User login
AVAHO
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]


Rapid update to ASCO breast cancer guidelines after OlympiA data
The American Society of Clinical Oncology (ASCO) now recommends offering 1 year of adjuvant olaparib therapy to patients with early-stage HER2-negative, BRCA-mutated breast cancer who have completed chemotherapy and local treatment.
The change in management of hereditary breast cancer is outlined in an update to 2020 guidelines, and it comes as a “rapid recommendation” on the heels of the phase 3 OlympiA trial results, which indicated a 42% improvement in invasive and distant disease-free survival with the PARP inhibitor olaparib (Lynparza) in comparison with placebo.
The OlympiA trial results, as reported by this news organization, were presented during the plenary session of the ASCO 2021 annual meeting and were published June 3 in The New England Journal of Medicine.
“These clear and positive data prompted ASCO to issue a provisional update of the guideline recommendation focused specifically on the role of olaparib in this setting,” states an ASCO press release.
The previous 2020 guidelines stated: “There are insufficient data ... to recommend a PARP inhibitor for patients with nonmetastatic breast cancer.” The OlympiA trial changed that. ASCO now recommends that patients with early-stage, HER2-negative, BRCA-mutated breast cancer at high risk for recurrence be offered olaparib after completion of chemotherapy and local treatment, including radiotherapy.
The update states: “For those who had surgery first, adjuvant olaparib is recommended for patients with TNBC [triple-negative breast cancer] and tumor size greater than 2 cm or any involved axillary nodes. For patients with hormone receptor–positive disease, adjuvant olaparib is recommended for those with at least four involved axillary lymph nodes. For patients who had neoadjuvant chemotherapy, adjuvant olaparib is recommended for patients with TNBC and any residual cancer. Adjuvant olaparib is recommended for patients with residual disease and an estrogen receptor status and tumor grade (CSP+EG) score greater than or equal to 3.”
“The findings from the OlympiA trial – presented just last week – mark a significant improvement in the care of these patients,” Julie Garlow, MD, ASCO’s executive vice president and chief medical officer, states in the ASCO press release.
“ASCO’s Expert Guideline Panel and Evidence-based Medicine Committee noted this and then quickly produced and provisionally approved this guideline update to enable patients to begin to benefit from this research advance as quickly as possible,” she said.
A formal assessment and submission for publication in the Journal of Clinical Oncology will follow the release notes.
A version of this article first appeared on Medscape.com.
The American Society of Clinical Oncology (ASCO) now recommends offering 1 year of adjuvant olaparib therapy to patients with early-stage HER2-negative, BRCA-mutated breast cancer who have completed chemotherapy and local treatment.
The change in management of hereditary breast cancer is outlined in an update to 2020 guidelines, and it comes as a “rapid recommendation” on the heels of the phase 3 OlympiA trial results, which indicated a 42% improvement in invasive and distant disease-free survival with the PARP inhibitor olaparib (Lynparza) in comparison with placebo.
The OlympiA trial results, as reported by this news organization, were presented during the plenary session of the ASCO 2021 annual meeting and were published June 3 in The New England Journal of Medicine.
“These clear and positive data prompted ASCO to issue a provisional update of the guideline recommendation focused specifically on the role of olaparib in this setting,” states an ASCO press release.
The previous 2020 guidelines stated: “There are insufficient data ... to recommend a PARP inhibitor for patients with nonmetastatic breast cancer.” The OlympiA trial changed that. ASCO now recommends that patients with early-stage, HER2-negative, BRCA-mutated breast cancer at high risk for recurrence be offered olaparib after completion of chemotherapy and local treatment, including radiotherapy.
The update states: “For those who had surgery first, adjuvant olaparib is recommended for patients with TNBC [triple-negative breast cancer] and tumor size greater than 2 cm or any involved axillary nodes. For patients with hormone receptor–positive disease, adjuvant olaparib is recommended for those with at least four involved axillary lymph nodes. For patients who had neoadjuvant chemotherapy, adjuvant olaparib is recommended for patients with TNBC and any residual cancer. Adjuvant olaparib is recommended for patients with residual disease and an estrogen receptor status and tumor grade (CSP+EG) score greater than or equal to 3.”
“The findings from the OlympiA trial – presented just last week – mark a significant improvement in the care of these patients,” Julie Garlow, MD, ASCO’s executive vice president and chief medical officer, states in the ASCO press release.
“ASCO’s Expert Guideline Panel and Evidence-based Medicine Committee noted this and then quickly produced and provisionally approved this guideline update to enable patients to begin to benefit from this research advance as quickly as possible,” she said.
A formal assessment and submission for publication in the Journal of Clinical Oncology will follow the release notes.
A version of this article first appeared on Medscape.com.
The American Society of Clinical Oncology (ASCO) now recommends offering 1 year of adjuvant olaparib therapy to patients with early-stage HER2-negative, BRCA-mutated breast cancer who have completed chemotherapy and local treatment.
The change in management of hereditary breast cancer is outlined in an update to 2020 guidelines, and it comes as a “rapid recommendation” on the heels of the phase 3 OlympiA trial results, which indicated a 42% improvement in invasive and distant disease-free survival with the PARP inhibitor olaparib (Lynparza) in comparison with placebo.
The OlympiA trial results, as reported by this news organization, were presented during the plenary session of the ASCO 2021 annual meeting and were published June 3 in The New England Journal of Medicine.
“These clear and positive data prompted ASCO to issue a provisional update of the guideline recommendation focused specifically on the role of olaparib in this setting,” states an ASCO press release.
The previous 2020 guidelines stated: “There are insufficient data ... to recommend a PARP inhibitor for patients with nonmetastatic breast cancer.” The OlympiA trial changed that. ASCO now recommends that patients with early-stage, HER2-negative, BRCA-mutated breast cancer at high risk for recurrence be offered olaparib after completion of chemotherapy and local treatment, including radiotherapy.
The update states: “For those who had surgery first, adjuvant olaparib is recommended for patients with TNBC [triple-negative breast cancer] and tumor size greater than 2 cm or any involved axillary nodes. For patients with hormone receptor–positive disease, adjuvant olaparib is recommended for those with at least four involved axillary lymph nodes. For patients who had neoadjuvant chemotherapy, adjuvant olaparib is recommended for patients with TNBC and any residual cancer. Adjuvant olaparib is recommended for patients with residual disease and an estrogen receptor status and tumor grade (CSP+EG) score greater than or equal to 3.”
“The findings from the OlympiA trial – presented just last week – mark a significant improvement in the care of these patients,” Julie Garlow, MD, ASCO’s executive vice president and chief medical officer, states in the ASCO press release.
“ASCO’s Expert Guideline Panel and Evidence-based Medicine Committee noted this and then quickly produced and provisionally approved this guideline update to enable patients to begin to benefit from this research advance as quickly as possible,” she said.
A formal assessment and submission for publication in the Journal of Clinical Oncology will follow the release notes.
A version of this article first appeared on Medscape.com.
Reduced-intensity transplant benefits older patients with AML
Among older patients with acute myeloid leukemia (AML), survival is significantly better when they undergo reduced-intensity conditioning (RIC) before receiving an allogeneic hematopoietic cell transplant (HCT) at first remission. This improvement in survival is seen regardless of key factors such as genotype and the status of minimal residual disease (MRD) after initial chemotherapy, results from two large randomized trials show.
“Two consecutive trials of more than 1,500 older AML patients above 60 years of age demonstrate a consistent benefit for RIC transplant in first remission,” said first author Nigel Russell, MD, of Guy’s Hospital, London, and Nottingham University, England. “This benefit is seen independent of their post-course 1 MRD status,” he added.
Dr. Russell presented the new data at the European Hematology Association (EHA) 2021 Annual Meeting.
Commenting on the study, Charles Craddock, MD, said in an interview that the results “confirm the growing importance of RIC transplantation as a central treatment management strategy in high-risk AML and in this population high risk patients over 60.”
“[These findings] reinforce the evolving treatment paradigm that, in fit adults over 60 with AML, hematopoietic cell transplantation should be considered an essential component of their management plan,” said Dr. Craddock, academic director of the Center for Clinical Haematology, Queen Elizabeth Hospital, Birmingham, England.
Patients with AML who are older than 60 years can achieve complete remission with intensive chemotherapy alone; however, relapse is common, and only about 20% survive for 5 years, Dr. Russell explained.
HCT significantly improves survival outcomes, and the development of RIC has made transplantation accessible to high-risk patients by making the procedure more tolerable with lower toxicity in comparison with conventional conditioning regimens.
However, there is ongoing debate over the prognostic effect of key factors in pretransplant conditioning that may be predictive of the risk for post-transplant relapse – in particular, the presence of MRD after the first course of conditioning, he explained.
To more closely investigate those factors and the rate of survival of older patients with AML who undergo RIC transplant, Dr. Russell and his colleagues evaluated results from the National Cancer Research Institute’s (NCRI) AML16 trial, which was conducted from 2006 to 2012, and interim results from the NCRI AML18 trial, which started in 2015 and is ongoing.
Both trials employed double induction of daunorubicin and clofarabine or, in the AML16 trial, AraC ± gemtuzumab, and in the AML18 trial, daunorubicin and AraC (DA) + gemtuzumab.
In AML18, patients who were MRD positive after course 1 were randomly assigned to undergo either an intensification randomization after either FLAG-Ida or DA+cladribine or DA alone.
In AML16, of 983 patients in first complete response, 144 (15%) subsequently underwent RIC transplant. The median follow-up for survival from complete response was 45 months.
In the AML18 trial, of 847 patients, 648 patients achieved complete response. Among them, 201 (31%) underwent transplant. The median follow-up of survival was 45 months.
The results of both trials showed greater benefit with RIC transplant versus chemotherapy alone.
In the AML16 trial, among patients aged 60 to 70 who received RIC, survival at 5 years was significantly improved compared with chemotherapy alone (37% vs. 19%; hazard ratio, 0.65; 95% confidence interval, 0.52-0.82; P < .001).
In AML16, the higher survival benefit in comparison with chemotherapy alone was observed in the RIC group across subgroups of risk level, as stratified according to in the multivariate Wheatley risk group score. Subgroup stratification was based on age, cytogenics, and other factors (HR, 0.66; 95% CI, 0.53-0.83; P < .001).
Importantly, the survival benefits were significantly greater with RIC transplant regardless of MRD-negative or MRD-positive status after course 1 (HR, 0.68; 95% CI, 0.54-0.85; P < .001).
Allograft transplant was also more favorable regardless of FLT3 ITD or NPM1 mutation status (P for heterogeneity by genetic subgroups, 0.61).
In AML16, no groups were found to have benefited more with RIC. Consequently, the criteria for transplant in AML18 trial were based on patients’ health status and donor availability.
An interim analysis of the ongoing AML 18 trial further underscored an overall benefit of RIC transplant. Rates of 3-year survival from remission were 48% with RIC transplant, versus 37.4% with chemotherapy alone (P = .027). The benefit was independent of MRD status after conditioning course 1, similar to the AML16 results (HR, 0.71; 95% CI, 0.54-0.95; P = .02).
Although the rate of transplantation in the AML18 trial was higher among patients who were MRD positive in comparison with those who were MRD negative (36% vs. 24.8%), the rates of post-transplant survival were not significantly different between those who were MRD positive and those who were MRD negative after course 1 (51.1% vs. 46.6% at 3 years; P = .84).
The authors evaluated the effects of a second conditioning course on transplant outcomes among patients who did not initially achieve an MRD-negative complete remission.
They found that 60% of patients did convert from MRD-positive to MRD-negative status after course 2. Among those patients, the survival versus chemotherapy alone was substantially higher (HR, 0.32; 95% CI, 0.11-0.92) compared to those who remained MRD-negative (HR 0.74; 95% CI, 0.32-1.72).
However, the authors note that, owing to a lack of heterogeneity, the results don’t necessarily mean that the patients who remained MRD positive did not also benefit from transplant.
“There was a significant benefit for transplant in those who converted to MRD negativity,” Dr. Russell said.
“With a hazard ratio of .32, this was far superior to those who remained MRD-positive post course 2,” he said.
“These results show that MRD status after course 1 is important information in terms of response to therapy and can alter your treatment strategy if you’re considering a transplant as an option for these patients,” Dr. Russell told this news organization.
In further commenting, Dr. Craddock said the research highlights the importance of randomized trials with regard to whether patients who are MRD-positive before transplant will benefit from an additional course of therapy to reduce the MRD load.
“Most get two courses, but the question is, if they are still MRD positive, should they get a third course, and if so, what should that look like?” he said.
“There are currently no randomized controlled trials to address that ongoing question, and they need to be done,” he added.
Dr. Russell has relationships with Pfizer, Astellas, and Jazz Pharma. Dr. Craddock has a relationship with Bristol-Myers Squibb.
A version of this article first appeared on Medscape.com.
Among older patients with acute myeloid leukemia (AML), survival is significantly better when they undergo reduced-intensity conditioning (RIC) before receiving an allogeneic hematopoietic cell transplant (HCT) at first remission. This improvement in survival is seen regardless of key factors such as genotype and the status of minimal residual disease (MRD) after initial chemotherapy, results from two large randomized trials show.
“Two consecutive trials of more than 1,500 older AML patients above 60 years of age demonstrate a consistent benefit for RIC transplant in first remission,” said first author Nigel Russell, MD, of Guy’s Hospital, London, and Nottingham University, England. “This benefit is seen independent of their post-course 1 MRD status,” he added.
Dr. Russell presented the new data at the European Hematology Association (EHA) 2021 Annual Meeting.
Commenting on the study, Charles Craddock, MD, said in an interview that the results “confirm the growing importance of RIC transplantation as a central treatment management strategy in high-risk AML and in this population high risk patients over 60.”
“[These findings] reinforce the evolving treatment paradigm that, in fit adults over 60 with AML, hematopoietic cell transplantation should be considered an essential component of their management plan,” said Dr. Craddock, academic director of the Center for Clinical Haematology, Queen Elizabeth Hospital, Birmingham, England.
Patients with AML who are older than 60 years can achieve complete remission with intensive chemotherapy alone; however, relapse is common, and only about 20% survive for 5 years, Dr. Russell explained.
HCT significantly improves survival outcomes, and the development of RIC has made transplantation accessible to high-risk patients by making the procedure more tolerable with lower toxicity in comparison with conventional conditioning regimens.
However, there is ongoing debate over the prognostic effect of key factors in pretransplant conditioning that may be predictive of the risk for post-transplant relapse – in particular, the presence of MRD after the first course of conditioning, he explained.
To more closely investigate those factors and the rate of survival of older patients with AML who undergo RIC transplant, Dr. Russell and his colleagues evaluated results from the National Cancer Research Institute’s (NCRI) AML16 trial, which was conducted from 2006 to 2012, and interim results from the NCRI AML18 trial, which started in 2015 and is ongoing.
Both trials employed double induction of daunorubicin and clofarabine or, in the AML16 trial, AraC ± gemtuzumab, and in the AML18 trial, daunorubicin and AraC (DA) + gemtuzumab.
In AML18, patients who were MRD positive after course 1 were randomly assigned to undergo either an intensification randomization after either FLAG-Ida or DA+cladribine or DA alone.
In AML16, of 983 patients in first complete response, 144 (15%) subsequently underwent RIC transplant. The median follow-up for survival from complete response was 45 months.
In the AML18 trial, of 847 patients, 648 patients achieved complete response. Among them, 201 (31%) underwent transplant. The median follow-up of survival was 45 months.
The results of both trials showed greater benefit with RIC transplant versus chemotherapy alone.
In the AML16 trial, among patients aged 60 to 70 who received RIC, survival at 5 years was significantly improved compared with chemotherapy alone (37% vs. 19%; hazard ratio, 0.65; 95% confidence interval, 0.52-0.82; P < .001).
In AML16, the higher survival benefit in comparison with chemotherapy alone was observed in the RIC group across subgroups of risk level, as stratified according to in the multivariate Wheatley risk group score. Subgroup stratification was based on age, cytogenics, and other factors (HR, 0.66; 95% CI, 0.53-0.83; P < .001).
Importantly, the survival benefits were significantly greater with RIC transplant regardless of MRD-negative or MRD-positive status after course 1 (HR, 0.68; 95% CI, 0.54-0.85; P < .001).
Allograft transplant was also more favorable regardless of FLT3 ITD or NPM1 mutation status (P for heterogeneity by genetic subgroups, 0.61).
In AML16, no groups were found to have benefited more with RIC. Consequently, the criteria for transplant in AML18 trial were based on patients’ health status and donor availability.
An interim analysis of the ongoing AML 18 trial further underscored an overall benefit of RIC transplant. Rates of 3-year survival from remission were 48% with RIC transplant, versus 37.4% with chemotherapy alone (P = .027). The benefit was independent of MRD status after conditioning course 1, similar to the AML16 results (HR, 0.71; 95% CI, 0.54-0.95; P = .02).
Although the rate of transplantation in the AML18 trial was higher among patients who were MRD positive in comparison with those who were MRD negative (36% vs. 24.8%), the rates of post-transplant survival were not significantly different between those who were MRD positive and those who were MRD negative after course 1 (51.1% vs. 46.6% at 3 years; P = .84).
The authors evaluated the effects of a second conditioning course on transplant outcomes among patients who did not initially achieve an MRD-negative complete remission.
They found that 60% of patients did convert from MRD-positive to MRD-negative status after course 2. Among those patients, the survival versus chemotherapy alone was substantially higher (HR, 0.32; 95% CI, 0.11-0.92) compared to those who remained MRD-negative (HR 0.74; 95% CI, 0.32-1.72).
However, the authors note that, owing to a lack of heterogeneity, the results don’t necessarily mean that the patients who remained MRD positive did not also benefit from transplant.
“There was a significant benefit for transplant in those who converted to MRD negativity,” Dr. Russell said.
“With a hazard ratio of .32, this was far superior to those who remained MRD-positive post course 2,” he said.
“These results show that MRD status after course 1 is important information in terms of response to therapy and can alter your treatment strategy if you’re considering a transplant as an option for these patients,” Dr. Russell told this news organization.
In further commenting, Dr. Craddock said the research highlights the importance of randomized trials with regard to whether patients who are MRD-positive before transplant will benefit from an additional course of therapy to reduce the MRD load.
“Most get two courses, but the question is, if they are still MRD positive, should they get a third course, and if so, what should that look like?” he said.
“There are currently no randomized controlled trials to address that ongoing question, and they need to be done,” he added.
Dr. Russell has relationships with Pfizer, Astellas, and Jazz Pharma. Dr. Craddock has a relationship with Bristol-Myers Squibb.
A version of this article first appeared on Medscape.com.
Among older patients with acute myeloid leukemia (AML), survival is significantly better when they undergo reduced-intensity conditioning (RIC) before receiving an allogeneic hematopoietic cell transplant (HCT) at first remission. This improvement in survival is seen regardless of key factors such as genotype and the status of minimal residual disease (MRD) after initial chemotherapy, results from two large randomized trials show.
“Two consecutive trials of more than 1,500 older AML patients above 60 years of age demonstrate a consistent benefit for RIC transplant in first remission,” said first author Nigel Russell, MD, of Guy’s Hospital, London, and Nottingham University, England. “This benefit is seen independent of their post-course 1 MRD status,” he added.
Dr. Russell presented the new data at the European Hematology Association (EHA) 2021 Annual Meeting.
Commenting on the study, Charles Craddock, MD, said in an interview that the results “confirm the growing importance of RIC transplantation as a central treatment management strategy in high-risk AML and in this population high risk patients over 60.”
“[These findings] reinforce the evolving treatment paradigm that, in fit adults over 60 with AML, hematopoietic cell transplantation should be considered an essential component of their management plan,” said Dr. Craddock, academic director of the Center for Clinical Haematology, Queen Elizabeth Hospital, Birmingham, England.
Patients with AML who are older than 60 years can achieve complete remission with intensive chemotherapy alone; however, relapse is common, and only about 20% survive for 5 years, Dr. Russell explained.
HCT significantly improves survival outcomes, and the development of RIC has made transplantation accessible to high-risk patients by making the procedure more tolerable with lower toxicity in comparison with conventional conditioning regimens.
However, there is ongoing debate over the prognostic effect of key factors in pretransplant conditioning that may be predictive of the risk for post-transplant relapse – in particular, the presence of MRD after the first course of conditioning, he explained.
To more closely investigate those factors and the rate of survival of older patients with AML who undergo RIC transplant, Dr. Russell and his colleagues evaluated results from the National Cancer Research Institute’s (NCRI) AML16 trial, which was conducted from 2006 to 2012, and interim results from the NCRI AML18 trial, which started in 2015 and is ongoing.
Both trials employed double induction of daunorubicin and clofarabine or, in the AML16 trial, AraC ± gemtuzumab, and in the AML18 trial, daunorubicin and AraC (DA) + gemtuzumab.
In AML18, patients who were MRD positive after course 1 were randomly assigned to undergo either an intensification randomization after either FLAG-Ida or DA+cladribine or DA alone.
In AML16, of 983 patients in first complete response, 144 (15%) subsequently underwent RIC transplant. The median follow-up for survival from complete response was 45 months.
In the AML18 trial, of 847 patients, 648 patients achieved complete response. Among them, 201 (31%) underwent transplant. The median follow-up of survival was 45 months.
The results of both trials showed greater benefit with RIC transplant versus chemotherapy alone.
In the AML16 trial, among patients aged 60 to 70 who received RIC, survival at 5 years was significantly improved compared with chemotherapy alone (37% vs. 19%; hazard ratio, 0.65; 95% confidence interval, 0.52-0.82; P < .001).
In AML16, the higher survival benefit in comparison with chemotherapy alone was observed in the RIC group across subgroups of risk level, as stratified according to in the multivariate Wheatley risk group score. Subgroup stratification was based on age, cytogenics, and other factors (HR, 0.66; 95% CI, 0.53-0.83; P < .001).
Importantly, the survival benefits were significantly greater with RIC transplant regardless of MRD-negative or MRD-positive status after course 1 (HR, 0.68; 95% CI, 0.54-0.85; P < .001).
Allograft transplant was also more favorable regardless of FLT3 ITD or NPM1 mutation status (P for heterogeneity by genetic subgroups, 0.61).
In AML16, no groups were found to have benefited more with RIC. Consequently, the criteria for transplant in AML18 trial were based on patients’ health status and donor availability.
An interim analysis of the ongoing AML 18 trial further underscored an overall benefit of RIC transplant. Rates of 3-year survival from remission were 48% with RIC transplant, versus 37.4% with chemotherapy alone (P = .027). The benefit was independent of MRD status after conditioning course 1, similar to the AML16 results (HR, 0.71; 95% CI, 0.54-0.95; P = .02).
Although the rate of transplantation in the AML18 trial was higher among patients who were MRD positive in comparison with those who were MRD negative (36% vs. 24.8%), the rates of post-transplant survival were not significantly different between those who were MRD positive and those who were MRD negative after course 1 (51.1% vs. 46.6% at 3 years; P = .84).
The authors evaluated the effects of a second conditioning course on transplant outcomes among patients who did not initially achieve an MRD-negative complete remission.
They found that 60% of patients did convert from MRD-positive to MRD-negative status after course 2. Among those patients, the survival versus chemotherapy alone was substantially higher (HR, 0.32; 95% CI, 0.11-0.92) compared to those who remained MRD-negative (HR 0.74; 95% CI, 0.32-1.72).
However, the authors note that, owing to a lack of heterogeneity, the results don’t necessarily mean that the patients who remained MRD positive did not also benefit from transplant.
“There was a significant benefit for transplant in those who converted to MRD negativity,” Dr. Russell said.
“With a hazard ratio of .32, this was far superior to those who remained MRD-positive post course 2,” he said.
“These results show that MRD status after course 1 is important information in terms of response to therapy and can alter your treatment strategy if you’re considering a transplant as an option for these patients,” Dr. Russell told this news organization.
In further commenting, Dr. Craddock said the research highlights the importance of randomized trials with regard to whether patients who are MRD-positive before transplant will benefit from an additional course of therapy to reduce the MRD load.
“Most get two courses, but the question is, if they are still MRD positive, should they get a third course, and if so, what should that look like?” he said.
“There are currently no randomized controlled trials to address that ongoing question, and they need to be done,” he added.
Dr. Russell has relationships with Pfizer, Astellas, and Jazz Pharma. Dr. Craddock has a relationship with Bristol-Myers Squibb.
A version of this article first appeared on Medscape.com.
DCIS: Biosignature helps guide postlumpectomy decisions
A biosignature tool helps women avoid unnecessary radiotherapy after undergoing lumpectomy for ductal carcinoma in situ (DCIS) – and also identifies women who need more intense treatment.
The DCISionRT test (PreludeDx) and its response subtype (Rst) biosignature provide personalized risk assessment, explains Frank Vicini, MD, a radiation oncologist at GenesisCare and a member of NRG Oncology, Pontiac, Mich.
He presented data on the test at a poster at the recent American Society of Clinical Oncology Annual Meeting.
They can also identify patients who would likely benefit from radiotherapy, Dr. Vicini reported.
The tool shows promise for identifying those whose cancer is likely to recur despite undergoing postlumpectomy radiotherapy – women who might benefit from intensified or alternate treatment approaches, he added.
The latter finding is particularly provocative because it suggests that the biosignatures “may appropriately identify patients with very radioresistant ductal carcinoma in situ,” Benjamin D. Smith, MD, commented during a poster discussion session at the meeting.
“I think these findings merit validation in translational research models,” said Dr. Smith, a radiation oncologist and professor of radiation oncology and health services research at the University of Texas MD Anderson Cancer Center, Houston.
DCISionRT, Rst, and risk
DCISionRT combines molecular biology innovations with risk-based scores to assess risk for recurrence, which is classified as either low or elevated, according to the test developer, PreludeDx.
Dr. Vicini and colleagues used the test to classify tissue samples from 485 women who were part of previous DCISionRT validation cohorts in Sweden, Australia, and the United States. The patients underwent breast cancer surgery (BCS) with or without radiotherapy between 1996 and 2011.
The Rst biosignature was used to further categorize those in the elevated-risk group as having a good response subtype (good Rst) or a poor response subtype (poor Rst) after BCS plus radiotherapy.
Radiotherapy was associated with significantly reduced recurrence rates among women with elevated risk and a good Rst (the hazard ratios for ipsilateral breast tumor recurrence [IBTR] and invasive breast cancer [IBC] were 0.18 and 0.15, respectively).
No radiotherapy benefit was seen among those with elevated risk and poor Rst.
The investigators also reported that, among patients with a poor Rst, 10-year IBTR and IBC rates were 25% and 16%, respectively, regardless of whether they received radiotherapy. These rates were much higher than the rates among women with good Rst (6.6% and 4.5%; hazard ratio, 3.6 and 4.4, respectively).
No significant difference was seen in 10-year IBTR and IBC rates among patients in the low-risk group, with or without radiotherapy.
Traditional clinicopathologic risk factors, including age younger than 50 years, grade 3 disease, and tumor size greater than 2.5 cm, did not identify poor versus good response subtypes in this cohort, and on multivariable analysis, neither of these factors nor endocrine therapy was significantly associated with IBTR or IBC.
Prospective validation needed
In his discussion, Dr. Smith said that the study provides “important data” that further validate the DCISionRT platform alone for assessing risk among women with DCIS who undergo BCS. But it is the Rst biosignature, which allows clinicians to “predict radioresistance of residual malignant chromogens following lumpectomy plus radiation therapy,” that really stands out, he added.
From the data presented, “it is reasonable to conclude that patients with a poor Rst score treated with lumpectomy and radiation had a much higher risk of in-breast tumor recurrence than one might predict or anticipate based on existing published randomized clinical trial data,” he said.
“In my opinion, it is very important to prospectively validate this finding with other cohorts,” he said. “Moving forward, I think there may come a time where there may be interest in studying radiosensitizing agents for poor-Rst ductal carcinoma in situ that are resistant to standard doses of radiation, and it may be that we consider the Rst as a factor moving forward in selecting patients for BCT versus mastectomy.”
However, because 75% of patients at elevated risk with poor Rst who undergo lumpectomy and radiotherapy do not experience recurrence in the decade following their treatment, it would be “inappropriate and misguided” to start recommending mastectomy for patients at DCISionRT elevated risk who have poor Rst, he said.
The study was funded by PreludeDx. Dr. Vicini reported employment with 21st Century Oncology and financial relationships with ImpediMed, Prelude Therapeutics, and Concure Oncology. Dr. Smith, through his employer, has an equity interest in Oncora Medical through a partnership agreement. He also has an uncompensated relationship with the American Society for Radiation Oncology.
A version of this article first appeared on Medscape.com.
A biosignature tool helps women avoid unnecessary radiotherapy after undergoing lumpectomy for ductal carcinoma in situ (DCIS) – and also identifies women who need more intense treatment.
The DCISionRT test (PreludeDx) and its response subtype (Rst) biosignature provide personalized risk assessment, explains Frank Vicini, MD, a radiation oncologist at GenesisCare and a member of NRG Oncology, Pontiac, Mich.
He presented data on the test at a poster at the recent American Society of Clinical Oncology Annual Meeting.
They can also identify patients who would likely benefit from radiotherapy, Dr. Vicini reported.
The tool shows promise for identifying those whose cancer is likely to recur despite undergoing postlumpectomy radiotherapy – women who might benefit from intensified or alternate treatment approaches, he added.
The latter finding is particularly provocative because it suggests that the biosignatures “may appropriately identify patients with very radioresistant ductal carcinoma in situ,” Benjamin D. Smith, MD, commented during a poster discussion session at the meeting.
“I think these findings merit validation in translational research models,” said Dr. Smith, a radiation oncologist and professor of radiation oncology and health services research at the University of Texas MD Anderson Cancer Center, Houston.
DCISionRT, Rst, and risk
DCISionRT combines molecular biology innovations with risk-based scores to assess risk for recurrence, which is classified as either low or elevated, according to the test developer, PreludeDx.
Dr. Vicini and colleagues used the test to classify tissue samples from 485 women who were part of previous DCISionRT validation cohorts in Sweden, Australia, and the United States. The patients underwent breast cancer surgery (BCS) with or without radiotherapy between 1996 and 2011.
The Rst biosignature was used to further categorize those in the elevated-risk group as having a good response subtype (good Rst) or a poor response subtype (poor Rst) after BCS plus radiotherapy.
Radiotherapy was associated with significantly reduced recurrence rates among women with elevated risk and a good Rst (the hazard ratios for ipsilateral breast tumor recurrence [IBTR] and invasive breast cancer [IBC] were 0.18 and 0.15, respectively).
No radiotherapy benefit was seen among those with elevated risk and poor Rst.
The investigators also reported that, among patients with a poor Rst, 10-year IBTR and IBC rates were 25% and 16%, respectively, regardless of whether they received radiotherapy. These rates were much higher than the rates among women with good Rst (6.6% and 4.5%; hazard ratio, 3.6 and 4.4, respectively).
No significant difference was seen in 10-year IBTR and IBC rates among patients in the low-risk group, with or without radiotherapy.
Traditional clinicopathologic risk factors, including age younger than 50 years, grade 3 disease, and tumor size greater than 2.5 cm, did not identify poor versus good response subtypes in this cohort, and on multivariable analysis, neither of these factors nor endocrine therapy was significantly associated with IBTR or IBC.
Prospective validation needed
In his discussion, Dr. Smith said that the study provides “important data” that further validate the DCISionRT platform alone for assessing risk among women with DCIS who undergo BCS. But it is the Rst biosignature, which allows clinicians to “predict radioresistance of residual malignant chromogens following lumpectomy plus radiation therapy,” that really stands out, he added.
From the data presented, “it is reasonable to conclude that patients with a poor Rst score treated with lumpectomy and radiation had a much higher risk of in-breast tumor recurrence than one might predict or anticipate based on existing published randomized clinical trial data,” he said.
“In my opinion, it is very important to prospectively validate this finding with other cohorts,” he said. “Moving forward, I think there may come a time where there may be interest in studying radiosensitizing agents for poor-Rst ductal carcinoma in situ that are resistant to standard doses of radiation, and it may be that we consider the Rst as a factor moving forward in selecting patients for BCT versus mastectomy.”
However, because 75% of patients at elevated risk with poor Rst who undergo lumpectomy and radiotherapy do not experience recurrence in the decade following their treatment, it would be “inappropriate and misguided” to start recommending mastectomy for patients at DCISionRT elevated risk who have poor Rst, he said.
The study was funded by PreludeDx. Dr. Vicini reported employment with 21st Century Oncology and financial relationships with ImpediMed, Prelude Therapeutics, and Concure Oncology. Dr. Smith, through his employer, has an equity interest in Oncora Medical through a partnership agreement. He also has an uncompensated relationship with the American Society for Radiation Oncology.
A version of this article first appeared on Medscape.com.
A biosignature tool helps women avoid unnecessary radiotherapy after undergoing lumpectomy for ductal carcinoma in situ (DCIS) – and also identifies women who need more intense treatment.
The DCISionRT test (PreludeDx) and its response subtype (Rst) biosignature provide personalized risk assessment, explains Frank Vicini, MD, a radiation oncologist at GenesisCare and a member of NRG Oncology, Pontiac, Mich.
He presented data on the test at a poster at the recent American Society of Clinical Oncology Annual Meeting.
They can also identify patients who would likely benefit from radiotherapy, Dr. Vicini reported.
The tool shows promise for identifying those whose cancer is likely to recur despite undergoing postlumpectomy radiotherapy – women who might benefit from intensified or alternate treatment approaches, he added.
The latter finding is particularly provocative because it suggests that the biosignatures “may appropriately identify patients with very radioresistant ductal carcinoma in situ,” Benjamin D. Smith, MD, commented during a poster discussion session at the meeting.
“I think these findings merit validation in translational research models,” said Dr. Smith, a radiation oncologist and professor of radiation oncology and health services research at the University of Texas MD Anderson Cancer Center, Houston.
DCISionRT, Rst, and risk
DCISionRT combines molecular biology innovations with risk-based scores to assess risk for recurrence, which is classified as either low or elevated, according to the test developer, PreludeDx.
Dr. Vicini and colleagues used the test to classify tissue samples from 485 women who were part of previous DCISionRT validation cohorts in Sweden, Australia, and the United States. The patients underwent breast cancer surgery (BCS) with or without radiotherapy between 1996 and 2011.
The Rst biosignature was used to further categorize those in the elevated-risk group as having a good response subtype (good Rst) or a poor response subtype (poor Rst) after BCS plus radiotherapy.
Radiotherapy was associated with significantly reduced recurrence rates among women with elevated risk and a good Rst (the hazard ratios for ipsilateral breast tumor recurrence [IBTR] and invasive breast cancer [IBC] were 0.18 and 0.15, respectively).
No radiotherapy benefit was seen among those with elevated risk and poor Rst.
The investigators also reported that, among patients with a poor Rst, 10-year IBTR and IBC rates were 25% and 16%, respectively, regardless of whether they received radiotherapy. These rates were much higher than the rates among women with good Rst (6.6% and 4.5%; hazard ratio, 3.6 and 4.4, respectively).
No significant difference was seen in 10-year IBTR and IBC rates among patients in the low-risk group, with or without radiotherapy.
Traditional clinicopathologic risk factors, including age younger than 50 years, grade 3 disease, and tumor size greater than 2.5 cm, did not identify poor versus good response subtypes in this cohort, and on multivariable analysis, neither of these factors nor endocrine therapy was significantly associated with IBTR or IBC.
Prospective validation needed
In his discussion, Dr. Smith said that the study provides “important data” that further validate the DCISionRT platform alone for assessing risk among women with DCIS who undergo BCS. But it is the Rst biosignature, which allows clinicians to “predict radioresistance of residual malignant chromogens following lumpectomy plus radiation therapy,” that really stands out, he added.
From the data presented, “it is reasonable to conclude that patients with a poor Rst score treated with lumpectomy and radiation had a much higher risk of in-breast tumor recurrence than one might predict or anticipate based on existing published randomized clinical trial data,” he said.
“In my opinion, it is very important to prospectively validate this finding with other cohorts,” he said. “Moving forward, I think there may come a time where there may be interest in studying radiosensitizing agents for poor-Rst ductal carcinoma in situ that are resistant to standard doses of radiation, and it may be that we consider the Rst as a factor moving forward in selecting patients for BCT versus mastectomy.”
However, because 75% of patients at elevated risk with poor Rst who undergo lumpectomy and radiotherapy do not experience recurrence in the decade following their treatment, it would be “inappropriate and misguided” to start recommending mastectomy for patients at DCISionRT elevated risk who have poor Rst, he said.
The study was funded by PreludeDx. Dr. Vicini reported employment with 21st Century Oncology and financial relationships with ImpediMed, Prelude Therapeutics, and Concure Oncology. Dr. Smith, through his employer, has an equity interest in Oncora Medical through a partnership agreement. He also has an uncompensated relationship with the American Society for Radiation Oncology.
A version of this article first appeared on Medscape.com.
Americans’ sun protection practices fall short of intentions
commissioned by the American Academy of Dermatology.
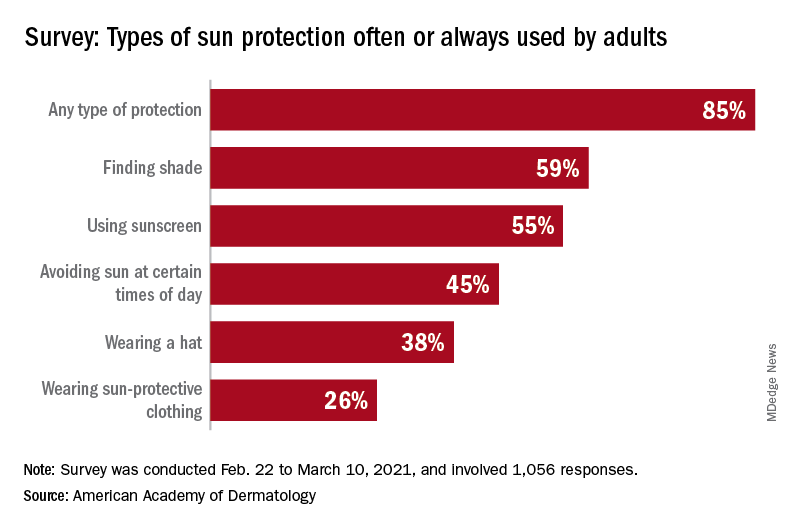
With the pandemic seemingly behind it, the United States enters the summer months facing the paradox of sun protection. Four out of five adults know that sunscreen should be reapplied every 2 hours when they’re outdoors, but only one in three make the actual effort, and 77% are likely to use sunscreen at the beach or a pool, compared with 41% when they’re gardening or working outside on their homes, the AAD reported.
“These findings are surprising and seem to suggest that many people do not take skin cancer seriously or perhaps believe skin cancer won’t happen to them,” Robert T. Brodell, MD, professor of dermatology at the University of Mississippi Medical Center, Jackson, said in a written statement from the AAD, adding that “unprotected exposure to ultraviolet rays is the most preventable risk factor for skin cancer, including melanoma.”
A quarter of all survey respondents reported getting sunburned in 2020, with the youngest adults most likely to feel the wrath of the sun. Sunburn was reported by 43% of those aged 18-23 years, 37% of those aged 24-39, 25% of the 40- to 55-year-olds, 12% of the 56- to 74-year-olds, and 7% of those aged 75 and older. More than a quarter of those who got sunburned said that it was bad enough to make their clothes feel uncomfortable, the academy said.
“Americans see the damaging effects of the sun on their skin as they get older, and two out of three look back and wish they had been more careful. But when it comes to cancer, specifically, most feel unconcerned in spite of their own risk,” according to a statement from Versta Research, which conducted the poll on behalf of the AAD. The survey was conducted from Feb. 22 to March 10, 2021, and involved 1,056 respondents, with a ±3% margin of error.
The lack of concern for skin cancer looks like this: More than two-thirds of the respondents (69%) have at least one possible risk factor – lighter skin tone, blue or green eyes, more than 50 moles, family history – but only 36% expressed concern about developing it. “Indeed, half of survey respondents (49%) say they are more worried about avoiding sunburn than they are about preventing skin cancer, and a third (32%) are more worried about avoiding premature wrinkles than they are about preventing cancer,” the AAD said.
The AAD is considering the creation of a social media quiz or interactive tool, and if the results of this survey were recast as a potential “Knowledge and Awareness Quiz” and graded with a traditional scheme (A = 90%-100%, B = 80%-89%, etc.), then 34% of the respondents would have failed, 15% would have gotten a D, and only 5% would have earned an A, the academy noted.
commissioned by the American Academy of Dermatology.

With the pandemic seemingly behind it, the United States enters the summer months facing the paradox of sun protection. Four out of five adults know that sunscreen should be reapplied every 2 hours when they’re outdoors, but only one in three make the actual effort, and 77% are likely to use sunscreen at the beach or a pool, compared with 41% when they’re gardening or working outside on their homes, the AAD reported.
“These findings are surprising and seem to suggest that many people do not take skin cancer seriously or perhaps believe skin cancer won’t happen to them,” Robert T. Brodell, MD, professor of dermatology at the University of Mississippi Medical Center, Jackson, said in a written statement from the AAD, adding that “unprotected exposure to ultraviolet rays is the most preventable risk factor for skin cancer, including melanoma.”
A quarter of all survey respondents reported getting sunburned in 2020, with the youngest adults most likely to feel the wrath of the sun. Sunburn was reported by 43% of those aged 18-23 years, 37% of those aged 24-39, 25% of the 40- to 55-year-olds, 12% of the 56- to 74-year-olds, and 7% of those aged 75 and older. More than a quarter of those who got sunburned said that it was bad enough to make their clothes feel uncomfortable, the academy said.
“Americans see the damaging effects of the sun on their skin as they get older, and two out of three look back and wish they had been more careful. But when it comes to cancer, specifically, most feel unconcerned in spite of their own risk,” according to a statement from Versta Research, which conducted the poll on behalf of the AAD. The survey was conducted from Feb. 22 to March 10, 2021, and involved 1,056 respondents, with a ±3% margin of error.
The lack of concern for skin cancer looks like this: More than two-thirds of the respondents (69%) have at least one possible risk factor – lighter skin tone, blue or green eyes, more than 50 moles, family history – but only 36% expressed concern about developing it. “Indeed, half of survey respondents (49%) say they are more worried about avoiding sunburn than they are about preventing skin cancer, and a third (32%) are more worried about avoiding premature wrinkles than they are about preventing cancer,” the AAD said.
The AAD is considering the creation of a social media quiz or interactive tool, and if the results of this survey were recast as a potential “Knowledge and Awareness Quiz” and graded with a traditional scheme (A = 90%-100%, B = 80%-89%, etc.), then 34% of the respondents would have failed, 15% would have gotten a D, and only 5% would have earned an A, the academy noted.
commissioned by the American Academy of Dermatology.

With the pandemic seemingly behind it, the United States enters the summer months facing the paradox of sun protection. Four out of five adults know that sunscreen should be reapplied every 2 hours when they’re outdoors, but only one in three make the actual effort, and 77% are likely to use sunscreen at the beach or a pool, compared with 41% when they’re gardening or working outside on their homes, the AAD reported.
“These findings are surprising and seem to suggest that many people do not take skin cancer seriously or perhaps believe skin cancer won’t happen to them,” Robert T. Brodell, MD, professor of dermatology at the University of Mississippi Medical Center, Jackson, said in a written statement from the AAD, adding that “unprotected exposure to ultraviolet rays is the most preventable risk factor for skin cancer, including melanoma.”
A quarter of all survey respondents reported getting sunburned in 2020, with the youngest adults most likely to feel the wrath of the sun. Sunburn was reported by 43% of those aged 18-23 years, 37% of those aged 24-39, 25% of the 40- to 55-year-olds, 12% of the 56- to 74-year-olds, and 7% of those aged 75 and older. More than a quarter of those who got sunburned said that it was bad enough to make their clothes feel uncomfortable, the academy said.
“Americans see the damaging effects of the sun on their skin as they get older, and two out of three look back and wish they had been more careful. But when it comes to cancer, specifically, most feel unconcerned in spite of their own risk,” according to a statement from Versta Research, which conducted the poll on behalf of the AAD. The survey was conducted from Feb. 22 to March 10, 2021, and involved 1,056 respondents, with a ±3% margin of error.
The lack of concern for skin cancer looks like this: More than two-thirds of the respondents (69%) have at least one possible risk factor – lighter skin tone, blue or green eyes, more than 50 moles, family history – but only 36% expressed concern about developing it. “Indeed, half of survey respondents (49%) say they are more worried about avoiding sunburn than they are about preventing skin cancer, and a third (32%) are more worried about avoiding premature wrinkles than they are about preventing cancer,” the AAD said.
The AAD is considering the creation of a social media quiz or interactive tool, and if the results of this survey were recast as a potential “Knowledge and Awareness Quiz” and graded with a traditional scheme (A = 90%-100%, B = 80%-89%, etc.), then 34% of the respondents would have failed, 15% would have gotten a D, and only 5% would have earned an A, the academy noted.
Freezing breast cancer to death avoids surgery: Why not further along?
In the United States, cryoablation or freezing tissue to death is a primary treatment option for a variety of cancers, including those originating in or spread to the bone, cervix, eye, kidney, liver, lung, pancreas, prostate, and skin.
Cryoablation for prostate cancer, one of the most common cancers in men, was first approved in the 1990s.
But unlike in Europe, this nonsurgical approach is not approved for breast cancer in the United States; it is one of the most common cancers in women.
So why is this approach still experimental for breast cancer?
“I don’t know,” answered cryoablation researcher Richard Fine, MD, of West Cancer Center in Germantown, Tenn., when asked by this news organization.
“It’s very interesting how slow the [Food and Drug Administration] is in approving devices for breast cancer [when compared with] other cancers,” he said.
New clinical data
Perhaps new clinical data will eventually lead to approval of this nonsurgical technique for use in low-risk breast cancer. However, the related trial had a controversial design that might discourage uptake by practitioners if it is approved, said an expert not involved in the study.
Nevertheless, the new data show that cryoablation can be an effective treatment for small, low-risk, early-stage breast cancers in older patients.
The findings come from ICE-3, a multicenter single-arm study of cryoablation in 194 such patients with mean follow-up of roughly 3 years.
It used liquid nitrogen-based cryoablation technology from IceCure Medical Ltd., an Israeli company and the study sponsor.
The results show that 2.06% (n = 4) of patients had a recurrence in the same breast, which is “basically the same” as lumpectomy, the surgical standard for this patient group, said Dr. Fine, the lead investigator on the trial.
These are interim data, Dr. Fine said at the American Society of Breast Surgeons annual meeting, held virtually.
The primary outcome is the 5-year recurrence rate, and this is the first-ever cryoablation trial that does not involve follow-up surgery, he said.
Cryoablation, which delivers a gas to a tumor via a thin needle-like probe that is guided by ultrasound, has multiple advantages over surgery, Dr. Fine said.
“The noninvasive procedure is fast, painless, and can be delivered under local anesthesia in a doctor’s office. Recovery time is minimal and cosmetic outcomes are excellent with little loss of breast tissue and no scarring,” he said in a meeting press statement.
The potential market for cryoablation in breast cancer is large, as it is intended for tumors ≤1.5 cm, which comprise approximately 60%-70% of stage 1 breast cancers that are hormone receptor–positive (HR+), and HER2-negative (HER2–), Dr. Fine said in an interview.
Cryoablation is part of a logical, de-escalation of breast cancer care, he added. “We have moved from radical mastectomy to modified mastectomy to lumpectomy – so the next step in that evolution is ablative technology, which is ‘nonsurgical.’ ”
There are other experimental ablative treatments for breast cancer including high-frequency ultrasound and laser, but cryoablation is the furthest along in development.
Cryoablation as a primary cancer treatment was first approved for coverage by the Centers for Medicare & Medicaid Services for localized prostate cancer in 1999.
But the concept extends back to 1845, when English physician James Arnott first used iced salt solutions (about –20 °C or – 4 °F) to induce tissue necrosis, reducing tumor size and ameliorating pain. Because the crude cryogen needed to be applied topically, the pioneering technique was limited to breast and cervical cancers because of their accessibility.
Not likely to show superiority
The new study’s population was composed of women aged 60 years or older (mean of 75 years) with unifocal invasive ductal cancers measuring ≤1.5 cm or less that were all low-grade, HR+, and HER2–, as noted.
The liquid nitrogen–based cryoablation consisted of a freeze-thaw-freeze cycle that totals 20-40 minutes, with freezing temperatures targeting the tumor area and turning it into an “ice ball.”
That ice ball eventually surrounds the tumor, creating a “lethal zone,” and thus a margin in which no cancer exists, akin to surgery, said Dr. Fine.
There were no significant device-related adverse events or complications reported, say the investigators. Most of the adverse events were minor and included bruising, localized edema, minor skin freeze burn, rash, minor bleeding from needle insertion, minor local hematoma, skin induration, minor infection, and pruritis.
Two of 15 patients who underwent sentinel lymph node biopsies had a positive sentinel node. At the discretion of their treating physician, 27 patients underwent adjuvant radiation, 1 patient received chemotherapy, and 148 began endocrine therapy. More than 95% of the patients and 98% of physicians reported satisfaction from the cosmetic results during follow-up visits.
Because not all patients underwent sentinel lymph node biopsy and adjuvant radiation, there is likely to be controversy about this approach, suggested Deanna J. Attai, MD, a breast surgeon at the University of California, Los Angeles, and past president of the American Society of Breast Surgeons, who was asked for comment.
“We have studies that [indicate that] these treatments don’t add significant benefit [in this patient population] but there still is this hesitation [to forgo them],” she told this news organization.
“The patients in this study were exceedingly low risk,” she emphasized.
“Is 5 years enough to assess recurrence rates? The answer is probably no. Recurrences or distant metastases are more likely to happen 10-20 years later.”
Thus, it will be difficult to show that cryoablation is superior to surgery, she said.
“You can show that cryoablation is not inferior to lumpectomy alone – which allows patients to avoid the operating room,” Dr. Attai summarized.
The surgical mindset and breast cancer
Dr. Attai, who was not involved in the current trial, was an investigator in an earlier single-arm cooperative group study of cryoablation for breast cancer, which had the rate of complete tumor ablation as the primary outcome. The study, known as the American College of Surgeons Oncology Group Z1072 trial, enrolled 99 patients, all of whom underwent ablation followed by surgery. The study reported results in 2014 but was very slow to develop, she observed.
“I did my first training in 2004 and I don’t think the study opened for several years after that. I think there’s been a lot of hesitation to change the mindset that every cancer needs to be removed surgically,” Dr. Attai stated.
“When you put breast cancer in the context of the other organs, we are lagging behind a bit [with cryoablation],” she added.
“I don’t want to go there but … the innovation for male diseases and procedures sometimes surpasses that of women’s diseases,” she said.
But she also defended her fellow practitioners. “There’s been tremendous changes in management over the 27 years I’ve been in practice,” she said, citing the movement from mastectomy to lumpectomy as one of multiple big changes.
The disparity between the development of cryoablation for breast and prostate cancer is a mystery when you contemplate the potential side effects, Dr. Fine observed. “There’s not a lot of vital structures inside the breast, so you don’t have risks that you have with the prostate, including urinary incontinence and impotence.”
As a next move, the American Society of Breast Surgeons is planning to establish a cryoablation registry and aims to enroll 50 sites and 500 patients who are aged 55-85 years; for those aged 65-70, radiation therapy will be required, said Dr. Fine.
Currently, cryoablation for breast cancer is allowed only in a clinical trial, so a registry would expand usage considerably, he said.
However, cryoablation, including from IceCure, has FDA clearance for ablating cancerous tissue in general (but not breast cancer specifically).
Dr. Attai hopes the field is ready for the nonsurgical approach.
“Halsted died in 1922 and the Halsted radical mastectomy really didn’t start to fall out of favor until the 1950s, 1960,” said Dr. Attai, referring to Dr William Halsted, who pioneered the procedure in the 1890s. “I would hope we are better at speeding up our progress. Changing the surgical mindset takes time,” she said.
Dr. Fine was an investigator in the ICE3 trial, which is funded by IceCure Medical. Dr. Attai has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the United States, cryoablation or freezing tissue to death is a primary treatment option for a variety of cancers, including those originating in or spread to the bone, cervix, eye, kidney, liver, lung, pancreas, prostate, and skin.
Cryoablation for prostate cancer, one of the most common cancers in men, was first approved in the 1990s.
But unlike in Europe, this nonsurgical approach is not approved for breast cancer in the United States; it is one of the most common cancers in women.
So why is this approach still experimental for breast cancer?
“I don’t know,” answered cryoablation researcher Richard Fine, MD, of West Cancer Center in Germantown, Tenn., when asked by this news organization.
“It’s very interesting how slow the [Food and Drug Administration] is in approving devices for breast cancer [when compared with] other cancers,” he said.
New clinical data
Perhaps new clinical data will eventually lead to approval of this nonsurgical technique for use in low-risk breast cancer. However, the related trial had a controversial design that might discourage uptake by practitioners if it is approved, said an expert not involved in the study.
Nevertheless, the new data show that cryoablation can be an effective treatment for small, low-risk, early-stage breast cancers in older patients.
The findings come from ICE-3, a multicenter single-arm study of cryoablation in 194 such patients with mean follow-up of roughly 3 years.
It used liquid nitrogen-based cryoablation technology from IceCure Medical Ltd., an Israeli company and the study sponsor.
The results show that 2.06% (n = 4) of patients had a recurrence in the same breast, which is “basically the same” as lumpectomy, the surgical standard for this patient group, said Dr. Fine, the lead investigator on the trial.
These are interim data, Dr. Fine said at the American Society of Breast Surgeons annual meeting, held virtually.
The primary outcome is the 5-year recurrence rate, and this is the first-ever cryoablation trial that does not involve follow-up surgery, he said.
Cryoablation, which delivers a gas to a tumor via a thin needle-like probe that is guided by ultrasound, has multiple advantages over surgery, Dr. Fine said.
“The noninvasive procedure is fast, painless, and can be delivered under local anesthesia in a doctor’s office. Recovery time is minimal and cosmetic outcomes are excellent with little loss of breast tissue and no scarring,” he said in a meeting press statement.
The potential market for cryoablation in breast cancer is large, as it is intended for tumors ≤1.5 cm, which comprise approximately 60%-70% of stage 1 breast cancers that are hormone receptor–positive (HR+), and HER2-negative (HER2–), Dr. Fine said in an interview.
Cryoablation is part of a logical, de-escalation of breast cancer care, he added. “We have moved from radical mastectomy to modified mastectomy to lumpectomy – so the next step in that evolution is ablative technology, which is ‘nonsurgical.’ ”
There are other experimental ablative treatments for breast cancer including high-frequency ultrasound and laser, but cryoablation is the furthest along in development.
Cryoablation as a primary cancer treatment was first approved for coverage by the Centers for Medicare & Medicaid Services for localized prostate cancer in 1999.
But the concept extends back to 1845, when English physician James Arnott first used iced salt solutions (about –20 °C or – 4 °F) to induce tissue necrosis, reducing tumor size and ameliorating pain. Because the crude cryogen needed to be applied topically, the pioneering technique was limited to breast and cervical cancers because of their accessibility.
Not likely to show superiority
The new study’s population was composed of women aged 60 years or older (mean of 75 years) with unifocal invasive ductal cancers measuring ≤1.5 cm or less that were all low-grade, HR+, and HER2–, as noted.
The liquid nitrogen–based cryoablation consisted of a freeze-thaw-freeze cycle that totals 20-40 minutes, with freezing temperatures targeting the tumor area and turning it into an “ice ball.”
That ice ball eventually surrounds the tumor, creating a “lethal zone,” and thus a margin in which no cancer exists, akin to surgery, said Dr. Fine.
There were no significant device-related adverse events or complications reported, say the investigators. Most of the adverse events were minor and included bruising, localized edema, minor skin freeze burn, rash, minor bleeding from needle insertion, minor local hematoma, skin induration, minor infection, and pruritis.
Two of 15 patients who underwent sentinel lymph node biopsies had a positive sentinel node. At the discretion of their treating physician, 27 patients underwent adjuvant radiation, 1 patient received chemotherapy, and 148 began endocrine therapy. More than 95% of the patients and 98% of physicians reported satisfaction from the cosmetic results during follow-up visits.
Because not all patients underwent sentinel lymph node biopsy and adjuvant radiation, there is likely to be controversy about this approach, suggested Deanna J. Attai, MD, a breast surgeon at the University of California, Los Angeles, and past president of the American Society of Breast Surgeons, who was asked for comment.
“We have studies that [indicate that] these treatments don’t add significant benefit [in this patient population] but there still is this hesitation [to forgo them],” she told this news organization.
“The patients in this study were exceedingly low risk,” she emphasized.
“Is 5 years enough to assess recurrence rates? The answer is probably no. Recurrences or distant metastases are more likely to happen 10-20 years later.”
Thus, it will be difficult to show that cryoablation is superior to surgery, she said.
“You can show that cryoablation is not inferior to lumpectomy alone – which allows patients to avoid the operating room,” Dr. Attai summarized.
The surgical mindset and breast cancer
Dr. Attai, who was not involved in the current trial, was an investigator in an earlier single-arm cooperative group study of cryoablation for breast cancer, which had the rate of complete tumor ablation as the primary outcome. The study, known as the American College of Surgeons Oncology Group Z1072 trial, enrolled 99 patients, all of whom underwent ablation followed by surgery. The study reported results in 2014 but was very slow to develop, she observed.
“I did my first training in 2004 and I don’t think the study opened for several years after that. I think there’s been a lot of hesitation to change the mindset that every cancer needs to be removed surgically,” Dr. Attai stated.
“When you put breast cancer in the context of the other organs, we are lagging behind a bit [with cryoablation],” she added.
“I don’t want to go there but … the innovation for male diseases and procedures sometimes surpasses that of women’s diseases,” she said.
But she also defended her fellow practitioners. “There’s been tremendous changes in management over the 27 years I’ve been in practice,” she said, citing the movement from mastectomy to lumpectomy as one of multiple big changes.
The disparity between the development of cryoablation for breast and prostate cancer is a mystery when you contemplate the potential side effects, Dr. Fine observed. “There’s not a lot of vital structures inside the breast, so you don’t have risks that you have with the prostate, including urinary incontinence and impotence.”
As a next move, the American Society of Breast Surgeons is planning to establish a cryoablation registry and aims to enroll 50 sites and 500 patients who are aged 55-85 years; for those aged 65-70, radiation therapy will be required, said Dr. Fine.
Currently, cryoablation for breast cancer is allowed only in a clinical trial, so a registry would expand usage considerably, he said.
However, cryoablation, including from IceCure, has FDA clearance for ablating cancerous tissue in general (but not breast cancer specifically).
Dr. Attai hopes the field is ready for the nonsurgical approach.
“Halsted died in 1922 and the Halsted radical mastectomy really didn’t start to fall out of favor until the 1950s, 1960,” said Dr. Attai, referring to Dr William Halsted, who pioneered the procedure in the 1890s. “I would hope we are better at speeding up our progress. Changing the surgical mindset takes time,” she said.
Dr. Fine was an investigator in the ICE3 trial, which is funded by IceCure Medical. Dr. Attai has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the United States, cryoablation or freezing tissue to death is a primary treatment option for a variety of cancers, including those originating in or spread to the bone, cervix, eye, kidney, liver, lung, pancreas, prostate, and skin.
Cryoablation for prostate cancer, one of the most common cancers in men, was first approved in the 1990s.
But unlike in Europe, this nonsurgical approach is not approved for breast cancer in the United States; it is one of the most common cancers in women.
So why is this approach still experimental for breast cancer?
“I don’t know,” answered cryoablation researcher Richard Fine, MD, of West Cancer Center in Germantown, Tenn., when asked by this news organization.
“It’s very interesting how slow the [Food and Drug Administration] is in approving devices for breast cancer [when compared with] other cancers,” he said.
New clinical data
Perhaps new clinical data will eventually lead to approval of this nonsurgical technique for use in low-risk breast cancer. However, the related trial had a controversial design that might discourage uptake by practitioners if it is approved, said an expert not involved in the study.
Nevertheless, the new data show that cryoablation can be an effective treatment for small, low-risk, early-stage breast cancers in older patients.
The findings come from ICE-3, a multicenter single-arm study of cryoablation in 194 such patients with mean follow-up of roughly 3 years.
It used liquid nitrogen-based cryoablation technology from IceCure Medical Ltd., an Israeli company and the study sponsor.
The results show that 2.06% (n = 4) of patients had a recurrence in the same breast, which is “basically the same” as lumpectomy, the surgical standard for this patient group, said Dr. Fine, the lead investigator on the trial.
These are interim data, Dr. Fine said at the American Society of Breast Surgeons annual meeting, held virtually.
The primary outcome is the 5-year recurrence rate, and this is the first-ever cryoablation trial that does not involve follow-up surgery, he said.
Cryoablation, which delivers a gas to a tumor via a thin needle-like probe that is guided by ultrasound, has multiple advantages over surgery, Dr. Fine said.
“The noninvasive procedure is fast, painless, and can be delivered under local anesthesia in a doctor’s office. Recovery time is minimal and cosmetic outcomes are excellent with little loss of breast tissue and no scarring,” he said in a meeting press statement.
The potential market for cryoablation in breast cancer is large, as it is intended for tumors ≤1.5 cm, which comprise approximately 60%-70% of stage 1 breast cancers that are hormone receptor–positive (HR+), and HER2-negative (HER2–), Dr. Fine said in an interview.
Cryoablation is part of a logical, de-escalation of breast cancer care, he added. “We have moved from radical mastectomy to modified mastectomy to lumpectomy – so the next step in that evolution is ablative technology, which is ‘nonsurgical.’ ”
There are other experimental ablative treatments for breast cancer including high-frequency ultrasound and laser, but cryoablation is the furthest along in development.
Cryoablation as a primary cancer treatment was first approved for coverage by the Centers for Medicare & Medicaid Services for localized prostate cancer in 1999.
But the concept extends back to 1845, when English physician James Arnott first used iced salt solutions (about –20 °C or – 4 °F) to induce tissue necrosis, reducing tumor size and ameliorating pain. Because the crude cryogen needed to be applied topically, the pioneering technique was limited to breast and cervical cancers because of their accessibility.
Not likely to show superiority
The new study’s population was composed of women aged 60 years or older (mean of 75 years) with unifocal invasive ductal cancers measuring ≤1.5 cm or less that were all low-grade, HR+, and HER2–, as noted.
The liquid nitrogen–based cryoablation consisted of a freeze-thaw-freeze cycle that totals 20-40 minutes, with freezing temperatures targeting the tumor area and turning it into an “ice ball.”
That ice ball eventually surrounds the tumor, creating a “lethal zone,” and thus a margin in which no cancer exists, akin to surgery, said Dr. Fine.
There were no significant device-related adverse events or complications reported, say the investigators. Most of the adverse events were minor and included bruising, localized edema, minor skin freeze burn, rash, minor bleeding from needle insertion, minor local hematoma, skin induration, minor infection, and pruritis.
Two of 15 patients who underwent sentinel lymph node biopsies had a positive sentinel node. At the discretion of their treating physician, 27 patients underwent adjuvant radiation, 1 patient received chemotherapy, and 148 began endocrine therapy. More than 95% of the patients and 98% of physicians reported satisfaction from the cosmetic results during follow-up visits.
Because not all patients underwent sentinel lymph node biopsy and adjuvant radiation, there is likely to be controversy about this approach, suggested Deanna J. Attai, MD, a breast surgeon at the University of California, Los Angeles, and past president of the American Society of Breast Surgeons, who was asked for comment.
“We have studies that [indicate that] these treatments don’t add significant benefit [in this patient population] but there still is this hesitation [to forgo them],” she told this news organization.
“The patients in this study were exceedingly low risk,” she emphasized.
“Is 5 years enough to assess recurrence rates? The answer is probably no. Recurrences or distant metastases are more likely to happen 10-20 years later.”
Thus, it will be difficult to show that cryoablation is superior to surgery, she said.
“You can show that cryoablation is not inferior to lumpectomy alone – which allows patients to avoid the operating room,” Dr. Attai summarized.
The surgical mindset and breast cancer
Dr. Attai, who was not involved in the current trial, was an investigator in an earlier single-arm cooperative group study of cryoablation for breast cancer, which had the rate of complete tumor ablation as the primary outcome. The study, known as the American College of Surgeons Oncology Group Z1072 trial, enrolled 99 patients, all of whom underwent ablation followed by surgery. The study reported results in 2014 but was very slow to develop, she observed.
“I did my first training in 2004 and I don’t think the study opened for several years after that. I think there’s been a lot of hesitation to change the mindset that every cancer needs to be removed surgically,” Dr. Attai stated.
“When you put breast cancer in the context of the other organs, we are lagging behind a bit [with cryoablation],” she added.
“I don’t want to go there but … the innovation for male diseases and procedures sometimes surpasses that of women’s diseases,” she said.
But she also defended her fellow practitioners. “There’s been tremendous changes in management over the 27 years I’ve been in practice,” she said, citing the movement from mastectomy to lumpectomy as one of multiple big changes.
The disparity between the development of cryoablation for breast and prostate cancer is a mystery when you contemplate the potential side effects, Dr. Fine observed. “There’s not a lot of vital structures inside the breast, so you don’t have risks that you have with the prostate, including urinary incontinence and impotence.”
As a next move, the American Society of Breast Surgeons is planning to establish a cryoablation registry and aims to enroll 50 sites and 500 patients who are aged 55-85 years; for those aged 65-70, radiation therapy will be required, said Dr. Fine.
Currently, cryoablation for breast cancer is allowed only in a clinical trial, so a registry would expand usage considerably, he said.
However, cryoablation, including from IceCure, has FDA clearance for ablating cancerous tissue in general (but not breast cancer specifically).
Dr. Attai hopes the field is ready for the nonsurgical approach.
“Halsted died in 1922 and the Halsted radical mastectomy really didn’t start to fall out of favor until the 1950s, 1960,” said Dr. Attai, referring to Dr William Halsted, who pioneered the procedure in the 1890s. “I would hope we are better at speeding up our progress. Changing the surgical mindset takes time,” she said.
Dr. Fine was an investigator in the ICE3 trial, which is funded by IceCure Medical. Dr. Attai has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘Remarkable’ results for targeted therapy of rare CNS tumors
The results from three small studies of targeted therapy for rare brain tumors were “remarkable,” according to Jaishri Blakeley, MD, a neurology professor at Johns Hopkins Medicine, Baltimore, who discussed the studies after they were presented at the American Society of Clinical Oncology meeting.
Although most patients don’t have targetable mutations, molecular testing “is well worth the effort,” for those that do. “I think it’s fair to say that precision medicine” – well established in other tumor types – “is finally here in full force for neuro-oncology,” Dr. Blakeley said.
A promising start
Fifteen of 16 patients (94%) in one study had newly diagnosed and untreated papillary craniopharyngiomas (PCPs) that harbored BRAF V600E mutations, a common finding in PCPs, which have no effective medical treatment.
Tumors shrunk 68%-99% in 14 patients (93%) after treatment with the BRAF inhibitor vemurafenib plus the MEK inhibitor cobimetinib, which was included to stave off resistance to vemurafenib. The 24-month progression free survival was 93%.
The combination resulted in significant response in all patients who received at least one cycle of therapy, with a median 91% volume reduction. “Our study indicates that BRAF/MEK inhibitors could be a powerful tool in the treatment of previously untreated PCP, with the potential to avoid the morbidity associated with radiation and surgery,” concluded lead investigator and presenter Priscilla K. Brastianos, MD, associate professor of medicine at Mass General Cancer Center, Boston.
Thirty-three people in the second study had a mix of high and low grade gliomas or other CNS tumors positive for TRK gene fusions, a known oncogenic driver; the majority were children. They were treated with the TRK inhibitor larotrectinib after progressing on other systemic therapies.
The objective response rate was 30%, and the disease control rate was 73% at 24 weeks, with a median time to best response of 1.9 months. Tumors shrank in 82% of evaluable patients. Median progression-free survival was 18.3 months, and overall survival was not reached.
“These results support testing for TRK gene fusions for all patients with CNS tumors, especially if there is no known driver and especially in infants,” concluded lead investigator and presenter Sebastien Perreault, MD, a clinical assistant neurosciences professor at the University of Montreal.
The third study tested ALK inhibitors such as crizotinib in seven patients with adult-onset neuroblastoma, a rare and almost invariably fatal tumor known to be enriched for ALK mutations; the subjects were positive for them.
Their disease remained stable anywhere from 3.4 to 37.4 months. Median time to progression was 15.5 months, and median overall survival was 46.5 months.
ALK inhibitors “can be a well-tolerated options for treatment, improving time to progression. Development of resistance to one agent does not preclude use of other agents in the same drug class. ALK inhibitors should be considered when treating patients with this diagnosis,” said lead investigator and presenter Jessica Stiefel, MD, a pediatric hematology oncology fellow at Memorial Sloan Kettering Cancer Center, New York.
A ‘strong’ recommendation
The data “are great news” across the board. Targeted therapy applied to the right CNS tumor can have “dramatic” benefit for tumor control, Dr. Blakeley said.
But organizing molecular testing is not straightforward and requires strategies to balance “the use of precious resources, such as time money, and tissue,” with the potential benefit. Interpretation of testing results isn’t straightforward either, and is best handled by a molecular tumor board. Clinical pharmacists are also key to accessing expensive medications off label for CNS tumors.
Adverse events are also a consideration. Most of the subjects in the PCP study had grade 3/4 toxicity. Three patients in the ALK inhibitor study had to stop because of adverse events. Almost 40% on larotrectinib had grade 3 or 4 toxicity; nobody came off treatment, but a third had to skip doses.
Once an actionable mutation is identified, Dr. Blakeley’s “strong recommendation” is to enroll patients in a clinical trial that targets it, to take advantage the structure already in place to secure treatment, managed patients, and assess outcomes.
The National Cancer Institute’s MATCH trial is one of several options.
The BRAF/MEK inhibitor study was funded by Genentech and the National Institutes of Health. Dr. Brastianos had ties to numerous companies, including Pfizer, Lilly, and Merck. The TRK inhibitor study was funded by Bayer/Lilly. Dr. Perreault is a speaker and researcher for the company and has other ties. Dr. Blakeley is an adviser and/or researcher for a number of companies, including AbbVie, Astellas, BMS, and Exelixis. Dr. Stiefel didn’t have any disclosures, and didn’t report outside funding.
The results from three small studies of targeted therapy for rare brain tumors were “remarkable,” according to Jaishri Blakeley, MD, a neurology professor at Johns Hopkins Medicine, Baltimore, who discussed the studies after they were presented at the American Society of Clinical Oncology meeting.
Although most patients don’t have targetable mutations, molecular testing “is well worth the effort,” for those that do. “I think it’s fair to say that precision medicine” – well established in other tumor types – “is finally here in full force for neuro-oncology,” Dr. Blakeley said.
A promising start
Fifteen of 16 patients (94%) in one study had newly diagnosed and untreated papillary craniopharyngiomas (PCPs) that harbored BRAF V600E mutations, a common finding in PCPs, which have no effective medical treatment.
Tumors shrunk 68%-99% in 14 patients (93%) after treatment with the BRAF inhibitor vemurafenib plus the MEK inhibitor cobimetinib, which was included to stave off resistance to vemurafenib. The 24-month progression free survival was 93%.
The combination resulted in significant response in all patients who received at least one cycle of therapy, with a median 91% volume reduction. “Our study indicates that BRAF/MEK inhibitors could be a powerful tool in the treatment of previously untreated PCP, with the potential to avoid the morbidity associated with radiation and surgery,” concluded lead investigator and presenter Priscilla K. Brastianos, MD, associate professor of medicine at Mass General Cancer Center, Boston.
Thirty-three people in the second study had a mix of high and low grade gliomas or other CNS tumors positive for TRK gene fusions, a known oncogenic driver; the majority were children. They were treated with the TRK inhibitor larotrectinib after progressing on other systemic therapies.
The objective response rate was 30%, and the disease control rate was 73% at 24 weeks, with a median time to best response of 1.9 months. Tumors shrank in 82% of evaluable patients. Median progression-free survival was 18.3 months, and overall survival was not reached.
“These results support testing for TRK gene fusions for all patients with CNS tumors, especially if there is no known driver and especially in infants,” concluded lead investigator and presenter Sebastien Perreault, MD, a clinical assistant neurosciences professor at the University of Montreal.
The third study tested ALK inhibitors such as crizotinib in seven patients with adult-onset neuroblastoma, a rare and almost invariably fatal tumor known to be enriched for ALK mutations; the subjects were positive for them.
Their disease remained stable anywhere from 3.4 to 37.4 months. Median time to progression was 15.5 months, and median overall survival was 46.5 months.
ALK inhibitors “can be a well-tolerated options for treatment, improving time to progression. Development of resistance to one agent does not preclude use of other agents in the same drug class. ALK inhibitors should be considered when treating patients with this diagnosis,” said lead investigator and presenter Jessica Stiefel, MD, a pediatric hematology oncology fellow at Memorial Sloan Kettering Cancer Center, New York.
A ‘strong’ recommendation
The data “are great news” across the board. Targeted therapy applied to the right CNS tumor can have “dramatic” benefit for tumor control, Dr. Blakeley said.
But organizing molecular testing is not straightforward and requires strategies to balance “the use of precious resources, such as time money, and tissue,” with the potential benefit. Interpretation of testing results isn’t straightforward either, and is best handled by a molecular tumor board. Clinical pharmacists are also key to accessing expensive medications off label for CNS tumors.
Adverse events are also a consideration. Most of the subjects in the PCP study had grade 3/4 toxicity. Three patients in the ALK inhibitor study had to stop because of adverse events. Almost 40% on larotrectinib had grade 3 or 4 toxicity; nobody came off treatment, but a third had to skip doses.
Once an actionable mutation is identified, Dr. Blakeley’s “strong recommendation” is to enroll patients in a clinical trial that targets it, to take advantage the structure already in place to secure treatment, managed patients, and assess outcomes.
The National Cancer Institute’s MATCH trial is one of several options.
The BRAF/MEK inhibitor study was funded by Genentech and the National Institutes of Health. Dr. Brastianos had ties to numerous companies, including Pfizer, Lilly, and Merck. The TRK inhibitor study was funded by Bayer/Lilly. Dr. Perreault is a speaker and researcher for the company and has other ties. Dr. Blakeley is an adviser and/or researcher for a number of companies, including AbbVie, Astellas, BMS, and Exelixis. Dr. Stiefel didn’t have any disclosures, and didn’t report outside funding.
The results from three small studies of targeted therapy for rare brain tumors were “remarkable,” according to Jaishri Blakeley, MD, a neurology professor at Johns Hopkins Medicine, Baltimore, who discussed the studies after they were presented at the American Society of Clinical Oncology meeting.
Although most patients don’t have targetable mutations, molecular testing “is well worth the effort,” for those that do. “I think it’s fair to say that precision medicine” – well established in other tumor types – “is finally here in full force for neuro-oncology,” Dr. Blakeley said.
A promising start
Fifteen of 16 patients (94%) in one study had newly diagnosed and untreated papillary craniopharyngiomas (PCPs) that harbored BRAF V600E mutations, a common finding in PCPs, which have no effective medical treatment.
Tumors shrunk 68%-99% in 14 patients (93%) after treatment with the BRAF inhibitor vemurafenib plus the MEK inhibitor cobimetinib, which was included to stave off resistance to vemurafenib. The 24-month progression free survival was 93%.
The combination resulted in significant response in all patients who received at least one cycle of therapy, with a median 91% volume reduction. “Our study indicates that BRAF/MEK inhibitors could be a powerful tool in the treatment of previously untreated PCP, with the potential to avoid the morbidity associated with radiation and surgery,” concluded lead investigator and presenter Priscilla K. Brastianos, MD, associate professor of medicine at Mass General Cancer Center, Boston.
Thirty-three people in the second study had a mix of high and low grade gliomas or other CNS tumors positive for TRK gene fusions, a known oncogenic driver; the majority were children. They were treated with the TRK inhibitor larotrectinib after progressing on other systemic therapies.
The objective response rate was 30%, and the disease control rate was 73% at 24 weeks, with a median time to best response of 1.9 months. Tumors shrank in 82% of evaluable patients. Median progression-free survival was 18.3 months, and overall survival was not reached.
“These results support testing for TRK gene fusions for all patients with CNS tumors, especially if there is no known driver and especially in infants,” concluded lead investigator and presenter Sebastien Perreault, MD, a clinical assistant neurosciences professor at the University of Montreal.
The third study tested ALK inhibitors such as crizotinib in seven patients with adult-onset neuroblastoma, a rare and almost invariably fatal tumor known to be enriched for ALK mutations; the subjects were positive for them.
Their disease remained stable anywhere from 3.4 to 37.4 months. Median time to progression was 15.5 months, and median overall survival was 46.5 months.
ALK inhibitors “can be a well-tolerated options for treatment, improving time to progression. Development of resistance to one agent does not preclude use of other agents in the same drug class. ALK inhibitors should be considered when treating patients with this diagnosis,” said lead investigator and presenter Jessica Stiefel, MD, a pediatric hematology oncology fellow at Memorial Sloan Kettering Cancer Center, New York.
A ‘strong’ recommendation
The data “are great news” across the board. Targeted therapy applied to the right CNS tumor can have “dramatic” benefit for tumor control, Dr. Blakeley said.
But organizing molecular testing is not straightforward and requires strategies to balance “the use of precious resources, such as time money, and tissue,” with the potential benefit. Interpretation of testing results isn’t straightforward either, and is best handled by a molecular tumor board. Clinical pharmacists are also key to accessing expensive medications off label for CNS tumors.
Adverse events are also a consideration. Most of the subjects in the PCP study had grade 3/4 toxicity. Three patients in the ALK inhibitor study had to stop because of adverse events. Almost 40% on larotrectinib had grade 3 or 4 toxicity; nobody came off treatment, but a third had to skip doses.
Once an actionable mutation is identified, Dr. Blakeley’s “strong recommendation” is to enroll patients in a clinical trial that targets it, to take advantage the structure already in place to secure treatment, managed patients, and assess outcomes.
The National Cancer Institute’s MATCH trial is one of several options.
The BRAF/MEK inhibitor study was funded by Genentech and the National Institutes of Health. Dr. Brastianos had ties to numerous companies, including Pfizer, Lilly, and Merck. The TRK inhibitor study was funded by Bayer/Lilly. Dr. Perreault is a speaker and researcher for the company and has other ties. Dr. Blakeley is an adviser and/or researcher for a number of companies, including AbbVie, Astellas, BMS, and Exelixis. Dr. Stiefel didn’t have any disclosures, and didn’t report outside funding.
FROM ASCO 2021
Experimental antibody-drug conjugate shown active against r/r DLBCL
Patients with relapsed or refractory B-cell non-Hodgkin lymphomas who are not candidates for hematopoietic stem cell transplant have a generally poor prognosis and few treatment options, but an experimental combination of the antibody-drug conjugate naratuximab with rituximab showed promising efficacy and acceptable safety in these patients in a phase 2 trial.
Among patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) the combination was associated with a 44.7% overall response rate, including 31.6% complete responses, and two-thirds of patients had responses lasting more than 12 months, reported Moshe Yair Levy, MD, from Texas Oncology–Baylor Charles A Sammons Cancer Center in Dallas.
“This is, in my viewpoint, very exciting therapy,” he said in a question-and-answer session following his presentation of the data in a late-breaking abstract session during the European Hematology Association annual congress. (Abstract LB1903).
Naratuximab emtansine is an investigational antibody-drug conjugate (ADC) consisting of a humanized monoclonal antibody against CD37, a surface marker on B lymphocytes that is highly expressed in non-Hodgkin lymphoma (NHL), conjugated to a cytotoxic derivative of maitansine.
CD37 is also an internalizable cell-surface antigen, making it an attractive candidate for an ADC approach.
In a phase 1 trial, naratuximab monotherapy showed a good safety profile and a 22% overall response rate, Dr. Levy noted.
“What they found is that, if you coadminister this ADC with rituximab, you’re actually going to get more internalization of the CD37 monoclonal, therefore more payload delivered to your target cells,” he said.
He reported results of a multicenter, adaptive phase 2 study of the combination in patients with DLBCL and other relapsed/refractory NHL.
DLBCL and others
The trial was divided into two parts, with the first consisting of a safety run-in phase with expansion in patients with confirmed diagnoses of relapsed/refractory NHL, including DLBCL, follicular lymphoma, mantle cell lymphoma, and marginal zone lymphoma.
Patients with double- or triple-hit disease (with translocations in MYC plus either BCL2 and/or BCL6), bulky disease, or transformed lymphoma were eligible.
The second part consisted of two cohorts of patients with DLBCL treated with naratuximab and rituximab either weekly or every 3 weeks.
All patients in the study had received one to six prior lines of therapy, and had Eastern Cooperative Oncology Group performance status of 0-2. Patients with CNS lymphomas or prior anti-CD37 targeting therapy were excluded.
The safety population included 50 patients with DLBCL assigned to therapy every 3 weeks, 30 assigned to weekly therapy, and 20 patients with other NHL.
DLBCL efficacy
A total of 76 patients with DLBCL were evaluable for efficacy.
The ORR was 44% for patients in both the weekly and every 3 week cohorts, with 31.6% having complete responses.
Among 61 patients with nonbulky disease (longest diameter 7.5 cm or less), the ORR was 50.8%, and among 28 patients who had three or more prior lines of therapy the ORR was 46.4%, with 32.1% having a complete response.
Among responders followed for a median of 15 months, the median duration of response was not reached, and 66% had responses lasting beyond 12 months.
In the weekly dosing DLBCL cohort, 53.3% of patients discontinued treatment of both study drugs because of disease progression, as did 58% of those in the every 3 week cohort, and 30% of patients with other lymphomas. Only eight patients discontinued the combination because of treatment-emergent adverse events. Six patients had treatment-emergent adverse events leading to naratuximab dose reduction.
The most common grade 3 or 4 adverse events were neutropenia, leukopenia, lymphopenia and thrombocytopenias. Dr. Levy commented that the use of granulocyte colony-stimulating factor, which was not mandatory in the study, would likely have lowered the incidence of cytopenias.
There were 10 deaths during the study, 2 of which were considered to be treatment related, occurring in 1 patient each in the DLBCL dosing cohorts; 1 of the patients died from pneumonitis, and the other from left ventricular heart failure.
Other patients deaths were attributed to non–treatment-related cardiac arrest, acute renal failure, exacerbation of chronic heart failure, respiratory failure, multiorgan failure, lung infection, or colon adenocarcinoma.
Q 3 weeks suffices
In the question-and-answer session following the presentation, Kenny Lei, MD, from the Chinese University of Hong Kong asked Dr. Levy what the half-life of naratuximab is, and what was the investigator’s rationale for testing a weekly dosing schedule.
“I think the reason they checked the two different regimens, the Q week and the Q 3-week group, is that they noted that [naratuximab] was cleared relatively quickly, and they wanted to see whether or not, by giving Q weekly, when you get a continuous CD37 site occupancy if they would have a better outcome. But as you saw, in the groups there was really no clinically relevant difference in outcome,” Dr. Levy said.
Andrew Davies, MD, PhD, from the University of Southampton (England), asked whether the neutropenia seen in the study was related to myeloid expression of the target of from the off-target deconjugated payload.
“I don’t know that I necessarily have the answer to that,” Dr. Levy replied. “Remember there is the CD20 monoclonal rituximab which we know can cause neutropenia, as well as the CD37 and the target payload. I don’t know if we have enough information to attribute it to one specific component of the therapy,” he said.
The study was funded by Debiopharm International. Dr. Levy disclosed speaker activities for multiple companies, not including Debiopharm. Dr. Lei and Dr. Davies had no disclosures relevant to the study.
Patients with relapsed or refractory B-cell non-Hodgkin lymphomas who are not candidates for hematopoietic stem cell transplant have a generally poor prognosis and few treatment options, but an experimental combination of the antibody-drug conjugate naratuximab with rituximab showed promising efficacy and acceptable safety in these patients in a phase 2 trial.
Among patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) the combination was associated with a 44.7% overall response rate, including 31.6% complete responses, and two-thirds of patients had responses lasting more than 12 months, reported Moshe Yair Levy, MD, from Texas Oncology–Baylor Charles A Sammons Cancer Center in Dallas.
“This is, in my viewpoint, very exciting therapy,” he said in a question-and-answer session following his presentation of the data in a late-breaking abstract session during the European Hematology Association annual congress. (Abstract LB1903).
Naratuximab emtansine is an investigational antibody-drug conjugate (ADC) consisting of a humanized monoclonal antibody against CD37, a surface marker on B lymphocytes that is highly expressed in non-Hodgkin lymphoma (NHL), conjugated to a cytotoxic derivative of maitansine.
CD37 is also an internalizable cell-surface antigen, making it an attractive candidate for an ADC approach.
In a phase 1 trial, naratuximab monotherapy showed a good safety profile and a 22% overall response rate, Dr. Levy noted.
“What they found is that, if you coadminister this ADC with rituximab, you’re actually going to get more internalization of the CD37 monoclonal, therefore more payload delivered to your target cells,” he said.
He reported results of a multicenter, adaptive phase 2 study of the combination in patients with DLBCL and other relapsed/refractory NHL.
DLBCL and others
The trial was divided into two parts, with the first consisting of a safety run-in phase with expansion in patients with confirmed diagnoses of relapsed/refractory NHL, including DLBCL, follicular lymphoma, mantle cell lymphoma, and marginal zone lymphoma.
Patients with double- or triple-hit disease (with translocations in MYC plus either BCL2 and/or BCL6), bulky disease, or transformed lymphoma were eligible.
The second part consisted of two cohorts of patients with DLBCL treated with naratuximab and rituximab either weekly or every 3 weeks.
All patients in the study had received one to six prior lines of therapy, and had Eastern Cooperative Oncology Group performance status of 0-2. Patients with CNS lymphomas or prior anti-CD37 targeting therapy were excluded.
The safety population included 50 patients with DLBCL assigned to therapy every 3 weeks, 30 assigned to weekly therapy, and 20 patients with other NHL.
DLBCL efficacy
A total of 76 patients with DLBCL were evaluable for efficacy.
The ORR was 44% for patients in both the weekly and every 3 week cohorts, with 31.6% having complete responses.
Among 61 patients with nonbulky disease (longest diameter 7.5 cm or less), the ORR was 50.8%, and among 28 patients who had three or more prior lines of therapy the ORR was 46.4%, with 32.1% having a complete response.
Among responders followed for a median of 15 months, the median duration of response was not reached, and 66% had responses lasting beyond 12 months.
In the weekly dosing DLBCL cohort, 53.3% of patients discontinued treatment of both study drugs because of disease progression, as did 58% of those in the every 3 week cohort, and 30% of patients with other lymphomas. Only eight patients discontinued the combination because of treatment-emergent adverse events. Six patients had treatment-emergent adverse events leading to naratuximab dose reduction.
The most common grade 3 or 4 adverse events were neutropenia, leukopenia, lymphopenia and thrombocytopenias. Dr. Levy commented that the use of granulocyte colony-stimulating factor, which was not mandatory in the study, would likely have lowered the incidence of cytopenias.
There were 10 deaths during the study, 2 of which were considered to be treatment related, occurring in 1 patient each in the DLBCL dosing cohorts; 1 of the patients died from pneumonitis, and the other from left ventricular heart failure.
Other patients deaths were attributed to non–treatment-related cardiac arrest, acute renal failure, exacerbation of chronic heart failure, respiratory failure, multiorgan failure, lung infection, or colon adenocarcinoma.
Q 3 weeks suffices
In the question-and-answer session following the presentation, Kenny Lei, MD, from the Chinese University of Hong Kong asked Dr. Levy what the half-life of naratuximab is, and what was the investigator’s rationale for testing a weekly dosing schedule.
“I think the reason they checked the two different regimens, the Q week and the Q 3-week group, is that they noted that [naratuximab] was cleared relatively quickly, and they wanted to see whether or not, by giving Q weekly, when you get a continuous CD37 site occupancy if they would have a better outcome. But as you saw, in the groups there was really no clinically relevant difference in outcome,” Dr. Levy said.
Andrew Davies, MD, PhD, from the University of Southampton (England), asked whether the neutropenia seen in the study was related to myeloid expression of the target of from the off-target deconjugated payload.
“I don’t know that I necessarily have the answer to that,” Dr. Levy replied. “Remember there is the CD20 monoclonal rituximab which we know can cause neutropenia, as well as the CD37 and the target payload. I don’t know if we have enough information to attribute it to one specific component of the therapy,” he said.
The study was funded by Debiopharm International. Dr. Levy disclosed speaker activities for multiple companies, not including Debiopharm. Dr. Lei and Dr. Davies had no disclosures relevant to the study.
Patients with relapsed or refractory B-cell non-Hodgkin lymphomas who are not candidates for hematopoietic stem cell transplant have a generally poor prognosis and few treatment options, but an experimental combination of the antibody-drug conjugate naratuximab with rituximab showed promising efficacy and acceptable safety in these patients in a phase 2 trial.
Among patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) the combination was associated with a 44.7% overall response rate, including 31.6% complete responses, and two-thirds of patients had responses lasting more than 12 months, reported Moshe Yair Levy, MD, from Texas Oncology–Baylor Charles A Sammons Cancer Center in Dallas.
“This is, in my viewpoint, very exciting therapy,” he said in a question-and-answer session following his presentation of the data in a late-breaking abstract session during the European Hematology Association annual congress. (Abstract LB1903).
Naratuximab emtansine is an investigational antibody-drug conjugate (ADC) consisting of a humanized monoclonal antibody against CD37, a surface marker on B lymphocytes that is highly expressed in non-Hodgkin lymphoma (NHL), conjugated to a cytotoxic derivative of maitansine.
CD37 is also an internalizable cell-surface antigen, making it an attractive candidate for an ADC approach.
In a phase 1 trial, naratuximab monotherapy showed a good safety profile and a 22% overall response rate, Dr. Levy noted.
“What they found is that, if you coadminister this ADC with rituximab, you’re actually going to get more internalization of the CD37 monoclonal, therefore more payload delivered to your target cells,” he said.
He reported results of a multicenter, adaptive phase 2 study of the combination in patients with DLBCL and other relapsed/refractory NHL.
DLBCL and others
The trial was divided into two parts, with the first consisting of a safety run-in phase with expansion in patients with confirmed diagnoses of relapsed/refractory NHL, including DLBCL, follicular lymphoma, mantle cell lymphoma, and marginal zone lymphoma.
Patients with double- or triple-hit disease (with translocations in MYC plus either BCL2 and/or BCL6), bulky disease, or transformed lymphoma were eligible.
The second part consisted of two cohorts of patients with DLBCL treated with naratuximab and rituximab either weekly or every 3 weeks.
All patients in the study had received one to six prior lines of therapy, and had Eastern Cooperative Oncology Group performance status of 0-2. Patients with CNS lymphomas or prior anti-CD37 targeting therapy were excluded.
The safety population included 50 patients with DLBCL assigned to therapy every 3 weeks, 30 assigned to weekly therapy, and 20 patients with other NHL.
DLBCL efficacy
A total of 76 patients with DLBCL were evaluable for efficacy.
The ORR was 44% for patients in both the weekly and every 3 week cohorts, with 31.6% having complete responses.
Among 61 patients with nonbulky disease (longest diameter 7.5 cm or less), the ORR was 50.8%, and among 28 patients who had three or more prior lines of therapy the ORR was 46.4%, with 32.1% having a complete response.
Among responders followed for a median of 15 months, the median duration of response was not reached, and 66% had responses lasting beyond 12 months.
In the weekly dosing DLBCL cohort, 53.3% of patients discontinued treatment of both study drugs because of disease progression, as did 58% of those in the every 3 week cohort, and 30% of patients with other lymphomas. Only eight patients discontinued the combination because of treatment-emergent adverse events. Six patients had treatment-emergent adverse events leading to naratuximab dose reduction.
The most common grade 3 or 4 adverse events were neutropenia, leukopenia, lymphopenia and thrombocytopenias. Dr. Levy commented that the use of granulocyte colony-stimulating factor, which was not mandatory in the study, would likely have lowered the incidence of cytopenias.
There were 10 deaths during the study, 2 of which were considered to be treatment related, occurring in 1 patient each in the DLBCL dosing cohorts; 1 of the patients died from pneumonitis, and the other from left ventricular heart failure.
Other patients deaths were attributed to non–treatment-related cardiac arrest, acute renal failure, exacerbation of chronic heart failure, respiratory failure, multiorgan failure, lung infection, or colon adenocarcinoma.
Q 3 weeks suffices
In the question-and-answer session following the presentation, Kenny Lei, MD, from the Chinese University of Hong Kong asked Dr. Levy what the half-life of naratuximab is, and what was the investigator’s rationale for testing a weekly dosing schedule.
“I think the reason they checked the two different regimens, the Q week and the Q 3-week group, is that they noted that [naratuximab] was cleared relatively quickly, and they wanted to see whether or not, by giving Q weekly, when you get a continuous CD37 site occupancy if they would have a better outcome. But as you saw, in the groups there was really no clinically relevant difference in outcome,” Dr. Levy said.
Andrew Davies, MD, PhD, from the University of Southampton (England), asked whether the neutropenia seen in the study was related to myeloid expression of the target of from the off-target deconjugated payload.
“I don’t know that I necessarily have the answer to that,” Dr. Levy replied. “Remember there is the CD20 monoclonal rituximab which we know can cause neutropenia, as well as the CD37 and the target payload. I don’t know if we have enough information to attribute it to one specific component of the therapy,” he said.
The study was funded by Debiopharm International. Dr. Levy disclosed speaker activities for multiple companies, not including Debiopharm. Dr. Lei and Dr. Davies had no disclosures relevant to the study.
FROM EHA 2021
Promising HER2+/HR– breast cancer survival with de-escalated therapy
It may not be always necessary to approach the treatment of HER2-positive, hormone receptor–negative (HER2+/HR–) early breast cancer with added chemotherapy, survival results of a prospective multicenter randomized trial suggest.
In the ADAPT-HER2+/HR– trial, comparing a de-escalated 12-week neoadjuvant regimen consisting of dual HER2 blockade with trastuzumab (Herceptin) and pertuzumab (Perjeta) with or without weekly paclitaxel, the three-drug regimen was associated with high pathologic complete response(pCR) rates and excellent 5-year survival, irrespective of whether patients received additional chemotherapy, reported Nadia Harbeck, MD, PhD, of the University of Munich.
“Chemotherapy-free regimens are promising in highly sensitive tumors with early response, but future investigation of such chemotherapy-free regimens need to be focused on selected patients, like those with HER2 3+ tumors, non–basal-like tumors, those showing early response to the de-escalated therapy, and those with predictive RNA signatures such as immune signatures,” she said in an oral abstract session during the American Society of Clinical Oncology annual meeting (Abstract 503).
Under the WGS umbrella
The ADAPT HER2+/HR– trial (NCT01779206) is one of several conducted by the West German Study Group (WGS) on therapy for intrinsic breast cancer types.
In this study, 134 patients with HER2-positive, estrogen and progesterone receptor–negative tumors with no metastatic disease and good performance status were assigned on a 5:2 basis to neoadjuvant therapy with trastuzumab at a loading dose of 8 mg/kg for the first cycle followed by 6 mg/kg for subsequent cycles every 3 weeks x 4, plus pertuzumab at a loading dose of 840 mg followed by 420 mg every 3 weeks x 4 (92 patients), or to trastuzumab and pertuzumab at the same dose and schedule plus paclitaxel 80 mg/m2 once weekly for 12 weeks.
Patients had surgery within 3 weeks of the end of study therapy unless they did not have a histologically confirmed pCR, in which case they went on to receive standard neoadjuvant therapy prior to surgery.
Adjuvant therapy was performed according to national guidelines, although patients with a pCR after 12 weeks of study therapy could be spared from adjuvant chemotherapy at the investigator’s discretion.
Patients underwent biopsy at 3 weeks for therapy for early response assessment, defined as either a Ki67 decrease of at least 30% from baseline, or low cellularity (less than 500 invasive tumor cells).
First survival results
The investigators previously reported the primary pCR endpoint from the trial, which showed a rate of 90% after 12 weeks in the three-drug arm, and a “substantial and clinically meaningful” pCR rate of 34% after the trastuzumab plus pertuzumab alone.
At ASCO 2021, Dr. Harbeck reported the first survival data from the trial.
After a median follow-up of 59.9 months, there were no statistically significant differences between trial arms in either overall survival, invasive disease-free survival (iDFS), or distant disease-free survival (dDFS).
The 5-year iDFS rate in the three-drug arm was 98%, compared with 87% for the dual HER2 blockade-only arm, a difference that was not statistically significant.
The 5-year dDFS rates were 98% and 92% respectively. There were only seven dDFS events during follow-up, Dr. Harbeck noted.
There were only six deaths during follow-up, with overall survival rates of 98% in the paclitaxel-containing arm, and 94% in the anti-HER2 antibodies–only arm, a difference of one overall survival event, Dr. Harbeck said.
pCR counts
However, patients who did not have pathologic complete responses at the end of first-line de-escalated therapy had worse outcomes, with a 5-year iDFS rate of 82%, compared with 98% for patients who had achieved a pCR. This translated into a hazard ratio for invasive disease in patients with pCRs of 0.14 (P = .011).
This difference occurred despite the study requirement that all patients who did not have pCR after 12 weeks of initial therapy would receive additional chemotherapy.
Looking at the tumor subtype among patients in the paclitaxel-free arm to see whether they could identify predictors of early response, the researchers found a pCR rate of 36.5% among 85 patients with nonbasal tumors, but 0% among 7 patients with basal tumors.
The investigators identified a population of patients whose tumors could be considered nonsensitive to dual HER2 blockade alone: Those with basal tumors, those tumors with low immunohistochemical HER2 expression, and those without an early response to therapy on biopsy 3 weeks into initial therapy. Among 31 of the 92 patients in the dual HER2 arm who met this description, 2 had pCRs, Dr. Harbeck noted.
The 5-year iDFS rate among patients in the dual blockade–only arm with nonsensitive tumors was 79%, compared with 93% for patients with treatment-responsive types, although there were only 13 invasive events total in this arm.
“If we look at the whole trial population, the negative prognostic impact of what we termed nonsensitive tumors was even significant regarding dDFS, with a hazard ratio of about 5,” she said.
‘A consistent theme’
Invited discussant Lisa A. Carey, MD, ScM, of the University of North Carolina Lineberger Comprehensive Cancer Center in Chapel Hill, noted that the trial was underpowered for outcomes, but that results nonetheless suggest that patients with strongly HER2-driven tumors might get comparable benefits from less chemotherapy.
“This trial included only hormone receptor–negative, HER2-positive tumors, and these we know are likely to be HER2-enriched in terms of subtype, about three-quarters of them,”she said.
The previously reported pCR rate of 90% in the paclitaxel-containing arm, with 80% of patients requiring no further chemotherapy, resulted in the excellent 5-year iDFS and dDFS in this group, despite the relatively highly clinical stage, with about 60% of patients having clinical stage 2 or higher tumors, and more than 40% being node positive.
The idea that pCR itself can predict which patients could be spared from more intensive chemotherapy “is starting to look like a consistent theme,” she said.
Dr. Carey pointed out that in the KRISTINE trial comparing the combination of trastuzumab emtansine (T-DM1) and pertuzumab with standard chemotherapy in patients with HER2-positive stage I-III breast cancer, although the experimental combination was associated with lower pCR rates and worse event-free survival, rates of iDFS/dDFS were virtually identical for patients in both arms who achieved a pCR.
“So the question is can pCR mean that we can either eliminate additional therapy,” she said, noting that the question is currently being addressed prospectively in two clinical trials, COMPASS-pCR and DECRESCENDO.
ADAPT HER2+/HR- is sponsored by F. Hoffman-La Roche. Dr. Harbeck disclosed institutional research funding from Roche/Genentech, as well as honoraria and consulting/advising for multiple companies. Dr. Carey disclosed institutional research funding and other relationships with various companies.
It may not be always necessary to approach the treatment of HER2-positive, hormone receptor–negative (HER2+/HR–) early breast cancer with added chemotherapy, survival results of a prospective multicenter randomized trial suggest.
In the ADAPT-HER2+/HR– trial, comparing a de-escalated 12-week neoadjuvant regimen consisting of dual HER2 blockade with trastuzumab (Herceptin) and pertuzumab (Perjeta) with or without weekly paclitaxel, the three-drug regimen was associated with high pathologic complete response(pCR) rates and excellent 5-year survival, irrespective of whether patients received additional chemotherapy, reported Nadia Harbeck, MD, PhD, of the University of Munich.
“Chemotherapy-free regimens are promising in highly sensitive tumors with early response, but future investigation of such chemotherapy-free regimens need to be focused on selected patients, like those with HER2 3+ tumors, non–basal-like tumors, those showing early response to the de-escalated therapy, and those with predictive RNA signatures such as immune signatures,” she said in an oral abstract session during the American Society of Clinical Oncology annual meeting (Abstract 503).
Under the WGS umbrella
The ADAPT HER2+/HR– trial (NCT01779206) is one of several conducted by the West German Study Group (WGS) on therapy for intrinsic breast cancer types.
In this study, 134 patients with HER2-positive, estrogen and progesterone receptor–negative tumors with no metastatic disease and good performance status were assigned on a 5:2 basis to neoadjuvant therapy with trastuzumab at a loading dose of 8 mg/kg for the first cycle followed by 6 mg/kg for subsequent cycles every 3 weeks x 4, plus pertuzumab at a loading dose of 840 mg followed by 420 mg every 3 weeks x 4 (92 patients), or to trastuzumab and pertuzumab at the same dose and schedule plus paclitaxel 80 mg/m2 once weekly for 12 weeks.
Patients had surgery within 3 weeks of the end of study therapy unless they did not have a histologically confirmed pCR, in which case they went on to receive standard neoadjuvant therapy prior to surgery.
Adjuvant therapy was performed according to national guidelines, although patients with a pCR after 12 weeks of study therapy could be spared from adjuvant chemotherapy at the investigator’s discretion.
Patients underwent biopsy at 3 weeks for therapy for early response assessment, defined as either a Ki67 decrease of at least 30% from baseline, or low cellularity (less than 500 invasive tumor cells).
First survival results
The investigators previously reported the primary pCR endpoint from the trial, which showed a rate of 90% after 12 weeks in the three-drug arm, and a “substantial and clinically meaningful” pCR rate of 34% after the trastuzumab plus pertuzumab alone.
At ASCO 2021, Dr. Harbeck reported the first survival data from the trial.
After a median follow-up of 59.9 months, there were no statistically significant differences between trial arms in either overall survival, invasive disease-free survival (iDFS), or distant disease-free survival (dDFS).
The 5-year iDFS rate in the three-drug arm was 98%, compared with 87% for the dual HER2 blockade-only arm, a difference that was not statistically significant.
The 5-year dDFS rates were 98% and 92% respectively. There were only seven dDFS events during follow-up, Dr. Harbeck noted.
There were only six deaths during follow-up, with overall survival rates of 98% in the paclitaxel-containing arm, and 94% in the anti-HER2 antibodies–only arm, a difference of one overall survival event, Dr. Harbeck said.
pCR counts
However, patients who did not have pathologic complete responses at the end of first-line de-escalated therapy had worse outcomes, with a 5-year iDFS rate of 82%, compared with 98% for patients who had achieved a pCR. This translated into a hazard ratio for invasive disease in patients with pCRs of 0.14 (P = .011).
This difference occurred despite the study requirement that all patients who did not have pCR after 12 weeks of initial therapy would receive additional chemotherapy.
Looking at the tumor subtype among patients in the paclitaxel-free arm to see whether they could identify predictors of early response, the researchers found a pCR rate of 36.5% among 85 patients with nonbasal tumors, but 0% among 7 patients with basal tumors.
The investigators identified a population of patients whose tumors could be considered nonsensitive to dual HER2 blockade alone: Those with basal tumors, those tumors with low immunohistochemical HER2 expression, and those without an early response to therapy on biopsy 3 weeks into initial therapy. Among 31 of the 92 patients in the dual HER2 arm who met this description, 2 had pCRs, Dr. Harbeck noted.
The 5-year iDFS rate among patients in the dual blockade–only arm with nonsensitive tumors was 79%, compared with 93% for patients with treatment-responsive types, although there were only 13 invasive events total in this arm.
“If we look at the whole trial population, the negative prognostic impact of what we termed nonsensitive tumors was even significant regarding dDFS, with a hazard ratio of about 5,” she said.
‘A consistent theme’
Invited discussant Lisa A. Carey, MD, ScM, of the University of North Carolina Lineberger Comprehensive Cancer Center in Chapel Hill, noted that the trial was underpowered for outcomes, but that results nonetheless suggest that patients with strongly HER2-driven tumors might get comparable benefits from less chemotherapy.
“This trial included only hormone receptor–negative, HER2-positive tumors, and these we know are likely to be HER2-enriched in terms of subtype, about three-quarters of them,”she said.
The previously reported pCR rate of 90% in the paclitaxel-containing arm, with 80% of patients requiring no further chemotherapy, resulted in the excellent 5-year iDFS and dDFS in this group, despite the relatively highly clinical stage, with about 60% of patients having clinical stage 2 or higher tumors, and more than 40% being node positive.
The idea that pCR itself can predict which patients could be spared from more intensive chemotherapy “is starting to look like a consistent theme,” she said.
Dr. Carey pointed out that in the KRISTINE trial comparing the combination of trastuzumab emtansine (T-DM1) and pertuzumab with standard chemotherapy in patients with HER2-positive stage I-III breast cancer, although the experimental combination was associated with lower pCR rates and worse event-free survival, rates of iDFS/dDFS were virtually identical for patients in both arms who achieved a pCR.
“So the question is can pCR mean that we can either eliminate additional therapy,” she said, noting that the question is currently being addressed prospectively in two clinical trials, COMPASS-pCR and DECRESCENDO.
ADAPT HER2+/HR- is sponsored by F. Hoffman-La Roche. Dr. Harbeck disclosed institutional research funding from Roche/Genentech, as well as honoraria and consulting/advising for multiple companies. Dr. Carey disclosed institutional research funding and other relationships with various companies.
It may not be always necessary to approach the treatment of HER2-positive, hormone receptor–negative (HER2+/HR–) early breast cancer with added chemotherapy, survival results of a prospective multicenter randomized trial suggest.
In the ADAPT-HER2+/HR– trial, comparing a de-escalated 12-week neoadjuvant regimen consisting of dual HER2 blockade with trastuzumab (Herceptin) and pertuzumab (Perjeta) with or without weekly paclitaxel, the three-drug regimen was associated with high pathologic complete response(pCR) rates and excellent 5-year survival, irrespective of whether patients received additional chemotherapy, reported Nadia Harbeck, MD, PhD, of the University of Munich.
“Chemotherapy-free regimens are promising in highly sensitive tumors with early response, but future investigation of such chemotherapy-free regimens need to be focused on selected patients, like those with HER2 3+ tumors, non–basal-like tumors, those showing early response to the de-escalated therapy, and those with predictive RNA signatures such as immune signatures,” she said in an oral abstract session during the American Society of Clinical Oncology annual meeting (Abstract 503).
Under the WGS umbrella
The ADAPT HER2+/HR– trial (NCT01779206) is one of several conducted by the West German Study Group (WGS) on therapy for intrinsic breast cancer types.
In this study, 134 patients with HER2-positive, estrogen and progesterone receptor–negative tumors with no metastatic disease and good performance status were assigned on a 5:2 basis to neoadjuvant therapy with trastuzumab at a loading dose of 8 mg/kg for the first cycle followed by 6 mg/kg for subsequent cycles every 3 weeks x 4, plus pertuzumab at a loading dose of 840 mg followed by 420 mg every 3 weeks x 4 (92 patients), or to trastuzumab and pertuzumab at the same dose and schedule plus paclitaxel 80 mg/m2 once weekly for 12 weeks.
Patients had surgery within 3 weeks of the end of study therapy unless they did not have a histologically confirmed pCR, in which case they went on to receive standard neoadjuvant therapy prior to surgery.
Adjuvant therapy was performed according to national guidelines, although patients with a pCR after 12 weeks of study therapy could be spared from adjuvant chemotherapy at the investigator’s discretion.
Patients underwent biopsy at 3 weeks for therapy for early response assessment, defined as either a Ki67 decrease of at least 30% from baseline, or low cellularity (less than 500 invasive tumor cells).
First survival results
The investigators previously reported the primary pCR endpoint from the trial, which showed a rate of 90% after 12 weeks in the three-drug arm, and a “substantial and clinically meaningful” pCR rate of 34% after the trastuzumab plus pertuzumab alone.
At ASCO 2021, Dr. Harbeck reported the first survival data from the trial.
After a median follow-up of 59.9 months, there were no statistically significant differences between trial arms in either overall survival, invasive disease-free survival (iDFS), or distant disease-free survival (dDFS).
The 5-year iDFS rate in the three-drug arm was 98%, compared with 87% for the dual HER2 blockade-only arm, a difference that was not statistically significant.
The 5-year dDFS rates were 98% and 92% respectively. There were only seven dDFS events during follow-up, Dr. Harbeck noted.
There were only six deaths during follow-up, with overall survival rates of 98% in the paclitaxel-containing arm, and 94% in the anti-HER2 antibodies–only arm, a difference of one overall survival event, Dr. Harbeck said.
pCR counts
However, patients who did not have pathologic complete responses at the end of first-line de-escalated therapy had worse outcomes, with a 5-year iDFS rate of 82%, compared with 98% for patients who had achieved a pCR. This translated into a hazard ratio for invasive disease in patients with pCRs of 0.14 (P = .011).
This difference occurred despite the study requirement that all patients who did not have pCR after 12 weeks of initial therapy would receive additional chemotherapy.
Looking at the tumor subtype among patients in the paclitaxel-free arm to see whether they could identify predictors of early response, the researchers found a pCR rate of 36.5% among 85 patients with nonbasal tumors, but 0% among 7 patients with basal tumors.
The investigators identified a population of patients whose tumors could be considered nonsensitive to dual HER2 blockade alone: Those with basal tumors, those tumors with low immunohistochemical HER2 expression, and those without an early response to therapy on biopsy 3 weeks into initial therapy. Among 31 of the 92 patients in the dual HER2 arm who met this description, 2 had pCRs, Dr. Harbeck noted.
The 5-year iDFS rate among patients in the dual blockade–only arm with nonsensitive tumors was 79%, compared with 93% for patients with treatment-responsive types, although there were only 13 invasive events total in this arm.
“If we look at the whole trial population, the negative prognostic impact of what we termed nonsensitive tumors was even significant regarding dDFS, with a hazard ratio of about 5,” she said.
‘A consistent theme’
Invited discussant Lisa A. Carey, MD, ScM, of the University of North Carolina Lineberger Comprehensive Cancer Center in Chapel Hill, noted that the trial was underpowered for outcomes, but that results nonetheless suggest that patients with strongly HER2-driven tumors might get comparable benefits from less chemotherapy.
“This trial included only hormone receptor–negative, HER2-positive tumors, and these we know are likely to be HER2-enriched in terms of subtype, about three-quarters of them,”she said.
The previously reported pCR rate of 90% in the paclitaxel-containing arm, with 80% of patients requiring no further chemotherapy, resulted in the excellent 5-year iDFS and dDFS in this group, despite the relatively highly clinical stage, with about 60% of patients having clinical stage 2 or higher tumors, and more than 40% being node positive.
The idea that pCR itself can predict which patients could be spared from more intensive chemotherapy “is starting to look like a consistent theme,” she said.
Dr. Carey pointed out that in the KRISTINE trial comparing the combination of trastuzumab emtansine (T-DM1) and pertuzumab with standard chemotherapy in patients with HER2-positive stage I-III breast cancer, although the experimental combination was associated with lower pCR rates and worse event-free survival, rates of iDFS/dDFS were virtually identical for patients in both arms who achieved a pCR.
“So the question is can pCR mean that we can either eliminate additional therapy,” she said, noting that the question is currently being addressed prospectively in two clinical trials, COMPASS-pCR and DECRESCENDO.
ADAPT HER2+/HR- is sponsored by F. Hoffman-La Roche. Dr. Harbeck disclosed institutional research funding from Roche/Genentech, as well as honoraria and consulting/advising for multiple companies. Dr. Carey disclosed institutional research funding and other relationships with various companies.
FROM ASCO 2021
In the previous year, 70% of oncologists reported sexual harassment
A new survey of sexual harassment among U.S. oncologists has found that 70% reported incidents from peers and/or supervisors in the previous 12 months.
The incidence was higher among women than men (80% vs. 56%), a difference that was statistically significant (P < .0001).
However, after experiencing sexual harassment from coworkers, men and women were alike in terms of reporting similarly negative outcomes in mental health, sense of safety, and turnover intentions (e.g., leaving or quitting).
“Our findings demonstrate that the impact of sexual harassment on both men and women is tangible and is not different,” said lead author Ishwaria Subbiah, MD, a medical oncologist at the University of Texas MD Anderson Cancer Center, Houston, during her presentation of the study on June 5 at the American Society of Clinical Oncology (ASCO) 2021. The meeting was held virtually because of the pandemic.
“The survey’s recall period [about harassment] was in the previous 12 months. The respondents weren’t reflecting on a lifetime of events,” Dr. Subbiah said in an interview. “That’s part of what makes the findings that much more sobering.”
The release of the survey results roughly coincided with a furor within oncology circles over details that have now come to light about Axel Grothey, MD, a high-profile medical oncologist who was forced out of the Mayo Clinic in Rochester, Minn., after having unethical sexual relations with mentees – only to move on to another major center with more mentees.
The new survey, which included 153 women and 118 men, was conducted in 2020.
Overall, 69% of respondents reported gender-based harassment, 17% reported unwanted sexual attention, and 3% reported sexual coercion from peers/supervisors. For the three types of sexual harassment, women reported higher rates of incidence; the greatest proportional disparity was in unwanted sexual attention (22% of women vs. 9% of men).
The types of sexual harassment are defined in a landmark 2018 report from the National Academies of Sciences, Engineering, and Medicine. Gender harassment is nonverbal or verbal behaviors that are hostile, objectifying, and excluding of or conveying second-class status about a gender. Unwanted sexual attention is advances, including touching, and seeking a sexual relationship despite discouragement. Sexual coercion involves seeking compliance with sexual demands by making job-related threats or promising job-related benefits.
The commonality in the three harassments is their being “unwanted,” Dr. Subbiah explained.
Another commonality is that “sexual harassment is a tool of power that one person yields over another,” commented Marina Stasenko, MD, a gynecologic oncologist at NYU Langone’s Perlmutter Cancer Center.
Dr. Stasenko led a 2018 study that found that 64% of U.S. gynecologic oncologists reported sexual harassment during training or practice, a much longer recall period than the 1 year in Dr. Subbiah’s study.
However, things may be changing regarding sexual harassment – at least in terms of victims speaking out, said Dr. Stasenko. Perhaps discussing personal experience “is becoming less taboo,” she told this news organization. “The media spotlight on sexual harassment within medicine has been bright [recently].”
That was borne out last week – a number of oncologists who had been harassed told their stories on Twitter in reaction to the report about Dr. Grothey at one of America’s top medical centers. Also, in another sign of the moment, an academic oncologist publicly said that rumors about Dr. Grothey were long-standing. “Heard from many colleagues that this behavior was known in the field and went on for years. Years,” tweeted Charu Aggarwal, MD, MPH, from the University of Pennsylvania, Philadelphia.
Other outcomes seem to make Dr. Grothey’s behavior at Mayo, which multiple oncologists said has occurred at every center, a watershed moment. Namely, he has been muted or dismissed by an array of organizations since the story broke.
ASCO disallowed Dr. Grothey from making presentations at the annual meeting (he was an author on 12 studies), the National Cancer Institute removed him from his position as co-chair of an influential steering committee that helps determine grant funding for research, and the OneOncology community care network dropped him as medical director of their research arm, as reported by The Cancer Letter. He was also removed from the OncoAlert Network, a global network of oncology professionals, and from the medical advisory board of Fight CRC, an advocacy group for patients with colorectal cancer, as reported by this news organization. His current employer, West Cancer Center, in Germantown, Tennessee, has also started an investigation.
In her presentation, Dr. Subbiah acknowledged a changing landscape, with “increasing attention in recent years” to sexual harassment thanks to the “broader cultural movements” of #metoo and #TIMESUP social media–based campaigns.
Another oncologist nodded to the recent news about Dr. Grothey at the Mayo Clinic and suggested Dr. Subbiah’s study was part of a historic struggle for equity for women. “Sadly, both timely and timeless,” tweeted medical oncologist Tatiana Prowell, MD, of Johns Hopkins University, Baltimore, about the new study.
Academia has a problem
To conduct their survey, Dr. Subbiah and her coinvestigators reached out to 1,000 randomly selected U.S. members of ASCO via the organization’s research survey pool, as well as through Twitter and Facebook. The invitation to participate described the survey as being about the “workplace experience of oncologists” and that it aimed to mitigate response bias.
Of the 271 survey respondents, 250 were oncologists in practice and 21 were residents/fellows. Nearly all were heterosexual (94%) and U.S. citizens (87%). A majority (53%) were White, 35% were Asian/Pacific Islander, and 11% were Black or Hispanic. Most (68%) were more than 5 years out from training.
Most of the respondents (62%) were from academia.
“There is a big problem of sexual harassment in academic medicine,” said Pamela Kunz, MD, of Yale Cancer Center, New Haven, Conn., who was asked for comment. Dr. Kunz left Stanford University in 2020 after 19 years, citing repeated harassment.
“The institutions tend to protect the brand rather than the victim. Perpetrators are often not disciplined and may leave an institution under cover of a resignation only to go on and receive a better leadership role at another institution,” she said in an interview.
A “revolution” is needed to address the problem, Dr. Kunz said, citing needs to routinely discuss the topic, systems to measure and track it, methods to hold perpetrators accountable, and meaningful educational opportunities.
Harassment from patients/families also tallied
The new survey also queried participants with regard to sexual harassment from patients and/or families, which was reported by 67% of women and 35% of men (P < .0001).
As with harassment from peers/supervisors, gender harassment was the most common form and was reported by significantly higher percentages of women.
And as with coworkers, sexual harassment from patients/families was also significantly associated with detriments to mental health, workplace safety, and turnover intentions.
Sexual harassment from “insiders” (P = .001) but not patients (P = .55) was significantly associated with a decrease in a fourth metric in the study – job satisfaction.
“The goal of medicine and oncology is ‘ultimately to ease suffering,’” Dr. Subbiah said. That holds true for workplace wellness, including addressing harassment. “There should be no hesitation to go there and look at what is truly impacting the workplace,” she said.
“This is a difficult topic,” Dr. Subbiah acknowledged, adding that “the findings are sobering and merit open, global conversation among all oncology stakeholders.”
The study authors, Dr. Ganz, and Dr. Stasenko have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new survey of sexual harassment among U.S. oncologists has found that 70% reported incidents from peers and/or supervisors in the previous 12 months.
The incidence was higher among women than men (80% vs. 56%), a difference that was statistically significant (P < .0001).
However, after experiencing sexual harassment from coworkers, men and women were alike in terms of reporting similarly negative outcomes in mental health, sense of safety, and turnover intentions (e.g., leaving or quitting).
“Our findings demonstrate that the impact of sexual harassment on both men and women is tangible and is not different,” said lead author Ishwaria Subbiah, MD, a medical oncologist at the University of Texas MD Anderson Cancer Center, Houston, during her presentation of the study on June 5 at the American Society of Clinical Oncology (ASCO) 2021. The meeting was held virtually because of the pandemic.
“The survey’s recall period [about harassment] was in the previous 12 months. The respondents weren’t reflecting on a lifetime of events,” Dr. Subbiah said in an interview. “That’s part of what makes the findings that much more sobering.”
The release of the survey results roughly coincided with a furor within oncology circles over details that have now come to light about Axel Grothey, MD, a high-profile medical oncologist who was forced out of the Mayo Clinic in Rochester, Minn., after having unethical sexual relations with mentees – only to move on to another major center with more mentees.
The new survey, which included 153 women and 118 men, was conducted in 2020.
Overall, 69% of respondents reported gender-based harassment, 17% reported unwanted sexual attention, and 3% reported sexual coercion from peers/supervisors. For the three types of sexual harassment, women reported higher rates of incidence; the greatest proportional disparity was in unwanted sexual attention (22% of women vs. 9% of men).
The types of sexual harassment are defined in a landmark 2018 report from the National Academies of Sciences, Engineering, and Medicine. Gender harassment is nonverbal or verbal behaviors that are hostile, objectifying, and excluding of or conveying second-class status about a gender. Unwanted sexual attention is advances, including touching, and seeking a sexual relationship despite discouragement. Sexual coercion involves seeking compliance with sexual demands by making job-related threats or promising job-related benefits.
The commonality in the three harassments is their being “unwanted,” Dr. Subbiah explained.
Another commonality is that “sexual harassment is a tool of power that one person yields over another,” commented Marina Stasenko, MD, a gynecologic oncologist at NYU Langone’s Perlmutter Cancer Center.
Dr. Stasenko led a 2018 study that found that 64% of U.S. gynecologic oncologists reported sexual harassment during training or practice, a much longer recall period than the 1 year in Dr. Subbiah’s study.
However, things may be changing regarding sexual harassment – at least in terms of victims speaking out, said Dr. Stasenko. Perhaps discussing personal experience “is becoming less taboo,” she told this news organization. “The media spotlight on sexual harassment within medicine has been bright [recently].”
That was borne out last week – a number of oncologists who had been harassed told their stories on Twitter in reaction to the report about Dr. Grothey at one of America’s top medical centers. Also, in another sign of the moment, an academic oncologist publicly said that rumors about Dr. Grothey were long-standing. “Heard from many colleagues that this behavior was known in the field and went on for years. Years,” tweeted Charu Aggarwal, MD, MPH, from the University of Pennsylvania, Philadelphia.
Other outcomes seem to make Dr. Grothey’s behavior at Mayo, which multiple oncologists said has occurred at every center, a watershed moment. Namely, he has been muted or dismissed by an array of organizations since the story broke.
ASCO disallowed Dr. Grothey from making presentations at the annual meeting (he was an author on 12 studies), the National Cancer Institute removed him from his position as co-chair of an influential steering committee that helps determine grant funding for research, and the OneOncology community care network dropped him as medical director of their research arm, as reported by The Cancer Letter. He was also removed from the OncoAlert Network, a global network of oncology professionals, and from the medical advisory board of Fight CRC, an advocacy group for patients with colorectal cancer, as reported by this news organization. His current employer, West Cancer Center, in Germantown, Tennessee, has also started an investigation.
In her presentation, Dr. Subbiah acknowledged a changing landscape, with “increasing attention in recent years” to sexual harassment thanks to the “broader cultural movements” of #metoo and #TIMESUP social media–based campaigns.
Another oncologist nodded to the recent news about Dr. Grothey at the Mayo Clinic and suggested Dr. Subbiah’s study was part of a historic struggle for equity for women. “Sadly, both timely and timeless,” tweeted medical oncologist Tatiana Prowell, MD, of Johns Hopkins University, Baltimore, about the new study.
Academia has a problem
To conduct their survey, Dr. Subbiah and her coinvestigators reached out to 1,000 randomly selected U.S. members of ASCO via the organization’s research survey pool, as well as through Twitter and Facebook. The invitation to participate described the survey as being about the “workplace experience of oncologists” and that it aimed to mitigate response bias.
Of the 271 survey respondents, 250 were oncologists in practice and 21 were residents/fellows. Nearly all were heterosexual (94%) and U.S. citizens (87%). A majority (53%) were White, 35% were Asian/Pacific Islander, and 11% were Black or Hispanic. Most (68%) were more than 5 years out from training.
Most of the respondents (62%) were from academia.
“There is a big problem of sexual harassment in academic medicine,” said Pamela Kunz, MD, of Yale Cancer Center, New Haven, Conn., who was asked for comment. Dr. Kunz left Stanford University in 2020 after 19 years, citing repeated harassment.
“The institutions tend to protect the brand rather than the victim. Perpetrators are often not disciplined and may leave an institution under cover of a resignation only to go on and receive a better leadership role at another institution,” she said in an interview.
A “revolution” is needed to address the problem, Dr. Kunz said, citing needs to routinely discuss the topic, systems to measure and track it, methods to hold perpetrators accountable, and meaningful educational opportunities.
Harassment from patients/families also tallied
The new survey also queried participants with regard to sexual harassment from patients and/or families, which was reported by 67% of women and 35% of men (P < .0001).
As with harassment from peers/supervisors, gender harassment was the most common form and was reported by significantly higher percentages of women.
And as with coworkers, sexual harassment from patients/families was also significantly associated with detriments to mental health, workplace safety, and turnover intentions.
Sexual harassment from “insiders” (P = .001) but not patients (P = .55) was significantly associated with a decrease in a fourth metric in the study – job satisfaction.
“The goal of medicine and oncology is ‘ultimately to ease suffering,’” Dr. Subbiah said. That holds true for workplace wellness, including addressing harassment. “There should be no hesitation to go there and look at what is truly impacting the workplace,” she said.
“This is a difficult topic,” Dr. Subbiah acknowledged, adding that “the findings are sobering and merit open, global conversation among all oncology stakeholders.”
The study authors, Dr. Ganz, and Dr. Stasenko have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new survey of sexual harassment among U.S. oncologists has found that 70% reported incidents from peers and/or supervisors in the previous 12 months.
The incidence was higher among women than men (80% vs. 56%), a difference that was statistically significant (P < .0001).
However, after experiencing sexual harassment from coworkers, men and women were alike in terms of reporting similarly negative outcomes in mental health, sense of safety, and turnover intentions (e.g., leaving or quitting).
“Our findings demonstrate that the impact of sexual harassment on both men and women is tangible and is not different,” said lead author Ishwaria Subbiah, MD, a medical oncologist at the University of Texas MD Anderson Cancer Center, Houston, during her presentation of the study on June 5 at the American Society of Clinical Oncology (ASCO) 2021. The meeting was held virtually because of the pandemic.
“The survey’s recall period [about harassment] was in the previous 12 months. The respondents weren’t reflecting on a lifetime of events,” Dr. Subbiah said in an interview. “That’s part of what makes the findings that much more sobering.”
The release of the survey results roughly coincided with a furor within oncology circles over details that have now come to light about Axel Grothey, MD, a high-profile medical oncologist who was forced out of the Mayo Clinic in Rochester, Minn., after having unethical sexual relations with mentees – only to move on to another major center with more mentees.
The new survey, which included 153 women and 118 men, was conducted in 2020.
Overall, 69% of respondents reported gender-based harassment, 17% reported unwanted sexual attention, and 3% reported sexual coercion from peers/supervisors. For the three types of sexual harassment, women reported higher rates of incidence; the greatest proportional disparity was in unwanted sexual attention (22% of women vs. 9% of men).
The types of sexual harassment are defined in a landmark 2018 report from the National Academies of Sciences, Engineering, and Medicine. Gender harassment is nonverbal or verbal behaviors that are hostile, objectifying, and excluding of or conveying second-class status about a gender. Unwanted sexual attention is advances, including touching, and seeking a sexual relationship despite discouragement. Sexual coercion involves seeking compliance with sexual demands by making job-related threats or promising job-related benefits.
The commonality in the three harassments is their being “unwanted,” Dr. Subbiah explained.
Another commonality is that “sexual harassment is a tool of power that one person yields over another,” commented Marina Stasenko, MD, a gynecologic oncologist at NYU Langone’s Perlmutter Cancer Center.
Dr. Stasenko led a 2018 study that found that 64% of U.S. gynecologic oncologists reported sexual harassment during training or practice, a much longer recall period than the 1 year in Dr. Subbiah’s study.
However, things may be changing regarding sexual harassment – at least in terms of victims speaking out, said Dr. Stasenko. Perhaps discussing personal experience “is becoming less taboo,” she told this news organization. “The media spotlight on sexual harassment within medicine has been bright [recently].”
That was borne out last week – a number of oncologists who had been harassed told their stories on Twitter in reaction to the report about Dr. Grothey at one of America’s top medical centers. Also, in another sign of the moment, an academic oncologist publicly said that rumors about Dr. Grothey were long-standing. “Heard from many colleagues that this behavior was known in the field and went on for years. Years,” tweeted Charu Aggarwal, MD, MPH, from the University of Pennsylvania, Philadelphia.
Other outcomes seem to make Dr. Grothey’s behavior at Mayo, which multiple oncologists said has occurred at every center, a watershed moment. Namely, he has been muted or dismissed by an array of organizations since the story broke.
ASCO disallowed Dr. Grothey from making presentations at the annual meeting (he was an author on 12 studies), the National Cancer Institute removed him from his position as co-chair of an influential steering committee that helps determine grant funding for research, and the OneOncology community care network dropped him as medical director of their research arm, as reported by The Cancer Letter. He was also removed from the OncoAlert Network, a global network of oncology professionals, and from the medical advisory board of Fight CRC, an advocacy group for patients with colorectal cancer, as reported by this news organization. His current employer, West Cancer Center, in Germantown, Tennessee, has also started an investigation.
In her presentation, Dr. Subbiah acknowledged a changing landscape, with “increasing attention in recent years” to sexual harassment thanks to the “broader cultural movements” of #metoo and #TIMESUP social media–based campaigns.
Another oncologist nodded to the recent news about Dr. Grothey at the Mayo Clinic and suggested Dr. Subbiah’s study was part of a historic struggle for equity for women. “Sadly, both timely and timeless,” tweeted medical oncologist Tatiana Prowell, MD, of Johns Hopkins University, Baltimore, about the new study.
Academia has a problem
To conduct their survey, Dr. Subbiah and her coinvestigators reached out to 1,000 randomly selected U.S. members of ASCO via the organization’s research survey pool, as well as through Twitter and Facebook. The invitation to participate described the survey as being about the “workplace experience of oncologists” and that it aimed to mitigate response bias.
Of the 271 survey respondents, 250 were oncologists in practice and 21 were residents/fellows. Nearly all were heterosexual (94%) and U.S. citizens (87%). A majority (53%) were White, 35% were Asian/Pacific Islander, and 11% were Black or Hispanic. Most (68%) were more than 5 years out from training.
Most of the respondents (62%) were from academia.
“There is a big problem of sexual harassment in academic medicine,” said Pamela Kunz, MD, of Yale Cancer Center, New Haven, Conn., who was asked for comment. Dr. Kunz left Stanford University in 2020 after 19 years, citing repeated harassment.
“The institutions tend to protect the brand rather than the victim. Perpetrators are often not disciplined and may leave an institution under cover of a resignation only to go on and receive a better leadership role at another institution,” she said in an interview.
A “revolution” is needed to address the problem, Dr. Kunz said, citing needs to routinely discuss the topic, systems to measure and track it, methods to hold perpetrators accountable, and meaningful educational opportunities.
Harassment from patients/families also tallied
The new survey also queried participants with regard to sexual harassment from patients and/or families, which was reported by 67% of women and 35% of men (P < .0001).
As with harassment from peers/supervisors, gender harassment was the most common form and was reported by significantly higher percentages of women.
And as with coworkers, sexual harassment from patients/families was also significantly associated with detriments to mental health, workplace safety, and turnover intentions.
Sexual harassment from “insiders” (P = .001) but not patients (P = .55) was significantly associated with a decrease in a fourth metric in the study – job satisfaction.
“The goal of medicine and oncology is ‘ultimately to ease suffering,’” Dr. Subbiah said. That holds true for workplace wellness, including addressing harassment. “There should be no hesitation to go there and look at what is truly impacting the workplace,” she said.
“This is a difficult topic,” Dr. Subbiah acknowledged, adding that “the findings are sobering and merit open, global conversation among all oncology stakeholders.”
The study authors, Dr. Ganz, and Dr. Stasenko have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Surgical outcomes favor addition of nivolumab to neoadjuvant chemo in resectable lung cancers
The addition of nivolumab to neoadjuvant chemotherapy did not impede the feasibility or timing of surgery in patients with resectable lung cancer, according to results from the phase 3 CheckMate 816 trial.
Adding nivolumab to chemotherapy was tolerable and did not increase the rate of surgical complications, investigator Jonathan Spicer, FRCPC, MD, PhD, of McGill University, Montreal, said in his presentation at the annual meeting of the American Society of Clinical Oncology.
His presentation comes about 2 months after the reporting of primary endpoint results of CheckMate 816 (NCT02998528). CheckMate 816 demonstrated that adding nivolumab to neoadjuvant chemotherapy significantly improved pathological complete response (pCR) in patients with resectable non–small cell lung cancer (NSCLC), according to results presented earlier at the American Association for Cancer Research annual meeting.
“The safety and surgical outcome data reported thus far from CheckMate 816, along with significant improvement in pathological complete response, support nivolumab in combination with chemotherapy as an attractive neoadjuvant option for patients with resectable NSCLC,” said Dr. Spicer (Abstract 8503).
Building on previous experience
The CheckMate 816 study builds on extensive experience in advanced NSCLC that has consistently shown better outcomes, including overall survival, with combinations of chemotherapy and immuno-oncology (IO) agents, compared to chemotherapy alone, said discussant Valerie W. Rusch, MD, of Memorial Sloan Kettering Cancer Center in New York.
Dr. Rusch called out “salient and interesting results” regarding surgical management in CheckMate 816, including a lower rate of surgery cancellations and shorter surgical duration in the chemotherapy-plus-IO arm, compared to the chemotherapy-alone arm.
Furthermore, fewer patients required a pneumonectomy and more patients had a complete resection in the chemotherapy-plus-IO arm, compared to chemotherapy alone, she noted.
“These excellent surgical results, along with the data previously presented at AACR regarding the primary endpoint, help to establish a new standard of neoadjuvant care,” Dr. Rusch said in her presentation.
Study details
CheckMate 816 included 358 patients with newly diagnosed, resectable, stage IB-IIIA NSCLC, Eastern Cooperative Oncology Group (ECOG) performance status of 0-1, and no known EGFR mutations or ALK alterations. Patients were randomized to receive nivolumab and platinum-doublet chemotherapy (nivolumab/chemotherapy) or chemotherapy alone every 3 weeks, with surgery to be performed within 6 weeks of the last dose of neoadjuvant treatment.
The median age of patients was 64 years in the nivolumab/chemotherapy arm and 65 years in the chemotherapy-alone arm. About one-third of patients had ECOG performance status of one, and about half had squamous tumor histology, Dr. Spicer said in his report. Almost two-thirds of patients had stage IIIA disease.
In the study results previously presented at the AACR meeting, both pCR and major pathologic response were significantly better following neoadjuvant chemotherapy and IO treatment, compared to chemotherapy alone.
In the intention-to-treat analysis, 24.0% of patients treated with nivolumab/chemotherapy achieved a pCR, compared to 2.2% in the chemotherapy arm, amounting to an approximate 12-fold increase in pCR, Dr. Spicer said. Similarly, the rate of major pathologic response in the intention-to-treat analysis was 36.9% and 8.9% for the nivolumab/chemotherapy and chemotherapy arms, respectively.
Surgical results
In his ASCO presentation, Dr. Spicer reported that definitive surgery was canceled in 16% of patients in the nivolumab/chemotherapy arm, and 21% of the chemotherapy arm. Reasons for surgery cancellation generally included patients declining surgery, unresectable disease, and poor lung function. “Cancellation of surgery due to neoadjuvant therapy toxicity was rare,” Dr. Spicer said in his presentation.
Among patients who did proceed to surgery, the median duration of the procedure was 184 minutes in the nivolumab/chemotherapy arm and 217 minutes in the chemotherapy arm. That half-hour difference in favor of the combination arm suggests that the complexity of surgery was not increased by the addition of nivolumab, Dr. Spicer said.
Median time to surgery was about 5 weeks in both arms, which was “well within accepted standards for a neoadjuvant therapeutic approach,” Dr. Spicer said. Most delays beyond 6 weeks were due to administrative issues, and occurred in similar proportions (21% of the nivolumab/chemotherapy arm and 18% of the chemotherapy arm).
The addition of nivolumab to chemotherapy improved pCR rates regardless of baseline stage of disease, according to Dr. Spicer. Furthermore, the depth of pathological regression in the primary tumor was “dramatically different” across stage groupings, he said. Median residual viable tumor percentage in stage IB/II patients was 28% for nivolumab/chemotherapy and 79% for chemotherapy, and in stage IIIA patients, it was 8% for nivolumab/chemotherapy and 70% for chemotherapy.
Overall, thoracotomy was the most frequent surgical approach in this international phase 3 trial, Dr. Spicer said. However, among patients with stage IIIA disease, minimally invasive approaches were used 30% of the time in the nivolumab/chemotherapy arm and 19% in the chemotherapy arm. Conversely, the rate of conversion from a minimally invasive to open approach in patients with stage IIIA disease was 11% for nivolumab/chemotherapy and 20% for chemotherapy alone.
Lobectomy was more frequent in the nivolumab/chemotherapy arm (77%) compared to the chemotherapy arm (61%), a difference that Dr. Spicer described as clinically important. He said the difference appears to be attributable to a lower rate of pneumonectomy in the nivolumab/chemotherapy arm (17%) than in the chemotherapy arm (25%).
Despite less extensive lung resection being required, the rate of R0 resection was numerically higher in the nivolumab/chemotherapy arm (83%) than in the chemotherapy arm (78%), said Dr. Spicer.
Length of hospital stay was “within expected ranges” from geographic regions represented in the trial, Dr. Spicer said. Median length of stay was 4.0 and 6.0 days, respectively, for nivolumab/chemotherapy and chemotherapy alone in North America, 9.5 and 13.0 days in Europe, and 11.0 and 13.0 days in Asia.
Likewise, 90-day surgical complications were well within expected ranges, according to the investigator, with anemia, pain, and wound complications being the most commonly reported. Rates were generally similar between study arms, other than a twofold higher rate of pain in the chemotherapy arm, possibly due to the lower rate of minimally invasive surgery or higher rate of conversion to an open procedure, compared to the nivolumab/chemotherapy arm, he said.
Awaiting survival
Rates of 30- and 90-day mortality are expected to be evaluated when survival endpoints are available, according to Dr. Spicer. Beyond pCR rate, event-free survival is also a primary endpoint of the study, while overall survival is a secondary endpoint.
The study was supported by Bristol Myers Squibb. Dr. Spicer reported disclosures related to AstraZeneca, Bristol Myers Squibb Foundation, Merck, and Roche. Dr. Rusch reported research funding with Genelux and Genentech, and travel expenses from Intuitive Surgical.
The addition of nivolumab to neoadjuvant chemotherapy did not impede the feasibility or timing of surgery in patients with resectable lung cancer, according to results from the phase 3 CheckMate 816 trial.
Adding nivolumab to chemotherapy was tolerable and did not increase the rate of surgical complications, investigator Jonathan Spicer, FRCPC, MD, PhD, of McGill University, Montreal, said in his presentation at the annual meeting of the American Society of Clinical Oncology.
His presentation comes about 2 months after the reporting of primary endpoint results of CheckMate 816 (NCT02998528). CheckMate 816 demonstrated that adding nivolumab to neoadjuvant chemotherapy significantly improved pathological complete response (pCR) in patients with resectable non–small cell lung cancer (NSCLC), according to results presented earlier at the American Association for Cancer Research annual meeting.
“The safety and surgical outcome data reported thus far from CheckMate 816, along with significant improvement in pathological complete response, support nivolumab in combination with chemotherapy as an attractive neoadjuvant option for patients with resectable NSCLC,” said Dr. Spicer (Abstract 8503).
Building on previous experience
The CheckMate 816 study builds on extensive experience in advanced NSCLC that has consistently shown better outcomes, including overall survival, with combinations of chemotherapy and immuno-oncology (IO) agents, compared to chemotherapy alone, said discussant Valerie W. Rusch, MD, of Memorial Sloan Kettering Cancer Center in New York.
Dr. Rusch called out “salient and interesting results” regarding surgical management in CheckMate 816, including a lower rate of surgery cancellations and shorter surgical duration in the chemotherapy-plus-IO arm, compared to the chemotherapy-alone arm.
Furthermore, fewer patients required a pneumonectomy and more patients had a complete resection in the chemotherapy-plus-IO arm, compared to chemotherapy alone, she noted.
“These excellent surgical results, along with the data previously presented at AACR regarding the primary endpoint, help to establish a new standard of neoadjuvant care,” Dr. Rusch said in her presentation.
Study details
CheckMate 816 included 358 patients with newly diagnosed, resectable, stage IB-IIIA NSCLC, Eastern Cooperative Oncology Group (ECOG) performance status of 0-1, and no known EGFR mutations or ALK alterations. Patients were randomized to receive nivolumab and platinum-doublet chemotherapy (nivolumab/chemotherapy) or chemotherapy alone every 3 weeks, with surgery to be performed within 6 weeks of the last dose of neoadjuvant treatment.
The median age of patients was 64 years in the nivolumab/chemotherapy arm and 65 years in the chemotherapy-alone arm. About one-third of patients had ECOG performance status of one, and about half had squamous tumor histology, Dr. Spicer said in his report. Almost two-thirds of patients had stage IIIA disease.
In the study results previously presented at the AACR meeting, both pCR and major pathologic response were significantly better following neoadjuvant chemotherapy and IO treatment, compared to chemotherapy alone.
In the intention-to-treat analysis, 24.0% of patients treated with nivolumab/chemotherapy achieved a pCR, compared to 2.2% in the chemotherapy arm, amounting to an approximate 12-fold increase in pCR, Dr. Spicer said. Similarly, the rate of major pathologic response in the intention-to-treat analysis was 36.9% and 8.9% for the nivolumab/chemotherapy and chemotherapy arms, respectively.
Surgical results
In his ASCO presentation, Dr. Spicer reported that definitive surgery was canceled in 16% of patients in the nivolumab/chemotherapy arm, and 21% of the chemotherapy arm. Reasons for surgery cancellation generally included patients declining surgery, unresectable disease, and poor lung function. “Cancellation of surgery due to neoadjuvant therapy toxicity was rare,” Dr. Spicer said in his presentation.
Among patients who did proceed to surgery, the median duration of the procedure was 184 minutes in the nivolumab/chemotherapy arm and 217 minutes in the chemotherapy arm. That half-hour difference in favor of the combination arm suggests that the complexity of surgery was not increased by the addition of nivolumab, Dr. Spicer said.
Median time to surgery was about 5 weeks in both arms, which was “well within accepted standards for a neoadjuvant therapeutic approach,” Dr. Spicer said. Most delays beyond 6 weeks were due to administrative issues, and occurred in similar proportions (21% of the nivolumab/chemotherapy arm and 18% of the chemotherapy arm).
The addition of nivolumab to chemotherapy improved pCR rates regardless of baseline stage of disease, according to Dr. Spicer. Furthermore, the depth of pathological regression in the primary tumor was “dramatically different” across stage groupings, he said. Median residual viable tumor percentage in stage IB/II patients was 28% for nivolumab/chemotherapy and 79% for chemotherapy, and in stage IIIA patients, it was 8% for nivolumab/chemotherapy and 70% for chemotherapy.
Overall, thoracotomy was the most frequent surgical approach in this international phase 3 trial, Dr. Spicer said. However, among patients with stage IIIA disease, minimally invasive approaches were used 30% of the time in the nivolumab/chemotherapy arm and 19% in the chemotherapy arm. Conversely, the rate of conversion from a minimally invasive to open approach in patients with stage IIIA disease was 11% for nivolumab/chemotherapy and 20% for chemotherapy alone.
Lobectomy was more frequent in the nivolumab/chemotherapy arm (77%) compared to the chemotherapy arm (61%), a difference that Dr. Spicer described as clinically important. He said the difference appears to be attributable to a lower rate of pneumonectomy in the nivolumab/chemotherapy arm (17%) than in the chemotherapy arm (25%).
Despite less extensive lung resection being required, the rate of R0 resection was numerically higher in the nivolumab/chemotherapy arm (83%) than in the chemotherapy arm (78%), said Dr. Spicer.
Length of hospital stay was “within expected ranges” from geographic regions represented in the trial, Dr. Spicer said. Median length of stay was 4.0 and 6.0 days, respectively, for nivolumab/chemotherapy and chemotherapy alone in North America, 9.5 and 13.0 days in Europe, and 11.0 and 13.0 days in Asia.
Likewise, 90-day surgical complications were well within expected ranges, according to the investigator, with anemia, pain, and wound complications being the most commonly reported. Rates were generally similar between study arms, other than a twofold higher rate of pain in the chemotherapy arm, possibly due to the lower rate of minimally invasive surgery or higher rate of conversion to an open procedure, compared to the nivolumab/chemotherapy arm, he said.
Awaiting survival
Rates of 30- and 90-day mortality are expected to be evaluated when survival endpoints are available, according to Dr. Spicer. Beyond pCR rate, event-free survival is also a primary endpoint of the study, while overall survival is a secondary endpoint.
The study was supported by Bristol Myers Squibb. Dr. Spicer reported disclosures related to AstraZeneca, Bristol Myers Squibb Foundation, Merck, and Roche. Dr. Rusch reported research funding with Genelux and Genentech, and travel expenses from Intuitive Surgical.
The addition of nivolumab to neoadjuvant chemotherapy did not impede the feasibility or timing of surgery in patients with resectable lung cancer, according to results from the phase 3 CheckMate 816 trial.
Adding nivolumab to chemotherapy was tolerable and did not increase the rate of surgical complications, investigator Jonathan Spicer, FRCPC, MD, PhD, of McGill University, Montreal, said in his presentation at the annual meeting of the American Society of Clinical Oncology.
His presentation comes about 2 months after the reporting of primary endpoint results of CheckMate 816 (NCT02998528). CheckMate 816 demonstrated that adding nivolumab to neoadjuvant chemotherapy significantly improved pathological complete response (pCR) in patients with resectable non–small cell lung cancer (NSCLC), according to results presented earlier at the American Association for Cancer Research annual meeting.
“The safety and surgical outcome data reported thus far from CheckMate 816, along with significant improvement in pathological complete response, support nivolumab in combination with chemotherapy as an attractive neoadjuvant option for patients with resectable NSCLC,” said Dr. Spicer (Abstract 8503).
Building on previous experience
The CheckMate 816 study builds on extensive experience in advanced NSCLC that has consistently shown better outcomes, including overall survival, with combinations of chemotherapy and immuno-oncology (IO) agents, compared to chemotherapy alone, said discussant Valerie W. Rusch, MD, of Memorial Sloan Kettering Cancer Center in New York.
Dr. Rusch called out “salient and interesting results” regarding surgical management in CheckMate 816, including a lower rate of surgery cancellations and shorter surgical duration in the chemotherapy-plus-IO arm, compared to the chemotherapy-alone arm.
Furthermore, fewer patients required a pneumonectomy and more patients had a complete resection in the chemotherapy-plus-IO arm, compared to chemotherapy alone, she noted.
“These excellent surgical results, along with the data previously presented at AACR regarding the primary endpoint, help to establish a new standard of neoadjuvant care,” Dr. Rusch said in her presentation.
Study details
CheckMate 816 included 358 patients with newly diagnosed, resectable, stage IB-IIIA NSCLC, Eastern Cooperative Oncology Group (ECOG) performance status of 0-1, and no known EGFR mutations or ALK alterations. Patients were randomized to receive nivolumab and platinum-doublet chemotherapy (nivolumab/chemotherapy) or chemotherapy alone every 3 weeks, with surgery to be performed within 6 weeks of the last dose of neoadjuvant treatment.
The median age of patients was 64 years in the nivolumab/chemotherapy arm and 65 years in the chemotherapy-alone arm. About one-third of patients had ECOG performance status of one, and about half had squamous tumor histology, Dr. Spicer said in his report. Almost two-thirds of patients had stage IIIA disease.
In the study results previously presented at the AACR meeting, both pCR and major pathologic response were significantly better following neoadjuvant chemotherapy and IO treatment, compared to chemotherapy alone.
In the intention-to-treat analysis, 24.0% of patients treated with nivolumab/chemotherapy achieved a pCR, compared to 2.2% in the chemotherapy arm, amounting to an approximate 12-fold increase in pCR, Dr. Spicer said. Similarly, the rate of major pathologic response in the intention-to-treat analysis was 36.9% and 8.9% for the nivolumab/chemotherapy and chemotherapy arms, respectively.
Surgical results
In his ASCO presentation, Dr. Spicer reported that definitive surgery was canceled in 16% of patients in the nivolumab/chemotherapy arm, and 21% of the chemotherapy arm. Reasons for surgery cancellation generally included patients declining surgery, unresectable disease, and poor lung function. “Cancellation of surgery due to neoadjuvant therapy toxicity was rare,” Dr. Spicer said in his presentation.
Among patients who did proceed to surgery, the median duration of the procedure was 184 minutes in the nivolumab/chemotherapy arm and 217 minutes in the chemotherapy arm. That half-hour difference in favor of the combination arm suggests that the complexity of surgery was not increased by the addition of nivolumab, Dr. Spicer said.
Median time to surgery was about 5 weeks in both arms, which was “well within accepted standards for a neoadjuvant therapeutic approach,” Dr. Spicer said. Most delays beyond 6 weeks were due to administrative issues, and occurred in similar proportions (21% of the nivolumab/chemotherapy arm and 18% of the chemotherapy arm).
The addition of nivolumab to chemotherapy improved pCR rates regardless of baseline stage of disease, according to Dr. Spicer. Furthermore, the depth of pathological regression in the primary tumor was “dramatically different” across stage groupings, he said. Median residual viable tumor percentage in stage IB/II patients was 28% for nivolumab/chemotherapy and 79% for chemotherapy, and in stage IIIA patients, it was 8% for nivolumab/chemotherapy and 70% for chemotherapy.
Overall, thoracotomy was the most frequent surgical approach in this international phase 3 trial, Dr. Spicer said. However, among patients with stage IIIA disease, minimally invasive approaches were used 30% of the time in the nivolumab/chemotherapy arm and 19% in the chemotherapy arm. Conversely, the rate of conversion from a minimally invasive to open approach in patients with stage IIIA disease was 11% for nivolumab/chemotherapy and 20% for chemotherapy alone.
Lobectomy was more frequent in the nivolumab/chemotherapy arm (77%) compared to the chemotherapy arm (61%), a difference that Dr. Spicer described as clinically important. He said the difference appears to be attributable to a lower rate of pneumonectomy in the nivolumab/chemotherapy arm (17%) than in the chemotherapy arm (25%).
Despite less extensive lung resection being required, the rate of R0 resection was numerically higher in the nivolumab/chemotherapy arm (83%) than in the chemotherapy arm (78%), said Dr. Spicer.
Length of hospital stay was “within expected ranges” from geographic regions represented in the trial, Dr. Spicer said. Median length of stay was 4.0 and 6.0 days, respectively, for nivolumab/chemotherapy and chemotherapy alone in North America, 9.5 and 13.0 days in Europe, and 11.0 and 13.0 days in Asia.
Likewise, 90-day surgical complications were well within expected ranges, according to the investigator, with anemia, pain, and wound complications being the most commonly reported. Rates were generally similar between study arms, other than a twofold higher rate of pain in the chemotherapy arm, possibly due to the lower rate of minimally invasive surgery or higher rate of conversion to an open procedure, compared to the nivolumab/chemotherapy arm, he said.
Awaiting survival
Rates of 30- and 90-day mortality are expected to be evaluated when survival endpoints are available, according to Dr. Spicer. Beyond pCR rate, event-free survival is also a primary endpoint of the study, while overall survival is a secondary endpoint.
The study was supported by Bristol Myers Squibb. Dr. Spicer reported disclosures related to AstraZeneca, Bristol Myers Squibb Foundation, Merck, and Roche. Dr. Rusch reported research funding with Genelux and Genentech, and travel expenses from Intuitive Surgical.
FROM ASCO 2021