User login
The Ruth Leff Siegel Award for Excellence in Pancreatic Cancer Research
The Pancreas Center at Columbia University and the Herbert Irving Comprehensive Cancer Center recently announced a call for nominations for the first annual Ruth Leff Siegel Award for Excellence in Pancreatic Cancer Research. Nominators should identify the investigator who has made an impactful contribution to the understanding and/or treatment of pancreatic cancer over the past year and who had a track record of high-quality work in the field. The work can be in any field of pancreatic cancer research including but not limited to basic biology, population biology, public health, and translational science. For more details, contact award coordinator Colina Chapman at 212-305-9468 or [email protected].
The Pancreas Center at Columbia University and the Herbert Irving Comprehensive Cancer Center recently announced a call for nominations for the first annual Ruth Leff Siegel Award for Excellence in Pancreatic Cancer Research. Nominators should identify the investigator who has made an impactful contribution to the understanding and/or treatment of pancreatic cancer over the past year and who had a track record of high-quality work in the field. The work can be in any field of pancreatic cancer research including but not limited to basic biology, population biology, public health, and translational science. For more details, contact award coordinator Colina Chapman at 212-305-9468 or [email protected].
The Pancreas Center at Columbia University and the Herbert Irving Comprehensive Cancer Center recently announced a call for nominations for the first annual Ruth Leff Siegel Award for Excellence in Pancreatic Cancer Research. Nominators should identify the investigator who has made an impactful contribution to the understanding and/or treatment of pancreatic cancer over the past year and who had a track record of high-quality work in the field. The work can be in any field of pancreatic cancer research including but not limited to basic biology, population biology, public health, and translational science. For more details, contact award coordinator Colina Chapman at 212-305-9468 or [email protected].
AGA creates Center for Gut Microbiome Research and Education
In a healthy adult, microbial cells are estimated to outnumber human cells 10 to 1. Many microbes maintain our health, while others cause illness. Recent investigations of the human gut microbiome have discovered important ways in which gut microbes may influence important disease states such as obesity, inflammatory bowel disease, functional GI disorders, GI cancers, and liver disease. Recognizing the major influence the gut microbiome is likely to have on the future of GI research and patient care, the AGA Governing Board is pleased to announce the creation of the AGA Center for Gut Microbiome Research and Education.
"The gut microbiome is among the most exciting and promising areas of research today. As gastroenterologists we are in an excellent position to perform gut microbiome research and translate basic discoveries into new methods to maintain and improve the health of our patients," said Loren Laine, M.D., president of the AGA Institute. "The gut microbiome offers a myriad of possibilities to GI basic and translational researchers, clinicians and patients."
The AGA Center for Microbiome Research and Education will be a virtual "home" for the AGA’s activities related to the gut microbiome. The mission of the Center is "To advance research and education on the gut microbiome in human health and disease."
To provide guidance on gut microbiome-related issues, the AGA has convened a scientific advisory board comprised of world leaders in computational biology and metagenomics, microbiome animal models, microbiology translational research, nutrition, and pertinent regulatory and policy issues. These advisors will make strategic and programmatic recommendations to the AGA Governing Board.
"This is an incredibly exciting time in science, where technological advances in DNA sequencing, transcriptomics, proteomics, and metabolomics provide an unprecedented opportunity to explore not only the composition, but also the function of the microbial communities that live in our intestinal tract. It is hoped that the knowledge gained will provide new insights into disease pathogenesis and innovative therapeutic modalities. The membership of the AGA is ideally suited to translate these findings from the bench to the bedside," said Gary Wu, M.D., chair of the scientific advisory board.
Scientific advisory board members:
• Gary D. Wu, M.D., chair
Professor of Medicine, Perelman School of Medicine at the University of Pennsylvania, Philadelphia
• Martin J. Blaser, M.D.
George and Muriel Singer Professor of Medicine, Professor of Microbiology and Director, Human Microbiome Program at the New York University Langone Medical Center, New York
• Jeffrey I. Gordon, M.D.
Dr. Robert J. Glaser Distinguished University Professor and Director of the Center for Genome Sciences & Systems Biology at Washington University, St. Louis
• Gail Hecht, M.D.
Professor of Medicine; Microbiology/Immunology and Chief, Gastroenterology and Nutrition at Loyola University Medical Center, Maywood, IL
• Lee M. Kaplan, M.D., Ph.D.
Director, Obesity, Metabolism & Nutrition Institute, Gastrointestinal Unit at Massachusetts General Hospital, Boston
• Rob Knight, Ph.D.
Associate Professor, University of Colorado, Boulder
• Mary Ellen Sanders, Ph.D.
Executive Director of the International Scientific Association for Probiotics and Prebiotics, Centennial, CO
• Ryan Balfour Sartor, M.D.
Distinguished Professor, Medicine, Microbiology and Immunology at the University of North Carolina – Chapel Hill
The AGA will fund the center’s activities and will seek funds from private, nonprofit, and governmental sources to help support the center.
Danone is a founding supporter of the center.
In a healthy adult, microbial cells are estimated to outnumber human cells 10 to 1. Many microbes maintain our health, while others cause illness. Recent investigations of the human gut microbiome have discovered important ways in which gut microbes may influence important disease states such as obesity, inflammatory bowel disease, functional GI disorders, GI cancers, and liver disease. Recognizing the major influence the gut microbiome is likely to have on the future of GI research and patient care, the AGA Governing Board is pleased to announce the creation of the AGA Center for Gut Microbiome Research and Education.
"The gut microbiome is among the most exciting and promising areas of research today. As gastroenterologists we are in an excellent position to perform gut microbiome research and translate basic discoveries into new methods to maintain and improve the health of our patients," said Loren Laine, M.D., president of the AGA Institute. "The gut microbiome offers a myriad of possibilities to GI basic and translational researchers, clinicians and patients."
The AGA Center for Microbiome Research and Education will be a virtual "home" for the AGA’s activities related to the gut microbiome. The mission of the Center is "To advance research and education on the gut microbiome in human health and disease."
To provide guidance on gut microbiome-related issues, the AGA has convened a scientific advisory board comprised of world leaders in computational biology and metagenomics, microbiome animal models, microbiology translational research, nutrition, and pertinent regulatory and policy issues. These advisors will make strategic and programmatic recommendations to the AGA Governing Board.
"This is an incredibly exciting time in science, where technological advances in DNA sequencing, transcriptomics, proteomics, and metabolomics provide an unprecedented opportunity to explore not only the composition, but also the function of the microbial communities that live in our intestinal tract. It is hoped that the knowledge gained will provide new insights into disease pathogenesis and innovative therapeutic modalities. The membership of the AGA is ideally suited to translate these findings from the bench to the bedside," said Gary Wu, M.D., chair of the scientific advisory board.
Scientific advisory board members:
• Gary D. Wu, M.D., chair
Professor of Medicine, Perelman School of Medicine at the University of Pennsylvania, Philadelphia
• Martin J. Blaser, M.D.
George and Muriel Singer Professor of Medicine, Professor of Microbiology and Director, Human Microbiome Program at the New York University Langone Medical Center, New York
• Jeffrey I. Gordon, M.D.
Dr. Robert J. Glaser Distinguished University Professor and Director of the Center for Genome Sciences & Systems Biology at Washington University, St. Louis
• Gail Hecht, M.D.
Professor of Medicine; Microbiology/Immunology and Chief, Gastroenterology and Nutrition at Loyola University Medical Center, Maywood, IL
• Lee M. Kaplan, M.D., Ph.D.
Director, Obesity, Metabolism & Nutrition Institute, Gastrointestinal Unit at Massachusetts General Hospital, Boston
• Rob Knight, Ph.D.
Associate Professor, University of Colorado, Boulder
• Mary Ellen Sanders, Ph.D.
Executive Director of the International Scientific Association for Probiotics and Prebiotics, Centennial, CO
• Ryan Balfour Sartor, M.D.
Distinguished Professor, Medicine, Microbiology and Immunology at the University of North Carolina – Chapel Hill
The AGA will fund the center’s activities and will seek funds from private, nonprofit, and governmental sources to help support the center.
Danone is a founding supporter of the center.
In a healthy adult, microbial cells are estimated to outnumber human cells 10 to 1. Many microbes maintain our health, while others cause illness. Recent investigations of the human gut microbiome have discovered important ways in which gut microbes may influence important disease states such as obesity, inflammatory bowel disease, functional GI disorders, GI cancers, and liver disease. Recognizing the major influence the gut microbiome is likely to have on the future of GI research and patient care, the AGA Governing Board is pleased to announce the creation of the AGA Center for Gut Microbiome Research and Education.
"The gut microbiome is among the most exciting and promising areas of research today. As gastroenterologists we are in an excellent position to perform gut microbiome research and translate basic discoveries into new methods to maintain and improve the health of our patients," said Loren Laine, M.D., president of the AGA Institute. "The gut microbiome offers a myriad of possibilities to GI basic and translational researchers, clinicians and patients."
The AGA Center for Microbiome Research and Education will be a virtual "home" for the AGA’s activities related to the gut microbiome. The mission of the Center is "To advance research and education on the gut microbiome in human health and disease."
To provide guidance on gut microbiome-related issues, the AGA has convened a scientific advisory board comprised of world leaders in computational biology and metagenomics, microbiome animal models, microbiology translational research, nutrition, and pertinent regulatory and policy issues. These advisors will make strategic and programmatic recommendations to the AGA Governing Board.
"This is an incredibly exciting time in science, where technological advances in DNA sequencing, transcriptomics, proteomics, and metabolomics provide an unprecedented opportunity to explore not only the composition, but also the function of the microbial communities that live in our intestinal tract. It is hoped that the knowledge gained will provide new insights into disease pathogenesis and innovative therapeutic modalities. The membership of the AGA is ideally suited to translate these findings from the bench to the bedside," said Gary Wu, M.D., chair of the scientific advisory board.
Scientific advisory board members:
• Gary D. Wu, M.D., chair
Professor of Medicine, Perelman School of Medicine at the University of Pennsylvania, Philadelphia
• Martin J. Blaser, M.D.
George and Muriel Singer Professor of Medicine, Professor of Microbiology and Director, Human Microbiome Program at the New York University Langone Medical Center, New York
• Jeffrey I. Gordon, M.D.
Dr. Robert J. Glaser Distinguished University Professor and Director of the Center for Genome Sciences & Systems Biology at Washington University, St. Louis
• Gail Hecht, M.D.
Professor of Medicine; Microbiology/Immunology and Chief, Gastroenterology and Nutrition at Loyola University Medical Center, Maywood, IL
• Lee M. Kaplan, M.D., Ph.D.
Director, Obesity, Metabolism & Nutrition Institute, Gastrointestinal Unit at Massachusetts General Hospital, Boston
• Rob Knight, Ph.D.
Associate Professor, University of Colorado, Boulder
• Mary Ellen Sanders, Ph.D.
Executive Director of the International Scientific Association for Probiotics and Prebiotics, Centennial, CO
• Ryan Balfour Sartor, M.D.
Distinguished Professor, Medicine, Microbiology and Immunology at the University of North Carolina – Chapel Hill
The AGA will fund the center’s activities and will seek funds from private, nonprofit, and governmental sources to help support the center.
Danone is a founding supporter of the center.
AGA Legacy Society members sustain GI research
Research creates successful practices. Patients benefit from GI research daily in practices. Scientists are working hard to develop new treatments and therapies and to discover cures to advance the field and better patient care. But they can’t do this without research funding.
AGA Legacy Society members have answered this call for support. They recognize the value that research has had in their profession, both in academic medicine and in private practice, and are showing their appreciation by giving back.
"I give back because I have a firsthand knowledge of what it will mean to a young investigator’s career," said Legacy Society member Shrikant Anant, Ph.D., AGAF, of the University of Kansas, Kansas City. "I was propelled in my career when I received the 2002 Research Scholar Award from the AGA Research Foundation. The funds helped me develop my independent research that has led to many NIH grants and, associated with it, career advancement. I still vividly remember the day I received the notice of award and how my whole life changed. Today, I am proud to be a donor myself because I know it is making a difference [for] yet another young investigator."
The AGA Legacy Society boasts 110 members. Members of the AGA Legacy Society provide tax-deductible gifts to the AGA Research Foundation of $25,000 (payable over 5 years) or $50,000 in a planned gift, such as a bequest.
The AGA Research Foundation’s mission is to raise money to support young researchers in gastroenterology and hepatology. More than 700 researchers have benefited from our support since 1984 – with more than 90% of AGA Research Scholar Award recipients in the past 10 years continuing on to exceptional research careers. These research grants are funded through the generosity of donors.
Recently, the AGA Research Foundation has awarded nearly $1.2 million in grants each year. In the last 5 years, however, over $30 million in qualified applications went unfunded. The AGA Research Foundation seeks to do more.
AGA Legacy Society members see the promise the future holds and are committed to furthering research in gastroenterology and hepatology.
"I donate because I look forward to seeing more and more innovation in gastroenterology," states Dr. John Allen, AGAF, AGA Vice President and Legacy Society member. "Our specialty has been so good to me and my partners. We provide care to over 120,000 patients each year. We see so many areas where research and new knowledge are needed. The least I can do is give back and support new science."
A worthy acknowledgement
Beginning with a memorable gathering at the United States Library of Congress in 2007, the Benefactor’s Dinner has welcomed members of the AGA Legacy Society and other AGA dignitaries to special locations nationwide. Rosen Shingle Creek will be the location of the 2013 Benefactor’s Dinner of the AGA Research Foundation during Digestive Diseases Week this year. The magical location pays tribute to Florida’s rich history. Members of the AGA Legacy Society will be among the distinguished honorees at the annual event.
Donors who make gifts at the Legacy Society level anytime before Digestive Diseases Week will receive an invitation to the Benefactor’s Dinner in Orlando. Individuals interested in learning more about Legacy Society membership may contact Stacey Hinton Tuneski, Senior Director of Development, at [email protected] or via phone at (301) 222-4005. Legacy Society information and acceptance forms are also available on the foundation’s website at www.gastro.org/legacysociety.
Research creates successful practices. Patients benefit from GI research daily in practices. Scientists are working hard to develop new treatments and therapies and to discover cures to advance the field and better patient care. But they can’t do this without research funding.
AGA Legacy Society members have answered this call for support. They recognize the value that research has had in their profession, both in academic medicine and in private practice, and are showing their appreciation by giving back.
"I give back because I have a firsthand knowledge of what it will mean to a young investigator’s career," said Legacy Society member Shrikant Anant, Ph.D., AGAF, of the University of Kansas, Kansas City. "I was propelled in my career when I received the 2002 Research Scholar Award from the AGA Research Foundation. The funds helped me develop my independent research that has led to many NIH grants and, associated with it, career advancement. I still vividly remember the day I received the notice of award and how my whole life changed. Today, I am proud to be a donor myself because I know it is making a difference [for] yet another young investigator."
The AGA Legacy Society boasts 110 members. Members of the AGA Legacy Society provide tax-deductible gifts to the AGA Research Foundation of $25,000 (payable over 5 years) or $50,000 in a planned gift, such as a bequest.
The AGA Research Foundation’s mission is to raise money to support young researchers in gastroenterology and hepatology. More than 700 researchers have benefited from our support since 1984 – with more than 90% of AGA Research Scholar Award recipients in the past 10 years continuing on to exceptional research careers. These research grants are funded through the generosity of donors.
Recently, the AGA Research Foundation has awarded nearly $1.2 million in grants each year. In the last 5 years, however, over $30 million in qualified applications went unfunded. The AGA Research Foundation seeks to do more.
AGA Legacy Society members see the promise the future holds and are committed to furthering research in gastroenterology and hepatology.
"I donate because I look forward to seeing more and more innovation in gastroenterology," states Dr. John Allen, AGAF, AGA Vice President and Legacy Society member. "Our specialty has been so good to me and my partners. We provide care to over 120,000 patients each year. We see so many areas where research and new knowledge are needed. The least I can do is give back and support new science."
A worthy acknowledgement
Beginning with a memorable gathering at the United States Library of Congress in 2007, the Benefactor’s Dinner has welcomed members of the AGA Legacy Society and other AGA dignitaries to special locations nationwide. Rosen Shingle Creek will be the location of the 2013 Benefactor’s Dinner of the AGA Research Foundation during Digestive Diseases Week this year. The magical location pays tribute to Florida’s rich history. Members of the AGA Legacy Society will be among the distinguished honorees at the annual event.
Donors who make gifts at the Legacy Society level anytime before Digestive Diseases Week will receive an invitation to the Benefactor’s Dinner in Orlando. Individuals interested in learning more about Legacy Society membership may contact Stacey Hinton Tuneski, Senior Director of Development, at [email protected] or via phone at (301) 222-4005. Legacy Society information and acceptance forms are also available on the foundation’s website at www.gastro.org/legacysociety.
Research creates successful practices. Patients benefit from GI research daily in practices. Scientists are working hard to develop new treatments and therapies and to discover cures to advance the field and better patient care. But they can’t do this without research funding.
AGA Legacy Society members have answered this call for support. They recognize the value that research has had in their profession, both in academic medicine and in private practice, and are showing their appreciation by giving back.
"I give back because I have a firsthand knowledge of what it will mean to a young investigator’s career," said Legacy Society member Shrikant Anant, Ph.D., AGAF, of the University of Kansas, Kansas City. "I was propelled in my career when I received the 2002 Research Scholar Award from the AGA Research Foundation. The funds helped me develop my independent research that has led to many NIH grants and, associated with it, career advancement. I still vividly remember the day I received the notice of award and how my whole life changed. Today, I am proud to be a donor myself because I know it is making a difference [for] yet another young investigator."
The AGA Legacy Society boasts 110 members. Members of the AGA Legacy Society provide tax-deductible gifts to the AGA Research Foundation of $25,000 (payable over 5 years) or $50,000 in a planned gift, such as a bequest.
The AGA Research Foundation’s mission is to raise money to support young researchers in gastroenterology and hepatology. More than 700 researchers have benefited from our support since 1984 – with more than 90% of AGA Research Scholar Award recipients in the past 10 years continuing on to exceptional research careers. These research grants are funded through the generosity of donors.
Recently, the AGA Research Foundation has awarded nearly $1.2 million in grants each year. In the last 5 years, however, over $30 million in qualified applications went unfunded. The AGA Research Foundation seeks to do more.
AGA Legacy Society members see the promise the future holds and are committed to furthering research in gastroenterology and hepatology.
"I donate because I look forward to seeing more and more innovation in gastroenterology," states Dr. John Allen, AGAF, AGA Vice President and Legacy Society member. "Our specialty has been so good to me and my partners. We provide care to over 120,000 patients each year. We see so many areas where research and new knowledge are needed. The least I can do is give back and support new science."
A worthy acknowledgement
Beginning with a memorable gathering at the United States Library of Congress in 2007, the Benefactor’s Dinner has welcomed members of the AGA Legacy Society and other AGA dignitaries to special locations nationwide. Rosen Shingle Creek will be the location of the 2013 Benefactor’s Dinner of the AGA Research Foundation during Digestive Diseases Week this year. The magical location pays tribute to Florida’s rich history. Members of the AGA Legacy Society will be among the distinguished honorees at the annual event.
Donors who make gifts at the Legacy Society level anytime before Digestive Diseases Week will receive an invitation to the Benefactor’s Dinner in Orlando. Individuals interested in learning more about Legacy Society membership may contact Stacey Hinton Tuneski, Senior Director of Development, at [email protected] or via phone at (301) 222-4005. Legacy Society information and acceptance forms are also available on the foundation’s website at www.gastro.org/legacysociety.
New opportunities arise for infection prevention in endoscopy
More than 3 million colonoscopies were performed in freestanding ambulatory facilities in 2006. Very specific national clinical practice guideline recommendations and manufacturers’ directions address instruction and competency testing for all staff who process endoscopes, as well as sterilization of reusable forceps/cutting instruments that break the mucosal barrier, before each use, and high-level disinfection (HLD) fluid testing and management. All accrediting bodies require ambulatory surgical centers to adhere to professional society guidelines and manufacturer recommendations for cleaning, disinfection, and sterilization of instruments and equipment.
The research literature indicates that, with the exception of unsafe injection practices, infectious outbreaks associated with colonoscopy procedures have been tied to lapses in instrument processing. Although the overall number of infectious outbreaks associated with colonoscopies is relatively low, these incidents were repeatedly reported in the research literature over the last couple of decades. In addition to the direct costs of infectious outbreaks, the expenses of patient follow-up, testing, and treatment/management are very high.
"Due to several unfortunate and highly publicized lapses, public scrutiny is focused upon infection control practices in endoscopy," said Dr. Lawrence S. Kim, AGAF, AGA representative to the Accreditation Association for Ambulatory Health Care (AAAHC). "AGA is committed to ensuring patient safety during endoscopic procedures; to this end, we have played an integral role in developing evidence-based guidelines and communicate regularly with the Food and Drug Administration (FDA), manufacturers, and accrediting bodies regarding reprocessing and disinfection issues. It is incumbent upon all gastroenterologists to follow recognized standards for cleaning and disinfection to protect the well-being of our patients as well as the integrity of our specialty."
From July to December 2012, the AAAHC Institute gathered information on organizational attributes and processes of 100 organizations that perform 60-12,800 colonoscopies each year, representing a total of more than 353,307 annually.
The AAAHC Institute found that:
• Ninety-nine percent of responding organizations complied with all of the following clinical practice guideline-recommended processes prior to sterilization or HLD: leak testing; cleaning with an enzyme cleaner that is compatible with the scope; flushing and brushing all channels and ports; cleaning all external surfaces and accessories; and cleaning residue/debris until no more debris appears on cleaning brushes.
• However, only 88% of responding organizations indicated that they follow all of the recommended steps for testing fluids used for sterilization or HLD: test at least every day of use; test prior to each cycle/use; use the manufacturer’s recommended chemical indicator; document the results of testing; discard the solution if the chemical indicator shows the concentration is less than the manufacturer’s minimum effective concentration; and discard the solution if it is beyond the manufacturer’s recommended shelf or use life.
• And, only 59% of the 27 responding organizations that reported the use of reusable cutting instruments indicated that they follow both recommended processes (mechanically cleaning reusable biopsy forceps or other cutting instruments that break the mucosal barrier and sterilizing these) between each patient.
Although study results indicate a high percentage of compliance with presterilization/high-level disinfection processes, there are clear opportunities to increase compliance with national guidelines and manufacturers’ recommendations for sterilant fluid testing and management, as well as processing of reusable cutting instruments. This improvement will result in increasing patient safety by lowering the rates of health care–acquired infections (HAIs) and the cost associated with testing for and treating or managing HAIs.
AGA was the first gastroenterology member organization of AAAHC, and AGA’s representatives have played important leadership roles in this accrediting body. In October, AGA was a supporting organization in the FDA-Association for the Advancement of Medical Instrumentation Reprocessing Summit, which addressed not only the regulatory and economic environments, but also the current science of reprocessing.
Please review the AGA-endorsed 2011 revised multisociety guideline for reprocessing of flexible endoscopes at http://ow.ly/hxQC5.
More than 3 million colonoscopies were performed in freestanding ambulatory facilities in 2006. Very specific national clinical practice guideline recommendations and manufacturers’ directions address instruction and competency testing for all staff who process endoscopes, as well as sterilization of reusable forceps/cutting instruments that break the mucosal barrier, before each use, and high-level disinfection (HLD) fluid testing and management. All accrediting bodies require ambulatory surgical centers to adhere to professional society guidelines and manufacturer recommendations for cleaning, disinfection, and sterilization of instruments and equipment.
The research literature indicates that, with the exception of unsafe injection practices, infectious outbreaks associated with colonoscopy procedures have been tied to lapses in instrument processing. Although the overall number of infectious outbreaks associated with colonoscopies is relatively low, these incidents were repeatedly reported in the research literature over the last couple of decades. In addition to the direct costs of infectious outbreaks, the expenses of patient follow-up, testing, and treatment/management are very high.
"Due to several unfortunate and highly publicized lapses, public scrutiny is focused upon infection control practices in endoscopy," said Dr. Lawrence S. Kim, AGAF, AGA representative to the Accreditation Association for Ambulatory Health Care (AAAHC). "AGA is committed to ensuring patient safety during endoscopic procedures; to this end, we have played an integral role in developing evidence-based guidelines and communicate regularly with the Food and Drug Administration (FDA), manufacturers, and accrediting bodies regarding reprocessing and disinfection issues. It is incumbent upon all gastroenterologists to follow recognized standards for cleaning and disinfection to protect the well-being of our patients as well as the integrity of our specialty."
From July to December 2012, the AAAHC Institute gathered information on organizational attributes and processes of 100 organizations that perform 60-12,800 colonoscopies each year, representing a total of more than 353,307 annually.
The AAAHC Institute found that:
• Ninety-nine percent of responding organizations complied with all of the following clinical practice guideline-recommended processes prior to sterilization or HLD: leak testing; cleaning with an enzyme cleaner that is compatible with the scope; flushing and brushing all channels and ports; cleaning all external surfaces and accessories; and cleaning residue/debris until no more debris appears on cleaning brushes.
• However, only 88% of responding organizations indicated that they follow all of the recommended steps for testing fluids used for sterilization or HLD: test at least every day of use; test prior to each cycle/use; use the manufacturer’s recommended chemical indicator; document the results of testing; discard the solution if the chemical indicator shows the concentration is less than the manufacturer’s minimum effective concentration; and discard the solution if it is beyond the manufacturer’s recommended shelf or use life.
• And, only 59% of the 27 responding organizations that reported the use of reusable cutting instruments indicated that they follow both recommended processes (mechanically cleaning reusable biopsy forceps or other cutting instruments that break the mucosal barrier and sterilizing these) between each patient.
Although study results indicate a high percentage of compliance with presterilization/high-level disinfection processes, there are clear opportunities to increase compliance with national guidelines and manufacturers’ recommendations for sterilant fluid testing and management, as well as processing of reusable cutting instruments. This improvement will result in increasing patient safety by lowering the rates of health care–acquired infections (HAIs) and the cost associated with testing for and treating or managing HAIs.
AGA was the first gastroenterology member organization of AAAHC, and AGA’s representatives have played important leadership roles in this accrediting body. In October, AGA was a supporting organization in the FDA-Association for the Advancement of Medical Instrumentation Reprocessing Summit, which addressed not only the regulatory and economic environments, but also the current science of reprocessing.
Please review the AGA-endorsed 2011 revised multisociety guideline for reprocessing of flexible endoscopes at http://ow.ly/hxQC5.
More than 3 million colonoscopies were performed in freestanding ambulatory facilities in 2006. Very specific national clinical practice guideline recommendations and manufacturers’ directions address instruction and competency testing for all staff who process endoscopes, as well as sterilization of reusable forceps/cutting instruments that break the mucosal barrier, before each use, and high-level disinfection (HLD) fluid testing and management. All accrediting bodies require ambulatory surgical centers to adhere to professional society guidelines and manufacturer recommendations for cleaning, disinfection, and sterilization of instruments and equipment.
The research literature indicates that, with the exception of unsafe injection practices, infectious outbreaks associated with colonoscopy procedures have been tied to lapses in instrument processing. Although the overall number of infectious outbreaks associated with colonoscopies is relatively low, these incidents were repeatedly reported in the research literature over the last couple of decades. In addition to the direct costs of infectious outbreaks, the expenses of patient follow-up, testing, and treatment/management are very high.
"Due to several unfortunate and highly publicized lapses, public scrutiny is focused upon infection control practices in endoscopy," said Dr. Lawrence S. Kim, AGAF, AGA representative to the Accreditation Association for Ambulatory Health Care (AAAHC). "AGA is committed to ensuring patient safety during endoscopic procedures; to this end, we have played an integral role in developing evidence-based guidelines and communicate regularly with the Food and Drug Administration (FDA), manufacturers, and accrediting bodies regarding reprocessing and disinfection issues. It is incumbent upon all gastroenterologists to follow recognized standards for cleaning and disinfection to protect the well-being of our patients as well as the integrity of our specialty."
From July to December 2012, the AAAHC Institute gathered information on organizational attributes and processes of 100 organizations that perform 60-12,800 colonoscopies each year, representing a total of more than 353,307 annually.
The AAAHC Institute found that:
• Ninety-nine percent of responding organizations complied with all of the following clinical practice guideline-recommended processes prior to sterilization or HLD: leak testing; cleaning with an enzyme cleaner that is compatible with the scope; flushing and brushing all channels and ports; cleaning all external surfaces and accessories; and cleaning residue/debris until no more debris appears on cleaning brushes.
• However, only 88% of responding organizations indicated that they follow all of the recommended steps for testing fluids used for sterilization or HLD: test at least every day of use; test prior to each cycle/use; use the manufacturer’s recommended chemical indicator; document the results of testing; discard the solution if the chemical indicator shows the concentration is less than the manufacturer’s minimum effective concentration; and discard the solution if it is beyond the manufacturer’s recommended shelf or use life.
• And, only 59% of the 27 responding organizations that reported the use of reusable cutting instruments indicated that they follow both recommended processes (mechanically cleaning reusable biopsy forceps or other cutting instruments that break the mucosal barrier and sterilizing these) between each patient.
Although study results indicate a high percentage of compliance with presterilization/high-level disinfection processes, there are clear opportunities to increase compliance with national guidelines and manufacturers’ recommendations for sterilant fluid testing and management, as well as processing of reusable cutting instruments. This improvement will result in increasing patient safety by lowering the rates of health care–acquired infections (HAIs) and the cost associated with testing for and treating or managing HAIs.
AGA was the first gastroenterology member organization of AAAHC, and AGA’s representatives have played important leadership roles in this accrediting body. In October, AGA was a supporting organization in the FDA-Association for the Advancement of Medical Instrumentation Reprocessing Summit, which addressed not only the regulatory and economic environments, but also the current science of reprocessing.
Please review the AGA-endorsed 2011 revised multisociety guideline for reprocessing of flexible endoscopes at http://ow.ly/hxQC5.
Introducing the GI Coding Advisor
AGA, Ask Mueller Consulting, LLC, and Medical Code Solutions recently launched the GI Coding Advisor. This web-based tool allows practices to maximize reimbursements, reduce denials, and improve compliance by providing easy access to the most current and authoritative coding information available.
Designed and regularly updated by the foremost coding expert in the field, AGA’s own coding and billing specialist Kathleen Mueller, the GI Coding Advisor was created specifically for GI practices and is sure to help improve your bottom line.
The GI Coding Advisor is web based, always updated, and quick and easy to use. AGA members save 15%.
These are just a few of the many benefits:
• Easily identify missed billing opportunities by using Kathy’s expert advice associated to your GI codes.
• Customize your coding for local/regional/state and national regulations with instant access to national coverage determinations and local coverage determinations.
• Avoid costly denials as you tap into a vast database that explains how to properly code your diagnoses and procedures.
• Protect your practice’s profits by using Kathy’s documentation tips that help you dodge auditing flags.
• Get ready for ICD-10 implementation by using the GI Coding Advisor ICD-10 translator.
To receive a free 14-day subscription and register for the online demo, visit www.gicodingadvisor.org.
AGA, Ask Mueller Consulting, LLC, and Medical Code Solutions recently launched the GI Coding Advisor. This web-based tool allows practices to maximize reimbursements, reduce denials, and improve compliance by providing easy access to the most current and authoritative coding information available.
Designed and regularly updated by the foremost coding expert in the field, AGA’s own coding and billing specialist Kathleen Mueller, the GI Coding Advisor was created specifically for GI practices and is sure to help improve your bottom line.
The GI Coding Advisor is web based, always updated, and quick and easy to use. AGA members save 15%.
These are just a few of the many benefits:
• Easily identify missed billing opportunities by using Kathy’s expert advice associated to your GI codes.
• Customize your coding for local/regional/state and national regulations with instant access to national coverage determinations and local coverage determinations.
• Avoid costly denials as you tap into a vast database that explains how to properly code your diagnoses and procedures.
• Protect your practice’s profits by using Kathy’s documentation tips that help you dodge auditing flags.
• Get ready for ICD-10 implementation by using the GI Coding Advisor ICD-10 translator.
To receive a free 14-day subscription and register for the online demo, visit www.gicodingadvisor.org.
AGA, Ask Mueller Consulting, LLC, and Medical Code Solutions recently launched the GI Coding Advisor. This web-based tool allows practices to maximize reimbursements, reduce denials, and improve compliance by providing easy access to the most current and authoritative coding information available.
Designed and regularly updated by the foremost coding expert in the field, AGA’s own coding and billing specialist Kathleen Mueller, the GI Coding Advisor was created specifically for GI practices and is sure to help improve your bottom line.
The GI Coding Advisor is web based, always updated, and quick and easy to use. AGA members save 15%.
These are just a few of the many benefits:
• Easily identify missed billing opportunities by using Kathy’s expert advice associated to your GI codes.
• Customize your coding for local/regional/state and national regulations with instant access to national coverage determinations and local coverage determinations.
• Avoid costly denials as you tap into a vast database that explains how to properly code your diagnoses and procedures.
• Protect your practice’s profits by using Kathy’s documentation tips that help you dodge auditing flags.
• Get ready for ICD-10 implementation by using the GI Coding Advisor ICD-10 translator.
To receive a free 14-day subscription and register for the online demo, visit www.gicodingadvisor.org.
Memorial and honorary gifts: A special tribute
Did you know you can honor a family member, friend, or colleague whose life has been touched by GI research through a gift to the AGA Research Foundation? Your gift will honor a loved one or yourself and support the AGA Research Awards Program, while giving you a tax benefit.
• Giving now or later. Any charitable gift can be made in honor or memory of someone.
• A gift today. An outright gift will help fund the AGA Research Awards Program. Your gift will assist in furthering basic digestive disease research which can ultimately advance research into all digestive diseases. The financial benefits include an income tax deduction and possible elimination of capital gains tax. A cash gift of $25,000 or more qualifies for membership in the AGA Legacy Society, which recognizes the foundation’s most generous individual donors.
• A gift through your will or living trust. You can include a bequest in your will or living trust stating that a specific asset, certain dollar amount, or more commonly a percentage of your estate will pass to the AGA Research Foundation at your death in honor of your loved one. A bequest gift of $50,000 or more qualifies for membership in the AGA Legacy Society.
• Named funds. A named fund, which can be named to honor or memorialize a loved one, can be established with a minimum gift of $100,000 over the course of 5 years or through an estate gift. Gifts of cash, appreciated securities, life insurance, or property are gift vehicles that may be used to establish a fund. Donors receive a tax deduction at the time a fund is established and when additional contributions are made to the fund.
Because the principal remains intact, the fund will support our mission in perpetuity. The larger the fund, the more impact it has on the program it is designed to benefit.
Your Next Step
An honorary gift is a wonderful way to acknowledge someone’s vision for the future. To learn more about ways to recognize your honoree, visit our website at www.gastro.org/contribute or contact Stacey Hinton Tuneski at 301-222-4005 or [email protected].
Did you know you can honor a family member, friend, or colleague whose life has been touched by GI research through a gift to the AGA Research Foundation? Your gift will honor a loved one or yourself and support the AGA Research Awards Program, while giving you a tax benefit.
• Giving now or later. Any charitable gift can be made in honor or memory of someone.
• A gift today. An outright gift will help fund the AGA Research Awards Program. Your gift will assist in furthering basic digestive disease research which can ultimately advance research into all digestive diseases. The financial benefits include an income tax deduction and possible elimination of capital gains tax. A cash gift of $25,000 or more qualifies for membership in the AGA Legacy Society, which recognizes the foundation’s most generous individual donors.
• A gift through your will or living trust. You can include a bequest in your will or living trust stating that a specific asset, certain dollar amount, or more commonly a percentage of your estate will pass to the AGA Research Foundation at your death in honor of your loved one. A bequest gift of $50,000 or more qualifies for membership in the AGA Legacy Society.
• Named funds. A named fund, which can be named to honor or memorialize a loved one, can be established with a minimum gift of $100,000 over the course of 5 years or through an estate gift. Gifts of cash, appreciated securities, life insurance, or property are gift vehicles that may be used to establish a fund. Donors receive a tax deduction at the time a fund is established and when additional contributions are made to the fund.
Because the principal remains intact, the fund will support our mission in perpetuity. The larger the fund, the more impact it has on the program it is designed to benefit.
Your Next Step
An honorary gift is a wonderful way to acknowledge someone’s vision for the future. To learn more about ways to recognize your honoree, visit our website at www.gastro.org/contribute or contact Stacey Hinton Tuneski at 301-222-4005 or [email protected].
Did you know you can honor a family member, friend, or colleague whose life has been touched by GI research through a gift to the AGA Research Foundation? Your gift will honor a loved one or yourself and support the AGA Research Awards Program, while giving you a tax benefit.
• Giving now or later. Any charitable gift can be made in honor or memory of someone.
• A gift today. An outright gift will help fund the AGA Research Awards Program. Your gift will assist in furthering basic digestive disease research which can ultimately advance research into all digestive diseases. The financial benefits include an income tax deduction and possible elimination of capital gains tax. A cash gift of $25,000 or more qualifies for membership in the AGA Legacy Society, which recognizes the foundation’s most generous individual donors.
• A gift through your will or living trust. You can include a bequest in your will or living trust stating that a specific asset, certain dollar amount, or more commonly a percentage of your estate will pass to the AGA Research Foundation at your death in honor of your loved one. A bequest gift of $50,000 or more qualifies for membership in the AGA Legacy Society.
• Named funds. A named fund, which can be named to honor or memorialize a loved one, can be established with a minimum gift of $100,000 over the course of 5 years or through an estate gift. Gifts of cash, appreciated securities, life insurance, or property are gift vehicles that may be used to establish a fund. Donors receive a tax deduction at the time a fund is established and when additional contributions are made to the fund.
Because the principal remains intact, the fund will support our mission in perpetuity. The larger the fund, the more impact it has on the program it is designed to benefit.
Your Next Step
An honorary gift is a wonderful way to acknowledge someone’s vision for the future. To learn more about ways to recognize your honoree, visit our website at www.gastro.org/contribute or contact Stacey Hinton Tuneski at 301-222-4005 or [email protected].
Ensure GI services are fairly and accurately valued
The GI community needs your help.
In a request from the Centers for Medicare and Medicaid Services to review physician services, the American Medical Association’s (AMA’s) Relative Value Update Committee (RUC) is performing a comprehensive review of the valuation of physician work and practice expense for more than 100 endoscopy procedures. To provide fair and accurate valuations, the American Gastroenterological Association and the American Society for Gastrointestinal Endoscopy are asking physicians to complete surveys of these endoscopic procedures.
These surveys will directly impact how codes and procedures are "valued" and ultimately reimbursed. The GI community needs significant participation from gastroenterologists to complete these surveys of physician work in order to provide realistic recommendations to the AMA’s RUC. If a minimum number of responses for each survey are not obtained, GIs are at risk that the Centers for Medicare and Medicaid Services will make final decisions regarding valuation without consideration of our input.
How can you help?
If you are selected for a survey, please participate. It is critical that you respond if you are selected to participate in any survey. Do not forfeit this important opportunity to provide input into the valuation of GI services. Whether you perform routine esophagogastroduodenoscopy (EGD) and colonoscopy, or more complex procedures such as endoscopic retrograde cholangiopancreatography (ERCP), endoscopic ultrasound or stents, your help is needed.
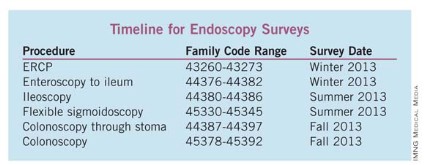
On the following page is a list of upcoming surveys. We also urge you to add your name to the roster of individuals to be contacted to complete the RUC surveys of physician work for endoscopic procedures shown in the timeline.
To volunteer, e-mail [email protected] with your name and contact information. Based on the list of surveys in the timeline, please specify which procedure surveys apply to you. Additional information will be provided prior to the start of each survey. To learn more, visit www.gastro.org/RUC or contact Leslie D. Narramore of the AGA ([email protected]; 410-349-7455) or Sam Reynolds of the ASGE ([email protected]; 630-570-5643).
Sincerely,
Loren Laine, M.D., AGAF President, The AGA Institute
Thomas M. Deas Jr., M.D., MMM, FASGE President, American Society for Gastrointestinal Endoscopy
The GI community needs your help.
In a request from the Centers for Medicare and Medicaid Services to review physician services, the American Medical Association’s (AMA’s) Relative Value Update Committee (RUC) is performing a comprehensive review of the valuation of physician work and practice expense for more than 100 endoscopy procedures. To provide fair and accurate valuations, the American Gastroenterological Association and the American Society for Gastrointestinal Endoscopy are asking physicians to complete surveys of these endoscopic procedures.
These surveys will directly impact how codes and procedures are "valued" and ultimately reimbursed. The GI community needs significant participation from gastroenterologists to complete these surveys of physician work in order to provide realistic recommendations to the AMA’s RUC. If a minimum number of responses for each survey are not obtained, GIs are at risk that the Centers for Medicare and Medicaid Services will make final decisions regarding valuation without consideration of our input.
How can you help?
If you are selected for a survey, please participate. It is critical that you respond if you are selected to participate in any survey. Do not forfeit this important opportunity to provide input into the valuation of GI services. Whether you perform routine esophagogastroduodenoscopy (EGD) and colonoscopy, or more complex procedures such as endoscopic retrograde cholangiopancreatography (ERCP), endoscopic ultrasound or stents, your help is needed.
On the following page is a list of upcoming surveys. We also urge you to add your name to the roster of individuals to be contacted to complete the RUC surveys of physician work for endoscopic procedures shown in the timeline.
To volunteer, e-mail [email protected] with your name and contact information. Based on the list of surveys in the timeline, please specify which procedure surveys apply to you. Additional information will be provided prior to the start of each survey. To learn more, visit www.gastro.org/RUC or contact Leslie D. Narramore of the AGA ([email protected]; 410-349-7455) or Sam Reynolds of the ASGE ([email protected]; 630-570-5643).
Sincerely,
Loren Laine, M.D., AGAF President, The AGA Institute
Thomas M. Deas Jr., M.D., MMM, FASGE President, American Society for Gastrointestinal Endoscopy
The GI community needs your help.
In a request from the Centers for Medicare and Medicaid Services to review physician services, the American Medical Association’s (AMA’s) Relative Value Update Committee (RUC) is performing a comprehensive review of the valuation of physician work and practice expense for more than 100 endoscopy procedures. To provide fair and accurate valuations, the American Gastroenterological Association and the American Society for Gastrointestinal Endoscopy are asking physicians to complete surveys of these endoscopic procedures.
These surveys will directly impact how codes and procedures are "valued" and ultimately reimbursed. The GI community needs significant participation from gastroenterologists to complete these surveys of physician work in order to provide realistic recommendations to the AMA’s RUC. If a minimum number of responses for each survey are not obtained, GIs are at risk that the Centers for Medicare and Medicaid Services will make final decisions regarding valuation without consideration of our input.
How can you help?
If you are selected for a survey, please participate. It is critical that you respond if you are selected to participate in any survey. Do not forfeit this important opportunity to provide input into the valuation of GI services. Whether you perform routine esophagogastroduodenoscopy (EGD) and colonoscopy, or more complex procedures such as endoscopic retrograde cholangiopancreatography (ERCP), endoscopic ultrasound or stents, your help is needed.
On the following page is a list of upcoming surveys. We also urge you to add your name to the roster of individuals to be contacted to complete the RUC surveys of physician work for endoscopic procedures shown in the timeline.
To volunteer, e-mail [email protected] with your name and contact information. Based on the list of surveys in the timeline, please specify which procedure surveys apply to you. Additional information will be provided prior to the start of each survey. To learn more, visit www.gastro.org/RUC or contact Leslie D. Narramore of the AGA ([email protected]; 410-349-7455) or Sam Reynolds of the ASGE ([email protected]; 630-570-5643).
Sincerely,
Loren Laine, M.D., AGAF President, The AGA Institute
Thomas M. Deas Jr., M.D., MMM, FASGE President, American Society for Gastrointestinal Endoscopy
AGA Tech Summit focuses on advancing innovation in GI
Register today for the fourth annual AGA Tech Summit, which will be held March 15-16, 2013, at the Four Seasons Hotel Silicon Valley in East Palo Alto, Calif.
The summit addresses critical elements that affect the likelihood of success in developing and obtaining adoption and coverage of new medical technologies in today’s GI market. Sessions explore the latest developments in reimbursement, the regulatory and legal environment, intellectual property, and more. You will hear directly from the Food and Drug Administration, private payers, and medical device executives, who will address specific requirements. Areas of unmet need and clinical and research opportunities will be discussed in areas such as the intestinal microbiome, inflammatory bowel disease, pancreatitis, and the liver.
You will also learn about what the AGA is doing to help create a more supportive environment for new technologies, like the AGA Technology Center and AGA initiatives that support innovation in digestive and metabolic diseases.
A new addition to the program, the physician innovator track, offers GI MD inventors beneficial information on how to obtain funding for their idea. This specialized track also gives insight on what investors are looking for, how to protect intellectual property, and how physician-investors can be successful in academic, private practice, or industry settings.
To register and view the program, visit www.gastro.org/tech-summit.
The AGA Institute and Kleiner Perkins Caufield & Byers gratefully acknowledge our corporate sponsors.
medical technologies, reimbursement, regulatory, legal environment, intellectual property, the Food and Drug Administration, private payers, medical device executives, requirements, clinical, research, intestinal microbiome, inflammatory bowel disease, pancreatitis, liver.
Register today for the fourth annual AGA Tech Summit, which will be held March 15-16, 2013, at the Four Seasons Hotel Silicon Valley in East Palo Alto, Calif.
The summit addresses critical elements that affect the likelihood of success in developing and obtaining adoption and coverage of new medical technologies in today’s GI market. Sessions explore the latest developments in reimbursement, the regulatory and legal environment, intellectual property, and more. You will hear directly from the Food and Drug Administration, private payers, and medical device executives, who will address specific requirements. Areas of unmet need and clinical and research opportunities will be discussed in areas such as the intestinal microbiome, inflammatory bowel disease, pancreatitis, and the liver.
You will also learn about what the AGA is doing to help create a more supportive environment for new technologies, like the AGA Technology Center and AGA initiatives that support innovation in digestive and metabolic diseases.
A new addition to the program, the physician innovator track, offers GI MD inventors beneficial information on how to obtain funding for their idea. This specialized track also gives insight on what investors are looking for, how to protect intellectual property, and how physician-investors can be successful in academic, private practice, or industry settings.
To register and view the program, visit www.gastro.org/tech-summit.
The AGA Institute and Kleiner Perkins Caufield & Byers gratefully acknowledge our corporate sponsors.
Register today for the fourth annual AGA Tech Summit, which will be held March 15-16, 2013, at the Four Seasons Hotel Silicon Valley in East Palo Alto, Calif.
The summit addresses critical elements that affect the likelihood of success in developing and obtaining adoption and coverage of new medical technologies in today’s GI market. Sessions explore the latest developments in reimbursement, the regulatory and legal environment, intellectual property, and more. You will hear directly from the Food and Drug Administration, private payers, and medical device executives, who will address specific requirements. Areas of unmet need and clinical and research opportunities will be discussed in areas such as the intestinal microbiome, inflammatory bowel disease, pancreatitis, and the liver.
You will also learn about what the AGA is doing to help create a more supportive environment for new technologies, like the AGA Technology Center and AGA initiatives that support innovation in digestive and metabolic diseases.
A new addition to the program, the physician innovator track, offers GI MD inventors beneficial information on how to obtain funding for their idea. This specialized track also gives insight on what investors are looking for, how to protect intellectual property, and how physician-investors can be successful in academic, private practice, or industry settings.
To register and view the program, visit www.gastro.org/tech-summit.
The AGA Institute and Kleiner Perkins Caufield & Byers gratefully acknowledge our corporate sponsors.
medical technologies, reimbursement, regulatory, legal environment, intellectual property, the Food and Drug Administration, private payers, medical device executives, requirements, clinical, research, intestinal microbiome, inflammatory bowel disease, pancreatitis, liver.
medical technologies, reimbursement, regulatory, legal environment, intellectual property, the Food and Drug Administration, private payers, medical device executives, requirements, clinical, research, intestinal microbiome, inflammatory bowel disease, pancreatitis, liver.
AGA remembers former Japanese society president
Kenji Fujiwara, M.D., Ph.D., former president of the Japanese Society of Gastroenterology (JSGE) and the honorary director of Yokohama Rosai Hospital, passed away at the age of 74 on Nov. 4, 2012. He graduated from the faculty of medicine, University of Tokyo, in 1964 and received a doctoral degree in 1970. As assistant professor in internal medicine at the University of Tokyo (1986-1994) and professor and chair of internal medicine at Saitama (Japan) Medical School (1994-2005), he published more than 250 original articles in English, including seminal studies on viral and nonviral fulminant hepatitis.
During his tenure as president of JSGE (2001-2004), he contributed significantly to the society’s management and foreign relations. In particular, his leadership solidified the AGA-JSGE relationship, which resulted in annual joint symposia. Dr. Fujiwara was appointed by the Japanese Ministry of Health, Labor, and Welfare (JMHLW) to chair the Council of Inspection for Cadaveric Organ Transplantation and Committee for Evaluation of Recipient of Cadaveric Liver Transplantation. In these roles, he made historic contributions to the establishment of national consensus and guidelines for cadaveric liver transplantation, which led to the dawn of a new era for orthotopic liver transplantation in Japan.
In recent years, Dr. Fujiwara worked tirelessly to establish a revised international classification of diseases system for the World Health Organization and for JMHLW. He is survived by his wife Mariko, three sons, a daughter, and 10 grandchildren.
Kenji Fujiwara, M.D., Ph.D., former president of the Japanese Society of Gastroenterology (JSGE) and the honorary director of Yokohama Rosai Hospital, passed away at the age of 74 on Nov. 4, 2012. He graduated from the faculty of medicine, University of Tokyo, in 1964 and received a doctoral degree in 1970. As assistant professor in internal medicine at the University of Tokyo (1986-1994) and professor and chair of internal medicine at Saitama (Japan) Medical School (1994-2005), he published more than 250 original articles in English, including seminal studies on viral and nonviral fulminant hepatitis.
During his tenure as president of JSGE (2001-2004), he contributed significantly to the society’s management and foreign relations. In particular, his leadership solidified the AGA-JSGE relationship, which resulted in annual joint symposia. Dr. Fujiwara was appointed by the Japanese Ministry of Health, Labor, and Welfare (JMHLW) to chair the Council of Inspection for Cadaveric Organ Transplantation and Committee for Evaluation of Recipient of Cadaveric Liver Transplantation. In these roles, he made historic contributions to the establishment of national consensus and guidelines for cadaveric liver transplantation, which led to the dawn of a new era for orthotopic liver transplantation in Japan.
In recent years, Dr. Fujiwara worked tirelessly to establish a revised international classification of diseases system for the World Health Organization and for JMHLW. He is survived by his wife Mariko, three sons, a daughter, and 10 grandchildren.
Kenji Fujiwara, M.D., Ph.D., former president of the Japanese Society of Gastroenterology (JSGE) and the honorary director of Yokohama Rosai Hospital, passed away at the age of 74 on Nov. 4, 2012. He graduated from the faculty of medicine, University of Tokyo, in 1964 and received a doctoral degree in 1970. As assistant professor in internal medicine at the University of Tokyo (1986-1994) and professor and chair of internal medicine at Saitama (Japan) Medical School (1994-2005), he published more than 250 original articles in English, including seminal studies on viral and nonviral fulminant hepatitis.
During his tenure as president of JSGE (2001-2004), he contributed significantly to the society’s management and foreign relations. In particular, his leadership solidified the AGA-JSGE relationship, which resulted in annual joint symposia. Dr. Fujiwara was appointed by the Japanese Ministry of Health, Labor, and Welfare (JMHLW) to chair the Council of Inspection for Cadaveric Organ Transplantation and Committee for Evaluation of Recipient of Cadaveric Liver Transplantation. In these roles, he made historic contributions to the establishment of national consensus and guidelines for cadaveric liver transplantation, which led to the dawn of a new era for orthotopic liver transplantation in Japan.
In recent years, Dr. Fujiwara worked tirelessly to establish a revised international classification of diseases system for the World Health Organization and for JMHLW. He is survived by his wife Mariko, three sons, a daughter, and 10 grandchildren.
Trainee and young GI track returns to DDW 2013
At Digestive Disease Week (DDW) 2013, the American Gastroenterological Association will again offer special sessions designed to meet the unique needs of physicians who are new to the field. Participants will learn about all aspects of starting a career in clinical practice or research, have the opportunity to network with mentors and peers, and review board material. Save the date for these trainee track sessions:
• AGA Spring Postgraduate Course: Putting Patients First: Actionable Evidence for Clinical Practice, Saturday, May 18, and Sunday, May 19. This clinically focused course offers you immediately applicable information. Trainees may register at a reduced registration fee.
• Mentor and Advisor Program: A Networking Event for Trainees, Saturday, May 18. Meet your peers and more established colleagues who serve as mentors, while enjoying refreshments.
• Board Review Session, Monday, May 20. Designed around content from the new Digestive Diseases Self-Education Program (DDSEP) 7, this session serves as a primer for third-year fellows preparing for the board exam, and as a review course for others. Discount coupons for DDSEP 7 will be available to attendees.
• Career and Professional Related Issues, Monday, May 20. Get advice on common career issues such as choosing a practice type and location, developing your CV, interviewing, negotiating contracts, and work-life balance.
• Maximizing Opportunities Within Fellowship Training: Advice From Fellows and Faculty, Monday, May 20. Learn how to create a niche within GI, discuss career options, and explore advanced clinical training opportunities.
With the exception of the Spring Postgraduate Course, these sessions are free, but you must be registered for DDW to attend. DDW registration opens Jan. 9 for AGA members. AGA trainee members can attend for free if they register by April 10. More information is available on the DDW website.
At Digestive Disease Week (DDW) 2013, the American Gastroenterological Association will again offer special sessions designed to meet the unique needs of physicians who are new to the field. Participants will learn about all aspects of starting a career in clinical practice or research, have the opportunity to network with mentors and peers, and review board material. Save the date for these trainee track sessions:
• AGA Spring Postgraduate Course: Putting Patients First: Actionable Evidence for Clinical Practice, Saturday, May 18, and Sunday, May 19. This clinically focused course offers you immediately applicable information. Trainees may register at a reduced registration fee.
• Mentor and Advisor Program: A Networking Event for Trainees, Saturday, May 18. Meet your peers and more established colleagues who serve as mentors, while enjoying refreshments.
• Board Review Session, Monday, May 20. Designed around content from the new Digestive Diseases Self-Education Program (DDSEP) 7, this session serves as a primer for third-year fellows preparing for the board exam, and as a review course for others. Discount coupons for DDSEP 7 will be available to attendees.
• Career and Professional Related Issues, Monday, May 20. Get advice on common career issues such as choosing a practice type and location, developing your CV, interviewing, negotiating contracts, and work-life balance.
• Maximizing Opportunities Within Fellowship Training: Advice From Fellows and Faculty, Monday, May 20. Learn how to create a niche within GI, discuss career options, and explore advanced clinical training opportunities.
With the exception of the Spring Postgraduate Course, these sessions are free, but you must be registered for DDW to attend. DDW registration opens Jan. 9 for AGA members. AGA trainee members can attend for free if they register by April 10. More information is available on the DDW website.
At Digestive Disease Week (DDW) 2013, the American Gastroenterological Association will again offer special sessions designed to meet the unique needs of physicians who are new to the field. Participants will learn about all aspects of starting a career in clinical practice or research, have the opportunity to network with mentors and peers, and review board material. Save the date for these trainee track sessions:
• AGA Spring Postgraduate Course: Putting Patients First: Actionable Evidence for Clinical Practice, Saturday, May 18, and Sunday, May 19. This clinically focused course offers you immediately applicable information. Trainees may register at a reduced registration fee.
• Mentor and Advisor Program: A Networking Event for Trainees, Saturday, May 18. Meet your peers and more established colleagues who serve as mentors, while enjoying refreshments.
• Board Review Session, Monday, May 20. Designed around content from the new Digestive Diseases Self-Education Program (DDSEP) 7, this session serves as a primer for third-year fellows preparing for the board exam, and as a review course for others. Discount coupons for DDSEP 7 will be available to attendees.
• Career and Professional Related Issues, Monday, May 20. Get advice on common career issues such as choosing a practice type and location, developing your CV, interviewing, negotiating contracts, and work-life balance.
• Maximizing Opportunities Within Fellowship Training: Advice From Fellows and Faculty, Monday, May 20. Learn how to create a niche within GI, discuss career options, and explore advanced clinical training opportunities.
With the exception of the Spring Postgraduate Course, these sessions are free, but you must be registered for DDW to attend. DDW registration opens Jan. 9 for AGA members. AGA trainee members can attend for free if they register by April 10. More information is available on the DDW website.