User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Real-World Study Confirms Short-Term Efficacy of Guselkumab in PsA
Key clinical point: Guselkumab treatment for 4 months was effective in patients with psoriatic arthritis (PsA) who had inflammatory back pain associated with suggestive axial involvement assessed by MRI or X-ray.
Major finding: At 4 months, the Bath Ankylosing Spondylitis Disease Activity Index scores reduced significantly in patients receiving guselkumab (mean difference, −2.11 ± 0.43; P < .05), with nearly half of the patients achieving a BASDAI score ≤ 4. Similar outcomes were observed in terms of Ankylosing Spondylitis Disease Activity Scores (P = .021) and Disease Activity in PsA scores (P = .001).
Study details: Findings are from a real-life, multicenter study including 67 patients with PsA and suggestive features of axial involvement as shown by x-ray or MRI who were treated with guselkumab for ≥ 4 months.
Disclosures: This study did not receive any funding. The authors declared no conflict of interests.
Source: Ruscitti P, Pantano I, Cataldi G, et al. Short-term effectiveness of guselkumab in psoriatic arthritis patients and suggestive features of axial involvement: Results from a real-life multicentre cohort. Rheumatology (Oxford). Published online April 10, 2024. Source
Key clinical point: Guselkumab treatment for 4 months was effective in patients with psoriatic arthritis (PsA) who had inflammatory back pain associated with suggestive axial involvement assessed by MRI or X-ray.
Major finding: At 4 months, the Bath Ankylosing Spondylitis Disease Activity Index scores reduced significantly in patients receiving guselkumab (mean difference, −2.11 ± 0.43; P < .05), with nearly half of the patients achieving a BASDAI score ≤ 4. Similar outcomes were observed in terms of Ankylosing Spondylitis Disease Activity Scores (P = .021) and Disease Activity in PsA scores (P = .001).
Study details: Findings are from a real-life, multicenter study including 67 patients with PsA and suggestive features of axial involvement as shown by x-ray or MRI who were treated with guselkumab for ≥ 4 months.
Disclosures: This study did not receive any funding. The authors declared no conflict of interests.
Source: Ruscitti P, Pantano I, Cataldi G, et al. Short-term effectiveness of guselkumab in psoriatic arthritis patients and suggestive features of axial involvement: Results from a real-life multicentre cohort. Rheumatology (Oxford). Published online April 10, 2024. Source
Key clinical point: Guselkumab treatment for 4 months was effective in patients with psoriatic arthritis (PsA) who had inflammatory back pain associated with suggestive axial involvement assessed by MRI or X-ray.
Major finding: At 4 months, the Bath Ankylosing Spondylitis Disease Activity Index scores reduced significantly in patients receiving guselkumab (mean difference, −2.11 ± 0.43; P < .05), with nearly half of the patients achieving a BASDAI score ≤ 4. Similar outcomes were observed in terms of Ankylosing Spondylitis Disease Activity Scores (P = .021) and Disease Activity in PsA scores (P = .001).
Study details: Findings are from a real-life, multicenter study including 67 patients with PsA and suggestive features of axial involvement as shown by x-ray or MRI who were treated with guselkumab for ≥ 4 months.
Disclosures: This study did not receive any funding. The authors declared no conflict of interests.
Source: Ruscitti P, Pantano I, Cataldi G, et al. Short-term effectiveness of guselkumab in psoriatic arthritis patients and suggestive features of axial involvement: Results from a real-life multicentre cohort. Rheumatology (Oxford). Published online April 10, 2024. Source
Study Identifies Risk Factors for Development of Psoriasis To Clinical PsA
Key clinical point: Risk factors for psoriasis developing into clinical psoriatic arthritis (PsA) include higher age, nail involvement, increased erythrocyte sedimentation rate (ESR), and elevated high-sensitivity C-reactive protein (hs-CRP) levels.
Major finding: Age ≥ 40 years (odds ratio [OR], 1.04; P < .01), nail involvement (OR, 1.17; P < .01), increased ESR (OR, 1.03; P < .05), and elevated hs-CRP levels (OR, 1.31; P < .01) increased the risk for incident PsA in patients with psoriasis. Moreover, MRI-detected enthesitis and tenosynovitis combined with these risk factors vs the risk factors alone showed better specificity (94.3% vs 69.0%) and similar sensitivity (89.0% vs 84.6%) in diagnosing PsA.
Study details: This longitudinal case-control study included 75 patients diagnosed with clinical PsA who were compared with 345 patients with psoriasis and without PsA, all of whom were aged 18-65 years.
Disclosures: This study was supported by the Beijing Municipal Science and Technology Project. The authors declared no conflict of interests.
Source: Yao A, Wang L, Qi F, et al. Risk factors and early detection of joint damage in patients with psoriasis: A case–control study. Int J Dermatol. Published online April 29, 2024. Source
Key clinical point: Risk factors for psoriasis developing into clinical psoriatic arthritis (PsA) include higher age, nail involvement, increased erythrocyte sedimentation rate (ESR), and elevated high-sensitivity C-reactive protein (hs-CRP) levels.
Major finding: Age ≥ 40 years (odds ratio [OR], 1.04; P < .01), nail involvement (OR, 1.17; P < .01), increased ESR (OR, 1.03; P < .05), and elevated hs-CRP levels (OR, 1.31; P < .01) increased the risk for incident PsA in patients with psoriasis. Moreover, MRI-detected enthesitis and tenosynovitis combined with these risk factors vs the risk factors alone showed better specificity (94.3% vs 69.0%) and similar sensitivity (89.0% vs 84.6%) in diagnosing PsA.
Study details: This longitudinal case-control study included 75 patients diagnosed with clinical PsA who were compared with 345 patients with psoriasis and without PsA, all of whom were aged 18-65 years.
Disclosures: This study was supported by the Beijing Municipal Science and Technology Project. The authors declared no conflict of interests.
Source: Yao A, Wang L, Qi F, et al. Risk factors and early detection of joint damage in patients with psoriasis: A case–control study. Int J Dermatol. Published online April 29, 2024. Source
Key clinical point: Risk factors for psoriasis developing into clinical psoriatic arthritis (PsA) include higher age, nail involvement, increased erythrocyte sedimentation rate (ESR), and elevated high-sensitivity C-reactive protein (hs-CRP) levels.
Major finding: Age ≥ 40 years (odds ratio [OR], 1.04; P < .01), nail involvement (OR, 1.17; P < .01), increased ESR (OR, 1.03; P < .05), and elevated hs-CRP levels (OR, 1.31; P < .01) increased the risk for incident PsA in patients with psoriasis. Moreover, MRI-detected enthesitis and tenosynovitis combined with these risk factors vs the risk factors alone showed better specificity (94.3% vs 69.0%) and similar sensitivity (89.0% vs 84.6%) in diagnosing PsA.
Study details: This longitudinal case-control study included 75 patients diagnosed with clinical PsA who were compared with 345 patients with psoriasis and without PsA, all of whom were aged 18-65 years.
Disclosures: This study was supported by the Beijing Municipal Science and Technology Project. The authors declared no conflict of interests.
Source: Yao A, Wang L, Qi F, et al. Risk factors and early detection of joint damage in patients with psoriasis: A case–control study. Int J Dermatol. Published online April 29, 2024. Source
Elevated Risk for PsA in Patients With Psoriasis and Arthralgia
Key clinical point: Patients with psoriasis who had subclinical psoriatic arthritis (PsA) based on the presence of arthralgia had a higher likelihood of developing PsA than those with only psoriasis.
Major finding: At a mean follow-up of 33 months, 80.9% of patients had subclinical PsA, with the incidence rate of new-onset PsA being 7.7 per 100 patients-years in this group and the most predominant presentation being peripheral arthritis (82.1%). The risk of developing PsA was significantly higher in patients with subclinical PsA vs psoriasis (hazard ratio, 11.7; P = .016).
Study details: This study included 384 patients with psoriasis from two European cohorts of whom 56 patients developed new-onset PsA.
Disclosures: This study did not disclose any specific funding. Six authors declared being consultants for or having other ties with various sources. The other authors declared no conflict of interests.
Source: Zabotti A, Fagni F, Gossec L, et al. Risk of developing psoriatic arthritis in psoriasis cohorts with arthralgia: Exploring the subclinical psoriatic arthritis stage. RMD Open. 2024;10:e004314. Source
Key clinical point: Patients with psoriasis who had subclinical psoriatic arthritis (PsA) based on the presence of arthralgia had a higher likelihood of developing PsA than those with only psoriasis.
Major finding: At a mean follow-up of 33 months, 80.9% of patients had subclinical PsA, with the incidence rate of new-onset PsA being 7.7 per 100 patients-years in this group and the most predominant presentation being peripheral arthritis (82.1%). The risk of developing PsA was significantly higher in patients with subclinical PsA vs psoriasis (hazard ratio, 11.7; P = .016).
Study details: This study included 384 patients with psoriasis from two European cohorts of whom 56 patients developed new-onset PsA.
Disclosures: This study did not disclose any specific funding. Six authors declared being consultants for or having other ties with various sources. The other authors declared no conflict of interests.
Source: Zabotti A, Fagni F, Gossec L, et al. Risk of developing psoriatic arthritis in psoriasis cohorts with arthralgia: Exploring the subclinical psoriatic arthritis stage. RMD Open. 2024;10:e004314. Source
Key clinical point: Patients with psoriasis who had subclinical psoriatic arthritis (PsA) based on the presence of arthralgia had a higher likelihood of developing PsA than those with only psoriasis.
Major finding: At a mean follow-up of 33 months, 80.9% of patients had subclinical PsA, with the incidence rate of new-onset PsA being 7.7 per 100 patients-years in this group and the most predominant presentation being peripheral arthritis (82.1%). The risk of developing PsA was significantly higher in patients with subclinical PsA vs psoriasis (hazard ratio, 11.7; P = .016).
Study details: This study included 384 patients with psoriasis from two European cohorts of whom 56 patients developed new-onset PsA.
Disclosures: This study did not disclose any specific funding. Six authors declared being consultants for or having other ties with various sources. The other authors declared no conflict of interests.
Source: Zabotti A, Fagni F, Gossec L, et al. Risk of developing psoriatic arthritis in psoriasis cohorts with arthralgia: Exploring the subclinical psoriatic arthritis stage. RMD Open. 2024;10:e004314. Source
Risankizumab Shows Long-Term Efficacy in PsA Regardless of Baseline Characteristics
Key clinical point: Risankizumab showed consistent long-term efficacy in patients with active psoriatic arthritis (PsA) regardless of baseline demographic, disease characteristics, and prior medication use.
Major finding: At week 24, a higher number of patients receiving risankizumab vs placebo achieved ≥ 20% improvement in the American College of Rheumatology response (ACR20; 46.3%-60.1% vs 15.5%-36.2%) despite varying demographic and disease characteristics. At week 52, similar proportions of patients who were randomized to risankizumab (48.6%-75.8%) or who switched from placebo to risankizumab (43.7%-63.9%) achieved ACR20.
Study details: This post hoc integrated analysis of the phase 3 KEEPsAKE 1 and KEEPsAKE 2 trials included 1235 patients with active PsA and prior inadequate response or intolerance to conventional synthetic or biologic disease-modifying antirheumatic drugs who received risankizumab or placebo with crossover to risankizumab at week 24.
Disclosures: This study was funded by AbbVie. Four authors declared being employees of and holding stocks or stock options with AbbVie. The other authors declared having ties with AbbVie and various sources.
Source: Merola JF, Armstrong A, Khattri S, et al. Efficacy of risankizumab across subgroups in patients with active psoriatic arthritis: A post hoc integrated analysis of the phase 3 KEEPsAKE 1 and KEEPsAKE 2 randomized controlled trials. J Dermatolog Treat. 2024;35:2342383. Source
Key clinical point: Risankizumab showed consistent long-term efficacy in patients with active psoriatic arthritis (PsA) regardless of baseline demographic, disease characteristics, and prior medication use.
Major finding: At week 24, a higher number of patients receiving risankizumab vs placebo achieved ≥ 20% improvement in the American College of Rheumatology response (ACR20; 46.3%-60.1% vs 15.5%-36.2%) despite varying demographic and disease characteristics. At week 52, similar proportions of patients who were randomized to risankizumab (48.6%-75.8%) or who switched from placebo to risankizumab (43.7%-63.9%) achieved ACR20.
Study details: This post hoc integrated analysis of the phase 3 KEEPsAKE 1 and KEEPsAKE 2 trials included 1235 patients with active PsA and prior inadequate response or intolerance to conventional synthetic or biologic disease-modifying antirheumatic drugs who received risankizumab or placebo with crossover to risankizumab at week 24.
Disclosures: This study was funded by AbbVie. Four authors declared being employees of and holding stocks or stock options with AbbVie. The other authors declared having ties with AbbVie and various sources.
Source: Merola JF, Armstrong A, Khattri S, et al. Efficacy of risankizumab across subgroups in patients with active psoriatic arthritis: A post hoc integrated analysis of the phase 3 KEEPsAKE 1 and KEEPsAKE 2 randomized controlled trials. J Dermatolog Treat. 2024;35:2342383. Source
Key clinical point: Risankizumab showed consistent long-term efficacy in patients with active psoriatic arthritis (PsA) regardless of baseline demographic, disease characteristics, and prior medication use.
Major finding: At week 24, a higher number of patients receiving risankizumab vs placebo achieved ≥ 20% improvement in the American College of Rheumatology response (ACR20; 46.3%-60.1% vs 15.5%-36.2%) despite varying demographic and disease characteristics. At week 52, similar proportions of patients who were randomized to risankizumab (48.6%-75.8%) or who switched from placebo to risankizumab (43.7%-63.9%) achieved ACR20.
Study details: This post hoc integrated analysis of the phase 3 KEEPsAKE 1 and KEEPsAKE 2 trials included 1235 patients with active PsA and prior inadequate response or intolerance to conventional synthetic or biologic disease-modifying antirheumatic drugs who received risankizumab or placebo with crossover to risankizumab at week 24.
Disclosures: This study was funded by AbbVie. Four authors declared being employees of and holding stocks or stock options with AbbVie. The other authors declared having ties with AbbVie and various sources.
Source: Merola JF, Armstrong A, Khattri S, et al. Efficacy of risankizumab across subgroups in patients with active psoriatic arthritis: A post hoc integrated analysis of the phase 3 KEEPsAKE 1 and KEEPsAKE 2 randomized controlled trials. J Dermatolog Treat. 2024;35:2342383. Source
Secukinumab Safe and Effective in Challenging-to-Treat Patients With PsA
Key clinical point: Secukinumab demonstrated rapid improvements in clinical outcomes and showed a consistent safety profile in challenging-to-treat patients with psoriatic arthritis (PsA) who had high BMI and showed higher tender and swollen joints despite previous treatment.
Major finding: At week 16, patients receiving secukinumab 300 mg and 150 mg with loading dose vs placebo had a higher rate of achieving the American College of Rheumatology 20 response (59.7% and 43.4%, respectively, vs 15.6%; both P < .0001) and the Psoriasis Area and Severity Index 90 response (47.1% and 22.2%, respectively, vs 5.3%; both P < .05).
Study details: This subgroup analysis of four phase 3 FUTURE studies included 279 patients with challenging-to-treat PsA from the United States who received secukinumab or placebo and had undergone prior treatment with nonsteroidal anti-inflammatory drugs, disease-modifying antirheumatic drugs, or corticosteroids.
Disclosures: This study was supported by Novartis Pharmaceuticals Corporation, United States. Luminita Pricop declared being an employee and stockholder of Novartis. The other authors declared receiving research grants from or having other ties with Novartis or others.
Source: Kivitz AJ, Kremer JM, Legerton CW 3rd, Pricop L, Singhal A. Efficacy and safety of secukinumab in US patients with psoriatic arthritis: A subgroup analysis of the phase 3 FUTURE studies. Rheumatol Ther. Published online April 16, 2024. Source
Key clinical point: Secukinumab demonstrated rapid improvements in clinical outcomes and showed a consistent safety profile in challenging-to-treat patients with psoriatic arthritis (PsA) who had high BMI and showed higher tender and swollen joints despite previous treatment.
Major finding: At week 16, patients receiving secukinumab 300 mg and 150 mg with loading dose vs placebo had a higher rate of achieving the American College of Rheumatology 20 response (59.7% and 43.4%, respectively, vs 15.6%; both P < .0001) and the Psoriasis Area and Severity Index 90 response (47.1% and 22.2%, respectively, vs 5.3%; both P < .05).
Study details: This subgroup analysis of four phase 3 FUTURE studies included 279 patients with challenging-to-treat PsA from the United States who received secukinumab or placebo and had undergone prior treatment with nonsteroidal anti-inflammatory drugs, disease-modifying antirheumatic drugs, or corticosteroids.
Disclosures: This study was supported by Novartis Pharmaceuticals Corporation, United States. Luminita Pricop declared being an employee and stockholder of Novartis. The other authors declared receiving research grants from or having other ties with Novartis or others.
Source: Kivitz AJ, Kremer JM, Legerton CW 3rd, Pricop L, Singhal A. Efficacy and safety of secukinumab in US patients with psoriatic arthritis: A subgroup analysis of the phase 3 FUTURE studies. Rheumatol Ther. Published online April 16, 2024. Source
Key clinical point: Secukinumab demonstrated rapid improvements in clinical outcomes and showed a consistent safety profile in challenging-to-treat patients with psoriatic arthritis (PsA) who had high BMI and showed higher tender and swollen joints despite previous treatment.
Major finding: At week 16, patients receiving secukinumab 300 mg and 150 mg with loading dose vs placebo had a higher rate of achieving the American College of Rheumatology 20 response (59.7% and 43.4%, respectively, vs 15.6%; both P < .0001) and the Psoriasis Area and Severity Index 90 response (47.1% and 22.2%, respectively, vs 5.3%; both P < .05).
Study details: This subgroup analysis of four phase 3 FUTURE studies included 279 patients with challenging-to-treat PsA from the United States who received secukinumab or placebo and had undergone prior treatment with nonsteroidal anti-inflammatory drugs, disease-modifying antirheumatic drugs, or corticosteroids.
Disclosures: This study was supported by Novartis Pharmaceuticals Corporation, United States. Luminita Pricop declared being an employee and stockholder of Novartis. The other authors declared receiving research grants from or having other ties with Novartis or others.
Source: Kivitz AJ, Kremer JM, Legerton CW 3rd, Pricop L, Singhal A. Efficacy and safety of secukinumab in US patients with psoriatic arthritis: A subgroup analysis of the phase 3 FUTURE studies. Rheumatol Ther. Published online April 16, 2024. Source
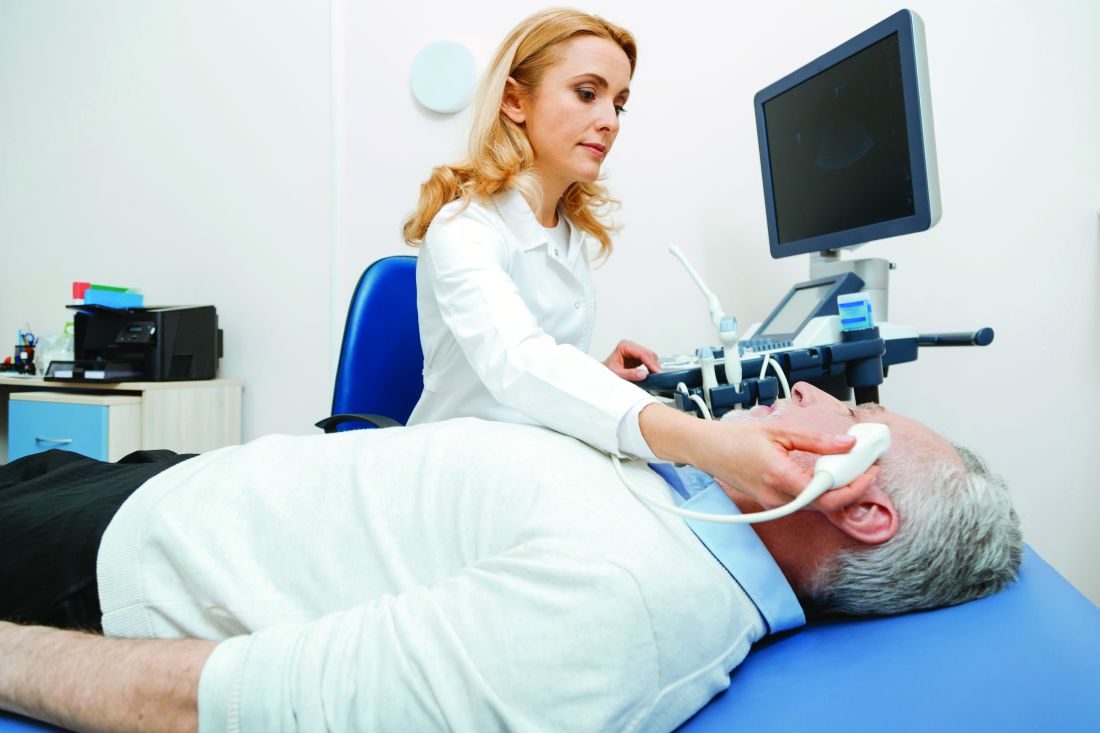
Diagnosing Giant Cell Arteritis Using Ultrasound First Proves Accurate, Avoids Biopsy in Many Cases
Temporal artery ultrasound alone was sufficient to accurately diagnose giant cell arteritis (GCA) in over half of patients in a new prospective study.
The findings provide further evidence that “[ultrasound] of temporal arteries could really take the place of traditional temporal artery biopsy (TAB)” in patients with high clinical suspicion of GCA, lead author Guillaume Denis, MD, of the Centre Hospitalier de Rochefort in Rochefort, France, told this news organization.
The European Alliance of Associations for Rheumatology (EULAR) already recommends ultrasound as a first-line diagnostic tool for patients with suspected large vessel vasculitis, and the 2022 American College of Rheumatology (ACR)/EULAR classification criteria for GCA weighs positive TAB or temporal artery halo sign on ultrasound equally.
Guidelines from the ACR and the Vasculitis Foundation still recommend TAB over ultrasound.
“In general, rheumatologists and radiologists in the US are less experienced in using ultrasound to diagnose temporal artery involvement in GCA compared to their counterparts in Europe,” the 2021 guidelines stated. “In centers with appropriate training and expertise in using temporal artery ultrasound, ultrasound may be a useful and complementary tool for diagnosing GCA.”
Methodology
In the study, researchers recruited 165 individuals with high clinical suspicion of GCA from August 2016 through February 2020 at six French hospitals. Only patients older than 50 years of age and with biologic inflammatory syndrome with C-reactive protein elevation (≥ 6 mg/L) qualified for the study. Patients also needed to have at least one of these factors:
- Clinical signs of GCA (abnormal temporal arteries, scalp hyperesthesia, jaw claudication, or vision loss)
- General signs of GCA (headache, fever, or impaired general condition)
- Large-vessel vasculitis visible on imaging (CT angiography [CTA], MR angiography [MRA], and/or PET/CT)
All participants underwent a color Doppler ultrasound of the temporal artery, performed less than 1 week after the initiation of corticosteroid therapy. (Previous research demonstrated that corticosteroids can change the hallmark halo sign of vasculitis detectable via ultrasound as early as 1 week after initiation of therapy, the authors noted.) In this study, the time between consultation with a specialist and ultrasound was less than 1 day.
“Patients with halo signs detected around the lumen of both temporal arteries (that is, bilateral temporal halo sign) were considered as ultrasound-positive,” Guillaume Denis, MD, and colleagues explained. “Patients with no halo sign, or bilateral halo signs in the axillary arteries, or a unilateral halo sign in the temporal artery were considered as ultrasound-negative.”
The findings were published in Annals of Internal Medicine on May 7.
Results
In total, 73 participants (44%) had positive ultrasounds and were diagnosed with GCA. These patients also underwent a second ultrasound a month later to document if the halo sign remained unchanged, reduced, or disappeared.
The remaining 92 patients with negative ultrasound results underwent TAB, which was conducted on average 4.5 days after the ultrasound. A total of 28 patients (30%) had a positive TAB result. Physicians diagnosed 35 TAB-negative patients with GCA using clinical, imaging, and biologic data, and 29 patients received alternative diagnoses. These other diagnoses included polymyalgia rheumatica, infectious diseases, cancer, and other systemic inflammatory rheumatic diseases.
All patients diagnosed with GCA via ultrasound had their diagnoses reconfirmed at 1 month and for up to 2 years of follow-up.
“In summary, our study showed that the use of temporal artery ultrasound may be an efficient way to make the diagnosis of GCA in patients with high clinical suspicion and to reduce imaging costs and the need for biopsy, thereby limiting complications and the need for a surgeon,” the authors concluded.
Qualifications and Limitations
While over half of patients ultimately diagnosed with GCA were diagnosed using ultrasound, that percentage was “a bit lower than expected,” said Mark Matza, MD, MBA, the co-clinical director of rheumatology at Massachusetts General Hospital in Boston. By comparison, one systematic review calculated ultrasound’s pooled sensitivity at 88% and pooled specificity at 96% for the diagnosis of GCA.
“In this [current] study, 30% of patients who had negative ultrasound were then found to have positive biopsy, indicating that ultrasound missed a substantial portion of patients who were ultimately diagnosed with GCA,” he continued.
Ultrasound is “very operator dependent,” he added, and there has been “variability in test performance of ultrasound.”
The authors acknowledged that techniques for ultrasound of the temporal arteries have also evolved over the study period, and thus, findings may not have been consistent.
However, about one in four patients with GCA were diagnosed after having both negative ultrasound and TAB results.
“One of the things that this paper shows is that even the gold standard of temporal artery biopsy isn’t 100% either,” noted Minna Kohler, MD, who directs the rheumatology musculoskeletal ultrasound program at Massachusetts General Hospital. “That’s why clinically, there is an increasing emphasis on using multimodality imaging to assist in the diagnosis of GCA along with a physician’s clinical intuition,” she said.
While ultrasound can visualize axillary, subclavian, and carotid arteries, other imaging modalities such as CTA, MRA, and PET/CT are better to fully assess supra-aortic and aortic vessels, she continued. However, “this imaging is more expensive and takes more time to coordinate, schedule, whereas ultrasound of temporal and axillary arteries can easily be done within the clinic with an immediate answer.”
This study was supported by a grant from “Recherche CH-CHU Poitou-Charentes 2014.” Dr. Denis disclosed relationships with Leo Pharma, Janssen, Novartis, Takeda, and Sanofi. Dr. Matza reported honoraria from the Ultrasound School of North American Rheumatologists. Kohler had no relevant disclosures.
A version of this article appeared on Medscape.com.
Temporal artery ultrasound alone was sufficient to accurately diagnose giant cell arteritis (GCA) in over half of patients in a new prospective study.
The findings provide further evidence that “[ultrasound] of temporal arteries could really take the place of traditional temporal artery biopsy (TAB)” in patients with high clinical suspicion of GCA, lead author Guillaume Denis, MD, of the Centre Hospitalier de Rochefort in Rochefort, France, told this news organization.
The European Alliance of Associations for Rheumatology (EULAR) already recommends ultrasound as a first-line diagnostic tool for patients with suspected large vessel vasculitis, and the 2022 American College of Rheumatology (ACR)/EULAR classification criteria for GCA weighs positive TAB or temporal artery halo sign on ultrasound equally.
Guidelines from the ACR and the Vasculitis Foundation still recommend TAB over ultrasound.
“In general, rheumatologists and radiologists in the US are less experienced in using ultrasound to diagnose temporal artery involvement in GCA compared to their counterparts in Europe,” the 2021 guidelines stated. “In centers with appropriate training and expertise in using temporal artery ultrasound, ultrasound may be a useful and complementary tool for diagnosing GCA.”
Methodology
In the study, researchers recruited 165 individuals with high clinical suspicion of GCA from August 2016 through February 2020 at six French hospitals. Only patients older than 50 years of age and with biologic inflammatory syndrome with C-reactive protein elevation (≥ 6 mg/L) qualified for the study. Patients also needed to have at least one of these factors:
- Clinical signs of GCA (abnormal temporal arteries, scalp hyperesthesia, jaw claudication, or vision loss)
- General signs of GCA (headache, fever, or impaired general condition)
- Large-vessel vasculitis visible on imaging (CT angiography [CTA], MR angiography [MRA], and/or PET/CT)
All participants underwent a color Doppler ultrasound of the temporal artery, performed less than 1 week after the initiation of corticosteroid therapy. (Previous research demonstrated that corticosteroids can change the hallmark halo sign of vasculitis detectable via ultrasound as early as 1 week after initiation of therapy, the authors noted.) In this study, the time between consultation with a specialist and ultrasound was less than 1 day.
“Patients with halo signs detected around the lumen of both temporal arteries (that is, bilateral temporal halo sign) were considered as ultrasound-positive,” Guillaume Denis, MD, and colleagues explained. “Patients with no halo sign, or bilateral halo signs in the axillary arteries, or a unilateral halo sign in the temporal artery were considered as ultrasound-negative.”
The findings were published in Annals of Internal Medicine on May 7.
Results
In total, 73 participants (44%) had positive ultrasounds and were diagnosed with GCA. These patients also underwent a second ultrasound a month later to document if the halo sign remained unchanged, reduced, or disappeared.
The remaining 92 patients with negative ultrasound results underwent TAB, which was conducted on average 4.5 days after the ultrasound. A total of 28 patients (30%) had a positive TAB result. Physicians diagnosed 35 TAB-negative patients with GCA using clinical, imaging, and biologic data, and 29 patients received alternative diagnoses. These other diagnoses included polymyalgia rheumatica, infectious diseases, cancer, and other systemic inflammatory rheumatic diseases.
All patients diagnosed with GCA via ultrasound had their diagnoses reconfirmed at 1 month and for up to 2 years of follow-up.
“In summary, our study showed that the use of temporal artery ultrasound may be an efficient way to make the diagnosis of GCA in patients with high clinical suspicion and to reduce imaging costs and the need for biopsy, thereby limiting complications and the need for a surgeon,” the authors concluded.
Qualifications and Limitations
While over half of patients ultimately diagnosed with GCA were diagnosed using ultrasound, that percentage was “a bit lower than expected,” said Mark Matza, MD, MBA, the co-clinical director of rheumatology at Massachusetts General Hospital in Boston. By comparison, one systematic review calculated ultrasound’s pooled sensitivity at 88% and pooled specificity at 96% for the diagnosis of GCA.
“In this [current] study, 30% of patients who had negative ultrasound were then found to have positive biopsy, indicating that ultrasound missed a substantial portion of patients who were ultimately diagnosed with GCA,” he continued.
Ultrasound is “very operator dependent,” he added, and there has been “variability in test performance of ultrasound.”
The authors acknowledged that techniques for ultrasound of the temporal arteries have also evolved over the study period, and thus, findings may not have been consistent.
However, about one in four patients with GCA were diagnosed after having both negative ultrasound and TAB results.
“One of the things that this paper shows is that even the gold standard of temporal artery biopsy isn’t 100% either,” noted Minna Kohler, MD, who directs the rheumatology musculoskeletal ultrasound program at Massachusetts General Hospital. “That’s why clinically, there is an increasing emphasis on using multimodality imaging to assist in the diagnosis of GCA along with a physician’s clinical intuition,” she said.
While ultrasound can visualize axillary, subclavian, and carotid arteries, other imaging modalities such as CTA, MRA, and PET/CT are better to fully assess supra-aortic and aortic vessels, she continued. However, “this imaging is more expensive and takes more time to coordinate, schedule, whereas ultrasound of temporal and axillary arteries can easily be done within the clinic with an immediate answer.”
This study was supported by a grant from “Recherche CH-CHU Poitou-Charentes 2014.” Dr. Denis disclosed relationships with Leo Pharma, Janssen, Novartis, Takeda, and Sanofi. Dr. Matza reported honoraria from the Ultrasound School of North American Rheumatologists. Kohler had no relevant disclosures.
A version of this article appeared on Medscape.com.
Temporal artery ultrasound alone was sufficient to accurately diagnose giant cell arteritis (GCA) in over half of patients in a new prospective study.
The findings provide further evidence that “[ultrasound] of temporal arteries could really take the place of traditional temporal artery biopsy (TAB)” in patients with high clinical suspicion of GCA, lead author Guillaume Denis, MD, of the Centre Hospitalier de Rochefort in Rochefort, France, told this news organization.
The European Alliance of Associations for Rheumatology (EULAR) already recommends ultrasound as a first-line diagnostic tool for patients with suspected large vessel vasculitis, and the 2022 American College of Rheumatology (ACR)/EULAR classification criteria for GCA weighs positive TAB or temporal artery halo sign on ultrasound equally.
Guidelines from the ACR and the Vasculitis Foundation still recommend TAB over ultrasound.
“In general, rheumatologists and radiologists in the US are less experienced in using ultrasound to diagnose temporal artery involvement in GCA compared to their counterparts in Europe,” the 2021 guidelines stated. “In centers with appropriate training and expertise in using temporal artery ultrasound, ultrasound may be a useful and complementary tool for diagnosing GCA.”
Methodology
In the study, researchers recruited 165 individuals with high clinical suspicion of GCA from August 2016 through February 2020 at six French hospitals. Only patients older than 50 years of age and with biologic inflammatory syndrome with C-reactive protein elevation (≥ 6 mg/L) qualified for the study. Patients also needed to have at least one of these factors:
- Clinical signs of GCA (abnormal temporal arteries, scalp hyperesthesia, jaw claudication, or vision loss)
- General signs of GCA (headache, fever, or impaired general condition)
- Large-vessel vasculitis visible on imaging (CT angiography [CTA], MR angiography [MRA], and/or PET/CT)
All participants underwent a color Doppler ultrasound of the temporal artery, performed less than 1 week after the initiation of corticosteroid therapy. (Previous research demonstrated that corticosteroids can change the hallmark halo sign of vasculitis detectable via ultrasound as early as 1 week after initiation of therapy, the authors noted.) In this study, the time between consultation with a specialist and ultrasound was less than 1 day.
“Patients with halo signs detected around the lumen of both temporal arteries (that is, bilateral temporal halo sign) were considered as ultrasound-positive,” Guillaume Denis, MD, and colleagues explained. “Patients with no halo sign, or bilateral halo signs in the axillary arteries, or a unilateral halo sign in the temporal artery were considered as ultrasound-negative.”
The findings were published in Annals of Internal Medicine on May 7.
Results
In total, 73 participants (44%) had positive ultrasounds and were diagnosed with GCA. These patients also underwent a second ultrasound a month later to document if the halo sign remained unchanged, reduced, or disappeared.
The remaining 92 patients with negative ultrasound results underwent TAB, which was conducted on average 4.5 days after the ultrasound. A total of 28 patients (30%) had a positive TAB result. Physicians diagnosed 35 TAB-negative patients with GCA using clinical, imaging, and biologic data, and 29 patients received alternative diagnoses. These other diagnoses included polymyalgia rheumatica, infectious diseases, cancer, and other systemic inflammatory rheumatic diseases.
All patients diagnosed with GCA via ultrasound had their diagnoses reconfirmed at 1 month and for up to 2 years of follow-up.
“In summary, our study showed that the use of temporal artery ultrasound may be an efficient way to make the diagnosis of GCA in patients with high clinical suspicion and to reduce imaging costs and the need for biopsy, thereby limiting complications and the need for a surgeon,” the authors concluded.
Qualifications and Limitations
While over half of patients ultimately diagnosed with GCA were diagnosed using ultrasound, that percentage was “a bit lower than expected,” said Mark Matza, MD, MBA, the co-clinical director of rheumatology at Massachusetts General Hospital in Boston. By comparison, one systematic review calculated ultrasound’s pooled sensitivity at 88% and pooled specificity at 96% for the diagnosis of GCA.
“In this [current] study, 30% of patients who had negative ultrasound were then found to have positive biopsy, indicating that ultrasound missed a substantial portion of patients who were ultimately diagnosed with GCA,” he continued.
Ultrasound is “very operator dependent,” he added, and there has been “variability in test performance of ultrasound.”
The authors acknowledged that techniques for ultrasound of the temporal arteries have also evolved over the study period, and thus, findings may not have been consistent.
However, about one in four patients with GCA were diagnosed after having both negative ultrasound and TAB results.
“One of the things that this paper shows is that even the gold standard of temporal artery biopsy isn’t 100% either,” noted Minna Kohler, MD, who directs the rheumatology musculoskeletal ultrasound program at Massachusetts General Hospital. “That’s why clinically, there is an increasing emphasis on using multimodality imaging to assist in the diagnosis of GCA along with a physician’s clinical intuition,” she said.
While ultrasound can visualize axillary, subclavian, and carotid arteries, other imaging modalities such as CTA, MRA, and PET/CT are better to fully assess supra-aortic and aortic vessels, she continued. However, “this imaging is more expensive and takes more time to coordinate, schedule, whereas ultrasound of temporal and axillary arteries can easily be done within the clinic with an immediate answer.”
This study was supported by a grant from “Recherche CH-CHU Poitou-Charentes 2014.” Dr. Denis disclosed relationships with Leo Pharma, Janssen, Novartis, Takeda, and Sanofi. Dr. Matza reported honoraria from the Ultrasound School of North American Rheumatologists. Kohler had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
Monoclonal Antibody With Unique Mechanism Gets Second Chance in RA
LIVERPOOL, ENGLAND — The IRIS-RA study of the investigational monoclonal antibody drug nipocalimab in patients with rheumatoid arthritis (RA) did not meet its primary endpoint, but there could still be people with moderate to severe RA who might benefit from treatment with the drug, according to information reported at the British Society for Rheumatology annual meeting.
The primary endpoint for the phase 2A trial was the least squares mean change in Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) from baseline to 12 weeks of treatment. This was reduced by −1.03 with nipocalimab and by −0.58 with placebo, giving a mean difference of just −0.45 (P = .224).
However, one of the key secondary endpoints was the proportion of patients who had 20% improvement in American College of Rheumatology response criteria (ACR20). Results for this endpoint showed a greater difference in response to nipocalimab vs placebo, with a respective 45.5% and 20.0% (P = .055) of individuals achieving ACR20.
Moreover, an analysis stratifying for anti-citrullinated protein autoantibody (ACPA) levels at baseline found that people with higher levels had a better response to nipocalimab.
Choice of Endpoint
“The way this study was powered was to look at a change between the treatment groups of a DAS28-CRP reduction of 1.0,” said Peter C. Taylor, BMBCh, PhD, the Norman Collisson chair of musculoskeletal medicine at the University of Oxford in Oxford, England.
DAS28-CRP was often chosen as the primary endpoint in small proof-of-concept studies, such as IRIS-RA, because it was a “measure of continuous change [that] theoretically, would allow greater sensitivity to change,” Dr. Taylor added.
“Ironically, it has to be said that had we chosen ACR20, we would have hit the primary endpoint. One lives and learns,” noted Dr. Taylor.
Proof of Concept
IRIS-RA was billed as a “proof-of-concept” study because it was the first time that a monoclonal antibody targeting the neonatal fragment crystallizable receptor (FcRn) was being tested in an RA population.
The study was a randomized double-blind trial in which 33 people with moderate to severe RA who had an inadequate response to tumor necrosis factor (TNF) inhibitors were treated with nipocalimab at a dose of 15 mg/kg given intravenously every 2 weeks, and 20 received a matching placebo. Participants were treated for 10 weeks, and then the primary follow-up was at 12 weeks, with additional follow-up for safety undertaken at 18 weeks.
Nipocalimab is a fully human, immunoglobulin G1 (IgG1) monoclonal antibody that is designed to selectively block the FcRn. By doing so, it essentially stops IgG from being recycled within the immune system, and this in turn lowers IgG levels. That includes potentially harmful ACPAs, among other pathogenic antibodies, Dr. Taylor and fellow investigators explained in their abstract.
“We’ve known for a long time that ACPA have prognostic value, but there’s been controversy about whether or not ACPA are actually pathogenic,” Dr. Taylor said. “So, one of the hypotheses that this study gives rise to is that by blocking FcRn, and thereby reducing, potentially, the concentration of ACPA in the blood, will we actually have efficacy in patients?”
Are ACPA Really Lowered?
Paul Emery, MD, Versus Arthritis professor of rheumatology and director of the Leeds Biomedical Research Centre at the University of Leeds in Leeds, England, questioned the reduction in antibody levels during the discussion that followed.
Although these data had not been presented, Dr. Emery observed that the reduction in IgG was actually greater than that in ACPA, “which is fairly critical. Is it feasible to look to selectively lower normal immunoglobulin over pathogenic autoantibodies?”
Dr. Emery also wanted to know if there “was a floor on the reduction of immunoglobulin” with long-term therapy, “which would be a worry.”
Dr. Taylor responded that total IgG had been reduced by about 65% and ACPA by about 40%. Why this difference exists is not yet clear. It could be because ACPA are part of complexed antibodies.
“Most of these patients are rheumatoid factor [RF]–positive,” said Taylor, pointing out that although IgM “wouldn’t normally be affected by FcRn blockade,” there was a 10% reduction in RF IgM, probably because it was complexed to IgG.
“So, the hypothesis here is that if you look at the clearance of complexes, they’re handled differently in the cytoplasm from the clearance of monomeric IgG. But that’s a hypothesis. It needs further investigation. In vitro, there’s very good, confirmatory evidence to support that. But we’ve yet to explore that more fully in vivo,” Dr. Taylor said.
As for long-term effects, Dr. Taylor responded: “All I can tell you is [that] after the 10-week intervention, that up to an 18-week observation period, immunoglobulin levels recovered very rapidly afterwards. And you mustn’t forget that other isotypes are not affected, unlike rituximab.”
Safety and Other Results
With regard to safety, 27 (82%) of nipocalimab- and 12 (60%) of placebo-treated participants experienced at least one treatment-emergent adverse event (TEAE). The most common, occurring in 10% or more of cases, were RA flares (36.4% for nipocalimab vs 15.0% with placebo), headache (12.1% vs 5.0%), and COVID-19 (12.1% vs 0.0%).
There were three serious TEAEs, all in the nipocalimab-treatment group: One was an infection of a burn that had been present at inclusion, another was a deep vein thrombosis that resolved with apixaban treatment, and the other was an infusion-related reaction that resolved with supportive treatment.
Another notable efficacy finding was the proportion of patients achieving DAS28-CRP remission at 12 weeks in the nipocalimab vs the placebo group was substantially greater if considering only people with high baseline ACPA levels, at a respective 40.0% vs 16.7%, when compared with the total population (21.2% vs 10.0%).
Similar findings were seen for the proportion of patients achieving an ACR50, and there were numerically greater reductions in the components of the ACR response criteria such as tender and swollen joints with nipocalimab vs placebo. All of these were exploratory observations, Dr. Taylor emphasized.
Combination and Further Trials
Further trials of nipocalimab are planned or are already ongoing in systemic lupus erythematosus, active lupus nephritis, Sjögren disease, and five other diseases.
In RA, nipocalimab is now being tested in combination with the TNF inhibitor certolizumab pegol (Cimzia) in the DAISY-RA trial. This is another proof-of-concept, phase 2A trial with a target accrual of 104 patients.
The IRIS-RA study was funded by Janssen Research & Development. Dr. Taylor serves as a consultant to AbbVie, Biogen, Eli Lilly, Fresenius, Galapagos, Gilead Sciences, GlaxoSmithKline, Janssen, Nordic Pharma, Pfizer, Sanofi, Aqtual, and UCB and received research funding from Galapagos, among others. Dr. Emery received research grants paid to his institution from AbbVie, Bristol Myers Squibb (BMS), Pfizer, MSD, and Roche; received consultant fees from BMS, AbbVie, Pfizer, MSD, Novartis, Roche, and UCB; and has undertaken clinical trials and provided expert advice to Pfizer, MSD, AbbVie, BMS, UCB, Roche, Novartis, Samsung, Sandoz, and Lilly.
A version of this article appeared on Medscape.com.
LIVERPOOL, ENGLAND — The IRIS-RA study of the investigational monoclonal antibody drug nipocalimab in patients with rheumatoid arthritis (RA) did not meet its primary endpoint, but there could still be people with moderate to severe RA who might benefit from treatment with the drug, according to information reported at the British Society for Rheumatology annual meeting.
The primary endpoint for the phase 2A trial was the least squares mean change in Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) from baseline to 12 weeks of treatment. This was reduced by −1.03 with nipocalimab and by −0.58 with placebo, giving a mean difference of just −0.45 (P = .224).
However, one of the key secondary endpoints was the proportion of patients who had 20% improvement in American College of Rheumatology response criteria (ACR20). Results for this endpoint showed a greater difference in response to nipocalimab vs placebo, with a respective 45.5% and 20.0% (P = .055) of individuals achieving ACR20.
Moreover, an analysis stratifying for anti-citrullinated protein autoantibody (ACPA) levels at baseline found that people with higher levels had a better response to nipocalimab.
Choice of Endpoint
“The way this study was powered was to look at a change between the treatment groups of a DAS28-CRP reduction of 1.0,” said Peter C. Taylor, BMBCh, PhD, the Norman Collisson chair of musculoskeletal medicine at the University of Oxford in Oxford, England.
DAS28-CRP was often chosen as the primary endpoint in small proof-of-concept studies, such as IRIS-RA, because it was a “measure of continuous change [that] theoretically, would allow greater sensitivity to change,” Dr. Taylor added.
“Ironically, it has to be said that had we chosen ACR20, we would have hit the primary endpoint. One lives and learns,” noted Dr. Taylor.
Proof of Concept
IRIS-RA was billed as a “proof-of-concept” study because it was the first time that a monoclonal antibody targeting the neonatal fragment crystallizable receptor (FcRn) was being tested in an RA population.
The study was a randomized double-blind trial in which 33 people with moderate to severe RA who had an inadequate response to tumor necrosis factor (TNF) inhibitors were treated with nipocalimab at a dose of 15 mg/kg given intravenously every 2 weeks, and 20 received a matching placebo. Participants were treated for 10 weeks, and then the primary follow-up was at 12 weeks, with additional follow-up for safety undertaken at 18 weeks.
Nipocalimab is a fully human, immunoglobulin G1 (IgG1) monoclonal antibody that is designed to selectively block the FcRn. By doing so, it essentially stops IgG from being recycled within the immune system, and this in turn lowers IgG levels. That includes potentially harmful ACPAs, among other pathogenic antibodies, Dr. Taylor and fellow investigators explained in their abstract.
“We’ve known for a long time that ACPA have prognostic value, but there’s been controversy about whether or not ACPA are actually pathogenic,” Dr. Taylor said. “So, one of the hypotheses that this study gives rise to is that by blocking FcRn, and thereby reducing, potentially, the concentration of ACPA in the blood, will we actually have efficacy in patients?”
Are ACPA Really Lowered?
Paul Emery, MD, Versus Arthritis professor of rheumatology and director of the Leeds Biomedical Research Centre at the University of Leeds in Leeds, England, questioned the reduction in antibody levels during the discussion that followed.
Although these data had not been presented, Dr. Emery observed that the reduction in IgG was actually greater than that in ACPA, “which is fairly critical. Is it feasible to look to selectively lower normal immunoglobulin over pathogenic autoantibodies?”
Dr. Emery also wanted to know if there “was a floor on the reduction of immunoglobulin” with long-term therapy, “which would be a worry.”
Dr. Taylor responded that total IgG had been reduced by about 65% and ACPA by about 40%. Why this difference exists is not yet clear. It could be because ACPA are part of complexed antibodies.
“Most of these patients are rheumatoid factor [RF]–positive,” said Taylor, pointing out that although IgM “wouldn’t normally be affected by FcRn blockade,” there was a 10% reduction in RF IgM, probably because it was complexed to IgG.
“So, the hypothesis here is that if you look at the clearance of complexes, they’re handled differently in the cytoplasm from the clearance of monomeric IgG. But that’s a hypothesis. It needs further investigation. In vitro, there’s very good, confirmatory evidence to support that. But we’ve yet to explore that more fully in vivo,” Dr. Taylor said.
As for long-term effects, Dr. Taylor responded: “All I can tell you is [that] after the 10-week intervention, that up to an 18-week observation period, immunoglobulin levels recovered very rapidly afterwards. And you mustn’t forget that other isotypes are not affected, unlike rituximab.”
Safety and Other Results
With regard to safety, 27 (82%) of nipocalimab- and 12 (60%) of placebo-treated participants experienced at least one treatment-emergent adverse event (TEAE). The most common, occurring in 10% or more of cases, were RA flares (36.4% for nipocalimab vs 15.0% with placebo), headache (12.1% vs 5.0%), and COVID-19 (12.1% vs 0.0%).
There were three serious TEAEs, all in the nipocalimab-treatment group: One was an infection of a burn that had been present at inclusion, another was a deep vein thrombosis that resolved with apixaban treatment, and the other was an infusion-related reaction that resolved with supportive treatment.
Another notable efficacy finding was the proportion of patients achieving DAS28-CRP remission at 12 weeks in the nipocalimab vs the placebo group was substantially greater if considering only people with high baseline ACPA levels, at a respective 40.0% vs 16.7%, when compared with the total population (21.2% vs 10.0%).
Similar findings were seen for the proportion of patients achieving an ACR50, and there were numerically greater reductions in the components of the ACR response criteria such as tender and swollen joints with nipocalimab vs placebo. All of these were exploratory observations, Dr. Taylor emphasized.
Combination and Further Trials
Further trials of nipocalimab are planned or are already ongoing in systemic lupus erythematosus, active lupus nephritis, Sjögren disease, and five other diseases.
In RA, nipocalimab is now being tested in combination with the TNF inhibitor certolizumab pegol (Cimzia) in the DAISY-RA trial. This is another proof-of-concept, phase 2A trial with a target accrual of 104 patients.
The IRIS-RA study was funded by Janssen Research & Development. Dr. Taylor serves as a consultant to AbbVie, Biogen, Eli Lilly, Fresenius, Galapagos, Gilead Sciences, GlaxoSmithKline, Janssen, Nordic Pharma, Pfizer, Sanofi, Aqtual, and UCB and received research funding from Galapagos, among others. Dr. Emery received research grants paid to his institution from AbbVie, Bristol Myers Squibb (BMS), Pfizer, MSD, and Roche; received consultant fees from BMS, AbbVie, Pfizer, MSD, Novartis, Roche, and UCB; and has undertaken clinical trials and provided expert advice to Pfizer, MSD, AbbVie, BMS, UCB, Roche, Novartis, Samsung, Sandoz, and Lilly.
A version of this article appeared on Medscape.com.
LIVERPOOL, ENGLAND — The IRIS-RA study of the investigational monoclonal antibody drug nipocalimab in patients with rheumatoid arthritis (RA) did not meet its primary endpoint, but there could still be people with moderate to severe RA who might benefit from treatment with the drug, according to information reported at the British Society for Rheumatology annual meeting.
The primary endpoint for the phase 2A trial was the least squares mean change in Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) from baseline to 12 weeks of treatment. This was reduced by −1.03 with nipocalimab and by −0.58 with placebo, giving a mean difference of just −0.45 (P = .224).
However, one of the key secondary endpoints was the proportion of patients who had 20% improvement in American College of Rheumatology response criteria (ACR20). Results for this endpoint showed a greater difference in response to nipocalimab vs placebo, with a respective 45.5% and 20.0% (P = .055) of individuals achieving ACR20.
Moreover, an analysis stratifying for anti-citrullinated protein autoantibody (ACPA) levels at baseline found that people with higher levels had a better response to nipocalimab.
Choice of Endpoint
“The way this study was powered was to look at a change between the treatment groups of a DAS28-CRP reduction of 1.0,” said Peter C. Taylor, BMBCh, PhD, the Norman Collisson chair of musculoskeletal medicine at the University of Oxford in Oxford, England.
DAS28-CRP was often chosen as the primary endpoint in small proof-of-concept studies, such as IRIS-RA, because it was a “measure of continuous change [that] theoretically, would allow greater sensitivity to change,” Dr. Taylor added.
“Ironically, it has to be said that had we chosen ACR20, we would have hit the primary endpoint. One lives and learns,” noted Dr. Taylor.
Proof of Concept
IRIS-RA was billed as a “proof-of-concept” study because it was the first time that a monoclonal antibody targeting the neonatal fragment crystallizable receptor (FcRn) was being tested in an RA population.
The study was a randomized double-blind trial in which 33 people with moderate to severe RA who had an inadequate response to tumor necrosis factor (TNF) inhibitors were treated with nipocalimab at a dose of 15 mg/kg given intravenously every 2 weeks, and 20 received a matching placebo. Participants were treated for 10 weeks, and then the primary follow-up was at 12 weeks, with additional follow-up for safety undertaken at 18 weeks.
Nipocalimab is a fully human, immunoglobulin G1 (IgG1) monoclonal antibody that is designed to selectively block the FcRn. By doing so, it essentially stops IgG from being recycled within the immune system, and this in turn lowers IgG levels. That includes potentially harmful ACPAs, among other pathogenic antibodies, Dr. Taylor and fellow investigators explained in their abstract.
“We’ve known for a long time that ACPA have prognostic value, but there’s been controversy about whether or not ACPA are actually pathogenic,” Dr. Taylor said. “So, one of the hypotheses that this study gives rise to is that by blocking FcRn, and thereby reducing, potentially, the concentration of ACPA in the blood, will we actually have efficacy in patients?”
Are ACPA Really Lowered?
Paul Emery, MD, Versus Arthritis professor of rheumatology and director of the Leeds Biomedical Research Centre at the University of Leeds in Leeds, England, questioned the reduction in antibody levels during the discussion that followed.
Although these data had not been presented, Dr. Emery observed that the reduction in IgG was actually greater than that in ACPA, “which is fairly critical. Is it feasible to look to selectively lower normal immunoglobulin over pathogenic autoantibodies?”
Dr. Emery also wanted to know if there “was a floor on the reduction of immunoglobulin” with long-term therapy, “which would be a worry.”
Dr. Taylor responded that total IgG had been reduced by about 65% and ACPA by about 40%. Why this difference exists is not yet clear. It could be because ACPA are part of complexed antibodies.
“Most of these patients are rheumatoid factor [RF]–positive,” said Taylor, pointing out that although IgM “wouldn’t normally be affected by FcRn blockade,” there was a 10% reduction in RF IgM, probably because it was complexed to IgG.
“So, the hypothesis here is that if you look at the clearance of complexes, they’re handled differently in the cytoplasm from the clearance of monomeric IgG. But that’s a hypothesis. It needs further investigation. In vitro, there’s very good, confirmatory evidence to support that. But we’ve yet to explore that more fully in vivo,” Dr. Taylor said.
As for long-term effects, Dr. Taylor responded: “All I can tell you is [that] after the 10-week intervention, that up to an 18-week observation period, immunoglobulin levels recovered very rapidly afterwards. And you mustn’t forget that other isotypes are not affected, unlike rituximab.”
Safety and Other Results
With regard to safety, 27 (82%) of nipocalimab- and 12 (60%) of placebo-treated participants experienced at least one treatment-emergent adverse event (TEAE). The most common, occurring in 10% or more of cases, were RA flares (36.4% for nipocalimab vs 15.0% with placebo), headache (12.1% vs 5.0%), and COVID-19 (12.1% vs 0.0%).
There were three serious TEAEs, all in the nipocalimab-treatment group: One was an infection of a burn that had been present at inclusion, another was a deep vein thrombosis that resolved with apixaban treatment, and the other was an infusion-related reaction that resolved with supportive treatment.
Another notable efficacy finding was the proportion of patients achieving DAS28-CRP remission at 12 weeks in the nipocalimab vs the placebo group was substantially greater if considering only people with high baseline ACPA levels, at a respective 40.0% vs 16.7%, when compared with the total population (21.2% vs 10.0%).
Similar findings were seen for the proportion of patients achieving an ACR50, and there were numerically greater reductions in the components of the ACR response criteria such as tender and swollen joints with nipocalimab vs placebo. All of these were exploratory observations, Dr. Taylor emphasized.
Combination and Further Trials
Further trials of nipocalimab are planned or are already ongoing in systemic lupus erythematosus, active lupus nephritis, Sjögren disease, and five other diseases.
In RA, nipocalimab is now being tested in combination with the TNF inhibitor certolizumab pegol (Cimzia) in the DAISY-RA trial. This is another proof-of-concept, phase 2A trial with a target accrual of 104 patients.
The IRIS-RA study was funded by Janssen Research & Development. Dr. Taylor serves as a consultant to AbbVie, Biogen, Eli Lilly, Fresenius, Galapagos, Gilead Sciences, GlaxoSmithKline, Janssen, Nordic Pharma, Pfizer, Sanofi, Aqtual, and UCB and received research funding from Galapagos, among others. Dr. Emery received research grants paid to his institution from AbbVie, Bristol Myers Squibb (BMS), Pfizer, MSD, and Roche; received consultant fees from BMS, AbbVie, Pfizer, MSD, Novartis, Roche, and UCB; and has undertaken clinical trials and provided expert advice to Pfizer, MSD, AbbVie, BMS, UCB, Roche, Novartis, Samsung, Sandoz, and Lilly.
A version of this article appeared on Medscape.com.
FROM BSR 2024
Rural Health System ‘Teetering on Brink’ of Collapse, Says AMA
Physicians are leaving healthcare in droves, “not because they don’t want to practice ... but because the system is making it more and more difficult for them to care for their patients,” Bruce Scott, MD, president-elect of the American Medical Association (AMA), said at a press conference May 9 at the National Rural Health Association’s Annual Conference in New Orleans.
He said that shrinking reimbursement rates and excessive administrative tasks are pushing doctors out of the workforce, exacerbating physician shortages in rural locations where 46 million Americans live.
A recent Centers for Disease Control and Prevention report found that people living in rural areas are more likely to die early from preventable causes than their urban counterparts, said Dr. Scott.
He said the AMA wants Congress to pass legislation to incentivize more physicians to work in rural areas and expand the number of rural and primary care residency spots. Historically, 80% of residents practice within 80 miles of where they complete residency, he said.
Dr. Scott also hopes Congress will revise the J-1 visa rules to allow qualified international medical graduates to continue to practice in the United States. He’d like to see the pandemic telehealth flexibilities made permanent because these loosened guidelines greatly improved care access for rural areas in recent years.
Lower Pay Affects Care in Rural, Urban Areas
Decreased reimbursements also have hit rural and urban doctors in independent practice particularly hard, Dr. Scott said. When adjusted for inflation, the current Medicare payment rate for physicians has dropped 29% since 2001, he said. Now that commercial payers tie their reimbursement models to the Medicare rate, physicians are experiencing “severe” financial stress amid rising practice costs and student loan debt.
He shared anecdotes about how these issues have affected his private otolaryngology practice in Louisville, Kentucky, a state where more than 2 million people live in federally designated primary care professional shortage areas.
“A major insurance company that controls over 60% of the private payer market in rural Kentucky [recently] offered us ... surgical rates less than they paid us 6 years ago,” he said.
Dr. Scott said physicians must make difficult choices. “Do we not invest in the latest physical equipment? Do we reduce our number of employees? Do we perhaps stop accepting new Medicare patients?”
He noted that physicians now spend twice as much time on prior authorizations and other administrative tasks as they do on direct patient care. According to a 2022 AMA survey, 33% of physicians reported that the cumbersome prior authorization process led to a serious adverse event for a patient. Eighty percent reported it caused their patient to forgo treatment altogether.
Dr. Scott, who will be sworn in as AMA president in June, said he experiences the frustration daily.
“I have to get on the phone and justify to an insurance person who rarely has gone to medical school, has never seen the patient, and heck, in my case, sometimes they can’t even say otolaryngology, much less tell me what the appropriate care is for my patient,” he said.
When asked about the impact of private equity in healthcare, Dr. Scott said there is room for all different modes of practice, but private equity could bring a unique benefit.
“They have deeper pockets to potentially invest in telehealth technology, AI, and better computer systems,” he said.
But, he said, some private equity-owned systems have abandoned rural areas, and in other regions they “push the physicians to move faster, see more patients, and do the things that are profit-driven.
“The key is to continue to provide ... quality medical care that is determined by an individual physician in consultation with the patient.”
A version of this article appeared on Medscape.com.
Physicians are leaving healthcare in droves, “not because they don’t want to practice ... but because the system is making it more and more difficult for them to care for their patients,” Bruce Scott, MD, president-elect of the American Medical Association (AMA), said at a press conference May 9 at the National Rural Health Association’s Annual Conference in New Orleans.
He said that shrinking reimbursement rates and excessive administrative tasks are pushing doctors out of the workforce, exacerbating physician shortages in rural locations where 46 million Americans live.
A recent Centers for Disease Control and Prevention report found that people living in rural areas are more likely to die early from preventable causes than their urban counterparts, said Dr. Scott.
He said the AMA wants Congress to pass legislation to incentivize more physicians to work in rural areas and expand the number of rural and primary care residency spots. Historically, 80% of residents practice within 80 miles of where they complete residency, he said.
Dr. Scott also hopes Congress will revise the J-1 visa rules to allow qualified international medical graduates to continue to practice in the United States. He’d like to see the pandemic telehealth flexibilities made permanent because these loosened guidelines greatly improved care access for rural areas in recent years.
Lower Pay Affects Care in Rural, Urban Areas
Decreased reimbursements also have hit rural and urban doctors in independent practice particularly hard, Dr. Scott said. When adjusted for inflation, the current Medicare payment rate for physicians has dropped 29% since 2001, he said. Now that commercial payers tie their reimbursement models to the Medicare rate, physicians are experiencing “severe” financial stress amid rising practice costs and student loan debt.
He shared anecdotes about how these issues have affected his private otolaryngology practice in Louisville, Kentucky, a state where more than 2 million people live in federally designated primary care professional shortage areas.
“A major insurance company that controls over 60% of the private payer market in rural Kentucky [recently] offered us ... surgical rates less than they paid us 6 years ago,” he said.
Dr. Scott said physicians must make difficult choices. “Do we not invest in the latest physical equipment? Do we reduce our number of employees? Do we perhaps stop accepting new Medicare patients?”
He noted that physicians now spend twice as much time on prior authorizations and other administrative tasks as they do on direct patient care. According to a 2022 AMA survey, 33% of physicians reported that the cumbersome prior authorization process led to a serious adverse event for a patient. Eighty percent reported it caused their patient to forgo treatment altogether.
Dr. Scott, who will be sworn in as AMA president in June, said he experiences the frustration daily.
“I have to get on the phone and justify to an insurance person who rarely has gone to medical school, has never seen the patient, and heck, in my case, sometimes they can’t even say otolaryngology, much less tell me what the appropriate care is for my patient,” he said.
When asked about the impact of private equity in healthcare, Dr. Scott said there is room for all different modes of practice, but private equity could bring a unique benefit.
“They have deeper pockets to potentially invest in telehealth technology, AI, and better computer systems,” he said.
But, he said, some private equity-owned systems have abandoned rural areas, and in other regions they “push the physicians to move faster, see more patients, and do the things that are profit-driven.
“The key is to continue to provide ... quality medical care that is determined by an individual physician in consultation with the patient.”
A version of this article appeared on Medscape.com.
Physicians are leaving healthcare in droves, “not because they don’t want to practice ... but because the system is making it more and more difficult for them to care for their patients,” Bruce Scott, MD, president-elect of the American Medical Association (AMA), said at a press conference May 9 at the National Rural Health Association’s Annual Conference in New Orleans.
He said that shrinking reimbursement rates and excessive administrative tasks are pushing doctors out of the workforce, exacerbating physician shortages in rural locations where 46 million Americans live.
A recent Centers for Disease Control and Prevention report found that people living in rural areas are more likely to die early from preventable causes than their urban counterparts, said Dr. Scott.
He said the AMA wants Congress to pass legislation to incentivize more physicians to work in rural areas and expand the number of rural and primary care residency spots. Historically, 80% of residents practice within 80 miles of where they complete residency, he said.
Dr. Scott also hopes Congress will revise the J-1 visa rules to allow qualified international medical graduates to continue to practice in the United States. He’d like to see the pandemic telehealth flexibilities made permanent because these loosened guidelines greatly improved care access for rural areas in recent years.
Lower Pay Affects Care in Rural, Urban Areas
Decreased reimbursements also have hit rural and urban doctors in independent practice particularly hard, Dr. Scott said. When adjusted for inflation, the current Medicare payment rate for physicians has dropped 29% since 2001, he said. Now that commercial payers tie their reimbursement models to the Medicare rate, physicians are experiencing “severe” financial stress amid rising practice costs and student loan debt.
He shared anecdotes about how these issues have affected his private otolaryngology practice in Louisville, Kentucky, a state where more than 2 million people live in federally designated primary care professional shortage areas.
“A major insurance company that controls over 60% of the private payer market in rural Kentucky [recently] offered us ... surgical rates less than they paid us 6 years ago,” he said.
Dr. Scott said physicians must make difficult choices. “Do we not invest in the latest physical equipment? Do we reduce our number of employees? Do we perhaps stop accepting new Medicare patients?”
He noted that physicians now spend twice as much time on prior authorizations and other administrative tasks as they do on direct patient care. According to a 2022 AMA survey, 33% of physicians reported that the cumbersome prior authorization process led to a serious adverse event for a patient. Eighty percent reported it caused their patient to forgo treatment altogether.
Dr. Scott, who will be sworn in as AMA president in June, said he experiences the frustration daily.
“I have to get on the phone and justify to an insurance person who rarely has gone to medical school, has never seen the patient, and heck, in my case, sometimes they can’t even say otolaryngology, much less tell me what the appropriate care is for my patient,” he said.
When asked about the impact of private equity in healthcare, Dr. Scott said there is room for all different modes of practice, but private equity could bring a unique benefit.
“They have deeper pockets to potentially invest in telehealth technology, AI, and better computer systems,” he said.
But, he said, some private equity-owned systems have abandoned rural areas, and in other regions they “push the physicians to move faster, see more patients, and do the things that are profit-driven.
“The key is to continue to provide ... quality medical care that is determined by an individual physician in consultation with the patient.”
A version of this article appeared on Medscape.com.
Jumpstart Your AI Learning: The Very Best Resources for Doctors
Like it or not, artificial intelligence (AI) is coming to medicine. For many physicians — maybe you — it’s already here.
More than a third of physicians use AI in their practice. And the vast majority of healthcare companies — 94%, according to Morgan Stanley — use some kind of AI machine learning.
“It’s incumbent on physicians, as well as physicians in training, to become familiar with at least the basics [of AI],” said internist Matthew DeCamp, MD, PhD, an associate professor in the Center for Bioethics and Humanities at the University of Colorado Anschutz Medical Campus, Aurora, Colorado.
“Frankly, the people who are deciding whether to implement algorithms in our day-to-day lives are oftentimes not physicians,” noted Ravi B. Parikh, MD, an assistant professor at the University of Pennsylvania and director of augmented and artificial intelligence at the Penn Center for Cancer Care Innovation, Philadelphia. Yet, physicians are most qualified to assess an AI tool’s usefulness in clinical practice.
That brings us to the best starting place for your AI education: Your own institution. Find out what AI tools your organization is implementing — and how you can influence them.
“Getting involved with our hospital data governance is the best way not only to learn practically what these AI tools do but also to influence the development process in positive ways,” Dr. Parikh said.
From there, consider the following resources to enhance your AI knowledge.
Get a Lay of the Land: Free Primers
Many clinical societies and interest groups have put out AI primers, an easy way to get a broad overview of the technology. The following were recommended or developed by the experts we spoke to, and all are free:
- The American Medical Association’s (AMA’s) framework for advancing healthcare AI lays out actionable guidance. Ask three key questions, the AMA recommends: Does it work? Does it work for my patients? Does it improve health outcomes?
- The Coalition for Health AI’s Blueprint for Trustworthy AI Implementation Guidance and Assurance for Healthcare provides a high-level summary of how to evaluate AI in healthcare, plus steps for implementing it. AI systems should be useful, safe, accountable, explainable, fair, and secure, the report asserted.
- The National Academy of Medicine’s draft code of conduct for AI in healthcare proposes core principles and commitments. These “reflect simple guideposts to guide and gauge behavior in a complex system and provide a starting point for real-time decision-making,” the report said.
- Health AI Partnership — a collaboration of Duke Health and Microsoft — outlines eight key decision points to consider at any stage of AI implementation, whether you’re still planning how to use it or you’ve started but want to improve it. The site also provides a breakdown of standards by regulatory agencies, organizations, and oversight bodies — so you can make sure your practices align with their guidance.
Make the Most of Conferences
Next time you’re at a conference, check the agenda for sessions on AI. “For someone who’s interested in this, I would be looking for content in my next national meeting because, undoubtedly, it’s going to be there,” said Dr. DeCamp. In a fast-moving field like AI, it’s a great way to get fresh, up-to-the-moment insights.
Listen to This Podcast
The New England Journal of Medicine’s free monthly podcast AI Grand Rounds is made for researchers and clinicians. Available on Apple, Spotify, and YouTube, the pod is good for “someone who’s looking to see both where the field is going [and to hear] a retrospective on big-name papers,” said Dr. Parikh . Episodes run for about an hour.
To learn about the challenges of applying AI to biology: Listen to Daphne Koller, PhD, founder of AI-driven drug discovery and development company insitro. For insights on the potential of AI in medicine, tune into the one with Eric Horvitz, MD, PhD, Microsoft’s chief scientific officer.
Consider a Class
Look for courses that focus on AI applications in clinical practice rather than a deep dive into theory. (You need to understand how these tools will influence your work, not the intricacies of large language model development.) Be wary of corporate-funded training that centers on one product , which could present conflicts of interest, said Dr. DeCamp. See the chart for courses that meet these criteria.
A version of this article appeared on Medscape.com.
Like it or not, artificial intelligence (AI) is coming to medicine. For many physicians — maybe you — it’s already here.
More than a third of physicians use AI in their practice. And the vast majority of healthcare companies — 94%, according to Morgan Stanley — use some kind of AI machine learning.
“It’s incumbent on physicians, as well as physicians in training, to become familiar with at least the basics [of AI],” said internist Matthew DeCamp, MD, PhD, an associate professor in the Center for Bioethics and Humanities at the University of Colorado Anschutz Medical Campus, Aurora, Colorado.
“Frankly, the people who are deciding whether to implement algorithms in our day-to-day lives are oftentimes not physicians,” noted Ravi B. Parikh, MD, an assistant professor at the University of Pennsylvania and director of augmented and artificial intelligence at the Penn Center for Cancer Care Innovation, Philadelphia. Yet, physicians are most qualified to assess an AI tool’s usefulness in clinical practice.
That brings us to the best starting place for your AI education: Your own institution. Find out what AI tools your organization is implementing — and how you can influence them.
“Getting involved with our hospital data governance is the best way not only to learn practically what these AI tools do but also to influence the development process in positive ways,” Dr. Parikh said.
From there, consider the following resources to enhance your AI knowledge.
Get a Lay of the Land: Free Primers
Many clinical societies and interest groups have put out AI primers, an easy way to get a broad overview of the technology. The following were recommended or developed by the experts we spoke to, and all are free:
- The American Medical Association’s (AMA’s) framework for advancing healthcare AI lays out actionable guidance. Ask three key questions, the AMA recommends: Does it work? Does it work for my patients? Does it improve health outcomes?
- The Coalition for Health AI’s Blueprint for Trustworthy AI Implementation Guidance and Assurance for Healthcare provides a high-level summary of how to evaluate AI in healthcare, plus steps for implementing it. AI systems should be useful, safe, accountable, explainable, fair, and secure, the report asserted.
- The National Academy of Medicine’s draft code of conduct for AI in healthcare proposes core principles and commitments. These “reflect simple guideposts to guide and gauge behavior in a complex system and provide a starting point for real-time decision-making,” the report said.
- Health AI Partnership — a collaboration of Duke Health and Microsoft — outlines eight key decision points to consider at any stage of AI implementation, whether you’re still planning how to use it or you’ve started but want to improve it. The site also provides a breakdown of standards by regulatory agencies, organizations, and oversight bodies — so you can make sure your practices align with their guidance.
Make the Most of Conferences
Next time you’re at a conference, check the agenda for sessions on AI. “For someone who’s interested in this, I would be looking for content in my next national meeting because, undoubtedly, it’s going to be there,” said Dr. DeCamp. In a fast-moving field like AI, it’s a great way to get fresh, up-to-the-moment insights.
Listen to This Podcast
The New England Journal of Medicine’s free monthly podcast AI Grand Rounds is made for researchers and clinicians. Available on Apple, Spotify, and YouTube, the pod is good for “someone who’s looking to see both where the field is going [and to hear] a retrospective on big-name papers,” said Dr. Parikh . Episodes run for about an hour.
To learn about the challenges of applying AI to biology: Listen to Daphne Koller, PhD, founder of AI-driven drug discovery and development company insitro. For insights on the potential of AI in medicine, tune into the one with Eric Horvitz, MD, PhD, Microsoft’s chief scientific officer.
Consider a Class
Look for courses that focus on AI applications in clinical practice rather than a deep dive into theory. (You need to understand how these tools will influence your work, not the intricacies of large language model development.) Be wary of corporate-funded training that centers on one product , which could present conflicts of interest, said Dr. DeCamp. See the chart for courses that meet these criteria.
A version of this article appeared on Medscape.com.
Like it or not, artificial intelligence (AI) is coming to medicine. For many physicians — maybe you — it’s already here.
More than a third of physicians use AI in their practice. And the vast majority of healthcare companies — 94%, according to Morgan Stanley — use some kind of AI machine learning.
“It’s incumbent on physicians, as well as physicians in training, to become familiar with at least the basics [of AI],” said internist Matthew DeCamp, MD, PhD, an associate professor in the Center for Bioethics and Humanities at the University of Colorado Anschutz Medical Campus, Aurora, Colorado.
“Frankly, the people who are deciding whether to implement algorithms in our day-to-day lives are oftentimes not physicians,” noted Ravi B. Parikh, MD, an assistant professor at the University of Pennsylvania and director of augmented and artificial intelligence at the Penn Center for Cancer Care Innovation, Philadelphia. Yet, physicians are most qualified to assess an AI tool’s usefulness in clinical practice.
That brings us to the best starting place for your AI education: Your own institution. Find out what AI tools your organization is implementing — and how you can influence them.
“Getting involved with our hospital data governance is the best way not only to learn practically what these AI tools do but also to influence the development process in positive ways,” Dr. Parikh said.
From there, consider the following resources to enhance your AI knowledge.
Get a Lay of the Land: Free Primers
Many clinical societies and interest groups have put out AI primers, an easy way to get a broad overview of the technology. The following were recommended or developed by the experts we spoke to, and all are free:
- The American Medical Association’s (AMA’s) framework for advancing healthcare AI lays out actionable guidance. Ask three key questions, the AMA recommends: Does it work? Does it work for my patients? Does it improve health outcomes?
- The Coalition for Health AI’s Blueprint for Trustworthy AI Implementation Guidance and Assurance for Healthcare provides a high-level summary of how to evaluate AI in healthcare, plus steps for implementing it. AI systems should be useful, safe, accountable, explainable, fair, and secure, the report asserted.
- The National Academy of Medicine’s draft code of conduct for AI in healthcare proposes core principles and commitments. These “reflect simple guideposts to guide and gauge behavior in a complex system and provide a starting point for real-time decision-making,” the report said.
- Health AI Partnership — a collaboration of Duke Health and Microsoft — outlines eight key decision points to consider at any stage of AI implementation, whether you’re still planning how to use it or you’ve started but want to improve it. The site also provides a breakdown of standards by regulatory agencies, organizations, and oversight bodies — so you can make sure your practices align with their guidance.
Make the Most of Conferences
Next time you’re at a conference, check the agenda for sessions on AI. “For someone who’s interested in this, I would be looking for content in my next national meeting because, undoubtedly, it’s going to be there,” said Dr. DeCamp. In a fast-moving field like AI, it’s a great way to get fresh, up-to-the-moment insights.
Listen to This Podcast
The New England Journal of Medicine’s free monthly podcast AI Grand Rounds is made for researchers and clinicians. Available on Apple, Spotify, and YouTube, the pod is good for “someone who’s looking to see both where the field is going [and to hear] a retrospective on big-name papers,” said Dr. Parikh . Episodes run for about an hour.
To learn about the challenges of applying AI to biology: Listen to Daphne Koller, PhD, founder of AI-driven drug discovery and development company insitro. For insights on the potential of AI in medicine, tune into the one with Eric Horvitz, MD, PhD, Microsoft’s chief scientific officer.
Consider a Class
Look for courses that focus on AI applications in clinical practice rather than a deep dive into theory. (You need to understand how these tools will influence your work, not the intricacies of large language model development.) Be wary of corporate-funded training that centers on one product , which could present conflicts of interest, said Dr. DeCamp. See the chart for courses that meet these criteria.
A version of this article appeared on Medscape.com.
Diacerein, Resveratrol, Botulinum Toxin Disappoint in Knee Osteoarthritis
VIENNA — Data do not back the use of diacerein or resveratrol for managing the pain of knee osteoarthritis (OA), according to the results of two well-performed, multicenter, double-blind, randomized controlled clinical trials.
During the News in Therapies session at the OARSI 2024 World Congress, the null findings of the DICKENS study and ARTHROL trial were presented alongside a reappraisal of the possible role of botulinum toxin.
DICKENS Study of Diacerein
“The role of diacerein in the treatment of OA is controversial,” acknowledged Dawn Aitken, PhD, associate professor at the University of Tasmania in Hobart, Tasmania, Australia. “There are only a few acceptable quality trials to date, and the results are inconsistent,” Dr. Aitken added.
Indeed, a Cochrane review performed in 2014 had concluded that there was “low-quality evidence that diacerein had a small beneficial effect on pain,” she said. The reported overall effect size on a 100-mm visual analog scale, based on a meta-analysis of 10 trials, has been just −8.65 mm, equating to just a 9% pain reduction.
At the time the DICKENS study was conceived, diacerein was recommended by a number of international guidelines for the management of hip and knee OA, although further, higher-quality studies were needed.
Diacerein blocks interleukin-1 beta, which is one of the key inflammatory markers of OA, so Dr. Aitken and collaborators postulated that perhaps it would work better if used in patients with an inflammatory phenotype.
They set about to test their hypothesis by recruiting 260 individuals with knee OA and MRI-detected effusion synovitis. The participants were then randomly allocated to treatment with either diacerein or a matching placebo for 24 weeks.
Individuals in the diacerein group were treated with an oral dose of 50 mg once daily for the first 2 weeks. If tolerated, the dose was increased to 50 mg twice daily.
No significant improvement in the primary endpoint of knee pain was seen comparing diacerein with placebo, with mean values of 53.2 mm and 56.4 mm, respectively, at 24 weeks using a 0-100 mm visual analog scale where 0 represented no pain and 100 represented the worst pain. It followed that there was no significant difference in the change from baseline to week 24 (−19.9 mm vs −18.6 mm; P = .77).
There was also no difference in the secondary endpoints, which included Western Ontario and McMaster Universities Arthritis Index pain, function, and stiffness. In fact, placebo-treated patients appeared to do better in terms of resolution of effusion synovitis as measured by a repeat MRI and quality of life, Dr. Aitken reported.
“These findings do not support the use of diacerein in treating patients with knee OA and effusion synovitis,” Dr. Aitken concluded.
ARTHROL Trial of Resveratrol
Similarly, negative results were reported for resveratrol from the ARTHROL trial, with 55% of the resveratrol- and 55% of placebo-treated individuals achieving a 20% reduction in knee pain intensity at 3 months. The actual change in knee pain from baseline to 3 months was −15.7 for resveratrol and −15.2 for placebo on a numerical rating scale that went from 0 (no pain) up to 100 (worst pain).
Resveratrol is found naturally in grapes, peanuts, pine cones, and Chinese knotweed, and there is a growing body of evidence that it may have pleiotropic effects, said investigator Christelle Nguyen, PhD, MD, a professor of physical and rehabilitation medicine at Université Paris Cité, Paris, France.
It’s available in a powder form over the counter as a treatment for multiple ailments, but more recently, became available as an oral formulation. Dr. Nguyen and colleagues wanted to know if this would make a difference to OA knee pain when added to usual care.
A double-blind, multicenter, placebo-controlled randomized trial was therefore conducted that involved 142 people with knee OA who had been experiencing knee pain for at least 1 month. The participants were equally randomly allocated to receive either oral resveratrol given as two caplets of 20 mg twice daily for the first week, then once daily for a total of 6 months, or a matched placebo.
There was also no effect of resveratrol vs placebo on a host of secondary outcomes measured at 3 and 6 months.
The interpretation is that oral resveratrol may not be effective in this indication or have a biologic effect on the pain pathway, Dr. Nguyen said.
“Our findings do not support the use of [trans-resveratrol] supplementation in this patented formulation for reducing knee pain in adults with painful knee OA,” she concluded.
Botulinum Toxin: Over But Not Out?
Dr. Nguyen separately reported data from a new systematic review and meta-analysis on the use of intra-articular (IA) botulinum toxin type A (BoNT-A) for knee OA pain.
Seven of the 14 randomized controlled trials included in the meta-analysis had looked specifically at knee OA outcomes in the short, intermediate, and long term.
Results showed a nonsignificant trend favoring BoNT-A use, with the standard mean difference in pain of 0.35 (−0.82; 0.12), −0.27 (−0.61; 0.08), and −0.43 (−1.12; 0.26) for short-, intermediate-, and long-term use, respectively.
In contrast, pain reductions were seen with BoNT-A in three trials that included people with OA of the shoulder or base of the thumb. This begs the question as to whether botulinum toxin may still have a role to play, Dr. Nguyen said in an interview.
“It seems like there may be a positive effect for the shoulder joint and base of the thumb,” she told this news organization.
“So, basically, we found differences between large and small to intermediate joints,” Dr. Nguyen added. “It questions the dilution of botulinum toxin into the joint. If it’s a big joint, maybe the dilution is too high,” she suggested.
This hypothesis will be tested in the upcoming RHIBOT II trial that will begin recruitment later this year. This is a follow-on from the RHIBOT trial that was published in The Lancet Rheumatology 2 years ago.
Meanwhile, the use of botulinum toxin is off-label, Dr. Nguyen said. “We use it in our clinics only when first-line treatment had failed for base of thumb OA.” It’s not offered as a stand-alone intervention, and the IA injections need to be given by someone with experience, she said.
Methodologically Sound Studies
Commenting on the studies, Nancy E. Lane, MD, said: “There have been small botulinum studies before but not powered enough so that you could confirm or refute hypotheses.”
Dr. Lane, endowed professor of medicine, rheumatology, and aging research and director for the Center for Musculoskeletal Health at the University of California Davis School of Medicine, Sacramento, California, added: “Similarly for resveratrol, there have been lots of studies.”
Moreover, Dr. Lane observed that the studies were “really well-designed. They were well-powered. The subjects were selected in such a way that was good rigor in the methodologic design, and there were enough people in the studies so that you could really believe the results.”
The take-home is probably that these approaches do not work, Dr. Lane said, “at least when you apply them to moderate-severe knee OA patients, they don’t seem to make a difference.”
The congress was sponsored by the Osteoarthritis Research Society International.
The DICKENS study of diacerein was an investigator-initiated trial that was funded by the National Health and Medical Research Council of Australia. TRB Chemedica International S.A. provided diacerein free of charge for the trial but was not involved in the implementation or data analysis. Dr. Aitken had no conflicts of interest to disclose.
The ARTHROL trial of oral resveratrol was funded by the French Ministry of Health and Solidarity (Ministré des Solidarités et de la Santé). Yvery Laboratory provided the resveratrol caplet and matching placebo free of charge. Dr. Nguyen has financial relationships with Actelion, Grünenthal, Ipsen, Lilly, Meda, Merz, Novartis, Preciphar, Sandoz, Takeda, Thuasne, and UCB.
Dr. Lane had no relevant conflicts of interest to declare.
A version of this article appeared on Medscape.com.
VIENNA — Data do not back the use of diacerein or resveratrol for managing the pain of knee osteoarthritis (OA), according to the results of two well-performed, multicenter, double-blind, randomized controlled clinical trials.
During the News in Therapies session at the OARSI 2024 World Congress, the null findings of the DICKENS study and ARTHROL trial were presented alongside a reappraisal of the possible role of botulinum toxin.
DICKENS Study of Diacerein
“The role of diacerein in the treatment of OA is controversial,” acknowledged Dawn Aitken, PhD, associate professor at the University of Tasmania in Hobart, Tasmania, Australia. “There are only a few acceptable quality trials to date, and the results are inconsistent,” Dr. Aitken added.
Indeed, a Cochrane review performed in 2014 had concluded that there was “low-quality evidence that diacerein had a small beneficial effect on pain,” she said. The reported overall effect size on a 100-mm visual analog scale, based on a meta-analysis of 10 trials, has been just −8.65 mm, equating to just a 9% pain reduction.
At the time the DICKENS study was conceived, diacerein was recommended by a number of international guidelines for the management of hip and knee OA, although further, higher-quality studies were needed.
Diacerein blocks interleukin-1 beta, which is one of the key inflammatory markers of OA, so Dr. Aitken and collaborators postulated that perhaps it would work better if used in patients with an inflammatory phenotype.
They set about to test their hypothesis by recruiting 260 individuals with knee OA and MRI-detected effusion synovitis. The participants were then randomly allocated to treatment with either diacerein or a matching placebo for 24 weeks.
Individuals in the diacerein group were treated with an oral dose of 50 mg once daily for the first 2 weeks. If tolerated, the dose was increased to 50 mg twice daily.
No significant improvement in the primary endpoint of knee pain was seen comparing diacerein with placebo, with mean values of 53.2 mm and 56.4 mm, respectively, at 24 weeks using a 0-100 mm visual analog scale where 0 represented no pain and 100 represented the worst pain. It followed that there was no significant difference in the change from baseline to week 24 (−19.9 mm vs −18.6 mm; P = .77).
There was also no difference in the secondary endpoints, which included Western Ontario and McMaster Universities Arthritis Index pain, function, and stiffness. In fact, placebo-treated patients appeared to do better in terms of resolution of effusion synovitis as measured by a repeat MRI and quality of life, Dr. Aitken reported.
“These findings do not support the use of diacerein in treating patients with knee OA and effusion synovitis,” Dr. Aitken concluded.
ARTHROL Trial of Resveratrol
Similarly, negative results were reported for resveratrol from the ARTHROL trial, with 55% of the resveratrol- and 55% of placebo-treated individuals achieving a 20% reduction in knee pain intensity at 3 months. The actual change in knee pain from baseline to 3 months was −15.7 for resveratrol and −15.2 for placebo on a numerical rating scale that went from 0 (no pain) up to 100 (worst pain).
Resveratrol is found naturally in grapes, peanuts, pine cones, and Chinese knotweed, and there is a growing body of evidence that it may have pleiotropic effects, said investigator Christelle Nguyen, PhD, MD, a professor of physical and rehabilitation medicine at Université Paris Cité, Paris, France.
It’s available in a powder form over the counter as a treatment for multiple ailments, but more recently, became available as an oral formulation. Dr. Nguyen and colleagues wanted to know if this would make a difference to OA knee pain when added to usual care.
A double-blind, multicenter, placebo-controlled randomized trial was therefore conducted that involved 142 people with knee OA who had been experiencing knee pain for at least 1 month. The participants were equally randomly allocated to receive either oral resveratrol given as two caplets of 20 mg twice daily for the first week, then once daily for a total of 6 months, or a matched placebo.
There was also no effect of resveratrol vs placebo on a host of secondary outcomes measured at 3 and 6 months.
The interpretation is that oral resveratrol may not be effective in this indication or have a biologic effect on the pain pathway, Dr. Nguyen said.
“Our findings do not support the use of [trans-resveratrol] supplementation in this patented formulation for reducing knee pain in adults with painful knee OA,” she concluded.
Botulinum Toxin: Over But Not Out?
Dr. Nguyen separately reported data from a new systematic review and meta-analysis on the use of intra-articular (IA) botulinum toxin type A (BoNT-A) for knee OA pain.
Seven of the 14 randomized controlled trials included in the meta-analysis had looked specifically at knee OA outcomes in the short, intermediate, and long term.
Results showed a nonsignificant trend favoring BoNT-A use, with the standard mean difference in pain of 0.35 (−0.82; 0.12), −0.27 (−0.61; 0.08), and −0.43 (−1.12; 0.26) for short-, intermediate-, and long-term use, respectively.
In contrast, pain reductions were seen with BoNT-A in three trials that included people with OA of the shoulder or base of the thumb. This begs the question as to whether botulinum toxin may still have a role to play, Dr. Nguyen said in an interview.
“It seems like there may be a positive effect for the shoulder joint and base of the thumb,” she told this news organization.
“So, basically, we found differences between large and small to intermediate joints,” Dr. Nguyen added. “It questions the dilution of botulinum toxin into the joint. If it’s a big joint, maybe the dilution is too high,” she suggested.
This hypothesis will be tested in the upcoming RHIBOT II trial that will begin recruitment later this year. This is a follow-on from the RHIBOT trial that was published in The Lancet Rheumatology 2 years ago.
Meanwhile, the use of botulinum toxin is off-label, Dr. Nguyen said. “We use it in our clinics only when first-line treatment had failed for base of thumb OA.” It’s not offered as a stand-alone intervention, and the IA injections need to be given by someone with experience, she said.
Methodologically Sound Studies
Commenting on the studies, Nancy E. Lane, MD, said: “There have been small botulinum studies before but not powered enough so that you could confirm or refute hypotheses.”
Dr. Lane, endowed professor of medicine, rheumatology, and aging research and director for the Center for Musculoskeletal Health at the University of California Davis School of Medicine, Sacramento, California, added: “Similarly for resveratrol, there have been lots of studies.”
Moreover, Dr. Lane observed that the studies were “really well-designed. They were well-powered. The subjects were selected in such a way that was good rigor in the methodologic design, and there were enough people in the studies so that you could really believe the results.”
The take-home is probably that these approaches do not work, Dr. Lane said, “at least when you apply them to moderate-severe knee OA patients, they don’t seem to make a difference.”
The congress was sponsored by the Osteoarthritis Research Society International.
The DICKENS study of diacerein was an investigator-initiated trial that was funded by the National Health and Medical Research Council of Australia. TRB Chemedica International S.A. provided diacerein free of charge for the trial but was not involved in the implementation or data analysis. Dr. Aitken had no conflicts of interest to disclose.
The ARTHROL trial of oral resveratrol was funded by the French Ministry of Health and Solidarity (Ministré des Solidarités et de la Santé). Yvery Laboratory provided the resveratrol caplet and matching placebo free of charge. Dr. Nguyen has financial relationships with Actelion, Grünenthal, Ipsen, Lilly, Meda, Merz, Novartis, Preciphar, Sandoz, Takeda, Thuasne, and UCB.
Dr. Lane had no relevant conflicts of interest to declare.
A version of this article appeared on Medscape.com.
VIENNA — Data do not back the use of diacerein or resveratrol for managing the pain of knee osteoarthritis (OA), according to the results of two well-performed, multicenter, double-blind, randomized controlled clinical trials.
During the News in Therapies session at the OARSI 2024 World Congress, the null findings of the DICKENS study and ARTHROL trial were presented alongside a reappraisal of the possible role of botulinum toxin.
DICKENS Study of Diacerein
“The role of diacerein in the treatment of OA is controversial,” acknowledged Dawn Aitken, PhD, associate professor at the University of Tasmania in Hobart, Tasmania, Australia. “There are only a few acceptable quality trials to date, and the results are inconsistent,” Dr. Aitken added.
Indeed, a Cochrane review performed in 2014 had concluded that there was “low-quality evidence that diacerein had a small beneficial effect on pain,” she said. The reported overall effect size on a 100-mm visual analog scale, based on a meta-analysis of 10 trials, has been just −8.65 mm, equating to just a 9% pain reduction.
At the time the DICKENS study was conceived, diacerein was recommended by a number of international guidelines for the management of hip and knee OA, although further, higher-quality studies were needed.
Diacerein blocks interleukin-1 beta, which is one of the key inflammatory markers of OA, so Dr. Aitken and collaborators postulated that perhaps it would work better if used in patients with an inflammatory phenotype.
They set about to test their hypothesis by recruiting 260 individuals with knee OA and MRI-detected effusion synovitis. The participants were then randomly allocated to treatment with either diacerein or a matching placebo for 24 weeks.
Individuals in the diacerein group were treated with an oral dose of 50 mg once daily for the first 2 weeks. If tolerated, the dose was increased to 50 mg twice daily.
No significant improvement in the primary endpoint of knee pain was seen comparing diacerein with placebo, with mean values of 53.2 mm and 56.4 mm, respectively, at 24 weeks using a 0-100 mm visual analog scale where 0 represented no pain and 100 represented the worst pain. It followed that there was no significant difference in the change from baseline to week 24 (−19.9 mm vs −18.6 mm; P = .77).
There was also no difference in the secondary endpoints, which included Western Ontario and McMaster Universities Arthritis Index pain, function, and stiffness. In fact, placebo-treated patients appeared to do better in terms of resolution of effusion synovitis as measured by a repeat MRI and quality of life, Dr. Aitken reported.
“These findings do not support the use of diacerein in treating patients with knee OA and effusion synovitis,” Dr. Aitken concluded.
ARTHROL Trial of Resveratrol
Similarly, negative results were reported for resveratrol from the ARTHROL trial, with 55% of the resveratrol- and 55% of placebo-treated individuals achieving a 20% reduction in knee pain intensity at 3 months. The actual change in knee pain from baseline to 3 months was −15.7 for resveratrol and −15.2 for placebo on a numerical rating scale that went from 0 (no pain) up to 100 (worst pain).
Resveratrol is found naturally in grapes, peanuts, pine cones, and Chinese knotweed, and there is a growing body of evidence that it may have pleiotropic effects, said investigator Christelle Nguyen, PhD, MD, a professor of physical and rehabilitation medicine at Université Paris Cité, Paris, France.
It’s available in a powder form over the counter as a treatment for multiple ailments, but more recently, became available as an oral formulation. Dr. Nguyen and colleagues wanted to know if this would make a difference to OA knee pain when added to usual care.
A double-blind, multicenter, placebo-controlled randomized trial was therefore conducted that involved 142 people with knee OA who had been experiencing knee pain for at least 1 month. The participants were equally randomly allocated to receive either oral resveratrol given as two caplets of 20 mg twice daily for the first week, then once daily for a total of 6 months, or a matched placebo.
There was also no effect of resveratrol vs placebo on a host of secondary outcomes measured at 3 and 6 months.
The interpretation is that oral resveratrol may not be effective in this indication or have a biologic effect on the pain pathway, Dr. Nguyen said.
“Our findings do not support the use of [trans-resveratrol] supplementation in this patented formulation for reducing knee pain in adults with painful knee OA,” she concluded.
Botulinum Toxin: Over But Not Out?
Dr. Nguyen separately reported data from a new systematic review and meta-analysis on the use of intra-articular (IA) botulinum toxin type A (BoNT-A) for knee OA pain.
Seven of the 14 randomized controlled trials included in the meta-analysis had looked specifically at knee OA outcomes in the short, intermediate, and long term.
Results showed a nonsignificant trend favoring BoNT-A use, with the standard mean difference in pain of 0.35 (−0.82; 0.12), −0.27 (−0.61; 0.08), and −0.43 (−1.12; 0.26) for short-, intermediate-, and long-term use, respectively.
In contrast, pain reductions were seen with BoNT-A in three trials that included people with OA of the shoulder or base of the thumb. This begs the question as to whether botulinum toxin may still have a role to play, Dr. Nguyen said in an interview.
“It seems like there may be a positive effect for the shoulder joint and base of the thumb,” she told this news organization.
“So, basically, we found differences between large and small to intermediate joints,” Dr. Nguyen added. “It questions the dilution of botulinum toxin into the joint. If it’s a big joint, maybe the dilution is too high,” she suggested.
This hypothesis will be tested in the upcoming RHIBOT II trial that will begin recruitment later this year. This is a follow-on from the RHIBOT trial that was published in The Lancet Rheumatology 2 years ago.
Meanwhile, the use of botulinum toxin is off-label, Dr. Nguyen said. “We use it in our clinics only when first-line treatment had failed for base of thumb OA.” It’s not offered as a stand-alone intervention, and the IA injections need to be given by someone with experience, she said.
Methodologically Sound Studies
Commenting on the studies, Nancy E. Lane, MD, said: “There have been small botulinum studies before but not powered enough so that you could confirm or refute hypotheses.”
Dr. Lane, endowed professor of medicine, rheumatology, and aging research and director for the Center for Musculoskeletal Health at the University of California Davis School of Medicine, Sacramento, California, added: “Similarly for resveratrol, there have been lots of studies.”
Moreover, Dr. Lane observed that the studies were “really well-designed. They were well-powered. The subjects were selected in such a way that was good rigor in the methodologic design, and there were enough people in the studies so that you could really believe the results.”
The take-home is probably that these approaches do not work, Dr. Lane said, “at least when you apply them to moderate-severe knee OA patients, they don’t seem to make a difference.”
The congress was sponsored by the Osteoarthritis Research Society International.
The DICKENS study of diacerein was an investigator-initiated trial that was funded by the National Health and Medical Research Council of Australia. TRB Chemedica International S.A. provided diacerein free of charge for the trial but was not involved in the implementation or data analysis. Dr. Aitken had no conflicts of interest to disclose.
The ARTHROL trial of oral resveratrol was funded by the French Ministry of Health and Solidarity (Ministré des Solidarités et de la Santé). Yvery Laboratory provided the resveratrol caplet and matching placebo free of charge. Dr. Nguyen has financial relationships with Actelion, Grünenthal, Ipsen, Lilly, Meda, Merz, Novartis, Preciphar, Sandoz, Takeda, Thuasne, and UCB.
Dr. Lane had no relevant conflicts of interest to declare.
A version of this article appeared on Medscape.com.
FROM OARSI 2024