User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
House passes prior authorization bill, Senate path unclear
The path through the U.S. Senate is not yet certain for a bill intended to speed the prior authorization process of insurer-run Medicare Advantage plans, despite the measure having breezed through the House.
House leaders opted to move the Improving Seniors’ Timely Access to Care Act of 2021 (HR 3173) without requiring a roll-call vote. The measure was passed on Sept. 14 by a voice vote, an approach used in general with only uncontroversial measures that have broad support. The bill has 191 Democratic and 135 Republican sponsors, representing about three-quarters of the members of the House.
“There is no reason that patients should be waiting for medically appropriate care, especially when we know that this can lead to worse outcomes,” Rep. Earl Blumenauer (D-Ore.) said in a Sept. 14 speech on the House floor. “The fundamental promise of Medicare Advantage is undermined when people are delaying care, getting sicker, and ultimately costing Medicare more money.”
Rep. Greg Murphy, MD (R-N.C.), spoke on the House floor that day as well, bringing up cases he has seen in his own urology practice in which prior authorization delays disrupted medical care. One patient wound up in the hospital with abscess after an insurer denied an antibiotic prescription, Rep. Murphy said.
But the Senate appears unlikely at this time to move the prior authorization bill as a standalone measure. Instead, the bill may become part of a larger legislative package focused on health care that the Senate Finance Committee intends to prepare later this year.
The House-passed bill would require insurer-run Medicare plans to respond to expedited requests for prior authorization of services within 24 hours and to other requests within 7 days. This bill also would establish an electronic program for prior authorizations and mandate increased transparency as to how insurers use this tool.
CBO: Cost of change would be billions
In seeking to mandate changes in prior authorization, lawmakers likely will need to contend with the issue of a $16 billion cumulative cost estimate for the bill from the Congressional Budget Office. Members of Congress often seek to offset new spending by pairing bills that add to expected costs for the federal government with ones expected to produce savings.
Unlike Rep. Blumenauer, Rep. Murphy, and other backers of the prior authorization streamlining bill, CBO staff estimates that making the mandated changes would raise federal spending, inasmuch as there would be “a greater use of services.”
On Sept. 14, CBO issued a one-page report on the costs of the bill. The CBO report concerns only the bill in question, as is common practice with the office’s estimates.
Prior authorization changes would begin in fiscal 2025 and would add $899 million in spending, or outlays, that year, CBO said. The annual costs from the streamlined prior authorization practices through fiscal 2026 to 2032 range from $1.6 billion to $2.7 billion.
Looking at the CBO estimate against a backdrop of total Medicare Advantage costs, though, may provide important context.
The increases in spending estimated by CBO may suggest that there would be little change in federal spending as a result of streamlining prior authorization practices. These estimates of increased annual spending of $1.6 billion–$2.7 billion are only a small fraction of the current annual cost of insurer-run Medicare, and they represent an even smaller share of the projected expense.
The federal government last year spent about $350 billion on insurer-run plans, excluding Part D drug plan payments, according to the Medicare Advisory Payment Commission (MedPAC).
As of 2021, about 27 million people were enrolled in these plans, accounting for about 46% of the total Medicare population. Enrollment has doubled since 2010, MedPAC said, and it is expected to continue to grow. By 2027, insurer-run Medicare could cover 50% of the program’s population, a figure that may reach 53% by 2031.
Federal payments to these plans will accelerate in the years ahead as insurers attract more people eligible for Medicare as customers. Payments to these private health plans could rise from an expected $418 billion this year to $940.6 billion by 2031, according to the most recent Medicare trustees report.
Good intentions, poor implementation?
Insurer-run Medicare has long enjoyed deep bipartisan support in Congress. That’s due in part to its potential for reducing spending on what are considered low-value treatments, or ones considered unlikely to provide a significant medical benefit, but Rep. Blumenauer is among the members of Congress who see insurer-run Medicare as a path for preserving the giant federal health program. Traditional Medicare has far fewer restrictions on services, which sometimes opens a path for tests and treatments that offer less value for patients.
“I believe that the way traditional fee-for-service Medicare operates is not sustainable and that Medicare Advantage is one of the tools we can use to demonstrate how we can incentivize value,” Rep. Blumenauer said on the House floor. “But this is only possible when the program operates as intended. I have been deeply concerned about the reports of delays in care” caused by the clunky prior authorization processes.
He highlighted a recent report from the internal watchdog group for the Department of Health & Human Services that raises concerns about denials of appropriate care. About 18% of a set of payment denials examined by the Office of Inspector General of HHS in April actually met Medicare coverage rules and plan billing rules.
“For patients and their families, being told that you need to wait longer for care that your doctor tells you that you need is incredibly frustrating and frightening,” Rep. Blumenauer said. “There’s no comfort to be found in the fact that your insurance company needs time to decide if your doctor is right.”
Trends in prior authorization
The CBO report does not provide detail on what kind of medical spending would increase under a streamlined prior authorization process in insurer-run Medicare plans.
From trends reported in prior authorization, though, two factors could be at play in what appear to be relatively small estimated increases in Medicare spending from streamlined prior authorization.
One is the work already underway to create less burdensome electronic systems for these requests, such as the Fast Prior Authorization Technology Highway initiative run by the trade association America’s Health Insurance Plans.
The other factor could be the number of cases in which prior authorization merely causes delays in treatments and tests and thus simply postpones spending while adding to clinicians’ administrative work.
An analysis of prior authorization requests for dermatologic practices affiliated with the University of Utah may represent an extreme example. In a report published in JAMA Dermatology in 2020, researchers described what happened with requests made during 1 month, September 2016.
The approval rate for procedures was 99.6% – 100% (95 of 95) for Mohs surgery, and 96% (130 of 131, with 4 additional cases pending) for excisions. These findings supported calls for simplifying prior authorization procedures, “perhaps first by eliminating unnecessary PAs [prior authorizations] and appeals,” Aaron M. Secrest, MD, PhD, of the University of Utah, Salt Lake City, and coauthors wrote in the article.
Still, there is some evidence that insurer-run Medicare policies reduce the use of low-value care.
In a study published in JAMA Health Forum, Emily Boudreau, PhD, of insurer Humana Inc, and coauthors from Tufts University, Boston, and the University of Pennsylvania, Philadelphia investigated whether insurer-run Medicare could do a better job in reducing the amount of low-value care delivered than the traditional program. They analyzed a set of claims data from 2017 to 2019 for people enrolled in insurer-run and traditional Medicare.
They reported a rate of 23.07 low-value services provided per 100 people in insurer-run Medicare, compared with 25.39 for those in traditional Medicare. Some of the biggest differences reported in the article were in cancer screenings for older people.
As an example, the U.S. Preventive Services Task Force recommends that women older than 65 years not be screened for cervical cancer if they have undergone adequate screening in the past and are not at high risk for cervical cancer. There was an annual count of 1.76 screenings for cervical cancer per 100 women older than 65 in the insurer-run Medicare group versus 3.18 for those in traditional Medicare.
The Better Medicare Alliance issued a statement in favor of the House passage of the Improving Seniors’ Timely Access to Care Act.
In it, the group said the measure would “modernize prior authorization while protecting its essential function in facilitating safe, high-value, evidence-based care.” The alliance promotes use of insurer-run Medicare. The board of the Better Medicare Alliance includes executives who serve with firms that run Advantage plans as well as medical organizations and universities.
“With studies showing that up to one-quarter of all health care expenditures are wasted on services with no benefit to the patient, we need a robust, next-generation prior authorization program to deter low-value, and even harmful, care while protecting access to needed treatment and effective therapies,” said A. Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, in a statement issued by the Better Medicare Alliance. He is a member of the group’s council of scholars.
On the House floor on September 14, Rep. Ami Bera, MD (D-Calif.), said he has heard from former colleagues and his medical school classmates that they now spend as much as 40% of their time on administrative work. These distractions from patient care are helping drive physicians away from the practice of medicine.
Still, the internist defended the basic premise of prior authorization while strongly appealing for better systems of handling it.
“Yes, there is a role for prior authorization in limited cases. There is also a role to go back and retrospectively look at how care is being delivered,” Rep. Bera said. “But what is happening today is a travesty. It wasn’t the intention of prior authorization. It is a prior authorization process gone awry.”
A version of this article first appeared on Medscape.com.
The path through the U.S. Senate is not yet certain for a bill intended to speed the prior authorization process of insurer-run Medicare Advantage plans, despite the measure having breezed through the House.
House leaders opted to move the Improving Seniors’ Timely Access to Care Act of 2021 (HR 3173) without requiring a roll-call vote. The measure was passed on Sept. 14 by a voice vote, an approach used in general with only uncontroversial measures that have broad support. The bill has 191 Democratic and 135 Republican sponsors, representing about three-quarters of the members of the House.
“There is no reason that patients should be waiting for medically appropriate care, especially when we know that this can lead to worse outcomes,” Rep. Earl Blumenauer (D-Ore.) said in a Sept. 14 speech on the House floor. “The fundamental promise of Medicare Advantage is undermined when people are delaying care, getting sicker, and ultimately costing Medicare more money.”
Rep. Greg Murphy, MD (R-N.C.), spoke on the House floor that day as well, bringing up cases he has seen in his own urology practice in which prior authorization delays disrupted medical care. One patient wound up in the hospital with abscess after an insurer denied an antibiotic prescription, Rep. Murphy said.
But the Senate appears unlikely at this time to move the prior authorization bill as a standalone measure. Instead, the bill may become part of a larger legislative package focused on health care that the Senate Finance Committee intends to prepare later this year.
The House-passed bill would require insurer-run Medicare plans to respond to expedited requests for prior authorization of services within 24 hours and to other requests within 7 days. This bill also would establish an electronic program for prior authorizations and mandate increased transparency as to how insurers use this tool.
CBO: Cost of change would be billions
In seeking to mandate changes in prior authorization, lawmakers likely will need to contend with the issue of a $16 billion cumulative cost estimate for the bill from the Congressional Budget Office. Members of Congress often seek to offset new spending by pairing bills that add to expected costs for the federal government with ones expected to produce savings.
Unlike Rep. Blumenauer, Rep. Murphy, and other backers of the prior authorization streamlining bill, CBO staff estimates that making the mandated changes would raise federal spending, inasmuch as there would be “a greater use of services.”
On Sept. 14, CBO issued a one-page report on the costs of the bill. The CBO report concerns only the bill in question, as is common practice with the office’s estimates.
Prior authorization changes would begin in fiscal 2025 and would add $899 million in spending, or outlays, that year, CBO said. The annual costs from the streamlined prior authorization practices through fiscal 2026 to 2032 range from $1.6 billion to $2.7 billion.
Looking at the CBO estimate against a backdrop of total Medicare Advantage costs, though, may provide important context.
The increases in spending estimated by CBO may suggest that there would be little change in federal spending as a result of streamlining prior authorization practices. These estimates of increased annual spending of $1.6 billion–$2.7 billion are only a small fraction of the current annual cost of insurer-run Medicare, and they represent an even smaller share of the projected expense.
The federal government last year spent about $350 billion on insurer-run plans, excluding Part D drug plan payments, according to the Medicare Advisory Payment Commission (MedPAC).
As of 2021, about 27 million people were enrolled in these plans, accounting for about 46% of the total Medicare population. Enrollment has doubled since 2010, MedPAC said, and it is expected to continue to grow. By 2027, insurer-run Medicare could cover 50% of the program’s population, a figure that may reach 53% by 2031.
Federal payments to these plans will accelerate in the years ahead as insurers attract more people eligible for Medicare as customers. Payments to these private health plans could rise from an expected $418 billion this year to $940.6 billion by 2031, according to the most recent Medicare trustees report.
Good intentions, poor implementation?
Insurer-run Medicare has long enjoyed deep bipartisan support in Congress. That’s due in part to its potential for reducing spending on what are considered low-value treatments, or ones considered unlikely to provide a significant medical benefit, but Rep. Blumenauer is among the members of Congress who see insurer-run Medicare as a path for preserving the giant federal health program. Traditional Medicare has far fewer restrictions on services, which sometimes opens a path for tests and treatments that offer less value for patients.
“I believe that the way traditional fee-for-service Medicare operates is not sustainable and that Medicare Advantage is one of the tools we can use to demonstrate how we can incentivize value,” Rep. Blumenauer said on the House floor. “But this is only possible when the program operates as intended. I have been deeply concerned about the reports of delays in care” caused by the clunky prior authorization processes.
He highlighted a recent report from the internal watchdog group for the Department of Health & Human Services that raises concerns about denials of appropriate care. About 18% of a set of payment denials examined by the Office of Inspector General of HHS in April actually met Medicare coverage rules and plan billing rules.
“For patients and their families, being told that you need to wait longer for care that your doctor tells you that you need is incredibly frustrating and frightening,” Rep. Blumenauer said. “There’s no comfort to be found in the fact that your insurance company needs time to decide if your doctor is right.”
Trends in prior authorization
The CBO report does not provide detail on what kind of medical spending would increase under a streamlined prior authorization process in insurer-run Medicare plans.
From trends reported in prior authorization, though, two factors could be at play in what appear to be relatively small estimated increases in Medicare spending from streamlined prior authorization.
One is the work already underway to create less burdensome electronic systems for these requests, such as the Fast Prior Authorization Technology Highway initiative run by the trade association America’s Health Insurance Plans.
The other factor could be the number of cases in which prior authorization merely causes delays in treatments and tests and thus simply postpones spending while adding to clinicians’ administrative work.
An analysis of prior authorization requests for dermatologic practices affiliated with the University of Utah may represent an extreme example. In a report published in JAMA Dermatology in 2020, researchers described what happened with requests made during 1 month, September 2016.
The approval rate for procedures was 99.6% – 100% (95 of 95) for Mohs surgery, and 96% (130 of 131, with 4 additional cases pending) for excisions. These findings supported calls for simplifying prior authorization procedures, “perhaps first by eliminating unnecessary PAs [prior authorizations] and appeals,” Aaron M. Secrest, MD, PhD, of the University of Utah, Salt Lake City, and coauthors wrote in the article.
Still, there is some evidence that insurer-run Medicare policies reduce the use of low-value care.
In a study published in JAMA Health Forum, Emily Boudreau, PhD, of insurer Humana Inc, and coauthors from Tufts University, Boston, and the University of Pennsylvania, Philadelphia investigated whether insurer-run Medicare could do a better job in reducing the amount of low-value care delivered than the traditional program. They analyzed a set of claims data from 2017 to 2019 for people enrolled in insurer-run and traditional Medicare.
They reported a rate of 23.07 low-value services provided per 100 people in insurer-run Medicare, compared with 25.39 for those in traditional Medicare. Some of the biggest differences reported in the article were in cancer screenings for older people.
As an example, the U.S. Preventive Services Task Force recommends that women older than 65 years not be screened for cervical cancer if they have undergone adequate screening in the past and are not at high risk for cervical cancer. There was an annual count of 1.76 screenings for cervical cancer per 100 women older than 65 in the insurer-run Medicare group versus 3.18 for those in traditional Medicare.
The Better Medicare Alliance issued a statement in favor of the House passage of the Improving Seniors’ Timely Access to Care Act.
In it, the group said the measure would “modernize prior authorization while protecting its essential function in facilitating safe, high-value, evidence-based care.” The alliance promotes use of insurer-run Medicare. The board of the Better Medicare Alliance includes executives who serve with firms that run Advantage plans as well as medical organizations and universities.
“With studies showing that up to one-quarter of all health care expenditures are wasted on services with no benefit to the patient, we need a robust, next-generation prior authorization program to deter low-value, and even harmful, care while protecting access to needed treatment and effective therapies,” said A. Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, in a statement issued by the Better Medicare Alliance. He is a member of the group’s council of scholars.
On the House floor on September 14, Rep. Ami Bera, MD (D-Calif.), said he has heard from former colleagues and his medical school classmates that they now spend as much as 40% of their time on administrative work. These distractions from patient care are helping drive physicians away from the practice of medicine.
Still, the internist defended the basic premise of prior authorization while strongly appealing for better systems of handling it.
“Yes, there is a role for prior authorization in limited cases. There is also a role to go back and retrospectively look at how care is being delivered,” Rep. Bera said. “But what is happening today is a travesty. It wasn’t the intention of prior authorization. It is a prior authorization process gone awry.”
A version of this article first appeared on Medscape.com.
The path through the U.S. Senate is not yet certain for a bill intended to speed the prior authorization process of insurer-run Medicare Advantage plans, despite the measure having breezed through the House.
House leaders opted to move the Improving Seniors’ Timely Access to Care Act of 2021 (HR 3173) without requiring a roll-call vote. The measure was passed on Sept. 14 by a voice vote, an approach used in general with only uncontroversial measures that have broad support. The bill has 191 Democratic and 135 Republican sponsors, representing about three-quarters of the members of the House.
“There is no reason that patients should be waiting for medically appropriate care, especially when we know that this can lead to worse outcomes,” Rep. Earl Blumenauer (D-Ore.) said in a Sept. 14 speech on the House floor. “The fundamental promise of Medicare Advantage is undermined when people are delaying care, getting sicker, and ultimately costing Medicare more money.”
Rep. Greg Murphy, MD (R-N.C.), spoke on the House floor that day as well, bringing up cases he has seen in his own urology practice in which prior authorization delays disrupted medical care. One patient wound up in the hospital with abscess after an insurer denied an antibiotic prescription, Rep. Murphy said.
But the Senate appears unlikely at this time to move the prior authorization bill as a standalone measure. Instead, the bill may become part of a larger legislative package focused on health care that the Senate Finance Committee intends to prepare later this year.
The House-passed bill would require insurer-run Medicare plans to respond to expedited requests for prior authorization of services within 24 hours and to other requests within 7 days. This bill also would establish an electronic program for prior authorizations and mandate increased transparency as to how insurers use this tool.
CBO: Cost of change would be billions
In seeking to mandate changes in prior authorization, lawmakers likely will need to contend with the issue of a $16 billion cumulative cost estimate for the bill from the Congressional Budget Office. Members of Congress often seek to offset new spending by pairing bills that add to expected costs for the federal government with ones expected to produce savings.
Unlike Rep. Blumenauer, Rep. Murphy, and other backers of the prior authorization streamlining bill, CBO staff estimates that making the mandated changes would raise federal spending, inasmuch as there would be “a greater use of services.”
On Sept. 14, CBO issued a one-page report on the costs of the bill. The CBO report concerns only the bill in question, as is common practice with the office’s estimates.
Prior authorization changes would begin in fiscal 2025 and would add $899 million in spending, or outlays, that year, CBO said. The annual costs from the streamlined prior authorization practices through fiscal 2026 to 2032 range from $1.6 billion to $2.7 billion.
Looking at the CBO estimate against a backdrop of total Medicare Advantage costs, though, may provide important context.
The increases in spending estimated by CBO may suggest that there would be little change in federal spending as a result of streamlining prior authorization practices. These estimates of increased annual spending of $1.6 billion–$2.7 billion are only a small fraction of the current annual cost of insurer-run Medicare, and they represent an even smaller share of the projected expense.
The federal government last year spent about $350 billion on insurer-run plans, excluding Part D drug plan payments, according to the Medicare Advisory Payment Commission (MedPAC).
As of 2021, about 27 million people were enrolled in these plans, accounting for about 46% of the total Medicare population. Enrollment has doubled since 2010, MedPAC said, and it is expected to continue to grow. By 2027, insurer-run Medicare could cover 50% of the program’s population, a figure that may reach 53% by 2031.
Federal payments to these plans will accelerate in the years ahead as insurers attract more people eligible for Medicare as customers. Payments to these private health plans could rise from an expected $418 billion this year to $940.6 billion by 2031, according to the most recent Medicare trustees report.
Good intentions, poor implementation?
Insurer-run Medicare has long enjoyed deep bipartisan support in Congress. That’s due in part to its potential for reducing spending on what are considered low-value treatments, or ones considered unlikely to provide a significant medical benefit, but Rep. Blumenauer is among the members of Congress who see insurer-run Medicare as a path for preserving the giant federal health program. Traditional Medicare has far fewer restrictions on services, which sometimes opens a path for tests and treatments that offer less value for patients.
“I believe that the way traditional fee-for-service Medicare operates is not sustainable and that Medicare Advantage is one of the tools we can use to demonstrate how we can incentivize value,” Rep. Blumenauer said on the House floor. “But this is only possible when the program operates as intended. I have been deeply concerned about the reports of delays in care” caused by the clunky prior authorization processes.
He highlighted a recent report from the internal watchdog group for the Department of Health & Human Services that raises concerns about denials of appropriate care. About 18% of a set of payment denials examined by the Office of Inspector General of HHS in April actually met Medicare coverage rules and plan billing rules.
“For patients and their families, being told that you need to wait longer for care that your doctor tells you that you need is incredibly frustrating and frightening,” Rep. Blumenauer said. “There’s no comfort to be found in the fact that your insurance company needs time to decide if your doctor is right.”
Trends in prior authorization
The CBO report does not provide detail on what kind of medical spending would increase under a streamlined prior authorization process in insurer-run Medicare plans.
From trends reported in prior authorization, though, two factors could be at play in what appear to be relatively small estimated increases in Medicare spending from streamlined prior authorization.
One is the work already underway to create less burdensome electronic systems for these requests, such as the Fast Prior Authorization Technology Highway initiative run by the trade association America’s Health Insurance Plans.
The other factor could be the number of cases in which prior authorization merely causes delays in treatments and tests and thus simply postpones spending while adding to clinicians’ administrative work.
An analysis of prior authorization requests for dermatologic practices affiliated with the University of Utah may represent an extreme example. In a report published in JAMA Dermatology in 2020, researchers described what happened with requests made during 1 month, September 2016.
The approval rate for procedures was 99.6% – 100% (95 of 95) for Mohs surgery, and 96% (130 of 131, with 4 additional cases pending) for excisions. These findings supported calls for simplifying prior authorization procedures, “perhaps first by eliminating unnecessary PAs [prior authorizations] and appeals,” Aaron M. Secrest, MD, PhD, of the University of Utah, Salt Lake City, and coauthors wrote in the article.
Still, there is some evidence that insurer-run Medicare policies reduce the use of low-value care.
In a study published in JAMA Health Forum, Emily Boudreau, PhD, of insurer Humana Inc, and coauthors from Tufts University, Boston, and the University of Pennsylvania, Philadelphia investigated whether insurer-run Medicare could do a better job in reducing the amount of low-value care delivered than the traditional program. They analyzed a set of claims data from 2017 to 2019 for people enrolled in insurer-run and traditional Medicare.
They reported a rate of 23.07 low-value services provided per 100 people in insurer-run Medicare, compared with 25.39 for those in traditional Medicare. Some of the biggest differences reported in the article were in cancer screenings for older people.
As an example, the U.S. Preventive Services Task Force recommends that women older than 65 years not be screened for cervical cancer if they have undergone adequate screening in the past and are not at high risk for cervical cancer. There was an annual count of 1.76 screenings for cervical cancer per 100 women older than 65 in the insurer-run Medicare group versus 3.18 for those in traditional Medicare.
The Better Medicare Alliance issued a statement in favor of the House passage of the Improving Seniors’ Timely Access to Care Act.
In it, the group said the measure would “modernize prior authorization while protecting its essential function in facilitating safe, high-value, evidence-based care.” The alliance promotes use of insurer-run Medicare. The board of the Better Medicare Alliance includes executives who serve with firms that run Advantage plans as well as medical organizations and universities.
“With studies showing that up to one-quarter of all health care expenditures are wasted on services with no benefit to the patient, we need a robust, next-generation prior authorization program to deter low-value, and even harmful, care while protecting access to needed treatment and effective therapies,” said A. Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, in a statement issued by the Better Medicare Alliance. He is a member of the group’s council of scholars.
On the House floor on September 14, Rep. Ami Bera, MD (D-Calif.), said he has heard from former colleagues and his medical school classmates that they now spend as much as 40% of their time on administrative work. These distractions from patient care are helping drive physicians away from the practice of medicine.
Still, the internist defended the basic premise of prior authorization while strongly appealing for better systems of handling it.
“Yes, there is a role for prior authorization in limited cases. There is also a role to go back and retrospectively look at how care is being delivered,” Rep. Bera said. “But what is happening today is a travesty. It wasn’t the intention of prior authorization. It is a prior authorization process gone awry.”
A version of this article first appeared on Medscape.com.
Ultrasonic renal denervation passes 2-month test in uncontrolled HTN: RADIANCE II
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.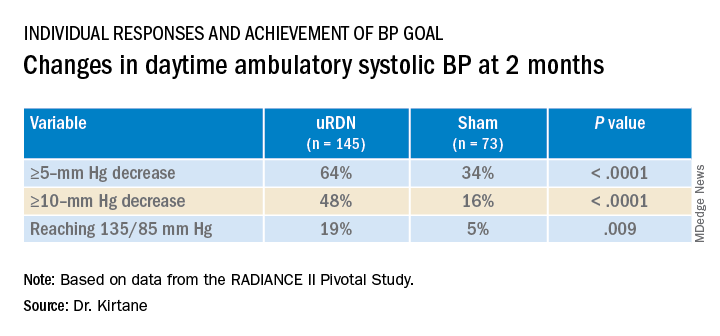
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).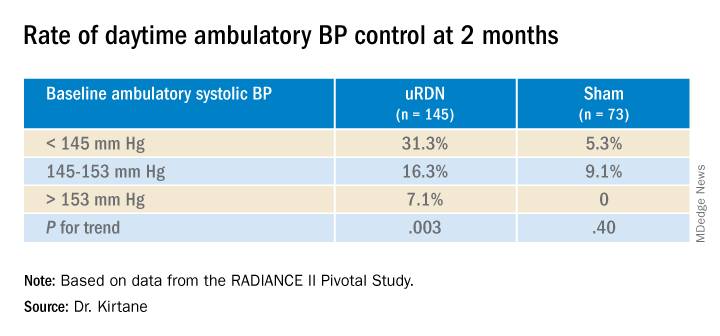
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
TAVR now used in almost 50% of younger severe aortic stenosis patients
Among patients with severe isolated aortic stenosis younger than 65, the rate of transcatheter aortic valve replacement (TAVR) now almost matches that of surgical aortic valve replacement (SAVR), despite guideline recommendations to the contrary, a study in a national U.S. population shows.
The 2020 American Heart Association/American College of Cardiology (AHA/ACC) valve guideline recommends SAVR for patients younger than 65 with severe aortic stenosis, the researchers note, but their study showed “near equal utilization between TAVR and SAVR in these younger patients by 2021,” at 48% and 52% respectively.
Toishi Sharma, MD, and colleagues presented these findings in an oral poster session at Transcatheter Cardiovascular Therapeutics 2022, and the study was simultaneously published as a Research Letter in the Journal of the American College of Cardiology (JACC).
“To our knowledge, the current findings represent the first national temporal trends study stratifying [aortic stenosis] therapies according to guideline-recommended age groups: our observations demonstrate the dramatic growth of TAVR in all age groups, including young patients,” the researchers conclude.
They analyzed changes in rates of TAVR and SAVR in a U.S. sample stratified by age: younger than 65 years, 65-80, and older than 80 years.
These findings have implications for lifetime management of younger patients who undergo TAVR, they write, “including issues related to lifetime coronary access, valve durability, and the potential for subsequent TAVR procedures over time.”
Three age groups
In a study published in JACC, this group examined changes in uptake of TAVR versus SAVR in 4,161 patients with aortic stenosis in Vermont, New Hampshire, and Maine, senior author Harold L. Dauerman, MD, said in an interview.
The greatest rate of rise of TAVR was in the group younger than 65, but that study ended in 2019, said Dr. Dauerman, from the University of Vermont Health Network, Burlington.
The 2020 guideline stratifies TAVR and SAVR recommendations such that “less than 65 should primarily be a surgical approach and greater than 80 primarily a TAVR approach, while 65 to 80 is a gray zone, and shared decision-making becomes important,” he noted.
The group hypothesized that recent trials and technology have led to a national increase in TAVR in people younger than 65.
From the Vizient clinical database, including more than 250 U.S. academic centers that perform both TAVR and SAVR, the researchers identified 142,953 patients who underwent TAVR or SAVR for isolated aortic stenosis from Oct. 1, 2015, to Dec. 31, 2021. From 2015 to 2021, the valve replacement rates in the three age groups changed as follows:
- Age less than 65: TAVR rose from 17% to 48%; SAVR fell from 83% to 52%.
- Age 65-80: TAVR rose from 46% to 87%; SAVR fell from 54% to 12%.
- Age greater than 80: TAVR rose from 83% to 99%; SAVR fell from 16% to 1%.
“All ages have grown in the last 7 years in TAVR,” Dr. Dauerman summarized. “The one that’s surprising, and in contradiction to the guideline, is the growth of TAVR in young patients less than 65.”
Among patients younger than 65, prior bypass surgery and congestive heart failure predicted the use of TAVR instead of surgery, whereas bicuspid aortic valve disease was the biggest predictor of surgery instead of TAVR.
Most studies on TAVR valve durability are limited to patients in the randomized trials who were primarily in their mid-70s to mid-80s, some of whom died before a 10-year follow-up, Dr. Dauerman noted.
European guidelines recommend surgery for patients younger than 70, and it would be interesting to see if clinicians there follow this recommendation or if TAVR is now the preferred approach, he added.
There is a need for further, longer study of TAVR in younger patients, he said, to determine whether there are long-term clinical issues of concern.
Strategy depends on more than age
The “findings are not too surprising,” John Carroll, MD, who was not involved in this research, said in an email.
“Age is only one of multiple patient characteristics that enter into consideration of TAVR versus SAVR,” said Dr. Carroll, from Anschutz Medical Campus, University of Colorado, Aurora.
“As the article reports,” he noted, “those less than 65 having TAVR are more likely to have comorbid conditions that likely made the risk of SAVR higher.”
Dr. Carroll was lead author of a review article published in 2020 based on data from the ACC–Society of Thoracic Surgeons (STS)–Transcatheter Valve Therapy (TVT) registry on 276,316 patients who had TAVR in the United States from 2011-2019.
He pointed out that Figure 2 in that review shows that “SAVR is often performed in conjunction with other surgical procedures – another major reason why SAVR remains an important treatment for valvular heart disease.”
“We are awaiting long-term data comparing TAVR to SAVR durability,” Dr. Carroll added, echoing Dr. Dauerman. “So far [there are] no major differences, but it remains a key need to fully understand TAVR and the various models in commercial use.”
“Both TAVR and SAVR used in adults are tissue valves (SAVR with mechanical valves is used in younger patients),” Dr. Carroll noted, “and all tissue valves will eventually fail if the patient lives long enough.”
Patient management strategies need to consider what treatment options exist when the first valve fails. “If the first valve is SAVR, there is now extensive experience with placing a TAVR valve inside a failing SAVR valve, so called Valve-in-Valve or TAVR-in-SAVR. This is the preferred treatment in most patients with failing SAVR valves,” he said.
“On the other hand,” he continued, “we are just beginning to see more and more patients with failing TAVR valves, and the TAVR-in-TAVR procedure is less well understood.”
“Issues such as acute coronary occlusion and long-term difficulty in accessing coronary arteries are being encountered in some patients having TAVR-in-TAVR,” Dr. Carroll noted, which he discusses in a recent editorial he coauthored about the complexities of redo TAVR, published in JACC: Cardiovascular Interventions.
The study received no funding. Dr. Dauerman has research grants and is a consultant for Medtronic and Boston Scientific. Dr. Carroll is a local principal investigator in trials sponsored by Medtronic, Abbott, and Edwards Lifesciences.
A version of this article first appeared on Medscape.com.
Among patients with severe isolated aortic stenosis younger than 65, the rate of transcatheter aortic valve replacement (TAVR) now almost matches that of surgical aortic valve replacement (SAVR), despite guideline recommendations to the contrary, a study in a national U.S. population shows.
The 2020 American Heart Association/American College of Cardiology (AHA/ACC) valve guideline recommends SAVR for patients younger than 65 with severe aortic stenosis, the researchers note, but their study showed “near equal utilization between TAVR and SAVR in these younger patients by 2021,” at 48% and 52% respectively.
Toishi Sharma, MD, and colleagues presented these findings in an oral poster session at Transcatheter Cardiovascular Therapeutics 2022, and the study was simultaneously published as a Research Letter in the Journal of the American College of Cardiology (JACC).
“To our knowledge, the current findings represent the first national temporal trends study stratifying [aortic stenosis] therapies according to guideline-recommended age groups: our observations demonstrate the dramatic growth of TAVR in all age groups, including young patients,” the researchers conclude.
They analyzed changes in rates of TAVR and SAVR in a U.S. sample stratified by age: younger than 65 years, 65-80, and older than 80 years.
These findings have implications for lifetime management of younger patients who undergo TAVR, they write, “including issues related to lifetime coronary access, valve durability, and the potential for subsequent TAVR procedures over time.”
Three age groups
In a study published in JACC, this group examined changes in uptake of TAVR versus SAVR in 4,161 patients with aortic stenosis in Vermont, New Hampshire, and Maine, senior author Harold L. Dauerman, MD, said in an interview.
The greatest rate of rise of TAVR was in the group younger than 65, but that study ended in 2019, said Dr. Dauerman, from the University of Vermont Health Network, Burlington.
The 2020 guideline stratifies TAVR and SAVR recommendations such that “less than 65 should primarily be a surgical approach and greater than 80 primarily a TAVR approach, while 65 to 80 is a gray zone, and shared decision-making becomes important,” he noted.
The group hypothesized that recent trials and technology have led to a national increase in TAVR in people younger than 65.
From the Vizient clinical database, including more than 250 U.S. academic centers that perform both TAVR and SAVR, the researchers identified 142,953 patients who underwent TAVR or SAVR for isolated aortic stenosis from Oct. 1, 2015, to Dec. 31, 2021. From 2015 to 2021, the valve replacement rates in the three age groups changed as follows:
- Age less than 65: TAVR rose from 17% to 48%; SAVR fell from 83% to 52%.
- Age 65-80: TAVR rose from 46% to 87%; SAVR fell from 54% to 12%.
- Age greater than 80: TAVR rose from 83% to 99%; SAVR fell from 16% to 1%.
“All ages have grown in the last 7 years in TAVR,” Dr. Dauerman summarized. “The one that’s surprising, and in contradiction to the guideline, is the growth of TAVR in young patients less than 65.”
Among patients younger than 65, prior bypass surgery and congestive heart failure predicted the use of TAVR instead of surgery, whereas bicuspid aortic valve disease was the biggest predictor of surgery instead of TAVR.
Most studies on TAVR valve durability are limited to patients in the randomized trials who were primarily in their mid-70s to mid-80s, some of whom died before a 10-year follow-up, Dr. Dauerman noted.
European guidelines recommend surgery for patients younger than 70, and it would be interesting to see if clinicians there follow this recommendation or if TAVR is now the preferred approach, he added.
There is a need for further, longer study of TAVR in younger patients, he said, to determine whether there are long-term clinical issues of concern.
Strategy depends on more than age
The “findings are not too surprising,” John Carroll, MD, who was not involved in this research, said in an email.
“Age is only one of multiple patient characteristics that enter into consideration of TAVR versus SAVR,” said Dr. Carroll, from Anschutz Medical Campus, University of Colorado, Aurora.
“As the article reports,” he noted, “those less than 65 having TAVR are more likely to have comorbid conditions that likely made the risk of SAVR higher.”
Dr. Carroll was lead author of a review article published in 2020 based on data from the ACC–Society of Thoracic Surgeons (STS)–Transcatheter Valve Therapy (TVT) registry on 276,316 patients who had TAVR in the United States from 2011-2019.
He pointed out that Figure 2 in that review shows that “SAVR is often performed in conjunction with other surgical procedures – another major reason why SAVR remains an important treatment for valvular heart disease.”
“We are awaiting long-term data comparing TAVR to SAVR durability,” Dr. Carroll added, echoing Dr. Dauerman. “So far [there are] no major differences, but it remains a key need to fully understand TAVR and the various models in commercial use.”
“Both TAVR and SAVR used in adults are tissue valves (SAVR with mechanical valves is used in younger patients),” Dr. Carroll noted, “and all tissue valves will eventually fail if the patient lives long enough.”
Patient management strategies need to consider what treatment options exist when the first valve fails. “If the first valve is SAVR, there is now extensive experience with placing a TAVR valve inside a failing SAVR valve, so called Valve-in-Valve or TAVR-in-SAVR. This is the preferred treatment in most patients with failing SAVR valves,” he said.
“On the other hand,” he continued, “we are just beginning to see more and more patients with failing TAVR valves, and the TAVR-in-TAVR procedure is less well understood.”
“Issues such as acute coronary occlusion and long-term difficulty in accessing coronary arteries are being encountered in some patients having TAVR-in-TAVR,” Dr. Carroll noted, which he discusses in a recent editorial he coauthored about the complexities of redo TAVR, published in JACC: Cardiovascular Interventions.
The study received no funding. Dr. Dauerman has research grants and is a consultant for Medtronic and Boston Scientific. Dr. Carroll is a local principal investigator in trials sponsored by Medtronic, Abbott, and Edwards Lifesciences.
A version of this article first appeared on Medscape.com.
Among patients with severe isolated aortic stenosis younger than 65, the rate of transcatheter aortic valve replacement (TAVR) now almost matches that of surgical aortic valve replacement (SAVR), despite guideline recommendations to the contrary, a study in a national U.S. population shows.
The 2020 American Heart Association/American College of Cardiology (AHA/ACC) valve guideline recommends SAVR for patients younger than 65 with severe aortic stenosis, the researchers note, but their study showed “near equal utilization between TAVR and SAVR in these younger patients by 2021,” at 48% and 52% respectively.
Toishi Sharma, MD, and colleagues presented these findings in an oral poster session at Transcatheter Cardiovascular Therapeutics 2022, and the study was simultaneously published as a Research Letter in the Journal of the American College of Cardiology (JACC).
“To our knowledge, the current findings represent the first national temporal trends study stratifying [aortic stenosis] therapies according to guideline-recommended age groups: our observations demonstrate the dramatic growth of TAVR in all age groups, including young patients,” the researchers conclude.
They analyzed changes in rates of TAVR and SAVR in a U.S. sample stratified by age: younger than 65 years, 65-80, and older than 80 years.
These findings have implications for lifetime management of younger patients who undergo TAVR, they write, “including issues related to lifetime coronary access, valve durability, and the potential for subsequent TAVR procedures over time.”
Three age groups
In a study published in JACC, this group examined changes in uptake of TAVR versus SAVR in 4,161 patients with aortic stenosis in Vermont, New Hampshire, and Maine, senior author Harold L. Dauerman, MD, said in an interview.
The greatest rate of rise of TAVR was in the group younger than 65, but that study ended in 2019, said Dr. Dauerman, from the University of Vermont Health Network, Burlington.
The 2020 guideline stratifies TAVR and SAVR recommendations such that “less than 65 should primarily be a surgical approach and greater than 80 primarily a TAVR approach, while 65 to 80 is a gray zone, and shared decision-making becomes important,” he noted.
The group hypothesized that recent trials and technology have led to a national increase in TAVR in people younger than 65.
From the Vizient clinical database, including more than 250 U.S. academic centers that perform both TAVR and SAVR, the researchers identified 142,953 patients who underwent TAVR or SAVR for isolated aortic stenosis from Oct. 1, 2015, to Dec. 31, 2021. From 2015 to 2021, the valve replacement rates in the three age groups changed as follows:
- Age less than 65: TAVR rose from 17% to 48%; SAVR fell from 83% to 52%.
- Age 65-80: TAVR rose from 46% to 87%; SAVR fell from 54% to 12%.
- Age greater than 80: TAVR rose from 83% to 99%; SAVR fell from 16% to 1%.
“All ages have grown in the last 7 years in TAVR,” Dr. Dauerman summarized. “The one that’s surprising, and in contradiction to the guideline, is the growth of TAVR in young patients less than 65.”
Among patients younger than 65, prior bypass surgery and congestive heart failure predicted the use of TAVR instead of surgery, whereas bicuspid aortic valve disease was the biggest predictor of surgery instead of TAVR.
Most studies on TAVR valve durability are limited to patients in the randomized trials who were primarily in their mid-70s to mid-80s, some of whom died before a 10-year follow-up, Dr. Dauerman noted.
European guidelines recommend surgery for patients younger than 70, and it would be interesting to see if clinicians there follow this recommendation or if TAVR is now the preferred approach, he added.
There is a need for further, longer study of TAVR in younger patients, he said, to determine whether there are long-term clinical issues of concern.
Strategy depends on more than age
The “findings are not too surprising,” John Carroll, MD, who was not involved in this research, said in an email.
“Age is only one of multiple patient characteristics that enter into consideration of TAVR versus SAVR,” said Dr. Carroll, from Anschutz Medical Campus, University of Colorado, Aurora.
“As the article reports,” he noted, “those less than 65 having TAVR are more likely to have comorbid conditions that likely made the risk of SAVR higher.”
Dr. Carroll was lead author of a review article published in 2020 based on data from the ACC–Society of Thoracic Surgeons (STS)–Transcatheter Valve Therapy (TVT) registry on 276,316 patients who had TAVR in the United States from 2011-2019.
He pointed out that Figure 2 in that review shows that “SAVR is often performed in conjunction with other surgical procedures – another major reason why SAVR remains an important treatment for valvular heart disease.”
“We are awaiting long-term data comparing TAVR to SAVR durability,” Dr. Carroll added, echoing Dr. Dauerman. “So far [there are] no major differences, but it remains a key need to fully understand TAVR and the various models in commercial use.”
“Both TAVR and SAVR used in adults are tissue valves (SAVR with mechanical valves is used in younger patients),” Dr. Carroll noted, “and all tissue valves will eventually fail if the patient lives long enough.”
Patient management strategies need to consider what treatment options exist when the first valve fails. “If the first valve is SAVR, there is now extensive experience with placing a TAVR valve inside a failing SAVR valve, so called Valve-in-Valve or TAVR-in-SAVR. This is the preferred treatment in most patients with failing SAVR valves,” he said.
“On the other hand,” he continued, “we are just beginning to see more and more patients with failing TAVR valves, and the TAVR-in-TAVR procedure is less well understood.”
“Issues such as acute coronary occlusion and long-term difficulty in accessing coronary arteries are being encountered in some patients having TAVR-in-TAVR,” Dr. Carroll noted, which he discusses in a recent editorial he coauthored about the complexities of redo TAVR, published in JACC: Cardiovascular Interventions.
The study received no funding. Dr. Dauerman has research grants and is a consultant for Medtronic and Boston Scientific. Dr. Carroll is a local principal investigator in trials sponsored by Medtronic, Abbott, and Edwards Lifesciences.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
Obstructive sleep apnea linked to unprovoked VTE
Add unprovoked venous thromboembolic events to the list of potential consequences of severe obstructive sleep apnea.
That conclusion comes from a study showing that patients with obstructive sleep apnea (OSA) who had the longest nocturnal hypoxemia episodes had a twofold risk for venous thromboembolic events.
systems, reported Wojciech Trzepizur, MD, of Angers University Hospital, France.
Previous studies have suggested links between OSA and both cancer and cognitive decline, but this is the first study to investigate the association between OSA and the incidence of unprovoked VTE, he reported in an oral abstract session at the annual congress of the European Respiratory Society.
“We found that those who spent more than 6% of their nighttime with levels of oxygen in their blood below 90% of normal had an almost twofold risk of developing VTEs compared to patients without oxygen deprivation,” he said.
Dr. Trzepizur and colleagues conducted a retrospective study linking cohort data to an administrative health database. They identified unprovoked VTE in patients with a suspicion for OSA and no previous VTE.
They created Cox proportional hazard models to assess the association of unprovoked VTE with apnea hypopnea index (AHI) measures and nocturnal hypoxemia markers, including the time patients spent below 90% oxygen saturation (T90), oxygen desaturation index (ODI), and hypoxic burden, defined as the total area under the respiratory event-related desaturation curve.
They found that after a median follow-up of 6.3 years, 104 out of 7,355 patients had an unprovoked VTE. In an unadjusted hazard model, there were significant associations between VTE and T90, as well as with hypoxic burden, but not with either AHI or ODI.
However, in an analysis adjusted for age, gender, body mass index, alcohol intake, hypertension, depression, history of cardiovascular disease, statin use, type of sleep study, study site, and CPAP adherence, the investigators found that only T90 remained a significant independent predictor of VTE, with a hazard ratio of 1.06, P = .02.
The association between T90 and VTE strengthened as the time spent below 90% saturation increased. Patients in the highest tercile, who spent more than 6% of the time undersaturated, had an HR for VTE of 1.95 (P = .02), compared with patients with a T90 less than 1%.
There were no significant differences in VTE risk between patients who used CPAP for more than 4 hours per night and those who either used the devices for less than 4 hours or refused CPAP.
“We see that T90 seems to be a strong parameter,” said session comoderator Raphael Heinzer, MD, MPH, of Lausanne University Hospital, Switzerland.
Dr. Heinzer’s comoderator, Silke Ryan, MD, of University College Dublin, pointed out that although T90 was the main predictor of responses, Dr. Trzepizur and colleagues did not control for other pulmonary diseases.
“Obviously, there could be an influence of other hypoxic-related diseases,” she said, and recommended controlling for this in future studies.
Winfried Randerath, MD, of the Bethanien Hospital at the University of Cologne, Germany, head of the ERS specialist group on sleep disordered breathing, said that this study and others presented at the meeting “show worrying associations between obstructive sleep apnea and important diseases that affect survival and quality of life.
“While they cannot prove that OSA causes any of these health problems, people should be made aware of these links and should try to make lifestyle changes in order to reduce their risk of OSA, for instance, by maintaining a healthy weight. However, if OSA is suspected, definite diagnosis and treatment should be initiated. We look forward to further research that may help to clarify whether OSA may be causing some of the health problems seen in these studies,” said Dr. Randerath, who was not involved with the study.
The study was supported by a grant from Institut de Recherche en Santé Respiratoire des Pays de la Loire (IRSR), Beaucouzé, France. Dr. Trzepizur, Dr. Heinzer, Dr. Ryan and Dr. Randerath reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Add unprovoked venous thromboembolic events to the list of potential consequences of severe obstructive sleep apnea.
That conclusion comes from a study showing that patients with obstructive sleep apnea (OSA) who had the longest nocturnal hypoxemia episodes had a twofold risk for venous thromboembolic events.
systems, reported Wojciech Trzepizur, MD, of Angers University Hospital, France.
Previous studies have suggested links between OSA and both cancer and cognitive decline, but this is the first study to investigate the association between OSA and the incidence of unprovoked VTE, he reported in an oral abstract session at the annual congress of the European Respiratory Society.
“We found that those who spent more than 6% of their nighttime with levels of oxygen in their blood below 90% of normal had an almost twofold risk of developing VTEs compared to patients without oxygen deprivation,” he said.
Dr. Trzepizur and colleagues conducted a retrospective study linking cohort data to an administrative health database. They identified unprovoked VTE in patients with a suspicion for OSA and no previous VTE.
They created Cox proportional hazard models to assess the association of unprovoked VTE with apnea hypopnea index (AHI) measures and nocturnal hypoxemia markers, including the time patients spent below 90% oxygen saturation (T90), oxygen desaturation index (ODI), and hypoxic burden, defined as the total area under the respiratory event-related desaturation curve.
They found that after a median follow-up of 6.3 years, 104 out of 7,355 patients had an unprovoked VTE. In an unadjusted hazard model, there were significant associations between VTE and T90, as well as with hypoxic burden, but not with either AHI or ODI.
However, in an analysis adjusted for age, gender, body mass index, alcohol intake, hypertension, depression, history of cardiovascular disease, statin use, type of sleep study, study site, and CPAP adherence, the investigators found that only T90 remained a significant independent predictor of VTE, with a hazard ratio of 1.06, P = .02.
The association between T90 and VTE strengthened as the time spent below 90% saturation increased. Patients in the highest tercile, who spent more than 6% of the time undersaturated, had an HR for VTE of 1.95 (P = .02), compared with patients with a T90 less than 1%.
There were no significant differences in VTE risk between patients who used CPAP for more than 4 hours per night and those who either used the devices for less than 4 hours or refused CPAP.
“We see that T90 seems to be a strong parameter,” said session comoderator Raphael Heinzer, MD, MPH, of Lausanne University Hospital, Switzerland.
Dr. Heinzer’s comoderator, Silke Ryan, MD, of University College Dublin, pointed out that although T90 was the main predictor of responses, Dr. Trzepizur and colleagues did not control for other pulmonary diseases.
“Obviously, there could be an influence of other hypoxic-related diseases,” she said, and recommended controlling for this in future studies.
Winfried Randerath, MD, of the Bethanien Hospital at the University of Cologne, Germany, head of the ERS specialist group on sleep disordered breathing, said that this study and others presented at the meeting “show worrying associations between obstructive sleep apnea and important diseases that affect survival and quality of life.
“While they cannot prove that OSA causes any of these health problems, people should be made aware of these links and should try to make lifestyle changes in order to reduce their risk of OSA, for instance, by maintaining a healthy weight. However, if OSA is suspected, definite diagnosis and treatment should be initiated. We look forward to further research that may help to clarify whether OSA may be causing some of the health problems seen in these studies,” said Dr. Randerath, who was not involved with the study.
The study was supported by a grant from Institut de Recherche en Santé Respiratoire des Pays de la Loire (IRSR), Beaucouzé, France. Dr. Trzepizur, Dr. Heinzer, Dr. Ryan and Dr. Randerath reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Add unprovoked venous thromboembolic events to the list of potential consequences of severe obstructive sleep apnea.
That conclusion comes from a study showing that patients with obstructive sleep apnea (OSA) who had the longest nocturnal hypoxemia episodes had a twofold risk for venous thromboembolic events.
systems, reported Wojciech Trzepizur, MD, of Angers University Hospital, France.
Previous studies have suggested links between OSA and both cancer and cognitive decline, but this is the first study to investigate the association between OSA and the incidence of unprovoked VTE, he reported in an oral abstract session at the annual congress of the European Respiratory Society.
“We found that those who spent more than 6% of their nighttime with levels of oxygen in their blood below 90% of normal had an almost twofold risk of developing VTEs compared to patients without oxygen deprivation,” he said.
Dr. Trzepizur and colleagues conducted a retrospective study linking cohort data to an administrative health database. They identified unprovoked VTE in patients with a suspicion for OSA and no previous VTE.
They created Cox proportional hazard models to assess the association of unprovoked VTE with apnea hypopnea index (AHI) measures and nocturnal hypoxemia markers, including the time patients spent below 90% oxygen saturation (T90), oxygen desaturation index (ODI), and hypoxic burden, defined as the total area under the respiratory event-related desaturation curve.
They found that after a median follow-up of 6.3 years, 104 out of 7,355 patients had an unprovoked VTE. In an unadjusted hazard model, there were significant associations between VTE and T90, as well as with hypoxic burden, but not with either AHI or ODI.
However, in an analysis adjusted for age, gender, body mass index, alcohol intake, hypertension, depression, history of cardiovascular disease, statin use, type of sleep study, study site, and CPAP adherence, the investigators found that only T90 remained a significant independent predictor of VTE, with a hazard ratio of 1.06, P = .02.
The association between T90 and VTE strengthened as the time spent below 90% saturation increased. Patients in the highest tercile, who spent more than 6% of the time undersaturated, had an HR for VTE of 1.95 (P = .02), compared with patients with a T90 less than 1%.
There were no significant differences in VTE risk between patients who used CPAP for more than 4 hours per night and those who either used the devices for less than 4 hours or refused CPAP.
“We see that T90 seems to be a strong parameter,” said session comoderator Raphael Heinzer, MD, MPH, of Lausanne University Hospital, Switzerland.
Dr. Heinzer’s comoderator, Silke Ryan, MD, of University College Dublin, pointed out that although T90 was the main predictor of responses, Dr. Trzepizur and colleagues did not control for other pulmonary diseases.
“Obviously, there could be an influence of other hypoxic-related diseases,” she said, and recommended controlling for this in future studies.
Winfried Randerath, MD, of the Bethanien Hospital at the University of Cologne, Germany, head of the ERS specialist group on sleep disordered breathing, said that this study and others presented at the meeting “show worrying associations between obstructive sleep apnea and important diseases that affect survival and quality of life.
“While they cannot prove that OSA causes any of these health problems, people should be made aware of these links and should try to make lifestyle changes in order to reduce their risk of OSA, for instance, by maintaining a healthy weight. However, if OSA is suspected, definite diagnosis and treatment should be initiated. We look forward to further research that may help to clarify whether OSA may be causing some of the health problems seen in these studies,” said Dr. Randerath, who was not involved with the study.
The study was supported by a grant from Institut de Recherche en Santé Respiratoire des Pays de la Loire (IRSR), Beaucouzé, France. Dr. Trzepizur, Dr. Heinzer, Dr. Ryan and Dr. Randerath reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ERS 2022
In cardiogenic shock, edge-to-edge mitral valve repair improves outcome
In patients with severe mitral regurgitation (MR) and cardiogenic shock, successful transcatheter edge-to-edge repair (TEER) is associated with a substantial reduction in all-cause mortality and lower morbidity at 1 year, according to an analysis of registry data.
The data from this analysis also confirm that “successful reduction of MR is achievable with TEER in most patients with cardiogenic shock,” reported Mohamad A. Alkhouli, MD, an interventional cardiologist and professor of medicine at the Mayo Clinic, Rochester, Minn.
In those with device success, achieved in 85.6% of patients, all-cause mortality was about 21% lower (34.6% vs. 55.5%; P < .001) at 1 year than in those who were not successfully repaired, according to Dr. Alkhouli, who presented the findings at the Transcatheter Cardiovascular Therapeutics annual meeting in Boston. This translated into a reduction in the hazard ratio for death of nearly 50% (hazard ratio, 0.52; 95% confidence interval, 0.43-0.63).
A similar relative benefit was found for the composite endpoint of mortality and heart failure admissions at 1 year. Whether unadjusted (HR, 0.54; 95% CI, 0.45-0.66) or adjusted (HR, 0.51; 95% CI, 0.42-0.62), risk reductions with successful MR reduction, defined as greater than or equal to 1 grade improvement and a final MR grade of less than or equal to 2+, indicated that major adverse outcomes are reduced by about half.
STS/ACC TCT registry data queried
Drawn from the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry, 3,797 patients with cardiogenic shock underwent MR repair between November 2013 and December 2021. Outcomes at 1 year were evaluable in 2,773 of these patients. For inclusion, all had to meet at least one of the definitions of cardiogenic shock, such as inotrope use or mechanical circulatory support.
At baseline, 94.5% had a MR severity of at least 3+, and most of these had 4+. Thirty days after treatment, 88.8% had MR severity of 2+ or less, the majority of which had a severity of 1+.
These data address an important question not previously well studied, according to Dr. Alkhouli. In MR patients, cardiogenic shock is associated with a high risk of death, but there has been little evidence that valve repair does not exacerbate, let alone modify, this risk.
These data support the value of intervention, which was performed in almost all patients with MitraClipä (Abbott), the only device available for most of the period in which the registry was queried. However, Dr. Alkhouli cautioned that his data are best considered “hypothesis generating.”
“We need a randomized trial,” he said at the meeting sponsored by the Cardiovascular Research Foundation. He pointed out that this is a complex population for which multiple variables might have skewed results when data are analyzed retrospectively. Not least, those MR patients with cardiogenic shock in the database considered for TEER might well have been relatively healthy and not representative of an unselected population with both MR and cardiogenic shock.
The question might be better answered by the multicenter Canadian trial CAPITAL MINOS, which has just started. Described in an article in the American Heart Journal, it has a planned enrollment of about 150 MR patients with cardiogenic shock randomized to TEER or medical therapy. Results are expected in about 1 year, according to Dr. Alkhouli.
But regarding the present analysis, Dr. Alkhouli did note that sensitivity analyses conducted within his data across risk factors, such as degenerative versus nondegenerative MR, low (< 30%) versus higher left ventricular ejection fraction (LVEF), and presence or absence of an acute coronary syndrome (ACS), consistently supported a benefit from intervention.
Also, cardiogenic shock did not appear to be a factor in device failure, according to Dr. Alkhouli, addressing a potential criticism that cardiogenic shock was an underlying reason for device failure.
More than 90% in NYHA class III or IV heart failure
In this study, the mean age was 73 years. More than 90% were in class III or IV heart failure in the 2 weeks prior to TEER. More than half had established coronary artery disease. Other concomitant cardiovascular morbidities, including atrial fibrillation or flutter (65%), prior MI (39%), and prior stroke or transient ischemic attach (> 10%) were well represented.
When those with device success were compared with those with device failure, the risk profile was comparable. The predicted STS (Society of Thoracic Surgeons) mortality for mitral valve repair among these two groups was 14.8% versus 15% (P = 0.97), respectively.
However, those with device failure did have a lower baseline left ventricular ejection fraction (40.7% vs. 42.9%; P = .009) and a greater prevalence of moderate-to-severe or severe MR (96.1% vs. 84.9%; P < 0.001).
The growing experience with TEER means that benefit has now been shown in several complicated MR groups, such as those with severe ventricular dysfunction, renal insufficiency, and obstructive lung disease. This was a rationale for looking at the impact or repairing MR in patients with cardiogenic shock.
It is a pressing question, according to Dr. Alkhouli. He cited studies suggesting that up to 20% of patients hospitalized for cardiogenic shock have at least moderate-to-severe MR. Conversely, cardiogenic shock is not an uncommon finding in patients with MR.
While Dr. Alkhouli acknowledged that the many variables influencing outcome in patients with MR and cardiogenic shock will make a randomized trial “challenging,” many experts echoed this concern and even expressed some skepticism about the potential for an unbiased trial.
Data confirm MR repair is safe during shock
“These data do show that repair of MR is safe in patients safe in patients with cardiogenic shock,” said Anita W. Asgar, MD, an interventional cardiologist associated with the Montreal Heart Institute. She noted that there was a 5- to 6-day delay among the cardiogenic shock patients prior to undergoing MR repair in this analysis, potentially reflecting an elimination of those at very high risk. Similarly, she suggested that many interventionalists are likely to consider multiple variables before proceeding.
As a result, MR repair may not be amenable to randomization in a cardiogenic shock population, given that this decision is not typically undertaken out of the context of multiple variables.
“I am not sure that a clinical trial is ethical,” she said. She would expect that clinicians enrolling patients would only do so on a selective basis.
Alexandra J. Lansky, MD, Director of the Yale Heart and Vascular Research Program, Yale University, New Haven, Conn., also emphasized the difficulty of controlling for variables, such as the duration of cardiogenic shock, that influence decision-making.
Nevertheless, she called the data “very important” in that they at least lend some objective data for deciding whether to intervene a group of “challenging” patients not uncommonly faced in clinical practice.
Dr. Alkhouli reports financial relationships with Abbott Vascular, Boston Scientific, Johnson & Johnson, and Phillips. Dr. Asgar reports financial relationships with Abbott Vascular, Edwards Lifesciences, W.L. Gore & Associates, and Medtronic. Dr. Lasky reports no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
In patients with severe mitral regurgitation (MR) and cardiogenic shock, successful transcatheter edge-to-edge repair (TEER) is associated with a substantial reduction in all-cause mortality and lower morbidity at 1 year, according to an analysis of registry data.
The data from this analysis also confirm that “successful reduction of MR is achievable with TEER in most patients with cardiogenic shock,” reported Mohamad A. Alkhouli, MD, an interventional cardiologist and professor of medicine at the Mayo Clinic, Rochester, Minn.
In those with device success, achieved in 85.6% of patients, all-cause mortality was about 21% lower (34.6% vs. 55.5%; P < .001) at 1 year than in those who were not successfully repaired, according to Dr. Alkhouli, who presented the findings at the Transcatheter Cardiovascular Therapeutics annual meeting in Boston. This translated into a reduction in the hazard ratio for death of nearly 50% (hazard ratio, 0.52; 95% confidence interval, 0.43-0.63).
A similar relative benefit was found for the composite endpoint of mortality and heart failure admissions at 1 year. Whether unadjusted (HR, 0.54; 95% CI, 0.45-0.66) or adjusted (HR, 0.51; 95% CI, 0.42-0.62), risk reductions with successful MR reduction, defined as greater than or equal to 1 grade improvement and a final MR grade of less than or equal to 2+, indicated that major adverse outcomes are reduced by about half.
STS/ACC TCT registry data queried
Drawn from the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry, 3,797 patients with cardiogenic shock underwent MR repair between November 2013 and December 2021. Outcomes at 1 year were evaluable in 2,773 of these patients. For inclusion, all had to meet at least one of the definitions of cardiogenic shock, such as inotrope use or mechanical circulatory support.
At baseline, 94.5% had a MR severity of at least 3+, and most of these had 4+. Thirty days after treatment, 88.8% had MR severity of 2+ or less, the majority of which had a severity of 1+.
These data address an important question not previously well studied, according to Dr. Alkhouli. In MR patients, cardiogenic shock is associated with a high risk of death, but there has been little evidence that valve repair does not exacerbate, let alone modify, this risk.
These data support the value of intervention, which was performed in almost all patients with MitraClipä (Abbott), the only device available for most of the period in which the registry was queried. However, Dr. Alkhouli cautioned that his data are best considered “hypothesis generating.”
“We need a randomized trial,” he said at the meeting sponsored by the Cardiovascular Research Foundation. He pointed out that this is a complex population for which multiple variables might have skewed results when data are analyzed retrospectively. Not least, those MR patients with cardiogenic shock in the database considered for TEER might well have been relatively healthy and not representative of an unselected population with both MR and cardiogenic shock.
The question might be better answered by the multicenter Canadian trial CAPITAL MINOS, which has just started. Described in an article in the American Heart Journal, it has a planned enrollment of about 150 MR patients with cardiogenic shock randomized to TEER or medical therapy. Results are expected in about 1 year, according to Dr. Alkhouli.
But regarding the present analysis, Dr. Alkhouli did note that sensitivity analyses conducted within his data across risk factors, such as degenerative versus nondegenerative MR, low (< 30%) versus higher left ventricular ejection fraction (LVEF), and presence or absence of an acute coronary syndrome (ACS), consistently supported a benefit from intervention.
Also, cardiogenic shock did not appear to be a factor in device failure, according to Dr. Alkhouli, addressing a potential criticism that cardiogenic shock was an underlying reason for device failure.
More than 90% in NYHA class III or IV heart failure
In this study, the mean age was 73 years. More than 90% were in class III or IV heart failure in the 2 weeks prior to TEER. More than half had established coronary artery disease. Other concomitant cardiovascular morbidities, including atrial fibrillation or flutter (65%), prior MI (39%), and prior stroke or transient ischemic attach (> 10%) were well represented.
When those with device success were compared with those with device failure, the risk profile was comparable. The predicted STS (Society of Thoracic Surgeons) mortality for mitral valve repair among these two groups was 14.8% versus 15% (P = 0.97), respectively.
However, those with device failure did have a lower baseline left ventricular ejection fraction (40.7% vs. 42.9%; P = .009) and a greater prevalence of moderate-to-severe or severe MR (96.1% vs. 84.9%; P < 0.001).
The growing experience with TEER means that benefit has now been shown in several complicated MR groups, such as those with severe ventricular dysfunction, renal insufficiency, and obstructive lung disease. This was a rationale for looking at the impact or repairing MR in patients with cardiogenic shock.
It is a pressing question, according to Dr. Alkhouli. He cited studies suggesting that up to 20% of patients hospitalized for cardiogenic shock have at least moderate-to-severe MR. Conversely, cardiogenic shock is not an uncommon finding in patients with MR.
While Dr. Alkhouli acknowledged that the many variables influencing outcome in patients with MR and cardiogenic shock will make a randomized trial “challenging,” many experts echoed this concern and even expressed some skepticism about the potential for an unbiased trial.
Data confirm MR repair is safe during shock
“These data do show that repair of MR is safe in patients safe in patients with cardiogenic shock,” said Anita W. Asgar, MD, an interventional cardiologist associated with the Montreal Heart Institute. She noted that there was a 5- to 6-day delay among the cardiogenic shock patients prior to undergoing MR repair in this analysis, potentially reflecting an elimination of those at very high risk. Similarly, she suggested that many interventionalists are likely to consider multiple variables before proceeding.
As a result, MR repair may not be amenable to randomization in a cardiogenic shock population, given that this decision is not typically undertaken out of the context of multiple variables.
“I am not sure that a clinical trial is ethical,” she said. She would expect that clinicians enrolling patients would only do so on a selective basis.
Alexandra J. Lansky, MD, Director of the Yale Heart and Vascular Research Program, Yale University, New Haven, Conn., also emphasized the difficulty of controlling for variables, such as the duration of cardiogenic shock, that influence decision-making.
Nevertheless, she called the data “very important” in that they at least lend some objective data for deciding whether to intervene a group of “challenging” patients not uncommonly faced in clinical practice.
Dr. Alkhouli reports financial relationships with Abbott Vascular, Boston Scientific, Johnson & Johnson, and Phillips. Dr. Asgar reports financial relationships with Abbott Vascular, Edwards Lifesciences, W.L. Gore & Associates, and Medtronic. Dr. Lasky reports no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
In patients with severe mitral regurgitation (MR) and cardiogenic shock, successful transcatheter edge-to-edge repair (TEER) is associated with a substantial reduction in all-cause mortality and lower morbidity at 1 year, according to an analysis of registry data.
The data from this analysis also confirm that “successful reduction of MR is achievable with TEER in most patients with cardiogenic shock,” reported Mohamad A. Alkhouli, MD, an interventional cardiologist and professor of medicine at the Mayo Clinic, Rochester, Minn.
In those with device success, achieved in 85.6% of patients, all-cause mortality was about 21% lower (34.6% vs. 55.5%; P < .001) at 1 year than in those who were not successfully repaired, according to Dr. Alkhouli, who presented the findings at the Transcatheter Cardiovascular Therapeutics annual meeting in Boston. This translated into a reduction in the hazard ratio for death of nearly 50% (hazard ratio, 0.52; 95% confidence interval, 0.43-0.63).
A similar relative benefit was found for the composite endpoint of mortality and heart failure admissions at 1 year. Whether unadjusted (HR, 0.54; 95% CI, 0.45-0.66) or adjusted (HR, 0.51; 95% CI, 0.42-0.62), risk reductions with successful MR reduction, defined as greater than or equal to 1 grade improvement and a final MR grade of less than or equal to 2+, indicated that major adverse outcomes are reduced by about half.
STS/ACC TCT registry data queried
Drawn from the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry, 3,797 patients with cardiogenic shock underwent MR repair between November 2013 and December 2021. Outcomes at 1 year were evaluable in 2,773 of these patients. For inclusion, all had to meet at least one of the definitions of cardiogenic shock, such as inotrope use or mechanical circulatory support.
At baseline, 94.5% had a MR severity of at least 3+, and most of these had 4+. Thirty days after treatment, 88.8% had MR severity of 2+ or less, the majority of which had a severity of 1+.
These data address an important question not previously well studied, according to Dr. Alkhouli. In MR patients, cardiogenic shock is associated with a high risk of death, but there has been little evidence that valve repair does not exacerbate, let alone modify, this risk.
These data support the value of intervention, which was performed in almost all patients with MitraClipä (Abbott), the only device available for most of the period in which the registry was queried. However, Dr. Alkhouli cautioned that his data are best considered “hypothesis generating.”
“We need a randomized trial,” he said at the meeting sponsored by the Cardiovascular Research Foundation. He pointed out that this is a complex population for which multiple variables might have skewed results when data are analyzed retrospectively. Not least, those MR patients with cardiogenic shock in the database considered for TEER might well have been relatively healthy and not representative of an unselected population with both MR and cardiogenic shock.
The question might be better answered by the multicenter Canadian trial CAPITAL MINOS, which has just started. Described in an article in the American Heart Journal, it has a planned enrollment of about 150 MR patients with cardiogenic shock randomized to TEER or medical therapy. Results are expected in about 1 year, according to Dr. Alkhouli.
But regarding the present analysis, Dr. Alkhouli did note that sensitivity analyses conducted within his data across risk factors, such as degenerative versus nondegenerative MR, low (< 30%) versus higher left ventricular ejection fraction (LVEF), and presence or absence of an acute coronary syndrome (ACS), consistently supported a benefit from intervention.
Also, cardiogenic shock did not appear to be a factor in device failure, according to Dr. Alkhouli, addressing a potential criticism that cardiogenic shock was an underlying reason for device failure.
More than 90% in NYHA class III or IV heart failure
In this study, the mean age was 73 years. More than 90% were in class III or IV heart failure in the 2 weeks prior to TEER. More than half had established coronary artery disease. Other concomitant cardiovascular morbidities, including atrial fibrillation or flutter (65%), prior MI (39%), and prior stroke or transient ischemic attach (> 10%) were well represented.
When those with device success were compared with those with device failure, the risk profile was comparable. The predicted STS (Society of Thoracic Surgeons) mortality for mitral valve repair among these two groups was 14.8% versus 15% (P = 0.97), respectively.
However, those with device failure did have a lower baseline left ventricular ejection fraction (40.7% vs. 42.9%; P = .009) and a greater prevalence of moderate-to-severe or severe MR (96.1% vs. 84.9%; P < 0.001).
The growing experience with TEER means that benefit has now been shown in several complicated MR groups, such as those with severe ventricular dysfunction, renal insufficiency, and obstructive lung disease. This was a rationale for looking at the impact or repairing MR in patients with cardiogenic shock.
It is a pressing question, according to Dr. Alkhouli. He cited studies suggesting that up to 20% of patients hospitalized for cardiogenic shock have at least moderate-to-severe MR. Conversely, cardiogenic shock is not an uncommon finding in patients with MR.
While Dr. Alkhouli acknowledged that the many variables influencing outcome in patients with MR and cardiogenic shock will make a randomized trial “challenging,” many experts echoed this concern and even expressed some skepticism about the potential for an unbiased trial.
Data confirm MR repair is safe during shock
“These data do show that repair of MR is safe in patients safe in patients with cardiogenic shock,” said Anita W. Asgar, MD, an interventional cardiologist associated with the Montreal Heart Institute. She noted that there was a 5- to 6-day delay among the cardiogenic shock patients prior to undergoing MR repair in this analysis, potentially reflecting an elimination of those at very high risk. Similarly, she suggested that many interventionalists are likely to consider multiple variables before proceeding.
As a result, MR repair may not be amenable to randomization in a cardiogenic shock population, given that this decision is not typically undertaken out of the context of multiple variables.
“I am not sure that a clinical trial is ethical,” she said. She would expect that clinicians enrolling patients would only do so on a selective basis.
Alexandra J. Lansky, MD, Director of the Yale Heart and Vascular Research Program, Yale University, New Haven, Conn., also emphasized the difficulty of controlling for variables, such as the duration of cardiogenic shock, that influence decision-making.
Nevertheless, she called the data “very important” in that they at least lend some objective data for deciding whether to intervene a group of “challenging” patients not uncommonly faced in clinical practice.
Dr. Alkhouli reports financial relationships with Abbott Vascular, Boston Scientific, Johnson & Johnson, and Phillips. Dr. Asgar reports financial relationships with Abbott Vascular, Edwards Lifesciences, W.L. Gore & Associates, and Medtronic. Dr. Lasky reports no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
Fourth-gen transcatheter mitral valve shows clinical, procedural improvements
The design improvements introduced in the fourth-generation device for transcatheter mitral valve repair, called the MitraClip G4 (Abbott), appears to yield better outcomes than previous iterations, according to a multinational postapproval study with more than 1,000 patients.
Not least, the 1.3% all-cause mortality at 30 days in this series, called EXPAND G4, “is the lowest that has been reported to date,” reported Ralph Stephan von Bardeleben, MD, at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
The evidence of relative advantages was based on comparisons with historical data and a similar study of the previous-generation device. That previous study, called EXPAND, evaluated the MitraClip NTR and ETR systems.
Device times shorter with new device
“There were shorter device times with MitraClip G4,” said Dr. von Bardeleben, referring to a more than 10-minute advantage over the previous generation device (35 minutes in EXPAND G4 vs. 46 min in EXPAND). Although the reduction in overall median procedure time was more modest (77 vs. 80 minutes), Dr. von Bardeleben said these are “the shortest device and procedural times reported to date.”
He also reported what appeared to be incremental advantages across multiple other endpoints, such as procedural success (96.2% vs. 95.8%) and a reduction in the mean clip rate (1.4 vs. 1.5).
Compared with historical outcomes with other devices employed in transcatheter edge-to-edge repair (TEER) of mitral valves, Dr. von Bardeleben contended that the results support the premise that the MitraClip G4 system is a meaningful advance by incorporating such features as an expanded choice of clip sizes, a greater coaptation area, and a more advanced gripper actuation for leaflet grasping.
Over 90% achieve MR 1+
Not least, it appears to increase the proportion of patients who achieve a mitral regurgitation grade of 1+ (MR1+) or lower, which is increasingly recognized as the goal of TEER, said Dr. von Bardeleben, head of the Centre of Structure Heart Disease Interventions, Heart Valve Centre, Mainz, Germany.
He said the rates of 91% achieving MR1+ or less and 98% achieved 2+ or lower compare favorably with most other series and exceeds levels achieved with surgery.
Dr. von Bardeleben also contended that, because of its design features, the MitraClip G4 “expands the spectrum of TEER-suitable patients.” He noted that 5% of the patients in this real-world series had a high risk of stenosis owing to such issues as severe annular or leaflet calcification and another 5% had factors that would predict inadequate MR reduction, such as Barlow’s disease, bi-leaflet prolapse, and severe leaflet degeneration.
The 1,164 patients in EXPAND G4 were enrolled from sites in the United States, Europe, Canada, and Japan. For the key outcome measure of procedural success, echocardiograms were assessed by an independent core laboratory. Of the 1,164 patients enrolled, 1,044 (91%) had complete follow-up data at 30 days.
The procedural success rates were reflected in improvements in New York Heart Association (NYHA) functional classes and in the Kansas City Cardiomyopathy Questionnaire (KCCQ), a quality of life instrument. Prior to treatment, 69% were in NYHA class III or greater. Following treatment, the proportion was 17% (P < .0001). The 18-point improvement in the KCCQ was characterized by Dr. von Bardeleben as “both clinically and statistically significant [P < .0001].”
There were no strokes in this series, and the 30-day incidence of myocardial infarction was 0.2%. The proportion requiring cardiovascular surgery within 30 days was less than 1%. The rate of bleeding episodes, all of which were nonserious, was 7%.
The “EXPAND G4 study confirms the safety and effectiveness of the next generation MitraClip G4 system,” according to Dr. von Bardeleben, and it did so “in a contemporary real-world setting.”
Outcome data characterized as ‘excellent’
Several invited panelists participating in a discussion following the presentation agreed.
“These results are excellent,” said Raj Makkar, MD, associate director of interventional technologies at Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles. While he was impressed with the fact that only 2% missed the primary endpoint of MR 2+ or lower, he indicated that the 91% achieving MR 1+ or lower might be an even more apt signal that newer-generation devices are improving.
This was echoed by other panelists who appeared to form a general consensus over the premise that the target in TEER should no longer be MR 2+ for most patients.
“We should now be aiming for MR grade of 0-1,” stated panelist Stephan Windecker, MD, chairman, department of cardiology, University of Bern (Switzerland). He indicated that this goal is increasingly reasonable given the advances in device design and greater operator experience.
Dr. von Bardeleben reported financial relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, and Neochord. Dr. Makkar reported financial relationships with Abbott Vascular, Cordis, Edwards Lifesciences, and Medtronic. Dr. Windecker reported financial relationships with more than 30 pharmaceutical companies, including Abbott Vascular, which manufactures MitraClip G4.
The design improvements introduced in the fourth-generation device for transcatheter mitral valve repair, called the MitraClip G4 (Abbott), appears to yield better outcomes than previous iterations, according to a multinational postapproval study with more than 1,000 patients.
Not least, the 1.3% all-cause mortality at 30 days in this series, called EXPAND G4, “is the lowest that has been reported to date,” reported Ralph Stephan von Bardeleben, MD, at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
The evidence of relative advantages was based on comparisons with historical data and a similar study of the previous-generation device. That previous study, called EXPAND, evaluated the MitraClip NTR and ETR systems.
Device times shorter with new device
“There were shorter device times with MitraClip G4,” said Dr. von Bardeleben, referring to a more than 10-minute advantage over the previous generation device (35 minutes in EXPAND G4 vs. 46 min in EXPAND). Although the reduction in overall median procedure time was more modest (77 vs. 80 minutes), Dr. von Bardeleben said these are “the shortest device and procedural times reported to date.”
He also reported what appeared to be incremental advantages across multiple other endpoints, such as procedural success (96.2% vs. 95.8%) and a reduction in the mean clip rate (1.4 vs. 1.5).
Compared with historical outcomes with other devices employed in transcatheter edge-to-edge repair (TEER) of mitral valves, Dr. von Bardeleben contended that the results support the premise that the MitraClip G4 system is a meaningful advance by incorporating such features as an expanded choice of clip sizes, a greater coaptation area, and a more advanced gripper actuation for leaflet grasping.
Over 90% achieve MR 1+
Not least, it appears to increase the proportion of patients who achieve a mitral regurgitation grade of 1+ (MR1+) or lower, which is increasingly recognized as the goal of TEER, said Dr. von Bardeleben, head of the Centre of Structure Heart Disease Interventions, Heart Valve Centre, Mainz, Germany.
He said the rates of 91% achieving MR1+ or less and 98% achieved 2+ or lower compare favorably with most other series and exceeds levels achieved with surgery.
Dr. von Bardeleben also contended that, because of its design features, the MitraClip G4 “expands the spectrum of TEER-suitable patients.” He noted that 5% of the patients in this real-world series had a high risk of stenosis owing to such issues as severe annular or leaflet calcification and another 5% had factors that would predict inadequate MR reduction, such as Barlow’s disease, bi-leaflet prolapse, and severe leaflet degeneration.
The 1,164 patients in EXPAND G4 were enrolled from sites in the United States, Europe, Canada, and Japan. For the key outcome measure of procedural success, echocardiograms were assessed by an independent core laboratory. Of the 1,164 patients enrolled, 1,044 (91%) had complete follow-up data at 30 days.
The procedural success rates were reflected in improvements in New York Heart Association (NYHA) functional classes and in the Kansas City Cardiomyopathy Questionnaire (KCCQ), a quality of life instrument. Prior to treatment, 69% were in NYHA class III or greater. Following treatment, the proportion was 17% (P < .0001). The 18-point improvement in the KCCQ was characterized by Dr. von Bardeleben as “both clinically and statistically significant [P < .0001].”
There were no strokes in this series, and the 30-day incidence of myocardial infarction was 0.2%. The proportion requiring cardiovascular surgery within 30 days was less than 1%. The rate of bleeding episodes, all of which were nonserious, was 7%.
The “EXPAND G4 study confirms the safety and effectiveness of the next generation MitraClip G4 system,” according to Dr. von Bardeleben, and it did so “in a contemporary real-world setting.”
Outcome data characterized as ‘excellent’
Several invited panelists participating in a discussion following the presentation agreed.
“These results are excellent,” said Raj Makkar, MD, associate director of interventional technologies at Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles. While he was impressed with the fact that only 2% missed the primary endpoint of MR 2+ or lower, he indicated that the 91% achieving MR 1+ or lower might be an even more apt signal that newer-generation devices are improving.
This was echoed by other panelists who appeared to form a general consensus over the premise that the target in TEER should no longer be MR 2+ for most patients.
“We should now be aiming for MR grade of 0-1,” stated panelist Stephan Windecker, MD, chairman, department of cardiology, University of Bern (Switzerland). He indicated that this goal is increasingly reasonable given the advances in device design and greater operator experience.
Dr. von Bardeleben reported financial relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, and Neochord. Dr. Makkar reported financial relationships with Abbott Vascular, Cordis, Edwards Lifesciences, and Medtronic. Dr. Windecker reported financial relationships with more than 30 pharmaceutical companies, including Abbott Vascular, which manufactures MitraClip G4.
The design improvements introduced in the fourth-generation device for transcatheter mitral valve repair, called the MitraClip G4 (Abbott), appears to yield better outcomes than previous iterations, according to a multinational postapproval study with more than 1,000 patients.
Not least, the 1.3% all-cause mortality at 30 days in this series, called EXPAND G4, “is the lowest that has been reported to date,” reported Ralph Stephan von Bardeleben, MD, at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
The evidence of relative advantages was based on comparisons with historical data and a similar study of the previous-generation device. That previous study, called EXPAND, evaluated the MitraClip NTR and ETR systems.
Device times shorter with new device
“There were shorter device times with MitraClip G4,” said Dr. von Bardeleben, referring to a more than 10-minute advantage over the previous generation device (35 minutes in EXPAND G4 vs. 46 min in EXPAND). Although the reduction in overall median procedure time was more modest (77 vs. 80 minutes), Dr. von Bardeleben said these are “the shortest device and procedural times reported to date.”
He also reported what appeared to be incremental advantages across multiple other endpoints, such as procedural success (96.2% vs. 95.8%) and a reduction in the mean clip rate (1.4 vs. 1.5).
Compared with historical outcomes with other devices employed in transcatheter edge-to-edge repair (TEER) of mitral valves, Dr. von Bardeleben contended that the results support the premise that the MitraClip G4 system is a meaningful advance by incorporating such features as an expanded choice of clip sizes, a greater coaptation area, and a more advanced gripper actuation for leaflet grasping.
Over 90% achieve MR 1+
Not least, it appears to increase the proportion of patients who achieve a mitral regurgitation grade of 1+ (MR1+) or lower, which is increasingly recognized as the goal of TEER, said Dr. von Bardeleben, head of the Centre of Structure Heart Disease Interventions, Heart Valve Centre, Mainz, Germany.
He said the rates of 91% achieving MR1+ or less and 98% achieved 2+ or lower compare favorably with most other series and exceeds levels achieved with surgery.
Dr. von Bardeleben also contended that, because of its design features, the MitraClip G4 “expands the spectrum of TEER-suitable patients.” He noted that 5% of the patients in this real-world series had a high risk of stenosis owing to such issues as severe annular or leaflet calcification and another 5% had factors that would predict inadequate MR reduction, such as Barlow’s disease, bi-leaflet prolapse, and severe leaflet degeneration.
The 1,164 patients in EXPAND G4 were enrolled from sites in the United States, Europe, Canada, and Japan. For the key outcome measure of procedural success, echocardiograms were assessed by an independent core laboratory. Of the 1,164 patients enrolled, 1,044 (91%) had complete follow-up data at 30 days.
The procedural success rates were reflected in improvements in New York Heart Association (NYHA) functional classes and in the Kansas City Cardiomyopathy Questionnaire (KCCQ), a quality of life instrument. Prior to treatment, 69% were in NYHA class III or greater. Following treatment, the proportion was 17% (P < .0001). The 18-point improvement in the KCCQ was characterized by Dr. von Bardeleben as “both clinically and statistically significant [P < .0001].”
There were no strokes in this series, and the 30-day incidence of myocardial infarction was 0.2%. The proportion requiring cardiovascular surgery within 30 days was less than 1%. The rate of bleeding episodes, all of which were nonserious, was 7%.
The “EXPAND G4 study confirms the safety and effectiveness of the next generation MitraClip G4 system,” according to Dr. von Bardeleben, and it did so “in a contemporary real-world setting.”
Outcome data characterized as ‘excellent’
Several invited panelists participating in a discussion following the presentation agreed.
“These results are excellent,” said Raj Makkar, MD, associate director of interventional technologies at Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles. While he was impressed with the fact that only 2% missed the primary endpoint of MR 2+ or lower, he indicated that the 91% achieving MR 1+ or lower might be an even more apt signal that newer-generation devices are improving.
This was echoed by other panelists who appeared to form a general consensus over the premise that the target in TEER should no longer be MR 2+ for most patients.
“We should now be aiming for MR grade of 0-1,” stated panelist Stephan Windecker, MD, chairman, department of cardiology, University of Bern (Switzerland). He indicated that this goal is increasingly reasonable given the advances in device design and greater operator experience.
Dr. von Bardeleben reported financial relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, and Neochord. Dr. Makkar reported financial relationships with Abbott Vascular, Cordis, Edwards Lifesciences, and Medtronic. Dr. Windecker reported financial relationships with more than 30 pharmaceutical companies, including Abbott Vascular, which manufactures MitraClip G4.
FROM TCT 2022
Physicians can’t be bystanders in ‘silent scourge’ of medical bullying
Maya Iyer, MD, MEd, experienced bullying as a faculty member, and she sensed that she wasn’t alone. “The best ideas for research often come from individual experiences, in both personal and the professional academic medicine setting,” she said in an interview.
“And I was correct. I was not the only one who experienced bullying. In fact, the most severe bullying experiences among ... women physician leaders occurred when they were in leadership positions,” said Dr. Iyer, a pediatric emergency medicine physician at Nationwide Children’s Hospital in Columbus, Ohio.
She is a coauthor of a study that was published in JAMA Network Open in which investigators surveyed the existence of antibullying policies for faculty at almost 100 U.S. medical schools.
The researchers defined bullying as “a severe form of mistreatment [that] occurs in the medical setting when a power differential allows offenders to consciously target individuals through persistent negative actions to impede the education or career of the target.”
The study included 91 medical schools, of which 4 schools had antibullying policies that included the reporting of procedures. Of the 87 medical schools without antibullying policies, 60 had antiharrassment policies; of those schools, 10 of the schools’ websites cited bullying and antiharassment policies. Five schools required a login to access policies, and one school’s website had a broken webpage link, per the study.
“We need to bring the silent scourge of bullying to the forefront because bullying is causing a brain drain on the medical profession,” said Dr. Iyer. “Bullying has numerous downstream negative effects, including depression, anxiety, burnout stress, decreased patient care satisfaction, increased medical errors, and job attrition.”
She added: “Through bullying, we are losing voices in medicine just at that point in time where we are trying to diversify the workforce to improve representation of all physicians.”
Dr. Iyer’s team sampled the top 25 schools for research and the top 25 schools for primary care. They also took a random sampling from 25 schools for research and a random sampling from top 25 schools for primary care. They assessed antibullying policies, antiharassment policies that mentioned bullying, antiharrassment policies that did not mention bullying, and the absence of policies addressing these issues.
Policy comprehensiveness was another focus for the researchers. They evaluated whether the relevant policies included faculty members and articulated the institution’s commitment to providing a safe and healthy workplace. Other factors included defining bullying and the roles and responsibilities of employees and procedures for reporting bullying.
Physicians can’t be bystanders to bullying
This means transitioning from being a bystander to an upstander.”
She doesn’t let medical schools off the hook, however. Instead, she advocated having institutions “provide safe spaces and opportunities for near-peer mentoring so that targets of bullying can share stories.”
Regarding who is responsible for addressing bullying, Dr. Iyer is emphatic. “I do want to be clear that the onus of disrupting does not fall on the targets. Rather, we need to fix the systems in which such behavior is tolerated.”
Her advice to leaders in academic medicine is to create comprehensive, zero-retaliation bullying policies that include detailed reporting procedures. Dr. Iyer advised leaders to partner with colleagues in human resources, offices of equity, and ombudspersons to develop, implement, and enforce these policies.
The study authors reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Maya Iyer, MD, MEd, experienced bullying as a faculty member, and she sensed that she wasn’t alone. “The best ideas for research often come from individual experiences, in both personal and the professional academic medicine setting,” she said in an interview.
“And I was correct. I was not the only one who experienced bullying. In fact, the most severe bullying experiences among ... women physician leaders occurred when they were in leadership positions,” said Dr. Iyer, a pediatric emergency medicine physician at Nationwide Children’s Hospital in Columbus, Ohio.
She is a coauthor of a study that was published in JAMA Network Open in which investigators surveyed the existence of antibullying policies for faculty at almost 100 U.S. medical schools.
The researchers defined bullying as “a severe form of mistreatment [that] occurs in the medical setting when a power differential allows offenders to consciously target individuals through persistent negative actions to impede the education or career of the target.”
The study included 91 medical schools, of which 4 schools had antibullying policies that included the reporting of procedures. Of the 87 medical schools without antibullying policies, 60 had antiharrassment policies; of those schools, 10 of the schools’ websites cited bullying and antiharassment policies. Five schools required a login to access policies, and one school’s website had a broken webpage link, per the study.
“We need to bring the silent scourge of bullying to the forefront because bullying is causing a brain drain on the medical profession,” said Dr. Iyer. “Bullying has numerous downstream negative effects, including depression, anxiety, burnout stress, decreased patient care satisfaction, increased medical errors, and job attrition.”
She added: “Through bullying, we are losing voices in medicine just at that point in time where we are trying to diversify the workforce to improve representation of all physicians.”
Dr. Iyer’s team sampled the top 25 schools for research and the top 25 schools for primary care. They also took a random sampling from 25 schools for research and a random sampling from top 25 schools for primary care. They assessed antibullying policies, antiharassment policies that mentioned bullying, antiharrassment policies that did not mention bullying, and the absence of policies addressing these issues.
Policy comprehensiveness was another focus for the researchers. They evaluated whether the relevant policies included faculty members and articulated the institution’s commitment to providing a safe and healthy workplace. Other factors included defining bullying and the roles and responsibilities of employees and procedures for reporting bullying.
Physicians can’t be bystanders to bullying
This means transitioning from being a bystander to an upstander.”
She doesn’t let medical schools off the hook, however. Instead, she advocated having institutions “provide safe spaces and opportunities for near-peer mentoring so that targets of bullying can share stories.”
Regarding who is responsible for addressing bullying, Dr. Iyer is emphatic. “I do want to be clear that the onus of disrupting does not fall on the targets. Rather, we need to fix the systems in which such behavior is tolerated.”
Her advice to leaders in academic medicine is to create comprehensive, zero-retaliation bullying policies that include detailed reporting procedures. Dr. Iyer advised leaders to partner with colleagues in human resources, offices of equity, and ombudspersons to develop, implement, and enforce these policies.
The study authors reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Maya Iyer, MD, MEd, experienced bullying as a faculty member, and she sensed that she wasn’t alone. “The best ideas for research often come from individual experiences, in both personal and the professional academic medicine setting,” she said in an interview.
“And I was correct. I was not the only one who experienced bullying. In fact, the most severe bullying experiences among ... women physician leaders occurred when they were in leadership positions,” said Dr. Iyer, a pediatric emergency medicine physician at Nationwide Children’s Hospital in Columbus, Ohio.
She is a coauthor of a study that was published in JAMA Network Open in which investigators surveyed the existence of antibullying policies for faculty at almost 100 U.S. medical schools.
The researchers defined bullying as “a severe form of mistreatment [that] occurs in the medical setting when a power differential allows offenders to consciously target individuals through persistent negative actions to impede the education or career of the target.”
The study included 91 medical schools, of which 4 schools had antibullying policies that included the reporting of procedures. Of the 87 medical schools without antibullying policies, 60 had antiharrassment policies; of those schools, 10 of the schools’ websites cited bullying and antiharassment policies. Five schools required a login to access policies, and one school’s website had a broken webpage link, per the study.
“We need to bring the silent scourge of bullying to the forefront because bullying is causing a brain drain on the medical profession,” said Dr. Iyer. “Bullying has numerous downstream negative effects, including depression, anxiety, burnout stress, decreased patient care satisfaction, increased medical errors, and job attrition.”
She added: “Through bullying, we are losing voices in medicine just at that point in time where we are trying to diversify the workforce to improve representation of all physicians.”
Dr. Iyer’s team sampled the top 25 schools for research and the top 25 schools for primary care. They also took a random sampling from 25 schools for research and a random sampling from top 25 schools for primary care. They assessed antibullying policies, antiharassment policies that mentioned bullying, antiharrassment policies that did not mention bullying, and the absence of policies addressing these issues.
Policy comprehensiveness was another focus for the researchers. They evaluated whether the relevant policies included faculty members and articulated the institution’s commitment to providing a safe and healthy workplace. Other factors included defining bullying and the roles and responsibilities of employees and procedures for reporting bullying.
Physicians can’t be bystanders to bullying
This means transitioning from being a bystander to an upstander.”
She doesn’t let medical schools off the hook, however. Instead, she advocated having institutions “provide safe spaces and opportunities for near-peer mentoring so that targets of bullying can share stories.”
Regarding who is responsible for addressing bullying, Dr. Iyer is emphatic. “I do want to be clear that the onus of disrupting does not fall on the targets. Rather, we need to fix the systems in which such behavior is tolerated.”
Her advice to leaders in academic medicine is to create comprehensive, zero-retaliation bullying policies that include detailed reporting procedures. Dr. Iyer advised leaders to partner with colleagues in human resources, offices of equity, and ombudspersons to develop, implement, and enforce these policies.
The study authors reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Advances in Diabetes and Heart Disease From ESC 2022
Professor Pardeep Jhund, from the University of Glasgow, United Kingdom, discusses highlights in diabetes and heart disease presented at the European Society of Cardiology Congress 2022.
He begins with a Danish study of the causes of excess mortality in individuals who have diabetes but do not have coronary artery disease. This showed that, even in these patients, greater efforts are required to improve outcomes.
A second Danish epidemiologic study examines the prevalence of diabetic neuropathy and cardiovascular outcomes. Worryingly, it found that around half of patients did not have their urinary albumin measured.
Next, Prof Jhund examines a follow-up analysis of the DANISH trial, which showed that implantable cardioverter-defibrillators were effective in reducing mortality, but only in individuals without diabetes.
He moves on to a pooled analysis of DECLARE-TIMI 58 and DAPA-CKD, which revealed that dapagliflozin consistently reduced kidney and cardiovascular events regardless of baseline eGFR and urinary albumin.
Finally, he discusses a large study addressing adherence rates for drugs in type 2 diabetes. The data on more than 38,000 patients showed that initiating therapy with an SGLT2 inhibitor or a GLP-1R agonist was associated with comparably high rates of adherence, which were unaffected by the presence or absence of cardiovascular disease.
--
Professor of Cardiology and Epidemiology, University of Glasgow; Honorary Consultant Cardiologist, Queen Elizabeth University Hospital, Glasgow, Scotland
Pardeep Jhund, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: Global Clinical Trial Partners
Serve(d) as a speaker or a member of a speakers bureau for: AstraZeneca; Novartis
Received research grant from: AstraZeneca; Boehringer Ingelheim; Analog Devices
Professor Pardeep Jhund, from the University of Glasgow, United Kingdom, discusses highlights in diabetes and heart disease presented at the European Society of Cardiology Congress 2022.
He begins with a Danish study of the causes of excess mortality in individuals who have diabetes but do not have coronary artery disease. This showed that, even in these patients, greater efforts are required to improve outcomes.
A second Danish epidemiologic study examines the prevalence of diabetic neuropathy and cardiovascular outcomes. Worryingly, it found that around half of patients did not have their urinary albumin measured.
Next, Prof Jhund examines a follow-up analysis of the DANISH trial, which showed that implantable cardioverter-defibrillators were effective in reducing mortality, but only in individuals without diabetes.
He moves on to a pooled analysis of DECLARE-TIMI 58 and DAPA-CKD, which revealed that dapagliflozin consistently reduced kidney and cardiovascular events regardless of baseline eGFR and urinary albumin.
Finally, he discusses a large study addressing adherence rates for drugs in type 2 diabetes. The data on more than 38,000 patients showed that initiating therapy with an SGLT2 inhibitor or a GLP-1R agonist was associated with comparably high rates of adherence, which were unaffected by the presence or absence of cardiovascular disease.
--
Professor of Cardiology and Epidemiology, University of Glasgow; Honorary Consultant Cardiologist, Queen Elizabeth University Hospital, Glasgow, Scotland
Pardeep Jhund, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: Global Clinical Trial Partners
Serve(d) as a speaker or a member of a speakers bureau for: AstraZeneca; Novartis
Received research grant from: AstraZeneca; Boehringer Ingelheim; Analog Devices
Professor Pardeep Jhund, from the University of Glasgow, United Kingdom, discusses highlights in diabetes and heart disease presented at the European Society of Cardiology Congress 2022.
He begins with a Danish study of the causes of excess mortality in individuals who have diabetes but do not have coronary artery disease. This showed that, even in these patients, greater efforts are required to improve outcomes.
A second Danish epidemiologic study examines the prevalence of diabetic neuropathy and cardiovascular outcomes. Worryingly, it found that around half of patients did not have their urinary albumin measured.
Next, Prof Jhund examines a follow-up analysis of the DANISH trial, which showed that implantable cardioverter-defibrillators were effective in reducing mortality, but only in individuals without diabetes.
He moves on to a pooled analysis of DECLARE-TIMI 58 and DAPA-CKD, which revealed that dapagliflozin consistently reduced kidney and cardiovascular events regardless of baseline eGFR and urinary albumin.
Finally, he discusses a large study addressing adherence rates for drugs in type 2 diabetes. The data on more than 38,000 patients showed that initiating therapy with an SGLT2 inhibitor or a GLP-1R agonist was associated with comparably high rates of adherence, which were unaffected by the presence or absence of cardiovascular disease.
--
Professor of Cardiology and Epidemiology, University of Glasgow; Honorary Consultant Cardiologist, Queen Elizabeth University Hospital, Glasgow, Scotland
Pardeep Jhund, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: Global Clinical Trial Partners
Serve(d) as a speaker or a member of a speakers bureau for: AstraZeneca; Novartis
Received research grant from: AstraZeneca; Boehringer Ingelheim; Analog Devices

Experts express caution over type 2 diabetes/tea-drinking claim
type 2 diabetes/tea-drinking claim
A claim that drinking tea might protect people against developing type 2 diabetes has been met with caution from multiple experts ahead of the annual meeting of the European Association for the Study of Diabetes.
The claim is that people who drink four or more cups of tea every day – specifically green, Oolong, or black tea – are 17% less likely to develop type 2 diabetes than those who do not drink tea. Drinking fewer cups of tea per day was not found to confer any benefit.
“Our results are exciting because they suggest that people can do something as simple as drinking four cups of tea a day to potentially lessen their risk of developing type 2 diabetes,” Xiaying Li of Wuhan (China) University of Science and Technology is quoted as saying in an official EASD press release.
“It is possible that particular components in tea, such as polyphenols, may reduce blood glucose levels, but a sufficient amount of these bioactive compounds may be needed to be effective,” Dr. Li added.
“The words ‘suggest’ and ‘potentially’ are crucial here,” said Kevin McConway, PhD, MSc, MBA, emeritus professor of applied statistics at The Open University, said in a separate statement to the press that reeled in Dr. Li’s enthusiasm.
“Tea drinking would only be useful for reducing diabetes risk if the tea drinking causes reductions in risk, that is, if the risk is reduced if you drink the tea and not if you don’t – and this study simply can’t show whether it does this or not,” Dr. Conway stressed.
Naveed Sattar, FMedSci FRCPath FRCPGlas FRSE, professor of metabolic medicine at the University of Glasgow, was also cautiously critical. “There is no good trial evidence whatsoever that the chemicals in tea prevent diabetes,” he observed separately.
“So, I suspect its more about tea being healthier (less calorific) than many alternative drinks or tea drinkers leading healthier lives more generally.”
Dr. Sattar added that it could be that people who drink tea might also be avoiding drinking more harmful sugary drinks and have other health behaviors that might lead them to have a lower risk for type 2 diabetes.
Time for tea?
Dr. Li will present the findings of two analyses on Sept. 21 at the EASD meeting: the first a large observational cohort study and the second an updated systematic review and meta-analysis.
For the cohort study, Dr. Li and her coauthors took data on more than 5,100 adults who had participated in the long-running and ongoing China Health and Nutrition Survey (CHNS). Information on tea drinking behavior was extracted from questionnaires that had been filled out at two time points – 1997 and 2009 – and they determined whether people had developed type 2 diabetes according to American Diabetes Association criteria.
Nearly half, 45.8%, were found to be tea drinkers, and 10% of the population they sampled had developed type 2 diabetes. No association between tea drinking and type 2 diabetes development was found, however, with the hazard ratio comparing tea drinkers and non–tea drinkers sitting firmly at 1.02. Moreover, a sensitivity analysis that excluded participants who had developed type 2 diabetes in the first 3 years of follow-up did not change the result.
Things were slightly different when Dr. Li and associates performed their meta-analysis that involved analyzing data on more than 1 million participants in 19 studies conducted in eight countries that had been published up to September 2021.
Here, they found there was a significant (P < .003) linear association between tea consumption and having type 2 diabetes, with the relative risk of developing type 2 diabetes decreasing by 0.986 for every additional cup of tea that was drunk.
HRs for the development of type 2 diabetes in tea drinkers versus non–tea drinkers were 1.00 for those who drank less than one cup per day, 0.96 for those who had one to two cups, and 0.84 for those who drank four or more cups.
“While more research needs to be done to determine the exact dosage and mechanisms behind these observations, our findings suggest that drinking tea is beneficial in reducing the risk of type 2 diabetes, but only at high doses (at least 4 cups a day)”, said Dr. Li.
Perhaps, “we did not find an association between tea drinking and type 2 diabetes in our cohort study because we did not look at higher tea consumption,” she added.
Tempest in a teacup
“This is large, observational data. It’s not a randomized controlled trial so there’s plenty of room for data to be misunderstood,” warned Matt Sydes, MSc, professor of clinical trials & methodology at the MRC Clinical Trials Unit, University College London.
“Everyone drinks fluids. If there is an effect here (and that’s a big if), it might be not about the tea they drink, but about what they don’t drink. One can’t tell at the moment. It seems unlikely that a large randomized controlled trial could be done to disambiguate” added Dr. Sydes
“Being only a conference abstract, it is difficult to assess the quality of this research,” Baptiste Leurent, PhD, a medical statistician also working at University College London, said. Not only was the cohort study observational, so were all the other studies included in the meta-analysis, he pointed out.
“Therefore, no cause-effect conclusions can be drawn. The association could simply be due to other factors, such as those drinking more tea having a healthier lifestyle. It does not seem that the authors tried to control for confounders, which is usually difficult in meta-analysis,” Dr. Leurent said.
“There is reason to be a bit skeptical at this point; we really need to have the full details to assess it properly,” said Jonathan Cook of the Centre for Statistics in Medicine at the University of Oxford (England). “It’s a fair attempt to look at this, but not cutting edge, [using] fairly standard approaches.”
Similar studies have shown a reduced risk associated with coffee drinking, noted Duane Mellor, PhD, a registered dietitian and senior teaching fellow at Aston University in Birmingham.
“The important take-home message is that lifestyle is important in managing risk of developing type 2 diabetes,” Dr. Mellor said.
“That includes choosing low-calorie drinks including mainly water as well as unsweetened tea and coffee as your drinks of choice as part of a healthy lifestyle.”
The study was funded by the Young Talents Project of Hubei Provincial Health Commission, the Science and Technology Research Key Project of Education Department of Hubei Province, the Sanuo Diabetes Charity Foundation, and the Xiangyang Science and Technology Plan Project, all based in China. Dr. Li had no conflicts of interest to disclose. Dr. McConway is a Trustee and on the advisory committee of The Science Media Centre. Dr. Sattar has consulted for many companies that make diabetes and cardiovascular drugs and has been involved in multiple trials of lifestyle approaches for the prevention and remission of diabetes. Dr. Sydes, Dr. Leurent, Dr. Cook, and Dr. Mellor had no conflicts of interest to report.
A claim that drinking tea might protect people against developing type 2 diabetes has been met with caution from multiple experts ahead of the annual meeting of the European Association for the Study of Diabetes.
The claim is that people who drink four or more cups of tea every day – specifically green, Oolong, or black tea – are 17% less likely to develop type 2 diabetes than those who do not drink tea. Drinking fewer cups of tea per day was not found to confer any benefit.
“Our results are exciting because they suggest that people can do something as simple as drinking four cups of tea a day to potentially lessen their risk of developing type 2 diabetes,” Xiaying Li of Wuhan (China) University of Science and Technology is quoted as saying in an official EASD press release.
“It is possible that particular components in tea, such as polyphenols, may reduce blood glucose levels, but a sufficient amount of these bioactive compounds may be needed to be effective,” Dr. Li added.
“The words ‘suggest’ and ‘potentially’ are crucial here,” said Kevin McConway, PhD, MSc, MBA, emeritus professor of applied statistics at The Open University, said in a separate statement to the press that reeled in Dr. Li’s enthusiasm.
“Tea drinking would only be useful for reducing diabetes risk if the tea drinking causes reductions in risk, that is, if the risk is reduced if you drink the tea and not if you don’t – and this study simply can’t show whether it does this or not,” Dr. Conway stressed.
Naveed Sattar, FMedSci FRCPath FRCPGlas FRSE, professor of metabolic medicine at the University of Glasgow, was also cautiously critical. “There is no good trial evidence whatsoever that the chemicals in tea prevent diabetes,” he observed separately.
“So, I suspect its more about tea being healthier (less calorific) than many alternative drinks or tea drinkers leading healthier lives more generally.”
Dr. Sattar added that it could be that people who drink tea might also be avoiding drinking more harmful sugary drinks and have other health behaviors that might lead them to have a lower risk for type 2 diabetes.
Time for tea?
Dr. Li will present the findings of two analyses on Sept. 21 at the EASD meeting: the first a large observational cohort study and the second an updated systematic review and meta-analysis.
For the cohort study, Dr. Li and her coauthors took data on more than 5,100 adults who had participated in the long-running and ongoing China Health and Nutrition Survey (CHNS). Information on tea drinking behavior was extracted from questionnaires that had been filled out at two time points – 1997 and 2009 – and they determined whether people had developed type 2 diabetes according to American Diabetes Association criteria.
Nearly half, 45.8%, were found to be tea drinkers, and 10% of the population they sampled had developed type 2 diabetes. No association between tea drinking and type 2 diabetes development was found, however, with the hazard ratio comparing tea drinkers and non–tea drinkers sitting firmly at 1.02. Moreover, a sensitivity analysis that excluded participants who had developed type 2 diabetes in the first 3 years of follow-up did not change the result.
Things were slightly different when Dr. Li and associates performed their meta-analysis that involved analyzing data on more than 1 million participants in 19 studies conducted in eight countries that had been published up to September 2021.
Here, they found there was a significant (P < .003) linear association between tea consumption and having type 2 diabetes, with the relative risk of developing type 2 diabetes decreasing by 0.986 for every additional cup of tea that was drunk.
HRs for the development of type 2 diabetes in tea drinkers versus non–tea drinkers were 1.00 for those who drank less than one cup per day, 0.96 for those who had one to two cups, and 0.84 for those who drank four or more cups.
“While more research needs to be done to determine the exact dosage and mechanisms behind these observations, our findings suggest that drinking tea is beneficial in reducing the risk of type 2 diabetes, but only at high doses (at least 4 cups a day)”, said Dr. Li.
Perhaps, “we did not find an association between tea drinking and type 2 diabetes in our cohort study because we did not look at higher tea consumption,” she added.
Tempest in a teacup
“This is large, observational data. It’s not a randomized controlled trial so there’s plenty of room for data to be misunderstood,” warned Matt Sydes, MSc, professor of clinical trials & methodology at the MRC Clinical Trials Unit, University College London.
“Everyone drinks fluids. If there is an effect here (and that’s a big if), it might be not about the tea they drink, but about what they don’t drink. One can’t tell at the moment. It seems unlikely that a large randomized controlled trial could be done to disambiguate” added Dr. Sydes
“Being only a conference abstract, it is difficult to assess the quality of this research,” Baptiste Leurent, PhD, a medical statistician also working at University College London, said. Not only was the cohort study observational, so were all the other studies included in the meta-analysis, he pointed out.
“Therefore, no cause-effect conclusions can be drawn. The association could simply be due to other factors, such as those drinking more tea having a healthier lifestyle. It does not seem that the authors tried to control for confounders, which is usually difficult in meta-analysis,” Dr. Leurent said.
“There is reason to be a bit skeptical at this point; we really need to have the full details to assess it properly,” said Jonathan Cook of the Centre for Statistics in Medicine at the University of Oxford (England). “It’s a fair attempt to look at this, but not cutting edge, [using] fairly standard approaches.”
Similar studies have shown a reduced risk associated with coffee drinking, noted Duane Mellor, PhD, a registered dietitian and senior teaching fellow at Aston University in Birmingham.
“The important take-home message is that lifestyle is important in managing risk of developing type 2 diabetes,” Dr. Mellor said.
“That includes choosing low-calorie drinks including mainly water as well as unsweetened tea and coffee as your drinks of choice as part of a healthy lifestyle.”
The study was funded by the Young Talents Project of Hubei Provincial Health Commission, the Science and Technology Research Key Project of Education Department of Hubei Province, the Sanuo Diabetes Charity Foundation, and the Xiangyang Science and Technology Plan Project, all based in China. Dr. Li had no conflicts of interest to disclose. Dr. McConway is a Trustee and on the advisory committee of The Science Media Centre. Dr. Sattar has consulted for many companies that make diabetes and cardiovascular drugs and has been involved in multiple trials of lifestyle approaches for the prevention and remission of diabetes. Dr. Sydes, Dr. Leurent, Dr. Cook, and Dr. Mellor had no conflicts of interest to report.
A claim that drinking tea might protect people against developing type 2 diabetes has been met with caution from multiple experts ahead of the annual meeting of the European Association for the Study of Diabetes.
The claim is that people who drink four or more cups of tea every day – specifically green, Oolong, or black tea – are 17% less likely to develop type 2 diabetes than those who do not drink tea. Drinking fewer cups of tea per day was not found to confer any benefit.
“Our results are exciting because they suggest that people can do something as simple as drinking four cups of tea a day to potentially lessen their risk of developing type 2 diabetes,” Xiaying Li of Wuhan (China) University of Science and Technology is quoted as saying in an official EASD press release.
“It is possible that particular components in tea, such as polyphenols, may reduce blood glucose levels, but a sufficient amount of these bioactive compounds may be needed to be effective,” Dr. Li added.
“The words ‘suggest’ and ‘potentially’ are crucial here,” said Kevin McConway, PhD, MSc, MBA, emeritus professor of applied statistics at The Open University, said in a separate statement to the press that reeled in Dr. Li’s enthusiasm.
“Tea drinking would only be useful for reducing diabetes risk if the tea drinking causes reductions in risk, that is, if the risk is reduced if you drink the tea and not if you don’t – and this study simply can’t show whether it does this or not,” Dr. Conway stressed.
Naveed Sattar, FMedSci FRCPath FRCPGlas FRSE, professor of metabolic medicine at the University of Glasgow, was also cautiously critical. “There is no good trial evidence whatsoever that the chemicals in tea prevent diabetes,” he observed separately.
“So, I suspect its more about tea being healthier (less calorific) than many alternative drinks or tea drinkers leading healthier lives more generally.”
Dr. Sattar added that it could be that people who drink tea might also be avoiding drinking more harmful sugary drinks and have other health behaviors that might lead them to have a lower risk for type 2 diabetes.
Time for tea?
Dr. Li will present the findings of two analyses on Sept. 21 at the EASD meeting: the first a large observational cohort study and the second an updated systematic review and meta-analysis.
For the cohort study, Dr. Li and her coauthors took data on more than 5,100 adults who had participated in the long-running and ongoing China Health and Nutrition Survey (CHNS). Information on tea drinking behavior was extracted from questionnaires that had been filled out at two time points – 1997 and 2009 – and they determined whether people had developed type 2 diabetes according to American Diabetes Association criteria.
Nearly half, 45.8%, were found to be tea drinkers, and 10% of the population they sampled had developed type 2 diabetes. No association between tea drinking and type 2 diabetes development was found, however, with the hazard ratio comparing tea drinkers and non–tea drinkers sitting firmly at 1.02. Moreover, a sensitivity analysis that excluded participants who had developed type 2 diabetes in the first 3 years of follow-up did not change the result.
Things were slightly different when Dr. Li and associates performed their meta-analysis that involved analyzing data on more than 1 million participants in 19 studies conducted in eight countries that had been published up to September 2021.
Here, they found there was a significant (P < .003) linear association between tea consumption and having type 2 diabetes, with the relative risk of developing type 2 diabetes decreasing by 0.986 for every additional cup of tea that was drunk.
HRs for the development of type 2 diabetes in tea drinkers versus non–tea drinkers were 1.00 for those who drank less than one cup per day, 0.96 for those who had one to two cups, and 0.84 for those who drank four or more cups.
“While more research needs to be done to determine the exact dosage and mechanisms behind these observations, our findings suggest that drinking tea is beneficial in reducing the risk of type 2 diabetes, but only at high doses (at least 4 cups a day)”, said Dr. Li.
Perhaps, “we did not find an association between tea drinking and type 2 diabetes in our cohort study because we did not look at higher tea consumption,” she added.
Tempest in a teacup
“This is large, observational data. It’s not a randomized controlled trial so there’s plenty of room for data to be misunderstood,” warned Matt Sydes, MSc, professor of clinical trials & methodology at the MRC Clinical Trials Unit, University College London.
“Everyone drinks fluids. If there is an effect here (and that’s a big if), it might be not about the tea they drink, but about what they don’t drink. One can’t tell at the moment. It seems unlikely that a large randomized controlled trial could be done to disambiguate” added Dr. Sydes
“Being only a conference abstract, it is difficult to assess the quality of this research,” Baptiste Leurent, PhD, a medical statistician also working at University College London, said. Not only was the cohort study observational, so were all the other studies included in the meta-analysis, he pointed out.
“Therefore, no cause-effect conclusions can be drawn. The association could simply be due to other factors, such as those drinking more tea having a healthier lifestyle. It does not seem that the authors tried to control for confounders, which is usually difficult in meta-analysis,” Dr. Leurent said.
“There is reason to be a bit skeptical at this point; we really need to have the full details to assess it properly,” said Jonathan Cook of the Centre for Statistics in Medicine at the University of Oxford (England). “It’s a fair attempt to look at this, but not cutting edge, [using] fairly standard approaches.”
Similar studies have shown a reduced risk associated with coffee drinking, noted Duane Mellor, PhD, a registered dietitian and senior teaching fellow at Aston University in Birmingham.
“The important take-home message is that lifestyle is important in managing risk of developing type 2 diabetes,” Dr. Mellor said.
“That includes choosing low-calorie drinks including mainly water as well as unsweetened tea and coffee as your drinks of choice as part of a healthy lifestyle.”
The study was funded by the Young Talents Project of Hubei Provincial Health Commission, the Science and Technology Research Key Project of Education Department of Hubei Province, the Sanuo Diabetes Charity Foundation, and the Xiangyang Science and Technology Plan Project, all based in China. Dr. Li had no conflicts of interest to disclose. Dr. McConway is a Trustee and on the advisory committee of The Science Media Centre. Dr. Sattar has consulted for many companies that make diabetes and cardiovascular drugs and has been involved in multiple trials of lifestyle approaches for the prevention and remission of diabetes. Dr. Sydes, Dr. Leurent, Dr. Cook, and Dr. Mellor had no conflicts of interest to report.
type 2 diabetes/tea-drinking claim
type 2 diabetes/tea-drinking claim
FROM EASD 2022
EHR: A progress report
I wrote my first column on electronic health records in the mid-1990s. At the time, it seemed like an idea whose time had come. After all, in an era when just about every essential process in medicine had already been computerized, we physicians continued to process clinical data – our key asset – with pen and paper. Most of us were reluctant to make the switch, and for good reason:
Then, the government stepped in. Shortly after his inauguration in 2000, President George W. Bush outlined a plan to ensure that most Americans had electronic health records within 10 years. “By computerizing health records,” the president said, “we can avoid dangerous medical mistakes, reduce costs, and improve care.” The goal was to eliminate missing charts, duplication of lab testing, ineffective documentation, and inordinate amounts of time spent on paperwork, not to mention illegible handwriting, poor coordination of care between physicians, and many other problems. Studies were quoted, suggesting that EHR shortened inpatient stays, decreased risk of adverse drug interactions, improved the consistency and content of records, and improved continuity of care and follow-up.
The EHR Incentive Program (later renamed the Promoting Interoperability Program) was introduced to encourage physicians and hospitals “to adopt, implement, upgrade, and demonstrate meaningful use of certified electronic health record technology.”
Nearly a quarter-century later, implementation is well behind schedule. According to a 2019 federal study, while nearly all hospitals (96%) have adopted a certified EHR, only 72% of office-based physicians have done so.
There are multiple reasons for this. For one thing, EHR is still by and large slower than pen and paper, because direct data entry is still primarily done by keyboard. Voice recognition, hand-held and wireless devices have been developed, but most work only on specialized tasks. Even the best systems take more clinician time per encounter than the manual processes they replace.
Physicians have been slow to warm to a system that slows them down and forces them to change the way they think and work. In addition, paper systems never crash; the prospect of a server malfunction or Internet failure bringing an entire clinic to a grinding halt is not particularly inviting.
The special needs of dermatology – high patient volumes, multiple diagnoses and prescriptions per patient, the wide variety of procedures we perform, and digital image storage – present further hurdles.
Nevertheless, the march toward electronic record keeping continues, and I continue to receive many questions about choosing a good EHR system. As always, I cannot recommend any specific products since every office has unique needs and requirements.
The key phrase to keep in mind is caveat emptor. Several regulatory bodies exist to test vendor claims and certify system behaviors, but different agencies use different criteria that may or may not be relevant to your requirements. Vaporware is still as common as real software; beware the “feature in the next release” that might never appear, particularly if you need it right now.
Avoid the temptation to buy a flashy new system and then try to adapt it to your office; figure out your needs first, then find a system that meets them.
Unfortunately, there is no easy way around doing the work of comparing one system with another. The most important information a vendor can give you is the names and addresses of two or more offices where you can go watch their system in action. Site visits are time-consuming, but they are only way to pick the best EHR the first time around.
Don’t be the first office using a new system. Let the vendor work out the bugs somewhere else.
Above all, if you have disorganized paper records, don’t count on EHR to automatically solve your problems. Well-designed paper systems usually lend themselves to effective automation, but automating a poorly designed system just increases the chaos. If your paper system is in disarray, solve that problem before considering EHR.
With all of its problems and hurdles, EHRs will inevitably be a part of most of our lives. And for those who take the time to do it right, it will ultimately be an improvement.
Think of information technologies as power tools: They can help you to do things better, but they can also amplify your errors. So choose carefully.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
I wrote my first column on electronic health records in the mid-1990s. At the time, it seemed like an idea whose time had come. After all, in an era when just about every essential process in medicine had already been computerized, we physicians continued to process clinical data – our key asset – with pen and paper. Most of us were reluctant to make the switch, and for good reason:
Then, the government stepped in. Shortly after his inauguration in 2000, President George W. Bush outlined a plan to ensure that most Americans had electronic health records within 10 years. “By computerizing health records,” the president said, “we can avoid dangerous medical mistakes, reduce costs, and improve care.” The goal was to eliminate missing charts, duplication of lab testing, ineffective documentation, and inordinate amounts of time spent on paperwork, not to mention illegible handwriting, poor coordination of care between physicians, and many other problems. Studies were quoted, suggesting that EHR shortened inpatient stays, decreased risk of adverse drug interactions, improved the consistency and content of records, and improved continuity of care and follow-up.
The EHR Incentive Program (later renamed the Promoting Interoperability Program) was introduced to encourage physicians and hospitals “to adopt, implement, upgrade, and demonstrate meaningful use of certified electronic health record technology.”
Nearly a quarter-century later, implementation is well behind schedule. According to a 2019 federal study, while nearly all hospitals (96%) have adopted a certified EHR, only 72% of office-based physicians have done so.
There are multiple reasons for this. For one thing, EHR is still by and large slower than pen and paper, because direct data entry is still primarily done by keyboard. Voice recognition, hand-held and wireless devices have been developed, but most work only on specialized tasks. Even the best systems take more clinician time per encounter than the manual processes they replace.
Physicians have been slow to warm to a system that slows them down and forces them to change the way they think and work. In addition, paper systems never crash; the prospect of a server malfunction or Internet failure bringing an entire clinic to a grinding halt is not particularly inviting.
The special needs of dermatology – high patient volumes, multiple diagnoses and prescriptions per patient, the wide variety of procedures we perform, and digital image storage – present further hurdles.
Nevertheless, the march toward electronic record keeping continues, and I continue to receive many questions about choosing a good EHR system. As always, I cannot recommend any specific products since every office has unique needs and requirements.
The key phrase to keep in mind is caveat emptor. Several regulatory bodies exist to test vendor claims and certify system behaviors, but different agencies use different criteria that may or may not be relevant to your requirements. Vaporware is still as common as real software; beware the “feature in the next release” that might never appear, particularly if you need it right now.
Avoid the temptation to buy a flashy new system and then try to adapt it to your office; figure out your needs first, then find a system that meets them.
Unfortunately, there is no easy way around doing the work of comparing one system with another. The most important information a vendor can give you is the names and addresses of two or more offices where you can go watch their system in action. Site visits are time-consuming, but they are only way to pick the best EHR the first time around.
Don’t be the first office using a new system. Let the vendor work out the bugs somewhere else.
Above all, if you have disorganized paper records, don’t count on EHR to automatically solve your problems. Well-designed paper systems usually lend themselves to effective automation, but automating a poorly designed system just increases the chaos. If your paper system is in disarray, solve that problem before considering EHR.
With all of its problems and hurdles, EHRs will inevitably be a part of most of our lives. And for those who take the time to do it right, it will ultimately be an improvement.
Think of information technologies as power tools: They can help you to do things better, but they can also amplify your errors. So choose carefully.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
I wrote my first column on electronic health records in the mid-1990s. At the time, it seemed like an idea whose time had come. After all, in an era when just about every essential process in medicine had already been computerized, we physicians continued to process clinical data – our key asset – with pen and paper. Most of us were reluctant to make the switch, and for good reason:
Then, the government stepped in. Shortly after his inauguration in 2000, President George W. Bush outlined a plan to ensure that most Americans had electronic health records within 10 years. “By computerizing health records,” the president said, “we can avoid dangerous medical mistakes, reduce costs, and improve care.” The goal was to eliminate missing charts, duplication of lab testing, ineffective documentation, and inordinate amounts of time spent on paperwork, not to mention illegible handwriting, poor coordination of care between physicians, and many other problems. Studies were quoted, suggesting that EHR shortened inpatient stays, decreased risk of adverse drug interactions, improved the consistency and content of records, and improved continuity of care and follow-up.
The EHR Incentive Program (later renamed the Promoting Interoperability Program) was introduced to encourage physicians and hospitals “to adopt, implement, upgrade, and demonstrate meaningful use of certified electronic health record technology.”
Nearly a quarter-century later, implementation is well behind schedule. According to a 2019 federal study, while nearly all hospitals (96%) have adopted a certified EHR, only 72% of office-based physicians have done so.
There are multiple reasons for this. For one thing, EHR is still by and large slower than pen and paper, because direct data entry is still primarily done by keyboard. Voice recognition, hand-held and wireless devices have been developed, but most work only on specialized tasks. Even the best systems take more clinician time per encounter than the manual processes they replace.
Physicians have been slow to warm to a system that slows them down and forces them to change the way they think and work. In addition, paper systems never crash; the prospect of a server malfunction or Internet failure bringing an entire clinic to a grinding halt is not particularly inviting.
The special needs of dermatology – high patient volumes, multiple diagnoses and prescriptions per patient, the wide variety of procedures we perform, and digital image storage – present further hurdles.
Nevertheless, the march toward electronic record keeping continues, and I continue to receive many questions about choosing a good EHR system. As always, I cannot recommend any specific products since every office has unique needs and requirements.
The key phrase to keep in mind is caveat emptor. Several regulatory bodies exist to test vendor claims and certify system behaviors, but different agencies use different criteria that may or may not be relevant to your requirements. Vaporware is still as common as real software; beware the “feature in the next release” that might never appear, particularly if you need it right now.
Avoid the temptation to buy a flashy new system and then try to adapt it to your office; figure out your needs first, then find a system that meets them.
Unfortunately, there is no easy way around doing the work of comparing one system with another. The most important information a vendor can give you is the names and addresses of two or more offices where you can go watch their system in action. Site visits are time-consuming, but they are only way to pick the best EHR the first time around.
Don’t be the first office using a new system. Let the vendor work out the bugs somewhere else.
Above all, if you have disorganized paper records, don’t count on EHR to automatically solve your problems. Well-designed paper systems usually lend themselves to effective automation, but automating a poorly designed system just increases the chaos. If your paper system is in disarray, solve that problem before considering EHR.
With all of its problems and hurdles, EHRs will inevitably be a part of most of our lives. And for those who take the time to do it right, it will ultimately be an improvement.
Think of information technologies as power tools: They can help you to do things better, but they can also amplify your errors. So choose carefully.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].














