User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
2022 Update on gynecologic cancer
Despite the challenges of an ongoing COVID-19 pandemic, researchers in 2021 delivered practice-changing studies in gynecologic oncology. In this cancer Update, we highlight 4 studies that shed light on the surgical and systemic therapies that may improve outcomes for patients with cancers of the ovary, endometrium, and cervix. We review DESKTOP III, a trial that investigated the role of cytoreductive surgery in patients with recurrent ovarian cancer, and SENTOR, a study that evaluated the performance of sentinel lymph node biopsy in patients with high-grade endometrial cancers. Additionally, we examine 2 studies of systemic therapy that reveal the growing role of targeted therapies and immuno-oncology in the treatment of gynecologic malignancies.
A new era for patients with BRCA mutation–associated ovarian cancer
Banerjee S, Moore KN, Colombo N, et al. Maintenance olaparib for patients with newly diagnosed advanced ovarian cancer and a BRCA mutation (SOLO1/GOG 3004): 5-year follow-up of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2021;22:1721-1731.
Ovarian cancer remains the most lethal gynecologic malignancy due to the frequency of advanced-stage diagnosis and frequent relapse after primary therapy. But for ovarian cancer patients with inherited mutations of the BRCA1 or BRCA2 genes, poly(ADP-ribose) polymerase (PARP) inhibitors, a class of oral anticancer medicines that target DNA repair, have ushered in a new era in which the possibility of long-term remission, and even cure, is more likely than at any other time.
Olaparib trial details
The SOLO1 study was a double-blind, placebo-controlled, phase 3 trial that investigated the role of PARP inhibitor maintenance therapy with olaparib in patients with pathologic BRCA1 or BRCA2 mutations who responded to platinum-based chemotherapy administered for a newly diagnosed, advanced-stage ovarian cancer.1 The study enrolled 391 patients, with 260 randomly assigned to receive olaparib for 24 months and 131 patients randomly assigned to receive placebo tablets. Most patients in the study had a mutation in the BRCA1 gene (72%), 27% had a BRCA2 mutation, and 1% had mutations in both genes.
The primary analysis of SOLO1 was published in 2018 and was based on a median follow-up of 3.4 years.1 That study showed that olaparib maintenance therapy resulted in a large progression-free survival benefit and led to its approval by the US Food and Drug Administration (FDA) as a maintenance therapy for patients with BRCA-mutated advanced ovarian cancer who responded to first-line platinum-based chemotherapy.
In 2021, Banerjee and colleagues updated the progression-free survival results for the SOLO1 trial after 5 years of follow-up.2 In this study, the patients randomly assigned to olaparib maintenance therapy had a persistent and statistically significant progression-free survival benefit, with the median progression-free survival reaching 56 months among the olaparib group compared with 13.8 months in the placebo group (hazard ratio [HR], 0.33; 95% confidence interval [CI], 0.25–0.43).2 Olaparib maintenance therapy resulted in more clinically significant adverse events, including anemia and neutropenia. Serious adverse events occurred in 55 (21%) of the olaparib-treated patients and 17 (13%) of the placebo-treated patients, but no treatment-related adverse events were fatal.
The updated progression-free survival data from the SOLO1 study provides important and promising evidence that frontline PARP inhibitor maintenance therapy may affect long-term remission in an unprecedented proportion of patients with BRCA-related ovarian cancer. Significant, sustained benefit was seen well beyond the end of treatment, and median progression-free survival was an astonishing 3.5 years longer in the olaparib treatment group than among patients who received placebo therapy.
Continue to: Cytoreductive surgery for recurrent ovarian cancer improves survival in well-selected patients...
Cytoreductive surgery for recurrent ovarian cancer improves survival in well-selected patients
Harter P, Sehouli J, Vergote I, et al; DESKTOP III Investigators. Randomized trial of cytoreductive surgery for relapsed ovarian cancer. N Engl J Med. 2021;385:2123- 2131.
In the DESKTOP III trial, Harter and colleagues contribute results to the ongoing discourse surrounding treatment options for patients with recurrent, platinum-sensitive ovarian cancer.3 Systemic therapies continue to be the mainstay of treatment in this setting; however, several groups have attempted to evaluate the role of secondary cytoreductive surgery in this setting.4,5
Specific inclusion criteria employed
The DESKTOP III investigators randomly assigned 407 patients with platinum-sensitive recurrent ovarian cancer to secondary cytoreductive surgery followed by platinum-based chemotherapy (n = 206) or platinum-based chemotherapy alone (n = 201).3 An essential aspect of the study’s design was the use of specific inclusion criteria known to identify patients with a high likelihood of complete resection at the time of secondary cytoreduction.6,7 Patients were eligible only if they had at least a 6-month remission following platinum-based chemotherapy, had a complete resection at their previous surgery, had no restriction on physical activity, and had ascites of no more than 500 mL.
Surgery group had superior overall and progression-free survival
After a median follow-up of approximately 70 months, patients randomly assigned to surgery had superior overall survival (53.7 months) compared with those assigned to chemotherapy alone (46.0 months; HR, 0.75; 95% CI, 0.59–0.96).3 Progression-free survival also was improved among patients who underwent surgery (median 18.4 vs 12.7 months; HR, 0.66; 95% CI, 0.54–0.82). Subgroup analyses did not identify any subset of patients who did not benefit from surgery. Whether a complete resection was achieved at secondary cytoreduction was highly prognostic: Patients who had a complete resection had a median overall survival of 61.9 months compared with 27.7 months in patients with residual disease. There were no deaths within 90 days of surgery.
The DESKTOP III trial provides compelling evidence that secondary cytoreductive surgery improves overall and progression-free survival among well-selected patients with recurrent, platinum-sensitive ovarian cancer. These results differ from those of a recently reported Gynecologic Oncology Group (GOG) trial that failed to detect a survival benefit for secondary cytoreductive surgery among patients with platinum-sensitive recurrent ovarian cancer.5 Key differences, which might explain the studies’ seemingly contradictory results, were that the GOG study had fewer specific eligibility criteria than the DESKTOP III trial, and that bevacizumab was administered much more frequently in the GOG study. It is therefore reasonable to discuss the possible benefits of secondary cytoreductive surgery with patients who meet DESKTOP III eligibility criteria, with a focus toward shared decision making and a candid discussion of the potential risks and benefits of secondary cytoreduction.
Continue to: Immunotherapy enters first-line treatment regimen for advanced cervical cancer...
Immunotherapy enters first-line treatment regimen for advanced cervical cancer
Colombo N, Dubot C, Lorusso D, et al; KEYNOTE-826 Investigators. Pembrolizumab for persistent, recurrent, or metastatic cervical cancer. N Engl J Med. 2021;385:1856-1867.
Persistent, recurrent, and metastatic cervical cancer carries a very poor prognosis: Most patients progress less than a year after starting treatment, and fewer than half survive for 2 years. First-line treatment in this setting has been platinum-based chemotherapy, often given with bevacizumab, a humanized monoclonal antibody that inhibits tumor growth by blocking angiogenesis.8 Pembrolizumab, an immune checkpoint inhibitor, targets cancer cells by blocking their ability to evade the immune system, and it is FDA approved and widely administered to patients with advanced cervical cancer who progress after first-line treatment.9
Addition of pembrolizumab extended survival
In the KEYNOTE-826 trial, Colombo and colleagues investigated the efficacy of incorporating an immune checkpoint inhibitor into the first-line treatment regimen for patients with persistent, recurrent, and metastatic cervical cancer.10 Researchers in this double-blinded, phase 3, randomized controlled trial assigned 617 patients to receive pembrolizumab or placebo concurrently with the investigator’s choice platinum-based chemotherapy. Bevacizumab was administered at the discretion of the treating oncologist.
The proportion of patients who survived at least 2 years following randomization was significantly higher among those assigned to pembrolizumab compared with placebo (53% vs 42%; HR, 0.67, 95% CI, 0.54–0.84).10 Similarly, median progression-free survival was superior among patients who received pembrolizumab compared with those who received placebo (10.4 months vs 8.2 months; HR, 0.65; 95% CI, 0.53–0.79). The role of bevacizumab in conjunction with pembrolizumab and platinum-based chemotherapy was not elucidated in this study because bevacizumab administration was not randomly assigned.
Anemia and neutropenia were the most common adverse events and were more frequent in the pembrolizumab group, but there were no new safety concerns related to concurrent use of pembrolizumab with cytotoxic chemotherapy and bevacizumab. Importantly, subgroup analysis results suggested that pembrolizumab was effective only in patients whose tumors expressed PD-L1 (programmed death ligand 1), a biomarker of pembrolizumab sensitivity in cervical cancer.
In light of the significant improvements in overall and progression-free survival demonstrated in the KEYNOTE-826 trial, in October 2021, the FDA approved the use of frontline pembrolizumab alongside platinum-based chemotherapy, with or without bevacizumab, for treatment of patients with persistent, recurrent, or metastatic cervical cancers that express PD-L1.
Continue to: Endometrial cancer surgical staging...
Endometrial cancer surgical staging: Is sentinel lymph node biopsy a viable option for high-risk histologies?
Cusimano MC, Vicus D, Pulman K, et al. Assessment of sentinel lymph node biopsy vs lymphadenectomy for intermediate- and high-grade endometrial cancer staging. JAMA Surg. 2021;156:157-164.
The use of intraoperative sentinel lymph node mapping and biopsy to identify lymph node metastases among patients undergoing surgical staging for endometrial cancer has become increasingly common. Lymph node status is an important prognostic factor, and it guides adjuvant treatment decisions in endometrial cancer. However, traditional pelvic and para-aortic lymphadenectomy is associated with increased risk of lower-extremity lymphedema, postoperative complications, and intraoperative injury.
Sentinel lymph node biopsy seeks to identify lymph node metastases while minimizing surgical morbidity by identifying and excising only lymph nodes that directly receive lymphatic drainage from the uterus. The combination of a fluorescent dye (indocyanine green) and near infrared cameras have led to the broad adoption of sentinel lymph node biopsy in endometrial cancer staging surgery. This practice is supported by prospective studies that demonstrate the high diagnostic accuracy of this approach.11,12 However, because most patients included in prior studies had low-grade endometrial cancer, the utility of sentinel lymph node biopsy in cases of high-grade histology has been less clear.
Sentinel lymph node biopsy vs lymphadenectomy for staging
In the SENTOR trial, Cusimano and colleagues examined the diagnostic accuracy of sentinel lymph node mapping and biopsy, using indocyanine green, in patients with intermediate- or high-grade early-stage endometrial cancer.13
All eligible patients (N = 156) underwent traditional or robot-assisted laparoscopic hysterectomy with sentinel lymph node biopsy. Subsequently, patients with grade 2 endometrioid carcinoma underwent bilateral pelvic lymphadenectomy, and those with high-grade histology (grade 3 endometrioid, serous, carcinosarcoma, clear cell, undifferentiated or dedifferentiated, and mixed high grade) underwent bilateral pelvic and para-aortic lymphadenectomy. The investigators evaluated the diagnostic characteristics of sentinel lymph node biopsy, treating complete lymphadenectomy as the gold standard.
Of the 156 patients enrolled, the median age was 65.5 and median body mass index was 27.5; 126 patients (81%) had high-grade histology. The sentinel lymph node biopsy had a sensitivity of 96% (95% CI, 81%–100%), identifying 26 of the 27 patients with nodal metastases. The false-negative rate was 4% (95% CI, 0%–9%) and the negative predictive value was 99% (95% CI, 96%–100%). Intraoperative adverse events occurred in 5 patients (3%), but none occurred during the sentinel lymph node biopsy. ●
The high sensitivity and negative predictive value of sentinel lymph node biopsy in the intermediate- and high-grade cohort included in the SENTOR trial are concordant with prior studies that predominantly included patients with low-grade endometrial cancer. These findings suggest that sentinel lymph node mapping and biopsy is a reasonable option for surgical staging, not only for patients with low-grade endometrial cancer but also for those with intermediate- and high-grade disease.
- Moore K, Colombo N, Scambia G, et al. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 2018;379:2495-2505.
- Banerjee S, Moore KN, Colombo N, et al. Maintenance olaparib for patients with newly diagnosed advanced ovarian cancer and a BRCA mutation (SOLO1/GOG 3004): 5-year follow-up of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2021;22:1721-1731.
- Harter P, Sehouli J, Vergote I, et al; DESKTOP III Investigators. Randomized trial of cytoreductive surgery for relapsed ovarian cancer. N Engl J Med. 2021;385:2123-2131.
- Shi T, Zhu J, Feng Y, et al. Secondary cytoreduction followed by chemotherapy versus chemotherapy alone in platinum-sensitive relapsed ovarian cancer (SOC-1): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2021;22:439-449.
- Coleman RL, Spiritos NM, Enserro D, et al. Secondary surgical cytoreduction for recurrent ovarian cancer. N Engl J Med. 2019;381:1929-1939.
- Harter P, du Bois A, Hahmann M, et al; Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Committee; AGO Ovarian Cancer Study Group. Surgery in recurrent ovarian cancer: the Arbeitsgemeinschaft Gynaekologische Onkologie (AGO) DESKTOP OVAR trial. Ann Surg Oncol. 2006;13:1702-1710.
- Harter P, Sehouli J, Reuss A, et al. Prospective validation study of a predictive score for operability of recurrent ovarian cancer: the Multicenter Intergroup Study DESKTOP II. A project of the AGO Kommission OVAR, AGO Study Group, NOGGO, AGO-Austria, and MITO. Int J Gynecol Cancer. 2011;21: 289-295.
- Tewari KS, Sill MW, Penson RT, et al. Bevacizumab for advanced cervical cancer: final overall survival and adverse event analysis of a randomised, controlled, open-label, phase 3 trial (Gynecologic Oncology Group 240). Lancet. 2017;390:1654-1663.
- Frenel JS, Le Tourneau C, O’Neil B, et al. Safety and efficacy of pembrolizumab in advanced, programmed death ligand 1-positive cervical cancer: results from the phase Ib KEYNOTE-028 trial. J Clin Oncol. 2017;35:4035-4041.
- Colombo N, Dubot C, Lorusso D, et al; KEYNOTE-826 Investigators. Pembrolizumab for persistent, recurrent, or metastatic cervical cancer. N Engl J Med. 2021;385:1856-1867.
- Rossi EC, Kowalski L, Scalici J, et al. A comparison of sentinel lymph node biopsy to lymphadenectomy for endometrial cancer staging (FIRES trial): a multicentre, prospective, cohort study. Lancet Oncol. 2017;18:384-392.
- Ballester M, Dubernard G, Lecuru F, et al. Detection rate and diagnostic accuracy of sentinel-node biopsy in early stage endometrial cancer: a prospective multicentre study (SENTIENDO). Lancet Oncol. 2011;12: 469-476.
- Cusimano MC, Vicus D, Pulman K, et al. Assessment of sentinel lymph node biopsy vs lymphadenectomy for intermediate- and high-grade endometrial cancer staging. JAMA Surg. 2021;156:157-164.
Despite the challenges of an ongoing COVID-19 pandemic, researchers in 2021 delivered practice-changing studies in gynecologic oncology. In this cancer Update, we highlight 4 studies that shed light on the surgical and systemic therapies that may improve outcomes for patients with cancers of the ovary, endometrium, and cervix. We review DESKTOP III, a trial that investigated the role of cytoreductive surgery in patients with recurrent ovarian cancer, and SENTOR, a study that evaluated the performance of sentinel lymph node biopsy in patients with high-grade endometrial cancers. Additionally, we examine 2 studies of systemic therapy that reveal the growing role of targeted therapies and immuno-oncology in the treatment of gynecologic malignancies.
A new era for patients with BRCA mutation–associated ovarian cancer
Banerjee S, Moore KN, Colombo N, et al. Maintenance olaparib for patients with newly diagnosed advanced ovarian cancer and a BRCA mutation (SOLO1/GOG 3004): 5-year follow-up of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2021;22:1721-1731.
Ovarian cancer remains the most lethal gynecologic malignancy due to the frequency of advanced-stage diagnosis and frequent relapse after primary therapy. But for ovarian cancer patients with inherited mutations of the BRCA1 or BRCA2 genes, poly(ADP-ribose) polymerase (PARP) inhibitors, a class of oral anticancer medicines that target DNA repair, have ushered in a new era in which the possibility of long-term remission, and even cure, is more likely than at any other time.
Olaparib trial details
The SOLO1 study was a double-blind, placebo-controlled, phase 3 trial that investigated the role of PARP inhibitor maintenance therapy with olaparib in patients with pathologic BRCA1 or BRCA2 mutations who responded to platinum-based chemotherapy administered for a newly diagnosed, advanced-stage ovarian cancer.1 The study enrolled 391 patients, with 260 randomly assigned to receive olaparib for 24 months and 131 patients randomly assigned to receive placebo tablets. Most patients in the study had a mutation in the BRCA1 gene (72%), 27% had a BRCA2 mutation, and 1% had mutations in both genes.
The primary analysis of SOLO1 was published in 2018 and was based on a median follow-up of 3.4 years.1 That study showed that olaparib maintenance therapy resulted in a large progression-free survival benefit and led to its approval by the US Food and Drug Administration (FDA) as a maintenance therapy for patients with BRCA-mutated advanced ovarian cancer who responded to first-line platinum-based chemotherapy.
In 2021, Banerjee and colleagues updated the progression-free survival results for the SOLO1 trial after 5 years of follow-up.2 In this study, the patients randomly assigned to olaparib maintenance therapy had a persistent and statistically significant progression-free survival benefit, with the median progression-free survival reaching 56 months among the olaparib group compared with 13.8 months in the placebo group (hazard ratio [HR], 0.33; 95% confidence interval [CI], 0.25–0.43).2 Olaparib maintenance therapy resulted in more clinically significant adverse events, including anemia and neutropenia. Serious adverse events occurred in 55 (21%) of the olaparib-treated patients and 17 (13%) of the placebo-treated patients, but no treatment-related adverse events were fatal.
The updated progression-free survival data from the SOLO1 study provides important and promising evidence that frontline PARP inhibitor maintenance therapy may affect long-term remission in an unprecedented proportion of patients with BRCA-related ovarian cancer. Significant, sustained benefit was seen well beyond the end of treatment, and median progression-free survival was an astonishing 3.5 years longer in the olaparib treatment group than among patients who received placebo therapy.
Continue to: Cytoreductive surgery for recurrent ovarian cancer improves survival in well-selected patients...
Cytoreductive surgery for recurrent ovarian cancer improves survival in well-selected patients
Harter P, Sehouli J, Vergote I, et al; DESKTOP III Investigators. Randomized trial of cytoreductive surgery for relapsed ovarian cancer. N Engl J Med. 2021;385:2123- 2131.
In the DESKTOP III trial, Harter and colleagues contribute results to the ongoing discourse surrounding treatment options for patients with recurrent, platinum-sensitive ovarian cancer.3 Systemic therapies continue to be the mainstay of treatment in this setting; however, several groups have attempted to evaluate the role of secondary cytoreductive surgery in this setting.4,5
Specific inclusion criteria employed
The DESKTOP III investigators randomly assigned 407 patients with platinum-sensitive recurrent ovarian cancer to secondary cytoreductive surgery followed by platinum-based chemotherapy (n = 206) or platinum-based chemotherapy alone (n = 201).3 An essential aspect of the study’s design was the use of specific inclusion criteria known to identify patients with a high likelihood of complete resection at the time of secondary cytoreduction.6,7 Patients were eligible only if they had at least a 6-month remission following platinum-based chemotherapy, had a complete resection at their previous surgery, had no restriction on physical activity, and had ascites of no more than 500 mL.
Surgery group had superior overall and progression-free survival
After a median follow-up of approximately 70 months, patients randomly assigned to surgery had superior overall survival (53.7 months) compared with those assigned to chemotherapy alone (46.0 months; HR, 0.75; 95% CI, 0.59–0.96).3 Progression-free survival also was improved among patients who underwent surgery (median 18.4 vs 12.7 months; HR, 0.66; 95% CI, 0.54–0.82). Subgroup analyses did not identify any subset of patients who did not benefit from surgery. Whether a complete resection was achieved at secondary cytoreduction was highly prognostic: Patients who had a complete resection had a median overall survival of 61.9 months compared with 27.7 months in patients with residual disease. There were no deaths within 90 days of surgery.
The DESKTOP III trial provides compelling evidence that secondary cytoreductive surgery improves overall and progression-free survival among well-selected patients with recurrent, platinum-sensitive ovarian cancer. These results differ from those of a recently reported Gynecologic Oncology Group (GOG) trial that failed to detect a survival benefit for secondary cytoreductive surgery among patients with platinum-sensitive recurrent ovarian cancer.5 Key differences, which might explain the studies’ seemingly contradictory results, were that the GOG study had fewer specific eligibility criteria than the DESKTOP III trial, and that bevacizumab was administered much more frequently in the GOG study. It is therefore reasonable to discuss the possible benefits of secondary cytoreductive surgery with patients who meet DESKTOP III eligibility criteria, with a focus toward shared decision making and a candid discussion of the potential risks and benefits of secondary cytoreduction.
Continue to: Immunotherapy enters first-line treatment regimen for advanced cervical cancer...
Immunotherapy enters first-line treatment regimen for advanced cervical cancer
Colombo N, Dubot C, Lorusso D, et al; KEYNOTE-826 Investigators. Pembrolizumab for persistent, recurrent, or metastatic cervical cancer. N Engl J Med. 2021;385:1856-1867.
Persistent, recurrent, and metastatic cervical cancer carries a very poor prognosis: Most patients progress less than a year after starting treatment, and fewer than half survive for 2 years. First-line treatment in this setting has been platinum-based chemotherapy, often given with bevacizumab, a humanized monoclonal antibody that inhibits tumor growth by blocking angiogenesis.8 Pembrolizumab, an immune checkpoint inhibitor, targets cancer cells by blocking their ability to evade the immune system, and it is FDA approved and widely administered to patients with advanced cervical cancer who progress after first-line treatment.9
Addition of pembrolizumab extended survival
In the KEYNOTE-826 trial, Colombo and colleagues investigated the efficacy of incorporating an immune checkpoint inhibitor into the first-line treatment regimen for patients with persistent, recurrent, and metastatic cervical cancer.10 Researchers in this double-blinded, phase 3, randomized controlled trial assigned 617 patients to receive pembrolizumab or placebo concurrently with the investigator’s choice platinum-based chemotherapy. Bevacizumab was administered at the discretion of the treating oncologist.
The proportion of patients who survived at least 2 years following randomization was significantly higher among those assigned to pembrolizumab compared with placebo (53% vs 42%; HR, 0.67, 95% CI, 0.54–0.84).10 Similarly, median progression-free survival was superior among patients who received pembrolizumab compared with those who received placebo (10.4 months vs 8.2 months; HR, 0.65; 95% CI, 0.53–0.79). The role of bevacizumab in conjunction with pembrolizumab and platinum-based chemotherapy was not elucidated in this study because bevacizumab administration was not randomly assigned.
Anemia and neutropenia were the most common adverse events and were more frequent in the pembrolizumab group, but there were no new safety concerns related to concurrent use of pembrolizumab with cytotoxic chemotherapy and bevacizumab. Importantly, subgroup analysis results suggested that pembrolizumab was effective only in patients whose tumors expressed PD-L1 (programmed death ligand 1), a biomarker of pembrolizumab sensitivity in cervical cancer.
In light of the significant improvements in overall and progression-free survival demonstrated in the KEYNOTE-826 trial, in October 2021, the FDA approved the use of frontline pembrolizumab alongside platinum-based chemotherapy, with or without bevacizumab, for treatment of patients with persistent, recurrent, or metastatic cervical cancers that express PD-L1.
Continue to: Endometrial cancer surgical staging...
Endometrial cancer surgical staging: Is sentinel lymph node biopsy a viable option for high-risk histologies?
Cusimano MC, Vicus D, Pulman K, et al. Assessment of sentinel lymph node biopsy vs lymphadenectomy for intermediate- and high-grade endometrial cancer staging. JAMA Surg. 2021;156:157-164.
The use of intraoperative sentinel lymph node mapping and biopsy to identify lymph node metastases among patients undergoing surgical staging for endometrial cancer has become increasingly common. Lymph node status is an important prognostic factor, and it guides adjuvant treatment decisions in endometrial cancer. However, traditional pelvic and para-aortic lymphadenectomy is associated with increased risk of lower-extremity lymphedema, postoperative complications, and intraoperative injury.
Sentinel lymph node biopsy seeks to identify lymph node metastases while minimizing surgical morbidity by identifying and excising only lymph nodes that directly receive lymphatic drainage from the uterus. The combination of a fluorescent dye (indocyanine green) and near infrared cameras have led to the broad adoption of sentinel lymph node biopsy in endometrial cancer staging surgery. This practice is supported by prospective studies that demonstrate the high diagnostic accuracy of this approach.11,12 However, because most patients included in prior studies had low-grade endometrial cancer, the utility of sentinel lymph node biopsy in cases of high-grade histology has been less clear.
Sentinel lymph node biopsy vs lymphadenectomy for staging
In the SENTOR trial, Cusimano and colleagues examined the diagnostic accuracy of sentinel lymph node mapping and biopsy, using indocyanine green, in patients with intermediate- or high-grade early-stage endometrial cancer.13
All eligible patients (N = 156) underwent traditional or robot-assisted laparoscopic hysterectomy with sentinel lymph node biopsy. Subsequently, patients with grade 2 endometrioid carcinoma underwent bilateral pelvic lymphadenectomy, and those with high-grade histology (grade 3 endometrioid, serous, carcinosarcoma, clear cell, undifferentiated or dedifferentiated, and mixed high grade) underwent bilateral pelvic and para-aortic lymphadenectomy. The investigators evaluated the diagnostic characteristics of sentinel lymph node biopsy, treating complete lymphadenectomy as the gold standard.
Of the 156 patients enrolled, the median age was 65.5 and median body mass index was 27.5; 126 patients (81%) had high-grade histology. The sentinel lymph node biopsy had a sensitivity of 96% (95% CI, 81%–100%), identifying 26 of the 27 patients with nodal metastases. The false-negative rate was 4% (95% CI, 0%–9%) and the negative predictive value was 99% (95% CI, 96%–100%). Intraoperative adverse events occurred in 5 patients (3%), but none occurred during the sentinel lymph node biopsy. ●
The high sensitivity and negative predictive value of sentinel lymph node biopsy in the intermediate- and high-grade cohort included in the SENTOR trial are concordant with prior studies that predominantly included patients with low-grade endometrial cancer. These findings suggest that sentinel lymph node mapping and biopsy is a reasonable option for surgical staging, not only for patients with low-grade endometrial cancer but also for those with intermediate- and high-grade disease.
Despite the challenges of an ongoing COVID-19 pandemic, researchers in 2021 delivered practice-changing studies in gynecologic oncology. In this cancer Update, we highlight 4 studies that shed light on the surgical and systemic therapies that may improve outcomes for patients with cancers of the ovary, endometrium, and cervix. We review DESKTOP III, a trial that investigated the role of cytoreductive surgery in patients with recurrent ovarian cancer, and SENTOR, a study that evaluated the performance of sentinel lymph node biopsy in patients with high-grade endometrial cancers. Additionally, we examine 2 studies of systemic therapy that reveal the growing role of targeted therapies and immuno-oncology in the treatment of gynecologic malignancies.
A new era for patients with BRCA mutation–associated ovarian cancer
Banerjee S, Moore KN, Colombo N, et al. Maintenance olaparib for patients with newly diagnosed advanced ovarian cancer and a BRCA mutation (SOLO1/GOG 3004): 5-year follow-up of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2021;22:1721-1731.
Ovarian cancer remains the most lethal gynecologic malignancy due to the frequency of advanced-stage diagnosis and frequent relapse after primary therapy. But for ovarian cancer patients with inherited mutations of the BRCA1 or BRCA2 genes, poly(ADP-ribose) polymerase (PARP) inhibitors, a class of oral anticancer medicines that target DNA repair, have ushered in a new era in which the possibility of long-term remission, and even cure, is more likely than at any other time.
Olaparib trial details
The SOLO1 study was a double-blind, placebo-controlled, phase 3 trial that investigated the role of PARP inhibitor maintenance therapy with olaparib in patients with pathologic BRCA1 or BRCA2 mutations who responded to platinum-based chemotherapy administered for a newly diagnosed, advanced-stage ovarian cancer.1 The study enrolled 391 patients, with 260 randomly assigned to receive olaparib for 24 months and 131 patients randomly assigned to receive placebo tablets. Most patients in the study had a mutation in the BRCA1 gene (72%), 27% had a BRCA2 mutation, and 1% had mutations in both genes.
The primary analysis of SOLO1 was published in 2018 and was based on a median follow-up of 3.4 years.1 That study showed that olaparib maintenance therapy resulted in a large progression-free survival benefit and led to its approval by the US Food and Drug Administration (FDA) as a maintenance therapy for patients with BRCA-mutated advanced ovarian cancer who responded to first-line platinum-based chemotherapy.
In 2021, Banerjee and colleagues updated the progression-free survival results for the SOLO1 trial after 5 years of follow-up.2 In this study, the patients randomly assigned to olaparib maintenance therapy had a persistent and statistically significant progression-free survival benefit, with the median progression-free survival reaching 56 months among the olaparib group compared with 13.8 months in the placebo group (hazard ratio [HR], 0.33; 95% confidence interval [CI], 0.25–0.43).2 Olaparib maintenance therapy resulted in more clinically significant adverse events, including anemia and neutropenia. Serious adverse events occurred in 55 (21%) of the olaparib-treated patients and 17 (13%) of the placebo-treated patients, but no treatment-related adverse events were fatal.
The updated progression-free survival data from the SOLO1 study provides important and promising evidence that frontline PARP inhibitor maintenance therapy may affect long-term remission in an unprecedented proportion of patients with BRCA-related ovarian cancer. Significant, sustained benefit was seen well beyond the end of treatment, and median progression-free survival was an astonishing 3.5 years longer in the olaparib treatment group than among patients who received placebo therapy.
Continue to: Cytoreductive surgery for recurrent ovarian cancer improves survival in well-selected patients...
Cytoreductive surgery for recurrent ovarian cancer improves survival in well-selected patients
Harter P, Sehouli J, Vergote I, et al; DESKTOP III Investigators. Randomized trial of cytoreductive surgery for relapsed ovarian cancer. N Engl J Med. 2021;385:2123- 2131.
In the DESKTOP III trial, Harter and colleagues contribute results to the ongoing discourse surrounding treatment options for patients with recurrent, platinum-sensitive ovarian cancer.3 Systemic therapies continue to be the mainstay of treatment in this setting; however, several groups have attempted to evaluate the role of secondary cytoreductive surgery in this setting.4,5
Specific inclusion criteria employed
The DESKTOP III investigators randomly assigned 407 patients with platinum-sensitive recurrent ovarian cancer to secondary cytoreductive surgery followed by platinum-based chemotherapy (n = 206) or platinum-based chemotherapy alone (n = 201).3 An essential aspect of the study’s design was the use of specific inclusion criteria known to identify patients with a high likelihood of complete resection at the time of secondary cytoreduction.6,7 Patients were eligible only if they had at least a 6-month remission following platinum-based chemotherapy, had a complete resection at their previous surgery, had no restriction on physical activity, and had ascites of no more than 500 mL.
Surgery group had superior overall and progression-free survival
After a median follow-up of approximately 70 months, patients randomly assigned to surgery had superior overall survival (53.7 months) compared with those assigned to chemotherapy alone (46.0 months; HR, 0.75; 95% CI, 0.59–0.96).3 Progression-free survival also was improved among patients who underwent surgery (median 18.4 vs 12.7 months; HR, 0.66; 95% CI, 0.54–0.82). Subgroup analyses did not identify any subset of patients who did not benefit from surgery. Whether a complete resection was achieved at secondary cytoreduction was highly prognostic: Patients who had a complete resection had a median overall survival of 61.9 months compared with 27.7 months in patients with residual disease. There were no deaths within 90 days of surgery.
The DESKTOP III trial provides compelling evidence that secondary cytoreductive surgery improves overall and progression-free survival among well-selected patients with recurrent, platinum-sensitive ovarian cancer. These results differ from those of a recently reported Gynecologic Oncology Group (GOG) trial that failed to detect a survival benefit for secondary cytoreductive surgery among patients with platinum-sensitive recurrent ovarian cancer.5 Key differences, which might explain the studies’ seemingly contradictory results, were that the GOG study had fewer specific eligibility criteria than the DESKTOP III trial, and that bevacizumab was administered much more frequently in the GOG study. It is therefore reasonable to discuss the possible benefits of secondary cytoreductive surgery with patients who meet DESKTOP III eligibility criteria, with a focus toward shared decision making and a candid discussion of the potential risks and benefits of secondary cytoreduction.
Continue to: Immunotherapy enters first-line treatment regimen for advanced cervical cancer...
Immunotherapy enters first-line treatment regimen for advanced cervical cancer
Colombo N, Dubot C, Lorusso D, et al; KEYNOTE-826 Investigators. Pembrolizumab for persistent, recurrent, or metastatic cervical cancer. N Engl J Med. 2021;385:1856-1867.
Persistent, recurrent, and metastatic cervical cancer carries a very poor prognosis: Most patients progress less than a year after starting treatment, and fewer than half survive for 2 years. First-line treatment in this setting has been platinum-based chemotherapy, often given with bevacizumab, a humanized monoclonal antibody that inhibits tumor growth by blocking angiogenesis.8 Pembrolizumab, an immune checkpoint inhibitor, targets cancer cells by blocking their ability to evade the immune system, and it is FDA approved and widely administered to patients with advanced cervical cancer who progress after first-line treatment.9
Addition of pembrolizumab extended survival
In the KEYNOTE-826 trial, Colombo and colleagues investigated the efficacy of incorporating an immune checkpoint inhibitor into the first-line treatment regimen for patients with persistent, recurrent, and metastatic cervical cancer.10 Researchers in this double-blinded, phase 3, randomized controlled trial assigned 617 patients to receive pembrolizumab or placebo concurrently with the investigator’s choice platinum-based chemotherapy. Bevacizumab was administered at the discretion of the treating oncologist.
The proportion of patients who survived at least 2 years following randomization was significantly higher among those assigned to pembrolizumab compared with placebo (53% vs 42%; HR, 0.67, 95% CI, 0.54–0.84).10 Similarly, median progression-free survival was superior among patients who received pembrolizumab compared with those who received placebo (10.4 months vs 8.2 months; HR, 0.65; 95% CI, 0.53–0.79). The role of bevacizumab in conjunction with pembrolizumab and platinum-based chemotherapy was not elucidated in this study because bevacizumab administration was not randomly assigned.
Anemia and neutropenia were the most common adverse events and were more frequent in the pembrolizumab group, but there were no new safety concerns related to concurrent use of pembrolizumab with cytotoxic chemotherapy and bevacizumab. Importantly, subgroup analysis results suggested that pembrolizumab was effective only in patients whose tumors expressed PD-L1 (programmed death ligand 1), a biomarker of pembrolizumab sensitivity in cervical cancer.
In light of the significant improvements in overall and progression-free survival demonstrated in the KEYNOTE-826 trial, in October 2021, the FDA approved the use of frontline pembrolizumab alongside platinum-based chemotherapy, with or without bevacizumab, for treatment of patients with persistent, recurrent, or metastatic cervical cancers that express PD-L1.
Continue to: Endometrial cancer surgical staging...
Endometrial cancer surgical staging: Is sentinel lymph node biopsy a viable option for high-risk histologies?
Cusimano MC, Vicus D, Pulman K, et al. Assessment of sentinel lymph node biopsy vs lymphadenectomy for intermediate- and high-grade endometrial cancer staging. JAMA Surg. 2021;156:157-164.
The use of intraoperative sentinel lymph node mapping and biopsy to identify lymph node metastases among patients undergoing surgical staging for endometrial cancer has become increasingly common. Lymph node status is an important prognostic factor, and it guides adjuvant treatment decisions in endometrial cancer. However, traditional pelvic and para-aortic lymphadenectomy is associated with increased risk of lower-extremity lymphedema, postoperative complications, and intraoperative injury.
Sentinel lymph node biopsy seeks to identify lymph node metastases while minimizing surgical morbidity by identifying and excising only lymph nodes that directly receive lymphatic drainage from the uterus. The combination of a fluorescent dye (indocyanine green) and near infrared cameras have led to the broad adoption of sentinel lymph node biopsy in endometrial cancer staging surgery. This practice is supported by prospective studies that demonstrate the high diagnostic accuracy of this approach.11,12 However, because most patients included in prior studies had low-grade endometrial cancer, the utility of sentinel lymph node biopsy in cases of high-grade histology has been less clear.
Sentinel lymph node biopsy vs lymphadenectomy for staging
In the SENTOR trial, Cusimano and colleagues examined the diagnostic accuracy of sentinel lymph node mapping and biopsy, using indocyanine green, in patients with intermediate- or high-grade early-stage endometrial cancer.13
All eligible patients (N = 156) underwent traditional or robot-assisted laparoscopic hysterectomy with sentinel lymph node biopsy. Subsequently, patients with grade 2 endometrioid carcinoma underwent bilateral pelvic lymphadenectomy, and those with high-grade histology (grade 3 endometrioid, serous, carcinosarcoma, clear cell, undifferentiated or dedifferentiated, and mixed high grade) underwent bilateral pelvic and para-aortic lymphadenectomy. The investigators evaluated the diagnostic characteristics of sentinel lymph node biopsy, treating complete lymphadenectomy as the gold standard.
Of the 156 patients enrolled, the median age was 65.5 and median body mass index was 27.5; 126 patients (81%) had high-grade histology. The sentinel lymph node biopsy had a sensitivity of 96% (95% CI, 81%–100%), identifying 26 of the 27 patients with nodal metastases. The false-negative rate was 4% (95% CI, 0%–9%) and the negative predictive value was 99% (95% CI, 96%–100%). Intraoperative adverse events occurred in 5 patients (3%), but none occurred during the sentinel lymph node biopsy. ●
The high sensitivity and negative predictive value of sentinel lymph node biopsy in the intermediate- and high-grade cohort included in the SENTOR trial are concordant with prior studies that predominantly included patients with low-grade endometrial cancer. These findings suggest that sentinel lymph node mapping and biopsy is a reasonable option for surgical staging, not only for patients with low-grade endometrial cancer but also for those with intermediate- and high-grade disease.
- Moore K, Colombo N, Scambia G, et al. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 2018;379:2495-2505.
- Banerjee S, Moore KN, Colombo N, et al. Maintenance olaparib for patients with newly diagnosed advanced ovarian cancer and a BRCA mutation (SOLO1/GOG 3004): 5-year follow-up of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2021;22:1721-1731.
- Harter P, Sehouli J, Vergote I, et al; DESKTOP III Investigators. Randomized trial of cytoreductive surgery for relapsed ovarian cancer. N Engl J Med. 2021;385:2123-2131.
- Shi T, Zhu J, Feng Y, et al. Secondary cytoreduction followed by chemotherapy versus chemotherapy alone in platinum-sensitive relapsed ovarian cancer (SOC-1): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2021;22:439-449.
- Coleman RL, Spiritos NM, Enserro D, et al. Secondary surgical cytoreduction for recurrent ovarian cancer. N Engl J Med. 2019;381:1929-1939.
- Harter P, du Bois A, Hahmann M, et al; Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Committee; AGO Ovarian Cancer Study Group. Surgery in recurrent ovarian cancer: the Arbeitsgemeinschaft Gynaekologische Onkologie (AGO) DESKTOP OVAR trial. Ann Surg Oncol. 2006;13:1702-1710.
- Harter P, Sehouli J, Reuss A, et al. Prospective validation study of a predictive score for operability of recurrent ovarian cancer: the Multicenter Intergroup Study DESKTOP II. A project of the AGO Kommission OVAR, AGO Study Group, NOGGO, AGO-Austria, and MITO. Int J Gynecol Cancer. 2011;21: 289-295.
- Tewari KS, Sill MW, Penson RT, et al. Bevacizumab for advanced cervical cancer: final overall survival and adverse event analysis of a randomised, controlled, open-label, phase 3 trial (Gynecologic Oncology Group 240). Lancet. 2017;390:1654-1663.
- Frenel JS, Le Tourneau C, O’Neil B, et al. Safety and efficacy of pembrolizumab in advanced, programmed death ligand 1-positive cervical cancer: results from the phase Ib KEYNOTE-028 trial. J Clin Oncol. 2017;35:4035-4041.
- Colombo N, Dubot C, Lorusso D, et al; KEYNOTE-826 Investigators. Pembrolizumab for persistent, recurrent, or metastatic cervical cancer. N Engl J Med. 2021;385:1856-1867.
- Rossi EC, Kowalski L, Scalici J, et al. A comparison of sentinel lymph node biopsy to lymphadenectomy for endometrial cancer staging (FIRES trial): a multicentre, prospective, cohort study. Lancet Oncol. 2017;18:384-392.
- Ballester M, Dubernard G, Lecuru F, et al. Detection rate and diagnostic accuracy of sentinel-node biopsy in early stage endometrial cancer: a prospective multicentre study (SENTIENDO). Lancet Oncol. 2011;12: 469-476.
- Cusimano MC, Vicus D, Pulman K, et al. Assessment of sentinel lymph node biopsy vs lymphadenectomy for intermediate- and high-grade endometrial cancer staging. JAMA Surg. 2021;156:157-164.
- Moore K, Colombo N, Scambia G, et al. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 2018;379:2495-2505.
- Banerjee S, Moore KN, Colombo N, et al. Maintenance olaparib for patients with newly diagnosed advanced ovarian cancer and a BRCA mutation (SOLO1/GOG 3004): 5-year follow-up of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2021;22:1721-1731.
- Harter P, Sehouli J, Vergote I, et al; DESKTOP III Investigators. Randomized trial of cytoreductive surgery for relapsed ovarian cancer. N Engl J Med. 2021;385:2123-2131.
- Shi T, Zhu J, Feng Y, et al. Secondary cytoreduction followed by chemotherapy versus chemotherapy alone in platinum-sensitive relapsed ovarian cancer (SOC-1): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2021;22:439-449.
- Coleman RL, Spiritos NM, Enserro D, et al. Secondary surgical cytoreduction for recurrent ovarian cancer. N Engl J Med. 2019;381:1929-1939.
- Harter P, du Bois A, Hahmann M, et al; Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Committee; AGO Ovarian Cancer Study Group. Surgery in recurrent ovarian cancer: the Arbeitsgemeinschaft Gynaekologische Onkologie (AGO) DESKTOP OVAR trial. Ann Surg Oncol. 2006;13:1702-1710.
- Harter P, Sehouli J, Reuss A, et al. Prospective validation study of a predictive score for operability of recurrent ovarian cancer: the Multicenter Intergroup Study DESKTOP II. A project of the AGO Kommission OVAR, AGO Study Group, NOGGO, AGO-Austria, and MITO. Int J Gynecol Cancer. 2011;21: 289-295.
- Tewari KS, Sill MW, Penson RT, et al. Bevacizumab for advanced cervical cancer: final overall survival and adverse event analysis of a randomised, controlled, open-label, phase 3 trial (Gynecologic Oncology Group 240). Lancet. 2017;390:1654-1663.
- Frenel JS, Le Tourneau C, O’Neil B, et al. Safety and efficacy of pembrolizumab in advanced, programmed death ligand 1-positive cervical cancer: results from the phase Ib KEYNOTE-028 trial. J Clin Oncol. 2017;35:4035-4041.
- Colombo N, Dubot C, Lorusso D, et al; KEYNOTE-826 Investigators. Pembrolizumab for persistent, recurrent, or metastatic cervical cancer. N Engl J Med. 2021;385:1856-1867.
- Rossi EC, Kowalski L, Scalici J, et al. A comparison of sentinel lymph node biopsy to lymphadenectomy for endometrial cancer staging (FIRES trial): a multicentre, prospective, cohort study. Lancet Oncol. 2017;18:384-392.
- Ballester M, Dubernard G, Lecuru F, et al. Detection rate and diagnostic accuracy of sentinel-node biopsy in early stage endometrial cancer: a prospective multicentre study (SENTIENDO). Lancet Oncol. 2011;12: 469-476.
- Cusimano MC, Vicus D, Pulman K, et al. Assessment of sentinel lymph node biopsy vs lymphadenectomy for intermediate- and high-grade endometrial cancer staging. JAMA Surg. 2021;156:157-164.
Nonstress test and maximal vertical pocket vs the biophysical profile: Equivocal or equivalent?
CASE 1 Pregnant patient endures extensive wait and travel times to have antenatal testing
Pregnant at age 35 without comorbidities, Ms. H was instructed to schedule weekly biophysical profiles (BPP) after 36 weeks’ gestation for advanced maternal age. She receives care at a community office 25 miles from the hospital where she will deliver. Ms. H must complete her antenatal testing at the hospital where the sonographer performs BPPs. She sees her physician at the nearby clinic and then takes public transit to the hospital. She waits 2 hours to be seen then makes her way back home. Her prenatal care visit, which usually takes 30 minutes, turns into a 5-hour ordeal. Ms. H delivered a healthy baby at 39 weeks. Unfortunately, she was fired from her job for missing too many workdays.
Antenatal testing has become routine, and it is costly
For the prescriber, antenatal testing is simple: Order a weekly ultrasound exam to reduce the risk of stillbirth, decrease litigation, generate income, and maximize patient satisfaction (with the assumption that everyone likes to peek at their baby). Recommending antenatal testing has—with the best intentions—become a habit and therefore is difficult to break. However, the American College of Obstetricians and Gynecologists (ACOG) recognizes that “there is a paucity of evidenced-based recommendations on the timing and frequency of antenatal fetal surveillance because of the challenges of conducting prospective trials in pregnancies complicated by stillbirths and the varying conditions that place pregnancies at high risk for stillbirth. As a result, evidence for the efficacy of antenatal fetal surveillance, when available, is largely circumstantial.”1
Antenatal testing without an evidence-based indication can be costly for the health care system, insurance companies, and patients. Many clinics, especially those in rural communities, do not have the equipment or personnel to complete antenatal testing on site. Asking a pregnant patient to travel repeatedly to another location for antenatal testing can increase her time off from work, complicate childcare, pose a financial burden, and lead to nonadherence. As clinicians, it is imperative that we work with our patients to create an individualized care plan to minimize these burdens and increase adherence.
Antenatal fetal surveillance can be considered for conditions in which stillbirth is reported more frequently than 0.8 per 1,000.
Advanced maternal age and stillbirth risk
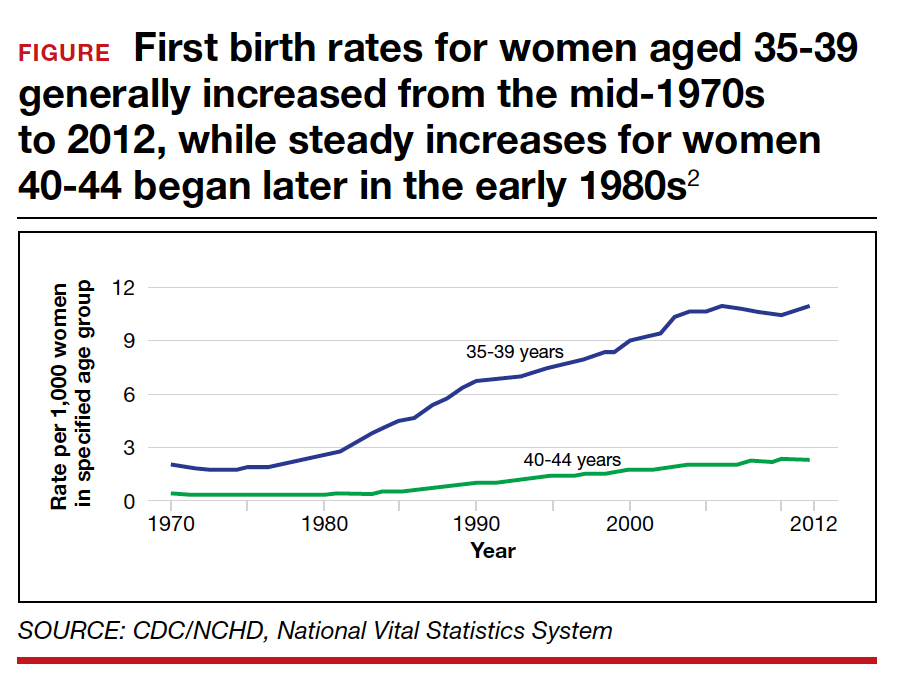
One of the most common reasons for antenatal testing is advanced maternal age, that is, age older than 35. According to the Centers for Disease Control and Prevention and the National Vital Statistics System, from 2000 to 2012, 46 states and the District of Columbia (DC) reported an increase in first birth rates for women aged 35 to 39. Thirty-one states and DC saw a rise among women aged 40 to 44 in the same period (FIGURE).2
Advanced maternal age is an independent risk factor for stillbirth, with women aged 35 to 39 at 1.9-fold increased risk and women older than age 40 with a 2.4-fold higher risk compared with women younger than age 30.3 In a review of 44 studies including nearly 45,000,000 births, case-control studies, versus cohort studies, demonstrated a higher odds for stillbirth among women aged 35 and older (odds ratio [OR], 2.39; 95% confidence interval [CI], 1.57-3.66 vs OR, 1.73; 95% CI, 1.6-1.87).4 Now, many women older than age 35 may have a concomitant risk factor, such as diabetes or hypertension, that requires antenatal testing. However, for those without other risk factors, nearly 863 antenatal tests and 71 inductions would need to be completed to reduce the number of stillbirths by 1. Antenatal testing for women older than age 35 without other risk factors should be individualized through shared decision making.5 See the ACOG committee opinion for a table that outlines factors associated with an increased risk of stillbirth and suggested strategies for antenatal surveillance after viability.1
Continue to: CASE 2 Patient with high BPP score and altered...
CASE 2 Patient with high BPP score and altered fetal movements delivered for nonreassuring fetal heart rate
Ms. Q was undergoing weekly BPPs for diet-controlled gestational diabetes and a prepregnancy body mass index (BMI) of 52. At 37 weeks’ gestation, she had a BPP score of 8/8. However, it took almost 30 minutes to see 2 discrete body or limb movements. Ms. Q mentioned to the nurse taking her vitals after the BPP that the baby’s movements had changed over the previous few days, especially after contractions. Ms. Q then completed a nonstress test (NST); she had 2 contractions and 2 fetal heart rate decelerations, each lasting approximately 60 seconds. Ms. Q was sent to labor and delivery for prolonged monitoring, and she was delivered that day for a nonreassuring fetal heart rate tracing. Meconium-stained amniotic fluid and a tight triple nuchal cord were noted at delivery.
BPP considerations
While considered an in-depth look at the fetal status, BPPs may not predict overall fetal well-being during acute changes, such as umbilical cord compression or placental abruption. BPPs take longer to complete, require a trained sonographer, and include components like fetal breathing that may be influenced by such factors as nicotine,6-8 labor,9 rupture of membranes,10 magnesium sulfate,11 and infection.12
If medically indicated, which antenatal surveillance technique is right for your patient?
Frequently used antepartum fetal surveillance techniques include maternal perception of fetal movement or “kick counting,” NST, BPP, modified BPP, contraction stress test (CST), and umbilical artery Doppler velocimetry.
Worldwide, the most common form of antenatal surveillance is fetal kick counting. It is noninvasive, can be completed frequently, may decrease maternal anxiety, may improve maternal-fetal bonding, and is free.13 According to the results of a 2020 meta-analysis of 468,601 fetuses, however, there was no difference in perinatal death among patients who assessed fetal movements (0.54%) and those who did not (0.59%).14 There was a statistically significant increase in induction of labor, cesarean delivery, and preterm delivery among patients who counted fetal movements. Women who perceive a decrease in fetal movement should seek medical attention from a health care provider.
An evaluation for decreased fetal movement typically includes taking a history that focuses on risk factors that may increase stillbirth, including hypertension, growth restriction, fetal anomalies, diabetes, and substance use, and auscultation with a fetal Doppler. In the absence of risk factors and the presence of a normal fetal heartbeat, pregnant women should be reassured of fetal well-being. In a pregnancy at greater than 28 weeks, a 20-minute NST can be completed as well; this has become part of the standard workup of decreased fetal movement in developed countries. A reactive NST indicates normal fetal autonomic function in real time and a low incidence of stillbirth (1.9/1,000) within 1 week.15
Additionally, by measuring the amniotic fluid volume using the largest maximal vertical pocket (MVP), clinicians can gain insight into overall uteroplacental function. The combination of the NST and the MVP—otherwise known as a modified BPP—provides both short-term acid-base status and long-term uteroplacental function. The incidence of stillbirth in the 1 week after a modified BPP has been reported to be 0.8/1,000, which is equivalent to stillbirth incidence using a full BPP (0.8/1,000).16 The negative predictive value for both the modified BPP and the BPP is 99.9%—equivalent.
The case for modified BPP use
The modified BPP requires less time, is less costly (cost savings of approximately 50%), does not require a specialized sonographer, and can be performed in local community clinics.
Perhaps the initial antepartum surveillance test of choice should be the modified BPP, with the BPP used in cases in which the results of a modified BPP are abnormal. ●
- American College of Obstetricians and Gynecologists’ Committee on Obstetric Practice, Society for MaternalFetal Medicine. Indications for outpatient antenatal fetal surveillance: ACOG committee opinion no. 828. Obstet Gynecol. 2021;137:e177-197.
- Mathews TJ, Hamilton BE. First births to older women continue to rise. NCHS Data Brief, No. 152. Hyattsville, MD: National Center for Health Statistics; 2014.
- Fretts RC, Schmittdiel J, McLean FH, et al. Increased maternal age and the risk of fetal death. N Engl J Med. 1995;333: 953-957.
- Lean SC, Derricott H, Jones RL, et al. Advanced maternal age and adverse pregnancy outcomes: a systematic review and meta-analysis. PLoS One. 2017;12:e0186287.
- Fretts RC, Elkins EB, Myers ER, et al. Should older women have antepartum testing to prevent unexplained stillbirth? Obstet Gynecol. 2004;104:56-64.
- Manning F, Wyn Pugh E, Boddy K. Effect of cigarette smoking on fetal breathing movements in normal pregnancies. Br Med J. 1975;1:552-553.
- Manning FA, Feyerabend C. Cigarette smoking and fetal breathing movements. Br J Obstet Gynecol. 1976;83:262-270.
- Gennser G, Marsal K, Brantmark B. Maternal smoking and fetal breathing movements. Am J Obstet Gynecol. 1975;123:861-867.
- Boylan P, O’Donovan P, Owens OJ. Fetal breathing movements and the diagnosis of labor: a prospective analysis of 100 cases. Obstet Gynecol. 1985;66:517-520.
- Kivikoski AI, Amon E, Vaalamo PO, et al. Effect of thirdtrimester premature rupture of membranes on fetal breathing movements: a prospective case-control study. Am J Obstet Gynecol. 1988;159:1474-1477.
- Peaceman AM, Meyer BA, Thorp JA, et al. The effect of magnesium sulfate tocolysis on the fetal biophysical profile. Am J Obstet Gynecol. 1989;161:771-774.
- Vintzileos AM, Campbell WA, Nochimson DJ, et al. The fetal biophysical profile in patients with premature rupture of the membranes—an early predictor of fetal infection. Am J Obstet Gynecol. 1985;152:501-516.
- Liston RM, Bloom K, Zimmer P. The psychological effects of counting fetal movements. Birth. 1994;21:135-140.
- Bellussi F, Po’ G, Livi A, et al. Fetal movement counting and perinatal mortality: a systematic review and meta-analysis. Obstet Gynecol. 2020;135:453-462.
- Freeman RK, Anderson G, Dorchester W. A prospective multiinstitutional study of antepartum fetal heart rate monitoring. I. Risk of perinatal mortality and morbidity according to antepartum fetal heart rate test results. Am J Obstet Gynecol. 1982;143:771-777.
- Miller DA , Rabello YA, Paul RH. The modified biophysical profile: antepartum testing in the 1990s. Am J Obstet Gynecol. 1996;174:812-817.
CASE 1 Pregnant patient endures extensive wait and travel times to have antenatal testing
Pregnant at age 35 without comorbidities, Ms. H was instructed to schedule weekly biophysical profiles (BPP) after 36 weeks’ gestation for advanced maternal age. She receives care at a community office 25 miles from the hospital where she will deliver. Ms. H must complete her antenatal testing at the hospital where the sonographer performs BPPs. She sees her physician at the nearby clinic and then takes public transit to the hospital. She waits 2 hours to be seen then makes her way back home. Her prenatal care visit, which usually takes 30 minutes, turns into a 5-hour ordeal. Ms. H delivered a healthy baby at 39 weeks. Unfortunately, she was fired from her job for missing too many workdays.
Antenatal testing has become routine, and it is costly
For the prescriber, antenatal testing is simple: Order a weekly ultrasound exam to reduce the risk of stillbirth, decrease litigation, generate income, and maximize patient satisfaction (with the assumption that everyone likes to peek at their baby). Recommending antenatal testing has—with the best intentions—become a habit and therefore is difficult to break. However, the American College of Obstetricians and Gynecologists (ACOG) recognizes that “there is a paucity of evidenced-based recommendations on the timing and frequency of antenatal fetal surveillance because of the challenges of conducting prospective trials in pregnancies complicated by stillbirths and the varying conditions that place pregnancies at high risk for stillbirth. As a result, evidence for the efficacy of antenatal fetal surveillance, when available, is largely circumstantial.”1
Antenatal testing without an evidence-based indication can be costly for the health care system, insurance companies, and patients. Many clinics, especially those in rural communities, do not have the equipment or personnel to complete antenatal testing on site. Asking a pregnant patient to travel repeatedly to another location for antenatal testing can increase her time off from work, complicate childcare, pose a financial burden, and lead to nonadherence. As clinicians, it is imperative that we work with our patients to create an individualized care plan to minimize these burdens and increase adherence.
Antenatal fetal surveillance can be considered for conditions in which stillbirth is reported more frequently than 0.8 per 1,000.
Advanced maternal age and stillbirth risk
One of the most common reasons for antenatal testing is advanced maternal age, that is, age older than 35. According to the Centers for Disease Control and Prevention and the National Vital Statistics System, from 2000 to 2012, 46 states and the District of Columbia (DC) reported an increase in first birth rates for women aged 35 to 39. Thirty-one states and DC saw a rise among women aged 40 to 44 in the same period (FIGURE).2

Advanced maternal age is an independent risk factor for stillbirth, with women aged 35 to 39 at 1.9-fold increased risk and women older than age 40 with a 2.4-fold higher risk compared with women younger than age 30.3 In a review of 44 studies including nearly 45,000,000 births, case-control studies, versus cohort studies, demonstrated a higher odds for stillbirth among women aged 35 and older (odds ratio [OR], 2.39; 95% confidence interval [CI], 1.57-3.66 vs OR, 1.73; 95% CI, 1.6-1.87).4 Now, many women older than age 35 may have a concomitant risk factor, such as diabetes or hypertension, that requires antenatal testing. However, for those without other risk factors, nearly 863 antenatal tests and 71 inductions would need to be completed to reduce the number of stillbirths by 1. Antenatal testing for women older than age 35 without other risk factors should be individualized through shared decision making.5 See the ACOG committee opinion for a table that outlines factors associated with an increased risk of stillbirth and suggested strategies for antenatal surveillance after viability.1
Continue to: CASE 2 Patient with high BPP score and altered...
CASE 2 Patient with high BPP score and altered fetal movements delivered for nonreassuring fetal heart rate
Ms. Q was undergoing weekly BPPs for diet-controlled gestational diabetes and a prepregnancy body mass index (BMI) of 52. At 37 weeks’ gestation, she had a BPP score of 8/8. However, it took almost 30 minutes to see 2 discrete body or limb movements. Ms. Q mentioned to the nurse taking her vitals after the BPP that the baby’s movements had changed over the previous few days, especially after contractions. Ms. Q then completed a nonstress test (NST); she had 2 contractions and 2 fetal heart rate decelerations, each lasting approximately 60 seconds. Ms. Q was sent to labor and delivery for prolonged monitoring, and she was delivered that day for a nonreassuring fetal heart rate tracing. Meconium-stained amniotic fluid and a tight triple nuchal cord were noted at delivery.
BPP considerations
While considered an in-depth look at the fetal status, BPPs may not predict overall fetal well-being during acute changes, such as umbilical cord compression or placental abruption. BPPs take longer to complete, require a trained sonographer, and include components like fetal breathing that may be influenced by such factors as nicotine,6-8 labor,9 rupture of membranes,10 magnesium sulfate,11 and infection.12
If medically indicated, which antenatal surveillance technique is right for your patient?
Frequently used antepartum fetal surveillance techniques include maternal perception of fetal movement or “kick counting,” NST, BPP, modified BPP, contraction stress test (CST), and umbilical artery Doppler velocimetry.
Worldwide, the most common form of antenatal surveillance is fetal kick counting. It is noninvasive, can be completed frequently, may decrease maternal anxiety, may improve maternal-fetal bonding, and is free.13 According to the results of a 2020 meta-analysis of 468,601 fetuses, however, there was no difference in perinatal death among patients who assessed fetal movements (0.54%) and those who did not (0.59%).14 There was a statistically significant increase in induction of labor, cesarean delivery, and preterm delivery among patients who counted fetal movements. Women who perceive a decrease in fetal movement should seek medical attention from a health care provider.
An evaluation for decreased fetal movement typically includes taking a history that focuses on risk factors that may increase stillbirth, including hypertension, growth restriction, fetal anomalies, diabetes, and substance use, and auscultation with a fetal Doppler. In the absence of risk factors and the presence of a normal fetal heartbeat, pregnant women should be reassured of fetal well-being. In a pregnancy at greater than 28 weeks, a 20-minute NST can be completed as well; this has become part of the standard workup of decreased fetal movement in developed countries. A reactive NST indicates normal fetal autonomic function in real time and a low incidence of stillbirth (1.9/1,000) within 1 week.15
Additionally, by measuring the amniotic fluid volume using the largest maximal vertical pocket (MVP), clinicians can gain insight into overall uteroplacental function. The combination of the NST and the MVP—otherwise known as a modified BPP—provides both short-term acid-base status and long-term uteroplacental function. The incidence of stillbirth in the 1 week after a modified BPP has been reported to be 0.8/1,000, which is equivalent to stillbirth incidence using a full BPP (0.8/1,000).16 The negative predictive value for both the modified BPP and the BPP is 99.9%—equivalent.
The case for modified BPP use
The modified BPP requires less time, is less costly (cost savings of approximately 50%), does not require a specialized sonographer, and can be performed in local community clinics.
Perhaps the initial antepartum surveillance test of choice should be the modified BPP, with the BPP used in cases in which the results of a modified BPP are abnormal. ●
CASE 1 Pregnant patient endures extensive wait and travel times to have antenatal testing
Pregnant at age 35 without comorbidities, Ms. H was instructed to schedule weekly biophysical profiles (BPP) after 36 weeks’ gestation for advanced maternal age. She receives care at a community office 25 miles from the hospital where she will deliver. Ms. H must complete her antenatal testing at the hospital where the sonographer performs BPPs. She sees her physician at the nearby clinic and then takes public transit to the hospital. She waits 2 hours to be seen then makes her way back home. Her prenatal care visit, which usually takes 30 minutes, turns into a 5-hour ordeal. Ms. H delivered a healthy baby at 39 weeks. Unfortunately, she was fired from her job for missing too many workdays.
Antenatal testing has become routine, and it is costly
For the prescriber, antenatal testing is simple: Order a weekly ultrasound exam to reduce the risk of stillbirth, decrease litigation, generate income, and maximize patient satisfaction (with the assumption that everyone likes to peek at their baby). Recommending antenatal testing has—with the best intentions—become a habit and therefore is difficult to break. However, the American College of Obstetricians and Gynecologists (ACOG) recognizes that “there is a paucity of evidenced-based recommendations on the timing and frequency of antenatal fetal surveillance because of the challenges of conducting prospective trials in pregnancies complicated by stillbirths and the varying conditions that place pregnancies at high risk for stillbirth. As a result, evidence for the efficacy of antenatal fetal surveillance, when available, is largely circumstantial.”1
Antenatal testing without an evidence-based indication can be costly for the health care system, insurance companies, and patients. Many clinics, especially those in rural communities, do not have the equipment or personnel to complete antenatal testing on site. Asking a pregnant patient to travel repeatedly to another location for antenatal testing can increase her time off from work, complicate childcare, pose a financial burden, and lead to nonadherence. As clinicians, it is imperative that we work with our patients to create an individualized care plan to minimize these burdens and increase adherence.
Antenatal fetal surveillance can be considered for conditions in which stillbirth is reported more frequently than 0.8 per 1,000.
Advanced maternal age and stillbirth risk
One of the most common reasons for antenatal testing is advanced maternal age, that is, age older than 35. According to the Centers for Disease Control and Prevention and the National Vital Statistics System, from 2000 to 2012, 46 states and the District of Columbia (DC) reported an increase in first birth rates for women aged 35 to 39. Thirty-one states and DC saw a rise among women aged 40 to 44 in the same period (FIGURE).2

Advanced maternal age is an independent risk factor for stillbirth, with women aged 35 to 39 at 1.9-fold increased risk and women older than age 40 with a 2.4-fold higher risk compared with women younger than age 30.3 In a review of 44 studies including nearly 45,000,000 births, case-control studies, versus cohort studies, demonstrated a higher odds for stillbirth among women aged 35 and older (odds ratio [OR], 2.39; 95% confidence interval [CI], 1.57-3.66 vs OR, 1.73; 95% CI, 1.6-1.87).4 Now, many women older than age 35 may have a concomitant risk factor, such as diabetes or hypertension, that requires antenatal testing. However, for those without other risk factors, nearly 863 antenatal tests and 71 inductions would need to be completed to reduce the number of stillbirths by 1. Antenatal testing for women older than age 35 without other risk factors should be individualized through shared decision making.5 See the ACOG committee opinion for a table that outlines factors associated with an increased risk of stillbirth and suggested strategies for antenatal surveillance after viability.1
Continue to: CASE 2 Patient with high BPP score and altered...
CASE 2 Patient with high BPP score and altered fetal movements delivered for nonreassuring fetal heart rate
Ms. Q was undergoing weekly BPPs for diet-controlled gestational diabetes and a prepregnancy body mass index (BMI) of 52. At 37 weeks’ gestation, she had a BPP score of 8/8. However, it took almost 30 minutes to see 2 discrete body or limb movements. Ms. Q mentioned to the nurse taking her vitals after the BPP that the baby’s movements had changed over the previous few days, especially after contractions. Ms. Q then completed a nonstress test (NST); she had 2 contractions and 2 fetal heart rate decelerations, each lasting approximately 60 seconds. Ms. Q was sent to labor and delivery for prolonged monitoring, and she was delivered that day for a nonreassuring fetal heart rate tracing. Meconium-stained amniotic fluid and a tight triple nuchal cord were noted at delivery.
BPP considerations
While considered an in-depth look at the fetal status, BPPs may not predict overall fetal well-being during acute changes, such as umbilical cord compression or placental abruption. BPPs take longer to complete, require a trained sonographer, and include components like fetal breathing that may be influenced by such factors as nicotine,6-8 labor,9 rupture of membranes,10 magnesium sulfate,11 and infection.12
If medically indicated, which antenatal surveillance technique is right for your patient?
Frequently used antepartum fetal surveillance techniques include maternal perception of fetal movement or “kick counting,” NST, BPP, modified BPP, contraction stress test (CST), and umbilical artery Doppler velocimetry.
Worldwide, the most common form of antenatal surveillance is fetal kick counting. It is noninvasive, can be completed frequently, may decrease maternal anxiety, may improve maternal-fetal bonding, and is free.13 According to the results of a 2020 meta-analysis of 468,601 fetuses, however, there was no difference in perinatal death among patients who assessed fetal movements (0.54%) and those who did not (0.59%).14 There was a statistically significant increase in induction of labor, cesarean delivery, and preterm delivery among patients who counted fetal movements. Women who perceive a decrease in fetal movement should seek medical attention from a health care provider.
An evaluation for decreased fetal movement typically includes taking a history that focuses on risk factors that may increase stillbirth, including hypertension, growth restriction, fetal anomalies, diabetes, and substance use, and auscultation with a fetal Doppler. In the absence of risk factors and the presence of a normal fetal heartbeat, pregnant women should be reassured of fetal well-being. In a pregnancy at greater than 28 weeks, a 20-minute NST can be completed as well; this has become part of the standard workup of decreased fetal movement in developed countries. A reactive NST indicates normal fetal autonomic function in real time and a low incidence of stillbirth (1.9/1,000) within 1 week.15
Additionally, by measuring the amniotic fluid volume using the largest maximal vertical pocket (MVP), clinicians can gain insight into overall uteroplacental function. The combination of the NST and the MVP—otherwise known as a modified BPP—provides both short-term acid-base status and long-term uteroplacental function. The incidence of stillbirth in the 1 week after a modified BPP has been reported to be 0.8/1,000, which is equivalent to stillbirth incidence using a full BPP (0.8/1,000).16 The negative predictive value for both the modified BPP and the BPP is 99.9%—equivalent.
The case for modified BPP use
The modified BPP requires less time, is less costly (cost savings of approximately 50%), does not require a specialized sonographer, and can be performed in local community clinics.
Perhaps the initial antepartum surveillance test of choice should be the modified BPP, with the BPP used in cases in which the results of a modified BPP are abnormal. ●
- American College of Obstetricians and Gynecologists’ Committee on Obstetric Practice, Society for MaternalFetal Medicine. Indications for outpatient antenatal fetal surveillance: ACOG committee opinion no. 828. Obstet Gynecol. 2021;137:e177-197.
- Mathews TJ, Hamilton BE. First births to older women continue to rise. NCHS Data Brief, No. 152. Hyattsville, MD: National Center for Health Statistics; 2014.
- Fretts RC, Schmittdiel J, McLean FH, et al. Increased maternal age and the risk of fetal death. N Engl J Med. 1995;333: 953-957.
- Lean SC, Derricott H, Jones RL, et al. Advanced maternal age and adverse pregnancy outcomes: a systematic review and meta-analysis. PLoS One. 2017;12:e0186287.
- Fretts RC, Elkins EB, Myers ER, et al. Should older women have antepartum testing to prevent unexplained stillbirth? Obstet Gynecol. 2004;104:56-64.
- Manning F, Wyn Pugh E, Boddy K. Effect of cigarette smoking on fetal breathing movements in normal pregnancies. Br Med J. 1975;1:552-553.
- Manning FA, Feyerabend C. Cigarette smoking and fetal breathing movements. Br J Obstet Gynecol. 1976;83:262-270.
- Gennser G, Marsal K, Brantmark B. Maternal smoking and fetal breathing movements. Am J Obstet Gynecol. 1975;123:861-867.
- Boylan P, O’Donovan P, Owens OJ. Fetal breathing movements and the diagnosis of labor: a prospective analysis of 100 cases. Obstet Gynecol. 1985;66:517-520.
- Kivikoski AI, Amon E, Vaalamo PO, et al. Effect of thirdtrimester premature rupture of membranes on fetal breathing movements: a prospective case-control study. Am J Obstet Gynecol. 1988;159:1474-1477.
- Peaceman AM, Meyer BA, Thorp JA, et al. The effect of magnesium sulfate tocolysis on the fetal biophysical profile. Am J Obstet Gynecol. 1989;161:771-774.
- Vintzileos AM, Campbell WA, Nochimson DJ, et al. The fetal biophysical profile in patients with premature rupture of the membranes—an early predictor of fetal infection. Am J Obstet Gynecol. 1985;152:501-516.
- Liston RM, Bloom K, Zimmer P. The psychological effects of counting fetal movements. Birth. 1994;21:135-140.
- Bellussi F, Po’ G, Livi A, et al. Fetal movement counting and perinatal mortality: a systematic review and meta-analysis. Obstet Gynecol. 2020;135:453-462.
- Freeman RK, Anderson G, Dorchester W. A prospective multiinstitutional study of antepartum fetal heart rate monitoring. I. Risk of perinatal mortality and morbidity according to antepartum fetal heart rate test results. Am J Obstet Gynecol. 1982;143:771-777.
- Miller DA , Rabello YA, Paul RH. The modified biophysical profile: antepartum testing in the 1990s. Am J Obstet Gynecol. 1996;174:812-817.
- American College of Obstetricians and Gynecologists’ Committee on Obstetric Practice, Society for MaternalFetal Medicine. Indications for outpatient antenatal fetal surveillance: ACOG committee opinion no. 828. Obstet Gynecol. 2021;137:e177-197.
- Mathews TJ, Hamilton BE. First births to older women continue to rise. NCHS Data Brief, No. 152. Hyattsville, MD: National Center for Health Statistics; 2014.
- Fretts RC, Schmittdiel J, McLean FH, et al. Increased maternal age and the risk of fetal death. N Engl J Med. 1995;333: 953-957.
- Lean SC, Derricott H, Jones RL, et al. Advanced maternal age and adverse pregnancy outcomes: a systematic review and meta-analysis. PLoS One. 2017;12:e0186287.
- Fretts RC, Elkins EB, Myers ER, et al. Should older women have antepartum testing to prevent unexplained stillbirth? Obstet Gynecol. 2004;104:56-64.
- Manning F, Wyn Pugh E, Boddy K. Effect of cigarette smoking on fetal breathing movements in normal pregnancies. Br Med J. 1975;1:552-553.
- Manning FA, Feyerabend C. Cigarette smoking and fetal breathing movements. Br J Obstet Gynecol. 1976;83:262-270.
- Gennser G, Marsal K, Brantmark B. Maternal smoking and fetal breathing movements. Am J Obstet Gynecol. 1975;123:861-867.
- Boylan P, O’Donovan P, Owens OJ. Fetal breathing movements and the diagnosis of labor: a prospective analysis of 100 cases. Obstet Gynecol. 1985;66:517-520.
- Kivikoski AI, Amon E, Vaalamo PO, et al. Effect of thirdtrimester premature rupture of membranes on fetal breathing movements: a prospective case-control study. Am J Obstet Gynecol. 1988;159:1474-1477.
- Peaceman AM, Meyer BA, Thorp JA, et al. The effect of magnesium sulfate tocolysis on the fetal biophysical profile. Am J Obstet Gynecol. 1989;161:771-774.
- Vintzileos AM, Campbell WA, Nochimson DJ, et al. The fetal biophysical profile in patients with premature rupture of the membranes—an early predictor of fetal infection. Am J Obstet Gynecol. 1985;152:501-516.
- Liston RM, Bloom K, Zimmer P. The psychological effects of counting fetal movements. Birth. 1994;21:135-140.
- Bellussi F, Po’ G, Livi A, et al. Fetal movement counting and perinatal mortality: a systematic review and meta-analysis. Obstet Gynecol. 2020;135:453-462.
- Freeman RK, Anderson G, Dorchester W. A prospective multiinstitutional study of antepartum fetal heart rate monitoring. I. Risk of perinatal mortality and morbidity according to antepartum fetal heart rate test results. Am J Obstet Gynecol. 1982;143:771-777.
- Miller DA , Rabello YA, Paul RH. The modified biophysical profile: antepartum testing in the 1990s. Am J Obstet Gynecol. 1996;174:812-817.
COVID-19 vaccination and pregnancy: What’s the latest?

COVID-19 vaccination is recommended for all reproductive-aged women, regardless of pregnancy status.1 Yet, national vaccination rates in pregnancy remain woefully low—lower than vaccine coverage rates for other recommended vaccines during pregnancy.2,3 COVID-19 infection has clearly documented risks for maternal and fetal health, and data continue to accumulate on the maternal and neonatal benefits of COVID-19 vaccination in pregnancy, as well as the safety of vaccination during pregnancy.
Maternal and neonatal benefits of COVID-19 vaccination
Does vaccination in pregnancy result in decreased rates of severe COVID-19 infection? Results from a study from a Louisiana health system comparing maternal outcomes between fully vaccinated (defined as 2 weeks after the final vaccine dose) and unvaccinated or partially vaccinated pregnant women during the delta variant—predominant COVID-19 surge clearly answer this question. Vaccination in pregnancy resulted in a 90% risk reduction in severe or critical COVID-19 infection and a 70% risk reduction in COVID-19 infection of any severity among fully vaccinated women. The study also provides some useful absolute numbers for patient counseling: Although none of the 1,332 vaccinated pregnant women in the study required supplemental oxygen or intensive care unit (ICU) admission, there was 1 maternal death, 5 ICU admissions, and 6 stillbirths among the 8,760 unvaccinated pregnant women.4
A larger population-based data set from Scotland and Israel demonstrated similar findings.5 Most importantly, the Scotland data, with most patients having had an mRNA-based vaccine, showed that, while 77% of all COVID-19 infections occurred in unvaccinated pregnant women, 91% of all hospital admissions occurred in unvaccinated women, and 98% of all critical care admissions occurred in unvaccinated women. Furthermore, although 13% of all COVID-19 hospitalizations in pregnancy occurred among vaccinated women, only 2% of critical care admissions occurred among vaccinated women. The Israeli experience (which identified nearly 30,000 eligible pregnancies from 1 of 4 state-mandated health funds in the country), demonstrated that the efficacy of the Pfizer/BioNTech vaccine to prevent a SARS-CoV-2 infection of any severity once fully vaccinated is more than 80%.6
Breakthrough infections, which were more prevalent during the omicron surge, have caused some patients to question the utility of COVID-19 vaccination. Recent data from South Africa, where the omicron variant was first identified, noted that efficacy of the Pfizer/ BioNTech vaccine to prevent hospitalization with COVID-19 infection during an omicron-predominant period was 70%—versus 93% efficacy in a delta-predominant period.7 These data, however, were in the absence of a booster dose, and in vitro studies suggest increased vaccine efficacy with a booster dose.8
Continue to: Counseling women on vaccination benefits and risks...
Counseling women on vaccination benefits and risks. No matter the specific numeric rate of efficacy against a COVID-19 infection, it is important to counsel women that the goal of vaccination is to prevent severe or critical COVID-19 infections, and these data all demonstrate that COVID-19 vaccination meets this goal. However, women may have additional questions regarding both fetal/neonatal benefits and safety with immunization in pregnancy.
Let us address the question of benefit first. In a large cohort of more than 1,300 women vaccinated during pregnancy and delivering at >34 weeks’ gestation, a few observations are worth noting.9 The first is that women who were fully vaccinated by the time of delivery had detectable antibodies at birth, even with first trimester vaccination, and these antibodies did cross the placenta to the neonate. Although higher maternal and neonatal antibody levels are achieved with early third trimester vaccination, it is key that women interpret this finding in light of 2 important points:
- women cannot know what gestational age they will deliver, thus waiting until the early third trimester for vaccination to optimize neonatal antibody levels could result in delivery prior to planned vaccination, with benefit for neither the woman nor the baby
- partial vaccination in the early third trimester resulted in lower maternal and neonatal antibody levels than full vaccination in the first trimester.
In addition, while the data were limited, a booster dose in the third trimester results in the highest antibody levels at delivery. Given the recommendation to initiate a booster dose 5 months after the completion of the primary vaccine series,10 many women will be eligible for a booster prior to delivery and thus can achieve the goals of high maternal and neonatal antibody levels simultaneously. One caveat to these data is that, while higher antibody levels seem comforting and may be better, we do not yet know the level of neonatal antibody necessary to decrease risks of COVID-19 infection in early newborn life.9 Recent data from the Centers for Disease Control and Prevention provide real-world evidence that maternal vaccination decreases the risk of hospitalization from COVID-19 for infants aged <6 months, with vaccine efficacy estimated to be 61% during a period of both Delta and Omicron predominance.11
The evidence is clear—the time for COVID-19 vaccination is now. There is no “optimal” time of vaccination in pregnancy for neonatal benefit that would be worth risking any amount of time a woman is susceptible to COVID-19, especially given the promising data regarding maternal and neonatal antibody levels achieved after a booster dose.
Although the COVID-19 vaccine is currently approved by the US Food and Drug Administration for ages 5 and above, Pfizer-BioNTech has plans to submit for approval for their vaccine’s use among kids as young as 6 months.1 Assuming that this approval occurs, this will leave newborns as the only group without possible vaccination against COVID-19. But can vaccination during pregnancy protect these infants against infection, as vaccination with the flu vaccine during pregnancy confers protective benefit to newborns?2
In a recent research letter published in Journal of the American Medical Association, Shook and colleagues present their data on antibody levels against COVID-19 present in newborns of women who were either naturally infected with COVID-19 at 20 to 32 weeks’ gestation (12 women) or who received mRNA vaccination during pregnancy at 20 to 32 weeks’ gestation (77 women).3 (They chose the 20- to 32-week timeframe during pregnancy because it had “demonstrated superior transplacental transfer of antibodies during this window.”)
They found that COVID-19 antibody levels were higher in both maternal and cord blood at birth in the women who were vaccinated versus the women who had infection. At 6 months, 16 of the 28 infants from the vaccinated-mother group had detectable antibodies compared with 1 of 12 infants from the infected-mother group. The researchers pointed out that the “antibody titer known to be protective against COVID-19 in infants is unknown;” however, they say that their findings provide further supportive evidence for COVID-19 vaccination in pregnant women.3
References
- Pfizer-BioNTech coronavirus vaccine for children under 5 could be available by the end of February, people with knowledge say. The Washington Post. https://www.washingtonpost.com /health/2022/01/31/coronavirus-vaccine-children-under-5/. Accessed February 11, 2022.
- Sakala IG, Honda-Okubo Y, Fung J, et al. Influenza immunization during pregnancy: benefits for mother and infant. Hum Vaccin Immunother. 2016;12:3065-3071. doi:10.1080/21645515.2016 .1215392.
- Shook LL, Atyeo CG, Yonker LM, et al. Durability of anti-spike antibodies in infants after maternal COVID-19 vaccination or natural infection. JAMA. doi:10.1001/jama.2022.1206.
Safety of COVID-19 vaccination: Current data
Risks for pregnancy loss, birth defects, and preterm delivery often are concerns of pregnant women considering a COVID-19 vaccination. Data from more than 2,400 women who submitted their information to the v-SAFE registry demonstrated a 14% risk for pregnancy loss between 6 and 20 weeks’ gestation—well within the expected rate of pregnancy loss in this gestational age range.12
Data from more than 46,000 pregnancies included in the Vaccine Safety Datalink, which includes data from health care organizations in 6 states, demonstrated a preterm birth rate of 6.6% and a small-for-gestational-age rate of 8.2% among fully vaccinated women, rates that were no different among unvaccinated women. There were no differences in the outcomes by trimester of vaccination, and these rates are comparable to the expected rates of these outcomes.13
Women also worry about the risks of vaccine side effects, such as fever or rare adverse events. Although all adverse events (ie, Guillain-Barre syndrome, pericarditis/myocarditis, thrombosis with thrombocytopenia syndrome [TTS]) are very rare, the American College of Obstetricians and Gynecologists does recommend that women get an mRNA COVID-19 vaccine, as the Johnson & Johnson/Janssen vaccine is associated with TTS, which occurred more commonly (although still rare) in women of reproductive age.14
Two large studies of typical side effects experienced after COVID-19 vaccination in pregnancy are incredibly reassuring. In the first, authors of a large study of more than 12,000 pregnant women enrolled in the v-SAFE registry reported that the most common side effect after each mRNA dose was injection site pain (88% after dose 1, 92% after dose 2).15 Self-reported fever occurred in 4% of women after dose 1 and 35% after dose 2. Although this frequency may seem high, a fever of 38.0°C (100.4°F) or higher only occurred among 8% of all participants.
In another study of almost 8,000 women self-reporting side effects (some of whom also may have contributed data to the v-SAFE study), fever occurred in approximately 5% after dose 1 and in about 20% after dose 2.16 In this study, the highest mean temperature was 38.1°C (100.6°F) after dose 1 and 38.2°C (100.7°F) after dose 2. Although it is a reasonable expectation for fever to follow COVID-19 vaccination, particularly after the second dose, the typical fever is a low-grade temperature that will not harm a developing fetus and will be responsive to acetaminophen administration. Moreover, if the fever were the harbinger of harm, then it might stand to reason that an increased signal of preterm delivery may be observed, but data from nearly 10,000 pregnant women vaccinated during the second or third trimesters showed no association with preterm birth (adjusted hazard ratio, 0.91; 95% confidence interval, 0.82–1.01).13
The bottom line
The data are clear. COVID-19 vaccination decreases the risks of severe infection in pregnancy, confers antibodies to neonates with at least some level of protection, and has no demonstrated harmful side effects in pregnancy. ●
- Interim clinical considerations for use of COVID-19 vaccines. CDC website. Published January 24, 2022. Accessed February 22, 2022. https://www.cdc.gov/vaccines/covid-19/clinical-considerations/covid-19-vaccines-us.html
- Cumulative data: percent of pregnant people aged 18-49 years receiving at least one dose of a COVID-19 vaccine during pregnancy overall, by race/ethnicity, and date reported to CDC—Vaccine Safety Datalink, United States. CDC website. Accessed February 22, 2022. https://data.cdc.gov/Vaccinations/Cumulative-Data-Percent-of-Pregnant-People-aged-18/4ht3-nbmd/data
- Razzaghi H, Kahn KE, Black CL, et al. Influenza and Tdap vaccination coverage among pregnant women—United States, April 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1391-1397.
- Morgan JA, Biggio JRJ, Martin JK, et al. Maternal outcomes after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in vaccinated compared with unvaccinated pregnant patients. Obstet Gynecol. 2022;139:107-109.
- Stock SJ, Carruthers J, Calvert C, et al. SARS-CoV-2 infection and COVID-19 vaccination rates in pregnant women in Scotland [published online January 13, 2022]. Nat Med. doi:10.1038/s41591-021-01666-2
- Goldshtein I, Nevo D, Steinberg DM, et al. Association between BNT162b2 vaccination and incidence of SARS-CoV-2 infection in pregnant women. JAMA. 2021;326:728-735.
- Collie S, Champion J, Moultrie H, et al. Effectiveness of BNT162b2 vaccine against omicron variant in South Africa [published online December 29, 2021]. N Engl J Med. doi:10.1056/NEJMc2119270
- Nemet I, Kliker L, Lustig Y, et al. Third BNT162b2 vaccination neutralization of SARS-CoV-2 omicron infection [published online December 29, 2021]. N Engl J Med. doi:10.1056/NEJMc2119358
- Yang YJ, Murphy EA, Singh S, et al. Association of gestational age at coronavirus disease 2019 (COVID-19) vaccination, history of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, and a vaccine booster dose with maternal and umbilical cord antibody levels at delivery [published online December 28, 2021]. Obstet Gynecol. doi:10.1097/AOG.0000000000004693
- COVID-19 vaccine booster shots. Centers for Disease Control and Prevention web site. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/booster-shot.html. Accessed March 2, 2022.
- Effectiveness of maternal vaccination with mRNA COVID-19 vaccine during pregnancy against COVID-19–associated hospitalization in infants aged <6 months—17 states, July 2021–January 2022. MMWR Morb Mortal Wkly Rep. 2022;71:264–270. doi: http://dx.doi.org/10.15585/mmwr.mm7107e3external icon.
- Zauche LH, Wallace B, Smoots AN, et al. Receipt of mRNA COVID-19 vaccines and risk of spontaneous abortion. N Engl J Med. 2021;385:1533-1535.
- Lipkind HS. Receipt of COVID-19 vaccine during pregnancy and preterm or small-for-gestational-age at birth—eight integrated health care organizations, United States, December 15, 2020–July 22, 2021. MMWR Morb Mortal Wkly Rep. doi:10.15585/mmwr.mm7101e1
- COVID-19 vaccination considerations for obstetric-gynecologic care. ACOG website. Updated February 8, 2022. Accessed February 22, 2022. https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2020/12/covid-19-vaccination-considerations-for-obstetric-gynecologic-care
- Shimabukuro TT, Kim SY, Myers TR, et al. Preliminary findings of mRNA COVID-19 vaccine safety in pregnant persons. N Engl J Med. 2021;384:2273-2282.
- Kachikis A, Englund JA, Singleton M, et al. Short-term reactions among pregnant and lactating individuals in the first wave of the COVID-19 vaccine rollout. JAMA Netw Open. 2021;4:E2121310.

COVID-19 vaccination is recommended for all reproductive-aged women, regardless of pregnancy status.1 Yet, national vaccination rates in pregnancy remain woefully low—lower than vaccine coverage rates for other recommended vaccines during pregnancy.2,3 COVID-19 infection has clearly documented risks for maternal and fetal health, and data continue to accumulate on the maternal and neonatal benefits of COVID-19 vaccination in pregnancy, as well as the safety of vaccination during pregnancy.
Maternal and neonatal benefits of COVID-19 vaccination
Does vaccination in pregnancy result in decreased rates of severe COVID-19 infection? Results from a study from a Louisiana health system comparing maternal outcomes between fully vaccinated (defined as 2 weeks after the final vaccine dose) and unvaccinated or partially vaccinated pregnant women during the delta variant—predominant COVID-19 surge clearly answer this question. Vaccination in pregnancy resulted in a 90% risk reduction in severe or critical COVID-19 infection and a 70% risk reduction in COVID-19 infection of any severity among fully vaccinated women. The study also provides some useful absolute numbers for patient counseling: Although none of the 1,332 vaccinated pregnant women in the study required supplemental oxygen or intensive care unit (ICU) admission, there was 1 maternal death, 5 ICU admissions, and 6 stillbirths among the 8,760 unvaccinated pregnant women.4
A larger population-based data set from Scotland and Israel demonstrated similar findings.5 Most importantly, the Scotland data, with most patients having had an mRNA-based vaccine, showed that, while 77% of all COVID-19 infections occurred in unvaccinated pregnant women, 91% of all hospital admissions occurred in unvaccinated women, and 98% of all critical care admissions occurred in unvaccinated women. Furthermore, although 13% of all COVID-19 hospitalizations in pregnancy occurred among vaccinated women, only 2% of critical care admissions occurred among vaccinated women. The Israeli experience (which identified nearly 30,000 eligible pregnancies from 1 of 4 state-mandated health funds in the country), demonstrated that the efficacy of the Pfizer/BioNTech vaccine to prevent a SARS-CoV-2 infection of any severity once fully vaccinated is more than 80%.6
Breakthrough infections, which were more prevalent during the omicron surge, have caused some patients to question the utility of COVID-19 vaccination. Recent data from South Africa, where the omicron variant was first identified, noted that efficacy of the Pfizer/ BioNTech vaccine to prevent hospitalization with COVID-19 infection during an omicron-predominant period was 70%—versus 93% efficacy in a delta-predominant period.7 These data, however, were in the absence of a booster dose, and in vitro studies suggest increased vaccine efficacy with a booster dose.8
Continue to: Counseling women on vaccination benefits and risks...
Counseling women on vaccination benefits and risks. No matter the specific numeric rate of efficacy against a COVID-19 infection, it is important to counsel women that the goal of vaccination is to prevent severe or critical COVID-19 infections, and these data all demonstrate that COVID-19 vaccination meets this goal. However, women may have additional questions regarding both fetal/neonatal benefits and safety with immunization in pregnancy.
Let us address the question of benefit first. In a large cohort of more than 1,300 women vaccinated during pregnancy and delivering at >34 weeks’ gestation, a few observations are worth noting.9 The first is that women who were fully vaccinated by the time of delivery had detectable antibodies at birth, even with first trimester vaccination, and these antibodies did cross the placenta to the neonate. Although higher maternal and neonatal antibody levels are achieved with early third trimester vaccination, it is key that women interpret this finding in light of 2 important points:
- women cannot know what gestational age they will deliver, thus waiting until the early third trimester for vaccination to optimize neonatal antibody levels could result in delivery prior to planned vaccination, with benefit for neither the woman nor the baby
- partial vaccination in the early third trimester resulted in lower maternal and neonatal antibody levels than full vaccination in the first trimester.
In addition, while the data were limited, a booster dose in the third trimester results in the highest antibody levels at delivery. Given the recommendation to initiate a booster dose 5 months after the completion of the primary vaccine series,10 many women will be eligible for a booster prior to delivery and thus can achieve the goals of high maternal and neonatal antibody levels simultaneously. One caveat to these data is that, while higher antibody levels seem comforting and may be better, we do not yet know the level of neonatal antibody necessary to decrease risks of COVID-19 infection in early newborn life.9 Recent data from the Centers for Disease Control and Prevention provide real-world evidence that maternal vaccination decreases the risk of hospitalization from COVID-19 for infants aged <6 months, with vaccine efficacy estimated to be 61% during a period of both Delta and Omicron predominance.11
The evidence is clear—the time for COVID-19 vaccination is now. There is no “optimal” time of vaccination in pregnancy for neonatal benefit that would be worth risking any amount of time a woman is susceptible to COVID-19, especially given the promising data regarding maternal and neonatal antibody levels achieved after a booster dose.
Although the COVID-19 vaccine is currently approved by the US Food and Drug Administration for ages 5 and above, Pfizer-BioNTech has plans to submit for approval for their vaccine’s use among kids as young as 6 months.1 Assuming that this approval occurs, this will leave newborns as the only group without possible vaccination against COVID-19. But can vaccination during pregnancy protect these infants against infection, as vaccination with the flu vaccine during pregnancy confers protective benefit to newborns?2
In a recent research letter published in Journal of the American Medical Association, Shook and colleagues present their data on antibody levels against COVID-19 present in newborns of women who were either naturally infected with COVID-19 at 20 to 32 weeks’ gestation (12 women) or who received mRNA vaccination during pregnancy at 20 to 32 weeks’ gestation (77 women).3 (They chose the 20- to 32-week timeframe during pregnancy because it had “demonstrated superior transplacental transfer of antibodies during this window.”)
They found that COVID-19 antibody levels were higher in both maternal and cord blood at birth in the women who were vaccinated versus the women who had infection. At 6 months, 16 of the 28 infants from the vaccinated-mother group had detectable antibodies compared with 1 of 12 infants from the infected-mother group. The researchers pointed out that the “antibody titer known to be protective against COVID-19 in infants is unknown;” however, they say that their findings provide further supportive evidence for COVID-19 vaccination in pregnant women.3
References
- Pfizer-BioNTech coronavirus vaccine for children under 5 could be available by the end of February, people with knowledge say. The Washington Post. https://www.washingtonpost.com /health/2022/01/31/coronavirus-vaccine-children-under-5/. Accessed February 11, 2022.
- Sakala IG, Honda-Okubo Y, Fung J, et al. Influenza immunization during pregnancy: benefits for mother and infant. Hum Vaccin Immunother. 2016;12:3065-3071. doi:10.1080/21645515.2016 .1215392.
- Shook LL, Atyeo CG, Yonker LM, et al. Durability of anti-spike antibodies in infants after maternal COVID-19 vaccination or natural infection. JAMA. doi:10.1001/jama.2022.1206.
Safety of COVID-19 vaccination: Current data
Risks for pregnancy loss, birth defects, and preterm delivery often are concerns of pregnant women considering a COVID-19 vaccination. Data from more than 2,400 women who submitted their information to the v-SAFE registry demonstrated a 14% risk for pregnancy loss between 6 and 20 weeks’ gestation—well within the expected rate of pregnancy loss in this gestational age range.12
Data from more than 46,000 pregnancies included in the Vaccine Safety Datalink, which includes data from health care organizations in 6 states, demonstrated a preterm birth rate of 6.6% and a small-for-gestational-age rate of 8.2% among fully vaccinated women, rates that were no different among unvaccinated women. There were no differences in the outcomes by trimester of vaccination, and these rates are comparable to the expected rates of these outcomes.13
Women also worry about the risks of vaccine side effects, such as fever or rare adverse events. Although all adverse events (ie, Guillain-Barre syndrome, pericarditis/myocarditis, thrombosis with thrombocytopenia syndrome [TTS]) are very rare, the American College of Obstetricians and Gynecologists does recommend that women get an mRNA COVID-19 vaccine, as the Johnson & Johnson/Janssen vaccine is associated with TTS, which occurred more commonly (although still rare) in women of reproductive age.14
Two large studies of typical side effects experienced after COVID-19 vaccination in pregnancy are incredibly reassuring. In the first, authors of a large study of more than 12,000 pregnant women enrolled in the v-SAFE registry reported that the most common side effect after each mRNA dose was injection site pain (88% after dose 1, 92% after dose 2).15 Self-reported fever occurred in 4% of women after dose 1 and 35% after dose 2. Although this frequency may seem high, a fever of 38.0°C (100.4°F) or higher only occurred among 8% of all participants.
In another study of almost 8,000 women self-reporting side effects (some of whom also may have contributed data to the v-SAFE study), fever occurred in approximately 5% after dose 1 and in about 20% after dose 2.16 In this study, the highest mean temperature was 38.1°C (100.6°F) after dose 1 and 38.2°C (100.7°F) after dose 2. Although it is a reasonable expectation for fever to follow COVID-19 vaccination, particularly after the second dose, the typical fever is a low-grade temperature that will not harm a developing fetus and will be responsive to acetaminophen administration. Moreover, if the fever were the harbinger of harm, then it might stand to reason that an increased signal of preterm delivery may be observed, but data from nearly 10,000 pregnant women vaccinated during the second or third trimesters showed no association with preterm birth (adjusted hazard ratio, 0.91; 95% confidence interval, 0.82–1.01).13
The bottom line
The data are clear. COVID-19 vaccination decreases the risks of severe infection in pregnancy, confers antibodies to neonates with at least some level of protection, and has no demonstrated harmful side effects in pregnancy. ●

COVID-19 vaccination is recommended for all reproductive-aged women, regardless of pregnancy status.1 Yet, national vaccination rates in pregnancy remain woefully low—lower than vaccine coverage rates for other recommended vaccines during pregnancy.2,3 COVID-19 infection has clearly documented risks for maternal and fetal health, and data continue to accumulate on the maternal and neonatal benefits of COVID-19 vaccination in pregnancy, as well as the safety of vaccination during pregnancy.
Maternal and neonatal benefits of COVID-19 vaccination
Does vaccination in pregnancy result in decreased rates of severe COVID-19 infection? Results from a study from a Louisiana health system comparing maternal outcomes between fully vaccinated (defined as 2 weeks after the final vaccine dose) and unvaccinated or partially vaccinated pregnant women during the delta variant—predominant COVID-19 surge clearly answer this question. Vaccination in pregnancy resulted in a 90% risk reduction in severe or critical COVID-19 infection and a 70% risk reduction in COVID-19 infection of any severity among fully vaccinated women. The study also provides some useful absolute numbers for patient counseling: Although none of the 1,332 vaccinated pregnant women in the study required supplemental oxygen or intensive care unit (ICU) admission, there was 1 maternal death, 5 ICU admissions, and 6 stillbirths among the 8,760 unvaccinated pregnant women.4
A larger population-based data set from Scotland and Israel demonstrated similar findings.5 Most importantly, the Scotland data, with most patients having had an mRNA-based vaccine, showed that, while 77% of all COVID-19 infections occurred in unvaccinated pregnant women, 91% of all hospital admissions occurred in unvaccinated women, and 98% of all critical care admissions occurred in unvaccinated women. Furthermore, although 13% of all COVID-19 hospitalizations in pregnancy occurred among vaccinated women, only 2% of critical care admissions occurred among vaccinated women. The Israeli experience (which identified nearly 30,000 eligible pregnancies from 1 of 4 state-mandated health funds in the country), demonstrated that the efficacy of the Pfizer/BioNTech vaccine to prevent a SARS-CoV-2 infection of any severity once fully vaccinated is more than 80%.6
Breakthrough infections, which were more prevalent during the omicron surge, have caused some patients to question the utility of COVID-19 vaccination. Recent data from South Africa, where the omicron variant was first identified, noted that efficacy of the Pfizer/ BioNTech vaccine to prevent hospitalization with COVID-19 infection during an omicron-predominant period was 70%—versus 93% efficacy in a delta-predominant period.7 These data, however, were in the absence of a booster dose, and in vitro studies suggest increased vaccine efficacy with a booster dose.8
Continue to: Counseling women on vaccination benefits and risks...
Counseling women on vaccination benefits and risks. No matter the specific numeric rate of efficacy against a COVID-19 infection, it is important to counsel women that the goal of vaccination is to prevent severe or critical COVID-19 infections, and these data all demonstrate that COVID-19 vaccination meets this goal. However, women may have additional questions regarding both fetal/neonatal benefits and safety with immunization in pregnancy.
Let us address the question of benefit first. In a large cohort of more than 1,300 women vaccinated during pregnancy and delivering at >34 weeks’ gestation, a few observations are worth noting.9 The first is that women who were fully vaccinated by the time of delivery had detectable antibodies at birth, even with first trimester vaccination, and these antibodies did cross the placenta to the neonate. Although higher maternal and neonatal antibody levels are achieved with early third trimester vaccination, it is key that women interpret this finding in light of 2 important points:
- women cannot know what gestational age they will deliver, thus waiting until the early third trimester for vaccination to optimize neonatal antibody levels could result in delivery prior to planned vaccination, with benefit for neither the woman nor the baby
- partial vaccination in the early third trimester resulted in lower maternal and neonatal antibody levels than full vaccination in the first trimester.
In addition, while the data were limited, a booster dose in the third trimester results in the highest antibody levels at delivery. Given the recommendation to initiate a booster dose 5 months after the completion of the primary vaccine series,10 many women will be eligible for a booster prior to delivery and thus can achieve the goals of high maternal and neonatal antibody levels simultaneously. One caveat to these data is that, while higher antibody levels seem comforting and may be better, we do not yet know the level of neonatal antibody necessary to decrease risks of COVID-19 infection in early newborn life.9 Recent data from the Centers for Disease Control and Prevention provide real-world evidence that maternal vaccination decreases the risk of hospitalization from COVID-19 for infants aged <6 months, with vaccine efficacy estimated to be 61% during a period of both Delta and Omicron predominance.11
The evidence is clear—the time for COVID-19 vaccination is now. There is no “optimal” time of vaccination in pregnancy for neonatal benefit that would be worth risking any amount of time a woman is susceptible to COVID-19, especially given the promising data regarding maternal and neonatal antibody levels achieved after a booster dose.
Although the COVID-19 vaccine is currently approved by the US Food and Drug Administration for ages 5 and above, Pfizer-BioNTech has plans to submit for approval for their vaccine’s use among kids as young as 6 months.1 Assuming that this approval occurs, this will leave newborns as the only group without possible vaccination against COVID-19. But can vaccination during pregnancy protect these infants against infection, as vaccination with the flu vaccine during pregnancy confers protective benefit to newborns?2
In a recent research letter published in Journal of the American Medical Association, Shook and colleagues present their data on antibody levels against COVID-19 present in newborns of women who were either naturally infected with COVID-19 at 20 to 32 weeks’ gestation (12 women) or who received mRNA vaccination during pregnancy at 20 to 32 weeks’ gestation (77 women).3 (They chose the 20- to 32-week timeframe during pregnancy because it had “demonstrated superior transplacental transfer of antibodies during this window.”)
They found that COVID-19 antibody levels were higher in both maternal and cord blood at birth in the women who were vaccinated versus the women who had infection. At 6 months, 16 of the 28 infants from the vaccinated-mother group had detectable antibodies compared with 1 of 12 infants from the infected-mother group. The researchers pointed out that the “antibody titer known to be protective against COVID-19 in infants is unknown;” however, they say that their findings provide further supportive evidence for COVID-19 vaccination in pregnant women.3
References
- Pfizer-BioNTech coronavirus vaccine for children under 5 could be available by the end of February, people with knowledge say. The Washington Post. https://www.washingtonpost.com /health/2022/01/31/coronavirus-vaccine-children-under-5/. Accessed February 11, 2022.
- Sakala IG, Honda-Okubo Y, Fung J, et al. Influenza immunization during pregnancy: benefits for mother and infant. Hum Vaccin Immunother. 2016;12:3065-3071. doi:10.1080/21645515.2016 .1215392.
- Shook LL, Atyeo CG, Yonker LM, et al. Durability of anti-spike antibodies in infants after maternal COVID-19 vaccination or natural infection. JAMA. doi:10.1001/jama.2022.1206.
Safety of COVID-19 vaccination: Current data
Risks for pregnancy loss, birth defects, and preterm delivery often are concerns of pregnant women considering a COVID-19 vaccination. Data from more than 2,400 women who submitted their information to the v-SAFE registry demonstrated a 14% risk for pregnancy loss between 6 and 20 weeks’ gestation—well within the expected rate of pregnancy loss in this gestational age range.12
Data from more than 46,000 pregnancies included in the Vaccine Safety Datalink, which includes data from health care organizations in 6 states, demonstrated a preterm birth rate of 6.6% and a small-for-gestational-age rate of 8.2% among fully vaccinated women, rates that were no different among unvaccinated women. There were no differences in the outcomes by trimester of vaccination, and these rates are comparable to the expected rates of these outcomes.13
Women also worry about the risks of vaccine side effects, such as fever or rare adverse events. Although all adverse events (ie, Guillain-Barre syndrome, pericarditis/myocarditis, thrombosis with thrombocytopenia syndrome [TTS]) are very rare, the American College of Obstetricians and Gynecologists does recommend that women get an mRNA COVID-19 vaccine, as the Johnson & Johnson/Janssen vaccine is associated with TTS, which occurred more commonly (although still rare) in women of reproductive age.14
Two large studies of typical side effects experienced after COVID-19 vaccination in pregnancy are incredibly reassuring. In the first, authors of a large study of more than 12,000 pregnant women enrolled in the v-SAFE registry reported that the most common side effect after each mRNA dose was injection site pain (88% after dose 1, 92% after dose 2).15 Self-reported fever occurred in 4% of women after dose 1 and 35% after dose 2. Although this frequency may seem high, a fever of 38.0°C (100.4°F) or higher only occurred among 8% of all participants.
In another study of almost 8,000 women self-reporting side effects (some of whom also may have contributed data to the v-SAFE study), fever occurred in approximately 5% after dose 1 and in about 20% after dose 2.16 In this study, the highest mean temperature was 38.1°C (100.6°F) after dose 1 and 38.2°C (100.7°F) after dose 2. Although it is a reasonable expectation for fever to follow COVID-19 vaccination, particularly after the second dose, the typical fever is a low-grade temperature that will not harm a developing fetus and will be responsive to acetaminophen administration. Moreover, if the fever were the harbinger of harm, then it might stand to reason that an increased signal of preterm delivery may be observed, but data from nearly 10,000 pregnant women vaccinated during the second or third trimesters showed no association with preterm birth (adjusted hazard ratio, 0.91; 95% confidence interval, 0.82–1.01).13
The bottom line
The data are clear. COVID-19 vaccination decreases the risks of severe infection in pregnancy, confers antibodies to neonates with at least some level of protection, and has no demonstrated harmful side effects in pregnancy. ●
- Interim clinical considerations for use of COVID-19 vaccines. CDC website. Published January 24, 2022. Accessed February 22, 2022. https://www.cdc.gov/vaccines/covid-19/clinical-considerations/covid-19-vaccines-us.html
- Cumulative data: percent of pregnant people aged 18-49 years receiving at least one dose of a COVID-19 vaccine during pregnancy overall, by race/ethnicity, and date reported to CDC—Vaccine Safety Datalink, United States. CDC website. Accessed February 22, 2022. https://data.cdc.gov/Vaccinations/Cumulative-Data-Percent-of-Pregnant-People-aged-18/4ht3-nbmd/data
- Razzaghi H, Kahn KE, Black CL, et al. Influenza and Tdap vaccination coverage among pregnant women—United States, April 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1391-1397.
- Morgan JA, Biggio JRJ, Martin JK, et al. Maternal outcomes after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in vaccinated compared with unvaccinated pregnant patients. Obstet Gynecol. 2022;139:107-109.
- Stock SJ, Carruthers J, Calvert C, et al. SARS-CoV-2 infection and COVID-19 vaccination rates in pregnant women in Scotland [published online January 13, 2022]. Nat Med. doi:10.1038/s41591-021-01666-2
- Goldshtein I, Nevo D, Steinberg DM, et al. Association between BNT162b2 vaccination and incidence of SARS-CoV-2 infection in pregnant women. JAMA. 2021;326:728-735.
- Collie S, Champion J, Moultrie H, et al. Effectiveness of BNT162b2 vaccine against omicron variant in South Africa [published online December 29, 2021]. N Engl J Med. doi:10.1056/NEJMc2119270
- Nemet I, Kliker L, Lustig Y, et al. Third BNT162b2 vaccination neutralization of SARS-CoV-2 omicron infection [published online December 29, 2021]. N Engl J Med. doi:10.1056/NEJMc2119358
- Yang YJ, Murphy EA, Singh S, et al. Association of gestational age at coronavirus disease 2019 (COVID-19) vaccination, history of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, and a vaccine booster dose with maternal and umbilical cord antibody levels at delivery [published online December 28, 2021]. Obstet Gynecol. doi:10.1097/AOG.0000000000004693
- COVID-19 vaccine booster shots. Centers for Disease Control and Prevention web site. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/booster-shot.html. Accessed March 2, 2022.
- Effectiveness of maternal vaccination with mRNA COVID-19 vaccine during pregnancy against COVID-19–associated hospitalization in infants aged <6 months—17 states, July 2021–January 2022. MMWR Morb Mortal Wkly Rep. 2022;71:264–270. doi: http://dx.doi.org/10.15585/mmwr.mm7107e3external icon.
- Zauche LH, Wallace B, Smoots AN, et al. Receipt of mRNA COVID-19 vaccines and risk of spontaneous abortion. N Engl J Med. 2021;385:1533-1535.
- Lipkind HS. Receipt of COVID-19 vaccine during pregnancy and preterm or small-for-gestational-age at birth—eight integrated health care organizations, United States, December 15, 2020–July 22, 2021. MMWR Morb Mortal Wkly Rep. doi:10.15585/mmwr.mm7101e1
- COVID-19 vaccination considerations for obstetric-gynecologic care. ACOG website. Updated February 8, 2022. Accessed February 22, 2022. https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2020/12/covid-19-vaccination-considerations-for-obstetric-gynecologic-care
- Shimabukuro TT, Kim SY, Myers TR, et al. Preliminary findings of mRNA COVID-19 vaccine safety in pregnant persons. N Engl J Med. 2021;384:2273-2282.
- Kachikis A, Englund JA, Singleton M, et al. Short-term reactions among pregnant and lactating individuals in the first wave of the COVID-19 vaccine rollout. JAMA Netw Open. 2021;4:E2121310.
- Interim clinical considerations for use of COVID-19 vaccines. CDC website. Published January 24, 2022. Accessed February 22, 2022. https://www.cdc.gov/vaccines/covid-19/clinical-considerations/covid-19-vaccines-us.html
- Cumulative data: percent of pregnant people aged 18-49 years receiving at least one dose of a COVID-19 vaccine during pregnancy overall, by race/ethnicity, and date reported to CDC—Vaccine Safety Datalink, United States. CDC website. Accessed February 22, 2022. https://data.cdc.gov/Vaccinations/Cumulative-Data-Percent-of-Pregnant-People-aged-18/4ht3-nbmd/data
- Razzaghi H, Kahn KE, Black CL, et al. Influenza and Tdap vaccination coverage among pregnant women—United States, April 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1391-1397.
- Morgan JA, Biggio JRJ, Martin JK, et al. Maternal outcomes after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in vaccinated compared with unvaccinated pregnant patients. Obstet Gynecol. 2022;139:107-109.
- Stock SJ, Carruthers J, Calvert C, et al. SARS-CoV-2 infection and COVID-19 vaccination rates in pregnant women in Scotland [published online January 13, 2022]. Nat Med. doi:10.1038/s41591-021-01666-2
- Goldshtein I, Nevo D, Steinberg DM, et al. Association between BNT162b2 vaccination and incidence of SARS-CoV-2 infection in pregnant women. JAMA. 2021;326:728-735.
- Collie S, Champion J, Moultrie H, et al. Effectiveness of BNT162b2 vaccine against omicron variant in South Africa [published online December 29, 2021]. N Engl J Med. doi:10.1056/NEJMc2119270
- Nemet I, Kliker L, Lustig Y, et al. Third BNT162b2 vaccination neutralization of SARS-CoV-2 omicron infection [published online December 29, 2021]. N Engl J Med. doi:10.1056/NEJMc2119358
- Yang YJ, Murphy EA, Singh S, et al. Association of gestational age at coronavirus disease 2019 (COVID-19) vaccination, history of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, and a vaccine booster dose with maternal and umbilical cord antibody levels at delivery [published online December 28, 2021]. Obstet Gynecol. doi:10.1097/AOG.0000000000004693
- COVID-19 vaccine booster shots. Centers for Disease Control and Prevention web site. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/booster-shot.html. Accessed March 2, 2022.
- Effectiveness of maternal vaccination with mRNA COVID-19 vaccine during pregnancy against COVID-19–associated hospitalization in infants aged <6 months—17 states, July 2021–January 2022. MMWR Morb Mortal Wkly Rep. 2022;71:264–270. doi: http://dx.doi.org/10.15585/mmwr.mm7107e3external icon.
- Zauche LH, Wallace B, Smoots AN, et al. Receipt of mRNA COVID-19 vaccines and risk of spontaneous abortion. N Engl J Med. 2021;385:1533-1535.
- Lipkind HS. Receipt of COVID-19 vaccine during pregnancy and preterm or small-for-gestational-age at birth—eight integrated health care organizations, United States, December 15, 2020–July 22, 2021. MMWR Morb Mortal Wkly Rep. doi:10.15585/mmwr.mm7101e1
- COVID-19 vaccination considerations for obstetric-gynecologic care. ACOG website. Updated February 8, 2022. Accessed February 22, 2022. https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2020/12/covid-19-vaccination-considerations-for-obstetric-gynecologic-care
- Shimabukuro TT, Kim SY, Myers TR, et al. Preliminary findings of mRNA COVID-19 vaccine safety in pregnant persons. N Engl J Med. 2021;384:2273-2282.
- Kachikis A, Englund JA, Singleton M, et al. Short-term reactions among pregnant and lactating individuals in the first wave of the COVID-19 vaccine rollout. JAMA Netw Open. 2021;4:E2121310.
Is the United States addressing maternal mortality rates from preeclampsia/eclampsia and chronic hypertension?
Ananth CV, Brandt JS, Hill J, et al. Historical and recent changes in maternal mortality due to hypertensive disorders in the United States, 1979 to 2018. Hypertension. 2021;78:1414–1422. doi: 10.1161/HYPERTENSIONAHA.121.17661.
EXPERT COMMENTARY
Maternal mortality is a pressing public health issue and is largely preventable. Up to 10% of all US pregnancies are complicated by a hypertensive disorder, and rates of chronic hypertension and severe preeclampsia have steadily increased over the last 4 decades. However, maternal mortality is an outcome in a population with advancing maternal age, increasing obesity, and undermanaged chronic disease. The MMR due to hypertension is substantially higher among Black women compared with White women. Countless studies attribute systemic racism to these disparities.
Details of the study
Spanning 40 years, a recent study by Ananth and colleagues included live births across all 50 United States and Washington, DC. Of the 1.5 million live births examined, there were 3,287 hypertension-related maternal deaths.
Data were deidentified and available in the public domain. The researchers compiled mortality data and live births among women aged 15 to 49. The MMR was considered the death of a woman during pregnancy or within the 42 days following a live birth.
Key points of the study included:
- An estimated two-thirds of maternal deaths are preventable.
- The hypertension-related MMR was 2.1 per 100,000 live births.
- Preeclampsia-related MMR decreased, while hypertension-related MMR increased.
- The MMR from chronic hypertension has increased annually by 9.2%.
- Pregnancies among women with advanced maternal age have grown, especially among those over age 40.
- The MMR due to hypertension increases with age and is highest among women age 45 to 49.
Study strengths and limitations
A major strength of this study is the sheer size of the sample. This is one of the largest studies that examined changes in the MMR in the United States.
As with any study that spans a long period, a primary limitation is inconsistencies in the data collected. In 2003, the US death certificate was revised to include a set of “pregnancy checkboxes” indicating pregnancy at the time of death.
There also have been shifts in diagnostic coding and criteria for preeclampsia.
Classification of race and ethnicity has improved and broadened over time. Despite these limitations, the overarching trends are compelling. ●
This study’s authors note that maternal mortality is largely preventable. Patients need to be aware of their health and how to adopt healthy behaviors long before pregnancy is even a consideration. Primary and secondary prevention are essential for reducing the MMR.
Clinicians who care for women have an opportunity to emphasize cardiac health at every visit. This includes strict blood pressure control through modifiable behaviors like diet and exercise. The busy clinician could consider a 1- to 2-minute pitch to emphasize that heart disease is the leading cause of death in women both during pregnancy and later in life. A tool from the American Heart Association, Life’s Simple 7 (https://www.heart .org/en/healthy-living/healthy-lifestyle/my-life-check--lifes-sim ple-7), can help guide this language.
In office and clinical settings, consider strategies to raise awareness among staff and colleagues about cultural sensitivities to improve the health of all patients. Addressing systemic racism in the US health care system is critical to mitigate racial inequities in the rates of MMR. An editorial in The New England Journal of Medicine urges clinicians to observe patient color rather than be “color blind.”1 The editorialists note that “physician-citizens must recognize the harm inflicted by discrimination and racism and consider this environmental agent of disease as a vital sign— alongside blood pressure, pulse, weight, and temperature—that provides important information about a patient’s condition.”1
LAUREN B. GOLFER, WHNP-BC, AND MARY L. ROSSER, MD, PHD
- Evans MK, Rosenbaum L, Malina D, et al. Diagnosing and treating systemic racism. N Engl J Med. 2020;383:274-276. https://www.nejm.org/doi/full/10.1056/NEJMe2021693. Accessed February 24, 2022.
Ananth CV, Brandt JS, Hill J, et al. Historical and recent changes in maternal mortality due to hypertensive disorders in the United States, 1979 to 2018. Hypertension. 2021;78:1414–1422. doi: 10.1161/HYPERTENSIONAHA.121.17661.
EXPERT COMMENTARY
Maternal mortality is a pressing public health issue and is largely preventable. Up to 10% of all US pregnancies are complicated by a hypertensive disorder, and rates of chronic hypertension and severe preeclampsia have steadily increased over the last 4 decades. However, maternal mortality is an outcome in a population with advancing maternal age, increasing obesity, and undermanaged chronic disease. The MMR due to hypertension is substantially higher among Black women compared with White women. Countless studies attribute systemic racism to these disparities.
Details of the study
Spanning 40 years, a recent study by Ananth and colleagues included live births across all 50 United States and Washington, DC. Of the 1.5 million live births examined, there were 3,287 hypertension-related maternal deaths.
Data were deidentified and available in the public domain. The researchers compiled mortality data and live births among women aged 15 to 49. The MMR was considered the death of a woman during pregnancy or within the 42 days following a live birth.
Key points of the study included:
- An estimated two-thirds of maternal deaths are preventable.
- The hypertension-related MMR was 2.1 per 100,000 live births.
- Preeclampsia-related MMR decreased, while hypertension-related MMR increased.
- The MMR from chronic hypertension has increased annually by 9.2%.
- Pregnancies among women with advanced maternal age have grown, especially among those over age 40.
- The MMR due to hypertension increases with age and is highest among women age 45 to 49.
Study strengths and limitations
A major strength of this study is the sheer size of the sample. This is one of the largest studies that examined changes in the MMR in the United States.
As with any study that spans a long period, a primary limitation is inconsistencies in the data collected. In 2003, the US death certificate was revised to include a set of “pregnancy checkboxes” indicating pregnancy at the time of death.
There also have been shifts in diagnostic coding and criteria for preeclampsia.
Classification of race and ethnicity has improved and broadened over time. Despite these limitations, the overarching trends are compelling. ●
This study’s authors note that maternal mortality is largely preventable. Patients need to be aware of their health and how to adopt healthy behaviors long before pregnancy is even a consideration. Primary and secondary prevention are essential for reducing the MMR.
Clinicians who care for women have an opportunity to emphasize cardiac health at every visit. This includes strict blood pressure control through modifiable behaviors like diet and exercise. The busy clinician could consider a 1- to 2-minute pitch to emphasize that heart disease is the leading cause of death in women both during pregnancy and later in life. A tool from the American Heart Association, Life’s Simple 7 (https://www.heart .org/en/healthy-living/healthy-lifestyle/my-life-check--lifes-sim ple-7), can help guide this language.
In office and clinical settings, consider strategies to raise awareness among staff and colleagues about cultural sensitivities to improve the health of all patients. Addressing systemic racism in the US health care system is critical to mitigate racial inequities in the rates of MMR. An editorial in The New England Journal of Medicine urges clinicians to observe patient color rather than be “color blind.”1 The editorialists note that “physician-citizens must recognize the harm inflicted by discrimination and racism and consider this environmental agent of disease as a vital sign— alongside blood pressure, pulse, weight, and temperature—that provides important information about a patient’s condition.”1
LAUREN B. GOLFER, WHNP-BC, AND MARY L. ROSSER, MD, PHD
Ananth CV, Brandt JS, Hill J, et al. Historical and recent changes in maternal mortality due to hypertensive disorders in the United States, 1979 to 2018. Hypertension. 2021;78:1414–1422. doi: 10.1161/HYPERTENSIONAHA.121.17661.
EXPERT COMMENTARY
Maternal mortality is a pressing public health issue and is largely preventable. Up to 10% of all US pregnancies are complicated by a hypertensive disorder, and rates of chronic hypertension and severe preeclampsia have steadily increased over the last 4 decades. However, maternal mortality is an outcome in a population with advancing maternal age, increasing obesity, and undermanaged chronic disease. The MMR due to hypertension is substantially higher among Black women compared with White women. Countless studies attribute systemic racism to these disparities.
Details of the study
Spanning 40 years, a recent study by Ananth and colleagues included live births across all 50 United States and Washington, DC. Of the 1.5 million live births examined, there were 3,287 hypertension-related maternal deaths.
Data were deidentified and available in the public domain. The researchers compiled mortality data and live births among women aged 15 to 49. The MMR was considered the death of a woman during pregnancy or within the 42 days following a live birth.
Key points of the study included:
- An estimated two-thirds of maternal deaths are preventable.
- The hypertension-related MMR was 2.1 per 100,000 live births.
- Preeclampsia-related MMR decreased, while hypertension-related MMR increased.
- The MMR from chronic hypertension has increased annually by 9.2%.
- Pregnancies among women with advanced maternal age have grown, especially among those over age 40.
- The MMR due to hypertension increases with age and is highest among women age 45 to 49.
Study strengths and limitations
A major strength of this study is the sheer size of the sample. This is one of the largest studies that examined changes in the MMR in the United States.
As with any study that spans a long period, a primary limitation is inconsistencies in the data collected. In 2003, the US death certificate was revised to include a set of “pregnancy checkboxes” indicating pregnancy at the time of death.
There also have been shifts in diagnostic coding and criteria for preeclampsia.
Classification of race and ethnicity has improved and broadened over time. Despite these limitations, the overarching trends are compelling. ●
This study’s authors note that maternal mortality is largely preventable. Patients need to be aware of their health and how to adopt healthy behaviors long before pregnancy is even a consideration. Primary and secondary prevention are essential for reducing the MMR.
Clinicians who care for women have an opportunity to emphasize cardiac health at every visit. This includes strict blood pressure control through modifiable behaviors like diet and exercise. The busy clinician could consider a 1- to 2-minute pitch to emphasize that heart disease is the leading cause of death in women both during pregnancy and later in life. A tool from the American Heart Association, Life’s Simple 7 (https://www.heart .org/en/healthy-living/healthy-lifestyle/my-life-check--lifes-sim ple-7), can help guide this language.
In office and clinical settings, consider strategies to raise awareness among staff and colleagues about cultural sensitivities to improve the health of all patients. Addressing systemic racism in the US health care system is critical to mitigate racial inequities in the rates of MMR. An editorial in The New England Journal of Medicine urges clinicians to observe patient color rather than be “color blind.”1 The editorialists note that “physician-citizens must recognize the harm inflicted by discrimination and racism and consider this environmental agent of disease as a vital sign— alongside blood pressure, pulse, weight, and temperature—that provides important information about a patient’s condition.”1
LAUREN B. GOLFER, WHNP-BC, AND MARY L. ROSSER, MD, PHD
- Evans MK, Rosenbaum L, Malina D, et al. Diagnosing and treating systemic racism. N Engl J Med. 2020;383:274-276. https://www.nejm.org/doi/full/10.1056/NEJMe2021693. Accessed February 24, 2022.
- Evans MK, Rosenbaum L, Malina D, et al. Diagnosing and treating systemic racism. N Engl J Med. 2020;383:274-276. https://www.nejm.org/doi/full/10.1056/NEJMe2021693. Accessed February 24, 2022.
Will NAAT replace microscopy for the identification of organisms causing vaginitis?
Over the past 200 years, identification of the specific organism causing an infection has evolved from a reliance on patient history and physical examination to the use of microscopic examination of relevant biological samples to the rise of microbial culture and immunological testing as the gold standards for diagnosis. More recently, advances in nucleic acid testing have made nucleic acid amplification testing (NAAT) a primary method for identifying the specific organism causing an infection.
The evolution of the diagnosis of gonorrhea in clinical practice is a good example of the inexorable evolution of diagnostic techniques from physical examination to microscopic analysis to culture and finally to NAAT. Neiseer discovered Neisseria gonorrhea in 1879.1 In 19th century general medical practice gonorrhea was often diagnosed based on history and physical examination and sometimes microscopy was also utilized.2 In the mid-20th century, it was realized that culture was a superior approach to diagnosis of gonorrhea, and it became the gold standard for diagnosis in general practice.3 NAAT has now replaced culture as the gold standard for the diagnosis of gonorrhea because of its superior performance in clinical practice.4 It may now be time to consider using NAAT rather than microscopy and culture in general practice for the identification of specific microorganisms causing vaginitis.
Trichomoniasis
Vaginitis caused by Trichomonas vaginalis is characterized by a discharge that is foamy and green-yellow in color, with a vaginal pH that is >4.5. Microscopy of a vaginal specimen has low sensitivity, in the range of 50%, for detecting T vaginalis.5-7 There are many factors that make microscopy a poor approach to the diagnosis of T vaginalis, including the rapid decrease in protozoan motility once a vaginal specimen is placed on a glass slide and the similar size of non-motile T vaginalis and other cells in the vagina.
Given the low sensitivity of microscopy for the diagnosis of trichomoniasis, the American College of Obstetricians and Gynecologists (ACOG) recommends NAAT as a primary approach to test for T vaginalis, with culture or NAAT testing as alternative approaches.8 The Centers for Disease Control and Prevention (CDC) recommends that if a wet mount is negative for T vaginalis that NAAT should be utilized.9
In this 2-step testing process, the first step is to test the vaginal pH and perform a microscopic examination of a vaginal specimen for T vaginalis. If T vaginalis organisms are detected, the diagnosis of trichomoniasis is confirmed. If organisms are not detected the second step would be to send a vaginal or urine specimen for NAAT for T vaginalis or for culture. An advantage of NAAT over culture is that urine specimens can be used for diagnosis of T vaginalis while urine specimens are not suitable for culture because of low sensitivity. For patients diagnosed with trichomoniasis, the CDC recommends that testing be repeated in 3 months because of high rates of reinfection. NAAT would be an optimal test to use in this situation.
Continue to: Bacterial vaginosis and candidiasis...
Bacterial vaginosis and candidiasis
ACOG recommends using Amsel criteria or Nugent scoring of a specimen colorized with a Gram stain for the diagnosis of bacterial vaginosis and microscopy or culture for the diagnosis of candidiasis.8 Recent research reports that NAAT testing for bacterial vaginosis and candidiasis may be more sensitive than standard office-based approaches for detecting these two causes of vaginitis. In a study of approximately 1,740 patients with symptoms of vaginitis, vaginal specimens were analyzed using NAAT or standard office approaches to diagnosis.10 In this study the diagnostic gold standards were Nugent scoring with Amsel criteria to resolve intermediate Nugent scores for bacterial vaginosis and culture for Candida. The study demonstrated the superiority of NAAT testing over standard office approaches for the identification of the cause of the vaginitis. NAAT testing was reported to have superior sensitivity for diagnosing bacterial vaginosis compared with the original Amsel criteria (93% vs 76%, respectively (P <.0001), with similar respective specificities of 92% and 94% .10 NAAT testing also had superior sensitivity for diagnosing Candidiasis compared with microscopy after potassium hydroxide treatment of a vaginal specimen (91% vs 58%, respectively (P <.0001).10 NAAT testing also had superior specificity compared with microscopy after potassium hydroxide treatment of a vaginal specimen (94% vs 89%, respectively (P < .0005).10
In another study comparing NAAT with clinical diagnosis for 466 patients with symptoms of vaginitis, standard office approaches to the diagnosis of vaginitis resulted in the failure to identify the correct infection in a large number of cases. For the diagnosis of bacterial vaginosis, clinicians missed 42% of the cases identified by NAAT. For the diagnosis of Candida, clinicians missed 46% of the cases identified by NAAT. For T vaginalis diagnosis, clinicians missed 72% of the cases identified by NAAT. Clearly, this resulted in clinicians not treating many infections detected by NAAT.11
Continue to: One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis...
One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis
In a recent study, 1,471 patients with a symptom of vaginitis (abnormal vaginal discharge, itching or irritation, or odor) self-collected a vaginal swab and had a vaginal swab collected by a clinician.12 The swabs were placed in buffer and the samples were tested by NAAT using the BD Max system (Franklin Lakes, New Jersey) for the presence of nucleic acid sequences of the microorganisms responsible for the most common causes of vaginitis. In this cohort, using the clinician collected vaginal swabs for NAAT, the investigators reported the following pattern of detection of nucleic acid sequences: 36.1%, bacterial vaginosis pattern; 16.2%, Candida spp.; 1.6%, T vaginalis; 0.7%, Candida glabrata; and 0.1%, Candida krusei. Nucleic acid sequences of multiple organisms were detected in 21.7% of patients, including 13.9% with bacterial vaginosis pattern plus Candida spp., 4.9% with bacterial vaginosis pattern plus T vaginalis, 0.3% with Candida spp. plus T vaginalis, 0.2% with Candida spp. plus Candida glabrata, 0.2% with bacterial vaginosis pattern plus Candida glabrata, and 2.2% with all 3 organisms. A total of 23.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.
In another study of 1,491 patients with a symptom of vaginitis, clinician-collected vaginal swabs were tested by NAAT using the Aptima BV and Aptima Candida/Trichomonas systems (Hologic, Marlborough, Massachusetts) for the presence of nucleic acid sequences of microorganisms responsible for most cases of vaginitis.13 The investigators reported the following pattern of detection of nucleic acid sequences: 28.6%, bacterial vaginosis pattern; 14.2%, Candida spp.; 3%, T vaginalis; 1.9%, Candida glabrata.13 Nucleic acid sequences from multiple organisms were detected in 23.3% of patients. Nucleic acid sequences suggesting the presence of two different causes of vaginitis were detected among 20.8% of patients, including bacterial vaginosis plus Candida spp., 11.1%; bacterial vaginosis plus T vaginalis, 7.2%; Candida spp. plus T vaginalis, 1.0%; Candida spp. plus Candida glabrata, 0.9%; bacterial vaginosis plus Candida spp., 0.5%; Candida glabrata plus T vaginalis, 0.1%. Nucleic acid sequences suggesting the presence of 3 different causes of vaginitis were detected in 2.4% of patients, the most common being the combination of bacterial vaginosis plus Candida spp. plus T vaginalis, 1.7% and bacterial vaginosis plus Candida spp. plus Candida glabrata, 0.5%. Nucleic acid sequences suggesting the presence of 4 different causes of vaginitis were detected in 0.1% of patients. A total of 28.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.13
In clinical practice it is uncommon to see the diagnosis of multiple causes of vaginitis recorded in the medical record of a patient. This suggests that we are not effectively identifying the 20% of patients with multiple causes of vaginitis.
When multiple organisms that cause vaginitis are present, NAAT is superior to clinical evaluation for diagnosis
In a study of 1,264 patients with symptoms of vaginitis who had an identified microbial cause, more than 20% had multiple organisms detected by NAAT
Patient collection of a vaginal swab for NAAT
Multiple studies have reported that collection of a vaginal swab for NAAT by the patient or a clinician results in similar excellent test performance.4,12,13 This observation might catalyze the development of clinical protocols where patients with vaginitis could collect the swab for NAAT analysis, without needing to have a speculum examination by a clinician.
When collecting a vaginal specimen for NAAT it is important that no vaginal lubricants or creams contaminate the collection swab. Vaginal lubricants and creams may inhibit the polymerase chain reaction enzymes resulting in a false negative. The swab may be directly inserted into the vagina to collect the specimen or a speculum without a lubricant, except water can be used to facilitate specimen collection. To collect a specimen without a speculum the swab is inserted 2 inches into the vagina and rotated for 10 to 15 seconds.
What should clinicians do while waiting for a NAAT result?
A major problem with NAAT testing for vaginitis is that the results are not available at the initial patient visit, impacting the ability to make an immediate diagnosis and provide targeted antibiotic treatment. Given that bacterial vaginosis and Candida species are the most common causes of infectious vaginitis in many populations of gynecology patients, one approach is to initiate treatment with one dose of an oral antifungal agent and a multiday course of vaginal metronidazole. Once the NAAT test results are available, the treatment can be refined to specific infectious agents identified by the test, or the antibiotics can be discontinued if no relevant microorganisms are detected. Another approach would be to wait until the NAAT test is completed and then prescribe the appropriate antibiotic. My sense is that most patients would not favor this wait and see approach.
Barriers to the use of NAAT for vaginitis
A barrier to the use of NAAT for the diagnosis of vaginitis is that leading organizations do not currently recommend NAAT as a primary approach to diagnosis, favoring microscopy and measurement of vaginal pH.9 In addition, clinicians and patients may be rightfully concerned about the cost of NAAT, which can be substantial.
Vaginitis, especially when it is recurrent, can be stressful14 and have an impact on a patient’s quality of life15,16 and sexual health.17 Arguably, our current practice algorithms for diagnosing the cause of vaginitis are not optimized.18 Our failure to accurately diagnose the cause of vaginitis contributes to inappropriate antibiotic treatment and return visits because of inadequate initial treatment.18 We can improve and simplify our approach to the diagnosis of vaginitis by prioritizing the use of NAAT.19 In turn, reliably making the right diagnosis will result in the optimization of treatment. ●
- Jose PP, Vivekanandan V, Sobhanakumari K. Gonorrhea: Historical outlook. J Skin Sex Transm Dis. 2020;2:110-114.
- Bayly HW. The diagnosis and treatment of chronic gonorrhoea and its local complications. Br Med J. 1914;14:584-587.
- Stuart RD. The diagnosis and control of gonorrhoea by bacteriological cultures: with a preliminary report on a new method for transporting clinical material. Glasgow Med J. 1946;27:131-142.
- Wilson JD, Wallace HE, Loftus-Keeling M, et al. Swab-yourself trial with economic monitoring and testing for infections collectively (SYSTEMATIC): Part 2. A diagnostic accuracy and cost-effectiveness study comparing rectal, pharyngeal and urogenital samples analyzed individually, versus as a pooled specimen, for the diagnosis of gonorrhea and chlamydia. Clin Infect Dis. 2021;73:e3183-3193.
- Hollman D, Coupey SM, Fox AS, et al. Screening for Trichomonas vaginalis in high-risk adolescent females with a new NAAT: association with ethnicity, symptoms and prior and current STIs. J Pediatr Adolesc Gynecol. 2010;23:312-316.
- Roth AM, Williams JA, Ly R. et al. Changing sexually transmitted infection screening protocol will result in improved case finding for Trichomonas vaginalis among high-risk female populations. Sex Transm Dis. 2011;38:398-400.
- Hobbs MM, Sena AC. Modern diagnosis of Trichomonas vaginalis infection. Sex Transm Infection. 2013;89:434-438.
- Vaginitis in nonpregnant patients. ACOG Practice Bulletin No 215. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2020;135:e1-e17.
- Workowksi KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines 2021. MMWR. 2021;70:1-187.
- Schwebke JR, Gaydos CA, Hyirjesy P, et al. Diagnostic performance of a molecular test versus clinician assessment of vaginitis. J Clin Microbiol. 2018;56:e00252-18.
- Broache M, Cammarata CL, Stonebraker E, et al. Performance of vaginal panel assay compared with clinical diagnosis of vaginitis. Obstet Gynecol. 2021;138:853-859.
- Gaydos CA, Beqaj S, Schwebke JR, et al. Clinical validation of a test for the diagnosis of vaginitis. Obstet Gynecol. 2017;130:181-189.
- Schwebke JR, Taylor SN, Ackerman N, et al. Clinical validation of the Aptima bacterial vaginosis and Aptima Candida/Trichomonas vaginalis assays: results from a prospective multi-center study. J Clin Microbiol. 2020;58:e01643-19.
- Ehrstrom S, Kornfeld D, Rylander E. Perceived stress in women with recurrent vulvovaginal candidiasis. J Psychosomatic Obstet Gynecol. 2007;28:169-176.
- Abellea S, Guelfucci F, Wagner J, et al. Subjective health status and health-related quality of life among women with recurrent vulvovaginal candidosis in Europe and the USA. Health Quality Life Outcomes. 2013;11:169.
- Fukazawa EI, Witkin SS, Robial R, et al. Influence of recurrent vulvovaginal candidiasis on quality of life issues. Arch Gynecol Obstet. 2019;300:647-650.
- Giraldo PC, Polpeta NC, Juliato CT, et al. Evaluation of sexual function in Brazilian women with recurrent vulvovaginal candidiasis and localized provoked vulvodynia. J Sex Med. 2012;9:805-811.
- Hillier SL, Austin M, Macio I, et al. Diagnosis and treatment of vaginal discharge syndromes in community practice settings. Clin Infect Dis. 2021;72:1538-1543.
- . Sobel JD. Syndromic treatment of women with vulvovaginal symptoms in the United States: a call to action. Clin Infect Dis. 2021;72:1544-1545.
Over the past 200 years, identification of the specific organism causing an infection has evolved from a reliance on patient history and physical examination to the use of microscopic examination of relevant biological samples to the rise of microbial culture and immunological testing as the gold standards for diagnosis. More recently, advances in nucleic acid testing have made nucleic acid amplification testing (NAAT) a primary method for identifying the specific organism causing an infection.
The evolution of the diagnosis of gonorrhea in clinical practice is a good example of the inexorable evolution of diagnostic techniques from physical examination to microscopic analysis to culture and finally to NAAT. Neiseer discovered Neisseria gonorrhea in 1879.1 In 19th century general medical practice gonorrhea was often diagnosed based on history and physical examination and sometimes microscopy was also utilized.2 In the mid-20th century, it was realized that culture was a superior approach to diagnosis of gonorrhea, and it became the gold standard for diagnosis in general practice.3 NAAT has now replaced culture as the gold standard for the diagnosis of gonorrhea because of its superior performance in clinical practice.4 It may now be time to consider using NAAT rather than microscopy and culture in general practice for the identification of specific microorganisms causing vaginitis.
Trichomoniasis
Vaginitis caused by Trichomonas vaginalis is characterized by a discharge that is foamy and green-yellow in color, with a vaginal pH that is >4.5. Microscopy of a vaginal specimen has low sensitivity, in the range of 50%, for detecting T vaginalis.5-7 There are many factors that make microscopy a poor approach to the diagnosis of T vaginalis, including the rapid decrease in protozoan motility once a vaginal specimen is placed on a glass slide and the similar size of non-motile T vaginalis and other cells in the vagina.
Given the low sensitivity of microscopy for the diagnosis of trichomoniasis, the American College of Obstetricians and Gynecologists (ACOG) recommends NAAT as a primary approach to test for T vaginalis, with culture or NAAT testing as alternative approaches.8 The Centers for Disease Control and Prevention (CDC) recommends that if a wet mount is negative for T vaginalis that NAAT should be utilized.9
In this 2-step testing process, the first step is to test the vaginal pH and perform a microscopic examination of a vaginal specimen for T vaginalis. If T vaginalis organisms are detected, the diagnosis of trichomoniasis is confirmed. If organisms are not detected the second step would be to send a vaginal or urine specimen for NAAT for T vaginalis or for culture. An advantage of NAAT over culture is that urine specimens can be used for diagnosis of T vaginalis while urine specimens are not suitable for culture because of low sensitivity. For patients diagnosed with trichomoniasis, the CDC recommends that testing be repeated in 3 months because of high rates of reinfection. NAAT would be an optimal test to use in this situation.
Continue to: Bacterial vaginosis and candidiasis...
Bacterial vaginosis and candidiasis
ACOG recommends using Amsel criteria or Nugent scoring of a specimen colorized with a Gram stain for the diagnosis of bacterial vaginosis and microscopy or culture for the diagnosis of candidiasis.8 Recent research reports that NAAT testing for bacterial vaginosis and candidiasis may be more sensitive than standard office-based approaches for detecting these two causes of vaginitis. In a study of approximately 1,740 patients with symptoms of vaginitis, vaginal specimens were analyzed using NAAT or standard office approaches to diagnosis.10 In this study the diagnostic gold standards were Nugent scoring with Amsel criteria to resolve intermediate Nugent scores for bacterial vaginosis and culture for Candida. The study demonstrated the superiority of NAAT testing over standard office approaches for the identification of the cause of the vaginitis. NAAT testing was reported to have superior sensitivity for diagnosing bacterial vaginosis compared with the original Amsel criteria (93% vs 76%, respectively (P <.0001), with similar respective specificities of 92% and 94% .10 NAAT testing also had superior sensitivity for diagnosing Candidiasis compared with microscopy after potassium hydroxide treatment of a vaginal specimen (91% vs 58%, respectively (P <.0001).10 NAAT testing also had superior specificity compared with microscopy after potassium hydroxide treatment of a vaginal specimen (94% vs 89%, respectively (P < .0005).10
In another study comparing NAAT with clinical diagnosis for 466 patients with symptoms of vaginitis, standard office approaches to the diagnosis of vaginitis resulted in the failure to identify the correct infection in a large number of cases. For the diagnosis of bacterial vaginosis, clinicians missed 42% of the cases identified by NAAT. For the diagnosis of Candida, clinicians missed 46% of the cases identified by NAAT. For T vaginalis diagnosis, clinicians missed 72% of the cases identified by NAAT. Clearly, this resulted in clinicians not treating many infections detected by NAAT.11
Continue to: One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis...
One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis
In a recent study, 1,471 patients with a symptom of vaginitis (abnormal vaginal discharge, itching or irritation, or odor) self-collected a vaginal swab and had a vaginal swab collected by a clinician.12 The swabs were placed in buffer and the samples were tested by NAAT using the BD Max system (Franklin Lakes, New Jersey) for the presence of nucleic acid sequences of the microorganisms responsible for the most common causes of vaginitis. In this cohort, using the clinician collected vaginal swabs for NAAT, the investigators reported the following pattern of detection of nucleic acid sequences: 36.1%, bacterial vaginosis pattern; 16.2%, Candida spp.; 1.6%, T vaginalis; 0.7%, Candida glabrata; and 0.1%, Candida krusei. Nucleic acid sequences of multiple organisms were detected in 21.7% of patients, including 13.9% with bacterial vaginosis pattern plus Candida spp., 4.9% with bacterial vaginosis pattern plus T vaginalis, 0.3% with Candida spp. plus T vaginalis, 0.2% with Candida spp. plus Candida glabrata, 0.2% with bacterial vaginosis pattern plus Candida glabrata, and 2.2% with all 3 organisms. A total of 23.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.
In another study of 1,491 patients with a symptom of vaginitis, clinician-collected vaginal swabs were tested by NAAT using the Aptima BV and Aptima Candida/Trichomonas systems (Hologic, Marlborough, Massachusetts) for the presence of nucleic acid sequences of microorganisms responsible for most cases of vaginitis.13 The investigators reported the following pattern of detection of nucleic acid sequences: 28.6%, bacterial vaginosis pattern; 14.2%, Candida spp.; 3%, T vaginalis; 1.9%, Candida glabrata.13 Nucleic acid sequences from multiple organisms were detected in 23.3% of patients. Nucleic acid sequences suggesting the presence of two different causes of vaginitis were detected among 20.8% of patients, including bacterial vaginosis plus Candida spp., 11.1%; bacterial vaginosis plus T vaginalis, 7.2%; Candida spp. plus T vaginalis, 1.0%; Candida spp. plus Candida glabrata, 0.9%; bacterial vaginosis plus Candida spp., 0.5%; Candida glabrata plus T vaginalis, 0.1%. Nucleic acid sequences suggesting the presence of 3 different causes of vaginitis were detected in 2.4% of patients, the most common being the combination of bacterial vaginosis plus Candida spp. plus T vaginalis, 1.7% and bacterial vaginosis plus Candida spp. plus Candida glabrata, 0.5%. Nucleic acid sequences suggesting the presence of 4 different causes of vaginitis were detected in 0.1% of patients. A total of 28.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.13
In clinical practice it is uncommon to see the diagnosis of multiple causes of vaginitis recorded in the medical record of a patient. This suggests that we are not effectively identifying the 20% of patients with multiple causes of vaginitis.
When multiple organisms that cause vaginitis are present, NAAT is superior to clinical evaluation for diagnosis
In a study of 1,264 patients with symptoms of vaginitis who had an identified microbial cause, more than 20% had multiple organisms detected by NAAT
Patient collection of a vaginal swab for NAAT
Multiple studies have reported that collection of a vaginal swab for NAAT by the patient or a clinician results in similar excellent test performance.4,12,13 This observation might catalyze the development of clinical protocols where patients with vaginitis could collect the swab for NAAT analysis, without needing to have a speculum examination by a clinician.
When collecting a vaginal specimen for NAAT it is important that no vaginal lubricants or creams contaminate the collection swab. Vaginal lubricants and creams may inhibit the polymerase chain reaction enzymes resulting in a false negative. The swab may be directly inserted into the vagina to collect the specimen or a speculum without a lubricant, except water can be used to facilitate specimen collection. To collect a specimen without a speculum the swab is inserted 2 inches into the vagina and rotated for 10 to 15 seconds.
What should clinicians do while waiting for a NAAT result?
A major problem with NAAT testing for vaginitis is that the results are not available at the initial patient visit, impacting the ability to make an immediate diagnosis and provide targeted antibiotic treatment. Given that bacterial vaginosis and Candida species are the most common causes of infectious vaginitis in many populations of gynecology patients, one approach is to initiate treatment with one dose of an oral antifungal agent and a multiday course of vaginal metronidazole. Once the NAAT test results are available, the treatment can be refined to specific infectious agents identified by the test, or the antibiotics can be discontinued if no relevant microorganisms are detected. Another approach would be to wait until the NAAT test is completed and then prescribe the appropriate antibiotic. My sense is that most patients would not favor this wait and see approach.
Barriers to the use of NAAT for vaginitis
A barrier to the use of NAAT for the diagnosis of vaginitis is that leading organizations do not currently recommend NAAT as a primary approach to diagnosis, favoring microscopy and measurement of vaginal pH.9 In addition, clinicians and patients may be rightfully concerned about the cost of NAAT, which can be substantial.
Vaginitis, especially when it is recurrent, can be stressful14 and have an impact on a patient’s quality of life15,16 and sexual health.17 Arguably, our current practice algorithms for diagnosing the cause of vaginitis are not optimized.18 Our failure to accurately diagnose the cause of vaginitis contributes to inappropriate antibiotic treatment and return visits because of inadequate initial treatment.18 We can improve and simplify our approach to the diagnosis of vaginitis by prioritizing the use of NAAT.19 In turn, reliably making the right diagnosis will result in the optimization of treatment. ●
Over the past 200 years, identification of the specific organism causing an infection has evolved from a reliance on patient history and physical examination to the use of microscopic examination of relevant biological samples to the rise of microbial culture and immunological testing as the gold standards for diagnosis. More recently, advances in nucleic acid testing have made nucleic acid amplification testing (NAAT) a primary method for identifying the specific organism causing an infection.
The evolution of the diagnosis of gonorrhea in clinical practice is a good example of the inexorable evolution of diagnostic techniques from physical examination to microscopic analysis to culture and finally to NAAT. Neiseer discovered Neisseria gonorrhea in 1879.1 In 19th century general medical practice gonorrhea was often diagnosed based on history and physical examination and sometimes microscopy was also utilized.2 In the mid-20th century, it was realized that culture was a superior approach to diagnosis of gonorrhea, and it became the gold standard for diagnosis in general practice.3 NAAT has now replaced culture as the gold standard for the diagnosis of gonorrhea because of its superior performance in clinical practice.4 It may now be time to consider using NAAT rather than microscopy and culture in general practice for the identification of specific microorganisms causing vaginitis.
Trichomoniasis
Vaginitis caused by Trichomonas vaginalis is characterized by a discharge that is foamy and green-yellow in color, with a vaginal pH that is >4.5. Microscopy of a vaginal specimen has low sensitivity, in the range of 50%, for detecting T vaginalis.5-7 There are many factors that make microscopy a poor approach to the diagnosis of T vaginalis, including the rapid decrease in protozoan motility once a vaginal specimen is placed on a glass slide and the similar size of non-motile T vaginalis and other cells in the vagina.
Given the low sensitivity of microscopy for the diagnosis of trichomoniasis, the American College of Obstetricians and Gynecologists (ACOG) recommends NAAT as a primary approach to test for T vaginalis, with culture or NAAT testing as alternative approaches.8 The Centers for Disease Control and Prevention (CDC) recommends that if a wet mount is negative for T vaginalis that NAAT should be utilized.9
In this 2-step testing process, the first step is to test the vaginal pH and perform a microscopic examination of a vaginal specimen for T vaginalis. If T vaginalis organisms are detected, the diagnosis of trichomoniasis is confirmed. If organisms are not detected the second step would be to send a vaginal or urine specimen for NAAT for T vaginalis or for culture. An advantage of NAAT over culture is that urine specimens can be used for diagnosis of T vaginalis while urine specimens are not suitable for culture because of low sensitivity. For patients diagnosed with trichomoniasis, the CDC recommends that testing be repeated in 3 months because of high rates of reinfection. NAAT would be an optimal test to use in this situation.
Continue to: Bacterial vaginosis and candidiasis...
Bacterial vaginosis and candidiasis
ACOG recommends using Amsel criteria or Nugent scoring of a specimen colorized with a Gram stain for the diagnosis of bacterial vaginosis and microscopy or culture for the diagnosis of candidiasis.8 Recent research reports that NAAT testing for bacterial vaginosis and candidiasis may be more sensitive than standard office-based approaches for detecting these two causes of vaginitis. In a study of approximately 1,740 patients with symptoms of vaginitis, vaginal specimens were analyzed using NAAT or standard office approaches to diagnosis.10 In this study the diagnostic gold standards were Nugent scoring with Amsel criteria to resolve intermediate Nugent scores for bacterial vaginosis and culture for Candida. The study demonstrated the superiority of NAAT testing over standard office approaches for the identification of the cause of the vaginitis. NAAT testing was reported to have superior sensitivity for diagnosing bacterial vaginosis compared with the original Amsel criteria (93% vs 76%, respectively (P <.0001), with similar respective specificities of 92% and 94% .10 NAAT testing also had superior sensitivity for diagnosing Candidiasis compared with microscopy after potassium hydroxide treatment of a vaginal specimen (91% vs 58%, respectively (P <.0001).10 NAAT testing also had superior specificity compared with microscopy after potassium hydroxide treatment of a vaginal specimen (94% vs 89%, respectively (P < .0005).10
In another study comparing NAAT with clinical diagnosis for 466 patients with symptoms of vaginitis, standard office approaches to the diagnosis of vaginitis resulted in the failure to identify the correct infection in a large number of cases. For the diagnosis of bacterial vaginosis, clinicians missed 42% of the cases identified by NAAT. For the diagnosis of Candida, clinicians missed 46% of the cases identified by NAAT. For T vaginalis diagnosis, clinicians missed 72% of the cases identified by NAAT. Clearly, this resulted in clinicians not treating many infections detected by NAAT.11
Continue to: One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis...
One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis
In a recent study, 1,471 patients with a symptom of vaginitis (abnormal vaginal discharge, itching or irritation, or odor) self-collected a vaginal swab and had a vaginal swab collected by a clinician.12 The swabs were placed in buffer and the samples were tested by NAAT using the BD Max system (Franklin Lakes, New Jersey) for the presence of nucleic acid sequences of the microorganisms responsible for the most common causes of vaginitis. In this cohort, using the clinician collected vaginal swabs for NAAT, the investigators reported the following pattern of detection of nucleic acid sequences: 36.1%, bacterial vaginosis pattern; 16.2%, Candida spp.; 1.6%, T vaginalis; 0.7%, Candida glabrata; and 0.1%, Candida krusei. Nucleic acid sequences of multiple organisms were detected in 21.7% of patients, including 13.9% with bacterial vaginosis pattern plus Candida spp., 4.9% with bacterial vaginosis pattern plus T vaginalis, 0.3% with Candida spp. plus T vaginalis, 0.2% with Candida spp. plus Candida glabrata, 0.2% with bacterial vaginosis pattern plus Candida glabrata, and 2.2% with all 3 organisms. A total of 23.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.
In another study of 1,491 patients with a symptom of vaginitis, clinician-collected vaginal swabs were tested by NAAT using the Aptima BV and Aptima Candida/Trichomonas systems (Hologic, Marlborough, Massachusetts) for the presence of nucleic acid sequences of microorganisms responsible for most cases of vaginitis.13 The investigators reported the following pattern of detection of nucleic acid sequences: 28.6%, bacterial vaginosis pattern; 14.2%, Candida spp.; 3%, T vaginalis; 1.9%, Candida glabrata.13 Nucleic acid sequences from multiple organisms were detected in 23.3% of patients. Nucleic acid sequences suggesting the presence of two different causes of vaginitis were detected among 20.8% of patients, including bacterial vaginosis plus Candida spp., 11.1%; bacterial vaginosis plus T vaginalis, 7.2%; Candida spp. plus T vaginalis, 1.0%; Candida spp. plus Candida glabrata, 0.9%; bacterial vaginosis plus Candida spp., 0.5%; Candida glabrata plus T vaginalis, 0.1%. Nucleic acid sequences suggesting the presence of 3 different causes of vaginitis were detected in 2.4% of patients, the most common being the combination of bacterial vaginosis plus Candida spp. plus T vaginalis, 1.7% and bacterial vaginosis plus Candida spp. plus Candida glabrata, 0.5%. Nucleic acid sequences suggesting the presence of 4 different causes of vaginitis were detected in 0.1% of patients. A total of 28.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.13
In clinical practice it is uncommon to see the diagnosis of multiple causes of vaginitis recorded in the medical record of a patient. This suggests that we are not effectively identifying the 20% of patients with multiple causes of vaginitis.
When multiple organisms that cause vaginitis are present, NAAT is superior to clinical evaluation for diagnosis
In a study of 1,264 patients with symptoms of vaginitis who had an identified microbial cause, more than 20% had multiple organisms detected by NAAT
Patient collection of a vaginal swab for NAAT
Multiple studies have reported that collection of a vaginal swab for NAAT by the patient or a clinician results in similar excellent test performance.4,12,13 This observation might catalyze the development of clinical protocols where patients with vaginitis could collect the swab for NAAT analysis, without needing to have a speculum examination by a clinician.
When collecting a vaginal specimen for NAAT it is important that no vaginal lubricants or creams contaminate the collection swab. Vaginal lubricants and creams may inhibit the polymerase chain reaction enzymes resulting in a false negative. The swab may be directly inserted into the vagina to collect the specimen or a speculum without a lubricant, except water can be used to facilitate specimen collection. To collect a specimen without a speculum the swab is inserted 2 inches into the vagina and rotated for 10 to 15 seconds.
What should clinicians do while waiting for a NAAT result?
A major problem with NAAT testing for vaginitis is that the results are not available at the initial patient visit, impacting the ability to make an immediate diagnosis and provide targeted antibiotic treatment. Given that bacterial vaginosis and Candida species are the most common causes of infectious vaginitis in many populations of gynecology patients, one approach is to initiate treatment with one dose of an oral antifungal agent and a multiday course of vaginal metronidazole. Once the NAAT test results are available, the treatment can be refined to specific infectious agents identified by the test, or the antibiotics can be discontinued if no relevant microorganisms are detected. Another approach would be to wait until the NAAT test is completed and then prescribe the appropriate antibiotic. My sense is that most patients would not favor this wait and see approach.
Barriers to the use of NAAT for vaginitis
A barrier to the use of NAAT for the diagnosis of vaginitis is that leading organizations do not currently recommend NAAT as a primary approach to diagnosis, favoring microscopy and measurement of vaginal pH.9 In addition, clinicians and patients may be rightfully concerned about the cost of NAAT, which can be substantial.
Vaginitis, especially when it is recurrent, can be stressful14 and have an impact on a patient’s quality of life15,16 and sexual health.17 Arguably, our current practice algorithms for diagnosing the cause of vaginitis are not optimized.18 Our failure to accurately diagnose the cause of vaginitis contributes to inappropriate antibiotic treatment and return visits because of inadequate initial treatment.18 We can improve and simplify our approach to the diagnosis of vaginitis by prioritizing the use of NAAT.19 In turn, reliably making the right diagnosis will result in the optimization of treatment. ●
- Jose PP, Vivekanandan V, Sobhanakumari K. Gonorrhea: Historical outlook. J Skin Sex Transm Dis. 2020;2:110-114.
- Bayly HW. The diagnosis and treatment of chronic gonorrhoea and its local complications. Br Med J. 1914;14:584-587.
- Stuart RD. The diagnosis and control of gonorrhoea by bacteriological cultures: with a preliminary report on a new method for transporting clinical material. Glasgow Med J. 1946;27:131-142.
- Wilson JD, Wallace HE, Loftus-Keeling M, et al. Swab-yourself trial with economic monitoring and testing for infections collectively (SYSTEMATIC): Part 2. A diagnostic accuracy and cost-effectiveness study comparing rectal, pharyngeal and urogenital samples analyzed individually, versus as a pooled specimen, for the diagnosis of gonorrhea and chlamydia. Clin Infect Dis. 2021;73:e3183-3193.
- Hollman D, Coupey SM, Fox AS, et al. Screening for Trichomonas vaginalis in high-risk adolescent females with a new NAAT: association with ethnicity, symptoms and prior and current STIs. J Pediatr Adolesc Gynecol. 2010;23:312-316.
- Roth AM, Williams JA, Ly R. et al. Changing sexually transmitted infection screening protocol will result in improved case finding for Trichomonas vaginalis among high-risk female populations. Sex Transm Dis. 2011;38:398-400.
- Hobbs MM, Sena AC. Modern diagnosis of Trichomonas vaginalis infection. Sex Transm Infection. 2013;89:434-438.
- Vaginitis in nonpregnant patients. ACOG Practice Bulletin No 215. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2020;135:e1-e17.
- Workowksi KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines 2021. MMWR. 2021;70:1-187.
- Schwebke JR, Gaydos CA, Hyirjesy P, et al. Diagnostic performance of a molecular test versus clinician assessment of vaginitis. J Clin Microbiol. 2018;56:e00252-18.
- Broache M, Cammarata CL, Stonebraker E, et al. Performance of vaginal panel assay compared with clinical diagnosis of vaginitis. Obstet Gynecol. 2021;138:853-859.
- Gaydos CA, Beqaj S, Schwebke JR, et al. Clinical validation of a test for the diagnosis of vaginitis. Obstet Gynecol. 2017;130:181-189.
- Schwebke JR, Taylor SN, Ackerman N, et al. Clinical validation of the Aptima bacterial vaginosis and Aptima Candida/Trichomonas vaginalis assays: results from a prospective multi-center study. J Clin Microbiol. 2020;58:e01643-19.
- Ehrstrom S, Kornfeld D, Rylander E. Perceived stress in women with recurrent vulvovaginal candidiasis. J Psychosomatic Obstet Gynecol. 2007;28:169-176.
- Abellea S, Guelfucci F, Wagner J, et al. Subjective health status and health-related quality of life among women with recurrent vulvovaginal candidosis in Europe and the USA. Health Quality Life Outcomes. 2013;11:169.
- Fukazawa EI, Witkin SS, Robial R, et al. Influence of recurrent vulvovaginal candidiasis on quality of life issues. Arch Gynecol Obstet. 2019;300:647-650.
- Giraldo PC, Polpeta NC, Juliato CT, et al. Evaluation of sexual function in Brazilian women with recurrent vulvovaginal candidiasis and localized provoked vulvodynia. J Sex Med. 2012;9:805-811.
- Hillier SL, Austin M, Macio I, et al. Diagnosis and treatment of vaginal discharge syndromes in community practice settings. Clin Infect Dis. 2021;72:1538-1543.
- . Sobel JD. Syndromic treatment of women with vulvovaginal symptoms in the United States: a call to action. Clin Infect Dis. 2021;72:1544-1545.
- Jose PP, Vivekanandan V, Sobhanakumari K. Gonorrhea: Historical outlook. J Skin Sex Transm Dis. 2020;2:110-114.
- Bayly HW. The diagnosis and treatment of chronic gonorrhoea and its local complications. Br Med J. 1914;14:584-587.
- Stuart RD. The diagnosis and control of gonorrhoea by bacteriological cultures: with a preliminary report on a new method for transporting clinical material. Glasgow Med J. 1946;27:131-142.
- Wilson JD, Wallace HE, Loftus-Keeling M, et al. Swab-yourself trial with economic monitoring and testing for infections collectively (SYSTEMATIC): Part 2. A diagnostic accuracy and cost-effectiveness study comparing rectal, pharyngeal and urogenital samples analyzed individually, versus as a pooled specimen, for the diagnosis of gonorrhea and chlamydia. Clin Infect Dis. 2021;73:e3183-3193.
- Hollman D, Coupey SM, Fox AS, et al. Screening for Trichomonas vaginalis in high-risk adolescent females with a new NAAT: association with ethnicity, symptoms and prior and current STIs. J Pediatr Adolesc Gynecol. 2010;23:312-316.
- Roth AM, Williams JA, Ly R. et al. Changing sexually transmitted infection screening protocol will result in improved case finding for Trichomonas vaginalis among high-risk female populations. Sex Transm Dis. 2011;38:398-400.
- Hobbs MM, Sena AC. Modern diagnosis of Trichomonas vaginalis infection. Sex Transm Infection. 2013;89:434-438.
- Vaginitis in nonpregnant patients. ACOG Practice Bulletin No 215. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2020;135:e1-e17.
- Workowksi KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines 2021. MMWR. 2021;70:1-187.
- Schwebke JR, Gaydos CA, Hyirjesy P, et al. Diagnostic performance of a molecular test versus clinician assessment of vaginitis. J Clin Microbiol. 2018;56:e00252-18.
- Broache M, Cammarata CL, Stonebraker E, et al. Performance of vaginal panel assay compared with clinical diagnosis of vaginitis. Obstet Gynecol. 2021;138:853-859.
- Gaydos CA, Beqaj S, Schwebke JR, et al. Clinical validation of a test for the diagnosis of vaginitis. Obstet Gynecol. 2017;130:181-189.
- Schwebke JR, Taylor SN, Ackerman N, et al. Clinical validation of the Aptima bacterial vaginosis and Aptima Candida/Trichomonas vaginalis assays: results from a prospective multi-center study. J Clin Microbiol. 2020;58:e01643-19.
- Ehrstrom S, Kornfeld D, Rylander E. Perceived stress in women with recurrent vulvovaginal candidiasis. J Psychosomatic Obstet Gynecol. 2007;28:169-176.
- Abellea S, Guelfucci F, Wagner J, et al. Subjective health status and health-related quality of life among women with recurrent vulvovaginal candidosis in Europe and the USA. Health Quality Life Outcomes. 2013;11:169.
- Fukazawa EI, Witkin SS, Robial R, et al. Influence of recurrent vulvovaginal candidiasis on quality of life issues. Arch Gynecol Obstet. 2019;300:647-650.
- Giraldo PC, Polpeta NC, Juliato CT, et al. Evaluation of sexual function in Brazilian women with recurrent vulvovaginal candidiasis and localized provoked vulvodynia. J Sex Med. 2012;9:805-811.
- Hillier SL, Austin M, Macio I, et al. Diagnosis and treatment of vaginal discharge syndromes in community practice settings. Clin Infect Dis. 2021;72:1538-1543.
- . Sobel JD. Syndromic treatment of women with vulvovaginal symptoms in the United States: a call to action. Clin Infect Dis. 2021;72:1544-1545.
Appropriate cancer screening for women with dense breasts
We have been interested in the quiz series focused on breast cancer screening for women with dense breasts presented in
The concerns with breast cancer in particular
Breast cancer is not cervical cancer. It isn’t one disease. It is a multitude of diseases that happen to show up in the breast. Some are relatively slow-growing—the kinds of cancers that lend themselves to screening and to early intervention. But other cancers are rapidly-growing; they show up no matter how often or what modality we use for screening. Our goal should be to find an approach to screening that can diagnose breast cancer at a stage where we can intervene and positively impact breast cancer specific and overall mortality.
Screening guidelines vary
The variety of screening guidelines published by different professional organizations reflect differing assumptions and sets of values related to the early diagnosis and treatment of breast cancer. (For a comprehensive table of current screening guidelines, see https://www.cdc.gov/cancer/breast/pdf/breast-cancer-screening-guidelines-508.pdf.)
ACOG’s approach—to offer screening at age 40 but to begin by at least age 50 and, through shared decision making with the patient, screen every 1 or 2 years—is focused on capturing as many cases as we can identify, while minimizing the harms of false-positives.1 The perspective of the US Preventive Services Task Force (USPSTF) recommendations (to screen every 2 years beginning at age 50) is at the population level, a cost-effective approach that will have the greatest benefit while minimizing harms in the population at large.2 The American Society of Breast Surgeons recommends screening to begin by age 40.3 Like the breast surgeons, radiologists dedicated to breast imaging are focused on an individual rather than a population level. They strive to identify each and every instance of possible cancer, and therefore recommend annual screening beginning at age 40.4 However, with more aggressive screening in average-risk women many cases of ductal carcinoma in situ (DCIS) are identified—a lesion that, if not detected, may not impact the woman’s health during her lifetime—representing what some might call “overdiagnosis.” Yet there may be some instances in which the DCIS might affect an individual woman’s health. Unfortunately, we can’t prospectively distinguish between the first and the second types of cases.
We follow American College of Obstetricians and Gynecologists and US Preventive Services Task Force guidelines in discussing screening (both its hazards and benefits) with our average-risk patients beginning at age 40. We talk about risk factors for breast cancer, including breast density, but let patients know that no specific additional imaging is advised, and that density is more common in younger women (one consideration in earlier screening) and is quite common in general. Although we do not send follow-up letters to patients with dense breasts, we do educate our staff so that they can respond appropriately should patients call with questions.
Of course, we all bring to the table values that will impact the decisions that we make for ourselves and for our patients. What an ObGyn might suggest may differ from what a radiologist might suggest. Although we follow recommendations made by the radiologist at screening, an ObGyn wants to take care of the whole human being. We are concerned with bones, heart, everything about the patient, so we approach a patient in a different way. These priorities are reflected in the current varying breast cancer screening guidelines.
Continue to: Research on breast cancer screening varies by design...
Research on breast cancer screening varies by design
There has not been a randomized clinical trial conducted on screening mammography since the days of the analog mammogram. The research that has been conducted is difficult to compare due to variations in screening ages and intervals, technology sensitivity, and patient adherence with recommended screening. Treatments for breast cancer also have changed dramatically over time, so the findings of older studies may no longer be relevant to current breast cancer screening. The kind of analysis that needs to be done is an interrupted time series, where you can look at the trajectory of breast cancer survival and whether screening mammography shifts that survival in any way.
One specific study from Australia measured the impact of newer available breast cancer treatments, including tamoxifen for women with receptor-positive tumors and newer chemotherapy strategies.5 The authors analyzed screening mammography trends in one large province where women aged 50 to 69 were offered biennial screening. Trends from the 1990s showed that more women were being screened over time. Simultaneously, however, advances in therapy were entering clinical practice. The researchers pointed to a substantial decline in mortality from breast cancer from the early 1980s until 2013. But their conclusion was that none of the decline in mortality for breast cancer could be attributed to screening mammography when they looked at time trends; from their perspective all of the important decline in breast cancer mortality resulted from better treatment. They concluded that government programs should not support screening mammography.5
That is a recommendation that we do not support. However, we do recognize the conundrum that mammography is less sensitive among those who have dense breasts. In order to have congruent professional guidelines, we support research funding to determine which types, starting ages, and intervals of screening would be best in various patient populations. The USPSTF cites data from studies performed in the 1980s based on outdated technology; more recent (and relevant) randomized clinical trials have not been performed, and yet this information is critical to provide sufficient evidence to develop appropriate guidelines.
Our recommendations for gathering new data
The kind of data we would find most valuable would assess how different screening strategies impact overall mortality and breast cancer-specific mortality. It would require decades of follow-up—which of course means that screening technology will change over that time. A surrogate for evaluating overall survival is to look at interval cancers, which are all breast cancers diagnosed following negative mammograms and prior to the next screening. These cancers may or may not be biologically active, again focusing us on the need to look at overall survival of the patient. In addition, reducing breast cancer mortality may not reduce overall mortality, because the treatment for breast cancer may cause heart disease, or osteoporosis, or something else that impacts overall survival. These are important considerations for women and physicians who are making choices on treatment. What matters to a patient are 2 overlapping questions:
- Do I have a life-threatening condition or do I not?
- Has screening identified a condition that might lead to treatment that’s unnecessary?
The problem is that with breast cancer we can’t tell the difference. We do not understand the biological potential of a lesion when we evaluate an image on MRI, or computed tomography (CT), or mammography.
A re-look at presented data
A trial conducted by Bakker and colleagues6 was discussed by the authors of the DenseBreast-info.org quiz in which they recommended breast MRI for all women with extremely dense breasts (but no other risk factors for breast cancer) detected on screening mammograms.7 The Bakker study was large and conducted in the Netherlands. The primary outcome of the trial was to compare the incidence of interval breast cancers of women aged 50 to 75 randomly assigned to MRI versus those assigned to continued screening mammography every 2 years. Importantly, among the more than 8,000 women who were assigned to MRI, 59%, or fewer than two-thirds, chose to actually undergo MRI.
Among women randomized to MRI, 20 interval cancers were found—4 were diagnosed in those who actually had MRIs, and 16 were diagnosed among women who were randomized to MRI but didn’t undergo the study. Among women assigned to screening mammography only, 161 interval cancers were diagnosed among more than 32,000 women screened. The primary outcome findings were 2.5 interval cancers per 1,000 screenings among women randomly assigned to MRI, and 5 interval cancers per 1,000 screenings among those randomly assigned to mammography only.6
Because the trial included women aged 50 and older, we can’t apply these results to younger women, who often undergo screening mammography in the United States. In addition, the majority of the population in the Netherlands are of Western European ethnicity, a less-diverse population of women than in the United States. Furthermore, among the tumors that were detected in the MRI group, a larger proportion were DCIS, early-stage tumors, well differentiated, and hormone receptor-positive. This observation supports that many of the MRI-detected tumors were cases of overdiagnosis, or the detection of tumors destined not to cause clinical problems for the patient during her lifetime, or for which earlier diagnosis would impact survival.
We also know that treatment of these small ER-positive tumors carries risks for patients, as we may treat them by depriving a patient of estrogen for the rest of her life, with potential consequences of sexual dysfunction, osteoporosis, and perhaps cardiovascular disease depending on her age at the time of that diagnosis. Weighing the risks and benefits of not only treatment but also use of more sensitive screening techniques such as MRI is extremely important. Although Bakker and colleagues’ study results are interesting, we do not feel they support routinely recommending MRI for women found to have extremely dense breasts with mammography.
Overdiagnosis: A difficult concept
One reason overdiagnosis is so challenging to understand is that it can’t be directly measured, which makes comprehending it that much more problematic for clinicians and our patients.
One way to help grasp the overall issue is to compare screening mammography with cervical and colon cancer screening.
We are well aware that cervical cancer screening has reduced the incidence of mortality from invasive cervical cancer.8 We can argue very validly that the biggest success in any cancer screening program in history and globally has been cervical cancer screening. Our specialty, in particular, should feel proud about this. Screening colonoscopy also has repeatedly been found to reduce colon cancer mortality.9 For breast cancer, decades of media messaging have emphasized the benefits of screening mammograms; however, in contrast with cervical cancer screening and colonoscopy, screening mammography has not reduced the incidence of breast cancer presenting with metastatic or advanced disease. Danish authors pointed out in 2017 that screening mammography has not achieved the hoped for or the promised reduction in breast cancer mortality.10
A report published in the March 2022, issue of Annals of Internal Medicine used modeling techniques to estimate the incidence of overdiagnosis and concluded that, among women aged 50-74 years receiving biennial screening mammograms (consistent with USPSTF recommendations), more than 15% of screen-detected breast cancers would represent cases of overdiagnosis. Of note, the study authors found that, among screen-detected cancers, the proportion representing overdiagnosis among women in their 60s (16.7%) and early 70s (23.6%) was higher than among women in their 50s-60s (11.5%-11.6%).11
The former Chief Medical and Scientific Officer for the American Cancer Society Otis Brawley, MD, has stated that, at the same time that breast cancer screening should not be abandoned, “We must acknowledge that overdiagnosis is common. The benefits of screening have been overstated, and some patients considered as ‘cured’ from breast cancer have, in fact, been harmed by unneeded treatment.”12
“Everybody loves early detection,” said Donald Berry, PhD, from MD Anderson Cancer Center, “but it comes with harms.” He points out that mortality rates have improved for breast cancer, but he attributes it to improved treatment. “The harms [of screening] we know, but the benefits of screening are very uncertain.”13
The importance of health equity is receiving more attention. When examining equity according to breast cancer mortality, ethnic minority populations have worse cancer survival outcomes than White women; the mortality rate is 40% higher among Black women than among White women.1 Lower survival rates are also noted among lower socioeconomic groups and among women who live in rural areas. Lower survival rates among ethnic minority women are also noted for cervical and colorectal cancers.2
In the past, these disparities in mortality were attributed to the historically lower breast cancer screening rates among Black women compared with White women. However, decades of efforts to increase mammography rates have effectively addressed much of the racial/ethnic gap in screening rates.1 In fact, a 2021 study showed Black and Hispanic women to have 6% to 10% higher rates of breast, cervical, and colorectal cancer screening than White women according to US Preventive Services Task Force guidelines.2 The study authors point out that other national data have demonstrated similar results and conclude that “higher cancer mortality among racial/ethnic minority groups will not be reduced solely by increasing rates of cancer screening. Although preventive screenings and timely diagnosis are important elements of prognosis, they are just 2 elements of many along the cancer care continuum that need to be addressed to eliminate disparities in cancer mortality.”
Unfortunately, the randomized trials that have been conducted on mammography have been conducted overwhelmingly in White populations. National registry studies from the Netherlands and Sweden are not representative patient populations for the United States. Recently, the US government proposed an ambitious plan to cut cancer mortality rates and has promised vast amounts of research funding to achieve that goal.3 Hopefully, this funding will support studies which enroll diverse patient populations. We hope to gain knowledge on what elements along the cancer care continuum can be addressed to better reduce or eliminate cancer mortality inequities.
References
1. National Cancer Institute. SEER Explorer. https://seer.cancer.gov/explorer/. Accessed February 9, 2022.
2. Benavidez GA, Zgodic A, Zahnd WE, Eberth JM. Disparities in Meeting USPSTF Breast, Cervical, and Colorectal Cancer Screening Guidelines Among Women in the United States. Prev Chronic Dis. 2021;18:200315. doi: http://dx.doi.org/10.5888/pcd18.200315.
3. Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
Continue to: Limitations of breast MRI...
Limitations of breast MRI
Overall, MRI is a diagnostic and monitoring test. It is costlier than mammography, and because it is not recommended in guidelines as a screening modality for most women, it is not typically covered by insurance. Abbreviated (rapid) MRI is a non-standardized imaging strategy being used at a few health centers. It has a shorter protocol overall than MRI, so it takes less time than current MRI and is less expensive, but there are few data on sensitivity and specificity. It is yet to be determined which populations could benefit from this newer technology.
As mentioned, 41% of women in the Bakker et al trial who were randomly assigned to breast MRI chose not to proceed with that exam even though it would have been at no cost to them.6 Anecdotally, some patients who have undergone MRI say they would forgo it a second time as a screening modality because it was a very unpleasant, stressful experience. It’s not a perfect test, although it is more sensitive than mammography.
Other options for following up dense-breast screening. Besides MRI and abbreviated MRI, the following modalities can be used to evaluate women found to have dense breasts with screening mammograms: CT mammography with contrast, molecular breast imaging, and ultrasonography.
Screening and treatment advances
3D mammography. In the US, the great majority of screening mammography now is performed with tomosynthesis, or what our patients sometimes call 3D mammography. In fact, it is approaching standard of care. Women whose screening mammography includes tomosynthesis are less likely to experience a so-called callback for additional imaging with diagnostic mammography or breast ultrasonography.14
Liquid biopsy. A potential major advancement for making decisions about when to treat cancers in general involves determining the biological behavior of a tumor, based on analysis of either circulating tumor DNA or proteins in the blood. As more experience with this new technology accumulates, the role of liquid biopsies for breast cancer will expand.15 Liquid biopsies for screening remain investigational for now, but they hold tremendous potential.
Noninvasive proteomics. With the development of noninvasive proteomic biomarkers obtained from blood, saliva, or nipple aspiration fluid, there exists the possibility of not just evaluating an image of a tumor seen on a mammogram, but actually studying the biological characteristics of that lesion.16 The cost of this technology is far less in terms of resources than MRI or molecular-based imaging, and actually reveals the flaws with using image-based screening. With proteomics, we can tell whether or not a lump is generating proteins that are going to make that disease biologically meaningful, and treatment decisions can be based on that information. This idea has the potential to disrupt our current breast cancer screening paradigm.
Advocacy’s role in mandating legislation
Many advocacy groups lobby on Capitol Hill for legislation related to health care, but we don’t feel that is the best way to make scientific decisions, and it’s not the way to do medicine. Passionate people, who truly believe that their outcome would have been different had something else been done, have every right to advocate, and should. However, without longer-term data focusing on breast cancer and overall mortality, rather than surrogate outcomes like interval cancers, it is not clear that routinely recommending supplemental MRI will improve survival for women with extremely dense breasts. Unfortunately, overall, earlier diagnosis of highly aggressive breast cancer tumors does not result in better outcomes for patients. ●
- American College of Obstetricians and Gynecologists. Practice Bulletin number 179: breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130: e1-e16. doi: 10.1097/AOG.0000000000002158.
- Sui AL, U.S. Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279-296. doi: 10.7326/M15-2886.
- The American Society of Breast Surgeons. Position statement on screening mammography. https://www.breastsurgeons.org/docs /statements/Position-Statement-on-ScreeningMammography.pdf. Accessed February 15, 2022.
- Monticciolo DL, Malak SF, Friedewald SM, et al. Breast cancer screening recommendations inclusive of all women at average-risk: update from the ACR and Society of Breast Imaging. J Am College Radiol. 2021;18:1280-1288.
- Burton R, Stevenson C. Assessment of breast cancer mortality trends associated with mammographic screening and adjuvant therapy from 1986 to 2013 in the state of Victoria, Australia. JAMA Netw Open. 2020;3:e208249.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102. doi: 10.1056/NEJMoa1903986.
- Seitzman R, Berg W. Average-risk women with dense breasts—what breast screening is appropriate? OBG Manag. 2021;33:18-19. doi: 10.12788/obgm.0155.
- Gopalani SV, Janitz AE, Campbell JE. Cervical cancer incidence and mortality among non-hispanic African American and White women, United States, 1999-2015. J Natl Med Assoc. 2020;112:632-638. doi: 10.1016 /j.jnma.2020.06.007.
- Niikura R, Hirata Y, Suzuki N, et al. Colonoscopy reduces colorectal cancer mortality: a multicenter, long-term, colonoscopy-based cohort study. PLoS One. 2017;12:e0185294.
- Jørgensen KJ, Gøtzsche PC, Kalager M, et al. Breast cancer screening in Denmark. Ann Intern Med. 2017;167:524. doi: 10.7326/L17-0270.
- Ryser MD, Lange J, Inoue IL, et al. Estimation of breast cancer overdiagnosis in a U.S. breast screening cohort. Ann Intern Med. 2022 March 1. doi: 10.7326/M21-3577.
- Brawley OW. Accepting the existence of breast cancer overdiagnosis. Ann Intern Med. 2017;166:364-365. doi:10.7326/M16-2850.
- Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
- Conant EF, Barlow WE, Herschorn SD, et al. Association of digital breast tomosynthesis vs digital mammography with cancer detection and recall rates by age and breast density. JAMA Oncol. 2019;5:635-642. doi: 10.1001 /jamaoncol.2018.7078.
- Tay TK, Tan PH. Liquid biopsy in breast cancer: a focused review. Arch Pathol Lab Med. 2021;145: 678-686. doi: 10.5858/arpa.2019-0559-RA.
- Debald M, Wolgarten M, Walgenbach-Brunagel G, et al. Non-invasive proteomics—thinking about personalized breast cancer screening and treatment. EPMA J. 2010;1:413-420. doi: 10.1007 /s13167-010-0039-9.
We have been interested in the quiz series focused on breast cancer screening for women with dense breasts presented in
The concerns with breast cancer in particular
Breast cancer is not cervical cancer. It isn’t one disease. It is a multitude of diseases that happen to show up in the breast. Some are relatively slow-growing—the kinds of cancers that lend themselves to screening and to early intervention. But other cancers are rapidly-growing; they show up no matter how often or what modality we use for screening. Our goal should be to find an approach to screening that can diagnose breast cancer at a stage where we can intervene and positively impact breast cancer specific and overall mortality.
Screening guidelines vary
The variety of screening guidelines published by different professional organizations reflect differing assumptions and sets of values related to the early diagnosis and treatment of breast cancer. (For a comprehensive table of current screening guidelines, see https://www.cdc.gov/cancer/breast/pdf/breast-cancer-screening-guidelines-508.pdf.)
ACOG’s approach—to offer screening at age 40 but to begin by at least age 50 and, through shared decision making with the patient, screen every 1 or 2 years—is focused on capturing as many cases as we can identify, while minimizing the harms of false-positives.1 The perspective of the US Preventive Services Task Force (USPSTF) recommendations (to screen every 2 years beginning at age 50) is at the population level, a cost-effective approach that will have the greatest benefit while minimizing harms in the population at large.2 The American Society of Breast Surgeons recommends screening to begin by age 40.3 Like the breast surgeons, radiologists dedicated to breast imaging are focused on an individual rather than a population level. They strive to identify each and every instance of possible cancer, and therefore recommend annual screening beginning at age 40.4 However, with more aggressive screening in average-risk women many cases of ductal carcinoma in situ (DCIS) are identified—a lesion that, if not detected, may not impact the woman’s health during her lifetime—representing what some might call “overdiagnosis.” Yet there may be some instances in which the DCIS might affect an individual woman’s health. Unfortunately, we can’t prospectively distinguish between the first and the second types of cases.
We follow American College of Obstetricians and Gynecologists and US Preventive Services Task Force guidelines in discussing screening (both its hazards and benefits) with our average-risk patients beginning at age 40. We talk about risk factors for breast cancer, including breast density, but let patients know that no specific additional imaging is advised, and that density is more common in younger women (one consideration in earlier screening) and is quite common in general. Although we do not send follow-up letters to patients with dense breasts, we do educate our staff so that they can respond appropriately should patients call with questions.
Of course, we all bring to the table values that will impact the decisions that we make for ourselves and for our patients. What an ObGyn might suggest may differ from what a radiologist might suggest. Although we follow recommendations made by the radiologist at screening, an ObGyn wants to take care of the whole human being. We are concerned with bones, heart, everything about the patient, so we approach a patient in a different way. These priorities are reflected in the current varying breast cancer screening guidelines.
Continue to: Research on breast cancer screening varies by design...
Research on breast cancer screening varies by design
There has not been a randomized clinical trial conducted on screening mammography since the days of the analog mammogram. The research that has been conducted is difficult to compare due to variations in screening ages and intervals, technology sensitivity, and patient adherence with recommended screening. Treatments for breast cancer also have changed dramatically over time, so the findings of older studies may no longer be relevant to current breast cancer screening. The kind of analysis that needs to be done is an interrupted time series, where you can look at the trajectory of breast cancer survival and whether screening mammography shifts that survival in any way.
One specific study from Australia measured the impact of newer available breast cancer treatments, including tamoxifen for women with receptor-positive tumors and newer chemotherapy strategies.5 The authors analyzed screening mammography trends in one large province where women aged 50 to 69 were offered biennial screening. Trends from the 1990s showed that more women were being screened over time. Simultaneously, however, advances in therapy were entering clinical practice. The researchers pointed to a substantial decline in mortality from breast cancer from the early 1980s until 2013. But their conclusion was that none of the decline in mortality for breast cancer could be attributed to screening mammography when they looked at time trends; from their perspective all of the important decline in breast cancer mortality resulted from better treatment. They concluded that government programs should not support screening mammography.5
That is a recommendation that we do not support. However, we do recognize the conundrum that mammography is less sensitive among those who have dense breasts. In order to have congruent professional guidelines, we support research funding to determine which types, starting ages, and intervals of screening would be best in various patient populations. The USPSTF cites data from studies performed in the 1980s based on outdated technology; more recent (and relevant) randomized clinical trials have not been performed, and yet this information is critical to provide sufficient evidence to develop appropriate guidelines.
Our recommendations for gathering new data
The kind of data we would find most valuable would assess how different screening strategies impact overall mortality and breast cancer-specific mortality. It would require decades of follow-up—which of course means that screening technology will change over that time. A surrogate for evaluating overall survival is to look at interval cancers, which are all breast cancers diagnosed following negative mammograms and prior to the next screening. These cancers may or may not be biologically active, again focusing us on the need to look at overall survival of the patient. In addition, reducing breast cancer mortality may not reduce overall mortality, because the treatment for breast cancer may cause heart disease, or osteoporosis, or something else that impacts overall survival. These are important considerations for women and physicians who are making choices on treatment. What matters to a patient are 2 overlapping questions:
- Do I have a life-threatening condition or do I not?
- Has screening identified a condition that might lead to treatment that’s unnecessary?
The problem is that with breast cancer we can’t tell the difference. We do not understand the biological potential of a lesion when we evaluate an image on MRI, or computed tomography (CT), or mammography.
A re-look at presented data
A trial conducted by Bakker and colleagues6 was discussed by the authors of the DenseBreast-info.org quiz in which they recommended breast MRI for all women with extremely dense breasts (but no other risk factors for breast cancer) detected on screening mammograms.7 The Bakker study was large and conducted in the Netherlands. The primary outcome of the trial was to compare the incidence of interval breast cancers of women aged 50 to 75 randomly assigned to MRI versus those assigned to continued screening mammography every 2 years. Importantly, among the more than 8,000 women who were assigned to MRI, 59%, or fewer than two-thirds, chose to actually undergo MRI.
Among women randomized to MRI, 20 interval cancers were found—4 were diagnosed in those who actually had MRIs, and 16 were diagnosed among women who were randomized to MRI but didn’t undergo the study. Among women assigned to screening mammography only, 161 interval cancers were diagnosed among more than 32,000 women screened. The primary outcome findings were 2.5 interval cancers per 1,000 screenings among women randomly assigned to MRI, and 5 interval cancers per 1,000 screenings among those randomly assigned to mammography only.6
Because the trial included women aged 50 and older, we can’t apply these results to younger women, who often undergo screening mammography in the United States. In addition, the majority of the population in the Netherlands are of Western European ethnicity, a less-diverse population of women than in the United States. Furthermore, among the tumors that were detected in the MRI group, a larger proportion were DCIS, early-stage tumors, well differentiated, and hormone receptor-positive. This observation supports that many of the MRI-detected tumors were cases of overdiagnosis, or the detection of tumors destined not to cause clinical problems for the patient during her lifetime, or for which earlier diagnosis would impact survival.
We also know that treatment of these small ER-positive tumors carries risks for patients, as we may treat them by depriving a patient of estrogen for the rest of her life, with potential consequences of sexual dysfunction, osteoporosis, and perhaps cardiovascular disease depending on her age at the time of that diagnosis. Weighing the risks and benefits of not only treatment but also use of more sensitive screening techniques such as MRI is extremely important. Although Bakker and colleagues’ study results are interesting, we do not feel they support routinely recommending MRI for women found to have extremely dense breasts with mammography.
Overdiagnosis: A difficult concept
One reason overdiagnosis is so challenging to understand is that it can’t be directly measured, which makes comprehending it that much more problematic for clinicians and our patients.
One way to help grasp the overall issue is to compare screening mammography with cervical and colon cancer screening.
We are well aware that cervical cancer screening has reduced the incidence of mortality from invasive cervical cancer.8 We can argue very validly that the biggest success in any cancer screening program in history and globally has been cervical cancer screening. Our specialty, in particular, should feel proud about this. Screening colonoscopy also has repeatedly been found to reduce colon cancer mortality.9 For breast cancer, decades of media messaging have emphasized the benefits of screening mammograms; however, in contrast with cervical cancer screening and colonoscopy, screening mammography has not reduced the incidence of breast cancer presenting with metastatic or advanced disease. Danish authors pointed out in 2017 that screening mammography has not achieved the hoped for or the promised reduction in breast cancer mortality.10
A report published in the March 2022, issue of Annals of Internal Medicine used modeling techniques to estimate the incidence of overdiagnosis and concluded that, among women aged 50-74 years receiving biennial screening mammograms (consistent with USPSTF recommendations), more than 15% of screen-detected breast cancers would represent cases of overdiagnosis. Of note, the study authors found that, among screen-detected cancers, the proportion representing overdiagnosis among women in their 60s (16.7%) and early 70s (23.6%) was higher than among women in their 50s-60s (11.5%-11.6%).11
The former Chief Medical and Scientific Officer for the American Cancer Society Otis Brawley, MD, has stated that, at the same time that breast cancer screening should not be abandoned, “We must acknowledge that overdiagnosis is common. The benefits of screening have been overstated, and some patients considered as ‘cured’ from breast cancer have, in fact, been harmed by unneeded treatment.”12
“Everybody loves early detection,” said Donald Berry, PhD, from MD Anderson Cancer Center, “but it comes with harms.” He points out that mortality rates have improved for breast cancer, but he attributes it to improved treatment. “The harms [of screening] we know, but the benefits of screening are very uncertain.”13
The importance of health equity is receiving more attention. When examining equity according to breast cancer mortality, ethnic minority populations have worse cancer survival outcomes than White women; the mortality rate is 40% higher among Black women than among White women.1 Lower survival rates are also noted among lower socioeconomic groups and among women who live in rural areas. Lower survival rates among ethnic minority women are also noted for cervical and colorectal cancers.2
In the past, these disparities in mortality were attributed to the historically lower breast cancer screening rates among Black women compared with White women. However, decades of efforts to increase mammography rates have effectively addressed much of the racial/ethnic gap in screening rates.1 In fact, a 2021 study showed Black and Hispanic women to have 6% to 10% higher rates of breast, cervical, and colorectal cancer screening than White women according to US Preventive Services Task Force guidelines.2 The study authors point out that other national data have demonstrated similar results and conclude that “higher cancer mortality among racial/ethnic minority groups will not be reduced solely by increasing rates of cancer screening. Although preventive screenings and timely diagnosis are important elements of prognosis, they are just 2 elements of many along the cancer care continuum that need to be addressed to eliminate disparities in cancer mortality.”
Unfortunately, the randomized trials that have been conducted on mammography have been conducted overwhelmingly in White populations. National registry studies from the Netherlands and Sweden are not representative patient populations for the United States. Recently, the US government proposed an ambitious plan to cut cancer mortality rates and has promised vast amounts of research funding to achieve that goal.3 Hopefully, this funding will support studies which enroll diverse patient populations. We hope to gain knowledge on what elements along the cancer care continuum can be addressed to better reduce or eliminate cancer mortality inequities.
References
1. National Cancer Institute. SEER Explorer. https://seer.cancer.gov/explorer/. Accessed February 9, 2022.
2. Benavidez GA, Zgodic A, Zahnd WE, Eberth JM. Disparities in Meeting USPSTF Breast, Cervical, and Colorectal Cancer Screening Guidelines Among Women in the United States. Prev Chronic Dis. 2021;18:200315. doi: http://dx.doi.org/10.5888/pcd18.200315.
3. Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
Continue to: Limitations of breast MRI...
Limitations of breast MRI
Overall, MRI is a diagnostic and monitoring test. It is costlier than mammography, and because it is not recommended in guidelines as a screening modality for most women, it is not typically covered by insurance. Abbreviated (rapid) MRI is a non-standardized imaging strategy being used at a few health centers. It has a shorter protocol overall than MRI, so it takes less time than current MRI and is less expensive, but there are few data on sensitivity and specificity. It is yet to be determined which populations could benefit from this newer technology.
As mentioned, 41% of women in the Bakker et al trial who were randomly assigned to breast MRI chose not to proceed with that exam even though it would have been at no cost to them.6 Anecdotally, some patients who have undergone MRI say they would forgo it a second time as a screening modality because it was a very unpleasant, stressful experience. It’s not a perfect test, although it is more sensitive than mammography.
Other options for following up dense-breast screening. Besides MRI and abbreviated MRI, the following modalities can be used to evaluate women found to have dense breasts with screening mammograms: CT mammography with contrast, molecular breast imaging, and ultrasonography.
Screening and treatment advances
3D mammography. In the US, the great majority of screening mammography now is performed with tomosynthesis, or what our patients sometimes call 3D mammography. In fact, it is approaching standard of care. Women whose screening mammography includes tomosynthesis are less likely to experience a so-called callback for additional imaging with diagnostic mammography or breast ultrasonography.14
Liquid biopsy. A potential major advancement for making decisions about when to treat cancers in general involves determining the biological behavior of a tumor, based on analysis of either circulating tumor DNA or proteins in the blood. As more experience with this new technology accumulates, the role of liquid biopsies for breast cancer will expand.15 Liquid biopsies for screening remain investigational for now, but they hold tremendous potential.
Noninvasive proteomics. With the development of noninvasive proteomic biomarkers obtained from blood, saliva, or nipple aspiration fluid, there exists the possibility of not just evaluating an image of a tumor seen on a mammogram, but actually studying the biological characteristics of that lesion.16 The cost of this technology is far less in terms of resources than MRI or molecular-based imaging, and actually reveals the flaws with using image-based screening. With proteomics, we can tell whether or not a lump is generating proteins that are going to make that disease biologically meaningful, and treatment decisions can be based on that information. This idea has the potential to disrupt our current breast cancer screening paradigm.
Advocacy’s role in mandating legislation
Many advocacy groups lobby on Capitol Hill for legislation related to health care, but we don’t feel that is the best way to make scientific decisions, and it’s not the way to do medicine. Passionate people, who truly believe that their outcome would have been different had something else been done, have every right to advocate, and should. However, without longer-term data focusing on breast cancer and overall mortality, rather than surrogate outcomes like interval cancers, it is not clear that routinely recommending supplemental MRI will improve survival for women with extremely dense breasts. Unfortunately, overall, earlier diagnosis of highly aggressive breast cancer tumors does not result in better outcomes for patients. ●
We have been interested in the quiz series focused on breast cancer screening for women with dense breasts presented in
The concerns with breast cancer in particular
Breast cancer is not cervical cancer. It isn’t one disease. It is a multitude of diseases that happen to show up in the breast. Some are relatively slow-growing—the kinds of cancers that lend themselves to screening and to early intervention. But other cancers are rapidly-growing; they show up no matter how often or what modality we use for screening. Our goal should be to find an approach to screening that can diagnose breast cancer at a stage where we can intervene and positively impact breast cancer specific and overall mortality.
Screening guidelines vary
The variety of screening guidelines published by different professional organizations reflect differing assumptions and sets of values related to the early diagnosis and treatment of breast cancer. (For a comprehensive table of current screening guidelines, see https://www.cdc.gov/cancer/breast/pdf/breast-cancer-screening-guidelines-508.pdf.)
ACOG’s approach—to offer screening at age 40 but to begin by at least age 50 and, through shared decision making with the patient, screen every 1 or 2 years—is focused on capturing as many cases as we can identify, while minimizing the harms of false-positives.1 The perspective of the US Preventive Services Task Force (USPSTF) recommendations (to screen every 2 years beginning at age 50) is at the population level, a cost-effective approach that will have the greatest benefit while minimizing harms in the population at large.2 The American Society of Breast Surgeons recommends screening to begin by age 40.3 Like the breast surgeons, radiologists dedicated to breast imaging are focused on an individual rather than a population level. They strive to identify each and every instance of possible cancer, and therefore recommend annual screening beginning at age 40.4 However, with more aggressive screening in average-risk women many cases of ductal carcinoma in situ (DCIS) are identified—a lesion that, if not detected, may not impact the woman’s health during her lifetime—representing what some might call “overdiagnosis.” Yet there may be some instances in which the DCIS might affect an individual woman’s health. Unfortunately, we can’t prospectively distinguish between the first and the second types of cases.
We follow American College of Obstetricians and Gynecologists and US Preventive Services Task Force guidelines in discussing screening (both its hazards and benefits) with our average-risk patients beginning at age 40. We talk about risk factors for breast cancer, including breast density, but let patients know that no specific additional imaging is advised, and that density is more common in younger women (one consideration in earlier screening) and is quite common in general. Although we do not send follow-up letters to patients with dense breasts, we do educate our staff so that they can respond appropriately should patients call with questions.
Of course, we all bring to the table values that will impact the decisions that we make for ourselves and for our patients. What an ObGyn might suggest may differ from what a radiologist might suggest. Although we follow recommendations made by the radiologist at screening, an ObGyn wants to take care of the whole human being. We are concerned with bones, heart, everything about the patient, so we approach a patient in a different way. These priorities are reflected in the current varying breast cancer screening guidelines.
Continue to: Research on breast cancer screening varies by design...
Research on breast cancer screening varies by design
There has not been a randomized clinical trial conducted on screening mammography since the days of the analog mammogram. The research that has been conducted is difficult to compare due to variations in screening ages and intervals, technology sensitivity, and patient adherence with recommended screening. Treatments for breast cancer also have changed dramatically over time, so the findings of older studies may no longer be relevant to current breast cancer screening. The kind of analysis that needs to be done is an interrupted time series, where you can look at the trajectory of breast cancer survival and whether screening mammography shifts that survival in any way.
One specific study from Australia measured the impact of newer available breast cancer treatments, including tamoxifen for women with receptor-positive tumors and newer chemotherapy strategies.5 The authors analyzed screening mammography trends in one large province where women aged 50 to 69 were offered biennial screening. Trends from the 1990s showed that more women were being screened over time. Simultaneously, however, advances in therapy were entering clinical practice. The researchers pointed to a substantial decline in mortality from breast cancer from the early 1980s until 2013. But their conclusion was that none of the decline in mortality for breast cancer could be attributed to screening mammography when they looked at time trends; from their perspective all of the important decline in breast cancer mortality resulted from better treatment. They concluded that government programs should not support screening mammography.5
That is a recommendation that we do not support. However, we do recognize the conundrum that mammography is less sensitive among those who have dense breasts. In order to have congruent professional guidelines, we support research funding to determine which types, starting ages, and intervals of screening would be best in various patient populations. The USPSTF cites data from studies performed in the 1980s based on outdated technology; more recent (and relevant) randomized clinical trials have not been performed, and yet this information is critical to provide sufficient evidence to develop appropriate guidelines.
Our recommendations for gathering new data
The kind of data we would find most valuable would assess how different screening strategies impact overall mortality and breast cancer-specific mortality. It would require decades of follow-up—which of course means that screening technology will change over that time. A surrogate for evaluating overall survival is to look at interval cancers, which are all breast cancers diagnosed following negative mammograms and prior to the next screening. These cancers may or may not be biologically active, again focusing us on the need to look at overall survival of the patient. In addition, reducing breast cancer mortality may not reduce overall mortality, because the treatment for breast cancer may cause heart disease, or osteoporosis, or something else that impacts overall survival. These are important considerations for women and physicians who are making choices on treatment. What matters to a patient are 2 overlapping questions:
- Do I have a life-threatening condition or do I not?
- Has screening identified a condition that might lead to treatment that’s unnecessary?
The problem is that with breast cancer we can’t tell the difference. We do not understand the biological potential of a lesion when we evaluate an image on MRI, or computed tomography (CT), or mammography.
A re-look at presented data
A trial conducted by Bakker and colleagues6 was discussed by the authors of the DenseBreast-info.org quiz in which they recommended breast MRI for all women with extremely dense breasts (but no other risk factors for breast cancer) detected on screening mammograms.7 The Bakker study was large and conducted in the Netherlands. The primary outcome of the trial was to compare the incidence of interval breast cancers of women aged 50 to 75 randomly assigned to MRI versus those assigned to continued screening mammography every 2 years. Importantly, among the more than 8,000 women who were assigned to MRI, 59%, or fewer than two-thirds, chose to actually undergo MRI.
Among women randomized to MRI, 20 interval cancers were found—4 were diagnosed in those who actually had MRIs, and 16 were diagnosed among women who were randomized to MRI but didn’t undergo the study. Among women assigned to screening mammography only, 161 interval cancers were diagnosed among more than 32,000 women screened. The primary outcome findings were 2.5 interval cancers per 1,000 screenings among women randomly assigned to MRI, and 5 interval cancers per 1,000 screenings among those randomly assigned to mammography only.6
Because the trial included women aged 50 and older, we can’t apply these results to younger women, who often undergo screening mammography in the United States. In addition, the majority of the population in the Netherlands are of Western European ethnicity, a less-diverse population of women than in the United States. Furthermore, among the tumors that were detected in the MRI group, a larger proportion were DCIS, early-stage tumors, well differentiated, and hormone receptor-positive. This observation supports that many of the MRI-detected tumors were cases of overdiagnosis, or the detection of tumors destined not to cause clinical problems for the patient during her lifetime, or for which earlier diagnosis would impact survival.
We also know that treatment of these small ER-positive tumors carries risks for patients, as we may treat them by depriving a patient of estrogen for the rest of her life, with potential consequences of sexual dysfunction, osteoporosis, and perhaps cardiovascular disease depending on her age at the time of that diagnosis. Weighing the risks and benefits of not only treatment but also use of more sensitive screening techniques such as MRI is extremely important. Although Bakker and colleagues’ study results are interesting, we do not feel they support routinely recommending MRI for women found to have extremely dense breasts with mammography.
Overdiagnosis: A difficult concept
One reason overdiagnosis is so challenging to understand is that it can’t be directly measured, which makes comprehending it that much more problematic for clinicians and our patients.
One way to help grasp the overall issue is to compare screening mammography with cervical and colon cancer screening.
We are well aware that cervical cancer screening has reduced the incidence of mortality from invasive cervical cancer.8 We can argue very validly that the biggest success in any cancer screening program in history and globally has been cervical cancer screening. Our specialty, in particular, should feel proud about this. Screening colonoscopy also has repeatedly been found to reduce colon cancer mortality.9 For breast cancer, decades of media messaging have emphasized the benefits of screening mammograms; however, in contrast with cervical cancer screening and colonoscopy, screening mammography has not reduced the incidence of breast cancer presenting with metastatic or advanced disease. Danish authors pointed out in 2017 that screening mammography has not achieved the hoped for or the promised reduction in breast cancer mortality.10
A report published in the March 2022, issue of Annals of Internal Medicine used modeling techniques to estimate the incidence of overdiagnosis and concluded that, among women aged 50-74 years receiving biennial screening mammograms (consistent with USPSTF recommendations), more than 15% of screen-detected breast cancers would represent cases of overdiagnosis. Of note, the study authors found that, among screen-detected cancers, the proportion representing overdiagnosis among women in their 60s (16.7%) and early 70s (23.6%) was higher than among women in their 50s-60s (11.5%-11.6%).11
The former Chief Medical and Scientific Officer for the American Cancer Society Otis Brawley, MD, has stated that, at the same time that breast cancer screening should not be abandoned, “We must acknowledge that overdiagnosis is common. The benefits of screening have been overstated, and some patients considered as ‘cured’ from breast cancer have, in fact, been harmed by unneeded treatment.”12
“Everybody loves early detection,” said Donald Berry, PhD, from MD Anderson Cancer Center, “but it comes with harms.” He points out that mortality rates have improved for breast cancer, but he attributes it to improved treatment. “The harms [of screening] we know, but the benefits of screening are very uncertain.”13
The importance of health equity is receiving more attention. When examining equity according to breast cancer mortality, ethnic minority populations have worse cancer survival outcomes than White women; the mortality rate is 40% higher among Black women than among White women.1 Lower survival rates are also noted among lower socioeconomic groups and among women who live in rural areas. Lower survival rates among ethnic minority women are also noted for cervical and colorectal cancers.2
In the past, these disparities in mortality were attributed to the historically lower breast cancer screening rates among Black women compared with White women. However, decades of efforts to increase mammography rates have effectively addressed much of the racial/ethnic gap in screening rates.1 In fact, a 2021 study showed Black and Hispanic women to have 6% to 10% higher rates of breast, cervical, and colorectal cancer screening than White women according to US Preventive Services Task Force guidelines.2 The study authors point out that other national data have demonstrated similar results and conclude that “higher cancer mortality among racial/ethnic minority groups will not be reduced solely by increasing rates of cancer screening. Although preventive screenings and timely diagnosis are important elements of prognosis, they are just 2 elements of many along the cancer care continuum that need to be addressed to eliminate disparities in cancer mortality.”
Unfortunately, the randomized trials that have been conducted on mammography have been conducted overwhelmingly in White populations. National registry studies from the Netherlands and Sweden are not representative patient populations for the United States. Recently, the US government proposed an ambitious plan to cut cancer mortality rates and has promised vast amounts of research funding to achieve that goal.3 Hopefully, this funding will support studies which enroll diverse patient populations. We hope to gain knowledge on what elements along the cancer care continuum can be addressed to better reduce or eliminate cancer mortality inequities.
References
1. National Cancer Institute. SEER Explorer. https://seer.cancer.gov/explorer/. Accessed February 9, 2022.
2. Benavidez GA, Zgodic A, Zahnd WE, Eberth JM. Disparities in Meeting USPSTF Breast, Cervical, and Colorectal Cancer Screening Guidelines Among Women in the United States. Prev Chronic Dis. 2021;18:200315. doi: http://dx.doi.org/10.5888/pcd18.200315.
3. Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
Continue to: Limitations of breast MRI...
Limitations of breast MRI
Overall, MRI is a diagnostic and monitoring test. It is costlier than mammography, and because it is not recommended in guidelines as a screening modality for most women, it is not typically covered by insurance. Abbreviated (rapid) MRI is a non-standardized imaging strategy being used at a few health centers. It has a shorter protocol overall than MRI, so it takes less time than current MRI and is less expensive, but there are few data on sensitivity and specificity. It is yet to be determined which populations could benefit from this newer technology.
As mentioned, 41% of women in the Bakker et al trial who were randomly assigned to breast MRI chose not to proceed with that exam even though it would have been at no cost to them.6 Anecdotally, some patients who have undergone MRI say they would forgo it a second time as a screening modality because it was a very unpleasant, stressful experience. It’s not a perfect test, although it is more sensitive than mammography.
Other options for following up dense-breast screening. Besides MRI and abbreviated MRI, the following modalities can be used to evaluate women found to have dense breasts with screening mammograms: CT mammography with contrast, molecular breast imaging, and ultrasonography.
Screening and treatment advances
3D mammography. In the US, the great majority of screening mammography now is performed with tomosynthesis, or what our patients sometimes call 3D mammography. In fact, it is approaching standard of care. Women whose screening mammography includes tomosynthesis are less likely to experience a so-called callback for additional imaging with diagnostic mammography or breast ultrasonography.14
Liquid biopsy. A potential major advancement for making decisions about when to treat cancers in general involves determining the biological behavior of a tumor, based on analysis of either circulating tumor DNA or proteins in the blood. As more experience with this new technology accumulates, the role of liquid biopsies for breast cancer will expand.15 Liquid biopsies for screening remain investigational for now, but they hold tremendous potential.
Noninvasive proteomics. With the development of noninvasive proteomic biomarkers obtained from blood, saliva, or nipple aspiration fluid, there exists the possibility of not just evaluating an image of a tumor seen on a mammogram, but actually studying the biological characteristics of that lesion.16 The cost of this technology is far less in terms of resources than MRI or molecular-based imaging, and actually reveals the flaws with using image-based screening. With proteomics, we can tell whether or not a lump is generating proteins that are going to make that disease biologically meaningful, and treatment decisions can be based on that information. This idea has the potential to disrupt our current breast cancer screening paradigm.
Advocacy’s role in mandating legislation
Many advocacy groups lobby on Capitol Hill for legislation related to health care, but we don’t feel that is the best way to make scientific decisions, and it’s not the way to do medicine. Passionate people, who truly believe that their outcome would have been different had something else been done, have every right to advocate, and should. However, without longer-term data focusing on breast cancer and overall mortality, rather than surrogate outcomes like interval cancers, it is not clear that routinely recommending supplemental MRI will improve survival for women with extremely dense breasts. Unfortunately, overall, earlier diagnosis of highly aggressive breast cancer tumors does not result in better outcomes for patients. ●
- American College of Obstetricians and Gynecologists. Practice Bulletin number 179: breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130: e1-e16. doi: 10.1097/AOG.0000000000002158.
- Sui AL, U.S. Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279-296. doi: 10.7326/M15-2886.
- The American Society of Breast Surgeons. Position statement on screening mammography. https://www.breastsurgeons.org/docs /statements/Position-Statement-on-ScreeningMammography.pdf. Accessed February 15, 2022.
- Monticciolo DL, Malak SF, Friedewald SM, et al. Breast cancer screening recommendations inclusive of all women at average-risk: update from the ACR and Society of Breast Imaging. J Am College Radiol. 2021;18:1280-1288.
- Burton R, Stevenson C. Assessment of breast cancer mortality trends associated with mammographic screening and adjuvant therapy from 1986 to 2013 in the state of Victoria, Australia. JAMA Netw Open. 2020;3:e208249.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102. doi: 10.1056/NEJMoa1903986.
- Seitzman R, Berg W. Average-risk women with dense breasts—what breast screening is appropriate? OBG Manag. 2021;33:18-19. doi: 10.12788/obgm.0155.
- Gopalani SV, Janitz AE, Campbell JE. Cervical cancer incidence and mortality among non-hispanic African American and White women, United States, 1999-2015. J Natl Med Assoc. 2020;112:632-638. doi: 10.1016 /j.jnma.2020.06.007.
- Niikura R, Hirata Y, Suzuki N, et al. Colonoscopy reduces colorectal cancer mortality: a multicenter, long-term, colonoscopy-based cohort study. PLoS One. 2017;12:e0185294.
- Jørgensen KJ, Gøtzsche PC, Kalager M, et al. Breast cancer screening in Denmark. Ann Intern Med. 2017;167:524. doi: 10.7326/L17-0270.
- Ryser MD, Lange J, Inoue IL, et al. Estimation of breast cancer overdiagnosis in a U.S. breast screening cohort. Ann Intern Med. 2022 March 1. doi: 10.7326/M21-3577.
- Brawley OW. Accepting the existence of breast cancer overdiagnosis. Ann Intern Med. 2017;166:364-365. doi:10.7326/M16-2850.
- Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
- Conant EF, Barlow WE, Herschorn SD, et al. Association of digital breast tomosynthesis vs digital mammography with cancer detection and recall rates by age and breast density. JAMA Oncol. 2019;5:635-642. doi: 10.1001 /jamaoncol.2018.7078.
- Tay TK, Tan PH. Liquid biopsy in breast cancer: a focused review. Arch Pathol Lab Med. 2021;145: 678-686. doi: 10.5858/arpa.2019-0559-RA.
- Debald M, Wolgarten M, Walgenbach-Brunagel G, et al. Non-invasive proteomics—thinking about personalized breast cancer screening and treatment. EPMA J. 2010;1:413-420. doi: 10.1007 /s13167-010-0039-9.
- American College of Obstetricians and Gynecologists. Practice Bulletin number 179: breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130: e1-e16. doi: 10.1097/AOG.0000000000002158.
- Sui AL, U.S. Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279-296. doi: 10.7326/M15-2886.
- The American Society of Breast Surgeons. Position statement on screening mammography. https://www.breastsurgeons.org/docs /statements/Position-Statement-on-ScreeningMammography.pdf. Accessed February 15, 2022.
- Monticciolo DL, Malak SF, Friedewald SM, et al. Breast cancer screening recommendations inclusive of all women at average-risk: update from the ACR and Society of Breast Imaging. J Am College Radiol. 2021;18:1280-1288.
- Burton R, Stevenson C. Assessment of breast cancer mortality trends associated with mammographic screening and adjuvant therapy from 1986 to 2013 in the state of Victoria, Australia. JAMA Netw Open. 2020;3:e208249.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102. doi: 10.1056/NEJMoa1903986.
- Seitzman R, Berg W. Average-risk women with dense breasts—what breast screening is appropriate? OBG Manag. 2021;33:18-19. doi: 10.12788/obgm.0155.
- Gopalani SV, Janitz AE, Campbell JE. Cervical cancer incidence and mortality among non-hispanic African American and White women, United States, 1999-2015. J Natl Med Assoc. 2020;112:632-638. doi: 10.1016 /j.jnma.2020.06.007.
- Niikura R, Hirata Y, Suzuki N, et al. Colonoscopy reduces colorectal cancer mortality: a multicenter, long-term, colonoscopy-based cohort study. PLoS One. 2017;12:e0185294.
- Jørgensen KJ, Gøtzsche PC, Kalager M, et al. Breast cancer screening in Denmark. Ann Intern Med. 2017;167:524. doi: 10.7326/L17-0270.
- Ryser MD, Lange J, Inoue IL, et al. Estimation of breast cancer overdiagnosis in a U.S. breast screening cohort. Ann Intern Med. 2022 March 1. doi: 10.7326/M21-3577.
- Brawley OW. Accepting the existence of breast cancer overdiagnosis. Ann Intern Med. 2017;166:364-365. doi:10.7326/M16-2850.
- Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
- Conant EF, Barlow WE, Herschorn SD, et al. Association of digital breast tomosynthesis vs digital mammography with cancer detection and recall rates by age and breast density. JAMA Oncol. 2019;5:635-642. doi: 10.1001 /jamaoncol.2018.7078.
- Tay TK, Tan PH. Liquid biopsy in breast cancer: a focused review. Arch Pathol Lab Med. 2021;145: 678-686. doi: 10.5858/arpa.2019-0559-RA.
- Debald M, Wolgarten M, Walgenbach-Brunagel G, et al. Non-invasive proteomics—thinking about personalized breast cancer screening and treatment. EPMA J. 2010;1:413-420. doi: 10.1007 /s13167-010-0039-9.
Telehealth apps in ObGyn practice
The COVID-19 pandemic has presented increasing demands on health care systems internationally. In addition to redistribution of inpatient health care resources, outpatient care practices evolved, with health care providers offering streamlined access to care to patients via telehealth.
Due to updated insurance practices, physicians now can receive reimbursement via private insurers, Medicare, and Medicaid (as determined by states) for telehealth visits both related and unrelated to COVID-19 care. Increased telehealth use has advantages, including increased health care access, reduced in-clinic wait times, and reduced patient and physician travel time. Within the field of obstetrics and gynecology, clinicians have used telehealth to maintain access to prenatal maternity care while redirecting resources and minimizing the risk of COVID-19 transmission. Additional advantages include provision of care during expanded hours, including evenings and weekends, to increase patient access without increasing the demand on office support staff and the ability to bill for 5- to 10-minute phone counseling encounters.1 Research shows that patients express satisfaction regarding the quality of telehealth care in the setting of prenatal care.2
In February 2020, the American College of Obstetricians and Gynecologists (ACOG) released a Committee Opinion regarding telehealth use in ObGyn, a sign of telehealth’s likely long-standing role within the field.3 Within the statement, ACOG commented on the increasing application of telemedicine in all aspects of obstetrics and gynecology and recommended that physicians become acquainted with new technologies and consider using them in their practice.
There is a large opportunity for development of mobile applications (apps) to further streamline telehealth-based medical care. During the pandemic, the Centers for Medicare and Medicaid Services instituted waivers for telemedicine use on non-HIPAA (Health Insurance Portability and Accountability Act) compliant video communications products, such as Google+ Hangout and Skype. However, HIPAA-compliant video services are preferred, and many virtual apps have released methods for patient communication that meet HIPAA guidelines.1,4 These apps offer services such as phone- and video-based patient visits, appointment scheduling, secure physician-patient messaging, and electronic health record (EHR) documentation.
App recommendations
To identify current mobile apps with clinical use for the ObGyn, we conducted a search of the Apple App Store using the term “telehealth” between December 1, 2021 and January 1, 2022. We limited search results to apps that had at least 1,000 user ratings and to HIPAA-compliant user communication apps. Based on our review, we selected 4 apps to highlight here: Doximity, OhMD, Spruce, and Telehealth by SimplePractice (TABLE). We excluded apps that were advertised as having internal medical clinicians with first patient encounter on-demand through the app or that were associated with a singular insurance company or hospital system.
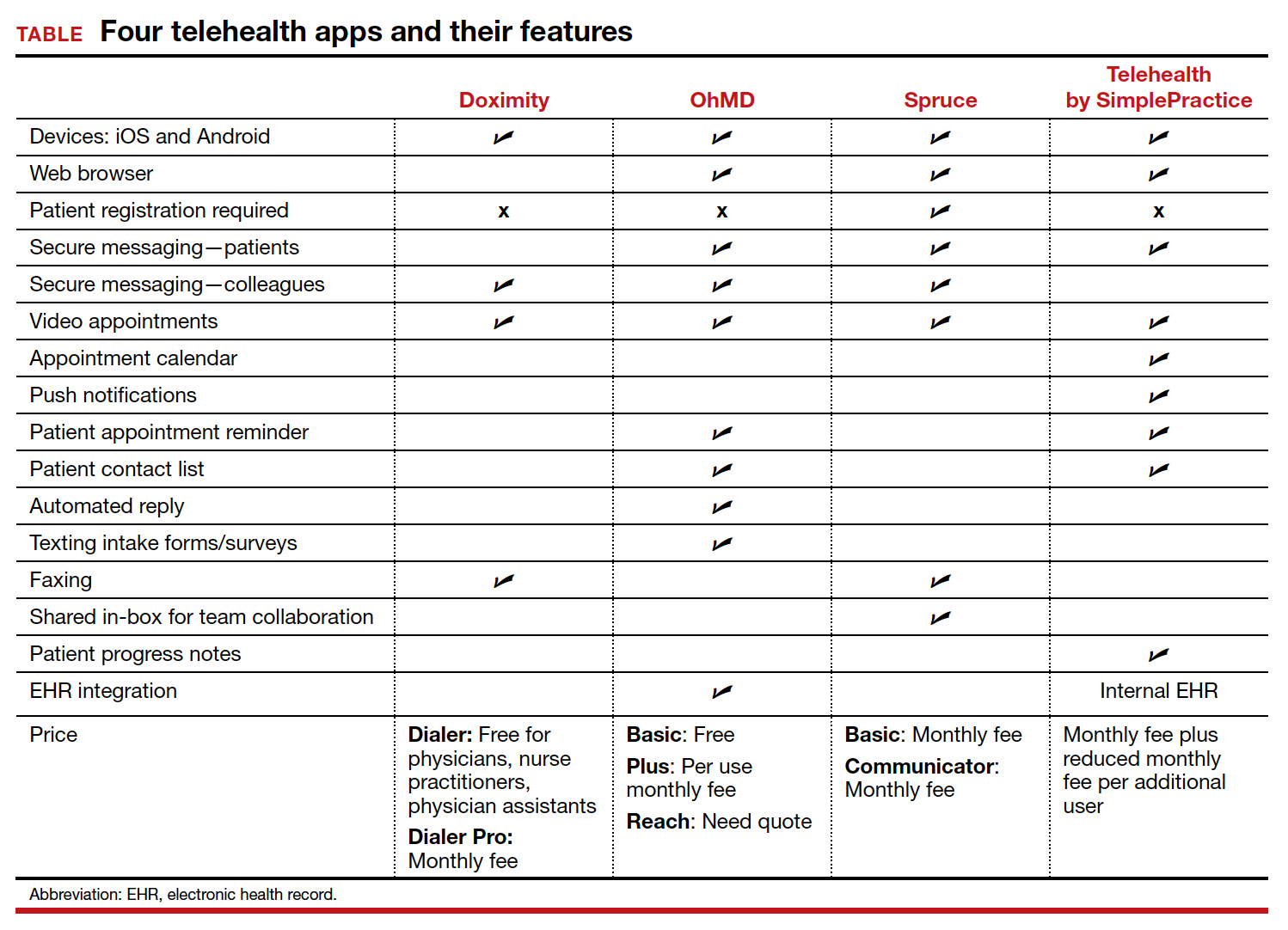
These apps are largely enabled for iOS and Android mobile devices and are offered at a range of price points for individual physician and practice-scale clinical implementation. Most apps offer secure messaging services between health care practitioners in addition to HIPAA-compliant patient messaging. Some apps offer additional features with the aim to increase patient attendance; these include push notifications, appointment reminders, and an option for automated replies with clinic information. For an additional fee, several apps offer integration to established EHR systems.
An additional tool
The COVID-19 pandemic caused health care systems and individual clinicians to rapidly evolve their practices to maintain patient access to essential health care. Notably, the pandemic led to accelerated implementation of virtual health care services. Telehealth apps likely will become another tool that ObGyns can use to improve the efficiency of their clinical practice and expand patient access to care. ●
- Karram M, Baum N. Telemedicine: a primer for today’s ObGyn. OBG Manag. 2020;32:28-32.
- Marko KI, Ganju N, Krapf JM, et al. A mobile prenatal care app to reduce in-person visits: prospective controlled trial. JMIR Mhealth Uhealth. 2019;7:e10520.
- American College of Obstetricians and Gynecologists. Implementing telehealth in practice: committee opinion no. 798. Obstet Gynecol. 2020;135:e73-e79.
- Karram M, Dooley A, de la Houssaye N, et al. Telemedicine: navigating legal issues. OBG Manag. 2020;32:18-24.
The COVID-19 pandemic has presented increasing demands on health care systems internationally. In addition to redistribution of inpatient health care resources, outpatient care practices evolved, with health care providers offering streamlined access to care to patients via telehealth.
Due to updated insurance practices, physicians now can receive reimbursement via private insurers, Medicare, and Medicaid (as determined by states) for telehealth visits both related and unrelated to COVID-19 care. Increased telehealth use has advantages, including increased health care access, reduced in-clinic wait times, and reduced patient and physician travel time. Within the field of obstetrics and gynecology, clinicians have used telehealth to maintain access to prenatal maternity care while redirecting resources and minimizing the risk of COVID-19 transmission. Additional advantages include provision of care during expanded hours, including evenings and weekends, to increase patient access without increasing the demand on office support staff and the ability to bill for 5- to 10-minute phone counseling encounters.1 Research shows that patients express satisfaction regarding the quality of telehealth care in the setting of prenatal care.2
In February 2020, the American College of Obstetricians and Gynecologists (ACOG) released a Committee Opinion regarding telehealth use in ObGyn, a sign of telehealth’s likely long-standing role within the field.3 Within the statement, ACOG commented on the increasing application of telemedicine in all aspects of obstetrics and gynecology and recommended that physicians become acquainted with new technologies and consider using them in their practice.
There is a large opportunity for development of mobile applications (apps) to further streamline telehealth-based medical care. During the pandemic, the Centers for Medicare and Medicaid Services instituted waivers for telemedicine use on non-HIPAA (Health Insurance Portability and Accountability Act) compliant video communications products, such as Google+ Hangout and Skype. However, HIPAA-compliant video services are preferred, and many virtual apps have released methods for patient communication that meet HIPAA guidelines.1,4 These apps offer services such as phone- and video-based patient visits, appointment scheduling, secure physician-patient messaging, and electronic health record (EHR) documentation.
App recommendations
To identify current mobile apps with clinical use for the ObGyn, we conducted a search of the Apple App Store using the term “telehealth” between December 1, 2021 and January 1, 2022. We limited search results to apps that had at least 1,000 user ratings and to HIPAA-compliant user communication apps. Based on our review, we selected 4 apps to highlight here: Doximity, OhMD, Spruce, and Telehealth by SimplePractice (TABLE). We excluded apps that were advertised as having internal medical clinicians with first patient encounter on-demand through the app or that were associated with a singular insurance company or hospital system.

These apps are largely enabled for iOS and Android mobile devices and are offered at a range of price points for individual physician and practice-scale clinical implementation. Most apps offer secure messaging services between health care practitioners in addition to HIPAA-compliant patient messaging. Some apps offer additional features with the aim to increase patient attendance; these include push notifications, appointment reminders, and an option for automated replies with clinic information. For an additional fee, several apps offer integration to established EHR systems.
An additional tool
The COVID-19 pandemic caused health care systems and individual clinicians to rapidly evolve their practices to maintain patient access to essential health care. Notably, the pandemic led to accelerated implementation of virtual health care services. Telehealth apps likely will become another tool that ObGyns can use to improve the efficiency of their clinical practice and expand patient access to care. ●
The COVID-19 pandemic has presented increasing demands on health care systems internationally. In addition to redistribution of inpatient health care resources, outpatient care practices evolved, with health care providers offering streamlined access to care to patients via telehealth.
Due to updated insurance practices, physicians now can receive reimbursement via private insurers, Medicare, and Medicaid (as determined by states) for telehealth visits both related and unrelated to COVID-19 care. Increased telehealth use has advantages, including increased health care access, reduced in-clinic wait times, and reduced patient and physician travel time. Within the field of obstetrics and gynecology, clinicians have used telehealth to maintain access to prenatal maternity care while redirecting resources and minimizing the risk of COVID-19 transmission. Additional advantages include provision of care during expanded hours, including evenings and weekends, to increase patient access without increasing the demand on office support staff and the ability to bill for 5- to 10-minute phone counseling encounters.1 Research shows that patients express satisfaction regarding the quality of telehealth care in the setting of prenatal care.2
In February 2020, the American College of Obstetricians and Gynecologists (ACOG) released a Committee Opinion regarding telehealth use in ObGyn, a sign of telehealth’s likely long-standing role within the field.3 Within the statement, ACOG commented on the increasing application of telemedicine in all aspects of obstetrics and gynecology and recommended that physicians become acquainted with new technologies and consider using them in their practice.
There is a large opportunity for development of mobile applications (apps) to further streamline telehealth-based medical care. During the pandemic, the Centers for Medicare and Medicaid Services instituted waivers for telemedicine use on non-HIPAA (Health Insurance Portability and Accountability Act) compliant video communications products, such as Google+ Hangout and Skype. However, HIPAA-compliant video services are preferred, and many virtual apps have released methods for patient communication that meet HIPAA guidelines.1,4 These apps offer services such as phone- and video-based patient visits, appointment scheduling, secure physician-patient messaging, and electronic health record (EHR) documentation.
App recommendations
To identify current mobile apps with clinical use for the ObGyn, we conducted a search of the Apple App Store using the term “telehealth” between December 1, 2021 and January 1, 2022. We limited search results to apps that had at least 1,000 user ratings and to HIPAA-compliant user communication apps. Based on our review, we selected 4 apps to highlight here: Doximity, OhMD, Spruce, and Telehealth by SimplePractice (TABLE). We excluded apps that were advertised as having internal medical clinicians with first patient encounter on-demand through the app or that were associated with a singular insurance company or hospital system.

These apps are largely enabled for iOS and Android mobile devices and are offered at a range of price points for individual physician and practice-scale clinical implementation. Most apps offer secure messaging services between health care practitioners in addition to HIPAA-compliant patient messaging. Some apps offer additional features with the aim to increase patient attendance; these include push notifications, appointment reminders, and an option for automated replies with clinic information. For an additional fee, several apps offer integration to established EHR systems.
An additional tool
The COVID-19 pandemic caused health care systems and individual clinicians to rapidly evolve their practices to maintain patient access to essential health care. Notably, the pandemic led to accelerated implementation of virtual health care services. Telehealth apps likely will become another tool that ObGyns can use to improve the efficiency of their clinical practice and expand patient access to care. ●
- Karram M, Baum N. Telemedicine: a primer for today’s ObGyn. OBG Manag. 2020;32:28-32.
- Marko KI, Ganju N, Krapf JM, et al. A mobile prenatal care app to reduce in-person visits: prospective controlled trial. JMIR Mhealth Uhealth. 2019;7:e10520.
- American College of Obstetricians and Gynecologists. Implementing telehealth in practice: committee opinion no. 798. Obstet Gynecol. 2020;135:e73-e79.
- Karram M, Dooley A, de la Houssaye N, et al. Telemedicine: navigating legal issues. OBG Manag. 2020;32:18-24.
- Karram M, Baum N. Telemedicine: a primer for today’s ObGyn. OBG Manag. 2020;32:28-32.
- Marko KI, Ganju N, Krapf JM, et al. A mobile prenatal care app to reduce in-person visits: prospective controlled trial. JMIR Mhealth Uhealth. 2019;7:e10520.
- American College of Obstetricians and Gynecologists. Implementing telehealth in practice: committee opinion no. 798. Obstet Gynecol. 2020;135:e73-e79.
- Karram M, Dooley A, de la Houssaye N, et al. Telemedicine: navigating legal issues. OBG Manag. 2020;32:18-24.
Big missed opportunities for BP control in premenopausal women
A new report shows considerable gaps in the awareness, treatment, and control of hypertension in premenopausal women in the United States, with a key driver being regular access to health care.
In a nationally representative sample of women ages 35-54 with no prior cardiovascular disease, the prevalence of hypertension increased 8% from an estimated 15.2 million women between 2011 and 2014 to 16.4 million women between 2015 and 2018.
What’s more, the percentage of women with controlled hypertension dropped over the two time periods from 55% to 50%, which is well below the government’s Million Hearts target of 70%.
Missed opportunities for hypertension control in these premenopausal women were a lack of awareness of their hypertension in 23%, ineffective treatment in 34%, and a lack of health care access in 43%; increasing to 51% in non-Hispanic Black patients and 56% in Hispanic patients.
Notably, lack of health care access affected an estimated 3.1 million women (45%) in 2011-2014 and 3.5 million women (43%) in 2015-2018.
Equally stubborn over the two time periods was the lack of effective treatment, affecting 2.1 million (31%) versus 2.8 million (34%) women, and lack of awareness, affecting 1.6 million (24%) versus 1.9 million (23%) women.
“There’s been no improvement over the past decade, and there is evidence of race/ethnic disparities,” study author Susan Hennessy, PhD, said at the recent Epidemiology, Prevention/Lifestyle & Cardiometabolic Health (EPI|Lifestyle) 2022 conference sponsored by the American Heart Association.
The prevalence of uncontrolled hypertension among non-Hispanic Whites was less than that of the U.S. population, at 44%, and most of the missed opportunities were due to uncontrolled blood pressure (BP), noted Dr. Hennessy, a researcher with the University of California, San Francisco School of Medicine.
However, the uncontrolled prevalence was 54% in non-Hispanic Black women and 66% in Hispanic women. “In both of these subgroups, over half of the missed opportunities occur because these women have no regular access to health care,” she said.
In women who identified as “other,” which includes non-Hispanic Asian and mixed-race populations, the uncontrolled prevalence reached 70%, and the biggest missed opportunity was in those who were untreated.
Raising awareness, empowering women, and delivery of guideline-concordant care will help premenopausal women gain control of their blood pressure, Dr. Hennessy said. “But underpinning all of this is ensuring equitable health care access, because if we fail to get women into the system, then we have no opportunity to help them lower their blood pressure.”
She reminded the audience that cardiovascular disease (CVD) is the number one killer of women in the United States and that CVD risk, mediated through hypertension, increases after menopause. Thus, managing hypertension prior to this life event is an important element of primary prevention of CVD and should be a priority.
Session moderator Sadiya S. Khan, MD, Northwestern University Feinberg School of Medicine, Chicago, told this news organization that the findings should raise “alarm and concern that hypertension is not just a disease of the old but very prevalent in younger women, particularly around the time of pregnancy. And this is a clear driver of maternal morbidity and mortality as well.”
“This idea that patients should ‘Know Your Numbers’ is really important, and we talk a lot about that for hypertension, but if you don’t have a doctor, if you don’t have someone to go to, it’s very hard to know or understand what your numbers mean,” she said. “I think that’s really the main message.”
Speaking to this news organization, Dr. Hennessy said there’s no simple solution to the problem, given that some women are not even in the system, whereas others are not being treated effectively, but that increasing opportunities to screen BP would be a start. That could be through community programs, similar to the Barbershop Hypertension trial, or by making BP devices available for home monitoring.
“Again, this is about empowering ourselves to take some level of control, but, as a system, we have to be able to make it equitable for everyone and make sure they have the right equipment, the right cuff size,” she said. “The disparities arise because of the social determinants of health, so if these women are struggling to put food on the table, they aren’t going to be able to afford a blood pressure cuff.”
During a discussion of the findings, audience members noted that the National Health and Nutrition Examination Survey (NHANES) data used for the analysis were somewhat dated. Dr. Hennessy also pointed out that NHANES blood pressure is measured up to three times during a single visit, which differs from clinical practice, and that responses were based on self-report and thus subject to recall bias.
The sample included 3,343 women aged 35-54 years with no prior cardiovascular disease, representing an estimated 31.6 million American women. Hypertension was defined by a systolic BP of at least 140 mm Hg or a diastolic BP of at least 90 mm Hg or current BP medication use.
The authors and Dr. Khan report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new report shows considerable gaps in the awareness, treatment, and control of hypertension in premenopausal women in the United States, with a key driver being regular access to health care.
In a nationally representative sample of women ages 35-54 with no prior cardiovascular disease, the prevalence of hypertension increased 8% from an estimated 15.2 million women between 2011 and 2014 to 16.4 million women between 2015 and 2018.
What’s more, the percentage of women with controlled hypertension dropped over the two time periods from 55% to 50%, which is well below the government’s Million Hearts target of 70%.
Missed opportunities for hypertension control in these premenopausal women were a lack of awareness of their hypertension in 23%, ineffective treatment in 34%, and a lack of health care access in 43%; increasing to 51% in non-Hispanic Black patients and 56% in Hispanic patients.
Notably, lack of health care access affected an estimated 3.1 million women (45%) in 2011-2014 and 3.5 million women (43%) in 2015-2018.
Equally stubborn over the two time periods was the lack of effective treatment, affecting 2.1 million (31%) versus 2.8 million (34%) women, and lack of awareness, affecting 1.6 million (24%) versus 1.9 million (23%) women.
“There’s been no improvement over the past decade, and there is evidence of race/ethnic disparities,” study author Susan Hennessy, PhD, said at the recent Epidemiology, Prevention/Lifestyle & Cardiometabolic Health (EPI|Lifestyle) 2022 conference sponsored by the American Heart Association.
The prevalence of uncontrolled hypertension among non-Hispanic Whites was less than that of the U.S. population, at 44%, and most of the missed opportunities were due to uncontrolled blood pressure (BP), noted Dr. Hennessy, a researcher with the University of California, San Francisco School of Medicine.
However, the uncontrolled prevalence was 54% in non-Hispanic Black women and 66% in Hispanic women. “In both of these subgroups, over half of the missed opportunities occur because these women have no regular access to health care,” she said.
In women who identified as “other,” which includes non-Hispanic Asian and mixed-race populations, the uncontrolled prevalence reached 70%, and the biggest missed opportunity was in those who were untreated.
Raising awareness, empowering women, and delivery of guideline-concordant care will help premenopausal women gain control of their blood pressure, Dr. Hennessy said. “But underpinning all of this is ensuring equitable health care access, because if we fail to get women into the system, then we have no opportunity to help them lower their blood pressure.”
She reminded the audience that cardiovascular disease (CVD) is the number one killer of women in the United States and that CVD risk, mediated through hypertension, increases after menopause. Thus, managing hypertension prior to this life event is an important element of primary prevention of CVD and should be a priority.
Session moderator Sadiya S. Khan, MD, Northwestern University Feinberg School of Medicine, Chicago, told this news organization that the findings should raise “alarm and concern that hypertension is not just a disease of the old but very prevalent in younger women, particularly around the time of pregnancy. And this is a clear driver of maternal morbidity and mortality as well.”
“This idea that patients should ‘Know Your Numbers’ is really important, and we talk a lot about that for hypertension, but if you don’t have a doctor, if you don’t have someone to go to, it’s very hard to know or understand what your numbers mean,” she said. “I think that’s really the main message.”
Speaking to this news organization, Dr. Hennessy said there’s no simple solution to the problem, given that some women are not even in the system, whereas others are not being treated effectively, but that increasing opportunities to screen BP would be a start. That could be through community programs, similar to the Barbershop Hypertension trial, or by making BP devices available for home monitoring.
“Again, this is about empowering ourselves to take some level of control, but, as a system, we have to be able to make it equitable for everyone and make sure they have the right equipment, the right cuff size,” she said. “The disparities arise because of the social determinants of health, so if these women are struggling to put food on the table, they aren’t going to be able to afford a blood pressure cuff.”
During a discussion of the findings, audience members noted that the National Health and Nutrition Examination Survey (NHANES) data used for the analysis were somewhat dated. Dr. Hennessy also pointed out that NHANES blood pressure is measured up to three times during a single visit, which differs from clinical practice, and that responses were based on self-report and thus subject to recall bias.
The sample included 3,343 women aged 35-54 years with no prior cardiovascular disease, representing an estimated 31.6 million American women. Hypertension was defined by a systolic BP of at least 140 mm Hg or a diastolic BP of at least 90 mm Hg or current BP medication use.
The authors and Dr. Khan report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new report shows considerable gaps in the awareness, treatment, and control of hypertension in premenopausal women in the United States, with a key driver being regular access to health care.
In a nationally representative sample of women ages 35-54 with no prior cardiovascular disease, the prevalence of hypertension increased 8% from an estimated 15.2 million women between 2011 and 2014 to 16.4 million women between 2015 and 2018.
What’s more, the percentage of women with controlled hypertension dropped over the two time periods from 55% to 50%, which is well below the government’s Million Hearts target of 70%.
Missed opportunities for hypertension control in these premenopausal women were a lack of awareness of their hypertension in 23%, ineffective treatment in 34%, and a lack of health care access in 43%; increasing to 51% in non-Hispanic Black patients and 56% in Hispanic patients.
Notably, lack of health care access affected an estimated 3.1 million women (45%) in 2011-2014 and 3.5 million women (43%) in 2015-2018.
Equally stubborn over the two time periods was the lack of effective treatment, affecting 2.1 million (31%) versus 2.8 million (34%) women, and lack of awareness, affecting 1.6 million (24%) versus 1.9 million (23%) women.
“There’s been no improvement over the past decade, and there is evidence of race/ethnic disparities,” study author Susan Hennessy, PhD, said at the recent Epidemiology, Prevention/Lifestyle & Cardiometabolic Health (EPI|Lifestyle) 2022 conference sponsored by the American Heart Association.
The prevalence of uncontrolled hypertension among non-Hispanic Whites was less than that of the U.S. population, at 44%, and most of the missed opportunities were due to uncontrolled blood pressure (BP), noted Dr. Hennessy, a researcher with the University of California, San Francisco School of Medicine.
However, the uncontrolled prevalence was 54% in non-Hispanic Black women and 66% in Hispanic women. “In both of these subgroups, over half of the missed opportunities occur because these women have no regular access to health care,” she said.
In women who identified as “other,” which includes non-Hispanic Asian and mixed-race populations, the uncontrolled prevalence reached 70%, and the biggest missed opportunity was in those who were untreated.
Raising awareness, empowering women, and delivery of guideline-concordant care will help premenopausal women gain control of their blood pressure, Dr. Hennessy said. “But underpinning all of this is ensuring equitable health care access, because if we fail to get women into the system, then we have no opportunity to help them lower their blood pressure.”
She reminded the audience that cardiovascular disease (CVD) is the number one killer of women in the United States and that CVD risk, mediated through hypertension, increases after menopause. Thus, managing hypertension prior to this life event is an important element of primary prevention of CVD and should be a priority.
Session moderator Sadiya S. Khan, MD, Northwestern University Feinberg School of Medicine, Chicago, told this news organization that the findings should raise “alarm and concern that hypertension is not just a disease of the old but very prevalent in younger women, particularly around the time of pregnancy. And this is a clear driver of maternal morbidity and mortality as well.”
“This idea that patients should ‘Know Your Numbers’ is really important, and we talk a lot about that for hypertension, but if you don’t have a doctor, if you don’t have someone to go to, it’s very hard to know or understand what your numbers mean,” she said. “I think that’s really the main message.”
Speaking to this news organization, Dr. Hennessy said there’s no simple solution to the problem, given that some women are not even in the system, whereas others are not being treated effectively, but that increasing opportunities to screen BP would be a start. That could be through community programs, similar to the Barbershop Hypertension trial, or by making BP devices available for home monitoring.
“Again, this is about empowering ourselves to take some level of control, but, as a system, we have to be able to make it equitable for everyone and make sure they have the right equipment, the right cuff size,” she said. “The disparities arise because of the social determinants of health, so if these women are struggling to put food on the table, they aren’t going to be able to afford a blood pressure cuff.”
During a discussion of the findings, audience members noted that the National Health and Nutrition Examination Survey (NHANES) data used for the analysis were somewhat dated. Dr. Hennessy also pointed out that NHANES blood pressure is measured up to three times during a single visit, which differs from clinical practice, and that responses were based on self-report and thus subject to recall bias.
The sample included 3,343 women aged 35-54 years with no prior cardiovascular disease, representing an estimated 31.6 million American women. Hypertension was defined by a systolic BP of at least 140 mm Hg or a diastolic BP of at least 90 mm Hg or current BP medication use.
The authors and Dr. Khan report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Biden administration’s new test-to-treat program pits pharmacists against physicians
The Biden administration’s new test-to-treat program is simple on the surface: if you feel like you may have COVID-19, go to a pharmacy, get tested, and, if positive, get treated with an antiviral medication on the spot.
One large physicians’ group is concerned that the program leaves doctors on the margins, and may put patients at risk if there are adverse effects from the medications. Pharmacists groups, on the other hand, say the program is too restrictive, according to an article by the research group Advisory Board.
Recently, the White House announced that more than 1,000 pharmacy clinics across the United States had registered to participate in the initiative, according to CNN. Ordering of the drugs is underway in many of these clinics, a White House official told the network.
Besides retail clinics in chain pharmacies, the antivirals will also be available in community health centers, long-term-care facilities, and Veterans Health Administration clinics, according to a statement from the U.S. Department of Health and Human Services.
The two antiviral pills authorized by the U.S. Food and Drug Administration include Pfizer’s Paxlovid, for people 12 and older, and Merck’s molnupiravir, for adults. Either drug has to be taken within 5 days after symptoms appear to be effective in preventing serious illness.
The need for speed is a major reason why the government chose to work with retail clinics that are more accessible than most primary care offices. However, the American Medical Association (AMA), the National Community Pharmacists Association (NCPA), and the American Pharmacists Association (APhA) have publicly criticized the administration’s approach.
The pharmacists’ groups are concerned that the program is limited only to pharmacies with clinics on site, thus restricting the number of pharmacies qualified to participate. Fourteen pharmacy groups, including the NCPA and the APhA, have also sent a letter to the Biden administration urging it to remove barriers to pharmacies ordering the medications.
The groups also want permission as “clinically trained medication experts” to prescribe the drugs and ensure their safe use.
The AMA on March 4 took issue with the prescribing component, saying that “the pharmacy-based clinic component of the test-to-treat plan flouts patient safety and risks significant negative health outcomes.”
In the AMA’s view, prescribing Paxlovid without a patient’s physician being present poses a risk for adverse drug interactions, as neither the nurse practitioners in retail clinics nor the pharmacists who dispense the drug have full knowledge of a patient›s medical history.
The next day, the AMA released another statement, saying it was reassured by comments from administration officials “that patients who have access to a regular source of care should contact their physician shortly after testing positive for COVID-19 to assess their treatment options.”
“Traditional doctor-only approach”
Having patients call their doctors after testing positive for COVID in a pharmacy “strikes me as unnecessary in the vast majority of cases, and it will delay treatment,” Robert Wachter, MD, professor and chair of the department of medicine at the University of California San Francisco, said in an interview. “In this case, it seems like the AMA is taking a very traditional doctor-only approach. And the world has changed. It’s much more of a team sport than an individual sport, the way it was years ago.”
Dr. Wachter said he has the utmost respect for pharmacists’ ability to screen prescriptions for adverse drug interactions. “We’re required to do medication reconciliation when patients see us,” he says. “And in many hospitals, we delegate that to pharmacists. They’re at least as good at it if not better than physicians are.”
While it’s essential to know what other medications a patient is taking, he noted, pharmacies have computer records of all the prescriptions they’ve filled for patients. In addition, pharmacies have access to complete medication histories through Surescripts, the company that enables electronic prescribing transactions between prescribers and pharmacies.
Drug interactions “not trivial”
Preeti Malani, MD, the chief health officer and a professor of medicine in the division of infectious diseases at the University of Michigan in Ann Arbor, told this news organization that the potential interactions between Paxlovid and some other medications are “not trivial.”
However, she said, “The really dangerous drugs are the ones for people who have had organ transplants and the like. Those aren’t individuals who are going to shop at a pharmacy.”
Besides the antirejection drugs, Dr. Wachter said, there can be serious interactions with cholesterol-lowering medications. If a person is taking Lipitor, for instance, “Someone would have to make the decision on whether it’s ok for me to stop it for a while, or to lower the dose. But I trust the pharmacist to do that as well as anybody.”
Except for these potential drug interactions with Paxlovid, the antiviral medications are “quite safe,” he said, adding that being able to treat people who test positive for COVID-19 right away is a big advantage of the test-to-treat program, considering how difficult it is for many people to get access to a doctor. That delay could mean that the antivirals are not prescribed and taken until they are no longer effective.
Both Dr. Wachter and Dr. Malani said that the widespread distribution of pharmacies and their extended hours are other big pluses, especially for people who can’t easily leave work or travel far to visit a physician.
Dr. Malani cautioned that there are still kinks to work out in the test-to-treat program. It will be a while before the retail clinics all have the antiviral drugs, and many pharmacies don’t have clinics on site.
Still, she said people can still go to their physicians to be tested, and presumably those doctors can also write antiviral prescriptions. But it’s not clear where the antivirals will be available in the near term.
“Right now, we’re playing catch-up,” Dr. Malani said. “But pharmacies are an important piece of the puzzle.”
Looking at the big picture, she said, “We know that neither vaccination nor natural infection provides long lasting immunity, and so there will be a role for antivirals in order to make this a manageable illness. And when you’re talking about millions of cases, as we were having a few months ago, the health system can’t field all those patients. So we do need a system where I can go to a pharmacy and get a test and treatment.”
A version of this article first appeared on Medscape.com.
The Biden administration’s new test-to-treat program is simple on the surface: if you feel like you may have COVID-19, go to a pharmacy, get tested, and, if positive, get treated with an antiviral medication on the spot.
One large physicians’ group is concerned that the program leaves doctors on the margins, and may put patients at risk if there are adverse effects from the medications. Pharmacists groups, on the other hand, say the program is too restrictive, according to an article by the research group Advisory Board.
Recently, the White House announced that more than 1,000 pharmacy clinics across the United States had registered to participate in the initiative, according to CNN. Ordering of the drugs is underway in many of these clinics, a White House official told the network.
Besides retail clinics in chain pharmacies, the antivirals will also be available in community health centers, long-term-care facilities, and Veterans Health Administration clinics, according to a statement from the U.S. Department of Health and Human Services.
The two antiviral pills authorized by the U.S. Food and Drug Administration include Pfizer’s Paxlovid, for people 12 and older, and Merck’s molnupiravir, for adults. Either drug has to be taken within 5 days after symptoms appear to be effective in preventing serious illness.
The need for speed is a major reason why the government chose to work with retail clinics that are more accessible than most primary care offices. However, the American Medical Association (AMA), the National Community Pharmacists Association (NCPA), and the American Pharmacists Association (APhA) have publicly criticized the administration’s approach.
The pharmacists’ groups are concerned that the program is limited only to pharmacies with clinics on site, thus restricting the number of pharmacies qualified to participate. Fourteen pharmacy groups, including the NCPA and the APhA, have also sent a letter to the Biden administration urging it to remove barriers to pharmacies ordering the medications.
The groups also want permission as “clinically trained medication experts” to prescribe the drugs and ensure their safe use.
The AMA on March 4 took issue with the prescribing component, saying that “the pharmacy-based clinic component of the test-to-treat plan flouts patient safety and risks significant negative health outcomes.”
In the AMA’s view, prescribing Paxlovid without a patient’s physician being present poses a risk for adverse drug interactions, as neither the nurse practitioners in retail clinics nor the pharmacists who dispense the drug have full knowledge of a patient›s medical history.
The next day, the AMA released another statement, saying it was reassured by comments from administration officials “that patients who have access to a regular source of care should contact their physician shortly after testing positive for COVID-19 to assess their treatment options.”
“Traditional doctor-only approach”
Having patients call their doctors after testing positive for COVID in a pharmacy “strikes me as unnecessary in the vast majority of cases, and it will delay treatment,” Robert Wachter, MD, professor and chair of the department of medicine at the University of California San Francisco, said in an interview. “In this case, it seems like the AMA is taking a very traditional doctor-only approach. And the world has changed. It’s much more of a team sport than an individual sport, the way it was years ago.”
Dr. Wachter said he has the utmost respect for pharmacists’ ability to screen prescriptions for adverse drug interactions. “We’re required to do medication reconciliation when patients see us,” he says. “And in many hospitals, we delegate that to pharmacists. They’re at least as good at it if not better than physicians are.”
While it’s essential to know what other medications a patient is taking, he noted, pharmacies have computer records of all the prescriptions they’ve filled for patients. In addition, pharmacies have access to complete medication histories through Surescripts, the company that enables electronic prescribing transactions between prescribers and pharmacies.
Drug interactions “not trivial”
Preeti Malani, MD, the chief health officer and a professor of medicine in the division of infectious diseases at the University of Michigan in Ann Arbor, told this news organization that the potential interactions between Paxlovid and some other medications are “not trivial.”
However, she said, “The really dangerous drugs are the ones for people who have had organ transplants and the like. Those aren’t individuals who are going to shop at a pharmacy.”
Besides the antirejection drugs, Dr. Wachter said, there can be serious interactions with cholesterol-lowering medications. If a person is taking Lipitor, for instance, “Someone would have to make the decision on whether it’s ok for me to stop it for a while, or to lower the dose. But I trust the pharmacist to do that as well as anybody.”
Except for these potential drug interactions with Paxlovid, the antiviral medications are “quite safe,” he said, adding that being able to treat people who test positive for COVID-19 right away is a big advantage of the test-to-treat program, considering how difficult it is for many people to get access to a doctor. That delay could mean that the antivirals are not prescribed and taken until they are no longer effective.
Both Dr. Wachter and Dr. Malani said that the widespread distribution of pharmacies and their extended hours are other big pluses, especially for people who can’t easily leave work or travel far to visit a physician.
Dr. Malani cautioned that there are still kinks to work out in the test-to-treat program. It will be a while before the retail clinics all have the antiviral drugs, and many pharmacies don’t have clinics on site.
Still, she said people can still go to their physicians to be tested, and presumably those doctors can also write antiviral prescriptions. But it’s not clear where the antivirals will be available in the near term.
“Right now, we’re playing catch-up,” Dr. Malani said. “But pharmacies are an important piece of the puzzle.”
Looking at the big picture, she said, “We know that neither vaccination nor natural infection provides long lasting immunity, and so there will be a role for antivirals in order to make this a manageable illness. And when you’re talking about millions of cases, as we were having a few months ago, the health system can’t field all those patients. So we do need a system where I can go to a pharmacy and get a test and treatment.”
A version of this article first appeared on Medscape.com.
The Biden administration’s new test-to-treat program is simple on the surface: if you feel like you may have COVID-19, go to a pharmacy, get tested, and, if positive, get treated with an antiviral medication on the spot.
One large physicians’ group is concerned that the program leaves doctors on the margins, and may put patients at risk if there are adverse effects from the medications. Pharmacists groups, on the other hand, say the program is too restrictive, according to an article by the research group Advisory Board.
Recently, the White House announced that more than 1,000 pharmacy clinics across the United States had registered to participate in the initiative, according to CNN. Ordering of the drugs is underway in many of these clinics, a White House official told the network.
Besides retail clinics in chain pharmacies, the antivirals will also be available in community health centers, long-term-care facilities, and Veterans Health Administration clinics, according to a statement from the U.S. Department of Health and Human Services.
The two antiviral pills authorized by the U.S. Food and Drug Administration include Pfizer’s Paxlovid, for people 12 and older, and Merck’s molnupiravir, for adults. Either drug has to be taken within 5 days after symptoms appear to be effective in preventing serious illness.
The need for speed is a major reason why the government chose to work with retail clinics that are more accessible than most primary care offices. However, the American Medical Association (AMA), the National Community Pharmacists Association (NCPA), and the American Pharmacists Association (APhA) have publicly criticized the administration’s approach.
The pharmacists’ groups are concerned that the program is limited only to pharmacies with clinics on site, thus restricting the number of pharmacies qualified to participate. Fourteen pharmacy groups, including the NCPA and the APhA, have also sent a letter to the Biden administration urging it to remove barriers to pharmacies ordering the medications.
The groups also want permission as “clinically trained medication experts” to prescribe the drugs and ensure their safe use.
The AMA on March 4 took issue with the prescribing component, saying that “the pharmacy-based clinic component of the test-to-treat plan flouts patient safety and risks significant negative health outcomes.”
In the AMA’s view, prescribing Paxlovid without a patient’s physician being present poses a risk for adverse drug interactions, as neither the nurse practitioners in retail clinics nor the pharmacists who dispense the drug have full knowledge of a patient›s medical history.
The next day, the AMA released another statement, saying it was reassured by comments from administration officials “that patients who have access to a regular source of care should contact their physician shortly after testing positive for COVID-19 to assess their treatment options.”
“Traditional doctor-only approach”
Having patients call their doctors after testing positive for COVID in a pharmacy “strikes me as unnecessary in the vast majority of cases, and it will delay treatment,” Robert Wachter, MD, professor and chair of the department of medicine at the University of California San Francisco, said in an interview. “In this case, it seems like the AMA is taking a very traditional doctor-only approach. And the world has changed. It’s much more of a team sport than an individual sport, the way it was years ago.”
Dr. Wachter said he has the utmost respect for pharmacists’ ability to screen prescriptions for adverse drug interactions. “We’re required to do medication reconciliation when patients see us,” he says. “And in many hospitals, we delegate that to pharmacists. They’re at least as good at it if not better than physicians are.”
While it’s essential to know what other medications a patient is taking, he noted, pharmacies have computer records of all the prescriptions they’ve filled for patients. In addition, pharmacies have access to complete medication histories through Surescripts, the company that enables electronic prescribing transactions between prescribers and pharmacies.
Drug interactions “not trivial”
Preeti Malani, MD, the chief health officer and a professor of medicine in the division of infectious diseases at the University of Michigan in Ann Arbor, told this news organization that the potential interactions between Paxlovid and some other medications are “not trivial.”
However, she said, “The really dangerous drugs are the ones for people who have had organ transplants and the like. Those aren’t individuals who are going to shop at a pharmacy.”
Besides the antirejection drugs, Dr. Wachter said, there can be serious interactions with cholesterol-lowering medications. If a person is taking Lipitor, for instance, “Someone would have to make the decision on whether it’s ok for me to stop it for a while, or to lower the dose. But I trust the pharmacist to do that as well as anybody.”
Except for these potential drug interactions with Paxlovid, the antiviral medications are “quite safe,” he said, adding that being able to treat people who test positive for COVID-19 right away is a big advantage of the test-to-treat program, considering how difficult it is for many people to get access to a doctor. That delay could mean that the antivirals are not prescribed and taken until they are no longer effective.
Both Dr. Wachter and Dr. Malani said that the widespread distribution of pharmacies and their extended hours are other big pluses, especially for people who can’t easily leave work or travel far to visit a physician.
Dr. Malani cautioned that there are still kinks to work out in the test-to-treat program. It will be a while before the retail clinics all have the antiviral drugs, and many pharmacies don’t have clinics on site.
Still, she said people can still go to their physicians to be tested, and presumably those doctors can also write antiviral prescriptions. But it’s not clear where the antivirals will be available in the near term.
“Right now, we’re playing catch-up,” Dr. Malani said. “But pharmacies are an important piece of the puzzle.”
Looking at the big picture, she said, “We know that neither vaccination nor natural infection provides long lasting immunity, and so there will be a role for antivirals in order to make this a manageable illness. And when you’re talking about millions of cases, as we were having a few months ago, the health system can’t field all those patients. So we do need a system where I can go to a pharmacy and get a test and treatment.”
A version of this article first appeared on Medscape.com.
Pharma should stop doing business in Russia, says ethicist
Should pharmaceutical companies continue to do business in Russia, running ongoing clinical trials, starting new ones, or continuing to sell their products there?
Some argue that medicine and science must not get enmeshed in politics, staying above the fray to protect their independence and credibility. Other defenders of business-as-usual say the pharmaceutical industry deals in health and aids the vulnerable. Humanitarianism requires continued interaction with Russia.
I think both arguments fail.
We are fighting a war with Russia. It is a war of economic strangulation, social isolation, and pushing Russia as hard as we can to become a pariah state so that internal pressure on Putin will cause him to rethink his cruel, unjustified invasion or the Russian people to replace him. This pressure must be harsh and it must happen quickly. Why?
Having failed to rapidly defeat the Ukrainian army in the war’s first weeks, Russian commanders are now resorting to the horrible barbarism they used in previous wars in Chechnya and Syria: flattening cities, attacking civilians, killing children with massive and indiscriminate firepower.
To mention one recent horror among many, Russian shelling destroyed a maternity hospital in Mariupol. Ukraine’s president, Volodymyr Zelensky, in bemoaning the Russians for their continuing series of war crimes called on the world to act.
“Mariupol. Direct Strike of Russian troops at the maternity hospital,” he wrote in a Twitter post. “People, children are under the wreckage. Atrocity! How much longer will the world be an accomplice ignoring terror?”
The Russian government’s response: “It is not the first time we have seen pathetic outcries concerning the so-called atrocities,” said Minister of Foreign Affairs Sergei Lavrov, claiming the hospital was being used as a base by an “ultra-radical” Ukrainian battalion.
Health and its preservation are key parts of the aim of medicine and science. There is no way that medicine and science can ignore what war does to health, what attacks on hospitals do to the sick and those who serve them there, the psychological toll that intentional terrorism takes on civilians and their defenders, and what the destruction of infrastructure means for the long-term well-being of Ukrainians.
There can be no collusion with war criminals. There can be no denial of the inextricable link between medicine, science, and politics. Medicine and science are controlled by political forces; their use for good or evil is driven by political considerations, and each doctor, scientist, and scientific society must take a stand when politics corrodes the underlying aims of research and healing.
How far does noncooperation with Russia go? Very, very far. All research, both ongoing and new, must cease immediately. Whatever can be done to minimize harm to existing subjects in a short period of time ought to be done, but that is it.
Similarly, no sale of medicines or therapies ought to be occurring, be they life-saving or consumer products. Putin will see to it that such shipments go to the military or are sold on the black market for revenue, and there is nothing pharma companies can do to stop that.
The Russian people need to be pinched not only by the loss of cheeseburgers and boutique coffee but by products they use to maintain their well-being. War is cruel that way, but if you tolerate a government that is bombing and shelling a peaceful neighbor to oblivion, then pharma must ensure that efforts to make Putin and his kleptocratic goons feel the wrath of their fellow citizens.
Given the realities of nuclear Armageddon, the civilized world must fight obvious barbarity as best it can with sanctions, financial assaults, property seizures, and forgoing commerce, including important raw materials and health products. War, even in a fiscal form, is not without terrible costs; but achieving a rapid, just resolution against tyranny permits no exceptions for pharma or any other business if it is a war that must be fought.
Dr. Caplan is director of the division of medical ethics at New York University. He has consulted with Johnson & Johnson’s Panel for Compassionate Drug Use.
A version of this article first appeared on Medscape.com.
Should pharmaceutical companies continue to do business in Russia, running ongoing clinical trials, starting new ones, or continuing to sell their products there?
Some argue that medicine and science must not get enmeshed in politics, staying above the fray to protect their independence and credibility. Other defenders of business-as-usual say the pharmaceutical industry deals in health and aids the vulnerable. Humanitarianism requires continued interaction with Russia.
I think both arguments fail.
We are fighting a war with Russia. It is a war of economic strangulation, social isolation, and pushing Russia as hard as we can to become a pariah state so that internal pressure on Putin will cause him to rethink his cruel, unjustified invasion or the Russian people to replace him. This pressure must be harsh and it must happen quickly. Why?
Having failed to rapidly defeat the Ukrainian army in the war’s first weeks, Russian commanders are now resorting to the horrible barbarism they used in previous wars in Chechnya and Syria: flattening cities, attacking civilians, killing children with massive and indiscriminate firepower.
To mention one recent horror among many, Russian shelling destroyed a maternity hospital in Mariupol. Ukraine’s president, Volodymyr Zelensky, in bemoaning the Russians for their continuing series of war crimes called on the world to act.
“Mariupol. Direct Strike of Russian troops at the maternity hospital,” he wrote in a Twitter post. “People, children are under the wreckage. Atrocity! How much longer will the world be an accomplice ignoring terror?”
The Russian government’s response: “It is not the first time we have seen pathetic outcries concerning the so-called atrocities,” said Minister of Foreign Affairs Sergei Lavrov, claiming the hospital was being used as a base by an “ultra-radical” Ukrainian battalion.
Health and its preservation are key parts of the aim of medicine and science. There is no way that medicine and science can ignore what war does to health, what attacks on hospitals do to the sick and those who serve them there, the psychological toll that intentional terrorism takes on civilians and their defenders, and what the destruction of infrastructure means for the long-term well-being of Ukrainians.
There can be no collusion with war criminals. There can be no denial of the inextricable link between medicine, science, and politics. Medicine and science are controlled by political forces; their use for good or evil is driven by political considerations, and each doctor, scientist, and scientific society must take a stand when politics corrodes the underlying aims of research and healing.
How far does noncooperation with Russia go? Very, very far. All research, both ongoing and new, must cease immediately. Whatever can be done to minimize harm to existing subjects in a short period of time ought to be done, but that is it.
Similarly, no sale of medicines or therapies ought to be occurring, be they life-saving or consumer products. Putin will see to it that such shipments go to the military or are sold on the black market for revenue, and there is nothing pharma companies can do to stop that.
The Russian people need to be pinched not only by the loss of cheeseburgers and boutique coffee but by products they use to maintain their well-being. War is cruel that way, but if you tolerate a government that is bombing and shelling a peaceful neighbor to oblivion, then pharma must ensure that efforts to make Putin and his kleptocratic goons feel the wrath of their fellow citizens.
Given the realities of nuclear Armageddon, the civilized world must fight obvious barbarity as best it can with sanctions, financial assaults, property seizures, and forgoing commerce, including important raw materials and health products. War, even in a fiscal form, is not without terrible costs; but achieving a rapid, just resolution against tyranny permits no exceptions for pharma or any other business if it is a war that must be fought.
Dr. Caplan is director of the division of medical ethics at New York University. He has consulted with Johnson & Johnson’s Panel for Compassionate Drug Use.
A version of this article first appeared on Medscape.com.
Should pharmaceutical companies continue to do business in Russia, running ongoing clinical trials, starting new ones, or continuing to sell their products there?
Some argue that medicine and science must not get enmeshed in politics, staying above the fray to protect their independence and credibility. Other defenders of business-as-usual say the pharmaceutical industry deals in health and aids the vulnerable. Humanitarianism requires continued interaction with Russia.
I think both arguments fail.
We are fighting a war with Russia. It is a war of economic strangulation, social isolation, and pushing Russia as hard as we can to become a pariah state so that internal pressure on Putin will cause him to rethink his cruel, unjustified invasion or the Russian people to replace him. This pressure must be harsh and it must happen quickly. Why?
Having failed to rapidly defeat the Ukrainian army in the war’s first weeks, Russian commanders are now resorting to the horrible barbarism they used in previous wars in Chechnya and Syria: flattening cities, attacking civilians, killing children with massive and indiscriminate firepower.
To mention one recent horror among many, Russian shelling destroyed a maternity hospital in Mariupol. Ukraine’s president, Volodymyr Zelensky, in bemoaning the Russians for their continuing series of war crimes called on the world to act.
“Mariupol. Direct Strike of Russian troops at the maternity hospital,” he wrote in a Twitter post. “People, children are under the wreckage. Atrocity! How much longer will the world be an accomplice ignoring terror?”
The Russian government’s response: “It is not the first time we have seen pathetic outcries concerning the so-called atrocities,” said Minister of Foreign Affairs Sergei Lavrov, claiming the hospital was being used as a base by an “ultra-radical” Ukrainian battalion.
Health and its preservation are key parts of the aim of medicine and science. There is no way that medicine and science can ignore what war does to health, what attacks on hospitals do to the sick and those who serve them there, the psychological toll that intentional terrorism takes on civilians and their defenders, and what the destruction of infrastructure means for the long-term well-being of Ukrainians.
There can be no collusion with war criminals. There can be no denial of the inextricable link between medicine, science, and politics. Medicine and science are controlled by political forces; their use for good or evil is driven by political considerations, and each doctor, scientist, and scientific society must take a stand when politics corrodes the underlying aims of research and healing.
How far does noncooperation with Russia go? Very, very far. All research, both ongoing and new, must cease immediately. Whatever can be done to minimize harm to existing subjects in a short period of time ought to be done, but that is it.
Similarly, no sale of medicines or therapies ought to be occurring, be they life-saving or consumer products. Putin will see to it that such shipments go to the military or are sold on the black market for revenue, and there is nothing pharma companies can do to stop that.
The Russian people need to be pinched not only by the loss of cheeseburgers and boutique coffee but by products they use to maintain their well-being. War is cruel that way, but if you tolerate a government that is bombing and shelling a peaceful neighbor to oblivion, then pharma must ensure that efforts to make Putin and his kleptocratic goons feel the wrath of their fellow citizens.
Given the realities of nuclear Armageddon, the civilized world must fight obvious barbarity as best it can with sanctions, financial assaults, property seizures, and forgoing commerce, including important raw materials and health products. War, even in a fiscal form, is not without terrible costs; but achieving a rapid, just resolution against tyranny permits no exceptions for pharma or any other business if it is a war that must be fought.
Dr. Caplan is director of the division of medical ethics at New York University. He has consulted with Johnson & Johnson’s Panel for Compassionate Drug Use.
A version of this article first appeared on Medscape.com.