User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
FDA okays transcatheter pulmonary valve for congenital heart disease
The Food and Drug Administration has approved Medtronic’s Harmony Transcatheter Pulmonary Valve (TPV) System to treat severe pulmonary regurgitation in pediatric and adult patients who have a native or surgically repaired right ventricular outflow tract (RVOT).
The Harmony TPV is the first nonsurgical heart valve to treat severe pulmonary valve regurgitation, which is common in patients with congenital heart disease, the agency said in a news release. Its use can delay the time before a patient needs open-heart surgery and potentially reduce the number of these surgeries required over a lifetime.
“The Harmony TPV provides a new treatment option for adult and pediatric patients with certain types of congenital heart disease,” Bram Zuckerman, MD, director of the Office of Cardiovascular Devices in the FDA’s Center for Devices and Radiological Health, said in the statement.
“It offers a less-invasive treatment alternative to open-heart surgery to patients with a leaky native or surgically repaired RVOT and may help patients improve their quality of life and return to their normal activities more quickly, thus fulfilling an unmet clinical need of many patients with congenital heart disease,” he said.
The Harmony valve, which was granted breakthrough device designation, is a 22-mm or 25-mm porcine pericardium valve, sewn to a nitinol frame. It is implanted with a 25-French delivery system using a coil-loading catheter.
The FDA approval was based on the 70-patient prospective, nonrandomized, multicenter Harmony TPV Clinical study, in which 100% of patients achieved the primary safety endpoint of no procedure or device-related deaths 30 days after implantation.
Among 65 patients with evaluable echocardiographic data, 89.2% met the primary effectiveness endpoint of no additional surgical or interventional device-related procedures and acceptable heart blood flow at 6 months.
Adverse events included irregular or abnormal heart rhythms in 23.9% of patients, including 14.1% ventricular tachycardia; leakage around the valve in 8.5%, including 1.4% major leakage; minor bleeding in 7.0%, narrowing of the pulmonary valve in 4.2%, and movement of the implant in 4.2%.
Follow-up was scheduled annually through 5 years and has been extended to 10 years as part of the postapproval study, the FDA noted.
The Harmony TPV device is contraindicated for patients with an infection in the heart or elsewhere, for patients who cannot tolerate blood-thinning medicines, and for those with a sensitivity to nitinol (titanium or nickel).
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved Medtronic’s Harmony Transcatheter Pulmonary Valve (TPV) System to treat severe pulmonary regurgitation in pediatric and adult patients who have a native or surgically repaired right ventricular outflow tract (RVOT).
The Harmony TPV is the first nonsurgical heart valve to treat severe pulmonary valve regurgitation, which is common in patients with congenital heart disease, the agency said in a news release. Its use can delay the time before a patient needs open-heart surgery and potentially reduce the number of these surgeries required over a lifetime.
“The Harmony TPV provides a new treatment option for adult and pediatric patients with certain types of congenital heart disease,” Bram Zuckerman, MD, director of the Office of Cardiovascular Devices in the FDA’s Center for Devices and Radiological Health, said in the statement.
“It offers a less-invasive treatment alternative to open-heart surgery to patients with a leaky native or surgically repaired RVOT and may help patients improve their quality of life and return to their normal activities more quickly, thus fulfilling an unmet clinical need of many patients with congenital heart disease,” he said.
The Harmony valve, which was granted breakthrough device designation, is a 22-mm or 25-mm porcine pericardium valve, sewn to a nitinol frame. It is implanted with a 25-French delivery system using a coil-loading catheter.
The FDA approval was based on the 70-patient prospective, nonrandomized, multicenter Harmony TPV Clinical study, in which 100% of patients achieved the primary safety endpoint of no procedure or device-related deaths 30 days after implantation.
Among 65 patients with evaluable echocardiographic data, 89.2% met the primary effectiveness endpoint of no additional surgical or interventional device-related procedures and acceptable heart blood flow at 6 months.
Adverse events included irregular or abnormal heart rhythms in 23.9% of patients, including 14.1% ventricular tachycardia; leakage around the valve in 8.5%, including 1.4% major leakage; minor bleeding in 7.0%, narrowing of the pulmonary valve in 4.2%, and movement of the implant in 4.2%.
Follow-up was scheduled annually through 5 years and has been extended to 10 years as part of the postapproval study, the FDA noted.
The Harmony TPV device is contraindicated for patients with an infection in the heart or elsewhere, for patients who cannot tolerate blood-thinning medicines, and for those with a sensitivity to nitinol (titanium or nickel).
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved Medtronic’s Harmony Transcatheter Pulmonary Valve (TPV) System to treat severe pulmonary regurgitation in pediatric and adult patients who have a native or surgically repaired right ventricular outflow tract (RVOT).
The Harmony TPV is the first nonsurgical heart valve to treat severe pulmonary valve regurgitation, which is common in patients with congenital heart disease, the agency said in a news release. Its use can delay the time before a patient needs open-heart surgery and potentially reduce the number of these surgeries required over a lifetime.
“The Harmony TPV provides a new treatment option for adult and pediatric patients with certain types of congenital heart disease,” Bram Zuckerman, MD, director of the Office of Cardiovascular Devices in the FDA’s Center for Devices and Radiological Health, said in the statement.
“It offers a less-invasive treatment alternative to open-heart surgery to patients with a leaky native or surgically repaired RVOT and may help patients improve their quality of life and return to their normal activities more quickly, thus fulfilling an unmet clinical need of many patients with congenital heart disease,” he said.
The Harmony valve, which was granted breakthrough device designation, is a 22-mm or 25-mm porcine pericardium valve, sewn to a nitinol frame. It is implanted with a 25-French delivery system using a coil-loading catheter.
The FDA approval was based on the 70-patient prospective, nonrandomized, multicenter Harmony TPV Clinical study, in which 100% of patients achieved the primary safety endpoint of no procedure or device-related deaths 30 days after implantation.
Among 65 patients with evaluable echocardiographic data, 89.2% met the primary effectiveness endpoint of no additional surgical or interventional device-related procedures and acceptable heart blood flow at 6 months.
Adverse events included irregular or abnormal heart rhythms in 23.9% of patients, including 14.1% ventricular tachycardia; leakage around the valve in 8.5%, including 1.4% major leakage; minor bleeding in 7.0%, narrowing of the pulmonary valve in 4.2%, and movement of the implant in 4.2%.
Follow-up was scheduled annually through 5 years and has been extended to 10 years as part of the postapproval study, the FDA noted.
The Harmony TPV device is contraindicated for patients with an infection in the heart or elsewhere, for patients who cannot tolerate blood-thinning medicines, and for those with a sensitivity to nitinol (titanium or nickel).
A version of this article first appeared on Medscape.com.
Step therapy: Inside the fight against insurance companies and fail-first medicine
Every day Melissa Fulton, RN, MSN, FNP, APRN-C, shows up to work, she’s ready for another fight. An advanced practice nurse who specializes in multiple sclerosis care, Ms. Fulton said she typically spends more than a third of her time battling it out with insurance companies over drugs she knows her patients need but that insurers don’t want to cover. Instead, they want the patient to first receive less expensive and often less efficacious drugs, even if that goes against recommendations and, in some cases, against the patient’s medical history.
The maddening protocol – familiar to health care providers everywhere – is known as “step therapy.” It forces patients to try alternative medications – medications that often fail – before receiving the one initially prescribed. The process can take weeks or months, which is time that some patients don’t have. Step therapy was sold as a way to lower costs. However, beyond the ethically problematic notion of forcing sick patients to receiver cheaper alternatives that are ineffective, research has also shown it may actually be more costly in the long run.
Ms. Fulton, who works at Saunders Medical Center in Wahoo, Neb., is a veteran in the war against step therapy. She is used to pushing her appeals up the insurance company chain of command, past nonmedical reviewers, until her patient’s case finally lands on the desk of someone with a neurology background. She said that can take three or four appeals – a judge might even get involved – and the patient could still lose. “This happens constantly,” she said, “but we fight like hell.”
Fortunately, life may soon get a little easier for Ms. Fulton. In late March, a bill to restrict step therapy made it through the Nebraska state legislature and is on its way to the governor’s desk. The Step Therapy Reform Act doesn’t outright ban the practice; however, it will put guardrails in place. It requires that insurers respond to appeals within certain time frames, and it creates key exemptions.
When the governor signs off, Nebraska will join more than two dozen other states that already have step therapy restrictions on the books, according to Hannah Lynch, MPS, associate director of federal government relations and health policy at the National Psoriasis Foundation, a leading advocate to reform and protect against the insurance practice. “There’s a lot of frustration out there,” Ms. Lynch said. “It really hinders providers’ ability to make decisions they think will have the best outcomes.”
Driven by coalitions of doctors, nurses, and patients, laws reining in step therapy have been adopted at a relatively quick clip, mostly within the past 5 years. Recent additions include South Dakota and North Carolina, which adopted step therapy laws in 2020, and Arkansas, which passed a law earlier this year.
Ms. Lynch attributed growing support to rising out-of-pocket drug costs and the introduction of biologic drugs, which are often more effective but also more expensive. Like Nebraska’s law, most step therapy reform legislation carves out exemptions and requires timely appeals processes; however, many of the laws still have significant gaps, such as not including certain types of insurance plans.
Ideally, Ms. Lynch said, the protections would apply to all types of health plans that are regulated at the state level, such as Medicaid, state employee health plans, and coverage sold through state insurance exchanges. Closing loopholes in the laws is a top priority for advocates, she added, pointing to work currently underway in Arkansas to extend its new protections to Medicaid expansion patients.
“With so many outside stakeholders, you have to compromise – it’s a give and take,” Ms. Lynch said. Still, when it comes to fighting step therapy, she says, “Any protection on the books is always our first goal when we go into a state.”
Putting patients first
Lisa Arkin, MD, a pediatric dermatologist at the University of Wisconsin–Madison, said she finds herself “swimming upstream every day in the fight with insurance.” Her patients are typically on their second or third stop and have more complex disorders. Dr. Arkin said that the problem with step therapy is that it tries to squeeze all patients into the same box, even if the circumstances don’t fit.
Her state passed restrictions on step therapy in 2019, but the measures only went into effect last year. Under the Wisconsin law, patients can be granted an exemption if an alternative treatment is contraindicated, likely to cause harm, or expected to be ineffective. Patients can also be exempt if their current treatment is working.
Dr. Arkin, an outspoken advocate for curbing step therapy, says the Wisconsin law is “very strong.” However, because it only applies to certain health plans – state employee health plans and those purchased in the state’s health insurance exchange – fewer than half the state’s patients benefit from its protections. She notes that some of the most severe presentations she treats occur in patients who rely on Medicaid coverage and already face barriers to care.
“I’m a doctor who puts up a fuss [with insurers], but that’s not fair – we shouldn’t have to do that,” Dr. Arkin said. “To me, it’s really critical to make this an even playing field so this law affords protection to everyone I see in the clinic.”
Major medical associations caution against step therapy as well. The American Society of Clinical Oncology and the American Medical Association have called out the risks to patient safety and health. In fact, in 2019, after the Centers for Medicare & Medicaid Services gave new authority to Medicare Advantage plans to start using step therapy, dozens of national medical groups called out the agency for allowing a practice that could potentially hurt patients and undercut the physician-patient decision-making process.
Last year, in a new position paper from the American College of Physicians, authors laid out recommendations for combating step therapy’s side effects. These recommendations included making related data transparent to the public and minimizing the policy’s disruptions to care. Jacqueline W. Fincher, MD, MACP, a member of the committee that issued the position paper and who is a primary care physician in Georgia, said such insurance practices need to be designed with “strong input from frontline physicians, not clipboard physicians.
“What we want from insurers is understanding, transparency, and the least burdensome protocol to provide patients the care they need at a cost-effective price they can afford,” said Dr. Fincher, who is also the current president of the ACP. “The focus needs to be on what’s in the patient’s best interest.”
Every year a new fight
“We all dread January,” said Dr. Fincher. That is the worst month, she added, because new health benefits go into effect, which means patients who are responding well to certain treatments may suddenly face new restrictions.
Another aggravating aspect of step therapy? It is often difficult – if not impossible – to access information on specific step therapy protocols in a patient’s health plan in real time in the exam room, where treatment conversations actually take place. In a more patient-centered world, Dr. Fincher said, she would be able to use the electronic health record system to quickly identify whether a patient’s plan covers a particular treatment and, if not, what the alternatives are.
Georgia’s new step therapy law went into effect last year. Like laws in other states, it spells out step therapy exemptions and sets time frames in which insurers must respond to exceptions and appeals. Dr. Fincher, who spoke in favor of the new law, said she’s “happy for any step forward.” Still, the growing burden of prior authorization rules are an utter “time sink” for her and her staff.
“I have to justify my decisions to nondoctors before I even get to a doctor, and that’s really frustrating,” she said. “We’re talking about people here, not widgets.”
Advocates in Nevada are hoping this is the year a step therapy bill will make it into law in their state. As of March, one had yet to be introduced in the state legislature. Tom McCoy, director of state government affairs at the Nevada Chronic Care Collaborative, said existing Nevada law already prohibits nonmedical drug switching during a policy year; however, insurers can still make changes the following year.
A bill to rein in step therapy was proposed previously, Mr. McCoy said, but it never got off the ground. The collaborative, as well as about two dozen organizations representing Nevada providers and patients, are now calling on state lawmakers to make the issue a priority in the current session.
“The health plans have a lot of power – a lot,” Mr. McCoy said. “We’re hoping to get a [legislative] sponsor in 2021 ... but it’s also been a really hard year to connect legislators with patients and doctors, and being able to hear their stories really does make a difference.”
In Nebraska, Marcus Snow, MD, a rheumatologist at Nebraska Medicine, in Omaha, said that the state’s new step therapy law will be a “great first step in helping to provide some guardrails” around the practice. He noted that turnaround requirements for insurer responses are “sorely needed.” However, he said that, because the bill doesn’t apply to all health plans, many Nebraskans still won’t benefit.
Dealing with step therapy is a daily “headache” for Dr. Snow, who says navigating the bureaucracy of prior authorization seems to be getting worse every year. Like his peers around the country, he spends an inordinate amount of time pushing appeals up the insurance company ranks to get access to treatments he believes will be most effective. But Snow says that, more than just being a mountain of tiresome red tape, these practices also intrude on the patient-provider relationship, casting an unsettling sense of uncertainty that the ultimate decision about the best course of action isn’t up to the doctor and patient at all.
“In the end, the insurance company is the judge and jury of my prescription,” Dr. Snow said. “They’d argue I can still prescribe it, but if it costs $70,000 a year – I don’t know who can afford that.”
Ms. Lynch, at the National Psoriasis Foundation, said their step therapy advocacy will continue to take a two-pronged approach. They will push for new and expanded protections at both state and federal levels. Protections are needed at both levels to make sure that all health plans regulated by all entities are covered. In the U.S. Senate and the House, step therapy bills were reintroduced this year. They would apply to health plans subject to the federal Employee Retirement Income Security Act, which governs employer-sponsored health coverage, and could close a big gap in existing protections. Oregon, New Jersey, and Arizona are at the top of the foundation’s advocacy list this year, according to Ms. Lynch.
“Folks are really starting to pay more attention to this issue,” she said. “And hearing those real-world stories and frustrations is definitely one of the most effective tools we have.”
A version of this article first appeared on Medscape.com.
Every day Melissa Fulton, RN, MSN, FNP, APRN-C, shows up to work, she’s ready for another fight. An advanced practice nurse who specializes in multiple sclerosis care, Ms. Fulton said she typically spends more than a third of her time battling it out with insurance companies over drugs she knows her patients need but that insurers don’t want to cover. Instead, they want the patient to first receive less expensive and often less efficacious drugs, even if that goes against recommendations and, in some cases, against the patient’s medical history.
The maddening protocol – familiar to health care providers everywhere – is known as “step therapy.” It forces patients to try alternative medications – medications that often fail – before receiving the one initially prescribed. The process can take weeks or months, which is time that some patients don’t have. Step therapy was sold as a way to lower costs. However, beyond the ethically problematic notion of forcing sick patients to receiver cheaper alternatives that are ineffective, research has also shown it may actually be more costly in the long run.
Ms. Fulton, who works at Saunders Medical Center in Wahoo, Neb., is a veteran in the war against step therapy. She is used to pushing her appeals up the insurance company chain of command, past nonmedical reviewers, until her patient’s case finally lands on the desk of someone with a neurology background. She said that can take three or four appeals – a judge might even get involved – and the patient could still lose. “This happens constantly,” she said, “but we fight like hell.”
Fortunately, life may soon get a little easier for Ms. Fulton. In late March, a bill to restrict step therapy made it through the Nebraska state legislature and is on its way to the governor’s desk. The Step Therapy Reform Act doesn’t outright ban the practice; however, it will put guardrails in place. It requires that insurers respond to appeals within certain time frames, and it creates key exemptions.
When the governor signs off, Nebraska will join more than two dozen other states that already have step therapy restrictions on the books, according to Hannah Lynch, MPS, associate director of federal government relations and health policy at the National Psoriasis Foundation, a leading advocate to reform and protect against the insurance practice. “There’s a lot of frustration out there,” Ms. Lynch said. “It really hinders providers’ ability to make decisions they think will have the best outcomes.”
Driven by coalitions of doctors, nurses, and patients, laws reining in step therapy have been adopted at a relatively quick clip, mostly within the past 5 years. Recent additions include South Dakota and North Carolina, which adopted step therapy laws in 2020, and Arkansas, which passed a law earlier this year.
Ms. Lynch attributed growing support to rising out-of-pocket drug costs and the introduction of biologic drugs, which are often more effective but also more expensive. Like Nebraska’s law, most step therapy reform legislation carves out exemptions and requires timely appeals processes; however, many of the laws still have significant gaps, such as not including certain types of insurance plans.
Ideally, Ms. Lynch said, the protections would apply to all types of health plans that are regulated at the state level, such as Medicaid, state employee health plans, and coverage sold through state insurance exchanges. Closing loopholes in the laws is a top priority for advocates, she added, pointing to work currently underway in Arkansas to extend its new protections to Medicaid expansion patients.
“With so many outside stakeholders, you have to compromise – it’s a give and take,” Ms. Lynch said. Still, when it comes to fighting step therapy, she says, “Any protection on the books is always our first goal when we go into a state.”
Putting patients first
Lisa Arkin, MD, a pediatric dermatologist at the University of Wisconsin–Madison, said she finds herself “swimming upstream every day in the fight with insurance.” Her patients are typically on their second or third stop and have more complex disorders. Dr. Arkin said that the problem with step therapy is that it tries to squeeze all patients into the same box, even if the circumstances don’t fit.
Her state passed restrictions on step therapy in 2019, but the measures only went into effect last year. Under the Wisconsin law, patients can be granted an exemption if an alternative treatment is contraindicated, likely to cause harm, or expected to be ineffective. Patients can also be exempt if their current treatment is working.
Dr. Arkin, an outspoken advocate for curbing step therapy, says the Wisconsin law is “very strong.” However, because it only applies to certain health plans – state employee health plans and those purchased in the state’s health insurance exchange – fewer than half the state’s patients benefit from its protections. She notes that some of the most severe presentations she treats occur in patients who rely on Medicaid coverage and already face barriers to care.
“I’m a doctor who puts up a fuss [with insurers], but that’s not fair – we shouldn’t have to do that,” Dr. Arkin said. “To me, it’s really critical to make this an even playing field so this law affords protection to everyone I see in the clinic.”
Major medical associations caution against step therapy as well. The American Society of Clinical Oncology and the American Medical Association have called out the risks to patient safety and health. In fact, in 2019, after the Centers for Medicare & Medicaid Services gave new authority to Medicare Advantage plans to start using step therapy, dozens of national medical groups called out the agency for allowing a practice that could potentially hurt patients and undercut the physician-patient decision-making process.
Last year, in a new position paper from the American College of Physicians, authors laid out recommendations for combating step therapy’s side effects. These recommendations included making related data transparent to the public and minimizing the policy’s disruptions to care. Jacqueline W. Fincher, MD, MACP, a member of the committee that issued the position paper and who is a primary care physician in Georgia, said such insurance practices need to be designed with “strong input from frontline physicians, not clipboard physicians.
“What we want from insurers is understanding, transparency, and the least burdensome protocol to provide patients the care they need at a cost-effective price they can afford,” said Dr. Fincher, who is also the current president of the ACP. “The focus needs to be on what’s in the patient’s best interest.”
Every year a new fight
“We all dread January,” said Dr. Fincher. That is the worst month, she added, because new health benefits go into effect, which means patients who are responding well to certain treatments may suddenly face new restrictions.
Another aggravating aspect of step therapy? It is often difficult – if not impossible – to access information on specific step therapy protocols in a patient’s health plan in real time in the exam room, where treatment conversations actually take place. In a more patient-centered world, Dr. Fincher said, she would be able to use the electronic health record system to quickly identify whether a patient’s plan covers a particular treatment and, if not, what the alternatives are.
Georgia’s new step therapy law went into effect last year. Like laws in other states, it spells out step therapy exemptions and sets time frames in which insurers must respond to exceptions and appeals. Dr. Fincher, who spoke in favor of the new law, said she’s “happy for any step forward.” Still, the growing burden of prior authorization rules are an utter “time sink” for her and her staff.
“I have to justify my decisions to nondoctors before I even get to a doctor, and that’s really frustrating,” she said. “We’re talking about people here, not widgets.”
Advocates in Nevada are hoping this is the year a step therapy bill will make it into law in their state. As of March, one had yet to be introduced in the state legislature. Tom McCoy, director of state government affairs at the Nevada Chronic Care Collaborative, said existing Nevada law already prohibits nonmedical drug switching during a policy year; however, insurers can still make changes the following year.
A bill to rein in step therapy was proposed previously, Mr. McCoy said, but it never got off the ground. The collaborative, as well as about two dozen organizations representing Nevada providers and patients, are now calling on state lawmakers to make the issue a priority in the current session.
“The health plans have a lot of power – a lot,” Mr. McCoy said. “We’re hoping to get a [legislative] sponsor in 2021 ... but it’s also been a really hard year to connect legislators with patients and doctors, and being able to hear their stories really does make a difference.”
In Nebraska, Marcus Snow, MD, a rheumatologist at Nebraska Medicine, in Omaha, said that the state’s new step therapy law will be a “great first step in helping to provide some guardrails” around the practice. He noted that turnaround requirements for insurer responses are “sorely needed.” However, he said that, because the bill doesn’t apply to all health plans, many Nebraskans still won’t benefit.
Dealing with step therapy is a daily “headache” for Dr. Snow, who says navigating the bureaucracy of prior authorization seems to be getting worse every year. Like his peers around the country, he spends an inordinate amount of time pushing appeals up the insurance company ranks to get access to treatments he believes will be most effective. But Snow says that, more than just being a mountain of tiresome red tape, these practices also intrude on the patient-provider relationship, casting an unsettling sense of uncertainty that the ultimate decision about the best course of action isn’t up to the doctor and patient at all.
“In the end, the insurance company is the judge and jury of my prescription,” Dr. Snow said. “They’d argue I can still prescribe it, but if it costs $70,000 a year – I don’t know who can afford that.”
Ms. Lynch, at the National Psoriasis Foundation, said their step therapy advocacy will continue to take a two-pronged approach. They will push for new and expanded protections at both state and federal levels. Protections are needed at both levels to make sure that all health plans regulated by all entities are covered. In the U.S. Senate and the House, step therapy bills were reintroduced this year. They would apply to health plans subject to the federal Employee Retirement Income Security Act, which governs employer-sponsored health coverage, and could close a big gap in existing protections. Oregon, New Jersey, and Arizona are at the top of the foundation’s advocacy list this year, according to Ms. Lynch.
“Folks are really starting to pay more attention to this issue,” she said. “And hearing those real-world stories and frustrations is definitely one of the most effective tools we have.”
A version of this article first appeared on Medscape.com.
Every day Melissa Fulton, RN, MSN, FNP, APRN-C, shows up to work, she’s ready for another fight. An advanced practice nurse who specializes in multiple sclerosis care, Ms. Fulton said she typically spends more than a third of her time battling it out with insurance companies over drugs she knows her patients need but that insurers don’t want to cover. Instead, they want the patient to first receive less expensive and often less efficacious drugs, even if that goes against recommendations and, in some cases, against the patient’s medical history.
The maddening protocol – familiar to health care providers everywhere – is known as “step therapy.” It forces patients to try alternative medications – medications that often fail – before receiving the one initially prescribed. The process can take weeks or months, which is time that some patients don’t have. Step therapy was sold as a way to lower costs. However, beyond the ethically problematic notion of forcing sick patients to receiver cheaper alternatives that are ineffective, research has also shown it may actually be more costly in the long run.
Ms. Fulton, who works at Saunders Medical Center in Wahoo, Neb., is a veteran in the war against step therapy. She is used to pushing her appeals up the insurance company chain of command, past nonmedical reviewers, until her patient’s case finally lands on the desk of someone with a neurology background. She said that can take three or four appeals – a judge might even get involved – and the patient could still lose. “This happens constantly,” she said, “but we fight like hell.”
Fortunately, life may soon get a little easier for Ms. Fulton. In late March, a bill to restrict step therapy made it through the Nebraska state legislature and is on its way to the governor’s desk. The Step Therapy Reform Act doesn’t outright ban the practice; however, it will put guardrails in place. It requires that insurers respond to appeals within certain time frames, and it creates key exemptions.
When the governor signs off, Nebraska will join more than two dozen other states that already have step therapy restrictions on the books, according to Hannah Lynch, MPS, associate director of federal government relations and health policy at the National Psoriasis Foundation, a leading advocate to reform and protect against the insurance practice. “There’s a lot of frustration out there,” Ms. Lynch said. “It really hinders providers’ ability to make decisions they think will have the best outcomes.”
Driven by coalitions of doctors, nurses, and patients, laws reining in step therapy have been adopted at a relatively quick clip, mostly within the past 5 years. Recent additions include South Dakota and North Carolina, which adopted step therapy laws in 2020, and Arkansas, which passed a law earlier this year.
Ms. Lynch attributed growing support to rising out-of-pocket drug costs and the introduction of biologic drugs, which are often more effective but also more expensive. Like Nebraska’s law, most step therapy reform legislation carves out exemptions and requires timely appeals processes; however, many of the laws still have significant gaps, such as not including certain types of insurance plans.
Ideally, Ms. Lynch said, the protections would apply to all types of health plans that are regulated at the state level, such as Medicaid, state employee health plans, and coverage sold through state insurance exchanges. Closing loopholes in the laws is a top priority for advocates, she added, pointing to work currently underway in Arkansas to extend its new protections to Medicaid expansion patients.
“With so many outside stakeholders, you have to compromise – it’s a give and take,” Ms. Lynch said. Still, when it comes to fighting step therapy, she says, “Any protection on the books is always our first goal when we go into a state.”
Putting patients first
Lisa Arkin, MD, a pediatric dermatologist at the University of Wisconsin–Madison, said she finds herself “swimming upstream every day in the fight with insurance.” Her patients are typically on their second or third stop and have more complex disorders. Dr. Arkin said that the problem with step therapy is that it tries to squeeze all patients into the same box, even if the circumstances don’t fit.
Her state passed restrictions on step therapy in 2019, but the measures only went into effect last year. Under the Wisconsin law, patients can be granted an exemption if an alternative treatment is contraindicated, likely to cause harm, or expected to be ineffective. Patients can also be exempt if their current treatment is working.
Dr. Arkin, an outspoken advocate for curbing step therapy, says the Wisconsin law is “very strong.” However, because it only applies to certain health plans – state employee health plans and those purchased in the state’s health insurance exchange – fewer than half the state’s patients benefit from its protections. She notes that some of the most severe presentations she treats occur in patients who rely on Medicaid coverage and already face barriers to care.
“I’m a doctor who puts up a fuss [with insurers], but that’s not fair – we shouldn’t have to do that,” Dr. Arkin said. “To me, it’s really critical to make this an even playing field so this law affords protection to everyone I see in the clinic.”
Major medical associations caution against step therapy as well. The American Society of Clinical Oncology and the American Medical Association have called out the risks to patient safety and health. In fact, in 2019, after the Centers for Medicare & Medicaid Services gave new authority to Medicare Advantage plans to start using step therapy, dozens of national medical groups called out the agency for allowing a practice that could potentially hurt patients and undercut the physician-patient decision-making process.
Last year, in a new position paper from the American College of Physicians, authors laid out recommendations for combating step therapy’s side effects. These recommendations included making related data transparent to the public and minimizing the policy’s disruptions to care. Jacqueline W. Fincher, MD, MACP, a member of the committee that issued the position paper and who is a primary care physician in Georgia, said such insurance practices need to be designed with “strong input from frontline physicians, not clipboard physicians.
“What we want from insurers is understanding, transparency, and the least burdensome protocol to provide patients the care they need at a cost-effective price they can afford,” said Dr. Fincher, who is also the current president of the ACP. “The focus needs to be on what’s in the patient’s best interest.”
Every year a new fight
“We all dread January,” said Dr. Fincher. That is the worst month, she added, because new health benefits go into effect, which means patients who are responding well to certain treatments may suddenly face new restrictions.
Another aggravating aspect of step therapy? It is often difficult – if not impossible – to access information on specific step therapy protocols in a patient’s health plan in real time in the exam room, where treatment conversations actually take place. In a more patient-centered world, Dr. Fincher said, she would be able to use the electronic health record system to quickly identify whether a patient’s plan covers a particular treatment and, if not, what the alternatives are.
Georgia’s new step therapy law went into effect last year. Like laws in other states, it spells out step therapy exemptions and sets time frames in which insurers must respond to exceptions and appeals. Dr. Fincher, who spoke in favor of the new law, said she’s “happy for any step forward.” Still, the growing burden of prior authorization rules are an utter “time sink” for her and her staff.
“I have to justify my decisions to nondoctors before I even get to a doctor, and that’s really frustrating,” she said. “We’re talking about people here, not widgets.”
Advocates in Nevada are hoping this is the year a step therapy bill will make it into law in their state. As of March, one had yet to be introduced in the state legislature. Tom McCoy, director of state government affairs at the Nevada Chronic Care Collaborative, said existing Nevada law already prohibits nonmedical drug switching during a policy year; however, insurers can still make changes the following year.
A bill to rein in step therapy was proposed previously, Mr. McCoy said, but it never got off the ground. The collaborative, as well as about two dozen organizations representing Nevada providers and patients, are now calling on state lawmakers to make the issue a priority in the current session.
“The health plans have a lot of power – a lot,” Mr. McCoy said. “We’re hoping to get a [legislative] sponsor in 2021 ... but it’s also been a really hard year to connect legislators with patients and doctors, and being able to hear their stories really does make a difference.”
In Nebraska, Marcus Snow, MD, a rheumatologist at Nebraska Medicine, in Omaha, said that the state’s new step therapy law will be a “great first step in helping to provide some guardrails” around the practice. He noted that turnaround requirements for insurer responses are “sorely needed.” However, he said that, because the bill doesn’t apply to all health plans, many Nebraskans still won’t benefit.
Dealing with step therapy is a daily “headache” for Dr. Snow, who says navigating the bureaucracy of prior authorization seems to be getting worse every year. Like his peers around the country, he spends an inordinate amount of time pushing appeals up the insurance company ranks to get access to treatments he believes will be most effective. But Snow says that, more than just being a mountain of tiresome red tape, these practices also intrude on the patient-provider relationship, casting an unsettling sense of uncertainty that the ultimate decision about the best course of action isn’t up to the doctor and patient at all.
“In the end, the insurance company is the judge and jury of my prescription,” Dr. Snow said. “They’d argue I can still prescribe it, but if it costs $70,000 a year – I don’t know who can afford that.”
Ms. Lynch, at the National Psoriasis Foundation, said their step therapy advocacy will continue to take a two-pronged approach. They will push for new and expanded protections at both state and federal levels. Protections are needed at both levels to make sure that all health plans regulated by all entities are covered. In the U.S. Senate and the House, step therapy bills were reintroduced this year. They would apply to health plans subject to the federal Employee Retirement Income Security Act, which governs employer-sponsored health coverage, and could close a big gap in existing protections. Oregon, New Jersey, and Arizona are at the top of the foundation’s advocacy list this year, according to Ms. Lynch.
“Folks are really starting to pay more attention to this issue,” she said. “And hearing those real-world stories and frustrations is definitely one of the most effective tools we have.”
A version of this article first appeared on Medscape.com.
Encephalopathy common, often lethal in hospitalized patients with COVID-19
, new research shows. Results of a retrospective study show that of almost 4,500 patients with COVID-19, 12% were diagnosed with TME. Of these, 78% developed encephalopathy immediately prior to hospital admission. Septic encephalopathy, hypoxic-ischemic encephalopathy (HIE), and uremia were the most common causes, although multiple causes were present in close to 80% of patients. TME was also associated with a 24% higher risk of in-hospital death.
“We found that close to one in eight patients who were hospitalized with COVID-19 had TME that was not attributed to the effects of sedatives, and that this is incredibly common among these patients who are critically ill” said lead author Jennifer A. Frontera, MD, New York University.
“The general principle of our findings is to be more aggressive in TME; and from a neurologist perspective, the way to do this is to eliminate the effects of sedation, which is a confounder,” she said.
The study was published online March 16 in Neurocritical Care.
Drilling down
“Many neurological complications of COVID-19 are sequelae of severe illness or secondary effects of multisystem organ failure, but our previous work identified TME as the most common neurological complication,” Dr. Frontera said.
Previous research investigating encephalopathy among patients with COVID-19 included patients who may have been sedated or have had a positive Confusion Assessment Method (CAM) result.
“A lot of the delirium literature is effectively heterogeneous because there are a number of patients who are on sedative medication that, if you could turn it off, these patients would return to normal. Some may have underlying neurological issues that can be addressed, but you can›t get to the bottom of this unless you turn off the sedation,” Dr. Frontera noted.
“We wanted to be specific and try to drill down to see what the underlying cause of the encephalopathy was,” she said.
The researchers retrospectively analyzed data on 4,491 patients (≥ 18 years old) with COVID-19 who were admitted to four New York City hospitals between March 1, 2020, and May 20, 2020. Of these, 559 (12%) with TME were compared with 3,932 patients without TME.
The researchers looked at index admissions and included patients who had:
- New changes in mental status or significant worsening of mental status (in patients with baseline abnormal mental status).
- Hyperglycemia or with transient focal neurologic deficits that resolved with glucose correction.
- An adequate washout of sedating medications (when relevant) prior to mental status assessment.
Potential etiologies included electrolyte abnormalities, organ failure, hypertensive encephalopathy, sepsis or active infection, fever, nutritional deficiency, and environmental injury.
Foreign environment
Most (78%) of the 559 patients diagnosed with TME had already developed encephalopathy immediately prior to hospital admission, the authors report. The most common etiologies of TME among hospitalized patients with COVID-19 are listed below.
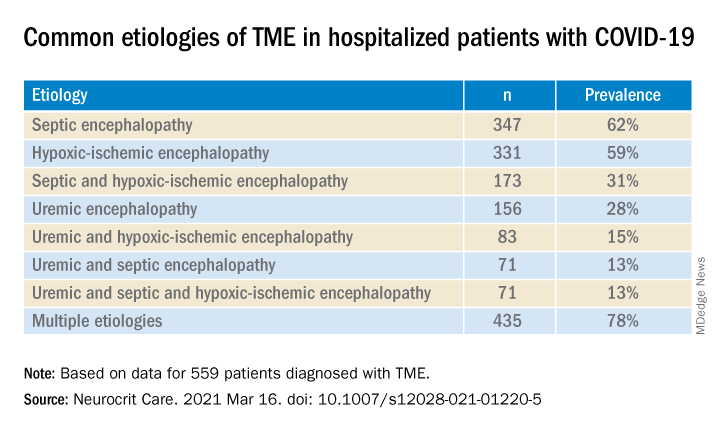
Compared with patients without TME, those with TME – (all Ps < .001):
- Were older (76 vs. 62 years).
- Had higher rates of dementia (27% vs. 3%).
- Had higher rates of psychiatric history (20% vs. 10%).
- Were more often intubated (37% vs. 20%).
- Had a longer length of hospital stay (7.9 vs. 6.0 days).
- Were less often discharged home (25% vs. 66%).
“It’s no surprise that older patients and people with dementia or psychiatric illness are predisposed to becoming encephalopathic,” said Dr. Frontera. “Being in a foreign environment, such as a hospital, or being sleep-deprived in the ICU is likely to make them more confused during their hospital stay.”
Delirium as a symptom
In-hospital mortality or discharge to hospice was considerably higher in the TME versus non-TME patients (44% vs. 18%, respectively).
When the researchers adjusted for confounders (age, sex, race, worse Sequential Organ Failure Assessment score during hospitalization, ventilator status, study week, hospital location, and ICU care level) and excluded patients receiving only comfort care, they found that TME was associated with a 24% increased risk of in-hospital death (30% in patients with TME vs. 16% in those without TME).
The highest mortality risk was associated with hypoxemia, with 42% of patients with HIE dying during hospitalization, compared with 16% of patients without HIE (adjusted hazard ratio 1.56; 95% confidence interval, 1.21-2.00; P = .001).
“Not all patients who are intubated require sedation, but there’s generally a lot of hesitation in reducing or stopping sedation in some patients,” Dr. Frontera observed.
She acknowledged there are “many extremely sick patients whom you can’t ventilate without sedation.”
Nevertheless, “delirium in and of itself does not cause death. It’s a symptom, not a disease, and we have to figure out what causes it. Delirium might not need to be sedated, and it’s more important to see what the causal problem is.”
Independent predictor of death
Commenting on the study, Panayiotis N. Varelas, MD, PhD, vice president of the Neurocritical Care Society, said the study “approached the TME issue better than previously, namely allowing time for sedatives to wear off to have a better sample of patients with this syndrome.”
Dr. Varelas, who is chairman of the department of neurology and professor of neurology at Albany (N.Y.) Medical College, emphasized that TME “is not benign and, in patients with COVID-19, it is an independent predictor of in-hospital mortality.”
“One should take all possible measures … to avoid desaturation and hypotensive episodes and also aggressively treat SAE and uremic encephalopathy in hopes of improving the outcomes,” added Dr. Varelas, who was not involved with the study.
Also commenting on the study, Mitchell Elkind, MD, professor of neurology and epidemiology at Columbia University in New York, who was not associated with the research, said it “nicely distinguishes among the different causes of encephalopathy, including sepsis, hypoxia, and kidney failure … emphasizing just how sick these patients are.”
The study received no direct funding. Individual investigators were supported by grants from the National Institute on Aging and the National Institute of Neurological Disorders and Stroke. The investigators, Dr. Varelas, and Dr. Elkind have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows. Results of a retrospective study show that of almost 4,500 patients with COVID-19, 12% were diagnosed with TME. Of these, 78% developed encephalopathy immediately prior to hospital admission. Septic encephalopathy, hypoxic-ischemic encephalopathy (HIE), and uremia were the most common causes, although multiple causes were present in close to 80% of patients. TME was also associated with a 24% higher risk of in-hospital death.
“We found that close to one in eight patients who were hospitalized with COVID-19 had TME that was not attributed to the effects of sedatives, and that this is incredibly common among these patients who are critically ill” said lead author Jennifer A. Frontera, MD, New York University.
“The general principle of our findings is to be more aggressive in TME; and from a neurologist perspective, the way to do this is to eliminate the effects of sedation, which is a confounder,” she said.
The study was published online March 16 in Neurocritical Care.
Drilling down
“Many neurological complications of COVID-19 are sequelae of severe illness or secondary effects of multisystem organ failure, but our previous work identified TME as the most common neurological complication,” Dr. Frontera said.
Previous research investigating encephalopathy among patients with COVID-19 included patients who may have been sedated or have had a positive Confusion Assessment Method (CAM) result.
“A lot of the delirium literature is effectively heterogeneous because there are a number of patients who are on sedative medication that, if you could turn it off, these patients would return to normal. Some may have underlying neurological issues that can be addressed, but you can›t get to the bottom of this unless you turn off the sedation,” Dr. Frontera noted.
“We wanted to be specific and try to drill down to see what the underlying cause of the encephalopathy was,” she said.
The researchers retrospectively analyzed data on 4,491 patients (≥ 18 years old) with COVID-19 who were admitted to four New York City hospitals between March 1, 2020, and May 20, 2020. Of these, 559 (12%) with TME were compared with 3,932 patients without TME.
The researchers looked at index admissions and included patients who had:
- New changes in mental status or significant worsening of mental status (in patients with baseline abnormal mental status).
- Hyperglycemia or with transient focal neurologic deficits that resolved with glucose correction.
- An adequate washout of sedating medications (when relevant) prior to mental status assessment.
Potential etiologies included electrolyte abnormalities, organ failure, hypertensive encephalopathy, sepsis or active infection, fever, nutritional deficiency, and environmental injury.
Foreign environment
Most (78%) of the 559 patients diagnosed with TME had already developed encephalopathy immediately prior to hospital admission, the authors report. The most common etiologies of TME among hospitalized patients with COVID-19 are listed below.

Compared with patients without TME, those with TME – (all Ps < .001):
- Were older (76 vs. 62 years).
- Had higher rates of dementia (27% vs. 3%).
- Had higher rates of psychiatric history (20% vs. 10%).
- Were more often intubated (37% vs. 20%).
- Had a longer length of hospital stay (7.9 vs. 6.0 days).
- Were less often discharged home (25% vs. 66%).
“It’s no surprise that older patients and people with dementia or psychiatric illness are predisposed to becoming encephalopathic,” said Dr. Frontera. “Being in a foreign environment, such as a hospital, or being sleep-deprived in the ICU is likely to make them more confused during their hospital stay.”
Delirium as a symptom
In-hospital mortality or discharge to hospice was considerably higher in the TME versus non-TME patients (44% vs. 18%, respectively).
When the researchers adjusted for confounders (age, sex, race, worse Sequential Organ Failure Assessment score during hospitalization, ventilator status, study week, hospital location, and ICU care level) and excluded patients receiving only comfort care, they found that TME was associated with a 24% increased risk of in-hospital death (30% in patients with TME vs. 16% in those without TME).
The highest mortality risk was associated with hypoxemia, with 42% of patients with HIE dying during hospitalization, compared with 16% of patients without HIE (adjusted hazard ratio 1.56; 95% confidence interval, 1.21-2.00; P = .001).
“Not all patients who are intubated require sedation, but there’s generally a lot of hesitation in reducing or stopping sedation in some patients,” Dr. Frontera observed.
She acknowledged there are “many extremely sick patients whom you can’t ventilate without sedation.”
Nevertheless, “delirium in and of itself does not cause death. It’s a symptom, not a disease, and we have to figure out what causes it. Delirium might not need to be sedated, and it’s more important to see what the causal problem is.”
Independent predictor of death
Commenting on the study, Panayiotis N. Varelas, MD, PhD, vice president of the Neurocritical Care Society, said the study “approached the TME issue better than previously, namely allowing time for sedatives to wear off to have a better sample of patients with this syndrome.”
Dr. Varelas, who is chairman of the department of neurology and professor of neurology at Albany (N.Y.) Medical College, emphasized that TME “is not benign and, in patients with COVID-19, it is an independent predictor of in-hospital mortality.”
“One should take all possible measures … to avoid desaturation and hypotensive episodes and also aggressively treat SAE and uremic encephalopathy in hopes of improving the outcomes,” added Dr. Varelas, who was not involved with the study.
Also commenting on the study, Mitchell Elkind, MD, professor of neurology and epidemiology at Columbia University in New York, who was not associated with the research, said it “nicely distinguishes among the different causes of encephalopathy, including sepsis, hypoxia, and kidney failure … emphasizing just how sick these patients are.”
The study received no direct funding. Individual investigators were supported by grants from the National Institute on Aging and the National Institute of Neurological Disorders and Stroke. The investigators, Dr. Varelas, and Dr. Elkind have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows. Results of a retrospective study show that of almost 4,500 patients with COVID-19, 12% were diagnosed with TME. Of these, 78% developed encephalopathy immediately prior to hospital admission. Septic encephalopathy, hypoxic-ischemic encephalopathy (HIE), and uremia were the most common causes, although multiple causes were present in close to 80% of patients. TME was also associated with a 24% higher risk of in-hospital death.
“We found that close to one in eight patients who were hospitalized with COVID-19 had TME that was not attributed to the effects of sedatives, and that this is incredibly common among these patients who are critically ill” said lead author Jennifer A. Frontera, MD, New York University.
“The general principle of our findings is to be more aggressive in TME; and from a neurologist perspective, the way to do this is to eliminate the effects of sedation, which is a confounder,” she said.
The study was published online March 16 in Neurocritical Care.
Drilling down
“Many neurological complications of COVID-19 are sequelae of severe illness or secondary effects of multisystem organ failure, but our previous work identified TME as the most common neurological complication,” Dr. Frontera said.
Previous research investigating encephalopathy among patients with COVID-19 included patients who may have been sedated or have had a positive Confusion Assessment Method (CAM) result.
“A lot of the delirium literature is effectively heterogeneous because there are a number of patients who are on sedative medication that, if you could turn it off, these patients would return to normal. Some may have underlying neurological issues that can be addressed, but you can›t get to the bottom of this unless you turn off the sedation,” Dr. Frontera noted.
“We wanted to be specific and try to drill down to see what the underlying cause of the encephalopathy was,” she said.
The researchers retrospectively analyzed data on 4,491 patients (≥ 18 years old) with COVID-19 who were admitted to four New York City hospitals between March 1, 2020, and May 20, 2020. Of these, 559 (12%) with TME were compared with 3,932 patients without TME.
The researchers looked at index admissions and included patients who had:
- New changes in mental status or significant worsening of mental status (in patients with baseline abnormal mental status).
- Hyperglycemia or with transient focal neurologic deficits that resolved with glucose correction.
- An adequate washout of sedating medications (when relevant) prior to mental status assessment.
Potential etiologies included electrolyte abnormalities, organ failure, hypertensive encephalopathy, sepsis or active infection, fever, nutritional deficiency, and environmental injury.
Foreign environment
Most (78%) of the 559 patients diagnosed with TME had already developed encephalopathy immediately prior to hospital admission, the authors report. The most common etiologies of TME among hospitalized patients with COVID-19 are listed below.

Compared with patients without TME, those with TME – (all Ps < .001):
- Were older (76 vs. 62 years).
- Had higher rates of dementia (27% vs. 3%).
- Had higher rates of psychiatric history (20% vs. 10%).
- Were more often intubated (37% vs. 20%).
- Had a longer length of hospital stay (7.9 vs. 6.0 days).
- Were less often discharged home (25% vs. 66%).
“It’s no surprise that older patients and people with dementia or psychiatric illness are predisposed to becoming encephalopathic,” said Dr. Frontera. “Being in a foreign environment, such as a hospital, or being sleep-deprived in the ICU is likely to make them more confused during their hospital stay.”
Delirium as a symptom
In-hospital mortality or discharge to hospice was considerably higher in the TME versus non-TME patients (44% vs. 18%, respectively).
When the researchers adjusted for confounders (age, sex, race, worse Sequential Organ Failure Assessment score during hospitalization, ventilator status, study week, hospital location, and ICU care level) and excluded patients receiving only comfort care, they found that TME was associated with a 24% increased risk of in-hospital death (30% in patients with TME vs. 16% in those without TME).
The highest mortality risk was associated with hypoxemia, with 42% of patients with HIE dying during hospitalization, compared with 16% of patients without HIE (adjusted hazard ratio 1.56; 95% confidence interval, 1.21-2.00; P = .001).
“Not all patients who are intubated require sedation, but there’s generally a lot of hesitation in reducing or stopping sedation in some patients,” Dr. Frontera observed.
She acknowledged there are “many extremely sick patients whom you can’t ventilate without sedation.”
Nevertheless, “delirium in and of itself does not cause death. It’s a symptom, not a disease, and we have to figure out what causes it. Delirium might not need to be sedated, and it’s more important to see what the causal problem is.”
Independent predictor of death
Commenting on the study, Panayiotis N. Varelas, MD, PhD, vice president of the Neurocritical Care Society, said the study “approached the TME issue better than previously, namely allowing time for sedatives to wear off to have a better sample of patients with this syndrome.”
Dr. Varelas, who is chairman of the department of neurology and professor of neurology at Albany (N.Y.) Medical College, emphasized that TME “is not benign and, in patients with COVID-19, it is an independent predictor of in-hospital mortality.”
“One should take all possible measures … to avoid desaturation and hypotensive episodes and also aggressively treat SAE and uremic encephalopathy in hopes of improving the outcomes,” added Dr. Varelas, who was not involved with the study.
Also commenting on the study, Mitchell Elkind, MD, professor of neurology and epidemiology at Columbia University in New York, who was not associated with the research, said it “nicely distinguishes among the different causes of encephalopathy, including sepsis, hypoxia, and kidney failure … emphasizing just how sick these patients are.”
The study received no direct funding. Individual investigators were supported by grants from the National Institute on Aging and the National Institute of Neurological Disorders and Stroke. The investigators, Dr. Varelas, and Dr. Elkind have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROCRITICAL CARE
STEP 4: Ongoing semaglutide treatment extends weight loss
Weekly injections with the GLP-1 receptor agonist semaglutide helped people maintain, and even increase, their initial weight loss on the agent when they continued treatment beyond 20 weeks in results from an international, multicenter trial with 803 randomized subjects.
The study “reflects what we always see in practice, that when people lose weight their body then fights to regain it. The results underscore this” by showing what happens when people stop the drug, Domenica M. Rubino, MD, reported at the annual meeting of the Endocrine Society.
The STEP 4 study began with 902 obese or higher-risk people with an average body mass index of about 38 kg/m2 who underwent a 20-week, open-label, run-in phase of weekly subcutaneous injections of semaglutide (Ozempic), during which all subjects gradually up-titrated to the study’s maintenance dosage of 2.4 mg/week and allowing investigators to weed out intolerant, noncompliant, or nonresponsive people. After this phase excluded 99 subjects from continuing, and documented that the remaining 803 patients had already lost an average of 11% of their starting weight, the core of the study kicked in by randomizing them 2:1 to either maintain their weekly semaglutide injections for another 48 weeks or change to placebo injections.
After 48 more weeks, the 535 people who continued active semaglutide treatment lost on average an additional 8% of their weight. Meanwhile, the 268 who switched to placebo gained 7% of the weight they had reached at the 20-week point, for a significant between-group weight-loss difference of about 15% for the study’s primary endpoint. Those maintained on semaglutide for the full 68 weeks had a cumulative average weight loss of about 17%, compared with when they first began treatment, Dr. Rubino said. Concurrently with her report, the results also appeared in an article published online in JAMA.
“It’s reassuring that people who remain on this treatment can sustain weight losses of 15%, and in some cases 20% or more. That’s huge,” Dr. Rubino said in an interview. . After 68 weeks, 40% of the people who maintained their semaglutide treatment had lost at least 20% of their weight, compared with when they first started treatment.
“Preventing weight regain following initial weight loss is a well-known major challenge for people who lose weight,” commented John Clark III, MD, PhD, a weight management specialist at the University of Texas Southwestern Medical Center in Dallas who was not involved with the study. The findings from STEP 4 will be “helpful to have a discussion [with weight-loss patients] about the risks and benefits of continuing to take this medication longer than just a few months and if they want to continue taking the medication after they reach their goal weight,” Dr. Clark noted in an interview. “This new information reinforces that treatment continues to be effective after the short term.”
“This is obesity 101. If a treatment is provided that targets mechanisms of obesity, and then the treatment stops, we should not be surprised that weight regain occurs,” commented Ania M. Jastreboff, MD, PhD, codirector of the Yale Center for Weight Management in New Haven, Conn. “It’s tragic to see patients who, after successful weight loss, suffer regain because the treatment by which they lost weight stopped,” she said in an interview.
The STEP 4 study ran at 73 centers in 10 countries during 2018-2020. It enrolled adults without diabetes and with a BMI of at least 30, or at least 27 if they also had at least one weight-related comorbidity such as hypertension, dyslipidemia, or obstructive sleep apnea. Participants averaged about 47 years of age, almost 80% were women, and about 84% were White, including 8% of Hispanic or Latinx ethnicity.
The adverse-event profile was consistent with findings from trials where semaglutide treated hyperglycemia in patients with type 2 diabetes (semaglutide at a maximum once-weekly dosage of 1 mg has Food and Drug Administration approval for controlling hyperglycemia in patients with type 2 diabetes), as well results from other semaglutide studies and from studies of other agents in the GLP-1 receptor agonist class.
In STEP 4 9% of patients who received semaglutide during the randomized phase and 7% of those randomized to placebo had a serious adverse reaction, and about 2% of those in both treatment arms stopped treatment because of an adverse event. The most common adverse events on semaglutide were gastrointestinal, with diarrhea in 14%, nausea in 14%, constipation in 12%, and vomiting in 10%.
These GI effects are often mitigated by slower dose escalation, eating smaller amounts of food at a time, and not eating beyond the point of feeling full, noted Dr. Jastreboff.
The STEP 4 results follow prior reports from three other large trials – STEP 1, STEP 2, and STEP 3 – that studied the weight-loss effects of weekly semaglutide treatment in adults using varying enrollment criteria and treatment designs. “We’ve seen very consistent results [across all four studies] for efficacy and safety,” said Dr. Rubino, who owns and directs the Washington Center for Weight Management & Research in Arlington, Va.
NovoNordisk, the company that markets semaglutide, submitted data from all four studies to the FDA late last year in an application for a new label for a weight loss indication at the 2.4-mg/week dosage. The company has said it expects an agency decision by June 2021.
Dr. Rubino has been an adviser and consultant to and a speaker on behalf of Novo Nordisk, and she has also been an investigator for studies sponsored by AstraZeneca, Boehringer Ingelheim, and Novo Nordisk. Dr. Clark had no disclosures. Dr. Jastreboff is consultant for and has received research funding from NovoNordisk, and she has also been a consultant to and/or received research from Eli Lilly and Boehringer Ingelheim.
Weekly injections with the GLP-1 receptor agonist semaglutide helped people maintain, and even increase, their initial weight loss on the agent when they continued treatment beyond 20 weeks in results from an international, multicenter trial with 803 randomized subjects.
The study “reflects what we always see in practice, that when people lose weight their body then fights to regain it. The results underscore this” by showing what happens when people stop the drug, Domenica M. Rubino, MD, reported at the annual meeting of the Endocrine Society.
The STEP 4 study began with 902 obese or higher-risk people with an average body mass index of about 38 kg/m2 who underwent a 20-week, open-label, run-in phase of weekly subcutaneous injections of semaglutide (Ozempic), during which all subjects gradually up-titrated to the study’s maintenance dosage of 2.4 mg/week and allowing investigators to weed out intolerant, noncompliant, or nonresponsive people. After this phase excluded 99 subjects from continuing, and documented that the remaining 803 patients had already lost an average of 11% of their starting weight, the core of the study kicked in by randomizing them 2:1 to either maintain their weekly semaglutide injections for another 48 weeks or change to placebo injections.
After 48 more weeks, the 535 people who continued active semaglutide treatment lost on average an additional 8% of their weight. Meanwhile, the 268 who switched to placebo gained 7% of the weight they had reached at the 20-week point, for a significant between-group weight-loss difference of about 15% for the study’s primary endpoint. Those maintained on semaglutide for the full 68 weeks had a cumulative average weight loss of about 17%, compared with when they first began treatment, Dr. Rubino said. Concurrently with her report, the results also appeared in an article published online in JAMA.
“It’s reassuring that people who remain on this treatment can sustain weight losses of 15%, and in some cases 20% or more. That’s huge,” Dr. Rubino said in an interview. . After 68 weeks, 40% of the people who maintained their semaglutide treatment had lost at least 20% of their weight, compared with when they first started treatment.
“Preventing weight regain following initial weight loss is a well-known major challenge for people who lose weight,” commented John Clark III, MD, PhD, a weight management specialist at the University of Texas Southwestern Medical Center in Dallas who was not involved with the study. The findings from STEP 4 will be “helpful to have a discussion [with weight-loss patients] about the risks and benefits of continuing to take this medication longer than just a few months and if they want to continue taking the medication after they reach their goal weight,” Dr. Clark noted in an interview. “This new information reinforces that treatment continues to be effective after the short term.”
“This is obesity 101. If a treatment is provided that targets mechanisms of obesity, and then the treatment stops, we should not be surprised that weight regain occurs,” commented Ania M. Jastreboff, MD, PhD, codirector of the Yale Center for Weight Management in New Haven, Conn. “It’s tragic to see patients who, after successful weight loss, suffer regain because the treatment by which they lost weight stopped,” she said in an interview.
The STEP 4 study ran at 73 centers in 10 countries during 2018-2020. It enrolled adults without diabetes and with a BMI of at least 30, or at least 27 if they also had at least one weight-related comorbidity such as hypertension, dyslipidemia, or obstructive sleep apnea. Participants averaged about 47 years of age, almost 80% were women, and about 84% were White, including 8% of Hispanic or Latinx ethnicity.
The adverse-event profile was consistent with findings from trials where semaglutide treated hyperglycemia in patients with type 2 diabetes (semaglutide at a maximum once-weekly dosage of 1 mg has Food and Drug Administration approval for controlling hyperglycemia in patients with type 2 diabetes), as well results from other semaglutide studies and from studies of other agents in the GLP-1 receptor agonist class.
In STEP 4 9% of patients who received semaglutide during the randomized phase and 7% of those randomized to placebo had a serious adverse reaction, and about 2% of those in both treatment arms stopped treatment because of an adverse event. The most common adverse events on semaglutide were gastrointestinal, with diarrhea in 14%, nausea in 14%, constipation in 12%, and vomiting in 10%.
These GI effects are often mitigated by slower dose escalation, eating smaller amounts of food at a time, and not eating beyond the point of feeling full, noted Dr. Jastreboff.
The STEP 4 results follow prior reports from three other large trials – STEP 1, STEP 2, and STEP 3 – that studied the weight-loss effects of weekly semaglutide treatment in adults using varying enrollment criteria and treatment designs. “We’ve seen very consistent results [across all four studies] for efficacy and safety,” said Dr. Rubino, who owns and directs the Washington Center for Weight Management & Research in Arlington, Va.
NovoNordisk, the company that markets semaglutide, submitted data from all four studies to the FDA late last year in an application for a new label for a weight loss indication at the 2.4-mg/week dosage. The company has said it expects an agency decision by June 2021.
Dr. Rubino has been an adviser and consultant to and a speaker on behalf of Novo Nordisk, and she has also been an investigator for studies sponsored by AstraZeneca, Boehringer Ingelheim, and Novo Nordisk. Dr. Clark had no disclosures. Dr. Jastreboff is consultant for and has received research funding from NovoNordisk, and she has also been a consultant to and/or received research from Eli Lilly and Boehringer Ingelheim.
Weekly injections with the GLP-1 receptor agonist semaglutide helped people maintain, and even increase, their initial weight loss on the agent when they continued treatment beyond 20 weeks in results from an international, multicenter trial with 803 randomized subjects.
The study “reflects what we always see in practice, that when people lose weight their body then fights to regain it. The results underscore this” by showing what happens when people stop the drug, Domenica M. Rubino, MD, reported at the annual meeting of the Endocrine Society.
The STEP 4 study began with 902 obese or higher-risk people with an average body mass index of about 38 kg/m2 who underwent a 20-week, open-label, run-in phase of weekly subcutaneous injections of semaglutide (Ozempic), during which all subjects gradually up-titrated to the study’s maintenance dosage of 2.4 mg/week and allowing investigators to weed out intolerant, noncompliant, or nonresponsive people. After this phase excluded 99 subjects from continuing, and documented that the remaining 803 patients had already lost an average of 11% of their starting weight, the core of the study kicked in by randomizing them 2:1 to either maintain their weekly semaglutide injections for another 48 weeks or change to placebo injections.
After 48 more weeks, the 535 people who continued active semaglutide treatment lost on average an additional 8% of their weight. Meanwhile, the 268 who switched to placebo gained 7% of the weight they had reached at the 20-week point, for a significant between-group weight-loss difference of about 15% for the study’s primary endpoint. Those maintained on semaglutide for the full 68 weeks had a cumulative average weight loss of about 17%, compared with when they first began treatment, Dr. Rubino said. Concurrently with her report, the results also appeared in an article published online in JAMA.
“It’s reassuring that people who remain on this treatment can sustain weight losses of 15%, and in some cases 20% or more. That’s huge,” Dr. Rubino said in an interview. . After 68 weeks, 40% of the people who maintained their semaglutide treatment had lost at least 20% of their weight, compared with when they first started treatment.
“Preventing weight regain following initial weight loss is a well-known major challenge for people who lose weight,” commented John Clark III, MD, PhD, a weight management specialist at the University of Texas Southwestern Medical Center in Dallas who was not involved with the study. The findings from STEP 4 will be “helpful to have a discussion [with weight-loss patients] about the risks and benefits of continuing to take this medication longer than just a few months and if they want to continue taking the medication after they reach their goal weight,” Dr. Clark noted in an interview. “This new information reinforces that treatment continues to be effective after the short term.”
“This is obesity 101. If a treatment is provided that targets mechanisms of obesity, and then the treatment stops, we should not be surprised that weight regain occurs,” commented Ania M. Jastreboff, MD, PhD, codirector of the Yale Center for Weight Management in New Haven, Conn. “It’s tragic to see patients who, after successful weight loss, suffer regain because the treatment by which they lost weight stopped,” she said in an interview.
The STEP 4 study ran at 73 centers in 10 countries during 2018-2020. It enrolled adults without diabetes and with a BMI of at least 30, or at least 27 if they also had at least one weight-related comorbidity such as hypertension, dyslipidemia, or obstructive sleep apnea. Participants averaged about 47 years of age, almost 80% were women, and about 84% were White, including 8% of Hispanic or Latinx ethnicity.
The adverse-event profile was consistent with findings from trials where semaglutide treated hyperglycemia in patients with type 2 diabetes (semaglutide at a maximum once-weekly dosage of 1 mg has Food and Drug Administration approval for controlling hyperglycemia in patients with type 2 diabetes), as well results from other semaglutide studies and from studies of other agents in the GLP-1 receptor agonist class.
In STEP 4 9% of patients who received semaglutide during the randomized phase and 7% of those randomized to placebo had a serious adverse reaction, and about 2% of those in both treatment arms stopped treatment because of an adverse event. The most common adverse events on semaglutide were gastrointestinal, with diarrhea in 14%, nausea in 14%, constipation in 12%, and vomiting in 10%.
These GI effects are often mitigated by slower dose escalation, eating smaller amounts of food at a time, and not eating beyond the point of feeling full, noted Dr. Jastreboff.
The STEP 4 results follow prior reports from three other large trials – STEP 1, STEP 2, and STEP 3 – that studied the weight-loss effects of weekly semaglutide treatment in adults using varying enrollment criteria and treatment designs. “We’ve seen very consistent results [across all four studies] for efficacy and safety,” said Dr. Rubino, who owns and directs the Washington Center for Weight Management & Research in Arlington, Va.
NovoNordisk, the company that markets semaglutide, submitted data from all four studies to the FDA late last year in an application for a new label for a weight loss indication at the 2.4-mg/week dosage. The company has said it expects an agency decision by June 2021.
Dr. Rubino has been an adviser and consultant to and a speaker on behalf of Novo Nordisk, and she has also been an investigator for studies sponsored by AstraZeneca, Boehringer Ingelheim, and Novo Nordisk. Dr. Clark had no disclosures. Dr. Jastreboff is consultant for and has received research funding from NovoNordisk, and she has also been a consultant to and/or received research from Eli Lilly and Boehringer Ingelheim.
FROM ENDO 2021
Arcalyst gets FDA nod as first therapy for recurrent pericarditis
The Food and Drug Administration has approved rilonacept (Arcalyst) to treat recurrent pericarditis and reduce the risk for recurrence in adults and children 12 years and older.
Approval of the weekly subcutaneous injection offers patients the first and only FDA-approved therapy for recurrent pericarditis, the agency said in a release.
Recurrent pericarditis is characterized by a remitting relapsing inflammation of the pericardium, and therapeutic options have been limited to NSAIDs, colchicine, and corticosteroids.
Rilonacept is a recombinant fusion protein that blocks interleukin-1 alpha and interleukin-1 beta signaling. It is already approved by the FDA to treat a group of rare inherited inflammatory diseases called cryopyrin-associated periodic syndromes.
The new indication is based on the pivotal phase 3 RHAPSODY trial in 86 patients with acute symptoms of recurrent pericarditis and systemic inflammation. After randomization, pericarditis recurred in 2 of 30 patients (7%) treated with rilonacept and in 23 of 31 patients (74%) treated with placebo, representing a 96% reduction in the relative risk for recurrence with rilonacept.
Patients who received rilonacept were also pain free or had minimal pain on 98% of trial days, whereas those who received placebo had minimal or no pain on 46% of trial days.
The most common adverse effects of rilonacept are injection-site reactions and upper-respiratory tract infections.
Serious, life-threatening infections have been reported in patients taking rilonacept, according to the FDA. Patients with active or chronic infections should not take the drug.
The FDA label also advises that patients should avoid live vaccines while taking rilonacept and that it should be discontinued if a hypersensitivity reaction occurs.
The commercial launch is expected in April, according to the company.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved rilonacept (Arcalyst) to treat recurrent pericarditis and reduce the risk for recurrence in adults and children 12 years and older.
Approval of the weekly subcutaneous injection offers patients the first and only FDA-approved therapy for recurrent pericarditis, the agency said in a release.
Recurrent pericarditis is characterized by a remitting relapsing inflammation of the pericardium, and therapeutic options have been limited to NSAIDs, colchicine, and corticosteroids.
Rilonacept is a recombinant fusion protein that blocks interleukin-1 alpha and interleukin-1 beta signaling. It is already approved by the FDA to treat a group of rare inherited inflammatory diseases called cryopyrin-associated periodic syndromes.
The new indication is based on the pivotal phase 3 RHAPSODY trial in 86 patients with acute symptoms of recurrent pericarditis and systemic inflammation. After randomization, pericarditis recurred in 2 of 30 patients (7%) treated with rilonacept and in 23 of 31 patients (74%) treated with placebo, representing a 96% reduction in the relative risk for recurrence with rilonacept.
Patients who received rilonacept were also pain free or had minimal pain on 98% of trial days, whereas those who received placebo had minimal or no pain on 46% of trial days.
The most common adverse effects of rilonacept are injection-site reactions and upper-respiratory tract infections.
Serious, life-threatening infections have been reported in patients taking rilonacept, according to the FDA. Patients with active or chronic infections should not take the drug.
The FDA label also advises that patients should avoid live vaccines while taking rilonacept and that it should be discontinued if a hypersensitivity reaction occurs.
The commercial launch is expected in April, according to the company.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved rilonacept (Arcalyst) to treat recurrent pericarditis and reduce the risk for recurrence in adults and children 12 years and older.
Approval of the weekly subcutaneous injection offers patients the first and only FDA-approved therapy for recurrent pericarditis, the agency said in a release.
Recurrent pericarditis is characterized by a remitting relapsing inflammation of the pericardium, and therapeutic options have been limited to NSAIDs, colchicine, and corticosteroids.
Rilonacept is a recombinant fusion protein that blocks interleukin-1 alpha and interleukin-1 beta signaling. It is already approved by the FDA to treat a group of rare inherited inflammatory diseases called cryopyrin-associated periodic syndromes.
The new indication is based on the pivotal phase 3 RHAPSODY trial in 86 patients with acute symptoms of recurrent pericarditis and systemic inflammation. After randomization, pericarditis recurred in 2 of 30 patients (7%) treated with rilonacept and in 23 of 31 patients (74%) treated with placebo, representing a 96% reduction in the relative risk for recurrence with rilonacept.
Patients who received rilonacept were also pain free or had minimal pain on 98% of trial days, whereas those who received placebo had minimal or no pain on 46% of trial days.
The most common adverse effects of rilonacept are injection-site reactions and upper-respiratory tract infections.
Serious, life-threatening infections have been reported in patients taking rilonacept, according to the FDA. Patients with active or chronic infections should not take the drug.
The FDA label also advises that patients should avoid live vaccines while taking rilonacept and that it should be discontinued if a hypersensitivity reaction occurs.
The commercial launch is expected in April, according to the company.
A version of this article first appeared on Medscape.com.
In U.S., lockdowns added 2 pounds per month
Americans gained nearly 2 pounds per month under COVID-19 shelter-in-place orders in 2020, according to a new study published March 22, 2021, in JAMA Network Open.
Those who kept the same lockdown habits could have gained 20 pounds during the past year, the study authors said.
“We know that weight gain is a public health problem in the U.S. already, so anything making it worse is definitely concerning, and shelter-in-place orders are so ubiquitous that the sheer number of people affected by this makes it extremely relevant,” Gregory Marcus, MD, the senior author and a cardiologist at the University of California, San Francisco, told the New York Times.
Dr. Marcus and colleagues analyzed more than 7,000 weight measurements from 269 people in 37 states who used Bluetooth-connected scales from Feb. 1 to June 1, 2020. Among the participants, about 52% were women, 77% were White, and they had an average age of 52 years.
The research team found that participants had a steady weight gain of more than half a pound every 10 days. That equals about 1.5-2 pounds per month.
Many of the participants were losing weight before the shelter-in-place orders went into effect, Dr. Marcus said. The lockdown effects could be even greater for those who weren’t losing weight before.
“It’s reasonable to assume these individuals are more engaged with their health in general, and more disciplined and on top of things,” he said. “That suggests we could be underestimating – that this is the tip of the iceberg.”
The small study doesn’t represent all of the nation and can’t be generalized to the U.S. population, the study authors noted, but it’s an indicator of what happened during the pandemic. The participants’ weight increased regardless of their location and chronic medical conditions.
Overall, people don’t move around as much during lockdowns, the UCSF researchers reported in another study published in Annals of Internal Medicine in November 2020. According to smartphone data, daily step counts decreased by 27% in March 2020. The step counts increased again throughout the summer but still remained lower than before the COVID-19 pandemic.
“The detrimental health outcomes suggested by these data demonstrate a need to identify concurrent strategies to mitigate weight gain,” the authors wrote in the JAMA Network Open study, “such as encouraging healthy diets and exploring ways to enhance physical activity, as local governments consider new constraints in response to SARS-CoV-2 and potential future pandemics.”
A version of this article first appeared on WebMD.com.
Americans gained nearly 2 pounds per month under COVID-19 shelter-in-place orders in 2020, according to a new study published March 22, 2021, in JAMA Network Open.
Those who kept the same lockdown habits could have gained 20 pounds during the past year, the study authors said.
“We know that weight gain is a public health problem in the U.S. already, so anything making it worse is definitely concerning, and shelter-in-place orders are so ubiquitous that the sheer number of people affected by this makes it extremely relevant,” Gregory Marcus, MD, the senior author and a cardiologist at the University of California, San Francisco, told the New York Times.
Dr. Marcus and colleagues analyzed more than 7,000 weight measurements from 269 people in 37 states who used Bluetooth-connected scales from Feb. 1 to June 1, 2020. Among the participants, about 52% were women, 77% were White, and they had an average age of 52 years.
The research team found that participants had a steady weight gain of more than half a pound every 10 days. That equals about 1.5-2 pounds per month.
Many of the participants were losing weight before the shelter-in-place orders went into effect, Dr. Marcus said. The lockdown effects could be even greater for those who weren’t losing weight before.
“It’s reasonable to assume these individuals are more engaged with their health in general, and more disciplined and on top of things,” he said. “That suggests we could be underestimating – that this is the tip of the iceberg.”
The small study doesn’t represent all of the nation and can’t be generalized to the U.S. population, the study authors noted, but it’s an indicator of what happened during the pandemic. The participants’ weight increased regardless of their location and chronic medical conditions.
Overall, people don’t move around as much during lockdowns, the UCSF researchers reported in another study published in Annals of Internal Medicine in November 2020. According to smartphone data, daily step counts decreased by 27% in March 2020. The step counts increased again throughout the summer but still remained lower than before the COVID-19 pandemic.
“The detrimental health outcomes suggested by these data demonstrate a need to identify concurrent strategies to mitigate weight gain,” the authors wrote in the JAMA Network Open study, “such as encouraging healthy diets and exploring ways to enhance physical activity, as local governments consider new constraints in response to SARS-CoV-2 and potential future pandemics.”
A version of this article first appeared on WebMD.com.
Americans gained nearly 2 pounds per month under COVID-19 shelter-in-place orders in 2020, according to a new study published March 22, 2021, in JAMA Network Open.
Those who kept the same lockdown habits could have gained 20 pounds during the past year, the study authors said.
“We know that weight gain is a public health problem in the U.S. already, so anything making it worse is definitely concerning, and shelter-in-place orders are so ubiquitous that the sheer number of people affected by this makes it extremely relevant,” Gregory Marcus, MD, the senior author and a cardiologist at the University of California, San Francisco, told the New York Times.
Dr. Marcus and colleagues analyzed more than 7,000 weight measurements from 269 people in 37 states who used Bluetooth-connected scales from Feb. 1 to June 1, 2020. Among the participants, about 52% were women, 77% were White, and they had an average age of 52 years.
The research team found that participants had a steady weight gain of more than half a pound every 10 days. That equals about 1.5-2 pounds per month.
Many of the participants were losing weight before the shelter-in-place orders went into effect, Dr. Marcus said. The lockdown effects could be even greater for those who weren’t losing weight before.
“It’s reasonable to assume these individuals are more engaged with their health in general, and more disciplined and on top of things,” he said. “That suggests we could be underestimating – that this is the tip of the iceberg.”
The small study doesn’t represent all of the nation and can’t be generalized to the U.S. population, the study authors noted, but it’s an indicator of what happened during the pandemic. The participants’ weight increased regardless of their location and chronic medical conditions.
Overall, people don’t move around as much during lockdowns, the UCSF researchers reported in another study published in Annals of Internal Medicine in November 2020. According to smartphone data, daily step counts decreased by 27% in March 2020. The step counts increased again throughout the summer but still remained lower than before the COVID-19 pandemic.
“The detrimental health outcomes suggested by these data demonstrate a need to identify concurrent strategies to mitigate weight gain,” the authors wrote in the JAMA Network Open study, “such as encouraging healthy diets and exploring ways to enhance physical activity, as local governments consider new constraints in response to SARS-CoV-2 and potential future pandemics.”
A version of this article first appeared on WebMD.com.
Vitamin D may protect against COVID-19, especially in Black patients
Higher levels of vitamin D than traditionally considered sufficient may help prevent COVID-19 infection – particularly in Black patients, shows a new single-center, retrospective study looking at the role of vitamin D in prevention of infection.
The study, published recently in JAMA Network Open, noted that expert opinion varies as to what “sufficient” levels of vitamin D are, some define this as 30 ng/mL, while others cite 40 ng/mL or greater.
In their discussion, the authors also noted that their results showed the “risk of positive COVID-19 test results decreased significantly with increased vitamin D level of 30 ng/mL or greater when measured as a continuous variable.”
“These new results tell us that having vitamin D levels above those normally considered sufficient is associated with decreased risk of testing positive for COVID-19, at least in Black individuals,” lead author, David Meltzer, MD, chief of hospital medicine at the University of Chicago, said in a press release from his institution.
“These findings suggest that randomized clinical trials to determine whether increasing vitamin D levels to greater than 30-40 ng/mL affect COVID-19 risk are warranted, especially in Black individuals,” he and his coauthors said.
Vitamin D at time of testing most strongly associated with COVID risk
An earlier study by the same researchers found that vitamin D deficiency (less than 20 ng/mL) may raise the risk of testing positive for COVID-19 in people from various ethnicities, as reported by this news organization.
Data for this latest study were drawn from electronic health records for 4,638 individuals at the University of Chicago Medicine and were used to examine whether the likelihood of a positive COVID-19 test was associated with a person’s most recent vitamin D level (within the previous year), and whether there was any effect of ethnicity on this outcome.
Mean age was 52.8 years, 69% were women, 49% were Black, 43% White, and 8% were another race/ethnicity. A total of 27% of the individuals were deficient in vitamin D (less than 20 ng/mL), 27% had insufficient levels (20-30 ng/mL), 22% had sufficient levels (30-40 ng/mL), and the remaining 24% had levels of 40 ng/mL or greater.
In total, 333 (7%) of people tested positive for COVID-19, including 102 (5%) Whites and 211 (9%) Blacks. And 36% of Black individuals who tested positive for COVID-19 were classified as vitamin D deficient, compared with 16% of Whites.
A positive test result for COVID-19 was not significantly associated with vitamin D levels in white individuals but was in Black individuals.
In Black people, compared with levels of at least 40 ng/mL, vitamin D levels of 30-40 ng/mL were associated with an incidence rate ratio of 2.64 for COVID-19 positivity (P = .01). For levels of 20-30 ng/mL, the IRR was 1.69 (P = 0.21); and for less than 20 ng/mL the IRR was 2.55 (P = .009).
The researchers also found that the risk of positive test results with lower vitamin D levels increased when those levels were lower just prior to the positive COVID-19 test, lending “support [to] the idea that vitamin D level at the time of testing is most strongly associated with COVID-19 risk,” they wrote.
Try upping vitamin D levels to 40 ng/mL or greater to prevent COVID?
In their discussion, the authors noted that significant association of vitamin D levels with COVID-19 risk in Blacks but not in Whites, “could reflect their higher COVID-19 risk, to which socioeconomic factors and structural inequities clearly contribute.
“Biological susceptibility to vitamin D deficiency may also be less frequent in White than Black individuals, since lighter skin increases vitamin D production in response to sunlight, and vitamin D binding proteins may vary by race and affect vitamin D bioavailability.”
Given less than 10% of U.S. adults have a vitamin D level greater than 40 ng/mL, the study findings increase the urgency to consider whether increased sun exposure or supplementation could reduce COVID-19 risk, according to the authors.
“When increased sun exposure is impractical, achieving vitamin D levels of 40 ng/mL or greater typically requires greater supplementation than currently recommended for most individuals of 600-800 IU/d vitamin D3,” they added.
However, Dr. Meltzer also acknowledged that “this is an observational study. We can see that there’s an association between vitamin D levels and likelihood of a COVID-19 diagnosis, but we don’t know exactly why that is, or whether these results are due to the vitamin D directly or other related biological factors.”
All in all, the authors suggested that randomized clinical trials are needed to understand if vitamin D can reduce COVID-19 risk, and as such they should include doses of supplements likely to increase vitamin D to at least 40 ng/mL, and perhaps even higher, although they pointed out that the latter must be achieved safely.
“Studies should also consider the role of vitamin D testing, loading doses, dose adjustments for individuals who are obese or overweight, risks for hypercalcemia, and strategies to monitor for and mitigate hypercalcemia, and that non-White populations, such as Black individuals, may have greater needs for supplementation,” they outlined.
They are now recruiting participants for two separate clinical trials testing the efficacy of vitamin D supplements for preventing COVID-19.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Higher levels of vitamin D than traditionally considered sufficient may help prevent COVID-19 infection – particularly in Black patients, shows a new single-center, retrospective study looking at the role of vitamin D in prevention of infection.
The study, published recently in JAMA Network Open, noted that expert opinion varies as to what “sufficient” levels of vitamin D are, some define this as 30 ng/mL, while others cite 40 ng/mL or greater.
In their discussion, the authors also noted that their results showed the “risk of positive COVID-19 test results decreased significantly with increased vitamin D level of 30 ng/mL or greater when measured as a continuous variable.”
“These new results tell us that having vitamin D levels above those normally considered sufficient is associated with decreased risk of testing positive for COVID-19, at least in Black individuals,” lead author, David Meltzer, MD, chief of hospital medicine at the University of Chicago, said in a press release from his institution.
“These findings suggest that randomized clinical trials to determine whether increasing vitamin D levels to greater than 30-40 ng/mL affect COVID-19 risk are warranted, especially in Black individuals,” he and his coauthors said.
Vitamin D at time of testing most strongly associated with COVID risk
An earlier study by the same researchers found that vitamin D deficiency (less than 20 ng/mL) may raise the risk of testing positive for COVID-19 in people from various ethnicities, as reported by this news organization.
Data for this latest study were drawn from electronic health records for 4,638 individuals at the University of Chicago Medicine and were used to examine whether the likelihood of a positive COVID-19 test was associated with a person’s most recent vitamin D level (within the previous year), and whether there was any effect of ethnicity on this outcome.
Mean age was 52.8 years, 69% were women, 49% were Black, 43% White, and 8% were another race/ethnicity. A total of 27% of the individuals were deficient in vitamin D (less than 20 ng/mL), 27% had insufficient levels (20-30 ng/mL), 22% had sufficient levels (30-40 ng/mL), and the remaining 24% had levels of 40 ng/mL or greater.
In total, 333 (7%) of people tested positive for COVID-19, including 102 (5%) Whites and 211 (9%) Blacks. And 36% of Black individuals who tested positive for COVID-19 were classified as vitamin D deficient, compared with 16% of Whites.
A positive test result for COVID-19 was not significantly associated with vitamin D levels in white individuals but was in Black individuals.
In Black people, compared with levels of at least 40 ng/mL, vitamin D levels of 30-40 ng/mL were associated with an incidence rate ratio of 2.64 for COVID-19 positivity (P = .01). For levels of 20-30 ng/mL, the IRR was 1.69 (P = 0.21); and for less than 20 ng/mL the IRR was 2.55 (P = .009).
The researchers also found that the risk of positive test results with lower vitamin D levels increased when those levels were lower just prior to the positive COVID-19 test, lending “support [to] the idea that vitamin D level at the time of testing is most strongly associated with COVID-19 risk,” they wrote.
Try upping vitamin D levels to 40 ng/mL or greater to prevent COVID?
In their discussion, the authors noted that significant association of vitamin D levels with COVID-19 risk in Blacks but not in Whites, “could reflect their higher COVID-19 risk, to which socioeconomic factors and structural inequities clearly contribute.
“Biological susceptibility to vitamin D deficiency may also be less frequent in White than Black individuals, since lighter skin increases vitamin D production in response to sunlight, and vitamin D binding proteins may vary by race and affect vitamin D bioavailability.”
Given less than 10% of U.S. adults have a vitamin D level greater than 40 ng/mL, the study findings increase the urgency to consider whether increased sun exposure or supplementation could reduce COVID-19 risk, according to the authors.
“When increased sun exposure is impractical, achieving vitamin D levels of 40 ng/mL or greater typically requires greater supplementation than currently recommended for most individuals of 600-800 IU/d vitamin D3,” they added.
However, Dr. Meltzer also acknowledged that “this is an observational study. We can see that there’s an association between vitamin D levels and likelihood of a COVID-19 diagnosis, but we don’t know exactly why that is, or whether these results are due to the vitamin D directly or other related biological factors.”
All in all, the authors suggested that randomized clinical trials are needed to understand if vitamin D can reduce COVID-19 risk, and as such they should include doses of supplements likely to increase vitamin D to at least 40 ng/mL, and perhaps even higher, although they pointed out that the latter must be achieved safely.
“Studies should also consider the role of vitamin D testing, loading doses, dose adjustments for individuals who are obese or overweight, risks for hypercalcemia, and strategies to monitor for and mitigate hypercalcemia, and that non-White populations, such as Black individuals, may have greater needs for supplementation,” they outlined.
They are now recruiting participants for two separate clinical trials testing the efficacy of vitamin D supplements for preventing COVID-19.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Higher levels of vitamin D than traditionally considered sufficient may help prevent COVID-19 infection – particularly in Black patients, shows a new single-center, retrospective study looking at the role of vitamin D in prevention of infection.
The study, published recently in JAMA Network Open, noted that expert opinion varies as to what “sufficient” levels of vitamin D are, some define this as 30 ng/mL, while others cite 40 ng/mL or greater.
In their discussion, the authors also noted that their results showed the “risk of positive COVID-19 test results decreased significantly with increased vitamin D level of 30 ng/mL or greater when measured as a continuous variable.”
“These new results tell us that having vitamin D levels above those normally considered sufficient is associated with decreased risk of testing positive for COVID-19, at least in Black individuals,” lead author, David Meltzer, MD, chief of hospital medicine at the University of Chicago, said in a press release from his institution.
“These findings suggest that randomized clinical trials to determine whether increasing vitamin D levels to greater than 30-40 ng/mL affect COVID-19 risk are warranted, especially in Black individuals,” he and his coauthors said.
Vitamin D at time of testing most strongly associated with COVID risk
An earlier study by the same researchers found that vitamin D deficiency (less than 20 ng/mL) may raise the risk of testing positive for COVID-19 in people from various ethnicities, as reported by this news organization.
Data for this latest study were drawn from electronic health records for 4,638 individuals at the University of Chicago Medicine and were used to examine whether the likelihood of a positive COVID-19 test was associated with a person’s most recent vitamin D level (within the previous year), and whether there was any effect of ethnicity on this outcome.
Mean age was 52.8 years, 69% were women, 49% were Black, 43% White, and 8% were another race/ethnicity. A total of 27% of the individuals were deficient in vitamin D (less than 20 ng/mL), 27% had insufficient levels (20-30 ng/mL), 22% had sufficient levels (30-40 ng/mL), and the remaining 24% had levels of 40 ng/mL or greater.
In total, 333 (7%) of people tested positive for COVID-19, including 102 (5%) Whites and 211 (9%) Blacks. And 36% of Black individuals who tested positive for COVID-19 were classified as vitamin D deficient, compared with 16% of Whites.
A positive test result for COVID-19 was not significantly associated with vitamin D levels in white individuals but was in Black individuals.
In Black people, compared with levels of at least 40 ng/mL, vitamin D levels of 30-40 ng/mL were associated with an incidence rate ratio of 2.64 for COVID-19 positivity (P = .01). For levels of 20-30 ng/mL, the IRR was 1.69 (P = 0.21); and for less than 20 ng/mL the IRR was 2.55 (P = .009).
The researchers also found that the risk of positive test results with lower vitamin D levels increased when those levels were lower just prior to the positive COVID-19 test, lending “support [to] the idea that vitamin D level at the time of testing is most strongly associated with COVID-19 risk,” they wrote.
Try upping vitamin D levels to 40 ng/mL or greater to prevent COVID?
In their discussion, the authors noted that significant association of vitamin D levels with COVID-19 risk in Blacks but not in Whites, “could reflect their higher COVID-19 risk, to which socioeconomic factors and structural inequities clearly contribute.
“Biological susceptibility to vitamin D deficiency may also be less frequent in White than Black individuals, since lighter skin increases vitamin D production in response to sunlight, and vitamin D binding proteins may vary by race and affect vitamin D bioavailability.”
Given less than 10% of U.S. adults have a vitamin D level greater than 40 ng/mL, the study findings increase the urgency to consider whether increased sun exposure or supplementation could reduce COVID-19 risk, according to the authors.
“When increased sun exposure is impractical, achieving vitamin D levels of 40 ng/mL or greater typically requires greater supplementation than currently recommended for most individuals of 600-800 IU/d vitamin D3,” they added.
However, Dr. Meltzer also acknowledged that “this is an observational study. We can see that there’s an association between vitamin D levels and likelihood of a COVID-19 diagnosis, but we don’t know exactly why that is, or whether these results are due to the vitamin D directly or other related biological factors.”
All in all, the authors suggested that randomized clinical trials are needed to understand if vitamin D can reduce COVID-19 risk, and as such they should include doses of supplements likely to increase vitamin D to at least 40 ng/mL, and perhaps even higher, although they pointed out that the latter must be achieved safely.
“Studies should also consider the role of vitamin D testing, loading doses, dose adjustments for individuals who are obese or overweight, risks for hypercalcemia, and strategies to monitor for and mitigate hypercalcemia, and that non-White populations, such as Black individuals, may have greater needs for supplementation,” they outlined.
They are now recruiting participants for two separate clinical trials testing the efficacy of vitamin D supplements for preventing COVID-19.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Change is hard: Lessons from an EHR conversion
During this “go-live,” 5 hospitals and approximately 300 ambulatory service and physician practice locations made the transition, consolidating over 100 disparate electronic systems and dozens of interfaces into one world-class medical record.
If you’ve ever been part of such an event, you know it is anything but simple. On the contrary, it requires an enormous financial investment along with years of planning, hours of meetings, and months of training. No matter how much preparation goes into it, there are sure to be bumps along the way. It is a traumatic and stressful time for all involved, but the end result is well worth the effort. Still, there are lessons to be learned and wisdom to be gleaned, and this month we’d like to share a few that we found most important. We believe that many of these are useful lessons even to those who will never live through a go-live.
Safety always comes first
Patient safety is a term so often used that it has a tendency to be taken for granted. Health systems build processes and procedures to ensure safety – some even win awards and recognition for their efforts. But the best (and safest) health care institutions build patient safety into their cultures. More than just being taught to use checklists or buzzwords, the staff at these institutions are encouraged to put the welfare of patients first, making all other activities secondary to this pursuit. We had the opportunity to witness the benefits of such a culture during this go-live and were incredibly impressed with the results.
To be successful in an EHR transition of any magnitude, an organization needs to hold patient safety as a core value and provide its employees with the tools to execute on that value. This enables staff to prepare adequately and to identify risks and opportunities before the conversion takes place. Once go-live occurs, staff also must feel empowered to speak up when they identify problem areas that might jeopardize patients’ care. They also must be given a clear escalation path to ensure their voices can be heard. Most importantly, everyone must understand that the electronic health record itself is just one piece of a major operational change.
As workflows are modified to adapt to the new technology, unsafe processes should be called out and fixed quickly. While the EHR may offer the latest in decision support and system integration, no advancement in technology can make up for bad outcomes, nor justify processes that lead to patient harm.
Training is no substitute for good support
It takes a long time to train thousands of employees, especially when that training must occur during the era of social distancing in the midst of a pandemic. Still, even in the best of times, education should be married to hands-on experience in order to have a real impact. Unfortunately, this is extremely challenging.
Trainees forget much of what they’ve learned in the weeks or months between education and go-live, so they must be given immediately accessible support to bridge the gap. This is known as “at-the-elbow” (ATE) support, and as the name implies, it consists of individuals who are familiar with the new system and are always available to end users, answering their questions and helping them navigate. Since health care never sleeps, this support needs to be offered 24/7, and it should also be flexible and plentiful.
There are many areas that will require more support than anticipated to accommodate the number of clinical and other staff who will use the system, so support staff must be nimble and available for redeployment. In addition, ensuring high-quality support is essential. As many ATE experts are hired contractors, their knowledge base and communications skills can vary widely. Accountability is key, and end users should feel empowered to identify gaps in coverage and deficits in knowledge base in the ATE.
As employees become more familiar with the new system, the need for ATE will wane, but there will still be questions that arise for many weeks to months, and new EHR users will also be added all the time. A good after–go-live support system should remain available so clinical and clerical employees can get just-in-time assistance whenever they need it.
Users should be given clear expectations
Clinicians going through an EHR conversion may be frustrated to discover that the data transferred from their old system into the new one is not quite what they expected. While structured elements such as allergies and immunizations may transfer, unstructured patient histories may not come over at all.
There may be gaps in data, or the opposite may even be true: an overabundance of useless information may transfer over, leaving doctors with dozens of meaningless data points to sift through and eliminate to clean up the chart. This can be extremely time-consuming and discouraging and may jeopardize the success of the go-live.
Providers deserve clear expectations prior to conversion. They should be told what will and will not transfer and be informed that there will be extra work required for documentation at the outset. They may also want the option to preemptively reduce patient volumes to accommodate the additional effort involved in preparing charts. No matter what, this will be a heavy lift, and physicians should understand the implications long before go-live to prepare accordingly.
Old habits die hard
One of the most common complaints we’ve heard following EHR conversions is that “things just worked better in the old system.” We always respond with a question: “Were things better, or just different?” The truth may lie somewhere in the middle, but there is no question that muscle memory develops over many years, and change is difficult no matter how much better the new system is. Still, appropriate expectations, access to just-in-time support, and a continual focus on safety will ensure that the long-term benefits of a patient-centered and integrated electronic record will far outweigh the initial challenges of go-live.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
During this “go-live,” 5 hospitals and approximately 300 ambulatory service and physician practice locations made the transition, consolidating over 100 disparate electronic systems and dozens of interfaces into one world-class medical record.
If you’ve ever been part of such an event, you know it is anything but simple. On the contrary, it requires an enormous financial investment along with years of planning, hours of meetings, and months of training. No matter how much preparation goes into it, there are sure to be bumps along the way. It is a traumatic and stressful time for all involved, but the end result is well worth the effort. Still, there are lessons to be learned and wisdom to be gleaned, and this month we’d like to share a few that we found most important. We believe that many of these are useful lessons even to those who will never live through a go-live.
Safety always comes first
Patient safety is a term so often used that it has a tendency to be taken for granted. Health systems build processes and procedures to ensure safety – some even win awards and recognition for their efforts. But the best (and safest) health care institutions build patient safety into their cultures. More than just being taught to use checklists or buzzwords, the staff at these institutions are encouraged to put the welfare of patients first, making all other activities secondary to this pursuit. We had the opportunity to witness the benefits of such a culture during this go-live and were incredibly impressed with the results.
To be successful in an EHR transition of any magnitude, an organization needs to hold patient safety as a core value and provide its employees with the tools to execute on that value. This enables staff to prepare adequately and to identify risks and opportunities before the conversion takes place. Once go-live occurs, staff also must feel empowered to speak up when they identify problem areas that might jeopardize patients’ care. They also must be given a clear escalation path to ensure their voices can be heard. Most importantly, everyone must understand that the electronic health record itself is just one piece of a major operational change.
As workflows are modified to adapt to the new technology, unsafe processes should be called out and fixed quickly. While the EHR may offer the latest in decision support and system integration, no advancement in technology can make up for bad outcomes, nor justify processes that lead to patient harm.
Training is no substitute for good support
It takes a long time to train thousands of employees, especially when that training must occur during the era of social distancing in the midst of a pandemic. Still, even in the best of times, education should be married to hands-on experience in order to have a real impact. Unfortunately, this is extremely challenging.
Trainees forget much of what they’ve learned in the weeks or months between education and go-live, so they must be given immediately accessible support to bridge the gap. This is known as “at-the-elbow” (ATE) support, and as the name implies, it consists of individuals who are familiar with the new system and are always available to end users, answering their questions and helping them navigate. Since health care never sleeps, this support needs to be offered 24/7, and it should also be flexible and plentiful.
There are many areas that will require more support than anticipated to accommodate the number of clinical and other staff who will use the system, so support staff must be nimble and available for redeployment. In addition, ensuring high-quality support is essential. As many ATE experts are hired contractors, their knowledge base and communications skills can vary widely. Accountability is key, and end users should feel empowered to identify gaps in coverage and deficits in knowledge base in the ATE.
As employees become more familiar with the new system, the need for ATE will wane, but there will still be questions that arise for many weeks to months, and new EHR users will also be added all the time. A good after–go-live support system should remain available so clinical and clerical employees can get just-in-time assistance whenever they need it.
Users should be given clear expectations
Clinicians going through an EHR conversion may be frustrated to discover that the data transferred from their old system into the new one is not quite what they expected. While structured elements such as allergies and immunizations may transfer, unstructured patient histories may not come over at all.
There may be gaps in data, or the opposite may even be true: an overabundance of useless information may transfer over, leaving doctors with dozens of meaningless data points to sift through and eliminate to clean up the chart. This can be extremely time-consuming and discouraging and may jeopardize the success of the go-live.
Providers deserve clear expectations prior to conversion. They should be told what will and will not transfer and be informed that there will be extra work required for documentation at the outset. They may also want the option to preemptively reduce patient volumes to accommodate the additional effort involved in preparing charts. No matter what, this will be a heavy lift, and physicians should understand the implications long before go-live to prepare accordingly.
Old habits die hard
One of the most common complaints we’ve heard following EHR conversions is that “things just worked better in the old system.” We always respond with a question: “Were things better, or just different?” The truth may lie somewhere in the middle, but there is no question that muscle memory develops over many years, and change is difficult no matter how much better the new system is. Still, appropriate expectations, access to just-in-time support, and a continual focus on safety will ensure that the long-term benefits of a patient-centered and integrated electronic record will far outweigh the initial challenges of go-live.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
During this “go-live,” 5 hospitals and approximately 300 ambulatory service and physician practice locations made the transition, consolidating over 100 disparate electronic systems and dozens of interfaces into one world-class medical record.
If you’ve ever been part of such an event, you know it is anything but simple. On the contrary, it requires an enormous financial investment along with years of planning, hours of meetings, and months of training. No matter how much preparation goes into it, there are sure to be bumps along the way. It is a traumatic and stressful time for all involved, but the end result is well worth the effort. Still, there are lessons to be learned and wisdom to be gleaned, and this month we’d like to share a few that we found most important. We believe that many of these are useful lessons even to those who will never live through a go-live.
Safety always comes first
Patient safety is a term so often used that it has a tendency to be taken for granted. Health systems build processes and procedures to ensure safety – some even win awards and recognition for their efforts. But the best (and safest) health care institutions build patient safety into their cultures. More than just being taught to use checklists or buzzwords, the staff at these institutions are encouraged to put the welfare of patients first, making all other activities secondary to this pursuit. We had the opportunity to witness the benefits of such a culture during this go-live and were incredibly impressed with the results.
To be successful in an EHR transition of any magnitude, an organization needs to hold patient safety as a core value and provide its employees with the tools to execute on that value. This enables staff to prepare adequately and to identify risks and opportunities before the conversion takes place. Once go-live occurs, staff also must feel empowered to speak up when they identify problem areas that might jeopardize patients’ care. They also must be given a clear escalation path to ensure their voices can be heard. Most importantly, everyone must understand that the electronic health record itself is just one piece of a major operational change.
As workflows are modified to adapt to the new technology, unsafe processes should be called out and fixed quickly. While the EHR may offer the latest in decision support and system integration, no advancement in technology can make up for bad outcomes, nor justify processes that lead to patient harm.
Training is no substitute for good support
It takes a long time to train thousands of employees, especially when that training must occur during the era of social distancing in the midst of a pandemic. Still, even in the best of times, education should be married to hands-on experience in order to have a real impact. Unfortunately, this is extremely challenging.
Trainees forget much of what they’ve learned in the weeks or months between education and go-live, so they must be given immediately accessible support to bridge the gap. This is known as “at-the-elbow” (ATE) support, and as the name implies, it consists of individuals who are familiar with the new system and are always available to end users, answering their questions and helping them navigate. Since health care never sleeps, this support needs to be offered 24/7, and it should also be flexible and plentiful.
There are many areas that will require more support than anticipated to accommodate the number of clinical and other staff who will use the system, so support staff must be nimble and available for redeployment. In addition, ensuring high-quality support is essential. As many ATE experts are hired contractors, their knowledge base and communications skills can vary widely. Accountability is key, and end users should feel empowered to identify gaps in coverage and deficits in knowledge base in the ATE.
As employees become more familiar with the new system, the need for ATE will wane, but there will still be questions that arise for many weeks to months, and new EHR users will also be added all the time. A good after–go-live support system should remain available so clinical and clerical employees can get just-in-time assistance whenever they need it.
Users should be given clear expectations
Clinicians going through an EHR conversion may be frustrated to discover that the data transferred from their old system into the new one is not quite what they expected. While structured elements such as allergies and immunizations may transfer, unstructured patient histories may not come over at all.
There may be gaps in data, or the opposite may even be true: an overabundance of useless information may transfer over, leaving doctors with dozens of meaningless data points to sift through and eliminate to clean up the chart. This can be extremely time-consuming and discouraging and may jeopardize the success of the go-live.
Providers deserve clear expectations prior to conversion. They should be told what will and will not transfer and be informed that there will be extra work required for documentation at the outset. They may also want the option to preemptively reduce patient volumes to accommodate the additional effort involved in preparing charts. No matter what, this will be a heavy lift, and physicians should understand the implications long before go-live to prepare accordingly.
Old habits die hard
One of the most common complaints we’ve heard following EHR conversions is that “things just worked better in the old system.” We always respond with a question: “Were things better, or just different?” The truth may lie somewhere in the middle, but there is no question that muscle memory develops over many years, and change is difficult no matter how much better the new system is. Still, appropriate expectations, access to just-in-time support, and a continual focus on safety will ensure that the long-term benefits of a patient-centered and integrated electronic record will far outweigh the initial challenges of go-live.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
Top JAMA editor on leave amid podcast investigation
The American Medical Association’s Joint Oversight Committee announced that Howard Bauchner, MD, is on leave beginning at the end of the day on March 25. Dr. Bauchner is the top editor at JAMA, the journal of the AMA.
“The decision to place the editor-in-chief on administrative leave neither implicates nor exonerates individuals and is standard operating procedure for such investigations,” the committee said in a statement.
More than 2,000 people signed a petition on Change.org calling for an investigation at JAMA over the February podcast episode, called “Structural Racism for Doctors: What Is It?”
Already, Edward H. Livingston, MD, the host of the podcast, has resigned as deputy editor of the journal.
During the podcast, Dr. Livingston, who is White, said, “Structural racism is an unfortunate term. Personally, I think taking racism out of the conversation will help. Many of us are offended by the concept that we are racist.”
The audio of the podcast has been deleted from JAMA’s website. In its place is audio of a statement from Dr. Bauchner. In his statement, which he released in the week prior to his being on leave, he said the comments in the podcast, which also featured Mitch Katz, MD, were “inaccurate, offensive, hurtful, and inconsistent with the standards of JAMA.”
Also deleted was a JAMA tweet promoting the podcast episode. The tweet said: “No physician is racist, so how can there be structural racism in health care? An explanation of the idea by doctors for doctors in this user-friendly podcast.”
This story will be updated.
A version of this article first appeared on WedMD.com.
The American Medical Association’s Joint Oversight Committee announced that Howard Bauchner, MD, is on leave beginning at the end of the day on March 25. Dr. Bauchner is the top editor at JAMA, the journal of the AMA.
“The decision to place the editor-in-chief on administrative leave neither implicates nor exonerates individuals and is standard operating procedure for such investigations,” the committee said in a statement.
More than 2,000 people signed a petition on Change.org calling for an investigation at JAMA over the February podcast episode, called “Structural Racism for Doctors: What Is It?”
Already, Edward H. Livingston, MD, the host of the podcast, has resigned as deputy editor of the journal.
During the podcast, Dr. Livingston, who is White, said, “Structural racism is an unfortunate term. Personally, I think taking racism out of the conversation will help. Many of us are offended by the concept that we are racist.”
The audio of the podcast has been deleted from JAMA’s website. In its place is audio of a statement from Dr. Bauchner. In his statement, which he released in the week prior to his being on leave, he said the comments in the podcast, which also featured Mitch Katz, MD, were “inaccurate, offensive, hurtful, and inconsistent with the standards of JAMA.”
Also deleted was a JAMA tweet promoting the podcast episode. The tweet said: “No physician is racist, so how can there be structural racism in health care? An explanation of the idea by doctors for doctors in this user-friendly podcast.”
This story will be updated.
A version of this article first appeared on WedMD.com.
The American Medical Association’s Joint Oversight Committee announced that Howard Bauchner, MD, is on leave beginning at the end of the day on March 25. Dr. Bauchner is the top editor at JAMA, the journal of the AMA.
“The decision to place the editor-in-chief on administrative leave neither implicates nor exonerates individuals and is standard operating procedure for such investigations,” the committee said in a statement.
More than 2,000 people signed a petition on Change.org calling for an investigation at JAMA over the February podcast episode, called “Structural Racism for Doctors: What Is It?”
Already, Edward H. Livingston, MD, the host of the podcast, has resigned as deputy editor of the journal.
During the podcast, Dr. Livingston, who is White, said, “Structural racism is an unfortunate term. Personally, I think taking racism out of the conversation will help. Many of us are offended by the concept that we are racist.”
The audio of the podcast has been deleted from JAMA’s website. In its place is audio of a statement from Dr. Bauchner. In his statement, which he released in the week prior to his being on leave, he said the comments in the podcast, which also featured Mitch Katz, MD, were “inaccurate, offensive, hurtful, and inconsistent with the standards of JAMA.”
Also deleted was a JAMA tweet promoting the podcast episode. The tweet said: “No physician is racist, so how can there be structural racism in health care? An explanation of the idea by doctors for doctors in this user-friendly podcast.”
This story will be updated.
A version of this article first appeared on WedMD.com.
Low concordance between troponin assays for ACS
Clinicians should be aware that the discordance between high-sensitivity cardiac troponin (hs-cTn) assays is significant enough that management recommendations may change, for example, for a patient assessed for suspected acute coronary syndrome (ACS) in one hospital and transferred to another that uses a different assay, according to a team of international researchers.
When hs-cTn concentrations were measured using the three Food and Drug Administration–approved assays, only 37.4% (384 of 1,027 samples) of blood samples were classified into the same analytical benchmark category.
“We didn’t expect such low concordance, to be honest, but I have to stress that this first assessment used just one-time blood testing and serial testing is what is more commonly recommended now,” said Júlia Karády, MD, from Massachusetts General Hospital and Harvard Medical School, both in Boston.
To see if concordance improved with serial testing, the researchers looked at the 242 patients for whom serial samples were available and saw concordance of management recommendations across assays rise to 74.8%.
“We tested the 0/2-hour algorithm and found that the overall agreement almost doubled, so I think that a very important message from our study is that serial testing improves the agreement between the assays in terms of clinical management and patient stratification,” said Dr. Karády.
Dr. Karády and colleagues published their findings in the Journal of the American College of Cardiology.
The researchers tested three assays referred to clinically as high-sensitivity assays: Elecsys 2010 platform (Roche Diagnostics); ARCHITECT i2000SR (Abbott Diagnostics); and hsVista (Siemens Diagnostics). All three have received FDA approval, starting with Elecsys in 2017.
The proportion of patients with similar management recommendations differed between the assays for both “rule-out” (87.2%, 73.1%, and 78.5% for Roche, Abbott, and Siemens, respectively) and “observe” (9.5%, 24%, and 17.8%; both P < .001). For the purposes of “rule-in,” no difference was noted (3.3%, 2.9%, and 3.7%).
“It’s important to note that this was a highly selected population of patients with an intermediate likelihood for ACS, not an all-comer population. This group comprises about 20% of the [emergency department] population and actually is the group we struggle with the most, which is hardest to diagnose because it excludes the very low– and very high–risk patients,” said Dr. Karády.
The patients included in this study all had suspected ACS and were enrolled in the ROMICAT-I and II trials.
Among 1,027 samples from 624 patients (mean age, 52.8 years; 39.4% women), samples were classified as below the limit of detection (LOD) in 56.3%, 10.4%, and 41.2% (P < .001) by Roche, Abbott, and Siemens, respectively.
The proportion of sample with a troponin measurement between LOD to the 99th percentile also differed significantly between the assays at 36.5%, 83.5%, and 52.6%, respectively (P < .001).
Only the proportion classified greater than 99th percentile did not differ (7.2%, 6.0%, and 6.2%; P = .114).
When the researchers looked at sex-specific difference, no differences were seen in rule-in numbers for men, but significant differences were seen for women.
“One possible explanation for this could be differences in the representation of men and women in the various reference populations used to develop the 99th percentile values for these assays,” suggested Dr. Karády.
They estimate around 30%-40% of U.S. centers are currently using high-sensitivity troponin assays and this number is “rapidly rising.”
The diagnostic algorithms developed for use with high-sensitivity assays, such as the 0/2-h algorithm, acknowledge differences in performance characteristics and recommend that assay-specific cut points be used for clinical decision-making rather than relying on generally applicable thresholds.
Joseph S. Alpert, MD, University of Arizona, Tucson, and coauthors of an accompanying editorial said the take-home message here is caveat emptor.
“First, ‘let the buyer (i.e., the clinician) beware’ when patients are transferred from one hospital to another, where different hs-cTn assays may be used,” they wrote. This is particularly true in women and in those with troponin levels in the “observe (gray zone)” clinical management recommendation.
Dr. Karády has received grant support from the Fulbright Visiting Researcher Grant and the Rosztoczy Foundation. One of the coauthors of the editorial comment consults or has consulted for most of the major diagnostic companies, including the manufacturers of the three assays tested in this study. Dr. Alpert disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clinicians should be aware that the discordance between high-sensitivity cardiac troponin (hs-cTn) assays is significant enough that management recommendations may change, for example, for a patient assessed for suspected acute coronary syndrome (ACS) in one hospital and transferred to another that uses a different assay, according to a team of international researchers.
When hs-cTn concentrations were measured using the three Food and Drug Administration–approved assays, only 37.4% (384 of 1,027 samples) of blood samples were classified into the same analytical benchmark category.
“We didn’t expect such low concordance, to be honest, but I have to stress that this first assessment used just one-time blood testing and serial testing is what is more commonly recommended now,” said Júlia Karády, MD, from Massachusetts General Hospital and Harvard Medical School, both in Boston.
To see if concordance improved with serial testing, the researchers looked at the 242 patients for whom serial samples were available and saw concordance of management recommendations across assays rise to 74.8%.
“We tested the 0/2-hour algorithm and found that the overall agreement almost doubled, so I think that a very important message from our study is that serial testing improves the agreement between the assays in terms of clinical management and patient stratification,” said Dr. Karády.
Dr. Karády and colleagues published their findings in the Journal of the American College of Cardiology.
The researchers tested three assays referred to clinically as high-sensitivity assays: Elecsys 2010 platform (Roche Diagnostics); ARCHITECT i2000SR (Abbott Diagnostics); and hsVista (Siemens Diagnostics). All three have received FDA approval, starting with Elecsys in 2017.
The proportion of patients with similar management recommendations differed between the assays for both “rule-out” (87.2%, 73.1%, and 78.5% for Roche, Abbott, and Siemens, respectively) and “observe” (9.5%, 24%, and 17.8%; both P < .001). For the purposes of “rule-in,” no difference was noted (3.3%, 2.9%, and 3.7%).
“It’s important to note that this was a highly selected population of patients with an intermediate likelihood for ACS, not an all-comer population. This group comprises about 20% of the [emergency department] population and actually is the group we struggle with the most, which is hardest to diagnose because it excludes the very low– and very high–risk patients,” said Dr. Karády.
The patients included in this study all had suspected ACS and were enrolled in the ROMICAT-I and II trials.
Among 1,027 samples from 624 patients (mean age, 52.8 years; 39.4% women), samples were classified as below the limit of detection (LOD) in 56.3%, 10.4%, and 41.2% (P < .001) by Roche, Abbott, and Siemens, respectively.
The proportion of sample with a troponin measurement between LOD to the 99th percentile also differed significantly between the assays at 36.5%, 83.5%, and 52.6%, respectively (P < .001).
Only the proportion classified greater than 99th percentile did not differ (7.2%, 6.0%, and 6.2%; P = .114).
When the researchers looked at sex-specific difference, no differences were seen in rule-in numbers for men, but significant differences were seen for women.
“One possible explanation for this could be differences in the representation of men and women in the various reference populations used to develop the 99th percentile values for these assays,” suggested Dr. Karády.
They estimate around 30%-40% of U.S. centers are currently using high-sensitivity troponin assays and this number is “rapidly rising.”
The diagnostic algorithms developed for use with high-sensitivity assays, such as the 0/2-h algorithm, acknowledge differences in performance characteristics and recommend that assay-specific cut points be used for clinical decision-making rather than relying on generally applicable thresholds.
Joseph S. Alpert, MD, University of Arizona, Tucson, and coauthors of an accompanying editorial said the take-home message here is caveat emptor.
“First, ‘let the buyer (i.e., the clinician) beware’ when patients are transferred from one hospital to another, where different hs-cTn assays may be used,” they wrote. This is particularly true in women and in those with troponin levels in the “observe (gray zone)” clinical management recommendation.
Dr. Karády has received grant support from the Fulbright Visiting Researcher Grant and the Rosztoczy Foundation. One of the coauthors of the editorial comment consults or has consulted for most of the major diagnostic companies, including the manufacturers of the three assays tested in this study. Dr. Alpert disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clinicians should be aware that the discordance between high-sensitivity cardiac troponin (hs-cTn) assays is significant enough that management recommendations may change, for example, for a patient assessed for suspected acute coronary syndrome (ACS) in one hospital and transferred to another that uses a different assay, according to a team of international researchers.
When hs-cTn concentrations were measured using the three Food and Drug Administration–approved assays, only 37.4% (384 of 1,027 samples) of blood samples were classified into the same analytical benchmark category.
“We didn’t expect such low concordance, to be honest, but I have to stress that this first assessment used just one-time blood testing and serial testing is what is more commonly recommended now,” said Júlia Karády, MD, from Massachusetts General Hospital and Harvard Medical School, both in Boston.
To see if concordance improved with serial testing, the researchers looked at the 242 patients for whom serial samples were available and saw concordance of management recommendations across assays rise to 74.8%.
“We tested the 0/2-hour algorithm and found that the overall agreement almost doubled, so I think that a very important message from our study is that serial testing improves the agreement between the assays in terms of clinical management and patient stratification,” said Dr. Karády.
Dr. Karády and colleagues published their findings in the Journal of the American College of Cardiology.
The researchers tested three assays referred to clinically as high-sensitivity assays: Elecsys 2010 platform (Roche Diagnostics); ARCHITECT i2000SR (Abbott Diagnostics); and hsVista (Siemens Diagnostics). All three have received FDA approval, starting with Elecsys in 2017.
The proportion of patients with similar management recommendations differed between the assays for both “rule-out” (87.2%, 73.1%, and 78.5% for Roche, Abbott, and Siemens, respectively) and “observe” (9.5%, 24%, and 17.8%; both P < .001). For the purposes of “rule-in,” no difference was noted (3.3%, 2.9%, and 3.7%).
“It’s important to note that this was a highly selected population of patients with an intermediate likelihood for ACS, not an all-comer population. This group comprises about 20% of the [emergency department] population and actually is the group we struggle with the most, which is hardest to diagnose because it excludes the very low– and very high–risk patients,” said Dr. Karády.
The patients included in this study all had suspected ACS and were enrolled in the ROMICAT-I and II trials.
Among 1,027 samples from 624 patients (mean age, 52.8 years; 39.4% women), samples were classified as below the limit of detection (LOD) in 56.3%, 10.4%, and 41.2% (P < .001) by Roche, Abbott, and Siemens, respectively.
The proportion of sample with a troponin measurement between LOD to the 99th percentile also differed significantly between the assays at 36.5%, 83.5%, and 52.6%, respectively (P < .001).
Only the proportion classified greater than 99th percentile did not differ (7.2%, 6.0%, and 6.2%; P = .114).
When the researchers looked at sex-specific difference, no differences were seen in rule-in numbers for men, but significant differences were seen for women.
“One possible explanation for this could be differences in the representation of men and women in the various reference populations used to develop the 99th percentile values for these assays,” suggested Dr. Karády.
They estimate around 30%-40% of U.S. centers are currently using high-sensitivity troponin assays and this number is “rapidly rising.”
The diagnostic algorithms developed for use with high-sensitivity assays, such as the 0/2-h algorithm, acknowledge differences in performance characteristics and recommend that assay-specific cut points be used for clinical decision-making rather than relying on generally applicable thresholds.
Joseph S. Alpert, MD, University of Arizona, Tucson, and coauthors of an accompanying editorial said the take-home message here is caveat emptor.
“First, ‘let the buyer (i.e., the clinician) beware’ when patients are transferred from one hospital to another, where different hs-cTn assays may be used,” they wrote. This is particularly true in women and in those with troponin levels in the “observe (gray zone)” clinical management recommendation.
Dr. Karády has received grant support from the Fulbright Visiting Researcher Grant and the Rosztoczy Foundation. One of the coauthors of the editorial comment consults or has consulted for most of the major diagnostic companies, including the manufacturers of the three assays tested in this study. Dr. Alpert disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.

