User login
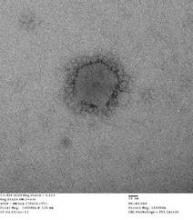
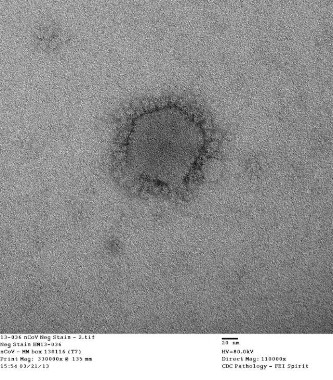
An Indiana man infected with Middle East Respiratory Syndrome Coronavirus had not transmitted the virus to a colleague in Illinois, as was previously suspected, Centers for Disease Control and Prevention officials said May 28.
On May 17, the CDC disclosed that the Illinois resident, who had attended an extended business meeting with the confirmed Indiana MERS-CoV patient prior to that patient’s hospitalization, had tested positive for MERS-CoV antibodies on serological assays. The case was particularly worrisome because it suggested transmission through a more casual level of contact than has been seen in most MERS cases to date.
However, CDC officials now say that additional testing with a neutralizing antibody assay, a more definitive blood test that takes 5 or more days to produce results, revealed that the Illinois man had not been infected with MERS-CoV.
Thus far only two U.S. residents have been confirmed with MERS-CoV, the Indiana patient and a Florida patient. Both became ill after travel to Saudi Arabia. None of their stateside contacts has yet tested positive, though voluntary testing of family members, colleagues, fellow passengers on flights, and attending health care workers continues.
In a news conference May 28, Mark Pallansch, Ph.D., director of CDC’s Division of Viral Diseases at the National Center for Immunization and Respiratory Diseases, acknowledged that the two serological assays used in preliminary testing – the enzyme-linked immunosorbent assay (ELISA) and the immunofluorescent assay (IFA), have high specificity in excluding infection, but can result in false positives.
In the case of the Illinois resident, "there are several potential explanations for our preliminary results – that is still something not completely understood," Dr. Pallansch said. The most common and likely explanation, he said, is some cross-reactivity with another type of coronavirus.
Dr. Pallansch said that CDC would continue its protocol of using ELISA and IFA, with any positive results followed by the neutralizing antibody assay.
The neutralizing antibody assay, he said, uses live MERS-CoV and must be conducted in a high-containment lab, making it difficult to test hundreds of samples; thus initial screening with the other assays would continue.
Various serology assays are being developed around the world, including by the CDC, that may supplant the assays currently being used in the initial serological detection of MERS-CoV, Dr. Pallansch reported.
Dr. David Swerdlow, who is leading CDC’s MERS-CoV response, said during the press conference that the CDC would continue to maintain its high level of vigilance, which includes disclosing to the public any initial positive results pending confirmation, and asking individuals who test positive on first assays to take significant measures to avoid contacts that could result in transmission.
Before definitive serological results were returned, the Illinois man was asked to wear a mask, to avoid close contact with other people, and to not come in contact with crowds. He was fully compliant with those requests, Dr. Swerdlow said.
"We can’t have all the tests back in order to take public health action," Dr. Swerdlow said, noting that MERS-CoV has been seen associated with a 30% fatality rate.
"It’s our priority to make sure the public is protected," he added. While there have been only two imported cases to date in the United States, and no infections yet found to have occurred as a result of local transmission, MERS-CoV "can and likely will enter our country again."
An Indiana man infected with Middle East Respiratory Syndrome Coronavirus had not transmitted the virus to a colleague in Illinois, as was previously suspected, Centers for Disease Control and Prevention officials said May 28.
On May 17, the CDC disclosed that the Illinois resident, who had attended an extended business meeting with the confirmed Indiana MERS-CoV patient prior to that patient’s hospitalization, had tested positive for MERS-CoV antibodies on serological assays. The case was particularly worrisome because it suggested transmission through a more casual level of contact than has been seen in most MERS cases to date.
However, CDC officials now say that additional testing with a neutralizing antibody assay, a more definitive blood test that takes 5 or more days to produce results, revealed that the Illinois man had not been infected with MERS-CoV.
Thus far only two U.S. residents have been confirmed with MERS-CoV, the Indiana patient and a Florida patient. Both became ill after travel to Saudi Arabia. None of their stateside contacts has yet tested positive, though voluntary testing of family members, colleagues, fellow passengers on flights, and attending health care workers continues.
In a news conference May 28, Mark Pallansch, Ph.D., director of CDC’s Division of Viral Diseases at the National Center for Immunization and Respiratory Diseases, acknowledged that the two serological assays used in preliminary testing – the enzyme-linked immunosorbent assay (ELISA) and the immunofluorescent assay (IFA), have high specificity in excluding infection, but can result in false positives.
In the case of the Illinois resident, "there are several potential explanations for our preliminary results – that is still something not completely understood," Dr. Pallansch said. The most common and likely explanation, he said, is some cross-reactivity with another type of coronavirus.
Dr. Pallansch said that CDC would continue its protocol of using ELISA and IFA, with any positive results followed by the neutralizing antibody assay.
The neutralizing antibody assay, he said, uses live MERS-CoV and must be conducted in a high-containment lab, making it difficult to test hundreds of samples; thus initial screening with the other assays would continue.
Various serology assays are being developed around the world, including by the CDC, that may supplant the assays currently being used in the initial serological detection of MERS-CoV, Dr. Pallansch reported.
Dr. David Swerdlow, who is leading CDC’s MERS-CoV response, said during the press conference that the CDC would continue to maintain its high level of vigilance, which includes disclosing to the public any initial positive results pending confirmation, and asking individuals who test positive on first assays to take significant measures to avoid contacts that could result in transmission.
Before definitive serological results were returned, the Illinois man was asked to wear a mask, to avoid close contact with other people, and to not come in contact with crowds. He was fully compliant with those requests, Dr. Swerdlow said.
"We can’t have all the tests back in order to take public health action," Dr. Swerdlow said, noting that MERS-CoV has been seen associated with a 30% fatality rate.
"It’s our priority to make sure the public is protected," he added. While there have been only two imported cases to date in the United States, and no infections yet found to have occurred as a result of local transmission, MERS-CoV "can and likely will enter our country again."
An Indiana man infected with Middle East Respiratory Syndrome Coronavirus had not transmitted the virus to a colleague in Illinois, as was previously suspected, Centers for Disease Control and Prevention officials said May 28.
On May 17, the CDC disclosed that the Illinois resident, who had attended an extended business meeting with the confirmed Indiana MERS-CoV patient prior to that patient’s hospitalization, had tested positive for MERS-CoV antibodies on serological assays. The case was particularly worrisome because it suggested transmission through a more casual level of contact than has been seen in most MERS cases to date.
However, CDC officials now say that additional testing with a neutralizing antibody assay, a more definitive blood test that takes 5 or more days to produce results, revealed that the Illinois man had not been infected with MERS-CoV.
Thus far only two U.S. residents have been confirmed with MERS-CoV, the Indiana patient and a Florida patient. Both became ill after travel to Saudi Arabia. None of their stateside contacts has yet tested positive, though voluntary testing of family members, colleagues, fellow passengers on flights, and attending health care workers continues.
In a news conference May 28, Mark Pallansch, Ph.D., director of CDC’s Division of Viral Diseases at the National Center for Immunization and Respiratory Diseases, acknowledged that the two serological assays used in preliminary testing – the enzyme-linked immunosorbent assay (ELISA) and the immunofluorescent assay (IFA), have high specificity in excluding infection, but can result in false positives.
In the case of the Illinois resident, "there are several potential explanations for our preliminary results – that is still something not completely understood," Dr. Pallansch said. The most common and likely explanation, he said, is some cross-reactivity with another type of coronavirus.
Dr. Pallansch said that CDC would continue its protocol of using ELISA and IFA, with any positive results followed by the neutralizing antibody assay.
The neutralizing antibody assay, he said, uses live MERS-CoV and must be conducted in a high-containment lab, making it difficult to test hundreds of samples; thus initial screening with the other assays would continue.
Various serology assays are being developed around the world, including by the CDC, that may supplant the assays currently being used in the initial serological detection of MERS-CoV, Dr. Pallansch reported.
Dr. David Swerdlow, who is leading CDC’s MERS-CoV response, said during the press conference that the CDC would continue to maintain its high level of vigilance, which includes disclosing to the public any initial positive results pending confirmation, and asking individuals who test positive on first assays to take significant measures to avoid contacts that could result in transmission.
Before definitive serological results were returned, the Illinois man was asked to wear a mask, to avoid close contact with other people, and to not come in contact with crowds. He was fully compliant with those requests, Dr. Swerdlow said.
"We can’t have all the tests back in order to take public health action," Dr. Swerdlow said, noting that MERS-CoV has been seen associated with a 30% fatality rate.
"It’s our priority to make sure the public is protected," he added. While there have been only two imported cases to date in the United States, and no infections yet found to have occurred as a result of local transmission, MERS-CoV "can and likely will enter our country again."