User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'main-prefix')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
Antidepressants for patients who are breastfeeding: What to consider
Ms. D, age 32, recently gave birth to her second child. Her psychiatric history includes major depressive disorder. She had been stable on mirtazapine 30 mg at bedtime for 3 years. Based on clinical stability and patient preference, Ms. D elected to taper off mirtazapine 1 month prior to delivery. Now at 1 month postdelivery, Ms. D notes the reemergence of her depressive symptoms; during her child’s latest pediatrician visit, she scores 15 on the Edinburgh Postnatal Depression Scale (EPDS). She breastfeeds her baby and wants more information on the safety of taking an antidepressant while breastfeeding.
Ms. D discusses her previous use of mirtazapine with her treatment team. The team reviews the available resources with Ms. D and together they plan to make a shared decision regarding treatment of her depression at her next appointment.
The American Academy of Pediatrics1 and World Health Organization2 recommend exclusive breastfeeding of infants for their first 6 months of life and support it as a complement to other foods through and beyond age 2. Untreated conditions such as postpartum depression impact maternal well-being and may interfere with parenting and child development. In fact, untreated maternal mental health leads to an increased risk of suicide, reduced maternal economic productivity, and worsened health for both mother and child.3
Because many women experience psychiatric symptoms before they become pregnant as well as during the perinatal period, questions often arise regardingthe use of psychiatric medications—specifically antidepressants—and their safety in patients who are breastfeeding. Key considerations regarding medication management should include the patient’s previous response to medications, the risks of untreated maternal mental illness, and evidence regarding risks and benefits in lactation. This article summarizes where to find evidence-based lactation information, how to interpret that information, and what information is available for select antidepressants.
Locating lactation information
Start by checking the manufacturer’s medication labeling (“prescribing information”) and medication information resources such as Micromedex (www.micromedexsolutions.com) and Lexicomp (www.wolterskluwer.com/en/solutions/lexicomp). The updated labeling includes a risk/benefit assessment of available data on the risk for continued use of a medication during pregnancy compared to the risk if a medication is discontinued and the disorder goes untreated.4 The “breastfeeding considerations” section of medication labeling include details regarding the presence of the medication and the amount of it in breastmilk, adverse events in infants exposed to the medication through breastmilk, and additional pertinent data as applicable. Lexicomp includes information regarding breastfeeding considerations, and a subscription may also include access to Briggs Drugs in Pregnancy and Lactation’s information pages. Micromedex includes its own lactation safety rating scale score.
Several other resources can help guide clinicians toward patient-specific recommendations. From the National Library of Medicine, LactMed (https://www.ncbi.nlm.nih.gov/books/NBK501922/) allows clinicians to search for specific medications to see what information exists pertaining to medication levels in breastmilk and infant blood as well as potential adverse effects in the nursing infant and/or on lactation and breastmilk.5 LactMed provides information regarding alternative medications to consider and references from which the information was gathered.
Another helpful resource is the InfantRisk Center from Texas Tech University Health Sciences Center, which includes a free call center for parents and clinicians who have questions about medications and breastfeeding (806-352-2519; Monday through Friday, 8
Continue to: How to interpret the information
How to interpret the information
Medication levels in breastmilk are affected by several properties, such as the medication’s molecular weight, protein binding, pKa, and volume of distribution. A few commonly used terms in lactation literature for medications include the relative infant dose (RID) and milk/plasma (M/P) ratio.
RID provides information about relative medication exposure for the infant. It is calculated by dividing the infant’s dose of a medication via breastmilk (mg/kg/d) by the mother’s dose (mg/kg/d).7 Most consider an RID <10% to be safe.7
M/P is the ratio of medication concentration in the mother’s milk divided by the medication concentration in the mother’s plasma. A ratio <1 is preferable and generally indicates that a low level of medication has been transferred to human milk.7
Another factor that can be evaluated is protein binding. Medications that are highly protein-bound do not tend to pass as easily into breastmilk and can minimize infant exposure.
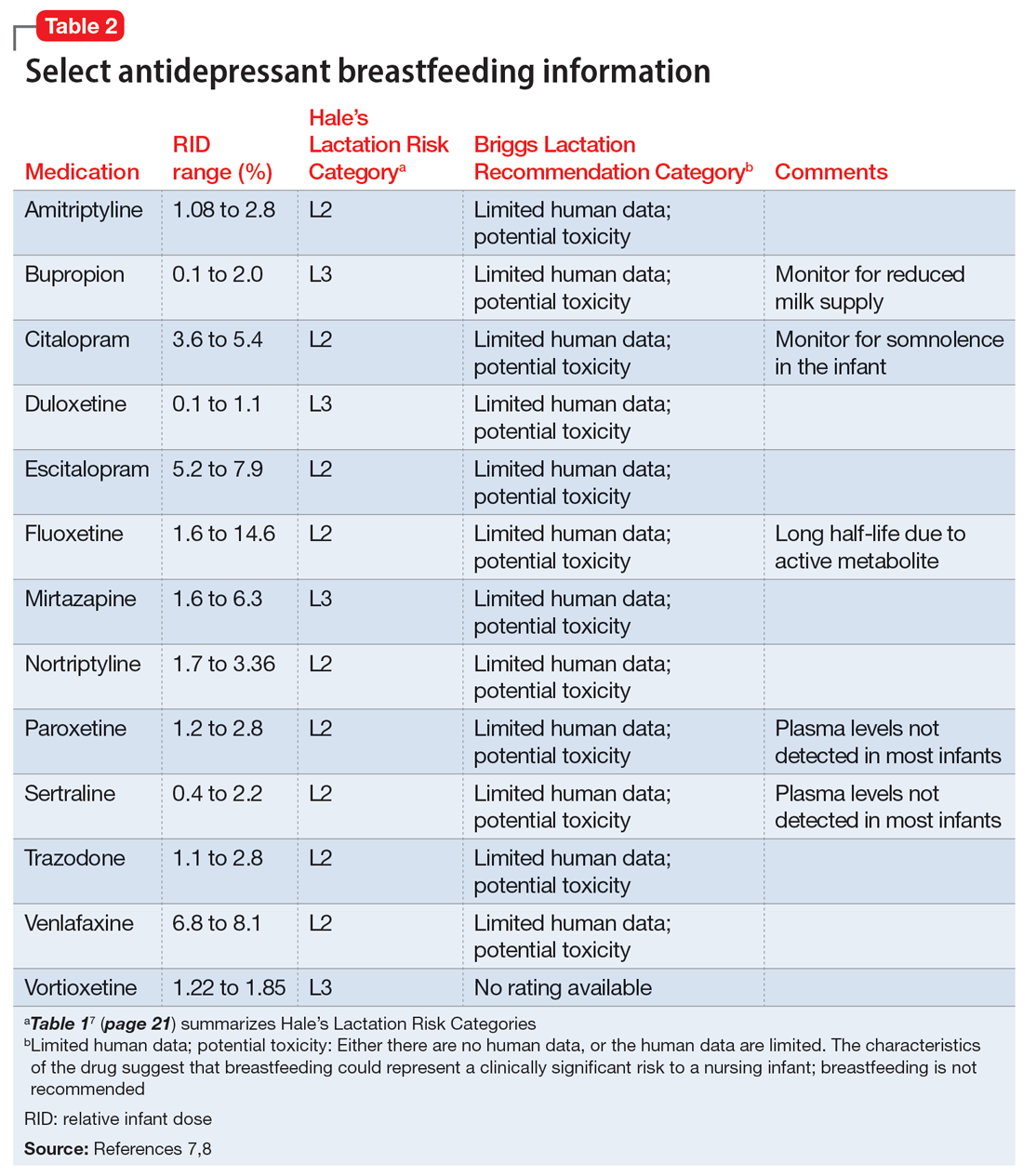
Several risk categorization systems are available, depending upon the resource used to obtain lactation information. One common system is Hale’s Lactation Risk Categories, with 5 safety levels ranging from L1 (breastfeeding compatible) to L5 (hazardous) (Table 17). Briggs et al8 utilize 7 categories to summarize recommendations ranging from breastfeeding-compatible to contraindicated; however, it is important to read the full medication monograph in the context of the rating provided. Table 27,8 provides breastfeeding information from Hale’s7 and from Briggs et al8 for some commonly used antidepressants.

In addition to interpreting available literature, it is also important to consider patient-specific factors, including (but not limited to) the severity of the patient’s psychiatric disorder and their previous response to medication. If a mother achieved remission on a particular antidepressant in the past, it may be preferable to restart that agent rather than trial a new medication.
CASE CONTINUED
Two weeks later and following the use of a variety of resources, Ms. D’s treatment team finds that mirtazapine is rated Probably Compatible (L3 in Hale’s Lactation Risk Categories), with an M/P ratio of 0.76.7 The RID of mirtazapine ranges from 1.6% to 6.3%, and limited data from infants exposed to maternal use of mirtazapine during breastfeeding have not shown adverse effects.5 The treatment team administers the EDPS to Ms. D again and she scores 18. Given Ms. D’s previous remission with mirtazapine, current severity of depressive symptoms, and the risk/benefit assessment from lactation resources, the decision is made to restart mirtazapine 15 mg/d at bedtime with the option to titrate up if indicated. Ms. D plans to continue breastfeeding and will monitor for signs of any adverse effects in her infant. The Figure provides a summary of navigating this individualized decision with patients.

Related Resources
- MotherToBaby. Medication fact sheets, option to contact for no-charge consultation, free patient education information materials. www.mothertobaby.org
- Reprotox. Summaries on effects of medications on pregnancy, reproduction, and development (subscription required). www.reprotox.org
Drug Brand Names
Bupropion • Wellbutrin
Citalopram • Celexa
Duloxetine • Cymbalta
Escitalopram • Lexapro
Fluoxetine • Prozac
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine • Effexor
Vortioxetine • Trintellix
1. American Academy of Pediatrics. American Academy of Pediatrics calls for more support for breastfeeding mothers within updated policy recommendations. June 27, 2022. Accessed April 7, 2023. https://www.aap.org/en/news-room/news-releases/aap/2022/american-academy-of-pediatrics-calls-for-more-support-for-breastfeeding-mothers-within-updated-policy-recommendations
2. World Health Organization. Breastfeeding recommendations. Accessed April 7, 2023. https://www.who.int/health-topics/breastfeeding#tab=tab_2
3. Margiotta C, Gao J, O’Neil S, et al. The economic impact of untreated maternal mental health conditions in Texas. BMC Pregnancy Childbirth. 2022;22(1):700. doi:10.1186/s12884-022-05001-6
4. Freeman MP, Farchione T, Yao L, et al. Psychiatric medications and reproductive safety: scientific and clinical perspectives pertaining to the US FDA pregnancy and lactation labeling rule. J Clin Psychiatry. 2018;79(4):18ah38120.
5. Drugs and Lactation Database (LactMed). National Library of Medicine (US); 2011. Updated April 18, 2016. Accessed September 29, 2022. https://www.ncbi.nlm.nih.gov/books/NBK501922/
6. InfantRisk Center Resources. InfantRisk Center at Texas Tech University Health Sciences Center. Accessed September 29, 2022. https://www.infantrisk.com/infantrisk-center-resources
7. Hale TW, Krutsch K. Hale’s Medications and Mother’s Milk 2023: A Manual of Lactational Pharmacology. Springer Publishing; 2023.
8. Briggs GG, Freeman RK, Towers CV, et al. Briggs Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. 12th ed. Lippincott Williams & Wilkins; 2021.
Ms. D, age 32, recently gave birth to her second child. Her psychiatric history includes major depressive disorder. She had been stable on mirtazapine 30 mg at bedtime for 3 years. Based on clinical stability and patient preference, Ms. D elected to taper off mirtazapine 1 month prior to delivery. Now at 1 month postdelivery, Ms. D notes the reemergence of her depressive symptoms; during her child’s latest pediatrician visit, she scores 15 on the Edinburgh Postnatal Depression Scale (EPDS). She breastfeeds her baby and wants more information on the safety of taking an antidepressant while breastfeeding.
Ms. D discusses her previous use of mirtazapine with her treatment team. The team reviews the available resources with Ms. D and together they plan to make a shared decision regarding treatment of her depression at her next appointment.
The American Academy of Pediatrics1 and World Health Organization2 recommend exclusive breastfeeding of infants for their first 6 months of life and support it as a complement to other foods through and beyond age 2. Untreated conditions such as postpartum depression impact maternal well-being and may interfere with parenting and child development. In fact, untreated maternal mental health leads to an increased risk of suicide, reduced maternal economic productivity, and worsened health for both mother and child.3
Because many women experience psychiatric symptoms before they become pregnant as well as during the perinatal period, questions often arise regardingthe use of psychiatric medications—specifically antidepressants—and their safety in patients who are breastfeeding. Key considerations regarding medication management should include the patient’s previous response to medications, the risks of untreated maternal mental illness, and evidence regarding risks and benefits in lactation. This article summarizes where to find evidence-based lactation information, how to interpret that information, and what information is available for select antidepressants.
Locating lactation information
Start by checking the manufacturer’s medication labeling (“prescribing information”) and medication information resources such as Micromedex (www.micromedexsolutions.com) and Lexicomp (www.wolterskluwer.com/en/solutions/lexicomp). The updated labeling includes a risk/benefit assessment of available data on the risk for continued use of a medication during pregnancy compared to the risk if a medication is discontinued and the disorder goes untreated.4 The “breastfeeding considerations” section of medication labeling include details regarding the presence of the medication and the amount of it in breastmilk, adverse events in infants exposed to the medication through breastmilk, and additional pertinent data as applicable. Lexicomp includes information regarding breastfeeding considerations, and a subscription may also include access to Briggs Drugs in Pregnancy and Lactation’s information pages. Micromedex includes its own lactation safety rating scale score.
Several other resources can help guide clinicians toward patient-specific recommendations. From the National Library of Medicine, LactMed (https://www.ncbi.nlm.nih.gov/books/NBK501922/) allows clinicians to search for specific medications to see what information exists pertaining to medication levels in breastmilk and infant blood as well as potential adverse effects in the nursing infant and/or on lactation and breastmilk.5 LactMed provides information regarding alternative medications to consider and references from which the information was gathered.
Another helpful resource is the InfantRisk Center from Texas Tech University Health Sciences Center, which includes a free call center for parents and clinicians who have questions about medications and breastfeeding (806-352-2519; Monday through Friday, 8
Continue to: How to interpret the information
How to interpret the information
Medication levels in breastmilk are affected by several properties, such as the medication’s molecular weight, protein binding, pKa, and volume of distribution. A few commonly used terms in lactation literature for medications include the relative infant dose (RID) and milk/plasma (M/P) ratio.
RID provides information about relative medication exposure for the infant. It is calculated by dividing the infant’s dose of a medication via breastmilk (mg/kg/d) by the mother’s dose (mg/kg/d).7 Most consider an RID <10% to be safe.7
M/P is the ratio of medication concentration in the mother’s milk divided by the medication concentration in the mother’s plasma. A ratio <1 is preferable and generally indicates that a low level of medication has been transferred to human milk.7
Another factor that can be evaluated is protein binding. Medications that are highly protein-bound do not tend to pass as easily into breastmilk and can minimize infant exposure.
Several risk categorization systems are available, depending upon the resource used to obtain lactation information. One common system is Hale’s Lactation Risk Categories, with 5 safety levels ranging from L1 (breastfeeding compatible) to L5 (hazardous) (Table 17). Briggs et al8 utilize 7 categories to summarize recommendations ranging from breastfeeding-compatible to contraindicated; however, it is important to read the full medication monograph in the context of the rating provided. Table 27,8 provides breastfeeding information from Hale’s7 and from Briggs et al8 for some commonly used antidepressants.

In addition to interpreting available literature, it is also important to consider patient-specific factors, including (but not limited to) the severity of the patient’s psychiatric disorder and their previous response to medication. If a mother achieved remission on a particular antidepressant in the past, it may be preferable to restart that agent rather than trial a new medication.

CASE CONTINUED
Two weeks later and following the use of a variety of resources, Ms. D’s treatment team finds that mirtazapine is rated Probably Compatible (L3 in Hale’s Lactation Risk Categories), with an M/P ratio of 0.76.7 The RID of mirtazapine ranges from 1.6% to 6.3%, and limited data from infants exposed to maternal use of mirtazapine during breastfeeding have not shown adverse effects.5 The treatment team administers the EDPS to Ms. D again and she scores 18. Given Ms. D’s previous remission with mirtazapine, current severity of depressive symptoms, and the risk/benefit assessment from lactation resources, the decision is made to restart mirtazapine 15 mg/d at bedtime with the option to titrate up if indicated. Ms. D plans to continue breastfeeding and will monitor for signs of any adverse effects in her infant. The Figure provides a summary of navigating this individualized decision with patients.

Related Resources
- MotherToBaby. Medication fact sheets, option to contact for no-charge consultation, free patient education information materials. www.mothertobaby.org
- Reprotox. Summaries on effects of medications on pregnancy, reproduction, and development (subscription required). www.reprotox.org
Drug Brand Names
Bupropion • Wellbutrin
Citalopram • Celexa
Duloxetine • Cymbalta
Escitalopram • Lexapro
Fluoxetine • Prozac
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine • Effexor
Vortioxetine • Trintellix
Ms. D, age 32, recently gave birth to her second child. Her psychiatric history includes major depressive disorder. She had been stable on mirtazapine 30 mg at bedtime for 3 years. Based on clinical stability and patient preference, Ms. D elected to taper off mirtazapine 1 month prior to delivery. Now at 1 month postdelivery, Ms. D notes the reemergence of her depressive symptoms; during her child’s latest pediatrician visit, she scores 15 on the Edinburgh Postnatal Depression Scale (EPDS). She breastfeeds her baby and wants more information on the safety of taking an antidepressant while breastfeeding.
Ms. D discusses her previous use of mirtazapine with her treatment team. The team reviews the available resources with Ms. D and together they plan to make a shared decision regarding treatment of her depression at her next appointment.
The American Academy of Pediatrics1 and World Health Organization2 recommend exclusive breastfeeding of infants for their first 6 months of life and support it as a complement to other foods through and beyond age 2. Untreated conditions such as postpartum depression impact maternal well-being and may interfere with parenting and child development. In fact, untreated maternal mental health leads to an increased risk of suicide, reduced maternal economic productivity, and worsened health for both mother and child.3
Because many women experience psychiatric symptoms before they become pregnant as well as during the perinatal period, questions often arise regardingthe use of psychiatric medications—specifically antidepressants—and their safety in patients who are breastfeeding. Key considerations regarding medication management should include the patient’s previous response to medications, the risks of untreated maternal mental illness, and evidence regarding risks and benefits in lactation. This article summarizes where to find evidence-based lactation information, how to interpret that information, and what information is available for select antidepressants.
Locating lactation information
Start by checking the manufacturer’s medication labeling (“prescribing information”) and medication information resources such as Micromedex (www.micromedexsolutions.com) and Lexicomp (www.wolterskluwer.com/en/solutions/lexicomp). The updated labeling includes a risk/benefit assessment of available data on the risk for continued use of a medication during pregnancy compared to the risk if a medication is discontinued and the disorder goes untreated.4 The “breastfeeding considerations” section of medication labeling include details regarding the presence of the medication and the amount of it in breastmilk, adverse events in infants exposed to the medication through breastmilk, and additional pertinent data as applicable. Lexicomp includes information regarding breastfeeding considerations, and a subscription may also include access to Briggs Drugs in Pregnancy and Lactation’s information pages. Micromedex includes its own lactation safety rating scale score.
Several other resources can help guide clinicians toward patient-specific recommendations. From the National Library of Medicine, LactMed (https://www.ncbi.nlm.nih.gov/books/NBK501922/) allows clinicians to search for specific medications to see what information exists pertaining to medication levels in breastmilk and infant blood as well as potential adverse effects in the nursing infant and/or on lactation and breastmilk.5 LactMed provides information regarding alternative medications to consider and references from which the information was gathered.
Another helpful resource is the InfantRisk Center from Texas Tech University Health Sciences Center, which includes a free call center for parents and clinicians who have questions about medications and breastfeeding (806-352-2519; Monday through Friday, 8
Continue to: How to interpret the information
How to interpret the information
Medication levels in breastmilk are affected by several properties, such as the medication’s molecular weight, protein binding, pKa, and volume of distribution. A few commonly used terms in lactation literature for medications include the relative infant dose (RID) and milk/plasma (M/P) ratio.
RID provides information about relative medication exposure for the infant. It is calculated by dividing the infant’s dose of a medication via breastmilk (mg/kg/d) by the mother’s dose (mg/kg/d).7 Most consider an RID <10% to be safe.7
M/P is the ratio of medication concentration in the mother’s milk divided by the medication concentration in the mother’s plasma. A ratio <1 is preferable and generally indicates that a low level of medication has been transferred to human milk.7
Another factor that can be evaluated is protein binding. Medications that are highly protein-bound do not tend to pass as easily into breastmilk and can minimize infant exposure.
Several risk categorization systems are available, depending upon the resource used to obtain lactation information. One common system is Hale’s Lactation Risk Categories, with 5 safety levels ranging from L1 (breastfeeding compatible) to L5 (hazardous) (Table 17). Briggs et al8 utilize 7 categories to summarize recommendations ranging from breastfeeding-compatible to contraindicated; however, it is important to read the full medication monograph in the context of the rating provided. Table 27,8 provides breastfeeding information from Hale’s7 and from Briggs et al8 for some commonly used antidepressants.

In addition to interpreting available literature, it is also important to consider patient-specific factors, including (but not limited to) the severity of the patient’s psychiatric disorder and their previous response to medication. If a mother achieved remission on a particular antidepressant in the past, it may be preferable to restart that agent rather than trial a new medication.

CASE CONTINUED
Two weeks later and following the use of a variety of resources, Ms. D’s treatment team finds that mirtazapine is rated Probably Compatible (L3 in Hale’s Lactation Risk Categories), with an M/P ratio of 0.76.7 The RID of mirtazapine ranges from 1.6% to 6.3%, and limited data from infants exposed to maternal use of mirtazapine during breastfeeding have not shown adverse effects.5 The treatment team administers the EDPS to Ms. D again and she scores 18. Given Ms. D’s previous remission with mirtazapine, current severity of depressive symptoms, and the risk/benefit assessment from lactation resources, the decision is made to restart mirtazapine 15 mg/d at bedtime with the option to titrate up if indicated. Ms. D plans to continue breastfeeding and will monitor for signs of any adverse effects in her infant. The Figure provides a summary of navigating this individualized decision with patients.

Related Resources
- MotherToBaby. Medication fact sheets, option to contact for no-charge consultation, free patient education information materials. www.mothertobaby.org
- Reprotox. Summaries on effects of medications on pregnancy, reproduction, and development (subscription required). www.reprotox.org
Drug Brand Names
Bupropion • Wellbutrin
Citalopram • Celexa
Duloxetine • Cymbalta
Escitalopram • Lexapro
Fluoxetine • Prozac
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine • Effexor
Vortioxetine • Trintellix
1. American Academy of Pediatrics. American Academy of Pediatrics calls for more support for breastfeeding mothers within updated policy recommendations. June 27, 2022. Accessed April 7, 2023. https://www.aap.org/en/news-room/news-releases/aap/2022/american-academy-of-pediatrics-calls-for-more-support-for-breastfeeding-mothers-within-updated-policy-recommendations
2. World Health Organization. Breastfeeding recommendations. Accessed April 7, 2023. https://www.who.int/health-topics/breastfeeding#tab=tab_2
3. Margiotta C, Gao J, O’Neil S, et al. The economic impact of untreated maternal mental health conditions in Texas. BMC Pregnancy Childbirth. 2022;22(1):700. doi:10.1186/s12884-022-05001-6
4. Freeman MP, Farchione T, Yao L, et al. Psychiatric medications and reproductive safety: scientific and clinical perspectives pertaining to the US FDA pregnancy and lactation labeling rule. J Clin Psychiatry. 2018;79(4):18ah38120.
5. Drugs and Lactation Database (LactMed). National Library of Medicine (US); 2011. Updated April 18, 2016. Accessed September 29, 2022. https://www.ncbi.nlm.nih.gov/books/NBK501922/
6. InfantRisk Center Resources. InfantRisk Center at Texas Tech University Health Sciences Center. Accessed September 29, 2022. https://www.infantrisk.com/infantrisk-center-resources
7. Hale TW, Krutsch K. Hale’s Medications and Mother’s Milk 2023: A Manual of Lactational Pharmacology. Springer Publishing; 2023.
8. Briggs GG, Freeman RK, Towers CV, et al. Briggs Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. 12th ed. Lippincott Williams & Wilkins; 2021.
1. American Academy of Pediatrics. American Academy of Pediatrics calls for more support for breastfeeding mothers within updated policy recommendations. June 27, 2022. Accessed April 7, 2023. https://www.aap.org/en/news-room/news-releases/aap/2022/american-academy-of-pediatrics-calls-for-more-support-for-breastfeeding-mothers-within-updated-policy-recommendations
2. World Health Organization. Breastfeeding recommendations. Accessed April 7, 2023. https://www.who.int/health-topics/breastfeeding#tab=tab_2
3. Margiotta C, Gao J, O’Neil S, et al. The economic impact of untreated maternal mental health conditions in Texas. BMC Pregnancy Childbirth. 2022;22(1):700. doi:10.1186/s12884-022-05001-6
4. Freeman MP, Farchione T, Yao L, et al. Psychiatric medications and reproductive safety: scientific and clinical perspectives pertaining to the US FDA pregnancy and lactation labeling rule. J Clin Psychiatry. 2018;79(4):18ah38120.
5. Drugs and Lactation Database (LactMed). National Library of Medicine (US); 2011. Updated April 18, 2016. Accessed September 29, 2022. https://www.ncbi.nlm.nih.gov/books/NBK501922/
6. InfantRisk Center Resources. InfantRisk Center at Texas Tech University Health Sciences Center. Accessed September 29, 2022. https://www.infantrisk.com/infantrisk-center-resources
7. Hale TW, Krutsch K. Hale’s Medications and Mother’s Milk 2023: A Manual of Lactational Pharmacology. Springer Publishing; 2023.
8. Briggs GG, Freeman RK, Towers CV, et al. Briggs Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. 12th ed. Lippincott Williams & Wilkins; 2021.
Bipolar disorder: The foundational role of mood stabilizers
Bipolar disorder (BD) is a recurrent, life-long psychiatric illness affecting nearly 2% of the world population1,2 that is characterized by episodes of mania and depression interspersed among periods of relative mood stability.3 The illness causes an enormous health burden, which makes understanding its pathophysiology and treatment patterns a substantial priority.4 In the 1950s, lithium was found to be effective for treating acute manic episodes and preventing relapse in BD.5 Since then, valproate and carbamazepine also have been FDA-approved for treating mania.6,7 Antipsychotics have also shown evidence of efficacy in BD treatment,8,9 particularly for use in acute settings for more rapid effect or for a limited duration,10 which has led some to refer to them as “mood stabilizers.”11
In this article, we describe changes in trends of prescribing medications to treat BD, the role of ion dysregulation in the disorder, and how a better understanding of this dysregulation might impact the choice of treatment.
Changes in pharmacotherapy for bipolar disorder
From 1997 through 2016, the use of lithium for BD decreased from >30% of patients to 17.6% (with a nadir of 13.9% from 2009 to 2012).12 Over the same period, the use of nonlithium mood stabilizers decreased from 30.4% to approximately 4.8%, while second-generation antipsychotic (SGAs) use increased from 12.4% to 50.4%.12 Distressingly, antidepressant use increased from approximately 47% to 56.8%, and antidepressant use without concomitant mood stabilizers increased from 38% to 40.8%, although the rate of antidepressants without either a mood stabilizer or an antipsychotic remained relatively stable (14.9% to 16.8%).12 In randomized trials, when added to mood stabilizers, antidepressants have consistently failed to separate from placebo,13-15 but they can destabilize the illness, resulting in increases in mania, depression, and subsyndromal mixed symptoms.16-18
It is easy to understand clinicians’ attempts to address their patients’ distress due to depressive symptoms that do not resolve with mood stabilizers.19,20 Similarly, the increased use of antipsychotics is driven by evidence that antipsychotics are effective for treating bipolar depression and preventing the recurrence of manic and (for some antipsychotics) depressive episodes.21,22 However, long-term antipsychotic use causes brain volume change in patients with schizophrenia23 or major depressive disorder24 and in nonhuman primates25,26; metabolic abnormalities27-31; and cardiovascular adverse effects.32 Antipsychotics are believed to be associated with withdrawal psychosis.33,34 In the head-to-head Clinical Health Outcomes Initiative in Comparative Effectiveness for Bipolar Disorder (Bipolar CHOICE) study, quetiapine was as effective as lithium but associated with more adverse effects.35 Importantly, the estimated disability-adjusted life years of patients with BD increased by 54.4% from 6.02 million in 1990 to 9.29 million in 2017, which is greater than the increase in the incidence of BD (47.74%) over the same time.36 This means that despite the dramatic increase in treatment options for people with BD, functional outcomes have declined.
One major difference between antipsychotics and mood stabilizers is that antipsychotics do not alter the underlying abnormal pathology of BD.37 An ideal pharmacologic intervention is one that corrects a known pathophysiologic anomaly of the condition being treated. There are no demonstrated abnormalities in the dopamine or serotonin systems in individuals with BD, but long-term use of antipsychotics may create dopaminergic alterations.33 One of the most reproducible biomarkers associated with manic and bipolar depressed mood states is increased intracellular sodium38,39 and reduced ability to correct a sodium challenge.40-42 By normalizing intracellular sodium levels, lithium and the mood-stabilizing anticonvulsants uniquely and specifically counter known physiologic abnormalities in patients with BD.37,43
The role of ion dysregulation
The pathophysiology of BD remains elusive. A multitude of lines of evidence link BD to abnormal neuroimaging findings,22,44,45 oxidative stress,46 inflammation,47 and mitochondrial disease,48 but there is still no unifying understanding of these findings. Ion dysregulation appears to be central to understanding and treating BD.38,39
Despite extensive genetic studies, no genes have been identified that mediate >5% of the risk for BD. Nonetheless, 74% of all genes identified as mediating risk for BD code for proteins essential for the regulation of ion transport and membrane potential.49 The 2 genes that contribute the greatest risk are CACNA1C and ANK3, which code for a calcium channel and a cytoskeletal protein, respectively.50 ANK3 codes for ankyrin G, which plays a role in proper coupling of the voltage-gated sodium channels to the cytoskeleton.51 An additional risk gene, TRANK1, contains multiple ankyrin-like repeat domains, which suggests some shared functions with ANK3.52 More importantly, the most reproducible pathophysiologic findings in BD are dysregulation of sodium, potassium, hydrogen, and calcium transport, with consequent alteration of depolarization potential, neuronal excitability, and calcium-mediated processes.38,39,53-56 For example, increased sodium and calcium within cells have been observed in both mania and bipolar depression, and these levels normalize during euthymia. All medications that are effective for treating BD may reduce intracellular sodium or calcium; traditional mood stabilizers do so directly by inhibiting voltage-sensitive sodium channels in an activity-dependent manner or displacing intracellular sodium,43,57 whereas antipsychotics do so indirectly by increasing sodium pump activity through inhibition of second messengers of the dopamine D2 family of receptors.37
Continue to: The extent of ion dysregulation...
The extent of ion dysregulation is directly associated with the expressed mood state of the illness. A small reduction in the activity of the sodium pump results in a small increase in intracellular sodium (approximately 10 mM).39,58 This led to the hypothesis that increased intracellular sodium causes the transmembrane potential to increase closer to membrane depolarization threshold, which increases excitability of affected neurons.38,39,58 Neurons are likely to fire and propagate signals more easily, which may manifest as symptoms of mania, such as increased energy, activity, lability, excitability, irritability, tangentiality, and looseness of associations. As the process of increased intracellular sodium progresses, a minority of neurons are expected to have their transmembrane potentials depolarize sufficiently for the resting membrane potential to go beyond threshold potential.59 Such neurons are in a state of constant depolarization (also known as depolarization block), which disrupts neuronal circuits. The difficulty in progression of these signals results in the classic bipolar depression symptoms of low energy, reduced activity, and slowing of all brain activity that is seen as psychomotor slowing.38
Implications for treatment
Medications for treating bipolar illness include lithium, anticonvulsants, benzodiazepines, first-generation antipsychotics, and SGAs.37,43
Mood stabilizers (lithium and certain anticonvulsants) correct the previously mentioned sodium abnormality by reducing sodium entry into the cell in an activity-dependent manner.43 As the only agents that directly address a known pathophysiologic abnormality, they are foundational in the treatment of BD.60 Lithium effectively treats acute mania and prevents relapse.61 It preferentially targets the active neurons, entering through both voltage-responsive and neurotransmitter-coupled channels.43,62 This results in an increase of intracellular lithium concentrations to as much as 8 times that of the extracellular concentration.63 These ions displace intracellular sodium ions in a 1:1 ratio, which results in a reduced intracellular sodium concentration that reduces the excitability of neurons.43,57,62
Substantial evidence supports the use of valproic acid for initial and maintenance treatment of BD.64 It inhibits the voltage-sensitive sodium channel when the channel is open, which results in an activity-dependent action that selectively impacts rapidly firing neurons.43 The voltage-gated sodium channels exist nearly exclusively on the axon, beyond the hillock65; as such, valproic acid will only inhibit neurons that fire, whereas lithium accumulates throughout the neuron and will affect depolarization in the neuronal soma as well as the firing in the axon.43 Additionally, valproic acid has been observed to enhance gamma-aminobutyric acid (GABA) levels and transmission.43,66,67 A meta-analysis that included 6 randomized controlled trials illustrated that, acutely, valproate was not different from lithium’s overall efficacy (RR 1.02; 95% CI, 0.87 to 1.20), but was associated with reduced dropout rates compared with placebo or lithium (RR 0.82; 95% CI, 0.71 to 0.95 and RR 0.87; 95% CI, 0.77 to 0.98, respectively).64
Lamotrigine is an anticonvulsant used for initial and maintenance treatment of BD, with greater efficacy for depressive episodes68; it also has notable effect for treating bipolar depression, although it is not FDA-approved for this indication.69 Lamotrigine inhibits sodium influx by binding to open voltage-gated sodium channels70 but also appears to reduce N-methyl-D-aspartate–mediated sodium entry,71 thereby acting both prehillock and posthillock.
Continue to: Carbamazepine is an anticonvulsant...
Carbamazepine is an anticonvulsant FDA-approved for treating BD.7 Like valproate, it acts by inhibiting voltage-gated sodium channels in an activity-dependent manner,72 which means it preferentially inhibits the most active neurons and those with higher intracellular sodium.43
Benzodiazepines, which have shown to be effective for treating acute mania,73 potentiate synaptic GABA receptors accruing an elevation in intracellular chloride influx.74 Despite acute efficacy, benzodiazepine use is limited because these agents are associated with worsening long-term, substance use–related outcomes.75,76
Antipsychotics are effective for treating mood disorders,60,76 and their use has been rising dramatically.12 The antimanic effect of all antipsychotics is believed to be mediated through dopamine D2 blockade, since use of a dose sufficient to block D2 receptors is required, and haloperidol, which acts exclusively on the D2 receptor, is equal to SGAs in its antimanic effect.77 Blockade of the D2 receptor will increase the activity of the sodium pump (sodium and potassium-activated adenosine triphosphatase) thus reducing intracellular sodium and calcium concentrations.37 When antipsychotics are used as antidepressants, they are generally used at doses lower than those used to treat mania.78
Antipsychotics are effective for treating BD, and may work more quickly than other agents for treating acute mania.79 However, maintenance or prevention trials tend to favor mood stabilizers.35,60,80 Several add-on studies have found the combination of a mood stabilizer plus an antipsychotic is superior to a mood stabilizer alone or an antipsychotic alone.81
An argument for mood stabilizers
Evidence suggests mood stabilizers and other approaches, such as antipsychotics, are almost equivalent for treating acute mania, with a small clinical advantage of mood stabilizers for preventing relapse. In general, current treatment guidelines do not distinguish mood stabilizers from antipsychotics as the first-line treatment.82 Over the past 20 years, antipsychotic use has increased while mood stabilizer use has decreased, so that presently a patient with BD is more likely to be prescribed an antipsychotic than a mood stabilizer.12 Over the same time, dysfunction among patients with BD has increased.33 Antipsychotics are appealing because they appear to be equally effective and generally well tolerated. But these agents cause problems that are difficult to see in routine visits, such as metabolic27-31 and cardiovascular adverse effects29 as well as reductions in brain volume.23-26 Mechanistic research suggests that mood stabilizers directly correct known pathophysiologic anomalies with additional protective effects, whereas antipsychotics appear to create new abnormalities and contribute to medical problems. Clinicians need to look beyond the similarities in acute efficacy and make a more broadly supported, evidence-based choice for managing BD, which clearly places mood stabilizers as the first-line agent and antipsychotics as reasonable alternatives. At a minimum, mood stabilizers should be viewed as the foundation to which antipsychotics can be added.
Bottom Line
Traditional mood stabilizers—lithium and some anticonvulsants—are the only agents that directly address physiologic abnormalities associated with both mania and bipolar depression, including mood state–associated elevations of intracellular sodium. Because of their specificity, these agents maximize mood stabilization and minimize adverse effects.
Related Resources
- Karas A, Stummer L, Freedberg A. Psychiatric and nonpsychiatric indications for mood stabilizers and select antiepileptics. Current Psychiatry. 2022;21(4):34-38. doi:10.12788/cp.0230
- Koch J. Mood stabilizers: balancing tolerability, serum levels, and dosage. Current Psychiatry. 2021;20(7):37-40. doi:10.12788/cp.0147
Drug Brand Names
Carbamazepine • Tegretol
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Quetiapine • Seroquel
Valproate • Depakote, Depakene
1. Whiteford HA, Degenhardt L, Rehm J, et al. Global burden of disease attributable to mental and substance use disorders: findings from the Global Burden of Disease Study 2010. Lancet. 2013;382(9904):1575-1586. doi:10.1016/S0140-6736(13)61611-6
2. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the world mental health survey initiative. Arch Gen Psychiatry. 2011;68(3):241-251. doi:10.1001/archgenpsychiatry.2011.12
3. Müller JK, Leweke FM. Bipolar disorder: clinical overview. Article in English, German. Med Monatsschr Pharm. 2016;39(9):363-369.
4. Smith DJ, Whitham EA, Ghaemi SN. Bipolar disorder. Handb Clin Neurol. 2012;106:251-263. doi:10.1016/B978-0-444-52002-9.00015-2
5. Goodwin FK, Ghaemi SN. The impact of the discovery of lithium on psychiatric thought and practice in the USA and Europe. Aust N Z J Psychiatry. 1999;33 Suppl:S54-S64. doi:10.1111/j.1440-1614.1999.00669.x
6. Pope HG, McElroy SL, Keck PE, et al. Valproate in the treatment of acute mania. A placebo-controlled study. Arch Gen Psychiatry. 1991;48(1):62-68. doi:10.1001/archpsyc.1991.01810250064008
7. Weisler RH, Keck PE Jr, Swann AC, et al. Extended-release carbamazepine capsules as monotherapy for acute mania in bipolar disorder: a multicenter, randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2005;66(3):323-330. doi:10.4088/jcp.v66n0308
8. Tarr GP, Glue P, Herbison P. Comparative efficacy and acceptability of mood stabilizer and second generation antipsychotic monotherapy for acute mania--a systematic review and meta-analysis. J Affect Disord. 2011;134(1-3):14-19. doi:10.1016/j.jad.2010.11.009
9. Pahwa M, Sleem A, Elsayed OH, et al. New antipsychotic medications in the last decade. Curr Psychiatry Rep. 2021;23(12):87.
10. Correll CU, Sheridan EM, DelBello MP. Antipsychotic and mood stabilizer efficacy and tolerability in pediatric and adult patients with bipolar I mania: a comparative analysis of acute, randomized, placebo-controlled trials. Bipolar Disord. 2010;12(2):116-141. doi:10.1111/j.1399-5618.2010.00798.x
11. Rybakowski JK. Two generations of mood stabilizers. Int J Neuropsychopharmacol. 2007;10:709-711. doi:10.1017/s146114570700795x
12. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715. doi:10.1176/appi.ajp.2020.19091000
13. El-Mallakh RS. Adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;357(6):615; author reply 615-616.
14. Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;356(17):1711-1722. doi:10.1056/NEJMoa064135
15. Ghaemi SN, Whitham EA, Vohringer PA, et al. Citalopram for acute and preventive efficacy in bipolar depression (CAPE-BD): a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2021;82(1):19m13136. doi:10.4088/JCP.19m13136
16. El-Mallakh RS, Ghaemi SN, Sagduyu K, et al. Antidepressant-associated chronic irritable dysphoria (ACID) in STEP-BD patients. J Affect Disord. 2008;111(2-3):372-377. doi:10.1016/j.jad.2008.03.025
17. Ghaemi SN, Ostacher MM, El-Mallakh RS, et al. Antidepressant discontinuation in bipolar depression: a systematic treatment enhancement program for bipolar disorder (STEP-BD) randomized clinical trial of long-term effectiveness and safety. J Clin Psychiatry. 2010;71(4):372-380.
18. Strejilevich SA, Martino DJ, Marengo E, et al. Long-term worsening of bipolar disorder related with frequency of antidepressant exposure. Ann Clin Psychiatry. 2011;23(3):186-192.
19. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society of Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262. doi:10.1176/appi.ajp.2013.13020185
20. McIntyre RS, Calabrese JR. Bipolar depression: the clinical characteristics and unmet needs of a complex disorder. Curr Med Res Opin. 2019;35(11):1993-2005.
21. Fornaro M, Stubbs B, De Berardis D, et al. Atypical antipsychotics in the treatment of acute bipolar depression with mixed features: a systematic review and exploratory meta-analysis of placebo-controlled clinical trials. Int J Mol Sci. 2016;17(2):241. doi:10.3390/ijms17020241
22. Lindström L, Lindström E, Nilsson M, et al. Maintenance therapy with second generation antipsychotics for bipolar disorder – a systematic review and meta-analysis. J Affect Disord. 2017;213:138-150. doi:10.1016/j.jad.2017.02.012
23. Ho BC, Andreasen NC, Ziebell S, et al. Long-term antipsychotic treatment and brain volumes: a longitudinal study of first-episode schizophrenia. Arch Gen Psychiatry. 2011;68(2):128-137. doi:010.1001/archgenpsychiatry.2010.199
24. Voineskos AN, Mulsant BH, Dickie EW, et al. Effects of antipsychotic medication on brain structure in patients with major depressive disorder and psychotic features: neuroimaging findings in the context of a randomized placebo-controlled clinical trial. JAMA Psychiatry. 2020;77(7):674-683. doi:10.1001/jamapsychiatry.2020.0036
25. Konopaske GT, Bolo NR, Basu AC, et al. Time-dependent effects of haloperidol on glutamine and GABA homeostasis and astrocyte activity in the rat brain. Psychopharmacology (Berl). 2013;230(1):57-67. doi:10.1007/s00213-013-3136-3
26. Dorph-Petersen KA, Pierri JN, Perel JM, et al. The influence of chronic exposure to antipsychotic medications on brain size before and after tissue fixation: a comparison of haloperidol and olanzapine in macaque monkeys. Neuropsychopharmacology. 2005;30(9):1649-1661. doi:10.1038/sj.npp.1300710
27. McIntyre RS, Mancini DA, Basile VS, et al. Antipsychotic-induced weight gain: bipolar disorder and leptin. J Clin Psychopharmacol. 2003;23(4):323-327. doi:10.1097/01.jcp.0000085403.08426.f4
28. McIntyre RS, Konarski JZ, Wilkins K, et al. Obesity in bipolar disorder and major depressive disorder: results from a national community health survey on mental health and well-being. Can J Psychiatry. 2006;51(5):274-280. doi:10.1177/070674370605100502
29. McIntyre RS, Cha DS, Kim RD, et al. A review of FDA-approved treatment options in bipolar depression. CNS Spectr. 2013;18(Suppl 1):4-20. doi:10.1017/S1092852913000746
30. Barton BB, Segger F, Fischer K, et al. Update on weight-gain caused by antipsychotics: a systematic review and meta-analysis. Expert Opin Drug Saf. 2020;19(3):295-314. doi:10.1080/14740338.2020.1713091
31. Doane MJ, Bessonova L, Friedler HS, et al. Weight gain and comorbidities associated with oral second-generation antipsychotics: analysis of real-world data for patients with schizophrenia or bipolar I disorder. BMC Psychiatry. 2022;22(1):114. doi:10.1186/s12888-022-03758-w
32. Buckley NA, Sanders P. Cardiovascular adverse effects of antipsychotic drugs. Drug Saf. 2000;23(3):215-228. doi:10.2165/00002018-200023030-00004
33. Ali Z, Roque A, El-Mallakh RS. A unifying theory for the pathoetiologic mechanism of tardive dyskinesia. Med Hypotheses. 2020;140:109682. doi:10.1016/j.mehy.2020.109682
34. Sleem A, El-Mallakh RS. Adaptive changes to antipsychotics: their consequences and how to avoid them. Curr Psychiatry. 2022;21(7):46-50,52. doi: 10.12788/cp.0262
35. Nierenberg AA, McElroy SL, Friedman ES, et al. Bipolar CHOICE (Clinical Health Outcomes Initiative in Comparative Effectiveness): a pragmatic 6-month trial of lithium versus quetiapine for bipolar disorder. J Clin Psychiatry. 2016;77(1):90-99. doi:10.4088/JCP.14m09349
36. He H, Hu C, Ren Z, et al. Trends in the incidence and DALYs of bipolar disorder at global, regional, and national levels: results from the global burden of disease study 2017. J Psychiatr Res. 2020;125:96-105. doi:10.1016/j.jpsychires.2020.03.015
37. Roberts RJ, Repass R, El-Mallakh RS. Effect of dopamine on intracellular sodium: a common pathway for pharmacological mechanism of action in bipolar illness. World J Biol Psychiatry. 2010;11(2 Pt 2):181-187. doi:10.1080/15622970902718774
38. El-Mallakh RS, Wyatt RJ. The Na, K-ATPase hypothesis for bipolar illness. Biol Psychiatry. 1995;37(4):235-244. doi:10.1016/0006-3223(94)00201-D
39. El-Mallakh RS, Yff T, Gao Y. Ion dysregulation in the pathogenesis of bipolar disorder. Ann Depress Anxiety. 2016;3(1):1076.
40. Li R, El-Mallakh RS. Differential response of bipolar and normal control lymphoblastoid cell sodium pump to ethacrynic acid. J Affect Disord. 2004;80(1):11-17. doi:10.1016/S0165-0327(03)00044-2
41. Woodruff DB, El-Mallakh RS, Thiruvengadam AP. Validation of a diagnostic screening blood test for bipolar disorder. Ann Clin Psychiatry. 2012;24(2):135-139.
42. Gao Y, Lohano K, Delamere NA, et al. Ethanol normalizes glutamate-induced elevation of intracellular sodium in olfactory neuroepithelial progenitors from subjects with bipolar illness but not nonbipolar controls: biologic evidence for the self-medication hypothesis. Bipolar Disord. 2019;21(2):179-181. doi:10.1111/bdi.12737
43. El-Mallakh RS, Huff MO. Mood stabilizers and ion regulation. Harv Rev Psychiatry. 2001;9(1):23-32. doi:10.1080/10673220127873
44. Phillips ML, Swartz HA. A critical appraisal of neuroimaging studies of bipolar disorder: toward a new conceptualization of underlying neural circuitry and a road map for future research. Am J Psychiatry. 2014;171(8):829-843. doi:10.1176/appi.ajp.2014.13081008
45. Hibar DP, Westlye LT, Doan NT, et al. Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA Bipolar Disorder Working Group. Mol Psychiatry. 2018;23(4):932-942. doi:10.1038/mp.2017.73
46. Brown NC, Andreazza AC, Young LT. An updated meta-analysis of oxidative stress markers in bipolar disorder. Psychiatry Res. 2014;218(1-2):61-68. doi:10.1016/j.psychres.2014.04.005
47. Benedetti F, Aggio V, Pratesi ML, et al. Neuroinflammation in bipolar depression. Front Psychiatry. 2020;11:71. doi:10.3389/fpsyt.2020.00071
48. Andreazza AC, Duong A, Young LT. Bipolar disorder as a mitochondrial disease. Biol Psychiatry. 2018;83(9):720-721. doi:10.1016/j.biopsych.2017.09.018
49. Askland KD. Toward a biaxial model of “bipolar” affective disorders: further exploration of genetic, molecular and cellular substrates. J Affect Disord. 2006;94(1-3):35-66. doi:10.1016/j.jad.2006.01.033
50. Ferreira MA, O’Donovan MC, Meng YA, et al; Wellcome Trust Case Control Consortium. Collaborative genome-wide association analysis supports a role for ANK3 and CACNA1C in bipolar disorder. Nat Genet 2008;40(9):1056-1058. doi:10.1038/ng.209
51. Salvi AM, Bays JL, Mackin SR, et al. Ankyrin G organizes membrane components to promote coupling of cell mechanics and glucose uptake. Nat Cell Biol. 2021;23(5):457-466. doi:10.1038/s41556-021-00677-y
52. Gargus JJ. Ion channel functional candidate genes in multigenic neuropsychiatric disease. Biol Psychiatry. 2006;60(2):177-185. doi:10.1016/j.biopsych.2005.12.008
53. Dubovsky SL, Murphy J, Thomas M, et al. Abnormal intracellular calcium ion concentration in platelets and lymphocytes of bipolar patients. Am J Psychiatry 1992;149(1):118-120. doi:10.1176/ajp.149.1.118
54. Blaustein MP. Physiological effects of endogenous ouabain: control of intracellular Ca2+ stores and cell responsiveness. Am J Physiol. 1993;264(6 Pt 1):C1367–C1387. doi:10.1152/ajpcell.1993.264.6.C1367
55. El-Mallakh RS, Li R, Worth CA, et al. Leukocyte transmembrane potential in bipolar illness. J Affect Disord. 1996;41(1):33-37. doi:10.1016/0165-0327(96)00063-8
56. El-Mallakh RS, Gao Y, You P. Role of endogenous ouabain in the etiology of bipolar disorder. Int J Bipolar Disord. 2021;9(1):6. doi:10.1186/s40345-020-00213-1
57. Huang X, Lei Z, El‐Mallakh RS. Lithium normalizes elevated intracellular sodium. Bipolar Disord. 2007;9(3):298-300. doi:10.1111/j.1399-5618.2007.00429.x
58. Shaw DM. Mineral metabolism, mania, and melancholia. Br Med J. 1966;2(5508):262-267. doi:10.1136/bmj.2.5508.262
59. Qian K, Yu N, Tucker KR, et al. Mathematical analysis of depolarization block mediated by slow inactivation of fast sodium channels in midbrain dopamine neurons. J Neurophysiol. 2014;112(11):2779-2790. doi:10.1152/jn.00578.2014
60. Sleem A, El-Mallakh RS. Advances in the psychopharmacotherapy of bipolar disorder type I. Exp Opin Pharmacother. 2021;22(10):1267-1290. doi:10.1080/14656566.2021.1893306
61. Malhi GS., Tanious M, Das P, et al. Potential mechanisms of action of lithium in bipolar disorder. CNS Drugs. 2013;27(2):135-153. doi:10.1007/s40263-013-0039-0
62. Armett CJ, Ritchie JM. On the permeability of mammalian non-myelinated fibers to sodium and to lithium ions. J Physiol. 1963;165(1):130-140. doi:10.1113/jphysiol.1963.sp007047
63. Kabakov AY, Karkanias NB, Lenox RH, et al. Synapse-specific accumulation of lithium in intracellular microdomains: a model for uncoupling coincidence detection in the brain. Synapse. 1998;28(4):271-279. doi:10.1002/(SICI)1098-2396(199804)28:4<271::AID-SYN2>3.0.CO;2-6
64. Cipriani A, Reid K, Young AH, et al. Valproic acid, valproate and divalproex in the maintenance treatment of bipolar disorder. Cochrane Database Syst Rev. 2013;2013(10):CD003196. doi:10.1002/14651858.CD003196.pub2
65. Lai HC, Jan LY. The distribution and targeting of neuronal voltage-gated ion channels. Nat Rev Neurosci. 2006;7(7):548-562. doi:10.1038/nrn1938
66. Löscher W, Schmidt D. Increase of human plasma GABA by sodium valproate. Epilepsia. 1980;21(6):611-615. doi:10.1111/j.1528-1157.1980.tb04314.x
67. Owens MJ, Nemeroff CB. Pharmacology of valproate. Psychopharmacol Bull. 2003;37(Suppl 2):17-24.
68. Calabrese JR, Vieta E, Shelton MD. Latest maintenance data on lamotrigine in bipolar disorder. Eur Neuropsychopharmacol. 2003;13(Suppl 2):S57-S66. doi:10.1016/s0924-977x(03)00079-8
69. Geddes JR, Calabrese JR, Goodwin GM. Lamotrigine for treatment of bipolar depression: independent meta-analysis and meta-regression of individual patient data from five randomised trials. Br J Psychiatry. 2009;194(1):4-9. doi:10.1192/bjp.bp.107.048504
70. Nakatani Y, Masuko H, Amano T. Effect of lamotrigine on Na(v)1.4 voltage-gated sodium channels. J Pharmacol Sci. 2013;123(2):203-206. doi:10.1254/jphs.13116sc
71. Ramadan E, Basselin M, Rao JS, et al. Lamotrigine blocks NMDA receptor-initiated arachidonic acid signalling in rat brain: implications for its efficacy in bipolar disorder. Int J Neuropsychopharmacol. 2012;15(7):931-943. doi:10.1017/S1461145711001003
72. Jo S, Bean BP. Sidedness of carbamazepine accessibility to voltage-gated sodium channels. Mol Pharmacol. 2014;85(2):381-387. doi:10.1124/mol.113.090472
73. Curtin F, Schulz P. Clonazepam and lorazepam in acute mania: a Bayesian meta-analysis. J Affect Disord 2004;78(3):201-208. doi:10.1016/S0165-0327(02)00317-8
74. Edwards R, Stephenson U, Flewett T. Clonazepam in acute mania: a double blind trial. Aust N Z J Psychiatry 1991;25(2):238-242. doi:10.3109/00048679109077740
75. Lin SC, Chen CC, Chen YH, et al. Benzodiazepine prescription among patients with severe mental illness and co-occurring alcohol abuse/dependence in Taiwan. Hum Psychopharmacol. 2011;26(3):201-207. doi:10.1002/hup.1193
76. Prisciandaro JJ, Brown DG, Brady KT, et al. Comorbid anxiety disorders and baseline medication regimens predict clinical outcomes in individuals with co-occurring bipolar disorder and alcohol dependence: results of a randomized controlled trial. Psychiatry Res. 2011;188(3):361-365. doi:10.1016/j.psychres.2011.04.030
77. Ashok AH, Marques TR, Jauhar S, et al. The dopamine hypothesis of bipolar affective disorder: the state of the art and implications for treatment. Mol Psychiatry. 2017;22(5):666-679. doi:10.1038/mp.2017.16
78. Roberts RJ, Lohano KK, El-Mallakh RS. Antipsychotics as antidepressants. Asia Pac Psychiatry. 2016;8(3):179-188. doi:10.1111/appy.12186
79. Cipriani A, Barbui C, Salanti G, et al. Comparative efficacy and acceptability of antimanic drugs in acute mania: a multiple-treatments meta-analysis. Lancet. 2011;378(9799):1306-1315. doi:10.1016/S0140-6736(11)60873-8
80. Hayes JF, Marston L, Walters K, et al. Lithium vs. valproate vs. olanzapine vs. quetiapine as maintenance monotherapy for bipolar disorder: a population-based UK cohort study using electronic health records. World Psychiatry. 2016;15(1):53-58. doi:10.1002/wps.20298
81. Geddes JR, Gardiner A, Rendell J, et al. Comparative evaluation of quetiapine plus lamotrigine combination versus quetiapine monotherapy (and folic acid versus placebo) in bipolar depression (CEQUEL): a 2 × 2 factorial randomised trial. Lancet Psychiatry. 2016;3(1):31239. doi:10.1016/S2215-0366(15)00450-2
82. Goodwin GM, Haddad PM, Ferrier IN, et al. Evidence-based guidelines for treating bipolar disorder: revised third edition recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2016;30(6):495-553. doi:10.1177/0269881116636545
Bipolar disorder (BD) is a recurrent, life-long psychiatric illness affecting nearly 2% of the world population1,2 that is characterized by episodes of mania and depression interspersed among periods of relative mood stability.3 The illness causes an enormous health burden, which makes understanding its pathophysiology and treatment patterns a substantial priority.4 In the 1950s, lithium was found to be effective for treating acute manic episodes and preventing relapse in BD.5 Since then, valproate and carbamazepine also have been FDA-approved for treating mania.6,7 Antipsychotics have also shown evidence of efficacy in BD treatment,8,9 particularly for use in acute settings for more rapid effect or for a limited duration,10 which has led some to refer to them as “mood stabilizers.”11
In this article, we describe changes in trends of prescribing medications to treat BD, the role of ion dysregulation in the disorder, and how a better understanding of this dysregulation might impact the choice of treatment.
Changes in pharmacotherapy for bipolar disorder
From 1997 through 2016, the use of lithium for BD decreased from >30% of patients to 17.6% (with a nadir of 13.9% from 2009 to 2012).12 Over the same period, the use of nonlithium mood stabilizers decreased from 30.4% to approximately 4.8%, while second-generation antipsychotic (SGAs) use increased from 12.4% to 50.4%.12 Distressingly, antidepressant use increased from approximately 47% to 56.8%, and antidepressant use without concomitant mood stabilizers increased from 38% to 40.8%, although the rate of antidepressants without either a mood stabilizer or an antipsychotic remained relatively stable (14.9% to 16.8%).12 In randomized trials, when added to mood stabilizers, antidepressants have consistently failed to separate from placebo,13-15 but they can destabilize the illness, resulting in increases in mania, depression, and subsyndromal mixed symptoms.16-18
It is easy to understand clinicians’ attempts to address their patients’ distress due to depressive symptoms that do not resolve with mood stabilizers.19,20 Similarly, the increased use of antipsychotics is driven by evidence that antipsychotics are effective for treating bipolar depression and preventing the recurrence of manic and (for some antipsychotics) depressive episodes.21,22 However, long-term antipsychotic use causes brain volume change in patients with schizophrenia23 or major depressive disorder24 and in nonhuman primates25,26; metabolic abnormalities27-31; and cardiovascular adverse effects.32 Antipsychotics are believed to be associated with withdrawal psychosis.33,34 In the head-to-head Clinical Health Outcomes Initiative in Comparative Effectiveness for Bipolar Disorder (Bipolar CHOICE) study, quetiapine was as effective as lithium but associated with more adverse effects.35 Importantly, the estimated disability-adjusted life years of patients with BD increased by 54.4% from 6.02 million in 1990 to 9.29 million in 2017, which is greater than the increase in the incidence of BD (47.74%) over the same time.36 This means that despite the dramatic increase in treatment options for people with BD, functional outcomes have declined.
One major difference between antipsychotics and mood stabilizers is that antipsychotics do not alter the underlying abnormal pathology of BD.37 An ideal pharmacologic intervention is one that corrects a known pathophysiologic anomaly of the condition being treated. There are no demonstrated abnormalities in the dopamine or serotonin systems in individuals with BD, but long-term use of antipsychotics may create dopaminergic alterations.33 One of the most reproducible biomarkers associated with manic and bipolar depressed mood states is increased intracellular sodium38,39 and reduced ability to correct a sodium challenge.40-42 By normalizing intracellular sodium levels, lithium and the mood-stabilizing anticonvulsants uniquely and specifically counter known physiologic abnormalities in patients with BD.37,43
The role of ion dysregulation
The pathophysiology of BD remains elusive. A multitude of lines of evidence link BD to abnormal neuroimaging findings,22,44,45 oxidative stress,46 inflammation,47 and mitochondrial disease,48 but there is still no unifying understanding of these findings. Ion dysregulation appears to be central to understanding and treating BD.38,39
Despite extensive genetic studies, no genes have been identified that mediate >5% of the risk for BD. Nonetheless, 74% of all genes identified as mediating risk for BD code for proteins essential for the regulation of ion transport and membrane potential.49 The 2 genes that contribute the greatest risk are CACNA1C and ANK3, which code for a calcium channel and a cytoskeletal protein, respectively.50 ANK3 codes for ankyrin G, which plays a role in proper coupling of the voltage-gated sodium channels to the cytoskeleton.51 An additional risk gene, TRANK1, contains multiple ankyrin-like repeat domains, which suggests some shared functions with ANK3.52 More importantly, the most reproducible pathophysiologic findings in BD are dysregulation of sodium, potassium, hydrogen, and calcium transport, with consequent alteration of depolarization potential, neuronal excitability, and calcium-mediated processes.38,39,53-56 For example, increased sodium and calcium within cells have been observed in both mania and bipolar depression, and these levels normalize during euthymia. All medications that are effective for treating BD may reduce intracellular sodium or calcium; traditional mood stabilizers do so directly by inhibiting voltage-sensitive sodium channels in an activity-dependent manner or displacing intracellular sodium,43,57 whereas antipsychotics do so indirectly by increasing sodium pump activity through inhibition of second messengers of the dopamine D2 family of receptors.37
Continue to: The extent of ion dysregulation...
The extent of ion dysregulation is directly associated with the expressed mood state of the illness. A small reduction in the activity of the sodium pump results in a small increase in intracellular sodium (approximately 10 mM).39,58 This led to the hypothesis that increased intracellular sodium causes the transmembrane potential to increase closer to membrane depolarization threshold, which increases excitability of affected neurons.38,39,58 Neurons are likely to fire and propagate signals more easily, which may manifest as symptoms of mania, such as increased energy, activity, lability, excitability, irritability, tangentiality, and looseness of associations. As the process of increased intracellular sodium progresses, a minority of neurons are expected to have their transmembrane potentials depolarize sufficiently for the resting membrane potential to go beyond threshold potential.59 Such neurons are in a state of constant depolarization (also known as depolarization block), which disrupts neuronal circuits. The difficulty in progression of these signals results in the classic bipolar depression symptoms of low energy, reduced activity, and slowing of all brain activity that is seen as psychomotor slowing.38
Implications for treatment
Medications for treating bipolar illness include lithium, anticonvulsants, benzodiazepines, first-generation antipsychotics, and SGAs.37,43
Mood stabilizers (lithium and certain anticonvulsants) correct the previously mentioned sodium abnormality by reducing sodium entry into the cell in an activity-dependent manner.43 As the only agents that directly address a known pathophysiologic abnormality, they are foundational in the treatment of BD.60 Lithium effectively treats acute mania and prevents relapse.61 It preferentially targets the active neurons, entering through both voltage-responsive and neurotransmitter-coupled channels.43,62 This results in an increase of intracellular lithium concentrations to as much as 8 times that of the extracellular concentration.63 These ions displace intracellular sodium ions in a 1:1 ratio, which results in a reduced intracellular sodium concentration that reduces the excitability of neurons.43,57,62
Substantial evidence supports the use of valproic acid for initial and maintenance treatment of BD.64 It inhibits the voltage-sensitive sodium channel when the channel is open, which results in an activity-dependent action that selectively impacts rapidly firing neurons.43 The voltage-gated sodium channels exist nearly exclusively on the axon, beyond the hillock65; as such, valproic acid will only inhibit neurons that fire, whereas lithium accumulates throughout the neuron and will affect depolarization in the neuronal soma as well as the firing in the axon.43 Additionally, valproic acid has been observed to enhance gamma-aminobutyric acid (GABA) levels and transmission.43,66,67 A meta-analysis that included 6 randomized controlled trials illustrated that, acutely, valproate was not different from lithium’s overall efficacy (RR 1.02; 95% CI, 0.87 to 1.20), but was associated with reduced dropout rates compared with placebo or lithium (RR 0.82; 95% CI, 0.71 to 0.95 and RR 0.87; 95% CI, 0.77 to 0.98, respectively).64
Lamotrigine is an anticonvulsant used for initial and maintenance treatment of BD, with greater efficacy for depressive episodes68; it also has notable effect for treating bipolar depression, although it is not FDA-approved for this indication.69 Lamotrigine inhibits sodium influx by binding to open voltage-gated sodium channels70 but also appears to reduce N-methyl-D-aspartate–mediated sodium entry,71 thereby acting both prehillock and posthillock.
Continue to: Carbamazepine is an anticonvulsant...
Carbamazepine is an anticonvulsant FDA-approved for treating BD.7 Like valproate, it acts by inhibiting voltage-gated sodium channels in an activity-dependent manner,72 which means it preferentially inhibits the most active neurons and those with higher intracellular sodium.43
Benzodiazepines, which have shown to be effective for treating acute mania,73 potentiate synaptic GABA receptors accruing an elevation in intracellular chloride influx.74 Despite acute efficacy, benzodiazepine use is limited because these agents are associated with worsening long-term, substance use–related outcomes.75,76
Antipsychotics are effective for treating mood disorders,60,76 and their use has been rising dramatically.12 The antimanic effect of all antipsychotics is believed to be mediated through dopamine D2 blockade, since use of a dose sufficient to block D2 receptors is required, and haloperidol, which acts exclusively on the D2 receptor, is equal to SGAs in its antimanic effect.77 Blockade of the D2 receptor will increase the activity of the sodium pump (sodium and potassium-activated adenosine triphosphatase) thus reducing intracellular sodium and calcium concentrations.37 When antipsychotics are used as antidepressants, they are generally used at doses lower than those used to treat mania.78
Antipsychotics are effective for treating BD, and may work more quickly than other agents for treating acute mania.79 However, maintenance or prevention trials tend to favor mood stabilizers.35,60,80 Several add-on studies have found the combination of a mood stabilizer plus an antipsychotic is superior to a mood stabilizer alone or an antipsychotic alone.81
An argument for mood stabilizers
Evidence suggests mood stabilizers and other approaches, such as antipsychotics, are almost equivalent for treating acute mania, with a small clinical advantage of mood stabilizers for preventing relapse. In general, current treatment guidelines do not distinguish mood stabilizers from antipsychotics as the first-line treatment.82 Over the past 20 years, antipsychotic use has increased while mood stabilizer use has decreased, so that presently a patient with BD is more likely to be prescribed an antipsychotic than a mood stabilizer.12 Over the same time, dysfunction among patients with BD has increased.33 Antipsychotics are appealing because they appear to be equally effective and generally well tolerated. But these agents cause problems that are difficult to see in routine visits, such as metabolic27-31 and cardiovascular adverse effects29 as well as reductions in brain volume.23-26 Mechanistic research suggests that mood stabilizers directly correct known pathophysiologic anomalies with additional protective effects, whereas antipsychotics appear to create new abnormalities and contribute to medical problems. Clinicians need to look beyond the similarities in acute efficacy and make a more broadly supported, evidence-based choice for managing BD, which clearly places mood stabilizers as the first-line agent and antipsychotics as reasonable alternatives. At a minimum, mood stabilizers should be viewed as the foundation to which antipsychotics can be added.
Bottom Line
Traditional mood stabilizers—lithium and some anticonvulsants—are the only agents that directly address physiologic abnormalities associated with both mania and bipolar depression, including mood state–associated elevations of intracellular sodium. Because of their specificity, these agents maximize mood stabilization and minimize adverse effects.
Related Resources
- Karas A, Stummer L, Freedberg A. Psychiatric and nonpsychiatric indications for mood stabilizers and select antiepileptics. Current Psychiatry. 2022;21(4):34-38. doi:10.12788/cp.0230
- Koch J. Mood stabilizers: balancing tolerability, serum levels, and dosage. Current Psychiatry. 2021;20(7):37-40. doi:10.12788/cp.0147
Drug Brand Names
Carbamazepine • Tegretol
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Quetiapine • Seroquel
Valproate • Depakote, Depakene
Bipolar disorder (BD) is a recurrent, life-long psychiatric illness affecting nearly 2% of the world population1,2 that is characterized by episodes of mania and depression interspersed among periods of relative mood stability.3 The illness causes an enormous health burden, which makes understanding its pathophysiology and treatment patterns a substantial priority.4 In the 1950s, lithium was found to be effective for treating acute manic episodes and preventing relapse in BD.5 Since then, valproate and carbamazepine also have been FDA-approved for treating mania.6,7 Antipsychotics have also shown evidence of efficacy in BD treatment,8,9 particularly for use in acute settings for more rapid effect or for a limited duration,10 which has led some to refer to them as “mood stabilizers.”11
In this article, we describe changes in trends of prescribing medications to treat BD, the role of ion dysregulation in the disorder, and how a better understanding of this dysregulation might impact the choice of treatment.
Changes in pharmacotherapy for bipolar disorder
From 1997 through 2016, the use of lithium for BD decreased from >30% of patients to 17.6% (with a nadir of 13.9% from 2009 to 2012).12 Over the same period, the use of nonlithium mood stabilizers decreased from 30.4% to approximately 4.8%, while second-generation antipsychotic (SGAs) use increased from 12.4% to 50.4%.12 Distressingly, antidepressant use increased from approximately 47% to 56.8%, and antidepressant use without concomitant mood stabilizers increased from 38% to 40.8%, although the rate of antidepressants without either a mood stabilizer or an antipsychotic remained relatively stable (14.9% to 16.8%).12 In randomized trials, when added to mood stabilizers, antidepressants have consistently failed to separate from placebo,13-15 but they can destabilize the illness, resulting in increases in mania, depression, and subsyndromal mixed symptoms.16-18
It is easy to understand clinicians’ attempts to address their patients’ distress due to depressive symptoms that do not resolve with mood stabilizers.19,20 Similarly, the increased use of antipsychotics is driven by evidence that antipsychotics are effective for treating bipolar depression and preventing the recurrence of manic and (for some antipsychotics) depressive episodes.21,22 However, long-term antipsychotic use causes brain volume change in patients with schizophrenia23 or major depressive disorder24 and in nonhuman primates25,26; metabolic abnormalities27-31; and cardiovascular adverse effects.32 Antipsychotics are believed to be associated with withdrawal psychosis.33,34 In the head-to-head Clinical Health Outcomes Initiative in Comparative Effectiveness for Bipolar Disorder (Bipolar CHOICE) study, quetiapine was as effective as lithium but associated with more adverse effects.35 Importantly, the estimated disability-adjusted life years of patients with BD increased by 54.4% from 6.02 million in 1990 to 9.29 million in 2017, which is greater than the increase in the incidence of BD (47.74%) over the same time.36 This means that despite the dramatic increase in treatment options for people with BD, functional outcomes have declined.
One major difference between antipsychotics and mood stabilizers is that antipsychotics do not alter the underlying abnormal pathology of BD.37 An ideal pharmacologic intervention is one that corrects a known pathophysiologic anomaly of the condition being treated. There are no demonstrated abnormalities in the dopamine or serotonin systems in individuals with BD, but long-term use of antipsychotics may create dopaminergic alterations.33 One of the most reproducible biomarkers associated with manic and bipolar depressed mood states is increased intracellular sodium38,39 and reduced ability to correct a sodium challenge.40-42 By normalizing intracellular sodium levels, lithium and the mood-stabilizing anticonvulsants uniquely and specifically counter known physiologic abnormalities in patients with BD.37,43
The role of ion dysregulation
The pathophysiology of BD remains elusive. A multitude of lines of evidence link BD to abnormal neuroimaging findings,22,44,45 oxidative stress,46 inflammation,47 and mitochondrial disease,48 but there is still no unifying understanding of these findings. Ion dysregulation appears to be central to understanding and treating BD.38,39
Despite extensive genetic studies, no genes have been identified that mediate >5% of the risk for BD. Nonetheless, 74% of all genes identified as mediating risk for BD code for proteins essential for the regulation of ion transport and membrane potential.49 The 2 genes that contribute the greatest risk are CACNA1C and ANK3, which code for a calcium channel and a cytoskeletal protein, respectively.50 ANK3 codes for ankyrin G, which plays a role in proper coupling of the voltage-gated sodium channels to the cytoskeleton.51 An additional risk gene, TRANK1, contains multiple ankyrin-like repeat domains, which suggests some shared functions with ANK3.52 More importantly, the most reproducible pathophysiologic findings in BD are dysregulation of sodium, potassium, hydrogen, and calcium transport, with consequent alteration of depolarization potential, neuronal excitability, and calcium-mediated processes.38,39,53-56 For example, increased sodium and calcium within cells have been observed in both mania and bipolar depression, and these levels normalize during euthymia. All medications that are effective for treating BD may reduce intracellular sodium or calcium; traditional mood stabilizers do so directly by inhibiting voltage-sensitive sodium channels in an activity-dependent manner or displacing intracellular sodium,43,57 whereas antipsychotics do so indirectly by increasing sodium pump activity through inhibition of second messengers of the dopamine D2 family of receptors.37
Continue to: The extent of ion dysregulation...
The extent of ion dysregulation is directly associated with the expressed mood state of the illness. A small reduction in the activity of the sodium pump results in a small increase in intracellular sodium (approximately 10 mM).39,58 This led to the hypothesis that increased intracellular sodium causes the transmembrane potential to increase closer to membrane depolarization threshold, which increases excitability of affected neurons.38,39,58 Neurons are likely to fire and propagate signals more easily, which may manifest as symptoms of mania, such as increased energy, activity, lability, excitability, irritability, tangentiality, and looseness of associations. As the process of increased intracellular sodium progresses, a minority of neurons are expected to have their transmembrane potentials depolarize sufficiently for the resting membrane potential to go beyond threshold potential.59 Such neurons are in a state of constant depolarization (also known as depolarization block), which disrupts neuronal circuits. The difficulty in progression of these signals results in the classic bipolar depression symptoms of low energy, reduced activity, and slowing of all brain activity that is seen as psychomotor slowing.38
Implications for treatment
Medications for treating bipolar illness include lithium, anticonvulsants, benzodiazepines, first-generation antipsychotics, and SGAs.37,43
Mood stabilizers (lithium and certain anticonvulsants) correct the previously mentioned sodium abnormality by reducing sodium entry into the cell in an activity-dependent manner.43 As the only agents that directly address a known pathophysiologic abnormality, they are foundational in the treatment of BD.60 Lithium effectively treats acute mania and prevents relapse.61 It preferentially targets the active neurons, entering through both voltage-responsive and neurotransmitter-coupled channels.43,62 This results in an increase of intracellular lithium concentrations to as much as 8 times that of the extracellular concentration.63 These ions displace intracellular sodium ions in a 1:1 ratio, which results in a reduced intracellular sodium concentration that reduces the excitability of neurons.43,57,62
Substantial evidence supports the use of valproic acid for initial and maintenance treatment of BD.64 It inhibits the voltage-sensitive sodium channel when the channel is open, which results in an activity-dependent action that selectively impacts rapidly firing neurons.43 The voltage-gated sodium channels exist nearly exclusively on the axon, beyond the hillock65; as such, valproic acid will only inhibit neurons that fire, whereas lithium accumulates throughout the neuron and will affect depolarization in the neuronal soma as well as the firing in the axon.43 Additionally, valproic acid has been observed to enhance gamma-aminobutyric acid (GABA) levels and transmission.43,66,67 A meta-analysis that included 6 randomized controlled trials illustrated that, acutely, valproate was not different from lithium’s overall efficacy (RR 1.02; 95% CI, 0.87 to 1.20), but was associated with reduced dropout rates compared with placebo or lithium (RR 0.82; 95% CI, 0.71 to 0.95 and RR 0.87; 95% CI, 0.77 to 0.98, respectively).64
Lamotrigine is an anticonvulsant used for initial and maintenance treatment of BD, with greater efficacy for depressive episodes68; it also has notable effect for treating bipolar depression, although it is not FDA-approved for this indication.69 Lamotrigine inhibits sodium influx by binding to open voltage-gated sodium channels70 but also appears to reduce N-methyl-D-aspartate–mediated sodium entry,71 thereby acting both prehillock and posthillock.
Continue to: Carbamazepine is an anticonvulsant...
Carbamazepine is an anticonvulsant FDA-approved for treating BD.7 Like valproate, it acts by inhibiting voltage-gated sodium channels in an activity-dependent manner,72 which means it preferentially inhibits the most active neurons and those with higher intracellular sodium.43
Benzodiazepines, which have shown to be effective for treating acute mania,73 potentiate synaptic GABA receptors accruing an elevation in intracellular chloride influx.74 Despite acute efficacy, benzodiazepine use is limited because these agents are associated with worsening long-term, substance use–related outcomes.75,76
Antipsychotics are effective for treating mood disorders,60,76 and their use has been rising dramatically.12 The antimanic effect of all antipsychotics is believed to be mediated through dopamine D2 blockade, since use of a dose sufficient to block D2 receptors is required, and haloperidol, which acts exclusively on the D2 receptor, is equal to SGAs in its antimanic effect.77 Blockade of the D2 receptor will increase the activity of the sodium pump (sodium and potassium-activated adenosine triphosphatase) thus reducing intracellular sodium and calcium concentrations.37 When antipsychotics are used as antidepressants, they are generally used at doses lower than those used to treat mania.78
Antipsychotics are effective for treating BD, and may work more quickly than other agents for treating acute mania.79 However, maintenance or prevention trials tend to favor mood stabilizers.35,60,80 Several add-on studies have found the combination of a mood stabilizer plus an antipsychotic is superior to a mood stabilizer alone or an antipsychotic alone.81
An argument for mood stabilizers
Evidence suggests mood stabilizers and other approaches, such as antipsychotics, are almost equivalent for treating acute mania, with a small clinical advantage of mood stabilizers for preventing relapse. In general, current treatment guidelines do not distinguish mood stabilizers from antipsychotics as the first-line treatment.82 Over the past 20 years, antipsychotic use has increased while mood stabilizer use has decreased, so that presently a patient with BD is more likely to be prescribed an antipsychotic than a mood stabilizer.12 Over the same time, dysfunction among patients with BD has increased.33 Antipsychotics are appealing because they appear to be equally effective and generally well tolerated. But these agents cause problems that are difficult to see in routine visits, such as metabolic27-31 and cardiovascular adverse effects29 as well as reductions in brain volume.23-26 Mechanistic research suggests that mood stabilizers directly correct known pathophysiologic anomalies with additional protective effects, whereas antipsychotics appear to create new abnormalities and contribute to medical problems. Clinicians need to look beyond the similarities in acute efficacy and make a more broadly supported, evidence-based choice for managing BD, which clearly places mood stabilizers as the first-line agent and antipsychotics as reasonable alternatives. At a minimum, mood stabilizers should be viewed as the foundation to which antipsychotics can be added.
Bottom Line
Traditional mood stabilizers—lithium and some anticonvulsants—are the only agents that directly address physiologic abnormalities associated with both mania and bipolar depression, including mood state–associated elevations of intracellular sodium. Because of their specificity, these agents maximize mood stabilization and minimize adverse effects.
Related Resources
- Karas A, Stummer L, Freedberg A. Psychiatric and nonpsychiatric indications for mood stabilizers and select antiepileptics. Current Psychiatry. 2022;21(4):34-38. doi:10.12788/cp.0230
- Koch J. Mood stabilizers: balancing tolerability, serum levels, and dosage. Current Psychiatry. 2021;20(7):37-40. doi:10.12788/cp.0147
Drug Brand Names
Carbamazepine • Tegretol
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Quetiapine • Seroquel
Valproate • Depakote, Depakene
1. Whiteford HA, Degenhardt L, Rehm J, et al. Global burden of disease attributable to mental and substance use disorders: findings from the Global Burden of Disease Study 2010. Lancet. 2013;382(9904):1575-1586. doi:10.1016/S0140-6736(13)61611-6
2. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the world mental health survey initiative. Arch Gen Psychiatry. 2011;68(3):241-251. doi:10.1001/archgenpsychiatry.2011.12
3. Müller JK, Leweke FM. Bipolar disorder: clinical overview. Article in English, German. Med Monatsschr Pharm. 2016;39(9):363-369.
4. Smith DJ, Whitham EA, Ghaemi SN. Bipolar disorder. Handb Clin Neurol. 2012;106:251-263. doi:10.1016/B978-0-444-52002-9.00015-2
5. Goodwin FK, Ghaemi SN. The impact of the discovery of lithium on psychiatric thought and practice in the USA and Europe. Aust N Z J Psychiatry. 1999;33 Suppl:S54-S64. doi:10.1111/j.1440-1614.1999.00669.x
6. Pope HG, McElroy SL, Keck PE, et al. Valproate in the treatment of acute mania. A placebo-controlled study. Arch Gen Psychiatry. 1991;48(1):62-68. doi:10.1001/archpsyc.1991.01810250064008
7. Weisler RH, Keck PE Jr, Swann AC, et al. Extended-release carbamazepine capsules as monotherapy for acute mania in bipolar disorder: a multicenter, randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2005;66(3):323-330. doi:10.4088/jcp.v66n0308
8. Tarr GP, Glue P, Herbison P. Comparative efficacy and acceptability of mood stabilizer and second generation antipsychotic monotherapy for acute mania--a systematic review and meta-analysis. J Affect Disord. 2011;134(1-3):14-19. doi:10.1016/j.jad.2010.11.009
9. Pahwa M, Sleem A, Elsayed OH, et al. New antipsychotic medications in the last decade. Curr Psychiatry Rep. 2021;23(12):87.
10. Correll CU, Sheridan EM, DelBello MP. Antipsychotic and mood stabilizer efficacy and tolerability in pediatric and adult patients with bipolar I mania: a comparative analysis of acute, randomized, placebo-controlled trials. Bipolar Disord. 2010;12(2):116-141. doi:10.1111/j.1399-5618.2010.00798.x
11. Rybakowski JK. Two generations of mood stabilizers. Int J Neuropsychopharmacol. 2007;10:709-711. doi:10.1017/s146114570700795x
12. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715. doi:10.1176/appi.ajp.2020.19091000
13. El-Mallakh RS. Adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;357(6):615; author reply 615-616.
14. Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;356(17):1711-1722. doi:10.1056/NEJMoa064135
15. Ghaemi SN, Whitham EA, Vohringer PA, et al. Citalopram for acute and preventive efficacy in bipolar depression (CAPE-BD): a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2021;82(1):19m13136. doi:10.4088/JCP.19m13136
16. El-Mallakh RS, Ghaemi SN, Sagduyu K, et al. Antidepressant-associated chronic irritable dysphoria (ACID) in STEP-BD patients. J Affect Disord. 2008;111(2-3):372-377. doi:10.1016/j.jad.2008.03.025
17. Ghaemi SN, Ostacher MM, El-Mallakh RS, et al. Antidepressant discontinuation in bipolar depression: a systematic treatment enhancement program for bipolar disorder (STEP-BD) randomized clinical trial of long-term effectiveness and safety. J Clin Psychiatry. 2010;71(4):372-380.
18. Strejilevich SA, Martino DJ, Marengo E, et al. Long-term worsening of bipolar disorder related with frequency of antidepressant exposure. Ann Clin Psychiatry. 2011;23(3):186-192.
19. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society of Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262. doi:10.1176/appi.ajp.2013.13020185
20. McIntyre RS, Calabrese JR. Bipolar depression: the clinical characteristics and unmet needs of a complex disorder. Curr Med Res Opin. 2019;35(11):1993-2005.
21. Fornaro M, Stubbs B, De Berardis D, et al. Atypical antipsychotics in the treatment of acute bipolar depression with mixed features: a systematic review and exploratory meta-analysis of placebo-controlled clinical trials. Int J Mol Sci. 2016;17(2):241. doi:10.3390/ijms17020241
22. Lindström L, Lindström E, Nilsson M, et al. Maintenance therapy with second generation antipsychotics for bipolar disorder – a systematic review and meta-analysis. J Affect Disord. 2017;213:138-150. doi:10.1016/j.jad.2017.02.012
23. Ho BC, Andreasen NC, Ziebell S, et al. Long-term antipsychotic treatment and brain volumes: a longitudinal study of first-episode schizophrenia. Arch Gen Psychiatry. 2011;68(2):128-137. doi:010.1001/archgenpsychiatry.2010.199
24. Voineskos AN, Mulsant BH, Dickie EW, et al. Effects of antipsychotic medication on brain structure in patients with major depressive disorder and psychotic features: neuroimaging findings in the context of a randomized placebo-controlled clinical trial. JAMA Psychiatry. 2020;77(7):674-683. doi:10.1001/jamapsychiatry.2020.0036
25. Konopaske GT, Bolo NR, Basu AC, et al. Time-dependent effects of haloperidol on glutamine and GABA homeostasis and astrocyte activity in the rat brain. Psychopharmacology (Berl). 2013;230(1):57-67. doi:10.1007/s00213-013-3136-3
26. Dorph-Petersen KA, Pierri JN, Perel JM, et al. The influence of chronic exposure to antipsychotic medications on brain size before and after tissue fixation: a comparison of haloperidol and olanzapine in macaque monkeys. Neuropsychopharmacology. 2005;30(9):1649-1661. doi:10.1038/sj.npp.1300710
27. McIntyre RS, Mancini DA, Basile VS, et al. Antipsychotic-induced weight gain: bipolar disorder and leptin. J Clin Psychopharmacol. 2003;23(4):323-327. doi:10.1097/01.jcp.0000085403.08426.f4
28. McIntyre RS, Konarski JZ, Wilkins K, et al. Obesity in bipolar disorder and major depressive disorder: results from a national community health survey on mental health and well-being. Can J Psychiatry. 2006;51(5):274-280. doi:10.1177/070674370605100502
29. McIntyre RS, Cha DS, Kim RD, et al. A review of FDA-approved treatment options in bipolar depression. CNS Spectr. 2013;18(Suppl 1):4-20. doi:10.1017/S1092852913000746
30. Barton BB, Segger F, Fischer K, et al. Update on weight-gain caused by antipsychotics: a systematic review and meta-analysis. Expert Opin Drug Saf. 2020;19(3):295-314. doi:10.1080/14740338.2020.1713091
31. Doane MJ, Bessonova L, Friedler HS, et al. Weight gain and comorbidities associated with oral second-generation antipsychotics: analysis of real-world data for patients with schizophrenia or bipolar I disorder. BMC Psychiatry. 2022;22(1):114. doi:10.1186/s12888-022-03758-w
32. Buckley NA, Sanders P. Cardiovascular adverse effects of antipsychotic drugs. Drug Saf. 2000;23(3):215-228. doi:10.2165/00002018-200023030-00004
33. Ali Z, Roque A, El-Mallakh RS. A unifying theory for the pathoetiologic mechanism of tardive dyskinesia. Med Hypotheses. 2020;140:109682. doi:10.1016/j.mehy.2020.109682
34. Sleem A, El-Mallakh RS. Adaptive changes to antipsychotics: their consequences and how to avoid them. Curr Psychiatry. 2022;21(7):46-50,52. doi: 10.12788/cp.0262
35. Nierenberg AA, McElroy SL, Friedman ES, et al. Bipolar CHOICE (Clinical Health Outcomes Initiative in Comparative Effectiveness): a pragmatic 6-month trial of lithium versus quetiapine for bipolar disorder. J Clin Psychiatry. 2016;77(1):90-99. doi:10.4088/JCP.14m09349
36. He H, Hu C, Ren Z, et al. Trends in the incidence and DALYs of bipolar disorder at global, regional, and national levels: results from the global burden of disease study 2017. J Psychiatr Res. 2020;125:96-105. doi:10.1016/j.jpsychires.2020.03.015
37. Roberts RJ, Repass R, El-Mallakh RS. Effect of dopamine on intracellular sodium: a common pathway for pharmacological mechanism of action in bipolar illness. World J Biol Psychiatry. 2010;11(2 Pt 2):181-187. doi:10.1080/15622970902718774
38. El-Mallakh RS, Wyatt RJ. The Na, K-ATPase hypothesis for bipolar illness. Biol Psychiatry. 1995;37(4):235-244. doi:10.1016/0006-3223(94)00201-D
39. El-Mallakh RS, Yff T, Gao Y. Ion dysregulation in the pathogenesis of bipolar disorder. Ann Depress Anxiety. 2016;3(1):1076.
40. Li R, El-Mallakh RS. Differential response of bipolar and normal control lymphoblastoid cell sodium pump to ethacrynic acid. J Affect Disord. 2004;80(1):11-17. doi:10.1016/S0165-0327(03)00044-2
41. Woodruff DB, El-Mallakh RS, Thiruvengadam AP. Validation of a diagnostic screening blood test for bipolar disorder. Ann Clin Psychiatry. 2012;24(2):135-139.
42. Gao Y, Lohano K, Delamere NA, et al. Ethanol normalizes glutamate-induced elevation of intracellular sodium in olfactory neuroepithelial progenitors from subjects with bipolar illness but not nonbipolar controls: biologic evidence for the self-medication hypothesis. Bipolar Disord. 2019;21(2):179-181. doi:10.1111/bdi.12737
43. El-Mallakh RS, Huff MO. Mood stabilizers and ion regulation. Harv Rev Psychiatry. 2001;9(1):23-32. doi:10.1080/10673220127873
44. Phillips ML, Swartz HA. A critical appraisal of neuroimaging studies of bipolar disorder: toward a new conceptualization of underlying neural circuitry and a road map for future research. Am J Psychiatry. 2014;171(8):829-843. doi:10.1176/appi.ajp.2014.13081008
45. Hibar DP, Westlye LT, Doan NT, et al. Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA Bipolar Disorder Working Group. Mol Psychiatry. 2018;23(4):932-942. doi:10.1038/mp.2017.73
46. Brown NC, Andreazza AC, Young LT. An updated meta-analysis of oxidative stress markers in bipolar disorder. Psychiatry Res. 2014;218(1-2):61-68. doi:10.1016/j.psychres.2014.04.005
47. Benedetti F, Aggio V, Pratesi ML, et al. Neuroinflammation in bipolar depression. Front Psychiatry. 2020;11:71. doi:10.3389/fpsyt.2020.00071
48. Andreazza AC, Duong A, Young LT. Bipolar disorder as a mitochondrial disease. Biol Psychiatry. 2018;83(9):720-721. doi:10.1016/j.biopsych.2017.09.018
49. Askland KD. Toward a biaxial model of “bipolar” affective disorders: further exploration of genetic, molecular and cellular substrates. J Affect Disord. 2006;94(1-3):35-66. doi:10.1016/j.jad.2006.01.033
50. Ferreira MA, O’Donovan MC, Meng YA, et al; Wellcome Trust Case Control Consortium. Collaborative genome-wide association analysis supports a role for ANK3 and CACNA1C in bipolar disorder. Nat Genet 2008;40(9):1056-1058. doi:10.1038/ng.209
51. Salvi AM, Bays JL, Mackin SR, et al. Ankyrin G organizes membrane components to promote coupling of cell mechanics and glucose uptake. Nat Cell Biol. 2021;23(5):457-466. doi:10.1038/s41556-021-00677-y
52. Gargus JJ. Ion channel functional candidate genes in multigenic neuropsychiatric disease. Biol Psychiatry. 2006;60(2):177-185. doi:10.1016/j.biopsych.2005.12.008
53. Dubovsky SL, Murphy J, Thomas M, et al. Abnormal intracellular calcium ion concentration in platelets and lymphocytes of bipolar patients. Am J Psychiatry 1992;149(1):118-120. doi:10.1176/ajp.149.1.118
54. Blaustein MP. Physiological effects of endogenous ouabain: control of intracellular Ca2+ stores and cell responsiveness. Am J Physiol. 1993;264(6 Pt 1):C1367–C1387. doi:10.1152/ajpcell.1993.264.6.C1367
55. El-Mallakh RS, Li R, Worth CA, et al. Leukocyte transmembrane potential in bipolar illness. J Affect Disord. 1996;41(1):33-37. doi:10.1016/0165-0327(96)00063-8
56. El-Mallakh RS, Gao Y, You P. Role of endogenous ouabain in the etiology of bipolar disorder. Int J Bipolar Disord. 2021;9(1):6. doi:10.1186/s40345-020-00213-1
57. Huang X, Lei Z, El‐Mallakh RS. Lithium normalizes elevated intracellular sodium. Bipolar Disord. 2007;9(3):298-300. doi:10.1111/j.1399-5618.2007.00429.x
58. Shaw DM. Mineral metabolism, mania, and melancholia. Br Med J. 1966;2(5508):262-267. doi:10.1136/bmj.2.5508.262
59. Qian K, Yu N, Tucker KR, et al. Mathematical analysis of depolarization block mediated by slow inactivation of fast sodium channels in midbrain dopamine neurons. J Neurophysiol. 2014;112(11):2779-2790. doi:10.1152/jn.00578.2014
60. Sleem A, El-Mallakh RS. Advances in the psychopharmacotherapy of bipolar disorder type I. Exp Opin Pharmacother. 2021;22(10):1267-1290. doi:10.1080/14656566.2021.1893306
61. Malhi GS., Tanious M, Das P, et al. Potential mechanisms of action of lithium in bipolar disorder. CNS Drugs. 2013;27(2):135-153. doi:10.1007/s40263-013-0039-0
62. Armett CJ, Ritchie JM. On the permeability of mammalian non-myelinated fibers to sodium and to lithium ions. J Physiol. 1963;165(1):130-140. doi:10.1113/jphysiol.1963.sp007047
63. Kabakov AY, Karkanias NB, Lenox RH, et al. Synapse-specific accumulation of lithium in intracellular microdomains: a model for uncoupling coincidence detection in the brain. Synapse. 1998;28(4):271-279. doi:10.1002/(SICI)1098-2396(199804)28:4<271::AID-SYN2>3.0.CO;2-6
64. Cipriani A, Reid K, Young AH, et al. Valproic acid, valproate and divalproex in the maintenance treatment of bipolar disorder. Cochrane Database Syst Rev. 2013;2013(10):CD003196. doi:10.1002/14651858.CD003196.pub2
65. Lai HC, Jan LY. The distribution and targeting of neuronal voltage-gated ion channels. Nat Rev Neurosci. 2006;7(7):548-562. doi:10.1038/nrn1938
66. Löscher W, Schmidt D. Increase of human plasma GABA by sodium valproate. Epilepsia. 1980;21(6):611-615. doi:10.1111/j.1528-1157.1980.tb04314.x
67. Owens MJ, Nemeroff CB. Pharmacology of valproate. Psychopharmacol Bull. 2003;37(Suppl 2):17-24.
68. Calabrese JR, Vieta E, Shelton MD. Latest maintenance data on lamotrigine in bipolar disorder. Eur Neuropsychopharmacol. 2003;13(Suppl 2):S57-S66. doi:10.1016/s0924-977x(03)00079-8
69. Geddes JR, Calabrese JR, Goodwin GM. Lamotrigine for treatment of bipolar depression: independent meta-analysis and meta-regression of individual patient data from five randomised trials. Br J Psychiatry. 2009;194(1):4-9. doi:10.1192/bjp.bp.107.048504
70. Nakatani Y, Masuko H, Amano T. Effect of lamotrigine on Na(v)1.4 voltage-gated sodium channels. J Pharmacol Sci. 2013;123(2):203-206. doi:10.1254/jphs.13116sc
71. Ramadan E, Basselin M, Rao JS, et al. Lamotrigine blocks NMDA receptor-initiated arachidonic acid signalling in rat brain: implications for its efficacy in bipolar disorder. Int J Neuropsychopharmacol. 2012;15(7):931-943. doi:10.1017/S1461145711001003
72. Jo S, Bean BP. Sidedness of carbamazepine accessibility to voltage-gated sodium channels. Mol Pharmacol. 2014;85(2):381-387. doi:10.1124/mol.113.090472
73. Curtin F, Schulz P. Clonazepam and lorazepam in acute mania: a Bayesian meta-analysis. J Affect Disord 2004;78(3):201-208. doi:10.1016/S0165-0327(02)00317-8
74. Edwards R, Stephenson U, Flewett T. Clonazepam in acute mania: a double blind trial. Aust N Z J Psychiatry 1991;25(2):238-242. doi:10.3109/00048679109077740
75. Lin SC, Chen CC, Chen YH, et al. Benzodiazepine prescription among patients with severe mental illness and co-occurring alcohol abuse/dependence in Taiwan. Hum Psychopharmacol. 2011;26(3):201-207. doi:10.1002/hup.1193
76. Prisciandaro JJ, Brown DG, Brady KT, et al. Comorbid anxiety disorders and baseline medication regimens predict clinical outcomes in individuals with co-occurring bipolar disorder and alcohol dependence: results of a randomized controlled trial. Psychiatry Res. 2011;188(3):361-365. doi:10.1016/j.psychres.2011.04.030
77. Ashok AH, Marques TR, Jauhar S, et al. The dopamine hypothesis of bipolar affective disorder: the state of the art and implications for treatment. Mol Psychiatry. 2017;22(5):666-679. doi:10.1038/mp.2017.16
78. Roberts RJ, Lohano KK, El-Mallakh RS. Antipsychotics as antidepressants. Asia Pac Psychiatry. 2016;8(3):179-188. doi:10.1111/appy.12186
79. Cipriani A, Barbui C, Salanti G, et al. Comparative efficacy and acceptability of antimanic drugs in acute mania: a multiple-treatments meta-analysis. Lancet. 2011;378(9799):1306-1315. doi:10.1016/S0140-6736(11)60873-8
80. Hayes JF, Marston L, Walters K, et al. Lithium vs. valproate vs. olanzapine vs. quetiapine as maintenance monotherapy for bipolar disorder: a population-based UK cohort study using electronic health records. World Psychiatry. 2016;15(1):53-58. doi:10.1002/wps.20298
81. Geddes JR, Gardiner A, Rendell J, et al. Comparative evaluation of quetiapine plus lamotrigine combination versus quetiapine monotherapy (and folic acid versus placebo) in bipolar depression (CEQUEL): a 2 × 2 factorial randomised trial. Lancet Psychiatry. 2016;3(1):31239. doi:10.1016/S2215-0366(15)00450-2
82. Goodwin GM, Haddad PM, Ferrier IN, et al. Evidence-based guidelines for treating bipolar disorder: revised third edition recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2016;30(6):495-553. doi:10.1177/0269881116636545
1. Whiteford HA, Degenhardt L, Rehm J, et al. Global burden of disease attributable to mental and substance use disorders: findings from the Global Burden of Disease Study 2010. Lancet. 2013;382(9904):1575-1586. doi:10.1016/S0140-6736(13)61611-6
2. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the world mental health survey initiative. Arch Gen Psychiatry. 2011;68(3):241-251. doi:10.1001/archgenpsychiatry.2011.12
3. Müller JK, Leweke FM. Bipolar disorder: clinical overview. Article in English, German. Med Monatsschr Pharm. 2016;39(9):363-369.
4. Smith DJ, Whitham EA, Ghaemi SN. Bipolar disorder. Handb Clin Neurol. 2012;106:251-263. doi:10.1016/B978-0-444-52002-9.00015-2
5. Goodwin FK, Ghaemi SN. The impact of the discovery of lithium on psychiatric thought and practice in the USA and Europe. Aust N Z J Psychiatry. 1999;33 Suppl:S54-S64. doi:10.1111/j.1440-1614.1999.00669.x
6. Pope HG, McElroy SL, Keck PE, et al. Valproate in the treatment of acute mania. A placebo-controlled study. Arch Gen Psychiatry. 1991;48(1):62-68. doi:10.1001/archpsyc.1991.01810250064008
7. Weisler RH, Keck PE Jr, Swann AC, et al. Extended-release carbamazepine capsules as monotherapy for acute mania in bipolar disorder: a multicenter, randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2005;66(3):323-330. doi:10.4088/jcp.v66n0308
8. Tarr GP, Glue P, Herbison P. Comparative efficacy and acceptability of mood stabilizer and second generation antipsychotic monotherapy for acute mania--a systematic review and meta-analysis. J Affect Disord. 2011;134(1-3):14-19. doi:10.1016/j.jad.2010.11.009
9. Pahwa M, Sleem A, Elsayed OH, et al. New antipsychotic medications in the last decade. Curr Psychiatry Rep. 2021;23(12):87.
10. Correll CU, Sheridan EM, DelBello MP. Antipsychotic and mood stabilizer efficacy and tolerability in pediatric and adult patients with bipolar I mania: a comparative analysis of acute, randomized, placebo-controlled trials. Bipolar Disord. 2010;12(2):116-141. doi:10.1111/j.1399-5618.2010.00798.x
11. Rybakowski JK. Two generations of mood stabilizers. Int J Neuropsychopharmacol. 2007;10:709-711. doi:10.1017/s146114570700795x
12. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715. doi:10.1176/appi.ajp.2020.19091000
13. El-Mallakh RS. Adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;357(6):615; author reply 615-616.
14. Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;356(17):1711-1722. doi:10.1056/NEJMoa064135
15. Ghaemi SN, Whitham EA, Vohringer PA, et al. Citalopram for acute and preventive efficacy in bipolar depression (CAPE-BD): a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2021;82(1):19m13136. doi:10.4088/JCP.19m13136
16. El-Mallakh RS, Ghaemi SN, Sagduyu K, et al. Antidepressant-associated chronic irritable dysphoria (ACID) in STEP-BD patients. J Affect Disord. 2008;111(2-3):372-377. doi:10.1016/j.jad.2008.03.025
17. Ghaemi SN, Ostacher MM, El-Mallakh RS, et al. Antidepressant discontinuation in bipolar depression: a systematic treatment enhancement program for bipolar disorder (STEP-BD) randomized clinical trial of long-term effectiveness and safety. J Clin Psychiatry. 2010;71(4):372-380.
18. Strejilevich SA, Martino DJ, Marengo E, et al. Long-term worsening of bipolar disorder related with frequency of antidepressant exposure. Ann Clin Psychiatry. 2011;23(3):186-192.
19. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society of Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262. doi:10.1176/appi.ajp.2013.13020185
20. McIntyre RS, Calabrese JR. Bipolar depression: the clinical characteristics and unmet needs of a complex disorder. Curr Med Res Opin. 2019;35(11):1993-2005.
21. Fornaro M, Stubbs B, De Berardis D, et al. Atypical antipsychotics in the treatment of acute bipolar depression with mixed features: a systematic review and exploratory meta-analysis of placebo-controlled clinical trials. Int J Mol Sci. 2016;17(2):241. doi:10.3390/ijms17020241
22. Lindström L, Lindström E, Nilsson M, et al. Maintenance therapy with second generation antipsychotics for bipolar disorder – a systematic review and meta-analysis. J Affect Disord. 2017;213:138-150. doi:10.1016/j.jad.2017.02.012
23. Ho BC, Andreasen NC, Ziebell S, et al. Long-term antipsychotic treatment and brain volumes: a longitudinal study of first-episode schizophrenia. Arch Gen Psychiatry. 2011;68(2):128-137. doi:010.1001/archgenpsychiatry.2010.199
24. Voineskos AN, Mulsant BH, Dickie EW, et al. Effects of antipsychotic medication on brain structure in patients with major depressive disorder and psychotic features: neuroimaging findings in the context of a randomized placebo-controlled clinical trial. JAMA Psychiatry. 2020;77(7):674-683. doi:10.1001/jamapsychiatry.2020.0036
25. Konopaske GT, Bolo NR, Basu AC, et al. Time-dependent effects of haloperidol on glutamine and GABA homeostasis and astrocyte activity in the rat brain. Psychopharmacology (Berl). 2013;230(1):57-67. doi:10.1007/s00213-013-3136-3
26. Dorph-Petersen KA, Pierri JN, Perel JM, et al. The influence of chronic exposure to antipsychotic medications on brain size before and after tissue fixation: a comparison of haloperidol and olanzapine in macaque monkeys. Neuropsychopharmacology. 2005;30(9):1649-1661. doi:10.1038/sj.npp.1300710
27. McIntyre RS, Mancini DA, Basile VS, et al. Antipsychotic-induced weight gain: bipolar disorder and leptin. J Clin Psychopharmacol. 2003;23(4):323-327. doi:10.1097/01.jcp.0000085403.08426.f4
28. McIntyre RS, Konarski JZ, Wilkins K, et al. Obesity in bipolar disorder and major depressive disorder: results from a national community health survey on mental health and well-being. Can J Psychiatry. 2006;51(5):274-280. doi:10.1177/070674370605100502
29. McIntyre RS, Cha DS, Kim RD, et al. A review of FDA-approved treatment options in bipolar depression. CNS Spectr. 2013;18(Suppl 1):4-20. doi:10.1017/S1092852913000746
30. Barton BB, Segger F, Fischer K, et al. Update on weight-gain caused by antipsychotics: a systematic review and meta-analysis. Expert Opin Drug Saf. 2020;19(3):295-314. doi:10.1080/14740338.2020.1713091
31. Doane MJ, Bessonova L, Friedler HS, et al. Weight gain and comorbidities associated with oral second-generation antipsychotics: analysis of real-world data for patients with schizophrenia or bipolar I disorder. BMC Psychiatry. 2022;22(1):114. doi:10.1186/s12888-022-03758-w
32. Buckley NA, Sanders P. Cardiovascular adverse effects of antipsychotic drugs. Drug Saf. 2000;23(3):215-228. doi:10.2165/00002018-200023030-00004
33. Ali Z, Roque A, El-Mallakh RS. A unifying theory for the pathoetiologic mechanism of tardive dyskinesia. Med Hypotheses. 2020;140:109682. doi:10.1016/j.mehy.2020.109682
34. Sleem A, El-Mallakh RS. Adaptive changes to antipsychotics: their consequences and how to avoid them. Curr Psychiatry. 2022;21(7):46-50,52. doi: 10.12788/cp.0262
35. Nierenberg AA, McElroy SL, Friedman ES, et al. Bipolar CHOICE (Clinical Health Outcomes Initiative in Comparative Effectiveness): a pragmatic 6-month trial of lithium versus quetiapine for bipolar disorder. J Clin Psychiatry. 2016;77(1):90-99. doi:10.4088/JCP.14m09349
36. He H, Hu C, Ren Z, et al. Trends in the incidence and DALYs of bipolar disorder at global, regional, and national levels: results from the global burden of disease study 2017. J Psychiatr Res. 2020;125:96-105. doi:10.1016/j.jpsychires.2020.03.015
37. Roberts RJ, Repass R, El-Mallakh RS. Effect of dopamine on intracellular sodium: a common pathway for pharmacological mechanism of action in bipolar illness. World J Biol Psychiatry. 2010;11(2 Pt 2):181-187. doi:10.1080/15622970902718774
38. El-Mallakh RS, Wyatt RJ. The Na, K-ATPase hypothesis for bipolar illness. Biol Psychiatry. 1995;37(4):235-244. doi:10.1016/0006-3223(94)00201-D
39. El-Mallakh RS, Yff T, Gao Y. Ion dysregulation in the pathogenesis of bipolar disorder. Ann Depress Anxiety. 2016;3(1):1076.
40. Li R, El-Mallakh RS. Differential response of bipolar and normal control lymphoblastoid cell sodium pump to ethacrynic acid. J Affect Disord. 2004;80(1):11-17. doi:10.1016/S0165-0327(03)00044-2
41. Woodruff DB, El-Mallakh RS, Thiruvengadam AP. Validation of a diagnostic screening blood test for bipolar disorder. Ann Clin Psychiatry. 2012;24(2):135-139.
42. Gao Y, Lohano K, Delamere NA, et al. Ethanol normalizes glutamate-induced elevation of intracellular sodium in olfactory neuroepithelial progenitors from subjects with bipolar illness but not nonbipolar controls: biologic evidence for the self-medication hypothesis. Bipolar Disord. 2019;21(2):179-181. doi:10.1111/bdi.12737
43. El-Mallakh RS, Huff MO. Mood stabilizers and ion regulation. Harv Rev Psychiatry. 2001;9(1):23-32. doi:10.1080/10673220127873
44. Phillips ML, Swartz HA. A critical appraisal of neuroimaging studies of bipolar disorder: toward a new conceptualization of underlying neural circuitry and a road map for future research. Am J Psychiatry. 2014;171(8):829-843. doi:10.1176/appi.ajp.2014.13081008
45. Hibar DP, Westlye LT, Doan NT, et al. Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA Bipolar Disorder Working Group. Mol Psychiatry. 2018;23(4):932-942. doi:10.1038/mp.2017.73
46. Brown NC, Andreazza AC, Young LT. An updated meta-analysis of oxidative stress markers in bipolar disorder. Psychiatry Res. 2014;218(1-2):61-68. doi:10.1016/j.psychres.2014.04.005
47. Benedetti F, Aggio V, Pratesi ML, et al. Neuroinflammation in bipolar depression. Front Psychiatry. 2020;11:71. doi:10.3389/fpsyt.2020.00071
48. Andreazza AC, Duong A, Young LT. Bipolar disorder as a mitochondrial disease. Biol Psychiatry. 2018;83(9):720-721. doi:10.1016/j.biopsych.2017.09.018
49. Askland KD. Toward a biaxial model of “bipolar” affective disorders: further exploration of genetic, molecular and cellular substrates. J Affect Disord. 2006;94(1-3):35-66. doi:10.1016/j.jad.2006.01.033
50. Ferreira MA, O’Donovan MC, Meng YA, et al; Wellcome Trust Case Control Consortium. Collaborative genome-wide association analysis supports a role for ANK3 and CACNA1C in bipolar disorder. Nat Genet 2008;40(9):1056-1058. doi:10.1038/ng.209
51. Salvi AM, Bays JL, Mackin SR, et al. Ankyrin G organizes membrane components to promote coupling of cell mechanics and glucose uptake. Nat Cell Biol. 2021;23(5):457-466. doi:10.1038/s41556-021-00677-y
52. Gargus JJ. Ion channel functional candidate genes in multigenic neuropsychiatric disease. Biol Psychiatry. 2006;60(2):177-185. doi:10.1016/j.biopsych.2005.12.008
53. Dubovsky SL, Murphy J, Thomas M, et al. Abnormal intracellular calcium ion concentration in platelets and lymphocytes of bipolar patients. Am J Psychiatry 1992;149(1):118-120. doi:10.1176/ajp.149.1.118
54. Blaustein MP. Physiological effects of endogenous ouabain: control of intracellular Ca2+ stores and cell responsiveness. Am J Physiol. 1993;264(6 Pt 1):C1367–C1387. doi:10.1152/ajpcell.1993.264.6.C1367
55. El-Mallakh RS, Li R, Worth CA, et al. Leukocyte transmembrane potential in bipolar illness. J Affect Disord. 1996;41(1):33-37. doi:10.1016/0165-0327(96)00063-8
56. El-Mallakh RS, Gao Y, You P. Role of endogenous ouabain in the etiology of bipolar disorder. Int J Bipolar Disord. 2021;9(1):6. doi:10.1186/s40345-020-00213-1
57. Huang X, Lei Z, El‐Mallakh RS. Lithium normalizes elevated intracellular sodium. Bipolar Disord. 2007;9(3):298-300. doi:10.1111/j.1399-5618.2007.00429.x
58. Shaw DM. Mineral metabolism, mania, and melancholia. Br Med J. 1966;2(5508):262-267. doi:10.1136/bmj.2.5508.262
59. Qian K, Yu N, Tucker KR, et al. Mathematical analysis of depolarization block mediated by slow inactivation of fast sodium channels in midbrain dopamine neurons. J Neurophysiol. 2014;112(11):2779-2790. doi:10.1152/jn.00578.2014
60. Sleem A, El-Mallakh RS. Advances in the psychopharmacotherapy of bipolar disorder type I. Exp Opin Pharmacother. 2021;22(10):1267-1290. doi:10.1080/14656566.2021.1893306
61. Malhi GS., Tanious M, Das P, et al. Potential mechanisms of action of lithium in bipolar disorder. CNS Drugs. 2013;27(2):135-153. doi:10.1007/s40263-013-0039-0
62. Armett CJ, Ritchie JM. On the permeability of mammalian non-myelinated fibers to sodium and to lithium ions. J Physiol. 1963;165(1):130-140. doi:10.1113/jphysiol.1963.sp007047
63. Kabakov AY, Karkanias NB, Lenox RH, et al. Synapse-specific accumulation of lithium in intracellular microdomains: a model for uncoupling coincidence detection in the brain. Synapse. 1998;28(4):271-279. doi:10.1002/(SICI)1098-2396(199804)28:4<271::AID-SYN2>3.0.CO;2-6
64. Cipriani A, Reid K, Young AH, et al. Valproic acid, valproate and divalproex in the maintenance treatment of bipolar disorder. Cochrane Database Syst Rev. 2013;2013(10):CD003196. doi:10.1002/14651858.CD003196.pub2
65. Lai HC, Jan LY. The distribution and targeting of neuronal voltage-gated ion channels. Nat Rev Neurosci. 2006;7(7):548-562. doi:10.1038/nrn1938
66. Löscher W, Schmidt D. Increase of human plasma GABA by sodium valproate. Epilepsia. 1980;21(6):611-615. doi:10.1111/j.1528-1157.1980.tb04314.x
67. Owens MJ, Nemeroff CB. Pharmacology of valproate. Psychopharmacol Bull. 2003;37(Suppl 2):17-24.
68. Calabrese JR, Vieta E, Shelton MD. Latest maintenance data on lamotrigine in bipolar disorder. Eur Neuropsychopharmacol. 2003;13(Suppl 2):S57-S66. doi:10.1016/s0924-977x(03)00079-8
69. Geddes JR, Calabrese JR, Goodwin GM. Lamotrigine for treatment of bipolar depression: independent meta-analysis and meta-regression of individual patient data from five randomised trials. Br J Psychiatry. 2009;194(1):4-9. doi:10.1192/bjp.bp.107.048504
70. Nakatani Y, Masuko H, Amano T. Effect of lamotrigine on Na(v)1.4 voltage-gated sodium channels. J Pharmacol Sci. 2013;123(2):203-206. doi:10.1254/jphs.13116sc
71. Ramadan E, Basselin M, Rao JS, et al. Lamotrigine blocks NMDA receptor-initiated arachidonic acid signalling in rat brain: implications for its efficacy in bipolar disorder. Int J Neuropsychopharmacol. 2012;15(7):931-943. doi:10.1017/S1461145711001003
72. Jo S, Bean BP. Sidedness of carbamazepine accessibility to voltage-gated sodium channels. Mol Pharmacol. 2014;85(2):381-387. doi:10.1124/mol.113.090472
73. Curtin F, Schulz P. Clonazepam and lorazepam in acute mania: a Bayesian meta-analysis. J Affect Disord 2004;78(3):201-208. doi:10.1016/S0165-0327(02)00317-8
74. Edwards R, Stephenson U, Flewett T. Clonazepam in acute mania: a double blind trial. Aust N Z J Psychiatry 1991;25(2):238-242. doi:10.3109/00048679109077740
75. Lin SC, Chen CC, Chen YH, et al. Benzodiazepine prescription among patients with severe mental illness and co-occurring alcohol abuse/dependence in Taiwan. Hum Psychopharmacol. 2011;26(3):201-207. doi:10.1002/hup.1193
76. Prisciandaro JJ, Brown DG, Brady KT, et al. Comorbid anxiety disorders and baseline medication regimens predict clinical outcomes in individuals with co-occurring bipolar disorder and alcohol dependence: results of a randomized controlled trial. Psychiatry Res. 2011;188(3):361-365. doi:10.1016/j.psychres.2011.04.030
77. Ashok AH, Marques TR, Jauhar S, et al. The dopamine hypothesis of bipolar affective disorder: the state of the art and implications for treatment. Mol Psychiatry. 2017;22(5):666-679. doi:10.1038/mp.2017.16
78. Roberts RJ, Lohano KK, El-Mallakh RS. Antipsychotics as antidepressants. Asia Pac Psychiatry. 2016;8(3):179-188. doi:10.1111/appy.12186
79. Cipriani A, Barbui C, Salanti G, et al. Comparative efficacy and acceptability of antimanic drugs in acute mania: a multiple-treatments meta-analysis. Lancet. 2011;378(9799):1306-1315. doi:10.1016/S0140-6736(11)60873-8
80. Hayes JF, Marston L, Walters K, et al. Lithium vs. valproate vs. olanzapine vs. quetiapine as maintenance monotherapy for bipolar disorder: a population-based UK cohort study using electronic health records. World Psychiatry. 2016;15(1):53-58. doi:10.1002/wps.20298
81. Geddes JR, Gardiner A, Rendell J, et al. Comparative evaluation of quetiapine plus lamotrigine combination versus quetiapine monotherapy (and folic acid versus placebo) in bipolar depression (CEQUEL): a 2 × 2 factorial randomised trial. Lancet Psychiatry. 2016;3(1):31239. doi:10.1016/S2215-0366(15)00450-2
82. Goodwin GM, Haddad PM, Ferrier IN, et al. Evidence-based guidelines for treating bipolar disorder: revised third edition recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2016;30(6):495-553. doi:10.1177/0269881116636545
Depressed and cognitively impaired
CASE Depressed and anxious
Five years ago, Ms. X, age 60, was diagnosed with treatment-resistant major depressive disorder (MDD) with anxiety. This diagnosis was established by a previous psychiatrist. She presents to a clinic for a second opinion.
Since her diagnosis, Ms. X has experienced sad mood, anhedonia, difficulty falling asleep, increased appetite and weight, and decreased concentration and attention. Her anxiety stems from her inability to work, which causes her to worry about her children. In the clinic, the treatment team conducts the Patient Health Questionnaire-9 (PHQ-9) and Generalized Anxiety Disorder-7 item scale (GAD-7) with Ms. X. She scores 16 on the PHQ-9, indicating moderately severe depression, and scores 12 on the GAD-7, indicating moderate anxiety.
Ms. X’s current medication regimen consists of venlafaxine extended-release (XR) 225 mg/d, trazodone 100 mg/d at bedtime, and clonazepam 1 mg twice daily. She reports no significant improvement of her symptoms from these medications. Additionally, Ms. X reports that in the past she had been prescribed fluoxetine, citalopram, and duloxetine, but she cannot recall the dosages.
Ms. X appears appropriately groomed, maintains appropriate eye contact, has clear speech, and does not show evidence of internal stimulation; however, she has difficulty following instructions. She makes negative comments about herself such as “I’m worthless” and “Nobody cares about me.” The treatment team decides to taper Ms. X off venlafaxine XR and initiates sertraline 50 mg/d, while continuing trazodone 50 mg/d at bedtime and clonazepam 1 mg twice daily. The team refers her for cognitive-behavioral therapy (CBT) to address her cognitive distortions, sad mood, and anxiety. Ms. X is asked to follow up with Psychiatry in 1 week.
EVALUATION Unusual behavior
At her CBT intake, Ms. X endorses depression and anxiety. Her PHQ-9 score at this visit is 19 (moderately severe depression) and GAD-7 score is 16 (severe anxiety). The psychologist notes that Ms. X is able to complete activities of daily living and instrumental activities of daily living without assistance. Ms. X denies any use of illicit substances or alcohol. No gross memory impairment is noted during this appointment, though Ms. X exhibits unusual behavior, including exiting and re-entering the clinic multiple times to repeatedly ask about follow-up appointments. The psychologist concludes that Ms. X’s presentation and behavior can be explained by MDD and pseudodementia.
[polldaddy:12189562]
The authors’ observations
Pseudodementia gained recognition in clinical research >100 years ago.1 Officially coined by Kiloh in 1961, the term was used broadly to categorize psychiatric cases that present like dementia but are the result of reversible causes. More recently, it has been used to describe older adults who present with cognitive deficits in the context of depressive symptoms.2 The goal of evaluation is to determine if the primary issue is a cognitive disorder or a depressive episode. DSM-5-TR does not classify pseudodementia as a distinct diagnosis, but instead categorizes its symptoms as components under other major diagnostic categories. Patients can present with MDD and associated cognitive symptoms, or with a cognitive disorder with depressive symptoms, which would be diagnosed as a cognitive disorder with a major depressive-like episode.3
Pseudodementia is rare. Brodaty et al4 found the prevalence of pseudodementia in primary care settings was 0.6%. Older adults (age >65) who live alone are at increased risk of developing pseudodementia, which can be worsened by poor social support and acute psychosocial and environmental changes.5 A key characteristic of this disorder is that as the patient’s depressed mood improves, their memory and cognition also improve.6 Table 13,6 outlines overlapping features of MDD and pseudodementia.
Continue to: EVALUATION Worsening depression
EVALUATION Worsening depression
At her Psychiatry follow-up appointment, Ms. X reports that her mood is worse since she ended the relationship with her partner and she feels anxious because the partner was financially supporting her. Her PHQ-9 score is 24 (severe depression) and her GAD-7 score is 12 (moderate anxiety). Ms. X reports tolerating her transition from venlafaxine XR 225 mg/d to sertraline 50 mg/d well.
Additionally, Ms. X reports her children have called her “useless” since she continues to have difficulties following through on household tasks, even though she has no physical impairments that prevent her from completing them. The Psychiatry team observes that Ms. X has no problems walking or moving her arms or legs.
The Psychiatry team administers the Montreal Cognitive Assessment (MoCA). Ms. X scores 22, indicating mild impairment.
The team recommends a neuropsychological assessment to determine if this MoCA score is due to a cognitive disorder or is rooted in her mood symptoms. The team also recommends an MRI of the brain, complete blood count (CBC), comprehensive metabolic panel (CMP), and urinalysis (UA).
[polldaddy:12189567]
Continue to: The authors' observations
The authors’ observations
Neuropsychological assessments are important tools for exploring the behavioral manifestations of brain dysfunction (Table 2).7 These assessments factor in elements of neurology, psychiatry, and psychology to provide information about the diagnosis, prognosis, and functional status of patients with medical conditions, especially those with neurocognitive and psychiatric disorders. They combine information from the patient and collateral interviews, behavioral observations, a review of patient records, and objective tests of motor, emotional, and cognitive function.
Among other uses, neuropsychological assessments can help identify depression in patients with neurologic impairment, determine the diagnosis and plan of care for patients with concussions, determine the risk of a motor vehicle crash in patients with cognitive impairment, and distinguish Alzheimer disease from vascular dementia.8 Components of such assessments include the Beck Anxiety Inventory (BAI) to assess anxiety, the Dementia Rating Scale-2 and Neuropsychological Assessment Battery-Screening Module to assess dementia, and the Beck Depression Inventory (BDI) to assess depression.9
EVALUATION Continued cognitive decline
A different psychologist performs the neuropsychological assessment, who conducts the Repeatable Battery for the Assessment of Neuropsychological Status Update to determine if Ms. X is experiencing cognitive impairment. Her immediate memory, visuospatial/constructions, language, attention, and delayed memory are significantly impaired for someone her age. The psychologist also administers the Wechsler Adult Intelligence Scale IV and finds Ms. X’s general cognitive ability is within the low average range of intellectual functioning as measured by Full-Scale IQ. Ms. X scores 29 on the BDI-II, indicating significant depressive symptoms, and 13 on the BAI, indicating mild anxiety symptoms.
Ms. X is diagnosed with MDD and an unspecified neurocognitive disorder. The psychologist recommends she start CBT to address her mood and anxiety symptoms.
Upon reviewing the results with Ms. X, the treatment team again recommends a brain MRI, CBC, CMP, and UA to rule out organic causes of her cognitive decline. Ms. X decides against the MRI and laboratory workup and elects to continue her present medication regimen and CBT.
Several weeks later, Ms. X’s family brings her to the emergency department (ED) for evaluation of worsening mood, decreased personal hygiene, increased irritability, and further cognitive decline. They report she is having an increasingly difficult time remembering things such as where she parked her car. The ED team decides to discontinue clonazepam but continues sertraline and trazodone.
Continue to: CBC, CMP, and UA...
CBC, CMP, and UA are unremarkable. Ms. X undergoes a brain CT scan without contrast, which reveals hyperdense lesions in the inferior left tentorium, posterior fossa. A subsequent brain MRI with contrast reveals a dural-based enhancing mass, inferior to the left tentorium, in the left posterior fossa measuring 2.2 cm x 2.1 cm, suggestive of a meningioma. The team orders a Neurosurgery consult.
[polldaddy:12189571]
The authors’ observations
While most brain tumors are secondary to metastasis, meningiomas are the most common primary CNS tumor. Typically, they are asymptomatic; their diagnosis is often delayed until the patient presents with psychiatric symptoms without any focal neurologic findings. The frontal lobe is the most common location of meningioma. Data from 48 case reports of patients with meningiomas and psychiatric symptoms suggest symptoms do not always correlate with specific brain regions.10,11
Indications for neuroimaging in cases such as Ms. X include an abrupt change in behavior or personality, lack of response to psychiatric treatment, presence of focal neurologic signs, and an unusual psychiatric presentation and development of symptoms.11
TREATMENT Neurosurgery
Neurosurgery recommends and performs a suboccipital craniotomy for biopsy and resection. Ms. X tolerates the procedure well. A meningioma is found in the posterior fossa, near the cerebellar convexity. A biopsy finds no evidence of malignancies.
At her postoperative follow-up appointment several days after the procedure, Ms. X reports new-onset hearing loss and tinnitus.
[polldaddy:12189747]
Continue to: The authors' observations
The authors’ observations
Patients who require neurosurgery typically already carry a heavy psychiatric burden, which makes it challenging to determine the exact psychiatric consequences of neurosurgery.12-14 For example, research shows that temporal lobe resection and temporal lobectomy for treatment-resistant epilepsy can lead to an exacerbation of baseline psychiatric symptoms and the development of new symptoms (31% to 34%).15,16 However, Bommakanti et al13 found no new psychiatric symptoms after resection of meningiomas, and surgery seemed to play a role in ameliorating psychiatric symptoms in patients with intracranial tumors. Research attempting to document the psychiatric sequelae of neurosurgery has had mixed results, and it is difficult to determine what effects brain surgery has on mental health.
OUTCOME Minimal improvement
Several weeks after neurosurgery, Ms. X and her family report her mood is improved. Her PHQ-9 score improves to 15, but her GAD-7 score increases to 13, 1 point above her previous score.
The treatment team recommends Ms. X continue taking sertraline 50 mg/d and trazodone 50 mg/d at bedtime. Ms. X’s family reports her cognition and memory have not improved; her MoCA score increases by 1 point to 23. The treatment team discusses with Ms. X and her family the possibility that her cognitive problems maybe better explained as a neurocognitive disorder rather than as a result of the meningioma, since her MoCA score has not significantly improved. Ms. X and her family decide to seek a second opinion from a neurologist.
Bottom Line
Pseudodementia is a term used to describe older adults who present with cognitive issues in the context of depressive symptoms. Even in the absence of focal findings, neuroimaging should be considered as part of the workup in patients who continue to experience a progressive decline in mood and cognitive function.
Related Resources
- Moller MD, Parmenter BA, Lane DW. Neuropsychological testing: a useful but underutilized resource. Current Psychiatry. 2019;18(11):40-46,51.
- Pollak J. Psychological/neuropsychological testing: when to refer for reexamination. Current Psychiatry. 2021;20(9):18- 19,30-31,37. doi:10.12788/cp.0157
Drug Brand Names
Citalopram • Celexa
Clonazepam • Klonopin
Duloxetine • Cymbalta
Fluoxetine • Prozac
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine extended- release • Effexor XR
1. Nussbaum PD. (1994). Pseudodementia: a slow death. Neuropsychol Rev. 1994;4(2):71-90. doi:10.1007/BF01874829
2. Kang H, Zhao F, You L, et al. (2014). Pseudo-dementia: a neuropsychological review. Ann Indian Acad Neurol. 17(2):147-154. doi:10.4103/0972-2327.132613
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, text revision. American Psychiatric Association; 2022.
4. Brodaty H, Connors MH. Pseudodementia, pseudo-pseudodementia, and pseudodepression. Alzheimers Dement (Amst). 2020;12(1):e12027. doi:10.1002/dad2.12027
5. Sekhon S, Marwaha R. Depressive Cognitive Disorders. StatPearls Publishing; 2022. https://www.ncbi.nlm.nih.gov/books/NBK559256/
6. Brown WA. Pseudodementia: issues in diagnosis. Psychiatric Times. April 9, 2005. Accessed February 3, 2023. www.psychiatrictimes.com/view/pseudodementia-issues-diagnosis
7. Kulas JF, Naugle RI. (2003). Indications for neuropsychological assessment. Cleve Clin J Med. 2003;70(9):785-792.
8. Braun M, Tupper D, Kaufmann P, et al. Neuropsychological assessment: a valuable tool in the diagnosis and management of neurological, neurodevelopmental, medical, and psychiatric disorders. Cogn Behav Neurol. 2011;24(3):107-114.
9. Michels TC, Tiu AY, Graver CJ. Neuropsychological evaluation in primary care. Am Fam Physician. 2010;82(5):495-502.
10. Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010;99(3):307-314. doi:10.1007/s11060-010-0386-3
11. Gyawali S, Sharma P, Mahapatra A. Meningioma and psychiatric symptoms: an individual patient data analysis. Asian J Psychiatr. 2019;42:94-103. doi:10.1016/j.ajp.2019.03.029
12. McAllister TW. Neurobehavioral sequelae of traumatic brain injury: evaluation and management. World Psychiatry. 2008;7(1):3-10. doi:10.1002/j.2051-5545.2008.tb00139.x
13. Bommakanti K, Gaddamanugu P, Alladi S, et al. Pre-operative and post-operative psychiatric manifestations in patients with supratentorial meningiomas. Clin Neurol Neurosurg. 2016;147:24-29. doi:10.1016/j.clineuro.2016.05.018
14. Devinsky O, Barr WB, Vickrey BG, et al. Changes in depression and anxiety after resective surgery for epilepsy. Neurology. 2005;65(11):1744-1749. doi:10.1212/01.wnl.0000187114.71524.c3
15. Blumer D, Wakhlu S, Davies K, et al. Psychiatric outcome of temporal lobectomy for epilepsy: incidence and treatment of psychiatric complications. Epilepsia. 1998;39(5):478-486. doi:10.1111/j.1528-1157.1998.tb01409.x
16. Glosser G, Zwil AS, Glosser DS, et al. Psychiatric aspects of temporal lobe epilepsy before and after anterior temporal lobectomy. J Neurol Neurosurg Psychiatry. 2000;68(1):53-58. doi:10.1136/jnnp.68.1.53
CASE Depressed and anxious
Five years ago, Ms. X, age 60, was diagnosed with treatment-resistant major depressive disorder (MDD) with anxiety. This diagnosis was established by a previous psychiatrist. She presents to a clinic for a second opinion.
Since her diagnosis, Ms. X has experienced sad mood, anhedonia, difficulty falling asleep, increased appetite and weight, and decreased concentration and attention. Her anxiety stems from her inability to work, which causes her to worry about her children. In the clinic, the treatment team conducts the Patient Health Questionnaire-9 (PHQ-9) and Generalized Anxiety Disorder-7 item scale (GAD-7) with Ms. X. She scores 16 on the PHQ-9, indicating moderately severe depression, and scores 12 on the GAD-7, indicating moderate anxiety.
Ms. X’s current medication regimen consists of venlafaxine extended-release (XR) 225 mg/d, trazodone 100 mg/d at bedtime, and clonazepam 1 mg twice daily. She reports no significant improvement of her symptoms from these medications. Additionally, Ms. X reports that in the past she had been prescribed fluoxetine, citalopram, and duloxetine, but she cannot recall the dosages.
Ms. X appears appropriately groomed, maintains appropriate eye contact, has clear speech, and does not show evidence of internal stimulation; however, she has difficulty following instructions. She makes negative comments about herself such as “I’m worthless” and “Nobody cares about me.” The treatment team decides to taper Ms. X off venlafaxine XR and initiates sertraline 50 mg/d, while continuing trazodone 50 mg/d at bedtime and clonazepam 1 mg twice daily. The team refers her for cognitive-behavioral therapy (CBT) to address her cognitive distortions, sad mood, and anxiety. Ms. X is asked to follow up with Psychiatry in 1 week.
EVALUATION Unusual behavior
At her CBT intake, Ms. X endorses depression and anxiety. Her PHQ-9 score at this visit is 19 (moderately severe depression) and GAD-7 score is 16 (severe anxiety). The psychologist notes that Ms. X is able to complete activities of daily living and instrumental activities of daily living without assistance. Ms. X denies any use of illicit substances or alcohol. No gross memory impairment is noted during this appointment, though Ms. X exhibits unusual behavior, including exiting and re-entering the clinic multiple times to repeatedly ask about follow-up appointments. The psychologist concludes that Ms. X’s presentation and behavior can be explained by MDD and pseudodementia.
[polldaddy:12189562]
The authors’ observations
Pseudodementia gained recognition in clinical research >100 years ago.1 Officially coined by Kiloh in 1961, the term was used broadly to categorize psychiatric cases that present like dementia but are the result of reversible causes. More recently, it has been used to describe older adults who present with cognitive deficits in the context of depressive symptoms.2 The goal of evaluation is to determine if the primary issue is a cognitive disorder or a depressive episode. DSM-5-TR does not classify pseudodementia as a distinct diagnosis, but instead categorizes its symptoms as components under other major diagnostic categories. Patients can present with MDD and associated cognitive symptoms, or with a cognitive disorder with depressive symptoms, which would be diagnosed as a cognitive disorder with a major depressive-like episode.3
Pseudodementia is rare. Brodaty et al4 found the prevalence of pseudodementia in primary care settings was 0.6%. Older adults (age >65) who live alone are at increased risk of developing pseudodementia, which can be worsened by poor social support and acute psychosocial and environmental changes.5 A key characteristic of this disorder is that as the patient’s depressed mood improves, their memory and cognition also improve.6 Table 13,6 outlines overlapping features of MDD and pseudodementia.
Continue to: EVALUATION Worsening depression
EVALUATION Worsening depression
At her Psychiatry follow-up appointment, Ms. X reports that her mood is worse since she ended the relationship with her partner and she feels anxious because the partner was financially supporting her. Her PHQ-9 score is 24 (severe depression) and her GAD-7 score is 12 (moderate anxiety). Ms. X reports tolerating her transition from venlafaxine XR 225 mg/d to sertraline 50 mg/d well.
Additionally, Ms. X reports her children have called her “useless” since she continues to have difficulties following through on household tasks, even though she has no physical impairments that prevent her from completing them. The Psychiatry team observes that Ms. X has no problems walking or moving her arms or legs.
The Psychiatry team administers the Montreal Cognitive Assessment (MoCA). Ms. X scores 22, indicating mild impairment.
The team recommends a neuropsychological assessment to determine if this MoCA score is due to a cognitive disorder or is rooted in her mood symptoms. The team also recommends an MRI of the brain, complete blood count (CBC), comprehensive metabolic panel (CMP), and urinalysis (UA).
[polldaddy:12189567]
Continue to: The authors' observations
The authors’ observations
Neuropsychological assessments are important tools for exploring the behavioral manifestations of brain dysfunction (Table 2).7 These assessments factor in elements of neurology, psychiatry, and psychology to provide information about the diagnosis, prognosis, and functional status of patients with medical conditions, especially those with neurocognitive and psychiatric disorders. They combine information from the patient and collateral interviews, behavioral observations, a review of patient records, and objective tests of motor, emotional, and cognitive function.
Among other uses, neuropsychological assessments can help identify depression in patients with neurologic impairment, determine the diagnosis and plan of care for patients with concussions, determine the risk of a motor vehicle crash in patients with cognitive impairment, and distinguish Alzheimer disease from vascular dementia.8 Components of such assessments include the Beck Anxiety Inventory (BAI) to assess anxiety, the Dementia Rating Scale-2 and Neuropsychological Assessment Battery-Screening Module to assess dementia, and the Beck Depression Inventory (BDI) to assess depression.9
EVALUATION Continued cognitive decline
A different psychologist performs the neuropsychological assessment, who conducts the Repeatable Battery for the Assessment of Neuropsychological Status Update to determine if Ms. X is experiencing cognitive impairment. Her immediate memory, visuospatial/constructions, language, attention, and delayed memory are significantly impaired for someone her age. The psychologist also administers the Wechsler Adult Intelligence Scale IV and finds Ms. X’s general cognitive ability is within the low average range of intellectual functioning as measured by Full-Scale IQ. Ms. X scores 29 on the BDI-II, indicating significant depressive symptoms, and 13 on the BAI, indicating mild anxiety symptoms.
Ms. X is diagnosed with MDD and an unspecified neurocognitive disorder. The psychologist recommends she start CBT to address her mood and anxiety symptoms.
Upon reviewing the results with Ms. X, the treatment team again recommends a brain MRI, CBC, CMP, and UA to rule out organic causes of her cognitive decline. Ms. X decides against the MRI and laboratory workup and elects to continue her present medication regimen and CBT.
Several weeks later, Ms. X’s family brings her to the emergency department (ED) for evaluation of worsening mood, decreased personal hygiene, increased irritability, and further cognitive decline. They report she is having an increasingly difficult time remembering things such as where she parked her car. The ED team decides to discontinue clonazepam but continues sertraline and trazodone.
Continue to: CBC, CMP, and UA...
CBC, CMP, and UA are unremarkable. Ms. X undergoes a brain CT scan without contrast, which reveals hyperdense lesions in the inferior left tentorium, posterior fossa. A subsequent brain MRI with contrast reveals a dural-based enhancing mass, inferior to the left tentorium, in the left posterior fossa measuring 2.2 cm x 2.1 cm, suggestive of a meningioma. The team orders a Neurosurgery consult.
[polldaddy:12189571]
The authors’ observations
While most brain tumors are secondary to metastasis, meningiomas are the most common primary CNS tumor. Typically, they are asymptomatic; their diagnosis is often delayed until the patient presents with psychiatric symptoms without any focal neurologic findings. The frontal lobe is the most common location of meningioma. Data from 48 case reports of patients with meningiomas and psychiatric symptoms suggest symptoms do not always correlate with specific brain regions.10,11
Indications for neuroimaging in cases such as Ms. X include an abrupt change in behavior or personality, lack of response to psychiatric treatment, presence of focal neurologic signs, and an unusual psychiatric presentation and development of symptoms.11
TREATMENT Neurosurgery
Neurosurgery recommends and performs a suboccipital craniotomy for biopsy and resection. Ms. X tolerates the procedure well. A meningioma is found in the posterior fossa, near the cerebellar convexity. A biopsy finds no evidence of malignancies.
At her postoperative follow-up appointment several days after the procedure, Ms. X reports new-onset hearing loss and tinnitus.
[polldaddy:12189747]
Continue to: The authors' observations
The authors’ observations
Patients who require neurosurgery typically already carry a heavy psychiatric burden, which makes it challenging to determine the exact psychiatric consequences of neurosurgery.12-14 For example, research shows that temporal lobe resection and temporal lobectomy for treatment-resistant epilepsy can lead to an exacerbation of baseline psychiatric symptoms and the development of new symptoms (31% to 34%).15,16 However, Bommakanti et al13 found no new psychiatric symptoms after resection of meningiomas, and surgery seemed to play a role in ameliorating psychiatric symptoms in patients with intracranial tumors. Research attempting to document the psychiatric sequelae of neurosurgery has had mixed results, and it is difficult to determine what effects brain surgery has on mental health.
OUTCOME Minimal improvement
Several weeks after neurosurgery, Ms. X and her family report her mood is improved. Her PHQ-9 score improves to 15, but her GAD-7 score increases to 13, 1 point above her previous score.
The treatment team recommends Ms. X continue taking sertraline 50 mg/d and trazodone 50 mg/d at bedtime. Ms. X’s family reports her cognition and memory have not improved; her MoCA score increases by 1 point to 23. The treatment team discusses with Ms. X and her family the possibility that her cognitive problems maybe better explained as a neurocognitive disorder rather than as a result of the meningioma, since her MoCA score has not significantly improved. Ms. X and her family decide to seek a second opinion from a neurologist.
Bottom Line
Pseudodementia is a term used to describe older adults who present with cognitive issues in the context of depressive symptoms. Even in the absence of focal findings, neuroimaging should be considered as part of the workup in patients who continue to experience a progressive decline in mood and cognitive function.
Related Resources
- Moller MD, Parmenter BA, Lane DW. Neuropsychological testing: a useful but underutilized resource. Current Psychiatry. 2019;18(11):40-46,51.
- Pollak J. Psychological/neuropsychological testing: when to refer for reexamination. Current Psychiatry. 2021;20(9):18- 19,30-31,37. doi:10.12788/cp.0157
Drug Brand Names
Citalopram • Celexa
Clonazepam • Klonopin
Duloxetine • Cymbalta
Fluoxetine • Prozac
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine extended- release • Effexor XR
CASE Depressed and anxious
Five years ago, Ms. X, age 60, was diagnosed with treatment-resistant major depressive disorder (MDD) with anxiety. This diagnosis was established by a previous psychiatrist. She presents to a clinic for a second opinion.
Since her diagnosis, Ms. X has experienced sad mood, anhedonia, difficulty falling asleep, increased appetite and weight, and decreased concentration and attention. Her anxiety stems from her inability to work, which causes her to worry about her children. In the clinic, the treatment team conducts the Patient Health Questionnaire-9 (PHQ-9) and Generalized Anxiety Disorder-7 item scale (GAD-7) with Ms. X. She scores 16 on the PHQ-9, indicating moderately severe depression, and scores 12 on the GAD-7, indicating moderate anxiety.
Ms. X’s current medication regimen consists of venlafaxine extended-release (XR) 225 mg/d, trazodone 100 mg/d at bedtime, and clonazepam 1 mg twice daily. She reports no significant improvement of her symptoms from these medications. Additionally, Ms. X reports that in the past she had been prescribed fluoxetine, citalopram, and duloxetine, but she cannot recall the dosages.
Ms. X appears appropriately groomed, maintains appropriate eye contact, has clear speech, and does not show evidence of internal stimulation; however, she has difficulty following instructions. She makes negative comments about herself such as “I’m worthless” and “Nobody cares about me.” The treatment team decides to taper Ms. X off venlafaxine XR and initiates sertraline 50 mg/d, while continuing trazodone 50 mg/d at bedtime and clonazepam 1 mg twice daily. The team refers her for cognitive-behavioral therapy (CBT) to address her cognitive distortions, sad mood, and anxiety. Ms. X is asked to follow up with Psychiatry in 1 week.
EVALUATION Unusual behavior
At her CBT intake, Ms. X endorses depression and anxiety. Her PHQ-9 score at this visit is 19 (moderately severe depression) and GAD-7 score is 16 (severe anxiety). The psychologist notes that Ms. X is able to complete activities of daily living and instrumental activities of daily living without assistance. Ms. X denies any use of illicit substances or alcohol. No gross memory impairment is noted during this appointment, though Ms. X exhibits unusual behavior, including exiting and re-entering the clinic multiple times to repeatedly ask about follow-up appointments. The psychologist concludes that Ms. X’s presentation and behavior can be explained by MDD and pseudodementia.
[polldaddy:12189562]
The authors’ observations
Pseudodementia gained recognition in clinical research >100 years ago.1 Officially coined by Kiloh in 1961, the term was used broadly to categorize psychiatric cases that present like dementia but are the result of reversible causes. More recently, it has been used to describe older adults who present with cognitive deficits in the context of depressive symptoms.2 The goal of evaluation is to determine if the primary issue is a cognitive disorder or a depressive episode. DSM-5-TR does not classify pseudodementia as a distinct diagnosis, but instead categorizes its symptoms as components under other major diagnostic categories. Patients can present with MDD and associated cognitive symptoms, or with a cognitive disorder with depressive symptoms, which would be diagnosed as a cognitive disorder with a major depressive-like episode.3
Pseudodementia is rare. Brodaty et al4 found the prevalence of pseudodementia in primary care settings was 0.6%. Older adults (age >65) who live alone are at increased risk of developing pseudodementia, which can be worsened by poor social support and acute psychosocial and environmental changes.5 A key characteristic of this disorder is that as the patient’s depressed mood improves, their memory and cognition also improve.6 Table 13,6 outlines overlapping features of MDD and pseudodementia.
Continue to: EVALUATION Worsening depression
EVALUATION Worsening depression
At her Psychiatry follow-up appointment, Ms. X reports that her mood is worse since she ended the relationship with her partner and she feels anxious because the partner was financially supporting her. Her PHQ-9 score is 24 (severe depression) and her GAD-7 score is 12 (moderate anxiety). Ms. X reports tolerating her transition from venlafaxine XR 225 mg/d to sertraline 50 mg/d well.
Additionally, Ms. X reports her children have called her “useless” since she continues to have difficulties following through on household tasks, even though she has no physical impairments that prevent her from completing them. The Psychiatry team observes that Ms. X has no problems walking or moving her arms or legs.
The Psychiatry team administers the Montreal Cognitive Assessment (MoCA). Ms. X scores 22, indicating mild impairment.
The team recommends a neuropsychological assessment to determine if this MoCA score is due to a cognitive disorder or is rooted in her mood symptoms. The team also recommends an MRI of the brain, complete blood count (CBC), comprehensive metabolic panel (CMP), and urinalysis (UA).
[polldaddy:12189567]
Continue to: The authors' observations
The authors’ observations
Neuropsychological assessments are important tools for exploring the behavioral manifestations of brain dysfunction (Table 2).7 These assessments factor in elements of neurology, psychiatry, and psychology to provide information about the diagnosis, prognosis, and functional status of patients with medical conditions, especially those with neurocognitive and psychiatric disorders. They combine information from the patient and collateral interviews, behavioral observations, a review of patient records, and objective tests of motor, emotional, and cognitive function.
Among other uses, neuropsychological assessments can help identify depression in patients with neurologic impairment, determine the diagnosis and plan of care for patients with concussions, determine the risk of a motor vehicle crash in patients with cognitive impairment, and distinguish Alzheimer disease from vascular dementia.8 Components of such assessments include the Beck Anxiety Inventory (BAI) to assess anxiety, the Dementia Rating Scale-2 and Neuropsychological Assessment Battery-Screening Module to assess dementia, and the Beck Depression Inventory (BDI) to assess depression.9
EVALUATION Continued cognitive decline
A different psychologist performs the neuropsychological assessment, who conducts the Repeatable Battery for the Assessment of Neuropsychological Status Update to determine if Ms. X is experiencing cognitive impairment. Her immediate memory, visuospatial/constructions, language, attention, and delayed memory are significantly impaired for someone her age. The psychologist also administers the Wechsler Adult Intelligence Scale IV and finds Ms. X’s general cognitive ability is within the low average range of intellectual functioning as measured by Full-Scale IQ. Ms. X scores 29 on the BDI-II, indicating significant depressive symptoms, and 13 on the BAI, indicating mild anxiety symptoms.
Ms. X is diagnosed with MDD and an unspecified neurocognitive disorder. The psychologist recommends she start CBT to address her mood and anxiety symptoms.
Upon reviewing the results with Ms. X, the treatment team again recommends a brain MRI, CBC, CMP, and UA to rule out organic causes of her cognitive decline. Ms. X decides against the MRI and laboratory workup and elects to continue her present medication regimen and CBT.
Several weeks later, Ms. X’s family brings her to the emergency department (ED) for evaluation of worsening mood, decreased personal hygiene, increased irritability, and further cognitive decline. They report she is having an increasingly difficult time remembering things such as where she parked her car. The ED team decides to discontinue clonazepam but continues sertraline and trazodone.
Continue to: CBC, CMP, and UA...
CBC, CMP, and UA are unremarkable. Ms. X undergoes a brain CT scan without contrast, which reveals hyperdense lesions in the inferior left tentorium, posterior fossa. A subsequent brain MRI with contrast reveals a dural-based enhancing mass, inferior to the left tentorium, in the left posterior fossa measuring 2.2 cm x 2.1 cm, suggestive of a meningioma. The team orders a Neurosurgery consult.
[polldaddy:12189571]
The authors’ observations
While most brain tumors are secondary to metastasis, meningiomas are the most common primary CNS tumor. Typically, they are asymptomatic; their diagnosis is often delayed until the patient presents with psychiatric symptoms without any focal neurologic findings. The frontal lobe is the most common location of meningioma. Data from 48 case reports of patients with meningiomas and psychiatric symptoms suggest symptoms do not always correlate with specific brain regions.10,11
Indications for neuroimaging in cases such as Ms. X include an abrupt change in behavior or personality, lack of response to psychiatric treatment, presence of focal neurologic signs, and an unusual psychiatric presentation and development of symptoms.11
TREATMENT Neurosurgery
Neurosurgery recommends and performs a suboccipital craniotomy for biopsy and resection. Ms. X tolerates the procedure well. A meningioma is found in the posterior fossa, near the cerebellar convexity. A biopsy finds no evidence of malignancies.
At her postoperative follow-up appointment several days after the procedure, Ms. X reports new-onset hearing loss and tinnitus.
[polldaddy:12189747]
Continue to: The authors' observations
The authors’ observations
Patients who require neurosurgery typically already carry a heavy psychiatric burden, which makes it challenging to determine the exact psychiatric consequences of neurosurgery.12-14 For example, research shows that temporal lobe resection and temporal lobectomy for treatment-resistant epilepsy can lead to an exacerbation of baseline psychiatric symptoms and the development of new symptoms (31% to 34%).15,16 However, Bommakanti et al13 found no new psychiatric symptoms after resection of meningiomas, and surgery seemed to play a role in ameliorating psychiatric symptoms in patients with intracranial tumors. Research attempting to document the psychiatric sequelae of neurosurgery has had mixed results, and it is difficult to determine what effects brain surgery has on mental health.
OUTCOME Minimal improvement
Several weeks after neurosurgery, Ms. X and her family report her mood is improved. Her PHQ-9 score improves to 15, but her GAD-7 score increases to 13, 1 point above her previous score.
The treatment team recommends Ms. X continue taking sertraline 50 mg/d and trazodone 50 mg/d at bedtime. Ms. X’s family reports her cognition and memory have not improved; her MoCA score increases by 1 point to 23. The treatment team discusses with Ms. X and her family the possibility that her cognitive problems maybe better explained as a neurocognitive disorder rather than as a result of the meningioma, since her MoCA score has not significantly improved. Ms. X and her family decide to seek a second opinion from a neurologist.
Bottom Line
Pseudodementia is a term used to describe older adults who present with cognitive issues in the context of depressive symptoms. Even in the absence of focal findings, neuroimaging should be considered as part of the workup in patients who continue to experience a progressive decline in mood and cognitive function.
Related Resources
- Moller MD, Parmenter BA, Lane DW. Neuropsychological testing: a useful but underutilized resource. Current Psychiatry. 2019;18(11):40-46,51.
- Pollak J. Psychological/neuropsychological testing: when to refer for reexamination. Current Psychiatry. 2021;20(9):18- 19,30-31,37. doi:10.12788/cp.0157
Drug Brand Names
Citalopram • Celexa
Clonazepam • Klonopin
Duloxetine • Cymbalta
Fluoxetine • Prozac
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine extended- release • Effexor XR
1. Nussbaum PD. (1994). Pseudodementia: a slow death. Neuropsychol Rev. 1994;4(2):71-90. doi:10.1007/BF01874829
2. Kang H, Zhao F, You L, et al. (2014). Pseudo-dementia: a neuropsychological review. Ann Indian Acad Neurol. 17(2):147-154. doi:10.4103/0972-2327.132613
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, text revision. American Psychiatric Association; 2022.
4. Brodaty H, Connors MH. Pseudodementia, pseudo-pseudodementia, and pseudodepression. Alzheimers Dement (Amst). 2020;12(1):e12027. doi:10.1002/dad2.12027
5. Sekhon S, Marwaha R. Depressive Cognitive Disorders. StatPearls Publishing; 2022. https://www.ncbi.nlm.nih.gov/books/NBK559256/
6. Brown WA. Pseudodementia: issues in diagnosis. Psychiatric Times. April 9, 2005. Accessed February 3, 2023. www.psychiatrictimes.com/view/pseudodementia-issues-diagnosis
7. Kulas JF, Naugle RI. (2003). Indications for neuropsychological assessment. Cleve Clin J Med. 2003;70(9):785-792.
8. Braun M, Tupper D, Kaufmann P, et al. Neuropsychological assessment: a valuable tool in the diagnosis and management of neurological, neurodevelopmental, medical, and psychiatric disorders. Cogn Behav Neurol. 2011;24(3):107-114.
9. Michels TC, Tiu AY, Graver CJ. Neuropsychological evaluation in primary care. Am Fam Physician. 2010;82(5):495-502.
10. Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010;99(3):307-314. doi:10.1007/s11060-010-0386-3
11. Gyawali S, Sharma P, Mahapatra A. Meningioma and psychiatric symptoms: an individual patient data analysis. Asian J Psychiatr. 2019;42:94-103. doi:10.1016/j.ajp.2019.03.029
12. McAllister TW. Neurobehavioral sequelae of traumatic brain injury: evaluation and management. World Psychiatry. 2008;7(1):3-10. doi:10.1002/j.2051-5545.2008.tb00139.x
13. Bommakanti K, Gaddamanugu P, Alladi S, et al. Pre-operative and post-operative psychiatric manifestations in patients with supratentorial meningiomas. Clin Neurol Neurosurg. 2016;147:24-29. doi:10.1016/j.clineuro.2016.05.018
14. Devinsky O, Barr WB, Vickrey BG, et al. Changes in depression and anxiety after resective surgery for epilepsy. Neurology. 2005;65(11):1744-1749. doi:10.1212/01.wnl.0000187114.71524.c3
15. Blumer D, Wakhlu S, Davies K, et al. Psychiatric outcome of temporal lobectomy for epilepsy: incidence and treatment of psychiatric complications. Epilepsia. 1998;39(5):478-486. doi:10.1111/j.1528-1157.1998.tb01409.x
16. Glosser G, Zwil AS, Glosser DS, et al. Psychiatric aspects of temporal lobe epilepsy before and after anterior temporal lobectomy. J Neurol Neurosurg Psychiatry. 2000;68(1):53-58. doi:10.1136/jnnp.68.1.53
1. Nussbaum PD. (1994). Pseudodementia: a slow death. Neuropsychol Rev. 1994;4(2):71-90. doi:10.1007/BF01874829
2. Kang H, Zhao F, You L, et al. (2014). Pseudo-dementia: a neuropsychological review. Ann Indian Acad Neurol. 17(2):147-154. doi:10.4103/0972-2327.132613
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, text revision. American Psychiatric Association; 2022.
4. Brodaty H, Connors MH. Pseudodementia, pseudo-pseudodementia, and pseudodepression. Alzheimers Dement (Amst). 2020;12(1):e12027. doi:10.1002/dad2.12027
5. Sekhon S, Marwaha R. Depressive Cognitive Disorders. StatPearls Publishing; 2022. https://www.ncbi.nlm.nih.gov/books/NBK559256/
6. Brown WA. Pseudodementia: issues in diagnosis. Psychiatric Times. April 9, 2005. Accessed February 3, 2023. www.psychiatrictimes.com/view/pseudodementia-issues-diagnosis
7. Kulas JF, Naugle RI. (2003). Indications for neuropsychological assessment. Cleve Clin J Med. 2003;70(9):785-792.
8. Braun M, Tupper D, Kaufmann P, et al. Neuropsychological assessment: a valuable tool in the diagnosis and management of neurological, neurodevelopmental, medical, and psychiatric disorders. Cogn Behav Neurol. 2011;24(3):107-114.
9. Michels TC, Tiu AY, Graver CJ. Neuropsychological evaluation in primary care. Am Fam Physician. 2010;82(5):495-502.
10. Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010;99(3):307-314. doi:10.1007/s11060-010-0386-3
11. Gyawali S, Sharma P, Mahapatra A. Meningioma and psychiatric symptoms: an individual patient data analysis. Asian J Psychiatr. 2019;42:94-103. doi:10.1016/j.ajp.2019.03.029
12. McAllister TW. Neurobehavioral sequelae of traumatic brain injury: evaluation and management. World Psychiatry. 2008;7(1):3-10. doi:10.1002/j.2051-5545.2008.tb00139.x
13. Bommakanti K, Gaddamanugu P, Alladi S, et al. Pre-operative and post-operative psychiatric manifestations in patients with supratentorial meningiomas. Clin Neurol Neurosurg. 2016;147:24-29. doi:10.1016/j.clineuro.2016.05.018
14. Devinsky O, Barr WB, Vickrey BG, et al. Changes in depression and anxiety after resective surgery for epilepsy. Neurology. 2005;65(11):1744-1749. doi:10.1212/01.wnl.0000187114.71524.c3
15. Blumer D, Wakhlu S, Davies K, et al. Psychiatric outcome of temporal lobectomy for epilepsy: incidence and treatment of psychiatric complications. Epilepsia. 1998;39(5):478-486. doi:10.1111/j.1528-1157.1998.tb01409.x
16. Glosser G, Zwil AS, Glosser DS, et al. Psychiatric aspects of temporal lobe epilepsy before and after anterior temporal lobectomy. J Neurol Neurosurg Psychiatry. 2000;68(1):53-58. doi:10.1136/jnnp.68.1.53
Tips for efficient night shift work in a psychiatric ED
Attending psychiatrists who work night shift in a psychiatric emergency department (ED) or medical ED require a different set of skills than when working daytime or evening shifts, especially when working full-time or solo. While all patients should be treated carefully and meticulously regardless of the shift, this article offers tips for efficiency for solo attending psychiatrists who work night shift in an ED.
Check orders. Typically, multiple psychiatric clinicians are available on other shifts, but only 1 at night. This can lead to significant variability and potential errors in patients’ orders. Such errors filter down to night shift and often must be addressed by the solo clinician, who can’t say “that person is not my patient” because there are no other clinicians available to help. Carefully check orders (ideally, on all patients every shift) to ensure there are no errors or omissions.
Use note templates. While it is important to avoid using mere checklists, with electronic medical record systems, create templates for typical notes. This will save time when the pace of patients increases.
Be brief in your documentation. Brevity is key when documenting at night. Focus on what is necessary and sufficient.
Conduct thorough but efficient interviews. Be aware of how much time you spend on patient interviews. While still thorough, interviews must often be shorter due to a higher staff-to-patient ratio at night.
Be aware of potential medical issues. Many psychiatric EDs are not attached to a hospital. With other medical consultants not readily available in the middle of the night, be particularly alert for any acute medical issues that may arise, and act accordingly.
Focus on the order of tasks. Be aware of which tasks you complete and in what order. For example, at night you may need to medicate sooner for agitation because other patients are sleeping, instead of letting one patient’s agitation disrupt the entire night milieu.
Continue to: Don't let tasks pile up
Don’t let tasks pile up. Time management and multitasking are key skills at night. Take care of clinical issues as they arise. Finish documentation as you go along. Don’t let things pile up throughout your shift and then spend significant time after your shift to catch up.
Know your staff. The staff around you are your eyes and ears. Get to know your clinical and nonclinical staff’s tendencies. This can be immensely helpful in picking up any different patterns when interviewing and observing patients.
Know your limits. You may not be able to solve everything or obtain the ideal collateral at night. Don’t get caught up in definitively trying to resolve things and end up wasting precious time at night. Let it go. Don’t overthink. If all else fails, hold the patient overnight.
Prioritize self-care. Night shift work has been shown to negatively impact one’s health.1-3 If you choose this type of work, either part-time or full-time, maintain your own health by exercising regularly, eating a healthy diet, obtaining adequate rest between shifts, and seeing your health care team often.
1. Wu QJ, Sun H, Wen ZY, et al. Shift work and health outcomes: an umbrella review of systematic reviews and meta-analyses of epidemiological studies. J Clin Sleep Med. 2022;18(2):653-662. doi:10.5664/jcsm.9642
2. Kecklund G, Axelsson J. Health consequences of shift work and insufficient sleep. BMJ. 2016;355:i5210. doi:10.1136/bmj.i5210
3. Boivin DB, Boudreau P. Impacts of shift work on sleep and circadian rhythms. Pathol Biol (Paris). 2014;62(5):292-301. doi:10.1016/j.patbio.2014.08.001
Attending psychiatrists who work night shift in a psychiatric emergency department (ED) or medical ED require a different set of skills than when working daytime or evening shifts, especially when working full-time or solo. While all patients should be treated carefully and meticulously regardless of the shift, this article offers tips for efficiency for solo attending psychiatrists who work night shift in an ED.
Check orders. Typically, multiple psychiatric clinicians are available on other shifts, but only 1 at night. This can lead to significant variability and potential errors in patients’ orders. Such errors filter down to night shift and often must be addressed by the solo clinician, who can’t say “that person is not my patient” because there are no other clinicians available to help. Carefully check orders (ideally, on all patients every shift) to ensure there are no errors or omissions.
Use note templates. While it is important to avoid using mere checklists, with electronic medical record systems, create templates for typical notes. This will save time when the pace of patients increases.
Be brief in your documentation. Brevity is key when documenting at night. Focus on what is necessary and sufficient.
Conduct thorough but efficient interviews. Be aware of how much time you spend on patient interviews. While still thorough, interviews must often be shorter due to a higher staff-to-patient ratio at night.
Be aware of potential medical issues. Many psychiatric EDs are not attached to a hospital. With other medical consultants not readily available in the middle of the night, be particularly alert for any acute medical issues that may arise, and act accordingly.
Focus on the order of tasks. Be aware of which tasks you complete and in what order. For example, at night you may need to medicate sooner for agitation because other patients are sleeping, instead of letting one patient’s agitation disrupt the entire night milieu.
Continue to: Don't let tasks pile up
Don’t let tasks pile up. Time management and multitasking are key skills at night. Take care of clinical issues as they arise. Finish documentation as you go along. Don’t let things pile up throughout your shift and then spend significant time after your shift to catch up.
Know your staff. The staff around you are your eyes and ears. Get to know your clinical and nonclinical staff’s tendencies. This can be immensely helpful in picking up any different patterns when interviewing and observing patients.
Know your limits. You may not be able to solve everything or obtain the ideal collateral at night. Don’t get caught up in definitively trying to resolve things and end up wasting precious time at night. Let it go. Don’t overthink. If all else fails, hold the patient overnight.
Prioritize self-care. Night shift work has been shown to negatively impact one’s health.1-3 If you choose this type of work, either part-time or full-time, maintain your own health by exercising regularly, eating a healthy diet, obtaining adequate rest between shifts, and seeing your health care team often.
Attending psychiatrists who work night shift in a psychiatric emergency department (ED) or medical ED require a different set of skills than when working daytime or evening shifts, especially when working full-time or solo. While all patients should be treated carefully and meticulously regardless of the shift, this article offers tips for efficiency for solo attending psychiatrists who work night shift in an ED.
Check orders. Typically, multiple psychiatric clinicians are available on other shifts, but only 1 at night. This can lead to significant variability and potential errors in patients’ orders. Such errors filter down to night shift and often must be addressed by the solo clinician, who can’t say “that person is not my patient” because there are no other clinicians available to help. Carefully check orders (ideally, on all patients every shift) to ensure there are no errors or omissions.
Use note templates. While it is important to avoid using mere checklists, with electronic medical record systems, create templates for typical notes. This will save time when the pace of patients increases.
Be brief in your documentation. Brevity is key when documenting at night. Focus on what is necessary and sufficient.
Conduct thorough but efficient interviews. Be aware of how much time you spend on patient interviews. While still thorough, interviews must often be shorter due to a higher staff-to-patient ratio at night.
Be aware of potential medical issues. Many psychiatric EDs are not attached to a hospital. With other medical consultants not readily available in the middle of the night, be particularly alert for any acute medical issues that may arise, and act accordingly.
Focus on the order of tasks. Be aware of which tasks you complete and in what order. For example, at night you may need to medicate sooner for agitation because other patients are sleeping, instead of letting one patient’s agitation disrupt the entire night milieu.
Continue to: Don't let tasks pile up
Don’t let tasks pile up. Time management and multitasking are key skills at night. Take care of clinical issues as they arise. Finish documentation as you go along. Don’t let things pile up throughout your shift and then spend significant time after your shift to catch up.
Know your staff. The staff around you are your eyes and ears. Get to know your clinical and nonclinical staff’s tendencies. This can be immensely helpful in picking up any different patterns when interviewing and observing patients.
Know your limits. You may not be able to solve everything or obtain the ideal collateral at night. Don’t get caught up in definitively trying to resolve things and end up wasting precious time at night. Let it go. Don’t overthink. If all else fails, hold the patient overnight.
Prioritize self-care. Night shift work has been shown to negatively impact one’s health.1-3 If you choose this type of work, either part-time or full-time, maintain your own health by exercising regularly, eating a healthy diet, obtaining adequate rest between shifts, and seeing your health care team often.
1. Wu QJ, Sun H, Wen ZY, et al. Shift work and health outcomes: an umbrella review of systematic reviews and meta-analyses of epidemiological studies. J Clin Sleep Med. 2022;18(2):653-662. doi:10.5664/jcsm.9642
2. Kecklund G, Axelsson J. Health consequences of shift work and insufficient sleep. BMJ. 2016;355:i5210. doi:10.1136/bmj.i5210
3. Boivin DB, Boudreau P. Impacts of shift work on sleep and circadian rhythms. Pathol Biol (Paris). 2014;62(5):292-301. doi:10.1016/j.patbio.2014.08.001
1. Wu QJ, Sun H, Wen ZY, et al. Shift work and health outcomes: an umbrella review of systematic reviews and meta-analyses of epidemiological studies. J Clin Sleep Med. 2022;18(2):653-662. doi:10.5664/jcsm.9642
2. Kecklund G, Axelsson J. Health consequences of shift work and insufficient sleep. BMJ. 2016;355:i5210. doi:10.1136/bmj.i5210
3. Boivin DB, Boudreau P. Impacts of shift work on sleep and circadian rhythms. Pathol Biol (Paris). 2014;62(5):292-301. doi:10.1016/j.patbio.2014.08.001
Ethics do not end at the bedside: A commentary about scientific authorship
Sound moral principles are essential in the development of all physicians. Given how heavily each clinical encounter is laden with ethical implications, this is taught early in medical school. The medical student and resident physician must be able to make ethical and moral decisions on a consistent basis.
Speaking as a psychiatrist in training, there is an intimate relationship between psychiatry and moral questions.1 Issues such as determining an individual’s ability to make decisions about their medical care, hospitalizing patients against their will, and involuntarily administering medication are an almost-daily occurrence.2 Physicians, especially those who practice psychiatric medicine, must be ethically grounded to properly make these difficult but common decisions. It is also imperative that residents are given proper guidance in ethical practice in structured didactics and hands-on training.
However, many residents may be unfamiliar with ethics in research, more specifically ethical authorship. While some trainees might have participated in scholarly activities before residency, residency is the time to discover one’s interests, and residents are encouraged to engage in research. Unfortunately, many of the considerations surrounding ethical authorship are not emphasized, and questionable practices are common.3 In this article, I summarize the different faces of unethical authorship, and call for a greater emphasis on ethical authorship in medical residency training programs.
What drives unethical authorship practices
One of the main drivers for the increase in unethical practices is the need to publish to advance one’s academic career. The academic principle of “publish or perish” pressures many faculty researchers.3 The impact of this expectation plays a significant role in potentially unethical authorship practices, and also has increased the number of publications of mediocre quality or fraudulent data.4 This mindset has also seeped into the clinical world because promotions and financial bonuses are incentives for attending physicians to perform scholarly work. Due to these incentives and pressures, a senior academician might compel a junior researcher to include them as a coauthor on the junior researcher’s paper, even when the senior’s contributions to the paper might be limited.5
Most journals have specific criteria for authorship. The International Committee of Medical Journal Editors (ICMJE) has 4 core criteria for authorship: 1) substantial contributions to the conception or design of the work, or the acquisition, analysis, or interpretation of data for the work; 2) drafting the work or revising it critically for important intellectual content; 3) providing final approval of the version to be published, and 4) agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.5,6 One survey found that in certain journals, approximately 15% of authors met full ICMJE authorship criteria, while one-half claimed there were substantial contributions but did not state anything more specific.7
There are several types of authorship abuse.5 Gift authorship is when authorship is awarded to a friend either out of respect or in hopes that friend will return the favor (quid pro quo). Ghost authorship occurs when a third party commissions an author to write or help write a paper (eg, when a pharmaceutical company hires writers to produce a paper about a medication they manufacture) or when legitimate authors are denied recognition on a paper. Honorary authorship occurs when authorship is granted with the hope that the reputation of the honorary author will increase the chances of the paper getting published and possibly boost citations.
While these forms of authorship abuse occur with unsettling frequency, they might not be common among physician trainees who do not engage in full-time research.5 Resident authors might be more likely to experience coercive authorship.
Continue to: Coercive authorship is when...
Coercive authorship is when an individual in a superior position (such as an attending physician) forces their name onto a paper of a junior individual (such as a resident). Kwok8 called this “The White Bull effect,” based on Greek mythology in which Zeus transformed himself into a white bull to seduce Europa. The White Bull represents the predatory nature of the senior individual who exploits ambiguous institutional research regulations to their benefit.8 They stretch out the ICMJE criteria, only superficially satisfying them to justify authorship. In this scenario, the attending physician with promotional incentives notices the work of a resident and demands authorship, given their role as the “supervising” physician (akin to general supervision of a research group). This is not justification for authorship per the ICMJE or any major medical journal criteria. However, a resident with limited research experience may agree to include the attending as a coauthor for a variety of reasons, including fear of a poor performance evaluation or professionalism complaints, or just to maintain a positive working relationship.
Serious implications
While there are countless reasons to be concerned about this behavior, the central issue is the attending physician’s role to train and/or mentor the resident. As previously stated, a physician—especially one practicing psychiatric medicine—must be of morally sound mind. A resident being taught unethical behaviors by their attending physician has dangerous implications. Academic dishonesty does not occur in vacuum. It is likely that dishonest and unethical behavior in research matters can cross over into the clinical arena. One study found that individuals who exhibit dishonest academic behavior are more likely to violate workplace policies.9 Also, these behaviors lead to increased moral disengagement in all areas.10,11 Imagining a morally disengaged attending psychiatrist practicing medicine and training the next generation of psychiatrists is unsettling.
My hope is that residency programs discourage this detrimental conduct in their departments and support those trying to uphold integrity.
1. Scher S, Kozlowska K. Teaching ethics in psychiatry: time to reset. Harv Rev Psychiatry. 2020;28(5):328-333. doi:10.1097/HRP.0000000000000258
2. Allen NG, Khan JS, Alzahri MS, et al. Ethical issues in emergency psychiatry. Emerg Med Clin North Am. 2015;33(4):863-874. doi:10.1016/j.emc.2015.07.012
3. Pfleegor AG, Katz M, Bowers MT. Publish, perish, or salami slice? Authorship ethics in an emerging field. Journal of Business Ethics. 2019;156(1):189-208.
4. Rivera H. Fake peer review and inappropriate authorship are real evils. J Korean Med Sci. 2018;34(2):e6. doi:10.3346/jkms.2019.34.e6
5. Strange K. Authorship: why not just toss a coin? Am J Physiol Cell Physiol. 2008;295(3):C567-C575. doi:10.1152/ajpcell.00208.2008
6. Ali MJ. ICMJE criteria for authorship: why the criticisms are not justified? Graefes Arch Clin Exp Ophthalmol. 2021;259(2):289-290. doi:10.1007/s00417-020-04825-2
7. Malički M, Jerončić A, Marušić M, et al. Why do you think you should be the author on this manuscript? Analysis of open-ended responses of authors in a general medical journal. BMC Med Res Methodol. 2012;12:189. doi:10.1186/1471-2288-12-189
8. Kwok LS. The White Bull effect: abusive coauthorship and publication parasitism. J Med Ethics. 2005;31(9):554-556. doi:10.1136/jme.2004.010553
9. Harding TS, Carpenter DD, Finelli CJ, et al. Does academic dishonesty relate to unethical behavior in professional practice? An exploratory study. Sci Eng Ethics. 2004;10(2):311-324. doi:10.1007/s11948-004-0027-3
10. Shu LL, Gino F. Sweeping dishonesty under the rug: how unethical actions lead to forgetting of moral rules. J Pers Soc Psychol. 2012;102(6):1164-1177. doi:10.1037/a0028381
11. Shu LL, Gino F, Bazerman MH. Dishonest deed, clear conscience: when cheating leads to moral disengagement and motivated forgetting. Pers Soc Psychol Bull. 2011;37(3):330-349. doi:10.1177/0146167211398138
Sound moral principles are essential in the development of all physicians. Given how heavily each clinical encounter is laden with ethical implications, this is taught early in medical school. The medical student and resident physician must be able to make ethical and moral decisions on a consistent basis.
Speaking as a psychiatrist in training, there is an intimate relationship between psychiatry and moral questions.1 Issues such as determining an individual’s ability to make decisions about their medical care, hospitalizing patients against their will, and involuntarily administering medication are an almost-daily occurrence.2 Physicians, especially those who practice psychiatric medicine, must be ethically grounded to properly make these difficult but common decisions. It is also imperative that residents are given proper guidance in ethical practice in structured didactics and hands-on training.
However, many residents may be unfamiliar with ethics in research, more specifically ethical authorship. While some trainees might have participated in scholarly activities before residency, residency is the time to discover one’s interests, and residents are encouraged to engage in research. Unfortunately, many of the considerations surrounding ethical authorship are not emphasized, and questionable practices are common.3 In this article, I summarize the different faces of unethical authorship, and call for a greater emphasis on ethical authorship in medical residency training programs.
What drives unethical authorship practices
One of the main drivers for the increase in unethical practices is the need to publish to advance one’s academic career. The academic principle of “publish or perish” pressures many faculty researchers.3 The impact of this expectation plays a significant role in potentially unethical authorship practices, and also has increased the number of publications of mediocre quality or fraudulent data.4 This mindset has also seeped into the clinical world because promotions and financial bonuses are incentives for attending physicians to perform scholarly work. Due to these incentives and pressures, a senior academician might compel a junior researcher to include them as a coauthor on the junior researcher’s paper, even when the senior’s contributions to the paper might be limited.5
Most journals have specific criteria for authorship. The International Committee of Medical Journal Editors (ICMJE) has 4 core criteria for authorship: 1) substantial contributions to the conception or design of the work, or the acquisition, analysis, or interpretation of data for the work; 2) drafting the work or revising it critically for important intellectual content; 3) providing final approval of the version to be published, and 4) agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.5,6 One survey found that in certain journals, approximately 15% of authors met full ICMJE authorship criteria, while one-half claimed there were substantial contributions but did not state anything more specific.7
There are several types of authorship abuse.5 Gift authorship is when authorship is awarded to a friend either out of respect or in hopes that friend will return the favor (quid pro quo). Ghost authorship occurs when a third party commissions an author to write or help write a paper (eg, when a pharmaceutical company hires writers to produce a paper about a medication they manufacture) or when legitimate authors are denied recognition on a paper. Honorary authorship occurs when authorship is granted with the hope that the reputation of the honorary author will increase the chances of the paper getting published and possibly boost citations.
While these forms of authorship abuse occur with unsettling frequency, they might not be common among physician trainees who do not engage in full-time research.5 Resident authors might be more likely to experience coercive authorship.
Continue to: Coercive authorship is when...
Coercive authorship is when an individual in a superior position (such as an attending physician) forces their name onto a paper of a junior individual (such as a resident). Kwok8 called this “The White Bull effect,” based on Greek mythology in which Zeus transformed himself into a white bull to seduce Europa. The White Bull represents the predatory nature of the senior individual who exploits ambiguous institutional research regulations to their benefit.8 They stretch out the ICMJE criteria, only superficially satisfying them to justify authorship. In this scenario, the attending physician with promotional incentives notices the work of a resident and demands authorship, given their role as the “supervising” physician (akin to general supervision of a research group). This is not justification for authorship per the ICMJE or any major medical journal criteria. However, a resident with limited research experience may agree to include the attending as a coauthor for a variety of reasons, including fear of a poor performance evaluation or professionalism complaints, or just to maintain a positive working relationship.
Serious implications
While there are countless reasons to be concerned about this behavior, the central issue is the attending physician’s role to train and/or mentor the resident. As previously stated, a physician—especially one practicing psychiatric medicine—must be of morally sound mind. A resident being taught unethical behaviors by their attending physician has dangerous implications. Academic dishonesty does not occur in vacuum. It is likely that dishonest and unethical behavior in research matters can cross over into the clinical arena. One study found that individuals who exhibit dishonest academic behavior are more likely to violate workplace policies.9 Also, these behaviors lead to increased moral disengagement in all areas.10,11 Imagining a morally disengaged attending psychiatrist practicing medicine and training the next generation of psychiatrists is unsettling.
My hope is that residency programs discourage this detrimental conduct in their departments and support those trying to uphold integrity.
Sound moral principles are essential in the development of all physicians. Given how heavily each clinical encounter is laden with ethical implications, this is taught early in medical school. The medical student and resident physician must be able to make ethical and moral decisions on a consistent basis.
Speaking as a psychiatrist in training, there is an intimate relationship between psychiatry and moral questions.1 Issues such as determining an individual’s ability to make decisions about their medical care, hospitalizing patients against their will, and involuntarily administering medication are an almost-daily occurrence.2 Physicians, especially those who practice psychiatric medicine, must be ethically grounded to properly make these difficult but common decisions. It is also imperative that residents are given proper guidance in ethical practice in structured didactics and hands-on training.
However, many residents may be unfamiliar with ethics in research, more specifically ethical authorship. While some trainees might have participated in scholarly activities before residency, residency is the time to discover one’s interests, and residents are encouraged to engage in research. Unfortunately, many of the considerations surrounding ethical authorship are not emphasized, and questionable practices are common.3 In this article, I summarize the different faces of unethical authorship, and call for a greater emphasis on ethical authorship in medical residency training programs.
What drives unethical authorship practices
One of the main drivers for the increase in unethical practices is the need to publish to advance one’s academic career. The academic principle of “publish or perish” pressures many faculty researchers.3 The impact of this expectation plays a significant role in potentially unethical authorship practices, and also has increased the number of publications of mediocre quality or fraudulent data.4 This mindset has also seeped into the clinical world because promotions and financial bonuses are incentives for attending physicians to perform scholarly work. Due to these incentives and pressures, a senior academician might compel a junior researcher to include them as a coauthor on the junior researcher’s paper, even when the senior’s contributions to the paper might be limited.5
Most journals have specific criteria for authorship. The International Committee of Medical Journal Editors (ICMJE) has 4 core criteria for authorship: 1) substantial contributions to the conception or design of the work, or the acquisition, analysis, or interpretation of data for the work; 2) drafting the work or revising it critically for important intellectual content; 3) providing final approval of the version to be published, and 4) agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.5,6 One survey found that in certain journals, approximately 15% of authors met full ICMJE authorship criteria, while one-half claimed there were substantial contributions but did not state anything more specific.7
There are several types of authorship abuse.5 Gift authorship is when authorship is awarded to a friend either out of respect or in hopes that friend will return the favor (quid pro quo). Ghost authorship occurs when a third party commissions an author to write or help write a paper (eg, when a pharmaceutical company hires writers to produce a paper about a medication they manufacture) or when legitimate authors are denied recognition on a paper. Honorary authorship occurs when authorship is granted with the hope that the reputation of the honorary author will increase the chances of the paper getting published and possibly boost citations.
While these forms of authorship abuse occur with unsettling frequency, they might not be common among physician trainees who do not engage in full-time research.5 Resident authors might be more likely to experience coercive authorship.
Continue to: Coercive authorship is when...
Coercive authorship is when an individual in a superior position (such as an attending physician) forces their name onto a paper of a junior individual (such as a resident). Kwok8 called this “The White Bull effect,” based on Greek mythology in which Zeus transformed himself into a white bull to seduce Europa. The White Bull represents the predatory nature of the senior individual who exploits ambiguous institutional research regulations to their benefit.8 They stretch out the ICMJE criteria, only superficially satisfying them to justify authorship. In this scenario, the attending physician with promotional incentives notices the work of a resident and demands authorship, given their role as the “supervising” physician (akin to general supervision of a research group). This is not justification for authorship per the ICMJE or any major medical journal criteria. However, a resident with limited research experience may agree to include the attending as a coauthor for a variety of reasons, including fear of a poor performance evaluation or professionalism complaints, or just to maintain a positive working relationship.
Serious implications
While there are countless reasons to be concerned about this behavior, the central issue is the attending physician’s role to train and/or mentor the resident. As previously stated, a physician—especially one practicing psychiatric medicine—must be of morally sound mind. A resident being taught unethical behaviors by their attending physician has dangerous implications. Academic dishonesty does not occur in vacuum. It is likely that dishonest and unethical behavior in research matters can cross over into the clinical arena. One study found that individuals who exhibit dishonest academic behavior are more likely to violate workplace policies.9 Also, these behaviors lead to increased moral disengagement in all areas.10,11 Imagining a morally disengaged attending psychiatrist practicing medicine and training the next generation of psychiatrists is unsettling.
My hope is that residency programs discourage this detrimental conduct in their departments and support those trying to uphold integrity.
1. Scher S, Kozlowska K. Teaching ethics in psychiatry: time to reset. Harv Rev Psychiatry. 2020;28(5):328-333. doi:10.1097/HRP.0000000000000258
2. Allen NG, Khan JS, Alzahri MS, et al. Ethical issues in emergency psychiatry. Emerg Med Clin North Am. 2015;33(4):863-874. doi:10.1016/j.emc.2015.07.012
3. Pfleegor AG, Katz M, Bowers MT. Publish, perish, or salami slice? Authorship ethics in an emerging field. Journal of Business Ethics. 2019;156(1):189-208.
4. Rivera H. Fake peer review and inappropriate authorship are real evils. J Korean Med Sci. 2018;34(2):e6. doi:10.3346/jkms.2019.34.e6
5. Strange K. Authorship: why not just toss a coin? Am J Physiol Cell Physiol. 2008;295(3):C567-C575. doi:10.1152/ajpcell.00208.2008
6. Ali MJ. ICMJE criteria for authorship: why the criticisms are not justified? Graefes Arch Clin Exp Ophthalmol. 2021;259(2):289-290. doi:10.1007/s00417-020-04825-2
7. Malički M, Jerončić A, Marušić M, et al. Why do you think you should be the author on this manuscript? Analysis of open-ended responses of authors in a general medical journal. BMC Med Res Methodol. 2012;12:189. doi:10.1186/1471-2288-12-189
8. Kwok LS. The White Bull effect: abusive coauthorship and publication parasitism. J Med Ethics. 2005;31(9):554-556. doi:10.1136/jme.2004.010553
9. Harding TS, Carpenter DD, Finelli CJ, et al. Does academic dishonesty relate to unethical behavior in professional practice? An exploratory study. Sci Eng Ethics. 2004;10(2):311-324. doi:10.1007/s11948-004-0027-3
10. Shu LL, Gino F. Sweeping dishonesty under the rug: how unethical actions lead to forgetting of moral rules. J Pers Soc Psychol. 2012;102(6):1164-1177. doi:10.1037/a0028381
11. Shu LL, Gino F, Bazerman MH. Dishonest deed, clear conscience: when cheating leads to moral disengagement and motivated forgetting. Pers Soc Psychol Bull. 2011;37(3):330-349. doi:10.1177/0146167211398138
1. Scher S, Kozlowska K. Teaching ethics in psychiatry: time to reset. Harv Rev Psychiatry. 2020;28(5):328-333. doi:10.1097/HRP.0000000000000258
2. Allen NG, Khan JS, Alzahri MS, et al. Ethical issues in emergency psychiatry. Emerg Med Clin North Am. 2015;33(4):863-874. doi:10.1016/j.emc.2015.07.012
3. Pfleegor AG, Katz M, Bowers MT. Publish, perish, or salami slice? Authorship ethics in an emerging field. Journal of Business Ethics. 2019;156(1):189-208.
4. Rivera H. Fake peer review and inappropriate authorship are real evils. J Korean Med Sci. 2018;34(2):e6. doi:10.3346/jkms.2019.34.e6
5. Strange K. Authorship: why not just toss a coin? Am J Physiol Cell Physiol. 2008;295(3):C567-C575. doi:10.1152/ajpcell.00208.2008
6. Ali MJ. ICMJE criteria for authorship: why the criticisms are not justified? Graefes Arch Clin Exp Ophthalmol. 2021;259(2):289-290. doi:10.1007/s00417-020-04825-2
7. Malički M, Jerončić A, Marušić M, et al. Why do you think you should be the author on this manuscript? Analysis of open-ended responses of authors in a general medical journal. BMC Med Res Methodol. 2012;12:189. doi:10.1186/1471-2288-12-189
8. Kwok LS. The White Bull effect: abusive coauthorship and publication parasitism. J Med Ethics. 2005;31(9):554-556. doi:10.1136/jme.2004.010553
9. Harding TS, Carpenter DD, Finelli CJ, et al. Does academic dishonesty relate to unethical behavior in professional practice? An exploratory study. Sci Eng Ethics. 2004;10(2):311-324. doi:10.1007/s11948-004-0027-3
10. Shu LL, Gino F. Sweeping dishonesty under the rug: how unethical actions lead to forgetting of moral rules. J Pers Soc Psychol. 2012;102(6):1164-1177. doi:10.1037/a0028381
11. Shu LL, Gino F, Bazerman MH. Dishonest deed, clear conscience: when cheating leads to moral disengagement and motivated forgetting. Pers Soc Psychol Bull. 2011;37(3):330-349. doi:10.1177/0146167211398138
Erratic sleep, lack of activity tied to worsening schizophrenia symptoms
The findings also showed that people with schizophrenia spectrum disorders (SSDs) who lived in residential facilities experienced rigid routines, which correlated with a higher degree of negative symptoms.
The rigid routines were problematic for the patients living in residential settings, lead investigator Fabio Ferrarelli, MD, PhD, told this news organization. Dr. Ferrarelli is an associate professor of psychiatry at the University of Pittsburgh.
“Engaging in different activities at different times in activities associated with motivation and social interaction – this helps to ameliorate difficult-to-treat negative symptoms,” he said.
The findings were published online in Molecular Psychiatry.
Need to increase activity levels
While there is no shortage of research on sleep disturbances among people with schizophrenia, research focusing specifically on rest-activity rhythm disturbances and their relationships to symptoms of schizophrenia has been limited by small sample sizes or the lack of a control group, the investigators note.
To address this research gap, the investigators recruited 230 patients with SSD from participating residential facilities and communities throughout Italy. The participants included 108 healthy control participants, 54 community-dwelling patients with SSD who were receiving outpatient services, and 68 patients with SSD who were living in residential facilities.
All participants wore an actigraph for 7 consecutive days so that investigators could monitor sleep-wake patterns.
Compared with healthy control participants, both SSD groups had more total sleep time and spent more time resting or being passive (P < .001). In contrast, healthy control participants were much more active.
Part of the explanation for this may be that most of the control participants had jobs or attended school. In addition, the investigators note that many medications used to treat SSD can be highly sedating, causing some patients to sleep up to 15 hours per day.
Among residential participants with SSD, there was a higher level of inter-daily stability and higher daily rest-activity-rest fragmentation than occurred among healthy control participants or community-dwelling patients with SSD (P < .001). There was also a higher level of negative symptoms among residential participants with SSD than among the community-dwelling group with SSD.
When the findings were taken together, Dr. Ferrarelli and his team interpreted them to mean that inter-daily stability could reflect premature aging or neurodegenerative processes in patients with more severe forms of schizophrenia.
Another explanation could be that the rigid routine of the residential facility was making negative symptoms worse, Dr. Ferrarelli said. It is important to add variety into the mix – getting people to engage in different activities at different times of day would likely help residential SSD patients overcome negative symptoms of the disorder.
Although participants were recruited in Italy, Dr. Ferrarelli said he believes the findings are generalizable.
Bidirectional relationship?
Commenting on the findings, Matcheri Keshavan, MD, professor of psychiatry at Harvard Medical School in Boston, said the results are consistent with “well-known clinical observations that SSD patients tend to spend more time in bed and have more dysregulated sleep.
“Negative symptoms are also common, especially in residential patients. However, it is difficult to determine causality, as we do not know whether excessive sleepiness and decreased physical activity cause negative symptoms, or vice versa, or whether this is a bidirectional relationship,” Dr. Keshavan said.
He emphasized that physical exercise is known to increase sleep quality for people with mental illness and may also improve negative symptoms. “A useful approach in clinical practice is to increase activity levels, especially physical activities like walking and gardening.”
Dr. Keshavan said he would like to see future research that focuses on whether an intervention such as aerobic exercise would improve sleep quality as well as negative symptoms.
He also said that future research should ideally examine the characteristics of sleep alterations in schizophrenia.
“For example, while sleep duration is increased in schizophrenia, studies suggest that time spent in deep sleep is reduced; sleep spindles, which are important for consolidating memory during sleep, are also reduced. Correcting these deficits may improve negative symptoms and cognitive deficits,” he added.
The study was funded by the Italian Ministry of Health and the National Institute of Mental Health. There were no conflicts of interest.
A version of this article first appeared on Medscape.com.
The findings also showed that people with schizophrenia spectrum disorders (SSDs) who lived in residential facilities experienced rigid routines, which correlated with a higher degree of negative symptoms.
The rigid routines were problematic for the patients living in residential settings, lead investigator Fabio Ferrarelli, MD, PhD, told this news organization. Dr. Ferrarelli is an associate professor of psychiatry at the University of Pittsburgh.
“Engaging in different activities at different times in activities associated with motivation and social interaction – this helps to ameliorate difficult-to-treat negative symptoms,” he said.
The findings were published online in Molecular Psychiatry.
Need to increase activity levels
While there is no shortage of research on sleep disturbances among people with schizophrenia, research focusing specifically on rest-activity rhythm disturbances and their relationships to symptoms of schizophrenia has been limited by small sample sizes or the lack of a control group, the investigators note.
To address this research gap, the investigators recruited 230 patients with SSD from participating residential facilities and communities throughout Italy. The participants included 108 healthy control participants, 54 community-dwelling patients with SSD who were receiving outpatient services, and 68 patients with SSD who were living in residential facilities.
All participants wore an actigraph for 7 consecutive days so that investigators could monitor sleep-wake patterns.
Compared with healthy control participants, both SSD groups had more total sleep time and spent more time resting or being passive (P < .001). In contrast, healthy control participants were much more active.
Part of the explanation for this may be that most of the control participants had jobs or attended school. In addition, the investigators note that many medications used to treat SSD can be highly sedating, causing some patients to sleep up to 15 hours per day.
Among residential participants with SSD, there was a higher level of inter-daily stability and higher daily rest-activity-rest fragmentation than occurred among healthy control participants or community-dwelling patients with SSD (P < .001). There was also a higher level of negative symptoms among residential participants with SSD than among the community-dwelling group with SSD.
When the findings were taken together, Dr. Ferrarelli and his team interpreted them to mean that inter-daily stability could reflect premature aging or neurodegenerative processes in patients with more severe forms of schizophrenia.
Another explanation could be that the rigid routine of the residential facility was making negative symptoms worse, Dr. Ferrarelli said. It is important to add variety into the mix – getting people to engage in different activities at different times of day would likely help residential SSD patients overcome negative symptoms of the disorder.
Although participants were recruited in Italy, Dr. Ferrarelli said he believes the findings are generalizable.
Bidirectional relationship?
Commenting on the findings, Matcheri Keshavan, MD, professor of psychiatry at Harvard Medical School in Boston, said the results are consistent with “well-known clinical observations that SSD patients tend to spend more time in bed and have more dysregulated sleep.
“Negative symptoms are also common, especially in residential patients. However, it is difficult to determine causality, as we do not know whether excessive sleepiness and decreased physical activity cause negative symptoms, or vice versa, or whether this is a bidirectional relationship,” Dr. Keshavan said.
He emphasized that physical exercise is known to increase sleep quality for people with mental illness and may also improve negative symptoms. “A useful approach in clinical practice is to increase activity levels, especially physical activities like walking and gardening.”
Dr. Keshavan said he would like to see future research that focuses on whether an intervention such as aerobic exercise would improve sleep quality as well as negative symptoms.
He also said that future research should ideally examine the characteristics of sleep alterations in schizophrenia.
“For example, while sleep duration is increased in schizophrenia, studies suggest that time spent in deep sleep is reduced; sleep spindles, which are important for consolidating memory during sleep, are also reduced. Correcting these deficits may improve negative symptoms and cognitive deficits,” he added.
The study was funded by the Italian Ministry of Health and the National Institute of Mental Health. There were no conflicts of interest.
A version of this article first appeared on Medscape.com.
The findings also showed that people with schizophrenia spectrum disorders (SSDs) who lived in residential facilities experienced rigid routines, which correlated with a higher degree of negative symptoms.
The rigid routines were problematic for the patients living in residential settings, lead investigator Fabio Ferrarelli, MD, PhD, told this news organization. Dr. Ferrarelli is an associate professor of psychiatry at the University of Pittsburgh.
“Engaging in different activities at different times in activities associated with motivation and social interaction – this helps to ameliorate difficult-to-treat negative symptoms,” he said.
The findings were published online in Molecular Psychiatry.
Need to increase activity levels
While there is no shortage of research on sleep disturbances among people with schizophrenia, research focusing specifically on rest-activity rhythm disturbances and their relationships to symptoms of schizophrenia has been limited by small sample sizes or the lack of a control group, the investigators note.
To address this research gap, the investigators recruited 230 patients with SSD from participating residential facilities and communities throughout Italy. The participants included 108 healthy control participants, 54 community-dwelling patients with SSD who were receiving outpatient services, and 68 patients with SSD who were living in residential facilities.
All participants wore an actigraph for 7 consecutive days so that investigators could monitor sleep-wake patterns.
Compared with healthy control participants, both SSD groups had more total sleep time and spent more time resting or being passive (P < .001). In contrast, healthy control participants were much more active.
Part of the explanation for this may be that most of the control participants had jobs or attended school. In addition, the investigators note that many medications used to treat SSD can be highly sedating, causing some patients to sleep up to 15 hours per day.
Among residential participants with SSD, there was a higher level of inter-daily stability and higher daily rest-activity-rest fragmentation than occurred among healthy control participants or community-dwelling patients with SSD (P < .001). There was also a higher level of negative symptoms among residential participants with SSD than among the community-dwelling group with SSD.
When the findings were taken together, Dr. Ferrarelli and his team interpreted them to mean that inter-daily stability could reflect premature aging or neurodegenerative processes in patients with more severe forms of schizophrenia.
Another explanation could be that the rigid routine of the residential facility was making negative symptoms worse, Dr. Ferrarelli said. It is important to add variety into the mix – getting people to engage in different activities at different times of day would likely help residential SSD patients overcome negative symptoms of the disorder.
Although participants were recruited in Italy, Dr. Ferrarelli said he believes the findings are generalizable.
Bidirectional relationship?
Commenting on the findings, Matcheri Keshavan, MD, professor of psychiatry at Harvard Medical School in Boston, said the results are consistent with “well-known clinical observations that SSD patients tend to spend more time in bed and have more dysregulated sleep.
“Negative symptoms are also common, especially in residential patients. However, it is difficult to determine causality, as we do not know whether excessive sleepiness and decreased physical activity cause negative symptoms, or vice versa, or whether this is a bidirectional relationship,” Dr. Keshavan said.
He emphasized that physical exercise is known to increase sleep quality for people with mental illness and may also improve negative symptoms. “A useful approach in clinical practice is to increase activity levels, especially physical activities like walking and gardening.”
Dr. Keshavan said he would like to see future research that focuses on whether an intervention such as aerobic exercise would improve sleep quality as well as negative symptoms.
He also said that future research should ideally examine the characteristics of sleep alterations in schizophrenia.
“For example, while sleep duration is increased in schizophrenia, studies suggest that time spent in deep sleep is reduced; sleep spindles, which are important for consolidating memory during sleep, are also reduced. Correcting these deficits may improve negative symptoms and cognitive deficits,” he added.
The study was funded by the Italian Ministry of Health and the National Institute of Mental Health. There were no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM MOLECULAR PSYCHIATRY
Drive, chip, and putt your way to osteoarthritis relief
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
‘Shocking’ data on what’s really in melatonin gummies
New data may explain the recent massive jump in pediatric hospitalizations.
Thenvestigators found that consuming some products as directed could expose consumers, including children, to doses that are 40-130 times greater than what’s recommended.
“The results were quite shocking,” lead researcher Pieter Cohen, MD, with Harvard Medical School, Boston, and Cambridge Health Alliance, Somerville, Mass., said in an interview.
“Melatonin gummies contained up to 347% more melatonin than what was listed on the label, and some products also contained cannabidiol; in one brand of melatonin gummies, there was zero melatonin, just CBD,” Dr. Cohen said.
The study was published online in JAMA.
530% jump in pediatric hospitalizations
Melatonin products are not approved by the Food and Drug Administration but are sold over the counter or online.
Previous research from JAMA has shown the use of melatonin has increased over the past 2 decades among people of all ages.
With increased use has come a spike in reports of melatonin overdose, calls to poison control centers, and related ED visits for children.
Federal data show the number of U.S. children who unintentionally ingested melatonin supplements jumped 530% from 2012 to 2021. More than 4,000 of the reported ingestions led to a hospital stay; 287 children required intensive care, and two children died.
It was unclear why melatonin supplements were causing these harms, which led Dr. Cohen’s team to analyze 25 unique brands of “melatonin” gummies purchased online.
One product didn’t contain any melatonin but did contain 31.3 mg of CBD.
In the remaining products, the quantity of melatonin ranged from 1.3 mg to 13.1 mg per serving. The actual quantity of melatonin ranged from 74% to 347% of the labeled quantity, the researchers found.
They note that for a young adult who takes as little as 0.1-0.3 mg of melatonin, plasma concentrations can increase into the normal night-time range.
Of the 25 products (88%) analyzed, 22 were inaccurately labeled, and only 3 (12%) contained a quantity of melatonin that was within 10% (plus or minus) of the declared quantity.
Five products listed CBD as an ingredient. The listed quantity ranged from 10.6 mg to 31.3 mg per serving, although the actual quantity of CBD ranged from 104% to 118% of the labeled quantity.
Inquire about use in kids
A limitation of the study is that only one sample of each brand was analyzed, and only gummies were analyzed. It is not known whether the results are generalizable to melatonin products sold as tablets and capsules in the United States or whether the quantity of melatonin within an individual brand may vary from batch to batch.
A recent study from Canada showed similar results. In an analysis of 16 Canadian melatonin brands, the actual dose of melatonin ranged from 17% to 478% of the declared quantity.
It’s estimated that more than 1% of all U.S. children use melatonin supplements, most commonly for sleep, stress, and relaxation.
“Given new research as to the excessive quantities of melatonin in gummies, caution should be used if considering their use,” said Dr. Cohen.
“It’s important to inquire about melatonin use when caring for children, particularly when parents express concerns about their child’s sleep,” he added.
The American Academy of Sleep Medicine recently issued a health advisory encouraging parents to talk to a health care professional before giving melatonin or any supplement to children.
Children don’t need melatonin
Commenting on the study, Michael Breus, PhD, clinical psychologist and founder of TheSleepDoctor.com, agreed that analyzing only one sample of each brand is a key limitation “because supplements are made in batches, and gummies in particular are difficult to distribute the active ingredient evenly.
“But even with that being said, 88% of them were labeled incorrectly, so even if there were a few single-sample issues, I kind of doubt its all of them,” Dr. Breus said.
“Kids as a general rule do not need melatonin. Their brains make almost four times the necessary amount already. If you start giving kids pills to help them sleep, then they start to have a pill problem, causing another issue,” Dr. Breus added.
“Most children’s falling asleep and staying sleep issues can be treated with behavioral measures like cognitive-behavioral therapy for insomnia,” he said.
The study had no specific funding. Dr. Cohen has received research support from Consumers Union and PEW Charitable Trusts and royalties from UptoDate. Dr. Breus disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New data may explain the recent massive jump in pediatric hospitalizations.
Thenvestigators found that consuming some products as directed could expose consumers, including children, to doses that are 40-130 times greater than what’s recommended.
“The results were quite shocking,” lead researcher Pieter Cohen, MD, with Harvard Medical School, Boston, and Cambridge Health Alliance, Somerville, Mass., said in an interview.
“Melatonin gummies contained up to 347% more melatonin than what was listed on the label, and some products also contained cannabidiol; in one brand of melatonin gummies, there was zero melatonin, just CBD,” Dr. Cohen said.
The study was published online in JAMA.
530% jump in pediatric hospitalizations
Melatonin products are not approved by the Food and Drug Administration but are sold over the counter or online.
Previous research from JAMA has shown the use of melatonin has increased over the past 2 decades among people of all ages.
With increased use has come a spike in reports of melatonin overdose, calls to poison control centers, and related ED visits for children.
Federal data show the number of U.S. children who unintentionally ingested melatonin supplements jumped 530% from 2012 to 2021. More than 4,000 of the reported ingestions led to a hospital stay; 287 children required intensive care, and two children died.
It was unclear why melatonin supplements were causing these harms, which led Dr. Cohen’s team to analyze 25 unique brands of “melatonin” gummies purchased online.
One product didn’t contain any melatonin but did contain 31.3 mg of CBD.
In the remaining products, the quantity of melatonin ranged from 1.3 mg to 13.1 mg per serving. The actual quantity of melatonin ranged from 74% to 347% of the labeled quantity, the researchers found.
They note that for a young adult who takes as little as 0.1-0.3 mg of melatonin, plasma concentrations can increase into the normal night-time range.
Of the 25 products (88%) analyzed, 22 were inaccurately labeled, and only 3 (12%) contained a quantity of melatonin that was within 10% (plus or minus) of the declared quantity.
Five products listed CBD as an ingredient. The listed quantity ranged from 10.6 mg to 31.3 mg per serving, although the actual quantity of CBD ranged from 104% to 118% of the labeled quantity.
Inquire about use in kids
A limitation of the study is that only one sample of each brand was analyzed, and only gummies were analyzed. It is not known whether the results are generalizable to melatonin products sold as tablets and capsules in the United States or whether the quantity of melatonin within an individual brand may vary from batch to batch.
A recent study from Canada showed similar results. In an analysis of 16 Canadian melatonin brands, the actual dose of melatonin ranged from 17% to 478% of the declared quantity.
It’s estimated that more than 1% of all U.S. children use melatonin supplements, most commonly for sleep, stress, and relaxation.
“Given new research as to the excessive quantities of melatonin in gummies, caution should be used if considering their use,” said Dr. Cohen.
“It’s important to inquire about melatonin use when caring for children, particularly when parents express concerns about their child’s sleep,” he added.
The American Academy of Sleep Medicine recently issued a health advisory encouraging parents to talk to a health care professional before giving melatonin or any supplement to children.
Children don’t need melatonin
Commenting on the study, Michael Breus, PhD, clinical psychologist and founder of TheSleepDoctor.com, agreed that analyzing only one sample of each brand is a key limitation “because supplements are made in batches, and gummies in particular are difficult to distribute the active ingredient evenly.
“But even with that being said, 88% of them were labeled incorrectly, so even if there were a few single-sample issues, I kind of doubt its all of them,” Dr. Breus said.
“Kids as a general rule do not need melatonin. Their brains make almost four times the necessary amount already. If you start giving kids pills to help them sleep, then they start to have a pill problem, causing another issue,” Dr. Breus added.
“Most children’s falling asleep and staying sleep issues can be treated with behavioral measures like cognitive-behavioral therapy for insomnia,” he said.
The study had no specific funding. Dr. Cohen has received research support from Consumers Union and PEW Charitable Trusts and royalties from UptoDate. Dr. Breus disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New data may explain the recent massive jump in pediatric hospitalizations.
Thenvestigators found that consuming some products as directed could expose consumers, including children, to doses that are 40-130 times greater than what’s recommended.
“The results were quite shocking,” lead researcher Pieter Cohen, MD, with Harvard Medical School, Boston, and Cambridge Health Alliance, Somerville, Mass., said in an interview.
“Melatonin gummies contained up to 347% more melatonin than what was listed on the label, and some products also contained cannabidiol; in one brand of melatonin gummies, there was zero melatonin, just CBD,” Dr. Cohen said.
The study was published online in JAMA.
530% jump in pediatric hospitalizations
Melatonin products are not approved by the Food and Drug Administration but are sold over the counter or online.
Previous research from JAMA has shown the use of melatonin has increased over the past 2 decades among people of all ages.
With increased use has come a spike in reports of melatonin overdose, calls to poison control centers, and related ED visits for children.
Federal data show the number of U.S. children who unintentionally ingested melatonin supplements jumped 530% from 2012 to 2021. More than 4,000 of the reported ingestions led to a hospital stay; 287 children required intensive care, and two children died.
It was unclear why melatonin supplements were causing these harms, which led Dr. Cohen’s team to analyze 25 unique brands of “melatonin” gummies purchased online.
One product didn’t contain any melatonin but did contain 31.3 mg of CBD.
In the remaining products, the quantity of melatonin ranged from 1.3 mg to 13.1 mg per serving. The actual quantity of melatonin ranged from 74% to 347% of the labeled quantity, the researchers found.
They note that for a young adult who takes as little as 0.1-0.3 mg of melatonin, plasma concentrations can increase into the normal night-time range.
Of the 25 products (88%) analyzed, 22 were inaccurately labeled, and only 3 (12%) contained a quantity of melatonin that was within 10% (plus or minus) of the declared quantity.
Five products listed CBD as an ingredient. The listed quantity ranged from 10.6 mg to 31.3 mg per serving, although the actual quantity of CBD ranged from 104% to 118% of the labeled quantity.
Inquire about use in kids
A limitation of the study is that only one sample of each brand was analyzed, and only gummies were analyzed. It is not known whether the results are generalizable to melatonin products sold as tablets and capsules in the United States or whether the quantity of melatonin within an individual brand may vary from batch to batch.
A recent study from Canada showed similar results. In an analysis of 16 Canadian melatonin brands, the actual dose of melatonin ranged from 17% to 478% of the declared quantity.
It’s estimated that more than 1% of all U.S. children use melatonin supplements, most commonly for sleep, stress, and relaxation.
“Given new research as to the excessive quantities of melatonin in gummies, caution should be used if considering their use,” said Dr. Cohen.
“It’s important to inquire about melatonin use when caring for children, particularly when parents express concerns about their child’s sleep,” he added.
The American Academy of Sleep Medicine recently issued a health advisory encouraging parents to talk to a health care professional before giving melatonin or any supplement to children.
Children don’t need melatonin
Commenting on the study, Michael Breus, PhD, clinical psychologist and founder of TheSleepDoctor.com, agreed that analyzing only one sample of each brand is a key limitation “because supplements are made in batches, and gummies in particular are difficult to distribute the active ingredient evenly.
“But even with that being said, 88% of them were labeled incorrectly, so even if there were a few single-sample issues, I kind of doubt its all of them,” Dr. Breus said.
“Kids as a general rule do not need melatonin. Their brains make almost four times the necessary amount already. If you start giving kids pills to help them sleep, then they start to have a pill problem, causing another issue,” Dr. Breus added.
“Most children’s falling asleep and staying sleep issues can be treated with behavioral measures like cognitive-behavioral therapy for insomnia,” he said.
The study had no specific funding. Dr. Cohen has received research support from Consumers Union and PEW Charitable Trusts and royalties from UptoDate. Dr. Breus disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA
The newest form of mommy shaming: The 'narcissistic mother'
Narcissists appear to be everywhere. A few minutes on the Internet shows the dangers of narcissistic romantic partners, friends, and employers. Identifying and limiting the reach of their manipulative and self-centered endeavors is cast as both urgent and necessary. The destructive powers of the narcissistic mother are viewed as especially in need of remedy, and any bookstore can reveal the risks they pose: “Will I Ever Be Good Enough? Healing the Daughters of Narcissistic Mothers;” “You’re Not Crazy – It’s Your Mother: Freedom for Daughters of Narcissistic Mothers;” “Healing for Daughters of Narcissistic Mothers: A Practical Guide on How to Recover from the Childhood Trauma of Toxic Relationship with Your Mother and How You Can Handle Her Abuse Now As An Adult” – to name just a few (there are more).
As a psychologist specializing in parental estrangement, I (Dr. Coleman) regularly see letters from adult children explaining their discovery-through-therapy that their mother is a narcissist. The proclamation often comes when the therapist has never met the mother. Typically, the discovery is presented as a justification for ending the relationship with the parent. While these mothers could rightly be accused of being anxious, over-involved, depressed, or hurt by the lack of gratitude or reciprocity, the vast majority are not narcissists.
Which begs the question, why are so many being labeled in this way? Are therapists only now discovering the power of narcissistic mothers? Have they always existed, casting their spells upon unwary children? Are those now-grown children only today able to disentangle themselves from the longstanding, pervasive, and harmful influence of these parents, with the help of therapy? Or is this the newest form of mommy shaming as it engages head-on with our Diagnostic and Statistical Manuals?
We believe it is the latter.
Blaming mothers has a long reach. Mothers have been blamed for causing schizophrenia, autism, homosexuality, and effeminacy in men. While we used to call people selfish and “controlling,” narcissism is a more consequential label as it confers diagnostic validity from the mental health profession. Worse, it suggests an individual beyond reach, where the only answer is distance, containment, or estrangement.
The rise of the narcissistic mother comes during a time when, for the past 4 decades, the average working mother spends more time with her children than stay-at-home moms did in the supposed halcyon days of the 1960s’ middle class, before “parenting” was a common term. A variety of economists and sociologists observed that an increase in parental effort became necessary to launch children into adulthood given the retreat of governmental and corporate support for parents that began in the 1980s.
“The financial and emotional burden on families has grown in ways that were almost unimaginable just a half-century ago,” writes the University of Pennsylvania sociologist Frank Furstenberg in “On a New Schedule: Transitions to Adulthood and Family Change.” In addition, a view of children as vulnerable and in need of intense parental investment gained momentum over the course of the 20th century and has continued unabated into the present. As a result, an environment of intense maternal preoccupation, worry, guilt, and involvement with children’s grades, safety, health, and emotional states – referred to as “helicopter” and “tiger” mothering – grew into the norm across the classes.
While prior generations of parents could, by today’s standards, be viewed as being insufficiently involved, today’s parents have become “over-involved” – aided by the ability of parents to be in constant contact with their adult children through technology. While this shift to a more hands-on, more conscientious parenting has been a boon to parent–adult child relationships in the main, the downside has meant, for some, too much of a good thing. From that perspective, pathologizing a mother’s involvement or her expressions of hurt for that child’s lack of availability provides a shield against the child’s feelings of guilt or obligation.
Diagnoses can serve a social purpose: They can allow individuals to use the authority of our profession to decide who to be close to and who to let go. They can provide insulation against feelings of obligation or guilt. They create a way to label behavior as dysfunctional that in other eras or cultures would be considered normal, even valued. To that extent, diagnoses don’t occur in a cultural void. They are inextricably tied to larger ideals, be they individualistic – as exists in the United States – or collectivist, as exists in many other parts of the world.
While we have decided what parents owe our children, it is unclear what parents might ask in return. To that end, mothers who want more interest, availability, or gratitude today are vulnerable to being cast as selfish, uncaring, needy, and controlling. They can now be viewed as failing in their task of selfless devotion. Their desires for closeness or repair can be regarded as incompatible with the quest for the adult child’s self-fulfillment and identity; her identification with her children too great a barrier to their individuation.
There may well be good reasons to estrange family members for their intolerable behaviors, especially ones who have threatened personal safety. Yet, while there are plenty of problematic parents, few meet the diagnostic criteria of narcissistic personality disorder. More important, such labels can discourage a discussion of boundaries that both the parents and the adult children might find acceptable – which sometimes means asking family members to tolerate behavior or individuals not to their liking.
Diagnoses carry enormous social weight and can facilitate estrangements or negativity to mothers that are far more workable than our patients’ characterization of them might lead them or us to believe. ; it devalues their years of love and dedication, however flawed; and it weakens the fabric of connection that could otherwise exist. Rather than provide a path toward compassion or understanding, “narcissistic mother” just becomes the latest form of mommy shaming.
Dr. Coleman is a clinical psychologist and author of “Rules of Estrangement: Why Adult Children Cut Ties and How to Heal the Conflict” (New York: Penguin Random House, 2021). Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is an assistant professor of psychiatry and behavioral sciences at Johns Hopkins University in Baltimore.
Narcissists appear to be everywhere. A few minutes on the Internet shows the dangers of narcissistic romantic partners, friends, and employers. Identifying and limiting the reach of their manipulative and self-centered endeavors is cast as both urgent and necessary. The destructive powers of the narcissistic mother are viewed as especially in need of remedy, and any bookstore can reveal the risks they pose: “Will I Ever Be Good Enough? Healing the Daughters of Narcissistic Mothers;” “You’re Not Crazy – It’s Your Mother: Freedom for Daughters of Narcissistic Mothers;” “Healing for Daughters of Narcissistic Mothers: A Practical Guide on How to Recover from the Childhood Trauma of Toxic Relationship with Your Mother and How You Can Handle Her Abuse Now As An Adult” – to name just a few (there are more).
As a psychologist specializing in parental estrangement, I (Dr. Coleman) regularly see letters from adult children explaining their discovery-through-therapy that their mother is a narcissist. The proclamation often comes when the therapist has never met the mother. Typically, the discovery is presented as a justification for ending the relationship with the parent. While these mothers could rightly be accused of being anxious, over-involved, depressed, or hurt by the lack of gratitude or reciprocity, the vast majority are not narcissists.
Which begs the question, why are so many being labeled in this way? Are therapists only now discovering the power of narcissistic mothers? Have they always existed, casting their spells upon unwary children? Are those now-grown children only today able to disentangle themselves from the longstanding, pervasive, and harmful influence of these parents, with the help of therapy? Or is this the newest form of mommy shaming as it engages head-on with our Diagnostic and Statistical Manuals?
We believe it is the latter.
Blaming mothers has a long reach. Mothers have been blamed for causing schizophrenia, autism, homosexuality, and effeminacy in men. While we used to call people selfish and “controlling,” narcissism is a more consequential label as it confers diagnostic validity from the mental health profession. Worse, it suggests an individual beyond reach, where the only answer is distance, containment, or estrangement.
The rise of the narcissistic mother comes during a time when, for the past 4 decades, the average working mother spends more time with her children than stay-at-home moms did in the supposed halcyon days of the 1960s’ middle class, before “parenting” was a common term. A variety of economists and sociologists observed that an increase in parental effort became necessary to launch children into adulthood given the retreat of governmental and corporate support for parents that began in the 1980s.
“The financial and emotional burden on families has grown in ways that were almost unimaginable just a half-century ago,” writes the University of Pennsylvania sociologist Frank Furstenberg in “On a New Schedule: Transitions to Adulthood and Family Change.” In addition, a view of children as vulnerable and in need of intense parental investment gained momentum over the course of the 20th century and has continued unabated into the present. As a result, an environment of intense maternal preoccupation, worry, guilt, and involvement with children’s grades, safety, health, and emotional states – referred to as “helicopter” and “tiger” mothering – grew into the norm across the classes.
While prior generations of parents could, by today’s standards, be viewed as being insufficiently involved, today’s parents have become “over-involved” – aided by the ability of parents to be in constant contact with their adult children through technology. While this shift to a more hands-on, more conscientious parenting has been a boon to parent–adult child relationships in the main, the downside has meant, for some, too much of a good thing. From that perspective, pathologizing a mother’s involvement or her expressions of hurt for that child’s lack of availability provides a shield against the child’s feelings of guilt or obligation.
Diagnoses can serve a social purpose: They can allow individuals to use the authority of our profession to decide who to be close to and who to let go. They can provide insulation against feelings of obligation or guilt. They create a way to label behavior as dysfunctional that in other eras or cultures would be considered normal, even valued. To that extent, diagnoses don’t occur in a cultural void. They are inextricably tied to larger ideals, be they individualistic – as exists in the United States – or collectivist, as exists in many other parts of the world.
While we have decided what parents owe our children, it is unclear what parents might ask in return. To that end, mothers who want more interest, availability, or gratitude today are vulnerable to being cast as selfish, uncaring, needy, and controlling. They can now be viewed as failing in their task of selfless devotion. Their desires for closeness or repair can be regarded as incompatible with the quest for the adult child’s self-fulfillment and identity; her identification with her children too great a barrier to their individuation.
There may well be good reasons to estrange family members for their intolerable behaviors, especially ones who have threatened personal safety. Yet, while there are plenty of problematic parents, few meet the diagnostic criteria of narcissistic personality disorder. More important, such labels can discourage a discussion of boundaries that both the parents and the adult children might find acceptable – which sometimes means asking family members to tolerate behavior or individuals not to their liking.
Diagnoses carry enormous social weight and can facilitate estrangements or negativity to mothers that are far more workable than our patients’ characterization of them might lead them or us to believe. ; it devalues their years of love and dedication, however flawed; and it weakens the fabric of connection that could otherwise exist. Rather than provide a path toward compassion or understanding, “narcissistic mother” just becomes the latest form of mommy shaming.
Dr. Coleman is a clinical psychologist and author of “Rules of Estrangement: Why Adult Children Cut Ties and How to Heal the Conflict” (New York: Penguin Random House, 2021). Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is an assistant professor of psychiatry and behavioral sciences at Johns Hopkins University in Baltimore.
Narcissists appear to be everywhere. A few minutes on the Internet shows the dangers of narcissistic romantic partners, friends, and employers. Identifying and limiting the reach of their manipulative and self-centered endeavors is cast as both urgent and necessary. The destructive powers of the narcissistic mother are viewed as especially in need of remedy, and any bookstore can reveal the risks they pose: “Will I Ever Be Good Enough? Healing the Daughters of Narcissistic Mothers;” “You’re Not Crazy – It’s Your Mother: Freedom for Daughters of Narcissistic Mothers;” “Healing for Daughters of Narcissistic Mothers: A Practical Guide on How to Recover from the Childhood Trauma of Toxic Relationship with Your Mother and How You Can Handle Her Abuse Now As An Adult” – to name just a few (there are more).
As a psychologist specializing in parental estrangement, I (Dr. Coleman) regularly see letters from adult children explaining their discovery-through-therapy that their mother is a narcissist. The proclamation often comes when the therapist has never met the mother. Typically, the discovery is presented as a justification for ending the relationship with the parent. While these mothers could rightly be accused of being anxious, over-involved, depressed, or hurt by the lack of gratitude or reciprocity, the vast majority are not narcissists.
Which begs the question, why are so many being labeled in this way? Are therapists only now discovering the power of narcissistic mothers? Have they always existed, casting their spells upon unwary children? Are those now-grown children only today able to disentangle themselves from the longstanding, pervasive, and harmful influence of these parents, with the help of therapy? Or is this the newest form of mommy shaming as it engages head-on with our Diagnostic and Statistical Manuals?
We believe it is the latter.
Blaming mothers has a long reach. Mothers have been blamed for causing schizophrenia, autism, homosexuality, and effeminacy in men. While we used to call people selfish and “controlling,” narcissism is a more consequential label as it confers diagnostic validity from the mental health profession. Worse, it suggests an individual beyond reach, where the only answer is distance, containment, or estrangement.
The rise of the narcissistic mother comes during a time when, for the past 4 decades, the average working mother spends more time with her children than stay-at-home moms did in the supposed halcyon days of the 1960s’ middle class, before “parenting” was a common term. A variety of economists and sociologists observed that an increase in parental effort became necessary to launch children into adulthood given the retreat of governmental and corporate support for parents that began in the 1980s.
“The financial and emotional burden on families has grown in ways that were almost unimaginable just a half-century ago,” writes the University of Pennsylvania sociologist Frank Furstenberg in “On a New Schedule: Transitions to Adulthood and Family Change.” In addition, a view of children as vulnerable and in need of intense parental investment gained momentum over the course of the 20th century and has continued unabated into the present. As a result, an environment of intense maternal preoccupation, worry, guilt, and involvement with children’s grades, safety, health, and emotional states – referred to as “helicopter” and “tiger” mothering – grew into the norm across the classes.
While prior generations of parents could, by today’s standards, be viewed as being insufficiently involved, today’s parents have become “over-involved” – aided by the ability of parents to be in constant contact with their adult children through technology. While this shift to a more hands-on, more conscientious parenting has been a boon to parent–adult child relationships in the main, the downside has meant, for some, too much of a good thing. From that perspective, pathologizing a mother’s involvement or her expressions of hurt for that child’s lack of availability provides a shield against the child’s feelings of guilt or obligation.
Diagnoses can serve a social purpose: They can allow individuals to use the authority of our profession to decide who to be close to and who to let go. They can provide insulation against feelings of obligation or guilt. They create a way to label behavior as dysfunctional that in other eras or cultures would be considered normal, even valued. To that extent, diagnoses don’t occur in a cultural void. They are inextricably tied to larger ideals, be they individualistic – as exists in the United States – or collectivist, as exists in many other parts of the world.
While we have decided what parents owe our children, it is unclear what parents might ask in return. To that end, mothers who want more interest, availability, or gratitude today are vulnerable to being cast as selfish, uncaring, needy, and controlling. They can now be viewed as failing in their task of selfless devotion. Their desires for closeness or repair can be regarded as incompatible with the quest for the adult child’s self-fulfillment and identity; her identification with her children too great a barrier to their individuation.
There may well be good reasons to estrange family members for their intolerable behaviors, especially ones who have threatened personal safety. Yet, while there are plenty of problematic parents, few meet the diagnostic criteria of narcissistic personality disorder. More important, such labels can discourage a discussion of boundaries that both the parents and the adult children might find acceptable – which sometimes means asking family members to tolerate behavior or individuals not to their liking.
Diagnoses carry enormous social weight and can facilitate estrangements or negativity to mothers that are far more workable than our patients’ characterization of them might lead them or us to believe. ; it devalues their years of love and dedication, however flawed; and it weakens the fabric of connection that could otherwise exist. Rather than provide a path toward compassion or understanding, “narcissistic mother” just becomes the latest form of mommy shaming.
Dr. Coleman is a clinical psychologist and author of “Rules of Estrangement: Why Adult Children Cut Ties and How to Heal the Conflict” (New York: Penguin Random House, 2021). Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is an assistant professor of psychiatry and behavioral sciences at Johns Hopkins University in Baltimore.
Strong need for eating disorder screening in patients with PTSD
WASHINGTON –
“Eating-related and body-image concerns may be more prevalent than we think, and if not considered, these concerns can make psychotherapy treatment less effective,” study author Nick Powers, a doctoral student in clinical psychology, La Salle University, Philadelphia, told this news organization.
The findings were presented as part of the Anxiety and Depression Association of America Anxiety & Depression conference.
Common bedfellows
Although many patients with PTSD also have an eating disorder, they are not always properly assessed for eating pathology and related functional impairment.
Some therapists don’t feel adequately equipped to target eating-related concerns in these patients and so may refer them to other providers. This, said Mr. Powers, can prolong symptoms and further distress patients.
Mr. Powers noted childhood physical or sexual abuse may affect eating patterns in patients with PTSD. “The evidence suggests these types of trauma exposure can be risk factors for the development of an eating disorder.”
Undiagnosed eating pathology may exacerbate functional impairment from PTSD and weaken the impact of evidence-based treatment.
Such patients are challenging to treat as they may not have the requisite skills to fully engage in exposure therapy, an evidence-based approach to treat PTSD, said Mr. Powers.
To determine whether PTSD would be significantly linked to greater eating disorder impairment (EDI) compared with other anxiety-related diagnoses and whether this would impair treatment, investigators studied 748 patients with an anxiety disorder who were attending a cognitive behavioral therapy (CBT) clinic. Anxiety disorders included PTSD, obsessive-compulsive disorder (OCD), social anxiety, and panic disorder.
Participants completed the 16-item Clinical Impairment Assessment (CIA) questionnaire, which includes questions about eating habits and feelings about food, body shape, and weight over the previous 4 weeks. Participants also reported anxiety symptom severity at the beginning, during, and end of treatment.
Need for better screening
Results showed that compared with those who had other anxiety disorders, patients with PTSD were three times more likely to have disordered eating (odds ratio [OR], 3.06; 95% confidence interval [CI], 1.47-6.37; P = .003).
In addition, higher baseline CIA scores predicted poorer PTSD treatment outcome (beta = –1.4; 95% CI, –1.67 to –1.10; P < .01).
“Having higher baseline CIA scores meant that patients’ PTSD symptoms did not remit as strongly compared to those with lower scores,” said Mr. Powers.
Patients with both PTSD and an eating disorder may have difficulty with regulating emotions and tolerating distress, he said.
“They may use binge eating, purging, or food restriction as strategies to regulate emotions. These behaviors may allow patients to become numb to or avoid heightened emotions that come from having PTSD and an eating disorder.”
Prior research linked perfectionism tendencies to poorer response to PTSD treatment. Those with an eating disorder may share similar tendencies, said Mr. Powers.
“If someone is consistently thinking negatively about their eating or body to the point where it interrupts their functioning, they may not be as likely to fully engage with PTSD treatment,” he said.
Ideally, clinicians would screen all patients with PTSD for an eating disorder, said Mr. Powers. “If screening instruments aren’t feasible or available, even just inquiring about body image or history of maladaptive eating behaviors can be helpful.”
He added this could open up a conversation about a traumatic event in the patient’s past.
Confirmatory research
Commenting on the study, Karen S. Mitchell, PhD, clinical research psychologist, National Center for PTSD, VA Boston Healthcare System, and associate professor in psychiatry, Boston University, said she was “excited” to see this research.
“Very few studies have examined the impact of baseline eating disorder symptoms on PTSD treatment outcomes or vice versa,” she said.
The study findings “add to the small but growing body of evidence suggesting that comorbid PTSD and eating disorder symptoms can impact recovery from each disorder,” she said.
She noted the importance of assessing comorbidity in patients presenting for treatment and of addressing comorbidity in both eating disorders and PTSD treatment. “But we need more research on how best to do this.”
Mr. Powers and Dr. Mitchell have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON –
“Eating-related and body-image concerns may be more prevalent than we think, and if not considered, these concerns can make psychotherapy treatment less effective,” study author Nick Powers, a doctoral student in clinical psychology, La Salle University, Philadelphia, told this news organization.
The findings were presented as part of the Anxiety and Depression Association of America Anxiety & Depression conference.
Common bedfellows
Although many patients with PTSD also have an eating disorder, they are not always properly assessed for eating pathology and related functional impairment.
Some therapists don’t feel adequately equipped to target eating-related concerns in these patients and so may refer them to other providers. This, said Mr. Powers, can prolong symptoms and further distress patients.
Mr. Powers noted childhood physical or sexual abuse may affect eating patterns in patients with PTSD. “The evidence suggests these types of trauma exposure can be risk factors for the development of an eating disorder.”
Undiagnosed eating pathology may exacerbate functional impairment from PTSD and weaken the impact of evidence-based treatment.
Such patients are challenging to treat as they may not have the requisite skills to fully engage in exposure therapy, an evidence-based approach to treat PTSD, said Mr. Powers.
To determine whether PTSD would be significantly linked to greater eating disorder impairment (EDI) compared with other anxiety-related diagnoses and whether this would impair treatment, investigators studied 748 patients with an anxiety disorder who were attending a cognitive behavioral therapy (CBT) clinic. Anxiety disorders included PTSD, obsessive-compulsive disorder (OCD), social anxiety, and panic disorder.
Participants completed the 16-item Clinical Impairment Assessment (CIA) questionnaire, which includes questions about eating habits and feelings about food, body shape, and weight over the previous 4 weeks. Participants also reported anxiety symptom severity at the beginning, during, and end of treatment.
Need for better screening
Results showed that compared with those who had other anxiety disorders, patients with PTSD were three times more likely to have disordered eating (odds ratio [OR], 3.06; 95% confidence interval [CI], 1.47-6.37; P = .003).
In addition, higher baseline CIA scores predicted poorer PTSD treatment outcome (beta = –1.4; 95% CI, –1.67 to –1.10; P < .01).
“Having higher baseline CIA scores meant that patients’ PTSD symptoms did not remit as strongly compared to those with lower scores,” said Mr. Powers.
Patients with both PTSD and an eating disorder may have difficulty with regulating emotions and tolerating distress, he said.
“They may use binge eating, purging, or food restriction as strategies to regulate emotions. These behaviors may allow patients to become numb to or avoid heightened emotions that come from having PTSD and an eating disorder.”
Prior research linked perfectionism tendencies to poorer response to PTSD treatment. Those with an eating disorder may share similar tendencies, said Mr. Powers.
“If someone is consistently thinking negatively about their eating or body to the point where it interrupts their functioning, they may not be as likely to fully engage with PTSD treatment,” he said.
Ideally, clinicians would screen all patients with PTSD for an eating disorder, said Mr. Powers. “If screening instruments aren’t feasible or available, even just inquiring about body image or history of maladaptive eating behaviors can be helpful.”
He added this could open up a conversation about a traumatic event in the patient’s past.
Confirmatory research
Commenting on the study, Karen S. Mitchell, PhD, clinical research psychologist, National Center for PTSD, VA Boston Healthcare System, and associate professor in psychiatry, Boston University, said she was “excited” to see this research.
“Very few studies have examined the impact of baseline eating disorder symptoms on PTSD treatment outcomes or vice versa,” she said.
The study findings “add to the small but growing body of evidence suggesting that comorbid PTSD and eating disorder symptoms can impact recovery from each disorder,” she said.
She noted the importance of assessing comorbidity in patients presenting for treatment and of addressing comorbidity in both eating disorders and PTSD treatment. “But we need more research on how best to do this.”
Mr. Powers and Dr. Mitchell have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON –
“Eating-related and body-image concerns may be more prevalent than we think, and if not considered, these concerns can make psychotherapy treatment less effective,” study author Nick Powers, a doctoral student in clinical psychology, La Salle University, Philadelphia, told this news organization.
The findings were presented as part of the Anxiety and Depression Association of America Anxiety & Depression conference.
Common bedfellows
Although many patients with PTSD also have an eating disorder, they are not always properly assessed for eating pathology and related functional impairment.
Some therapists don’t feel adequately equipped to target eating-related concerns in these patients and so may refer them to other providers. This, said Mr. Powers, can prolong symptoms and further distress patients.
Mr. Powers noted childhood physical or sexual abuse may affect eating patterns in patients with PTSD. “The evidence suggests these types of trauma exposure can be risk factors for the development of an eating disorder.”
Undiagnosed eating pathology may exacerbate functional impairment from PTSD and weaken the impact of evidence-based treatment.
Such patients are challenging to treat as they may not have the requisite skills to fully engage in exposure therapy, an evidence-based approach to treat PTSD, said Mr. Powers.
To determine whether PTSD would be significantly linked to greater eating disorder impairment (EDI) compared with other anxiety-related diagnoses and whether this would impair treatment, investigators studied 748 patients with an anxiety disorder who were attending a cognitive behavioral therapy (CBT) clinic. Anxiety disorders included PTSD, obsessive-compulsive disorder (OCD), social anxiety, and panic disorder.
Participants completed the 16-item Clinical Impairment Assessment (CIA) questionnaire, which includes questions about eating habits and feelings about food, body shape, and weight over the previous 4 weeks. Participants also reported anxiety symptom severity at the beginning, during, and end of treatment.
Need for better screening
Results showed that compared with those who had other anxiety disorders, patients with PTSD were three times more likely to have disordered eating (odds ratio [OR], 3.06; 95% confidence interval [CI], 1.47-6.37; P = .003).
In addition, higher baseline CIA scores predicted poorer PTSD treatment outcome (beta = –1.4; 95% CI, –1.67 to –1.10; P < .01).
“Having higher baseline CIA scores meant that patients’ PTSD symptoms did not remit as strongly compared to those with lower scores,” said Mr. Powers.
Patients with both PTSD and an eating disorder may have difficulty with regulating emotions and tolerating distress, he said.
“They may use binge eating, purging, or food restriction as strategies to regulate emotions. These behaviors may allow patients to become numb to or avoid heightened emotions that come from having PTSD and an eating disorder.”
Prior research linked perfectionism tendencies to poorer response to PTSD treatment. Those with an eating disorder may share similar tendencies, said Mr. Powers.
“If someone is consistently thinking negatively about their eating or body to the point where it interrupts their functioning, they may not be as likely to fully engage with PTSD treatment,” he said.
Ideally, clinicians would screen all patients with PTSD for an eating disorder, said Mr. Powers. “If screening instruments aren’t feasible or available, even just inquiring about body image or history of maladaptive eating behaviors can be helpful.”
He added this could open up a conversation about a traumatic event in the patient’s past.
Confirmatory research
Commenting on the study, Karen S. Mitchell, PhD, clinical research psychologist, National Center for PTSD, VA Boston Healthcare System, and associate professor in psychiatry, Boston University, said she was “excited” to see this research.
“Very few studies have examined the impact of baseline eating disorder symptoms on PTSD treatment outcomes or vice versa,” she said.
The study findings “add to the small but growing body of evidence suggesting that comorbid PTSD and eating disorder symptoms can impact recovery from each disorder,” she said.
She noted the importance of assessing comorbidity in patients presenting for treatment and of addressing comorbidity in both eating disorders and PTSD treatment. “But we need more research on how best to do this.”
Mr. Powers and Dr. Mitchell have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ADAA 2023




















