User login
GI Doc Empowers Female Patients ‘To Be Themselves’
Pooja Singhal, MD, AGAF, will never forget the time a female patient came in for gastroesophageal reflux disease and dysphagia treatment, revealing that she had already gone through multiple gastroenterologists to help diagnose and treat her ailments.
“We spent a whole visit talking about it,” said Dr. Singhal, a gastroenterologist, hepatologist, and obesity medicine specialist at Oklahoma Gastro Health and Wellness in Oklahoma City. During the exam, she discovered that her middle-aged patient was wearing an adult diaper for diarrhea and leakage.
Previous GI doctors told the patient they couldn’t help her and that she had to live with these symptoms. “I was just so shocked. I told her: This is not normal. Let’s talk more about it. Let’s figure out how we can manage it,” said Dr. Singhal, who has spent her career advocating for more education about GI conditions.
There are real barriers to patients opening up and sharing their symptoms, especially if they’re female. while ensuring that the correct knowledge gets across to the public, said Dr. Singhal.
An alumna of the American Gastroenterological Association’s (AGA) Future Leaders Program, Dr. Singhal has served as the private practice course director for AGA’s Midwest Women in GI Workshop. She is a also a four-time recipient of the SCOPY award for her work in raising community awareness of colorectal cancer prevention in Oklahoma. In an interview, she discussed the critical role women GI doctors play in assisting the unique needs of female patients, and why it takes a village of doctors to treat the complexities of GI disorders.
Why did you choose GI, and more specifically, what brought about your interest in women’s GI issues?
GI is simply the best field. While I was doing my rotation in GI as a resident, I was enthralled and humbled that the field of gastroenterology offered an opportunity to prevent cancer. Colon cancer is the second leading cause of cancer related deaths, and when I realized that we could do these micro-interventions during a procedure to remove polyps that could potentially turn into cancer — or give us an opportunity to remove carcinoma in situ — that’s what really inspired me and piqued my interest in GI. As I continued to learn and explore GI more, I appreciated the opportunity the field gave us in terms of using both sides of our brains equally, the right side and the left side.
I love the diagnostic part of medicine. You have this privilege to be able to diagnose so many different diseases and perform procedures using technical skills, exploring everything from the esophagus, liver, pancreas, small bowel, and colon.
But what I really appreciate about gastroenterology is how it’s piqued my interest in women’s digestive health. How it became very close to my heart is really from my patients. I’ve learned a lot from my patients throughout the years. When I was much younger, I don’t know if I really appreciated the vulnerability it takes as a woman to go to a physician and talk about hemorrhoids and diarrhea.
One of the comments I often receive is: ‘Oh, thank God you’re a female GI. I can be myself. I can share something personal and you would understand.’
Your practice places a specific emphasis on health and wellness. Can you provide some examples of how you incorporate wellness into treatment?
I feel like wellness is very commonplace now. To me, the definition of wellness is about practicing healthy habits to attain your maximum potential, both physically and mentally — to feel the best you can. My practice specifically tries to achieve that goal by placing a strong emphasis on education and communication. We provide journals where patients can keep track of their symptoms. We encourage a lot of discussion during visits, where we talk about GI diseases and how to prevent them, or to prevent them from happening again. If you’re going to do a hemorrhoid treatment that offers hemorrhoid banding, we talk about it in detail with the patient; we don’t just do the procedure.
We have a dietitian on staff for conditions like inflammatory bowel disease, Crohn’s and ulcerative colitis, celiac disease, IBS. Some of our older patients have pelvic organ prolapse and fecal incontinence. We have a pelvic floor therapist and a urogynecologist, and we work very closely with ob-gyn teams. My practice also takes pride in communicating with primary care physicians. We’ve had patients who have had memory loss or dementia or are grieving the loss of a loved one. And we prioritize communicating and treating patients as a whole and not focusing on just their GI symptoms.
As an advocate for community education on GI disorders, where is education lacking in this field?
I think education is lacking because there is an information delivery gap. I feel the public consumes information in the form of short social media reels. The attention span is so short and any scientific information, especially around diseases, can be scary and overwhelming. Whereas I think a lot of the medical community still interacts and exchanges information in terms of journals and publications. So, we are not really trained necessarily to talk about diseases in very simple terms.
We need more advocacy efforts on Capitol Hill. AGA has been good about doing advocacy work. I had an opportunity to go to Capitol Hill a couple of times and really advocate for policy around obesity medicine coverage and procedure coverage. I was fortunate to learn so much about healthcare policy, but it also made me appreciate that there are a lot of gaps in terms of understanding common medical diseases.
You’re trained in the Orbera Intragastric balloon system for weight reduction. How does this procedure differentiate from other bariatric procedures?
Intragastric balloon is Food and Drug Administration approved for weight loss. It’s a temporary medical device, so it’s reversible. No. 2, it’s a nonsurgical intervention, so it’s usually done in an outpatient setting. We basically place a deflated gastric balloon endoscopically, similar to an upper endoscopy method. We take a pin endoscope, a deflated balloon, which is made of medical-grade material, and we inflate it with adequate fluid. The concept is when the balloon is inflated, it provides satiety. It reduces the amount of space in the stomach for food. It slows down how quickly the food is going to leave. So you feel full much of the time. And it also helps decrease a hormone called ghrelin, which is responsible for hunger. It can make a big difference when people are gaining weight and in that category of overweight before they progress to obese.
As I tell everybody, obesity is a chronic lifelong disease that is very complex and requires lifelong efforts. So, it’s truly a journey. What’s made this procedure a success is follow-up and the continued efforts of dietitians and counseling and incorporating physical exercise, because maintenance of that weight loss is also very important. Our goal is always sustained weight loss and not just short-term weight loss.
As the practice course director for the AGA’s Midwest Women in GI Workshop, can you tell me how this course came about? What does the workshop cover?
This workshop is a brainchild of AGA. This will be the third year of having these workshops. It’s been divided into regional workshops, so more people can attend. But it arose from the recognition that there is a need to have a support system, a forum where discussions on navigating career and life transitions with grace can happen, and more resources for success can be provided.
There is so much power in learning from shared experiences. And I think that was huge, to realize that we are not alone. We can celebrate our achievements together and acknowledge our challenges together, and then come together to brainstorm and innovate to solve problems and advocate for health equity.
You’ve been involved with community, non-profit organizations like the Homeless Alliance in Oklahoma City. How has this work enriched your life outside of medicine?
I feel like we sometimes get tunnel vision, talking to people in the same line of work. It was extremely important for me to broaden my horizons by learning from people outside of the medical community and from organizations like Homeless Alliance, which allowed me a platform to understand what my community needs. It’s an incredible organization that helps provide shelter not only for human beings, but also pets. The freezing temperatures over the last few months provided unique challenges like overflow in homeless shelters. I’ve learned so many things, such as how to ask for grants and how to allocate those funds. It has been absolutely enriching to me to learn about my community needs and see what an amazing difference people in the community are making.
Pooja Singhal, MD, AGAF, will never forget the time a female patient came in for gastroesophageal reflux disease and dysphagia treatment, revealing that she had already gone through multiple gastroenterologists to help diagnose and treat her ailments.
“We spent a whole visit talking about it,” said Dr. Singhal, a gastroenterologist, hepatologist, and obesity medicine specialist at Oklahoma Gastro Health and Wellness in Oklahoma City. During the exam, she discovered that her middle-aged patient was wearing an adult diaper for diarrhea and leakage.
Previous GI doctors told the patient they couldn’t help her and that she had to live with these symptoms. “I was just so shocked. I told her: This is not normal. Let’s talk more about it. Let’s figure out how we can manage it,” said Dr. Singhal, who has spent her career advocating for more education about GI conditions.
There are real barriers to patients opening up and sharing their symptoms, especially if they’re female. while ensuring that the correct knowledge gets across to the public, said Dr. Singhal.
An alumna of the American Gastroenterological Association’s (AGA) Future Leaders Program, Dr. Singhal has served as the private practice course director for AGA’s Midwest Women in GI Workshop. She is a also a four-time recipient of the SCOPY award for her work in raising community awareness of colorectal cancer prevention in Oklahoma. In an interview, she discussed the critical role women GI doctors play in assisting the unique needs of female patients, and why it takes a village of doctors to treat the complexities of GI disorders.
Why did you choose GI, and more specifically, what brought about your interest in women’s GI issues?
GI is simply the best field. While I was doing my rotation in GI as a resident, I was enthralled and humbled that the field of gastroenterology offered an opportunity to prevent cancer. Colon cancer is the second leading cause of cancer related deaths, and when I realized that we could do these micro-interventions during a procedure to remove polyps that could potentially turn into cancer — or give us an opportunity to remove carcinoma in situ — that’s what really inspired me and piqued my interest in GI. As I continued to learn and explore GI more, I appreciated the opportunity the field gave us in terms of using both sides of our brains equally, the right side and the left side.
I love the diagnostic part of medicine. You have this privilege to be able to diagnose so many different diseases and perform procedures using technical skills, exploring everything from the esophagus, liver, pancreas, small bowel, and colon.
But what I really appreciate about gastroenterology is how it’s piqued my interest in women’s digestive health. How it became very close to my heart is really from my patients. I’ve learned a lot from my patients throughout the years. When I was much younger, I don’t know if I really appreciated the vulnerability it takes as a woman to go to a physician and talk about hemorrhoids and diarrhea.
One of the comments I often receive is: ‘Oh, thank God you’re a female GI. I can be myself. I can share something personal and you would understand.’
Your practice places a specific emphasis on health and wellness. Can you provide some examples of how you incorporate wellness into treatment?
I feel like wellness is very commonplace now. To me, the definition of wellness is about practicing healthy habits to attain your maximum potential, both physically and mentally — to feel the best you can. My practice specifically tries to achieve that goal by placing a strong emphasis on education and communication. We provide journals where patients can keep track of their symptoms. We encourage a lot of discussion during visits, where we talk about GI diseases and how to prevent them, or to prevent them from happening again. If you’re going to do a hemorrhoid treatment that offers hemorrhoid banding, we talk about it in detail with the patient; we don’t just do the procedure.
We have a dietitian on staff for conditions like inflammatory bowel disease, Crohn’s and ulcerative colitis, celiac disease, IBS. Some of our older patients have pelvic organ prolapse and fecal incontinence. We have a pelvic floor therapist and a urogynecologist, and we work very closely with ob-gyn teams. My practice also takes pride in communicating with primary care physicians. We’ve had patients who have had memory loss or dementia or are grieving the loss of a loved one. And we prioritize communicating and treating patients as a whole and not focusing on just their GI symptoms.
As an advocate for community education on GI disorders, where is education lacking in this field?
I think education is lacking because there is an information delivery gap. I feel the public consumes information in the form of short social media reels. The attention span is so short and any scientific information, especially around diseases, can be scary and overwhelming. Whereas I think a lot of the medical community still interacts and exchanges information in terms of journals and publications. So, we are not really trained necessarily to talk about diseases in very simple terms.
We need more advocacy efforts on Capitol Hill. AGA has been good about doing advocacy work. I had an opportunity to go to Capitol Hill a couple of times and really advocate for policy around obesity medicine coverage and procedure coverage. I was fortunate to learn so much about healthcare policy, but it also made me appreciate that there are a lot of gaps in terms of understanding common medical diseases.
You’re trained in the Orbera Intragastric balloon system for weight reduction. How does this procedure differentiate from other bariatric procedures?
Intragastric balloon is Food and Drug Administration approved for weight loss. It’s a temporary medical device, so it’s reversible. No. 2, it’s a nonsurgical intervention, so it’s usually done in an outpatient setting. We basically place a deflated gastric balloon endoscopically, similar to an upper endoscopy method. We take a pin endoscope, a deflated balloon, which is made of medical-grade material, and we inflate it with adequate fluid. The concept is when the balloon is inflated, it provides satiety. It reduces the amount of space in the stomach for food. It slows down how quickly the food is going to leave. So you feel full much of the time. And it also helps decrease a hormone called ghrelin, which is responsible for hunger. It can make a big difference when people are gaining weight and in that category of overweight before they progress to obese.
As I tell everybody, obesity is a chronic lifelong disease that is very complex and requires lifelong efforts. So, it’s truly a journey. What’s made this procedure a success is follow-up and the continued efforts of dietitians and counseling and incorporating physical exercise, because maintenance of that weight loss is also very important. Our goal is always sustained weight loss and not just short-term weight loss.
As the practice course director for the AGA’s Midwest Women in GI Workshop, can you tell me how this course came about? What does the workshop cover?
This workshop is a brainchild of AGA. This will be the third year of having these workshops. It’s been divided into regional workshops, so more people can attend. But it arose from the recognition that there is a need to have a support system, a forum where discussions on navigating career and life transitions with grace can happen, and more resources for success can be provided.
There is so much power in learning from shared experiences. And I think that was huge, to realize that we are not alone. We can celebrate our achievements together and acknowledge our challenges together, and then come together to brainstorm and innovate to solve problems and advocate for health equity.
You’ve been involved with community, non-profit organizations like the Homeless Alliance in Oklahoma City. How has this work enriched your life outside of medicine?
I feel like we sometimes get tunnel vision, talking to people in the same line of work. It was extremely important for me to broaden my horizons by learning from people outside of the medical community and from organizations like Homeless Alliance, which allowed me a platform to understand what my community needs. It’s an incredible organization that helps provide shelter not only for human beings, but also pets. The freezing temperatures over the last few months provided unique challenges like overflow in homeless shelters. I’ve learned so many things, such as how to ask for grants and how to allocate those funds. It has been absolutely enriching to me to learn about my community needs and see what an amazing difference people in the community are making.
Pooja Singhal, MD, AGAF, will never forget the time a female patient came in for gastroesophageal reflux disease and dysphagia treatment, revealing that she had already gone through multiple gastroenterologists to help diagnose and treat her ailments.
“We spent a whole visit talking about it,” said Dr. Singhal, a gastroenterologist, hepatologist, and obesity medicine specialist at Oklahoma Gastro Health and Wellness in Oklahoma City. During the exam, she discovered that her middle-aged patient was wearing an adult diaper for diarrhea and leakage.
Previous GI doctors told the patient they couldn’t help her and that she had to live with these symptoms. “I was just so shocked. I told her: This is not normal. Let’s talk more about it. Let’s figure out how we can manage it,” said Dr. Singhal, who has spent her career advocating for more education about GI conditions.
There are real barriers to patients opening up and sharing their symptoms, especially if they’re female. while ensuring that the correct knowledge gets across to the public, said Dr. Singhal.
An alumna of the American Gastroenterological Association’s (AGA) Future Leaders Program, Dr. Singhal has served as the private practice course director for AGA’s Midwest Women in GI Workshop. She is a also a four-time recipient of the SCOPY award for her work in raising community awareness of colorectal cancer prevention in Oklahoma. In an interview, she discussed the critical role women GI doctors play in assisting the unique needs of female patients, and why it takes a village of doctors to treat the complexities of GI disorders.
Why did you choose GI, and more specifically, what brought about your interest in women’s GI issues?
GI is simply the best field. While I was doing my rotation in GI as a resident, I was enthralled and humbled that the field of gastroenterology offered an opportunity to prevent cancer. Colon cancer is the second leading cause of cancer related deaths, and when I realized that we could do these micro-interventions during a procedure to remove polyps that could potentially turn into cancer — or give us an opportunity to remove carcinoma in situ — that’s what really inspired me and piqued my interest in GI. As I continued to learn and explore GI more, I appreciated the opportunity the field gave us in terms of using both sides of our brains equally, the right side and the left side.
I love the diagnostic part of medicine. You have this privilege to be able to diagnose so many different diseases and perform procedures using technical skills, exploring everything from the esophagus, liver, pancreas, small bowel, and colon.
But what I really appreciate about gastroenterology is how it’s piqued my interest in women’s digestive health. How it became very close to my heart is really from my patients. I’ve learned a lot from my patients throughout the years. When I was much younger, I don’t know if I really appreciated the vulnerability it takes as a woman to go to a physician and talk about hemorrhoids and diarrhea.
One of the comments I often receive is: ‘Oh, thank God you’re a female GI. I can be myself. I can share something personal and you would understand.’
Your practice places a specific emphasis on health and wellness. Can you provide some examples of how you incorporate wellness into treatment?
I feel like wellness is very commonplace now. To me, the definition of wellness is about practicing healthy habits to attain your maximum potential, both physically and mentally — to feel the best you can. My practice specifically tries to achieve that goal by placing a strong emphasis on education and communication. We provide journals where patients can keep track of their symptoms. We encourage a lot of discussion during visits, where we talk about GI diseases and how to prevent them, or to prevent them from happening again. If you’re going to do a hemorrhoid treatment that offers hemorrhoid banding, we talk about it in detail with the patient; we don’t just do the procedure.
We have a dietitian on staff for conditions like inflammatory bowel disease, Crohn’s and ulcerative colitis, celiac disease, IBS. Some of our older patients have pelvic organ prolapse and fecal incontinence. We have a pelvic floor therapist and a urogynecologist, and we work very closely with ob-gyn teams. My practice also takes pride in communicating with primary care physicians. We’ve had patients who have had memory loss or dementia or are grieving the loss of a loved one. And we prioritize communicating and treating patients as a whole and not focusing on just their GI symptoms.
As an advocate for community education on GI disorders, where is education lacking in this field?
I think education is lacking because there is an information delivery gap. I feel the public consumes information in the form of short social media reels. The attention span is so short and any scientific information, especially around diseases, can be scary and overwhelming. Whereas I think a lot of the medical community still interacts and exchanges information in terms of journals and publications. So, we are not really trained necessarily to talk about diseases in very simple terms.
We need more advocacy efforts on Capitol Hill. AGA has been good about doing advocacy work. I had an opportunity to go to Capitol Hill a couple of times and really advocate for policy around obesity medicine coverage and procedure coverage. I was fortunate to learn so much about healthcare policy, but it also made me appreciate that there are a lot of gaps in terms of understanding common medical diseases.
You’re trained in the Orbera Intragastric balloon system for weight reduction. How does this procedure differentiate from other bariatric procedures?
Intragastric balloon is Food and Drug Administration approved for weight loss. It’s a temporary medical device, so it’s reversible. No. 2, it’s a nonsurgical intervention, so it’s usually done in an outpatient setting. We basically place a deflated gastric balloon endoscopically, similar to an upper endoscopy method. We take a pin endoscope, a deflated balloon, which is made of medical-grade material, and we inflate it with adequate fluid. The concept is when the balloon is inflated, it provides satiety. It reduces the amount of space in the stomach for food. It slows down how quickly the food is going to leave. So you feel full much of the time. And it also helps decrease a hormone called ghrelin, which is responsible for hunger. It can make a big difference when people are gaining weight and in that category of overweight before they progress to obese.
As I tell everybody, obesity is a chronic lifelong disease that is very complex and requires lifelong efforts. So, it’s truly a journey. What’s made this procedure a success is follow-up and the continued efforts of dietitians and counseling and incorporating physical exercise, because maintenance of that weight loss is also very important. Our goal is always sustained weight loss and not just short-term weight loss.
As the practice course director for the AGA’s Midwest Women in GI Workshop, can you tell me how this course came about? What does the workshop cover?
This workshop is a brainchild of AGA. This will be the third year of having these workshops. It’s been divided into regional workshops, so more people can attend. But it arose from the recognition that there is a need to have a support system, a forum where discussions on navigating career and life transitions with grace can happen, and more resources for success can be provided.
There is so much power in learning from shared experiences. And I think that was huge, to realize that we are not alone. We can celebrate our achievements together and acknowledge our challenges together, and then come together to brainstorm and innovate to solve problems and advocate for health equity.
You’ve been involved with community, non-profit organizations like the Homeless Alliance in Oklahoma City. How has this work enriched your life outside of medicine?
I feel like we sometimes get tunnel vision, talking to people in the same line of work. It was extremely important for me to broaden my horizons by learning from people outside of the medical community and from organizations like Homeless Alliance, which allowed me a platform to understand what my community needs. It’s an incredible organization that helps provide shelter not only for human beings, but also pets. The freezing temperatures over the last few months provided unique challenges like overflow in homeless shelters. I’ve learned so many things, such as how to ask for grants and how to allocate those funds. It has been absolutely enriching to me to learn about my community needs and see what an amazing difference people in the community are making.

Endoscopic Sleeve Gastroplasty is an Effective Treatment for Obesity in a Veteran With Metabolic and Psychiatric Comorbidities
Endoscopic Sleeve Gastroplasty is an Effective Treatment for Obesity in a Veteran With Metabolic and Psychiatric Comorbidities
Obesity is a growing worldwide epidemic with significant implications for individual health and public health care costs. It is also associated with several medical conditions, including diabetes, cardiovascular disease, cancer, and mental health disorders.1 Comprehensive lifestyle intervention is a first-line therapy for obesity consisting of dietary and exercise interventions. Despite initial success, long-term results and durability of weight loss with lifestyle modifications are limited. 2 Bariatric surgery, including sleeve gastrectomy and gastric bypass surgery, is a more invasive approach that is highly effective in weight loss. However, these operations are not reversible, and patients may not be eligible for or may not desire surgery. Overall, bariatric surgery is widely underutilized, with < 1% of eligible patients ultimately undergoing surgery.3,4
Endoscopic bariatric therapies are increasingly popular procedures that address the need for additional treatments for obesity among individuals who have not had success with lifestyle changes and are not surgical candidates. The most common procedure is the endoscopic sleeve gastroplasty (ESG), which applies full-thickness sutures in the stomach to reduce gastric volume, delay gastric emptying, and limit food intake while keeping the fundus intact compared with sleeve gastrectomy. This procedure is typically considered in patients with body mass index (BMI) ≥ 30, who do not qualify for or do not want traditional bariatric surgery. The literature supports robust outcomes after ESG, with studies demonstrating significant and sustained total body weight loss of up to 14% to 16% at 5 years and significant improvement in ≥ 1 metabolic comorbidities in 80% of patients.5,6 ESG adverse events (AEs) include abdominal pain, nausea, and vomiting that are typically self-limited to 1 week. Rarer but more serious AEs include bleeding, perforation, or infection, and occur in 2% of cases based on large trial data.5,7
Although the weight loss benefits of ESG are well established, to date, there are limited data on the effects of endoscopic bariatric therapies like ESG on mental health conditions. Here, we describe a case of a veteran with a history of mental health disorders that prevented him from completing bariatric surgery. The patient underwent ESG and had a successful clinical course.
CASE PRESENTATION
A 59-year-old male veteran with a medical history of class III obesity (42.4 BMI), obstructive sleep apnea, hypothyroidism, hypertension, type 2 diabetes mellitus, and a large ventral hernia was referred to the MOVE! (Management of Overweight/ Obese Veterans Everywhere!) multidisciplinary high-intensity weight loss program at the US Department of Veterans Affairs (VA) West Los Angeles VA Medical Center (WLAVAMC). His psychiatric history included generalized anxiety disorder, posttraumatic stress disorder (PTSD), and panic disorder, managed by the Psychiatry Service and treated with sertraline 25 mg daily, lorazepam 0.5 mg twice daily, and hydroxyzine 20 mg nightly. He had previously implemented lifestyle changes and attended MOVE! classes and nutrition coaching for 1 year but was unsuccessful in losing weight. He had also tried liraglutide 3 mg daily for weight loss but was unable to tolerate it and reported worsening medication-related anxiety.
The patient declined further weight loss pharmacotherapy and was referred to bariatric surgery. He was scheduled for a surgical sleeve gastrectomy. However, on the day he arrived at the hospital for surgery, he developed severe anxiety and had a panic attack, and it was canceled. Due to his mental health issues, he was no longer comfortable proceeding with surgery and was left without other options for obesity treatment. The veteran was extremely disappointed because the ventral hernia caused significant quality of life impairment, limited his ability to exercise, and caused him embarrassment in public settings. The hernia could not be surgically repaired until there was significant weight loss.
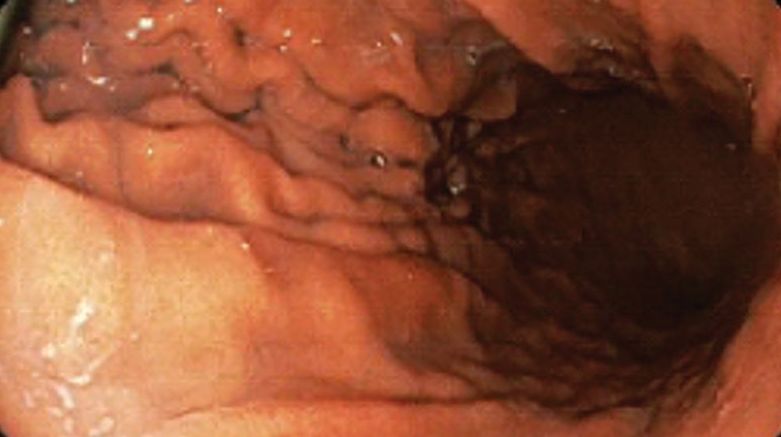
A bariatric endoscopy program within the Division of Gastroenterology was developed and implemented at the WLAVAMC in February 2023 in conjunction with MOVE! The patient was referred for consideration of an endoscopic weight loss procedure. He was determined to be a suitable candidate for ESG based on his BMI being > 40 and personal preference not to proceed with surgery to lose enough weight to qualify for hernia repair. The veteran underwent an endoscopy, which showed normal anatomy and gastric mucosa. ESG was performed in standard fashion (Figure).8 Three vertical lines were made using argon plasma coagulation from the incisura to 2 cm below the gastroesophageal junction along the anterior, posterior, and greater curvature of the stomach to mark the area for endoscopic suture placement. Starting at the incisura, 7 full-thickness sutures were placed to create a volume reduction plication, with preservation of the fundus. The patient did well postprocedure with no immediate or delayed AEs and was discharged home the same day.
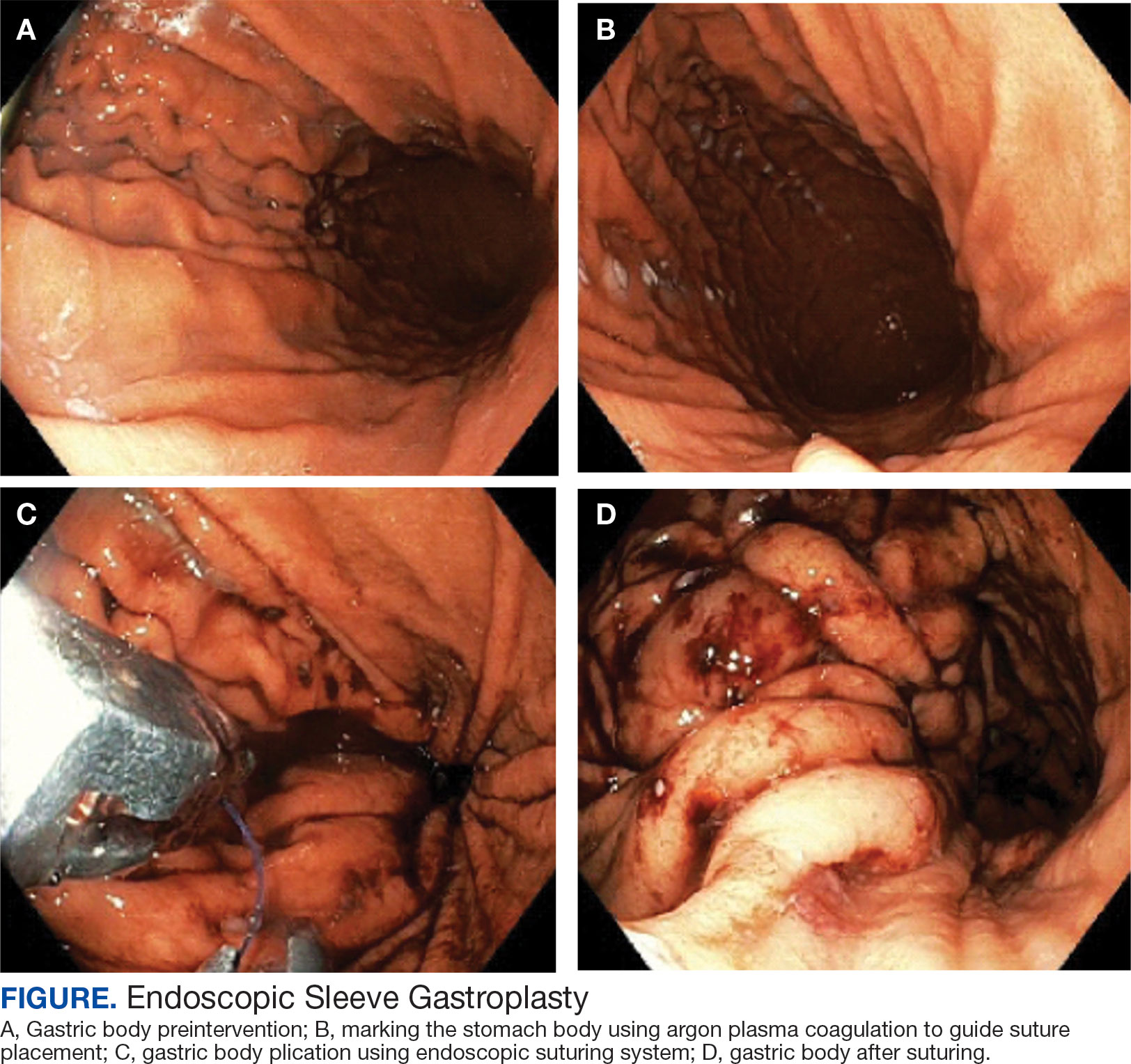
Follow-up
The veteran followed a gradual dietary advancement from a clear liquid diet to pureed and soft texture food. The patient’s weight dropped from 359 lbs preprocedure to 304 lbs 6 months postprocedure, a total body weight loss (TWBL) of 15.3%. At 12 months the veteran weighed 299 lbs (16.7% TBWL). He also had notable improvements in metabolic parameters. His systolic blood pressure decreased from ≥ 140 mm Hg to 120 to 130 mm Hg and hemoglobin A1c dropped from 7.0% to 6.3%. Remarkably, his psychiatrist noted significant improvement in his overall mental health. The veteran reported complete cessation of panic attacks since the ESG, improvements in PTSD and anxiety, and was able to discontinue lorazepam and decrease his dose of sertraline to 12.5 mg daily. He reported feeling more energetic and goal-oriented with increased clarity of thought. Perhaps the most significant outcome was that after the 55-lb weight loss at 6 months, the patient was eligible to undergo ventral hernia surgical repair, which had previously contributed to shame and social isolation. This, in turn, improved his quality of life, allowed him to start walking again, up to 8 miles daily, and to feel comfortable again going out in public settings.
DISCUSSION
Bariatric surgeries are an effective method of achieving weight loss and improving obesity-related comorbidities. However, only a small percentage of individuals with obesity are candidates for bariatric surgery. Given the dramatic increase in the prevalence of obesity, other options are needed. Specifically, within the VA, an estimated 80% of veterans are overweight or obese, but only about 500 bariatric surgeries are performed annually.9 With the need for additional weight loss therapies, VA programs are starting to offer endoscopic bariatric procedures as an alternative option. This may be a desirable choice for patients with obesity (BMI > 30), with or without associated metabolic comorbidities, who need more aggressive intervention beyond dietary and lifestyle changes and are either not interested in or not eligible for bariatric surgery or weight loss medications.
Although there is evidence that metabolic comorbidities are associated with obesity, there has been less research on obesity and mental health comorbidities such as depression and anxiety. These psychiatric conditions may even be more common among patients seeking weight loss procedures and more prominent in certain groups such as veterans, which may ultimately exclude these patients from bariatric surgery.10 Prior studies suggest that bariatric surgery can reduce the severity of depression and, to a lesser extent, anxiety symptoms at 2 years following the initial surgery; however, there is limited literature describing the impact of weight loss procedure on panic disorders.11-14 We suspect that a weight loss procedure such as ESG may have indirectly improved the veteran’s mood disorder due to the weight loss it induced, increasing the ability to exercise, quality of sleep, and participation in public settings.
This case highlights a veteran who did not tolerate weight loss medication and had severe anxiety and PTSD that prevented him from going through with bariatric surgery. He then underwent an endoscopic weight loss procedure. The ESG helped him successfully achieve significant weight loss, increase his physical activity, reduce his anxiety and panic disorder, and overall, significantly improve his quality of life. More than 1 year after the procedure, the patient has sustained improvements in his psychiatric and emotional health along with durable weight loss, maintaining > 15% of his total weight lost. Additional studies are needed to further understand the prevalence and long-term outcomes of mental health comorbidities, as well as weight loss outcomes in this group of patients who undergo endoscopic bariatric procedures.
CONCLUSIONS
We describe a case of a veteran with severe obesity and significant psychiatric comorbidities that prevented him from undergoing bariatric surgery, who underwent an ESG. This procedure led to significant weight loss, improvement of metabolic parameters, reduction in anxiety and PTSD, and enhancement of his quality of life. This case emphasizes the unique advantages of ESG and supports the expansion of endoscopic bariatric programs in the VA.
- Ritchie SA, Connell JM. The link between abdominal obesity, metabolic syndrome and cardiovascular disease. Nutr Metab Cardiovasc Dis. 2007;17(4):319-326. doi:10.1016/j.numecd.2006.07.005
- Bray GA, Kim KK, Wilding JPH; World Obesity Federation. Obesity: a chronic relapsing progressive disease process. A position statement of the World Obesity Federation. Obes Rev. 2017;18(7):715-723. doi:10.1111/obr.12551
- Imbus JR, Voils CI, Funk LM. Bariatric surgery barriers: a review using andersen’s model of health services use. Surg Obes Relat Dis. 2018;14(3):404-412. doi:10.1016/j.soard.2017.11.012
- Dawes AJ, Maggard-Gibbons M, Maher AR, et al. Mental health conditions among patients seeking and undergoing bariatric surgery: a meta-analysis. JAMA. 2016;315(2):150- 163. doi:10.1001/jama.2015.18118
- Abu Dayyeh BK, Bazerbachi F, Vargas EJ, et al.. Endoscopic sleeve gastroplasty for treatment of class 1 and 2 obesity (MERIT): a prospective, multicentre, randomised trial. Lancet. 2022;400(10350):441-451. doi:10.1016/S0140-6736(22)01280-6
- Matteo MV, Bove V, Ciasca G, et al. Success predictors of endoscopic sleeve gastroplasty. Obes Surg. 2024;34(5):1496-1504. doi:10.1007/s11695-024-07109-4
- Maselli DB, Hoff AC, Kucera A, et al. Endoscopic sleeve gastroplasty in class III obesity: efficacy, safety, and durability outcomes in 404 consecutive patients. World J Gastrointest Endosc. 2023;15(6):469-479. doi:10.4253/wjge.v15.i6.469
- Kumar N, Abu Dayyeh BK, Lopez-Nava Breviere G, et al. Endoscopic sutured gastroplasty: procedure evolution from first-in-man cases through current technique. Surg Endosc. 2018;32(4):2159-2164. doi:10.1007/s00464-017-5869-2
- Maggard-Gibbons M, Shekelle PG, Girgis MD, et al. Endoscopic Bariatric Interventions versus lifestyle interventions or surgery for weight loss in patients with obesity: a systematic review and meta-analysis. Department of Veterans Affairs (US); 2022. https://www.ncbi.nlm.nih.gov/books/NBK587943/
- Maggard Gibbons MA, Maher AM, Dawes AJ, et al. Psychological clearance for bariatric surgery: a systematic review. VA-ESP project #05-2262014.
- van Hout GC, Verschure SK, van Heck GL. Psychosocial predictors of success following bariatric surgery. Obes Surg. 2005;15(4):552-560. doi:10.1381/0960892053723484
- Hudson JI, Hiripi E, Pope HG Jr, Kessler RC. The prevalence and correlates of eating disorders in the national comorbidity survey replication. Biol Psychiatry. 2007;61(3):348-358. doi:10.1016/j.biopsych.2006.03.040
- Aylward L, Lilly C, Konsor M, et al. How soon do depression and anxiety symptoms improve after bariatric surgery?. Healthcare (Basel). 2023;11(6):862. doi:10.3390/healthcare11060862
- Law S, Dong S, Zhou F, Zheng D, Wang C, Dong Z. Bariatric surgery and mental health outcomes: an umbrella review. Front Endocrinol (Lausanne). 2023;14:1283621. doi:10.3389/fendo.2023.1283621
Obesity is a growing worldwide epidemic with significant implications for individual health and public health care costs. It is also associated with several medical conditions, including diabetes, cardiovascular disease, cancer, and mental health disorders.1 Comprehensive lifestyle intervention is a first-line therapy for obesity consisting of dietary and exercise interventions. Despite initial success, long-term results and durability of weight loss with lifestyle modifications are limited. 2 Bariatric surgery, including sleeve gastrectomy and gastric bypass surgery, is a more invasive approach that is highly effective in weight loss. However, these operations are not reversible, and patients may not be eligible for or may not desire surgery. Overall, bariatric surgery is widely underutilized, with < 1% of eligible patients ultimately undergoing surgery.3,4
Endoscopic bariatric therapies are increasingly popular procedures that address the need for additional treatments for obesity among individuals who have not had success with lifestyle changes and are not surgical candidates. The most common procedure is the endoscopic sleeve gastroplasty (ESG), which applies full-thickness sutures in the stomach to reduce gastric volume, delay gastric emptying, and limit food intake while keeping the fundus intact compared with sleeve gastrectomy. This procedure is typically considered in patients with body mass index (BMI) ≥ 30, who do not qualify for or do not want traditional bariatric surgery. The literature supports robust outcomes after ESG, with studies demonstrating significant and sustained total body weight loss of up to 14% to 16% at 5 years and significant improvement in ≥ 1 metabolic comorbidities in 80% of patients.5,6 ESG adverse events (AEs) include abdominal pain, nausea, and vomiting that are typically self-limited to 1 week. Rarer but more serious AEs include bleeding, perforation, or infection, and occur in 2% of cases based on large trial data.5,7
Although the weight loss benefits of ESG are well established, to date, there are limited data on the effects of endoscopic bariatric therapies like ESG on mental health conditions. Here, we describe a case of a veteran with a history of mental health disorders that prevented him from completing bariatric surgery. The patient underwent ESG and had a successful clinical course.
CASE PRESENTATION
A 59-year-old male veteran with a medical history of class III obesity (42.4 BMI), obstructive sleep apnea, hypothyroidism, hypertension, type 2 diabetes mellitus, and a large ventral hernia was referred to the MOVE! (Management of Overweight/ Obese Veterans Everywhere!) multidisciplinary high-intensity weight loss program at the US Department of Veterans Affairs (VA) West Los Angeles VA Medical Center (WLAVAMC). His psychiatric history included generalized anxiety disorder, posttraumatic stress disorder (PTSD), and panic disorder, managed by the Psychiatry Service and treated with sertraline 25 mg daily, lorazepam 0.5 mg twice daily, and hydroxyzine 20 mg nightly. He had previously implemented lifestyle changes and attended MOVE! classes and nutrition coaching for 1 year but was unsuccessful in losing weight. He had also tried liraglutide 3 mg daily for weight loss but was unable to tolerate it and reported worsening medication-related anxiety.
The patient declined further weight loss pharmacotherapy and was referred to bariatric surgery. He was scheduled for a surgical sleeve gastrectomy. However, on the day he arrived at the hospital for surgery, he developed severe anxiety and had a panic attack, and it was canceled. Due to his mental health issues, he was no longer comfortable proceeding with surgery and was left without other options for obesity treatment. The veteran was extremely disappointed because the ventral hernia caused significant quality of life impairment, limited his ability to exercise, and caused him embarrassment in public settings. The hernia could not be surgically repaired until there was significant weight loss.
A bariatric endoscopy program within the Division of Gastroenterology was developed and implemented at the WLAVAMC in February 2023 in conjunction with MOVE! The patient was referred for consideration of an endoscopic weight loss procedure. He was determined to be a suitable candidate for ESG based on his BMI being > 40 and personal preference not to proceed with surgery to lose enough weight to qualify for hernia repair. The veteran underwent an endoscopy, which showed normal anatomy and gastric mucosa. ESG was performed in standard fashion (Figure).8 Three vertical lines were made using argon plasma coagulation from the incisura to 2 cm below the gastroesophageal junction along the anterior, posterior, and greater curvature of the stomach to mark the area for endoscopic suture placement. Starting at the incisura, 7 full-thickness sutures were placed to create a volume reduction plication, with preservation of the fundus. The patient did well postprocedure with no immediate or delayed AEs and was discharged home the same day.

Follow-up
The veteran followed a gradual dietary advancement from a clear liquid diet to pureed and soft texture food. The patient’s weight dropped from 359 lbs preprocedure to 304 lbs 6 months postprocedure, a total body weight loss (TWBL) of 15.3%. At 12 months the veteran weighed 299 lbs (16.7% TBWL). He also had notable improvements in metabolic parameters. His systolic blood pressure decreased from ≥ 140 mm Hg to 120 to 130 mm Hg and hemoglobin A1c dropped from 7.0% to 6.3%. Remarkably, his psychiatrist noted significant improvement in his overall mental health. The veteran reported complete cessation of panic attacks since the ESG, improvements in PTSD and anxiety, and was able to discontinue lorazepam and decrease his dose of sertraline to 12.5 mg daily. He reported feeling more energetic and goal-oriented with increased clarity of thought. Perhaps the most significant outcome was that after the 55-lb weight loss at 6 months, the patient was eligible to undergo ventral hernia surgical repair, which had previously contributed to shame and social isolation. This, in turn, improved his quality of life, allowed him to start walking again, up to 8 miles daily, and to feel comfortable again going out in public settings.
DISCUSSION
Bariatric surgeries are an effective method of achieving weight loss and improving obesity-related comorbidities. However, only a small percentage of individuals with obesity are candidates for bariatric surgery. Given the dramatic increase in the prevalence of obesity, other options are needed. Specifically, within the VA, an estimated 80% of veterans are overweight or obese, but only about 500 bariatric surgeries are performed annually.9 With the need for additional weight loss therapies, VA programs are starting to offer endoscopic bariatric procedures as an alternative option. This may be a desirable choice for patients with obesity (BMI > 30), with or without associated metabolic comorbidities, who need more aggressive intervention beyond dietary and lifestyle changes and are either not interested in or not eligible for bariatric surgery or weight loss medications.
Although there is evidence that metabolic comorbidities are associated with obesity, there has been less research on obesity and mental health comorbidities such as depression and anxiety. These psychiatric conditions may even be more common among patients seeking weight loss procedures and more prominent in certain groups such as veterans, which may ultimately exclude these patients from bariatric surgery.10 Prior studies suggest that bariatric surgery can reduce the severity of depression and, to a lesser extent, anxiety symptoms at 2 years following the initial surgery; however, there is limited literature describing the impact of weight loss procedure on panic disorders.11-14 We suspect that a weight loss procedure such as ESG may have indirectly improved the veteran’s mood disorder due to the weight loss it induced, increasing the ability to exercise, quality of sleep, and participation in public settings.
This case highlights a veteran who did not tolerate weight loss medication and had severe anxiety and PTSD that prevented him from going through with bariatric surgery. He then underwent an endoscopic weight loss procedure. The ESG helped him successfully achieve significant weight loss, increase his physical activity, reduce his anxiety and panic disorder, and overall, significantly improve his quality of life. More than 1 year after the procedure, the patient has sustained improvements in his psychiatric and emotional health along with durable weight loss, maintaining > 15% of his total weight lost. Additional studies are needed to further understand the prevalence and long-term outcomes of mental health comorbidities, as well as weight loss outcomes in this group of patients who undergo endoscopic bariatric procedures.
CONCLUSIONS
We describe a case of a veteran with severe obesity and significant psychiatric comorbidities that prevented him from undergoing bariatric surgery, who underwent an ESG. This procedure led to significant weight loss, improvement of metabolic parameters, reduction in anxiety and PTSD, and enhancement of his quality of life. This case emphasizes the unique advantages of ESG and supports the expansion of endoscopic bariatric programs in the VA.
Obesity is a growing worldwide epidemic with significant implications for individual health and public health care costs. It is also associated with several medical conditions, including diabetes, cardiovascular disease, cancer, and mental health disorders.1 Comprehensive lifestyle intervention is a first-line therapy for obesity consisting of dietary and exercise interventions. Despite initial success, long-term results and durability of weight loss with lifestyle modifications are limited. 2 Bariatric surgery, including sleeve gastrectomy and gastric bypass surgery, is a more invasive approach that is highly effective in weight loss. However, these operations are not reversible, and patients may not be eligible for or may not desire surgery. Overall, bariatric surgery is widely underutilized, with < 1% of eligible patients ultimately undergoing surgery.3,4
Endoscopic bariatric therapies are increasingly popular procedures that address the need for additional treatments for obesity among individuals who have not had success with lifestyle changes and are not surgical candidates. The most common procedure is the endoscopic sleeve gastroplasty (ESG), which applies full-thickness sutures in the stomach to reduce gastric volume, delay gastric emptying, and limit food intake while keeping the fundus intact compared with sleeve gastrectomy. This procedure is typically considered in patients with body mass index (BMI) ≥ 30, who do not qualify for or do not want traditional bariatric surgery. The literature supports robust outcomes after ESG, with studies demonstrating significant and sustained total body weight loss of up to 14% to 16% at 5 years and significant improvement in ≥ 1 metabolic comorbidities in 80% of patients.5,6 ESG adverse events (AEs) include abdominal pain, nausea, and vomiting that are typically self-limited to 1 week. Rarer but more serious AEs include bleeding, perforation, or infection, and occur in 2% of cases based on large trial data.5,7
Although the weight loss benefits of ESG are well established, to date, there are limited data on the effects of endoscopic bariatric therapies like ESG on mental health conditions. Here, we describe a case of a veteran with a history of mental health disorders that prevented him from completing bariatric surgery. The patient underwent ESG and had a successful clinical course.
CASE PRESENTATION
A 59-year-old male veteran with a medical history of class III obesity (42.4 BMI), obstructive sleep apnea, hypothyroidism, hypertension, type 2 diabetes mellitus, and a large ventral hernia was referred to the MOVE! (Management of Overweight/ Obese Veterans Everywhere!) multidisciplinary high-intensity weight loss program at the US Department of Veterans Affairs (VA) West Los Angeles VA Medical Center (WLAVAMC). His psychiatric history included generalized anxiety disorder, posttraumatic stress disorder (PTSD), and panic disorder, managed by the Psychiatry Service and treated with sertraline 25 mg daily, lorazepam 0.5 mg twice daily, and hydroxyzine 20 mg nightly. He had previously implemented lifestyle changes and attended MOVE! classes and nutrition coaching for 1 year but was unsuccessful in losing weight. He had also tried liraglutide 3 mg daily for weight loss but was unable to tolerate it and reported worsening medication-related anxiety.
The patient declined further weight loss pharmacotherapy and was referred to bariatric surgery. He was scheduled for a surgical sleeve gastrectomy. However, on the day he arrived at the hospital for surgery, he developed severe anxiety and had a panic attack, and it was canceled. Due to his mental health issues, he was no longer comfortable proceeding with surgery and was left without other options for obesity treatment. The veteran was extremely disappointed because the ventral hernia caused significant quality of life impairment, limited his ability to exercise, and caused him embarrassment in public settings. The hernia could not be surgically repaired until there was significant weight loss.
A bariatric endoscopy program within the Division of Gastroenterology was developed and implemented at the WLAVAMC in February 2023 in conjunction with MOVE! The patient was referred for consideration of an endoscopic weight loss procedure. He was determined to be a suitable candidate for ESG based on his BMI being > 40 and personal preference not to proceed with surgery to lose enough weight to qualify for hernia repair. The veteran underwent an endoscopy, which showed normal anatomy and gastric mucosa. ESG was performed in standard fashion (Figure).8 Three vertical lines were made using argon plasma coagulation from the incisura to 2 cm below the gastroesophageal junction along the anterior, posterior, and greater curvature of the stomach to mark the area for endoscopic suture placement. Starting at the incisura, 7 full-thickness sutures were placed to create a volume reduction plication, with preservation of the fundus. The patient did well postprocedure with no immediate or delayed AEs and was discharged home the same day.

Follow-up
The veteran followed a gradual dietary advancement from a clear liquid diet to pureed and soft texture food. The patient’s weight dropped from 359 lbs preprocedure to 304 lbs 6 months postprocedure, a total body weight loss (TWBL) of 15.3%. At 12 months the veteran weighed 299 lbs (16.7% TBWL). He also had notable improvements in metabolic parameters. His systolic blood pressure decreased from ≥ 140 mm Hg to 120 to 130 mm Hg and hemoglobin A1c dropped from 7.0% to 6.3%. Remarkably, his psychiatrist noted significant improvement in his overall mental health. The veteran reported complete cessation of panic attacks since the ESG, improvements in PTSD and anxiety, and was able to discontinue lorazepam and decrease his dose of sertraline to 12.5 mg daily. He reported feeling more energetic and goal-oriented with increased clarity of thought. Perhaps the most significant outcome was that after the 55-lb weight loss at 6 months, the patient was eligible to undergo ventral hernia surgical repair, which had previously contributed to shame and social isolation. This, in turn, improved his quality of life, allowed him to start walking again, up to 8 miles daily, and to feel comfortable again going out in public settings.
DISCUSSION
Bariatric surgeries are an effective method of achieving weight loss and improving obesity-related comorbidities. However, only a small percentage of individuals with obesity are candidates for bariatric surgery. Given the dramatic increase in the prevalence of obesity, other options are needed. Specifically, within the VA, an estimated 80% of veterans are overweight or obese, but only about 500 bariatric surgeries are performed annually.9 With the need for additional weight loss therapies, VA programs are starting to offer endoscopic bariatric procedures as an alternative option. This may be a desirable choice for patients with obesity (BMI > 30), with or without associated metabolic comorbidities, who need more aggressive intervention beyond dietary and lifestyle changes and are either not interested in or not eligible for bariatric surgery or weight loss medications.
Although there is evidence that metabolic comorbidities are associated with obesity, there has been less research on obesity and mental health comorbidities such as depression and anxiety. These psychiatric conditions may even be more common among patients seeking weight loss procedures and more prominent in certain groups such as veterans, which may ultimately exclude these patients from bariatric surgery.10 Prior studies suggest that bariatric surgery can reduce the severity of depression and, to a lesser extent, anxiety symptoms at 2 years following the initial surgery; however, there is limited literature describing the impact of weight loss procedure on panic disorders.11-14 We suspect that a weight loss procedure such as ESG may have indirectly improved the veteran’s mood disorder due to the weight loss it induced, increasing the ability to exercise, quality of sleep, and participation in public settings.
This case highlights a veteran who did not tolerate weight loss medication and had severe anxiety and PTSD that prevented him from going through with bariatric surgery. He then underwent an endoscopic weight loss procedure. The ESG helped him successfully achieve significant weight loss, increase his physical activity, reduce his anxiety and panic disorder, and overall, significantly improve his quality of life. More than 1 year after the procedure, the patient has sustained improvements in his psychiatric and emotional health along with durable weight loss, maintaining > 15% of his total weight lost. Additional studies are needed to further understand the prevalence and long-term outcomes of mental health comorbidities, as well as weight loss outcomes in this group of patients who undergo endoscopic bariatric procedures.
CONCLUSIONS
We describe a case of a veteran with severe obesity and significant psychiatric comorbidities that prevented him from undergoing bariatric surgery, who underwent an ESG. This procedure led to significant weight loss, improvement of metabolic parameters, reduction in anxiety and PTSD, and enhancement of his quality of life. This case emphasizes the unique advantages of ESG and supports the expansion of endoscopic bariatric programs in the VA.
- Ritchie SA, Connell JM. The link between abdominal obesity, metabolic syndrome and cardiovascular disease. Nutr Metab Cardiovasc Dis. 2007;17(4):319-326. doi:10.1016/j.numecd.2006.07.005
- Bray GA, Kim KK, Wilding JPH; World Obesity Federation. Obesity: a chronic relapsing progressive disease process. A position statement of the World Obesity Federation. Obes Rev. 2017;18(7):715-723. doi:10.1111/obr.12551
- Imbus JR, Voils CI, Funk LM. Bariatric surgery barriers: a review using andersen’s model of health services use. Surg Obes Relat Dis. 2018;14(3):404-412. doi:10.1016/j.soard.2017.11.012
- Dawes AJ, Maggard-Gibbons M, Maher AR, et al. Mental health conditions among patients seeking and undergoing bariatric surgery: a meta-analysis. JAMA. 2016;315(2):150- 163. doi:10.1001/jama.2015.18118
- Abu Dayyeh BK, Bazerbachi F, Vargas EJ, et al.. Endoscopic sleeve gastroplasty for treatment of class 1 and 2 obesity (MERIT): a prospective, multicentre, randomised trial. Lancet. 2022;400(10350):441-451. doi:10.1016/S0140-6736(22)01280-6
- Matteo MV, Bove V, Ciasca G, et al. Success predictors of endoscopic sleeve gastroplasty. Obes Surg. 2024;34(5):1496-1504. doi:10.1007/s11695-024-07109-4
- Maselli DB, Hoff AC, Kucera A, et al. Endoscopic sleeve gastroplasty in class III obesity: efficacy, safety, and durability outcomes in 404 consecutive patients. World J Gastrointest Endosc. 2023;15(6):469-479. doi:10.4253/wjge.v15.i6.469
- Kumar N, Abu Dayyeh BK, Lopez-Nava Breviere G, et al. Endoscopic sutured gastroplasty: procedure evolution from first-in-man cases through current technique. Surg Endosc. 2018;32(4):2159-2164. doi:10.1007/s00464-017-5869-2
- Maggard-Gibbons M, Shekelle PG, Girgis MD, et al. Endoscopic Bariatric Interventions versus lifestyle interventions or surgery for weight loss in patients with obesity: a systematic review and meta-analysis. Department of Veterans Affairs (US); 2022. https://www.ncbi.nlm.nih.gov/books/NBK587943/
- Maggard Gibbons MA, Maher AM, Dawes AJ, et al. Psychological clearance for bariatric surgery: a systematic review. VA-ESP project #05-2262014.
- van Hout GC, Verschure SK, van Heck GL. Psychosocial predictors of success following bariatric surgery. Obes Surg. 2005;15(4):552-560. doi:10.1381/0960892053723484
- Hudson JI, Hiripi E, Pope HG Jr, Kessler RC. The prevalence and correlates of eating disorders in the national comorbidity survey replication. Biol Psychiatry. 2007;61(3):348-358. doi:10.1016/j.biopsych.2006.03.040
- Aylward L, Lilly C, Konsor M, et al. How soon do depression and anxiety symptoms improve after bariatric surgery?. Healthcare (Basel). 2023;11(6):862. doi:10.3390/healthcare11060862
- Law S, Dong S, Zhou F, Zheng D, Wang C, Dong Z. Bariatric surgery and mental health outcomes: an umbrella review. Front Endocrinol (Lausanne). 2023;14:1283621. doi:10.3389/fendo.2023.1283621
- Ritchie SA, Connell JM. The link between abdominal obesity, metabolic syndrome and cardiovascular disease. Nutr Metab Cardiovasc Dis. 2007;17(4):319-326. doi:10.1016/j.numecd.2006.07.005
- Bray GA, Kim KK, Wilding JPH; World Obesity Federation. Obesity: a chronic relapsing progressive disease process. A position statement of the World Obesity Federation. Obes Rev. 2017;18(7):715-723. doi:10.1111/obr.12551
- Imbus JR, Voils CI, Funk LM. Bariatric surgery barriers: a review using andersen’s model of health services use. Surg Obes Relat Dis. 2018;14(3):404-412. doi:10.1016/j.soard.2017.11.012
- Dawes AJ, Maggard-Gibbons M, Maher AR, et al. Mental health conditions among patients seeking and undergoing bariatric surgery: a meta-analysis. JAMA. 2016;315(2):150- 163. doi:10.1001/jama.2015.18118
- Abu Dayyeh BK, Bazerbachi F, Vargas EJ, et al.. Endoscopic sleeve gastroplasty for treatment of class 1 and 2 obesity (MERIT): a prospective, multicentre, randomised trial. Lancet. 2022;400(10350):441-451. doi:10.1016/S0140-6736(22)01280-6
- Matteo MV, Bove V, Ciasca G, et al. Success predictors of endoscopic sleeve gastroplasty. Obes Surg. 2024;34(5):1496-1504. doi:10.1007/s11695-024-07109-4
- Maselli DB, Hoff AC, Kucera A, et al. Endoscopic sleeve gastroplasty in class III obesity: efficacy, safety, and durability outcomes in 404 consecutive patients. World J Gastrointest Endosc. 2023;15(6):469-479. doi:10.4253/wjge.v15.i6.469
- Kumar N, Abu Dayyeh BK, Lopez-Nava Breviere G, et al. Endoscopic sutured gastroplasty: procedure evolution from first-in-man cases through current technique. Surg Endosc. 2018;32(4):2159-2164. doi:10.1007/s00464-017-5869-2
- Maggard-Gibbons M, Shekelle PG, Girgis MD, et al. Endoscopic Bariatric Interventions versus lifestyle interventions or surgery for weight loss in patients with obesity: a systematic review and meta-analysis. Department of Veterans Affairs (US); 2022. https://www.ncbi.nlm.nih.gov/books/NBK587943/
- Maggard Gibbons MA, Maher AM, Dawes AJ, et al. Psychological clearance for bariatric surgery: a systematic review. VA-ESP project #05-2262014.
- van Hout GC, Verschure SK, van Heck GL. Psychosocial predictors of success following bariatric surgery. Obes Surg. 2005;15(4):552-560. doi:10.1381/0960892053723484
- Hudson JI, Hiripi E, Pope HG Jr, Kessler RC. The prevalence and correlates of eating disorders in the national comorbidity survey replication. Biol Psychiatry. 2007;61(3):348-358. doi:10.1016/j.biopsych.2006.03.040
- Aylward L, Lilly C, Konsor M, et al. How soon do depression and anxiety symptoms improve after bariatric surgery?. Healthcare (Basel). 2023;11(6):862. doi:10.3390/healthcare11060862
- Law S, Dong S, Zhou F, Zheng D, Wang C, Dong Z. Bariatric surgery and mental health outcomes: an umbrella review. Front Endocrinol (Lausanne). 2023;14:1283621. doi:10.3389/fendo.2023.1283621
Endoscopic Sleeve Gastroplasty is an Effective Treatment for Obesity in a Veteran With Metabolic and Psychiatric Comorbidities
Endoscopic Sleeve Gastroplasty is an Effective Treatment for Obesity in a Veteran With Metabolic and Psychiatric Comorbidities
Weight Loss May Cut Cancer Risk in Adults With Obesity
Weight Loss May Cut Cancer Risk in Adults With Obesity
TOPLINE:
Among adults with obesity, nonsurgical weight loss was significantly associated with reduced odds for developing obesity-related and other cancers at 3 and 5 years, a study of real-world data found.
METHODOLOGY:
- Although weight loss after bariatric surgery is linked to a reduced risk for cancer, the effect of nonsurgical weight loss on cancer risk remains unclear.
- Researchers conducted a retrospective observational study using electronic health record data from a US health system to assess the association between nonsurgical weight loss and the risk for cancer among adults with obesity.
- The inclusion criteria were age of ≥ 20 years, BMI > 30, and at least seven health system visits over 3 years. Patients with a history of alcohol or substance abuse, amputations, HIV infection, organ transplant, thyroid problems, or those who underwent bariatric surgery were excluded.
- The 143,630 patients who met inclusion criteria (7703 cancer cases and 135,927 controls) were divided into 3 cohorts based on weight change over time intervals of 3 years (115,942 patients), 5 years (105,472 patients), and 10 years (59,112 patients).
- Primary endpoints included obesity-related cancers (esophageal cancer, liver cancer, gallbladder cancer, pancreatic cancer, colorectal cancer, renal cell carcinoma, endometrial cancer, multiple myeloma, and postmenopausal breast cancer), and secondary endpoints included all malignant neoplasms.
TAKEAWAY:
- Each 1% reduction in BMI was associated with reduced odds of obesity-related cancers at 3 years and 5 years (odds ratio [OR], 0.99 and 0.989, respectively; P < .001 for both). These results translate to 5% weight loss corresponding to 4.9% and 5.4% reductions in obesity-related cancer odds at 3 and 5 years, respectively.
- Weight loss was associated with reduced odds of endometrial cancer at 3, 5, and 10 years (OR, 0.978; P < .05), of renal cell carcinoma at 3 and 5 years (OR, 0.983; P < .05), and of multiple myeloma at 10 years (OR, 0.969; P = .004).
- Weight loss was also associated with reduced odds of developing any malignancy at 3 years (OR, 0.992), 5 years (OR, 0.994), and 10 years (OR, 0.991; P = .001 for all). These results translate into a 5% weight loss corresponding to 3.9%, 3%, and 4.4% lower odds of any malignancy at 3, 5, and 10 years, respectively.
IN PRACTICE:
"Real-world weight loss was associated with a decreased risk of developing obesity-related cancers and all other cancers. Our study serves as a call for action and a strong public health message to healthcare stakeholders to intensify efforts and resources to treat obesity as a chronic disease to help reduce the risk of developing cancer," the author wrote.
SOURCE:
This study, led by endocrinologist Kenda Alkwatli, MD, Starling Physicians, Wethersfield, Connecticut, was published online in Obesity.
LIMITATIONS:
The study included only individuals with sufficient longitudinal health records, which may have introduced selection bias. It could not distinguish between intentional and unintentional weight loss or differentiate between fat and lean mass. Due to its observational nature, the study could not assess whether weight loss preceding cancer diagnosis was related to delay in diagnosis.
DISCLOSURES:
The study was funded in part by the Cleveland Clinic Center for Quantitative Metabolic Research. Three authors reported receiving research funding, consulting fees, honoraria, grants, or research support and holding patent applications, license agreements, leadership roles, or equity in healthcare and biotechnology companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Among adults with obesity, nonsurgical weight loss was significantly associated with reduced odds for developing obesity-related and other cancers at 3 and 5 years, a study of real-world data found.
METHODOLOGY:
- Although weight loss after bariatric surgery is linked to a reduced risk for cancer, the effect of nonsurgical weight loss on cancer risk remains unclear.
- Researchers conducted a retrospective observational study using electronic health record data from a US health system to assess the association between nonsurgical weight loss and the risk for cancer among adults with obesity.
- The inclusion criteria were age of ≥ 20 years, BMI > 30, and at least seven health system visits over 3 years. Patients with a history of alcohol or substance abuse, amputations, HIV infection, organ transplant, thyroid problems, or those who underwent bariatric surgery were excluded.
- The 143,630 patients who met inclusion criteria (7703 cancer cases and 135,927 controls) were divided into 3 cohorts based on weight change over time intervals of 3 years (115,942 patients), 5 years (105,472 patients), and 10 years (59,112 patients).
- Primary endpoints included obesity-related cancers (esophageal cancer, liver cancer, gallbladder cancer, pancreatic cancer, colorectal cancer, renal cell carcinoma, endometrial cancer, multiple myeloma, and postmenopausal breast cancer), and secondary endpoints included all malignant neoplasms.
TAKEAWAY:
- Each 1% reduction in BMI was associated with reduced odds of obesity-related cancers at 3 years and 5 years (odds ratio [OR], 0.99 and 0.989, respectively; P < .001 for both). These results translate to 5% weight loss corresponding to 4.9% and 5.4% reductions in obesity-related cancer odds at 3 and 5 years, respectively.
- Weight loss was associated with reduced odds of endometrial cancer at 3, 5, and 10 years (OR, 0.978; P < .05), of renal cell carcinoma at 3 and 5 years (OR, 0.983; P < .05), and of multiple myeloma at 10 years (OR, 0.969; P = .004).
- Weight loss was also associated with reduced odds of developing any malignancy at 3 years (OR, 0.992), 5 years (OR, 0.994), and 10 years (OR, 0.991; P = .001 for all). These results translate into a 5% weight loss corresponding to 3.9%, 3%, and 4.4% lower odds of any malignancy at 3, 5, and 10 years, respectively.
IN PRACTICE:
"Real-world weight loss was associated with a decreased risk of developing obesity-related cancers and all other cancers. Our study serves as a call for action and a strong public health message to healthcare stakeholders to intensify efforts and resources to treat obesity as a chronic disease to help reduce the risk of developing cancer," the author wrote.
SOURCE:
This study, led by endocrinologist Kenda Alkwatli, MD, Starling Physicians, Wethersfield, Connecticut, was published online in Obesity.
LIMITATIONS:
The study included only individuals with sufficient longitudinal health records, which may have introduced selection bias. It could not distinguish between intentional and unintentional weight loss or differentiate between fat and lean mass. Due to its observational nature, the study could not assess whether weight loss preceding cancer diagnosis was related to delay in diagnosis.
DISCLOSURES:
The study was funded in part by the Cleveland Clinic Center for Quantitative Metabolic Research. Three authors reported receiving research funding, consulting fees, honoraria, grants, or research support and holding patent applications, license agreements, leadership roles, or equity in healthcare and biotechnology companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Among adults with obesity, nonsurgical weight loss was significantly associated with reduced odds for developing obesity-related and other cancers at 3 and 5 years, a study of real-world data found.
METHODOLOGY:
- Although weight loss after bariatric surgery is linked to a reduced risk for cancer, the effect of nonsurgical weight loss on cancer risk remains unclear.
- Researchers conducted a retrospective observational study using electronic health record data from a US health system to assess the association between nonsurgical weight loss and the risk for cancer among adults with obesity.
- The inclusion criteria were age of ≥ 20 years, BMI > 30, and at least seven health system visits over 3 years. Patients with a history of alcohol or substance abuse, amputations, HIV infection, organ transplant, thyroid problems, or those who underwent bariatric surgery were excluded.
- The 143,630 patients who met inclusion criteria (7703 cancer cases and 135,927 controls) were divided into 3 cohorts based on weight change over time intervals of 3 years (115,942 patients), 5 years (105,472 patients), and 10 years (59,112 patients).
- Primary endpoints included obesity-related cancers (esophageal cancer, liver cancer, gallbladder cancer, pancreatic cancer, colorectal cancer, renal cell carcinoma, endometrial cancer, multiple myeloma, and postmenopausal breast cancer), and secondary endpoints included all malignant neoplasms.
TAKEAWAY:
- Each 1% reduction in BMI was associated with reduced odds of obesity-related cancers at 3 years and 5 years (odds ratio [OR], 0.99 and 0.989, respectively; P < .001 for both). These results translate to 5% weight loss corresponding to 4.9% and 5.4% reductions in obesity-related cancer odds at 3 and 5 years, respectively.
- Weight loss was associated with reduced odds of endometrial cancer at 3, 5, and 10 years (OR, 0.978; P < .05), of renal cell carcinoma at 3 and 5 years (OR, 0.983; P < .05), and of multiple myeloma at 10 years (OR, 0.969; P = .004).
- Weight loss was also associated with reduced odds of developing any malignancy at 3 years (OR, 0.992), 5 years (OR, 0.994), and 10 years (OR, 0.991; P = .001 for all). These results translate into a 5% weight loss corresponding to 3.9%, 3%, and 4.4% lower odds of any malignancy at 3, 5, and 10 years, respectively.
IN PRACTICE:
"Real-world weight loss was associated with a decreased risk of developing obesity-related cancers and all other cancers. Our study serves as a call for action and a strong public health message to healthcare stakeholders to intensify efforts and resources to treat obesity as a chronic disease to help reduce the risk of developing cancer," the author wrote.
SOURCE:
This study, led by endocrinologist Kenda Alkwatli, MD, Starling Physicians, Wethersfield, Connecticut, was published online in Obesity.
LIMITATIONS:
The study included only individuals with sufficient longitudinal health records, which may have introduced selection bias. It could not distinguish between intentional and unintentional weight loss or differentiate between fat and lean mass. Due to its observational nature, the study could not assess whether weight loss preceding cancer diagnosis was related to delay in diagnosis.
DISCLOSURES:
The study was funded in part by the Cleveland Clinic Center for Quantitative Metabolic Research. Three authors reported receiving research funding, consulting fees, honoraria, grants, or research support and holding patent applications, license agreements, leadership roles, or equity in healthcare and biotechnology companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Weight Loss May Cut Cancer Risk in Adults With Obesity
Weight Loss May Cut Cancer Risk in Adults With Obesity
GLP-1s Lower Risk of SUDs in VA Studies
Two studies published in March by researchers at the Veterans Affairs Saint Louis Healthcare System highlight the clinical significance of glucagon-like peptide 1 receptor agonists (GLP-1s) and their impact on reducing substance use disorder (SUD) risks. The studies also explore the impact of GLP-1 discontinuation or interruption on their effectiveness in protection against the cardiovascular events.
In one study, Al-Aly et al assigned 606,434 veterans with type 2 diabetes to 1 of 2 protocols, comparing GLP-1s with sodium-glucose cotransporter-2 (SGLT-2) inhibitors, and followed the patients for up to 3 years. Al-Aly et al found that GLP-1s were “consistently associated” with a lower risk of developing SUDs, including those involving alcohol, cannabis, cocaine, nicotine, and opioids. The findings suggested “potential preventive effects across a broad range of addictive substances.”
In participants with pre-existing SUDs, GLP-1s were also associated with reduced risks of SUD-related emergency department visits, hospital admissions, and mortality, in addition to drug overdoses and suicidal behaviors. A study published in 2025 from the same research group reported that GLP-1s could have a variety of health benefits, including reducing the risk of incident alcohol and cannabis disorders, neurocognitive disorders (such as Alzheimer's disease and dementia), coagulation disorders, cardiometabolic disorders, infectious illnesses and several respiratory conditions, but less was known about the potential for preventing development of opioid use disorder and other SUDs.
GLP-1s target the brain’s reward pathways and have recently made attention-grabbing headlines regarding celebrity weight loss, with social media boosting public interest. One study, for example, found 100 videos on TikTok with the #Ozempic viewed nearly 70 million times.
Al-Aly et al used SGLT-2 inhibitors as active comparators because “they have no established direct actions on mesolimbic reward circuits in the brain, whereas GLP-1 receptors are present in areas of the brain involved in impulse control and reward signaling.”
The second study found that quitting or pausing GLP-1 treatment for 6 months could have a rebound effect and possibly reverse any progress. Discontinuing GLP-1 treatment is common, with rates ranging from 36% to 81% in the first year. Stopping or interrupting the treatment is often followed by weight regain and a rebound in inflammation, both major drivers in cardiovascular disease risk.
The study followed 132,551 VA patients using GLP-1s and 201,136 using sulfonylureas from 2017 through 2023. About two-thirds of participants took semaglutide, prescribed as Ozempic to treat diabetes and Wegovy to reduce obesity. A total of 26% of the participants stopped GLP-1 treatment during the follow-up period, with 64% occurring during the first year. Most (67%) treatment interruptions also came in the first year.
Compared with incident use of sulfonylureas, incident use of GLP-1s was associated with a reduced risk of heart attack, stroke, or death. Patients who took the GLP-1s without interruption > 3 years experienced an 18% lower risk for heart attack or stroke.
Cardiovascular benefits accumulated with continuous use over 3 years, but even brief periods of discontinuations or interruptions could progressively erode and ultimately reverse this protection, the researchers found. Discontinuing treatment for half a year was associated with an increased risk of major adverse cardiovascular events (incidence risk ratio [IRR], 1.04), while longer gaps were progressively associated with a higher risk of disease (IRR, 1.12 for 1 year; IRR, 1.16 for 2 years of interrupted use, respectively).
Dr. Ziyad Al-Aly, a study author and Chief of the Research and Education Service at the Veterans Affairs Saint Louis Healthcare System, called it “metabolic whiplash.” In an interview, he said it was important to caution patients that these medications “need to be taken for the long haul. This is not something (patients) can take for a month or 2 or 3 and get off of it. It's not going to work like that.”
Two studies published in March by researchers at the Veterans Affairs Saint Louis Healthcare System highlight the clinical significance of glucagon-like peptide 1 receptor agonists (GLP-1s) and their impact on reducing substance use disorder (SUD) risks. The studies also explore the impact of GLP-1 discontinuation or interruption on their effectiveness in protection against the cardiovascular events.
In one study, Al-Aly et al assigned 606,434 veterans with type 2 diabetes to 1 of 2 protocols, comparing GLP-1s with sodium-glucose cotransporter-2 (SGLT-2) inhibitors, and followed the patients for up to 3 years. Al-Aly et al found that GLP-1s were “consistently associated” with a lower risk of developing SUDs, including those involving alcohol, cannabis, cocaine, nicotine, and opioids. The findings suggested “potential preventive effects across a broad range of addictive substances.”
In participants with pre-existing SUDs, GLP-1s were also associated with reduced risks of SUD-related emergency department visits, hospital admissions, and mortality, in addition to drug overdoses and suicidal behaviors. A study published in 2025 from the same research group reported that GLP-1s could have a variety of health benefits, including reducing the risk of incident alcohol and cannabis disorders, neurocognitive disorders (such as Alzheimer's disease and dementia), coagulation disorders, cardiometabolic disorders, infectious illnesses and several respiratory conditions, but less was known about the potential for preventing development of opioid use disorder and other SUDs.
GLP-1s target the brain’s reward pathways and have recently made attention-grabbing headlines regarding celebrity weight loss, with social media boosting public interest. One study, for example, found 100 videos on TikTok with the #Ozempic viewed nearly 70 million times.
Al-Aly et al used SGLT-2 inhibitors as active comparators because “they have no established direct actions on mesolimbic reward circuits in the brain, whereas GLP-1 receptors are present in areas of the brain involved in impulse control and reward signaling.”
The second study found that quitting or pausing GLP-1 treatment for 6 months could have a rebound effect and possibly reverse any progress. Discontinuing GLP-1 treatment is common, with rates ranging from 36% to 81% in the first year. Stopping or interrupting the treatment is often followed by weight regain and a rebound in inflammation, both major drivers in cardiovascular disease risk.
The study followed 132,551 VA patients using GLP-1s and 201,136 using sulfonylureas from 2017 through 2023. About two-thirds of participants took semaglutide, prescribed as Ozempic to treat diabetes and Wegovy to reduce obesity. A total of 26% of the participants stopped GLP-1 treatment during the follow-up period, with 64% occurring during the first year. Most (67%) treatment interruptions also came in the first year.
Compared with incident use of sulfonylureas, incident use of GLP-1s was associated with a reduced risk of heart attack, stroke, or death. Patients who took the GLP-1s without interruption > 3 years experienced an 18% lower risk for heart attack or stroke.
Cardiovascular benefits accumulated with continuous use over 3 years, but even brief periods of discontinuations or interruptions could progressively erode and ultimately reverse this protection, the researchers found. Discontinuing treatment for half a year was associated with an increased risk of major adverse cardiovascular events (incidence risk ratio [IRR], 1.04), while longer gaps were progressively associated with a higher risk of disease (IRR, 1.12 for 1 year; IRR, 1.16 for 2 years of interrupted use, respectively).
Dr. Ziyad Al-Aly, a study author and Chief of the Research and Education Service at the Veterans Affairs Saint Louis Healthcare System, called it “metabolic whiplash.” In an interview, he said it was important to caution patients that these medications “need to be taken for the long haul. This is not something (patients) can take for a month or 2 or 3 and get off of it. It's not going to work like that.”
Two studies published in March by researchers at the Veterans Affairs Saint Louis Healthcare System highlight the clinical significance of glucagon-like peptide 1 receptor agonists (GLP-1s) and their impact on reducing substance use disorder (SUD) risks. The studies also explore the impact of GLP-1 discontinuation or interruption on their effectiveness in protection against the cardiovascular events.
In one study, Al-Aly et al assigned 606,434 veterans with type 2 diabetes to 1 of 2 protocols, comparing GLP-1s with sodium-glucose cotransporter-2 (SGLT-2) inhibitors, and followed the patients for up to 3 years. Al-Aly et al found that GLP-1s were “consistently associated” with a lower risk of developing SUDs, including those involving alcohol, cannabis, cocaine, nicotine, and opioids. The findings suggested “potential preventive effects across a broad range of addictive substances.”
In participants with pre-existing SUDs, GLP-1s were also associated with reduced risks of SUD-related emergency department visits, hospital admissions, and mortality, in addition to drug overdoses and suicidal behaviors. A study published in 2025 from the same research group reported that GLP-1s could have a variety of health benefits, including reducing the risk of incident alcohol and cannabis disorders, neurocognitive disorders (such as Alzheimer's disease and dementia), coagulation disorders, cardiometabolic disorders, infectious illnesses and several respiratory conditions, but less was known about the potential for preventing development of opioid use disorder and other SUDs.
GLP-1s target the brain’s reward pathways and have recently made attention-grabbing headlines regarding celebrity weight loss, with social media boosting public interest. One study, for example, found 100 videos on TikTok with the #Ozempic viewed nearly 70 million times.
Al-Aly et al used SGLT-2 inhibitors as active comparators because “they have no established direct actions on mesolimbic reward circuits in the brain, whereas GLP-1 receptors are present in areas of the brain involved in impulse control and reward signaling.”
The second study found that quitting or pausing GLP-1 treatment for 6 months could have a rebound effect and possibly reverse any progress. Discontinuing GLP-1 treatment is common, with rates ranging from 36% to 81% in the first year. Stopping or interrupting the treatment is often followed by weight regain and a rebound in inflammation, both major drivers in cardiovascular disease risk.
The study followed 132,551 VA patients using GLP-1s and 201,136 using sulfonylureas from 2017 through 2023. About two-thirds of participants took semaglutide, prescribed as Ozempic to treat diabetes and Wegovy to reduce obesity. A total of 26% of the participants stopped GLP-1 treatment during the follow-up period, with 64% occurring during the first year. Most (67%) treatment interruptions also came in the first year.
Compared with incident use of sulfonylureas, incident use of GLP-1s was associated with a reduced risk of heart attack, stroke, or death. Patients who took the GLP-1s without interruption > 3 years experienced an 18% lower risk for heart attack or stroke.
Cardiovascular benefits accumulated with continuous use over 3 years, but even brief periods of discontinuations or interruptions could progressively erode and ultimately reverse this protection, the researchers found. Discontinuing treatment for half a year was associated with an increased risk of major adverse cardiovascular events (incidence risk ratio [IRR], 1.04), while longer gaps were progressively associated with a higher risk of disease (IRR, 1.12 for 1 year; IRR, 1.16 for 2 years of interrupted use, respectively).
Dr. Ziyad Al-Aly, a study author and Chief of the Research and Education Service at the Veterans Affairs Saint Louis Healthcare System, called it “metabolic whiplash.” In an interview, he said it was important to caution patients that these medications “need to be taken for the long haul. This is not something (patients) can take for a month or 2 or 3 and get off of it. It's not going to work like that.”
Seventy-Five Percent of Total Energy Intake Comes From Ultra-Processed Foods Among a Sample of Veterans With Overweight and Obesity: An Exploratory Analysis of Three-Day Food Records
Seventy-Five Percent of Total Energy Intake Comes From Ultra-Processed Foods Among a Sample of Veterans With Overweight and Obesity: An Exploratory Analysis of Three-Day Food Records
Roughly 8.6% of the 17.4 million US veterans live in poverty. About 11.1% are considered food insecure (ie, unable to acquire adequate food for ≥1 household members), with another 5.3% considered very food insecure (ie, eating patterns of ≥1 household members were disrupted and their food intake was reduced at least some time during the year). Compared with nonveterans, veterans are 7.4% more likely to be food insecure.1 This high prevalence of food insecurity and poverty has a negative impact on veteran diets.
Veterans’ diets contained more added sugars and solid fats and scored lower compared with nonveterans when assessed for diet quality with the Healthy Eating Index.2 Veterans have a higher prevalence of diet-related chronic disease, including diabetes, hypertension, and obesity compared with the nonveterans.3-5 Given the critical role of diet in health and disease risk, enhancing diet quality among veterans has garnered significant attention and calls to action.2,6,7 While there are many factors that contribute to diet, any veteran can receive a consultation or self-refer to receive nutrition counseling effective for improving diet quality, within the US Department of Veterans Affairs (VA).
The NOVA food classification system describes diet quality by categorizing food items by processing methods and ingredients into 4 food groups.8 The first is unprocessed and minimally processed items (MPFs) such as fresh fruits, vegetables, and meats. MPFs consist of whole foods which can also be minimally processed (eg, chopping, drying, grinding, heating, chilling). Culinary processed foods (CPFs) are processed foods for cooking (eg, salt, butter, and vinegar) and are typically eaten in small quantities along with MPFs. Processed foods (PRFs) include canned and smoked foods, while ultra-processed foods (UPFs) are distinguished by industrial ingredients, requiring specialized tools and processing techniques, and hyper-palatability related to color, flavor, and packaging.8 Examples of UPFs include mass-produced breads found at grocery stores, prepackaged snacks and meals, and hydrogenated oils. UPF consumption is associated with higher risk for negative cardiometabolic outcomes, common mental disorders, and all-cause mortality.9 To date, only a study by Powell et al has used the NOVA classification system in a veteran population, and it was limited to a comparison of the price of UPFs and veteran body mass index (BMI).10 Therefore, it remains unknown what percentage of total energy intake (TEI) comes from UPFs in the diets of veterans.
This study sought to quantify the proportion of TEI from UPFs among a sample of patients from the VA Phoenix Health Care System (VAPHCS). Results from a 2021 global meta-analysis reveal that the US and United Kingdom have the highest intakes of UPFs in the world.11 Specifically, within the US, 15 studies with 234,890 participants reveal that the majority of TEI (about 55%) comes from UPFs.11 We hypothesized that this veteran sample would have a higher proportion of TEI from UPFs, possibly due to a higher prevalence of poverty and food insecurity among veterans compared with nonveterans.1 If the percentage of TEI coming from UPF is higher or even similar to nonveterans, further efforts to increase veterans’ use of the available nutritional services would be warranted to minimize nutrition-related disease among veterans.
Methods
This is a cross-sectional, secondary data analysis of baseline 3-day food records collected from 2017 to 2020 from 92 patients recruited at VAPHCS to participate in a whole-food plant-based diet study.12 The original study was reviewed and approved by the VAPHCS Institutional Review Board (1593830). Recruitment methods included clinician recommendation, a recorded advertisement played while phone calls were on hold, and flyers distributed throughout VAPHCS. Patients were included if they were aged 18 to 90 years, had a BMI 25.1 to 39.9, had a diagnosis of nutrition-related chronic disease (hypertension, diabetes, or hyperlipidemia), an interest and desire to make a lifestyle change, active telephone contact information (either landline or cell phone), no contraindication to be on a whole-food plant-based diet, access to transportation and a functioning kitchen, ability to prepare meals independently, access to a computer or tablet with internet access, and a digital camera or smartphone. Exclusion criteria included significant unplanned weight loss within 6 months, uncontrolled insulin-dependent diabetes with a current hemoglobin A1c > 9%, pregnancy/lactation, taking prescribed weight loss medication, currently following a diet (eg, plant-based diet, vegan, or medical weight loss program diet), celiac disease diagnosed within 6 months, end-stage hepatic disease or renal disease requiring dialysis, active cancer or receiving chemotherapy or radiation therapy, active alcohol or substance use disorder, history of eating disorders, fasting triglyceride level > 350 mg/dL, any psychological issues that prevent adherence, inability to speak English, limited mobility, and homeless or in housing with limited kitchen access. A baseline 3-day food record was collected from the participants and used in this secondary analysis.
Diet Analysis
Food records were analyzed using Esha Research Food Processor 4.0 to identify calorie and macronutrient information. To limit bias, food items were coded independently by 2 researchers into 4 food processing groups determined by the NOVA classification: MPF, CPF, PRF, and UPF.8 When possible, specific ingredient information was collected using internet searches for brand product websites. Initial coding had an 89% agreement rate for food item coding between the 2 researchers. As coding was done in duplicate, a third researcher resolved disagreements. The number of food items for each processing group was determined and the mean (SD) percentage of TEI for each NOVA group was provided across participants. A 1-way analysis of variance and Tukey Multiple Comparisons Test were used to determine significance between groups with an α = .05 using Prism V9.
Results
Of the 92 participants in the original study, only 79 met inclusion criteria and had baseline diet data. The 79 veterans had a mean (SD) age of 61 (13) years and 59 (75%) were male (Table 1). Mean (SD) TEI was 1921 (815) kcal. The mean (SD) percentage of calories from carbohydrate, fat, and protein were 46% (21%), 39% (20%), and 16% (6%), respectively (Table 2).
A mean (SD) of 36 (12) food items were analyzed from the 3-day food records. The majority of food items were UPFs (56%), 33% were MPFs, 8% were PRFs, and 3% were CPFs. In total, 75% of TEI came from UPFs (P < .001); only 14% of TEI came from minimally processed foods (Figure).
Discussion
To our knowledge, this is the first analysis of UPF consumption among US veterans. TEIs coming from UPFs appear to be about 20% higher among veterans compared to nonveterans: 75% vs 55%.11 Coupled with high UPF consumption, MPFs (14%) and PRFs (9%) represent smaller sources of TEI among surveyed veterans. Top caloric sources of UPFs in the US include sandwiches (including burgers), sweet bakery products, savory snacks, pizza, sweetened beverages, and breads, rolls, and tortillas, and likely reflect the major sources of UPFs in the veteran diet.13 As the statistical comparison between the veteran data and nonveteran data is not feasible in the present study, a future study with a much larger sample size would be needed for a direct comparison.
While the exact cause of higher UPF consumption among sampled veterans remains unknown and likely multifactorial (eg, cost, food insecurity, access, cooking skills, nutrition knowledge), veterans can receive a consult or self-refer to a registered dietitian nutritionist (RDN) for nutrition education. Counseling has been shown to be an effective way to improve diet quality and increase daily fruit and vegetable intake.14 High consumption of UPFs, which are generally energy-dense and nutrient-poor, contributes to the low diet quality observed in veterans, and future research examining the relationship between UPF intake and overall diet quality among veterans is warranted.2,15 As nutrition knowledge is associated with higher diet quality among veterans, increased use of nutrition services (ie, nutrition education or food supplement programs) has the potential to influence consumption of MPFs and decrease consumption of UPFs.16 Subsequently, UPF-targeted interventions developed by VA RDNs hold the promise to decrease consumption of UPFs and increase intake of MPFs and PRFs.
Veterans have a high prevalence of diabetes, hypertension, and obesity.9 The high UPF intake observed in this sample of veterans may increase the risk for these chronic diseases and overall mortality. The high percentage of TEI from UPFs among veterans is also of concern not only due to potential negative health outcomes, but also associated costs of treating veterans with multimorbidities.17 Targeting UPF intake via nutritional education may promote health and decrease the financial burden needed to support the health of veterans.
Improving veteran health and well-being, including enhancing health care accessibility in underserved areas, are pivotal objectives of the VA strategic plan for 2026 to 2030. Public policy aims to tackle food insecurity within the veteran population during the first 5 years of civilian life.18 In alignment with the White House Strategy on Hunger, Nutrition, and Health, VA established a Food Security Office (FSO) in 2023. The FSO mission is to use an interdisciplinary approach to provide resources to ensure veteran food security and create an environment where all veterans are food and nutrition secure.
Limitations
This study has several limitations. As the Food Processor software database does not include all brand items, similar brands were used to mirror the nutrient profile. While food records are common among veteran diet studies, accuracy may be reduced due to self-reporting bias.19 Different interpretation of the NOVA classification designation for various food items is possible, however, 89% of foods were coded the same by the research team which suggests high accuracy in food coding. Specific ingredient information was not collected from the 3-day food records; thus, these records were not produced in such a way to improve the accuracy of the NOVA classification designation. This study was limited by its small sample size (N = 79); although, this analysis is larger than other studies of UPF consumption in the US.20,21 In addition, the generalizability of this study is limited as this population sample was from a single VA hospital and may not reflect the overall veteran population. Participants in this study were recruited only from those receiving VA care, thus their diet quality may not represent the quality consumed by veterans not participating in VA services. Further research on UPF consumption among veterans is warranted with a larger, more representative study sample size.
Conclusions
As this is the highest observed UPF intake documented in the US, these results should be of concern for the VA and its RDNs. More research is needed to better understand why UPF consumption is so high among veterans, what barriers veterans face to decreasing UPF consumption, and what intervention(s) veterans would welcome to improve their diet quality. Presently, veterans are provided with access to a variety of effective nutrition education and counseling options and should be encouraged to use these services. VA RDNs should be aware of the high intake of UPFs in the veteran population and familiarize themselves with education and counseling strategies that promote behavior change to replace UPFs with more nutrient-dense foods choices.
- Rabbitt MP, Smith MD. Food insecurity among workingage veterans. US Dept of Agriculture, Economic Research Service; 2021. Accessed January 26, 2026. https://www.ers.usda.gov/publications/pub-details/?pubid=101268
- Dong D, Stewart H, Carlson AC. An examination of veterans’ diet quality. US Dept of Agriculture, Economic Research Service; 2019. Accessed January 26, 2026. https:// www.ers.usda.gov/publications/pub-details/?pubid=95608
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of adult overweight and obesity. 2020. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/obesity/
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. 2023. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/diabetes/
- Boersma P, Cohen R, Zelaya C, et al. Multiple chronic conditions among veterans and nonveterans: United States, 2015–2018. Natl Health Stat Rep. 2021. doi:10.15620/cdc:101659
- Hoerster KD, Wilson S, Nelson KM, et al. Diet quality is associated with mental health, social support, and neighborhood factors among veterans. Eat Behav. 2016;23:168- 173. doi:10.1016/j.eatbeh.2016.10.003
- Becerra MB, Hassija CM, Becerra BJ. Food insecurity is associated with unhealthy dietary practices among US veterans in California. Public Health Nutr. 2017;20:2569-2576. doi:10.1017/S1368980016002147
- Monteiro CA, Cannon G, Levy RB, et al. Ultra-processed foods: what they are and how to identify them. Public Health Nutr. 2019;22:936-941. doi:10.1017/S1368980018003762
- Lane MM, Gamage E, Du S, et al. Ultra-processed food exposure and adverse health outcomes: umbrella review of epidemiological meta-analyses. BMJ. February 2024:e077310. doi:10.1136/bmj-2023-077310
- Powell LM, Jones K, Duran AC, et al. The price of ultra-processed foods and beverages and adult body weight: Evidence from U.S. veterans. Econ Hum Biol. 2019;34:39- 48. doi:10.1016/j.ehb.2019.05.006
- Marino M, Puppo F, Del Bo’ C, et al. A systematic review of worldwide consumption of ultra-processed foods: findings and criticisms. Nutrients. 2021;13. doi:10.3390/nu13082778
- Parrington D, Kurtz J, Fawcett J, et al. Pilot study on the effects of a whole-food, plant-strong diet on cardiovascular risk factors in veterans: part 3. Curr Dev Nutr. 2022;6:385. doi:10.1093/cdn/nzac054.040
- Williams AM, Couch CA, Emmerich SD, et al. Ultra-processed Food Consumption in Youth and Adults: United States, August 2021-August 2023. NCHS Data Brief. 2025. doi:10.15620/cdc/174612
- Serra MC, Addison O, Giffuni J, et al. Changes in self-reported fruit and vegetable intake following nutritional modification in high risk older veterans. J Nutr Gerontol Geriatr. 2021;40:1-8. doi:10.1080/21551197.2020.1863892
- Gupta S, Hawk T, Aggarwal A, et al. Characterizing ultra-processed foods by energy density, nutrient density, and cost. Front Nutr. 2019;6:1-9. doi:10.3389/fnut.2019.00070
- Robinson LA, Colin CR, Smith KS, et al. Diet quality is associated with nutrition knowledge and physical activity in the US military veterans enrolled in university programmes. BMJ Mil Heal. 2023:e002525. doi:10.1136/military-2023-002525
- Yoon J, Zulman D, Scott JY, et al. Costs associated with multimorbidity among VA patients. Med Care. 2014;52:S31-6. doi:10.1097/MLR.0000000000000061
- End Veteran Hunger Act of 2022, H.R. 8852, 117th Cong. (2022). Accessed January 26, 2026. https://www.congress.gov/bill/117th-congress/house-bill/8852.
- Collins RA, Baker B, Coyle DH, et al. Dietary assessment methods in military and veteran populations: a scoping review. Nutrients. 2020;12:1-21. doi:10.3390/nu12030769
- Smiljanec K, Mbakwe AU, Ramos-Gonzalez M, et al. Associations of ultra-processed and unprocessed/minimally processed food consumption with peripheral and central hemodynamics, and arterial stiffness in young healthy adults. Nutrients. 2020;12. doi:10.3390/nu12113229
- Rohatgi KW, Tinius RA, Cade WT, et al. Relationships between consumption of ultra-processed foods, gestational weight gain and neonatal outcomes in a sample of US pregnant women. PeerJ. 2017;5:e4091. doi:10.7717/peerj.4091
Roughly 8.6% of the 17.4 million US veterans live in poverty. About 11.1% are considered food insecure (ie, unable to acquire adequate food for ≥1 household members), with another 5.3% considered very food insecure (ie, eating patterns of ≥1 household members were disrupted and their food intake was reduced at least some time during the year). Compared with nonveterans, veterans are 7.4% more likely to be food insecure.1 This high prevalence of food insecurity and poverty has a negative impact on veteran diets.
Veterans’ diets contained more added sugars and solid fats and scored lower compared with nonveterans when assessed for diet quality with the Healthy Eating Index.2 Veterans have a higher prevalence of diet-related chronic disease, including diabetes, hypertension, and obesity compared with the nonveterans.3-5 Given the critical role of diet in health and disease risk, enhancing diet quality among veterans has garnered significant attention and calls to action.2,6,7 While there are many factors that contribute to diet, any veteran can receive a consultation or self-refer to receive nutrition counseling effective for improving diet quality, within the US Department of Veterans Affairs (VA).
The NOVA food classification system describes diet quality by categorizing food items by processing methods and ingredients into 4 food groups.8 The first is unprocessed and minimally processed items (MPFs) such as fresh fruits, vegetables, and meats. MPFs consist of whole foods which can also be minimally processed (eg, chopping, drying, grinding, heating, chilling). Culinary processed foods (CPFs) are processed foods for cooking (eg, salt, butter, and vinegar) and are typically eaten in small quantities along with MPFs. Processed foods (PRFs) include canned and smoked foods, while ultra-processed foods (UPFs) are distinguished by industrial ingredients, requiring specialized tools and processing techniques, and hyper-palatability related to color, flavor, and packaging.8 Examples of UPFs include mass-produced breads found at grocery stores, prepackaged snacks and meals, and hydrogenated oils. UPF consumption is associated with higher risk for negative cardiometabolic outcomes, common mental disorders, and all-cause mortality.9 To date, only a study by Powell et al has used the NOVA classification system in a veteran population, and it was limited to a comparison of the price of UPFs and veteran body mass index (BMI).10 Therefore, it remains unknown what percentage of total energy intake (TEI) comes from UPFs in the diets of veterans.
This study sought to quantify the proportion of TEI from UPFs among a sample of patients from the VA Phoenix Health Care System (VAPHCS). Results from a 2021 global meta-analysis reveal that the US and United Kingdom have the highest intakes of UPFs in the world.11 Specifically, within the US, 15 studies with 234,890 participants reveal that the majority of TEI (about 55%) comes from UPFs.11 We hypothesized that this veteran sample would have a higher proportion of TEI from UPFs, possibly due to a higher prevalence of poverty and food insecurity among veterans compared with nonveterans.1 If the percentage of TEI coming from UPF is higher or even similar to nonveterans, further efforts to increase veterans’ use of the available nutritional services would be warranted to minimize nutrition-related disease among veterans.
Methods
This is a cross-sectional, secondary data analysis of baseline 3-day food records collected from 2017 to 2020 from 92 patients recruited at VAPHCS to participate in a whole-food plant-based diet study.12 The original study was reviewed and approved by the VAPHCS Institutional Review Board (1593830). Recruitment methods included clinician recommendation, a recorded advertisement played while phone calls were on hold, and flyers distributed throughout VAPHCS. Patients were included if they were aged 18 to 90 years, had a BMI 25.1 to 39.9, had a diagnosis of nutrition-related chronic disease (hypertension, diabetes, or hyperlipidemia), an interest and desire to make a lifestyle change, active telephone contact information (either landline or cell phone), no contraindication to be on a whole-food plant-based diet, access to transportation and a functioning kitchen, ability to prepare meals independently, access to a computer or tablet with internet access, and a digital camera or smartphone. Exclusion criteria included significant unplanned weight loss within 6 months, uncontrolled insulin-dependent diabetes with a current hemoglobin A1c > 9%, pregnancy/lactation, taking prescribed weight loss medication, currently following a diet (eg, plant-based diet, vegan, or medical weight loss program diet), celiac disease diagnosed within 6 months, end-stage hepatic disease or renal disease requiring dialysis, active cancer or receiving chemotherapy or radiation therapy, active alcohol or substance use disorder, history of eating disorders, fasting triglyceride level > 350 mg/dL, any psychological issues that prevent adherence, inability to speak English, limited mobility, and homeless or in housing with limited kitchen access. A baseline 3-day food record was collected from the participants and used in this secondary analysis.
Diet Analysis
Food records were analyzed using Esha Research Food Processor 4.0 to identify calorie and macronutrient information. To limit bias, food items were coded independently by 2 researchers into 4 food processing groups determined by the NOVA classification: MPF, CPF, PRF, and UPF.8 When possible, specific ingredient information was collected using internet searches for brand product websites. Initial coding had an 89% agreement rate for food item coding between the 2 researchers. As coding was done in duplicate, a third researcher resolved disagreements. The number of food items for each processing group was determined and the mean (SD) percentage of TEI for each NOVA group was provided across participants. A 1-way analysis of variance and Tukey Multiple Comparisons Test were used to determine significance between groups with an α = .05 using Prism V9.
Results
Of the 92 participants in the original study, only 79 met inclusion criteria and had baseline diet data. The 79 veterans had a mean (SD) age of 61 (13) years and 59 (75%) were male (Table 1). Mean (SD) TEI was 1921 (815) kcal. The mean (SD) percentage of calories from carbohydrate, fat, and protein were 46% (21%), 39% (20%), and 16% (6%), respectively (Table 2).


A mean (SD) of 36 (12) food items were analyzed from the 3-day food records. The majority of food items were UPFs (56%), 33% were MPFs, 8% were PRFs, and 3% were CPFs. In total, 75% of TEI came from UPFs (P < .001); only 14% of TEI came from minimally processed foods (Figure).

Discussion
To our knowledge, this is the first analysis of UPF consumption among US veterans. TEIs coming from UPFs appear to be about 20% higher among veterans compared to nonveterans: 75% vs 55%.11 Coupled with high UPF consumption, MPFs (14%) and PRFs (9%) represent smaller sources of TEI among surveyed veterans. Top caloric sources of UPFs in the US include sandwiches (including burgers), sweet bakery products, savory snacks, pizza, sweetened beverages, and breads, rolls, and tortillas, and likely reflect the major sources of UPFs in the veteran diet.13 As the statistical comparison between the veteran data and nonveteran data is not feasible in the present study, a future study with a much larger sample size would be needed for a direct comparison.
While the exact cause of higher UPF consumption among sampled veterans remains unknown and likely multifactorial (eg, cost, food insecurity, access, cooking skills, nutrition knowledge), veterans can receive a consult or self-refer to a registered dietitian nutritionist (RDN) for nutrition education. Counseling has been shown to be an effective way to improve diet quality and increase daily fruit and vegetable intake.14 High consumption of UPFs, which are generally energy-dense and nutrient-poor, contributes to the low diet quality observed in veterans, and future research examining the relationship between UPF intake and overall diet quality among veterans is warranted.2,15 As nutrition knowledge is associated with higher diet quality among veterans, increased use of nutrition services (ie, nutrition education or food supplement programs) has the potential to influence consumption of MPFs and decrease consumption of UPFs.16 Subsequently, UPF-targeted interventions developed by VA RDNs hold the promise to decrease consumption of UPFs and increase intake of MPFs and PRFs.
Veterans have a high prevalence of diabetes, hypertension, and obesity.9 The high UPF intake observed in this sample of veterans may increase the risk for these chronic diseases and overall mortality. The high percentage of TEI from UPFs among veterans is also of concern not only due to potential negative health outcomes, but also associated costs of treating veterans with multimorbidities.17 Targeting UPF intake via nutritional education may promote health and decrease the financial burden needed to support the health of veterans.
Improving veteran health and well-being, including enhancing health care accessibility in underserved areas, are pivotal objectives of the VA strategic plan for 2026 to 2030. Public policy aims to tackle food insecurity within the veteran population during the first 5 years of civilian life.18 In alignment with the White House Strategy on Hunger, Nutrition, and Health, VA established a Food Security Office (FSO) in 2023. The FSO mission is to use an interdisciplinary approach to provide resources to ensure veteran food security and create an environment where all veterans are food and nutrition secure.
Limitations
This study has several limitations. As the Food Processor software database does not include all brand items, similar brands were used to mirror the nutrient profile. While food records are common among veteran diet studies, accuracy may be reduced due to self-reporting bias.19 Different interpretation of the NOVA classification designation for various food items is possible, however, 89% of foods were coded the same by the research team which suggests high accuracy in food coding. Specific ingredient information was not collected from the 3-day food records; thus, these records were not produced in such a way to improve the accuracy of the NOVA classification designation. This study was limited by its small sample size (N = 79); although, this analysis is larger than other studies of UPF consumption in the US.20,21 In addition, the generalizability of this study is limited as this population sample was from a single VA hospital and may not reflect the overall veteran population. Participants in this study were recruited only from those receiving VA care, thus their diet quality may not represent the quality consumed by veterans not participating in VA services. Further research on UPF consumption among veterans is warranted with a larger, more representative study sample size.
Conclusions
As this is the highest observed UPF intake documented in the US, these results should be of concern for the VA and its RDNs. More research is needed to better understand why UPF consumption is so high among veterans, what barriers veterans face to decreasing UPF consumption, and what intervention(s) veterans would welcome to improve their diet quality. Presently, veterans are provided with access to a variety of effective nutrition education and counseling options and should be encouraged to use these services. VA RDNs should be aware of the high intake of UPFs in the veteran population and familiarize themselves with education and counseling strategies that promote behavior change to replace UPFs with more nutrient-dense foods choices.
Roughly 8.6% of the 17.4 million US veterans live in poverty. About 11.1% are considered food insecure (ie, unable to acquire adequate food for ≥1 household members), with another 5.3% considered very food insecure (ie, eating patterns of ≥1 household members were disrupted and their food intake was reduced at least some time during the year). Compared with nonveterans, veterans are 7.4% more likely to be food insecure.1 This high prevalence of food insecurity and poverty has a negative impact on veteran diets.
Veterans’ diets contained more added sugars and solid fats and scored lower compared with nonveterans when assessed for diet quality with the Healthy Eating Index.2 Veterans have a higher prevalence of diet-related chronic disease, including diabetes, hypertension, and obesity compared with the nonveterans.3-5 Given the critical role of diet in health and disease risk, enhancing diet quality among veterans has garnered significant attention and calls to action.2,6,7 While there are many factors that contribute to diet, any veteran can receive a consultation or self-refer to receive nutrition counseling effective for improving diet quality, within the US Department of Veterans Affairs (VA).
The NOVA food classification system describes diet quality by categorizing food items by processing methods and ingredients into 4 food groups.8 The first is unprocessed and minimally processed items (MPFs) such as fresh fruits, vegetables, and meats. MPFs consist of whole foods which can also be minimally processed (eg, chopping, drying, grinding, heating, chilling). Culinary processed foods (CPFs) are processed foods for cooking (eg, salt, butter, and vinegar) and are typically eaten in small quantities along with MPFs. Processed foods (PRFs) include canned and smoked foods, while ultra-processed foods (UPFs) are distinguished by industrial ingredients, requiring specialized tools and processing techniques, and hyper-palatability related to color, flavor, and packaging.8 Examples of UPFs include mass-produced breads found at grocery stores, prepackaged snacks and meals, and hydrogenated oils. UPF consumption is associated with higher risk for negative cardiometabolic outcomes, common mental disorders, and all-cause mortality.9 To date, only a study by Powell et al has used the NOVA classification system in a veteran population, and it was limited to a comparison of the price of UPFs and veteran body mass index (BMI).10 Therefore, it remains unknown what percentage of total energy intake (TEI) comes from UPFs in the diets of veterans.
This study sought to quantify the proportion of TEI from UPFs among a sample of patients from the VA Phoenix Health Care System (VAPHCS). Results from a 2021 global meta-analysis reveal that the US and United Kingdom have the highest intakes of UPFs in the world.11 Specifically, within the US, 15 studies with 234,890 participants reveal that the majority of TEI (about 55%) comes from UPFs.11 We hypothesized that this veteran sample would have a higher proportion of TEI from UPFs, possibly due to a higher prevalence of poverty and food insecurity among veterans compared with nonveterans.1 If the percentage of TEI coming from UPF is higher or even similar to nonveterans, further efforts to increase veterans’ use of the available nutritional services would be warranted to minimize nutrition-related disease among veterans.
Methods
This is a cross-sectional, secondary data analysis of baseline 3-day food records collected from 2017 to 2020 from 92 patients recruited at VAPHCS to participate in a whole-food plant-based diet study.12 The original study was reviewed and approved by the VAPHCS Institutional Review Board (1593830). Recruitment methods included clinician recommendation, a recorded advertisement played while phone calls were on hold, and flyers distributed throughout VAPHCS. Patients were included if they were aged 18 to 90 years, had a BMI 25.1 to 39.9, had a diagnosis of nutrition-related chronic disease (hypertension, diabetes, or hyperlipidemia), an interest and desire to make a lifestyle change, active telephone contact information (either landline or cell phone), no contraindication to be on a whole-food plant-based diet, access to transportation and a functioning kitchen, ability to prepare meals independently, access to a computer or tablet with internet access, and a digital camera or smartphone. Exclusion criteria included significant unplanned weight loss within 6 months, uncontrolled insulin-dependent diabetes with a current hemoglobin A1c > 9%, pregnancy/lactation, taking prescribed weight loss medication, currently following a diet (eg, plant-based diet, vegan, or medical weight loss program diet), celiac disease diagnosed within 6 months, end-stage hepatic disease or renal disease requiring dialysis, active cancer or receiving chemotherapy or radiation therapy, active alcohol or substance use disorder, history of eating disorders, fasting triglyceride level > 350 mg/dL, any psychological issues that prevent adherence, inability to speak English, limited mobility, and homeless or in housing with limited kitchen access. A baseline 3-day food record was collected from the participants and used in this secondary analysis.
Diet Analysis
Food records were analyzed using Esha Research Food Processor 4.0 to identify calorie and macronutrient information. To limit bias, food items were coded independently by 2 researchers into 4 food processing groups determined by the NOVA classification: MPF, CPF, PRF, and UPF.8 When possible, specific ingredient information was collected using internet searches for brand product websites. Initial coding had an 89% agreement rate for food item coding between the 2 researchers. As coding was done in duplicate, a third researcher resolved disagreements. The number of food items for each processing group was determined and the mean (SD) percentage of TEI for each NOVA group was provided across participants. A 1-way analysis of variance and Tukey Multiple Comparisons Test were used to determine significance between groups with an α = .05 using Prism V9.
Results
Of the 92 participants in the original study, only 79 met inclusion criteria and had baseline diet data. The 79 veterans had a mean (SD) age of 61 (13) years and 59 (75%) were male (Table 1). Mean (SD) TEI was 1921 (815) kcal. The mean (SD) percentage of calories from carbohydrate, fat, and protein were 46% (21%), 39% (20%), and 16% (6%), respectively (Table 2).


A mean (SD) of 36 (12) food items were analyzed from the 3-day food records. The majority of food items were UPFs (56%), 33% were MPFs, 8% were PRFs, and 3% were CPFs. In total, 75% of TEI came from UPFs (P < .001); only 14% of TEI came from minimally processed foods (Figure).

Discussion
To our knowledge, this is the first analysis of UPF consumption among US veterans. TEIs coming from UPFs appear to be about 20% higher among veterans compared to nonveterans: 75% vs 55%.11 Coupled with high UPF consumption, MPFs (14%) and PRFs (9%) represent smaller sources of TEI among surveyed veterans. Top caloric sources of UPFs in the US include sandwiches (including burgers), sweet bakery products, savory snacks, pizza, sweetened beverages, and breads, rolls, and tortillas, and likely reflect the major sources of UPFs in the veteran diet.13 As the statistical comparison between the veteran data and nonveteran data is not feasible in the present study, a future study with a much larger sample size would be needed for a direct comparison.
While the exact cause of higher UPF consumption among sampled veterans remains unknown and likely multifactorial (eg, cost, food insecurity, access, cooking skills, nutrition knowledge), veterans can receive a consult or self-refer to a registered dietitian nutritionist (RDN) for nutrition education. Counseling has been shown to be an effective way to improve diet quality and increase daily fruit and vegetable intake.14 High consumption of UPFs, which are generally energy-dense and nutrient-poor, contributes to the low diet quality observed in veterans, and future research examining the relationship between UPF intake and overall diet quality among veterans is warranted.2,15 As nutrition knowledge is associated with higher diet quality among veterans, increased use of nutrition services (ie, nutrition education or food supplement programs) has the potential to influence consumption of MPFs and decrease consumption of UPFs.16 Subsequently, UPF-targeted interventions developed by VA RDNs hold the promise to decrease consumption of UPFs and increase intake of MPFs and PRFs.
Veterans have a high prevalence of diabetes, hypertension, and obesity.9 The high UPF intake observed in this sample of veterans may increase the risk for these chronic diseases and overall mortality. The high percentage of TEI from UPFs among veterans is also of concern not only due to potential negative health outcomes, but also associated costs of treating veterans with multimorbidities.17 Targeting UPF intake via nutritional education may promote health and decrease the financial burden needed to support the health of veterans.
Improving veteran health and well-being, including enhancing health care accessibility in underserved areas, are pivotal objectives of the VA strategic plan for 2026 to 2030. Public policy aims to tackle food insecurity within the veteran population during the first 5 years of civilian life.18 In alignment with the White House Strategy on Hunger, Nutrition, and Health, VA established a Food Security Office (FSO) in 2023. The FSO mission is to use an interdisciplinary approach to provide resources to ensure veteran food security and create an environment where all veterans are food and nutrition secure.
Limitations
This study has several limitations. As the Food Processor software database does not include all brand items, similar brands were used to mirror the nutrient profile. While food records are common among veteran diet studies, accuracy may be reduced due to self-reporting bias.19 Different interpretation of the NOVA classification designation for various food items is possible, however, 89% of foods were coded the same by the research team which suggests high accuracy in food coding. Specific ingredient information was not collected from the 3-day food records; thus, these records were not produced in such a way to improve the accuracy of the NOVA classification designation. This study was limited by its small sample size (N = 79); although, this analysis is larger than other studies of UPF consumption in the US.20,21 In addition, the generalizability of this study is limited as this population sample was from a single VA hospital and may not reflect the overall veteran population. Participants in this study were recruited only from those receiving VA care, thus their diet quality may not represent the quality consumed by veterans not participating in VA services. Further research on UPF consumption among veterans is warranted with a larger, more representative study sample size.
Conclusions
As this is the highest observed UPF intake documented in the US, these results should be of concern for the VA and its RDNs. More research is needed to better understand why UPF consumption is so high among veterans, what barriers veterans face to decreasing UPF consumption, and what intervention(s) veterans would welcome to improve their diet quality. Presently, veterans are provided with access to a variety of effective nutrition education and counseling options and should be encouraged to use these services. VA RDNs should be aware of the high intake of UPFs in the veteran population and familiarize themselves with education and counseling strategies that promote behavior change to replace UPFs with more nutrient-dense foods choices.
- Rabbitt MP, Smith MD. Food insecurity among workingage veterans. US Dept of Agriculture, Economic Research Service; 2021. Accessed January 26, 2026. https://www.ers.usda.gov/publications/pub-details/?pubid=101268
- Dong D, Stewart H, Carlson AC. An examination of veterans’ diet quality. US Dept of Agriculture, Economic Research Service; 2019. Accessed January 26, 2026. https:// www.ers.usda.gov/publications/pub-details/?pubid=95608
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of adult overweight and obesity. 2020. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/obesity/
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. 2023. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/diabetes/
- Boersma P, Cohen R, Zelaya C, et al. Multiple chronic conditions among veterans and nonveterans: United States, 2015–2018. Natl Health Stat Rep. 2021. doi:10.15620/cdc:101659
- Hoerster KD, Wilson S, Nelson KM, et al. Diet quality is associated with mental health, social support, and neighborhood factors among veterans. Eat Behav. 2016;23:168- 173. doi:10.1016/j.eatbeh.2016.10.003
- Becerra MB, Hassija CM, Becerra BJ. Food insecurity is associated with unhealthy dietary practices among US veterans in California. Public Health Nutr. 2017;20:2569-2576. doi:10.1017/S1368980016002147
- Monteiro CA, Cannon G, Levy RB, et al. Ultra-processed foods: what they are and how to identify them. Public Health Nutr. 2019;22:936-941. doi:10.1017/S1368980018003762
- Lane MM, Gamage E, Du S, et al. Ultra-processed food exposure and adverse health outcomes: umbrella review of epidemiological meta-analyses. BMJ. February 2024:e077310. doi:10.1136/bmj-2023-077310
- Powell LM, Jones K, Duran AC, et al. The price of ultra-processed foods and beverages and adult body weight: Evidence from U.S. veterans. Econ Hum Biol. 2019;34:39- 48. doi:10.1016/j.ehb.2019.05.006
- Marino M, Puppo F, Del Bo’ C, et al. A systematic review of worldwide consumption of ultra-processed foods: findings and criticisms. Nutrients. 2021;13. doi:10.3390/nu13082778
- Parrington D, Kurtz J, Fawcett J, et al. Pilot study on the effects of a whole-food, plant-strong diet on cardiovascular risk factors in veterans: part 3. Curr Dev Nutr. 2022;6:385. doi:10.1093/cdn/nzac054.040
- Williams AM, Couch CA, Emmerich SD, et al. Ultra-processed Food Consumption in Youth and Adults: United States, August 2021-August 2023. NCHS Data Brief. 2025. doi:10.15620/cdc/174612
- Serra MC, Addison O, Giffuni J, et al. Changes in self-reported fruit and vegetable intake following nutritional modification in high risk older veterans. J Nutr Gerontol Geriatr. 2021;40:1-8. doi:10.1080/21551197.2020.1863892
- Gupta S, Hawk T, Aggarwal A, et al. Characterizing ultra-processed foods by energy density, nutrient density, and cost. Front Nutr. 2019;6:1-9. doi:10.3389/fnut.2019.00070
- Robinson LA, Colin CR, Smith KS, et al. Diet quality is associated with nutrition knowledge and physical activity in the US military veterans enrolled in university programmes. BMJ Mil Heal. 2023:e002525. doi:10.1136/military-2023-002525
- Yoon J, Zulman D, Scott JY, et al. Costs associated with multimorbidity among VA patients. Med Care. 2014;52:S31-6. doi:10.1097/MLR.0000000000000061
- End Veteran Hunger Act of 2022, H.R. 8852, 117th Cong. (2022). Accessed January 26, 2026. https://www.congress.gov/bill/117th-congress/house-bill/8852.
- Collins RA, Baker B, Coyle DH, et al. Dietary assessment methods in military and veteran populations: a scoping review. Nutrients. 2020;12:1-21. doi:10.3390/nu12030769
- Smiljanec K, Mbakwe AU, Ramos-Gonzalez M, et al. Associations of ultra-processed and unprocessed/minimally processed food consumption with peripheral and central hemodynamics, and arterial stiffness in young healthy adults. Nutrients. 2020;12. doi:10.3390/nu12113229
- Rohatgi KW, Tinius RA, Cade WT, et al. Relationships between consumption of ultra-processed foods, gestational weight gain and neonatal outcomes in a sample of US pregnant women. PeerJ. 2017;5:e4091. doi:10.7717/peerj.4091
- Rabbitt MP, Smith MD. Food insecurity among workingage veterans. US Dept of Agriculture, Economic Research Service; 2021. Accessed January 26, 2026. https://www.ers.usda.gov/publications/pub-details/?pubid=101268
- Dong D, Stewart H, Carlson AC. An examination of veterans’ diet quality. US Dept of Agriculture, Economic Research Service; 2019. Accessed January 26, 2026. https:// www.ers.usda.gov/publications/pub-details/?pubid=95608
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of adult overweight and obesity. 2020. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/obesity/
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. 2023. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/diabetes/
- Boersma P, Cohen R, Zelaya C, et al. Multiple chronic conditions among veterans and nonveterans: United States, 2015–2018. Natl Health Stat Rep. 2021. doi:10.15620/cdc:101659
- Hoerster KD, Wilson S, Nelson KM, et al. Diet quality is associated with mental health, social support, and neighborhood factors among veterans. Eat Behav. 2016;23:168- 173. doi:10.1016/j.eatbeh.2016.10.003
- Becerra MB, Hassija CM, Becerra BJ. Food insecurity is associated with unhealthy dietary practices among US veterans in California. Public Health Nutr. 2017;20:2569-2576. doi:10.1017/S1368980016002147
- Monteiro CA, Cannon G, Levy RB, et al. Ultra-processed foods: what they are and how to identify them. Public Health Nutr. 2019;22:936-941. doi:10.1017/S1368980018003762
- Lane MM, Gamage E, Du S, et al. Ultra-processed food exposure and adverse health outcomes: umbrella review of epidemiological meta-analyses. BMJ. February 2024:e077310. doi:10.1136/bmj-2023-077310
- Powell LM, Jones K, Duran AC, et al. The price of ultra-processed foods and beverages and adult body weight: Evidence from U.S. veterans. Econ Hum Biol. 2019;34:39- 48. doi:10.1016/j.ehb.2019.05.006
- Marino M, Puppo F, Del Bo’ C, et al. A systematic review of worldwide consumption of ultra-processed foods: findings and criticisms. Nutrients. 2021;13. doi:10.3390/nu13082778
- Parrington D, Kurtz J, Fawcett J, et al. Pilot study on the effects of a whole-food, plant-strong diet on cardiovascular risk factors in veterans: part 3. Curr Dev Nutr. 2022;6:385. doi:10.1093/cdn/nzac054.040
- Williams AM, Couch CA, Emmerich SD, et al. Ultra-processed Food Consumption in Youth and Adults: United States, August 2021-August 2023. NCHS Data Brief. 2025. doi:10.15620/cdc/174612
- Serra MC, Addison O, Giffuni J, et al. Changes in self-reported fruit and vegetable intake following nutritional modification in high risk older veterans. J Nutr Gerontol Geriatr. 2021;40:1-8. doi:10.1080/21551197.2020.1863892
- Gupta S, Hawk T, Aggarwal A, et al. Characterizing ultra-processed foods by energy density, nutrient density, and cost. Front Nutr. 2019;6:1-9. doi:10.3389/fnut.2019.00070
- Robinson LA, Colin CR, Smith KS, et al. Diet quality is associated with nutrition knowledge and physical activity in the US military veterans enrolled in university programmes. BMJ Mil Heal. 2023:e002525. doi:10.1136/military-2023-002525
- Yoon J, Zulman D, Scott JY, et al. Costs associated with multimorbidity among VA patients. Med Care. 2014;52:S31-6. doi:10.1097/MLR.0000000000000061
- End Veteran Hunger Act of 2022, H.R. 8852, 117th Cong. (2022). Accessed January 26, 2026. https://www.congress.gov/bill/117th-congress/house-bill/8852.
- Collins RA, Baker B, Coyle DH, et al. Dietary assessment methods in military and veteran populations: a scoping review. Nutrients. 2020;12:1-21. doi:10.3390/nu12030769
- Smiljanec K, Mbakwe AU, Ramos-Gonzalez M, et al. Associations of ultra-processed and unprocessed/minimally processed food consumption with peripheral and central hemodynamics, and arterial stiffness in young healthy adults. Nutrients. 2020;12. doi:10.3390/nu12113229
- Rohatgi KW, Tinius RA, Cade WT, et al. Relationships between consumption of ultra-processed foods, gestational weight gain and neonatal outcomes in a sample of US pregnant women. PeerJ. 2017;5:e4091. doi:10.7717/peerj.4091
Seventy-Five Percent of Total Energy Intake Comes From Ultra-Processed Foods Among a Sample of Veterans With Overweight and Obesity: An Exploratory Analysis of Three-Day Food Records
Seventy-Five Percent of Total Energy Intake Comes From Ultra-Processed Foods Among a Sample of Veterans With Overweight and Obesity: An Exploratory Analysis of Three-Day Food Records
New Drug Eases Side Effects of Weight-Loss Meds
, based on data from a phase 2 trial presented at the Obesity Society’s Obesity Week 2025 in Atlanta.
Previous research published in JAMA Network Open showed a nearly 65% discontinuation rate for three GLP-1s (liraglutide, semaglutide, or tirzepatide) among adults with overweight or obesity and without type 2 diabetes. Gastrointestinal (GI) side effects topped the list of reasons for dropping the medications.
Given the impact of nausea and vomiting on discontinuation, there is an unmet need for therapies to manage GI symptoms, said Kimberley Cummings, PhD, of Neurogastrx, Inc., in her presentation.
In the new study, Cummings and colleagues randomly assigned 90 adults aged 18-55 years with overweight or obesity (defined as a BMI ranging from 22.0 to 35.0) to receive a single subcutaneous dose of semaglutide (0.5 mg) plus 5 days of NG101 at 20 mg twice daily, or a placebo.
NG101 is a peripherally acting D2 antagonist designed to reduce nausea and vomiting associated with GLP-1 use, Cummings said. NG101 targets the nausea center of the brain but is peripherally restricted to prevent central nervous system side effects, she explained.
Compared with placebo, NG101 significantly reduced the incidence of nausea and vomiting by 40% and 67%, respectively. Use of NG101 also was associated with a significant reduction in the duration of nausea and vomiting; GI events lasting longer than 1 day were reported in 22% and 51% of the NG101 patients and placebo patients, respectively.
In addition, participants who received NG101 reported a 70% decrease in nausea severity from baseline.
Overall, patients in the NG101 group also reported significantly fewer adverse events than those in the placebo group (74 vs 135), suggesting an improved safety profile when semaglutide is administered in conjunction with NG101, the researchers noted. No serious adverse events related to the study drug were reported in either group.
The findings were limited by several factors including the relatively small sample size. Additional research is needed with other GLP-1 agonists in larger populations with longer follow-up periods, Cummings said. However, the results suggest that NG101 was safe and effectively improved side effects associated with GLP-1 agonists.
“We know there are receptors for GLP-1 in the area postrema (nausea center of the brain), and that NG101 works on this area to reduce nausea and vomiting, so the study findings were not unexpected,” said Jim O’Mara, president and CEO of Neurogastrx, in an interview.
The study was a single-dose study designed to show proof of concept, and future studies would involve treating patients going through the recommended titration schedule for their GLP-1s, O’Mara said. However, NG101 offers an opportunity to keep more patients on GLP-1 therapy and help them reach their long-term therapeutic goals, he said.
Decrease Side Effects for Weight-Loss Success
“GI side effects are often the rate-limiting step in implementing an effective medication that patients want to take but may not be able to tolerate,” Sean Wharton, MD, PharmD, medical director of the Wharton Medical Clinic for Weight and Diabetes Management, Burlington, Ontario, Canada, said in an interview. “If we can decrease side effects, these medications could improve patients’ lives,” said Wharton, who was not involved in the study.
The improvement after a single dose of NG101 in patients receiving a single dose of semaglutide was impressive and in keeping with the mechanism of the drug action, said Wharton. “I was not surprised by the result but pleased that this single dose was shown to reduce the overall incidence of nausea and vomiting, the duration of nausea, the severity of nausea as rated by the study participants compared to placebo,” he said.
Ultimately, the clinical implications for NG101 are improved patient tolerance for GLP-1s and the ability to titrate and stay on them long term, incurring greater cardiometabolic benefit, Wharton told GI & Hepatology News.
The current trial was limited to GLP1-1s on the market; newer medications may have fewer side effects, Wharton noted. “In clinical practice, patients often decrease the medication or titrate slower, and this could be the comparator,” he added.
The study was funded by Neurogastrx.
Wharton disclosed serving as a consultant for Neurogastrx but not as an investigator on the current study. He also reported having disclosed research on various GLP-1 medications.
A version of this article appeared on Medscape.com.
, based on data from a phase 2 trial presented at the Obesity Society’s Obesity Week 2025 in Atlanta.
Previous research published in JAMA Network Open showed a nearly 65% discontinuation rate for three GLP-1s (liraglutide, semaglutide, or tirzepatide) among adults with overweight or obesity and without type 2 diabetes. Gastrointestinal (GI) side effects topped the list of reasons for dropping the medications.
Given the impact of nausea and vomiting on discontinuation, there is an unmet need for therapies to manage GI symptoms, said Kimberley Cummings, PhD, of Neurogastrx, Inc., in her presentation.
In the new study, Cummings and colleagues randomly assigned 90 adults aged 18-55 years with overweight or obesity (defined as a BMI ranging from 22.0 to 35.0) to receive a single subcutaneous dose of semaglutide (0.5 mg) plus 5 days of NG101 at 20 mg twice daily, or a placebo.
NG101 is a peripherally acting D2 antagonist designed to reduce nausea and vomiting associated with GLP-1 use, Cummings said. NG101 targets the nausea center of the brain but is peripherally restricted to prevent central nervous system side effects, she explained.
Compared with placebo, NG101 significantly reduced the incidence of nausea and vomiting by 40% and 67%, respectively. Use of NG101 also was associated with a significant reduction in the duration of nausea and vomiting; GI events lasting longer than 1 day were reported in 22% and 51% of the NG101 patients and placebo patients, respectively.
In addition, participants who received NG101 reported a 70% decrease in nausea severity from baseline.
Overall, patients in the NG101 group also reported significantly fewer adverse events than those in the placebo group (74 vs 135), suggesting an improved safety profile when semaglutide is administered in conjunction with NG101, the researchers noted. No serious adverse events related to the study drug were reported in either group.
The findings were limited by several factors including the relatively small sample size. Additional research is needed with other GLP-1 agonists in larger populations with longer follow-up periods, Cummings said. However, the results suggest that NG101 was safe and effectively improved side effects associated with GLP-1 agonists.
“We know there are receptors for GLP-1 in the area postrema (nausea center of the brain), and that NG101 works on this area to reduce nausea and vomiting, so the study findings were not unexpected,” said Jim O’Mara, president and CEO of Neurogastrx, in an interview.
The study was a single-dose study designed to show proof of concept, and future studies would involve treating patients going through the recommended titration schedule for their GLP-1s, O’Mara said. However, NG101 offers an opportunity to keep more patients on GLP-1 therapy and help them reach their long-term therapeutic goals, he said.
Decrease Side Effects for Weight-Loss Success
“GI side effects are often the rate-limiting step in implementing an effective medication that patients want to take but may not be able to tolerate,” Sean Wharton, MD, PharmD, medical director of the Wharton Medical Clinic for Weight and Diabetes Management, Burlington, Ontario, Canada, said in an interview. “If we can decrease side effects, these medications could improve patients’ lives,” said Wharton, who was not involved in the study.
The improvement after a single dose of NG101 in patients receiving a single dose of semaglutide was impressive and in keeping with the mechanism of the drug action, said Wharton. “I was not surprised by the result but pleased that this single dose was shown to reduce the overall incidence of nausea and vomiting, the duration of nausea, the severity of nausea as rated by the study participants compared to placebo,” he said.
Ultimately, the clinical implications for NG101 are improved patient tolerance for GLP-1s and the ability to titrate and stay on them long term, incurring greater cardiometabolic benefit, Wharton told GI & Hepatology News.
The current trial was limited to GLP1-1s on the market; newer medications may have fewer side effects, Wharton noted. “In clinical practice, patients often decrease the medication or titrate slower, and this could be the comparator,” he added.
The study was funded by Neurogastrx.
Wharton disclosed serving as a consultant for Neurogastrx but not as an investigator on the current study. He also reported having disclosed research on various GLP-1 medications.
A version of this article appeared on Medscape.com.
, based on data from a phase 2 trial presented at the Obesity Society’s Obesity Week 2025 in Atlanta.
Previous research published in JAMA Network Open showed a nearly 65% discontinuation rate for three GLP-1s (liraglutide, semaglutide, or tirzepatide) among adults with overweight or obesity and without type 2 diabetes. Gastrointestinal (GI) side effects topped the list of reasons for dropping the medications.
Given the impact of nausea and vomiting on discontinuation, there is an unmet need for therapies to manage GI symptoms, said Kimberley Cummings, PhD, of Neurogastrx, Inc., in her presentation.
In the new study, Cummings and colleagues randomly assigned 90 adults aged 18-55 years with overweight or obesity (defined as a BMI ranging from 22.0 to 35.0) to receive a single subcutaneous dose of semaglutide (0.5 mg) plus 5 days of NG101 at 20 mg twice daily, or a placebo.
NG101 is a peripherally acting D2 antagonist designed to reduce nausea and vomiting associated with GLP-1 use, Cummings said. NG101 targets the nausea center of the brain but is peripherally restricted to prevent central nervous system side effects, she explained.
Compared with placebo, NG101 significantly reduced the incidence of nausea and vomiting by 40% and 67%, respectively. Use of NG101 also was associated with a significant reduction in the duration of nausea and vomiting; GI events lasting longer than 1 day were reported in 22% and 51% of the NG101 patients and placebo patients, respectively.
In addition, participants who received NG101 reported a 70% decrease in nausea severity from baseline.
Overall, patients in the NG101 group also reported significantly fewer adverse events than those in the placebo group (74 vs 135), suggesting an improved safety profile when semaglutide is administered in conjunction with NG101, the researchers noted. No serious adverse events related to the study drug were reported in either group.
The findings were limited by several factors including the relatively small sample size. Additional research is needed with other GLP-1 agonists in larger populations with longer follow-up periods, Cummings said. However, the results suggest that NG101 was safe and effectively improved side effects associated with GLP-1 agonists.
“We know there are receptors for GLP-1 in the area postrema (nausea center of the brain), and that NG101 works on this area to reduce nausea and vomiting, so the study findings were not unexpected,” said Jim O’Mara, president and CEO of Neurogastrx, in an interview.
The study was a single-dose study designed to show proof of concept, and future studies would involve treating patients going through the recommended titration schedule for their GLP-1s, O’Mara said. However, NG101 offers an opportunity to keep more patients on GLP-1 therapy and help them reach their long-term therapeutic goals, he said.
Decrease Side Effects for Weight-Loss Success
“GI side effects are often the rate-limiting step in implementing an effective medication that patients want to take but may not be able to tolerate,” Sean Wharton, MD, PharmD, medical director of the Wharton Medical Clinic for Weight and Diabetes Management, Burlington, Ontario, Canada, said in an interview. “If we can decrease side effects, these medications could improve patients’ lives,” said Wharton, who was not involved in the study.
The improvement after a single dose of NG101 in patients receiving a single dose of semaglutide was impressive and in keeping with the mechanism of the drug action, said Wharton. “I was not surprised by the result but pleased that this single dose was shown to reduce the overall incidence of nausea and vomiting, the duration of nausea, the severity of nausea as rated by the study participants compared to placebo,” he said.
Ultimately, the clinical implications for NG101 are improved patient tolerance for GLP-1s and the ability to titrate and stay on them long term, incurring greater cardiometabolic benefit, Wharton told GI & Hepatology News.
The current trial was limited to GLP1-1s on the market; newer medications may have fewer side effects, Wharton noted. “In clinical practice, patients often decrease the medication or titrate slower, and this could be the comparator,” he added.
The study was funded by Neurogastrx.
Wharton disclosed serving as a consultant for Neurogastrx but not as an investigator on the current study. He also reported having disclosed research on various GLP-1 medications.
A version of this article appeared on Medscape.com.
Duodenal Mucosal Resurfacing Curbs Weight Gain Post-GLP-1
, initial results of the open-label, multistage REMAIN-1 trial showed.
In addition, “the procedure was well tolerated, with only minor, transient TEAEs [treatment-emergent adverse events] consistent with routine upper endoscopy,” said Shailendra Singh, MD, of West Virginia University in Morgantown, West Virginia, who presented the findings at The Obesity Society’s Obesity Week 2025 meeting in Atlanta.
DMR uses hydrothermal ablation to treat the duodenal mucosa, which may be dysfunctional in both obesity and impaired glucose tolerance. A previous pooled clinical trial analysis of more than 100 patients with type 2 diabetes demonstrated that DMR helped patients maintain body weight loss up to 48 weeks post-procedure.
Metabolic therapeutics company Fractyl Health, Burlington, Massachusetts, developed the procedure, called Revita, and is sponsoring the current study. The trial’s aim is to determine the effect of DMR on weight-loss maintenance in patients with ≥ 15% total body weight loss using a GLP-1 RA in both an open-label arm and a prospective, randomized, double-blind, sham-controlled multicenter arm.
‘Encouraging Preliminary Findings’
The open-label arm included 15 DMR-treated participants (mean age, 49 years, 87% female ), all of whom had taken tirzepatide for a minimum of 5 months and a maximum of 3 years prior to DMR and had lost at least 15% of their total body weight.
Participants had a mean pre-GLP-1 RA weight of 104.8 kg and a mean weight prior to DMR of 79.4 kg, for a mean total body weight loss from the start of GLP-1 RA of 23.8%. Weight loss was heterogeneous and reflective of the real-world patient population taking GLP-1 medications, according to the poster presentation.
Participants discontinued their GLP-1 medication, underwent the DMR procedure, and were followed for 3 months. A total of 12 of 13 patients maintained or lost weight at that point, with 6 of 13 losing additional weight.
Specifically, participants experienced a median of 0.46% weight change (approximately 1 lb) compared with the 5%-6% weight regain (10-15 lb) observed after GLP-1 discontinuation in the literature.
The procedure was well tolerated, with most patients experiencing no TEAEs and none experiencing an event greater than grade 1. Grade 1 events occurred in three patients; 23% were transient in nature, lasting 2-5 days, and were similar to those typically seen with a routine upper endoscopy.
“These encouraging preliminary findings suggest that DMR may safely achieve durable weight maintenance for patients who wish to discontinue GLP-1 RA therapy,” the study authors stated.
Randomization is anticipated in early 2026, with 6-month topline data and a potential premarket approval filing expected in the second half of 2026.
A version of this article appeared on Medscape.com.
, initial results of the open-label, multistage REMAIN-1 trial showed.
In addition, “the procedure was well tolerated, with only minor, transient TEAEs [treatment-emergent adverse events] consistent with routine upper endoscopy,” said Shailendra Singh, MD, of West Virginia University in Morgantown, West Virginia, who presented the findings at The Obesity Society’s Obesity Week 2025 meeting in Atlanta.
DMR uses hydrothermal ablation to treat the duodenal mucosa, which may be dysfunctional in both obesity and impaired glucose tolerance. A previous pooled clinical trial analysis of more than 100 patients with type 2 diabetes demonstrated that DMR helped patients maintain body weight loss up to 48 weeks post-procedure.
Metabolic therapeutics company Fractyl Health, Burlington, Massachusetts, developed the procedure, called Revita, and is sponsoring the current study. The trial’s aim is to determine the effect of DMR on weight-loss maintenance in patients with ≥ 15% total body weight loss using a GLP-1 RA in both an open-label arm and a prospective, randomized, double-blind, sham-controlled multicenter arm.
‘Encouraging Preliminary Findings’
The open-label arm included 15 DMR-treated participants (mean age, 49 years, 87% female ), all of whom had taken tirzepatide for a minimum of 5 months and a maximum of 3 years prior to DMR and had lost at least 15% of their total body weight.
Participants had a mean pre-GLP-1 RA weight of 104.8 kg and a mean weight prior to DMR of 79.4 kg, for a mean total body weight loss from the start of GLP-1 RA of 23.8%. Weight loss was heterogeneous and reflective of the real-world patient population taking GLP-1 medications, according to the poster presentation.
Participants discontinued their GLP-1 medication, underwent the DMR procedure, and were followed for 3 months. A total of 12 of 13 patients maintained or lost weight at that point, with 6 of 13 losing additional weight.
Specifically, participants experienced a median of 0.46% weight change (approximately 1 lb) compared with the 5%-6% weight regain (10-15 lb) observed after GLP-1 discontinuation in the literature.
The procedure was well tolerated, with most patients experiencing no TEAEs and none experiencing an event greater than grade 1. Grade 1 events occurred in three patients; 23% were transient in nature, lasting 2-5 days, and were similar to those typically seen with a routine upper endoscopy.
“These encouraging preliminary findings suggest that DMR may safely achieve durable weight maintenance for patients who wish to discontinue GLP-1 RA therapy,” the study authors stated.
Randomization is anticipated in early 2026, with 6-month topline data and a potential premarket approval filing expected in the second half of 2026.
A version of this article appeared on Medscape.com.
, initial results of the open-label, multistage REMAIN-1 trial showed.
In addition, “the procedure was well tolerated, with only minor, transient TEAEs [treatment-emergent adverse events] consistent with routine upper endoscopy,” said Shailendra Singh, MD, of West Virginia University in Morgantown, West Virginia, who presented the findings at The Obesity Society’s Obesity Week 2025 meeting in Atlanta.
DMR uses hydrothermal ablation to treat the duodenal mucosa, which may be dysfunctional in both obesity and impaired glucose tolerance. A previous pooled clinical trial analysis of more than 100 patients with type 2 diabetes demonstrated that DMR helped patients maintain body weight loss up to 48 weeks post-procedure.
Metabolic therapeutics company Fractyl Health, Burlington, Massachusetts, developed the procedure, called Revita, and is sponsoring the current study. The trial’s aim is to determine the effect of DMR on weight-loss maintenance in patients with ≥ 15% total body weight loss using a GLP-1 RA in both an open-label arm and a prospective, randomized, double-blind, sham-controlled multicenter arm.
‘Encouraging Preliminary Findings’
The open-label arm included 15 DMR-treated participants (mean age, 49 years, 87% female ), all of whom had taken tirzepatide for a minimum of 5 months and a maximum of 3 years prior to DMR and had lost at least 15% of their total body weight.
Participants had a mean pre-GLP-1 RA weight of 104.8 kg and a mean weight prior to DMR of 79.4 kg, for a mean total body weight loss from the start of GLP-1 RA of 23.8%. Weight loss was heterogeneous and reflective of the real-world patient population taking GLP-1 medications, according to the poster presentation.
Participants discontinued their GLP-1 medication, underwent the DMR procedure, and were followed for 3 months. A total of 12 of 13 patients maintained or lost weight at that point, with 6 of 13 losing additional weight.
Specifically, participants experienced a median of 0.46% weight change (approximately 1 lb) compared with the 5%-6% weight regain (10-15 lb) observed after GLP-1 discontinuation in the literature.
The procedure was well tolerated, with most patients experiencing no TEAEs and none experiencing an event greater than grade 1. Grade 1 events occurred in three patients; 23% were transient in nature, lasting 2-5 days, and were similar to those typically seen with a routine upper endoscopy.
“These encouraging preliminary findings suggest that DMR may safely achieve durable weight maintenance for patients who wish to discontinue GLP-1 RA therapy,” the study authors stated.
Randomization is anticipated in early 2026, with 6-month topline data and a potential premarket approval filing expected in the second half of 2026.
A version of this article appeared on Medscape.com.
New Drug Eases Side Effects of Weight-Loss Meds
A new drug currently known as NG101 reduced nausea and vomiting in patients with obesity using GLP-1s by 40% and 67%, respectively, based on data from a phase 2 trial presented at the Obesity Society’s Obesity Week 2025 in Atlanta.
Previous research published in JAMA Network Open showed a nearly 65% discontinuation rate for three GLP-1s (liraglutide, semaglutide, or tirzepatide) among adults with overweight or obesity and without type 2 diabetes. Gastrointestinal (GI) side effects topped the list of reasons for dropping the medications.
Given the impact of nausea and vomiting on discontinuation, there is an unmet need for therapies to manage GI symptoms, said Kimberley Cummings, PhD, of Neurogastrx, Inc., in her presentation.
In the new study, Cummings and colleagues randomly assigned 90 adults aged 18-55 years with overweight or obesity (defined as a BMI ranging from 22.0 to 35.0) to receive a single subcutaneous dose of semaglutide (0.5 mg) plus 5 days of NG101 at 20 mg twice daily, or a placebo.
NG101 is a peripherally acting D2 antagonist designed to reduce nausea and vomiting associated with GLP-1 use, Cummings said. NG101 targets the nausea center of the brain but is peripherally restricted to prevent central nervous system side effects, she explained.
Compared with placebo, NG101 significantly reduced the incidence of nausea and vomiting by 40% and 67%, respectively. Use of NG101 also was associated with a significant reduction in the duration of nausea and vomiting; GI events lasting longer than 1 day were reported in 22% and 51% of the NG101 patients and placebo patients, respectively.
In addition, participants who received NG101 reported a 70% decrease in nausea severity from baseline.
Overall, patients in the NG101 group also reported significantly fewer adverse events than those in the placebo group (74 vs 135), suggesting an improved safety profile when semaglutide is administered in conjunction with NG101, the researchers noted. No serious adverse events related to the study drug were reported in either group.
The findings were limited by several factors including the relatively small sample size. Additional research is needed with other GLP-1 agonists in larger populations with longer follow-up periods, Cummings said. However, the results suggest that NG101 was safe and effectively improved side effects associated with GLP-1 agonists.
“We know there are receptors for GLP-1 in the area postrema (nausea center of the brain), and that NG101 works on this area to reduce nausea and vomiting, so the study findings were not unexpected,” said Jim O’Mara, president and CEO of Neurogastrx, in an interview.
The study was a single-dose study designed to show proof of concept, and future studies would involve treating patients going through the recommended titration schedule for their GLP-1s, O’Mara said. However, NG101 offers an opportunity to keep more patients on GLP-1 therapy and help them reach their long-term therapeutic goals, he said.
Decrease Side Effects for Weight-Loss Success
“GI side effects are often the rate-limiting step in implementing an effective medication that patients want to take but may not be able to tolerate,” said Sean Wharton, MD, PharmD, medical director of the Wharton Medical Clinic for Weight and Diabetes Management, Burlington, Ontario, Canada, in an interview. “If we can decrease side effects, these medications could improve patients’ lives,” said Wharton, who was not involved in the study.
The improvement after a single dose of NG101 in patients receiving a single dose of semaglutide was impressive and in keeping with the mechanism of the drug action, said Wharton. “I was not surprised by the result but pleased that this single dose was shown to reduce the overall incidence of nausea and vomiting, the duration of nausea, the severity of nausea as rated by the study participants compared to placebo,” he said.
Ultimately, the clinical implications for NG101 are improved patient tolerance for GLP-1s and the ability to titrate and stay on them long term, incurring greater cardiometabolic benefit, Wharton told this news organization.
The current trial was limited to GLP1-1s on the market; newer medications may have fewer side effects, Wharton noted. “In clinical practice, patients often decrease the medication or titrate slower, and this could be the comparator,” he added.
The study was funded by Neurogastrx.
Wharton disclosed serving as a consultant for Neurogastrx but not as an investigator on the current study. He also reported having disclosed research on various GLP-1 medications.
A version of this article first appeared on Medscape.com.
A new drug currently known as NG101 reduced nausea and vomiting in patients with obesity using GLP-1s by 40% and 67%, respectively, based on data from a phase 2 trial presented at the Obesity Society’s Obesity Week 2025 in Atlanta.
Previous research published in JAMA Network Open showed a nearly 65% discontinuation rate for three GLP-1s (liraglutide, semaglutide, or tirzepatide) among adults with overweight or obesity and without type 2 diabetes. Gastrointestinal (GI) side effects topped the list of reasons for dropping the medications.
Given the impact of nausea and vomiting on discontinuation, there is an unmet need for therapies to manage GI symptoms, said Kimberley Cummings, PhD, of Neurogastrx, Inc., in her presentation.
In the new study, Cummings and colleagues randomly assigned 90 adults aged 18-55 years with overweight or obesity (defined as a BMI ranging from 22.0 to 35.0) to receive a single subcutaneous dose of semaglutide (0.5 mg) plus 5 days of NG101 at 20 mg twice daily, or a placebo.
NG101 is a peripherally acting D2 antagonist designed to reduce nausea and vomiting associated with GLP-1 use, Cummings said. NG101 targets the nausea center of the brain but is peripherally restricted to prevent central nervous system side effects, she explained.
Compared with placebo, NG101 significantly reduced the incidence of nausea and vomiting by 40% and 67%, respectively. Use of NG101 also was associated with a significant reduction in the duration of nausea and vomiting; GI events lasting longer than 1 day were reported in 22% and 51% of the NG101 patients and placebo patients, respectively.
In addition, participants who received NG101 reported a 70% decrease in nausea severity from baseline.
Overall, patients in the NG101 group also reported significantly fewer adverse events than those in the placebo group (74 vs 135), suggesting an improved safety profile when semaglutide is administered in conjunction with NG101, the researchers noted. No serious adverse events related to the study drug were reported in either group.
The findings were limited by several factors including the relatively small sample size. Additional research is needed with other GLP-1 agonists in larger populations with longer follow-up periods, Cummings said. However, the results suggest that NG101 was safe and effectively improved side effects associated with GLP-1 agonists.
“We know there are receptors for GLP-1 in the area postrema (nausea center of the brain), and that NG101 works on this area to reduce nausea and vomiting, so the study findings were not unexpected,” said Jim O’Mara, president and CEO of Neurogastrx, in an interview.
The study was a single-dose study designed to show proof of concept, and future studies would involve treating patients going through the recommended titration schedule for their GLP-1s, O’Mara said. However, NG101 offers an opportunity to keep more patients on GLP-1 therapy and help them reach their long-term therapeutic goals, he said.
Decrease Side Effects for Weight-Loss Success
“GI side effects are often the rate-limiting step in implementing an effective medication that patients want to take but may not be able to tolerate,” said Sean Wharton, MD, PharmD, medical director of the Wharton Medical Clinic for Weight and Diabetes Management, Burlington, Ontario, Canada, in an interview. “If we can decrease side effects, these medications could improve patients’ lives,” said Wharton, who was not involved in the study.
The improvement after a single dose of NG101 in patients receiving a single dose of semaglutide was impressive and in keeping with the mechanism of the drug action, said Wharton. “I was not surprised by the result but pleased that this single dose was shown to reduce the overall incidence of nausea and vomiting, the duration of nausea, the severity of nausea as rated by the study participants compared to placebo,” he said.
Ultimately, the clinical implications for NG101 are improved patient tolerance for GLP-1s and the ability to titrate and stay on them long term, incurring greater cardiometabolic benefit, Wharton told this news organization.
The current trial was limited to GLP1-1s on the market; newer medications may have fewer side effects, Wharton noted. “In clinical practice, patients often decrease the medication or titrate slower, and this could be the comparator,” he added.
The study was funded by Neurogastrx.
Wharton disclosed serving as a consultant for Neurogastrx but not as an investigator on the current study. He also reported having disclosed research on various GLP-1 medications.
A version of this article first appeared on Medscape.com.
A new drug currently known as NG101 reduced nausea and vomiting in patients with obesity using GLP-1s by 40% and 67%, respectively, based on data from a phase 2 trial presented at the Obesity Society’s Obesity Week 2025 in Atlanta.
Previous research published in JAMA Network Open showed a nearly 65% discontinuation rate for three GLP-1s (liraglutide, semaglutide, or tirzepatide) among adults with overweight or obesity and without type 2 diabetes. Gastrointestinal (GI) side effects topped the list of reasons for dropping the medications.
Given the impact of nausea and vomiting on discontinuation, there is an unmet need for therapies to manage GI symptoms, said Kimberley Cummings, PhD, of Neurogastrx, Inc., in her presentation.
In the new study, Cummings and colleagues randomly assigned 90 adults aged 18-55 years with overweight or obesity (defined as a BMI ranging from 22.0 to 35.0) to receive a single subcutaneous dose of semaglutide (0.5 mg) plus 5 days of NG101 at 20 mg twice daily, or a placebo.
NG101 is a peripherally acting D2 antagonist designed to reduce nausea and vomiting associated with GLP-1 use, Cummings said. NG101 targets the nausea center of the brain but is peripherally restricted to prevent central nervous system side effects, she explained.
Compared with placebo, NG101 significantly reduced the incidence of nausea and vomiting by 40% and 67%, respectively. Use of NG101 also was associated with a significant reduction in the duration of nausea and vomiting; GI events lasting longer than 1 day were reported in 22% and 51% of the NG101 patients and placebo patients, respectively.
In addition, participants who received NG101 reported a 70% decrease in nausea severity from baseline.
Overall, patients in the NG101 group also reported significantly fewer adverse events than those in the placebo group (74 vs 135), suggesting an improved safety profile when semaglutide is administered in conjunction with NG101, the researchers noted. No serious adverse events related to the study drug were reported in either group.
The findings were limited by several factors including the relatively small sample size. Additional research is needed with other GLP-1 agonists in larger populations with longer follow-up periods, Cummings said. However, the results suggest that NG101 was safe and effectively improved side effects associated with GLP-1 agonists.
“We know there are receptors for GLP-1 in the area postrema (nausea center of the brain), and that NG101 works on this area to reduce nausea and vomiting, so the study findings were not unexpected,” said Jim O’Mara, president and CEO of Neurogastrx, in an interview.
The study was a single-dose study designed to show proof of concept, and future studies would involve treating patients going through the recommended titration schedule for their GLP-1s, O’Mara said. However, NG101 offers an opportunity to keep more patients on GLP-1 therapy and help them reach their long-term therapeutic goals, he said.
Decrease Side Effects for Weight-Loss Success
“GI side effects are often the rate-limiting step in implementing an effective medication that patients want to take but may not be able to tolerate,” said Sean Wharton, MD, PharmD, medical director of the Wharton Medical Clinic for Weight and Diabetes Management, Burlington, Ontario, Canada, in an interview. “If we can decrease side effects, these medications could improve patients’ lives,” said Wharton, who was not involved in the study.
The improvement after a single dose of NG101 in patients receiving a single dose of semaglutide was impressive and in keeping with the mechanism of the drug action, said Wharton. “I was not surprised by the result but pleased that this single dose was shown to reduce the overall incidence of nausea and vomiting, the duration of nausea, the severity of nausea as rated by the study participants compared to placebo,” he said.
Ultimately, the clinical implications for NG101 are improved patient tolerance for GLP-1s and the ability to titrate and stay on them long term, incurring greater cardiometabolic benefit, Wharton told this news organization.
The current trial was limited to GLP1-1s on the market; newer medications may have fewer side effects, Wharton noted. “In clinical practice, patients often decrease the medication or titrate slower, and this could be the comparator,” he added.
The study was funded by Neurogastrx.
Wharton disclosed serving as a consultant for Neurogastrx but not as an investigator on the current study. He also reported having disclosed research on various GLP-1 medications.
A version of this article first appeared on Medscape.com.
FROM OBESITY WEEK 2025
Approach to Weight Management in GI Practice
Introduction
The majority of patients in the United States are now overweight or obese, and as gastroenterologists we treat a number of conditions that are caused or worsened by obesity.1 Cirrhosis related to metabolic associated fatty liver disease (MAFLD) is now a leading indication for liver transplantation in the US2 and obesity is a clear risk factor for all major malignancies of the GI tract, including esophageal, gastric cardia, pancreatic, liver, gallbladder, colon, and rectum.3 Obesity is associated with dysbiosis and impacts barrier function: increasing permeability, abnormal gut bacterial translocation, and inflammation.4 It is more common than malnutrition in our patients with inflammatory bowel disease (IBD), where it impacts response to biologic drugs, increases the technical difficulty of surgeries, such as IPAA, and is associated with worse surgical outcomes.5 Furthermore, patients with obesity may be less likely to undergo preventative cancer screenings and are at increased risk related to sedation for endoscopic procedures.6 With over 40% of Americans suffering from obesity, and increasingly effective treatments available,
Understanding the Mechanisms of Obesity
There are complex orexigenic and anorexigenic brain pathways in the hypothalamus which control global energy balance.7 Obesity results when energy intake exceeds energy expenditure. While overeating and a sedentary lifestyle are commonly blamed, there are a number of elements that contribute, including genetics, medical conditions, medications, psychosocial factors, and environmental components. For example, sleep loss contributes to weight gain by several mechanisms including increasing ghrelin and decreasing leptin levels, thereby increasing hunger and appetite, as well as by decreasing insulin sensitivity and increasing cortisol. Subjects exposed to sleep deprivation in research settings take in 550 kcal more the following day.8 Medications used commonly in GI practice including corticosteroids, antihistamines, propranolol, and amitriptyline, are obesogenic9 and cannabis can impact hypothalamic pathways to stimulate hunger.10
When patients diet or exercise to lose weight, as we have traditionally advised, there are strong hormonal changes and metabolic adaptations that occur to preserve the defended fat mass or “set point.” Loss of adipose tissue results in decreased production of leptin, a hormone that stimulates satiety pathways and inhibits orexigenic pathways, greatly increasing hunger and cravings. Increases in ghrelin production by the stomach decreases perceptions of fullness. With weight loss, energy requirements decrease, and muscles become more efficient, meaning fewer kcal are needed to maintain bodily processes.11 Eventually a plateau is reached, while motivation to diet and restraint around food wane, and hedonistic (reward) pathways are activated. These powerful factors result in the regain of lost weight within one year in the majority of patients.
Implementing Weight Management into GI Practice
Given the stigma and bias around obesity, patients often feel shame and vulnerability around the condition. It is important to have empathy in your approach, asking permission to discuss weight and using patient-first language (e.g. “patient with obesity” not “obese patient”). While BMI is predictive of health outcomes, it does not measure body fat percentage and may be misleading, such as in muscular individuals. Other measures of adiposity including waist circumference and body composition testing, such as with DEXA, may provide additional data. A BMI of 30 or above defines obesity, though newer definitions incorporate related symptoms, organ disfunction, and metabolic abnormalities into the term “clinical obesity.”12 Asian patients experience metabolic complications at a lower BMI, and therefore the definition of obese is 27.5kg/m2 in this population.
Begin by taking a weight history. Has this been a lifelong struggle or is there a particular life circumstance, such as working a third shift or recent pregnancy which precipitated weight gain? Patients should be asked about binge eating or eating late into the evening or waking at night to eat, as these disordered eating behaviors are managed with specific medications and behavioral therapies. Inquire about sleep duration and quality and refer for a sleep study if there is suspicion for obstructive sleep apnea. Other weight-related comorbidities including hyperlipidemia, type 2 diabetes mellitus (T2DM), and MAFLD should be considered and merit a more aggressive approach, as does more severe obesity (class III, BMI ≥40). Questions about marijuana and alcohol use as well as review of the medication list for obesogenic medications can provide further insight into modifiable contributing factors.
Pillars of Weight Management
The internet is awash with trendy diet recommendations, and widespread misconceptions about obesity management are even ingrained into how physicians approach the disease. It is critical to remember that this is not a consequence of bad choices or lack of self-control. Exercise alone is insufficient to result in significant weight loss.13 Furthermore, whether it is through low fat, low carb, or intermittent fasting, weight loss will occur with calorie deficit.14 Evidence-based diet and lifestyle recommendations to lay the groundwork for success should be discussed at each visit (see Table 1). The Mediterranean diet is recommended for weight loss as well as for several GI disorders (i.e., MAFLD and IBD) and is the optimal eating strategy for cardiovascular health.15 Patients should be advised to engage in 150 minutes of moderate exercise per week, such as brisk walking, and should incorporate resistance training to build muscle and maintain bone density.
Anti-obesity Medications
There are a number of medications, either FDA approved or used off label, for treatment of obesity (see Table 2).16 All are indicated for patients with a BMI of ≥ 30 kg/m2 or for those with a BMI between 27-29 kg/m2 with weight-related comorbidities and should be used in combination with diet and lifestyle interventions. None are approved or safe in pregnancy. Mechanisms of action vary by type and include decreased appetite, increased energy expenditure, improved insulin sensitivity, and interfere with absorption.
The newest and most effective anti-obesity medications (AOM), the glucagon-like peptide-1 receptor agonists (GLP-1 RA) are derived from gut hormones secreted in the distal small bowel and colon in response to a meal, which function to delay gastric emptying, increase insulin release from the pancreas, and reduce hepatic gluconeogenesis. Central nervous system effects are not yet entirely understood, but function to decrease appetite and increase satiety. Initially developed for treatment of T2DM, observed weight reduction in patients treated with GLP-1 RA led to clinical trials for treatment of obesity. Semaglutide treatment resulted in weight reduction of 16.9% of total body weight (TBW), and one third of subjects lost ≥ 20% of TBW.17 Tirzepatide combines GLP-1 RA and a gastric inhibitory polypeptide (GIP) receptor agonist, which also has an incretin effect and functions to slow gastric emptying. In the pivotal SURMOUNT trial, approximately 58% of patients achieved ≥20% loss of TBW18 with 15mg weekly dosing of tirzepatide. This class of drugs is a logical choice in patients with T2DM and obesity. Long-term treatment appears necessary, as patients typically regain two-thirds of lost weight within a year after GLP-1 RA are stopped.
Based on tumors observed in rodents, GLP-1 RA are contraindicated in patients with a personal or family history of multiple endocrine neoplasia type 2 (MEN II) or medullary thyroid cancer. These tumors have not been observed in humans treated with GLP-1 RA. They should be used with caution in patients with history of pancreatitis, gastroparesis, or diabetic retinopathy, though a recent systematic review and meta-analysis suggests showed little to no increased risk for biliary events from GLP-1 RA.19 Side effects are most commonly gastrointestinal in nature (nausea, reflux, constipation or diarrhea) and are typically most severe with initiation of the drug and with dose escalation. Side effects can be mitigated by initiating these drugs at lowest doses and gradually titrating up (every four weeks) based on effectiveness and tolerability. Antisecretory, antiemetic, and laxative medications can also be used to help manage GLP-1 RA related side effects.
There is no reason to escalate to highest doses if patients are experiencing weight loss and reduction in food cravings at lower doses. Both semaglutide and tirzepatide are administered subcutaneously every seven days. Once patients have reached goal weight, they can either continue maintenance therapy at that same dose/interval, or if motivated to do so, may gradually reduce the weekly dose in a stepwise approach to determine the minimally effective dose to maintain weight loss. There are not yet published maintenance studies to guide this process. Currently the price of GLP-1 RA and inconsistent insurance coverage make them inaccessible to many patients. The manufacturers of both semaglutide and tirzepatide offer direct to consumer pricing and home delivery.
Bariatric Surgery
In patients with higher BMI (≥35kg/m2) or those with BMI ≥30kg/m2 and obesity-related metabolic disease and the desire to avoid lifelong medications or who fail or are intolerant of AOM, bariatric options should be considered.20 Sleeve gastrectomy has become the most performed surgery for treatment of obesity. It is a restrictive procedure, removing 80% of the stomach, but a drop in circulating levels of ghrelin afterwards also leads to decreased feelings of hunger. It results in weight loss of 25-30% TBW loss. It is not a good choice for patients who suffer from severe GERD, as this typically worsens afterwards; furthermore, de novo Barrett’s has been observed in nearly 6% of patients who undergo sleeve gastrectomy.21
Roux-en-Y gastric bypass is a restrictive and malabsorptive procedure, resulting in 30-35% TBW loss. It has beneficial and immediate metabolic effects, including increased release of endogenous GLP-1, which leads to improvements in weight-related T2DM. The newer single anastomosis duodenal-ileal bypass with sleeve gastrectomy (SADI-S) starts with a sleeve gastrectomy, making a smaller tube-shaped stomach. The duodenum is divided just after the stomach and then a loop of ileum is brought up and connected to the stomach (see Figure 1). This procedure is highly effective, with patients losing 75-95% of excess body weight and is becoming a preferred option for patients with greater BMI (≥50kg/m2). It is also an option for patients who have already had a sleeve gastrectomy and are seeking further weight loss. Because there is only one anastomosis, perioperative complications, such as anastomotic leaks, are reduced. The risk of micronutrient deficiencies is present with all malabsorptive procedures, and these patients must supplement with multivitamins, iron, vitamin D, and calcium.
Endoscopic Therapies
Endoscopic bariatric and metabolic therapies (EBMTs) have been increasingly studied and utilized, and this less invasive option may be more appropriate for or attractive to many patients. Intragastric balloons, which reduce meal volume and delay gastric emptying, can be used short term only (six months) resulting in loss of about 6.9% of total body weight (TBW) greater than lifestyle modification (LM) alone, and may be considered in limited situations, such as need for pre-operative weight loss to reduce risks in very obese individuals.22
Endoscopic gastric remodeling (EGR), also known as endoscopic sleeve gastrectomy (ESG), is a purely restrictive procedure in which the stomach is cinched to resize and reshape using an endoscopic suturing device (see Figure 2).23 It is an option for patients with class 1 or 2 obesity, with data from a randomized controlled trial in this population demonstrating mean percentage of TBW loss of 13.6% at 52 weeks compared to 0.8% in those treated with LM alone.24 A recent meta-analysis of 21 observational studies, including patients with higher BMIs (32.5 to 49.9 kg/m2) showed pooled average weight loss of 17.3% TBW at 12 months with EGR.22 This procedure has potential advantages of fewer complications, quicker recovery, and much less new-onset GERD compared to laparoscopic sleeve gastrectomy. Furthermore, it may be utilized in combination with AOMs to achieve optimum weight loss and metabolic outcomes.25,26 Potential adverse events include abdominal pain, nausea and vomiting (which may be severe), as well as rare instances of intra/extra luminal bleeding or abdominal abscess requiring drainage.22
Recent joint American/European Gastrointestinal Endoscopy guidelines suggest the use of EBMTs plus lifestyle modification in patients with a BMI of ≥ 30 kg/m2, or with a BMI of 27.0-29.9 kg/m2 with at least 1 obesity-related comorbidity.22 Small bowel interventions including duodenal-jejunal bypass liner and duodenal mucosal resurfacing are being investigated for patients with obesity and type 2 diabetes but not yet commercially available.
Conclusion
Given the overlap of obesity with many GI disorders, it is entirely appropriate for gastroenterologists to consider it worthy of aggressive treatment, particularly in patients with MAFLD and other serious weight related comorbidities. With a compassionate and empathetic approach, and a number of highly effective medical, endoscopic, and surgical therapies now available, weight management has the potential to be extremely rewarding when implemented in GI practice.
Dr. Kelly is based in the Department of Medicine, Division of Gastroenterology, Brigham and Women’s Hospital, and Harvard Medical School, both in Boston, Massachusetts. She serves on the clinical advisory board for OpenBiome (unpaid) and has served on an advisory board for Eli Lilly and Company.
References
1. Hales CM, et al. Prevalence of Obesity and Severe Obesity Among Adults: United States, 2017-2018. NCHS Data Brief 2020 Feb:(360):1–8.
2. Pais R, et al. NAFLD and liver transplantation: Current burden and expected challenges. J Hepatol. 2016 Dec. doi: 10.1016/j.jhep.2016.07.033.
3. Lauby-Secretan B, et al. Body Fatness and Cancer--Viewpoint of the IARC Working Group. N Engl J Med. 2016 Aug. doi: 10.1056/NEJMsr1606602.
4. Kim A. Dysbiosis: A Review Highlighting Obesity and Inflammatory Bowel Disease. J Clin Gastroenterol. 2015 Nov-Dec. doi: 10.1097/MCG.0000000000000356.
5. Singh S, et al. Obesity in IBD: epidemiology, pathogenesis, disease course and treatment outcomes. Nat Rev Gastroenterol Hepatol. 2017 Feb. doi: 10.1038/nrgastro.2016.181.
6. Sundararaman L, Goudra B. Sedation for GI Endoscopy in the Morbidly Obese: Challenges and Possible Solutions. J Clin Med. 2024 Aug. doi: 10.3390/jcm13164635.
7. Bombassaro B, et al. The hypothalamus as the central regulator of energy balance and its impact on current and future obesity treatments. Arch Endocrinol Metab. 2024 Nov. doi: 10.20945/2359-4292-2024-0082.
8. Beccuti G, Pannain S. Sleep and obesity. Curr Opin Clin Nutr Metab Care. 2011 Jul. doi: 10.1097/MCO.0b013e3283479109.
9. Desalermos A, et al. Effect of Obesogenic Medications on Weight-Loss Outcomes in a Behavioral Weight-Management Program. Obesity (Silver Spring). 2019 May. doi: 10.1002/oby.22444.
10. Lord MN, Noble EE. Hypothalamic cannabinoid signaling: Consequences for eating behavior. Pharmacol Res Perspect. 2024 Oct. doi: 10.1002/prp2.1251.
11. Farhana A, Rehman A. Metabolic Consequences of Weight Reduction. [Updated 2023 Jul 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan. Available from: https://www.ncbi.nlm.nih.gov/books/NBK572145/.
12. Rubino F, et al. Definition and diagnostic criteria of clinical obesity. Lancet Diabetes Endocrinol. 2025 Mar. doi: 10.1016/S2213-8587(24)00316-4.
13. Cox CE. Role of Physical Activity for Weight Loss and Weight Maintenance. Diabetes Spectr. 2017 Aug. doi: 10.2337/ds17-0013.
14. Chaput JP, et al. Widespread misconceptions about obesity. Can Fam Physician. 2014 Nov. PMID: 25392431.
15. Muscogiuri G, et al. Mediterranean Diet and Obesity-related Disorders: What is the Evidence? Curr Obes Rep. 2022 Dec. doi: 10.1007/s13679-022-00481-1.
16. Gudzune KA, Kushner RF. Medications for Obesity: A Review. JAMA. 2024 Aug. doi: 10.1001/jama.2024.10816.
17. Wilding JPH, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021 Feb. doi: 10.1056/NEJMoa2032183.
18. Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022 Jun. doi: 10.1056/NEJMoa2206038.
19. Chiang CH, et al. Glucagon-Like Peptide-1 Receptor Agonists and Gastrointestinal Adverse Events: A Systematic Review and Meta-Analysis. Gastroenterology. 2025 Nov. doi: 10.1053/j.gastro.2025.06.003.
20. Aderinto N, et al. Recent advances in bariatric surgery: a narrative review of weight loss procedures. Ann Med Surg (Lond). 2023 Nov. doi: 10.1097/MS9.0000000000001472.
21. Chandan S, et al. Risk of De Novo Barrett’s Esophagus Post Sleeve Gastrectomy: A Systematic Review and Meta-Analysis of Studies With Long-Term Follow-Up. Clin Gastroenterol Hepatol. 2025 Jan. doi: 10.1016/j.cgh.2024.06.041.
22. Jirapinyo P, et al. American Society for Gastrointestinal Endoscopy-European Society of Gastrointestinal Endoscopy guideline on primary endoscopic bariatric and metabolic therapies for adults with obesity. Gastrointest Endosc. 2024 Jun. doi: 10.1016/j.gie.2023.12.004.
23. Nduma BN, et al. Endoscopic Gastric Sleeve: A Review of Literature. Cureus. 2023 Mar. doi: 10.7759/cureus.36353.
24. Abu Dayyeh BK, et al. Endoscopic sleeve gastroplasty for treatment of class 1 and 2 obesity (MERIT): a prospective, multicentre, randomised trial. Lancet. 2022 Aug. doi: 10.1016/S0140-6736(22)01280-6.
25. Gala K, et al. Outcomes of concomitant antiobesity medication use with endoscopic sleeve gastroplasty in clinical US settings. Obes Pillars. 2024 May. doi: 10.1016/j.obpill.2024.100112.
26. Chung CS, et al. Endoscopic sleeve gastroplasty combined with anti-obesity medication for better control of weight and diabetes. Clin Endosc. 2025 May. doi: 10.5946/ce.2024.274.
Introduction
The majority of patients in the United States are now overweight or obese, and as gastroenterologists we treat a number of conditions that are caused or worsened by obesity.1 Cirrhosis related to metabolic associated fatty liver disease (MAFLD) is now a leading indication for liver transplantation in the US2 and obesity is a clear risk factor for all major malignancies of the GI tract, including esophageal, gastric cardia, pancreatic, liver, gallbladder, colon, and rectum.3 Obesity is associated with dysbiosis and impacts barrier function: increasing permeability, abnormal gut bacterial translocation, and inflammation.4 It is more common than malnutrition in our patients with inflammatory bowel disease (IBD), where it impacts response to biologic drugs, increases the technical difficulty of surgeries, such as IPAA, and is associated with worse surgical outcomes.5 Furthermore, patients with obesity may be less likely to undergo preventative cancer screenings and are at increased risk related to sedation for endoscopic procedures.6 With over 40% of Americans suffering from obesity, and increasingly effective treatments available,
Understanding the Mechanisms of Obesity
There are complex orexigenic and anorexigenic brain pathways in the hypothalamus which control global energy balance.7 Obesity results when energy intake exceeds energy expenditure. While overeating and a sedentary lifestyle are commonly blamed, there are a number of elements that contribute, including genetics, medical conditions, medications, psychosocial factors, and environmental components. For example, sleep loss contributes to weight gain by several mechanisms including increasing ghrelin and decreasing leptin levels, thereby increasing hunger and appetite, as well as by decreasing insulin sensitivity and increasing cortisol. Subjects exposed to sleep deprivation in research settings take in 550 kcal more the following day.8 Medications used commonly in GI practice including corticosteroids, antihistamines, propranolol, and amitriptyline, are obesogenic9 and cannabis can impact hypothalamic pathways to stimulate hunger.10
When patients diet or exercise to lose weight, as we have traditionally advised, there are strong hormonal changes and metabolic adaptations that occur to preserve the defended fat mass or “set point.” Loss of adipose tissue results in decreased production of leptin, a hormone that stimulates satiety pathways and inhibits orexigenic pathways, greatly increasing hunger and cravings. Increases in ghrelin production by the stomach decreases perceptions of fullness. With weight loss, energy requirements decrease, and muscles become more efficient, meaning fewer kcal are needed to maintain bodily processes.11 Eventually a plateau is reached, while motivation to diet and restraint around food wane, and hedonistic (reward) pathways are activated. These powerful factors result in the regain of lost weight within one year in the majority of patients.
Implementing Weight Management into GI Practice
Given the stigma and bias around obesity, patients often feel shame and vulnerability around the condition. It is important to have empathy in your approach, asking permission to discuss weight and using patient-first language (e.g. “patient with obesity” not “obese patient”). While BMI is predictive of health outcomes, it does not measure body fat percentage and may be misleading, such as in muscular individuals. Other measures of adiposity including waist circumference and body composition testing, such as with DEXA, may provide additional data. A BMI of 30 or above defines obesity, though newer definitions incorporate related symptoms, organ disfunction, and metabolic abnormalities into the term “clinical obesity.”12 Asian patients experience metabolic complications at a lower BMI, and therefore the definition of obese is 27.5kg/m2 in this population.
Begin by taking a weight history. Has this been a lifelong struggle or is there a particular life circumstance, such as working a third shift or recent pregnancy which precipitated weight gain? Patients should be asked about binge eating or eating late into the evening or waking at night to eat, as these disordered eating behaviors are managed with specific medications and behavioral therapies. Inquire about sleep duration and quality and refer for a sleep study if there is suspicion for obstructive sleep apnea. Other weight-related comorbidities including hyperlipidemia, type 2 diabetes mellitus (T2DM), and MAFLD should be considered and merit a more aggressive approach, as does more severe obesity (class III, BMI ≥40). Questions about marijuana and alcohol use as well as review of the medication list for obesogenic medications can provide further insight into modifiable contributing factors.
Pillars of Weight Management
The internet is awash with trendy diet recommendations, and widespread misconceptions about obesity management are even ingrained into how physicians approach the disease. It is critical to remember that this is not a consequence of bad choices or lack of self-control. Exercise alone is insufficient to result in significant weight loss.13 Furthermore, whether it is through low fat, low carb, or intermittent fasting, weight loss will occur with calorie deficit.14 Evidence-based diet and lifestyle recommendations to lay the groundwork for success should be discussed at each visit (see Table 1). The Mediterranean diet is recommended for weight loss as well as for several GI disorders (i.e., MAFLD and IBD) and is the optimal eating strategy for cardiovascular health.15 Patients should be advised to engage in 150 minutes of moderate exercise per week, such as brisk walking, and should incorporate resistance training to build muscle and maintain bone density.
Anti-obesity Medications
There are a number of medications, either FDA approved or used off label, for treatment of obesity (see Table 2).16 All are indicated for patients with a BMI of ≥ 30 kg/m2 or for those with a BMI between 27-29 kg/m2 with weight-related comorbidities and should be used in combination with diet and lifestyle interventions. None are approved or safe in pregnancy. Mechanisms of action vary by type and include decreased appetite, increased energy expenditure, improved insulin sensitivity, and interfere with absorption.
The newest and most effective anti-obesity medications (AOM), the glucagon-like peptide-1 receptor agonists (GLP-1 RA) are derived from gut hormones secreted in the distal small bowel and colon in response to a meal, which function to delay gastric emptying, increase insulin release from the pancreas, and reduce hepatic gluconeogenesis. Central nervous system effects are not yet entirely understood, but function to decrease appetite and increase satiety. Initially developed for treatment of T2DM, observed weight reduction in patients treated with GLP-1 RA led to clinical trials for treatment of obesity. Semaglutide treatment resulted in weight reduction of 16.9% of total body weight (TBW), and one third of subjects lost ≥ 20% of TBW.17 Tirzepatide combines GLP-1 RA and a gastric inhibitory polypeptide (GIP) receptor agonist, which also has an incretin effect and functions to slow gastric emptying. In the pivotal SURMOUNT trial, approximately 58% of patients achieved ≥20% loss of TBW18 with 15mg weekly dosing of tirzepatide. This class of drugs is a logical choice in patients with T2DM and obesity. Long-term treatment appears necessary, as patients typically regain two-thirds of lost weight within a year after GLP-1 RA are stopped.
Based on tumors observed in rodents, GLP-1 RA are contraindicated in patients with a personal or family history of multiple endocrine neoplasia type 2 (MEN II) or medullary thyroid cancer. These tumors have not been observed in humans treated with GLP-1 RA. They should be used with caution in patients with history of pancreatitis, gastroparesis, or diabetic retinopathy, though a recent systematic review and meta-analysis suggests showed little to no increased risk for biliary events from GLP-1 RA.19 Side effects are most commonly gastrointestinal in nature (nausea, reflux, constipation or diarrhea) and are typically most severe with initiation of the drug and with dose escalation. Side effects can be mitigated by initiating these drugs at lowest doses and gradually titrating up (every four weeks) based on effectiveness and tolerability. Antisecretory, antiemetic, and laxative medications can also be used to help manage GLP-1 RA related side effects.
There is no reason to escalate to highest doses if patients are experiencing weight loss and reduction in food cravings at lower doses. Both semaglutide and tirzepatide are administered subcutaneously every seven days. Once patients have reached goal weight, they can either continue maintenance therapy at that same dose/interval, or if motivated to do so, may gradually reduce the weekly dose in a stepwise approach to determine the minimally effective dose to maintain weight loss. There are not yet published maintenance studies to guide this process. Currently the price of GLP-1 RA and inconsistent insurance coverage make them inaccessible to many patients. The manufacturers of both semaglutide and tirzepatide offer direct to consumer pricing and home delivery.
Bariatric Surgery
In patients with higher BMI (≥35kg/m2) or those with BMI ≥30kg/m2 and obesity-related metabolic disease and the desire to avoid lifelong medications or who fail or are intolerant of AOM, bariatric options should be considered.20 Sleeve gastrectomy has become the most performed surgery for treatment of obesity. It is a restrictive procedure, removing 80% of the stomach, but a drop in circulating levels of ghrelin afterwards also leads to decreased feelings of hunger. It results in weight loss of 25-30% TBW loss. It is not a good choice for patients who suffer from severe GERD, as this typically worsens afterwards; furthermore, de novo Barrett’s has been observed in nearly 6% of patients who undergo sleeve gastrectomy.21
Roux-en-Y gastric bypass is a restrictive and malabsorptive procedure, resulting in 30-35% TBW loss. It has beneficial and immediate metabolic effects, including increased release of endogenous GLP-1, which leads to improvements in weight-related T2DM. The newer single anastomosis duodenal-ileal bypass with sleeve gastrectomy (SADI-S) starts with a sleeve gastrectomy, making a smaller tube-shaped stomach. The duodenum is divided just after the stomach and then a loop of ileum is brought up and connected to the stomach (see Figure 1). This procedure is highly effective, with patients losing 75-95% of excess body weight and is becoming a preferred option for patients with greater BMI (≥50kg/m2). It is also an option for patients who have already had a sleeve gastrectomy and are seeking further weight loss. Because there is only one anastomosis, perioperative complications, such as anastomotic leaks, are reduced. The risk of micronutrient deficiencies is present with all malabsorptive procedures, and these patients must supplement with multivitamins, iron, vitamin D, and calcium.
Endoscopic Therapies
Endoscopic bariatric and metabolic therapies (EBMTs) have been increasingly studied and utilized, and this less invasive option may be more appropriate for or attractive to many patients. Intragastric balloons, which reduce meal volume and delay gastric emptying, can be used short term only (six months) resulting in loss of about 6.9% of total body weight (TBW) greater than lifestyle modification (LM) alone, and may be considered in limited situations, such as need for pre-operative weight loss to reduce risks in very obese individuals.22
Endoscopic gastric remodeling (EGR), also known as endoscopic sleeve gastrectomy (ESG), is a purely restrictive procedure in which the stomach is cinched to resize and reshape using an endoscopic suturing device (see Figure 2).23 It is an option for patients with class 1 or 2 obesity, with data from a randomized controlled trial in this population demonstrating mean percentage of TBW loss of 13.6% at 52 weeks compared to 0.8% in those treated with LM alone.24 A recent meta-analysis of 21 observational studies, including patients with higher BMIs (32.5 to 49.9 kg/m2) showed pooled average weight loss of 17.3% TBW at 12 months with EGR.22 This procedure has potential advantages of fewer complications, quicker recovery, and much less new-onset GERD compared to laparoscopic sleeve gastrectomy. Furthermore, it may be utilized in combination with AOMs to achieve optimum weight loss and metabolic outcomes.25,26 Potential adverse events include abdominal pain, nausea and vomiting (which may be severe), as well as rare instances of intra/extra luminal bleeding or abdominal abscess requiring drainage.22
Recent joint American/European Gastrointestinal Endoscopy guidelines suggest the use of EBMTs plus lifestyle modification in patients with a BMI of ≥ 30 kg/m2, or with a BMI of 27.0-29.9 kg/m2 with at least 1 obesity-related comorbidity.22 Small bowel interventions including duodenal-jejunal bypass liner and duodenal mucosal resurfacing are being investigated for patients with obesity and type 2 diabetes but not yet commercially available.
Conclusion
Given the overlap of obesity with many GI disorders, it is entirely appropriate for gastroenterologists to consider it worthy of aggressive treatment, particularly in patients with MAFLD and other serious weight related comorbidities. With a compassionate and empathetic approach, and a number of highly effective medical, endoscopic, and surgical therapies now available, weight management has the potential to be extremely rewarding when implemented in GI practice.
Dr. Kelly is based in the Department of Medicine, Division of Gastroenterology, Brigham and Women’s Hospital, and Harvard Medical School, both in Boston, Massachusetts. She serves on the clinical advisory board for OpenBiome (unpaid) and has served on an advisory board for Eli Lilly and Company.
References
1. Hales CM, et al. Prevalence of Obesity and Severe Obesity Among Adults: United States, 2017-2018. NCHS Data Brief 2020 Feb:(360):1–8.
2. Pais R, et al. NAFLD and liver transplantation: Current burden and expected challenges. J Hepatol. 2016 Dec. doi: 10.1016/j.jhep.2016.07.033.
3. Lauby-Secretan B, et al. Body Fatness and Cancer--Viewpoint of the IARC Working Group. N Engl J Med. 2016 Aug. doi: 10.1056/NEJMsr1606602.
4. Kim A. Dysbiosis: A Review Highlighting Obesity and Inflammatory Bowel Disease. J Clin Gastroenterol. 2015 Nov-Dec. doi: 10.1097/MCG.0000000000000356.
5. Singh S, et al. Obesity in IBD: epidemiology, pathogenesis, disease course and treatment outcomes. Nat Rev Gastroenterol Hepatol. 2017 Feb. doi: 10.1038/nrgastro.2016.181.
6. Sundararaman L, Goudra B. Sedation for GI Endoscopy in the Morbidly Obese: Challenges and Possible Solutions. J Clin Med. 2024 Aug. doi: 10.3390/jcm13164635.
7. Bombassaro B, et al. The hypothalamus as the central regulator of energy balance and its impact on current and future obesity treatments. Arch Endocrinol Metab. 2024 Nov. doi: 10.20945/2359-4292-2024-0082.
8. Beccuti G, Pannain S. Sleep and obesity. Curr Opin Clin Nutr Metab Care. 2011 Jul. doi: 10.1097/MCO.0b013e3283479109.
9. Desalermos A, et al. Effect of Obesogenic Medications on Weight-Loss Outcomes in a Behavioral Weight-Management Program. Obesity (Silver Spring). 2019 May. doi: 10.1002/oby.22444.
10. Lord MN, Noble EE. Hypothalamic cannabinoid signaling: Consequences for eating behavior. Pharmacol Res Perspect. 2024 Oct. doi: 10.1002/prp2.1251.
11. Farhana A, Rehman A. Metabolic Consequences of Weight Reduction. [Updated 2023 Jul 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan. Available from: https://www.ncbi.nlm.nih.gov/books/NBK572145/.
12. Rubino F, et al. Definition and diagnostic criteria of clinical obesity. Lancet Diabetes Endocrinol. 2025 Mar. doi: 10.1016/S2213-8587(24)00316-4.
13. Cox CE. Role of Physical Activity for Weight Loss and Weight Maintenance. Diabetes Spectr. 2017 Aug. doi: 10.2337/ds17-0013.
14. Chaput JP, et al. Widespread misconceptions about obesity. Can Fam Physician. 2014 Nov. PMID: 25392431.
15. Muscogiuri G, et al. Mediterranean Diet and Obesity-related Disorders: What is the Evidence? Curr Obes Rep. 2022 Dec. doi: 10.1007/s13679-022-00481-1.
16. Gudzune KA, Kushner RF. Medications for Obesity: A Review. JAMA. 2024 Aug. doi: 10.1001/jama.2024.10816.
17. Wilding JPH, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021 Feb. doi: 10.1056/NEJMoa2032183.
18. Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022 Jun. doi: 10.1056/NEJMoa2206038.
19. Chiang CH, et al. Glucagon-Like Peptide-1 Receptor Agonists and Gastrointestinal Adverse Events: A Systematic Review and Meta-Analysis. Gastroenterology. 2025 Nov. doi: 10.1053/j.gastro.2025.06.003.
20. Aderinto N, et al. Recent advances in bariatric surgery: a narrative review of weight loss procedures. Ann Med Surg (Lond). 2023 Nov. doi: 10.1097/MS9.0000000000001472.
21. Chandan S, et al. Risk of De Novo Barrett’s Esophagus Post Sleeve Gastrectomy: A Systematic Review and Meta-Analysis of Studies With Long-Term Follow-Up. Clin Gastroenterol Hepatol. 2025 Jan. doi: 10.1016/j.cgh.2024.06.041.
22. Jirapinyo P, et al. American Society for Gastrointestinal Endoscopy-European Society of Gastrointestinal Endoscopy guideline on primary endoscopic bariatric and metabolic therapies for adults with obesity. Gastrointest Endosc. 2024 Jun. doi: 10.1016/j.gie.2023.12.004.
23. Nduma BN, et al. Endoscopic Gastric Sleeve: A Review of Literature. Cureus. 2023 Mar. doi: 10.7759/cureus.36353.
24. Abu Dayyeh BK, et al. Endoscopic sleeve gastroplasty for treatment of class 1 and 2 obesity (MERIT): a prospective, multicentre, randomised trial. Lancet. 2022 Aug. doi: 10.1016/S0140-6736(22)01280-6.
25. Gala K, et al. Outcomes of concomitant antiobesity medication use with endoscopic sleeve gastroplasty in clinical US settings. Obes Pillars. 2024 May. doi: 10.1016/j.obpill.2024.100112.
26. Chung CS, et al. Endoscopic sleeve gastroplasty combined with anti-obesity medication for better control of weight and diabetes. Clin Endosc. 2025 May. doi: 10.5946/ce.2024.274.
Introduction
The majority of patients in the United States are now overweight or obese, and as gastroenterologists we treat a number of conditions that are caused or worsened by obesity.1 Cirrhosis related to metabolic associated fatty liver disease (MAFLD) is now a leading indication for liver transplantation in the US2 and obesity is a clear risk factor for all major malignancies of the GI tract, including esophageal, gastric cardia, pancreatic, liver, gallbladder, colon, and rectum.3 Obesity is associated with dysbiosis and impacts barrier function: increasing permeability, abnormal gut bacterial translocation, and inflammation.4 It is more common than malnutrition in our patients with inflammatory bowel disease (IBD), where it impacts response to biologic drugs, increases the technical difficulty of surgeries, such as IPAA, and is associated with worse surgical outcomes.5 Furthermore, patients with obesity may be less likely to undergo preventative cancer screenings and are at increased risk related to sedation for endoscopic procedures.6 With over 40% of Americans suffering from obesity, and increasingly effective treatments available,
Understanding the Mechanisms of Obesity
There are complex orexigenic and anorexigenic brain pathways in the hypothalamus which control global energy balance.7 Obesity results when energy intake exceeds energy expenditure. While overeating and a sedentary lifestyle are commonly blamed, there are a number of elements that contribute, including genetics, medical conditions, medications, psychosocial factors, and environmental components. For example, sleep loss contributes to weight gain by several mechanisms including increasing ghrelin and decreasing leptin levels, thereby increasing hunger and appetite, as well as by decreasing insulin sensitivity and increasing cortisol. Subjects exposed to sleep deprivation in research settings take in 550 kcal more the following day.8 Medications used commonly in GI practice including corticosteroids, antihistamines, propranolol, and amitriptyline, are obesogenic9 and cannabis can impact hypothalamic pathways to stimulate hunger.10
When patients diet or exercise to lose weight, as we have traditionally advised, there are strong hormonal changes and metabolic adaptations that occur to preserve the defended fat mass or “set point.” Loss of adipose tissue results in decreased production of leptin, a hormone that stimulates satiety pathways and inhibits orexigenic pathways, greatly increasing hunger and cravings. Increases in ghrelin production by the stomach decreases perceptions of fullness. With weight loss, energy requirements decrease, and muscles become more efficient, meaning fewer kcal are needed to maintain bodily processes.11 Eventually a plateau is reached, while motivation to diet and restraint around food wane, and hedonistic (reward) pathways are activated. These powerful factors result in the regain of lost weight within one year in the majority of patients.
Implementing Weight Management into GI Practice
Given the stigma and bias around obesity, patients often feel shame and vulnerability around the condition. It is important to have empathy in your approach, asking permission to discuss weight and using patient-first language (e.g. “patient with obesity” not “obese patient”). While BMI is predictive of health outcomes, it does not measure body fat percentage and may be misleading, such as in muscular individuals. Other measures of adiposity including waist circumference and body composition testing, such as with DEXA, may provide additional data. A BMI of 30 or above defines obesity, though newer definitions incorporate related symptoms, organ disfunction, and metabolic abnormalities into the term “clinical obesity.”12 Asian patients experience metabolic complications at a lower BMI, and therefore the definition of obese is 27.5kg/m2 in this population.
Begin by taking a weight history. Has this been a lifelong struggle or is there a particular life circumstance, such as working a third shift or recent pregnancy which precipitated weight gain? Patients should be asked about binge eating or eating late into the evening or waking at night to eat, as these disordered eating behaviors are managed with specific medications and behavioral therapies. Inquire about sleep duration and quality and refer for a sleep study if there is suspicion for obstructive sleep apnea. Other weight-related comorbidities including hyperlipidemia, type 2 diabetes mellitus (T2DM), and MAFLD should be considered and merit a more aggressive approach, as does more severe obesity (class III, BMI ≥40). Questions about marijuana and alcohol use as well as review of the medication list for obesogenic medications can provide further insight into modifiable contributing factors.
Pillars of Weight Management
The internet is awash with trendy diet recommendations, and widespread misconceptions about obesity management are even ingrained into how physicians approach the disease. It is critical to remember that this is not a consequence of bad choices or lack of self-control. Exercise alone is insufficient to result in significant weight loss.13 Furthermore, whether it is through low fat, low carb, or intermittent fasting, weight loss will occur with calorie deficit.14 Evidence-based diet and lifestyle recommendations to lay the groundwork for success should be discussed at each visit (see Table 1). The Mediterranean diet is recommended for weight loss as well as for several GI disorders (i.e., MAFLD and IBD) and is the optimal eating strategy for cardiovascular health.15 Patients should be advised to engage in 150 minutes of moderate exercise per week, such as brisk walking, and should incorporate resistance training to build muscle and maintain bone density.
Anti-obesity Medications
There are a number of medications, either FDA approved or used off label, for treatment of obesity (see Table 2).16 All are indicated for patients with a BMI of ≥ 30 kg/m2 or for those with a BMI between 27-29 kg/m2 with weight-related comorbidities and should be used in combination with diet and lifestyle interventions. None are approved or safe in pregnancy. Mechanisms of action vary by type and include decreased appetite, increased energy expenditure, improved insulin sensitivity, and interfere with absorption.
The newest and most effective anti-obesity medications (AOM), the glucagon-like peptide-1 receptor agonists (GLP-1 RA) are derived from gut hormones secreted in the distal small bowel and colon in response to a meal, which function to delay gastric emptying, increase insulin release from the pancreas, and reduce hepatic gluconeogenesis. Central nervous system effects are not yet entirely understood, but function to decrease appetite and increase satiety. Initially developed for treatment of T2DM, observed weight reduction in patients treated with GLP-1 RA led to clinical trials for treatment of obesity. Semaglutide treatment resulted in weight reduction of 16.9% of total body weight (TBW), and one third of subjects lost ≥ 20% of TBW.17 Tirzepatide combines GLP-1 RA and a gastric inhibitory polypeptide (GIP) receptor agonist, which also has an incretin effect and functions to slow gastric emptying. In the pivotal SURMOUNT trial, approximately 58% of patients achieved ≥20% loss of TBW18 with 15mg weekly dosing of tirzepatide. This class of drugs is a logical choice in patients with T2DM and obesity. Long-term treatment appears necessary, as patients typically regain two-thirds of lost weight within a year after GLP-1 RA are stopped.
Based on tumors observed in rodents, GLP-1 RA are contraindicated in patients with a personal or family history of multiple endocrine neoplasia type 2 (MEN II) or medullary thyroid cancer. These tumors have not been observed in humans treated with GLP-1 RA. They should be used with caution in patients with history of pancreatitis, gastroparesis, or diabetic retinopathy, though a recent systematic review and meta-analysis suggests showed little to no increased risk for biliary events from GLP-1 RA.19 Side effects are most commonly gastrointestinal in nature (nausea, reflux, constipation or diarrhea) and are typically most severe with initiation of the drug and with dose escalation. Side effects can be mitigated by initiating these drugs at lowest doses and gradually titrating up (every four weeks) based on effectiveness and tolerability. Antisecretory, antiemetic, and laxative medications can also be used to help manage GLP-1 RA related side effects.
There is no reason to escalate to highest doses if patients are experiencing weight loss and reduction in food cravings at lower doses. Both semaglutide and tirzepatide are administered subcutaneously every seven days. Once patients have reached goal weight, they can either continue maintenance therapy at that same dose/interval, or if motivated to do so, may gradually reduce the weekly dose in a stepwise approach to determine the minimally effective dose to maintain weight loss. There are not yet published maintenance studies to guide this process. Currently the price of GLP-1 RA and inconsistent insurance coverage make them inaccessible to many patients. The manufacturers of both semaglutide and tirzepatide offer direct to consumer pricing and home delivery.
Bariatric Surgery
In patients with higher BMI (≥35kg/m2) or those with BMI ≥30kg/m2 and obesity-related metabolic disease and the desire to avoid lifelong medications or who fail or are intolerant of AOM, bariatric options should be considered.20 Sleeve gastrectomy has become the most performed surgery for treatment of obesity. It is a restrictive procedure, removing 80% of the stomach, but a drop in circulating levels of ghrelin afterwards also leads to decreased feelings of hunger. It results in weight loss of 25-30% TBW loss. It is not a good choice for patients who suffer from severe GERD, as this typically worsens afterwards; furthermore, de novo Barrett’s has been observed in nearly 6% of patients who undergo sleeve gastrectomy.21
Roux-en-Y gastric bypass is a restrictive and malabsorptive procedure, resulting in 30-35% TBW loss. It has beneficial and immediate metabolic effects, including increased release of endogenous GLP-1, which leads to improvements in weight-related T2DM. The newer single anastomosis duodenal-ileal bypass with sleeve gastrectomy (SADI-S) starts with a sleeve gastrectomy, making a smaller tube-shaped stomach. The duodenum is divided just after the stomach and then a loop of ileum is brought up and connected to the stomach (see Figure 1). This procedure is highly effective, with patients losing 75-95% of excess body weight and is becoming a preferred option for patients with greater BMI (≥50kg/m2). It is also an option for patients who have already had a sleeve gastrectomy and are seeking further weight loss. Because there is only one anastomosis, perioperative complications, such as anastomotic leaks, are reduced. The risk of micronutrient deficiencies is present with all malabsorptive procedures, and these patients must supplement with multivitamins, iron, vitamin D, and calcium.
Endoscopic Therapies
Endoscopic bariatric and metabolic therapies (EBMTs) have been increasingly studied and utilized, and this less invasive option may be more appropriate for or attractive to many patients. Intragastric balloons, which reduce meal volume and delay gastric emptying, can be used short term only (six months) resulting in loss of about 6.9% of total body weight (TBW) greater than lifestyle modification (LM) alone, and may be considered in limited situations, such as need for pre-operative weight loss to reduce risks in very obese individuals.22
Endoscopic gastric remodeling (EGR), also known as endoscopic sleeve gastrectomy (ESG), is a purely restrictive procedure in which the stomach is cinched to resize and reshape using an endoscopic suturing device (see Figure 2).23 It is an option for patients with class 1 or 2 obesity, with data from a randomized controlled trial in this population demonstrating mean percentage of TBW loss of 13.6% at 52 weeks compared to 0.8% in those treated with LM alone.24 A recent meta-analysis of 21 observational studies, including patients with higher BMIs (32.5 to 49.9 kg/m2) showed pooled average weight loss of 17.3% TBW at 12 months with EGR.22 This procedure has potential advantages of fewer complications, quicker recovery, and much less new-onset GERD compared to laparoscopic sleeve gastrectomy. Furthermore, it may be utilized in combination with AOMs to achieve optimum weight loss and metabolic outcomes.25,26 Potential adverse events include abdominal pain, nausea and vomiting (which may be severe), as well as rare instances of intra/extra luminal bleeding or abdominal abscess requiring drainage.22
Recent joint American/European Gastrointestinal Endoscopy guidelines suggest the use of EBMTs plus lifestyle modification in patients with a BMI of ≥ 30 kg/m2, or with a BMI of 27.0-29.9 kg/m2 with at least 1 obesity-related comorbidity.22 Small bowel interventions including duodenal-jejunal bypass liner and duodenal mucosal resurfacing are being investigated for patients with obesity and type 2 diabetes but not yet commercially available.
Conclusion
Given the overlap of obesity with many GI disorders, it is entirely appropriate for gastroenterologists to consider it worthy of aggressive treatment, particularly in patients with MAFLD and other serious weight related comorbidities. With a compassionate and empathetic approach, and a number of highly effective medical, endoscopic, and surgical therapies now available, weight management has the potential to be extremely rewarding when implemented in GI practice.
Dr. Kelly is based in the Department of Medicine, Division of Gastroenterology, Brigham and Women’s Hospital, and Harvard Medical School, both in Boston, Massachusetts. She serves on the clinical advisory board for OpenBiome (unpaid) and has served on an advisory board for Eli Lilly and Company.
References
1. Hales CM, et al. Prevalence of Obesity and Severe Obesity Among Adults: United States, 2017-2018. NCHS Data Brief 2020 Feb:(360):1–8.
2. Pais R, et al. NAFLD and liver transplantation: Current burden and expected challenges. J Hepatol. 2016 Dec. doi: 10.1016/j.jhep.2016.07.033.
3. Lauby-Secretan B, et al. Body Fatness and Cancer--Viewpoint of the IARC Working Group. N Engl J Med. 2016 Aug. doi: 10.1056/NEJMsr1606602.
4. Kim A. Dysbiosis: A Review Highlighting Obesity and Inflammatory Bowel Disease. J Clin Gastroenterol. 2015 Nov-Dec. doi: 10.1097/MCG.0000000000000356.
5. Singh S, et al. Obesity in IBD: epidemiology, pathogenesis, disease course and treatment outcomes. Nat Rev Gastroenterol Hepatol. 2017 Feb. doi: 10.1038/nrgastro.2016.181.
6. Sundararaman L, Goudra B. Sedation for GI Endoscopy in the Morbidly Obese: Challenges and Possible Solutions. J Clin Med. 2024 Aug. doi: 10.3390/jcm13164635.
7. Bombassaro B, et al. The hypothalamus as the central regulator of energy balance and its impact on current and future obesity treatments. Arch Endocrinol Metab. 2024 Nov. doi: 10.20945/2359-4292-2024-0082.
8. Beccuti G, Pannain S. Sleep and obesity. Curr Opin Clin Nutr Metab Care. 2011 Jul. doi: 10.1097/MCO.0b013e3283479109.
9. Desalermos A, et al. Effect of Obesogenic Medications on Weight-Loss Outcomes in a Behavioral Weight-Management Program. Obesity (Silver Spring). 2019 May. doi: 10.1002/oby.22444.
10. Lord MN, Noble EE. Hypothalamic cannabinoid signaling: Consequences for eating behavior. Pharmacol Res Perspect. 2024 Oct. doi: 10.1002/prp2.1251.
11. Farhana A, Rehman A. Metabolic Consequences of Weight Reduction. [Updated 2023 Jul 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan. Available from: https://www.ncbi.nlm.nih.gov/books/NBK572145/.
12. Rubino F, et al. Definition and diagnostic criteria of clinical obesity. Lancet Diabetes Endocrinol. 2025 Mar. doi: 10.1016/S2213-8587(24)00316-4.
13. Cox CE. Role of Physical Activity for Weight Loss and Weight Maintenance. Diabetes Spectr. 2017 Aug. doi: 10.2337/ds17-0013.
14. Chaput JP, et al. Widespread misconceptions about obesity. Can Fam Physician. 2014 Nov. PMID: 25392431.
15. Muscogiuri G, et al. Mediterranean Diet and Obesity-related Disorders: What is the Evidence? Curr Obes Rep. 2022 Dec. doi: 10.1007/s13679-022-00481-1.
16. Gudzune KA, Kushner RF. Medications for Obesity: A Review. JAMA. 2024 Aug. doi: 10.1001/jama.2024.10816.
17. Wilding JPH, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021 Feb. doi: 10.1056/NEJMoa2032183.
18. Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022 Jun. doi: 10.1056/NEJMoa2206038.
19. Chiang CH, et al. Glucagon-Like Peptide-1 Receptor Agonists and Gastrointestinal Adverse Events: A Systematic Review and Meta-Analysis. Gastroenterology. 2025 Nov. doi: 10.1053/j.gastro.2025.06.003.
20. Aderinto N, et al. Recent advances in bariatric surgery: a narrative review of weight loss procedures. Ann Med Surg (Lond). 2023 Nov. doi: 10.1097/MS9.0000000000001472.
21. Chandan S, et al. Risk of De Novo Barrett’s Esophagus Post Sleeve Gastrectomy: A Systematic Review and Meta-Analysis of Studies With Long-Term Follow-Up. Clin Gastroenterol Hepatol. 2025 Jan. doi: 10.1016/j.cgh.2024.06.041.
22. Jirapinyo P, et al. American Society for Gastrointestinal Endoscopy-European Society of Gastrointestinal Endoscopy guideline on primary endoscopic bariatric and metabolic therapies for adults with obesity. Gastrointest Endosc. 2024 Jun. doi: 10.1016/j.gie.2023.12.004.
23. Nduma BN, et al. Endoscopic Gastric Sleeve: A Review of Literature. Cureus. 2023 Mar. doi: 10.7759/cureus.36353.
24. Abu Dayyeh BK, et al. Endoscopic sleeve gastroplasty for treatment of class 1 and 2 obesity (MERIT): a prospective, multicentre, randomised trial. Lancet. 2022 Aug. doi: 10.1016/S0140-6736(22)01280-6.
25. Gala K, et al. Outcomes of concomitant antiobesity medication use with endoscopic sleeve gastroplasty in clinical US settings. Obes Pillars. 2024 May. doi: 10.1016/j.obpill.2024.100112.
26. Chung CS, et al. Endoscopic sleeve gastroplasty combined with anti-obesity medication for better control of weight and diabetes. Clin Endosc. 2025 May. doi: 10.5946/ce.2024.274.
Factors Influencing Outcomes of a Telehealth-Based Physical Activity Program in Older Veterans Postdischarge
Factors Influencing Outcomes of a Telehealth-Based Physical Activity Program in Older Veterans Postdischarge
Deconditioning among hospitalized older adults contributes to significant decline in posthospitalization functional ability, physical performance, and physical activity.1-10 Previous hospital-to-home interventions have targeted improving function and physical activity, including recent programs leveraging home telehealth as a feasible and potentially effective mode of delivering in-home exercise and rehabilitation.11-14 However, pilot interventions have shown mixed effectiveness.11,12,14 This study expands on a previously published intervention describing a pilot home telehealth program for veterans posthospital discharge that demonstrated significant 6-month improvement in physical activity as well as trends in physical function improvement, including among those with cognitive impairment.15 Factors that contribute to improved outcomes are the focus of the present study.
Key factors underlying the complexity of hospital-to-home transitions include hospitalization elements (ie, reason for admission and length of stay), associated posthospital syndromes (ie, postdischarge falls, medication changes, cognitive impairment, and pain), and postdischarge health care application (ie, physical therapy and hospital readmission).16-18 These factors may be associated with postdischarge functional ability, physical performance, and physical activity, but their direct influence on intervention outcomes is unclear (Figure 1).5,7,9,16-20 The objective of this study was to examine the influence of hospitalization, posthospital syndrome, and postdischarge health care application factors on outcomes of a US Department of Veterans Affairs (VA) Video Connect (VVC) intervention to enhance function and physical activity in older adults posthospital discharge.
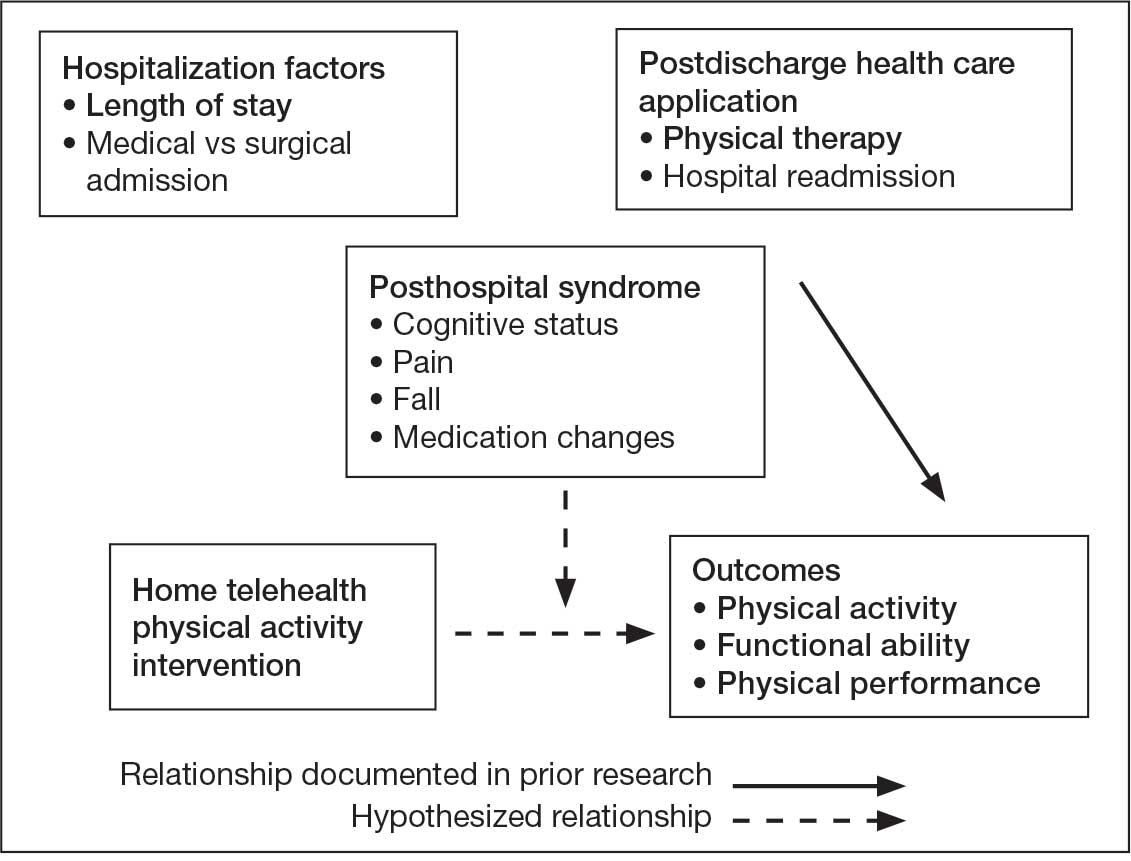
health care application factors on physical activity, functional ability, and
physical performance intervention outcomes.
Methods
The previous analysis reported on patient characteristics, program feasibility, and preliminary outcomes.13,15 The current study reports on relationships between hospitalization, posthospital syndrome, and postdischarge health care application factors and change in key outcomes, namely postdischarge self-reported functional ability, physical performance, and physical activity from baseline to endpoint.
Participants provided written informed consent. The protocol and consent forms were approved by the VA Ann Arbor Healthcare System (VAAAHS) Research and Development Committee, and the project was registered on clinicaltrials.gov (NCT04045054).
Intervention
The pilot program targeted older adults following recent hospital discharge from VAAAHS. Participants were eligible if they were aged ≥ 50 years, had been discharged following an inpatient stay in the past 1 to 2 weeks, evaluated by physical therapy during hospitalization with stated rehabilitation goals on discharge, and followed by a VAAAHS primary care physician. Participants were either recruited during hospital admission or shortly after discharge.13
An experienced physical activity trainer (PAT) supported the progression of participants’ rehabilitation goals via a home exercise program and coached the patient and caregiver to optimize functional ability, physical performance, and physical activity. The PAT was a nonlicensed research assistant with extensive experience in applying standard physical activity enhancement protocols (eg, increased walking) to older adults with comorbidities. Participation in the program lasted about 6 months. Initiation of the PAT program was delayed if the patient was already receiving postdischarge home-based or outpatient physical therapy. The PAT contacted the patient weekly via VVC for the first 6 weeks, then monthly for a total of 6 months. Each contact included information on optimal walking form, injury prevention, program progression, and ways to incorporate sit-to-stand transitions, nonsitting behavior, and walking into daily routines. The initial VVC contact lasted about 60 minutes and subsequent sessions lasted about 30 minutes.13
Demographic characteristics were self-reported by participants and included age, sex, race, years of education, and marital status. Clinical characteristics were obtained from each participant’s electronic health record (EHR), including copay status, index hospitalization length of stay, admission diagnosis, and postsurgery status (postsurgery vs nonpostsurgery). Intervention adherence was tracked as the number of PAT sessions attended.
Posthospital Syndrome Factors
Participant falls (categorized as those who reported a fall vs those who did not) and medication changes (number of changes reported, including new medication, discontinued medication, dose changes, medication changes, or changes in medication schedule) were reported by participants or caregivers during each VVC contact. Participants completed the Montreal Cognitive Assessment (MoCA) at baseline, and were dichotomized into 2 groups: no cognitive impairment (MoCA score ≥ 26) and mild to moderate cognitive impairment (MoCA score 10-25).13,21
Participants rated how much pain interfered with their normal daily activities since the previous VVC session on a 5-point Likert scale (1, not at all; to 5, extremely).22 Similar to prior research, participants were placed into 2 groups based on their mean pain interference score (individuals with scores from 1.0 to 2.0 in 1 group, and individuals with > 2.0 in another).23-25 Participants were separated into a no or mild pain interference group and a moderate to severe pain interference group. Hospital readmissions (VA and non-VA) and postdischarge physical therapy outcomes were obtained from the participant’s EHR, including primary care visits.
Outcomes
Outcomes were collected at baseline (posthospital discharge) and 6 months postenrollment.
Self-Reported Functional Ability. This measure is provided by participants or caregivers and measured by the Katz Index of Independence in Activities of Daily Living (ADL), Lawton and Brody Instrumental ADL Scale (IADL), Nagi Disability Model, and Rosow-Breslau Scale. The Katz ADL assesses the ability to complete 6 self-care activities and awards 1 point for independence and 0 if the individual is dependent (total score range, 0-6).26 The Lawton and Brody IADL measures an individual’s independence in 8 instrumental ADLs; it awards 1 point for independence and 0 if the individual is dependent (total score range, 0-8).27 The Nagi Disability Model evaluates an individual’s difficulty performing 5 tasks (total score range, 0-5) and tallies the number of items with a response other than “no difficulty at all” (higher total score indicates greater difficulty). 28 The Rosow-Breslau Scale is a 3-item measure of mobility disability; individual responses are 0 (no help) and 1 (requires help or unable); higher total score (range, 0-3) indicates greater disability.29
Physical Performance. Measured using the Short Physical Performance Battery (SPPB), which evaluates standing balance, sit to stand, and walking performance. Scores range from 0 to 4 on the balance, gait speed, and chair stand tests, for a total composite score between 0 and 12 (higher score indicates better performance).30
Physical Activity. Measured using actigraphy, namely a physical activity monitor adherent to the thigh (activ-PAL3TM, PAL Technologies Ltd., Glasgow, UK).31 Participants were instructed to wear the activPal for ≥ 1 week. Participants with a minimum of 5 days of wear were included in this analysis.
Data Analyses
Analyses were performed using SPSS software version 29.0.32 Continuous variables were summarized using mean (SD) or median and IQR using the weighted average method; categorical variables were summarized using frequencies and percentages. Baseline scores on outcome variables were compared by categorical hospitalization, posthospital syndrome, and postdischarge health care application factor variables using Mann-Whitney U tests. The differences between outcome variables from baseline to endpoint were then calculated to produce change scores. Relationships between the number of PAT sessions attended and baseline outcomes and outcome change scores were estimated using Spearman correlations. Relationships between categorical factors (hospitalization, posthospital syndrome, and postdischarge health care application) and outcome variable change scores (which were normally distributed) were examined using Mann-Whitney U tests. Relationships with continuous hospitalization (length of stay) and posthospital syndrome factors (medication changes) were estimated using Spearman correlations. Effect sizes (ES) were estimated with Cohen d; small (d = 0.2), medium (d = 0.5), or large (d ≥ 0.8). Missing data were handled using pairwise deletion.33 Therefore, sample sizes were reported for each analysis. For all statistical tests, P < .05 was considered significant.
Results
Twenty-four individuals completed the pilot intervention.15 Mean (SD) age was 73.6 (8.1) years (range, 64-93 years) and participants were predominantly White males (Table 1). Eight participants had a high school education only and 13 had more than a high school education. Diagnoses at admission included 9 patients with orthopedic/musculoskeletal conditions (6 were for joint replacement), 6 patients with vascular/pulmonary conditions, and 4 with gastrointestinal/renal/urological conditions. Of the 11 postsurgery participants, 7 were orthopedic, 4 were gastrointestinal, and 1 was peripheral vascular.
Baseline outcome scores did not differ significantly between groups, except individuals with moderate to severe pain interference reported a significantly lower IADL score (median [IQR] 4 [2-7]) than individuals with mild or moderate pain interference (median [IQR] 8 [7-8]; P = .02) (Table 2). The mean (SD) number of PAT sessions attended was 9.3 (3.7) (range, 3-19). There were no significant relationships between number of sessions attended and any baseline outcome variables or outcome change scores.
Hospitalization Factors
Participants who were postsurgery tended to have greater improvement than individuals who were nonpostsurgery in ADLs (median [IQR] 0 [0-1.5]; ES, 0.6; P = .10) and SPPB (median [IQR] 2 [1.5-9]; ES, 0.9; P = .07), but the improvements were not statistically significant (Table 3). Mean (SD) length of stay of the index hospitalization was 6.7 (6.1) days. Longer length of stay was significantly correlated with an increase in Nagi score (ρ, 0.45; 95% CI, 0.01-0.75). There were no other significant or trending relationships between length of stay and outcome variables.
Posthospital Syndrome Factors
The 16 participants with mild to moderate cognitive impairment had less improvement in ADLs (median [IQR] 0 [0-1]) than the 8 participants with no impairment (median [IQR] 0 [-0.75 to 0]; ES, -1.1; P = .04). Change in outcome variables from baseline to endpoint did not significantly differ between the 8 patients who reported a fall compared with the 13 who did not, nor were any trends observed. Change in outcome variables from baseline to endpoint also did not significantly differ between the 8 participants who reported no or mild pain interference compared with the 10 patients with moderate to severe pain interference, nor were any trends observed. Mean (SD) number of medication changes was 2.5 (1.6). Higher number of medication changes was significantly correlated with a decrease in Rosow-Breslau score (ρ, -0.47; 95% CI, -0.76 to -0.02). There were no other significant or trending relationships between number of medication changes and outcome variables.
Postdischarge Health Care Application Factors
The 16 participants who attended posthospital physical therapy trended towards less improvement in IADLs (median [IQR] 0 [-0.5 to 1.5]; ES, -0.7; P = .11) and SPPB (median [IQR] 2 [-3.0 to 4.5]; ES, -0.5; P = .15) than the 8 patients with no postdischarge physical therapy. Eleven participants were readmitted, while 13 had no readmissions in their medical records between baseline and endpoint. Participants with ≥ 1 readmission experienced a greater increase in Rosow-Breslau score (median [IQR] 0 [-0.5 to 1.0]) than those not readmitted (median [IQR] 0 [-1.25 to 0.25]; ES, 1.0; P = .03). Borderline greater improvement in number of steps was found in those not readmitted (median [IQR] 3365.6 [274.4-7710.9]) compared with those readmitted (median [IQR] 319.9 [-136.1 to 774.5]; ES, -1.3; P = .05). Patients who were readmitted also tended to have lower and not statistically significant improvements in SPPB (median [IQR] 1 [-4.0 to 5.3]) compared with those not readmitted (median [IQR] 2 [0.3-3.8]; ES, -0.5; P = .17) (Table 3).
Discussion
This study examined the association between hospitalization, posthospital syndrome, and postdischarge health care use in patients undergoing a VVC-based intervention following hospital discharge. Participants who had no or mild cognitive impairment, no readmissions, higher medication changes, and a shorter hospital length of stay tended to experience lower disability, including in mobility and ADLs. This suggests individuals who are less clinically complex may be more likely to benefit from this type of virtual rehabilitation program. These findings are consistent with clinical experiences; home-based programs to improve physical activity posthospital discharge can be challenging for those who were medically ill (and did not undergo a specific surgical procedure), cognitively impaired, and become acutely ill and trigger hospital readmission. 15 For example, the sample in this study had higher rates of falls, pain, and readmissions compared to previous research.2,3,34-39
The importance of posthospital syndrome in the context of recovery of function and health at home following hospitalization is well documented.16-18 The potential impact of posthospital syndrome on physical activity-focused interventions is less understood. In our analysis, participants with mild or moderate cognitive impairment tended to become more dependent in their ADLs, while those with no cognitive impairment tended to become more independent in their ADLs. This functional decline over time is perhaps expected in persons with cognitive impairment, but the significant difference with a large ES warrants further consideration on how to tailor interventions to better promote functional recovery in these individuals.40,41 While some cognitive decline may not be preventable, this finding supports the need to promote healthy cognitive aging, identify declines in cognition, and work to mitigate additional decline. Programs specifically designed to promote function and physical activity in older adults with cognitive impairment are needed, especially during care transitions.41-43
While participants reported that falls and pain interference did not have a significant impact on change in outcomes between baseline and endpoint, these areas need further investigation. Falls and pain have been associated with function and physical activity in older adults.42-46 Pain is common, yet underappreciated during older adult hospital-to-home transitions.11,12,45,46 There is a need for more comprehensive assessment of pain (including pain intensity) and qualitative research.
Hospitalization and postdischarge health care application factors may have a significant impact on home-telehealth physical activity intervention success. Individuals who were postsurgery tended to have greater improvements in ADLs and physical performance. Most postsurgery participants had joint replacement surgery. Postsurgery status may not be modifiable, but it is important to note expected differences in recovery between medical and surgical admissions and the need to tailor care based on admission diagnosis. Those with a longer length of hospital stay may be considered at higher risk of suboptimal outcomes postdischarge, which indicates an opportunity for targeting resources and support, in addition to efforts of reducing length of stay where possible.47
Readmissions were significantly related to a change in Rosow-Breslau mobility disability score. This may indicate the detrimental impact a readmission can have on increasing mobility and physical activity postdischarge, or the potential of this pilot program to impact readmissions by increasing mobility and physical activity, contrary to prior physical exercise interventions.5,7,9,48 With 5% to 79% of readmissions considered preventable, continued efforts and program dissemination and implementation to address preventable readmissions are warranted.49 Individuals with postdischarge physical therapy (prior to beginning the pilot program) tended to demonstrate less improvement in disability and physical performance. This relationship needs further investigation; the 2 groups did not appear to have significant differences at baseline, albeit with a small sample size. It is possible they experienced initial improvements with postdischarge physical therapy and plateaued or had little further reserve to improve upon entering the VVC program.
Strengths and Limitations
This pilot program provided evaluative data on the use of VVC to enhance function and physical activity in older adults posthospital discharge. It included individual (eg, fall, pain, cognitive impairment) and health service (eg, readmission, physical therapy) level factors as predictors of function and physical activity posthospitalization.5,7,9,15-19
The results of this pilot project stem from a small sample lacking diversity in terms of race, ethnicity, and sex. There was some variation in baseline and endpoints between participants, and when hospitalization, posthospital syndrome, and postdischarge health care application factors were collected. The majority of participants were recruited within a month postdischarge, and the program lasted about 6 months. Data collection was attempted at regular PAT contacts, but there was some variation in when visits occurred based on participant availability and preference. Some participants had missing data, which was handled using pairwise deletion.33 Larger studies are needed to confirm the findings of this study, particularly the trends that did not reach statistical significance. Home health services other than physical therapy (eg, nursing, occupational therapy) were not fully accounted for and should be considered in future research.
Conclusions
In patients undergoing a 6-month pilot VVC-based physical activity intervention posthospital discharge, improvements in mobility and disability were most likely in those who had no cognitive impairment and were not readmitted. Larger sample and qualitative investigations are necessary to optimize outcomes for patients who meet these clinical profiles.
- Liebzeit D, Bratzke L, Boltz M, Purvis S, King B. Getting back to normal: a grounded theory study of function in post-hospitalized older adults. Gerontologist. 2020;60:704-714. doi:10.1093/geront/gnz057
- Ponzetto M, Zanocchi M, Maero B, et al. Post-hospitalization mortality in the elderly. Arch Gerontol Geriatr. 2003;36:83-91. doi:10.1016/s0167-4943(02)00061-4
- Buurman BM, Hoogerduijn JG, de Haan RJ, et al. Geriatric conditions in acutely hospitalized older patients: prevalence and one-year survival and functional decline. PLoS One. 2011;6:e26951. doi:10.1371/journal.pone.0026951
- Ponzetto M, Maero B, Maina P, et al. Risk factors for early and late mortality in hospitalized older patients: the continuing importance of functional status. J Gerontol A Biol Sci Med Sci. 2003;58:1049-1054. doi:10.1093/gerona/58.11.m1049
- Huang HT, Chang CM, Liu LF, Lin HS, Chen CH. Trajectories and predictors of functional decline of hospitalised older patients. J Clin Nurs. 2013;22:1322-1331. doi:10.1111/jocn.12055
- Boyd CM, Landefeld CS, Counsell SR, et al. Recovery of activities of daily living in older adults after hospitalization for acute medical illness. J Am Geriatr Soc. 2008;56:2171- 2179. doi:10.1111/j.1532-5415.2008.02023.x
- Helvik AS, Selbæk G, Engedal K. Functional decline in older adults one year after hospitalization. Arch Gerontol Geriatr. 2013;57:305-310. doi:10.1016/j.archger.2013.05.008
- Zaslavsky O, Zisberg A, Shadmi E. Impact of functional change before and during hospitalization on functional recovery 1 month following hospitalization. J Gerontol Biol Sci Med Sci. 2015;70:381-386. doi:10.1093/gerona/glu168
- Chen CC, Wang C, Huang GH. Functional trajectory 6 months posthospitalization: a cohort study of older hospitalized patients in Taiwan. Nurs Res. 2008;57:93-100. doi:10.1097/01.NNR.0000313485.18670.e2
- Kleinpell RM, Fletcher K, Jennings BM. Reducing functional decline in hospitalized elderly. In: Hughes RG, ed. Patient Safety and Quality: An Evidence-Based Handbook for Nurses. Agency for Healthcare Research and Quality (US); 2008. Accessed September 3, 2025. http://www.ncbi.nlm.nih.gov/books/NBK2629/
- Liebzeit D, Rutkowski R, Arbaje AI, Fields B, Werner NE. A scoping review of interventions for older adults transitioning from hospital to home. J Am Geriatr Soc. 2021;69:2950-2962. doi:10.1111/jgs.17323
- Hladkowicz E, Dumitrascu F, Auais M, et al. Evaluations of postoperative transitions in care for older adults: a scoping review. BMC Geriatr. 2022;22:329. doi:10.1186/s12877-022-02989-6
- Alexander NB, Phillips K, Wagner-Felkey J, et al. Team VA Video Connect (VVC) to optimize mobility and physical activity in post-hospital discharge older veterans: baseline assessment. BMC Geriatr. 2021;21:502. doi:10.1186/s12877-021-02454-w
- Dawson R, Oliveira JS, Kwok WS, et al. Exercise interventions delivered through telehealth to improve physical functioning for older adults with frailty, cognitive, or mobility disability: a systematic review and meta-analysis. Telemed J E Health. 2024;30:940-950. doi:10.1089/tmj.2023.0177
- Liebzeit D, Phillips KK, Hogikyan RV, Cigolle CT, Alexander NB. A pilot home-telehealth program to enhance functional ability, physical performance, and physical activity in older adult veterans post-hospital discharge. Res Gerontol Nurs. 2024;17:271-279. doi:10.3928/19404921-20241105-01
- Krumholz HM. Post-hospital syndrome—an acquired, transient condition of generalized risk. N Engl J Med. 2013;368:100-102. doi:10.1056/NEJMp1212324
- Caraballo C, Dharmarajan K, Krumholz HM. Post hospital syndrome: is the stress of hospitalization causing harm? Rev Esp Cardiol (Engl Ed). 2019;72:896-898. doi:10.1016/j.rec.2019.04.010
- Rawal S, Kwan JL, Razak F, et al. Association of the trauma of hospitalization with 30-day readmission or emergency department visit. JAMA Intern Med. 2019;179:38- 45. doi:10.1001/jamainternmed.2018.5100
- Dutzi I, Schwenk M, Kirchner M, Jooss E, Bauer JM, Hauer K. Influence of cognitive impairment on rehabilitation received and its mediating effect on functional recovery. J Alzheimers Dis. 2021;84:745-756. doi:10.3233/JAD-210620
- Uriz-Otano F, Uriz-Otano JI, Malafarina V. Factors associated with short-term functional recovery in elderly people with a hip fracture. Influence ofcognitiveimpairment. JAmMedDirAssoc. 2015;16:215-220. doi:10.1016/j.jamda.2014.09.009
- Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695-699. doi:10.1111/j.1532-5415.2005.53221.x
- Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30:473-483.
- White RS, Jiang J, Hall CB, et al. Higher perceived stress scale scores are associated with higher pain intensity and pain interference levels in older adults. J Am Geriatr Soc. 2014;62:2350-2356. doi:10.1111/jgs.13135
- Blyth FM, March LM, Brnabic AJ, et al. Chronic pain in Australia: a prevalence study. Pain. 2001;89:127-134. doi:10.1016/s0304-3959(00)00355-9
- Thomas E, Peat G, Harris L, Wilkie R, Croft PR. The prevalence of pain and pain interference in a general population of older adults: cross-sectional findings from the North Staffordshire Osteoarthritis Project (NorStOP). Pain. 2004;110:361-368. doi:10.1016/j.pain.2004.04.017
- Katz S, Ford AB, Moskowitz RW, Jackson BA, Jaffe MW. Studies of illness in the aged. The index of ADL: a standardized measure of biological and psychosocial function. JAMA. 1963;185:914-919. doi:10.1001/jama.1963.03060120024016
- Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. 1969;9:179-186.
- Alexander NB, Guire KE, Thelen DG, et al. Self-reported walking ability predicts functional mobility performance in frail older adults. J Am Geriatr Soc. 2000;48:1408-1413. doi:10.1111/j.1532-5415.2000.tb02630.x
- Rosow I, Breslau N. A Guttman health scale for the aged. J Gerontol. 1966;21:556-559. doi:10.1093/geronj/21.4.556
- Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49:M85-M94. doi:10.1093/geronj/49.2.m85
- Chan CS, Slaughter SE, Jones CA, Ickert C, Wagg AS. Measuring activity performance of older adults using the activPAL: a rapid review. Healthcare (Basel). 2017;5:94. doi:10.3390/healthcare5040094
- IBM SPSS software. IBM Corp; 2019. Accessed September 3, 2025. https://www.ibm.com/spss
- Kang H. The prevention and handling of the missing data. Korean J Anesthesiol. 2013;64:402-406. doi:10.4097/kjae.2013.64.5.402
- Epstein AM, Jha AK, Orav EJ. The relationship between hospital admission rates and rehospitalizations. N Engl J Med. 2011;365:2287-2295. doi:10.1056/NEJMsa1101942
- Bogaisky M, Dezieck L. Early hospital readmission of nursing home residents and community-dwelling elderly adults discharged from the geriatrics service of an urban teaching hospital: patterns and risk factors. J Am Geriatr Soc. 2015;63:548-552. doi:10.1111/jgs.13317
- Jencks SF, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009;360:1418-1428. doi:10.1056/NEJMsa0803563
- Hoyer EH, Needham DM, Atanelov L, Knox B, Friedman M, Brotman DJ. Association of impaired functional status at hospital discharge and subsequent rehospitalization. J Hosp Med. 2014;9:277-282. doi:10.1002/jhm.2152
- Mahoney J, Sager M, Dunham NC, Johnson J. Risk of falls after hospital discharge. J Am Geriatr Soc. 1994;42:269- 274. doi:10.1111/j.1532-5415.1994.tb01750.x
- Hoffman GJ, Liu H, Alexander NB, Tinetti M, Braun TM, Min LC. Posthospital fall injuries and 30-day readmissions in adults 65 years and older. JAMA Netw Open. 2019;2:e194276. doi:10.1001/jamanetworkopen.2019.4276
- Gill DP, Hubbard RA, Koepsell TD, et al. Differences in rate of functional decline across three dementia types. Alzheimers Dement. 2013;9:S63-S71. doi:10.1016/j.jalz.2012.10.010
- Auyeung TW, Kwok T, Lee J, Leung PC, Leung J, Woo J. Functional decline in cognitive impairment–the relationship between physical and cognitive function. Neuroepidemiology. 2008;31:167-173. doi:10.1159/000154929
- Patti A, Zangla D, Sahin FN, et al. Physical exercise and prevention of falls. Effects of a Pilates training method compared with a general physical activity program. Medicine (Baltimore). 2021;100:e25289. doi:10.1097/MD.0000000000025289
- Nagarkar A, Kulkarni S. Association between daily activities and fall in older adults: an analysis of longitudinal ageing study in India (2017-18). BMC Geriatr. 2022;22:203. doi:10.1186/s12877-022-02879-x
- Ek S, Rizzuto D, Xu W, Calderón-Larrañaga A, Welmer AK. Predictors for functional decline after an injurious fall: a population-based cohort study. Aging Clin Exp Res. 2021;33:2183-2190. doi:10.1007/s40520-020-01747-1
- Dagnino APA, Campos MM. Chronic pain in the elderly: mechanisms and perspectives. Front Hum Neurosci. 2022;16:736688. doi:10.3389/fnhum.2022.736688
- Ritchie CS, Patel K, Boscardin J, et al. Impact of persistent pain on function, cognition, and well-being of older adults. J Am Geriatr Soc. 2023;71:26-35. doi:10.1111/jgs.18125
- Han TS, Murray P, Robin J, et al. Evaluation of the association of length of stay in hospital and outcomes. Int J Qual Health Care. 2022;34:mzab160. doi:10.1093/intqhc/ mzab160
- Lærum-Onsager E, Molin M, Olsen CF, et al. Effect of nutritional and physical exercise intervention on hospital readmission for patients aged 65 or older: a systematic review and meta-analysis of randomized controlled trials. Int J Behav Nutr Phys Act. 2021;18:62. doi:10.1186/s12966-021-01123-w
- Van Walraven C, Bennett C, Jennings A, Austin PC, Forster AJ. Proportion of hospital readmissions deemed avoidable: a systematic review. CMAJ. 2011;183:E391-E402. doi:10.1503/cmaj.101860
Deconditioning among hospitalized older adults contributes to significant decline in posthospitalization functional ability, physical performance, and physical activity.1-10 Previous hospital-to-home interventions have targeted improving function and physical activity, including recent programs leveraging home telehealth as a feasible and potentially effective mode of delivering in-home exercise and rehabilitation.11-14 However, pilot interventions have shown mixed effectiveness.11,12,14 This study expands on a previously published intervention describing a pilot home telehealth program for veterans posthospital discharge that demonstrated significant 6-month improvement in physical activity as well as trends in physical function improvement, including among those with cognitive impairment.15 Factors that contribute to improved outcomes are the focus of the present study.
Key factors underlying the complexity of hospital-to-home transitions include hospitalization elements (ie, reason for admission and length of stay), associated posthospital syndromes (ie, postdischarge falls, medication changes, cognitive impairment, and pain), and postdischarge health care application (ie, physical therapy and hospital readmission).16-18 These factors may be associated with postdischarge functional ability, physical performance, and physical activity, but their direct influence on intervention outcomes is unclear (Figure 1).5,7,9,16-20 The objective of this study was to examine the influence of hospitalization, posthospital syndrome, and postdischarge health care application factors on outcomes of a US Department of Veterans Affairs (VA) Video Connect (VVC) intervention to enhance function and physical activity in older adults posthospital discharge.

health care application factors on physical activity, functional ability, and
physical performance intervention outcomes.
Methods
The previous analysis reported on patient characteristics, program feasibility, and preliminary outcomes.13,15 The current study reports on relationships between hospitalization, posthospital syndrome, and postdischarge health care application factors and change in key outcomes, namely postdischarge self-reported functional ability, physical performance, and physical activity from baseline to endpoint.
Participants provided written informed consent. The protocol and consent forms were approved by the VA Ann Arbor Healthcare System (VAAAHS) Research and Development Committee, and the project was registered on clinicaltrials.gov (NCT04045054).
Intervention
The pilot program targeted older adults following recent hospital discharge from VAAAHS. Participants were eligible if they were aged ≥ 50 years, had been discharged following an inpatient stay in the past 1 to 2 weeks, evaluated by physical therapy during hospitalization with stated rehabilitation goals on discharge, and followed by a VAAAHS primary care physician. Participants were either recruited during hospital admission or shortly after discharge.13
An experienced physical activity trainer (PAT) supported the progression of participants’ rehabilitation goals via a home exercise program and coached the patient and caregiver to optimize functional ability, physical performance, and physical activity. The PAT was a nonlicensed research assistant with extensive experience in applying standard physical activity enhancement protocols (eg, increased walking) to older adults with comorbidities. Participation in the program lasted about 6 months. Initiation of the PAT program was delayed if the patient was already receiving postdischarge home-based or outpatient physical therapy. The PAT contacted the patient weekly via VVC for the first 6 weeks, then monthly for a total of 6 months. Each contact included information on optimal walking form, injury prevention, program progression, and ways to incorporate sit-to-stand transitions, nonsitting behavior, and walking into daily routines. The initial VVC contact lasted about 60 minutes and subsequent sessions lasted about 30 minutes.13
Demographic characteristics were self-reported by participants and included age, sex, race, years of education, and marital status. Clinical characteristics were obtained from each participant’s electronic health record (EHR), including copay status, index hospitalization length of stay, admission diagnosis, and postsurgery status (postsurgery vs nonpostsurgery). Intervention adherence was tracked as the number of PAT sessions attended.
Posthospital Syndrome Factors
Participant falls (categorized as those who reported a fall vs those who did not) and medication changes (number of changes reported, including new medication, discontinued medication, dose changes, medication changes, or changes in medication schedule) were reported by participants or caregivers during each VVC contact. Participants completed the Montreal Cognitive Assessment (MoCA) at baseline, and were dichotomized into 2 groups: no cognitive impairment (MoCA score ≥ 26) and mild to moderate cognitive impairment (MoCA score 10-25).13,21
Participants rated how much pain interfered with their normal daily activities since the previous VVC session on a 5-point Likert scale (1, not at all; to 5, extremely).22 Similar to prior research, participants were placed into 2 groups based on their mean pain interference score (individuals with scores from 1.0 to 2.0 in 1 group, and individuals with > 2.0 in another).23-25 Participants were separated into a no or mild pain interference group and a moderate to severe pain interference group. Hospital readmissions (VA and non-VA) and postdischarge physical therapy outcomes were obtained from the participant’s EHR, including primary care visits.
Outcomes
Outcomes were collected at baseline (posthospital discharge) and 6 months postenrollment.
Self-Reported Functional Ability. This measure is provided by participants or caregivers and measured by the Katz Index of Independence in Activities of Daily Living (ADL), Lawton and Brody Instrumental ADL Scale (IADL), Nagi Disability Model, and Rosow-Breslau Scale. The Katz ADL assesses the ability to complete 6 self-care activities and awards 1 point for independence and 0 if the individual is dependent (total score range, 0-6).26 The Lawton and Brody IADL measures an individual’s independence in 8 instrumental ADLs; it awards 1 point for independence and 0 if the individual is dependent (total score range, 0-8).27 The Nagi Disability Model evaluates an individual’s difficulty performing 5 tasks (total score range, 0-5) and tallies the number of items with a response other than “no difficulty at all” (higher total score indicates greater difficulty). 28 The Rosow-Breslau Scale is a 3-item measure of mobility disability; individual responses are 0 (no help) and 1 (requires help or unable); higher total score (range, 0-3) indicates greater disability.29
Physical Performance. Measured using the Short Physical Performance Battery (SPPB), which evaluates standing balance, sit to stand, and walking performance. Scores range from 0 to 4 on the balance, gait speed, and chair stand tests, for a total composite score between 0 and 12 (higher score indicates better performance).30
Physical Activity. Measured using actigraphy, namely a physical activity monitor adherent to the thigh (activ-PAL3TM, PAL Technologies Ltd., Glasgow, UK).31 Participants were instructed to wear the activPal for ≥ 1 week. Participants with a minimum of 5 days of wear were included in this analysis.
Data Analyses
Analyses were performed using SPSS software version 29.0.32 Continuous variables were summarized using mean (SD) or median and IQR using the weighted average method; categorical variables were summarized using frequencies and percentages. Baseline scores on outcome variables were compared by categorical hospitalization, posthospital syndrome, and postdischarge health care application factor variables using Mann-Whitney U tests. The differences between outcome variables from baseline to endpoint were then calculated to produce change scores. Relationships between the number of PAT sessions attended and baseline outcomes and outcome change scores were estimated using Spearman correlations. Relationships between categorical factors (hospitalization, posthospital syndrome, and postdischarge health care application) and outcome variable change scores (which were normally distributed) were examined using Mann-Whitney U tests. Relationships with continuous hospitalization (length of stay) and posthospital syndrome factors (medication changes) were estimated using Spearman correlations. Effect sizes (ES) were estimated with Cohen d; small (d = 0.2), medium (d = 0.5), or large (d ≥ 0.8). Missing data were handled using pairwise deletion.33 Therefore, sample sizes were reported for each analysis. For all statistical tests, P < .05 was considered significant.
Results
Twenty-four individuals completed the pilot intervention.15 Mean (SD) age was 73.6 (8.1) years (range, 64-93 years) and participants were predominantly White males (Table 1). Eight participants had a high school education only and 13 had more than a high school education. Diagnoses at admission included 9 patients with orthopedic/musculoskeletal conditions (6 were for joint replacement), 6 patients with vascular/pulmonary conditions, and 4 with gastrointestinal/renal/urological conditions. Of the 11 postsurgery participants, 7 were orthopedic, 4 were gastrointestinal, and 1 was peripheral vascular.

Baseline outcome scores did not differ significantly between groups, except individuals with moderate to severe pain interference reported a significantly lower IADL score (median [IQR] 4 [2-7]) than individuals with mild or moderate pain interference (median [IQR] 8 [7-8]; P = .02) (Table 2). The mean (SD) number of PAT sessions attended was 9.3 (3.7) (range, 3-19). There were no significant relationships between number of sessions attended and any baseline outcome variables or outcome change scores.

Hospitalization Factors
Participants who were postsurgery tended to have greater improvement than individuals who were nonpostsurgery in ADLs (median [IQR] 0 [0-1.5]; ES, 0.6; P = .10) and SPPB (median [IQR] 2 [1.5-9]; ES, 0.9; P = .07), but the improvements were not statistically significant (Table 3). Mean (SD) length of stay of the index hospitalization was 6.7 (6.1) days. Longer length of stay was significantly correlated with an increase in Nagi score (ρ, 0.45; 95% CI, 0.01-0.75). There were no other significant or trending relationships between length of stay and outcome variables.

Posthospital Syndrome Factors
The 16 participants with mild to moderate cognitive impairment had less improvement in ADLs (median [IQR] 0 [0-1]) than the 8 participants with no impairment (median [IQR] 0 [-0.75 to 0]; ES, -1.1; P = .04). Change in outcome variables from baseline to endpoint did not significantly differ between the 8 patients who reported a fall compared with the 13 who did not, nor were any trends observed. Change in outcome variables from baseline to endpoint also did not significantly differ between the 8 participants who reported no or mild pain interference compared with the 10 patients with moderate to severe pain interference, nor were any trends observed. Mean (SD) number of medication changes was 2.5 (1.6). Higher number of medication changes was significantly correlated with a decrease in Rosow-Breslau score (ρ, -0.47; 95% CI, -0.76 to -0.02). There were no other significant or trending relationships between number of medication changes and outcome variables.
Postdischarge Health Care Application Factors
The 16 participants who attended posthospital physical therapy trended towards less improvement in IADLs (median [IQR] 0 [-0.5 to 1.5]; ES, -0.7; P = .11) and SPPB (median [IQR] 2 [-3.0 to 4.5]; ES, -0.5; P = .15) than the 8 patients with no postdischarge physical therapy. Eleven participants were readmitted, while 13 had no readmissions in their medical records between baseline and endpoint. Participants with ≥ 1 readmission experienced a greater increase in Rosow-Breslau score (median [IQR] 0 [-0.5 to 1.0]) than those not readmitted (median [IQR] 0 [-1.25 to 0.25]; ES, 1.0; P = .03). Borderline greater improvement in number of steps was found in those not readmitted (median [IQR] 3365.6 [274.4-7710.9]) compared with those readmitted (median [IQR] 319.9 [-136.1 to 774.5]; ES, -1.3; P = .05). Patients who were readmitted also tended to have lower and not statistically significant improvements in SPPB (median [IQR] 1 [-4.0 to 5.3]) compared with those not readmitted (median [IQR] 2 [0.3-3.8]; ES, -0.5; P = .17) (Table 3).
Discussion
This study examined the association between hospitalization, posthospital syndrome, and postdischarge health care use in patients undergoing a VVC-based intervention following hospital discharge. Participants who had no or mild cognitive impairment, no readmissions, higher medication changes, and a shorter hospital length of stay tended to experience lower disability, including in mobility and ADLs. This suggests individuals who are less clinically complex may be more likely to benefit from this type of virtual rehabilitation program. These findings are consistent with clinical experiences; home-based programs to improve physical activity posthospital discharge can be challenging for those who were medically ill (and did not undergo a specific surgical procedure), cognitively impaired, and become acutely ill and trigger hospital readmission. 15 For example, the sample in this study had higher rates of falls, pain, and readmissions compared to previous research.2,3,34-39
The importance of posthospital syndrome in the context of recovery of function and health at home following hospitalization is well documented.16-18 The potential impact of posthospital syndrome on physical activity-focused interventions is less understood. In our analysis, participants with mild or moderate cognitive impairment tended to become more dependent in their ADLs, while those with no cognitive impairment tended to become more independent in their ADLs. This functional decline over time is perhaps expected in persons with cognitive impairment, but the significant difference with a large ES warrants further consideration on how to tailor interventions to better promote functional recovery in these individuals.40,41 While some cognitive decline may not be preventable, this finding supports the need to promote healthy cognitive aging, identify declines in cognition, and work to mitigate additional decline. Programs specifically designed to promote function and physical activity in older adults with cognitive impairment are needed, especially during care transitions.41-43
While participants reported that falls and pain interference did not have a significant impact on change in outcomes between baseline and endpoint, these areas need further investigation. Falls and pain have been associated with function and physical activity in older adults.42-46 Pain is common, yet underappreciated during older adult hospital-to-home transitions.11,12,45,46 There is a need for more comprehensive assessment of pain (including pain intensity) and qualitative research.
Hospitalization and postdischarge health care application factors may have a significant impact on home-telehealth physical activity intervention success. Individuals who were postsurgery tended to have greater improvements in ADLs and physical performance. Most postsurgery participants had joint replacement surgery. Postsurgery status may not be modifiable, but it is important to note expected differences in recovery between medical and surgical admissions and the need to tailor care based on admission diagnosis. Those with a longer length of hospital stay may be considered at higher risk of suboptimal outcomes postdischarge, which indicates an opportunity for targeting resources and support, in addition to efforts of reducing length of stay where possible.47
Readmissions were significantly related to a change in Rosow-Breslau mobility disability score. This may indicate the detrimental impact a readmission can have on increasing mobility and physical activity postdischarge, or the potential of this pilot program to impact readmissions by increasing mobility and physical activity, contrary to prior physical exercise interventions.5,7,9,48 With 5% to 79% of readmissions considered preventable, continued efforts and program dissemination and implementation to address preventable readmissions are warranted.49 Individuals with postdischarge physical therapy (prior to beginning the pilot program) tended to demonstrate less improvement in disability and physical performance. This relationship needs further investigation; the 2 groups did not appear to have significant differences at baseline, albeit with a small sample size. It is possible they experienced initial improvements with postdischarge physical therapy and plateaued or had little further reserve to improve upon entering the VVC program.
Strengths and Limitations
This pilot program provided evaluative data on the use of VVC to enhance function and physical activity in older adults posthospital discharge. It included individual (eg, fall, pain, cognitive impairment) and health service (eg, readmission, physical therapy) level factors as predictors of function and physical activity posthospitalization.5,7,9,15-19
The results of this pilot project stem from a small sample lacking diversity in terms of race, ethnicity, and sex. There was some variation in baseline and endpoints between participants, and when hospitalization, posthospital syndrome, and postdischarge health care application factors were collected. The majority of participants were recruited within a month postdischarge, and the program lasted about 6 months. Data collection was attempted at regular PAT contacts, but there was some variation in when visits occurred based on participant availability and preference. Some participants had missing data, which was handled using pairwise deletion.33 Larger studies are needed to confirm the findings of this study, particularly the trends that did not reach statistical significance. Home health services other than physical therapy (eg, nursing, occupational therapy) were not fully accounted for and should be considered in future research.
Conclusions
In patients undergoing a 6-month pilot VVC-based physical activity intervention posthospital discharge, improvements in mobility and disability were most likely in those who had no cognitive impairment and were not readmitted. Larger sample and qualitative investigations are necessary to optimize outcomes for patients who meet these clinical profiles.
Deconditioning among hospitalized older adults contributes to significant decline in posthospitalization functional ability, physical performance, and physical activity.1-10 Previous hospital-to-home interventions have targeted improving function and physical activity, including recent programs leveraging home telehealth as a feasible and potentially effective mode of delivering in-home exercise and rehabilitation.11-14 However, pilot interventions have shown mixed effectiveness.11,12,14 This study expands on a previously published intervention describing a pilot home telehealth program for veterans posthospital discharge that demonstrated significant 6-month improvement in physical activity as well as trends in physical function improvement, including among those with cognitive impairment.15 Factors that contribute to improved outcomes are the focus of the present study.
Key factors underlying the complexity of hospital-to-home transitions include hospitalization elements (ie, reason for admission and length of stay), associated posthospital syndromes (ie, postdischarge falls, medication changes, cognitive impairment, and pain), and postdischarge health care application (ie, physical therapy and hospital readmission).16-18 These factors may be associated with postdischarge functional ability, physical performance, and physical activity, but their direct influence on intervention outcomes is unclear (Figure 1).5,7,9,16-20 The objective of this study was to examine the influence of hospitalization, posthospital syndrome, and postdischarge health care application factors on outcomes of a US Department of Veterans Affairs (VA) Video Connect (VVC) intervention to enhance function and physical activity in older adults posthospital discharge.

health care application factors on physical activity, functional ability, and
physical performance intervention outcomes.
Methods
The previous analysis reported on patient characteristics, program feasibility, and preliminary outcomes.13,15 The current study reports on relationships between hospitalization, posthospital syndrome, and postdischarge health care application factors and change in key outcomes, namely postdischarge self-reported functional ability, physical performance, and physical activity from baseline to endpoint.
Participants provided written informed consent. The protocol and consent forms were approved by the VA Ann Arbor Healthcare System (VAAAHS) Research and Development Committee, and the project was registered on clinicaltrials.gov (NCT04045054).
Intervention
The pilot program targeted older adults following recent hospital discharge from VAAAHS. Participants were eligible if they were aged ≥ 50 years, had been discharged following an inpatient stay in the past 1 to 2 weeks, evaluated by physical therapy during hospitalization with stated rehabilitation goals on discharge, and followed by a VAAAHS primary care physician. Participants were either recruited during hospital admission or shortly after discharge.13
An experienced physical activity trainer (PAT) supported the progression of participants’ rehabilitation goals via a home exercise program and coached the patient and caregiver to optimize functional ability, physical performance, and physical activity. The PAT was a nonlicensed research assistant with extensive experience in applying standard physical activity enhancement protocols (eg, increased walking) to older adults with comorbidities. Participation in the program lasted about 6 months. Initiation of the PAT program was delayed if the patient was already receiving postdischarge home-based or outpatient physical therapy. The PAT contacted the patient weekly via VVC for the first 6 weeks, then monthly for a total of 6 months. Each contact included information on optimal walking form, injury prevention, program progression, and ways to incorporate sit-to-stand transitions, nonsitting behavior, and walking into daily routines. The initial VVC contact lasted about 60 minutes and subsequent sessions lasted about 30 minutes.13
Demographic characteristics were self-reported by participants and included age, sex, race, years of education, and marital status. Clinical characteristics were obtained from each participant’s electronic health record (EHR), including copay status, index hospitalization length of stay, admission diagnosis, and postsurgery status (postsurgery vs nonpostsurgery). Intervention adherence was tracked as the number of PAT sessions attended.
Posthospital Syndrome Factors
Participant falls (categorized as those who reported a fall vs those who did not) and medication changes (number of changes reported, including new medication, discontinued medication, dose changes, medication changes, or changes in medication schedule) were reported by participants or caregivers during each VVC contact. Participants completed the Montreal Cognitive Assessment (MoCA) at baseline, and were dichotomized into 2 groups: no cognitive impairment (MoCA score ≥ 26) and mild to moderate cognitive impairment (MoCA score 10-25).13,21
Participants rated how much pain interfered with their normal daily activities since the previous VVC session on a 5-point Likert scale (1, not at all; to 5, extremely).22 Similar to prior research, participants were placed into 2 groups based on their mean pain interference score (individuals with scores from 1.0 to 2.0 in 1 group, and individuals with > 2.0 in another).23-25 Participants were separated into a no or mild pain interference group and a moderate to severe pain interference group. Hospital readmissions (VA and non-VA) and postdischarge physical therapy outcomes were obtained from the participant’s EHR, including primary care visits.
Outcomes
Outcomes were collected at baseline (posthospital discharge) and 6 months postenrollment.
Self-Reported Functional Ability. This measure is provided by participants or caregivers and measured by the Katz Index of Independence in Activities of Daily Living (ADL), Lawton and Brody Instrumental ADL Scale (IADL), Nagi Disability Model, and Rosow-Breslau Scale. The Katz ADL assesses the ability to complete 6 self-care activities and awards 1 point for independence and 0 if the individual is dependent (total score range, 0-6).26 The Lawton and Brody IADL measures an individual’s independence in 8 instrumental ADLs; it awards 1 point for independence and 0 if the individual is dependent (total score range, 0-8).27 The Nagi Disability Model evaluates an individual’s difficulty performing 5 tasks (total score range, 0-5) and tallies the number of items with a response other than “no difficulty at all” (higher total score indicates greater difficulty). 28 The Rosow-Breslau Scale is a 3-item measure of mobility disability; individual responses are 0 (no help) and 1 (requires help or unable); higher total score (range, 0-3) indicates greater disability.29
Physical Performance. Measured using the Short Physical Performance Battery (SPPB), which evaluates standing balance, sit to stand, and walking performance. Scores range from 0 to 4 on the balance, gait speed, and chair stand tests, for a total composite score between 0 and 12 (higher score indicates better performance).30
Physical Activity. Measured using actigraphy, namely a physical activity monitor adherent to the thigh (activ-PAL3TM, PAL Technologies Ltd., Glasgow, UK).31 Participants were instructed to wear the activPal for ≥ 1 week. Participants with a minimum of 5 days of wear were included in this analysis.
Data Analyses
Analyses were performed using SPSS software version 29.0.32 Continuous variables were summarized using mean (SD) or median and IQR using the weighted average method; categorical variables were summarized using frequencies and percentages. Baseline scores on outcome variables were compared by categorical hospitalization, posthospital syndrome, and postdischarge health care application factor variables using Mann-Whitney U tests. The differences between outcome variables from baseline to endpoint were then calculated to produce change scores. Relationships between the number of PAT sessions attended and baseline outcomes and outcome change scores were estimated using Spearman correlations. Relationships between categorical factors (hospitalization, posthospital syndrome, and postdischarge health care application) and outcome variable change scores (which were normally distributed) were examined using Mann-Whitney U tests. Relationships with continuous hospitalization (length of stay) and posthospital syndrome factors (medication changes) were estimated using Spearman correlations. Effect sizes (ES) were estimated with Cohen d; small (d = 0.2), medium (d = 0.5), or large (d ≥ 0.8). Missing data were handled using pairwise deletion.33 Therefore, sample sizes were reported for each analysis. For all statistical tests, P < .05 was considered significant.
Results
Twenty-four individuals completed the pilot intervention.15 Mean (SD) age was 73.6 (8.1) years (range, 64-93 years) and participants were predominantly White males (Table 1). Eight participants had a high school education only and 13 had more than a high school education. Diagnoses at admission included 9 patients with orthopedic/musculoskeletal conditions (6 were for joint replacement), 6 patients with vascular/pulmonary conditions, and 4 with gastrointestinal/renal/urological conditions. Of the 11 postsurgery participants, 7 were orthopedic, 4 were gastrointestinal, and 1 was peripheral vascular.

Baseline outcome scores did not differ significantly between groups, except individuals with moderate to severe pain interference reported a significantly lower IADL score (median [IQR] 4 [2-7]) than individuals with mild or moderate pain interference (median [IQR] 8 [7-8]; P = .02) (Table 2). The mean (SD) number of PAT sessions attended was 9.3 (3.7) (range, 3-19). There were no significant relationships between number of sessions attended and any baseline outcome variables or outcome change scores.

Hospitalization Factors
Participants who were postsurgery tended to have greater improvement than individuals who were nonpostsurgery in ADLs (median [IQR] 0 [0-1.5]; ES, 0.6; P = .10) and SPPB (median [IQR] 2 [1.5-9]; ES, 0.9; P = .07), but the improvements were not statistically significant (Table 3). Mean (SD) length of stay of the index hospitalization was 6.7 (6.1) days. Longer length of stay was significantly correlated with an increase in Nagi score (ρ, 0.45; 95% CI, 0.01-0.75). There were no other significant or trending relationships between length of stay and outcome variables.

Posthospital Syndrome Factors
The 16 participants with mild to moderate cognitive impairment had less improvement in ADLs (median [IQR] 0 [0-1]) than the 8 participants with no impairment (median [IQR] 0 [-0.75 to 0]; ES, -1.1; P = .04). Change in outcome variables from baseline to endpoint did not significantly differ between the 8 patients who reported a fall compared with the 13 who did not, nor were any trends observed. Change in outcome variables from baseline to endpoint also did not significantly differ between the 8 participants who reported no or mild pain interference compared with the 10 patients with moderate to severe pain interference, nor were any trends observed. Mean (SD) number of medication changes was 2.5 (1.6). Higher number of medication changes was significantly correlated with a decrease in Rosow-Breslau score (ρ, -0.47; 95% CI, -0.76 to -0.02). There were no other significant or trending relationships between number of medication changes and outcome variables.
Postdischarge Health Care Application Factors
The 16 participants who attended posthospital physical therapy trended towards less improvement in IADLs (median [IQR] 0 [-0.5 to 1.5]; ES, -0.7; P = .11) and SPPB (median [IQR] 2 [-3.0 to 4.5]; ES, -0.5; P = .15) than the 8 patients with no postdischarge physical therapy. Eleven participants were readmitted, while 13 had no readmissions in their medical records between baseline and endpoint. Participants with ≥ 1 readmission experienced a greater increase in Rosow-Breslau score (median [IQR] 0 [-0.5 to 1.0]) than those not readmitted (median [IQR] 0 [-1.25 to 0.25]; ES, 1.0; P = .03). Borderline greater improvement in number of steps was found in those not readmitted (median [IQR] 3365.6 [274.4-7710.9]) compared with those readmitted (median [IQR] 319.9 [-136.1 to 774.5]; ES, -1.3; P = .05). Patients who were readmitted also tended to have lower and not statistically significant improvements in SPPB (median [IQR] 1 [-4.0 to 5.3]) compared with those not readmitted (median [IQR] 2 [0.3-3.8]; ES, -0.5; P = .17) (Table 3).
Discussion
This study examined the association between hospitalization, posthospital syndrome, and postdischarge health care use in patients undergoing a VVC-based intervention following hospital discharge. Participants who had no or mild cognitive impairment, no readmissions, higher medication changes, and a shorter hospital length of stay tended to experience lower disability, including in mobility and ADLs. This suggests individuals who are less clinically complex may be more likely to benefit from this type of virtual rehabilitation program. These findings are consistent with clinical experiences; home-based programs to improve physical activity posthospital discharge can be challenging for those who were medically ill (and did not undergo a specific surgical procedure), cognitively impaired, and become acutely ill and trigger hospital readmission. 15 For example, the sample in this study had higher rates of falls, pain, and readmissions compared to previous research.2,3,34-39
The importance of posthospital syndrome in the context of recovery of function and health at home following hospitalization is well documented.16-18 The potential impact of posthospital syndrome on physical activity-focused interventions is less understood. In our analysis, participants with mild or moderate cognitive impairment tended to become more dependent in their ADLs, while those with no cognitive impairment tended to become more independent in their ADLs. This functional decline over time is perhaps expected in persons with cognitive impairment, but the significant difference with a large ES warrants further consideration on how to tailor interventions to better promote functional recovery in these individuals.40,41 While some cognitive decline may not be preventable, this finding supports the need to promote healthy cognitive aging, identify declines in cognition, and work to mitigate additional decline. Programs specifically designed to promote function and physical activity in older adults with cognitive impairment are needed, especially during care transitions.41-43
While participants reported that falls and pain interference did not have a significant impact on change in outcomes between baseline and endpoint, these areas need further investigation. Falls and pain have been associated with function and physical activity in older adults.42-46 Pain is common, yet underappreciated during older adult hospital-to-home transitions.11,12,45,46 There is a need for more comprehensive assessment of pain (including pain intensity) and qualitative research.
Hospitalization and postdischarge health care application factors may have a significant impact on home-telehealth physical activity intervention success. Individuals who were postsurgery tended to have greater improvements in ADLs and physical performance. Most postsurgery participants had joint replacement surgery. Postsurgery status may not be modifiable, but it is important to note expected differences in recovery between medical and surgical admissions and the need to tailor care based on admission diagnosis. Those with a longer length of hospital stay may be considered at higher risk of suboptimal outcomes postdischarge, which indicates an opportunity for targeting resources and support, in addition to efforts of reducing length of stay where possible.47
Readmissions were significantly related to a change in Rosow-Breslau mobility disability score. This may indicate the detrimental impact a readmission can have on increasing mobility and physical activity postdischarge, or the potential of this pilot program to impact readmissions by increasing mobility and physical activity, contrary to prior physical exercise interventions.5,7,9,48 With 5% to 79% of readmissions considered preventable, continued efforts and program dissemination and implementation to address preventable readmissions are warranted.49 Individuals with postdischarge physical therapy (prior to beginning the pilot program) tended to demonstrate less improvement in disability and physical performance. This relationship needs further investigation; the 2 groups did not appear to have significant differences at baseline, albeit with a small sample size. It is possible they experienced initial improvements with postdischarge physical therapy and plateaued or had little further reserve to improve upon entering the VVC program.
Strengths and Limitations
This pilot program provided evaluative data on the use of VVC to enhance function and physical activity in older adults posthospital discharge. It included individual (eg, fall, pain, cognitive impairment) and health service (eg, readmission, physical therapy) level factors as predictors of function and physical activity posthospitalization.5,7,9,15-19
The results of this pilot project stem from a small sample lacking diversity in terms of race, ethnicity, and sex. There was some variation in baseline and endpoints between participants, and when hospitalization, posthospital syndrome, and postdischarge health care application factors were collected. The majority of participants were recruited within a month postdischarge, and the program lasted about 6 months. Data collection was attempted at regular PAT contacts, but there was some variation in when visits occurred based on participant availability and preference. Some participants had missing data, which was handled using pairwise deletion.33 Larger studies are needed to confirm the findings of this study, particularly the trends that did not reach statistical significance. Home health services other than physical therapy (eg, nursing, occupational therapy) were not fully accounted for and should be considered in future research.
Conclusions
In patients undergoing a 6-month pilot VVC-based physical activity intervention posthospital discharge, improvements in mobility and disability were most likely in those who had no cognitive impairment and were not readmitted. Larger sample and qualitative investigations are necessary to optimize outcomes for patients who meet these clinical profiles.
- Liebzeit D, Bratzke L, Boltz M, Purvis S, King B. Getting back to normal: a grounded theory study of function in post-hospitalized older adults. Gerontologist. 2020;60:704-714. doi:10.1093/geront/gnz057
- Ponzetto M, Zanocchi M, Maero B, et al. Post-hospitalization mortality in the elderly. Arch Gerontol Geriatr. 2003;36:83-91. doi:10.1016/s0167-4943(02)00061-4
- Buurman BM, Hoogerduijn JG, de Haan RJ, et al. Geriatric conditions in acutely hospitalized older patients: prevalence and one-year survival and functional decline. PLoS One. 2011;6:e26951. doi:10.1371/journal.pone.0026951
- Ponzetto M, Maero B, Maina P, et al. Risk factors for early and late mortality in hospitalized older patients: the continuing importance of functional status. J Gerontol A Biol Sci Med Sci. 2003;58:1049-1054. doi:10.1093/gerona/58.11.m1049
- Huang HT, Chang CM, Liu LF, Lin HS, Chen CH. Trajectories and predictors of functional decline of hospitalised older patients. J Clin Nurs. 2013;22:1322-1331. doi:10.1111/jocn.12055
- Boyd CM, Landefeld CS, Counsell SR, et al. Recovery of activities of daily living in older adults after hospitalization for acute medical illness. J Am Geriatr Soc. 2008;56:2171- 2179. doi:10.1111/j.1532-5415.2008.02023.x
- Helvik AS, Selbæk G, Engedal K. Functional decline in older adults one year after hospitalization. Arch Gerontol Geriatr. 2013;57:305-310. doi:10.1016/j.archger.2013.05.008
- Zaslavsky O, Zisberg A, Shadmi E. Impact of functional change before and during hospitalization on functional recovery 1 month following hospitalization. J Gerontol Biol Sci Med Sci. 2015;70:381-386. doi:10.1093/gerona/glu168
- Chen CC, Wang C, Huang GH. Functional trajectory 6 months posthospitalization: a cohort study of older hospitalized patients in Taiwan. Nurs Res. 2008;57:93-100. doi:10.1097/01.NNR.0000313485.18670.e2
- Kleinpell RM, Fletcher K, Jennings BM. Reducing functional decline in hospitalized elderly. In: Hughes RG, ed. Patient Safety and Quality: An Evidence-Based Handbook for Nurses. Agency for Healthcare Research and Quality (US); 2008. Accessed September 3, 2025. http://www.ncbi.nlm.nih.gov/books/NBK2629/
- Liebzeit D, Rutkowski R, Arbaje AI, Fields B, Werner NE. A scoping review of interventions for older adults transitioning from hospital to home. J Am Geriatr Soc. 2021;69:2950-2962. doi:10.1111/jgs.17323
- Hladkowicz E, Dumitrascu F, Auais M, et al. Evaluations of postoperative transitions in care for older adults: a scoping review. BMC Geriatr. 2022;22:329. doi:10.1186/s12877-022-02989-6
- Alexander NB, Phillips K, Wagner-Felkey J, et al. Team VA Video Connect (VVC) to optimize mobility and physical activity in post-hospital discharge older veterans: baseline assessment. BMC Geriatr. 2021;21:502. doi:10.1186/s12877-021-02454-w
- Dawson R, Oliveira JS, Kwok WS, et al. Exercise interventions delivered through telehealth to improve physical functioning for older adults with frailty, cognitive, or mobility disability: a systematic review and meta-analysis. Telemed J E Health. 2024;30:940-950. doi:10.1089/tmj.2023.0177
- Liebzeit D, Phillips KK, Hogikyan RV, Cigolle CT, Alexander NB. A pilot home-telehealth program to enhance functional ability, physical performance, and physical activity in older adult veterans post-hospital discharge. Res Gerontol Nurs. 2024;17:271-279. doi:10.3928/19404921-20241105-01
- Krumholz HM. Post-hospital syndrome—an acquired, transient condition of generalized risk. N Engl J Med. 2013;368:100-102. doi:10.1056/NEJMp1212324
- Caraballo C, Dharmarajan K, Krumholz HM. Post hospital syndrome: is the stress of hospitalization causing harm? Rev Esp Cardiol (Engl Ed). 2019;72:896-898. doi:10.1016/j.rec.2019.04.010
- Rawal S, Kwan JL, Razak F, et al. Association of the trauma of hospitalization with 30-day readmission or emergency department visit. JAMA Intern Med. 2019;179:38- 45. doi:10.1001/jamainternmed.2018.5100
- Dutzi I, Schwenk M, Kirchner M, Jooss E, Bauer JM, Hauer K. Influence of cognitive impairment on rehabilitation received and its mediating effect on functional recovery. J Alzheimers Dis. 2021;84:745-756. doi:10.3233/JAD-210620
- Uriz-Otano F, Uriz-Otano JI, Malafarina V. Factors associated with short-term functional recovery in elderly people with a hip fracture. Influence ofcognitiveimpairment. JAmMedDirAssoc. 2015;16:215-220. doi:10.1016/j.jamda.2014.09.009
- Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695-699. doi:10.1111/j.1532-5415.2005.53221.x
- Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30:473-483.
- White RS, Jiang J, Hall CB, et al. Higher perceived stress scale scores are associated with higher pain intensity and pain interference levels in older adults. J Am Geriatr Soc. 2014;62:2350-2356. doi:10.1111/jgs.13135
- Blyth FM, March LM, Brnabic AJ, et al. Chronic pain in Australia: a prevalence study. Pain. 2001;89:127-134. doi:10.1016/s0304-3959(00)00355-9
- Thomas E, Peat G, Harris L, Wilkie R, Croft PR. The prevalence of pain and pain interference in a general population of older adults: cross-sectional findings from the North Staffordshire Osteoarthritis Project (NorStOP). Pain. 2004;110:361-368. doi:10.1016/j.pain.2004.04.017
- Katz S, Ford AB, Moskowitz RW, Jackson BA, Jaffe MW. Studies of illness in the aged. The index of ADL: a standardized measure of biological and psychosocial function. JAMA. 1963;185:914-919. doi:10.1001/jama.1963.03060120024016
- Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. 1969;9:179-186.
- Alexander NB, Guire KE, Thelen DG, et al. Self-reported walking ability predicts functional mobility performance in frail older adults. J Am Geriatr Soc. 2000;48:1408-1413. doi:10.1111/j.1532-5415.2000.tb02630.x
- Rosow I, Breslau N. A Guttman health scale for the aged. J Gerontol. 1966;21:556-559. doi:10.1093/geronj/21.4.556
- Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49:M85-M94. doi:10.1093/geronj/49.2.m85
- Chan CS, Slaughter SE, Jones CA, Ickert C, Wagg AS. Measuring activity performance of older adults using the activPAL: a rapid review. Healthcare (Basel). 2017;5:94. doi:10.3390/healthcare5040094
- IBM SPSS software. IBM Corp; 2019. Accessed September 3, 2025. https://www.ibm.com/spss
- Kang H. The prevention and handling of the missing data. Korean J Anesthesiol. 2013;64:402-406. doi:10.4097/kjae.2013.64.5.402
- Epstein AM, Jha AK, Orav EJ. The relationship between hospital admission rates and rehospitalizations. N Engl J Med. 2011;365:2287-2295. doi:10.1056/NEJMsa1101942
- Bogaisky M, Dezieck L. Early hospital readmission of nursing home residents and community-dwelling elderly adults discharged from the geriatrics service of an urban teaching hospital: patterns and risk factors. J Am Geriatr Soc. 2015;63:548-552. doi:10.1111/jgs.13317
- Jencks SF, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009;360:1418-1428. doi:10.1056/NEJMsa0803563
- Hoyer EH, Needham DM, Atanelov L, Knox B, Friedman M, Brotman DJ. Association of impaired functional status at hospital discharge and subsequent rehospitalization. J Hosp Med. 2014;9:277-282. doi:10.1002/jhm.2152
- Mahoney J, Sager M, Dunham NC, Johnson J. Risk of falls after hospital discharge. J Am Geriatr Soc. 1994;42:269- 274. doi:10.1111/j.1532-5415.1994.tb01750.x
- Hoffman GJ, Liu H, Alexander NB, Tinetti M, Braun TM, Min LC. Posthospital fall injuries and 30-day readmissions in adults 65 years and older. JAMA Netw Open. 2019;2:e194276. doi:10.1001/jamanetworkopen.2019.4276
- Gill DP, Hubbard RA, Koepsell TD, et al. Differences in rate of functional decline across three dementia types. Alzheimers Dement. 2013;9:S63-S71. doi:10.1016/j.jalz.2012.10.010
- Auyeung TW, Kwok T, Lee J, Leung PC, Leung J, Woo J. Functional decline in cognitive impairment–the relationship between physical and cognitive function. Neuroepidemiology. 2008;31:167-173. doi:10.1159/000154929
- Patti A, Zangla D, Sahin FN, et al. Physical exercise and prevention of falls. Effects of a Pilates training method compared with a general physical activity program. Medicine (Baltimore). 2021;100:e25289. doi:10.1097/MD.0000000000025289
- Nagarkar A, Kulkarni S. Association between daily activities and fall in older adults: an analysis of longitudinal ageing study in India (2017-18). BMC Geriatr. 2022;22:203. doi:10.1186/s12877-022-02879-x
- Ek S, Rizzuto D, Xu W, Calderón-Larrañaga A, Welmer AK. Predictors for functional decline after an injurious fall: a population-based cohort study. Aging Clin Exp Res. 2021;33:2183-2190. doi:10.1007/s40520-020-01747-1
- Dagnino APA, Campos MM. Chronic pain in the elderly: mechanisms and perspectives. Front Hum Neurosci. 2022;16:736688. doi:10.3389/fnhum.2022.736688
- Ritchie CS, Patel K, Boscardin J, et al. Impact of persistent pain on function, cognition, and well-being of older adults. J Am Geriatr Soc. 2023;71:26-35. doi:10.1111/jgs.18125
- Han TS, Murray P, Robin J, et al. Evaluation of the association of length of stay in hospital and outcomes. Int J Qual Health Care. 2022;34:mzab160. doi:10.1093/intqhc/ mzab160
- Lærum-Onsager E, Molin M, Olsen CF, et al. Effect of nutritional and physical exercise intervention on hospital readmission for patients aged 65 or older: a systematic review and meta-analysis of randomized controlled trials. Int J Behav Nutr Phys Act. 2021;18:62. doi:10.1186/s12966-021-01123-w
- Van Walraven C, Bennett C, Jennings A, Austin PC, Forster AJ. Proportion of hospital readmissions deemed avoidable: a systematic review. CMAJ. 2011;183:E391-E402. doi:10.1503/cmaj.101860
- Liebzeit D, Bratzke L, Boltz M, Purvis S, King B. Getting back to normal: a grounded theory study of function in post-hospitalized older adults. Gerontologist. 2020;60:704-714. doi:10.1093/geront/gnz057
- Ponzetto M, Zanocchi M, Maero B, et al. Post-hospitalization mortality in the elderly. Arch Gerontol Geriatr. 2003;36:83-91. doi:10.1016/s0167-4943(02)00061-4
- Buurman BM, Hoogerduijn JG, de Haan RJ, et al. Geriatric conditions in acutely hospitalized older patients: prevalence and one-year survival and functional decline. PLoS One. 2011;6:e26951. doi:10.1371/journal.pone.0026951
- Ponzetto M, Maero B, Maina P, et al. Risk factors for early and late mortality in hospitalized older patients: the continuing importance of functional status. J Gerontol A Biol Sci Med Sci. 2003;58:1049-1054. doi:10.1093/gerona/58.11.m1049
- Huang HT, Chang CM, Liu LF, Lin HS, Chen CH. Trajectories and predictors of functional decline of hospitalised older patients. J Clin Nurs. 2013;22:1322-1331. doi:10.1111/jocn.12055
- Boyd CM, Landefeld CS, Counsell SR, et al. Recovery of activities of daily living in older adults after hospitalization for acute medical illness. J Am Geriatr Soc. 2008;56:2171- 2179. doi:10.1111/j.1532-5415.2008.02023.x
- Helvik AS, Selbæk G, Engedal K. Functional decline in older adults one year after hospitalization. Arch Gerontol Geriatr. 2013;57:305-310. doi:10.1016/j.archger.2013.05.008
- Zaslavsky O, Zisberg A, Shadmi E. Impact of functional change before and during hospitalization on functional recovery 1 month following hospitalization. J Gerontol Biol Sci Med Sci. 2015;70:381-386. doi:10.1093/gerona/glu168
- Chen CC, Wang C, Huang GH. Functional trajectory 6 months posthospitalization: a cohort study of older hospitalized patients in Taiwan. Nurs Res. 2008;57:93-100. doi:10.1097/01.NNR.0000313485.18670.e2
- Kleinpell RM, Fletcher K, Jennings BM. Reducing functional decline in hospitalized elderly. In: Hughes RG, ed. Patient Safety and Quality: An Evidence-Based Handbook for Nurses. Agency for Healthcare Research and Quality (US); 2008. Accessed September 3, 2025. http://www.ncbi.nlm.nih.gov/books/NBK2629/
- Liebzeit D, Rutkowski R, Arbaje AI, Fields B, Werner NE. A scoping review of interventions for older adults transitioning from hospital to home. J Am Geriatr Soc. 2021;69:2950-2962. doi:10.1111/jgs.17323
- Hladkowicz E, Dumitrascu F, Auais M, et al. Evaluations of postoperative transitions in care for older adults: a scoping review. BMC Geriatr. 2022;22:329. doi:10.1186/s12877-022-02989-6
- Alexander NB, Phillips K, Wagner-Felkey J, et al. Team VA Video Connect (VVC) to optimize mobility and physical activity in post-hospital discharge older veterans: baseline assessment. BMC Geriatr. 2021;21:502. doi:10.1186/s12877-021-02454-w
- Dawson R, Oliveira JS, Kwok WS, et al. Exercise interventions delivered through telehealth to improve physical functioning for older adults with frailty, cognitive, or mobility disability: a systematic review and meta-analysis. Telemed J E Health. 2024;30:940-950. doi:10.1089/tmj.2023.0177
- Liebzeit D, Phillips KK, Hogikyan RV, Cigolle CT, Alexander NB. A pilot home-telehealth program to enhance functional ability, physical performance, and physical activity in older adult veterans post-hospital discharge. Res Gerontol Nurs. 2024;17:271-279. doi:10.3928/19404921-20241105-01
- Krumholz HM. Post-hospital syndrome—an acquired, transient condition of generalized risk. N Engl J Med. 2013;368:100-102. doi:10.1056/NEJMp1212324
- Caraballo C, Dharmarajan K, Krumholz HM. Post hospital syndrome: is the stress of hospitalization causing harm? Rev Esp Cardiol (Engl Ed). 2019;72:896-898. doi:10.1016/j.rec.2019.04.010
- Rawal S, Kwan JL, Razak F, et al. Association of the trauma of hospitalization with 30-day readmission or emergency department visit. JAMA Intern Med. 2019;179:38- 45. doi:10.1001/jamainternmed.2018.5100
- Dutzi I, Schwenk M, Kirchner M, Jooss E, Bauer JM, Hauer K. Influence of cognitive impairment on rehabilitation received and its mediating effect on functional recovery. J Alzheimers Dis. 2021;84:745-756. doi:10.3233/JAD-210620
- Uriz-Otano F, Uriz-Otano JI, Malafarina V. Factors associated with short-term functional recovery in elderly people with a hip fracture. Influence ofcognitiveimpairment. JAmMedDirAssoc. 2015;16:215-220. doi:10.1016/j.jamda.2014.09.009
- Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695-699. doi:10.1111/j.1532-5415.2005.53221.x
- Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30:473-483.
- White RS, Jiang J, Hall CB, et al. Higher perceived stress scale scores are associated with higher pain intensity and pain interference levels in older adults. J Am Geriatr Soc. 2014;62:2350-2356. doi:10.1111/jgs.13135
- Blyth FM, March LM, Brnabic AJ, et al. Chronic pain in Australia: a prevalence study. Pain. 2001;89:127-134. doi:10.1016/s0304-3959(00)00355-9
- Thomas E, Peat G, Harris L, Wilkie R, Croft PR. The prevalence of pain and pain interference in a general population of older adults: cross-sectional findings from the North Staffordshire Osteoarthritis Project (NorStOP). Pain. 2004;110:361-368. doi:10.1016/j.pain.2004.04.017
- Katz S, Ford AB, Moskowitz RW, Jackson BA, Jaffe MW. Studies of illness in the aged. The index of ADL: a standardized measure of biological and psychosocial function. JAMA. 1963;185:914-919. doi:10.1001/jama.1963.03060120024016
- Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. 1969;9:179-186.
- Alexander NB, Guire KE, Thelen DG, et al. Self-reported walking ability predicts functional mobility performance in frail older adults. J Am Geriatr Soc. 2000;48:1408-1413. doi:10.1111/j.1532-5415.2000.tb02630.x
- Rosow I, Breslau N. A Guttman health scale for the aged. J Gerontol. 1966;21:556-559. doi:10.1093/geronj/21.4.556
- Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49:M85-M94. doi:10.1093/geronj/49.2.m85
- Chan CS, Slaughter SE, Jones CA, Ickert C, Wagg AS. Measuring activity performance of older adults using the activPAL: a rapid review. Healthcare (Basel). 2017;5:94. doi:10.3390/healthcare5040094
- IBM SPSS software. IBM Corp; 2019. Accessed September 3, 2025. https://www.ibm.com/spss
- Kang H. The prevention and handling of the missing data. Korean J Anesthesiol. 2013;64:402-406. doi:10.4097/kjae.2013.64.5.402
- Epstein AM, Jha AK, Orav EJ. The relationship between hospital admission rates and rehospitalizations. N Engl J Med. 2011;365:2287-2295. doi:10.1056/NEJMsa1101942
- Bogaisky M, Dezieck L. Early hospital readmission of nursing home residents and community-dwelling elderly adults discharged from the geriatrics service of an urban teaching hospital: patterns and risk factors. J Am Geriatr Soc. 2015;63:548-552. doi:10.1111/jgs.13317
- Jencks SF, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009;360:1418-1428. doi:10.1056/NEJMsa0803563
- Hoyer EH, Needham DM, Atanelov L, Knox B, Friedman M, Brotman DJ. Association of impaired functional status at hospital discharge and subsequent rehospitalization. J Hosp Med. 2014;9:277-282. doi:10.1002/jhm.2152
- Mahoney J, Sager M, Dunham NC, Johnson J. Risk of falls after hospital discharge. J Am Geriatr Soc. 1994;42:269- 274. doi:10.1111/j.1532-5415.1994.tb01750.x
- Hoffman GJ, Liu H, Alexander NB, Tinetti M, Braun TM, Min LC. Posthospital fall injuries and 30-day readmissions in adults 65 years and older. JAMA Netw Open. 2019;2:e194276. doi:10.1001/jamanetworkopen.2019.4276
- Gill DP, Hubbard RA, Koepsell TD, et al. Differences in rate of functional decline across three dementia types. Alzheimers Dement. 2013;9:S63-S71. doi:10.1016/j.jalz.2012.10.010
- Auyeung TW, Kwok T, Lee J, Leung PC, Leung J, Woo J. Functional decline in cognitive impairment–the relationship between physical and cognitive function. Neuroepidemiology. 2008;31:167-173. doi:10.1159/000154929
- Patti A, Zangla D, Sahin FN, et al. Physical exercise and prevention of falls. Effects of a Pilates training method compared with a general physical activity program. Medicine (Baltimore). 2021;100:e25289. doi:10.1097/MD.0000000000025289
- Nagarkar A, Kulkarni S. Association between daily activities and fall in older adults: an analysis of longitudinal ageing study in India (2017-18). BMC Geriatr. 2022;22:203. doi:10.1186/s12877-022-02879-x
- Ek S, Rizzuto D, Xu W, Calderón-Larrañaga A, Welmer AK. Predictors for functional decline after an injurious fall: a population-based cohort study. Aging Clin Exp Res. 2021;33:2183-2190. doi:10.1007/s40520-020-01747-1
- Dagnino APA, Campos MM. Chronic pain in the elderly: mechanisms and perspectives. Front Hum Neurosci. 2022;16:736688. doi:10.3389/fnhum.2022.736688
- Ritchie CS, Patel K, Boscardin J, et al. Impact of persistent pain on function, cognition, and well-being of older adults. J Am Geriatr Soc. 2023;71:26-35. doi:10.1111/jgs.18125
- Han TS, Murray P, Robin J, et al. Evaluation of the association of length of stay in hospital and outcomes. Int J Qual Health Care. 2022;34:mzab160. doi:10.1093/intqhc/ mzab160
- Lærum-Onsager E, Molin M, Olsen CF, et al. Effect of nutritional and physical exercise intervention on hospital readmission for patients aged 65 or older: a systematic review and meta-analysis of randomized controlled trials. Int J Behav Nutr Phys Act. 2021;18:62. doi:10.1186/s12966-021-01123-w
- Van Walraven C, Bennett C, Jennings A, Austin PC, Forster AJ. Proportion of hospital readmissions deemed avoidable: a systematic review. CMAJ. 2011;183:E391-E402. doi:10.1503/cmaj.101860
Factors Influencing Outcomes of a Telehealth-Based Physical Activity Program in Older Veterans Postdischarge
Factors Influencing Outcomes of a Telehealth-Based Physical Activity Program in Older Veterans Postdischarge