User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
Drug cocktail significantly reduced severe COVID, death in outpatients
A monoclonal antibody combination of casirivimab and imdevimab (REGEN-COV) significantly reduced the risk of COVID-19–related hospitalizations and death from any cause in the phase 3 portion of an adaptive trial of outpatients.
Researchers, led by David Weinreich, MD, MBA, executive vice president of the drug cocktail’s manufacturer Regeneron, found in the randomized trial that the combination also resolved symptoms and reduced the SARS-CoV-2 viral load more quickly, compared with placebo.
Findings were published in the New England Journal of Medicine.
COVID-related hospitalization or death from any cause occurred in 18 of 1,355 patients (1.3%) in the group getting 2,400 mg infusions of the study drug, compared with 62 (4.6%) of 1,341 in the matching placebo group, indicating a relative risk reduction of 71.3%; P < .001.
Sunil Joshi, MD, president of the Duval County Medical Society Foundation and an immunologist in Jacksonville, Fla., said in an interview that these findings confirm benefits of REGEN-COV and are very good news for a patient group that includes those age 65 and older with high blood pressure, diabetes, or obesity; and for people not vaccinated, who are all at high risk of hospitalization or death if they get COVID-19.
“Vaccines are critically important,” he said, “but if you were to be infected and know that there’s a way to keep yourself out of the hospital, this is very good news.”
Researchers seek lowest doses
This trial found that the effect was similar when researchers cut the doses in half. These outcomes occurred in 7 of 736 (1%) of patients given 1,200 mg of REGEN-COV and in 24 (3.2%) of 748 in the matching placebo group (relative risk reduction, 70.4%; P = .002).
Symptoms were resolved on average 4 days earlier with each REGEN-COV dose than with placebo (10 days vs. 14 days; P < .001 for both comparisons).
Dr. Weinreich said in an interview that trials will continue to find the lowest effective doses that can stand up to all evolving variants.
“This is one of those settings where you don’t want to underdose. You’ve got one shot at this,” he said. “We’d love to do lower doses. It would be more convenient and we could treat more patients, but if it generates more clinical failures or doesn’t work with certain variants, then you’ve done a huge disservice to the world.”
Also new in this study is that researchers tested not only seronegative patients, but patients at high risk regardless of blood antibody status, he said.
“It’s the first suggestion of data that if you’re breaking through a vaccine and you’re at high risk, the use of the cocktail is something to strongly consider because treatment early is better than treatment later,” Dr. Weinreich said.
In addition to efficacy, the phase 3 trial demonstrated the cocktail had a good safety profile. Serious adverse events occurred more often in the placebo group (4%) than in the 1,200-mg group (1.1%) and the 2,400-mg group (1.3%). Infusion reactions (grade 2 or higher) occurred in less than 0.3% of patients in all groups.
William Fales, MD, state medical director for the Michigan Department of Health and Human Services, said the results confirm the promise of REGEN-COV for reducing hospitalizations and death in a peer-reviewed publication.
COVID-19 a moving target
However, Dr. Fales noted that COVID-19 is a moving target with emerging variants. The criteria for populations at high risk have also broadened since the start of the study, he said.
“A great example is pregnancy is now included as high risk, and that would have likely been a specific contraindication of patients in this clinical trial,” he said.
Dr. Fales said Michigan has been using both REGEN-COV and the Eli Lilly combination of bamlanivimab and etesevimab, which also has an emergency use authorization (EUA) from the Food and Drug Administration, with positive results.
REGEN-COV has an EUA to treat people who are at high risk of serious consequences from COVID-19, including those who are already infected (nonhospitalized) or those in certain postexposure prophylaxis settings.
“We’re seeing very low hospitalization rates and few deaths in a state that is predominately Delta,” Dr. Fales said. “So, this makes us feel that we’re doing the right thing and supports the current efforts around the country to make monoclonal antibody therapy available to high-risk patients.”
Dr. Joshi noted that trial results have been emerging from other monoclonal antibody cocktails with different COVID-19 patient groups.
However, he said in an interview, “how much more effective they would be than this is something we’d have to look at, as 71% effectiveness in keeping people out of the hospital is pretty good for any treatment.”
“These are great numbers, but vaccination itself keeps you from getting the disease in the first place and not just for a short time period. This treatment is just that – a treatment. It gets you through that episode but it doesn’t mean you won’t get sick again. You don’t develop an immune response as you do with the vaccine,” he said.
Dr. Weinreich agreed: “This is not a substitute for a vaccine except for the small group who get the vaccine and their bodies can’t respond to it because they’re significantly immunocompromised.”
The results from this paper “are one piece of a large, multistudy, phase 3 program that basically spans from prophylaxis all the way to hospitalization and pretty much the gamut – all of them – have worked. All of these studies have shown dramatic improvement in whatever the definitive regulatory endpoint is,” Dr. Weinreich said.
He said discussions are ongoing for full regulatory approval in the United States and for expanding the EUA for other populations, including pre-exposure prophylaxis, “which the [United Kingdom’s] authority has already granted us but the FDA has not.”
The study is funded by Regeneron and the Department of Health & Human Services. Dr. Weinreich is a vice president of Regeneron. Dr. Joshi reported no relevant financial relationships. Dr. Fales holds stock in Eli Lilly.
A version of this article first appeared on Medscape.com.
A monoclonal antibody combination of casirivimab and imdevimab (REGEN-COV) significantly reduced the risk of COVID-19–related hospitalizations and death from any cause in the phase 3 portion of an adaptive trial of outpatients.
Researchers, led by David Weinreich, MD, MBA, executive vice president of the drug cocktail’s manufacturer Regeneron, found in the randomized trial that the combination also resolved symptoms and reduced the SARS-CoV-2 viral load more quickly, compared with placebo.
Findings were published in the New England Journal of Medicine.
COVID-related hospitalization or death from any cause occurred in 18 of 1,355 patients (1.3%) in the group getting 2,400 mg infusions of the study drug, compared with 62 (4.6%) of 1,341 in the matching placebo group, indicating a relative risk reduction of 71.3%; P < .001.
Sunil Joshi, MD, president of the Duval County Medical Society Foundation and an immunologist in Jacksonville, Fla., said in an interview that these findings confirm benefits of REGEN-COV and are very good news for a patient group that includes those age 65 and older with high blood pressure, diabetes, or obesity; and for people not vaccinated, who are all at high risk of hospitalization or death if they get COVID-19.
“Vaccines are critically important,” he said, “but if you were to be infected and know that there’s a way to keep yourself out of the hospital, this is very good news.”
Researchers seek lowest doses
This trial found that the effect was similar when researchers cut the doses in half. These outcomes occurred in 7 of 736 (1%) of patients given 1,200 mg of REGEN-COV and in 24 (3.2%) of 748 in the matching placebo group (relative risk reduction, 70.4%; P = .002).
Symptoms were resolved on average 4 days earlier with each REGEN-COV dose than with placebo (10 days vs. 14 days; P < .001 for both comparisons).
Dr. Weinreich said in an interview that trials will continue to find the lowest effective doses that can stand up to all evolving variants.
“This is one of those settings where you don’t want to underdose. You’ve got one shot at this,” he said. “We’d love to do lower doses. It would be more convenient and we could treat more patients, but if it generates more clinical failures or doesn’t work with certain variants, then you’ve done a huge disservice to the world.”
Also new in this study is that researchers tested not only seronegative patients, but patients at high risk regardless of blood antibody status, he said.
“It’s the first suggestion of data that if you’re breaking through a vaccine and you’re at high risk, the use of the cocktail is something to strongly consider because treatment early is better than treatment later,” Dr. Weinreich said.
In addition to efficacy, the phase 3 trial demonstrated the cocktail had a good safety profile. Serious adverse events occurred more often in the placebo group (4%) than in the 1,200-mg group (1.1%) and the 2,400-mg group (1.3%). Infusion reactions (grade 2 or higher) occurred in less than 0.3% of patients in all groups.
William Fales, MD, state medical director for the Michigan Department of Health and Human Services, said the results confirm the promise of REGEN-COV for reducing hospitalizations and death in a peer-reviewed publication.
COVID-19 a moving target
However, Dr. Fales noted that COVID-19 is a moving target with emerging variants. The criteria for populations at high risk have also broadened since the start of the study, he said.
“A great example is pregnancy is now included as high risk, and that would have likely been a specific contraindication of patients in this clinical trial,” he said.
Dr. Fales said Michigan has been using both REGEN-COV and the Eli Lilly combination of bamlanivimab and etesevimab, which also has an emergency use authorization (EUA) from the Food and Drug Administration, with positive results.
REGEN-COV has an EUA to treat people who are at high risk of serious consequences from COVID-19, including those who are already infected (nonhospitalized) or those in certain postexposure prophylaxis settings.
“We’re seeing very low hospitalization rates and few deaths in a state that is predominately Delta,” Dr. Fales said. “So, this makes us feel that we’re doing the right thing and supports the current efforts around the country to make monoclonal antibody therapy available to high-risk patients.”
Dr. Joshi noted that trial results have been emerging from other monoclonal antibody cocktails with different COVID-19 patient groups.
However, he said in an interview, “how much more effective they would be than this is something we’d have to look at, as 71% effectiveness in keeping people out of the hospital is pretty good for any treatment.”
“These are great numbers, but vaccination itself keeps you from getting the disease in the first place and not just for a short time period. This treatment is just that – a treatment. It gets you through that episode but it doesn’t mean you won’t get sick again. You don’t develop an immune response as you do with the vaccine,” he said.
Dr. Weinreich agreed: “This is not a substitute for a vaccine except for the small group who get the vaccine and their bodies can’t respond to it because they’re significantly immunocompromised.”
The results from this paper “are one piece of a large, multistudy, phase 3 program that basically spans from prophylaxis all the way to hospitalization and pretty much the gamut – all of them – have worked. All of these studies have shown dramatic improvement in whatever the definitive regulatory endpoint is,” Dr. Weinreich said.
He said discussions are ongoing for full regulatory approval in the United States and for expanding the EUA for other populations, including pre-exposure prophylaxis, “which the [United Kingdom’s] authority has already granted us but the FDA has not.”
The study is funded by Regeneron and the Department of Health & Human Services. Dr. Weinreich is a vice president of Regeneron. Dr. Joshi reported no relevant financial relationships. Dr. Fales holds stock in Eli Lilly.
A version of this article first appeared on Medscape.com.
A monoclonal antibody combination of casirivimab and imdevimab (REGEN-COV) significantly reduced the risk of COVID-19–related hospitalizations and death from any cause in the phase 3 portion of an adaptive trial of outpatients.
Researchers, led by David Weinreich, MD, MBA, executive vice president of the drug cocktail’s manufacturer Regeneron, found in the randomized trial that the combination also resolved symptoms and reduced the SARS-CoV-2 viral load more quickly, compared with placebo.
Findings were published in the New England Journal of Medicine.
COVID-related hospitalization or death from any cause occurred in 18 of 1,355 patients (1.3%) in the group getting 2,400 mg infusions of the study drug, compared with 62 (4.6%) of 1,341 in the matching placebo group, indicating a relative risk reduction of 71.3%; P < .001.
Sunil Joshi, MD, president of the Duval County Medical Society Foundation and an immunologist in Jacksonville, Fla., said in an interview that these findings confirm benefits of REGEN-COV and are very good news for a patient group that includes those age 65 and older with high blood pressure, diabetes, or obesity; and for people not vaccinated, who are all at high risk of hospitalization or death if they get COVID-19.
“Vaccines are critically important,” he said, “but if you were to be infected and know that there’s a way to keep yourself out of the hospital, this is very good news.”
Researchers seek lowest doses
This trial found that the effect was similar when researchers cut the doses in half. These outcomes occurred in 7 of 736 (1%) of patients given 1,200 mg of REGEN-COV and in 24 (3.2%) of 748 in the matching placebo group (relative risk reduction, 70.4%; P = .002).
Symptoms were resolved on average 4 days earlier with each REGEN-COV dose than with placebo (10 days vs. 14 days; P < .001 for both comparisons).
Dr. Weinreich said in an interview that trials will continue to find the lowest effective doses that can stand up to all evolving variants.
“This is one of those settings where you don’t want to underdose. You’ve got one shot at this,” he said. “We’d love to do lower doses. It would be more convenient and we could treat more patients, but if it generates more clinical failures or doesn’t work with certain variants, then you’ve done a huge disservice to the world.”
Also new in this study is that researchers tested not only seronegative patients, but patients at high risk regardless of blood antibody status, he said.
“It’s the first suggestion of data that if you’re breaking through a vaccine and you’re at high risk, the use of the cocktail is something to strongly consider because treatment early is better than treatment later,” Dr. Weinreich said.
In addition to efficacy, the phase 3 trial demonstrated the cocktail had a good safety profile. Serious adverse events occurred more often in the placebo group (4%) than in the 1,200-mg group (1.1%) and the 2,400-mg group (1.3%). Infusion reactions (grade 2 or higher) occurred in less than 0.3% of patients in all groups.
William Fales, MD, state medical director for the Michigan Department of Health and Human Services, said the results confirm the promise of REGEN-COV for reducing hospitalizations and death in a peer-reviewed publication.
COVID-19 a moving target
However, Dr. Fales noted that COVID-19 is a moving target with emerging variants. The criteria for populations at high risk have also broadened since the start of the study, he said.
“A great example is pregnancy is now included as high risk, and that would have likely been a specific contraindication of patients in this clinical trial,” he said.
Dr. Fales said Michigan has been using both REGEN-COV and the Eli Lilly combination of bamlanivimab and etesevimab, which also has an emergency use authorization (EUA) from the Food and Drug Administration, with positive results.
REGEN-COV has an EUA to treat people who are at high risk of serious consequences from COVID-19, including those who are already infected (nonhospitalized) or those in certain postexposure prophylaxis settings.
“We’re seeing very low hospitalization rates and few deaths in a state that is predominately Delta,” Dr. Fales said. “So, this makes us feel that we’re doing the right thing and supports the current efforts around the country to make monoclonal antibody therapy available to high-risk patients.”
Dr. Joshi noted that trial results have been emerging from other monoclonal antibody cocktails with different COVID-19 patient groups.
However, he said in an interview, “how much more effective they would be than this is something we’d have to look at, as 71% effectiveness in keeping people out of the hospital is pretty good for any treatment.”
“These are great numbers, but vaccination itself keeps you from getting the disease in the first place and not just for a short time period. This treatment is just that – a treatment. It gets you through that episode but it doesn’t mean you won’t get sick again. You don’t develop an immune response as you do with the vaccine,” he said.
Dr. Weinreich agreed: “This is not a substitute for a vaccine except for the small group who get the vaccine and their bodies can’t respond to it because they’re significantly immunocompromised.”
The results from this paper “are one piece of a large, multistudy, phase 3 program that basically spans from prophylaxis all the way to hospitalization and pretty much the gamut – all of them – have worked. All of these studies have shown dramatic improvement in whatever the definitive regulatory endpoint is,” Dr. Weinreich said.
He said discussions are ongoing for full regulatory approval in the United States and for expanding the EUA for other populations, including pre-exposure prophylaxis, “which the [United Kingdom’s] authority has already granted us but the FDA has not.”
The study is funded by Regeneron and the Department of Health & Human Services. Dr. Weinreich is a vice president of Regeneron. Dr. Joshi reported no relevant financial relationships. Dr. Fales holds stock in Eli Lilly.
A version of this article first appeared on Medscape.com.
New data illustrate pandemic pivot to telehealth by patients, physicians
Telehealth use, although much higher than before the COVID-19 pandemic, accounted for less than 20% of weekly outpatient visits 6 months into the pandemic, according to a new report from the American Medical Association. Ten percent of weekly visits were conducted via videoconferencing, and 8.1% of visits were conducted using the telephone.
Those figures may overstate the true level of telehealth use in fall 2020. A study by the Commonwealth Fund, Harvard University, Boston, and Phreesia found that in December of that year, only 8% of outpatient visits involved the use of telemedicine – and that was up from 6% in October. In contrast to the AMA results, which came from its 2020 benchmark survey of physicians, the Commonwealth Fund study used data from practice management systems and an online patient registration platform, as well as electronic health record data.
A more recent survey of hospital executives found that as of September 2021, hospital telehealth visits had leveled off at 10% to 20% of appointments. Similarly, a McKinsey survey in July showed that telehealth encounters made up 13% to 17% of evaluation and management visits across all specialties.
Big jump during pandemic
The AMA report offers a wealth of data on how physicians use telehealth and the differences between specialties in this area.
The report found that 70.3% of physicians worked in practices that used videoconferencing to provide patient visits in September 2020, compared to 14.3% of physicians in September 2018. Sixty-seven percent of physicians worked in practices that used telephone visits (the comparable figure for 2018 was unavailable).
Overall, 79% of physicians worked in a practice that used telehealth, compared to 25% in 2018.
Not every doctor in practices that utilized telehealth conducted virtual visits. In contrast to the 70.3% of doctors who were in practices that had video visits, only 59.1% of the respondents had personally conducted a videoconferencing visit in the previous week. The average numbers of weekly video and telephone visits per physician were 9.9 and 7.6, respectively, including those who did none.
There were big differences in virtual visit use among specialties as well. Eighty-five percent of psychiatrists were in practices that provided online appointments, according to the AMA survey, and three-quarters of primary care physicians said their practices offered telehealth appointments. Pediatricians were much less likely than family practice/general practice physicians (FPs/GPs) or general internists to do so.
The practices of many medical specialists were also highly likely to provide telehealth. Over 75% of practices in cardiology, endocrinology/diabetes, gastroenterology, nephrology, and neurology offered telehealth visits. About 88% of hematologists/oncologists offered video visits. Far fewer surgeons reported that their practice used virtual visits; the exceptions were urologists and dermatologists, 87% of whose practices used telehealth.
How telehealth was used
Across all specialties, 58% of physicians said clinicians in their practices used it to diagnose or treat patients; 59.2%, to manage patients with chronic disease; 50.4%, to provide acute care; and 34.3%, to provide preventive care.
Seventy-two percent of FP/GP and pediatric practices used telehealth to diagnose or treat patients. Just 64.9% of internists said their practices did so, and only 61.9% of them said their practices provided acute care via telehealth, versus 70% of FPs/GPs and pediatricians.
Among medical specialties, endocrinologists/diabetes physicians were those most likely to report the practice-level use of telehealth to diagnose or treat patients (71.9%), manage patients with chronic disease (92.1%), and provide preventive care (52.6%).
Significantly, 33% of medical specialists said their practices used remote patient monitoring. This finding was driven by high rates of use among cardiology practices (63.3%) and endocrinology practices (41.6%). Overall, the practice-level use of remote patient monitoring rose from 10.4% of practices in 2018 to 19.9% in 2020.
Virtual consults with peers
Some practices used telehealth to enable physicians to consult with colleagues. Twelve percent of respondents said their practices used telehealth to seek a second opinion from a health care professional in 2020, compared to 6.9% in 2018. Formal consultations via telehealth were also increasingly common: 17.2% of doctors said their practices did this in 2020, compared to 11.3% in 2018.
Also of note, 22.4% of physicians said their practices used telehealth for after-hours care or night calls in 2020, versus 9.9% in 2018.
The AMA report credited telehealth and expanded coverage and payment rules for enabling physician practices to keep their revenue streams positive and their practices open. However, the Commonwealth Fund study found “a substantial cumulative reduction in visits across all specialties over the course of the pandemic in 2020.” These ranged from a drop of 27% in pediatric visits to a decline of 8% in rheumatology visits during the period from March to December 2020.
A version of this article first appeared on Medscape.com.
Telehealth use, although much higher than before the COVID-19 pandemic, accounted for less than 20% of weekly outpatient visits 6 months into the pandemic, according to a new report from the American Medical Association. Ten percent of weekly visits were conducted via videoconferencing, and 8.1% of visits were conducted using the telephone.
Those figures may overstate the true level of telehealth use in fall 2020. A study by the Commonwealth Fund, Harvard University, Boston, and Phreesia found that in December of that year, only 8% of outpatient visits involved the use of telemedicine – and that was up from 6% in October. In contrast to the AMA results, which came from its 2020 benchmark survey of physicians, the Commonwealth Fund study used data from practice management systems and an online patient registration platform, as well as electronic health record data.
A more recent survey of hospital executives found that as of September 2021, hospital telehealth visits had leveled off at 10% to 20% of appointments. Similarly, a McKinsey survey in July showed that telehealth encounters made up 13% to 17% of evaluation and management visits across all specialties.
Big jump during pandemic
The AMA report offers a wealth of data on how physicians use telehealth and the differences between specialties in this area.
The report found that 70.3% of physicians worked in practices that used videoconferencing to provide patient visits in September 2020, compared to 14.3% of physicians in September 2018. Sixty-seven percent of physicians worked in practices that used telephone visits (the comparable figure for 2018 was unavailable).
Overall, 79% of physicians worked in a practice that used telehealth, compared to 25% in 2018.
Not every doctor in practices that utilized telehealth conducted virtual visits. In contrast to the 70.3% of doctors who were in practices that had video visits, only 59.1% of the respondents had personally conducted a videoconferencing visit in the previous week. The average numbers of weekly video and telephone visits per physician were 9.9 and 7.6, respectively, including those who did none.
There were big differences in virtual visit use among specialties as well. Eighty-five percent of psychiatrists were in practices that provided online appointments, according to the AMA survey, and three-quarters of primary care physicians said their practices offered telehealth appointments. Pediatricians were much less likely than family practice/general practice physicians (FPs/GPs) or general internists to do so.
The practices of many medical specialists were also highly likely to provide telehealth. Over 75% of practices in cardiology, endocrinology/diabetes, gastroenterology, nephrology, and neurology offered telehealth visits. About 88% of hematologists/oncologists offered video visits. Far fewer surgeons reported that their practice used virtual visits; the exceptions were urologists and dermatologists, 87% of whose practices used telehealth.
How telehealth was used
Across all specialties, 58% of physicians said clinicians in their practices used it to diagnose or treat patients; 59.2%, to manage patients with chronic disease; 50.4%, to provide acute care; and 34.3%, to provide preventive care.
Seventy-two percent of FP/GP and pediatric practices used telehealth to diagnose or treat patients. Just 64.9% of internists said their practices did so, and only 61.9% of them said their practices provided acute care via telehealth, versus 70% of FPs/GPs and pediatricians.
Among medical specialties, endocrinologists/diabetes physicians were those most likely to report the practice-level use of telehealth to diagnose or treat patients (71.9%), manage patients with chronic disease (92.1%), and provide preventive care (52.6%).
Significantly, 33% of medical specialists said their practices used remote patient monitoring. This finding was driven by high rates of use among cardiology practices (63.3%) and endocrinology practices (41.6%). Overall, the practice-level use of remote patient monitoring rose from 10.4% of practices in 2018 to 19.9% in 2020.
Virtual consults with peers
Some practices used telehealth to enable physicians to consult with colleagues. Twelve percent of respondents said their practices used telehealth to seek a second opinion from a health care professional in 2020, compared to 6.9% in 2018. Formal consultations via telehealth were also increasingly common: 17.2% of doctors said their practices did this in 2020, compared to 11.3% in 2018.
Also of note, 22.4% of physicians said their practices used telehealth for after-hours care or night calls in 2020, versus 9.9% in 2018.
The AMA report credited telehealth and expanded coverage and payment rules for enabling physician practices to keep their revenue streams positive and their practices open. However, the Commonwealth Fund study found “a substantial cumulative reduction in visits across all specialties over the course of the pandemic in 2020.” These ranged from a drop of 27% in pediatric visits to a decline of 8% in rheumatology visits during the period from March to December 2020.
A version of this article first appeared on Medscape.com.
Telehealth use, although much higher than before the COVID-19 pandemic, accounted for less than 20% of weekly outpatient visits 6 months into the pandemic, according to a new report from the American Medical Association. Ten percent of weekly visits were conducted via videoconferencing, and 8.1% of visits were conducted using the telephone.
Those figures may overstate the true level of telehealth use in fall 2020. A study by the Commonwealth Fund, Harvard University, Boston, and Phreesia found that in December of that year, only 8% of outpatient visits involved the use of telemedicine – and that was up from 6% in October. In contrast to the AMA results, which came from its 2020 benchmark survey of physicians, the Commonwealth Fund study used data from practice management systems and an online patient registration platform, as well as electronic health record data.
A more recent survey of hospital executives found that as of September 2021, hospital telehealth visits had leveled off at 10% to 20% of appointments. Similarly, a McKinsey survey in July showed that telehealth encounters made up 13% to 17% of evaluation and management visits across all specialties.
Big jump during pandemic
The AMA report offers a wealth of data on how physicians use telehealth and the differences between specialties in this area.
The report found that 70.3% of physicians worked in practices that used videoconferencing to provide patient visits in September 2020, compared to 14.3% of physicians in September 2018. Sixty-seven percent of physicians worked in practices that used telephone visits (the comparable figure for 2018 was unavailable).
Overall, 79% of physicians worked in a practice that used telehealth, compared to 25% in 2018.
Not every doctor in practices that utilized telehealth conducted virtual visits. In contrast to the 70.3% of doctors who were in practices that had video visits, only 59.1% of the respondents had personally conducted a videoconferencing visit in the previous week. The average numbers of weekly video and telephone visits per physician were 9.9 and 7.6, respectively, including those who did none.
There were big differences in virtual visit use among specialties as well. Eighty-five percent of psychiatrists were in practices that provided online appointments, according to the AMA survey, and three-quarters of primary care physicians said their practices offered telehealth appointments. Pediatricians were much less likely than family practice/general practice physicians (FPs/GPs) or general internists to do so.
The practices of many medical specialists were also highly likely to provide telehealth. Over 75% of practices in cardiology, endocrinology/diabetes, gastroenterology, nephrology, and neurology offered telehealth visits. About 88% of hematologists/oncologists offered video visits. Far fewer surgeons reported that their practice used virtual visits; the exceptions were urologists and dermatologists, 87% of whose practices used telehealth.
How telehealth was used
Across all specialties, 58% of physicians said clinicians in their practices used it to diagnose or treat patients; 59.2%, to manage patients with chronic disease; 50.4%, to provide acute care; and 34.3%, to provide preventive care.
Seventy-two percent of FP/GP and pediatric practices used telehealth to diagnose or treat patients. Just 64.9% of internists said their practices did so, and only 61.9% of them said their practices provided acute care via telehealth, versus 70% of FPs/GPs and pediatricians.
Among medical specialties, endocrinologists/diabetes physicians were those most likely to report the practice-level use of telehealth to diagnose or treat patients (71.9%), manage patients with chronic disease (92.1%), and provide preventive care (52.6%).
Significantly, 33% of medical specialists said their practices used remote patient monitoring. This finding was driven by high rates of use among cardiology practices (63.3%) and endocrinology practices (41.6%). Overall, the practice-level use of remote patient monitoring rose from 10.4% of practices in 2018 to 19.9% in 2020.
Virtual consults with peers
Some practices used telehealth to enable physicians to consult with colleagues. Twelve percent of respondents said their practices used telehealth to seek a second opinion from a health care professional in 2020, compared to 6.9% in 2018. Formal consultations via telehealth were also increasingly common: 17.2% of doctors said their practices did this in 2020, compared to 11.3% in 2018.
Also of note, 22.4% of physicians said their practices used telehealth for after-hours care or night calls in 2020, versus 9.9% in 2018.
The AMA report credited telehealth and expanded coverage and payment rules for enabling physician practices to keep their revenue streams positive and their practices open. However, the Commonwealth Fund study found “a substantial cumulative reduction in visits across all specialties over the course of the pandemic in 2020.” These ranged from a drop of 27% in pediatric visits to a decline of 8% in rheumatology visits during the period from March to December 2020.
A version of this article first appeared on Medscape.com.
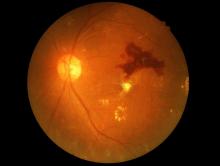
Retinopathy risk higher in young-onset T2D, more so in men
Men diagnosed with type 2 diabetes (T2D) by the age of 40 years appear significantly more likely to develop retinopathy than men who are diagnosed at an older age, Norwegian researchers report.
In a cross-sectional study of about 10,000 people, men with young-onset T2D were 72% more likely than men aged 50 years or older to have retinopathy.
While an increased retinopathy risk was also seen in women with young-onset T2D versus older women at first, this difference was not significant after adjusting for various confounding factors.
The effect of young-onset diabetes on retinopathy seems to be gender specific, Katrina Tibballs, MD, of the department of general practice at the University of Oslo, reported at the annual meeting of the European Association for the Study of Diabetes.
“In the unadjusted analysis, the odds ratio for retinopathy was substantially higher in both [young-onset] men [odds ratio, 3.0] and women [OR, 2.46], compared with those 50 or older at diabetes diagnosis,” Dr. Tibballs said.
That relationship was not substantially altered after adjustment for variables such as level of education, country background, gender, and body mass index, with adjusted ORs of 2.56 and 2.55 for men and women, respectively.
However, further adjustment to include current age, duration of diabetes, and blood lipids and glycated hemoglobin levels, led to the difference no longer holding for women (OR, 1.34; 95% confidence interval, 0.95-1.89) as it did for men (OR 1.72; 95% CI, 1.29-2.29).
First data in Norwegian population
Cross-sectional data on more than 10,000 people with T2D were used for the analysis. These came from the ROSA4 study, a general practice study conducted across Norway in 2014.
Just over 10% of the study population used in the analysis was under the age of 40 years at diagnosis of T2D; 21% were aged between 40 and 49 years, and 69% were at least 50 years old.
The mean age of those with young-onset T2D, defined as a diagnosis before the age of 40 years, was 33 years. These individuals had a longer disease duration than those in the other age groups (11.4 vs. 10.0 vs. 7.8 years).
“Looking at clinical characteristics, we say that individuals [with young-onset T2D] have a higher level of hemoglobin A1c than those with diabetes onset later in life,” Dr. Tibballs said.
“This is despite a substantially higher proportion [being] treated with insulin and fewer on lifestyle interventions alone.”
Gender differences were seen in A1c levels, with men with young-onset T2D having consistently higher levels than women, with levels increasing with diabetes duration.
Rise in retinopathy faster in men than in women
Dr. Tibballs reported that, not only did the prevalence of retinopathy rise faster in those of a younger age, but it also rose more quickly in men with young-onset T2D than it in their female counterparts.
“Comparing that [young-onset diabetes] and later-onset diabetes in men and women separately, we see a clearly higher prevalence of retinopathy with increasing diabetes duration for [young-onset] men,” she said.
In women, on the other hand, there was “no clear indication of a higher retinopathy prevalence in [young-onset diabetes], except in those with the longest diabetes duration.”
So, what do the results mean for practice? First, they confirm prior work showing that there is a strong association between retinopathy and age at diagnosis of T2D. Second, they suggest that this is despite intensive glucose-lowering treatment.
She speculated that men with young-onset T2D may have had a delayed diagnosis when compared with women and individuals with later onset diabetes, Dr. Tibballs said.
“This may in turn lead to delayed onset of glucose-lowering treatment, allowing for more time with high glycemic exposure and increased risk of acquiring complications, such as retinopathy at the time of diagnosis, or in the first years after,” said Dr. Tibballs.
These are cross-sectional data, “so we can’t say anything about whether this treatment is sufficient, but it is obviously not reducing HbA1c levels as much as we would like” added Dr. Tibballs, who is a primary care physician and PhD student.
The study was supported by The Norwegian Research Fund for General Practice. Dr. Tibballs had no conflicts of interest to disclose.
Men diagnosed with type 2 diabetes (T2D) by the age of 40 years appear significantly more likely to develop retinopathy than men who are diagnosed at an older age, Norwegian researchers report.
In a cross-sectional study of about 10,000 people, men with young-onset T2D were 72% more likely than men aged 50 years or older to have retinopathy.
While an increased retinopathy risk was also seen in women with young-onset T2D versus older women at first, this difference was not significant after adjusting for various confounding factors.
The effect of young-onset diabetes on retinopathy seems to be gender specific, Katrina Tibballs, MD, of the department of general practice at the University of Oslo, reported at the annual meeting of the European Association for the Study of Diabetes.
“In the unadjusted analysis, the odds ratio for retinopathy was substantially higher in both [young-onset] men [odds ratio, 3.0] and women [OR, 2.46], compared with those 50 or older at diabetes diagnosis,” Dr. Tibballs said.
That relationship was not substantially altered after adjustment for variables such as level of education, country background, gender, and body mass index, with adjusted ORs of 2.56 and 2.55 for men and women, respectively.
However, further adjustment to include current age, duration of diabetes, and blood lipids and glycated hemoglobin levels, led to the difference no longer holding for women (OR, 1.34; 95% confidence interval, 0.95-1.89) as it did for men (OR 1.72; 95% CI, 1.29-2.29).
First data in Norwegian population
Cross-sectional data on more than 10,000 people with T2D were used for the analysis. These came from the ROSA4 study, a general practice study conducted across Norway in 2014.
Just over 10% of the study population used in the analysis was under the age of 40 years at diagnosis of T2D; 21% were aged between 40 and 49 years, and 69% were at least 50 years old.
The mean age of those with young-onset T2D, defined as a diagnosis before the age of 40 years, was 33 years. These individuals had a longer disease duration than those in the other age groups (11.4 vs. 10.0 vs. 7.8 years).
“Looking at clinical characteristics, we say that individuals [with young-onset T2D] have a higher level of hemoglobin A1c than those with diabetes onset later in life,” Dr. Tibballs said.
“This is despite a substantially higher proportion [being] treated with insulin and fewer on lifestyle interventions alone.”
Gender differences were seen in A1c levels, with men with young-onset T2D having consistently higher levels than women, with levels increasing with diabetes duration.
Rise in retinopathy faster in men than in women
Dr. Tibballs reported that, not only did the prevalence of retinopathy rise faster in those of a younger age, but it also rose more quickly in men with young-onset T2D than it in their female counterparts.
“Comparing that [young-onset diabetes] and later-onset diabetes in men and women separately, we see a clearly higher prevalence of retinopathy with increasing diabetes duration for [young-onset] men,” she said.
In women, on the other hand, there was “no clear indication of a higher retinopathy prevalence in [young-onset diabetes], except in those with the longest diabetes duration.”
So, what do the results mean for practice? First, they confirm prior work showing that there is a strong association between retinopathy and age at diagnosis of T2D. Second, they suggest that this is despite intensive glucose-lowering treatment.
She speculated that men with young-onset T2D may have had a delayed diagnosis when compared with women and individuals with later onset diabetes, Dr. Tibballs said.
“This may in turn lead to delayed onset of glucose-lowering treatment, allowing for more time with high glycemic exposure and increased risk of acquiring complications, such as retinopathy at the time of diagnosis, or in the first years after,” said Dr. Tibballs.
These are cross-sectional data, “so we can’t say anything about whether this treatment is sufficient, but it is obviously not reducing HbA1c levels as much as we would like” added Dr. Tibballs, who is a primary care physician and PhD student.
The study was supported by The Norwegian Research Fund for General Practice. Dr. Tibballs had no conflicts of interest to disclose.
Men diagnosed with type 2 diabetes (T2D) by the age of 40 years appear significantly more likely to develop retinopathy than men who are diagnosed at an older age, Norwegian researchers report.
In a cross-sectional study of about 10,000 people, men with young-onset T2D were 72% more likely than men aged 50 years or older to have retinopathy.
While an increased retinopathy risk was also seen in women with young-onset T2D versus older women at first, this difference was not significant after adjusting for various confounding factors.
The effect of young-onset diabetes on retinopathy seems to be gender specific, Katrina Tibballs, MD, of the department of general practice at the University of Oslo, reported at the annual meeting of the European Association for the Study of Diabetes.
“In the unadjusted analysis, the odds ratio for retinopathy was substantially higher in both [young-onset] men [odds ratio, 3.0] and women [OR, 2.46], compared with those 50 or older at diabetes diagnosis,” Dr. Tibballs said.
That relationship was not substantially altered after adjustment for variables such as level of education, country background, gender, and body mass index, with adjusted ORs of 2.56 and 2.55 for men and women, respectively.
However, further adjustment to include current age, duration of diabetes, and blood lipids and glycated hemoglobin levels, led to the difference no longer holding for women (OR, 1.34; 95% confidence interval, 0.95-1.89) as it did for men (OR 1.72; 95% CI, 1.29-2.29).
First data in Norwegian population
Cross-sectional data on more than 10,000 people with T2D were used for the analysis. These came from the ROSA4 study, a general practice study conducted across Norway in 2014.
Just over 10% of the study population used in the analysis was under the age of 40 years at diagnosis of T2D; 21% were aged between 40 and 49 years, and 69% were at least 50 years old.
The mean age of those with young-onset T2D, defined as a diagnosis before the age of 40 years, was 33 years. These individuals had a longer disease duration than those in the other age groups (11.4 vs. 10.0 vs. 7.8 years).
“Looking at clinical characteristics, we say that individuals [with young-onset T2D] have a higher level of hemoglobin A1c than those with diabetes onset later in life,” Dr. Tibballs said.
“This is despite a substantially higher proportion [being] treated with insulin and fewer on lifestyle interventions alone.”
Gender differences were seen in A1c levels, with men with young-onset T2D having consistently higher levels than women, with levels increasing with diabetes duration.
Rise in retinopathy faster in men than in women
Dr. Tibballs reported that, not only did the prevalence of retinopathy rise faster in those of a younger age, but it also rose more quickly in men with young-onset T2D than it in their female counterparts.
“Comparing that [young-onset diabetes] and later-onset diabetes in men and women separately, we see a clearly higher prevalence of retinopathy with increasing diabetes duration for [young-onset] men,” she said.
In women, on the other hand, there was “no clear indication of a higher retinopathy prevalence in [young-onset diabetes], except in those with the longest diabetes duration.”
So, what do the results mean for practice? First, they confirm prior work showing that there is a strong association between retinopathy and age at diagnosis of T2D. Second, they suggest that this is despite intensive glucose-lowering treatment.
She speculated that men with young-onset T2D may have had a delayed diagnosis when compared with women and individuals with later onset diabetes, Dr. Tibballs said.
“This may in turn lead to delayed onset of glucose-lowering treatment, allowing for more time with high glycemic exposure and increased risk of acquiring complications, such as retinopathy at the time of diagnosis, or in the first years after,” said Dr. Tibballs.
These are cross-sectional data, “so we can’t say anything about whether this treatment is sufficient, but it is obviously not reducing HbA1c levels as much as we would like” added Dr. Tibballs, who is a primary care physician and PhD student.
The study was supported by The Norwegian Research Fund for General Practice. Dr. Tibballs had no conflicts of interest to disclose.
FROM EASD 2021
Study finds paying people to participate in clinical trials is not unethical
Paying people to participate in clinical trials remains controversial. But to date, most reservations are based on hypothetical scenarios or expert opinion with few real-world data to support them.
Research released this week could change that.
Investigators offered nearly 1,300 participants in two clinical trials either no payment or incentives up to $500 to partake in a smoking cessation study or an analysis of a behavioral intervention to increase ambulation in hospitalized patients.
More cash was associated with greater agreement to participate in the smoking cessation study but not the ambulation trial.
But the bigger news may be that offering payment did not appear to get people to accept more risks or skew participation to lower-income individuals, as some ethicists have warned.
“With the publication of our study, investigators finally have data that they can cite to put to rest any lingering concerns about offering moderate incentives in low-risk trials,” lead author Scott D. Halpern, MD, PhD, the John M. Eisenberg Professor of Medicine, Epidemiology, and Medical Ethics & Health Policy at the University of Pennsylvania, Philadelphia, told this news organization.
This initial real-world data centers on low-risk interventions and more research is needed to analyze the ethics and effectiveness of paying people to join clinical trials with more inherent risk, the researchers note.
The study was published online Sept. 20 in JAMA Internal Medicine.
A good first step?
“Payments to research participants are notoriously controversial. Many people oppose payments altogether or insist on minimal payments out of concern that people might be unduly influenced to participate,” Ana S. Iltis, PhD, told this news organization when asked for comment. “Others worry that incentives will disproportionately motivate the less well-off to participate.”
“This is an important study that begins to assess whether these concerns are justified in a real-world context,” added Dr. Iltis, director of the Center for Bioethics, Health and Society and professor of philosophy at Wake Forest University in Winston-Salem, N.C.
In an accompanying invited commentary, Sang Ngo, Anthony S. Kim, MD, and Winston Chiong, MD, PhD, write: “This work is welcome, as it presents experimental data to a bioethical debate that so far has been largely driven by conjecture and competing suppositions.”
The commentary authors, however, question the conclusiveness of the findings. “Interpreting the authors’ findings is complex and illustrates some of the challenges inherent to applying empirical data to ethical problems,” they write.
Recruitment realities
When asked his advice for researchers considering financial incentives, Dr. Halpern said: “All researchers would happily include incentives in their trial budgets if not for concerns that the sponsor or institutional review board might not approve of them.”
“By far the biggest threat to a trial’s success is the inability to enroll enough participants,” he added.
Dr. Iltis agreed, framing the need to boost enrollment in ethical terms. “There is another important ethical issue that often gets ignored, and that is the issue of studies that fail to enroll enough participants and are never completed or are underpowered,” she said.
“These studies end up exposing people to research risks and burdens without a compensating social benefit.”
“If incentives help to increase enrollment and do not necessarily result in undue influence or unfair participant selection, then there might be ethical reasons to offer incentives,” Dr. Iltis added.
Building on previous work assessing financial incentives in hypothetical clinical trials, Dr. Halpern and colleagues studied 654 participants with major depressive disorder in a smoking cessation trial. They also studied another 642 participants in a study that compared a gamification strategy to usual care for encouraging hospitalized patients to get out of bed and walk.
Dr. Halpern and colleagues randomly assigned people in the smoking cessation study to receive no financial compensation, $200, or $500. In the ambulation trial, participants were randomly allocated to receive no compensation, $100, or $300.
Key findings
A total of 22% of those offered no incentive enrolled in the smoking cessation study. In contrast, 36% offered $200 agreed, as did 47% of those offered $500, which the investigators say supports offering cash incentives to boost enrollment. The differences were significant (P < .001).
In contrast, the amount offered did not significantly incentivize more people to participate in the ambulation trial (P = .62). Rates were 45% with no compensation, 48% with $100 payment, and 43% with $300 payment.
In an analysis that adjusted for demographic differences, financial well-being, and Research Attitudes Questionnaire (RAQ-7) scores, each increase in cash incentive increased the odds of enrollment in the smoking cessation trial by 70% (adjusted odds ratio, 1.70; 95% confidence interval, 1.34-2.17).
The same effect was not seen in the ambulation trial, where each higher cash incentive did not make a significant difference (aOR, 0.88; 95% CI, 0.64-1.22).
“The ambulation trial was a lower-risk trial in which patients’ willingness to participate was higher in general. So there were likely fewer people whose participation decisions could be influenced by offers of money,” Dr. Halpern said.
Inducement vs. coercion
The incentives in the study “did not function as unjust inducements, as they were not preferentially motivating across groups with different income levels or financial well-being in either trial,” the researchers note.
Dr. Halpern and colleagues also checked for any perceptions of coercion. More than 70% of participants in each smoking cessation trial group perceived no coercion, as did more than 93% of participants in each ambulation trial group, according to scores on a modified Perceived Coercion Scale of the MacArthur Admission Experience Survey.
Furthermore, perception of risks did not significantly alter the association between cash incentives and enrollment in either trial.
After collecting the findings, Dr. Halpern and colleagues informed participants about their participation in RETAIN and explained the rationale for using different cash incentives. They also let all participants know they would ultimately receive the maximum incentive – either $500 or $300, depending on the trial.
Research implications
A study limitation was reliance on participant risk perception, as was an inability to measure perceived coercion among people who chose not to participant in the trials. Another potential limitation is that “neither of these parent trials posed particularly high risks. Future tests of incentives of different sizes, and in the context of higher-risk parent trials, including trials that test treatments of serious illnesses, are warranted,” the researchers note.
“While there are many more questions to ask and contexts in which to study the effects of incentives, this study calls on opponents of incentivizing research participants with money to be more humble,” Dr. Iltis said. “Incentives might not have the effects they assume they have and which they have long held make such incentives unethical.”
“I encourage researchers who are offering incentives to consider working with people doing ethics research to assess the effects of incentives in their studies,” Dr. Halpern said. “Real-world, as opposed to hypothetical studies that can improve our understanding of the impact of incentives can improve the ethical conduct of research over time.”
Responding to criticism
The authors of the invited commentary questioned the definitions Dr. Halpern and colleagues used for undue or unjust inducement. “Among bioethicists, there is no consensus about what counts as undue inducement or an unjust distribution of research burdens. In this article, the authors have operationalized these constructs based on their own interpretations of undue and unjust inducement, which may not capture all the concerns that scholars have raised about inducement.”
Asked to respond to this and other criticisms raised in the commentary, Dr. Halpern said: “Did our study answer all possible questions about incentives? Absolutely not. But when it comes to incentives for research participation, an ounce of data is worth a pound of conjecture.”
There was agreement, however, that the findings could now put the onus on opponents of financial incentives for trial participants.
“I agree with the commentary’s authors that our study essentially shifts the burden of proof, such that, as they say, ‘those who would limit [incentives’] application may owe us an applicable criterion,’ ” Dr. Halpern said.
The authors of the invited commentary also criticized use of the study’s noninferiority design to rule out undue or unjust inducement. They note this design “may be unfamiliar to many bioethicists and can place substantial evaluative demands on readers.”
“As for the authors’ claim that noninferiority designs are difficult to interpret and unfamiliar to most clinicians and ethicists, I certainly agree,” Dr. Halpern said. “But that is hardly a reason to not employ the most rigorous methods possible to answer important questions.”
The study was supported by funding from the National Cancer Institute.
A version of this article first appeared on Medscape.com.
Paying people to participate in clinical trials remains controversial. But to date, most reservations are based on hypothetical scenarios or expert opinion with few real-world data to support them.
Research released this week could change that.
Investigators offered nearly 1,300 participants in two clinical trials either no payment or incentives up to $500 to partake in a smoking cessation study or an analysis of a behavioral intervention to increase ambulation in hospitalized patients.
More cash was associated with greater agreement to participate in the smoking cessation study but not the ambulation trial.
But the bigger news may be that offering payment did not appear to get people to accept more risks or skew participation to lower-income individuals, as some ethicists have warned.
“With the publication of our study, investigators finally have data that they can cite to put to rest any lingering concerns about offering moderate incentives in low-risk trials,” lead author Scott D. Halpern, MD, PhD, the John M. Eisenberg Professor of Medicine, Epidemiology, and Medical Ethics & Health Policy at the University of Pennsylvania, Philadelphia, told this news organization.
This initial real-world data centers on low-risk interventions and more research is needed to analyze the ethics and effectiveness of paying people to join clinical trials with more inherent risk, the researchers note.
The study was published online Sept. 20 in JAMA Internal Medicine.
A good first step?
“Payments to research participants are notoriously controversial. Many people oppose payments altogether or insist on minimal payments out of concern that people might be unduly influenced to participate,” Ana S. Iltis, PhD, told this news organization when asked for comment. “Others worry that incentives will disproportionately motivate the less well-off to participate.”
“This is an important study that begins to assess whether these concerns are justified in a real-world context,” added Dr. Iltis, director of the Center for Bioethics, Health and Society and professor of philosophy at Wake Forest University in Winston-Salem, N.C.
In an accompanying invited commentary, Sang Ngo, Anthony S. Kim, MD, and Winston Chiong, MD, PhD, write: “This work is welcome, as it presents experimental data to a bioethical debate that so far has been largely driven by conjecture and competing suppositions.”
The commentary authors, however, question the conclusiveness of the findings. “Interpreting the authors’ findings is complex and illustrates some of the challenges inherent to applying empirical data to ethical problems,” they write.
Recruitment realities
When asked his advice for researchers considering financial incentives, Dr. Halpern said: “All researchers would happily include incentives in their trial budgets if not for concerns that the sponsor or institutional review board might not approve of them.”
“By far the biggest threat to a trial’s success is the inability to enroll enough participants,” he added.
Dr. Iltis agreed, framing the need to boost enrollment in ethical terms. “There is another important ethical issue that often gets ignored, and that is the issue of studies that fail to enroll enough participants and are never completed or are underpowered,” she said.
“These studies end up exposing people to research risks and burdens without a compensating social benefit.”
“If incentives help to increase enrollment and do not necessarily result in undue influence or unfair participant selection, then there might be ethical reasons to offer incentives,” Dr. Iltis added.
Building on previous work assessing financial incentives in hypothetical clinical trials, Dr. Halpern and colleagues studied 654 participants with major depressive disorder in a smoking cessation trial. They also studied another 642 participants in a study that compared a gamification strategy to usual care for encouraging hospitalized patients to get out of bed and walk.
Dr. Halpern and colleagues randomly assigned people in the smoking cessation study to receive no financial compensation, $200, or $500. In the ambulation trial, participants were randomly allocated to receive no compensation, $100, or $300.
Key findings
A total of 22% of those offered no incentive enrolled in the smoking cessation study. In contrast, 36% offered $200 agreed, as did 47% of those offered $500, which the investigators say supports offering cash incentives to boost enrollment. The differences were significant (P < .001).
In contrast, the amount offered did not significantly incentivize more people to participate in the ambulation trial (P = .62). Rates were 45% with no compensation, 48% with $100 payment, and 43% with $300 payment.
In an analysis that adjusted for demographic differences, financial well-being, and Research Attitudes Questionnaire (RAQ-7) scores, each increase in cash incentive increased the odds of enrollment in the smoking cessation trial by 70% (adjusted odds ratio, 1.70; 95% confidence interval, 1.34-2.17).
The same effect was not seen in the ambulation trial, where each higher cash incentive did not make a significant difference (aOR, 0.88; 95% CI, 0.64-1.22).
“The ambulation trial was a lower-risk trial in which patients’ willingness to participate was higher in general. So there were likely fewer people whose participation decisions could be influenced by offers of money,” Dr. Halpern said.
Inducement vs. coercion
The incentives in the study “did not function as unjust inducements, as they were not preferentially motivating across groups with different income levels or financial well-being in either trial,” the researchers note.
Dr. Halpern and colleagues also checked for any perceptions of coercion. More than 70% of participants in each smoking cessation trial group perceived no coercion, as did more than 93% of participants in each ambulation trial group, according to scores on a modified Perceived Coercion Scale of the MacArthur Admission Experience Survey.
Furthermore, perception of risks did not significantly alter the association between cash incentives and enrollment in either trial.
After collecting the findings, Dr. Halpern and colleagues informed participants about their participation in RETAIN and explained the rationale for using different cash incentives. They also let all participants know they would ultimately receive the maximum incentive – either $500 or $300, depending on the trial.
Research implications
A study limitation was reliance on participant risk perception, as was an inability to measure perceived coercion among people who chose not to participant in the trials. Another potential limitation is that “neither of these parent trials posed particularly high risks. Future tests of incentives of different sizes, and in the context of higher-risk parent trials, including trials that test treatments of serious illnesses, are warranted,” the researchers note.
“While there are many more questions to ask and contexts in which to study the effects of incentives, this study calls on opponents of incentivizing research participants with money to be more humble,” Dr. Iltis said. “Incentives might not have the effects they assume they have and which they have long held make such incentives unethical.”
“I encourage researchers who are offering incentives to consider working with people doing ethics research to assess the effects of incentives in their studies,” Dr. Halpern said. “Real-world, as opposed to hypothetical studies that can improve our understanding of the impact of incentives can improve the ethical conduct of research over time.”
Responding to criticism
The authors of the invited commentary questioned the definitions Dr. Halpern and colleagues used for undue or unjust inducement. “Among bioethicists, there is no consensus about what counts as undue inducement or an unjust distribution of research burdens. In this article, the authors have operationalized these constructs based on their own interpretations of undue and unjust inducement, which may not capture all the concerns that scholars have raised about inducement.”
Asked to respond to this and other criticisms raised in the commentary, Dr. Halpern said: “Did our study answer all possible questions about incentives? Absolutely not. But when it comes to incentives for research participation, an ounce of data is worth a pound of conjecture.”
There was agreement, however, that the findings could now put the onus on opponents of financial incentives for trial participants.
“I agree with the commentary’s authors that our study essentially shifts the burden of proof, such that, as they say, ‘those who would limit [incentives’] application may owe us an applicable criterion,’ ” Dr. Halpern said.
The authors of the invited commentary also criticized use of the study’s noninferiority design to rule out undue or unjust inducement. They note this design “may be unfamiliar to many bioethicists and can place substantial evaluative demands on readers.”
“As for the authors’ claim that noninferiority designs are difficult to interpret and unfamiliar to most clinicians and ethicists, I certainly agree,” Dr. Halpern said. “But that is hardly a reason to not employ the most rigorous methods possible to answer important questions.”
The study was supported by funding from the National Cancer Institute.
A version of this article first appeared on Medscape.com.
Paying people to participate in clinical trials remains controversial. But to date, most reservations are based on hypothetical scenarios or expert opinion with few real-world data to support them.
Research released this week could change that.
Investigators offered nearly 1,300 participants in two clinical trials either no payment or incentives up to $500 to partake in a smoking cessation study or an analysis of a behavioral intervention to increase ambulation in hospitalized patients.
More cash was associated with greater agreement to participate in the smoking cessation study but not the ambulation trial.
But the bigger news may be that offering payment did not appear to get people to accept more risks or skew participation to lower-income individuals, as some ethicists have warned.
“With the publication of our study, investigators finally have data that they can cite to put to rest any lingering concerns about offering moderate incentives in low-risk trials,” lead author Scott D. Halpern, MD, PhD, the John M. Eisenberg Professor of Medicine, Epidemiology, and Medical Ethics & Health Policy at the University of Pennsylvania, Philadelphia, told this news organization.
This initial real-world data centers on low-risk interventions and more research is needed to analyze the ethics and effectiveness of paying people to join clinical trials with more inherent risk, the researchers note.
The study was published online Sept. 20 in JAMA Internal Medicine.
A good first step?
“Payments to research participants are notoriously controversial. Many people oppose payments altogether or insist on minimal payments out of concern that people might be unduly influenced to participate,” Ana S. Iltis, PhD, told this news organization when asked for comment. “Others worry that incentives will disproportionately motivate the less well-off to participate.”
“This is an important study that begins to assess whether these concerns are justified in a real-world context,” added Dr. Iltis, director of the Center for Bioethics, Health and Society and professor of philosophy at Wake Forest University in Winston-Salem, N.C.
In an accompanying invited commentary, Sang Ngo, Anthony S. Kim, MD, and Winston Chiong, MD, PhD, write: “This work is welcome, as it presents experimental data to a bioethical debate that so far has been largely driven by conjecture and competing suppositions.”
The commentary authors, however, question the conclusiveness of the findings. “Interpreting the authors’ findings is complex and illustrates some of the challenges inherent to applying empirical data to ethical problems,” they write.
Recruitment realities
When asked his advice for researchers considering financial incentives, Dr. Halpern said: “All researchers would happily include incentives in their trial budgets if not for concerns that the sponsor or institutional review board might not approve of them.”
“By far the biggest threat to a trial’s success is the inability to enroll enough participants,” he added.
Dr. Iltis agreed, framing the need to boost enrollment in ethical terms. “There is another important ethical issue that often gets ignored, and that is the issue of studies that fail to enroll enough participants and are never completed or are underpowered,” she said.
“These studies end up exposing people to research risks and burdens without a compensating social benefit.”
“If incentives help to increase enrollment and do not necessarily result in undue influence or unfair participant selection, then there might be ethical reasons to offer incentives,” Dr. Iltis added.
Building on previous work assessing financial incentives in hypothetical clinical trials, Dr. Halpern and colleagues studied 654 participants with major depressive disorder in a smoking cessation trial. They also studied another 642 participants in a study that compared a gamification strategy to usual care for encouraging hospitalized patients to get out of bed and walk.
Dr. Halpern and colleagues randomly assigned people in the smoking cessation study to receive no financial compensation, $200, or $500. In the ambulation trial, participants were randomly allocated to receive no compensation, $100, or $300.
Key findings
A total of 22% of those offered no incentive enrolled in the smoking cessation study. In contrast, 36% offered $200 agreed, as did 47% of those offered $500, which the investigators say supports offering cash incentives to boost enrollment. The differences were significant (P < .001).
In contrast, the amount offered did not significantly incentivize more people to participate in the ambulation trial (P = .62). Rates were 45% with no compensation, 48% with $100 payment, and 43% with $300 payment.
In an analysis that adjusted for demographic differences, financial well-being, and Research Attitudes Questionnaire (RAQ-7) scores, each increase in cash incentive increased the odds of enrollment in the smoking cessation trial by 70% (adjusted odds ratio, 1.70; 95% confidence interval, 1.34-2.17).
The same effect was not seen in the ambulation trial, where each higher cash incentive did not make a significant difference (aOR, 0.88; 95% CI, 0.64-1.22).
“The ambulation trial was a lower-risk trial in which patients’ willingness to participate was higher in general. So there were likely fewer people whose participation decisions could be influenced by offers of money,” Dr. Halpern said.
Inducement vs. coercion
The incentives in the study “did not function as unjust inducements, as they were not preferentially motivating across groups with different income levels or financial well-being in either trial,” the researchers note.
Dr. Halpern and colleagues also checked for any perceptions of coercion. More than 70% of participants in each smoking cessation trial group perceived no coercion, as did more than 93% of participants in each ambulation trial group, according to scores on a modified Perceived Coercion Scale of the MacArthur Admission Experience Survey.
Furthermore, perception of risks did not significantly alter the association between cash incentives and enrollment in either trial.
After collecting the findings, Dr. Halpern and colleagues informed participants about their participation in RETAIN and explained the rationale for using different cash incentives. They also let all participants know they would ultimately receive the maximum incentive – either $500 or $300, depending on the trial.
Research implications
A study limitation was reliance on participant risk perception, as was an inability to measure perceived coercion among people who chose not to participant in the trials. Another potential limitation is that “neither of these parent trials posed particularly high risks. Future tests of incentives of different sizes, and in the context of higher-risk parent trials, including trials that test treatments of serious illnesses, are warranted,” the researchers note.
“While there are many more questions to ask and contexts in which to study the effects of incentives, this study calls on opponents of incentivizing research participants with money to be more humble,” Dr. Iltis said. “Incentives might not have the effects they assume they have and which they have long held make such incentives unethical.”
“I encourage researchers who are offering incentives to consider working with people doing ethics research to assess the effects of incentives in their studies,” Dr. Halpern said. “Real-world, as opposed to hypothetical studies that can improve our understanding of the impact of incentives can improve the ethical conduct of research over time.”
Responding to criticism
The authors of the invited commentary questioned the definitions Dr. Halpern and colleagues used for undue or unjust inducement. “Among bioethicists, there is no consensus about what counts as undue inducement or an unjust distribution of research burdens. In this article, the authors have operationalized these constructs based on their own interpretations of undue and unjust inducement, which may not capture all the concerns that scholars have raised about inducement.”
Asked to respond to this and other criticisms raised in the commentary, Dr. Halpern said: “Did our study answer all possible questions about incentives? Absolutely not. But when it comes to incentives for research participation, an ounce of data is worth a pound of conjecture.”
There was agreement, however, that the findings could now put the onus on opponents of financial incentives for trial participants.
“I agree with the commentary’s authors that our study essentially shifts the burden of proof, such that, as they say, ‘those who would limit [incentives’] application may owe us an applicable criterion,’ ” Dr. Halpern said.
The authors of the invited commentary also criticized use of the study’s noninferiority design to rule out undue or unjust inducement. They note this design “may be unfamiliar to many bioethicists and can place substantial evaluative demands on readers.”
“As for the authors’ claim that noninferiority designs are difficult to interpret and unfamiliar to most clinicians and ethicists, I certainly agree,” Dr. Halpern said. “But that is hardly a reason to not employ the most rigorous methods possible to answer important questions.”
The study was supported by funding from the National Cancer Institute.
A version of this article first appeared on Medscape.com.
Dr. Judy C. Washington shows URM physicians how to lead
For URM physicians, she also imparts a shared experience of being a minority in the field and helps prepare them for the challenges of facing racism or feeling marginalized or not equitably supported in academic life – and for making change.
While family medicine’s demographics have become more diverse over time, and more so than other specialties, they are not yet representative of the U.S. population. Within academia, male physicians who are Black or African American, or Hispanic or Latino, comprised about 4% and 5% of family medicine faculty, respectively, at the end of 2019, according to data from the Association of American Medical Colleges. For women, these numbers were about 9% and 4%, respectively. (Only those with an MD degree exclusively were included in the report.)
“When you have the privilege to serve in leadership, you have the responsibility to reach back and identify and help others who would not otherwise have the opportunity to be recognized,” Dr. Washington said.
Her mentorship work stems in large part from her long-time involvement and leadership roles in the Society of Teachers of Family Medicine (STFM) – roles she considers a pillar of her professional life. She currently serves as president of the STFM Foundation and is associate chief medical officer of the Atlantic Medical Group, a large multisite physician-led organization. She is also coordinator of women’s health for the Overlook Family Medicine Residency Program, which is affiliated with Atlantic Medical Group.
In Dr. Washington’s role as associate chief medical officer of Atlantic Medical Group in Summit, N.J., she focuses on physician engagement, satisfaction, and diversity. She also assists in areas such as population health. For the Overlook Family Medicine Residency Program also in Summit, she precepts residents in the obstetrics clinic and in the family medicine outpatient clinic.
Diana N. Carvajal, MD, MPH, one of Dr. Washington’s mentees, called her an “inspirational leader” for young academic faculty and said she is a familiar speaker at STFM meetings on topics of workforce diversity, equity, and leadership. She is “passionate” about mentorship, Dr. Carvajal said, and has understood “that URMs and women of color were not always getting [the mentorship they need to be successful].”
Guiding future leaders
Ivonne McLean, MD, assistant professor of family and community medicine at Icahn School of Medicine at Mount Sinai, New York, and an attending at a community health center in the Bronx, called Dr. Washington for advice a couple of years ago when she was considering her next career move.
“She took a genuine interest in me. She never said, this is what you should do. But the questions she asked and the examples she gave from her own life were incredibly helpful to me [in deciding to pursue a research fellowship] ... it was a pivotal conversation,” said Dr. McLean, associate director of a reproductive health fellowship and a research fellow in a New York State–funded program.
“From a lived experience angle, she also told me, here are some of the challenges you’ll have as a woman of color, and here are some of the ways you can approach that,” she said.
Dr. Carvajal, also a URM family physician, credits Dr. Washington’s mentorship with the development of a day-long workshop – held before the annual Society of Teachers of Family Medicine (STFM) meeting – on the low and declining rates of Black males in medicine. “We’d planned it as a presentation, and [she heard of it and] helped us expand it,” she said, calling Dr. Washington “warm, welcoming, and encouraging.
“That work and collaboration with her and the others she brought [into the process] have resulted in publications and more presentations and strategy building for diversifying the workforce,” said Dr. Carvajal, assistant professor, director of reproductive health education in family medicine, and codirector of the research section, all in the department of family and community medicine at the University of Maryland, Baltimore.
STFM involvement
Dr. Washington, who says that all or almost all of her mentees are now leaders in their academic institutions and communities, has been instrumental in developing STFM’s mentoring programming and in facilitating the organization’s multifaceted URM Initiative.
She has been active in STFM since the start of her academic career, and in 2009, while serving as assistant program director for the residency program in which she’d trained, she joined two other African American women, Monique Y. Davis-Smith, MD, and Joedrecka Brown-Speights, MD, in cochairing the society’s Group on Minority and Multicultural Health.
It was in this space, that Dr. Washington said she “heard people’s stories of being in major academic institutions and not feeling supported, not being given roadmaps to success, not getting assistance with publishing, or just kind of feeling like an outsider ... of not being pulled in.” Hispanic and African American females, in particular, “were feeling marginalized,” she said.
In 2018, having co-led development of the STFM Quality Mentoring Program for URM faculty, Dr. Washington was asked to join the STFM Foundation and subsequently led the STFM Foundation’s fundraising campaign for a new URM Initiative. She exceeded her goal, increasing support for URM participation in meetings and activities, and then participated in an STFM steering committee to create broader and longer-lasting support for URM faculty, community teachers, and medical students and residents going into academic family medicine.
Increasing the percentage of URM family medicine faculty in leadership positions – and raising awareness of structural barriers to achievement – is one of the current pillars of the URM Initiative.
Navigating the ‘minority tax’
As part of her mentoring, Dr. Washington helps URM physicians navigate the minority tax – a term referring to the uncompensated citizenship tasks that are more often assigned to Black and other URM physicians than to White physicians, and that take time away from scholarship, further perpetuating inequities.
“Some of our young faculty members find themselves thrust into being the diversity and inclusion leaders in their institutions at a level at which they feel little power and little buy-in from [leadership],” she noted.
A commentary written by Dr. Washington and several colleagues on the minority tax as it impacts women – and the need to build a “tax shelter” to make academic medicine a more just environment for URM women – was published earlier this year in the Journal of Women’s Health.
She also answers e-mails and fields phone calls from young URM faculty who are mulling career moves and facing other familiar challenges.
Physicians who are URM, and African American physicians in particular, tend to “get pulled into the [often underserved] communities, into the patient care and community service areas,” Dr. Washington explained. “But unless you convert these projects into scholarship and publications, and unless you serve on a national committee outside of your institution, you’re not going to be promoted.”
Dr. Washington helps junior faculty envision themselves 5-plus years down the road, find what she calls scholarly “passion projects,” and prepare themselves for their next steps.
She helps her mentees navigate other parts of the continuum of unconscious bias and racism as well, from microaggressions from colleagues to overt discrimination from patients.
“I spend countless minutes fielding texts and phone calls from those who need support,” she wrote in a blog post. “They are a constant reminder that I must continue to speak up when I get the opportunity to do so.”
A journey through family medicine, and through bias and racism
Dr. Washington’s early days in medicine included graduating from Meharry Medical College in 1983 and the Mountainside Family Practice Residency Program in 1990. Following 6 years of working in a private practice in rural Maryland, she moved to academia, spending 6 years at East Tennessee State University and 4 years at the UMDNJ–New Jersey Medical School in Newark as an assistant professor of family medicine.
As had happened in rural Maryland, bias and racism have too often lurked during her career as a physician.
“I grew up in Alabama so I was pretty much ready to deal with racism in the South,” Dr. Washington said. “What I was not ready for was coming to the Northeast and seeing that you’re marginalized because you’re not invited into the room. Or if you do go into spaces when you’re the only one, you often don’t feel as welcomed as you thought you might be.”
Her ideas and contributions were too often dismissed, she wrote in a 2020 blog entry posted on her LinkedIn page. And during contract negotiations, “I was not aware of all the information that my White colleagues had. They had the advantage of inside information.”
Dr. Washington says that “it took a village” to make her who she is today: teachers in her segregated schools in Alabama, one of her college professors, her best friend in medical school – and STFM, “where the list [of her own mentors] is long.”
For URM physicians, she also imparts a shared experience of being a minority in the field and helps prepare them for the challenges of facing racism or feeling marginalized or not equitably supported in academic life – and for making change.
While family medicine’s demographics have become more diverse over time, and more so than other specialties, they are not yet representative of the U.S. population. Within academia, male physicians who are Black or African American, or Hispanic or Latino, comprised about 4% and 5% of family medicine faculty, respectively, at the end of 2019, according to data from the Association of American Medical Colleges. For women, these numbers were about 9% and 4%, respectively. (Only those with an MD degree exclusively were included in the report.)
“When you have the privilege to serve in leadership, you have the responsibility to reach back and identify and help others who would not otherwise have the opportunity to be recognized,” Dr. Washington said.
Her mentorship work stems in large part from her long-time involvement and leadership roles in the Society of Teachers of Family Medicine (STFM) – roles she considers a pillar of her professional life. She currently serves as president of the STFM Foundation and is associate chief medical officer of the Atlantic Medical Group, a large multisite physician-led organization. She is also coordinator of women’s health for the Overlook Family Medicine Residency Program, which is affiliated with Atlantic Medical Group.
In Dr. Washington’s role as associate chief medical officer of Atlantic Medical Group in Summit, N.J., she focuses on physician engagement, satisfaction, and diversity. She also assists in areas such as population health. For the Overlook Family Medicine Residency Program also in Summit, she precepts residents in the obstetrics clinic and in the family medicine outpatient clinic.
Diana N. Carvajal, MD, MPH, one of Dr. Washington’s mentees, called her an “inspirational leader” for young academic faculty and said she is a familiar speaker at STFM meetings on topics of workforce diversity, equity, and leadership. She is “passionate” about mentorship, Dr. Carvajal said, and has understood “that URMs and women of color were not always getting [the mentorship they need to be successful].”
Guiding future leaders
Ivonne McLean, MD, assistant professor of family and community medicine at Icahn School of Medicine at Mount Sinai, New York, and an attending at a community health center in the Bronx, called Dr. Washington for advice a couple of years ago when she was considering her next career move.
“She took a genuine interest in me. She never said, this is what you should do. But the questions she asked and the examples she gave from her own life were incredibly helpful to me [in deciding to pursue a research fellowship] ... it was a pivotal conversation,” said Dr. McLean, associate director of a reproductive health fellowship and a research fellow in a New York State–funded program.
“From a lived experience angle, she also told me, here are some of the challenges you’ll have as a woman of color, and here are some of the ways you can approach that,” she said.
Dr. Carvajal, also a URM family physician, credits Dr. Washington’s mentorship with the development of a day-long workshop – held before the annual Society of Teachers of Family Medicine (STFM) meeting – on the low and declining rates of Black males in medicine. “We’d planned it as a presentation, and [she heard of it and] helped us expand it,” she said, calling Dr. Washington “warm, welcoming, and encouraging.
“That work and collaboration with her and the others she brought [into the process] have resulted in publications and more presentations and strategy building for diversifying the workforce,” said Dr. Carvajal, assistant professor, director of reproductive health education in family medicine, and codirector of the research section, all in the department of family and community medicine at the University of Maryland, Baltimore.
STFM involvement
Dr. Washington, who says that all or almost all of her mentees are now leaders in their academic institutions and communities, has been instrumental in developing STFM’s mentoring programming and in facilitating the organization’s multifaceted URM Initiative.
She has been active in STFM since the start of her academic career, and in 2009, while serving as assistant program director for the residency program in which she’d trained, she joined two other African American women, Monique Y. Davis-Smith, MD, and Joedrecka Brown-Speights, MD, in cochairing the society’s Group on Minority and Multicultural Health.
It was in this space, that Dr. Washington said she “heard people’s stories of being in major academic institutions and not feeling supported, not being given roadmaps to success, not getting assistance with publishing, or just kind of feeling like an outsider ... of not being pulled in.” Hispanic and African American females, in particular, “were feeling marginalized,” she said.
In 2018, having co-led development of the STFM Quality Mentoring Program for URM faculty, Dr. Washington was asked to join the STFM Foundation and subsequently led the STFM Foundation’s fundraising campaign for a new URM Initiative. She exceeded her goal, increasing support for URM participation in meetings and activities, and then participated in an STFM steering committee to create broader and longer-lasting support for URM faculty, community teachers, and medical students and residents going into academic family medicine.
Increasing the percentage of URM family medicine faculty in leadership positions – and raising awareness of structural barriers to achievement – is one of the current pillars of the URM Initiative.
Navigating the ‘minority tax’
As part of her mentoring, Dr. Washington helps URM physicians navigate the minority tax – a term referring to the uncompensated citizenship tasks that are more often assigned to Black and other URM physicians than to White physicians, and that take time away from scholarship, further perpetuating inequities.
“Some of our young faculty members find themselves thrust into being the diversity and inclusion leaders in their institutions at a level at which they feel little power and little buy-in from [leadership],” she noted.
A commentary written by Dr. Washington and several colleagues on the minority tax as it impacts women – and the need to build a “tax shelter” to make academic medicine a more just environment for URM women – was published earlier this year in the Journal of Women’s Health.
She also answers e-mails and fields phone calls from young URM faculty who are mulling career moves and facing other familiar challenges.
Physicians who are URM, and African American physicians in particular, tend to “get pulled into the [often underserved] communities, into the patient care and community service areas,” Dr. Washington explained. “But unless you convert these projects into scholarship and publications, and unless you serve on a national committee outside of your institution, you’re not going to be promoted.”
Dr. Washington helps junior faculty envision themselves 5-plus years down the road, find what she calls scholarly “passion projects,” and prepare themselves for their next steps.
She helps her mentees navigate other parts of the continuum of unconscious bias and racism as well, from microaggressions from colleagues to overt discrimination from patients.
“I spend countless minutes fielding texts and phone calls from those who need support,” she wrote in a blog post. “They are a constant reminder that I must continue to speak up when I get the opportunity to do so.”
A journey through family medicine, and through bias and racism
Dr. Washington’s early days in medicine included graduating from Meharry Medical College in 1983 and the Mountainside Family Practice Residency Program in 1990. Following 6 years of working in a private practice in rural Maryland, she moved to academia, spending 6 years at East Tennessee State University and 4 years at the UMDNJ–New Jersey Medical School in Newark as an assistant professor of family medicine.
As had happened in rural Maryland, bias and racism have too often lurked during her career as a physician.
“I grew up in Alabama so I was pretty much ready to deal with racism in the South,” Dr. Washington said. “What I was not ready for was coming to the Northeast and seeing that you’re marginalized because you’re not invited into the room. Or if you do go into spaces when you’re the only one, you often don’t feel as welcomed as you thought you might be.”
Her ideas and contributions were too often dismissed, she wrote in a 2020 blog entry posted on her LinkedIn page. And during contract negotiations, “I was not aware of all the information that my White colleagues had. They had the advantage of inside information.”
Dr. Washington says that “it took a village” to make her who she is today: teachers in her segregated schools in Alabama, one of her college professors, her best friend in medical school – and STFM, “where the list [of her own mentors] is long.”
For URM physicians, she also imparts a shared experience of being a minority in the field and helps prepare them for the challenges of facing racism or feeling marginalized or not equitably supported in academic life – and for making change.
While family medicine’s demographics have become more diverse over time, and more so than other specialties, they are not yet representative of the U.S. population. Within academia, male physicians who are Black or African American, or Hispanic or Latino, comprised about 4% and 5% of family medicine faculty, respectively, at the end of 2019, according to data from the Association of American Medical Colleges. For women, these numbers were about 9% and 4%, respectively. (Only those with an MD degree exclusively were included in the report.)
“When you have the privilege to serve in leadership, you have the responsibility to reach back and identify and help others who would not otherwise have the opportunity to be recognized,” Dr. Washington said.
Her mentorship work stems in large part from her long-time involvement and leadership roles in the Society of Teachers of Family Medicine (STFM) – roles she considers a pillar of her professional life. She currently serves as president of the STFM Foundation and is associate chief medical officer of the Atlantic Medical Group, a large multisite physician-led organization. She is also coordinator of women’s health for the Overlook Family Medicine Residency Program, which is affiliated with Atlantic Medical Group.
In Dr. Washington’s role as associate chief medical officer of Atlantic Medical Group in Summit, N.J., she focuses on physician engagement, satisfaction, and diversity. She also assists in areas such as population health. For the Overlook Family Medicine Residency Program also in Summit, she precepts residents in the obstetrics clinic and in the family medicine outpatient clinic.
Diana N. Carvajal, MD, MPH, one of Dr. Washington’s mentees, called her an “inspirational leader” for young academic faculty and said she is a familiar speaker at STFM meetings on topics of workforce diversity, equity, and leadership. She is “passionate” about mentorship, Dr. Carvajal said, and has understood “that URMs and women of color were not always getting [the mentorship they need to be successful].”
Guiding future leaders
Ivonne McLean, MD, assistant professor of family and community medicine at Icahn School of Medicine at Mount Sinai, New York, and an attending at a community health center in the Bronx, called Dr. Washington for advice a couple of years ago when she was considering her next career move.
“She took a genuine interest in me. She never said, this is what you should do. But the questions she asked and the examples she gave from her own life were incredibly helpful to me [in deciding to pursue a research fellowship] ... it was a pivotal conversation,” said Dr. McLean, associate director of a reproductive health fellowship and a research fellow in a New York State–funded program.
“From a lived experience angle, she also told me, here are some of the challenges you’ll have as a woman of color, and here are some of the ways you can approach that,” she said.
Dr. Carvajal, also a URM family physician, credits Dr. Washington’s mentorship with the development of a day-long workshop – held before the annual Society of Teachers of Family Medicine (STFM) meeting – on the low and declining rates of Black males in medicine. “We’d planned it as a presentation, and [she heard of it and] helped us expand it,” she said, calling Dr. Washington “warm, welcoming, and encouraging.
“That work and collaboration with her and the others she brought [into the process] have resulted in publications and more presentations and strategy building for diversifying the workforce,” said Dr. Carvajal, assistant professor, director of reproductive health education in family medicine, and codirector of the research section, all in the department of family and community medicine at the University of Maryland, Baltimore.
STFM involvement
Dr. Washington, who says that all or almost all of her mentees are now leaders in their academic institutions and communities, has been instrumental in developing STFM’s mentoring programming and in facilitating the organization’s multifaceted URM Initiative.
She has been active in STFM since the start of her academic career, and in 2009, while serving as assistant program director for the residency program in which she’d trained, she joined two other African American women, Monique Y. Davis-Smith, MD, and Joedrecka Brown-Speights, MD, in cochairing the society’s Group on Minority and Multicultural Health.
It was in this space, that Dr. Washington said she “heard people’s stories of being in major academic institutions and not feeling supported, not being given roadmaps to success, not getting assistance with publishing, or just kind of feeling like an outsider ... of not being pulled in.” Hispanic and African American females, in particular, “were feeling marginalized,” she said.
In 2018, having co-led development of the STFM Quality Mentoring Program for URM faculty, Dr. Washington was asked to join the STFM Foundation and subsequently led the STFM Foundation’s fundraising campaign for a new URM Initiative. She exceeded her goal, increasing support for URM participation in meetings and activities, and then participated in an STFM steering committee to create broader and longer-lasting support for URM faculty, community teachers, and medical students and residents going into academic family medicine.
Increasing the percentage of URM family medicine faculty in leadership positions – and raising awareness of structural barriers to achievement – is one of the current pillars of the URM Initiative.
Navigating the ‘minority tax’
As part of her mentoring, Dr. Washington helps URM physicians navigate the minority tax – a term referring to the uncompensated citizenship tasks that are more often assigned to Black and other URM physicians than to White physicians, and that take time away from scholarship, further perpetuating inequities.
“Some of our young faculty members find themselves thrust into being the diversity and inclusion leaders in their institutions at a level at which they feel little power and little buy-in from [leadership],” she noted.
A commentary written by Dr. Washington and several colleagues on the minority tax as it impacts women – and the need to build a “tax shelter” to make academic medicine a more just environment for URM women – was published earlier this year in the Journal of Women’s Health.
She also answers e-mails and fields phone calls from young URM faculty who are mulling career moves and facing other familiar challenges.
Physicians who are URM, and African American physicians in particular, tend to “get pulled into the [often underserved] communities, into the patient care and community service areas,” Dr. Washington explained. “But unless you convert these projects into scholarship and publications, and unless you serve on a national committee outside of your institution, you’re not going to be promoted.”
Dr. Washington helps junior faculty envision themselves 5-plus years down the road, find what she calls scholarly “passion projects,” and prepare themselves for their next steps.
She helps her mentees navigate other parts of the continuum of unconscious bias and racism as well, from microaggressions from colleagues to overt discrimination from patients.
“I spend countless minutes fielding texts and phone calls from those who need support,” she wrote in a blog post. “They are a constant reminder that I must continue to speak up when I get the opportunity to do so.”
A journey through family medicine, and through bias and racism
Dr. Washington’s early days in medicine included graduating from Meharry Medical College in 1983 and the Mountainside Family Practice Residency Program in 1990. Following 6 years of working in a private practice in rural Maryland, she moved to academia, spending 6 years at East Tennessee State University and 4 years at the UMDNJ–New Jersey Medical School in Newark as an assistant professor of family medicine.
As had happened in rural Maryland, bias and racism have too often lurked during her career as a physician.
“I grew up in Alabama so I was pretty much ready to deal with racism in the South,” Dr. Washington said. “What I was not ready for was coming to the Northeast and seeing that you’re marginalized because you’re not invited into the room. Or if you do go into spaces when you’re the only one, you often don’t feel as welcomed as you thought you might be.”
Her ideas and contributions were too often dismissed, she wrote in a 2020 blog entry posted on her LinkedIn page. And during contract negotiations, “I was not aware of all the information that my White colleagues had. They had the advantage of inside information.”
Dr. Washington says that “it took a village” to make her who she is today: teachers in her segregated schools in Alabama, one of her college professors, her best friend in medical school – and STFM, “where the list [of her own mentors] is long.”
Military sexual trauma tied to risk for hypertension
a new study suggests.
“Understanding a patient’s trauma history is invaluable for treating the whole person,” Allison E. Gaffey, PhD, Yale University, New Haven, Conn., and the Veterans Affairs Connecticut Healthcare System, told this news organization.
“Assessing men and women’s history of trauma, including sexual trauma, is critical for recognizing nontraditional factors that contribute to their cardiovascular risk and to gain a more comprehensive understanding of their mental and physical health,” Dr. Gaffey added.
She presented her research at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
Lasting impact on physical health
Dr. Gaffey and colleagues analyzed data from the VA for roughly 1.2 million veterans (mean age, 30.2 years; 12% female) who were discharged from the military after Sept. 30, 2001, and who received health care services at VA medical centers from 2001 to 2017.
All were screened for sexual harassment and assault, known as military sexual trauma (MST), when they first began receiving VA care.
During 16 years of follow-up, 33,881 veterans screened positive for MST (65% women), and 307,332 developed hypertension (15% women).
Overall, MST was associated with a 30% increase in risk for incident hypertension in unadjusted models (hazard ratio, 1.30; 95% confidence interval, 1.28-1.33; P < .001).
After adjustment for demographic characteristics, lifestyle factors, cardiovascular comorbidities, PTSD, anxiety, and depression, MST remained significantly associated with hypertension (adjusted HR, 1.10; 95% CI, 1.08-1.12; P < .001).
When women and men were examined separately, the link between MST and risk for hypertension remained for both groups, but was slightly stronger among women.
“Sexual trauma has been associated with autonomic dysfunction, inflammation, and dysregulation in the hypothalamic pituitary adrenal axis and renin-angiotensin-aldosterone system, which could lead to elevations in BP over time,” Dr. Gaffey told this news organization.
“These findings show that even many years after being discharged from military service, exposure to military sexual trauma can continue to significantly influence veterans’ physical health,” she added.
Dr. Gaffey said it will be important to determine if early treatment of MST improves hypertension risk, particularly among those showing elevated blood pressure or stage 1 hypertension.
Social determinants of health
Willie Lawrence Jr., MD, head of the AHA National Hypertension Control Initiative oversight committee, said the findings in this study are “in line with what we know about the impact of social determinants of health on high blood pressure.”
“There are studies that suggest that things that we historically don’t look at as risk factors for hypertension – lifelong racism, crime, mental health status – do in fact predict your risk of developing hypertension,” Dr. Lawrence, from Spectrum Health in Benton Harbor, Mich., told this news organization.
“It’s not just your genetics that will determine your health, and there are a lot of things that will affect your blood pressure. Your blood pressure is really just a barometer of everything that’s going on in your life and some of the things that have gone on in your life in the past,” added Dr. Lawrence, who wasn’t involved in the study.
Funding for the study was provided by the Department of Veterans Affairs. Dr. Gaffey and Dr. Lawrence have no relevant disclosures.
A version of this article first appeared on Medscape.com.
a new study suggests.
“Understanding a patient’s trauma history is invaluable for treating the whole person,” Allison E. Gaffey, PhD, Yale University, New Haven, Conn., and the Veterans Affairs Connecticut Healthcare System, told this news organization.
“Assessing men and women’s history of trauma, including sexual trauma, is critical for recognizing nontraditional factors that contribute to their cardiovascular risk and to gain a more comprehensive understanding of their mental and physical health,” Dr. Gaffey added.
She presented her research at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
Lasting impact on physical health
Dr. Gaffey and colleagues analyzed data from the VA for roughly 1.2 million veterans (mean age, 30.2 years; 12% female) who were discharged from the military after Sept. 30, 2001, and who received health care services at VA medical centers from 2001 to 2017.
All were screened for sexual harassment and assault, known as military sexual trauma (MST), when they first began receiving VA care.
During 16 years of follow-up, 33,881 veterans screened positive for MST (65% women), and 307,332 developed hypertension (15% women).
Overall, MST was associated with a 30% increase in risk for incident hypertension in unadjusted models (hazard ratio, 1.30; 95% confidence interval, 1.28-1.33; P < .001).
After adjustment for demographic characteristics, lifestyle factors, cardiovascular comorbidities, PTSD, anxiety, and depression, MST remained significantly associated with hypertension (adjusted HR, 1.10; 95% CI, 1.08-1.12; P < .001).
When women and men were examined separately, the link between MST and risk for hypertension remained for both groups, but was slightly stronger among women.
“Sexual trauma has been associated with autonomic dysfunction, inflammation, and dysregulation in the hypothalamic pituitary adrenal axis and renin-angiotensin-aldosterone system, which could lead to elevations in BP over time,” Dr. Gaffey told this news organization.
“These findings show that even many years after being discharged from military service, exposure to military sexual trauma can continue to significantly influence veterans’ physical health,” she added.
Dr. Gaffey said it will be important to determine if early treatment of MST improves hypertension risk, particularly among those showing elevated blood pressure or stage 1 hypertension.
Social determinants of health
Willie Lawrence Jr., MD, head of the AHA National Hypertension Control Initiative oversight committee, said the findings in this study are “in line with what we know about the impact of social determinants of health on high blood pressure.”
“There are studies that suggest that things that we historically don’t look at as risk factors for hypertension – lifelong racism, crime, mental health status – do in fact predict your risk of developing hypertension,” Dr. Lawrence, from Spectrum Health in Benton Harbor, Mich., told this news organization.
“It’s not just your genetics that will determine your health, and there are a lot of things that will affect your blood pressure. Your blood pressure is really just a barometer of everything that’s going on in your life and some of the things that have gone on in your life in the past,” added Dr. Lawrence, who wasn’t involved in the study.
Funding for the study was provided by the Department of Veterans Affairs. Dr. Gaffey and Dr. Lawrence have no relevant disclosures.
A version of this article first appeared on Medscape.com.
a new study suggests.
“Understanding a patient’s trauma history is invaluable for treating the whole person,” Allison E. Gaffey, PhD, Yale University, New Haven, Conn., and the Veterans Affairs Connecticut Healthcare System, told this news organization.
“Assessing men and women’s history of trauma, including sexual trauma, is critical for recognizing nontraditional factors that contribute to their cardiovascular risk and to gain a more comprehensive understanding of their mental and physical health,” Dr. Gaffey added.
She presented her research at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
Lasting impact on physical health
Dr. Gaffey and colleagues analyzed data from the VA for roughly 1.2 million veterans (mean age, 30.2 years; 12% female) who were discharged from the military after Sept. 30, 2001, and who received health care services at VA medical centers from 2001 to 2017.
All were screened for sexual harassment and assault, known as military sexual trauma (MST), when they first began receiving VA care.
During 16 years of follow-up, 33,881 veterans screened positive for MST (65% women), and 307,332 developed hypertension (15% women).
Overall, MST was associated with a 30% increase in risk for incident hypertension in unadjusted models (hazard ratio, 1.30; 95% confidence interval, 1.28-1.33; P < .001).
After adjustment for demographic characteristics, lifestyle factors, cardiovascular comorbidities, PTSD, anxiety, and depression, MST remained significantly associated with hypertension (adjusted HR, 1.10; 95% CI, 1.08-1.12; P < .001).
When women and men were examined separately, the link between MST and risk for hypertension remained for both groups, but was slightly stronger among women.
“Sexual trauma has been associated with autonomic dysfunction, inflammation, and dysregulation in the hypothalamic pituitary adrenal axis and renin-angiotensin-aldosterone system, which could lead to elevations in BP over time,” Dr. Gaffey told this news organization.
“These findings show that even many years after being discharged from military service, exposure to military sexual trauma can continue to significantly influence veterans’ physical health,” she added.
Dr. Gaffey said it will be important to determine if early treatment of MST improves hypertension risk, particularly among those showing elevated blood pressure or stage 1 hypertension.
Social determinants of health
Willie Lawrence Jr., MD, head of the AHA National Hypertension Control Initiative oversight committee, said the findings in this study are “in line with what we know about the impact of social determinants of health on high blood pressure.”
“There are studies that suggest that things that we historically don’t look at as risk factors for hypertension – lifelong racism, crime, mental health status – do in fact predict your risk of developing hypertension,” Dr. Lawrence, from Spectrum Health in Benton Harbor, Mich., told this news organization.
“It’s not just your genetics that will determine your health, and there are a lot of things that will affect your blood pressure. Your blood pressure is really just a barometer of everything that’s going on in your life and some of the things that have gone on in your life in the past,” added Dr. Lawrence, who wasn’t involved in the study.
Funding for the study was provided by the Department of Veterans Affairs. Dr. Gaffey and Dr. Lawrence have no relevant disclosures.
A version of this article first appeared on Medscape.com.
Booster shot back-and-forth creates uncertainty, confusion
Many people are confused — patients and healthcare providers alike — in the wake of the U.S. Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) announcements about who is authorized to get a third or ‘booster’ shot of the Pfizer/BioNTech COVID-19 vaccine.
, healthcare providers report. At the same time, the uncertainty from changing federal messages about boosters is causing some chaos, especially in the form of vaccine misinformation.
The confusion started, in part, with the August 13 announcement that immunocompromised Americans were eligible for a booster shot. Next came the initial Biden administration intention to provide most U.S. adults with a third shot starting September 20 — an announcement later rolled back — followed by the Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) limiting boosters to select groups last week.
“It was only 3% of the population that was going to be getting a third dose, then it was back to everyone being able to get the booster, and then it’s back to a select crew,” Louito Edje, MD, a family physician in private practice in Cincinnati, said in an interview with this news organization.
This kind of mixed messaging is generating more questions than answers.
“Even though that is following the science, translating the science into policy, it’s really fraught with confusion for patients, especially,” added Dr. Edje, professor educator in the departments of medical education and family and community medicine at UC Health and a fellow of the American Academy of Family Physicians.
When asked if he’s seeing more uncertainty about boosters, community pharmacist Brian Caswell, RPh, said: “I’m going to have to say yes because I’ve been confused myself at times.”
“Yes, there is a lot of confusion,” added Mr. Caswell, owner or co-owner of four pharmacies in Kansas and Missouri and president of the National Community Pharmacists Association.
Boosting misinformation?
“Unfortunately, confusion leads to an acceleration of misinformation,” Mr. Caswell said.
Dr. Edje shared an example. “The folks who have been hesitant to even get the first vaccine appear now a little less likely to want to go ahead and get vaccinated.”
These patients point to breakthrough COVID-19 cases of the Delta variant, which “reinforces that they don’t need to get vaccinated in the first place,” Dr. Edje said.
“That’s unfortunate because it’s a complete fallacy.”
Clearer communication from the federal government could help alleviate confusion, Mr. Caswell said. “I would like to see an official CDC chart that states who is eligible as of a certain date. Something that is accessible through their webpage or a social media source that can be updated. That would help all of us.”
“For myself, I’ve got patients from Kansas, Oklahoma, and Missouri that might be operating under different guidelines. That makes it even more confusing,” he said.
More clarity is needed for individuals seeking boosters as well. “It would help to be very clear with the general public, who are becoming very knowledgeable within this vaccine realm,” Mr. Caswell said.
‘Gaming the system’
Although most people seeking a booster shot at one of Caswell’s pharmacies are following official recommendations, there are some who remain ineligible but nonetheless come in for an additional vaccine.
“Even before this announcement last Friday, in the latter part of August when the CDC talked about a booster for immunocompromised, we had interest from people who did not meet the criteria,” Mr. Caswell said.
To the ineligible, he and his staff explain the approval process, why certain decisions are made, and point out that the number of eligible Americans is likely to expand in the future.
“The vast majority of them are understanding,” Mr. Caswell said. “But we’ve had some people who really didn’t want to accept the information, and I don’t know what they’ve done.”
“Some people are gaming the system to get their booster or second shot of J&J,” he said.
For example, Mr. Caswell had a patient who crossed over state lines from Missouri seeking a vaccine booster at Wolkar Drug, a pharmacy in Baxter Springs, Kan. “We found out later he had a J&J shot at a facility or provider in Missouri. He came over to Kansas, signed up for it and got a booster with Moderna.”
“We called and asked him if he was aware of it. He said, ‘yes.’ When we questioned him more about it, he hung up.”
Dr. Edje is likewise seeing interest from some ineligible patients, she said.
Crossing a liability line?
Mr. Caswell has asked for advice from lawyers and the State Board of Pharmacy on potential liability if a pharmacist gives a booster to a patient not eligible under the official FDA and CDC guidance.
“We ask patients direct questions about whether they’ve had the COVID vaccine, COVID, and a whole litany of questions they must answer. And we’re assuming they are going to be honest and forthright,” he said. “The pharmacist needs to make sure they make every effort to get that information from the patient.”
Normally, healthcare providers like Mr. Caswell report each COVID-19 vaccination to the state registry after administration. “We have not gone through a police action and checked the registry first,” he said.
But, if people continue to try ‘gaming the system,’ he said, he might have to start checking the state registry before giving someone a booster.
The American Academy of Family Physicians offers advice from the CDC about legal protections for providers.
“As outlined by CDC, any off-label use of the Comirnaty/Pfizer-BioNTech COVID-19 vaccine is not authorized at this time and may not be covered under the PREP Act or the PREP Act declaration. This means that clinicians providing the vaccine outside of the authorized/approved use may not have immunity from claims,” the AAFP website states.
“Per CDC, individuals who receive a third dose may not be eligible for compensation after a possible adverse event. Such use would be in violation of the CDC COVID-19 vaccination program provider agreement and therefore may not be reimbursable and may impact the ability of a provider to remain in the CDC program, in addition to other potential sanctions. Administration fees for off-label doses may not be reimbursed by payers.”
Despite confusion, demand is up
Even amid all the uncertainty, there appears to be a jump in enthusiasm for the booster shots.
“The requests have gone up quite a bit. We’ve seen a number of requests from people in person and over the phone looking to get a booster,” Mr. Caswell said. “Since the discussion at the federal level...there has been a lot of interest in the third shot booster, itself, as well as about a booster for J&J.”
“There is quite a bit of excitement out there,” he said.
Dr. Edje agreed: “I take care of a fair number of folks...including the elderly and healthcare professionals. They are already asking for the booster.”
Interestingly, Dr. Edje would like to get a booster herself but is not eligible for the Pfizer third shot. She is a participant in a Moderna vaccine trial and can only receive additional immunization as part of the study.
‘Walk, don’t run’
To quell any potential early rush to get a third shot, U.S. health officials are reminding booster-ineligible people that they still have some protection against COVID-19.
“If you’re a person who ultimately might get a booster that will make you optimally protected, you don’t necessarily need to get it tomorrow,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases told CNN.
CDC Director Rochelle Walensky, MD, also weighed in. She told ABC that boosters for people who received a Moderna or Johnson & Johnson vaccine will be addressed with urgency.
“I want to reiterate that this is a very slow wane. There is no urgency here to go and get your booster immediately. You know, walk don’t run to your booster appointment,” she said.
“We will come and look at the data for Moderna and J&J in very short order.”
Dr. Edje and Mr. Caswell have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Many people are confused — patients and healthcare providers alike — in the wake of the U.S. Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) announcements about who is authorized to get a third or ‘booster’ shot of the Pfizer/BioNTech COVID-19 vaccine.
, healthcare providers report. At the same time, the uncertainty from changing federal messages about boosters is causing some chaos, especially in the form of vaccine misinformation.
The confusion started, in part, with the August 13 announcement that immunocompromised Americans were eligible for a booster shot. Next came the initial Biden administration intention to provide most U.S. adults with a third shot starting September 20 — an announcement later rolled back — followed by the Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) limiting boosters to select groups last week.
“It was only 3% of the population that was going to be getting a third dose, then it was back to everyone being able to get the booster, and then it’s back to a select crew,” Louito Edje, MD, a family physician in private practice in Cincinnati, said in an interview with this news organization.
This kind of mixed messaging is generating more questions than answers.
“Even though that is following the science, translating the science into policy, it’s really fraught with confusion for patients, especially,” added Dr. Edje, professor educator in the departments of medical education and family and community medicine at UC Health and a fellow of the American Academy of Family Physicians.
When asked if he’s seeing more uncertainty about boosters, community pharmacist Brian Caswell, RPh, said: “I’m going to have to say yes because I’ve been confused myself at times.”
“Yes, there is a lot of confusion,” added Mr. Caswell, owner or co-owner of four pharmacies in Kansas and Missouri and president of the National Community Pharmacists Association.
Boosting misinformation?
“Unfortunately, confusion leads to an acceleration of misinformation,” Mr. Caswell said.
Dr. Edje shared an example. “The folks who have been hesitant to even get the first vaccine appear now a little less likely to want to go ahead and get vaccinated.”
These patients point to breakthrough COVID-19 cases of the Delta variant, which “reinforces that they don’t need to get vaccinated in the first place,” Dr. Edje said.
“That’s unfortunate because it’s a complete fallacy.”
Clearer communication from the federal government could help alleviate confusion, Mr. Caswell said. “I would like to see an official CDC chart that states who is eligible as of a certain date. Something that is accessible through their webpage or a social media source that can be updated. That would help all of us.”
“For myself, I’ve got patients from Kansas, Oklahoma, and Missouri that might be operating under different guidelines. That makes it even more confusing,” he said.
More clarity is needed for individuals seeking boosters as well. “It would help to be very clear with the general public, who are becoming very knowledgeable within this vaccine realm,” Mr. Caswell said.
‘Gaming the system’
Although most people seeking a booster shot at one of Caswell’s pharmacies are following official recommendations, there are some who remain ineligible but nonetheless come in for an additional vaccine.
“Even before this announcement last Friday, in the latter part of August when the CDC talked about a booster for immunocompromised, we had interest from people who did not meet the criteria,” Mr. Caswell said.
To the ineligible, he and his staff explain the approval process, why certain decisions are made, and point out that the number of eligible Americans is likely to expand in the future.
“The vast majority of them are understanding,” Mr. Caswell said. “But we’ve had some people who really didn’t want to accept the information, and I don’t know what they’ve done.”
“Some people are gaming the system to get their booster or second shot of J&J,” he said.
For example, Mr. Caswell had a patient who crossed over state lines from Missouri seeking a vaccine booster at Wolkar Drug, a pharmacy in Baxter Springs, Kan. “We found out later he had a J&J shot at a facility or provider in Missouri. He came over to Kansas, signed up for it and got a booster with Moderna.”
“We called and asked him if he was aware of it. He said, ‘yes.’ When we questioned him more about it, he hung up.”
Dr. Edje is likewise seeing interest from some ineligible patients, she said.
Crossing a liability line?
Mr. Caswell has asked for advice from lawyers and the State Board of Pharmacy on potential liability if a pharmacist gives a booster to a patient not eligible under the official FDA and CDC guidance.
“We ask patients direct questions about whether they’ve had the COVID vaccine, COVID, and a whole litany of questions they must answer. And we’re assuming they are going to be honest and forthright,” he said. “The pharmacist needs to make sure they make every effort to get that information from the patient.”
Normally, healthcare providers like Mr. Caswell report each COVID-19 vaccination to the state registry after administration. “We have not gone through a police action and checked the registry first,” he said.
But, if people continue to try ‘gaming the system,’ he said, he might have to start checking the state registry before giving someone a booster.
The American Academy of Family Physicians offers advice from the CDC about legal protections for providers.
“As outlined by CDC, any off-label use of the Comirnaty/Pfizer-BioNTech COVID-19 vaccine is not authorized at this time and may not be covered under the PREP Act or the PREP Act declaration. This means that clinicians providing the vaccine outside of the authorized/approved use may not have immunity from claims,” the AAFP website states.
“Per CDC, individuals who receive a third dose may not be eligible for compensation after a possible adverse event. Such use would be in violation of the CDC COVID-19 vaccination program provider agreement and therefore may not be reimbursable and may impact the ability of a provider to remain in the CDC program, in addition to other potential sanctions. Administration fees for off-label doses may not be reimbursed by payers.”
Despite confusion, demand is up
Even amid all the uncertainty, there appears to be a jump in enthusiasm for the booster shots.
“The requests have gone up quite a bit. We’ve seen a number of requests from people in person and over the phone looking to get a booster,” Mr. Caswell said. “Since the discussion at the federal level...there has been a lot of interest in the third shot booster, itself, as well as about a booster for J&J.”
“There is quite a bit of excitement out there,” he said.
Dr. Edje agreed: “I take care of a fair number of folks...including the elderly and healthcare professionals. They are already asking for the booster.”
Interestingly, Dr. Edje would like to get a booster herself but is not eligible for the Pfizer third shot. She is a participant in a Moderna vaccine trial and can only receive additional immunization as part of the study.
‘Walk, don’t run’
To quell any potential early rush to get a third shot, U.S. health officials are reminding booster-ineligible people that they still have some protection against COVID-19.
“If you’re a person who ultimately might get a booster that will make you optimally protected, you don’t necessarily need to get it tomorrow,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases told CNN.
CDC Director Rochelle Walensky, MD, also weighed in. She told ABC that boosters for people who received a Moderna or Johnson & Johnson vaccine will be addressed with urgency.
“I want to reiterate that this is a very slow wane. There is no urgency here to go and get your booster immediately. You know, walk don’t run to your booster appointment,” she said.
“We will come and look at the data for Moderna and J&J in very short order.”
Dr. Edje and Mr. Caswell have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Many people are confused — patients and healthcare providers alike — in the wake of the U.S. Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) announcements about who is authorized to get a third or ‘booster’ shot of the Pfizer/BioNTech COVID-19 vaccine.
, healthcare providers report. At the same time, the uncertainty from changing federal messages about boosters is causing some chaos, especially in the form of vaccine misinformation.
The confusion started, in part, with the August 13 announcement that immunocompromised Americans were eligible for a booster shot. Next came the initial Biden administration intention to provide most U.S. adults with a third shot starting September 20 — an announcement later rolled back — followed by the Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) limiting boosters to select groups last week.
“It was only 3% of the population that was going to be getting a third dose, then it was back to everyone being able to get the booster, and then it’s back to a select crew,” Louito Edje, MD, a family physician in private practice in Cincinnati, said in an interview with this news organization.
This kind of mixed messaging is generating more questions than answers.
“Even though that is following the science, translating the science into policy, it’s really fraught with confusion for patients, especially,” added Dr. Edje, professor educator in the departments of medical education and family and community medicine at UC Health and a fellow of the American Academy of Family Physicians.
When asked if he’s seeing more uncertainty about boosters, community pharmacist Brian Caswell, RPh, said: “I’m going to have to say yes because I’ve been confused myself at times.”
“Yes, there is a lot of confusion,” added Mr. Caswell, owner or co-owner of four pharmacies in Kansas and Missouri and president of the National Community Pharmacists Association.
Boosting misinformation?
“Unfortunately, confusion leads to an acceleration of misinformation,” Mr. Caswell said.
Dr. Edje shared an example. “The folks who have been hesitant to even get the first vaccine appear now a little less likely to want to go ahead and get vaccinated.”
These patients point to breakthrough COVID-19 cases of the Delta variant, which “reinforces that they don’t need to get vaccinated in the first place,” Dr. Edje said.
“That’s unfortunate because it’s a complete fallacy.”
Clearer communication from the federal government could help alleviate confusion, Mr. Caswell said. “I would like to see an official CDC chart that states who is eligible as of a certain date. Something that is accessible through their webpage or a social media source that can be updated. That would help all of us.”
“For myself, I’ve got patients from Kansas, Oklahoma, and Missouri that might be operating under different guidelines. That makes it even more confusing,” he said.
More clarity is needed for individuals seeking boosters as well. “It would help to be very clear with the general public, who are becoming very knowledgeable within this vaccine realm,” Mr. Caswell said.
‘Gaming the system’
Although most people seeking a booster shot at one of Caswell’s pharmacies are following official recommendations, there are some who remain ineligible but nonetheless come in for an additional vaccine.
“Even before this announcement last Friday, in the latter part of August when the CDC talked about a booster for immunocompromised, we had interest from people who did not meet the criteria,” Mr. Caswell said.
To the ineligible, he and his staff explain the approval process, why certain decisions are made, and point out that the number of eligible Americans is likely to expand in the future.
“The vast majority of them are understanding,” Mr. Caswell said. “But we’ve had some people who really didn’t want to accept the information, and I don’t know what they’ve done.”
“Some people are gaming the system to get their booster or second shot of J&J,” he said.
For example, Mr. Caswell had a patient who crossed over state lines from Missouri seeking a vaccine booster at Wolkar Drug, a pharmacy in Baxter Springs, Kan. “We found out later he had a J&J shot at a facility or provider in Missouri. He came over to Kansas, signed up for it and got a booster with Moderna.”
“We called and asked him if he was aware of it. He said, ‘yes.’ When we questioned him more about it, he hung up.”
Dr. Edje is likewise seeing interest from some ineligible patients, she said.
Crossing a liability line?
Mr. Caswell has asked for advice from lawyers and the State Board of Pharmacy on potential liability if a pharmacist gives a booster to a patient not eligible under the official FDA and CDC guidance.
“We ask patients direct questions about whether they’ve had the COVID vaccine, COVID, and a whole litany of questions they must answer. And we’re assuming they are going to be honest and forthright,” he said. “The pharmacist needs to make sure they make every effort to get that information from the patient.”
Normally, healthcare providers like Mr. Caswell report each COVID-19 vaccination to the state registry after administration. “We have not gone through a police action and checked the registry first,” he said.
But, if people continue to try ‘gaming the system,’ he said, he might have to start checking the state registry before giving someone a booster.
The American Academy of Family Physicians offers advice from the CDC about legal protections for providers.
“As outlined by CDC, any off-label use of the Comirnaty/Pfizer-BioNTech COVID-19 vaccine is not authorized at this time and may not be covered under the PREP Act or the PREP Act declaration. This means that clinicians providing the vaccine outside of the authorized/approved use may not have immunity from claims,” the AAFP website states.
“Per CDC, individuals who receive a third dose may not be eligible for compensation after a possible adverse event. Such use would be in violation of the CDC COVID-19 vaccination program provider agreement and therefore may not be reimbursable and may impact the ability of a provider to remain in the CDC program, in addition to other potential sanctions. Administration fees for off-label doses may not be reimbursed by payers.”
Despite confusion, demand is up
Even amid all the uncertainty, there appears to be a jump in enthusiasm for the booster shots.
“The requests have gone up quite a bit. We’ve seen a number of requests from people in person and over the phone looking to get a booster,” Mr. Caswell said. “Since the discussion at the federal level...there has been a lot of interest in the third shot booster, itself, as well as about a booster for J&J.”
“There is quite a bit of excitement out there,” he said.
Dr. Edje agreed: “I take care of a fair number of folks...including the elderly and healthcare professionals. They are already asking for the booster.”
Interestingly, Dr. Edje would like to get a booster herself but is not eligible for the Pfizer third shot. She is a participant in a Moderna vaccine trial and can only receive additional immunization as part of the study.
‘Walk, don’t run’
To quell any potential early rush to get a third shot, U.S. health officials are reminding booster-ineligible people that they still have some protection against COVID-19.
“If you’re a person who ultimately might get a booster that will make you optimally protected, you don’t necessarily need to get it tomorrow,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases told CNN.
CDC Director Rochelle Walensky, MD, also weighed in. She told ABC that boosters for people who received a Moderna or Johnson & Johnson vaccine will be addressed with urgency.
“I want to reiterate that this is a very slow wane. There is no urgency here to go and get your booster immediately. You know, walk don’t run to your booster appointment,” she said.
“We will come and look at the data for Moderna and J&J in very short order.”
Dr. Edje and Mr. Caswell have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cook your amphibians before you eat them
Novel food for thought
When you were growing up, your parents probably told you to brush your teeth before you went to bed, warned you not to run with the scissors or play with matches, and punished you whenever you used the neighbor children to play Schrödinger’s cat.
They did those things for your own good, of course, and now the nation’s mother – the Centers for Disease Control and Prevention – is doing the same by warning us about novel outbreak–associated foods. As in, “Put down that novel outbreak–associated food! You don’t know where it’s been!”
Seriously, you don’t know where it’s been. CDC investigators identified 28 novel foods that were linked to 36 foodborne-disease outbreaks that occurred during 2007-2016, including moringa leaf (herb/spice), tempeh (grain), frog, sprouted nut butter, and skate.
The novel foods implicated in these outbreaks were more likely to be imported, compared with 14,216 outbreaks that occurred from 1973 to 2016, and about half didn’t require refrigeration. Two-thirds did not need to be cooked after purchase. Another thing your parents wouldn’t like: Some can’t be washed, like sheep milk, sugar cane, or the aforementioned nut butter.
We wanted to get a food expert to comment on these novel foods, but our editor said that the assistant manager of our local Burger King wasn’t expert enough, so we’ve commandeered someone else’s expert. Cynthia Sears, MD, of Johns Hopkins University in Baltimore, told Today.com all about the dangers of frogs: “Essentially all amphibians are contaminated, often with salmonella. Eating any amphibian that is not thoroughly cooked is a risk.”
Be sure to cook your amphibians before you eat them. Advice that your parents would be proud to share.
Dieters should stay away from diet drinks
When a drink is labeled “diet” many assume that the calorie-free beverage is the best choice. However, one of the largest studies to date on artificial sweeteners is out to set the record straight.
Artificial sweeteners, or nonnutritive sweeteners (NNS), are used in most if not all diet products to give the illusion of sweetness without the caloric guilt. Some studies say they help with weight loss for that very reason, but others say they can contribute to weight gain. So which is it?
Researchers at the University of Southern California sought to add some clarity to the research already out there.
They looked at an even-gendered split of 74 participants who drank 300 mL of drinks sweetened with NNS, table sugar, or water. The researchers then used functional MRI to see how parts of the brain responsible for appetite and cravings responded to images of high-calorie foods. They also looked at glucose, insulin, and other metabolic hormone levels, as well as how much food the participants ate at their free buffet. (In the participants’ defense, who can say no to a free buffet?)
The researchers made some interesting observations:
- Women who drank the NNS drink ate more than did the table-sugar group, but all men ate the same.
- Images of those calorie-packed goodies increased cravings and appetite for obese men and women in the NNS group, compared with the table-sugar group.
- For all participants who drank the NNS drink, there was a decrease in the hormone that tells the body it’s full.
“By studying different groups we were able to show that females and people with obesity may be more sensitive to artificial sweeteners. For these groups, drinking artificially sweetened drinks may trick the brain into feeling hungry, which may in turn result in more calories being consumed,” Kathleen Page, MD, the study’s corresponding author, said in a separate statement.
Today’s lesson? Don’t believe every label you read.
Instagram vegetables and the triumph of peer pressure
You and your family are sitting down for dinner. You’ve taken the time to prepare a healthy, nutritious meal. Vegetables, rice, seafood – all the right things. But the children around you refuse to partake. What can you do? Why, show them a highly liked photo of broccoli on Instagram!
In reality, kids will probably never like to eat their vegetables, but according to a study published in Appetite, viewing highly liked images on social media can compel adults to eat theirs.
The investigators recruited a group of 169 adults aged 18-28 (average age, 21) and showed them a series of mock Instagram posts of all sorts of food, everything from Brussels sprouts to chocolate cake, as well as nonfood images to act as a baseline. The images had a varying amount of likes. After viewing the images, study participants were offered a snack buffet consisting of grapes and cookies.
The results were a triumph of peer pressure. Those who viewed highly liked images of nutritious foods ate a significantly larger proportion of grapes, compared with those who saw highly liked images of unhealthy food or nonfood.
The authors cautioned that more research is needed, but they said that they’re onto something in the eternal struggle of getting people to eat better. If Mikey liked it, maybe you should, too. Just as long as you don’t try to encourage the eating of peas. That is a dark road none should take, and no one should ever be subjected to that cursed food.
It’s nice to share … hypertension?
You may have heard that, over time, you begin to resemble your spouse. You may have also heard that, as time goes by, your pet might start to resemble you, but that is a story for another time.
A lot of the time, it’s human nature that people partner with someone who is similar to them in physical and environmental status. If you like to go jogging at 5 a.m., you might want a spouse who does the same. A study done using data from couples in Japan and the Netherlands found that couples who had the same lifestyle had similar levels of blood pressure, cholesterol, and triglycerides. They also had similar illnesses such as hypertension and diabetes.
It’s important to note that many of the couples were not very genetically similar but had similar lifestyles. Encourage your partner to have a healthier lifestyle, so you can live on for many years to come!
Novel food for thought
When you were growing up, your parents probably told you to brush your teeth before you went to bed, warned you not to run with the scissors or play with matches, and punished you whenever you used the neighbor children to play Schrödinger’s cat.
They did those things for your own good, of course, and now the nation’s mother – the Centers for Disease Control and Prevention – is doing the same by warning us about novel outbreak–associated foods. As in, “Put down that novel outbreak–associated food! You don’t know where it’s been!”
Seriously, you don’t know where it’s been. CDC investigators identified 28 novel foods that were linked to 36 foodborne-disease outbreaks that occurred during 2007-2016, including moringa leaf (herb/spice), tempeh (grain), frog, sprouted nut butter, and skate.
The novel foods implicated in these outbreaks were more likely to be imported, compared with 14,216 outbreaks that occurred from 1973 to 2016, and about half didn’t require refrigeration. Two-thirds did not need to be cooked after purchase. Another thing your parents wouldn’t like: Some can’t be washed, like sheep milk, sugar cane, or the aforementioned nut butter.
We wanted to get a food expert to comment on these novel foods, but our editor said that the assistant manager of our local Burger King wasn’t expert enough, so we’ve commandeered someone else’s expert. Cynthia Sears, MD, of Johns Hopkins University in Baltimore, told Today.com all about the dangers of frogs: “Essentially all amphibians are contaminated, often with salmonella. Eating any amphibian that is not thoroughly cooked is a risk.”
Be sure to cook your amphibians before you eat them. Advice that your parents would be proud to share.
Dieters should stay away from diet drinks
When a drink is labeled “diet” many assume that the calorie-free beverage is the best choice. However, one of the largest studies to date on artificial sweeteners is out to set the record straight.
Artificial sweeteners, or nonnutritive sweeteners (NNS), are used in most if not all diet products to give the illusion of sweetness without the caloric guilt. Some studies say they help with weight loss for that very reason, but others say they can contribute to weight gain. So which is it?
Researchers at the University of Southern California sought to add some clarity to the research already out there.
They looked at an even-gendered split of 74 participants who drank 300 mL of drinks sweetened with NNS, table sugar, or water. The researchers then used functional MRI to see how parts of the brain responsible for appetite and cravings responded to images of high-calorie foods. They also looked at glucose, insulin, and other metabolic hormone levels, as well as how much food the participants ate at their free buffet. (In the participants’ defense, who can say no to a free buffet?)
The researchers made some interesting observations:
- Women who drank the NNS drink ate more than did the table-sugar group, but all men ate the same.
- Images of those calorie-packed goodies increased cravings and appetite for obese men and women in the NNS group, compared with the table-sugar group.
- For all participants who drank the NNS drink, there was a decrease in the hormone that tells the body it’s full.
“By studying different groups we were able to show that females and people with obesity may be more sensitive to artificial sweeteners. For these groups, drinking artificially sweetened drinks may trick the brain into feeling hungry, which may in turn result in more calories being consumed,” Kathleen Page, MD, the study’s corresponding author, said in a separate statement.
Today’s lesson? Don’t believe every label you read.
Instagram vegetables and the triumph of peer pressure
You and your family are sitting down for dinner. You’ve taken the time to prepare a healthy, nutritious meal. Vegetables, rice, seafood – all the right things. But the children around you refuse to partake. What can you do? Why, show them a highly liked photo of broccoli on Instagram!
In reality, kids will probably never like to eat their vegetables, but according to a study published in Appetite, viewing highly liked images on social media can compel adults to eat theirs.
The investigators recruited a group of 169 adults aged 18-28 (average age, 21) and showed them a series of mock Instagram posts of all sorts of food, everything from Brussels sprouts to chocolate cake, as well as nonfood images to act as a baseline. The images had a varying amount of likes. After viewing the images, study participants were offered a snack buffet consisting of grapes and cookies.
The results were a triumph of peer pressure. Those who viewed highly liked images of nutritious foods ate a significantly larger proportion of grapes, compared with those who saw highly liked images of unhealthy food or nonfood.
The authors cautioned that more research is needed, but they said that they’re onto something in the eternal struggle of getting people to eat better. If Mikey liked it, maybe you should, too. Just as long as you don’t try to encourage the eating of peas. That is a dark road none should take, and no one should ever be subjected to that cursed food.
It’s nice to share … hypertension?
You may have heard that, over time, you begin to resemble your spouse. You may have also heard that, as time goes by, your pet might start to resemble you, but that is a story for another time.
A lot of the time, it’s human nature that people partner with someone who is similar to them in physical and environmental status. If you like to go jogging at 5 a.m., you might want a spouse who does the same. A study done using data from couples in Japan and the Netherlands found that couples who had the same lifestyle had similar levels of blood pressure, cholesterol, and triglycerides. They also had similar illnesses such as hypertension and diabetes.
It’s important to note that many of the couples were not very genetically similar but had similar lifestyles. Encourage your partner to have a healthier lifestyle, so you can live on for many years to come!
Novel food for thought
When you were growing up, your parents probably told you to brush your teeth before you went to bed, warned you not to run with the scissors or play with matches, and punished you whenever you used the neighbor children to play Schrödinger’s cat.
They did those things for your own good, of course, and now the nation’s mother – the Centers for Disease Control and Prevention – is doing the same by warning us about novel outbreak–associated foods. As in, “Put down that novel outbreak–associated food! You don’t know where it’s been!”
Seriously, you don’t know where it’s been. CDC investigators identified 28 novel foods that were linked to 36 foodborne-disease outbreaks that occurred during 2007-2016, including moringa leaf (herb/spice), tempeh (grain), frog, sprouted nut butter, and skate.
The novel foods implicated in these outbreaks were more likely to be imported, compared with 14,216 outbreaks that occurred from 1973 to 2016, and about half didn’t require refrigeration. Two-thirds did not need to be cooked after purchase. Another thing your parents wouldn’t like: Some can’t be washed, like sheep milk, sugar cane, or the aforementioned nut butter.
We wanted to get a food expert to comment on these novel foods, but our editor said that the assistant manager of our local Burger King wasn’t expert enough, so we’ve commandeered someone else’s expert. Cynthia Sears, MD, of Johns Hopkins University in Baltimore, told Today.com all about the dangers of frogs: “Essentially all amphibians are contaminated, often with salmonella. Eating any amphibian that is not thoroughly cooked is a risk.”
Be sure to cook your amphibians before you eat them. Advice that your parents would be proud to share.
Dieters should stay away from diet drinks
When a drink is labeled “diet” many assume that the calorie-free beverage is the best choice. However, one of the largest studies to date on artificial sweeteners is out to set the record straight.
Artificial sweeteners, or nonnutritive sweeteners (NNS), are used in most if not all diet products to give the illusion of sweetness without the caloric guilt. Some studies say they help with weight loss for that very reason, but others say they can contribute to weight gain. So which is it?
Researchers at the University of Southern California sought to add some clarity to the research already out there.
They looked at an even-gendered split of 74 participants who drank 300 mL of drinks sweetened with NNS, table sugar, or water. The researchers then used functional MRI to see how parts of the brain responsible for appetite and cravings responded to images of high-calorie foods. They also looked at glucose, insulin, and other metabolic hormone levels, as well as how much food the participants ate at their free buffet. (In the participants’ defense, who can say no to a free buffet?)
The researchers made some interesting observations:
- Women who drank the NNS drink ate more than did the table-sugar group, but all men ate the same.
- Images of those calorie-packed goodies increased cravings and appetite for obese men and women in the NNS group, compared with the table-sugar group.
- For all participants who drank the NNS drink, there was a decrease in the hormone that tells the body it’s full.
“By studying different groups we were able to show that females and people with obesity may be more sensitive to artificial sweeteners. For these groups, drinking artificially sweetened drinks may trick the brain into feeling hungry, which may in turn result in more calories being consumed,” Kathleen Page, MD, the study’s corresponding author, said in a separate statement.
Today’s lesson? Don’t believe every label you read.
Instagram vegetables and the triumph of peer pressure
You and your family are sitting down for dinner. You’ve taken the time to prepare a healthy, nutritious meal. Vegetables, rice, seafood – all the right things. But the children around you refuse to partake. What can you do? Why, show them a highly liked photo of broccoli on Instagram!
In reality, kids will probably never like to eat their vegetables, but according to a study published in Appetite, viewing highly liked images on social media can compel adults to eat theirs.
The investigators recruited a group of 169 adults aged 18-28 (average age, 21) and showed them a series of mock Instagram posts of all sorts of food, everything from Brussels sprouts to chocolate cake, as well as nonfood images to act as a baseline. The images had a varying amount of likes. After viewing the images, study participants were offered a snack buffet consisting of grapes and cookies.
The results were a triumph of peer pressure. Those who viewed highly liked images of nutritious foods ate a significantly larger proportion of grapes, compared with those who saw highly liked images of unhealthy food or nonfood.
The authors cautioned that more research is needed, but they said that they’re onto something in the eternal struggle of getting people to eat better. If Mikey liked it, maybe you should, too. Just as long as you don’t try to encourage the eating of peas. That is a dark road none should take, and no one should ever be subjected to that cursed food.
It’s nice to share … hypertension?
You may have heard that, over time, you begin to resemble your spouse. You may have also heard that, as time goes by, your pet might start to resemble you, but that is a story for another time.
A lot of the time, it’s human nature that people partner with someone who is similar to them in physical and environmental status. If you like to go jogging at 5 a.m., you might want a spouse who does the same. A study done using data from couples in Japan and the Netherlands found that couples who had the same lifestyle had similar levels of blood pressure, cholesterol, and triglycerides. They also had similar illnesses such as hypertension and diabetes.
It’s important to note that many of the couples were not very genetically similar but had similar lifestyles. Encourage your partner to have a healthier lifestyle, so you can live on for many years to come!
Greater portal use gives patients access, doctors headaches
The use of patient portals that provide access to electronic health records has dramatically increased in the past several years, and patients whose health care practitioner encouraged them to use their online portal accessed them at a higher rate than those who were not encouraged to do so.
These were among the top-line results of a national survey of U.S. adults conducted by the National Institutes of Health from January 2020 to April 2020. Although the COVID-19 pandemic hit the United States in the middle of that period, a report on the survey by the Office of the National Coordinator for Health IT stated, “These findings largely reflect prepandemic rates of individuals being offered and subsequently using their online medical record, also known as a patient portal.”
But with more patient access can come additional work for physicians and other health care practitioners, ranging from an onslaught of patient communications to managing data sent to them by patients.
According to the report, 59% of individuals were offered access to their patient portal, and 38% accessed their record at least once in 2020. By comparison, in 2014, just 42% were offered access to their portal, and 25% used it. But these percentages hardly changed from 2019 to 2020.
The increase in the percentage of people who accessed portals reflects the fact that more people were offered access. In addition, there were signs of rising activity among portal users.
Among patients offered access to their patient portal, 64% accessed it at least once in 2020 – 11 percentage points more than in 2017. Twenty-seven percent of those who had access to a portal used it once or twice; 20% accessed it three to five times; and 18% used it six or more times. The latter two percentages were significantly higher than in 2017.
Of the respondents who were offered access to portals but didn’t use them, 69% said they didn’t access the portal because they preferred to speak with their health care practitioner directly. Sixty-three percent said they didn’t see a need to use their online medical record. This was similar to the percentage 3 years earlier. Other reasons included respondents’ concerns about the privacy/security of online medical records (24%), their lack of comfort with computers (20%), and their lack of Internet access (13%).
The pros and cons of patient portals, greater access
Among portal users who accessed their records through a mobile health app, 51% used the app to facilitate discussions with their health care practitioner in 2020, an 8–percentage point increase from 2017. Fifty-percent of the mobile health app users utilized it to make a decision about how to treat an illness or condition, up from 45% in 2017. And 71% of these individuals used their app to track progress on a health-related goal, just a bit more than in 2017.
Individuals who were encouraged by their health care practitioner to use their patient portal viewed clinical notes and exchanged secure messages with their practitioner at higher rates than those who had not been encouraged. This is not surprising, but it reflects an unintended result of patient portals that many physicians have found burdensome, especially during the pandemic: overflowing electronic in-boxes.
Robert Wachter, MD, chairman of the department of medicine at the University of California, San Francisco, recently tweeted, “We’re seeing huge uptick in in-box messages for MDs during COVID – now seems like biggest driver of MD burnout. The fundamental problem: We turned on 24/7/365 access for patients (who of course like it) with no operational or business model to handle it. Crucial that we fix this.”
Steven Waldren, MD, vice president and chief medical informatics officer at the American Academy of Family Physicians, told this news organization that he agrees that this is a major challenge. “In-box management is a burden on physicians and practices,” he said. “However, it can be done better, either through a team in-box or through better use of technology.”
The team in-box he refers to is a mechanism for triaging patient messages. For example, a triage nurse can look at the messages and decide which ones can be handled by staff and which ones the doctor needs to see. Or physicians and front office staff can see the messages at the same time; a nurse can triage some messages according to protocols, and the physician can respond to any message, depending on what he or she knows about the patient.
Technology can also be enlisted in the effort, he suggested, perhaps by automating the triaging of messages such as prescription refill requests or using artificial intelligence to sort messages by content.
Making patient records portable
Nearly 40% of portal users accessed it using a smartphone app (17%) or with both their smartphone app and their computer (22%). Sixty-one percent of users relied exclusively on computers to access their portals.
About a third of patient portal users downloaded their online medical records in 2020. This proportion has nearly doubled from 17% since 2017, the ONC report noted.
Although the survey didn’t ask about multiple downloads, it appears that most people had to download their records separately from the patient portal of each practitioner who cared for them. Although the Apple Health app allows people to download records to their iPhones from multiple portals using a standard application programming interface, the ONC report says that only 5% of respondents transmitted their records to a service or app, up slightly from 3% in 2017.
Dr. Waldren hopes most patients will have the ability to download and integrate records from multiple practitioners in a few years, but he wouldn’t bet on it.
“A fair amount of work needs to be done on the business side and on figuring out how the data get connected together,” he said. “And there are still privacy concerns with apps.”
Overall, 21% of portal users transmitted their data to at least one outside party in 2020, compared with 14% in 2017. Seventeen percent of them sent their records to another health care practitioner, up from 10% in 2017. Five percent of the users transmitted their records to a caregiver, slightly more than in 2017.
Managing data is a challenge
Asked how physicians feel about portal users adding information to their record or correcting inaccurate information, Dr. Waldren says, “Doctors are already comfortable with patient-generated data. The challenge is managing it. If the patient provides data that’s not easy to put in the EHR, that’s going to add work, and they don’t want to see 100 blood pressure readings.
“You’d be hard-pressed to find a doctor who doesn’t welcome additional information about the patient’s health, but it can be onerous and can take time to enter the data,” Dr. Waldren said.
Overall, he said, “Giving patients the ability to take more ownership of their health and participate in their own care is good and can help us move forward. How this will be integrated into patient care is another question.”
A version of this article first appeared on Medscape.com.
The use of patient portals that provide access to electronic health records has dramatically increased in the past several years, and patients whose health care practitioner encouraged them to use their online portal accessed them at a higher rate than those who were not encouraged to do so.
These were among the top-line results of a national survey of U.S. adults conducted by the National Institutes of Health from January 2020 to April 2020. Although the COVID-19 pandemic hit the United States in the middle of that period, a report on the survey by the Office of the National Coordinator for Health IT stated, “These findings largely reflect prepandemic rates of individuals being offered and subsequently using their online medical record, also known as a patient portal.”
But with more patient access can come additional work for physicians and other health care practitioners, ranging from an onslaught of patient communications to managing data sent to them by patients.
According to the report, 59% of individuals were offered access to their patient portal, and 38% accessed their record at least once in 2020. By comparison, in 2014, just 42% were offered access to their portal, and 25% used it. But these percentages hardly changed from 2019 to 2020.
The increase in the percentage of people who accessed portals reflects the fact that more people were offered access. In addition, there were signs of rising activity among portal users.
Among patients offered access to their patient portal, 64% accessed it at least once in 2020 – 11 percentage points more than in 2017. Twenty-seven percent of those who had access to a portal used it once or twice; 20% accessed it three to five times; and 18% used it six or more times. The latter two percentages were significantly higher than in 2017.
Of the respondents who were offered access to portals but didn’t use them, 69% said they didn’t access the portal because they preferred to speak with their health care practitioner directly. Sixty-three percent said they didn’t see a need to use their online medical record. This was similar to the percentage 3 years earlier. Other reasons included respondents’ concerns about the privacy/security of online medical records (24%), their lack of comfort with computers (20%), and their lack of Internet access (13%).
The pros and cons of patient portals, greater access
Among portal users who accessed their records through a mobile health app, 51% used the app to facilitate discussions with their health care practitioner in 2020, an 8–percentage point increase from 2017. Fifty-percent of the mobile health app users utilized it to make a decision about how to treat an illness or condition, up from 45% in 2017. And 71% of these individuals used their app to track progress on a health-related goal, just a bit more than in 2017.
Individuals who were encouraged by their health care practitioner to use their patient portal viewed clinical notes and exchanged secure messages with their practitioner at higher rates than those who had not been encouraged. This is not surprising, but it reflects an unintended result of patient portals that many physicians have found burdensome, especially during the pandemic: overflowing electronic in-boxes.
Robert Wachter, MD, chairman of the department of medicine at the University of California, San Francisco, recently tweeted, “We’re seeing huge uptick in in-box messages for MDs during COVID – now seems like biggest driver of MD burnout. The fundamental problem: We turned on 24/7/365 access for patients (who of course like it) with no operational or business model to handle it. Crucial that we fix this.”
Steven Waldren, MD, vice president and chief medical informatics officer at the American Academy of Family Physicians, told this news organization that he agrees that this is a major challenge. “In-box management is a burden on physicians and practices,” he said. “However, it can be done better, either through a team in-box or through better use of technology.”
The team in-box he refers to is a mechanism for triaging patient messages. For example, a triage nurse can look at the messages and decide which ones can be handled by staff and which ones the doctor needs to see. Or physicians and front office staff can see the messages at the same time; a nurse can triage some messages according to protocols, and the physician can respond to any message, depending on what he or she knows about the patient.
Technology can also be enlisted in the effort, he suggested, perhaps by automating the triaging of messages such as prescription refill requests or using artificial intelligence to sort messages by content.
Making patient records portable
Nearly 40% of portal users accessed it using a smartphone app (17%) or with both their smartphone app and their computer (22%). Sixty-one percent of users relied exclusively on computers to access their portals.
About a third of patient portal users downloaded their online medical records in 2020. This proportion has nearly doubled from 17% since 2017, the ONC report noted.
Although the survey didn’t ask about multiple downloads, it appears that most people had to download their records separately from the patient portal of each practitioner who cared for them. Although the Apple Health app allows people to download records to their iPhones from multiple portals using a standard application programming interface, the ONC report says that only 5% of respondents transmitted their records to a service or app, up slightly from 3% in 2017.
Dr. Waldren hopes most patients will have the ability to download and integrate records from multiple practitioners in a few years, but he wouldn’t bet on it.
“A fair amount of work needs to be done on the business side and on figuring out how the data get connected together,” he said. “And there are still privacy concerns with apps.”
Overall, 21% of portal users transmitted their data to at least one outside party in 2020, compared with 14% in 2017. Seventeen percent of them sent their records to another health care practitioner, up from 10% in 2017. Five percent of the users transmitted their records to a caregiver, slightly more than in 2017.
Managing data is a challenge
Asked how physicians feel about portal users adding information to their record or correcting inaccurate information, Dr. Waldren says, “Doctors are already comfortable with patient-generated data. The challenge is managing it. If the patient provides data that’s not easy to put in the EHR, that’s going to add work, and they don’t want to see 100 blood pressure readings.
“You’d be hard-pressed to find a doctor who doesn’t welcome additional information about the patient’s health, but it can be onerous and can take time to enter the data,” Dr. Waldren said.
Overall, he said, “Giving patients the ability to take more ownership of their health and participate in their own care is good and can help us move forward. How this will be integrated into patient care is another question.”
A version of this article first appeared on Medscape.com.
The use of patient portals that provide access to electronic health records has dramatically increased in the past several years, and patients whose health care practitioner encouraged them to use their online portal accessed them at a higher rate than those who were not encouraged to do so.
These were among the top-line results of a national survey of U.S. adults conducted by the National Institutes of Health from January 2020 to April 2020. Although the COVID-19 pandemic hit the United States in the middle of that period, a report on the survey by the Office of the National Coordinator for Health IT stated, “These findings largely reflect prepandemic rates of individuals being offered and subsequently using their online medical record, also known as a patient portal.”
But with more patient access can come additional work for physicians and other health care practitioners, ranging from an onslaught of patient communications to managing data sent to them by patients.
According to the report, 59% of individuals were offered access to their patient portal, and 38% accessed their record at least once in 2020. By comparison, in 2014, just 42% were offered access to their portal, and 25% used it. But these percentages hardly changed from 2019 to 2020.
The increase in the percentage of people who accessed portals reflects the fact that more people were offered access. In addition, there were signs of rising activity among portal users.
Among patients offered access to their patient portal, 64% accessed it at least once in 2020 – 11 percentage points more than in 2017. Twenty-seven percent of those who had access to a portal used it once or twice; 20% accessed it three to five times; and 18% used it six or more times. The latter two percentages were significantly higher than in 2017.
Of the respondents who were offered access to portals but didn’t use them, 69% said they didn’t access the portal because they preferred to speak with their health care practitioner directly. Sixty-three percent said they didn’t see a need to use their online medical record. This was similar to the percentage 3 years earlier. Other reasons included respondents’ concerns about the privacy/security of online medical records (24%), their lack of comfort with computers (20%), and their lack of Internet access (13%).
The pros and cons of patient portals, greater access
Among portal users who accessed their records through a mobile health app, 51% used the app to facilitate discussions with their health care practitioner in 2020, an 8–percentage point increase from 2017. Fifty-percent of the mobile health app users utilized it to make a decision about how to treat an illness or condition, up from 45% in 2017. And 71% of these individuals used their app to track progress on a health-related goal, just a bit more than in 2017.
Individuals who were encouraged by their health care practitioner to use their patient portal viewed clinical notes and exchanged secure messages with their practitioner at higher rates than those who had not been encouraged. This is not surprising, but it reflects an unintended result of patient portals that many physicians have found burdensome, especially during the pandemic: overflowing electronic in-boxes.
Robert Wachter, MD, chairman of the department of medicine at the University of California, San Francisco, recently tweeted, “We’re seeing huge uptick in in-box messages for MDs during COVID – now seems like biggest driver of MD burnout. The fundamental problem: We turned on 24/7/365 access for patients (who of course like it) with no operational or business model to handle it. Crucial that we fix this.”
Steven Waldren, MD, vice president and chief medical informatics officer at the American Academy of Family Physicians, told this news organization that he agrees that this is a major challenge. “In-box management is a burden on physicians and practices,” he said. “However, it can be done better, either through a team in-box or through better use of technology.”
The team in-box he refers to is a mechanism for triaging patient messages. For example, a triage nurse can look at the messages and decide which ones can be handled by staff and which ones the doctor needs to see. Or physicians and front office staff can see the messages at the same time; a nurse can triage some messages according to protocols, and the physician can respond to any message, depending on what he or she knows about the patient.
Technology can also be enlisted in the effort, he suggested, perhaps by automating the triaging of messages such as prescription refill requests or using artificial intelligence to sort messages by content.
Making patient records portable
Nearly 40% of portal users accessed it using a smartphone app (17%) or with both their smartphone app and their computer (22%). Sixty-one percent of users relied exclusively on computers to access their portals.
About a third of patient portal users downloaded their online medical records in 2020. This proportion has nearly doubled from 17% since 2017, the ONC report noted.
Although the survey didn’t ask about multiple downloads, it appears that most people had to download their records separately from the patient portal of each practitioner who cared for them. Although the Apple Health app allows people to download records to their iPhones from multiple portals using a standard application programming interface, the ONC report says that only 5% of respondents transmitted their records to a service or app, up slightly from 3% in 2017.
Dr. Waldren hopes most patients will have the ability to download and integrate records from multiple practitioners in a few years, but he wouldn’t bet on it.
“A fair amount of work needs to be done on the business side and on figuring out how the data get connected together,” he said. “And there are still privacy concerns with apps.”
Overall, 21% of portal users transmitted their data to at least one outside party in 2020, compared with 14% in 2017. Seventeen percent of them sent their records to another health care practitioner, up from 10% in 2017. Five percent of the users transmitted their records to a caregiver, slightly more than in 2017.
Managing data is a challenge
Asked how physicians feel about portal users adding information to their record or correcting inaccurate information, Dr. Waldren says, “Doctors are already comfortable with patient-generated data. The challenge is managing it. If the patient provides data that’s not easy to put in the EHR, that’s going to add work, and they don’t want to see 100 blood pressure readings.
“You’d be hard-pressed to find a doctor who doesn’t welcome additional information about the patient’s health, but it can be onerous and can take time to enter the data,” Dr. Waldren said.
Overall, he said, “Giving patients the ability to take more ownership of their health and participate in their own care is good and can help us move forward. How this will be integrated into patient care is another question.”
A version of this article first appeared on Medscape.com.
FDA investigating potential sex differences in LAAO adverse outcomes
The agency said it is evaluating a real-world study, published in JAMA Cardiology, of 49,357 patients in the National Cardiovascular Data Registry LAAO Registry that suggested women might be at greater risk than men for procedural outcomes, including major adverse events after device implant.
“The FDA recognizes the limitations of these data, including that the study was not randomized, only included one LAAO device (the first-generation Watchman device), and did not include longer-term outcomes beyond in-hospital events. However, the analysis provides results from a large registry of patients treated with LAAO implants in the U.S.,” the agency said in its letter to health care providers Sept. 27.
As reported last month, the study by Darden et al showed a significantly higher rate of adverse procedural events in women than in men, including any adverse events (6.3% vs. 3.9%; P < .001), any major adverse events (4.1% vs. 2.0%; P < .001), and hospital stay longer than 1 day (16.0% vs. 11.6%; P < .001). Procedure-associated death was 0.3% in women and 0.1% in men.
The agency noted that the number of patients in the LAAO Registry analysis was much larger than the number of patients in the premarket studies of the Watchman device that supported its approval and in the premarket studies for the other approved LAAO devices.
LAAO devices are indicated to reduce the risk for thromboembolism from the left atrial appendage in patients with nonvalvular atrial fibrillation who are not good candidates for long-term anticoagulation. LAAO devices currently marketed in the United States are Boston Scientific’s Watchman and Watchman FLX devices and Abbott Medical’s Amplatzer Amulet device.
“Currently, the FDA believes the benefits continue to outweigh the risks for approved LAAO devices when used in accordance with their instructions for use,” the letter states.
The FDA will work with device manufacturers to evaluate the potential issue, including a review of available premarket and postapproval study data and other available real-world, postmarket datasets, the letter notes.
The agency will also work with device manufacturers, investigators, and the LAAO Registry to try to identify the causes of procedural outcome differences between women and men.
In the letter, the FDA recommends health care providers continue monitoring patients who have been treated with LAAO devices in accordance with the current standard of care. They should also discuss the risks and benefits of all available options for stroke prevention in patients with atrial fibrillation as part of shared clinical decision-making.
Any adverse events or suspected adverse events experienced by patients with LAAO devices should also be reported to the FDA through MedWatch, its adverse event reporting program.
A version of this article first appeared on Medscape.com.
The agency said it is evaluating a real-world study, published in JAMA Cardiology, of 49,357 patients in the National Cardiovascular Data Registry LAAO Registry that suggested women might be at greater risk than men for procedural outcomes, including major adverse events after device implant.
“The FDA recognizes the limitations of these data, including that the study was not randomized, only included one LAAO device (the first-generation Watchman device), and did not include longer-term outcomes beyond in-hospital events. However, the analysis provides results from a large registry of patients treated with LAAO implants in the U.S.,” the agency said in its letter to health care providers Sept. 27.
As reported last month, the study by Darden et al showed a significantly higher rate of adverse procedural events in women than in men, including any adverse events (6.3% vs. 3.9%; P < .001), any major adverse events (4.1% vs. 2.0%; P < .001), and hospital stay longer than 1 day (16.0% vs. 11.6%; P < .001). Procedure-associated death was 0.3% in women and 0.1% in men.
The agency noted that the number of patients in the LAAO Registry analysis was much larger than the number of patients in the premarket studies of the Watchman device that supported its approval and in the premarket studies for the other approved LAAO devices.
LAAO devices are indicated to reduce the risk for thromboembolism from the left atrial appendage in patients with nonvalvular atrial fibrillation who are not good candidates for long-term anticoagulation. LAAO devices currently marketed in the United States are Boston Scientific’s Watchman and Watchman FLX devices and Abbott Medical’s Amplatzer Amulet device.
“Currently, the FDA believes the benefits continue to outweigh the risks for approved LAAO devices when used in accordance with their instructions for use,” the letter states.
The FDA will work with device manufacturers to evaluate the potential issue, including a review of available premarket and postapproval study data and other available real-world, postmarket datasets, the letter notes.
The agency will also work with device manufacturers, investigators, and the LAAO Registry to try to identify the causes of procedural outcome differences between women and men.
In the letter, the FDA recommends health care providers continue monitoring patients who have been treated with LAAO devices in accordance with the current standard of care. They should also discuss the risks and benefits of all available options for stroke prevention in patients with atrial fibrillation as part of shared clinical decision-making.
Any adverse events or suspected adverse events experienced by patients with LAAO devices should also be reported to the FDA through MedWatch, its adverse event reporting program.
A version of this article first appeared on Medscape.com.
The agency said it is evaluating a real-world study, published in JAMA Cardiology, of 49,357 patients in the National Cardiovascular Data Registry LAAO Registry that suggested women might be at greater risk than men for procedural outcomes, including major adverse events after device implant.
“The FDA recognizes the limitations of these data, including that the study was not randomized, only included one LAAO device (the first-generation Watchman device), and did not include longer-term outcomes beyond in-hospital events. However, the analysis provides results from a large registry of patients treated with LAAO implants in the U.S.,” the agency said in its letter to health care providers Sept. 27.
As reported last month, the study by Darden et al showed a significantly higher rate of adverse procedural events in women than in men, including any adverse events (6.3% vs. 3.9%; P < .001), any major adverse events (4.1% vs. 2.0%; P < .001), and hospital stay longer than 1 day (16.0% vs. 11.6%; P < .001). Procedure-associated death was 0.3% in women and 0.1% in men.
The agency noted that the number of patients in the LAAO Registry analysis was much larger than the number of patients in the premarket studies of the Watchman device that supported its approval and in the premarket studies for the other approved LAAO devices.
LAAO devices are indicated to reduce the risk for thromboembolism from the left atrial appendage in patients with nonvalvular atrial fibrillation who are not good candidates for long-term anticoagulation. LAAO devices currently marketed in the United States are Boston Scientific’s Watchman and Watchman FLX devices and Abbott Medical’s Amplatzer Amulet device.
“Currently, the FDA believes the benefits continue to outweigh the risks for approved LAAO devices when used in accordance with their instructions for use,” the letter states.
The FDA will work with device manufacturers to evaluate the potential issue, including a review of available premarket and postapproval study data and other available real-world, postmarket datasets, the letter notes.
The agency will also work with device manufacturers, investigators, and the LAAO Registry to try to identify the causes of procedural outcome differences between women and men.
In the letter, the FDA recommends health care providers continue monitoring patients who have been treated with LAAO devices in accordance with the current standard of care. They should also discuss the risks and benefits of all available options for stroke prevention in patients with atrial fibrillation as part of shared clinical decision-making.
Any adverse events or suspected adverse events experienced by patients with LAAO devices should also be reported to the FDA through MedWatch, its adverse event reporting program.
A version of this article first appeared on Medscape.com.