User login
-
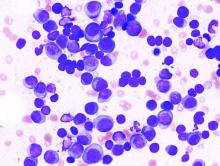
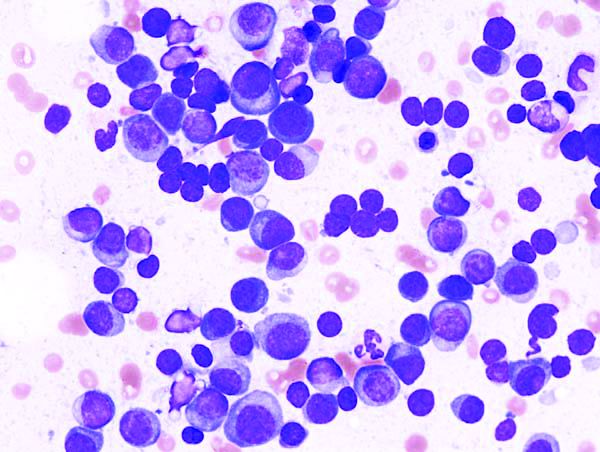
New CAR T-cell therapy eliminates MM and tumor propagating cells without fratricide in lab study
These cells proved to be active in vitro and in vivo against MM plasma cells, memory B cells, and MM-propagating cells, according to a report in Nature Communications.
This research is important because most MM patients eventually succumb to the disease and previously developed CAR T cells targeting B-cell maturation antigen (BCMA) on MM cells have shown high-response rates but limited durability.
Previous research showed that CD229/LY9 is a potential target for CAR T-cell therapy in MM because of its strong and homogeneous expression on the bulk of tumor cells, as well as chemotherapy-resistant myeloma progenitors; its absence from most normal cells; and dependence of MM cells on CD229 for their survival, according to Sabarinath V. Radhakrishnan, MD, of the University of Utah, Salt Lake City, and colleagues.
Using primary CD138+ tumor cells from three patients with plasma cell leukemia, a highly aggressive form of MM, which all showed high expression of CD229, the researchers found that CD229 CAR T cells exhibited high cytotoxic activity against these cells. In addition, when assessing two MM cell lines, U-266 and RPMI-8226, expressing different levels of CD229, they found that CD229 CAR T cells efficiently killed both cell lines in vitro.
“We do not observe fratricide during CD229 CAR T-cell production, as CD229 is downregulated in T cells during activation. In addition, while CD229 CAR T cells target normal CD229high T cells, they spare functional CD229neg/low T cells. These findings indicate that CD229 CAR T cells may be an effective treatment for patients with MM,” the authors concluded.
The study was funded by several nongovernmental organizations and the National Cancer Institute. Three of the authors are inventors on PCT application US2017/42840 “Antibodies and CAR T Cells for the Treatment of Multiple Myeloma” describing the therapeutic use of CD229 CAR T cells.
SOURCE: Radhakrishnan SV et al. Nat Commun. 2020 Feb 7;11(1):798. doi: 10.1038/s41467-020-14619-z.
These cells proved to be active in vitro and in vivo against MM plasma cells, memory B cells, and MM-propagating cells, according to a report in Nature Communications.
This research is important because most MM patients eventually succumb to the disease and previously developed CAR T cells targeting B-cell maturation antigen (BCMA) on MM cells have shown high-response rates but limited durability.
Previous research showed that CD229/LY9 is a potential target for CAR T-cell therapy in MM because of its strong and homogeneous expression on the bulk of tumor cells, as well as chemotherapy-resistant myeloma progenitors; its absence from most normal cells; and dependence of MM cells on CD229 for their survival, according to Sabarinath V. Radhakrishnan, MD, of the University of Utah, Salt Lake City, and colleagues.
Using primary CD138+ tumor cells from three patients with plasma cell leukemia, a highly aggressive form of MM, which all showed high expression of CD229, the researchers found that CD229 CAR T cells exhibited high cytotoxic activity against these cells. In addition, when assessing two MM cell lines, U-266 and RPMI-8226, expressing different levels of CD229, they found that CD229 CAR T cells efficiently killed both cell lines in vitro.
“We do not observe fratricide during CD229 CAR T-cell production, as CD229 is downregulated in T cells during activation. In addition, while CD229 CAR T cells target normal CD229high T cells, they spare functional CD229neg/low T cells. These findings indicate that CD229 CAR T cells may be an effective treatment for patients with MM,” the authors concluded.
The study was funded by several nongovernmental organizations and the National Cancer Institute. Three of the authors are inventors on PCT application US2017/42840 “Antibodies and CAR T Cells for the Treatment of Multiple Myeloma” describing the therapeutic use of CD229 CAR T cells.
SOURCE: Radhakrishnan SV et al. Nat Commun. 2020 Feb 7;11(1):798. doi: 10.1038/s41467-020-14619-z.
These cells proved to be active in vitro and in vivo against MM plasma cells, memory B cells, and MM-propagating cells, according to a report in Nature Communications.
This research is important because most MM patients eventually succumb to the disease and previously developed CAR T cells targeting B-cell maturation antigen (BCMA) on MM cells have shown high-response rates but limited durability.
Previous research showed that CD229/LY9 is a potential target for CAR T-cell therapy in MM because of its strong and homogeneous expression on the bulk of tumor cells, as well as chemotherapy-resistant myeloma progenitors; its absence from most normal cells; and dependence of MM cells on CD229 for their survival, according to Sabarinath V. Radhakrishnan, MD, of the University of Utah, Salt Lake City, and colleagues.
Using primary CD138+ tumor cells from three patients with plasma cell leukemia, a highly aggressive form of MM, which all showed high expression of CD229, the researchers found that CD229 CAR T cells exhibited high cytotoxic activity against these cells. In addition, when assessing two MM cell lines, U-266 and RPMI-8226, expressing different levels of CD229, they found that CD229 CAR T cells efficiently killed both cell lines in vitro.
“We do not observe fratricide during CD229 CAR T-cell production, as CD229 is downregulated in T cells during activation. In addition, while CD229 CAR T cells target normal CD229high T cells, they spare functional CD229neg/low T cells. These findings indicate that CD229 CAR T cells may be an effective treatment for patients with MM,” the authors concluded.
The study was funded by several nongovernmental organizations and the National Cancer Institute. Three of the authors are inventors on PCT application US2017/42840 “Antibodies and CAR T Cells for the Treatment of Multiple Myeloma” describing the therapeutic use of CD229 CAR T cells.
SOURCE: Radhakrishnan SV et al. Nat Commun. 2020 Feb 7;11(1):798. doi: 10.1038/s41467-020-14619-z.
FROM NATURE COMMUNICATIONS
Early results with ‘optimized’ gene therapy in hemophilia A
First-in-human results suggest a gene therapy, BAY 2599023, can increase factor VIII levels in patients with hemophilia A, but it isn’t clear if BAY 2599023 can eliminate the need for prophylaxis.
Of four patients who received BAY 2599023, three achieved clinically meaningful factor VIII levels, and all were able to stop prophylaxis at some point. However, one patient had to resume prophylaxis, and all four have experienced bleeds since receiving BAY 2599023.
This is a dose-escalating trial (NCT03588299), so researchers are still attempting to determine the optimal dose of BAY 2599023. Upcoming results in two patients treated at the highest dose should provide more insight, according to Steven W. Pipe, MD, of the University of Michigan in Ann Arbor.
Dr. Pipe presented results with BAY 2599023 at the annual congress of the European Association for Haemophilia and Allied Disorders.
He noted that between 30% and 70% of hemophilia patients may have preexisting neutralizing antibodies to specific adeno-associated virus (AAV) serotypes. Developing gene therapies with varying serotypes could reduce this problem and increase eligibility for gene therapy. Bayer had this in mind when developing BAY 2599023.
BAY 2599023 is a non-replicating AAV vector, based on the AAV serotype hu37, which contains a single-stranded DNA genome encoding a B-domain deleted factor VIII, under the control of a liver-specific promotor/enhancer combination optimized for transgenic expression, Dr. Pipe explained.
He presented results with BAY 2599023 in four patients with severe hemophilia A. At baseline, patients were receiving prophylaxis and did not have factor VIII inhibitors or detectable immunity to the AAVhu37 capsid.
First cohort
Two patients received BAY 2599023 at a dose of 0.5 × 1013 GC/kg. One patient achieved a factor VIII level above 5% at this dose and was able to stop prophylaxis temporarily. The patient experienced two bleeds on study and had to resume prophylaxis because of a target joint.
“He came into the study with a particularly challenging target joint in his ankle, and he had known from experience on prophylaxis that, if his factor level got too low, he would have breakthrough bleeds,” Dr. Pipe explained. “So we agreed that if he didn’t achieve a sustained level above 5%, he would go back on prophylaxis. He was able to stay off prophylaxis for 6 weeks, but we had to put him back on because of breakthrough bleeding.”
Dr. Pipe and colleagues have been following this patient for about a year, and the patient still has factor VIII levels of about 2% to 3% after a washout of prophylaxis.
The other patient who received a dose of 0.5 × 1013 GC/kg came off prophylaxis early on and has sustained factor VIII levels of 20%. This patient had four bleeds early in the trial but has since done very well, Dr. Pipe said. The patient had 99 bleeds in the year prior to receiving BAY 2599023.
Second cohort
Two subsequent patients received BAY 2599023 at a dose of 1 × 1013 GC/kg. Both patients have stopped prophylaxis.
One patient achieved factor VIII levels in the 5%-10% range and stopped prophylaxis at day 49. The patient has received on-demand treatment for four bleeds. The other patient achieved “robust” factor VIII expression levels “well above 20%,” according to Dr. Pipe. This patient stopped prophylaxis at day 10 and experienced bleeds early on but has remained bleed-free and treatment-free for 7 months.
Dr. Pipe noted that this patient experienced a spike in transaminases “very early on” after receiving BAY 2599023.
“So he was put on prednisone, but his liver enzymes almost immediately corrected to within the normal range,” Dr. Pipe said. “He has finished the cascading dose of prednisone, his liver enzymes have remained stable, his factor VIII expression has remained stable, and he continues to do very well.”
Next steps
Based on results in the first two cohorts, researchers are proceeding with a higher dose of BAY 2599023.
“The overall goal of this study is to identify the optimal dose that can be taken forward to the planned phase 3 study,” Dr. Pipe said. “What they [Bayer] would like to bring to the community is a product that offers a chance for maximum eligibility based on low screen out because of preexisting immunity and maximize the number of patients who are in the normal range.”
“For patients to embrace gene therapy, it really has to get them at least the possibility to land within the normal range and, hopefully, be not just liberated from prophylaxis but maybe liberated from the need for factor VIII at all,” Dr. Pipe added. “I think if these AAV platform therapies can’t deliver that on a relatively consistent basis, it’s going to be challenging to get patients to embrace this technology.”
The phase 1/2 trial is sponsored by Bayer in collaboration with Ultragenyx Pharmaceutical Inc. Dr. Pipe disclosed relationships with Bayer and other companies.
SOURCE: Pipe SW et al. EAHAD 2020. Abstract P190.
First-in-human results suggest a gene therapy, BAY 2599023, can increase factor VIII levels in patients with hemophilia A, but it isn’t clear if BAY 2599023 can eliminate the need for prophylaxis.
Of four patients who received BAY 2599023, three achieved clinically meaningful factor VIII levels, and all were able to stop prophylaxis at some point. However, one patient had to resume prophylaxis, and all four have experienced bleeds since receiving BAY 2599023.
This is a dose-escalating trial (NCT03588299), so researchers are still attempting to determine the optimal dose of BAY 2599023. Upcoming results in two patients treated at the highest dose should provide more insight, according to Steven W. Pipe, MD, of the University of Michigan in Ann Arbor.
Dr. Pipe presented results with BAY 2599023 at the annual congress of the European Association for Haemophilia and Allied Disorders.
He noted that between 30% and 70% of hemophilia patients may have preexisting neutralizing antibodies to specific adeno-associated virus (AAV) serotypes. Developing gene therapies with varying serotypes could reduce this problem and increase eligibility for gene therapy. Bayer had this in mind when developing BAY 2599023.
BAY 2599023 is a non-replicating AAV vector, based on the AAV serotype hu37, which contains a single-stranded DNA genome encoding a B-domain deleted factor VIII, under the control of a liver-specific promotor/enhancer combination optimized for transgenic expression, Dr. Pipe explained.
He presented results with BAY 2599023 in four patients with severe hemophilia A. At baseline, patients were receiving prophylaxis and did not have factor VIII inhibitors or detectable immunity to the AAVhu37 capsid.
First cohort
Two patients received BAY 2599023 at a dose of 0.5 × 1013 GC/kg. One patient achieved a factor VIII level above 5% at this dose and was able to stop prophylaxis temporarily. The patient experienced two bleeds on study and had to resume prophylaxis because of a target joint.
“He came into the study with a particularly challenging target joint in his ankle, and he had known from experience on prophylaxis that, if his factor level got too low, he would have breakthrough bleeds,” Dr. Pipe explained. “So we agreed that if he didn’t achieve a sustained level above 5%, he would go back on prophylaxis. He was able to stay off prophylaxis for 6 weeks, but we had to put him back on because of breakthrough bleeding.”
Dr. Pipe and colleagues have been following this patient for about a year, and the patient still has factor VIII levels of about 2% to 3% after a washout of prophylaxis.
The other patient who received a dose of 0.5 × 1013 GC/kg came off prophylaxis early on and has sustained factor VIII levels of 20%. This patient had four bleeds early in the trial but has since done very well, Dr. Pipe said. The patient had 99 bleeds in the year prior to receiving BAY 2599023.
Second cohort
Two subsequent patients received BAY 2599023 at a dose of 1 × 1013 GC/kg. Both patients have stopped prophylaxis.
One patient achieved factor VIII levels in the 5%-10% range and stopped prophylaxis at day 49. The patient has received on-demand treatment for four bleeds. The other patient achieved “robust” factor VIII expression levels “well above 20%,” according to Dr. Pipe. This patient stopped prophylaxis at day 10 and experienced bleeds early on but has remained bleed-free and treatment-free for 7 months.
Dr. Pipe noted that this patient experienced a spike in transaminases “very early on” after receiving BAY 2599023.
“So he was put on prednisone, but his liver enzymes almost immediately corrected to within the normal range,” Dr. Pipe said. “He has finished the cascading dose of prednisone, his liver enzymes have remained stable, his factor VIII expression has remained stable, and he continues to do very well.”
Next steps
Based on results in the first two cohorts, researchers are proceeding with a higher dose of BAY 2599023.
“The overall goal of this study is to identify the optimal dose that can be taken forward to the planned phase 3 study,” Dr. Pipe said. “What they [Bayer] would like to bring to the community is a product that offers a chance for maximum eligibility based on low screen out because of preexisting immunity and maximize the number of patients who are in the normal range.”
“For patients to embrace gene therapy, it really has to get them at least the possibility to land within the normal range and, hopefully, be not just liberated from prophylaxis but maybe liberated from the need for factor VIII at all,” Dr. Pipe added. “I think if these AAV platform therapies can’t deliver that on a relatively consistent basis, it’s going to be challenging to get patients to embrace this technology.”
The phase 1/2 trial is sponsored by Bayer in collaboration with Ultragenyx Pharmaceutical Inc. Dr. Pipe disclosed relationships with Bayer and other companies.
SOURCE: Pipe SW et al. EAHAD 2020. Abstract P190.
First-in-human results suggest a gene therapy, BAY 2599023, can increase factor VIII levels in patients with hemophilia A, but it isn’t clear if BAY 2599023 can eliminate the need for prophylaxis.
Of four patients who received BAY 2599023, three achieved clinically meaningful factor VIII levels, and all were able to stop prophylaxis at some point. However, one patient had to resume prophylaxis, and all four have experienced bleeds since receiving BAY 2599023.
This is a dose-escalating trial (NCT03588299), so researchers are still attempting to determine the optimal dose of BAY 2599023. Upcoming results in two patients treated at the highest dose should provide more insight, according to Steven W. Pipe, MD, of the University of Michigan in Ann Arbor.
Dr. Pipe presented results with BAY 2599023 at the annual congress of the European Association for Haemophilia and Allied Disorders.
He noted that between 30% and 70% of hemophilia patients may have preexisting neutralizing antibodies to specific adeno-associated virus (AAV) serotypes. Developing gene therapies with varying serotypes could reduce this problem and increase eligibility for gene therapy. Bayer had this in mind when developing BAY 2599023.
BAY 2599023 is a non-replicating AAV vector, based on the AAV serotype hu37, which contains a single-stranded DNA genome encoding a B-domain deleted factor VIII, under the control of a liver-specific promotor/enhancer combination optimized for transgenic expression, Dr. Pipe explained.
He presented results with BAY 2599023 in four patients with severe hemophilia A. At baseline, patients were receiving prophylaxis and did not have factor VIII inhibitors or detectable immunity to the AAVhu37 capsid.
First cohort
Two patients received BAY 2599023 at a dose of 0.5 × 1013 GC/kg. One patient achieved a factor VIII level above 5% at this dose and was able to stop prophylaxis temporarily. The patient experienced two bleeds on study and had to resume prophylaxis because of a target joint.
“He came into the study with a particularly challenging target joint in his ankle, and he had known from experience on prophylaxis that, if his factor level got too low, he would have breakthrough bleeds,” Dr. Pipe explained. “So we agreed that if he didn’t achieve a sustained level above 5%, he would go back on prophylaxis. He was able to stay off prophylaxis for 6 weeks, but we had to put him back on because of breakthrough bleeding.”
Dr. Pipe and colleagues have been following this patient for about a year, and the patient still has factor VIII levels of about 2% to 3% after a washout of prophylaxis.
The other patient who received a dose of 0.5 × 1013 GC/kg came off prophylaxis early on and has sustained factor VIII levels of 20%. This patient had four bleeds early in the trial but has since done very well, Dr. Pipe said. The patient had 99 bleeds in the year prior to receiving BAY 2599023.
Second cohort
Two subsequent patients received BAY 2599023 at a dose of 1 × 1013 GC/kg. Both patients have stopped prophylaxis.
One patient achieved factor VIII levels in the 5%-10% range and stopped prophylaxis at day 49. The patient has received on-demand treatment for four bleeds. The other patient achieved “robust” factor VIII expression levels “well above 20%,” according to Dr. Pipe. This patient stopped prophylaxis at day 10 and experienced bleeds early on but has remained bleed-free and treatment-free for 7 months.
Dr. Pipe noted that this patient experienced a spike in transaminases “very early on” after receiving BAY 2599023.
“So he was put on prednisone, but his liver enzymes almost immediately corrected to within the normal range,” Dr. Pipe said. “He has finished the cascading dose of prednisone, his liver enzymes have remained stable, his factor VIII expression has remained stable, and he continues to do very well.”
Next steps
Based on results in the first two cohorts, researchers are proceeding with a higher dose of BAY 2599023.
“The overall goal of this study is to identify the optimal dose that can be taken forward to the planned phase 3 study,” Dr. Pipe said. “What they [Bayer] would like to bring to the community is a product that offers a chance for maximum eligibility based on low screen out because of preexisting immunity and maximize the number of patients who are in the normal range.”
“For patients to embrace gene therapy, it really has to get them at least the possibility to land within the normal range and, hopefully, be not just liberated from prophylaxis but maybe liberated from the need for factor VIII at all,” Dr. Pipe added. “I think if these AAV platform therapies can’t deliver that on a relatively consistent basis, it’s going to be challenging to get patients to embrace this technology.”
The phase 1/2 trial is sponsored by Bayer in collaboration with Ultragenyx Pharmaceutical Inc. Dr. Pipe disclosed relationships with Bayer and other companies.
SOURCE: Pipe SW et al. EAHAD 2020. Abstract P190.
REPORTING FROM EAHAD 2020
Are patient portals living up to the hype? Ask your mother-in-law!
While preparing to write this technology column, I received a great deal of insight from the unlikeliest of sources: my mother-in-law.
Now don’t get me wrong – she’s a truly lovely, intelligent, and capable woman. I have sought her advice often on many things and have always been impressed by her wisdom and pragmatism, but I’ve just never thought of asking her for her opinion on medicine or technology, as I considered her knowledge of both subjects to be limited.
This occasion changed my opinion. In fact, I believe that, as health care IT becomes more complex, people like my mother-in-law may be exactly who we should be looking to for answers.
A few weeks ago, my mother-in-law and I were discussing her recent trip to the doctor. When she mentioned some lab tests, I suggested that we log in to her patient portal to view the results. This elicited several questions and a declaration of frustration.
“Which portal?” she asked. “I have so many and can’t keep all of the websites and passwords straight! Why can’t all of my doctors use the same portal, and why do they all have different password requirements?”
As she spoke these words, I was immediately struck with an unfortunate reality of EHRs: We have done a brilliant job creating state-of-the-art digital castles and have filled them with the data needed to revolutionize care and improve population health – but we haven’t given our patients the keys to get inside.
We must ask ourselves if, in trying to construct fortresses of information around our patients, we have lost sight of the individuals in the center. I believe that we can answer this question and improve the benefits of patient portals, but we all must agree to a few simple steps to streamline the experience for everyone.
Make it easy
A study recently published in the Journal of General Internal Medicine surveyed several hospitals on their usage of patient portals. After determining whether or not the institutions had such portals, the authors then investigated to find out what, if any, guidance was provided to patients about how to use them.
Their findings are frustrating, though not surprising. While 89% of hospitals had some form of patient portal, only 65% of those “had links that were easily found, defined as links accessible within two clicks from the home page.”
Furthermore, even in cases where portals were easily found, good instructions on how to use them were missing. Those instructions that did exist centered on rules and restrictions and laying out “terms and conditions” and informing patients on “what not to do,” rather than explaining how to make the most of the experience.
According to the authors, “this focus on curtailing behavior, and the hurdles placed on finding and understanding guidance, suggest that some hospitals may be prioritizing reducing liability over improving the patient experience with portals.”
If we want our patients to use them, portals must be easy to access and intuitive to use. They also must provide value.
Make it meaningful
Patient portals have proliferated exponentially over the last 10 years, thanks to government incentive programs. One such program, known as “meaningful use,” is primarily responsible for this, as it made implementation of a patient portal one of its core requirements.
Sadly, in spite of its oft-reviled name, the meaningful use program never defined patient-friendly standards of usability for patient portals. As a result, current portals just aren’t very good. Patients like my mother-in-law find them to be too numerous, too unfriendly to use, and too limited, so they are not being used to their full potential.
In fact, many institutions may choose not to enable all of the available features in order to limit technical issues and reduce the burden on providers. In the study referenced above, only 63% of portals offered the ability for patients to communicate directly with their physicians, and only 43% offered the ability to refill prescriptions.
When enabled, these functions improve patient engagement and efficiency. Without them, patients are less likely to log on, and physicians are forced to rely on less-efficient telephone calls or traditional letters to communicate results to their patients.
Put the patient, not the portal, at the center
History has all but forgotten the attempts by tech giants such as Google and Microsoft to create personal health records. While these initially seemed like a wonderful concept, they sadly proved to be a total flop. Some patients embraced the idea, but security concerns and the lack of buy-in from EHR vendors significantly limited their uptake.
They may simply have been ahead of their time.
A decade later, wearable technology and telemedicine are ushering in a new era of patient-centric care. Individuals have been embracing a greater share of the responsibility for their own personal health information, yet most EHRs lack the ability to easily incorporate data acquired outside physicians’ offices.
It’s time for EHR vendors to go all in and change that. Instead of enslaving patients to the tyranny of fragmented health records, they should prioritize the creation of a robust, standardized, and portable health record that travels with the patient, not the other way around.
Have any other ideas on how to improve patient engagement? We’d love to hear about them and share them in a future column.
If you want to contribute but don’t have any ideas, we have a suggestion: Ask your mother-in-law. You may be surprised at what you learn!
Dr. Notte is a family physician and associate chief medical information officer for Abington (Pa.) Hospital–Jefferson Health. Follow him on twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health.
Reference
Lee JL et al. J Gen Intern Med. 2019 Nov 12. doi: 10.1007/s11606-019-05528-z.
While preparing to write this technology column, I received a great deal of insight from the unlikeliest of sources: my mother-in-law.
Now don’t get me wrong – she’s a truly lovely, intelligent, and capable woman. I have sought her advice often on many things and have always been impressed by her wisdom and pragmatism, but I’ve just never thought of asking her for her opinion on medicine or technology, as I considered her knowledge of both subjects to be limited.
This occasion changed my opinion. In fact, I believe that, as health care IT becomes more complex, people like my mother-in-law may be exactly who we should be looking to for answers.
A few weeks ago, my mother-in-law and I were discussing her recent trip to the doctor. When she mentioned some lab tests, I suggested that we log in to her patient portal to view the results. This elicited several questions and a declaration of frustration.
“Which portal?” she asked. “I have so many and can’t keep all of the websites and passwords straight! Why can’t all of my doctors use the same portal, and why do they all have different password requirements?”
As she spoke these words, I was immediately struck with an unfortunate reality of EHRs: We have done a brilliant job creating state-of-the-art digital castles and have filled them with the data needed to revolutionize care and improve population health – but we haven’t given our patients the keys to get inside.
We must ask ourselves if, in trying to construct fortresses of information around our patients, we have lost sight of the individuals in the center. I believe that we can answer this question and improve the benefits of patient portals, but we all must agree to a few simple steps to streamline the experience for everyone.
Make it easy
A study recently published in the Journal of General Internal Medicine surveyed several hospitals on their usage of patient portals. After determining whether or not the institutions had such portals, the authors then investigated to find out what, if any, guidance was provided to patients about how to use them.
Their findings are frustrating, though not surprising. While 89% of hospitals had some form of patient portal, only 65% of those “had links that were easily found, defined as links accessible within two clicks from the home page.”
Furthermore, even in cases where portals were easily found, good instructions on how to use them were missing. Those instructions that did exist centered on rules and restrictions and laying out “terms and conditions” and informing patients on “what not to do,” rather than explaining how to make the most of the experience.
According to the authors, “this focus on curtailing behavior, and the hurdles placed on finding and understanding guidance, suggest that some hospitals may be prioritizing reducing liability over improving the patient experience with portals.”
If we want our patients to use them, portals must be easy to access and intuitive to use. They also must provide value.
Make it meaningful
Patient portals have proliferated exponentially over the last 10 years, thanks to government incentive programs. One such program, known as “meaningful use,” is primarily responsible for this, as it made implementation of a patient portal one of its core requirements.
Sadly, in spite of its oft-reviled name, the meaningful use program never defined patient-friendly standards of usability for patient portals. As a result, current portals just aren’t very good. Patients like my mother-in-law find them to be too numerous, too unfriendly to use, and too limited, so they are not being used to their full potential.
In fact, many institutions may choose not to enable all of the available features in order to limit technical issues and reduce the burden on providers. In the study referenced above, only 63% of portals offered the ability for patients to communicate directly with their physicians, and only 43% offered the ability to refill prescriptions.
When enabled, these functions improve patient engagement and efficiency. Without them, patients are less likely to log on, and physicians are forced to rely on less-efficient telephone calls or traditional letters to communicate results to their patients.
Put the patient, not the portal, at the center
History has all but forgotten the attempts by tech giants such as Google and Microsoft to create personal health records. While these initially seemed like a wonderful concept, they sadly proved to be a total flop. Some patients embraced the idea, but security concerns and the lack of buy-in from EHR vendors significantly limited their uptake.
They may simply have been ahead of their time.
A decade later, wearable technology and telemedicine are ushering in a new era of patient-centric care. Individuals have been embracing a greater share of the responsibility for their own personal health information, yet most EHRs lack the ability to easily incorporate data acquired outside physicians’ offices.
It’s time for EHR vendors to go all in and change that. Instead of enslaving patients to the tyranny of fragmented health records, they should prioritize the creation of a robust, standardized, and portable health record that travels with the patient, not the other way around.
Have any other ideas on how to improve patient engagement? We’d love to hear about them and share them in a future column.
If you want to contribute but don’t have any ideas, we have a suggestion: Ask your mother-in-law. You may be surprised at what you learn!
Dr. Notte is a family physician and associate chief medical information officer for Abington (Pa.) Hospital–Jefferson Health. Follow him on twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health.
Reference
Lee JL et al. J Gen Intern Med. 2019 Nov 12. doi: 10.1007/s11606-019-05528-z.
While preparing to write this technology column, I received a great deal of insight from the unlikeliest of sources: my mother-in-law.
Now don’t get me wrong – she’s a truly lovely, intelligent, and capable woman. I have sought her advice often on many things and have always been impressed by her wisdom and pragmatism, but I’ve just never thought of asking her for her opinion on medicine or technology, as I considered her knowledge of both subjects to be limited.
This occasion changed my opinion. In fact, I believe that, as health care IT becomes more complex, people like my mother-in-law may be exactly who we should be looking to for answers.
A few weeks ago, my mother-in-law and I were discussing her recent trip to the doctor. When she mentioned some lab tests, I suggested that we log in to her patient portal to view the results. This elicited several questions and a declaration of frustration.
“Which portal?” she asked. “I have so many and can’t keep all of the websites and passwords straight! Why can’t all of my doctors use the same portal, and why do they all have different password requirements?”
As she spoke these words, I was immediately struck with an unfortunate reality of EHRs: We have done a brilliant job creating state-of-the-art digital castles and have filled them with the data needed to revolutionize care and improve population health – but we haven’t given our patients the keys to get inside.
We must ask ourselves if, in trying to construct fortresses of information around our patients, we have lost sight of the individuals in the center. I believe that we can answer this question and improve the benefits of patient portals, but we all must agree to a few simple steps to streamline the experience for everyone.
Make it easy
A study recently published in the Journal of General Internal Medicine surveyed several hospitals on their usage of patient portals. After determining whether or not the institutions had such portals, the authors then investigated to find out what, if any, guidance was provided to patients about how to use them.
Their findings are frustrating, though not surprising. While 89% of hospitals had some form of patient portal, only 65% of those “had links that were easily found, defined as links accessible within two clicks from the home page.”
Furthermore, even in cases where portals were easily found, good instructions on how to use them were missing. Those instructions that did exist centered on rules and restrictions and laying out “terms and conditions” and informing patients on “what not to do,” rather than explaining how to make the most of the experience.
According to the authors, “this focus on curtailing behavior, and the hurdles placed on finding and understanding guidance, suggest that some hospitals may be prioritizing reducing liability over improving the patient experience with portals.”
If we want our patients to use them, portals must be easy to access and intuitive to use. They also must provide value.
Make it meaningful
Patient portals have proliferated exponentially over the last 10 years, thanks to government incentive programs. One such program, known as “meaningful use,” is primarily responsible for this, as it made implementation of a patient portal one of its core requirements.
Sadly, in spite of its oft-reviled name, the meaningful use program never defined patient-friendly standards of usability for patient portals. As a result, current portals just aren’t very good. Patients like my mother-in-law find them to be too numerous, too unfriendly to use, and too limited, so they are not being used to their full potential.
In fact, many institutions may choose not to enable all of the available features in order to limit technical issues and reduce the burden on providers. In the study referenced above, only 63% of portals offered the ability for patients to communicate directly with their physicians, and only 43% offered the ability to refill prescriptions.
When enabled, these functions improve patient engagement and efficiency. Without them, patients are less likely to log on, and physicians are forced to rely on less-efficient telephone calls or traditional letters to communicate results to their patients.
Put the patient, not the portal, at the center
History has all but forgotten the attempts by tech giants such as Google and Microsoft to create personal health records. While these initially seemed like a wonderful concept, they sadly proved to be a total flop. Some patients embraced the idea, but security concerns and the lack of buy-in from EHR vendors significantly limited their uptake.
They may simply have been ahead of their time.
A decade later, wearable technology and telemedicine are ushering in a new era of patient-centric care. Individuals have been embracing a greater share of the responsibility for their own personal health information, yet most EHRs lack the ability to easily incorporate data acquired outside physicians’ offices.
It’s time for EHR vendors to go all in and change that. Instead of enslaving patients to the tyranny of fragmented health records, they should prioritize the creation of a robust, standardized, and portable health record that travels with the patient, not the other way around.
Have any other ideas on how to improve patient engagement? We’d love to hear about them and share them in a future column.
If you want to contribute but don’t have any ideas, we have a suggestion: Ask your mother-in-law. You may be surprised at what you learn!
Dr. Notte is a family physician and associate chief medical information officer for Abington (Pa.) Hospital–Jefferson Health. Follow him on twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health.
Reference
Lee JL et al. J Gen Intern Med. 2019 Nov 12. doi: 10.1007/s11606-019-05528-z.
Supreme Court roundup: Latest health care decisions
The Trump administration can move forward with expanding a rule that makes it more difficult for immigrants to remain in the United States if they receive health care assistance, the U.S. Supreme Court ruled in a 5-4 vote.
The Feb. 21 order allows the administration to broaden the so-called “public charge rule” while legal challenges against the expanded regulation continue in the lower courts. The Supreme Court’s decision, which lifts a preliminary injunction against the expansion, applies to enforcement only in Illinois, where a district court blocked the revised rule from moving forward in October 2019. The Supreme Court’s measure follows another 5-4 order in January, in which justices lifted a nationwide injunction against the revised rule.
Under the long-standing public charge rule, immigration officials can refuse to admit immigrants into the United States or can deny them permanent legal status if they are deemed likely to become a public charge. Previously, immigration officers considered cash aid, such as Temporary Assistance for Needy Families or long-term institutionalized care, as potential public charge reasons for denial.
The revised regulation allows officials to consider previously excluded programs in their determination, including nonemergency Medicaid, the Supplemental Nutrition Assistance Program, and several housing programs. Use of these programs for more than 12 months in the aggregate during a 36-month period may result in a “public charge” designation and lead to green card denial.
Eight legal challenges were immediately filed against the rule changes, including a complaint issued by 14 states. At least five trial courts have since blocked the measure, while appeals courts have lifted some of the injunctions and upheld enforcement.
In its Jan. 27 order lifting the nationwide injunction, Associate Justice Neil M. Gorsuch wrote that nationwide injunctions are being overused by trial courts with negative consequences.
“The real problem here is the increasingly common practice of trial courts ordering relief that transcends the cases before them. Whether framed as injunctions of ‘nationwide,’ ‘universal,’ or ‘cosmic’ scope, these orders share the same basic flaw – they direct how the defendant must act toward persons who are not parties to the case,” he wrote. “It has become increasingly apparent that this court must, at some point, confront these important objections to this increasingly widespread practice. As the brief and furious history of the regulation before us illustrates, the routine issuance of universal injunctions is patently unworkable, sowing chaos for litigants, the government, courts, and all those affected by these conflicting decisions.”
In the court’s Feb. 21 order lifting the injunction in Illinois, justices gave no explanation for overturning the lower court’s injunction. However, Associate Justice Sonia Sotomayor issued a sharply-worded dissent, criticizing her fellow justices for allowing the rule to proceed.
“In sum, the government’s only claimed hardship is that it must enforce an existing interpretation of an immigration rule in one state – just as it has done for the past 20 years – while an updated version of the rule takes effect in the remaining 49,” she wrote. “The government has not quantified or explained any burdens that would arise from this state of the world.”
ACA cases still in limbo
Meanwhile, the Supreme Court still has not decided whether it will hear Texas v. United States, a case that could effectively dismantle the Affordable Care Act.
The high court was expected to announce whether it would take the high-profile case at a private Feb. 21 conference, but the justices have released no update. The case was relisted for consideration at the court’s Feb. 28 conference.
Texas v. United States stems from a lawsuit by 20 Republican state attorneys general and governors that was filed after Congress zeroed out the ACA’s individual mandate penalty in 2017. The plaintiffs contend the now-valueless mandate is no longer constitutional and thus, the entire ACA should be struck down. Because the Trump administration declined to defend the law, a coalition of Democratic attorneys general and governors intervened in the case as defendants.
In 2018, a Texas district court ruled in favor of the plaintiffs and declared the entire health care law invalid. The 5th U.S. Circuit Court of Appeals partially affirmed the district court’s decision, ruling that the mandate was unconstitutional, but sending the case back to the lower court for more analysis on severability. The Democratic attorneys general and governors appealed the decision to the U.S. Supreme Court.
If the Supreme Court agrees to hear the challenge, the court could fast-track the case and schedule arguments for the current term or wait until its next term, which starts in October 2020. If justices decline to hear the case, the challenge will remain with the district court for more analysis about the law’s severability.
Another ACA-related case – Maine Community Health Options v. U.S. – also remains in limbo. Justices heard the case, which was consolidated with two similar challenges, on Dec. 10, 2019, but still have not issued a decision.
The consolidated challenges center on whether the federal government owes insurers billions based on an Affordable Care Act provision intended to help health plans mitigate risk under the law. The ACA’s risk corridor program required the U.S. Department of Health & Human Services to collect funds from profitable insurers that offered qualified health plans under the exchanges and distribute the funds to insurers with excessive losses. Collections from profitable insurers under the program fell short in 2014, 2015, and 2016, while losses steadily grew, resulting in the HHS paying about 12 cents on the dollar in payments to insurers. More than 150 insurers now allege they were shortchanged and they want the Supreme Court to force the government to reimburse them to the tune of $12 billion.
The Department of Justice counters that the government is not required to pay the insurers because of appropriations measures passed by Congress in 2014 and in later years that limited the funding available to compensate insurers for their losses.
The federal government and insurers have each experienced wins and losses at the lower court level. Most recently, the U.S. Court of Appeals for the Federal Circuit decided in favor of the government, ruling that while the ACA required the government to compensate the insurers for their losses, the appropriations measures repealed or suspended that requirement.
A Supreme Court decision in the case could come as soon as Feb. 26.
Court to hear women’s health cases
Two closely watched reproductive health cases will go before the court this spring.
On March 4, justices will hear oral arguments in June Medical Services v. Russo, regarding the constitutionality of a Louisiana law that requires physicians performing abortions to have admitting privileges at a nearby hospital. Doctors who perform abortions without admitting privileges at a hospital within 30 miles face fines and imprisonment, according to the state law, originally passed in 2014. Clinics that employ such doctors can also have their licenses revoked.
June Medical Services LLC, a women’s health clinic, sued over the law. A district court ruled in favor of the plaintiff, but the 5th U.S. Circuit Court of Appeals reversed and upheld Louisiana’s law. The clinic appealed to the U.S. Supreme Court. Louisiana officials argue the challenge should be dismissed, and the law allowed to proceed, because the plaintiffs lack standing.
The Supreme Court in 2016 heard a similar case – Whole Woman’s Health v. Hellerstedt – concerning a comparable law in Texas. In that case, justices struck down the measure as unconstitutional.
And on April 29, justices will hear arguments in Little Sisters of the Poor v. Pennsylvania, a consolidated case about whether the Trump administration acted properly when it expanded exemptions under the Affordable Care Act’s contraceptive mandate. Entities that object to providing contraception on the basis of religious beliefs can opt out of complying with the mandate, according to the 2018 regulations. Additionally, nonprofit organizations and small businesses that have nonreligious moral convictions against the mandate can skip compliance. A number of states and entities sued over the new rules.
A federal appeals court temporarily barred the regulations from moving forward, ruling the plaintiffs were likely to succeed in proving the Trump administration did not follow appropriate procedures when it promulgated the new rules and that the regulations were not authorized under the ACA.
Justices will decide whether the parties have standing in the case, whether the Trump administration followed correct rule-making procedures, and if the regulations can stand.
The Trump administration can move forward with expanding a rule that makes it more difficult for immigrants to remain in the United States if they receive health care assistance, the U.S. Supreme Court ruled in a 5-4 vote.
The Feb. 21 order allows the administration to broaden the so-called “public charge rule” while legal challenges against the expanded regulation continue in the lower courts. The Supreme Court’s decision, which lifts a preliminary injunction against the expansion, applies to enforcement only in Illinois, where a district court blocked the revised rule from moving forward in October 2019. The Supreme Court’s measure follows another 5-4 order in January, in which justices lifted a nationwide injunction against the revised rule.
Under the long-standing public charge rule, immigration officials can refuse to admit immigrants into the United States or can deny them permanent legal status if they are deemed likely to become a public charge. Previously, immigration officers considered cash aid, such as Temporary Assistance for Needy Families or long-term institutionalized care, as potential public charge reasons for denial.
The revised regulation allows officials to consider previously excluded programs in their determination, including nonemergency Medicaid, the Supplemental Nutrition Assistance Program, and several housing programs. Use of these programs for more than 12 months in the aggregate during a 36-month period may result in a “public charge” designation and lead to green card denial.
Eight legal challenges were immediately filed against the rule changes, including a complaint issued by 14 states. At least five trial courts have since blocked the measure, while appeals courts have lifted some of the injunctions and upheld enforcement.
In its Jan. 27 order lifting the nationwide injunction, Associate Justice Neil M. Gorsuch wrote that nationwide injunctions are being overused by trial courts with negative consequences.
“The real problem here is the increasingly common practice of trial courts ordering relief that transcends the cases before them. Whether framed as injunctions of ‘nationwide,’ ‘universal,’ or ‘cosmic’ scope, these orders share the same basic flaw – they direct how the defendant must act toward persons who are not parties to the case,” he wrote. “It has become increasingly apparent that this court must, at some point, confront these important objections to this increasingly widespread practice. As the brief and furious history of the regulation before us illustrates, the routine issuance of universal injunctions is patently unworkable, sowing chaos for litigants, the government, courts, and all those affected by these conflicting decisions.”
In the court’s Feb. 21 order lifting the injunction in Illinois, justices gave no explanation for overturning the lower court’s injunction. However, Associate Justice Sonia Sotomayor issued a sharply-worded dissent, criticizing her fellow justices for allowing the rule to proceed.
“In sum, the government’s only claimed hardship is that it must enforce an existing interpretation of an immigration rule in one state – just as it has done for the past 20 years – while an updated version of the rule takes effect in the remaining 49,” she wrote. “The government has not quantified or explained any burdens that would arise from this state of the world.”
ACA cases still in limbo
Meanwhile, the Supreme Court still has not decided whether it will hear Texas v. United States, a case that could effectively dismantle the Affordable Care Act.
The high court was expected to announce whether it would take the high-profile case at a private Feb. 21 conference, but the justices have released no update. The case was relisted for consideration at the court’s Feb. 28 conference.
Texas v. United States stems from a lawsuit by 20 Republican state attorneys general and governors that was filed after Congress zeroed out the ACA’s individual mandate penalty in 2017. The plaintiffs contend the now-valueless mandate is no longer constitutional and thus, the entire ACA should be struck down. Because the Trump administration declined to defend the law, a coalition of Democratic attorneys general and governors intervened in the case as defendants.
In 2018, a Texas district court ruled in favor of the plaintiffs and declared the entire health care law invalid. The 5th U.S. Circuit Court of Appeals partially affirmed the district court’s decision, ruling that the mandate was unconstitutional, but sending the case back to the lower court for more analysis on severability. The Democratic attorneys general and governors appealed the decision to the U.S. Supreme Court.
If the Supreme Court agrees to hear the challenge, the court could fast-track the case and schedule arguments for the current term or wait until its next term, which starts in October 2020. If justices decline to hear the case, the challenge will remain with the district court for more analysis about the law’s severability.
Another ACA-related case – Maine Community Health Options v. U.S. – also remains in limbo. Justices heard the case, which was consolidated with two similar challenges, on Dec. 10, 2019, but still have not issued a decision.
The consolidated challenges center on whether the federal government owes insurers billions based on an Affordable Care Act provision intended to help health plans mitigate risk under the law. The ACA’s risk corridor program required the U.S. Department of Health & Human Services to collect funds from profitable insurers that offered qualified health plans under the exchanges and distribute the funds to insurers with excessive losses. Collections from profitable insurers under the program fell short in 2014, 2015, and 2016, while losses steadily grew, resulting in the HHS paying about 12 cents on the dollar in payments to insurers. More than 150 insurers now allege they were shortchanged and they want the Supreme Court to force the government to reimburse them to the tune of $12 billion.
The Department of Justice counters that the government is not required to pay the insurers because of appropriations measures passed by Congress in 2014 and in later years that limited the funding available to compensate insurers for their losses.
The federal government and insurers have each experienced wins and losses at the lower court level. Most recently, the U.S. Court of Appeals for the Federal Circuit decided in favor of the government, ruling that while the ACA required the government to compensate the insurers for their losses, the appropriations measures repealed or suspended that requirement.
A Supreme Court decision in the case could come as soon as Feb. 26.
Court to hear women’s health cases
Two closely watched reproductive health cases will go before the court this spring.
On March 4, justices will hear oral arguments in June Medical Services v. Russo, regarding the constitutionality of a Louisiana law that requires physicians performing abortions to have admitting privileges at a nearby hospital. Doctors who perform abortions without admitting privileges at a hospital within 30 miles face fines and imprisonment, according to the state law, originally passed in 2014. Clinics that employ such doctors can also have their licenses revoked.
June Medical Services LLC, a women’s health clinic, sued over the law. A district court ruled in favor of the plaintiff, but the 5th U.S. Circuit Court of Appeals reversed and upheld Louisiana’s law. The clinic appealed to the U.S. Supreme Court. Louisiana officials argue the challenge should be dismissed, and the law allowed to proceed, because the plaintiffs lack standing.
The Supreme Court in 2016 heard a similar case – Whole Woman’s Health v. Hellerstedt – concerning a comparable law in Texas. In that case, justices struck down the measure as unconstitutional.
And on April 29, justices will hear arguments in Little Sisters of the Poor v. Pennsylvania, a consolidated case about whether the Trump administration acted properly when it expanded exemptions under the Affordable Care Act’s contraceptive mandate. Entities that object to providing contraception on the basis of religious beliefs can opt out of complying with the mandate, according to the 2018 regulations. Additionally, nonprofit organizations and small businesses that have nonreligious moral convictions against the mandate can skip compliance. A number of states and entities sued over the new rules.
A federal appeals court temporarily barred the regulations from moving forward, ruling the plaintiffs were likely to succeed in proving the Trump administration did not follow appropriate procedures when it promulgated the new rules and that the regulations were not authorized under the ACA.
Justices will decide whether the parties have standing in the case, whether the Trump administration followed correct rule-making procedures, and if the regulations can stand.
The Trump administration can move forward with expanding a rule that makes it more difficult for immigrants to remain in the United States if they receive health care assistance, the U.S. Supreme Court ruled in a 5-4 vote.
The Feb. 21 order allows the administration to broaden the so-called “public charge rule” while legal challenges against the expanded regulation continue in the lower courts. The Supreme Court’s decision, which lifts a preliminary injunction against the expansion, applies to enforcement only in Illinois, where a district court blocked the revised rule from moving forward in October 2019. The Supreme Court’s measure follows another 5-4 order in January, in which justices lifted a nationwide injunction against the revised rule.
Under the long-standing public charge rule, immigration officials can refuse to admit immigrants into the United States or can deny them permanent legal status if they are deemed likely to become a public charge. Previously, immigration officers considered cash aid, such as Temporary Assistance for Needy Families or long-term institutionalized care, as potential public charge reasons for denial.
The revised regulation allows officials to consider previously excluded programs in their determination, including nonemergency Medicaid, the Supplemental Nutrition Assistance Program, and several housing programs. Use of these programs for more than 12 months in the aggregate during a 36-month period may result in a “public charge” designation and lead to green card denial.
Eight legal challenges were immediately filed against the rule changes, including a complaint issued by 14 states. At least five trial courts have since blocked the measure, while appeals courts have lifted some of the injunctions and upheld enforcement.
In its Jan. 27 order lifting the nationwide injunction, Associate Justice Neil M. Gorsuch wrote that nationwide injunctions are being overused by trial courts with negative consequences.
“The real problem here is the increasingly common practice of trial courts ordering relief that transcends the cases before them. Whether framed as injunctions of ‘nationwide,’ ‘universal,’ or ‘cosmic’ scope, these orders share the same basic flaw – they direct how the defendant must act toward persons who are not parties to the case,” he wrote. “It has become increasingly apparent that this court must, at some point, confront these important objections to this increasingly widespread practice. As the brief and furious history of the regulation before us illustrates, the routine issuance of universal injunctions is patently unworkable, sowing chaos for litigants, the government, courts, and all those affected by these conflicting decisions.”
In the court’s Feb. 21 order lifting the injunction in Illinois, justices gave no explanation for overturning the lower court’s injunction. However, Associate Justice Sonia Sotomayor issued a sharply-worded dissent, criticizing her fellow justices for allowing the rule to proceed.
“In sum, the government’s only claimed hardship is that it must enforce an existing interpretation of an immigration rule in one state – just as it has done for the past 20 years – while an updated version of the rule takes effect in the remaining 49,” she wrote. “The government has not quantified or explained any burdens that would arise from this state of the world.”
ACA cases still in limbo
Meanwhile, the Supreme Court still has not decided whether it will hear Texas v. United States, a case that could effectively dismantle the Affordable Care Act.
The high court was expected to announce whether it would take the high-profile case at a private Feb. 21 conference, but the justices have released no update. The case was relisted for consideration at the court’s Feb. 28 conference.
Texas v. United States stems from a lawsuit by 20 Republican state attorneys general and governors that was filed after Congress zeroed out the ACA’s individual mandate penalty in 2017. The plaintiffs contend the now-valueless mandate is no longer constitutional and thus, the entire ACA should be struck down. Because the Trump administration declined to defend the law, a coalition of Democratic attorneys general and governors intervened in the case as defendants.
In 2018, a Texas district court ruled in favor of the plaintiffs and declared the entire health care law invalid. The 5th U.S. Circuit Court of Appeals partially affirmed the district court’s decision, ruling that the mandate was unconstitutional, but sending the case back to the lower court for more analysis on severability. The Democratic attorneys general and governors appealed the decision to the U.S. Supreme Court.
If the Supreme Court agrees to hear the challenge, the court could fast-track the case and schedule arguments for the current term or wait until its next term, which starts in October 2020. If justices decline to hear the case, the challenge will remain with the district court for more analysis about the law’s severability.
Another ACA-related case – Maine Community Health Options v. U.S. – also remains in limbo. Justices heard the case, which was consolidated with two similar challenges, on Dec. 10, 2019, but still have not issued a decision.
The consolidated challenges center on whether the federal government owes insurers billions based on an Affordable Care Act provision intended to help health plans mitigate risk under the law. The ACA’s risk corridor program required the U.S. Department of Health & Human Services to collect funds from profitable insurers that offered qualified health plans under the exchanges and distribute the funds to insurers with excessive losses. Collections from profitable insurers under the program fell short in 2014, 2015, and 2016, while losses steadily grew, resulting in the HHS paying about 12 cents on the dollar in payments to insurers. More than 150 insurers now allege they were shortchanged and they want the Supreme Court to force the government to reimburse them to the tune of $12 billion.
The Department of Justice counters that the government is not required to pay the insurers because of appropriations measures passed by Congress in 2014 and in later years that limited the funding available to compensate insurers for their losses.
The federal government and insurers have each experienced wins and losses at the lower court level. Most recently, the U.S. Court of Appeals for the Federal Circuit decided in favor of the government, ruling that while the ACA required the government to compensate the insurers for their losses, the appropriations measures repealed or suspended that requirement.
A Supreme Court decision in the case could come as soon as Feb. 26.
Court to hear women’s health cases
Two closely watched reproductive health cases will go before the court this spring.
On March 4, justices will hear oral arguments in June Medical Services v. Russo, regarding the constitutionality of a Louisiana law that requires physicians performing abortions to have admitting privileges at a nearby hospital. Doctors who perform abortions without admitting privileges at a hospital within 30 miles face fines and imprisonment, according to the state law, originally passed in 2014. Clinics that employ such doctors can also have their licenses revoked.
June Medical Services LLC, a women’s health clinic, sued over the law. A district court ruled in favor of the plaintiff, but the 5th U.S. Circuit Court of Appeals reversed and upheld Louisiana’s law. The clinic appealed to the U.S. Supreme Court. Louisiana officials argue the challenge should be dismissed, and the law allowed to proceed, because the plaintiffs lack standing.
The Supreme Court in 2016 heard a similar case – Whole Woman’s Health v. Hellerstedt – concerning a comparable law in Texas. In that case, justices struck down the measure as unconstitutional.
And on April 29, justices will hear arguments in Little Sisters of the Poor v. Pennsylvania, a consolidated case about whether the Trump administration acted properly when it expanded exemptions under the Affordable Care Act’s contraceptive mandate. Entities that object to providing contraception on the basis of religious beliefs can opt out of complying with the mandate, according to the 2018 regulations. Additionally, nonprofit organizations and small businesses that have nonreligious moral convictions against the mandate can skip compliance. A number of states and entities sued over the new rules.
A federal appeals court temporarily barred the regulations from moving forward, ruling the plaintiffs were likely to succeed in proving the Trump administration did not follow appropriate procedures when it promulgated the new rules and that the regulations were not authorized under the ACA.
Justices will decide whether the parties have standing in the case, whether the Trump administration followed correct rule-making procedures, and if the regulations can stand.
Initial FLAG-Ida outperforms 7+3 for high-risk AML
A treatment commonly used as a salvage regimen for relapsed/refractory acute myelogenous leukemia (AML) showed better results than did standard treatment when used as initial induction therapy.
Researchers found that the use of fludarabine, high-dose cytarabine, with granulocyte-colony stimulating factor (FLAG) – with or without idarubicin (Ida) – resulted in higher remission rates for nonfavorable risk AML patients after one course of induction, compared with standard 7+3 (anthracycline plus cytarabine) treatment.
The use of FLAG+/-Ida also resulted in a shorter time to complete remission (CR) and a shorter time to transplant, compared with 7+3. Additionally, postremission overall survival (OS) and disease-free survival (DFS) were better after achieving CR from FLAG-Ida, compared with 7+3, Melhem Solh, MD, of the Blood & Marrow Transplant Program at Northside Hospital, Atlanta, and colleagues reported in Leukemia Research.
The researchers retrospectively analyzed 304 consecutive AML patients, with nonfavorable National Comprehensive Cancer Network (NCCN) risk who received initial treatment at their center with either 7+3 (86 patients) or FLAG+/-Ida (218 patients). They found that patients in the FLAG+/-Ida group were more likely to achieve remission after one course of induction (74% vs. 62%; P less than .001) and had a faster time to achieve CR (30 days vs. 37.5 days; P less than .001), compared with patients receiving 7+3.
The time from diagnosis to allogeneic hematopoietic cell transplantation was shorter among CR patients after FLAG+/-Ida, compared with 7+3 (115 days vs. 151 days; P less than .003).
Additionally, the 3-year postremission OS and DFS were significantly better for patients receiving FLAG-Ida at 54% and 49%, compared with 39% and 32% for 7+3, respectively (P = .01).
Dr. Solh and colleagues found that the factors associated with postremission survival included age at first CR, NCCN risk, induction regimen (FLAG+/-Ida vs. 7+3; hazard ratio, 0.62; P = .01), and receipt of allogeneic hematopoietic cell transplantation.
“FLAG-Ida is a more efficacious regimen when used for initial induction compared to 3+7. It appears to produce better survival and improved postremission survival for AML patients with intermediate and poor risk AML without increasing toxicity,” Dr. Solh and colleagues wrote. “Further validation of these results in a well-designed randomized prospective trial will help define the best induction approaches for AML.”
There was no outside funding for this research and the authors reported that they had no relevant financial conflicts.
SOURCE: Solh MM et al. Leuk Res. 2020 Feb 14. doi: 10.1016/j.leukres.2020.106318.
A treatment commonly used as a salvage regimen for relapsed/refractory acute myelogenous leukemia (AML) showed better results than did standard treatment when used as initial induction therapy.
Researchers found that the use of fludarabine, high-dose cytarabine, with granulocyte-colony stimulating factor (FLAG) – with or without idarubicin (Ida) – resulted in higher remission rates for nonfavorable risk AML patients after one course of induction, compared with standard 7+3 (anthracycline plus cytarabine) treatment.
The use of FLAG+/-Ida also resulted in a shorter time to complete remission (CR) and a shorter time to transplant, compared with 7+3. Additionally, postremission overall survival (OS) and disease-free survival (DFS) were better after achieving CR from FLAG-Ida, compared with 7+3, Melhem Solh, MD, of the Blood & Marrow Transplant Program at Northside Hospital, Atlanta, and colleagues reported in Leukemia Research.
The researchers retrospectively analyzed 304 consecutive AML patients, with nonfavorable National Comprehensive Cancer Network (NCCN) risk who received initial treatment at their center with either 7+3 (86 patients) or FLAG+/-Ida (218 patients). They found that patients in the FLAG+/-Ida group were more likely to achieve remission after one course of induction (74% vs. 62%; P less than .001) and had a faster time to achieve CR (30 days vs. 37.5 days; P less than .001), compared with patients receiving 7+3.
The time from diagnosis to allogeneic hematopoietic cell transplantation was shorter among CR patients after FLAG+/-Ida, compared with 7+3 (115 days vs. 151 days; P less than .003).
Additionally, the 3-year postremission OS and DFS were significantly better for patients receiving FLAG-Ida at 54% and 49%, compared with 39% and 32% for 7+3, respectively (P = .01).
Dr. Solh and colleagues found that the factors associated with postremission survival included age at first CR, NCCN risk, induction regimen (FLAG+/-Ida vs. 7+3; hazard ratio, 0.62; P = .01), and receipt of allogeneic hematopoietic cell transplantation.
“FLAG-Ida is a more efficacious regimen when used for initial induction compared to 3+7. It appears to produce better survival and improved postremission survival for AML patients with intermediate and poor risk AML without increasing toxicity,” Dr. Solh and colleagues wrote. “Further validation of these results in a well-designed randomized prospective trial will help define the best induction approaches for AML.”
There was no outside funding for this research and the authors reported that they had no relevant financial conflicts.
SOURCE: Solh MM et al. Leuk Res. 2020 Feb 14. doi: 10.1016/j.leukres.2020.106318.
A treatment commonly used as a salvage regimen for relapsed/refractory acute myelogenous leukemia (AML) showed better results than did standard treatment when used as initial induction therapy.
Researchers found that the use of fludarabine, high-dose cytarabine, with granulocyte-colony stimulating factor (FLAG) – with or without idarubicin (Ida) – resulted in higher remission rates for nonfavorable risk AML patients after one course of induction, compared with standard 7+3 (anthracycline plus cytarabine) treatment.
The use of FLAG+/-Ida also resulted in a shorter time to complete remission (CR) and a shorter time to transplant, compared with 7+3. Additionally, postremission overall survival (OS) and disease-free survival (DFS) were better after achieving CR from FLAG-Ida, compared with 7+3, Melhem Solh, MD, of the Blood & Marrow Transplant Program at Northside Hospital, Atlanta, and colleagues reported in Leukemia Research.
The researchers retrospectively analyzed 304 consecutive AML patients, with nonfavorable National Comprehensive Cancer Network (NCCN) risk who received initial treatment at their center with either 7+3 (86 patients) or FLAG+/-Ida (218 patients). They found that patients in the FLAG+/-Ida group were more likely to achieve remission after one course of induction (74% vs. 62%; P less than .001) and had a faster time to achieve CR (30 days vs. 37.5 days; P less than .001), compared with patients receiving 7+3.
The time from diagnosis to allogeneic hematopoietic cell transplantation was shorter among CR patients after FLAG+/-Ida, compared with 7+3 (115 days vs. 151 days; P less than .003).
Additionally, the 3-year postremission OS and DFS were significantly better for patients receiving FLAG-Ida at 54% and 49%, compared with 39% and 32% for 7+3, respectively (P = .01).
Dr. Solh and colleagues found that the factors associated with postremission survival included age at first CR, NCCN risk, induction regimen (FLAG+/-Ida vs. 7+3; hazard ratio, 0.62; P = .01), and receipt of allogeneic hematopoietic cell transplantation.
“FLAG-Ida is a more efficacious regimen when used for initial induction compared to 3+7. It appears to produce better survival and improved postremission survival for AML patients with intermediate and poor risk AML without increasing toxicity,” Dr. Solh and colleagues wrote. “Further validation of these results in a well-designed randomized prospective trial will help define the best induction approaches for AML.”
There was no outside funding for this research and the authors reported that they had no relevant financial conflicts.
SOURCE: Solh MM et al. Leuk Res. 2020 Feb 14. doi: 10.1016/j.leukres.2020.106318.
FROM LEUKEMIA RESEARCH
Welcome Editorial
It’s an honor and a great pleasure to take on this new role as editor in chief for Hematology News. When I got the call from our outgoing editor Matt Kalaycio, MD, a year ago asking me to consider stepping into his shoes a few things flashed through my mind. Will I do this role justice? Do I have what it takes to be a great editor in chief?
Then I thought – will there be time to learn the ropes or will this be like most of my career positions where you jump into the water first and figure out how to swim later? I never once thought: “Oh no … I cannot do this and I’m going to say no!” So here I am today, reporting for duty as the editor in chief of Hematology News.
I was once accused of being “intellectually restless” which is a badge I wear with honor and is perhaps a trait I learned from my mom who was a public health nurse in Nigeria back in the seventies. She broke a lot of glass ceilings in her day – Cornell University–trained advanced practice nurse, mother of five girls, with a degree in textile and design and business accounting. She also got a certificate in baking and cake decorating and she used all her skills and certifications to raise her daughters to believe the sky was the limit.
Mom started one of the first licensed practical nursing (LPN) schools in Nigeria and I learned from her to never back down from a challenge – on a dare I got my LPN certification before I went to medical school.
You see I love a challenge and an adventure and serving as the editor in chief for Hematology News provides me with an amazing platform and opportunity to achieve a lot of firsts and satisfy that hunger to make a global difference that has always guided my career.
I’ve thought long and hard about what and how I envision this role shaping out. What do I want our readers to take away from this newspaper under my leadership? What common themes will be woven in every edition? I want our readers to be challenged and keep learning. Not just about hematologic disorders and the latest scientific breakthroughs that drive improved patient outcomes for blood disorders but also about the intersectionality between hematology and other life disciplines.
I remember taking an art class in high school learning about dimensions and proportions of buildings and I dreamt of becoming an architect. Fast forward 2 decades later, I attended a medical conference at Georgia Institute of Technology in Atlanta and was enthralled at how various sessions demonstrated how art, engineering and architecture played a role in the development and design of orthopedic prosthesis used in amputees. I learned how engineering shaped our understanding of microfluidics, something that is now being leveraged in drug delivery science and in the field of hematology.
I want our readers to keep learning not just from esteemed scientists and clinicians but from various stakeholders – the patient, the high school student, the spouse of the hematologist, not to mention our residents and fellows, who are the future of our discipline. Furthermore, I want our readers to see the human side of hematology – the face behind the scientist or clinician and the reality of what joys and tolls we experience in this field.
A Fall 2019 Medscape survey cited the prevalence of physician burnout among hematology oncology physicians: 32% of oncologists were burned out, 4% were depressed, and 9% were both burned out and depressed. These are statistics that cannot be ignored or minimized a they ultimately have a profound impact on patient outcomes. You see, I really believe that much of the success we have in healing our patients relies not just on the medications we prescribe or on the procedures we perform or the science we leverage. Much of healing in medicine and in hematology is based on the secret sauce of being humane – defined by Merriam-Webster dictionary as the character trait that is “marked by demonstrating compassion, sympathy, or consideration for humans or animals.”
So, to sum up what to expect in the coming year from your editor in chief? Look out for some thought-provoking, fun, and unusual perspectives that are aimed to keep us learning, growing, and remaining humane in our interactions with our patients, each other, but more importantly with ourselves. #bringit2020 #HematologyNews #NewEditorInChiefPerspectives.
Ifeyinwa (Ify) Osunkwo, MD, MPH, is a professor of medicine and the director of the Sickle Cell Disease Enterprise at the Levine Cancer Institute, Atrium Health, Charlotte, N.C. She is the editor in chief of Hematology News.
It’s an honor and a great pleasure to take on this new role as editor in chief for Hematology News. When I got the call from our outgoing editor Matt Kalaycio, MD, a year ago asking me to consider stepping into his shoes a few things flashed through my mind. Will I do this role justice? Do I have what it takes to be a great editor in chief?
Then I thought – will there be time to learn the ropes or will this be like most of my career positions where you jump into the water first and figure out how to swim later? I never once thought: “Oh no … I cannot do this and I’m going to say no!” So here I am today, reporting for duty as the editor in chief of Hematology News.
I was once accused of being “intellectually restless” which is a badge I wear with honor and is perhaps a trait I learned from my mom who was a public health nurse in Nigeria back in the seventies. She broke a lot of glass ceilings in her day – Cornell University–trained advanced practice nurse, mother of five girls, with a degree in textile and design and business accounting. She also got a certificate in baking and cake decorating and she used all her skills and certifications to raise her daughters to believe the sky was the limit.
Mom started one of the first licensed practical nursing (LPN) schools in Nigeria and I learned from her to never back down from a challenge – on a dare I got my LPN certification before I went to medical school.
You see I love a challenge and an adventure and serving as the editor in chief for Hematology News provides me with an amazing platform and opportunity to achieve a lot of firsts and satisfy that hunger to make a global difference that has always guided my career.
I’ve thought long and hard about what and how I envision this role shaping out. What do I want our readers to take away from this newspaper under my leadership? What common themes will be woven in every edition? I want our readers to be challenged and keep learning. Not just about hematologic disorders and the latest scientific breakthroughs that drive improved patient outcomes for blood disorders but also about the intersectionality between hematology and other life disciplines.
I remember taking an art class in high school learning about dimensions and proportions of buildings and I dreamt of becoming an architect. Fast forward 2 decades later, I attended a medical conference at Georgia Institute of Technology in Atlanta and was enthralled at how various sessions demonstrated how art, engineering and architecture played a role in the development and design of orthopedic prosthesis used in amputees. I learned how engineering shaped our understanding of microfluidics, something that is now being leveraged in drug delivery science and in the field of hematology.
I want our readers to keep learning not just from esteemed scientists and clinicians but from various stakeholders – the patient, the high school student, the spouse of the hematologist, not to mention our residents and fellows, who are the future of our discipline. Furthermore, I want our readers to see the human side of hematology – the face behind the scientist or clinician and the reality of what joys and tolls we experience in this field.
A Fall 2019 Medscape survey cited the prevalence of physician burnout among hematology oncology physicians: 32% of oncologists were burned out, 4% were depressed, and 9% were both burned out and depressed. These are statistics that cannot be ignored or minimized a they ultimately have a profound impact on patient outcomes. You see, I really believe that much of the success we have in healing our patients relies not just on the medications we prescribe or on the procedures we perform or the science we leverage. Much of healing in medicine and in hematology is based on the secret sauce of being humane – defined by Merriam-Webster dictionary as the character trait that is “marked by demonstrating compassion, sympathy, or consideration for humans or animals.”
So, to sum up what to expect in the coming year from your editor in chief? Look out for some thought-provoking, fun, and unusual perspectives that are aimed to keep us learning, growing, and remaining humane in our interactions with our patients, each other, but more importantly with ourselves. #bringit2020 #HematologyNews #NewEditorInChiefPerspectives.
Ifeyinwa (Ify) Osunkwo, MD, MPH, is a professor of medicine and the director of the Sickle Cell Disease Enterprise at the Levine Cancer Institute, Atrium Health, Charlotte, N.C. She is the editor in chief of Hematology News.
It’s an honor and a great pleasure to take on this new role as editor in chief for Hematology News. When I got the call from our outgoing editor Matt Kalaycio, MD, a year ago asking me to consider stepping into his shoes a few things flashed through my mind. Will I do this role justice? Do I have what it takes to be a great editor in chief?
Then I thought – will there be time to learn the ropes or will this be like most of my career positions where you jump into the water first and figure out how to swim later? I never once thought: “Oh no … I cannot do this and I’m going to say no!” So here I am today, reporting for duty as the editor in chief of Hematology News.
I was once accused of being “intellectually restless” which is a badge I wear with honor and is perhaps a trait I learned from my mom who was a public health nurse in Nigeria back in the seventies. She broke a lot of glass ceilings in her day – Cornell University–trained advanced practice nurse, mother of five girls, with a degree in textile and design and business accounting. She also got a certificate in baking and cake decorating and she used all her skills and certifications to raise her daughters to believe the sky was the limit.
Mom started one of the first licensed practical nursing (LPN) schools in Nigeria and I learned from her to never back down from a challenge – on a dare I got my LPN certification before I went to medical school.
You see I love a challenge and an adventure and serving as the editor in chief for Hematology News provides me with an amazing platform and opportunity to achieve a lot of firsts and satisfy that hunger to make a global difference that has always guided my career.
I’ve thought long and hard about what and how I envision this role shaping out. What do I want our readers to take away from this newspaper under my leadership? What common themes will be woven in every edition? I want our readers to be challenged and keep learning. Not just about hematologic disorders and the latest scientific breakthroughs that drive improved patient outcomes for blood disorders but also about the intersectionality between hematology and other life disciplines.
I remember taking an art class in high school learning about dimensions and proportions of buildings and I dreamt of becoming an architect. Fast forward 2 decades later, I attended a medical conference at Georgia Institute of Technology in Atlanta and was enthralled at how various sessions demonstrated how art, engineering and architecture played a role in the development and design of orthopedic prosthesis used in amputees. I learned how engineering shaped our understanding of microfluidics, something that is now being leveraged in drug delivery science and in the field of hematology.
I want our readers to keep learning not just from esteemed scientists and clinicians but from various stakeholders – the patient, the high school student, the spouse of the hematologist, not to mention our residents and fellows, who are the future of our discipline. Furthermore, I want our readers to see the human side of hematology – the face behind the scientist or clinician and the reality of what joys and tolls we experience in this field.
A Fall 2019 Medscape survey cited the prevalence of physician burnout among hematology oncology physicians: 32% of oncologists were burned out, 4% were depressed, and 9% were both burned out and depressed. These are statistics that cannot be ignored or minimized a they ultimately have a profound impact on patient outcomes. You see, I really believe that much of the success we have in healing our patients relies not just on the medications we prescribe or on the procedures we perform or the science we leverage. Much of healing in medicine and in hematology is based on the secret sauce of being humane – defined by Merriam-Webster dictionary as the character trait that is “marked by demonstrating compassion, sympathy, or consideration for humans or animals.”
So, to sum up what to expect in the coming year from your editor in chief? Look out for some thought-provoking, fun, and unusual perspectives that are aimed to keep us learning, growing, and remaining humane in our interactions with our patients, each other, but more importantly with ourselves. #bringit2020 #HematologyNews #NewEditorInChiefPerspectives.
Ifeyinwa (Ify) Osunkwo, MD, MPH, is a professor of medicine and the director of the Sickle Cell Disease Enterprise at the Levine Cancer Institute, Atrium Health, Charlotte, N.C. She is the editor in chief of Hematology News.
Late effects in young cancer survivors underscore importance of high-risk screening
according to data from the Childhood Cancer Survivor Study.
At a median follow-up of 21 years, the SMR for all-cause mortality was 5.9 among survivors aged 15-20 years and 6.2 among diagnosis-matched children under 15 years, compared with expected rates at the same ages in the general population. For health-related causes – excluding primary cancer recurrence or progression but including late effects of cancer therapy – the SMRs were 4.8 in the older group and 6.8 in the younger group.
Eugene Suh, MD, of Loyola University Chicago Medical Center, Maywood, Ill., and colleagues reported these results in Lancet Oncology.
The difference between the older and younger survivors (n = 5,804 in each group) was most evident at least 20 years after cancer diagnosis, the authors noted.
For both groups, but more so for childhood cancer survivors, the risk of developing any chronic health condition and any grade 3-5 health condition was greater than for siblings of the same age who did not have cancer (hazard ratios, 4.2 for adolescents/young adults and 5.6 for childhood survivors). The same was true for grade 3-5 cardiac conditions (HRs, 4.3 and 5.6, respectively), endocrine conditions (HRs, 3.9 and 6.4, respectively), and musculoskeletal conditions (HRs, 6.5 and 8.0, respectively).
These findings, which confirm those of previous studies suggesting that younger children might be more vulnerable to the adverse effects of cancer treatment, “underscore that focused efforts are needed to ensure early-adolescent and young adult cancer survivors are receiving recommended risk-based care, with a focus on high-risk cancer screening, to reduce morbidity and premature mortality,” the researchers concluded, noting that “studies to date indicate that adherence to such high-risk screening is poor.”
In a related editorial, Päivi Lähteenmäki, MD, PhD, of University of Turku (Finland) and Turku University Hospital, wrote that these findings warrant long-term follow-up of adolescent and young adult cancer survivors. She also argued that the results “might not be fully generalizable to patients treated today who might be on different treatment regimens to those treated in previous decades” and that “[m]ore prospectively collected objective data focusing on survivors ... are needed.”
Accurate characterization of patients at high risk who would benefit from a tailored screening program is most important, and identifying underlying genetic or molecular factors that confer higher risk for late sequelae would be useful for “planning approaches to survivorship,” Dr. Lähteenmäki added.
This study was funded by the National Cancer Institute and American Lebanese-Syrian Associated Charities. Dr. Suh and Dr. Lähteenmäki reported having no competing interests.
SOURCES: Suh E et al. Lancet Oncology. 2020 Feb 14. doi: 10.1016/S1470-2045(19)30800-9;Lähteenmäki P. Lancet Oncol. 2020 Feb 14. doi: 10.106/S1470-2045(19)30858-7.
according to data from the Childhood Cancer Survivor Study.
At a median follow-up of 21 years, the SMR for all-cause mortality was 5.9 among survivors aged 15-20 years and 6.2 among diagnosis-matched children under 15 years, compared with expected rates at the same ages in the general population. For health-related causes – excluding primary cancer recurrence or progression but including late effects of cancer therapy – the SMRs were 4.8 in the older group and 6.8 in the younger group.
Eugene Suh, MD, of Loyola University Chicago Medical Center, Maywood, Ill., and colleagues reported these results in Lancet Oncology.
The difference between the older and younger survivors (n = 5,804 in each group) was most evident at least 20 years after cancer diagnosis, the authors noted.
For both groups, but more so for childhood cancer survivors, the risk of developing any chronic health condition and any grade 3-5 health condition was greater than for siblings of the same age who did not have cancer (hazard ratios, 4.2 for adolescents/young adults and 5.6 for childhood survivors). The same was true for grade 3-5 cardiac conditions (HRs, 4.3 and 5.6, respectively), endocrine conditions (HRs, 3.9 and 6.4, respectively), and musculoskeletal conditions (HRs, 6.5 and 8.0, respectively).
These findings, which confirm those of previous studies suggesting that younger children might be more vulnerable to the adverse effects of cancer treatment, “underscore that focused efforts are needed to ensure early-adolescent and young adult cancer survivors are receiving recommended risk-based care, with a focus on high-risk cancer screening, to reduce morbidity and premature mortality,” the researchers concluded, noting that “studies to date indicate that adherence to such high-risk screening is poor.”
In a related editorial, Päivi Lähteenmäki, MD, PhD, of University of Turku (Finland) and Turku University Hospital, wrote that these findings warrant long-term follow-up of adolescent and young adult cancer survivors. She also argued that the results “might not be fully generalizable to patients treated today who might be on different treatment regimens to those treated in previous decades” and that “[m]ore prospectively collected objective data focusing on survivors ... are needed.”
Accurate characterization of patients at high risk who would benefit from a tailored screening program is most important, and identifying underlying genetic or molecular factors that confer higher risk for late sequelae would be useful for “planning approaches to survivorship,” Dr. Lähteenmäki added.
This study was funded by the National Cancer Institute and American Lebanese-Syrian Associated Charities. Dr. Suh and Dr. Lähteenmäki reported having no competing interests.
SOURCES: Suh E et al. Lancet Oncology. 2020 Feb 14. doi: 10.1016/S1470-2045(19)30800-9;Lähteenmäki P. Lancet Oncol. 2020 Feb 14. doi: 10.106/S1470-2045(19)30858-7.
according to data from the Childhood Cancer Survivor Study.
At a median follow-up of 21 years, the SMR for all-cause mortality was 5.9 among survivors aged 15-20 years and 6.2 among diagnosis-matched children under 15 years, compared with expected rates at the same ages in the general population. For health-related causes – excluding primary cancer recurrence or progression but including late effects of cancer therapy – the SMRs were 4.8 in the older group and 6.8 in the younger group.
Eugene Suh, MD, of Loyola University Chicago Medical Center, Maywood, Ill., and colleagues reported these results in Lancet Oncology.
The difference between the older and younger survivors (n = 5,804 in each group) was most evident at least 20 years after cancer diagnosis, the authors noted.
For both groups, but more so for childhood cancer survivors, the risk of developing any chronic health condition and any grade 3-5 health condition was greater than for siblings of the same age who did not have cancer (hazard ratios, 4.2 for adolescents/young adults and 5.6 for childhood survivors). The same was true for grade 3-5 cardiac conditions (HRs, 4.3 and 5.6, respectively), endocrine conditions (HRs, 3.9 and 6.4, respectively), and musculoskeletal conditions (HRs, 6.5 and 8.0, respectively).
These findings, which confirm those of previous studies suggesting that younger children might be more vulnerable to the adverse effects of cancer treatment, “underscore that focused efforts are needed to ensure early-adolescent and young adult cancer survivors are receiving recommended risk-based care, with a focus on high-risk cancer screening, to reduce morbidity and premature mortality,” the researchers concluded, noting that “studies to date indicate that adherence to such high-risk screening is poor.”
In a related editorial, Päivi Lähteenmäki, MD, PhD, of University of Turku (Finland) and Turku University Hospital, wrote that these findings warrant long-term follow-up of adolescent and young adult cancer survivors. She also argued that the results “might not be fully generalizable to patients treated today who might be on different treatment regimens to those treated in previous decades” and that “[m]ore prospectively collected objective data focusing on survivors ... are needed.”
Accurate characterization of patients at high risk who would benefit from a tailored screening program is most important, and identifying underlying genetic or molecular factors that confer higher risk for late sequelae would be useful for “planning approaches to survivorship,” Dr. Lähteenmäki added.
This study was funded by the National Cancer Institute and American Lebanese-Syrian Associated Charities. Dr. Suh and Dr. Lähteenmäki reported having no competing interests.
SOURCES: Suh E et al. Lancet Oncology. 2020 Feb 14. doi: 10.1016/S1470-2045(19)30800-9;Lähteenmäki P. Lancet Oncol. 2020 Feb 14. doi: 10.106/S1470-2045(19)30858-7.
FROM LANCET ONCOLOGY
Adolescent obesity linked with midlife cancer risk
Obesity during adolescence is associated with increased midlife cancer risk, according to findings from a large population-based cohort of Israeli teens examined between 1967 and 2010.
The association, which was stronger in individuals in the later period of the cohort than in those in the earlier years, suggests that the burden of obesity-related cancers might increase over time, given the increasing prevalence of adolescent obesity, wrote Ariel Furer, MD, of Israel Defense Forces Medical Corps, Ramat Gan, and colleagues. Their report is in The Lancet.
Obesity is a known causal factor for several types of cancer, but most studies have looked at middle-age or older individuals and had relatively short follow-up, and period effects are rarely assessed, the investigators said, noting that “the attributable burden of obesity-related cancer was previously calculated with an unverified assumption that the association remained unchanged over time.
“In contrast to this paucity of knowledge, the prevalence of youth obesity – particularly severe obesity – has increased worldwide, which parallels the rise in youth cancer incidence,” they wrote.
To address this paucity of data, the researchers reviewed medical and sociodemographic data for adolescents who were assessed at age 17 years for medical eligibility for mandatory military service, and linked that information with data from the National Cancer Registry to create a unified file. The primary study outcome was any cancer diagnosis between Jan. 1, 1967, and Dec. 31, 2012, and a secondary endpoint was all-cause mortality through Dec. 31, 2017, among those who developed cancer.
Among nearly 2.3 million participating adolescents who were evaluated for associations between body mass index at age 17 years and later cancer incidence, 1,370,020 were men with more than 29.5 million person-years of follow-up, and 928,110 were women with more than 18 million person-years of follow-up. The numbers of incident cancer cases in the men and women were 26,353 and 29,488, and the mean ages at diagnosis were 43.2 and 40.0 years, respectively, the investigators reported (Lancet. 2020 Feb 3. doi: 10.1016/S2213-8587(20)30019-X).
Adolescent obesity in men was significantly associated with midlife cancer incidence (hazard ratio, 1.26), but in women, no association was seen due to the previously reported inverse associations between obesity and cervical and breast cancers, they said.
However, when those cancers were excluded for women, the adjusted hazard ratio was similar to that for men (HR, 1.27).
Cancer incidence in both men and women increased gradually across BMI percentiles, and for both sexes, overweight BMI was associated with an increased cancer risk after 10 years of follow-up (HR, 1.14 for men, 1.22 for women after exclusion of cervical and breast cancer). Therefore, in some cases the increased cancer risk in those who were overweight as teens was evident before age 30 years, the authors noted.
Further, BMI was positively associated with greater mortality risk. For men, 5-year survival rates were 75.2% in those with adolescent BMI in the 5th-49th percentile, compared with 72.2% in those with BMI in the obesity range (95th percentile or greater), and the corresponding rates in women were 89.3% and 83.1% (HR, 1.33 and 1.89, respectively).
Of note, the investigators identified a period effect. That is, after stratification by enrollment period/cancer recording period (1967-1981/1982-1996 vs. 1982-1996/1997-2011), a stronger association was noted in individuals who entered the study during the later period, compared with those who entered in the early period (HR, 1.36 vs. 1.13; adjusted HR, 1.11 vs. 1.07 per 5 kg/m2). Possible mechanisms for this finding include environmental and nutritional factors, increased use of medical services, and changes in early cancer screening techniques, but further study is needed to verify the trend and “refine the exact nature of carcinogenic elements, compared with earlier periods,” they said.
Also of note, some cancers that were not associated with BMI in the early period, including stomach cancer, non-Hodgkin lymphoma, thyroid cancer, and colorectal and oral cavity cancers, became significantly associated with BMI in the late period.
“The projected population attributable risk percentage, using 2017 prevalence data of high BMI, was 5.1% for any cancer in men and 5.7% for cancers other than breast and cervical in women,” the researchers wrote, noting that this “is probably an underestimation, given the accentuation of the BMI-cancer association and the rapid increase in adolescent obesity prevalence within the past decade in Israel and worldwide.”
In an accompanying editorial, the journal editors noted that the findings by Dr. Furer and colleagues highlight the need to tackle obesity early in life and the need for obesity prevention strategies to reduce cancer incidence and mortality for those cancers that can be prevented by lifestyle modifications. They added, however, that care would be needed to avoid stigmatizing those with obesity, as obesity itself is a “multifactorial condition driven by social injustice and health inequalities” that most often affect those who are least able to implement lifestyle change (Lancet. 2020 Feb 3. doi: 10.106/S2213-8587(20)30031-0).
They also emphasized that the links between obesity and cancer, like those between obesity and other diseases such as diabetes, underscore the fact that noncommunicable diseases do not exist in isolation, and that tackling them requires bold action, a consolidated approach, and elimination of the environmental and social factors driving the epidemic.
The study was limited by a number of factors, including the lack of data on lifestyle factors, underrepresentation of some ethnicities, and lack of data on BMI and medical comorbidities at the time of cancer diagnosis. However, strengths of the study include the systematic data collection, narrow range of age at study entry, strict control of coexisting conditions, and high statistical power, which strengthen the generalizability of the results, the investigators said, concluding, therefore, that “[c]urrent trends of rising BMI among adolescents could constitute an important intervention target for cancer prevention.”
The authors reported having no disclosures.
SOURCE: Furer A et al. Lancet Diabetes Endocrinol. 2020 Feb 3. doi: 10.1016/S2213-8587(20)30019-X.
Obesity during adolescence is associated with increased midlife cancer risk, according to findings from a large population-based cohort of Israeli teens examined between 1967 and 2010.
The association, which was stronger in individuals in the later period of the cohort than in those in the earlier years, suggests that the burden of obesity-related cancers might increase over time, given the increasing prevalence of adolescent obesity, wrote Ariel Furer, MD, of Israel Defense Forces Medical Corps, Ramat Gan, and colleagues. Their report is in The Lancet.
Obesity is a known causal factor for several types of cancer, but most studies have looked at middle-age or older individuals and had relatively short follow-up, and period effects are rarely assessed, the investigators said, noting that “the attributable burden of obesity-related cancer was previously calculated with an unverified assumption that the association remained unchanged over time.
“In contrast to this paucity of knowledge, the prevalence of youth obesity – particularly severe obesity – has increased worldwide, which parallels the rise in youth cancer incidence,” they wrote.
To address this paucity of data, the researchers reviewed medical and sociodemographic data for adolescents who were assessed at age 17 years for medical eligibility for mandatory military service, and linked that information with data from the National Cancer Registry to create a unified file. The primary study outcome was any cancer diagnosis between Jan. 1, 1967, and Dec. 31, 2012, and a secondary endpoint was all-cause mortality through Dec. 31, 2017, among those who developed cancer.
Among nearly 2.3 million participating adolescents who were evaluated for associations between body mass index at age 17 years and later cancer incidence, 1,370,020 were men with more than 29.5 million person-years of follow-up, and 928,110 were women with more than 18 million person-years of follow-up. The numbers of incident cancer cases in the men and women were 26,353 and 29,488, and the mean ages at diagnosis were 43.2 and 40.0 years, respectively, the investigators reported (Lancet. 2020 Feb 3. doi: 10.1016/S2213-8587(20)30019-X).
Adolescent obesity in men was significantly associated with midlife cancer incidence (hazard ratio, 1.26), but in women, no association was seen due to the previously reported inverse associations between obesity and cervical and breast cancers, they said.
However, when those cancers were excluded for women, the adjusted hazard ratio was similar to that for men (HR, 1.27).
Cancer incidence in both men and women increased gradually across BMI percentiles, and for both sexes, overweight BMI was associated with an increased cancer risk after 10 years of follow-up (HR, 1.14 for men, 1.22 for women after exclusion of cervical and breast cancer). Therefore, in some cases the increased cancer risk in those who were overweight as teens was evident before age 30 years, the authors noted.
Further, BMI was positively associated with greater mortality risk. For men, 5-year survival rates were 75.2% in those with adolescent BMI in the 5th-49th percentile, compared with 72.2% in those with BMI in the obesity range (95th percentile or greater), and the corresponding rates in women were 89.3% and 83.1% (HR, 1.33 and 1.89, respectively).
Of note, the investigators identified a period effect. That is, after stratification by enrollment period/cancer recording period (1967-1981/1982-1996 vs. 1982-1996/1997-2011), a stronger association was noted in individuals who entered the study during the later period, compared with those who entered in the early period (HR, 1.36 vs. 1.13; adjusted HR, 1.11 vs. 1.07 per 5 kg/m2). Possible mechanisms for this finding include environmental and nutritional factors, increased use of medical services, and changes in early cancer screening techniques, but further study is needed to verify the trend and “refine the exact nature of carcinogenic elements, compared with earlier periods,” they said.
Also of note, some cancers that were not associated with BMI in the early period, including stomach cancer, non-Hodgkin lymphoma, thyroid cancer, and colorectal and oral cavity cancers, became significantly associated with BMI in the late period.
“The projected population attributable risk percentage, using 2017 prevalence data of high BMI, was 5.1% for any cancer in men and 5.7% for cancers other than breast and cervical in women,” the researchers wrote, noting that this “is probably an underestimation, given the accentuation of the BMI-cancer association and the rapid increase in adolescent obesity prevalence within the past decade in Israel and worldwide.”
In an accompanying editorial, the journal editors noted that the findings by Dr. Furer and colleagues highlight the need to tackle obesity early in life and the need for obesity prevention strategies to reduce cancer incidence and mortality for those cancers that can be prevented by lifestyle modifications. They added, however, that care would be needed to avoid stigmatizing those with obesity, as obesity itself is a “multifactorial condition driven by social injustice and health inequalities” that most often affect those who are least able to implement lifestyle change (Lancet. 2020 Feb 3. doi: 10.106/S2213-8587(20)30031-0).
They also emphasized that the links between obesity and cancer, like those between obesity and other diseases such as diabetes, underscore the fact that noncommunicable diseases do not exist in isolation, and that tackling them requires bold action, a consolidated approach, and elimination of the environmental and social factors driving the epidemic.
The study was limited by a number of factors, including the lack of data on lifestyle factors, underrepresentation of some ethnicities, and lack of data on BMI and medical comorbidities at the time of cancer diagnosis. However, strengths of the study include the systematic data collection, narrow range of age at study entry, strict control of coexisting conditions, and high statistical power, which strengthen the generalizability of the results, the investigators said, concluding, therefore, that “[c]urrent trends of rising BMI among adolescents could constitute an important intervention target for cancer prevention.”
The authors reported having no disclosures.
SOURCE: Furer A et al. Lancet Diabetes Endocrinol. 2020 Feb 3. doi: 10.1016/S2213-8587(20)30019-X.
Obesity during adolescence is associated with increased midlife cancer risk, according to findings from a large population-based cohort of Israeli teens examined between 1967 and 2010.
The association, which was stronger in individuals in the later period of the cohort than in those in the earlier years, suggests that the burden of obesity-related cancers might increase over time, given the increasing prevalence of adolescent obesity, wrote Ariel Furer, MD, of Israel Defense Forces Medical Corps, Ramat Gan, and colleagues. Their report is in The Lancet.
Obesity is a known causal factor for several types of cancer, but most studies have looked at middle-age or older individuals and had relatively short follow-up, and period effects are rarely assessed, the investigators said, noting that “the attributable burden of obesity-related cancer was previously calculated with an unverified assumption that the association remained unchanged over time.
“In contrast to this paucity of knowledge, the prevalence of youth obesity – particularly severe obesity – has increased worldwide, which parallels the rise in youth cancer incidence,” they wrote.
To address this paucity of data, the researchers reviewed medical and sociodemographic data for adolescents who were assessed at age 17 years for medical eligibility for mandatory military service, and linked that information with data from the National Cancer Registry to create a unified file. The primary study outcome was any cancer diagnosis between Jan. 1, 1967, and Dec. 31, 2012, and a secondary endpoint was all-cause mortality through Dec. 31, 2017, among those who developed cancer.
Among nearly 2.3 million participating adolescents who were evaluated for associations between body mass index at age 17 years and later cancer incidence, 1,370,020 were men with more than 29.5 million person-years of follow-up, and 928,110 were women with more than 18 million person-years of follow-up. The numbers of incident cancer cases in the men and women were 26,353 and 29,488, and the mean ages at diagnosis were 43.2 and 40.0 years, respectively, the investigators reported (Lancet. 2020 Feb 3. doi: 10.1016/S2213-8587(20)30019-X).
Adolescent obesity in men was significantly associated with midlife cancer incidence (hazard ratio, 1.26), but in women, no association was seen due to the previously reported inverse associations between obesity and cervical and breast cancers, they said.
However, when those cancers were excluded for women, the adjusted hazard ratio was similar to that for men (HR, 1.27).
Cancer incidence in both men and women increased gradually across BMI percentiles, and for both sexes, overweight BMI was associated with an increased cancer risk after 10 years of follow-up (HR, 1.14 for men, 1.22 for women after exclusion of cervical and breast cancer). Therefore, in some cases the increased cancer risk in those who were overweight as teens was evident before age 30 years, the authors noted.
Further, BMI was positively associated with greater mortality risk. For men, 5-year survival rates were 75.2% in those with adolescent BMI in the 5th-49th percentile, compared with 72.2% in those with BMI in the obesity range (95th percentile or greater), and the corresponding rates in women were 89.3% and 83.1% (HR, 1.33 and 1.89, respectively).
Of note, the investigators identified a period effect. That is, after stratification by enrollment period/cancer recording period (1967-1981/1982-1996 vs. 1982-1996/1997-2011), a stronger association was noted in individuals who entered the study during the later period, compared with those who entered in the early period (HR, 1.36 vs. 1.13; adjusted HR, 1.11 vs. 1.07 per 5 kg/m2). Possible mechanisms for this finding include environmental and nutritional factors, increased use of medical services, and changes in early cancer screening techniques, but further study is needed to verify the trend and “refine the exact nature of carcinogenic elements, compared with earlier periods,” they said.
Also of note, some cancers that were not associated with BMI in the early period, including stomach cancer, non-Hodgkin lymphoma, thyroid cancer, and colorectal and oral cavity cancers, became significantly associated with BMI in the late period.
“The projected population attributable risk percentage, using 2017 prevalence data of high BMI, was 5.1% for any cancer in men and 5.7% for cancers other than breast and cervical in women,” the researchers wrote, noting that this “is probably an underestimation, given the accentuation of the BMI-cancer association and the rapid increase in adolescent obesity prevalence within the past decade in Israel and worldwide.”
In an accompanying editorial, the journal editors noted that the findings by Dr. Furer and colleagues highlight the need to tackle obesity early in life and the need for obesity prevention strategies to reduce cancer incidence and mortality for those cancers that can be prevented by lifestyle modifications. They added, however, that care would be needed to avoid stigmatizing those with obesity, as obesity itself is a “multifactorial condition driven by social injustice and health inequalities” that most often affect those who are least able to implement lifestyle change (Lancet. 2020 Feb 3. doi: 10.106/S2213-8587(20)30031-0).
They also emphasized that the links between obesity and cancer, like those between obesity and other diseases such as diabetes, underscore the fact that noncommunicable diseases do not exist in isolation, and that tackling them requires bold action, a consolidated approach, and elimination of the environmental and social factors driving the epidemic.
The study was limited by a number of factors, including the lack of data on lifestyle factors, underrepresentation of some ethnicities, and lack of data on BMI and medical comorbidities at the time of cancer diagnosis. However, strengths of the study include the systematic data collection, narrow range of age at study entry, strict control of coexisting conditions, and high statistical power, which strengthen the generalizability of the results, the investigators said, concluding, therefore, that “[c]urrent trends of rising BMI among adolescents could constitute an important intervention target for cancer prevention.”
The authors reported having no disclosures.
SOURCE: Furer A et al. Lancet Diabetes Endocrinol. 2020 Feb 3. doi: 10.1016/S2213-8587(20)30019-X.
FROM THE LANCET
‘Momentous’ USMLE change: New pass/fail format stuns medicine
News that the United States Medical Licensing Examination (USMLE) program will change its Step 1 scoring from a 3-digit number to pass/fail starting Jan. 1, 2022, has set off a flurry of shocked responses from students and physicians.
J. Bryan Carmody, MD, MPH, an assistant professor at Eastern Virginia Medical School in Norfolk, said in an interview that he was “stunned” when he heard the news on Wednesday and said the switch presents “the single biggest opportunity for medical school education reform since the Flexner Report,” which in 1910 established standards for modern medical education.
Numbers will continue for some tests
The USMLE cosponsors – the Federation of State Medical Boards (FSMB) and the National Board of Medical Examiners (NBME) – said that the Step 2 Clinical Knowledge (CK) exam and Step 3 will continue to be scored numerically. Step 2 Clinical Skills (CS) will continue its pass/fail system.
The change was made after Step 1 had been roundly criticized as playing too big a role in the process of becoming a physician and for causing students to study for the test instead of engaging fully in their medical education.
Ramie Fathy, a third-year medical student at the University of Pennsylvania, Philadelphia, currently studying for Step 1, said in an interview that it would have been nice personally to have the pass/fail choice, but he predicts both good and unintended consequences in the change.
The positive news, Mr. Fathy said, is that less emphasis will be put on the Step 1 test, which includes memorizing basic science details that may or not be relevant depending on later specialty choice.
“It’s not necessarily measuring what the test makers intended, which was whether or not a student can understand and apply basic science concepts to the practice of medicine,” he said.
“The current system encourages students to get as high a score as possible, which – after a certain point – translates to memorizing many little details that become increasingly less practically relevant,” Mr. Fathy said.
Pressure may move elsewhere?
However, Mr. Fathy worries that, without a scoring system to help decide who stands out in Step 1, residency program directors will depend more on the reputation of candidates’ medical school and the clout of the person writing a letter of recommendation – factors that are often influenced by family resources and social standing. That could wedge a further economic divide into the path to becoming a physician.
Mr. Fathy said he and fellow students are watching for information on what the passing bar will be and what happens with Step 2 Clinical Knowledge exam. USMLE has promised more information as soon as it is available.
“The question is whether that test will replace Step 1 as the standardized metric of student competency,” Mr. Fathy said, which would put more pressure on students further down the medical path.
Will Step 2 anxiety increase?
Dr. Carmody agreed that there is the danger that students now will spend their time studying for Step 2 CK at the expense of other parts of their education.
Meaningful reform will depend on the pass/fail move being coupled with other reforms, most importantly application caps, said Dr. Carmody, who teaches preclinical medical students and works with the residency program.
He has been blogging about Step 1 pass/fail for the past year.
Currently students can apply for as many residencies as they can pay for and Carmody said the number of applications per student has been rising over the past decade.
“That puts program directors under an impossible burden,” he said. “With our Step 1-based system, there’s significant inequality in the number of interviews people get. Programs end up overinviting the same group of people who look good on paper.”
People outside that group respond by sending more applications than they need to just to get a few interviews, Dr. Carmody added.
With caps, students would have an incentive to apply to only those programs in which they had a sincere interest, he said. Program directors also would then be better able to evaluate each application.
Switching Step 1 to pass/fail may have some effect on medical school burnout, Dr. Carmody said.
“It’s one thing to work hard when you’re on call and your patients depend on it,” he said. “But I would have a hard time staying up late every night studying something that I know in my heart is not going to help my patients, but I have to do it because I have to do better than the person who’s studying in the apartment next to me.”
Test has strayed from original purpose
Joseph Safdieh, MD, an assistant dean for clinical curriculum and director of the medical student neurology clerkship for the Weill Cornell Medicine, New York, sees the move as positive overall.
“We should not be using any single metric to define or describe our students’ overall profile,” he said in an interview.
“This has been a very significant anxiety point for our medical students for quite a number of years,” Dr. Safdieh said. “They were frustrated that their entire 4 years of medical school seemingly came down to one number.”
The test was created originally as one of three parts of licensure, he pointed out.
“Over the past 10 or 15 years, the exam has morphed to become a litmus test for very specific residency programs,” he said.
However, Dr. Safdieh has concerns that Step 2 will cultivate the same anxiety and may get too big a spotlight without the Step 1 metric, “although one could argue that test does more accurately reflect clinical material,” he said.
He also worries that students who have selected a specialty by the time they take Step 2 may find late in the game that they are less competitive in their field than they thought they were and may have to make a last-minute switch.
Dr. Safdieh said he thinks Step 2 will be next to go the pass/fail route. In reading between the lines of the announcement, he believes the test cosponsors didn’t make both pass/fail at once because it would have been “a nuclear bomb to the system.”
He credited the cosponsors with making what he called a “bold and momentous decision to initiate radical change in the overall transition between undergraduate and graduate medical education.”
Dr. Safdieh added that few in medicine were expecting Wednesday’s announcement.
“I think many of us were expecting them to go to quartile grading, not to go this far,” he said.
Dr. Safdieh suggested that, among those who may see downstream effects from the pass/fail move are offshore schools, such as those in the Caribbean. “Those schools rely on Step 1 to demonstrate that their students are meeting the rigor,” he said. But he hopes that this will lead to more holistic review.
“We’re hoping that this will force change in the system so that residency directors will look at more than just test-taking ability. They’ll look at publications and scholarship, community service and advocacy and performance in medical school,” Dr. Safdieh said.
Alison J. Whelan, MD, chief medical education officer of the Association of American Medical Colleges said in a statement, “The transition from medical school to residency training is a matter of great concern throughout academic medicine.
“The decision by the NBME and FSMB to change USMLE Step 1 score reporting to pass/fail was very carefully considered to balance student learning and student well-being,” she said. “The medical education community must now work together to identify and implement additional changes to improve the overall UME-GME [undergraduate and graduate medical education] transition system for all stakeholders and the AAMC is committed to helping lead this work.”
Dr. Fathy, Dr. Carmody, and Dr. Safdieh have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
News that the United States Medical Licensing Examination (USMLE) program will change its Step 1 scoring from a 3-digit number to pass/fail starting Jan. 1, 2022, has set off a flurry of shocked responses from students and physicians.
J. Bryan Carmody, MD, MPH, an assistant professor at Eastern Virginia Medical School in Norfolk, said in an interview that he was “stunned” when he heard the news on Wednesday and said the switch presents “the single biggest opportunity for medical school education reform since the Flexner Report,” which in 1910 established standards for modern medical education.
Numbers will continue for some tests
The USMLE cosponsors – the Federation of State Medical Boards (FSMB) and the National Board of Medical Examiners (NBME) – said that the Step 2 Clinical Knowledge (CK) exam and Step 3 will continue to be scored numerically. Step 2 Clinical Skills (CS) will continue its pass/fail system.
The change was made after Step 1 had been roundly criticized as playing too big a role in the process of becoming a physician and for causing students to study for the test instead of engaging fully in their medical education.
Ramie Fathy, a third-year medical student at the University of Pennsylvania, Philadelphia, currently studying for Step 1, said in an interview that it would have been nice personally to have the pass/fail choice, but he predicts both good and unintended consequences in the change.
The positive news, Mr. Fathy said, is that less emphasis will be put on the Step 1 test, which includes memorizing basic science details that may or not be relevant depending on later specialty choice.
“It’s not necessarily measuring what the test makers intended, which was whether or not a student can understand and apply basic science concepts to the practice of medicine,” he said.
“The current system encourages students to get as high a score as possible, which – after a certain point – translates to memorizing many little details that become increasingly less practically relevant,” Mr. Fathy said.
Pressure may move elsewhere?
However, Mr. Fathy worries that, without a scoring system to help decide who stands out in Step 1, residency program directors will depend more on the reputation of candidates’ medical school and the clout of the person writing a letter of recommendation – factors that are often influenced by family resources and social standing. That could wedge a further economic divide into the path to becoming a physician.
Mr. Fathy said he and fellow students are watching for information on what the passing bar will be and what happens with Step 2 Clinical Knowledge exam. USMLE has promised more information as soon as it is available.
“The question is whether that test will replace Step 1 as the standardized metric of student competency,” Mr. Fathy said, which would put more pressure on students further down the medical path.
Will Step 2 anxiety increase?
Dr. Carmody agreed that there is the danger that students now will spend their time studying for Step 2 CK at the expense of other parts of their education.
Meaningful reform will depend on the pass/fail move being coupled with other reforms, most importantly application caps, said Dr. Carmody, who teaches preclinical medical students and works with the residency program.
He has been blogging about Step 1 pass/fail for the past year.
Currently students can apply for as many residencies as they can pay for and Carmody said the number of applications per student has been rising over the past decade.
“That puts program directors under an impossible burden,” he said. “With our Step 1-based system, there’s significant inequality in the number of interviews people get. Programs end up overinviting the same group of people who look good on paper.”
People outside that group respond by sending more applications than they need to just to get a few interviews, Dr. Carmody added.
With caps, students would have an incentive to apply to only those programs in which they had a sincere interest, he said. Program directors also would then be better able to evaluate each application.
Switching Step 1 to pass/fail may have some effect on medical school burnout, Dr. Carmody said.
“It’s one thing to work hard when you’re on call and your patients depend on it,” he said. “But I would have a hard time staying up late every night studying something that I know in my heart is not going to help my patients, but I have to do it because I have to do better than the person who’s studying in the apartment next to me.”
Test has strayed from original purpose
Joseph Safdieh, MD, an assistant dean for clinical curriculum and director of the medical student neurology clerkship for the Weill Cornell Medicine, New York, sees the move as positive overall.
“We should not be using any single metric to define or describe our students’ overall profile,” he said in an interview.
“This has been a very significant anxiety point for our medical students for quite a number of years,” Dr. Safdieh said. “They were frustrated that their entire 4 years of medical school seemingly came down to one number.”
The test was created originally as one of three parts of licensure, he pointed out.
“Over the past 10 or 15 years, the exam has morphed to become a litmus test for very specific residency programs,” he said.
However, Dr. Safdieh has concerns that Step 2 will cultivate the same anxiety and may get too big a spotlight without the Step 1 metric, “although one could argue that test does more accurately reflect clinical material,” he said.
He also worries that students who have selected a specialty by the time they take Step 2 may find late in the game that they are less competitive in their field than they thought they were and may have to make a last-minute switch.
Dr. Safdieh said he thinks Step 2 will be next to go the pass/fail route. In reading between the lines of the announcement, he believes the test cosponsors didn’t make both pass/fail at once because it would have been “a nuclear bomb to the system.”
He credited the cosponsors with making what he called a “bold and momentous decision to initiate radical change in the overall transition between undergraduate and graduate medical education.”
Dr. Safdieh added that few in medicine were expecting Wednesday’s announcement.
“I think many of us were expecting them to go to quartile grading, not to go this far,” he said.
Dr. Safdieh suggested that, among those who may see downstream effects from the pass/fail move are offshore schools, such as those in the Caribbean. “Those schools rely on Step 1 to demonstrate that their students are meeting the rigor,” he said. But he hopes that this will lead to more holistic review.
“We’re hoping that this will force change in the system so that residency directors will look at more than just test-taking ability. They’ll look at publications and scholarship, community service and advocacy and performance in medical school,” Dr. Safdieh said.
Alison J. Whelan, MD, chief medical education officer of the Association of American Medical Colleges said in a statement, “The transition from medical school to residency training is a matter of great concern throughout academic medicine.
“The decision by the NBME and FSMB to change USMLE Step 1 score reporting to pass/fail was very carefully considered to balance student learning and student well-being,” she said. “The medical education community must now work together to identify and implement additional changes to improve the overall UME-GME [undergraduate and graduate medical education] transition system for all stakeholders and the AAMC is committed to helping lead this work.”
Dr. Fathy, Dr. Carmody, and Dr. Safdieh have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
News that the United States Medical Licensing Examination (USMLE) program will change its Step 1 scoring from a 3-digit number to pass/fail starting Jan. 1, 2022, has set off a flurry of shocked responses from students and physicians.
J. Bryan Carmody, MD, MPH, an assistant professor at Eastern Virginia Medical School in Norfolk, said in an interview that he was “stunned” when he heard the news on Wednesday and said the switch presents “the single biggest opportunity for medical school education reform since the Flexner Report,” which in 1910 established standards for modern medical education.
Numbers will continue for some tests
The USMLE cosponsors – the Federation of State Medical Boards (FSMB) and the National Board of Medical Examiners (NBME) – said that the Step 2 Clinical Knowledge (CK) exam and Step 3 will continue to be scored numerically. Step 2 Clinical Skills (CS) will continue its pass/fail system.
The change was made after Step 1 had been roundly criticized as playing too big a role in the process of becoming a physician and for causing students to study for the test instead of engaging fully in their medical education.
Ramie Fathy, a third-year medical student at the University of Pennsylvania, Philadelphia, currently studying for Step 1, said in an interview that it would have been nice personally to have the pass/fail choice, but he predicts both good and unintended consequences in the change.
The positive news, Mr. Fathy said, is that less emphasis will be put on the Step 1 test, which includes memorizing basic science details that may or not be relevant depending on later specialty choice.
“It’s not necessarily measuring what the test makers intended, which was whether or not a student can understand and apply basic science concepts to the practice of medicine,” he said.
“The current system encourages students to get as high a score as possible, which – after a certain point – translates to memorizing many little details that become increasingly less practically relevant,” Mr. Fathy said.
Pressure may move elsewhere?
However, Mr. Fathy worries that, without a scoring system to help decide who stands out in Step 1, residency program directors will depend more on the reputation of candidates’ medical school and the clout of the person writing a letter of recommendation – factors that are often influenced by family resources and social standing. That could wedge a further economic divide into the path to becoming a physician.
Mr. Fathy said he and fellow students are watching for information on what the passing bar will be and what happens with Step 2 Clinical Knowledge exam. USMLE has promised more information as soon as it is available.
“The question is whether that test will replace Step 1 as the standardized metric of student competency,” Mr. Fathy said, which would put more pressure on students further down the medical path.
Will Step 2 anxiety increase?
Dr. Carmody agreed that there is the danger that students now will spend their time studying for Step 2 CK at the expense of other parts of their education.
Meaningful reform will depend on the pass/fail move being coupled with other reforms, most importantly application caps, said Dr. Carmody, who teaches preclinical medical students and works with the residency program.
He has been blogging about Step 1 pass/fail for the past year.
Currently students can apply for as many residencies as they can pay for and Carmody said the number of applications per student has been rising over the past decade.
“That puts program directors under an impossible burden,” he said. “With our Step 1-based system, there’s significant inequality in the number of interviews people get. Programs end up overinviting the same group of people who look good on paper.”
People outside that group respond by sending more applications than they need to just to get a few interviews, Dr. Carmody added.
With caps, students would have an incentive to apply to only those programs in which they had a sincere interest, he said. Program directors also would then be better able to evaluate each application.
Switching Step 1 to pass/fail may have some effect on medical school burnout, Dr. Carmody said.
“It’s one thing to work hard when you’re on call and your patients depend on it,” he said. “But I would have a hard time staying up late every night studying something that I know in my heart is not going to help my patients, but I have to do it because I have to do better than the person who’s studying in the apartment next to me.”
Test has strayed from original purpose
Joseph Safdieh, MD, an assistant dean for clinical curriculum and director of the medical student neurology clerkship for the Weill Cornell Medicine, New York, sees the move as positive overall.
“We should not be using any single metric to define or describe our students’ overall profile,” he said in an interview.
“This has been a very significant anxiety point for our medical students for quite a number of years,” Dr. Safdieh said. “They were frustrated that their entire 4 years of medical school seemingly came down to one number.”
The test was created originally as one of three parts of licensure, he pointed out.
“Over the past 10 or 15 years, the exam has morphed to become a litmus test for very specific residency programs,” he said.
However, Dr. Safdieh has concerns that Step 2 will cultivate the same anxiety and may get too big a spotlight without the Step 1 metric, “although one could argue that test does more accurately reflect clinical material,” he said.
He also worries that students who have selected a specialty by the time they take Step 2 may find late in the game that they are less competitive in their field than they thought they were and may have to make a last-minute switch.
Dr. Safdieh said he thinks Step 2 will be next to go the pass/fail route. In reading between the lines of the announcement, he believes the test cosponsors didn’t make both pass/fail at once because it would have been “a nuclear bomb to the system.”
He credited the cosponsors with making what he called a “bold and momentous decision to initiate radical change in the overall transition between undergraduate and graduate medical education.”
Dr. Safdieh added that few in medicine were expecting Wednesday’s announcement.
“I think many of us were expecting them to go to quartile grading, not to go this far,” he said.
Dr. Safdieh suggested that, among those who may see downstream effects from the pass/fail move are offshore schools, such as those in the Caribbean. “Those schools rely on Step 1 to demonstrate that their students are meeting the rigor,” he said. But he hopes that this will lead to more holistic review.
“We’re hoping that this will force change in the system so that residency directors will look at more than just test-taking ability. They’ll look at publications and scholarship, community service and advocacy and performance in medical school,” Dr. Safdieh said.
Alison J. Whelan, MD, chief medical education officer of the Association of American Medical Colleges said in a statement, “The transition from medical school to residency training is a matter of great concern throughout academic medicine.
“The decision by the NBME and FSMB to change USMLE Step 1 score reporting to pass/fail was very carefully considered to balance student learning and student well-being,” she said. “The medical education community must now work together to identify and implement additional changes to improve the overall UME-GME [undergraduate and graduate medical education] transition system for all stakeholders and the AAMC is committed to helping lead this work.”
Dr. Fathy, Dr. Carmody, and Dr. Safdieh have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Safer CAR uses modified NK cells for advanced CLL, NHL
A chimeric antigen receptor (CAR) construct using transduced natural killer cells instead of T cells was associated with a high complete remission rate without the cytokine release syndrome frequently seen with CAR T cell therapy, early clinical trial results show.
The construct, consisting of natural killer (NK) cells derived from umbilical cord blood that have been transduced to target CD19-expressing cells combined with interleukin 15 and equipped with an “off” switch, offers the prospect of an off-the-shelf CAR product, reported Enli Liu, MD, and colleagues at the University of Texas MD Anderson Cancer Center in Houston.
“We found that allogeneic CAR-NK cells can be delivered in adoptive transfer without the serious cytokine release syndrome and neurologic toxic effects that have been associated with CAR T-cell therapy,” they wrote in The New England Journal of Medicine.
The modified NK cells were delivered to 9 of 11 patients with only partial human leukocyte antigen (HLA) matching, and in 2 patients with no matching, yet there were no cases of graft-versus host disease (GvHD), and no patients had symptoms of cytokine release syndrome (CRS), neurotoxicity, or hemophagocytic lymphohistiocytosis.
CAR T cell production “is a cumbersome process that requires coordination and collection of the cells and there’s several weeks of manufacturing, during which time patients often can have their lymphoma worsen, and so at times it’s a little bit of a race against the clock to get those cells manufactured,” Brian Hill, MD, PhD, director of the lymphoid malignancies program at Taussig Cancer Institute at Cleveland Clinic, said in an interview.
Dr. Hill, who was not involved in the study, said that the proof-of-principle study shows promising early results and offers the prospect of an effective and safe off-the-shelf therapeutic option for patients with lymphoid malignancies.
Advanced B-cell cancers
The investigators conducted a phase 1/2 trial in patients with B-cell lymphoid malignancies, including five patients with chronic lymphocytic leukemia (CLL), one patient with Richter’s transformation and one with accelerated CLL, three with transformed follicular lymphoma, two with diffuse large B-cell lymphoma (DLBCL), and one with follicular lymphoma (focally grade 3B).
The patients were all heavily pretreated, with 3 to as many as 11 prior lines of therapy.
The patients received cord blood-derived NK cells that had been transduced with a retroviral vector expressing genes that encode anti-CD19 CAR, interleukin-15, and inducible caspase 9 as a safety switch.
The cells were expanded in the lab and after the patients underwent lymphodepleting chemotherapy, they received the cells in a single infusion at one of three doses, either 1×105, 1×106, or 1×107 CAR-NK cells per kilogram of body weight.
As noted before, there were no cases of CRS, neurotoxicity, or GvHD and no increase over baseline in inflammatory cytokines, including interleukin-6, a key factor in the development and severity of CRS. The maximum tolerated dose was not reached.
Early efficacy
Of the 11 patients, 8 had a clinical response, and 7 had a complete remission, including 4 patients with lymphomas and 3 with CLL.
The patient with CLL with Richter’s transformation had a remission of the Richter’s component, but not of the CLL itself.
“This is particularly remarkable, because these patients are notoriously very difficult to treat, and the efficacy of autologous CAR T cell therapy in CLL and Richter’s patients has been hampered by lack of fitness of the patient’s own T cells when manufacturing the CAR T cell product, so this approach may obviate the need for autologous T cells in these patients,” Dr. Hill said.
The responses were rapid and occurred within 30 days of infusion at all dose levels. In addition, there was evidence of expansion and persistence of the modified NK cells at low levels for at least 1 year, despite the HLA mismatches between the NK cells and the recipients. The investigators speculated that the inclusion of interleukin-15 in the NL construct may at least partially account for the persistence of the cells and their antitumor activity.
Of the eight patients with a response, five had postremission therapy, including two patients with CLL who had minimal residual disease (MRD), one patient with follicular lymphoma and one with transformed follicular lymphoma who underwent hematopoietic stem-cell transplantation while in complete remission without evidence of MRD, and the patient with CLLL with Richter’s transformation with remission of the lymphoma component, who received a course of venetoclax.
The authors acknowledged that it may be difficult to assess the durability of response after CAR NK therapy in this study because of the allowed consolidation therapy for patients in remission.
They noted that although the patients in the current study each had a fresh CAR NK product manufactured for them, “we have shown that it is possible to produce more than 100 doses of CAR-NK cells from a single cord-blood unit. This capability, together with the apparently minimal HLA-matching requirements between the donor of CAR-NK cells and the patient, may pave the way for a truly off-the-shelf product that could increase treatment accessibility for many more patients.”
The National Institutes of Health supported the study. Dr. Liu disclosed a pending patent for methods of production of CAR-NK cells, and a patent held by MD Anderson for methods of treatment with NK cells. Dr. Hill is a member of the Hematology News editorial advisory board.
SOURCE: Liu E et al. N Engl J Med. 2020 Feb 6;382:545-53.
A chimeric antigen receptor (CAR) construct using transduced natural killer cells instead of T cells was associated with a high complete remission rate without the cytokine release syndrome frequently seen with CAR T cell therapy, early clinical trial results show.
The construct, consisting of natural killer (NK) cells derived from umbilical cord blood that have been transduced to target CD19-expressing cells combined with interleukin 15 and equipped with an “off” switch, offers the prospect of an off-the-shelf CAR product, reported Enli Liu, MD, and colleagues at the University of Texas MD Anderson Cancer Center in Houston.
“We found that allogeneic CAR-NK cells can be delivered in adoptive transfer without the serious cytokine release syndrome and neurologic toxic effects that have been associated with CAR T-cell therapy,” they wrote in The New England Journal of Medicine.
The modified NK cells were delivered to 9 of 11 patients with only partial human leukocyte antigen (HLA) matching, and in 2 patients with no matching, yet there were no cases of graft-versus host disease (GvHD), and no patients had symptoms of cytokine release syndrome (CRS), neurotoxicity, or hemophagocytic lymphohistiocytosis.
CAR T cell production “is a cumbersome process that requires coordination and collection of the cells and there’s several weeks of manufacturing, during which time patients often can have their lymphoma worsen, and so at times it’s a little bit of a race against the clock to get those cells manufactured,” Brian Hill, MD, PhD, director of the lymphoid malignancies program at Taussig Cancer Institute at Cleveland Clinic, said in an interview.
Dr. Hill, who was not involved in the study, said that the proof-of-principle study shows promising early results and offers the prospect of an effective and safe off-the-shelf therapeutic option for patients with lymphoid malignancies.
Advanced B-cell cancers
The investigators conducted a phase 1/2 trial in patients with B-cell lymphoid malignancies, including five patients with chronic lymphocytic leukemia (CLL), one patient with Richter’s transformation and one with accelerated CLL, three with transformed follicular lymphoma, two with diffuse large B-cell lymphoma (DLBCL), and one with follicular lymphoma (focally grade 3B).
The patients were all heavily pretreated, with 3 to as many as 11 prior lines of therapy.
The patients received cord blood-derived NK cells that had been transduced with a retroviral vector expressing genes that encode anti-CD19 CAR, interleukin-15, and inducible caspase 9 as a safety switch.
The cells were expanded in the lab and after the patients underwent lymphodepleting chemotherapy, they received the cells in a single infusion at one of three doses, either 1×105, 1×106, or 1×107 CAR-NK cells per kilogram of body weight.
As noted before, there were no cases of CRS, neurotoxicity, or GvHD and no increase over baseline in inflammatory cytokines, including interleukin-6, a key factor in the development and severity of CRS. The maximum tolerated dose was not reached.
Early efficacy
Of the 11 patients, 8 had a clinical response, and 7 had a complete remission, including 4 patients with lymphomas and 3 with CLL.
The patient with CLL with Richter’s transformation had a remission of the Richter’s component, but not of the CLL itself.
“This is particularly remarkable, because these patients are notoriously very difficult to treat, and the efficacy of autologous CAR T cell therapy in CLL and Richter’s patients has been hampered by lack of fitness of the patient’s own T cells when manufacturing the CAR T cell product, so this approach may obviate the need for autologous T cells in these patients,” Dr. Hill said.
The responses were rapid and occurred within 30 days of infusion at all dose levels. In addition, there was evidence of expansion and persistence of the modified NK cells at low levels for at least 1 year, despite the HLA mismatches between the NK cells and the recipients. The investigators speculated that the inclusion of interleukin-15 in the NL construct may at least partially account for the persistence of the cells and their antitumor activity.
Of the eight patients with a response, five had postremission therapy, including two patients with CLL who had minimal residual disease (MRD), one patient with follicular lymphoma and one with transformed follicular lymphoma who underwent hematopoietic stem-cell transplantation while in complete remission without evidence of MRD, and the patient with CLLL with Richter’s transformation with remission of the lymphoma component, who received a course of venetoclax.
The authors acknowledged that it may be difficult to assess the durability of response after CAR NK therapy in this study because of the allowed consolidation therapy for patients in remission.
They noted that although the patients in the current study each had a fresh CAR NK product manufactured for them, “we have shown that it is possible to produce more than 100 doses of CAR-NK cells from a single cord-blood unit. This capability, together with the apparently minimal HLA-matching requirements between the donor of CAR-NK cells and the patient, may pave the way for a truly off-the-shelf product that could increase treatment accessibility for many more patients.”
The National Institutes of Health supported the study. Dr. Liu disclosed a pending patent for methods of production of CAR-NK cells, and a patent held by MD Anderson for methods of treatment with NK cells. Dr. Hill is a member of the Hematology News editorial advisory board.
SOURCE: Liu E et al. N Engl J Med. 2020 Feb 6;382:545-53.
A chimeric antigen receptor (CAR) construct using transduced natural killer cells instead of T cells was associated with a high complete remission rate without the cytokine release syndrome frequently seen with CAR T cell therapy, early clinical trial results show.
The construct, consisting of natural killer (NK) cells derived from umbilical cord blood that have been transduced to target CD19-expressing cells combined with interleukin 15 and equipped with an “off” switch, offers the prospect of an off-the-shelf CAR product, reported Enli Liu, MD, and colleagues at the University of Texas MD Anderson Cancer Center in Houston.
“We found that allogeneic CAR-NK cells can be delivered in adoptive transfer without the serious cytokine release syndrome and neurologic toxic effects that have been associated with CAR T-cell therapy,” they wrote in The New England Journal of Medicine.
The modified NK cells were delivered to 9 of 11 patients with only partial human leukocyte antigen (HLA) matching, and in 2 patients with no matching, yet there were no cases of graft-versus host disease (GvHD), and no patients had symptoms of cytokine release syndrome (CRS), neurotoxicity, or hemophagocytic lymphohistiocytosis.
CAR T cell production “is a cumbersome process that requires coordination and collection of the cells and there’s several weeks of manufacturing, during which time patients often can have their lymphoma worsen, and so at times it’s a little bit of a race against the clock to get those cells manufactured,” Brian Hill, MD, PhD, director of the lymphoid malignancies program at Taussig Cancer Institute at Cleveland Clinic, said in an interview.
Dr. Hill, who was not involved in the study, said that the proof-of-principle study shows promising early results and offers the prospect of an effective and safe off-the-shelf therapeutic option for patients with lymphoid malignancies.
Advanced B-cell cancers
The investigators conducted a phase 1/2 trial in patients with B-cell lymphoid malignancies, including five patients with chronic lymphocytic leukemia (CLL), one patient with Richter’s transformation and one with accelerated CLL, three with transformed follicular lymphoma, two with diffuse large B-cell lymphoma (DLBCL), and one with follicular lymphoma (focally grade 3B).
The patients were all heavily pretreated, with 3 to as many as 11 prior lines of therapy.
The patients received cord blood-derived NK cells that had been transduced with a retroviral vector expressing genes that encode anti-CD19 CAR, interleukin-15, and inducible caspase 9 as a safety switch.
The cells were expanded in the lab and after the patients underwent lymphodepleting chemotherapy, they received the cells in a single infusion at one of three doses, either 1×105, 1×106, or 1×107 CAR-NK cells per kilogram of body weight.
As noted before, there were no cases of CRS, neurotoxicity, or GvHD and no increase over baseline in inflammatory cytokines, including interleukin-6, a key factor in the development and severity of CRS. The maximum tolerated dose was not reached.
Early efficacy
Of the 11 patients, 8 had a clinical response, and 7 had a complete remission, including 4 patients with lymphomas and 3 with CLL.
The patient with CLL with Richter’s transformation had a remission of the Richter’s component, but not of the CLL itself.
“This is particularly remarkable, because these patients are notoriously very difficult to treat, and the efficacy of autologous CAR T cell therapy in CLL and Richter’s patients has been hampered by lack of fitness of the patient’s own T cells when manufacturing the CAR T cell product, so this approach may obviate the need for autologous T cells in these patients,” Dr. Hill said.
The responses were rapid and occurred within 30 days of infusion at all dose levels. In addition, there was evidence of expansion and persistence of the modified NK cells at low levels for at least 1 year, despite the HLA mismatches between the NK cells and the recipients. The investigators speculated that the inclusion of interleukin-15 in the NL construct may at least partially account for the persistence of the cells and their antitumor activity.
Of the eight patients with a response, five had postremission therapy, including two patients with CLL who had minimal residual disease (MRD), one patient with follicular lymphoma and one with transformed follicular lymphoma who underwent hematopoietic stem-cell transplantation while in complete remission without evidence of MRD, and the patient with CLLL with Richter’s transformation with remission of the lymphoma component, who received a course of venetoclax.
The authors acknowledged that it may be difficult to assess the durability of response after CAR NK therapy in this study because of the allowed consolidation therapy for patients in remission.
They noted that although the patients in the current study each had a fresh CAR NK product manufactured for them, “we have shown that it is possible to produce more than 100 doses of CAR-NK cells from a single cord-blood unit. This capability, together with the apparently minimal HLA-matching requirements between the donor of CAR-NK cells and the patient, may pave the way for a truly off-the-shelf product that could increase treatment accessibility for many more patients.”
The National Institutes of Health supported the study. Dr. Liu disclosed a pending patent for methods of production of CAR-NK cells, and a patent held by MD Anderson for methods of treatment with NK cells. Dr. Hill is a member of the Hematology News editorial advisory board.
SOURCE: Liu E et al. N Engl J Med. 2020 Feb 6;382:545-53.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE