User login
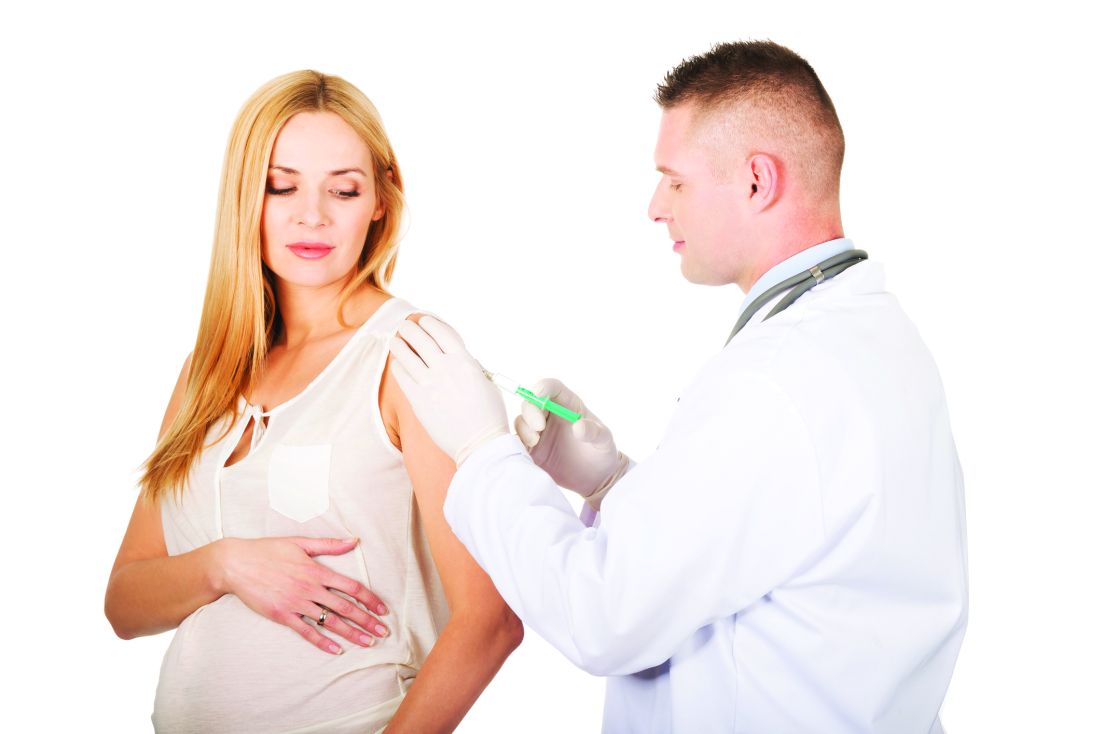
The combined tetanus, diphtheria, and acellular pertussis (Tdap) vaccine is not associated with an increased risk of microcephaly and other structural birth defects when administered during pregnancy, according to findings from a large, retrospective cohort study.
The U.S. Advisory Committee on Immunization Practices currently recommends administration of the Tdap vaccine between 27 and 36 weeks’ gestation in every pregnancy. However, the overlap of the start of Brazil’s maternal Tdap immunization in November 2014 with the substantial increase in microcephaly cases in 2015 prompted concerns of an association between the vaccine and structural birth defects.
They found that Tdap immunization was not significantly associated with an increased risk for microcephaly during any week of pregnancy (adjusted prevalence ratio, 0.86; 95% CI, 0.60-1.24). They also saw no increased risk of microcephaly when vaccinations occurred before 14 weeks’ gestation (adjusted prevalence ratio, 0.96; 95% CI, 0.36-2.58), or when vaccinations were administered between 27 weeks’ and 36 weeks’ gestation (adjusted prevalence ratio, 1.01; 95% CI, 0.63-1.61). The findings were similar for other structural defects, including congenital heart defects, spina bifida, encephalocele, and anophthalmia (JAMA. 2016;316[17]:1823-5).
“These results expand upon what is known about maternal Tdap vaccination safety to include information about structural birth defects and microcephaly in offspring,” the investigators wrote. “The findings support recommendations for routine Tdap administration during pregnancy.”
However, they noted that the study findings may have been limited by incomplete data on women’s immunization status, birth defects, and defects that may have resulted in pregnancy loss or elective termination.
The study was funded by the Centers for Disease Control and Prevention. The investigators reported having no relevant financial disclosures.
The combined tetanus, diphtheria, and acellular pertussis (Tdap) vaccine is not associated with an increased risk of microcephaly and other structural birth defects when administered during pregnancy, according to findings from a large, retrospective cohort study.
The U.S. Advisory Committee on Immunization Practices currently recommends administration of the Tdap vaccine between 27 and 36 weeks’ gestation in every pregnancy. However, the overlap of the start of Brazil’s maternal Tdap immunization in November 2014 with the substantial increase in microcephaly cases in 2015 prompted concerns of an association between the vaccine and structural birth defects.
They found that Tdap immunization was not significantly associated with an increased risk for microcephaly during any week of pregnancy (adjusted prevalence ratio, 0.86; 95% CI, 0.60-1.24). They also saw no increased risk of microcephaly when vaccinations occurred before 14 weeks’ gestation (adjusted prevalence ratio, 0.96; 95% CI, 0.36-2.58), or when vaccinations were administered between 27 weeks’ and 36 weeks’ gestation (adjusted prevalence ratio, 1.01; 95% CI, 0.63-1.61). The findings were similar for other structural defects, including congenital heart defects, spina bifida, encephalocele, and anophthalmia (JAMA. 2016;316[17]:1823-5).
“These results expand upon what is known about maternal Tdap vaccination safety to include information about structural birth defects and microcephaly in offspring,” the investigators wrote. “The findings support recommendations for routine Tdap administration during pregnancy.”
However, they noted that the study findings may have been limited by incomplete data on women’s immunization status, birth defects, and defects that may have resulted in pregnancy loss or elective termination.
The study was funded by the Centers for Disease Control and Prevention. The investigators reported having no relevant financial disclosures.
The combined tetanus, diphtheria, and acellular pertussis (Tdap) vaccine is not associated with an increased risk of microcephaly and other structural birth defects when administered during pregnancy, according to findings from a large, retrospective cohort study.
The U.S. Advisory Committee on Immunization Practices currently recommends administration of the Tdap vaccine between 27 and 36 weeks’ gestation in every pregnancy. However, the overlap of the start of Brazil’s maternal Tdap immunization in November 2014 with the substantial increase in microcephaly cases in 2015 prompted concerns of an association between the vaccine and structural birth defects.
They found that Tdap immunization was not significantly associated with an increased risk for microcephaly during any week of pregnancy (adjusted prevalence ratio, 0.86; 95% CI, 0.60-1.24). They also saw no increased risk of microcephaly when vaccinations occurred before 14 weeks’ gestation (adjusted prevalence ratio, 0.96; 95% CI, 0.36-2.58), or when vaccinations were administered between 27 weeks’ and 36 weeks’ gestation (adjusted prevalence ratio, 1.01; 95% CI, 0.63-1.61). The findings were similar for other structural defects, including congenital heart defects, spina bifida, encephalocele, and anophthalmia (JAMA. 2016;316[17]:1823-5).
“These results expand upon what is known about maternal Tdap vaccination safety to include information about structural birth defects and microcephaly in offspring,” the investigators wrote. “The findings support recommendations for routine Tdap administration during pregnancy.”
However, they noted that the study findings may have been limited by incomplete data on women’s immunization status, birth defects, and defects that may have resulted in pregnancy loss or elective termination.
The study was funded by the Centers for Disease Control and Prevention. The investigators reported having no relevant financial disclosures.
FROM JAMA
Key clinical point:
Major finding: Tdap immunization was not significantly associated with an increased risk for microcephaly during any week of pregnancy (adjusted prevalence ratio, 0.86; 95% CI, 0.60-1.24).
Data source: A retrospective cohort study in 41,654 singleton infants born to women who received Tdap during pregnancy and a control group of 282,809 babies born to unvaccinated women.
Disclosures: The study was funded by the Centers for Disease Control and Prevention. The investigators reported having no relevant financial disclosures.