User login
Can biomarkers detect concussions? It’s complicated
A series of three studies in college students showed that some serum markers are associated with concussion but the background level of the markers can vary considerably. There was no association between the markers and history of concussion, and they markers varied significantly by sex and race.
The work, published in Neurology, suggests that there is hope for finding biomarkers for concussion, but much more work needs to be done.
Serum levels of amyloid beta 42 (Abeta42), total tau, and S100 calcium binding protein B (S100B) were associated with concussion, especially when tests were performed within 4 hours of the injury. However, the varying background levels indicate that these biomarkers are not yet ready for clinical application.
All three studies looked at serum levels of Abeta42, total tau, S100B, ubiquitin carboxyl-terminal hydrolase L1 (UCH-L1), glial fibrillary acidic protein (GFAP), microtubule-associated protein 2 (MAP2), and 2’,3’-cyclic-nucleotide 3’-phosphodiesterase (CNPase).
In the first study, researchers recruited 415 college athletes without a concussion (61% male, 40% white). The researchers took measurements outside of the athletes’ competitive sports season to maximize the odds that the levels would represent a true baseline. The median time between blood draw and the last risk of head impact was 80 days (mean, 98.4 days; interquartile range, 38-204 days).
Males had higher levels of UCH-L1 (Cohen d = 0.75; P less than .001) and S100B (Cohen d = 0.56; P less than .001), while females had higher levels of CNPase (Cohen d = 0.43; P less than .001). White subjects had higher levels of Abeta42 (Cohen d = .28; P = .005) and CNPase (Cohen d = 0.46; P less than .001). Black subjects had higher levels of UCH-L1 (Cohen d = 0.61; P less than .001) and S100 B (Cohen d = 1.1; P less than .001).
The measurements were not particularly reliable, with retests over 6- to 12-month periods yielding varying results such that none of the test/retest cutoff points reached the cutoff for acceptable reliability.
The second study was an observational cohort study of the same 415 subjects. The researchers assessed the self-reported concussion history and the cumulative exposure to collision sports with serum levels of the above biomarkers. The only relationship between a biomarker history and self-reported concussions was higher baseline Abeta42, but that had a small effect size (P = .005). Among football players, there was no association between approximate number of head impacts and any baseline biomarker.
The third study looked at 31 subjects who had experienced a sports-related concussion, 29 of whom had had both a baseline and a postconcussion blood draw, and compared them with nonconcussed, demographically matched athletes.
Of all the biomarkers studied, only levels of S100B rose following a concussion, with 67% of concussed subjects experiencing such a change (P = .003). When the researchers restricted the analysis to subjects who had a blood draw within 4 hours of the concussion, 88% of the tests showed an increase (P = .001). UCH-L1 also rose in 86% of subjects, but this change was not significant after adjustment for multiple comparisons (P greater than .007).
Compared with controls, concussed individuals had significantly higher levels of Abeta42, total tau, S100B, and GFAP. Of the concussed patients, 79.4% had Abeta42 levels higher than the median of controls, 67.6% had higher levels of total tau than the median of controls, and 83.3% had higher levels of S100B. Restriction of analysis to blood drawn within 4 hours of the injury yielded values of 81.3%, 75.0%, and 88.2%, respectively.
When limited to blood draws taken within 4 hours of injury, the researchers found fair diagnostic accuracy for measurements of Abeta42 (area under the curve, 0.75; 95% confidence interval, 0.59-0.91), total tau (AUC, 0.74; 95% CI, 0.58-0.90), and S100B (AUC, 0.75; 95% CI, 0.64-0.85). Abeta42 concentrations higher than 13.7 pg/mL were 75.0% sensitive and 82.4% specific to a sports-related concussion. Total tau concentrations higher than 1.7 pg/mL detected sports-related concussions at 75.0% sensitivity and 66.3% specificity, with acceptable diagnostic accuracy for white subjects (AUC, 0.82, 95% CI, 0.72-0.93). Also for white participants, S100B concentrations higher than 53 pg/mL predicted sports-related concussions with 83.3% sensitivity and 74.6% specificity.
The researchers found no associations between biomarkers and performance on clinical tests or time away from sports.
SOURCE: BM Asken et al. Neurology. 2018. doi: 10.1212/WNL.0000000000006613.
Concussion diagnosis has been constrained by reliance on subjective evidence, particularly in mild cases. Concussions also often result from a wide range of injuries, but focusing on sports-related concussions offers a chance to study biomarkers in a more controlled way.
These three studies represent the most comprehensive sports-related concussion biomarker work to date. The message may be that, for sports-related concussions, serum biomarkers may be able to detect the occurrence of a concussion, but they cannot predict motor, neurobehavioral, or neurocognitive outcome measures.
The study results also underline the need for larger, more complex prospective studies.
Erin Bigler, PhD, is a professor of psychology and neuroscience at Brigham Young University. Ellen Deibert, MD, is a neurologist in York, Pa. These comments were taken from an accompanying editorial (Neurology. 2018. doi: 10.1212/WNL.0000000000006609 ). Dr. Bigler and Dr. Deibert have no relevant conflicts of interest.
Concussion diagnosis has been constrained by reliance on subjective evidence, particularly in mild cases. Concussions also often result from a wide range of injuries, but focusing on sports-related concussions offers a chance to study biomarkers in a more controlled way.
These three studies represent the most comprehensive sports-related concussion biomarker work to date. The message may be that, for sports-related concussions, serum biomarkers may be able to detect the occurrence of a concussion, but they cannot predict motor, neurobehavioral, or neurocognitive outcome measures.
The study results also underline the need for larger, more complex prospective studies.
Erin Bigler, PhD, is a professor of psychology and neuroscience at Brigham Young University. Ellen Deibert, MD, is a neurologist in York, Pa. These comments were taken from an accompanying editorial (Neurology. 2018. doi: 10.1212/WNL.0000000000006609 ). Dr. Bigler and Dr. Deibert have no relevant conflicts of interest.
Concussion diagnosis has been constrained by reliance on subjective evidence, particularly in mild cases. Concussions also often result from a wide range of injuries, but focusing on sports-related concussions offers a chance to study biomarkers in a more controlled way.
These three studies represent the most comprehensive sports-related concussion biomarker work to date. The message may be that, for sports-related concussions, serum biomarkers may be able to detect the occurrence of a concussion, but they cannot predict motor, neurobehavioral, or neurocognitive outcome measures.
The study results also underline the need for larger, more complex prospective studies.
Erin Bigler, PhD, is a professor of psychology and neuroscience at Brigham Young University. Ellen Deibert, MD, is a neurologist in York, Pa. These comments were taken from an accompanying editorial (Neurology. 2018. doi: 10.1212/WNL.0000000000006609 ). Dr. Bigler and Dr. Deibert have no relevant conflicts of interest.
A series of three studies in college students showed that some serum markers are associated with concussion but the background level of the markers can vary considerably. There was no association between the markers and history of concussion, and they markers varied significantly by sex and race.
The work, published in Neurology, suggests that there is hope for finding biomarkers for concussion, but much more work needs to be done.
Serum levels of amyloid beta 42 (Abeta42), total tau, and S100 calcium binding protein B (S100B) were associated with concussion, especially when tests were performed within 4 hours of the injury. However, the varying background levels indicate that these biomarkers are not yet ready for clinical application.
All three studies looked at serum levels of Abeta42, total tau, S100B, ubiquitin carboxyl-terminal hydrolase L1 (UCH-L1), glial fibrillary acidic protein (GFAP), microtubule-associated protein 2 (MAP2), and 2’,3’-cyclic-nucleotide 3’-phosphodiesterase (CNPase).
In the first study, researchers recruited 415 college athletes without a concussion (61% male, 40% white). The researchers took measurements outside of the athletes’ competitive sports season to maximize the odds that the levels would represent a true baseline. The median time between blood draw and the last risk of head impact was 80 days (mean, 98.4 days; interquartile range, 38-204 days).
Males had higher levels of UCH-L1 (Cohen d = 0.75; P less than .001) and S100B (Cohen d = 0.56; P less than .001), while females had higher levels of CNPase (Cohen d = 0.43; P less than .001). White subjects had higher levels of Abeta42 (Cohen d = .28; P = .005) and CNPase (Cohen d = 0.46; P less than .001). Black subjects had higher levels of UCH-L1 (Cohen d = 0.61; P less than .001) and S100 B (Cohen d = 1.1; P less than .001).
The measurements were not particularly reliable, with retests over 6- to 12-month periods yielding varying results such that none of the test/retest cutoff points reached the cutoff for acceptable reliability.
The second study was an observational cohort study of the same 415 subjects. The researchers assessed the self-reported concussion history and the cumulative exposure to collision sports with serum levels of the above biomarkers. The only relationship between a biomarker history and self-reported concussions was higher baseline Abeta42, but that had a small effect size (P = .005). Among football players, there was no association between approximate number of head impacts and any baseline biomarker.
The third study looked at 31 subjects who had experienced a sports-related concussion, 29 of whom had had both a baseline and a postconcussion blood draw, and compared them with nonconcussed, demographically matched athletes.
Of all the biomarkers studied, only levels of S100B rose following a concussion, with 67% of concussed subjects experiencing such a change (P = .003). When the researchers restricted the analysis to subjects who had a blood draw within 4 hours of the concussion, 88% of the tests showed an increase (P = .001). UCH-L1 also rose in 86% of subjects, but this change was not significant after adjustment for multiple comparisons (P greater than .007).
Compared with controls, concussed individuals had significantly higher levels of Abeta42, total tau, S100B, and GFAP. Of the concussed patients, 79.4% had Abeta42 levels higher than the median of controls, 67.6% had higher levels of total tau than the median of controls, and 83.3% had higher levels of S100B. Restriction of analysis to blood drawn within 4 hours of the injury yielded values of 81.3%, 75.0%, and 88.2%, respectively.
When limited to blood draws taken within 4 hours of injury, the researchers found fair diagnostic accuracy for measurements of Abeta42 (area under the curve, 0.75; 95% confidence interval, 0.59-0.91), total tau (AUC, 0.74; 95% CI, 0.58-0.90), and S100B (AUC, 0.75; 95% CI, 0.64-0.85). Abeta42 concentrations higher than 13.7 pg/mL were 75.0% sensitive and 82.4% specific to a sports-related concussion. Total tau concentrations higher than 1.7 pg/mL detected sports-related concussions at 75.0% sensitivity and 66.3% specificity, with acceptable diagnostic accuracy for white subjects (AUC, 0.82, 95% CI, 0.72-0.93). Also for white participants, S100B concentrations higher than 53 pg/mL predicted sports-related concussions with 83.3% sensitivity and 74.6% specificity.
The researchers found no associations between biomarkers and performance on clinical tests or time away from sports.
SOURCE: BM Asken et al. Neurology. 2018. doi: 10.1212/WNL.0000000000006613.
A series of three studies in college students showed that some serum markers are associated with concussion but the background level of the markers can vary considerably. There was no association between the markers and history of concussion, and they markers varied significantly by sex and race.
The work, published in Neurology, suggests that there is hope for finding biomarkers for concussion, but much more work needs to be done.
Serum levels of amyloid beta 42 (Abeta42), total tau, and S100 calcium binding protein B (S100B) were associated with concussion, especially when tests were performed within 4 hours of the injury. However, the varying background levels indicate that these biomarkers are not yet ready for clinical application.
All three studies looked at serum levels of Abeta42, total tau, S100B, ubiquitin carboxyl-terminal hydrolase L1 (UCH-L1), glial fibrillary acidic protein (GFAP), microtubule-associated protein 2 (MAP2), and 2’,3’-cyclic-nucleotide 3’-phosphodiesterase (CNPase).
In the first study, researchers recruited 415 college athletes without a concussion (61% male, 40% white). The researchers took measurements outside of the athletes’ competitive sports season to maximize the odds that the levels would represent a true baseline. The median time between blood draw and the last risk of head impact was 80 days (mean, 98.4 days; interquartile range, 38-204 days).
Males had higher levels of UCH-L1 (Cohen d = 0.75; P less than .001) and S100B (Cohen d = 0.56; P less than .001), while females had higher levels of CNPase (Cohen d = 0.43; P less than .001). White subjects had higher levels of Abeta42 (Cohen d = .28; P = .005) and CNPase (Cohen d = 0.46; P less than .001). Black subjects had higher levels of UCH-L1 (Cohen d = 0.61; P less than .001) and S100 B (Cohen d = 1.1; P less than .001).
The measurements were not particularly reliable, with retests over 6- to 12-month periods yielding varying results such that none of the test/retest cutoff points reached the cutoff for acceptable reliability.
The second study was an observational cohort study of the same 415 subjects. The researchers assessed the self-reported concussion history and the cumulative exposure to collision sports with serum levels of the above biomarkers. The only relationship between a biomarker history and self-reported concussions was higher baseline Abeta42, but that had a small effect size (P = .005). Among football players, there was no association between approximate number of head impacts and any baseline biomarker.
The third study looked at 31 subjects who had experienced a sports-related concussion, 29 of whom had had both a baseline and a postconcussion blood draw, and compared them with nonconcussed, demographically matched athletes.
Of all the biomarkers studied, only levels of S100B rose following a concussion, with 67% of concussed subjects experiencing such a change (P = .003). When the researchers restricted the analysis to subjects who had a blood draw within 4 hours of the concussion, 88% of the tests showed an increase (P = .001). UCH-L1 also rose in 86% of subjects, but this change was not significant after adjustment for multiple comparisons (P greater than .007).
Compared with controls, concussed individuals had significantly higher levels of Abeta42, total tau, S100B, and GFAP. Of the concussed patients, 79.4% had Abeta42 levels higher than the median of controls, 67.6% had higher levels of total tau than the median of controls, and 83.3% had higher levels of S100B. Restriction of analysis to blood drawn within 4 hours of the injury yielded values of 81.3%, 75.0%, and 88.2%, respectively.
When limited to blood draws taken within 4 hours of injury, the researchers found fair diagnostic accuracy for measurements of Abeta42 (area under the curve, 0.75; 95% confidence interval, 0.59-0.91), total tau (AUC, 0.74; 95% CI, 0.58-0.90), and S100B (AUC, 0.75; 95% CI, 0.64-0.85). Abeta42 concentrations higher than 13.7 pg/mL were 75.0% sensitive and 82.4% specific to a sports-related concussion. Total tau concentrations higher than 1.7 pg/mL detected sports-related concussions at 75.0% sensitivity and 66.3% specificity, with acceptable diagnostic accuracy for white subjects (AUC, 0.82, 95% CI, 0.72-0.93). Also for white participants, S100B concentrations higher than 53 pg/mL predicted sports-related concussions with 83.3% sensitivity and 74.6% specificity.
The researchers found no associations between biomarkers and performance on clinical tests or time away from sports.
SOURCE: BM Asken et al. Neurology. 2018. doi: 10.1212/WNL.0000000000006613.
FROM NEUROLOGY
Key clinical point: Serum biomarkers show promise in concussion diagnosis, but much work remains.
Major finding: Serum levels of Abeta42, total tau, and S100B were elevated after concussions.
Study details: Prospective studies on 415 college athletes.
Disclosures: The study was funded by the Head Health Initiative, Banyan Biomarkers, and the United States Army Medical Research and Materiel Command.
Sources: BM Asken et al. Neurology. 2018. doi: 10.1212/WNL.0000000000006613.
Circulating tumor DNA identified by fragment size
Circulating tumor DNA could be effectively isolated from plasma by focusing on a particular range of fragment sizes, which paves the way for noninvasive genomic analysis of tumor DNA, new research suggests.
In a study of 344 plasma samples from 200 patients with 18 cancer types and 65 samples from healthy controls, DNA fragment length could be used to distinguish circulating tumor DNA (ctDNA) from other cell-free DNA (cfDNA), investigators reported in Science Translational Medicine.
“We hypothesized that we could improve the sensitivity for noninvasive cancer genomics by selective sequencing of ctDNA fragments and by leveraging differences in the biology that determine DNA fragmentation,” wrote Florent Mouliere, PhD, from the Cancer Research UK Cambridge Institute, and coauthors.
Cell-free plasma fragments are often cleaved at around 167 base pairs in length and differences in length between circulating fetal and maternal DNA are already used for noninvasive prenatal diagnosis. However, the authors said that only a few studies, with conflicting results, have looked at the size distribution of tumor-derived cfDNA.
The study used two approaches to determining the size profile of mutant ctDNA. The first looked at tumor and nontumor cfDNA in mice with human ovarian cancer xenografts and the second approach used deep sequencing in 19 cancer patients. This revealed that tumor-derived cfDNA was most commonly found in fragments between 90-150 base pairs or 250-320 base pairs in size.
The researchers also noted that mutant circulating tumor DNA was generally more fragmented than nonmutant cfDNA and that patients with untreated advanced cancer showed consistently shorter lengths of mutant DNA.
The next question was whether size selection and other biological properties – such as somatic alterations – of the cfDNA could be used to enhance detection of ctDNA via machine learning technology.
Two models, designed to distinguish between healthy and cancerous samples, were developed using 153 samples, then validated on two datasets of 94 and 83 samples.
One of these models correctly classified cancerous samples in 94% of samples from patients with cancers known to have high levels of ctDNA – colorectal, cholangiocarcinoma, ovarian, breast, and melanoma – and in 65% of samples from low-ctDNA cancers – pancreatic, renal, and glioma.
Another model focused just on fragmentation patterns and was still able to distinguish cancer samples from those of healthy controls, although with slightly reduced area under the curve.
“Our results indicate that exploiting fundamental properties of cfDNA with fragment-specific analyses can allow more sensitive evaluation of ctDNA,” the authors wrote. “We identified features that could determine the presence and amount of ctDNA in plasma samples, without a prior knowledge of somatic aberrations.”
The authors pointed out that size selection of DNA fragments was relatively simple and cheap, and was also compatible with other genome-wide and targeted genomic analyses, “greatly increasing the potential value and utility of liquid biopsies as well as the cost-effectiveness of cfDNA sequencing.”
However, they cautioned that their catalogue had focused solely on double-stranded DNA and was subject to potential biases from the DNA extraction and sequencing methods they used in the study. They also commented that other biological effects could help refine the analysis of ctDNA.
“Other bodily fluids [urine, cerebrospinal fluid, and saliva], different nucleic acids and structures, altered mechanisms of release into circulation, or sample processing methods could exhibit varying fragment size signatures and could offer additional exploitable biological patterns for selective sequencing,” they wrote.
The study was supported by the University of Cambridge, Cancer Research UK, and the Engineering and Physical Sciences Research Council. Research supporting the study was also funded by the European Research Council, the National Institute for Health Research Cambridge, National Cancer Research Network, Cambridge Experimental Cancer Medicine Centre, Hutchison Whampoa, Target Ovarian Cancer, the Medical Research Council, and AstraZeneca. Three authors are cofounders, shareholders, and officers/consultants in a company specializing in ctDNA analysis. One author declared research funding and advisory board fees from private industry. Seven authors are listed on related patents.
SOURCE: Mouliere F et al. Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aat4921.
Cell-free DNA analysis has tremendous diagnostic potential and so is a very active area of research. In this study, researchers were able to identify five variables and develop models for the detection of cancer following analysis of circulating tumor DNA. One of these models based on DNA fragmentation pattern performed very well, and so fragment length analyses could develop into a general test for the presence of cancer.
However confirmation of these findings in large, multicenter clinical trials is still needed. There is also the problem that size selection can result in a loss of circulating tumor DNA for analysis or may introduce biases. We also need to understand the mechanisms underpinning the different fragment size patterns seen in the study. But this study still substantially extends the potential of cell-free, DNA-based diagnostic tests.
Ellen Heitzer, PhD, and Michael R. Speicher, MD, are from the Medical University of Graz (Austria). These comments are taken from an accompanying editorial (Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aav3873). Both authors declared research funding from Servier and Dr. Heitzer declared laboratory research funding from Freenome and PreAnalytiX.
Cell-free DNA analysis has tremendous diagnostic potential and so is a very active area of research. In this study, researchers were able to identify five variables and develop models for the detection of cancer following analysis of circulating tumor DNA. One of these models based on DNA fragmentation pattern performed very well, and so fragment length analyses could develop into a general test for the presence of cancer.
However confirmation of these findings in large, multicenter clinical trials is still needed. There is also the problem that size selection can result in a loss of circulating tumor DNA for analysis or may introduce biases. We also need to understand the mechanisms underpinning the different fragment size patterns seen in the study. But this study still substantially extends the potential of cell-free, DNA-based diagnostic tests.
Ellen Heitzer, PhD, and Michael R. Speicher, MD, are from the Medical University of Graz (Austria). These comments are taken from an accompanying editorial (Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aav3873). Both authors declared research funding from Servier and Dr. Heitzer declared laboratory research funding from Freenome and PreAnalytiX.
Cell-free DNA analysis has tremendous diagnostic potential and so is a very active area of research. In this study, researchers were able to identify five variables and develop models for the detection of cancer following analysis of circulating tumor DNA. One of these models based on DNA fragmentation pattern performed very well, and so fragment length analyses could develop into a general test for the presence of cancer.
However confirmation of these findings in large, multicenter clinical trials is still needed. There is also the problem that size selection can result in a loss of circulating tumor DNA for analysis or may introduce biases. We also need to understand the mechanisms underpinning the different fragment size patterns seen in the study. But this study still substantially extends the potential of cell-free, DNA-based diagnostic tests.
Ellen Heitzer, PhD, and Michael R. Speicher, MD, are from the Medical University of Graz (Austria). These comments are taken from an accompanying editorial (Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aav3873). Both authors declared research funding from Servier and Dr. Heitzer declared laboratory research funding from Freenome and PreAnalytiX.
Circulating tumor DNA could be effectively isolated from plasma by focusing on a particular range of fragment sizes, which paves the way for noninvasive genomic analysis of tumor DNA, new research suggests.
In a study of 344 plasma samples from 200 patients with 18 cancer types and 65 samples from healthy controls, DNA fragment length could be used to distinguish circulating tumor DNA (ctDNA) from other cell-free DNA (cfDNA), investigators reported in Science Translational Medicine.
“We hypothesized that we could improve the sensitivity for noninvasive cancer genomics by selective sequencing of ctDNA fragments and by leveraging differences in the biology that determine DNA fragmentation,” wrote Florent Mouliere, PhD, from the Cancer Research UK Cambridge Institute, and coauthors.
Cell-free plasma fragments are often cleaved at around 167 base pairs in length and differences in length between circulating fetal and maternal DNA are already used for noninvasive prenatal diagnosis. However, the authors said that only a few studies, with conflicting results, have looked at the size distribution of tumor-derived cfDNA.
The study used two approaches to determining the size profile of mutant ctDNA. The first looked at tumor and nontumor cfDNA in mice with human ovarian cancer xenografts and the second approach used deep sequencing in 19 cancer patients. This revealed that tumor-derived cfDNA was most commonly found in fragments between 90-150 base pairs or 250-320 base pairs in size.
The researchers also noted that mutant circulating tumor DNA was generally more fragmented than nonmutant cfDNA and that patients with untreated advanced cancer showed consistently shorter lengths of mutant DNA.
The next question was whether size selection and other biological properties – such as somatic alterations – of the cfDNA could be used to enhance detection of ctDNA via machine learning technology.
Two models, designed to distinguish between healthy and cancerous samples, were developed using 153 samples, then validated on two datasets of 94 and 83 samples.
One of these models correctly classified cancerous samples in 94% of samples from patients with cancers known to have high levels of ctDNA – colorectal, cholangiocarcinoma, ovarian, breast, and melanoma – and in 65% of samples from low-ctDNA cancers – pancreatic, renal, and glioma.
Another model focused just on fragmentation patterns and was still able to distinguish cancer samples from those of healthy controls, although with slightly reduced area under the curve.
“Our results indicate that exploiting fundamental properties of cfDNA with fragment-specific analyses can allow more sensitive evaluation of ctDNA,” the authors wrote. “We identified features that could determine the presence and amount of ctDNA in plasma samples, without a prior knowledge of somatic aberrations.”
The authors pointed out that size selection of DNA fragments was relatively simple and cheap, and was also compatible with other genome-wide and targeted genomic analyses, “greatly increasing the potential value and utility of liquid biopsies as well as the cost-effectiveness of cfDNA sequencing.”
However, they cautioned that their catalogue had focused solely on double-stranded DNA and was subject to potential biases from the DNA extraction and sequencing methods they used in the study. They also commented that other biological effects could help refine the analysis of ctDNA.
“Other bodily fluids [urine, cerebrospinal fluid, and saliva], different nucleic acids and structures, altered mechanisms of release into circulation, or sample processing methods could exhibit varying fragment size signatures and could offer additional exploitable biological patterns for selective sequencing,” they wrote.
The study was supported by the University of Cambridge, Cancer Research UK, and the Engineering and Physical Sciences Research Council. Research supporting the study was also funded by the European Research Council, the National Institute for Health Research Cambridge, National Cancer Research Network, Cambridge Experimental Cancer Medicine Centre, Hutchison Whampoa, Target Ovarian Cancer, the Medical Research Council, and AstraZeneca. Three authors are cofounders, shareholders, and officers/consultants in a company specializing in ctDNA analysis. One author declared research funding and advisory board fees from private industry. Seven authors are listed on related patents.
SOURCE: Mouliere F et al. Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aat4921.
Circulating tumor DNA could be effectively isolated from plasma by focusing on a particular range of fragment sizes, which paves the way for noninvasive genomic analysis of tumor DNA, new research suggests.
In a study of 344 plasma samples from 200 patients with 18 cancer types and 65 samples from healthy controls, DNA fragment length could be used to distinguish circulating tumor DNA (ctDNA) from other cell-free DNA (cfDNA), investigators reported in Science Translational Medicine.
“We hypothesized that we could improve the sensitivity for noninvasive cancer genomics by selective sequencing of ctDNA fragments and by leveraging differences in the biology that determine DNA fragmentation,” wrote Florent Mouliere, PhD, from the Cancer Research UK Cambridge Institute, and coauthors.
Cell-free plasma fragments are often cleaved at around 167 base pairs in length and differences in length between circulating fetal and maternal DNA are already used for noninvasive prenatal diagnosis. However, the authors said that only a few studies, with conflicting results, have looked at the size distribution of tumor-derived cfDNA.
The study used two approaches to determining the size profile of mutant ctDNA. The first looked at tumor and nontumor cfDNA in mice with human ovarian cancer xenografts and the second approach used deep sequencing in 19 cancer patients. This revealed that tumor-derived cfDNA was most commonly found in fragments between 90-150 base pairs or 250-320 base pairs in size.
The researchers also noted that mutant circulating tumor DNA was generally more fragmented than nonmutant cfDNA and that patients with untreated advanced cancer showed consistently shorter lengths of mutant DNA.
The next question was whether size selection and other biological properties – such as somatic alterations – of the cfDNA could be used to enhance detection of ctDNA via machine learning technology.
Two models, designed to distinguish between healthy and cancerous samples, were developed using 153 samples, then validated on two datasets of 94 and 83 samples.
One of these models correctly classified cancerous samples in 94% of samples from patients with cancers known to have high levels of ctDNA – colorectal, cholangiocarcinoma, ovarian, breast, and melanoma – and in 65% of samples from low-ctDNA cancers – pancreatic, renal, and glioma.
Another model focused just on fragmentation patterns and was still able to distinguish cancer samples from those of healthy controls, although with slightly reduced area under the curve.
“Our results indicate that exploiting fundamental properties of cfDNA with fragment-specific analyses can allow more sensitive evaluation of ctDNA,” the authors wrote. “We identified features that could determine the presence and amount of ctDNA in plasma samples, without a prior knowledge of somatic aberrations.”
The authors pointed out that size selection of DNA fragments was relatively simple and cheap, and was also compatible with other genome-wide and targeted genomic analyses, “greatly increasing the potential value and utility of liquid biopsies as well as the cost-effectiveness of cfDNA sequencing.”
However, they cautioned that their catalogue had focused solely on double-stranded DNA and was subject to potential biases from the DNA extraction and sequencing methods they used in the study. They also commented that other biological effects could help refine the analysis of ctDNA.
“Other bodily fluids [urine, cerebrospinal fluid, and saliva], different nucleic acids and structures, altered mechanisms of release into circulation, or sample processing methods could exhibit varying fragment size signatures and could offer additional exploitable biological patterns for selective sequencing,” they wrote.
The study was supported by the University of Cambridge, Cancer Research UK, and the Engineering and Physical Sciences Research Council. Research supporting the study was also funded by the European Research Council, the National Institute for Health Research Cambridge, National Cancer Research Network, Cambridge Experimental Cancer Medicine Centre, Hutchison Whampoa, Target Ovarian Cancer, the Medical Research Council, and AstraZeneca. Three authors are cofounders, shareholders, and officers/consultants in a company specializing in ctDNA analysis. One author declared research funding and advisory board fees from private industry. Seven authors are listed on related patents.
SOURCE: Mouliere F et al. Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aat4921.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: The size of cell-free DNA could be used to single out circulating tumor DNA.
Major finding: Circulating tumor DNA fragments are more commonly found in the 90-150 base pair range.
Study details: A study of 344 plasma samples from 200 patients with 18 cancer types and 65 samples from healthy controls.
Disclosures: The study was supported by the University of Cambridge, Cancer Research UK, and the Engineering and Physical Sciences Research Council. Research supporting the study was also funded by the European Research Council, the National Institute for Health Research Cambridge, National Cancer Research Network, Cambridge Experimental Cancer Medicine Centre, Hutchison Whampoa, Target Ovarian Cancer, the Medical Research Council, and AstraZeneca. Three authors are cofounders, shareholders, and officers/consultants in a company specializing in circulating tumor DNA analysis. One author declared research funding and advisory board fees from private industry. Seven authors are listed on related patents.
Source: Mouliere F et al. Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aat4921.
Cognitive and Behavioral Problems Increase With ALS Disease Stage
Data raise the question of whether cognitive and behavioral change should be included in ALS diagnostic criteria.
Cognitive deficits and behavioral symptoms that are specific to amyotrophic lateral sclerosis (ALS) occur more frequently as the disease progresses, according to research published online ahead of print September 12 in Neurology. Few patients are free of neuropsychologic impairment when they reach the final stage of the disease. It might be appropriate to include cognitive and behavioral change in the diagnostic criteria and future staging systems for ALS, said the authors.
In 2013, Elamin et al suggested that cognitive change in ALS may be associated with indirect measures of disease progression, such as total score on the ALS Functional Rating Scale-Revised. Christopher Crockford, PhD, a researcher at the University of Edinburgh, and colleagues sought to determine whether the cognitive and behavioral symptoms in ALS were more prevalent at more advanced stages of disease.
The Role of Letter Fluency Impairment
They conducted a multicenter, cross-sectional, observational study that included 161 patients from Edinburgh, Dublin, and London with possible, probable, or definite ALS, according to revised El Escorial diagnostic criteria. The researchers also recruited 80 healthy, matched controls. Through interviews, Dr. Crockford and colleagues elicited demographic and clinical data. They measured participants’ clinical staging with the King’s Clinical Staging System and neuropsychologic status with the Edinburgh Cognitive and Behavioral ALS Screen (ECAS).
The investigators did not observe significant differences between the patient and control groups in background variables. Approximately 67% of patients with ALS were male, compared with 60% of controls. Mean age at testing was approximately 61 in both groups. Among patients with ALS, 40 were in Stage 1, 45 were in Stage 2, 22 were in Stage 3, and 54 were in Stage 4.
Compared with controls, patients with ALS had significantly worse cognitive performance on all domains of the ECAS except visuospatial functioning. Dr. Crockford’s group found a significant cross-sectional effect across disease stages for ALS-specific functions (eg, executive, language, and letter fluency) and ECAS total score. They did not find a similar effect for ALS-nonspecific functions (eg, memory and visuospatial). In addition, rates of ALS-specific impairment and behavioral change increased with increasing disease stage.
Letter fluency impairment may explain the relationship between cognitive function and disease stage, said the authors. They observed higher rates of all behavioral problems in later King’s stages. Bulbar signs were significantly related to ALS-specific scores, ECAS total score, and behavioral scores. Site of onset was not related to these scores, however.
Intervention programs to alleviate the effect of patients’ neuropsychologic impairment on caregivers may be appropriate, said the authors. “Furthermore, clinicians should be cognizant of current neuropsychologic status when prescribing life-prolonging interventions to patients and implement support structures for those with a neuropsychologic impairment,” they added.
Informing Patients and Caregivers
Although Dr. Crockford and colleagues focused on the behavioral and cognitive effects of ALS, the disease may affect mental health as well, said Paul Wicks, PhD, Vice President of Innovation at PatientsLikeMe in Cambridge, Massachusetts, and Steven M. Albert, PhD, Professor and Chair of Behavioral and Community Health Sciences at the University of Pittsburgh, in an accompanying editorial. The data show that the rates of major depression and depressed mood increase with increasing disease stage.
Drs. Wicks and Albert cited a survey in which 90% of patients and caregivers reported that their doctors had not told them that cognitive or psychologic symptoms can arise in ALS. “In our experience, colleagues report keeping the information from patients in order to spare them further distress,” they said. Yet most respondents to this survey reported that they would have liked to have been informed about these symptoms.
“Educating patients and caregivers that cognitive change is a part of ALS should be no different from similar discussions to be had in multiple sclerosis, Parkinson disease, and a range of other conditions,” said Drs. Wicks and Albert. “Keeping the truth from patients and caregivers is not protective; it is paternalistic, and it is time to stop. Only by facing up to the hard truth that one of the most dreaded conditions in medicine is even worse than we previously acknowledged can we take stock, marshal our resources, and make renewed plans to defeat our common enemy.”
Suggested Reading
Crockford C, Newton J, Lonergan K, et al. ALS-specific cognitive and behavior changes associated with advancing disease stage in ALS. Neurology. 2018 Sep 12 [Epub ahead of print].
Wicks P, Albert SM. It’s time to stop saying “the mind is unaffected” in ALS. Neurology. 2018 Sep 12 [Epub ahead of print].
Data raise the question of whether cognitive and behavioral change should be included in ALS diagnostic criteria.
Data raise the question of whether cognitive and behavioral change should be included in ALS diagnostic criteria.
Cognitive deficits and behavioral symptoms that are specific to amyotrophic lateral sclerosis (ALS) occur more frequently as the disease progresses, according to research published online ahead of print September 12 in Neurology. Few patients are free of neuropsychologic impairment when they reach the final stage of the disease. It might be appropriate to include cognitive and behavioral change in the diagnostic criteria and future staging systems for ALS, said the authors.
In 2013, Elamin et al suggested that cognitive change in ALS may be associated with indirect measures of disease progression, such as total score on the ALS Functional Rating Scale-Revised. Christopher Crockford, PhD, a researcher at the University of Edinburgh, and colleagues sought to determine whether the cognitive and behavioral symptoms in ALS were more prevalent at more advanced stages of disease.
The Role of Letter Fluency Impairment
They conducted a multicenter, cross-sectional, observational study that included 161 patients from Edinburgh, Dublin, and London with possible, probable, or definite ALS, according to revised El Escorial diagnostic criteria. The researchers also recruited 80 healthy, matched controls. Through interviews, Dr. Crockford and colleagues elicited demographic and clinical data. They measured participants’ clinical staging with the King’s Clinical Staging System and neuropsychologic status with the Edinburgh Cognitive and Behavioral ALS Screen (ECAS).
The investigators did not observe significant differences between the patient and control groups in background variables. Approximately 67% of patients with ALS were male, compared with 60% of controls. Mean age at testing was approximately 61 in both groups. Among patients with ALS, 40 were in Stage 1, 45 were in Stage 2, 22 were in Stage 3, and 54 were in Stage 4.
Compared with controls, patients with ALS had significantly worse cognitive performance on all domains of the ECAS except visuospatial functioning. Dr. Crockford’s group found a significant cross-sectional effect across disease stages for ALS-specific functions (eg, executive, language, and letter fluency) and ECAS total score. They did not find a similar effect for ALS-nonspecific functions (eg, memory and visuospatial). In addition, rates of ALS-specific impairment and behavioral change increased with increasing disease stage.
Letter fluency impairment may explain the relationship between cognitive function and disease stage, said the authors. They observed higher rates of all behavioral problems in later King’s stages. Bulbar signs were significantly related to ALS-specific scores, ECAS total score, and behavioral scores. Site of onset was not related to these scores, however.
Intervention programs to alleviate the effect of patients’ neuropsychologic impairment on caregivers may be appropriate, said the authors. “Furthermore, clinicians should be cognizant of current neuropsychologic status when prescribing life-prolonging interventions to patients and implement support structures for those with a neuropsychologic impairment,” they added.
Informing Patients and Caregivers
Although Dr. Crockford and colleagues focused on the behavioral and cognitive effects of ALS, the disease may affect mental health as well, said Paul Wicks, PhD, Vice President of Innovation at PatientsLikeMe in Cambridge, Massachusetts, and Steven M. Albert, PhD, Professor and Chair of Behavioral and Community Health Sciences at the University of Pittsburgh, in an accompanying editorial. The data show that the rates of major depression and depressed mood increase with increasing disease stage.
Drs. Wicks and Albert cited a survey in which 90% of patients and caregivers reported that their doctors had not told them that cognitive or psychologic symptoms can arise in ALS. “In our experience, colleagues report keeping the information from patients in order to spare them further distress,” they said. Yet most respondents to this survey reported that they would have liked to have been informed about these symptoms.
“Educating patients and caregivers that cognitive change is a part of ALS should be no different from similar discussions to be had in multiple sclerosis, Parkinson disease, and a range of other conditions,” said Drs. Wicks and Albert. “Keeping the truth from patients and caregivers is not protective; it is paternalistic, and it is time to stop. Only by facing up to the hard truth that one of the most dreaded conditions in medicine is even worse than we previously acknowledged can we take stock, marshal our resources, and make renewed plans to defeat our common enemy.”
Suggested Reading
Crockford C, Newton J, Lonergan K, et al. ALS-specific cognitive and behavior changes associated with advancing disease stage in ALS. Neurology. 2018 Sep 12 [Epub ahead of print].
Wicks P, Albert SM. It’s time to stop saying “the mind is unaffected” in ALS. Neurology. 2018 Sep 12 [Epub ahead of print].
Cognitive deficits and behavioral symptoms that are specific to amyotrophic lateral sclerosis (ALS) occur more frequently as the disease progresses, according to research published online ahead of print September 12 in Neurology. Few patients are free of neuropsychologic impairment when they reach the final stage of the disease. It might be appropriate to include cognitive and behavioral change in the diagnostic criteria and future staging systems for ALS, said the authors.
In 2013, Elamin et al suggested that cognitive change in ALS may be associated with indirect measures of disease progression, such as total score on the ALS Functional Rating Scale-Revised. Christopher Crockford, PhD, a researcher at the University of Edinburgh, and colleagues sought to determine whether the cognitive and behavioral symptoms in ALS were more prevalent at more advanced stages of disease.
The Role of Letter Fluency Impairment
They conducted a multicenter, cross-sectional, observational study that included 161 patients from Edinburgh, Dublin, and London with possible, probable, or definite ALS, according to revised El Escorial diagnostic criteria. The researchers also recruited 80 healthy, matched controls. Through interviews, Dr. Crockford and colleagues elicited demographic and clinical data. They measured participants’ clinical staging with the King’s Clinical Staging System and neuropsychologic status with the Edinburgh Cognitive and Behavioral ALS Screen (ECAS).
The investigators did not observe significant differences between the patient and control groups in background variables. Approximately 67% of patients with ALS were male, compared with 60% of controls. Mean age at testing was approximately 61 in both groups. Among patients with ALS, 40 were in Stage 1, 45 were in Stage 2, 22 were in Stage 3, and 54 were in Stage 4.
Compared with controls, patients with ALS had significantly worse cognitive performance on all domains of the ECAS except visuospatial functioning. Dr. Crockford’s group found a significant cross-sectional effect across disease stages for ALS-specific functions (eg, executive, language, and letter fluency) and ECAS total score. They did not find a similar effect for ALS-nonspecific functions (eg, memory and visuospatial). In addition, rates of ALS-specific impairment and behavioral change increased with increasing disease stage.
Letter fluency impairment may explain the relationship between cognitive function and disease stage, said the authors. They observed higher rates of all behavioral problems in later King’s stages. Bulbar signs were significantly related to ALS-specific scores, ECAS total score, and behavioral scores. Site of onset was not related to these scores, however.
Intervention programs to alleviate the effect of patients’ neuropsychologic impairment on caregivers may be appropriate, said the authors. “Furthermore, clinicians should be cognizant of current neuropsychologic status when prescribing life-prolonging interventions to patients and implement support structures for those with a neuropsychologic impairment,” they added.
Informing Patients and Caregivers
Although Dr. Crockford and colleagues focused on the behavioral and cognitive effects of ALS, the disease may affect mental health as well, said Paul Wicks, PhD, Vice President of Innovation at PatientsLikeMe in Cambridge, Massachusetts, and Steven M. Albert, PhD, Professor and Chair of Behavioral and Community Health Sciences at the University of Pittsburgh, in an accompanying editorial. The data show that the rates of major depression and depressed mood increase with increasing disease stage.
Drs. Wicks and Albert cited a survey in which 90% of patients and caregivers reported that their doctors had not told them that cognitive or psychologic symptoms can arise in ALS. “In our experience, colleagues report keeping the information from patients in order to spare them further distress,” they said. Yet most respondents to this survey reported that they would have liked to have been informed about these symptoms.
“Educating patients and caregivers that cognitive change is a part of ALS should be no different from similar discussions to be had in multiple sclerosis, Parkinson disease, and a range of other conditions,” said Drs. Wicks and Albert. “Keeping the truth from patients and caregivers is not protective; it is paternalistic, and it is time to stop. Only by facing up to the hard truth that one of the most dreaded conditions in medicine is even worse than we previously acknowledged can we take stock, marshal our resources, and make renewed plans to defeat our common enemy.”
Suggested Reading
Crockford C, Newton J, Lonergan K, et al. ALS-specific cognitive and behavior changes associated with advancing disease stage in ALS. Neurology. 2018 Sep 12 [Epub ahead of print].
Wicks P, Albert SM. It’s time to stop saying “the mind is unaffected” in ALS. Neurology. 2018 Sep 12 [Epub ahead of print].
DEA Reclassifies Epidiolex as Schedule V
The regulatory action acknowledges the drug’s medical use and clears the way for it to be marketed.
WASHINGTON, DC
The DEA’s final rescheduling order is limited to drugs approved by the FDA that contain cannabis-derived CBD and no more than 0.1% tetrahydrocannabinol (THC). In practice, this means that the rescheduling currently applies only to Epidiolex, since this is the only formulation of CBD that has received FDA approval.
A Low Potential for Abuse
“We are pleased that the DEA has placed Epidiolex in the lowest restriction schedule, because it will help ensure that patients with LGS and Dravet syndrome, two of the most debilitating forms of epilepsy, can access this important new treatment option through their physicians,” said GW Pharmaceutical’s Chief Executive Officer Justin Gover in a statement.
During the FDA advisory committee meeting for the approval of Epidiolex to treat LGS and Dravet syndrome, the FDA and GW Pharmaceuticals concluded that the potential to abuse CBD was low, since it does not contain THC, the primary psychoactive component of cannabis.
All other marijuana products are currently classified as Schedule I, along with illegal substances such as heroin and cocaine.
An Established Medical Use
Epidiolex had received fast track and rare pediatric designations from the FDA for LGS and Dravet syndrome; the approval was based on three pivotal randomized, double-blind, placebo-controlled clinical trials. The drug met its primary end point of reduced seizure frequency in all trials when added to standard of care for patients with drug-resistant LGS and those with Dravet syndrome.
Safety evaluations assessed data from 1,756 patients and found that the 20 deaths seen during the study period were not clearly linked to Epidiolex and may be expected for children with severe seizure disorders.
In supplementary information accompanying the order, the DEA’s Acting Administrator Uttam Dhillon noted that the FDA’s approval of Epidiolex means that “it has a currently accepted medical use in treatment for purposes of the Controlled Substances Act [CSA]. Accordingly, Epidiolex no longer meets the criteria for placement in Schedule I of the CSA.” Schedule I drugs do not have a currently accepted medical use.
Schedule V drugs, according to the DEA, are defined as “drugs with lower potential for abuse than Schedule IV and [that] consist of preparations containing limited quantities of certain narcotics.” Other Schedule V drugs include cough medicine with less than 200 mg of codeine or per 100 mL, antidiarrheal medications, pregabalin, and the antiepileptic drugs brivaracetam and lacosamide. “Schedule V drugs represent the least potential for abuse,” according to the DEA.
Initial dosing recommendations for Epidiolex are to titrate to a dose of 10 mg/kg/day. Dose adjustments to 20 mg/kg/day are permissible, depending on clinical response and tolerability. The manufacturer has submitted a marketing agreement to the European Medicines Agency and has received orphan drug designation for Epidiolex in the treatment of tuberous sclerosis complex.
—Kari Oakes
The regulatory action acknowledges the drug’s medical use and clears the way for it to be marketed.
The regulatory action acknowledges the drug’s medical use and clears the way for it to be marketed.
WASHINGTON, DC
The DEA’s final rescheduling order is limited to drugs approved by the FDA that contain cannabis-derived CBD and no more than 0.1% tetrahydrocannabinol (THC). In practice, this means that the rescheduling currently applies only to Epidiolex, since this is the only formulation of CBD that has received FDA approval.
A Low Potential for Abuse
“We are pleased that the DEA has placed Epidiolex in the lowest restriction schedule, because it will help ensure that patients with LGS and Dravet syndrome, two of the most debilitating forms of epilepsy, can access this important new treatment option through their physicians,” said GW Pharmaceutical’s Chief Executive Officer Justin Gover in a statement.
During the FDA advisory committee meeting for the approval of Epidiolex to treat LGS and Dravet syndrome, the FDA and GW Pharmaceuticals concluded that the potential to abuse CBD was low, since it does not contain THC, the primary psychoactive component of cannabis.
All other marijuana products are currently classified as Schedule I, along with illegal substances such as heroin and cocaine.
An Established Medical Use
Epidiolex had received fast track and rare pediatric designations from the FDA for LGS and Dravet syndrome; the approval was based on three pivotal randomized, double-blind, placebo-controlled clinical trials. The drug met its primary end point of reduced seizure frequency in all trials when added to standard of care for patients with drug-resistant LGS and those with Dravet syndrome.
Safety evaluations assessed data from 1,756 patients and found that the 20 deaths seen during the study period were not clearly linked to Epidiolex and may be expected for children with severe seizure disorders.
In supplementary information accompanying the order, the DEA’s Acting Administrator Uttam Dhillon noted that the FDA’s approval of Epidiolex means that “it has a currently accepted medical use in treatment for purposes of the Controlled Substances Act [CSA]. Accordingly, Epidiolex no longer meets the criteria for placement in Schedule I of the CSA.” Schedule I drugs do not have a currently accepted medical use.
Schedule V drugs, according to the DEA, are defined as “drugs with lower potential for abuse than Schedule IV and [that] consist of preparations containing limited quantities of certain narcotics.” Other Schedule V drugs include cough medicine with less than 200 mg of codeine or per 100 mL, antidiarrheal medications, pregabalin, and the antiepileptic drugs brivaracetam and lacosamide. “Schedule V drugs represent the least potential for abuse,” according to the DEA.
Initial dosing recommendations for Epidiolex are to titrate to a dose of 10 mg/kg/day. Dose adjustments to 20 mg/kg/day are permissible, depending on clinical response and tolerability. The manufacturer has submitted a marketing agreement to the European Medicines Agency and has received orphan drug designation for Epidiolex in the treatment of tuberous sclerosis complex.
—Kari Oakes
WASHINGTON, DC
The DEA’s final rescheduling order is limited to drugs approved by the FDA that contain cannabis-derived CBD and no more than 0.1% tetrahydrocannabinol (THC). In practice, this means that the rescheduling currently applies only to Epidiolex, since this is the only formulation of CBD that has received FDA approval.
A Low Potential for Abuse
“We are pleased that the DEA has placed Epidiolex in the lowest restriction schedule, because it will help ensure that patients with LGS and Dravet syndrome, two of the most debilitating forms of epilepsy, can access this important new treatment option through their physicians,” said GW Pharmaceutical’s Chief Executive Officer Justin Gover in a statement.
During the FDA advisory committee meeting for the approval of Epidiolex to treat LGS and Dravet syndrome, the FDA and GW Pharmaceuticals concluded that the potential to abuse CBD was low, since it does not contain THC, the primary psychoactive component of cannabis.
All other marijuana products are currently classified as Schedule I, along with illegal substances such as heroin and cocaine.
An Established Medical Use
Epidiolex had received fast track and rare pediatric designations from the FDA for LGS and Dravet syndrome; the approval was based on three pivotal randomized, double-blind, placebo-controlled clinical trials. The drug met its primary end point of reduced seizure frequency in all trials when added to standard of care for patients with drug-resistant LGS and those with Dravet syndrome.
Safety evaluations assessed data from 1,756 patients and found that the 20 deaths seen during the study period were not clearly linked to Epidiolex and may be expected for children with severe seizure disorders.
In supplementary information accompanying the order, the DEA’s Acting Administrator Uttam Dhillon noted that the FDA’s approval of Epidiolex means that “it has a currently accepted medical use in treatment for purposes of the Controlled Substances Act [CSA]. Accordingly, Epidiolex no longer meets the criteria for placement in Schedule I of the CSA.” Schedule I drugs do not have a currently accepted medical use.
Schedule V drugs, according to the DEA, are defined as “drugs with lower potential for abuse than Schedule IV and [that] consist of preparations containing limited quantities of certain narcotics.” Other Schedule V drugs include cough medicine with less than 200 mg of codeine or per 100 mL, antidiarrheal medications, pregabalin, and the antiepileptic drugs brivaracetam and lacosamide. “Schedule V drugs represent the least potential for abuse,” according to the DEA.
Initial dosing recommendations for Epidiolex are to titrate to a dose of 10 mg/kg/day. Dose adjustments to 20 mg/kg/day are permissible, depending on clinical response and tolerability. The manufacturer has submitted a marketing agreement to the European Medicines Agency and has received orphan drug designation for Epidiolex in the treatment of tuberous sclerosis complex.
—Kari Oakes
Nipple-sparing mastectomy safe in older patients
BOSTON – For women undergoing results of recent studies suggest.
The procedure was “surgically safe” in older patients, with complication rates comparable to those seen in younger patients, Solange E. Cox, MD, of MedStar Georgetown University Hospital, Washington, said in a presentation of one those two retrospective analyses at the annual clinical congress of the American College of Surgeons.
“From this, we think that eligible older patients should be offered a nipple-sparing mastectomy as a surgical option for breast cancer, and age alone should not be used as criteria to exclude these patients from the option,” she said.
The second retrospective study showed that patients undergoing neoadjuvant chemotherapy had a rate of surgical complications and unintended reoperations comparable to what was seen in women undergoing primary surgery.
“Our big-picture takeaway from this study is that receipt of neoadjuvant chemotherapy is not a contraindication for nipple-sparing mastectomy,” said investigator Alex J. Bartholomew, MS, also of Medstar Georgetown University Hospital.
Mr. Bartholomew’s conclusion was based on an analysis of the nipple-sparing mastectomy registry of the American Society of Breast Surgeons that included a total of 3,125 breasts. Neoadjuvant chemotherapy was used in 528, or 16.9%, while primary surgery was performed in 2,597, or 83.1%.
The overall rate of complications was 11%, with nonsignificant differences between the neoadjuvant chemotherapy and primary surgery groups at 12.7% and 10.7%, respectively.
The rate of unintended reoperation, at 4.9%, was not significantly different in the neoadjuvant chemotherapy and primary surgery groups, at 5.2% and 4.8%, Mr. Bartholomew said. Similarly, he found that the rate of nipple areolar complex loss of 1% overall was not different between groups.
Advanced age was likewise not associated with increased complications in the study presented by Dr. Cox, which was a retrospective review of data for patients undergoing nipple-sparing mastectomy from 1998 to 2015 at a single institution. That cohort included 38 patients age 60 years or older, and 358 younger patients.
The rate of complications was 15.5% for patients over age 60 years, and similarly, 13.0% for their younger counterparts (P = .590), Dr. Cox reported. Likewise, the rate of unintended operations was 13.3% and 15.3% for older and younger patients, respectively (P = .274).
These findings are important because advancing age has been associated with a decrease in the likelihood of nipple-sparing mastectomy, according to Dr. Cox.
For mastectomies in general, advanced age has been implicated as a potential risk factor for necrosis, technical complications, and poor outcomes with mastectomies. However, no prior studies had been done specifically to evaluate nipple-sparing mastectomies in older breast cancer patients, Dr. Cox said.
Nipple-sparing mastectomy provides both cosmetic and psychosocial benefits to patients, according to the researchers, because the procedure spares the nipple-areolar complex.
The researchers who had no relevant disclosures.
SOURCES: Cox S et al. SF310 abstract; Bartholomew AJ et al. SF310 abstract ACS Clinical Congress 2018
BOSTON – For women undergoing results of recent studies suggest.
The procedure was “surgically safe” in older patients, with complication rates comparable to those seen in younger patients, Solange E. Cox, MD, of MedStar Georgetown University Hospital, Washington, said in a presentation of one those two retrospective analyses at the annual clinical congress of the American College of Surgeons.
“From this, we think that eligible older patients should be offered a nipple-sparing mastectomy as a surgical option for breast cancer, and age alone should not be used as criteria to exclude these patients from the option,” she said.
The second retrospective study showed that patients undergoing neoadjuvant chemotherapy had a rate of surgical complications and unintended reoperations comparable to what was seen in women undergoing primary surgery.
“Our big-picture takeaway from this study is that receipt of neoadjuvant chemotherapy is not a contraindication for nipple-sparing mastectomy,” said investigator Alex J. Bartholomew, MS, also of Medstar Georgetown University Hospital.
Mr. Bartholomew’s conclusion was based on an analysis of the nipple-sparing mastectomy registry of the American Society of Breast Surgeons that included a total of 3,125 breasts. Neoadjuvant chemotherapy was used in 528, or 16.9%, while primary surgery was performed in 2,597, or 83.1%.
The overall rate of complications was 11%, with nonsignificant differences between the neoadjuvant chemotherapy and primary surgery groups at 12.7% and 10.7%, respectively.
The rate of unintended reoperation, at 4.9%, was not significantly different in the neoadjuvant chemotherapy and primary surgery groups, at 5.2% and 4.8%, Mr. Bartholomew said. Similarly, he found that the rate of nipple areolar complex loss of 1% overall was not different between groups.
Advanced age was likewise not associated with increased complications in the study presented by Dr. Cox, which was a retrospective review of data for patients undergoing nipple-sparing mastectomy from 1998 to 2015 at a single institution. That cohort included 38 patients age 60 years or older, and 358 younger patients.
The rate of complications was 15.5% for patients over age 60 years, and similarly, 13.0% for their younger counterparts (P = .590), Dr. Cox reported. Likewise, the rate of unintended operations was 13.3% and 15.3% for older and younger patients, respectively (P = .274).
These findings are important because advancing age has been associated with a decrease in the likelihood of nipple-sparing mastectomy, according to Dr. Cox.
For mastectomies in general, advanced age has been implicated as a potential risk factor for necrosis, technical complications, and poor outcomes with mastectomies. However, no prior studies had been done specifically to evaluate nipple-sparing mastectomies in older breast cancer patients, Dr. Cox said.
Nipple-sparing mastectomy provides both cosmetic and psychosocial benefits to patients, according to the researchers, because the procedure spares the nipple-areolar complex.
The researchers who had no relevant disclosures.
SOURCES: Cox S et al. SF310 abstract; Bartholomew AJ et al. SF310 abstract ACS Clinical Congress 2018
BOSTON – For women undergoing results of recent studies suggest.
The procedure was “surgically safe” in older patients, with complication rates comparable to those seen in younger patients, Solange E. Cox, MD, of MedStar Georgetown University Hospital, Washington, said in a presentation of one those two retrospective analyses at the annual clinical congress of the American College of Surgeons.
“From this, we think that eligible older patients should be offered a nipple-sparing mastectomy as a surgical option for breast cancer, and age alone should not be used as criteria to exclude these patients from the option,” she said.
The second retrospective study showed that patients undergoing neoadjuvant chemotherapy had a rate of surgical complications and unintended reoperations comparable to what was seen in women undergoing primary surgery.
“Our big-picture takeaway from this study is that receipt of neoadjuvant chemotherapy is not a contraindication for nipple-sparing mastectomy,” said investigator Alex J. Bartholomew, MS, also of Medstar Georgetown University Hospital.
Mr. Bartholomew’s conclusion was based on an analysis of the nipple-sparing mastectomy registry of the American Society of Breast Surgeons that included a total of 3,125 breasts. Neoadjuvant chemotherapy was used in 528, or 16.9%, while primary surgery was performed in 2,597, or 83.1%.
The overall rate of complications was 11%, with nonsignificant differences between the neoadjuvant chemotherapy and primary surgery groups at 12.7% and 10.7%, respectively.
The rate of unintended reoperation, at 4.9%, was not significantly different in the neoadjuvant chemotherapy and primary surgery groups, at 5.2% and 4.8%, Mr. Bartholomew said. Similarly, he found that the rate of nipple areolar complex loss of 1% overall was not different between groups.
Advanced age was likewise not associated with increased complications in the study presented by Dr. Cox, which was a retrospective review of data for patients undergoing nipple-sparing mastectomy from 1998 to 2015 at a single institution. That cohort included 38 patients age 60 years or older, and 358 younger patients.
The rate of complications was 15.5% for patients over age 60 years, and similarly, 13.0% for their younger counterparts (P = .590), Dr. Cox reported. Likewise, the rate of unintended operations was 13.3% and 15.3% for older and younger patients, respectively (P = .274).
These findings are important because advancing age has been associated with a decrease in the likelihood of nipple-sparing mastectomy, according to Dr. Cox.
For mastectomies in general, advanced age has been implicated as a potential risk factor for necrosis, technical complications, and poor outcomes with mastectomies. However, no prior studies had been done specifically to evaluate nipple-sparing mastectomies in older breast cancer patients, Dr. Cox said.
Nipple-sparing mastectomy provides both cosmetic and psychosocial benefits to patients, according to the researchers, because the procedure spares the nipple-areolar complex.
The researchers who had no relevant disclosures.
SOURCES: Cox S et al. SF310 abstract; Bartholomew AJ et al. SF310 abstract ACS Clinical Congress 2018
REPORTING FROM THE ACS CLINICAL CONGRESS
Key clinical point: Neoadjuvant chemotherapy and advancing age were not associated with increased rates of complications in women undergoing nipple-sparing mastectomy in two recent studies.
Major finding: The overall rate of complications was not significantly different for neoadjuvant chemotherapy vs. primary surgery (12.7% vs. 10.7%) or for age over 60 years vs. younger age (15.5% vs. 13.0%).
Study details: Retrospective studies of a nipple-sparing mastectomy registry including more than 3,000 breasts (neoadjuvant chemotherapy vs. primary surgery study) and a single-institution study of nearly 400 patients (older vs. younger study).
Disclosures: The authors reported no conflicts of interest.
Source: Cox S et al. and Bartholomew AJ et al. Session SF310 ACS Clinical Congress 2018.
FDA approves second pegfilgrastim biosimilar
The Food and Drug Administration has approved a second biosimilar to pegfilgrastim (Neulasta) to decrease the chance of infection in patients with nonmyeloid cancer who are receiving myelosuppressive chemotherapy and are at risk of febrile neutropenia.
Approval of pegfilgrastim-cbqv, previously known as CHS-1701, was based on analyses establishing biosimilarity, including pharmacokinetic, pharmacodynamic, and immunogenicity studies. Clinical trial results were presented at the 2017 ASCO Annual Meeting.
The most common adverse reactions with pegfilgrastim-cbqv are bone pain and pain in extremities.
The FDA approved the first pegfilgrastim biosimilar, pegfilgrastim-jmdb (Fulphila) in June.
Pegfilgrastim-cbqv will be marketed as Udenyca by Coherus BioSciences.
“Udenyca’s robust clinical package includes a dedicated immunogenicity similarity study in over 300 healthy subjects,” Barbara Finck, MD, chief medical officer of Coherus BioSciences, said in a press release.
“In support of that study, and as part of our commitment to ensuring patient safety, we deployed a battery of sensitive immunogenicity assays. This effort not only supported the biosimilarity of Udenyca but also advanced the understanding of the immunogenic response of pegfilgrastim products.”
Coherus BioSciences plans to provide details about pricing and the launch of pegfilgrastim-cbqv during an earnings call on Nov. 8.
The Food and Drug Administration has approved a second biosimilar to pegfilgrastim (Neulasta) to decrease the chance of infection in patients with nonmyeloid cancer who are receiving myelosuppressive chemotherapy and are at risk of febrile neutropenia.
Approval of pegfilgrastim-cbqv, previously known as CHS-1701, was based on analyses establishing biosimilarity, including pharmacokinetic, pharmacodynamic, and immunogenicity studies. Clinical trial results were presented at the 2017 ASCO Annual Meeting.
The most common adverse reactions with pegfilgrastim-cbqv are bone pain and pain in extremities.
The FDA approved the first pegfilgrastim biosimilar, pegfilgrastim-jmdb (Fulphila) in June.
Pegfilgrastim-cbqv will be marketed as Udenyca by Coherus BioSciences.
“Udenyca’s robust clinical package includes a dedicated immunogenicity similarity study in over 300 healthy subjects,” Barbara Finck, MD, chief medical officer of Coherus BioSciences, said in a press release.
“In support of that study, and as part of our commitment to ensuring patient safety, we deployed a battery of sensitive immunogenicity assays. This effort not only supported the biosimilarity of Udenyca but also advanced the understanding of the immunogenic response of pegfilgrastim products.”
Coherus BioSciences plans to provide details about pricing and the launch of pegfilgrastim-cbqv during an earnings call on Nov. 8.
The Food and Drug Administration has approved a second biosimilar to pegfilgrastim (Neulasta) to decrease the chance of infection in patients with nonmyeloid cancer who are receiving myelosuppressive chemotherapy and are at risk of febrile neutropenia.
Approval of pegfilgrastim-cbqv, previously known as CHS-1701, was based on analyses establishing biosimilarity, including pharmacokinetic, pharmacodynamic, and immunogenicity studies. Clinical trial results were presented at the 2017 ASCO Annual Meeting.
The most common adverse reactions with pegfilgrastim-cbqv are bone pain and pain in extremities.
The FDA approved the first pegfilgrastim biosimilar, pegfilgrastim-jmdb (Fulphila) in June.
Pegfilgrastim-cbqv will be marketed as Udenyca by Coherus BioSciences.
“Udenyca’s robust clinical package includes a dedicated immunogenicity similarity study in over 300 healthy subjects,” Barbara Finck, MD, chief medical officer of Coherus BioSciences, said in a press release.
“In support of that study, and as part of our commitment to ensuring patient safety, we deployed a battery of sensitive immunogenicity assays. This effort not only supported the biosimilarity of Udenyca but also advanced the understanding of the immunogenic response of pegfilgrastim products.”
Coherus BioSciences plans to provide details about pricing and the launch of pegfilgrastim-cbqv during an earnings call on Nov. 8.
Debunking Psoriasis Myths: Remove Psoriasis Scales Gently
Myth: Pick Psoriasis Scales to Remove Them
Patients may be inclined to pick psoriasis scales that appear in noticeable areas or on the scalp. However, they should be counseled to avoid this practice, which could cause an infection. Instead, Dr. Steven Feldman (Winston-Salem, North Carolina) suggests putting on an ointment or oil-like medication to soften the scale. “Almost any kind of moisturizer will change the reflective properties of the scale so that you don’t see the scale,” he advised. He also suggested descaling agents such as topical salicylic acid or lactic acid. His patient education video is available on the American Academy of Dermatology website should you wish to direct your patients to it.
Because salicylic acid is a keratolytic (or peeling agent), it works by causing the outer layer of skin to shed. When applied topically, it helps to soften and lift psoriasis scales. Coal tar over-the-counter products also can be used for the same purpose. The over-the-counter product guide from the National Psoriasis Foundation is a valuable resource to share with patients.
Expert Commentary
I agree that it is very important to treat scale very gently. In addition to risk for infection, picking and traumatizing scale can lead to worsening of the psoriasis. This is known as the Koebner phenomenon. The phenomenon was first described by Heinrich Koebner in 1876 as the formation of psoriatic lesions in uninvolved skin of patients with psoriasis after cutaneous trauma. This isomorphic phenomenon is now known to involve numerous diseases, among them vitiligo, lichen planus, and Darier disease.
—Jeffrey M. Weinberg, MD (New York, New York)
Feldman S. How should I remove psoriasis scale? American Academy of Dermatology website. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Accessed October 31, 2018.
National Psoriasis Foundation. Over-the-counter products. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Published June 2017. Accessed October 31, 2018.
Sagi L, Trau H. The Koebner phenomenon. Clin Dermatol. 2011;29:231-236.
Myth: Pick Psoriasis Scales to Remove Them
Patients may be inclined to pick psoriasis scales that appear in noticeable areas or on the scalp. However, they should be counseled to avoid this practice, which could cause an infection. Instead, Dr. Steven Feldman (Winston-Salem, North Carolina) suggests putting on an ointment or oil-like medication to soften the scale. “Almost any kind of moisturizer will change the reflective properties of the scale so that you don’t see the scale,” he advised. He also suggested descaling agents such as topical salicylic acid or lactic acid. His patient education video is available on the American Academy of Dermatology website should you wish to direct your patients to it.
Because salicylic acid is a keratolytic (or peeling agent), it works by causing the outer layer of skin to shed. When applied topically, it helps to soften and lift psoriasis scales. Coal tar over-the-counter products also can be used for the same purpose. The over-the-counter product guide from the National Psoriasis Foundation is a valuable resource to share with patients.
Expert Commentary
I agree that it is very important to treat scale very gently. In addition to risk for infection, picking and traumatizing scale can lead to worsening of the psoriasis. This is known as the Koebner phenomenon. The phenomenon was first described by Heinrich Koebner in 1876 as the formation of psoriatic lesions in uninvolved skin of patients with psoriasis after cutaneous trauma. This isomorphic phenomenon is now known to involve numerous diseases, among them vitiligo, lichen planus, and Darier disease.
—Jeffrey M. Weinberg, MD (New York, New York)
Myth: Pick Psoriasis Scales to Remove Them
Patients may be inclined to pick psoriasis scales that appear in noticeable areas or on the scalp. However, they should be counseled to avoid this practice, which could cause an infection. Instead, Dr. Steven Feldman (Winston-Salem, North Carolina) suggests putting on an ointment or oil-like medication to soften the scale. “Almost any kind of moisturizer will change the reflective properties of the scale so that you don’t see the scale,” he advised. He also suggested descaling agents such as topical salicylic acid or lactic acid. His patient education video is available on the American Academy of Dermatology website should you wish to direct your patients to it.
Because salicylic acid is a keratolytic (or peeling agent), it works by causing the outer layer of skin to shed. When applied topically, it helps to soften and lift psoriasis scales. Coal tar over-the-counter products also can be used for the same purpose. The over-the-counter product guide from the National Psoriasis Foundation is a valuable resource to share with patients.
Expert Commentary
I agree that it is very important to treat scale very gently. In addition to risk for infection, picking and traumatizing scale can lead to worsening of the psoriasis. This is known as the Koebner phenomenon. The phenomenon was first described by Heinrich Koebner in 1876 as the formation of psoriatic lesions in uninvolved skin of patients with psoriasis after cutaneous trauma. This isomorphic phenomenon is now known to involve numerous diseases, among them vitiligo, lichen planus, and Darier disease.
—Jeffrey M. Weinberg, MD (New York, New York)
Feldman S. How should I remove psoriasis scale? American Academy of Dermatology website. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Accessed October 31, 2018.
National Psoriasis Foundation. Over-the-counter products. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Published June 2017. Accessed October 31, 2018.
Sagi L, Trau H. The Koebner phenomenon. Clin Dermatol. 2011;29:231-236.
Feldman S. How should I remove psoriasis scale? American Academy of Dermatology website. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Accessed October 31, 2018.
National Psoriasis Foundation. Over-the-counter products. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Published June 2017. Accessed October 31, 2018.
Sagi L, Trau H. The Koebner phenomenon. Clin Dermatol. 2011;29:231-236.
Modeling Favors Immediate AED Treatment After an Unprovoked First Seizure
Factoring in quality of life, seizure risk, and side effects, a model prefers immediate over delayed treatment.
Immediate treatment of a first unprovoked seizure may be preferable to delayed treatment over a wide range of patients, including those who are at low risk of recurrent seizures, results of a decision analysis suggest.
Taking into account quality of life, seizure risk, and antiepileptic drug (AED) side effects, a model favored treatment of a patient with a single unprovoked seizure who did not meet the International League Against Epilepsy (ILAE) definition of epilepsy, investigators reported.
The model also favored treatment of patients who did meet ILAE criteria, namely, a 10-year recurrence risk greater than 60% in a patient with a single unprovoked seizure, according to the analysis, which was published in the October 9 issue of Neurology.
Together, these findings suggest that the current ILAE epilepsy definition is “too simplistic” for deciding whether to start or withhold AED treatment after a first unprovoked seizure, said M. Brandon Westover, MD, PhD, Associate Professor of Neurology at Massachusetts General Hospital in Boston, and his coauthors.
“A more precise and patient-personalized definition of epilepsy should encompass not only seizure recurrence probability but also a multitude of other risks and benefits associated with AED treatment,” they said.
Weighing Risks and Benefits
To determine which patients with a first unprovoked seizure might benefit from immediate AED treatment, Dr. Westover and his colleagues used a decision model with measures constructed from retrospective clinical trial data.
The goal of the simulation was to determine which treatment strategy—immediate or delayed AED treatment—would maximize the patient’s expected quality-adjusted life years (QALYs). Toward that end, Dr. Westover and his coinvestigators considered three base cases, which represented various degrees of seizure-recurrence risk.
The first case was a 30-year-old man with no risk factors for recurrent seizure other than having had a first seizure. In that case, immediate and deferred AED treatment resulted in 19.04 and 18.65 QALYs, respectively.
“In dollar values, using the conservative approximation of $50,000/QALY gained, this difference in treatment outcomes would amount to $19,500 gained per individual,” Dr. Westover and his coauthors wrote in their report.
The second case was a 30-year-old woman who presented with a first unprovoked seizure and had positive MRI results that establish a high risk of recurrence. As expected, because of the high recurrence risk, this scenario also favored immediate treatment, with 15.23 and 14.75 QALYs, respectively, for the immediate and deferred strategies.
The final case was a wheelchair-bound 60-year-old woman with a first unprovoked seizure and high risk of recurrence, but also a high risk of AED adverse effects and a smaller expected quality of life reduction from further seizures. In this scenario, in which treatment might be “intuitively discouraged” because of the AED side-effect risk, the cohort simulation favored deferred AED treatment by a small margin, the investigators said.
“A high baseline risk for recurrent seizures does not by itself always favor immediate AED treatment,” they said.
Findings May Shift Discussions About Therapy
The conclusion of this decision analysis by Dr. Westover and colleagues is “likely correct” that early treatment of a first unprovoked seizure could be favorable in a wide range of clinical scenarios, according to the authors of an accompanying editorial.
The decision analysis is based on a reasonable, though not comprehensive, set of parameters to simulate base cases representative of common first-ever seizure clinical scenarios, said editorialists Claire S. Jacobs, MD, PhD, and Jong Woo Lee, MD, PhD, both with the Department of Neurology at Brigham and Women’s Hospital in Boston.
Potentially the most controversial scenario addressed in the decision model, they noted, is the patient with low seizure recurrence risk but substantial quality of life decline upon recurrence. While that patient would not meet the commonly accepted 60% recurrence risk threshold that would indicate that treatment is warranted, this model favors immediate treatment because of the potentially disruptive effect of recurrence.
This study does not address important issues such as the cost of medication and patient preferences, they pointed out, and furthermore, QALYs can be difficult to integrate into clinical decision making.
Nonetheless, the findings are worth considering in clinical practice, the authors suggested. “At the very least, this study should, however subtly, shift the starting point of discussion with the patient toward a default of immediate, rather than deferred, treatment after a first unprovoked seizure and apparent absence of disease,” said Drs. Jacob and Lee.
The study was supported by NINDS.
—Andrew D. Bowser
Suggested Reading
Bao EL, Chao LY, Ni P, et al. Antiepileptic drug treatment after an unprovoked first seizure: a decision analysis. Neurology. 2018;91(15):e1429-e1439.
Jacobs CS, Lee JW. Immediate vs delayed treatment of first unprovoked seizure: to treat, or not to treat? Neurology. 2018;91(15):684-685.
Factoring in quality of life, seizure risk, and side effects, a model prefers immediate over delayed treatment.
Factoring in quality of life, seizure risk, and side effects, a model prefers immediate over delayed treatment.
Immediate treatment of a first unprovoked seizure may be preferable to delayed treatment over a wide range of patients, including those who are at low risk of recurrent seizures, results of a decision analysis suggest.
Taking into account quality of life, seizure risk, and antiepileptic drug (AED) side effects, a model favored treatment of a patient with a single unprovoked seizure who did not meet the International League Against Epilepsy (ILAE) definition of epilepsy, investigators reported.
The model also favored treatment of patients who did meet ILAE criteria, namely, a 10-year recurrence risk greater than 60% in a patient with a single unprovoked seizure, according to the analysis, which was published in the October 9 issue of Neurology.
Together, these findings suggest that the current ILAE epilepsy definition is “too simplistic” for deciding whether to start or withhold AED treatment after a first unprovoked seizure, said M. Brandon Westover, MD, PhD, Associate Professor of Neurology at Massachusetts General Hospital in Boston, and his coauthors.
“A more precise and patient-personalized definition of epilepsy should encompass not only seizure recurrence probability but also a multitude of other risks and benefits associated with AED treatment,” they said.
Weighing Risks and Benefits
To determine which patients with a first unprovoked seizure might benefit from immediate AED treatment, Dr. Westover and his colleagues used a decision model with measures constructed from retrospective clinical trial data.
The goal of the simulation was to determine which treatment strategy—immediate or delayed AED treatment—would maximize the patient’s expected quality-adjusted life years (QALYs). Toward that end, Dr. Westover and his coinvestigators considered three base cases, which represented various degrees of seizure-recurrence risk.
The first case was a 30-year-old man with no risk factors for recurrent seizure other than having had a first seizure. In that case, immediate and deferred AED treatment resulted in 19.04 and 18.65 QALYs, respectively.
“In dollar values, using the conservative approximation of $50,000/QALY gained, this difference in treatment outcomes would amount to $19,500 gained per individual,” Dr. Westover and his coauthors wrote in their report.
The second case was a 30-year-old woman who presented with a first unprovoked seizure and had positive MRI results that establish a high risk of recurrence. As expected, because of the high recurrence risk, this scenario also favored immediate treatment, with 15.23 and 14.75 QALYs, respectively, for the immediate and deferred strategies.
The final case was a wheelchair-bound 60-year-old woman with a first unprovoked seizure and high risk of recurrence, but also a high risk of AED adverse effects and a smaller expected quality of life reduction from further seizures. In this scenario, in which treatment might be “intuitively discouraged” because of the AED side-effect risk, the cohort simulation favored deferred AED treatment by a small margin, the investigators said.
“A high baseline risk for recurrent seizures does not by itself always favor immediate AED treatment,” they said.
Findings May Shift Discussions About Therapy
The conclusion of this decision analysis by Dr. Westover and colleagues is “likely correct” that early treatment of a first unprovoked seizure could be favorable in a wide range of clinical scenarios, according to the authors of an accompanying editorial.
The decision analysis is based on a reasonable, though not comprehensive, set of parameters to simulate base cases representative of common first-ever seizure clinical scenarios, said editorialists Claire S. Jacobs, MD, PhD, and Jong Woo Lee, MD, PhD, both with the Department of Neurology at Brigham and Women’s Hospital in Boston.
Potentially the most controversial scenario addressed in the decision model, they noted, is the patient with low seizure recurrence risk but substantial quality of life decline upon recurrence. While that patient would not meet the commonly accepted 60% recurrence risk threshold that would indicate that treatment is warranted, this model favors immediate treatment because of the potentially disruptive effect of recurrence.
This study does not address important issues such as the cost of medication and patient preferences, they pointed out, and furthermore, QALYs can be difficult to integrate into clinical decision making.
Nonetheless, the findings are worth considering in clinical practice, the authors suggested. “At the very least, this study should, however subtly, shift the starting point of discussion with the patient toward a default of immediate, rather than deferred, treatment after a first unprovoked seizure and apparent absence of disease,” said Drs. Jacob and Lee.
The study was supported by NINDS.
—Andrew D. Bowser
Suggested Reading
Bao EL, Chao LY, Ni P, et al. Antiepileptic drug treatment after an unprovoked first seizure: a decision analysis. Neurology. 2018;91(15):e1429-e1439.
Jacobs CS, Lee JW. Immediate vs delayed treatment of first unprovoked seizure: to treat, or not to treat? Neurology. 2018;91(15):684-685.
Immediate treatment of a first unprovoked seizure may be preferable to delayed treatment over a wide range of patients, including those who are at low risk of recurrent seizures, results of a decision analysis suggest.
Taking into account quality of life, seizure risk, and antiepileptic drug (AED) side effects, a model favored treatment of a patient with a single unprovoked seizure who did not meet the International League Against Epilepsy (ILAE) definition of epilepsy, investigators reported.
The model also favored treatment of patients who did meet ILAE criteria, namely, a 10-year recurrence risk greater than 60% in a patient with a single unprovoked seizure, according to the analysis, which was published in the October 9 issue of Neurology.
Together, these findings suggest that the current ILAE epilepsy definition is “too simplistic” for deciding whether to start or withhold AED treatment after a first unprovoked seizure, said M. Brandon Westover, MD, PhD, Associate Professor of Neurology at Massachusetts General Hospital in Boston, and his coauthors.
“A more precise and patient-personalized definition of epilepsy should encompass not only seizure recurrence probability but also a multitude of other risks and benefits associated with AED treatment,” they said.
Weighing Risks and Benefits
To determine which patients with a first unprovoked seizure might benefit from immediate AED treatment, Dr. Westover and his colleagues used a decision model with measures constructed from retrospective clinical trial data.
The goal of the simulation was to determine which treatment strategy—immediate or delayed AED treatment—would maximize the patient’s expected quality-adjusted life years (QALYs). Toward that end, Dr. Westover and his coinvestigators considered three base cases, which represented various degrees of seizure-recurrence risk.
The first case was a 30-year-old man with no risk factors for recurrent seizure other than having had a first seizure. In that case, immediate and deferred AED treatment resulted in 19.04 and 18.65 QALYs, respectively.
“In dollar values, using the conservative approximation of $50,000/QALY gained, this difference in treatment outcomes would amount to $19,500 gained per individual,” Dr. Westover and his coauthors wrote in their report.
The second case was a 30-year-old woman who presented with a first unprovoked seizure and had positive MRI results that establish a high risk of recurrence. As expected, because of the high recurrence risk, this scenario also favored immediate treatment, with 15.23 and 14.75 QALYs, respectively, for the immediate and deferred strategies.
The final case was a wheelchair-bound 60-year-old woman with a first unprovoked seizure and high risk of recurrence, but also a high risk of AED adverse effects and a smaller expected quality of life reduction from further seizures. In this scenario, in which treatment might be “intuitively discouraged” because of the AED side-effect risk, the cohort simulation favored deferred AED treatment by a small margin, the investigators said.
“A high baseline risk for recurrent seizures does not by itself always favor immediate AED treatment,” they said.
Findings May Shift Discussions About Therapy
The conclusion of this decision analysis by Dr. Westover and colleagues is “likely correct” that early treatment of a first unprovoked seizure could be favorable in a wide range of clinical scenarios, according to the authors of an accompanying editorial.
The decision analysis is based on a reasonable, though not comprehensive, set of parameters to simulate base cases representative of common first-ever seizure clinical scenarios, said editorialists Claire S. Jacobs, MD, PhD, and Jong Woo Lee, MD, PhD, both with the Department of Neurology at Brigham and Women’s Hospital in Boston.
Potentially the most controversial scenario addressed in the decision model, they noted, is the patient with low seizure recurrence risk but substantial quality of life decline upon recurrence. While that patient would not meet the commonly accepted 60% recurrence risk threshold that would indicate that treatment is warranted, this model favors immediate treatment because of the potentially disruptive effect of recurrence.
This study does not address important issues such as the cost of medication and patient preferences, they pointed out, and furthermore, QALYs can be difficult to integrate into clinical decision making.
Nonetheless, the findings are worth considering in clinical practice, the authors suggested. “At the very least, this study should, however subtly, shift the starting point of discussion with the patient toward a default of immediate, rather than deferred, treatment after a first unprovoked seizure and apparent absence of disease,” said Drs. Jacob and Lee.
The study was supported by NINDS.
—Andrew D. Bowser
Suggested Reading
Bao EL, Chao LY, Ni P, et al. Antiepileptic drug treatment after an unprovoked first seizure: a decision analysis. Neurology. 2018;91(15):e1429-e1439.
Jacobs CS, Lee JW. Immediate vs delayed treatment of first unprovoked seizure: to treat, or not to treat? Neurology. 2018;91(15):684-685.
Psychosocial Impact of Psoriasis: A Review for Dermatology Residents
The psychosocial impact of psoriasis is a critical component of disease burden. Psoriatic patients have high rates of depression and anxiety, problems at work, and difficulties with interpersonal relationships and intimacy.1 A National Psoriasis Foundation (NPF) survey from 2003 to 2011 reported that psoriasis affects overall emotional well-being in 88% of patients and enjoyment of life in 82% of patients.2
The reasons for psychosocial burden stem from public misconceptions and disease stigma. A survey of 1005 individuals (age range, 16–64 years) about their perceptions of psoriasis revealed that 16.5% believed that psoriasis is contagious and 6.8% believed that psoriasis is related to personal hygiene.3 Fifty percent practiced discriminatory behavior toward psoriatic patients, including reluctance to shake hands (28.8%) and engage in sexual relations/intercourse (44.1%). Sixty-five percent of psoriatic patients felt their appearance is unsightly, and 73% felt self-conscious about having psoriasis.2
The psychosocial burden exists despite medical treatment of the disease. In a cross-sectional study of 1184 psoriatic patients, 70.2% had impaired quality of life (QOL) as measured by the dermatology life quality index (DLQI), even after receiving a 4-week treatment for psoriasis.4 Medical treatment of psoriasis is not enough; providers need to assess overall QOL and provide treatment and resources for these patients in addition to symptomatic management.
There have been many studies on the psychosocial burden of psoriasis, but few have focused on a dermatology resident’s role in addressing this issue. This article will review psychosocial domains—psychiatric comorbidities and social functioning including occupational functioning, interpersonal relationships, and sexual functioning— and discuss a dermatology resident’s role in assessing and addressing each of these areas.
Methods
A PubMed search of articles indexed for MEDLINE was conducted using the following terms: psoriasis, depression, anxiety, work productivity, sexual functioning, and interpersonal relationships. Selected articles covered prevalence, assessment, and management of each psychosocial domain.
Results
Psychiatric Comorbidities
Prevalence
A high prevalence of psychiatric comorbidities exists in psoriatic patients. In a study of 469,097 patients with psoriasis, depression was the third most prevalent comorbidity (17.91%), following hyperlipidemia (45.64%) and hypertension (42.19%).5 In a 10-year longitudinal, population-based, prospective cohort study, antidepressant prescriptions were twice as frequent in psoriatic patients (17.8%) compared to control (7.9%)(P<.001).6 In a meta-analysis of 98 studies investigating psoriatic patients and psychiatric comorbidities, patients with psoriasis were 1.5 times more likely to experience depression (odds ratio [OR]: 1.57; 95% CI, 1.40-1.76) and use antidepressants (OR: 4.24; 95% CI, 1.53-11.76) compared to control.7 Patients with psoriasis were more likely to attempt suicide (OR: 1.32; 95% CI, 1.14-1.54) and complete suicide (OR: 1.20; 95% CI, 1.04-1.39) compared to people without psoriasis.8 A 1-year cross-sectional study of 90 psoriatic patients reported 78.7% were diagnosed with depression and 76.7% were diagnosed with anxiety. Seventy-two percent reported both anxiety and depression, correlating with worse QOL (χ2=26.7; P<.05).9
Assessment
Psychiatric comorbidities are assessed using clinical judgment and formal screening questionnaires in research studies. Signs of depression in patients with psoriasis can manifest as poor treatment adherence and recurrent flares of psoriasis.10,11 Psoriatic patients with psychiatric comorbidities were less likely to be adherent to treatment (risk ratio: 0.35; P<.003).10 The patient health questionnaire (PHQ) 9 and generalized anxiety disorder scale (GAD) 7 are validated and reliable questionnaires. The first 2 questions in PHQ-9 and GAD-7 screen for depression and anxiety, respectively.12-14 These 2-question screens are practical in a fast-paced dermatology outpatient setting. Systematic questionnaires specifically targeting mood disorders may be more beneficial than the widely used DLQI, which may not adequately capture mood disorders. Over the course of 10 months, 607 patients with psoriasis were asked to fill out the PHQ-9, GAD-7, and DLQI. Thirty-eight percent of patients with major depressive disorder had a DLQI score lower than 10, while 46% of patients with generalized anxiety disorder had a DLQI score lower than 10.15 Other questionnaires, including the hospital anxiety and depression scale and Beck depression inventory, are valid instruments with high sensitivity but are commonly used for research purposes and may not be clinically feasible.16
Management
Dermatologists should refer patients with depression and/or anxiety to psychiatry. Interventions include pharmacologic and nonpharmacologic management. First-line therapy for depression and anxiety is a combination of selective serotonin reuptake inhibitors and cognitive behavioral therapy.17 In addition, providers can direct patients to online resources such as the NPF website, where patients with psoriasis can access information about the signs and symptoms of mood disorders and contact the patient navigation center for further help.18
Social Functioning
Occupational Prevalence
The NPF found that 92% of patients with psoriasis or psoriatic arthritis (PsA) surveyed between 2003 and 2011 cited their psoriasis as reason for unemployment.2 In a survey of 43 patients asked about social and occupational functioning using the social and occupational assessment scale, 62.5% of psoriatic patients reported distress at work and 51.1% reported decreased efficiency at work.19 A national online survey that was conducted in France and issued to patients with and without psoriasis assessed overall QOL and work productivity using the work productivity and activity impairment questionnaire for psoriasis (WPAI-PSO). Of 714 patients with psoriasis and PsA, the latter had a 57.6% decrease in work productivity over 7 days compared to 27.9% in controls (P<.05).20 Occupational impairment leads to lost wages and hinders advancement, further exacerbating the psychosocial burden of psoriasis.21
Occupational Assessment
Formal assessment of occupational function can be done with the WPAI-PSO, a 6-question valid instrument.22 Providers may look for risk factors associated with greater loss in work productivity to help identify and offer support for patients. Patients with increased severity of itching, pain, and scaling experienced a greater decrease in work productivity.21,23 Patients with PsA warrant early detection and treatment because they experience greater physical restraints that can interfere with work activities. Of the 459 psoriatic patients without a prior diagnosis of PsA who filled out the PsA screening and evaluation questionnaire, 144 (31.4%) received a score of 44 or higher and were referred to rheumatology for further evaluation with the classification criteria for PsA. Nine percent of patients failed to be screened and remained undiagnosed with PsA.24 In a study using the health assessment questionnaire to assess 400 patients with PsA, those with worse physical function due to joint pain and stiffness were less likely to remain employed (OR: 0.56; P=.02).25
Occupational Management
Identifying and coordinating symptoms of PsA between dermatology and rheumatology is beneficial for patients who experience debilitating symptoms. There are a variety of treatments available for PsA. According to the European League Against Rheumatism 2015 guidelines developed from expert opinion and systematic reviews for PsA management, there are 4 phases of treatment, with reassessment every 3 to 6 months for effectiveness of therapy.26,27 Phase I involves initiating nonsteroidal anti-inflammatory drugs with or without glucocorticoid injections. Phase II involves synthetic disease-modifying drugs, including methotrexate, leflunomide, sulfasalazine, or cyclosporine. Phase III involves adding a second synthetic disease-modifying drug or starting a biologic, such as an anti–tumor necrosis factor, IL-12/IL-23, or IL-17 inhibitor. Phase IV involves switching to a different drug in either aforementioned class.26,27 Treatment with biologics improves work productivity as assessed by WPAI-PSO for psoriasis and PsA.28-30 Encouraging patients to speak up in the workplace and request small accommodations such as timely breaks or ergonomic chairs can help patients feel more comfortable and supported in the work environment.18 Patients who felt supported at work were more likely to remain employed.25
Interpersonal Relationships Prevalence
Misinformation about psoriasis, fear of rejection, and feelings of isolation may contribute to interpersonal conflict. Patients have feelings of shame and self-consciousness that hinder them from engaging in social activities and seeking out relationships.31 Twenty-nine percent of patients feel that psoriasis has interfered with establishing relationships because of negative self-esteem associated with the disease,32 and 26.3% have experienced people avoiding physical contact.33 Family and spouses of patients with psoriasis may be secondarily affected due to economic and emotional distress. Ninety-eight percent of family members of psoriatic patients experienced emotional distress and 54% experienced the burden of care.34 In a survey of 63 relatives and partners of patients with psoriasis, 57% experienced psychological distress, including anxiety and worry over a psoriatic patient’s future.35
Interpersonal Relationships Assessment
Current available tools, including the DLQI and short form health survey, measure overall QOL, including social functioning, but may not be practical in a clinic setting. Although no quick-screening test to assess for this domain exists, providers are encouraged to ask patients about disease impact on interpersonal relationships. The family DLQI questionnaire, adapted from the DLQI, may help physicians and social workers evaluate the burden on a patient’s family members.34
Interpersonal Relationships Management
It may be difficult for providers to address problems with interpersonal relationships without accessible tools. Patients may not be accompanied by family or friends during appointments, and it is difficult to screen for these issues during visits. Providers may offer resources such as the NPF website, which provides information about support groups. It also provides tips on dating and connecting to others in the community who share similar experiences.18 Encouraging patients to seek family or couples therapy also may be beneficial. Increased social support can lead to better QOL and fewer depressive symptoms.36
Sexual Functioning Prevalence
Psoriasis affects both physical and psychological components of sexual function. Among 3485 patients with skin conditions who were surveyed about sexual function, 34% of psoriatic patients reported that psoriasis interfered with sexual functioning at least to a certain degree.37 Sexual impairment was strongly associated with depression, anxiety, and suicidal ideation; 24% of depressed patients and 20% of anxious patients experienced sexual problems a lot or very much, based on the DLQI.37 Depending on the questionnaire used, the prevalence of sexual dysfunction due to psoriasis ranged from 35.5% to 71.3%.38 In an observational cohort study of 158 participants (n=79 psoriasis patients and n=79 controls), 34.2% of patients with psoriasis experienced erectile dysfunction compared to 17.7% of controls.39 Forty-two percent of psoriatic patients with genital involvement reported dyspareunia, 32% reported worsening of genital psoriasis after intercourse, and 43% reported decreased frequency of intercourse.40
Sexual Functioning Assessment
The Skindex-29, DLQI, and psoriasis disability index are available QOL tools that include one question evaluating difficulties with sexual function. The
Sexual Functioning Management
Better disease control leads to improved sexual function, as patients experience fewer feelings of shame, anxiety, and depression, as well as improvement of physical symptoms that can interfere with sexual functioning.38,43,44 Reducing friction, warmth, and moisture, as well as avoiding tight clothing, can help those with genital psoriasis. Patients are advised to reapply topical medications after sexual intercourse. Patients also can apply makeup to disguise psoriasis and help reduce feelings of self-consciousness that can impede sexual intimacy.18
Comment
The psychosocial burden of psoriasis penetrates many facets of patient lives. Psoriasis can invoke feelings of shame and embarrassment that are worsened by the public’s misconceptions about psoriasis, resulting in serious mental health issues that can cause even greater disability. Depression and anxiety are prevalent in patients with psoriasis. The characteristic symptoms of pain and pruritus along with psychiatric comorbidities can have an underestimated impact on daily activities, including employment, interpersonal relationships, and sexual function. Such dysfunctions have serious implications toward wages, professional advancement, social support, and overall QOL.
Dermatology providers play an important role in screening for these problems through validated questionnaires and identifying risks. Simple screening questions such as the PHQ-9 can be beneficial and feasible during dermatology visits. Screening for PsA can help patients avoid problems at work. Sexual dysfunction is a sensitive topic; however, providers can use a 1-question screen from valid questionnaires and inquire about the location of lesions as opportunities to address this issue.
Interventions lead to better disease control, which concurrently improves overall QOL. These interventions depend on both patient adherence and a physician’s commitment to finding an optimal treatment regimen for each individual. Medical management; coordinating care; developing treatment plans with psychiatry, rheumatology, and primary care providers; and psychological counseling and services may be necessary and beneficial (Table). Offering accessible resources such as the NPF website helps patients access information outside the clinic when it is not feasible to address all these concerns in a single visit. Psoriasis requires more than just medical management; it requires dermatology providers to use a multidisciplinary approach to address the psychosocial aspects of the disease.
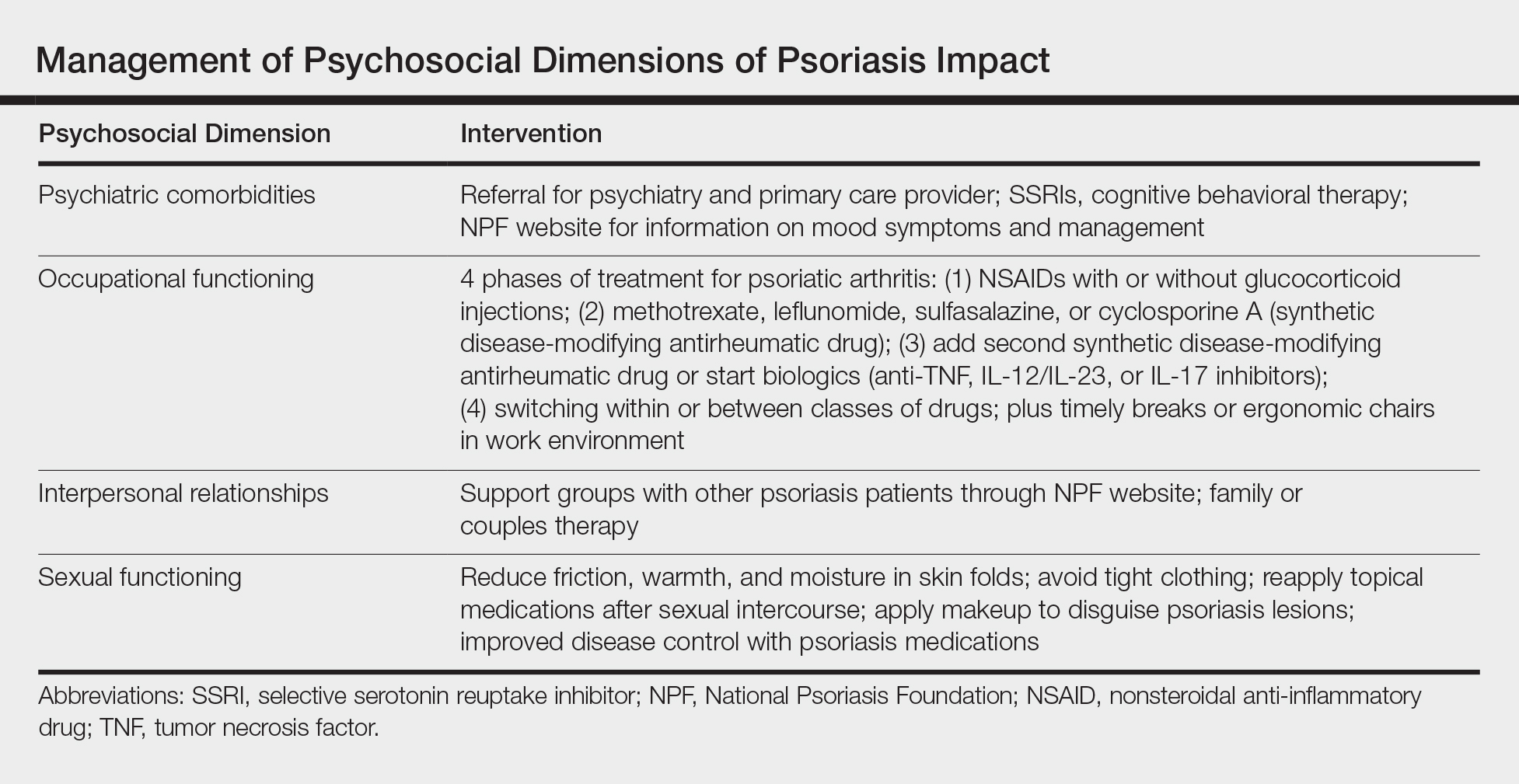
Conclusion
The psychosocial burden of psoriasis is immense. Stigma, public misconception, mental health concerns, and occupational and interpersonal difficulty are the basis of disease burden. Providers play a vital role in assessing the effect psoriasis has on different areas of patients’ lives and providing appropriate interventions and resources to reduce disease burden.
- Kimball AB, Jacobson C, Weiss S, et al. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383-392.
- Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PloS One. 2012;7:e52935.
- Halioua B, Sid-Mohand D, Roussel ME, et al. Extent of misconceptions, negative prejudices and discriminatory behaviour to psoriasis patients in France. J Eur Acad Dermatol Venereol. 2016;30:650-654.
- Wolf P, Weger W, Legat F, et al. Quality of life and treatment goals in psoriasis from the patient perspective: results of an Austrian cross-sectional survey. J Dtsch Dermatol Ges. 2018;16:981-990.
- Shah K, Mellars L, Changolkar A, et al. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77:287-292.e4.
- Dowlatshahi EA, Wakkee M, Herings RM, et al. Increased antidepressant drug exposure in psoriasis patients: a longitudinal population-based cohort study. Acta Derm Venereol. 2013;93:544-550.
- Dowlatshahi EA, Wakkee M, Arends LR, et al. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542-1551.
- Singh S, Taylor C, Kornmehl H, et al. Psoriasis and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77:425.e2-440.e2.
- Lakshmy S, Balasundaram S, Sarkar S, et al. A cross-sectional study of prevalence and implications of depression and anxiety in psoriasis. Indian J Psychol Med. 2015;37:434-440.
- Renzi C, Picardi A, Abeni D, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337-342.
- Kulkarni AS, Balkrishnan R, Camacho FT, et al. Medication and health care service utilization related to depressive symptoms in older adults with psoriasis. J Drugs Dermatol. 2004;3:661-666.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Lamb RC, Matcham F, Turner MA, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol. 2017;176:1028-1034.
- Law M, Naughton MT, Dhar A, et al. Validation of two depression screening instruments in a sleep disorders clinic. J Clin Sleep Med. 2014;10:683-688.
- Cuijpers P, Dekker J, Hollon SD, et al. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: a meta-analysis. J Clin Psychiatry. 2009;70:1219-1229.
- National Psoriasis Foundation. Living with psoriatic arthritis. https://www.psoriasis.org/life-with-psoriatic-arthritis. Accessed September 23, 2018.
- Gaikwad R, Deshpande S, Raje S, et al. Evaluation of functional impairment in psoriasis. Indian J Dermatol Venereol Leprol. 2006;72:37-40.
- Claudepierre P, Lahfa M, Levy P, et al. The impact of psoriasis on professional life: PsoPRO, a French national survey [published online April 6, 2018]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.14986.
- Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41:514-521.
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. PharmacoEconomics. 1993;4:353-365.
- Korman NJ, Zhao Y, Pike J, et al. Increased severity of itching, pain, and scaling in psoriasis patients is associated with increased disease severity, reduced quality of life, and reduced work productivity. Dermatol Online J. 2015;21.
- Spelman L, Su JC, Fernandez-Penas P, et al. Frequency of undiagnosed psoriatic arthritis among psoriasis patients in Australian dermatology practice. J Eur Acad Dermatol Venereol. 2015;29:2184-2191.
- Tillett W, Shaddick G, Askari A, et al. Factors influencing work disability in psoriatic arthritis: first results from a large UK multicentre study. Rheumatology (Oxford). 2015;54:157-162.
- Raychaudhuri SP, Wilken R, Sukhov AC, et al. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J Autoimmun. 2017;76:21-37.
- Gossec L, Smolen JS, Ramiro S, et al. European League Against Rheumatism (EULAR) recommendations for the manegement of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499-510.
- Beroukhim K, Danesh M, Nguyen C, et al. A prospective, interventional assessment of the impact of ustekinumab treatment on psoriasis-related work productivity and activity impairment. J Dermatol Treat. 2016;27:552-555.
- Armstrong AW, Lynde CW, McBride SR, et al. Effect of ixekizumab treatment on work productivity for patients with moderate-to-severe plaque psoriasis: analysis of results from 3 randomized phase 3 clinical trials. JAMA Dermatol. 2016;152:661-669.
- Kimball AB, Yu AP, Signorovitch J, et al. The effects of adalimumab treatment and psoriasis severity on self-reported work productivity and activity impairment for patients with moderate to severe psoriasis. J Am Acad Dermatol. 2012;66:e67-76.
- Feldman SR, Malakouti M, Koo JY. Social impact of the burden of psoriasis: effects on patients and practice. Dermatol Online J. 2014;20.
- Reich A, Welz-Kubiak K, Rams Ł. Apprehension of the disease by patients suffering from psoriasis. Postepy Dermatol Alergol. 2014;31:289-293.
- Gupta MA, Gupta AK, Watteel GN. Perceived deprivation of social touch in psoriasis is associated with greater psychologic morbidity: an index of the stigma experience in dermatologic disorders. Cutis. 1998;61:339-342.
- Basra MK, Finlay AY. The family impact of skin diseases: the Greater Patient concept. Br J Dermatol. 2007;156:929-937.
- Eghlileb AM, Davies EE, Finlay AY. Psoriasis has a major secondary impact on the lives of family members and partners. Br J Dermatol. 2007;156:1245-1250.
- Janowski K, Steuden S, Pietrzak A, et al. Social support and adaptation to the disease in men and women with psoriasis. Arch Dermatol Res. 2012;304:421-432.
- Sampogna F, Abeni D, Gieler U, et al. Impairment of sexual life in 3,485 dermatological outpatients from a multicentre study in 13 European countries. Acta Derm Venereol. 2017;97:478-482.
- Sampogna F, Gisondi P, Tabolli S, et al. Impairment of sexual life in patients with psoriasis. Dermatology. 2007;214:144-150.
- Molina-Leyva A, Molina-Leyva I, Almodovar-Real A, et al. Prevalence and associated factors of erectile dysfunction in patients with moderate to severe psoriasis and healthy population: a comparative study considering physical and psychological factors. Arch Sex Behav. 2016;45:2047-2055.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Labbate LA, Lare SB. Sexual dysfunction in male psychiatric outpatients: validity of the Massachusetts General Hospital Sexual Functioning Questionnaire. Psychother Psychosom. 2001;70:221-225.
- Molina-Leyva A, Almodovar-Real A, Ruiz-Carrascosa JC, et al. Distribution pattern of psoriasis affects sexual function in moderate to severe psoriasis: a prospective case series study. J Sex Med. 2014;11:2882-2889.
- Guenther L, Han C, Szapary P, et al. Impact of ustekinumab on health-related quality of life and sexual difficulties associated with psoriasis: results from two phase III clinical trials. J Eur Acad Dermatol Venereol. 2011;25:851-857.
- Guenther L, Warren RB, Cather JC, et al. Impact of ixekizumab treatment on skin-related personal relationship difficulties in moderate-to-severe psoriasis patients: 12-week results from two Phase 3 trials. J Eur Acad Dermatol Venereol. 2017;31:1867-1875.
The psychosocial impact of psoriasis is a critical component of disease burden. Psoriatic patients have high rates of depression and anxiety, problems at work, and difficulties with interpersonal relationships and intimacy.1 A National Psoriasis Foundation (NPF) survey from 2003 to 2011 reported that psoriasis affects overall emotional well-being in 88% of patients and enjoyment of life in 82% of patients.2
The reasons for psychosocial burden stem from public misconceptions and disease stigma. A survey of 1005 individuals (age range, 16–64 years) about their perceptions of psoriasis revealed that 16.5% believed that psoriasis is contagious and 6.8% believed that psoriasis is related to personal hygiene.3 Fifty percent practiced discriminatory behavior toward psoriatic patients, including reluctance to shake hands (28.8%) and engage in sexual relations/intercourse (44.1%). Sixty-five percent of psoriatic patients felt their appearance is unsightly, and 73% felt self-conscious about having psoriasis.2
The psychosocial burden exists despite medical treatment of the disease. In a cross-sectional study of 1184 psoriatic patients, 70.2% had impaired quality of life (QOL) as measured by the dermatology life quality index (DLQI), even after receiving a 4-week treatment for psoriasis.4 Medical treatment of psoriasis is not enough; providers need to assess overall QOL and provide treatment and resources for these patients in addition to symptomatic management.
There have been many studies on the psychosocial burden of psoriasis, but few have focused on a dermatology resident’s role in addressing this issue. This article will review psychosocial domains—psychiatric comorbidities and social functioning including occupational functioning, interpersonal relationships, and sexual functioning— and discuss a dermatology resident’s role in assessing and addressing each of these areas.
Methods
A PubMed search of articles indexed for MEDLINE was conducted using the following terms: psoriasis, depression, anxiety, work productivity, sexual functioning, and interpersonal relationships. Selected articles covered prevalence, assessment, and management of each psychosocial domain.
Results
Psychiatric Comorbidities
Prevalence
A high prevalence of psychiatric comorbidities exists in psoriatic patients. In a study of 469,097 patients with psoriasis, depression was the third most prevalent comorbidity (17.91%), following hyperlipidemia (45.64%) and hypertension (42.19%).5 In a 10-year longitudinal, population-based, prospective cohort study, antidepressant prescriptions were twice as frequent in psoriatic patients (17.8%) compared to control (7.9%)(P<.001).6 In a meta-analysis of 98 studies investigating psoriatic patients and psychiatric comorbidities, patients with psoriasis were 1.5 times more likely to experience depression (odds ratio [OR]: 1.57; 95% CI, 1.40-1.76) and use antidepressants (OR: 4.24; 95% CI, 1.53-11.76) compared to control.7 Patients with psoriasis were more likely to attempt suicide (OR: 1.32; 95% CI, 1.14-1.54) and complete suicide (OR: 1.20; 95% CI, 1.04-1.39) compared to people without psoriasis.8 A 1-year cross-sectional study of 90 psoriatic patients reported 78.7% were diagnosed with depression and 76.7% were diagnosed with anxiety. Seventy-two percent reported both anxiety and depression, correlating with worse QOL (χ2=26.7; P<.05).9
Assessment
Psychiatric comorbidities are assessed using clinical judgment and formal screening questionnaires in research studies. Signs of depression in patients with psoriasis can manifest as poor treatment adherence and recurrent flares of psoriasis.10,11 Psoriatic patients with psychiatric comorbidities were less likely to be adherent to treatment (risk ratio: 0.35; P<.003).10 The patient health questionnaire (PHQ) 9 and generalized anxiety disorder scale (GAD) 7 are validated and reliable questionnaires. The first 2 questions in PHQ-9 and GAD-7 screen for depression and anxiety, respectively.12-14 These 2-question screens are practical in a fast-paced dermatology outpatient setting. Systematic questionnaires specifically targeting mood disorders may be more beneficial than the widely used DLQI, which may not adequately capture mood disorders. Over the course of 10 months, 607 patients with psoriasis were asked to fill out the PHQ-9, GAD-7, and DLQI. Thirty-eight percent of patients with major depressive disorder had a DLQI score lower than 10, while 46% of patients with generalized anxiety disorder had a DLQI score lower than 10.15 Other questionnaires, including the hospital anxiety and depression scale and Beck depression inventory, are valid instruments with high sensitivity but are commonly used for research purposes and may not be clinically feasible.16
Management
Dermatologists should refer patients with depression and/or anxiety to psychiatry. Interventions include pharmacologic and nonpharmacologic management. First-line therapy for depression and anxiety is a combination of selective serotonin reuptake inhibitors and cognitive behavioral therapy.17 In addition, providers can direct patients to online resources such as the NPF website, where patients with psoriasis can access information about the signs and symptoms of mood disorders and contact the patient navigation center for further help.18
Social Functioning
Occupational Prevalence
The NPF found that 92% of patients with psoriasis or psoriatic arthritis (PsA) surveyed between 2003 and 2011 cited their psoriasis as reason for unemployment.2 In a survey of 43 patients asked about social and occupational functioning using the social and occupational assessment scale, 62.5% of psoriatic patients reported distress at work and 51.1% reported decreased efficiency at work.19 A national online survey that was conducted in France and issued to patients with and without psoriasis assessed overall QOL and work productivity using the work productivity and activity impairment questionnaire for psoriasis (WPAI-PSO). Of 714 patients with psoriasis and PsA, the latter had a 57.6% decrease in work productivity over 7 days compared to 27.9% in controls (P<.05).20 Occupational impairment leads to lost wages and hinders advancement, further exacerbating the psychosocial burden of psoriasis.21
Occupational Assessment
Formal assessment of occupational function can be done with the WPAI-PSO, a 6-question valid instrument.22 Providers may look for risk factors associated with greater loss in work productivity to help identify and offer support for patients. Patients with increased severity of itching, pain, and scaling experienced a greater decrease in work productivity.21,23 Patients with PsA warrant early detection and treatment because they experience greater physical restraints that can interfere with work activities. Of the 459 psoriatic patients without a prior diagnosis of PsA who filled out the PsA screening and evaluation questionnaire, 144 (31.4%) received a score of 44 or higher and were referred to rheumatology for further evaluation with the classification criteria for PsA. Nine percent of patients failed to be screened and remained undiagnosed with PsA.24 In a study using the health assessment questionnaire to assess 400 patients with PsA, those with worse physical function due to joint pain and stiffness were less likely to remain employed (OR: 0.56; P=.02).25
Occupational Management
Identifying and coordinating symptoms of PsA between dermatology and rheumatology is beneficial for patients who experience debilitating symptoms. There are a variety of treatments available for PsA. According to the European League Against Rheumatism 2015 guidelines developed from expert opinion and systematic reviews for PsA management, there are 4 phases of treatment, with reassessment every 3 to 6 months for effectiveness of therapy.26,27 Phase I involves initiating nonsteroidal anti-inflammatory drugs with or without glucocorticoid injections. Phase II involves synthetic disease-modifying drugs, including methotrexate, leflunomide, sulfasalazine, or cyclosporine. Phase III involves adding a second synthetic disease-modifying drug or starting a biologic, such as an anti–tumor necrosis factor, IL-12/IL-23, or IL-17 inhibitor. Phase IV involves switching to a different drug in either aforementioned class.26,27 Treatment with biologics improves work productivity as assessed by WPAI-PSO for psoriasis and PsA.28-30 Encouraging patients to speak up in the workplace and request small accommodations such as timely breaks or ergonomic chairs can help patients feel more comfortable and supported in the work environment.18 Patients who felt supported at work were more likely to remain employed.25
Interpersonal Relationships Prevalence
Misinformation about psoriasis, fear of rejection, and feelings of isolation may contribute to interpersonal conflict. Patients have feelings of shame and self-consciousness that hinder them from engaging in social activities and seeking out relationships.31 Twenty-nine percent of patients feel that psoriasis has interfered with establishing relationships because of negative self-esteem associated with the disease,32 and 26.3% have experienced people avoiding physical contact.33 Family and spouses of patients with psoriasis may be secondarily affected due to economic and emotional distress. Ninety-eight percent of family members of psoriatic patients experienced emotional distress and 54% experienced the burden of care.34 In a survey of 63 relatives and partners of patients with psoriasis, 57% experienced psychological distress, including anxiety and worry over a psoriatic patient’s future.35
Interpersonal Relationships Assessment
Current available tools, including the DLQI and short form health survey, measure overall QOL, including social functioning, but may not be practical in a clinic setting. Although no quick-screening test to assess for this domain exists, providers are encouraged to ask patients about disease impact on interpersonal relationships. The family DLQI questionnaire, adapted from the DLQI, may help physicians and social workers evaluate the burden on a patient’s family members.34
Interpersonal Relationships Management
It may be difficult for providers to address problems with interpersonal relationships without accessible tools. Patients may not be accompanied by family or friends during appointments, and it is difficult to screen for these issues during visits. Providers may offer resources such as the NPF website, which provides information about support groups. It also provides tips on dating and connecting to others in the community who share similar experiences.18 Encouraging patients to seek family or couples therapy also may be beneficial. Increased social support can lead to better QOL and fewer depressive symptoms.36
Sexual Functioning Prevalence
Psoriasis affects both physical and psychological components of sexual function. Among 3485 patients with skin conditions who were surveyed about sexual function, 34% of psoriatic patients reported that psoriasis interfered with sexual functioning at least to a certain degree.37 Sexual impairment was strongly associated with depression, anxiety, and suicidal ideation; 24% of depressed patients and 20% of anxious patients experienced sexual problems a lot or very much, based on the DLQI.37 Depending on the questionnaire used, the prevalence of sexual dysfunction due to psoriasis ranged from 35.5% to 71.3%.38 In an observational cohort study of 158 participants (n=79 psoriasis patients and n=79 controls), 34.2% of patients with psoriasis experienced erectile dysfunction compared to 17.7% of controls.39 Forty-two percent of psoriatic patients with genital involvement reported dyspareunia, 32% reported worsening of genital psoriasis after intercourse, and 43% reported decreased frequency of intercourse.40
Sexual Functioning Assessment
The Skindex-29, DLQI, and psoriasis disability index are available QOL tools that include one question evaluating difficulties with sexual function. The
Sexual Functioning Management
Better disease control leads to improved sexual function, as patients experience fewer feelings of shame, anxiety, and depression, as well as improvement of physical symptoms that can interfere with sexual functioning.38,43,44 Reducing friction, warmth, and moisture, as well as avoiding tight clothing, can help those with genital psoriasis. Patients are advised to reapply topical medications after sexual intercourse. Patients also can apply makeup to disguise psoriasis and help reduce feelings of self-consciousness that can impede sexual intimacy.18
Comment
The psychosocial burden of psoriasis penetrates many facets of patient lives. Psoriasis can invoke feelings of shame and embarrassment that are worsened by the public’s misconceptions about psoriasis, resulting in serious mental health issues that can cause even greater disability. Depression and anxiety are prevalent in patients with psoriasis. The characteristic symptoms of pain and pruritus along with psychiatric comorbidities can have an underestimated impact on daily activities, including employment, interpersonal relationships, and sexual function. Such dysfunctions have serious implications toward wages, professional advancement, social support, and overall QOL.
Dermatology providers play an important role in screening for these problems through validated questionnaires and identifying risks. Simple screening questions such as the PHQ-9 can be beneficial and feasible during dermatology visits. Screening for PsA can help patients avoid problems at work. Sexual dysfunction is a sensitive topic; however, providers can use a 1-question screen from valid questionnaires and inquire about the location of lesions as opportunities to address this issue.
Interventions lead to better disease control, which concurrently improves overall QOL. These interventions depend on both patient adherence and a physician’s commitment to finding an optimal treatment regimen for each individual. Medical management; coordinating care; developing treatment plans with psychiatry, rheumatology, and primary care providers; and psychological counseling and services may be necessary and beneficial (Table). Offering accessible resources such as the NPF website helps patients access information outside the clinic when it is not feasible to address all these concerns in a single visit. Psoriasis requires more than just medical management; it requires dermatology providers to use a multidisciplinary approach to address the psychosocial aspects of the disease.

Conclusion
The psychosocial burden of psoriasis is immense. Stigma, public misconception, mental health concerns, and occupational and interpersonal difficulty are the basis of disease burden. Providers play a vital role in assessing the effect psoriasis has on different areas of patients’ lives and providing appropriate interventions and resources to reduce disease burden.
The psychosocial impact of psoriasis is a critical component of disease burden. Psoriatic patients have high rates of depression and anxiety, problems at work, and difficulties with interpersonal relationships and intimacy.1 A National Psoriasis Foundation (NPF) survey from 2003 to 2011 reported that psoriasis affects overall emotional well-being in 88% of patients and enjoyment of life in 82% of patients.2
The reasons for psychosocial burden stem from public misconceptions and disease stigma. A survey of 1005 individuals (age range, 16–64 years) about their perceptions of psoriasis revealed that 16.5% believed that psoriasis is contagious and 6.8% believed that psoriasis is related to personal hygiene.3 Fifty percent practiced discriminatory behavior toward psoriatic patients, including reluctance to shake hands (28.8%) and engage in sexual relations/intercourse (44.1%). Sixty-five percent of psoriatic patients felt their appearance is unsightly, and 73% felt self-conscious about having psoriasis.2
The psychosocial burden exists despite medical treatment of the disease. In a cross-sectional study of 1184 psoriatic patients, 70.2% had impaired quality of life (QOL) as measured by the dermatology life quality index (DLQI), even after receiving a 4-week treatment for psoriasis.4 Medical treatment of psoriasis is not enough; providers need to assess overall QOL and provide treatment and resources for these patients in addition to symptomatic management.
There have been many studies on the psychosocial burden of psoriasis, but few have focused on a dermatology resident’s role in addressing this issue. This article will review psychosocial domains—psychiatric comorbidities and social functioning including occupational functioning, interpersonal relationships, and sexual functioning— and discuss a dermatology resident’s role in assessing and addressing each of these areas.
Methods
A PubMed search of articles indexed for MEDLINE was conducted using the following terms: psoriasis, depression, anxiety, work productivity, sexual functioning, and interpersonal relationships. Selected articles covered prevalence, assessment, and management of each psychosocial domain.
Results
Psychiatric Comorbidities
Prevalence
A high prevalence of psychiatric comorbidities exists in psoriatic patients. In a study of 469,097 patients with psoriasis, depression was the third most prevalent comorbidity (17.91%), following hyperlipidemia (45.64%) and hypertension (42.19%).5 In a 10-year longitudinal, population-based, prospective cohort study, antidepressant prescriptions were twice as frequent in psoriatic patients (17.8%) compared to control (7.9%)(P<.001).6 In a meta-analysis of 98 studies investigating psoriatic patients and psychiatric comorbidities, patients with psoriasis were 1.5 times more likely to experience depression (odds ratio [OR]: 1.57; 95% CI, 1.40-1.76) and use antidepressants (OR: 4.24; 95% CI, 1.53-11.76) compared to control.7 Patients with psoriasis were more likely to attempt suicide (OR: 1.32; 95% CI, 1.14-1.54) and complete suicide (OR: 1.20; 95% CI, 1.04-1.39) compared to people without psoriasis.8 A 1-year cross-sectional study of 90 psoriatic patients reported 78.7% were diagnosed with depression and 76.7% were diagnosed with anxiety. Seventy-two percent reported both anxiety and depression, correlating with worse QOL (χ2=26.7; P<.05).9
Assessment
Psychiatric comorbidities are assessed using clinical judgment and formal screening questionnaires in research studies. Signs of depression in patients with psoriasis can manifest as poor treatment adherence and recurrent flares of psoriasis.10,11 Psoriatic patients with psychiatric comorbidities were less likely to be adherent to treatment (risk ratio: 0.35; P<.003).10 The patient health questionnaire (PHQ) 9 and generalized anxiety disorder scale (GAD) 7 are validated and reliable questionnaires. The first 2 questions in PHQ-9 and GAD-7 screen for depression and anxiety, respectively.12-14 These 2-question screens are practical in a fast-paced dermatology outpatient setting. Systematic questionnaires specifically targeting mood disorders may be more beneficial than the widely used DLQI, which may not adequately capture mood disorders. Over the course of 10 months, 607 patients with psoriasis were asked to fill out the PHQ-9, GAD-7, and DLQI. Thirty-eight percent of patients with major depressive disorder had a DLQI score lower than 10, while 46% of patients with generalized anxiety disorder had a DLQI score lower than 10.15 Other questionnaires, including the hospital anxiety and depression scale and Beck depression inventory, are valid instruments with high sensitivity but are commonly used for research purposes and may not be clinically feasible.16
Management
Dermatologists should refer patients with depression and/or anxiety to psychiatry. Interventions include pharmacologic and nonpharmacologic management. First-line therapy for depression and anxiety is a combination of selective serotonin reuptake inhibitors and cognitive behavioral therapy.17 In addition, providers can direct patients to online resources such as the NPF website, where patients with psoriasis can access information about the signs and symptoms of mood disorders and contact the patient navigation center for further help.18
Social Functioning
Occupational Prevalence
The NPF found that 92% of patients with psoriasis or psoriatic arthritis (PsA) surveyed between 2003 and 2011 cited their psoriasis as reason for unemployment.2 In a survey of 43 patients asked about social and occupational functioning using the social and occupational assessment scale, 62.5% of psoriatic patients reported distress at work and 51.1% reported decreased efficiency at work.19 A national online survey that was conducted in France and issued to patients with and without psoriasis assessed overall QOL and work productivity using the work productivity and activity impairment questionnaire for psoriasis (WPAI-PSO). Of 714 patients with psoriasis and PsA, the latter had a 57.6% decrease in work productivity over 7 days compared to 27.9% in controls (P<.05).20 Occupational impairment leads to lost wages and hinders advancement, further exacerbating the psychosocial burden of psoriasis.21
Occupational Assessment
Formal assessment of occupational function can be done with the WPAI-PSO, a 6-question valid instrument.22 Providers may look for risk factors associated with greater loss in work productivity to help identify and offer support for patients. Patients with increased severity of itching, pain, and scaling experienced a greater decrease in work productivity.21,23 Patients with PsA warrant early detection and treatment because they experience greater physical restraints that can interfere with work activities. Of the 459 psoriatic patients without a prior diagnosis of PsA who filled out the PsA screening and evaluation questionnaire, 144 (31.4%) received a score of 44 or higher and were referred to rheumatology for further evaluation with the classification criteria for PsA. Nine percent of patients failed to be screened and remained undiagnosed with PsA.24 In a study using the health assessment questionnaire to assess 400 patients with PsA, those with worse physical function due to joint pain and stiffness were less likely to remain employed (OR: 0.56; P=.02).25
Occupational Management
Identifying and coordinating symptoms of PsA between dermatology and rheumatology is beneficial for patients who experience debilitating symptoms. There are a variety of treatments available for PsA. According to the European League Against Rheumatism 2015 guidelines developed from expert opinion and systematic reviews for PsA management, there are 4 phases of treatment, with reassessment every 3 to 6 months for effectiveness of therapy.26,27 Phase I involves initiating nonsteroidal anti-inflammatory drugs with or without glucocorticoid injections. Phase II involves synthetic disease-modifying drugs, including methotrexate, leflunomide, sulfasalazine, or cyclosporine. Phase III involves adding a second synthetic disease-modifying drug or starting a biologic, such as an anti–tumor necrosis factor, IL-12/IL-23, or IL-17 inhibitor. Phase IV involves switching to a different drug in either aforementioned class.26,27 Treatment with biologics improves work productivity as assessed by WPAI-PSO for psoriasis and PsA.28-30 Encouraging patients to speak up in the workplace and request small accommodations such as timely breaks or ergonomic chairs can help patients feel more comfortable and supported in the work environment.18 Patients who felt supported at work were more likely to remain employed.25
Interpersonal Relationships Prevalence
Misinformation about psoriasis, fear of rejection, and feelings of isolation may contribute to interpersonal conflict. Patients have feelings of shame and self-consciousness that hinder them from engaging in social activities and seeking out relationships.31 Twenty-nine percent of patients feel that psoriasis has interfered with establishing relationships because of negative self-esteem associated with the disease,32 and 26.3% have experienced people avoiding physical contact.33 Family and spouses of patients with psoriasis may be secondarily affected due to economic and emotional distress. Ninety-eight percent of family members of psoriatic patients experienced emotional distress and 54% experienced the burden of care.34 In a survey of 63 relatives and partners of patients with psoriasis, 57% experienced psychological distress, including anxiety and worry over a psoriatic patient’s future.35
Interpersonal Relationships Assessment
Current available tools, including the DLQI and short form health survey, measure overall QOL, including social functioning, but may not be practical in a clinic setting. Although no quick-screening test to assess for this domain exists, providers are encouraged to ask patients about disease impact on interpersonal relationships. The family DLQI questionnaire, adapted from the DLQI, may help physicians and social workers evaluate the burden on a patient’s family members.34
Interpersonal Relationships Management
It may be difficult for providers to address problems with interpersonal relationships without accessible tools. Patients may not be accompanied by family or friends during appointments, and it is difficult to screen for these issues during visits. Providers may offer resources such as the NPF website, which provides information about support groups. It also provides tips on dating and connecting to others in the community who share similar experiences.18 Encouraging patients to seek family or couples therapy also may be beneficial. Increased social support can lead to better QOL and fewer depressive symptoms.36
Sexual Functioning Prevalence
Psoriasis affects both physical and psychological components of sexual function. Among 3485 patients with skin conditions who were surveyed about sexual function, 34% of psoriatic patients reported that psoriasis interfered with sexual functioning at least to a certain degree.37 Sexual impairment was strongly associated with depression, anxiety, and suicidal ideation; 24% of depressed patients and 20% of anxious patients experienced sexual problems a lot or very much, based on the DLQI.37 Depending on the questionnaire used, the prevalence of sexual dysfunction due to psoriasis ranged from 35.5% to 71.3%.38 In an observational cohort study of 158 participants (n=79 psoriasis patients and n=79 controls), 34.2% of patients with psoriasis experienced erectile dysfunction compared to 17.7% of controls.39 Forty-two percent of psoriatic patients with genital involvement reported dyspareunia, 32% reported worsening of genital psoriasis after intercourse, and 43% reported decreased frequency of intercourse.40
Sexual Functioning Assessment
The Skindex-29, DLQI, and psoriasis disability index are available QOL tools that include one question evaluating difficulties with sexual function. The
Sexual Functioning Management
Better disease control leads to improved sexual function, as patients experience fewer feelings of shame, anxiety, and depression, as well as improvement of physical symptoms that can interfere with sexual functioning.38,43,44 Reducing friction, warmth, and moisture, as well as avoiding tight clothing, can help those with genital psoriasis. Patients are advised to reapply topical medications after sexual intercourse. Patients also can apply makeup to disguise psoriasis and help reduce feelings of self-consciousness that can impede sexual intimacy.18
Comment
The psychosocial burden of psoriasis penetrates many facets of patient lives. Psoriasis can invoke feelings of shame and embarrassment that are worsened by the public’s misconceptions about psoriasis, resulting in serious mental health issues that can cause even greater disability. Depression and anxiety are prevalent in patients with psoriasis. The characteristic symptoms of pain and pruritus along with psychiatric comorbidities can have an underestimated impact on daily activities, including employment, interpersonal relationships, and sexual function. Such dysfunctions have serious implications toward wages, professional advancement, social support, and overall QOL.
Dermatology providers play an important role in screening for these problems through validated questionnaires and identifying risks. Simple screening questions such as the PHQ-9 can be beneficial and feasible during dermatology visits. Screening for PsA can help patients avoid problems at work. Sexual dysfunction is a sensitive topic; however, providers can use a 1-question screen from valid questionnaires and inquire about the location of lesions as opportunities to address this issue.
Interventions lead to better disease control, which concurrently improves overall QOL. These interventions depend on both patient adherence and a physician’s commitment to finding an optimal treatment regimen for each individual. Medical management; coordinating care; developing treatment plans with psychiatry, rheumatology, and primary care providers; and psychological counseling and services may be necessary and beneficial (Table). Offering accessible resources such as the NPF website helps patients access information outside the clinic when it is not feasible to address all these concerns in a single visit. Psoriasis requires more than just medical management; it requires dermatology providers to use a multidisciplinary approach to address the psychosocial aspects of the disease.

Conclusion
The psychosocial burden of psoriasis is immense. Stigma, public misconception, mental health concerns, and occupational and interpersonal difficulty are the basis of disease burden. Providers play a vital role in assessing the effect psoriasis has on different areas of patients’ lives and providing appropriate interventions and resources to reduce disease burden.
- Kimball AB, Jacobson C, Weiss S, et al. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383-392.
- Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PloS One. 2012;7:e52935.
- Halioua B, Sid-Mohand D, Roussel ME, et al. Extent of misconceptions, negative prejudices and discriminatory behaviour to psoriasis patients in France. J Eur Acad Dermatol Venereol. 2016;30:650-654.
- Wolf P, Weger W, Legat F, et al. Quality of life and treatment goals in psoriasis from the patient perspective: results of an Austrian cross-sectional survey. J Dtsch Dermatol Ges. 2018;16:981-990.
- Shah K, Mellars L, Changolkar A, et al. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77:287-292.e4.
- Dowlatshahi EA, Wakkee M, Herings RM, et al. Increased antidepressant drug exposure in psoriasis patients: a longitudinal population-based cohort study. Acta Derm Venereol. 2013;93:544-550.
- Dowlatshahi EA, Wakkee M, Arends LR, et al. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542-1551.
- Singh S, Taylor C, Kornmehl H, et al. Psoriasis and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77:425.e2-440.e2.
- Lakshmy S, Balasundaram S, Sarkar S, et al. A cross-sectional study of prevalence and implications of depression and anxiety in psoriasis. Indian J Psychol Med. 2015;37:434-440.
- Renzi C, Picardi A, Abeni D, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337-342.
- Kulkarni AS, Balkrishnan R, Camacho FT, et al. Medication and health care service utilization related to depressive symptoms in older adults with psoriasis. J Drugs Dermatol. 2004;3:661-666.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Lamb RC, Matcham F, Turner MA, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol. 2017;176:1028-1034.
- Law M, Naughton MT, Dhar A, et al. Validation of two depression screening instruments in a sleep disorders clinic. J Clin Sleep Med. 2014;10:683-688.
- Cuijpers P, Dekker J, Hollon SD, et al. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: a meta-analysis. J Clin Psychiatry. 2009;70:1219-1229.
- National Psoriasis Foundation. Living with psoriatic arthritis. https://www.psoriasis.org/life-with-psoriatic-arthritis. Accessed September 23, 2018.
- Gaikwad R, Deshpande S, Raje S, et al. Evaluation of functional impairment in psoriasis. Indian J Dermatol Venereol Leprol. 2006;72:37-40.
- Claudepierre P, Lahfa M, Levy P, et al. The impact of psoriasis on professional life: PsoPRO, a French national survey [published online April 6, 2018]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.14986.
- Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41:514-521.
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. PharmacoEconomics. 1993;4:353-365.
- Korman NJ, Zhao Y, Pike J, et al. Increased severity of itching, pain, and scaling in psoriasis patients is associated with increased disease severity, reduced quality of life, and reduced work productivity. Dermatol Online J. 2015;21.
- Spelman L, Su JC, Fernandez-Penas P, et al. Frequency of undiagnosed psoriatic arthritis among psoriasis patients in Australian dermatology practice. J Eur Acad Dermatol Venereol. 2015;29:2184-2191.
- Tillett W, Shaddick G, Askari A, et al. Factors influencing work disability in psoriatic arthritis: first results from a large UK multicentre study. Rheumatology (Oxford). 2015;54:157-162.
- Raychaudhuri SP, Wilken R, Sukhov AC, et al. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J Autoimmun. 2017;76:21-37.
- Gossec L, Smolen JS, Ramiro S, et al. European League Against Rheumatism (EULAR) recommendations for the manegement of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499-510.
- Beroukhim K, Danesh M, Nguyen C, et al. A prospective, interventional assessment of the impact of ustekinumab treatment on psoriasis-related work productivity and activity impairment. J Dermatol Treat. 2016;27:552-555.
- Armstrong AW, Lynde CW, McBride SR, et al. Effect of ixekizumab treatment on work productivity for patients with moderate-to-severe plaque psoriasis: analysis of results from 3 randomized phase 3 clinical trials. JAMA Dermatol. 2016;152:661-669.
- Kimball AB, Yu AP, Signorovitch J, et al. The effects of adalimumab treatment and psoriasis severity on self-reported work productivity and activity impairment for patients with moderate to severe psoriasis. J Am Acad Dermatol. 2012;66:e67-76.
- Feldman SR, Malakouti M, Koo JY. Social impact of the burden of psoriasis: effects on patients and practice. Dermatol Online J. 2014;20.
- Reich A, Welz-Kubiak K, Rams Ł. Apprehension of the disease by patients suffering from psoriasis. Postepy Dermatol Alergol. 2014;31:289-293.
- Gupta MA, Gupta AK, Watteel GN. Perceived deprivation of social touch in psoriasis is associated with greater psychologic morbidity: an index of the stigma experience in dermatologic disorders. Cutis. 1998;61:339-342.
- Basra MK, Finlay AY. The family impact of skin diseases: the Greater Patient concept. Br J Dermatol. 2007;156:929-937.
- Eghlileb AM, Davies EE, Finlay AY. Psoriasis has a major secondary impact on the lives of family members and partners. Br J Dermatol. 2007;156:1245-1250.
- Janowski K, Steuden S, Pietrzak A, et al. Social support and adaptation to the disease in men and women with psoriasis. Arch Dermatol Res. 2012;304:421-432.
- Sampogna F, Abeni D, Gieler U, et al. Impairment of sexual life in 3,485 dermatological outpatients from a multicentre study in 13 European countries. Acta Derm Venereol. 2017;97:478-482.
- Sampogna F, Gisondi P, Tabolli S, et al. Impairment of sexual life in patients with psoriasis. Dermatology. 2007;214:144-150.
- Molina-Leyva A, Molina-Leyva I, Almodovar-Real A, et al. Prevalence and associated factors of erectile dysfunction in patients with moderate to severe psoriasis and healthy population: a comparative study considering physical and psychological factors. Arch Sex Behav. 2016;45:2047-2055.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Labbate LA, Lare SB. Sexual dysfunction in male psychiatric outpatients: validity of the Massachusetts General Hospital Sexual Functioning Questionnaire. Psychother Psychosom. 2001;70:221-225.
- Molina-Leyva A, Almodovar-Real A, Ruiz-Carrascosa JC, et al. Distribution pattern of psoriasis affects sexual function in moderate to severe psoriasis: a prospective case series study. J Sex Med. 2014;11:2882-2889.
- Guenther L, Han C, Szapary P, et al. Impact of ustekinumab on health-related quality of life and sexual difficulties associated with psoriasis: results from two phase III clinical trials. J Eur Acad Dermatol Venereol. 2011;25:851-857.
- Guenther L, Warren RB, Cather JC, et al. Impact of ixekizumab treatment on skin-related personal relationship difficulties in moderate-to-severe psoriasis patients: 12-week results from two Phase 3 trials. J Eur Acad Dermatol Venereol. 2017;31:1867-1875.
- Kimball AB, Jacobson C, Weiss S, et al. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383-392.
- Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PloS One. 2012;7:e52935.
- Halioua B, Sid-Mohand D, Roussel ME, et al. Extent of misconceptions, negative prejudices and discriminatory behaviour to psoriasis patients in France. J Eur Acad Dermatol Venereol. 2016;30:650-654.
- Wolf P, Weger W, Legat F, et al. Quality of life and treatment goals in psoriasis from the patient perspective: results of an Austrian cross-sectional survey. J Dtsch Dermatol Ges. 2018;16:981-990.
- Shah K, Mellars L, Changolkar A, et al. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77:287-292.e4.
- Dowlatshahi EA, Wakkee M, Herings RM, et al. Increased antidepressant drug exposure in psoriasis patients: a longitudinal population-based cohort study. Acta Derm Venereol. 2013;93:544-550.
- Dowlatshahi EA, Wakkee M, Arends LR, et al. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542-1551.
- Singh S, Taylor C, Kornmehl H, et al. Psoriasis and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77:425.e2-440.e2.
- Lakshmy S, Balasundaram S, Sarkar S, et al. A cross-sectional study of prevalence and implications of depression and anxiety in psoriasis. Indian J Psychol Med. 2015;37:434-440.
- Renzi C, Picardi A, Abeni D, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337-342.
- Kulkarni AS, Balkrishnan R, Camacho FT, et al. Medication and health care service utilization related to depressive symptoms in older adults with psoriasis. J Drugs Dermatol. 2004;3:661-666.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Lamb RC, Matcham F, Turner MA, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol. 2017;176:1028-1034.
- Law M, Naughton MT, Dhar A, et al. Validation of two depression screening instruments in a sleep disorders clinic. J Clin Sleep Med. 2014;10:683-688.
- Cuijpers P, Dekker J, Hollon SD, et al. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: a meta-analysis. J Clin Psychiatry. 2009;70:1219-1229.
- National Psoriasis Foundation. Living with psoriatic arthritis. https://www.psoriasis.org/life-with-psoriatic-arthritis. Accessed September 23, 2018.
- Gaikwad R, Deshpande S, Raje S, et al. Evaluation of functional impairment in psoriasis. Indian J Dermatol Venereol Leprol. 2006;72:37-40.
- Claudepierre P, Lahfa M, Levy P, et al. The impact of psoriasis on professional life: PsoPRO, a French national survey [published online April 6, 2018]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.14986.
- Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41:514-521.
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. PharmacoEconomics. 1993;4:353-365.
- Korman NJ, Zhao Y, Pike J, et al. Increased severity of itching, pain, and scaling in psoriasis patients is associated with increased disease severity, reduced quality of life, and reduced work productivity. Dermatol Online J. 2015;21.
- Spelman L, Su JC, Fernandez-Penas P, et al. Frequency of undiagnosed psoriatic arthritis among psoriasis patients in Australian dermatology practice. J Eur Acad Dermatol Venereol. 2015;29:2184-2191.
- Tillett W, Shaddick G, Askari A, et al. Factors influencing work disability in psoriatic arthritis: first results from a large UK multicentre study. Rheumatology (Oxford). 2015;54:157-162.
- Raychaudhuri SP, Wilken R, Sukhov AC, et al. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J Autoimmun. 2017;76:21-37.
- Gossec L, Smolen JS, Ramiro S, et al. European League Against Rheumatism (EULAR) recommendations for the manegement of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499-510.
- Beroukhim K, Danesh M, Nguyen C, et al. A prospective, interventional assessment of the impact of ustekinumab treatment on psoriasis-related work productivity and activity impairment. J Dermatol Treat. 2016;27:552-555.
- Armstrong AW, Lynde CW, McBride SR, et al. Effect of ixekizumab treatment on work productivity for patients with moderate-to-severe plaque psoriasis: analysis of results from 3 randomized phase 3 clinical trials. JAMA Dermatol. 2016;152:661-669.
- Kimball AB, Yu AP, Signorovitch J, et al. The effects of adalimumab treatment and psoriasis severity on self-reported work productivity and activity impairment for patients with moderate to severe psoriasis. J Am Acad Dermatol. 2012;66:e67-76.
- Feldman SR, Malakouti M, Koo JY. Social impact of the burden of psoriasis: effects on patients and practice. Dermatol Online J. 2014;20.
- Reich A, Welz-Kubiak K, Rams Ł. Apprehension of the disease by patients suffering from psoriasis. Postepy Dermatol Alergol. 2014;31:289-293.
- Gupta MA, Gupta AK, Watteel GN. Perceived deprivation of social touch in psoriasis is associated with greater psychologic morbidity: an index of the stigma experience in dermatologic disorders. Cutis. 1998;61:339-342.
- Basra MK, Finlay AY. The family impact of skin diseases: the Greater Patient concept. Br J Dermatol. 2007;156:929-937.
- Eghlileb AM, Davies EE, Finlay AY. Psoriasis has a major secondary impact on the lives of family members and partners. Br J Dermatol. 2007;156:1245-1250.
- Janowski K, Steuden S, Pietrzak A, et al. Social support and adaptation to the disease in men and women with psoriasis. Arch Dermatol Res. 2012;304:421-432.
- Sampogna F, Abeni D, Gieler U, et al. Impairment of sexual life in 3,485 dermatological outpatients from a multicentre study in 13 European countries. Acta Derm Venereol. 2017;97:478-482.
- Sampogna F, Gisondi P, Tabolli S, et al. Impairment of sexual life in patients with psoriasis. Dermatology. 2007;214:144-150.
- Molina-Leyva A, Molina-Leyva I, Almodovar-Real A, et al. Prevalence and associated factors of erectile dysfunction in patients with moderate to severe psoriasis and healthy population: a comparative study considering physical and psychological factors. Arch Sex Behav. 2016;45:2047-2055.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Labbate LA, Lare SB. Sexual dysfunction in male psychiatric outpatients: validity of the Massachusetts General Hospital Sexual Functioning Questionnaire. Psychother Psychosom. 2001;70:221-225.
- Molina-Leyva A, Almodovar-Real A, Ruiz-Carrascosa JC, et al. Distribution pattern of psoriasis affects sexual function in moderate to severe psoriasis: a prospective case series study. J Sex Med. 2014;11:2882-2889.
- Guenther L, Han C, Szapary P, et al. Impact of ustekinumab on health-related quality of life and sexual difficulties associated with psoriasis: results from two phase III clinical trials. J Eur Acad Dermatol Venereol. 2011;25:851-857.
- Guenther L, Warren RB, Cather JC, et al. Impact of ixekizumab treatment on skin-related personal relationship difficulties in moderate-to-severe psoriasis patients: 12-week results from two Phase 3 trials. J Eur Acad Dermatol Venereol. 2017;31:1867-1875.
Practice Points
- The psychosocial impact of psoriasis is an important component of the disease burden leading to reduced quality of life.
- Assessment of psychosocial dysfunction can be done through short questionnaires, asking patients directly about these issues and anticipating these problems in patients who are most vulnerable.
- Management of psychosocial impact ranges from pharmacological interventions to helpful resources such as the National Psoriasis Foundation website.
Psoriasis Risk Factors and Triggers
Psoriasis is a chronic autoimmune skin disease affecting approximately 6.7 million adults in the United States.1 Although its pathogenesis is not yet clear, risk factors and triggers provide insight into potential pathways by which psoriasis can occur. There is notable overlap between risk factors and triggers of psoriasis; perceived risk factors might, in fact, be triggers causing manifestation of disease in predisposed persons. In this review, we summarize the key factors contributing to onset and exacerbation of psoriasis. When learning to manage this chronic disease, it also may be helpful to educate patients about how these elements may affect the course of psoriasis.
Genetics
The pathogenesis of psoriasis has a strong genetic component, with approximately 70% and 20% concordance rates in monozygotic and dizygotic twins, respectively.2 Moreover, studies have shown a positive family history in approximately 35% of patients.3,4 Family-based studies have found a 50% risk of developing psoriasis in patients with 2 affected parents.5 However, the genetics of psoriasis are complex and are attributed to many different genes. Thus far, genes involving antigen presentation, T-cell receptor development and polarization, and the nuclear factor κβ (NF-κβ) pathway have been identified.6
HLA-Cw6
The most well-studied gene implicated in psoriasis is HLA-Cw6, which encodes a major histocompatibility complex class I allele supporting psoriasis as a T cell–mediated reaction to an autoantigen.6 Two potential antigens for HLA-Cw6 recently have been identified: LL-37, a cathelicidin-related antimicrobial peptide, and the A disintegrin and metalloproteinase with thrombospondin motifs-like protein 5 (ADAMTSL5), found on melanocytes and keratinocytes.7 The percentage of psoriasis patients with HLA-Cw6 ranges from 10.5% to 77.2%, with higher frequency in white individuals than in Asians.7
HLA-Cw6 manifests as specific features in psoriasis, including onset of disease before 21 years of age.8 It also is more strongly associated with guttate-type psoriasis, greater body surface area involvement, and higher incidence of Köbner phenomenon. Patients with positive HLA-Cw6 also reported worsening of psoriasis during and after throat infection.9
Caspase Recruitment Domain Family Member 14
Another gene mutation implicated in psoriasis pathogenesis is caspase recruitment domain family member 14, CARD14 (formerly PSORS2), a gene encoding a scaffolding protein important in the activation of NF-κβ.10,11 Missense CARD14 mutations cause upregulation of NF-κβ through formation of a complex with adapter protein B-cell lymphoma 10 (BCL10) and mucosa-associated lymphoid tissue lymphoma translocation protein 1 (MALT1),12 which, in turn, causes increased transcription of cytokines IL-8, C-C motif chemokine ligand 20 (CCL-20), and IL-36 gamma in the keratinocyte.13 Mutations in CARD14 alone lead to psoriasiform skin in mice through amplified activation of the IL-23/IL-17 axis.14,15 Patients with a mutation in a CARD14 variant (p.Arg820Trp) have demonstrated better response to tumor necrosis factor (TNF) inhibitors.16
Further characterization of the genetic pathogenesis of psoriasis might lead to better targeted therapies, including the possibility of MALT1 inhibitors as a treatment option.12
Infection
Streptococcus
The association between streptococcal infection and psoriasis was first documented more than 100 years ago, specifically the onset of acute guttate psoriasis.17,18 Although classically described following throat infection, psoriasis also occurs following streptococcal vulvovaginitis and perianal streptococcal infection.19,20
This type of psoriasis is typically self-limited but can recur with subsequent streptococcal infections or initiate a more chronic plaque psoriasis. Patients have a 1 in 3 risk of developing chronic psoriasis within 10 years of a single episode of acute guttate psoriasis.21 Moreover, in many patients with existing plaque psoriasis, throat infection exacerbates psoriatic symptoms.22 The mechanism of exacerbation is likely due to cross-reactivity between streptococcal M surface antigen and human keratinocytes and might also be influenced by inherited abnormalities in immune response.23-26 Therefore, tonsillectomy has been studied as a possible treatment of psoriasis but is likely helpful only in patients with exacerbations of disease that are closely associated with recurrent tonsillitis.27
Human Immunodeficiency Virus
The prevalence of psoriasis in human immunodeficiency virus (HIV) patients is similar to or greater than the general population.28 Human immunodeficiency virus infection causes new onset of psoriasis and exacerbation of existing psoriasis; severity often is correlated with worsening immune function.28,29
The clinical subtypes of psoriasis that occur most frequently with HIV include guttate, inverse, and erythrodermic, though patients may present with any subtype.28 The mechanism is puzzling because HIV is primarily mediated by helper T cell 2 (TH2) cytokines, whereas psoriasis is mainly driven by helper T cell 1 (TH1) cytokines.30 Furthermore, despite increased severity with lower CD4+ counts, treatments further lowering T-cell counts paradoxically improve symptoms.31 Current literature suggests that expansion of CD8+ memory T cells might be the primary mechanism in the exacerbation of psoriasis in HIV-mediated immunosuppression.30
Treatment of HIV-associated psoriasis presents challenges because many therapeutics cause further immunosuppression. The National Psoriasis Foundation recommends topical preparations as first-line agents for mild to moderate psoriasis.32 For moderate to severe psoriasis, retroviral agents may be effective as first-line monotherapy or when supplemented by phototherapy with UVB or psoralen plus UVA. Retinoids can be used as second-line agents.32 For cases of severe refractory psoriasis, cyclosporine, methotrexate, TNF inhibitors, or hydroxyurea can be considered. There also is evidence that apremilast is effective without risk for worsening immune function.33
Other Infections
Other bacteria associated with triggering or exacerbating psoriasis include Staphylococcus aureus and Helicobacter pylori.34,35 Fungi, such as species of the genera Malassezia and Candida, and other viruses, including papillomaviruses and retroviruses, also have been implicated.34
Medications
Numerous medications can trigger psoriasis, including lithium, nonsteroidal anti-inflammatory drugs, antimalarials, beta-blockers, and angiotensin-converting enzyme inhibitors.34 More recent literature suggests that TNF inhibitors also can paradoxically induce psoriasis in rare cases.35
Lithium
Psoriasis is the most common cutaneous adverse effect of lithium.34 It is more likely to exacerbate existing disease but also can induce onset of psoriasis; it also can cause disease that is more refractory to treatment.34,36 Current literature hypothesizes that lithium triggers psoriasis by interference of intracellular calcium channels through reduction of inositol, thereby affecting keratinocyte proliferation and differentiation.34 Lithium also inhibits glycogen synthase kinase-3 (GSK-3), a serine threonine kinase, which, in turn, induces human keratinocyte proliferation.37 However, it is unlikely lithium alone can induce psoriasis; genetic predisposition is necessary.
TNF Inhibitors
Tumor necrosis factor inhibitors such as adalimumab, etanercept, certolizumab pegol, golimumab, and infliximab are used in various inflammatory diseases, including psoriasis. Interestingly, there have been more than 200 reported cases of suspected TNF inhibitor–induced or –exacerbated psoriasis.38 This phenomenon appears to occur more frequently with infliximab and is most likely to occur in the first year of treatment of Crohn disease and rheumatoid arthritis.38 Plaque psoriasis is the most common form, but 15% to 26% of cases presented with 2 or more morphologies.38,39
Treatment options include discontinuing therapy, though many patients experience resolution while continuing treatment or switching to another TNF inhibitor.38-40 Traditional topical therapies also have been used with success.40 The pathogenesis of this phenomenon is still unclear but is thought to involve both the IL-23/helper T cell 17 (TH17) axis and dysregulation of IFN-α in the setting of TNF suppression.38
Lifestyle
Obesity is a chronic low-grade inflammatory state that can contribute to the onset of psoriasis or exacerbation of exist
The relationship between psoriasis and alcohol consumption is less clear than it is between psoriasis and obesity or smoking; greater consumption is found in psoriasis patients, but evidence is insufficient to deem alcohol a risk factor.44
Conclusion
Various factors, including genetics, infection, pharmacotherapeutic, and lifestyle, can all contribute to the induction or exacerbation of psoriasis. These factors can provide clues to the pathogenesis of psoriasis as well as help clinicians better counsel patients about their disease.
- Helmick CG, Lee-Han H, Hirsch SC, et al. Prevalence of psoriasis among adults in the U.S.: 2003-2006 and 2009-2010 National Health and Nutrition Examination Surveys. Am J Prev Med. 2014;47:37-45.
- Bowcock AM. The genetics of psoriasis and autoimmunity. Annu Rev Genomics Hum Genet. 2005;6:93-122.
- Swanbeck G, Inerot A, Martinsson T, et al. A population genetic study of psoriasis. Br J Dermatol. 1994;131:32-39.
- Kimberling W, Dobson RL. The inheritance of psoriasis. J Invest Dermatol. 1973;60:538-540.
- Gupta R, Debbaneh MG, Liao W. Genetic epidemiology of psoriasis. Curr Dermatol Rep. 2014;3:61-78.
- Harden JL, Krueger JG, Bowcock AM. The immunogenetics of psoriasis: a comprehensive review. J Autoimmun. 2015;64:66-73.
- Chen L, Tsai TF. HLA-Cw6 and psoriasis. Br J Dermatol. 2018;178:854-862.
- Enerbäck C, Martinsson T, Ineraot A, et al. Evidence that HLA-Cw6 determines early onset of psoriasis, obtained using sequence-specific primers (PCR-SSP). Acta Derm Venereol. 1997;77:273-276.
- Gudjónsson JE, Kárason A, Antonsdóttir EH, et al. HLA-Cw6-positive and HLA-Cw6-negative patients with psoriasis vulgaris have distinct clinical features. J Invest Dermatol. 2002;118:362-365.
- Tomfohrde J, Silverman A, Barnes R, et al. Gene for familial psoriasis susceptibility mapped to distal end of human chromosome 17q. Science. 1994;264:1141-1145.
- Blonska M, Lin X. NF-κB signaling pathways regulated by CARMA family of scaffold proteins. Cell Res. 2011;21:55-70.
- Van Nuffel E, Schmitt A, Afonina IS, et al. CARD14-mediated activation of paracaspase MALT1 in keratinocytes: implications for psoriasis. J Invest Dermatol. 2017;137:569-575.
- Jordan CT, Cao L, Roberson ED, et al. PSORS2 is due to mutations in CARD14. Am J Hum Genet. 2012;90:784-795.
- Wang M, Zhang S, Zheng G, et al. Gain-of-function mutation of Card14 leads to spontaneous psoriasis-like skin inflammation through enhanced keratinocyte response to IL-17A. Immunity. 2018;49:66-79.
- Mellet M, Meier B, Mohanan D, et al. CARD14 gain-of-function mutation alone is sufficient to drive IL-23/IL-17-mediated psoriasiform skin inflammation in vivo. J Invest Dermatol. 2018;138:2010-2023.
- Coto-Segura P, González-Fernández D, Batalla A, et al. Common and rare CARD14 gene variants affect the antitumour necrosis factor response among patients with psoriasis. Br J Dermatol. 2016;175:134-141.
- Winfield JM. Psoriasis as a sequel to acute inflammations of the tonsils: a clinical note. J Cutan Dis. 1916;34:441-443.
- Telfer NR, Chalmers RJG, Whale K, et al. The role of streptococcal infection in the initiation of guttate psoriasis. Arch Dermatol. 1992;128:39-42.
- Hernandez M, Simms-Cendan J, Zendell K. Guttate psoriasis following streptococcal vulvovaginitis in a five-year-old girl. J Pediatr Adolesc Gynecol. 2015;28:e127-e129.
- Herbst RA, Hoch O, Kapp A, et al. Guttate psoriasis triggered by perianal streptococcal dermatitis in a four-year-old boy. J Am Acad Dermatol. 2000;42(5, pt 2):885-887.
- Martin BA, Chalmers RJ, Telfer NR. How great is the risk of further psoriasis following a single episode of acute guttate psoriasis? Arch Dermatol. 1996;132:717-718.
- Thorleifsdottir RH, Eysteinsdóttir, Olafsson JH, et al. Throat infections are associated with exacerbation in a substantial proportion of patients with chronic plaque psoriasis. Acta Derm Venereol. 2016;96:788-791.
- McFadden J, Valdimarsson H, Fry L. Cross-reactivity between streptococcal M surface antigen and human skin. Br J Dermatol. 1991;125:443-447.
- Validmarsson H, Thorleifsdottir RH, Sigurdardottir SL, et al. Psoriasis—as an autoimmune disease caused by molecular mimicry. Trends Immunol. 2009;30:494-501.
- Muto M, Fujikara Y, Hamamoto Y, et al. Immune response to Streptococcus pyogenes and the susceptibility to psoriasis. Australas J Dermatol. 1996;37(suppl 1):S54-S55.
- Weisenseel P, Laumbacher B, Besgen P, et al. Streptococcal infection distinguishes different types of psoriasis. J Med Genet. 2002;39:767-768.
- Rachakonda TD, Dhillon JS, Florek AG, et al. Effect of tonsillectomy on psoriasis: a systematic review. J Am Acad Dermatol. 2015;72:261-275.
- Mallon E, Bunker CB. HIV-associated psoriasis. AIDS Patient Care STDS. 2000;14:239-246.
- Duvic M, Johnson TM, Rapini RP, et al. Acquired immunodeficiency syndrome-associated psoriasis and Reiter’s syndrome. Arch Dermatol. 1987;123:1622-1632.
- Fife DJ, Waller JM, Jeffes EW, et al. Unraveling the paradoxes of HIV-associated psoriasis: a review of T-cell subsets and cytokine profiles. Dermatol Online J. 2007;13:4.
- Ortonne JP, Lebwohl M, Em Griffiths C; Alefacept Clinical Study Group. Alefacept-induced decreases in circulating blood lymphocyte counts correlate with clinical response in patients with chronic plaque psoriasis. Eur J Dermatol. 2003;13:117-123.
- Menon K, Van Voorhees AS, Bebo BF Jr, et al; National Psoriasis Foundation. Psoriasis in patients with HIV infection: from the medical board of the National Psoriasis Foundation. J Am Acad Dermatol. 2010;62:291-299.
- Reddy SP, Shah VV, Wu JJ. Apremilast for a psoriasis patient with HIV and hepatitis C. J Eur Acad Dermatol Venereol. 2017;31:e481-e482.
- Fry L, Baker BS. Triggering psoriasis: the role of infections and medications. Clin Dermatol. 2007;25:606-615.
- Sfikakis PP, Iliopoulos A, Elezoglou A, et al. Psoriasis induced by anti-tumor necrosis factor therapy: a paradoxical adverse reaction. Arthritis Rheum. 2005;52:2513-2518.
- Yeung CK, Chan HH. Cutaneous adverse effects of lithium: epidemiology and management. Am J Clin Dermatol. 2004;5:3-8.
- Hampton PJ, Jans R, Flockhart RJ, et al. Lithium regulates keratinocyte proliferation via glycogen synthase kinase 3 and NFAT 2 (nuclear factor of activated T cells 2). J Cell Physiol. 2012;227:1529-1537.
- Brown G, Wang E, Leon A, et al. Tumor necrosis factor-α inhibitor-induced psoriasis: systematic review of clinical features, histopathological findings, and management experience. J Am Acad Dermatol. 2017;76:334-341.
- Collamer AN, Battafarano DF. Psoriatic skin lesions induced by tumor necrosis factor antagonist therapy: clinical features and possible immunopathogenesis. Semin Arthritis Rheum. 2010;40:233-240.
- Collamer AN, Guerrero KT, Henning JS, et al. Psoriatic skin lesions induced by tumor antagonist therapy: a literature review and potential mechanisms of action. Arthritis Rheum. 2008;59:996-1001.
- Jensen P, Skov L. Psoriasis and obesity. Dermatology. 2016;232:633-639.
- Barrea L, Nappi F, Di Somma C, et al. Environmental risk factors in psoriasis: the point of view of the nutritionist. Int J Environ Res Public Health. 2016;13:743.
- Lee EJ, Han KD, Han JH, et al. Smoking and risk of psoriasis: a nationwide cohort study. J Am Acad Dermatol. 2017;77:573-575.
- Brenaut E, Horreau C, Pouplard C, et al. Alcohol consumption and psoriasis: a systematic literature review. J Eur Acad Dermatol Venereol. 2013;27(suppl 3):30-35.
Psoriasis is a chronic autoimmune skin disease affecting approximately 6.7 million adults in the United States.1 Although its pathogenesis is not yet clear, risk factors and triggers provide insight into potential pathways by which psoriasis can occur. There is notable overlap between risk factors and triggers of psoriasis; perceived risk factors might, in fact, be triggers causing manifestation of disease in predisposed persons. In this review, we summarize the key factors contributing to onset and exacerbation of psoriasis. When learning to manage this chronic disease, it also may be helpful to educate patients about how these elements may affect the course of psoriasis.
Genetics
The pathogenesis of psoriasis has a strong genetic component, with approximately 70% and 20% concordance rates in monozygotic and dizygotic twins, respectively.2 Moreover, studies have shown a positive family history in approximately 35% of patients.3,4 Family-based studies have found a 50% risk of developing psoriasis in patients with 2 affected parents.5 However, the genetics of psoriasis are complex and are attributed to many different genes. Thus far, genes involving antigen presentation, T-cell receptor development and polarization, and the nuclear factor κβ (NF-κβ) pathway have been identified.6
HLA-Cw6
The most well-studied gene implicated in psoriasis is HLA-Cw6, which encodes a major histocompatibility complex class I allele supporting psoriasis as a T cell–mediated reaction to an autoantigen.6 Two potential antigens for HLA-Cw6 recently have been identified: LL-37, a cathelicidin-related antimicrobial peptide, and the A disintegrin and metalloproteinase with thrombospondin motifs-like protein 5 (ADAMTSL5), found on melanocytes and keratinocytes.7 The percentage of psoriasis patients with HLA-Cw6 ranges from 10.5% to 77.2%, with higher frequency in white individuals than in Asians.7
HLA-Cw6 manifests as specific features in psoriasis, including onset of disease before 21 years of age.8 It also is more strongly associated with guttate-type psoriasis, greater body surface area involvement, and higher incidence of Köbner phenomenon. Patients with positive HLA-Cw6 also reported worsening of psoriasis during and after throat infection.9
Caspase Recruitment Domain Family Member 14
Another gene mutation implicated in psoriasis pathogenesis is caspase recruitment domain family member 14, CARD14 (formerly PSORS2), a gene encoding a scaffolding protein important in the activation of NF-κβ.10,11 Missense CARD14 mutations cause upregulation of NF-κβ through formation of a complex with adapter protein B-cell lymphoma 10 (BCL10) and mucosa-associated lymphoid tissue lymphoma translocation protein 1 (MALT1),12 which, in turn, causes increased transcription of cytokines IL-8, C-C motif chemokine ligand 20 (CCL-20), and IL-36 gamma in the keratinocyte.13 Mutations in CARD14 alone lead to psoriasiform skin in mice through amplified activation of the IL-23/IL-17 axis.14,15 Patients with a mutation in a CARD14 variant (p.Arg820Trp) have demonstrated better response to tumor necrosis factor (TNF) inhibitors.16
Further characterization of the genetic pathogenesis of psoriasis might lead to better targeted therapies, including the possibility of MALT1 inhibitors as a treatment option.12
Infection
Streptococcus
The association between streptococcal infection and psoriasis was first documented more than 100 years ago, specifically the onset of acute guttate psoriasis.17,18 Although classically described following throat infection, psoriasis also occurs following streptococcal vulvovaginitis and perianal streptococcal infection.19,20
This type of psoriasis is typically self-limited but can recur with subsequent streptococcal infections or initiate a more chronic plaque psoriasis. Patients have a 1 in 3 risk of developing chronic psoriasis within 10 years of a single episode of acute guttate psoriasis.21 Moreover, in many patients with existing plaque psoriasis, throat infection exacerbates psoriatic symptoms.22 The mechanism of exacerbation is likely due to cross-reactivity between streptococcal M surface antigen and human keratinocytes and might also be influenced by inherited abnormalities in immune response.23-26 Therefore, tonsillectomy has been studied as a possible treatment of psoriasis but is likely helpful only in patients with exacerbations of disease that are closely associated with recurrent tonsillitis.27
Human Immunodeficiency Virus
The prevalence of psoriasis in human immunodeficiency virus (HIV) patients is similar to or greater than the general population.28 Human immunodeficiency virus infection causes new onset of psoriasis and exacerbation of existing psoriasis; severity often is correlated with worsening immune function.28,29
The clinical subtypes of psoriasis that occur most frequently with HIV include guttate, inverse, and erythrodermic, though patients may present with any subtype.28 The mechanism is puzzling because HIV is primarily mediated by helper T cell 2 (TH2) cytokines, whereas psoriasis is mainly driven by helper T cell 1 (TH1) cytokines.30 Furthermore, despite increased severity with lower CD4+ counts, treatments further lowering T-cell counts paradoxically improve symptoms.31 Current literature suggests that expansion of CD8+ memory T cells might be the primary mechanism in the exacerbation of psoriasis in HIV-mediated immunosuppression.30
Treatment of HIV-associated psoriasis presents challenges because many therapeutics cause further immunosuppression. The National Psoriasis Foundation recommends topical preparations as first-line agents for mild to moderate psoriasis.32 For moderate to severe psoriasis, retroviral agents may be effective as first-line monotherapy or when supplemented by phototherapy with UVB or psoralen plus UVA. Retinoids can be used as second-line agents.32 For cases of severe refractory psoriasis, cyclosporine, methotrexate, TNF inhibitors, or hydroxyurea can be considered. There also is evidence that apremilast is effective without risk for worsening immune function.33
Other Infections
Other bacteria associated with triggering or exacerbating psoriasis include Staphylococcus aureus and Helicobacter pylori.34,35 Fungi, such as species of the genera Malassezia and Candida, and other viruses, including papillomaviruses and retroviruses, also have been implicated.34
Medications
Numerous medications can trigger psoriasis, including lithium, nonsteroidal anti-inflammatory drugs, antimalarials, beta-blockers, and angiotensin-converting enzyme inhibitors.34 More recent literature suggests that TNF inhibitors also can paradoxically induce psoriasis in rare cases.35
Lithium
Psoriasis is the most common cutaneous adverse effect of lithium.34 It is more likely to exacerbate existing disease but also can induce onset of psoriasis; it also can cause disease that is more refractory to treatment.34,36 Current literature hypothesizes that lithium triggers psoriasis by interference of intracellular calcium channels through reduction of inositol, thereby affecting keratinocyte proliferation and differentiation.34 Lithium also inhibits glycogen synthase kinase-3 (GSK-3), a serine threonine kinase, which, in turn, induces human keratinocyte proliferation.37 However, it is unlikely lithium alone can induce psoriasis; genetic predisposition is necessary.
TNF Inhibitors
Tumor necrosis factor inhibitors such as adalimumab, etanercept, certolizumab pegol, golimumab, and infliximab are used in various inflammatory diseases, including psoriasis. Interestingly, there have been more than 200 reported cases of suspected TNF inhibitor–induced or –exacerbated psoriasis.38 This phenomenon appears to occur more frequently with infliximab and is most likely to occur in the first year of treatment of Crohn disease and rheumatoid arthritis.38 Plaque psoriasis is the most common form, but 15% to 26% of cases presented with 2 or more morphologies.38,39
Treatment options include discontinuing therapy, though many patients experience resolution while continuing treatment or switching to another TNF inhibitor.38-40 Traditional topical therapies also have been used with success.40 The pathogenesis of this phenomenon is still unclear but is thought to involve both the IL-23/helper T cell 17 (TH17) axis and dysregulation of IFN-α in the setting of TNF suppression.38
Lifestyle
Obesity is a chronic low-grade inflammatory state that can contribute to the onset of psoriasis or exacerbation of exist
The relationship between psoriasis and alcohol consumption is less clear than it is between psoriasis and obesity or smoking; greater consumption is found in psoriasis patients, but evidence is insufficient to deem alcohol a risk factor.44
Conclusion
Various factors, including genetics, infection, pharmacotherapeutic, and lifestyle, can all contribute to the induction or exacerbation of psoriasis. These factors can provide clues to the pathogenesis of psoriasis as well as help clinicians better counsel patients about their disease.
Psoriasis is a chronic autoimmune skin disease affecting approximately 6.7 million adults in the United States.1 Although its pathogenesis is not yet clear, risk factors and triggers provide insight into potential pathways by which psoriasis can occur. There is notable overlap between risk factors and triggers of psoriasis; perceived risk factors might, in fact, be triggers causing manifestation of disease in predisposed persons. In this review, we summarize the key factors contributing to onset and exacerbation of psoriasis. When learning to manage this chronic disease, it also may be helpful to educate patients about how these elements may affect the course of psoriasis.
Genetics
The pathogenesis of psoriasis has a strong genetic component, with approximately 70% and 20% concordance rates in monozygotic and dizygotic twins, respectively.2 Moreover, studies have shown a positive family history in approximately 35% of patients.3,4 Family-based studies have found a 50% risk of developing psoriasis in patients with 2 affected parents.5 However, the genetics of psoriasis are complex and are attributed to many different genes. Thus far, genes involving antigen presentation, T-cell receptor development and polarization, and the nuclear factor κβ (NF-κβ) pathway have been identified.6
HLA-Cw6
The most well-studied gene implicated in psoriasis is HLA-Cw6, which encodes a major histocompatibility complex class I allele supporting psoriasis as a T cell–mediated reaction to an autoantigen.6 Two potential antigens for HLA-Cw6 recently have been identified: LL-37, a cathelicidin-related antimicrobial peptide, and the A disintegrin and metalloproteinase with thrombospondin motifs-like protein 5 (ADAMTSL5), found on melanocytes and keratinocytes.7 The percentage of psoriasis patients with HLA-Cw6 ranges from 10.5% to 77.2%, with higher frequency in white individuals than in Asians.7
HLA-Cw6 manifests as specific features in psoriasis, including onset of disease before 21 years of age.8 It also is more strongly associated with guttate-type psoriasis, greater body surface area involvement, and higher incidence of Köbner phenomenon. Patients with positive HLA-Cw6 also reported worsening of psoriasis during and after throat infection.9
Caspase Recruitment Domain Family Member 14
Another gene mutation implicated in psoriasis pathogenesis is caspase recruitment domain family member 14, CARD14 (formerly PSORS2), a gene encoding a scaffolding protein important in the activation of NF-κβ.10,11 Missense CARD14 mutations cause upregulation of NF-κβ through formation of a complex with adapter protein B-cell lymphoma 10 (BCL10) and mucosa-associated lymphoid tissue lymphoma translocation protein 1 (MALT1),12 which, in turn, causes increased transcription of cytokines IL-8, C-C motif chemokine ligand 20 (CCL-20), and IL-36 gamma in the keratinocyte.13 Mutations in CARD14 alone lead to psoriasiform skin in mice through amplified activation of the IL-23/IL-17 axis.14,15 Patients with a mutation in a CARD14 variant (p.Arg820Trp) have demonstrated better response to tumor necrosis factor (TNF) inhibitors.16
Further characterization of the genetic pathogenesis of psoriasis might lead to better targeted therapies, including the possibility of MALT1 inhibitors as a treatment option.12
Infection
Streptococcus
The association between streptococcal infection and psoriasis was first documented more than 100 years ago, specifically the onset of acute guttate psoriasis.17,18 Although classically described following throat infection, psoriasis also occurs following streptococcal vulvovaginitis and perianal streptococcal infection.19,20
This type of psoriasis is typically self-limited but can recur with subsequent streptococcal infections or initiate a more chronic plaque psoriasis. Patients have a 1 in 3 risk of developing chronic psoriasis within 10 years of a single episode of acute guttate psoriasis.21 Moreover, in many patients with existing plaque psoriasis, throat infection exacerbates psoriatic symptoms.22 The mechanism of exacerbation is likely due to cross-reactivity between streptococcal M surface antigen and human keratinocytes and might also be influenced by inherited abnormalities in immune response.23-26 Therefore, tonsillectomy has been studied as a possible treatment of psoriasis but is likely helpful only in patients with exacerbations of disease that are closely associated with recurrent tonsillitis.27
Human Immunodeficiency Virus
The prevalence of psoriasis in human immunodeficiency virus (HIV) patients is similar to or greater than the general population.28 Human immunodeficiency virus infection causes new onset of psoriasis and exacerbation of existing psoriasis; severity often is correlated with worsening immune function.28,29
The clinical subtypes of psoriasis that occur most frequently with HIV include guttate, inverse, and erythrodermic, though patients may present with any subtype.28 The mechanism is puzzling because HIV is primarily mediated by helper T cell 2 (TH2) cytokines, whereas psoriasis is mainly driven by helper T cell 1 (TH1) cytokines.30 Furthermore, despite increased severity with lower CD4+ counts, treatments further lowering T-cell counts paradoxically improve symptoms.31 Current literature suggests that expansion of CD8+ memory T cells might be the primary mechanism in the exacerbation of psoriasis in HIV-mediated immunosuppression.30
Treatment of HIV-associated psoriasis presents challenges because many therapeutics cause further immunosuppression. The National Psoriasis Foundation recommends topical preparations as first-line agents for mild to moderate psoriasis.32 For moderate to severe psoriasis, retroviral agents may be effective as first-line monotherapy or when supplemented by phototherapy with UVB or psoralen plus UVA. Retinoids can be used as second-line agents.32 For cases of severe refractory psoriasis, cyclosporine, methotrexate, TNF inhibitors, or hydroxyurea can be considered. There also is evidence that apremilast is effective without risk for worsening immune function.33
Other Infections
Other bacteria associated with triggering or exacerbating psoriasis include Staphylococcus aureus and Helicobacter pylori.34,35 Fungi, such as species of the genera Malassezia and Candida, and other viruses, including papillomaviruses and retroviruses, also have been implicated.34
Medications
Numerous medications can trigger psoriasis, including lithium, nonsteroidal anti-inflammatory drugs, antimalarials, beta-blockers, and angiotensin-converting enzyme inhibitors.34 More recent literature suggests that TNF inhibitors also can paradoxically induce psoriasis in rare cases.35
Lithium
Psoriasis is the most common cutaneous adverse effect of lithium.34 It is more likely to exacerbate existing disease but also can induce onset of psoriasis; it also can cause disease that is more refractory to treatment.34,36 Current literature hypothesizes that lithium triggers psoriasis by interference of intracellular calcium channels through reduction of inositol, thereby affecting keratinocyte proliferation and differentiation.34 Lithium also inhibits glycogen synthase kinase-3 (GSK-3), a serine threonine kinase, which, in turn, induces human keratinocyte proliferation.37 However, it is unlikely lithium alone can induce psoriasis; genetic predisposition is necessary.
TNF Inhibitors
Tumor necrosis factor inhibitors such as adalimumab, etanercept, certolizumab pegol, golimumab, and infliximab are used in various inflammatory diseases, including psoriasis. Interestingly, there have been more than 200 reported cases of suspected TNF inhibitor–induced or –exacerbated psoriasis.38 This phenomenon appears to occur more frequently with infliximab and is most likely to occur in the first year of treatment of Crohn disease and rheumatoid arthritis.38 Plaque psoriasis is the most common form, but 15% to 26% of cases presented with 2 or more morphologies.38,39
Treatment options include discontinuing therapy, though many patients experience resolution while continuing treatment or switching to another TNF inhibitor.38-40 Traditional topical therapies also have been used with success.40 The pathogenesis of this phenomenon is still unclear but is thought to involve both the IL-23/helper T cell 17 (TH17) axis and dysregulation of IFN-α in the setting of TNF suppression.38
Lifestyle
Obesity is a chronic low-grade inflammatory state that can contribute to the onset of psoriasis or exacerbation of exist
The relationship between psoriasis and alcohol consumption is less clear than it is between psoriasis and obesity or smoking; greater consumption is found in psoriasis patients, but evidence is insufficient to deem alcohol a risk factor.44
Conclusion
Various factors, including genetics, infection, pharmacotherapeutic, and lifestyle, can all contribute to the induction or exacerbation of psoriasis. These factors can provide clues to the pathogenesis of psoriasis as well as help clinicians better counsel patients about their disease.
- Helmick CG, Lee-Han H, Hirsch SC, et al. Prevalence of psoriasis among adults in the U.S.: 2003-2006 and 2009-2010 National Health and Nutrition Examination Surveys. Am J Prev Med. 2014;47:37-45.
- Bowcock AM. The genetics of psoriasis and autoimmunity. Annu Rev Genomics Hum Genet. 2005;6:93-122.
- Swanbeck G, Inerot A, Martinsson T, et al. A population genetic study of psoriasis. Br J Dermatol. 1994;131:32-39.
- Kimberling W, Dobson RL. The inheritance of psoriasis. J Invest Dermatol. 1973;60:538-540.
- Gupta R, Debbaneh MG, Liao W. Genetic epidemiology of psoriasis. Curr Dermatol Rep. 2014;3:61-78.
- Harden JL, Krueger JG, Bowcock AM. The immunogenetics of psoriasis: a comprehensive review. J Autoimmun. 2015;64:66-73.
- Chen L, Tsai TF. HLA-Cw6 and psoriasis. Br J Dermatol. 2018;178:854-862.
- Enerbäck C, Martinsson T, Ineraot A, et al. Evidence that HLA-Cw6 determines early onset of psoriasis, obtained using sequence-specific primers (PCR-SSP). Acta Derm Venereol. 1997;77:273-276.
- Gudjónsson JE, Kárason A, Antonsdóttir EH, et al. HLA-Cw6-positive and HLA-Cw6-negative patients with psoriasis vulgaris have distinct clinical features. J Invest Dermatol. 2002;118:362-365.
- Tomfohrde J, Silverman A, Barnes R, et al. Gene for familial psoriasis susceptibility mapped to distal end of human chromosome 17q. Science. 1994;264:1141-1145.
- Blonska M, Lin X. NF-κB signaling pathways regulated by CARMA family of scaffold proteins. Cell Res. 2011;21:55-70.
- Van Nuffel E, Schmitt A, Afonina IS, et al. CARD14-mediated activation of paracaspase MALT1 in keratinocytes: implications for psoriasis. J Invest Dermatol. 2017;137:569-575.
- Jordan CT, Cao L, Roberson ED, et al. PSORS2 is due to mutations in CARD14. Am J Hum Genet. 2012;90:784-795.
- Wang M, Zhang S, Zheng G, et al. Gain-of-function mutation of Card14 leads to spontaneous psoriasis-like skin inflammation through enhanced keratinocyte response to IL-17A. Immunity. 2018;49:66-79.
- Mellet M, Meier B, Mohanan D, et al. CARD14 gain-of-function mutation alone is sufficient to drive IL-23/IL-17-mediated psoriasiform skin inflammation in vivo. J Invest Dermatol. 2018;138:2010-2023.
- Coto-Segura P, González-Fernández D, Batalla A, et al. Common and rare CARD14 gene variants affect the antitumour necrosis factor response among patients with psoriasis. Br J Dermatol. 2016;175:134-141.
- Winfield JM. Psoriasis as a sequel to acute inflammations of the tonsils: a clinical note. J Cutan Dis. 1916;34:441-443.
- Telfer NR, Chalmers RJG, Whale K, et al. The role of streptococcal infection in the initiation of guttate psoriasis. Arch Dermatol. 1992;128:39-42.
- Hernandez M, Simms-Cendan J, Zendell K. Guttate psoriasis following streptococcal vulvovaginitis in a five-year-old girl. J Pediatr Adolesc Gynecol. 2015;28:e127-e129.
- Herbst RA, Hoch O, Kapp A, et al. Guttate psoriasis triggered by perianal streptococcal dermatitis in a four-year-old boy. J Am Acad Dermatol. 2000;42(5, pt 2):885-887.
- Martin BA, Chalmers RJ, Telfer NR. How great is the risk of further psoriasis following a single episode of acute guttate psoriasis? Arch Dermatol. 1996;132:717-718.
- Thorleifsdottir RH, Eysteinsdóttir, Olafsson JH, et al. Throat infections are associated with exacerbation in a substantial proportion of patients with chronic plaque psoriasis. Acta Derm Venereol. 2016;96:788-791.
- McFadden J, Valdimarsson H, Fry L. Cross-reactivity between streptococcal M surface antigen and human skin. Br J Dermatol. 1991;125:443-447.
- Validmarsson H, Thorleifsdottir RH, Sigurdardottir SL, et al. Psoriasis—as an autoimmune disease caused by molecular mimicry. Trends Immunol. 2009;30:494-501.
- Muto M, Fujikara Y, Hamamoto Y, et al. Immune response to Streptococcus pyogenes and the susceptibility to psoriasis. Australas J Dermatol. 1996;37(suppl 1):S54-S55.
- Weisenseel P, Laumbacher B, Besgen P, et al. Streptococcal infection distinguishes different types of psoriasis. J Med Genet. 2002;39:767-768.
- Rachakonda TD, Dhillon JS, Florek AG, et al. Effect of tonsillectomy on psoriasis: a systematic review. J Am Acad Dermatol. 2015;72:261-275.
- Mallon E, Bunker CB. HIV-associated psoriasis. AIDS Patient Care STDS. 2000;14:239-246.
- Duvic M, Johnson TM, Rapini RP, et al. Acquired immunodeficiency syndrome-associated psoriasis and Reiter’s syndrome. Arch Dermatol. 1987;123:1622-1632.
- Fife DJ, Waller JM, Jeffes EW, et al. Unraveling the paradoxes of HIV-associated psoriasis: a review of T-cell subsets and cytokine profiles. Dermatol Online J. 2007;13:4.
- Ortonne JP, Lebwohl M, Em Griffiths C; Alefacept Clinical Study Group. Alefacept-induced decreases in circulating blood lymphocyte counts correlate with clinical response in patients with chronic plaque psoriasis. Eur J Dermatol. 2003;13:117-123.
- Menon K, Van Voorhees AS, Bebo BF Jr, et al; National Psoriasis Foundation. Psoriasis in patients with HIV infection: from the medical board of the National Psoriasis Foundation. J Am Acad Dermatol. 2010;62:291-299.
- Reddy SP, Shah VV, Wu JJ. Apremilast for a psoriasis patient with HIV and hepatitis C. J Eur Acad Dermatol Venereol. 2017;31:e481-e482.
- Fry L, Baker BS. Triggering psoriasis: the role of infections and medications. Clin Dermatol. 2007;25:606-615.
- Sfikakis PP, Iliopoulos A, Elezoglou A, et al. Psoriasis induced by anti-tumor necrosis factor therapy: a paradoxical adverse reaction. Arthritis Rheum. 2005;52:2513-2518.
- Yeung CK, Chan HH. Cutaneous adverse effects of lithium: epidemiology and management. Am J Clin Dermatol. 2004;5:3-8.
- Hampton PJ, Jans R, Flockhart RJ, et al. Lithium regulates keratinocyte proliferation via glycogen synthase kinase 3 and NFAT 2 (nuclear factor of activated T cells 2). J Cell Physiol. 2012;227:1529-1537.
- Brown G, Wang E, Leon A, et al. Tumor necrosis factor-α inhibitor-induced psoriasis: systematic review of clinical features, histopathological findings, and management experience. J Am Acad Dermatol. 2017;76:334-341.
- Collamer AN, Battafarano DF. Psoriatic skin lesions induced by tumor necrosis factor antagonist therapy: clinical features and possible immunopathogenesis. Semin Arthritis Rheum. 2010;40:233-240.
- Collamer AN, Guerrero KT, Henning JS, et al. Psoriatic skin lesions induced by tumor antagonist therapy: a literature review and potential mechanisms of action. Arthritis Rheum. 2008;59:996-1001.
- Jensen P, Skov L. Psoriasis and obesity. Dermatology. 2016;232:633-639.
- Barrea L, Nappi F, Di Somma C, et al. Environmental risk factors in psoriasis: the point of view of the nutritionist. Int J Environ Res Public Health. 2016;13:743.
- Lee EJ, Han KD, Han JH, et al. Smoking and risk of psoriasis: a nationwide cohort study. J Am Acad Dermatol. 2017;77:573-575.
- Brenaut E, Horreau C, Pouplard C, et al. Alcohol consumption and psoriasis: a systematic literature review. J Eur Acad Dermatol Venereol. 2013;27(suppl 3):30-35.
- Helmick CG, Lee-Han H, Hirsch SC, et al. Prevalence of psoriasis among adults in the U.S.: 2003-2006 and 2009-2010 National Health and Nutrition Examination Surveys. Am J Prev Med. 2014;47:37-45.
- Bowcock AM. The genetics of psoriasis and autoimmunity. Annu Rev Genomics Hum Genet. 2005;6:93-122.
- Swanbeck G, Inerot A, Martinsson T, et al. A population genetic study of psoriasis. Br J Dermatol. 1994;131:32-39.
- Kimberling W, Dobson RL. The inheritance of psoriasis. J Invest Dermatol. 1973;60:538-540.
- Gupta R, Debbaneh MG, Liao W. Genetic epidemiology of psoriasis. Curr Dermatol Rep. 2014;3:61-78.
- Harden JL, Krueger JG, Bowcock AM. The immunogenetics of psoriasis: a comprehensive review. J Autoimmun. 2015;64:66-73.
- Chen L, Tsai TF. HLA-Cw6 and psoriasis. Br J Dermatol. 2018;178:854-862.
- Enerbäck C, Martinsson T, Ineraot A, et al. Evidence that HLA-Cw6 determines early onset of psoriasis, obtained using sequence-specific primers (PCR-SSP). Acta Derm Venereol. 1997;77:273-276.
- Gudjónsson JE, Kárason A, Antonsdóttir EH, et al. HLA-Cw6-positive and HLA-Cw6-negative patients with psoriasis vulgaris have distinct clinical features. J Invest Dermatol. 2002;118:362-365.
- Tomfohrde J, Silverman A, Barnes R, et al. Gene for familial psoriasis susceptibility mapped to distal end of human chromosome 17q. Science. 1994;264:1141-1145.
- Blonska M, Lin X. NF-κB signaling pathways regulated by CARMA family of scaffold proteins. Cell Res. 2011;21:55-70.
- Van Nuffel E, Schmitt A, Afonina IS, et al. CARD14-mediated activation of paracaspase MALT1 in keratinocytes: implications for psoriasis. J Invest Dermatol. 2017;137:569-575.
- Jordan CT, Cao L, Roberson ED, et al. PSORS2 is due to mutations in CARD14. Am J Hum Genet. 2012;90:784-795.
- Wang M, Zhang S, Zheng G, et al. Gain-of-function mutation of Card14 leads to spontaneous psoriasis-like skin inflammation through enhanced keratinocyte response to IL-17A. Immunity. 2018;49:66-79.
- Mellet M, Meier B, Mohanan D, et al. CARD14 gain-of-function mutation alone is sufficient to drive IL-23/IL-17-mediated psoriasiform skin inflammation in vivo. J Invest Dermatol. 2018;138:2010-2023.
- Coto-Segura P, González-Fernández D, Batalla A, et al. Common and rare CARD14 gene variants affect the antitumour necrosis factor response among patients with psoriasis. Br J Dermatol. 2016;175:134-141.
- Winfield JM. Psoriasis as a sequel to acute inflammations of the tonsils: a clinical note. J Cutan Dis. 1916;34:441-443.
- Telfer NR, Chalmers RJG, Whale K, et al. The role of streptococcal infection in the initiation of guttate psoriasis. Arch Dermatol. 1992;128:39-42.
- Hernandez M, Simms-Cendan J, Zendell K. Guttate psoriasis following streptococcal vulvovaginitis in a five-year-old girl. J Pediatr Adolesc Gynecol. 2015;28:e127-e129.
- Herbst RA, Hoch O, Kapp A, et al. Guttate psoriasis triggered by perianal streptococcal dermatitis in a four-year-old boy. J Am Acad Dermatol. 2000;42(5, pt 2):885-887.
- Martin BA, Chalmers RJ, Telfer NR. How great is the risk of further psoriasis following a single episode of acute guttate psoriasis? Arch Dermatol. 1996;132:717-718.
- Thorleifsdottir RH, Eysteinsdóttir, Olafsson JH, et al. Throat infections are associated with exacerbation in a substantial proportion of patients with chronic plaque psoriasis. Acta Derm Venereol. 2016;96:788-791.
- McFadden J, Valdimarsson H, Fry L. Cross-reactivity between streptococcal M surface antigen and human skin. Br J Dermatol. 1991;125:443-447.
- Validmarsson H, Thorleifsdottir RH, Sigurdardottir SL, et al. Psoriasis—as an autoimmune disease caused by molecular mimicry. Trends Immunol. 2009;30:494-501.
- Muto M, Fujikara Y, Hamamoto Y, et al. Immune response to Streptococcus pyogenes and the susceptibility to psoriasis. Australas J Dermatol. 1996;37(suppl 1):S54-S55.
- Weisenseel P, Laumbacher B, Besgen P, et al. Streptococcal infection distinguishes different types of psoriasis. J Med Genet. 2002;39:767-768.
- Rachakonda TD, Dhillon JS, Florek AG, et al. Effect of tonsillectomy on psoriasis: a systematic review. J Am Acad Dermatol. 2015;72:261-275.
- Mallon E, Bunker CB. HIV-associated psoriasis. AIDS Patient Care STDS. 2000;14:239-246.
- Duvic M, Johnson TM, Rapini RP, et al. Acquired immunodeficiency syndrome-associated psoriasis and Reiter’s syndrome. Arch Dermatol. 1987;123:1622-1632.
- Fife DJ, Waller JM, Jeffes EW, et al. Unraveling the paradoxes of HIV-associated psoriasis: a review of T-cell subsets and cytokine profiles. Dermatol Online J. 2007;13:4.
- Ortonne JP, Lebwohl M, Em Griffiths C; Alefacept Clinical Study Group. Alefacept-induced decreases in circulating blood lymphocyte counts correlate with clinical response in patients with chronic plaque psoriasis. Eur J Dermatol. 2003;13:117-123.
- Menon K, Van Voorhees AS, Bebo BF Jr, et al; National Psoriasis Foundation. Psoriasis in patients with HIV infection: from the medical board of the National Psoriasis Foundation. J Am Acad Dermatol. 2010;62:291-299.
- Reddy SP, Shah VV, Wu JJ. Apremilast for a psoriasis patient with HIV and hepatitis C. J Eur Acad Dermatol Venereol. 2017;31:e481-e482.
- Fry L, Baker BS. Triggering psoriasis: the role of infections and medications. Clin Dermatol. 2007;25:606-615.
- Sfikakis PP, Iliopoulos A, Elezoglou A, et al. Psoriasis induced by anti-tumor necrosis factor therapy: a paradoxical adverse reaction. Arthritis Rheum. 2005;52:2513-2518.
- Yeung CK, Chan HH. Cutaneous adverse effects of lithium: epidemiology and management. Am J Clin Dermatol. 2004;5:3-8.
- Hampton PJ, Jans R, Flockhart RJ, et al. Lithium regulates keratinocyte proliferation via glycogen synthase kinase 3 and NFAT 2 (nuclear factor of activated T cells 2). J Cell Physiol. 2012;227:1529-1537.
- Brown G, Wang E, Leon A, et al. Tumor necrosis factor-α inhibitor-induced psoriasis: systematic review of clinical features, histopathological findings, and management experience. J Am Acad Dermatol. 2017;76:334-341.
- Collamer AN, Battafarano DF. Psoriatic skin lesions induced by tumor necrosis factor antagonist therapy: clinical features and possible immunopathogenesis. Semin Arthritis Rheum. 2010;40:233-240.
- Collamer AN, Guerrero KT, Henning JS, et al. Psoriatic skin lesions induced by tumor antagonist therapy: a literature review and potential mechanisms of action. Arthritis Rheum. 2008;59:996-1001.
- Jensen P, Skov L. Psoriasis and obesity. Dermatology. 2016;232:633-639.
- Barrea L, Nappi F, Di Somma C, et al. Environmental risk factors in psoriasis: the point of view of the nutritionist. Int J Environ Res Public Health. 2016;13:743.
- Lee EJ, Han KD, Han JH, et al. Smoking and risk of psoriasis: a nationwide cohort study. J Am Acad Dermatol. 2017;77:573-575.
- Brenaut E, Horreau C, Pouplard C, et al. Alcohol consumption and psoriasis: a systematic literature review. J Eur Acad Dermatol Venereol. 2013;27(suppl 3):30-35.
Practice Points
- HLA-Cw6 and CARD14 are genetic factors associated with psoriasis.
- Psoriasis in the setting of human immunodeficiency virus infection may be treated with topical steroids, phototherapy, systemic retinoids, or apremilast.
- Psoriasis is a potential adverse effect in patients taking lithium or tumor necrosis factor inhibitors.
- Patients should be counseled about the role of obesity and smoking on psoriasis.







