User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Should There Be a Mandatory Retirement Age for Physicians?
This transcript has been edited for clarity.
I’d like to pose a question: When should doctors retire? When, as practicing physicians or surgeons, do we become too old to deliver competent service?
You will be amazed to hear, those of you who have listened to my videos before — and although it is a matter of public knowledge — that I’m 68. I know it’s impossible to imagine, due to this youthful appearance, visage, and so on, but I am. I’ve been a cancer doctor for 40 years; therefore, I need to think a little about retirement.
There are two elements of this for me. I’m a university professor, and in Oxford we did vote, as a democracy of scholars, to have a mandatory retirement age around 68. This is so that we can bring new blood forward so that we can create the space to promote new professors, to bring youngsters in to make new ideas, and to get rid of us fusty old lot.
The other argument would be, of course, that we are wise, we’re experienced, we are world-weary, and we’re successful — otherwise, we wouldn’t have lasted as academics as long. Nevertheless, we voted to do that.
It’s possible to have a discussion with the university to extend this, and for those of us who are clinical academics, I have an honorary appointment as a consultant cancer physician in the hospital and my university professorial appointment, too.
I can extend it probably until I’m about 70. It feels like a nice, round number at which to retire — somewhat arbitrarily, one would admit. But does that feel right?
In the United States, more than 25% of the physician workforce is over the age of 65. There are many studies showing that there is a 20% cognitive decline for most individuals between the ages of 45 and 65.
Are we as capable as an elderly workforce as once we were? Clearly, it’s hardly individualistic. It depends on each of our own health status, where we started from, and so on, but are there any general rules that we can apply? I think these are starting to creep in around the sense of revalidation.
In the United Kingdom, we have a General Medical Council (GMC). I need to have a license to practice from the GMC and a sense of fitness to practice. I have annual appraisals within the hospital system, in which I explore delivery of care, how I’m doing as a mentor, am I reaching the milestones I’ve set in terms of academic achievements, and so on.
This is a peer-to-peer process. We have senior physicians — people like myself — who act as appraisers to support our colleagues and to maintain that sense of fitness to practice. Every 5 years, I’m revalidated by the GMC. They take account of the annual appraisals and a report made by the senior physician within my hospital network who’s a so-called designated person.
These two elements come together with patient feedback, with 360-degree feedback from colleagues, and so on. This is quite a firmly regulated system that I think works. Our mandatory retirement age of 65 has gone. That was phased out by the government. In fact, our NHS is making an effort to retain older elders in the workforce.
They see the benefits of mentorship, experience, leadership, and networks. At a time when the majority of NHS are actively seeking to retire when 65, the NHS is trying to retain and pull back those of us who have been around for that wee bit longer and who still feel committed to doing it.
I’d be really interested to see what you think. There’s variation from country to country. I know that, in Australia, they’re talking about annual appraisals of doctors over the age of 70. I’d be very interested to hear what you think is likely to happen in the United States.
I think our system works pretty well, as long as you’re within the NHS and hospital system. If you wanted to still practice, but practice privately, you would still have to find somebody who’d be prepared to conduct appraisals and so on outside of the NHS. It’s an interesting area.
For myself, I still feel competent. Patients seem to like me. That’s an objective assessment by this 360-degree thing in which patients reflected very positively, indeed, in my approach to the delivery of the care and so on, as did colleagues. I’m still publishing, I go to meetings, I cheer things, bits and bobs. I’d say I’m a wee bit unusual in terms of still having a strong academic profile in doing stuff.
It’s an interesting question. Richard Doll, one of the world’s great epidemiologists who, of course, was the dominant discoverer of the link between smoking and lung cancer, was attending seminars, sitting in the front row, and coming into university 3 days a week at age 90, continuing to be contributory with his extraordinarily sharp intellect and vast, vast experience.
When I think of experience, all young cancer doctors are now immunologists. When I was a young doctor, I was a clinical pharmacologist. There are many lessons and tricks that I learned which I do need to pass on to the younger generation of today. What do you think? Should there be a mandatory retirement age? How do we best measure, assess, and revalidate elderly physicians and surgeons? How can we continue to contribute to those who choose to do so? For the time being, as always, thanks for listening.
Dr. Kerr is professor, Nuffield Department of Clinical Laboratory Science, University of Oxford, and professor of cancer medicine, Oxford Cancer Centre, Oxford, United Kingdom. He has disclosed ties with Celleron Therapeutics, Oxford Cancer Biomarkers (Board of Directors); Afrox (charity; Trustee); GlaxoSmithKline and Bayer HealthCare Pharmaceuticals (Consultant), Genomic Health; Merck Serono, and Roche.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I’d like to pose a question: When should doctors retire? When, as practicing physicians or surgeons, do we become too old to deliver competent service?
You will be amazed to hear, those of you who have listened to my videos before — and although it is a matter of public knowledge — that I’m 68. I know it’s impossible to imagine, due to this youthful appearance, visage, and so on, but I am. I’ve been a cancer doctor for 40 years; therefore, I need to think a little about retirement.
There are two elements of this for me. I’m a university professor, and in Oxford we did vote, as a democracy of scholars, to have a mandatory retirement age around 68. This is so that we can bring new blood forward so that we can create the space to promote new professors, to bring youngsters in to make new ideas, and to get rid of us fusty old lot.
The other argument would be, of course, that we are wise, we’re experienced, we are world-weary, and we’re successful — otherwise, we wouldn’t have lasted as academics as long. Nevertheless, we voted to do that.
It’s possible to have a discussion with the university to extend this, and for those of us who are clinical academics, I have an honorary appointment as a consultant cancer physician in the hospital and my university professorial appointment, too.
I can extend it probably until I’m about 70. It feels like a nice, round number at which to retire — somewhat arbitrarily, one would admit. But does that feel right?
In the United States, more than 25% of the physician workforce is over the age of 65. There are many studies showing that there is a 20% cognitive decline for most individuals between the ages of 45 and 65.
Are we as capable as an elderly workforce as once we were? Clearly, it’s hardly individualistic. It depends on each of our own health status, where we started from, and so on, but are there any general rules that we can apply? I think these are starting to creep in around the sense of revalidation.
In the United Kingdom, we have a General Medical Council (GMC). I need to have a license to practice from the GMC and a sense of fitness to practice. I have annual appraisals within the hospital system, in which I explore delivery of care, how I’m doing as a mentor, am I reaching the milestones I’ve set in terms of academic achievements, and so on.
This is a peer-to-peer process. We have senior physicians — people like myself — who act as appraisers to support our colleagues and to maintain that sense of fitness to practice. Every 5 years, I’m revalidated by the GMC. They take account of the annual appraisals and a report made by the senior physician within my hospital network who’s a so-called designated person.
These two elements come together with patient feedback, with 360-degree feedback from colleagues, and so on. This is quite a firmly regulated system that I think works. Our mandatory retirement age of 65 has gone. That was phased out by the government. In fact, our NHS is making an effort to retain older elders in the workforce.
They see the benefits of mentorship, experience, leadership, and networks. At a time when the majority of NHS are actively seeking to retire when 65, the NHS is trying to retain and pull back those of us who have been around for that wee bit longer and who still feel committed to doing it.
I’d be really interested to see what you think. There’s variation from country to country. I know that, in Australia, they’re talking about annual appraisals of doctors over the age of 70. I’d be very interested to hear what you think is likely to happen in the United States.
I think our system works pretty well, as long as you’re within the NHS and hospital system. If you wanted to still practice, but practice privately, you would still have to find somebody who’d be prepared to conduct appraisals and so on outside of the NHS. It’s an interesting area.
For myself, I still feel competent. Patients seem to like me. That’s an objective assessment by this 360-degree thing in which patients reflected very positively, indeed, in my approach to the delivery of the care and so on, as did colleagues. I’m still publishing, I go to meetings, I cheer things, bits and bobs. I’d say I’m a wee bit unusual in terms of still having a strong academic profile in doing stuff.
It’s an interesting question. Richard Doll, one of the world’s great epidemiologists who, of course, was the dominant discoverer of the link between smoking and lung cancer, was attending seminars, sitting in the front row, and coming into university 3 days a week at age 90, continuing to be contributory with his extraordinarily sharp intellect and vast, vast experience.
When I think of experience, all young cancer doctors are now immunologists. When I was a young doctor, I was a clinical pharmacologist. There are many lessons and tricks that I learned which I do need to pass on to the younger generation of today. What do you think? Should there be a mandatory retirement age? How do we best measure, assess, and revalidate elderly physicians and surgeons? How can we continue to contribute to those who choose to do so? For the time being, as always, thanks for listening.
Dr. Kerr is professor, Nuffield Department of Clinical Laboratory Science, University of Oxford, and professor of cancer medicine, Oxford Cancer Centre, Oxford, United Kingdom. He has disclosed ties with Celleron Therapeutics, Oxford Cancer Biomarkers (Board of Directors); Afrox (charity; Trustee); GlaxoSmithKline and Bayer HealthCare Pharmaceuticals (Consultant), Genomic Health; Merck Serono, and Roche.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I’d like to pose a question: When should doctors retire? When, as practicing physicians or surgeons, do we become too old to deliver competent service?
You will be amazed to hear, those of you who have listened to my videos before — and although it is a matter of public knowledge — that I’m 68. I know it’s impossible to imagine, due to this youthful appearance, visage, and so on, but I am. I’ve been a cancer doctor for 40 years; therefore, I need to think a little about retirement.
There are two elements of this for me. I’m a university professor, and in Oxford we did vote, as a democracy of scholars, to have a mandatory retirement age around 68. This is so that we can bring new blood forward so that we can create the space to promote new professors, to bring youngsters in to make new ideas, and to get rid of us fusty old lot.
The other argument would be, of course, that we are wise, we’re experienced, we are world-weary, and we’re successful — otherwise, we wouldn’t have lasted as academics as long. Nevertheless, we voted to do that.
It’s possible to have a discussion with the university to extend this, and for those of us who are clinical academics, I have an honorary appointment as a consultant cancer physician in the hospital and my university professorial appointment, too.
I can extend it probably until I’m about 70. It feels like a nice, round number at which to retire — somewhat arbitrarily, one would admit. But does that feel right?
In the United States, more than 25% of the physician workforce is over the age of 65. There are many studies showing that there is a 20% cognitive decline for most individuals between the ages of 45 and 65.
Are we as capable as an elderly workforce as once we were? Clearly, it’s hardly individualistic. It depends on each of our own health status, where we started from, and so on, but are there any general rules that we can apply? I think these are starting to creep in around the sense of revalidation.
In the United Kingdom, we have a General Medical Council (GMC). I need to have a license to practice from the GMC and a sense of fitness to practice. I have annual appraisals within the hospital system, in which I explore delivery of care, how I’m doing as a mentor, am I reaching the milestones I’ve set in terms of academic achievements, and so on.
This is a peer-to-peer process. We have senior physicians — people like myself — who act as appraisers to support our colleagues and to maintain that sense of fitness to practice. Every 5 years, I’m revalidated by the GMC. They take account of the annual appraisals and a report made by the senior physician within my hospital network who’s a so-called designated person.
These two elements come together with patient feedback, with 360-degree feedback from colleagues, and so on. This is quite a firmly regulated system that I think works. Our mandatory retirement age of 65 has gone. That was phased out by the government. In fact, our NHS is making an effort to retain older elders in the workforce.
They see the benefits of mentorship, experience, leadership, and networks. At a time when the majority of NHS are actively seeking to retire when 65, the NHS is trying to retain and pull back those of us who have been around for that wee bit longer and who still feel committed to doing it.
I’d be really interested to see what you think. There’s variation from country to country. I know that, in Australia, they’re talking about annual appraisals of doctors over the age of 70. I’d be very interested to hear what you think is likely to happen in the United States.
I think our system works pretty well, as long as you’re within the NHS and hospital system. If you wanted to still practice, but practice privately, you would still have to find somebody who’d be prepared to conduct appraisals and so on outside of the NHS. It’s an interesting area.
For myself, I still feel competent. Patients seem to like me. That’s an objective assessment by this 360-degree thing in which patients reflected very positively, indeed, in my approach to the delivery of the care and so on, as did colleagues. I’m still publishing, I go to meetings, I cheer things, bits and bobs. I’d say I’m a wee bit unusual in terms of still having a strong academic profile in doing stuff.
It’s an interesting question. Richard Doll, one of the world’s great epidemiologists who, of course, was the dominant discoverer of the link between smoking and lung cancer, was attending seminars, sitting in the front row, and coming into university 3 days a week at age 90, continuing to be contributory with his extraordinarily sharp intellect and vast, vast experience.
When I think of experience, all young cancer doctors are now immunologists. When I was a young doctor, I was a clinical pharmacologist. There are many lessons and tricks that I learned which I do need to pass on to the younger generation of today. What do you think? Should there be a mandatory retirement age? How do we best measure, assess, and revalidate elderly physicians and surgeons? How can we continue to contribute to those who choose to do so? For the time being, as always, thanks for listening.
Dr. Kerr is professor, Nuffield Department of Clinical Laboratory Science, University of Oxford, and professor of cancer medicine, Oxford Cancer Centre, Oxford, United Kingdom. He has disclosed ties with Celleron Therapeutics, Oxford Cancer Biomarkers (Board of Directors); Afrox (charity; Trustee); GlaxoSmithKline and Bayer HealthCare Pharmaceuticals (Consultant), Genomic Health; Merck Serono, and Roche.
A version of this article appeared on Medscape.com.
Triptans Trump Newer, More Expensive Meds for Acute Migraine
new research suggested.
Results of a large systematic review and meta-analysis showed that eletriptan, rizatriptan, sumatriptan, and zolmitriptan were more effective than lasmiditan, rimegepant, and ubrogepant, which investigators found were as effective as nonsteroidal anti-inflammatory drugs (NSAIDs).
International guidelines generally endorse NSAIDs as the first-line treatment for migraine and recommend triptans for moderate to severe episodes or when the response to NSAIDs is insufficient.
However, based on the study’s findings, these four triptans should be considered the treatment of choice for migraine, study investigator Andrea Cipriani, MD, PhD, professor of psychiatry at the University of Oxford in England and director of the Oxford Health Clinical Research Facility, told a press briefing.
The investigators added that these particular triptans should be “included in the WHO [World Health Organization] List of Essential Medicines to promote global accessibility and uniform standards of care.”
The study was published online in The BMJ.
Filling the Knowledge Gap
To date, almost all migraine studies have compared migraine drugs with placebo, so there’s a knowledge gap, said Dr. Cipriani. As a result, “there’s no clear consensus among experts and guidelines about which specific drug classes should be prescribed initially, and this is a clinical problem.”
The researchers pointed out that, in recent years, lasmiditan and gepants have been introduced as further treatment options, especially for patients in whom triptans are contraindicated because of their potential vasoconstrictive effects and higher risk for ischemic events.
The analysis included 137 double-blind, randomized, controlled trials that were primarily sponsored by the pharmaceutical industry. It included 89,445 adult outpatients with migraine (mean age, 40.3 years; 85.6% women).
Only drugs licensed for migraine or headache that are recommended in at least one country were included. Researchers divided these 17 drugs into five categories: Antipyretics (paracetamol), ditans (lasmiditan), gepants (rimegepant and ubrogepant), NSAIDs (acetylsalicylic acid, celecoxib, diclofenac potassium, ibuprofen, naproxen sodium, and phenazone), and triptans (almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, sumatriptan, and zolmitriptan).
The study’s primary outcomes were freedom from pain at 2 hours and at 2-24 hours, without the use of rescue drugs.
Investigators used sumatriptan as the reference intervention because it is the most commonly prescribed migraine drug and is included in the WHO Model Lists of Essential Medicines.
The study showed all active interventions were better than placebo for pain freedom at 2 hours; with the exception of paracetamol and naratriptan, all were better for sustained pain freedom from 2 to 24 hours.
When the active interventions were compared with each other, eletriptan outperformed other drugs for achieving pain freedom at 2 hours. It was followed by rizatriptan, sumatriptan, and zolmitriptan (odds ratio [OR], 1.35-3.01). For sustained pain freedom up to 24 hours, the most efficacious interventions were eletriptan (OR, 1.41-2.73) and ibuprofen (OR, 3.16-4.82).
As for secondary efficacy outcomes, in head-to-head comparisons, eletriptan was superior to nearly all other active interventions for pain relief at 2 hours and for the use of rescue drugs.
As for adverse events, dizziness was more commonly associated with lasmiditan, eletriptan, sumatriptan, and zolmitriptan, while fatigue and sedation occurred more frequently with eletriptan and lasmiditan. Nausea was more often associated with lasmiditan, sumatriptan, zolmitriptan, and ubrogepant. Eletriptan was the only intervention most frequently associated with chest pain or discomfort.
Need to Update Guidelines?
Considering the new results, Dr. Cipriani and study coauthor Messoud Ashina, MD, PhD, professor of neurology, University of Copenhagen in Denmark, said clinical guidelines for acute migraine management should be updated.
While triptans are contraindicated in patients with vascular disease, the researchers noted that “cerebrovascular events may present primarily as migraine-like headaches, and misdiagnosis of transient ischemic attack and minor stroke as migraine is not rare.”
Recently, lasmiditan, rimegepant, and ubrogepant — which are not associated with vasoconstrictive effects — have been promoted as alternatives in patients for whom triptans are contraindicated or not tolerated. But the high costs of these drugs put them out of reach for some patients, the investigators noted.
Triptans are widely underutilized, Dr. Ashina noted during the press briefing. Current use ranges from 17% to 22% in the United States and from 3% to 22.5% in Europe.
The investigators said that triptans have been shown to be superior and should be promoted globally, adding that the limited access and substantial underutilization of these medications are “missed opportunities to offer more effective treatments.”
The new results underscore the importance of head-to-head trials, which is the gold standard, said Dr. Cipriani.
The investigators noted that the study’s main limitation was the quality of the data, which was deemed to be low, or very low, for most comparisons. Other potential limitations included lack of individual patient data; exclusion of combination drugs; inclusion of only oral treatments; and not considering type of oral formulation, consistency in response across migraine episodes, or cost-effectiveness.
The study also did not cover important clinical issues that might inform treatment decision-making, including drug overuse headache or potential withdrawal symptoms. And the authors were unable to quantify some outcomes, such as global functioning.
‘Best Profile’?
Reached for comment, Neurologist Nina Riggins, MD, PhD, Headache Center of Excellence, Palo Alto VA Medical Center in California, praised the authors for a “great job” of bringing attention to the topic.
However, she noted that the investigators’ characterization of the four triptans as having the “best profile” for acute migraine gave her pause.
“Calling triptans the medication with the ‘best profile’ might be not applicable in many cases,” she said. For example, those who need acute medication more than two to three times a week in addition to those with cardiovascular contraindications to triptans may fall outside of that category.
Dr. Riggins said that “it makes sense” that longer-acting triptans like frovatriptan and naratriptan may not rank highly for efficacy within the first 2 hours. However, these agents likely offer a superior therapeutic profile in specific contexts, such as menstrual-related migraine.
In addition, while triptans are known to cause medication overuse headache, this may not be the case with gepants, she noted.
In a release from the Science Media Center, a nonprofit organization promoting voices and views of the scientific community, Eloísa Rubio-Beltrán, PhD, research associate with The Migraine Trust at the Wolfson Sensory, Pain and Regeneration Centre, King’s College London in England, said the findings should affect migraine treatment guidelines.
“As the study highlights, due to their high efficacy and low cost, triptans should be the first-line treatment option for the acute treatment of migraine. These results should inform treatment guidelines and support the inclusion of the best performing triptans into the List of Essential Medicines, to optimize treatment, allowing patients to access more efficacious options,” said Dr. Rubio-Beltrán.
It is also important to note that gepants and ditans were developed to offer alternatives for patients who show no improvement from triptans, she added.
She pointed out that these medications were not developed as a substitute for triptans, but rather to expand the number of treatment options for migraine.
“Nonetheless,” she added, “this study highlights the need for further research on the pathophysiology of migraine, which will allow the discovery of novel targets, and thus, novel treatments options that will benefit all patient populations.”
The study was funded by the NIHR Oxford Health Biomedical Research Centre and the Lundbeck Foundation. Dr. Cipriani reported receiving research, educational, and consultancy fees from Italian Network for Pediatric Clinical Trials, Fondazione Cariplo, Lundbeck, and Angelini Pharma. Dr. Ashina is a consultant, speaker, or scientific adviser for AbbVie, Amgen, AstraZeneca, Eli Lilly, GSK, Lundbeck, Novartis, Pfizer, and Teva; is the past president of the International Headache Society; and an associate editor of The Journal of Headache and Pain and Brain. Dr. Riggins has consulted for Gerson Lehrman Group; participated in compensated work with AcademicCME and Association of Migraine Disorders; was a principal investigator on research with electroCore, Theranica, and Eli Lilly; serves on advisory boards for Theranica, Teva, Lundbeck, Amneal Pharmaceuticals, NeurologyLive, and Miles for Migraine; and is a project advisor for Clinical Awareness Initiative with Clinical Neurological Society of America. Dr. Rubio-Beltrán reported serving as a junior editorial board member of The Journal of Headache and Pain and a junior representative of the International Headache Society; receiving research support from The Migraine Trust, Eli Lilly, CoLucid Pharmaceuticals, Amgen, Novartis, and Kallyope; and receiving travel support from CoLucid Pharmaceuticals, Allergan, and Novartis.
A version of this article first appeared on Medscape.com.
new research suggested.
Results of a large systematic review and meta-analysis showed that eletriptan, rizatriptan, sumatriptan, and zolmitriptan were more effective than lasmiditan, rimegepant, and ubrogepant, which investigators found were as effective as nonsteroidal anti-inflammatory drugs (NSAIDs).
International guidelines generally endorse NSAIDs as the first-line treatment for migraine and recommend triptans for moderate to severe episodes or when the response to NSAIDs is insufficient.
However, based on the study’s findings, these four triptans should be considered the treatment of choice for migraine, study investigator Andrea Cipriani, MD, PhD, professor of psychiatry at the University of Oxford in England and director of the Oxford Health Clinical Research Facility, told a press briefing.
The investigators added that these particular triptans should be “included in the WHO [World Health Organization] List of Essential Medicines to promote global accessibility and uniform standards of care.”
The study was published online in The BMJ.
Filling the Knowledge Gap
To date, almost all migraine studies have compared migraine drugs with placebo, so there’s a knowledge gap, said Dr. Cipriani. As a result, “there’s no clear consensus among experts and guidelines about which specific drug classes should be prescribed initially, and this is a clinical problem.”
The researchers pointed out that, in recent years, lasmiditan and gepants have been introduced as further treatment options, especially for patients in whom triptans are contraindicated because of their potential vasoconstrictive effects and higher risk for ischemic events.
The analysis included 137 double-blind, randomized, controlled trials that were primarily sponsored by the pharmaceutical industry. It included 89,445 adult outpatients with migraine (mean age, 40.3 years; 85.6% women).
Only drugs licensed for migraine or headache that are recommended in at least one country were included. Researchers divided these 17 drugs into five categories: Antipyretics (paracetamol), ditans (lasmiditan), gepants (rimegepant and ubrogepant), NSAIDs (acetylsalicylic acid, celecoxib, diclofenac potassium, ibuprofen, naproxen sodium, and phenazone), and triptans (almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, sumatriptan, and zolmitriptan).
The study’s primary outcomes were freedom from pain at 2 hours and at 2-24 hours, without the use of rescue drugs.
Investigators used sumatriptan as the reference intervention because it is the most commonly prescribed migraine drug and is included in the WHO Model Lists of Essential Medicines.
The study showed all active interventions were better than placebo for pain freedom at 2 hours; with the exception of paracetamol and naratriptan, all were better for sustained pain freedom from 2 to 24 hours.
When the active interventions were compared with each other, eletriptan outperformed other drugs for achieving pain freedom at 2 hours. It was followed by rizatriptan, sumatriptan, and zolmitriptan (odds ratio [OR], 1.35-3.01). For sustained pain freedom up to 24 hours, the most efficacious interventions were eletriptan (OR, 1.41-2.73) and ibuprofen (OR, 3.16-4.82).
As for secondary efficacy outcomes, in head-to-head comparisons, eletriptan was superior to nearly all other active interventions for pain relief at 2 hours and for the use of rescue drugs.
As for adverse events, dizziness was more commonly associated with lasmiditan, eletriptan, sumatriptan, and zolmitriptan, while fatigue and sedation occurred more frequently with eletriptan and lasmiditan. Nausea was more often associated with lasmiditan, sumatriptan, zolmitriptan, and ubrogepant. Eletriptan was the only intervention most frequently associated with chest pain or discomfort.
Need to Update Guidelines?
Considering the new results, Dr. Cipriani and study coauthor Messoud Ashina, MD, PhD, professor of neurology, University of Copenhagen in Denmark, said clinical guidelines for acute migraine management should be updated.
While triptans are contraindicated in patients with vascular disease, the researchers noted that “cerebrovascular events may present primarily as migraine-like headaches, and misdiagnosis of transient ischemic attack and minor stroke as migraine is not rare.”
Recently, lasmiditan, rimegepant, and ubrogepant — which are not associated with vasoconstrictive effects — have been promoted as alternatives in patients for whom triptans are contraindicated or not tolerated. But the high costs of these drugs put them out of reach for some patients, the investigators noted.
Triptans are widely underutilized, Dr. Ashina noted during the press briefing. Current use ranges from 17% to 22% in the United States and from 3% to 22.5% in Europe.
The investigators said that triptans have been shown to be superior and should be promoted globally, adding that the limited access and substantial underutilization of these medications are “missed opportunities to offer more effective treatments.”
The new results underscore the importance of head-to-head trials, which is the gold standard, said Dr. Cipriani.
The investigators noted that the study’s main limitation was the quality of the data, which was deemed to be low, or very low, for most comparisons. Other potential limitations included lack of individual patient data; exclusion of combination drugs; inclusion of only oral treatments; and not considering type of oral formulation, consistency in response across migraine episodes, or cost-effectiveness.
The study also did not cover important clinical issues that might inform treatment decision-making, including drug overuse headache or potential withdrawal symptoms. And the authors were unable to quantify some outcomes, such as global functioning.
‘Best Profile’?
Reached for comment, Neurologist Nina Riggins, MD, PhD, Headache Center of Excellence, Palo Alto VA Medical Center in California, praised the authors for a “great job” of bringing attention to the topic.
However, she noted that the investigators’ characterization of the four triptans as having the “best profile” for acute migraine gave her pause.
“Calling triptans the medication with the ‘best profile’ might be not applicable in many cases,” she said. For example, those who need acute medication more than two to three times a week in addition to those with cardiovascular contraindications to triptans may fall outside of that category.
Dr. Riggins said that “it makes sense” that longer-acting triptans like frovatriptan and naratriptan may not rank highly for efficacy within the first 2 hours. However, these agents likely offer a superior therapeutic profile in specific contexts, such as menstrual-related migraine.
In addition, while triptans are known to cause medication overuse headache, this may not be the case with gepants, she noted.
In a release from the Science Media Center, a nonprofit organization promoting voices and views of the scientific community, Eloísa Rubio-Beltrán, PhD, research associate with The Migraine Trust at the Wolfson Sensory, Pain and Regeneration Centre, King’s College London in England, said the findings should affect migraine treatment guidelines.
“As the study highlights, due to their high efficacy and low cost, triptans should be the first-line treatment option for the acute treatment of migraine. These results should inform treatment guidelines and support the inclusion of the best performing triptans into the List of Essential Medicines, to optimize treatment, allowing patients to access more efficacious options,” said Dr. Rubio-Beltrán.
It is also important to note that gepants and ditans were developed to offer alternatives for patients who show no improvement from triptans, she added.
She pointed out that these medications were not developed as a substitute for triptans, but rather to expand the number of treatment options for migraine.
“Nonetheless,” she added, “this study highlights the need for further research on the pathophysiology of migraine, which will allow the discovery of novel targets, and thus, novel treatments options that will benefit all patient populations.”
The study was funded by the NIHR Oxford Health Biomedical Research Centre and the Lundbeck Foundation. Dr. Cipriani reported receiving research, educational, and consultancy fees from Italian Network for Pediatric Clinical Trials, Fondazione Cariplo, Lundbeck, and Angelini Pharma. Dr. Ashina is a consultant, speaker, or scientific adviser for AbbVie, Amgen, AstraZeneca, Eli Lilly, GSK, Lundbeck, Novartis, Pfizer, and Teva; is the past president of the International Headache Society; and an associate editor of The Journal of Headache and Pain and Brain. Dr. Riggins has consulted for Gerson Lehrman Group; participated in compensated work with AcademicCME and Association of Migraine Disorders; was a principal investigator on research with electroCore, Theranica, and Eli Lilly; serves on advisory boards for Theranica, Teva, Lundbeck, Amneal Pharmaceuticals, NeurologyLive, and Miles for Migraine; and is a project advisor for Clinical Awareness Initiative with Clinical Neurological Society of America. Dr. Rubio-Beltrán reported serving as a junior editorial board member of The Journal of Headache and Pain and a junior representative of the International Headache Society; receiving research support from The Migraine Trust, Eli Lilly, CoLucid Pharmaceuticals, Amgen, Novartis, and Kallyope; and receiving travel support from CoLucid Pharmaceuticals, Allergan, and Novartis.
A version of this article first appeared on Medscape.com.
new research suggested.
Results of a large systematic review and meta-analysis showed that eletriptan, rizatriptan, sumatriptan, and zolmitriptan were more effective than lasmiditan, rimegepant, and ubrogepant, which investigators found were as effective as nonsteroidal anti-inflammatory drugs (NSAIDs).
International guidelines generally endorse NSAIDs as the first-line treatment for migraine and recommend triptans for moderate to severe episodes or when the response to NSAIDs is insufficient.
However, based on the study’s findings, these four triptans should be considered the treatment of choice for migraine, study investigator Andrea Cipriani, MD, PhD, professor of psychiatry at the University of Oxford in England and director of the Oxford Health Clinical Research Facility, told a press briefing.
The investigators added that these particular triptans should be “included in the WHO [World Health Organization] List of Essential Medicines to promote global accessibility and uniform standards of care.”
The study was published online in The BMJ.
Filling the Knowledge Gap
To date, almost all migraine studies have compared migraine drugs with placebo, so there’s a knowledge gap, said Dr. Cipriani. As a result, “there’s no clear consensus among experts and guidelines about which specific drug classes should be prescribed initially, and this is a clinical problem.”
The researchers pointed out that, in recent years, lasmiditan and gepants have been introduced as further treatment options, especially for patients in whom triptans are contraindicated because of their potential vasoconstrictive effects and higher risk for ischemic events.
The analysis included 137 double-blind, randomized, controlled trials that were primarily sponsored by the pharmaceutical industry. It included 89,445 adult outpatients with migraine (mean age, 40.3 years; 85.6% women).
Only drugs licensed for migraine or headache that are recommended in at least one country were included. Researchers divided these 17 drugs into five categories: Antipyretics (paracetamol), ditans (lasmiditan), gepants (rimegepant and ubrogepant), NSAIDs (acetylsalicylic acid, celecoxib, diclofenac potassium, ibuprofen, naproxen sodium, and phenazone), and triptans (almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, sumatriptan, and zolmitriptan).
The study’s primary outcomes were freedom from pain at 2 hours and at 2-24 hours, without the use of rescue drugs.
Investigators used sumatriptan as the reference intervention because it is the most commonly prescribed migraine drug and is included in the WHO Model Lists of Essential Medicines.
The study showed all active interventions were better than placebo for pain freedom at 2 hours; with the exception of paracetamol and naratriptan, all were better for sustained pain freedom from 2 to 24 hours.
When the active interventions were compared with each other, eletriptan outperformed other drugs for achieving pain freedom at 2 hours. It was followed by rizatriptan, sumatriptan, and zolmitriptan (odds ratio [OR], 1.35-3.01). For sustained pain freedom up to 24 hours, the most efficacious interventions were eletriptan (OR, 1.41-2.73) and ibuprofen (OR, 3.16-4.82).
As for secondary efficacy outcomes, in head-to-head comparisons, eletriptan was superior to nearly all other active interventions for pain relief at 2 hours and for the use of rescue drugs.
As for adverse events, dizziness was more commonly associated with lasmiditan, eletriptan, sumatriptan, and zolmitriptan, while fatigue and sedation occurred more frequently with eletriptan and lasmiditan. Nausea was more often associated with lasmiditan, sumatriptan, zolmitriptan, and ubrogepant. Eletriptan was the only intervention most frequently associated with chest pain or discomfort.
Need to Update Guidelines?
Considering the new results, Dr. Cipriani and study coauthor Messoud Ashina, MD, PhD, professor of neurology, University of Copenhagen in Denmark, said clinical guidelines for acute migraine management should be updated.
While triptans are contraindicated in patients with vascular disease, the researchers noted that “cerebrovascular events may present primarily as migraine-like headaches, and misdiagnosis of transient ischemic attack and minor stroke as migraine is not rare.”
Recently, lasmiditan, rimegepant, and ubrogepant — which are not associated with vasoconstrictive effects — have been promoted as alternatives in patients for whom triptans are contraindicated or not tolerated. But the high costs of these drugs put them out of reach for some patients, the investigators noted.
Triptans are widely underutilized, Dr. Ashina noted during the press briefing. Current use ranges from 17% to 22% in the United States and from 3% to 22.5% in Europe.
The investigators said that triptans have been shown to be superior and should be promoted globally, adding that the limited access and substantial underutilization of these medications are “missed opportunities to offer more effective treatments.”
The new results underscore the importance of head-to-head trials, which is the gold standard, said Dr. Cipriani.
The investigators noted that the study’s main limitation was the quality of the data, which was deemed to be low, or very low, for most comparisons. Other potential limitations included lack of individual patient data; exclusion of combination drugs; inclusion of only oral treatments; and not considering type of oral formulation, consistency in response across migraine episodes, or cost-effectiveness.
The study also did not cover important clinical issues that might inform treatment decision-making, including drug overuse headache or potential withdrawal symptoms. And the authors were unable to quantify some outcomes, such as global functioning.
‘Best Profile’?
Reached for comment, Neurologist Nina Riggins, MD, PhD, Headache Center of Excellence, Palo Alto VA Medical Center in California, praised the authors for a “great job” of bringing attention to the topic.
However, she noted that the investigators’ characterization of the four triptans as having the “best profile” for acute migraine gave her pause.
“Calling triptans the medication with the ‘best profile’ might be not applicable in many cases,” she said. For example, those who need acute medication more than two to three times a week in addition to those with cardiovascular contraindications to triptans may fall outside of that category.
Dr. Riggins said that “it makes sense” that longer-acting triptans like frovatriptan and naratriptan may not rank highly for efficacy within the first 2 hours. However, these agents likely offer a superior therapeutic profile in specific contexts, such as menstrual-related migraine.
In addition, while triptans are known to cause medication overuse headache, this may not be the case with gepants, she noted.
In a release from the Science Media Center, a nonprofit organization promoting voices and views of the scientific community, Eloísa Rubio-Beltrán, PhD, research associate with The Migraine Trust at the Wolfson Sensory, Pain and Regeneration Centre, King’s College London in England, said the findings should affect migraine treatment guidelines.
“As the study highlights, due to their high efficacy and low cost, triptans should be the first-line treatment option for the acute treatment of migraine. These results should inform treatment guidelines and support the inclusion of the best performing triptans into the List of Essential Medicines, to optimize treatment, allowing patients to access more efficacious options,” said Dr. Rubio-Beltrán.
It is also important to note that gepants and ditans were developed to offer alternatives for patients who show no improvement from triptans, she added.
She pointed out that these medications were not developed as a substitute for triptans, but rather to expand the number of treatment options for migraine.
“Nonetheless,” she added, “this study highlights the need for further research on the pathophysiology of migraine, which will allow the discovery of novel targets, and thus, novel treatments options that will benefit all patient populations.”
The study was funded by the NIHR Oxford Health Biomedical Research Centre and the Lundbeck Foundation. Dr. Cipriani reported receiving research, educational, and consultancy fees from Italian Network for Pediatric Clinical Trials, Fondazione Cariplo, Lundbeck, and Angelini Pharma. Dr. Ashina is a consultant, speaker, or scientific adviser for AbbVie, Amgen, AstraZeneca, Eli Lilly, GSK, Lundbeck, Novartis, Pfizer, and Teva; is the past president of the International Headache Society; and an associate editor of The Journal of Headache and Pain and Brain. Dr. Riggins has consulted for Gerson Lehrman Group; participated in compensated work with AcademicCME and Association of Migraine Disorders; was a principal investigator on research with electroCore, Theranica, and Eli Lilly; serves on advisory boards for Theranica, Teva, Lundbeck, Amneal Pharmaceuticals, NeurologyLive, and Miles for Migraine; and is a project advisor for Clinical Awareness Initiative with Clinical Neurological Society of America. Dr. Rubio-Beltrán reported serving as a junior editorial board member of The Journal of Headache and Pain and a junior representative of the International Headache Society; receiving research support from The Migraine Trust, Eli Lilly, CoLucid Pharmaceuticals, Amgen, Novartis, and Kallyope; and receiving travel support from CoLucid Pharmaceuticals, Allergan, and Novartis.
A version of this article first appeared on Medscape.com.
FROM THE BMJ
Positive Stem Cell Transplant Data Is Increasing Its Use
COPENHAGEN —
With only one completed randomized trial available, HSCT remains experimental but the number of patients treated with this approach has now reached substantial numbers over 20 years of experience at multiple centers, according to two representative real-world studies presented at the 2024 ECTRIMS annual meeting.
The latest data are wholly consistent with a 2019 multinational randomized trial that found HSCT, which is a one-time intervention, to be relatively well tolerated and more effective than disease-modifying therapy (DMT) for median time to progression in patients refractory to DMT.
Relative to 24 months in the DMT group, median time to relapse was not reached among those randomized to HSCT because there were too few events. The hazard ratio (HR) reduction for progression was greater than 90% (HR, 0.07; P < 0.001). Other endpoints, such as EDSS, which improved in the group receiving HSCT but declined on DMT, also favored the single-treatment therapy.
Two Real-World Experiences With HSCT Reported
Of the two multicenter real world studies presented at ECTRIMS, one included 363 patients treated at one of 14 participating public hospitals in the United Kingdom since 2002. This analysis was uncontrolled. The other, with 97 patients treated at one of 20 participating centers in Italy since 1999, compared HSCT to alemtuzumab retrospectively.
In the UK data, presented by Paolo Muraro, MD, PhD, Senior Consultant, Division of Brain Sciences, Imperial College, London, England, 94.6% were in relapse-free survival (RFS) at 2 years and 88.6% at 5 years after undergoing HSCT. He called these numbers “impressive.”
In addition, MRI-free activity survival (MFS) was 88.2% and 78.8% at 2 and 5 years, respectively, Dr. Muraro said.
On the Expanded Disability Status Scale (EDSS), the cumulative incidence of improvement was 24.6% at 2 years and 28.6% at 5 years. There was no evidence of disease activity on the endpoints of symptoms, relapse, and MRI (NEDA-3) in 72% of patients at 2 years and 48.5% at 5 years.
Relative to historical response rates in a refractory population, Dr. Muraro considered these results favorable. Although there were four deaths, producing a treatment-related mortality of 1.1%, all occurred at an early stage of the HSCT program when there was limited experience in the management of cytopenias and other acute complications of HSCT.
“In this real-world cohort, stem cell transplant led to a durable remission of inflammatory activity and to clinical stability even though this included patients with a high EDSS at baseline [median 6.0] and a substantial proportion [39%) with progressive disease,” Dr. Muraro said.
In the Italian data, presented by Alessio Signori, MD, PhD, associate professor, Department of Health Sciences, University of Genoa, Italy, the 97 HSCT patients were matched with 314 treated with alemtuzumab over the same period and compared with propensity score overlap weighting. Baseline features were comparable.
HSCT Outperforms Alemtuzumab on All Measures
After a median follow-up of 62 months in the HSCT group and 30 months in the alemtuzumab group, HSCT outperformed drug therapy on all efficacy measures. When translated into HR, HSCT relative to alemtuzumab was associated with a 50% reduction in the probability of disability progression (HR, 0.50; P = 0.025), a 66% reduction in probability of relapse (HR, 0.34; P < 0.001), and a 62% reduction in the probability of MRI activity (HR, 0.38; P < 0.001).
“At 5 years, 58.3% of patients in the HSCT group versus 22.3% of the patients in the alemtuzumab group maintained NEDA-3,” said Dr. Signori, who reported that this difference represented a greater than 50% reduction (HR, 0.48; P < 0.001).
In this study there were two treatment-related deaths. Both occurred within 30 days of HSCT and, again, were confined to the early experience with HSCT.
There are questions that remain unanswered, such as whether there are predictors of response to HSCT. Although sustained responses have been greater on HSCT than drug therapy on average, poor responses appeared to be more common among those with a progressive phenotype in the UK experience.
Although patients with progressive disease were excluded from the Italian study, a real-world experience published 2 years ago also indicated that patients with progressive forms of MS respond less well to HSCT. Conducted in the United States at a single center (Northwestern University, Chicago) with 414 patients of whom 93 had progressive disease, EDSS progressively declined over the 5 years of follow-up among those with secondary progressive MS even as it improved in those with relapsing-remitting multiple sclerosis.
HSCT Considered on Compassionate Basis
Overall, the HSCT experience as a rescue therapy for MS patients with an inadequate response to DMT has led a growing number of centers active in this area to offer this option on a compassionate basis, according to Dr. Muraro and Dr. Signori. Joachim Burman, MD, PhD, who was the moderator of the scientific session at ECTRIMS and the leader of a research program into HSCT for MS at Uppsala University, Uppsala, Sweden, agreed.
The increasing number of centers offering HSCT to MS patients does not preclude the need or the value of the ongoing phase 3 trials, according to Dr. Burman, but he suggested that there is a growing focus of how it should be used, not whether it will be used.
Ultimately, he speculated that HSCT, once accepted as a mainstream option for MS, will probably be confined to the 5%-10% of patients with very aggressive disease. This is not for lack of safety or efficacy, but he sees several barriers to using this approach first-line, outside of special situations.
“You need a team of several different specialists to offer HSCT,” he said, suggesting that this approach to MS is much more complicated than a visit to a neurologist’s office for a DMT prescription. However, he thinks other barriers, such as concern about safety, are dissipating.
HSCT “is still being characterized as a high-risk procedure, but I would object to that,” he said. “The deaths associated with HSCT largely occurred 20 years ago when the field was new.”
Dr. Muraro reported a financial relationship with Cellerys AG that is unrelated to this study. Dr. Signori reported financial relationships with Chiesi, Horizon, and Novartis. Dr. Burman reports no potential conflicts of interest.
COPENHAGEN —
With only one completed randomized trial available, HSCT remains experimental but the number of patients treated with this approach has now reached substantial numbers over 20 years of experience at multiple centers, according to two representative real-world studies presented at the 2024 ECTRIMS annual meeting.
The latest data are wholly consistent with a 2019 multinational randomized trial that found HSCT, which is a one-time intervention, to be relatively well tolerated and more effective than disease-modifying therapy (DMT) for median time to progression in patients refractory to DMT.
Relative to 24 months in the DMT group, median time to relapse was not reached among those randomized to HSCT because there were too few events. The hazard ratio (HR) reduction for progression was greater than 90% (HR, 0.07; P < 0.001). Other endpoints, such as EDSS, which improved in the group receiving HSCT but declined on DMT, also favored the single-treatment therapy.
Two Real-World Experiences With HSCT Reported
Of the two multicenter real world studies presented at ECTRIMS, one included 363 patients treated at one of 14 participating public hospitals in the United Kingdom since 2002. This analysis was uncontrolled. The other, with 97 patients treated at one of 20 participating centers in Italy since 1999, compared HSCT to alemtuzumab retrospectively.
In the UK data, presented by Paolo Muraro, MD, PhD, Senior Consultant, Division of Brain Sciences, Imperial College, London, England, 94.6% were in relapse-free survival (RFS) at 2 years and 88.6% at 5 years after undergoing HSCT. He called these numbers “impressive.”
In addition, MRI-free activity survival (MFS) was 88.2% and 78.8% at 2 and 5 years, respectively, Dr. Muraro said.
On the Expanded Disability Status Scale (EDSS), the cumulative incidence of improvement was 24.6% at 2 years and 28.6% at 5 years. There was no evidence of disease activity on the endpoints of symptoms, relapse, and MRI (NEDA-3) in 72% of patients at 2 years and 48.5% at 5 years.
Relative to historical response rates in a refractory population, Dr. Muraro considered these results favorable. Although there were four deaths, producing a treatment-related mortality of 1.1%, all occurred at an early stage of the HSCT program when there was limited experience in the management of cytopenias and other acute complications of HSCT.
“In this real-world cohort, stem cell transplant led to a durable remission of inflammatory activity and to clinical stability even though this included patients with a high EDSS at baseline [median 6.0] and a substantial proportion [39%) with progressive disease,” Dr. Muraro said.
In the Italian data, presented by Alessio Signori, MD, PhD, associate professor, Department of Health Sciences, University of Genoa, Italy, the 97 HSCT patients were matched with 314 treated with alemtuzumab over the same period and compared with propensity score overlap weighting. Baseline features were comparable.
HSCT Outperforms Alemtuzumab on All Measures
After a median follow-up of 62 months in the HSCT group and 30 months in the alemtuzumab group, HSCT outperformed drug therapy on all efficacy measures. When translated into HR, HSCT relative to alemtuzumab was associated with a 50% reduction in the probability of disability progression (HR, 0.50; P = 0.025), a 66% reduction in probability of relapse (HR, 0.34; P < 0.001), and a 62% reduction in the probability of MRI activity (HR, 0.38; P < 0.001).
“At 5 years, 58.3% of patients in the HSCT group versus 22.3% of the patients in the alemtuzumab group maintained NEDA-3,” said Dr. Signori, who reported that this difference represented a greater than 50% reduction (HR, 0.48; P < 0.001).
In this study there were two treatment-related deaths. Both occurred within 30 days of HSCT and, again, were confined to the early experience with HSCT.
There are questions that remain unanswered, such as whether there are predictors of response to HSCT. Although sustained responses have been greater on HSCT than drug therapy on average, poor responses appeared to be more common among those with a progressive phenotype in the UK experience.
Although patients with progressive disease were excluded from the Italian study, a real-world experience published 2 years ago also indicated that patients with progressive forms of MS respond less well to HSCT. Conducted in the United States at a single center (Northwestern University, Chicago) with 414 patients of whom 93 had progressive disease, EDSS progressively declined over the 5 years of follow-up among those with secondary progressive MS even as it improved in those with relapsing-remitting multiple sclerosis.
HSCT Considered on Compassionate Basis
Overall, the HSCT experience as a rescue therapy for MS patients with an inadequate response to DMT has led a growing number of centers active in this area to offer this option on a compassionate basis, according to Dr. Muraro and Dr. Signori. Joachim Burman, MD, PhD, who was the moderator of the scientific session at ECTRIMS and the leader of a research program into HSCT for MS at Uppsala University, Uppsala, Sweden, agreed.
The increasing number of centers offering HSCT to MS patients does not preclude the need or the value of the ongoing phase 3 trials, according to Dr. Burman, but he suggested that there is a growing focus of how it should be used, not whether it will be used.
Ultimately, he speculated that HSCT, once accepted as a mainstream option for MS, will probably be confined to the 5%-10% of patients with very aggressive disease. This is not for lack of safety or efficacy, but he sees several barriers to using this approach first-line, outside of special situations.
“You need a team of several different specialists to offer HSCT,” he said, suggesting that this approach to MS is much more complicated than a visit to a neurologist’s office for a DMT prescription. However, he thinks other barriers, such as concern about safety, are dissipating.
HSCT “is still being characterized as a high-risk procedure, but I would object to that,” he said. “The deaths associated with HSCT largely occurred 20 years ago when the field was new.”
Dr. Muraro reported a financial relationship with Cellerys AG that is unrelated to this study. Dr. Signori reported financial relationships with Chiesi, Horizon, and Novartis. Dr. Burman reports no potential conflicts of interest.
COPENHAGEN —
With only one completed randomized trial available, HSCT remains experimental but the number of patients treated with this approach has now reached substantial numbers over 20 years of experience at multiple centers, according to two representative real-world studies presented at the 2024 ECTRIMS annual meeting.
The latest data are wholly consistent with a 2019 multinational randomized trial that found HSCT, which is a one-time intervention, to be relatively well tolerated and more effective than disease-modifying therapy (DMT) for median time to progression in patients refractory to DMT.
Relative to 24 months in the DMT group, median time to relapse was not reached among those randomized to HSCT because there were too few events. The hazard ratio (HR) reduction for progression was greater than 90% (HR, 0.07; P < 0.001). Other endpoints, such as EDSS, which improved in the group receiving HSCT but declined on DMT, also favored the single-treatment therapy.
Two Real-World Experiences With HSCT Reported
Of the two multicenter real world studies presented at ECTRIMS, one included 363 patients treated at one of 14 participating public hospitals in the United Kingdom since 2002. This analysis was uncontrolled. The other, with 97 patients treated at one of 20 participating centers in Italy since 1999, compared HSCT to alemtuzumab retrospectively.
In the UK data, presented by Paolo Muraro, MD, PhD, Senior Consultant, Division of Brain Sciences, Imperial College, London, England, 94.6% were in relapse-free survival (RFS) at 2 years and 88.6% at 5 years after undergoing HSCT. He called these numbers “impressive.”
In addition, MRI-free activity survival (MFS) was 88.2% and 78.8% at 2 and 5 years, respectively, Dr. Muraro said.
On the Expanded Disability Status Scale (EDSS), the cumulative incidence of improvement was 24.6% at 2 years and 28.6% at 5 years. There was no evidence of disease activity on the endpoints of symptoms, relapse, and MRI (NEDA-3) in 72% of patients at 2 years and 48.5% at 5 years.
Relative to historical response rates in a refractory population, Dr. Muraro considered these results favorable. Although there were four deaths, producing a treatment-related mortality of 1.1%, all occurred at an early stage of the HSCT program when there was limited experience in the management of cytopenias and other acute complications of HSCT.
“In this real-world cohort, stem cell transplant led to a durable remission of inflammatory activity and to clinical stability even though this included patients with a high EDSS at baseline [median 6.0] and a substantial proportion [39%) with progressive disease,” Dr. Muraro said.
In the Italian data, presented by Alessio Signori, MD, PhD, associate professor, Department of Health Sciences, University of Genoa, Italy, the 97 HSCT patients were matched with 314 treated with alemtuzumab over the same period and compared with propensity score overlap weighting. Baseline features were comparable.
HSCT Outperforms Alemtuzumab on All Measures
After a median follow-up of 62 months in the HSCT group and 30 months in the alemtuzumab group, HSCT outperformed drug therapy on all efficacy measures. When translated into HR, HSCT relative to alemtuzumab was associated with a 50% reduction in the probability of disability progression (HR, 0.50; P = 0.025), a 66% reduction in probability of relapse (HR, 0.34; P < 0.001), and a 62% reduction in the probability of MRI activity (HR, 0.38; P < 0.001).
“At 5 years, 58.3% of patients in the HSCT group versus 22.3% of the patients in the alemtuzumab group maintained NEDA-3,” said Dr. Signori, who reported that this difference represented a greater than 50% reduction (HR, 0.48; P < 0.001).
In this study there were two treatment-related deaths. Both occurred within 30 days of HSCT and, again, were confined to the early experience with HSCT.
There are questions that remain unanswered, such as whether there are predictors of response to HSCT. Although sustained responses have been greater on HSCT than drug therapy on average, poor responses appeared to be more common among those with a progressive phenotype in the UK experience.
Although patients with progressive disease were excluded from the Italian study, a real-world experience published 2 years ago also indicated that patients with progressive forms of MS respond less well to HSCT. Conducted in the United States at a single center (Northwestern University, Chicago) with 414 patients of whom 93 had progressive disease, EDSS progressively declined over the 5 years of follow-up among those with secondary progressive MS even as it improved in those with relapsing-remitting multiple sclerosis.
HSCT Considered on Compassionate Basis
Overall, the HSCT experience as a rescue therapy for MS patients with an inadequate response to DMT has led a growing number of centers active in this area to offer this option on a compassionate basis, according to Dr. Muraro and Dr. Signori. Joachim Burman, MD, PhD, who was the moderator of the scientific session at ECTRIMS and the leader of a research program into HSCT for MS at Uppsala University, Uppsala, Sweden, agreed.
The increasing number of centers offering HSCT to MS patients does not preclude the need or the value of the ongoing phase 3 trials, according to Dr. Burman, but he suggested that there is a growing focus of how it should be used, not whether it will be used.
Ultimately, he speculated that HSCT, once accepted as a mainstream option for MS, will probably be confined to the 5%-10% of patients with very aggressive disease. This is not for lack of safety or efficacy, but he sees several barriers to using this approach first-line, outside of special situations.
“You need a team of several different specialists to offer HSCT,” he said, suggesting that this approach to MS is much more complicated than a visit to a neurologist’s office for a DMT prescription. However, he thinks other barriers, such as concern about safety, are dissipating.
HSCT “is still being characterized as a high-risk procedure, but I would object to that,” he said. “The deaths associated with HSCT largely occurred 20 years ago when the field was new.”
Dr. Muraro reported a financial relationship with Cellerys AG that is unrelated to this study. Dr. Signori reported financial relationships with Chiesi, Horizon, and Novartis. Dr. Burman reports no potential conflicts of interest.
FROM ECTRIMS 2024
Comorbidity Control Might Slow MS Activity
COPENHAGEN — The largest and perhaps most rigorous study to demonstrate an association between the presence of comorbidities and accelerated progression of multiple sclerosis (MS) is sufficiently compelling that both the study author and an independent expert maintained clinical practice should be adjusted.
Even while acknowledging that “it is hard to make causative statements” on the basis of these types of data, the findings are sufficiently compelling to suggest that comorbidities “should be a pressing concern” in MS management, according to Amber Salter, PhD, an associate professor of biostatistics at the University of Texas Southwestern Medical School, Dallas.
The strong association in this meta-analysis, presented at the 2024 ECTRIMS annual meeting, were drawn from 15 multicenter phase 3 treatment trials with 16,794 participants followed for at least 2 years, Dr. Salter reported. Her data were published simultaneously in JAMA Neurology.
“One of the strengths of this study is that the data on comorbidities were collected prospectively as part of these trials,” explained Mark S. Freedman, MD, director of the Multiple Sclerosis Research Center at Ottawa Hospital in Canada. He agreed with Dr. Salter that it is reasonable to apply these findings to clinical practice given their consistency with numerous other studies and the value of what he termed as “a holistic approach” to improving outcomes in MS.
Meta-Analysis Avoids Weaknesses of Previous Data
There are many potential weaknesses of past observational studies that the authors of this meta-analysis hoped to avoid. These include the possibility that MS patients with comorbidities might be less likely to take or adhere to disease-modifying therapies (DMT) or that comorbidity burden might masquerade or be misinterpreted as MS progression. By employing data from phase 3 DMT trials, Dr. Salter maintained that prospectively collected data monitored carefully over an extended follow-up allows the impact of comorbidities on outcome to be evaluated in a more controlled fashion.
Dr. Freedman liked the design of this study, but he admitted that he was surprised by the result.
“Phase 3 trials typically include exclusion criteria for significant comorbidities, so I did not think they would be able to show any meaningful differences,” Dr. Freedman said in an interview.
For the main outcome of evidence of disease activity (EDA), defined as confirmed disability worsening measured with the Expanded Disability Status Scale (EDSS), relapse activity, or any new or enlarging lesions on MRI, the differences reached significance even after adjustments for multiple potentially confounding factors.
MS Activity Increases Significantly with More Comorbidities
Compared with no comorbidity, the presence of three or more comorbidities were associated with a significant 14% increase in the adjusted hazard ratio (aHR) of EDA (aHR, 1.14; 95% CI, 1.02-1.28), Dr. Salter reported. If there were two or more cardiometabolic comorbidities, the risk of EDA was increased 21% (aHR, 1.21; 95% CI 1.08-1.37).
The list of comorbidities considered in this study was drawn from the International Advisory Committee on Clinical Trials in MS. It included numerous cardiometabolic comorbidities, such as hypertension, hyperlipidemia, diabetes, ischemic heart disease, cerebrovascular disease, and peripheral vascular disease. It also included chronic lung diseases, such as asthma and chronic obstructive pulmonary disease; psychiatric diseases, such as depression and anxiety; and miscellaneous autoimmune conditions.
The number of comorbidities was categorized for analysis as zero, one, two, or three or more. However, Dr. Salter acknowledged that these phase 3 trials did include comorbidity exclusion criteria. In fact, severe forms of most of these comorbidities were exclusion criteria in at least some studies. Yet, the prevalence of one or more comorbidities was still 45.4% in the total population from this meta-analysis.
By themselves alone, ischemic heart disease (aHR, 1.63), cerebrovascular disease (aHR, 1.70) and at least one psychiatric disorder (aHR, 1.14) were all significant for increased MS activity at the end of 2 years by a 95% confidence interval that did not cross the line of unity.
When the EDA endpoints were evaluated individually, not even three or more comorbidities was associated with an increased rate of active lesions on MRI at the end of follow-up, but two or more and three or more comorbidities were associated with a significantly increased risk of disability worsening (aHR, 1.16 and aHR, 1.31, respectively) and relapse (aHR, 1.16 for both).
An Underestimation of Associations?
Prospective trials are still needed to show that treating comorbidities improves outcome in MS, but randomization will be problematic if it means withholding treatment for conditions with risks independent of MS, Dr. Salter said. Although the data from this analysis did not permit an analysis of how relative severity of comorbidities affected MS outcome, she reiterated that most patients with severe comorbidities were likely excluded from inclusion in the studies anyway.
“We think that we are probably seeing an underestimation of an associations between comorbidity and increased MS activity,” Dr. Salter said. While she reported that confounding cannot be ruled out, the robust associations identified in a meta-analysis “limit the possibility of bias or chance findings.”
Asked if the message that clinicians should treat comorbidities to reduce MS activity is a reasonable conclusion in the absence of proof that treatment is beneficial, Dr. Freedman looked both to the body of evidence and to the common sense behind the recommendation.
Basically, Dr. Freedman believes that comorbidities should be addressed routinely and rigorously even if there was no evidence that they improve MS outcome. These data provide just one other source of support for a practice that should be conducted anyway.
Dr. Salter reported financial relationships with Abata Therapeutics, Gryphon Bio, and Owl Therapeutics. Dr. Freedman reported financial relationships with more than 10 pharmaceutical companies.
COPENHAGEN — The largest and perhaps most rigorous study to demonstrate an association between the presence of comorbidities and accelerated progression of multiple sclerosis (MS) is sufficiently compelling that both the study author and an independent expert maintained clinical practice should be adjusted.
Even while acknowledging that “it is hard to make causative statements” on the basis of these types of data, the findings are sufficiently compelling to suggest that comorbidities “should be a pressing concern” in MS management, according to Amber Salter, PhD, an associate professor of biostatistics at the University of Texas Southwestern Medical School, Dallas.
The strong association in this meta-analysis, presented at the 2024 ECTRIMS annual meeting, were drawn from 15 multicenter phase 3 treatment trials with 16,794 participants followed for at least 2 years, Dr. Salter reported. Her data were published simultaneously in JAMA Neurology.
“One of the strengths of this study is that the data on comorbidities were collected prospectively as part of these trials,” explained Mark S. Freedman, MD, director of the Multiple Sclerosis Research Center at Ottawa Hospital in Canada. He agreed with Dr. Salter that it is reasonable to apply these findings to clinical practice given their consistency with numerous other studies and the value of what he termed as “a holistic approach” to improving outcomes in MS.
Meta-Analysis Avoids Weaknesses of Previous Data
There are many potential weaknesses of past observational studies that the authors of this meta-analysis hoped to avoid. These include the possibility that MS patients with comorbidities might be less likely to take or adhere to disease-modifying therapies (DMT) or that comorbidity burden might masquerade or be misinterpreted as MS progression. By employing data from phase 3 DMT trials, Dr. Salter maintained that prospectively collected data monitored carefully over an extended follow-up allows the impact of comorbidities on outcome to be evaluated in a more controlled fashion.
Dr. Freedman liked the design of this study, but he admitted that he was surprised by the result.
“Phase 3 trials typically include exclusion criteria for significant comorbidities, so I did not think they would be able to show any meaningful differences,” Dr. Freedman said in an interview.
For the main outcome of evidence of disease activity (EDA), defined as confirmed disability worsening measured with the Expanded Disability Status Scale (EDSS), relapse activity, or any new or enlarging lesions on MRI, the differences reached significance even after adjustments for multiple potentially confounding factors.
MS Activity Increases Significantly with More Comorbidities
Compared with no comorbidity, the presence of three or more comorbidities were associated with a significant 14% increase in the adjusted hazard ratio (aHR) of EDA (aHR, 1.14; 95% CI, 1.02-1.28), Dr. Salter reported. If there were two or more cardiometabolic comorbidities, the risk of EDA was increased 21% (aHR, 1.21; 95% CI 1.08-1.37).
The list of comorbidities considered in this study was drawn from the International Advisory Committee on Clinical Trials in MS. It included numerous cardiometabolic comorbidities, such as hypertension, hyperlipidemia, diabetes, ischemic heart disease, cerebrovascular disease, and peripheral vascular disease. It also included chronic lung diseases, such as asthma and chronic obstructive pulmonary disease; psychiatric diseases, such as depression and anxiety; and miscellaneous autoimmune conditions.
The number of comorbidities was categorized for analysis as zero, one, two, or three or more. However, Dr. Salter acknowledged that these phase 3 trials did include comorbidity exclusion criteria. In fact, severe forms of most of these comorbidities were exclusion criteria in at least some studies. Yet, the prevalence of one or more comorbidities was still 45.4% in the total population from this meta-analysis.
By themselves alone, ischemic heart disease (aHR, 1.63), cerebrovascular disease (aHR, 1.70) and at least one psychiatric disorder (aHR, 1.14) were all significant for increased MS activity at the end of 2 years by a 95% confidence interval that did not cross the line of unity.
When the EDA endpoints were evaluated individually, not even three or more comorbidities was associated with an increased rate of active lesions on MRI at the end of follow-up, but two or more and three or more comorbidities were associated with a significantly increased risk of disability worsening (aHR, 1.16 and aHR, 1.31, respectively) and relapse (aHR, 1.16 for both).
An Underestimation of Associations?
Prospective trials are still needed to show that treating comorbidities improves outcome in MS, but randomization will be problematic if it means withholding treatment for conditions with risks independent of MS, Dr. Salter said. Although the data from this analysis did not permit an analysis of how relative severity of comorbidities affected MS outcome, she reiterated that most patients with severe comorbidities were likely excluded from inclusion in the studies anyway.
“We think that we are probably seeing an underestimation of an associations between comorbidity and increased MS activity,” Dr. Salter said. While she reported that confounding cannot be ruled out, the robust associations identified in a meta-analysis “limit the possibility of bias or chance findings.”
Asked if the message that clinicians should treat comorbidities to reduce MS activity is a reasonable conclusion in the absence of proof that treatment is beneficial, Dr. Freedman looked both to the body of evidence and to the common sense behind the recommendation.
Basically, Dr. Freedman believes that comorbidities should be addressed routinely and rigorously even if there was no evidence that they improve MS outcome. These data provide just one other source of support for a practice that should be conducted anyway.
Dr. Salter reported financial relationships with Abata Therapeutics, Gryphon Bio, and Owl Therapeutics. Dr. Freedman reported financial relationships with more than 10 pharmaceutical companies.
COPENHAGEN — The largest and perhaps most rigorous study to demonstrate an association between the presence of comorbidities and accelerated progression of multiple sclerosis (MS) is sufficiently compelling that both the study author and an independent expert maintained clinical practice should be adjusted.
Even while acknowledging that “it is hard to make causative statements” on the basis of these types of data, the findings are sufficiently compelling to suggest that comorbidities “should be a pressing concern” in MS management, according to Amber Salter, PhD, an associate professor of biostatistics at the University of Texas Southwestern Medical School, Dallas.
The strong association in this meta-analysis, presented at the 2024 ECTRIMS annual meeting, were drawn from 15 multicenter phase 3 treatment trials with 16,794 participants followed for at least 2 years, Dr. Salter reported. Her data were published simultaneously in JAMA Neurology.
“One of the strengths of this study is that the data on comorbidities were collected prospectively as part of these trials,” explained Mark S. Freedman, MD, director of the Multiple Sclerosis Research Center at Ottawa Hospital in Canada. He agreed with Dr. Salter that it is reasonable to apply these findings to clinical practice given their consistency with numerous other studies and the value of what he termed as “a holistic approach” to improving outcomes in MS.
Meta-Analysis Avoids Weaknesses of Previous Data
There are many potential weaknesses of past observational studies that the authors of this meta-analysis hoped to avoid. These include the possibility that MS patients with comorbidities might be less likely to take or adhere to disease-modifying therapies (DMT) or that comorbidity burden might masquerade or be misinterpreted as MS progression. By employing data from phase 3 DMT trials, Dr. Salter maintained that prospectively collected data monitored carefully over an extended follow-up allows the impact of comorbidities on outcome to be evaluated in a more controlled fashion.
Dr. Freedman liked the design of this study, but he admitted that he was surprised by the result.
“Phase 3 trials typically include exclusion criteria for significant comorbidities, so I did not think they would be able to show any meaningful differences,” Dr. Freedman said in an interview.
For the main outcome of evidence of disease activity (EDA), defined as confirmed disability worsening measured with the Expanded Disability Status Scale (EDSS), relapse activity, or any new or enlarging lesions on MRI, the differences reached significance even after adjustments for multiple potentially confounding factors.
MS Activity Increases Significantly with More Comorbidities
Compared with no comorbidity, the presence of three or more comorbidities were associated with a significant 14% increase in the adjusted hazard ratio (aHR) of EDA (aHR, 1.14; 95% CI, 1.02-1.28), Dr. Salter reported. If there were two or more cardiometabolic comorbidities, the risk of EDA was increased 21% (aHR, 1.21; 95% CI 1.08-1.37).
The list of comorbidities considered in this study was drawn from the International Advisory Committee on Clinical Trials in MS. It included numerous cardiometabolic comorbidities, such as hypertension, hyperlipidemia, diabetes, ischemic heart disease, cerebrovascular disease, and peripheral vascular disease. It also included chronic lung diseases, such as asthma and chronic obstructive pulmonary disease; psychiatric diseases, such as depression and anxiety; and miscellaneous autoimmune conditions.
The number of comorbidities was categorized for analysis as zero, one, two, or three or more. However, Dr. Salter acknowledged that these phase 3 trials did include comorbidity exclusion criteria. In fact, severe forms of most of these comorbidities were exclusion criteria in at least some studies. Yet, the prevalence of one or more comorbidities was still 45.4% in the total population from this meta-analysis.
By themselves alone, ischemic heart disease (aHR, 1.63), cerebrovascular disease (aHR, 1.70) and at least one psychiatric disorder (aHR, 1.14) were all significant for increased MS activity at the end of 2 years by a 95% confidence interval that did not cross the line of unity.
When the EDA endpoints were evaluated individually, not even three or more comorbidities was associated with an increased rate of active lesions on MRI at the end of follow-up, but two or more and three or more comorbidities were associated with a significantly increased risk of disability worsening (aHR, 1.16 and aHR, 1.31, respectively) and relapse (aHR, 1.16 for both).
An Underestimation of Associations?
Prospective trials are still needed to show that treating comorbidities improves outcome in MS, but randomization will be problematic if it means withholding treatment for conditions with risks independent of MS, Dr. Salter said. Although the data from this analysis did not permit an analysis of how relative severity of comorbidities affected MS outcome, she reiterated that most patients with severe comorbidities were likely excluded from inclusion in the studies anyway.
“We think that we are probably seeing an underestimation of an associations between comorbidity and increased MS activity,” Dr. Salter said. While she reported that confounding cannot be ruled out, the robust associations identified in a meta-analysis “limit the possibility of bias or chance findings.”
Asked if the message that clinicians should treat comorbidities to reduce MS activity is a reasonable conclusion in the absence of proof that treatment is beneficial, Dr. Freedman looked both to the body of evidence and to the common sense behind the recommendation.
Basically, Dr. Freedman believes that comorbidities should be addressed routinely and rigorously even if there was no evidence that they improve MS outcome. These data provide just one other source of support for a practice that should be conducted anyway.
Dr. Salter reported financial relationships with Abata Therapeutics, Gryphon Bio, and Owl Therapeutics. Dr. Freedman reported financial relationships with more than 10 pharmaceutical companies.
FROM ECTRIMS 2024
When You and Your Malpractice Insurer Disagree on Your Case
You’ve been sued for medical malpractice. If you are a physician in the United States, that is not an unlikely scenario.
An analysis by the American Medical Association shows that almost half of all physicians are sued by the time they reach 54. In some specialties, such as ob.gyn., one is almost guaranteed to be sued at some point.
But that’s what medical malpractice insurance is for, right? Your medical malpractice insurer will assign an attorney to take care of you and help you through this situation. Won’t they?
Maybe so, but the attorney and the claims representative your insurer assigns to your case may have a different idea about how to proceed than you do. Though the defense attorney assigned to you represents you, he or she gets paid by the insurance carrier.
This can create a conflict when your defense counsel and your insurance claims representative aim to take your case in a direction you don’t like.
Disagreements might include:
- Choice of expert witnesses
- Tactical decisions related to trial strategy
- Public relations considerations
- Admissions of liability
- Allocation of resources
To Settle or Not?
One of the most challenging — and common — disagreements is whether to settle the case.
Sometimes a malpractice insurer wants to settle the case against the defendant doctor’s wishes. Or the doctor wants to settle but is pushed into going to trial. In the following case, one doctor had to face the consequences of a decision he didn’t even make.
The Underlying Medical Malpractice Case
Dr. D was sued by a patient who had allegedly called Dr. D’s office six times in 2 days complaining of intermittent chest pain.
Dr. D had been swamped with patients and couldn’t squeeze this patient in for an office visit, but he did call back. The patient later claimed that during the call he told the doctor he was suffering from chest pain. The doctor recalled that the patient had complained of abdominal discomfort that began after he had exercised.
The physician wrote a prescription for an ECG at the local hospital and called to ensure that the patient could just walk in. The ECG was allegedly abnormal but was not read as representing an impending or current heart attack. Later that evening, however, the patient went to the emergency department of another hospital where it was confirmed that he had suffered a heart attack. The patient underwent cardiac catheterization and stent placement to address a blockage in his left anterior descending artery.
The patient subsequently sued Dr. D and the hospital where he had the original ECG. Dr. D contacted his medical malpractice insurance company. The insurance company assigned an attorney to represent Dr. D. Discovery in the case began.
The plaintiff’s own medical expert testified in a deposition that there was no way for the heart attack to have been prevented and that the treatment would have been the same either way. But Dr. D could not find a record of the phone calls with the patient, and he had not noted his conversation the patient in their medical records.
Dr. D held a policy for $1 million, and his state had a fund that would kick in an additional $1 million. But the plaintiffs demanded $4 million to settle.
A month before trial, the plaintiff’s attorney sent a threatening letter to Dr. D’s attorney warning him that Dr. D was underinsured and suggesting that it would be in the physician’s best interests to settle.
“I want to stress to you that it is not my desire to harm your client’s reputation or to destroy his business,” wrote the plaintiff’s attorney. “However, now is the time to avoid consequences such as these by making a good faith effort to get this case resolved.”
The letter went on to note that the defense attorney should give Dr. D a copy of the letter so that everyone would be aware of the potential consequences of an award against Dr. D in excess of his limits of insurance coverage. The plaintiff’s attorney even suggested that Dr. D should retain personal counsel.
Dr. D’s defense attorney downplayed the letter and assured him that there was no reason to worry.
Meanwhile the case inched closer to trial.
The codefendant hospital settled with the plaintiff on the night before jury selection, leaving Dr. D in the uncomfortable position of being the only defendant in the case. At this point, Dr. D decided he would like to settle, and he sent his attorney an email telling him so. But the attorney instead referred him to an insurance company claims.
Just days before the trial was to start, Dr. D repeatedly told the claims representative assigned to his claim that he did not want to go to trial but rather wanted to settle. The representative told Dr. D that he had no choice in whether the action settled.
A committee at the insurance company had decided to proceed with the trial rather than settle.
The trial proved a painful debacle for Dr. D. His attorney’s idea of showing a “gotcha” video of the allegedly permanently injured plaintiff carrying a large, heavy box backfired when the jury was shown by the plaintiff that the box actually contained ice cream cones and weighed very little.
Prior to trial, the plaintiff offered to settle for $1 million. On the first day of trial, they lowered that amount to $750,000, yet the defense attorney did not settle the case, and it proceeded to a jury verdict. The jury awarded the plaintiff over $4 million — well in excess of Dr. D’s policy limits.
The Follow-up
Dr. D was horrified, but the insurance company claims representative said the insurer would promptly offer $2 million in available insurance coverage to settle the case post verdict. This did not happen. Instead, the insurer chose to appeal the verdict against Dr. D’s wishes.
Ultimately, Dr. D was forced to hire his own lawyer. He ultimately sued the insurance company for breach of contract and bad faith.
The insurance company eventually attempted to settle with the plaintiffs’ counsel, but the plaintiff refused to accept the available insurance coverage. The insurance carrier still has not posted the entire appeal bond. The case is still pending.
Protecting Yourself
The lesson from Dr. D’s experience: Understand that the insurance company is not your friend. It’s a business looking out for its own interests.
The plaintiff’s attorney was absolutely correct in suggesting that Dr. D retain his own attorney to represent his own interests. You should hire your own lawyer when:
- You disagree with your insurer on how to proceed in a case.
- You receive a demand that exceeds your available insurance coverage or for damages that may not be covered by your policy, such as punitive damages.
- Your insurance carrier attempts to deny insurance coverage for your claim or sends you a letter stating that it is “reserving its rights” not to cover or to limit coverage for your claim.
Retaining independent counsel protects your interests, not those of your insurance company.
Independent counsel can give you a second opinion on the strengths and weaknesses of your claim, help you prepare for your deposition, and attend court dates with you to ensure that you are completely protected.
Independent counsel can challenge your insurance company’s decision to deny or limit your insurance coverage and ensure that you receive all of the benefits to which you are entitled under your insurance policy. Some policies may include an independent lawyer to be paid for by your insurance carrier in case of a conflicts.
The most important takeaway? Your medical malpractice insurance carrier is not your friend, so act accordingly in times of conflict.
A version of this article first appeared on Medscape.com.
You’ve been sued for medical malpractice. If you are a physician in the United States, that is not an unlikely scenario.
An analysis by the American Medical Association shows that almost half of all physicians are sued by the time they reach 54. In some specialties, such as ob.gyn., one is almost guaranteed to be sued at some point.
But that’s what medical malpractice insurance is for, right? Your medical malpractice insurer will assign an attorney to take care of you and help you through this situation. Won’t they?
Maybe so, but the attorney and the claims representative your insurer assigns to your case may have a different idea about how to proceed than you do. Though the defense attorney assigned to you represents you, he or she gets paid by the insurance carrier.
This can create a conflict when your defense counsel and your insurance claims representative aim to take your case in a direction you don’t like.
Disagreements might include:
- Choice of expert witnesses
- Tactical decisions related to trial strategy
- Public relations considerations
- Admissions of liability
- Allocation of resources
To Settle or Not?
One of the most challenging — and common — disagreements is whether to settle the case.
Sometimes a malpractice insurer wants to settle the case against the defendant doctor’s wishes. Or the doctor wants to settle but is pushed into going to trial. In the following case, one doctor had to face the consequences of a decision he didn’t even make.
The Underlying Medical Malpractice Case
Dr. D was sued by a patient who had allegedly called Dr. D’s office six times in 2 days complaining of intermittent chest pain.
Dr. D had been swamped with patients and couldn’t squeeze this patient in for an office visit, but he did call back. The patient later claimed that during the call he told the doctor he was suffering from chest pain. The doctor recalled that the patient had complained of abdominal discomfort that began after he had exercised.
The physician wrote a prescription for an ECG at the local hospital and called to ensure that the patient could just walk in. The ECG was allegedly abnormal but was not read as representing an impending or current heart attack. Later that evening, however, the patient went to the emergency department of another hospital where it was confirmed that he had suffered a heart attack. The patient underwent cardiac catheterization and stent placement to address a blockage in his left anterior descending artery.
The patient subsequently sued Dr. D and the hospital where he had the original ECG. Dr. D contacted his medical malpractice insurance company. The insurance company assigned an attorney to represent Dr. D. Discovery in the case began.
The plaintiff’s own medical expert testified in a deposition that there was no way for the heart attack to have been prevented and that the treatment would have been the same either way. But Dr. D could not find a record of the phone calls with the patient, and he had not noted his conversation the patient in their medical records.
Dr. D held a policy for $1 million, and his state had a fund that would kick in an additional $1 million. But the plaintiffs demanded $4 million to settle.
A month before trial, the plaintiff’s attorney sent a threatening letter to Dr. D’s attorney warning him that Dr. D was underinsured and suggesting that it would be in the physician’s best interests to settle.
“I want to stress to you that it is not my desire to harm your client’s reputation or to destroy his business,” wrote the plaintiff’s attorney. “However, now is the time to avoid consequences such as these by making a good faith effort to get this case resolved.”
The letter went on to note that the defense attorney should give Dr. D a copy of the letter so that everyone would be aware of the potential consequences of an award against Dr. D in excess of his limits of insurance coverage. The plaintiff’s attorney even suggested that Dr. D should retain personal counsel.
Dr. D’s defense attorney downplayed the letter and assured him that there was no reason to worry.
Meanwhile the case inched closer to trial.
The codefendant hospital settled with the plaintiff on the night before jury selection, leaving Dr. D in the uncomfortable position of being the only defendant in the case. At this point, Dr. D decided he would like to settle, and he sent his attorney an email telling him so. But the attorney instead referred him to an insurance company claims.
Just days before the trial was to start, Dr. D repeatedly told the claims representative assigned to his claim that he did not want to go to trial but rather wanted to settle. The representative told Dr. D that he had no choice in whether the action settled.
A committee at the insurance company had decided to proceed with the trial rather than settle.
The trial proved a painful debacle for Dr. D. His attorney’s idea of showing a “gotcha” video of the allegedly permanently injured plaintiff carrying a large, heavy box backfired when the jury was shown by the plaintiff that the box actually contained ice cream cones and weighed very little.
Prior to trial, the plaintiff offered to settle for $1 million. On the first day of trial, they lowered that amount to $750,000, yet the defense attorney did not settle the case, and it proceeded to a jury verdict. The jury awarded the plaintiff over $4 million — well in excess of Dr. D’s policy limits.
The Follow-up
Dr. D was horrified, but the insurance company claims representative said the insurer would promptly offer $2 million in available insurance coverage to settle the case post verdict. This did not happen. Instead, the insurer chose to appeal the verdict against Dr. D’s wishes.
Ultimately, Dr. D was forced to hire his own lawyer. He ultimately sued the insurance company for breach of contract and bad faith.
The insurance company eventually attempted to settle with the plaintiffs’ counsel, but the plaintiff refused to accept the available insurance coverage. The insurance carrier still has not posted the entire appeal bond. The case is still pending.
Protecting Yourself
The lesson from Dr. D’s experience: Understand that the insurance company is not your friend. It’s a business looking out for its own interests.
The plaintiff’s attorney was absolutely correct in suggesting that Dr. D retain his own attorney to represent his own interests. You should hire your own lawyer when:
- You disagree with your insurer on how to proceed in a case.
- You receive a demand that exceeds your available insurance coverage or for damages that may not be covered by your policy, such as punitive damages.
- Your insurance carrier attempts to deny insurance coverage for your claim or sends you a letter stating that it is “reserving its rights” not to cover or to limit coverage for your claim.
Retaining independent counsel protects your interests, not those of your insurance company.
Independent counsel can give you a second opinion on the strengths and weaknesses of your claim, help you prepare for your deposition, and attend court dates with you to ensure that you are completely protected.
Independent counsel can challenge your insurance company’s decision to deny or limit your insurance coverage and ensure that you receive all of the benefits to which you are entitled under your insurance policy. Some policies may include an independent lawyer to be paid for by your insurance carrier in case of a conflicts.
The most important takeaway? Your medical malpractice insurance carrier is not your friend, so act accordingly in times of conflict.
A version of this article first appeared on Medscape.com.
You’ve been sued for medical malpractice. If you are a physician in the United States, that is not an unlikely scenario.
An analysis by the American Medical Association shows that almost half of all physicians are sued by the time they reach 54. In some specialties, such as ob.gyn., one is almost guaranteed to be sued at some point.
But that’s what medical malpractice insurance is for, right? Your medical malpractice insurer will assign an attorney to take care of you and help you through this situation. Won’t they?
Maybe so, but the attorney and the claims representative your insurer assigns to your case may have a different idea about how to proceed than you do. Though the defense attorney assigned to you represents you, he or she gets paid by the insurance carrier.
This can create a conflict when your defense counsel and your insurance claims representative aim to take your case in a direction you don’t like.
Disagreements might include:
- Choice of expert witnesses
- Tactical decisions related to trial strategy
- Public relations considerations
- Admissions of liability
- Allocation of resources
To Settle or Not?
One of the most challenging — and common — disagreements is whether to settle the case.
Sometimes a malpractice insurer wants to settle the case against the defendant doctor’s wishes. Or the doctor wants to settle but is pushed into going to trial. In the following case, one doctor had to face the consequences of a decision he didn’t even make.
The Underlying Medical Malpractice Case
Dr. D was sued by a patient who had allegedly called Dr. D’s office six times in 2 days complaining of intermittent chest pain.
Dr. D had been swamped with patients and couldn’t squeeze this patient in for an office visit, but he did call back. The patient later claimed that during the call he told the doctor he was suffering from chest pain. The doctor recalled that the patient had complained of abdominal discomfort that began after he had exercised.
The physician wrote a prescription for an ECG at the local hospital and called to ensure that the patient could just walk in. The ECG was allegedly abnormal but was not read as representing an impending or current heart attack. Later that evening, however, the patient went to the emergency department of another hospital where it was confirmed that he had suffered a heart attack. The patient underwent cardiac catheterization and stent placement to address a blockage in his left anterior descending artery.
The patient subsequently sued Dr. D and the hospital where he had the original ECG. Dr. D contacted his medical malpractice insurance company. The insurance company assigned an attorney to represent Dr. D. Discovery in the case began.
The plaintiff’s own medical expert testified in a deposition that there was no way for the heart attack to have been prevented and that the treatment would have been the same either way. But Dr. D could not find a record of the phone calls with the patient, and he had not noted his conversation the patient in their medical records.
Dr. D held a policy for $1 million, and his state had a fund that would kick in an additional $1 million. But the plaintiffs demanded $4 million to settle.
A month before trial, the plaintiff’s attorney sent a threatening letter to Dr. D’s attorney warning him that Dr. D was underinsured and suggesting that it would be in the physician’s best interests to settle.
“I want to stress to you that it is not my desire to harm your client’s reputation or to destroy his business,” wrote the plaintiff’s attorney. “However, now is the time to avoid consequences such as these by making a good faith effort to get this case resolved.”
The letter went on to note that the defense attorney should give Dr. D a copy of the letter so that everyone would be aware of the potential consequences of an award against Dr. D in excess of his limits of insurance coverage. The plaintiff’s attorney even suggested that Dr. D should retain personal counsel.
Dr. D’s defense attorney downplayed the letter and assured him that there was no reason to worry.
Meanwhile the case inched closer to trial.
The codefendant hospital settled with the plaintiff on the night before jury selection, leaving Dr. D in the uncomfortable position of being the only defendant in the case. At this point, Dr. D decided he would like to settle, and he sent his attorney an email telling him so. But the attorney instead referred him to an insurance company claims.
Just days before the trial was to start, Dr. D repeatedly told the claims representative assigned to his claim that he did not want to go to trial but rather wanted to settle. The representative told Dr. D that he had no choice in whether the action settled.
A committee at the insurance company had decided to proceed with the trial rather than settle.
The trial proved a painful debacle for Dr. D. His attorney’s idea of showing a “gotcha” video of the allegedly permanently injured plaintiff carrying a large, heavy box backfired when the jury was shown by the plaintiff that the box actually contained ice cream cones and weighed very little.
Prior to trial, the plaintiff offered to settle for $1 million. On the first day of trial, they lowered that amount to $750,000, yet the defense attorney did not settle the case, and it proceeded to a jury verdict. The jury awarded the plaintiff over $4 million — well in excess of Dr. D’s policy limits.
The Follow-up
Dr. D was horrified, but the insurance company claims representative said the insurer would promptly offer $2 million in available insurance coverage to settle the case post verdict. This did not happen. Instead, the insurer chose to appeal the verdict against Dr. D’s wishes.
Ultimately, Dr. D was forced to hire his own lawyer. He ultimately sued the insurance company for breach of contract and bad faith.
The insurance company eventually attempted to settle with the plaintiffs’ counsel, but the plaintiff refused to accept the available insurance coverage. The insurance carrier still has not posted the entire appeal bond. The case is still pending.
Protecting Yourself
The lesson from Dr. D’s experience: Understand that the insurance company is not your friend. It’s a business looking out for its own interests.
The plaintiff’s attorney was absolutely correct in suggesting that Dr. D retain his own attorney to represent his own interests. You should hire your own lawyer when:
- You disagree with your insurer on how to proceed in a case.
- You receive a demand that exceeds your available insurance coverage or for damages that may not be covered by your policy, such as punitive damages.
- Your insurance carrier attempts to deny insurance coverage for your claim or sends you a letter stating that it is “reserving its rights” not to cover or to limit coverage for your claim.
Retaining independent counsel protects your interests, not those of your insurance company.
Independent counsel can give you a second opinion on the strengths and weaknesses of your claim, help you prepare for your deposition, and attend court dates with you to ensure that you are completely protected.
Independent counsel can challenge your insurance company’s decision to deny or limit your insurance coverage and ensure that you receive all of the benefits to which you are entitled under your insurance policy. Some policies may include an independent lawyer to be paid for by your insurance carrier in case of a conflicts.
The most important takeaway? Your medical malpractice insurance carrier is not your friend, so act accordingly in times of conflict.
A version of this article first appeared on Medscape.com.
‘Reform School’ for Pharmacy Benefit Managers: How Might Legislation Help Patients?
The term “reform school” is a bit outdated. It used to refer to institutions where young offenders were sent instead of prison. Some argue that pharmacy benefit managers (PBMs) should bypass reform school and go straight to prison. “PBM reform” has become a ubiquitous term, encompassing any legislative or regulatory efforts aimed at curbing PBMs’ bad behavior. When discussing PBM reform, it’s crucial to understand the various segments of the healthcare system affected by PBMs. This complexity often makes it challenging to determine what these reform packages would actually achieve and who they would benefit.
Pharmacists have long been vocal critics of PBMs, and while their issues are extremely important, it is essential to remember that the ultimate victims of PBM misconduct, in terms of access to care, are patients. At some point, we will all be patients, making this issue universally relevant. It has been quite challenging to follow federal legislation on this topic as these packages attempt to address a number of bad behaviors by PBMs affecting a variety of victims. This discussion will examine those reforms that would directly improve patient’s access to available and affordable medications.
Policy Categories of PBM Reform
There are five policy categories of PBM reform legislation overall, including three that have the greatest potential to directly address patient needs. The first is patient access to medications (utilization management, copay assistance, prior authorization, etc.), followed by delinking drug list prices from PBM income and pass-through of price concessions from the manufacturer. The remaining two categories involve transparency and pharmacy-facing reform, both of which are very important. However, this discussion will revolve around the first three categories. It should be noted that many of the legislation packages addressing the categories of patient access, delinking, and pass-through also include transparency issues, particularly as they relate to pharmacy-facing issues.
Patient Access to Medications — Step Therapy Legislation
One of the major obstacles to patient access to medications is the use of PBM utilization management tools such as step therapy (“fail first”), prior authorizations, nonmedical switching, and formulary exclusions. These tools dictate when patients can obtain necessary medications and for how long patients who are stable on their current treatments can remain on them.
While many states have enacted step therapy reforms to prevent stable patients from being whip-sawed between medications that maximize PBM profits (often labeled as “savings”), these state protections apply only to state-regulated health plans. These include fully insured health plans and those offered through the Affordable Care Act’s Health Insurance Marketplace. It also includes state employees, state corrections, and, in some cases, state labor unions. State legislation does not extend to patients covered by employer self-insured health plans, called ERISA plans for the federal law that governs employee benefit plans, the Employee Retirement Income Security Act. These ERISA plans include nearly 35 million people nationwide.
This is where the Safe Step Act (S.652/H.R.2630) becomes crucial, as it allows employees to request exceptions to harmful fail-first protocols. The bill has gained significant momentum, having been reported out of the Senate HELP Committee and discussed in House markups. The Safe Step Act would mandate that an exception to a step therapy protocol must be granted if:
- The required treatment has been ineffective
- The treatment is expected to be ineffective, and delaying effective treatment would lead to irreversible consequences
- The treatment will cause or is likely to cause an adverse reaction
- The treatment is expected to prevent the individual from performing daily activities or occupational responsibilities
- The individual is stable on their current prescription drugs
- There are other circumstances as determined by the Employee Benefits Security Administration
This legislation is vital for ensuring that patients have timely access to the medications they need without unnecessary delays or disruptions.
Patient Access to Medications — Prior Authorizations
Another significant issue affecting patient access to medications is prior authorizations (PAs). According to an American Medical Association survey, nearly one in four physicians (24%) report that a PA has led to a serious adverse event for a patient in their care. In rheumatology, PAs often result in delays in care (even for those initially approved) and a significant increase in steroid usage. In particular, PAs in Medicare Advantage (MA) plans are harmful to Medicare beneficiaries.
The Improving Seniors’ Timely Access to Care Act (H.R.8702 / S.4532) aims to reform PAs used in MA plans, making the process more efficient and transparent to improve access to care for seniors. Unfortunately, it does not cover Part D drugs and may only cover Part B drugs depending on the MA plan’s benefit package. Here are the key provisions of the act:
- Electronic PA: Implementing real-time decisions for routinely approved items and services.
- Transparency: Requiring annual publication of PA information, such as the percentage of requests approved and the average response time.
- Quality and Timeliness Standards: The Centers for Medicare & Medicaid Services (CMS) will set standards for the quality and timeliness of PA determinations.
- Streamlining Approvals: Simplifying the approval process and reducing the time allowed for health plans to consider PA requests.
This bill passed the House in September 2022 but stalled in the Senate because of an unfavorable Congressional Budget Office score. CMS has since finalized portions of this bill via regulation, zeroing out the CBO score and increasing the chances of its passage.
Delinking Drug Prices from PBM Income and Pass-Through of Price Concessions
Affordability is a crucial aspect of accessibility, especially when it comes to medications. Over the years, we’ve learned that PBMs often favor placing the highest list price drugs on formularies because the rebates and various fees they receive from manufacturers are based on a percentage of the list price. In other words, the higher the medication’s price, the more money the PBM makes.
This practice is evident in both commercial and government formularies, where brand-name drugs are often preferred, while lower-priced generics are either excluded or placed on higher tiers. As a result, while major PBMs benefit from these rebates and fees, patients continue to pay their cost share based on the list price of the medication.
To improve the affordability of medications, a key aspect of PBM reform should be to disincentivize PBMs from selecting higher-priced medications and/or require the pass-through of manufacturer price concessions to patients.
Several major PBM reform bills are currently being considered that address either the delinking of price concessions from the list price of the drug or some form of pass-through of these concessions. These reforms are essential to ensure that patients can access affordable medications without being burdened by inflated costs.
The legislation includes the Pharmacy Benefit Manager Reform Act (S.1339); the Modernizing & Ensuring PBM Accountability Act (S.2973); the Better Mental Health Care, Lower Cost Drugs, and Extenders Act (S.3430); the Protecting Patients Against PBM Abuses Act (H.R. 2880); the DRUG Act (S.2474 / H.R.6283); and the Share the Savings with Seniors Act (S.2474 / H.R.5376).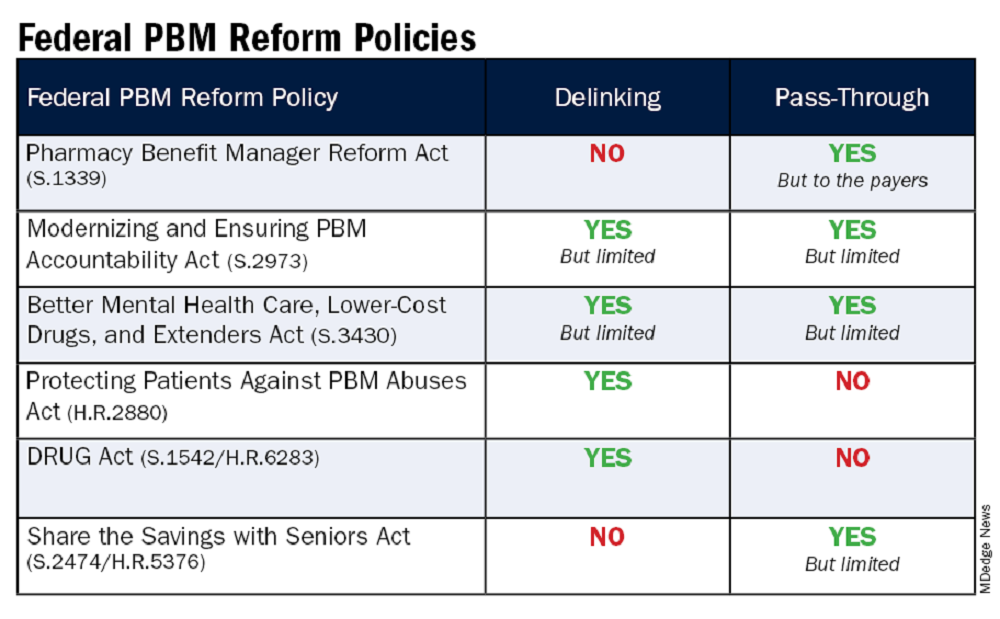
As with all legislation, there are limitations and compromises in each of these. However, these bills are a good first step in addressing PBM remuneration (rebates and fees) based on the list price of the drug and/or passing through to the patient the benefit of manufacturer price concessions. By focusing on key areas like utilization management, delinking drug prices from PBM income, and allowing patients to directly benefit from manufacturer price concessions, we can work toward a more equitable and efficient healthcare system. Reigning in PBM bad behavior is a challenge, but the potential benefits for patient care and access make it a crucial fight worth pursuing.
Please help in efforts to improve patients’ access to available and affordable medications by contacting your representatives in Congress to impart to them the importance of passing legislation. The CSRO’s legislative map tool can help to inform you of the latest information on these and other bills and assist you in engaging with your representatives on them.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of Advocacy and Government Affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. She has no relevant conflicts of interest to disclose. You can reach her at [email protected].
The term “reform school” is a bit outdated. It used to refer to institutions where young offenders were sent instead of prison. Some argue that pharmacy benefit managers (PBMs) should bypass reform school and go straight to prison. “PBM reform” has become a ubiquitous term, encompassing any legislative or regulatory efforts aimed at curbing PBMs’ bad behavior. When discussing PBM reform, it’s crucial to understand the various segments of the healthcare system affected by PBMs. This complexity often makes it challenging to determine what these reform packages would actually achieve and who they would benefit.
Pharmacists have long been vocal critics of PBMs, and while their issues are extremely important, it is essential to remember that the ultimate victims of PBM misconduct, in terms of access to care, are patients. At some point, we will all be patients, making this issue universally relevant. It has been quite challenging to follow federal legislation on this topic as these packages attempt to address a number of bad behaviors by PBMs affecting a variety of victims. This discussion will examine those reforms that would directly improve patient’s access to available and affordable medications.
Policy Categories of PBM Reform
There are five policy categories of PBM reform legislation overall, including three that have the greatest potential to directly address patient needs. The first is patient access to medications (utilization management, copay assistance, prior authorization, etc.), followed by delinking drug list prices from PBM income and pass-through of price concessions from the manufacturer. The remaining two categories involve transparency and pharmacy-facing reform, both of which are very important. However, this discussion will revolve around the first three categories. It should be noted that many of the legislation packages addressing the categories of patient access, delinking, and pass-through also include transparency issues, particularly as they relate to pharmacy-facing issues.
Patient Access to Medications — Step Therapy Legislation
One of the major obstacles to patient access to medications is the use of PBM utilization management tools such as step therapy (“fail first”), prior authorizations, nonmedical switching, and formulary exclusions. These tools dictate when patients can obtain necessary medications and for how long patients who are stable on their current treatments can remain on them.
While many states have enacted step therapy reforms to prevent stable patients from being whip-sawed between medications that maximize PBM profits (often labeled as “savings”), these state protections apply only to state-regulated health plans. These include fully insured health plans and those offered through the Affordable Care Act’s Health Insurance Marketplace. It also includes state employees, state corrections, and, in some cases, state labor unions. State legislation does not extend to patients covered by employer self-insured health plans, called ERISA plans for the federal law that governs employee benefit plans, the Employee Retirement Income Security Act. These ERISA plans include nearly 35 million people nationwide.
This is where the Safe Step Act (S.652/H.R.2630) becomes crucial, as it allows employees to request exceptions to harmful fail-first protocols. The bill has gained significant momentum, having been reported out of the Senate HELP Committee and discussed in House markups. The Safe Step Act would mandate that an exception to a step therapy protocol must be granted if:
- The required treatment has been ineffective
- The treatment is expected to be ineffective, and delaying effective treatment would lead to irreversible consequences
- The treatment will cause or is likely to cause an adverse reaction
- The treatment is expected to prevent the individual from performing daily activities or occupational responsibilities
- The individual is stable on their current prescription drugs
- There are other circumstances as determined by the Employee Benefits Security Administration
This legislation is vital for ensuring that patients have timely access to the medications they need without unnecessary delays or disruptions.
Patient Access to Medications — Prior Authorizations
Another significant issue affecting patient access to medications is prior authorizations (PAs). According to an American Medical Association survey, nearly one in four physicians (24%) report that a PA has led to a serious adverse event for a patient in their care. In rheumatology, PAs often result in delays in care (even for those initially approved) and a significant increase in steroid usage. In particular, PAs in Medicare Advantage (MA) plans are harmful to Medicare beneficiaries.
The Improving Seniors’ Timely Access to Care Act (H.R.8702 / S.4532) aims to reform PAs used in MA plans, making the process more efficient and transparent to improve access to care for seniors. Unfortunately, it does not cover Part D drugs and may only cover Part B drugs depending on the MA plan’s benefit package. Here are the key provisions of the act:
- Electronic PA: Implementing real-time decisions for routinely approved items and services.
- Transparency: Requiring annual publication of PA information, such as the percentage of requests approved and the average response time.
- Quality and Timeliness Standards: The Centers for Medicare & Medicaid Services (CMS) will set standards for the quality and timeliness of PA determinations.
- Streamlining Approvals: Simplifying the approval process and reducing the time allowed for health plans to consider PA requests.
This bill passed the House in September 2022 but stalled in the Senate because of an unfavorable Congressional Budget Office score. CMS has since finalized portions of this bill via regulation, zeroing out the CBO score and increasing the chances of its passage.
Delinking Drug Prices from PBM Income and Pass-Through of Price Concessions
Affordability is a crucial aspect of accessibility, especially when it comes to medications. Over the years, we’ve learned that PBMs often favor placing the highest list price drugs on formularies because the rebates and various fees they receive from manufacturers are based on a percentage of the list price. In other words, the higher the medication’s price, the more money the PBM makes.
This practice is evident in both commercial and government formularies, where brand-name drugs are often preferred, while lower-priced generics are either excluded or placed on higher tiers. As a result, while major PBMs benefit from these rebates and fees, patients continue to pay their cost share based on the list price of the medication.
To improve the affordability of medications, a key aspect of PBM reform should be to disincentivize PBMs from selecting higher-priced medications and/or require the pass-through of manufacturer price concessions to patients.
Several major PBM reform bills are currently being considered that address either the delinking of price concessions from the list price of the drug or some form of pass-through of these concessions. These reforms are essential to ensure that patients can access affordable medications without being burdened by inflated costs.
The legislation includes the Pharmacy Benefit Manager Reform Act (S.1339); the Modernizing & Ensuring PBM Accountability Act (S.2973); the Better Mental Health Care, Lower Cost Drugs, and Extenders Act (S.3430); the Protecting Patients Against PBM Abuses Act (H.R. 2880); the DRUG Act (S.2474 / H.R.6283); and the Share the Savings with Seniors Act (S.2474 / H.R.5376).
As with all legislation, there are limitations and compromises in each of these. However, these bills are a good first step in addressing PBM remuneration (rebates and fees) based on the list price of the drug and/or passing through to the patient the benefit of manufacturer price concessions. By focusing on key areas like utilization management, delinking drug prices from PBM income, and allowing patients to directly benefit from manufacturer price concessions, we can work toward a more equitable and efficient healthcare system. Reigning in PBM bad behavior is a challenge, but the potential benefits for patient care and access make it a crucial fight worth pursuing.
Please help in efforts to improve patients’ access to available and affordable medications by contacting your representatives in Congress to impart to them the importance of passing legislation. The CSRO’s legislative map tool can help to inform you of the latest information on these and other bills and assist you in engaging with your representatives on them.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of Advocacy and Government Affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. She has no relevant conflicts of interest to disclose. You can reach her at [email protected].
The term “reform school” is a bit outdated. It used to refer to institutions where young offenders were sent instead of prison. Some argue that pharmacy benefit managers (PBMs) should bypass reform school and go straight to prison. “PBM reform” has become a ubiquitous term, encompassing any legislative or regulatory efforts aimed at curbing PBMs’ bad behavior. When discussing PBM reform, it’s crucial to understand the various segments of the healthcare system affected by PBMs. This complexity often makes it challenging to determine what these reform packages would actually achieve and who they would benefit.
Pharmacists have long been vocal critics of PBMs, and while their issues are extremely important, it is essential to remember that the ultimate victims of PBM misconduct, in terms of access to care, are patients. At some point, we will all be patients, making this issue universally relevant. It has been quite challenging to follow federal legislation on this topic as these packages attempt to address a number of bad behaviors by PBMs affecting a variety of victims. This discussion will examine those reforms that would directly improve patient’s access to available and affordable medications.
Policy Categories of PBM Reform
There are five policy categories of PBM reform legislation overall, including three that have the greatest potential to directly address patient needs. The first is patient access to medications (utilization management, copay assistance, prior authorization, etc.), followed by delinking drug list prices from PBM income and pass-through of price concessions from the manufacturer. The remaining two categories involve transparency and pharmacy-facing reform, both of which are very important. However, this discussion will revolve around the first three categories. It should be noted that many of the legislation packages addressing the categories of patient access, delinking, and pass-through also include transparency issues, particularly as they relate to pharmacy-facing issues.
Patient Access to Medications — Step Therapy Legislation
One of the major obstacles to patient access to medications is the use of PBM utilization management tools such as step therapy (“fail first”), prior authorizations, nonmedical switching, and formulary exclusions. These tools dictate when patients can obtain necessary medications and for how long patients who are stable on their current treatments can remain on them.
While many states have enacted step therapy reforms to prevent stable patients from being whip-sawed between medications that maximize PBM profits (often labeled as “savings”), these state protections apply only to state-regulated health plans. These include fully insured health plans and those offered through the Affordable Care Act’s Health Insurance Marketplace. It also includes state employees, state corrections, and, in some cases, state labor unions. State legislation does not extend to patients covered by employer self-insured health plans, called ERISA plans for the federal law that governs employee benefit plans, the Employee Retirement Income Security Act. These ERISA plans include nearly 35 million people nationwide.
This is where the Safe Step Act (S.652/H.R.2630) becomes crucial, as it allows employees to request exceptions to harmful fail-first protocols. The bill has gained significant momentum, having been reported out of the Senate HELP Committee and discussed in House markups. The Safe Step Act would mandate that an exception to a step therapy protocol must be granted if:
- The required treatment has been ineffective
- The treatment is expected to be ineffective, and delaying effective treatment would lead to irreversible consequences
- The treatment will cause or is likely to cause an adverse reaction
- The treatment is expected to prevent the individual from performing daily activities or occupational responsibilities
- The individual is stable on their current prescription drugs
- There are other circumstances as determined by the Employee Benefits Security Administration
This legislation is vital for ensuring that patients have timely access to the medications they need without unnecessary delays or disruptions.
Patient Access to Medications — Prior Authorizations
Another significant issue affecting patient access to medications is prior authorizations (PAs). According to an American Medical Association survey, nearly one in four physicians (24%) report that a PA has led to a serious adverse event for a patient in their care. In rheumatology, PAs often result in delays in care (even for those initially approved) and a significant increase in steroid usage. In particular, PAs in Medicare Advantage (MA) plans are harmful to Medicare beneficiaries.
The Improving Seniors’ Timely Access to Care Act (H.R.8702 / S.4532) aims to reform PAs used in MA plans, making the process more efficient and transparent to improve access to care for seniors. Unfortunately, it does not cover Part D drugs and may only cover Part B drugs depending on the MA plan’s benefit package. Here are the key provisions of the act:
- Electronic PA: Implementing real-time decisions for routinely approved items and services.
- Transparency: Requiring annual publication of PA information, such as the percentage of requests approved and the average response time.
- Quality and Timeliness Standards: The Centers for Medicare & Medicaid Services (CMS) will set standards for the quality and timeliness of PA determinations.
- Streamlining Approvals: Simplifying the approval process and reducing the time allowed for health plans to consider PA requests.
This bill passed the House in September 2022 but stalled in the Senate because of an unfavorable Congressional Budget Office score. CMS has since finalized portions of this bill via regulation, zeroing out the CBO score and increasing the chances of its passage.
Delinking Drug Prices from PBM Income and Pass-Through of Price Concessions
Affordability is a crucial aspect of accessibility, especially when it comes to medications. Over the years, we’ve learned that PBMs often favor placing the highest list price drugs on formularies because the rebates and various fees they receive from manufacturers are based on a percentage of the list price. In other words, the higher the medication’s price, the more money the PBM makes.
This practice is evident in both commercial and government formularies, where brand-name drugs are often preferred, while lower-priced generics are either excluded or placed on higher tiers. As a result, while major PBMs benefit from these rebates and fees, patients continue to pay their cost share based on the list price of the medication.
To improve the affordability of medications, a key aspect of PBM reform should be to disincentivize PBMs from selecting higher-priced medications and/or require the pass-through of manufacturer price concessions to patients.
Several major PBM reform bills are currently being considered that address either the delinking of price concessions from the list price of the drug or some form of pass-through of these concessions. These reforms are essential to ensure that patients can access affordable medications without being burdened by inflated costs.
The legislation includes the Pharmacy Benefit Manager Reform Act (S.1339); the Modernizing & Ensuring PBM Accountability Act (S.2973); the Better Mental Health Care, Lower Cost Drugs, and Extenders Act (S.3430); the Protecting Patients Against PBM Abuses Act (H.R. 2880); the DRUG Act (S.2474 / H.R.6283); and the Share the Savings with Seniors Act (S.2474 / H.R.5376).
As with all legislation, there are limitations and compromises in each of these. However, these bills are a good first step in addressing PBM remuneration (rebates and fees) based on the list price of the drug and/or passing through to the patient the benefit of manufacturer price concessions. By focusing on key areas like utilization management, delinking drug prices from PBM income, and allowing patients to directly benefit from manufacturer price concessions, we can work toward a more equitable and efficient healthcare system. Reigning in PBM bad behavior is a challenge, but the potential benefits for patient care and access make it a crucial fight worth pursuing.
Please help in efforts to improve patients’ access to available and affordable medications by contacting your representatives in Congress to impart to them the importance of passing legislation. The CSRO’s legislative map tool can help to inform you of the latest information on these and other bills and assist you in engaging with your representatives on them.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of Advocacy and Government Affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. She has no relevant conflicts of interest to disclose. You can reach her at [email protected].
FDA Okays Subcutaneous Ocrelizumab for MS
The subcutaneous (SC) injection can be administered by a healthcare professional in approximately 10 minutes and is the first and only twice-a-year SC injection approved for both RMS and PPMS, according to a company news release.
The FDA approval is based on pivotal data from the phase 3 OCARINA II trial, which showed no clinically significant difference in blood levels of ocrelizumab when administered subcutaneously and an efficacy profile consistent with the intravenous (IV) formulation.
“The trial met its primary and secondary endpoints, demonstrating SC injection was noninferior to IV infusion based on [ocrelizumab] levels in the blood, and consistent control of clinical (relapses) and radiological (MRI lesions) disease activity,” the company said in the release.
The safety profile of SC ocrelizumab was consistent with the safety profile of IV ocrelizumab, with the exception of injection site reactions, the most common adverse event.
Injection reactions were more often reported with the first injection, with 49% of trial participants experiencing an injection reaction after the first injection. All injection reactions were mild or moderate, and none led to treatment withdrawal.
Ocrevus Zunovo “may offer greater flexibility for healthcare providers and people living with multiple sclerosis, based on their individual treatment needs,” Levi Garraway, MD, PhD, chief medical officer for Genentech, said in the press release. “We are pleased that with a new method of delivery, there is now an additional option for those who need flexibility in the route of administration or treatment time,” Natalie Blake, executive director of the MS Foundation, said in the release.
The SC formulation of ocrelizumab was approved by the European Commission in June.
Complete prescribing information is available online.
A version of this article appeared on Medscape.com.
The subcutaneous (SC) injection can be administered by a healthcare professional in approximately 10 minutes and is the first and only twice-a-year SC injection approved for both RMS and PPMS, according to a company news release.
The FDA approval is based on pivotal data from the phase 3 OCARINA II trial, which showed no clinically significant difference in blood levels of ocrelizumab when administered subcutaneously and an efficacy profile consistent with the intravenous (IV) formulation.
“The trial met its primary and secondary endpoints, demonstrating SC injection was noninferior to IV infusion based on [ocrelizumab] levels in the blood, and consistent control of clinical (relapses) and radiological (MRI lesions) disease activity,” the company said in the release.
The safety profile of SC ocrelizumab was consistent with the safety profile of IV ocrelizumab, with the exception of injection site reactions, the most common adverse event.
Injection reactions were more often reported with the first injection, with 49% of trial participants experiencing an injection reaction after the first injection. All injection reactions were mild or moderate, and none led to treatment withdrawal.
Ocrevus Zunovo “may offer greater flexibility for healthcare providers and people living with multiple sclerosis, based on their individual treatment needs,” Levi Garraway, MD, PhD, chief medical officer for Genentech, said in the press release. “We are pleased that with a new method of delivery, there is now an additional option for those who need flexibility in the route of administration or treatment time,” Natalie Blake, executive director of the MS Foundation, said in the release.
The SC formulation of ocrelizumab was approved by the European Commission in June.
Complete prescribing information is available online.
A version of this article appeared on Medscape.com.
The subcutaneous (SC) injection can be administered by a healthcare professional in approximately 10 minutes and is the first and only twice-a-year SC injection approved for both RMS and PPMS, according to a company news release.
The FDA approval is based on pivotal data from the phase 3 OCARINA II trial, which showed no clinically significant difference in blood levels of ocrelizumab when administered subcutaneously and an efficacy profile consistent with the intravenous (IV) formulation.
“The trial met its primary and secondary endpoints, demonstrating SC injection was noninferior to IV infusion based on [ocrelizumab] levels in the blood, and consistent control of clinical (relapses) and radiological (MRI lesions) disease activity,” the company said in the release.
The safety profile of SC ocrelizumab was consistent with the safety profile of IV ocrelizumab, with the exception of injection site reactions, the most common adverse event.
Injection reactions were more often reported with the first injection, with 49% of trial participants experiencing an injection reaction after the first injection. All injection reactions were mild or moderate, and none led to treatment withdrawal.
Ocrevus Zunovo “may offer greater flexibility for healthcare providers and people living with multiple sclerosis, based on their individual treatment needs,” Levi Garraway, MD, PhD, chief medical officer for Genentech, said in the press release. “We are pleased that with a new method of delivery, there is now an additional option for those who need flexibility in the route of administration or treatment time,” Natalie Blake, executive director of the MS Foundation, said in the release.
The SC formulation of ocrelizumab was approved by the European Commission in June.
Complete prescribing information is available online.
A version of this article appeared on Medscape.com.
SGLT2 Inhibitor Reduces Risk for Neurodegenerative Diseases in T2D
MADRID — Patients with type 2 diabetes treated with sodium-glucose cotransporter 2 inhibitors (SGLT2is) show significant reductions in the risk of developing neurodegenerative disorders including Alzheimer’s disease, vascular dementia, and Parkinson’s disease, compared with those treated with other antidiabetic drugs, results from a large population-based cohort show.
“This was the largest nationwide population-based longitudinal cohort study to investigate the association between the use of SGLT2 inhibitors and the incidence of all-cause dementia and Parkinson’s disease,” said first author Hae Kyung Kim, MD, of the Department of Internal Medicine, Yonsei University College of Medicine, Seoul, South Korea, in presenting the findings at the annual meeting of the European Association for the Study of Diabetes.
Type 2 diabetes is known to increase the risk for neurodegenerative diseases such as dementia or Alzheimer’s disease, said Dr. Kim. Key factors attributed to the risk include shared pathophysiological mechanisms such as central nervous system insulin resistance and reduced cerebral glucose metabolism.
While research is lacking on the role of antidiabetic drugs in the treatment of neurodegenerative diseases, the researcher noted that “SGLT2 inhibitors, which have shown significant cardiorenal benefits and enhanced energy metabolism through ketogenesis, offer promise.”
To further investigate, Dr. Kim and her colleagues conducted the retrospective study, evaluating data on more than 1.3 million enrollees in Korea’s National Health Insurance Service Database who were aged 40 years or older, diagnosed with type 2 diabetes, and had initiated antidiabetic drugs between September 2014 and December 2019.
In the propensity score analysis, 358,862 patients were matched 1:1, in groups of 179,431 participants each, based on whether they were treated with SGLT2is or other oral antidiabetic drugs. Patients with a history of neurodegenerative disease, cancer, or use of glucagon-like peptide 1 receptor agonists were excluded.
The patients had a mean age of 57.8 years, 57.9% were men, and 6837 had incident dementia or Parkinson’s disease events reported.
With a mean follow-up of 2.88 years, after adjustment for key variables, those treated with SGLT2is had a 19% reduced risk of developing Alzheimer’s disease (adjusted hazard ratio [aHR], 0.81), a 31% reduced risk for vascular dementia (aHR, 0.69), and a 20% reduced risk for Parkinson’s disease (aHR, 0.80) compared with the non-SGLT2i group.
Furthermore, those receiving SGLT2i treatment had a 21% reduced risk for all-cause dementia (aHR, 0.79) and a 22% reduced risk for all-cause dementia and Parkinson’s disease compared with the oral antidiabetic drug group (aHR, 0.78) with a 6-month drug use lag period.
The association was observed regardless of SGLT2i exposure duration. Subgroup analyses indicated that the reductions in neurodegenerative disorders among those receiving SGLT2is were not associated with factors including age, sex, body mass index, blood pressure, glucose, lipid profiles, kidney function, health behaviors, comorbidities, diabetic complications, or other medication use.
Dr. Kim speculated that mechanisms underlying the reduced dementia risk could include SGLT2i effects of mitigating the common severe risk factors of type 2 diabetes and neurodegenerative diseases, including hypertension, heart failure, and chronic kidney disease, and improving hyperperfusion in the heart and cerebral vascular insufficiency.
Commenting on the study to this news organization, Erik H. Serné, MD, of the VU University Medical Centre, Amsterdam, the Netherlands, who comoderated the session, noted that “people with type 2 diabetes have a 50%-100% increased risk of developing dementia, particularly Alzheimer’s disease and vascular dementia.”
“The increasing prevalence of both conditions poses significant public health challenges, highlighting the need for effective prevention strategies and interventions.”
Currently, treatments for dementia are limited, with most primarily addressing symptoms and not the underlying cause of the neurodegenerative disease, he said.
He noted that, in addition to the effects mentioned by Dr. Kim, SGLT2is are also speculated to provide potential neuroprotective effects through improved glycemic control and insulin sensitivity, reduced inflammation and oxidative stress, enhanced mitochondrial function and energy metabolism, and reduced beta-amyloid and tau pathology.
“These mechanisms collectively may reduce the risk of cognitive decline, particularly in diabetic patients, and warrant further investigation in clinical trials to solidify the neuroprotective role of SGLT2 inhibitors,” said Dr. Serné.
In addition to their benefits in type 2 diabetes, SGLT2is “now offer hope in the prevention of dementia, a disease that has very limited therapeutic options thus far. The current data [presented by Dr. Kim] seem to corroborate this,” he added.
Dr. Kim and Dr. Serné had no disclosures to report.
A version of this article first appeared on Medscape.com.
MADRID — Patients with type 2 diabetes treated with sodium-glucose cotransporter 2 inhibitors (SGLT2is) show significant reductions in the risk of developing neurodegenerative disorders including Alzheimer’s disease, vascular dementia, and Parkinson’s disease, compared with those treated with other antidiabetic drugs, results from a large population-based cohort show.
“This was the largest nationwide population-based longitudinal cohort study to investigate the association between the use of SGLT2 inhibitors and the incidence of all-cause dementia and Parkinson’s disease,” said first author Hae Kyung Kim, MD, of the Department of Internal Medicine, Yonsei University College of Medicine, Seoul, South Korea, in presenting the findings at the annual meeting of the European Association for the Study of Diabetes.
Type 2 diabetes is known to increase the risk for neurodegenerative diseases such as dementia or Alzheimer’s disease, said Dr. Kim. Key factors attributed to the risk include shared pathophysiological mechanisms such as central nervous system insulin resistance and reduced cerebral glucose metabolism.
While research is lacking on the role of antidiabetic drugs in the treatment of neurodegenerative diseases, the researcher noted that “SGLT2 inhibitors, which have shown significant cardiorenal benefits and enhanced energy metabolism through ketogenesis, offer promise.”
To further investigate, Dr. Kim and her colleagues conducted the retrospective study, evaluating data on more than 1.3 million enrollees in Korea’s National Health Insurance Service Database who were aged 40 years or older, diagnosed with type 2 diabetes, and had initiated antidiabetic drugs between September 2014 and December 2019.
In the propensity score analysis, 358,862 patients were matched 1:1, in groups of 179,431 participants each, based on whether they were treated with SGLT2is or other oral antidiabetic drugs. Patients with a history of neurodegenerative disease, cancer, or use of glucagon-like peptide 1 receptor agonists were excluded.
The patients had a mean age of 57.8 years, 57.9% were men, and 6837 had incident dementia or Parkinson’s disease events reported.
With a mean follow-up of 2.88 years, after adjustment for key variables, those treated with SGLT2is had a 19% reduced risk of developing Alzheimer’s disease (adjusted hazard ratio [aHR], 0.81), a 31% reduced risk for vascular dementia (aHR, 0.69), and a 20% reduced risk for Parkinson’s disease (aHR, 0.80) compared with the non-SGLT2i group.
Furthermore, those receiving SGLT2i treatment had a 21% reduced risk for all-cause dementia (aHR, 0.79) and a 22% reduced risk for all-cause dementia and Parkinson’s disease compared with the oral antidiabetic drug group (aHR, 0.78) with a 6-month drug use lag period.
The association was observed regardless of SGLT2i exposure duration. Subgroup analyses indicated that the reductions in neurodegenerative disorders among those receiving SGLT2is were not associated with factors including age, sex, body mass index, blood pressure, glucose, lipid profiles, kidney function, health behaviors, comorbidities, diabetic complications, or other medication use.
Dr. Kim speculated that mechanisms underlying the reduced dementia risk could include SGLT2i effects of mitigating the common severe risk factors of type 2 diabetes and neurodegenerative diseases, including hypertension, heart failure, and chronic kidney disease, and improving hyperperfusion in the heart and cerebral vascular insufficiency.
Commenting on the study to this news organization, Erik H. Serné, MD, of the VU University Medical Centre, Amsterdam, the Netherlands, who comoderated the session, noted that “people with type 2 diabetes have a 50%-100% increased risk of developing dementia, particularly Alzheimer’s disease and vascular dementia.”
“The increasing prevalence of both conditions poses significant public health challenges, highlighting the need for effective prevention strategies and interventions.”
Currently, treatments for dementia are limited, with most primarily addressing symptoms and not the underlying cause of the neurodegenerative disease, he said.
He noted that, in addition to the effects mentioned by Dr. Kim, SGLT2is are also speculated to provide potential neuroprotective effects through improved glycemic control and insulin sensitivity, reduced inflammation and oxidative stress, enhanced mitochondrial function and energy metabolism, and reduced beta-amyloid and tau pathology.
“These mechanisms collectively may reduce the risk of cognitive decline, particularly in diabetic patients, and warrant further investigation in clinical trials to solidify the neuroprotective role of SGLT2 inhibitors,” said Dr. Serné.
In addition to their benefits in type 2 diabetes, SGLT2is “now offer hope in the prevention of dementia, a disease that has very limited therapeutic options thus far. The current data [presented by Dr. Kim] seem to corroborate this,” he added.
Dr. Kim and Dr. Serné had no disclosures to report.
A version of this article first appeared on Medscape.com.
MADRID — Patients with type 2 diabetes treated with sodium-glucose cotransporter 2 inhibitors (SGLT2is) show significant reductions in the risk of developing neurodegenerative disorders including Alzheimer’s disease, vascular dementia, and Parkinson’s disease, compared with those treated with other antidiabetic drugs, results from a large population-based cohort show.
“This was the largest nationwide population-based longitudinal cohort study to investigate the association between the use of SGLT2 inhibitors and the incidence of all-cause dementia and Parkinson’s disease,” said first author Hae Kyung Kim, MD, of the Department of Internal Medicine, Yonsei University College of Medicine, Seoul, South Korea, in presenting the findings at the annual meeting of the European Association for the Study of Diabetes.
Type 2 diabetes is known to increase the risk for neurodegenerative diseases such as dementia or Alzheimer’s disease, said Dr. Kim. Key factors attributed to the risk include shared pathophysiological mechanisms such as central nervous system insulin resistance and reduced cerebral glucose metabolism.
While research is lacking on the role of antidiabetic drugs in the treatment of neurodegenerative diseases, the researcher noted that “SGLT2 inhibitors, which have shown significant cardiorenal benefits and enhanced energy metabolism through ketogenesis, offer promise.”
To further investigate, Dr. Kim and her colleagues conducted the retrospective study, evaluating data on more than 1.3 million enrollees in Korea’s National Health Insurance Service Database who were aged 40 years or older, diagnosed with type 2 diabetes, and had initiated antidiabetic drugs between September 2014 and December 2019.
In the propensity score analysis, 358,862 patients were matched 1:1, in groups of 179,431 participants each, based on whether they were treated with SGLT2is or other oral antidiabetic drugs. Patients with a history of neurodegenerative disease, cancer, or use of glucagon-like peptide 1 receptor agonists were excluded.
The patients had a mean age of 57.8 years, 57.9% were men, and 6837 had incident dementia or Parkinson’s disease events reported.
With a mean follow-up of 2.88 years, after adjustment for key variables, those treated with SGLT2is had a 19% reduced risk of developing Alzheimer’s disease (adjusted hazard ratio [aHR], 0.81), a 31% reduced risk for vascular dementia (aHR, 0.69), and a 20% reduced risk for Parkinson’s disease (aHR, 0.80) compared with the non-SGLT2i group.
Furthermore, those receiving SGLT2i treatment had a 21% reduced risk for all-cause dementia (aHR, 0.79) and a 22% reduced risk for all-cause dementia and Parkinson’s disease compared with the oral antidiabetic drug group (aHR, 0.78) with a 6-month drug use lag period.
The association was observed regardless of SGLT2i exposure duration. Subgroup analyses indicated that the reductions in neurodegenerative disorders among those receiving SGLT2is were not associated with factors including age, sex, body mass index, blood pressure, glucose, lipid profiles, kidney function, health behaviors, comorbidities, diabetic complications, or other medication use.
Dr. Kim speculated that mechanisms underlying the reduced dementia risk could include SGLT2i effects of mitigating the common severe risk factors of type 2 diabetes and neurodegenerative diseases, including hypertension, heart failure, and chronic kidney disease, and improving hyperperfusion in the heart and cerebral vascular insufficiency.
Commenting on the study to this news organization, Erik H. Serné, MD, of the VU University Medical Centre, Amsterdam, the Netherlands, who comoderated the session, noted that “people with type 2 diabetes have a 50%-100% increased risk of developing dementia, particularly Alzheimer’s disease and vascular dementia.”
“The increasing prevalence of both conditions poses significant public health challenges, highlighting the need for effective prevention strategies and interventions.”
Currently, treatments for dementia are limited, with most primarily addressing symptoms and not the underlying cause of the neurodegenerative disease, he said.
He noted that, in addition to the effects mentioned by Dr. Kim, SGLT2is are also speculated to provide potential neuroprotective effects through improved glycemic control and insulin sensitivity, reduced inflammation and oxidative stress, enhanced mitochondrial function and energy metabolism, and reduced beta-amyloid and tau pathology.
“These mechanisms collectively may reduce the risk of cognitive decline, particularly in diabetic patients, and warrant further investigation in clinical trials to solidify the neuroprotective role of SGLT2 inhibitors,” said Dr. Serné.
In addition to their benefits in type 2 diabetes, SGLT2is “now offer hope in the prevention of dementia, a disease that has very limited therapeutic options thus far. The current data [presented by Dr. Kim] seem to corroborate this,” he added.
Dr. Kim and Dr. Serné had no disclosures to report.
A version of this article first appeared on Medscape.com.
FROM EASD 2024
Mom’s Potato Salad
Outside of caffeine, I have very few addictions. One of them is “Midnight Diner.”
“Midnight Diner” is a quirky, sometimes funny, sometimes bittersweet, Japanese series on Netflix. It’s about a small diner in Tokyo, open only in the wee hours of the morning, its enigmatic owner/cook, and the eclectic patrons that come and go. Each is seeking a dish that means something to them.
One episode (spoiler alert, in case you’re planning to watch it) deals with the regulars realizing a fellow who frequently comes in and orders potato salad is secretly Japan’s most famous porn actor, Erect Oki. This revelation garners him the respect, awe, and envy of the other male patrons, though Mr. Oki would rather be left to his potato salad.
The jokes are there ... but as things develop, we learn he has the potato salad because it reminds him of his mother’s potato salad — and that he’s been cut off from his family for more than 20 years because of his career path. The potato salad is all he has left.
While preparing for a shoot, he learns his mother has Alzheimer’s disease, and immediately returns home. As they sit talking on the patio of a care center, she tells him about her son, who lives in Tokyo, and loves her potato salad. The show doesn’t make it clear if she ever remembers who he is.
In the darkening hallways of her mind, she asks his sister for help in making potato salad for her visitor. It’s too salty, though whether this is from the ingredients or his tears is also never stated.
The episode is a poignant reminder of how Alzheimer’s disease is a worldwide human problem. Not American. Not western. Not restricted by race, or ethnicity, or continent. It effects us all as a species, as families, and as individuals. No matter what our jobs or backgrounds are.
For those of us on this side of the desk, it’s a reminder that Yes, we have all kinds of new toys, but from a practical viewpoint it’s hard to say that we’ve made any major advances. I’m sure my drug reps will disagree with me, and I’m not saying any of the treatments of the last 28 years are worthless, but even now we’re still far from a cure, or even something that stops progression.
That’s not from lack of trying, either.
For all the jokes about his job, Mr. Oki is no different from any other children trying to hold onto their parents as the disease slowly takes them away.
I hope we have real answers, soon.
Dr. Block has a solo neurology practice in Scottsdale, Arizona.
Outside of caffeine, I have very few addictions. One of them is “Midnight Diner.”
“Midnight Diner” is a quirky, sometimes funny, sometimes bittersweet, Japanese series on Netflix. It’s about a small diner in Tokyo, open only in the wee hours of the morning, its enigmatic owner/cook, and the eclectic patrons that come and go. Each is seeking a dish that means something to them.
One episode (spoiler alert, in case you’re planning to watch it) deals with the regulars realizing a fellow who frequently comes in and orders potato salad is secretly Japan’s most famous porn actor, Erect Oki. This revelation garners him the respect, awe, and envy of the other male patrons, though Mr. Oki would rather be left to his potato salad.
The jokes are there ... but as things develop, we learn he has the potato salad because it reminds him of his mother’s potato salad — and that he’s been cut off from his family for more than 20 years because of his career path. The potato salad is all he has left.
While preparing for a shoot, he learns his mother has Alzheimer’s disease, and immediately returns home. As they sit talking on the patio of a care center, she tells him about her son, who lives in Tokyo, and loves her potato salad. The show doesn’t make it clear if she ever remembers who he is.
In the darkening hallways of her mind, she asks his sister for help in making potato salad for her visitor. It’s too salty, though whether this is from the ingredients or his tears is also never stated.
The episode is a poignant reminder of how Alzheimer’s disease is a worldwide human problem. Not American. Not western. Not restricted by race, or ethnicity, or continent. It effects us all as a species, as families, and as individuals. No matter what our jobs or backgrounds are.
For those of us on this side of the desk, it’s a reminder that Yes, we have all kinds of new toys, but from a practical viewpoint it’s hard to say that we’ve made any major advances. I’m sure my drug reps will disagree with me, and I’m not saying any of the treatments of the last 28 years are worthless, but even now we’re still far from a cure, or even something that stops progression.
That’s not from lack of trying, either.
For all the jokes about his job, Mr. Oki is no different from any other children trying to hold onto their parents as the disease slowly takes them away.
I hope we have real answers, soon.
Dr. Block has a solo neurology practice in Scottsdale, Arizona.
Outside of caffeine, I have very few addictions. One of them is “Midnight Diner.”
“Midnight Diner” is a quirky, sometimes funny, sometimes bittersweet, Japanese series on Netflix. It’s about a small diner in Tokyo, open only in the wee hours of the morning, its enigmatic owner/cook, and the eclectic patrons that come and go. Each is seeking a dish that means something to them.
One episode (spoiler alert, in case you’re planning to watch it) deals with the regulars realizing a fellow who frequently comes in and orders potato salad is secretly Japan’s most famous porn actor, Erect Oki. This revelation garners him the respect, awe, and envy of the other male patrons, though Mr. Oki would rather be left to his potato salad.
The jokes are there ... but as things develop, we learn he has the potato salad because it reminds him of his mother’s potato salad — and that he’s been cut off from his family for more than 20 years because of his career path. The potato salad is all he has left.
While preparing for a shoot, he learns his mother has Alzheimer’s disease, and immediately returns home. As they sit talking on the patio of a care center, she tells him about her son, who lives in Tokyo, and loves her potato salad. The show doesn’t make it clear if she ever remembers who he is.
In the darkening hallways of her mind, she asks his sister for help in making potato salad for her visitor. It’s too salty, though whether this is from the ingredients or his tears is also never stated.
The episode is a poignant reminder of how Alzheimer’s disease is a worldwide human problem. Not American. Not western. Not restricted by race, or ethnicity, or continent. It effects us all as a species, as families, and as individuals. No matter what our jobs or backgrounds are.
For those of us on this side of the desk, it’s a reminder that Yes, we have all kinds of new toys, but from a practical viewpoint it’s hard to say that we’ve made any major advances. I’m sure my drug reps will disagree with me, and I’m not saying any of the treatments of the last 28 years are worthless, but even now we’re still far from a cure, or even something that stops progression.
That’s not from lack of trying, either.
For all the jokes about his job, Mr. Oki is no different from any other children trying to hold onto their parents as the disease slowly takes them away.
I hope we have real answers, soon.
Dr. Block has a solo neurology practice in Scottsdale, Arizona.
UVA Defends Medical School Dean, Hospital CEO After Docs Call for Their Removal
The University of Virginia (UVA) is defending the CEO of its health system and its medical school dean in the wake of a very public call for their removal.
At least 128 members of the University of Virginia faculty who are employed by both the medical school and the UVA Physicians Group wrote to the UVA Board of Visitors and its peer-elected faculty leaders, expressing no confidence in K. Craig Kent, MD, CEO of UVA Health and executive vice president for health affairs, and Melina Kibbe, MD, dean of the medical school and chief health affairs officer.
Dr. Kibbe, a vascular surgeon and researcher, is also the editor in chief of JAMA Surgery.
“We call for the immediate removal of Craig Kent and Melina Kibbe,” wrote the physicians.
The letter alleged that patient safety was compromised because doctors, nurses, and other staff were pressured to abstain from reporting safety concerns and that physicians had been hired “despite concerns regarding integrity and quality.” Those who raised safety concerns faced “explicit and implicit threats and retaliation,” including delays and denials of promotion and tenure, said the letter.
The September 5 letter did not include signatures. The authors said that names were being protected, but that they would share the names with a limited audience.
UVA President Jim Ryan took issue with the notion that the signees were anonymous. He said in his own letter to medical school faculty that some of the accusations were about matters that had already been addressed or that were being worked on. As far as allegations that he was not previously aware of, “we will do our best to investigate,” he said.
The faculty who signed the letter “have besmirched the reputations of not just Melina and Craig,” wrote Mr. Ryan. “They have unfairly — and I trust unwittingly — cast a shadow over the great work of the entire health system and medical school.”
The authors claimed that reports about bullying and harassment of trainees had been “suppressed, minimized, and subsequently altered.”
And they said that spending on leadership was prioritized over addressing clinical and technical staff shortages. Whistleblowers who reported fraud were not protected, and clinicians were pressured to modify patient records to “obfuscate adverse outcomes and boost productivity metrics,” they wrote.
The 128 members of the UVA Physicians Group who signed the letter represent about 10% of the 1400 medical school faculty members.
It is not the first time that Dr. Kent has been given a vote of no confidence. In 2017, when he was the dean of the College of Medicine at the Ohio State University, Dr. Kent was accused in a “no confidence” letter from 25 physicians and faculty of helping to undermine the school’s mission and taking actions that led to resignations and early retirements of many staff, the Columbus Dispatch reported.
William G. Crutchfield Jr., a member of the UVA Health System Board, defended Dr. Kent and Dr. Kibbe in a lengthy statement shared with this news organization. He said that UVA Health’s four hospitals had received “A” ratings for safety, and that the system has a 5.1% turnover rate compared with a national average of 8.3%.
Dr. Kent and Dr. Kibbe have recruited faculty from top academic medical centers, Mr. Crutchfield wrote.
“If our work environment were so toxic, these people would not have joined our faculty,” he wrote.
Mr. Crutchfield credited Dr. Kent and Dr. Kibbe with crafting a new 10-year strategic plan and for hiring a chief strategy officer to lead the plan — a move that replaced “expensive outside consultants.”
Mr. Ryan said in his letter that his inbox “is overflowing with testimonials from some of the 1200-plus faculty who did not sign the letter, who attest that the health system today — under Melina and Craig’s leadership — is in the best shape it has ever been in, and that they have addressed changes that have needed to be made for more than two decades.”
A request to see some of these positive testimonials was not answered by press time.
Mr. Crutchfield, like Mr. Ryan, said that the letter writers were doing more harm than good.
“If a small cabal of people hiding behind anonymity can force outstanding leaders out of UVA, it will make it extremely difficult to recruit outstanding new physicians, nurses, technicians, and administrators,” he wrote.
A version of this article first appeared on Medscape.com.
The University of Virginia (UVA) is defending the CEO of its health system and its medical school dean in the wake of a very public call for their removal.
At least 128 members of the University of Virginia faculty who are employed by both the medical school and the UVA Physicians Group wrote to the UVA Board of Visitors and its peer-elected faculty leaders, expressing no confidence in K. Craig Kent, MD, CEO of UVA Health and executive vice president for health affairs, and Melina Kibbe, MD, dean of the medical school and chief health affairs officer.
Dr. Kibbe, a vascular surgeon and researcher, is also the editor in chief of JAMA Surgery.
“We call for the immediate removal of Craig Kent and Melina Kibbe,” wrote the physicians.
The letter alleged that patient safety was compromised because doctors, nurses, and other staff were pressured to abstain from reporting safety concerns and that physicians had been hired “despite concerns regarding integrity and quality.” Those who raised safety concerns faced “explicit and implicit threats and retaliation,” including delays and denials of promotion and tenure, said the letter.
The September 5 letter did not include signatures. The authors said that names were being protected, but that they would share the names with a limited audience.
UVA President Jim Ryan took issue with the notion that the signees were anonymous. He said in his own letter to medical school faculty that some of the accusations were about matters that had already been addressed or that were being worked on. As far as allegations that he was not previously aware of, “we will do our best to investigate,” he said.
The faculty who signed the letter “have besmirched the reputations of not just Melina and Craig,” wrote Mr. Ryan. “They have unfairly — and I trust unwittingly — cast a shadow over the great work of the entire health system and medical school.”
The authors claimed that reports about bullying and harassment of trainees had been “suppressed, minimized, and subsequently altered.”
And they said that spending on leadership was prioritized over addressing clinical and technical staff shortages. Whistleblowers who reported fraud were not protected, and clinicians were pressured to modify patient records to “obfuscate adverse outcomes and boost productivity metrics,” they wrote.
The 128 members of the UVA Physicians Group who signed the letter represent about 10% of the 1400 medical school faculty members.
It is not the first time that Dr. Kent has been given a vote of no confidence. In 2017, when he was the dean of the College of Medicine at the Ohio State University, Dr. Kent was accused in a “no confidence” letter from 25 physicians and faculty of helping to undermine the school’s mission and taking actions that led to resignations and early retirements of many staff, the Columbus Dispatch reported.
William G. Crutchfield Jr., a member of the UVA Health System Board, defended Dr. Kent and Dr. Kibbe in a lengthy statement shared with this news organization. He said that UVA Health’s four hospitals had received “A” ratings for safety, and that the system has a 5.1% turnover rate compared with a national average of 8.3%.
Dr. Kent and Dr. Kibbe have recruited faculty from top academic medical centers, Mr. Crutchfield wrote.
“If our work environment were so toxic, these people would not have joined our faculty,” he wrote.
Mr. Crutchfield credited Dr. Kent and Dr. Kibbe with crafting a new 10-year strategic plan and for hiring a chief strategy officer to lead the plan — a move that replaced “expensive outside consultants.”
Mr. Ryan said in his letter that his inbox “is overflowing with testimonials from some of the 1200-plus faculty who did not sign the letter, who attest that the health system today — under Melina and Craig’s leadership — is in the best shape it has ever been in, and that they have addressed changes that have needed to be made for more than two decades.”
A request to see some of these positive testimonials was not answered by press time.
Mr. Crutchfield, like Mr. Ryan, said that the letter writers were doing more harm than good.
“If a small cabal of people hiding behind anonymity can force outstanding leaders out of UVA, it will make it extremely difficult to recruit outstanding new physicians, nurses, technicians, and administrators,” he wrote.
A version of this article first appeared on Medscape.com.
The University of Virginia (UVA) is defending the CEO of its health system and its medical school dean in the wake of a very public call for their removal.
At least 128 members of the University of Virginia faculty who are employed by both the medical school and the UVA Physicians Group wrote to the UVA Board of Visitors and its peer-elected faculty leaders, expressing no confidence in K. Craig Kent, MD, CEO of UVA Health and executive vice president for health affairs, and Melina Kibbe, MD, dean of the medical school and chief health affairs officer.
Dr. Kibbe, a vascular surgeon and researcher, is also the editor in chief of JAMA Surgery.
“We call for the immediate removal of Craig Kent and Melina Kibbe,” wrote the physicians.
The letter alleged that patient safety was compromised because doctors, nurses, and other staff were pressured to abstain from reporting safety concerns and that physicians had been hired “despite concerns regarding integrity and quality.” Those who raised safety concerns faced “explicit and implicit threats and retaliation,” including delays and denials of promotion and tenure, said the letter.
The September 5 letter did not include signatures. The authors said that names were being protected, but that they would share the names with a limited audience.
UVA President Jim Ryan took issue with the notion that the signees were anonymous. He said in his own letter to medical school faculty that some of the accusations were about matters that had already been addressed or that were being worked on. As far as allegations that he was not previously aware of, “we will do our best to investigate,” he said.
The faculty who signed the letter “have besmirched the reputations of not just Melina and Craig,” wrote Mr. Ryan. “They have unfairly — and I trust unwittingly — cast a shadow over the great work of the entire health system and medical school.”
The authors claimed that reports about bullying and harassment of trainees had been “suppressed, minimized, and subsequently altered.”
And they said that spending on leadership was prioritized over addressing clinical and technical staff shortages. Whistleblowers who reported fraud were not protected, and clinicians were pressured to modify patient records to “obfuscate adverse outcomes and boost productivity metrics,” they wrote.
The 128 members of the UVA Physicians Group who signed the letter represent about 10% of the 1400 medical school faculty members.
It is not the first time that Dr. Kent has been given a vote of no confidence. In 2017, when he was the dean of the College of Medicine at the Ohio State University, Dr. Kent was accused in a “no confidence” letter from 25 physicians and faculty of helping to undermine the school’s mission and taking actions that led to resignations and early retirements of many staff, the Columbus Dispatch reported.
William G. Crutchfield Jr., a member of the UVA Health System Board, defended Dr. Kent and Dr. Kibbe in a lengthy statement shared with this news organization. He said that UVA Health’s four hospitals had received “A” ratings for safety, and that the system has a 5.1% turnover rate compared with a national average of 8.3%.
Dr. Kent and Dr. Kibbe have recruited faculty from top academic medical centers, Mr. Crutchfield wrote.
“If our work environment were so toxic, these people would not have joined our faculty,” he wrote.
Mr. Crutchfield credited Dr. Kent and Dr. Kibbe with crafting a new 10-year strategic plan and for hiring a chief strategy officer to lead the plan — a move that replaced “expensive outside consultants.”
Mr. Ryan said in his letter that his inbox “is overflowing with testimonials from some of the 1200-plus faculty who did not sign the letter, who attest that the health system today — under Melina and Craig’s leadership — is in the best shape it has ever been in, and that they have addressed changes that have needed to be made for more than two decades.”
A request to see some of these positive testimonials was not answered by press time.
Mr. Crutchfield, like Mr. Ryan, said that the letter writers were doing more harm than good.
“If a small cabal of people hiding behind anonymity can force outstanding leaders out of UVA, it will make it extremely difficult to recruit outstanding new physicians, nurses, technicians, and administrators,” he wrote.
A version of this article first appeared on Medscape.com.






