User login
Long-acting injectable antipsychotics during COVID-19
Long-acting injectable antipsychotics (LAIs) are an essential tool in the treatment of patients with psychotic disorders, allowing for periods of stable drug plasma concentration and confirmed adherence.1 The current coronavirus disease 2019 (COVID-19) pandemic presents unique challenges for administering LAIs and requires a thoughtful and prospective approach in order to ensure continuity of psychiatric care while minimizing the risk of infection with COVID-19. Ideally, patients should be seen in person as infrequently as clinically prudent during this public health emergency; however, LAI administration necessitates direct physical contact between patient and clinician.
Patients with serious mental illness (SMI), who comprise the majority of individuals who receive LAIs, are at heightened risk for cardiovascular and pulmonary comorbidities. These factors are the primary reason the life expectancy of a patient with SMI is nearly 30 years shorter than that of the general population.2-5 The risk of health care workers becoming infected or inadvertently spreading COVID-19 is heightened when working with patients in group living environments (ie, a shelter or group home), who have both increased exposure and increased risk of further transmission.6 Additional patient populations, including older adults, immunocompromised individuals, and those with preexisting conditions, are at heightened risk for serious complications if they were to contract COVID-19.7,8
Thus, the questions of whether LAIs should be administered, and how to do so safely (both during the ongoing, acute phase of the pandemic as well as during the subsequent recovery period until the pandemic abates) need to be carefully considered. In this article, we provide concrete advice for clinicians and clinics on these topics, with the goal of maintaining patients’ psychiatric stability while protecting patients, health care workers, and the broader society from COVID-19 infection. Table 1 summarizes the questions regarding LAIs that clinicians need to address during this crisis. While we focus on outpatient care, inpatient teams should keep these considerations in mind if they are starting and discharging a patient on an LAI. More than ever, close collaboration and communication between inpatient and outpatient teams is critical.
Should an LAI be continued?
An important first step to approaching this challenge is to create a spreadsheet for all patients receiving LAIs. Focusing on a population-based approach is helpful to be systematic and ensure that no patients fall through the cracks during this public health emergency.9 Once all patients have been identified, the treatment team should review each patient to determine if continuing to administer the antipsychotic as an LAI formulation is essential, taking into account the patient’s current psychiatric status, historical medication adherence, potential severity and dangerousness of decompensation if nonadherent, and structures to support stability. For example, can a patient move in with family who can monitor medication adherence during the pandemic? Is it possible for the group home to assume medication administration? Additional consideration should be given to the living environment and health-vulnerability of the patient and the individuals living with them.
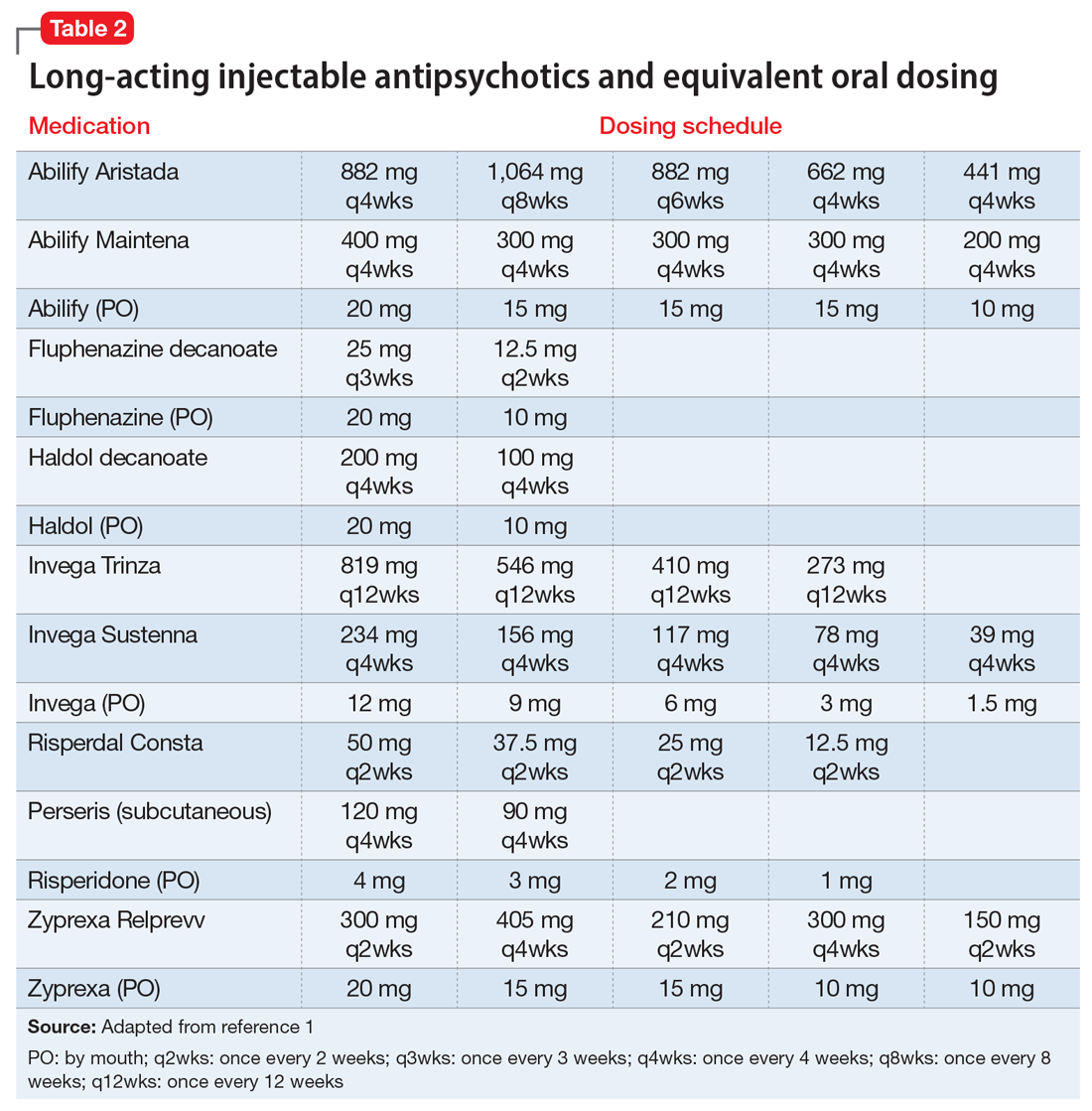
If the risk calculation does not point strongly towards a need for continuing the LAI, it may be prudent to temporarily transition the patient to the corresponding oral antipsychotic preparation. Table 2 lists all LAIs available in the United States and their approximate equivalent oral dosing. It is important to note that such transitions are not without clinical risk, to emphasize to the patient that the transition is intended as a temporary measure, and to discuss a proposed timeline for re-initiating the LAI. Also, emphasize to the patient and family that this transition does not diminish the previous reasoning for needing an LAI, but is a temporary measure taken in light of weighing the risks and benefits during a pandemic.
Which LAI should be administered?
If continuing the LAI is determined to be clinically necessary, consider switching the patient to a longer-acting preparation to maximize intervals between administrations and minimize the potential for infection. From a public health perspective, the longest clinically prudent interval between injections may be the most important consideration, provided the patient can receive a dose necessary to retain stability, and the LAI should be chosen accordingly. Deltoid injections may be able to be administered with reduced contact, or on a “drive-up” basis.10 Consider transitioning a patient who is receiving olanzapine pamoate to an alternate LAI or oral formulation, because the 3-hour observation period that is required after olanzapine pamoate administration is particularly problematic. While it may not be ideal to make medication changes during a pandemic, it is worth carefully weighing the patient’s stability and historical experience with other LAIs to determine if a safer/longer-spaced option is worth trying.11
We recommend only switching among similar antipsychotics (ie, risperidone to paliperidone), or between different preparations of the same drug (ie, Abilify Maintena to Aristada), if possible, as these are the lowest risk transitions with regards to relapse. Table 3 provides examples.

Continue to: When should the LAI be administered?
When should the LAI be administered?
The pharmacokinetics of LAIs allow for some flexibility in terms of when an LAI needs to be administered. The package inserts of all second-generation LAIs include missed-dose guidelines. These guidelines provide information on how long one can wait before the next injection is due, and what additional measures must be taken when beyond that date. Delaying an injection may be prudent, and the missed dose guidelines will indicate when one must consider supplementing with oral medications. For patients who are in quarantine, it may be better to delay an injection until the patient ends their quarantine than to deliver the dose during quarantine. Administering an injection earlier also is usually safe; off-cycle visits may help minimize patient contact (ie, if the patient happens to be coming into the vicinity of the clinic, or requires phlebotomy for therapeutic drug monitoring), and assist in planning for possible resurgences. When appropriate, and after considering the risk of worsening adverse effects, administering a higher dose than the usual maintenance dose would provide a buffer if the next injection was to be delayed. Therapeutic drug monitoring can help to optimize dosing and avoid low plasma drug levels, which may be not be sufficient, particularly during this time of stress.12 To provide optimal protection against relapse, consider administering a dose that puts patients at the higher range of plasma drug levels.
Where can the LAI be administered, and who can give it?
For patients who usually travel to a clinic, consider arranging for a more local injection (ie, at the patient’s primary care clinic in their hometown, or at a local mental health center), and explore if the patient may be able to receive their injection in their home through a visiting nurse association (VNA). In many states (approximately 30 currently), clinicians at pharmacies are also able to administer patient injections. Clinics would do well to at least plan for alternate staffing models in the event of staff illness. A pool of individuals should be available to give injections; consider training additional staff members (including MDs who may have never previously administered an LAI but could be quickly instructed to do so) to administer LAIs. Theoretically, during a public health emergency, family members, particularly those who have a background in health care, could be trained to give an injection and provided education on LAI storage and post-injection monitoring. This approach would not be consistent with FDA labeling, however, and should only be considered as a last resort.
What safety measures can be put in place?
Face-to-face time for injection administration should be kept as brief as possible. Before the encounter, obtain the patient’s clinical information, ideally through telehealth or from an acceptable distance. Medication should be drawn ahead of time, and not in an enclosed space with the patient present. Strongly consider abandoning the traditional enclosed room for the injection, and instead use larger spaces, doorways, or outside, if feasible. As previously noted, some clinics and clinicians have used a drive-up approach for LAI administration, particularly for deltoid injections.10 Individuals who administer the injections should wear personal protective equipment, and the clinic should obtain an adequate supply of this equipment well in advance.
Lessons learned at our clinic
In our community mental health center clinic, planning around these questions has allowed us to provide safe and continuous psychiatric care with LAIs during this public health emergency while reducing the risk of infection. We have worked to transfer LAI administration to VNAs and transition patients to longer-lasting formulations or oral medications where appropriate, which has resulted in an approximately 50% decrease in in-person visits. Reducing the number of in-person visits does not need to result in less frequent clinical follow-up. Telepsychiatry visits can make up for lost in-person visits and have generally been well accepted.
As we are preparing for the next phase, routine medical health monitoring (eg, metabolic monitoring, monitoring for tardive dyskinesia) that has not been at the forefront of concerns should be carefully reintroduced. Challenges encountered have included difficulty in having VNA accept patients for short-term LAI visits, changes to where on the body the injection is delivered, and patients with SMI and their families being reluctant to depart from previous routines and administration schedules.
Continue to: There is great value...
There is great value in the collective lessons learned during this public health emergency (eg, the need for a flexible, population health-based approach; acceptability of combination telehealth and in-person visits) that can lead to more person-centered and accessible care for patients with SMI.
Acknowledgments
The authors thank North Suffolk Mental Health Association, the Freedom Trail Clinic, and their patients.
Bottom Line
When caring for a patient with a psychotic illness during the coronavirus disease 2019 (COVID-19) pandemic, evaluate whether it is necessary to continue a longacting injectable antipsychotic (LAI). If yes, reconsider which LAI should be administered, when and where it should be given, and by whom. Implement safety measures to minimize the risk of COVID-19 exposure and transmission.
Related Resources
- American Association of Community Psychiatrists. Clinical Tip Series. Long acting antipsychotic medications. https://drive.google.com/file/d/1unigjmjFJkqZMbaZ_ftdj8oqog49awZs/view?usp=sharing
- SMI Adviser. What are clinical considerations for giving LAIs during the COVID-19 public health emergency? https://smiadviser.org/knowledge_post/what-are-clinical-considerations-for-giving-lais-during-the-covid-19-public-health-emergency
- CDC. Infection control basics. https://www.cdc.gov/infectioncontrol/basics/index.html
Drug Brand Names
Aripiprazole • Abilify
Aripiprazole for extended- release injectable suspension • Abilify Maintena
Aripiprazole lauroxil • Aristada
Haloperidol • Haldol
Haloperidol injection • Haldol decanoate
Olanzapine • Zyprexa
Olanzapine for extended-release injectable suspension • Zyprexa Relprevv
Paliperidone • Invega
Paliperidone palmitate extended-release injectable suspension • Invega Sustenna
Paliperidone palmitate extended-release injectable suspension • Invega Trinza
Risperidone • Risperdal
Risperidone for extended- release injectable suspension • Perseris
Risperidone injection • Risperdal Consta
1. Freudenreich O. Long-acting injectable antipsychotics. In: Freudenreich O. Psychotic disorders: a practical guide. Springer; 2020:249-261.
2. Olfson M, Gerhard T, Huang C, et al. Premature mortality among adults with schizophrenia in the United States. JAMA Psychiatry. 2015;72(12):1172-1181.
3. Reilly S, Olier I, Planner C, et al. Inequalities in physical comorbidity: a longitudinal comparative cohort study of people with severe mental illness in the UK. BMJ Open. 2015;5(12):e009010.
4. Brown S, Inskip H, Barraclough B. Causes of the excess mortality of schizophrenia. Br J Psychiatry. 2000;177:212-217.
5. Goff DC, Cather C, Evins AE, et al. Medical morbidity and mortality in schizophrenia: guidelines for psychiatrists. J Clin Psychiatry. 2005;66(2):183-194.
6. Baggett TP, Keyes H, Sporn N, et al. Prevalence of SARS-CoV-2 infection in residents of a large homeless shelter in Boston. JAMA. 2020;323(21):2191-2192.
7. CDC COVID-19 Response Team. Severe outcomes among patients with coronavirus disease 2019 (COVID-19)—United States, February 12–March 16, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(12):343-346.
8. Docherty AB, Harrison EM, Green CA, et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985. doi: 10.1136/bmj.m1985
9. Etches V, Frank J, Di Ruggiero E, et al. Measuring population health: a review of indicators. Annu Rev Public Health. 2006;27:29-55.
10. Chepke C. Drive-up pharmacotherapy during the COVID-19 pandemic. Current Psychiatry. 2020;19(5):29-30.
11. Sajatovic M, Ross R, Legacy SN, et al. Initiating/maintaining long-acting injectable antipsychotics in schizophrenia/schizoaffective or bipolar disorder - expert consensus survey part 2. Neuropsychiatr Dis Treat. 2018;14:1475-1492.
12. Schoretsanitis G, Kane JM, Correll CU, et al; American Society of Clinical Psychopharmacology, Pharmakopsychiatrie TTDMTFOTAFNU. Blood levels to optimize antipsychotic treatment in clinical practice: a joint consensus statement of the American Society of Clinical Psychopharmacology and the Therapeutic Drug Monitoring Task Force of the Arbeitsgemeinschaft für Neuropsychopharmakologie und Pharmakopsychiatrie. J Clin Psychiatry. 2020;81(3):19cs13169. doi: 10.4088/JCP.19cs13169
Long-acting injectable antipsychotics (LAIs) are an essential tool in the treatment of patients with psychotic disorders, allowing for periods of stable drug plasma concentration and confirmed adherence.1 The current coronavirus disease 2019 (COVID-19) pandemic presents unique challenges for administering LAIs and requires a thoughtful and prospective approach in order to ensure continuity of psychiatric care while minimizing the risk of infection with COVID-19. Ideally, patients should be seen in person as infrequently as clinically prudent during this public health emergency; however, LAI administration necessitates direct physical contact between patient and clinician.
Patients with serious mental illness (SMI), who comprise the majority of individuals who receive LAIs, are at heightened risk for cardiovascular and pulmonary comorbidities. These factors are the primary reason the life expectancy of a patient with SMI is nearly 30 years shorter than that of the general population.2-5 The risk of health care workers becoming infected or inadvertently spreading COVID-19 is heightened when working with patients in group living environments (ie, a shelter or group home), who have both increased exposure and increased risk of further transmission.6 Additional patient populations, including older adults, immunocompromised individuals, and those with preexisting conditions, are at heightened risk for serious complications if they were to contract COVID-19.7,8
Thus, the questions of whether LAIs should be administered, and how to do so safely (both during the ongoing, acute phase of the pandemic as well as during the subsequent recovery period until the pandemic abates) need to be carefully considered. In this article, we provide concrete advice for clinicians and clinics on these topics, with the goal of maintaining patients’ psychiatric stability while protecting patients, health care workers, and the broader society from COVID-19 infection. Table 1 summarizes the questions regarding LAIs that clinicians need to address during this crisis. While we focus on outpatient care, inpatient teams should keep these considerations in mind if they are starting and discharging a patient on an LAI. More than ever, close collaboration and communication between inpatient and outpatient teams is critical.
Should an LAI be continued?
An important first step to approaching this challenge is to create a spreadsheet for all patients receiving LAIs. Focusing on a population-based approach is helpful to be systematic and ensure that no patients fall through the cracks during this public health emergency.9 Once all patients have been identified, the treatment team should review each patient to determine if continuing to administer the antipsychotic as an LAI formulation is essential, taking into account the patient’s current psychiatric status, historical medication adherence, potential severity and dangerousness of decompensation if nonadherent, and structures to support stability. For example, can a patient move in with family who can monitor medication adherence during the pandemic? Is it possible for the group home to assume medication administration? Additional consideration should be given to the living environment and health-vulnerability of the patient and the individuals living with them.
If the risk calculation does not point strongly towards a need for continuing the LAI, it may be prudent to temporarily transition the patient to the corresponding oral antipsychotic preparation. Table 2 lists all LAIs available in the United States and their approximate equivalent oral dosing. It is important to note that such transitions are not without clinical risk, to emphasize to the patient that the transition is intended as a temporary measure, and to discuss a proposed timeline for re-initiating the LAI. Also, emphasize to the patient and family that this transition does not diminish the previous reasoning for needing an LAI, but is a temporary measure taken in light of weighing the risks and benefits during a pandemic.

Which LAI should be administered?
If continuing the LAI is determined to be clinically necessary, consider switching the patient to a longer-acting preparation to maximize intervals between administrations and minimize the potential for infection. From a public health perspective, the longest clinically prudent interval between injections may be the most important consideration, provided the patient can receive a dose necessary to retain stability, and the LAI should be chosen accordingly. Deltoid injections may be able to be administered with reduced contact, or on a “drive-up” basis.10 Consider transitioning a patient who is receiving olanzapine pamoate to an alternate LAI or oral formulation, because the 3-hour observation period that is required after olanzapine pamoate administration is particularly problematic. While it may not be ideal to make medication changes during a pandemic, it is worth carefully weighing the patient’s stability and historical experience with other LAIs to determine if a safer/longer-spaced option is worth trying.11
We recommend only switching among similar antipsychotics (ie, risperidone to paliperidone), or between different preparations of the same drug (ie, Abilify Maintena to Aristada), if possible, as these are the lowest risk transitions with regards to relapse. Table 3 provides examples.

Continue to: When should the LAI be administered?
When should the LAI be administered?
The pharmacokinetics of LAIs allow for some flexibility in terms of when an LAI needs to be administered. The package inserts of all second-generation LAIs include missed-dose guidelines. These guidelines provide information on how long one can wait before the next injection is due, and what additional measures must be taken when beyond that date. Delaying an injection may be prudent, and the missed dose guidelines will indicate when one must consider supplementing with oral medications. For patients who are in quarantine, it may be better to delay an injection until the patient ends their quarantine than to deliver the dose during quarantine. Administering an injection earlier also is usually safe; off-cycle visits may help minimize patient contact (ie, if the patient happens to be coming into the vicinity of the clinic, or requires phlebotomy for therapeutic drug monitoring), and assist in planning for possible resurgences. When appropriate, and after considering the risk of worsening adverse effects, administering a higher dose than the usual maintenance dose would provide a buffer if the next injection was to be delayed. Therapeutic drug monitoring can help to optimize dosing and avoid low plasma drug levels, which may be not be sufficient, particularly during this time of stress.12 To provide optimal protection against relapse, consider administering a dose that puts patients at the higher range of plasma drug levels.
Where can the LAI be administered, and who can give it?
For patients who usually travel to a clinic, consider arranging for a more local injection (ie, at the patient’s primary care clinic in their hometown, or at a local mental health center), and explore if the patient may be able to receive their injection in their home through a visiting nurse association (VNA). In many states (approximately 30 currently), clinicians at pharmacies are also able to administer patient injections. Clinics would do well to at least plan for alternate staffing models in the event of staff illness. A pool of individuals should be available to give injections; consider training additional staff members (including MDs who may have never previously administered an LAI but could be quickly instructed to do so) to administer LAIs. Theoretically, during a public health emergency, family members, particularly those who have a background in health care, could be trained to give an injection and provided education on LAI storage and post-injection monitoring. This approach would not be consistent with FDA labeling, however, and should only be considered as a last resort.
What safety measures can be put in place?
Face-to-face time for injection administration should be kept as brief as possible. Before the encounter, obtain the patient’s clinical information, ideally through telehealth or from an acceptable distance. Medication should be drawn ahead of time, and not in an enclosed space with the patient present. Strongly consider abandoning the traditional enclosed room for the injection, and instead use larger spaces, doorways, or outside, if feasible. As previously noted, some clinics and clinicians have used a drive-up approach for LAI administration, particularly for deltoid injections.10 Individuals who administer the injections should wear personal protective equipment, and the clinic should obtain an adequate supply of this equipment well in advance.
Lessons learned at our clinic
In our community mental health center clinic, planning around these questions has allowed us to provide safe and continuous psychiatric care with LAIs during this public health emergency while reducing the risk of infection. We have worked to transfer LAI administration to VNAs and transition patients to longer-lasting formulations or oral medications where appropriate, which has resulted in an approximately 50% decrease in in-person visits. Reducing the number of in-person visits does not need to result in less frequent clinical follow-up. Telepsychiatry visits can make up for lost in-person visits and have generally been well accepted.
As we are preparing for the next phase, routine medical health monitoring (eg, metabolic monitoring, monitoring for tardive dyskinesia) that has not been at the forefront of concerns should be carefully reintroduced. Challenges encountered have included difficulty in having VNA accept patients for short-term LAI visits, changes to where on the body the injection is delivered, and patients with SMI and their families being reluctant to depart from previous routines and administration schedules.
Continue to: There is great value...
There is great value in the collective lessons learned during this public health emergency (eg, the need for a flexible, population health-based approach; acceptability of combination telehealth and in-person visits) that can lead to more person-centered and accessible care for patients with SMI.
Acknowledgments
The authors thank North Suffolk Mental Health Association, the Freedom Trail Clinic, and their patients.
Bottom Line
When caring for a patient with a psychotic illness during the coronavirus disease 2019 (COVID-19) pandemic, evaluate whether it is necessary to continue a longacting injectable antipsychotic (LAI). If yes, reconsider which LAI should be administered, when and where it should be given, and by whom. Implement safety measures to minimize the risk of COVID-19 exposure and transmission.
Related Resources
- American Association of Community Psychiatrists. Clinical Tip Series. Long acting antipsychotic medications. https://drive.google.com/file/d/1unigjmjFJkqZMbaZ_ftdj8oqog49awZs/view?usp=sharing
- SMI Adviser. What are clinical considerations for giving LAIs during the COVID-19 public health emergency? https://smiadviser.org/knowledge_post/what-are-clinical-considerations-for-giving-lais-during-the-covid-19-public-health-emergency
- CDC. Infection control basics. https://www.cdc.gov/infectioncontrol/basics/index.html
Drug Brand Names
Aripiprazole • Abilify
Aripiprazole for extended- release injectable suspension • Abilify Maintena
Aripiprazole lauroxil • Aristada
Haloperidol • Haldol
Haloperidol injection • Haldol decanoate
Olanzapine • Zyprexa
Olanzapine for extended-release injectable suspension • Zyprexa Relprevv
Paliperidone • Invega
Paliperidone palmitate extended-release injectable suspension • Invega Sustenna
Paliperidone palmitate extended-release injectable suspension • Invega Trinza
Risperidone • Risperdal
Risperidone for extended- release injectable suspension • Perseris
Risperidone injection • Risperdal Consta
Long-acting injectable antipsychotics (LAIs) are an essential tool in the treatment of patients with psychotic disorders, allowing for periods of stable drug plasma concentration and confirmed adherence.1 The current coronavirus disease 2019 (COVID-19) pandemic presents unique challenges for administering LAIs and requires a thoughtful and prospective approach in order to ensure continuity of psychiatric care while minimizing the risk of infection with COVID-19. Ideally, patients should be seen in person as infrequently as clinically prudent during this public health emergency; however, LAI administration necessitates direct physical contact between patient and clinician.
Patients with serious mental illness (SMI), who comprise the majority of individuals who receive LAIs, are at heightened risk for cardiovascular and pulmonary comorbidities. These factors are the primary reason the life expectancy of a patient with SMI is nearly 30 years shorter than that of the general population.2-5 The risk of health care workers becoming infected or inadvertently spreading COVID-19 is heightened when working with patients in group living environments (ie, a shelter or group home), who have both increased exposure and increased risk of further transmission.6 Additional patient populations, including older adults, immunocompromised individuals, and those with preexisting conditions, are at heightened risk for serious complications if they were to contract COVID-19.7,8
Thus, the questions of whether LAIs should be administered, and how to do so safely (both during the ongoing, acute phase of the pandemic as well as during the subsequent recovery period until the pandemic abates) need to be carefully considered. In this article, we provide concrete advice for clinicians and clinics on these topics, with the goal of maintaining patients’ psychiatric stability while protecting patients, health care workers, and the broader society from COVID-19 infection. Table 1 summarizes the questions regarding LAIs that clinicians need to address during this crisis. While we focus on outpatient care, inpatient teams should keep these considerations in mind if they are starting and discharging a patient on an LAI. More than ever, close collaboration and communication between inpatient and outpatient teams is critical.
Should an LAI be continued?
An important first step to approaching this challenge is to create a spreadsheet for all patients receiving LAIs. Focusing on a population-based approach is helpful to be systematic and ensure that no patients fall through the cracks during this public health emergency.9 Once all patients have been identified, the treatment team should review each patient to determine if continuing to administer the antipsychotic as an LAI formulation is essential, taking into account the patient’s current psychiatric status, historical medication adherence, potential severity and dangerousness of decompensation if nonadherent, and structures to support stability. For example, can a patient move in with family who can monitor medication adherence during the pandemic? Is it possible for the group home to assume medication administration? Additional consideration should be given to the living environment and health-vulnerability of the patient and the individuals living with them.
If the risk calculation does not point strongly towards a need for continuing the LAI, it may be prudent to temporarily transition the patient to the corresponding oral antipsychotic preparation. Table 2 lists all LAIs available in the United States and their approximate equivalent oral dosing. It is important to note that such transitions are not without clinical risk, to emphasize to the patient that the transition is intended as a temporary measure, and to discuss a proposed timeline for re-initiating the LAI. Also, emphasize to the patient and family that this transition does not diminish the previous reasoning for needing an LAI, but is a temporary measure taken in light of weighing the risks and benefits during a pandemic.

Which LAI should be administered?
If continuing the LAI is determined to be clinically necessary, consider switching the patient to a longer-acting preparation to maximize intervals between administrations and minimize the potential for infection. From a public health perspective, the longest clinically prudent interval between injections may be the most important consideration, provided the patient can receive a dose necessary to retain stability, and the LAI should be chosen accordingly. Deltoid injections may be able to be administered with reduced contact, or on a “drive-up” basis.10 Consider transitioning a patient who is receiving olanzapine pamoate to an alternate LAI or oral formulation, because the 3-hour observation period that is required after olanzapine pamoate administration is particularly problematic. While it may not be ideal to make medication changes during a pandemic, it is worth carefully weighing the patient’s stability and historical experience with other LAIs to determine if a safer/longer-spaced option is worth trying.11
We recommend only switching among similar antipsychotics (ie, risperidone to paliperidone), or between different preparations of the same drug (ie, Abilify Maintena to Aristada), if possible, as these are the lowest risk transitions with regards to relapse. Table 3 provides examples.

Continue to: When should the LAI be administered?
When should the LAI be administered?
The pharmacokinetics of LAIs allow for some flexibility in terms of when an LAI needs to be administered. The package inserts of all second-generation LAIs include missed-dose guidelines. These guidelines provide information on how long one can wait before the next injection is due, and what additional measures must be taken when beyond that date. Delaying an injection may be prudent, and the missed dose guidelines will indicate when one must consider supplementing with oral medications. For patients who are in quarantine, it may be better to delay an injection until the patient ends their quarantine than to deliver the dose during quarantine. Administering an injection earlier also is usually safe; off-cycle visits may help minimize patient contact (ie, if the patient happens to be coming into the vicinity of the clinic, or requires phlebotomy for therapeutic drug monitoring), and assist in planning for possible resurgences. When appropriate, and after considering the risk of worsening adverse effects, administering a higher dose than the usual maintenance dose would provide a buffer if the next injection was to be delayed. Therapeutic drug monitoring can help to optimize dosing and avoid low plasma drug levels, which may be not be sufficient, particularly during this time of stress.12 To provide optimal protection against relapse, consider administering a dose that puts patients at the higher range of plasma drug levels.
Where can the LAI be administered, and who can give it?
For patients who usually travel to a clinic, consider arranging for a more local injection (ie, at the patient’s primary care clinic in their hometown, or at a local mental health center), and explore if the patient may be able to receive their injection in their home through a visiting nurse association (VNA). In many states (approximately 30 currently), clinicians at pharmacies are also able to administer patient injections. Clinics would do well to at least plan for alternate staffing models in the event of staff illness. A pool of individuals should be available to give injections; consider training additional staff members (including MDs who may have never previously administered an LAI but could be quickly instructed to do so) to administer LAIs. Theoretically, during a public health emergency, family members, particularly those who have a background in health care, could be trained to give an injection and provided education on LAI storage and post-injection monitoring. This approach would not be consistent with FDA labeling, however, and should only be considered as a last resort.
What safety measures can be put in place?
Face-to-face time for injection administration should be kept as brief as possible. Before the encounter, obtain the patient’s clinical information, ideally through telehealth or from an acceptable distance. Medication should be drawn ahead of time, and not in an enclosed space with the patient present. Strongly consider abandoning the traditional enclosed room for the injection, and instead use larger spaces, doorways, or outside, if feasible. As previously noted, some clinics and clinicians have used a drive-up approach for LAI administration, particularly for deltoid injections.10 Individuals who administer the injections should wear personal protective equipment, and the clinic should obtain an adequate supply of this equipment well in advance.
Lessons learned at our clinic
In our community mental health center clinic, planning around these questions has allowed us to provide safe and continuous psychiatric care with LAIs during this public health emergency while reducing the risk of infection. We have worked to transfer LAI administration to VNAs and transition patients to longer-lasting formulations or oral medications where appropriate, which has resulted in an approximately 50% decrease in in-person visits. Reducing the number of in-person visits does not need to result in less frequent clinical follow-up. Telepsychiatry visits can make up for lost in-person visits and have generally been well accepted.
As we are preparing for the next phase, routine medical health monitoring (eg, metabolic monitoring, monitoring for tardive dyskinesia) that has not been at the forefront of concerns should be carefully reintroduced. Challenges encountered have included difficulty in having VNA accept patients for short-term LAI visits, changes to where on the body the injection is delivered, and patients with SMI and their families being reluctant to depart from previous routines and administration schedules.
Continue to: There is great value...
There is great value in the collective lessons learned during this public health emergency (eg, the need for a flexible, population health-based approach; acceptability of combination telehealth and in-person visits) that can lead to more person-centered and accessible care for patients with SMI.
Acknowledgments
The authors thank North Suffolk Mental Health Association, the Freedom Trail Clinic, and their patients.
Bottom Line
When caring for a patient with a psychotic illness during the coronavirus disease 2019 (COVID-19) pandemic, evaluate whether it is necessary to continue a longacting injectable antipsychotic (LAI). If yes, reconsider which LAI should be administered, when and where it should be given, and by whom. Implement safety measures to minimize the risk of COVID-19 exposure and transmission.
Related Resources
- American Association of Community Psychiatrists. Clinical Tip Series. Long acting antipsychotic medications. https://drive.google.com/file/d/1unigjmjFJkqZMbaZ_ftdj8oqog49awZs/view?usp=sharing
- SMI Adviser. What are clinical considerations for giving LAIs during the COVID-19 public health emergency? https://smiadviser.org/knowledge_post/what-are-clinical-considerations-for-giving-lais-during-the-covid-19-public-health-emergency
- CDC. Infection control basics. https://www.cdc.gov/infectioncontrol/basics/index.html
Drug Brand Names
Aripiprazole • Abilify
Aripiprazole for extended- release injectable suspension • Abilify Maintena
Aripiprazole lauroxil • Aristada
Haloperidol • Haldol
Haloperidol injection • Haldol decanoate
Olanzapine • Zyprexa
Olanzapine for extended-release injectable suspension • Zyprexa Relprevv
Paliperidone • Invega
Paliperidone palmitate extended-release injectable suspension • Invega Sustenna
Paliperidone palmitate extended-release injectable suspension • Invega Trinza
Risperidone • Risperdal
Risperidone for extended- release injectable suspension • Perseris
Risperidone injection • Risperdal Consta
1. Freudenreich O. Long-acting injectable antipsychotics. In: Freudenreich O. Psychotic disorders: a practical guide. Springer; 2020:249-261.
2. Olfson M, Gerhard T, Huang C, et al. Premature mortality among adults with schizophrenia in the United States. JAMA Psychiatry. 2015;72(12):1172-1181.
3. Reilly S, Olier I, Planner C, et al. Inequalities in physical comorbidity: a longitudinal comparative cohort study of people with severe mental illness in the UK. BMJ Open. 2015;5(12):e009010.
4. Brown S, Inskip H, Barraclough B. Causes of the excess mortality of schizophrenia. Br J Psychiatry. 2000;177:212-217.
5. Goff DC, Cather C, Evins AE, et al. Medical morbidity and mortality in schizophrenia: guidelines for psychiatrists. J Clin Psychiatry. 2005;66(2):183-194.
6. Baggett TP, Keyes H, Sporn N, et al. Prevalence of SARS-CoV-2 infection in residents of a large homeless shelter in Boston. JAMA. 2020;323(21):2191-2192.
7. CDC COVID-19 Response Team. Severe outcomes among patients with coronavirus disease 2019 (COVID-19)—United States, February 12–March 16, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(12):343-346.
8. Docherty AB, Harrison EM, Green CA, et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985. doi: 10.1136/bmj.m1985
9. Etches V, Frank J, Di Ruggiero E, et al. Measuring population health: a review of indicators. Annu Rev Public Health. 2006;27:29-55.
10. Chepke C. Drive-up pharmacotherapy during the COVID-19 pandemic. Current Psychiatry. 2020;19(5):29-30.
11. Sajatovic M, Ross R, Legacy SN, et al. Initiating/maintaining long-acting injectable antipsychotics in schizophrenia/schizoaffective or bipolar disorder - expert consensus survey part 2. Neuropsychiatr Dis Treat. 2018;14:1475-1492.
12. Schoretsanitis G, Kane JM, Correll CU, et al; American Society of Clinical Psychopharmacology, Pharmakopsychiatrie TTDMTFOTAFNU. Blood levels to optimize antipsychotic treatment in clinical practice: a joint consensus statement of the American Society of Clinical Psychopharmacology and the Therapeutic Drug Monitoring Task Force of the Arbeitsgemeinschaft für Neuropsychopharmakologie und Pharmakopsychiatrie. J Clin Psychiatry. 2020;81(3):19cs13169. doi: 10.4088/JCP.19cs13169
1. Freudenreich O. Long-acting injectable antipsychotics. In: Freudenreich O. Psychotic disorders: a practical guide. Springer; 2020:249-261.
2. Olfson M, Gerhard T, Huang C, et al. Premature mortality among adults with schizophrenia in the United States. JAMA Psychiatry. 2015;72(12):1172-1181.
3. Reilly S, Olier I, Planner C, et al. Inequalities in physical comorbidity: a longitudinal comparative cohort study of people with severe mental illness in the UK. BMJ Open. 2015;5(12):e009010.
4. Brown S, Inskip H, Barraclough B. Causes of the excess mortality of schizophrenia. Br J Psychiatry. 2000;177:212-217.
5. Goff DC, Cather C, Evins AE, et al. Medical morbidity and mortality in schizophrenia: guidelines for psychiatrists. J Clin Psychiatry. 2005;66(2):183-194.
6. Baggett TP, Keyes H, Sporn N, et al. Prevalence of SARS-CoV-2 infection in residents of a large homeless shelter in Boston. JAMA. 2020;323(21):2191-2192.
7. CDC COVID-19 Response Team. Severe outcomes among patients with coronavirus disease 2019 (COVID-19)—United States, February 12–March 16, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(12):343-346.
8. Docherty AB, Harrison EM, Green CA, et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985. doi: 10.1136/bmj.m1985
9. Etches V, Frank J, Di Ruggiero E, et al. Measuring population health: a review of indicators. Annu Rev Public Health. 2006;27:29-55.
10. Chepke C. Drive-up pharmacotherapy during the COVID-19 pandemic. Current Psychiatry. 2020;19(5):29-30.
11. Sajatovic M, Ross R, Legacy SN, et al. Initiating/maintaining long-acting injectable antipsychotics in schizophrenia/schizoaffective or bipolar disorder - expert consensus survey part 2. Neuropsychiatr Dis Treat. 2018;14:1475-1492.
12. Schoretsanitis G, Kane JM, Correll CU, et al; American Society of Clinical Psychopharmacology, Pharmakopsychiatrie TTDMTFOTAFNU. Blood levels to optimize antipsychotic treatment in clinical practice: a joint consensus statement of the American Society of Clinical Psychopharmacology and the Therapeutic Drug Monitoring Task Force of the Arbeitsgemeinschaft für Neuropsychopharmakologie und Pharmakopsychiatrie. J Clin Psychiatry. 2020;81(3):19cs13169. doi: 10.4088/JCP.19cs13169
COVID-19 and the risk of homicide-suicide among older adults
On March 25, 2020, in Cambridge, United Kingdom, a 71-year-old man stabbed his 71-year-old wife before suffocating himself to death. The couple was reportedly anxious about the coronavirus disease 2019 (COVID-19) pandemic lockdown measures and were on the verge of running out of food and medicine.1
One week later, in Chicago, Illinois, a 54-year-old man shot and killed his female partner, age 54, before killing himself. The couple was tested for COVID-19 2 days earlier and the man believed they had contracted the virus; however, the test results for both of them had come back negative.2
Intimate partner homicide-suicide is the most dramatic domestic abuse outcome.3 Homicide-suicide is defined as “homicide committed by a person who subsequently commits suicide within one week of the homicide. In most cases the subsequent suicide occurs within a 24-hour period.”4 Approximately one-quarter of all homicide-suicides are committed by persons age ≥55 years.5,6 We believe that during the COVID-19 pandemic, the risk of homicide-suicide among older adults may be increased due to several factors, including:
- physical distancing and quarantine measures. Protocols established to slow the spread of the virus may be associated with increased rates of depression and anxiety7 and an increased risk of suicide among older adults8
- increased intimate partner violence9
- increased firearm ownership rates in the United States.10
In this article, we review studies that identified risk factors for homicide-suicide among older adults, discuss the impact the COVID-19 pandemic has had on these risks, and describe steps clinicians can take to intervene.
A review of the literature
To better characterize the perpetrators of older adult homicide-suicide, we conducted a literature search of relevant terms. We identified 9 original research publications that examined homicide-suicide in older adults.
Bourget et al11 (2010) reviewed coroners’ charts of individuals killed by an older (age ≥65) spouse or family member from 1992 through 2007 in Quebec, Canada. They identified 19 cases of homicide-suicide, 17 (90%) of which were perpetrated by men. Perpetrators and victims were married (63%), in common-law relationships (16%), or separated/divorced (16%). A history of domestic violence was documented in 4 (21%) cases. The authors found that 13 of 15 perpetrators (87%) had “major depression” and 2 perpetrators had a psychotic disorder. Substance use at the time of the event was confirmed in 6 (32%) cases. Firearms and strangulation were the top methods used to carry out the homicide-suicide.11
Cheung et al12 (2016) conducted a review of coroners’ records of homicide-suicide cases among individuals age ≥65 in New Zealand from 2007 through 2012. In all 4 cases, the perpetrators were men, and their victims were predominantly female, live-in family members. Two cases involved men with a history of domestic violence who were undergoing significant changes in their home and social lives. Both men had a history suggestive of depression and used a firearm to carry out the homicide-suicide.12
Continue to: Cohen et al
Cohen et al13 (1998) conducted a review of coroners’ records from 1988 through 1994 in 2 regions in Florida. They found 48 intimate partner homicide-suicide cases among “old couples” (age ≥55). All were perpetrated by men. The authors identified sociocultural differences in risk factors between the 2 regions. In west-central Florida, perpetrators and victims were predominantly white and in a spousal relationship. Domestic violence was documented in <4% of cases. Approximately 55% of the couples were reported to be ill, and a substantial proportion were documented to be declining in health. One-quarter of the perpetrators and one-third of the victims had “pain and suffering.” More than one-third of perpetrators were reported to have “depression,” 15% were reported to have talked about suicide, and 4% had a history of a suicide attempt. Only 11% of perpetrators were described as abusing substances.
The authors noted several differences in cases in southeastern Florida. Approximately two-thirds of the couples were Hispanic, and 14% had a history of domestic violence. A minority of the couples were in a live-in relationship. Less than 15% of the perpetrators and victims were described as having a decline in health. Additionally, only 19% of perpetrators were reported to have “depression,” and none of the perpetrators had a documented history of attempted suicide or substance abuse. No information was provided regarding the methods used to carry out the homicide-suicide in the southeastern region.13 Financial stress was not a factor in either region.
Malphurs et al14 (2001) used the same database described in the Cohen et al13 study to compare 27 perpetrators of homicide-suicide to 36 age-matched suicide decedents in west central Florida. They found that homicide-suicide perpetrators were significantly less likely to have health problems and were 3 times more likely to be caregivers to their spouses. Approximately 52% of perpetrators had at least 1 documented psychiatric symptom (“depression” and/or substance abuse or other), but only 5% were seeking mental health services at the time of death.14
De Koning and Piette15 (2014) conducted a retrospective medicolegal chart review from 1935 to 2010 to identify homicide-suicide cases in West and East Flanders, Belgium. They found 19 cases of intimate partner homicide-suicide committed by offenders age ≥55 years. Ninety-five percent of the perpetrators were men who killed their female partners. In one-quarter of the cases, either the perpetrator or the victim had a health issue; 21% of the perpetrators were documented as having depression and 27% had alcohol intoxication at the time of death. A motive was documented in 14 out of 19 cases; “mercy killing” was determined as the motive in 6 (43%) cases and “amorous jealousy” in 5 cases (36%).15 Starting in the 1970s, firearms were the most prevalent method used to kill a partner.
Logan et al16 (2019) used data from the National Violent Death Reporting System between 2003 and 2015 to identify characteristics that differentiated male suicide decedents from male perpetrators of intimate partner homicide-suicide. They found that men age 50 to 64 years were 3 times more likely than men age 18 to 34 years to commit intimate partner homicide-suicide, and that men age ≥65 years were approximately 5 times more likely than men age 18 to 34 years to commit intimate partner homicide-suicide. The authors found that approximately 22% of all perpetrators had a documented history of physical domestic violence, and close to 17% had a prior interaction with the criminal justice system. Furthermore, one-third of perpetrators had relationship difficulties and were in the process of a breakup. Health issues were prevalent in 34% of the victims and 26% of the perpetrators. Perpetrator-caregiver burden was reported as a contributing factor for homicide-suicide in 16% of cases. In 27% of cases, multiple health-related contributing factors were mentioned.16
Continue to: Malphurs and Cohen
Malphurs and Cohen5 (2002) reviewed American newspapers from 1997 through 1999 and identified 673 homicide-suicide events, of which 152 (27%) were committed by individuals age ≥55 years. The victims and perpetrators (95% of which were men) were intimate partners in three-quarters of cases. In nearly one-third of cases, caregiving was a contributing factor for the homicide-suicide. A history of or a pending divorce was reported in nearly 14% of cases. Substance use history was rarely recorded. Firearms were used in 88% of the homicide-suicide cases.5
Malphurs and Cohen17 (2005) reviewed coroner records between 1998 and 1999 in Florida and compared 20 cases of intimate partner homicide-suicide involving perpetrators age ≥55 years with matched suicide decedents. They found that 60% of homicide-suicide perpetrators had documented health issues. The authors reported that a “recent change in health status” was more prevalent among perpetrators compared with decedents. Perpetrators were also more likely to be caregivers to their spouses. The authors found that 65% of perpetrators were reported to have a “depressed mood” and 15% of perpetrators had reportedly threatened suicide prior to the incident. However, none of the perpetrators tested positive for antidepressants as documented on post-mortem toxicology reports. Firearms were used in 100% of homicide-suicide cases.17
Salari3 (2007) reviewed multiple American media sources and published police reports between 1999 and 2005 to retrieve data about intimate partner homicide-suicide events in the United States. There were 225 events identified where the perpetrator and/or the victim were age ≥60 years. Ninety-six percent of the perpetrators were men and most homicide-suicide events were committed at the home. A history of domestic violence was reported in 14% of homicide-suicide cases. Thirteen percent of couples were separated or divorced. The perpetrator and/or victim had health issues in 124 (55%) events. Dementia was reported in 7.5% of cases, but overwhelmingly among the victims. Substance abuse was rarely mentioned as a contributing factor. In three-quarters of cases where a motive was described, the perpetrator was “suicidal”; however, a “suicide pact” was mentioned in only 4% of cases. Firearms were used in 87% of cases.3
Focus on common risk factors
The scarcity and heterogeneity of research regarding older adult homicide-suicide were major limitations to our review. Because most of the studies we identified had a small sample size and limited information regarding the mental health of victims and perpetrators, it would be an overreach to claim to have identified a typical profile of an older perpetrator of homicide-suicide. However, the literature has repeatedly identified several common characteristics of such perpetrators. They are significantly more likely to be men who use firearms to murder their intimate partners and then die by suicide in their home (Table3,5,11-17). Health issues afflicting 1 or both individuals in the couple appear to be a contributing factor, particularly when the perpetrator is in a caregiving role. Relational discord, with or without a history of domestic violence, increases the risk of homicide-suicide. Finally, older perpetrators are highly likely to be depressed and have suicidal ideations.
How COVID-19 affects these risks
Although it is too early to determine if there is a causal relationship between the COVID-19 pandemic and an increase in homicide-suicide, the pandemic is likely to promote risk factors for these events, especially among older adults. Confinement measures put into place during the pandemic context have already been shown to increase rates of domestic violence18 and depression and anxiety among older individuals.7 Furthermore,
Continue to: Late-life psychiatric disorders
Late-life psychiatric disorders
Early recognition and effective treatment of late-life psychiatric disorders is crucial. Unfortunately, depression in geriatric patients is often underdiagnosed and undertreated.20 Older adults have more frequent contact with their primary care physicians, and rarely consult mental health professionals.21,22 Several models of integrated depression care within primary care settings have shown the positive impact of this collaborative approach in treating late-life depression and preventing suicide in older individuals.23 Additionally, because alcohol abuse is also a risk factor for domestic violence and breaking the law in this population,24,25 older adults should be screened for alcohol use disorders, and referred to treatment when necessary.
Take steps to keep patients safe
In the context of the COVID-19 pandemic, there are several steps clinicians need to keep in mind when interacting with older patients:
- Screen for depressive symptoms, suicidality, and alcohol and substance use disorders. Individuals who have tested positive for COVID-19 or who have been in contact with a carrier are a particularly vulnerable population.
- Screen for domestic violence and access to weapons at home.4 Any older adult who has a psychiatric disorder and/or suicide ideation should receive immediate intervention through a social worker that includes providing gun-risk education to other family members or contacting law-enforcement officials.26
- Refer patients with a suspected psychiatric disorder to specialized mental health clinicians. Telemental health services can provide rapid access to subspecialists, allowing patients to be treated from their homes.27 These services need to be promoted among older adults during this critical period and reimbursed by public and private insurance systems to ensure accessibility and affordability.28
- Create psychiatric inpatient units specifically designed for suicidal and/or homicidal patients with COVID-19.
Additionally, informing the public about these major health issues is crucial. The media can raise awareness about domestic violence and depression among older adults; however, this should be done responsibly and with accuracy to prevent the spread of misinformation, confusion, fear, and panic.29
Bottom Line
The mental health burden of the coronavirus disease 2019 pandemic has significantly impacted individuals who are older and most vulnerable. Reducing the incidence of homicide-suicide among older adults requires timely screening and interventions by primary care providers, mental health specialists, social workers, media, and governmental agencies.
Related Resources
- Saeed SA, Hebishi K. The psychiatric consequences of COVID-19: 8 studies. Current Psychiatry. 2020;19(11):22-24,28-30,32-35.
- Schwab-Reese LM, Murfree L, Coppola EC, et al. Homicidesuicide across the lifespan: a mixed methods examination of factors contributing to older adult perpetration. Aging Ment Health. 2020;20:1-9.
1. Christodoulou H. LOCKDOWN ‘MURDER-SUICIDE’ OAP, 71, ‘stabbed wife to death then killed himself as he worried about coping with coronavirus lockdown.’ The Sun. Updated April 4, 2020. Accessed December 22, 2020. https://www.thesun.co.uk/news/11327095/coronavirus-lockdown-murder-suicide-cambridge/
2. Farberov S. Illinois man, 54, shoots dead his wife then kills himself in murder-suicide because he feared they had coronavirus - but tests later show the couple were NOT ill. Updated April 6, 2020. Accessed December 22, 2020. https://www.dailymail.co.uk/news/article-8191933/Man-kills-wife-feared-coronavirus.html
3. Salari S. Patterns of intimate partner homicide suicide in later life: strategies for prevention. Clin Interv Aging. 2007;2(3):441-452.
4. Kotzé C, Roos JL. Homicide–suicide: practical implications for risk reduction and support services at primary care level. South African Family Practice. 2019;61(4):165-169.
5. Malphurs JE, Cohen D. A newspaper surveillance study of homicide-suicide in the United States. Am J Forensic Med Pathol. 2002;23(2):142-148.
6. Eliason S. Murder-suicide: a review of the recent literature. J Am Acad Psychiatry Law. 2009;37(3):371-376.
7. Armitage R, Nellums LB. COVID-19 and the consequences of isolating the elderly. Lancet Public Health. 2020;5(5):e256. doi: 10.1016/S2468-2667(20)30061-X
8. Gunnell D, Appleby L, Arensman E, et al. Suicide risk and prevention during the COVID-19 pandemic. Lancet Psychiatry 2020;7(6):468-471.
9. Gosangi B, Park H, Thomas R, et al. Exacerbation of physical intimate partner violence during COVID-19 pandemic. Radiology. 2021;298(1):E38-E45.
10. Mannix R, Lee LK, Fleegler EW. Coronavirus disease 2019 (COVID-19) and firearms in the United States: will an epidemic of suicide follow? Ann Intern Med. 2020;173(3):228-229.
11. Bourget D, Gagne P, Whitehurst L. Domestic homicide and homicide-suicide: the older offender. J Am Acad Psychiatry Law. 2010;38(3):305-311.
12. Cheung G, Hatters Friedman S, Sundram F. Late-life homicide-suicide: a national case series in New Zealand. Psychogeriatrics. 2016;16(1):76-81.
13. Cohen D, Llorente M, Eisdorfer C. Homicide-suicide in older persons. Am J Psychiatry. 1998;155(3):390-396.
14. Malphurs JE, Eisdorfer C, Cohen D. A comparison of antecedents of homicide-suicide and suicide in older married men. Am J Geriatr Psychiatry. 2001;9(1):49-57.
15. De Koning E, Piette MHA. A retrospective study of murder–suicide at the Forensic Institute of Ghent University, Belgium: 1935–2010. Med Sci Law. 2014;54(2):88-98.
16. Logan JE, Ertl A, Bossarte R. Correlates of intimate partner homicide among male suicide decedents with known intimate partner problems. Suicide Life Threat Behav. 2019;49(6):1693-1706.
17. Malphurs JE, Cohen D. A statewide case-control study of spousal homicide-suicide in older persons. Am J Geriatr Psychiatry. 2005;13(3):211-217.
18. Sanford A. ‘Horrifying surge in domestic violence’ against women amid coronavirus-lockdowns, UN chief warns. Euronews. Published June 4, 2020. Accessed December 22, 2020. https://www.euronews.com/2020/04/06/horrifying-surge-in-domestic-violence-against-women-amid-coronavirus-lockdowns-un-chief-w
19. Appel JM. Intimate partner homicide in elderly populations. In: Friedman SH, ed. Family murder: pathologies of love and hate. American Psychiatric Association Publishing; 2019:131-142.
20. Hall CA, Reynolds-III CF. Late-life depression in the primary care setting: challenges, collaborative care, and prevention. Maturitas. 2014;79(2):147-152.
21. Unützer J. Diagnosis and treatment of older adults with depression in primary care. Biological Psychiatry. 2002;52(3):285-292.
22. Byers AL, Arean PA, Yaffe K. Low use of mental health services among older Americans with mood and anxiety disorders. Psychiatr Serv. 2012;63(1):66-72.
23. Bruce ML, Sirey JA. Integrated care for depression in older primary care patients. Can J Psychiatry. 2018;63(7):439-446.
24. Rao R, Roche A. Substance misuse in older people. BMJ. 2017;358:j3885. doi: 10.1136/bmj.j3885
25. Ghossoub E, Khoury R. Prevalence and correlates of criminal behavior among the non-institutionalized elderly: results from the National Survey on Drug Use and Health. J Geriatr Psychiatry Neurol. 2018;31(4):211-222.
26. Slater MAG. Older adults at risk for suicide. In: Berkman B. Handbook of social work in health and aging. Oxford University Press; 2006:149-161.
27. Hollander JE, Carr BG. Virtually perfect? Telemedicine for Covid-19. N Engl J Med. 2020;382(18):1679-1681.
28. Centers for Medicare & Medicaid Services. President Trump expands telehealth benefits for Medicare beneficiaries during COVID-19 outbreak. Published March 17, 2020. Accessed December 23, 2020. https://www.cms.gov/newsroom/press-releases/president-trump-expands-telehealth-benefits-medicare-beneficiaries-during-covid-19-outbreak
29. Mian A, Khan S. Coronavirus: the spread of misinformation. BMC Med. 2020;18(1):89.
On March 25, 2020, in Cambridge, United Kingdom, a 71-year-old man stabbed his 71-year-old wife before suffocating himself to death. The couple was reportedly anxious about the coronavirus disease 2019 (COVID-19) pandemic lockdown measures and were on the verge of running out of food and medicine.1
One week later, in Chicago, Illinois, a 54-year-old man shot and killed his female partner, age 54, before killing himself. The couple was tested for COVID-19 2 days earlier and the man believed they had contracted the virus; however, the test results for both of them had come back negative.2
Intimate partner homicide-suicide is the most dramatic domestic abuse outcome.3 Homicide-suicide is defined as “homicide committed by a person who subsequently commits suicide within one week of the homicide. In most cases the subsequent suicide occurs within a 24-hour period.”4 Approximately one-quarter of all homicide-suicides are committed by persons age ≥55 years.5,6 We believe that during the COVID-19 pandemic, the risk of homicide-suicide among older adults may be increased due to several factors, including:
- physical distancing and quarantine measures. Protocols established to slow the spread of the virus may be associated with increased rates of depression and anxiety7 and an increased risk of suicide among older adults8
- increased intimate partner violence9
- increased firearm ownership rates in the United States.10
In this article, we review studies that identified risk factors for homicide-suicide among older adults, discuss the impact the COVID-19 pandemic has had on these risks, and describe steps clinicians can take to intervene.
A review of the literature
To better characterize the perpetrators of older adult homicide-suicide, we conducted a literature search of relevant terms. We identified 9 original research publications that examined homicide-suicide in older adults.
Bourget et al11 (2010) reviewed coroners’ charts of individuals killed by an older (age ≥65) spouse or family member from 1992 through 2007 in Quebec, Canada. They identified 19 cases of homicide-suicide, 17 (90%) of which were perpetrated by men. Perpetrators and victims were married (63%), in common-law relationships (16%), or separated/divorced (16%). A history of domestic violence was documented in 4 (21%) cases. The authors found that 13 of 15 perpetrators (87%) had “major depression” and 2 perpetrators had a psychotic disorder. Substance use at the time of the event was confirmed in 6 (32%) cases. Firearms and strangulation were the top methods used to carry out the homicide-suicide.11
Cheung et al12 (2016) conducted a review of coroners’ records of homicide-suicide cases among individuals age ≥65 in New Zealand from 2007 through 2012. In all 4 cases, the perpetrators were men, and their victims were predominantly female, live-in family members. Two cases involved men with a history of domestic violence who were undergoing significant changes in their home and social lives. Both men had a history suggestive of depression and used a firearm to carry out the homicide-suicide.12
Continue to: Cohen et al
Cohen et al13 (1998) conducted a review of coroners’ records from 1988 through 1994 in 2 regions in Florida. They found 48 intimate partner homicide-suicide cases among “old couples” (age ≥55). All were perpetrated by men. The authors identified sociocultural differences in risk factors between the 2 regions. In west-central Florida, perpetrators and victims were predominantly white and in a spousal relationship. Domestic violence was documented in <4% of cases. Approximately 55% of the couples were reported to be ill, and a substantial proportion were documented to be declining in health. One-quarter of the perpetrators and one-third of the victims had “pain and suffering.” More than one-third of perpetrators were reported to have “depression,” 15% were reported to have talked about suicide, and 4% had a history of a suicide attempt. Only 11% of perpetrators were described as abusing substances.
The authors noted several differences in cases in southeastern Florida. Approximately two-thirds of the couples were Hispanic, and 14% had a history of domestic violence. A minority of the couples were in a live-in relationship. Less than 15% of the perpetrators and victims were described as having a decline in health. Additionally, only 19% of perpetrators were reported to have “depression,” and none of the perpetrators had a documented history of attempted suicide or substance abuse. No information was provided regarding the methods used to carry out the homicide-suicide in the southeastern region.13 Financial stress was not a factor in either region.
Malphurs et al14 (2001) used the same database described in the Cohen et al13 study to compare 27 perpetrators of homicide-suicide to 36 age-matched suicide decedents in west central Florida. They found that homicide-suicide perpetrators were significantly less likely to have health problems and were 3 times more likely to be caregivers to their spouses. Approximately 52% of perpetrators had at least 1 documented psychiatric symptom (“depression” and/or substance abuse or other), but only 5% were seeking mental health services at the time of death.14
De Koning and Piette15 (2014) conducted a retrospective medicolegal chart review from 1935 to 2010 to identify homicide-suicide cases in West and East Flanders, Belgium. They found 19 cases of intimate partner homicide-suicide committed by offenders age ≥55 years. Ninety-five percent of the perpetrators were men who killed their female partners. In one-quarter of the cases, either the perpetrator or the victim had a health issue; 21% of the perpetrators were documented as having depression and 27% had alcohol intoxication at the time of death. A motive was documented in 14 out of 19 cases; “mercy killing” was determined as the motive in 6 (43%) cases and “amorous jealousy” in 5 cases (36%).15 Starting in the 1970s, firearms were the most prevalent method used to kill a partner.
Logan et al16 (2019) used data from the National Violent Death Reporting System between 2003 and 2015 to identify characteristics that differentiated male suicide decedents from male perpetrators of intimate partner homicide-suicide. They found that men age 50 to 64 years were 3 times more likely than men age 18 to 34 years to commit intimate partner homicide-suicide, and that men age ≥65 years were approximately 5 times more likely than men age 18 to 34 years to commit intimate partner homicide-suicide. The authors found that approximately 22% of all perpetrators had a documented history of physical domestic violence, and close to 17% had a prior interaction with the criminal justice system. Furthermore, one-third of perpetrators had relationship difficulties and were in the process of a breakup. Health issues were prevalent in 34% of the victims and 26% of the perpetrators. Perpetrator-caregiver burden was reported as a contributing factor for homicide-suicide in 16% of cases. In 27% of cases, multiple health-related contributing factors were mentioned.16
Continue to: Malphurs and Cohen
Malphurs and Cohen5 (2002) reviewed American newspapers from 1997 through 1999 and identified 673 homicide-suicide events, of which 152 (27%) were committed by individuals age ≥55 years. The victims and perpetrators (95% of which were men) were intimate partners in three-quarters of cases. In nearly one-third of cases, caregiving was a contributing factor for the homicide-suicide. A history of or a pending divorce was reported in nearly 14% of cases. Substance use history was rarely recorded. Firearms were used in 88% of the homicide-suicide cases.5
Malphurs and Cohen17 (2005) reviewed coroner records between 1998 and 1999 in Florida and compared 20 cases of intimate partner homicide-suicide involving perpetrators age ≥55 years with matched suicide decedents. They found that 60% of homicide-suicide perpetrators had documented health issues. The authors reported that a “recent change in health status” was more prevalent among perpetrators compared with decedents. Perpetrators were also more likely to be caregivers to their spouses. The authors found that 65% of perpetrators were reported to have a “depressed mood” and 15% of perpetrators had reportedly threatened suicide prior to the incident. However, none of the perpetrators tested positive for antidepressants as documented on post-mortem toxicology reports. Firearms were used in 100% of homicide-suicide cases.17
Salari3 (2007) reviewed multiple American media sources and published police reports between 1999 and 2005 to retrieve data about intimate partner homicide-suicide events in the United States. There were 225 events identified where the perpetrator and/or the victim were age ≥60 years. Ninety-six percent of the perpetrators were men and most homicide-suicide events were committed at the home. A history of domestic violence was reported in 14% of homicide-suicide cases. Thirteen percent of couples were separated or divorced. The perpetrator and/or victim had health issues in 124 (55%) events. Dementia was reported in 7.5% of cases, but overwhelmingly among the victims. Substance abuse was rarely mentioned as a contributing factor. In three-quarters of cases where a motive was described, the perpetrator was “suicidal”; however, a “suicide pact” was mentioned in only 4% of cases. Firearms were used in 87% of cases.3
Focus on common risk factors
The scarcity and heterogeneity of research regarding older adult homicide-suicide were major limitations to our review. Because most of the studies we identified had a small sample size and limited information regarding the mental health of victims and perpetrators, it would be an overreach to claim to have identified a typical profile of an older perpetrator of homicide-suicide. However, the literature has repeatedly identified several common characteristics of such perpetrators. They are significantly more likely to be men who use firearms to murder their intimate partners and then die by suicide in their home (Table3,5,11-17). Health issues afflicting 1 or both individuals in the couple appear to be a contributing factor, particularly when the perpetrator is in a caregiving role. Relational discord, with or without a history of domestic violence, increases the risk of homicide-suicide. Finally, older perpetrators are highly likely to be depressed and have suicidal ideations.
How COVID-19 affects these risks
Although it is too early to determine if there is a causal relationship between the COVID-19 pandemic and an increase in homicide-suicide, the pandemic is likely to promote risk factors for these events, especially among older adults. Confinement measures put into place during the pandemic context have already been shown to increase rates of domestic violence18 and depression and anxiety among older individuals.7 Furthermore,
Continue to: Late-life psychiatric disorders
Late-life psychiatric disorders
Early recognition and effective treatment of late-life psychiatric disorders is crucial. Unfortunately, depression in geriatric patients is often underdiagnosed and undertreated.20 Older adults have more frequent contact with their primary care physicians, and rarely consult mental health professionals.21,22 Several models of integrated depression care within primary care settings have shown the positive impact of this collaborative approach in treating late-life depression and preventing suicide in older individuals.23 Additionally, because alcohol abuse is also a risk factor for domestic violence and breaking the law in this population,24,25 older adults should be screened for alcohol use disorders, and referred to treatment when necessary.
Take steps to keep patients safe
In the context of the COVID-19 pandemic, there are several steps clinicians need to keep in mind when interacting with older patients:
- Screen for depressive symptoms, suicidality, and alcohol and substance use disorders. Individuals who have tested positive for COVID-19 or who have been in contact with a carrier are a particularly vulnerable population.
- Screen for domestic violence and access to weapons at home.4 Any older adult who has a psychiatric disorder and/or suicide ideation should receive immediate intervention through a social worker that includes providing gun-risk education to other family members or contacting law-enforcement officials.26
- Refer patients with a suspected psychiatric disorder to specialized mental health clinicians. Telemental health services can provide rapid access to subspecialists, allowing patients to be treated from their homes.27 These services need to be promoted among older adults during this critical period and reimbursed by public and private insurance systems to ensure accessibility and affordability.28
- Create psychiatric inpatient units specifically designed for suicidal and/or homicidal patients with COVID-19.
Additionally, informing the public about these major health issues is crucial. The media can raise awareness about domestic violence and depression among older adults; however, this should be done responsibly and with accuracy to prevent the spread of misinformation, confusion, fear, and panic.29
Bottom Line
The mental health burden of the coronavirus disease 2019 pandemic has significantly impacted individuals who are older and most vulnerable. Reducing the incidence of homicide-suicide among older adults requires timely screening and interventions by primary care providers, mental health specialists, social workers, media, and governmental agencies.
Related Resources
- Saeed SA, Hebishi K. The psychiatric consequences of COVID-19: 8 studies. Current Psychiatry. 2020;19(11):22-24,28-30,32-35.
- Schwab-Reese LM, Murfree L, Coppola EC, et al. Homicidesuicide across the lifespan: a mixed methods examination of factors contributing to older adult perpetration. Aging Ment Health. 2020;20:1-9.
On March 25, 2020, in Cambridge, United Kingdom, a 71-year-old man stabbed his 71-year-old wife before suffocating himself to death. The couple was reportedly anxious about the coronavirus disease 2019 (COVID-19) pandemic lockdown measures and were on the verge of running out of food and medicine.1
One week later, in Chicago, Illinois, a 54-year-old man shot and killed his female partner, age 54, before killing himself. The couple was tested for COVID-19 2 days earlier and the man believed they had contracted the virus; however, the test results for both of them had come back negative.2
Intimate partner homicide-suicide is the most dramatic domestic abuse outcome.3 Homicide-suicide is defined as “homicide committed by a person who subsequently commits suicide within one week of the homicide. In most cases the subsequent suicide occurs within a 24-hour period.”4 Approximately one-quarter of all homicide-suicides are committed by persons age ≥55 years.5,6 We believe that during the COVID-19 pandemic, the risk of homicide-suicide among older adults may be increased due to several factors, including:
- physical distancing and quarantine measures. Protocols established to slow the spread of the virus may be associated with increased rates of depression and anxiety7 and an increased risk of suicide among older adults8
- increased intimate partner violence9
- increased firearm ownership rates in the United States.10
In this article, we review studies that identified risk factors for homicide-suicide among older adults, discuss the impact the COVID-19 pandemic has had on these risks, and describe steps clinicians can take to intervene.
A review of the literature
To better characterize the perpetrators of older adult homicide-suicide, we conducted a literature search of relevant terms. We identified 9 original research publications that examined homicide-suicide in older adults.
Bourget et al11 (2010) reviewed coroners’ charts of individuals killed by an older (age ≥65) spouse or family member from 1992 through 2007 in Quebec, Canada. They identified 19 cases of homicide-suicide, 17 (90%) of which were perpetrated by men. Perpetrators and victims were married (63%), in common-law relationships (16%), or separated/divorced (16%). A history of domestic violence was documented in 4 (21%) cases. The authors found that 13 of 15 perpetrators (87%) had “major depression” and 2 perpetrators had a psychotic disorder. Substance use at the time of the event was confirmed in 6 (32%) cases. Firearms and strangulation were the top methods used to carry out the homicide-suicide.11
Cheung et al12 (2016) conducted a review of coroners’ records of homicide-suicide cases among individuals age ≥65 in New Zealand from 2007 through 2012. In all 4 cases, the perpetrators were men, and their victims were predominantly female, live-in family members. Two cases involved men with a history of domestic violence who were undergoing significant changes in their home and social lives. Both men had a history suggestive of depression and used a firearm to carry out the homicide-suicide.12
Continue to: Cohen et al
Cohen et al13 (1998) conducted a review of coroners’ records from 1988 through 1994 in 2 regions in Florida. They found 48 intimate partner homicide-suicide cases among “old couples” (age ≥55). All were perpetrated by men. The authors identified sociocultural differences in risk factors between the 2 regions. In west-central Florida, perpetrators and victims were predominantly white and in a spousal relationship. Domestic violence was documented in <4% of cases. Approximately 55% of the couples were reported to be ill, and a substantial proportion were documented to be declining in health. One-quarter of the perpetrators and one-third of the victims had “pain and suffering.” More than one-third of perpetrators were reported to have “depression,” 15% were reported to have talked about suicide, and 4% had a history of a suicide attempt. Only 11% of perpetrators were described as abusing substances.
The authors noted several differences in cases in southeastern Florida. Approximately two-thirds of the couples were Hispanic, and 14% had a history of domestic violence. A minority of the couples were in a live-in relationship. Less than 15% of the perpetrators and victims were described as having a decline in health. Additionally, only 19% of perpetrators were reported to have “depression,” and none of the perpetrators had a documented history of attempted suicide or substance abuse. No information was provided regarding the methods used to carry out the homicide-suicide in the southeastern region.13 Financial stress was not a factor in either region.
Malphurs et al14 (2001) used the same database described in the Cohen et al13 study to compare 27 perpetrators of homicide-suicide to 36 age-matched suicide decedents in west central Florida. They found that homicide-suicide perpetrators were significantly less likely to have health problems and were 3 times more likely to be caregivers to their spouses. Approximately 52% of perpetrators had at least 1 documented psychiatric symptom (“depression” and/or substance abuse or other), but only 5% were seeking mental health services at the time of death.14
De Koning and Piette15 (2014) conducted a retrospective medicolegal chart review from 1935 to 2010 to identify homicide-suicide cases in West and East Flanders, Belgium. They found 19 cases of intimate partner homicide-suicide committed by offenders age ≥55 years. Ninety-five percent of the perpetrators were men who killed their female partners. In one-quarter of the cases, either the perpetrator or the victim had a health issue; 21% of the perpetrators were documented as having depression and 27% had alcohol intoxication at the time of death. A motive was documented in 14 out of 19 cases; “mercy killing” was determined as the motive in 6 (43%) cases and “amorous jealousy” in 5 cases (36%).15 Starting in the 1970s, firearms were the most prevalent method used to kill a partner.
Logan et al16 (2019) used data from the National Violent Death Reporting System between 2003 and 2015 to identify characteristics that differentiated male suicide decedents from male perpetrators of intimate partner homicide-suicide. They found that men age 50 to 64 years were 3 times more likely than men age 18 to 34 years to commit intimate partner homicide-suicide, and that men age ≥65 years were approximately 5 times more likely than men age 18 to 34 years to commit intimate partner homicide-suicide. The authors found that approximately 22% of all perpetrators had a documented history of physical domestic violence, and close to 17% had a prior interaction with the criminal justice system. Furthermore, one-third of perpetrators had relationship difficulties and were in the process of a breakup. Health issues were prevalent in 34% of the victims and 26% of the perpetrators. Perpetrator-caregiver burden was reported as a contributing factor for homicide-suicide in 16% of cases. In 27% of cases, multiple health-related contributing factors were mentioned.16
Continue to: Malphurs and Cohen
Malphurs and Cohen5 (2002) reviewed American newspapers from 1997 through 1999 and identified 673 homicide-suicide events, of which 152 (27%) were committed by individuals age ≥55 years. The victims and perpetrators (95% of which were men) were intimate partners in three-quarters of cases. In nearly one-third of cases, caregiving was a contributing factor for the homicide-suicide. A history of or a pending divorce was reported in nearly 14% of cases. Substance use history was rarely recorded. Firearms were used in 88% of the homicide-suicide cases.5
Malphurs and Cohen17 (2005) reviewed coroner records between 1998 and 1999 in Florida and compared 20 cases of intimate partner homicide-suicide involving perpetrators age ≥55 years with matched suicide decedents. They found that 60% of homicide-suicide perpetrators had documented health issues. The authors reported that a “recent change in health status” was more prevalent among perpetrators compared with decedents. Perpetrators were also more likely to be caregivers to their spouses. The authors found that 65% of perpetrators were reported to have a “depressed mood” and 15% of perpetrators had reportedly threatened suicide prior to the incident. However, none of the perpetrators tested positive for antidepressants as documented on post-mortem toxicology reports. Firearms were used in 100% of homicide-suicide cases.17
Salari3 (2007) reviewed multiple American media sources and published police reports between 1999 and 2005 to retrieve data about intimate partner homicide-suicide events in the United States. There were 225 events identified where the perpetrator and/or the victim were age ≥60 years. Ninety-six percent of the perpetrators were men and most homicide-suicide events were committed at the home. A history of domestic violence was reported in 14% of homicide-suicide cases. Thirteen percent of couples were separated or divorced. The perpetrator and/or victim had health issues in 124 (55%) events. Dementia was reported in 7.5% of cases, but overwhelmingly among the victims. Substance abuse was rarely mentioned as a contributing factor. In three-quarters of cases where a motive was described, the perpetrator was “suicidal”; however, a “suicide pact” was mentioned in only 4% of cases. Firearms were used in 87% of cases.3
Focus on common risk factors
The scarcity and heterogeneity of research regarding older adult homicide-suicide were major limitations to our review. Because most of the studies we identified had a small sample size and limited information regarding the mental health of victims and perpetrators, it would be an overreach to claim to have identified a typical profile of an older perpetrator of homicide-suicide. However, the literature has repeatedly identified several common characteristics of such perpetrators. They are significantly more likely to be men who use firearms to murder their intimate partners and then die by suicide in their home (Table3,5,11-17). Health issues afflicting 1 or both individuals in the couple appear to be a contributing factor, particularly when the perpetrator is in a caregiving role. Relational discord, with or without a history of domestic violence, increases the risk of homicide-suicide. Finally, older perpetrators are highly likely to be depressed and have suicidal ideations.
How COVID-19 affects these risks
Although it is too early to determine if there is a causal relationship between the COVID-19 pandemic and an increase in homicide-suicide, the pandemic is likely to promote risk factors for these events, especially among older adults. Confinement measures put into place during the pandemic context have already been shown to increase rates of domestic violence18 and depression and anxiety among older individuals.7 Furthermore,
Continue to: Late-life psychiatric disorders
Late-life psychiatric disorders
Early recognition and effective treatment of late-life psychiatric disorders is crucial. Unfortunately, depression in geriatric patients is often underdiagnosed and undertreated.20 Older adults have more frequent contact with their primary care physicians, and rarely consult mental health professionals.21,22 Several models of integrated depression care within primary care settings have shown the positive impact of this collaborative approach in treating late-life depression and preventing suicide in older individuals.23 Additionally, because alcohol abuse is also a risk factor for domestic violence and breaking the law in this population,24,25 older adults should be screened for alcohol use disorders, and referred to treatment when necessary.
Take steps to keep patients safe
In the context of the COVID-19 pandemic, there are several steps clinicians need to keep in mind when interacting with older patients:
- Screen for depressive symptoms, suicidality, and alcohol and substance use disorders. Individuals who have tested positive for COVID-19 or who have been in contact with a carrier are a particularly vulnerable population.
- Screen for domestic violence and access to weapons at home.4 Any older adult who has a psychiatric disorder and/or suicide ideation should receive immediate intervention through a social worker that includes providing gun-risk education to other family members or contacting law-enforcement officials.26
- Refer patients with a suspected psychiatric disorder to specialized mental health clinicians. Telemental health services can provide rapid access to subspecialists, allowing patients to be treated from their homes.27 These services need to be promoted among older adults during this critical period and reimbursed by public and private insurance systems to ensure accessibility and affordability.28
- Create psychiatric inpatient units specifically designed for suicidal and/or homicidal patients with COVID-19.
Additionally, informing the public about these major health issues is crucial. The media can raise awareness about domestic violence and depression among older adults; however, this should be done responsibly and with accuracy to prevent the spread of misinformation, confusion, fear, and panic.29
Bottom Line
The mental health burden of the coronavirus disease 2019 pandemic has significantly impacted individuals who are older and most vulnerable. Reducing the incidence of homicide-suicide among older adults requires timely screening and interventions by primary care providers, mental health specialists, social workers, media, and governmental agencies.
Related Resources
- Saeed SA, Hebishi K. The psychiatric consequences of COVID-19: 8 studies. Current Psychiatry. 2020;19(11):22-24,28-30,32-35.
- Schwab-Reese LM, Murfree L, Coppola EC, et al. Homicidesuicide across the lifespan: a mixed methods examination of factors contributing to older adult perpetration. Aging Ment Health. 2020;20:1-9.
1. Christodoulou H. LOCKDOWN ‘MURDER-SUICIDE’ OAP, 71, ‘stabbed wife to death then killed himself as he worried about coping with coronavirus lockdown.’ The Sun. Updated April 4, 2020. Accessed December 22, 2020. https://www.thesun.co.uk/news/11327095/coronavirus-lockdown-murder-suicide-cambridge/
2. Farberov S. Illinois man, 54, shoots dead his wife then kills himself in murder-suicide because he feared they had coronavirus - but tests later show the couple were NOT ill. Updated April 6, 2020. Accessed December 22, 2020. https://www.dailymail.co.uk/news/article-8191933/Man-kills-wife-feared-coronavirus.html
3. Salari S. Patterns of intimate partner homicide suicide in later life: strategies for prevention. Clin Interv Aging. 2007;2(3):441-452.
4. Kotzé C, Roos JL. Homicide–suicide: practical implications for risk reduction and support services at primary care level. South African Family Practice. 2019;61(4):165-169.
5. Malphurs JE, Cohen D. A newspaper surveillance study of homicide-suicide in the United States. Am J Forensic Med Pathol. 2002;23(2):142-148.
6. Eliason S. Murder-suicide: a review of the recent literature. J Am Acad Psychiatry Law. 2009;37(3):371-376.
7. Armitage R, Nellums LB. COVID-19 and the consequences of isolating the elderly. Lancet Public Health. 2020;5(5):e256. doi: 10.1016/S2468-2667(20)30061-X
8. Gunnell D, Appleby L, Arensman E, et al. Suicide risk and prevention during the COVID-19 pandemic. Lancet Psychiatry 2020;7(6):468-471.
9. Gosangi B, Park H, Thomas R, et al. Exacerbation of physical intimate partner violence during COVID-19 pandemic. Radiology. 2021;298(1):E38-E45.
10. Mannix R, Lee LK, Fleegler EW. Coronavirus disease 2019 (COVID-19) and firearms in the United States: will an epidemic of suicide follow? Ann Intern Med. 2020;173(3):228-229.
11. Bourget D, Gagne P, Whitehurst L. Domestic homicide and homicide-suicide: the older offender. J Am Acad Psychiatry Law. 2010;38(3):305-311.
12. Cheung G, Hatters Friedman S, Sundram F. Late-life homicide-suicide: a national case series in New Zealand. Psychogeriatrics. 2016;16(1):76-81.
13. Cohen D, Llorente M, Eisdorfer C. Homicide-suicide in older persons. Am J Psychiatry. 1998;155(3):390-396.
14. Malphurs JE, Eisdorfer C, Cohen D. A comparison of antecedents of homicide-suicide and suicide in older married men. Am J Geriatr Psychiatry. 2001;9(1):49-57.
15. De Koning E, Piette MHA. A retrospective study of murder–suicide at the Forensic Institute of Ghent University, Belgium: 1935–2010. Med Sci Law. 2014;54(2):88-98.
16. Logan JE, Ertl A, Bossarte R. Correlates of intimate partner homicide among male suicide decedents with known intimate partner problems. Suicide Life Threat Behav. 2019;49(6):1693-1706.
17. Malphurs JE, Cohen D. A statewide case-control study of spousal homicide-suicide in older persons. Am J Geriatr Psychiatry. 2005;13(3):211-217.
18. Sanford A. ‘Horrifying surge in domestic violence’ against women amid coronavirus-lockdowns, UN chief warns. Euronews. Published June 4, 2020. Accessed December 22, 2020. https://www.euronews.com/2020/04/06/horrifying-surge-in-domestic-violence-against-women-amid-coronavirus-lockdowns-un-chief-w
19. Appel JM. Intimate partner homicide in elderly populations. In: Friedman SH, ed. Family murder: pathologies of love and hate. American Psychiatric Association Publishing; 2019:131-142.
20. Hall CA, Reynolds-III CF. Late-life depression in the primary care setting: challenges, collaborative care, and prevention. Maturitas. 2014;79(2):147-152.
21. Unützer J. Diagnosis and treatment of older adults with depression in primary care. Biological Psychiatry. 2002;52(3):285-292.
22. Byers AL, Arean PA, Yaffe K. Low use of mental health services among older Americans with mood and anxiety disorders. Psychiatr Serv. 2012;63(1):66-72.
23. Bruce ML, Sirey JA. Integrated care for depression in older primary care patients. Can J Psychiatry. 2018;63(7):439-446.
24. Rao R, Roche A. Substance misuse in older people. BMJ. 2017;358:j3885. doi: 10.1136/bmj.j3885
25. Ghossoub E, Khoury R. Prevalence and correlates of criminal behavior among the non-institutionalized elderly: results from the National Survey on Drug Use and Health. J Geriatr Psychiatry Neurol. 2018;31(4):211-222.
26. Slater MAG. Older adults at risk for suicide. In: Berkman B. Handbook of social work in health and aging. Oxford University Press; 2006:149-161.
27. Hollander JE, Carr BG. Virtually perfect? Telemedicine for Covid-19. N Engl J Med. 2020;382(18):1679-1681.
28. Centers for Medicare & Medicaid Services. President Trump expands telehealth benefits for Medicare beneficiaries during COVID-19 outbreak. Published March 17, 2020. Accessed December 23, 2020. https://www.cms.gov/newsroom/press-releases/president-trump-expands-telehealth-benefits-medicare-beneficiaries-during-covid-19-outbreak
29. Mian A, Khan S. Coronavirus: the spread of misinformation. BMC Med. 2020;18(1):89.
1. Christodoulou H. LOCKDOWN ‘MURDER-SUICIDE’ OAP, 71, ‘stabbed wife to death then killed himself as he worried about coping with coronavirus lockdown.’ The Sun. Updated April 4, 2020. Accessed December 22, 2020. https://www.thesun.co.uk/news/11327095/coronavirus-lockdown-murder-suicide-cambridge/
2. Farberov S. Illinois man, 54, shoots dead his wife then kills himself in murder-suicide because he feared they had coronavirus - but tests later show the couple were NOT ill. Updated April 6, 2020. Accessed December 22, 2020. https://www.dailymail.co.uk/news/article-8191933/Man-kills-wife-feared-coronavirus.html
3. Salari S. Patterns of intimate partner homicide suicide in later life: strategies for prevention. Clin Interv Aging. 2007;2(3):441-452.
4. Kotzé C, Roos JL. Homicide–suicide: practical implications for risk reduction and support services at primary care level. South African Family Practice. 2019;61(4):165-169.
5. Malphurs JE, Cohen D. A newspaper surveillance study of homicide-suicide in the United States. Am J Forensic Med Pathol. 2002;23(2):142-148.
6. Eliason S. Murder-suicide: a review of the recent literature. J Am Acad Psychiatry Law. 2009;37(3):371-376.
7. Armitage R, Nellums LB. COVID-19 and the consequences of isolating the elderly. Lancet Public Health. 2020;5(5):e256. doi: 10.1016/S2468-2667(20)30061-X
8. Gunnell D, Appleby L, Arensman E, et al. Suicide risk and prevention during the COVID-19 pandemic. Lancet Psychiatry 2020;7(6):468-471.
9. Gosangi B, Park H, Thomas R, et al. Exacerbation of physical intimate partner violence during COVID-19 pandemic. Radiology. 2021;298(1):E38-E45.
10. Mannix R, Lee LK, Fleegler EW. Coronavirus disease 2019 (COVID-19) and firearms in the United States: will an epidemic of suicide follow? Ann Intern Med. 2020;173(3):228-229.
11. Bourget D, Gagne P, Whitehurst L. Domestic homicide and homicide-suicide: the older offender. J Am Acad Psychiatry Law. 2010;38(3):305-311.
12. Cheung G, Hatters Friedman S, Sundram F. Late-life homicide-suicide: a national case series in New Zealand. Psychogeriatrics. 2016;16(1):76-81.
13. Cohen D, Llorente M, Eisdorfer C. Homicide-suicide in older persons. Am J Psychiatry. 1998;155(3):390-396.
14. Malphurs JE, Eisdorfer C, Cohen D. A comparison of antecedents of homicide-suicide and suicide in older married men. Am J Geriatr Psychiatry. 2001;9(1):49-57.
15. De Koning E, Piette MHA. A retrospective study of murder–suicide at the Forensic Institute of Ghent University, Belgium: 1935–2010. Med Sci Law. 2014;54(2):88-98.
16. Logan JE, Ertl A, Bossarte R. Correlates of intimate partner homicide among male suicide decedents with known intimate partner problems. Suicide Life Threat Behav. 2019;49(6):1693-1706.
17. Malphurs JE, Cohen D. A statewide case-control study of spousal homicide-suicide in older persons. Am J Geriatr Psychiatry. 2005;13(3):211-217.
18. Sanford A. ‘Horrifying surge in domestic violence’ against women amid coronavirus-lockdowns, UN chief warns. Euronews. Published June 4, 2020. Accessed December 22, 2020. https://www.euronews.com/2020/04/06/horrifying-surge-in-domestic-violence-against-women-amid-coronavirus-lockdowns-un-chief-w
19. Appel JM. Intimate partner homicide in elderly populations. In: Friedman SH, ed. Family murder: pathologies of love and hate. American Psychiatric Association Publishing; 2019:131-142.
20. Hall CA, Reynolds-III CF. Late-life depression in the primary care setting: challenges, collaborative care, and prevention. Maturitas. 2014;79(2):147-152.
21. Unützer J. Diagnosis and treatment of older adults with depression in primary care. Biological Psychiatry. 2002;52(3):285-292.
22. Byers AL, Arean PA, Yaffe K. Low use of mental health services among older Americans with mood and anxiety disorders. Psychiatr Serv. 2012;63(1):66-72.
23. Bruce ML, Sirey JA. Integrated care for depression in older primary care patients. Can J Psychiatry. 2018;63(7):439-446.
24. Rao R, Roche A. Substance misuse in older people. BMJ. 2017;358:j3885. doi: 10.1136/bmj.j3885
25. Ghossoub E, Khoury R. Prevalence and correlates of criminal behavior among the non-institutionalized elderly: results from the National Survey on Drug Use and Health. J Geriatr Psychiatry Neurol. 2018;31(4):211-222.
26. Slater MAG. Older adults at risk for suicide. In: Berkman B. Handbook of social work in health and aging. Oxford University Press; 2006:149-161.
27. Hollander JE, Carr BG. Virtually perfect? Telemedicine for Covid-19. N Engl J Med. 2020;382(18):1679-1681.
28. Centers for Medicare & Medicaid Services. President Trump expands telehealth benefits for Medicare beneficiaries during COVID-19 outbreak. Published March 17, 2020. Accessed December 23, 2020. https://www.cms.gov/newsroom/press-releases/president-trump-expands-telehealth-benefits-medicare-beneficiaries-during-covid-19-outbreak
29. Mian A, Khan S. Coronavirus: the spread of misinformation. BMC Med. 2020;18(1):89.
Anticonvulsants for alcohol withdrawal: A review of the evidence
Abrupt cessation or reduction of alcohol consumption may result in alcohol withdrawal syndrome (AWS), which is a medical emergency that can lead to serious complications when unrecognized or treatment is delayed. Symptoms of AWS include tremors, anxiety attacks, cognitive impairment, hallucinations, seizures, delirium tremens (DT), and in severe, untreated cases, death.1 Low to moderate alcohol consumption produces euphoria and excitation via activation of glutamatergic neurotransmission, while higher concentrations produce severe intoxication via GABAergic mechanisms. Acute withdrawal unmasks the hyper-excitatory state of the brain, causing anxiety, agitation, and autonomic activation characteristic of AWS, which typically begins 1 to 3 days after the last drink.2 In the 2012-2013 National Epidemiologic Survey on Alcohol and Related Conditions conducted by the National Institute on Alcohol Abuse and Alcoholism (NIAAA), the 12-month and lifetime prevalences of AWS were 13.9% and 29.1%, respectively.3 Within the general inpatient population, AWS can be present in nearly 30% of patients; if left untreated, AWS has a 15% mortality rate, although when AWS is recognized early and treated, the mortality rate falls dramatically to 2%.4
AWS has most commonly been treated with benzodiazepines.5 However, benzodiazepines have the potential for significant adverse effects when used in older adults and in individuals with complicated medical issues, such as obstructive lung disease and sleep apnea.6 Anticonvulsants have been increasingly used to treat alcohol withdrawal, and their use is supported by several retrospective and prospective studies. In this article, we review the data from randomized control trials (RCTs) on the use of anticonvulsants for the treatment of AWS to see if we can make any recommendations for the use of anticonvulsants for treating AWS.
Our literature search
We searched 5 databases (PubMed, Cochrane, Medline, PsycInfo, and Embase) using the following terms: “alcohol withdrawal syndrome treatment”, “anticonvulsants”, “anti-epileptic”, “gabapentin”, “carbamazepine”, “sodium valproate”, “oxcarbazepine”, “phenytoin”, “levetiracetam”, and “lamotrigine.” We included only double-blind RCTs published between January 1, 1976 and September 30, 2016 in English-language journals or that had an official English translation. There were no restrictions on patient age or location of treatment (inpatient vs outpatient). All RCTs that compared anticonvulsants or a combination of an anticonvulsant and an active pharmacotherapeutic agent with either placebo or gold standard treatment for AWS were included. Database reviews, systematic reviews, and meta-analyses were excluded.
We identified 662 articles that met these criteria. However, most were duplicates, review articles, systematic reviews, meta-analyses, case reports, or open-label or non-randomized trials. Only 16 articles met our inclusion criteria. In the following sections, we discuss these 16 studies by medication type and in chronological order.
Gabapentin
The characteristics of the gabapentin studies included in this review are summarized in Table 1.7-13
Bonnet et al7 (2003) examined 61 adults who met the clinical criteria for alcohol dependence and displayed moderate or severe AWS according to their Mainz Alcohol Withdrawal Score (MAWS ≥4). They were randomized to receive placebo or gabapentin, 400 mg 4 times a day, along with clomethiazole. The attrition rate was not significantly different between the 2 groups (P = .66). The difference in the number of clomethiazole capsules taken during the first 24 hours between the groups was small and not significant (P = .96). Analysis of MAWS over time revealed no significant main effect for group (P = .26) and a significant effect for the time variable (P < .001). The interaction between group and time was not significant (P =.4). This means that there was a significant decrease in MAWS from baseline over 48 hours, and this decrease in MAWS was considered equal for both study groups. Adverse clinical events were observed in both groups, and there was no significant difference (P = .74) between the groups. Nausea and ataxia, which are specific to gabapentin, were observed more frequently in this group.
Conclusion: The authors concluded that gabapentin, 400 mg 4 times a day, is no better than placebo in reducing the amount of clomethiazole required to treat acute AWS.7
Continue to: Bonnet et al
Bonnet et al8 (2007) also conducted a study examining 59 patients with alcohol dependence who displayed moderate or severe AWS. Participants received placebo or gabapentin, 400 mg, and a rescue medication, clomethiazole, if needed. Subsequently, a capsule of study medication was administered every 6 hours for 2 days and then tapered. During the study, mood was measured by Profile of Mood States (POMS), and subjective complaints of withdrawal were measured using the Essen Self-Assessment of Alcohol Withdrawal Scale (ESA). Of the 59 patients, only 46 were analyzed; 5 patients dropped out, and 8 patients were missing data. Compared with the placebo group, the gabapentin group displayed less dejection, fatigue, and anger, and more vigor. Analysis of variance (ANOVA) measures revealed significant overall changes over time on all 4 scales (all P < .001). A significant (F = 3.62, df 2;43, P = .035) group × time interaction resulted exclusively for vigor. Analysis was repeated using rank-transformed data, resulting in a significant (P = .046) interaction effect. The significant increase in vigor was not apparent after tapering off gabapentin, which suggests gabapentin has a reversible effect on vigor. There was a significant (P < .001) overall decline of subjective withdrawal symptoms complaints, but no group × time interaction (P = .62). Analysis of 11 patients with comorbid mild depression revealed no significant time × group interaction for dejection, fatigue, anger, or subjective withdrawal (all P > .05). However, for vigor, the group × time interaction was significant (P = .022). Throughout the treatment, vigor scores of those mild depressive patients who received gabapentin increased to a level comparable to that of patients without a mood disorder.
Conclusion: The authors authors concluded that gabapentin was markedly more efficacious in improving vigor in the small subgroup of patients with mild depression.8
Myrick et al9 (2007) evaluated the safety and tolerability of gabapentin in patients who abused alcohol, as well as the ability of gabapentin to reduce alcohol craving and consumption. This study included 35 participants randomly assigned to receive gabapentin (n = 17) or placebo (n = 18) for 7 days. All medications were administered in standard gel caps with riboflavin, 25 mg, to assess for compliance via a laboratory-based urinary fluorescence assay. Urine samples were assessed for riboflavin at baseline and Day 6, and a reading of 1,500 ng/mL of riboflavin on Day 6 was interpreted as being compliant. Participants were required to abstain completely from drinking alcohol on Day 6 and the morning of Day 7. At the first session, the following measures were completed: demographic form, alcohol and drug section of the Structured Clinical Interview (SCID), Obsessive-Compulsive Drinking Scale, Self-Administered Alcohol Screening Test (SAAST), and Alcohol Dependence Scale (ADS); there also was collection of a urine sample for detection of abused drugs and a blood sample for liver function and general health screening.
At the second session, patients completed the psychiatric sections of the SCID and the Alcohol Craving Questionnaire, and received a physical exam. To assess the negative clinical effects of gabapentin and alcohol on the CNS, the Epworth Sleepiness Scale (ESS) and POMS were administered at baseline and on Day 6. Also, several other scales were used to identify any impact of gabapentin on acute alcohol effects and craving: the Clinical Institute Withdrawal Assessment of Alcohol, Revised (CIWA-Ar), Biphasic Alcohol Effects Scale (BAES), Subjective High Assessment Scale (SHAS), and Alcohol Urge Questionnaire (AUQ).
Conclusion: Gabapentin was well tolerated, but compared with placebo, gabapentin had no effect on alcohol stimulation (P = .75) or sedation (P = .99) as measured by the BAES. The difference in SHAS scores was also not significant (P = .19). There was also no significant reduction in craving for alcohol as measured on the AUQ scale
Continue to: Malcolm et al
Malcolm et al10 conducted an outpatient treatment study. Patients were men and women age 21 to 70 years from multiple ethnic groups. They were randomized to receive gabapentin or lorazepam; 449 patients were screened and 68 completed the follow-up. Scales used included the CIWA-Ar, Beck Depression Inventory (BDI), and ESS.
Patients receiving lorazepam reported less insomnia and more sleepiness early in treatment than patients receiving gabapentin. However, upon completing treatment and discontinuing medication administration, patients previously treated with lorazepam reported increased insomnia and daytime sleepiness, while patients previously treated with gabapentin continued to report improvements in these self-reported sleep measures. The differences between lorazepam and gabapentin were further evidenced in BDI scores at Day 5, Day 7, and Day 12 in patients who had previously experienced multiple withdrawals. Gabapentin was superior to lorazepam in reducing insomnia as assessed by BDI score, an effect that was sustained throughout the post-treatment week. Participants’ ESS scores indicated less daytime sleepiness in the gabapentin group than in the lorazepam group.
Conclusion: Among patients who abused alcohol and had a history of multiple withdrawals, lorazepam is less effective than gabapentin in reducing insomnia.10 However, this study had several limitations: <25% of individuals who were initially screened were enrolled in the study, and it used subjective tests such as BDI. Objective electrophysiologic measures of sleep and daytime sleepiness would have been very helpful.
Myrick et al11 (2009) also compared gabapentin and lorazepam for treating alcohol withdrawal. One hundred patients were randomized to receive 4 days of fixed-dose taper of gabapentin or lorazepam. Patients could receive 1 of 3 gabapentin dosing regimens (600 mg/d, 900 mg/d, or 1,200 mg/d) for 3 days. Participants who were randomized to receive lorazepam were given 6 mg/d for 3 days and then tapered to 4 mg/d. Also, blinded supplemental medications (rescue packs) were given to each patient on Days 1 to 4 to treat subjective feelings of alcohol withdrawal. All patients also received thiamine for 12 days. Assessment of severity of alcohol withdrawal was measured by the CIWA-Ar. To quantify the severity of alcohol dependence and alcohol use, patients were asked to complete the ADS and Time-Line Follow-Back (TLFB) scales, respectively. Other scales administered included the BDI, Zung Anxiety Scale (ZAS), ESS, and visual analogue scales that assessed craving, ability to perform work, and need for additional medication.
There was a decrease in CIWA-Ar scores over time in all groups. High-dose gabapentin was found to be statistically superior but clinically similar to lorazepam (P = .009). Researchers also found that compared with patients who were treated with lorazepam, patients who were treated with gabapentin experienced reduced craving and anxiety/depressive symptoms, and complained of less subjective discomfort. Compared to patients who were treated with gabapentin, patients who were treated with lorazepam had higher probabilities of drinking on the first day of dose decrease (Day 2) and the second day off medication (Day 6) (P = .0002). During post-treatment, patients who were treated with gabapentin had less probability of drinking during the follow-up post-treatment period (P = .2 for 900 mg/d and P = .3 for 1,200 mg/d) compared with patients who were treated with lorazepam (P = .55).
Continue to: Conclusion
Conclusion: The researchers concluded that gabapentin was well tolerated and effectively diminished the symptoms of alcohol withdrawal, especially at the higher target dose (1,200 mg/d), and that compared with lorazepam, gabapentin decreased the probability of drinking during alcohol withdrawal and in the immediate post-withdrawal week.11
Stock et al12 randomized 26 patients who met criteria for AWS to receive gabapentin or chlordiazepoxide. Gabapentin doses were 1,200 mg/d orally for 3 days, followed by 900 mg/d, 600 mg/d, and 300 mg/d for 1 day each. Chlordiazepoxide doses were 100 mg/d orally for 3 days, followed by 75 mg/d, 50 mg/d, and 25 mg/d for 1 day each. The ESS, Penn Alcohol Craving Scale (PACS), ataxia rating, and CIWA-Ar were administered daily. Thirty-five percent of participants dropped out at the end of the 7-day treatment period. Days 1 to 4 were considered the early treatment period, and Days 5 to 7 were considered the late treatment period. The adjusted mean ESS score did not differ significantly between the randomized groups during the early stage (P = .61) vs the late stage, in which the adjusted mean ESS score was significantly lower with gabapentin compared with chlordiazepoxide (P = .04). The differences in PACS scores between the groups were not statistically significant in either stage (early stage P = .59 vs late stage P = .08), but a trend of lower PACS scores was noted with gabapentin in the later stage. No participant in either group had ataxia during the study. In both groups, CIWA-Ar scores were reduced similarly.
Conclusion: The researchers concluded that gabapentin treatment resulted in a significantly greater reduction in sedation (ESS) and a trend toward reduced alcohol craving (PACS) by the end of treatment compared with chlordiazepoxide treatment.12
Schacht et al13 analyzed functional magnetic resonance imaging data from 48 patients who were alcohol-dependent in a 6-week RCT. Patients were randomized to receive gabapentin up to 1,200 mg/d for 39 days plus flumazenil for 2 days (GP/FMZ group) or an oral placebo and placebo infusions on the same time course. Evaluations included the SCID, ADS, and Obsessive-Compulsive Drinking Scale (OCDS). On Day 1, the CIWA-Ar was administered; it was used to ensure equal distribution of individuals with higher alcohol withdrawal symptoms between medication groups. There were no significant effects of initial alcohol withdrawal symptom status or medication. However, there was a significant interaction between these factors: patients with higher alcohol withdrawal symptoms who received GP/FMZ and those with lower alcohol withdrawal symptoms who received placebo demonstrated greater cue-elicited activation, relative to the other groups, and had less subsequent drinking, which reflected differences in deactivation between alcohol and beverage stimuli, in a cluster that encompassed the dorsal ACC (dACC) (family-wise error-corrected cluster probability of P = .012; 99 voxels; local maxima at [-3, 39, 18] and [6, 33, 9]). In the GP/FMZ group, patients with higher alcohol withdrawal symptoms had significantly greater activation, while in the placebo group, patients with lower alcohol withdrawal symptoms had greater activation.
Conclusion: The researchers concluded that alterations in task-related deactivation of dACC, a component of the default mode network, may predict better alcohol treatment response, while activation of DLPFC, an area associated with selective attention, may predict relapse drinking.13
Continue to: Carbamazepine
Carbamazepine
The characteristics of the carbamazepine studies included in this review are summarized in Table 2.14-19
Björkqvist et al14 randomized 105 men with AWS to placebo or carbamazepine. On initial assessment, history, physical examination, relevant labs, and intoxication assessments were recorded. On subsequent visits, nursing staff recorded withdrawal symptoms for patients as 0 to 2 (0 = no specific symptoms, 1 = patient only complained when asked about specific symptoms, 2 = patient complained of withdrawal symptoms without being asked, or if symptoms were severe or obvious to others). Along with the above, vital signs and a visual analogue scale of 0 to 10 (0 = feeling could not be worse, 10 = feeling could not be better) were recorded at each visit. The dose was weight-dependent and administered as follows: on Days 1 and 2, 1+1+2 tablets of carbamazepine, 200 mg, or placebo; Days 3 and 4, 1+1+1 tablets; and Days 5 and 6, 1+0+1 tablets. Every patient received dichloralphenazone as needed. All patients were given vitamin B 3 times a day. Most withdrawal symptoms decreased faster in the carbamazepine group on Day 2 (P = .01) and on Day 4 (P = .1). On the visual analogue scale, scores varied between patients. On Day 1, the mean score was 2.5 times higher in the carbamazepine group compared with the placebo group, and this difference increased to 3 times by Day 7 (P < .01). The patient’s estimated ability to work improved significantly faster in the carbamazepine group than in the placebo group (P < .01).
Conclusion: The authors concluded that compared with placebo, carbamazepine was able to more quickly decrease withdrawal symptoms, especially insomnia and subjective recovery.14
Ritola et al15 randomized 68 hospitalized men with AWS to carbamazepine, 200 mg/d, or clomethiazole, 300 mg/d, for 1 week. The target withdrawal symptoms included gastrointestinal and sleep disturbances; anxiety; aggressiveness; and cardiovascular, depressive, psychotic, and neurologic symptoms. A 4-point rating scale was used for individual symptoms (0 = no symptom, 1 = mild symptom, 2 = moderate symptom, and 3 = severe symptom). On the day of admission (Day 0), all patients were given 50 to 100 mg of chlordiazepoxide IM and 2 tablets and 4 capsules of the trial preparations (either the tablets or capsules were active, and the others were placebos) in the evening. Five patients dropped out of the clomethiazole group and 1 from the carbamazepine group. No significant difference between the 2 treatments were found by the patient, nurse, or physician.
Conclusion: The authors concluded that carbamazepine seemed to be as effective as clomethiazole in the treatment of milder alcohol withdrawal symptoms. Final treatment results were equally good in both groups. Sleep disturbance resolved faster in the carbamazepine group.15
Continue to: Agricola et al
Agricola et al16 compared carbamazepine to tiapride for treatment of acute AWS. In this study, 60 patients were randomized to carbamazepine, 200 mg 3 times a day, or tiapride, 200 mg 3 times a day. All patients were hospitalized with severe AWS preceding DT. The patients were evaluated for withdrawal symptoms (gastrointestinal and cardiovascular symptoms, sleep disturbances, anxiety, aggression, fear, depression, psychotic symptoms, and certain neurologic symptoms). The severity of these symptoms was scored as follows: 0 = no symptoms; 1 = moderate symptoms; and 2 = severe symptoms. At each visit, an overall evaluation of the patient’s clinical condition was made according to a visual analogue scale (100 = worst condition, 0 = best condition). On Day 7, both the doctor and patient evaluated treatment efficacy according to a 4-point scale (1 = no efficacy, 4 = excellent efficacy). There was no significant difference between carbamazepine and tiapride in terms of total symptoms score and visual analogue scale assessment. Carbamazepine was found to have faster relief of symptoms and a significantly greater reduction in symptom score on Day 2 (P < .01). Carbamazepine had a preferential action on fear, nightmares, and hallucinations. The proportion of patients in whom anxiety improved after treatment was 96.2% for carbamazepine and 71.4% for tiapride (P < .05). Aggressiveness and gastrointestinal discomfort resolved faster in the tiapride group. No cases of DT were observed.
Conclusion: The researchers concluded that either carbamazepine or tiapride provides an appropriate alternative in the treatment of inpatients with severe AWS.16
Stuppaeck et al17 compared the efficacy of carbamazepine to oxazepam in 60 inpatients who had symptoms of alcohol withdrawal. Alcohol withdrawal was measured with the CIWA-A, and patients with scores >20 were enrolled in the study. The Clinical Global Impression (CGI) scale and self-rated Adjective Checklist (ACL) were also used. On Days 1 to 3, patients received oxazepam, 120 mg/d, or carbamazepine, 800 mg/d. From Day 4 to 7, doses were decreased to 90 mg/d and 600 mg/d, respectively. After the 7-day trial, all patients were treated with carbamazepine, 200 mg twice a day on Day 8 and 200 mg at night on Day 9. Two patients withdrew consent and 6 dropped out due to adverse effects. During the 7-day trial, when comparing all improvements on CIWA-A, ACL, and CGI scales, carbamazepine was equivalent to oxazepam up to Day 5, and then superior on Days 6 and 7 (P ≤ .05). No decrease in white blood cell count was found in the carbamazepine group.
Conclusion: The authors concluded that carbamazepine is as effective as oxazepam and may be a viable alternative that does not interact with alcohol or cause delirium.17
Malcolm et al18 compared the effects of carbamazepine and lorazepam in patients in an outpatient setting who had single vs multiple previous alcohol withdrawals. The study included 136 patients who satisfied DSM-IV criteria for alcohol dependence and alcohol withdrawal, with a blood alcohol level ≤0.1 g/dL, a Mini-Mental State Examination (MMSE) score ≤26, and a CIWA-Ar score ≤10 on admission. Patients also completed the ADS to quantify the severity of alcohol dependence. Daily drinking was measured by patient report using a daily drinking log and blood alcohol level. Heavy drinking was defined as ≥4 standard drinks per day for women and ≥5 drinks per day for men. On Day 1, patients were randomized to receive carbamazepine, 600 to 800 mg/d,or lorazepam, 6 to 8 mg/d, in divided doses, which was tapered to carbamazepine, 200 mg/d, or lorazepam, 2 mg/d, on Day 5. All patients received thiamine for 12 days. In the immediate post-detoxification period, carbamazepine-treated patients were less likely to relapse, and if they did drink, they drank less than those treated with lorazepam (P = .003). Even in patients who had multiple previous detoxifications, those randomized to carbamazepine drank less than those in lorazepam group (P = .004). Patients in the lorazepam group had significant higher rebound withdrawal symptoms (P = .007).
Continue to: Conclusion
Conclusion: The researchers concluded that carbamazepine and lorazepam were both effective in reducing alcohol withdrawal symptoms. They also concluded that carbamazepine was less likely to cause rebound withdrawal and more likely to reduce post-treatment drinking; among those who did drink, there was less heavy drinking.18
Malcolm et al19 conducted a 5-day double-blind RCT with 136 outpatients who met DSM-IV criteria for alcohol withdrawal. Patients were evaluated by CIWA before getting medications and then daily for 5 days. Patients were randomized to receive carbamazepine, 600 to 800 mg/d on Day 1, 200 mg 3 times a day on Day 2, 200 mg twice a day on Days 3 and 4, and 200 mg once on Day 5. Participants were randomized to receive lorazepam, 6 to 8 mg/d in divided doses on Day 1, 2 mg 3 times a day on Day 2, 2 mg twice a day on Days 3 and 4, and 2 mg once on Day 5. Ability to return to work was self-rated on a 100-mm visual analogue scale, with 0 being “totally unable to return to work’’ and 100 representing “being fully able to return to work.’’ Self-report measures of sleep quality were made using a 100-mm visual analogue scale, with 0 = “the very worst night’s sleep I’ve ever had’’ and 100 = “the very best night’s sleep I’ve ever had.’’ Carbamazepine significantly reduced anxiety (P = .0007). Visual analogue measures of sleep quality indicated a statistically significant main effect of medication on sleep that favored carbamazepine (P = .0186).
Conclusion: The authors concluded that when treating patients with mild to moderate alcohol withdrawal symptoms, carbamazepine was superior to lorazepam in reducing anxiety and improving sleep.19
Sodium valproate
The characteristics of the sodium valproate studies included in this review are summarized in Table 3.20,21
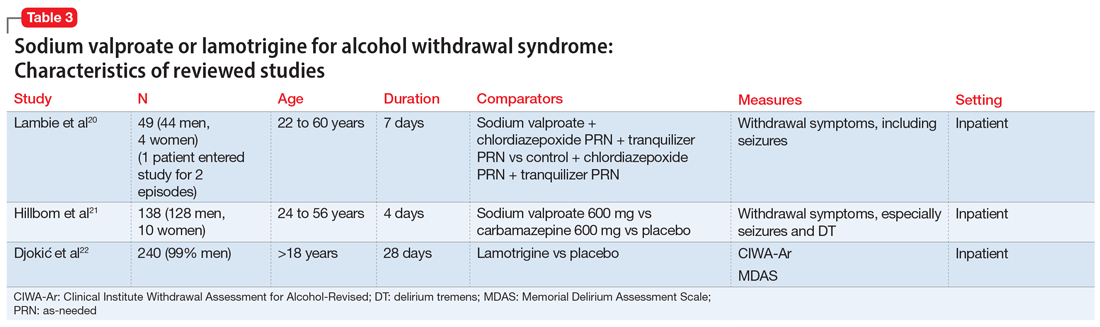
Lambie et al20 evaluated the use of sodium valproate in the treatment of AWS. A total of 49 patients were randomized to a sodium valproate group (n = 22) or a control group (n = 27). All participants were inpatients receiving treatment for alcohol use disorder and substance use disorder for 7 days. Patients in the sodium valproate group received 800 mg every 8 hours for 7 days. Patients were observed daily for occurrence of withdrawal symptoms. Nurses who were blinded to the group assignment graded the degree and severity of symptoms. The trial was initially designed so that chlormethiazole and/or tranquilizers were added to sodium valproate when withdrawal symptoms occurred. However, after treating the first few patients, it became evident that additional medications were not needed. In the treatment group, 13 participants received only sodium valproate, 4 patients needed a tranquilizer, 4 needed chlormethiazole, and 1 needed both. In the control group, 1 received only sodium valproate, 4 received a tranquilizer, 14 received chlormethiazole, and 8 needed both. One patient, who entered the study twice, had a withdrawal seizure when in control group and no seizure on second admission in the sodium valproate group. Physical symptoms disappeared quickly in the sodium valproate group (mean of 2 days vs 2.6 days in the control group). Fourteen patients in the control group received chlormethiazole, compared with only 4 patients in sodium valproate group.
Continue to: Conclusion
Conclusion: The researchers concluded that physical symptoms disappeared quicker in the sodium valproate group than in the control group.20
Hillbom et al21 evaluated the efficacy of sodium valproate vs carbamazepine vs placebo to prevent alcohol withdrawal seizures. A total of 138 participants were studied. Forty-three were assigned to the carbamazepine group, 46 to the sodium valproate group, and 49 to the placebo group. The RCT lasted 4 days. The initial medication doses were 1,200 mg/d. Participants in the carbamazepine group experienced more adverse effects than those in the sodium valproate or placebo groups (P < .001). As a result, approximately one-half of the participants in the carbamazepine group stopped taking the medication. This finding was dependent on the dose of carbamazepine; >800 mg/d resulted in poor tolerance to adverse effects. Seizures occurred among patients in all 3 arms of the study; in the sodium valproate group, 1 participant had a seizure vs 2 participants in the carbamazepine group and 3 in the placebo group. On the other hand, DT occurred only in the sodium valproate and placebo groups.
Conclusion: Researchers concluded that when using sodium valproate or carbamazepine to prevent alcohol withdrawal seizures in an outpatient setting, the adverse effects may outweigh the benefits.21
Lamotrigine
The characteristics of the lamotrigine study included in this review are summarized in Table 3.22
Djokić et al22 evaluated the efficiency of lamotrigine in the treatment of DT. A total of 240 participants who met International Classification of Diseases-10 criteria for DT were randomized to a control group that was treated with anticonvulsants according to an NIAAA protocol (2004), or to an experimental group that was treated with lamotrigine. The CIWA-Ar and the Memorial Delirium Assessment Scale (MDAS) were administered for objective assessment of clinical symptoms, superimposed medical complications, general condition of the patient, adverse effects, and mortality rate. Statistically significant differences between the experimental and control groups were apparent after the third day of therapy, when a drop in the average CIWA-Ar score was observed in the experimental group, while the control group still had high scores (P < .01). After the fifth day of treatment, the differences in scores were more apparent, with the experimental group showing CIWA-Ar scores equal to those of persons with mild/moderate DT, while those in the control group still had high scores. After the tenth day, participants in the experimental group did not have any alcohol withdrawal symptoms, while control group participants were just beginning to get out of life-threatening danger. Death occurred in 4.1% of control group participants and 3.4% of experimental group participants; this difference in mortality rate was not statistically significant.
Continue to: Conclusion
Conclusion: Researchers concluded that lamotrigine is significantly efficacious in the treatment of DT, but does not decrease the mortality rate.22
What to know before you prescribe
AWS is a medical emergency that if left untreated leads to several complications and possibly death. Although benzodiazepines are considered the gold standard for treating AWS, the adverse effects associated with their use advocates for finding alternatives. Anticonvulsants can be an effective alternative for treating AWS. In our literature review, we found 16 double-blind RCTs that used an anticonvulsant medication for the treatment of AWS. Of these, 7 involved gabapentin, 6 involved carbamazepine, 1 involved sodium valproate, 1 involved sodium valproate vs carbamazepine, and 1 involved lamotrigine. Overall, the use of anticonvulsants resulted in significant improvement of mild to moderate symptoms of AWS.
There were more studies of carbamazepine and gabapentin than of other anticonvulsants. All the anticonvulsants offered potential benefits. They decreased the probability of a withdrawal seizure and other complications and effectively reduced alcohol cravings. Anticonvulsants were useful for preventing rebound withdrawal symptoms and reducing post-treatment alcohol consumption, especially in patients who had multiple previous withdrawals. Anticonvulsants were particularly helpful for patients with mood disorders such as depression. In the studies we reviewed, anticonvulsants caused less sedation compared with benzodiazepines, and also decreased the occurrence of relapse.
Dosing recommendations. In the studies included in our review, gabapentin was effective at a dosage of 1,600 mg/d (given as 400 mg 4 times a day). This was tapered as follows: 400 mg 4 times a day on Days 1 to 3, 400 mg 3 times a day on Day 4, 400 mg twice a day on Day 5, and 400 mg once a day on Day 6. Carbamazepine was effective at 600 to 800 mg/d, and was tapered by decreasing by 200 mg as follows: 800 mg/d on Days 1 to 3, 600 mg/d on Day 4, 400 mg on Day 5, and 200 mg/d on Day 6. In the reviewed study, the maximum dose of lamotrigine never exceeded 200 mg/d and was administered for 28 days; the exact dosing and taper plan were not described. The dosing of sodium valproate ranged from 1,200 mg/d to 1600 mg/d for 7 days, followed by decreasing by 200 mg each day. The recommended duration of treatment varied; on average for all anticonvulsants, it was 7 to 12 days, followed by a taper. Carbamazepine was shown to be superior to oxazepam in ameliorating the symptoms of AWS.
Adverse effects. When considering the tolerability, adverse effect profile, duration of action, and effectiveness of the anticonvulsants included in our review, gabapentin appears to be the safest agent to choose. For the other anticonvulsants, the risks might outweigh the benefits. Specifically, in a comparison of sodium valproate and carbamazepine, Hillbom et al21 concluded that in doses >800 mg/d, carbamazepine has potential to cause more adverse effects than benefits. However, Agricola et al16 found that carbamazepine had a preferential action on fear, nightmares, and hallucinations.
Continue to: A few caveats
A few caveats
Our review focused a large collection of data from multiple databases and RCTs only. However, its limitations include:
- there was no measure of heterogeneity
- the studies had short treatment duration
- most studies evaluated predominantly male participants
- some studies were underpowered.
Our review laid a groundwork for future research that includes more well-designed RCTs and/or meta-analyses of recent studies that evaluated the use anticonvulsants for treating AWS.
Bottom Line
Evidence suggests certain anticonvulsants may be an effective alternative to benzodiazepines for the treatment of mild to moderate alcohol withdrawal syndrome. Gabapentin may be the safest anticonvulsant to prescribe. Other anticonvulsants to consider include carbamazepine, sodium valproate, and lamotrigine, but for these agents, the risks might outweigh the benefits.
Related Resources
- Myrick H, Anton RF. Treatment of alcohol withdrawal. Alcohol Health Res World. 1998;22(1):38-43. https://pubs.niaaa.nih.gov/publications/arh22-1/38-43.pdf
- World Health Organization. Management of alcohol withdrawal. Published 2012. https://www.who.int/mental_health/mhgap/evidence/alcohol/q2/en/
Drug Brand Names
Carbamazepine • Tegretol
Gabapentin • Neurontin
Lamotrigine • Lamictal
Levetiracetam • Keppra
Lorazepam • Ativan
Oxcarbazepine • Trileptal
Phenytoin • Dilantin
Sodium valproate • Depakote
Acknowledgments
The authors thank Geetha Manikkara, MD, Madhuri Jakkam Setty, MD, and Elizabeth DeOreo, MD, for their efforts with the systematic review research.
1. Trevisan LA, Boutros N, Petrakis IL, et al. Complications of alcohol withdrawal: pathophysiological insights. Alcohol Health Res World. 1998;22(1):61-66.
2. Borghesani P. Alcohol withdrawal. In: Nordstrom KD, Wilson MP, eds. Quick guide to psychiatric emergencies. Springer: 2018;209-215.
3. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 alcohol use disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766.
4. Ungur LA, Neuner B, John S, et al. Prevention and therapy of alcohol withdrawal on intensive care units: systematic review of controlled trials. Alcohol Clin Exp Res. 2013;37(4):675-686.
5. Sachdeva A, Choudhary M, Chandra M. Alcohol withdrawal syndrome: benzodiazepines and beyond. J Clin Diagn Res. 2015;9(9):VE01-VE07.
6. Ashton H. Toxicity and adverse consequences of benzodiazepine use. Psychiatr Ann. 1995;25:158-165.
7. Bonnet U, Banger M, Leweke FM, et al. Treatment of acute alcohol withdrawal with gabapentin: results from a controlled two-center trial. J Clin Psychopharmacol. 2003;23(5):514-519.
8. Bonnet U, Specka M, Leweke FM, et al. Gabapentin’s acute effect on mood profile--a controlled study on patients with alcohol withdrawal. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):434-438.
9. Myrick H, Anton R, Voronin K, et al. A double-blind evaluation of gabapentin on alcohol effects and drinking in a clinical laboratory paradigm. Alcohol Clin Exp Res. 2007;31(2):221-227.
10. Malcolm R, Myrick L, Veatch L, et al. Self-reported sleep, sleepiness, and repeated alcohol withdrawals: a randomized, double blind, controlled comparison of lorazepam vs gabapentin. J Clin Sleep Med. 2007;3(1):24-32.
11. Myrick H, Malcolm R, Randall PK, et al. A double-blind trial of gabapentin versus lorazepam in the treatment of alcohol withdrawal. Alcohol Clin Exp Res. 2009;33(9):1582-1588.
12. Stock CJ, Carpenter L, Ying J, et al. Gabapentin versus chlordiazepoxide for outpatient alcohol detoxification treatment. Ann Pharmacother. 2013;47(7-8):961-969.
13. Schacht JP, Anton RF, Randall PK, et al. Effects of a GABA-ergic medication combination and initial alcohol withdrawal severity on cue-elicited brain activation among treatment-seeking alcoholics. Psychopharmacol. 2013;227(4):627-637.
14. Björkqvist SE, Isohanni M, Mäkelä R, et al. Ambulant treatment of alcohol withdrawal symptoms with carbamazepine: a formal multicenter double blind comparison with placebo. Acta Psychiatr Scand. 1976;53(5):333-342.
15. Ritola E, Malinen L. A double-blind comparison of carbamazepine and clomethiazole in the treatment of alcohol withdrawal syndrome. Acta Psychiatr Scand. 1981;64(3):254-259.
16. Agricola R, Mazzarino M, Urani R, et al. Treatment of acute alcohol withdrawal syndrome with carbamazepine: a double-blind comparison with tiapride. J Int Med Res. 1982;10(3):160-165.
17. Stuppaeck CH, Pycha R, Miller C, et al. Carbamazepine versus oxazepam in the treatment of alcohol withdrawal: a double-blind study. Alcohol Alcohol. 1992;27(2):153-158.
18. Malcolm R, Myrick H, Roberts J, et al. The effects of carbamazepine and lorazepam on single vs multiple previous withdrawals in an outpatient randomized trial. J Gen Intern Med. 2002;17(5):349-355.
19. Malcolm R, Myrick H, Roberts J, et al. The differential effects of medications on mood, sleep disturbance, and work ability in outpatient alcohol detoxification. Am J Addict. 2002;11(2):141-150.
20. Lambie D, Johnson R, Vijayasenan M, et al. Sodium valproate in the treatment of the alcohol withdrawal syndrome. Aust N Z J. 1980;14(3):213-215.
21. Hillbom M, Tokola R, Kuusela V, et al. Prevention of alcohol withdrawal seizures with carbamazepine and valproic acid. Alcohol. 1989;6(3):223-226.
22. Djokic
Abrupt cessation or reduction of alcohol consumption may result in alcohol withdrawal syndrome (AWS), which is a medical emergency that can lead to serious complications when unrecognized or treatment is delayed. Symptoms of AWS include tremors, anxiety attacks, cognitive impairment, hallucinations, seizures, delirium tremens (DT), and in severe, untreated cases, death.1 Low to moderate alcohol consumption produces euphoria and excitation via activation of glutamatergic neurotransmission, while higher concentrations produce severe intoxication via GABAergic mechanisms. Acute withdrawal unmasks the hyper-excitatory state of the brain, causing anxiety, agitation, and autonomic activation characteristic of AWS, which typically begins 1 to 3 days after the last drink.2 In the 2012-2013 National Epidemiologic Survey on Alcohol and Related Conditions conducted by the National Institute on Alcohol Abuse and Alcoholism (NIAAA), the 12-month and lifetime prevalences of AWS were 13.9% and 29.1%, respectively.3 Within the general inpatient population, AWS can be present in nearly 30% of patients; if left untreated, AWS has a 15% mortality rate, although when AWS is recognized early and treated, the mortality rate falls dramatically to 2%.4
AWS has most commonly been treated with benzodiazepines.5 However, benzodiazepines have the potential for significant adverse effects when used in older adults and in individuals with complicated medical issues, such as obstructive lung disease and sleep apnea.6 Anticonvulsants have been increasingly used to treat alcohol withdrawal, and their use is supported by several retrospective and prospective studies. In this article, we review the data from randomized control trials (RCTs) on the use of anticonvulsants for the treatment of AWS to see if we can make any recommendations for the use of anticonvulsants for treating AWS.
Our literature search
We searched 5 databases (PubMed, Cochrane, Medline, PsycInfo, and Embase) using the following terms: “alcohol withdrawal syndrome treatment”, “anticonvulsants”, “anti-epileptic”, “gabapentin”, “carbamazepine”, “sodium valproate”, “oxcarbazepine”, “phenytoin”, “levetiracetam”, and “lamotrigine.” We included only double-blind RCTs published between January 1, 1976 and September 30, 2016 in English-language journals or that had an official English translation. There were no restrictions on patient age or location of treatment (inpatient vs outpatient). All RCTs that compared anticonvulsants or a combination of an anticonvulsant and an active pharmacotherapeutic agent with either placebo or gold standard treatment for AWS were included. Database reviews, systematic reviews, and meta-analyses were excluded.
We identified 662 articles that met these criteria. However, most were duplicates, review articles, systematic reviews, meta-analyses, case reports, or open-label or non-randomized trials. Only 16 articles met our inclusion criteria. In the following sections, we discuss these 16 studies by medication type and in chronological order.
Gabapentin
The characteristics of the gabapentin studies included in this review are summarized in Table 1.7-13

Bonnet et al7 (2003) examined 61 adults who met the clinical criteria for alcohol dependence and displayed moderate or severe AWS according to their Mainz Alcohol Withdrawal Score (MAWS ≥4). They were randomized to receive placebo or gabapentin, 400 mg 4 times a day, along with clomethiazole. The attrition rate was not significantly different between the 2 groups (P = .66). The difference in the number of clomethiazole capsules taken during the first 24 hours between the groups was small and not significant (P = .96). Analysis of MAWS over time revealed no significant main effect for group (P = .26) and a significant effect for the time variable (P < .001). The interaction between group and time was not significant (P =.4). This means that there was a significant decrease in MAWS from baseline over 48 hours, and this decrease in MAWS was considered equal for both study groups. Adverse clinical events were observed in both groups, and there was no significant difference (P = .74) between the groups. Nausea and ataxia, which are specific to gabapentin, were observed more frequently in this group.
Conclusion: The authors concluded that gabapentin, 400 mg 4 times a day, is no better than placebo in reducing the amount of clomethiazole required to treat acute AWS.7
Continue to: Bonnet et al
Bonnet et al8 (2007) also conducted a study examining 59 patients with alcohol dependence who displayed moderate or severe AWS. Participants received placebo or gabapentin, 400 mg, and a rescue medication, clomethiazole, if needed. Subsequently, a capsule of study medication was administered every 6 hours for 2 days and then tapered. During the study, mood was measured by Profile of Mood States (POMS), and subjective complaints of withdrawal were measured using the Essen Self-Assessment of Alcohol Withdrawal Scale (ESA). Of the 59 patients, only 46 were analyzed; 5 patients dropped out, and 8 patients were missing data. Compared with the placebo group, the gabapentin group displayed less dejection, fatigue, and anger, and more vigor. Analysis of variance (ANOVA) measures revealed significant overall changes over time on all 4 scales (all P < .001). A significant (F = 3.62, df 2;43, P = .035) group × time interaction resulted exclusively for vigor. Analysis was repeated using rank-transformed data, resulting in a significant (P = .046) interaction effect. The significant increase in vigor was not apparent after tapering off gabapentin, which suggests gabapentin has a reversible effect on vigor. There was a significant (P < .001) overall decline of subjective withdrawal symptoms complaints, but no group × time interaction (P = .62). Analysis of 11 patients with comorbid mild depression revealed no significant time × group interaction for dejection, fatigue, anger, or subjective withdrawal (all P > .05). However, for vigor, the group × time interaction was significant (P = .022). Throughout the treatment, vigor scores of those mild depressive patients who received gabapentin increased to a level comparable to that of patients without a mood disorder.
Conclusion: The authors authors concluded that gabapentin was markedly more efficacious in improving vigor in the small subgroup of patients with mild depression.8
Myrick et al9 (2007) evaluated the safety and tolerability of gabapentin in patients who abused alcohol, as well as the ability of gabapentin to reduce alcohol craving and consumption. This study included 35 participants randomly assigned to receive gabapentin (n = 17) or placebo (n = 18) for 7 days. All medications were administered in standard gel caps with riboflavin, 25 mg, to assess for compliance via a laboratory-based urinary fluorescence assay. Urine samples were assessed for riboflavin at baseline and Day 6, and a reading of 1,500 ng/mL of riboflavin on Day 6 was interpreted as being compliant. Participants were required to abstain completely from drinking alcohol on Day 6 and the morning of Day 7. At the first session, the following measures were completed: demographic form, alcohol and drug section of the Structured Clinical Interview (SCID), Obsessive-Compulsive Drinking Scale, Self-Administered Alcohol Screening Test (SAAST), and Alcohol Dependence Scale (ADS); there also was collection of a urine sample for detection of abused drugs and a blood sample for liver function and general health screening.
At the second session, patients completed the psychiatric sections of the SCID and the Alcohol Craving Questionnaire, and received a physical exam. To assess the negative clinical effects of gabapentin and alcohol on the CNS, the Epworth Sleepiness Scale (ESS) and POMS were administered at baseline and on Day 6. Also, several other scales were used to identify any impact of gabapentin on acute alcohol effects and craving: the Clinical Institute Withdrawal Assessment of Alcohol, Revised (CIWA-Ar), Biphasic Alcohol Effects Scale (BAES), Subjective High Assessment Scale (SHAS), and Alcohol Urge Questionnaire (AUQ).
Conclusion: Gabapentin was well tolerated, but compared with placebo, gabapentin had no effect on alcohol stimulation (P = .75) or sedation (P = .99) as measured by the BAES. The difference in SHAS scores was also not significant (P = .19). There was also no significant reduction in craving for alcohol as measured on the AUQ scale
Continue to: Malcolm et al
Malcolm et al10 conducted an outpatient treatment study. Patients were men and women age 21 to 70 years from multiple ethnic groups. They were randomized to receive gabapentin or lorazepam; 449 patients were screened and 68 completed the follow-up. Scales used included the CIWA-Ar, Beck Depression Inventory (BDI), and ESS.
Patients receiving lorazepam reported less insomnia and more sleepiness early in treatment than patients receiving gabapentin. However, upon completing treatment and discontinuing medication administration, patients previously treated with lorazepam reported increased insomnia and daytime sleepiness, while patients previously treated with gabapentin continued to report improvements in these self-reported sleep measures. The differences between lorazepam and gabapentin were further evidenced in BDI scores at Day 5, Day 7, and Day 12 in patients who had previously experienced multiple withdrawals. Gabapentin was superior to lorazepam in reducing insomnia as assessed by BDI score, an effect that was sustained throughout the post-treatment week. Participants’ ESS scores indicated less daytime sleepiness in the gabapentin group than in the lorazepam group.
Conclusion: Among patients who abused alcohol and had a history of multiple withdrawals, lorazepam is less effective than gabapentin in reducing insomnia.10 However, this study had several limitations: <25% of individuals who were initially screened were enrolled in the study, and it used subjective tests such as BDI. Objective electrophysiologic measures of sleep and daytime sleepiness would have been very helpful.
Myrick et al11 (2009) also compared gabapentin and lorazepam for treating alcohol withdrawal. One hundred patients were randomized to receive 4 days of fixed-dose taper of gabapentin or lorazepam. Patients could receive 1 of 3 gabapentin dosing regimens (600 mg/d, 900 mg/d, or 1,200 mg/d) for 3 days. Participants who were randomized to receive lorazepam were given 6 mg/d for 3 days and then tapered to 4 mg/d. Also, blinded supplemental medications (rescue packs) were given to each patient on Days 1 to 4 to treat subjective feelings of alcohol withdrawal. All patients also received thiamine for 12 days. Assessment of severity of alcohol withdrawal was measured by the CIWA-Ar. To quantify the severity of alcohol dependence and alcohol use, patients were asked to complete the ADS and Time-Line Follow-Back (TLFB) scales, respectively. Other scales administered included the BDI, Zung Anxiety Scale (ZAS), ESS, and visual analogue scales that assessed craving, ability to perform work, and need for additional medication.
There was a decrease in CIWA-Ar scores over time in all groups. High-dose gabapentin was found to be statistically superior but clinically similar to lorazepam (P = .009). Researchers also found that compared with patients who were treated with lorazepam, patients who were treated with gabapentin experienced reduced craving and anxiety/depressive symptoms, and complained of less subjective discomfort. Compared to patients who were treated with gabapentin, patients who were treated with lorazepam had higher probabilities of drinking on the first day of dose decrease (Day 2) and the second day off medication (Day 6) (P = .0002). During post-treatment, patients who were treated with gabapentin had less probability of drinking during the follow-up post-treatment period (P = .2 for 900 mg/d and P = .3 for 1,200 mg/d) compared with patients who were treated with lorazepam (P = .55).
Continue to: Conclusion
Conclusion: The researchers concluded that gabapentin was well tolerated and effectively diminished the symptoms of alcohol withdrawal, especially at the higher target dose (1,200 mg/d), and that compared with lorazepam, gabapentin decreased the probability of drinking during alcohol withdrawal and in the immediate post-withdrawal week.11
Stock et al12 randomized 26 patients who met criteria for AWS to receive gabapentin or chlordiazepoxide. Gabapentin doses were 1,200 mg/d orally for 3 days, followed by 900 mg/d, 600 mg/d, and 300 mg/d for 1 day each. Chlordiazepoxide doses were 100 mg/d orally for 3 days, followed by 75 mg/d, 50 mg/d, and 25 mg/d for 1 day each. The ESS, Penn Alcohol Craving Scale (PACS), ataxia rating, and CIWA-Ar were administered daily. Thirty-five percent of participants dropped out at the end of the 7-day treatment period. Days 1 to 4 were considered the early treatment period, and Days 5 to 7 were considered the late treatment period. The adjusted mean ESS score did not differ significantly between the randomized groups during the early stage (P = .61) vs the late stage, in which the adjusted mean ESS score was significantly lower with gabapentin compared with chlordiazepoxide (P = .04). The differences in PACS scores between the groups were not statistically significant in either stage (early stage P = .59 vs late stage P = .08), but a trend of lower PACS scores was noted with gabapentin in the later stage. No participant in either group had ataxia during the study. In both groups, CIWA-Ar scores were reduced similarly.
Conclusion: The researchers concluded that gabapentin treatment resulted in a significantly greater reduction in sedation (ESS) and a trend toward reduced alcohol craving (PACS) by the end of treatment compared with chlordiazepoxide treatment.12
Schacht et al13 analyzed functional magnetic resonance imaging data from 48 patients who were alcohol-dependent in a 6-week RCT. Patients were randomized to receive gabapentin up to 1,200 mg/d for 39 days plus flumazenil for 2 days (GP/FMZ group) or an oral placebo and placebo infusions on the same time course. Evaluations included the SCID, ADS, and Obsessive-Compulsive Drinking Scale (OCDS). On Day 1, the CIWA-Ar was administered; it was used to ensure equal distribution of individuals with higher alcohol withdrawal symptoms between medication groups. There were no significant effects of initial alcohol withdrawal symptom status or medication. However, there was a significant interaction between these factors: patients with higher alcohol withdrawal symptoms who received GP/FMZ and those with lower alcohol withdrawal symptoms who received placebo demonstrated greater cue-elicited activation, relative to the other groups, and had less subsequent drinking, which reflected differences in deactivation between alcohol and beverage stimuli, in a cluster that encompassed the dorsal ACC (dACC) (family-wise error-corrected cluster probability of P = .012; 99 voxels; local maxima at [-3, 39, 18] and [6, 33, 9]). In the GP/FMZ group, patients with higher alcohol withdrawal symptoms had significantly greater activation, while in the placebo group, patients with lower alcohol withdrawal symptoms had greater activation.
Conclusion: The researchers concluded that alterations in task-related deactivation of dACC, a component of the default mode network, may predict better alcohol treatment response, while activation of DLPFC, an area associated with selective attention, may predict relapse drinking.13
Continue to: Carbamazepine
Carbamazepine
The characteristics of the carbamazepine studies included in this review are summarized in Table 2.14-19

Björkqvist et al14 randomized 105 men with AWS to placebo or carbamazepine. On initial assessment, history, physical examination, relevant labs, and intoxication assessments were recorded. On subsequent visits, nursing staff recorded withdrawal symptoms for patients as 0 to 2 (0 = no specific symptoms, 1 = patient only complained when asked about specific symptoms, 2 = patient complained of withdrawal symptoms without being asked, or if symptoms were severe or obvious to others). Along with the above, vital signs and a visual analogue scale of 0 to 10 (0 = feeling could not be worse, 10 = feeling could not be better) were recorded at each visit. The dose was weight-dependent and administered as follows: on Days 1 and 2, 1+1+2 tablets of carbamazepine, 200 mg, or placebo; Days 3 and 4, 1+1+1 tablets; and Days 5 and 6, 1+0+1 tablets. Every patient received dichloralphenazone as needed. All patients were given vitamin B 3 times a day. Most withdrawal symptoms decreased faster in the carbamazepine group on Day 2 (P = .01) and on Day 4 (P = .1). On the visual analogue scale, scores varied between patients. On Day 1, the mean score was 2.5 times higher in the carbamazepine group compared with the placebo group, and this difference increased to 3 times by Day 7 (P < .01). The patient’s estimated ability to work improved significantly faster in the carbamazepine group than in the placebo group (P < .01).
Conclusion: The authors concluded that compared with placebo, carbamazepine was able to more quickly decrease withdrawal symptoms, especially insomnia and subjective recovery.14
Ritola et al15 randomized 68 hospitalized men with AWS to carbamazepine, 200 mg/d, or clomethiazole, 300 mg/d, for 1 week. The target withdrawal symptoms included gastrointestinal and sleep disturbances; anxiety; aggressiveness; and cardiovascular, depressive, psychotic, and neurologic symptoms. A 4-point rating scale was used for individual symptoms (0 = no symptom, 1 = mild symptom, 2 = moderate symptom, and 3 = severe symptom). On the day of admission (Day 0), all patients were given 50 to 100 mg of chlordiazepoxide IM and 2 tablets and 4 capsules of the trial preparations (either the tablets or capsules were active, and the others were placebos) in the evening. Five patients dropped out of the clomethiazole group and 1 from the carbamazepine group. No significant difference between the 2 treatments were found by the patient, nurse, or physician.
Conclusion: The authors concluded that carbamazepine seemed to be as effective as clomethiazole in the treatment of milder alcohol withdrawal symptoms. Final treatment results were equally good in both groups. Sleep disturbance resolved faster in the carbamazepine group.15
Continue to: Agricola et al
Agricola et al16 compared carbamazepine to tiapride for treatment of acute AWS. In this study, 60 patients were randomized to carbamazepine, 200 mg 3 times a day, or tiapride, 200 mg 3 times a day. All patients were hospitalized with severe AWS preceding DT. The patients were evaluated for withdrawal symptoms (gastrointestinal and cardiovascular symptoms, sleep disturbances, anxiety, aggression, fear, depression, psychotic symptoms, and certain neurologic symptoms). The severity of these symptoms was scored as follows: 0 = no symptoms; 1 = moderate symptoms; and 2 = severe symptoms. At each visit, an overall evaluation of the patient’s clinical condition was made according to a visual analogue scale (100 = worst condition, 0 = best condition). On Day 7, both the doctor and patient evaluated treatment efficacy according to a 4-point scale (1 = no efficacy, 4 = excellent efficacy). There was no significant difference between carbamazepine and tiapride in terms of total symptoms score and visual analogue scale assessment. Carbamazepine was found to have faster relief of symptoms and a significantly greater reduction in symptom score on Day 2 (P < .01). Carbamazepine had a preferential action on fear, nightmares, and hallucinations. The proportion of patients in whom anxiety improved after treatment was 96.2% for carbamazepine and 71.4% for tiapride (P < .05). Aggressiveness and gastrointestinal discomfort resolved faster in the tiapride group. No cases of DT were observed.
Conclusion: The researchers concluded that either carbamazepine or tiapride provides an appropriate alternative in the treatment of inpatients with severe AWS.16
Stuppaeck et al17 compared the efficacy of carbamazepine to oxazepam in 60 inpatients who had symptoms of alcohol withdrawal. Alcohol withdrawal was measured with the CIWA-A, and patients with scores >20 were enrolled in the study. The Clinical Global Impression (CGI) scale and self-rated Adjective Checklist (ACL) were also used. On Days 1 to 3, patients received oxazepam, 120 mg/d, or carbamazepine, 800 mg/d. From Day 4 to 7, doses were decreased to 90 mg/d and 600 mg/d, respectively. After the 7-day trial, all patients were treated with carbamazepine, 200 mg twice a day on Day 8 and 200 mg at night on Day 9. Two patients withdrew consent and 6 dropped out due to adverse effects. During the 7-day trial, when comparing all improvements on CIWA-A, ACL, and CGI scales, carbamazepine was equivalent to oxazepam up to Day 5, and then superior on Days 6 and 7 (P ≤ .05). No decrease in white blood cell count was found in the carbamazepine group.
Conclusion: The authors concluded that carbamazepine is as effective as oxazepam and may be a viable alternative that does not interact with alcohol or cause delirium.17
Malcolm et al18 compared the effects of carbamazepine and lorazepam in patients in an outpatient setting who had single vs multiple previous alcohol withdrawals. The study included 136 patients who satisfied DSM-IV criteria for alcohol dependence and alcohol withdrawal, with a blood alcohol level ≤0.1 g/dL, a Mini-Mental State Examination (MMSE) score ≤26, and a CIWA-Ar score ≤10 on admission. Patients also completed the ADS to quantify the severity of alcohol dependence. Daily drinking was measured by patient report using a daily drinking log and blood alcohol level. Heavy drinking was defined as ≥4 standard drinks per day for women and ≥5 drinks per day for men. On Day 1, patients were randomized to receive carbamazepine, 600 to 800 mg/d,or lorazepam, 6 to 8 mg/d, in divided doses, which was tapered to carbamazepine, 200 mg/d, or lorazepam, 2 mg/d, on Day 5. All patients received thiamine for 12 days. In the immediate post-detoxification period, carbamazepine-treated patients were less likely to relapse, and if they did drink, they drank less than those treated with lorazepam (P = .003). Even in patients who had multiple previous detoxifications, those randomized to carbamazepine drank less than those in lorazepam group (P = .004). Patients in the lorazepam group had significant higher rebound withdrawal symptoms (P = .007).
Continue to: Conclusion
Conclusion: The researchers concluded that carbamazepine and lorazepam were both effective in reducing alcohol withdrawal symptoms. They also concluded that carbamazepine was less likely to cause rebound withdrawal and more likely to reduce post-treatment drinking; among those who did drink, there was less heavy drinking.18
Malcolm et al19 conducted a 5-day double-blind RCT with 136 outpatients who met DSM-IV criteria for alcohol withdrawal. Patients were evaluated by CIWA before getting medications and then daily for 5 days. Patients were randomized to receive carbamazepine, 600 to 800 mg/d on Day 1, 200 mg 3 times a day on Day 2, 200 mg twice a day on Days 3 and 4, and 200 mg once on Day 5. Participants were randomized to receive lorazepam, 6 to 8 mg/d in divided doses on Day 1, 2 mg 3 times a day on Day 2, 2 mg twice a day on Days 3 and 4, and 2 mg once on Day 5. Ability to return to work was self-rated on a 100-mm visual analogue scale, with 0 being “totally unable to return to work’’ and 100 representing “being fully able to return to work.’’ Self-report measures of sleep quality were made using a 100-mm visual analogue scale, with 0 = “the very worst night’s sleep I’ve ever had’’ and 100 = “the very best night’s sleep I’ve ever had.’’ Carbamazepine significantly reduced anxiety (P = .0007). Visual analogue measures of sleep quality indicated a statistically significant main effect of medication on sleep that favored carbamazepine (P = .0186).
Conclusion: The authors concluded that when treating patients with mild to moderate alcohol withdrawal symptoms, carbamazepine was superior to lorazepam in reducing anxiety and improving sleep.19
Sodium valproate
The characteristics of the sodium valproate studies included in this review are summarized in Table 3.20,21

Lambie et al20 evaluated the use of sodium valproate in the treatment of AWS. A total of 49 patients were randomized to a sodium valproate group (n = 22) or a control group (n = 27). All participants were inpatients receiving treatment for alcohol use disorder and substance use disorder for 7 days. Patients in the sodium valproate group received 800 mg every 8 hours for 7 days. Patients were observed daily for occurrence of withdrawal symptoms. Nurses who were blinded to the group assignment graded the degree and severity of symptoms. The trial was initially designed so that chlormethiazole and/or tranquilizers were added to sodium valproate when withdrawal symptoms occurred. However, after treating the first few patients, it became evident that additional medications were not needed. In the treatment group, 13 participants received only sodium valproate, 4 patients needed a tranquilizer, 4 needed chlormethiazole, and 1 needed both. In the control group, 1 received only sodium valproate, 4 received a tranquilizer, 14 received chlormethiazole, and 8 needed both. One patient, who entered the study twice, had a withdrawal seizure when in control group and no seizure on second admission in the sodium valproate group. Physical symptoms disappeared quickly in the sodium valproate group (mean of 2 days vs 2.6 days in the control group). Fourteen patients in the control group received chlormethiazole, compared with only 4 patients in sodium valproate group.
Continue to: Conclusion
Conclusion: The researchers concluded that physical symptoms disappeared quicker in the sodium valproate group than in the control group.20
Hillbom et al21 evaluated the efficacy of sodium valproate vs carbamazepine vs placebo to prevent alcohol withdrawal seizures. A total of 138 participants were studied. Forty-three were assigned to the carbamazepine group, 46 to the sodium valproate group, and 49 to the placebo group. The RCT lasted 4 days. The initial medication doses were 1,200 mg/d. Participants in the carbamazepine group experienced more adverse effects than those in the sodium valproate or placebo groups (P < .001). As a result, approximately one-half of the participants in the carbamazepine group stopped taking the medication. This finding was dependent on the dose of carbamazepine; >800 mg/d resulted in poor tolerance to adverse effects. Seizures occurred among patients in all 3 arms of the study; in the sodium valproate group, 1 participant had a seizure vs 2 participants in the carbamazepine group and 3 in the placebo group. On the other hand, DT occurred only in the sodium valproate and placebo groups.
Conclusion: Researchers concluded that when using sodium valproate or carbamazepine to prevent alcohol withdrawal seizures in an outpatient setting, the adverse effects may outweigh the benefits.21
Lamotrigine
The characteristics of the lamotrigine study included in this review are summarized in Table 3.22
Djokić et al22 evaluated the efficiency of lamotrigine in the treatment of DT. A total of 240 participants who met International Classification of Diseases-10 criteria for DT were randomized to a control group that was treated with anticonvulsants according to an NIAAA protocol (2004), or to an experimental group that was treated with lamotrigine. The CIWA-Ar and the Memorial Delirium Assessment Scale (MDAS) were administered for objective assessment of clinical symptoms, superimposed medical complications, general condition of the patient, adverse effects, and mortality rate. Statistically significant differences between the experimental and control groups were apparent after the third day of therapy, when a drop in the average CIWA-Ar score was observed in the experimental group, while the control group still had high scores (P < .01). After the fifth day of treatment, the differences in scores were more apparent, with the experimental group showing CIWA-Ar scores equal to those of persons with mild/moderate DT, while those in the control group still had high scores. After the tenth day, participants in the experimental group did not have any alcohol withdrawal symptoms, while control group participants were just beginning to get out of life-threatening danger. Death occurred in 4.1% of control group participants and 3.4% of experimental group participants; this difference in mortality rate was not statistically significant.
Continue to: Conclusion
Conclusion: Researchers concluded that lamotrigine is significantly efficacious in the treatment of DT, but does not decrease the mortality rate.22
What to know before you prescribe
AWS is a medical emergency that if left untreated leads to several complications and possibly death. Although benzodiazepines are considered the gold standard for treating AWS, the adverse effects associated with their use advocates for finding alternatives. Anticonvulsants can be an effective alternative for treating AWS. In our literature review, we found 16 double-blind RCTs that used an anticonvulsant medication for the treatment of AWS. Of these, 7 involved gabapentin, 6 involved carbamazepine, 1 involved sodium valproate, 1 involved sodium valproate vs carbamazepine, and 1 involved lamotrigine. Overall, the use of anticonvulsants resulted in significant improvement of mild to moderate symptoms of AWS.
There were more studies of carbamazepine and gabapentin than of other anticonvulsants. All the anticonvulsants offered potential benefits. They decreased the probability of a withdrawal seizure and other complications and effectively reduced alcohol cravings. Anticonvulsants were useful for preventing rebound withdrawal symptoms and reducing post-treatment alcohol consumption, especially in patients who had multiple previous withdrawals. Anticonvulsants were particularly helpful for patients with mood disorders such as depression. In the studies we reviewed, anticonvulsants caused less sedation compared with benzodiazepines, and also decreased the occurrence of relapse.
Dosing recommendations. In the studies included in our review, gabapentin was effective at a dosage of 1,600 mg/d (given as 400 mg 4 times a day). This was tapered as follows: 400 mg 4 times a day on Days 1 to 3, 400 mg 3 times a day on Day 4, 400 mg twice a day on Day 5, and 400 mg once a day on Day 6. Carbamazepine was effective at 600 to 800 mg/d, and was tapered by decreasing by 200 mg as follows: 800 mg/d on Days 1 to 3, 600 mg/d on Day 4, 400 mg on Day 5, and 200 mg/d on Day 6. In the reviewed study, the maximum dose of lamotrigine never exceeded 200 mg/d and was administered for 28 days; the exact dosing and taper plan were not described. The dosing of sodium valproate ranged from 1,200 mg/d to 1600 mg/d for 7 days, followed by decreasing by 200 mg each day. The recommended duration of treatment varied; on average for all anticonvulsants, it was 7 to 12 days, followed by a taper. Carbamazepine was shown to be superior to oxazepam in ameliorating the symptoms of AWS.
Adverse effects. When considering the tolerability, adverse effect profile, duration of action, and effectiveness of the anticonvulsants included in our review, gabapentin appears to be the safest agent to choose. For the other anticonvulsants, the risks might outweigh the benefits. Specifically, in a comparison of sodium valproate and carbamazepine, Hillbom et al21 concluded that in doses >800 mg/d, carbamazepine has potential to cause more adverse effects than benefits. However, Agricola et al16 found that carbamazepine had a preferential action on fear, nightmares, and hallucinations.
Continue to: A few caveats
A few caveats
Our review focused a large collection of data from multiple databases and RCTs only. However, its limitations include:
- there was no measure of heterogeneity
- the studies had short treatment duration
- most studies evaluated predominantly male participants
- some studies were underpowered.
Our review laid a groundwork for future research that includes more well-designed RCTs and/or meta-analyses of recent studies that evaluated the use anticonvulsants for treating AWS.
Bottom Line
Evidence suggests certain anticonvulsants may be an effective alternative to benzodiazepines for the treatment of mild to moderate alcohol withdrawal syndrome. Gabapentin may be the safest anticonvulsant to prescribe. Other anticonvulsants to consider include carbamazepine, sodium valproate, and lamotrigine, but for these agents, the risks might outweigh the benefits.
Related Resources
- Myrick H, Anton RF. Treatment of alcohol withdrawal. Alcohol Health Res World. 1998;22(1):38-43. https://pubs.niaaa.nih.gov/publications/arh22-1/38-43.pdf
- World Health Organization. Management of alcohol withdrawal. Published 2012. https://www.who.int/mental_health/mhgap/evidence/alcohol/q2/en/
Drug Brand Names
Carbamazepine • Tegretol
Gabapentin • Neurontin
Lamotrigine • Lamictal
Levetiracetam • Keppra
Lorazepam • Ativan
Oxcarbazepine • Trileptal
Phenytoin • Dilantin
Sodium valproate • Depakote
Acknowledgments
The authors thank Geetha Manikkara, MD, Madhuri Jakkam Setty, MD, and Elizabeth DeOreo, MD, for their efforts with the systematic review research.
Abrupt cessation or reduction of alcohol consumption may result in alcohol withdrawal syndrome (AWS), which is a medical emergency that can lead to serious complications when unrecognized or treatment is delayed. Symptoms of AWS include tremors, anxiety attacks, cognitive impairment, hallucinations, seizures, delirium tremens (DT), and in severe, untreated cases, death.1 Low to moderate alcohol consumption produces euphoria and excitation via activation of glutamatergic neurotransmission, while higher concentrations produce severe intoxication via GABAergic mechanisms. Acute withdrawal unmasks the hyper-excitatory state of the brain, causing anxiety, agitation, and autonomic activation characteristic of AWS, which typically begins 1 to 3 days after the last drink.2 In the 2012-2013 National Epidemiologic Survey on Alcohol and Related Conditions conducted by the National Institute on Alcohol Abuse and Alcoholism (NIAAA), the 12-month and lifetime prevalences of AWS were 13.9% and 29.1%, respectively.3 Within the general inpatient population, AWS can be present in nearly 30% of patients; if left untreated, AWS has a 15% mortality rate, although when AWS is recognized early and treated, the mortality rate falls dramatically to 2%.4
AWS has most commonly been treated with benzodiazepines.5 However, benzodiazepines have the potential for significant adverse effects when used in older adults and in individuals with complicated medical issues, such as obstructive lung disease and sleep apnea.6 Anticonvulsants have been increasingly used to treat alcohol withdrawal, and their use is supported by several retrospective and prospective studies. In this article, we review the data from randomized control trials (RCTs) on the use of anticonvulsants for the treatment of AWS to see if we can make any recommendations for the use of anticonvulsants for treating AWS.
Our literature search
We searched 5 databases (PubMed, Cochrane, Medline, PsycInfo, and Embase) using the following terms: “alcohol withdrawal syndrome treatment”, “anticonvulsants”, “anti-epileptic”, “gabapentin”, “carbamazepine”, “sodium valproate”, “oxcarbazepine”, “phenytoin”, “levetiracetam”, and “lamotrigine.” We included only double-blind RCTs published between January 1, 1976 and September 30, 2016 in English-language journals or that had an official English translation. There were no restrictions on patient age or location of treatment (inpatient vs outpatient). All RCTs that compared anticonvulsants or a combination of an anticonvulsant and an active pharmacotherapeutic agent with either placebo or gold standard treatment for AWS were included. Database reviews, systematic reviews, and meta-analyses were excluded.
We identified 662 articles that met these criteria. However, most were duplicates, review articles, systematic reviews, meta-analyses, case reports, or open-label or non-randomized trials. Only 16 articles met our inclusion criteria. In the following sections, we discuss these 16 studies by medication type and in chronological order.
Gabapentin
The characteristics of the gabapentin studies included in this review are summarized in Table 1.7-13

Bonnet et al7 (2003) examined 61 adults who met the clinical criteria for alcohol dependence and displayed moderate or severe AWS according to their Mainz Alcohol Withdrawal Score (MAWS ≥4). They were randomized to receive placebo or gabapentin, 400 mg 4 times a day, along with clomethiazole. The attrition rate was not significantly different between the 2 groups (P = .66). The difference in the number of clomethiazole capsules taken during the first 24 hours between the groups was small and not significant (P = .96). Analysis of MAWS over time revealed no significant main effect for group (P = .26) and a significant effect for the time variable (P < .001). The interaction between group and time was not significant (P =.4). This means that there was a significant decrease in MAWS from baseline over 48 hours, and this decrease in MAWS was considered equal for both study groups. Adverse clinical events were observed in both groups, and there was no significant difference (P = .74) between the groups. Nausea and ataxia, which are specific to gabapentin, were observed more frequently in this group.
Conclusion: The authors concluded that gabapentin, 400 mg 4 times a day, is no better than placebo in reducing the amount of clomethiazole required to treat acute AWS.7
Continue to: Bonnet et al
Bonnet et al8 (2007) also conducted a study examining 59 patients with alcohol dependence who displayed moderate or severe AWS. Participants received placebo or gabapentin, 400 mg, and a rescue medication, clomethiazole, if needed. Subsequently, a capsule of study medication was administered every 6 hours for 2 days and then tapered. During the study, mood was measured by Profile of Mood States (POMS), and subjective complaints of withdrawal were measured using the Essen Self-Assessment of Alcohol Withdrawal Scale (ESA). Of the 59 patients, only 46 were analyzed; 5 patients dropped out, and 8 patients were missing data. Compared with the placebo group, the gabapentin group displayed less dejection, fatigue, and anger, and more vigor. Analysis of variance (ANOVA) measures revealed significant overall changes over time on all 4 scales (all P < .001). A significant (F = 3.62, df 2;43, P = .035) group × time interaction resulted exclusively for vigor. Analysis was repeated using rank-transformed data, resulting in a significant (P = .046) interaction effect. The significant increase in vigor was not apparent after tapering off gabapentin, which suggests gabapentin has a reversible effect on vigor. There was a significant (P < .001) overall decline of subjective withdrawal symptoms complaints, but no group × time interaction (P = .62). Analysis of 11 patients with comorbid mild depression revealed no significant time × group interaction for dejection, fatigue, anger, or subjective withdrawal (all P > .05). However, for vigor, the group × time interaction was significant (P = .022). Throughout the treatment, vigor scores of those mild depressive patients who received gabapentin increased to a level comparable to that of patients without a mood disorder.
Conclusion: The authors authors concluded that gabapentin was markedly more efficacious in improving vigor in the small subgroup of patients with mild depression.8
Myrick et al9 (2007) evaluated the safety and tolerability of gabapentin in patients who abused alcohol, as well as the ability of gabapentin to reduce alcohol craving and consumption. This study included 35 participants randomly assigned to receive gabapentin (n = 17) or placebo (n = 18) for 7 days. All medications were administered in standard gel caps with riboflavin, 25 mg, to assess for compliance via a laboratory-based urinary fluorescence assay. Urine samples were assessed for riboflavin at baseline and Day 6, and a reading of 1,500 ng/mL of riboflavin on Day 6 was interpreted as being compliant. Participants were required to abstain completely from drinking alcohol on Day 6 and the morning of Day 7. At the first session, the following measures were completed: demographic form, alcohol and drug section of the Structured Clinical Interview (SCID), Obsessive-Compulsive Drinking Scale, Self-Administered Alcohol Screening Test (SAAST), and Alcohol Dependence Scale (ADS); there also was collection of a urine sample for detection of abused drugs and a blood sample for liver function and general health screening.
At the second session, patients completed the psychiatric sections of the SCID and the Alcohol Craving Questionnaire, and received a physical exam. To assess the negative clinical effects of gabapentin and alcohol on the CNS, the Epworth Sleepiness Scale (ESS) and POMS were administered at baseline and on Day 6. Also, several other scales were used to identify any impact of gabapentin on acute alcohol effects and craving: the Clinical Institute Withdrawal Assessment of Alcohol, Revised (CIWA-Ar), Biphasic Alcohol Effects Scale (BAES), Subjective High Assessment Scale (SHAS), and Alcohol Urge Questionnaire (AUQ).
Conclusion: Gabapentin was well tolerated, but compared with placebo, gabapentin had no effect on alcohol stimulation (P = .75) or sedation (P = .99) as measured by the BAES. The difference in SHAS scores was also not significant (P = .19). There was also no significant reduction in craving for alcohol as measured on the AUQ scale
Continue to: Malcolm et al
Malcolm et al10 conducted an outpatient treatment study. Patients were men and women age 21 to 70 years from multiple ethnic groups. They were randomized to receive gabapentin or lorazepam; 449 patients were screened and 68 completed the follow-up. Scales used included the CIWA-Ar, Beck Depression Inventory (BDI), and ESS.
Patients receiving lorazepam reported less insomnia and more sleepiness early in treatment than patients receiving gabapentin. However, upon completing treatment and discontinuing medication administration, patients previously treated with lorazepam reported increased insomnia and daytime sleepiness, while patients previously treated with gabapentin continued to report improvements in these self-reported sleep measures. The differences between lorazepam and gabapentin were further evidenced in BDI scores at Day 5, Day 7, and Day 12 in patients who had previously experienced multiple withdrawals. Gabapentin was superior to lorazepam in reducing insomnia as assessed by BDI score, an effect that was sustained throughout the post-treatment week. Participants’ ESS scores indicated less daytime sleepiness in the gabapentin group than in the lorazepam group.
Conclusion: Among patients who abused alcohol and had a history of multiple withdrawals, lorazepam is less effective than gabapentin in reducing insomnia.10 However, this study had several limitations: <25% of individuals who were initially screened were enrolled in the study, and it used subjective tests such as BDI. Objective electrophysiologic measures of sleep and daytime sleepiness would have been very helpful.
Myrick et al11 (2009) also compared gabapentin and lorazepam for treating alcohol withdrawal. One hundred patients were randomized to receive 4 days of fixed-dose taper of gabapentin or lorazepam. Patients could receive 1 of 3 gabapentin dosing regimens (600 mg/d, 900 mg/d, or 1,200 mg/d) for 3 days. Participants who were randomized to receive lorazepam were given 6 mg/d for 3 days and then tapered to 4 mg/d. Also, blinded supplemental medications (rescue packs) were given to each patient on Days 1 to 4 to treat subjective feelings of alcohol withdrawal. All patients also received thiamine for 12 days. Assessment of severity of alcohol withdrawal was measured by the CIWA-Ar. To quantify the severity of alcohol dependence and alcohol use, patients were asked to complete the ADS and Time-Line Follow-Back (TLFB) scales, respectively. Other scales administered included the BDI, Zung Anxiety Scale (ZAS), ESS, and visual analogue scales that assessed craving, ability to perform work, and need for additional medication.
There was a decrease in CIWA-Ar scores over time in all groups. High-dose gabapentin was found to be statistically superior but clinically similar to lorazepam (P = .009). Researchers also found that compared with patients who were treated with lorazepam, patients who were treated with gabapentin experienced reduced craving and anxiety/depressive symptoms, and complained of less subjective discomfort. Compared to patients who were treated with gabapentin, patients who were treated with lorazepam had higher probabilities of drinking on the first day of dose decrease (Day 2) and the second day off medication (Day 6) (P = .0002). During post-treatment, patients who were treated with gabapentin had less probability of drinking during the follow-up post-treatment period (P = .2 for 900 mg/d and P = .3 for 1,200 mg/d) compared with patients who were treated with lorazepam (P = .55).
Continue to: Conclusion
Conclusion: The researchers concluded that gabapentin was well tolerated and effectively diminished the symptoms of alcohol withdrawal, especially at the higher target dose (1,200 mg/d), and that compared with lorazepam, gabapentin decreased the probability of drinking during alcohol withdrawal and in the immediate post-withdrawal week.11
Stock et al12 randomized 26 patients who met criteria for AWS to receive gabapentin or chlordiazepoxide. Gabapentin doses were 1,200 mg/d orally for 3 days, followed by 900 mg/d, 600 mg/d, and 300 mg/d for 1 day each. Chlordiazepoxide doses were 100 mg/d orally for 3 days, followed by 75 mg/d, 50 mg/d, and 25 mg/d for 1 day each. The ESS, Penn Alcohol Craving Scale (PACS), ataxia rating, and CIWA-Ar were administered daily. Thirty-five percent of participants dropped out at the end of the 7-day treatment period. Days 1 to 4 were considered the early treatment period, and Days 5 to 7 were considered the late treatment period. The adjusted mean ESS score did not differ significantly between the randomized groups during the early stage (P = .61) vs the late stage, in which the adjusted mean ESS score was significantly lower with gabapentin compared with chlordiazepoxide (P = .04). The differences in PACS scores between the groups were not statistically significant in either stage (early stage P = .59 vs late stage P = .08), but a trend of lower PACS scores was noted with gabapentin in the later stage. No participant in either group had ataxia during the study. In both groups, CIWA-Ar scores were reduced similarly.
Conclusion: The researchers concluded that gabapentin treatment resulted in a significantly greater reduction in sedation (ESS) and a trend toward reduced alcohol craving (PACS) by the end of treatment compared with chlordiazepoxide treatment.12
Schacht et al13 analyzed functional magnetic resonance imaging data from 48 patients who were alcohol-dependent in a 6-week RCT. Patients were randomized to receive gabapentin up to 1,200 mg/d for 39 days plus flumazenil for 2 days (GP/FMZ group) or an oral placebo and placebo infusions on the same time course. Evaluations included the SCID, ADS, and Obsessive-Compulsive Drinking Scale (OCDS). On Day 1, the CIWA-Ar was administered; it was used to ensure equal distribution of individuals with higher alcohol withdrawal symptoms between medication groups. There were no significant effects of initial alcohol withdrawal symptom status or medication. However, there was a significant interaction between these factors: patients with higher alcohol withdrawal symptoms who received GP/FMZ and those with lower alcohol withdrawal symptoms who received placebo demonstrated greater cue-elicited activation, relative to the other groups, and had less subsequent drinking, which reflected differences in deactivation between alcohol and beverage stimuli, in a cluster that encompassed the dorsal ACC (dACC) (family-wise error-corrected cluster probability of P = .012; 99 voxels; local maxima at [-3, 39, 18] and [6, 33, 9]). In the GP/FMZ group, patients with higher alcohol withdrawal symptoms had significantly greater activation, while in the placebo group, patients with lower alcohol withdrawal symptoms had greater activation.
Conclusion: The researchers concluded that alterations in task-related deactivation of dACC, a component of the default mode network, may predict better alcohol treatment response, while activation of DLPFC, an area associated with selective attention, may predict relapse drinking.13
Continue to: Carbamazepine
Carbamazepine
The characteristics of the carbamazepine studies included in this review are summarized in Table 2.14-19

Björkqvist et al14 randomized 105 men with AWS to placebo or carbamazepine. On initial assessment, history, physical examination, relevant labs, and intoxication assessments were recorded. On subsequent visits, nursing staff recorded withdrawal symptoms for patients as 0 to 2 (0 = no specific symptoms, 1 = patient only complained when asked about specific symptoms, 2 = patient complained of withdrawal symptoms without being asked, or if symptoms were severe or obvious to others). Along with the above, vital signs and a visual analogue scale of 0 to 10 (0 = feeling could not be worse, 10 = feeling could not be better) were recorded at each visit. The dose was weight-dependent and administered as follows: on Days 1 and 2, 1+1+2 tablets of carbamazepine, 200 mg, or placebo; Days 3 and 4, 1+1+1 tablets; and Days 5 and 6, 1+0+1 tablets. Every patient received dichloralphenazone as needed. All patients were given vitamin B 3 times a day. Most withdrawal symptoms decreased faster in the carbamazepine group on Day 2 (P = .01) and on Day 4 (P = .1). On the visual analogue scale, scores varied between patients. On Day 1, the mean score was 2.5 times higher in the carbamazepine group compared with the placebo group, and this difference increased to 3 times by Day 7 (P < .01). The patient’s estimated ability to work improved significantly faster in the carbamazepine group than in the placebo group (P < .01).
Conclusion: The authors concluded that compared with placebo, carbamazepine was able to more quickly decrease withdrawal symptoms, especially insomnia and subjective recovery.14
Ritola et al15 randomized 68 hospitalized men with AWS to carbamazepine, 200 mg/d, or clomethiazole, 300 mg/d, for 1 week. The target withdrawal symptoms included gastrointestinal and sleep disturbances; anxiety; aggressiveness; and cardiovascular, depressive, psychotic, and neurologic symptoms. A 4-point rating scale was used for individual symptoms (0 = no symptom, 1 = mild symptom, 2 = moderate symptom, and 3 = severe symptom). On the day of admission (Day 0), all patients were given 50 to 100 mg of chlordiazepoxide IM and 2 tablets and 4 capsules of the trial preparations (either the tablets or capsules were active, and the others were placebos) in the evening. Five patients dropped out of the clomethiazole group and 1 from the carbamazepine group. No significant difference between the 2 treatments were found by the patient, nurse, or physician.
Conclusion: The authors concluded that carbamazepine seemed to be as effective as clomethiazole in the treatment of milder alcohol withdrawal symptoms. Final treatment results were equally good in both groups. Sleep disturbance resolved faster in the carbamazepine group.15
Continue to: Agricola et al
Agricola et al16 compared carbamazepine to tiapride for treatment of acute AWS. In this study, 60 patients were randomized to carbamazepine, 200 mg 3 times a day, or tiapride, 200 mg 3 times a day. All patients were hospitalized with severe AWS preceding DT. The patients were evaluated for withdrawal symptoms (gastrointestinal and cardiovascular symptoms, sleep disturbances, anxiety, aggression, fear, depression, psychotic symptoms, and certain neurologic symptoms). The severity of these symptoms was scored as follows: 0 = no symptoms; 1 = moderate symptoms; and 2 = severe symptoms. At each visit, an overall evaluation of the patient’s clinical condition was made according to a visual analogue scale (100 = worst condition, 0 = best condition). On Day 7, both the doctor and patient evaluated treatment efficacy according to a 4-point scale (1 = no efficacy, 4 = excellent efficacy). There was no significant difference between carbamazepine and tiapride in terms of total symptoms score and visual analogue scale assessment. Carbamazepine was found to have faster relief of symptoms and a significantly greater reduction in symptom score on Day 2 (P < .01). Carbamazepine had a preferential action on fear, nightmares, and hallucinations. The proportion of patients in whom anxiety improved after treatment was 96.2% for carbamazepine and 71.4% for tiapride (P < .05). Aggressiveness and gastrointestinal discomfort resolved faster in the tiapride group. No cases of DT were observed.
Conclusion: The researchers concluded that either carbamazepine or tiapride provides an appropriate alternative in the treatment of inpatients with severe AWS.16
Stuppaeck et al17 compared the efficacy of carbamazepine to oxazepam in 60 inpatients who had symptoms of alcohol withdrawal. Alcohol withdrawal was measured with the CIWA-A, and patients with scores >20 were enrolled in the study. The Clinical Global Impression (CGI) scale and self-rated Adjective Checklist (ACL) were also used. On Days 1 to 3, patients received oxazepam, 120 mg/d, or carbamazepine, 800 mg/d. From Day 4 to 7, doses were decreased to 90 mg/d and 600 mg/d, respectively. After the 7-day trial, all patients were treated with carbamazepine, 200 mg twice a day on Day 8 and 200 mg at night on Day 9. Two patients withdrew consent and 6 dropped out due to adverse effects. During the 7-day trial, when comparing all improvements on CIWA-A, ACL, and CGI scales, carbamazepine was equivalent to oxazepam up to Day 5, and then superior on Days 6 and 7 (P ≤ .05). No decrease in white blood cell count was found in the carbamazepine group.
Conclusion: The authors concluded that carbamazepine is as effective as oxazepam and may be a viable alternative that does not interact with alcohol or cause delirium.17
Malcolm et al18 compared the effects of carbamazepine and lorazepam in patients in an outpatient setting who had single vs multiple previous alcohol withdrawals. The study included 136 patients who satisfied DSM-IV criteria for alcohol dependence and alcohol withdrawal, with a blood alcohol level ≤0.1 g/dL, a Mini-Mental State Examination (MMSE) score ≤26, and a CIWA-Ar score ≤10 on admission. Patients also completed the ADS to quantify the severity of alcohol dependence. Daily drinking was measured by patient report using a daily drinking log and blood alcohol level. Heavy drinking was defined as ≥4 standard drinks per day for women and ≥5 drinks per day for men. On Day 1, patients were randomized to receive carbamazepine, 600 to 800 mg/d,or lorazepam, 6 to 8 mg/d, in divided doses, which was tapered to carbamazepine, 200 mg/d, or lorazepam, 2 mg/d, on Day 5. All patients received thiamine for 12 days. In the immediate post-detoxification period, carbamazepine-treated patients were less likely to relapse, and if they did drink, they drank less than those treated with lorazepam (P = .003). Even in patients who had multiple previous detoxifications, those randomized to carbamazepine drank less than those in lorazepam group (P = .004). Patients in the lorazepam group had significant higher rebound withdrawal symptoms (P = .007).
Continue to: Conclusion
Conclusion: The researchers concluded that carbamazepine and lorazepam were both effective in reducing alcohol withdrawal symptoms. They also concluded that carbamazepine was less likely to cause rebound withdrawal and more likely to reduce post-treatment drinking; among those who did drink, there was less heavy drinking.18
Malcolm et al19 conducted a 5-day double-blind RCT with 136 outpatients who met DSM-IV criteria for alcohol withdrawal. Patients were evaluated by CIWA before getting medications and then daily for 5 days. Patients were randomized to receive carbamazepine, 600 to 800 mg/d on Day 1, 200 mg 3 times a day on Day 2, 200 mg twice a day on Days 3 and 4, and 200 mg once on Day 5. Participants were randomized to receive lorazepam, 6 to 8 mg/d in divided doses on Day 1, 2 mg 3 times a day on Day 2, 2 mg twice a day on Days 3 and 4, and 2 mg once on Day 5. Ability to return to work was self-rated on a 100-mm visual analogue scale, with 0 being “totally unable to return to work’’ and 100 representing “being fully able to return to work.’’ Self-report measures of sleep quality were made using a 100-mm visual analogue scale, with 0 = “the very worst night’s sleep I’ve ever had’’ and 100 = “the very best night’s sleep I’ve ever had.’’ Carbamazepine significantly reduced anxiety (P = .0007). Visual analogue measures of sleep quality indicated a statistically significant main effect of medication on sleep that favored carbamazepine (P = .0186).
Conclusion: The authors concluded that when treating patients with mild to moderate alcohol withdrawal symptoms, carbamazepine was superior to lorazepam in reducing anxiety and improving sleep.19
Sodium valproate
The characteristics of the sodium valproate studies included in this review are summarized in Table 3.20,21

Lambie et al20 evaluated the use of sodium valproate in the treatment of AWS. A total of 49 patients were randomized to a sodium valproate group (n = 22) or a control group (n = 27). All participants were inpatients receiving treatment for alcohol use disorder and substance use disorder for 7 days. Patients in the sodium valproate group received 800 mg every 8 hours for 7 days. Patients were observed daily for occurrence of withdrawal symptoms. Nurses who were blinded to the group assignment graded the degree and severity of symptoms. The trial was initially designed so that chlormethiazole and/or tranquilizers were added to sodium valproate when withdrawal symptoms occurred. However, after treating the first few patients, it became evident that additional medications were not needed. In the treatment group, 13 participants received only sodium valproate, 4 patients needed a tranquilizer, 4 needed chlormethiazole, and 1 needed both. In the control group, 1 received only sodium valproate, 4 received a tranquilizer, 14 received chlormethiazole, and 8 needed both. One patient, who entered the study twice, had a withdrawal seizure when in control group and no seizure on second admission in the sodium valproate group. Physical symptoms disappeared quickly in the sodium valproate group (mean of 2 days vs 2.6 days in the control group). Fourteen patients in the control group received chlormethiazole, compared with only 4 patients in sodium valproate group.
Continue to: Conclusion
Conclusion: The researchers concluded that physical symptoms disappeared quicker in the sodium valproate group than in the control group.20
Hillbom et al21 evaluated the efficacy of sodium valproate vs carbamazepine vs placebo to prevent alcohol withdrawal seizures. A total of 138 participants were studied. Forty-three were assigned to the carbamazepine group, 46 to the sodium valproate group, and 49 to the placebo group. The RCT lasted 4 days. The initial medication doses were 1,200 mg/d. Participants in the carbamazepine group experienced more adverse effects than those in the sodium valproate or placebo groups (P < .001). As a result, approximately one-half of the participants in the carbamazepine group stopped taking the medication. This finding was dependent on the dose of carbamazepine; >800 mg/d resulted in poor tolerance to adverse effects. Seizures occurred among patients in all 3 arms of the study; in the sodium valproate group, 1 participant had a seizure vs 2 participants in the carbamazepine group and 3 in the placebo group. On the other hand, DT occurred only in the sodium valproate and placebo groups.
Conclusion: Researchers concluded that when using sodium valproate or carbamazepine to prevent alcohol withdrawal seizures in an outpatient setting, the adverse effects may outweigh the benefits.21
Lamotrigine
The characteristics of the lamotrigine study included in this review are summarized in Table 3.22
Djokić et al22 evaluated the efficiency of lamotrigine in the treatment of DT. A total of 240 participants who met International Classification of Diseases-10 criteria for DT were randomized to a control group that was treated with anticonvulsants according to an NIAAA protocol (2004), or to an experimental group that was treated with lamotrigine. The CIWA-Ar and the Memorial Delirium Assessment Scale (MDAS) were administered for objective assessment of clinical symptoms, superimposed medical complications, general condition of the patient, adverse effects, and mortality rate. Statistically significant differences between the experimental and control groups were apparent after the third day of therapy, when a drop in the average CIWA-Ar score was observed in the experimental group, while the control group still had high scores (P < .01). After the fifth day of treatment, the differences in scores were more apparent, with the experimental group showing CIWA-Ar scores equal to those of persons with mild/moderate DT, while those in the control group still had high scores. After the tenth day, participants in the experimental group did not have any alcohol withdrawal symptoms, while control group participants were just beginning to get out of life-threatening danger. Death occurred in 4.1% of control group participants and 3.4% of experimental group participants; this difference in mortality rate was not statistically significant.
Continue to: Conclusion
Conclusion: Researchers concluded that lamotrigine is significantly efficacious in the treatment of DT, but does not decrease the mortality rate.22
What to know before you prescribe
AWS is a medical emergency that if left untreated leads to several complications and possibly death. Although benzodiazepines are considered the gold standard for treating AWS, the adverse effects associated with their use advocates for finding alternatives. Anticonvulsants can be an effective alternative for treating AWS. In our literature review, we found 16 double-blind RCTs that used an anticonvulsant medication for the treatment of AWS. Of these, 7 involved gabapentin, 6 involved carbamazepine, 1 involved sodium valproate, 1 involved sodium valproate vs carbamazepine, and 1 involved lamotrigine. Overall, the use of anticonvulsants resulted in significant improvement of mild to moderate symptoms of AWS.
There were more studies of carbamazepine and gabapentin than of other anticonvulsants. All the anticonvulsants offered potential benefits. They decreased the probability of a withdrawal seizure and other complications and effectively reduced alcohol cravings. Anticonvulsants were useful for preventing rebound withdrawal symptoms and reducing post-treatment alcohol consumption, especially in patients who had multiple previous withdrawals. Anticonvulsants were particularly helpful for patients with mood disorders such as depression. In the studies we reviewed, anticonvulsants caused less sedation compared with benzodiazepines, and also decreased the occurrence of relapse.
Dosing recommendations. In the studies included in our review, gabapentin was effective at a dosage of 1,600 mg/d (given as 400 mg 4 times a day). This was tapered as follows: 400 mg 4 times a day on Days 1 to 3, 400 mg 3 times a day on Day 4, 400 mg twice a day on Day 5, and 400 mg once a day on Day 6. Carbamazepine was effective at 600 to 800 mg/d, and was tapered by decreasing by 200 mg as follows: 800 mg/d on Days 1 to 3, 600 mg/d on Day 4, 400 mg on Day 5, and 200 mg/d on Day 6. In the reviewed study, the maximum dose of lamotrigine never exceeded 200 mg/d and was administered for 28 days; the exact dosing and taper plan were not described. The dosing of sodium valproate ranged from 1,200 mg/d to 1600 mg/d for 7 days, followed by decreasing by 200 mg each day. The recommended duration of treatment varied; on average for all anticonvulsants, it was 7 to 12 days, followed by a taper. Carbamazepine was shown to be superior to oxazepam in ameliorating the symptoms of AWS.
Adverse effects. When considering the tolerability, adverse effect profile, duration of action, and effectiveness of the anticonvulsants included in our review, gabapentin appears to be the safest agent to choose. For the other anticonvulsants, the risks might outweigh the benefits. Specifically, in a comparison of sodium valproate and carbamazepine, Hillbom et al21 concluded that in doses >800 mg/d, carbamazepine has potential to cause more adverse effects than benefits. However, Agricola et al16 found that carbamazepine had a preferential action on fear, nightmares, and hallucinations.
Continue to: A few caveats
A few caveats
Our review focused a large collection of data from multiple databases and RCTs only. However, its limitations include:
- there was no measure of heterogeneity
- the studies had short treatment duration
- most studies evaluated predominantly male participants
- some studies were underpowered.
Our review laid a groundwork for future research that includes more well-designed RCTs and/or meta-analyses of recent studies that evaluated the use anticonvulsants for treating AWS.
Bottom Line
Evidence suggests certain anticonvulsants may be an effective alternative to benzodiazepines for the treatment of mild to moderate alcohol withdrawal syndrome. Gabapentin may be the safest anticonvulsant to prescribe. Other anticonvulsants to consider include carbamazepine, sodium valproate, and lamotrigine, but for these agents, the risks might outweigh the benefits.
Related Resources
- Myrick H, Anton RF. Treatment of alcohol withdrawal. Alcohol Health Res World. 1998;22(1):38-43. https://pubs.niaaa.nih.gov/publications/arh22-1/38-43.pdf
- World Health Organization. Management of alcohol withdrawal. Published 2012. https://www.who.int/mental_health/mhgap/evidence/alcohol/q2/en/
Drug Brand Names
Carbamazepine • Tegretol
Gabapentin • Neurontin
Lamotrigine • Lamictal
Levetiracetam • Keppra
Lorazepam • Ativan
Oxcarbazepine • Trileptal
Phenytoin • Dilantin
Sodium valproate • Depakote
Acknowledgments
The authors thank Geetha Manikkara, MD, Madhuri Jakkam Setty, MD, and Elizabeth DeOreo, MD, for their efforts with the systematic review research.
1. Trevisan LA, Boutros N, Petrakis IL, et al. Complications of alcohol withdrawal: pathophysiological insights. Alcohol Health Res World. 1998;22(1):61-66.
2. Borghesani P. Alcohol withdrawal. In: Nordstrom KD, Wilson MP, eds. Quick guide to psychiatric emergencies. Springer: 2018;209-215.
3. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 alcohol use disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766.
4. Ungur LA, Neuner B, John S, et al. Prevention and therapy of alcohol withdrawal on intensive care units: systematic review of controlled trials. Alcohol Clin Exp Res. 2013;37(4):675-686.
5. Sachdeva A, Choudhary M, Chandra M. Alcohol withdrawal syndrome: benzodiazepines and beyond. J Clin Diagn Res. 2015;9(9):VE01-VE07.
6. Ashton H. Toxicity and adverse consequences of benzodiazepine use. Psychiatr Ann. 1995;25:158-165.
7. Bonnet U, Banger M, Leweke FM, et al. Treatment of acute alcohol withdrawal with gabapentin: results from a controlled two-center trial. J Clin Psychopharmacol. 2003;23(5):514-519.
8. Bonnet U, Specka M, Leweke FM, et al. Gabapentin’s acute effect on mood profile--a controlled study on patients with alcohol withdrawal. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):434-438.
9. Myrick H, Anton R, Voronin K, et al. A double-blind evaluation of gabapentin on alcohol effects and drinking in a clinical laboratory paradigm. Alcohol Clin Exp Res. 2007;31(2):221-227.
10. Malcolm R, Myrick L, Veatch L, et al. Self-reported sleep, sleepiness, and repeated alcohol withdrawals: a randomized, double blind, controlled comparison of lorazepam vs gabapentin. J Clin Sleep Med. 2007;3(1):24-32.
11. Myrick H, Malcolm R, Randall PK, et al. A double-blind trial of gabapentin versus lorazepam in the treatment of alcohol withdrawal. Alcohol Clin Exp Res. 2009;33(9):1582-1588.
12. Stock CJ, Carpenter L, Ying J, et al. Gabapentin versus chlordiazepoxide for outpatient alcohol detoxification treatment. Ann Pharmacother. 2013;47(7-8):961-969.
13. Schacht JP, Anton RF, Randall PK, et al. Effects of a GABA-ergic medication combination and initial alcohol withdrawal severity on cue-elicited brain activation among treatment-seeking alcoholics. Psychopharmacol. 2013;227(4):627-637.
14. Björkqvist SE, Isohanni M, Mäkelä R, et al. Ambulant treatment of alcohol withdrawal symptoms with carbamazepine: a formal multicenter double blind comparison with placebo. Acta Psychiatr Scand. 1976;53(5):333-342.
15. Ritola E, Malinen L. A double-blind comparison of carbamazepine and clomethiazole in the treatment of alcohol withdrawal syndrome. Acta Psychiatr Scand. 1981;64(3):254-259.
16. Agricola R, Mazzarino M, Urani R, et al. Treatment of acute alcohol withdrawal syndrome with carbamazepine: a double-blind comparison with tiapride. J Int Med Res. 1982;10(3):160-165.
17. Stuppaeck CH, Pycha R, Miller C, et al. Carbamazepine versus oxazepam in the treatment of alcohol withdrawal: a double-blind study. Alcohol Alcohol. 1992;27(2):153-158.
18. Malcolm R, Myrick H, Roberts J, et al. The effects of carbamazepine and lorazepam on single vs multiple previous withdrawals in an outpatient randomized trial. J Gen Intern Med. 2002;17(5):349-355.
19. Malcolm R, Myrick H, Roberts J, et al. The differential effects of medications on mood, sleep disturbance, and work ability in outpatient alcohol detoxification. Am J Addict. 2002;11(2):141-150.
20. Lambie D, Johnson R, Vijayasenan M, et al. Sodium valproate in the treatment of the alcohol withdrawal syndrome. Aust N Z J. 1980;14(3):213-215.
21. Hillbom M, Tokola R, Kuusela V, et al. Prevention of alcohol withdrawal seizures with carbamazepine and valproic acid. Alcohol. 1989;6(3):223-226.
22. Djokic
1. Trevisan LA, Boutros N, Petrakis IL, et al. Complications of alcohol withdrawal: pathophysiological insights. Alcohol Health Res World. 1998;22(1):61-66.
2. Borghesani P. Alcohol withdrawal. In: Nordstrom KD, Wilson MP, eds. Quick guide to psychiatric emergencies. Springer: 2018;209-215.
3. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 alcohol use disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766.
4. Ungur LA, Neuner B, John S, et al. Prevention and therapy of alcohol withdrawal on intensive care units: systematic review of controlled trials. Alcohol Clin Exp Res. 2013;37(4):675-686.
5. Sachdeva A, Choudhary M, Chandra M. Alcohol withdrawal syndrome: benzodiazepines and beyond. J Clin Diagn Res. 2015;9(9):VE01-VE07.
6. Ashton H. Toxicity and adverse consequences of benzodiazepine use. Psychiatr Ann. 1995;25:158-165.
7. Bonnet U, Banger M, Leweke FM, et al. Treatment of acute alcohol withdrawal with gabapentin: results from a controlled two-center trial. J Clin Psychopharmacol. 2003;23(5):514-519.
8. Bonnet U, Specka M, Leweke FM, et al. Gabapentin’s acute effect on mood profile--a controlled study on patients with alcohol withdrawal. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):434-438.
9. Myrick H, Anton R, Voronin K, et al. A double-blind evaluation of gabapentin on alcohol effects and drinking in a clinical laboratory paradigm. Alcohol Clin Exp Res. 2007;31(2):221-227.
10. Malcolm R, Myrick L, Veatch L, et al. Self-reported sleep, sleepiness, and repeated alcohol withdrawals: a randomized, double blind, controlled comparison of lorazepam vs gabapentin. J Clin Sleep Med. 2007;3(1):24-32.
11. Myrick H, Malcolm R, Randall PK, et al. A double-blind trial of gabapentin versus lorazepam in the treatment of alcohol withdrawal. Alcohol Clin Exp Res. 2009;33(9):1582-1588.
12. Stock CJ, Carpenter L, Ying J, et al. Gabapentin versus chlordiazepoxide for outpatient alcohol detoxification treatment. Ann Pharmacother. 2013;47(7-8):961-969.
13. Schacht JP, Anton RF, Randall PK, et al. Effects of a GABA-ergic medication combination and initial alcohol withdrawal severity on cue-elicited brain activation among treatment-seeking alcoholics. Psychopharmacol. 2013;227(4):627-637.
14. Björkqvist SE, Isohanni M, Mäkelä R, et al. Ambulant treatment of alcohol withdrawal symptoms with carbamazepine: a formal multicenter double blind comparison with placebo. Acta Psychiatr Scand. 1976;53(5):333-342.
15. Ritola E, Malinen L. A double-blind comparison of carbamazepine and clomethiazole in the treatment of alcohol withdrawal syndrome. Acta Psychiatr Scand. 1981;64(3):254-259.
16. Agricola R, Mazzarino M, Urani R, et al. Treatment of acute alcohol withdrawal syndrome with carbamazepine: a double-blind comparison with tiapride. J Int Med Res. 1982;10(3):160-165.
17. Stuppaeck CH, Pycha R, Miller C, et al. Carbamazepine versus oxazepam in the treatment of alcohol withdrawal: a double-blind study. Alcohol Alcohol. 1992;27(2):153-158.
18. Malcolm R, Myrick H, Roberts J, et al. The effects of carbamazepine and lorazepam on single vs multiple previous withdrawals in an outpatient randomized trial. J Gen Intern Med. 2002;17(5):349-355.
19. Malcolm R, Myrick H, Roberts J, et al. The differential effects of medications on mood, sleep disturbance, and work ability in outpatient alcohol detoxification. Am J Addict. 2002;11(2):141-150.
20. Lambie D, Johnson R, Vijayasenan M, et al. Sodium valproate in the treatment of the alcohol withdrawal syndrome. Aust N Z J. 1980;14(3):213-215.
21. Hillbom M, Tokola R, Kuusela V, et al. Prevention of alcohol withdrawal seizures with carbamazepine and valproic acid. Alcohol. 1989;6(3):223-226.
22. Djokic
Asenapine transdermal system for schizophrenia
The asenapine transdermal system is available in 3 patch sizes: 20, 30, and 40 cm2, which deliver 3.8, 5.7, and 7.6 mg/24 hours of asenapine, respectively.3 Based on the average exposure (area under the plasma concentration curve [AUC]) of asenapine, 3.8 mg/24 hours corresponds to 5 mg twice daily of sublingual asenapine, and 7.6 mg/24 hours corresponds to 10 mg twice daily of sublingual asenapine.3 The “in-between” dose strength of 5.7 mg/24 hours would correspond to exposure to a total of 15 mg/d of sublingual asenapine. The recommended starting dose for asenapine transdermal system is 3.8 mg/24 hours. The dosage may be increased to 5.7 mg/24 hours or 7.6 mg/24 hours, as needed, after 1 week. The safety of doses above 7.6 mg/24 hours has not been evaluated in clinical studies. Asenapine transdermal system is applied once daily and should be worn for 24 hours only, with only 1 patch at any time. Application sites include the upper arm, upper back, abdomen, and hip. A different application site of clean, dry, intact skin should be selected each time a new patch is applied. Although showering is permitted, the use of asenapine transdermal system during swimming or taking a bath has not been evaluated. Of note, prolonged application of heat over an asenapine transdermal system increases plasma concentrations of asenapine, and thus application of external heat sources (eg, heating pads) over the patch should be avoided.
How it works
Product labeling notes that asenapine is an atypical antipsychotic, and that its efficacy in schizophrenia could be mediated through a combination of antagonist activity at dopamine D2 and serotonin 5-HT2A receptors.3 The pharmacodynamic profile of asenapine is complex5 and receptor-binding assays performed using cloned human serotonin, norepinephrine, dopamine, histamine, and muscarinic receptors demonstrated picomolar affinity (extremely high) for 5-HT2C and 5-HT2A receptors, subnanomolar affinity (very high) for 5-HT7, 5-HT2B, 5-HT6, and D3 receptors, and nanomolar affinity (high) for D2 receptors, as well as histamine H1, D4, a1-adrenergic, a2-adrenergic, D1, 5-HT5, 5-HT1A, 5-HT1B, and histamine H2 receptors. Activity of asenapine is that of antagonism at these receptors. Asenapine has no appreciable affinity for muscarinic cholinergic receptors.
The asenapine receptor-binding “fingerprint” differs from that of other antipsychotics. Some of these receptor affinities are of special interest in terms of potential efficacy for pro-cognitive effects and amelioration of abnormal mood.5,9 In terms of tolerability, a relative absence of affinity to muscarinic receptors would predict a low risk for anticholinergic adverse effects, but antagonism at histamine H1 and at a1-adrenergic receptors, either alone or in combination, may cause sedation, and blockade of H1 receptors would also predict weight gain.9 Antagonism of a1-adrenergic receptors can be associated with orthostatic hypotension and neurally mediated reflex bradycardia.9
Clinical pharmacokinetics
Three open-label, randomized, phase 1 studies were conducted to assess the relative bioavailability of asenapine transdermal system vs sublingual asenapine.10 These included single- and multiple-dose studies and clinical trials that examined the effects of different application sites and ethnic groups, and the effect of external heat on medication absorption. Studies were conducted in healthy individuals, except for the multiple-dose study, which was performed in adults with schizophrenia. The AUC for asenapine transdermal system was within the range of that of equivalent doses of sublingual asenapine, but peak exposure (maximum concentration) was significantly lower. As already noted, the AUC of the asenapine patch for 3.8 mg/24 hours and 7.6 mg/24 hours corresponds to sublingual asenapine 5 mg and 10 mg twice daily, respectively. Maximum asenapine concentrations are typically reached between 12 and 24 hours, with sustained concentrations during the 24-hour wear time.3 On average, approximately 60% of the available asenapine is released from the transdermal system over 24 hours. Steady-state plasma concentrations for asenapine transdermal system were achieved approximately 72 hours after the first application and, in contrast to sublingual asenapine, the peak-trough fluctuations were small (peak-to-trough ratio is 1.5 for asenapine transdermal system compared with >3 for sublingual asenapine). Dose-proportionality at steady state was evident for asenapine transdermal system. This is in contrast to sublingual asenapine, where exposure increases 1.7-fold with a 2-fold increase in dose.4,5 Following patch removal, the apparent elimination half-life is approximately 30 hours.3 The pharmacokinetics of the patch did not vary with regards to the application site (upper arm, upper back, abdomen, or hip area), and the pharmacokinetic profile was similar across the ethnic groups that participated in the study. Direct exposure to external heat did increase both the rate and extent of absorption, so external heat sources should be avoided.3
Efficacy
The efficacy profile for asenapine transdermal system would be expected to mirror that for sublingual asenapine.6,7 In addition to data supporting the use of asenapine as administered sublingually, a phase 3 study specifically assessed efficacy and safety of asenapine transdermal system in adults with schizophrenia.11,12 This study was conducted in the United States and 4 other countries at a total of 59 study sites, and 616 patients with acutely exacerbated schizophrenia were enrolled. After a 3- to 14-day screening/single-blind run-in washout period, participants entered a 6-week inpatient double-blind period. Randomization was 1:1:1 to asenapine transdermal system 3.8 mg/24 hours, 7.6 mg/24 hours, or a placebo patch. Each of the patch doses demonstrated significant improvement vs placebo at Week 6 for the primary (change in Positive and Negative Syndrome Scale [PANSS] total score) and key secondary (change in Clinical Global Impression-Severity of Illness) endpoints. Response at endpoint, as defined by a ≥30% improvement from baseline PANSS total score, or by a Clinical Global Impression–Improvement score of 1 (very much improved) or 2 (much improved), was also assessed. For either definition of response, both doses of asenapine transdermal system were superior to placebo, with number needed to treat (NNT) (Box) values <10 for the 3.8 mg/24 hours dose (Table 2). These effect sizes are similar to what is known about sublingual asenapine as determined in a meta-analysis performed by the manufacturer and using individual patient data.13
Box
Clinical trials produce a mountain of data that can be difficult to interpret and apply to clinical practice. When reading about studies, you may wonder:
- How large is the effect being measured?
- Is it clinically important?
- Are we reviewing a result that may be statistically significant but irrelevant for day-today patient care?
Number needed to treat (NNT) and number needed to harm (NNH)—two tools of evidence-based medicine—can help answer these questions. NNT helps us gauge effect size or clinical significance. It is different from knowing if a clinical trial result is statistically significant. NNT allows us to place a number on how often we can expect to encounter a difference between two interventions. If we see a therapeutic difference once every 100 patients (NNT of 100), the difference between the treatments is not of great concern under most circumstances. But if a difference in outcome is seen once in every 7 patients being treated with an intervention vs another (NNT of 7), the result will likely influence dayto-day practice.
How to calculate NNT (or NNH):
What is the NNT for an outcome for drug A vs drug B?
fA = frequency of outcome for drug A
fB = frequency of outcome for drug B
NNT = 1/[ fA - fB]
By convention, we round up the NNT to the next higher whole number.
For example, let’s say drugs A and B are used to treat depression, and they result in 6-week response rates of 55% and 75%, respectively. The NNT to encounter a difference between drug B and drug A in terms of responders at 6 weeks can be calculated as follows:
- Difference in response rates: .75 -.55 = .20
- NNT: 1/.20 = 5
A rule of thumb: NNT values for a medication vs placebo <10 usually denote a medication we use on a regular basis to treat patients.
a Adapted from Citrome L. Dissecting clinical trials with ‘number needed to treat.’ Current Psychiatry. 2007;6(3):66-71. Citrome L. Can you interpret confidence intervals? It’s not that difficult. Current Psychiatry. 2007;6(8):77-82. Additional information can be found in Citrome L, Ketter TA. When does a difference make a difference? Interpretation of number needed to treat, number needed to harm, and likelihood to be helped or harmed. Int J Clin Pract. 2013;67(5):407-411 (free to access at onlinelibrary.wiley.com/doi/full/10.1111/ijcp.12142)
Overall tolerability and safety
The systemic safety and tolerability profile for asenapine transdermal system would be expected to be similar to that for sublingual asenapine, unless there are adverse events that are related to high peak plasma concentrations or large differences between peak and trough plasma concentrations.6 Nonsystemic local application site adverse events would, of course, differ between sublingual vs transdermal administration.
Continue to: Use of asenapine transdermal system...
Use of asenapine transdermal system avoids the dysgeusia and oral hypoesthesia that can be observed with sublingual asenapine4,6; however, dermal effects need to be considered (see Dermal safety). The most commonly observed adverse reactions (incidence ≥5% and at least twice that for placebo) for asenapine transdermal system are extrapyramidal disorder, application site reaction, and weight gain.3 For sublingual asenapine for adults with schizophrenia, the list includes akathisia, oral hypoesthesia, and somnolence.4 These adverse events can be further described using the metric of number needed to harm (NNH) as shown in Table 3.3,4,11,12,14 Of note, extrapyramidal disorder and weight gain appear to be dose-related for asenapine transdermal system. Akathisia appears to be dose-related for sublingual asenapine but not for asenapine transdermal system. Somnolence appears to be associated with sublingual asenapine but not necessarily with asenapine transdermal system.
For sublingual asenapine, the additional indications (bipolar I disorder as acute monotherapy treatment of manic or mixed episodes in adults and pediatric patients age 10 to 17, adjunctive treatment to lithium or valproate in adults, and maintenance monotherapy treatment in adults) have varying commonly encountered adverse reactions.4 Both transdermal asenapine system and sublingual asenapine are contraindicated in patients with severe hepatic impairment (Child-Pugh C) and those with known hypersensitivity to asenapine or to any components in the formulation. Both formulations carry similar warnings in their prescribing information regarding increased mortality in older patients with dementia-related psychosis, cerebrovascular adverse reactions in older patients with dementia-related psychosis, neuroleptic malignant syndrome, tardive dyskinesia, metabolic changes, orthostatic hypotension, leukopenia (and neutropenia and agranulocytosis), QT prolongation, seizures, and potential for cognitive and motor impairment.
Adverse events leading to discontinuation of study treatment in the asenapine transdermal system pivotal trial occurred in 4.9%, 7.8%, and 6.8% of participants in the 3.8 mg/24 hour, 7.6 mg/24 hour, and placebo groups, respectively.11
Dermal safety
In the pivotal efficacy study,11 the incidence of adverse events at patch application sites was higher in the active groups vs placebo (Table 33,4,11,12,14). The most frequently reported patch application site reactions were erythema and pruritus, occurring in approximately 10% and 4% in the active treatment arms vs 1.5% and 1.9% for placebo, respectively. With the exception of 1 adverse event of severe application site erythema during Week 2 (participant received 7.6 mg/24 hour, erythema resolved without intervention, and the patient continued the study), all other patch application site events were mild or moderate in severity. Rates of discontinuation due to application site reactions or skin disorders were ≤0.5% across all groups. In the pharmacokinetic studies,10 no patches were removed because of unacceptable irritation.
Why Rx?
Asenapine transdermal system is the first antipsychotic “patch” FDA-approved for the treatment of adults with schizophrenia. Asenapine has been available since 2009 as a sublingual formulation administered twice daily. The pharmacokinetic profile of the once-daily transdermal system demonstrates dose-proportional kinetics and sustained delivery of asenapine with a low peak-to-trough plasma level ratio. Three dosage strengths (3.8, 5.7, and 7.6 mg/24 hours) are available, corresponding to blood levels attained with sublingual asenapine exposures of 10, 15, and 20 mg/d, respectively. Application sites are rotated daily and include the upper arms, upper back, abdomen, or hip. Dysgeusia and hypoesthesia of the tongue are avoided with the use of the patch, and there are no food or drink restrictions. Attention will be needed in case of dermal reactions, similar to that observed with other medication patches.
Bottom Line
The asenapine transdermal drug delivery system appears to be efficacious and reasonably well tolerated. The treatment of schizophrenia is complex and requires individualized choices in order to optimize outcomes. A patch may be the preferred formulation for selected patients, and caregivers will have the ability to visually check if the medication is being used.
Related Resource
- Hisamitsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
Drug Brand Names
Asenapine sublingual • Saphris
Asenapine transdermal system • Secuado
Lithium • Eskalith, Lithobid
Valproate • Depakote
1. Noven. US FDA approves SECUADO® (asenapine) transdermal system, the first-and-only transdermal patch for the treatment of adults with schizophrenia. October 15, 2019. Accessed January 15, 2021. https://www.noven.com/wp-content/uploads/2020/04/PR101519.pdf
2. US Food and Drug Administration. Center for Drug Evaluation and Research. Approval Package for: APPLICATION NUMBER: 212268Orig1s000. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000Approv.pdf
3. Hisam itsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
4. Allergan USA, Inc. SAPHRIS® (asenapine) sublingual tablets prescribing information. February 2017. Accessed January 15, 2021. https://media.allergan.com/actavis/actavis/media/allergan-pdf-documents/product-prescribing/Final_labeling_text_SAPHRIS-clean-02-2017.pdf
5. Citrome L. Asenapine review, part I: chemistry, receptor affinity profile, pharmacokinetics and metabolism. Expert Opin Drug Metab Toxicol. 2014;10(6):893-903.
6. Citrome L. Asenapine review, part II: clinical efficacy, safety and tolerability. Expert Opin Drug Saf. 2014;13(6):803-830.
7. Citrome L. Chapter 31: Asenapine. In: Schatzberg AF, Nemeroff CB, eds. The American Psychiatric Association Publishing Textbook of Psychopharmacology, 5th ed. American Psychiatric Association Publishing; 2017:797-808.
8. Citrome L, Zeni CM, Correll CU. Patches: established and emerging transdermal treatments in psychiatry. J Clin Psychiatry. 2019;80(4):18nr12554. doi: 10.4088/JCP.18nr12554
9. Shayegan DK, Stahl SM. Atypical antipsychotics: matching receptor profile to individual patient’s clinical profile. CNS Spectr. 2004;9(10 suppl 11):6-14.
10. Castelli M, Suzuki K, Komaroff M, et al. Pharmacokinetic profile of asenapine transdermal system HP-3070: The first antipsychotic patch in the US. Poster presented virtually at the American Society for Clinical Psychopharmacology (ASCP) 2020 Annual Meeting, May 29-30, 2020. https://www.psychiatrist.com/ascpcorner/Documents/ascp2020/3_ASCP%20Poster%20Abstracts%202020-JCP.pdf
11. Citrome L, Walling DP, Zeni CM, et al. Efficacy and safety of HP-3070, an asenapine transdermal system, in patients with schizophrenia: a phase 3, randomized, placebo-controlled study. J Clin Psychiatry. 2020;82(1):20m13602. doi: 10.4088/JCP.20m13602
12. US Food and Drug Administration. Drug Approval Package: SECAUDO. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000TOC.cfm
13. Szegedi A, Verweij P, van Duijnhoven W, et al. Meta-analyses of the efficacy of asenapine for acute schizophrenia: comparisons with placebo and other antipsychotics. J Clin Psychiatry. 2012;73(12):1533-1540.
14. Citrome L. Asenapine for schizophrenia and bipolar disorder: a review of the efficacy and safety profile for this newly approved sublingually absorbed second-generation antipsychotic. Int J Clin Pract. 2009;63(12):1762-1784.
The asenapine transdermal system is available in 3 patch sizes: 20, 30, and 40 cm2, which deliver 3.8, 5.7, and 7.6 mg/24 hours of asenapine, respectively.3 Based on the average exposure (area under the plasma concentration curve [AUC]) of asenapine, 3.8 mg/24 hours corresponds to 5 mg twice daily of sublingual asenapine, and 7.6 mg/24 hours corresponds to 10 mg twice daily of sublingual asenapine.3 The “in-between” dose strength of 5.7 mg/24 hours would correspond to exposure to a total of 15 mg/d of sublingual asenapine. The recommended starting dose for asenapine transdermal system is 3.8 mg/24 hours. The dosage may be increased to 5.7 mg/24 hours or 7.6 mg/24 hours, as needed, after 1 week. The safety of doses above 7.6 mg/24 hours has not been evaluated in clinical studies. Asenapine transdermal system is applied once daily and should be worn for 24 hours only, with only 1 patch at any time. Application sites include the upper arm, upper back, abdomen, and hip. A different application site of clean, dry, intact skin should be selected each time a new patch is applied. Although showering is permitted, the use of asenapine transdermal system during swimming or taking a bath has not been evaluated. Of note, prolonged application of heat over an asenapine transdermal system increases plasma concentrations of asenapine, and thus application of external heat sources (eg, heating pads) over the patch should be avoided.
How it works
Product labeling notes that asenapine is an atypical antipsychotic, and that its efficacy in schizophrenia could be mediated through a combination of antagonist activity at dopamine D2 and serotonin 5-HT2A receptors.3 The pharmacodynamic profile of asenapine is complex5 and receptor-binding assays performed using cloned human serotonin, norepinephrine, dopamine, histamine, and muscarinic receptors demonstrated picomolar affinity (extremely high) for 5-HT2C and 5-HT2A receptors, subnanomolar affinity (very high) for 5-HT7, 5-HT2B, 5-HT6, and D3 receptors, and nanomolar affinity (high) for D2 receptors, as well as histamine H1, D4, a1-adrenergic, a2-adrenergic, D1, 5-HT5, 5-HT1A, 5-HT1B, and histamine H2 receptors. Activity of asenapine is that of antagonism at these receptors. Asenapine has no appreciable affinity for muscarinic cholinergic receptors.
The asenapine receptor-binding “fingerprint” differs from that of other antipsychotics. Some of these receptor affinities are of special interest in terms of potential efficacy for pro-cognitive effects and amelioration of abnormal mood.5,9 In terms of tolerability, a relative absence of affinity to muscarinic receptors would predict a low risk for anticholinergic adverse effects, but antagonism at histamine H1 and at a1-adrenergic receptors, either alone or in combination, may cause sedation, and blockade of H1 receptors would also predict weight gain.9 Antagonism of a1-adrenergic receptors can be associated with orthostatic hypotension and neurally mediated reflex bradycardia.9
Clinical pharmacokinetics
Three open-label, randomized, phase 1 studies were conducted to assess the relative bioavailability of asenapine transdermal system vs sublingual asenapine.10 These included single- and multiple-dose studies and clinical trials that examined the effects of different application sites and ethnic groups, and the effect of external heat on medication absorption. Studies were conducted in healthy individuals, except for the multiple-dose study, which was performed in adults with schizophrenia. The AUC for asenapine transdermal system was within the range of that of equivalent doses of sublingual asenapine, but peak exposure (maximum concentration) was significantly lower. As already noted, the AUC of the asenapine patch for 3.8 mg/24 hours and 7.6 mg/24 hours corresponds to sublingual asenapine 5 mg and 10 mg twice daily, respectively. Maximum asenapine concentrations are typically reached between 12 and 24 hours, with sustained concentrations during the 24-hour wear time.3 On average, approximately 60% of the available asenapine is released from the transdermal system over 24 hours. Steady-state plasma concentrations for asenapine transdermal system were achieved approximately 72 hours after the first application and, in contrast to sublingual asenapine, the peak-trough fluctuations were small (peak-to-trough ratio is 1.5 for asenapine transdermal system compared with >3 for sublingual asenapine). Dose-proportionality at steady state was evident for asenapine transdermal system. This is in contrast to sublingual asenapine, where exposure increases 1.7-fold with a 2-fold increase in dose.4,5 Following patch removal, the apparent elimination half-life is approximately 30 hours.3 The pharmacokinetics of the patch did not vary with regards to the application site (upper arm, upper back, abdomen, or hip area), and the pharmacokinetic profile was similar across the ethnic groups that participated in the study. Direct exposure to external heat did increase both the rate and extent of absorption, so external heat sources should be avoided.3
Efficacy
The efficacy profile for asenapine transdermal system would be expected to mirror that for sublingual asenapine.6,7 In addition to data supporting the use of asenapine as administered sublingually, a phase 3 study specifically assessed efficacy and safety of asenapine transdermal system in adults with schizophrenia.11,12 This study was conducted in the United States and 4 other countries at a total of 59 study sites, and 616 patients with acutely exacerbated schizophrenia were enrolled. After a 3- to 14-day screening/single-blind run-in washout period, participants entered a 6-week inpatient double-blind period. Randomization was 1:1:1 to asenapine transdermal system 3.8 mg/24 hours, 7.6 mg/24 hours, or a placebo patch. Each of the patch doses demonstrated significant improvement vs placebo at Week 6 for the primary (change in Positive and Negative Syndrome Scale [PANSS] total score) and key secondary (change in Clinical Global Impression-Severity of Illness) endpoints. Response at endpoint, as defined by a ≥30% improvement from baseline PANSS total score, or by a Clinical Global Impression–Improvement score of 1 (very much improved) or 2 (much improved), was also assessed. For either definition of response, both doses of asenapine transdermal system were superior to placebo, with number needed to treat (NNT) (Box) values <10 for the 3.8 mg/24 hours dose (Table 2). These effect sizes are similar to what is known about sublingual asenapine as determined in a meta-analysis performed by the manufacturer and using individual patient data.13
Box
Clinical trials produce a mountain of data that can be difficult to interpret and apply to clinical practice. When reading about studies, you may wonder:
- How large is the effect being measured?
- Is it clinically important?
- Are we reviewing a result that may be statistically significant but irrelevant for day-today patient care?
Number needed to treat (NNT) and number needed to harm (NNH)—two tools of evidence-based medicine—can help answer these questions. NNT helps us gauge effect size or clinical significance. It is different from knowing if a clinical trial result is statistically significant. NNT allows us to place a number on how often we can expect to encounter a difference between two interventions. If we see a therapeutic difference once every 100 patients (NNT of 100), the difference between the treatments is not of great concern under most circumstances. But if a difference in outcome is seen once in every 7 patients being treated with an intervention vs another (NNT of 7), the result will likely influence dayto-day practice.
How to calculate NNT (or NNH):
What is the NNT for an outcome for drug A vs drug B?
fA = frequency of outcome for drug A
fB = frequency of outcome for drug B
NNT = 1/[ fA - fB]
By convention, we round up the NNT to the next higher whole number.
For example, let’s say drugs A and B are used to treat depression, and they result in 6-week response rates of 55% and 75%, respectively. The NNT to encounter a difference between drug B and drug A in terms of responders at 6 weeks can be calculated as follows:
- Difference in response rates: .75 -.55 = .20
- NNT: 1/.20 = 5
A rule of thumb: NNT values for a medication vs placebo <10 usually denote a medication we use on a regular basis to treat patients.
a Adapted from Citrome L. Dissecting clinical trials with ‘number needed to treat.’ Current Psychiatry. 2007;6(3):66-71. Citrome L. Can you interpret confidence intervals? It’s not that difficult. Current Psychiatry. 2007;6(8):77-82. Additional information can be found in Citrome L, Ketter TA. When does a difference make a difference? Interpretation of number needed to treat, number needed to harm, and likelihood to be helped or harmed. Int J Clin Pract. 2013;67(5):407-411 (free to access at onlinelibrary.wiley.com/doi/full/10.1111/ijcp.12142)
Overall tolerability and safety
The systemic safety and tolerability profile for asenapine transdermal system would be expected to be similar to that for sublingual asenapine, unless there are adverse events that are related to high peak plasma concentrations or large differences between peak and trough plasma concentrations.6 Nonsystemic local application site adverse events would, of course, differ between sublingual vs transdermal administration.

Continue to: Use of asenapine transdermal system...
Use of asenapine transdermal system avoids the dysgeusia and oral hypoesthesia that can be observed with sublingual asenapine4,6; however, dermal effects need to be considered (see Dermal safety). The most commonly observed adverse reactions (incidence ≥5% and at least twice that for placebo) for asenapine transdermal system are extrapyramidal disorder, application site reaction, and weight gain.3 For sublingual asenapine for adults with schizophrenia, the list includes akathisia, oral hypoesthesia, and somnolence.4 These adverse events can be further described using the metric of number needed to harm (NNH) as shown in Table 3.3,4,11,12,14 Of note, extrapyramidal disorder and weight gain appear to be dose-related for asenapine transdermal system. Akathisia appears to be dose-related for sublingual asenapine but not for asenapine transdermal system. Somnolence appears to be associated with sublingual asenapine but not necessarily with asenapine transdermal system.

For sublingual asenapine, the additional indications (bipolar I disorder as acute monotherapy treatment of manic or mixed episodes in adults and pediatric patients age 10 to 17, adjunctive treatment to lithium or valproate in adults, and maintenance monotherapy treatment in adults) have varying commonly encountered adverse reactions.4 Both transdermal asenapine system and sublingual asenapine are contraindicated in patients with severe hepatic impairment (Child-Pugh C) and those with known hypersensitivity to asenapine or to any components in the formulation. Both formulations carry similar warnings in their prescribing information regarding increased mortality in older patients with dementia-related psychosis, cerebrovascular adverse reactions in older patients with dementia-related psychosis, neuroleptic malignant syndrome, tardive dyskinesia, metabolic changes, orthostatic hypotension, leukopenia (and neutropenia and agranulocytosis), QT prolongation, seizures, and potential for cognitive and motor impairment.
Adverse events leading to discontinuation of study treatment in the asenapine transdermal system pivotal trial occurred in 4.9%, 7.8%, and 6.8% of participants in the 3.8 mg/24 hour, 7.6 mg/24 hour, and placebo groups, respectively.11
Dermal safety
In the pivotal efficacy study,11 the incidence of adverse events at patch application sites was higher in the active groups vs placebo (Table 33,4,11,12,14). The most frequently reported patch application site reactions were erythema and pruritus, occurring in approximately 10% and 4% in the active treatment arms vs 1.5% and 1.9% for placebo, respectively. With the exception of 1 adverse event of severe application site erythema during Week 2 (participant received 7.6 mg/24 hour, erythema resolved without intervention, and the patient continued the study), all other patch application site events were mild or moderate in severity. Rates of discontinuation due to application site reactions or skin disorders were ≤0.5% across all groups. In the pharmacokinetic studies,10 no patches were removed because of unacceptable irritation.
Why Rx?
Asenapine transdermal system is the first antipsychotic “patch” FDA-approved for the treatment of adults with schizophrenia. Asenapine has been available since 2009 as a sublingual formulation administered twice daily. The pharmacokinetic profile of the once-daily transdermal system demonstrates dose-proportional kinetics and sustained delivery of asenapine with a low peak-to-trough plasma level ratio. Three dosage strengths (3.8, 5.7, and 7.6 mg/24 hours) are available, corresponding to blood levels attained with sublingual asenapine exposures of 10, 15, and 20 mg/d, respectively. Application sites are rotated daily and include the upper arms, upper back, abdomen, or hip. Dysgeusia and hypoesthesia of the tongue are avoided with the use of the patch, and there are no food or drink restrictions. Attention will be needed in case of dermal reactions, similar to that observed with other medication patches.
Bottom Line
The asenapine transdermal drug delivery system appears to be efficacious and reasonably well tolerated. The treatment of schizophrenia is complex and requires individualized choices in order to optimize outcomes. A patch may be the preferred formulation for selected patients, and caregivers will have the ability to visually check if the medication is being used.
Related Resource
- Hisamitsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
Drug Brand Names
Asenapine sublingual • Saphris
Asenapine transdermal system • Secuado
Lithium • Eskalith, Lithobid
Valproate • Depakote
The asenapine transdermal system is available in 3 patch sizes: 20, 30, and 40 cm2, which deliver 3.8, 5.7, and 7.6 mg/24 hours of asenapine, respectively.3 Based on the average exposure (area under the plasma concentration curve [AUC]) of asenapine, 3.8 mg/24 hours corresponds to 5 mg twice daily of sublingual asenapine, and 7.6 mg/24 hours corresponds to 10 mg twice daily of sublingual asenapine.3 The “in-between” dose strength of 5.7 mg/24 hours would correspond to exposure to a total of 15 mg/d of sublingual asenapine. The recommended starting dose for asenapine transdermal system is 3.8 mg/24 hours. The dosage may be increased to 5.7 mg/24 hours or 7.6 mg/24 hours, as needed, after 1 week. The safety of doses above 7.6 mg/24 hours has not been evaluated in clinical studies. Asenapine transdermal system is applied once daily and should be worn for 24 hours only, with only 1 patch at any time. Application sites include the upper arm, upper back, abdomen, and hip. A different application site of clean, dry, intact skin should be selected each time a new patch is applied. Although showering is permitted, the use of asenapine transdermal system during swimming or taking a bath has not been evaluated. Of note, prolonged application of heat over an asenapine transdermal system increases plasma concentrations of asenapine, and thus application of external heat sources (eg, heating pads) over the patch should be avoided.
How it works
Product labeling notes that asenapine is an atypical antipsychotic, and that its efficacy in schizophrenia could be mediated through a combination of antagonist activity at dopamine D2 and serotonin 5-HT2A receptors.3 The pharmacodynamic profile of asenapine is complex5 and receptor-binding assays performed using cloned human serotonin, norepinephrine, dopamine, histamine, and muscarinic receptors demonstrated picomolar affinity (extremely high) for 5-HT2C and 5-HT2A receptors, subnanomolar affinity (very high) for 5-HT7, 5-HT2B, 5-HT6, and D3 receptors, and nanomolar affinity (high) for D2 receptors, as well as histamine H1, D4, a1-adrenergic, a2-adrenergic, D1, 5-HT5, 5-HT1A, 5-HT1B, and histamine H2 receptors. Activity of asenapine is that of antagonism at these receptors. Asenapine has no appreciable affinity for muscarinic cholinergic receptors.
The asenapine receptor-binding “fingerprint” differs from that of other antipsychotics. Some of these receptor affinities are of special interest in terms of potential efficacy for pro-cognitive effects and amelioration of abnormal mood.5,9 In terms of tolerability, a relative absence of affinity to muscarinic receptors would predict a low risk for anticholinergic adverse effects, but antagonism at histamine H1 and at a1-adrenergic receptors, either alone or in combination, may cause sedation, and blockade of H1 receptors would also predict weight gain.9 Antagonism of a1-adrenergic receptors can be associated with orthostatic hypotension and neurally mediated reflex bradycardia.9
Clinical pharmacokinetics
Three open-label, randomized, phase 1 studies were conducted to assess the relative bioavailability of asenapine transdermal system vs sublingual asenapine.10 These included single- and multiple-dose studies and clinical trials that examined the effects of different application sites and ethnic groups, and the effect of external heat on medication absorption. Studies were conducted in healthy individuals, except for the multiple-dose study, which was performed in adults with schizophrenia. The AUC for asenapine transdermal system was within the range of that of equivalent doses of sublingual asenapine, but peak exposure (maximum concentration) was significantly lower. As already noted, the AUC of the asenapine patch for 3.8 mg/24 hours and 7.6 mg/24 hours corresponds to sublingual asenapine 5 mg and 10 mg twice daily, respectively. Maximum asenapine concentrations are typically reached between 12 and 24 hours, with sustained concentrations during the 24-hour wear time.3 On average, approximately 60% of the available asenapine is released from the transdermal system over 24 hours. Steady-state plasma concentrations for asenapine transdermal system were achieved approximately 72 hours after the first application and, in contrast to sublingual asenapine, the peak-trough fluctuations were small (peak-to-trough ratio is 1.5 for asenapine transdermal system compared with >3 for sublingual asenapine). Dose-proportionality at steady state was evident for asenapine transdermal system. This is in contrast to sublingual asenapine, where exposure increases 1.7-fold with a 2-fold increase in dose.4,5 Following patch removal, the apparent elimination half-life is approximately 30 hours.3 The pharmacokinetics of the patch did not vary with regards to the application site (upper arm, upper back, abdomen, or hip area), and the pharmacokinetic profile was similar across the ethnic groups that participated in the study. Direct exposure to external heat did increase both the rate and extent of absorption, so external heat sources should be avoided.3
Efficacy
The efficacy profile for asenapine transdermal system would be expected to mirror that for sublingual asenapine.6,7 In addition to data supporting the use of asenapine as administered sublingually, a phase 3 study specifically assessed efficacy and safety of asenapine transdermal system in adults with schizophrenia.11,12 This study was conducted in the United States and 4 other countries at a total of 59 study sites, and 616 patients with acutely exacerbated schizophrenia were enrolled. After a 3- to 14-day screening/single-blind run-in washout period, participants entered a 6-week inpatient double-blind period. Randomization was 1:1:1 to asenapine transdermal system 3.8 mg/24 hours, 7.6 mg/24 hours, or a placebo patch. Each of the patch doses demonstrated significant improvement vs placebo at Week 6 for the primary (change in Positive and Negative Syndrome Scale [PANSS] total score) and key secondary (change in Clinical Global Impression-Severity of Illness) endpoints. Response at endpoint, as defined by a ≥30% improvement from baseline PANSS total score, or by a Clinical Global Impression–Improvement score of 1 (very much improved) or 2 (much improved), was also assessed. For either definition of response, both doses of asenapine transdermal system were superior to placebo, with number needed to treat (NNT) (Box) values <10 for the 3.8 mg/24 hours dose (Table 2). These effect sizes are similar to what is known about sublingual asenapine as determined in a meta-analysis performed by the manufacturer and using individual patient data.13
Box
Clinical trials produce a mountain of data that can be difficult to interpret and apply to clinical practice. When reading about studies, you may wonder:
- How large is the effect being measured?
- Is it clinically important?
- Are we reviewing a result that may be statistically significant but irrelevant for day-today patient care?
Number needed to treat (NNT) and number needed to harm (NNH)—two tools of evidence-based medicine—can help answer these questions. NNT helps us gauge effect size or clinical significance. It is different from knowing if a clinical trial result is statistically significant. NNT allows us to place a number on how often we can expect to encounter a difference between two interventions. If we see a therapeutic difference once every 100 patients (NNT of 100), the difference between the treatments is not of great concern under most circumstances. But if a difference in outcome is seen once in every 7 patients being treated with an intervention vs another (NNT of 7), the result will likely influence dayto-day practice.
How to calculate NNT (or NNH):
What is the NNT for an outcome for drug A vs drug B?
fA = frequency of outcome for drug A
fB = frequency of outcome for drug B
NNT = 1/[ fA - fB]
By convention, we round up the NNT to the next higher whole number.
For example, let’s say drugs A and B are used to treat depression, and they result in 6-week response rates of 55% and 75%, respectively. The NNT to encounter a difference between drug B and drug A in terms of responders at 6 weeks can be calculated as follows:
- Difference in response rates: .75 -.55 = .20
- NNT: 1/.20 = 5
A rule of thumb: NNT values for a medication vs placebo <10 usually denote a medication we use on a regular basis to treat patients.
a Adapted from Citrome L. Dissecting clinical trials with ‘number needed to treat.’ Current Psychiatry. 2007;6(3):66-71. Citrome L. Can you interpret confidence intervals? It’s not that difficult. Current Psychiatry. 2007;6(8):77-82. Additional information can be found in Citrome L, Ketter TA. When does a difference make a difference? Interpretation of number needed to treat, number needed to harm, and likelihood to be helped or harmed. Int J Clin Pract. 2013;67(5):407-411 (free to access at onlinelibrary.wiley.com/doi/full/10.1111/ijcp.12142)
Overall tolerability and safety
The systemic safety and tolerability profile for asenapine transdermal system would be expected to be similar to that for sublingual asenapine, unless there are adverse events that are related to high peak plasma concentrations or large differences between peak and trough plasma concentrations.6 Nonsystemic local application site adverse events would, of course, differ between sublingual vs transdermal administration.

Continue to: Use of asenapine transdermal system...
Use of asenapine transdermal system avoids the dysgeusia and oral hypoesthesia that can be observed with sublingual asenapine4,6; however, dermal effects need to be considered (see Dermal safety). The most commonly observed adverse reactions (incidence ≥5% and at least twice that for placebo) for asenapine transdermal system are extrapyramidal disorder, application site reaction, and weight gain.3 For sublingual asenapine for adults with schizophrenia, the list includes akathisia, oral hypoesthesia, and somnolence.4 These adverse events can be further described using the metric of number needed to harm (NNH) as shown in Table 3.3,4,11,12,14 Of note, extrapyramidal disorder and weight gain appear to be dose-related for asenapine transdermal system. Akathisia appears to be dose-related for sublingual asenapine but not for asenapine transdermal system. Somnolence appears to be associated with sublingual asenapine but not necessarily with asenapine transdermal system.

For sublingual asenapine, the additional indications (bipolar I disorder as acute monotherapy treatment of manic or mixed episodes in adults and pediatric patients age 10 to 17, adjunctive treatment to lithium or valproate in adults, and maintenance monotherapy treatment in adults) have varying commonly encountered adverse reactions.4 Both transdermal asenapine system and sublingual asenapine are contraindicated in patients with severe hepatic impairment (Child-Pugh C) and those with known hypersensitivity to asenapine or to any components in the formulation. Both formulations carry similar warnings in their prescribing information regarding increased mortality in older patients with dementia-related psychosis, cerebrovascular adverse reactions in older patients with dementia-related psychosis, neuroleptic malignant syndrome, tardive dyskinesia, metabolic changes, orthostatic hypotension, leukopenia (and neutropenia and agranulocytosis), QT prolongation, seizures, and potential for cognitive and motor impairment.
Adverse events leading to discontinuation of study treatment in the asenapine transdermal system pivotal trial occurred in 4.9%, 7.8%, and 6.8% of participants in the 3.8 mg/24 hour, 7.6 mg/24 hour, and placebo groups, respectively.11
Dermal safety
In the pivotal efficacy study,11 the incidence of adverse events at patch application sites was higher in the active groups vs placebo (Table 33,4,11,12,14). The most frequently reported patch application site reactions were erythema and pruritus, occurring in approximately 10% and 4% in the active treatment arms vs 1.5% and 1.9% for placebo, respectively. With the exception of 1 adverse event of severe application site erythema during Week 2 (participant received 7.6 mg/24 hour, erythema resolved without intervention, and the patient continued the study), all other patch application site events were mild or moderate in severity. Rates of discontinuation due to application site reactions or skin disorders were ≤0.5% across all groups. In the pharmacokinetic studies,10 no patches were removed because of unacceptable irritation.
Why Rx?
Asenapine transdermal system is the first antipsychotic “patch” FDA-approved for the treatment of adults with schizophrenia. Asenapine has been available since 2009 as a sublingual formulation administered twice daily. The pharmacokinetic profile of the once-daily transdermal system demonstrates dose-proportional kinetics and sustained delivery of asenapine with a low peak-to-trough plasma level ratio. Three dosage strengths (3.8, 5.7, and 7.6 mg/24 hours) are available, corresponding to blood levels attained with sublingual asenapine exposures of 10, 15, and 20 mg/d, respectively. Application sites are rotated daily and include the upper arms, upper back, abdomen, or hip. Dysgeusia and hypoesthesia of the tongue are avoided with the use of the patch, and there are no food or drink restrictions. Attention will be needed in case of dermal reactions, similar to that observed with other medication patches.
Bottom Line
The asenapine transdermal drug delivery system appears to be efficacious and reasonably well tolerated. The treatment of schizophrenia is complex and requires individualized choices in order to optimize outcomes. A patch may be the preferred formulation for selected patients, and caregivers will have the ability to visually check if the medication is being used.
Related Resource
- Hisamitsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
Drug Brand Names
Asenapine sublingual • Saphris
Asenapine transdermal system • Secuado
Lithium • Eskalith, Lithobid
Valproate • Depakote
1. Noven. US FDA approves SECUADO® (asenapine) transdermal system, the first-and-only transdermal patch for the treatment of adults with schizophrenia. October 15, 2019. Accessed January 15, 2021. https://www.noven.com/wp-content/uploads/2020/04/PR101519.pdf
2. US Food and Drug Administration. Center for Drug Evaluation and Research. Approval Package for: APPLICATION NUMBER: 212268Orig1s000. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000Approv.pdf
3. Hisam itsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
4. Allergan USA, Inc. SAPHRIS® (asenapine) sublingual tablets prescribing information. February 2017. Accessed January 15, 2021. https://media.allergan.com/actavis/actavis/media/allergan-pdf-documents/product-prescribing/Final_labeling_text_SAPHRIS-clean-02-2017.pdf
5. Citrome L. Asenapine review, part I: chemistry, receptor affinity profile, pharmacokinetics and metabolism. Expert Opin Drug Metab Toxicol. 2014;10(6):893-903.
6. Citrome L. Asenapine review, part II: clinical efficacy, safety and tolerability. Expert Opin Drug Saf. 2014;13(6):803-830.
7. Citrome L. Chapter 31: Asenapine. In: Schatzberg AF, Nemeroff CB, eds. The American Psychiatric Association Publishing Textbook of Psychopharmacology, 5th ed. American Psychiatric Association Publishing; 2017:797-808.
8. Citrome L, Zeni CM, Correll CU. Patches: established and emerging transdermal treatments in psychiatry. J Clin Psychiatry. 2019;80(4):18nr12554. doi: 10.4088/JCP.18nr12554
9. Shayegan DK, Stahl SM. Atypical antipsychotics: matching receptor profile to individual patient’s clinical profile. CNS Spectr. 2004;9(10 suppl 11):6-14.
10. Castelli M, Suzuki K, Komaroff M, et al. Pharmacokinetic profile of asenapine transdermal system HP-3070: The first antipsychotic patch in the US. Poster presented virtually at the American Society for Clinical Psychopharmacology (ASCP) 2020 Annual Meeting, May 29-30, 2020. https://www.psychiatrist.com/ascpcorner/Documents/ascp2020/3_ASCP%20Poster%20Abstracts%202020-JCP.pdf
11. Citrome L, Walling DP, Zeni CM, et al. Efficacy and safety of HP-3070, an asenapine transdermal system, in patients with schizophrenia: a phase 3, randomized, placebo-controlled study. J Clin Psychiatry. 2020;82(1):20m13602. doi: 10.4088/JCP.20m13602
12. US Food and Drug Administration. Drug Approval Package: SECAUDO. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000TOC.cfm
13. Szegedi A, Verweij P, van Duijnhoven W, et al. Meta-analyses of the efficacy of asenapine for acute schizophrenia: comparisons with placebo and other antipsychotics. J Clin Psychiatry. 2012;73(12):1533-1540.
14. Citrome L. Asenapine for schizophrenia and bipolar disorder: a review of the efficacy and safety profile for this newly approved sublingually absorbed second-generation antipsychotic. Int J Clin Pract. 2009;63(12):1762-1784.
1. Noven. US FDA approves SECUADO® (asenapine) transdermal system, the first-and-only transdermal patch for the treatment of adults with schizophrenia. October 15, 2019. Accessed January 15, 2021. https://www.noven.com/wp-content/uploads/2020/04/PR101519.pdf
2. US Food and Drug Administration. Center for Drug Evaluation and Research. Approval Package for: APPLICATION NUMBER: 212268Orig1s000. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000Approv.pdf
3. Hisam itsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
4. Allergan USA, Inc. SAPHRIS® (asenapine) sublingual tablets prescribing information. February 2017. Accessed January 15, 2021. https://media.allergan.com/actavis/actavis/media/allergan-pdf-documents/product-prescribing/Final_labeling_text_SAPHRIS-clean-02-2017.pdf
5. Citrome L. Asenapine review, part I: chemistry, receptor affinity profile, pharmacokinetics and metabolism. Expert Opin Drug Metab Toxicol. 2014;10(6):893-903.
6. Citrome L. Asenapine review, part II: clinical efficacy, safety and tolerability. Expert Opin Drug Saf. 2014;13(6):803-830.
7. Citrome L. Chapter 31: Asenapine. In: Schatzberg AF, Nemeroff CB, eds. The American Psychiatric Association Publishing Textbook of Psychopharmacology, 5th ed. American Psychiatric Association Publishing; 2017:797-808.
8. Citrome L, Zeni CM, Correll CU. Patches: established and emerging transdermal treatments in psychiatry. J Clin Psychiatry. 2019;80(4):18nr12554. doi: 10.4088/JCP.18nr12554
9. Shayegan DK, Stahl SM. Atypical antipsychotics: matching receptor profile to individual patient’s clinical profile. CNS Spectr. 2004;9(10 suppl 11):6-14.
10. Castelli M, Suzuki K, Komaroff M, et al. Pharmacokinetic profile of asenapine transdermal system HP-3070: The first antipsychotic patch in the US. Poster presented virtually at the American Society for Clinical Psychopharmacology (ASCP) 2020 Annual Meeting, May 29-30, 2020. https://www.psychiatrist.com/ascpcorner/Documents/ascp2020/3_ASCP%20Poster%20Abstracts%202020-JCP.pdf
11. Citrome L, Walling DP, Zeni CM, et al. Efficacy and safety of HP-3070, an asenapine transdermal system, in patients with schizophrenia: a phase 3, randomized, placebo-controlled study. J Clin Psychiatry. 2020;82(1):20m13602. doi: 10.4088/JCP.20m13602
12. US Food and Drug Administration. Drug Approval Package: SECAUDO. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000TOC.cfm
13. Szegedi A, Verweij P, van Duijnhoven W, et al. Meta-analyses of the efficacy of asenapine for acute schizophrenia: comparisons with placebo and other antipsychotics. J Clin Psychiatry. 2012;73(12):1533-1540.
14. Citrome L. Asenapine for schizophrenia and bipolar disorder: a review of the efficacy and safety profile for this newly approved sublingually absorbed second-generation antipsychotic. Int J Clin Pract. 2009;63(12):1762-1784.
Suvorexant: An option for preventing delirium?
Delirium is characterized by a disturbance of consciousness or cognition that typically has a rapid onset and fluctuating course.1 Up to 42% of hospitalized geriatric patients experience delirium.1 Approximately 10% to 31% of these patients have the condition upon admission, and the remainder develop it during their hospitalization.1 Unfortunately, options for preventing or treating delirium are limited. Benzodiazepines and antipsychotic medications have been used to treat problematic behaviors associated with delirium, but they do not effectively reduce the occurrence, duration, or severity of this condition.2,3
Recent evidence suggests that suvorexant, which is FDA-approved for insomnia, may be useful for preventing delirium. Suvorexant—a dual orexin receptor (OX1R, OX2R) antagonist—promotes sleep onset and maintenance, and is associated with normal measures of sleep activity such as rapid eye movement (REM) sleep, non-REM sleep, and sleep stage–specific electroencephalographic profiles.4 Here we review 3 studies that evaluated suvorexant for preventing delirium.
Hatta et al.5 In this randomized, placebo-controlled, blinded, multicenter study, 72 patients (age 65 to 89) newly admitted to an ICU were randomized to suvorexant, 15 mg/d, (n = 36) or placebo (n = 36) for 3 days.5 None of the patients taking suvorexant developed delirium, whereas 17% (6 patients) in the placebo group did (P = .025).5
Azuma et al.6 In this 7-day, blinded, randomized study of 70 adult patients (age ≥20) admitted to an ICU, 34 participants received suvorexant (15 mg nightly for age <65, 20 mg nightly for age ≥65) and the rest received treatment as usual (TAU). Suvorexant was associated with a lower incidence of delirium symptoms (n = 6, 17.6%) compared with TAU (n = 17, 47.2%) (P = .011).6 The onset of delirium was earlier in the TAU group (P < .05).6
Hatta et al.7 In this large prospective, observational study of adults (age >65), 526 patients with significant risk factors for delirium were prescribed suvorexant and/or ramelteon. Approximately 16% of the patients who received either or both of these medications met DSM-5 criteria for delirium, compared with 24% who did not receive these medications (P = .005).7
Acknowledgment
The authors thank Jakob Evans, BS, for compiling much of the research for this article.
1. Siddiqi N, House AO, Holmes JD. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 2006;35(4):350-364.
2. Lonergan E, Luxenberg J, Areosa Sastre A. Benzodiazepines for delirium. Cochrane Database Syst Rev. 2009;2009(4):CD006379.
3. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2018;6(6):CD005594.
4. Coleman PJ, Gotter AL, Herring WJ, et al. The discovery of suvorexant, the first orexin receptor drug for insomnia. Annu Rev Pharmacol Toxicol. 2017;57:509-533.
5. Hatta K, Kishi Y, Wada K, et al. Preventive effects of suvorexant on delirium: a randomized placebo-controlled trial. J Clin Psychiatry. 2017;78(8):e970-e979.
6. Azuma K, Takaesu Y, Soeda H, et al. Ability of suvorexant to prevent delirium in patients in the intensive care unit: a randomized controlled trial. Acute Med Surg. 2018;5(4):362-368.
7. Hatta K, Kishi Y, Wada K, et al. Real-world effectiveness of ramelteon and suvorexant for delirium prevention in 948 patients with delirium risk factors. J Clin Psychiatry. 2019;81(1):19m12865. doi: 10.4088/JCP.19m12865
Delirium is characterized by a disturbance of consciousness or cognition that typically has a rapid onset and fluctuating course.1 Up to 42% of hospitalized geriatric patients experience delirium.1 Approximately 10% to 31% of these patients have the condition upon admission, and the remainder develop it during their hospitalization.1 Unfortunately, options for preventing or treating delirium are limited. Benzodiazepines and antipsychotic medications have been used to treat problematic behaviors associated with delirium, but they do not effectively reduce the occurrence, duration, or severity of this condition.2,3
Recent evidence suggests that suvorexant, which is FDA-approved for insomnia, may be useful for preventing delirium. Suvorexant—a dual orexin receptor (OX1R, OX2R) antagonist—promotes sleep onset and maintenance, and is associated with normal measures of sleep activity such as rapid eye movement (REM) sleep, non-REM sleep, and sleep stage–specific electroencephalographic profiles.4 Here we review 3 studies that evaluated suvorexant for preventing delirium.
Hatta et al.5 In this randomized, placebo-controlled, blinded, multicenter study, 72 patients (age 65 to 89) newly admitted to an ICU were randomized to suvorexant, 15 mg/d, (n = 36) or placebo (n = 36) for 3 days.5 None of the patients taking suvorexant developed delirium, whereas 17% (6 patients) in the placebo group did (P = .025).5
Azuma et al.6 In this 7-day, blinded, randomized study of 70 adult patients (age ≥20) admitted to an ICU, 34 participants received suvorexant (15 mg nightly for age <65, 20 mg nightly for age ≥65) and the rest received treatment as usual (TAU). Suvorexant was associated with a lower incidence of delirium symptoms (n = 6, 17.6%) compared with TAU (n = 17, 47.2%) (P = .011).6 The onset of delirium was earlier in the TAU group (P < .05).6
Hatta et al.7 In this large prospective, observational study of adults (age >65), 526 patients with significant risk factors for delirium were prescribed suvorexant and/or ramelteon. Approximately 16% of the patients who received either or both of these medications met DSM-5 criteria for delirium, compared with 24% who did not receive these medications (P = .005).7
Acknowledgment
The authors thank Jakob Evans, BS, for compiling much of the research for this article.
Delirium is characterized by a disturbance of consciousness or cognition that typically has a rapid onset and fluctuating course.1 Up to 42% of hospitalized geriatric patients experience delirium.1 Approximately 10% to 31% of these patients have the condition upon admission, and the remainder develop it during their hospitalization.1 Unfortunately, options for preventing or treating delirium are limited. Benzodiazepines and antipsychotic medications have been used to treat problematic behaviors associated with delirium, but they do not effectively reduce the occurrence, duration, or severity of this condition.2,3
Recent evidence suggests that suvorexant, which is FDA-approved for insomnia, may be useful for preventing delirium. Suvorexant—a dual orexin receptor (OX1R, OX2R) antagonist—promotes sleep onset and maintenance, and is associated with normal measures of sleep activity such as rapid eye movement (REM) sleep, non-REM sleep, and sleep stage–specific electroencephalographic profiles.4 Here we review 3 studies that evaluated suvorexant for preventing delirium.
Hatta et al.5 In this randomized, placebo-controlled, blinded, multicenter study, 72 patients (age 65 to 89) newly admitted to an ICU were randomized to suvorexant, 15 mg/d, (n = 36) or placebo (n = 36) for 3 days.5 None of the patients taking suvorexant developed delirium, whereas 17% (6 patients) in the placebo group did (P = .025).5
Azuma et al.6 In this 7-day, blinded, randomized study of 70 adult patients (age ≥20) admitted to an ICU, 34 participants received suvorexant (15 mg nightly for age <65, 20 mg nightly for age ≥65) and the rest received treatment as usual (TAU). Suvorexant was associated with a lower incidence of delirium symptoms (n = 6, 17.6%) compared with TAU (n = 17, 47.2%) (P = .011).6 The onset of delirium was earlier in the TAU group (P < .05).6
Hatta et al.7 In this large prospective, observational study of adults (age >65), 526 patients with significant risk factors for delirium were prescribed suvorexant and/or ramelteon. Approximately 16% of the patients who received either or both of these medications met DSM-5 criteria for delirium, compared with 24% who did not receive these medications (P = .005).7
Acknowledgment
The authors thank Jakob Evans, BS, for compiling much of the research for this article.
1. Siddiqi N, House AO, Holmes JD. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 2006;35(4):350-364.
2. Lonergan E, Luxenberg J, Areosa Sastre A. Benzodiazepines for delirium. Cochrane Database Syst Rev. 2009;2009(4):CD006379.
3. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2018;6(6):CD005594.
4. Coleman PJ, Gotter AL, Herring WJ, et al. The discovery of suvorexant, the first orexin receptor drug for insomnia. Annu Rev Pharmacol Toxicol. 2017;57:509-533.
5. Hatta K, Kishi Y, Wada K, et al. Preventive effects of suvorexant on delirium: a randomized placebo-controlled trial. J Clin Psychiatry. 2017;78(8):e970-e979.
6. Azuma K, Takaesu Y, Soeda H, et al. Ability of suvorexant to prevent delirium in patients in the intensive care unit: a randomized controlled trial. Acute Med Surg. 2018;5(4):362-368.
7. Hatta K, Kishi Y, Wada K, et al. Real-world effectiveness of ramelteon and suvorexant for delirium prevention in 948 patients with delirium risk factors. J Clin Psychiatry. 2019;81(1):19m12865. doi: 10.4088/JCP.19m12865
1. Siddiqi N, House AO, Holmes JD. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 2006;35(4):350-364.
2. Lonergan E, Luxenberg J, Areosa Sastre A. Benzodiazepines for delirium. Cochrane Database Syst Rev. 2009;2009(4):CD006379.
3. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2018;6(6):CD005594.
4. Coleman PJ, Gotter AL, Herring WJ, et al. The discovery of suvorexant, the first orexin receptor drug for insomnia. Annu Rev Pharmacol Toxicol. 2017;57:509-533.
5. Hatta K, Kishi Y, Wada K, et al. Preventive effects of suvorexant on delirium: a randomized placebo-controlled trial. J Clin Psychiatry. 2017;78(8):e970-e979.
6. Azuma K, Takaesu Y, Soeda H, et al. Ability of suvorexant to prevent delirium in patients in the intensive care unit: a randomized controlled trial. Acute Med Surg. 2018;5(4):362-368.
7. Hatta K, Kishi Y, Wada K, et al. Real-world effectiveness of ramelteon and suvorexant for delirium prevention in 948 patients with delirium risk factors. J Clin Psychiatry. 2019;81(1):19m12865. doi: 10.4088/JCP.19m12865
Key questions to ask patients who are veterans
The Mission Act—signed into law in 2018—recognizes that the health care needs of patients who are veterans can no longer be fully served by the Veterans Health Administration.1 This act allows some veterans who are enrolled in the Veterans Affairs (VA) health care system or otherwise entitled to VA care to access treatment outside of VA facilities.1 As a result, psychiatrists may treat veterans more frequently.
During such patients’ initial visit, obtaining a detailed history of their military service can reveal vital clinical information and establish a therapeutic alliance that can help foster positive treatment outcomes. Here we offer an A-to-L list of important questions to ask veterans about their military service, and explanations of why these questions are valuable.
Attained rank. What rank did you attain during your military service? Did you retire from the military? How many years did you serve?
Asking about your patient’s rank, retirement status, and time in service is vital to understanding their military experience. By military law, only individuals who retired from the military can use their rank as an identifier after they leave the military, although some veterans may not wish to be called by their rank in a clinical setting.
Branch. Which branch of the military did you serve? Were you in Active Duty, the Reserves, or the National Guard?
Military members often take great pride in service of their specific branch. Each branch has its own language, culture, values, and exposures. If your patient has served in a combination of Active Duty, Reserves, and/or National Guard, ask how much time they spent in each.
Culture. What part of the military culture was positive or negative for you?
Continue to: There is a clear culture...
There is a clear culture within the military. Some veterans may feel lost without the military structure, and even devalued without the respect of rank. Others may feel jaded and spiteful about the strict military culture, procedures, and expectations.
Discharge. When, why, and under what circumstances were you discharged? What type of discharge did you receive?
There are 6 types of discharge: Honorable, General, Other than Honorable (OTH), Entry Level Separation, Bad Conduct, and Dishonorable. The type of discharge a veteran received may impact what resources are available to them. It also can influence a veteran’s perception of their military career.
Exposures. Were you exposed to combat, death, explosive blasts, or hazardous chemicals?
Do not ask a veteran if they have killed anyone. This question is both disrespectful and highly presumptuous because most veterans have not killed anyone. Be respectful of their experiences. Depending on the veteran’s mission, they may have unique exposures (Agent Orange, burn pits, detainee camps, etc.). Consider asking follow-up questions to learn the details of these exposures.
Continue to: Family impact
Family impact. How has your military service impacted your family?
A veteran’s military service often affects family members. Deployments can cause strain on marital relationships, children’s birthdays and special events may be missed, and extended family may have negative reactions to military service. Understanding the impact on the veteran’s family members can help uncover potential stressful relationships as well as help enhance any positive support systems that are available at home.
Go. Where were you stationed? Were you deployed?
Training location, geography of combat theater, peace-keeping locations, and area of station can all profoundly impact a veteran’s military experience. Ask follow-up questions about their duty stations, deployment locations, and experiences with these locations.
Hot water. Did you ever get into “trouble” while serving the military (eg, lose rank, get arrested, etc.)? How did you respond to the military’s method of discipline?
Continue to: Although it may be difficult...
Although it may be difficult or uncomfortable to ask your patient if they experienced any disciplinary action, this information may prove useful. It can help provide context when you discuss the veteran’s ease of assimilation into civilian life and other important information regarding the type of discharge.
Injuries. Have you experienced any moral, physical, sexual, emotional, or concussive injuries?
Moral injury, guilt, and regret are common for veterans. Not all injuries are from combat. Your patient may have experienced sexual assault, hazing rituals, pranks, etc.
Job. What was your job in the military? What kind of security clearance did you have?
Note that not all veterans’ “jobs” in the military accurately reflect the duties and tasks that they actually performed. Security clearance will often influence the duties and tasks they were required to perform.
Continue to: Keeping it inside
Keeping it inside. Do you have anyone to talk with about your military experiences?
Many veterans feel uncomfortable discussing their experiences with others. Some veterans may be concerned that others will not understand what they went through. Some might perceive that disclosing their experiences could burden other people, or they may be concerned that explaining their experiences may be too shocking. Asking this question may present an opportunity for you to suggest psychotherapy for your patient.
Life as a civilian. How is your life different as a civilian? How have you adjusted to civilian life?
During the process of assimilation into civilian life, veterans may experience symptoms of depression, posttraumatic stress disorder, anxiety, or other disorders. These symptoms may emerge and/or become exacerbated during their transition to civilian life.
1. VA MISSION Act of 2018 (VA Maintaining Internal Systems and Strengthening Integrated Outside Networks Act), S 2372, 115th Cong, 2nd Sess, HR Doc No. 115-671 (2018).
The Mission Act—signed into law in 2018—recognizes that the health care needs of patients who are veterans can no longer be fully served by the Veterans Health Administration.1 This act allows some veterans who are enrolled in the Veterans Affairs (VA) health care system or otherwise entitled to VA care to access treatment outside of VA facilities.1 As a result, psychiatrists may treat veterans more frequently.
During such patients’ initial visit, obtaining a detailed history of their military service can reveal vital clinical information and establish a therapeutic alliance that can help foster positive treatment outcomes. Here we offer an A-to-L list of important questions to ask veterans about their military service, and explanations of why these questions are valuable.
Attained rank. What rank did you attain during your military service? Did you retire from the military? How many years did you serve?
Asking about your patient’s rank, retirement status, and time in service is vital to understanding their military experience. By military law, only individuals who retired from the military can use their rank as an identifier after they leave the military, although some veterans may not wish to be called by their rank in a clinical setting.
Branch. Which branch of the military did you serve? Were you in Active Duty, the Reserves, or the National Guard?
Military members often take great pride in service of their specific branch. Each branch has its own language, culture, values, and exposures. If your patient has served in a combination of Active Duty, Reserves, and/or National Guard, ask how much time they spent in each.
Culture. What part of the military culture was positive or negative for you?
Continue to: There is a clear culture...
There is a clear culture within the military. Some veterans may feel lost without the military structure, and even devalued without the respect of rank. Others may feel jaded and spiteful about the strict military culture, procedures, and expectations.
Discharge. When, why, and under what circumstances were you discharged? What type of discharge did you receive?
There are 6 types of discharge: Honorable, General, Other than Honorable (OTH), Entry Level Separation, Bad Conduct, and Dishonorable. The type of discharge a veteran received may impact what resources are available to them. It also can influence a veteran’s perception of their military career.
Exposures. Were you exposed to combat, death, explosive blasts, or hazardous chemicals?
Do not ask a veteran if they have killed anyone. This question is both disrespectful and highly presumptuous because most veterans have not killed anyone. Be respectful of their experiences. Depending on the veteran’s mission, they may have unique exposures (Agent Orange, burn pits, detainee camps, etc.). Consider asking follow-up questions to learn the details of these exposures.
Continue to: Family impact
Family impact. How has your military service impacted your family?
A veteran’s military service often affects family members. Deployments can cause strain on marital relationships, children’s birthdays and special events may be missed, and extended family may have negative reactions to military service. Understanding the impact on the veteran’s family members can help uncover potential stressful relationships as well as help enhance any positive support systems that are available at home.
Go. Where were you stationed? Were you deployed?
Training location, geography of combat theater, peace-keeping locations, and area of station can all profoundly impact a veteran’s military experience. Ask follow-up questions about their duty stations, deployment locations, and experiences with these locations.
Hot water. Did you ever get into “trouble” while serving the military (eg, lose rank, get arrested, etc.)? How did you respond to the military’s method of discipline?
Continue to: Although it may be difficult...
Although it may be difficult or uncomfortable to ask your patient if they experienced any disciplinary action, this information may prove useful. It can help provide context when you discuss the veteran’s ease of assimilation into civilian life and other important information regarding the type of discharge.
Injuries. Have you experienced any moral, physical, sexual, emotional, or concussive injuries?
Moral injury, guilt, and regret are common for veterans. Not all injuries are from combat. Your patient may have experienced sexual assault, hazing rituals, pranks, etc.
Job. What was your job in the military? What kind of security clearance did you have?
Note that not all veterans’ “jobs” in the military accurately reflect the duties and tasks that they actually performed. Security clearance will often influence the duties and tasks they were required to perform.
Continue to: Keeping it inside
Keeping it inside. Do you have anyone to talk with about your military experiences?
Many veterans feel uncomfortable discussing their experiences with others. Some veterans may be concerned that others will not understand what they went through. Some might perceive that disclosing their experiences could burden other people, or they may be concerned that explaining their experiences may be too shocking. Asking this question may present an opportunity for you to suggest psychotherapy for your patient.
Life as a civilian. How is your life different as a civilian? How have you adjusted to civilian life?
During the process of assimilation into civilian life, veterans may experience symptoms of depression, posttraumatic stress disorder, anxiety, or other disorders. These symptoms may emerge and/or become exacerbated during their transition to civilian life.
The Mission Act—signed into law in 2018—recognizes that the health care needs of patients who are veterans can no longer be fully served by the Veterans Health Administration.1 This act allows some veterans who are enrolled in the Veterans Affairs (VA) health care system or otherwise entitled to VA care to access treatment outside of VA facilities.1 As a result, psychiatrists may treat veterans more frequently.
During such patients’ initial visit, obtaining a detailed history of their military service can reveal vital clinical information and establish a therapeutic alliance that can help foster positive treatment outcomes. Here we offer an A-to-L list of important questions to ask veterans about their military service, and explanations of why these questions are valuable.
Attained rank. What rank did you attain during your military service? Did you retire from the military? How many years did you serve?
Asking about your patient’s rank, retirement status, and time in service is vital to understanding their military experience. By military law, only individuals who retired from the military can use their rank as an identifier after they leave the military, although some veterans may not wish to be called by their rank in a clinical setting.
Branch. Which branch of the military did you serve? Were you in Active Duty, the Reserves, or the National Guard?
Military members often take great pride in service of their specific branch. Each branch has its own language, culture, values, and exposures. If your patient has served in a combination of Active Duty, Reserves, and/or National Guard, ask how much time they spent in each.
Culture. What part of the military culture was positive or negative for you?
Continue to: There is a clear culture...
There is a clear culture within the military. Some veterans may feel lost without the military structure, and even devalued without the respect of rank. Others may feel jaded and spiteful about the strict military culture, procedures, and expectations.
Discharge. When, why, and under what circumstances were you discharged? What type of discharge did you receive?
There are 6 types of discharge: Honorable, General, Other than Honorable (OTH), Entry Level Separation, Bad Conduct, and Dishonorable. The type of discharge a veteran received may impact what resources are available to them. It also can influence a veteran’s perception of their military career.
Exposures. Were you exposed to combat, death, explosive blasts, or hazardous chemicals?
Do not ask a veteran if they have killed anyone. This question is both disrespectful and highly presumptuous because most veterans have not killed anyone. Be respectful of their experiences. Depending on the veteran’s mission, they may have unique exposures (Agent Orange, burn pits, detainee camps, etc.). Consider asking follow-up questions to learn the details of these exposures.
Continue to: Family impact
Family impact. How has your military service impacted your family?
A veteran’s military service often affects family members. Deployments can cause strain on marital relationships, children’s birthdays and special events may be missed, and extended family may have negative reactions to military service. Understanding the impact on the veteran’s family members can help uncover potential stressful relationships as well as help enhance any positive support systems that are available at home.
Go. Where were you stationed? Were you deployed?
Training location, geography of combat theater, peace-keeping locations, and area of station can all profoundly impact a veteran’s military experience. Ask follow-up questions about their duty stations, deployment locations, and experiences with these locations.
Hot water. Did you ever get into “trouble” while serving the military (eg, lose rank, get arrested, etc.)? How did you respond to the military’s method of discipline?
Continue to: Although it may be difficult...
Although it may be difficult or uncomfortable to ask your patient if they experienced any disciplinary action, this information may prove useful. It can help provide context when you discuss the veteran’s ease of assimilation into civilian life and other important information regarding the type of discharge.
Injuries. Have you experienced any moral, physical, sexual, emotional, or concussive injuries?
Moral injury, guilt, and regret are common for veterans. Not all injuries are from combat. Your patient may have experienced sexual assault, hazing rituals, pranks, etc.
Job. What was your job in the military? What kind of security clearance did you have?
Note that not all veterans’ “jobs” in the military accurately reflect the duties and tasks that they actually performed. Security clearance will often influence the duties and tasks they were required to perform.
Continue to: Keeping it inside
Keeping it inside. Do you have anyone to talk with about your military experiences?
Many veterans feel uncomfortable discussing their experiences with others. Some veterans may be concerned that others will not understand what they went through. Some might perceive that disclosing their experiences could burden other people, or they may be concerned that explaining their experiences may be too shocking. Asking this question may present an opportunity for you to suggest psychotherapy for your patient.
Life as a civilian. How is your life different as a civilian? How have you adjusted to civilian life?
During the process of assimilation into civilian life, veterans may experience symptoms of depression, posttraumatic stress disorder, anxiety, or other disorders. These symptoms may emerge and/or become exacerbated during their transition to civilian life.
1. VA MISSION Act of 2018 (VA Maintaining Internal Systems and Strengthening Integrated Outside Networks Act), S 2372, 115th Cong, 2nd Sess, HR Doc No. 115-671 (2018).
1. VA MISSION Act of 2018 (VA Maintaining Internal Systems and Strengthening Integrated Outside Networks Act), S 2372, 115th Cong, 2nd Sess, HR Doc No. 115-671 (2018).
Helping survivors of human trafficking
Human trafficking (HT) is a secretive, multibillion dollar criminal industry involving the use of coercion, threats, and fraud to force individuals to engage in labor or commercial sex acts. In 2017, the International Labour Organization estimated that 24.9 million people worldwide were victims of forced labor (ie, working under threat or coercion).1 Risk factors for individuals who are vulnerable to HT include recent migration, substance use, housing insecurity, runaway youth, and mental illness. Traffickers continue the cycle of HT through isolation and emotional, physical, financial, and verbal abuse.
Survivors of HT may avoid seeking health care due to cultural reasons or feelings of guilt, isolation, distrust, or fear of criminal sanctions. There can be missed opportunities for victims to obtain help through health care services, law enforcement, child welfare services, or even family or friends. In a study of 173 survivors of HT in the United States, 68% of those who were currently trafficked visited with a health care professional at least once and were not identified as being trafficked.2 Psychiatrists rarely receive education on HT, which can lead to missed opportunities for identifying victims. Table 1 lists screening questions psychiatrists can ask patients they suspect may be trafficked.
The psychiatric sequelae of trafficking
Survivors of HT commonly experience psychiatric illness, substance use, pain, sexually transmitted diseases, and unplanned pregnancies.3 Here we discuss some of the psychiatric conditions that are common among HT survivors, and outline a multidisciplinary approach to their care.
PTSD, mood disorders, and anxiety disorders. Studies suggest survivors of HT who seek care have a high prevalence of depression, anxiety, and posttraumatic stress disorder (PTSD).3 Survivors may have experienced multiple repetitive trauma, such as physical and sexual abuse.3 Compared with survivors of forced labor trafficking, survivors of sex trafficking have higher rates of childhood abuse, violence during trafficking, severe symptoms of PTSD, and comorbid depression and PTSD.4 For survivors with PTSD, consider psychosocial interventions that address social support, coping strategies, and community reintegration.5 Survivors can also benefit from trauma-informed care that focuses on the cognitive aspect of the trauma, such as cognitive processing therapy, which involves cognitive restructuring without a written account of the trauma.6
Substance use disorders. Some individuals who are trafficked may be forced to use drugs of abuse or alcohol, while others may use substances to help cope while they are being trafficked or afterwards.3 For these patients, motivational interviewing may be beneficial. Also, consider referring them to detoxification or rehabilitation programs.
Suicide and self-harm. In a study of 98 HT survivors in England, 33% reported a history of self-harm before receiving care and 25% engaged in self-harm during care.7 After engaging in self-harm, survivors of HT were more likely to be admitted to psychiatric inpatient units than were patients who had not been trafficked.7 It is crucial to conduct a suicide risk assessment as part of the trauma-informed care of these patients.
Other conditions. In addition to psychiatric illness, survivors of HT may experience physical symptoms such as headache, back pain, stomach pain, fatigue, dizziness, memory problems, and weight loss.3 Referral to other specialties may be necessary for addressing any of the patient’s other conditions.
Continue to: Use a multidisciplinary approach
Use a multidisciplinary approach
Treatment for survivors of HT should be tailored to their specific mental health needs by including psychopharmacology; individual, group, or family psychotherapy; and peer advocate support. Rehabilitation, social services, and case management should also be considered. The care of survivors of HT benefits from a multidisciplinary, culturally-sensitive, and trauma-informed approach. Table 28 describes the PEARR Tool (Provide privacy, Educate, Ask, Respect, and Respond), which offers physicians 4 steps for addressing abuse, neglect, or violence with their patients. Also, the National Human Trafficking Hotline (1-888-373-7888) is available 24/7 for trafficked persons, survivors, and health care professionals to provide guidance on reporting laws and finding additional resources such as housing and legal services.

1. International Labour Organization, the Walk Free Foundation. Global Estimates of Modern Slavery: forced labour and forced marriage. Published 2017. Accessed January 14, 2021. www.ilo.org/global/publications/books/WCMS_575479/lang--en/index.htm
2. Chisolm-Straker M, Baldwin S, Gaïgbé-Togbé B, et al. Health care and human trafficking: we are seeing the unseen. J Health Care Poor Underserved. 2016;27(3):1220-1233.
3. Ottisova L, Hemmings S, Howard LM, et al. Prevalence and risk of violence and the mental, physical and sexual health problems associated with human trafficking: an updated systematic review. Epidemiol Psychiatr Sci. 2016;25(4):317-341.
4. Hopper EK, Gonzalez LD. A comparison of psychological symptoms in survivors of sex and labor trafficking. Behav Med. 2018;44(3):177-188.
5. Okech D, Hanseen N, Howard W, et al. Social support, dysfunctional coping, and community reintegration as predictors of PTSD among human trafficking survivors. Behav Med. 2018;44(3):209-218.
6. Salami T, Gordon M, Coverdale J, et al. What therapies are favored in the treatment of the psychological sequelae of trauma in human trafficking victims? J Psychiatr Pract. 2018;24(2):87-96.
7. Borschmann R, Oram S, Kinner SA, et al. Self-harm among adult victims of human trafficking who accessed secondary mental health services in England. Psychiatr Serv. 2017;68(2):207-210.
8. Using the PEARR Tool. Dignity Health. Published 2019. Accessed January 14, 2021. https://www.dignityhealth.org/hello-humankindness/human-trafficking/victimcentered-and-trauma-informed/using-the-pearr-tool
Human trafficking (HT) is a secretive, multibillion dollar criminal industry involving the use of coercion, threats, and fraud to force individuals to engage in labor or commercial sex acts. In 2017, the International Labour Organization estimated that 24.9 million people worldwide were victims of forced labor (ie, working under threat or coercion).1 Risk factors for individuals who are vulnerable to HT include recent migration, substance use, housing insecurity, runaway youth, and mental illness. Traffickers continue the cycle of HT through isolation and emotional, physical, financial, and verbal abuse.
Survivors of HT may avoid seeking health care due to cultural reasons or feelings of guilt, isolation, distrust, or fear of criminal sanctions. There can be missed opportunities for victims to obtain help through health care services, law enforcement, child welfare services, or even family or friends. In a study of 173 survivors of HT in the United States, 68% of those who were currently trafficked visited with a health care professional at least once and were not identified as being trafficked.2 Psychiatrists rarely receive education on HT, which can lead to missed opportunities for identifying victims. Table 1 lists screening questions psychiatrists can ask patients they suspect may be trafficked.
The psychiatric sequelae of trafficking
Survivors of HT commonly experience psychiatric illness, substance use, pain, sexually transmitted diseases, and unplanned pregnancies.3 Here we discuss some of the psychiatric conditions that are common among HT survivors, and outline a multidisciplinary approach to their care.
PTSD, mood disorders, and anxiety disorders. Studies suggest survivors of HT who seek care have a high prevalence of depression, anxiety, and posttraumatic stress disorder (PTSD).3 Survivors may have experienced multiple repetitive trauma, such as physical and sexual abuse.3 Compared with survivors of forced labor trafficking, survivors of sex trafficking have higher rates of childhood abuse, violence during trafficking, severe symptoms of PTSD, and comorbid depression and PTSD.4 For survivors with PTSD, consider psychosocial interventions that address social support, coping strategies, and community reintegration.5 Survivors can also benefit from trauma-informed care that focuses on the cognitive aspect of the trauma, such as cognitive processing therapy, which involves cognitive restructuring without a written account of the trauma.6
Substance use disorders. Some individuals who are trafficked may be forced to use drugs of abuse or alcohol, while others may use substances to help cope while they are being trafficked or afterwards.3 For these patients, motivational interviewing may be beneficial. Also, consider referring them to detoxification or rehabilitation programs.
Suicide and self-harm. In a study of 98 HT survivors in England, 33% reported a history of self-harm before receiving care and 25% engaged in self-harm during care.7 After engaging in self-harm, survivors of HT were more likely to be admitted to psychiatric inpatient units than were patients who had not been trafficked.7 It is crucial to conduct a suicide risk assessment as part of the trauma-informed care of these patients.
Other conditions. In addition to psychiatric illness, survivors of HT may experience physical symptoms such as headache, back pain, stomach pain, fatigue, dizziness, memory problems, and weight loss.3 Referral to other specialties may be necessary for addressing any of the patient’s other conditions.
Continue to: Use a multidisciplinary approach
Use a multidisciplinary approach
Treatment for survivors of HT should be tailored to their specific mental health needs by including psychopharmacology; individual, group, or family psychotherapy; and peer advocate support. Rehabilitation, social services, and case management should also be considered. The care of survivors of HT benefits from a multidisciplinary, culturally-sensitive, and trauma-informed approach. Table 28 describes the PEARR Tool (Provide privacy, Educate, Ask, Respect, and Respond), which offers physicians 4 steps for addressing abuse, neglect, or violence with their patients. Also, the National Human Trafficking Hotline (1-888-373-7888) is available 24/7 for trafficked persons, survivors, and health care professionals to provide guidance on reporting laws and finding additional resources such as housing and legal services.

Human trafficking (HT) is a secretive, multibillion dollar criminal industry involving the use of coercion, threats, and fraud to force individuals to engage in labor or commercial sex acts. In 2017, the International Labour Organization estimated that 24.9 million people worldwide were victims of forced labor (ie, working under threat or coercion).1 Risk factors for individuals who are vulnerable to HT include recent migration, substance use, housing insecurity, runaway youth, and mental illness. Traffickers continue the cycle of HT through isolation and emotional, physical, financial, and verbal abuse.
Survivors of HT may avoid seeking health care due to cultural reasons or feelings of guilt, isolation, distrust, or fear of criminal sanctions. There can be missed opportunities for victims to obtain help through health care services, law enforcement, child welfare services, or even family or friends. In a study of 173 survivors of HT in the United States, 68% of those who were currently trafficked visited with a health care professional at least once and were not identified as being trafficked.2 Psychiatrists rarely receive education on HT, which can lead to missed opportunities for identifying victims. Table 1 lists screening questions psychiatrists can ask patients they suspect may be trafficked.
The psychiatric sequelae of trafficking
Survivors of HT commonly experience psychiatric illness, substance use, pain, sexually transmitted diseases, and unplanned pregnancies.3 Here we discuss some of the psychiatric conditions that are common among HT survivors, and outline a multidisciplinary approach to their care.
PTSD, mood disorders, and anxiety disorders. Studies suggest survivors of HT who seek care have a high prevalence of depression, anxiety, and posttraumatic stress disorder (PTSD).3 Survivors may have experienced multiple repetitive trauma, such as physical and sexual abuse.3 Compared with survivors of forced labor trafficking, survivors of sex trafficking have higher rates of childhood abuse, violence during trafficking, severe symptoms of PTSD, and comorbid depression and PTSD.4 For survivors with PTSD, consider psychosocial interventions that address social support, coping strategies, and community reintegration.5 Survivors can also benefit from trauma-informed care that focuses on the cognitive aspect of the trauma, such as cognitive processing therapy, which involves cognitive restructuring without a written account of the trauma.6
Substance use disorders. Some individuals who are trafficked may be forced to use drugs of abuse or alcohol, while others may use substances to help cope while they are being trafficked or afterwards.3 For these patients, motivational interviewing may be beneficial. Also, consider referring them to detoxification or rehabilitation programs.
Suicide and self-harm. In a study of 98 HT survivors in England, 33% reported a history of self-harm before receiving care and 25% engaged in self-harm during care.7 After engaging in self-harm, survivors of HT were more likely to be admitted to psychiatric inpatient units than were patients who had not been trafficked.7 It is crucial to conduct a suicide risk assessment as part of the trauma-informed care of these patients.
Other conditions. In addition to psychiatric illness, survivors of HT may experience physical symptoms such as headache, back pain, stomach pain, fatigue, dizziness, memory problems, and weight loss.3 Referral to other specialties may be necessary for addressing any of the patient’s other conditions.
Continue to: Use a multidisciplinary approach
Use a multidisciplinary approach
Treatment for survivors of HT should be tailored to their specific mental health needs by including psychopharmacology; individual, group, or family psychotherapy; and peer advocate support. Rehabilitation, social services, and case management should also be considered. The care of survivors of HT benefits from a multidisciplinary, culturally-sensitive, and trauma-informed approach. Table 28 describes the PEARR Tool (Provide privacy, Educate, Ask, Respect, and Respond), which offers physicians 4 steps for addressing abuse, neglect, or violence with their patients. Also, the National Human Trafficking Hotline (1-888-373-7888) is available 24/7 for trafficked persons, survivors, and health care professionals to provide guidance on reporting laws and finding additional resources such as housing and legal services.

1. International Labour Organization, the Walk Free Foundation. Global Estimates of Modern Slavery: forced labour and forced marriage. Published 2017. Accessed January 14, 2021. www.ilo.org/global/publications/books/WCMS_575479/lang--en/index.htm
2. Chisolm-Straker M, Baldwin S, Gaïgbé-Togbé B, et al. Health care and human trafficking: we are seeing the unseen. J Health Care Poor Underserved. 2016;27(3):1220-1233.
3. Ottisova L, Hemmings S, Howard LM, et al. Prevalence and risk of violence and the mental, physical and sexual health problems associated with human trafficking: an updated systematic review. Epidemiol Psychiatr Sci. 2016;25(4):317-341.
4. Hopper EK, Gonzalez LD. A comparison of psychological symptoms in survivors of sex and labor trafficking. Behav Med. 2018;44(3):177-188.
5. Okech D, Hanseen N, Howard W, et al. Social support, dysfunctional coping, and community reintegration as predictors of PTSD among human trafficking survivors. Behav Med. 2018;44(3):209-218.
6. Salami T, Gordon M, Coverdale J, et al. What therapies are favored in the treatment of the psychological sequelae of trauma in human trafficking victims? J Psychiatr Pract. 2018;24(2):87-96.
7. Borschmann R, Oram S, Kinner SA, et al. Self-harm among adult victims of human trafficking who accessed secondary mental health services in England. Psychiatr Serv. 2017;68(2):207-210.
8. Using the PEARR Tool. Dignity Health. Published 2019. Accessed January 14, 2021. https://www.dignityhealth.org/hello-humankindness/human-trafficking/victimcentered-and-trauma-informed/using-the-pearr-tool
1. International Labour Organization, the Walk Free Foundation. Global Estimates of Modern Slavery: forced labour and forced marriage. Published 2017. Accessed January 14, 2021. www.ilo.org/global/publications/books/WCMS_575479/lang--en/index.htm
2. Chisolm-Straker M, Baldwin S, Gaïgbé-Togbé B, et al. Health care and human trafficking: we are seeing the unseen. J Health Care Poor Underserved. 2016;27(3):1220-1233.
3. Ottisova L, Hemmings S, Howard LM, et al. Prevalence and risk of violence and the mental, physical and sexual health problems associated with human trafficking: an updated systematic review. Epidemiol Psychiatr Sci. 2016;25(4):317-341.
4. Hopper EK, Gonzalez LD. A comparison of psychological symptoms in survivors of sex and labor trafficking. Behav Med. 2018;44(3):177-188.
5. Okech D, Hanseen N, Howard W, et al. Social support, dysfunctional coping, and community reintegration as predictors of PTSD among human trafficking survivors. Behav Med. 2018;44(3):209-218.
6. Salami T, Gordon M, Coverdale J, et al. What therapies are favored in the treatment of the psychological sequelae of trauma in human trafficking victims? J Psychiatr Pract. 2018;24(2):87-96.
7. Borschmann R, Oram S, Kinner SA, et al. Self-harm among adult victims of human trafficking who accessed secondary mental health services in England. Psychiatr Serv. 2017;68(2):207-210.
8. Using the PEARR Tool. Dignity Health. Published 2019. Accessed January 14, 2021. https://www.dignityhealth.org/hello-humankindness/human-trafficking/victimcentered-and-trauma-informed/using-the-pearr-tool
Treating alcohol withdrawal syndrome: Going beyond ‘benzos’
Alcohol withdrawal syndrome (AWS) occurs when an individual who is alcohol-dependent suddenly stops or significantly reduces his/her alcohol intake.1 Symptoms of AWS, which can be fatal, include anxiety, restlessness, seizures, confusion, and delirium.1 Because benzodiazepines have been proven effective in improving symptoms of AWS, they are considered the first-line treatment, but they also carry the risk of abuse, psychomotor sedation, cognitive impairment, and interactions with alcohol.1
Non-benzodiazepine anticonvulsants (NBACs) such as valproic acid (VPA) and carbamazepine may offer benefit as alternatives or adjuncts to benzodiazepines.1 Many NBACs affect the functioning of glutamate and gamma-aminobutyric acid (GABA) neurotransmitters,2 which are particularly dysregulated in patients with AWS. Because NBACs help stabilize this imbalance, they may be useful for managing AWS and preventing relapse without the risks associated with benzodiazepines.2
Valproic acid: A better choice than carbamazepine
Compared with other NBACs, VPA and carbamazepine have been studied more extensively for treating patients with AWS, and their clinical effectiveness has been well documented.1 For mild-to-moderate AWS, VPA and carbamazepine may be as effective as benzodiazepines in reducing the severity of symptoms, and more potent for preventing withdrawal seizures.1
Increasing data suggests that compared with VPA, adjunctive treatment with carbamazepine for AWS may be more frequently associated with intolerable adverse effects such as ataxia, orthostatic hypotension, vertigo, nausea, and vomiting.1 The rapid onset of AWS requires rapid-acting pharmacotherapy.1 In attempting to rapidly achieve the desired plasma concentrations of carbamazepine, clinicians may risk inducing adverse effects. Compared with carbamazepine, VPA is associated with faster symptom resolution, a shorter duration of pharmacologic treatment, fewer transfers to the ICU, fewer withdrawal seizures, and a more favorable adverse effect profile.1 Likely due to its shorter half-life, VPA delivers its therapeutic effects without producing significant adverse effects.1
Early and aggressive treatment of AWS is needed to block kindling,3 which is characterized by the worsening of withdrawal symptoms each time an individual attempts to quit drinking alcohol. Compared with carbamazepine, VPA may be more helpful for blocking kindling.3
More data is needed
Due to ethical concerns, few studies have compared anticonvulsant medications with placebo for treating AWS.2 Most studies examining NBACs for AWS have either used the anticonvulsant as an adjunct to a benzodiazepine to examine improvement in withdrawal symptoms, or compared the anticonvulsant with placebo or another intervention to assess the amount of a benzodiazepine required for safe detoxification.2,4 Additionally, most studies examining NBACs have been short, and few followed patients after the active medication period, which limits our knowledge of the long-term effectiveness of NBACs.2 Before NBACs can replace benzodiazepines for managing AWS, further evidence from clinical trials is needed to assess their efficiency as a stand-alone treatment.
1. Maldonado JR. Novel algorithms for the prophylaxis and management of alcohol withdrawal syndromes–beyond benzodiazepines. Crit Care Clin. 2017;33(3):559-599.
2. Hammond CJ, Niciu MJ, Drew S, et al. Anticonvulsants for the treatment of alcohol withdrawal syndrome and alcohol use disorders. CNS Drugs. 2015;29(4):293-311.
3. Eyer F, Schreckenberg M, Hecht D, et al. Carbamazepine and valproate as adjuncts in the treatment of alcohol withdrawal syndrome: a retrospective cohort study. Alcohol Alcohol. 2011;46(2):177-184.
4. Guirguis E, Richardson J, Kuhn T, et al. Treatment of severe alcohol withdrawal: a focus on adjunctive agents. J Pharm Technol. 2017;33(5):204-212.
Alcohol withdrawal syndrome (AWS) occurs when an individual who is alcohol-dependent suddenly stops or significantly reduces his/her alcohol intake.1 Symptoms of AWS, which can be fatal, include anxiety, restlessness, seizures, confusion, and delirium.1 Because benzodiazepines have been proven effective in improving symptoms of AWS, they are considered the first-line treatment, but they also carry the risk of abuse, psychomotor sedation, cognitive impairment, and interactions with alcohol.1
Non-benzodiazepine anticonvulsants (NBACs) such as valproic acid (VPA) and carbamazepine may offer benefit as alternatives or adjuncts to benzodiazepines.1 Many NBACs affect the functioning of glutamate and gamma-aminobutyric acid (GABA) neurotransmitters,2 which are particularly dysregulated in patients with AWS. Because NBACs help stabilize this imbalance, they may be useful for managing AWS and preventing relapse without the risks associated with benzodiazepines.2
Valproic acid: A better choice than carbamazepine
Compared with other NBACs, VPA and carbamazepine have been studied more extensively for treating patients with AWS, and their clinical effectiveness has been well documented.1 For mild-to-moderate AWS, VPA and carbamazepine may be as effective as benzodiazepines in reducing the severity of symptoms, and more potent for preventing withdrawal seizures.1
Increasing data suggests that compared with VPA, adjunctive treatment with carbamazepine for AWS may be more frequently associated with intolerable adverse effects such as ataxia, orthostatic hypotension, vertigo, nausea, and vomiting.1 The rapid onset of AWS requires rapid-acting pharmacotherapy.1 In attempting to rapidly achieve the desired plasma concentrations of carbamazepine, clinicians may risk inducing adverse effects. Compared with carbamazepine, VPA is associated with faster symptom resolution, a shorter duration of pharmacologic treatment, fewer transfers to the ICU, fewer withdrawal seizures, and a more favorable adverse effect profile.1 Likely due to its shorter half-life, VPA delivers its therapeutic effects without producing significant adverse effects.1
Early and aggressive treatment of AWS is needed to block kindling,3 which is characterized by the worsening of withdrawal symptoms each time an individual attempts to quit drinking alcohol. Compared with carbamazepine, VPA may be more helpful for blocking kindling.3
More data is needed
Due to ethical concerns, few studies have compared anticonvulsant medications with placebo for treating AWS.2 Most studies examining NBACs for AWS have either used the anticonvulsant as an adjunct to a benzodiazepine to examine improvement in withdrawal symptoms, or compared the anticonvulsant with placebo or another intervention to assess the amount of a benzodiazepine required for safe detoxification.2,4 Additionally, most studies examining NBACs have been short, and few followed patients after the active medication period, which limits our knowledge of the long-term effectiveness of NBACs.2 Before NBACs can replace benzodiazepines for managing AWS, further evidence from clinical trials is needed to assess their efficiency as a stand-alone treatment.
Alcohol withdrawal syndrome (AWS) occurs when an individual who is alcohol-dependent suddenly stops or significantly reduces his/her alcohol intake.1 Symptoms of AWS, which can be fatal, include anxiety, restlessness, seizures, confusion, and delirium.1 Because benzodiazepines have been proven effective in improving symptoms of AWS, they are considered the first-line treatment, but they also carry the risk of abuse, psychomotor sedation, cognitive impairment, and interactions with alcohol.1
Non-benzodiazepine anticonvulsants (NBACs) such as valproic acid (VPA) and carbamazepine may offer benefit as alternatives or adjuncts to benzodiazepines.1 Many NBACs affect the functioning of glutamate and gamma-aminobutyric acid (GABA) neurotransmitters,2 which are particularly dysregulated in patients with AWS. Because NBACs help stabilize this imbalance, they may be useful for managing AWS and preventing relapse without the risks associated with benzodiazepines.2
Valproic acid: A better choice than carbamazepine
Compared with other NBACs, VPA and carbamazepine have been studied more extensively for treating patients with AWS, and their clinical effectiveness has been well documented.1 For mild-to-moderate AWS, VPA and carbamazepine may be as effective as benzodiazepines in reducing the severity of symptoms, and more potent for preventing withdrawal seizures.1
Increasing data suggests that compared with VPA, adjunctive treatment with carbamazepine for AWS may be more frequently associated with intolerable adverse effects such as ataxia, orthostatic hypotension, vertigo, nausea, and vomiting.1 The rapid onset of AWS requires rapid-acting pharmacotherapy.1 In attempting to rapidly achieve the desired plasma concentrations of carbamazepine, clinicians may risk inducing adverse effects. Compared with carbamazepine, VPA is associated with faster symptom resolution, a shorter duration of pharmacologic treatment, fewer transfers to the ICU, fewer withdrawal seizures, and a more favorable adverse effect profile.1 Likely due to its shorter half-life, VPA delivers its therapeutic effects without producing significant adverse effects.1
Early and aggressive treatment of AWS is needed to block kindling,3 which is characterized by the worsening of withdrawal symptoms each time an individual attempts to quit drinking alcohol. Compared with carbamazepine, VPA may be more helpful for blocking kindling.3
More data is needed
Due to ethical concerns, few studies have compared anticonvulsant medications with placebo for treating AWS.2 Most studies examining NBACs for AWS have either used the anticonvulsant as an adjunct to a benzodiazepine to examine improvement in withdrawal symptoms, or compared the anticonvulsant with placebo or another intervention to assess the amount of a benzodiazepine required for safe detoxification.2,4 Additionally, most studies examining NBACs have been short, and few followed patients after the active medication period, which limits our knowledge of the long-term effectiveness of NBACs.2 Before NBACs can replace benzodiazepines for managing AWS, further evidence from clinical trials is needed to assess their efficiency as a stand-alone treatment.
1. Maldonado JR. Novel algorithms for the prophylaxis and management of alcohol withdrawal syndromes–beyond benzodiazepines. Crit Care Clin. 2017;33(3):559-599.
2. Hammond CJ, Niciu MJ, Drew S, et al. Anticonvulsants for the treatment of alcohol withdrawal syndrome and alcohol use disorders. CNS Drugs. 2015;29(4):293-311.
3. Eyer F, Schreckenberg M, Hecht D, et al. Carbamazepine and valproate as adjuncts in the treatment of alcohol withdrawal syndrome: a retrospective cohort study. Alcohol Alcohol. 2011;46(2):177-184.
4. Guirguis E, Richardson J, Kuhn T, et al. Treatment of severe alcohol withdrawal: a focus on adjunctive agents. J Pharm Technol. 2017;33(5):204-212.
1. Maldonado JR. Novel algorithms for the prophylaxis and management of alcohol withdrawal syndromes–beyond benzodiazepines. Crit Care Clin. 2017;33(3):559-599.
2. Hammond CJ, Niciu MJ, Drew S, et al. Anticonvulsants for the treatment of alcohol withdrawal syndrome and alcohol use disorders. CNS Drugs. 2015;29(4):293-311.
3. Eyer F, Schreckenberg M, Hecht D, et al. Carbamazepine and valproate as adjuncts in the treatment of alcohol withdrawal syndrome: a retrospective cohort study. Alcohol Alcohol. 2011;46(2):177-184.
4. Guirguis E, Richardson J, Kuhn T, et al. Treatment of severe alcohol withdrawal: a focus on adjunctive agents. J Pharm Technol. 2017;33(5):204-212.