User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Real-world data generate debate on definition of flare in axial spondyloarthritis
How best to define axial spondyloarthritis (axSpA) flares in practice remains the subject of some debate as evidenced by the discussion that followed an abstract presentation at the 12th International Congress on Spondyloarthritides.
It’s an important topic, said Maxime Breban, MD, PhD, of Ambroise Paré Hospital in Paris, as flares can adversely affect patient outcomes. The absence of flares may also a useful measure of how well a patient is responding to treatment in clinical trials and whether a treatment can be tapered.
“There have been many ways to define flares in the past and there is no consensus,” he observed.
Although the Assessment of Spondyloarthritis International Society (ASAS) devised 12 preliminary definitions of flare in 2016, “these were not that good when we moved to patients,” Dr. Breban suggested.
The ASAS definitions were based on patient vignettes, he explained, and used a combination of variables from the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), a visual analog scale (VAS) of pain, and the Ankylosing Spondylitis Disease Activity Score based on C-reactive protein (ASDAS-CRP).
The study that Dr. Breban presented looked at the performance of the ASAS preliminary definitions of axSpA flares in a real-life patient population, as well as prospectively determining how variations in BASDAI and VAS pain were associated with patient-perceived flares of disease.
A total of 99 patients took part in the study, recruited through a secure e-health platform called SPONDY+. Once a week, patients completed the BASDAI questionnaire and the pain VAS, and stated whether their disease had flared in the past week.
Receiver operating characteristic (ROC) curves were calculated to see how well the BASDAI and pain VAS identified patients who were experiencing a flare or had a recently resolved flare of axSpA.
Dr. Breban reported that variation in the BASDAI “appears a suitable variable to monitor the occurrence and resolution of patient-reported flare in axial spondylarthritis.”
In predicting a flare, the area under the curve (AUC) was significantly higher for the change in BASDAI than for the change in pain VAS, at a respective 0.81 and 0.77 (P = .01). However, both variables were similarly accurate in predicting the resolution of a flare, with respective AUCs of 0.78 and 0.80 (P = .3).
A 0.22-point increase in BASDAI was reported to be the best balance between sensitivity (70%) and specificity (79%) for a flare. However, this is “outside of what is possible within a test–retest situation,” Désirée van der Heijde, MD, PhD, of Leiden (the Netherlands) University Medical Center, said during discussion.
Dr. van der Heijde told Dr. Breban: “I understand that that comes out of your data, that that’s the best combination for sensitivity and specificity, but the next step is to decide if that makes sense.”
The ROC curves that Dr. Breban presented showed the range of sensitivities and specificities that could be achieved. If the specificity was increased to be 90% or higher, the specificity fell to 55%, with the change in BASDAI being an increase of 0.8 points. Conversely, bringing the sensitivity above 90% meant the specificity dropped to 39% and the change in BASDAI was a decrease of 0.1 point.
“So that means you can choose whatever you want as a cutoff,” Dr. Breban said. It depends on what you are aiming to do. “If you want to identify a flare, you can increase sensitivity, or specificity, according to what your purpose is,” he suggested.
“The next step, of course, is what to choose as a flare. Then it depends on how you want to use a flare if you want to use a flare to change the treatment,” agreed Dr. van der Heijde. “That was why, in the ASAS group, it was decided to have a high specificity so that you are not changing treatment all the time.”
In the data that Dr. Breban presented, the ASAS preliminary definitions were highly specific but lacked sensitivity. None of the ASAS definitions yielded sensitivity values higher than 37%, whereas specificity was higher than 95% for all of them.
The study’s design did not allow researchers to test the ASDAS-CRP as a definition of flare in its real-world patient sample. Thus, it is looking only at the patient’s perspective on flare, and there is a “huge discrepancy” between patient and physician-reported disease activity, Dr. van der Heijde noted. “So, I think before using your data to really choose the flare definition, I think we need to take it all into account.”
Maxime Dougados, MD, PhD, of Cochin Hospital in Paris, who has been “deeply involved in the elaboration of the definition of flare” added his thoughts: “Flare means for me, not a status, but a change,” he observed.
But if the aim of treating people with axSpA is to achieve a good or acceptable state of health, he questioned whether work should be continued to define the concept of a flare.
The definition of a flare was conceived for use in clinical trials mainly, Dr. van der Heijde noted. It helped to assess how changes in treatment might affect the outcomes of patients. In clinical practice, especially now with treat-to-target gaining more and more traction in axSpA, she agreed that perhaps the goal should be to focus more on the health status of patients.
Dr. Breban acknowledged that the SPONDY+ platform has been developed by bepatient with support from Merck Sharp & Dohme. No other disclosures were made.
How best to define axial spondyloarthritis (axSpA) flares in practice remains the subject of some debate as evidenced by the discussion that followed an abstract presentation at the 12th International Congress on Spondyloarthritides.
It’s an important topic, said Maxime Breban, MD, PhD, of Ambroise Paré Hospital in Paris, as flares can adversely affect patient outcomes. The absence of flares may also a useful measure of how well a patient is responding to treatment in clinical trials and whether a treatment can be tapered.
“There have been many ways to define flares in the past and there is no consensus,” he observed.
Although the Assessment of Spondyloarthritis International Society (ASAS) devised 12 preliminary definitions of flare in 2016, “these were not that good when we moved to patients,” Dr. Breban suggested.
The ASAS definitions were based on patient vignettes, he explained, and used a combination of variables from the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), a visual analog scale (VAS) of pain, and the Ankylosing Spondylitis Disease Activity Score based on C-reactive protein (ASDAS-CRP).
The study that Dr. Breban presented looked at the performance of the ASAS preliminary definitions of axSpA flares in a real-life patient population, as well as prospectively determining how variations in BASDAI and VAS pain were associated with patient-perceived flares of disease.
A total of 99 patients took part in the study, recruited through a secure e-health platform called SPONDY+. Once a week, patients completed the BASDAI questionnaire and the pain VAS, and stated whether their disease had flared in the past week.
Receiver operating characteristic (ROC) curves were calculated to see how well the BASDAI and pain VAS identified patients who were experiencing a flare or had a recently resolved flare of axSpA.
Dr. Breban reported that variation in the BASDAI “appears a suitable variable to monitor the occurrence and resolution of patient-reported flare in axial spondylarthritis.”
In predicting a flare, the area under the curve (AUC) was significantly higher for the change in BASDAI than for the change in pain VAS, at a respective 0.81 and 0.77 (P = .01). However, both variables were similarly accurate in predicting the resolution of a flare, with respective AUCs of 0.78 and 0.80 (P = .3).
A 0.22-point increase in BASDAI was reported to be the best balance between sensitivity (70%) and specificity (79%) for a flare. However, this is “outside of what is possible within a test–retest situation,” Désirée van der Heijde, MD, PhD, of Leiden (the Netherlands) University Medical Center, said during discussion.
Dr. van der Heijde told Dr. Breban: “I understand that that comes out of your data, that that’s the best combination for sensitivity and specificity, but the next step is to decide if that makes sense.”
The ROC curves that Dr. Breban presented showed the range of sensitivities and specificities that could be achieved. If the specificity was increased to be 90% or higher, the specificity fell to 55%, with the change in BASDAI being an increase of 0.8 points. Conversely, bringing the sensitivity above 90% meant the specificity dropped to 39% and the change in BASDAI was a decrease of 0.1 point.
“So that means you can choose whatever you want as a cutoff,” Dr. Breban said. It depends on what you are aiming to do. “If you want to identify a flare, you can increase sensitivity, or specificity, according to what your purpose is,” he suggested.
“The next step, of course, is what to choose as a flare. Then it depends on how you want to use a flare if you want to use a flare to change the treatment,” agreed Dr. van der Heijde. “That was why, in the ASAS group, it was decided to have a high specificity so that you are not changing treatment all the time.”
In the data that Dr. Breban presented, the ASAS preliminary definitions were highly specific but lacked sensitivity. None of the ASAS definitions yielded sensitivity values higher than 37%, whereas specificity was higher than 95% for all of them.
The study’s design did not allow researchers to test the ASDAS-CRP as a definition of flare in its real-world patient sample. Thus, it is looking only at the patient’s perspective on flare, and there is a “huge discrepancy” between patient and physician-reported disease activity, Dr. van der Heijde noted. “So, I think before using your data to really choose the flare definition, I think we need to take it all into account.”
Maxime Dougados, MD, PhD, of Cochin Hospital in Paris, who has been “deeply involved in the elaboration of the definition of flare” added his thoughts: “Flare means for me, not a status, but a change,” he observed.
But if the aim of treating people with axSpA is to achieve a good or acceptable state of health, he questioned whether work should be continued to define the concept of a flare.
The definition of a flare was conceived for use in clinical trials mainly, Dr. van der Heijde noted. It helped to assess how changes in treatment might affect the outcomes of patients. In clinical practice, especially now with treat-to-target gaining more and more traction in axSpA, she agreed that perhaps the goal should be to focus more on the health status of patients.
Dr. Breban acknowledged that the SPONDY+ platform has been developed by bepatient with support from Merck Sharp & Dohme. No other disclosures were made.
How best to define axial spondyloarthritis (axSpA) flares in practice remains the subject of some debate as evidenced by the discussion that followed an abstract presentation at the 12th International Congress on Spondyloarthritides.
It’s an important topic, said Maxime Breban, MD, PhD, of Ambroise Paré Hospital in Paris, as flares can adversely affect patient outcomes. The absence of flares may also a useful measure of how well a patient is responding to treatment in clinical trials and whether a treatment can be tapered.
“There have been many ways to define flares in the past and there is no consensus,” he observed.
Although the Assessment of Spondyloarthritis International Society (ASAS) devised 12 preliminary definitions of flare in 2016, “these were not that good when we moved to patients,” Dr. Breban suggested.
The ASAS definitions were based on patient vignettes, he explained, and used a combination of variables from the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), a visual analog scale (VAS) of pain, and the Ankylosing Spondylitis Disease Activity Score based on C-reactive protein (ASDAS-CRP).
The study that Dr. Breban presented looked at the performance of the ASAS preliminary definitions of axSpA flares in a real-life patient population, as well as prospectively determining how variations in BASDAI and VAS pain were associated with patient-perceived flares of disease.
A total of 99 patients took part in the study, recruited through a secure e-health platform called SPONDY+. Once a week, patients completed the BASDAI questionnaire and the pain VAS, and stated whether their disease had flared in the past week.
Receiver operating characteristic (ROC) curves were calculated to see how well the BASDAI and pain VAS identified patients who were experiencing a flare or had a recently resolved flare of axSpA.
Dr. Breban reported that variation in the BASDAI “appears a suitable variable to monitor the occurrence and resolution of patient-reported flare in axial spondylarthritis.”
In predicting a flare, the area under the curve (AUC) was significantly higher for the change in BASDAI than for the change in pain VAS, at a respective 0.81 and 0.77 (P = .01). However, both variables were similarly accurate in predicting the resolution of a flare, with respective AUCs of 0.78 and 0.80 (P = .3).
A 0.22-point increase in BASDAI was reported to be the best balance between sensitivity (70%) and specificity (79%) for a flare. However, this is “outside of what is possible within a test–retest situation,” Désirée van der Heijde, MD, PhD, of Leiden (the Netherlands) University Medical Center, said during discussion.
Dr. van der Heijde told Dr. Breban: “I understand that that comes out of your data, that that’s the best combination for sensitivity and specificity, but the next step is to decide if that makes sense.”
The ROC curves that Dr. Breban presented showed the range of sensitivities and specificities that could be achieved. If the specificity was increased to be 90% or higher, the specificity fell to 55%, with the change in BASDAI being an increase of 0.8 points. Conversely, bringing the sensitivity above 90% meant the specificity dropped to 39% and the change in BASDAI was a decrease of 0.1 point.
“So that means you can choose whatever you want as a cutoff,” Dr. Breban said. It depends on what you are aiming to do. “If you want to identify a flare, you can increase sensitivity, or specificity, according to what your purpose is,” he suggested.
“The next step, of course, is what to choose as a flare. Then it depends on how you want to use a flare if you want to use a flare to change the treatment,” agreed Dr. van der Heijde. “That was why, in the ASAS group, it was decided to have a high specificity so that you are not changing treatment all the time.”
In the data that Dr. Breban presented, the ASAS preliminary definitions were highly specific but lacked sensitivity. None of the ASAS definitions yielded sensitivity values higher than 37%, whereas specificity was higher than 95% for all of them.
The study’s design did not allow researchers to test the ASDAS-CRP as a definition of flare in its real-world patient sample. Thus, it is looking only at the patient’s perspective on flare, and there is a “huge discrepancy” between patient and physician-reported disease activity, Dr. van der Heijde noted. “So, I think before using your data to really choose the flare definition, I think we need to take it all into account.”
Maxime Dougados, MD, PhD, of Cochin Hospital in Paris, who has been “deeply involved in the elaboration of the definition of flare” added his thoughts: “Flare means for me, not a status, but a change,” he observed.
But if the aim of treating people with axSpA is to achieve a good or acceptable state of health, he questioned whether work should be continued to define the concept of a flare.
The definition of a flare was conceived for use in clinical trials mainly, Dr. van der Heijde noted. It helped to assess how changes in treatment might affect the outcomes of patients. In clinical practice, especially now with treat-to-target gaining more and more traction in axSpA, she agreed that perhaps the goal should be to focus more on the health status of patients.
Dr. Breban acknowledged that the SPONDY+ platform has been developed by bepatient with support from Merck Sharp & Dohme. No other disclosures were made.
FROM THE 2021 SPA CONGRESS
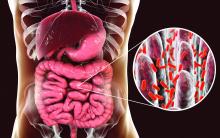
RA treatment responders show unique differences in gut microbiome
The gut microbiome, previously shown to have an association with rheumatoid arthritis, may also provide signals of a patient’s disease prognosis, researchers at the Mayo Clinic have reported.
“We found that the gut microbiome is linked to whether patients with RA improve in their clinical symptoms or not,” cosenior author Jaeyun Sung, PhD, said in an interview. “We found features of the gut microbiome that linked to improvement, and we also put those features in a machine-learning model that can actually predict improvement at a follow-up visit.” Dr. Sung is a computational biologist with Mayo Clinic’s Center for Individualized Medicine in Rochester, Minn.
The retrospective, observational cohort study included 32 patients diagnosed with RA between 1988 and 2014. The researchers performed meta-genome shotgun sequencing on 64 stool samples kept in a biobank and collected at two separate visits 6-12 months apart. Dr. Sung and colleagues observed significantly different microbiome traits between patients who eventually showed minimally clinically important improvement and those who didn’t.
The study also provided a proof of concept for using machine-learning technology to analyze the gut microbiome to predict the course of the disease, Dr. Sung said.
Cosenior author John M. Davis III, MD, a clinical rheumatologist and rheumatology research chair of the Mayo Clinic, noted that their own previous study had confirmed dysbiosis in people with RA when compared with controls. “We had some preliminary insight that it may be linked to some extent to the disease state and maybe treatments,” Dr. Davis said. “So that led us to hypothesize that there may be an association between the gut microbiome and response to treatment or disease activity over time.”
The study found that age was the dominant factor in determining variations in the gut microbiome composition, but the next prevailing factor was minimum clinically important improvement status, which 12 of the 32 study participants achieved at their follow-up visits. At baseline, all patients were on some type of treatment – either biologic or conventional disease-modifying antirheumatic drugs (DMARDs, 46.9% and 87.5%, respectively), or prednisone (46.9%).
Gut microbiome composition
The patients who achieved minimum clinically important improvement had an average decline in Clinical Disease Activity Index of 16.7 units (standard deviation, 12.8) versus a gain of 5.7 (SD, 8.9) in the remaining patients. The study found higher species-level alpha diversity and richness and higher beta diversity in the group that achieved minimum clinically important improvement, compared with those who did not.
They identified six microbial taxa as higher in abundance in the improved patients: Negativicutes (class); Selenomonadales (order); Prevotellaceae (family); Coprococcus (genus); Bacteroides sp. 3_1_19 (species); and Bilophila sp. 4_1_30 (species). In the patients who showed no improvement, Eubacterium sp. 3_1_31 (species) was found to be higher (P < .05). They also found 15 metabolic pathways that were differently abundant between the two groups at baseline.
Two things make this study different from other studies of the gut microbiome in RA, Dr. Sung said: It didn’t have a control group, only RA patients, and it didn’t evaluate a specific drug in RA patients.
“We’re thinking beyond just drug or treatment, independent of prior treatment, independent of prior clinical measurements, independent of age, sex, and other factors, can we predict RA response just using the gut microbiome alone?” Dr. Sung said. “Is there an association between clinical improvement and the gut microbiome?”
The study also showed that the microbiome may be a modifiable target for RA, Dr. Davis said.
“This research is attractive because it may complement medical treatment for RA if we can identify dietary modifications,” he said. “Still, there’s the question if probiotics or prebiotics can influence the gut. Can we modify the gut microbiome to further ameliorate the disease state? That’s something I think is an open question that’s specifically called out in our paper.”
This study included patients with long-term disease, but the group’s ongoing research is focusing on patients with earlier-stage RA, Dr. Davis said. “The next steps have to be in validating [the findings] in additional and external populations and looking at patients with very early disease where a lot of the decision-making is very active and happening in real time.”
James T. Rosenbaum, MD, an ophthalmologist and rheumatologist at Oregon Health & Science University, Portland, acknowledged that this is the first study in RA to find an effect on the gut microbiome using the minimum clinically important improvement endpoint.
“It also raises a ‘chicken-egg’ dilemma,” Dr. Rosenbaum said in an interview. “Did the patients improve and then their microbiome changed, or was the microbiome the first change that led to the clinical improvement? If the latter is correct, we potentially could alter the microbiome, for example, by diet, to treat rheumatic disease.”
He noted that studies with fecal transplants for ulcerative colitis support the therapeutic potential of microbiome modification. “But,” he added, “we are still a long way from putting this in practice.”
“The outcome is promising,” Claudia Mauri, PhD, a professor of immunology at University College London, said of the study. “Obviously, if this can be repeated in a very large cohort of patients, it would give us the possibility to be able to, based on the composition of the microbiota, to predict who is going to respond to treatment or not.”
She noted that, while RA has a broader array of available treatments than other autoimmune diseases, some RA patients don’t respond their first biologic treatment. “If from the outset we would be able to see who may not respond based on the microbiota, we may prepare physicians to better target these patients by, for example, offering them an alternative second biologic agent,” Dr. Mauri said.
Dr. Davis reported receiving research grants from Pfizer. Dr. Sung and other study coauthors have no financial relationships to disclose. Dr. Rosenbaum reported that the National Institutes of Health supports his research. Dr. Mauri has no financial relationships to disclose.
The gut microbiome, previously shown to have an association with rheumatoid arthritis, may also provide signals of a patient’s disease prognosis, researchers at the Mayo Clinic have reported.
“We found that the gut microbiome is linked to whether patients with RA improve in their clinical symptoms or not,” cosenior author Jaeyun Sung, PhD, said in an interview. “We found features of the gut microbiome that linked to improvement, and we also put those features in a machine-learning model that can actually predict improvement at a follow-up visit.” Dr. Sung is a computational biologist with Mayo Clinic’s Center for Individualized Medicine in Rochester, Minn.
The retrospective, observational cohort study included 32 patients diagnosed with RA between 1988 and 2014. The researchers performed meta-genome shotgun sequencing on 64 stool samples kept in a biobank and collected at two separate visits 6-12 months apart. Dr. Sung and colleagues observed significantly different microbiome traits between patients who eventually showed minimally clinically important improvement and those who didn’t.
The study also provided a proof of concept for using machine-learning technology to analyze the gut microbiome to predict the course of the disease, Dr. Sung said.
Cosenior author John M. Davis III, MD, a clinical rheumatologist and rheumatology research chair of the Mayo Clinic, noted that their own previous study had confirmed dysbiosis in people with RA when compared with controls. “We had some preliminary insight that it may be linked to some extent to the disease state and maybe treatments,” Dr. Davis said. “So that led us to hypothesize that there may be an association between the gut microbiome and response to treatment or disease activity over time.”
The study found that age was the dominant factor in determining variations in the gut microbiome composition, but the next prevailing factor was minimum clinically important improvement status, which 12 of the 32 study participants achieved at their follow-up visits. At baseline, all patients were on some type of treatment – either biologic or conventional disease-modifying antirheumatic drugs (DMARDs, 46.9% and 87.5%, respectively), or prednisone (46.9%).
Gut microbiome composition
The patients who achieved minimum clinically important improvement had an average decline in Clinical Disease Activity Index of 16.7 units (standard deviation, 12.8) versus a gain of 5.7 (SD, 8.9) in the remaining patients. The study found higher species-level alpha diversity and richness and higher beta diversity in the group that achieved minimum clinically important improvement, compared with those who did not.
They identified six microbial taxa as higher in abundance in the improved patients: Negativicutes (class); Selenomonadales (order); Prevotellaceae (family); Coprococcus (genus); Bacteroides sp. 3_1_19 (species); and Bilophila sp. 4_1_30 (species). In the patients who showed no improvement, Eubacterium sp. 3_1_31 (species) was found to be higher (P < .05). They also found 15 metabolic pathways that were differently abundant between the two groups at baseline.
Two things make this study different from other studies of the gut microbiome in RA, Dr. Sung said: It didn’t have a control group, only RA patients, and it didn’t evaluate a specific drug in RA patients.
“We’re thinking beyond just drug or treatment, independent of prior treatment, independent of prior clinical measurements, independent of age, sex, and other factors, can we predict RA response just using the gut microbiome alone?” Dr. Sung said. “Is there an association between clinical improvement and the gut microbiome?”
The study also showed that the microbiome may be a modifiable target for RA, Dr. Davis said.
“This research is attractive because it may complement medical treatment for RA if we can identify dietary modifications,” he said. “Still, there’s the question if probiotics or prebiotics can influence the gut. Can we modify the gut microbiome to further ameliorate the disease state? That’s something I think is an open question that’s specifically called out in our paper.”
This study included patients with long-term disease, but the group’s ongoing research is focusing on patients with earlier-stage RA, Dr. Davis said. “The next steps have to be in validating [the findings] in additional and external populations and looking at patients with very early disease where a lot of the decision-making is very active and happening in real time.”
James T. Rosenbaum, MD, an ophthalmologist and rheumatologist at Oregon Health & Science University, Portland, acknowledged that this is the first study in RA to find an effect on the gut microbiome using the minimum clinically important improvement endpoint.
“It also raises a ‘chicken-egg’ dilemma,” Dr. Rosenbaum said in an interview. “Did the patients improve and then their microbiome changed, or was the microbiome the first change that led to the clinical improvement? If the latter is correct, we potentially could alter the microbiome, for example, by diet, to treat rheumatic disease.”
He noted that studies with fecal transplants for ulcerative colitis support the therapeutic potential of microbiome modification. “But,” he added, “we are still a long way from putting this in practice.”
“The outcome is promising,” Claudia Mauri, PhD, a professor of immunology at University College London, said of the study. “Obviously, if this can be repeated in a very large cohort of patients, it would give us the possibility to be able to, based on the composition of the microbiota, to predict who is going to respond to treatment or not.”
She noted that, while RA has a broader array of available treatments than other autoimmune diseases, some RA patients don’t respond their first biologic treatment. “If from the outset we would be able to see who may not respond based on the microbiota, we may prepare physicians to better target these patients by, for example, offering them an alternative second biologic agent,” Dr. Mauri said.
Dr. Davis reported receiving research grants from Pfizer. Dr. Sung and other study coauthors have no financial relationships to disclose. Dr. Rosenbaum reported that the National Institutes of Health supports his research. Dr. Mauri has no financial relationships to disclose.
The gut microbiome, previously shown to have an association with rheumatoid arthritis, may also provide signals of a patient’s disease prognosis, researchers at the Mayo Clinic have reported.
“We found that the gut microbiome is linked to whether patients with RA improve in their clinical symptoms or not,” cosenior author Jaeyun Sung, PhD, said in an interview. “We found features of the gut microbiome that linked to improvement, and we also put those features in a machine-learning model that can actually predict improvement at a follow-up visit.” Dr. Sung is a computational biologist with Mayo Clinic’s Center for Individualized Medicine in Rochester, Minn.
The retrospective, observational cohort study included 32 patients diagnosed with RA between 1988 and 2014. The researchers performed meta-genome shotgun sequencing on 64 stool samples kept in a biobank and collected at two separate visits 6-12 months apart. Dr. Sung and colleagues observed significantly different microbiome traits between patients who eventually showed minimally clinically important improvement and those who didn’t.
The study also provided a proof of concept for using machine-learning technology to analyze the gut microbiome to predict the course of the disease, Dr. Sung said.
Cosenior author John M. Davis III, MD, a clinical rheumatologist and rheumatology research chair of the Mayo Clinic, noted that their own previous study had confirmed dysbiosis in people with RA when compared with controls. “We had some preliminary insight that it may be linked to some extent to the disease state and maybe treatments,” Dr. Davis said. “So that led us to hypothesize that there may be an association between the gut microbiome and response to treatment or disease activity over time.”
The study found that age was the dominant factor in determining variations in the gut microbiome composition, but the next prevailing factor was minimum clinically important improvement status, which 12 of the 32 study participants achieved at their follow-up visits. At baseline, all patients were on some type of treatment – either biologic or conventional disease-modifying antirheumatic drugs (DMARDs, 46.9% and 87.5%, respectively), or prednisone (46.9%).
Gut microbiome composition
The patients who achieved minimum clinically important improvement had an average decline in Clinical Disease Activity Index of 16.7 units (standard deviation, 12.8) versus a gain of 5.7 (SD, 8.9) in the remaining patients. The study found higher species-level alpha diversity and richness and higher beta diversity in the group that achieved minimum clinically important improvement, compared with those who did not.
They identified six microbial taxa as higher in abundance in the improved patients: Negativicutes (class); Selenomonadales (order); Prevotellaceae (family); Coprococcus (genus); Bacteroides sp. 3_1_19 (species); and Bilophila sp. 4_1_30 (species). In the patients who showed no improvement, Eubacterium sp. 3_1_31 (species) was found to be higher (P < .05). They also found 15 metabolic pathways that were differently abundant between the two groups at baseline.
Two things make this study different from other studies of the gut microbiome in RA, Dr. Sung said: It didn’t have a control group, only RA patients, and it didn’t evaluate a specific drug in RA patients.
“We’re thinking beyond just drug or treatment, independent of prior treatment, independent of prior clinical measurements, independent of age, sex, and other factors, can we predict RA response just using the gut microbiome alone?” Dr. Sung said. “Is there an association between clinical improvement and the gut microbiome?”
The study also showed that the microbiome may be a modifiable target for RA, Dr. Davis said.
“This research is attractive because it may complement medical treatment for RA if we can identify dietary modifications,” he said. “Still, there’s the question if probiotics or prebiotics can influence the gut. Can we modify the gut microbiome to further ameliorate the disease state? That’s something I think is an open question that’s specifically called out in our paper.”
This study included patients with long-term disease, but the group’s ongoing research is focusing on patients with earlier-stage RA, Dr. Davis said. “The next steps have to be in validating [the findings] in additional and external populations and looking at patients with very early disease where a lot of the decision-making is very active and happening in real time.”
James T. Rosenbaum, MD, an ophthalmologist and rheumatologist at Oregon Health & Science University, Portland, acknowledged that this is the first study in RA to find an effect on the gut microbiome using the minimum clinically important improvement endpoint.
“It also raises a ‘chicken-egg’ dilemma,” Dr. Rosenbaum said in an interview. “Did the patients improve and then their microbiome changed, or was the microbiome the first change that led to the clinical improvement? If the latter is correct, we potentially could alter the microbiome, for example, by diet, to treat rheumatic disease.”
He noted that studies with fecal transplants for ulcerative colitis support the therapeutic potential of microbiome modification. “But,” he added, “we are still a long way from putting this in practice.”
“The outcome is promising,” Claudia Mauri, PhD, a professor of immunology at University College London, said of the study. “Obviously, if this can be repeated in a very large cohort of patients, it would give us the possibility to be able to, based on the composition of the microbiota, to predict who is going to respond to treatment or not.”
She noted that, while RA has a broader array of available treatments than other autoimmune diseases, some RA patients don’t respond their first biologic treatment. “If from the outset we would be able to see who may not respond based on the microbiota, we may prepare physicians to better target these patients by, for example, offering them an alternative second biologic agent,” Dr. Mauri said.
Dr. Davis reported receiving research grants from Pfizer. Dr. Sung and other study coauthors have no financial relationships to disclose. Dr. Rosenbaum reported that the National Institutes of Health supports his research. Dr. Mauri has no financial relationships to disclose.
FROM GENOME MEDICINE
COVID-19 claims more than 675,000 U.S. lives, surpassing the 1918 flu
, according to data collected by Johns Hopkins University.
Although the raw numbers match, epidemiologists point out that 675,000 deaths in 1918 was a much greater proportion of the population. In 1918, the U.S. population was 105 million, less than one third of what it is today.
The AIDS pandemic of the 1980s remains the deadliest of the 20th Century, claiming the lives of 700,000 Americans. But at our current pace of 2,000 COVID deaths a day, we could quickly eclipse that death toll, too.
Even though the 1918 epidemic is often called the “Spanish Flu,” there is no universal consensus regarding where the virus originated, according to the Centers for Disease Control and Prevention.
Still, the almost incomprehensible loss harkens back to a time when medicine and technology were far less advanced than they are today.
In 1918, the United States didn’t have access to a vaccine, or near real-time tools to trace the spread and communicate the threat.
In some ways, the United States has failed to learn from the mistakes of the past.
There are many similarities between the two pandemics. In the spring of 1918, when the first wave of influenza hit, the United States and its allies were nearing victory in Europe in World War I. Just this summer the United States has ended its longest war, the conflict in Afghanistan, as COVID cases surge.
In both pandemics, hospitals and funeral homes were overrun and makeshift clinics were opened where space was available. Mask mandates were installed; schools, churches, and theaters closed; and social distancing was encouraged.
As is the case today, different jurisdictions took different steps to fight the pandemic and some were more successful than others.
According to History.com, in 1918, Philadelphia’s mayor said a popular annual parade could be held, and an estimated 200,000 people attended. In less than 2 weeks, more than 1,000 local residents were dead. But in St. Louis, public gatherings were banned, schools and theaters closed, and the death toll there was one eighth of Philadelphia’s.
Just as in 1918, America has at times continued to fan the flames of the epidemic by relaxing restrictions too quickly and relying on unproven treatments. Poor communication allowed younger people to feel that they wouldn’t necessarily face the worst consequences of the virus, contributing to a false sense of security in the age group that was fueling the spread.
“A lot of the mistakes that we definitely fell into in 1918, we hoped we wouldn’t fall into in 2020,” epidemiologist Stephen Kissler, PhD, of the Harvard T.H. Chan School of Public Health, told CNN. “We did.”
A version of this article first appeared on Medscape.com.
, according to data collected by Johns Hopkins University.
Although the raw numbers match, epidemiologists point out that 675,000 deaths in 1918 was a much greater proportion of the population. In 1918, the U.S. population was 105 million, less than one third of what it is today.
The AIDS pandemic of the 1980s remains the deadliest of the 20th Century, claiming the lives of 700,000 Americans. But at our current pace of 2,000 COVID deaths a day, we could quickly eclipse that death toll, too.
Even though the 1918 epidemic is often called the “Spanish Flu,” there is no universal consensus regarding where the virus originated, according to the Centers for Disease Control and Prevention.
Still, the almost incomprehensible loss harkens back to a time when medicine and technology were far less advanced than they are today.
In 1918, the United States didn’t have access to a vaccine, or near real-time tools to trace the spread and communicate the threat.
In some ways, the United States has failed to learn from the mistakes of the past.
There are many similarities between the two pandemics. In the spring of 1918, when the first wave of influenza hit, the United States and its allies were nearing victory in Europe in World War I. Just this summer the United States has ended its longest war, the conflict in Afghanistan, as COVID cases surge.
In both pandemics, hospitals and funeral homes were overrun and makeshift clinics were opened where space was available. Mask mandates were installed; schools, churches, and theaters closed; and social distancing was encouraged.
As is the case today, different jurisdictions took different steps to fight the pandemic and some were more successful than others.
According to History.com, in 1918, Philadelphia’s mayor said a popular annual parade could be held, and an estimated 200,000 people attended. In less than 2 weeks, more than 1,000 local residents were dead. But in St. Louis, public gatherings were banned, schools and theaters closed, and the death toll there was one eighth of Philadelphia’s.
Just as in 1918, America has at times continued to fan the flames of the epidemic by relaxing restrictions too quickly and relying on unproven treatments. Poor communication allowed younger people to feel that they wouldn’t necessarily face the worst consequences of the virus, contributing to a false sense of security in the age group that was fueling the spread.
“A lot of the mistakes that we definitely fell into in 1918, we hoped we wouldn’t fall into in 2020,” epidemiologist Stephen Kissler, PhD, of the Harvard T.H. Chan School of Public Health, told CNN. “We did.”
A version of this article first appeared on Medscape.com.
, according to data collected by Johns Hopkins University.
Although the raw numbers match, epidemiologists point out that 675,000 deaths in 1918 was a much greater proportion of the population. In 1918, the U.S. population was 105 million, less than one third of what it is today.
The AIDS pandemic of the 1980s remains the deadliest of the 20th Century, claiming the lives of 700,000 Americans. But at our current pace of 2,000 COVID deaths a day, we could quickly eclipse that death toll, too.
Even though the 1918 epidemic is often called the “Spanish Flu,” there is no universal consensus regarding where the virus originated, according to the Centers for Disease Control and Prevention.
Still, the almost incomprehensible loss harkens back to a time when medicine and technology were far less advanced than they are today.
In 1918, the United States didn’t have access to a vaccine, or near real-time tools to trace the spread and communicate the threat.
In some ways, the United States has failed to learn from the mistakes of the past.
There are many similarities between the two pandemics. In the spring of 1918, when the first wave of influenza hit, the United States and its allies were nearing victory in Europe in World War I. Just this summer the United States has ended its longest war, the conflict in Afghanistan, as COVID cases surge.
In both pandemics, hospitals and funeral homes were overrun and makeshift clinics were opened where space was available. Mask mandates were installed; schools, churches, and theaters closed; and social distancing was encouraged.
As is the case today, different jurisdictions took different steps to fight the pandemic and some were more successful than others.
According to History.com, in 1918, Philadelphia’s mayor said a popular annual parade could be held, and an estimated 200,000 people attended. In less than 2 weeks, more than 1,000 local residents were dead. But in St. Louis, public gatherings were banned, schools and theaters closed, and the death toll there was one eighth of Philadelphia’s.
Just as in 1918, America has at times continued to fan the flames of the epidemic by relaxing restrictions too quickly and relying on unproven treatments. Poor communication allowed younger people to feel that they wouldn’t necessarily face the worst consequences of the virus, contributing to a false sense of security in the age group that was fueling the spread.
“A lot of the mistakes that we definitely fell into in 1918, we hoped we wouldn’t fall into in 2020,” epidemiologist Stephen Kissler, PhD, of the Harvard T.H. Chan School of Public Health, told CNN. “We did.”
A version of this article first appeared on Medscape.com.
Guideline gives weak support to trying oral medical cannabis for chronic pain
“Evidence alone is not sufficient for clinical decision-making, particularly in chronic pain,” said Jason Busse, DC, PhD, director of Michael G. DeGroote Centre for Medicinal Cannabis Research at McMaster University, Hamilton, Ont., and lead author of a newly released rapid guideline on medical cannabis or cannabinoids for chronic pain.
The recommendations, published online Sept. 9, 2021 in the British Medical Journal, suggest that providers offer patients with chronic pain a trial of noninhaled medical cannabis or cannabinoids if standard care or management is ineffective. However, the “weak” rating attached to the recommendation may compel some clinicians to automatically write off the panel’s recommendations.
“Because of the close balance between benefits and harms and wide variability in patient attitudes, the panel came to the conclusion that [some] patients presented with the current best evidence would likely choose to engage in a trial of medicinal cannabis, if their current care was felt to be suboptimal,” Dr. Busse explained in an interview.
But more importantly, “the recommendation allows for shared decision making to occur, and for different patients to make different decisions based on individual preferences and circumstances,” he said.
Evidence supports improved pain and sleep quality, physical functioning
Evidence supporting the use of medical cannabis in chronic pain is derived from a rigorous systematic review and meta-analysis of 32 studies enrolling 5,174 patients randomized to oral (capsule, spray, sublingual drops) or topical (transdermal cream) medical cannabis or placebo. Of note, three types of cannabinoids were represented: phytocannabinoids, synthetic, and endocannabinoids.
The studies included both patients with chronic noncancer pain (28 studies, n = 3,812) and chronic cancer pain not receiving palliative care (4 studies, n = 1,362). On average, baseline pain scores were a median 6.28 cm on a 10-cm visual analog scale (VAS), and median participant age was 53 years. 60% of trials reporting sex differences enrolled female participants. Overall, patients were followed for roughly 2 months (median, 50 days).
Findings (27 studies, n = 3,939) showed that, compared with placebo, medical cannabis resulted in a small, albeit important, improvement in the proportion of patients experiencing pain relief at or above the minimally important difference (MID) (moderate-certainty evidence, 10% modeled risk difference [RD; 95% confidence interval, 5%-15%] for achieving at least the MID of 1 cm).
Medical cannabis (15 studies, n = 2,425) also provided a small increase in the proportion of patients experiencing improvements in physical functioning at or above the MID (high certainty evidence, 4% modeled RD [95% CI, 0.1%-8%] for achieving at least a MID of 10 points).
Additionally, participants experienced significant improvements in sleep quality, compared with placebo (16 studies, 3,124 participants, high-quality evidence), demonstrating a weighted mean difference of –0.53 cm on a 10-cm VAS (95% CI, –0.75 to –0.30 cm). A total of nine larger trials (n = 2,652, high-certainty evidence) saw a small increase in the proportion of patients experiencing improved sleep quality at or above the MID: 6% modeled RD (95% CI, 2%-9%).
On the other hand, benefits did not extend to emotional, role, or social functioning (high-certainty evidence).
First do no harm: Start low, go slow
While these findings provide a rationale for medical cannabis in chronic pain, exploring options with patients can be challenging. Studies on medical cannabis consistently note that patients want information, but data also show that many providers express a lack of knowledge to provide adequate counseling.
There are also legal hurdles. Despite the authorization of medicinal cannabis across a majority of states and territories, cannabis is still a schedule I substance under the Federal Controlled Substances Act. In addition, the absence of standards around formulations, potency, and dosing has also been cited as a major barrier to recommending medical cannabis, as have concerns about adverse events (AEs), especially with inhaled and tetrahydrocannabinol (THC)-predominant formulations.
Like most medications, medical cannabis dosing should be individualized depending on product, patient, and ability to titrate the dose, but the guidelines provide a general rule of thumb. Providers considering therapeutic noninhaled medical cannabis trials are encouraged to start with a low-dose cannabidiol (CBD) oral tablet, spray, or sublingual oil drops 5 mg twice daily, increasing it by 10 mg every 2-3 days depending on the clinical response (to a maximum daily dose of 40 mg/day). If patient response is unsatisfactory, they should consider adding 1-2.5 mg THC/daily, titrated every 2-7 days to a maximum of 40 mg/day.
Still, an important caveat is whether or not adjunctive CBD alone is effective for chronic pain.
“While we know that one out of seven U.S. adults are using cannabidiol, we know very little about its therapeutic effects when given by itself for pain,” Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, and an associate professor at-large of psychology and behavioral science, said in an interview. (Dr. Cooper was not involved in the guideline development.)
“But patients tend to self-report that CBD is helpful, and at low doses, we know that it is unlikely to have adverse effects of any significant concern,” Dr. Cooper noted.
Depending on its components, medical cannabis is associated with a wide range of AEs. Studies comprising the evidence base for the guideline reported transient cognitive impairment (relative risk, 2.39; 95% CI, 1.06-5.38), vomiting (RR, 1.46; 95% CI, 1.07-1.99), and drowsiness (RR, 2.14; 95% CI, 1.55-2.95), attention impairment (RR, 4.04; 95% CI, 1.67-9.74), and nausea (RR, 1.59; 95% CI, 1.28-1.99). Of note, findings of a subgroup analysis showed that the risk of dizziness increased with treatment duration, starting at 3 months (test of interaction P = .002).
However, Dr. Cooper explained that, because the included studies were inconsistent in terms of cannabis type (e.g., some looked at synthetic THC or THC-like substances where others looked at a THC/CBD combination) and formulation (capsules, oral mucosal sprays), it’s difficult to tease out component-specific AEs.
“These are really important things to note, especially when you think about different populations that might be using these types of medicines moving forward,” she said.
Toward that end, the guideline specifically states that there is “no reason why the expected benefits would be systematically different among adolescents and emerging adults.”
Among children with cancer, prior study findings reinforce the conclusion that benefits are similar to adults, but studies in this area are limited to end-of-life treatment, childhood cancer with primarily palliative intent, or progressive or relapsed cancer. Because THC’s safety profile is less certain in children, it’s also important to consider adverse neurocognitive effects before initiating a medical cannabis trial in this population.
Navigating the landscape
Although promising, the medical cannabis landscape is undoubtedly difficult to navigate, with land mines ranging from a limited inability to simply pick up a prescribing pad to quality control.
With the exception of three Food and Drug Administration–approved products – dronabinol, cannabidiol Rx, and nabilone – U.S. providers are only able to ‘certify,’ not prescribe, medical cannabis for chronic pain, and only if it is included within the state cannabis board’s list of eligible conditions. (A state-by-state guide is available.)
Quality control also varies by product but is critical. “You want to look for certificates of quality assurance,” Jenny Wilkerson, PhD, a research assistant professor of pharmacodynamics at the University of Florida, Gainesville, said in an interview. (Dr. Wilkerson was not involved in the guideline development.)
“A good dispensary should have that information or at least be willing to get that information, but generally speaking, that is something that patients need to ask for,” she emphasized, noting that “most available mass readouts are not divided by lots.”
Initial counseling and AE monitoring and regular follow-up is important, especially among patients who’ve never tried medical cannabis (or older patients whose prior experience may be limited to weaker recreational marijuana).
Notably, the reliance on medical dispensaries to deliver the right information at the right time may prove to be faulty. While recent data show that frontline dispensary workers regularly provide information to customers on their medical conditions and available products, they rarely, if ever, base recommendations on provider input, and never or rarely discuss potential AEs and other risks.
Per the new guideline, inexperienced patients should be seen monthly until a stable dose is achieved; longer times between visits can be considered in those who are more experienced. Still, patients should be advised to contact their provider when pain relief or other goals are insufficient, or when response or problematic AEs occur. This facilitates down-titration to a previously tolerated dose, up-titration in CBD and/or THC, or a different route of administration/formulation altogether.
Dr. Wilkerson pointed out that follow-up visits also provide an opportunity to do a blood draw and ask the lab to conduct pharmacokinetic analysis.
If possible, “ask patients to [ensure that they] take a standard dose before the visit so that the lab can assess the blood percentage of primary compounds and metabolites in the product that they are using,” she explained, noting that the information is helping to determine how “the different ratios may be affecting therapeutic response in individual patients.”
Granted, the guideline is only a start. But it is a good one.
“A lot of physicians want to be able to hang their hat on evidence of the safety and efficacy of these products, and the analysis that was leveraged for this guideline was very rigorous,” Dr. Cooper said.
Not only do they reinforce that “oral cannabinoids can produce small improvements in pain and provide a dosing structure that minimizes risk to the patient, [but they] should be able to help educate physicians who [are looking] for a sense of what the literature tells us at this time,” she added.
“With chronic pain, we often find that different treatments will show small potential benefits and they have a certain risk profile,” Dr. Busse said.
“It’s almost impossible to know what patients think about this option unless you present them with the evidence and ask them to make a decision based on their values and preferences,” he said.
The Michael G. DeGroote Centre for Medicinal Cannabis Research funded the MAGIC Evidence Ecosystem Foundation to support the creation of the guideline. The center receives no funding from industry Dr. Busse, Dr. Cooper, and Dr. Wilkerson reported having no relevant financial relationships.
“Evidence alone is not sufficient for clinical decision-making, particularly in chronic pain,” said Jason Busse, DC, PhD, director of Michael G. DeGroote Centre for Medicinal Cannabis Research at McMaster University, Hamilton, Ont., and lead author of a newly released rapid guideline on medical cannabis or cannabinoids for chronic pain.
The recommendations, published online Sept. 9, 2021 in the British Medical Journal, suggest that providers offer patients with chronic pain a trial of noninhaled medical cannabis or cannabinoids if standard care or management is ineffective. However, the “weak” rating attached to the recommendation may compel some clinicians to automatically write off the panel’s recommendations.
“Because of the close balance between benefits and harms and wide variability in patient attitudes, the panel came to the conclusion that [some] patients presented with the current best evidence would likely choose to engage in a trial of medicinal cannabis, if their current care was felt to be suboptimal,” Dr. Busse explained in an interview.
But more importantly, “the recommendation allows for shared decision making to occur, and for different patients to make different decisions based on individual preferences and circumstances,” he said.
Evidence supports improved pain and sleep quality, physical functioning
Evidence supporting the use of medical cannabis in chronic pain is derived from a rigorous systematic review and meta-analysis of 32 studies enrolling 5,174 patients randomized to oral (capsule, spray, sublingual drops) or topical (transdermal cream) medical cannabis or placebo. Of note, three types of cannabinoids were represented: phytocannabinoids, synthetic, and endocannabinoids.
The studies included both patients with chronic noncancer pain (28 studies, n = 3,812) and chronic cancer pain not receiving palliative care (4 studies, n = 1,362). On average, baseline pain scores were a median 6.28 cm on a 10-cm visual analog scale (VAS), and median participant age was 53 years. 60% of trials reporting sex differences enrolled female participants. Overall, patients were followed for roughly 2 months (median, 50 days).
Findings (27 studies, n = 3,939) showed that, compared with placebo, medical cannabis resulted in a small, albeit important, improvement in the proportion of patients experiencing pain relief at or above the minimally important difference (MID) (moderate-certainty evidence, 10% modeled risk difference [RD; 95% confidence interval, 5%-15%] for achieving at least the MID of 1 cm).
Medical cannabis (15 studies, n = 2,425) also provided a small increase in the proportion of patients experiencing improvements in physical functioning at or above the MID (high certainty evidence, 4% modeled RD [95% CI, 0.1%-8%] for achieving at least a MID of 10 points).
Additionally, participants experienced significant improvements in sleep quality, compared with placebo (16 studies, 3,124 participants, high-quality evidence), demonstrating a weighted mean difference of –0.53 cm on a 10-cm VAS (95% CI, –0.75 to –0.30 cm). A total of nine larger trials (n = 2,652, high-certainty evidence) saw a small increase in the proportion of patients experiencing improved sleep quality at or above the MID: 6% modeled RD (95% CI, 2%-9%).
On the other hand, benefits did not extend to emotional, role, or social functioning (high-certainty evidence).
First do no harm: Start low, go slow
While these findings provide a rationale for medical cannabis in chronic pain, exploring options with patients can be challenging. Studies on medical cannabis consistently note that patients want information, but data also show that many providers express a lack of knowledge to provide adequate counseling.
There are also legal hurdles. Despite the authorization of medicinal cannabis across a majority of states and territories, cannabis is still a schedule I substance under the Federal Controlled Substances Act. In addition, the absence of standards around formulations, potency, and dosing has also been cited as a major barrier to recommending medical cannabis, as have concerns about adverse events (AEs), especially with inhaled and tetrahydrocannabinol (THC)-predominant formulations.
Like most medications, medical cannabis dosing should be individualized depending on product, patient, and ability to titrate the dose, but the guidelines provide a general rule of thumb. Providers considering therapeutic noninhaled medical cannabis trials are encouraged to start with a low-dose cannabidiol (CBD) oral tablet, spray, or sublingual oil drops 5 mg twice daily, increasing it by 10 mg every 2-3 days depending on the clinical response (to a maximum daily dose of 40 mg/day). If patient response is unsatisfactory, they should consider adding 1-2.5 mg THC/daily, titrated every 2-7 days to a maximum of 40 mg/day.
Still, an important caveat is whether or not adjunctive CBD alone is effective for chronic pain.
“While we know that one out of seven U.S. adults are using cannabidiol, we know very little about its therapeutic effects when given by itself for pain,” Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, and an associate professor at-large of psychology and behavioral science, said in an interview. (Dr. Cooper was not involved in the guideline development.)
“But patients tend to self-report that CBD is helpful, and at low doses, we know that it is unlikely to have adverse effects of any significant concern,” Dr. Cooper noted.
Depending on its components, medical cannabis is associated with a wide range of AEs. Studies comprising the evidence base for the guideline reported transient cognitive impairment (relative risk, 2.39; 95% CI, 1.06-5.38), vomiting (RR, 1.46; 95% CI, 1.07-1.99), and drowsiness (RR, 2.14; 95% CI, 1.55-2.95), attention impairment (RR, 4.04; 95% CI, 1.67-9.74), and nausea (RR, 1.59; 95% CI, 1.28-1.99). Of note, findings of a subgroup analysis showed that the risk of dizziness increased with treatment duration, starting at 3 months (test of interaction P = .002).
However, Dr. Cooper explained that, because the included studies were inconsistent in terms of cannabis type (e.g., some looked at synthetic THC or THC-like substances where others looked at a THC/CBD combination) and formulation (capsules, oral mucosal sprays), it’s difficult to tease out component-specific AEs.
“These are really important things to note, especially when you think about different populations that might be using these types of medicines moving forward,” she said.
Toward that end, the guideline specifically states that there is “no reason why the expected benefits would be systematically different among adolescents and emerging adults.”
Among children with cancer, prior study findings reinforce the conclusion that benefits are similar to adults, but studies in this area are limited to end-of-life treatment, childhood cancer with primarily palliative intent, or progressive or relapsed cancer. Because THC’s safety profile is less certain in children, it’s also important to consider adverse neurocognitive effects before initiating a medical cannabis trial in this population.
Navigating the landscape
Although promising, the medical cannabis landscape is undoubtedly difficult to navigate, with land mines ranging from a limited inability to simply pick up a prescribing pad to quality control.
With the exception of three Food and Drug Administration–approved products – dronabinol, cannabidiol Rx, and nabilone – U.S. providers are only able to ‘certify,’ not prescribe, medical cannabis for chronic pain, and only if it is included within the state cannabis board’s list of eligible conditions. (A state-by-state guide is available.)
Quality control also varies by product but is critical. “You want to look for certificates of quality assurance,” Jenny Wilkerson, PhD, a research assistant professor of pharmacodynamics at the University of Florida, Gainesville, said in an interview. (Dr. Wilkerson was not involved in the guideline development.)
“A good dispensary should have that information or at least be willing to get that information, but generally speaking, that is something that patients need to ask for,” she emphasized, noting that “most available mass readouts are not divided by lots.”
Initial counseling and AE monitoring and regular follow-up is important, especially among patients who’ve never tried medical cannabis (or older patients whose prior experience may be limited to weaker recreational marijuana).
Notably, the reliance on medical dispensaries to deliver the right information at the right time may prove to be faulty. While recent data show that frontline dispensary workers regularly provide information to customers on their medical conditions and available products, they rarely, if ever, base recommendations on provider input, and never or rarely discuss potential AEs and other risks.
Per the new guideline, inexperienced patients should be seen monthly until a stable dose is achieved; longer times between visits can be considered in those who are more experienced. Still, patients should be advised to contact their provider when pain relief or other goals are insufficient, or when response or problematic AEs occur. This facilitates down-titration to a previously tolerated dose, up-titration in CBD and/or THC, or a different route of administration/formulation altogether.
Dr. Wilkerson pointed out that follow-up visits also provide an opportunity to do a blood draw and ask the lab to conduct pharmacokinetic analysis.
If possible, “ask patients to [ensure that they] take a standard dose before the visit so that the lab can assess the blood percentage of primary compounds and metabolites in the product that they are using,” she explained, noting that the information is helping to determine how “the different ratios may be affecting therapeutic response in individual patients.”
Granted, the guideline is only a start. But it is a good one.
“A lot of physicians want to be able to hang their hat on evidence of the safety and efficacy of these products, and the analysis that was leveraged for this guideline was very rigorous,” Dr. Cooper said.
Not only do they reinforce that “oral cannabinoids can produce small improvements in pain and provide a dosing structure that minimizes risk to the patient, [but they] should be able to help educate physicians who [are looking] for a sense of what the literature tells us at this time,” she added.
“With chronic pain, we often find that different treatments will show small potential benefits and they have a certain risk profile,” Dr. Busse said.
“It’s almost impossible to know what patients think about this option unless you present them with the evidence and ask them to make a decision based on their values and preferences,” he said.
The Michael G. DeGroote Centre for Medicinal Cannabis Research funded the MAGIC Evidence Ecosystem Foundation to support the creation of the guideline. The center receives no funding from industry Dr. Busse, Dr. Cooper, and Dr. Wilkerson reported having no relevant financial relationships.
“Evidence alone is not sufficient for clinical decision-making, particularly in chronic pain,” said Jason Busse, DC, PhD, director of Michael G. DeGroote Centre for Medicinal Cannabis Research at McMaster University, Hamilton, Ont., and lead author of a newly released rapid guideline on medical cannabis or cannabinoids for chronic pain.
The recommendations, published online Sept. 9, 2021 in the British Medical Journal, suggest that providers offer patients with chronic pain a trial of noninhaled medical cannabis or cannabinoids if standard care or management is ineffective. However, the “weak” rating attached to the recommendation may compel some clinicians to automatically write off the panel’s recommendations.
“Because of the close balance between benefits and harms and wide variability in patient attitudes, the panel came to the conclusion that [some] patients presented with the current best evidence would likely choose to engage in a trial of medicinal cannabis, if their current care was felt to be suboptimal,” Dr. Busse explained in an interview.
But more importantly, “the recommendation allows for shared decision making to occur, and for different patients to make different decisions based on individual preferences and circumstances,” he said.
Evidence supports improved pain and sleep quality, physical functioning
Evidence supporting the use of medical cannabis in chronic pain is derived from a rigorous systematic review and meta-analysis of 32 studies enrolling 5,174 patients randomized to oral (capsule, spray, sublingual drops) or topical (transdermal cream) medical cannabis or placebo. Of note, three types of cannabinoids were represented: phytocannabinoids, synthetic, and endocannabinoids.
The studies included both patients with chronic noncancer pain (28 studies, n = 3,812) and chronic cancer pain not receiving palliative care (4 studies, n = 1,362). On average, baseline pain scores were a median 6.28 cm on a 10-cm visual analog scale (VAS), and median participant age was 53 years. 60% of trials reporting sex differences enrolled female participants. Overall, patients were followed for roughly 2 months (median, 50 days).
Findings (27 studies, n = 3,939) showed that, compared with placebo, medical cannabis resulted in a small, albeit important, improvement in the proportion of patients experiencing pain relief at or above the minimally important difference (MID) (moderate-certainty evidence, 10% modeled risk difference [RD; 95% confidence interval, 5%-15%] for achieving at least the MID of 1 cm).
Medical cannabis (15 studies, n = 2,425) also provided a small increase in the proportion of patients experiencing improvements in physical functioning at or above the MID (high certainty evidence, 4% modeled RD [95% CI, 0.1%-8%] for achieving at least a MID of 10 points).
Additionally, participants experienced significant improvements in sleep quality, compared with placebo (16 studies, 3,124 participants, high-quality evidence), demonstrating a weighted mean difference of –0.53 cm on a 10-cm VAS (95% CI, –0.75 to –0.30 cm). A total of nine larger trials (n = 2,652, high-certainty evidence) saw a small increase in the proportion of patients experiencing improved sleep quality at or above the MID: 6% modeled RD (95% CI, 2%-9%).
On the other hand, benefits did not extend to emotional, role, or social functioning (high-certainty evidence).
First do no harm: Start low, go slow
While these findings provide a rationale for medical cannabis in chronic pain, exploring options with patients can be challenging. Studies on medical cannabis consistently note that patients want information, but data also show that many providers express a lack of knowledge to provide adequate counseling.
There are also legal hurdles. Despite the authorization of medicinal cannabis across a majority of states and territories, cannabis is still a schedule I substance under the Federal Controlled Substances Act. In addition, the absence of standards around formulations, potency, and dosing has also been cited as a major barrier to recommending medical cannabis, as have concerns about adverse events (AEs), especially with inhaled and tetrahydrocannabinol (THC)-predominant formulations.
Like most medications, medical cannabis dosing should be individualized depending on product, patient, and ability to titrate the dose, but the guidelines provide a general rule of thumb. Providers considering therapeutic noninhaled medical cannabis trials are encouraged to start with a low-dose cannabidiol (CBD) oral tablet, spray, or sublingual oil drops 5 mg twice daily, increasing it by 10 mg every 2-3 days depending on the clinical response (to a maximum daily dose of 40 mg/day). If patient response is unsatisfactory, they should consider adding 1-2.5 mg THC/daily, titrated every 2-7 days to a maximum of 40 mg/day.
Still, an important caveat is whether or not adjunctive CBD alone is effective for chronic pain.
“While we know that one out of seven U.S. adults are using cannabidiol, we know very little about its therapeutic effects when given by itself for pain,” Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, and an associate professor at-large of psychology and behavioral science, said in an interview. (Dr. Cooper was not involved in the guideline development.)
“But patients tend to self-report that CBD is helpful, and at low doses, we know that it is unlikely to have adverse effects of any significant concern,” Dr. Cooper noted.
Depending on its components, medical cannabis is associated with a wide range of AEs. Studies comprising the evidence base for the guideline reported transient cognitive impairment (relative risk, 2.39; 95% CI, 1.06-5.38), vomiting (RR, 1.46; 95% CI, 1.07-1.99), and drowsiness (RR, 2.14; 95% CI, 1.55-2.95), attention impairment (RR, 4.04; 95% CI, 1.67-9.74), and nausea (RR, 1.59; 95% CI, 1.28-1.99). Of note, findings of a subgroup analysis showed that the risk of dizziness increased with treatment duration, starting at 3 months (test of interaction P = .002).
However, Dr. Cooper explained that, because the included studies were inconsistent in terms of cannabis type (e.g., some looked at synthetic THC or THC-like substances where others looked at a THC/CBD combination) and formulation (capsules, oral mucosal sprays), it’s difficult to tease out component-specific AEs.
“These are really important things to note, especially when you think about different populations that might be using these types of medicines moving forward,” she said.
Toward that end, the guideline specifically states that there is “no reason why the expected benefits would be systematically different among adolescents and emerging adults.”
Among children with cancer, prior study findings reinforce the conclusion that benefits are similar to adults, but studies in this area are limited to end-of-life treatment, childhood cancer with primarily palliative intent, or progressive or relapsed cancer. Because THC’s safety profile is less certain in children, it’s also important to consider adverse neurocognitive effects before initiating a medical cannabis trial in this population.
Navigating the landscape
Although promising, the medical cannabis landscape is undoubtedly difficult to navigate, with land mines ranging from a limited inability to simply pick up a prescribing pad to quality control.
With the exception of three Food and Drug Administration–approved products – dronabinol, cannabidiol Rx, and nabilone – U.S. providers are only able to ‘certify,’ not prescribe, medical cannabis for chronic pain, and only if it is included within the state cannabis board’s list of eligible conditions. (A state-by-state guide is available.)
Quality control also varies by product but is critical. “You want to look for certificates of quality assurance,” Jenny Wilkerson, PhD, a research assistant professor of pharmacodynamics at the University of Florida, Gainesville, said in an interview. (Dr. Wilkerson was not involved in the guideline development.)
“A good dispensary should have that information or at least be willing to get that information, but generally speaking, that is something that patients need to ask for,” she emphasized, noting that “most available mass readouts are not divided by lots.”
Initial counseling and AE monitoring and regular follow-up is important, especially among patients who’ve never tried medical cannabis (or older patients whose prior experience may be limited to weaker recreational marijuana).
Notably, the reliance on medical dispensaries to deliver the right information at the right time may prove to be faulty. While recent data show that frontline dispensary workers regularly provide information to customers on their medical conditions and available products, they rarely, if ever, base recommendations on provider input, and never or rarely discuss potential AEs and other risks.
Per the new guideline, inexperienced patients should be seen monthly until a stable dose is achieved; longer times between visits can be considered in those who are more experienced. Still, patients should be advised to contact their provider when pain relief or other goals are insufficient, or when response or problematic AEs occur. This facilitates down-titration to a previously tolerated dose, up-titration in CBD and/or THC, or a different route of administration/formulation altogether.
Dr. Wilkerson pointed out that follow-up visits also provide an opportunity to do a blood draw and ask the lab to conduct pharmacokinetic analysis.
If possible, “ask patients to [ensure that they] take a standard dose before the visit so that the lab can assess the blood percentage of primary compounds and metabolites in the product that they are using,” she explained, noting that the information is helping to determine how “the different ratios may be affecting therapeutic response in individual patients.”
Granted, the guideline is only a start. But it is a good one.
“A lot of physicians want to be able to hang their hat on evidence of the safety and efficacy of these products, and the analysis that was leveraged for this guideline was very rigorous,” Dr. Cooper said.
Not only do they reinforce that “oral cannabinoids can produce small improvements in pain and provide a dosing structure that minimizes risk to the patient, [but they] should be able to help educate physicians who [are looking] for a sense of what the literature tells us at this time,” she added.
“With chronic pain, we often find that different treatments will show small potential benefits and they have a certain risk profile,” Dr. Busse said.
“It’s almost impossible to know what patients think about this option unless you present them with the evidence and ask them to make a decision based on their values and preferences,” he said.
The Michael G. DeGroote Centre for Medicinal Cannabis Research funded the MAGIC Evidence Ecosystem Foundation to support the creation of the guideline. The center receives no funding from industry Dr. Busse, Dr. Cooper, and Dr. Wilkerson reported having no relevant financial relationships.
FROM THE BMJ
Moderna vaccine more effective than Pfizer and J&J
the Centers for Disease Control and Protection has said.
“Among U.S. adults without immunocompromising conditions, vaccine effectiveness against COVID-19 hospitalization during March 11–Aug. 15, 2021, was higher for the Moderna vaccine (93%) than the Pfizer-BioNTech vaccine (88%) and the Janssen vaccine (71%),” the agency’s Morbidity and Mortality Weekly Report said. Janssen refers to the Johnson & Johnson vaccine.
The CDC said the data could help people make informed decisions.
“Understanding differences in VE [vaccine effectiveness] by vaccine product can guide individual choices and policy recommendations regarding vaccine boosters. All Food and Drug Administration–approved or authorized COVID-19 vaccines provide substantial protection against COVID-19 hospitalization,” the report said.
The study also broke down effectiveness for longer periods. Moderna came out on top again.
After 120 days, the Moderna vaccine provided 92% effectiveness against hospitalization, whereas the Pfizer vaccine’s effectiveness dropped to 77%, the CDC said. There was no similar calculation for the Johnson & Johnson vaccine.
The CDC studied 3,689 adults at 21 hospitals in 18 states who got the two-shot Pfizer or Moderna vaccine or the one-shot Johnson & Johnson vaccine between March and August.
The agency noted some factors that could have come into play.
“Differences in vaccine effectiveness between the Moderna and Pfizer-BioNTech vaccine might be due to higher mRNA content in the Moderna vaccine, differences in timing between doses (3 weeks for Pfizer-BioNTech vs. 4 weeks for Moderna), or possible differences between groups that received each vaccine that were not accounted for in the analysis,” the report said.
The CDC noted limitations in the findings. Children, immunocompromised adults, and vaccine effectiveness against COVID-19 that did not result in hospitalization were not studied.
Other studies have shown all three U.S. vaccines provide a high rate of protection against coronavirus.
A version of this article first appeared on WebMD.com.
the Centers for Disease Control and Protection has said.
“Among U.S. adults without immunocompromising conditions, vaccine effectiveness against COVID-19 hospitalization during March 11–Aug. 15, 2021, was higher for the Moderna vaccine (93%) than the Pfizer-BioNTech vaccine (88%) and the Janssen vaccine (71%),” the agency’s Morbidity and Mortality Weekly Report said. Janssen refers to the Johnson & Johnson vaccine.
The CDC said the data could help people make informed decisions.
“Understanding differences in VE [vaccine effectiveness] by vaccine product can guide individual choices and policy recommendations regarding vaccine boosters. All Food and Drug Administration–approved or authorized COVID-19 vaccines provide substantial protection against COVID-19 hospitalization,” the report said.
The study also broke down effectiveness for longer periods. Moderna came out on top again.
After 120 days, the Moderna vaccine provided 92% effectiveness against hospitalization, whereas the Pfizer vaccine’s effectiveness dropped to 77%, the CDC said. There was no similar calculation for the Johnson & Johnson vaccine.
The CDC studied 3,689 adults at 21 hospitals in 18 states who got the two-shot Pfizer or Moderna vaccine or the one-shot Johnson & Johnson vaccine between March and August.
The agency noted some factors that could have come into play.
“Differences in vaccine effectiveness between the Moderna and Pfizer-BioNTech vaccine might be due to higher mRNA content in the Moderna vaccine, differences in timing between doses (3 weeks for Pfizer-BioNTech vs. 4 weeks for Moderna), or possible differences between groups that received each vaccine that were not accounted for in the analysis,” the report said.
The CDC noted limitations in the findings. Children, immunocompromised adults, and vaccine effectiveness against COVID-19 that did not result in hospitalization were not studied.
Other studies have shown all three U.S. vaccines provide a high rate of protection against coronavirus.
A version of this article first appeared on WebMD.com.
the Centers for Disease Control and Protection has said.
“Among U.S. adults without immunocompromising conditions, vaccine effectiveness against COVID-19 hospitalization during March 11–Aug. 15, 2021, was higher for the Moderna vaccine (93%) than the Pfizer-BioNTech vaccine (88%) and the Janssen vaccine (71%),” the agency’s Morbidity and Mortality Weekly Report said. Janssen refers to the Johnson & Johnson vaccine.
The CDC said the data could help people make informed decisions.
“Understanding differences in VE [vaccine effectiveness] by vaccine product can guide individual choices and policy recommendations regarding vaccine boosters. All Food and Drug Administration–approved or authorized COVID-19 vaccines provide substantial protection against COVID-19 hospitalization,” the report said.
The study also broke down effectiveness for longer periods. Moderna came out on top again.
After 120 days, the Moderna vaccine provided 92% effectiveness against hospitalization, whereas the Pfizer vaccine’s effectiveness dropped to 77%, the CDC said. There was no similar calculation for the Johnson & Johnson vaccine.
The CDC studied 3,689 adults at 21 hospitals in 18 states who got the two-shot Pfizer or Moderna vaccine or the one-shot Johnson & Johnson vaccine between March and August.
The agency noted some factors that could have come into play.
“Differences in vaccine effectiveness between the Moderna and Pfizer-BioNTech vaccine might be due to higher mRNA content in the Moderna vaccine, differences in timing between doses (3 weeks for Pfizer-BioNTech vs. 4 weeks for Moderna), or possible differences between groups that received each vaccine that were not accounted for in the analysis,” the report said.
The CDC noted limitations in the findings. Children, immunocompromised adults, and vaccine effectiveness against COVID-19 that did not result in hospitalization were not studied.
Other studies have shown all three U.S. vaccines provide a high rate of protection against coronavirus.
A version of this article first appeared on WebMD.com.
Beware patients’ health plans that skirt state laws on specialty drug access
There is a dangerous trend in our country in which employers, seeking to reduce health plan costs they pay, enter into agreements with small third-party administrators that “carve out” specialty drug benefits from their self-funded health insurance plan. What employers are not told is that these spending reductions are accomplished by risking the health of their employees. It is the self-funded businesses that are being preyed upon by these administrators because there is a lot of money to be made by carving out the specialty drugs in their self-funded health plan.
Let’s start with a little primer on “fully insured” versus “self-funded” health plans. As a small business owner, I understand the need to make sure that expenses don’t outpace revenue if I want to keep my doors open. One of the largest expenses for any business is health insurance. My private rheumatology practice uses a fully insured health plan. In a fully insured plan, the insurer is the party taking the financial risk. We pay the premiums, and the insurance company pays the bills after the deductible is met. It may cost more in premiums than a self-funded plan, but if an employee has an accident or severe illness, our practice is not responsible for the cost of care.
On the other hand, large and small businesses that are self-funded cover the health costs of their employees themselves. These businesses will hire a third-party administrator to pay the bills out of an account that is supplied with money from the business owner. Looking at the insurance card of your patient is one way to tell if they are covered by a fully insured or self-funded plan. If the insurance card says the plan is “administered by” the insurer or “administrative services only,” it is most likely a self-funded plan. If their insurance card states “underwritten by” the insurer on the card then it is likely a fully insured plan. This becomes important because self-funded plans are not subject to the jurisdiction of state laws such as utilization management reform. These state laws are preempted from applying to self-funded plans by the Employee Retirement Income Security Act of 1974. The Rutledge v. Pharmaceutical Care Management Association Supreme Court case took up the question of whether certain state laws impermissibly applied or were connected to self-funded plans. The ruling in favor of Rutledge opens the door that certain state legislation may one day apply to self-funded plans.
Specialty drug benefit carve outs are not in best interests of employees, employers
This piece is not about Rutledge but about the small third-party administrators that are convincing self-funded businesses to let them “carve out” specialty drug benefits from the larger administrator of the plan by promising huge savings in the employer’s specialty drug spending. Two such companies that have come to the attention of the Coalition of State Rheumatology Organizations are Vivio Health and Archimedes. CSRO has received numerous complaints from rheumatologists regarding interference from these two entities with their clinical decision making and disregard for standard of care.
Vivio’s website reveals a disturbing approach to cost reduction. The website states that Vivio profiles physicians through ProPublica and Open Payments to determine if they are prescribing for the right reason and not for self-interest. This serves as an attempt to set up mistrust of the physician by the employer. Vivio’s website also states that the Food and Drug Administration has declining standards for approval of drugs, and consequently many approved drugs should be considered “experimental.” They say that business owners should not have to pay for “experimental” treatments. Through conversation with Vivio, it appears they believe that oncologists could be replaced by a primary care physician with the right algorithm.
Many of Vivio’s egregious behaviors are enumerated in our letter to Vivio: outrageous nonmedical switches, mandatory biologic tapering, and site of care changes. In all of the complaints that we received, Vivio attempted to switch patients to the same infusible medication, Renflexis, and also mandated white bagging, which means the payer has a specialty pharmacy ship a patient’s medication directly to the physician’s office for administration. This switch was made regardless of the mechanism of action or route of administration of the drug that had stabilized the patient. Peer-to-peer reviews with a retired radiologist led to routinely denied appeals and would even force the patient to a different site of care if the rheumatologist refused the new treatment or the mandated white bagging.
Our letter resulted in two conversations with Vivio. Vivio insisted that it was using American College of Rheumatology guidelines and comparisons between drug studies to make these decisions. The company stated that patients can be switched to any drug that has the same ACR 20, 50, and 70 response criteria outcomes as the drug that they are presently taking, even though these sorts of comparisons of results across completely different studies are invalid for a number of reasons, including because they do not have the same patient populations, protocols, and inclusion/exclusion criteria. These are dangerous policies and thus far we have not been able to find any rheumatologists who have gone along with these demands.
Companies such as Vivio are spreading, and employers are unaware that their policies are only paying lip service to “individualized care” while maintaining an approach to patient care that is focused only on cost cutting. Indeed, Archimedes represents one such metastasis. Their practices are similar to those of Vivio and of which CSRO has received complaints. Archimedes has similarly attempted to mandate white-bagging for the enrollees it manages and switch stable patients for nonmedical reasons to an entirely different molecule and mechanism of action.
Business owners do not understand the harm that these policies can cause their employees. This harm increases downstream medical spending as a result of loss of control of disease activity.
This is a call to action for all advocates and advocacy groups to get in the room with employer/business groups and explain how these third-party administrators, carving out specialty drug benefits, can ultimately cause physical harm to employees and increase monetary cost to the employer in the long run. Rheumatology as a specialty needs to educate employers and work out ways to save money for them and, at the same time, maintain excellence in care for their employees. CSRO has a letter it used successfully with the human resources department of Edward Jones to effect a change in its policies on this matter, which you are welcome to use to craft your own to businesses.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is President of the CSRO, past chair of the Alliance for Safe Biologic Medicines, and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
There is a dangerous trend in our country in which employers, seeking to reduce health plan costs they pay, enter into agreements with small third-party administrators that “carve out” specialty drug benefits from their self-funded health insurance plan. What employers are not told is that these spending reductions are accomplished by risking the health of their employees. It is the self-funded businesses that are being preyed upon by these administrators because there is a lot of money to be made by carving out the specialty drugs in their self-funded health plan.
Let’s start with a little primer on “fully insured” versus “self-funded” health plans. As a small business owner, I understand the need to make sure that expenses don’t outpace revenue if I want to keep my doors open. One of the largest expenses for any business is health insurance. My private rheumatology practice uses a fully insured health plan. In a fully insured plan, the insurer is the party taking the financial risk. We pay the premiums, and the insurance company pays the bills after the deductible is met. It may cost more in premiums than a self-funded plan, but if an employee has an accident or severe illness, our practice is not responsible for the cost of care.
On the other hand, large and small businesses that are self-funded cover the health costs of their employees themselves. These businesses will hire a third-party administrator to pay the bills out of an account that is supplied with money from the business owner. Looking at the insurance card of your patient is one way to tell if they are covered by a fully insured or self-funded plan. If the insurance card says the plan is “administered by” the insurer or “administrative services only,” it is most likely a self-funded plan. If their insurance card states “underwritten by” the insurer on the card then it is likely a fully insured plan. This becomes important because self-funded plans are not subject to the jurisdiction of state laws such as utilization management reform. These state laws are preempted from applying to self-funded plans by the Employee Retirement Income Security Act of 1974. The Rutledge v. Pharmaceutical Care Management Association Supreme Court case took up the question of whether certain state laws impermissibly applied or were connected to self-funded plans. The ruling in favor of Rutledge opens the door that certain state legislation may one day apply to self-funded plans.
Specialty drug benefit carve outs are not in best interests of employees, employers
This piece is not about Rutledge but about the small third-party administrators that are convincing self-funded businesses to let them “carve out” specialty drug benefits from the larger administrator of the plan by promising huge savings in the employer’s specialty drug spending. Two such companies that have come to the attention of the Coalition of State Rheumatology Organizations are Vivio Health and Archimedes. CSRO has received numerous complaints from rheumatologists regarding interference from these two entities with their clinical decision making and disregard for standard of care.
Vivio’s website reveals a disturbing approach to cost reduction. The website states that Vivio profiles physicians through ProPublica and Open Payments to determine if they are prescribing for the right reason and not for self-interest. This serves as an attempt to set up mistrust of the physician by the employer. Vivio’s website also states that the Food and Drug Administration has declining standards for approval of drugs, and consequently many approved drugs should be considered “experimental.” They say that business owners should not have to pay for “experimental” treatments. Through conversation with Vivio, it appears they believe that oncologists could be replaced by a primary care physician with the right algorithm.
Many of Vivio’s egregious behaviors are enumerated in our letter to Vivio: outrageous nonmedical switches, mandatory biologic tapering, and site of care changes. In all of the complaints that we received, Vivio attempted to switch patients to the same infusible medication, Renflexis, and also mandated white bagging, which means the payer has a specialty pharmacy ship a patient’s medication directly to the physician’s office for administration. This switch was made regardless of the mechanism of action or route of administration of the drug that had stabilized the patient. Peer-to-peer reviews with a retired radiologist led to routinely denied appeals and would even force the patient to a different site of care if the rheumatologist refused the new treatment or the mandated white bagging.
Our letter resulted in two conversations with Vivio. Vivio insisted that it was using American College of Rheumatology guidelines and comparisons between drug studies to make these decisions. The company stated that patients can be switched to any drug that has the same ACR 20, 50, and 70 response criteria outcomes as the drug that they are presently taking, even though these sorts of comparisons of results across completely different studies are invalid for a number of reasons, including because they do not have the same patient populations, protocols, and inclusion/exclusion criteria. These are dangerous policies and thus far we have not been able to find any rheumatologists who have gone along with these demands.
Companies such as Vivio are spreading, and employers are unaware that their policies are only paying lip service to “individualized care” while maintaining an approach to patient care that is focused only on cost cutting. Indeed, Archimedes represents one such metastasis. Their practices are similar to those of Vivio and of which CSRO has received complaints. Archimedes has similarly attempted to mandate white-bagging for the enrollees it manages and switch stable patients for nonmedical reasons to an entirely different molecule and mechanism of action.
Business owners do not understand the harm that these policies can cause their employees. This harm increases downstream medical spending as a result of loss of control of disease activity.
This is a call to action for all advocates and advocacy groups to get in the room with employer/business groups and explain how these third-party administrators, carving out specialty drug benefits, can ultimately cause physical harm to employees and increase monetary cost to the employer in the long run. Rheumatology as a specialty needs to educate employers and work out ways to save money for them and, at the same time, maintain excellence in care for their employees. CSRO has a letter it used successfully with the human resources department of Edward Jones to effect a change in its policies on this matter, which you are welcome to use to craft your own to businesses.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is President of the CSRO, past chair of the Alliance for Safe Biologic Medicines, and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
There is a dangerous trend in our country in which employers, seeking to reduce health plan costs they pay, enter into agreements with small third-party administrators that “carve out” specialty drug benefits from their self-funded health insurance plan. What employers are not told is that these spending reductions are accomplished by risking the health of their employees. It is the self-funded businesses that are being preyed upon by these administrators because there is a lot of money to be made by carving out the specialty drugs in their self-funded health plan.
Let’s start with a little primer on “fully insured” versus “self-funded” health plans. As a small business owner, I understand the need to make sure that expenses don’t outpace revenue if I want to keep my doors open. One of the largest expenses for any business is health insurance. My private rheumatology practice uses a fully insured health plan. In a fully insured plan, the insurer is the party taking the financial risk. We pay the premiums, and the insurance company pays the bills after the deductible is met. It may cost more in premiums than a self-funded plan, but if an employee has an accident or severe illness, our practice is not responsible for the cost of care.
On the other hand, large and small businesses that are self-funded cover the health costs of their employees themselves. These businesses will hire a third-party administrator to pay the bills out of an account that is supplied with money from the business owner. Looking at the insurance card of your patient is one way to tell if they are covered by a fully insured or self-funded plan. If the insurance card says the plan is “administered by” the insurer or “administrative services only,” it is most likely a self-funded plan. If their insurance card states “underwritten by” the insurer on the card then it is likely a fully insured plan. This becomes important because self-funded plans are not subject to the jurisdiction of state laws such as utilization management reform. These state laws are preempted from applying to self-funded plans by the Employee Retirement Income Security Act of 1974. The Rutledge v. Pharmaceutical Care Management Association Supreme Court case took up the question of whether certain state laws impermissibly applied or were connected to self-funded plans. The ruling in favor of Rutledge opens the door that certain state legislation may one day apply to self-funded plans.
Specialty drug benefit carve outs are not in best interests of employees, employers
This piece is not about Rutledge but about the small third-party administrators that are convincing self-funded businesses to let them “carve out” specialty drug benefits from the larger administrator of the plan by promising huge savings in the employer’s specialty drug spending. Two such companies that have come to the attention of the Coalition of State Rheumatology Organizations are Vivio Health and Archimedes. CSRO has received numerous complaints from rheumatologists regarding interference from these two entities with their clinical decision making and disregard for standard of care.
Vivio’s website reveals a disturbing approach to cost reduction. The website states that Vivio profiles physicians through ProPublica and Open Payments to determine if they are prescribing for the right reason and not for self-interest. This serves as an attempt to set up mistrust of the physician by the employer. Vivio’s website also states that the Food and Drug Administration has declining standards for approval of drugs, and consequently many approved drugs should be considered “experimental.” They say that business owners should not have to pay for “experimental” treatments. Through conversation with Vivio, it appears they believe that oncologists could be replaced by a primary care physician with the right algorithm.
Many of Vivio’s egregious behaviors are enumerated in our letter to Vivio: outrageous nonmedical switches, mandatory biologic tapering, and site of care changes. In all of the complaints that we received, Vivio attempted to switch patients to the same infusible medication, Renflexis, and also mandated white bagging, which means the payer has a specialty pharmacy ship a patient’s medication directly to the physician’s office for administration. This switch was made regardless of the mechanism of action or route of administration of the drug that had stabilized the patient. Peer-to-peer reviews with a retired radiologist led to routinely denied appeals and would even force the patient to a different site of care if the rheumatologist refused the new treatment or the mandated white bagging.
Our letter resulted in two conversations with Vivio. Vivio insisted that it was using American College of Rheumatology guidelines and comparisons between drug studies to make these decisions. The company stated that patients can be switched to any drug that has the same ACR 20, 50, and 70 response criteria outcomes as the drug that they are presently taking, even though these sorts of comparisons of results across completely different studies are invalid for a number of reasons, including because they do not have the same patient populations, protocols, and inclusion/exclusion criteria. These are dangerous policies and thus far we have not been able to find any rheumatologists who have gone along with these demands.
Companies such as Vivio are spreading, and employers are unaware that their policies are only paying lip service to “individualized care” while maintaining an approach to patient care that is focused only on cost cutting. Indeed, Archimedes represents one such metastasis. Their practices are similar to those of Vivio and of which CSRO has received complaints. Archimedes has similarly attempted to mandate white-bagging for the enrollees it manages and switch stable patients for nonmedical reasons to an entirely different molecule and mechanism of action.
Business owners do not understand the harm that these policies can cause their employees. This harm increases downstream medical spending as a result of loss of control of disease activity.
This is a call to action for all advocates and advocacy groups to get in the room with employer/business groups and explain how these third-party administrators, carving out specialty drug benefits, can ultimately cause physical harm to employees and increase monetary cost to the employer in the long run. Rheumatology as a specialty needs to educate employers and work out ways to save money for them and, at the same time, maintain excellence in care for their employees. CSRO has a letter it used successfully with the human resources department of Edward Jones to effect a change in its policies on this matter, which you are welcome to use to craft your own to businesses.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is President of the CSRO, past chair of the Alliance for Safe Biologic Medicines, and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Targeting IL-23 could still be important for axial spondyloarthritis treatment
Interleukin (IL)–23 inhibition may still have a role to play in the treatment of patients with axial spondyloarthritis (SpA), suggests research presented at the 12th International Congress on Spondyloarthritides.
There is a strong rationale for using IL-23 inhibitors in patients with axial SpA, and the IL-23/IL-17 axis has been proposed as a critical player in the pathophysiology of the disease. But around 2018 it became clear from randomized, controlled trials that IL-23 inhibition was ineffective at improving key clinical outcomes, at least in patients with axial disease.
Although the overall results of a systematic review and meta-analysis that was presented at the meeting corroborated the negative results seen with IL-23–inhibiting agents in clinical trials, there were some data showing benefits of the IL-23 inhibitor risankizumab on secondary outcomes in one trial.
To look at the available evidence, Louise Vanhoutte, a 2nd-year internal medicine student at University Hospitals Leuven (Belgium) worked under the guidance of Rik Lories, MD, PhD, head of the division of rheumatology at University Hospitals Leuven. Together they searched known databases for randomized, controlled trials investigating the use of IL-23 and IL-17 inhibitors for the treatment of adults with axial SpA or psoriatic arthritis. Studies could be either phase 2 or phase 3, but had to have included a placebo and used the ASAS40 (40% Improvement in Assessment of SpondyloArthritis International Society Response criteria), ASAS20, Bath Ankylosing Spondylitis Disease Activity Index, or SPARCC (Spondyloarthritis Research Consortium of Canada) index score to assess outcomes.
The systematic review whittled the number of clinical trials in the meta-analysis to 12, which concerned the use of ustekinumab, an IL-12/23 inhibitor, and risankizumab, along with two IL-17 inhibitors, ixekizumab and secukinumab. Data for the IL-23 inhibitors guselkumab and tildrakizumab were not available.
“To no surprise, Forest plots showed that there was a lack of efficacy for IL-23 agents in the treatment of axial spondyloarthritis and a superior efficacy for IL-17 inhibitors in the treatment of axial spondyloarthritis,” Ms. Vanhoutte reported.
The respective odds ratios for IL-23 and IL-17 inhibitors in getting patients to meet ASAS40 response criteria in comparison to baseline were 1.51 (95% confidence interval, 0.98-2.31) and 2.54 (95% CI, 2.02-3.19).
“Does this mean it is a dead-end street for all IL-23 inhibition?” she asked. Not necessarily. In the meta-analysis, not only did risankizumab lower the Ankylosing Spondylitis Disease Activity Score based on C-reactive protein (ASDAS-CRP) by a mean difference (MD) of –0.30 (95% CI, –0.41 to –0.19) from baseline values, but it also led to statistically significant reductions in SPARCC index score for the spine (MD, –3.10; 95% CI, –4.50 to –1.70) and high-sensitivity CRP (MD, –2.10; 95% CI, –2.56 to –1.64). The risankizumab findings might suggest there are potential disease-modifying properties for specifically targeting IL-23p19. There could also be a window of opportunity to use IL-23 inhibitors earlier.
“These are only results from one randomized, controlled trial in a small sample size where outcomes were reported as medians and interquartile ranges, so they had to be converted to means and standard deviations to have an odds ratio in the end,” she explained.
“Also, these were results from a radiographic axial spondyloarthritis population and not a nonradiographic axial spondyloarthritis population,” she added.
While that might limit the interpretation of the findings, “what we see here is both reduction in inflammation and reduction in structural disease progression as [measured] by SPARCC,” Ms. Vanhoutte said.
“Since IL-23 is an upstream molecule from IL-17 it’s probable that IL-23 is present in the prephase of the disease, in a prephase inflammation state,” she hypothesized. “This is especially interesting because there are very few randomized, controlled trials that examine therapeutic agents in nonradiographic axial spondyloarthritis,” she observed. Looking at IL-23 in radiographic, or established, disease therefore may not be as useful.
“I’m thinking you’re making actually a very important point for us,” commented Robert Landewé, MD, PhD, of Amsterdam University Medical Center.
“We are discussing whether or not IL-23 is important in inhibiting the disease activity of patients with axial spondyloarthritis, and we are surprised that it is not shown in RCTs.
“Why is it completely ineffective in axial spondyloarthritis? You show us that that is probably not the case,” Dr. Landewé suggested.
“What you make very clear here is that indeed there is some efficacy, and from a pathophysiological way of thinking it might be slightly different as compared with what most clinicians nowadays think.”
The study had no specific funding, and no disclosures were reported.
Interleukin (IL)–23 inhibition may still have a role to play in the treatment of patients with axial spondyloarthritis (SpA), suggests research presented at the 12th International Congress on Spondyloarthritides.
There is a strong rationale for using IL-23 inhibitors in patients with axial SpA, and the IL-23/IL-17 axis has been proposed as a critical player in the pathophysiology of the disease. But around 2018 it became clear from randomized, controlled trials that IL-23 inhibition was ineffective at improving key clinical outcomes, at least in patients with axial disease.
Although the overall results of a systematic review and meta-analysis that was presented at the meeting corroborated the negative results seen with IL-23–inhibiting agents in clinical trials, there were some data showing benefits of the IL-23 inhibitor risankizumab on secondary outcomes in one trial.
To look at the available evidence, Louise Vanhoutte, a 2nd-year internal medicine student at University Hospitals Leuven (Belgium) worked under the guidance of Rik Lories, MD, PhD, head of the division of rheumatology at University Hospitals Leuven. Together they searched known databases for randomized, controlled trials investigating the use of IL-23 and IL-17 inhibitors for the treatment of adults with axial SpA or psoriatic arthritis. Studies could be either phase 2 or phase 3, but had to have included a placebo and used the ASAS40 (40% Improvement in Assessment of SpondyloArthritis International Society Response criteria), ASAS20, Bath Ankylosing Spondylitis Disease Activity Index, or SPARCC (Spondyloarthritis Research Consortium of Canada) index score to assess outcomes.
The systematic review whittled the number of clinical trials in the meta-analysis to 12, which concerned the use of ustekinumab, an IL-12/23 inhibitor, and risankizumab, along with two IL-17 inhibitors, ixekizumab and secukinumab. Data for the IL-23 inhibitors guselkumab and tildrakizumab were not available.
“To no surprise, Forest plots showed that there was a lack of efficacy for IL-23 agents in the treatment of axial spondyloarthritis and a superior efficacy for IL-17 inhibitors in the treatment of axial spondyloarthritis,” Ms. Vanhoutte reported.
The respective odds ratios for IL-23 and IL-17 inhibitors in getting patients to meet ASAS40 response criteria in comparison to baseline were 1.51 (95% confidence interval, 0.98-2.31) and 2.54 (95% CI, 2.02-3.19).
“Does this mean it is a dead-end street for all IL-23 inhibition?” she asked. Not necessarily. In the meta-analysis, not only did risankizumab lower the Ankylosing Spondylitis Disease Activity Score based on C-reactive protein (ASDAS-CRP) by a mean difference (MD) of –0.30 (95% CI, –0.41 to –0.19) from baseline values, but it also led to statistically significant reductions in SPARCC index score for the spine (MD, –3.10; 95% CI, –4.50 to –1.70) and high-sensitivity CRP (MD, –2.10; 95% CI, –2.56 to –1.64). The risankizumab findings might suggest there are potential disease-modifying properties for specifically targeting IL-23p19. There could also be a window of opportunity to use IL-23 inhibitors earlier.
“These are only results from one randomized, controlled trial in a small sample size where outcomes were reported as medians and interquartile ranges, so they had to be converted to means and standard deviations to have an odds ratio in the end,” she explained.
“Also, these were results from a radiographic axial spondyloarthritis population and not a nonradiographic axial spondyloarthritis population,” she added.
While that might limit the interpretation of the findings, “what we see here is both reduction in inflammation and reduction in structural disease progression as [measured] by SPARCC,” Ms. Vanhoutte said.
“Since IL-23 is an upstream molecule from IL-17 it’s probable that IL-23 is present in the prephase of the disease, in a prephase inflammation state,” she hypothesized. “This is especially interesting because there are very few randomized, controlled trials that examine therapeutic agents in nonradiographic axial spondyloarthritis,” she observed. Looking at IL-23 in radiographic, or established, disease therefore may not be as useful.
“I’m thinking you’re making actually a very important point for us,” commented Robert Landewé, MD, PhD, of Amsterdam University Medical Center.
“We are discussing whether or not IL-23 is important in inhibiting the disease activity of patients with axial spondyloarthritis, and we are surprised that it is not shown in RCTs.
“Why is it completely ineffective in axial spondyloarthritis? You show us that that is probably not the case,” Dr. Landewé suggested.
“What you make very clear here is that indeed there is some efficacy, and from a pathophysiological way of thinking it might be slightly different as compared with what most clinicians nowadays think.”
The study had no specific funding, and no disclosures were reported.
Interleukin (IL)–23 inhibition may still have a role to play in the treatment of patients with axial spondyloarthritis (SpA), suggests research presented at the 12th International Congress on Spondyloarthritides.
There is a strong rationale for using IL-23 inhibitors in patients with axial SpA, and the IL-23/IL-17 axis has been proposed as a critical player in the pathophysiology of the disease. But around 2018 it became clear from randomized, controlled trials that IL-23 inhibition was ineffective at improving key clinical outcomes, at least in patients with axial disease.
Although the overall results of a systematic review and meta-analysis that was presented at the meeting corroborated the negative results seen with IL-23–inhibiting agents in clinical trials, there were some data showing benefits of the IL-23 inhibitor risankizumab on secondary outcomes in one trial.
To look at the available evidence, Louise Vanhoutte, a 2nd-year internal medicine student at University Hospitals Leuven (Belgium) worked under the guidance of Rik Lories, MD, PhD, head of the division of rheumatology at University Hospitals Leuven. Together they searched known databases for randomized, controlled trials investigating the use of IL-23 and IL-17 inhibitors for the treatment of adults with axial SpA or psoriatic arthritis. Studies could be either phase 2 or phase 3, but had to have included a placebo and used the ASAS40 (40% Improvement in Assessment of SpondyloArthritis International Society Response criteria), ASAS20, Bath Ankylosing Spondylitis Disease Activity Index, or SPARCC (Spondyloarthritis Research Consortium of Canada) index score to assess outcomes.
The systematic review whittled the number of clinical trials in the meta-analysis to 12, which concerned the use of ustekinumab, an IL-12/23 inhibitor, and risankizumab, along with two IL-17 inhibitors, ixekizumab and secukinumab. Data for the IL-23 inhibitors guselkumab and tildrakizumab were not available.
“To no surprise, Forest plots showed that there was a lack of efficacy for IL-23 agents in the treatment of axial spondyloarthritis and a superior efficacy for IL-17 inhibitors in the treatment of axial spondyloarthritis,” Ms. Vanhoutte reported.
The respective odds ratios for IL-23 and IL-17 inhibitors in getting patients to meet ASAS40 response criteria in comparison to baseline were 1.51 (95% confidence interval, 0.98-2.31) and 2.54 (95% CI, 2.02-3.19).
“Does this mean it is a dead-end street for all IL-23 inhibition?” she asked. Not necessarily. In the meta-analysis, not only did risankizumab lower the Ankylosing Spondylitis Disease Activity Score based on C-reactive protein (ASDAS-CRP) by a mean difference (MD) of –0.30 (95% CI, –0.41 to –0.19) from baseline values, but it also led to statistically significant reductions in SPARCC index score for the spine (MD, –3.10; 95% CI, –4.50 to –1.70) and high-sensitivity CRP (MD, –2.10; 95% CI, –2.56 to –1.64). The risankizumab findings might suggest there are potential disease-modifying properties for specifically targeting IL-23p19. There could also be a window of opportunity to use IL-23 inhibitors earlier.
“These are only results from one randomized, controlled trial in a small sample size where outcomes were reported as medians and interquartile ranges, so they had to be converted to means and standard deviations to have an odds ratio in the end,” she explained.
“Also, these were results from a radiographic axial spondyloarthritis population and not a nonradiographic axial spondyloarthritis population,” she added.
While that might limit the interpretation of the findings, “what we see here is both reduction in inflammation and reduction in structural disease progression as [measured] by SPARCC,” Ms. Vanhoutte said.
“Since IL-23 is an upstream molecule from IL-17 it’s probable that IL-23 is present in the prephase of the disease, in a prephase inflammation state,” she hypothesized. “This is especially interesting because there are very few randomized, controlled trials that examine therapeutic agents in nonradiographic axial spondyloarthritis,” she observed. Looking at IL-23 in radiographic, or established, disease therefore may not be as useful.
“I’m thinking you’re making actually a very important point for us,” commented Robert Landewé, MD, PhD, of Amsterdam University Medical Center.
“We are discussing whether or not IL-23 is important in inhibiting the disease activity of patients with axial spondyloarthritis, and we are surprised that it is not shown in RCTs.
“Why is it completely ineffective in axial spondyloarthritis? You show us that that is probably not the case,” Dr. Landewé suggested.
“What you make very clear here is that indeed there is some efficacy, and from a pathophysiological way of thinking it might be slightly different as compared with what most clinicians nowadays think.”
The study had no specific funding, and no disclosures were reported.
FROM THE 2021 SPA CONGRESS
European agency recommends two new adalimumab biosimilars
The European Medicines Agency’s Committee for Medicinal Products for Human Use recommended marketing authorization this week for two new adalimumab biosimilars, Hukyndra and Libmyris.
The biosimilars, both developed by STADA Arzneimittel AG, will be available as a 40-mg solution for injection in a pre-filled syringe and pre-filled pen and 80-mg solution for injection in a pre-filled syringe. Both biosimilars will have 15 indications:
- rheumatoid arthritis
- polyarticular juvenile idiopathic arthritis
- enthesitis-related arthritis
- ankylosing spondylitis
- axial spondyloarthritis without radiographic evidence of ankylosing spondylitis
- psoriatic arthritis
- chronic plaque psoriasis (adults and children)
- hidradenitis suppurativa
- Crohn’s disease (adults and children)
- ulcerative colitis (adults and children)
- uveitis (adults and children)
Data show that both Hukyndra and Libmyris are highly similar to the reference product Humira (adalimumab), a monoclonal antibody to tumor necrosis factor alpha, and have comparable quality, safety, and efficacy.
A version of this article first appeared on Medscape.com.
The European Medicines Agency’s Committee for Medicinal Products for Human Use recommended marketing authorization this week for two new adalimumab biosimilars, Hukyndra and Libmyris.
The biosimilars, both developed by STADA Arzneimittel AG, will be available as a 40-mg solution for injection in a pre-filled syringe and pre-filled pen and 80-mg solution for injection in a pre-filled syringe. Both biosimilars will have 15 indications:
- rheumatoid arthritis
- polyarticular juvenile idiopathic arthritis
- enthesitis-related arthritis
- ankylosing spondylitis
- axial spondyloarthritis without radiographic evidence of ankylosing spondylitis
- psoriatic arthritis
- chronic plaque psoriasis (adults and children)
- hidradenitis suppurativa
- Crohn’s disease (adults and children)
- ulcerative colitis (adults and children)
- uveitis (adults and children)
Data show that both Hukyndra and Libmyris are highly similar to the reference product Humira (adalimumab), a monoclonal antibody to tumor necrosis factor alpha, and have comparable quality, safety, and efficacy.
A version of this article first appeared on Medscape.com.
The European Medicines Agency’s Committee for Medicinal Products for Human Use recommended marketing authorization this week for two new adalimumab biosimilars, Hukyndra and Libmyris.
The biosimilars, both developed by STADA Arzneimittel AG, will be available as a 40-mg solution for injection in a pre-filled syringe and pre-filled pen and 80-mg solution for injection in a pre-filled syringe. Both biosimilars will have 15 indications:
- rheumatoid arthritis
- polyarticular juvenile idiopathic arthritis
- enthesitis-related arthritis
- ankylosing spondylitis
- axial spondyloarthritis without radiographic evidence of ankylosing spondylitis
- psoriatic arthritis
- chronic plaque psoriasis (adults and children)
- hidradenitis suppurativa
- Crohn’s disease (adults and children)
- ulcerative colitis (adults and children)
- uveitis (adults and children)
Data show that both Hukyndra and Libmyris are highly similar to the reference product Humira (adalimumab), a monoclonal antibody to tumor necrosis factor alpha, and have comparable quality, safety, and efficacy.
A version of this article first appeared on Medscape.com.
FDA panel backs Pfizer's COVID booster for 65 and older, those at high risk
An expert panel that advises the Food and Drug Administration on its regulatory decisions voted Sept. 17 against recommending third doses of Pfizer’s COVID-19 vaccine for younger Americans.
But they didn’t kill the idea of booster shots completely.
In a dramatic, last-minute pivot, the 18 members of the FDA’s Vaccines and Related Biological Products Advisory Committee unanimously voted to recommend the FDA make boosters available for seniors and others at high risk of severe outcomes from COVID-19, including health care workers.
The 16-2 vote was a rebuttal to Pfizer’s initial request. The company had asked the FDA to allow it to offer third doses to all Americans over the age of 16 at least six months after their second shot.
The company requested an amendment to the full approval the FDA granted in August. That is the typical way boosters are authorized in the U.S., but it requires a higher bar of evidence and more regulatory scrutiny than the agency had been able to give since Pfizer filed for the change just days after its vaccine was granted full approval.
The committee’s actions were also a rebuff to the Biden administration, which announced before the FDA approved them that boosters would be rolled out to the general public Sept. 20. The announcement triggered the resignations of two of the agency’s top vaccine reviewers, who both participated in the Sept. 17 meeting.
After initially voting against Pfizer’s request to amend its license, the committee then worked on the fly with FDA officials to craft a strategy that would allow third doses to be offered under an emergency use authorization (EUA).
An EUA requires a lower standard of evidence and is more specific. It will restrict third doses to a more defined population than a change to the license would. It will also require Pfizer to continue to monitor the safety of third doses as they begin to be administered.
“This should demonstrate to the public that the members of this committee are independent of the FDA and that we do, in fact, bring our voices to the table when we are asked to serve on this committee,” said Archana Chattergee, MD, a pediatric infectious disease specialist who is dean of the Chicago Medical School at Rosalind Franklin University in Illinois.
The FDA doesn’t have to follow the committee’s recommendation, but almost certainly will, though regulators said they may still make some changes.
“We are not bound at FDA by your vote, we can tweak this,” said Peter Marks, MD, director of the Center for Biologics Evaluation and Research at the FDA. Dr. Marks participated in the meeting and helped to draft the revised proposal.
If the FDA issues the recommended EUA, a council of independent advisors to the CDC will make specific recommendations about how the third doses should be given. After the CDC director weighs in, boosters will begin rolling out to the public.
Moderna submitted data to the FDA on Sept. 1 in support of adding a booster dose to its regimen. The agency has not yet scheduled a public review of that data.
The Biden administration is prepared to administer shots as soon as they get the green light, Surgeon General Vivek Murthy, MD, said at a White House briefing earlier Sept. 17.
"This process is consistent with what we outlined in August where our goals were to stay ahead of the virus," Dr. Murthy said. "Our goal then and now is to protect the health and well-being of the public. As soon as the FDA and CDC complete their evaluations, we will be ready to move forward accordingly."
He added, "We've used this time since our August announcement to communicate and coordinate with pharmacy partners, nursing homes, states, and localities."
White House COVID-19 Response Coordinator Jeff Zients said vaccine supply is "in good shape for all Americans to get boosters as recommended."
Taking cues from Israel
In considering Pfizer’s original request, the committee overwhelmingly felt that they didn’t have enough information to say that the benefits of an additional dose of vaccine in 16- and 17-year-olds would outweigh its risk. Teens have the highest risk of rare heart inflammation after vaccination, a side effect known as myocarditis. It is not known how the vaccines are causing these cases of heart swelling. Most who have been diagnosed with the condition have recovered, though some have needed hospital care.
Pfizer didn’t include 16- and 17-year-olds in its studies of boosters, which included about 300 people between the ages of 18 and 55. The company acknowledged that gap in its data but pointed to FDA guidance that said evidence from adults could be extrapolated to teens.
“We don’t know that much about risks,” said committee member Eric Rubin, MD, who is editor-in-chief of the New England Journal of Medicine.
Much of the data on the potential benefits and harms of third Pfizer doses comes from Israel, which first began rolling out boosters to older adults in July.
In a highly anticipated presentation, Sharon Alroy-Preis, Israel’s director of public health services, joined the meeting to describe Israel’s experience with boosters.
Israel began to see a third surge of COVID-19 cases in December.
“This was after having two waves and two lockdowns,” Ms. Alroy-Preis said. By the third surge, she said, Israelis were tired.
“We decided on a lockdown, but the compliance of the public wasn’t as it was in the previous two waves,” she said.
Then the vaccine arrived. Israel started vaccinations as soon as the FDA approved it, and they quickly vaccinated a high percentage of their population, about 3 months faster than the rest of the world.
All vaccinations are reported and tracked by the Ministry of Health, so the country is able to keep close tabs on how well the shots are working.
As vaccines rolled out, cases fell dramatically. The pandemic seemed to be behind them. Delta arrived in March. By June, their cases were doubling every 10 days, despite about 80% of their most vulnerable adults being fully vaccinated, she said.
Most concerning was that about 60% of severe cases were breakthrough cases in fully vaccinated individuals.
“We had to stop and figure out, was this a Delta issue,” she said. “Or was this a waning immunity issue.”
“We had some clue that it might not be the Delta variant, at least not alone,” she said.
People who had originally been first in line for the vaccines, seniors and health care workers, were having the highest rates of breakthrough infections. People further away from their second dose were more likely to get a breakthrough infection.
Ms. Alroy-Preis said that if they had not started booster doses in July, their hospitals would have been overwhelmed. They had projected that they would have 2,000 cases in the hospital each day.
Boosters have helped to flatten the curve, though they are still seeing a significant number of infections.
Data from Israel presented at the meeting show boosters are largely safe and effective at reducing severe outcomes in seniors. Israeli experience also showed that third doses, which generate very high levels of neutralizing antibodies—the first and fastest line of the body’s immune defense - -may also slow transmission of the virus.
Key differences in the U.S.
The benefit of slowing down the explosive spread of a highly contagious virus was tantalizing, but many members noted that circumstances in Israel are very different than in the United States. Israel went into its current Delta surge already having high levels of vaccination in its population. They also relied on the Pfizer vaccine almost exclusively for their campaign.
The United States used a different mix of vaccines – Pfizer, Moderna, and Johnson & Johnson -- and doesn’t have the same high level of vaccination coverage of its population.
In the United States, transmission is mainly being driven by unvaccinated people, Dr. Rubin noted.
“That really means the primary benefit is going to be in reducing disease,” he said, “And we know the people who are going to benefit from that … and those are the kinds of people the FDA has already approved a third dose for,” he said, referring to those with underlying health conditions.
But Israel only began vaccinating younger people a few weeks ago. Most are still within a window where rare risks like myocarditis could appear, Rubin noted.
He and other members of the committee said they wished they had more information about the safety of third doses in younger adults.
“We don’t have that right now, and I don’t think I would be comfortable giving it to a 16-year-old,” he said.
At the same time, the primary benefit for third doses would be in preventing severe disease, and overall, data from the United States and other countries show that two doses of the vaccines remain highly effective at preventing hospitalization and death.
Asked why Israel began to see more severe cases in fully vaccinated people, the CDC’s Sara Oliver, MD, a disease detective with the CDC, said it was probably due to a mix of factors including the fact that Israel defines severe cases a little differently.
In the United States, a severe case is generally a person who has to be hospitalized or who has died from the infection. In Israel, a person with a severe case is someone who has an elevated respiratory rate and someone who has a blood oxygen level less than 94%. In the United States, that kind of patient wouldn’t necessarily be hospitalized.
In the end, one of the two committee members who wanted full approval for Pfizer’s third doses said he was satisfied with the outcome.
Mark Sawyer, MD, a professor of pediatrics and infectious disease at the University of California at San Diego, said he voted yes on the first question because he thought full approval was the best way to give doctors the flexibility to prescribe the shots to vulnerable individuals.
“I’m really glad we authorized a vaccine for a third dose, and I plan to go out and get my vaccine this afternoon,” Dr. Sawyer said, noting that he was at high risk as a health care provider.
This article was updated 9/19/21.
A version of this article first appeared on Medscape.com.
An expert panel that advises the Food and Drug Administration on its regulatory decisions voted Sept. 17 against recommending third doses of Pfizer’s COVID-19 vaccine for younger Americans.
But they didn’t kill the idea of booster shots completely.
In a dramatic, last-minute pivot, the 18 members of the FDA’s Vaccines and Related Biological Products Advisory Committee unanimously voted to recommend the FDA make boosters available for seniors and others at high risk of severe outcomes from COVID-19, including health care workers.
The 16-2 vote was a rebuttal to Pfizer’s initial request. The company had asked the FDA to allow it to offer third doses to all Americans over the age of 16 at least six months after their second shot.
The company requested an amendment to the full approval the FDA granted in August. That is the typical way boosters are authorized in the U.S., but it requires a higher bar of evidence and more regulatory scrutiny than the agency had been able to give since Pfizer filed for the change just days after its vaccine was granted full approval.
The committee’s actions were also a rebuff to the Biden administration, which announced before the FDA approved them that boosters would be rolled out to the general public Sept. 20. The announcement triggered the resignations of two of the agency’s top vaccine reviewers, who both participated in the Sept. 17 meeting.
After initially voting against Pfizer’s request to amend its license, the committee then worked on the fly with FDA officials to craft a strategy that would allow third doses to be offered under an emergency use authorization (EUA).
An EUA requires a lower standard of evidence and is more specific. It will restrict third doses to a more defined population than a change to the license would. It will also require Pfizer to continue to monitor the safety of third doses as they begin to be administered.
“This should demonstrate to the public that the members of this committee are independent of the FDA and that we do, in fact, bring our voices to the table when we are asked to serve on this committee,” said Archana Chattergee, MD, a pediatric infectious disease specialist who is dean of the Chicago Medical School at Rosalind Franklin University in Illinois.
The FDA doesn’t have to follow the committee’s recommendation, but almost certainly will, though regulators said they may still make some changes.
“We are not bound at FDA by your vote, we can tweak this,” said Peter Marks, MD, director of the Center for Biologics Evaluation and Research at the FDA. Dr. Marks participated in the meeting and helped to draft the revised proposal.
If the FDA issues the recommended EUA, a council of independent advisors to the CDC will make specific recommendations about how the third doses should be given. After the CDC director weighs in, boosters will begin rolling out to the public.
Moderna submitted data to the FDA on Sept. 1 in support of adding a booster dose to its regimen. The agency has not yet scheduled a public review of that data.
The Biden administration is prepared to administer shots as soon as they get the green light, Surgeon General Vivek Murthy, MD, said at a White House briefing earlier Sept. 17.
"This process is consistent with what we outlined in August where our goals were to stay ahead of the virus," Dr. Murthy said. "Our goal then and now is to protect the health and well-being of the public. As soon as the FDA and CDC complete their evaluations, we will be ready to move forward accordingly."
He added, "We've used this time since our August announcement to communicate and coordinate with pharmacy partners, nursing homes, states, and localities."
White House COVID-19 Response Coordinator Jeff Zients said vaccine supply is "in good shape for all Americans to get boosters as recommended."
Taking cues from Israel
In considering Pfizer’s original request, the committee overwhelmingly felt that they didn’t have enough information to say that the benefits of an additional dose of vaccine in 16- and 17-year-olds would outweigh its risk. Teens have the highest risk of rare heart inflammation after vaccination, a side effect known as myocarditis. It is not known how the vaccines are causing these cases of heart swelling. Most who have been diagnosed with the condition have recovered, though some have needed hospital care.
Pfizer didn’t include 16- and 17-year-olds in its studies of boosters, which included about 300 people between the ages of 18 and 55. The company acknowledged that gap in its data but pointed to FDA guidance that said evidence from adults could be extrapolated to teens.
“We don’t know that much about risks,” said committee member Eric Rubin, MD, who is editor-in-chief of the New England Journal of Medicine.
Much of the data on the potential benefits and harms of third Pfizer doses comes from Israel, which first began rolling out boosters to older adults in July.
In a highly anticipated presentation, Sharon Alroy-Preis, Israel’s director of public health services, joined the meeting to describe Israel’s experience with boosters.
Israel began to see a third surge of COVID-19 cases in December.
“This was after having two waves and two lockdowns,” Ms. Alroy-Preis said. By the third surge, she said, Israelis were tired.
“We decided on a lockdown, but the compliance of the public wasn’t as it was in the previous two waves,” she said.
Then the vaccine arrived. Israel started vaccinations as soon as the FDA approved it, and they quickly vaccinated a high percentage of their population, about 3 months faster than the rest of the world.
All vaccinations are reported and tracked by the Ministry of Health, so the country is able to keep close tabs on how well the shots are working.
As vaccines rolled out, cases fell dramatically. The pandemic seemed to be behind them. Delta arrived in March. By June, their cases were doubling every 10 days, despite about 80% of their most vulnerable adults being fully vaccinated, she said.
Most concerning was that about 60% of severe cases were breakthrough cases in fully vaccinated individuals.
“We had to stop and figure out, was this a Delta issue,” she said. “Or was this a waning immunity issue.”
“We had some clue that it might not be the Delta variant, at least not alone,” she said.
People who had originally been first in line for the vaccines, seniors and health care workers, were having the highest rates of breakthrough infections. People further away from their second dose were more likely to get a breakthrough infection.
Ms. Alroy-Preis said that if they had not started booster doses in July, their hospitals would have been overwhelmed. They had projected that they would have 2,000 cases in the hospital each day.
Boosters have helped to flatten the curve, though they are still seeing a significant number of infections.
Data from Israel presented at the meeting show boosters are largely safe and effective at reducing severe outcomes in seniors. Israeli experience also showed that third doses, which generate very high levels of neutralizing antibodies—the first and fastest line of the body’s immune defense - -may also slow transmission of the virus.
Key differences in the U.S.
The benefit of slowing down the explosive spread of a highly contagious virus was tantalizing, but many members noted that circumstances in Israel are very different than in the United States. Israel went into its current Delta surge already having high levels of vaccination in its population. They also relied on the Pfizer vaccine almost exclusively for their campaign.
The United States used a different mix of vaccines – Pfizer, Moderna, and Johnson & Johnson -- and doesn’t have the same high level of vaccination coverage of its population.
In the United States, transmission is mainly being driven by unvaccinated people, Dr. Rubin noted.
“That really means the primary benefit is going to be in reducing disease,” he said, “And we know the people who are going to benefit from that … and those are the kinds of people the FDA has already approved a third dose for,” he said, referring to those with underlying health conditions.
But Israel only began vaccinating younger people a few weeks ago. Most are still within a window where rare risks like myocarditis could appear, Rubin noted.
He and other members of the committee said they wished they had more information about the safety of third doses in younger adults.
“We don’t have that right now, and I don’t think I would be comfortable giving it to a 16-year-old,” he said.
At the same time, the primary benefit for third doses would be in preventing severe disease, and overall, data from the United States and other countries show that two doses of the vaccines remain highly effective at preventing hospitalization and death.
Asked why Israel began to see more severe cases in fully vaccinated people, the CDC’s Sara Oliver, MD, a disease detective with the CDC, said it was probably due to a mix of factors including the fact that Israel defines severe cases a little differently.
In the United States, a severe case is generally a person who has to be hospitalized or who has died from the infection. In Israel, a person with a severe case is someone who has an elevated respiratory rate and someone who has a blood oxygen level less than 94%. In the United States, that kind of patient wouldn’t necessarily be hospitalized.
In the end, one of the two committee members who wanted full approval for Pfizer’s third doses said he was satisfied with the outcome.
Mark Sawyer, MD, a professor of pediatrics and infectious disease at the University of California at San Diego, said he voted yes on the first question because he thought full approval was the best way to give doctors the flexibility to prescribe the shots to vulnerable individuals.
“I’m really glad we authorized a vaccine for a third dose, and I plan to go out and get my vaccine this afternoon,” Dr. Sawyer said, noting that he was at high risk as a health care provider.
This article was updated 9/19/21.
A version of this article first appeared on Medscape.com.
An expert panel that advises the Food and Drug Administration on its regulatory decisions voted Sept. 17 against recommending third doses of Pfizer’s COVID-19 vaccine for younger Americans.
But they didn’t kill the idea of booster shots completely.
In a dramatic, last-minute pivot, the 18 members of the FDA’s Vaccines and Related Biological Products Advisory Committee unanimously voted to recommend the FDA make boosters available for seniors and others at high risk of severe outcomes from COVID-19, including health care workers.
The 16-2 vote was a rebuttal to Pfizer’s initial request. The company had asked the FDA to allow it to offer third doses to all Americans over the age of 16 at least six months after their second shot.
The company requested an amendment to the full approval the FDA granted in August. That is the typical way boosters are authorized in the U.S., but it requires a higher bar of evidence and more regulatory scrutiny than the agency had been able to give since Pfizer filed for the change just days after its vaccine was granted full approval.
The committee’s actions were also a rebuff to the Biden administration, which announced before the FDA approved them that boosters would be rolled out to the general public Sept. 20. The announcement triggered the resignations of two of the agency’s top vaccine reviewers, who both participated in the Sept. 17 meeting.
After initially voting against Pfizer’s request to amend its license, the committee then worked on the fly with FDA officials to craft a strategy that would allow third doses to be offered under an emergency use authorization (EUA).
An EUA requires a lower standard of evidence and is more specific. It will restrict third doses to a more defined population than a change to the license would. It will also require Pfizer to continue to monitor the safety of third doses as they begin to be administered.
“This should demonstrate to the public that the members of this committee are independent of the FDA and that we do, in fact, bring our voices to the table when we are asked to serve on this committee,” said Archana Chattergee, MD, a pediatric infectious disease specialist who is dean of the Chicago Medical School at Rosalind Franklin University in Illinois.
The FDA doesn’t have to follow the committee’s recommendation, but almost certainly will, though regulators said they may still make some changes.
“We are not bound at FDA by your vote, we can tweak this,” said Peter Marks, MD, director of the Center for Biologics Evaluation and Research at the FDA. Dr. Marks participated in the meeting and helped to draft the revised proposal.
If the FDA issues the recommended EUA, a council of independent advisors to the CDC will make specific recommendations about how the third doses should be given. After the CDC director weighs in, boosters will begin rolling out to the public.
Moderna submitted data to the FDA on Sept. 1 in support of adding a booster dose to its regimen. The agency has not yet scheduled a public review of that data.
The Biden administration is prepared to administer shots as soon as they get the green light, Surgeon General Vivek Murthy, MD, said at a White House briefing earlier Sept. 17.
"This process is consistent with what we outlined in August where our goals were to stay ahead of the virus," Dr. Murthy said. "Our goal then and now is to protect the health and well-being of the public. As soon as the FDA and CDC complete their evaluations, we will be ready to move forward accordingly."
He added, "We've used this time since our August announcement to communicate and coordinate with pharmacy partners, nursing homes, states, and localities."
White House COVID-19 Response Coordinator Jeff Zients said vaccine supply is "in good shape for all Americans to get boosters as recommended."
Taking cues from Israel
In considering Pfizer’s original request, the committee overwhelmingly felt that they didn’t have enough information to say that the benefits of an additional dose of vaccine in 16- and 17-year-olds would outweigh its risk. Teens have the highest risk of rare heart inflammation after vaccination, a side effect known as myocarditis. It is not known how the vaccines are causing these cases of heart swelling. Most who have been diagnosed with the condition have recovered, though some have needed hospital care.
Pfizer didn’t include 16- and 17-year-olds in its studies of boosters, which included about 300 people between the ages of 18 and 55. The company acknowledged that gap in its data but pointed to FDA guidance that said evidence from adults could be extrapolated to teens.
“We don’t know that much about risks,” said committee member Eric Rubin, MD, who is editor-in-chief of the New England Journal of Medicine.
Much of the data on the potential benefits and harms of third Pfizer doses comes from Israel, which first began rolling out boosters to older adults in July.
In a highly anticipated presentation, Sharon Alroy-Preis, Israel’s director of public health services, joined the meeting to describe Israel’s experience with boosters.
Israel began to see a third surge of COVID-19 cases in December.
“This was after having two waves and two lockdowns,” Ms. Alroy-Preis said. By the third surge, she said, Israelis were tired.
“We decided on a lockdown, but the compliance of the public wasn’t as it was in the previous two waves,” she said.
Then the vaccine arrived. Israel started vaccinations as soon as the FDA approved it, and they quickly vaccinated a high percentage of their population, about 3 months faster than the rest of the world.
All vaccinations are reported and tracked by the Ministry of Health, so the country is able to keep close tabs on how well the shots are working.
As vaccines rolled out, cases fell dramatically. The pandemic seemed to be behind them. Delta arrived in March. By June, their cases were doubling every 10 days, despite about 80% of their most vulnerable adults being fully vaccinated, she said.
Most concerning was that about 60% of severe cases were breakthrough cases in fully vaccinated individuals.
“We had to stop and figure out, was this a Delta issue,” she said. “Or was this a waning immunity issue.”
“We had some clue that it might not be the Delta variant, at least not alone,” she said.
People who had originally been first in line for the vaccines, seniors and health care workers, were having the highest rates of breakthrough infections. People further away from their second dose were more likely to get a breakthrough infection.
Ms. Alroy-Preis said that if they had not started booster doses in July, their hospitals would have been overwhelmed. They had projected that they would have 2,000 cases in the hospital each day.
Boosters have helped to flatten the curve, though they are still seeing a significant number of infections.
Data from Israel presented at the meeting show boosters are largely safe and effective at reducing severe outcomes in seniors. Israeli experience also showed that third doses, which generate very high levels of neutralizing antibodies—the first and fastest line of the body’s immune defense - -may also slow transmission of the virus.
Key differences in the U.S.
The benefit of slowing down the explosive spread of a highly contagious virus was tantalizing, but many members noted that circumstances in Israel are very different than in the United States. Israel went into its current Delta surge already having high levels of vaccination in its population. They also relied on the Pfizer vaccine almost exclusively for their campaign.
The United States used a different mix of vaccines – Pfizer, Moderna, and Johnson & Johnson -- and doesn’t have the same high level of vaccination coverage of its population.
In the United States, transmission is mainly being driven by unvaccinated people, Dr. Rubin noted.
“That really means the primary benefit is going to be in reducing disease,” he said, “And we know the people who are going to benefit from that … and those are the kinds of people the FDA has already approved a third dose for,” he said, referring to those with underlying health conditions.
But Israel only began vaccinating younger people a few weeks ago. Most are still within a window where rare risks like myocarditis could appear, Rubin noted.
He and other members of the committee said they wished they had more information about the safety of third doses in younger adults.
“We don’t have that right now, and I don’t think I would be comfortable giving it to a 16-year-old,” he said.
At the same time, the primary benefit for third doses would be in preventing severe disease, and overall, data from the United States and other countries show that two doses of the vaccines remain highly effective at preventing hospitalization and death.
Asked why Israel began to see more severe cases in fully vaccinated people, the CDC’s Sara Oliver, MD, a disease detective with the CDC, said it was probably due to a mix of factors including the fact that Israel defines severe cases a little differently.
In the United States, a severe case is generally a person who has to be hospitalized or who has died from the infection. In Israel, a person with a severe case is someone who has an elevated respiratory rate and someone who has a blood oxygen level less than 94%. In the United States, that kind of patient wouldn’t necessarily be hospitalized.
In the end, one of the two committee members who wanted full approval for Pfizer’s third doses said he was satisfied with the outcome.
Mark Sawyer, MD, a professor of pediatrics and infectious disease at the University of California at San Diego, said he voted yes on the first question because he thought full approval was the best way to give doctors the flexibility to prescribe the shots to vulnerable individuals.
“I’m really glad we authorized a vaccine for a third dose, and I plan to go out and get my vaccine this afternoon,” Dr. Sawyer said, noting that he was at high risk as a health care provider.
This article was updated 9/19/21.
A version of this article first appeared on Medscape.com.
As opioid deaths climb, human trials begin for vaccine
Opioid-related drug overdose deaths in the United States exploded to an estimated record high of 69,031 people in 2020, topping the 49,860 deaths logged in 2019, according to a new report from the Centers for Disease Control and Prevention. Most of the deaths involved synthetic opioids such as fentanyl.
President Joe Biden has pledged more than $10 billion to expand access to prevention, treatment, and recovery services. The money is important as people receiving treatment for opioid use disorder have a high risk for relapse, and that means a high risk for opioid overdose.
Now, researchers are studying a possible bridge to successful recovery: A vaccine that could blunt the drugs’ ability to cause harm.
The first such vaccines are now entering clinical trials, raising hopes of adding another tool to the antiaddiction armamentarium. But even if the vaccines prove safe and effective, their success could generate some new problems to solve.
An advantage of vaccines is that their effects can last for several months, said trial investigator Sandra Comer, PhD, professor of neurobiology and psychiatry at Columbia University Irving Medical Center, New York. Dropout rates for existing medical therapies for opioid use disorder are as high as 50% at 6 months, and a vaccine could protect people from overdose and give them time to re-enter treatment.
“It serves as a bit of a safety net,” she said.
The first vaccine to enter a trial targets oxycodone. Volunteers are being recruited who have a diagnosis of opioid use disorder but are not being medically treated and are still using opioids. A third of them will receive a placebo vaccine, a third will receive a low-dose injection of vaccine, and the other third will receive a high-dose vaccine.
A shot against oxycodone
Researchers are primarily tracking the safety of the shot, but they’re also looking at whether vaccination prevents the euphoria that opioids usually produce. They expect to enroll 24 people initially but expand to 45 if results look promising.
In response to the shot, the body produces antibodies, proteins that tag oxycodone and keep it from reaching the brain. If the drug can’t reach brain cells, it can’t produce euphoria. And more important for lifesaving effects, it can’t block the brain’s signals to the body to breathe. The vaccine has already performed well in animal studies.
Previous trials of vaccines for cocaine and nicotine failed. Those vaccines made it to the last clinical trial stage, but didn’t prove effective overall. So this time, investigators plan to track antibody levels in participants, examining blood samples for signs of a good immune response to the vaccine.
But even though earlier cocaine and nicotine vaccines didn’t work for everybody, there were some people they seemed to help. This is why investigators involved in opioid vaccine trials want to track immune responses, said Marco Pravetoni, PhD, associate professor of pharmacology and medicine at the University of Minnesota, Minneapolis, whose team will be assessing the blood samples. Ultimately, a doctor might even be able to use this information to tailor vaccine selection to a specific person.
Dr. Pravetoni also said that oxycodone is one of three vaccine targets – the other two are heroin and fentanyl – that researchers hope to combine into a single shot. Recipients might need to have one shot a month for the first 3 to 4 months and then receive annual boosters.
Stopping the pain
The vaccines also raise some issues that need attention, said Cody Wenthur, PharmD, PhD, assistant professor of pharmacy at the University of Wisconsin–Madison, who is not involved in the vaccine trials.
“If you’re vaccinated against oxycodone, you might not have access to adequate pain control if you get into a car accident, for example,” he said.
Clinicians could use other opioids for pain management, but limiting the opioids that the vaccine targets is a “double-edged sword,” said Dr. Wenthur, because vaccinated people could just switch their opioid of choice to one that a vaccine does not inhibit.
Although these issues need to be addressed, vaccines, if successful, will have an important role. Dr. Wenthur noted a survey of pharmacists and pharmacy students that he and his group conducted showing that respondents “overwhelmingly” viewed a potential vaccine as helpful.
said Dr. Pravetoni. He mentioned the 2002 incident when terrorists took over a theater in Moscow and Russian special forces are thought to have used an aerosolized form of fentanyl to incapacitate everyone in the room. More than 100 of the hostages died, and the episode raised the specter of opioids being used in chemical attacks.
Dr. Pravetoni said vaccination could offer protection for first responders, law enforcement or other people whose professions place them at risk for inhalation, either accidentally or through such attacks.
These or other real-world applications for people at risk for exposure are several years away. Dr. Pravetoni said it took 10 years to get to this phase and estimates that, in about 5 years, a vaccine that targets multiple opioid drugs might enter the first clinical trial.
A version of this article first appeared on WebMD.com.
Opioid-related drug overdose deaths in the United States exploded to an estimated record high of 69,031 people in 2020, topping the 49,860 deaths logged in 2019, according to a new report from the Centers for Disease Control and Prevention. Most of the deaths involved synthetic opioids such as fentanyl.
President Joe Biden has pledged more than $10 billion to expand access to prevention, treatment, and recovery services. The money is important as people receiving treatment for opioid use disorder have a high risk for relapse, and that means a high risk for opioid overdose.
Now, researchers are studying a possible bridge to successful recovery: A vaccine that could blunt the drugs’ ability to cause harm.
The first such vaccines are now entering clinical trials, raising hopes of adding another tool to the antiaddiction armamentarium. But even if the vaccines prove safe and effective, their success could generate some new problems to solve.
An advantage of vaccines is that their effects can last for several months, said trial investigator Sandra Comer, PhD, professor of neurobiology and psychiatry at Columbia University Irving Medical Center, New York. Dropout rates for existing medical therapies for opioid use disorder are as high as 50% at 6 months, and a vaccine could protect people from overdose and give them time to re-enter treatment.
“It serves as a bit of a safety net,” she said.
The first vaccine to enter a trial targets oxycodone. Volunteers are being recruited who have a diagnosis of opioid use disorder but are not being medically treated and are still using opioids. A third of them will receive a placebo vaccine, a third will receive a low-dose injection of vaccine, and the other third will receive a high-dose vaccine.
A shot against oxycodone
Researchers are primarily tracking the safety of the shot, but they’re also looking at whether vaccination prevents the euphoria that opioids usually produce. They expect to enroll 24 people initially but expand to 45 if results look promising.
In response to the shot, the body produces antibodies, proteins that tag oxycodone and keep it from reaching the brain. If the drug can’t reach brain cells, it can’t produce euphoria. And more important for lifesaving effects, it can’t block the brain’s signals to the body to breathe. The vaccine has already performed well in animal studies.
Previous trials of vaccines for cocaine and nicotine failed. Those vaccines made it to the last clinical trial stage, but didn’t prove effective overall. So this time, investigators plan to track antibody levels in participants, examining blood samples for signs of a good immune response to the vaccine.
But even though earlier cocaine and nicotine vaccines didn’t work for everybody, there were some people they seemed to help. This is why investigators involved in opioid vaccine trials want to track immune responses, said Marco Pravetoni, PhD, associate professor of pharmacology and medicine at the University of Minnesota, Minneapolis, whose team will be assessing the blood samples. Ultimately, a doctor might even be able to use this information to tailor vaccine selection to a specific person.
Dr. Pravetoni also said that oxycodone is one of three vaccine targets – the other two are heroin and fentanyl – that researchers hope to combine into a single shot. Recipients might need to have one shot a month for the first 3 to 4 months and then receive annual boosters.
Stopping the pain
The vaccines also raise some issues that need attention, said Cody Wenthur, PharmD, PhD, assistant professor of pharmacy at the University of Wisconsin–Madison, who is not involved in the vaccine trials.
“If you’re vaccinated against oxycodone, you might not have access to adequate pain control if you get into a car accident, for example,” he said.
Clinicians could use other opioids for pain management, but limiting the opioids that the vaccine targets is a “double-edged sword,” said Dr. Wenthur, because vaccinated people could just switch their opioid of choice to one that a vaccine does not inhibit.
Although these issues need to be addressed, vaccines, if successful, will have an important role. Dr. Wenthur noted a survey of pharmacists and pharmacy students that he and his group conducted showing that respondents “overwhelmingly” viewed a potential vaccine as helpful.
said Dr. Pravetoni. He mentioned the 2002 incident when terrorists took over a theater in Moscow and Russian special forces are thought to have used an aerosolized form of fentanyl to incapacitate everyone in the room. More than 100 of the hostages died, and the episode raised the specter of opioids being used in chemical attacks.
Dr. Pravetoni said vaccination could offer protection for first responders, law enforcement or other people whose professions place them at risk for inhalation, either accidentally or through such attacks.
These or other real-world applications for people at risk for exposure are several years away. Dr. Pravetoni said it took 10 years to get to this phase and estimates that, in about 5 years, a vaccine that targets multiple opioid drugs might enter the first clinical trial.
A version of this article first appeared on WebMD.com.
Opioid-related drug overdose deaths in the United States exploded to an estimated record high of 69,031 people in 2020, topping the 49,860 deaths logged in 2019, according to a new report from the Centers for Disease Control and Prevention. Most of the deaths involved synthetic opioids such as fentanyl.
President Joe Biden has pledged more than $10 billion to expand access to prevention, treatment, and recovery services. The money is important as people receiving treatment for opioid use disorder have a high risk for relapse, and that means a high risk for opioid overdose.
Now, researchers are studying a possible bridge to successful recovery: A vaccine that could blunt the drugs’ ability to cause harm.
The first such vaccines are now entering clinical trials, raising hopes of adding another tool to the antiaddiction armamentarium. But even if the vaccines prove safe and effective, their success could generate some new problems to solve.
An advantage of vaccines is that their effects can last for several months, said trial investigator Sandra Comer, PhD, professor of neurobiology and psychiatry at Columbia University Irving Medical Center, New York. Dropout rates for existing medical therapies for opioid use disorder are as high as 50% at 6 months, and a vaccine could protect people from overdose and give them time to re-enter treatment.
“It serves as a bit of a safety net,” she said.
The first vaccine to enter a trial targets oxycodone. Volunteers are being recruited who have a diagnosis of opioid use disorder but are not being medically treated and are still using opioids. A third of them will receive a placebo vaccine, a third will receive a low-dose injection of vaccine, and the other third will receive a high-dose vaccine.
A shot against oxycodone
Researchers are primarily tracking the safety of the shot, but they’re also looking at whether vaccination prevents the euphoria that opioids usually produce. They expect to enroll 24 people initially but expand to 45 if results look promising.
In response to the shot, the body produces antibodies, proteins that tag oxycodone and keep it from reaching the brain. If the drug can’t reach brain cells, it can’t produce euphoria. And more important for lifesaving effects, it can’t block the brain’s signals to the body to breathe. The vaccine has already performed well in animal studies.
Previous trials of vaccines for cocaine and nicotine failed. Those vaccines made it to the last clinical trial stage, but didn’t prove effective overall. So this time, investigators plan to track antibody levels in participants, examining blood samples for signs of a good immune response to the vaccine.
But even though earlier cocaine and nicotine vaccines didn’t work for everybody, there were some people they seemed to help. This is why investigators involved in opioid vaccine trials want to track immune responses, said Marco Pravetoni, PhD, associate professor of pharmacology and medicine at the University of Minnesota, Minneapolis, whose team will be assessing the blood samples. Ultimately, a doctor might even be able to use this information to tailor vaccine selection to a specific person.
Dr. Pravetoni also said that oxycodone is one of three vaccine targets – the other two are heroin and fentanyl – that researchers hope to combine into a single shot. Recipients might need to have one shot a month for the first 3 to 4 months and then receive annual boosters.
Stopping the pain
The vaccines also raise some issues that need attention, said Cody Wenthur, PharmD, PhD, assistant professor of pharmacy at the University of Wisconsin–Madison, who is not involved in the vaccine trials.
“If you’re vaccinated against oxycodone, you might not have access to adequate pain control if you get into a car accident, for example,” he said.
Clinicians could use other opioids for pain management, but limiting the opioids that the vaccine targets is a “double-edged sword,” said Dr. Wenthur, because vaccinated people could just switch their opioid of choice to one that a vaccine does not inhibit.
Although these issues need to be addressed, vaccines, if successful, will have an important role. Dr. Wenthur noted a survey of pharmacists and pharmacy students that he and his group conducted showing that respondents “overwhelmingly” viewed a potential vaccine as helpful.
said Dr. Pravetoni. He mentioned the 2002 incident when terrorists took over a theater in Moscow and Russian special forces are thought to have used an aerosolized form of fentanyl to incapacitate everyone in the room. More than 100 of the hostages died, and the episode raised the specter of opioids being used in chemical attacks.
Dr. Pravetoni said vaccination could offer protection for first responders, law enforcement or other people whose professions place them at risk for inhalation, either accidentally or through such attacks.
These or other real-world applications for people at risk for exposure are several years away. Dr. Pravetoni said it took 10 years to get to this phase and estimates that, in about 5 years, a vaccine that targets multiple opioid drugs might enter the first clinical trial.
A version of this article first appeared on WebMD.com.