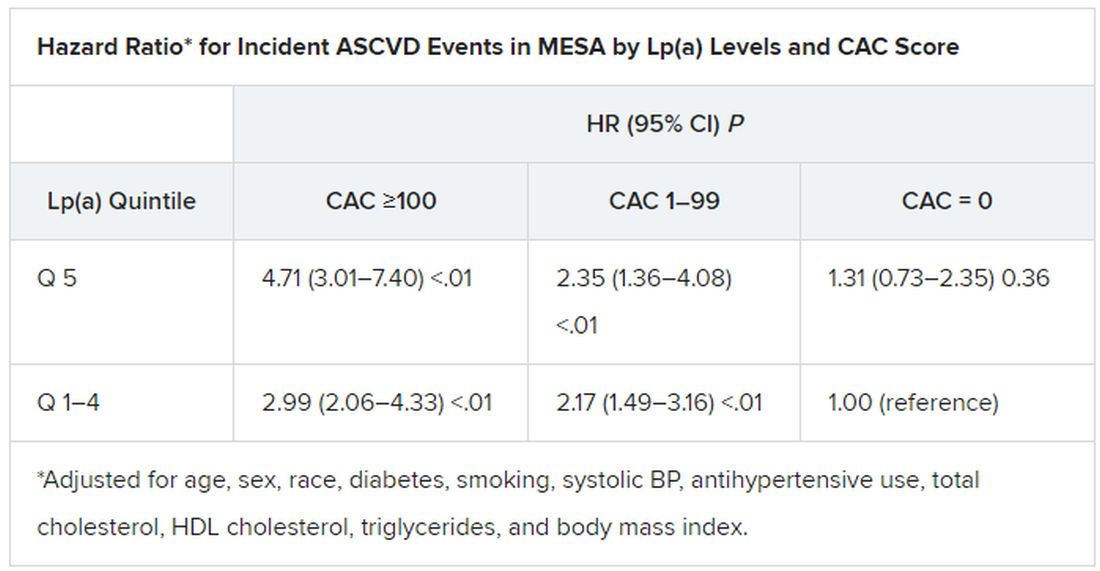
The findings were similar in the corresponding DHS analysis, the authors noted.
When both Lp(a) and CAC scores are considered as dichotomous variables, the highest 10-year ASCVD incidence in MESA was in participants with both elevated Lp(a) (≥50 mg/dL) and a high CAC score (≥100). The lowest risk was seen when Lp(a) was normal (<50 mg/dL) and the CAC score was no more than moderately high (<100).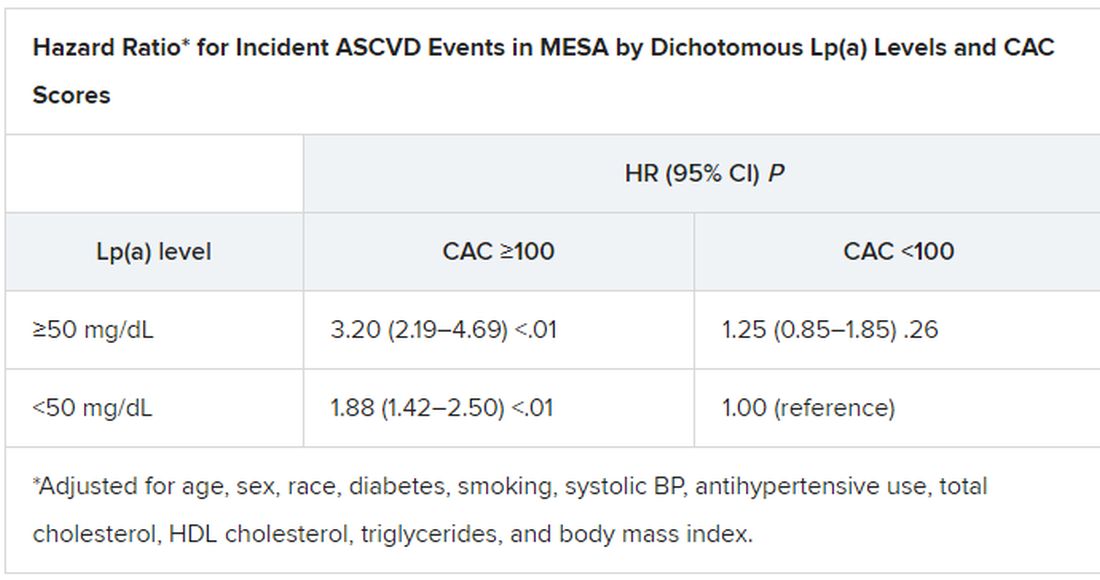
The results in the corresponding DHS analysis, according to the report, again mirrored those from MESA.
“This study has important implications for our patients and also potentially for future clinical trial design,” Dr. Blankstein noted. “A big part of developing a trial in this space is identifying the patients who are at higher risk,” and the current analysis supports CAC scores for identifying the highest-risk patient among those with elevated Lp(a).
Current wisdom is that, for the most part, Lp(a) levels are genetically mediated and are mostly unaffected by interventions such as diet management or exercise. It’s unknown whether reducing elevated Lp(a) levels pharmacologically will cut ASCVD risk, but there are a number of clinical trial programs currently aimed at learning just that. They include the Novartis-sponsored phase 3 HORIZON trial of the antisense agent pelacarsen (TQJ230), with an estimated enrollment of almost 7,700; a randomized, controlled dose-finding study of the small interfering RNA agent olpasiran (AMG890), with 290 patients and funded by Amgen; and an 88-patient phase 1 study of another siRNA agent, SLN360, supported by Silence Therapeutics.
Dr. Mehta reported no relevant relationships. Dr. Joshi has received grant support from Novo Nordisk and consulting income from Bayer and Regeneron; holds equity in G3 Therapeutics; and has served as site investigator for GlaxoSmithKline, Sanofi, AstraZeneca, and Novartis. Dr. Blankstein reported serving as a consultant to Amgen, Novartis, and Silence Therapeutics.
A version of this article first appeared on Medscape.com.
A look back at a pair of large cohort studies suggests a telling relation between two distinct predictors of atherosclerotic cardiovascular disease (ASCVD) risk and may offer guidance on how to interpret them together.
Elevated levels of lipoprotein(a), or Lp(a), and high coronary artery calcium (CAC) scores were both predictive of ASCVD risk over 10 years, but independent of each other and a host of more traditional cardiovascular risk factors, for example, in the analysis of data from the MESA (Multi-Ethnic Study of Atherosclerosis) and DHS (Dallas Heart Study) longitudinal cohorts.
Notably, the risk when both Lp(a) and CAC scores were high far exceeded that associated with either marker alone. But when CAC scores were less than 100 Agatston units, predicted ASCVD risk wasn’t influenced by levels of Lp(a). Indeed, a CAC score of 0 predicted the lowest levels of ASCVD risk, even with elevated Lp(a).
That is, the findings suggest, the addition of Lp(a) makes a difference to the risk assessment only when CAC scores are high, at least 100 units, and elevated Lp(a) doesn’t mean increased ASCVD risk in the absence of coronary calcium.
“Our novel findings indicate that elevated Lp(a) drives ASCVD risk independent of the subclinical coronary atherosclerosis burden captured by CAC score,” concluded a report on the analysis, published in the Journal of the American College of Cardiology, with lead author Anurag Mehta, MD, Emory University, Atlanta.
There are no formal recommendations on how to interpret Lp(a) and CAC scores together, but the current findings “provide impetus for measuring Lp(a) in more individuals as part of the shared decision-making process,” the authors contended.
“Really, the calcium score carries the majority of the information in terms of risk, except in the highest CAC score group. That is, if you have a high Lp(a) and a high burden of calcium, your risk is significantly higher than if you just have the high calcium score and the normal Lp(a),” senior author Parag H. Joshi, MD, MHS, said in an interview.
“We thought we would see that the group with higher Lp(a) would have more events over 10 years, even among those who didn’t have coronary calcium,” said Dr. Joshi, of the University of Texas Southwestern Medical Center, Dallas. “But we really don’t see that, at least in a statistically significant way.”
A CAC score of 0 would at least support a more conservative approach in a patient with elevated Lp(a) “who is hesitant to be on a statin or to be more aggressive managing their risk,” Dr. Joshi said.
“This study should be very reassuring for a patient like that,” Ron Blankstein, MD, director of cardiac computed tomography at Brigham and Women’s Hospital, Boston, said in an interview.
“If you have a high Lp(a) and you’re concerned, I think this study really supports the role of calcium scoring for further risk assessment,” said Dr. Blankstein, who is not associated with the new report. “We often check Lp(a) in individuals who perhaps have a family history or who come to see us in a preventive cardiology clinic. If it is high and there is concern, a calcium score can be very helpful. If it’s zero, that really means a very low risk of events. And if it’s elevated, I think we’re going to be more concerned about that patient.”
The current analysis suggests “that, when a patient without clinical cardiovascular disease is identified with either CAC ≥100 or Lp(a) >50 mg/dL, the next step in the risk evaluation should be to measure either Lp(a) or CAC, respectively – if not already performed – to identify the patients at highest risk,” Sotirios Tsimikas, MD, director of vascular medicine at University of California, San Diego, wrote in an accompanying editorial.
“Both Lp(a) and CAC should be more broadly applied in clinical care settings in patients without prior ASCVD to identify those that most likely will benefit from more aggressive therapy and, in the future, from Lp(a)-lowering therapies,” he wrote.
The analyses were conducted separately on data from 4,512 initially asymptomatic patients in MESA and 2,078 from the DHS cohort, who were followed for ASCVD events an average of 13 years and 11 years, respectively. Such events included coronary heart disease–related death, nonfatal MI, and fatal or nonfatal stroke.
In the MESA cohort – 52% women, 36.8% White, 29.3% Black, 22.2% Hispanic, and 11.7% Chinese – elevated Lp(a) (quintile 5 vs. quintiles 1-4) and CAC scores of 1-99 and above 100 (both compared with 0) were each independently associated with increased risk for ASCVD events. The hazard ratio was 1.29 (P = .02) for elevated Lp(a), 1.68 (P < .01) for a CAC score of 1-99, and 2.66 (P < .01) for a CAC score of at least 100.
The corresponding HRs in the DHS cohort were 1.54 (P = .07) for Lp(a), 3.32 (P < .01) for a CAC score of 1-99, and 5.21 (P < .01) for a CAC score of at least 100.
Of note, the authors wrote, ASCVD risk among MESA participants with a CAC score of 0 was not significantly different in those with normal and elevated Lp(a).
The findings were similar in the corresponding DHS analysis, the authors noted.
When both Lp(a) and CAC scores are considered as dichotomous variables, the highest 10-year ASCVD incidence in MESA was in participants with both elevated Lp(a) (≥50 mg/dL) and a high CAC score (≥100). The lowest risk was seen when Lp(a) was normal (<50 mg/dL) and the CAC score was no more than moderately high (<100).
The results in the corresponding DHS analysis, according to the report, again mirrored those from MESA.
“This study has important implications for our patients and also potentially for future clinical trial design,” Dr. Blankstein noted. “A big part of developing a trial in this space is identifying the patients who are at higher risk,” and the current analysis supports CAC scores for identifying the highest-risk patient among those with elevated Lp(a).
Current wisdom is that, for the most part, Lp(a) levels are genetically mediated and are mostly unaffected by interventions such as diet management or exercise. It’s unknown whether reducing elevated Lp(a) levels pharmacologically will cut ASCVD risk, but there are a number of clinical trial programs currently aimed at learning just that. They include the Novartis-sponsored phase 3 HORIZON trial of the antisense agent pelacarsen (TQJ230), with an estimated enrollment of almost 7,700; a randomized, controlled dose-finding study of the small interfering RNA agent olpasiran (AMG890), with 290 patients and funded by Amgen; and an 88-patient phase 1 study of another siRNA agent, SLN360, supported by Silence Therapeutics.
Dr. Mehta reported no relevant relationships. Dr. Joshi has received grant support from Novo Nordisk and consulting income from Bayer and Regeneron; holds equity in G3 Therapeutics; and has served as site investigator for GlaxoSmithKline, Sanofi, AstraZeneca, and Novartis. Dr. Blankstein reported serving as a consultant to Amgen, Novartis, and Silence Therapeutics.
A version of this article first appeared on Medscape.com.
A look back at a pair of large cohort studies suggests a telling relation between two distinct predictors of atherosclerotic cardiovascular disease (ASCVD) risk and may offer guidance on how to interpret them together.
Elevated levels of lipoprotein(a), or Lp(a), and high coronary artery calcium (CAC) scores were both predictive of ASCVD risk over 10 years, but independent of each other and a host of more traditional cardiovascular risk factors, for example, in the analysis of data from the MESA (Multi-Ethnic Study of Atherosclerosis) and DHS (Dallas Heart Study) longitudinal cohorts.
Notably, the risk when both Lp(a) and CAC scores were high far exceeded that associated with either marker alone. But when CAC scores were less than 100 Agatston units, predicted ASCVD risk wasn’t influenced by levels of Lp(a). Indeed, a CAC score of 0 predicted the lowest levels of ASCVD risk, even with elevated Lp(a).
That is, the findings suggest, the addition of Lp(a) makes a difference to the risk assessment only when CAC scores are high, at least 100 units, and elevated Lp(a) doesn’t mean increased ASCVD risk in the absence of coronary calcium.
“Our novel findings indicate that elevated Lp(a) drives ASCVD risk independent of the subclinical coronary atherosclerosis burden captured by CAC score,” concluded a report on the analysis, published in the Journal of the American College of Cardiology, with lead author Anurag Mehta, MD, Emory University, Atlanta.
There are no formal recommendations on how to interpret Lp(a) and CAC scores together, but the current findings “provide impetus for measuring Lp(a) in more individuals as part of the shared decision-making process,” the authors contended.
“Really, the calcium score carries the majority of the information in terms of risk, except in the highest CAC score group. That is, if you have a high Lp(a) and a high burden of calcium, your risk is significantly higher than if you just have the high calcium score and the normal Lp(a),” senior author Parag H. Joshi, MD, MHS, said in an interview.
“We thought we would see that the group with higher Lp(a) would have more events over 10 years, even among those who didn’t have coronary calcium,” said Dr. Joshi, of the University of Texas Southwestern Medical Center, Dallas. “But we really don’t see that, at least in a statistically significant way.”
A CAC score of 0 would at least support a more conservative approach in a patient with elevated Lp(a) “who is hesitant to be on a statin or to be more aggressive managing their risk,” Dr. Joshi said.
“This study should be very reassuring for a patient like that,” Ron Blankstein, MD, director of cardiac computed tomography at Brigham and Women’s Hospital, Boston, said in an interview.
“If you have a high Lp(a) and you’re concerned, I think this study really supports the role of calcium scoring for further risk assessment,” said Dr. Blankstein, who is not associated with the new report. “We often check Lp(a) in individuals who perhaps have a family history or who come to see us in a preventive cardiology clinic. If it is high and there is concern, a calcium score can be very helpful. If it’s zero, that really means a very low risk of events. And if it’s elevated, I think we’re going to be more concerned about that patient.”
The current analysis suggests “that, when a patient without clinical cardiovascular disease is identified with either CAC ≥100 or Lp(a) >50 mg/dL, the next step in the risk evaluation should be to measure either Lp(a) or CAC, respectively – if not already performed – to identify the patients at highest risk,” Sotirios Tsimikas, MD, director of vascular medicine at University of California, San Diego, wrote in an accompanying editorial.
“Both Lp(a) and CAC should be more broadly applied in clinical care settings in patients without prior ASCVD to identify those that most likely will benefit from more aggressive therapy and, in the future, from Lp(a)-lowering therapies,” he wrote.
The analyses were conducted separately on data from 4,512 initially asymptomatic patients in MESA and 2,078 from the DHS cohort, who were followed for ASCVD events an average of 13 years and 11 years, respectively. Such events included coronary heart disease–related death, nonfatal MI, and fatal or nonfatal stroke.
In the MESA cohort – 52% women, 36.8% White, 29.3% Black, 22.2% Hispanic, and 11.7% Chinese – elevated Lp(a) (quintile 5 vs. quintiles 1-4) and CAC scores of 1-99 and above 100 (both compared with 0) were each independently associated with increased risk for ASCVD events. The hazard ratio was 1.29 (P = .02) for elevated Lp(a), 1.68 (P < .01) for a CAC score of 1-99, and 2.66 (P < .01) for a CAC score of at least 100.
The corresponding HRs in the DHS cohort were 1.54 (P = .07) for Lp(a), 3.32 (P < .01) for a CAC score of 1-99, and 5.21 (P < .01) for a CAC score of at least 100.
Of note, the authors wrote, ASCVD risk among MESA participants with a CAC score of 0 was not significantly different in those with normal and elevated Lp(a).
The findings were similar in the corresponding DHS analysis, the authors noted.
When both Lp(a) and CAC scores are considered as dichotomous variables, the highest 10-year ASCVD incidence in MESA was in participants with both elevated Lp(a) (≥50 mg/dL) and a high CAC score (≥100). The lowest risk was seen when Lp(a) was normal (<50 mg/dL) and the CAC score was no more than moderately high (<100).
The results in the corresponding DHS analysis, according to the report, again mirrored those from MESA.
“This study has important implications for our patients and also potentially for future clinical trial design,” Dr. Blankstein noted. “A big part of developing a trial in this space is identifying the patients who are at higher risk,” and the current analysis supports CAC scores for identifying the highest-risk patient among those with elevated Lp(a).
Current wisdom is that, for the most part, Lp(a) levels are genetically mediated and are mostly unaffected by interventions such as diet management or exercise. It’s unknown whether reducing elevated Lp(a) levels pharmacologically will cut ASCVD risk, but there are a number of clinical trial programs currently aimed at learning just that. They include the Novartis-sponsored phase 3 HORIZON trial of the antisense agent pelacarsen (TQJ230), with an estimated enrollment of almost 7,700; a randomized, controlled dose-finding study of the small interfering RNA agent olpasiran (AMG890), with 290 patients and funded by Amgen; and an 88-patient phase 1 study of another siRNA agent, SLN360, supported by Silence Therapeutics.
Dr. Mehta reported no relevant relationships. Dr. Joshi has received grant support from Novo Nordisk and consulting income from Bayer and Regeneron; holds equity in G3 Therapeutics; and has served as site investigator for GlaxoSmithKline, Sanofi, AstraZeneca, and Novartis. Dr. Blankstein reported serving as a consultant to Amgen, Novartis, and Silence Therapeutics.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
AHA targets ‘low-value’ heart care in new scientific statement
Low-value health care services that provide little or no benefit to patients are “common, potentially harmful, and costly,” and there is a critical need to reduce this kind of care, the American Heart Association said in a newly released scientific statement.
Each year, nearly half of patients in the United States will receive at least one low-value test or procedure, with the attendant risk of avoidable complications from cascades of care and excess costs to individuals and society, the authors noted. Reducing low-value care is particularly important in cardiology, given the high prevalence and costs of cardiovascular disease in the United States.
The statement was published online Feb. 22, 2022, in Circulation: Cardiovascular Quality and Outcomes.
High burden with uncertain benefit
“Cardiovascular disease is common and can present suddenly, such as a heart attack or abnormal heart rhythm,” Vinay Kini, MD, chair of the statement writing group and assistant professor of medicine at Weill Cornell Medicine, New York, said in a news release.
“Our desire to be vigilant about treating and preventing cardiovascular disease may sometimes lead to use of tests and procedures where the benefits to patients may be uncertain,” Dr. Kini said. “This may impose burdens on patients, in the form of increased risk of physical harm from the low-value procedure or potential complications, as well as follow-up care and out-of-pocket financial costs.”
For example, studies have shown that up to one in five echocardiograms and up to half of all stress tests performed in the United States may be rated as rarely appropriate, based on established guidelines for their use.
In addition, up to 15% of percutaneous coronary interventions (PCIs) are classified as rarely appropriate, the writing group said.
Annually, among Medicare fee-for-service beneficiaries, low-value stress testing in patients with stable coronary artery disease is estimated to cost between $212 million and $2.1 billion, while costs of PCI for stable CAD range from $212 million to $2.8 billion, the writing group noted.
“At best, spending on low-value care potentially diverts resources from higher-value services that would benefit patients more effectively at the same or reduced cost. At worst, low-value care results in physical harm in the form of preventable morbidity and mortality,” they said.
“Thus, reducing low-value care is one of the few patient-centered solutions that directly address both the need to control health care spending and the societal imperative to devote its limited resources to beneficial health care services that improve health,” they added.
The group outlines several ways to reduce low-value cardiovascular care targeting patients, providers, and payers/policymakers.
For patients, education and shared decision-making may help reduce low-value care and dispel misconceptions about the intended purpose of test or treatment, they suggested.
For clinicians, a “layered” approach to reducing low-value care may be most effective, such as through education, audit and feedback, and behavioral science tools (“nudges”) to shift behaviors and practices, they said.
For payers and policy leaders, interventions to reduce low-value care include national insurance coverage determinations; prior authorization; alternative payment models that reward lower costs and higher-quality health care; value-based insurance designs that financially penalize low-value care; and medical liability reform to reduce defensive medical practices.
Low-value cardiovascular care is a complex problem, the writing group acknowledged, and achieving meaningful reductions in low-value cardiovascular care will require a multidisciplinary approach that includes continuous research, implementation, evaluation, and adjustment while ensuring equitable access to care.
“Each approach has benefits and drawbacks,” Dr. Kini said. “For example, prior authorization imposes a large burden on health care professionals to obtain insurance approval for tests and treatments. Prior authorization and some value-based payment models may unintentionally worsen existing racial and ethnic health care disparities.
“A one-size-fits-all approach to reducing low-value care is unlikely to succeed; rather, acting through multiple perspectives and frequently measuring impacts and potential unintended consequences is critical,” he concluded.
The scientific statement was prepared by the volunteer writing group on behalf of the AHA’s Council on Quality of Care and Outcomes Research.
The research had no commercial funding. Dr. Kini disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Low-value health care services that provide little or no benefit to patients are “common, potentially harmful, and costly,” and there is a critical need to reduce this kind of care, the American Heart Association said in a newly released scientific statement.
Each year, nearly half of patients in the United States will receive at least one low-value test or procedure, with the attendant risk of avoidable complications from cascades of care and excess costs to individuals and society, the authors noted. Reducing low-value care is particularly important in cardiology, given the high prevalence and costs of cardiovascular disease in the United States.
The statement was published online Feb. 22, 2022, in Circulation: Cardiovascular Quality and Outcomes.
High burden with uncertain benefit
“Cardiovascular disease is common and can present suddenly, such as a heart attack or abnormal heart rhythm,” Vinay Kini, MD, chair of the statement writing group and assistant professor of medicine at Weill Cornell Medicine, New York, said in a news release.
“Our desire to be vigilant about treating and preventing cardiovascular disease may sometimes lead to use of tests and procedures where the benefits to patients may be uncertain,” Dr. Kini said. “This may impose burdens on patients, in the form of increased risk of physical harm from the low-value procedure or potential complications, as well as follow-up care and out-of-pocket financial costs.”
For example, studies have shown that up to one in five echocardiograms and up to half of all stress tests performed in the United States may be rated as rarely appropriate, based on established guidelines for their use.
In addition, up to 15% of percutaneous coronary interventions (PCIs) are classified as rarely appropriate, the writing group said.
Annually, among Medicare fee-for-service beneficiaries, low-value stress testing in patients with stable coronary artery disease is estimated to cost between $212 million and $2.1 billion, while costs of PCI for stable CAD range from $212 million to $2.8 billion, the writing group noted.
“At best, spending on low-value care potentially diverts resources from higher-value services that would benefit patients more effectively at the same or reduced cost. At worst, low-value care results in physical harm in the form of preventable morbidity and mortality,” they said.
“Thus, reducing low-value care is one of the few patient-centered solutions that directly address both the need to control health care spending and the societal imperative to devote its limited resources to beneficial health care services that improve health,” they added.
The group outlines several ways to reduce low-value cardiovascular care targeting patients, providers, and payers/policymakers.
For patients, education and shared decision-making may help reduce low-value care and dispel misconceptions about the intended purpose of test or treatment, they suggested.
For clinicians, a “layered” approach to reducing low-value care may be most effective, such as through education, audit and feedback, and behavioral science tools (“nudges”) to shift behaviors and practices, they said.
For payers and policy leaders, interventions to reduce low-value care include national insurance coverage determinations; prior authorization; alternative payment models that reward lower costs and higher-quality health care; value-based insurance designs that financially penalize low-value care; and medical liability reform to reduce defensive medical practices.
Low-value cardiovascular care is a complex problem, the writing group acknowledged, and achieving meaningful reductions in low-value cardiovascular care will require a multidisciplinary approach that includes continuous research, implementation, evaluation, and adjustment while ensuring equitable access to care.
“Each approach has benefits and drawbacks,” Dr. Kini said. “For example, prior authorization imposes a large burden on health care professionals to obtain insurance approval for tests and treatments. Prior authorization and some value-based payment models may unintentionally worsen existing racial and ethnic health care disparities.
“A one-size-fits-all approach to reducing low-value care is unlikely to succeed; rather, acting through multiple perspectives and frequently measuring impacts and potential unintended consequences is critical,” he concluded.
The scientific statement was prepared by the volunteer writing group on behalf of the AHA’s Council on Quality of Care and Outcomes Research.
The research had no commercial funding. Dr. Kini disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Low-value health care services that provide little or no benefit to patients are “common, potentially harmful, and costly,” and there is a critical need to reduce this kind of care, the American Heart Association said in a newly released scientific statement.
Each year, nearly half of patients in the United States will receive at least one low-value test or procedure, with the attendant risk of avoidable complications from cascades of care and excess costs to individuals and society, the authors noted. Reducing low-value care is particularly important in cardiology, given the high prevalence and costs of cardiovascular disease in the United States.
The statement was published online Feb. 22, 2022, in Circulation: Cardiovascular Quality and Outcomes.
High burden with uncertain benefit
“Cardiovascular disease is common and can present suddenly, such as a heart attack or abnormal heart rhythm,” Vinay Kini, MD, chair of the statement writing group and assistant professor of medicine at Weill Cornell Medicine, New York, said in a news release.
“Our desire to be vigilant about treating and preventing cardiovascular disease may sometimes lead to use of tests and procedures where the benefits to patients may be uncertain,” Dr. Kini said. “This may impose burdens on patients, in the form of increased risk of physical harm from the low-value procedure or potential complications, as well as follow-up care and out-of-pocket financial costs.”
For example, studies have shown that up to one in five echocardiograms and up to half of all stress tests performed in the United States may be rated as rarely appropriate, based on established guidelines for their use.
In addition, up to 15% of percutaneous coronary interventions (PCIs) are classified as rarely appropriate, the writing group said.
Annually, among Medicare fee-for-service beneficiaries, low-value stress testing in patients with stable coronary artery disease is estimated to cost between $212 million and $2.1 billion, while costs of PCI for stable CAD range from $212 million to $2.8 billion, the writing group noted.
“At best, spending on low-value care potentially diverts resources from higher-value services that would benefit patients more effectively at the same or reduced cost. At worst, low-value care results in physical harm in the form of preventable morbidity and mortality,” they said.
“Thus, reducing low-value care is one of the few patient-centered solutions that directly address both the need to control health care spending and the societal imperative to devote its limited resources to beneficial health care services that improve health,” they added.
The group outlines several ways to reduce low-value cardiovascular care targeting patients, providers, and payers/policymakers.
For patients, education and shared decision-making may help reduce low-value care and dispel misconceptions about the intended purpose of test or treatment, they suggested.
For clinicians, a “layered” approach to reducing low-value care may be most effective, such as through education, audit and feedback, and behavioral science tools (“nudges”) to shift behaviors and practices, they said.
For payers and policy leaders, interventions to reduce low-value care include national insurance coverage determinations; prior authorization; alternative payment models that reward lower costs and higher-quality health care; value-based insurance designs that financially penalize low-value care; and medical liability reform to reduce defensive medical practices.
Low-value cardiovascular care is a complex problem, the writing group acknowledged, and achieving meaningful reductions in low-value cardiovascular care will require a multidisciplinary approach that includes continuous research, implementation, evaluation, and adjustment while ensuring equitable access to care.
“Each approach has benefits and drawbacks,” Dr. Kini said. “For example, prior authorization imposes a large burden on health care professionals to obtain insurance approval for tests and treatments. Prior authorization and some value-based payment models may unintentionally worsen existing racial and ethnic health care disparities.
“A one-size-fits-all approach to reducing low-value care is unlikely to succeed; rather, acting through multiple perspectives and frequently measuring impacts and potential unintended consequences is critical,” he concluded.
The scientific statement was prepared by the volunteer writing group on behalf of the AHA’s Council on Quality of Care and Outcomes Research.
The research had no commercial funding. Dr. Kini disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION: CARDIOVASCULAR QUALITY AND OUTCOMES
More evidence links MI to cognitive decline over time
new research suggests.
Although cognition in the acute phase after MI was not different than those without an MI in large observational cohorts, cognitive decline became significantly different over a median 6.5 years of follow-up.
The results reinforce the idea that heart health is closely tied to brain health, lead study author Michelle C. Johansen, MD, PhD, assistant professor of neurology cerebrovascular division, Johns Hopkins University, Baltimore, said in an interview. “From a clinical standpoint, heart health affects brain health and there may be effective interventions to prevent heart attack from happening that could reduce the rate of cognitive decline.”
The study was presented during the International Stroke Conference sponsored by the American Heart Association.
Researchers are increasingly recognizing the vascular contribution to cognitive impairment, said Dr. Johansen. This could involve “silent” or subclinical strokes that go unrecognized until seen on imaging.
The study included 31,377 adults free of MI and dementia from six large, well-known cohort studies: the Atherosclerosis Risk in Communities Study, the Coronary Artery Risk Development in Young Adults Study, the Cardiovascular Health Study, the Framingham Offspring Study, the Multi-Ethnic Study of Atherosclerosis, and the Northern Manhattan Study
About 56% of study participants were women, 23% were Black, 8% were Hispanic, and 69% were White.
They were followed from 1971 to 2017 with investigators repeatedly measuring vascular risk factors. The median study follow-up was 6.5 years, but some were followed for up to 20 years. During that time, there were 1,047 incident MIs.
The researchers performed a pooled analysis from these studies “using some fancy statistical techniques,” said Dr. Johansen. “The unique thing about this study was we were able to harmonize the cognitive measures.”
This allowed the researchers to determine if incident MI affected cognitive decline soon after the event and then long-term after the event. The primary outcome was change in global cognition. Additional outcomes were memory and executive function.
The median time between the first MI and the cognitive assessment was about 1.8 years but ranged from about 6 months to 4 years, said Dr. Johansen. Participants were a median age of 60 years at the time of the first cognitive assessment.
The researchers adjusted results for demographic factors, heart disease risk factors, and cognitive test results prior to the MI. Participants who had a stroke during the follow-up period were excluded from the analysis as stroke can affect cognition.
The study showed incident MI was associated with significant decline in global cognition (–0.71; 95% confidence interval, –1.02 to 0.42; P < .0001) and executive function (–0.68; 95% CI, –0.97 to 0.39; P < .004), but not memory, after the MI.
As cognition naturally declines with age, the researchers took that into consideration. “We anticipated cognition over time was going to go down, which it did, but the question we asked was: ‘How did the slope, which we knew was going to decline over time, compare in people who did not have a MI versus those that did?’ ” said Dr. Johansen.
After adjusting the model accordingly, the effect estimates indicating declines in global cognition and executive function were not significant.
However, another model that looked at the effect of incident MI on decline in cognitive function over the years following the event found significant differences.
Compared with participants without MI, those with incident MI had significantly faster declines in global cognition (–0.15 points/year faster, 95% CI, –0.21 to –0.10; P < .002), memory (–0.13 points/year faster, 95% CI, –0.23 to –0.04; P = .004), and executive function (–0.14 points/year faster, 95% CI, –0.20 to –0.08; P < .0001).
Dr. Johansen surmised that MI may result in subclinical infarcts or inflammation, or that MI and cognitive decline have shared vascular risk factors.
She said she can only speculate about why there was not more of a cognitive decline surrounding the MI. “It may be that right after the event, subjects are kind of sick from other things so it’s hard to see exactly what’s going on. Sometimes people can have other problems just from being in the hospital and having a heart attack may make cognition difficult to assess.”
The researchers also looked at those who had a second MI. “We asked whether the decline we saw after the first heart attack among those who had two heart attacks was explained by the fact they had more than one heart attack, and the answer to that question is no,” Dr. Johansen said.
The next research steps for Dr. Johansen and associates are to look at differences in race and sex.
Karen L. Furie, MD, chair, department of neurology, Brown University, and chief of neurology at Rhode Island Hospital, the Miriam Hospital, and the Bradley Hospital, all in Providence, provided a comment on the research.
MI and cognitive decline have a number of common risk factors, including hypertension, diabetes, high cholesterol, smoking, physical inactivity, and poor diet that can lead to obesity, said Dr. Furie.
“It’s critically important to identify these risk factors as early as possible,” she said. “People in early and middle life may not be receiving optimal medical management or engaging in ideal lifestyle choices and these contribute to the development and progression of atherosclerotic disease over the subsequent decades.”
In theory, she said, if these risk factors were eliminated or adequately treated earlier in life, “both the heart and brain could age naturally and in a healthy manner, enabling a higher functioning and better quality of life.”
The study was funded by the National Institute of Neurological Disorders and Stroke and the National Institute of Aging of the National Institutes of Health. Dr. Johansen receives research funding from NINDS.
A version of this article first appeared on Medscape.com.
new research suggests.
Although cognition in the acute phase after MI was not different than those without an MI in large observational cohorts, cognitive decline became significantly different over a median 6.5 years of follow-up.
The results reinforce the idea that heart health is closely tied to brain health, lead study author Michelle C. Johansen, MD, PhD, assistant professor of neurology cerebrovascular division, Johns Hopkins University, Baltimore, said in an interview. “From a clinical standpoint, heart health affects brain health and there may be effective interventions to prevent heart attack from happening that could reduce the rate of cognitive decline.”
The study was presented during the International Stroke Conference sponsored by the American Heart Association.
Researchers are increasingly recognizing the vascular contribution to cognitive impairment, said Dr. Johansen. This could involve “silent” or subclinical strokes that go unrecognized until seen on imaging.
The study included 31,377 adults free of MI and dementia from six large, well-known cohort studies: the Atherosclerosis Risk in Communities Study, the Coronary Artery Risk Development in Young Adults Study, the Cardiovascular Health Study, the Framingham Offspring Study, the Multi-Ethnic Study of Atherosclerosis, and the Northern Manhattan Study
About 56% of study participants were women, 23% were Black, 8% were Hispanic, and 69% were White.
They were followed from 1971 to 2017 with investigators repeatedly measuring vascular risk factors. The median study follow-up was 6.5 years, but some were followed for up to 20 years. During that time, there were 1,047 incident MIs.
The researchers performed a pooled analysis from these studies “using some fancy statistical techniques,” said Dr. Johansen. “The unique thing about this study was we were able to harmonize the cognitive measures.”
This allowed the researchers to determine if incident MI affected cognitive decline soon after the event and then long-term after the event. The primary outcome was change in global cognition. Additional outcomes were memory and executive function.
The median time between the first MI and the cognitive assessment was about 1.8 years but ranged from about 6 months to 4 years, said Dr. Johansen. Participants were a median age of 60 years at the time of the first cognitive assessment.
The researchers adjusted results for demographic factors, heart disease risk factors, and cognitive test results prior to the MI. Participants who had a stroke during the follow-up period were excluded from the analysis as stroke can affect cognition.
The study showed incident MI was associated with significant decline in global cognition (–0.71; 95% confidence interval, –1.02 to 0.42; P < .0001) and executive function (–0.68; 95% CI, –0.97 to 0.39; P < .004), but not memory, after the MI.
As cognition naturally declines with age, the researchers took that into consideration. “We anticipated cognition over time was going to go down, which it did, but the question we asked was: ‘How did the slope, which we knew was going to decline over time, compare in people who did not have a MI versus those that did?’ ” said Dr. Johansen.
After adjusting the model accordingly, the effect estimates indicating declines in global cognition and executive function were not significant.
However, another model that looked at the effect of incident MI on decline in cognitive function over the years following the event found significant differences.
Compared with participants without MI, those with incident MI had significantly faster declines in global cognition (–0.15 points/year faster, 95% CI, –0.21 to –0.10; P < .002), memory (–0.13 points/year faster, 95% CI, –0.23 to –0.04; P = .004), and executive function (–0.14 points/year faster, 95% CI, –0.20 to –0.08; P < .0001).
Dr. Johansen surmised that MI may result in subclinical infarcts or inflammation, or that MI and cognitive decline have shared vascular risk factors.
She said she can only speculate about why there was not more of a cognitive decline surrounding the MI. “It may be that right after the event, subjects are kind of sick from other things so it’s hard to see exactly what’s going on. Sometimes people can have other problems just from being in the hospital and having a heart attack may make cognition difficult to assess.”
The researchers also looked at those who had a second MI. “We asked whether the decline we saw after the first heart attack among those who had two heart attacks was explained by the fact they had more than one heart attack, and the answer to that question is no,” Dr. Johansen said.
The next research steps for Dr. Johansen and associates are to look at differences in race and sex.
Karen L. Furie, MD, chair, department of neurology, Brown University, and chief of neurology at Rhode Island Hospital, the Miriam Hospital, and the Bradley Hospital, all in Providence, provided a comment on the research.
MI and cognitive decline have a number of common risk factors, including hypertension, diabetes, high cholesterol, smoking, physical inactivity, and poor diet that can lead to obesity, said Dr. Furie.
“It’s critically important to identify these risk factors as early as possible,” she said. “People in early and middle life may not be receiving optimal medical management or engaging in ideal lifestyle choices and these contribute to the development and progression of atherosclerotic disease over the subsequent decades.”
In theory, she said, if these risk factors were eliminated or adequately treated earlier in life, “both the heart and brain could age naturally and in a healthy manner, enabling a higher functioning and better quality of life.”
The study was funded by the National Institute of Neurological Disorders and Stroke and the National Institute of Aging of the National Institutes of Health. Dr. Johansen receives research funding from NINDS.
A version of this article first appeared on Medscape.com.
new research suggests.
Although cognition in the acute phase after MI was not different than those without an MI in large observational cohorts, cognitive decline became significantly different over a median 6.5 years of follow-up.
The results reinforce the idea that heart health is closely tied to brain health, lead study author Michelle C. Johansen, MD, PhD, assistant professor of neurology cerebrovascular division, Johns Hopkins University, Baltimore, said in an interview. “From a clinical standpoint, heart health affects brain health and there may be effective interventions to prevent heart attack from happening that could reduce the rate of cognitive decline.”
The study was presented during the International Stroke Conference sponsored by the American Heart Association.
Researchers are increasingly recognizing the vascular contribution to cognitive impairment, said Dr. Johansen. This could involve “silent” or subclinical strokes that go unrecognized until seen on imaging.
The study included 31,377 adults free of MI and dementia from six large, well-known cohort studies: the Atherosclerosis Risk in Communities Study, the Coronary Artery Risk Development in Young Adults Study, the Cardiovascular Health Study, the Framingham Offspring Study, the Multi-Ethnic Study of Atherosclerosis, and the Northern Manhattan Study
About 56% of study participants were women, 23% were Black, 8% were Hispanic, and 69% were White.
They were followed from 1971 to 2017 with investigators repeatedly measuring vascular risk factors. The median study follow-up was 6.5 years, but some were followed for up to 20 years. During that time, there were 1,047 incident MIs.
The researchers performed a pooled analysis from these studies “using some fancy statistical techniques,” said Dr. Johansen. “The unique thing about this study was we were able to harmonize the cognitive measures.”
This allowed the researchers to determine if incident MI affected cognitive decline soon after the event and then long-term after the event. The primary outcome was change in global cognition. Additional outcomes were memory and executive function.
The median time between the first MI and the cognitive assessment was about 1.8 years but ranged from about 6 months to 4 years, said Dr. Johansen. Participants were a median age of 60 years at the time of the first cognitive assessment.
The researchers adjusted results for demographic factors, heart disease risk factors, and cognitive test results prior to the MI. Participants who had a stroke during the follow-up period were excluded from the analysis as stroke can affect cognition.
The study showed incident MI was associated with significant decline in global cognition (–0.71; 95% confidence interval, –1.02 to 0.42; P < .0001) and executive function (–0.68; 95% CI, –0.97 to 0.39; P < .004), but not memory, after the MI.
As cognition naturally declines with age, the researchers took that into consideration. “We anticipated cognition over time was going to go down, which it did, but the question we asked was: ‘How did the slope, which we knew was going to decline over time, compare in people who did not have a MI versus those that did?’ ” said Dr. Johansen.
After adjusting the model accordingly, the effect estimates indicating declines in global cognition and executive function were not significant.
However, another model that looked at the effect of incident MI on decline in cognitive function over the years following the event found significant differences.
Compared with participants without MI, those with incident MI had significantly faster declines in global cognition (–0.15 points/year faster, 95% CI, –0.21 to –0.10; P < .002), memory (–0.13 points/year faster, 95% CI, –0.23 to –0.04; P = .004), and executive function (–0.14 points/year faster, 95% CI, –0.20 to –0.08; P < .0001).
Dr. Johansen surmised that MI may result in subclinical infarcts or inflammation, or that MI and cognitive decline have shared vascular risk factors.
She said she can only speculate about why there was not more of a cognitive decline surrounding the MI. “It may be that right after the event, subjects are kind of sick from other things so it’s hard to see exactly what’s going on. Sometimes people can have other problems just from being in the hospital and having a heart attack may make cognition difficult to assess.”
The researchers also looked at those who had a second MI. “We asked whether the decline we saw after the first heart attack among those who had two heart attacks was explained by the fact they had more than one heart attack, and the answer to that question is no,” Dr. Johansen said.
The next research steps for Dr. Johansen and associates are to look at differences in race and sex.
Karen L. Furie, MD, chair, department of neurology, Brown University, and chief of neurology at Rhode Island Hospital, the Miriam Hospital, and the Bradley Hospital, all in Providence, provided a comment on the research.
MI and cognitive decline have a number of common risk factors, including hypertension, diabetes, high cholesterol, smoking, physical inactivity, and poor diet that can lead to obesity, said Dr. Furie.
“It’s critically important to identify these risk factors as early as possible,” she said. “People in early and middle life may not be receiving optimal medical management or engaging in ideal lifestyle choices and these contribute to the development and progression of atherosclerotic disease over the subsequent decades.”
In theory, she said, if these risk factors were eliminated or adequately treated earlier in life, “both the heart and brain could age naturally and in a healthy manner, enabling a higher functioning and better quality of life.”
The study was funded by the National Institute of Neurological Disorders and Stroke and the National Institute of Aging of the National Institutes of Health. Dr. Johansen receives research funding from NINDS.
A version of this article first appeared on Medscape.com.
FROM ISC 2022