User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Better to binge drink than regularly tipple, suggests GI cancer study
When weekly levels are similar
Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts – compared with higher amounts or binge drinking, given similar weekly levels.
“The novel finding of the current study is that frequent drinking may be more dangerous than binge drinking with regard to GI cancers. Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts -- compared with higher amounts or binge drinking, given similar weekly levels.” first author Jung Eun Yook, MD, of Seoul (South Korea) National University Hospital, and colleagues reported in an article published Aug. 18, 2021, in JAMA Network Open (doi: 10.1001/jamanetworkopen.2021.20382).
“This finding suggests that repeated alcohol consumption events even at lower amounts of alcohol may have a greater carcinogenic effect on GI organs than the consumption of larger amounts of alcohol at a lower frequency,” the investigators wrote.
A possible reason behind the difference in risk may be that the chronic “carcinogenic insult” from regular alcohol use may promote cancer development, whereas less frequent, episodic alcohol exposures may allow physiologic recovery, said the authors.
The results are from a population-based study that involved 11,737,467 participants in the Korean National Health System database who did not have cancer and who took part in a national screening program between January 2009 and December 2010.
They were followed from the year after their screening until either they had received a diagnosis of a GI cancer, death occurred, or the end of December 2017.
During a median follow-up of 6.4 years, 319,202 (2.7%) of those in the study developed a GI cancer.
The increase in the risk associated with alcohol consumption was dose dependent.
Compared with those who did not consume alcohol, the risk of developing GI cancer was higher for mild drinkers (adjusted hazard ratio, 1.04; 95% confidence interval, 1.03-1.05), moderate drinkers (aHR, 1.14; 95% CI, 1.12-1.15), and heavy drinkers (aHR, 1.28; 95% CI, 1.26-1.29), after adjusting for age, sex, income, smoking status with intensity, regular exercise, body mass index, diabetes, hypertension, and dyslipidemia.
There was a linear association between the frequency of drinking and GI cancer risk, with an aHR of 1.39 for individuals who reported drinking every day (95% CI, 1.36-1.41). The risk for GI cancer increased with consumption of five to seven units per occasion (aHR, 1.15). Notably, there were no similar increases with higher intake, including intake of 8-14 units per occasion (aHR, 1.11; 95% CI, 1.09-1.12), and even up to more than 14 units per occasion (aHR, 1.11; 95%CI, 1.08-1.14), in comparison with an intake of 5-7 units per occasion.
“Given similar weekly alcohol consumption levels, the risk of GI cancer increased with a higher frequency of drinking and decreased with a higher amount per occasion,” the authors write.
“Most previous studies just assess alcohol consumption as a total amount, [such as] drinks per occasion times occasion per week equals drinks per week [and] grams per week,” coauthor Dong Wook Shin, MD, DrPH, Samsung Medical Center, Sungkyunkwan University, Seoul, South Korea, said in an interview.
“But it was not known whether frequent drinking with small amount is more harmful than binge drinking, given a similar level of total drinking,” Dr. Shin said.
The increased risk associated with frequent drinking was generally similar with respect to esophageal, gastric, colorectal, biliary, and pancreatic cancer.
An exception was liver cancer, which showed a slightly decreased risk among mild drinkers (aHR, 0.91; 95% CI, 0.89-0.93).
Of note, the association between alcohol intake and the incidence of GI cancer was lower among women than men in terms of weekly consumption, frequency, and amount of alcohol consumed per occasion.
The associations between drinking and cancer type in terms of esophageal and liver cancers were similar between men and women. However, the alcohol-related risk for colorectal, biliary, and pancreatic cancers was less prominent for women.
Possible mechanisms related to regular drinking
A factor that might account for the increase in GI cancer risk with frequent drinking is that regular alcohol consumption “promotes the accumulation of cell divisions in the stem cells that maintain tissues in homeostasis,” the authors explained.
Another possible explanation is that long-term alcohol exposure may promote carcinogenesis, whereas less frequent exposure might allow “physiological homeostasis,” the authors wrote, adding that in vivo experiments have shown that duration and dose of alcohol exposure have been linked to cancer development.
Importantly, the findings support the importance of reducing the frequency of alcohol use to prevent cancer, the authors noted.
“Alcohol users who have a glass of wine or beer during dinner every day may develop more cancer than people who occasionally consume several drinks,” they cautioned.
Genetics, self-reporting considerations
In a related commentary, John D. Potter, MBBS, PhD, of the Research Center for Hauora and Health, Massey University, Wellington, New Zealand, noted that, in addition to supporting the known link between alcohol and cancers of the esophagus, colorectum, and liver, the study “strengthens evidence for a role of alcohol in stomach, biliary tract, and pancreas cancers.”
In comparison with nondrinkers, those who reported heavy drinking were much more likely to be smokers (51.6% vs. 9.0%); however, the study adjusted for smoking.
“Because the researchers were able to control for tobacco, this last finding [regarding the association with cancers of the stomach, biliary tract, and pancreas] is particularly informative,” Dr. Potter noted.
An important caveat is that more than a quarter of the Korean population is known to have an inactive form of the aldehyde dehydrogenase gene (ALDH2), which could have effects on alcohol metabolism as well as the risk for cancer, Dr. Potter wrote.
“This common polymorphism in ALDH2 (ALDH2 rs671 [c.1510G>A (Glu504Lys)]) has paradoxical effects,” he wrote.
“It increases the level of acetaldehyde in the blood of drinkers, which in turn increases the risk of cancer because acetaldehyde is a key player in the carcinogenicity of alcoholic beverages,” Dr. Potter explained. “On the other hand, the accumulation of acetaldehyde and the resultant flushing response are sufficiently unpleasant that they tend to reduce alcohol consumption among those with the Lys allele.”
The study results may therefore not be generalizable to a population in which the distribution of the variation in the ALDH2 enzyme differs, Dr. Potter added.
The lower prevalence of the inactive form (in North America, for instance) would mean that this lower prevalence was not a constraint on individuals’ drinking behavior as it is for some in Korea, Dr. Potter explained.
He noted another consideration: the underreporting of alcohol use is a well-known limitation of studies involving the assessment of alcohol consumption.
Dr. Shin agreed that underreporting is a limitation.
“People tend to underestimate their alcohol use,” Dr. Shin said in an interview.
However, he noted that “our study participants are health-screening participants aged 40 years and older, [and] people who participate in health screening tend to have higher awareness and better health behavior than nonparticipants.”
The authors and Dr. Potter disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
When weekly levels are similar
When weekly levels are similar
Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts – compared with higher amounts or binge drinking, given similar weekly levels.
“The novel finding of the current study is that frequent drinking may be more dangerous than binge drinking with regard to GI cancers. Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts -- compared with higher amounts or binge drinking, given similar weekly levels.” first author Jung Eun Yook, MD, of Seoul (South Korea) National University Hospital, and colleagues reported in an article published Aug. 18, 2021, in JAMA Network Open (doi: 10.1001/jamanetworkopen.2021.20382).
“This finding suggests that repeated alcohol consumption events even at lower amounts of alcohol may have a greater carcinogenic effect on GI organs than the consumption of larger amounts of alcohol at a lower frequency,” the investigators wrote.
A possible reason behind the difference in risk may be that the chronic “carcinogenic insult” from regular alcohol use may promote cancer development, whereas less frequent, episodic alcohol exposures may allow physiologic recovery, said the authors.
The results are from a population-based study that involved 11,737,467 participants in the Korean National Health System database who did not have cancer and who took part in a national screening program between January 2009 and December 2010.
They were followed from the year after their screening until either they had received a diagnosis of a GI cancer, death occurred, or the end of December 2017.
During a median follow-up of 6.4 years, 319,202 (2.7%) of those in the study developed a GI cancer.
The increase in the risk associated with alcohol consumption was dose dependent.
Compared with those who did not consume alcohol, the risk of developing GI cancer was higher for mild drinkers (adjusted hazard ratio, 1.04; 95% confidence interval, 1.03-1.05), moderate drinkers (aHR, 1.14; 95% CI, 1.12-1.15), and heavy drinkers (aHR, 1.28; 95% CI, 1.26-1.29), after adjusting for age, sex, income, smoking status with intensity, regular exercise, body mass index, diabetes, hypertension, and dyslipidemia.
There was a linear association between the frequency of drinking and GI cancer risk, with an aHR of 1.39 for individuals who reported drinking every day (95% CI, 1.36-1.41). The risk for GI cancer increased with consumption of five to seven units per occasion (aHR, 1.15). Notably, there were no similar increases with higher intake, including intake of 8-14 units per occasion (aHR, 1.11; 95% CI, 1.09-1.12), and even up to more than 14 units per occasion (aHR, 1.11; 95%CI, 1.08-1.14), in comparison with an intake of 5-7 units per occasion.
“Given similar weekly alcohol consumption levels, the risk of GI cancer increased with a higher frequency of drinking and decreased with a higher amount per occasion,” the authors write.
“Most previous studies just assess alcohol consumption as a total amount, [such as] drinks per occasion times occasion per week equals drinks per week [and] grams per week,” coauthor Dong Wook Shin, MD, DrPH, Samsung Medical Center, Sungkyunkwan University, Seoul, South Korea, said in an interview.
“But it was not known whether frequent drinking with small amount is more harmful than binge drinking, given a similar level of total drinking,” Dr. Shin said.
The increased risk associated with frequent drinking was generally similar with respect to esophageal, gastric, colorectal, biliary, and pancreatic cancer.
An exception was liver cancer, which showed a slightly decreased risk among mild drinkers (aHR, 0.91; 95% CI, 0.89-0.93).
Of note, the association between alcohol intake and the incidence of GI cancer was lower among women than men in terms of weekly consumption, frequency, and amount of alcohol consumed per occasion.
The associations between drinking and cancer type in terms of esophageal and liver cancers were similar between men and women. However, the alcohol-related risk for colorectal, biliary, and pancreatic cancers was less prominent for women.
Possible mechanisms related to regular drinking
A factor that might account for the increase in GI cancer risk with frequent drinking is that regular alcohol consumption “promotes the accumulation of cell divisions in the stem cells that maintain tissues in homeostasis,” the authors explained.
Another possible explanation is that long-term alcohol exposure may promote carcinogenesis, whereas less frequent exposure might allow “physiological homeostasis,” the authors wrote, adding that in vivo experiments have shown that duration and dose of alcohol exposure have been linked to cancer development.
Importantly, the findings support the importance of reducing the frequency of alcohol use to prevent cancer, the authors noted.
“Alcohol users who have a glass of wine or beer during dinner every day may develop more cancer than people who occasionally consume several drinks,” they cautioned.
Genetics, self-reporting considerations
In a related commentary, John D. Potter, MBBS, PhD, of the Research Center for Hauora and Health, Massey University, Wellington, New Zealand, noted that, in addition to supporting the known link between alcohol and cancers of the esophagus, colorectum, and liver, the study “strengthens evidence for a role of alcohol in stomach, biliary tract, and pancreas cancers.”
In comparison with nondrinkers, those who reported heavy drinking were much more likely to be smokers (51.6% vs. 9.0%); however, the study adjusted for smoking.
“Because the researchers were able to control for tobacco, this last finding [regarding the association with cancers of the stomach, biliary tract, and pancreas] is particularly informative,” Dr. Potter noted.
An important caveat is that more than a quarter of the Korean population is known to have an inactive form of the aldehyde dehydrogenase gene (ALDH2), which could have effects on alcohol metabolism as well as the risk for cancer, Dr. Potter wrote.
“This common polymorphism in ALDH2 (ALDH2 rs671 [c.1510G>A (Glu504Lys)]) has paradoxical effects,” he wrote.
“It increases the level of acetaldehyde in the blood of drinkers, which in turn increases the risk of cancer because acetaldehyde is a key player in the carcinogenicity of alcoholic beverages,” Dr. Potter explained. “On the other hand, the accumulation of acetaldehyde and the resultant flushing response are sufficiently unpleasant that they tend to reduce alcohol consumption among those with the Lys allele.”
The study results may therefore not be generalizable to a population in which the distribution of the variation in the ALDH2 enzyme differs, Dr. Potter added.
The lower prevalence of the inactive form (in North America, for instance) would mean that this lower prevalence was not a constraint on individuals’ drinking behavior as it is for some in Korea, Dr. Potter explained.
He noted another consideration: the underreporting of alcohol use is a well-known limitation of studies involving the assessment of alcohol consumption.
Dr. Shin agreed that underreporting is a limitation.
“People tend to underestimate their alcohol use,” Dr. Shin said in an interview.
However, he noted that “our study participants are health-screening participants aged 40 years and older, [and] people who participate in health screening tend to have higher awareness and better health behavior than nonparticipants.”
The authors and Dr. Potter disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts – compared with higher amounts or binge drinking, given similar weekly levels.
“The novel finding of the current study is that frequent drinking may be more dangerous than binge drinking with regard to GI cancers. Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts -- compared with higher amounts or binge drinking, given similar weekly levels.” first author Jung Eun Yook, MD, of Seoul (South Korea) National University Hospital, and colleagues reported in an article published Aug. 18, 2021, in JAMA Network Open (doi: 10.1001/jamanetworkopen.2021.20382).
“This finding suggests that repeated alcohol consumption events even at lower amounts of alcohol may have a greater carcinogenic effect on GI organs than the consumption of larger amounts of alcohol at a lower frequency,” the investigators wrote.
A possible reason behind the difference in risk may be that the chronic “carcinogenic insult” from regular alcohol use may promote cancer development, whereas less frequent, episodic alcohol exposures may allow physiologic recovery, said the authors.
The results are from a population-based study that involved 11,737,467 participants in the Korean National Health System database who did not have cancer and who took part in a national screening program between January 2009 and December 2010.
They were followed from the year after their screening until either they had received a diagnosis of a GI cancer, death occurred, or the end of December 2017.
During a median follow-up of 6.4 years, 319,202 (2.7%) of those in the study developed a GI cancer.
The increase in the risk associated with alcohol consumption was dose dependent.
Compared with those who did not consume alcohol, the risk of developing GI cancer was higher for mild drinkers (adjusted hazard ratio, 1.04; 95% confidence interval, 1.03-1.05), moderate drinkers (aHR, 1.14; 95% CI, 1.12-1.15), and heavy drinkers (aHR, 1.28; 95% CI, 1.26-1.29), after adjusting for age, sex, income, smoking status with intensity, regular exercise, body mass index, diabetes, hypertension, and dyslipidemia.
There was a linear association between the frequency of drinking and GI cancer risk, with an aHR of 1.39 for individuals who reported drinking every day (95% CI, 1.36-1.41). The risk for GI cancer increased with consumption of five to seven units per occasion (aHR, 1.15). Notably, there were no similar increases with higher intake, including intake of 8-14 units per occasion (aHR, 1.11; 95% CI, 1.09-1.12), and even up to more than 14 units per occasion (aHR, 1.11; 95%CI, 1.08-1.14), in comparison with an intake of 5-7 units per occasion.
“Given similar weekly alcohol consumption levels, the risk of GI cancer increased with a higher frequency of drinking and decreased with a higher amount per occasion,” the authors write.
“Most previous studies just assess alcohol consumption as a total amount, [such as] drinks per occasion times occasion per week equals drinks per week [and] grams per week,” coauthor Dong Wook Shin, MD, DrPH, Samsung Medical Center, Sungkyunkwan University, Seoul, South Korea, said in an interview.
“But it was not known whether frequent drinking with small amount is more harmful than binge drinking, given a similar level of total drinking,” Dr. Shin said.
The increased risk associated with frequent drinking was generally similar with respect to esophageal, gastric, colorectal, biliary, and pancreatic cancer.
An exception was liver cancer, which showed a slightly decreased risk among mild drinkers (aHR, 0.91; 95% CI, 0.89-0.93).
Of note, the association between alcohol intake and the incidence of GI cancer was lower among women than men in terms of weekly consumption, frequency, and amount of alcohol consumed per occasion.
The associations between drinking and cancer type in terms of esophageal and liver cancers were similar between men and women. However, the alcohol-related risk for colorectal, biliary, and pancreatic cancers was less prominent for women.
Possible mechanisms related to regular drinking
A factor that might account for the increase in GI cancer risk with frequent drinking is that regular alcohol consumption “promotes the accumulation of cell divisions in the stem cells that maintain tissues in homeostasis,” the authors explained.
Another possible explanation is that long-term alcohol exposure may promote carcinogenesis, whereas less frequent exposure might allow “physiological homeostasis,” the authors wrote, adding that in vivo experiments have shown that duration and dose of alcohol exposure have been linked to cancer development.
Importantly, the findings support the importance of reducing the frequency of alcohol use to prevent cancer, the authors noted.
“Alcohol users who have a glass of wine or beer during dinner every day may develop more cancer than people who occasionally consume several drinks,” they cautioned.
Genetics, self-reporting considerations
In a related commentary, John D. Potter, MBBS, PhD, of the Research Center for Hauora and Health, Massey University, Wellington, New Zealand, noted that, in addition to supporting the known link between alcohol and cancers of the esophagus, colorectum, and liver, the study “strengthens evidence for a role of alcohol in stomach, biliary tract, and pancreas cancers.”
In comparison with nondrinkers, those who reported heavy drinking were much more likely to be smokers (51.6% vs. 9.0%); however, the study adjusted for smoking.
“Because the researchers were able to control for tobacco, this last finding [regarding the association with cancers of the stomach, biliary tract, and pancreas] is particularly informative,” Dr. Potter noted.
An important caveat is that more than a quarter of the Korean population is known to have an inactive form of the aldehyde dehydrogenase gene (ALDH2), which could have effects on alcohol metabolism as well as the risk for cancer, Dr. Potter wrote.
“This common polymorphism in ALDH2 (ALDH2 rs671 [c.1510G>A (Glu504Lys)]) has paradoxical effects,” he wrote.
“It increases the level of acetaldehyde in the blood of drinkers, which in turn increases the risk of cancer because acetaldehyde is a key player in the carcinogenicity of alcoholic beverages,” Dr. Potter explained. “On the other hand, the accumulation of acetaldehyde and the resultant flushing response are sufficiently unpleasant that they tend to reduce alcohol consumption among those with the Lys allele.”
The study results may therefore not be generalizable to a population in which the distribution of the variation in the ALDH2 enzyme differs, Dr. Potter added.
The lower prevalence of the inactive form (in North America, for instance) would mean that this lower prevalence was not a constraint on individuals’ drinking behavior as it is for some in Korea, Dr. Potter explained.
He noted another consideration: the underreporting of alcohol use is a well-known limitation of studies involving the assessment of alcohol consumption.
Dr. Shin agreed that underreporting is a limitation.
“People tend to underestimate their alcohol use,” Dr. Shin said in an interview.
However, he noted that “our study participants are health-screening participants aged 40 years and older, [and] people who participate in health screening tend to have higher awareness and better health behavior than nonparticipants.”
The authors and Dr. Potter disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Study informs about risks of discontinuing meds in JIA
Flares are modest in preliminary data.
Many but not all children with juvenile idiopathic arthritis (JIA) can regain remission after stopping and then restarting treatment, according to preliminary data from the ongoing Recapture-JIA study that were presented in a symposium sponsored by the Rheumatology Research Foundation.
The aim of this study is to evaluate the risks of discontinuing treatment after a period when JIA has been well controlled. Such data are of increasing interest to parents now that many children with JIA are achieving sustained periods of remission, according to Sarah Ringold, MD, a pediatric rheumatologist and associate professor of pediatrics at Seattle Children’s Hospital.
In follow-up so far, “recapture rates range from 50% to 76%” depending on type of JIA, reported Dr. Ringold, who said that patients with systemic JIA have so far been the most likely to achieve a good response when treatment is restarted.
The study is being conducted through the Childhood Arthritis and Rheumatology Research Alliance, which has 71 participating centers and has accrued data on more than 10,000 children with rheumatic diseases. For the study, the researchers identified 384 children with JIA who were already enrolled in the CARRA registry and had discontinued medications and then subsequently restarted them, and they also enrolled a prospective cohort of patients new to the registry who presented with flare after discontinuing their medication. Dr. Ringold reported on 64 of the patients in the prospective cohort.
Median time to flare: 219 days
Of findings so far, disease recurrence after discontinuation has been generally characterized by flares “of moderate activity” several months to more than a year after treatment discontinuation, according to Dr. Ringold, who emphasized repeatedly that these data are preliminary. The median time to a flare after treatment discontinuation was approximately 7 months (219 days).
In the combined cohorts, the median age at onset of JIA was 4 years. The median age at time of discontinuation was 9 years. More than half (55%) were taking a conventional disease-modifying antirheumatic drug (DMARD) and 35% were taking a tumor necrosis factor inhibitor at the time that their therapy was discontinued.
Most JIA types are represented. The most common form is rheumatoid factor–negative oligoarticular JIA. The main outcome looked the rate of clinically inactive disease at 6 months in children who had discontinued therapy after a period of remission. They defined clinically inactive disease as a Physician’s Global Assessment of less than 1 and an active joint count of 0.
Systemic JIA recapture rate at 6 months: 76%
At the time of disease flare after treatment discontinuation across both the retrospective and prospective cohorts, the median clinical Juvenile Arthritis Disease Activity Score based on 10 joints (cJADAS10; score range of 0-30) was 3.5. The recapture rate to clinically inactive disease at 6 months was 76% in those with systemic JIA and 50% in those with rheumatoid factor–positive polyarticular JIA. Other subtypes fell within this range. Rates of inactive disease at 6 months according to cJADAS10 score were lower, ranging from 26% with enthesitis-related arthritis/juvenile psoriatic arthritis to 57% with systemic JIA.
About 40% of those who restarted on therapy after a flare took the same medication again. About one-third of patients were restarted on glucocorticoids, mostly involving injections to inflamed joints, and data are not yet in about whether these were restarted alone or with other drugs, according to Dr. Ringold.
The final analysis of this study will explore clinical and laboratory variables associated with disease recapture. In the prospective cohort, which did not reach its planned enrollment of 150 children because the COVID pandemic, a broad array of these variables was evaluated at baseline.
Numerous studies have already looked at predictors of sustained remission after stopping medications of JIA, according to Dr. Ringold, but she said that there is relatively little information about outcomes in children who stop medications, flare, and are retreated. Other experts agree.
“We know little about how successfully DMARDs can be discontinued and used again after a disease flare,” reported Jens Klotsche, MD, a researcher at the German Rheumatism Research Center, which is part of the Leibniz Institute in Berlin. Dr. Klotsche, who is an author of a recent study that found etanercept effective for retreatment when children with JIA had discontinued therapy, agreed that “data from large cohort studies are necessary to support the treatment decisions by clinicians, parents, and patients.”
JIA recurrence risk is unclear
In a systematic review published 2 years ago, rates of flare following discontinuation of treatment for JIA were relatively high, but there were some limitations to this analysis, according to the lead author, Olha Halyabar, MD, a pediatric rheumatologist at Boston Children’s Hospital.
“The data in our systematic review showed that overall quality of evidence was low, with large variations and sometimes very different conclusions,” Dr. Halyabar said in an interview. She believes that the data generated by the CARRA analysis will be valuable, particularly in evaluating outcomes across subtypes.
“Even though, at this point, [previously published] reports indicate overall high rates of recurrence (>50% for some JIA subtypes), there are some encouraging studies from early treat-to-target strategies,” she said, adding that large datasets like those from CARRA offer an opportunity to gather data likely to be clinically useful.
Dr. Ringold cautioned that there are some limitations to the CARRA analysis, including some missing data from the retrospective cohort. She also pointed out that patients have been assessed at routine clinical visits rather than at standardized intervals, introducing a potential for bias.
For parents concerned about the costs, inconvenience, and side effects from sustained JIA treatment once remission is achieved, data from CARRA will allow clinicians to provide evidence-based counseling on balancing the risks of discontinuing therapy, including the likelihood of regaining remission when disease returns, against the goals of stopping treatment.
“Parents are having more conversations about when to stop medications,” Dr. Ringold said. She indicated that these data should be helpful for providing guidance.
Dr. Ringold, Dr. Klotsche, and Dr. Halyabar reported having no potential conflicts of interest.
Flares are modest in preliminary data.
Flares are modest in preliminary data.
Many but not all children with juvenile idiopathic arthritis (JIA) can regain remission after stopping and then restarting treatment, according to preliminary data from the ongoing Recapture-JIA study that were presented in a symposium sponsored by the Rheumatology Research Foundation.
The aim of this study is to evaluate the risks of discontinuing treatment after a period when JIA has been well controlled. Such data are of increasing interest to parents now that many children with JIA are achieving sustained periods of remission, according to Sarah Ringold, MD, a pediatric rheumatologist and associate professor of pediatrics at Seattle Children’s Hospital.
In follow-up so far, “recapture rates range from 50% to 76%” depending on type of JIA, reported Dr. Ringold, who said that patients with systemic JIA have so far been the most likely to achieve a good response when treatment is restarted.
The study is being conducted through the Childhood Arthritis and Rheumatology Research Alliance, which has 71 participating centers and has accrued data on more than 10,000 children with rheumatic diseases. For the study, the researchers identified 384 children with JIA who were already enrolled in the CARRA registry and had discontinued medications and then subsequently restarted them, and they also enrolled a prospective cohort of patients new to the registry who presented with flare after discontinuing their medication. Dr. Ringold reported on 64 of the patients in the prospective cohort.
Median time to flare: 219 days
Of findings so far, disease recurrence after discontinuation has been generally characterized by flares “of moderate activity” several months to more than a year after treatment discontinuation, according to Dr. Ringold, who emphasized repeatedly that these data are preliminary. The median time to a flare after treatment discontinuation was approximately 7 months (219 days).
In the combined cohorts, the median age at onset of JIA was 4 years. The median age at time of discontinuation was 9 years. More than half (55%) were taking a conventional disease-modifying antirheumatic drug (DMARD) and 35% were taking a tumor necrosis factor inhibitor at the time that their therapy was discontinued.
Most JIA types are represented. The most common form is rheumatoid factor–negative oligoarticular JIA. The main outcome looked the rate of clinically inactive disease at 6 months in children who had discontinued therapy after a period of remission. They defined clinically inactive disease as a Physician’s Global Assessment of less than 1 and an active joint count of 0.
Systemic JIA recapture rate at 6 months: 76%
At the time of disease flare after treatment discontinuation across both the retrospective and prospective cohorts, the median clinical Juvenile Arthritis Disease Activity Score based on 10 joints (cJADAS10; score range of 0-30) was 3.5. The recapture rate to clinically inactive disease at 6 months was 76% in those with systemic JIA and 50% in those with rheumatoid factor–positive polyarticular JIA. Other subtypes fell within this range. Rates of inactive disease at 6 months according to cJADAS10 score were lower, ranging from 26% with enthesitis-related arthritis/juvenile psoriatic arthritis to 57% with systemic JIA.
About 40% of those who restarted on therapy after a flare took the same medication again. About one-third of patients were restarted on glucocorticoids, mostly involving injections to inflamed joints, and data are not yet in about whether these were restarted alone or with other drugs, according to Dr. Ringold.
The final analysis of this study will explore clinical and laboratory variables associated with disease recapture. In the prospective cohort, which did not reach its planned enrollment of 150 children because the COVID pandemic, a broad array of these variables was evaluated at baseline.
Numerous studies have already looked at predictors of sustained remission after stopping medications of JIA, according to Dr. Ringold, but she said that there is relatively little information about outcomes in children who stop medications, flare, and are retreated. Other experts agree.
“We know little about how successfully DMARDs can be discontinued and used again after a disease flare,” reported Jens Klotsche, MD, a researcher at the German Rheumatism Research Center, which is part of the Leibniz Institute in Berlin. Dr. Klotsche, who is an author of a recent study that found etanercept effective for retreatment when children with JIA had discontinued therapy, agreed that “data from large cohort studies are necessary to support the treatment decisions by clinicians, parents, and patients.”
JIA recurrence risk is unclear
In a systematic review published 2 years ago, rates of flare following discontinuation of treatment for JIA were relatively high, but there were some limitations to this analysis, according to the lead author, Olha Halyabar, MD, a pediatric rheumatologist at Boston Children’s Hospital.
“The data in our systematic review showed that overall quality of evidence was low, with large variations and sometimes very different conclusions,” Dr. Halyabar said in an interview. She believes that the data generated by the CARRA analysis will be valuable, particularly in evaluating outcomes across subtypes.
“Even though, at this point, [previously published] reports indicate overall high rates of recurrence (>50% for some JIA subtypes), there are some encouraging studies from early treat-to-target strategies,” she said, adding that large datasets like those from CARRA offer an opportunity to gather data likely to be clinically useful.
Dr. Ringold cautioned that there are some limitations to the CARRA analysis, including some missing data from the retrospective cohort. She also pointed out that patients have been assessed at routine clinical visits rather than at standardized intervals, introducing a potential for bias.
For parents concerned about the costs, inconvenience, and side effects from sustained JIA treatment once remission is achieved, data from CARRA will allow clinicians to provide evidence-based counseling on balancing the risks of discontinuing therapy, including the likelihood of regaining remission when disease returns, against the goals of stopping treatment.
“Parents are having more conversations about when to stop medications,” Dr. Ringold said. She indicated that these data should be helpful for providing guidance.
Dr. Ringold, Dr. Klotsche, and Dr. Halyabar reported having no potential conflicts of interest.
Many but not all children with juvenile idiopathic arthritis (JIA) can regain remission after stopping and then restarting treatment, according to preliminary data from the ongoing Recapture-JIA study that were presented in a symposium sponsored by the Rheumatology Research Foundation.
The aim of this study is to evaluate the risks of discontinuing treatment after a period when JIA has been well controlled. Such data are of increasing interest to parents now that many children with JIA are achieving sustained periods of remission, according to Sarah Ringold, MD, a pediatric rheumatologist and associate professor of pediatrics at Seattle Children’s Hospital.
In follow-up so far, “recapture rates range from 50% to 76%” depending on type of JIA, reported Dr. Ringold, who said that patients with systemic JIA have so far been the most likely to achieve a good response when treatment is restarted.
The study is being conducted through the Childhood Arthritis and Rheumatology Research Alliance, which has 71 participating centers and has accrued data on more than 10,000 children with rheumatic diseases. For the study, the researchers identified 384 children with JIA who were already enrolled in the CARRA registry and had discontinued medications and then subsequently restarted them, and they also enrolled a prospective cohort of patients new to the registry who presented with flare after discontinuing their medication. Dr. Ringold reported on 64 of the patients in the prospective cohort.
Median time to flare: 219 days
Of findings so far, disease recurrence after discontinuation has been generally characterized by flares “of moderate activity” several months to more than a year after treatment discontinuation, according to Dr. Ringold, who emphasized repeatedly that these data are preliminary. The median time to a flare after treatment discontinuation was approximately 7 months (219 days).
In the combined cohorts, the median age at onset of JIA was 4 years. The median age at time of discontinuation was 9 years. More than half (55%) were taking a conventional disease-modifying antirheumatic drug (DMARD) and 35% were taking a tumor necrosis factor inhibitor at the time that their therapy was discontinued.
Most JIA types are represented. The most common form is rheumatoid factor–negative oligoarticular JIA. The main outcome looked the rate of clinically inactive disease at 6 months in children who had discontinued therapy after a period of remission. They defined clinically inactive disease as a Physician’s Global Assessment of less than 1 and an active joint count of 0.
Systemic JIA recapture rate at 6 months: 76%
At the time of disease flare after treatment discontinuation across both the retrospective and prospective cohorts, the median clinical Juvenile Arthritis Disease Activity Score based on 10 joints (cJADAS10; score range of 0-30) was 3.5. The recapture rate to clinically inactive disease at 6 months was 76% in those with systemic JIA and 50% in those with rheumatoid factor–positive polyarticular JIA. Other subtypes fell within this range. Rates of inactive disease at 6 months according to cJADAS10 score were lower, ranging from 26% with enthesitis-related arthritis/juvenile psoriatic arthritis to 57% with systemic JIA.
About 40% of those who restarted on therapy after a flare took the same medication again. About one-third of patients were restarted on glucocorticoids, mostly involving injections to inflamed joints, and data are not yet in about whether these were restarted alone or with other drugs, according to Dr. Ringold.
The final analysis of this study will explore clinical and laboratory variables associated with disease recapture. In the prospective cohort, which did not reach its planned enrollment of 150 children because the COVID pandemic, a broad array of these variables was evaluated at baseline.
Numerous studies have already looked at predictors of sustained remission after stopping medications of JIA, according to Dr. Ringold, but she said that there is relatively little information about outcomes in children who stop medications, flare, and are retreated. Other experts agree.
“We know little about how successfully DMARDs can be discontinued and used again after a disease flare,” reported Jens Klotsche, MD, a researcher at the German Rheumatism Research Center, which is part of the Leibniz Institute in Berlin. Dr. Klotsche, who is an author of a recent study that found etanercept effective for retreatment when children with JIA had discontinued therapy, agreed that “data from large cohort studies are necessary to support the treatment decisions by clinicians, parents, and patients.”
JIA recurrence risk is unclear
In a systematic review published 2 years ago, rates of flare following discontinuation of treatment for JIA were relatively high, but there were some limitations to this analysis, according to the lead author, Olha Halyabar, MD, a pediatric rheumatologist at Boston Children’s Hospital.
“The data in our systematic review showed that overall quality of evidence was low, with large variations and sometimes very different conclusions,” Dr. Halyabar said in an interview. She believes that the data generated by the CARRA analysis will be valuable, particularly in evaluating outcomes across subtypes.
“Even though, at this point, [previously published] reports indicate overall high rates of recurrence (>50% for some JIA subtypes), there are some encouraging studies from early treat-to-target strategies,” she said, adding that large datasets like those from CARRA offer an opportunity to gather data likely to be clinically useful.
Dr. Ringold cautioned that there are some limitations to the CARRA analysis, including some missing data from the retrospective cohort. She also pointed out that patients have been assessed at routine clinical visits rather than at standardized intervals, introducing a potential for bias.
For parents concerned about the costs, inconvenience, and side effects from sustained JIA treatment once remission is achieved, data from CARRA will allow clinicians to provide evidence-based counseling on balancing the risks of discontinuing therapy, including the likelihood of regaining remission when disease returns, against the goals of stopping treatment.
“Parents are having more conversations about when to stop medications,” Dr. Ringold said. She indicated that these data should be helpful for providing guidance.
Dr. Ringold, Dr. Klotsche, and Dr. Halyabar reported having no potential conflicts of interest.
FROM RHEUMATOLOGY RESEARCH FOUNDATION SUMMER SERIES
New recommendations address ME/CFS diagnosis and management
New consensus recommendations address diagnosis and management of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), with advice that may also be helpful for patients with lingering symptoms following acute COVID-19 infection.
The document was published online Aug. 25, 2021, in the Mayo Clinic Proceedings by the 23-member U.S. ME/CFS Clinician Coalition, headed by Lucinda Bateman, MD, of the Bateman Horne Center of Excellence, Salt Lake City. The document is the culmination of work that began with a summit held at the center in March 2018.
The target audience is both generalist and specialist health care providers. While ME/CFS is estimated to affect up to 2.5 million Americans, more than 90% are either undiagnosed or misdiagnosed with other conditions such as depression. And those who are diagnosed often receive inappropriate, outdated treatments such as psychotherapy and exercise prescriptions.
“Despite myalgic encephalomyelitis/chronic fatigue syndrome affecting millions of people worldwide, many clinicians lack the knowledge to appropriately diagnose or manage ME/CFS. Unfortunately, clinical guidance has been scarce, obsolete, or potentially harmful,” Dr. Bateman and colleagues wrote.
The urgency of appropriate recognition and management of ME/CFS has increased as growing numbers of people are exhibiting signs and symptoms of ME/CFS following acute COVID-19 infection. This isn’t surprising because the illness has long been linked to other infections, including Epstein-Barr virus, the authors noted.
The document covers the epidemiology, impact, and prognosis of ME/CFS, as well as etiology and pathophysiology. “Scientific studies demonstrate multiple dysfunctional organ systems, including neuro, immune, and metabolic, in ME/CFS. These findings are not explained merely by deconditioning,” document coauthor Lily Chu, MD, an independent consultant in Burlingame, Calif., said in an interview.
The document reviews the 2015 U.S. Institute of Medicine (now Academy of Medicine) diagnostic criteria that are now also recommended by the Centers for Disease Control and Prevention. They are based on four main symptoms: substantial reduction or impairment in the ability to engage in preillness levels of occupational, educational, social or personal activities for longer than 6 months; postexertional malaise, a worsening of all current symptoms, that patients often describe as a “crash”; unrefreshing sleep; and cognitive impairment and/or orthostatic intolerance.
“The new diagnostic criteria focusing on the key symptom of postexertional malaise rather than chronic fatigue, which is common in many conditions, may make the diagnostic process quicker and more accurate. Diagnosis now is both an inclusionary and not just exclusionary process, so it’s not necessary to eliminate all causes of fatigue. Diagnose patients who fit the criteria and be alert for it in people with persistent symptoms post COVID,” Dr. Chu said.
The document provides advice for taking a clinical history to obtain the information necessary for making the diagnosis, including use of laboratory testing to rule out other conditions. Physical exams, while they may not reveal specific abnormalities, may help in identifying comorbidities and ruling out alternative diagnoses.
A long list of nonpharmacologic and pharmacologic treatment and management approaches is offered for each of the individual core and common ME/CFS symptoms, including postexertional malaise, orthostatic intolerance, sleep issues, cognitive dysfunction and fatigue, immune dysfunction, pain, and gastrointestinal issues.
The document recommends against using the “outdated standard of care” cognitive-behavioral therapy and graded exercise therapy as primary treatments for the illness. Instead, the authors recommend teaching patients “pacing,” an individualized approach to energy conservation aimed at minimizing the frequency, duration, and severity of postexertional malaise.
Clinicians are also advised to assess patients’ daily living needs and provide support, including acquiring handicap placards, work or school accommodations, and disability benefits.
“There are things clinicians can do now to help patients even without a disease-modifying treatment. These are actions they are already familiar with and carry out for people with other chronic diseases, which often have limited treatment options as well. Don’t underestimate the importance and value of supportive care for patients.” Dr. Chu said.
The recommendations are based primarily on clinical expertise because there are very few randomized trials, and much of the evidence from other types of trials has been flawed, document coauthor Anthony L. Komaroff, MD, of Brigham and Women’s Hospital and Harvard Medical School, both in Boston, said in an interview.
“The sad reality is there aren’t very many large randomized clinical trials with this illness and so what a group of very experienced clinicians did was to gather their collective experience and report it as that. It’s largely uncontrolled experience, but from people who have seen a lot of patients, for what it’s worth to the medical community.”
Dr. Komaroff also advised that clinicians watch out for ME/CFS in patients with long COVID. “If we find that those called long COVID meet ME/CFS criteria, the reason for knowing that is that there are already some treatments that according to experienced clinicians are helpful for ME/CFS, and it would be perfectly appropriate to try some of them in long COVID, particularly the ones that have minimal adverse reactions.”
The guidelines project was supported by the Open Medicine Foundation. Dr. Komaroff reported receiving personal fees from Serimmune outside the submitted work. Dr. Chu has no disclosures.
New consensus recommendations address diagnosis and management of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), with advice that may also be helpful for patients with lingering symptoms following acute COVID-19 infection.
The document was published online Aug. 25, 2021, in the Mayo Clinic Proceedings by the 23-member U.S. ME/CFS Clinician Coalition, headed by Lucinda Bateman, MD, of the Bateman Horne Center of Excellence, Salt Lake City. The document is the culmination of work that began with a summit held at the center in March 2018.
The target audience is both generalist and specialist health care providers. While ME/CFS is estimated to affect up to 2.5 million Americans, more than 90% are either undiagnosed or misdiagnosed with other conditions such as depression. And those who are diagnosed often receive inappropriate, outdated treatments such as psychotherapy and exercise prescriptions.
“Despite myalgic encephalomyelitis/chronic fatigue syndrome affecting millions of people worldwide, many clinicians lack the knowledge to appropriately diagnose or manage ME/CFS. Unfortunately, clinical guidance has been scarce, obsolete, or potentially harmful,” Dr. Bateman and colleagues wrote.
The urgency of appropriate recognition and management of ME/CFS has increased as growing numbers of people are exhibiting signs and symptoms of ME/CFS following acute COVID-19 infection. This isn’t surprising because the illness has long been linked to other infections, including Epstein-Barr virus, the authors noted.
The document covers the epidemiology, impact, and prognosis of ME/CFS, as well as etiology and pathophysiology. “Scientific studies demonstrate multiple dysfunctional organ systems, including neuro, immune, and metabolic, in ME/CFS. These findings are not explained merely by deconditioning,” document coauthor Lily Chu, MD, an independent consultant in Burlingame, Calif., said in an interview.
The document reviews the 2015 U.S. Institute of Medicine (now Academy of Medicine) diagnostic criteria that are now also recommended by the Centers for Disease Control and Prevention. They are based on four main symptoms: substantial reduction or impairment in the ability to engage in preillness levels of occupational, educational, social or personal activities for longer than 6 months; postexertional malaise, a worsening of all current symptoms, that patients often describe as a “crash”; unrefreshing sleep; and cognitive impairment and/or orthostatic intolerance.
“The new diagnostic criteria focusing on the key symptom of postexertional malaise rather than chronic fatigue, which is common in many conditions, may make the diagnostic process quicker and more accurate. Diagnosis now is both an inclusionary and not just exclusionary process, so it’s not necessary to eliminate all causes of fatigue. Diagnose patients who fit the criteria and be alert for it in people with persistent symptoms post COVID,” Dr. Chu said.
The document provides advice for taking a clinical history to obtain the information necessary for making the diagnosis, including use of laboratory testing to rule out other conditions. Physical exams, while they may not reveal specific abnormalities, may help in identifying comorbidities and ruling out alternative diagnoses.
A long list of nonpharmacologic and pharmacologic treatment and management approaches is offered for each of the individual core and common ME/CFS symptoms, including postexertional malaise, orthostatic intolerance, sleep issues, cognitive dysfunction and fatigue, immune dysfunction, pain, and gastrointestinal issues.
The document recommends against using the “outdated standard of care” cognitive-behavioral therapy and graded exercise therapy as primary treatments for the illness. Instead, the authors recommend teaching patients “pacing,” an individualized approach to energy conservation aimed at minimizing the frequency, duration, and severity of postexertional malaise.
Clinicians are also advised to assess patients’ daily living needs and provide support, including acquiring handicap placards, work or school accommodations, and disability benefits.
“There are things clinicians can do now to help patients even without a disease-modifying treatment. These are actions they are already familiar with and carry out for people with other chronic diseases, which often have limited treatment options as well. Don’t underestimate the importance and value of supportive care for patients.” Dr. Chu said.
The recommendations are based primarily on clinical expertise because there are very few randomized trials, and much of the evidence from other types of trials has been flawed, document coauthor Anthony L. Komaroff, MD, of Brigham and Women’s Hospital and Harvard Medical School, both in Boston, said in an interview.
“The sad reality is there aren’t very many large randomized clinical trials with this illness and so what a group of very experienced clinicians did was to gather their collective experience and report it as that. It’s largely uncontrolled experience, but from people who have seen a lot of patients, for what it’s worth to the medical community.”
Dr. Komaroff also advised that clinicians watch out for ME/CFS in patients with long COVID. “If we find that those called long COVID meet ME/CFS criteria, the reason for knowing that is that there are already some treatments that according to experienced clinicians are helpful for ME/CFS, and it would be perfectly appropriate to try some of them in long COVID, particularly the ones that have minimal adverse reactions.”
The guidelines project was supported by the Open Medicine Foundation. Dr. Komaroff reported receiving personal fees from Serimmune outside the submitted work. Dr. Chu has no disclosures.
New consensus recommendations address diagnosis and management of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), with advice that may also be helpful for patients with lingering symptoms following acute COVID-19 infection.
The document was published online Aug. 25, 2021, in the Mayo Clinic Proceedings by the 23-member U.S. ME/CFS Clinician Coalition, headed by Lucinda Bateman, MD, of the Bateman Horne Center of Excellence, Salt Lake City. The document is the culmination of work that began with a summit held at the center in March 2018.
The target audience is both generalist and specialist health care providers. While ME/CFS is estimated to affect up to 2.5 million Americans, more than 90% are either undiagnosed or misdiagnosed with other conditions such as depression. And those who are diagnosed often receive inappropriate, outdated treatments such as psychotherapy and exercise prescriptions.
“Despite myalgic encephalomyelitis/chronic fatigue syndrome affecting millions of people worldwide, many clinicians lack the knowledge to appropriately diagnose or manage ME/CFS. Unfortunately, clinical guidance has been scarce, obsolete, or potentially harmful,” Dr. Bateman and colleagues wrote.
The urgency of appropriate recognition and management of ME/CFS has increased as growing numbers of people are exhibiting signs and symptoms of ME/CFS following acute COVID-19 infection. This isn’t surprising because the illness has long been linked to other infections, including Epstein-Barr virus, the authors noted.
The document covers the epidemiology, impact, and prognosis of ME/CFS, as well as etiology and pathophysiology. “Scientific studies demonstrate multiple dysfunctional organ systems, including neuro, immune, and metabolic, in ME/CFS. These findings are not explained merely by deconditioning,” document coauthor Lily Chu, MD, an independent consultant in Burlingame, Calif., said in an interview.
The document reviews the 2015 U.S. Institute of Medicine (now Academy of Medicine) diagnostic criteria that are now also recommended by the Centers for Disease Control and Prevention. They are based on four main symptoms: substantial reduction or impairment in the ability to engage in preillness levels of occupational, educational, social or personal activities for longer than 6 months; postexertional malaise, a worsening of all current symptoms, that patients often describe as a “crash”; unrefreshing sleep; and cognitive impairment and/or orthostatic intolerance.
“The new diagnostic criteria focusing on the key symptom of postexertional malaise rather than chronic fatigue, which is common in many conditions, may make the diagnostic process quicker and more accurate. Diagnosis now is both an inclusionary and not just exclusionary process, so it’s not necessary to eliminate all causes of fatigue. Diagnose patients who fit the criteria and be alert for it in people with persistent symptoms post COVID,” Dr. Chu said.
The document provides advice for taking a clinical history to obtain the information necessary for making the diagnosis, including use of laboratory testing to rule out other conditions. Physical exams, while they may not reveal specific abnormalities, may help in identifying comorbidities and ruling out alternative diagnoses.
A long list of nonpharmacologic and pharmacologic treatment and management approaches is offered for each of the individual core and common ME/CFS symptoms, including postexertional malaise, orthostatic intolerance, sleep issues, cognitive dysfunction and fatigue, immune dysfunction, pain, and gastrointestinal issues.
The document recommends against using the “outdated standard of care” cognitive-behavioral therapy and graded exercise therapy as primary treatments for the illness. Instead, the authors recommend teaching patients “pacing,” an individualized approach to energy conservation aimed at minimizing the frequency, duration, and severity of postexertional malaise.
Clinicians are also advised to assess patients’ daily living needs and provide support, including acquiring handicap placards, work or school accommodations, and disability benefits.
“There are things clinicians can do now to help patients even without a disease-modifying treatment. These are actions they are already familiar with and carry out for people with other chronic diseases, which often have limited treatment options as well. Don’t underestimate the importance and value of supportive care for patients.” Dr. Chu said.
The recommendations are based primarily on clinical expertise because there are very few randomized trials, and much of the evidence from other types of trials has been flawed, document coauthor Anthony L. Komaroff, MD, of Brigham and Women’s Hospital and Harvard Medical School, both in Boston, said in an interview.
“The sad reality is there aren’t very many large randomized clinical trials with this illness and so what a group of very experienced clinicians did was to gather their collective experience and report it as that. It’s largely uncontrolled experience, but from people who have seen a lot of patients, for what it’s worth to the medical community.”
Dr. Komaroff also advised that clinicians watch out for ME/CFS in patients with long COVID. “If we find that those called long COVID meet ME/CFS criteria, the reason for knowing that is that there are already some treatments that according to experienced clinicians are helpful for ME/CFS, and it would be perfectly appropriate to try some of them in long COVID, particularly the ones that have minimal adverse reactions.”
The guidelines project was supported by the Open Medicine Foundation. Dr. Komaroff reported receiving personal fees from Serimmune outside the submitted work. Dr. Chu has no disclosures.
FROM THE MAYO CLINIC PROCEEDINGS
Bimekizumab approved in Europe for psoriasis treatment
, according to a statement from the manufacturer.
Bimekizumab (Bimzelx), a humanized IgG1 monoclonal antibody, is the first approved treatment for moderate to severe plaque psoriasis that selectively inhibits interleukin (IL)–17A and IL-17F, the statement from UCB said.
In the United States, the Food and Drug Administration is expected to make a decision on approval of bimekizumab for treating psoriasis on Oct. 15.
Approval in the EU was based on data from three phase 3 trials including a total of 1,480 adult patients with moderate to severe psoriasis, which found that those treated with bimekizumab experienced significantly greater skin clearance, compared with placebo, ustekinumab, and adalimumab, with a favorable safety profile, according to the company.
In all three studies (BE VIVID, BE READY, and BE SURE), more than 80% of patients treated with bimekizumab showed improved skin clearance after 16 weeks, significantly more than those treated with ustekinumab, placebo, or adalimumab, based on an improvement of at least 90% in the Psoriasis Area & Severity Index (PASI 90) and an Investigator’s Global Assessment (IGA) response of clear or almost clear skin (IGA 0/1). In all three studies, these clinical responses persisted after 1 year.
The recommended dose of bimekizumab is 320 mg, given in two subcutaneous injections every 4 weeks to week 16, then every 8 weeks. However, for “some patients” weighing 120 kg or more who have not achieved complete skin clearance at 16 weeks, 320 mg every 4 weeks after that time may improve response to treatment, according to the company statement.
The most common treatment-related adverse events in the studies were upper respiratory tract infections (a majority of which were nasopharyngitis), reported by 14.5% of patients, followed by oral candidiasis, reported by 7.3%.
Results of BE READY and BE VIVID were published in The Lancet. Results of the BE SURE study were published in The New England Journal of Medicine.
Bimekizumab is contraindicated for individuals with clinically important active infections such as tuberculosis, and for individuals with any hypersensitivity to the active substance. More details on bimekizumab are available on the website of the European Medicines Agency.
, according to a statement from the manufacturer.
Bimekizumab (Bimzelx), a humanized IgG1 monoclonal antibody, is the first approved treatment for moderate to severe plaque psoriasis that selectively inhibits interleukin (IL)–17A and IL-17F, the statement from UCB said.
In the United States, the Food and Drug Administration is expected to make a decision on approval of bimekizumab for treating psoriasis on Oct. 15.
Approval in the EU was based on data from three phase 3 trials including a total of 1,480 adult patients with moderate to severe psoriasis, which found that those treated with bimekizumab experienced significantly greater skin clearance, compared with placebo, ustekinumab, and adalimumab, with a favorable safety profile, according to the company.
In all three studies (BE VIVID, BE READY, and BE SURE), more than 80% of patients treated with bimekizumab showed improved skin clearance after 16 weeks, significantly more than those treated with ustekinumab, placebo, or adalimumab, based on an improvement of at least 90% in the Psoriasis Area & Severity Index (PASI 90) and an Investigator’s Global Assessment (IGA) response of clear or almost clear skin (IGA 0/1). In all three studies, these clinical responses persisted after 1 year.
The recommended dose of bimekizumab is 320 mg, given in two subcutaneous injections every 4 weeks to week 16, then every 8 weeks. However, for “some patients” weighing 120 kg or more who have not achieved complete skin clearance at 16 weeks, 320 mg every 4 weeks after that time may improve response to treatment, according to the company statement.
The most common treatment-related adverse events in the studies were upper respiratory tract infections (a majority of which were nasopharyngitis), reported by 14.5% of patients, followed by oral candidiasis, reported by 7.3%.
Results of BE READY and BE VIVID were published in The Lancet. Results of the BE SURE study were published in The New England Journal of Medicine.
Bimekizumab is contraindicated for individuals with clinically important active infections such as tuberculosis, and for individuals with any hypersensitivity to the active substance. More details on bimekizumab are available on the website of the European Medicines Agency.
, according to a statement from the manufacturer.
Bimekizumab (Bimzelx), a humanized IgG1 monoclonal antibody, is the first approved treatment for moderate to severe plaque psoriasis that selectively inhibits interleukin (IL)–17A and IL-17F, the statement from UCB said.
In the United States, the Food and Drug Administration is expected to make a decision on approval of bimekizumab for treating psoriasis on Oct. 15.
Approval in the EU was based on data from three phase 3 trials including a total of 1,480 adult patients with moderate to severe psoriasis, which found that those treated with bimekizumab experienced significantly greater skin clearance, compared with placebo, ustekinumab, and adalimumab, with a favorable safety profile, according to the company.
In all three studies (BE VIVID, BE READY, and BE SURE), more than 80% of patients treated with bimekizumab showed improved skin clearance after 16 weeks, significantly more than those treated with ustekinumab, placebo, or adalimumab, based on an improvement of at least 90% in the Psoriasis Area & Severity Index (PASI 90) and an Investigator’s Global Assessment (IGA) response of clear or almost clear skin (IGA 0/1). In all three studies, these clinical responses persisted after 1 year.
The recommended dose of bimekizumab is 320 mg, given in two subcutaneous injections every 4 weeks to week 16, then every 8 weeks. However, for “some patients” weighing 120 kg or more who have not achieved complete skin clearance at 16 weeks, 320 mg every 4 weeks after that time may improve response to treatment, according to the company statement.
The most common treatment-related adverse events in the studies were upper respiratory tract infections (a majority of which were nasopharyngitis), reported by 14.5% of patients, followed by oral candidiasis, reported by 7.3%.
Results of BE READY and BE VIVID were published in The Lancet. Results of the BE SURE study were published in The New England Journal of Medicine.
Bimekizumab is contraindicated for individuals with clinically important active infections such as tuberculosis, and for individuals with any hypersensitivity to the active substance. More details on bimekizumab are available on the website of the European Medicines Agency.
Psoriatic arthritis health care costs continue to rise over time
Annual health care costs for patients with psoriatic arthritis rose over recent 5-year periods across all categories of resource use to a significantly greater extent than among patients with psoriasis only or those without any psoriatic disease diagnoses, according to commercial insurance claims data.
Using an IBM MarketScan Commercial Database, researchers examined claims data for 208,434 patients with psoriasis, 47,274 with PsA, and 255,708 controls who had neither psoriasis nor PsA. Controls were matched for age and sex. Those with RA, ankylosing spondylitis, Crohn’s disease, or ulcerative colitis were excluded.
The investigators examined data for 2009-2020, following patients for 5 years within that period. They looked at hospitalizations, outpatient and pharmacy services, lab services, and office visits, Steven Peterson, director of market access for rheumatology at Janssen Pharmaceuticals, said in his presentation of the data at the Pan American League of Associations for Rheumatology 2021 annual meeting, held recently as a virtual event.
The research was also published online May 2, 2021, in Clinical Rheumatology.
Big differences between the groups were seen in the first year, when the average health care costs for the PsA group were $28,322, about half of which was outpatient drug costs. That compared with $12,039 for the psoriasis group and $6,672 for the control group.
The differences tended to widen over time. By the fifth year, average costs for the PsA group were $34,290, nearly 60% of which were drug costs. That compared with $12,877 for the psoriasis group and $8,569 for the control group. In each year examined, outpatient drug costs accounted for less than half of the expenses for the psoriasis group and about a quarter for the control group.
Researchers found that the PsA group needed 28.7 prescriptions per person per year, compared with 17.0 and 12.7 in the psoriasis and control groups, respectively, Mr. Peterson said. He also noted that patients with PsA and psoriasis tended to have higher rates of hypertension, depression, and anxiety.
“The cost and resource utilization disparity between these patient groups demonstrates the high remaining unmet medical need for patients with psoriasis and psoriatic arthritis,” Mr. Peterson said during the virtual proceedings.
Do findings reflect treatment advances?
Elaine Husni, MD, MPH, director of the Arthritis and Musculoskeletal Center at the Cleveland Clinic, where she studies health outcomes in PsA, said the findings are helpful in pointing to a trend across a large sample. But she added it’s important to remember that the increasing costs could reflect recent advances in PsA treatment, which include costly biologic drugs.
“There’s a ton more treatments for psoriasis and psoriatic arthritis than there were even just 5 years ago,” she said in an interview. She was not involved in the research.
Dr. Husni would like to see a more detailed look at the costs, from the categories of expenses to the patients who are incurring the highest costs.
“Is it just a couple of percent of really sick patients that are driving the psoriatic arthritis group?” she wondered.
She also pointed out that PsA is going to be more expensive by its very nature. PsA tends to develop 3-10 years after psoriasis, adding to the costs for someone who already has psoriasis and at a time when they are older and likely have higher health care costs because of comorbidities that develop with age.
Dr. Husni said she does think about treatment costs, in that a less expensive first-line drug might be more appropriate than going straight to a more expensive biologic, especially because they also tend to be safer. She said it’s not just a simple question of curbing costs.
“Is there a way that we can personalize medicine?” she asked. “Is there a way that we can be more accurate about which people may need the more expensive drugs, and which patients may need the less expensive drugs? Are we getting better at monitoring so we can avoid high-cost events?”
Mr. Peterson is an employee of Janssen Pharmaceuticals. Dr. Husni reported serving as a consultant to AbbVie, Amgen, Bristol-Myers Squibb, UCB, Novartis, Lilly, and Pfizer.
* Update, 9/28/21: The headline and parts of this story were updated to better reflect the study on which it reports.
A version of this article first appeared on Medscape.com.
Annual health care costs for patients with psoriatic arthritis rose over recent 5-year periods across all categories of resource use to a significantly greater extent than among patients with psoriasis only or those without any psoriatic disease diagnoses, according to commercial insurance claims data.
Using an IBM MarketScan Commercial Database, researchers examined claims data for 208,434 patients with psoriasis, 47,274 with PsA, and 255,708 controls who had neither psoriasis nor PsA. Controls were matched for age and sex. Those with RA, ankylosing spondylitis, Crohn’s disease, or ulcerative colitis were excluded.
The investigators examined data for 2009-2020, following patients for 5 years within that period. They looked at hospitalizations, outpatient and pharmacy services, lab services, and office visits, Steven Peterson, director of market access for rheumatology at Janssen Pharmaceuticals, said in his presentation of the data at the Pan American League of Associations for Rheumatology 2021 annual meeting, held recently as a virtual event.
The research was also published online May 2, 2021, in Clinical Rheumatology.
Big differences between the groups were seen in the first year, when the average health care costs for the PsA group were $28,322, about half of which was outpatient drug costs. That compared with $12,039 for the psoriasis group and $6,672 for the control group.
The differences tended to widen over time. By the fifth year, average costs for the PsA group were $34,290, nearly 60% of which were drug costs. That compared with $12,877 for the psoriasis group and $8,569 for the control group. In each year examined, outpatient drug costs accounted for less than half of the expenses for the psoriasis group and about a quarter for the control group.
Researchers found that the PsA group needed 28.7 prescriptions per person per year, compared with 17.0 and 12.7 in the psoriasis and control groups, respectively, Mr. Peterson said. He also noted that patients with PsA and psoriasis tended to have higher rates of hypertension, depression, and anxiety.
“The cost and resource utilization disparity between these patient groups demonstrates the high remaining unmet medical need for patients with psoriasis and psoriatic arthritis,” Mr. Peterson said during the virtual proceedings.
Do findings reflect treatment advances?
Elaine Husni, MD, MPH, director of the Arthritis and Musculoskeletal Center at the Cleveland Clinic, where she studies health outcomes in PsA, said the findings are helpful in pointing to a trend across a large sample. But she added it’s important to remember that the increasing costs could reflect recent advances in PsA treatment, which include costly biologic drugs.
“There’s a ton more treatments for psoriasis and psoriatic arthritis than there were even just 5 years ago,” she said in an interview. She was not involved in the research.
Dr. Husni would like to see a more detailed look at the costs, from the categories of expenses to the patients who are incurring the highest costs.
“Is it just a couple of percent of really sick patients that are driving the psoriatic arthritis group?” she wondered.
She also pointed out that PsA is going to be more expensive by its very nature. PsA tends to develop 3-10 years after psoriasis, adding to the costs for someone who already has psoriasis and at a time when they are older and likely have higher health care costs because of comorbidities that develop with age.
Dr. Husni said she does think about treatment costs, in that a less expensive first-line drug might be more appropriate than going straight to a more expensive biologic, especially because they also tend to be safer. She said it’s not just a simple question of curbing costs.
“Is there a way that we can personalize medicine?” she asked. “Is there a way that we can be more accurate about which people may need the more expensive drugs, and which patients may need the less expensive drugs? Are we getting better at monitoring so we can avoid high-cost events?”
Mr. Peterson is an employee of Janssen Pharmaceuticals. Dr. Husni reported serving as a consultant to AbbVie, Amgen, Bristol-Myers Squibb, UCB, Novartis, Lilly, and Pfizer.
* Update, 9/28/21: The headline and parts of this story were updated to better reflect the study on which it reports.
A version of this article first appeared on Medscape.com.
Annual health care costs for patients with psoriatic arthritis rose over recent 5-year periods across all categories of resource use to a significantly greater extent than among patients with psoriasis only or those without any psoriatic disease diagnoses, according to commercial insurance claims data.
Using an IBM MarketScan Commercial Database, researchers examined claims data for 208,434 patients with psoriasis, 47,274 with PsA, and 255,708 controls who had neither psoriasis nor PsA. Controls were matched for age and sex. Those with RA, ankylosing spondylitis, Crohn’s disease, or ulcerative colitis were excluded.
The investigators examined data for 2009-2020, following patients for 5 years within that period. They looked at hospitalizations, outpatient and pharmacy services, lab services, and office visits, Steven Peterson, director of market access for rheumatology at Janssen Pharmaceuticals, said in his presentation of the data at the Pan American League of Associations for Rheumatology 2021 annual meeting, held recently as a virtual event.
The research was also published online May 2, 2021, in Clinical Rheumatology.
Big differences between the groups were seen in the first year, when the average health care costs for the PsA group were $28,322, about half of which was outpatient drug costs. That compared with $12,039 for the psoriasis group and $6,672 for the control group.
The differences tended to widen over time. By the fifth year, average costs for the PsA group were $34,290, nearly 60% of which were drug costs. That compared with $12,877 for the psoriasis group and $8,569 for the control group. In each year examined, outpatient drug costs accounted for less than half of the expenses for the psoriasis group and about a quarter for the control group.
Researchers found that the PsA group needed 28.7 prescriptions per person per year, compared with 17.0 and 12.7 in the psoriasis and control groups, respectively, Mr. Peterson said. He also noted that patients with PsA and psoriasis tended to have higher rates of hypertension, depression, and anxiety.
“The cost and resource utilization disparity between these patient groups demonstrates the high remaining unmet medical need for patients with psoriasis and psoriatic arthritis,” Mr. Peterson said during the virtual proceedings.
Do findings reflect treatment advances?
Elaine Husni, MD, MPH, director of the Arthritis and Musculoskeletal Center at the Cleveland Clinic, where she studies health outcomes in PsA, said the findings are helpful in pointing to a trend across a large sample. But she added it’s important to remember that the increasing costs could reflect recent advances in PsA treatment, which include costly biologic drugs.
“There’s a ton more treatments for psoriasis and psoriatic arthritis than there were even just 5 years ago,” she said in an interview. She was not involved in the research.
Dr. Husni would like to see a more detailed look at the costs, from the categories of expenses to the patients who are incurring the highest costs.
“Is it just a couple of percent of really sick patients that are driving the psoriatic arthritis group?” she wondered.
She also pointed out that PsA is going to be more expensive by its very nature. PsA tends to develop 3-10 years after psoriasis, adding to the costs for someone who already has psoriasis and at a time when they are older and likely have higher health care costs because of comorbidities that develop with age.
Dr. Husni said she does think about treatment costs, in that a less expensive first-line drug might be more appropriate than going straight to a more expensive biologic, especially because they also tend to be safer. She said it’s not just a simple question of curbing costs.
“Is there a way that we can personalize medicine?” she asked. “Is there a way that we can be more accurate about which people may need the more expensive drugs, and which patients may need the less expensive drugs? Are we getting better at monitoring so we can avoid high-cost events?”
Mr. Peterson is an employee of Janssen Pharmaceuticals. Dr. Husni reported serving as a consultant to AbbVie, Amgen, Bristol-Myers Squibb, UCB, Novartis, Lilly, and Pfizer.
* Update, 9/28/21: The headline and parts of this story were updated to better reflect the study on which it reports.
A version of this article first appeared on Medscape.com.
Health care workers eager for COVID booster shots
As COVID vaccine boosters move closer to reality, most physicians and nurses are ready and willing to get another shot in the arm, according to a new Medscape survey.
Altogether, 93% of physicians and 87% of nurses/advanced practice nurses (APNs) said they wanted to get a booster, although the timing of when they wanted the shots differed somewhat between the two groups surveyed Aug. 4-15.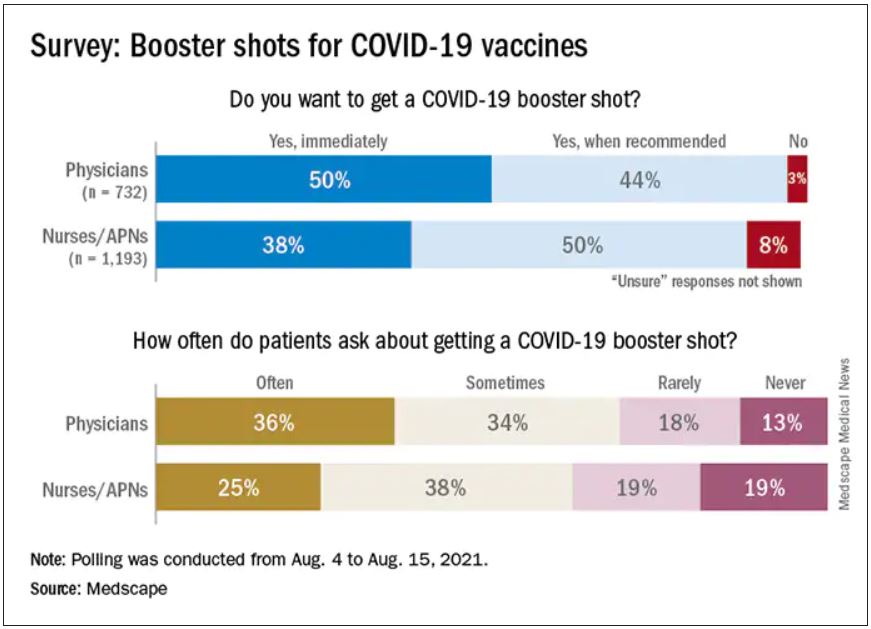
Among the 732 physicians polled, 50% wanted to get their shot immediately, compared with 38% of the 1,193 nurses/APNs who responded, while 44% of physicians and 50% of nurses/APNs said that they would wait until the vaccine booster was authorized and recommended.
At this point in time, almost all of the health care workers surveyed – 98% of physicians and 94% of nurses/APNs – have been fully vaccinated against COVID-19. A small proportion of each group, however, received the Johnson & Johnson vaccine (1% of physicians and 3% of nurses) and are not included in the current plan for booster shots.
The Medscape survey sample did include one group that is already eligible for a third dose: About 20% of physicians and 26% of nurses/ANPs said they have a condition or take a medication that compromises their immune system.
Respondents’ experiences with patient requests for boosters suggest a somewhat lower level of interest. About two-thirds of the health care workers (69% of physicians and 63% of nurses) said that patients frequently or sometimes asked about COVID boosters, compared with 13% (physicians) and 19% (nurses) who said their patients had never asked.
Interest lower among general population
In a separate survey conducted by WebMD, 82% of those who have been at least partially vaccinated said they want to get a COVID vaccine booster (14% immediately and 68% after authorization and recommendation). Of the remaining vaccinees, 7% said they do not want to get a booster and 11% were unsure.
The full sample of 592 respondents surveyed Aug. 5-10, however, included 19% who do not plan to get vaccinated and 6% who are planning to be vaccinated but have not yet done so.
The proportion of immunocompromised individuals in the two survey groups was similar, with about 25% of those in the WebMD survey reporting they have a condition or take a medication that compromises their immune system. Those respondents were more than twice as likely to want to get a booster immediately, compared to those with an uncompromised immune system (24% vs. 11%).
The distribution of vaccines received by brand was also comparable between the two groups surveyed. Of health care workers and readers, over half of each group received the Pfizer/BioNTech vaccine (59% vs. 54%), followed by Moderna (38% vs. 40%) and Johnson & Johnson (3% vs. 5%).
A version of this article first appeared on Medscape.com.
As COVID vaccine boosters move closer to reality, most physicians and nurses are ready and willing to get another shot in the arm, according to a new Medscape survey.
Altogether, 93% of physicians and 87% of nurses/advanced practice nurses (APNs) said they wanted to get a booster, although the timing of when they wanted the shots differed somewhat between the two groups surveyed Aug. 4-15.
Among the 732 physicians polled, 50% wanted to get their shot immediately, compared with 38% of the 1,193 nurses/APNs who responded, while 44% of physicians and 50% of nurses/APNs said that they would wait until the vaccine booster was authorized and recommended.
At this point in time, almost all of the health care workers surveyed – 98% of physicians and 94% of nurses/APNs – have been fully vaccinated against COVID-19. A small proportion of each group, however, received the Johnson & Johnson vaccine (1% of physicians and 3% of nurses) and are not included in the current plan for booster shots.
The Medscape survey sample did include one group that is already eligible for a third dose: About 20% of physicians and 26% of nurses/ANPs said they have a condition or take a medication that compromises their immune system.
Respondents’ experiences with patient requests for boosters suggest a somewhat lower level of interest. About two-thirds of the health care workers (69% of physicians and 63% of nurses) said that patients frequently or sometimes asked about COVID boosters, compared with 13% (physicians) and 19% (nurses) who said their patients had never asked.
Interest lower among general population
In a separate survey conducted by WebMD, 82% of those who have been at least partially vaccinated said they want to get a COVID vaccine booster (14% immediately and 68% after authorization and recommendation). Of the remaining vaccinees, 7% said they do not want to get a booster and 11% were unsure.
The full sample of 592 respondents surveyed Aug. 5-10, however, included 19% who do not plan to get vaccinated and 6% who are planning to be vaccinated but have not yet done so.
The proportion of immunocompromised individuals in the two survey groups was similar, with about 25% of those in the WebMD survey reporting they have a condition or take a medication that compromises their immune system. Those respondents were more than twice as likely to want to get a booster immediately, compared to those with an uncompromised immune system (24% vs. 11%).
The distribution of vaccines received by brand was also comparable between the two groups surveyed. Of health care workers and readers, over half of each group received the Pfizer/BioNTech vaccine (59% vs. 54%), followed by Moderna (38% vs. 40%) and Johnson & Johnson (3% vs. 5%).
A version of this article first appeared on Medscape.com.
As COVID vaccine boosters move closer to reality, most physicians and nurses are ready and willing to get another shot in the arm, according to a new Medscape survey.
Altogether, 93% of physicians and 87% of nurses/advanced practice nurses (APNs) said they wanted to get a booster, although the timing of when they wanted the shots differed somewhat between the two groups surveyed Aug. 4-15.
Among the 732 physicians polled, 50% wanted to get their shot immediately, compared with 38% of the 1,193 nurses/APNs who responded, while 44% of physicians and 50% of nurses/APNs said that they would wait until the vaccine booster was authorized and recommended.
At this point in time, almost all of the health care workers surveyed – 98% of physicians and 94% of nurses/APNs – have been fully vaccinated against COVID-19. A small proportion of each group, however, received the Johnson & Johnson vaccine (1% of physicians and 3% of nurses) and are not included in the current plan for booster shots.
The Medscape survey sample did include one group that is already eligible for a third dose: About 20% of physicians and 26% of nurses/ANPs said they have a condition or take a medication that compromises their immune system.
Respondents’ experiences with patient requests for boosters suggest a somewhat lower level of interest. About two-thirds of the health care workers (69% of physicians and 63% of nurses) said that patients frequently or sometimes asked about COVID boosters, compared with 13% (physicians) and 19% (nurses) who said their patients had never asked.
Interest lower among general population
In a separate survey conducted by WebMD, 82% of those who have been at least partially vaccinated said they want to get a COVID vaccine booster (14% immediately and 68% after authorization and recommendation). Of the remaining vaccinees, 7% said they do not want to get a booster and 11% were unsure.
The full sample of 592 respondents surveyed Aug. 5-10, however, included 19% who do not plan to get vaccinated and 6% who are planning to be vaccinated but have not yet done so.
The proportion of immunocompromised individuals in the two survey groups was similar, with about 25% of those in the WebMD survey reporting they have a condition or take a medication that compromises their immune system. Those respondents were more than twice as likely to want to get a booster immediately, compared to those with an uncompromised immune system (24% vs. 11%).
The distribution of vaccines received by brand was also comparable between the two groups surveyed. Of health care workers and readers, over half of each group received the Pfizer/BioNTech vaccine (59% vs. 54%), followed by Moderna (38% vs. 40%) and Johnson & Johnson (3% vs. 5%).
A version of this article first appeared on Medscape.com.
Plastic barriers may not stop COVID-19 spread, experts say
Plastic barriers that separate people in stores, restaurants, and classrooms may not be as effective at stopping the spread of COVID-19 as originally thought, according to The New York Times.
Scientists who study air flow, ventilation, and aerosol droplets say the barriers may not help, and in fact, could make the situation worse by blocking normal air flow, the newspaper reported.
Typically, as people interact and breathe in a room, currents and ventilation systems recirculate the air and disperse the exhaled particles. With plastic barriers, however, particles could get trapped in “dead zones” and build up.
“If you have a forest of barriers in a classroom, it’s going to interfere with proper ventilation of that room,” Linsey Marr, professor of civil and environmental engineering at Virginia Tech, told the newspaper.
“Everybody’s aerosols are going to be trapped and stuck there and building up, and they will end up spreading beyond your own desk,” she said.
Several variables factor into the efficacy of plastic barriers, The New York Times reported. Shields may stop big respiratory droplets from coughs and sneezes, for instance, but they may not do much to prevent small aerosol particles from viruses such as COVID-19 from spreading.
“We have shown this effect of blocking larger particles, but also that the smaller aerosols travel over the screen and become mixed in the room air within about 5 minutes,” Catherine Noakes, professor of environment engineering at the University of Leeds, told the newspaper.
“This means if people are interacting for more than a few minutes, they would likely be exposed to the virus regardless of the screen,” she said.
The effectiveness of plastic barriers likely also depends on the location and setup, the newspaper reported. A bus driver with a large barrier, for instance, may be able to avoid inhaling the particles that passengers are exhaling. A bank cashier or store clerk behind a large barrier may also be partly protected.
Even still, scientists say more research is needed. For instance, taller barriers are more likely to be effective. However, a large number of barriers in one room could likely block air flow.
Researchers have recommended that schools and offices focus on ventilation, masks, and vaccines to slow the spread of the coronavirus.
“Air flow in rooms is pretty complicated,” Richard Corsi, dean of engineering at the University of California at Davis, told the newspaper.
“Every room is different in terms of the arrangement of furniture, the height of the walls and ceilings, the vents, where the bookshelves are,” he said. “All of these things have a huge impact on the actual flow and air distribution in a room.”
A version of this article first appeared on WebMD.com.
Plastic barriers that separate people in stores, restaurants, and classrooms may not be as effective at stopping the spread of COVID-19 as originally thought, according to The New York Times.
Scientists who study air flow, ventilation, and aerosol droplets say the barriers may not help, and in fact, could make the situation worse by blocking normal air flow, the newspaper reported.
Typically, as people interact and breathe in a room, currents and ventilation systems recirculate the air and disperse the exhaled particles. With plastic barriers, however, particles could get trapped in “dead zones” and build up.
“If you have a forest of barriers in a classroom, it’s going to interfere with proper ventilation of that room,” Linsey Marr, professor of civil and environmental engineering at Virginia Tech, told the newspaper.
“Everybody’s aerosols are going to be trapped and stuck there and building up, and they will end up spreading beyond your own desk,” she said.
Several variables factor into the efficacy of plastic barriers, The New York Times reported. Shields may stop big respiratory droplets from coughs and sneezes, for instance, but they may not do much to prevent small aerosol particles from viruses such as COVID-19 from spreading.
“We have shown this effect of blocking larger particles, but also that the smaller aerosols travel over the screen and become mixed in the room air within about 5 minutes,” Catherine Noakes, professor of environment engineering at the University of Leeds, told the newspaper.
“This means if people are interacting for more than a few minutes, they would likely be exposed to the virus regardless of the screen,” she said.
The effectiveness of plastic barriers likely also depends on the location and setup, the newspaper reported. A bus driver with a large barrier, for instance, may be able to avoid inhaling the particles that passengers are exhaling. A bank cashier or store clerk behind a large barrier may also be partly protected.
Even still, scientists say more research is needed. For instance, taller barriers are more likely to be effective. However, a large number of barriers in one room could likely block air flow.
Researchers have recommended that schools and offices focus on ventilation, masks, and vaccines to slow the spread of the coronavirus.
“Air flow in rooms is pretty complicated,” Richard Corsi, dean of engineering at the University of California at Davis, told the newspaper.
“Every room is different in terms of the arrangement of furniture, the height of the walls and ceilings, the vents, where the bookshelves are,” he said. “All of these things have a huge impact on the actual flow and air distribution in a room.”
A version of this article first appeared on WebMD.com.
Plastic barriers that separate people in stores, restaurants, and classrooms may not be as effective at stopping the spread of COVID-19 as originally thought, according to The New York Times.
Scientists who study air flow, ventilation, and aerosol droplets say the barriers may not help, and in fact, could make the situation worse by blocking normal air flow, the newspaper reported.
Typically, as people interact and breathe in a room, currents and ventilation systems recirculate the air and disperse the exhaled particles. With plastic barriers, however, particles could get trapped in “dead zones” and build up.
“If you have a forest of barriers in a classroom, it’s going to interfere with proper ventilation of that room,” Linsey Marr, professor of civil and environmental engineering at Virginia Tech, told the newspaper.
“Everybody’s aerosols are going to be trapped and stuck there and building up, and they will end up spreading beyond your own desk,” she said.
Several variables factor into the efficacy of plastic barriers, The New York Times reported. Shields may stop big respiratory droplets from coughs and sneezes, for instance, but they may not do much to prevent small aerosol particles from viruses such as COVID-19 from spreading.
“We have shown this effect of blocking larger particles, but also that the smaller aerosols travel over the screen and become mixed in the room air within about 5 minutes,” Catherine Noakes, professor of environment engineering at the University of Leeds, told the newspaper.
“This means if people are interacting for more than a few minutes, they would likely be exposed to the virus regardless of the screen,” she said.
The effectiveness of plastic barriers likely also depends on the location and setup, the newspaper reported. A bus driver with a large barrier, for instance, may be able to avoid inhaling the particles that passengers are exhaling. A bank cashier or store clerk behind a large barrier may also be partly protected.
Even still, scientists say more research is needed. For instance, taller barriers are more likely to be effective. However, a large number of barriers in one room could likely block air flow.
Researchers have recommended that schools and offices focus on ventilation, masks, and vaccines to slow the spread of the coronavirus.
“Air flow in rooms is pretty complicated,” Richard Corsi, dean of engineering at the University of California at Davis, told the newspaper.
“Every room is different in terms of the arrangement of furniture, the height of the walls and ceilings, the vents, where the bookshelves are,” he said. “All of these things have a huge impact on the actual flow and air distribution in a room.”
A version of this article first appeared on WebMD.com.
In RA patients, multiple comorbidities lower odds of disease control
An increasing number of comorbidities in patients with rheumatoid arthritis correlates with a lower likelihood of reaching treatment targets, according to an analysis conducted with a series of large real-world datasets and presented in a symposium sponsored by the Rheumatology Research Foundation.
When compared to those with the lowest burden of comorbidity in one of these analyses, those with the highest had a nearly 50% lower likelihood (odds ratio, 0.54; 95% confidence interval [CI], 0.34-0.85) of achieving low disease activity or remission, according to Bryant England, MD, PhD, assistant professor in the division of rheumatology at the University of Nebraska, Omaha.
“Patients with more comorbidities struggle to reach treatment targets,” Dr. England said. In the treatment of RA, “we typically focus only on the joints, but these data suggest we need to begin to think more holistically about managing these patients.”
Both the American College of Rheumatology (ACR) and the European Alliance of Associations for Rheumatology (EULAR) endorse a treat-to-target management approach in RA guidelines, but only a proportion of patients reach their targets, according to Dr. England. In his series of analyses, Dr. England has been exploring the role of comorbidities as one of the contributing factors.
Looking for real-world data, Dr. England evaluated comorbidities in the Veterans Affairs Rheumatoid Arthritis Registry, which has a male predominant population, the National Databank for Rheumatic Diseases, which is female predominant, the Truven Health Analytics MarketScan Database, and the Rheumatology Informatics System for Effectiveness Registry (RISE).
Comorbidities accrue more quickly in RA patients
All of these real-world data support the premise that comorbidities are higher in patients with RA than in those without, and show that the burden of comorbidities rises more quickly in patients with RA. For example, the average number of comorbidities in the MarketScan database of recently diagnosed RA patients was 2.6. Five years later, the average doubled to more than 5. For those without RA, the average at the baseline evaluation was 1.6 and remained below 3 at 5 years.
For the burden of comorbidities in RA, Dr. England prefers the term “multimorbidity” because he believes it captures the interconnections of these chronic diseases, many of which trigger or exacerbate others. When he looked at health history 2 years before the RA diagnosis, multimorbidities were already somewhat higher, but he found that burden “takes off” in the peri-diagnostic period and climbs steeply thereafter.
“The data tell us that multimorbidity becomes more problematic throughout the RA disease course,” said Dr. England, who published some of these data only a few weeks prior to his presentation.
In one effort to evaluate how multimorbidity affects treatment choices and outcome, he selected patients with persistently active disease from the RISE registry, a group expected to be candidates for a treatment change or escalation. The data suggested patients with multimorbidity were less likely than were those without to receive a change of therapy in response to their active disease, but it also demonstrated that patients with multimorbidity were less likely to achieve remission or low disease activity even if the medications were changed.
Each comorbidity lowers odds of remission
When relative burden of comorbidities was assessed by RxRisk score, a validated medication-based measure of chronic disease that recognizes 46 categories of chronic conditions, there was about a 5% lower odds ratio for each RxRisk unit of increase in comorbidity. The relationship was consistent across various cohorts of patients evaluated, according to Dr. England.
When looking for patterns of comorbidities in these large datasets using machine learning, Dr. England reported that there were “striking” relationships between organ systems. This included a pattern of cardiometabolic multimorbidity, cardiopulmonary multimorbidity, and mental health and chronic pain multimorbidity. Surprisingly, the same patterns could be identified in those with or without RA, but the prevalence differed.
“RA was closely associated with all of these different multimorbidity patterns, but the odds of having these patterns were one- to threefold greater,” Dr. England reported.
“The multimorbidity pattern most closely associated with RA was mental health and chronic pain,” he added, noting that the same results were observed across the datasets evaluated.
The implication of this work is that multimorbidity exerts an adverse effect on the course of RA and might be an appropriate target of therapies to improve RA outcomes. Although Dr. England called for better tools to measure multimorbidity and consider how it can be addressed systematically in RA patients with the intention of improving RA control, he believes this is an important direction of research.
“What our data show is that we need to begin to think more holistically about these other diseases in RA patients,” he said.
Others support targeting of comorbidities
Vanessa L. Kronzer, MD, a rheumatology fellow at Mayo Clinic, Rochester, Minn., agreed. An author of a study that identified 11 comorbidities significantly associated with RA, either as conditions that predispose to RA or that commonly develop in patients with RA, Dr. Kronzer has drawn the same conclusion in regard to targeting comorbidities in the RA patient.
“Based on mounting evidence that multimorbidity is associated with RA and RA disease activity, taking a broader view of the patient as a whole and his or her comorbidities may help us to not only predict RA but also achieve over disease-specific goals,” Dr. Kronzer said in an interview.
“I suspect that certain comorbidities, perhaps depression as an example, may play a particularly strong role in perpetuating high RA disease activity,” she added. She considers this a ripe area of study for improving clinical strategies in RA.
“Finding out which ones [perpetuate RA] and targeting them could be a reasonable approach to moving forward,” Dr. Kronzer said.
Dr. England and Dr. Kronzer reported having no potential conflicts of interest.
An increasing number of comorbidities in patients with rheumatoid arthritis correlates with a lower likelihood of reaching treatment targets, according to an analysis conducted with a series of large real-world datasets and presented in a symposium sponsored by the Rheumatology Research Foundation.
When compared to those with the lowest burden of comorbidity in one of these analyses, those with the highest had a nearly 50% lower likelihood (odds ratio, 0.54; 95% confidence interval [CI], 0.34-0.85) of achieving low disease activity or remission, according to Bryant England, MD, PhD, assistant professor in the division of rheumatology at the University of Nebraska, Omaha.
“Patients with more comorbidities struggle to reach treatment targets,” Dr. England said. In the treatment of RA, “we typically focus only on the joints, but these data suggest we need to begin to think more holistically about managing these patients.”
Both the American College of Rheumatology (ACR) and the European Alliance of Associations for Rheumatology (EULAR) endorse a treat-to-target management approach in RA guidelines, but only a proportion of patients reach their targets, according to Dr. England. In his series of analyses, Dr. England has been exploring the role of comorbidities as one of the contributing factors.
Looking for real-world data, Dr. England evaluated comorbidities in the Veterans Affairs Rheumatoid Arthritis Registry, which has a male predominant population, the National Databank for Rheumatic Diseases, which is female predominant, the Truven Health Analytics MarketScan Database, and the Rheumatology Informatics System for Effectiveness Registry (RISE).
Comorbidities accrue more quickly in RA patients
All of these real-world data support the premise that comorbidities are higher in patients with RA than in those without, and show that the burden of comorbidities rises more quickly in patients with RA. For example, the average number of comorbidities in the MarketScan database of recently diagnosed RA patients was 2.6. Five years later, the average doubled to more than 5. For those without RA, the average at the baseline evaluation was 1.6 and remained below 3 at 5 years.
For the burden of comorbidities in RA, Dr. England prefers the term “multimorbidity” because he believes it captures the interconnections of these chronic diseases, many of which trigger or exacerbate others. When he looked at health history 2 years before the RA diagnosis, multimorbidities were already somewhat higher, but he found that burden “takes off” in the peri-diagnostic period and climbs steeply thereafter.
“The data tell us that multimorbidity becomes more problematic throughout the RA disease course,” said Dr. England, who published some of these data only a few weeks prior to his presentation.
In one effort to evaluate how multimorbidity affects treatment choices and outcome, he selected patients with persistently active disease from the RISE registry, a group expected to be candidates for a treatment change or escalation. The data suggested patients with multimorbidity were less likely than were those without to receive a change of therapy in response to their active disease, but it also demonstrated that patients with multimorbidity were less likely to achieve remission or low disease activity even if the medications were changed.
Each comorbidity lowers odds of remission
When relative burden of comorbidities was assessed by RxRisk score, a validated medication-based measure of chronic disease that recognizes 46 categories of chronic conditions, there was about a 5% lower odds ratio for each RxRisk unit of increase in comorbidity. The relationship was consistent across various cohorts of patients evaluated, according to Dr. England.
When looking for patterns of comorbidities in these large datasets using machine learning, Dr. England reported that there were “striking” relationships between organ systems. This included a pattern of cardiometabolic multimorbidity, cardiopulmonary multimorbidity, and mental health and chronic pain multimorbidity. Surprisingly, the same patterns could be identified in those with or without RA, but the prevalence differed.
“RA was closely associated with all of these different multimorbidity patterns, but the odds of having these patterns were one- to threefold greater,” Dr. England reported.
“The multimorbidity pattern most closely associated with RA was mental health and chronic pain,” he added, noting that the same results were observed across the datasets evaluated.
The implication of this work is that multimorbidity exerts an adverse effect on the course of RA and might be an appropriate target of therapies to improve RA outcomes. Although Dr. England called for better tools to measure multimorbidity and consider how it can be addressed systematically in RA patients with the intention of improving RA control, he believes this is an important direction of research.
“What our data show is that we need to begin to think more holistically about these other diseases in RA patients,” he said.
Others support targeting of comorbidities
Vanessa L. Kronzer, MD, a rheumatology fellow at Mayo Clinic, Rochester, Minn., agreed. An author of a study that identified 11 comorbidities significantly associated with RA, either as conditions that predispose to RA or that commonly develop in patients with RA, Dr. Kronzer has drawn the same conclusion in regard to targeting comorbidities in the RA patient.
“Based on mounting evidence that multimorbidity is associated with RA and RA disease activity, taking a broader view of the patient as a whole and his or her comorbidities may help us to not only predict RA but also achieve over disease-specific goals,” Dr. Kronzer said in an interview.
“I suspect that certain comorbidities, perhaps depression as an example, may play a particularly strong role in perpetuating high RA disease activity,” she added. She considers this a ripe area of study for improving clinical strategies in RA.
“Finding out which ones [perpetuate RA] and targeting them could be a reasonable approach to moving forward,” Dr. Kronzer said.
Dr. England and Dr. Kronzer reported having no potential conflicts of interest.
An increasing number of comorbidities in patients with rheumatoid arthritis correlates with a lower likelihood of reaching treatment targets, according to an analysis conducted with a series of large real-world datasets and presented in a symposium sponsored by the Rheumatology Research Foundation.
When compared to those with the lowest burden of comorbidity in one of these analyses, those with the highest had a nearly 50% lower likelihood (odds ratio, 0.54; 95% confidence interval [CI], 0.34-0.85) of achieving low disease activity or remission, according to Bryant England, MD, PhD, assistant professor in the division of rheumatology at the University of Nebraska, Omaha.
“Patients with more comorbidities struggle to reach treatment targets,” Dr. England said. In the treatment of RA, “we typically focus only on the joints, but these data suggest we need to begin to think more holistically about managing these patients.”
Both the American College of Rheumatology (ACR) and the European Alliance of Associations for Rheumatology (EULAR) endorse a treat-to-target management approach in RA guidelines, but only a proportion of patients reach their targets, according to Dr. England. In his series of analyses, Dr. England has been exploring the role of comorbidities as one of the contributing factors.
Looking for real-world data, Dr. England evaluated comorbidities in the Veterans Affairs Rheumatoid Arthritis Registry, which has a male predominant population, the National Databank for Rheumatic Diseases, which is female predominant, the Truven Health Analytics MarketScan Database, and the Rheumatology Informatics System for Effectiveness Registry (RISE).
Comorbidities accrue more quickly in RA patients
All of these real-world data support the premise that comorbidities are higher in patients with RA than in those without, and show that the burden of comorbidities rises more quickly in patients with RA. For example, the average number of comorbidities in the MarketScan database of recently diagnosed RA patients was 2.6. Five years later, the average doubled to more than 5. For those without RA, the average at the baseline evaluation was 1.6 and remained below 3 at 5 years.
For the burden of comorbidities in RA, Dr. England prefers the term “multimorbidity” because he believes it captures the interconnections of these chronic diseases, many of which trigger or exacerbate others. When he looked at health history 2 years before the RA diagnosis, multimorbidities were already somewhat higher, but he found that burden “takes off” in the peri-diagnostic period and climbs steeply thereafter.
“The data tell us that multimorbidity becomes more problematic throughout the RA disease course,” said Dr. England, who published some of these data only a few weeks prior to his presentation.
In one effort to evaluate how multimorbidity affects treatment choices and outcome, he selected patients with persistently active disease from the RISE registry, a group expected to be candidates for a treatment change or escalation. The data suggested patients with multimorbidity were less likely than were those without to receive a change of therapy in response to their active disease, but it also demonstrated that patients with multimorbidity were less likely to achieve remission or low disease activity even if the medications were changed.
Each comorbidity lowers odds of remission
When relative burden of comorbidities was assessed by RxRisk score, a validated medication-based measure of chronic disease that recognizes 46 categories of chronic conditions, there was about a 5% lower odds ratio for each RxRisk unit of increase in comorbidity. The relationship was consistent across various cohorts of patients evaluated, according to Dr. England.
When looking for patterns of comorbidities in these large datasets using machine learning, Dr. England reported that there were “striking” relationships between organ systems. This included a pattern of cardiometabolic multimorbidity, cardiopulmonary multimorbidity, and mental health and chronic pain multimorbidity. Surprisingly, the same patterns could be identified in those with or without RA, but the prevalence differed.
“RA was closely associated with all of these different multimorbidity patterns, but the odds of having these patterns were one- to threefold greater,” Dr. England reported.
“The multimorbidity pattern most closely associated with RA was mental health and chronic pain,” he added, noting that the same results were observed across the datasets evaluated.
The implication of this work is that multimorbidity exerts an adverse effect on the course of RA and might be an appropriate target of therapies to improve RA outcomes. Although Dr. England called for better tools to measure multimorbidity and consider how it can be addressed systematically in RA patients with the intention of improving RA control, he believes this is an important direction of research.
“What our data show is that we need to begin to think more holistically about these other diseases in RA patients,” he said.
Others support targeting of comorbidities
Vanessa L. Kronzer, MD, a rheumatology fellow at Mayo Clinic, Rochester, Minn., agreed. An author of a study that identified 11 comorbidities significantly associated with RA, either as conditions that predispose to RA or that commonly develop in patients with RA, Dr. Kronzer has drawn the same conclusion in regard to targeting comorbidities in the RA patient.
“Based on mounting evidence that multimorbidity is associated with RA and RA disease activity, taking a broader view of the patient as a whole and his or her comorbidities may help us to not only predict RA but also achieve over disease-specific goals,” Dr. Kronzer said in an interview.
“I suspect that certain comorbidities, perhaps depression as an example, may play a particularly strong role in perpetuating high RA disease activity,” she added. She considers this a ripe area of study for improving clinical strategies in RA.
“Finding out which ones [perpetuate RA] and targeting them could be a reasonable approach to moving forward,” Dr. Kronzer said.
Dr. England and Dr. Kronzer reported having no potential conflicts of interest.
FROM THE RHEUMATOLOGY RESEARCH FOUNDATION SUMMER SERIES
FDA fully approves Pfizer COVID-19 vaccine
The Food and Drug Administration has granted a biological license application, more commonly known as “full approval,” to the Pfizer-BioNTech COVID-19 vaccine.
It is the first COVID-19 vaccine to be fully licensed in the United States. It will be marketed under the trade name Comirnaty.
The approval applies to individuals ages 16 years and older. The vaccine is still available for emergency use for those ages 12-15.
The FDA’s stamp of approval is somewhat anticlimactic, following months of real-world use and millions of doses doled out to the general population. It comes after months of scrutiny by the agency of the clinical trial data.
Still, the approval puts the vaccines on firmer legal footing and is expected to spur a raft of new vaccination requirements by employers, schools, and universities.
“The FDA approval is the gold standard,” President Joe Biden said from the White House. “Those who have been waiting for full approval should go and get your shot now.”
“It could save your life or the lives of those you love,” he said.
Biden also called on businesses to mandate COVID vaccines for their employees.
Indeed, soon after the approval was announced, Defense Secretary Lloyd Austin said the vaccines would be required for all 1.4 million active duty service members.
Public health advocates have seen full approval as an important tool to increase U.S. vaccination rates and had criticized the FDA for taking so long to grant the license.
In a news briefing on the approval, Peter Marks, MD, director of the FDA’s Center for Biologics Evaluation and Research, said the agency had not dragged its feet.
Marks noted that his team had reviewed tens of thousands of pages of clinical trial data -- down to the level of individual patients. They also inspected clinical trial sites and manufacturing facilities, and reviewed information gathered after the vaccines were authorized for use.
“It’s been 97 days since Pfizer completed the role of its [application for approval] and the clock started, which means that we completed this in about 40% of the normal clock time for a submission of this magnitude,” he said. “People worked day and night.”
The agency resisted pressure to speed up its process, saying a thorough review was necessary to ensure public confidence.
“While millions of people have already safely received COVID-19 vaccines, we recognize that for some, the FDA approval of a vaccine may now instill additional confidence to get vaccinated. Today’s milestone puts us one step closer to altering the course of this pandemic in the U.S.,” acting FDA Commissioner Janet Woodcock said in a FDA news release.
Experts agreed the move would increase public confidence.
“I don't expect a big line outside of vaccination sites this afternoon or tomorrow morning, but it will persuade some,” said William Schaffner, MD, a professor of infectious diseases at Vanderbilt University in Nashville.
A recent Kaiser Family Foundation poll found that 3 in 10 unvaccinated adults said they would be more likely to get vaccinated if the vaccines were given full approval.
More importantly, Schaffner said, the FDA’s approval would lay the groundwork for vaccine mandates. “I think those kinds of mandates are going to be necessary to get us up over 80% vaccinated.”
In granting the approval, the agency reviewed a record amount of data from more than 40,000 people who took part in clinical trials. About 12,000 recipients have been followed for at least 6 months, the agency said.
The FDA also reviewed safety data collected since it issued its emergency use authorization for the shots in December.
Based on the results from the clinical trials, the vaccine was 91% effective at preventing COVID-19 disease. But that estimate came from data collected before the Delta variant became widespread.
The most commonly reported side effects in the clinical trials were pain, redness and swelling at the injection site, fatigue, headache, muscle or joint pain, chills, and fever.
The FDA said the vaccine is effective in preventing COVID-19 and potentially serious outcomes, including hospitalization and death.
Based on safety data reviewed since the two-dose vaccine was approved, the FDA said the data demonstrates a higher risk for heart inflammation -- clinically known as myocarditis or pericarditis -- especially within 7 days after the second dose of the shots. The risk is highest for men under age 40, compared to women and older men.
The prescription information includes warnings about these risks. The FDA said the drugmakers must continue to study the risks and long-term effects on people who have myocarditis after vaccination.
A version of this article first appeared on Medscape.com.
This article was updated on 8/24/21.
The Food and Drug Administration has granted a biological license application, more commonly known as “full approval,” to the Pfizer-BioNTech COVID-19 vaccine.
It is the first COVID-19 vaccine to be fully licensed in the United States. It will be marketed under the trade name Comirnaty.
The approval applies to individuals ages 16 years and older. The vaccine is still available for emergency use for those ages 12-15.
The FDA’s stamp of approval is somewhat anticlimactic, following months of real-world use and millions of doses doled out to the general population. It comes after months of scrutiny by the agency of the clinical trial data.
Still, the approval puts the vaccines on firmer legal footing and is expected to spur a raft of new vaccination requirements by employers, schools, and universities.
“The FDA approval is the gold standard,” President Joe Biden said from the White House. “Those who have been waiting for full approval should go and get your shot now.”
“It could save your life or the lives of those you love,” he said.
Biden also called on businesses to mandate COVID vaccines for their employees.
Indeed, soon after the approval was announced, Defense Secretary Lloyd Austin said the vaccines would be required for all 1.4 million active duty service members.
Public health advocates have seen full approval as an important tool to increase U.S. vaccination rates and had criticized the FDA for taking so long to grant the license.
In a news briefing on the approval, Peter Marks, MD, director of the FDA’s Center for Biologics Evaluation and Research, said the agency had not dragged its feet.
Marks noted that his team had reviewed tens of thousands of pages of clinical trial data -- down to the level of individual patients. They also inspected clinical trial sites and manufacturing facilities, and reviewed information gathered after the vaccines were authorized for use.
“It’s been 97 days since Pfizer completed the role of its [application for approval] and the clock started, which means that we completed this in about 40% of the normal clock time for a submission of this magnitude,” he said. “People worked day and night.”
The agency resisted pressure to speed up its process, saying a thorough review was necessary to ensure public confidence.
“While millions of people have already safely received COVID-19 vaccines, we recognize that for some, the FDA approval of a vaccine may now instill additional confidence to get vaccinated. Today’s milestone puts us one step closer to altering the course of this pandemic in the U.S.,” acting FDA Commissioner Janet Woodcock said in a FDA news release.
Experts agreed the move would increase public confidence.
“I don't expect a big line outside of vaccination sites this afternoon or tomorrow morning, but it will persuade some,” said William Schaffner, MD, a professor of infectious diseases at Vanderbilt University in Nashville.
A recent Kaiser Family Foundation poll found that 3 in 10 unvaccinated adults said they would be more likely to get vaccinated if the vaccines were given full approval.
More importantly, Schaffner said, the FDA’s approval would lay the groundwork for vaccine mandates. “I think those kinds of mandates are going to be necessary to get us up over 80% vaccinated.”
In granting the approval, the agency reviewed a record amount of data from more than 40,000 people who took part in clinical trials. About 12,000 recipients have been followed for at least 6 months, the agency said.
The FDA also reviewed safety data collected since it issued its emergency use authorization for the shots in December.
Based on the results from the clinical trials, the vaccine was 91% effective at preventing COVID-19 disease. But that estimate came from data collected before the Delta variant became widespread.
The most commonly reported side effects in the clinical trials were pain, redness and swelling at the injection site, fatigue, headache, muscle or joint pain, chills, and fever.
The FDA said the vaccine is effective in preventing COVID-19 and potentially serious outcomes, including hospitalization and death.
Based on safety data reviewed since the two-dose vaccine was approved, the FDA said the data demonstrates a higher risk for heart inflammation -- clinically known as myocarditis or pericarditis -- especially within 7 days after the second dose of the shots. The risk is highest for men under age 40, compared to women and older men.
The prescription information includes warnings about these risks. The FDA said the drugmakers must continue to study the risks and long-term effects on people who have myocarditis after vaccination.
A version of this article first appeared on Medscape.com.
This article was updated on 8/24/21.
The Food and Drug Administration has granted a biological license application, more commonly known as “full approval,” to the Pfizer-BioNTech COVID-19 vaccine.
It is the first COVID-19 vaccine to be fully licensed in the United States. It will be marketed under the trade name Comirnaty.
The approval applies to individuals ages 16 years and older. The vaccine is still available for emergency use for those ages 12-15.
The FDA’s stamp of approval is somewhat anticlimactic, following months of real-world use and millions of doses doled out to the general population. It comes after months of scrutiny by the agency of the clinical trial data.
Still, the approval puts the vaccines on firmer legal footing and is expected to spur a raft of new vaccination requirements by employers, schools, and universities.
“The FDA approval is the gold standard,” President Joe Biden said from the White House. “Those who have been waiting for full approval should go and get your shot now.”
“It could save your life or the lives of those you love,” he said.
Biden also called on businesses to mandate COVID vaccines for their employees.
Indeed, soon after the approval was announced, Defense Secretary Lloyd Austin said the vaccines would be required for all 1.4 million active duty service members.
Public health advocates have seen full approval as an important tool to increase U.S. vaccination rates and had criticized the FDA for taking so long to grant the license.
In a news briefing on the approval, Peter Marks, MD, director of the FDA’s Center for Biologics Evaluation and Research, said the agency had not dragged its feet.
Marks noted that his team had reviewed tens of thousands of pages of clinical trial data -- down to the level of individual patients. They also inspected clinical trial sites and manufacturing facilities, and reviewed information gathered after the vaccines were authorized for use.
“It’s been 97 days since Pfizer completed the role of its [application for approval] and the clock started, which means that we completed this in about 40% of the normal clock time for a submission of this magnitude,” he said. “People worked day and night.”
The agency resisted pressure to speed up its process, saying a thorough review was necessary to ensure public confidence.
“While millions of people have already safely received COVID-19 vaccines, we recognize that for some, the FDA approval of a vaccine may now instill additional confidence to get vaccinated. Today’s milestone puts us one step closer to altering the course of this pandemic in the U.S.,” acting FDA Commissioner Janet Woodcock said in a FDA news release.
Experts agreed the move would increase public confidence.
“I don't expect a big line outside of vaccination sites this afternoon or tomorrow morning, but it will persuade some,” said William Schaffner, MD, a professor of infectious diseases at Vanderbilt University in Nashville.
A recent Kaiser Family Foundation poll found that 3 in 10 unvaccinated adults said they would be more likely to get vaccinated if the vaccines were given full approval.
More importantly, Schaffner said, the FDA’s approval would lay the groundwork for vaccine mandates. “I think those kinds of mandates are going to be necessary to get us up over 80% vaccinated.”
In granting the approval, the agency reviewed a record amount of data from more than 40,000 people who took part in clinical trials. About 12,000 recipients have been followed for at least 6 months, the agency said.
The FDA also reviewed safety data collected since it issued its emergency use authorization for the shots in December.
Based on the results from the clinical trials, the vaccine was 91% effective at preventing COVID-19 disease. But that estimate came from data collected before the Delta variant became widespread.
The most commonly reported side effects in the clinical trials were pain, redness and swelling at the injection site, fatigue, headache, muscle or joint pain, chills, and fever.
The FDA said the vaccine is effective in preventing COVID-19 and potentially serious outcomes, including hospitalization and death.
Based on safety data reviewed since the two-dose vaccine was approved, the FDA said the data demonstrates a higher risk for heart inflammation -- clinically known as myocarditis or pericarditis -- especially within 7 days after the second dose of the shots. The risk is highest for men under age 40, compared to women and older men.
The prescription information includes warnings about these risks. The FDA said the drugmakers must continue to study the risks and long-term effects on people who have myocarditis after vaccination.
A version of this article first appeared on Medscape.com.
This article was updated on 8/24/21.
Shouldn’t docs who spread false COVID-19 info lose their licenses?
A tall, distinguished-looking physician in shirtsleeves and suspenders walked to the microphone at the Mt. Vernon, Ind., school board meeting on a Friday evening in early August. He launched into an impassioned, 7-minute attack on the public health establishment’s medical guidelines for COVID-19.
“The Center for Disease Control and the Indiana State [Department] of Health are giving you very bad scientific guidance,” said Daniel Stock, MD, a primary care physician with a concierge practice in Noblesville, Ind., He described himself as a “functional family medicine physician,” though he is not board certified in family medicine.
Dr. Stock told the school board members that COVID-19 vaccines are counterproductive because they make coronavirus infections worse. He claimed his treatment of “over 15” COVID-19 patients with vitamin D, ivermectin, and zinc has kept them out of the hospital, and that those treatments reduce mortality risk from the disease by 75%. (A study released in mid-August found that ivermectin is ineffective in treating COVID-19).
In response to Dr. Stock’s remarks, the state health department quickly issued a statement reaffirming that COVID-19 vaccines “are highly effective at preventing hospitalizations and deaths.” But by then, the YouTube video of Dr. Stock’s comments had garnered nearly 600,000 views as of Aug. 12 and had been shared over 10,000 times on Facebook. Opponents of COVID-19 vaccines and masking policies across the country have been citing his comments.
Across the country, state medical licensing boards and state and national medical associations are struggling with how to respond to scientifically baseless public statements about COVID-19 by some physicians such as Dr. Stock. They fear such statements are increasing public confusion and are heightening political conflict. Physicians accused of spreading false information include public officials such as Scott Atlas, MD, who served as President Donald Trump’s COVID-19 advisor, and Kentucky Sen. Rand Paul, an ophthalmologist, whose YouTube account was temporarily suspended in August after he posted a video disputing the effectiveness of masking in stopping the spread of COVID-19.
“That’s the problem – those types of viral videos of someone somewhere who thinks they know something the rest of us don’t,” lamented Jennifer Bryan, MD, board chair of the Mississippi State Medical Association. “I don’t know any good reason why a physician should be advising against vaccination. It’s appropriate for medical boards to look into those situations.”
The Federation of State Medical Boards agrees. In July, it warned that physicians who willfully spread false information about COVID-19 risk suspension or revocation of their medical license. The federation cited a “dramatic increase in the dissemination of COVID-19 vaccine misinformation and disinformation by physicians.” That’s particularly dangerous, it said, because physicians enjoy a high degree of public credibility.
Medical boards will particularly examine cases in which there is a pattern of misinformation or disinformation showing that a physician poses a continuing threat to public health, said Hank Chaudhry, DO, the federation’s CEO. In some cases, he said, boards have contacted physicians and have persuaded them to voluntarily refrain from making false public statements, without taking disciplinary action.
“Words matter,” he said. “Physicians have a really big platform, whether they realize it or not. Misinformation or disinformation in the context of COVID can not only cause harm but also death. We felt it was appropriate to remind physicians to be careful.”
Although medical leaders stress that most physicians are promoting solid science on COVID-19, the London-based Center for Countering Digital Hate, in a May report titled “The Disinformation Dozen,” named four U.S. physicians among 12 people who it said produce 65% of the misleading claims and lies about COVID-19 vaccines that abound on Facebook, Instagram, and Twitter. The leading spreader of false claims, the group said, is Joseph Mercola, MD, an Illinois-licensed osteopath living in Cape Coral, Fla. He did not respond to requests for comment.
But so far, state licensing boards and federal and state medical associations generally have been reluctant to discipline or publicly call out physicians who have spread misinformation about the causes, treatments, vaccines, and prevention strategies for COVID-19. Some of these physicians, such as Dr. Mercola, have a long history predating the COVID-19 pandemic of disseminating scientifically baseless information, often in connection with their marketing of products and services.
For instance, the Medical Licensing Board of Indiana and the state attorney general’s office, which brings medical disciplinary actions, declined to comment on Dr. Stock’s public statements at the August school board meeting. When asked about Dr. Stock, the Indiana State Medical Association, without mentioning his name, said: “We urge Hoosier physicians to share the proven facts [about public health measures recommended by the CDC and the Indiana Department of Health] with their patients and their communities.” Dr. Stock did not respond to a request for comment.
Experts say state medical boards are ill equipped and are often unwilling to address the challenge of disciplining physicians who disseminate dangerously false medical information. That enforcement gap is particularly troubling in the middle of a deadly pandemic such as this one.
“Unless you can show a harm to an individual patient, it’s pretty tough to get the boards to do much,” said Art Caplan, PhD, a professor of bioethics at New York University. “I wish they would, but they just don’t.”
That’s partly because state laws require the boards to engage in lengthy, confidential investigations and adversarial legal processes before imposing disciplinary actions. The laws generally require patients or members of the public to file a complaint before an investigation can start. Some states, however, do allow their medical boards to take rapid emergency action if a physician poses an immediate threat to patients or the public.
Another hurdle is that medical boards that seek to sanction physicians for making dangerously misleading public statements could face lawsuits alleging that such actions violate the physicians’ constitutional free speech rights or their professional autonomy.
“We have free speech, and you can get away with a lot of stuff,” said Stephen Barrett, MD, who for many years has critically documented examples of medical fraud on his website, Quackwatch. “Some doctors would sue if they were challenged by medical boards, and I’m not sure the boards would win that court fight. People have written books with advice that killed people, and I’m not aware of a single case where the author was disciplined.”
In addition, it’s not clear that U.S. physicians who are not government officials have any legal obligation – as opposed to a moral obligation – to the government or the public to promote public health, said Jonathan Moreno, PhD, a professor of medical ethics at University of Pennsylvania, Philadelphia. “Is transmitting misinformation about COVID-19 public health malpractice?” he asked. “Do we as a society see physicians having a special role as guides in an emergency? I’d like to think we do, but we don’t have a strong tradition like that in the U.S.”
But California State Sen. Richard Pan, MD, a pediatrician who represents the Sacramento area, doesn’t buy the arguments about why medical boards can’t discipline physicians for spreading misinformation. He successfully sponsored a 2019 bill that strengthens the medical board’s ability to discipline physicians who dole out medically unjustified vaccine exemptions to children.
“A medical license is a privilege. It’s an imprimatur from the state that the person is someone who upholds professional standards,” Dr. Pan said. “If someone is intentionally spreading disinformation for personal gain and that’s putting the public at risk, the medical board has a duty to act.”
There have been only a few publicly announced disciplinary actions related to COVID-19 misinformation so far.
Last December, the Oregon Medical Board, on an emergency basis, suspended the license of Steven LaTulippe, MD, of Dallas, Ore. He had publicly announced that he and his staff were not wearing masks in his clinic. In addition, he compared COVID-19 to the common cold and denied the governor’s legal authority to adopt public health protection measures. A recorded message on his office phone said he’s challenging the licensure action in court.
Last January, the Medical Board of California made Thomas Cowan, MD, of San Francisco surrender his license after Dr. Cowan posted a YouTube video, which went viral last year, that claimed that 5G Internet networks cause COVID-19. He did not respond to a request for comment.
In May, the College of Physicians and Surgeons of British Columbia reprimanded Stephen Malthouse, MD, and forbade him from speaking on issues related to COVID-19. He had written a widely circulated open letter to the province’s chief health office claiming that the pandemic was “over” and that measures to control the spread of COVID-19 were worse than the virus. He has challenged the disciplinary action in court, alleging it violates his right to free speech.
Attacking the problem from a different angle, the U.S. Federal Trade Commission has issued enforcement actions in cases in which physicians and other health care professionals engaged in deceptive business practices related to COVID-19. That approach may be applicable to a number of physicians accused of spreading COVID-19 misinformation, who allegedly have done so at least partly to sell unproven products and services to prevent or treat the disease.
In June, the FTC settled a case against Stephen Meis, MD, of Porterville, Calif. The settlement required that he stop making unsupported claims that his company’s dietary supplements effectively treat COVID-19 symptoms and that he pay $103,420 in refunds to defrauded customers.
State medical boards in the United States generally are not allowed to disclose investigations or disciplinary processes until they finalize a disciplinary action, so other investigations that have not been publicly disclosed may be pending.
A spokesperson for the Medical Board of California said the board is aware of questionable statements about COVID-19 made by several physicians and “will be looking into it.” That comment was in response to a question about statements made at a news conference last year by two Bakersfield emergency physicians, Artin Massihi, MD, and Dan Erickson, DO. They claimed that their COVID-19 testing data showed that the virus is not that dangerous. Dr. Erickson is an osteopath and is regulated by the Osteopathic Medical Board of California.
The two physicians’ news conference prompted an unusual joint statement from the American College of Emergency Medicine and the American Academy of Emergency Medicine in April 2020 declaring that they “emphatically condemn” Dr. Massihi’s and Dr. Erickson’s “reckless and untested musings.” The groups added that it appeared that the physicians issued the comments “to advance their personal financial interests without regard for the public’s health.”
Neither Dr. Massihi nor Dr. Erickson responded to a request for comment.
As for the physician dubbed by the Center for Countering Digital Hate as the world’s most influential spreader of COVID-19 misinformation on social media: No recent public complaints have been filed, and no disciplinary action has been taken against Dr. Mercola, according to a spokesman for the Illinois Department of Financial and Professional Regulation.
According to court records, Dr. Mercola faced disciplinary complaints from the Illinois board in the early 2000s for allegedly providing false and potentially harmful medical advice on his website. There is no record of any final disciplinary action taken against him.
In widely disseminated online posts, Dr. Mercola has called the COVID-19 pandemic a “scam” and said “forced vaccination” is part of a plan to re-set the global economic system. He called COVID-19 vaccines “a medical fraud,” claiming they “alter your genetic coding.” In February, the U.S. Food and Drug Administration ordered Dr. Mercola to stop saying on his website that various vitamins and dietary supplements he sells through his website are effective in preventing or treating COVID-19.
The New York Times reported in July that Dr. Mercola’s English-language Facebook page has more than 1.7 million followers, that his Spanish-language page has one million, and that he has 300,000 followers on Twitter and 400,000 on YouTube.
In August, Dr. Mercola announced that he was deleting the large archive of articles he’s written on his website but would continue to post articles every day that would be available on the site for only 48 hours. He explained his decision by saying he’s facing “blatant censorship” as part of a “McCarthyism-like attack” from “the sitting President of the United States.” He encouraged people to read his book, “The Truth about COVID-19.”
The lack of action against Dr. Mercola for his lengthy list of scientifically unfounded statements and marketing claims about COVID-19 and other medical conditions infuriates Quackwatch’s Dr. Barrett. He’s amazed that the Illinois board did not discipline Dr. Mercola despite a number of enforcement actions against him by the FTC and the FDA.
“If a doctor were to say to a patient, ‘Don’t wear a mask and don’t get vaccinated,’ the doctor would be held responsible for a bad outcome,” he said. “But if you say it to millions and as a direct result a dozen people die, shouldn’t the doctor also be held responsible for that misinformation? I think he should lose his license.”
Another of the four physicians cited in the “Disinformation Dozen” report is Sherri Tenpenny, DO, an osteopath licensed in Ohio, who has published posts on social media advocating against masking, testing, and vaccines to prevent COVID-19 infections. A spokesperson for the State Medical Board of Ohio said Dr. Tenpenny’s license expires on Oct. 1, 2021, and that any investigation would be confidential. She added that grounds for disciplinary action include “making a false, fraudulent, deceptive, or misleading statement in relation to the practice of medicine and surgery.” Dr. Tenpenny could not be reached for comment.
A third physician named in the report is Christiane Northrup, MD, an ob.gyn. formerly licensed in Maine, who has published posts advocating unproven cures for COVID-19 and claiming that vaccines increase chronic illness. Dennis Smith, executive director of the Maine Board of Licensure in Medicine, said the board received complaints about Dr. Northrup’s posts but can’t act because she withdrew her Maine license in 2015. He added that the Maine board can issue sanctions against physicians who engage in fraud, deceit, or misrepresentation or who post scientifically unfounded statements online.
The fourth physician identified in the “Disinformation Dozen” report is Rashid Buttar, DO, an osteopath practicing in Mooresville, N.C., who has claimed in social media posts that COVID-19 vaccines cause infertility and that COVID-19 tests contain living microorganisms. A spokeswoman for the North Carolina Medical Board said she could not confirm or deny the existence of any investigation of Dr. Buttar, who signed a consent order with the medical board in 2010 following charges of exorbitant fees, worthless tests and treatment, and false diagnoses. Undisclosed conditions were placed on his medical license in 2013. The spokesperson added that the board would investigate any information alleging that a physician spread false information about COVID-19.
Another physician who has caused widespread consternation over scientifically unfounded statements about COVID-19 is Simone Gold, MD, formerly an emergency department physician in Los Angeles. She founded a group called America’s Frontline Doctors, which filed a federal lawsuit in Alabama this spring to block the FDA from issuing an emergency use authorization allowing teenagers to receive COVID-19 vaccinations. She called the vaccines “an experimental biological agent whose harms are well-documented.”
Last summer, Dr. Gold and other physicians in her group held a news conference on the steps of the U.S. Supreme Court Building promoting hydroxychloroquine as a COVID-19 treatment. They declared that masks don’t work and that the virus isn’t deadly, and made other false claims. The news conference was livestreamed by conservative media outlets, was promoted on Twitter by then-President Trump and his family, and was viewed online more than 14 million times.
One of the participating physicians, Stella Immanuel, MD, of Houston, claimed in a video that went viral that she had successfully used hydroxychloroquine for more than 400 patients to cure the disease. In response, the Texas Medical Board, without naming Dr. Immanuel, warned that if it received a complaint about any physician who made a false claim about having a cure for COVID-19, it would investigate and potentially take disciplinary action.
Although no publicly known disciplinary action has been taken against Dr. Gold, she told The Washington Post last January that after participating in that July 2020 news conference, she was fired from her emergency department job at two hospitals and that she hasn’t worked as a physician since. Dr. Gold did not respond to a request for comment.
The outcome in her situation is consistent with the view of NYU’s Dr. Caplan that methods other than medical board discipline – such as action by employers, social media pressure, and reprimands from professional societies –will have to be used to hold physicians accountable for spreading COVID-19 misinformation.
“I’m disappointed to have to say it, but I don’t think medical boards are going to be effective,” he said. “We don’t know how to manage misinformation despite being in a plague. We just don’t.
A version of this article first appeared on Medscape.com.
A tall, distinguished-looking physician in shirtsleeves and suspenders walked to the microphone at the Mt. Vernon, Ind., school board meeting on a Friday evening in early August. He launched into an impassioned, 7-minute attack on the public health establishment’s medical guidelines for COVID-19.
“The Center for Disease Control and the Indiana State [Department] of Health are giving you very bad scientific guidance,” said Daniel Stock, MD, a primary care physician with a concierge practice in Noblesville, Ind., He described himself as a “functional family medicine physician,” though he is not board certified in family medicine.
Dr. Stock told the school board members that COVID-19 vaccines are counterproductive because they make coronavirus infections worse. He claimed his treatment of “over 15” COVID-19 patients with vitamin D, ivermectin, and zinc has kept them out of the hospital, and that those treatments reduce mortality risk from the disease by 75%. (A study released in mid-August found that ivermectin is ineffective in treating COVID-19).
In response to Dr. Stock’s remarks, the state health department quickly issued a statement reaffirming that COVID-19 vaccines “are highly effective at preventing hospitalizations and deaths.” But by then, the YouTube video of Dr. Stock’s comments had garnered nearly 600,000 views as of Aug. 12 and had been shared over 10,000 times on Facebook. Opponents of COVID-19 vaccines and masking policies across the country have been citing his comments.
Across the country, state medical licensing boards and state and national medical associations are struggling with how to respond to scientifically baseless public statements about COVID-19 by some physicians such as Dr. Stock. They fear such statements are increasing public confusion and are heightening political conflict. Physicians accused of spreading false information include public officials such as Scott Atlas, MD, who served as President Donald Trump’s COVID-19 advisor, and Kentucky Sen. Rand Paul, an ophthalmologist, whose YouTube account was temporarily suspended in August after he posted a video disputing the effectiveness of masking in stopping the spread of COVID-19.
“That’s the problem – those types of viral videos of someone somewhere who thinks they know something the rest of us don’t,” lamented Jennifer Bryan, MD, board chair of the Mississippi State Medical Association. “I don’t know any good reason why a physician should be advising against vaccination. It’s appropriate for medical boards to look into those situations.”
The Federation of State Medical Boards agrees. In July, it warned that physicians who willfully spread false information about COVID-19 risk suspension or revocation of their medical license. The federation cited a “dramatic increase in the dissemination of COVID-19 vaccine misinformation and disinformation by physicians.” That’s particularly dangerous, it said, because physicians enjoy a high degree of public credibility.
Medical boards will particularly examine cases in which there is a pattern of misinformation or disinformation showing that a physician poses a continuing threat to public health, said Hank Chaudhry, DO, the federation’s CEO. In some cases, he said, boards have contacted physicians and have persuaded them to voluntarily refrain from making false public statements, without taking disciplinary action.
“Words matter,” he said. “Physicians have a really big platform, whether they realize it or not. Misinformation or disinformation in the context of COVID can not only cause harm but also death. We felt it was appropriate to remind physicians to be careful.”
Although medical leaders stress that most physicians are promoting solid science on COVID-19, the London-based Center for Countering Digital Hate, in a May report titled “The Disinformation Dozen,” named four U.S. physicians among 12 people who it said produce 65% of the misleading claims and lies about COVID-19 vaccines that abound on Facebook, Instagram, and Twitter. The leading spreader of false claims, the group said, is Joseph Mercola, MD, an Illinois-licensed osteopath living in Cape Coral, Fla. He did not respond to requests for comment.
But so far, state licensing boards and federal and state medical associations generally have been reluctant to discipline or publicly call out physicians who have spread misinformation about the causes, treatments, vaccines, and prevention strategies for COVID-19. Some of these physicians, such as Dr. Mercola, have a long history predating the COVID-19 pandemic of disseminating scientifically baseless information, often in connection with their marketing of products and services.
For instance, the Medical Licensing Board of Indiana and the state attorney general’s office, which brings medical disciplinary actions, declined to comment on Dr. Stock’s public statements at the August school board meeting. When asked about Dr. Stock, the Indiana State Medical Association, without mentioning his name, said: “We urge Hoosier physicians to share the proven facts [about public health measures recommended by the CDC and the Indiana Department of Health] with their patients and their communities.” Dr. Stock did not respond to a request for comment.
Experts say state medical boards are ill equipped and are often unwilling to address the challenge of disciplining physicians who disseminate dangerously false medical information. That enforcement gap is particularly troubling in the middle of a deadly pandemic such as this one.
“Unless you can show a harm to an individual patient, it’s pretty tough to get the boards to do much,” said Art Caplan, PhD, a professor of bioethics at New York University. “I wish they would, but they just don’t.”
That’s partly because state laws require the boards to engage in lengthy, confidential investigations and adversarial legal processes before imposing disciplinary actions. The laws generally require patients or members of the public to file a complaint before an investigation can start. Some states, however, do allow their medical boards to take rapid emergency action if a physician poses an immediate threat to patients or the public.
Another hurdle is that medical boards that seek to sanction physicians for making dangerously misleading public statements could face lawsuits alleging that such actions violate the physicians’ constitutional free speech rights or their professional autonomy.
“We have free speech, and you can get away with a lot of stuff,” said Stephen Barrett, MD, who for many years has critically documented examples of medical fraud on his website, Quackwatch. “Some doctors would sue if they were challenged by medical boards, and I’m not sure the boards would win that court fight. People have written books with advice that killed people, and I’m not aware of a single case where the author was disciplined.”
In addition, it’s not clear that U.S. physicians who are not government officials have any legal obligation – as opposed to a moral obligation – to the government or the public to promote public health, said Jonathan Moreno, PhD, a professor of medical ethics at University of Pennsylvania, Philadelphia. “Is transmitting misinformation about COVID-19 public health malpractice?” he asked. “Do we as a society see physicians having a special role as guides in an emergency? I’d like to think we do, but we don’t have a strong tradition like that in the U.S.”
But California State Sen. Richard Pan, MD, a pediatrician who represents the Sacramento area, doesn’t buy the arguments about why medical boards can’t discipline physicians for spreading misinformation. He successfully sponsored a 2019 bill that strengthens the medical board’s ability to discipline physicians who dole out medically unjustified vaccine exemptions to children.
“A medical license is a privilege. It’s an imprimatur from the state that the person is someone who upholds professional standards,” Dr. Pan said. “If someone is intentionally spreading disinformation for personal gain and that’s putting the public at risk, the medical board has a duty to act.”
There have been only a few publicly announced disciplinary actions related to COVID-19 misinformation so far.
Last December, the Oregon Medical Board, on an emergency basis, suspended the license of Steven LaTulippe, MD, of Dallas, Ore. He had publicly announced that he and his staff were not wearing masks in his clinic. In addition, he compared COVID-19 to the common cold and denied the governor’s legal authority to adopt public health protection measures. A recorded message on his office phone said he’s challenging the licensure action in court.
Last January, the Medical Board of California made Thomas Cowan, MD, of San Francisco surrender his license after Dr. Cowan posted a YouTube video, which went viral last year, that claimed that 5G Internet networks cause COVID-19. He did not respond to a request for comment.
In May, the College of Physicians and Surgeons of British Columbia reprimanded Stephen Malthouse, MD, and forbade him from speaking on issues related to COVID-19. He had written a widely circulated open letter to the province’s chief health office claiming that the pandemic was “over” and that measures to control the spread of COVID-19 were worse than the virus. He has challenged the disciplinary action in court, alleging it violates his right to free speech.
Attacking the problem from a different angle, the U.S. Federal Trade Commission has issued enforcement actions in cases in which physicians and other health care professionals engaged in deceptive business practices related to COVID-19. That approach may be applicable to a number of physicians accused of spreading COVID-19 misinformation, who allegedly have done so at least partly to sell unproven products and services to prevent or treat the disease.
In June, the FTC settled a case against Stephen Meis, MD, of Porterville, Calif. The settlement required that he stop making unsupported claims that his company’s dietary supplements effectively treat COVID-19 symptoms and that he pay $103,420 in refunds to defrauded customers.
State medical boards in the United States generally are not allowed to disclose investigations or disciplinary processes until they finalize a disciplinary action, so other investigations that have not been publicly disclosed may be pending.
A spokesperson for the Medical Board of California said the board is aware of questionable statements about COVID-19 made by several physicians and “will be looking into it.” That comment was in response to a question about statements made at a news conference last year by two Bakersfield emergency physicians, Artin Massihi, MD, and Dan Erickson, DO. They claimed that their COVID-19 testing data showed that the virus is not that dangerous. Dr. Erickson is an osteopath and is regulated by the Osteopathic Medical Board of California.
The two physicians’ news conference prompted an unusual joint statement from the American College of Emergency Medicine and the American Academy of Emergency Medicine in April 2020 declaring that they “emphatically condemn” Dr. Massihi’s and Dr. Erickson’s “reckless and untested musings.” The groups added that it appeared that the physicians issued the comments “to advance their personal financial interests without regard for the public’s health.”
Neither Dr. Massihi nor Dr. Erickson responded to a request for comment.
As for the physician dubbed by the Center for Countering Digital Hate as the world’s most influential spreader of COVID-19 misinformation on social media: No recent public complaints have been filed, and no disciplinary action has been taken against Dr. Mercola, according to a spokesman for the Illinois Department of Financial and Professional Regulation.
According to court records, Dr. Mercola faced disciplinary complaints from the Illinois board in the early 2000s for allegedly providing false and potentially harmful medical advice on his website. There is no record of any final disciplinary action taken against him.
In widely disseminated online posts, Dr. Mercola has called the COVID-19 pandemic a “scam” and said “forced vaccination” is part of a plan to re-set the global economic system. He called COVID-19 vaccines “a medical fraud,” claiming they “alter your genetic coding.” In February, the U.S. Food and Drug Administration ordered Dr. Mercola to stop saying on his website that various vitamins and dietary supplements he sells through his website are effective in preventing or treating COVID-19.
The New York Times reported in July that Dr. Mercola’s English-language Facebook page has more than 1.7 million followers, that his Spanish-language page has one million, and that he has 300,000 followers on Twitter and 400,000 on YouTube.
In August, Dr. Mercola announced that he was deleting the large archive of articles he’s written on his website but would continue to post articles every day that would be available on the site for only 48 hours. He explained his decision by saying he’s facing “blatant censorship” as part of a “McCarthyism-like attack” from “the sitting President of the United States.” He encouraged people to read his book, “The Truth about COVID-19.”
The lack of action against Dr. Mercola for his lengthy list of scientifically unfounded statements and marketing claims about COVID-19 and other medical conditions infuriates Quackwatch’s Dr. Barrett. He’s amazed that the Illinois board did not discipline Dr. Mercola despite a number of enforcement actions against him by the FTC and the FDA.
“If a doctor were to say to a patient, ‘Don’t wear a mask and don’t get vaccinated,’ the doctor would be held responsible for a bad outcome,” he said. “But if you say it to millions and as a direct result a dozen people die, shouldn’t the doctor also be held responsible for that misinformation? I think he should lose his license.”
Another of the four physicians cited in the “Disinformation Dozen” report is Sherri Tenpenny, DO, an osteopath licensed in Ohio, who has published posts on social media advocating against masking, testing, and vaccines to prevent COVID-19 infections. A spokesperson for the State Medical Board of Ohio said Dr. Tenpenny’s license expires on Oct. 1, 2021, and that any investigation would be confidential. She added that grounds for disciplinary action include “making a false, fraudulent, deceptive, or misleading statement in relation to the practice of medicine and surgery.” Dr. Tenpenny could not be reached for comment.
A third physician named in the report is Christiane Northrup, MD, an ob.gyn. formerly licensed in Maine, who has published posts advocating unproven cures for COVID-19 and claiming that vaccines increase chronic illness. Dennis Smith, executive director of the Maine Board of Licensure in Medicine, said the board received complaints about Dr. Northrup’s posts but can’t act because she withdrew her Maine license in 2015. He added that the Maine board can issue sanctions against physicians who engage in fraud, deceit, or misrepresentation or who post scientifically unfounded statements online.
The fourth physician identified in the “Disinformation Dozen” report is Rashid Buttar, DO, an osteopath practicing in Mooresville, N.C., who has claimed in social media posts that COVID-19 vaccines cause infertility and that COVID-19 tests contain living microorganisms. A spokeswoman for the North Carolina Medical Board said she could not confirm or deny the existence of any investigation of Dr. Buttar, who signed a consent order with the medical board in 2010 following charges of exorbitant fees, worthless tests and treatment, and false diagnoses. Undisclosed conditions were placed on his medical license in 2013. The spokesperson added that the board would investigate any information alleging that a physician spread false information about COVID-19.
Another physician who has caused widespread consternation over scientifically unfounded statements about COVID-19 is Simone Gold, MD, formerly an emergency department physician in Los Angeles. She founded a group called America’s Frontline Doctors, which filed a federal lawsuit in Alabama this spring to block the FDA from issuing an emergency use authorization allowing teenagers to receive COVID-19 vaccinations. She called the vaccines “an experimental biological agent whose harms are well-documented.”
Last summer, Dr. Gold and other physicians in her group held a news conference on the steps of the U.S. Supreme Court Building promoting hydroxychloroquine as a COVID-19 treatment. They declared that masks don’t work and that the virus isn’t deadly, and made other false claims. The news conference was livestreamed by conservative media outlets, was promoted on Twitter by then-President Trump and his family, and was viewed online more than 14 million times.
One of the participating physicians, Stella Immanuel, MD, of Houston, claimed in a video that went viral that she had successfully used hydroxychloroquine for more than 400 patients to cure the disease. In response, the Texas Medical Board, without naming Dr. Immanuel, warned that if it received a complaint about any physician who made a false claim about having a cure for COVID-19, it would investigate and potentially take disciplinary action.
Although no publicly known disciplinary action has been taken against Dr. Gold, she told The Washington Post last January that after participating in that July 2020 news conference, she was fired from her emergency department job at two hospitals and that she hasn’t worked as a physician since. Dr. Gold did not respond to a request for comment.
The outcome in her situation is consistent with the view of NYU’s Dr. Caplan that methods other than medical board discipline – such as action by employers, social media pressure, and reprimands from professional societies –will have to be used to hold physicians accountable for spreading COVID-19 misinformation.
“I’m disappointed to have to say it, but I don’t think medical boards are going to be effective,” he said. “We don’t know how to manage misinformation despite being in a plague. We just don’t.
A version of this article first appeared on Medscape.com.
A tall, distinguished-looking physician in shirtsleeves and suspenders walked to the microphone at the Mt. Vernon, Ind., school board meeting on a Friday evening in early August. He launched into an impassioned, 7-minute attack on the public health establishment’s medical guidelines for COVID-19.
“The Center for Disease Control and the Indiana State [Department] of Health are giving you very bad scientific guidance,” said Daniel Stock, MD, a primary care physician with a concierge practice in Noblesville, Ind., He described himself as a “functional family medicine physician,” though he is not board certified in family medicine.
Dr. Stock told the school board members that COVID-19 vaccines are counterproductive because they make coronavirus infections worse. He claimed his treatment of “over 15” COVID-19 patients with vitamin D, ivermectin, and zinc has kept them out of the hospital, and that those treatments reduce mortality risk from the disease by 75%. (A study released in mid-August found that ivermectin is ineffective in treating COVID-19).
In response to Dr. Stock’s remarks, the state health department quickly issued a statement reaffirming that COVID-19 vaccines “are highly effective at preventing hospitalizations and deaths.” But by then, the YouTube video of Dr. Stock’s comments had garnered nearly 600,000 views as of Aug. 12 and had been shared over 10,000 times on Facebook. Opponents of COVID-19 vaccines and masking policies across the country have been citing his comments.
Across the country, state medical licensing boards and state and national medical associations are struggling with how to respond to scientifically baseless public statements about COVID-19 by some physicians such as Dr. Stock. They fear such statements are increasing public confusion and are heightening political conflict. Physicians accused of spreading false information include public officials such as Scott Atlas, MD, who served as President Donald Trump’s COVID-19 advisor, and Kentucky Sen. Rand Paul, an ophthalmologist, whose YouTube account was temporarily suspended in August after he posted a video disputing the effectiveness of masking in stopping the spread of COVID-19.
“That’s the problem – those types of viral videos of someone somewhere who thinks they know something the rest of us don’t,” lamented Jennifer Bryan, MD, board chair of the Mississippi State Medical Association. “I don’t know any good reason why a physician should be advising against vaccination. It’s appropriate for medical boards to look into those situations.”
The Federation of State Medical Boards agrees. In July, it warned that physicians who willfully spread false information about COVID-19 risk suspension or revocation of their medical license. The federation cited a “dramatic increase in the dissemination of COVID-19 vaccine misinformation and disinformation by physicians.” That’s particularly dangerous, it said, because physicians enjoy a high degree of public credibility.
Medical boards will particularly examine cases in which there is a pattern of misinformation or disinformation showing that a physician poses a continuing threat to public health, said Hank Chaudhry, DO, the federation’s CEO. In some cases, he said, boards have contacted physicians and have persuaded them to voluntarily refrain from making false public statements, without taking disciplinary action.
“Words matter,” he said. “Physicians have a really big platform, whether they realize it or not. Misinformation or disinformation in the context of COVID can not only cause harm but also death. We felt it was appropriate to remind physicians to be careful.”
Although medical leaders stress that most physicians are promoting solid science on COVID-19, the London-based Center for Countering Digital Hate, in a May report titled “The Disinformation Dozen,” named four U.S. physicians among 12 people who it said produce 65% of the misleading claims and lies about COVID-19 vaccines that abound on Facebook, Instagram, and Twitter. The leading spreader of false claims, the group said, is Joseph Mercola, MD, an Illinois-licensed osteopath living in Cape Coral, Fla. He did not respond to requests for comment.
But so far, state licensing boards and federal and state medical associations generally have been reluctant to discipline or publicly call out physicians who have spread misinformation about the causes, treatments, vaccines, and prevention strategies for COVID-19. Some of these physicians, such as Dr. Mercola, have a long history predating the COVID-19 pandemic of disseminating scientifically baseless information, often in connection with their marketing of products and services.
For instance, the Medical Licensing Board of Indiana and the state attorney general’s office, which brings medical disciplinary actions, declined to comment on Dr. Stock’s public statements at the August school board meeting. When asked about Dr. Stock, the Indiana State Medical Association, without mentioning his name, said: “We urge Hoosier physicians to share the proven facts [about public health measures recommended by the CDC and the Indiana Department of Health] with their patients and their communities.” Dr. Stock did not respond to a request for comment.
Experts say state medical boards are ill equipped and are often unwilling to address the challenge of disciplining physicians who disseminate dangerously false medical information. That enforcement gap is particularly troubling in the middle of a deadly pandemic such as this one.
“Unless you can show a harm to an individual patient, it’s pretty tough to get the boards to do much,” said Art Caplan, PhD, a professor of bioethics at New York University. “I wish they would, but they just don’t.”
That’s partly because state laws require the boards to engage in lengthy, confidential investigations and adversarial legal processes before imposing disciplinary actions. The laws generally require patients or members of the public to file a complaint before an investigation can start. Some states, however, do allow their medical boards to take rapid emergency action if a physician poses an immediate threat to patients or the public.
Another hurdle is that medical boards that seek to sanction physicians for making dangerously misleading public statements could face lawsuits alleging that such actions violate the physicians’ constitutional free speech rights or their professional autonomy.
“We have free speech, and you can get away with a lot of stuff,” said Stephen Barrett, MD, who for many years has critically documented examples of medical fraud on his website, Quackwatch. “Some doctors would sue if they were challenged by medical boards, and I’m not sure the boards would win that court fight. People have written books with advice that killed people, and I’m not aware of a single case where the author was disciplined.”
In addition, it’s not clear that U.S. physicians who are not government officials have any legal obligation – as opposed to a moral obligation – to the government or the public to promote public health, said Jonathan Moreno, PhD, a professor of medical ethics at University of Pennsylvania, Philadelphia. “Is transmitting misinformation about COVID-19 public health malpractice?” he asked. “Do we as a society see physicians having a special role as guides in an emergency? I’d like to think we do, but we don’t have a strong tradition like that in the U.S.”
But California State Sen. Richard Pan, MD, a pediatrician who represents the Sacramento area, doesn’t buy the arguments about why medical boards can’t discipline physicians for spreading misinformation. He successfully sponsored a 2019 bill that strengthens the medical board’s ability to discipline physicians who dole out medically unjustified vaccine exemptions to children.
“A medical license is a privilege. It’s an imprimatur from the state that the person is someone who upholds professional standards,” Dr. Pan said. “If someone is intentionally spreading disinformation for personal gain and that’s putting the public at risk, the medical board has a duty to act.”
There have been only a few publicly announced disciplinary actions related to COVID-19 misinformation so far.
Last December, the Oregon Medical Board, on an emergency basis, suspended the license of Steven LaTulippe, MD, of Dallas, Ore. He had publicly announced that he and his staff were not wearing masks in his clinic. In addition, he compared COVID-19 to the common cold and denied the governor’s legal authority to adopt public health protection measures. A recorded message on his office phone said he’s challenging the licensure action in court.
Last January, the Medical Board of California made Thomas Cowan, MD, of San Francisco surrender his license after Dr. Cowan posted a YouTube video, which went viral last year, that claimed that 5G Internet networks cause COVID-19. He did not respond to a request for comment.
In May, the College of Physicians and Surgeons of British Columbia reprimanded Stephen Malthouse, MD, and forbade him from speaking on issues related to COVID-19. He had written a widely circulated open letter to the province’s chief health office claiming that the pandemic was “over” and that measures to control the spread of COVID-19 were worse than the virus. He has challenged the disciplinary action in court, alleging it violates his right to free speech.
Attacking the problem from a different angle, the U.S. Federal Trade Commission has issued enforcement actions in cases in which physicians and other health care professionals engaged in deceptive business practices related to COVID-19. That approach may be applicable to a number of physicians accused of spreading COVID-19 misinformation, who allegedly have done so at least partly to sell unproven products and services to prevent or treat the disease.
In June, the FTC settled a case against Stephen Meis, MD, of Porterville, Calif. The settlement required that he stop making unsupported claims that his company’s dietary supplements effectively treat COVID-19 symptoms and that he pay $103,420 in refunds to defrauded customers.
State medical boards in the United States generally are not allowed to disclose investigations or disciplinary processes until they finalize a disciplinary action, so other investigations that have not been publicly disclosed may be pending.
A spokesperson for the Medical Board of California said the board is aware of questionable statements about COVID-19 made by several physicians and “will be looking into it.” That comment was in response to a question about statements made at a news conference last year by two Bakersfield emergency physicians, Artin Massihi, MD, and Dan Erickson, DO. They claimed that their COVID-19 testing data showed that the virus is not that dangerous. Dr. Erickson is an osteopath and is regulated by the Osteopathic Medical Board of California.
The two physicians’ news conference prompted an unusual joint statement from the American College of Emergency Medicine and the American Academy of Emergency Medicine in April 2020 declaring that they “emphatically condemn” Dr. Massihi’s and Dr. Erickson’s “reckless and untested musings.” The groups added that it appeared that the physicians issued the comments “to advance their personal financial interests without regard for the public’s health.”
Neither Dr. Massihi nor Dr. Erickson responded to a request for comment.
As for the physician dubbed by the Center for Countering Digital Hate as the world’s most influential spreader of COVID-19 misinformation on social media: No recent public complaints have been filed, and no disciplinary action has been taken against Dr. Mercola, according to a spokesman for the Illinois Department of Financial and Professional Regulation.
According to court records, Dr. Mercola faced disciplinary complaints from the Illinois board in the early 2000s for allegedly providing false and potentially harmful medical advice on his website. There is no record of any final disciplinary action taken against him.
In widely disseminated online posts, Dr. Mercola has called the COVID-19 pandemic a “scam” and said “forced vaccination” is part of a plan to re-set the global economic system. He called COVID-19 vaccines “a medical fraud,” claiming they “alter your genetic coding.” In February, the U.S. Food and Drug Administration ordered Dr. Mercola to stop saying on his website that various vitamins and dietary supplements he sells through his website are effective in preventing or treating COVID-19.
The New York Times reported in July that Dr. Mercola’s English-language Facebook page has more than 1.7 million followers, that his Spanish-language page has one million, and that he has 300,000 followers on Twitter and 400,000 on YouTube.
In August, Dr. Mercola announced that he was deleting the large archive of articles he’s written on his website but would continue to post articles every day that would be available on the site for only 48 hours. He explained his decision by saying he’s facing “blatant censorship” as part of a “McCarthyism-like attack” from “the sitting President of the United States.” He encouraged people to read his book, “The Truth about COVID-19.”
The lack of action against Dr. Mercola for his lengthy list of scientifically unfounded statements and marketing claims about COVID-19 and other medical conditions infuriates Quackwatch’s Dr. Barrett. He’s amazed that the Illinois board did not discipline Dr. Mercola despite a number of enforcement actions against him by the FTC and the FDA.
“If a doctor were to say to a patient, ‘Don’t wear a mask and don’t get vaccinated,’ the doctor would be held responsible for a bad outcome,” he said. “But if you say it to millions and as a direct result a dozen people die, shouldn’t the doctor also be held responsible for that misinformation? I think he should lose his license.”
Another of the four physicians cited in the “Disinformation Dozen” report is Sherri Tenpenny, DO, an osteopath licensed in Ohio, who has published posts on social media advocating against masking, testing, and vaccines to prevent COVID-19 infections. A spokesperson for the State Medical Board of Ohio said Dr. Tenpenny’s license expires on Oct. 1, 2021, and that any investigation would be confidential. She added that grounds for disciplinary action include “making a false, fraudulent, deceptive, or misleading statement in relation to the practice of medicine and surgery.” Dr. Tenpenny could not be reached for comment.
A third physician named in the report is Christiane Northrup, MD, an ob.gyn. formerly licensed in Maine, who has published posts advocating unproven cures for COVID-19 and claiming that vaccines increase chronic illness. Dennis Smith, executive director of the Maine Board of Licensure in Medicine, said the board received complaints about Dr. Northrup’s posts but can’t act because she withdrew her Maine license in 2015. He added that the Maine board can issue sanctions against physicians who engage in fraud, deceit, or misrepresentation or who post scientifically unfounded statements online.
The fourth physician identified in the “Disinformation Dozen” report is Rashid Buttar, DO, an osteopath practicing in Mooresville, N.C., who has claimed in social media posts that COVID-19 vaccines cause infertility and that COVID-19 tests contain living microorganisms. A spokeswoman for the North Carolina Medical Board said she could not confirm or deny the existence of any investigation of Dr. Buttar, who signed a consent order with the medical board in 2010 following charges of exorbitant fees, worthless tests and treatment, and false diagnoses. Undisclosed conditions were placed on his medical license in 2013. The spokesperson added that the board would investigate any information alleging that a physician spread false information about COVID-19.
Another physician who has caused widespread consternation over scientifically unfounded statements about COVID-19 is Simone Gold, MD, formerly an emergency department physician in Los Angeles. She founded a group called America’s Frontline Doctors, which filed a federal lawsuit in Alabama this spring to block the FDA from issuing an emergency use authorization allowing teenagers to receive COVID-19 vaccinations. She called the vaccines “an experimental biological agent whose harms are well-documented.”
Last summer, Dr. Gold and other physicians in her group held a news conference on the steps of the U.S. Supreme Court Building promoting hydroxychloroquine as a COVID-19 treatment. They declared that masks don’t work and that the virus isn’t deadly, and made other false claims. The news conference was livestreamed by conservative media outlets, was promoted on Twitter by then-President Trump and his family, and was viewed online more than 14 million times.
One of the participating physicians, Stella Immanuel, MD, of Houston, claimed in a video that went viral that she had successfully used hydroxychloroquine for more than 400 patients to cure the disease. In response, the Texas Medical Board, without naming Dr. Immanuel, warned that if it received a complaint about any physician who made a false claim about having a cure for COVID-19, it would investigate and potentially take disciplinary action.
Although no publicly known disciplinary action has been taken against Dr. Gold, she told The Washington Post last January that after participating in that July 2020 news conference, she was fired from her emergency department job at two hospitals and that she hasn’t worked as a physician since. Dr. Gold did not respond to a request for comment.
The outcome in her situation is consistent with the view of NYU’s Dr. Caplan that methods other than medical board discipline – such as action by employers, social media pressure, and reprimands from professional societies –will have to be used to hold physicians accountable for spreading COVID-19 misinformation.
“I’m disappointed to have to say it, but I don’t think medical boards are going to be effective,” he said. “We don’t know how to manage misinformation despite being in a plague. We just don’t.
A version of this article first appeared on Medscape.com.








