User login
Composite screening measures advocated for asymptomatic PAH
In patients at risk for pulmonary arterial hypertension (PAH) due to a connective tissue disease, composite novel screening methods are improving early detection when employed in the context of traditional tools, such as transthoracic echocardiography (TTE), according to a systematic review of studies published over the last 5 years.
The review was conducted to prepare for a guideline update, according to the authors of this recently published summary in Seminars in Arthritis and Rheumatism.
In a literature review for 2012-2015, the authors evaluated whether new tools or strategies have improved PAH screening in patients with connective tissue disease since the last review was undertaken (Semin Arthritis Rheum 2014;43:536-41).
The latest review found that although TTE and pulmonary function tests (PFT) remain a mainstay of screening, there is growing evidence that composite measures, such as the DETECT and ASIG algorithms, add sensitivity and specificity, compared with guidelines that rely on TTE and PFT alone.
After a literature search, the systematic review included 16 cohort studies and 6 case-control studies. Most of these evaluated PAH screening strategies for patients with systemic sclerosis specifically despite the potential for other connective tissue disease etiologies to lead to PAH.
“We need more longitudinal observational studies to develop and validate screening algorithms for non–systemic sclerosis connective tissue diseases,” stated the authors, led by senior investigator Dinesh Khanna, MD, medical director of ambulatory and chronic disease in the University of Michigan’s Office of Research, Ann Arbor.
Relative to screening primarily based on TTE and PFT as advocated in 2009 joint guidelines from the European Society of Cardiology and the European Respiratory Society (ESC/ERS), the preponderance of data supported the addition of DIRECT and ASIG algorithms to improve the sensitivity and specificity of traditional screening and diagnostic tools, according to the data reviewed.
Several of the studies evaluating DETECT and ASIG compared their sensitivities and specificities to the screening strategy recommended in the 2009 ESC/ERS guidelines because the 2015 ESC/ERS guidelines were not yet available at the time these studies were taking place.
In fact, the advantage of these algorithms has been acknowledged in a set of subsequent ESC/ERS guidelines issued in 2015. These specifically recommend DETECT in selected populations, such as adults with more than a 3-year history of systemic sclerosis and less than 60% diffusing capacity of the lung for carbon monoxide.
Based on the systematic review, the authors did not conclude that there is a single set of optimal tests for PAH screening whether in patients with systemic sclerosis or another connective tissue disease, but the authors did conclude that there has been progress in strategies for early PAH detection.
They also speculated that more progress might be coming. They noted that several newer tests, such as stress TTE to assess cardiopulmonary function during exercise, might further improve PAH detection at early stages.
According to the authors, this work is important, because better screening that results in earlier PAH detection means earlier treatment, which, in turn, “may improve survival.”
SOURCE: Young A et al. Semin Arthritis Rheum. 2018; Oct 14. doi: 10.1016/j.semarthrit.2018.10.010.
In patients at risk for pulmonary arterial hypertension (PAH) due to a connective tissue disease, composite novel screening methods are improving early detection when employed in the context of traditional tools, such as transthoracic echocardiography (TTE), according to a systematic review of studies published over the last 5 years.
The review was conducted to prepare for a guideline update, according to the authors of this recently published summary in Seminars in Arthritis and Rheumatism.
In a literature review for 2012-2015, the authors evaluated whether new tools or strategies have improved PAH screening in patients with connective tissue disease since the last review was undertaken (Semin Arthritis Rheum 2014;43:536-41).
The latest review found that although TTE and pulmonary function tests (PFT) remain a mainstay of screening, there is growing evidence that composite measures, such as the DETECT and ASIG algorithms, add sensitivity and specificity, compared with guidelines that rely on TTE and PFT alone.
After a literature search, the systematic review included 16 cohort studies and 6 case-control studies. Most of these evaluated PAH screening strategies for patients with systemic sclerosis specifically despite the potential for other connective tissue disease etiologies to lead to PAH.
“We need more longitudinal observational studies to develop and validate screening algorithms for non–systemic sclerosis connective tissue diseases,” stated the authors, led by senior investigator Dinesh Khanna, MD, medical director of ambulatory and chronic disease in the University of Michigan’s Office of Research, Ann Arbor.
Relative to screening primarily based on TTE and PFT as advocated in 2009 joint guidelines from the European Society of Cardiology and the European Respiratory Society (ESC/ERS), the preponderance of data supported the addition of DIRECT and ASIG algorithms to improve the sensitivity and specificity of traditional screening and diagnostic tools, according to the data reviewed.
Several of the studies evaluating DETECT and ASIG compared their sensitivities and specificities to the screening strategy recommended in the 2009 ESC/ERS guidelines because the 2015 ESC/ERS guidelines were not yet available at the time these studies were taking place.
In fact, the advantage of these algorithms has been acknowledged in a set of subsequent ESC/ERS guidelines issued in 2015. These specifically recommend DETECT in selected populations, such as adults with more than a 3-year history of systemic sclerosis and less than 60% diffusing capacity of the lung for carbon monoxide.
Based on the systematic review, the authors did not conclude that there is a single set of optimal tests for PAH screening whether in patients with systemic sclerosis or another connective tissue disease, but the authors did conclude that there has been progress in strategies for early PAH detection.
They also speculated that more progress might be coming. They noted that several newer tests, such as stress TTE to assess cardiopulmonary function during exercise, might further improve PAH detection at early stages.
According to the authors, this work is important, because better screening that results in earlier PAH detection means earlier treatment, which, in turn, “may improve survival.”
SOURCE: Young A et al. Semin Arthritis Rheum. 2018; Oct 14. doi: 10.1016/j.semarthrit.2018.10.010.
In patients at risk for pulmonary arterial hypertension (PAH) due to a connective tissue disease, composite novel screening methods are improving early detection when employed in the context of traditional tools, such as transthoracic echocardiography (TTE), according to a systematic review of studies published over the last 5 years.
The review was conducted to prepare for a guideline update, according to the authors of this recently published summary in Seminars in Arthritis and Rheumatism.
In a literature review for 2012-2015, the authors evaluated whether new tools or strategies have improved PAH screening in patients with connective tissue disease since the last review was undertaken (Semin Arthritis Rheum 2014;43:536-41).
The latest review found that although TTE and pulmonary function tests (PFT) remain a mainstay of screening, there is growing evidence that composite measures, such as the DETECT and ASIG algorithms, add sensitivity and specificity, compared with guidelines that rely on TTE and PFT alone.
After a literature search, the systematic review included 16 cohort studies and 6 case-control studies. Most of these evaluated PAH screening strategies for patients with systemic sclerosis specifically despite the potential for other connective tissue disease etiologies to lead to PAH.
“We need more longitudinal observational studies to develop and validate screening algorithms for non–systemic sclerosis connective tissue diseases,” stated the authors, led by senior investigator Dinesh Khanna, MD, medical director of ambulatory and chronic disease in the University of Michigan’s Office of Research, Ann Arbor.
Relative to screening primarily based on TTE and PFT as advocated in 2009 joint guidelines from the European Society of Cardiology and the European Respiratory Society (ESC/ERS), the preponderance of data supported the addition of DIRECT and ASIG algorithms to improve the sensitivity and specificity of traditional screening and diagnostic tools, according to the data reviewed.
Several of the studies evaluating DETECT and ASIG compared their sensitivities and specificities to the screening strategy recommended in the 2009 ESC/ERS guidelines because the 2015 ESC/ERS guidelines were not yet available at the time these studies were taking place.
In fact, the advantage of these algorithms has been acknowledged in a set of subsequent ESC/ERS guidelines issued in 2015. These specifically recommend DETECT in selected populations, such as adults with more than a 3-year history of systemic sclerosis and less than 60% diffusing capacity of the lung for carbon monoxide.
Based on the systematic review, the authors did not conclude that there is a single set of optimal tests for PAH screening whether in patients with systemic sclerosis or another connective tissue disease, but the authors did conclude that there has been progress in strategies for early PAH detection.
They also speculated that more progress might be coming. They noted that several newer tests, such as stress TTE to assess cardiopulmonary function during exercise, might further improve PAH detection at early stages.
According to the authors, this work is important, because better screening that results in earlier PAH detection means earlier treatment, which, in turn, “may improve survival.”
SOURCE: Young A et al. Semin Arthritis Rheum. 2018; Oct 14. doi: 10.1016/j.semarthrit.2018.10.010.
FROM SEMINARS IN ARTHRITIS AND RHEUMATISM
Key clinical point: Strategies for early detection of pulmonary arterial hypertension (PAH) are improving with new tools.
Major finding: Screening algorithms are improving sensitivity and specificity for PAH in patients with connective tissue diseases.
Study details: Systematic literature review.
Disclosures: Dr. Khanna has financial relationships with many companies that produce drugs for PAH but no conflicts relative to this study.
Source: Young A et al. Semin Arthritis Rheum. 2018 Oct 14. doi. 10.1016/j.semarthrit.2018.10.010.
In Medicare population, carotid revascularization has declined
NEW YORK – The rates of carotid artery revascularization with either endarterectomy or stenting declined precipitously over a recent 15-year period, at least among Medicare fee-for-service beneficiaries, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
A reduction in carotid endarterectomies (CEA) largely accounted for the decline during 1999-2014 although there was a cumulative decline in all carotid revascularization procedures when rates of CEA and stenting were combined, according to Brajesh K. Lal, MD, professor of surgery, University of Maryland Medical System, Baltimore.
In 1999, when enthusiasm for CEA appears to have peaked, 81,306 patients received this procedure, but a steady decline was observed until 2014, when 36,325 patients were being treated annually in the Medicare database. When calculated as endarterectomies per 100,000 beneficiaries, the rate declined from 298 to 128 (57%; P less than .001) over this 15-year period.
The number of stenting procedures had not reached its peak in 1999, when 10,416 were performed. Rather, the number performed annually nearly doubled to, 22.865 by 2006. However, it then began to decline and reached 10,208 by 2014, which was slightly fewer than in 1999, according to Dr. Lal.
These trends have been observed even though outcomes are getting better, at least for CEA, according to Dr. Lal. From the same pool of data, there was a 31% (1.1% vs. 1.6%) reduction from 1999 to 2014 in mortality at 30 days following CEA. For a composite of ischemic stroke and all-cause mortality, the rate fell 29.5% (3.1% vs. 4.4%). Both reductions were called statistically significant by Dr. Lal.
The improvements in CEA outcomes were observed even though “the treated patients got sicker when looking at comorbidities and risk factors, particularly hypertension, renal insufficiency, and diabetes,” Dr. Lal said.
Outcomes also improved among patients undergoing carotid stenting in general, although the patterns were described as “more complex.” In general, there was steady improvement on outcomes during 1999-2006, but there was no further gain and some lost ground during 2006-2014. For example, ischemic stroke or death fell from 7.0% in 1999 to 4.8% in 2006, but it had climbed back to 7.0% by 2014 with no net change when the first and last year were compared.
However, with risk adjustment, there was a reduction in in-hospital mortality (1.13% vs. 2.78%) over the study period for patients undergoing carotid stenting, according to Dr. Lal, who said this reached statistical significance. Like the CEA group, there was more comorbidity among those treated with stenting at the end, relative to the early part of the study period.
In the stenting group, patients with symptomatic carotid disease rose from 14.4% in 1999 to 25.9% in 2014. This tracks with Medicare policy, which required patients after 2005 to have symptomatic disease for reimbursement, according to Dr. Lal. Prior to 2005, reimbursement was granted for patients participating in clinical trials only.
The rates of carotid revascularization are not evenly distributed geographically in the United States, according to the Medicare data. Endarterectomy in particular has been more common in the south and Midwest than on either coast. This was true in 1999 and remained so in 2014. The distribution was similar for stenting, although it was also relatively common in the southwest in the early part of the study period.
In the beginning of the study, the increased rate of stenting might have contributed to the decline in endarterectomy, but there are several other factors that are implicated in the observed trends, according to Dr. Lal. He suggested that decreasing reimbursement for the performance of these procedures, better clinical management of risk factors, and advances in medical therapy. He cited a physician survey that showed a growing preference for medical management over invasive procedures in patients with high-grade stenosis and indicated that this last factor might be a particularly important driver of the decline in revascularization referrals for asymptomatic carotid disease.
The degree to which these Medicare data are representative of overall trends in the United States is unclear, but Dr. Lal called for further work to understand the forces that these data suggest are driving the changing patterns of carotid revascularization.
NEW YORK – The rates of carotid artery revascularization with either endarterectomy or stenting declined precipitously over a recent 15-year period, at least among Medicare fee-for-service beneficiaries, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
A reduction in carotid endarterectomies (CEA) largely accounted for the decline during 1999-2014 although there was a cumulative decline in all carotid revascularization procedures when rates of CEA and stenting were combined, according to Brajesh K. Lal, MD, professor of surgery, University of Maryland Medical System, Baltimore.
In 1999, when enthusiasm for CEA appears to have peaked, 81,306 patients received this procedure, but a steady decline was observed until 2014, when 36,325 patients were being treated annually in the Medicare database. When calculated as endarterectomies per 100,000 beneficiaries, the rate declined from 298 to 128 (57%; P less than .001) over this 15-year period.
The number of stenting procedures had not reached its peak in 1999, when 10,416 were performed. Rather, the number performed annually nearly doubled to, 22.865 by 2006. However, it then began to decline and reached 10,208 by 2014, which was slightly fewer than in 1999, according to Dr. Lal.
These trends have been observed even though outcomes are getting better, at least for CEA, according to Dr. Lal. From the same pool of data, there was a 31% (1.1% vs. 1.6%) reduction from 1999 to 2014 in mortality at 30 days following CEA. For a composite of ischemic stroke and all-cause mortality, the rate fell 29.5% (3.1% vs. 4.4%). Both reductions were called statistically significant by Dr. Lal.
The improvements in CEA outcomes were observed even though “the treated patients got sicker when looking at comorbidities and risk factors, particularly hypertension, renal insufficiency, and diabetes,” Dr. Lal said.
Outcomes also improved among patients undergoing carotid stenting in general, although the patterns were described as “more complex.” In general, there was steady improvement on outcomes during 1999-2006, but there was no further gain and some lost ground during 2006-2014. For example, ischemic stroke or death fell from 7.0% in 1999 to 4.8% in 2006, but it had climbed back to 7.0% by 2014 with no net change when the first and last year were compared.
However, with risk adjustment, there was a reduction in in-hospital mortality (1.13% vs. 2.78%) over the study period for patients undergoing carotid stenting, according to Dr. Lal, who said this reached statistical significance. Like the CEA group, there was more comorbidity among those treated with stenting at the end, relative to the early part of the study period.
In the stenting group, patients with symptomatic carotid disease rose from 14.4% in 1999 to 25.9% in 2014. This tracks with Medicare policy, which required patients after 2005 to have symptomatic disease for reimbursement, according to Dr. Lal. Prior to 2005, reimbursement was granted for patients participating in clinical trials only.
The rates of carotid revascularization are not evenly distributed geographically in the United States, according to the Medicare data. Endarterectomy in particular has been more common in the south and Midwest than on either coast. This was true in 1999 and remained so in 2014. The distribution was similar for stenting, although it was also relatively common in the southwest in the early part of the study period.
In the beginning of the study, the increased rate of stenting might have contributed to the decline in endarterectomy, but there are several other factors that are implicated in the observed trends, according to Dr. Lal. He suggested that decreasing reimbursement for the performance of these procedures, better clinical management of risk factors, and advances in medical therapy. He cited a physician survey that showed a growing preference for medical management over invasive procedures in patients with high-grade stenosis and indicated that this last factor might be a particularly important driver of the decline in revascularization referrals for asymptomatic carotid disease.
The degree to which these Medicare data are representative of overall trends in the United States is unclear, but Dr. Lal called for further work to understand the forces that these data suggest are driving the changing patterns of carotid revascularization.
NEW YORK – The rates of carotid artery revascularization with either endarterectomy or stenting declined precipitously over a recent 15-year period, at least among Medicare fee-for-service beneficiaries, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
A reduction in carotid endarterectomies (CEA) largely accounted for the decline during 1999-2014 although there was a cumulative decline in all carotid revascularization procedures when rates of CEA and stenting were combined, according to Brajesh K. Lal, MD, professor of surgery, University of Maryland Medical System, Baltimore.
In 1999, when enthusiasm for CEA appears to have peaked, 81,306 patients received this procedure, but a steady decline was observed until 2014, when 36,325 patients were being treated annually in the Medicare database. When calculated as endarterectomies per 100,000 beneficiaries, the rate declined from 298 to 128 (57%; P less than .001) over this 15-year period.
The number of stenting procedures had not reached its peak in 1999, when 10,416 were performed. Rather, the number performed annually nearly doubled to, 22.865 by 2006. However, it then began to decline and reached 10,208 by 2014, which was slightly fewer than in 1999, according to Dr. Lal.
These trends have been observed even though outcomes are getting better, at least for CEA, according to Dr. Lal. From the same pool of data, there was a 31% (1.1% vs. 1.6%) reduction from 1999 to 2014 in mortality at 30 days following CEA. For a composite of ischemic stroke and all-cause mortality, the rate fell 29.5% (3.1% vs. 4.4%). Both reductions were called statistically significant by Dr. Lal.
The improvements in CEA outcomes were observed even though “the treated patients got sicker when looking at comorbidities and risk factors, particularly hypertension, renal insufficiency, and diabetes,” Dr. Lal said.
Outcomes also improved among patients undergoing carotid stenting in general, although the patterns were described as “more complex.” In general, there was steady improvement on outcomes during 1999-2006, but there was no further gain and some lost ground during 2006-2014. For example, ischemic stroke or death fell from 7.0% in 1999 to 4.8% in 2006, but it had climbed back to 7.0% by 2014 with no net change when the first and last year were compared.
However, with risk adjustment, there was a reduction in in-hospital mortality (1.13% vs. 2.78%) over the study period for patients undergoing carotid stenting, according to Dr. Lal, who said this reached statistical significance. Like the CEA group, there was more comorbidity among those treated with stenting at the end, relative to the early part of the study period.
In the stenting group, patients with symptomatic carotid disease rose from 14.4% in 1999 to 25.9% in 2014. This tracks with Medicare policy, which required patients after 2005 to have symptomatic disease for reimbursement, according to Dr. Lal. Prior to 2005, reimbursement was granted for patients participating in clinical trials only.
The rates of carotid revascularization are not evenly distributed geographically in the United States, according to the Medicare data. Endarterectomy in particular has been more common in the south and Midwest than on either coast. This was true in 1999 and remained so in 2014. The distribution was similar for stenting, although it was also relatively common in the southwest in the early part of the study period.
In the beginning of the study, the increased rate of stenting might have contributed to the decline in endarterectomy, but there are several other factors that are implicated in the observed trends, according to Dr. Lal. He suggested that decreasing reimbursement for the performance of these procedures, better clinical management of risk factors, and advances in medical therapy. He cited a physician survey that showed a growing preference for medical management over invasive procedures in patients with high-grade stenosis and indicated that this last factor might be a particularly important driver of the decline in revascularization referrals for asymptomatic carotid disease.
The degree to which these Medicare data are representative of overall trends in the United States is unclear, but Dr. Lal called for further work to understand the forces that these data suggest are driving the changing patterns of carotid revascularization.
REPORTING FROM VEITHSYMPOSIUM
Key clinical point:
Major finding: During 1999-2014, the rate of carotid endarterectomy per 100,000 beneficiaries fell from 291 to 128 (57%; P less than .001).
Study details: Retrospective database review.
Disclosures: Dr. Lal reported having no financial conflicts relevant to the study.
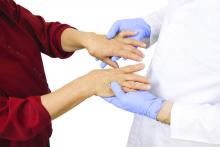
Tender joint count may confound assessment of RA inflammation
In RA, patients with predominantly tender joint counts were found to have lower levels of inflammation defined by ultrasound than those with predominantly swollen joints, according to an observational study that has implications for management decisions.
When patients do not achieve a remission defined by a composite disease activity score (CDAS) that includes tender joint counts, it suggests that inflammation persists, but this may be a “misinterpretation,” according to a report in Arthritis Care & Research by Hilde B. Hammer, MD, PhD, of the department of rheumatology at Diakonhjemmet Hospital, Oslo, and her associates.
In this observational study, 209 RA patients were evaluated at baseline and again at months 1, 2, 3, 6, and 12. The researchers compared 84 patients with predominantly tender joints, defined as tender-swollen joint count difference (TSJD) score of greater than zero, against 125 patients with predominantly swollen joints, defined as TSJD of zero or less.
Scores on specific CDAS measures, such as the Disease Activity Score based on 28 joints, the Clinical Disease Activity Index, and the Simplified Disease Activity Index, were significantly higher (P less than .001) at all visits in patients with predominantly tender joints, compared with those with predominantly swollen joints. Although laboratory markers, such as C-reactive protein and rheumatoid factor, were similar between groups, synovitis as scored with ultrasound assessment was significantly lower (P less than .001) among patients with predominantly tender joints than in those with predominantly swollen joints.
This disparity is important, according to the authors, who suggested that failure to reach a CDAS-defined remission solely on the basis of tender joints might complicate clinical assessment.
“Taking the high impact of CDAS levels in the treat-to-target management of RA into account, our results suggest a careful interpretation of CDAS levels in patients who have predominantly tender joints,” the authors reported.
Based on the evidence from this study, which found tender joints to be a positive predictor of CDAS and patient-reported outcomes but a negative predictor of ultrasound scoring of inflammation, “these results indicate that tender joints do not reflect the same pathology as found with ultrasound,” they wrote.
The research was supported by AbbVie, Pfizer, and Roche in the form of study grants awarded to the department of rheumatology at Diakonhjemmet Hospital via Dr. Hammer and two other authors. Dr. Hammer and the two other authors also reported financial relationships with those companies and others.
SOURCE: Hammer HB et al. Arthritis Care Res. 2018 Nov 26. doi: 10.1002/acr.23815.
In RA, patients with predominantly tender joint counts were found to have lower levels of inflammation defined by ultrasound than those with predominantly swollen joints, according to an observational study that has implications for management decisions.
When patients do not achieve a remission defined by a composite disease activity score (CDAS) that includes tender joint counts, it suggests that inflammation persists, but this may be a “misinterpretation,” according to a report in Arthritis Care & Research by Hilde B. Hammer, MD, PhD, of the department of rheumatology at Diakonhjemmet Hospital, Oslo, and her associates.
In this observational study, 209 RA patients were evaluated at baseline and again at months 1, 2, 3, 6, and 12. The researchers compared 84 patients with predominantly tender joints, defined as tender-swollen joint count difference (TSJD) score of greater than zero, against 125 patients with predominantly swollen joints, defined as TSJD of zero or less.
Scores on specific CDAS measures, such as the Disease Activity Score based on 28 joints, the Clinical Disease Activity Index, and the Simplified Disease Activity Index, were significantly higher (P less than .001) at all visits in patients with predominantly tender joints, compared with those with predominantly swollen joints. Although laboratory markers, such as C-reactive protein and rheumatoid factor, were similar between groups, synovitis as scored with ultrasound assessment was significantly lower (P less than .001) among patients with predominantly tender joints than in those with predominantly swollen joints.
This disparity is important, according to the authors, who suggested that failure to reach a CDAS-defined remission solely on the basis of tender joints might complicate clinical assessment.
“Taking the high impact of CDAS levels in the treat-to-target management of RA into account, our results suggest a careful interpretation of CDAS levels in patients who have predominantly tender joints,” the authors reported.
Based on the evidence from this study, which found tender joints to be a positive predictor of CDAS and patient-reported outcomes but a negative predictor of ultrasound scoring of inflammation, “these results indicate that tender joints do not reflect the same pathology as found with ultrasound,” they wrote.
The research was supported by AbbVie, Pfizer, and Roche in the form of study grants awarded to the department of rheumatology at Diakonhjemmet Hospital via Dr. Hammer and two other authors. Dr. Hammer and the two other authors also reported financial relationships with those companies and others.
SOURCE: Hammer HB et al. Arthritis Care Res. 2018 Nov 26. doi: 10.1002/acr.23815.
In RA, patients with predominantly tender joint counts were found to have lower levels of inflammation defined by ultrasound than those with predominantly swollen joints, according to an observational study that has implications for management decisions.
When patients do not achieve a remission defined by a composite disease activity score (CDAS) that includes tender joint counts, it suggests that inflammation persists, but this may be a “misinterpretation,” according to a report in Arthritis Care & Research by Hilde B. Hammer, MD, PhD, of the department of rheumatology at Diakonhjemmet Hospital, Oslo, and her associates.
In this observational study, 209 RA patients were evaluated at baseline and again at months 1, 2, 3, 6, and 12. The researchers compared 84 patients with predominantly tender joints, defined as tender-swollen joint count difference (TSJD) score of greater than zero, against 125 patients with predominantly swollen joints, defined as TSJD of zero or less.
Scores on specific CDAS measures, such as the Disease Activity Score based on 28 joints, the Clinical Disease Activity Index, and the Simplified Disease Activity Index, were significantly higher (P less than .001) at all visits in patients with predominantly tender joints, compared with those with predominantly swollen joints. Although laboratory markers, such as C-reactive protein and rheumatoid factor, were similar between groups, synovitis as scored with ultrasound assessment was significantly lower (P less than .001) among patients with predominantly tender joints than in those with predominantly swollen joints.
This disparity is important, according to the authors, who suggested that failure to reach a CDAS-defined remission solely on the basis of tender joints might complicate clinical assessment.
“Taking the high impact of CDAS levels in the treat-to-target management of RA into account, our results suggest a careful interpretation of CDAS levels in patients who have predominantly tender joints,” the authors reported.
Based on the evidence from this study, which found tender joints to be a positive predictor of CDAS and patient-reported outcomes but a negative predictor of ultrasound scoring of inflammation, “these results indicate that tender joints do not reflect the same pathology as found with ultrasound,” they wrote.
The research was supported by AbbVie, Pfizer, and Roche in the form of study grants awarded to the department of rheumatology at Diakonhjemmet Hospital via Dr. Hammer and two other authors. Dr. Hammer and the two other authors also reported financial relationships with those companies and others.
SOURCE: Hammer HB et al. Arthritis Care Res. 2018 Nov 26. doi: 10.1002/acr.23815.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point:
Major finding: Baseline tender joint count correlates with patient-reported outcomes (P less than .001) but not with ultrasound-assessed inflammation.
Study details: An observational study of 209 RA patients.
Disclosures: The research was supported by AbbVie, Pfizer, and Roche in the form of study grants awarded to the department of rheumatology at Diakonhjemmet Hospital via Dr. Hammer and two other authors. Dr. Hammer and the two other authors also reported financial relationships with those companies and others.
Source: Hammer HB et al. Arthritis Care Res. 2018 Nov 26. doi: 10.1002/acr.23815.
Redo carotid endarterectomy is more risky than previously estimated
NEW YORK – It is well known that reoperative carotid endarterectomy can be technically challenging because of the scarring left from the initial procedure, but an analysis of a large database presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation also revealed that the risk of complications, particularly stroke, is greater.
When “redo” carotid endarterectomies were compared with the index primary procedure collected in the American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP) database, the odds ratio for stroke was several times greater (odds ratio, 3.71; P = .002) on univariate analysis, reported Jeffrey J. Siracuse, MD, associate professor of surgery and radiology at Boston University.
Previous single-center reports of redo endarterectomies “showed terrific results, really no perioperative stroke or morbidity, but this is older data from a different era,” said Dr. Siracuse, who undertook this study to determine whether “real-world” data would tell a different story.
In this study, 75,943 primary carotid endarterectomies and 140 redo procedures were identified in the ACS NSQIP database and compared. The redo population had a significantly higher incidence of end-stage renal disease (3.6% vs. 1.1%; P = .004), but history of stroke, whether with deficit (20.8% vs. 15.4%) or without (11.5% vs. 9.1%), was numerically higher among those undergoing a primary procedure even though these differences did not reach statistical significance. Baseline demographics and comorbidities were otherwise similar.
Presumably because of the difficulty of recanalizing scarred tissue, the mean procedure time for redos was longer than that for the primary procedures (137 vs. 49 minutes; P less than .001), but there were no significant differences in the rate of surgical site infections (0.7% vs. 0.3%; P = .482), return to the operating room (3.6% vs. 4%; P = .853), or 30-day readmissions (2.1% vs. 6.9%; P = .810) for the redo and index procedures, respectively.
Although perioperative MI rates were higher in the redo group (2.1%) than in the primary endarterectomy group (0.9%), this difference did not reach statistical significance (P = .125). However, a multivariate analysis associated redo carotid endarterectomy procedures with a nearly threefold increase in risk of a composite of major adverse cardiovascular events when compared on a multivariate analysis (OR, 2.76; P = .007), Dr. Siracuse reported.
For the surgeons considering a redo carotid endarterectomy, these data “inform a risk-benefit analysis,” according to Dr. Siracuse, but he also said that redo procedures still should be considered a viable strategy when considered in the context of other options.
Presenting a case he performed just prior to the VEITHsymposium, Dr. Siracuse displayed CT images that showed internal and common carotids with more than 75% stenosis in an 80-year-old women 7 years after a primary carotid endarterectomy. The tight stenoses and the evidence of substantial intra-arterial debris were concerns, but a decision to perform a redo endarterectomy was reached after other options, including stenting, were considered.
“She did great. She went home and has had no more symptoms,” Dr. Siracuse reported. “The point is you still have to take these [potential redo endarterectomies] on a case-by case basis.”
Dr. Siracuse reported he had no financial relationships relevant to this study.
NEW YORK – It is well known that reoperative carotid endarterectomy can be technically challenging because of the scarring left from the initial procedure, but an analysis of a large database presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation also revealed that the risk of complications, particularly stroke, is greater.
When “redo” carotid endarterectomies were compared with the index primary procedure collected in the American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP) database, the odds ratio for stroke was several times greater (odds ratio, 3.71; P = .002) on univariate analysis, reported Jeffrey J. Siracuse, MD, associate professor of surgery and radiology at Boston University.
Previous single-center reports of redo endarterectomies “showed terrific results, really no perioperative stroke or morbidity, but this is older data from a different era,” said Dr. Siracuse, who undertook this study to determine whether “real-world” data would tell a different story.
In this study, 75,943 primary carotid endarterectomies and 140 redo procedures were identified in the ACS NSQIP database and compared. The redo population had a significantly higher incidence of end-stage renal disease (3.6% vs. 1.1%; P = .004), but history of stroke, whether with deficit (20.8% vs. 15.4%) or without (11.5% vs. 9.1%), was numerically higher among those undergoing a primary procedure even though these differences did not reach statistical significance. Baseline demographics and comorbidities were otherwise similar.
Presumably because of the difficulty of recanalizing scarred tissue, the mean procedure time for redos was longer than that for the primary procedures (137 vs. 49 minutes; P less than .001), but there were no significant differences in the rate of surgical site infections (0.7% vs. 0.3%; P = .482), return to the operating room (3.6% vs. 4%; P = .853), or 30-day readmissions (2.1% vs. 6.9%; P = .810) for the redo and index procedures, respectively.
Although perioperative MI rates were higher in the redo group (2.1%) than in the primary endarterectomy group (0.9%), this difference did not reach statistical significance (P = .125). However, a multivariate analysis associated redo carotid endarterectomy procedures with a nearly threefold increase in risk of a composite of major adverse cardiovascular events when compared on a multivariate analysis (OR, 2.76; P = .007), Dr. Siracuse reported.
For the surgeons considering a redo carotid endarterectomy, these data “inform a risk-benefit analysis,” according to Dr. Siracuse, but he also said that redo procedures still should be considered a viable strategy when considered in the context of other options.
Presenting a case he performed just prior to the VEITHsymposium, Dr. Siracuse displayed CT images that showed internal and common carotids with more than 75% stenosis in an 80-year-old women 7 years after a primary carotid endarterectomy. The tight stenoses and the evidence of substantial intra-arterial debris were concerns, but a decision to perform a redo endarterectomy was reached after other options, including stenting, were considered.
“She did great. She went home and has had no more symptoms,” Dr. Siracuse reported. “The point is you still have to take these [potential redo endarterectomies] on a case-by case basis.”
Dr. Siracuse reported he had no financial relationships relevant to this study.
NEW YORK – It is well known that reoperative carotid endarterectomy can be technically challenging because of the scarring left from the initial procedure, but an analysis of a large database presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation also revealed that the risk of complications, particularly stroke, is greater.
When “redo” carotid endarterectomies were compared with the index primary procedure collected in the American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP) database, the odds ratio for stroke was several times greater (odds ratio, 3.71; P = .002) on univariate analysis, reported Jeffrey J. Siracuse, MD, associate professor of surgery and radiology at Boston University.
Previous single-center reports of redo endarterectomies “showed terrific results, really no perioperative stroke or morbidity, but this is older data from a different era,” said Dr. Siracuse, who undertook this study to determine whether “real-world” data would tell a different story.
In this study, 75,943 primary carotid endarterectomies and 140 redo procedures were identified in the ACS NSQIP database and compared. The redo population had a significantly higher incidence of end-stage renal disease (3.6% vs. 1.1%; P = .004), but history of stroke, whether with deficit (20.8% vs. 15.4%) or without (11.5% vs. 9.1%), was numerically higher among those undergoing a primary procedure even though these differences did not reach statistical significance. Baseline demographics and comorbidities were otherwise similar.
Presumably because of the difficulty of recanalizing scarred tissue, the mean procedure time for redos was longer than that for the primary procedures (137 vs. 49 minutes; P less than .001), but there were no significant differences in the rate of surgical site infections (0.7% vs. 0.3%; P = .482), return to the operating room (3.6% vs. 4%; P = .853), or 30-day readmissions (2.1% vs. 6.9%; P = .810) for the redo and index procedures, respectively.
Although perioperative MI rates were higher in the redo group (2.1%) than in the primary endarterectomy group (0.9%), this difference did not reach statistical significance (P = .125). However, a multivariate analysis associated redo carotid endarterectomy procedures with a nearly threefold increase in risk of a composite of major adverse cardiovascular events when compared on a multivariate analysis (OR, 2.76; P = .007), Dr. Siracuse reported.
For the surgeons considering a redo carotid endarterectomy, these data “inform a risk-benefit analysis,” according to Dr. Siracuse, but he also said that redo procedures still should be considered a viable strategy when considered in the context of other options.
Presenting a case he performed just prior to the VEITHsymposium, Dr. Siracuse displayed CT images that showed internal and common carotids with more than 75% stenosis in an 80-year-old women 7 years after a primary carotid endarterectomy. The tight stenoses and the evidence of substantial intra-arterial debris were concerns, but a decision to perform a redo endarterectomy was reached after other options, including stenting, were considered.
“She did great. She went home and has had no more symptoms,” Dr. Siracuse reported. “The point is you still have to take these [potential redo endarterectomies] on a case-by case basis.”
Dr. Siracuse reported he had no financial relationships relevant to this study.
REPORTING FROM VEITHSYMPOSIUM
Key clinical point:
Major finding: The odds ratio for stroke is 3.71 times higher (P = .002) with redo than with primary carotid endarterectomy.
Study details: Multivariate retrospective database analysis.
Disclosures: Dr. Siracuse reported he had no financial relationships relevant to this study.
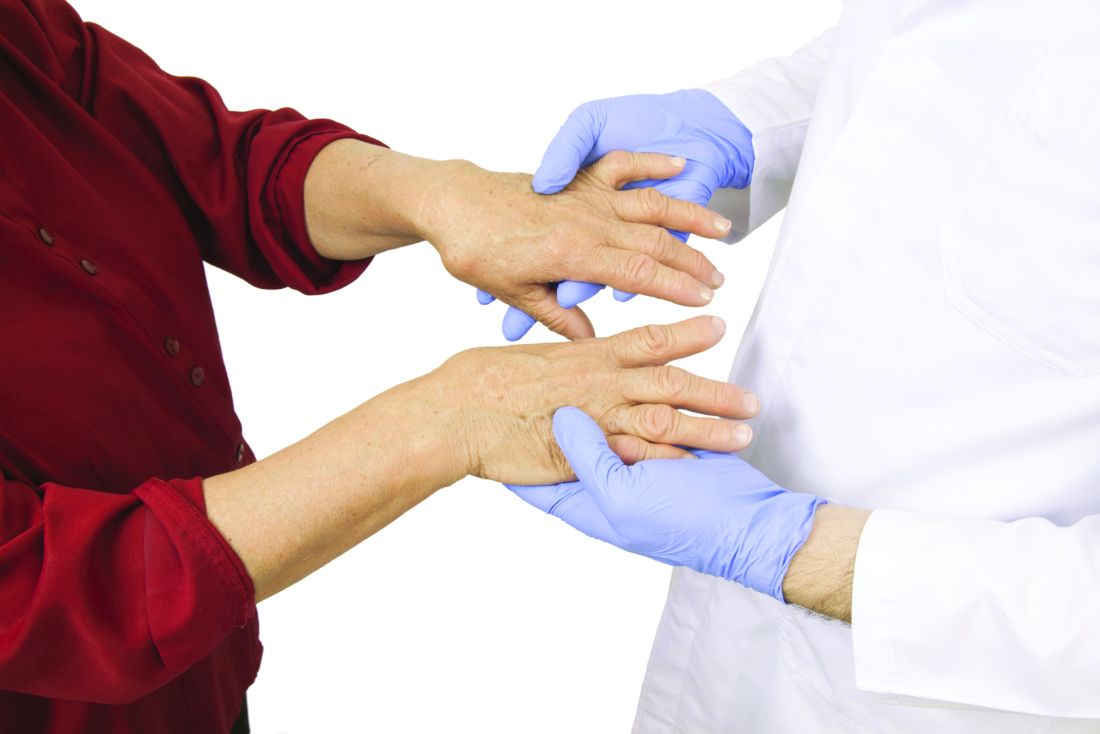
ICU-acquired pneumonia mortality risk may be underestimated
In a large prospectively collected database, the risk of death at 30 days in ICU patients was far greater in those with hospital-acquired pneumonia (HAP) than in those with ventilator-associated pneumonia (VAP) even after adjustment for prognostic factors, according to a large study that compared mortality risk for these complications.
The data for this newly published study were drawn from an evaluation of 14,212 patients treated at 23 ICUs participating in a collaborative French network OUTCOMEREA and published Critical Care Medicine.
HAP in ICU patients “was associated with an 82% increase in the risk of death at day 30,” reported a team of investigators led by Wafa Ibn Saied, MD, of the Université Paris Diderot. Although VAP and HAP were independent risk factors (P both less than .0001) for death at 30 days, VAP increased risk by 38%, less than half of HAP, which increased risk by 82%.
From an observational but prospective database initiated in 1997, this study evaluated 7,735 ICU patients at risk for VAP and 9,747 at risk for HAP. Of those at risk, defined by several factors including an ICU stay of more than 48 hours, HAP developed in 8% and VAP developed in 1%.
The 30-day mortality rates at 30 days after pneumonia were 23.9% for HAP and 28.4% for VAP. The greater risk of death by HR was identified after an analysis that adjusted for mortality risk factors, the adequacy of initial treatment, and other factors, such as prior history of pneumonia.
In HAP patients, the rate of mortality at 30 days was 32% in the 75 who were reintubated but only 16% in the 101 who were not. Adequate empirical therapy within the first 24 hours for HAP was not associated with a reduction in the risk of death.
As in the HAP patients, mortality was not significantly higher in VAP patients who received inadequate empirical therapy, compared with those who did, according to the authors.
Previous studies have suggested that both HAP and VAP increase risk of death in ICU patients, but the authors of this study believe that the relative risk of HAP “is underappreciated.” They asserted, based on these most recent data as well as on previously published analyses, that nonventilated HAP results in “significant increases in cost, length of stay, and mortality.”
The researchers had no disclosures.
SOURCE: Saied WI et al. Crit Care Med. 2018 Nov 7. doi: 10.1097/CCM.0000000000003553.
In a large prospectively collected database, the risk of death at 30 days in ICU patients was far greater in those with hospital-acquired pneumonia (HAP) than in those with ventilator-associated pneumonia (VAP) even after adjustment for prognostic factors, according to a large study that compared mortality risk for these complications.
The data for this newly published study were drawn from an evaluation of 14,212 patients treated at 23 ICUs participating in a collaborative French network OUTCOMEREA and published Critical Care Medicine.
HAP in ICU patients “was associated with an 82% increase in the risk of death at day 30,” reported a team of investigators led by Wafa Ibn Saied, MD, of the Université Paris Diderot. Although VAP and HAP were independent risk factors (P both less than .0001) for death at 30 days, VAP increased risk by 38%, less than half of HAP, which increased risk by 82%.
From an observational but prospective database initiated in 1997, this study evaluated 7,735 ICU patients at risk for VAP and 9,747 at risk for HAP. Of those at risk, defined by several factors including an ICU stay of more than 48 hours, HAP developed in 8% and VAP developed in 1%.
The 30-day mortality rates at 30 days after pneumonia were 23.9% for HAP and 28.4% for VAP. The greater risk of death by HR was identified after an analysis that adjusted for mortality risk factors, the adequacy of initial treatment, and other factors, such as prior history of pneumonia.
In HAP patients, the rate of mortality at 30 days was 32% in the 75 who were reintubated but only 16% in the 101 who were not. Adequate empirical therapy within the first 24 hours for HAP was not associated with a reduction in the risk of death.
As in the HAP patients, mortality was not significantly higher in VAP patients who received inadequate empirical therapy, compared with those who did, according to the authors.
Previous studies have suggested that both HAP and VAP increase risk of death in ICU patients, but the authors of this study believe that the relative risk of HAP “is underappreciated.” They asserted, based on these most recent data as well as on previously published analyses, that nonventilated HAP results in “significant increases in cost, length of stay, and mortality.”
The researchers had no disclosures.
SOURCE: Saied WI et al. Crit Care Med. 2018 Nov 7. doi: 10.1097/CCM.0000000000003553.
In a large prospectively collected database, the risk of death at 30 days in ICU patients was far greater in those with hospital-acquired pneumonia (HAP) than in those with ventilator-associated pneumonia (VAP) even after adjustment for prognostic factors, according to a large study that compared mortality risk for these complications.
The data for this newly published study were drawn from an evaluation of 14,212 patients treated at 23 ICUs participating in a collaborative French network OUTCOMEREA and published Critical Care Medicine.
HAP in ICU patients “was associated with an 82% increase in the risk of death at day 30,” reported a team of investigators led by Wafa Ibn Saied, MD, of the Université Paris Diderot. Although VAP and HAP were independent risk factors (P both less than .0001) for death at 30 days, VAP increased risk by 38%, less than half of HAP, which increased risk by 82%.
From an observational but prospective database initiated in 1997, this study evaluated 7,735 ICU patients at risk for VAP and 9,747 at risk for HAP. Of those at risk, defined by several factors including an ICU stay of more than 48 hours, HAP developed in 8% and VAP developed in 1%.
The 30-day mortality rates at 30 days after pneumonia were 23.9% for HAP and 28.4% for VAP. The greater risk of death by HR was identified after an analysis that adjusted for mortality risk factors, the adequacy of initial treatment, and other factors, such as prior history of pneumonia.
In HAP patients, the rate of mortality at 30 days was 32% in the 75 who were reintubated but only 16% in the 101 who were not. Adequate empirical therapy within the first 24 hours for HAP was not associated with a reduction in the risk of death.
As in the HAP patients, mortality was not significantly higher in VAP patients who received inadequate empirical therapy, compared with those who did, according to the authors.
Previous studies have suggested that both HAP and VAP increase risk of death in ICU patients, but the authors of this study believe that the relative risk of HAP “is underappreciated.” They asserted, based on these most recent data as well as on previously published analyses, that nonventilated HAP results in “significant increases in cost, length of stay, and mortality.”
The researchers had no disclosures.
SOURCE: Saied WI et al. Crit Care Med. 2018 Nov 7. doi: 10.1097/CCM.0000000000003553.
FROM CRITICAL CARE MEDICINE
Key clinical point: Hospital-acquired pneumonia poses a greater risk of death in the ICU than ventilator-associated pneumonia.
Major finding: After prognostic adjustment, the mortality hazard ratios were 1.82 and 1.38 for HAP and VAP, respectively.
Study details: Observational cohort study.
Disclosures: The researchers had no disclosures.
Source: Saied WI et al. Crit Care Med. 2018 Nov 7; doi: 10.1097/CCM.0000000000003553.
Open AAA repair mortality rates doubled for very-low-volume surgeons
NEW YORK – If New York State is representative, the risk of bad outcomes in patients undergoing open abdominal aortic aneurysm repair (OAR) or carotid endarterectomy (CEA), including death in the case of OAR, is about double when performed by very low- versus higher-volume surgeons, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
“What should we do to fix the problem? We could require surgeons to track their outcomes in quality improvement registry,” suggested Jack L. Cronenwett, MD, professor of surgery, Geisel School of Medicine at Dartmouth, Hanover, N.H.
The outcomes were evaluated from inpatient data gathered from patients undergoing OAR or CEA in an all-payer database involving every hospital discharge in New York State. Surgeons were defined as very-low-volume for a given procedure if they averaged one or less per year, though the results held true if very-low-volume was defined as less than three cases per year, according to Dr. Cronenwett.
The database had outcomes on 8,781 OAR procedures and 68,896 CEA procedures performed from 2000 to 2014.
Of the 614 surgeons who performed one or more OARs over this period, 318 (51.8%) were defined as low-volume surgeons. Despite their substantial representation, they performed just 7.6% of the procedures.
When outcomes from procedures performed by very-low-volume surgeons were compared to those done by higher-volume surgeons, the mortality rates without adjustments were nearly double (6.7% vs. 3.5%; P less than .001). Procedures performed by low-volume surgeons were associated with far higher rates of sepsis or shock (5.7% vs. 3.7%; P = .008), and patients treated by low-volume surgeons were more likely to spend 9 or more days in the hospital (39.3% vs. 30.1%; P less than .001).
When fully adjusted for other variables, “low-volume surgeons had twofold higher odds [OR 2.09] of postoperative death,” Dr. Cronenwett reported.
Of the 1,071 surgeons who performed CEA over this period, 512 (47.8%) were low-volume. They performed 1.3% of the procedures.
Mortality and sepsis or shock following CEA were less than 1% in procedures performed by either low- or higher-volume surgeons without significant differences. However, procedures performed by low-volume surgeons were associated with a three-times higher rate of myocardial infarction (1.5% vs. 0.5%; P less than .001) and a 65% higher rate of stroke (3.5% vs. 2.1%; P = .003).
In addition, patients who underwent CEA performed by a low-volume surgeon had a significantly higher rate of 30-day readmission (11.5% vs. 8.5%; P = .002) and a significantly longer median length of stay (2 days vs. 1 day; P less than .001) than did those treated by a higher-volume surgeon.
Whether OAR or CEA, patients treated by a low-volume surgeon were more likely to have Medicaid coverage. The fact that procedures by low-volume surgeons were more likely to be performed in New York City than other areas of the state suggest that access to care was not a variable, according to Dr. Cronenwett.
Surgeon volume was calculated in this study by dividing the total number of OAR or CEA procedures performed by the number of years that the surgeon was in practice in New York State. Surgeons were classified as vascular surgeons if 75% or more of their surgical practice involved vascular procedures, cardiac surgeons if more than 20% of their surgical practice involved cardiac procedures, and general surgeons if they did not meet either of these criteria.
Of OAR procedures were done by a higher-volume surgeon, approximately 65% were by vascular specialists, 5% were by cardiac specialists, and the remaining were by general surgeons.
Of OAR procedures were done by a low-volume surgeon, approximately 25% were by vascular surgeons, 20% were by cardiac surgeons, and the remaining were by general surgeons. For CEA, there was a somewhat greater representation of general surgeons in both categories, but the patterns were similar.
Dr. Cronenwett argued that more rigorous steps should be taken to ensure that those with proven skills perform OAR and CEA and that open abdominal aortic aneurysm repair should be performed only by high-volume surgeons and hospitals. He suggested there are a variety of incentives or disincentives that could help, but he stressed the importance of tracking results and making them available to referring physicians and to patients.
“Some of the low-volume surgeons are probably not tracking their results so are not even aware of these bad outcomes,” he added.
Dr. Cronenwett reported that he had no relevant disclosures.
NEW YORK – If New York State is representative, the risk of bad outcomes in patients undergoing open abdominal aortic aneurysm repair (OAR) or carotid endarterectomy (CEA), including death in the case of OAR, is about double when performed by very low- versus higher-volume surgeons, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
“What should we do to fix the problem? We could require surgeons to track their outcomes in quality improvement registry,” suggested Jack L. Cronenwett, MD, professor of surgery, Geisel School of Medicine at Dartmouth, Hanover, N.H.
The outcomes were evaluated from inpatient data gathered from patients undergoing OAR or CEA in an all-payer database involving every hospital discharge in New York State. Surgeons were defined as very-low-volume for a given procedure if they averaged one or less per year, though the results held true if very-low-volume was defined as less than three cases per year, according to Dr. Cronenwett.
The database had outcomes on 8,781 OAR procedures and 68,896 CEA procedures performed from 2000 to 2014.
Of the 614 surgeons who performed one or more OARs over this period, 318 (51.8%) were defined as low-volume surgeons. Despite their substantial representation, they performed just 7.6% of the procedures.
When outcomes from procedures performed by very-low-volume surgeons were compared to those done by higher-volume surgeons, the mortality rates without adjustments were nearly double (6.7% vs. 3.5%; P less than .001). Procedures performed by low-volume surgeons were associated with far higher rates of sepsis or shock (5.7% vs. 3.7%; P = .008), and patients treated by low-volume surgeons were more likely to spend 9 or more days in the hospital (39.3% vs. 30.1%; P less than .001).
When fully adjusted for other variables, “low-volume surgeons had twofold higher odds [OR 2.09] of postoperative death,” Dr. Cronenwett reported.
Of the 1,071 surgeons who performed CEA over this period, 512 (47.8%) were low-volume. They performed 1.3% of the procedures.
Mortality and sepsis or shock following CEA were less than 1% in procedures performed by either low- or higher-volume surgeons without significant differences. However, procedures performed by low-volume surgeons were associated with a three-times higher rate of myocardial infarction (1.5% vs. 0.5%; P less than .001) and a 65% higher rate of stroke (3.5% vs. 2.1%; P = .003).
In addition, patients who underwent CEA performed by a low-volume surgeon had a significantly higher rate of 30-day readmission (11.5% vs. 8.5%; P = .002) and a significantly longer median length of stay (2 days vs. 1 day; P less than .001) than did those treated by a higher-volume surgeon.
Whether OAR or CEA, patients treated by a low-volume surgeon were more likely to have Medicaid coverage. The fact that procedures by low-volume surgeons were more likely to be performed in New York City than other areas of the state suggest that access to care was not a variable, according to Dr. Cronenwett.
Surgeon volume was calculated in this study by dividing the total number of OAR or CEA procedures performed by the number of years that the surgeon was in practice in New York State. Surgeons were classified as vascular surgeons if 75% or more of their surgical practice involved vascular procedures, cardiac surgeons if more than 20% of their surgical practice involved cardiac procedures, and general surgeons if they did not meet either of these criteria.
Of OAR procedures were done by a higher-volume surgeon, approximately 65% were by vascular specialists, 5% were by cardiac specialists, and the remaining were by general surgeons.
Of OAR procedures were done by a low-volume surgeon, approximately 25% were by vascular surgeons, 20% were by cardiac surgeons, and the remaining were by general surgeons. For CEA, there was a somewhat greater representation of general surgeons in both categories, but the patterns were similar.
Dr. Cronenwett argued that more rigorous steps should be taken to ensure that those with proven skills perform OAR and CEA and that open abdominal aortic aneurysm repair should be performed only by high-volume surgeons and hospitals. He suggested there are a variety of incentives or disincentives that could help, but he stressed the importance of tracking results and making them available to referring physicians and to patients.
“Some of the low-volume surgeons are probably not tracking their results so are not even aware of these bad outcomes,” he added.
Dr. Cronenwett reported that he had no relevant disclosures.
NEW YORK – If New York State is representative, the risk of bad outcomes in patients undergoing open abdominal aortic aneurysm repair (OAR) or carotid endarterectomy (CEA), including death in the case of OAR, is about double when performed by very low- versus higher-volume surgeons, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
“What should we do to fix the problem? We could require surgeons to track their outcomes in quality improvement registry,” suggested Jack L. Cronenwett, MD, professor of surgery, Geisel School of Medicine at Dartmouth, Hanover, N.H.
The outcomes were evaluated from inpatient data gathered from patients undergoing OAR or CEA in an all-payer database involving every hospital discharge in New York State. Surgeons were defined as very-low-volume for a given procedure if they averaged one or less per year, though the results held true if very-low-volume was defined as less than three cases per year, according to Dr. Cronenwett.
The database had outcomes on 8,781 OAR procedures and 68,896 CEA procedures performed from 2000 to 2014.
Of the 614 surgeons who performed one or more OARs over this period, 318 (51.8%) were defined as low-volume surgeons. Despite their substantial representation, they performed just 7.6% of the procedures.
When outcomes from procedures performed by very-low-volume surgeons were compared to those done by higher-volume surgeons, the mortality rates without adjustments were nearly double (6.7% vs. 3.5%; P less than .001). Procedures performed by low-volume surgeons were associated with far higher rates of sepsis or shock (5.7% vs. 3.7%; P = .008), and patients treated by low-volume surgeons were more likely to spend 9 or more days in the hospital (39.3% vs. 30.1%; P less than .001).
When fully adjusted for other variables, “low-volume surgeons had twofold higher odds [OR 2.09] of postoperative death,” Dr. Cronenwett reported.
Of the 1,071 surgeons who performed CEA over this period, 512 (47.8%) were low-volume. They performed 1.3% of the procedures.
Mortality and sepsis or shock following CEA were less than 1% in procedures performed by either low- or higher-volume surgeons without significant differences. However, procedures performed by low-volume surgeons were associated with a three-times higher rate of myocardial infarction (1.5% vs. 0.5%; P less than .001) and a 65% higher rate of stroke (3.5% vs. 2.1%; P = .003).
In addition, patients who underwent CEA performed by a low-volume surgeon had a significantly higher rate of 30-day readmission (11.5% vs. 8.5%; P = .002) and a significantly longer median length of stay (2 days vs. 1 day; P less than .001) than did those treated by a higher-volume surgeon.
Whether OAR or CEA, patients treated by a low-volume surgeon were more likely to have Medicaid coverage. The fact that procedures by low-volume surgeons were more likely to be performed in New York City than other areas of the state suggest that access to care was not a variable, according to Dr. Cronenwett.
Surgeon volume was calculated in this study by dividing the total number of OAR or CEA procedures performed by the number of years that the surgeon was in practice in New York State. Surgeons were classified as vascular surgeons if 75% or more of their surgical practice involved vascular procedures, cardiac surgeons if more than 20% of their surgical practice involved cardiac procedures, and general surgeons if they did not meet either of these criteria.
Of OAR procedures were done by a higher-volume surgeon, approximately 65% were by vascular specialists, 5% were by cardiac specialists, and the remaining were by general surgeons.
Of OAR procedures were done by a low-volume surgeon, approximately 25% were by vascular surgeons, 20% were by cardiac surgeons, and the remaining were by general surgeons. For CEA, there was a somewhat greater representation of general surgeons in both categories, but the patterns were similar.
Dr. Cronenwett argued that more rigorous steps should be taken to ensure that those with proven skills perform OAR and CEA and that open abdominal aortic aneurysm repair should be performed only by high-volume surgeons and hospitals. He suggested there are a variety of incentives or disincentives that could help, but he stressed the importance of tracking results and making them available to referring physicians and to patients.
“Some of the low-volume surgeons are probably not tracking their results so are not even aware of these bad outcomes,” he added.
Dr. Cronenwett reported that he had no relevant disclosures.
REPORTING FROM VEITHSYMPOSIUM
Key clinical point:
Major finding: In-hospital mortality is approximately double (OR 2.09; P less than .001) for very low-volume relative to high-volume surgeon.
Study details: Retrospective database review.
Disclosures: Dr. Cronenwett reports no conflicts of interest.
Source: Cronenwett JL et al. 2018; 45th VEITHsymposium.
Leg ulceration guidelines expected to soon include endovascular ablation
NEW YORK – Guidelines for the management of leg ulcerations will be changed to accommodate the results of the Early Venous Reflux Ablation trial, according to this video interview with the senior author, Alun H Davies, DSc, professor of vascular surgery, Imperial College, London.
In this video interview, conducted at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation, Dr. Davies recaps the major results of the study, which associated immediate endovascular ablation (early intervention) with significantly faster healing than did compression therapy with ablation, considered only after 6 months (delayed intervention).
These data have been published (N Engl J Med 2018 May 31;378:2105-14), but Dr. Davies focused in this interview on the cost efficacy of early intervention with endovascular ablation. In the United Kingdom, where the study was conducted, the data support the cost efficacy, but Dr. Davies predicted even greater savings in the United States because of the expense of frequent wound care visits.
Based on data from a randomized trial, he expects guidelines, including those in the United States, to be revised to list early endovascular ablation as a 1b or 1A recommendation, thereby establishing this intervention as a standard.
If follow-up after 3 years confirms a lower rate of recurrence, an advantage previously shown for open surgery relative to compression healing, the case for early endovascular intervention will be even stronger, according to Dr. Davies.
NEW YORK – Guidelines for the management of leg ulcerations will be changed to accommodate the results of the Early Venous Reflux Ablation trial, according to this video interview with the senior author, Alun H Davies, DSc, professor of vascular surgery, Imperial College, London.
In this video interview, conducted at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation, Dr. Davies recaps the major results of the study, which associated immediate endovascular ablation (early intervention) with significantly faster healing than did compression therapy with ablation, considered only after 6 months (delayed intervention).
These data have been published (N Engl J Med 2018 May 31;378:2105-14), but Dr. Davies focused in this interview on the cost efficacy of early intervention with endovascular ablation. In the United Kingdom, where the study was conducted, the data support the cost efficacy, but Dr. Davies predicted even greater savings in the United States because of the expense of frequent wound care visits.
Based on data from a randomized trial, he expects guidelines, including those in the United States, to be revised to list early endovascular ablation as a 1b or 1A recommendation, thereby establishing this intervention as a standard.
If follow-up after 3 years confirms a lower rate of recurrence, an advantage previously shown for open surgery relative to compression healing, the case for early endovascular intervention will be even stronger, according to Dr. Davies.
NEW YORK – Guidelines for the management of leg ulcerations will be changed to accommodate the results of the Early Venous Reflux Ablation trial, according to this video interview with the senior author, Alun H Davies, DSc, professor of vascular surgery, Imperial College, London.
In this video interview, conducted at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation, Dr. Davies recaps the major results of the study, which associated immediate endovascular ablation (early intervention) with significantly faster healing than did compression therapy with ablation, considered only after 6 months (delayed intervention).
These data have been published (N Engl J Med 2018 May 31;378:2105-14), but Dr. Davies focused in this interview on the cost efficacy of early intervention with endovascular ablation. In the United Kingdom, where the study was conducted, the data support the cost efficacy, but Dr. Davies predicted even greater savings in the United States because of the expense of frequent wound care visits.
Based on data from a randomized trial, he expects guidelines, including those in the United States, to be revised to list early endovascular ablation as a 1b or 1A recommendation, thereby establishing this intervention as a standard.
If follow-up after 3 years confirms a lower rate of recurrence, an advantage previously shown for open surgery relative to compression healing, the case for early endovascular intervention will be even stronger, according to Dr. Davies.
REPORTING FROM VEITHSYMPOSIUM
Embolic protection devices advocated in some lower limb endovascular surgery
NEW YORK – Embolic protection devices to prevent debris from causing complications in percutaneous cardiac procedures also have a role when treating occlusions in the lower extremities, according to an expert who reviewed data and spoke about his experience at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
In this video interview, Peter Schneider, MD, chief of the division of vascular therapy at the Hawaii Kaiser Permanente Medical Group, Honolulu, provided an expert’s opinion about when filters can be helpful to reduce risk of complications.
According to Dr. Schneider, filters do have relative disadvantages so he does not advocate their use when risk is low. Among these disadvantages, the wire used to place the filter can make the endovascular procedure more difficult.
However, he said that there are well-documented cases in which debris in the runoff of a lower limb endovascular procedure resulted in adverse clinical consequences. Importantly, these may not be acute clinical events. Rather, occlusions in the tibial or pedal collaterals may remain asymptomatic for months or years.
Outlining those clinical situations that pose the greatest risk, Dr. Schneider identified several specific patient groups for whom he would advocate filters, particularly those with a large thrombotic burden. Based on his own experience, he provided some tips about situations in which he now employs embolic protection devices routinely.
NEW YORK – Embolic protection devices to prevent debris from causing complications in percutaneous cardiac procedures also have a role when treating occlusions in the lower extremities, according to an expert who reviewed data and spoke about his experience at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
In this video interview, Peter Schneider, MD, chief of the division of vascular therapy at the Hawaii Kaiser Permanente Medical Group, Honolulu, provided an expert’s opinion about when filters can be helpful to reduce risk of complications.
According to Dr. Schneider, filters do have relative disadvantages so he does not advocate their use when risk is low. Among these disadvantages, the wire used to place the filter can make the endovascular procedure more difficult.
However, he said that there are well-documented cases in which debris in the runoff of a lower limb endovascular procedure resulted in adverse clinical consequences. Importantly, these may not be acute clinical events. Rather, occlusions in the tibial or pedal collaterals may remain asymptomatic for months or years.
Outlining those clinical situations that pose the greatest risk, Dr. Schneider identified several specific patient groups for whom he would advocate filters, particularly those with a large thrombotic burden. Based on his own experience, he provided some tips about situations in which he now employs embolic protection devices routinely.
NEW YORK – Embolic protection devices to prevent debris from causing complications in percutaneous cardiac procedures also have a role when treating occlusions in the lower extremities, according to an expert who reviewed data and spoke about his experience at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
In this video interview, Peter Schneider, MD, chief of the division of vascular therapy at the Hawaii Kaiser Permanente Medical Group, Honolulu, provided an expert’s opinion about when filters can be helpful to reduce risk of complications.
According to Dr. Schneider, filters do have relative disadvantages so he does not advocate their use when risk is low. Among these disadvantages, the wire used to place the filter can make the endovascular procedure more difficult.
However, he said that there are well-documented cases in which debris in the runoff of a lower limb endovascular procedure resulted in adverse clinical consequences. Importantly, these may not be acute clinical events. Rather, occlusions in the tibial or pedal collaterals may remain asymptomatic for months or years.
Outlining those clinical situations that pose the greatest risk, Dr. Schneider identified several specific patient groups for whom he would advocate filters, particularly those with a large thrombotic burden. Based on his own experience, he provided some tips about situations in which he now employs embolic protection devices routinely.
REPORTING FROM VEITHSYMPOSIUM
At 10 years, reintervention rate for EVAR is 20%
NEW YORK – A clever strategy to evaluate long term outcomes in patients undergoing endovascular abdominal aortic aneurysm repair identified a 20% rate of reintervention, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation by Philip P. Goodney, MD.
In this video interview with Dr. Goodney, an associate professor of vascular surgery at Geisel School of Medicine at Dartmouth, Hanover, N.H., he explains how Medicare data were employed to track patients long term, even when they had moved to other hospital systems.
The main message from the long-term follow-up is that there is a persistent risk of recurrence and need for reintervention, according to Dr. Goodney. The hypothesis was that there would be an early risk of failure, followed by a diminishing need for reintervention over time, but this was not what was observed.Rather, the findings suggest that the rate of reinterventions was relatively steady over the course of follow-up, suggesting that patients should be informed of a persistent risk. However, Dr. Goodney reports that age was not a predictor of reintervention, so that older patients were at no greater risk.
NEW YORK – A clever strategy to evaluate long term outcomes in patients undergoing endovascular abdominal aortic aneurysm repair identified a 20% rate of reintervention, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation by Philip P. Goodney, MD.
In this video interview with Dr. Goodney, an associate professor of vascular surgery at Geisel School of Medicine at Dartmouth, Hanover, N.H., he explains how Medicare data were employed to track patients long term, even when they had moved to other hospital systems.
The main message from the long-term follow-up is that there is a persistent risk of recurrence and need for reintervention, according to Dr. Goodney. The hypothesis was that there would be an early risk of failure, followed by a diminishing need for reintervention over time, but this was not what was observed.Rather, the findings suggest that the rate of reinterventions was relatively steady over the course of follow-up, suggesting that patients should be informed of a persistent risk. However, Dr. Goodney reports that age was not a predictor of reintervention, so that older patients were at no greater risk.
NEW YORK – A clever strategy to evaluate long term outcomes in patients undergoing endovascular abdominal aortic aneurysm repair identified a 20% rate of reintervention, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation by Philip P. Goodney, MD.
In this video interview with Dr. Goodney, an associate professor of vascular surgery at Geisel School of Medicine at Dartmouth, Hanover, N.H., he explains how Medicare data were employed to track patients long term, even when they had moved to other hospital systems.
The main message from the long-term follow-up is that there is a persistent risk of recurrence and need for reintervention, according to Dr. Goodney. The hypothesis was that there would be an early risk of failure, followed by a diminishing need for reintervention over time, but this was not what was observed.Rather, the findings suggest that the rate of reinterventions was relatively steady over the course of follow-up, suggesting that patients should be informed of a persistent risk. However, Dr. Goodney reports that age was not a predictor of reintervention, so that older patients were at no greater risk.
FROM 2018 VEITH SYMPOSIUM
Snorkel/chimney repair of aortic aneurysms is still effective after 4 years
NEW YORK – With a median follow-up of almost 4 years in more than 200 patients, the snorkel/chimney technique of endovascular repair of complex abdominal aortic aneurysms continues to generate very good results, according data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
In a video interview with Jason T. Lee, MD, professor of vascular surgery, Stanford (Calif.) University, who presented the long-term results, he explains why these findings are important.
After several single-center studies associated the snorkel/chimney technique with good rates of patency and durability, the PERICLES Registry was created almost 10 years ago to test whether these data could be reproduced in the real world.The data presented by Dr. Lee involved outcomes in 244 patients who were followed for at least 2.5 years. The median follow-up is 47 months.
The results overall confirm that this is a viable technique, according to Dr. Lee who noted very little diminution of patency rates in this long-term cohort relative to the previously published follow-up of 17.1 months.
He acknowledged that the snorkel/chimney repair is not free of potential complications, particularly gutter endovascular leaks, but he recounted that at least some resolve over time. Moreover, he suggests that several strategies are being pursued that appear promising for avoiding this risk.
Most importantly, the registry, which captured the experience at 13 centers in the United States and Europe, shows outcomes that are similar to those reported at centers where the techniques were developed and championed.
NEW YORK – With a median follow-up of almost 4 years in more than 200 patients, the snorkel/chimney technique of endovascular repair of complex abdominal aortic aneurysms continues to generate very good results, according data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
In a video interview with Jason T. Lee, MD, professor of vascular surgery, Stanford (Calif.) University, who presented the long-term results, he explains why these findings are important.
After several single-center studies associated the snorkel/chimney technique with good rates of patency and durability, the PERICLES Registry was created almost 10 years ago to test whether these data could be reproduced in the real world.The data presented by Dr. Lee involved outcomes in 244 patients who were followed for at least 2.5 years. The median follow-up is 47 months.
The results overall confirm that this is a viable technique, according to Dr. Lee who noted very little diminution of patency rates in this long-term cohort relative to the previously published follow-up of 17.1 months.
He acknowledged that the snorkel/chimney repair is not free of potential complications, particularly gutter endovascular leaks, but he recounted that at least some resolve over time. Moreover, he suggests that several strategies are being pursued that appear promising for avoiding this risk.
Most importantly, the registry, which captured the experience at 13 centers in the United States and Europe, shows outcomes that are similar to those reported at centers where the techniques were developed and championed.
NEW YORK – With a median follow-up of almost 4 years in more than 200 patients, the snorkel/chimney technique of endovascular repair of complex abdominal aortic aneurysms continues to generate very good results, according data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
In a video interview with Jason T. Lee, MD, professor of vascular surgery, Stanford (Calif.) University, who presented the long-term results, he explains why these findings are important.
After several single-center studies associated the snorkel/chimney technique with good rates of patency and durability, the PERICLES Registry was created almost 10 years ago to test whether these data could be reproduced in the real world.The data presented by Dr. Lee involved outcomes in 244 patients who were followed for at least 2.5 years. The median follow-up is 47 months.
The results overall confirm that this is a viable technique, according to Dr. Lee who noted very little diminution of patency rates in this long-term cohort relative to the previously published follow-up of 17.1 months.
He acknowledged that the snorkel/chimney repair is not free of potential complications, particularly gutter endovascular leaks, but he recounted that at least some resolve over time. Moreover, he suggests that several strategies are being pursued that appear promising for avoiding this risk.
Most importantly, the registry, which captured the experience at 13 centers in the United States and Europe, shows outcomes that are similar to those reported at centers where the techniques were developed and championed.
FROM 2018 VEITH SYMPOSIUM