User login
-
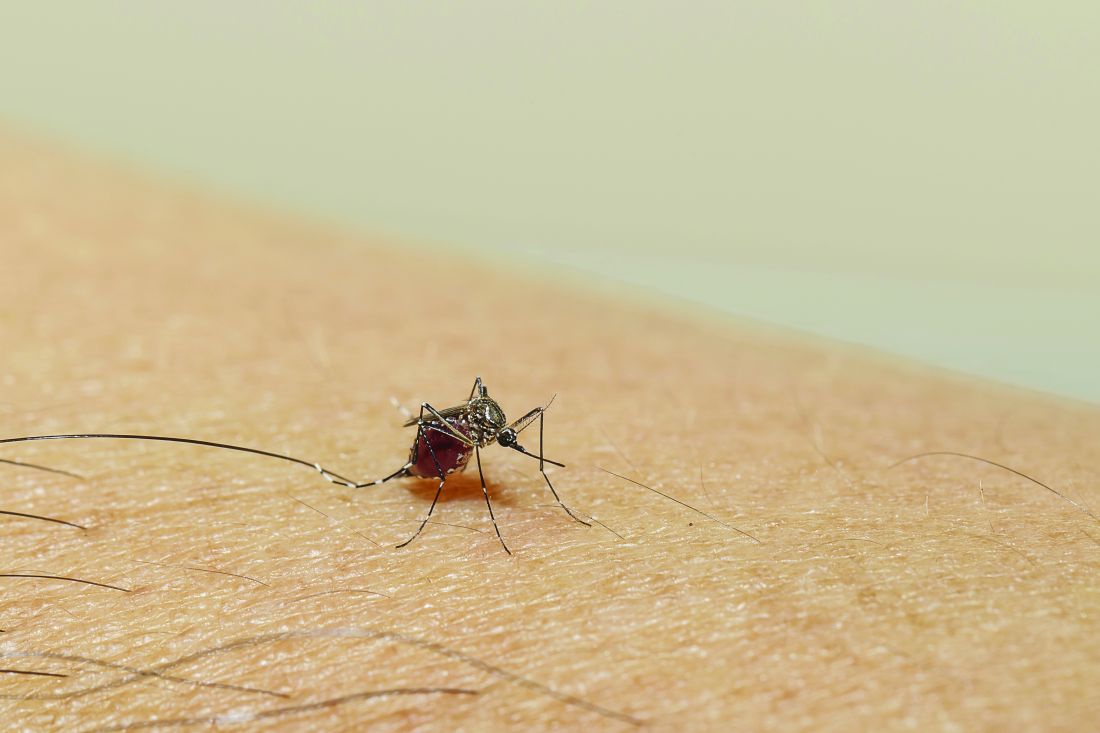
Mosquitoes and the vicious circle that’s gone viral
These viruses want mosquitoes with good taste
Taste can be a pretty subjective sense. Not everyone agrees on what tastes good and what tastes bad. Most people would agree that freshly baked cookies taste good, but what about lima beans? And what about mosquitoes? What tastes good to a mosquito?
The answer? Blood. Blood tastes good to a mosquito. That really wasn’t a very hard question, was it? You did know the answer, didn’t you? They don’t care about cookies, and they certainly don’t care about lima beans. It’s blood that they love.
That brings us back to subjectivity, because it is possible for blood to taste even better. The secret ingredient is dengue … and Zika.
A study just published in Cell demonstrates that mice infected with dengue and Zika viruses release a volatile compound called acetophenone. “We found that flavivirus [like dengue and Zika] can utilize the increased release of acetophenone to help itself achieve its lifecycles more effectively by making their hosts more attractive to mosquito vectors,” senior author Gong Cheng of Tsinghua University, Beijing, said in a written statement.
How do they do it? The viruses, he explained, promote the proliferation of acetophenone-producing skin bacteria. “As a result, some bacteria overreplicate and produce more acetophenone. Suddenly, these sick individuals smell as delicious to mosquitoes as a tray of freshly baked cookies to a group of five-year-old children,” the statement said.
And how do you stop a group of tiny, flying 5-year-olds? That’s right, with acne medication. Really? You knew that one but not the blood one before? The investigators fed isotretinoin to the infected mice, which led to reduced acetophenone release from skin bacteria and made the animals no more attractive to the mosquitoes than their uninfected counterparts.
The investigators are planning to take the next step – feeding isotretinoin to people with dengue and Zika – having gotten the official fictional taste-test approval of celebrity chef Gordon Ramsay, who said, “You’re going to feed this #$^% to sick people? ARE YOU &%*$@#& KIDDING ME?”
Okay, so maybe approval isn’t quite the right word.
Welcome to bladders of the rich and famous!
Don’t you hate it when you’re driving out to your multimillion-dollar second home in the Hamptons and traffic is so bad you absolutely have to find a place to “rest” along the way? But wouldn’t you know it, there just isn’t anywhere to stop! Geez, how do we live?
That’s where David Shusterman, MD, a urologist in New York City and a true American hero, comes in. He’s identified a market and positioned himself as the king of both bladder surgery and “bladder Botox” for the wealthy New Yorkers who regularly make long journeys from the city out to their second homes in the Hamptons. Traffic has increased dramatically on Long Island roads in recent years, and the journey can now taking upward of 4 hours. Some people just can’t make it that long without a bathroom break, and there are very few places to stop along the way.
Dr. Shusterman understands the plight of the Hamptons vacationer, as he told Insider.com: “I can’t tell you how many arguments I personally get into – I’ve lost three friends because I’m the driver and refuse to stop for them.” A tragedy worthy of Shakespeare himself.
During the summer season, Dr. Shusterman performs about 10 prostate artery embolizations a week, an hour-long procedure that shrinks the prostate, which is great for 50- to 60-year-old men with enlarged prostates that cause more frequent bathroom trips. He also performs Botox injections into the bladder once or twice a week for women, which reduces the need to urinate for roughly 6 months. The perfect amount of time to get them through the summer season.
These procedures are sometimes covered by insurance but can cost as much as $20,000 if paid out of pocket. That’s a lot of money to us, but if you’re the sort of person who has a second home in the Hamptons, $20,000 is chump change, especially if it means you won’t have to go 2 entire minutes out of your way to use a gas-station bathroom. Then again, having seen a more than a few gas-station bathrooms in our time, maybe they have a point.
Ditch the apples. Go for the avocados
We’ve all heard about “an apple a day,” but instead of apples you might want to go with avocados.
Avocados are generally thought to be a healthy fat. A study just published in the Journal of the American Heart Association proves that they actually don’t do anything for your waistline but will work wonders on your cholesterol level. The study involved 923 participants who were considered overweight/obese split into two groups: One was asked to consume an avocado a day, and the other continued their usual diets and were asked to consume fewer than two avocados a month.
At the end of the 6 months, the researchers found total cholesterol decreased by an additional 2.9 mg/dL and LDL cholesterol by 2.5 mg/dL in those who ate one avocado every day, compared with the usual-diet group. And even though avocados have a lot of calories, there was no clinical evidence that it impacted weight gain or any cardiometabolic risk factors, according to a statement from Penn State University.
Avocados, then, can be considered a guilt-free food. The findings from this study suggest it can give a substantial boost to your overall quality of diet, in turn lessening your risk of developing type 2 diabetes and some cancers, Kristina Peterson, PhD, assistant professor of nutritional sciences at Texas Tech University, said in the statement.
So get creative with your avocado recipes. You can only eat so much guacamole.
Your nose knows a good friend for you
You’ve probably noticed how dogs sniff other dogs and people before becoming friends. It would be pretty comical if people did the same thing, right? Just walked up to strangers and started sniffing them like dogs?
Well, apparently humans do go by smell when it comes to making friends, and they prefer people who smell like them. Maybe you’ve noticed that your friends look like you, share your values, and think the same way as you. You’re probably right, seeing as previous research has pointed to this.
For the current study, done to show how smell affects human behavior, researchers recruited people who befriended each other quickly, before knowing much about each other. They assumed that the relationships between these same-sex, nonromantic “click friends” relied more on physiological traits, including smell. After collecting samples from the click friends, researchers used an eNose to scan chemical signatures. In another experiment, human volunteers sniffed samples to determine if any were similar. Both experiments showed that click friends had more similar smells than pairs of random people.
“This is not to say that we act like goats or shrews – humans likely rely on other, far more dominant cues in their social decision-making. Nevertheless, our study’s results do suggest that our nose plays a bigger role than previously thought in our choice of friends,” said senior author Noam Sobel, PhD, of the Weizmann Institute of Science in Rehovot, Israel.
Lead author Inbal Ravreby, a graduate student at the institute, put it this way: “These results imply that, as the saying goes, there is chemistry in social chemistry.”
These viruses want mosquitoes with good taste
Taste can be a pretty subjective sense. Not everyone agrees on what tastes good and what tastes bad. Most people would agree that freshly baked cookies taste good, but what about lima beans? And what about mosquitoes? What tastes good to a mosquito?
The answer? Blood. Blood tastes good to a mosquito. That really wasn’t a very hard question, was it? You did know the answer, didn’t you? They don’t care about cookies, and they certainly don’t care about lima beans. It’s blood that they love.
That brings us back to subjectivity, because it is possible for blood to taste even better. The secret ingredient is dengue … and Zika.
A study just published in Cell demonstrates that mice infected with dengue and Zika viruses release a volatile compound called acetophenone. “We found that flavivirus [like dengue and Zika] can utilize the increased release of acetophenone to help itself achieve its lifecycles more effectively by making their hosts more attractive to mosquito vectors,” senior author Gong Cheng of Tsinghua University, Beijing, said in a written statement.
How do they do it? The viruses, he explained, promote the proliferation of acetophenone-producing skin bacteria. “As a result, some bacteria overreplicate and produce more acetophenone. Suddenly, these sick individuals smell as delicious to mosquitoes as a tray of freshly baked cookies to a group of five-year-old children,” the statement said.
And how do you stop a group of tiny, flying 5-year-olds? That’s right, with acne medication. Really? You knew that one but not the blood one before? The investigators fed isotretinoin to the infected mice, which led to reduced acetophenone release from skin bacteria and made the animals no more attractive to the mosquitoes than their uninfected counterparts.
The investigators are planning to take the next step – feeding isotretinoin to people with dengue and Zika – having gotten the official fictional taste-test approval of celebrity chef Gordon Ramsay, who said, “You’re going to feed this #$^% to sick people? ARE YOU &%*$@#& KIDDING ME?”
Okay, so maybe approval isn’t quite the right word.
Welcome to bladders of the rich and famous!
Don’t you hate it when you’re driving out to your multimillion-dollar second home in the Hamptons and traffic is so bad you absolutely have to find a place to “rest” along the way? But wouldn’t you know it, there just isn’t anywhere to stop! Geez, how do we live?
That’s where David Shusterman, MD, a urologist in New York City and a true American hero, comes in. He’s identified a market and positioned himself as the king of both bladder surgery and “bladder Botox” for the wealthy New Yorkers who regularly make long journeys from the city out to their second homes in the Hamptons. Traffic has increased dramatically on Long Island roads in recent years, and the journey can now taking upward of 4 hours. Some people just can’t make it that long without a bathroom break, and there are very few places to stop along the way.
Dr. Shusterman understands the plight of the Hamptons vacationer, as he told Insider.com: “I can’t tell you how many arguments I personally get into – I’ve lost three friends because I’m the driver and refuse to stop for them.” A tragedy worthy of Shakespeare himself.
During the summer season, Dr. Shusterman performs about 10 prostate artery embolizations a week, an hour-long procedure that shrinks the prostate, which is great for 50- to 60-year-old men with enlarged prostates that cause more frequent bathroom trips. He also performs Botox injections into the bladder once or twice a week for women, which reduces the need to urinate for roughly 6 months. The perfect amount of time to get them through the summer season.
These procedures are sometimes covered by insurance but can cost as much as $20,000 if paid out of pocket. That’s a lot of money to us, but if you’re the sort of person who has a second home in the Hamptons, $20,000 is chump change, especially if it means you won’t have to go 2 entire minutes out of your way to use a gas-station bathroom. Then again, having seen a more than a few gas-station bathrooms in our time, maybe they have a point.
Ditch the apples. Go for the avocados
We’ve all heard about “an apple a day,” but instead of apples you might want to go with avocados.
Avocados are generally thought to be a healthy fat. A study just published in the Journal of the American Heart Association proves that they actually don’t do anything for your waistline but will work wonders on your cholesterol level. The study involved 923 participants who were considered overweight/obese split into two groups: One was asked to consume an avocado a day, and the other continued their usual diets and were asked to consume fewer than two avocados a month.
At the end of the 6 months, the researchers found total cholesterol decreased by an additional 2.9 mg/dL and LDL cholesterol by 2.5 mg/dL in those who ate one avocado every day, compared with the usual-diet group. And even though avocados have a lot of calories, there was no clinical evidence that it impacted weight gain or any cardiometabolic risk factors, according to a statement from Penn State University.
Avocados, then, can be considered a guilt-free food. The findings from this study suggest it can give a substantial boost to your overall quality of diet, in turn lessening your risk of developing type 2 diabetes and some cancers, Kristina Peterson, PhD, assistant professor of nutritional sciences at Texas Tech University, said in the statement.
So get creative with your avocado recipes. You can only eat so much guacamole.
Your nose knows a good friend for you
You’ve probably noticed how dogs sniff other dogs and people before becoming friends. It would be pretty comical if people did the same thing, right? Just walked up to strangers and started sniffing them like dogs?
Well, apparently humans do go by smell when it comes to making friends, and they prefer people who smell like them. Maybe you’ve noticed that your friends look like you, share your values, and think the same way as you. You’re probably right, seeing as previous research has pointed to this.
For the current study, done to show how smell affects human behavior, researchers recruited people who befriended each other quickly, before knowing much about each other. They assumed that the relationships between these same-sex, nonromantic “click friends” relied more on physiological traits, including smell. After collecting samples from the click friends, researchers used an eNose to scan chemical signatures. In another experiment, human volunteers sniffed samples to determine if any were similar. Both experiments showed that click friends had more similar smells than pairs of random people.
“This is not to say that we act like goats or shrews – humans likely rely on other, far more dominant cues in their social decision-making. Nevertheless, our study’s results do suggest that our nose plays a bigger role than previously thought in our choice of friends,” said senior author Noam Sobel, PhD, of the Weizmann Institute of Science in Rehovot, Israel.
Lead author Inbal Ravreby, a graduate student at the institute, put it this way: “These results imply that, as the saying goes, there is chemistry in social chemistry.”
These viruses want mosquitoes with good taste
Taste can be a pretty subjective sense. Not everyone agrees on what tastes good and what tastes bad. Most people would agree that freshly baked cookies taste good, but what about lima beans? And what about mosquitoes? What tastes good to a mosquito?
The answer? Blood. Blood tastes good to a mosquito. That really wasn’t a very hard question, was it? You did know the answer, didn’t you? They don’t care about cookies, and they certainly don’t care about lima beans. It’s blood that they love.
That brings us back to subjectivity, because it is possible for blood to taste even better. The secret ingredient is dengue … and Zika.
A study just published in Cell demonstrates that mice infected with dengue and Zika viruses release a volatile compound called acetophenone. “We found that flavivirus [like dengue and Zika] can utilize the increased release of acetophenone to help itself achieve its lifecycles more effectively by making their hosts more attractive to mosquito vectors,” senior author Gong Cheng of Tsinghua University, Beijing, said in a written statement.
How do they do it? The viruses, he explained, promote the proliferation of acetophenone-producing skin bacteria. “As a result, some bacteria overreplicate and produce more acetophenone. Suddenly, these sick individuals smell as delicious to mosquitoes as a tray of freshly baked cookies to a group of five-year-old children,” the statement said.
And how do you stop a group of tiny, flying 5-year-olds? That’s right, with acne medication. Really? You knew that one but not the blood one before? The investigators fed isotretinoin to the infected mice, which led to reduced acetophenone release from skin bacteria and made the animals no more attractive to the mosquitoes than their uninfected counterparts.
The investigators are planning to take the next step – feeding isotretinoin to people with dengue and Zika – having gotten the official fictional taste-test approval of celebrity chef Gordon Ramsay, who said, “You’re going to feed this #$^% to sick people? ARE YOU &%*$@#& KIDDING ME?”
Okay, so maybe approval isn’t quite the right word.
Welcome to bladders of the rich and famous!
Don’t you hate it when you’re driving out to your multimillion-dollar second home in the Hamptons and traffic is so bad you absolutely have to find a place to “rest” along the way? But wouldn’t you know it, there just isn’t anywhere to stop! Geez, how do we live?
That’s where David Shusterman, MD, a urologist in New York City and a true American hero, comes in. He’s identified a market and positioned himself as the king of both bladder surgery and “bladder Botox” for the wealthy New Yorkers who regularly make long journeys from the city out to their second homes in the Hamptons. Traffic has increased dramatically on Long Island roads in recent years, and the journey can now taking upward of 4 hours. Some people just can’t make it that long without a bathroom break, and there are very few places to stop along the way.
Dr. Shusterman understands the plight of the Hamptons vacationer, as he told Insider.com: “I can’t tell you how many arguments I personally get into – I’ve lost three friends because I’m the driver and refuse to stop for them.” A tragedy worthy of Shakespeare himself.
During the summer season, Dr. Shusterman performs about 10 prostate artery embolizations a week, an hour-long procedure that shrinks the prostate, which is great for 50- to 60-year-old men with enlarged prostates that cause more frequent bathroom trips. He also performs Botox injections into the bladder once or twice a week for women, which reduces the need to urinate for roughly 6 months. The perfect amount of time to get them through the summer season.
These procedures are sometimes covered by insurance but can cost as much as $20,000 if paid out of pocket. That’s a lot of money to us, but if you’re the sort of person who has a second home in the Hamptons, $20,000 is chump change, especially if it means you won’t have to go 2 entire minutes out of your way to use a gas-station bathroom. Then again, having seen a more than a few gas-station bathrooms in our time, maybe they have a point.
Ditch the apples. Go for the avocados
We’ve all heard about “an apple a day,” but instead of apples you might want to go with avocados.
Avocados are generally thought to be a healthy fat. A study just published in the Journal of the American Heart Association proves that they actually don’t do anything for your waistline but will work wonders on your cholesterol level. The study involved 923 participants who were considered overweight/obese split into two groups: One was asked to consume an avocado a day, and the other continued their usual diets and were asked to consume fewer than two avocados a month.
At the end of the 6 months, the researchers found total cholesterol decreased by an additional 2.9 mg/dL and LDL cholesterol by 2.5 mg/dL in those who ate one avocado every day, compared with the usual-diet group. And even though avocados have a lot of calories, there was no clinical evidence that it impacted weight gain or any cardiometabolic risk factors, according to a statement from Penn State University.
Avocados, then, can be considered a guilt-free food. The findings from this study suggest it can give a substantial boost to your overall quality of diet, in turn lessening your risk of developing type 2 diabetes and some cancers, Kristina Peterson, PhD, assistant professor of nutritional sciences at Texas Tech University, said in the statement.
So get creative with your avocado recipes. You can only eat so much guacamole.
Your nose knows a good friend for you
You’ve probably noticed how dogs sniff other dogs and people before becoming friends. It would be pretty comical if people did the same thing, right? Just walked up to strangers and started sniffing them like dogs?
Well, apparently humans do go by smell when it comes to making friends, and they prefer people who smell like them. Maybe you’ve noticed that your friends look like you, share your values, and think the same way as you. You’re probably right, seeing as previous research has pointed to this.
For the current study, done to show how smell affects human behavior, researchers recruited people who befriended each other quickly, before knowing much about each other. They assumed that the relationships between these same-sex, nonromantic “click friends” relied more on physiological traits, including smell. After collecting samples from the click friends, researchers used an eNose to scan chemical signatures. In another experiment, human volunteers sniffed samples to determine if any were similar. Both experiments showed that click friends had more similar smells than pairs of random people.
“This is not to say that we act like goats or shrews – humans likely rely on other, far more dominant cues in their social decision-making. Nevertheless, our study’s results do suggest that our nose plays a bigger role than previously thought in our choice of friends,” said senior author Noam Sobel, PhD, of the Weizmann Institute of Science in Rehovot, Israel.
Lead author Inbal Ravreby, a graduate student at the institute, put it this way: “These results imply that, as the saying goes, there is chemistry in social chemistry.”
Will the headache field embrace rofecoxib?
In June, the Concord, Mass.–based company Tremeau Pharmaceuticals announced that the Food and Drug Administration was letting it proceed with a phase 3 clinical trial to test rofecoxib, the once-bestselling painkiller known as Vioxx, in patients with migraine.
The anti-inflammatory drug, a cyclooxygenase-2 (COX-2) inhibitor, received its first FDA approval in 1999 and became widely prescribed for arthritis and acute pain. In 2004 it was withdrawn by its manufacturer, Merck, after being shown to raise the risk of cardiovascular events.
In clinical trials and in real-world epidemiological studies, rofecoxib was associated with elevated heart attack, stroke, and related deaths; one 2005 study estimated that it had been responsible for some 38,000 excess deaths in the United States before being withdrawn. In 2007 Merck, beset with allegations that it had suppressed and mischaracterized rofecoxib’s safety data, paid out nearly $5 billion to settle thousands of lawsuits filed by patients and their families.
, an indication for which it received an orphan drug designation in 2017 and the agency’s green light for trials in 2020.
Brad Sippy, Tremeau’s chief executive officer, said that his company chose the two indications in part because both patient populations have low cardiovascular risk. Migraine patients are generally younger than the arthritis populations formerly treated with rofecoxib and are unlikely to take the drug for more than a day or 2 at time, avoiding the risks associated with extended exposure.
A crowded market
The past several years have seen the emergence of a cornucopia of new migraine treatments, including monoclonal antibodies such as erenumab (Aimovig, Amgen), which help prevent attacks by blocking the vasodilator calcitonin gene-related peptide, or CGRP. In addition to the standard arsenal of triptans and nonsteroidal anti-inflammatory drugs for acute pain relief, migraine patients can now choose among serotonin-blocking agents such as lasmiditan (Reyvow, Eli Lilly), known as “ditans,” and small-molecule CGRP antagonists such as ubrogepant (Ubrelvy, Abbie), known as “gepants.” Some NSAIDs, including one COX inhibitor, have been formulated into rapidly absorbed powders or liquids for migraine.
Mr. Sippy said he sees a role for rofecoxib even in this crowded space. “Migraine as you know is a multimodal situation – few people say that only one drug works for them,” he said. “We think this is an option that would basically be like a high dose of ibuprofen,” but with less frequent dosing and lower gastrointestinal and platelet effects compared with ibuprofen and other NSAIDs.
An improved formulation
Rofecoxib “crosses the blood brain barrier very readily – better than other COX inhibitors on the market,” Mr. Sippy added. “It was well absorbed in its original formulation, and our product is even better absorbed than the original – we estimate it’s probably an hour quicker to [peak concentration].” In addition, he said, “our formulation is more efficient at delivering the drug so we don’t need as much active ingredient – our 17.5 milligrams gets you the same systemic exposure as 25 milligrams of the old product.”
A different mechanism of action
Neurologist Alan M. Rapoport, MD, editor-in-chief of Neurology Reviews and professor of neurology at the University of California, Los Angeles, said that he was “cautiously optimistic” that “if used correctly and not too frequently, [rofecoxib] will find its niche in migraine treatment.”
“Patients liked Vioxx,” said Dr. Rapoport, past president of the International Headache Society. Even people currently on prevention “need to have an acute care drug handy.” While some patients on monoclonal antibodies have had success with gepants for acute care, “these both target the same pathway. It’s always nice to have options with a different mechanism of action.”
One of the arguments Tremeau has cited for reintroducing rofecoxib has been an urgent need for alternatives to opioid painkillers. Indeed some analysts have linked the demise of Vioxx with a subsequent increase in opioid prescribing.
Dr. Rapoport noted that he never prescribes opioids or butalbital, a barbiturate, for migraine, and that most headache specialists avoid them in clinical practice. But in the emergency setting, he said, patients receive them all too frequently.
Mr. Sippy said that opioid prescribing, while not unknown in migraine, was a bigger problem in hemophilic arthropathy, the first indication his company has pursued for rofecoxib. People with hemophilia “have a kind of arthritis that would respond well to an anti-inflammatory drug but they can’t take NSAIDs due to bleeding risk. This is why so many end up on opioids. Rofecoxib, as a COX-2 inhibitor, doesn’t have any effect on platelet aggregation, which would make it another option.”
No unique risks at prescribed doses
The migraine indication originally started out narrower: Patients with both migraine and bleeding disorders. “But in talking with the FDA, they encouraged us to develop it for migraine,” Mr. Sippy said. The company is considering pursuing a third indication: menstrual pain co-occurring with migraine. Tremeau has not ruled out seeking an indication in patients with arthritis who cannot take other painkillers, whether opioids or NSAIDs.
Five years ago, when Tremeau first announced its plans to bring rofecoxib back – indeed the company was set up for that purpose and has only this and another COX-2 inhibitor in development – some experts warned that there is little to prevent the drug from being used off-label, whether in higher doses or for other diseases.
“That’s something else we’re seeking to solve in addition to going for younger populations,” said Mr. Sippy, who worked at Merck during the Vioxx crisis and later headed neurology at Sunovion before starting his own company.
“We’re going for the former middle dose as our high dose and now we know that you don’t want to take more than the prescribed amount. If it doesn’t work you get off it; you don’t want to dose-creep on it. That’s been a key insight: At the appropriate dose, this product has no unique risk relative to the drug class and potentially some unique benefits,” he said.
Risk versus benefit
Joseph Ross, MD, a health policy researcher at Yale University in New Haven, Conn., who in a 2018 editorial expressed concerns about rofecoxib’s revival, said in an email that he felt its use in migraine could be justified, with caveats.
During Vioxx’s original approval and time on the market, “there was a cardiovascular risk associated with use that was not being transparently and clearly reported to patients and clinicians,” Dr. Ross said.
“In terms of testing the product for use in patients with migraine – a population of generally younger patients at lower risk of cardiovascular disease – my only concern is that the risk is clearly communicated and that there is adequate postmarket safety surveillance,” he said. “If patients are making fully informed decisions, the potential benefit of the drug with respect to pain control may be worth the risks.”
Dr. Rapoport serves as an adviser for AbbVie, Amgen, Biohaven, Cala Health, Collegium Pharmaceutical, Satsuma, Teva, Theranica and Xoc; he is on the speakers bureau of AbbVie, Amgen, Biohaven, Impel, Lundbeck, and Teva. Dr. Ross disclosed research support from Johnson and Johnson, the Medical Device Innovation Consortium, and the Laura and John Arnold Foundation, along with government grants; he is also an expert witness in a lawsuit against Biogen.
In June, the Concord, Mass.–based company Tremeau Pharmaceuticals announced that the Food and Drug Administration was letting it proceed with a phase 3 clinical trial to test rofecoxib, the once-bestselling painkiller known as Vioxx, in patients with migraine.
The anti-inflammatory drug, a cyclooxygenase-2 (COX-2) inhibitor, received its first FDA approval in 1999 and became widely prescribed for arthritis and acute pain. In 2004 it was withdrawn by its manufacturer, Merck, after being shown to raise the risk of cardiovascular events.
In clinical trials and in real-world epidemiological studies, rofecoxib was associated with elevated heart attack, stroke, and related deaths; one 2005 study estimated that it had been responsible for some 38,000 excess deaths in the United States before being withdrawn. In 2007 Merck, beset with allegations that it had suppressed and mischaracterized rofecoxib’s safety data, paid out nearly $5 billion to settle thousands of lawsuits filed by patients and their families.
, an indication for which it received an orphan drug designation in 2017 and the agency’s green light for trials in 2020.
Brad Sippy, Tremeau’s chief executive officer, said that his company chose the two indications in part because both patient populations have low cardiovascular risk. Migraine patients are generally younger than the arthritis populations formerly treated with rofecoxib and are unlikely to take the drug for more than a day or 2 at time, avoiding the risks associated with extended exposure.
A crowded market
The past several years have seen the emergence of a cornucopia of new migraine treatments, including monoclonal antibodies such as erenumab (Aimovig, Amgen), which help prevent attacks by blocking the vasodilator calcitonin gene-related peptide, or CGRP. In addition to the standard arsenal of triptans and nonsteroidal anti-inflammatory drugs for acute pain relief, migraine patients can now choose among serotonin-blocking agents such as lasmiditan (Reyvow, Eli Lilly), known as “ditans,” and small-molecule CGRP antagonists such as ubrogepant (Ubrelvy, Abbie), known as “gepants.” Some NSAIDs, including one COX inhibitor, have been formulated into rapidly absorbed powders or liquids for migraine.
Mr. Sippy said he sees a role for rofecoxib even in this crowded space. “Migraine as you know is a multimodal situation – few people say that only one drug works for them,” he said. “We think this is an option that would basically be like a high dose of ibuprofen,” but with less frequent dosing and lower gastrointestinal and platelet effects compared with ibuprofen and other NSAIDs.
An improved formulation
Rofecoxib “crosses the blood brain barrier very readily – better than other COX inhibitors on the market,” Mr. Sippy added. “It was well absorbed in its original formulation, and our product is even better absorbed than the original – we estimate it’s probably an hour quicker to [peak concentration].” In addition, he said, “our formulation is more efficient at delivering the drug so we don’t need as much active ingredient – our 17.5 milligrams gets you the same systemic exposure as 25 milligrams of the old product.”
A different mechanism of action
Neurologist Alan M. Rapoport, MD, editor-in-chief of Neurology Reviews and professor of neurology at the University of California, Los Angeles, said that he was “cautiously optimistic” that “if used correctly and not too frequently, [rofecoxib] will find its niche in migraine treatment.”
“Patients liked Vioxx,” said Dr. Rapoport, past president of the International Headache Society. Even people currently on prevention “need to have an acute care drug handy.” While some patients on monoclonal antibodies have had success with gepants for acute care, “these both target the same pathway. It’s always nice to have options with a different mechanism of action.”
One of the arguments Tremeau has cited for reintroducing rofecoxib has been an urgent need for alternatives to opioid painkillers. Indeed some analysts have linked the demise of Vioxx with a subsequent increase in opioid prescribing.
Dr. Rapoport noted that he never prescribes opioids or butalbital, a barbiturate, for migraine, and that most headache specialists avoid them in clinical practice. But in the emergency setting, he said, patients receive them all too frequently.
Mr. Sippy said that opioid prescribing, while not unknown in migraine, was a bigger problem in hemophilic arthropathy, the first indication his company has pursued for rofecoxib. People with hemophilia “have a kind of arthritis that would respond well to an anti-inflammatory drug but they can’t take NSAIDs due to bleeding risk. This is why so many end up on opioids. Rofecoxib, as a COX-2 inhibitor, doesn’t have any effect on platelet aggregation, which would make it another option.”
No unique risks at prescribed doses
The migraine indication originally started out narrower: Patients with both migraine and bleeding disorders. “But in talking with the FDA, they encouraged us to develop it for migraine,” Mr. Sippy said. The company is considering pursuing a third indication: menstrual pain co-occurring with migraine. Tremeau has not ruled out seeking an indication in patients with arthritis who cannot take other painkillers, whether opioids or NSAIDs.
Five years ago, when Tremeau first announced its plans to bring rofecoxib back – indeed the company was set up for that purpose and has only this and another COX-2 inhibitor in development – some experts warned that there is little to prevent the drug from being used off-label, whether in higher doses or for other diseases.
“That’s something else we’re seeking to solve in addition to going for younger populations,” said Mr. Sippy, who worked at Merck during the Vioxx crisis and later headed neurology at Sunovion before starting his own company.
“We’re going for the former middle dose as our high dose and now we know that you don’t want to take more than the prescribed amount. If it doesn’t work you get off it; you don’t want to dose-creep on it. That’s been a key insight: At the appropriate dose, this product has no unique risk relative to the drug class and potentially some unique benefits,” he said.
Risk versus benefit
Joseph Ross, MD, a health policy researcher at Yale University in New Haven, Conn., who in a 2018 editorial expressed concerns about rofecoxib’s revival, said in an email that he felt its use in migraine could be justified, with caveats.
During Vioxx’s original approval and time on the market, “there was a cardiovascular risk associated with use that was not being transparently and clearly reported to patients and clinicians,” Dr. Ross said.
“In terms of testing the product for use in patients with migraine – a population of generally younger patients at lower risk of cardiovascular disease – my only concern is that the risk is clearly communicated and that there is adequate postmarket safety surveillance,” he said. “If patients are making fully informed decisions, the potential benefit of the drug with respect to pain control may be worth the risks.”
Dr. Rapoport serves as an adviser for AbbVie, Amgen, Biohaven, Cala Health, Collegium Pharmaceutical, Satsuma, Teva, Theranica and Xoc; he is on the speakers bureau of AbbVie, Amgen, Biohaven, Impel, Lundbeck, and Teva. Dr. Ross disclosed research support from Johnson and Johnson, the Medical Device Innovation Consortium, and the Laura and John Arnold Foundation, along with government grants; he is also an expert witness in a lawsuit against Biogen.
In June, the Concord, Mass.–based company Tremeau Pharmaceuticals announced that the Food and Drug Administration was letting it proceed with a phase 3 clinical trial to test rofecoxib, the once-bestselling painkiller known as Vioxx, in patients with migraine.
The anti-inflammatory drug, a cyclooxygenase-2 (COX-2) inhibitor, received its first FDA approval in 1999 and became widely prescribed for arthritis and acute pain. In 2004 it was withdrawn by its manufacturer, Merck, after being shown to raise the risk of cardiovascular events.
In clinical trials and in real-world epidemiological studies, rofecoxib was associated with elevated heart attack, stroke, and related deaths; one 2005 study estimated that it had been responsible for some 38,000 excess deaths in the United States before being withdrawn. In 2007 Merck, beset with allegations that it had suppressed and mischaracterized rofecoxib’s safety data, paid out nearly $5 billion to settle thousands of lawsuits filed by patients and their families.
, an indication for which it received an orphan drug designation in 2017 and the agency’s green light for trials in 2020.
Brad Sippy, Tremeau’s chief executive officer, said that his company chose the two indications in part because both patient populations have low cardiovascular risk. Migraine patients are generally younger than the arthritis populations formerly treated with rofecoxib and are unlikely to take the drug for more than a day or 2 at time, avoiding the risks associated with extended exposure.
A crowded market
The past several years have seen the emergence of a cornucopia of new migraine treatments, including monoclonal antibodies such as erenumab (Aimovig, Amgen), which help prevent attacks by blocking the vasodilator calcitonin gene-related peptide, or CGRP. In addition to the standard arsenal of triptans and nonsteroidal anti-inflammatory drugs for acute pain relief, migraine patients can now choose among serotonin-blocking agents such as lasmiditan (Reyvow, Eli Lilly), known as “ditans,” and small-molecule CGRP antagonists such as ubrogepant (Ubrelvy, Abbie), known as “gepants.” Some NSAIDs, including one COX inhibitor, have been formulated into rapidly absorbed powders or liquids for migraine.
Mr. Sippy said he sees a role for rofecoxib even in this crowded space. “Migraine as you know is a multimodal situation – few people say that only one drug works for them,” he said. “We think this is an option that would basically be like a high dose of ibuprofen,” but with less frequent dosing and lower gastrointestinal and platelet effects compared with ibuprofen and other NSAIDs.
An improved formulation
Rofecoxib “crosses the blood brain barrier very readily – better than other COX inhibitors on the market,” Mr. Sippy added. “It was well absorbed in its original formulation, and our product is even better absorbed than the original – we estimate it’s probably an hour quicker to [peak concentration].” In addition, he said, “our formulation is more efficient at delivering the drug so we don’t need as much active ingredient – our 17.5 milligrams gets you the same systemic exposure as 25 milligrams of the old product.”
A different mechanism of action
Neurologist Alan M. Rapoport, MD, editor-in-chief of Neurology Reviews and professor of neurology at the University of California, Los Angeles, said that he was “cautiously optimistic” that “if used correctly and not too frequently, [rofecoxib] will find its niche in migraine treatment.”
“Patients liked Vioxx,” said Dr. Rapoport, past president of the International Headache Society. Even people currently on prevention “need to have an acute care drug handy.” While some patients on monoclonal antibodies have had success with gepants for acute care, “these both target the same pathway. It’s always nice to have options with a different mechanism of action.”
One of the arguments Tremeau has cited for reintroducing rofecoxib has been an urgent need for alternatives to opioid painkillers. Indeed some analysts have linked the demise of Vioxx with a subsequent increase in opioid prescribing.
Dr. Rapoport noted that he never prescribes opioids or butalbital, a barbiturate, for migraine, and that most headache specialists avoid them in clinical practice. But in the emergency setting, he said, patients receive them all too frequently.
Mr. Sippy said that opioid prescribing, while not unknown in migraine, was a bigger problem in hemophilic arthropathy, the first indication his company has pursued for rofecoxib. People with hemophilia “have a kind of arthritis that would respond well to an anti-inflammatory drug but they can’t take NSAIDs due to bleeding risk. This is why so many end up on opioids. Rofecoxib, as a COX-2 inhibitor, doesn’t have any effect on platelet aggregation, which would make it another option.”
No unique risks at prescribed doses
The migraine indication originally started out narrower: Patients with both migraine and bleeding disorders. “But in talking with the FDA, they encouraged us to develop it for migraine,” Mr. Sippy said. The company is considering pursuing a third indication: menstrual pain co-occurring with migraine. Tremeau has not ruled out seeking an indication in patients with arthritis who cannot take other painkillers, whether opioids or NSAIDs.
Five years ago, when Tremeau first announced its plans to bring rofecoxib back – indeed the company was set up for that purpose and has only this and another COX-2 inhibitor in development – some experts warned that there is little to prevent the drug from being used off-label, whether in higher doses or for other diseases.
“That’s something else we’re seeking to solve in addition to going for younger populations,” said Mr. Sippy, who worked at Merck during the Vioxx crisis and later headed neurology at Sunovion before starting his own company.
“We’re going for the former middle dose as our high dose and now we know that you don’t want to take more than the prescribed amount. If it doesn’t work you get off it; you don’t want to dose-creep on it. That’s been a key insight: At the appropriate dose, this product has no unique risk relative to the drug class and potentially some unique benefits,” he said.
Risk versus benefit
Joseph Ross, MD, a health policy researcher at Yale University in New Haven, Conn., who in a 2018 editorial expressed concerns about rofecoxib’s revival, said in an email that he felt its use in migraine could be justified, with caveats.
During Vioxx’s original approval and time on the market, “there was a cardiovascular risk associated with use that was not being transparently and clearly reported to patients and clinicians,” Dr. Ross said.
“In terms of testing the product for use in patients with migraine – a population of generally younger patients at lower risk of cardiovascular disease – my only concern is that the risk is clearly communicated and that there is adequate postmarket safety surveillance,” he said. “If patients are making fully informed decisions, the potential benefit of the drug with respect to pain control may be worth the risks.”
Dr. Rapoport serves as an adviser for AbbVie, Amgen, Biohaven, Cala Health, Collegium Pharmaceutical, Satsuma, Teva, Theranica and Xoc; he is on the speakers bureau of AbbVie, Amgen, Biohaven, Impel, Lundbeck, and Teva. Dr. Ross disclosed research support from Johnson and Johnson, the Medical Device Innovation Consortium, and the Laura and John Arnold Foundation, along with government grants; he is also an expert witness in a lawsuit against Biogen.
Study confirms increased CVT with AstraZeneca COVID vaccine
A new Scandinavian study has confirmed previous data showing increased rates of cerebral venous thrombosis and thrombocytopenia after the AstraZeneca COVID-19 vaccine.
The study also showed higher rates of several thromboembolic and thrombocytopenic outcomes after the Pfizer and Moderna mRNA vaccines, although these increases were less than the rates observed after the AstraZeneca vaccine, and sensitivity analyses were not consistent.
The researchers conclude that confirmatory analysis on the two mRNA vaccines by other methods are warranted.
The study was published in the June issue of JAMA Network Open.
“This study confirms what we know from other studies: that the AstraZeneca vaccine is associated with the rare but serious side effect of vaccine-induced immune thrombotic thrombocytopenia,” lead author Jacob Dag Berild, MD, Norwegian Institute of Public Health, Oslo, told this news organization.
he added.
Dr. Dag Berild noted that in the current study there was an excess of 1.6 events of cerebral venous thrombosis per 100,000 AstraZeneca vaccine doses, which is similar to what has been previously reported.
Asked how he saw these results affecting continued use of these vaccines, Dr. Dag Berild pointed out that the risk-benefit ratio of the vaccine depends on the risk of contracting COVID-19 and the risk for a severe outcome from COVID-19 weighed against the risk for an adverse event after vaccination.
“The European Medicines Agency has concluded that the overall risk-benefit ratio remains positive for the AstraZeneca vaccine, but Norway, Finland, and Denmark no longer use the AstraZeneca vaccine in their vaccination programs because of adequate availability of alternative vaccines. I think this is a reasonable decision,” he said.
For the current study, the researchers linked individual-level data separately from national population, patient, and vaccination registers in Norway, Finland, and Denmark. Patient registers were used to identify hospital visits and admissions related to thromboembolic and thrombocytopenic disease in all three countries.
The main outcomes were relative rates of coronary artery disease, coagulation disorders, and cerebrovascular disease in the 28-day period after vaccination, compared with the control period prior to vaccination.
The authors note that a strength of this study is the use of registers with full population coverage in three countries with universal health care, ensuring equal access to care for all permanent residents. At the end of the study period, from Jan. 1, 2020 to May 16, 2021, more than 5.3 million people in the three countries were vaccinated with one or two doses.
Another strength is the inherent adjustment for time-invariant confounders in the self-controlled case series design and the resulting control of confounders that can affect the more traditional observational studies when complete data for confounders are not available, they add.
Of the 265,339 hospital contacts, 43% were made by female patients and 93% by patients born in or before 1971, and 44% were for coronary artery disease, 21% for coagulation disorders, and 35% for cerebrovascular disease.
In the 28-day period after vaccination, there was an elevated rate of coronary artery disease after the Moderna vaccine (relative rate, 1.13) but not after the AstraZeneca (RR, 0.92) or Pfizer (RR, 0.96) vaccines.
There was an observed increase in the rate of coagulation disorders after all three vaccines (AstraZeneca RR, 2.01; Pfizer RR, 1.12; and Moderna RR, 1.26).
There was also an increase in the rate of cerebrovascular disease after all three vaccines (AstraZeneca RR, 1.32; Pfizer RR, 1.09; and Moderna RR, 1.21).
For individual diseases in the main outcomes, two notably high rates were observed after the AstraZeneca vaccine, with relative rates of 12.04 for cerebral venous thrombosis and 4.29 for thrombocytopenia, corresponding to 1.6 and 4.9 excess events per 100,000 doses, respectively.
The elevated risk after the AstraZeneca vaccine was consistent across all three countries and robust in sensitivity analyses.
The researchers report that they also observed statistically significant increases in hospital contacts for thrombocytopenic and thromboembolic events after the Pfizer and Moderna vaccines. However, the risk was smaller than after the AstraZeneca vaccine.
“Additionally, the national estimates varied, increased risk [was] observed only in the oldest cohorts, and sensitivity analysis checking underlying assumptions of the analyses were not consistent. Therefore, the overall and combined increased relative risks following the Pfizer and Moderna vaccinations should be interpreted with caution,” they say.
They note that their results with the AstraZeneca vaccine are in line with a comparison of observed and historic rates performed on partly the same population in Norway and Denmark and also with a Scottish national case-control study.
A version of this article first appeared on Medscape.com.
A new Scandinavian study has confirmed previous data showing increased rates of cerebral venous thrombosis and thrombocytopenia after the AstraZeneca COVID-19 vaccine.
The study also showed higher rates of several thromboembolic and thrombocytopenic outcomes after the Pfizer and Moderna mRNA vaccines, although these increases were less than the rates observed after the AstraZeneca vaccine, and sensitivity analyses were not consistent.
The researchers conclude that confirmatory analysis on the two mRNA vaccines by other methods are warranted.
The study was published in the June issue of JAMA Network Open.
“This study confirms what we know from other studies: that the AstraZeneca vaccine is associated with the rare but serious side effect of vaccine-induced immune thrombotic thrombocytopenia,” lead author Jacob Dag Berild, MD, Norwegian Institute of Public Health, Oslo, told this news organization.
he added.
Dr. Dag Berild noted that in the current study there was an excess of 1.6 events of cerebral venous thrombosis per 100,000 AstraZeneca vaccine doses, which is similar to what has been previously reported.
Asked how he saw these results affecting continued use of these vaccines, Dr. Dag Berild pointed out that the risk-benefit ratio of the vaccine depends on the risk of contracting COVID-19 and the risk for a severe outcome from COVID-19 weighed against the risk for an adverse event after vaccination.
“The European Medicines Agency has concluded that the overall risk-benefit ratio remains positive for the AstraZeneca vaccine, but Norway, Finland, and Denmark no longer use the AstraZeneca vaccine in their vaccination programs because of adequate availability of alternative vaccines. I think this is a reasonable decision,” he said.
For the current study, the researchers linked individual-level data separately from national population, patient, and vaccination registers in Norway, Finland, and Denmark. Patient registers were used to identify hospital visits and admissions related to thromboembolic and thrombocytopenic disease in all three countries.
The main outcomes were relative rates of coronary artery disease, coagulation disorders, and cerebrovascular disease in the 28-day period after vaccination, compared with the control period prior to vaccination.
The authors note that a strength of this study is the use of registers with full population coverage in three countries with universal health care, ensuring equal access to care for all permanent residents. At the end of the study period, from Jan. 1, 2020 to May 16, 2021, more than 5.3 million people in the three countries were vaccinated with one or two doses.
Another strength is the inherent adjustment for time-invariant confounders in the self-controlled case series design and the resulting control of confounders that can affect the more traditional observational studies when complete data for confounders are not available, they add.
Of the 265,339 hospital contacts, 43% were made by female patients and 93% by patients born in or before 1971, and 44% were for coronary artery disease, 21% for coagulation disorders, and 35% for cerebrovascular disease.
In the 28-day period after vaccination, there was an elevated rate of coronary artery disease after the Moderna vaccine (relative rate, 1.13) but not after the AstraZeneca (RR, 0.92) or Pfizer (RR, 0.96) vaccines.
There was an observed increase in the rate of coagulation disorders after all three vaccines (AstraZeneca RR, 2.01; Pfizer RR, 1.12; and Moderna RR, 1.26).
There was also an increase in the rate of cerebrovascular disease after all three vaccines (AstraZeneca RR, 1.32; Pfizer RR, 1.09; and Moderna RR, 1.21).
For individual diseases in the main outcomes, two notably high rates were observed after the AstraZeneca vaccine, with relative rates of 12.04 for cerebral venous thrombosis and 4.29 for thrombocytopenia, corresponding to 1.6 and 4.9 excess events per 100,000 doses, respectively.
The elevated risk after the AstraZeneca vaccine was consistent across all three countries and robust in sensitivity analyses.
The researchers report that they also observed statistically significant increases in hospital contacts for thrombocytopenic and thromboembolic events after the Pfizer and Moderna vaccines. However, the risk was smaller than after the AstraZeneca vaccine.
“Additionally, the national estimates varied, increased risk [was] observed only in the oldest cohorts, and sensitivity analysis checking underlying assumptions of the analyses were not consistent. Therefore, the overall and combined increased relative risks following the Pfizer and Moderna vaccinations should be interpreted with caution,” they say.
They note that their results with the AstraZeneca vaccine are in line with a comparison of observed and historic rates performed on partly the same population in Norway and Denmark and also with a Scottish national case-control study.
A version of this article first appeared on Medscape.com.
A new Scandinavian study has confirmed previous data showing increased rates of cerebral venous thrombosis and thrombocytopenia after the AstraZeneca COVID-19 vaccine.
The study also showed higher rates of several thromboembolic and thrombocytopenic outcomes after the Pfizer and Moderna mRNA vaccines, although these increases were less than the rates observed after the AstraZeneca vaccine, and sensitivity analyses were not consistent.
The researchers conclude that confirmatory analysis on the two mRNA vaccines by other methods are warranted.
The study was published in the June issue of JAMA Network Open.
“This study confirms what we know from other studies: that the AstraZeneca vaccine is associated with the rare but serious side effect of vaccine-induced immune thrombotic thrombocytopenia,” lead author Jacob Dag Berild, MD, Norwegian Institute of Public Health, Oslo, told this news organization.
he added.
Dr. Dag Berild noted that in the current study there was an excess of 1.6 events of cerebral venous thrombosis per 100,000 AstraZeneca vaccine doses, which is similar to what has been previously reported.
Asked how he saw these results affecting continued use of these vaccines, Dr. Dag Berild pointed out that the risk-benefit ratio of the vaccine depends on the risk of contracting COVID-19 and the risk for a severe outcome from COVID-19 weighed against the risk for an adverse event after vaccination.
“The European Medicines Agency has concluded that the overall risk-benefit ratio remains positive for the AstraZeneca vaccine, but Norway, Finland, and Denmark no longer use the AstraZeneca vaccine in their vaccination programs because of adequate availability of alternative vaccines. I think this is a reasonable decision,” he said.
For the current study, the researchers linked individual-level data separately from national population, patient, and vaccination registers in Norway, Finland, and Denmark. Patient registers were used to identify hospital visits and admissions related to thromboembolic and thrombocytopenic disease in all three countries.
The main outcomes were relative rates of coronary artery disease, coagulation disorders, and cerebrovascular disease in the 28-day period after vaccination, compared with the control period prior to vaccination.
The authors note that a strength of this study is the use of registers with full population coverage in three countries with universal health care, ensuring equal access to care for all permanent residents. At the end of the study period, from Jan. 1, 2020 to May 16, 2021, more than 5.3 million people in the three countries were vaccinated with one or two doses.
Another strength is the inherent adjustment for time-invariant confounders in the self-controlled case series design and the resulting control of confounders that can affect the more traditional observational studies when complete data for confounders are not available, they add.
Of the 265,339 hospital contacts, 43% were made by female patients and 93% by patients born in or before 1971, and 44% were for coronary artery disease, 21% for coagulation disorders, and 35% for cerebrovascular disease.
In the 28-day period after vaccination, there was an elevated rate of coronary artery disease after the Moderna vaccine (relative rate, 1.13) but not after the AstraZeneca (RR, 0.92) or Pfizer (RR, 0.96) vaccines.
There was an observed increase in the rate of coagulation disorders after all three vaccines (AstraZeneca RR, 2.01; Pfizer RR, 1.12; and Moderna RR, 1.26).
There was also an increase in the rate of cerebrovascular disease after all three vaccines (AstraZeneca RR, 1.32; Pfizer RR, 1.09; and Moderna RR, 1.21).
For individual diseases in the main outcomes, two notably high rates were observed after the AstraZeneca vaccine, with relative rates of 12.04 for cerebral venous thrombosis and 4.29 for thrombocytopenia, corresponding to 1.6 and 4.9 excess events per 100,000 doses, respectively.
The elevated risk after the AstraZeneca vaccine was consistent across all three countries and robust in sensitivity analyses.
The researchers report that they also observed statistically significant increases in hospital contacts for thrombocytopenic and thromboembolic events after the Pfizer and Moderna vaccines. However, the risk was smaller than after the AstraZeneca vaccine.
“Additionally, the national estimates varied, increased risk [was] observed only in the oldest cohorts, and sensitivity analysis checking underlying assumptions of the analyses were not consistent. Therefore, the overall and combined increased relative risks following the Pfizer and Moderna vaccinations should be interpreted with caution,” they say.
They note that their results with the AstraZeneca vaccine are in line with a comparison of observed and historic rates performed on partly the same population in Norway and Denmark and also with a Scottish national case-control study.
A version of this article first appeared on Medscape.com.
FDA warns of increased risk of death with CLL, lymphoma drug
Duvelisib was approved in 2018 to treat adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who had received at least two prior therapies that did not work or stopped working.
However, more recent 5-year overall survival results from the randomized phase 3 DUO clinical trial found a possible increased risk of death with duvelisib compared with another drug used to treat leukemia and lymphoma, according to an FDA Drug Safety Communication.
“The trial also found Copiktra was associated with a higher risk of serious side effects, including infections, diarrhea, inflammation of the intestines and lungs, skin reactions, and high liver enzyme levels in the blood,” states the warning, which advises prescribers to weigh the risks and benefits of continued use versus use of other treatments.
More specifically, median 5-year overall survival among 319 patients with CLL or SLL in the DUO trial was 52.3 months with duvelisib versus 63.3 months with the monoclonal antibody ofatumumab (hazard ratio, 1.09 overall and 1.06 among patients who received at least two prior lines of therapy).
Serious adverse events of grade 3 or higher were also more common in those treated with duvelisib.
Of note, in April, the FDA also announced it was withdrawing approval of the relapsed or refractory follicular lymphoma indication for duvelisib, following a voluntary request by the drug manufacturer, Secura Bio Inc.
A public meeting will be scheduled to discuss the findings of the trial and whether the drug should continue to be prescribed.
This FDA warning follows the agency’s June 1 withdrawal of approval for umbralisib (Ukoniq), another PI3 kinase inhibitor, following an investigation into a “possible increased risk of death.”
As reported by Medscape, umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
“These safety findings were similar for other medicines in the same PI3 kinase inhibitor class, which were discussed at an advisory committee meeting of non-FDA experts in April 2022,” according to the FDA warning.
The FDA urges patients and health care professionals to report side effects involving duvelisib or other medicines to the FDA MedWatch program.
A version of this article first appeared on Medscape.com.
Duvelisib was approved in 2018 to treat adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who had received at least two prior therapies that did not work or stopped working.
However, more recent 5-year overall survival results from the randomized phase 3 DUO clinical trial found a possible increased risk of death with duvelisib compared with another drug used to treat leukemia and lymphoma, according to an FDA Drug Safety Communication.
“The trial also found Copiktra was associated with a higher risk of serious side effects, including infections, diarrhea, inflammation of the intestines and lungs, skin reactions, and high liver enzyme levels in the blood,” states the warning, which advises prescribers to weigh the risks and benefits of continued use versus use of other treatments.
More specifically, median 5-year overall survival among 319 patients with CLL or SLL in the DUO trial was 52.3 months with duvelisib versus 63.3 months with the monoclonal antibody ofatumumab (hazard ratio, 1.09 overall and 1.06 among patients who received at least two prior lines of therapy).
Serious adverse events of grade 3 or higher were also more common in those treated with duvelisib.
Of note, in April, the FDA also announced it was withdrawing approval of the relapsed or refractory follicular lymphoma indication for duvelisib, following a voluntary request by the drug manufacturer, Secura Bio Inc.
A public meeting will be scheduled to discuss the findings of the trial and whether the drug should continue to be prescribed.
This FDA warning follows the agency’s June 1 withdrawal of approval for umbralisib (Ukoniq), another PI3 kinase inhibitor, following an investigation into a “possible increased risk of death.”
As reported by Medscape, umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
“These safety findings were similar for other medicines in the same PI3 kinase inhibitor class, which were discussed at an advisory committee meeting of non-FDA experts in April 2022,” according to the FDA warning.
The FDA urges patients and health care professionals to report side effects involving duvelisib or other medicines to the FDA MedWatch program.
A version of this article first appeared on Medscape.com.
Duvelisib was approved in 2018 to treat adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who had received at least two prior therapies that did not work or stopped working.
However, more recent 5-year overall survival results from the randomized phase 3 DUO clinical trial found a possible increased risk of death with duvelisib compared with another drug used to treat leukemia and lymphoma, according to an FDA Drug Safety Communication.
“The trial also found Copiktra was associated with a higher risk of serious side effects, including infections, diarrhea, inflammation of the intestines and lungs, skin reactions, and high liver enzyme levels in the blood,” states the warning, which advises prescribers to weigh the risks and benefits of continued use versus use of other treatments.
More specifically, median 5-year overall survival among 319 patients with CLL or SLL in the DUO trial was 52.3 months with duvelisib versus 63.3 months with the monoclonal antibody ofatumumab (hazard ratio, 1.09 overall and 1.06 among patients who received at least two prior lines of therapy).
Serious adverse events of grade 3 or higher were also more common in those treated with duvelisib.
Of note, in April, the FDA also announced it was withdrawing approval of the relapsed or refractory follicular lymphoma indication for duvelisib, following a voluntary request by the drug manufacturer, Secura Bio Inc.
A public meeting will be scheduled to discuss the findings of the trial and whether the drug should continue to be prescribed.
This FDA warning follows the agency’s June 1 withdrawal of approval for umbralisib (Ukoniq), another PI3 kinase inhibitor, following an investigation into a “possible increased risk of death.”
As reported by Medscape, umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
“These safety findings were similar for other medicines in the same PI3 kinase inhibitor class, which were discussed at an advisory committee meeting of non-FDA experts in April 2022,” according to the FDA warning.
The FDA urges patients and health care professionals to report side effects involving duvelisib or other medicines to the FDA MedWatch program.
A version of this article first appeared on Medscape.com.
FDA warning: Lymphoma drug heightens risk of death
The U.S. Food and Drug Administration issued a warning today that the cancer drug duvelisib (Copiktra, Verastem), a PI3 kinase inhibitor, may increase the risk of death and serious side effects.
Duvelisib was approved in 2018 to treat adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who had received at least two prior therapies that did not work or stopped working.
However, more recent 5-year overall survival results from the randomized phase 3 DUO clinical trial found a possible increased risk of death with duvelisib, compared with another drug used to treat leukemia and lymphoma, according to an FDA Drug Safety Communication.
“The trial also found Copiktra was associated with a higher risk of serious side effects, including infections, diarrhea, inflammation of the intestines and lungs, skin reactions, and high liver enzyme levels in the blood,” states the warning, which advises prescribers to weigh the risks and benefits of continued use versus use of other treatments.
More specifically, median 5-year overall survival among 319 patients with CLL or SLL in the DUO trial was 52.3 months with duvelisib versus 63.3 months with the monoclonal antibody ofatumumab (hazard ratio, 1.09 overall and 1.06 among patients who received at least two prior lines of therapy).
Serious adverse events of grade 3 or higher were also more common in those treated with duvelisib.
Of note, in April, the FDA also announced that it was withdrawing approval of the relapsed or refractory follicular lymphoma indication for duvelisib following a voluntary request by the drug manufacturer Secura Bio.
A public meeting will be scheduled to discuss the findings of the trial and whether the drug should continue to be prescribed.
This FDA warning follows the agency’s June 1 withdrawal of approval for umbralisib (Ukoniq), another PI3 kinase inhibitor, following an investigation into a “possible increased risk of death.”
As reported by this news organization, umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
“These safety findings were similar for other medicines in the same PI3 kinase inhibitor class, which were discussed at an advisory committee meeting of non-FDA experts in April 2022,” according to the FDA warning.
The FDA urges patients and health care professionals to report side effects involving duvelisib or other medicines to the FDA MedWatch program.
A version of this article first appeared on Medscape.com
The U.S. Food and Drug Administration issued a warning today that the cancer drug duvelisib (Copiktra, Verastem), a PI3 kinase inhibitor, may increase the risk of death and serious side effects.
Duvelisib was approved in 2018 to treat adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who had received at least two prior therapies that did not work or stopped working.
However, more recent 5-year overall survival results from the randomized phase 3 DUO clinical trial found a possible increased risk of death with duvelisib, compared with another drug used to treat leukemia and lymphoma, according to an FDA Drug Safety Communication.
“The trial also found Copiktra was associated with a higher risk of serious side effects, including infections, diarrhea, inflammation of the intestines and lungs, skin reactions, and high liver enzyme levels in the blood,” states the warning, which advises prescribers to weigh the risks and benefits of continued use versus use of other treatments.
More specifically, median 5-year overall survival among 319 patients with CLL or SLL in the DUO trial was 52.3 months with duvelisib versus 63.3 months with the monoclonal antibody ofatumumab (hazard ratio, 1.09 overall and 1.06 among patients who received at least two prior lines of therapy).
Serious adverse events of grade 3 or higher were also more common in those treated with duvelisib.
Of note, in April, the FDA also announced that it was withdrawing approval of the relapsed or refractory follicular lymphoma indication for duvelisib following a voluntary request by the drug manufacturer Secura Bio.
A public meeting will be scheduled to discuss the findings of the trial and whether the drug should continue to be prescribed.
This FDA warning follows the agency’s June 1 withdrawal of approval for umbralisib (Ukoniq), another PI3 kinase inhibitor, following an investigation into a “possible increased risk of death.”
As reported by this news organization, umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
“These safety findings were similar for other medicines in the same PI3 kinase inhibitor class, which were discussed at an advisory committee meeting of non-FDA experts in April 2022,” according to the FDA warning.
The FDA urges patients and health care professionals to report side effects involving duvelisib or other medicines to the FDA MedWatch program.
A version of this article first appeared on Medscape.com
The U.S. Food and Drug Administration issued a warning today that the cancer drug duvelisib (Copiktra, Verastem), a PI3 kinase inhibitor, may increase the risk of death and serious side effects.
Duvelisib was approved in 2018 to treat adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who had received at least two prior therapies that did not work or stopped working.
However, more recent 5-year overall survival results from the randomized phase 3 DUO clinical trial found a possible increased risk of death with duvelisib, compared with another drug used to treat leukemia and lymphoma, according to an FDA Drug Safety Communication.
“The trial also found Copiktra was associated with a higher risk of serious side effects, including infections, diarrhea, inflammation of the intestines and lungs, skin reactions, and high liver enzyme levels in the blood,” states the warning, which advises prescribers to weigh the risks and benefits of continued use versus use of other treatments.
More specifically, median 5-year overall survival among 319 patients with CLL or SLL in the DUO trial was 52.3 months with duvelisib versus 63.3 months with the monoclonal antibody ofatumumab (hazard ratio, 1.09 overall and 1.06 among patients who received at least two prior lines of therapy).
Serious adverse events of grade 3 or higher were also more common in those treated with duvelisib.
Of note, in April, the FDA also announced that it was withdrawing approval of the relapsed or refractory follicular lymphoma indication for duvelisib following a voluntary request by the drug manufacturer Secura Bio.
A public meeting will be scheduled to discuss the findings of the trial and whether the drug should continue to be prescribed.
This FDA warning follows the agency’s June 1 withdrawal of approval for umbralisib (Ukoniq), another PI3 kinase inhibitor, following an investigation into a “possible increased risk of death.”
As reported by this news organization, umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
“These safety findings were similar for other medicines in the same PI3 kinase inhibitor class, which were discussed at an advisory committee meeting of non-FDA experts in April 2022,” according to the FDA warning.
The FDA urges patients and health care professionals to report side effects involving duvelisib or other medicines to the FDA MedWatch program.
A version of this article first appeared on Medscape.com
Lawmakers argue for changes in prior authorization processes
Republican and Democratic members of the House called for changes in how insurer-run Medicare plans manage the prior authorization process, following testimony from a federal watchdog organization about improper denials of payment for care.
About 18% of payment denials in a sample examined by the Office of Inspector General (OIG) of the Department of Health and Human Services (HHS) either met Medicare coverage rules or the rules of the insurance plan.
As such, they should not have been denied, according to the OIG. That was the finding of an April OIG report, based on a sample of 2019 denials from large insurer-run Medicare plans.
Erin Bliss, an assistant inspector general with the OIG, appeared as a witness at a June 28 Energy and Commerce Subcommittee on Oversight and Investigations hearing to discuss this investigation and other issues with prior authorization and insurer-run Medicare, also known as the Advantage plans.
Most of these payment denials of appropriate services were due to human error during manual claims-processing reviews, Ms. Bliss told the subcommittee, such as overlooking a document, and to system processing errors, such as a Medicare insurance plan failing to program or update a system correctly.
In many cases, these denials were reversed, but patient care was still disrupted and clinicians lost time chasing clearances for services that plans already had covered, Ms. Bliss said in her testimony.
The April report was not the OIG’s first look into concerns about insurer-run plans inappropriately denying care through prior authorizations. The OIG in 2018 reported that insurer-run Medicare plans overturned 75% of their own denials during 2014-2016 when patients and clinicians appealed these decisions, overturning approximately 216,000 denials each year.
‘Numerous hoops’ unnecessary for doctors, patients
Lawmakers at the hearing supported the idea of the need for prior authorization as a screening tool to prevent unneeded care.
But they chided insurance companies for their execution of this process, with clinicians and patients often frustrated by complex steps needed. Medicare Advantage plans sometimes require prior authorization for “relatively standard medical services,” said Subcommittee on Oversight and Investigations Chair Diana DeGette (D-Colo.).
“Our seniors and their doctors should not be required to jump through numerous hoops to ensure coverage for straightforward and medically necessary procedures,” Rep. DeGette said.
Several lawmakers spoke at the hearing about the need for changes to prior authorization, including calling for action on a pending bill intended to compel insurers to streamline the review process. The Improving Seniors’ Timely Access to Care Act of 2021 already has attracted more than 300 bipartisan sponsors. A companion Senate bill has more than 30 sponsors.
The bill’s aim is to shift this process away from faxes and phone calls while also encouraging plans to adhere to evidence-based medical guidelines in consultation with physicians. The bill calls for the establishment of an electronic prior authorization program that could issue real-time decisions.
“The result will be less administrative burden for providers and more information in the hands of patients. It will allow more patients to receive care when they need it, reducing the likelihood of additional, often more severe complications,” said Rep. Larry Bucshon, MD, (R-Ind.) who is among the active sponsors of the bill.
“In the long term, I believe it would also result in cost savings for the health care system at large by identifying problems earlier and getting them treated before their patients have more complications,” Rep. Bucshon added.
Finding ‘room for improvement’ for prior authorizations
There’s strong bipartisan support in Congress for insurer-run Medicare, which has grown by 10% per year over the last several years and has doubled since 2010, according to the Medicare Payment Advisory Commission (MedPAC). About 27 million people are now enrolled in these plans.
But for that reason, insurer-run Medicare may also need more careful watching, lawmakers made clear at the hearing.
“We’ve heard quite a bit of evidence today that there is room for improvement,” said Rep. Bucshon, a strong supporter of insurer-run Medicare, which can offer patients added benefits such as dental coverage.
Rep. Ann Kuster (D-N.H.) said simplifying prior authorization would reduce stress on clinicians already dealing with burnout.
“They’re just so tired of all this paperwork and red tape,” Rep. Kuster said. “In 2022 can’t we at least consider electronic prior authorization?”
At the hearing, Rep. Michael C. Burgess, MD, (R-Tex.) noted that his home state already has taken a step toward reducing the burden of prior authorization with its “gold card” program.
In 2021, a new Texas law called on the state department of insurance to develop rules to require health plans to provide an exemption from preauthorization requirements for a particular health care service if the issuer has approved, or would have approved, at least 90% of the preauthorization requests submitted by the physician or provider for that service. The law also mandates that a physician participating in a peer-to-peer review on behalf of a health benefit plan issuer must be a Texas-licensed physician who has the same or similar specialty as the physician or clinician requesting the service, according to the state insurance department.
Separately, Rep. Suzan DelBene (D-Wash.), the sponsor of the Improving Seniors’ Timely Access to Care Act, told the American Medical Association in a recent interview that she expects the House Ways and Means Committee, on which she serves, to mark up her bill in July. (A mark-up is the process by which a House or Senate committee considers and often amends a bill and then sends it to the chamber’s leadership for a floor vote.)
In a statement issued about the hearing, America’s Health Insurance Plans (AHIP) noted that there has been work in recent years toward streamlining prior authorization. AHIP said it launched the Fast Prior Authorization Technology Highway (Fast PATH) initiative in 2020 to study electronic procedures for handling these reviews.
“The findings of this study showed that ePA delivered improvements with a strong majority of experienced providers reporting faster time to patient care, fewer phone calls and faxes, better understanding of [prior authorization] requirements, and faster time to decisions,” AHIP said.
A version of this article first appeared on Medscape.com.
Republican and Democratic members of the House called for changes in how insurer-run Medicare plans manage the prior authorization process, following testimony from a federal watchdog organization about improper denials of payment for care.
About 18% of payment denials in a sample examined by the Office of Inspector General (OIG) of the Department of Health and Human Services (HHS) either met Medicare coverage rules or the rules of the insurance plan.
As such, they should not have been denied, according to the OIG. That was the finding of an April OIG report, based on a sample of 2019 denials from large insurer-run Medicare plans.
Erin Bliss, an assistant inspector general with the OIG, appeared as a witness at a June 28 Energy and Commerce Subcommittee on Oversight and Investigations hearing to discuss this investigation and other issues with prior authorization and insurer-run Medicare, also known as the Advantage plans.
Most of these payment denials of appropriate services were due to human error during manual claims-processing reviews, Ms. Bliss told the subcommittee, such as overlooking a document, and to system processing errors, such as a Medicare insurance plan failing to program or update a system correctly.
In many cases, these denials were reversed, but patient care was still disrupted and clinicians lost time chasing clearances for services that plans already had covered, Ms. Bliss said in her testimony.
The April report was not the OIG’s first look into concerns about insurer-run plans inappropriately denying care through prior authorizations. The OIG in 2018 reported that insurer-run Medicare plans overturned 75% of their own denials during 2014-2016 when patients and clinicians appealed these decisions, overturning approximately 216,000 denials each year.
‘Numerous hoops’ unnecessary for doctors, patients
Lawmakers at the hearing supported the idea of the need for prior authorization as a screening tool to prevent unneeded care.
But they chided insurance companies for their execution of this process, with clinicians and patients often frustrated by complex steps needed. Medicare Advantage plans sometimes require prior authorization for “relatively standard medical services,” said Subcommittee on Oversight and Investigations Chair Diana DeGette (D-Colo.).
“Our seniors and their doctors should not be required to jump through numerous hoops to ensure coverage for straightforward and medically necessary procedures,” Rep. DeGette said.
Several lawmakers spoke at the hearing about the need for changes to prior authorization, including calling for action on a pending bill intended to compel insurers to streamline the review process. The Improving Seniors’ Timely Access to Care Act of 2021 already has attracted more than 300 bipartisan sponsors. A companion Senate bill has more than 30 sponsors.
The bill’s aim is to shift this process away from faxes and phone calls while also encouraging plans to adhere to evidence-based medical guidelines in consultation with physicians. The bill calls for the establishment of an electronic prior authorization program that could issue real-time decisions.
“The result will be less administrative burden for providers and more information in the hands of patients. It will allow more patients to receive care when they need it, reducing the likelihood of additional, often more severe complications,” said Rep. Larry Bucshon, MD, (R-Ind.) who is among the active sponsors of the bill.
“In the long term, I believe it would also result in cost savings for the health care system at large by identifying problems earlier and getting them treated before their patients have more complications,” Rep. Bucshon added.
Finding ‘room for improvement’ for prior authorizations
There’s strong bipartisan support in Congress for insurer-run Medicare, which has grown by 10% per year over the last several years and has doubled since 2010, according to the Medicare Payment Advisory Commission (MedPAC). About 27 million people are now enrolled in these plans.
But for that reason, insurer-run Medicare may also need more careful watching, lawmakers made clear at the hearing.
“We’ve heard quite a bit of evidence today that there is room for improvement,” said Rep. Bucshon, a strong supporter of insurer-run Medicare, which can offer patients added benefits such as dental coverage.
Rep. Ann Kuster (D-N.H.) said simplifying prior authorization would reduce stress on clinicians already dealing with burnout.
“They’re just so tired of all this paperwork and red tape,” Rep. Kuster said. “In 2022 can’t we at least consider electronic prior authorization?”
At the hearing, Rep. Michael C. Burgess, MD, (R-Tex.) noted that his home state already has taken a step toward reducing the burden of prior authorization with its “gold card” program.
In 2021, a new Texas law called on the state department of insurance to develop rules to require health plans to provide an exemption from preauthorization requirements for a particular health care service if the issuer has approved, or would have approved, at least 90% of the preauthorization requests submitted by the physician or provider for that service. The law also mandates that a physician participating in a peer-to-peer review on behalf of a health benefit plan issuer must be a Texas-licensed physician who has the same or similar specialty as the physician or clinician requesting the service, according to the state insurance department.
Separately, Rep. Suzan DelBene (D-Wash.), the sponsor of the Improving Seniors’ Timely Access to Care Act, told the American Medical Association in a recent interview that she expects the House Ways and Means Committee, on which she serves, to mark up her bill in July. (A mark-up is the process by which a House or Senate committee considers and often amends a bill and then sends it to the chamber’s leadership for a floor vote.)
In a statement issued about the hearing, America’s Health Insurance Plans (AHIP) noted that there has been work in recent years toward streamlining prior authorization. AHIP said it launched the Fast Prior Authorization Technology Highway (Fast PATH) initiative in 2020 to study electronic procedures for handling these reviews.
“The findings of this study showed that ePA delivered improvements with a strong majority of experienced providers reporting faster time to patient care, fewer phone calls and faxes, better understanding of [prior authorization] requirements, and faster time to decisions,” AHIP said.
A version of this article first appeared on Medscape.com.
Republican and Democratic members of the House called for changes in how insurer-run Medicare plans manage the prior authorization process, following testimony from a federal watchdog organization about improper denials of payment for care.
About 18% of payment denials in a sample examined by the Office of Inspector General (OIG) of the Department of Health and Human Services (HHS) either met Medicare coverage rules or the rules of the insurance plan.
As such, they should not have been denied, according to the OIG. That was the finding of an April OIG report, based on a sample of 2019 denials from large insurer-run Medicare plans.
Erin Bliss, an assistant inspector general with the OIG, appeared as a witness at a June 28 Energy and Commerce Subcommittee on Oversight and Investigations hearing to discuss this investigation and other issues with prior authorization and insurer-run Medicare, also known as the Advantage plans.
Most of these payment denials of appropriate services were due to human error during manual claims-processing reviews, Ms. Bliss told the subcommittee, such as overlooking a document, and to system processing errors, such as a Medicare insurance plan failing to program or update a system correctly.
In many cases, these denials were reversed, but patient care was still disrupted and clinicians lost time chasing clearances for services that plans already had covered, Ms. Bliss said in her testimony.
The April report was not the OIG’s first look into concerns about insurer-run plans inappropriately denying care through prior authorizations. The OIG in 2018 reported that insurer-run Medicare plans overturned 75% of their own denials during 2014-2016 when patients and clinicians appealed these decisions, overturning approximately 216,000 denials each year.
‘Numerous hoops’ unnecessary for doctors, patients
Lawmakers at the hearing supported the idea of the need for prior authorization as a screening tool to prevent unneeded care.
But they chided insurance companies for their execution of this process, with clinicians and patients often frustrated by complex steps needed. Medicare Advantage plans sometimes require prior authorization for “relatively standard medical services,” said Subcommittee on Oversight and Investigations Chair Diana DeGette (D-Colo.).
“Our seniors and their doctors should not be required to jump through numerous hoops to ensure coverage for straightforward and medically necessary procedures,” Rep. DeGette said.
Several lawmakers spoke at the hearing about the need for changes to prior authorization, including calling for action on a pending bill intended to compel insurers to streamline the review process. The Improving Seniors’ Timely Access to Care Act of 2021 already has attracted more than 300 bipartisan sponsors. A companion Senate bill has more than 30 sponsors.
The bill’s aim is to shift this process away from faxes and phone calls while also encouraging plans to adhere to evidence-based medical guidelines in consultation with physicians. The bill calls for the establishment of an electronic prior authorization program that could issue real-time decisions.
“The result will be less administrative burden for providers and more information in the hands of patients. It will allow more patients to receive care when they need it, reducing the likelihood of additional, often more severe complications,” said Rep. Larry Bucshon, MD, (R-Ind.) who is among the active sponsors of the bill.
“In the long term, I believe it would also result in cost savings for the health care system at large by identifying problems earlier and getting them treated before their patients have more complications,” Rep. Bucshon added.
Finding ‘room for improvement’ for prior authorizations
There’s strong bipartisan support in Congress for insurer-run Medicare, which has grown by 10% per year over the last several years and has doubled since 2010, according to the Medicare Payment Advisory Commission (MedPAC). About 27 million people are now enrolled in these plans.
But for that reason, insurer-run Medicare may also need more careful watching, lawmakers made clear at the hearing.
“We’ve heard quite a bit of evidence today that there is room for improvement,” said Rep. Bucshon, a strong supporter of insurer-run Medicare, which can offer patients added benefits such as dental coverage.
Rep. Ann Kuster (D-N.H.) said simplifying prior authorization would reduce stress on clinicians already dealing with burnout.
“They’re just so tired of all this paperwork and red tape,” Rep. Kuster said. “In 2022 can’t we at least consider electronic prior authorization?”
At the hearing, Rep. Michael C. Burgess, MD, (R-Tex.) noted that his home state already has taken a step toward reducing the burden of prior authorization with its “gold card” program.
In 2021, a new Texas law called on the state department of insurance to develop rules to require health plans to provide an exemption from preauthorization requirements for a particular health care service if the issuer has approved, or would have approved, at least 90% of the preauthorization requests submitted by the physician or provider for that service. The law also mandates that a physician participating in a peer-to-peer review on behalf of a health benefit plan issuer must be a Texas-licensed physician who has the same or similar specialty as the physician or clinician requesting the service, according to the state insurance department.
Separately, Rep. Suzan DelBene (D-Wash.), the sponsor of the Improving Seniors’ Timely Access to Care Act, told the American Medical Association in a recent interview that she expects the House Ways and Means Committee, on which she serves, to mark up her bill in July. (A mark-up is the process by which a House or Senate committee considers and often amends a bill and then sends it to the chamber’s leadership for a floor vote.)
In a statement issued about the hearing, America’s Health Insurance Plans (AHIP) noted that there has been work in recent years toward streamlining prior authorization. AHIP said it launched the Fast Prior Authorization Technology Highway (Fast PATH) initiative in 2020 to study electronic procedures for handling these reviews.
“The findings of this study showed that ePA delivered improvements with a strong majority of experienced providers reporting faster time to patient care, fewer phone calls and faxes, better understanding of [prior authorization] requirements, and faster time to decisions,” AHIP said.
A version of this article first appeared on Medscape.com.
Murder of physician raises the stress level for all clinicians
As if it weren’t enough that doctors work in a profession where it’s almost more a question of when they’ll be sued than if they’ll be sued – where COVID, staff shortages, long hours, and patients frustrated over canceled procedures have caused unrelenting fatigue and stress – they now have to worry that an unhappy patient is going to buy a gun, walk into their office, and kill them.
That’s exactly what happened in Tulsa, Okla., where a patient complaining of pain after back surgery murdered his doctor and several others who happened to be in the wrong place at the wrong time.
The temptation in the aftermath of such tragedies is to think about preventive measures: Make medical facilities “hardened” targets, like schools have become, with armed guards, metal detectors, automatically locking doors, physical barriers within, security cameras, and buzzers for entry – although hardening a large medical center where members of the community routinely come and go would be challenging.
What about the enormous stress on doctors, nurses, and others in the medical workplace? Physicians who have been sued for malpractice often describe how it changes the way they interact with patients: They now size patients up and make judgments about their potential litigiousness. Will the physicians now look over their patients’ shoulders at the video feed from a security camera when they’re taking a history? Will medical professionals be forced to make snap judgments about patients’ psychological state before deciding whether to treat them?
Remember, there was a time when school shootings were unimaginable. Once one person crosses that line, others inevitably follow.
It could be a drug-seeking patient complaining of ongoing pain, angry because he can’t get a new prescription. It could be a patient whose unpaid bill was turned over to a collection agency, angry because he’s now getting calls from collectors. It could be someone who blames a physician for the loss of a loved one. It could be someone who would otherwise have filed a lawsuit, who now thinks he has a more effective option for exacting retribution.
Most of us would find it unbearable to live and work under the kind of stress faced by medical professionals today. And unfortunately, there is no short-term, systemic relief on the horizon. But there are methods of relieving at least some of the psychological burden being carried by these dedicated individuals.
For starters, the government should provide funds to improve safety and security at medical facilities. It’s sad but it’s a fact of life. The physical structure of schools, along with emergency procedures, have been changed since Columbine and Sandy Hook, and our children and their teachers undergo active shooter drills. Health care facilities will need to adopt similar strategies.
But if we don’t also support the individuals who work in health care, we’ll no longer have even partially staffed health care facilities. Hospitals and medical groups need to be conscious of the effects stress may have on them. Medical staff and administrators need to recognize changes in their colleagues’ behavior and refer those cohorts to professional stress coaches who can get them back on track.
Medical personnel should be picking up on warning signs, like irritability, depression, sudden weight gain or loss, lack of motivation and job satisfaction, obsessiveness, unusual levels of fatigue, alcohol or drug use, and, of course, avoidable medical errors.
In addition, colleagues in the medical workplace need to know each other well. They are usually the first ones to notice if something is off and may be in the best position to refer coworkers for help. Also, medical malpractice insurance carriers should consider encouraging and covering coaching sessions, because helping physicians cope with this heightened stress will prevent medical errors and the lawsuits that inevitably accompany mistakes.
This needn’t be a long-term process like ongoing psychotherapy; a few sessions with a well-trained coach may help psychologically challenged peers restore their focus and perspective. It won’t eliminate the threat any more than litigation stress coaching eliminates the threat of being sued, but it can prevent that stress from leading to avoidable errors. It also can prevent physicians’ personal lives and relationships from going off the rails and driving them out of the medical profession.
None of us can afford to ignore the impacts that these new stressors are having and simply act as if it’s business as usual. The people in the trenches need our help.
Ms. Fiore is President of Winning Focus in Murrysville, Pa. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
As if it weren’t enough that doctors work in a profession where it’s almost more a question of when they’ll be sued than if they’ll be sued – where COVID, staff shortages, long hours, and patients frustrated over canceled procedures have caused unrelenting fatigue and stress – they now have to worry that an unhappy patient is going to buy a gun, walk into their office, and kill them.
That’s exactly what happened in Tulsa, Okla., where a patient complaining of pain after back surgery murdered his doctor and several others who happened to be in the wrong place at the wrong time.
The temptation in the aftermath of such tragedies is to think about preventive measures: Make medical facilities “hardened” targets, like schools have become, with armed guards, metal detectors, automatically locking doors, physical barriers within, security cameras, and buzzers for entry – although hardening a large medical center where members of the community routinely come and go would be challenging.
What about the enormous stress on doctors, nurses, and others in the medical workplace? Physicians who have been sued for malpractice often describe how it changes the way they interact with patients: They now size patients up and make judgments about their potential litigiousness. Will the physicians now look over their patients’ shoulders at the video feed from a security camera when they’re taking a history? Will medical professionals be forced to make snap judgments about patients’ psychological state before deciding whether to treat them?
Remember, there was a time when school shootings were unimaginable. Once one person crosses that line, others inevitably follow.
It could be a drug-seeking patient complaining of ongoing pain, angry because he can’t get a new prescription. It could be a patient whose unpaid bill was turned over to a collection agency, angry because he’s now getting calls from collectors. It could be someone who blames a physician for the loss of a loved one. It could be someone who would otherwise have filed a lawsuit, who now thinks he has a more effective option for exacting retribution.
Most of us would find it unbearable to live and work under the kind of stress faced by medical professionals today. And unfortunately, there is no short-term, systemic relief on the horizon. But there are methods of relieving at least some of the psychological burden being carried by these dedicated individuals.
For starters, the government should provide funds to improve safety and security at medical facilities. It’s sad but it’s a fact of life. The physical structure of schools, along with emergency procedures, have been changed since Columbine and Sandy Hook, and our children and their teachers undergo active shooter drills. Health care facilities will need to adopt similar strategies.
But if we don’t also support the individuals who work in health care, we’ll no longer have even partially staffed health care facilities. Hospitals and medical groups need to be conscious of the effects stress may have on them. Medical staff and administrators need to recognize changes in their colleagues’ behavior and refer those cohorts to professional stress coaches who can get them back on track.
Medical personnel should be picking up on warning signs, like irritability, depression, sudden weight gain or loss, lack of motivation and job satisfaction, obsessiveness, unusual levels of fatigue, alcohol or drug use, and, of course, avoidable medical errors.
In addition, colleagues in the medical workplace need to know each other well. They are usually the first ones to notice if something is off and may be in the best position to refer coworkers for help. Also, medical malpractice insurance carriers should consider encouraging and covering coaching sessions, because helping physicians cope with this heightened stress will prevent medical errors and the lawsuits that inevitably accompany mistakes.
This needn’t be a long-term process like ongoing psychotherapy; a few sessions with a well-trained coach may help psychologically challenged peers restore their focus and perspective. It won’t eliminate the threat any more than litigation stress coaching eliminates the threat of being sued, but it can prevent that stress from leading to avoidable errors. It also can prevent physicians’ personal lives and relationships from going off the rails and driving them out of the medical profession.
None of us can afford to ignore the impacts that these new stressors are having and simply act as if it’s business as usual. The people in the trenches need our help.
Ms. Fiore is President of Winning Focus in Murrysville, Pa. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
As if it weren’t enough that doctors work in a profession where it’s almost more a question of when they’ll be sued than if they’ll be sued – where COVID, staff shortages, long hours, and patients frustrated over canceled procedures have caused unrelenting fatigue and stress – they now have to worry that an unhappy patient is going to buy a gun, walk into their office, and kill them.
That’s exactly what happened in Tulsa, Okla., where a patient complaining of pain after back surgery murdered his doctor and several others who happened to be in the wrong place at the wrong time.
The temptation in the aftermath of such tragedies is to think about preventive measures: Make medical facilities “hardened” targets, like schools have become, with armed guards, metal detectors, automatically locking doors, physical barriers within, security cameras, and buzzers for entry – although hardening a large medical center where members of the community routinely come and go would be challenging.
What about the enormous stress on doctors, nurses, and others in the medical workplace? Physicians who have been sued for malpractice often describe how it changes the way they interact with patients: They now size patients up and make judgments about their potential litigiousness. Will the physicians now look over their patients’ shoulders at the video feed from a security camera when they’re taking a history? Will medical professionals be forced to make snap judgments about patients’ psychological state before deciding whether to treat them?
Remember, there was a time when school shootings were unimaginable. Once one person crosses that line, others inevitably follow.
It could be a drug-seeking patient complaining of ongoing pain, angry because he can’t get a new prescription. It could be a patient whose unpaid bill was turned over to a collection agency, angry because he’s now getting calls from collectors. It could be someone who blames a physician for the loss of a loved one. It could be someone who would otherwise have filed a lawsuit, who now thinks he has a more effective option for exacting retribution.
Most of us would find it unbearable to live and work under the kind of stress faced by medical professionals today. And unfortunately, there is no short-term, systemic relief on the horizon. But there are methods of relieving at least some of the psychological burden being carried by these dedicated individuals.
For starters, the government should provide funds to improve safety and security at medical facilities. It’s sad but it’s a fact of life. The physical structure of schools, along with emergency procedures, have been changed since Columbine and Sandy Hook, and our children and their teachers undergo active shooter drills. Health care facilities will need to adopt similar strategies.
But if we don’t also support the individuals who work in health care, we’ll no longer have even partially staffed health care facilities. Hospitals and medical groups need to be conscious of the effects stress may have on them. Medical staff and administrators need to recognize changes in their colleagues’ behavior and refer those cohorts to professional stress coaches who can get them back on track.
Medical personnel should be picking up on warning signs, like irritability, depression, sudden weight gain or loss, lack of motivation and job satisfaction, obsessiveness, unusual levels of fatigue, alcohol or drug use, and, of course, avoidable medical errors.
In addition, colleagues in the medical workplace need to know each other well. They are usually the first ones to notice if something is off and may be in the best position to refer coworkers for help. Also, medical malpractice insurance carriers should consider encouraging and covering coaching sessions, because helping physicians cope with this heightened stress will prevent medical errors and the lawsuits that inevitably accompany mistakes.
This needn’t be a long-term process like ongoing psychotherapy; a few sessions with a well-trained coach may help psychologically challenged peers restore their focus and perspective. It won’t eliminate the threat any more than litigation stress coaching eliminates the threat of being sued, but it can prevent that stress from leading to avoidable errors. It also can prevent physicians’ personal lives and relationships from going off the rails and driving them out of the medical profession.
None of us can afford to ignore the impacts that these new stressors are having and simply act as if it’s business as usual. The people in the trenches need our help.
Ms. Fiore is President of Winning Focus in Murrysville, Pa. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the Grand Canyon, norovirus gives new meaning to ‘leave no trace’
Ain’t gastroenteritis grand?
The Grand Canyon is perhaps America’s greatest natural wonder. The mile-deep gorge of epic proportions, carved over eons by the Colorado River, elicits superlatives of the highest order from those seeing it for the first time. In the past few months, though, visitors to the Grand Canyon have been experiencing a rather more unpleasant sort of reaction: Involuntary bowel evacuation.
Since April, more than 150 river rafters and backcountry campers have fallen ill with bouts of acute gastroenteritis, likely caused by norovirus. Hey, a viral outbreak and our old friend SARS-CoV-2 isn’t involved! Hopefully it won’t get jealous. Whatever the culprit is, however, it got everywhere, as clusters of illness have popped up in unconnected parts of the park and some hikers have been restricted to a smaller portion of the park to avoid further disease spread. The majority of cases occurred in May, so it’s hoped that the outbreak is dying down, but the park remains on alert.
Now, acute gastroenteritis is certainly an unpleasant disease, but it isn’t typically a life-threatening one. There are, however, a couple of unique factors complicating this outbreak. For one, the Grand Canyon is in Arizona (duh), which can get rather hot in the summer months. Expelling waste from both ends becomes rather more dangerous when the thermometer reads over a hundred degrees, and there have been reports of multiple helicopter rescues.
That’s pretty bad, but in a way, they’re the lucky ones. How can we explain this … see, when you visit the Grand Canyon, you’re expected to follow the general rules of Leave No Trace. That means several things, but essentially, if you bring it in, you have to bring it out. Yes, that includes the various consequences of an acute gastroenteritis attack.
Forget spooky campfire stories and hungry wildlife lurking in the night, because true horror is scraping your friend’s diarrhea off the walls of the Grand Canyon into a plastic bag and stuffing it into your backpack. Probably not the sublime one-on-one Grand Canyon experience that people are expecting.
Give us a pee! ... for stem cell retrieval
Getting cells for regenerative stem cell treatment has traditionally been painful and difficult – usually they are retrieved by surgical means from bone marrow or fat tissue – but there may be an easier way.
Just pee in a cup.
Apparently, human urine contains stem cells with the potential to be used for regenerative effects. The magic ingredient? The enzyme telomerase, which “is essential for the self-renewal and potential of different types of stem cells” and is related to longevity, according to researchers at Wake Forest Institute for Regenerative Medicine.
They looked into how regenerative telomerase activity is for various capabilities beyond chromosomal stability, and whether these stem cells can become other kinds of cells for optimal tissue repair. Turns out they could, acting as a “distinct subpopulation” that has the ability not only to grow cells but also to morph into other cells, they said in a written statement.
Safety is also an issue. “Being able to use a patient’s own stem cells for therapy is considered advantageous because they do not induce immune responses or rejection,” said Anthony Atala, MD, a coauthor of the study published in Frontiers in Cell and Developmental Biology.
So less risk, easier retrieval, and great regenerative results. If this takes off, the other methods of retrieval could get flushed down the toilet.
Politicians playing the long game, literally
Before we get started with actual information, here’s a joke about politicians:
What do you call a lawyer with an IQ of 100? Your Honor.
What do you call a lawyer with an IQ of 50? Senator.
Politics is a dirty business, no doubt, so why do people do it? Is it for the prestige? Seems like everyone hates politicians, so it’s probably not that. Is it their selfless concern for the well-being of others? Probably not that either. Is it for the money? Most members of Congress have more corporate sponsors than a NASCAR driver, but we’re going to pass on that one as well.
Once again, science gives us the real answer: Longevity. Politicians live longer than the rest of us, and that longevity gap is getting wider.
Investigators looked at data from 11 industrialized countries, some of it going back to 1817, and found that politicians in the United States can expect to live about 7 years longer than the national average. The difference is around 3 years in Switzerland, 4.5 years in Germany, and 6 years in France.
“For almost all countries, politicians had similar rates of mortality to the general population in the late 19th and early 20th centuries. Throughout the 20th century, differences in mortality rates widened significantly across all countries, so that politicians had an increasing survival advantage over the general population,” they said in a written statement.
Income inequality could be a factor, but the longevity gains made by politicians, which started before the 1940s, predate the rise of their earnings relative to the rest of the population, which didn’t really get going until the 1980s, the investigators noted.
Whatever the reason, we have this closing thought regarding our long-lived lawmakers: What’s the difference between a politician and a snail? One is a slimy pest that leaves a trail everywhere. The other is a snail.
Land of the free, home of obesity
In the United States, it seems, people are becoming more comfortable with obesity. TikTok and Instagram trends often try to show the world that all sizes are beautiful. There’s also the growing popularity of the dad bod.
America, it has been said, is the land of the free. We love our freedom, and we value our individualism. If an obese man orders three meals from McDonald’s just for himself, no one is going to stop him. Many Americans also have more access to the food they want at any given time, even while they are moving around a lot less because of their sedentary lifestyles.
According to a recent study cited by the New York Post, however, America is not the only country battling obesity. Egypt and Mexico, for example, also have men with higher BMIs who cherish their individualism and the right to eat what they want, Plamen Akaliyski, PhD, of University Carlos III of Madrid, and associates, said in Social Science & Medicine.
Women are not as likely to think the same way. “Men in particular think, ‘I’m an individual, don’t tell me what to do. I’m going to eat what I want,’ ” bariatric surgeon George A. Fielding, MD, said in the Post article. Dr. Fielding also noted that women are three times more likely than men to seek bariatric surgery.
Dr. Akaliyski and associates found that Asian countries such as Japan, Singapore, and South Korea – countries that value thrift, discipline, self control, and delaying gratification – have lower rates of obesity.
So yes, we can go to the drive through of a fast food restaurant whenever we want and order whatever we want, but can doesn’t always mean should.
Ain’t gastroenteritis grand?
The Grand Canyon is perhaps America’s greatest natural wonder. The mile-deep gorge of epic proportions, carved over eons by the Colorado River, elicits superlatives of the highest order from those seeing it for the first time. In the past few months, though, visitors to the Grand Canyon have been experiencing a rather more unpleasant sort of reaction: Involuntary bowel evacuation.
Since April, more than 150 river rafters and backcountry campers have fallen ill with bouts of acute gastroenteritis, likely caused by norovirus. Hey, a viral outbreak and our old friend SARS-CoV-2 isn’t involved! Hopefully it won’t get jealous. Whatever the culprit is, however, it got everywhere, as clusters of illness have popped up in unconnected parts of the park and some hikers have been restricted to a smaller portion of the park to avoid further disease spread. The majority of cases occurred in May, so it’s hoped that the outbreak is dying down, but the park remains on alert.
Now, acute gastroenteritis is certainly an unpleasant disease, but it isn’t typically a life-threatening one. There are, however, a couple of unique factors complicating this outbreak. For one, the Grand Canyon is in Arizona (duh), which can get rather hot in the summer months. Expelling waste from both ends becomes rather more dangerous when the thermometer reads over a hundred degrees, and there have been reports of multiple helicopter rescues.
That’s pretty bad, but in a way, they’re the lucky ones. How can we explain this … see, when you visit the Grand Canyon, you’re expected to follow the general rules of Leave No Trace. That means several things, but essentially, if you bring it in, you have to bring it out. Yes, that includes the various consequences of an acute gastroenteritis attack.
Forget spooky campfire stories and hungry wildlife lurking in the night, because true horror is scraping your friend’s diarrhea off the walls of the Grand Canyon into a plastic bag and stuffing it into your backpack. Probably not the sublime one-on-one Grand Canyon experience that people are expecting.
Give us a pee! ... for stem cell retrieval
Getting cells for regenerative stem cell treatment has traditionally been painful and difficult – usually they are retrieved by surgical means from bone marrow or fat tissue – but there may be an easier way.
Just pee in a cup.
Apparently, human urine contains stem cells with the potential to be used for regenerative effects. The magic ingredient? The enzyme telomerase, which “is essential for the self-renewal and potential of different types of stem cells” and is related to longevity, according to researchers at Wake Forest Institute for Regenerative Medicine.
They looked into how regenerative telomerase activity is for various capabilities beyond chromosomal stability, and whether these stem cells can become other kinds of cells for optimal tissue repair. Turns out they could, acting as a “distinct subpopulation” that has the ability not only to grow cells but also to morph into other cells, they said in a written statement.
Safety is also an issue. “Being able to use a patient’s own stem cells for therapy is considered advantageous because they do not induce immune responses or rejection,” said Anthony Atala, MD, a coauthor of the study published in Frontiers in Cell and Developmental Biology.
So less risk, easier retrieval, and great regenerative results. If this takes off, the other methods of retrieval could get flushed down the toilet.
Politicians playing the long game, literally
Before we get started with actual information, here’s a joke about politicians:
What do you call a lawyer with an IQ of 100? Your Honor.
What do you call a lawyer with an IQ of 50? Senator.
Politics is a dirty business, no doubt, so why do people do it? Is it for the prestige? Seems like everyone hates politicians, so it’s probably not that. Is it their selfless concern for the well-being of others? Probably not that either. Is it for the money? Most members of Congress have more corporate sponsors than a NASCAR driver, but we’re going to pass on that one as well.
Once again, science gives us the real answer: Longevity. Politicians live longer than the rest of us, and that longevity gap is getting wider.
Investigators looked at data from 11 industrialized countries, some of it going back to 1817, and found that politicians in the United States can expect to live about 7 years longer than the national average. The difference is around 3 years in Switzerland, 4.5 years in Germany, and 6 years in France.
“For almost all countries, politicians had similar rates of mortality to the general population in the late 19th and early 20th centuries. Throughout the 20th century, differences in mortality rates widened significantly across all countries, so that politicians had an increasing survival advantage over the general population,” they said in a written statement.
Income inequality could be a factor, but the longevity gains made by politicians, which started before the 1940s, predate the rise of their earnings relative to the rest of the population, which didn’t really get going until the 1980s, the investigators noted.
Whatever the reason, we have this closing thought regarding our long-lived lawmakers: What’s the difference between a politician and a snail? One is a slimy pest that leaves a trail everywhere. The other is a snail.
Land of the free, home of obesity
In the United States, it seems, people are becoming more comfortable with obesity. TikTok and Instagram trends often try to show the world that all sizes are beautiful. There’s also the growing popularity of the dad bod.
America, it has been said, is the land of the free. We love our freedom, and we value our individualism. If an obese man orders three meals from McDonald’s just for himself, no one is going to stop him. Many Americans also have more access to the food they want at any given time, even while they are moving around a lot less because of their sedentary lifestyles.
According to a recent study cited by the New York Post, however, America is not the only country battling obesity. Egypt and Mexico, for example, also have men with higher BMIs who cherish their individualism and the right to eat what they want, Plamen Akaliyski, PhD, of University Carlos III of Madrid, and associates, said in Social Science & Medicine.
Women are not as likely to think the same way. “Men in particular think, ‘I’m an individual, don’t tell me what to do. I’m going to eat what I want,’ ” bariatric surgeon George A. Fielding, MD, said in the Post article. Dr. Fielding also noted that women are three times more likely than men to seek bariatric surgery.
Dr. Akaliyski and associates found that Asian countries such as Japan, Singapore, and South Korea – countries that value thrift, discipline, self control, and delaying gratification – have lower rates of obesity.
So yes, we can go to the drive through of a fast food restaurant whenever we want and order whatever we want, but can doesn’t always mean should.
Ain’t gastroenteritis grand?
The Grand Canyon is perhaps America’s greatest natural wonder. The mile-deep gorge of epic proportions, carved over eons by the Colorado River, elicits superlatives of the highest order from those seeing it for the first time. In the past few months, though, visitors to the Grand Canyon have been experiencing a rather more unpleasant sort of reaction: Involuntary bowel evacuation.
Since April, more than 150 river rafters and backcountry campers have fallen ill with bouts of acute gastroenteritis, likely caused by norovirus. Hey, a viral outbreak and our old friend SARS-CoV-2 isn’t involved! Hopefully it won’t get jealous. Whatever the culprit is, however, it got everywhere, as clusters of illness have popped up in unconnected parts of the park and some hikers have been restricted to a smaller portion of the park to avoid further disease spread. The majority of cases occurred in May, so it’s hoped that the outbreak is dying down, but the park remains on alert.
Now, acute gastroenteritis is certainly an unpleasant disease, but it isn’t typically a life-threatening one. There are, however, a couple of unique factors complicating this outbreak. For one, the Grand Canyon is in Arizona (duh), which can get rather hot in the summer months. Expelling waste from both ends becomes rather more dangerous when the thermometer reads over a hundred degrees, and there have been reports of multiple helicopter rescues.
That’s pretty bad, but in a way, they’re the lucky ones. How can we explain this … see, when you visit the Grand Canyon, you’re expected to follow the general rules of Leave No Trace. That means several things, but essentially, if you bring it in, you have to bring it out. Yes, that includes the various consequences of an acute gastroenteritis attack.
Forget spooky campfire stories and hungry wildlife lurking in the night, because true horror is scraping your friend’s diarrhea off the walls of the Grand Canyon into a plastic bag and stuffing it into your backpack. Probably not the sublime one-on-one Grand Canyon experience that people are expecting.
Give us a pee! ... for stem cell retrieval
Getting cells for regenerative stem cell treatment has traditionally been painful and difficult – usually they are retrieved by surgical means from bone marrow or fat tissue – but there may be an easier way.
Just pee in a cup.
Apparently, human urine contains stem cells with the potential to be used for regenerative effects. The magic ingredient? The enzyme telomerase, which “is essential for the self-renewal and potential of different types of stem cells” and is related to longevity, according to researchers at Wake Forest Institute for Regenerative Medicine.
They looked into how regenerative telomerase activity is for various capabilities beyond chromosomal stability, and whether these stem cells can become other kinds of cells for optimal tissue repair. Turns out they could, acting as a “distinct subpopulation” that has the ability not only to grow cells but also to morph into other cells, they said in a written statement.
Safety is also an issue. “Being able to use a patient’s own stem cells for therapy is considered advantageous because they do not induce immune responses or rejection,” said Anthony Atala, MD, a coauthor of the study published in Frontiers in Cell and Developmental Biology.
So less risk, easier retrieval, and great regenerative results. If this takes off, the other methods of retrieval could get flushed down the toilet.
Politicians playing the long game, literally
Before we get started with actual information, here’s a joke about politicians:
What do you call a lawyer with an IQ of 100? Your Honor.
What do you call a lawyer with an IQ of 50? Senator.
Politics is a dirty business, no doubt, so why do people do it? Is it for the prestige? Seems like everyone hates politicians, so it’s probably not that. Is it their selfless concern for the well-being of others? Probably not that either. Is it for the money? Most members of Congress have more corporate sponsors than a NASCAR driver, but we’re going to pass on that one as well.
Once again, science gives us the real answer: Longevity. Politicians live longer than the rest of us, and that longevity gap is getting wider.
Investigators looked at data from 11 industrialized countries, some of it going back to 1817, and found that politicians in the United States can expect to live about 7 years longer than the national average. The difference is around 3 years in Switzerland, 4.5 years in Germany, and 6 years in France.
“For almost all countries, politicians had similar rates of mortality to the general population in the late 19th and early 20th centuries. Throughout the 20th century, differences in mortality rates widened significantly across all countries, so that politicians had an increasing survival advantage over the general population,” they said in a written statement.
Income inequality could be a factor, but the longevity gains made by politicians, which started before the 1940s, predate the rise of their earnings relative to the rest of the population, which didn’t really get going until the 1980s, the investigators noted.
Whatever the reason, we have this closing thought regarding our long-lived lawmakers: What’s the difference between a politician and a snail? One is a slimy pest that leaves a trail everywhere. The other is a snail.
Land of the free, home of obesity
In the United States, it seems, people are becoming more comfortable with obesity. TikTok and Instagram trends often try to show the world that all sizes are beautiful. There’s also the growing popularity of the dad bod.
America, it has been said, is the land of the free. We love our freedom, and we value our individualism. If an obese man orders three meals from McDonald’s just for himself, no one is going to stop him. Many Americans also have more access to the food they want at any given time, even while they are moving around a lot less because of their sedentary lifestyles.
According to a recent study cited by the New York Post, however, America is not the only country battling obesity. Egypt and Mexico, for example, also have men with higher BMIs who cherish their individualism and the right to eat what they want, Plamen Akaliyski, PhD, of University Carlos III of Madrid, and associates, said in Social Science & Medicine.
Women are not as likely to think the same way. “Men in particular think, ‘I’m an individual, don’t tell me what to do. I’m going to eat what I want,’ ” bariatric surgeon George A. Fielding, MD, said in the Post article. Dr. Fielding also noted that women are three times more likely than men to seek bariatric surgery.
Dr. Akaliyski and associates found that Asian countries such as Japan, Singapore, and South Korea – countries that value thrift, discipline, self control, and delaying gratification – have lower rates of obesity.
So yes, we can go to the drive through of a fast food restaurant whenever we want and order whatever we want, but can doesn’t always mean should.
‘Superior’ CLL regimen cuts chemo in half
“Overall, our data suggests that [the chemoimmunotherapy] regimen is very effective and appears superior to published six cycles of chemotherapy regimen for the same favorable risk features,” first author Dr. Nitin Jain, an associate professor in the department of leukemia at the University of Texas MD Anderson Cancer Center, Houston, told MDedge.
Chemoimmunotherapy with fludarabine, cyclophosphamide and rituximab (FCR) has been a standard frontline treatment for young, fit patients with CLL, resulting in 10-year PFS rates above 55% in patients with mutated IGHV status, said coauthor Dr. Alessandra Ferrajoli, also of the MD Anderson Cancer Center, in presenting the findings at the European Hematology Association annual congress.
The authors sought to investigate the efficacy of a targeted therapy combination of ibrutinib and obinutuzumab with fludarabine and cyclophosphamide (iFCG). They also sought to determine whether a three-cycle regimen of the chemotherapy, as compared to six cycles, could reduce the risk of myelodysplastic syndrome (MDS), which increases with chemotherapy in CLL patients who have mutated IGHV status.
For the phase 2 study, 45 previously untreated patients with CLL, who had mutated IGHV and an absence of del(17p)/TP53 mutation (both of which are associated with more favorable outcomes in CLL) were enrolled between March 2016 and August 2018. The patients were deemed fit for chemotherapy and had a median age of 60.
All patients were initially treated with three cycles of the iFCG regimen, and among them, 39 (87%) achieved undetectable measurable residual disease (MRD) in their bone marrow.
After the three cycles, an MRD-driven strategy was then used to determine subsequent treatment: All patients received nine courses of ibrutinib, and for those achieving complete remission (CR) or CR with incomplete count recovery (CRi) and undetectable MRD, three cycles of obinutuzumab were administered, while all others received nine additional cycles of obinutuzumab.
At completion of the 12 courses, those who still had MRD positivity continued on ibrutinib, while those with undetectable MRD discontinued ibrutinib.
By cycle six of iFCG, 40 (89%) of the patients achieved undetectable MRD. Overall, 44 of the 45 patients (98%) achieved undetectable MRD as their best response at any time during the study, with 69% of patients achieving CR/CRi. Four patients came off the study prior to cycle 12, including one death, one infection, and one patient who opted to pursue treatment locally. With a median follow-up of 59.6 months, there were no cases of CLL progression or Richter transformation and the lone death was from heart failure.
One patient developed treatment-related myelodysplastic syndrome (MDS), and that patient has maintained normal blood counts over 38 months of monitoring and has not required MDS therapy, Dr. Ferrajoli reported.
Over the follow-up, the six patients who were MRD positive after the completion of three cycles experienced a recurrence of MRD, defined as two consecutive values of 0.01% or higher in peripheral blood by flow cytometry, at a median of 27.2 months after stopping all therapy.
“Not unexpectedly, MRD recurrence during follow-up correlated with MRD positivity during therapy,” Dr. Ferrajoli said.
She noted that all six of the patients were being monitored, with no clinical progression or active therapy. However, with a median follow-up of 5 years, the progression-free survival (PFS) rate among the 45 patients was 97.7%, and the overall survival (OS) rate was 97.8%. Dr. Ferrajoli noted that, while the study population was clearly different, the results compare favorably with CLL clinical trial results that have previously shown a 5-year PFS of approximately 65% with FCR alone; approximately 70% with ibrutinib; and 81% with ibrutinib among patients with mutated IGHV status.
Furthermore, the rate of undetectable MRD status in mutated IGHV patients being 95% in evaluable patients in the current study is notably higher than rates of 51% through 67% reported in five other trials of CLL treatment with six cycles of FCR and with a rate of 79% in the DFCI trial of six-cycle chemotherapy plus ibrutinib.
And the current study’s undetectable MRD rate of 89% in the intention-to-treat population compares with just 13% though 40% in the five other chemotherapy trials and 79% in the DFCI trial, the authors note.
The current trial was the only one of any of their comparisons to utilize the three-cycle regimen.
Asked at the meeting about concerns of toxicities reported with obinutuzumab and chemotherapy, Dr. Ferrajoli said “the treatment was very well tolerated.”
“Myelosuppression is a concern with this combination, but we did make the use of prophylactic growth-factor mandatory in the study, so we were able to control that,” she said.
Dr. Jain noted that, while treatment trends have moved largely to chemo-free regimens, particularly in the United States because of concerns about the MDS, the current study’s results importantly shed light on a potentially beneficial approach of just three cycles of chemotherapy.
“In Europe and the rest of the world where chemo use is still common, this regimen could be considered,” he told MDedge. “The findings show that if you still use chemo in your practice, this regimen uses 50% less chemotherapy, yet seems to give higher response rates.”
“While MDS and acute myeloid leukemia (AML) remain a concern with any chemotherapy regimen, it is possible that 50% less chemo will lead to less risk of MDS AML, but longer-term follow-up [is needed],” he said.
Dr. Ferrajoli reported that she has received research support from Astra-Zeneca and Beigene. Dr. Jain has received research funding and honoraria from Genentech and Pharmacyclics.
“Overall, our data suggests that [the chemoimmunotherapy] regimen is very effective and appears superior to published six cycles of chemotherapy regimen for the same favorable risk features,” first author Dr. Nitin Jain, an associate professor in the department of leukemia at the University of Texas MD Anderson Cancer Center, Houston, told MDedge.
Chemoimmunotherapy with fludarabine, cyclophosphamide and rituximab (FCR) has been a standard frontline treatment for young, fit patients with CLL, resulting in 10-year PFS rates above 55% in patients with mutated IGHV status, said coauthor Dr. Alessandra Ferrajoli, also of the MD Anderson Cancer Center, in presenting the findings at the European Hematology Association annual congress.
The authors sought to investigate the efficacy of a targeted therapy combination of ibrutinib and obinutuzumab with fludarabine and cyclophosphamide (iFCG). They also sought to determine whether a three-cycle regimen of the chemotherapy, as compared to six cycles, could reduce the risk of myelodysplastic syndrome (MDS), which increases with chemotherapy in CLL patients who have mutated IGHV status.
For the phase 2 study, 45 previously untreated patients with CLL, who had mutated IGHV and an absence of del(17p)/TP53 mutation (both of which are associated with more favorable outcomes in CLL) were enrolled between March 2016 and August 2018. The patients were deemed fit for chemotherapy and had a median age of 60.
All patients were initially treated with three cycles of the iFCG regimen, and among them, 39 (87%) achieved undetectable measurable residual disease (MRD) in their bone marrow.
After the three cycles, an MRD-driven strategy was then used to determine subsequent treatment: All patients received nine courses of ibrutinib, and for those achieving complete remission (CR) or CR with incomplete count recovery (CRi) and undetectable MRD, three cycles of obinutuzumab were administered, while all others received nine additional cycles of obinutuzumab.
At completion of the 12 courses, those who still had MRD positivity continued on ibrutinib, while those with undetectable MRD discontinued ibrutinib.
By cycle six of iFCG, 40 (89%) of the patients achieved undetectable MRD. Overall, 44 of the 45 patients (98%) achieved undetectable MRD as their best response at any time during the study, with 69% of patients achieving CR/CRi. Four patients came off the study prior to cycle 12, including one death, one infection, and one patient who opted to pursue treatment locally. With a median follow-up of 59.6 months, there were no cases of CLL progression or Richter transformation and the lone death was from heart failure.
One patient developed treatment-related myelodysplastic syndrome (MDS), and that patient has maintained normal blood counts over 38 months of monitoring and has not required MDS therapy, Dr. Ferrajoli reported.
Over the follow-up, the six patients who were MRD positive after the completion of three cycles experienced a recurrence of MRD, defined as two consecutive values of 0.01% or higher in peripheral blood by flow cytometry, at a median of 27.2 months after stopping all therapy.
“Not unexpectedly, MRD recurrence during follow-up correlated with MRD positivity during therapy,” Dr. Ferrajoli said.
She noted that all six of the patients were being monitored, with no clinical progression or active therapy. However, with a median follow-up of 5 years, the progression-free survival (PFS) rate among the 45 patients was 97.7%, and the overall survival (OS) rate was 97.8%. Dr. Ferrajoli noted that, while the study population was clearly different, the results compare favorably with CLL clinical trial results that have previously shown a 5-year PFS of approximately 65% with FCR alone; approximately 70% with ibrutinib; and 81% with ibrutinib among patients with mutated IGHV status.
Furthermore, the rate of undetectable MRD status in mutated IGHV patients being 95% in evaluable patients in the current study is notably higher than rates of 51% through 67% reported in five other trials of CLL treatment with six cycles of FCR and with a rate of 79% in the DFCI trial of six-cycle chemotherapy plus ibrutinib.
And the current study’s undetectable MRD rate of 89% in the intention-to-treat population compares with just 13% though 40% in the five other chemotherapy trials and 79% in the DFCI trial, the authors note.
The current trial was the only one of any of their comparisons to utilize the three-cycle regimen.
Asked at the meeting about concerns of toxicities reported with obinutuzumab and chemotherapy, Dr. Ferrajoli said “the treatment was very well tolerated.”
“Myelosuppression is a concern with this combination, but we did make the use of prophylactic growth-factor mandatory in the study, so we were able to control that,” she said.
Dr. Jain noted that, while treatment trends have moved largely to chemo-free regimens, particularly in the United States because of concerns about the MDS, the current study’s results importantly shed light on a potentially beneficial approach of just three cycles of chemotherapy.
“In Europe and the rest of the world where chemo use is still common, this regimen could be considered,” he told MDedge. “The findings show that if you still use chemo in your practice, this regimen uses 50% less chemotherapy, yet seems to give higher response rates.”
“While MDS and acute myeloid leukemia (AML) remain a concern with any chemotherapy regimen, it is possible that 50% less chemo will lead to less risk of MDS AML, but longer-term follow-up [is needed],” he said.
Dr. Ferrajoli reported that she has received research support from Astra-Zeneca and Beigene. Dr. Jain has received research funding and honoraria from Genentech and Pharmacyclics.
“Overall, our data suggests that [the chemoimmunotherapy] regimen is very effective and appears superior to published six cycles of chemotherapy regimen for the same favorable risk features,” first author Dr. Nitin Jain, an associate professor in the department of leukemia at the University of Texas MD Anderson Cancer Center, Houston, told MDedge.
Chemoimmunotherapy with fludarabine, cyclophosphamide and rituximab (FCR) has been a standard frontline treatment for young, fit patients with CLL, resulting in 10-year PFS rates above 55% in patients with mutated IGHV status, said coauthor Dr. Alessandra Ferrajoli, also of the MD Anderson Cancer Center, in presenting the findings at the European Hematology Association annual congress.
The authors sought to investigate the efficacy of a targeted therapy combination of ibrutinib and obinutuzumab with fludarabine and cyclophosphamide (iFCG). They also sought to determine whether a three-cycle regimen of the chemotherapy, as compared to six cycles, could reduce the risk of myelodysplastic syndrome (MDS), which increases with chemotherapy in CLL patients who have mutated IGHV status.
For the phase 2 study, 45 previously untreated patients with CLL, who had mutated IGHV and an absence of del(17p)/TP53 mutation (both of which are associated with more favorable outcomes in CLL) were enrolled between March 2016 and August 2018. The patients were deemed fit for chemotherapy and had a median age of 60.
All patients were initially treated with three cycles of the iFCG regimen, and among them, 39 (87%) achieved undetectable measurable residual disease (MRD) in their bone marrow.
After the three cycles, an MRD-driven strategy was then used to determine subsequent treatment: All patients received nine courses of ibrutinib, and for those achieving complete remission (CR) or CR with incomplete count recovery (CRi) and undetectable MRD, three cycles of obinutuzumab were administered, while all others received nine additional cycles of obinutuzumab.
At completion of the 12 courses, those who still had MRD positivity continued on ibrutinib, while those with undetectable MRD discontinued ibrutinib.
By cycle six of iFCG, 40 (89%) of the patients achieved undetectable MRD. Overall, 44 of the 45 patients (98%) achieved undetectable MRD as their best response at any time during the study, with 69% of patients achieving CR/CRi. Four patients came off the study prior to cycle 12, including one death, one infection, and one patient who opted to pursue treatment locally. With a median follow-up of 59.6 months, there were no cases of CLL progression or Richter transformation and the lone death was from heart failure.
One patient developed treatment-related myelodysplastic syndrome (MDS), and that patient has maintained normal blood counts over 38 months of monitoring and has not required MDS therapy, Dr. Ferrajoli reported.
Over the follow-up, the six patients who were MRD positive after the completion of three cycles experienced a recurrence of MRD, defined as two consecutive values of 0.01% or higher in peripheral blood by flow cytometry, at a median of 27.2 months after stopping all therapy.
“Not unexpectedly, MRD recurrence during follow-up correlated with MRD positivity during therapy,” Dr. Ferrajoli said.
She noted that all six of the patients were being monitored, with no clinical progression or active therapy. However, with a median follow-up of 5 years, the progression-free survival (PFS) rate among the 45 patients was 97.7%, and the overall survival (OS) rate was 97.8%. Dr. Ferrajoli noted that, while the study population was clearly different, the results compare favorably with CLL clinical trial results that have previously shown a 5-year PFS of approximately 65% with FCR alone; approximately 70% with ibrutinib; and 81% with ibrutinib among patients with mutated IGHV status.
Furthermore, the rate of undetectable MRD status in mutated IGHV patients being 95% in evaluable patients in the current study is notably higher than rates of 51% through 67% reported in five other trials of CLL treatment with six cycles of FCR and with a rate of 79% in the DFCI trial of six-cycle chemotherapy plus ibrutinib.
And the current study’s undetectable MRD rate of 89% in the intention-to-treat population compares with just 13% though 40% in the five other chemotherapy trials and 79% in the DFCI trial, the authors note.
The current trial was the only one of any of their comparisons to utilize the three-cycle regimen.
Asked at the meeting about concerns of toxicities reported with obinutuzumab and chemotherapy, Dr. Ferrajoli said “the treatment was very well tolerated.”
“Myelosuppression is a concern with this combination, but we did make the use of prophylactic growth-factor mandatory in the study, so we were able to control that,” she said.
Dr. Jain noted that, while treatment trends have moved largely to chemo-free regimens, particularly in the United States because of concerns about the MDS, the current study’s results importantly shed light on a potentially beneficial approach of just three cycles of chemotherapy.
“In Europe and the rest of the world where chemo use is still common, this regimen could be considered,” he told MDedge. “The findings show that if you still use chemo in your practice, this regimen uses 50% less chemotherapy, yet seems to give higher response rates.”
“While MDS and acute myeloid leukemia (AML) remain a concern with any chemotherapy regimen, it is possible that 50% less chemo will lead to less risk of MDS AML, but longer-term follow-up [is needed],” he said.
Dr. Ferrajoli reported that she has received research support from Astra-Zeneca and Beigene. Dr. Jain has received research funding and honoraria from Genentech and Pharmacyclics.
FROM EHA 2022
FDA approves liso-cel as second-line therapy for LBCL
This expanded indication is based on findings from the pivotal phase 3 TRANSFORM study, which showed significant and clinically meaningful improvements with CD19-directed chimeric antigen receptor T-cell immunotherapy over salvage chemotherapy followed by high-dose chemotherapy plus autologous stem cell transplant. The latter course of treatment had been the standard of care for more than 2 decades.
Data from the global, randomized, multicenter TRANSFORM study, as reported in December 2021 at the annual meeting of the American Society of Hematology, showed that second-line treatment with liso-cel in 92 patients with r/r LBCL within 12 months after first-line therapy, compared with 92 patient who received standard of care therapy, was associated with highly statistically significant and clinically meaningful improvement in event-free survival (10.1 vs. 2.3 months; hazard ratio, 0.349), complete response rate (66% vs. 39%), and progression-free survival (14.8 vs. 5.7 months; HR, 0.406).
A positive trend in overall survival was also observed (HR, 0.509 at median follow-up of 6.2 months). No new liso-cel safety signals were detected in the second-line setting.
Liso-cel was initially approved in February 2021 for the treatment of adults with LBCL, including diffuse LBCL not otherwise specified (including DLBCL arising from indolent lymphoma), high-grade B-cell lymphoma, primary mediastinal large B-cell lymphoma, and follicular lymphoma grade 3B, who have:
- Refractory disease to first-line chemoimmunotherapy or relapse within 12 months of first-line chemoimmunotherapy.
- Refractory disease to first-line chemoimmunotherapy or relapse after first-line chemoimmunotherapy and are not eligible for hematopoietic stem cell transplant because of comorbidities or age.
Liso-cel is not indicated for the treatment of patients with primary central nervous system lymphoma.
In February 2022, the FDA granted Priority Review status for a Bristol-Myers Squibb supplemental Biologics License Application (sBLA), based on the TRANSFORM study data, to expand the indication to include use after the failure of first-line therapy.
The agent “now has the potential to be a new standard of care for patients after failure of first-line therapy, offering significantly improved outcomes beyond the current mainstay of care,” Anne Kerber, the BMS senior vice president of cell therapy development, said in a press release at that time.
The European Medicines Agency has also validated a type II variation application for extension of the indication for liso-cel in this setting. Validation of the application “confirms the submission is complete and begins the EMA’s centralized review procedure,” BMS announced in a June 20, 2022, press release.
Liso-cel, which has been available only through a restricted program under a Risk Evaluation and Mitigation Strategy, includes a boxed warning regarding the risk for cytokine release syndrome (CRS) and neurologic toxicities.
The warning states that liso-cel should not be administered to patients with active infection or inflammatory disorders, and that severe or life-threatening CRS should be treated with tocilizumab with or without corticosteroids.
Patients should also be monitored for neurologic events after treatment with liso-cel, and supportive care and/or corticosteroids should be administered as needed.
This expanded indication is based on findings from the pivotal phase 3 TRANSFORM study, which showed significant and clinically meaningful improvements with CD19-directed chimeric antigen receptor T-cell immunotherapy over salvage chemotherapy followed by high-dose chemotherapy plus autologous stem cell transplant. The latter course of treatment had been the standard of care for more than 2 decades.
Data from the global, randomized, multicenter TRANSFORM study, as reported in December 2021 at the annual meeting of the American Society of Hematology, showed that second-line treatment with liso-cel in 92 patients with r/r LBCL within 12 months after first-line therapy, compared with 92 patient who received standard of care therapy, was associated with highly statistically significant and clinically meaningful improvement in event-free survival (10.1 vs. 2.3 months; hazard ratio, 0.349), complete response rate (66% vs. 39%), and progression-free survival (14.8 vs. 5.7 months; HR, 0.406).
A positive trend in overall survival was also observed (HR, 0.509 at median follow-up of 6.2 months). No new liso-cel safety signals were detected in the second-line setting.
Liso-cel was initially approved in February 2021 for the treatment of adults with LBCL, including diffuse LBCL not otherwise specified (including DLBCL arising from indolent lymphoma), high-grade B-cell lymphoma, primary mediastinal large B-cell lymphoma, and follicular lymphoma grade 3B, who have:
- Refractory disease to first-line chemoimmunotherapy or relapse within 12 months of first-line chemoimmunotherapy.
- Refractory disease to first-line chemoimmunotherapy or relapse after first-line chemoimmunotherapy and are not eligible for hematopoietic stem cell transplant because of comorbidities or age.
Liso-cel is not indicated for the treatment of patients with primary central nervous system lymphoma.
In February 2022, the FDA granted Priority Review status for a Bristol-Myers Squibb supplemental Biologics License Application (sBLA), based on the TRANSFORM study data, to expand the indication to include use after the failure of first-line therapy.
The agent “now has the potential to be a new standard of care for patients after failure of first-line therapy, offering significantly improved outcomes beyond the current mainstay of care,” Anne Kerber, the BMS senior vice president of cell therapy development, said in a press release at that time.
The European Medicines Agency has also validated a type II variation application for extension of the indication for liso-cel in this setting. Validation of the application “confirms the submission is complete and begins the EMA’s centralized review procedure,” BMS announced in a June 20, 2022, press release.
Liso-cel, which has been available only through a restricted program under a Risk Evaluation and Mitigation Strategy, includes a boxed warning regarding the risk for cytokine release syndrome (CRS) and neurologic toxicities.
The warning states that liso-cel should not be administered to patients with active infection or inflammatory disorders, and that severe or life-threatening CRS should be treated with tocilizumab with or without corticosteroids.
Patients should also be monitored for neurologic events after treatment with liso-cel, and supportive care and/or corticosteroids should be administered as needed.
This expanded indication is based on findings from the pivotal phase 3 TRANSFORM study, which showed significant and clinically meaningful improvements with CD19-directed chimeric antigen receptor T-cell immunotherapy over salvage chemotherapy followed by high-dose chemotherapy plus autologous stem cell transplant. The latter course of treatment had been the standard of care for more than 2 decades.
Data from the global, randomized, multicenter TRANSFORM study, as reported in December 2021 at the annual meeting of the American Society of Hematology, showed that second-line treatment with liso-cel in 92 patients with r/r LBCL within 12 months after first-line therapy, compared with 92 patient who received standard of care therapy, was associated with highly statistically significant and clinically meaningful improvement in event-free survival (10.1 vs. 2.3 months; hazard ratio, 0.349), complete response rate (66% vs. 39%), and progression-free survival (14.8 vs. 5.7 months; HR, 0.406).
A positive trend in overall survival was also observed (HR, 0.509 at median follow-up of 6.2 months). No new liso-cel safety signals were detected in the second-line setting.
Liso-cel was initially approved in February 2021 for the treatment of adults with LBCL, including diffuse LBCL not otherwise specified (including DLBCL arising from indolent lymphoma), high-grade B-cell lymphoma, primary mediastinal large B-cell lymphoma, and follicular lymphoma grade 3B, who have:
- Refractory disease to first-line chemoimmunotherapy or relapse within 12 months of first-line chemoimmunotherapy.
- Refractory disease to first-line chemoimmunotherapy or relapse after first-line chemoimmunotherapy and are not eligible for hematopoietic stem cell transplant because of comorbidities or age.
Liso-cel is not indicated for the treatment of patients with primary central nervous system lymphoma.
In February 2022, the FDA granted Priority Review status for a Bristol-Myers Squibb supplemental Biologics License Application (sBLA), based on the TRANSFORM study data, to expand the indication to include use after the failure of first-line therapy.
The agent “now has the potential to be a new standard of care for patients after failure of first-line therapy, offering significantly improved outcomes beyond the current mainstay of care,” Anne Kerber, the BMS senior vice president of cell therapy development, said in a press release at that time.
The European Medicines Agency has also validated a type II variation application for extension of the indication for liso-cel in this setting. Validation of the application “confirms the submission is complete and begins the EMA’s centralized review procedure,” BMS announced in a June 20, 2022, press release.
Liso-cel, which has been available only through a restricted program under a Risk Evaluation and Mitigation Strategy, includes a boxed warning regarding the risk for cytokine release syndrome (CRS) and neurologic toxicities.
The warning states that liso-cel should not be administered to patients with active infection or inflammatory disorders, and that severe or life-threatening CRS should be treated with tocilizumab with or without corticosteroids.
Patients should also be monitored for neurologic events after treatment with liso-cel, and supportive care and/or corticosteroids should be administered as needed.