User login
AGA and CDC partner to expand access to CRC screening
The American Gastroenterological Association has a unique opportunity for you to use your skills to help prevent cancer in your community. It is once again working with the Centers for Disease Control and Prevention’s state Colorectal Cancer Control Programs (CRCCP) to conduct a screening and awareness event during Colorectal Cancer Awareness Month 2013. The partnership provides a great opportunity for the AGA to reach the underserved in select states and to raise awareness of the need for colorectal cancer screening around the country.
We are pleased to inform you that the CRCCP programs in the following states are interested in participating in this exciting screening and awareness event: Arizona, Florida, Massachusetts, Minnesota, Nebraska, Nevada, New Hampshire, Oregon, Pennsylvania, and Utah.
We are in the early stages of organizing the event, which will allow AGA members to provide colonoscopies to uninsured and underinsured individuals, aged 50 years and over, who could otherwise not afford this vital service. The state cancer control programs will match gastroenterologists in their areas with patients who are in need of free colonoscopy screenings.
We are looking for AGA members to take part in this exciting program. We hope you will volunteer to provide free colonoscopies in your communities during March 2013; the dates will be coordinated with the CRCCP program. If you would like to take a greater role in the management of the program, we are trying to identify members who are interested in helping to coordinate these efforts by working closely with the CRCCP program.
If you are interested in learning more, please contact Aimee Frank, AGA senior director of public relations, either via e-mail ([email protected]) or phone (301-941-2620).
The American Gastroenterological Association has a unique opportunity for you to use your skills to help prevent cancer in your community. It is once again working with the Centers for Disease Control and Prevention’s state Colorectal Cancer Control Programs (CRCCP) to conduct a screening and awareness event during Colorectal Cancer Awareness Month 2013. The partnership provides a great opportunity for the AGA to reach the underserved in select states and to raise awareness of the need for colorectal cancer screening around the country.
We are pleased to inform you that the CRCCP programs in the following states are interested in participating in this exciting screening and awareness event: Arizona, Florida, Massachusetts, Minnesota, Nebraska, Nevada, New Hampshire, Oregon, Pennsylvania, and Utah.
We are in the early stages of organizing the event, which will allow AGA members to provide colonoscopies to uninsured and underinsured individuals, aged 50 years and over, who could otherwise not afford this vital service. The state cancer control programs will match gastroenterologists in their areas with patients who are in need of free colonoscopy screenings.
We are looking for AGA members to take part in this exciting program. We hope you will volunteer to provide free colonoscopies in your communities during March 2013; the dates will be coordinated with the CRCCP program. If you would like to take a greater role in the management of the program, we are trying to identify members who are interested in helping to coordinate these efforts by working closely with the CRCCP program.
If you are interested in learning more, please contact Aimee Frank, AGA senior director of public relations, either via e-mail ([email protected]) or phone (301-941-2620).
The American Gastroenterological Association has a unique opportunity for you to use your skills to help prevent cancer in your community. It is once again working with the Centers for Disease Control and Prevention’s state Colorectal Cancer Control Programs (CRCCP) to conduct a screening and awareness event during Colorectal Cancer Awareness Month 2013. The partnership provides a great opportunity for the AGA to reach the underserved in select states and to raise awareness of the need for colorectal cancer screening around the country.
We are pleased to inform you that the CRCCP programs in the following states are interested in participating in this exciting screening and awareness event: Arizona, Florida, Massachusetts, Minnesota, Nebraska, Nevada, New Hampshire, Oregon, Pennsylvania, and Utah.
We are in the early stages of organizing the event, which will allow AGA members to provide colonoscopies to uninsured and underinsured individuals, aged 50 years and over, who could otherwise not afford this vital service. The state cancer control programs will match gastroenterologists in their areas with patients who are in need of free colonoscopy screenings.
We are looking for AGA members to take part in this exciting program. We hope you will volunteer to provide free colonoscopies in your communities during March 2013; the dates will be coordinated with the CRCCP program. If you would like to take a greater role in the management of the program, we are trying to identify members who are interested in helping to coordinate these efforts by working closely with the CRCCP program.
If you are interested in learning more, please contact Aimee Frank, AGA senior director of public relations, either via e-mail ([email protected]) or phone (301-941-2620).
Grant opportunities from the AGA Research Foundation
The AGA Research Foundation provides career development research grants to young investigators. The foundation offers a variety of grants, some of which are targeted to specific digestive diseases.
These awards are made possible through the generosity of AGA members, patients, foundations, and industry leaders. All gifts to the AGA Research Foundation go toward an endowment that will support young investigators on an ongoing basis, making it a gift that keeps on giving.
There are several new awards being offered this year:
– AGA–Caroline Craig Augustyn and Damian Augustyn Award in Digestive Cancer: This grant provides a one-time $40,000 award to a young investigator who currently holds a federal or nonfederal career development award devoted to conducting research related to digestive cancer. The application deadline is Jan. 13.
– AGA–Elsevier Gut Microbiome Pilot Research Award: This research initiative grant for $25,000 for 1 year is offered to early-stage and established investigators to support pilot research projects pertaining to the gut microbiome. The application deadline is Jan. 13.
– AGA Investing in the Future Student Research Fellowship: Awards are offered to support underrepresented minority students performing digestive disease or nutrition research for a minimum of 10 weeks. The award offers $5,000 and is supported by the National Institute of Diabetes and Digestive and Kidney Diseases (Award R25DK096968). The application deadline is Feb. 15.
In addition, we are continuing to offer the following awards with upcoming application deadlines:
– June & Donald O. Castell, MD Esophageal Clinical Research Award: This award provides 1 year of funding to a junior faculty member in that research area. This year marks the 13th annual Castell award supporting clinical esophageal researchers. Support of this program is made possible by the June and Donald O. Castell, MD, Gastroenterology Research and Education Trust. The application deadline is Jan. 20.
– Eli and Edythe L. Broad Student Research Fellowship Awards: This fellowship offers $2,500 stipends to high school and undergraduate students and $3,000 to medical and graduate students to spend time performing research in digestive diseases or nutrition for a minimum of 10 weeks. This award stimulates interest in research careers in digestive diseases by providing monetary support for research projects. The application deadline is March 15.
Over the last 28 years, the AGA and its foundations have supported 711 scientists with $40,569,000 in grants. This support is an investment in scientific discovery that ensures the ongoing pipeline of young investigators who will contribute to future advances.
For a complete list of AGA Research Foundation awards or to support the Research Awards Program with a tax-deductible gift, visit www.gastro.org/foundation.
The AGA Research Foundation provides career development research grants to young investigators. The foundation offers a variety of grants, some of which are targeted to specific digestive diseases.
These awards are made possible through the generosity of AGA members, patients, foundations, and industry leaders. All gifts to the AGA Research Foundation go toward an endowment that will support young investigators on an ongoing basis, making it a gift that keeps on giving.
There are several new awards being offered this year:
– AGA–Caroline Craig Augustyn and Damian Augustyn Award in Digestive Cancer: This grant provides a one-time $40,000 award to a young investigator who currently holds a federal or nonfederal career development award devoted to conducting research related to digestive cancer. The application deadline is Jan. 13.
– AGA–Elsevier Gut Microbiome Pilot Research Award: This research initiative grant for $25,000 for 1 year is offered to early-stage and established investigators to support pilot research projects pertaining to the gut microbiome. The application deadline is Jan. 13.
– AGA Investing in the Future Student Research Fellowship: Awards are offered to support underrepresented minority students performing digestive disease or nutrition research for a minimum of 10 weeks. The award offers $5,000 and is supported by the National Institute of Diabetes and Digestive and Kidney Diseases (Award R25DK096968). The application deadline is Feb. 15.
In addition, we are continuing to offer the following awards with upcoming application deadlines:
– June & Donald O. Castell, MD Esophageal Clinical Research Award: This award provides 1 year of funding to a junior faculty member in that research area. This year marks the 13th annual Castell award supporting clinical esophageal researchers. Support of this program is made possible by the June and Donald O. Castell, MD, Gastroenterology Research and Education Trust. The application deadline is Jan. 20.
– Eli and Edythe L. Broad Student Research Fellowship Awards: This fellowship offers $2,500 stipends to high school and undergraduate students and $3,000 to medical and graduate students to spend time performing research in digestive diseases or nutrition for a minimum of 10 weeks. This award stimulates interest in research careers in digestive diseases by providing monetary support for research projects. The application deadline is March 15.
Over the last 28 years, the AGA and its foundations have supported 711 scientists with $40,569,000 in grants. This support is an investment in scientific discovery that ensures the ongoing pipeline of young investigators who will contribute to future advances.
For a complete list of AGA Research Foundation awards or to support the Research Awards Program with a tax-deductible gift, visit www.gastro.org/foundation.
The AGA Research Foundation provides career development research grants to young investigators. The foundation offers a variety of grants, some of which are targeted to specific digestive diseases.
These awards are made possible through the generosity of AGA members, patients, foundations, and industry leaders. All gifts to the AGA Research Foundation go toward an endowment that will support young investigators on an ongoing basis, making it a gift that keeps on giving.
There are several new awards being offered this year:
– AGA–Caroline Craig Augustyn and Damian Augustyn Award in Digestive Cancer: This grant provides a one-time $40,000 award to a young investigator who currently holds a federal or nonfederal career development award devoted to conducting research related to digestive cancer. The application deadline is Jan. 13.
– AGA–Elsevier Gut Microbiome Pilot Research Award: This research initiative grant for $25,000 for 1 year is offered to early-stage and established investigators to support pilot research projects pertaining to the gut microbiome. The application deadline is Jan. 13.
– AGA Investing in the Future Student Research Fellowship: Awards are offered to support underrepresented minority students performing digestive disease or nutrition research for a minimum of 10 weeks. The award offers $5,000 and is supported by the National Institute of Diabetes and Digestive and Kidney Diseases (Award R25DK096968). The application deadline is Feb. 15.
In addition, we are continuing to offer the following awards with upcoming application deadlines:
– June & Donald O. Castell, MD Esophageal Clinical Research Award: This award provides 1 year of funding to a junior faculty member in that research area. This year marks the 13th annual Castell award supporting clinical esophageal researchers. Support of this program is made possible by the June and Donald O. Castell, MD, Gastroenterology Research and Education Trust. The application deadline is Jan. 20.
– Eli and Edythe L. Broad Student Research Fellowship Awards: This fellowship offers $2,500 stipends to high school and undergraduate students and $3,000 to medical and graduate students to spend time performing research in digestive diseases or nutrition for a minimum of 10 weeks. This award stimulates interest in research careers in digestive diseases by providing monetary support for research projects. The application deadline is March 15.
Over the last 28 years, the AGA and its foundations have supported 711 scientists with $40,569,000 in grants. This support is an investment in scientific discovery that ensures the ongoing pipeline of young investigators who will contribute to future advances.
For a complete list of AGA Research Foundation awards or to support the Research Awards Program with a tax-deductible gift, visit www.gastro.org/foundation.
Donate a Colonoscopy to the Research Foundation
I am asking you to support a cause important to me and equally important to you. Please ensure that scientific discoveries and medical breakthroughs in the field of digestive diseases continue by making a tax-deductible gift to the AGA Research Foundation today.
Scientific discovery helps you successfully practice medicine
As clinicians we benefit from advances in clinical care made possible by scientific discovery. Widespread use of fiber-optic endoscopes beginning in the 1960s later led to retrograde colonoscopy of the entire colon and endoscopic excision of polyps, helping to significantly reduce colon cancer mortality rates. Barry J. Marshall and J. Robin Warren’s discovery that peptic ulcer disease was primarily caused by Helicobacter pylori helped revolutionize the treatment of the disease.
These are just a couple of examples of the highest-profile advances in the field. There are many more. Most every diagnostic decision you make and every treatment protocol you follow has its origins in bench science and clinical trials.
Imagine your practice without these advances
Sometimes we take for granted the things we do every day. But, where would we be as clinicians without scientific discovery and the people behind these important medical breakthroughs? Would most internal conditions only be diagnosed or treated with open surgery? Would the treatment of patients with peptic ulcer disease focus on hospitalization, bed rest, and prescription of special bland food?
More importantly, where will the field be if we don’t continue to invest in scientific discovery and ensure the ongoing pipeline of young investigators who will contribute to future advances? The AGA Research Foundation funds promising young investigators who might not receive funding otherwise at crucial times in their early careers.
The research of these individuals, while important to the field, if left unfunded, could end prematurely. That’s something the field can’t afford, and that’s why I’ve supported the AGA Research Foundation over the years through my donations.
Show support, donate a colonoscopy
Donate a colonoscopy? Yes, symbolically. I earlier mentioned colonoscopy as one of my examples of how advances in the field help us deliver more effective patient care. Many of us perform colonoscopies on a regular basis. By donating your physician fee for just one screening colonoscopy (about $400-$800), you can make the difference in the lives of the young basic science and clinical investigators who are supported by the AGA Research Foundation through its research awards program. It’s a way for you to show your gratitude and give back to the field.
If you don’t perform colonoscopies, aren’t sure of the physician fee, or would like to give a different amount, that’s fine.
You can demonstrate your support by sending your tax-deductible gift of $400, $800, $1,000, or any amount you can give. You can make a secure online donation at www.gastro.org/contribute.
Your gift will go toward an endowment that will support young investigators on an ongoing basis, making it a gift that keeps on giving. Remember to give by Dec. 31, 2012, to receive a tax credit for this year.
Thank you in advance for your support. Best wishes for a happy, healthy holiday season and prosperous New Year.
Martin Brotman, M.D., AGAF, is Chair Elect of the AGA Research Foundation. He also is Senior Vice President of Education, Research, and Philanthropy at CPMC Sutter Health, San Francisco.
I am asking you to support a cause important to me and equally important to you. Please ensure that scientific discoveries and medical breakthroughs in the field of digestive diseases continue by making a tax-deductible gift to the AGA Research Foundation today.
Scientific discovery helps you successfully practice medicine
As clinicians we benefit from advances in clinical care made possible by scientific discovery. Widespread use of fiber-optic endoscopes beginning in the 1960s later led to retrograde colonoscopy of the entire colon and endoscopic excision of polyps, helping to significantly reduce colon cancer mortality rates. Barry J. Marshall and J. Robin Warren’s discovery that peptic ulcer disease was primarily caused by Helicobacter pylori helped revolutionize the treatment of the disease.
These are just a couple of examples of the highest-profile advances in the field. There are many more. Most every diagnostic decision you make and every treatment protocol you follow has its origins in bench science and clinical trials.
Imagine your practice without these advances
Sometimes we take for granted the things we do every day. But, where would we be as clinicians without scientific discovery and the people behind these important medical breakthroughs? Would most internal conditions only be diagnosed or treated with open surgery? Would the treatment of patients with peptic ulcer disease focus on hospitalization, bed rest, and prescription of special bland food?
More importantly, where will the field be if we don’t continue to invest in scientific discovery and ensure the ongoing pipeline of young investigators who will contribute to future advances? The AGA Research Foundation funds promising young investigators who might not receive funding otherwise at crucial times in their early careers.
The research of these individuals, while important to the field, if left unfunded, could end prematurely. That’s something the field can’t afford, and that’s why I’ve supported the AGA Research Foundation over the years through my donations.
Show support, donate a colonoscopy
Donate a colonoscopy? Yes, symbolically. I earlier mentioned colonoscopy as one of my examples of how advances in the field help us deliver more effective patient care. Many of us perform colonoscopies on a regular basis. By donating your physician fee for just one screening colonoscopy (about $400-$800), you can make the difference in the lives of the young basic science and clinical investigators who are supported by the AGA Research Foundation through its research awards program. It’s a way for you to show your gratitude and give back to the field.
If you don’t perform colonoscopies, aren’t sure of the physician fee, or would like to give a different amount, that’s fine.
You can demonstrate your support by sending your tax-deductible gift of $400, $800, $1,000, or any amount you can give. You can make a secure online donation at www.gastro.org/contribute.
Your gift will go toward an endowment that will support young investigators on an ongoing basis, making it a gift that keeps on giving. Remember to give by Dec. 31, 2012, to receive a tax credit for this year.
Thank you in advance for your support. Best wishes for a happy, healthy holiday season and prosperous New Year.
Martin Brotman, M.D., AGAF, is Chair Elect of the AGA Research Foundation. He also is Senior Vice President of Education, Research, and Philanthropy at CPMC Sutter Health, San Francisco.
I am asking you to support a cause important to me and equally important to you. Please ensure that scientific discoveries and medical breakthroughs in the field of digestive diseases continue by making a tax-deductible gift to the AGA Research Foundation today.
Scientific discovery helps you successfully practice medicine
As clinicians we benefit from advances in clinical care made possible by scientific discovery. Widespread use of fiber-optic endoscopes beginning in the 1960s later led to retrograde colonoscopy of the entire colon and endoscopic excision of polyps, helping to significantly reduce colon cancer mortality rates. Barry J. Marshall and J. Robin Warren’s discovery that peptic ulcer disease was primarily caused by Helicobacter pylori helped revolutionize the treatment of the disease.
These are just a couple of examples of the highest-profile advances in the field. There are many more. Most every diagnostic decision you make and every treatment protocol you follow has its origins in bench science and clinical trials.
Imagine your practice without these advances
Sometimes we take for granted the things we do every day. But, where would we be as clinicians without scientific discovery and the people behind these important medical breakthroughs? Would most internal conditions only be diagnosed or treated with open surgery? Would the treatment of patients with peptic ulcer disease focus on hospitalization, bed rest, and prescription of special bland food?
More importantly, where will the field be if we don’t continue to invest in scientific discovery and ensure the ongoing pipeline of young investigators who will contribute to future advances? The AGA Research Foundation funds promising young investigators who might not receive funding otherwise at crucial times in their early careers.
The research of these individuals, while important to the field, if left unfunded, could end prematurely. That’s something the field can’t afford, and that’s why I’ve supported the AGA Research Foundation over the years through my donations.
Show support, donate a colonoscopy
Donate a colonoscopy? Yes, symbolically. I earlier mentioned colonoscopy as one of my examples of how advances in the field help us deliver more effective patient care. Many of us perform colonoscopies on a regular basis. By donating your physician fee for just one screening colonoscopy (about $400-$800), you can make the difference in the lives of the young basic science and clinical investigators who are supported by the AGA Research Foundation through its research awards program. It’s a way for you to show your gratitude and give back to the field.
If you don’t perform colonoscopies, aren’t sure of the physician fee, or would like to give a different amount, that’s fine.
You can demonstrate your support by sending your tax-deductible gift of $400, $800, $1,000, or any amount you can give. You can make a secure online donation at www.gastro.org/contribute.
Your gift will go toward an endowment that will support young investigators on an ongoing basis, making it a gift that keeps on giving. Remember to give by Dec. 31, 2012, to receive a tax credit for this year.
Thank you in advance for your support. Best wishes for a happy, healthy holiday season and prosperous New Year.
Martin Brotman, M.D., AGAF, is Chair Elect of the AGA Research Foundation. He also is Senior Vice President of Education, Research, and Philanthropy at CPMC Sutter Health, San Francisco.
Could You Become a Preferred Provider?
Health payers are continually marching toward establishing quality reporting requirements to assess provider value and determine whether they are providing the best care for their patients. Preferred provider programs, such as UnitedHealthcare’s Premium program, will be used to distinguish high-value providers. Increasingly, these programs are migrating toward models that establish preferred status for providers, which can mean lower copays for patients, waiver of preauthorization for some services, and inclusion in narrow or tiered networks.
AGA has launched important quality measurement and reporting programs in recent years, with the aim of providing gastroenterologists with ways to report quality performance and earn various incentives from health payers. Through these programs, as well as the American Board of Internal Medicine Practice Improvement Modules, gastroenterologists have access to a broad menu of options to meet reporting requirements, allowing them to distinguish themselves among their peers and earn rewards.
The AGA Digestive Health Outcomes Registry provides a mechanism for reporting ongoing quality performance and improvement. AGA has partnered with UnitedHealthcare to use the registry as a component of the Premium program starting in 2013, and is pursuing other payer relationships that will be structured in the same fashion. Using the electronic medical record and endowriter integration offered as part of the registry, gastroenterologists can automatically feed data into the program. If they opt in to a payer reporting program, their aggregate performance will be automatically reported on their behalf to meet quality reporting requirements.
The AGA’s Bridges to Excellence (BTE) IBD Care Recognition, the first Digestive Health Recognition Program (DHRP) module, is a way to submit a small sample of patient data for assessment. Gastroenterologists who meet a minimum standard of quality performance using the Physician Consortium for Performance Improvement and CMS Physician Quality Reporting System (PQRS) IBD measures become recognized by BTE. This is a national program that counts among its participants nearly every national payer and a large group of regional payers and purchasers. BTE recognition leads to incentives from these payers and public recognition; the program can also be used to submit data to fulfill the requirements of the PQRS. The AGA is currently developing a quality improvement toolkit specifically aimed at helping gastroenterologists meet BTE standards.
It is crucial that gastroenterologists avail themselves of these programs so that they don’t get left behind and leave bonus money on the table. For more information about how you can meet these standards through AGA programs, visit www.agaregistry.org and www.agarecognition.org.
Health payers are continually marching toward establishing quality reporting requirements to assess provider value and determine whether they are providing the best care for their patients. Preferred provider programs, such as UnitedHealthcare’s Premium program, will be used to distinguish high-value providers. Increasingly, these programs are migrating toward models that establish preferred status for providers, which can mean lower copays for patients, waiver of preauthorization for some services, and inclusion in narrow or tiered networks.
AGA has launched important quality measurement and reporting programs in recent years, with the aim of providing gastroenterologists with ways to report quality performance and earn various incentives from health payers. Through these programs, as well as the American Board of Internal Medicine Practice Improvement Modules, gastroenterologists have access to a broad menu of options to meet reporting requirements, allowing them to distinguish themselves among their peers and earn rewards.
The AGA Digestive Health Outcomes Registry provides a mechanism for reporting ongoing quality performance and improvement. AGA has partnered with UnitedHealthcare to use the registry as a component of the Premium program starting in 2013, and is pursuing other payer relationships that will be structured in the same fashion. Using the electronic medical record and endowriter integration offered as part of the registry, gastroenterologists can automatically feed data into the program. If they opt in to a payer reporting program, their aggregate performance will be automatically reported on their behalf to meet quality reporting requirements.
The AGA’s Bridges to Excellence (BTE) IBD Care Recognition, the first Digestive Health Recognition Program (DHRP) module, is a way to submit a small sample of patient data for assessment. Gastroenterologists who meet a minimum standard of quality performance using the Physician Consortium for Performance Improvement and CMS Physician Quality Reporting System (PQRS) IBD measures become recognized by BTE. This is a national program that counts among its participants nearly every national payer and a large group of regional payers and purchasers. BTE recognition leads to incentives from these payers and public recognition; the program can also be used to submit data to fulfill the requirements of the PQRS. The AGA is currently developing a quality improvement toolkit specifically aimed at helping gastroenterologists meet BTE standards.
It is crucial that gastroenterologists avail themselves of these programs so that they don’t get left behind and leave bonus money on the table. For more information about how you can meet these standards through AGA programs, visit www.agaregistry.org and www.agarecognition.org.
Health payers are continually marching toward establishing quality reporting requirements to assess provider value and determine whether they are providing the best care for their patients. Preferred provider programs, such as UnitedHealthcare’s Premium program, will be used to distinguish high-value providers. Increasingly, these programs are migrating toward models that establish preferred status for providers, which can mean lower copays for patients, waiver of preauthorization for some services, and inclusion in narrow or tiered networks.
AGA has launched important quality measurement and reporting programs in recent years, with the aim of providing gastroenterologists with ways to report quality performance and earn various incentives from health payers. Through these programs, as well as the American Board of Internal Medicine Practice Improvement Modules, gastroenterologists have access to a broad menu of options to meet reporting requirements, allowing them to distinguish themselves among their peers and earn rewards.
The AGA Digestive Health Outcomes Registry provides a mechanism for reporting ongoing quality performance and improvement. AGA has partnered with UnitedHealthcare to use the registry as a component of the Premium program starting in 2013, and is pursuing other payer relationships that will be structured in the same fashion. Using the electronic medical record and endowriter integration offered as part of the registry, gastroenterologists can automatically feed data into the program. If they opt in to a payer reporting program, their aggregate performance will be automatically reported on their behalf to meet quality reporting requirements.
The AGA’s Bridges to Excellence (BTE) IBD Care Recognition, the first Digestive Health Recognition Program (DHRP) module, is a way to submit a small sample of patient data for assessment. Gastroenterologists who meet a minimum standard of quality performance using the Physician Consortium for Performance Improvement and CMS Physician Quality Reporting System (PQRS) IBD measures become recognized by BTE. This is a national program that counts among its participants nearly every national payer and a large group of regional payers and purchasers. BTE recognition leads to incentives from these payers and public recognition; the program can also be used to submit data to fulfill the requirements of the PQRS. The AGA is currently developing a quality improvement toolkit specifically aimed at helping gastroenterologists meet BTE standards.
It is crucial that gastroenterologists avail themselves of these programs so that they don’t get left behind and leave bonus money on the table. For more information about how you can meet these standards through AGA programs, visit www.agaregistry.org and www.agarecognition.org.
GI Research Needs Your Support
Now more than ever, the AGA Research Foundation needs your support. At a time when funds from NIH and other traditional sources of support are in decline, the foundation plays an important role in medical research by providing grants to young scientists at a critical time in their research careers.
Your contribution will help us fulfill our vision of fostering the future of young researchers in gastroenterology and hepatology. By joining others in donating to the AGA Research Foundation, you will ensure that researchers have opportunities to continue their life-saving work.
"The future of our field depends on advances made in research, bench, clinical and translational. I see firsthand that it’s increasingly difficult for investigators to secure funding, particularly those in the early stages of their career. We can’t afford to have our pipeline of researchers dry up. The AGA Research Foundation provides the funds that are necessary for these young investigators to continue their work and contribute to the field. That’s why I support the foundation."
– Mark Donowitz, M.D., AGAF
LeBoff Professor of Medicine, Johns Hopkins University, past AGA president and AGA Legacy Society member
Join your colleagues in supporting the foundation’s work. Demonstrate that you also see the value of research to the future of gastroenterology and as the basis of the work you do every day.
"I reached a point in my career where I realized how much of my ability to care for patients is a result of both clinical and basic research, and recognized that I have been fortunate enough to have the opportunity to give back and make a substantial gift to help continue research to benefit future generations."
– Patrick Quinn, M.D., AGAF
Practicing gastroenterologist at Northern New Mexico Gastroenterology, AGA Legacy Society member
As a medical oncologist I have witnessed firsthand the horrible burden of [gastric cancer] and our urgent need to make progress in our ability to prevent, detect and treat this disease. With this support from the AGA, my laboratory at the Dana-Farber Cancer Institute will be able to follow up on new results with immediate potential translational relevance to the care of patients with gastric cancer.
– Adam J. Bass, M.D.
Dana Farber Cancer Institute, 2012 R. Robert and Sally Funderburg Research Award in Gastric Cancer recipient
Your gift will make an immediate difference in the careers of young physician-scientists like these—and in the lives of countless patients who will benefit from their work.
"Thanks to an AGA Research Scholar Award I’m able to continue research that will help us better understand the environmental risk factors for the development of Crohn’s and ulcerative colitis. This award came at a critical next step in my development into a leader in clinical and translational IBD research."
– Ashwin Ananthakrishnan, M.D., M.P.H.
Massachusetts General Hospital, 2011 AGA Research Scholar Award recipient
Join your colleagues in making a gift to the future of GI. Your tax-deductible contribution supports the foundation’s portfolio of research awards, which ensures that studies are funded, discoveries are made and patients are treated.
Donate today at gastro.org/contribute.
Now more than ever, the AGA Research Foundation needs your support. At a time when funds from NIH and other traditional sources of support are in decline, the foundation plays an important role in medical research by providing grants to young scientists at a critical time in their research careers.
Your contribution will help us fulfill our vision of fostering the future of young researchers in gastroenterology and hepatology. By joining others in donating to the AGA Research Foundation, you will ensure that researchers have opportunities to continue their life-saving work.
"The future of our field depends on advances made in research, bench, clinical and translational. I see firsthand that it’s increasingly difficult for investigators to secure funding, particularly those in the early stages of their career. We can’t afford to have our pipeline of researchers dry up. The AGA Research Foundation provides the funds that are necessary for these young investigators to continue their work and contribute to the field. That’s why I support the foundation."
– Mark Donowitz, M.D., AGAF
LeBoff Professor of Medicine, Johns Hopkins University, past AGA president and AGA Legacy Society member
Join your colleagues in supporting the foundation’s work. Demonstrate that you also see the value of research to the future of gastroenterology and as the basis of the work you do every day.
"I reached a point in my career where I realized how much of my ability to care for patients is a result of both clinical and basic research, and recognized that I have been fortunate enough to have the opportunity to give back and make a substantial gift to help continue research to benefit future generations."
– Patrick Quinn, M.D., AGAF
Practicing gastroenterologist at Northern New Mexico Gastroenterology, AGA Legacy Society member
As a medical oncologist I have witnessed firsthand the horrible burden of [gastric cancer] and our urgent need to make progress in our ability to prevent, detect and treat this disease. With this support from the AGA, my laboratory at the Dana-Farber Cancer Institute will be able to follow up on new results with immediate potential translational relevance to the care of patients with gastric cancer.
– Adam J. Bass, M.D.
Dana Farber Cancer Institute, 2012 R. Robert and Sally Funderburg Research Award in Gastric Cancer recipient
Your gift will make an immediate difference in the careers of young physician-scientists like these—and in the lives of countless patients who will benefit from their work.
"Thanks to an AGA Research Scholar Award I’m able to continue research that will help us better understand the environmental risk factors for the development of Crohn’s and ulcerative colitis. This award came at a critical next step in my development into a leader in clinical and translational IBD research."
– Ashwin Ananthakrishnan, M.D., M.P.H.
Massachusetts General Hospital, 2011 AGA Research Scholar Award recipient
Join your colleagues in making a gift to the future of GI. Your tax-deductible contribution supports the foundation’s portfolio of research awards, which ensures that studies are funded, discoveries are made and patients are treated.
Donate today at gastro.org/contribute.
Now more than ever, the AGA Research Foundation needs your support. At a time when funds from NIH and other traditional sources of support are in decline, the foundation plays an important role in medical research by providing grants to young scientists at a critical time in their research careers.
Your contribution will help us fulfill our vision of fostering the future of young researchers in gastroenterology and hepatology. By joining others in donating to the AGA Research Foundation, you will ensure that researchers have opportunities to continue their life-saving work.
"The future of our field depends on advances made in research, bench, clinical and translational. I see firsthand that it’s increasingly difficult for investigators to secure funding, particularly those in the early stages of their career. We can’t afford to have our pipeline of researchers dry up. The AGA Research Foundation provides the funds that are necessary for these young investigators to continue their work and contribute to the field. That’s why I support the foundation."
– Mark Donowitz, M.D., AGAF
LeBoff Professor of Medicine, Johns Hopkins University, past AGA president and AGA Legacy Society member
Join your colleagues in supporting the foundation’s work. Demonstrate that you also see the value of research to the future of gastroenterology and as the basis of the work you do every day.
"I reached a point in my career where I realized how much of my ability to care for patients is a result of both clinical and basic research, and recognized that I have been fortunate enough to have the opportunity to give back and make a substantial gift to help continue research to benefit future generations."
– Patrick Quinn, M.D., AGAF
Practicing gastroenterologist at Northern New Mexico Gastroenterology, AGA Legacy Society member
As a medical oncologist I have witnessed firsthand the horrible burden of [gastric cancer] and our urgent need to make progress in our ability to prevent, detect and treat this disease. With this support from the AGA, my laboratory at the Dana-Farber Cancer Institute will be able to follow up on new results with immediate potential translational relevance to the care of patients with gastric cancer.
– Adam J. Bass, M.D.
Dana Farber Cancer Institute, 2012 R. Robert and Sally Funderburg Research Award in Gastric Cancer recipient
Your gift will make an immediate difference in the careers of young physician-scientists like these—and in the lives of countless patients who will benefit from their work.
"Thanks to an AGA Research Scholar Award I’m able to continue research that will help us better understand the environmental risk factors for the development of Crohn’s and ulcerative colitis. This award came at a critical next step in my development into a leader in clinical and translational IBD research."
– Ashwin Ananthakrishnan, M.D., M.P.H.
Massachusetts General Hospital, 2011 AGA Research Scholar Award recipient
Join your colleagues in making a gift to the future of GI. Your tax-deductible contribution supports the foundation’s portfolio of research awards, which ensures that studies are funded, discoveries are made and patients are treated.
Donate today at gastro.org/contribute.
CMS Targets Endoscopy Procedures for Evaluation
A systematic evaluation of physician services is currently underway that will have a significant impact on reimbursement for all who perform endoscopic procedures. The evaluation is the result of the Medicare physician fee schedule for calendar year 2012 in which the Centers for Medicare and Medicaid Services (CMS) asked the American Medical Association’s (AMA’s) Relative Value Update Committee (RUC) to review an extensive number of physician services.

The GI community is facing a comprehensive review of physician work and practice expense inputs on more than 100 endoscopy procedures. These inputs directly affect how codes and procedures are "valued" and ultimately reimbursed. The AGA and the American Society for Gastrointestinal Endoscopy (ASGE) are asking physicians to complete surveys of these endoscopic procedures. If a minimum number of responses for each survey are not obtained, the RUC could make recommendations to CMS about our codes without GI input.
The RUC is an advisory committee to CMS and is responsible for making relative value recommendations that directly affect the agency’s fee schedule decisions. If our recommendations for endoscopy procedures are not supported by adequate survey data from our membership, CMS will make the final determination regarding reimbursement of our procedures. Do not forfeit this important opportunity to provide input into the valuation of GI services.
Participation in RUC Surveys Is Critical
GI participation in the surveys is critical to obtaining equitable reimbursement of endoscopic procedures. Gastroenterologists must complete these surveys for physician work in order to provide realistic recommendations to the AMA’s RUC.
We realize that physicians are faced with ever-increasing demands for their time, but this is an important opportunity for all practicing gastroenterologists to have an impact on the future of our profession. Whether you perform routine esophagogastroduodenoscopy (EGD) and colonoscopy or more complex procedures such as endoscopic retrograde cholangiopancreatography (ERCP) and endoscopic ultrasound or stenting, your help is needed.
About RUC Surveys
The surveys are electronic and can be completed in a reasonable amount of time – no more than 1 or 2 hours. Only a physician can complete the surveys; they cannot be completed by office managers, nurses, fellows, nurse practitioners, or physician assistants.
When new codes are established or existing codes are revised, a survey of physicians providing that service is conducted by the relevant medical specialty society.
How Can You Help?
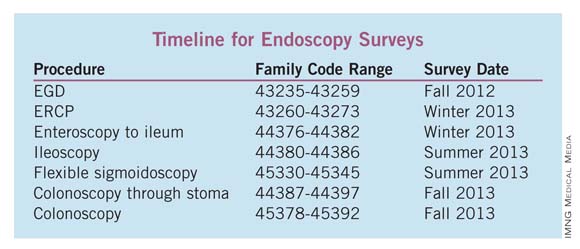
It is critical that you respond if you are selected to participate in any survey. We also urge you to add your name to the roster of individuals to be contacted to complete the RUC surveys of physician work for endoscopic procedures.
To volunteer, e-mail [email protected] with your name and contact information. Based on the list of surveys in the timeline, please specify which procedure surveys apply to you. Additional information will be provided prior to the start of each survey. To learn more visit www.gastro.org/RUC or e-mail Leslie D. Narramore, CPC, MPA, at [email protected].
A systematic evaluation of physician services is currently underway that will have a significant impact on reimbursement for all who perform endoscopic procedures. The evaluation is the result of the Medicare physician fee schedule for calendar year 2012 in which the Centers for Medicare and Medicaid Services (CMS) asked the American Medical Association’s (AMA’s) Relative Value Update Committee (RUC) to review an extensive number of physician services.

The GI community is facing a comprehensive review of physician work and practice expense inputs on more than 100 endoscopy procedures. These inputs directly affect how codes and procedures are "valued" and ultimately reimbursed. The AGA and the American Society for Gastrointestinal Endoscopy (ASGE) are asking physicians to complete surveys of these endoscopic procedures. If a minimum number of responses for each survey are not obtained, the RUC could make recommendations to CMS about our codes without GI input.
The RUC is an advisory committee to CMS and is responsible for making relative value recommendations that directly affect the agency’s fee schedule decisions. If our recommendations for endoscopy procedures are not supported by adequate survey data from our membership, CMS will make the final determination regarding reimbursement of our procedures. Do not forfeit this important opportunity to provide input into the valuation of GI services.
Participation in RUC Surveys Is Critical
GI participation in the surveys is critical to obtaining equitable reimbursement of endoscopic procedures. Gastroenterologists must complete these surveys for physician work in order to provide realistic recommendations to the AMA’s RUC.
We realize that physicians are faced with ever-increasing demands for their time, but this is an important opportunity for all practicing gastroenterologists to have an impact on the future of our profession. Whether you perform routine esophagogastroduodenoscopy (EGD) and colonoscopy or more complex procedures such as endoscopic retrograde cholangiopancreatography (ERCP) and endoscopic ultrasound or stenting, your help is needed.
About RUC Surveys
The surveys are electronic and can be completed in a reasonable amount of time – no more than 1 or 2 hours. Only a physician can complete the surveys; they cannot be completed by office managers, nurses, fellows, nurse practitioners, or physician assistants.
When new codes are established or existing codes are revised, a survey of physicians providing that service is conducted by the relevant medical specialty society.
How Can You Help?
It is critical that you respond if you are selected to participate in any survey. We also urge you to add your name to the roster of individuals to be contacted to complete the RUC surveys of physician work for endoscopic procedures.
To volunteer, e-mail [email protected] with your name and contact information. Based on the list of surveys in the timeline, please specify which procedure surveys apply to you. Additional information will be provided prior to the start of each survey. To learn more visit www.gastro.org/RUC or e-mail Leslie D. Narramore, CPC, MPA, at [email protected].
A systematic evaluation of physician services is currently underway that will have a significant impact on reimbursement for all who perform endoscopic procedures. The evaluation is the result of the Medicare physician fee schedule for calendar year 2012 in which the Centers for Medicare and Medicaid Services (CMS) asked the American Medical Association’s (AMA’s) Relative Value Update Committee (RUC) to review an extensive number of physician services.

The GI community is facing a comprehensive review of physician work and practice expense inputs on more than 100 endoscopy procedures. These inputs directly affect how codes and procedures are "valued" and ultimately reimbursed. The AGA and the American Society for Gastrointestinal Endoscopy (ASGE) are asking physicians to complete surveys of these endoscopic procedures. If a minimum number of responses for each survey are not obtained, the RUC could make recommendations to CMS about our codes without GI input.
The RUC is an advisory committee to CMS and is responsible for making relative value recommendations that directly affect the agency’s fee schedule decisions. If our recommendations for endoscopy procedures are not supported by adequate survey data from our membership, CMS will make the final determination regarding reimbursement of our procedures. Do not forfeit this important opportunity to provide input into the valuation of GI services.
Participation in RUC Surveys Is Critical
GI participation in the surveys is critical to obtaining equitable reimbursement of endoscopic procedures. Gastroenterologists must complete these surveys for physician work in order to provide realistic recommendations to the AMA’s RUC.
We realize that physicians are faced with ever-increasing demands for their time, but this is an important opportunity for all practicing gastroenterologists to have an impact on the future of our profession. Whether you perform routine esophagogastroduodenoscopy (EGD) and colonoscopy or more complex procedures such as endoscopic retrograde cholangiopancreatography (ERCP) and endoscopic ultrasound or stenting, your help is needed.
About RUC Surveys
The surveys are electronic and can be completed in a reasonable amount of time – no more than 1 or 2 hours. Only a physician can complete the surveys; they cannot be completed by office managers, nurses, fellows, nurse practitioners, or physician assistants.
When new codes are established or existing codes are revised, a survey of physicians providing that service is conducted by the relevant medical specialty society.
How Can You Help?
It is critical that you respond if you are selected to participate in any survey. We also urge you to add your name to the roster of individuals to be contacted to complete the RUC surveys of physician work for endoscopic procedures.
To volunteer, e-mail [email protected] with your name and contact information. Based on the list of surveys in the timeline, please specify which procedure surveys apply to you. Additional information will be provided prior to the start of each survey. To learn more visit www.gastro.org/RUC or e-mail Leslie D. Narramore, CPC, MPA, at [email protected].
IBD and IBS: Key Take Away Messages
At the AGA Spring Postgraduate Courses, Dr. Maria Abreu from the University of Miami began the session with updates on clinical aspects of inflammatory bowel disease. It has become increasingly apparent that Crohn's disease, in particular, is most often a chronic, progressive, transmural disease with a high lifetime risk for complications (e.g. strictures and fistulae) that often lead to the need for surgical resections. Furthermore, once initiated on corticosteroid therapy, a large proportion of individuals become steroid-dependent and are at risk for the multitude of steroid-related complications.
With the advent of more effective therapies, such as combination treatment with anti-tumor necrosis factor (anti-TNF) biologic therapy and immunosuppressives, it is important to risk stratify patients from the point of diagnosis or referral for the factors associated with progressive or complicated disease. Those factors include extensive disease presenting at a young age, deep mucosal ulcerations, fistulizing disease, high serologic titers of antibodies targeting intestinal microflora (pANCA, ASCA, etc), cigarette smoking, and the early need for corticosteroids). These are individuals for whom a heightened awareness of health maintenance issues (now incorporated into AGA quality guidelines) should be implemented, such asupdated vaccinations, ppd and chest x-rays prior to anti-TNF therapy, smoking cessation, dual x-ray absorptiometry scans, and early attempts at steroid-sparing.
Dr. Corey Siegel from the Dartmouth-Hitchcock Inflammatory Bowel Disease Center then segued into the (safety) and risks of immunosuppressive and biologic therapies for IBD, both alone and in combination. Dr. Siegel emphasized the underlying risks of progressive Crohn's disease and how "undertreatment" of the disease is substantial. He advised reviewing the much less common complications within the context of the chronic diseases and in terms that patients can comprehend; such as using absolute risks rather than risk ratios and utilization of tools such as a 10,000-patient diagram to demonstrate the rarity of many complications. Nevertheless, it is important to recognize that, aside from risks of intracellular pathogens, combination therapy is not associated with an increased risk of serious infections and that corticosteroids are associated with the biggest risk of infections. Viral infections such as Herpes zoster, non-melanotic skin cancer, Epstein-Barr virus-related and hepatosplenic T cell lymphomas are increased in patients receiving immunosuppressives. However, the absolute risk of those lymphomas is very small, approximately 6 in 10,000 in patients on combination therapy. Finally, Dr. Siegel described the serologic assay for the JC virus as being an important tool to determine and minimize the risk of developing progressive multifocal leukoencephalopathy for patients receiving, or about to receive natalizumab (Tysabri).
Dr. David Rubin from the University of Chicago then reviewed the prophylaxis against and the management of post-operative recurrence of Crohn's disease by discussing risk stratification based on factors associated with early disease recurrence in the setting of Crohn's disease and pouchitis in ulcerative colitis. Important factors related to the early recurrence of Crohn's disease include cigarette smoking, rapidly progressive or complicated disease, and extensive ileocolonic disease. Risks for pouchitis after ileo-anal anastomoses for ulcerative colitis include extraintestinal manifestations, smoking, and primary sclerosing cholangitis. Dr. Rubin emphasized the need to achieve mucosal healing and shared lessons he has learned related to the early endoscopic appearance of the pre-anastomotic ileum after an ileal resection. At present it appears that prophylaxis with combination immunosuppressives and antibiotics are effective for patients with a moderate risk of recurrence, whereas introduction or re-treatment with biologic therapies are looking to be the most effective prophylactic or therapeutic approach for patients with a high risk of clinical recurrence. While the probiotic VSL#3 has evidence for preventing pouchitis when administered at the time of re-anastomosis of the pouch to intestinal contiguity, optimal approaches at preventing pouchitis remain to be determined.
Dr. Darrell Pardi from the Mayo Clinic, Rochester, Minn., then provided an update on the overlapping spectrum of microscopic and collagenous colitis that seems to have peeked in incidence over the past decade, particularly in middle aged women. Risk factors include numerous medications but NSAIDs, in particular, as well as the presence of other autoimmune disorders, including celiac disease (particularly in patients with steatorrhea or weight loss). The most effective therapy has been budesonide, although some patients do become "budesonide-dependent." Dr. Pardi also favors bismuth therapy, but he says that the best evidence is in favor of budesonide for both short- and long-term use.
Finally, Dr. William Chey from the University of Michigan, Ann Arbor, ended the session by discussing some overlapping features between IBD and IBS, mucosal inflammation and related nerve-gut interactions, and the potential role of the intestinal microflora in initiating and perpetuating IBS symptoms. It is becoming apparent that the microbiota in IBS patients differs from that in healthy patients, and these differences may lead to novel diagnostic tests and treatments for IBS. He reviewed the spectrum of external factors affecting the gut microflora, including therapeutic foods (prebiotics), probiotics, and antibiotics. Recently, the non-absorbed antibiotic, rifaximin, has been demonstrated to have therapeutic potential in subgroups of IBS patients.
Stephen B. Hanauer, M.D., is the Joseph B. Kirsner Professor of Medicine and Clinical Pharmacology and chief of the section of gastroenterology, hepatology, and nutrition at the University of Chicago.
At the AGA Spring Postgraduate Courses, Dr. Maria Abreu from the University of Miami began the session with updates on clinical aspects of inflammatory bowel disease. It has become increasingly apparent that Crohn's disease, in particular, is most often a chronic, progressive, transmural disease with a high lifetime risk for complications (e.g. strictures and fistulae) that often lead to the need for surgical resections. Furthermore, once initiated on corticosteroid therapy, a large proportion of individuals become steroid-dependent and are at risk for the multitude of steroid-related complications.
With the advent of more effective therapies, such as combination treatment with anti-tumor necrosis factor (anti-TNF) biologic therapy and immunosuppressives, it is important to risk stratify patients from the point of diagnosis or referral for the factors associated with progressive or complicated disease. Those factors include extensive disease presenting at a young age, deep mucosal ulcerations, fistulizing disease, high serologic titers of antibodies targeting intestinal microflora (pANCA, ASCA, etc), cigarette smoking, and the early need for corticosteroids). These are individuals for whom a heightened awareness of health maintenance issues (now incorporated into AGA quality guidelines) should be implemented, such asupdated vaccinations, ppd and chest x-rays prior to anti-TNF therapy, smoking cessation, dual x-ray absorptiometry scans, and early attempts at steroid-sparing.
Dr. Corey Siegel from the Dartmouth-Hitchcock Inflammatory Bowel Disease Center then segued into the (safety) and risks of immunosuppressive and biologic therapies for IBD, both alone and in combination. Dr. Siegel emphasized the underlying risks of progressive Crohn's disease and how "undertreatment" of the disease is substantial. He advised reviewing the much less common complications within the context of the chronic diseases and in terms that patients can comprehend; such as using absolute risks rather than risk ratios and utilization of tools such as a 10,000-patient diagram to demonstrate the rarity of many complications. Nevertheless, it is important to recognize that, aside from risks of intracellular pathogens, combination therapy is not associated with an increased risk of serious infections and that corticosteroids are associated with the biggest risk of infections. Viral infections such as Herpes zoster, non-melanotic skin cancer, Epstein-Barr virus-related and hepatosplenic T cell lymphomas are increased in patients receiving immunosuppressives. However, the absolute risk of those lymphomas is very small, approximately 6 in 10,000 in patients on combination therapy. Finally, Dr. Siegel described the serologic assay for the JC virus as being an important tool to determine and minimize the risk of developing progressive multifocal leukoencephalopathy for patients receiving, or about to receive natalizumab (Tysabri).
Dr. David Rubin from the University of Chicago then reviewed the prophylaxis against and the management of post-operative recurrence of Crohn's disease by discussing risk stratification based on factors associated with early disease recurrence in the setting of Crohn's disease and pouchitis in ulcerative colitis. Important factors related to the early recurrence of Crohn's disease include cigarette smoking, rapidly progressive or complicated disease, and extensive ileocolonic disease. Risks for pouchitis after ileo-anal anastomoses for ulcerative colitis include extraintestinal manifestations, smoking, and primary sclerosing cholangitis. Dr. Rubin emphasized the need to achieve mucosal healing and shared lessons he has learned related to the early endoscopic appearance of the pre-anastomotic ileum after an ileal resection. At present it appears that prophylaxis with combination immunosuppressives and antibiotics are effective for patients with a moderate risk of recurrence, whereas introduction or re-treatment with biologic therapies are looking to be the most effective prophylactic or therapeutic approach for patients with a high risk of clinical recurrence. While the probiotic VSL#3 has evidence for preventing pouchitis when administered at the time of re-anastomosis of the pouch to intestinal contiguity, optimal approaches at preventing pouchitis remain to be determined.
Dr. Darrell Pardi from the Mayo Clinic, Rochester, Minn., then provided an update on the overlapping spectrum of microscopic and collagenous colitis that seems to have peeked in incidence over the past decade, particularly in middle aged women. Risk factors include numerous medications but NSAIDs, in particular, as well as the presence of other autoimmune disorders, including celiac disease (particularly in patients with steatorrhea or weight loss). The most effective therapy has been budesonide, although some patients do become "budesonide-dependent." Dr. Pardi also favors bismuth therapy, but he says that the best evidence is in favor of budesonide for both short- and long-term use.
Finally, Dr. William Chey from the University of Michigan, Ann Arbor, ended the session by discussing some overlapping features between IBD and IBS, mucosal inflammation and related nerve-gut interactions, and the potential role of the intestinal microflora in initiating and perpetuating IBS symptoms. It is becoming apparent that the microbiota in IBS patients differs from that in healthy patients, and these differences may lead to novel diagnostic tests and treatments for IBS. He reviewed the spectrum of external factors affecting the gut microflora, including therapeutic foods (prebiotics), probiotics, and antibiotics. Recently, the non-absorbed antibiotic, rifaximin, has been demonstrated to have therapeutic potential in subgroups of IBS patients.
Stephen B. Hanauer, M.D., is the Joseph B. Kirsner Professor of Medicine and Clinical Pharmacology and chief of the section of gastroenterology, hepatology, and nutrition at the University of Chicago.
At the AGA Spring Postgraduate Courses, Dr. Maria Abreu from the University of Miami began the session with updates on clinical aspects of inflammatory bowel disease. It has become increasingly apparent that Crohn's disease, in particular, is most often a chronic, progressive, transmural disease with a high lifetime risk for complications (e.g. strictures and fistulae) that often lead to the need for surgical resections. Furthermore, once initiated on corticosteroid therapy, a large proportion of individuals become steroid-dependent and are at risk for the multitude of steroid-related complications.
With the advent of more effective therapies, such as combination treatment with anti-tumor necrosis factor (anti-TNF) biologic therapy and immunosuppressives, it is important to risk stratify patients from the point of diagnosis or referral for the factors associated with progressive or complicated disease. Those factors include extensive disease presenting at a young age, deep mucosal ulcerations, fistulizing disease, high serologic titers of antibodies targeting intestinal microflora (pANCA, ASCA, etc), cigarette smoking, and the early need for corticosteroids). These are individuals for whom a heightened awareness of health maintenance issues (now incorporated into AGA quality guidelines) should be implemented, such asupdated vaccinations, ppd and chest x-rays prior to anti-TNF therapy, smoking cessation, dual x-ray absorptiometry scans, and early attempts at steroid-sparing.
Dr. Corey Siegel from the Dartmouth-Hitchcock Inflammatory Bowel Disease Center then segued into the (safety) and risks of immunosuppressive and biologic therapies for IBD, both alone and in combination. Dr. Siegel emphasized the underlying risks of progressive Crohn's disease and how "undertreatment" of the disease is substantial. He advised reviewing the much less common complications within the context of the chronic diseases and in terms that patients can comprehend; such as using absolute risks rather than risk ratios and utilization of tools such as a 10,000-patient diagram to demonstrate the rarity of many complications. Nevertheless, it is important to recognize that, aside from risks of intracellular pathogens, combination therapy is not associated with an increased risk of serious infections and that corticosteroids are associated with the biggest risk of infections. Viral infections such as Herpes zoster, non-melanotic skin cancer, Epstein-Barr virus-related and hepatosplenic T cell lymphomas are increased in patients receiving immunosuppressives. However, the absolute risk of those lymphomas is very small, approximately 6 in 10,000 in patients on combination therapy. Finally, Dr. Siegel described the serologic assay for the JC virus as being an important tool to determine and minimize the risk of developing progressive multifocal leukoencephalopathy for patients receiving, or about to receive natalizumab (Tysabri).
Dr. David Rubin from the University of Chicago then reviewed the prophylaxis against and the management of post-operative recurrence of Crohn's disease by discussing risk stratification based on factors associated with early disease recurrence in the setting of Crohn's disease and pouchitis in ulcerative colitis. Important factors related to the early recurrence of Crohn's disease include cigarette smoking, rapidly progressive or complicated disease, and extensive ileocolonic disease. Risks for pouchitis after ileo-anal anastomoses for ulcerative colitis include extraintestinal manifestations, smoking, and primary sclerosing cholangitis. Dr. Rubin emphasized the need to achieve mucosal healing and shared lessons he has learned related to the early endoscopic appearance of the pre-anastomotic ileum after an ileal resection. At present it appears that prophylaxis with combination immunosuppressives and antibiotics are effective for patients with a moderate risk of recurrence, whereas introduction or re-treatment with biologic therapies are looking to be the most effective prophylactic or therapeutic approach for patients with a high risk of clinical recurrence. While the probiotic VSL#3 has evidence for preventing pouchitis when administered at the time of re-anastomosis of the pouch to intestinal contiguity, optimal approaches at preventing pouchitis remain to be determined.
Dr. Darrell Pardi from the Mayo Clinic, Rochester, Minn., then provided an update on the overlapping spectrum of microscopic and collagenous colitis that seems to have peeked in incidence over the past decade, particularly in middle aged women. Risk factors include numerous medications but NSAIDs, in particular, as well as the presence of other autoimmune disorders, including celiac disease (particularly in patients with steatorrhea or weight loss). The most effective therapy has been budesonide, although some patients do become "budesonide-dependent." Dr. Pardi also favors bismuth therapy, but he says that the best evidence is in favor of budesonide for both short- and long-term use.
Finally, Dr. William Chey from the University of Michigan, Ann Arbor, ended the session by discussing some overlapping features between IBD and IBS, mucosal inflammation and related nerve-gut interactions, and the potential role of the intestinal microflora in initiating and perpetuating IBS symptoms. It is becoming apparent that the microbiota in IBS patients differs from that in healthy patients, and these differences may lead to novel diagnostic tests and treatments for IBS. He reviewed the spectrum of external factors affecting the gut microflora, including therapeutic foods (prebiotics), probiotics, and antibiotics. Recently, the non-absorbed antibiotic, rifaximin, has been demonstrated to have therapeutic potential in subgroups of IBS patients.
Stephen B. Hanauer, M.D., is the Joseph B. Kirsner Professor of Medicine and Clinical Pharmacology and chief of the section of gastroenterology, hepatology, and nutrition at the University of Chicago.
Light at the End of the Tunnel: Midgut Imaging and Beyond
AT the AGA Spring Postgraduate Courses the final session of the course focused on a variety of imaging and quality issues related to the small bowel and colon.
Dr. Brian Jacobson, associate professor of medicine at Boston University, emphasized the importance of grading colonoscopy preparations and the consequences of inadequate preparations. He stressed the proven superiority of split dosage preparation regimens to insure the optimal quality colon examination or the alternative of same-day preparations administered at 6:00 a.m. for afternoon cases. Lastly, he emphasized the misconception that monitored anesthesia care with propofol enhances adenoma detection rates.
Dr. Jonathan Leighton, chair of the division of gastroenterology at the Mayo Clinic Arizona, provided a state of the art perspective on the various imaging modalities of the small bowel in the setting of obscure bleeding, an area of considerable confusion. Capsule endoscopy is considered the initial step after a negative esophagogastroduodenoscopy and colonoscopy, although a second-look endoscopy is reasonable to consider first. If the capsule is found to be positive, Dr. Leighton advised using it to guide the deep enteroscopy approach. He said it is reasonable to consider an antegrade approach if at 0%-75% of small bowel and a retrograde approach if at 75-100% of the small bowel. In the setting of a negative capsule and a high index of suspicion, one should proceed right to deep enteroscopy. Lastly, newer cross sectional imaging tests such as CT angiography, CT enterography, or MR angiography are reasonable considerations in patients with ongoing bleeding despite negative endoscopic imaging.
Dr. Michael Wallace, chairman of the division of gastroenterology at the Mayo Clinic, Jacksonville, Fla., provided a perspective on advanced imaging techniques of the colon. First, he reminded attendees that chromoendoscopy improves neoplasia detection in inflammatory bowel disease and should be considered for cancer surveillance programs in those patients. Adenoma detection rates can be improved by using multimodal imaging platforms already available with an emphasis on high-definition, white light endoscopy with wide angle imaging accompanied by narrow band imaging. Finally, Dr. Wallace pointed out that confocal endomicroscopy, as part of a multimodality imaging platform, has the potential to reduce or even eliminate the need for biopsies, allow for real time diagnosis, and guide therapy.
The final presentation in the session came from Dr. Dawn Francis, associate professor of medicine at the Mayo Clinic, Jacksonville, Fla., who examined quality measures in colonoscopy. She emphasized the importance of appropriate screening intervals, commencing at 50 years for average risk individuals and then every 10 years until life expectancy is less than 5 years, although a starting age of 45 years has been recommended for African Americans. The appropriate surveillance interval depends on the type, number, and completeness of removal of any polyps. Finally, Dr. Francis emphasized the metric of a cecal intubation rate of at least 95% in healthy adults with an adenoma detection rate of 25% or greater in males and 15% or greater in females.
Gary W. Falk, M.D., M.S., is clinical co-director, Joint Center for Digestive, Liver and Pancreatic Medicine at the University of Pennsylvania and the Children’s Hospital of Philadelphia. He also is co-director of both the GI motility/physiology program and the GI physiology laboratory at the Hospital of the University of Pennsylvania, Philadelphia.
AT the AGA Spring Postgraduate Courses the final session of the course focused on a variety of imaging and quality issues related to the small bowel and colon.
Dr. Brian Jacobson, associate professor of medicine at Boston University, emphasized the importance of grading colonoscopy preparations and the consequences of inadequate preparations. He stressed the proven superiority of split dosage preparation regimens to insure the optimal quality colon examination or the alternative of same-day preparations administered at 6:00 a.m. for afternoon cases. Lastly, he emphasized the misconception that monitored anesthesia care with propofol enhances adenoma detection rates.
Dr. Jonathan Leighton, chair of the division of gastroenterology at the Mayo Clinic Arizona, provided a state of the art perspective on the various imaging modalities of the small bowel in the setting of obscure bleeding, an area of considerable confusion. Capsule endoscopy is considered the initial step after a negative esophagogastroduodenoscopy and colonoscopy, although a second-look endoscopy is reasonable to consider first. If the capsule is found to be positive, Dr. Leighton advised using it to guide the deep enteroscopy approach. He said it is reasonable to consider an antegrade approach if at 0%-75% of small bowel and a retrograde approach if at 75-100% of the small bowel. In the setting of a negative capsule and a high index of suspicion, one should proceed right to deep enteroscopy. Lastly, newer cross sectional imaging tests such as CT angiography, CT enterography, or MR angiography are reasonable considerations in patients with ongoing bleeding despite negative endoscopic imaging.
Dr. Michael Wallace, chairman of the division of gastroenterology at the Mayo Clinic, Jacksonville, Fla., provided a perspective on advanced imaging techniques of the colon. First, he reminded attendees that chromoendoscopy improves neoplasia detection in inflammatory bowel disease and should be considered for cancer surveillance programs in those patients. Adenoma detection rates can be improved by using multimodal imaging platforms already available with an emphasis on high-definition, white light endoscopy with wide angle imaging accompanied by narrow band imaging. Finally, Dr. Wallace pointed out that confocal endomicroscopy, as part of a multimodality imaging platform, has the potential to reduce or even eliminate the need for biopsies, allow for real time diagnosis, and guide therapy.
The final presentation in the session came from Dr. Dawn Francis, associate professor of medicine at the Mayo Clinic, Jacksonville, Fla., who examined quality measures in colonoscopy. She emphasized the importance of appropriate screening intervals, commencing at 50 years for average risk individuals and then every 10 years until life expectancy is less than 5 years, although a starting age of 45 years has been recommended for African Americans. The appropriate surveillance interval depends on the type, number, and completeness of removal of any polyps. Finally, Dr. Francis emphasized the metric of a cecal intubation rate of at least 95% in healthy adults with an adenoma detection rate of 25% or greater in males and 15% or greater in females.
Gary W. Falk, M.D., M.S., is clinical co-director, Joint Center for Digestive, Liver and Pancreatic Medicine at the University of Pennsylvania and the Children’s Hospital of Philadelphia. He also is co-director of both the GI motility/physiology program and the GI physiology laboratory at the Hospital of the University of Pennsylvania, Philadelphia.
AT the AGA Spring Postgraduate Courses the final session of the course focused on a variety of imaging and quality issues related to the small bowel and colon.
Dr. Brian Jacobson, associate professor of medicine at Boston University, emphasized the importance of grading colonoscopy preparations and the consequences of inadequate preparations. He stressed the proven superiority of split dosage preparation regimens to insure the optimal quality colon examination or the alternative of same-day preparations administered at 6:00 a.m. for afternoon cases. Lastly, he emphasized the misconception that monitored anesthesia care with propofol enhances adenoma detection rates.
Dr. Jonathan Leighton, chair of the division of gastroenterology at the Mayo Clinic Arizona, provided a state of the art perspective on the various imaging modalities of the small bowel in the setting of obscure bleeding, an area of considerable confusion. Capsule endoscopy is considered the initial step after a negative esophagogastroduodenoscopy and colonoscopy, although a second-look endoscopy is reasonable to consider first. If the capsule is found to be positive, Dr. Leighton advised using it to guide the deep enteroscopy approach. He said it is reasonable to consider an antegrade approach if at 0%-75% of small bowel and a retrograde approach if at 75-100% of the small bowel. In the setting of a negative capsule and a high index of suspicion, one should proceed right to deep enteroscopy. Lastly, newer cross sectional imaging tests such as CT angiography, CT enterography, or MR angiography are reasonable considerations in patients with ongoing bleeding despite negative endoscopic imaging.
Dr. Michael Wallace, chairman of the division of gastroenterology at the Mayo Clinic, Jacksonville, Fla., provided a perspective on advanced imaging techniques of the colon. First, he reminded attendees that chromoendoscopy improves neoplasia detection in inflammatory bowel disease and should be considered for cancer surveillance programs in those patients. Adenoma detection rates can be improved by using multimodal imaging platforms already available with an emphasis on high-definition, white light endoscopy with wide angle imaging accompanied by narrow band imaging. Finally, Dr. Wallace pointed out that confocal endomicroscopy, as part of a multimodality imaging platform, has the potential to reduce or even eliminate the need for biopsies, allow for real time diagnosis, and guide therapy.
The final presentation in the session came from Dr. Dawn Francis, associate professor of medicine at the Mayo Clinic, Jacksonville, Fla., who examined quality measures in colonoscopy. She emphasized the importance of appropriate screening intervals, commencing at 50 years for average risk individuals and then every 10 years until life expectancy is less than 5 years, although a starting age of 45 years has been recommended for African Americans. The appropriate surveillance interval depends on the type, number, and completeness of removal of any polyps. Finally, Dr. Francis emphasized the metric of a cecal intubation rate of at least 95% in healthy adults with an adenoma detection rate of 25% or greater in males and 15% or greater in females.
Gary W. Falk, M.D., M.S., is clinical co-director, Joint Center for Digestive, Liver and Pancreatic Medicine at the University of Pennsylvania and the Children’s Hospital of Philadelphia. He also is co-director of both the GI motility/physiology program and the GI physiology laboratory at the Hospital of the University of Pennsylvania, Philadelphia.
Intake of New Ideas in the Upper GI Tract
The session at the AGA Spring Postgraduate Courses entitled "Burning Issues in the Upper Gastrointestinal Tract" covered a number of hot topics in the management of patients with upper GI disorders.
Dr. Glenn Furuta, a pediatric gastroenterologist at Children’s Hospital Colorado, Aurora, presented a new definition of eosinophilic esophagitis (EoE) as a chronic, immune/antigen-mediated esophageal disease characterized clinically by symptoms related to esophageal dysfunction and histologically by eosinophil-predominant inflammation. He discussed the recently recognized condition called "PPI-responsive esophageal eosinophilia" in which patients have typical EoE symptoms and histology with no evidence of gastroesophageal reflux disease (GERD), and yet they respond to PPI therapy.
Dr. Furuta explained that these patients might have GERD with acid reflux causing esophageal eosinophilia, even though endoscopy and esophageal pH monitoring are normal. But he also proposed that they might have EoE responding to anti-inflammatory effects of PPIs that are independent of their effects on acid inhibition. And Dr. Furuta showed data that a six-food elimination diet that prohibits milk, soy, eggs, wheat, nuts, and seafood is effective therapy for adults with EoE.
Dr. Ronnie Fass, a professor of medicine at the University of Arizona in Tucson and director of the GI Motility Laboratory at the Southern Arizona VA Health Care System and head of the Neuro-Enteric Clinical Research Group, discussed the management of heartburn that is refractory to PPI therapy, and explained that potential underlying mechanisms include poor patient compliance, improper PPI dosing, persistent acidic reflux, non-acidic reflux, and the presence of non-GERD esophageal disorders like EoE. But he also noted that functional heartburn is the most common mechanism underlying PPI-refractory heartburn.
Although it is a common clinical practice to double the PPI dose for patients with this problem, Dr. Fass pointed out that there is little evidence to support this practice, and he encouraged clinicians instead to consider alternative strategies such as emphasizing proper compliance, implementing lifestyle modifications like weight loss, switching to another PPI, using baclofen (which reduces the transient LES relaxations that underlie most reflux episodes), and prescribing tricyclic antidepressants for pain modulation.
I presented recent data indicating that the risk of cancer is lower than we had thought it is for patients with non-dysplastic Barrett’s esophagus. Cancer affects approximately 1 in 400 such patients each year, and the risk appears to decrease over time. But for patients who have Barrett’s with high-grade dysplasia, the annual risk of cancer is approximately 6%, which warrants intervention. I discussed the AGA guideline recommending endoscopic eradication therapy rather than surveillance for the treatment of patients with confirmed high-grade dysplasia. This includes endoscopic mucosal resection of mucosal irregularities for staging, and ablation of all the remaining Barrett’s metaplasia. I showed the results of a recent systematic review that found that the risk of lymph node metastases for patients with high-grade dysplasia or intramucosal carcinoma in Barrett’s esophagus is in the range of 1%-2%. And finally, I pointed out that the AGA does not recommend endoscopic eradication therapy for the general population of patients with Barrett’s esophagus in the absence of dysplasia.
Dr. Nicholas Talley, professor of epidemiology and medicine in the department of gastroenterology and hepatology at the Mayo Clinic in Rochester, Minn., explained that the Rome III criteria for functional dyspepsia include one or more of the symptoms of epigastric pain, epigastric burning, early satiation and bothersome postprandial fullness. In addition, patients must have no evidence of structural disease that is likely to explain the symptoms.
Dr. Talley noted that patients with functional dyspepsia are often misdiagnosed as having GERD, and that one way to distinguish between those disorders is to look for meal-related symptoms – if symptoms like early satiation and postprandial fullness predominate, then functional dyspepsia is the likely diagnosis. The duodenum might play an important role in the pathogenesis of functional dyspepsia, he said, noting that the duodenum in these patients is hypersensitive to acid and distention, and that functional dyspepsia is associated with duodenal eosinophilia and with T-cells that home to the small bowel.
Finally, Dr. Nicholas Shaheen, professor of medicine and epidemiology and director of the Center for Esophageal Diseases & Swallowing at the University of North Carolina, Chapel Hill,discussed the management of non-variceal upper GI bleeding, encouraged clinicians to consider using a scoring system like the Rockall or Blatchford scores for patient risk stratification.
Dr. Shaheen said that patients with upper GI bleeding should have endoscopy within 24 hours of admission but, for patients who are hemodynamically stable, very early endoscopy within 12 hours does not reduce rebleeding or improve survival. This means that endoscopy safely can be postponed until the light of day for most patients, but they should be treated overnight with intravenous PPIs prior to the morning endoscopy because this treatment has been shown to decrease the need to administer endoscopic therapy. During the endoscopy, Dr. Shaheen recommended endoscopic therapy for ulcers that are actively spurting or oozing, and for non-bleeding visible vessels. He also recommended that we remove adherent clots to expose lesions that might need endoscopic therapy. He cautioned that injection therapy with epinephrine should not be used alone, but epinephrine injection can be combined with thermal therapy (heater probe and bipolar electrocoagulation), with the injection of sclerosants, and with the application of hemoclips, all of which are reasonable options that appear to have similar efficacy.
Stuart J. Spechler, M.D., is professor of medicine and the Berta M. and Cecil O. Patterson Chair in Gastroenterology at the University of Texas Southwestern Medical Center at Dallas, and chief of the division of gastroenterology at the Dallas VA Medical Center.
The session at the AGA Spring Postgraduate Courses entitled "Burning Issues in the Upper Gastrointestinal Tract" covered a number of hot topics in the management of patients with upper GI disorders.
Dr. Glenn Furuta, a pediatric gastroenterologist at Children’s Hospital Colorado, Aurora, presented a new definition of eosinophilic esophagitis (EoE) as a chronic, immune/antigen-mediated esophageal disease characterized clinically by symptoms related to esophageal dysfunction and histologically by eosinophil-predominant inflammation. He discussed the recently recognized condition called "PPI-responsive esophageal eosinophilia" in which patients have typical EoE symptoms and histology with no evidence of gastroesophageal reflux disease (GERD), and yet they respond to PPI therapy.
Dr. Furuta explained that these patients might have GERD with acid reflux causing esophageal eosinophilia, even though endoscopy and esophageal pH monitoring are normal. But he also proposed that they might have EoE responding to anti-inflammatory effects of PPIs that are independent of their effects on acid inhibition. And Dr. Furuta showed data that a six-food elimination diet that prohibits milk, soy, eggs, wheat, nuts, and seafood is effective therapy for adults with EoE.
Dr. Ronnie Fass, a professor of medicine at the University of Arizona in Tucson and director of the GI Motility Laboratory at the Southern Arizona VA Health Care System and head of the Neuro-Enteric Clinical Research Group, discussed the management of heartburn that is refractory to PPI therapy, and explained that potential underlying mechanisms include poor patient compliance, improper PPI dosing, persistent acidic reflux, non-acidic reflux, and the presence of non-GERD esophageal disorders like EoE. But he also noted that functional heartburn is the most common mechanism underlying PPI-refractory heartburn.
Although it is a common clinical practice to double the PPI dose for patients with this problem, Dr. Fass pointed out that there is little evidence to support this practice, and he encouraged clinicians instead to consider alternative strategies such as emphasizing proper compliance, implementing lifestyle modifications like weight loss, switching to another PPI, using baclofen (which reduces the transient LES relaxations that underlie most reflux episodes), and prescribing tricyclic antidepressants for pain modulation.
I presented recent data indicating that the risk of cancer is lower than we had thought it is for patients with non-dysplastic Barrett’s esophagus. Cancer affects approximately 1 in 400 such patients each year, and the risk appears to decrease over time. But for patients who have Barrett’s with high-grade dysplasia, the annual risk of cancer is approximately 6%, which warrants intervention. I discussed the AGA guideline recommending endoscopic eradication therapy rather than surveillance for the treatment of patients with confirmed high-grade dysplasia. This includes endoscopic mucosal resection of mucosal irregularities for staging, and ablation of all the remaining Barrett’s metaplasia. I showed the results of a recent systematic review that found that the risk of lymph node metastases for patients with high-grade dysplasia or intramucosal carcinoma in Barrett’s esophagus is in the range of 1%-2%. And finally, I pointed out that the AGA does not recommend endoscopic eradication therapy for the general population of patients with Barrett’s esophagus in the absence of dysplasia.
Dr. Nicholas Talley, professor of epidemiology and medicine in the department of gastroenterology and hepatology at the Mayo Clinic in Rochester, Minn., explained that the Rome III criteria for functional dyspepsia include one or more of the symptoms of epigastric pain, epigastric burning, early satiation and bothersome postprandial fullness. In addition, patients must have no evidence of structural disease that is likely to explain the symptoms.
Dr. Talley noted that patients with functional dyspepsia are often misdiagnosed as having GERD, and that one way to distinguish between those disorders is to look for meal-related symptoms – if symptoms like early satiation and postprandial fullness predominate, then functional dyspepsia is the likely diagnosis. The duodenum might play an important role in the pathogenesis of functional dyspepsia, he said, noting that the duodenum in these patients is hypersensitive to acid and distention, and that functional dyspepsia is associated with duodenal eosinophilia and with T-cells that home to the small bowel.
Finally, Dr. Nicholas Shaheen, professor of medicine and epidemiology and director of the Center for Esophageal Diseases & Swallowing at the University of North Carolina, Chapel Hill,discussed the management of non-variceal upper GI bleeding, encouraged clinicians to consider using a scoring system like the Rockall or Blatchford scores for patient risk stratification.
Dr. Shaheen said that patients with upper GI bleeding should have endoscopy within 24 hours of admission but, for patients who are hemodynamically stable, very early endoscopy within 12 hours does not reduce rebleeding or improve survival. This means that endoscopy safely can be postponed until the light of day for most patients, but they should be treated overnight with intravenous PPIs prior to the morning endoscopy because this treatment has been shown to decrease the need to administer endoscopic therapy. During the endoscopy, Dr. Shaheen recommended endoscopic therapy for ulcers that are actively spurting or oozing, and for non-bleeding visible vessels. He also recommended that we remove adherent clots to expose lesions that might need endoscopic therapy. He cautioned that injection therapy with epinephrine should not be used alone, but epinephrine injection can be combined with thermal therapy (heater probe and bipolar electrocoagulation), with the injection of sclerosants, and with the application of hemoclips, all of which are reasonable options that appear to have similar efficacy.
Stuart J. Spechler, M.D., is professor of medicine and the Berta M. and Cecil O. Patterson Chair in Gastroenterology at the University of Texas Southwestern Medical Center at Dallas, and chief of the division of gastroenterology at the Dallas VA Medical Center.
The session at the AGA Spring Postgraduate Courses entitled "Burning Issues in the Upper Gastrointestinal Tract" covered a number of hot topics in the management of patients with upper GI disorders.
Dr. Glenn Furuta, a pediatric gastroenterologist at Children’s Hospital Colorado, Aurora, presented a new definition of eosinophilic esophagitis (EoE) as a chronic, immune/antigen-mediated esophageal disease characterized clinically by symptoms related to esophageal dysfunction and histologically by eosinophil-predominant inflammation. He discussed the recently recognized condition called "PPI-responsive esophageal eosinophilia" in which patients have typical EoE symptoms and histology with no evidence of gastroesophageal reflux disease (GERD), and yet they respond to PPI therapy.
Dr. Furuta explained that these patients might have GERD with acid reflux causing esophageal eosinophilia, even though endoscopy and esophageal pH monitoring are normal. But he also proposed that they might have EoE responding to anti-inflammatory effects of PPIs that are independent of their effects on acid inhibition. And Dr. Furuta showed data that a six-food elimination diet that prohibits milk, soy, eggs, wheat, nuts, and seafood is effective therapy for adults with EoE.
Dr. Ronnie Fass, a professor of medicine at the University of Arizona in Tucson and director of the GI Motility Laboratory at the Southern Arizona VA Health Care System and head of the Neuro-Enteric Clinical Research Group, discussed the management of heartburn that is refractory to PPI therapy, and explained that potential underlying mechanisms include poor patient compliance, improper PPI dosing, persistent acidic reflux, non-acidic reflux, and the presence of non-GERD esophageal disorders like EoE. But he also noted that functional heartburn is the most common mechanism underlying PPI-refractory heartburn.
Although it is a common clinical practice to double the PPI dose for patients with this problem, Dr. Fass pointed out that there is little evidence to support this practice, and he encouraged clinicians instead to consider alternative strategies such as emphasizing proper compliance, implementing lifestyle modifications like weight loss, switching to another PPI, using baclofen (which reduces the transient LES relaxations that underlie most reflux episodes), and prescribing tricyclic antidepressants for pain modulation.
I presented recent data indicating that the risk of cancer is lower than we had thought it is for patients with non-dysplastic Barrett’s esophagus. Cancer affects approximately 1 in 400 such patients each year, and the risk appears to decrease over time. But for patients who have Barrett’s with high-grade dysplasia, the annual risk of cancer is approximately 6%, which warrants intervention. I discussed the AGA guideline recommending endoscopic eradication therapy rather than surveillance for the treatment of patients with confirmed high-grade dysplasia. This includes endoscopic mucosal resection of mucosal irregularities for staging, and ablation of all the remaining Barrett’s metaplasia. I showed the results of a recent systematic review that found that the risk of lymph node metastases for patients with high-grade dysplasia or intramucosal carcinoma in Barrett’s esophagus is in the range of 1%-2%. And finally, I pointed out that the AGA does not recommend endoscopic eradication therapy for the general population of patients with Barrett’s esophagus in the absence of dysplasia.
Dr. Nicholas Talley, professor of epidemiology and medicine in the department of gastroenterology and hepatology at the Mayo Clinic in Rochester, Minn., explained that the Rome III criteria for functional dyspepsia include one or more of the symptoms of epigastric pain, epigastric burning, early satiation and bothersome postprandial fullness. In addition, patients must have no evidence of structural disease that is likely to explain the symptoms.
Dr. Talley noted that patients with functional dyspepsia are often misdiagnosed as having GERD, and that one way to distinguish between those disorders is to look for meal-related symptoms – if symptoms like early satiation and postprandial fullness predominate, then functional dyspepsia is the likely diagnosis. The duodenum might play an important role in the pathogenesis of functional dyspepsia, he said, noting that the duodenum in these patients is hypersensitive to acid and distention, and that functional dyspepsia is associated with duodenal eosinophilia and with T-cells that home to the small bowel.
Finally, Dr. Nicholas Shaheen, professor of medicine and epidemiology and director of the Center for Esophageal Diseases & Swallowing at the University of North Carolina, Chapel Hill,discussed the management of non-variceal upper GI bleeding, encouraged clinicians to consider using a scoring system like the Rockall or Blatchford scores for patient risk stratification.
Dr. Shaheen said that patients with upper GI bleeding should have endoscopy within 24 hours of admission but, for patients who are hemodynamically stable, very early endoscopy within 12 hours does not reduce rebleeding or improve survival. This means that endoscopy safely can be postponed until the light of day for most patients, but they should be treated overnight with intravenous PPIs prior to the morning endoscopy because this treatment has been shown to decrease the need to administer endoscopic therapy. During the endoscopy, Dr. Shaheen recommended endoscopic therapy for ulcers that are actively spurting or oozing, and for non-bleeding visible vessels. He also recommended that we remove adherent clots to expose lesions that might need endoscopic therapy. He cautioned that injection therapy with epinephrine should not be used alone, but epinephrine injection can be combined with thermal therapy (heater probe and bipolar electrocoagulation), with the injection of sclerosants, and with the application of hemoclips, all of which are reasonable options that appear to have similar efficacy.
Stuart J. Spechler, M.D., is professor of medicine and the Berta M. and Cecil O. Patterson Chair in Gastroenterology at the University of Texas Southwestern Medical Center at Dallas, and chief of the division of gastroenterology at the Dallas VA Medical Center.
Screening, Surveillance, and Testing for GI Cancers
The Screening, Surveillance, and Molecular Testing for Gastrointestinal Cancers session of the 2012 AGA Spring Postgraduate Course contained five outstanding speakers who discussed cancers of the gastrointestinal tract or their precursor lesions, which gastroenterologists face frequently in the clinic.
Dr. John Inadomi from the University of Washington, Seattle, highlighted some evidence-based and practical approaches to care for patients who have developed or are at risk for Barrett’s esophagus.
Dr. Inadomi pointed out that our approach to screening should only involve those patients with multiple risk factors, such as white males with chronic heartburn and acid reflux, tobacco usage, and increased central obesity. Once Barrett’s is detected, there is low effectiveness of surveillance for Barrett’s to reduce esophageal cancer mortality, and is not cost-effective. This is largely due to the low observed conversion of Barrett’s esophagus to esophageal adenocarcinoma (less than 0.5% of patients/year), and that surveillance relies on repeated measures of a poor marker (i.e. random biopsies). Endoscopic ablation therapy is largely reserved for those patients with high-grade dysplasia (HGD), some patients with low-grade dysplasia (LGD), and is not for those with non-dysplastic Barrett’s esophagus. Ablation of patients with HGD and as well as some patients with LGD is cost-effective and may decrease cancer incidence. Dr. Inadomi also presented some new approaches for the management of Barrett’s and its potential complications, included advanced imaging techniques, the use of molecular diagnostics, and chemoprevention.
Dr. Asif Khalid from the University of Pittsburgh presented an approach to evaluation and care of the patient with cystic lesions of the pancreas. Dr. Khalid pointed out that serous cystic tumors are largely benign, are equal among males and females, and often have a central scar or honeycomb pattern on imaging. Mucinous neoplasms are a different story, and need to be carefully evaluated for potential resection. The mucinous cystic neoplasm (MCN) is premalignant, occurs more commonly in females, may present with septations on imaging, produces carcinoembryonic antigen (CEA), and needs resection. Intraductal papillary mucinous neoplasms (IPMNs) can be main duct, which requires resection as the only curative option, or branch duct, and requires surgical resection if at least 3 cm. IPMNs are equal between males and females, produce CEA, and may demonstrate mutation in the GNAS gene. Diagnostic differentiation between lesions can be challenging, but with imaging (endoscopic ultrasound, MRI, and CT) and aspiration of the lesion for cytology, amylase, CEA, and other molecular markers, differentiation is possible. Dr. Khalid stated that key features that differentiate mucinous from non-mucinous lesions are the presence of mutant KRAS and elevated CEA levels in the aspirate, and the presence of allelic loss in DNA taken in the sample. He also pointed out that key features to help differentiate malignant from non-malignant lesions include high DNA content, mutant KRAS, and allelic loss.
Dr. Hashem El-Serag from Baylor College of Medicine, Houston, showcased information on hepatocellular carcinoma (HCC). He showed evidence that the incidence of HCC is increasing, and that survival from HCC is marginally increasing as well with improved approaches to care. Dr. El-Serag reviewed the Milan criteria, where a person is potentially curative by resection or liver transplant if there are three or fewer nodules that are 3 cm or less in diameter. Dr. El-Serag presented data showing that surveillance for HCC reduces mortality from HCC. Efforts should be focused on Asian hepatitis B carriers, those with a family history of HCC, blacks from Africa, and patients with cirrhosis who have hepatitis B or C. Surveillance should be done every 6 months, and the combined use of ultrasound (with a sensitivity of 72%) with alpha-fetoprotein (AFP) is the best studied. Other biomarkers in the literature for HCC screening include des-gamma-carboxy prothrombin (DCP), golgi protein-73, and osteopontin. Dr. El-Serag also reviewed key aspects of imaging criteria for identifying HCC in cirrhotic patients, which includes hyper-enhancement by two imaging techniques if a lesion is at least 1 cm but less than 2 cm in diameter, and by one imaging technique if 2 cm or greater, with or without liver biopsy if the appearance is atypical.
Dr. Douglas Rex from Indiana University, Indianapolis, highlighted several quality issues in regard to screening for colorectal cancer. He stated that the primary contribution of gastroenterologists to colorectal cancer prevention is high quality colonoscopy. Dr. Rex pointed out recent evidence that colonoscopy is less effective in the right colon, compared with the left colon, and one contribution to this is the right-sided location of serrated, flat, and depressed lesions that blend in with the surrounding mucosa. For all colorectal screening tests, there is limited evidence regarding sensitivity for serrated lesions. Dr. Rex provided evidence that guaiac-based fecal occult blood test (FOBT) screening should all be now converted to fecal immunohistochemical test (FIT) screening due to enhanced sensitivity of the FIT tests. Dr. Rex also provided initial data on the first serum-screening assay for colorectal cancer, which was unable to detect precancerous lesions.
Dr. Sapna Syngal from the Dana-Farber Cancer Center in Boston presented on hereditary colorectal cancer syndromes. These syndromes elevate the risk for colorectal cancer, and if they are not recognized, their associated cancers present with a young age of onset. Patients and potential patients need to be referred for genetic counseling and testing, particularly those who present at a young age, have a high number of polyps, show upper GI lesions, and have strong family history of cancer or polyps. Dr. Syngal highlighted the issue of "genetic malpractice" that is growing in the United States due to a lack of recognition and referral for evaluation. These syndromes are managed much differently than sporadic cancer in their approach to surgery, surveillance for associated cancers, the screening of family members, and reproductive counseling and prenatal testing.
Dr. Syngal reviewed familial adenomatous polyposis (FAP), an autosomal dominant condition, and highlighted MYH associated polyposis (MAP), an autosomal recessive condition. Both FAP and MAP can present with colon polyps and cancer, upper GI lesions, form desmoid tumors, and are associated with thyroid, brain, and osteoma tumors. Care is through colectomy, upper GI surveillance, and chemoprevention for any lower GI remnant. Dr. Syngal also highlighted Lynch syndrome, which is associated with tumors of the colon and upper GI track, female reproductive track, urinary track, and sebaceous skin tumors. Lynch syndrome patients can undergo colonoscopic surveillance and subsequent colectomy, as well as endometrial aspiration and subsequent total abdominal hysterectomy-bilateral salpingo-oophorectomy. Dr. Syngal reviewed testing of tumors for microsatellite instability and immunohistochemistry for the DNA mismatch repair genes (mutation of which cause Lynch syndrome) and discussed prediction models for germline testing.
John M. Carethers, M.D., is the John G. Searle Professor and chair of the department of internal medicine at the University of Michigan, Ann Arbor.
The Screening, Surveillance, and Molecular Testing for Gastrointestinal Cancers session of the 2012 AGA Spring Postgraduate Course contained five outstanding speakers who discussed cancers of the gastrointestinal tract or their precursor lesions, which gastroenterologists face frequently in the clinic.
Dr. John Inadomi from the University of Washington, Seattle, highlighted some evidence-based and practical approaches to care for patients who have developed or are at risk for Barrett’s esophagus.
Dr. Inadomi pointed out that our approach to screening should only involve those patients with multiple risk factors, such as white males with chronic heartburn and acid reflux, tobacco usage, and increased central obesity. Once Barrett’s is detected, there is low effectiveness of surveillance for Barrett’s to reduce esophageal cancer mortality, and is not cost-effective. This is largely due to the low observed conversion of Barrett’s esophagus to esophageal adenocarcinoma (less than 0.5% of patients/year), and that surveillance relies on repeated measures of a poor marker (i.e. random biopsies). Endoscopic ablation therapy is largely reserved for those patients with high-grade dysplasia (HGD), some patients with low-grade dysplasia (LGD), and is not for those with non-dysplastic Barrett’s esophagus. Ablation of patients with HGD and as well as some patients with LGD is cost-effective and may decrease cancer incidence. Dr. Inadomi also presented some new approaches for the management of Barrett’s and its potential complications, included advanced imaging techniques, the use of molecular diagnostics, and chemoprevention.
Dr. Asif Khalid from the University of Pittsburgh presented an approach to evaluation and care of the patient with cystic lesions of the pancreas. Dr. Khalid pointed out that serous cystic tumors are largely benign, are equal among males and females, and often have a central scar or honeycomb pattern on imaging. Mucinous neoplasms are a different story, and need to be carefully evaluated for potential resection. The mucinous cystic neoplasm (MCN) is premalignant, occurs more commonly in females, may present with septations on imaging, produces carcinoembryonic antigen (CEA), and needs resection. Intraductal papillary mucinous neoplasms (IPMNs) can be main duct, which requires resection as the only curative option, or branch duct, and requires surgical resection if at least 3 cm. IPMNs are equal between males and females, produce CEA, and may demonstrate mutation in the GNAS gene. Diagnostic differentiation between lesions can be challenging, but with imaging (endoscopic ultrasound, MRI, and CT) and aspiration of the lesion for cytology, amylase, CEA, and other molecular markers, differentiation is possible. Dr. Khalid stated that key features that differentiate mucinous from non-mucinous lesions are the presence of mutant KRAS and elevated CEA levels in the aspirate, and the presence of allelic loss in DNA taken in the sample. He also pointed out that key features to help differentiate malignant from non-malignant lesions include high DNA content, mutant KRAS, and allelic loss.
Dr. Hashem El-Serag from Baylor College of Medicine, Houston, showcased information on hepatocellular carcinoma (HCC). He showed evidence that the incidence of HCC is increasing, and that survival from HCC is marginally increasing as well with improved approaches to care. Dr. El-Serag reviewed the Milan criteria, where a person is potentially curative by resection or liver transplant if there are three or fewer nodules that are 3 cm or less in diameter. Dr. El-Serag presented data showing that surveillance for HCC reduces mortality from HCC. Efforts should be focused on Asian hepatitis B carriers, those with a family history of HCC, blacks from Africa, and patients with cirrhosis who have hepatitis B or C. Surveillance should be done every 6 months, and the combined use of ultrasound (with a sensitivity of 72%) with alpha-fetoprotein (AFP) is the best studied. Other biomarkers in the literature for HCC screening include des-gamma-carboxy prothrombin (DCP), golgi protein-73, and osteopontin. Dr. El-Serag also reviewed key aspects of imaging criteria for identifying HCC in cirrhotic patients, which includes hyper-enhancement by two imaging techniques if a lesion is at least 1 cm but less than 2 cm in diameter, and by one imaging technique if 2 cm or greater, with or without liver biopsy if the appearance is atypical.
Dr. Douglas Rex from Indiana University, Indianapolis, highlighted several quality issues in regard to screening for colorectal cancer. He stated that the primary contribution of gastroenterologists to colorectal cancer prevention is high quality colonoscopy. Dr. Rex pointed out recent evidence that colonoscopy is less effective in the right colon, compared with the left colon, and one contribution to this is the right-sided location of serrated, flat, and depressed lesions that blend in with the surrounding mucosa. For all colorectal screening tests, there is limited evidence regarding sensitivity for serrated lesions. Dr. Rex provided evidence that guaiac-based fecal occult blood test (FOBT) screening should all be now converted to fecal immunohistochemical test (FIT) screening due to enhanced sensitivity of the FIT tests. Dr. Rex also provided initial data on the first serum-screening assay for colorectal cancer, which was unable to detect precancerous lesions.
Dr. Sapna Syngal from the Dana-Farber Cancer Center in Boston presented on hereditary colorectal cancer syndromes. These syndromes elevate the risk for colorectal cancer, and if they are not recognized, their associated cancers present with a young age of onset. Patients and potential patients need to be referred for genetic counseling and testing, particularly those who present at a young age, have a high number of polyps, show upper GI lesions, and have strong family history of cancer or polyps. Dr. Syngal highlighted the issue of "genetic malpractice" that is growing in the United States due to a lack of recognition and referral for evaluation. These syndromes are managed much differently than sporadic cancer in their approach to surgery, surveillance for associated cancers, the screening of family members, and reproductive counseling and prenatal testing.
Dr. Syngal reviewed familial adenomatous polyposis (FAP), an autosomal dominant condition, and highlighted MYH associated polyposis (MAP), an autosomal recessive condition. Both FAP and MAP can present with colon polyps and cancer, upper GI lesions, form desmoid tumors, and are associated with thyroid, brain, and osteoma tumors. Care is through colectomy, upper GI surveillance, and chemoprevention for any lower GI remnant. Dr. Syngal also highlighted Lynch syndrome, which is associated with tumors of the colon and upper GI track, female reproductive track, urinary track, and sebaceous skin tumors. Lynch syndrome patients can undergo colonoscopic surveillance and subsequent colectomy, as well as endometrial aspiration and subsequent total abdominal hysterectomy-bilateral salpingo-oophorectomy. Dr. Syngal reviewed testing of tumors for microsatellite instability and immunohistochemistry for the DNA mismatch repair genes (mutation of which cause Lynch syndrome) and discussed prediction models for germline testing.
John M. Carethers, M.D., is the John G. Searle Professor and chair of the department of internal medicine at the University of Michigan, Ann Arbor.
The Screening, Surveillance, and Molecular Testing for Gastrointestinal Cancers session of the 2012 AGA Spring Postgraduate Course contained five outstanding speakers who discussed cancers of the gastrointestinal tract or their precursor lesions, which gastroenterologists face frequently in the clinic.
Dr. John Inadomi from the University of Washington, Seattle, highlighted some evidence-based and practical approaches to care for patients who have developed or are at risk for Barrett’s esophagus.
Dr. Inadomi pointed out that our approach to screening should only involve those patients with multiple risk factors, such as white males with chronic heartburn and acid reflux, tobacco usage, and increased central obesity. Once Barrett’s is detected, there is low effectiveness of surveillance for Barrett’s to reduce esophageal cancer mortality, and is not cost-effective. This is largely due to the low observed conversion of Barrett’s esophagus to esophageal adenocarcinoma (less than 0.5% of patients/year), and that surveillance relies on repeated measures of a poor marker (i.e. random biopsies). Endoscopic ablation therapy is largely reserved for those patients with high-grade dysplasia (HGD), some patients with low-grade dysplasia (LGD), and is not for those with non-dysplastic Barrett’s esophagus. Ablation of patients with HGD and as well as some patients with LGD is cost-effective and may decrease cancer incidence. Dr. Inadomi also presented some new approaches for the management of Barrett’s and its potential complications, included advanced imaging techniques, the use of molecular diagnostics, and chemoprevention.
Dr. Asif Khalid from the University of Pittsburgh presented an approach to evaluation and care of the patient with cystic lesions of the pancreas. Dr. Khalid pointed out that serous cystic tumors are largely benign, are equal among males and females, and often have a central scar or honeycomb pattern on imaging. Mucinous neoplasms are a different story, and need to be carefully evaluated for potential resection. The mucinous cystic neoplasm (MCN) is premalignant, occurs more commonly in females, may present with septations on imaging, produces carcinoembryonic antigen (CEA), and needs resection. Intraductal papillary mucinous neoplasms (IPMNs) can be main duct, which requires resection as the only curative option, or branch duct, and requires surgical resection if at least 3 cm. IPMNs are equal between males and females, produce CEA, and may demonstrate mutation in the GNAS gene. Diagnostic differentiation between lesions can be challenging, but with imaging (endoscopic ultrasound, MRI, and CT) and aspiration of the lesion for cytology, amylase, CEA, and other molecular markers, differentiation is possible. Dr. Khalid stated that key features that differentiate mucinous from non-mucinous lesions are the presence of mutant KRAS and elevated CEA levels in the aspirate, and the presence of allelic loss in DNA taken in the sample. He also pointed out that key features to help differentiate malignant from non-malignant lesions include high DNA content, mutant KRAS, and allelic loss.
Dr. Hashem El-Serag from Baylor College of Medicine, Houston, showcased information on hepatocellular carcinoma (HCC). He showed evidence that the incidence of HCC is increasing, and that survival from HCC is marginally increasing as well with improved approaches to care. Dr. El-Serag reviewed the Milan criteria, where a person is potentially curative by resection or liver transplant if there are three or fewer nodules that are 3 cm or less in diameter. Dr. El-Serag presented data showing that surveillance for HCC reduces mortality from HCC. Efforts should be focused on Asian hepatitis B carriers, those with a family history of HCC, blacks from Africa, and patients with cirrhosis who have hepatitis B or C. Surveillance should be done every 6 months, and the combined use of ultrasound (with a sensitivity of 72%) with alpha-fetoprotein (AFP) is the best studied. Other biomarkers in the literature for HCC screening include des-gamma-carboxy prothrombin (DCP), golgi protein-73, and osteopontin. Dr. El-Serag also reviewed key aspects of imaging criteria for identifying HCC in cirrhotic patients, which includes hyper-enhancement by two imaging techniques if a lesion is at least 1 cm but less than 2 cm in diameter, and by one imaging technique if 2 cm or greater, with or without liver biopsy if the appearance is atypical.
Dr. Douglas Rex from Indiana University, Indianapolis, highlighted several quality issues in regard to screening for colorectal cancer. He stated that the primary contribution of gastroenterologists to colorectal cancer prevention is high quality colonoscopy. Dr. Rex pointed out recent evidence that colonoscopy is less effective in the right colon, compared with the left colon, and one contribution to this is the right-sided location of serrated, flat, and depressed lesions that blend in with the surrounding mucosa. For all colorectal screening tests, there is limited evidence regarding sensitivity for serrated lesions. Dr. Rex provided evidence that guaiac-based fecal occult blood test (FOBT) screening should all be now converted to fecal immunohistochemical test (FIT) screening due to enhanced sensitivity of the FIT tests. Dr. Rex also provided initial data on the first serum-screening assay for colorectal cancer, which was unable to detect precancerous lesions.
Dr. Sapna Syngal from the Dana-Farber Cancer Center in Boston presented on hereditary colorectal cancer syndromes. These syndromes elevate the risk for colorectal cancer, and if they are not recognized, their associated cancers present with a young age of onset. Patients and potential patients need to be referred for genetic counseling and testing, particularly those who present at a young age, have a high number of polyps, show upper GI lesions, and have strong family history of cancer or polyps. Dr. Syngal highlighted the issue of "genetic malpractice" that is growing in the United States due to a lack of recognition and referral for evaluation. These syndromes are managed much differently than sporadic cancer in their approach to surgery, surveillance for associated cancers, the screening of family members, and reproductive counseling and prenatal testing.
Dr. Syngal reviewed familial adenomatous polyposis (FAP), an autosomal dominant condition, and highlighted MYH associated polyposis (MAP), an autosomal recessive condition. Both FAP and MAP can present with colon polyps and cancer, upper GI lesions, form desmoid tumors, and are associated with thyroid, brain, and osteoma tumors. Care is through colectomy, upper GI surveillance, and chemoprevention for any lower GI remnant. Dr. Syngal also highlighted Lynch syndrome, which is associated with tumors of the colon and upper GI track, female reproductive track, urinary track, and sebaceous skin tumors. Lynch syndrome patients can undergo colonoscopic surveillance and subsequent colectomy, as well as endometrial aspiration and subsequent total abdominal hysterectomy-bilateral salpingo-oophorectomy. Dr. Syngal reviewed testing of tumors for microsatellite instability and immunohistochemistry for the DNA mismatch repair genes (mutation of which cause Lynch syndrome) and discussed prediction models for germline testing.
John M. Carethers, M.D., is the John G. Searle Professor and chair of the department of internal medicine at the University of Michigan, Ann Arbor.