User login
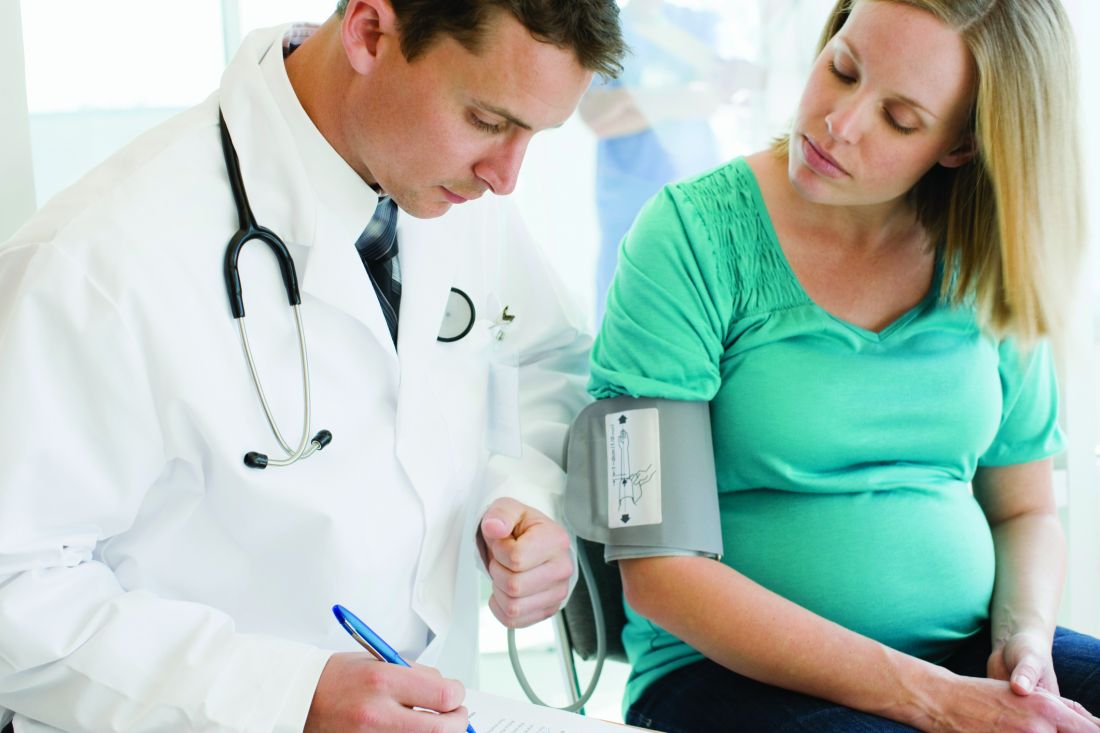
Teledermatology follow-up after Mohs surgery gets a thumbs up from patients
SEATTLE – The , according to new findings.
In addition, nearly all patients surveyed (91.4%) were willing to go through electronic follow-up again.
“A big takeaway from our study is that streamlining this process is really essential for successful implementation,” said study author Laura Rezac, MD, a PGY IV dermatology resident at the University of Mississippi, Jackson. “This study demonstrated the flexibility and convenience for both patients and surgeons and can serve as a prototype for future innovation.”
The study results were presented at the annual meeting of the American College of Mohs Surgery.
The role of telehealth has rapidly expanded over the past decade, with its use accelerating during the COVID-19 pandemic and transforming into an indispensable resource. It can be synchronous, Dr. Rezac explained, which is when telehealth happens in live, real-time settings where the patient interacts with a clinician. This usually occurs via phone or video, and providers and patients communicate directly.
Conversely, asynchronous telehealth, also known as “store-and-forward,” is often used for patient intake or follow-up care. For example, in dermatology, a patient can send a photo of a skin condition that is then reviewed by a dermatologist later.
“A pilot survey regarding the adoption of telemedicine in Mohs surgery found that, although most dermatologic surgeons felt that it can play a role, most said that they didn’t plan on using it after the pandemic,” said Dr. Rezac.
The survey, which was reported by this news organization, found that 80% of surveyed surgeons said that they turned to telemedicine during the pandemic, compared with just 23% who relied on the technology prior to the pandemic.
There were numerous perceived barriers to the use of telemedicine, and the one most commonly cited was the uncertainty of how telemedicine fits in the workflow of clinical practice. Other limitations reported were for physical exams (88%), patient response and training (57%), reimbursement concerns (50%), implementation of the technology (37%), regulations such as HIPAA (24%), training of staff (17%), and licensing (8%).
“The survey did identify one key use of telemedicine in Mohs and that was for [postoperative] visits,” she said. “But thus far, a postoperative evaluation after Mohs via an integrated asynchronous ‘store and forward’ teledermatology platform has not yet been evaluated.”
In the study, Dr. Rezac and colleagues sought to evaluate feasibility and efficacy, as well as patient attitudes, using a telemedicine platform for postoperative follow-up. A total of 163 patients who were treated with Mohs at a single academic institution during the 9-month study period (December 2021 through August 2022) responded to a survey and elected to participate in postoperative follow-up using telemedicine.
Dr. Rezac explained how their procedure was implemented for the patient. “On the day of the follow-up, the patient receives a text with a link that takes them to the MyChart website or app on their phone,” she said. “Once they log in, they see that they have a message telling them that they have a teledermatology message waiting for them. When they view it, they are taken to the curated message with instructions and a phone call if they need assistance, and then at the bottom, it shows they have a task to complete, which is the questionnaire.”
The patient will then be prompted to upload photos, which can be taken with their phone camera. The next step is to answer questions regarding the surgical site or pain concerns, and finally, patients are asked to respond to a few short questions about this type of follow-up. Once submitted, then they wait to be contacted by the surgeon.
On the surgeon’s side, these answers come into their EPIC inbox, and they can respond via a MyChart message.
Patient response was overwhelmingly positive, Dr. Rezac noted. Of the patients, 80.4% found the electronic surgery follow-up process to be “easy” or “very easy,” while only 4% found it “difficult” or “very difficult,” she said. “Also, 75.5% preferred electronic follow-up while 17.2% preferred in-person follow-up.”
There were limitations to this study, primarily that the asynchronous method does reduce live interaction, which could be an issue, depending on person’s needs, she pointed out. “But it is easy to schedule a phone call or video call or office visit.”
“The universal barrier is how to adopt it into the workflow, which includes training of staff,” she continued, “But this was a very streamlined process and gave very detailed instructions to the staff. Additionally, widespread use is limited to dermatological proficiency and access, and patients have to be amenable to it, so there is a selection bias since these patients chose to participate.”
Asked to comment on the study, Vishal Patel, MD, director of cutaneous oncology at George Washington University in Washington, said: “The COVID pandemic changed how practices and providers considered follow-up visits for small routine matters. Postoperative visits are often simple and do not require an in-depth, in-person evaluation.” Dr. Patel was not involved with this research.
“This study highlights the comfort of the vast majority of patients to have follow-up postoperative visits conducted via teledermatology – an approach that can help cut overall costs and also increase access for patients who are more in need of in-office care,” he added.
No external funding of the study was reported. Dr. Rezac reported no relevant financial relationships. Dr. Patel is a consultant for Sanofi, Regeneron, and Almirall.
A version of this article originally appeared on Medscape.com.
SEATTLE – The , according to new findings.
In addition, nearly all patients surveyed (91.4%) were willing to go through electronic follow-up again.
“A big takeaway from our study is that streamlining this process is really essential for successful implementation,” said study author Laura Rezac, MD, a PGY IV dermatology resident at the University of Mississippi, Jackson. “This study demonstrated the flexibility and convenience for both patients and surgeons and can serve as a prototype for future innovation.”
The study results were presented at the annual meeting of the American College of Mohs Surgery.
The role of telehealth has rapidly expanded over the past decade, with its use accelerating during the COVID-19 pandemic and transforming into an indispensable resource. It can be synchronous, Dr. Rezac explained, which is when telehealth happens in live, real-time settings where the patient interacts with a clinician. This usually occurs via phone or video, and providers and patients communicate directly.
Conversely, asynchronous telehealth, also known as “store-and-forward,” is often used for patient intake or follow-up care. For example, in dermatology, a patient can send a photo of a skin condition that is then reviewed by a dermatologist later.
“A pilot survey regarding the adoption of telemedicine in Mohs surgery found that, although most dermatologic surgeons felt that it can play a role, most said that they didn’t plan on using it after the pandemic,” said Dr. Rezac.
The survey, which was reported by this news organization, found that 80% of surveyed surgeons said that they turned to telemedicine during the pandemic, compared with just 23% who relied on the technology prior to the pandemic.
There were numerous perceived barriers to the use of telemedicine, and the one most commonly cited was the uncertainty of how telemedicine fits in the workflow of clinical practice. Other limitations reported were for physical exams (88%), patient response and training (57%), reimbursement concerns (50%), implementation of the technology (37%), regulations such as HIPAA (24%), training of staff (17%), and licensing (8%).
“The survey did identify one key use of telemedicine in Mohs and that was for [postoperative] visits,” she said. “But thus far, a postoperative evaluation after Mohs via an integrated asynchronous ‘store and forward’ teledermatology platform has not yet been evaluated.”
In the study, Dr. Rezac and colleagues sought to evaluate feasibility and efficacy, as well as patient attitudes, using a telemedicine platform for postoperative follow-up. A total of 163 patients who were treated with Mohs at a single academic institution during the 9-month study period (December 2021 through August 2022) responded to a survey and elected to participate in postoperative follow-up using telemedicine.
Dr. Rezac explained how their procedure was implemented for the patient. “On the day of the follow-up, the patient receives a text with a link that takes them to the MyChart website or app on their phone,” she said. “Once they log in, they see that they have a message telling them that they have a teledermatology message waiting for them. When they view it, they are taken to the curated message with instructions and a phone call if they need assistance, and then at the bottom, it shows they have a task to complete, which is the questionnaire.”
The patient will then be prompted to upload photos, which can be taken with their phone camera. The next step is to answer questions regarding the surgical site or pain concerns, and finally, patients are asked to respond to a few short questions about this type of follow-up. Once submitted, then they wait to be contacted by the surgeon.
On the surgeon’s side, these answers come into their EPIC inbox, and they can respond via a MyChart message.
Patient response was overwhelmingly positive, Dr. Rezac noted. Of the patients, 80.4% found the electronic surgery follow-up process to be “easy” or “very easy,” while only 4% found it “difficult” or “very difficult,” she said. “Also, 75.5% preferred electronic follow-up while 17.2% preferred in-person follow-up.”
There were limitations to this study, primarily that the asynchronous method does reduce live interaction, which could be an issue, depending on person’s needs, she pointed out. “But it is easy to schedule a phone call or video call or office visit.”
“The universal barrier is how to adopt it into the workflow, which includes training of staff,” she continued, “But this was a very streamlined process and gave very detailed instructions to the staff. Additionally, widespread use is limited to dermatological proficiency and access, and patients have to be amenable to it, so there is a selection bias since these patients chose to participate.”
Asked to comment on the study, Vishal Patel, MD, director of cutaneous oncology at George Washington University in Washington, said: “The COVID pandemic changed how practices and providers considered follow-up visits for small routine matters. Postoperative visits are often simple and do not require an in-depth, in-person evaluation.” Dr. Patel was not involved with this research.
“This study highlights the comfort of the vast majority of patients to have follow-up postoperative visits conducted via teledermatology – an approach that can help cut overall costs and also increase access for patients who are more in need of in-office care,” he added.
No external funding of the study was reported. Dr. Rezac reported no relevant financial relationships. Dr. Patel is a consultant for Sanofi, Regeneron, and Almirall.
A version of this article originally appeared on Medscape.com.
SEATTLE – The , according to new findings.
In addition, nearly all patients surveyed (91.4%) were willing to go through electronic follow-up again.
“A big takeaway from our study is that streamlining this process is really essential for successful implementation,” said study author Laura Rezac, MD, a PGY IV dermatology resident at the University of Mississippi, Jackson. “This study demonstrated the flexibility and convenience for both patients and surgeons and can serve as a prototype for future innovation.”
The study results were presented at the annual meeting of the American College of Mohs Surgery.
The role of telehealth has rapidly expanded over the past decade, with its use accelerating during the COVID-19 pandemic and transforming into an indispensable resource. It can be synchronous, Dr. Rezac explained, which is when telehealth happens in live, real-time settings where the patient interacts with a clinician. This usually occurs via phone or video, and providers and patients communicate directly.
Conversely, asynchronous telehealth, also known as “store-and-forward,” is often used for patient intake or follow-up care. For example, in dermatology, a patient can send a photo of a skin condition that is then reviewed by a dermatologist later.
“A pilot survey regarding the adoption of telemedicine in Mohs surgery found that, although most dermatologic surgeons felt that it can play a role, most said that they didn’t plan on using it after the pandemic,” said Dr. Rezac.
The survey, which was reported by this news organization, found that 80% of surveyed surgeons said that they turned to telemedicine during the pandemic, compared with just 23% who relied on the technology prior to the pandemic.
There were numerous perceived barriers to the use of telemedicine, and the one most commonly cited was the uncertainty of how telemedicine fits in the workflow of clinical practice. Other limitations reported were for physical exams (88%), patient response and training (57%), reimbursement concerns (50%), implementation of the technology (37%), regulations such as HIPAA (24%), training of staff (17%), and licensing (8%).
“The survey did identify one key use of telemedicine in Mohs and that was for [postoperative] visits,” she said. “But thus far, a postoperative evaluation after Mohs via an integrated asynchronous ‘store and forward’ teledermatology platform has not yet been evaluated.”
In the study, Dr. Rezac and colleagues sought to evaluate feasibility and efficacy, as well as patient attitudes, using a telemedicine platform for postoperative follow-up. A total of 163 patients who were treated with Mohs at a single academic institution during the 9-month study period (December 2021 through August 2022) responded to a survey and elected to participate in postoperative follow-up using telemedicine.
Dr. Rezac explained how their procedure was implemented for the patient. “On the day of the follow-up, the patient receives a text with a link that takes them to the MyChart website or app on their phone,” she said. “Once they log in, they see that they have a message telling them that they have a teledermatology message waiting for them. When they view it, they are taken to the curated message with instructions and a phone call if they need assistance, and then at the bottom, it shows they have a task to complete, which is the questionnaire.”
The patient will then be prompted to upload photos, which can be taken with their phone camera. The next step is to answer questions regarding the surgical site or pain concerns, and finally, patients are asked to respond to a few short questions about this type of follow-up. Once submitted, then they wait to be contacted by the surgeon.
On the surgeon’s side, these answers come into their EPIC inbox, and they can respond via a MyChart message.
Patient response was overwhelmingly positive, Dr. Rezac noted. Of the patients, 80.4% found the electronic surgery follow-up process to be “easy” or “very easy,” while only 4% found it “difficult” or “very difficult,” she said. “Also, 75.5% preferred electronic follow-up while 17.2% preferred in-person follow-up.”
There were limitations to this study, primarily that the asynchronous method does reduce live interaction, which could be an issue, depending on person’s needs, she pointed out. “But it is easy to schedule a phone call or video call or office visit.”
“The universal barrier is how to adopt it into the workflow, which includes training of staff,” she continued, “But this was a very streamlined process and gave very detailed instructions to the staff. Additionally, widespread use is limited to dermatological proficiency and access, and patients have to be amenable to it, so there is a selection bias since these patients chose to participate.”
Asked to comment on the study, Vishal Patel, MD, director of cutaneous oncology at George Washington University in Washington, said: “The COVID pandemic changed how practices and providers considered follow-up visits for small routine matters. Postoperative visits are often simple and do not require an in-depth, in-person evaluation.” Dr. Patel was not involved with this research.
“This study highlights the comfort of the vast majority of patients to have follow-up postoperative visits conducted via teledermatology – an approach that can help cut overall costs and also increase access for patients who are more in need of in-office care,” he added.
No external funding of the study was reported. Dr. Rezac reported no relevant financial relationships. Dr. Patel is a consultant for Sanofi, Regeneron, and Almirall.
A version of this article originally appeared on Medscape.com.
AT ACMS 2023
ASCO honors Hagop Kantarjian, MD, for leukemia research
This award is the society’s “highest scientific honor, and I am extremely happy and honored to receive it,” Dr. Kantarjian commented in an interview with this news organization.
Dr. Kantarjian serves as the chair of the department of leukemia and currently holds the Samsung Distinguished University Chair in Cancer Medicine at the University of Texas MD Anderson Cancer Center, Houston.
“No doubt that this is not an individual award. It represents an award for the accomplishments of all the leukemia faculty at MD Anderson across 4 decades. It’s really a teamwork effort that led to so many discoveries and improvements in treatment and care of patients with leukemia,” he commented.
The David A. Karnofsky Memorial Award has been presented annually since 1970 to recognize oncologists who have made outstanding contributions to cancer research, diagnosis, or treatment, ASCO noted.
From Lebanon to Texas
Dr. Kantarjian received his medical degree from the American University of Beirut, in Lebanon, in 1979 and completed his residency in internal medicine at the same institution in 1981.
It was his experience at MD Anderson as a young medical student and later as a fellow that fueled his interest and career in leukemia, he said.
“In 1978, I took a 4-month elective at MD Anderson, and I soon realized how different and innovative the atmosphere at MD Anderson was, compared to where I was training in Lebanon,” Dr. Kantarjian told this news organization.
Working with mentors that included MD Anderson heavyweights Emil Freireich, MD, Kenneth McCredie, MD, and Michael Keating, MD, helped shape his career and guide his leukemia research, he said.
Transformative impact on leukemia outcomes
The award citation notes that over the past 4 decades, Dr. Kantarjian’s research has transformed some standards of care and has dramatically improved survival in several leukemia subtypes, including chronic myeloid leukemia (CML), acute myeloid leukemia (AML), myelodysplastic syndrome (MDS), and acute lymphocytic leukemia (ALL).
“Four decades ago, most of the leukemias were incurable. Today, most of the leukemias are potentially curable with targeted therapies. That’s what I am most proud of,” Dr. Kantarjian told this news organization.
Among Dr. Kantarjian’s contributions to the field of leukemia:
- Developing the HYPER-CVAD regimen (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone) as a standard-of-care, frontline therapy for adults with ALL.
- Establishing clinical biology parameters of CML, including definitions of CML phases and cytogenetic responses, and establishing new prognostic factors that were subsequently adopted in studies of tyrosine kinase inhibitors.
- Leading the development of decitabine and epigenetic hypomethylation therapy for MDS and for older/unfit patients with AML.
- Pioneering research with hypomethylating agents (HMAs) in combination with venetoclax, which led to FDA approval of HMA-venetoclax combinations for older/unfit patients with AML.
- Championing the development of clofarabine, conducting animal toxicology studies, and leading subsequent phase 1 and 2 trials and pivotal phase 3 and 4 trials that led to FDA approval of clofarabine for pediatric ALL.
- Developing several FLT3 inhibitors, isocitrate dehydrogenase inhibitors, and venetoclax, which all received FDA approval for the treatment of AML and its subsets.
- Developing regimens for inotuzumab and blinatumomab combined with chemotherapy for adults with pre-B ALL.
- Working on the development of imatinib, dasatinib, nilotinib, bosutinib, ponatinib, and omacetaxine, which all received FDA approval for CML therapy.
“Dr. Kantarjian’s long list of accomplishments and groundbreaking discoveries are a testament to his lifelong commitment to impactful cancer research and patient care,” Giulio Draetta, MD, PhD, chief scientific officer at MD Anderson, said in a statement.
Giving back
Dr. Kantarjian has written more than 2,200 peer-reviewed articles and more than 100 book chapters. In 2012, he cofounded the Society of Hematologic Oncology, which has now expanded worldwide.
He has served on multiple ASCO committees throughout the years and served on the ASCO board of directors from 2010 to 2015.
Dr. Kantarjian is passionately involved in mentoring and education. In 2000 he created the MD Anderson Leukemia Fellowship, which now trains about 10 fellows in leukemia annually.
He is a nonresident fellow in health care at the Rice Baker Institute and has written extensively on important health care issues in cancer, including the importance of universal equitable health care, health care safety nets, health care as a human right, and the problem of drug shortages.
Dr. Kantarjian is a strong advocate for more affordable drug therapies. For years he has been outspoken about the high price of leukemia drugs and has written high-profile articles in medical journals. He has even appeared on a popular television program to publicize the issue.
“Drug costs have been increasing over time. If you think about it, even if you discover a drug that cures cancer, but the drug is affordable for the 1% of the patients, then you have no cure for cancer,” Dr. Kantarjian told this news organization.
“I started speaking about the issue of the cancer drug costs in 2012. Unfortunately, we have not made progress simply because of the for-profit nature of health care and the strong lobbying by drug companies,” he added. Dr. Kantarjian hopes new legislation will eventually turn the tide.
Dr. Kantarjian has received many other honors throughout his distinguished career, including the American Lebanese Medical Association’s Lifetime Achievement Award, the American Association for Cancer Research’s Joseph H. Burchenal Memorial Award, and the Leukemia Society of America’s Outstanding Service to Mankind Award. He also was named an ASCO Fellow and a Leukemia Society of America Special Fellow and Scholar.
Dr. Kantarjian will be presented with the 2023 David A. Karnofsky Memorial Award, which includes a $25,000 honorarium, and will give a scientific lecture about his research at the ASCO annual meeting in Chicago in early June.
A version of this article originally appeared on Medscape.com.
This award is the society’s “highest scientific honor, and I am extremely happy and honored to receive it,” Dr. Kantarjian commented in an interview with this news organization.
Dr. Kantarjian serves as the chair of the department of leukemia and currently holds the Samsung Distinguished University Chair in Cancer Medicine at the University of Texas MD Anderson Cancer Center, Houston.
“No doubt that this is not an individual award. It represents an award for the accomplishments of all the leukemia faculty at MD Anderson across 4 decades. It’s really a teamwork effort that led to so many discoveries and improvements in treatment and care of patients with leukemia,” he commented.
The David A. Karnofsky Memorial Award has been presented annually since 1970 to recognize oncologists who have made outstanding contributions to cancer research, diagnosis, or treatment, ASCO noted.
From Lebanon to Texas
Dr. Kantarjian received his medical degree from the American University of Beirut, in Lebanon, in 1979 and completed his residency in internal medicine at the same institution in 1981.
It was his experience at MD Anderson as a young medical student and later as a fellow that fueled his interest and career in leukemia, he said.
“In 1978, I took a 4-month elective at MD Anderson, and I soon realized how different and innovative the atmosphere at MD Anderson was, compared to where I was training in Lebanon,” Dr. Kantarjian told this news organization.
Working with mentors that included MD Anderson heavyweights Emil Freireich, MD, Kenneth McCredie, MD, and Michael Keating, MD, helped shape his career and guide his leukemia research, he said.
Transformative impact on leukemia outcomes
The award citation notes that over the past 4 decades, Dr. Kantarjian’s research has transformed some standards of care and has dramatically improved survival in several leukemia subtypes, including chronic myeloid leukemia (CML), acute myeloid leukemia (AML), myelodysplastic syndrome (MDS), and acute lymphocytic leukemia (ALL).
“Four decades ago, most of the leukemias were incurable. Today, most of the leukemias are potentially curable with targeted therapies. That’s what I am most proud of,” Dr. Kantarjian told this news organization.
Among Dr. Kantarjian’s contributions to the field of leukemia:
- Developing the HYPER-CVAD regimen (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone) as a standard-of-care, frontline therapy for adults with ALL.
- Establishing clinical biology parameters of CML, including definitions of CML phases and cytogenetic responses, and establishing new prognostic factors that were subsequently adopted in studies of tyrosine kinase inhibitors.
- Leading the development of decitabine and epigenetic hypomethylation therapy for MDS and for older/unfit patients with AML.
- Pioneering research with hypomethylating agents (HMAs) in combination with venetoclax, which led to FDA approval of HMA-venetoclax combinations for older/unfit patients with AML.
- Championing the development of clofarabine, conducting animal toxicology studies, and leading subsequent phase 1 and 2 trials and pivotal phase 3 and 4 trials that led to FDA approval of clofarabine for pediatric ALL.
- Developing several FLT3 inhibitors, isocitrate dehydrogenase inhibitors, and venetoclax, which all received FDA approval for the treatment of AML and its subsets.
- Developing regimens for inotuzumab and blinatumomab combined with chemotherapy for adults with pre-B ALL.
- Working on the development of imatinib, dasatinib, nilotinib, bosutinib, ponatinib, and omacetaxine, which all received FDA approval for CML therapy.
“Dr. Kantarjian’s long list of accomplishments and groundbreaking discoveries are a testament to his lifelong commitment to impactful cancer research and patient care,” Giulio Draetta, MD, PhD, chief scientific officer at MD Anderson, said in a statement.
Giving back
Dr. Kantarjian has written more than 2,200 peer-reviewed articles and more than 100 book chapters. In 2012, he cofounded the Society of Hematologic Oncology, which has now expanded worldwide.
He has served on multiple ASCO committees throughout the years and served on the ASCO board of directors from 2010 to 2015.
Dr. Kantarjian is passionately involved in mentoring and education. In 2000 he created the MD Anderson Leukemia Fellowship, which now trains about 10 fellows in leukemia annually.
He is a nonresident fellow in health care at the Rice Baker Institute and has written extensively on important health care issues in cancer, including the importance of universal equitable health care, health care safety nets, health care as a human right, and the problem of drug shortages.
Dr. Kantarjian is a strong advocate for more affordable drug therapies. For years he has been outspoken about the high price of leukemia drugs and has written high-profile articles in medical journals. He has even appeared on a popular television program to publicize the issue.
“Drug costs have been increasing over time. If you think about it, even if you discover a drug that cures cancer, but the drug is affordable for the 1% of the patients, then you have no cure for cancer,” Dr. Kantarjian told this news organization.
“I started speaking about the issue of the cancer drug costs in 2012. Unfortunately, we have not made progress simply because of the for-profit nature of health care and the strong lobbying by drug companies,” he added. Dr. Kantarjian hopes new legislation will eventually turn the tide.
Dr. Kantarjian has received many other honors throughout his distinguished career, including the American Lebanese Medical Association’s Lifetime Achievement Award, the American Association for Cancer Research’s Joseph H. Burchenal Memorial Award, and the Leukemia Society of America’s Outstanding Service to Mankind Award. He also was named an ASCO Fellow and a Leukemia Society of America Special Fellow and Scholar.
Dr. Kantarjian will be presented with the 2023 David A. Karnofsky Memorial Award, which includes a $25,000 honorarium, and will give a scientific lecture about his research at the ASCO annual meeting in Chicago in early June.
A version of this article originally appeared on Medscape.com.
This award is the society’s “highest scientific honor, and I am extremely happy and honored to receive it,” Dr. Kantarjian commented in an interview with this news organization.
Dr. Kantarjian serves as the chair of the department of leukemia and currently holds the Samsung Distinguished University Chair in Cancer Medicine at the University of Texas MD Anderson Cancer Center, Houston.
“No doubt that this is not an individual award. It represents an award for the accomplishments of all the leukemia faculty at MD Anderson across 4 decades. It’s really a teamwork effort that led to so many discoveries and improvements in treatment and care of patients with leukemia,” he commented.
The David A. Karnofsky Memorial Award has been presented annually since 1970 to recognize oncologists who have made outstanding contributions to cancer research, diagnosis, or treatment, ASCO noted.
From Lebanon to Texas
Dr. Kantarjian received his medical degree from the American University of Beirut, in Lebanon, in 1979 and completed his residency in internal medicine at the same institution in 1981.
It was his experience at MD Anderson as a young medical student and later as a fellow that fueled his interest and career in leukemia, he said.
“In 1978, I took a 4-month elective at MD Anderson, and I soon realized how different and innovative the atmosphere at MD Anderson was, compared to where I was training in Lebanon,” Dr. Kantarjian told this news organization.
Working with mentors that included MD Anderson heavyweights Emil Freireich, MD, Kenneth McCredie, MD, and Michael Keating, MD, helped shape his career and guide his leukemia research, he said.
Transformative impact on leukemia outcomes
The award citation notes that over the past 4 decades, Dr. Kantarjian’s research has transformed some standards of care and has dramatically improved survival in several leukemia subtypes, including chronic myeloid leukemia (CML), acute myeloid leukemia (AML), myelodysplastic syndrome (MDS), and acute lymphocytic leukemia (ALL).
“Four decades ago, most of the leukemias were incurable. Today, most of the leukemias are potentially curable with targeted therapies. That’s what I am most proud of,” Dr. Kantarjian told this news organization.
Among Dr. Kantarjian’s contributions to the field of leukemia:
- Developing the HYPER-CVAD regimen (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone) as a standard-of-care, frontline therapy for adults with ALL.
- Establishing clinical biology parameters of CML, including definitions of CML phases and cytogenetic responses, and establishing new prognostic factors that were subsequently adopted in studies of tyrosine kinase inhibitors.
- Leading the development of decitabine and epigenetic hypomethylation therapy for MDS and for older/unfit patients with AML.
- Pioneering research with hypomethylating agents (HMAs) in combination with venetoclax, which led to FDA approval of HMA-venetoclax combinations for older/unfit patients with AML.
- Championing the development of clofarabine, conducting animal toxicology studies, and leading subsequent phase 1 and 2 trials and pivotal phase 3 and 4 trials that led to FDA approval of clofarabine for pediatric ALL.
- Developing several FLT3 inhibitors, isocitrate dehydrogenase inhibitors, and venetoclax, which all received FDA approval for the treatment of AML and its subsets.
- Developing regimens for inotuzumab and blinatumomab combined with chemotherapy for adults with pre-B ALL.
- Working on the development of imatinib, dasatinib, nilotinib, bosutinib, ponatinib, and omacetaxine, which all received FDA approval for CML therapy.
“Dr. Kantarjian’s long list of accomplishments and groundbreaking discoveries are a testament to his lifelong commitment to impactful cancer research and patient care,” Giulio Draetta, MD, PhD, chief scientific officer at MD Anderson, said in a statement.
Giving back
Dr. Kantarjian has written more than 2,200 peer-reviewed articles and more than 100 book chapters. In 2012, he cofounded the Society of Hematologic Oncology, which has now expanded worldwide.
He has served on multiple ASCO committees throughout the years and served on the ASCO board of directors from 2010 to 2015.
Dr. Kantarjian is passionately involved in mentoring and education. In 2000 he created the MD Anderson Leukemia Fellowship, which now trains about 10 fellows in leukemia annually.
He is a nonresident fellow in health care at the Rice Baker Institute and has written extensively on important health care issues in cancer, including the importance of universal equitable health care, health care safety nets, health care as a human right, and the problem of drug shortages.
Dr. Kantarjian is a strong advocate for more affordable drug therapies. For years he has been outspoken about the high price of leukemia drugs and has written high-profile articles in medical journals. He has even appeared on a popular television program to publicize the issue.
“Drug costs have been increasing over time. If you think about it, even if you discover a drug that cures cancer, but the drug is affordable for the 1% of the patients, then you have no cure for cancer,” Dr. Kantarjian told this news organization.
“I started speaking about the issue of the cancer drug costs in 2012. Unfortunately, we have not made progress simply because of the for-profit nature of health care and the strong lobbying by drug companies,” he added. Dr. Kantarjian hopes new legislation will eventually turn the tide.
Dr. Kantarjian has received many other honors throughout his distinguished career, including the American Lebanese Medical Association’s Lifetime Achievement Award, the American Association for Cancer Research’s Joseph H. Burchenal Memorial Award, and the Leukemia Society of America’s Outstanding Service to Mankind Award. He also was named an ASCO Fellow and a Leukemia Society of America Special Fellow and Scholar.
Dr. Kantarjian will be presented with the 2023 David A. Karnofsky Memorial Award, which includes a $25,000 honorarium, and will give a scientific lecture about his research at the ASCO annual meeting in Chicago in early June.
A version of this article originally appeared on Medscape.com.
Novel insights on Takotsubo syndrome could lead to new therapies
Takotsubo syndrome is a form of acute heart failure that mimics acute coronary syndromes, with troponin elevation and symptoms including chest pain and dyspnea, but without a culprit lesion on coronary angiography.
However, echocardiography shows the heart to be massively enlarged. The condition was named by Japanese researchers as the shape of the left ventricle resembles the Takotsubo fishing pot used to trap octopi.
The condition affects mainly older women and accounts for about 6% of female patients presenting with acute coronary syndrome symptoms. In around two-thirds of cases there is a triggering stress event which can be physical, such as an acute disease, or emotional, such as an argument or the sudden death of someone close, hence the term “broken heart syndrome.” The emotional stress triggering the syndrome can also be positive such as a birthday party or the birth of a grandchild.
“The mechanisms involved in Takotsubo syndrome are unknown. Because there is often a stress trigger it is believed that sympathetic activation causes a surge of catecholamine release, but that is not fully understood,” lead author of the current study, Thomas Stiermaier, MD, University Heart Center Lübeck (Germany), explained in an interview.
“We wanted to look more closely at the hemodynamic effects in the hearts of patients with Takotsubo syndrome to see if we could identify novel mechanisms contributing to the condition,” he added.
The aptly named Optimized Characterization of Takotsubo Syndrome by Obtaining Pressure Volume Loops (OCTOPUS) study was published online in the Journal of the American College of Cardiology.
For the study, the researchers used a conductance catheter inserted into the left ventricle of the heart to analyze pressure-volume relationships in 24 consecutive patients with Takotsubo syndrome and a control population of 20 participants without cardiovascular disease.
These pressure-volume loops are “the gold standard for direct, real-time assessment of systolic and diastolic cardiac function independent of loading conditions,” and “provide in-depth information regarding ventricular-arterial coupling and cardiac energetics and efficiency,” the authors wrote.
“These parameters comprise a considerable amount of information on cardiac performance and help to advance our understanding of cardiac physiology and its pathophysiological role in various conditions,” they noted, adding that this is believed to be the first comprehensive hemodynamic analysis in patients with Takotsubo syndrome using such invasive tracing of pressure-volume loops.
Results showed that Takotsubo syndrome is associated with a severely impaired cardiac contractility and a shortened systolic period. In response, the heart compensates by increasing left ventricular end diastolic volume to preserve the stroke volume.
Diastolic function is characterized by prolonged active relaxation but unaltered passive elastic properties. The analysis of myocardial energetics revealed an inefficient system with increased potential and decreased kinetic energy (stroke work).
“These are new and important findings,” Dr. Stiermaier said, adding that these hemodynamic changes give clues as to the underlying mechanisms at play in Takotsubo syndrome, as well as possible treatment strategies that could be investigated.
“Taking all this information together, we believe that it is likely that decreased phosphorylation of myofilament proteins – which may be caused by some kind of disturbance in calcium metabolism – may partially account for the impaired contractility and shortened systolic period seen in Takotsubo syndrome,” he commented.
The researchers suggested that Takotsubo syndrome may therefore be treated with medications such as omecamtiv (a drug that increases systolic duration) or the calcium sensitizer levosimendan, which improves contractility, possibly in combination with beta-blockers to protect against the intense adrenergic activation.
They noted that several studies have reported the use of levosimendan in Takotsubo syndrome and have suggested positive effects by accelerating recovery of ventricular function. But they added that prospective data are lacking, and, to their knowledge, omecamtiv has not been tested in Takotsubo syndrome.
“We need to clearly identify the mechanism involved in these changes at the cellular level, and then test these medications to see if they can help prevent or reverse the hemodynamic changes seen in Takotsubo syndrome,” Dr. Stiermaier said.
He explained that the contractile abnormalities in Takotsubo syndrome are transient and generally normalize after a few weeks or months, but while systolic function may appear normal in the long term there are other more subtle changes that can persist, and these patients have an increased rate of cardiovascular events, compared with the healthy population over the long term.
However, because Takotsubo syndrome patients generally have a high rate of other comorbidities, it is not known whether their increased event rate is caused by the syndrome or by these other comorbidities.
While some patients with Takotsubo syndrome have a mild disease course and a good prognosis, others have more complications, with around 10%-15% going on to develop severe disease with cardiogenic shock or pleural effusion, Dr. Stiermaier noted.
“These patients have a bad prognosis. Our aim is to try to identify the patients who are at high risk of these complications and treat them early to prevent cardiogenic shock and pleural effusion from developing,” he said. “We are hopeful that by identifying the hemodynamic changes occurring in Takotsubo syndrome we can figure out the mechanisms involved and give medications in the acute setting to prevent the complications that can arise down the road.”
Mechanisms ‘appealing but speculative’
In an editorial (J Am Coll Cardiol. 2023 May;81[20]:1992-5), Jorge Salamanca, MD, and Fernando Alfonso, MD, Hospital Universitario de La Princesa, Madrid, described this new study as “an important piece of research, providing a careful, systematic, and comprehensive set of sophisticated invasive hemodynamic data that shed new light on our understanding of this unique clinical entity.”
They said the researchers have provided “robust data on the acute hemodynamic behavior of the left ventricle in patients with Takotsubo syndrome that clearly advance the field but also raise new questions.”
But the editorialists cautioned that the hypotheses of the potential mechanisms linking a molecular basis for the metabolic dysregulation, resulting in increased potential energy coupled with a decreased kinetic energy, “are appealing but largely speculative.”
“Whether these hemodynamic findings could be the foundation and would support the use of novel and attractive drugs in Takotsubo syndrome, remains unsettled and can only be considered as hypothesis generating,” they wrote.
“Further studies are required to elucidate factors associated with a more severe hemodynamic derangement and to devise therapeutic strategies helping to rapidly restore an efficient left ventricular function in these challenging patients,” they concluded.
The study authors and the editorialists reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Takotsubo syndrome is a form of acute heart failure that mimics acute coronary syndromes, with troponin elevation and symptoms including chest pain and dyspnea, but without a culprit lesion on coronary angiography.
However, echocardiography shows the heart to be massively enlarged. The condition was named by Japanese researchers as the shape of the left ventricle resembles the Takotsubo fishing pot used to trap octopi.
The condition affects mainly older women and accounts for about 6% of female patients presenting with acute coronary syndrome symptoms. In around two-thirds of cases there is a triggering stress event which can be physical, such as an acute disease, or emotional, such as an argument or the sudden death of someone close, hence the term “broken heart syndrome.” The emotional stress triggering the syndrome can also be positive such as a birthday party or the birth of a grandchild.
“The mechanisms involved in Takotsubo syndrome are unknown. Because there is often a stress trigger it is believed that sympathetic activation causes a surge of catecholamine release, but that is not fully understood,” lead author of the current study, Thomas Stiermaier, MD, University Heart Center Lübeck (Germany), explained in an interview.
“We wanted to look more closely at the hemodynamic effects in the hearts of patients with Takotsubo syndrome to see if we could identify novel mechanisms contributing to the condition,” he added.
The aptly named Optimized Characterization of Takotsubo Syndrome by Obtaining Pressure Volume Loops (OCTOPUS) study was published online in the Journal of the American College of Cardiology.
For the study, the researchers used a conductance catheter inserted into the left ventricle of the heart to analyze pressure-volume relationships in 24 consecutive patients with Takotsubo syndrome and a control population of 20 participants without cardiovascular disease.
These pressure-volume loops are “the gold standard for direct, real-time assessment of systolic and diastolic cardiac function independent of loading conditions,” and “provide in-depth information regarding ventricular-arterial coupling and cardiac energetics and efficiency,” the authors wrote.
“These parameters comprise a considerable amount of information on cardiac performance and help to advance our understanding of cardiac physiology and its pathophysiological role in various conditions,” they noted, adding that this is believed to be the first comprehensive hemodynamic analysis in patients with Takotsubo syndrome using such invasive tracing of pressure-volume loops.
Results showed that Takotsubo syndrome is associated with a severely impaired cardiac contractility and a shortened systolic period. In response, the heart compensates by increasing left ventricular end diastolic volume to preserve the stroke volume.
Diastolic function is characterized by prolonged active relaxation but unaltered passive elastic properties. The analysis of myocardial energetics revealed an inefficient system with increased potential and decreased kinetic energy (stroke work).
“These are new and important findings,” Dr. Stiermaier said, adding that these hemodynamic changes give clues as to the underlying mechanisms at play in Takotsubo syndrome, as well as possible treatment strategies that could be investigated.
“Taking all this information together, we believe that it is likely that decreased phosphorylation of myofilament proteins – which may be caused by some kind of disturbance in calcium metabolism – may partially account for the impaired contractility and shortened systolic period seen in Takotsubo syndrome,” he commented.
The researchers suggested that Takotsubo syndrome may therefore be treated with medications such as omecamtiv (a drug that increases systolic duration) or the calcium sensitizer levosimendan, which improves contractility, possibly in combination with beta-blockers to protect against the intense adrenergic activation.
They noted that several studies have reported the use of levosimendan in Takotsubo syndrome and have suggested positive effects by accelerating recovery of ventricular function. But they added that prospective data are lacking, and, to their knowledge, omecamtiv has not been tested in Takotsubo syndrome.
“We need to clearly identify the mechanism involved in these changes at the cellular level, and then test these medications to see if they can help prevent or reverse the hemodynamic changes seen in Takotsubo syndrome,” Dr. Stiermaier said.
He explained that the contractile abnormalities in Takotsubo syndrome are transient and generally normalize after a few weeks or months, but while systolic function may appear normal in the long term there are other more subtle changes that can persist, and these patients have an increased rate of cardiovascular events, compared with the healthy population over the long term.
However, because Takotsubo syndrome patients generally have a high rate of other comorbidities, it is not known whether their increased event rate is caused by the syndrome or by these other comorbidities.
While some patients with Takotsubo syndrome have a mild disease course and a good prognosis, others have more complications, with around 10%-15% going on to develop severe disease with cardiogenic shock or pleural effusion, Dr. Stiermaier noted.
“These patients have a bad prognosis. Our aim is to try to identify the patients who are at high risk of these complications and treat them early to prevent cardiogenic shock and pleural effusion from developing,” he said. “We are hopeful that by identifying the hemodynamic changes occurring in Takotsubo syndrome we can figure out the mechanisms involved and give medications in the acute setting to prevent the complications that can arise down the road.”
Mechanisms ‘appealing but speculative’
In an editorial (J Am Coll Cardiol. 2023 May;81[20]:1992-5), Jorge Salamanca, MD, and Fernando Alfonso, MD, Hospital Universitario de La Princesa, Madrid, described this new study as “an important piece of research, providing a careful, systematic, and comprehensive set of sophisticated invasive hemodynamic data that shed new light on our understanding of this unique clinical entity.”
They said the researchers have provided “robust data on the acute hemodynamic behavior of the left ventricle in patients with Takotsubo syndrome that clearly advance the field but also raise new questions.”
But the editorialists cautioned that the hypotheses of the potential mechanisms linking a molecular basis for the metabolic dysregulation, resulting in increased potential energy coupled with a decreased kinetic energy, “are appealing but largely speculative.”
“Whether these hemodynamic findings could be the foundation and would support the use of novel and attractive drugs in Takotsubo syndrome, remains unsettled and can only be considered as hypothesis generating,” they wrote.
“Further studies are required to elucidate factors associated with a more severe hemodynamic derangement and to devise therapeutic strategies helping to rapidly restore an efficient left ventricular function in these challenging patients,” they concluded.
The study authors and the editorialists reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Takotsubo syndrome is a form of acute heart failure that mimics acute coronary syndromes, with troponin elevation and symptoms including chest pain and dyspnea, but without a culprit lesion on coronary angiography.
However, echocardiography shows the heart to be massively enlarged. The condition was named by Japanese researchers as the shape of the left ventricle resembles the Takotsubo fishing pot used to trap octopi.
The condition affects mainly older women and accounts for about 6% of female patients presenting with acute coronary syndrome symptoms. In around two-thirds of cases there is a triggering stress event which can be physical, such as an acute disease, or emotional, such as an argument or the sudden death of someone close, hence the term “broken heart syndrome.” The emotional stress triggering the syndrome can also be positive such as a birthday party or the birth of a grandchild.
“The mechanisms involved in Takotsubo syndrome are unknown. Because there is often a stress trigger it is believed that sympathetic activation causes a surge of catecholamine release, but that is not fully understood,” lead author of the current study, Thomas Stiermaier, MD, University Heart Center Lübeck (Germany), explained in an interview.
“We wanted to look more closely at the hemodynamic effects in the hearts of patients with Takotsubo syndrome to see if we could identify novel mechanisms contributing to the condition,” he added.
The aptly named Optimized Characterization of Takotsubo Syndrome by Obtaining Pressure Volume Loops (OCTOPUS) study was published online in the Journal of the American College of Cardiology.
For the study, the researchers used a conductance catheter inserted into the left ventricle of the heart to analyze pressure-volume relationships in 24 consecutive patients with Takotsubo syndrome and a control population of 20 participants without cardiovascular disease.
These pressure-volume loops are “the gold standard for direct, real-time assessment of systolic and diastolic cardiac function independent of loading conditions,” and “provide in-depth information regarding ventricular-arterial coupling and cardiac energetics and efficiency,” the authors wrote.
“These parameters comprise a considerable amount of information on cardiac performance and help to advance our understanding of cardiac physiology and its pathophysiological role in various conditions,” they noted, adding that this is believed to be the first comprehensive hemodynamic analysis in patients with Takotsubo syndrome using such invasive tracing of pressure-volume loops.
Results showed that Takotsubo syndrome is associated with a severely impaired cardiac contractility and a shortened systolic period. In response, the heart compensates by increasing left ventricular end diastolic volume to preserve the stroke volume.
Diastolic function is characterized by prolonged active relaxation but unaltered passive elastic properties. The analysis of myocardial energetics revealed an inefficient system with increased potential and decreased kinetic energy (stroke work).
“These are new and important findings,” Dr. Stiermaier said, adding that these hemodynamic changes give clues as to the underlying mechanisms at play in Takotsubo syndrome, as well as possible treatment strategies that could be investigated.
“Taking all this information together, we believe that it is likely that decreased phosphorylation of myofilament proteins – which may be caused by some kind of disturbance in calcium metabolism – may partially account for the impaired contractility and shortened systolic period seen in Takotsubo syndrome,” he commented.
The researchers suggested that Takotsubo syndrome may therefore be treated with medications such as omecamtiv (a drug that increases systolic duration) or the calcium sensitizer levosimendan, which improves contractility, possibly in combination with beta-blockers to protect against the intense adrenergic activation.
They noted that several studies have reported the use of levosimendan in Takotsubo syndrome and have suggested positive effects by accelerating recovery of ventricular function. But they added that prospective data are lacking, and, to their knowledge, omecamtiv has not been tested in Takotsubo syndrome.
“We need to clearly identify the mechanism involved in these changes at the cellular level, and then test these medications to see if they can help prevent or reverse the hemodynamic changes seen in Takotsubo syndrome,” Dr. Stiermaier said.
He explained that the contractile abnormalities in Takotsubo syndrome are transient and generally normalize after a few weeks or months, but while systolic function may appear normal in the long term there are other more subtle changes that can persist, and these patients have an increased rate of cardiovascular events, compared with the healthy population over the long term.
However, because Takotsubo syndrome patients generally have a high rate of other comorbidities, it is not known whether their increased event rate is caused by the syndrome or by these other comorbidities.
While some patients with Takotsubo syndrome have a mild disease course and a good prognosis, others have more complications, with around 10%-15% going on to develop severe disease with cardiogenic shock or pleural effusion, Dr. Stiermaier noted.
“These patients have a bad prognosis. Our aim is to try to identify the patients who are at high risk of these complications and treat them early to prevent cardiogenic shock and pleural effusion from developing,” he said. “We are hopeful that by identifying the hemodynamic changes occurring in Takotsubo syndrome we can figure out the mechanisms involved and give medications in the acute setting to prevent the complications that can arise down the road.”
Mechanisms ‘appealing but speculative’
In an editorial (J Am Coll Cardiol. 2023 May;81[20]:1992-5), Jorge Salamanca, MD, and Fernando Alfonso, MD, Hospital Universitario de La Princesa, Madrid, described this new study as “an important piece of research, providing a careful, systematic, and comprehensive set of sophisticated invasive hemodynamic data that shed new light on our understanding of this unique clinical entity.”
They said the researchers have provided “robust data on the acute hemodynamic behavior of the left ventricle in patients with Takotsubo syndrome that clearly advance the field but also raise new questions.”
But the editorialists cautioned that the hypotheses of the potential mechanisms linking a molecular basis for the metabolic dysregulation, resulting in increased potential energy coupled with a decreased kinetic energy, “are appealing but largely speculative.”
“Whether these hemodynamic findings could be the foundation and would support the use of novel and attractive drugs in Takotsubo syndrome, remains unsettled and can only be considered as hypothesis generating,” they wrote.
“Further studies are required to elucidate factors associated with a more severe hemodynamic derangement and to devise therapeutic strategies helping to rapidly restore an efficient left ventricular function in these challenging patients,” they concluded.
The study authors and the editorialists reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN JOURNAL OF CARDIOLOGY
Gestational HTN, preeclampsia worsen long-term risk for ischemic, nonischemic heart failure
, an observational study suggests.
The risks were most pronounced, jumping more than sixfold in the case of ischemic HF, during the first 6 years after the pregnancy. They then receded to plateau at a lower, still significantly elevated level of risk that persisted even years later, in the analysis of women in a Swedish medical birth registry.
The case-matching study compared women with no history of cardiovascular (CV) disease and a first successful pregnancy during which they either developed or did not experience gestational hypertension or preeclampsia.
It’s among the first studies to explore the impact of pregnancy-induced hypertensive disease on subsequent HF risk separately for both ischemic and nonischemic HF and to find that the severity of such risk differs for the two HF etiologies, according to a report published in JACC: Heart Failure.
The adjusted risk for any HF during a median of 13 years after the pregnancy rose 70% for those who had developed gestational hypertension or preeclampsia. Their risk of nonischemic HF went up 60%, and their risk of ischemic HF more than doubled.
Hypertensive disorders of pregnancy “are so much more than short-term disorders confined to the pregnancy period. They have long-term implications throughout a lifetime,” lead author Ängla Mantel, MD, PhD, said in an interview.
Obstetric history doesn’t figure into any formal HF risk scoring systems, observed Dr. Mantel of Karolinska Institutet, Stockholm. Still, women who develop gestational hypertension, preeclampsia, or other pregnancy complications “should be considered a high-risk population even after the pregnancy and monitored for cardiovascular risk factors regularly throughout life.”
In many studies, she said, “knowledge of women-specific risk factors for cardiovascular disease is poor among both clinicians and patients.” The current findings should help raise awareness about such obstetric risk factors for HF, “especially” in patients with HF with preserved ejection fraction (HFpEF), which isn’t closely related to a number of traditional CV risk factors.
Even though pregnancy complications such as gestational hypertension and preeclampsia don’t feature in risk calculators, “they are actually risk enhancers per the 2019 primary prevention guidelines,” Natalie A. Bello, MD, MPH, who was not involved in the current study, said in an interview.
“We’re working to educate physicians and cardiovascular team members to take a pregnancy history” for risk stratification of women in primary prevention,” said Dr. Bello, director of hypertension research at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles.
The current study, she said, “is an important step” for its finding that hypertensive disorders of pregnancy are associated separately with both ischemic and nonischemic HF.
She pointed out, however, that because the study excluded women with peripartum cardiomyopathy, a form of nonischemic HF, it may “underestimate the impact of hypertensive disorders on the short-term risk of nonischemic heart failure.” Women who had peripartum cardiomyopathy were excluded to avoid misclassification of other HF outcomes, the authors stated.
Also, Dr. Bello said, the study’s inclusion of patients with either gestational hypertension or preeclampsia may complicate its interpretation. Compared with the former condition, she said, preeclampsia “involves more inflammation and more endothelial dysfunction. It may cause a different impact on the heart and the vasculature.”
In the analysis, about 79,000 women with gestational hypertension or preeclampsia were identified among more than 1.4 million primiparous women who entered the Swedish Medical Birth Register over a period of about 30 years. They were matched with about 396,000 women in the registry who had normotensive pregnancies.
Excluded, besides women with peripartum cardiomyopathy, were women with a prepregnancy history of HF, hypertension, ischemic heart disease, atrial fibrillation, or valvular heart disease.
Hazard ratios (HRs) for HF, ischemic HF, and nonischemic HF were significantly elevated over among the women with gestational hypertension or preeclampsia compared to those with normotensive pregnancies:
- Any HF: HR, 1.70 (95% confidence interval [CI], 1.51-1.91)
- Nonischemic HF: HR, 1.60 (95% CI, 1.40-1.83)
- Ischemic HF: HR, 2.28 (95% CI, 1.74-2.98)
The analyses were adjusted for maternal age at delivery, year of delivery, prepregnancy comorbidities, maternal education level, smoking status, and body mass index.
Sharper risk increases were seen among women with gestational hypertension or preeclampsia who delivered prior to gestational week 34:
- Any HF: HR, 2.46 (95% CI, 1.82-3.32)
- Nonischemic HF: HR, 2.33 (95% CI, 1.65-3.31)
- Ischemic HF: HR, 3.64 (95% CI, 1.97-6.74)
Risks for HF developing within 6 years of pregnancy characterized by gestational hypertension or preeclampsia were far more pronounced for ischemic HF than for nonischemic HF:
- Any HF: HR, 2.09 (95% CI, 1.52-2.89)
- Nonischemic HF: HR, 1.86 (95% CI, 1.32-2.61)
- Ischemic HF: HR, 6.52 (95% CI, 2.00-12.34).
The study couldn’t directly explore potential mechanisms for the associations between pregnancy-induced hypertensive disorders and different forms of HF, but it may have provided clues, Dr. Mantel said.
The hypertensive disorders and ischemic HF appear to share risk factors that could lead to both conditions, she noted. Also, hypertension itself is a risk factor for ischemic heart disease.
In contrast, “the risk of nonischemic heart failure might be driven by other factors, such as the inflammatory profile, endothelial dysfunction, and cardiac remodeling induced by preeclampsia or gestational hypertension.”
Those disorders, moreover, are associated with cardiac structural changes that are also seen in HFpEF, Dr. Mantel said. And both HFpEF and preeclampsia are characterized by systemic inflammation and endothelial dysfunction.
“These pathophysiological similarities,” she proposed, “might explain the link between pregnancy-induced hypertensive disorder and HFpEF.”
The authors have disclosed no relevant financial relationships. Dr. Bello has received grants from the National Institutes of Health.
A version of this article first appeared on Medscape.com.
, an observational study suggests.
The risks were most pronounced, jumping more than sixfold in the case of ischemic HF, during the first 6 years after the pregnancy. They then receded to plateau at a lower, still significantly elevated level of risk that persisted even years later, in the analysis of women in a Swedish medical birth registry.
The case-matching study compared women with no history of cardiovascular (CV) disease and a first successful pregnancy during which they either developed or did not experience gestational hypertension or preeclampsia.
It’s among the first studies to explore the impact of pregnancy-induced hypertensive disease on subsequent HF risk separately for both ischemic and nonischemic HF and to find that the severity of such risk differs for the two HF etiologies, according to a report published in JACC: Heart Failure.
The adjusted risk for any HF during a median of 13 years after the pregnancy rose 70% for those who had developed gestational hypertension or preeclampsia. Their risk of nonischemic HF went up 60%, and their risk of ischemic HF more than doubled.
Hypertensive disorders of pregnancy “are so much more than short-term disorders confined to the pregnancy period. They have long-term implications throughout a lifetime,” lead author Ängla Mantel, MD, PhD, said in an interview.
Obstetric history doesn’t figure into any formal HF risk scoring systems, observed Dr. Mantel of Karolinska Institutet, Stockholm. Still, women who develop gestational hypertension, preeclampsia, or other pregnancy complications “should be considered a high-risk population even after the pregnancy and monitored for cardiovascular risk factors regularly throughout life.”
In many studies, she said, “knowledge of women-specific risk factors for cardiovascular disease is poor among both clinicians and patients.” The current findings should help raise awareness about such obstetric risk factors for HF, “especially” in patients with HF with preserved ejection fraction (HFpEF), which isn’t closely related to a number of traditional CV risk factors.
Even though pregnancy complications such as gestational hypertension and preeclampsia don’t feature in risk calculators, “they are actually risk enhancers per the 2019 primary prevention guidelines,” Natalie A. Bello, MD, MPH, who was not involved in the current study, said in an interview.
“We’re working to educate physicians and cardiovascular team members to take a pregnancy history” for risk stratification of women in primary prevention,” said Dr. Bello, director of hypertension research at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles.
The current study, she said, “is an important step” for its finding that hypertensive disorders of pregnancy are associated separately with both ischemic and nonischemic HF.
She pointed out, however, that because the study excluded women with peripartum cardiomyopathy, a form of nonischemic HF, it may “underestimate the impact of hypertensive disorders on the short-term risk of nonischemic heart failure.” Women who had peripartum cardiomyopathy were excluded to avoid misclassification of other HF outcomes, the authors stated.
Also, Dr. Bello said, the study’s inclusion of patients with either gestational hypertension or preeclampsia may complicate its interpretation. Compared with the former condition, she said, preeclampsia “involves more inflammation and more endothelial dysfunction. It may cause a different impact on the heart and the vasculature.”
In the analysis, about 79,000 women with gestational hypertension or preeclampsia were identified among more than 1.4 million primiparous women who entered the Swedish Medical Birth Register over a period of about 30 years. They were matched with about 396,000 women in the registry who had normotensive pregnancies.
Excluded, besides women with peripartum cardiomyopathy, were women with a prepregnancy history of HF, hypertension, ischemic heart disease, atrial fibrillation, or valvular heart disease.
Hazard ratios (HRs) for HF, ischemic HF, and nonischemic HF were significantly elevated over among the women with gestational hypertension or preeclampsia compared to those with normotensive pregnancies:
- Any HF: HR, 1.70 (95% confidence interval [CI], 1.51-1.91)
- Nonischemic HF: HR, 1.60 (95% CI, 1.40-1.83)
- Ischemic HF: HR, 2.28 (95% CI, 1.74-2.98)
The analyses were adjusted for maternal age at delivery, year of delivery, prepregnancy comorbidities, maternal education level, smoking status, and body mass index.
Sharper risk increases were seen among women with gestational hypertension or preeclampsia who delivered prior to gestational week 34:
- Any HF: HR, 2.46 (95% CI, 1.82-3.32)
- Nonischemic HF: HR, 2.33 (95% CI, 1.65-3.31)
- Ischemic HF: HR, 3.64 (95% CI, 1.97-6.74)
Risks for HF developing within 6 years of pregnancy characterized by gestational hypertension or preeclampsia were far more pronounced for ischemic HF than for nonischemic HF:
- Any HF: HR, 2.09 (95% CI, 1.52-2.89)
- Nonischemic HF: HR, 1.86 (95% CI, 1.32-2.61)
- Ischemic HF: HR, 6.52 (95% CI, 2.00-12.34).
The study couldn’t directly explore potential mechanisms for the associations between pregnancy-induced hypertensive disorders and different forms of HF, but it may have provided clues, Dr. Mantel said.
The hypertensive disorders and ischemic HF appear to share risk factors that could lead to both conditions, she noted. Also, hypertension itself is a risk factor for ischemic heart disease.
In contrast, “the risk of nonischemic heart failure might be driven by other factors, such as the inflammatory profile, endothelial dysfunction, and cardiac remodeling induced by preeclampsia or gestational hypertension.”
Those disorders, moreover, are associated with cardiac structural changes that are also seen in HFpEF, Dr. Mantel said. And both HFpEF and preeclampsia are characterized by systemic inflammation and endothelial dysfunction.
“These pathophysiological similarities,” she proposed, “might explain the link between pregnancy-induced hypertensive disorder and HFpEF.”
The authors have disclosed no relevant financial relationships. Dr. Bello has received grants from the National Institutes of Health.
A version of this article first appeared on Medscape.com.
, an observational study suggests.
The risks were most pronounced, jumping more than sixfold in the case of ischemic HF, during the first 6 years after the pregnancy. They then receded to plateau at a lower, still significantly elevated level of risk that persisted even years later, in the analysis of women in a Swedish medical birth registry.
The case-matching study compared women with no history of cardiovascular (CV) disease and a first successful pregnancy during which they either developed or did not experience gestational hypertension or preeclampsia.
It’s among the first studies to explore the impact of pregnancy-induced hypertensive disease on subsequent HF risk separately for both ischemic and nonischemic HF and to find that the severity of such risk differs for the two HF etiologies, according to a report published in JACC: Heart Failure.
The adjusted risk for any HF during a median of 13 years after the pregnancy rose 70% for those who had developed gestational hypertension or preeclampsia. Their risk of nonischemic HF went up 60%, and their risk of ischemic HF more than doubled.
Hypertensive disorders of pregnancy “are so much more than short-term disorders confined to the pregnancy period. They have long-term implications throughout a lifetime,” lead author Ängla Mantel, MD, PhD, said in an interview.
Obstetric history doesn’t figure into any formal HF risk scoring systems, observed Dr. Mantel of Karolinska Institutet, Stockholm. Still, women who develop gestational hypertension, preeclampsia, or other pregnancy complications “should be considered a high-risk population even after the pregnancy and monitored for cardiovascular risk factors regularly throughout life.”
In many studies, she said, “knowledge of women-specific risk factors for cardiovascular disease is poor among both clinicians and patients.” The current findings should help raise awareness about such obstetric risk factors for HF, “especially” in patients with HF with preserved ejection fraction (HFpEF), which isn’t closely related to a number of traditional CV risk factors.
Even though pregnancy complications such as gestational hypertension and preeclampsia don’t feature in risk calculators, “they are actually risk enhancers per the 2019 primary prevention guidelines,” Natalie A. Bello, MD, MPH, who was not involved in the current study, said in an interview.
“We’re working to educate physicians and cardiovascular team members to take a pregnancy history” for risk stratification of women in primary prevention,” said Dr. Bello, director of hypertension research at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles.
The current study, she said, “is an important step” for its finding that hypertensive disorders of pregnancy are associated separately with both ischemic and nonischemic HF.
She pointed out, however, that because the study excluded women with peripartum cardiomyopathy, a form of nonischemic HF, it may “underestimate the impact of hypertensive disorders on the short-term risk of nonischemic heart failure.” Women who had peripartum cardiomyopathy were excluded to avoid misclassification of other HF outcomes, the authors stated.
Also, Dr. Bello said, the study’s inclusion of patients with either gestational hypertension or preeclampsia may complicate its interpretation. Compared with the former condition, she said, preeclampsia “involves more inflammation and more endothelial dysfunction. It may cause a different impact on the heart and the vasculature.”
In the analysis, about 79,000 women with gestational hypertension or preeclampsia were identified among more than 1.4 million primiparous women who entered the Swedish Medical Birth Register over a period of about 30 years. They were matched with about 396,000 women in the registry who had normotensive pregnancies.
Excluded, besides women with peripartum cardiomyopathy, were women with a prepregnancy history of HF, hypertension, ischemic heart disease, atrial fibrillation, or valvular heart disease.
Hazard ratios (HRs) for HF, ischemic HF, and nonischemic HF were significantly elevated over among the women with gestational hypertension or preeclampsia compared to those with normotensive pregnancies:
- Any HF: HR, 1.70 (95% confidence interval [CI], 1.51-1.91)
- Nonischemic HF: HR, 1.60 (95% CI, 1.40-1.83)
- Ischemic HF: HR, 2.28 (95% CI, 1.74-2.98)
The analyses were adjusted for maternal age at delivery, year of delivery, prepregnancy comorbidities, maternal education level, smoking status, and body mass index.
Sharper risk increases were seen among women with gestational hypertension or preeclampsia who delivered prior to gestational week 34:
- Any HF: HR, 2.46 (95% CI, 1.82-3.32)
- Nonischemic HF: HR, 2.33 (95% CI, 1.65-3.31)
- Ischemic HF: HR, 3.64 (95% CI, 1.97-6.74)
Risks for HF developing within 6 years of pregnancy characterized by gestational hypertension or preeclampsia were far more pronounced for ischemic HF than for nonischemic HF:
- Any HF: HR, 2.09 (95% CI, 1.52-2.89)
- Nonischemic HF: HR, 1.86 (95% CI, 1.32-2.61)
- Ischemic HF: HR, 6.52 (95% CI, 2.00-12.34).
The study couldn’t directly explore potential mechanisms for the associations between pregnancy-induced hypertensive disorders and different forms of HF, but it may have provided clues, Dr. Mantel said.
The hypertensive disorders and ischemic HF appear to share risk factors that could lead to both conditions, she noted. Also, hypertension itself is a risk factor for ischemic heart disease.
In contrast, “the risk of nonischemic heart failure might be driven by other factors, such as the inflammatory profile, endothelial dysfunction, and cardiac remodeling induced by preeclampsia or gestational hypertension.”
Those disorders, moreover, are associated with cardiac structural changes that are also seen in HFpEF, Dr. Mantel said. And both HFpEF and preeclampsia are characterized by systemic inflammation and endothelial dysfunction.
“These pathophysiological similarities,” she proposed, “might explain the link between pregnancy-induced hypertensive disorder and HFpEF.”
The authors have disclosed no relevant financial relationships. Dr. Bello has received grants from the National Institutes of Health.
A version of this article first appeared on Medscape.com.
FROM JACC: HEART FAILURE
Could vitamin D supplementation help in long COVID?
, in a retrospective, case-matched study.
The lower levels of vitamin D in patients with long COVID were most notable in those with brain fog.
These findings, by Luigi di Filippo, MD, and colleagues, were recently presented at the European Congress of Endocrinology and published in the Journal of Clinical Endocrinology & Metabolism.
“Our data suggest that vitamin D levels should be evaluated in COVID-19 patients after hospital discharge,” wrote the researchers, from San Raffaele Hospital, Milan.
“The role of vitamin D supplementation as a preventive strategy of COVID-19 sequelae should be tested in randomized controlled trials,” they urged.
The researchers also stressed that this was a controlled study in a homogeneous population, it included multiple signs and symptoms of long COVID, and it had a longer follow-up than most previous studies (6 vs. 3 months).
“The highly controlled nature of our study helps us better understand the role of vitamin D deficiency in long COVID and establish that there is likely a link between vitamin D deficiency and long COVID,” senior author Andrea Giustina, MD, said in a press release from the ECE.
“Our study shows that COVID-19 patients with low vitamin D levels are more likely to develop long COVID, but it is not yet known whether vitamin D supplements could improve the symptoms or reduce this risk altogether,” he cautioned.
“If confirmed in large, interventional, randomized controlled trials, [our data suggest] that vitamin D supplementation could represent a possible preventive strategy in reducing the burden of COVID-19 sequelae,” Dr. Giustina and colleagues wrote.
Reasonable to test vitamin D levels, consider supplementation
Invited to comment, Amiel Dror, MD, PhD, who led a related study that showed that people with a vitamin D deficiency were more likely to have severe COVID-19, agreed.
“The novelty and significance of this [new] study lie in the fact that it expands on our current understanding of the interplay between vitamin D and COVID-19, taking it beyond the acute phase of the disease,” said Dr. Dror, from Bar-Ilan University, Safed, Israel.
“It’s striking to see how vitamin D levels continue to influence patients’ health even after recovery from the initial infection,” he noted.
“The findings certainly add weight to the argument for conducting a randomized control trial [RCT],” he continued, which “would enable us to conclusively determine whether vitamin D supplementation can effectively reduce the risk or severity of long COVID.”
“In the interim,” Dr. Dror said, “given the safety profile of vitamin D and its broad health benefits, it could be reasonable to test for vitamin D levels in patients admitted with COVID-19. If levels are found to be low, supplementation could be considered.”
“However, it’s important to note that this should be done under medical supervision,” he cautioned, “and further studies are needed to establish the optimal timing and dosage of supplementation.”
“I anticipate that we’ll see more RCTs [of this] in the future,” he speculated.
Low vitamin D and risk of long COVID
Long COVID is an emerging syndrome that affects 50%-70% of COVID-19 survivors.
Low levels of vitamin D have been associated with increased likelihood of needing mechanical ventilation and worse survival in patients hospitalized with COVID-19, but the risk of long COVID associated with vitamin D has not been known.
Researchers analyzed data from adults aged 18 and older hospitalized at San Raffaele Hospital with a confirmed diagnosis of COVID-19 and discharged during the first pandemic wave, from March to May 2020, and then seen 6-months later for follow-up.
Patients were excluded if they had been admitted to the intensive care unit during hospitalization or had missing medical data or blood samples available to determine (OH) vitamin D levels, at admission and the 6-month follow-up.
Long COVID-19 was defined based on the U.K. National Institute for Health and Care Excellence guidelines as the concomitant presence of at least two or more of 17 signs and symptoms that were absent prior to the COVID-19 infection and could only be attributed to that acute disease.
Researchers identified 50 patients with long COVID at the 6-month follow-up and matched them with 50 patients without long COVID at that time point, based on age, sex, concomitant comorbidities, need for noninvasive mechanical ventilation, and week of evaluation.
Patients were a mean age of 61 years (range, 51-73) and 56% were men; 28% had been on a ventilator during hospitalization for COVID-19.
The most frequent signs and symptoms at 6 months in the patients with long COVID were asthenia (weakness, 38% of patients), dysgeusia (bad taste in the mouth, 34%), dyspnea (shortness of breath, 34%), and anosmia (loss of sense of smell, 24%).
Most symptoms were related to the cardiorespiratory system (42%), the feeling of well-being (42%), or the senses (36%), and fewer patients had symptoms related to neurocognitive impairment (headache or brain fog, 14%), or ear, nose, and throat (12%), or gastrointestinal system (4%).
Patients with long COVID had lower mean 25(OH) vitamin D levels than patients without long COVID (20.1 vs 23.2 ng/mL; P = .03). However, actual vitamin D deficiency levels were similar in both groups.
Two-thirds of patients with low vitamin D levels at hospital admission still presented with low levels at the 6-month follow-up.
Vitamin D levels were significantly lower in patients with neurocognitive symptoms at follow-up (n = 7) than in those without such symptoms (n = 93) (14.6 vs. 20.6 ng/mL; P = .042).
In patients with vitamin D deficiency (< 20 ng/mL) at admission and at follow-up (n = 42), those with long COVID (n = 22) had lower vitamin D levels at follow-up than those without long COVID (n = 20) (12.7 vs. 15.2 ng/mL; P = .041).
And in multiple regression analyses, a lower 25(OH) vitamin D level at follow-up was the only variable that was significantly associated with long COVID (odds ratio, 1.09; 95% confidence interval, 1.01-1.16; P = .008).
The findings “strongly reinforce the clinical usefulness of 25(OH) vitamin D evaluation as a possible modifiable pathophysiological factor underlying this emerging worldwide critical health issue,” the researchers concluded.
The study was supported by Abiogen Pharma. One study author is an employee at Abiogen. Dr. Giustina has reported being a consultant for Abiogen and Takeda and receiving a research grant to his institution from Takeda. Dr. Di Filippo and the other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, in a retrospective, case-matched study.
The lower levels of vitamin D in patients with long COVID were most notable in those with brain fog.
These findings, by Luigi di Filippo, MD, and colleagues, were recently presented at the European Congress of Endocrinology and published in the Journal of Clinical Endocrinology & Metabolism.
“Our data suggest that vitamin D levels should be evaluated in COVID-19 patients after hospital discharge,” wrote the researchers, from San Raffaele Hospital, Milan.
“The role of vitamin D supplementation as a preventive strategy of COVID-19 sequelae should be tested in randomized controlled trials,” they urged.
The researchers also stressed that this was a controlled study in a homogeneous population, it included multiple signs and symptoms of long COVID, and it had a longer follow-up than most previous studies (6 vs. 3 months).
“The highly controlled nature of our study helps us better understand the role of vitamin D deficiency in long COVID and establish that there is likely a link between vitamin D deficiency and long COVID,” senior author Andrea Giustina, MD, said in a press release from the ECE.
“Our study shows that COVID-19 patients with low vitamin D levels are more likely to develop long COVID, but it is not yet known whether vitamin D supplements could improve the symptoms or reduce this risk altogether,” he cautioned.
“If confirmed in large, interventional, randomized controlled trials, [our data suggest] that vitamin D supplementation could represent a possible preventive strategy in reducing the burden of COVID-19 sequelae,” Dr. Giustina and colleagues wrote.
Reasonable to test vitamin D levels, consider supplementation
Invited to comment, Amiel Dror, MD, PhD, who led a related study that showed that people with a vitamin D deficiency were more likely to have severe COVID-19, agreed.
“The novelty and significance of this [new] study lie in the fact that it expands on our current understanding of the interplay between vitamin D and COVID-19, taking it beyond the acute phase of the disease,” said Dr. Dror, from Bar-Ilan University, Safed, Israel.
“It’s striking to see how vitamin D levels continue to influence patients’ health even after recovery from the initial infection,” he noted.
“The findings certainly add weight to the argument for conducting a randomized control trial [RCT],” he continued, which “would enable us to conclusively determine whether vitamin D supplementation can effectively reduce the risk or severity of long COVID.”
“In the interim,” Dr. Dror said, “given the safety profile of vitamin D and its broad health benefits, it could be reasonable to test for vitamin D levels in patients admitted with COVID-19. If levels are found to be low, supplementation could be considered.”
“However, it’s important to note that this should be done under medical supervision,” he cautioned, “and further studies are needed to establish the optimal timing and dosage of supplementation.”
“I anticipate that we’ll see more RCTs [of this] in the future,” he speculated.
Low vitamin D and risk of long COVID
Long COVID is an emerging syndrome that affects 50%-70% of COVID-19 survivors.
Low levels of vitamin D have been associated with increased likelihood of needing mechanical ventilation and worse survival in patients hospitalized with COVID-19, but the risk of long COVID associated with vitamin D has not been known.
Researchers analyzed data from adults aged 18 and older hospitalized at San Raffaele Hospital with a confirmed diagnosis of COVID-19 and discharged during the first pandemic wave, from March to May 2020, and then seen 6-months later for follow-up.
Patients were excluded if they had been admitted to the intensive care unit during hospitalization or had missing medical data or blood samples available to determine (OH) vitamin D levels, at admission and the 6-month follow-up.
Long COVID-19 was defined based on the U.K. National Institute for Health and Care Excellence guidelines as the concomitant presence of at least two or more of 17 signs and symptoms that were absent prior to the COVID-19 infection and could only be attributed to that acute disease.
Researchers identified 50 patients with long COVID at the 6-month follow-up and matched them with 50 patients without long COVID at that time point, based on age, sex, concomitant comorbidities, need for noninvasive mechanical ventilation, and week of evaluation.
Patients were a mean age of 61 years (range, 51-73) and 56% were men; 28% had been on a ventilator during hospitalization for COVID-19.
The most frequent signs and symptoms at 6 months in the patients with long COVID were asthenia (weakness, 38% of patients), dysgeusia (bad taste in the mouth, 34%), dyspnea (shortness of breath, 34%), and anosmia (loss of sense of smell, 24%).
Most symptoms were related to the cardiorespiratory system (42%), the feeling of well-being (42%), or the senses (36%), and fewer patients had symptoms related to neurocognitive impairment (headache or brain fog, 14%), or ear, nose, and throat (12%), or gastrointestinal system (4%).
Patients with long COVID had lower mean 25(OH) vitamin D levels than patients without long COVID (20.1 vs 23.2 ng/mL; P = .03). However, actual vitamin D deficiency levels were similar in both groups.
Two-thirds of patients with low vitamin D levels at hospital admission still presented with low levels at the 6-month follow-up.
Vitamin D levels were significantly lower in patients with neurocognitive symptoms at follow-up (n = 7) than in those without such symptoms (n = 93) (14.6 vs. 20.6 ng/mL; P = .042).
In patients with vitamin D deficiency (< 20 ng/mL) at admission and at follow-up (n = 42), those with long COVID (n = 22) had lower vitamin D levels at follow-up than those without long COVID (n = 20) (12.7 vs. 15.2 ng/mL; P = .041).
And in multiple regression analyses, a lower 25(OH) vitamin D level at follow-up was the only variable that was significantly associated with long COVID (odds ratio, 1.09; 95% confidence interval, 1.01-1.16; P = .008).
The findings “strongly reinforce the clinical usefulness of 25(OH) vitamin D evaluation as a possible modifiable pathophysiological factor underlying this emerging worldwide critical health issue,” the researchers concluded.
The study was supported by Abiogen Pharma. One study author is an employee at Abiogen. Dr. Giustina has reported being a consultant for Abiogen and Takeda and receiving a research grant to his institution from Takeda. Dr. Di Filippo and the other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, in a retrospective, case-matched study.
The lower levels of vitamin D in patients with long COVID were most notable in those with brain fog.
These findings, by Luigi di Filippo, MD, and colleagues, were recently presented at the European Congress of Endocrinology and published in the Journal of Clinical Endocrinology & Metabolism.
“Our data suggest that vitamin D levels should be evaluated in COVID-19 patients after hospital discharge,” wrote the researchers, from San Raffaele Hospital, Milan.
“The role of vitamin D supplementation as a preventive strategy of COVID-19 sequelae should be tested in randomized controlled trials,” they urged.
The researchers also stressed that this was a controlled study in a homogeneous population, it included multiple signs and symptoms of long COVID, and it had a longer follow-up than most previous studies (6 vs. 3 months).
“The highly controlled nature of our study helps us better understand the role of vitamin D deficiency in long COVID and establish that there is likely a link between vitamin D deficiency and long COVID,” senior author Andrea Giustina, MD, said in a press release from the ECE.
“Our study shows that COVID-19 patients with low vitamin D levels are more likely to develop long COVID, but it is not yet known whether vitamin D supplements could improve the symptoms or reduce this risk altogether,” he cautioned.
“If confirmed in large, interventional, randomized controlled trials, [our data suggest] that vitamin D supplementation could represent a possible preventive strategy in reducing the burden of COVID-19 sequelae,” Dr. Giustina and colleagues wrote.
Reasonable to test vitamin D levels, consider supplementation
Invited to comment, Amiel Dror, MD, PhD, who led a related study that showed that people with a vitamin D deficiency were more likely to have severe COVID-19, agreed.
“The novelty and significance of this [new] study lie in the fact that it expands on our current understanding of the interplay between vitamin D and COVID-19, taking it beyond the acute phase of the disease,” said Dr. Dror, from Bar-Ilan University, Safed, Israel.
“It’s striking to see how vitamin D levels continue to influence patients’ health even after recovery from the initial infection,” he noted.
“The findings certainly add weight to the argument for conducting a randomized control trial [RCT],” he continued, which “would enable us to conclusively determine whether vitamin D supplementation can effectively reduce the risk or severity of long COVID.”
“In the interim,” Dr. Dror said, “given the safety profile of vitamin D and its broad health benefits, it could be reasonable to test for vitamin D levels in patients admitted with COVID-19. If levels are found to be low, supplementation could be considered.”
“However, it’s important to note that this should be done under medical supervision,” he cautioned, “and further studies are needed to establish the optimal timing and dosage of supplementation.”
“I anticipate that we’ll see more RCTs [of this] in the future,” he speculated.
Low vitamin D and risk of long COVID
Long COVID is an emerging syndrome that affects 50%-70% of COVID-19 survivors.
Low levels of vitamin D have been associated with increased likelihood of needing mechanical ventilation and worse survival in patients hospitalized with COVID-19, but the risk of long COVID associated with vitamin D has not been known.
Researchers analyzed data from adults aged 18 and older hospitalized at San Raffaele Hospital with a confirmed diagnosis of COVID-19 and discharged during the first pandemic wave, from March to May 2020, and then seen 6-months later for follow-up.
Patients were excluded if they had been admitted to the intensive care unit during hospitalization or had missing medical data or blood samples available to determine (OH) vitamin D levels, at admission and the 6-month follow-up.
Long COVID-19 was defined based on the U.K. National Institute for Health and Care Excellence guidelines as the concomitant presence of at least two or more of 17 signs and symptoms that were absent prior to the COVID-19 infection and could only be attributed to that acute disease.
Researchers identified 50 patients with long COVID at the 6-month follow-up and matched them with 50 patients without long COVID at that time point, based on age, sex, concomitant comorbidities, need for noninvasive mechanical ventilation, and week of evaluation.
Patients were a mean age of 61 years (range, 51-73) and 56% were men; 28% had been on a ventilator during hospitalization for COVID-19.
The most frequent signs and symptoms at 6 months in the patients with long COVID were asthenia (weakness, 38% of patients), dysgeusia (bad taste in the mouth, 34%), dyspnea (shortness of breath, 34%), and anosmia (loss of sense of smell, 24%).
Most symptoms were related to the cardiorespiratory system (42%), the feeling of well-being (42%), or the senses (36%), and fewer patients had symptoms related to neurocognitive impairment (headache or brain fog, 14%), or ear, nose, and throat (12%), or gastrointestinal system (4%).
Patients with long COVID had lower mean 25(OH) vitamin D levels than patients without long COVID (20.1 vs 23.2 ng/mL; P = .03). However, actual vitamin D deficiency levels were similar in both groups.
Two-thirds of patients with low vitamin D levels at hospital admission still presented with low levels at the 6-month follow-up.
Vitamin D levels were significantly lower in patients with neurocognitive symptoms at follow-up (n = 7) than in those without such symptoms (n = 93) (14.6 vs. 20.6 ng/mL; P = .042).
In patients with vitamin D deficiency (< 20 ng/mL) at admission and at follow-up (n = 42), those with long COVID (n = 22) had lower vitamin D levels at follow-up than those without long COVID (n = 20) (12.7 vs. 15.2 ng/mL; P = .041).
And in multiple regression analyses, a lower 25(OH) vitamin D level at follow-up was the only variable that was significantly associated with long COVID (odds ratio, 1.09; 95% confidence interval, 1.01-1.16; P = .008).
The findings “strongly reinforce the clinical usefulness of 25(OH) vitamin D evaluation as a possible modifiable pathophysiological factor underlying this emerging worldwide critical health issue,” the researchers concluded.
The study was supported by Abiogen Pharma. One study author is an employee at Abiogen. Dr. Giustina has reported being a consultant for Abiogen and Takeda and receiving a research grant to his institution from Takeda. Dr. Di Filippo and the other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ECE 2023
Mixed results on two treatments for erectile dysfunction
presented at the annual meeting of the American Urological Association.
In a single-blind prospective study that evaluated low-intensity shock-wave therapy, researchers randomly assigned 36 men with ED to receive mechanical therapy (n = 22) or sham treatment (n = 14) on their flaccid penises.
The patients in arm 1 of the study received three treatments of 5,000 shocks (4 Hz, 0.12 mJ/mm2) with the UroGold 1000 device (SoftWave) at weeks 0, 3, and 6. Those in arm 2 received a regimen of 5,000 shocks at week 0 and 3,000 at weeks 2 and 3, which was repeated 3 weeks later. Patients who completed sham treatment were unblinded and crossed over to the opposite arm for active treatment.
At weeks 20 and 32, the researchers assessed changes in gray-scale ultrasound erectile tissue homogeneity of the corpora cavernosa using visual grading scores as well as changes in color Duplex Doppler ultrasound assessments of artery blood flow parameters between baseline and follow-up.
Better blood flow – But is that enough?
After shock-wave therapy, more men experienced either improvements in or no worsening of blood flow parameters relative to baseline than after sham treatment. The decrease in end-diastolic volume was statistically significant for men in the active treatment arm 2 at week 32 (P = .003), according to the researchers.
The number of men whose visual grading scores for ultrasound gray-scale images improved in the proximal region was consistently higher with active treatment than with placebo (arm 1: 88.9% vs. 11.1%; arm 2: 40% vs. 20%), with statistical significance in arm 1 at weeks 20 (P = .005) and 32 (P = .001). Patients who received sham treatment and who subsequently received active shock-wave therapy also had improved scores on gray-scale ultrasound (arm 1: 33.3% vs. 11.1%; arm 2: 40% vs. 20%).
Scores on the International Index of Erectile Function (IIEF) were nominally higher for men in active treatment whose visual grading scores had improved compared with those who did not show improvement.
The most common adverse event was transient discomfort after the shock-wave treatment, according to the researchers.
The study provides “a glimpse into the concept” that the mechanotransduction from a shock wave results in biochemical changes, including “activation of stem cells within the corpus cavernosum,” said Irwin Goldstein, MD, director of San Diego Sexual Medicine and clinical professor of surgery at the University of California, San Diego, who led the trial. “If I can activate stem cells,” he added, “theoretically, I can improve the health of tissue.”
Dr. Goldstein noted that the study is the first to use before-and-after objective gray-scale ultrasound imaging along with color Doppler ultrasound. “We could see gray scale changes and peak systolic velocity changes even with a small group,” he said.
Dr. Goldstein added that the trial is the first in which zero energy was used in the sham phase instead of less energy than active treatment. With the sham treatment, there was no benefit on the gray scale, which he said is “very important.”
He said his team is in the process of submitting a proposal for a larger prospective trial to confirm the findings.
Although the results are promising, the study did not evaluate what matters most to men, said Louis Kuritzky, MD, a family medicine physician and assistant professor emeritus at HCA UCF Family Medicine Residency, in Gainesville, Fla.
“Men don’t care what the flow velocity is – they care [whether] they get an erection sufficient for penetration and completion of intercourse. That trial did not look at those endpoints. It looked at surrogates. Those are encouraging, but that’s not what I think a clinician would base their decision upon about whether or not a patient should possibly participate in shock therapy.”
Plasma injections a bust
The trial that assessed platelet-rich plasma was not encouraging. The results of the prospective, double-blind, randomized, placebo-controlled trial suggest that PRP is safe but not effective.
A proprietary version of the PRP injection is marketed as the “Priapus shot,” or the “P-shot,” despite a lack of solid evidence the therapy helps.
Brian Ledesma, MD, an andrology research fellow at the University of Miami, led the study, which received a “best abstract” award at the meeting. “We wanted to actually check and see – does this work or not?” Dr. Ledesma said of PRP injections generally.
Dr. Ledesma and his colleagues randomly assigned 61 men with mild to moderate ED to receive two intracavernosal injections of PRP 1 month apart (n = 28) or placebo treatment (n = 33). The primary outcome was change in IIEF score and the percentage of men meeting minimum clinically important difference (MCID) at 1 month. Complete data were available for 24 men who received PRP and for 28 who received placebo injections.
There was no significant difference in outcomes between the groups. IIEF scores changed from 17.4 (95% confidence interval [CI], –15.8 to 19.0) to 21 (95% CI, 17.9-24.0) for men who received PRP and from 18.6 (95% CI, 17.3-19.8) to 21.6 (95% CI, 19.1-24.1) for men in the placebo group (P = .756). Fourteen men (58.3%) in the PRP group, compared with 15 (53.6%) in the placebo group, met MCID. No differences were seen in mean penile Doppler parameters between PRP and placebo. The two adverse events reported in the trial were minor – a hematoma and “a new plaque that did not cause any curvature of the penis,” Dr. Ledesma said.
Platelet-rich plasma may be “really popular, but, objectively speaking, so far, we don’t have any evidence showing that it actually works.”
The study showed that “PRP was not more efficacious than placebo.” This treatment is “really popular, but, objectively speaking, so far, we don’t have any evidence showing that it actually works.”
Based on these findings, he said, “We would recommend sticking to the data primary care providers should tell their patients, ‘Don’t waste your money,’ because it’s pretty expensive.”
Dr. Kuritzky said more studies are needed for a definitive answer. “I think the results of PRP have been largely disappointing across most of the spheres of influence in which it’s been tried. So, it’s not so surprising to me that this trial would, again, not prove efficacious, but I’d have to hold judgment, dependent upon other trials,” he said.
Dr. Ledesma and his colleagues are conducting a prospective, randomized, double-blind trial “investigating whether PRP combined with shock-wave therapy could make a difference.” He said the trial, which is funded by the National Institutes of Health, is in the enrollment phase; results are expected in mid-2024.
Charles Runels, MD, who pioneered the P-Shot and other popular cosmetic procedures, defended the effectiveness of the injections.
“One of the legitimate criticisms of all of the review articles regarding PRP therapies in every field is that there is a significant variability in what people call PRP. The P-Shot represents a very specific protocol in the methods of preparing PRP, activating PRP, and injecting the PRP – all of which differ significantly from what was done in Ledesma’s study and which could account for their lack of results,” Dr. Runels said in an interview.
Dr. Runels added that “multiple studies” do show benefit of the injection of PRP for both erectile dysfunction and Peyronie’s disease and support the success of his protocol. “Also, all of our providers – there are over 3,000 people in our Cellular Medicine Association – offer money back to our patients if there are not satisfactory results,” he said.
Dr. Kuritzky said that when patients ask him about investigational treatments for ED, he tells them to stick to the more traditional approaches, such as phosphodiesterase type 5 inhibitors, intracorporeal injections, and vacuum devices.
But, he added, if other therapies are shown to be safe and effective “in a large population of men with diverse etiologies associated with their erectile dysfunction, including advanced age, diabetes, dyslipidemia, hypertension, cigarette smoking, then I think [they] could be recommended on a more consistent basis.”
Both studies were independently supported. Dr. Goldstein, Dr. Kuritzky, and Dr. Ledesma reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
presented at the annual meeting of the American Urological Association.
In a single-blind prospective study that evaluated low-intensity shock-wave therapy, researchers randomly assigned 36 men with ED to receive mechanical therapy (n = 22) or sham treatment (n = 14) on their flaccid penises.
The patients in arm 1 of the study received three treatments of 5,000 shocks (4 Hz, 0.12 mJ/mm2) with the UroGold 1000 device (SoftWave) at weeks 0, 3, and 6. Those in arm 2 received a regimen of 5,000 shocks at week 0 and 3,000 at weeks 2 and 3, which was repeated 3 weeks later. Patients who completed sham treatment were unblinded and crossed over to the opposite arm for active treatment.
At weeks 20 and 32, the researchers assessed changes in gray-scale ultrasound erectile tissue homogeneity of the corpora cavernosa using visual grading scores as well as changes in color Duplex Doppler ultrasound assessments of artery blood flow parameters between baseline and follow-up.
Better blood flow – But is that enough?
After shock-wave therapy, more men experienced either improvements in or no worsening of blood flow parameters relative to baseline than after sham treatment. The decrease in end-diastolic volume was statistically significant for men in the active treatment arm 2 at week 32 (P = .003), according to the researchers.
The number of men whose visual grading scores for ultrasound gray-scale images improved in the proximal region was consistently higher with active treatment than with placebo (arm 1: 88.9% vs. 11.1%; arm 2: 40% vs. 20%), with statistical significance in arm 1 at weeks 20 (P = .005) and 32 (P = .001). Patients who received sham treatment and who subsequently received active shock-wave therapy also had improved scores on gray-scale ultrasound (arm 1: 33.3% vs. 11.1%; arm 2: 40% vs. 20%).
Scores on the International Index of Erectile Function (IIEF) were nominally higher for men in active treatment whose visual grading scores had improved compared with those who did not show improvement.
The most common adverse event was transient discomfort after the shock-wave treatment, according to the researchers.
The study provides “a glimpse into the concept” that the mechanotransduction from a shock wave results in biochemical changes, including “activation of stem cells within the corpus cavernosum,” said Irwin Goldstein, MD, director of San Diego Sexual Medicine and clinical professor of surgery at the University of California, San Diego, who led the trial. “If I can activate stem cells,” he added, “theoretically, I can improve the health of tissue.”
Dr. Goldstein noted that the study is the first to use before-and-after objective gray-scale ultrasound imaging along with color Doppler ultrasound. “We could see gray scale changes and peak systolic velocity changes even with a small group,” he said.
Dr. Goldstein added that the trial is the first in which zero energy was used in the sham phase instead of less energy than active treatment. With the sham treatment, there was no benefit on the gray scale, which he said is “very important.”
He said his team is in the process of submitting a proposal for a larger prospective trial to confirm the findings.
Although the results are promising, the study did not evaluate what matters most to men, said Louis Kuritzky, MD, a family medicine physician and assistant professor emeritus at HCA UCF Family Medicine Residency, in Gainesville, Fla.
“Men don’t care what the flow velocity is – they care [whether] they get an erection sufficient for penetration and completion of intercourse. That trial did not look at those endpoints. It looked at surrogates. Those are encouraging, but that’s not what I think a clinician would base their decision upon about whether or not a patient should possibly participate in shock therapy.”
Plasma injections a bust
The trial that assessed platelet-rich plasma was not encouraging. The results of the prospective, double-blind, randomized, placebo-controlled trial suggest that PRP is safe but not effective.
A proprietary version of the PRP injection is marketed as the “Priapus shot,” or the “P-shot,” despite a lack of solid evidence the therapy helps.
Brian Ledesma, MD, an andrology research fellow at the University of Miami, led the study, which received a “best abstract” award at the meeting. “We wanted to actually check and see – does this work or not?” Dr. Ledesma said of PRP injections generally.
Dr. Ledesma and his colleagues randomly assigned 61 men with mild to moderate ED to receive two intracavernosal injections of PRP 1 month apart (n = 28) or placebo treatment (n = 33). The primary outcome was change in IIEF score and the percentage of men meeting minimum clinically important difference (MCID) at 1 month. Complete data were available for 24 men who received PRP and for 28 who received placebo injections.
There was no significant difference in outcomes between the groups. IIEF scores changed from 17.4 (95% confidence interval [CI], –15.8 to 19.0) to 21 (95% CI, 17.9-24.0) for men who received PRP and from 18.6 (95% CI, 17.3-19.8) to 21.6 (95% CI, 19.1-24.1) for men in the placebo group (P = .756). Fourteen men (58.3%) in the PRP group, compared with 15 (53.6%) in the placebo group, met MCID. No differences were seen in mean penile Doppler parameters between PRP and placebo. The two adverse events reported in the trial were minor – a hematoma and “a new plaque that did not cause any curvature of the penis,” Dr. Ledesma said.
Platelet-rich plasma may be “really popular, but, objectively speaking, so far, we don’t have any evidence showing that it actually works.”
The study showed that “PRP was not more efficacious than placebo.” This treatment is “really popular, but, objectively speaking, so far, we don’t have any evidence showing that it actually works.”
Based on these findings, he said, “We would recommend sticking to the data primary care providers should tell their patients, ‘Don’t waste your money,’ because it’s pretty expensive.”
Dr. Kuritzky said more studies are needed for a definitive answer. “I think the results of PRP have been largely disappointing across most of the spheres of influence in which it’s been tried. So, it’s not so surprising to me that this trial would, again, not prove efficacious, but I’d have to hold judgment, dependent upon other trials,” he said.
Dr. Ledesma and his colleagues are conducting a prospective, randomized, double-blind trial “investigating whether PRP combined with shock-wave therapy could make a difference.” He said the trial, which is funded by the National Institutes of Health, is in the enrollment phase; results are expected in mid-2024.
Charles Runels, MD, who pioneered the P-Shot and other popular cosmetic procedures, defended the effectiveness of the injections.
“One of the legitimate criticisms of all of the review articles regarding PRP therapies in every field is that there is a significant variability in what people call PRP. The P-Shot represents a very specific protocol in the methods of preparing PRP, activating PRP, and injecting the PRP – all of which differ significantly from what was done in Ledesma’s study and which could account for their lack of results,” Dr. Runels said in an interview.
Dr. Runels added that “multiple studies” do show benefit of the injection of PRP for both erectile dysfunction and Peyronie’s disease and support the success of his protocol. “Also, all of our providers – there are over 3,000 people in our Cellular Medicine Association – offer money back to our patients if there are not satisfactory results,” he said.
Dr. Kuritzky said that when patients ask him about investigational treatments for ED, he tells them to stick to the more traditional approaches, such as phosphodiesterase type 5 inhibitors, intracorporeal injections, and vacuum devices.
But, he added, if other therapies are shown to be safe and effective “in a large population of men with diverse etiologies associated with their erectile dysfunction, including advanced age, diabetes, dyslipidemia, hypertension, cigarette smoking, then I think [they] could be recommended on a more consistent basis.”
Both studies were independently supported. Dr. Goldstein, Dr. Kuritzky, and Dr. Ledesma reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
presented at the annual meeting of the American Urological Association.
In a single-blind prospective study that evaluated low-intensity shock-wave therapy, researchers randomly assigned 36 men with ED to receive mechanical therapy (n = 22) or sham treatment (n = 14) on their flaccid penises.
The patients in arm 1 of the study received three treatments of 5,000 shocks (4 Hz, 0.12 mJ/mm2) with the UroGold 1000 device (SoftWave) at weeks 0, 3, and 6. Those in arm 2 received a regimen of 5,000 shocks at week 0 and 3,000 at weeks 2 and 3, which was repeated 3 weeks later. Patients who completed sham treatment were unblinded and crossed over to the opposite arm for active treatment.
At weeks 20 and 32, the researchers assessed changes in gray-scale ultrasound erectile tissue homogeneity of the corpora cavernosa using visual grading scores as well as changes in color Duplex Doppler ultrasound assessments of artery blood flow parameters between baseline and follow-up.
Better blood flow – But is that enough?
After shock-wave therapy, more men experienced either improvements in or no worsening of blood flow parameters relative to baseline than after sham treatment. The decrease in end-diastolic volume was statistically significant for men in the active treatment arm 2 at week 32 (P = .003), according to the researchers.
The number of men whose visual grading scores for ultrasound gray-scale images improved in the proximal region was consistently higher with active treatment than with placebo (arm 1: 88.9% vs. 11.1%; arm 2: 40% vs. 20%), with statistical significance in arm 1 at weeks 20 (P = .005) and 32 (P = .001). Patients who received sham treatment and who subsequently received active shock-wave therapy also had improved scores on gray-scale ultrasound (arm 1: 33.3% vs. 11.1%; arm 2: 40% vs. 20%).
Scores on the International Index of Erectile Function (IIEF) were nominally higher for men in active treatment whose visual grading scores had improved compared with those who did not show improvement.
The most common adverse event was transient discomfort after the shock-wave treatment, according to the researchers.
The study provides “a glimpse into the concept” that the mechanotransduction from a shock wave results in biochemical changes, including “activation of stem cells within the corpus cavernosum,” said Irwin Goldstein, MD, director of San Diego Sexual Medicine and clinical professor of surgery at the University of California, San Diego, who led the trial. “If I can activate stem cells,” he added, “theoretically, I can improve the health of tissue.”
Dr. Goldstein noted that the study is the first to use before-and-after objective gray-scale ultrasound imaging along with color Doppler ultrasound. “We could see gray scale changes and peak systolic velocity changes even with a small group,” he said.
Dr. Goldstein added that the trial is the first in which zero energy was used in the sham phase instead of less energy than active treatment. With the sham treatment, there was no benefit on the gray scale, which he said is “very important.”
He said his team is in the process of submitting a proposal for a larger prospective trial to confirm the findings.
Although the results are promising, the study did not evaluate what matters most to men, said Louis Kuritzky, MD, a family medicine physician and assistant professor emeritus at HCA UCF Family Medicine Residency, in Gainesville, Fla.
“Men don’t care what the flow velocity is – they care [whether] they get an erection sufficient for penetration and completion of intercourse. That trial did not look at those endpoints. It looked at surrogates. Those are encouraging, but that’s not what I think a clinician would base their decision upon about whether or not a patient should possibly participate in shock therapy.”
Plasma injections a bust
The trial that assessed platelet-rich plasma was not encouraging. The results of the prospective, double-blind, randomized, placebo-controlled trial suggest that PRP is safe but not effective.
A proprietary version of the PRP injection is marketed as the “Priapus shot,” or the “P-shot,” despite a lack of solid evidence the therapy helps.
Brian Ledesma, MD, an andrology research fellow at the University of Miami, led the study, which received a “best abstract” award at the meeting. “We wanted to actually check and see – does this work or not?” Dr. Ledesma said of PRP injections generally.
Dr. Ledesma and his colleagues randomly assigned 61 men with mild to moderate ED to receive two intracavernosal injections of PRP 1 month apart (n = 28) or placebo treatment (n = 33). The primary outcome was change in IIEF score and the percentage of men meeting minimum clinically important difference (MCID) at 1 month. Complete data were available for 24 men who received PRP and for 28 who received placebo injections.
There was no significant difference in outcomes between the groups. IIEF scores changed from 17.4 (95% confidence interval [CI], –15.8 to 19.0) to 21 (95% CI, 17.9-24.0) for men who received PRP and from 18.6 (95% CI, 17.3-19.8) to 21.6 (95% CI, 19.1-24.1) for men in the placebo group (P = .756). Fourteen men (58.3%) in the PRP group, compared with 15 (53.6%) in the placebo group, met MCID. No differences were seen in mean penile Doppler parameters between PRP and placebo. The two adverse events reported in the trial were minor – a hematoma and “a new plaque that did not cause any curvature of the penis,” Dr. Ledesma said.
Platelet-rich plasma may be “really popular, but, objectively speaking, so far, we don’t have any evidence showing that it actually works.”
The study showed that “PRP was not more efficacious than placebo.” This treatment is “really popular, but, objectively speaking, so far, we don’t have any evidence showing that it actually works.”
Based on these findings, he said, “We would recommend sticking to the data primary care providers should tell their patients, ‘Don’t waste your money,’ because it’s pretty expensive.”
Dr. Kuritzky said more studies are needed for a definitive answer. “I think the results of PRP have been largely disappointing across most of the spheres of influence in which it’s been tried. So, it’s not so surprising to me that this trial would, again, not prove efficacious, but I’d have to hold judgment, dependent upon other trials,” he said.
Dr. Ledesma and his colleagues are conducting a prospective, randomized, double-blind trial “investigating whether PRP combined with shock-wave therapy could make a difference.” He said the trial, which is funded by the National Institutes of Health, is in the enrollment phase; results are expected in mid-2024.
Charles Runels, MD, who pioneered the P-Shot and other popular cosmetic procedures, defended the effectiveness of the injections.
“One of the legitimate criticisms of all of the review articles regarding PRP therapies in every field is that there is a significant variability in what people call PRP. The P-Shot represents a very specific protocol in the methods of preparing PRP, activating PRP, and injecting the PRP – all of which differ significantly from what was done in Ledesma’s study and which could account for their lack of results,” Dr. Runels said in an interview.
Dr. Runels added that “multiple studies” do show benefit of the injection of PRP for both erectile dysfunction and Peyronie’s disease and support the success of his protocol. “Also, all of our providers – there are over 3,000 people in our Cellular Medicine Association – offer money back to our patients if there are not satisfactory results,” he said.
Dr. Kuritzky said that when patients ask him about investigational treatments for ED, he tells them to stick to the more traditional approaches, such as phosphodiesterase type 5 inhibitors, intracorporeal injections, and vacuum devices.
But, he added, if other therapies are shown to be safe and effective “in a large population of men with diverse etiologies associated with their erectile dysfunction, including advanced age, diabetes, dyslipidemia, hypertension, cigarette smoking, then I think [they] could be recommended on a more consistent basis.”
Both studies were independently supported. Dr. Goldstein, Dr. Kuritzky, and Dr. Ledesma reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AUA 2023
Deep sleep may mitigate the impact of Alzheimer’s pathology
Investigators found that deep sleep, also known as non-REM (NREM) slow-wave sleep, can protect memory function in cognitively normal adults with a high beta-amyloid burden.
“Think of deep sleep almost like a life raft that keeps memory afloat, rather than memory getting dragged down by the weight of Alzheimer’s disease pathology,” senior investigator Matthew Walker, PhD, professor of neuroscience and psychology, University of California, Berkeley, said in a news release.
The study was published online in BMC Medicine.
Resilience factor
Studying resilience to existing brain pathology is “an exciting new research direction,” lead author Zsófia Zavecz, PhD, with the Center for Human Sleep Science at the University of California, Berkeley, said in an interview.
“That is, what factors explain the individual differences in cognitive function despite the same level of brain pathology, and how do some people with significant pathology have largely preserved memory?” she added.
The study included 62 cognitively normal older adults from the Berkeley Aging Cohort Study.
Sleep EEG recordings were obtained over 2 nights in a sleep lab and PET scans were used to quantify beta-amyloid. Half of the participants had high beta-amyloid burden and half were beta-amyloid negative.
After the sleep studies, all participants completed a memory task involving matching names to faces.
The results suggest that deep NREM slow-wave sleep significantly moderates the effect of beta-amyloid status on memory function.
Specifically, NREM slow-wave activity selectively supported superior memory function in adults with high beta-amyloid burden, who are most in need of cognitive reserve (B = 2.694, P = .019), the researchers report.
In contrast, adults without significant beta-amyloid pathological burden – and thus without the same need for cognitive reserve – did not similarly benefit from NREM slow-wave activity (B = –0.115, P = .876).
The findings remained significant after adjusting for age, sex, body mass index, gray matter atrophy, and previously identified cognitive reserve factors, such as education and physical activity.
Dr. Zavecz said there are several potential reasons why deep sleep may support cognitive reserve.
One is that during deep sleep specifically, memories are replayed in the brain, and this results in a “neural reorganization” that helps stabilize the memory and make it more permanent.
“Other explanations include deep sleep’s role in maintaining homeostasis in the brain’s capacity to form new neural connections and providing an optimal brain state for the clearance of toxins interfering with healthy brain functioning,” she noted.
“The extent to which sleep could offer a protective buffer against severe cognitive impairment remains to be tested. However, this study is the first step in hopefully a series of new research that will investigate sleep as a cognitive reserve factor,” said Dr. Zavecz.
Encouraging data
Reached for comment, Percy Griffin, PhD, Alzheimer’s Association director of scientific engagement, said although the study sample is small, the results are “encouraging because sleep is a modifiable factor and can therefore be targeted.”
“More work is needed in a larger population before we can fully leverage this stage of sleep to reduce the risk of developing cognitive decline,” Dr. Griffin said.
Also weighing in on this research, Shaheen Lakhan, MD, PhD, a neurologist and researcher in Boston, said the study is “exciting on two fronts – we may have an additional marker for the development of Alzheimer’s disease to predict risk and track disease, but also targets for early intervention with sleep architecture–enhancing therapies, be they drug, device, or digital.”
“For the sake of our brain health, we all must get very familiar with the concept of cognitive or brain reserve,” said Dr. Lakhan, who was not involved in the study.
“Brain reserve refers to our ability to buttress against the threat of dementia and classically it’s been associated with ongoing brain stimulation (i.e., higher education, cognitively demanding job),” he noted.
“This line of research now opens the door that optimal sleep health – especially deep NREM slow wave sleep – correlates with greater brain reserve against Alzheimer’s disease,” Dr. Lakhan said.
The study was supported by the National Institutes of Health and the University of California, Berkeley. Dr. Walker serves as an advisor to and has equity interest in Bryte, Shuni, Oura, and StimScience. Dr. Zavecz and Dr. Lakhan report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Investigators found that deep sleep, also known as non-REM (NREM) slow-wave sleep, can protect memory function in cognitively normal adults with a high beta-amyloid burden.
“Think of deep sleep almost like a life raft that keeps memory afloat, rather than memory getting dragged down by the weight of Alzheimer’s disease pathology,” senior investigator Matthew Walker, PhD, professor of neuroscience and psychology, University of California, Berkeley, said in a news release.
The study was published online in BMC Medicine.
Resilience factor
Studying resilience to existing brain pathology is “an exciting new research direction,” lead author Zsófia Zavecz, PhD, with the Center for Human Sleep Science at the University of California, Berkeley, said in an interview.
“That is, what factors explain the individual differences in cognitive function despite the same level of brain pathology, and how do some people with significant pathology have largely preserved memory?” she added.
The study included 62 cognitively normal older adults from the Berkeley Aging Cohort Study.
Sleep EEG recordings were obtained over 2 nights in a sleep lab and PET scans were used to quantify beta-amyloid. Half of the participants had high beta-amyloid burden and half were beta-amyloid negative.
After the sleep studies, all participants completed a memory task involving matching names to faces.
The results suggest that deep NREM slow-wave sleep significantly moderates the effect of beta-amyloid status on memory function.
Specifically, NREM slow-wave activity selectively supported superior memory function in adults with high beta-amyloid burden, who are most in need of cognitive reserve (B = 2.694, P = .019), the researchers report.
In contrast, adults without significant beta-amyloid pathological burden – and thus without the same need for cognitive reserve – did not similarly benefit from NREM slow-wave activity (B = –0.115, P = .876).
The findings remained significant after adjusting for age, sex, body mass index, gray matter atrophy, and previously identified cognitive reserve factors, such as education and physical activity.
Dr. Zavecz said there are several potential reasons why deep sleep may support cognitive reserve.
One is that during deep sleep specifically, memories are replayed in the brain, and this results in a “neural reorganization” that helps stabilize the memory and make it more permanent.
“Other explanations include deep sleep’s role in maintaining homeostasis in the brain’s capacity to form new neural connections and providing an optimal brain state for the clearance of toxins interfering with healthy brain functioning,” she noted.
“The extent to which sleep could offer a protective buffer against severe cognitive impairment remains to be tested. However, this study is the first step in hopefully a series of new research that will investigate sleep as a cognitive reserve factor,” said Dr. Zavecz.
Encouraging data
Reached for comment, Percy Griffin, PhD, Alzheimer’s Association director of scientific engagement, said although the study sample is small, the results are “encouraging because sleep is a modifiable factor and can therefore be targeted.”
“More work is needed in a larger population before we can fully leverage this stage of sleep to reduce the risk of developing cognitive decline,” Dr. Griffin said.
Also weighing in on this research, Shaheen Lakhan, MD, PhD, a neurologist and researcher in Boston, said the study is “exciting on two fronts – we may have an additional marker for the development of Alzheimer’s disease to predict risk and track disease, but also targets for early intervention with sleep architecture–enhancing therapies, be they drug, device, or digital.”
“For the sake of our brain health, we all must get very familiar with the concept of cognitive or brain reserve,” said Dr. Lakhan, who was not involved in the study.
“Brain reserve refers to our ability to buttress against the threat of dementia and classically it’s been associated with ongoing brain stimulation (i.e., higher education, cognitively demanding job),” he noted.
“This line of research now opens the door that optimal sleep health – especially deep NREM slow wave sleep – correlates with greater brain reserve against Alzheimer’s disease,” Dr. Lakhan said.
The study was supported by the National Institutes of Health and the University of California, Berkeley. Dr. Walker serves as an advisor to and has equity interest in Bryte, Shuni, Oura, and StimScience. Dr. Zavecz and Dr. Lakhan report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Investigators found that deep sleep, also known as non-REM (NREM) slow-wave sleep, can protect memory function in cognitively normal adults with a high beta-amyloid burden.
“Think of deep sleep almost like a life raft that keeps memory afloat, rather than memory getting dragged down by the weight of Alzheimer’s disease pathology,” senior investigator Matthew Walker, PhD, professor of neuroscience and psychology, University of California, Berkeley, said in a news release.
The study was published online in BMC Medicine.
Resilience factor
Studying resilience to existing brain pathology is “an exciting new research direction,” lead author Zsófia Zavecz, PhD, with the Center for Human Sleep Science at the University of California, Berkeley, said in an interview.
“That is, what factors explain the individual differences in cognitive function despite the same level of brain pathology, and how do some people with significant pathology have largely preserved memory?” she added.
The study included 62 cognitively normal older adults from the Berkeley Aging Cohort Study.
Sleep EEG recordings were obtained over 2 nights in a sleep lab and PET scans were used to quantify beta-amyloid. Half of the participants had high beta-amyloid burden and half were beta-amyloid negative.
After the sleep studies, all participants completed a memory task involving matching names to faces.
The results suggest that deep NREM slow-wave sleep significantly moderates the effect of beta-amyloid status on memory function.
Specifically, NREM slow-wave activity selectively supported superior memory function in adults with high beta-amyloid burden, who are most in need of cognitive reserve (B = 2.694, P = .019), the researchers report.
In contrast, adults without significant beta-amyloid pathological burden – and thus without the same need for cognitive reserve – did not similarly benefit from NREM slow-wave activity (B = –0.115, P = .876).
The findings remained significant after adjusting for age, sex, body mass index, gray matter atrophy, and previously identified cognitive reserve factors, such as education and physical activity.
Dr. Zavecz said there are several potential reasons why deep sleep may support cognitive reserve.
One is that during deep sleep specifically, memories are replayed in the brain, and this results in a “neural reorganization” that helps stabilize the memory and make it more permanent.
“Other explanations include deep sleep’s role in maintaining homeostasis in the brain’s capacity to form new neural connections and providing an optimal brain state for the clearance of toxins interfering with healthy brain functioning,” she noted.
“The extent to which sleep could offer a protective buffer against severe cognitive impairment remains to be tested. However, this study is the first step in hopefully a series of new research that will investigate sleep as a cognitive reserve factor,” said Dr. Zavecz.
Encouraging data
Reached for comment, Percy Griffin, PhD, Alzheimer’s Association director of scientific engagement, said although the study sample is small, the results are “encouraging because sleep is a modifiable factor and can therefore be targeted.”
“More work is needed in a larger population before we can fully leverage this stage of sleep to reduce the risk of developing cognitive decline,” Dr. Griffin said.
Also weighing in on this research, Shaheen Lakhan, MD, PhD, a neurologist and researcher in Boston, said the study is “exciting on two fronts – we may have an additional marker for the development of Alzheimer’s disease to predict risk and track disease, but also targets for early intervention with sleep architecture–enhancing therapies, be they drug, device, or digital.”
“For the sake of our brain health, we all must get very familiar with the concept of cognitive or brain reserve,” said Dr. Lakhan, who was not involved in the study.
“Brain reserve refers to our ability to buttress against the threat of dementia and classically it’s been associated with ongoing brain stimulation (i.e., higher education, cognitively demanding job),” he noted.
“This line of research now opens the door that optimal sleep health – especially deep NREM slow wave sleep – correlates with greater brain reserve against Alzheimer’s disease,” Dr. Lakhan said.
The study was supported by the National Institutes of Health and the University of California, Berkeley. Dr. Walker serves as an advisor to and has equity interest in Bryte, Shuni, Oura, and StimScience. Dr. Zavecz and Dr. Lakhan report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM BMC MEDICINE
Parkinson’s in Marines linked to toxic drinking water at Camp Lejeune
in Jacksonville, N.C.
In one of the best-documented, large-scale contaminations in U.S. history, the drinking water at the Marine Corps base was contaminated with TCE and other volatile organic compounds from about 1953 to 1987.
The new study of more than 340,000 service members found the risk of PD was 70% higher in Marines stationed at Camp Lejeune in North Carolina during the years 1975-1985, compared with Marines stationed at Camp Pendleton in Oceanside, Calif.
“This is by far the largest study to look at the association of TCE and PD and the evidence is pretty strong,” lead investigator Samuel M. Goldman, MD, MPH, with University of California, San Francisco, said in an interview.
The link is supported by animal models that show that TCE can induce a neurodegenerative syndrome that is “very similar pathologically to what we see in PD,” Dr. Goldman said.
The study was published online in JAMA Neurology.
‘Hundreds of thousands’ at risk
At Camp Lejeune during the years 1975-1985, the period of maximal contamination, the estimated monthly median TCE level was more than 70-fold the Environmental Protection Agency maximum contaminant level. Maximum contaminant levels were also exceeded for perchloroethylene (PCE) and vinyl chloride.
Dr. Goldman and colleagues had health data on 158,122 veterans – 84,824 from Camp Lejeune and 73,298 from Camp Pendleton – who served for at least 3 months between 1975 and 1985, with follow up from Jan. 1, 1997, to Feb. 17, 2021.
Demographic characteristics were similar between the two groups; most were White men with an average age of 59 years.
A total of 430 veterans had PD: 279 from Camp Lejeune (prevalence, 0.33%) and 151 from Camp Pendleton (prevalence, 0.21%).
In multivariable models, Camp Lejeune veterans had a 70% higher risk for PD (odds ratio, 1.70; 95% confidence interval, 1.39-2.07; P < .001).
“Remarkably,” the researchers noted, among veterans without PD, residence at Camp Lejeune was also associated with a significantly higher risk of having several well-established prodromal features of PD, including tremor, suggesting they may be in a prediagnostic phase of evolving PD pathology.
Importantly, they added, in addition to the exposed service members, “hundreds of thousands of family members and civilian workers exposed to contaminated water at Camp Lejeune may also be at increased risk of PD, cancers, and other health consequences. Continued prospective follow-up of this population is essential.”
‘An unreasonable risk’
The new study supports a prior, and much smaller, study by Dr. Goldman and colleagues showing TCE exposure was associated with a sixfold increased risk for PD.
TCE is a ubiquitous environmental contaminant. The EPA Toxics Release Inventory estimates 2.05 million pounds of TCE was released into the environment from industrial sites in 2017.
In an accompanying editorial, E. Ray Dorsey, MD, with the University of Rochester (N.Y.) and coauthors noted the work of Dr. Goldman and colleagues “increases the certainty” that environmental exposure to TCE and the similar compound PCE “contribute importantly to the cause of the world’s fastest-growing brain disease.”
In December, the EPA found that PCE posed “an unreasonable risk” to human health, and 1 month later, it reached the same conclusion for TCE.
“These actions could lay the foundation for increased regulation and possibly a ban of these two chemicals that have contributed to immeasurable death and disability for generations,” Dr. Dorsey and colleagues noted.
“A U.S. ban would be a step forward but would not address the tens of thousands of TCE/PCE-contaminated sites in the U.S. and around the world or the rising global use of the toxic solvents,” they added.
This research was supported by Department of Veterans Affairs. Dr. Goldman reported no relevant financial relationships. Dr. Dorsey has received personal fees from organizations including the American Neurological Association, Elsevier, International Parkinson and Movement Disorder Society, Massachusetts Medical Society, Michael J. Fox Foundation, National Institutes of Health, and WebMD, as well as numerous pharmaceutical companies.
A version of this article originally appeared on Medscape.com.
in Jacksonville, N.C.
In one of the best-documented, large-scale contaminations in U.S. history, the drinking water at the Marine Corps base was contaminated with TCE and other volatile organic compounds from about 1953 to 1987.
The new study of more than 340,000 service members found the risk of PD was 70% higher in Marines stationed at Camp Lejeune in North Carolina during the years 1975-1985, compared with Marines stationed at Camp Pendleton in Oceanside, Calif.
“This is by far the largest study to look at the association of TCE and PD and the evidence is pretty strong,” lead investigator Samuel M. Goldman, MD, MPH, with University of California, San Francisco, said in an interview.
The link is supported by animal models that show that TCE can induce a neurodegenerative syndrome that is “very similar pathologically to what we see in PD,” Dr. Goldman said.
The study was published online in JAMA Neurology.
‘Hundreds of thousands’ at risk
At Camp Lejeune during the years 1975-1985, the period of maximal contamination, the estimated monthly median TCE level was more than 70-fold the Environmental Protection Agency maximum contaminant level. Maximum contaminant levels were also exceeded for perchloroethylene (PCE) and vinyl chloride.
Dr. Goldman and colleagues had health data on 158,122 veterans – 84,824 from Camp Lejeune and 73,298 from Camp Pendleton – who served for at least 3 months between 1975 and 1985, with follow up from Jan. 1, 1997, to Feb. 17, 2021.
Demographic characteristics were similar between the two groups; most were White men with an average age of 59 years.
A total of 430 veterans had PD: 279 from Camp Lejeune (prevalence, 0.33%) and 151 from Camp Pendleton (prevalence, 0.21%).
In multivariable models, Camp Lejeune veterans had a 70% higher risk for PD (odds ratio, 1.70; 95% confidence interval, 1.39-2.07; P < .001).
“Remarkably,” the researchers noted, among veterans without PD, residence at Camp Lejeune was also associated with a significantly higher risk of having several well-established prodromal features of PD, including tremor, suggesting they may be in a prediagnostic phase of evolving PD pathology.
Importantly, they added, in addition to the exposed service members, “hundreds of thousands of family members and civilian workers exposed to contaminated water at Camp Lejeune may also be at increased risk of PD, cancers, and other health consequences. Continued prospective follow-up of this population is essential.”
‘An unreasonable risk’
The new study supports a prior, and much smaller, study by Dr. Goldman and colleagues showing TCE exposure was associated with a sixfold increased risk for PD.
TCE is a ubiquitous environmental contaminant. The EPA Toxics Release Inventory estimates 2.05 million pounds of TCE was released into the environment from industrial sites in 2017.
In an accompanying editorial, E. Ray Dorsey, MD, with the University of Rochester (N.Y.) and coauthors noted the work of Dr. Goldman and colleagues “increases the certainty” that environmental exposure to TCE and the similar compound PCE “contribute importantly to the cause of the world’s fastest-growing brain disease.”
In December, the EPA found that PCE posed “an unreasonable risk” to human health, and 1 month later, it reached the same conclusion for TCE.
“These actions could lay the foundation for increased regulation and possibly a ban of these two chemicals that have contributed to immeasurable death and disability for generations,” Dr. Dorsey and colleagues noted.
“A U.S. ban would be a step forward but would not address the tens of thousands of TCE/PCE-contaminated sites in the U.S. and around the world or the rising global use of the toxic solvents,” they added.
This research was supported by Department of Veterans Affairs. Dr. Goldman reported no relevant financial relationships. Dr. Dorsey has received personal fees from organizations including the American Neurological Association, Elsevier, International Parkinson and Movement Disorder Society, Massachusetts Medical Society, Michael J. Fox Foundation, National Institutes of Health, and WebMD, as well as numerous pharmaceutical companies.
A version of this article originally appeared on Medscape.com.
in Jacksonville, N.C.
In one of the best-documented, large-scale contaminations in U.S. history, the drinking water at the Marine Corps base was contaminated with TCE and other volatile organic compounds from about 1953 to 1987.
The new study of more than 340,000 service members found the risk of PD was 70% higher in Marines stationed at Camp Lejeune in North Carolina during the years 1975-1985, compared with Marines stationed at Camp Pendleton in Oceanside, Calif.
“This is by far the largest study to look at the association of TCE and PD and the evidence is pretty strong,” lead investigator Samuel M. Goldman, MD, MPH, with University of California, San Francisco, said in an interview.
The link is supported by animal models that show that TCE can induce a neurodegenerative syndrome that is “very similar pathologically to what we see in PD,” Dr. Goldman said.
The study was published online in JAMA Neurology.
‘Hundreds of thousands’ at risk
At Camp Lejeune during the years 1975-1985, the period of maximal contamination, the estimated monthly median TCE level was more than 70-fold the Environmental Protection Agency maximum contaminant level. Maximum contaminant levels were also exceeded for perchloroethylene (PCE) and vinyl chloride.
Dr. Goldman and colleagues had health data on 158,122 veterans – 84,824 from Camp Lejeune and 73,298 from Camp Pendleton – who served for at least 3 months between 1975 and 1985, with follow up from Jan. 1, 1997, to Feb. 17, 2021.
Demographic characteristics were similar between the two groups; most were White men with an average age of 59 years.
A total of 430 veterans had PD: 279 from Camp Lejeune (prevalence, 0.33%) and 151 from Camp Pendleton (prevalence, 0.21%).
In multivariable models, Camp Lejeune veterans had a 70% higher risk for PD (odds ratio, 1.70; 95% confidence interval, 1.39-2.07; P < .001).
“Remarkably,” the researchers noted, among veterans without PD, residence at Camp Lejeune was also associated with a significantly higher risk of having several well-established prodromal features of PD, including tremor, suggesting they may be in a prediagnostic phase of evolving PD pathology.
Importantly, they added, in addition to the exposed service members, “hundreds of thousands of family members and civilian workers exposed to contaminated water at Camp Lejeune may also be at increased risk of PD, cancers, and other health consequences. Continued prospective follow-up of this population is essential.”
‘An unreasonable risk’
The new study supports a prior, and much smaller, study by Dr. Goldman and colleagues showing TCE exposure was associated with a sixfold increased risk for PD.
TCE is a ubiquitous environmental contaminant. The EPA Toxics Release Inventory estimates 2.05 million pounds of TCE was released into the environment from industrial sites in 2017.
In an accompanying editorial, E. Ray Dorsey, MD, with the University of Rochester (N.Y.) and coauthors noted the work of Dr. Goldman and colleagues “increases the certainty” that environmental exposure to TCE and the similar compound PCE “contribute importantly to the cause of the world’s fastest-growing brain disease.”
In December, the EPA found that PCE posed “an unreasonable risk” to human health, and 1 month later, it reached the same conclusion for TCE.
“These actions could lay the foundation for increased regulation and possibly a ban of these two chemicals that have contributed to immeasurable death and disability for generations,” Dr. Dorsey and colleagues noted.
“A U.S. ban would be a step forward but would not address the tens of thousands of TCE/PCE-contaminated sites in the U.S. and around the world or the rising global use of the toxic solvents,” they added.
This research was supported by Department of Veterans Affairs. Dr. Goldman reported no relevant financial relationships. Dr. Dorsey has received personal fees from organizations including the American Neurological Association, Elsevier, International Parkinson and Movement Disorder Society, Massachusetts Medical Society, Michael J. Fox Foundation, National Institutes of Health, and WebMD, as well as numerous pharmaceutical companies.
A version of this article originally appeared on Medscape.com.
FROM JAMA NEUROLOGY
Video-based AI tool estimates LVEF from angiograms
a new study suggests.
In the test dataset, the video-based algorithm, called a deep neural network (DNN), discriminated reduced LVEF (< 40%) with an area under the receiver operating characteristic curve of 0.911.
In the external validation dataset, the DNN discriminated reduced LVEF with an AUROC of 0.906. However, the DNN tended to overestimate low LVEFs and to underestimate high LVEFs.
“We know the findings will be unexpected for cardiologists who don’t typically expect to get an estimate of systolic function or pump function just from an angiogram,” principal investigator Geoffrey H. Tison, MD, of the University of California, San Francisco, said in an interview.
In fact, he noted, “one of the challenges we face is a lack of trust by the health care community. They may not understand what drives the predictions behind our models. We have to translate that information in such a way that physicians trust that the algorithm is using the right features from the data they feed in to make the predictions.”
To help bolster that trust, “we display the ‘Model Facts,’ a nutrition-style label that describes how we train the algorithm, how it was validated, and the inclusion and exclusion criteria,” added lead author Robert Avram, MD, of the University of Montreal.
Model Facts is a safeguard against inappropriate use of the algorithm, Dr. Avram said. For example, if the algorithm was trained on patients between the ages of 40 and 90 and a clinician fed in data for a 35-year-old, a pop-up would appear warning the physician that the data being inputted are different from the data the algorithm was trained and validated on, and so any prediction “should be taken with a grain of salt.”
The study was published online in JAMA Cardiology.
Additional procedure
LVEF can be determined before coronary angiography with transthoracic echocardiography, but that is not always available, particularly for patients being seen emergently for acute coronary syndromes, the researchers wrote. LVEF can also be assessed using left ventriculography, an additional procedure that requires insertion of a pigtail catheter into the left ventricle and injection of more contrast and longer radiation exposure.
“Novel methods to assess LVEF at the point of care during coronary angiography would expand the available options to perform this important physiologic determination,” they wrote. “Video-based deep neural networks can learn subtle patterns from medical data to accomplish certain tasks beyond what physicians can achieve with that data, providing an opportunity to assess cardiac systolic function in real time from standard angiographic images without additional cost or procedures.”
The investigators conducted a cross-sectional study using UCSF patient data from 2012 to 2019. Data were randomly categorized into training, development, and test datasets.
External validation data were obtained from the University of Ottawa Heart Institute.
All adult patients who received a coronary angiogram and a transthoracic echocardiogram (TTE) within 3 months before or 1 month after receiving the angiogram were included.
A total of 4,042 angiograms with corresponding TTE LVEF from 3,679 UCSF patients were included in the analysis. The mean age of the patients was 64.3 years, and 65% were men.
The researchers’ video-based DNN, called CathEF, was used to discriminate reduced LVEF and to predict a continuous LVEF percentage from standard angiogram videos of the left coronary artery.
In the UCSF test dataset, CathEF discriminated reduced LVEF with an AUROC of 0.911; the diagnostic odds ratio for reduced LVEF was 22.7.
Furthermore, the CathEF-predicted that LVEF had a mean absolute error (MAE) of 8.5%, compared with TTE LVEF.
The CathEF-predicted LVEF differed 5% or less in comparison with the TTE LVEF in 38% of the test dataset studies; however, differences greater than 15% were seen in 15.2% of cases.
In the external validation, CathEF discriminated reduced LVEF with an AUROC of 0.906 and an MAE of 7%.
CathEF performance was consistent irrespective of patient characteristics, including sex, body mass index, low estimated glomerular filtration rate (< 45), acute coronary syndromes, obstructive coronary artery disease, and left ventricular hypertrophy.
However, as noted, it tended to overestimate low LVEFs and to underestimate high LVEFs.
“Further research can improve accuracy and reduce the variability of DNNs to maximize their clinical utility,” the authors concluded.
A validation study is underway at the Montreal Heart Institute, and similar studies are planned at UCSF and McGill University, Dr. Tison said. “We expect to present preliminary findings at medical conferences either before the end of the year or maybe for the American College of Cardiology meeting in March 2024.”
Potentially improved outcomes
In a comment, Alfonso H. Waller, MD, a member of the American College of Cardiology’s Imaging Council and director of cardiac imaging at New Jersey Medical School, Newark, said that, “at some centers, in patients presenting with an acute ST-segment elevation myocardial infarction, some argue that assessment of myocardial and valvular function with [left ventriculography] grams may provide important prognostic information and in part may help guide the management of the patient.
“Therefore, this novel approach may provide information that is not usually available without performing a classic LV gram ... [and] may lead to improved delivery of care, earlier therapies, and potentially improved outcomes and quality of life.”
If the technology is available in real time, “it could enable real-time, dynamic assessment of cardiac function during coronary angiography, which may be particularly helpful in acute STEMI cases where baseline cardiac function and renal function may be unknown and additional contrast may be detrimental,” he said.
However, patients who might benefit most from the technology are those with severely reduced LVEF, “and unfortunately, the LVEF may be overestimated in this group,” he said.
Dr. Waller also noted that the model was developed using echocardiograms obtained 3 months before or up to 1 month after the angiogram, during which time “LVEF may change significantly. Typically, if someone presents with an acute coronary syndrome, there can be myocardial stunning, which can lead to regional wall motion abnormalities and lowering of LVEF.”
The validation study is evaluating patients with acute coronary syndrome for whom an echocardiogram was performed within 48 hours of the angiogram, he added.
The study was supported by grants from the Fonds de la Recherche en Santé du Québec, the Montreal Heart Institute Research Centre, the Montreal Heart Institute Foundation, the Des Groseillers-Bérard Research Chair, the National Institutes of Health, and the Heart and Stroke Foundation of Ontario. Dr. Tison, Dr. Avram, and Dr. Waller disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
a new study suggests.
In the test dataset, the video-based algorithm, called a deep neural network (DNN), discriminated reduced LVEF (< 40%) with an area under the receiver operating characteristic curve of 0.911.
In the external validation dataset, the DNN discriminated reduced LVEF with an AUROC of 0.906. However, the DNN tended to overestimate low LVEFs and to underestimate high LVEFs.
“We know the findings will be unexpected for cardiologists who don’t typically expect to get an estimate of systolic function or pump function just from an angiogram,” principal investigator Geoffrey H. Tison, MD, of the University of California, San Francisco, said in an interview.
In fact, he noted, “one of the challenges we face is a lack of trust by the health care community. They may not understand what drives the predictions behind our models. We have to translate that information in such a way that physicians trust that the algorithm is using the right features from the data they feed in to make the predictions.”
To help bolster that trust, “we display the ‘Model Facts,’ a nutrition-style label that describes how we train the algorithm, how it was validated, and the inclusion and exclusion criteria,” added lead author Robert Avram, MD, of the University of Montreal.
Model Facts is a safeguard against inappropriate use of the algorithm, Dr. Avram said. For example, if the algorithm was trained on patients between the ages of 40 and 90 and a clinician fed in data for a 35-year-old, a pop-up would appear warning the physician that the data being inputted are different from the data the algorithm was trained and validated on, and so any prediction “should be taken with a grain of salt.”
The study was published online in JAMA Cardiology.
Additional procedure
LVEF can be determined before coronary angiography with transthoracic echocardiography, but that is not always available, particularly for patients being seen emergently for acute coronary syndromes, the researchers wrote. LVEF can also be assessed using left ventriculography, an additional procedure that requires insertion of a pigtail catheter into the left ventricle and injection of more contrast and longer radiation exposure.
“Novel methods to assess LVEF at the point of care during coronary angiography would expand the available options to perform this important physiologic determination,” they wrote. “Video-based deep neural networks can learn subtle patterns from medical data to accomplish certain tasks beyond what physicians can achieve with that data, providing an opportunity to assess cardiac systolic function in real time from standard angiographic images without additional cost or procedures.”
The investigators conducted a cross-sectional study using UCSF patient data from 2012 to 2019. Data were randomly categorized into training, development, and test datasets.
External validation data were obtained from the University of Ottawa Heart Institute.
All adult patients who received a coronary angiogram and a transthoracic echocardiogram (TTE) within 3 months before or 1 month after receiving the angiogram were included.
A total of 4,042 angiograms with corresponding TTE LVEF from 3,679 UCSF patients were included in the analysis. The mean age of the patients was 64.3 years, and 65% were men.
The researchers’ video-based DNN, called CathEF, was used to discriminate reduced LVEF and to predict a continuous LVEF percentage from standard angiogram videos of the left coronary artery.
In the UCSF test dataset, CathEF discriminated reduced LVEF with an AUROC of 0.911; the diagnostic odds ratio for reduced LVEF was 22.7.
Furthermore, the CathEF-predicted that LVEF had a mean absolute error (MAE) of 8.5%, compared with TTE LVEF.
The CathEF-predicted LVEF differed 5% or less in comparison with the TTE LVEF in 38% of the test dataset studies; however, differences greater than 15% were seen in 15.2% of cases.
In the external validation, CathEF discriminated reduced LVEF with an AUROC of 0.906 and an MAE of 7%.
CathEF performance was consistent irrespective of patient characteristics, including sex, body mass index, low estimated glomerular filtration rate (< 45), acute coronary syndromes, obstructive coronary artery disease, and left ventricular hypertrophy.
However, as noted, it tended to overestimate low LVEFs and to underestimate high LVEFs.
“Further research can improve accuracy and reduce the variability of DNNs to maximize their clinical utility,” the authors concluded.
A validation study is underway at the Montreal Heart Institute, and similar studies are planned at UCSF and McGill University, Dr. Tison said. “We expect to present preliminary findings at medical conferences either before the end of the year or maybe for the American College of Cardiology meeting in March 2024.”
Potentially improved outcomes
In a comment, Alfonso H. Waller, MD, a member of the American College of Cardiology’s Imaging Council and director of cardiac imaging at New Jersey Medical School, Newark, said that, “at some centers, in patients presenting with an acute ST-segment elevation myocardial infarction, some argue that assessment of myocardial and valvular function with [left ventriculography] grams may provide important prognostic information and in part may help guide the management of the patient.
“Therefore, this novel approach may provide information that is not usually available without performing a classic LV gram ... [and] may lead to improved delivery of care, earlier therapies, and potentially improved outcomes and quality of life.”
If the technology is available in real time, “it could enable real-time, dynamic assessment of cardiac function during coronary angiography, which may be particularly helpful in acute STEMI cases where baseline cardiac function and renal function may be unknown and additional contrast may be detrimental,” he said.
However, patients who might benefit most from the technology are those with severely reduced LVEF, “and unfortunately, the LVEF may be overestimated in this group,” he said.
Dr. Waller also noted that the model was developed using echocardiograms obtained 3 months before or up to 1 month after the angiogram, during which time “LVEF may change significantly. Typically, if someone presents with an acute coronary syndrome, there can be myocardial stunning, which can lead to regional wall motion abnormalities and lowering of LVEF.”
The validation study is evaluating patients with acute coronary syndrome for whom an echocardiogram was performed within 48 hours of the angiogram, he added.
The study was supported by grants from the Fonds de la Recherche en Santé du Québec, the Montreal Heart Institute Research Centre, the Montreal Heart Institute Foundation, the Des Groseillers-Bérard Research Chair, the National Institutes of Health, and the Heart and Stroke Foundation of Ontario. Dr. Tison, Dr. Avram, and Dr. Waller disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
a new study suggests.
In the test dataset, the video-based algorithm, called a deep neural network (DNN), discriminated reduced LVEF (< 40%) with an area under the receiver operating characteristic curve of 0.911.
In the external validation dataset, the DNN discriminated reduced LVEF with an AUROC of 0.906. However, the DNN tended to overestimate low LVEFs and to underestimate high LVEFs.
“We know the findings will be unexpected for cardiologists who don’t typically expect to get an estimate of systolic function or pump function just from an angiogram,” principal investigator Geoffrey H. Tison, MD, of the University of California, San Francisco, said in an interview.
In fact, he noted, “one of the challenges we face is a lack of trust by the health care community. They may not understand what drives the predictions behind our models. We have to translate that information in such a way that physicians trust that the algorithm is using the right features from the data they feed in to make the predictions.”
To help bolster that trust, “we display the ‘Model Facts,’ a nutrition-style label that describes how we train the algorithm, how it was validated, and the inclusion and exclusion criteria,” added lead author Robert Avram, MD, of the University of Montreal.
Model Facts is a safeguard against inappropriate use of the algorithm, Dr. Avram said. For example, if the algorithm was trained on patients between the ages of 40 and 90 and a clinician fed in data for a 35-year-old, a pop-up would appear warning the physician that the data being inputted are different from the data the algorithm was trained and validated on, and so any prediction “should be taken with a grain of salt.”
The study was published online in JAMA Cardiology.
Additional procedure
LVEF can be determined before coronary angiography with transthoracic echocardiography, but that is not always available, particularly for patients being seen emergently for acute coronary syndromes, the researchers wrote. LVEF can also be assessed using left ventriculography, an additional procedure that requires insertion of a pigtail catheter into the left ventricle and injection of more contrast and longer radiation exposure.
“Novel methods to assess LVEF at the point of care during coronary angiography would expand the available options to perform this important physiologic determination,” they wrote. “Video-based deep neural networks can learn subtle patterns from medical data to accomplish certain tasks beyond what physicians can achieve with that data, providing an opportunity to assess cardiac systolic function in real time from standard angiographic images without additional cost or procedures.”
The investigators conducted a cross-sectional study using UCSF patient data from 2012 to 2019. Data were randomly categorized into training, development, and test datasets.
External validation data were obtained from the University of Ottawa Heart Institute.
All adult patients who received a coronary angiogram and a transthoracic echocardiogram (TTE) within 3 months before or 1 month after receiving the angiogram were included.
A total of 4,042 angiograms with corresponding TTE LVEF from 3,679 UCSF patients were included in the analysis. The mean age of the patients was 64.3 years, and 65% were men.
The researchers’ video-based DNN, called CathEF, was used to discriminate reduced LVEF and to predict a continuous LVEF percentage from standard angiogram videos of the left coronary artery.
In the UCSF test dataset, CathEF discriminated reduced LVEF with an AUROC of 0.911; the diagnostic odds ratio for reduced LVEF was 22.7.
Furthermore, the CathEF-predicted that LVEF had a mean absolute error (MAE) of 8.5%, compared with TTE LVEF.
The CathEF-predicted LVEF differed 5% or less in comparison with the TTE LVEF in 38% of the test dataset studies; however, differences greater than 15% were seen in 15.2% of cases.
In the external validation, CathEF discriminated reduced LVEF with an AUROC of 0.906 and an MAE of 7%.
CathEF performance was consistent irrespective of patient characteristics, including sex, body mass index, low estimated glomerular filtration rate (< 45), acute coronary syndromes, obstructive coronary artery disease, and left ventricular hypertrophy.
However, as noted, it tended to overestimate low LVEFs and to underestimate high LVEFs.
“Further research can improve accuracy and reduce the variability of DNNs to maximize their clinical utility,” the authors concluded.
A validation study is underway at the Montreal Heart Institute, and similar studies are planned at UCSF and McGill University, Dr. Tison said. “We expect to present preliminary findings at medical conferences either before the end of the year or maybe for the American College of Cardiology meeting in March 2024.”
Potentially improved outcomes
In a comment, Alfonso H. Waller, MD, a member of the American College of Cardiology’s Imaging Council and director of cardiac imaging at New Jersey Medical School, Newark, said that, “at some centers, in patients presenting with an acute ST-segment elevation myocardial infarction, some argue that assessment of myocardial and valvular function with [left ventriculography] grams may provide important prognostic information and in part may help guide the management of the patient.
“Therefore, this novel approach may provide information that is not usually available without performing a classic LV gram ... [and] may lead to improved delivery of care, earlier therapies, and potentially improved outcomes and quality of life.”
If the technology is available in real time, “it could enable real-time, dynamic assessment of cardiac function during coronary angiography, which may be particularly helpful in acute STEMI cases where baseline cardiac function and renal function may be unknown and additional contrast may be detrimental,” he said.
However, patients who might benefit most from the technology are those with severely reduced LVEF, “and unfortunately, the LVEF may be overestimated in this group,” he said.
Dr. Waller also noted that the model was developed using echocardiograms obtained 3 months before or up to 1 month after the angiogram, during which time “LVEF may change significantly. Typically, if someone presents with an acute coronary syndrome, there can be myocardial stunning, which can lead to regional wall motion abnormalities and lowering of LVEF.”
The validation study is evaluating patients with acute coronary syndrome for whom an echocardiogram was performed within 48 hours of the angiogram, he added.
The study was supported by grants from the Fonds de la Recherche en Santé du Québec, the Montreal Heart Institute Research Centre, the Montreal Heart Institute Foundation, the Des Groseillers-Bérard Research Chair, the National Institutes of Health, and the Heart and Stroke Foundation of Ontario. Dr. Tison, Dr. Avram, and Dr. Waller disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA CARDIOLOGY
Foot ulcers red flag for eye disease in diabetes
Sores on the feet can signal problems with the eyes in patients with diabetes.
Prior research and anecdotal experience show that diabetic foot ulcers and diabetic retinopathy frequently co-occur.
David J. Ramsey, MD, PhD, MPH, director of ophthalmic research at Lahey Hospital & Medical Center, Burlington, Mass., said when clinicians detect either condition, they should involve a team that can intervene to help protect a patient’s vision and mobility.
For example, they should ensure patients receive comprehensive eye and foot evaluations and help them optimize diabetes management.
The new study, presented at the annual meeting of the Association for Research in Vision and Ophthalmology, “adds an important dimension” to understanding the association between the conditions, said Dr. Ramsey, who recently reviewed correlations between diabetic foot ulcers and diabetic retinopathy and their underlying causes.
“Patients with diabetic foot ulcers appear to receive less attention to their diabetic retinopathy and may receive fewer treatments with eye injections targeting vascular endothelial growth factor (VEGF), an important driver of progression of diabetic retinopathy,” said Dr. Ramsey, who is also an associate professor of ophthalmology at Tufts University School of Medicine, Boston. He was not involved in the study presented at ARVO 2023.
In the new study, Christopher T. Zhu, a medical student at UT Health San Antonio, and colleagues analyzed data from 426 eyes of 213 patients with type 2 diabetes who had had at least two eye exams between 2012 and 2022; 72 of the patients had diabetic foot ulcers. Patients were followed for about 4 years on average.
Patients with diabetic foot ulcers had a higher percentage of eyes with macular edema on their initial exam (32.6% vs. 28%). By the final exam, the percentage of eyes with macular edema was significantly greater in the group with diabetic foot ulcers (64.6% vs. 37.6%; P < .0001), Mr. Zhu’s group reported.
Eyes with nonproliferative diabetic retinopathy progressed to proliferative diabetic retinopathy, the worst grade, at a higher rate in the group with foot ulcers (50.6% vs. 35.6%; P = .03). In addition, patients with foot ulcers were more likely to experience vitreous hemorrhage (55.6% vs. 38.7%), the researchers found.
Despite patients with foot ulcers tending to have worse disease, they received fewer treatments for retinopathy. Those without ulcers received an average of 6.9 anti-VEGF injections per eye, while those with ulcers averaged 4.3.
Foot ulcers may hinder the ability of patients to get to appointments to receive the injections, Mr. Zhu and colleagues wrote. “For many patients in our part of the country [South Texas], a lack of transportation is a particular barrier to health care access,” Mr. Zhu told this news organization.
Mr. Zhu’s team conducted their study after noticing that patients with diabetes and foot ulcers who presented to their eye clinics “appeared to progress faster to worse grades of retinopathy” than patients with diabetes who did not have ulcers.
“Similar to how foot ulcers develop due to a severe disruption in blood flow [vascular] and a loss of sensation [neurologic], diabetic retinopathy may have a relation to microvascular disease, neurologic degeneration, and inflammation,” he said.
The findings confirm “that poor perfusion of the eye and foot are linked and can cause ischemic retinopathy leading to the development of proliferative diabetic retinopathy and vitreous hemorrhages, both serious, vision-threatening conditions,” Dr. Ramsey said.
To some extent, fewer treatments with anti-VEGF agents may account for why patients with foot ulcers have more eye complications, Dr. Ramsey added. “Additional research needs to be done to further dissect the cause and the effect, but it’s a very important finding that we need to increase awareness about,” he said.
Dr. Ramsey and Mr. Zhu reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sores on the feet can signal problems with the eyes in patients with diabetes.
Prior research and anecdotal experience show that diabetic foot ulcers and diabetic retinopathy frequently co-occur.
David J. Ramsey, MD, PhD, MPH, director of ophthalmic research at Lahey Hospital & Medical Center, Burlington, Mass., said when clinicians detect either condition, they should involve a team that can intervene to help protect a patient’s vision and mobility.
For example, they should ensure patients receive comprehensive eye and foot evaluations and help them optimize diabetes management.
The new study, presented at the annual meeting of the Association for Research in Vision and Ophthalmology, “adds an important dimension” to understanding the association between the conditions, said Dr. Ramsey, who recently reviewed correlations between diabetic foot ulcers and diabetic retinopathy and their underlying causes.
“Patients with diabetic foot ulcers appear to receive less attention to their diabetic retinopathy and may receive fewer treatments with eye injections targeting vascular endothelial growth factor (VEGF), an important driver of progression of diabetic retinopathy,” said Dr. Ramsey, who is also an associate professor of ophthalmology at Tufts University School of Medicine, Boston. He was not involved in the study presented at ARVO 2023.
In the new study, Christopher T. Zhu, a medical student at UT Health San Antonio, and colleagues analyzed data from 426 eyes of 213 patients with type 2 diabetes who had had at least two eye exams between 2012 and 2022; 72 of the patients had diabetic foot ulcers. Patients were followed for about 4 years on average.
Patients with diabetic foot ulcers had a higher percentage of eyes with macular edema on their initial exam (32.6% vs. 28%). By the final exam, the percentage of eyes with macular edema was significantly greater in the group with diabetic foot ulcers (64.6% vs. 37.6%; P < .0001), Mr. Zhu’s group reported.
Eyes with nonproliferative diabetic retinopathy progressed to proliferative diabetic retinopathy, the worst grade, at a higher rate in the group with foot ulcers (50.6% vs. 35.6%; P = .03). In addition, patients with foot ulcers were more likely to experience vitreous hemorrhage (55.6% vs. 38.7%), the researchers found.
Despite patients with foot ulcers tending to have worse disease, they received fewer treatments for retinopathy. Those without ulcers received an average of 6.9 anti-VEGF injections per eye, while those with ulcers averaged 4.3.
Foot ulcers may hinder the ability of patients to get to appointments to receive the injections, Mr. Zhu and colleagues wrote. “For many patients in our part of the country [South Texas], a lack of transportation is a particular barrier to health care access,” Mr. Zhu told this news organization.
Mr. Zhu’s team conducted their study after noticing that patients with diabetes and foot ulcers who presented to their eye clinics “appeared to progress faster to worse grades of retinopathy” than patients with diabetes who did not have ulcers.
“Similar to how foot ulcers develop due to a severe disruption in blood flow [vascular] and a loss of sensation [neurologic], diabetic retinopathy may have a relation to microvascular disease, neurologic degeneration, and inflammation,” he said.
The findings confirm “that poor perfusion of the eye and foot are linked and can cause ischemic retinopathy leading to the development of proliferative diabetic retinopathy and vitreous hemorrhages, both serious, vision-threatening conditions,” Dr. Ramsey said.
To some extent, fewer treatments with anti-VEGF agents may account for why patients with foot ulcers have more eye complications, Dr. Ramsey added. “Additional research needs to be done to further dissect the cause and the effect, but it’s a very important finding that we need to increase awareness about,” he said.
Dr. Ramsey and Mr. Zhu reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sores on the feet can signal problems with the eyes in patients with diabetes.
Prior research and anecdotal experience show that diabetic foot ulcers and diabetic retinopathy frequently co-occur.
David J. Ramsey, MD, PhD, MPH, director of ophthalmic research at Lahey Hospital & Medical Center, Burlington, Mass., said when clinicians detect either condition, they should involve a team that can intervene to help protect a patient’s vision and mobility.
For example, they should ensure patients receive comprehensive eye and foot evaluations and help them optimize diabetes management.
The new study, presented at the annual meeting of the Association for Research in Vision and Ophthalmology, “adds an important dimension” to understanding the association between the conditions, said Dr. Ramsey, who recently reviewed correlations between diabetic foot ulcers and diabetic retinopathy and their underlying causes.
“Patients with diabetic foot ulcers appear to receive less attention to their diabetic retinopathy and may receive fewer treatments with eye injections targeting vascular endothelial growth factor (VEGF), an important driver of progression of diabetic retinopathy,” said Dr. Ramsey, who is also an associate professor of ophthalmology at Tufts University School of Medicine, Boston. He was not involved in the study presented at ARVO 2023.
In the new study, Christopher T. Zhu, a medical student at UT Health San Antonio, and colleagues analyzed data from 426 eyes of 213 patients with type 2 diabetes who had had at least two eye exams between 2012 and 2022; 72 of the patients had diabetic foot ulcers. Patients were followed for about 4 years on average.
Patients with diabetic foot ulcers had a higher percentage of eyes with macular edema on their initial exam (32.6% vs. 28%). By the final exam, the percentage of eyes with macular edema was significantly greater in the group with diabetic foot ulcers (64.6% vs. 37.6%; P < .0001), Mr. Zhu’s group reported.
Eyes with nonproliferative diabetic retinopathy progressed to proliferative diabetic retinopathy, the worst grade, at a higher rate in the group with foot ulcers (50.6% vs. 35.6%; P = .03). In addition, patients with foot ulcers were more likely to experience vitreous hemorrhage (55.6% vs. 38.7%), the researchers found.
Despite patients with foot ulcers tending to have worse disease, they received fewer treatments for retinopathy. Those without ulcers received an average of 6.9 anti-VEGF injections per eye, while those with ulcers averaged 4.3.
Foot ulcers may hinder the ability of patients to get to appointments to receive the injections, Mr. Zhu and colleagues wrote. “For many patients in our part of the country [South Texas], a lack of transportation is a particular barrier to health care access,” Mr. Zhu told this news organization.
Mr. Zhu’s team conducted their study after noticing that patients with diabetes and foot ulcers who presented to their eye clinics “appeared to progress faster to worse grades of retinopathy” than patients with diabetes who did not have ulcers.
“Similar to how foot ulcers develop due to a severe disruption in blood flow [vascular] and a loss of sensation [neurologic], diabetic retinopathy may have a relation to microvascular disease, neurologic degeneration, and inflammation,” he said.
The findings confirm “that poor perfusion of the eye and foot are linked and can cause ischemic retinopathy leading to the development of proliferative diabetic retinopathy and vitreous hemorrhages, both serious, vision-threatening conditions,” Dr. Ramsey said.
To some extent, fewer treatments with anti-VEGF agents may account for why patients with foot ulcers have more eye complications, Dr. Ramsey added. “Additional research needs to be done to further dissect the cause and the effect, but it’s a very important finding that we need to increase awareness about,” he said.
Dr. Ramsey and Mr. Zhu reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ARVO 2023