User login
Novel agents hold promise for frontline AML treatment
Novel therapies are poised to dramatically change frontline therapy for acute myeloid leukemia (AML), and they have the potential to replace chemotherapy, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus.
But more work needs to be done, noted Alexander Perl, MD, MS, associate professor at the University of Pennsylvania, Philadelphia. While advances have transformed AML treatment in the relapsed/refractory setting, “we’re just not seeing that substantive improvement” for newly diagnosed patients, he said. “We need to find the disease-modifying drugs that work in the relapsed/refractory setting and move those frontline. That’s where we’re going to see the transformations.”
Research suggests that low-intensity therapy holds tremendous promise, he said, “with the idea that we could make therapy much more tolerable for the vast majority of patients affected by AML, who, as we know, are older patients.”
Dr. Perl highlighted the 2020 VIALE-A study – venetoclax/azacitidine versus azacitidine/placebo – which reported that “in previously untreated patients who were ineligible for intensive chemotherapy, overall survival was longer and the incidence of remission was higher among patients who received azacitidine plus venetoclax than among those who received azacitidine alone.”
Venetoclax promotes apoptosis in leukemia cells, Dr. Perl said. “To a certain extent, you can think of it as putting the rubber to the road in terms of what actually chemotherapy is designed to do, which is to make leukemic blasts apoptose. It does so without DNA damage and with much less toxicity to the patient. Therefore it can be added to any number of regimens – granted, with mild suppression, but with relatively little extramedullary toxicity.”
Dr. Perl noted that the venetoclax arm “showed a higher response rate than azacitidine in pretty much every subgroup that was looked at, whether patients had de novo leukemia, secondary leukemia, multiple mutational complements, various different karyotypes. The response rates on this study are as high as what we often will see with intensive chemotherapy.” He added that “the winning arm on this trial seems to hold up against any low-intensity therapy, and I would argue against many high-intensity therapies in older patients.”
As for other targeted agents, isocitrate dehydrogenase (IDH) inhibitors “are very promising drugs in the relapsed/refractory setting, which is primarily where these drugs are given. In regard to frontline treatment, “data are coming from a very small study, but they’re very encouraging. It’s hard to entirely say that we’re ready to change practice based on this. But it’s very encouraging – the idea that earlier use of a drug-targeting IDH mutation might lead to substantially better outcomes.”
Moving forward, he said, “we could put all of our eggs in one basket and use many active drugs [at] front line. Or we can perhaps be smart about sequencing these drugs one after another, or using more intensive approaches followed by maintenance approaches followed by more intensive approaches.”
This approach is similar to strategies in myeloma patients “who less and less are relying on an autologous transplant for durable control of their disease, and more and more are using low-intensity biologically targeted drugs,” he said.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Perl reported numerous disclosures, including relationships with Daiichi Sankyo, Abbvie, and Astellas.
Novel therapies are poised to dramatically change frontline therapy for acute myeloid leukemia (AML), and they have the potential to replace chemotherapy, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus.
But more work needs to be done, noted Alexander Perl, MD, MS, associate professor at the University of Pennsylvania, Philadelphia. While advances have transformed AML treatment in the relapsed/refractory setting, “we’re just not seeing that substantive improvement” for newly diagnosed patients, he said. “We need to find the disease-modifying drugs that work in the relapsed/refractory setting and move those frontline. That’s where we’re going to see the transformations.”
Research suggests that low-intensity therapy holds tremendous promise, he said, “with the idea that we could make therapy much more tolerable for the vast majority of patients affected by AML, who, as we know, are older patients.”
Dr. Perl highlighted the 2020 VIALE-A study – venetoclax/azacitidine versus azacitidine/placebo – which reported that “in previously untreated patients who were ineligible for intensive chemotherapy, overall survival was longer and the incidence of remission was higher among patients who received azacitidine plus venetoclax than among those who received azacitidine alone.”
Venetoclax promotes apoptosis in leukemia cells, Dr. Perl said. “To a certain extent, you can think of it as putting the rubber to the road in terms of what actually chemotherapy is designed to do, which is to make leukemic blasts apoptose. It does so without DNA damage and with much less toxicity to the patient. Therefore it can be added to any number of regimens – granted, with mild suppression, but with relatively little extramedullary toxicity.”
Dr. Perl noted that the venetoclax arm “showed a higher response rate than azacitidine in pretty much every subgroup that was looked at, whether patients had de novo leukemia, secondary leukemia, multiple mutational complements, various different karyotypes. The response rates on this study are as high as what we often will see with intensive chemotherapy.” He added that “the winning arm on this trial seems to hold up against any low-intensity therapy, and I would argue against many high-intensity therapies in older patients.”
As for other targeted agents, isocitrate dehydrogenase (IDH) inhibitors “are very promising drugs in the relapsed/refractory setting, which is primarily where these drugs are given. In regard to frontline treatment, “data are coming from a very small study, but they’re very encouraging. It’s hard to entirely say that we’re ready to change practice based on this. But it’s very encouraging – the idea that earlier use of a drug-targeting IDH mutation might lead to substantially better outcomes.”
Moving forward, he said, “we could put all of our eggs in one basket and use many active drugs [at] front line. Or we can perhaps be smart about sequencing these drugs one after another, or using more intensive approaches followed by maintenance approaches followed by more intensive approaches.”
This approach is similar to strategies in myeloma patients “who less and less are relying on an autologous transplant for durable control of their disease, and more and more are using low-intensity biologically targeted drugs,” he said.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Perl reported numerous disclosures, including relationships with Daiichi Sankyo, Abbvie, and Astellas.
Novel therapies are poised to dramatically change frontline therapy for acute myeloid leukemia (AML), and they have the potential to replace chemotherapy, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus.
But more work needs to be done, noted Alexander Perl, MD, MS, associate professor at the University of Pennsylvania, Philadelphia. While advances have transformed AML treatment in the relapsed/refractory setting, “we’re just not seeing that substantive improvement” for newly diagnosed patients, he said. “We need to find the disease-modifying drugs that work in the relapsed/refractory setting and move those frontline. That’s where we’re going to see the transformations.”
Research suggests that low-intensity therapy holds tremendous promise, he said, “with the idea that we could make therapy much more tolerable for the vast majority of patients affected by AML, who, as we know, are older patients.”
Dr. Perl highlighted the 2020 VIALE-A study – venetoclax/azacitidine versus azacitidine/placebo – which reported that “in previously untreated patients who were ineligible for intensive chemotherapy, overall survival was longer and the incidence of remission was higher among patients who received azacitidine plus venetoclax than among those who received azacitidine alone.”
Venetoclax promotes apoptosis in leukemia cells, Dr. Perl said. “To a certain extent, you can think of it as putting the rubber to the road in terms of what actually chemotherapy is designed to do, which is to make leukemic blasts apoptose. It does so without DNA damage and with much less toxicity to the patient. Therefore it can be added to any number of regimens – granted, with mild suppression, but with relatively little extramedullary toxicity.”
Dr. Perl noted that the venetoclax arm “showed a higher response rate than azacitidine in pretty much every subgroup that was looked at, whether patients had de novo leukemia, secondary leukemia, multiple mutational complements, various different karyotypes. The response rates on this study are as high as what we often will see with intensive chemotherapy.” He added that “the winning arm on this trial seems to hold up against any low-intensity therapy, and I would argue against many high-intensity therapies in older patients.”
As for other targeted agents, isocitrate dehydrogenase (IDH) inhibitors “are very promising drugs in the relapsed/refractory setting, which is primarily where these drugs are given. In regard to frontline treatment, “data are coming from a very small study, but they’re very encouraging. It’s hard to entirely say that we’re ready to change practice based on this. But it’s very encouraging – the idea that earlier use of a drug-targeting IDH mutation might lead to substantially better outcomes.”
Moving forward, he said, “we could put all of our eggs in one basket and use many active drugs [at] front line. Or we can perhaps be smart about sequencing these drugs one after another, or using more intensive approaches followed by maintenance approaches followed by more intensive approaches.”
This approach is similar to strategies in myeloma patients “who less and less are relying on an autologous transplant for durable control of their disease, and more and more are using low-intensity biologically targeted drugs,” he said.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Perl reported numerous disclosures, including relationships with Daiichi Sankyo, Abbvie, and Astellas.
FROM ALF 2020
Novel treatments under study for chronic graft-versus-host disease in allo-HCT
Physicians are gaining a greater understanding of the pathophysiology of chronic graft-versus-host disease (cGVHD) in allo-hematopoietic cell transplantation (allo-HCT), a hematologist/oncologist told colleagues, and novel treatments are being tested.
However, options remain limited. There’s only one Food and Drug Administration–approved therapy for cGVHD that’s failed one or more treatments, and clinical trials remain a crucial option in some cases, said Mary E.D. Flowers, MD, professor of medicine at the University of Washington’s Fred Hutchinson Cancer Research Center, Seattle, in a presentation at the virtual Acute Leukemia Forum of Hemedicus.
According to Dr. Flowers, cGVHD – a product of a graft’s “immunological assault” against the person receiving a transplant – occurs in 40% of patients within a year after allo-HCT. The disorder “is associated with a poor quality of life, disability, and increased mortality after allo-transplantation,” she said. “It’s a syndrome that can be inflammatory and fibrotic. It involves several organs – the skin, the mouth, the eyes, the lungs, the GI tract.”
The median length of treatment after peripheral blood stem cell transplant is 3.5 years, Dr. Flowers said. Seven years after treatment, 10% of those who are alive – and have avoided relapse – will still need treatment. “Corticosteroids remain the first-line [treatment], at 0.5-1.0 [mg/kg], but they do not control at least 40% of the patients with cGVHD.”
In regard to pathophysiology, she highlighted a 2017 report that presented findings about the pathophysiology of cGVHD. The findings, the report authors wrote, “have yielded a raft of potential new therapeutics, centered on naive T-cell depletion, interleukin-17/21 inhibition, kinase inhibition, regulatory T-cell restoration, and CSF-1 inhibition.”
For now, no agents other than corticosteroids have shown benefit in cGVHD as initial therapy, Dr. Flowers said. In fact, several trials closed early from lack of benefit. But trials continue, she said: Results are pending for a completed phase 3 trial of ibrutinib, a Bruton tyrosine kinase inhibitor, plus steroids for initial treatment of cGVHD. Nearly 500 patients were enrolled, she said. And there’s an ongoing phase 2/3 trial of itacitinib, a Janus kinase 1 inhibitor plus steroids as initial treatment.
Dr. Flowers highlighted the case of a patient with moderate cGVHD. The patient was treated with infection prophylaxis, supportive care for oral and eyes manifestations, and prednisone 0.5 mg/kg (at a lower dose because of diabetes) plus a substitution of tacrolimus with sirolimus, a calcineurin inhibitor.
Why sirolimus? At this early point in progression, she said, the patient didn’t necessarily need systemwide chemo-suppression, and calcineurin inhibitors can be “quite effective” in management of inflammation in the liver. “It would be a completely different story once the patient develops severe cGVHR.” In that case, she said, calcineurin inhibitors wouldn’t be appropriate.
The patient’s status deteriorated to severe cGVHD, and sirolimus was replaced with ibrutinib. Other drugs were added to prevent infection and treat bronchiolitis obliterans syndrome.
In general, “the goal of the treatment is get adequate control of clinical manifestations and prevent more severe disease from developing,” Dr. Flowers said.
In response to a question about polypharmacy in patient with advanced disease – “we tend not to peel those drugs off” – Dr. Flowers said she does see new patients who appear to be taking too many medications. “They are on five drugs, and I say, ‘What are we doing?’ ”
Quite often, Dr. Flowers said, she doesn’t add therapies to existing ones but instead looks for substitutes. “A clinical lesson that I feel that I learned over time is: Ask your questions first. What would you like to see in 3 months? Or 6 months? Before you just add another therapy, do you really know what the trajectory of a disease might be?”
Dr. Flowers discloses research support (Pharmacyclics, Incyte), speaker honorarium (Janssen, Johnson & Johnson, Astellas, Mallinckrodt), and consulting relationships (Pharmacyclics, CSL Behring, Fresenius Kabi).
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Physicians are gaining a greater understanding of the pathophysiology of chronic graft-versus-host disease (cGVHD) in allo-hematopoietic cell transplantation (allo-HCT), a hematologist/oncologist told colleagues, and novel treatments are being tested.
However, options remain limited. There’s only one Food and Drug Administration–approved therapy for cGVHD that’s failed one or more treatments, and clinical trials remain a crucial option in some cases, said Mary E.D. Flowers, MD, professor of medicine at the University of Washington’s Fred Hutchinson Cancer Research Center, Seattle, in a presentation at the virtual Acute Leukemia Forum of Hemedicus.
According to Dr. Flowers, cGVHD – a product of a graft’s “immunological assault” against the person receiving a transplant – occurs in 40% of patients within a year after allo-HCT. The disorder “is associated with a poor quality of life, disability, and increased mortality after allo-transplantation,” she said. “It’s a syndrome that can be inflammatory and fibrotic. It involves several organs – the skin, the mouth, the eyes, the lungs, the GI tract.”
The median length of treatment after peripheral blood stem cell transplant is 3.5 years, Dr. Flowers said. Seven years after treatment, 10% of those who are alive – and have avoided relapse – will still need treatment. “Corticosteroids remain the first-line [treatment], at 0.5-1.0 [mg/kg], but they do not control at least 40% of the patients with cGVHD.”
In regard to pathophysiology, she highlighted a 2017 report that presented findings about the pathophysiology of cGVHD. The findings, the report authors wrote, “have yielded a raft of potential new therapeutics, centered on naive T-cell depletion, interleukin-17/21 inhibition, kinase inhibition, regulatory T-cell restoration, and CSF-1 inhibition.”
For now, no agents other than corticosteroids have shown benefit in cGVHD as initial therapy, Dr. Flowers said. In fact, several trials closed early from lack of benefit. But trials continue, she said: Results are pending for a completed phase 3 trial of ibrutinib, a Bruton tyrosine kinase inhibitor, plus steroids for initial treatment of cGVHD. Nearly 500 patients were enrolled, she said. And there’s an ongoing phase 2/3 trial of itacitinib, a Janus kinase 1 inhibitor plus steroids as initial treatment.
Dr. Flowers highlighted the case of a patient with moderate cGVHD. The patient was treated with infection prophylaxis, supportive care for oral and eyes manifestations, and prednisone 0.5 mg/kg (at a lower dose because of diabetes) plus a substitution of tacrolimus with sirolimus, a calcineurin inhibitor.
Why sirolimus? At this early point in progression, she said, the patient didn’t necessarily need systemwide chemo-suppression, and calcineurin inhibitors can be “quite effective” in management of inflammation in the liver. “It would be a completely different story once the patient develops severe cGVHR.” In that case, she said, calcineurin inhibitors wouldn’t be appropriate.
The patient’s status deteriorated to severe cGVHD, and sirolimus was replaced with ibrutinib. Other drugs were added to prevent infection and treat bronchiolitis obliterans syndrome.
In general, “the goal of the treatment is get adequate control of clinical manifestations and prevent more severe disease from developing,” Dr. Flowers said.
In response to a question about polypharmacy in patient with advanced disease – “we tend not to peel those drugs off” – Dr. Flowers said she does see new patients who appear to be taking too many medications. “They are on five drugs, and I say, ‘What are we doing?’ ”
Quite often, Dr. Flowers said, she doesn’t add therapies to existing ones but instead looks for substitutes. “A clinical lesson that I feel that I learned over time is: Ask your questions first. What would you like to see in 3 months? Or 6 months? Before you just add another therapy, do you really know what the trajectory of a disease might be?”
Dr. Flowers discloses research support (Pharmacyclics, Incyte), speaker honorarium (Janssen, Johnson & Johnson, Astellas, Mallinckrodt), and consulting relationships (Pharmacyclics, CSL Behring, Fresenius Kabi).
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Physicians are gaining a greater understanding of the pathophysiology of chronic graft-versus-host disease (cGVHD) in allo-hematopoietic cell transplantation (allo-HCT), a hematologist/oncologist told colleagues, and novel treatments are being tested.
However, options remain limited. There’s only one Food and Drug Administration–approved therapy for cGVHD that’s failed one or more treatments, and clinical trials remain a crucial option in some cases, said Mary E.D. Flowers, MD, professor of medicine at the University of Washington’s Fred Hutchinson Cancer Research Center, Seattle, in a presentation at the virtual Acute Leukemia Forum of Hemedicus.
According to Dr. Flowers, cGVHD – a product of a graft’s “immunological assault” against the person receiving a transplant – occurs in 40% of patients within a year after allo-HCT. The disorder “is associated with a poor quality of life, disability, and increased mortality after allo-transplantation,” she said. “It’s a syndrome that can be inflammatory and fibrotic. It involves several organs – the skin, the mouth, the eyes, the lungs, the GI tract.”
The median length of treatment after peripheral blood stem cell transplant is 3.5 years, Dr. Flowers said. Seven years after treatment, 10% of those who are alive – and have avoided relapse – will still need treatment. “Corticosteroids remain the first-line [treatment], at 0.5-1.0 [mg/kg], but they do not control at least 40% of the patients with cGVHD.”
In regard to pathophysiology, she highlighted a 2017 report that presented findings about the pathophysiology of cGVHD. The findings, the report authors wrote, “have yielded a raft of potential new therapeutics, centered on naive T-cell depletion, interleukin-17/21 inhibition, kinase inhibition, regulatory T-cell restoration, and CSF-1 inhibition.”
For now, no agents other than corticosteroids have shown benefit in cGVHD as initial therapy, Dr. Flowers said. In fact, several trials closed early from lack of benefit. But trials continue, she said: Results are pending for a completed phase 3 trial of ibrutinib, a Bruton tyrosine kinase inhibitor, plus steroids for initial treatment of cGVHD. Nearly 500 patients were enrolled, she said. And there’s an ongoing phase 2/3 trial of itacitinib, a Janus kinase 1 inhibitor plus steroids as initial treatment.
Dr. Flowers highlighted the case of a patient with moderate cGVHD. The patient was treated with infection prophylaxis, supportive care for oral and eyes manifestations, and prednisone 0.5 mg/kg (at a lower dose because of diabetes) plus a substitution of tacrolimus with sirolimus, a calcineurin inhibitor.
Why sirolimus? At this early point in progression, she said, the patient didn’t necessarily need systemwide chemo-suppression, and calcineurin inhibitors can be “quite effective” in management of inflammation in the liver. “It would be a completely different story once the patient develops severe cGVHR.” In that case, she said, calcineurin inhibitors wouldn’t be appropriate.
The patient’s status deteriorated to severe cGVHD, and sirolimus was replaced with ibrutinib. Other drugs were added to prevent infection and treat bronchiolitis obliterans syndrome.
In general, “the goal of the treatment is get adequate control of clinical manifestations and prevent more severe disease from developing,” Dr. Flowers said.
In response to a question about polypharmacy in patient with advanced disease – “we tend not to peel those drugs off” – Dr. Flowers said she does see new patients who appear to be taking too many medications. “They are on five drugs, and I say, ‘What are we doing?’ ”
Quite often, Dr. Flowers said, she doesn’t add therapies to existing ones but instead looks for substitutes. “A clinical lesson that I feel that I learned over time is: Ask your questions first. What would you like to see in 3 months? Or 6 months? Before you just add another therapy, do you really know what the trajectory of a disease might be?”
Dr. Flowers discloses research support (Pharmacyclics, Incyte), speaker honorarium (Janssen, Johnson & Johnson, Astellas, Mallinckrodt), and consulting relationships (Pharmacyclics, CSL Behring, Fresenius Kabi).
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
FROM ALF 2020
Standard treatment lacking in relapsed refractory AML
Despite a variety of options, patients with relapsed/refractory acute myeloid leukemia (AML) continue to face poor prognoses, and a standard of care remains elusive, a hematologist/oncologist told colleagues.
“Clearly we have a problem with this group of patients,” Ehab Atallah, MD, professor of medicine at Medical College of Wisconsin, Milwaukee, said in a presentation at the virtual Acute Leukemia Forum of Hemedicus. In regard to treatments, he added, “we still have multiple unanswered questions.”
As Dr. Atallah noted, a 2018 study of 3,012 patients – in 9 successive ECOG‐ACRIN trials for newly diagnosed AML from 1984-2008 – showed poor outcomes for relapsed/refractory patients. At a median follow-up of 9.7 years, 59.1% reached first complete remission (CR1), and 58.9% of those relapsed. In the relapsed patients, the median overall survival from relapse was 0.5 years, and the overall survival (OS) over 5 years was 10%.
“Even among patients who relapsed with better prognostic factors – age < 40 and CR1 > 12 months – there was no significant OS difference between the studies,” the study noted. “In conclusion, this large cohort appears to confirm that the survival of AML patients post relapse continues to be dismal and has not improved during the past quarter of a century.”
There isn’t a clear standard of care, said Dr. Atallah, as shown by a 2014 phase 3 study of elacytarabine vs. investigator choice in relapsed/refractory AML patients. The investigators chose seven treatment options for the control arm.
So how can physicians make the best decisions about treatment? A 2018 report finds that some factors do offer guidance about how well relapsed patients will do, Dr. Atallah said, including worse prognoses for higher age (>50 years), time to relapse (< 1 year), number of cycles of treatment needed to achieve remission (more than 1), and unfavorable cytogenetics. And, he said, “practically no one is cured when their leukemia relapses without stem cell transplantation.”
Also keep comorbidities in mind, he said, and consider previous therapies – not just the ones implemented prior to their induction but from all treatments they received: “How much anthracycline did they get? Do they still have room to receive any more anthracycline? Do they have any pulmonary complications from GVHD [graft versus host disease]?”
Another tool may be helpful. A 2013 study found that geriatric assessment predicted survival for older adults with AML who took induction chemotherapy, he said. “I’m pretty sure that this geriatric assessment would also have significant prognostic information for patients with relapsed refractory AML.”
Molecular changes add to the complexity of treatment for relapsed/refractory AML, Dr. Atallah said, in light of new molecularly targeted drugs. He pointed to a 2019 study that showed a slight increase in median overall survival (9.3 months vs. 5.6 months) for gilteritinib vs. salvage chemotherapy in relapsed/refractory patients with FLT3-mutated AML. Other studies have shown limited effects of ID1 inhibitors, he said.
In the big picture, “there are many patient-, disease-, and prior-therapy-related variables that are involved in our decisions plus donor availability, social support, whether they have a transplant before, what kind of treatment they got before the functional assessment, and comorbidities. Even with the current choices for relapsed/refractory AML, the overall survival remains poor. Enrollment in clinical trials would be the best option for these patients.”
Dr. Atallah disclosed ties with Jazz, Abbvie, Takeda, Celgene, and Novartis.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
SOURCE: “Why Is There No Standard of Care for Relapsed AML?” Acute Leukemia Forum of Hemedicus, Oct. 15, 2020.
Despite a variety of options, patients with relapsed/refractory acute myeloid leukemia (AML) continue to face poor prognoses, and a standard of care remains elusive, a hematologist/oncologist told colleagues.
“Clearly we have a problem with this group of patients,” Ehab Atallah, MD, professor of medicine at Medical College of Wisconsin, Milwaukee, said in a presentation at the virtual Acute Leukemia Forum of Hemedicus. In regard to treatments, he added, “we still have multiple unanswered questions.”
As Dr. Atallah noted, a 2018 study of 3,012 patients – in 9 successive ECOG‐ACRIN trials for newly diagnosed AML from 1984-2008 – showed poor outcomes for relapsed/refractory patients. At a median follow-up of 9.7 years, 59.1% reached first complete remission (CR1), and 58.9% of those relapsed. In the relapsed patients, the median overall survival from relapse was 0.5 years, and the overall survival (OS) over 5 years was 10%.
“Even among patients who relapsed with better prognostic factors – age < 40 and CR1 > 12 months – there was no significant OS difference between the studies,” the study noted. “In conclusion, this large cohort appears to confirm that the survival of AML patients post relapse continues to be dismal and has not improved during the past quarter of a century.”
There isn’t a clear standard of care, said Dr. Atallah, as shown by a 2014 phase 3 study of elacytarabine vs. investigator choice in relapsed/refractory AML patients. The investigators chose seven treatment options for the control arm.
So how can physicians make the best decisions about treatment? A 2018 report finds that some factors do offer guidance about how well relapsed patients will do, Dr. Atallah said, including worse prognoses for higher age (>50 years), time to relapse (< 1 year), number of cycles of treatment needed to achieve remission (more than 1), and unfavorable cytogenetics. And, he said, “practically no one is cured when their leukemia relapses without stem cell transplantation.”
Also keep comorbidities in mind, he said, and consider previous therapies – not just the ones implemented prior to their induction but from all treatments they received: “How much anthracycline did they get? Do they still have room to receive any more anthracycline? Do they have any pulmonary complications from GVHD [graft versus host disease]?”
Another tool may be helpful. A 2013 study found that geriatric assessment predicted survival for older adults with AML who took induction chemotherapy, he said. “I’m pretty sure that this geriatric assessment would also have significant prognostic information for patients with relapsed refractory AML.”
Molecular changes add to the complexity of treatment for relapsed/refractory AML, Dr. Atallah said, in light of new molecularly targeted drugs. He pointed to a 2019 study that showed a slight increase in median overall survival (9.3 months vs. 5.6 months) for gilteritinib vs. salvage chemotherapy in relapsed/refractory patients with FLT3-mutated AML. Other studies have shown limited effects of ID1 inhibitors, he said.
In the big picture, “there are many patient-, disease-, and prior-therapy-related variables that are involved in our decisions plus donor availability, social support, whether they have a transplant before, what kind of treatment they got before the functional assessment, and comorbidities. Even with the current choices for relapsed/refractory AML, the overall survival remains poor. Enrollment in clinical trials would be the best option for these patients.”
Dr. Atallah disclosed ties with Jazz, Abbvie, Takeda, Celgene, and Novartis.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
SOURCE: “Why Is There No Standard of Care for Relapsed AML?” Acute Leukemia Forum of Hemedicus, Oct. 15, 2020.
Despite a variety of options, patients with relapsed/refractory acute myeloid leukemia (AML) continue to face poor prognoses, and a standard of care remains elusive, a hematologist/oncologist told colleagues.
“Clearly we have a problem with this group of patients,” Ehab Atallah, MD, professor of medicine at Medical College of Wisconsin, Milwaukee, said in a presentation at the virtual Acute Leukemia Forum of Hemedicus. In regard to treatments, he added, “we still have multiple unanswered questions.”
As Dr. Atallah noted, a 2018 study of 3,012 patients – in 9 successive ECOG‐ACRIN trials for newly diagnosed AML from 1984-2008 – showed poor outcomes for relapsed/refractory patients. At a median follow-up of 9.7 years, 59.1% reached first complete remission (CR1), and 58.9% of those relapsed. In the relapsed patients, the median overall survival from relapse was 0.5 years, and the overall survival (OS) over 5 years was 10%.
“Even among patients who relapsed with better prognostic factors – age < 40 and CR1 > 12 months – there was no significant OS difference between the studies,” the study noted. “In conclusion, this large cohort appears to confirm that the survival of AML patients post relapse continues to be dismal and has not improved during the past quarter of a century.”
There isn’t a clear standard of care, said Dr. Atallah, as shown by a 2014 phase 3 study of elacytarabine vs. investigator choice in relapsed/refractory AML patients. The investigators chose seven treatment options for the control arm.
So how can physicians make the best decisions about treatment? A 2018 report finds that some factors do offer guidance about how well relapsed patients will do, Dr. Atallah said, including worse prognoses for higher age (>50 years), time to relapse (< 1 year), number of cycles of treatment needed to achieve remission (more than 1), and unfavorable cytogenetics. And, he said, “practically no one is cured when their leukemia relapses without stem cell transplantation.”
Also keep comorbidities in mind, he said, and consider previous therapies – not just the ones implemented prior to their induction but from all treatments they received: “How much anthracycline did they get? Do they still have room to receive any more anthracycline? Do they have any pulmonary complications from GVHD [graft versus host disease]?”
Another tool may be helpful. A 2013 study found that geriatric assessment predicted survival for older adults with AML who took induction chemotherapy, he said. “I’m pretty sure that this geriatric assessment would also have significant prognostic information for patients with relapsed refractory AML.”
Molecular changes add to the complexity of treatment for relapsed/refractory AML, Dr. Atallah said, in light of new molecularly targeted drugs. He pointed to a 2019 study that showed a slight increase in median overall survival (9.3 months vs. 5.6 months) for gilteritinib vs. salvage chemotherapy in relapsed/refractory patients with FLT3-mutated AML. Other studies have shown limited effects of ID1 inhibitors, he said.
In the big picture, “there are many patient-, disease-, and prior-therapy-related variables that are involved in our decisions plus donor availability, social support, whether they have a transplant before, what kind of treatment they got before the functional assessment, and comorbidities. Even with the current choices for relapsed/refractory AML, the overall survival remains poor. Enrollment in clinical trials would be the best option for these patients.”
Dr. Atallah disclosed ties with Jazz, Abbvie, Takeda, Celgene, and Novartis.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
SOURCE: “Why Is There No Standard of Care for Relapsed AML?” Acute Leukemia Forum of Hemedicus, Oct. 15, 2020.
FROM ALF 2020
Can AML patients be too old for cell transplantation?
How old is too old for a patient to undergo hematopoietic cell transplantation (HCT)? That’s the wrong question to ask, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus. Instead, he said, look at other factors such as disease status and genetics.
“Transplantation for older patients, even beyond the age of 70, is acceptable, as long as it’s done with caution, care, and wisdom. So we’re all not too old for transplantation, at least not today,” said Daniel Weisdorf, MD, professor of medicine and deputy director of the University of Minnesota Clinical and Translational Science Institute.
As he noted, acute myeloid leukemia (AML) is often fatal. Among the general population, “the expected survival life expectancy at age 75 is 98% at 1 year, and most people living at 75 go on to live more than 10 years,” he said. “But if you have AML, at age 75, you have 20% survival at 1 year, 4% at 3 years. And since the median age of AML diagnosis is 68, and 75% of patients are diagnosed beyond the age of 55, this becomes relevant.”
Risk factors that affect survival after transplantation “certainly include age, but that interacts directly with the comorbidities people accumulate with age, their assessments of frailty, and their Karnofsky performance status, as well as the disease phenotype and molecular genetic markers,” Dr. Weisdorf said. “Perhaps most importantly, though not addressed very much, is patients’ willingness to undertake intensive therapy and their life outlook related to patient-reported outcomes when they get older.”
Despite the lack of indications that higher age by itself is an influential factor in survival after transplant, “we are generally reluctant to push the age of eligibility,” Dr. Weisdorf said. He noted that recently published American Society of Hematology guidelines for treatment of AML over the age of 55 “don’t discuss anything about transplantation fitness because they didn’t want to tackle that.”
Overall survival (OS) at 1 year after allogenic transplants only dipped slightly from ages 51-60 to 71 and above, according to Dr. Weisdorf’s analysis of U.S. data collected by the Center for International Blood and Marrow Transplant Research for the time period 2005-2019.
OS was 67.6% (66.8%-68.3%) for the 41-50 age group (n = 9,287) and 57.9% (56.1%-59.8%) for the 71 and older group, Dr. Weisdorf found. Overall, OS dropped by about 4 percentage points per decade of age, he said, revealing a “modest influence” of advancing years.
His analysis of autologous transplant data from the same source, also for 2005-2019, revealed “essentially no age influence.” OS was 90.8% (90.3%-91.2%) for the 41-50 age group (n = 15,075) and 86.6% (85.9%-87.3%) for the 71 and older group (n = 7,247).
Dr. Weisdorf also highlighted unpublished research that suggests that cord-blood transplant recipients older than 70 face a significantly higher risk of death than that of younger patients in the same category. Cord blood “may be option of last resort” because of a lack of other options, he explained. “And it may be part of the learning curve of cord blood transplantation, which grew a little bit in the early 2000s, and maybe past 2010, and then fell off as everybody got enamored with the haploidentical transplant option.”
How can physicians make decisions about transplants in older patients? “The transplant comorbidity index, the specific comorbidities themselves, performance score, and frailty are all measures of somebody’s fitness to be a good candidate for transplant, really at any age,” Dr. Weisdorf said. “But we also have to recognize that disease status, genetics, and the risk phenotype remain critical and should influence decision making.”
However, even as transplant survival improves overall, “very few people are incorporating any very specific biological markers” in decision-making, he said. “We’ve gotten to measures of frailty, but we haven’t gotten to any biologic measures of cytokines or other things that would predict poor chances for doing well. So I’m afraid we’re still standing at the foot of the bed saying: ‘You look okay.’ Or we’re measuring their comorbidity index. But it is disappointing that we’re using mostly very simple clinical measures to decide if somebody is sturdy enough to proceed, and we perhaps need something better. But I don’t have a great suggestion what it should be.”
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Weisdorf disclosed consulting fees from Fate Therapeutics and Incyte Corp.
SOURCE: “The Ever-Increasing Upper Age for Transplant: Is This Evidence-Based?” Acute Leukemia Forum of Hemedicus, Oct. 15, 2020.
How old is too old for a patient to undergo hematopoietic cell transplantation (HCT)? That’s the wrong question to ask, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus. Instead, he said, look at other factors such as disease status and genetics.
“Transplantation for older patients, even beyond the age of 70, is acceptable, as long as it’s done with caution, care, and wisdom. So we’re all not too old for transplantation, at least not today,” said Daniel Weisdorf, MD, professor of medicine and deputy director of the University of Minnesota Clinical and Translational Science Institute.
As he noted, acute myeloid leukemia (AML) is often fatal. Among the general population, “the expected survival life expectancy at age 75 is 98% at 1 year, and most people living at 75 go on to live more than 10 years,” he said. “But if you have AML, at age 75, you have 20% survival at 1 year, 4% at 3 years. And since the median age of AML diagnosis is 68, and 75% of patients are diagnosed beyond the age of 55, this becomes relevant.”
Risk factors that affect survival after transplantation “certainly include age, but that interacts directly with the comorbidities people accumulate with age, their assessments of frailty, and their Karnofsky performance status, as well as the disease phenotype and molecular genetic markers,” Dr. Weisdorf said. “Perhaps most importantly, though not addressed very much, is patients’ willingness to undertake intensive therapy and their life outlook related to patient-reported outcomes when they get older.”
Despite the lack of indications that higher age by itself is an influential factor in survival after transplant, “we are generally reluctant to push the age of eligibility,” Dr. Weisdorf said. He noted that recently published American Society of Hematology guidelines for treatment of AML over the age of 55 “don’t discuss anything about transplantation fitness because they didn’t want to tackle that.”
Overall survival (OS) at 1 year after allogenic transplants only dipped slightly from ages 51-60 to 71 and above, according to Dr. Weisdorf’s analysis of U.S. data collected by the Center for International Blood and Marrow Transplant Research for the time period 2005-2019.
OS was 67.6% (66.8%-68.3%) for the 41-50 age group (n = 9,287) and 57.9% (56.1%-59.8%) for the 71 and older group, Dr. Weisdorf found. Overall, OS dropped by about 4 percentage points per decade of age, he said, revealing a “modest influence” of advancing years.
His analysis of autologous transplant data from the same source, also for 2005-2019, revealed “essentially no age influence.” OS was 90.8% (90.3%-91.2%) for the 41-50 age group (n = 15,075) and 86.6% (85.9%-87.3%) for the 71 and older group (n = 7,247).
Dr. Weisdorf also highlighted unpublished research that suggests that cord-blood transplant recipients older than 70 face a significantly higher risk of death than that of younger patients in the same category. Cord blood “may be option of last resort” because of a lack of other options, he explained. “And it may be part of the learning curve of cord blood transplantation, which grew a little bit in the early 2000s, and maybe past 2010, and then fell off as everybody got enamored with the haploidentical transplant option.”
How can physicians make decisions about transplants in older patients? “The transplant comorbidity index, the specific comorbidities themselves, performance score, and frailty are all measures of somebody’s fitness to be a good candidate for transplant, really at any age,” Dr. Weisdorf said. “But we also have to recognize that disease status, genetics, and the risk phenotype remain critical and should influence decision making.”
However, even as transplant survival improves overall, “very few people are incorporating any very specific biological markers” in decision-making, he said. “We’ve gotten to measures of frailty, but we haven’t gotten to any biologic measures of cytokines or other things that would predict poor chances for doing well. So I’m afraid we’re still standing at the foot of the bed saying: ‘You look okay.’ Or we’re measuring their comorbidity index. But it is disappointing that we’re using mostly very simple clinical measures to decide if somebody is sturdy enough to proceed, and we perhaps need something better. But I don’t have a great suggestion what it should be.”
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Weisdorf disclosed consulting fees from Fate Therapeutics and Incyte Corp.
SOURCE: “The Ever-Increasing Upper Age for Transplant: Is This Evidence-Based?” Acute Leukemia Forum of Hemedicus, Oct. 15, 2020.
How old is too old for a patient to undergo hematopoietic cell transplantation (HCT)? That’s the wrong question to ask, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus. Instead, he said, look at other factors such as disease status and genetics.
“Transplantation for older patients, even beyond the age of 70, is acceptable, as long as it’s done with caution, care, and wisdom. So we’re all not too old for transplantation, at least not today,” said Daniel Weisdorf, MD, professor of medicine and deputy director of the University of Minnesota Clinical and Translational Science Institute.
As he noted, acute myeloid leukemia (AML) is often fatal. Among the general population, “the expected survival life expectancy at age 75 is 98% at 1 year, and most people living at 75 go on to live more than 10 years,” he said. “But if you have AML, at age 75, you have 20% survival at 1 year, 4% at 3 years. And since the median age of AML diagnosis is 68, and 75% of patients are diagnosed beyond the age of 55, this becomes relevant.”
Risk factors that affect survival after transplantation “certainly include age, but that interacts directly with the comorbidities people accumulate with age, their assessments of frailty, and their Karnofsky performance status, as well as the disease phenotype and molecular genetic markers,” Dr. Weisdorf said. “Perhaps most importantly, though not addressed very much, is patients’ willingness to undertake intensive therapy and their life outlook related to patient-reported outcomes when they get older.”
Despite the lack of indications that higher age by itself is an influential factor in survival after transplant, “we are generally reluctant to push the age of eligibility,” Dr. Weisdorf said. He noted that recently published American Society of Hematology guidelines for treatment of AML over the age of 55 “don’t discuss anything about transplantation fitness because they didn’t want to tackle that.”
Overall survival (OS) at 1 year after allogenic transplants only dipped slightly from ages 51-60 to 71 and above, according to Dr. Weisdorf’s analysis of U.S. data collected by the Center for International Blood and Marrow Transplant Research for the time period 2005-2019.
OS was 67.6% (66.8%-68.3%) for the 41-50 age group (n = 9,287) and 57.9% (56.1%-59.8%) for the 71 and older group, Dr. Weisdorf found. Overall, OS dropped by about 4 percentage points per decade of age, he said, revealing a “modest influence” of advancing years.
His analysis of autologous transplant data from the same source, also for 2005-2019, revealed “essentially no age influence.” OS was 90.8% (90.3%-91.2%) for the 41-50 age group (n = 15,075) and 86.6% (85.9%-87.3%) for the 71 and older group (n = 7,247).
Dr. Weisdorf also highlighted unpublished research that suggests that cord-blood transplant recipients older than 70 face a significantly higher risk of death than that of younger patients in the same category. Cord blood “may be option of last resort” because of a lack of other options, he explained. “And it may be part of the learning curve of cord blood transplantation, which grew a little bit in the early 2000s, and maybe past 2010, and then fell off as everybody got enamored with the haploidentical transplant option.”
How can physicians make decisions about transplants in older patients? “The transplant comorbidity index, the specific comorbidities themselves, performance score, and frailty are all measures of somebody’s fitness to be a good candidate for transplant, really at any age,” Dr. Weisdorf said. “But we also have to recognize that disease status, genetics, and the risk phenotype remain critical and should influence decision making.”
However, even as transplant survival improves overall, “very few people are incorporating any very specific biological markers” in decision-making, he said. “We’ve gotten to measures of frailty, but we haven’t gotten to any biologic measures of cytokines or other things that would predict poor chances for doing well. So I’m afraid we’re still standing at the foot of the bed saying: ‘You look okay.’ Or we’re measuring their comorbidity index. But it is disappointing that we’re using mostly very simple clinical measures to decide if somebody is sturdy enough to proceed, and we perhaps need something better. But I don’t have a great suggestion what it should be.”
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Weisdorf disclosed consulting fees from Fate Therapeutics and Incyte Corp.
SOURCE: “The Ever-Increasing Upper Age for Transplant: Is This Evidence-Based?” Acute Leukemia Forum of Hemedicus, Oct. 15, 2020.
FROM ALF 2020
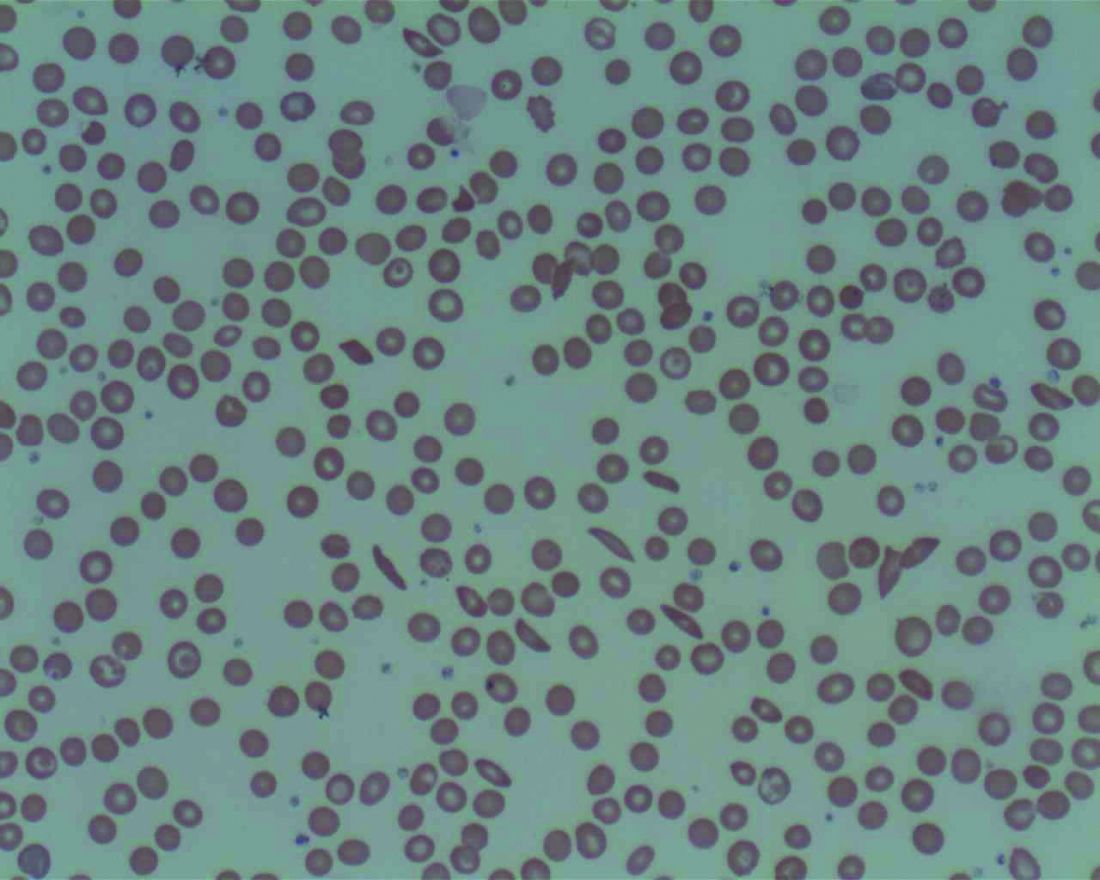
Landmark sickle cell report targets massive failures, calls for action
The National Academies of Science, Engineering, and Medicine have just released a 522-page report, but it’s not the usual compilation of guidelines for treatment of a disease. Instead, the authors of “Addressing Sickle Cell Disease: A Strategic Plan and Blueprint for Action” argue in stark terms that the American society has colossally failed individuals living with sickle cell disease (SCD), who are mostly Black or Brown. A dramatic overhaul of the country’s medical and societal priorities is needed to turn things around to improve health and longevity among this rare disease population.
The findings from the NASEM report are explicit: “There has been substantial success in increasing the survival of children with SCD, but this success had not been translated to similar success as they become adults.” One factor posited to contribute to the slow progress in the improvement of quality and quantity of life for adults living with this disease is the fact that “SCD is largely a disease of African Americans, and as such exists in a context of racial discrimination, health and other societal disparities, mistrust of the health care system, and the effects of poverty.” The report also cites the substantial evidence that those with SCD may receive poorer quality of care.
The report’s 14 authors were made up of an ad hoc committee formed at the request of the Department of Health & Human Services’ Office of Minority Health. The office asked NASEM to convene the committee to develop a strategic plan and blueprint for the United States and others regarding SCD.
The NASEM SCD committee members “realized that we can’t address the medical components of SCD if we don’t explore societal issues and why it’s been so hard to get good care for people with sickle cell disease,” hematologist and report coauthor Ifeyinwa (Ify) Osunkwo, MD, professor of medicine and pediatrics at Atrium Health and director of the Sickle Cell Disease Enterprise, Levine Cancer Institute, Charlotte, N.C., said in an interview. Dr. Osunkwo is also the medical editor of Hematology News.
“After almost a year of meetings and digging into the background and history of SCD care, we came out with very comprehensive summary of where we were and where we want to be,” she said. “The report provides short-, intermediate- and long-term recommendations and identifies which entity and organization should be responsible for implementing them.”
The report authors, led by pediatrician and committee chair Marie Clare McCormick, MD, of the Harvard School of Public Health, Boston, stated that about 100,000 people in the United States and millions worldwide live with SCD. The disease kills more than 700 people per year in the United States, and treatment costs an estimated $2 billion a year.
When judged by disability-adjusted life-years lost – a measurement of expected healthy years of life without an illness – the impact of SCD on individuals is estimated to be greater than a long list of other diseases such as Alzheimer’s disease, breast cancer, type 1 diabetes, and AIDS/HIV, the report noted.
“The health care needs of individuals living with SCD have been neglected by the U.S. and global health care systems, causing them and their families to suffer,” the report said. “Many of the complications that afflict individuals with SCD, particularly pain, are invisible. Pain is only diagnosed by self-reports, and in SCD there are few to no external indicators of the pain experience. Nevertheless, the pain can be excruciatingly severe and requires treatment with strong analgesics.”
There’s even more misery to the story of SCD, the report said, and Dr. Osunkwo agreed. “It’s not just about pain. These individuals suffer from multiple organ-system complications that are physical but also psychological and societal. They experience a lot of disparities in every aspect of their lives. You’re sick, so then you can’t get a job or health insurance, you can’t get Social Security benefits. You can’t get the type of health care you need nor can you access the other forms of support you need and often you are judged as a drug seeker for complaining of pain or repeatedly seeking acute care for unresolved pain.”
Multiple factors exacerbate the experience of people living with SCD in America, the report said. “Because of systemic racism, unconscious bias, and the stigma associated with the diagnosis, the disease brings with it a much broader burden.”
Dr. Osunkwo put it this way: “SCD is a disease that mostly affects Brown and Black people, and that gets layered into the whole discrimination issues that Black and Brown people face compounding the health burden from their disease.”
The report added that “the SCD community has developed a significant lack of trust in the health care system due to the nearly universal stigma and lack of belief in their reports of pain, a lack of trust that has been further reinforced by historical events, such as the Tuskegee experiment.”
The report highlighted research that finds that Blacks “are more likely to receive a lower quality of pain management than white patients and may be perceived as having drug-seeking behavior.”
The report also identified gaps in treatment, noting that “many SCD complications are not restricted to any one organ system, and the impact of the disease on [quality of life] can be profound but hard to define and compartmentalize.”
Dr. Osunkwo said medical professionals often fail to understand the full breadth of the disease. “There’s no particular look to SCD. When you have cancer, you come in, and you look like you’re sick because you’re bald. Everyone clues into that cancer look and knows it’s lethal, that you’re may likely die early. We don’t have that “look that generates empathy” for SCD, and people don’t understand the burden on those affected. They don’t understand or appreciate that SCD shortens your lifespan as well ... that people living with SCD die 3 decades earlier than their ethnically matched peers. Also, SCD is associated with a lot of pain, and pain and the treatment of pain with opioids makes people [health care providers] uncomfortable unless it’s cancer pain.”
She added: “People also assume that, if it’s not pain, it’s not SCD even though SCD can cause leg ulcers and blood clots and even affect the tonsils, or lead to a stroke. When a disease complexity is too difficult for providers to understand, they either avoid it or don’t do anything for the patient.”
Screening and surveillance for SCD and sickle cell trait is insufficient, the report said, and the potential cost of missed childhood cases is large. Detecting the condition at birth allows the implementation of appropriate comprehensive care and treatment to prevent early death from infections and strokes. As the authors noted, “tremendous strides have been made in the past few decades in the care of children with SCD, which have led to almost all children in high-income settings surviving to adulthood.” However, there remains gaps in care coordination and follow-up of babies screened at birth and even bigger gaps in translating these life span gains to adults particularly around the period of transition from pediatrics to adult care when there appears to be a spike in morbidity and mortality.
The report summarized current treatments for SCD and noted “an influx of pipeline products” after years of little progress and identifies “a need for targeted SCD therapies that address the underlying cause of the disease.”
While treatment recommendations exist, Dr. Osunkwo said, “the evidence for them is very poor and many SCD complications have no evidence-based guidelines for providers to follow. We need more research to provide high quality evidence to make guidelines for SCD treatment stronger and more robust.”
In its final section, the report offers a “strategic plan and blueprint for sickle cell disease action.” It offers several strategies to achieve the vision of “long healthy productive lives for those living with sickle cell disease and sickle cell trait”:
- Establish and fund a research agenda to inform effective programs and policies across the life span.
- Implement efforts to advance understanding of the full impact of sickle cell trait on individuals and society.
- Address barriers to accessing current and pipeline therapies for SCD.
- Improve SCD awareness and strengthen advocacy efforts.
- Increase the number of qualified health professionals providing SCD care.
- Strengthen the evidence base for interventions and disease management and implement widespread efforts to monitor the quality of SCD care.
- Establish organized systems of care assuring both clinical and nonclinical supportive services to all persons living with SCD.
- Establish a national system to collect and link data to characterize the burden of disease, outcomes, and the needs of those with SCD across the life span.
“Right now, the average lifespan for SCD is in the mid-40s to mid-50s,” Dr. Osunkwo said. “That’s a horrible statistic. Even if we just take up half of these recommendations, people will live longer with SCD, and they’ll be more productive and contribute more to society. If we value a cancer life the same as a sickle cell life, we’ll be halfway across the finish line. But the stigma of SCD being a Black and Brown problem is going to be the hardest to confront as it requires a systemic change in our culture as a country and a health care system.”
Still, she said, the commissioning of the report “shows that there is a desire to understand the issue in better detail and try to mitigate it.”
Dr. Osunkwo and Dr. McCormick had no relevant disclosures.
SOURCE: National Academies of Sciences, Engineering, and Medicine. Addressing Sickle Cell Disease: A Strategic Plan and Blueprint for Action. Washington, D.C.: National Academies Press, 2020.
The National Academies of Science, Engineering, and Medicine have just released a 522-page report, but it’s not the usual compilation of guidelines for treatment of a disease. Instead, the authors of “Addressing Sickle Cell Disease: A Strategic Plan and Blueprint for Action” argue in stark terms that the American society has colossally failed individuals living with sickle cell disease (SCD), who are mostly Black or Brown. A dramatic overhaul of the country’s medical and societal priorities is needed to turn things around to improve health and longevity among this rare disease population.
The findings from the NASEM report are explicit: “There has been substantial success in increasing the survival of children with SCD, but this success had not been translated to similar success as they become adults.” One factor posited to contribute to the slow progress in the improvement of quality and quantity of life for adults living with this disease is the fact that “SCD is largely a disease of African Americans, and as such exists in a context of racial discrimination, health and other societal disparities, mistrust of the health care system, and the effects of poverty.” The report also cites the substantial evidence that those with SCD may receive poorer quality of care.
The report’s 14 authors were made up of an ad hoc committee formed at the request of the Department of Health & Human Services’ Office of Minority Health. The office asked NASEM to convene the committee to develop a strategic plan and blueprint for the United States and others regarding SCD.
The NASEM SCD committee members “realized that we can’t address the medical components of SCD if we don’t explore societal issues and why it’s been so hard to get good care for people with sickle cell disease,” hematologist and report coauthor Ifeyinwa (Ify) Osunkwo, MD, professor of medicine and pediatrics at Atrium Health and director of the Sickle Cell Disease Enterprise, Levine Cancer Institute, Charlotte, N.C., said in an interview. Dr. Osunkwo is also the medical editor of Hematology News.
“After almost a year of meetings and digging into the background and history of SCD care, we came out with very comprehensive summary of where we were and where we want to be,” she said. “The report provides short-, intermediate- and long-term recommendations and identifies which entity and organization should be responsible for implementing them.”
The report authors, led by pediatrician and committee chair Marie Clare McCormick, MD, of the Harvard School of Public Health, Boston, stated that about 100,000 people in the United States and millions worldwide live with SCD. The disease kills more than 700 people per year in the United States, and treatment costs an estimated $2 billion a year.
When judged by disability-adjusted life-years lost – a measurement of expected healthy years of life without an illness – the impact of SCD on individuals is estimated to be greater than a long list of other diseases such as Alzheimer’s disease, breast cancer, type 1 diabetes, and AIDS/HIV, the report noted.
“The health care needs of individuals living with SCD have been neglected by the U.S. and global health care systems, causing them and their families to suffer,” the report said. “Many of the complications that afflict individuals with SCD, particularly pain, are invisible. Pain is only diagnosed by self-reports, and in SCD there are few to no external indicators of the pain experience. Nevertheless, the pain can be excruciatingly severe and requires treatment with strong analgesics.”
There’s even more misery to the story of SCD, the report said, and Dr. Osunkwo agreed. “It’s not just about pain. These individuals suffer from multiple organ-system complications that are physical but also psychological and societal. They experience a lot of disparities in every aspect of their lives. You’re sick, so then you can’t get a job or health insurance, you can’t get Social Security benefits. You can’t get the type of health care you need nor can you access the other forms of support you need and often you are judged as a drug seeker for complaining of pain or repeatedly seeking acute care for unresolved pain.”
Multiple factors exacerbate the experience of people living with SCD in America, the report said. “Because of systemic racism, unconscious bias, and the stigma associated with the diagnosis, the disease brings with it a much broader burden.”
Dr. Osunkwo put it this way: “SCD is a disease that mostly affects Brown and Black people, and that gets layered into the whole discrimination issues that Black and Brown people face compounding the health burden from their disease.”
The report added that “the SCD community has developed a significant lack of trust in the health care system due to the nearly universal stigma and lack of belief in their reports of pain, a lack of trust that has been further reinforced by historical events, such as the Tuskegee experiment.”
The report highlighted research that finds that Blacks “are more likely to receive a lower quality of pain management than white patients and may be perceived as having drug-seeking behavior.”
The report also identified gaps in treatment, noting that “many SCD complications are not restricted to any one organ system, and the impact of the disease on [quality of life] can be profound but hard to define and compartmentalize.”
Dr. Osunkwo said medical professionals often fail to understand the full breadth of the disease. “There’s no particular look to SCD. When you have cancer, you come in, and you look like you’re sick because you’re bald. Everyone clues into that cancer look and knows it’s lethal, that you’re may likely die early. We don’t have that “look that generates empathy” for SCD, and people don’t understand the burden on those affected. They don’t understand or appreciate that SCD shortens your lifespan as well ... that people living with SCD die 3 decades earlier than their ethnically matched peers. Also, SCD is associated with a lot of pain, and pain and the treatment of pain with opioids makes people [health care providers] uncomfortable unless it’s cancer pain.”
She added: “People also assume that, if it’s not pain, it’s not SCD even though SCD can cause leg ulcers and blood clots and even affect the tonsils, or lead to a stroke. When a disease complexity is too difficult for providers to understand, they either avoid it or don’t do anything for the patient.”
Screening and surveillance for SCD and sickle cell trait is insufficient, the report said, and the potential cost of missed childhood cases is large. Detecting the condition at birth allows the implementation of appropriate comprehensive care and treatment to prevent early death from infections and strokes. As the authors noted, “tremendous strides have been made in the past few decades in the care of children with SCD, which have led to almost all children in high-income settings surviving to adulthood.” However, there remains gaps in care coordination and follow-up of babies screened at birth and even bigger gaps in translating these life span gains to adults particularly around the period of transition from pediatrics to adult care when there appears to be a spike in morbidity and mortality.
The report summarized current treatments for SCD and noted “an influx of pipeline products” after years of little progress and identifies “a need for targeted SCD therapies that address the underlying cause of the disease.”
While treatment recommendations exist, Dr. Osunkwo said, “the evidence for them is very poor and many SCD complications have no evidence-based guidelines for providers to follow. We need more research to provide high quality evidence to make guidelines for SCD treatment stronger and more robust.”
In its final section, the report offers a “strategic plan and blueprint for sickle cell disease action.” It offers several strategies to achieve the vision of “long healthy productive lives for those living with sickle cell disease and sickle cell trait”:
- Establish and fund a research agenda to inform effective programs and policies across the life span.
- Implement efforts to advance understanding of the full impact of sickle cell trait on individuals and society.
- Address barriers to accessing current and pipeline therapies for SCD.
- Improve SCD awareness and strengthen advocacy efforts.
- Increase the number of qualified health professionals providing SCD care.
- Strengthen the evidence base for interventions and disease management and implement widespread efforts to monitor the quality of SCD care.
- Establish organized systems of care assuring both clinical and nonclinical supportive services to all persons living with SCD.
- Establish a national system to collect and link data to characterize the burden of disease, outcomes, and the needs of those with SCD across the life span.
“Right now, the average lifespan for SCD is in the mid-40s to mid-50s,” Dr. Osunkwo said. “That’s a horrible statistic. Even if we just take up half of these recommendations, people will live longer with SCD, and they’ll be more productive and contribute more to society. If we value a cancer life the same as a sickle cell life, we’ll be halfway across the finish line. But the stigma of SCD being a Black and Brown problem is going to be the hardest to confront as it requires a systemic change in our culture as a country and a health care system.”
Still, she said, the commissioning of the report “shows that there is a desire to understand the issue in better detail and try to mitigate it.”
Dr. Osunkwo and Dr. McCormick had no relevant disclosures.
SOURCE: National Academies of Sciences, Engineering, and Medicine. Addressing Sickle Cell Disease: A Strategic Plan and Blueprint for Action. Washington, D.C.: National Academies Press, 2020.
The National Academies of Science, Engineering, and Medicine have just released a 522-page report, but it’s not the usual compilation of guidelines for treatment of a disease. Instead, the authors of “Addressing Sickle Cell Disease: A Strategic Plan and Blueprint for Action” argue in stark terms that the American society has colossally failed individuals living with sickle cell disease (SCD), who are mostly Black or Brown. A dramatic overhaul of the country’s medical and societal priorities is needed to turn things around to improve health and longevity among this rare disease population.
The findings from the NASEM report are explicit: “There has been substantial success in increasing the survival of children with SCD, but this success had not been translated to similar success as they become adults.” One factor posited to contribute to the slow progress in the improvement of quality and quantity of life for adults living with this disease is the fact that “SCD is largely a disease of African Americans, and as such exists in a context of racial discrimination, health and other societal disparities, mistrust of the health care system, and the effects of poverty.” The report also cites the substantial evidence that those with SCD may receive poorer quality of care.
The report’s 14 authors were made up of an ad hoc committee formed at the request of the Department of Health & Human Services’ Office of Minority Health. The office asked NASEM to convene the committee to develop a strategic plan and blueprint for the United States and others regarding SCD.
The NASEM SCD committee members “realized that we can’t address the medical components of SCD if we don’t explore societal issues and why it’s been so hard to get good care for people with sickle cell disease,” hematologist and report coauthor Ifeyinwa (Ify) Osunkwo, MD, professor of medicine and pediatrics at Atrium Health and director of the Sickle Cell Disease Enterprise, Levine Cancer Institute, Charlotte, N.C., said in an interview. Dr. Osunkwo is also the medical editor of Hematology News.
“After almost a year of meetings and digging into the background and history of SCD care, we came out with very comprehensive summary of where we were and where we want to be,” she said. “The report provides short-, intermediate- and long-term recommendations and identifies which entity and organization should be responsible for implementing them.”
The report authors, led by pediatrician and committee chair Marie Clare McCormick, MD, of the Harvard School of Public Health, Boston, stated that about 100,000 people in the United States and millions worldwide live with SCD. The disease kills more than 700 people per year in the United States, and treatment costs an estimated $2 billion a year.
When judged by disability-adjusted life-years lost – a measurement of expected healthy years of life without an illness – the impact of SCD on individuals is estimated to be greater than a long list of other diseases such as Alzheimer’s disease, breast cancer, type 1 diabetes, and AIDS/HIV, the report noted.
“The health care needs of individuals living with SCD have been neglected by the U.S. and global health care systems, causing them and their families to suffer,” the report said. “Many of the complications that afflict individuals with SCD, particularly pain, are invisible. Pain is only diagnosed by self-reports, and in SCD there are few to no external indicators of the pain experience. Nevertheless, the pain can be excruciatingly severe and requires treatment with strong analgesics.”
There’s even more misery to the story of SCD, the report said, and Dr. Osunkwo agreed. “It’s not just about pain. These individuals suffer from multiple organ-system complications that are physical but also psychological and societal. They experience a lot of disparities in every aspect of their lives. You’re sick, so then you can’t get a job or health insurance, you can’t get Social Security benefits. You can’t get the type of health care you need nor can you access the other forms of support you need and often you are judged as a drug seeker for complaining of pain or repeatedly seeking acute care for unresolved pain.”
Multiple factors exacerbate the experience of people living with SCD in America, the report said. “Because of systemic racism, unconscious bias, and the stigma associated with the diagnosis, the disease brings with it a much broader burden.”
Dr. Osunkwo put it this way: “SCD is a disease that mostly affects Brown and Black people, and that gets layered into the whole discrimination issues that Black and Brown people face compounding the health burden from their disease.”
The report added that “the SCD community has developed a significant lack of trust in the health care system due to the nearly universal stigma and lack of belief in their reports of pain, a lack of trust that has been further reinforced by historical events, such as the Tuskegee experiment.”
The report highlighted research that finds that Blacks “are more likely to receive a lower quality of pain management than white patients and may be perceived as having drug-seeking behavior.”
The report also identified gaps in treatment, noting that “many SCD complications are not restricted to any one organ system, and the impact of the disease on [quality of life] can be profound but hard to define and compartmentalize.”
Dr. Osunkwo said medical professionals often fail to understand the full breadth of the disease. “There’s no particular look to SCD. When you have cancer, you come in, and you look like you’re sick because you’re bald. Everyone clues into that cancer look and knows it’s lethal, that you’re may likely die early. We don’t have that “look that generates empathy” for SCD, and people don’t understand the burden on those affected. They don’t understand or appreciate that SCD shortens your lifespan as well ... that people living with SCD die 3 decades earlier than their ethnically matched peers. Also, SCD is associated with a lot of pain, and pain and the treatment of pain with opioids makes people [health care providers] uncomfortable unless it’s cancer pain.”
She added: “People also assume that, if it’s not pain, it’s not SCD even though SCD can cause leg ulcers and blood clots and even affect the tonsils, or lead to a stroke. When a disease complexity is too difficult for providers to understand, they either avoid it or don’t do anything for the patient.”
Screening and surveillance for SCD and sickle cell trait is insufficient, the report said, and the potential cost of missed childhood cases is large. Detecting the condition at birth allows the implementation of appropriate comprehensive care and treatment to prevent early death from infections and strokes. As the authors noted, “tremendous strides have been made in the past few decades in the care of children with SCD, which have led to almost all children in high-income settings surviving to adulthood.” However, there remains gaps in care coordination and follow-up of babies screened at birth and even bigger gaps in translating these life span gains to adults particularly around the period of transition from pediatrics to adult care when there appears to be a spike in morbidity and mortality.
The report summarized current treatments for SCD and noted “an influx of pipeline products” after years of little progress and identifies “a need for targeted SCD therapies that address the underlying cause of the disease.”
While treatment recommendations exist, Dr. Osunkwo said, “the evidence for them is very poor and many SCD complications have no evidence-based guidelines for providers to follow. We need more research to provide high quality evidence to make guidelines for SCD treatment stronger and more robust.”
In its final section, the report offers a “strategic plan and blueprint for sickle cell disease action.” It offers several strategies to achieve the vision of “long healthy productive lives for those living with sickle cell disease and sickle cell trait”:
- Establish and fund a research agenda to inform effective programs and policies across the life span.
- Implement efforts to advance understanding of the full impact of sickle cell trait on individuals and society.
- Address barriers to accessing current and pipeline therapies for SCD.
- Improve SCD awareness and strengthen advocacy efforts.
- Increase the number of qualified health professionals providing SCD care.
- Strengthen the evidence base for interventions and disease management and implement widespread efforts to monitor the quality of SCD care.
- Establish organized systems of care assuring both clinical and nonclinical supportive services to all persons living with SCD.
- Establish a national system to collect and link data to characterize the burden of disease, outcomes, and the needs of those with SCD across the life span.
“Right now, the average lifespan for SCD is in the mid-40s to mid-50s,” Dr. Osunkwo said. “That’s a horrible statistic. Even if we just take up half of these recommendations, people will live longer with SCD, and they’ll be more productive and contribute more to society. If we value a cancer life the same as a sickle cell life, we’ll be halfway across the finish line. But the stigma of SCD being a Black and Brown problem is going to be the hardest to confront as it requires a systemic change in our culture as a country and a health care system.”
Still, she said, the commissioning of the report “shows that there is a desire to understand the issue in better detail and try to mitigate it.”
Dr. Osunkwo and Dr. McCormick had no relevant disclosures.
SOURCE: National Academies of Sciences, Engineering, and Medicine. Addressing Sickle Cell Disease: A Strategic Plan and Blueprint for Action. Washington, D.C.: National Academies Press, 2020.
Promising Trends Seen in AIDS-Associated NHL
Prognoses for patients with AIDS-associated, non-Hodgkin lymphomas (AIDS-NHLs) have improved dramatically as HIV/AIDS has become easier to treat, and “we’re actually seeing patients with long-term remissions that are translating to cure,” a hematologist told colleagues at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO).
“Even those with low CD4 counts initially have more chance of survival compared to the historic patients in the pre-HAART [highly active antiretroviral therapy] era,” explained Erin Reid, MD, MS, of the University of California at San Diego Moores Cancer Center. “They’re seeing complete-response rates and overall-survival rates that are nearly matching what we’re seeing in the non-HIV lymphoma cases. And aggressive infection prophylaxis has seemed to mitigate some of the infectious complications.”
Still, Reid said, a severe form of AIDS-NHL continues to have very poor outcomes, although specific regimens appear to be brightening the picture somewhat.
According to Dr. Reid, AIDS-NHLs are the most common malignancy in the HIV-positive population, and patients with these cancers are more likely to have aggressive lymphomas. These patients are also more likely to have lymphomas associated with Epstein-Barr virus—40 to 80%, depending on the subtype of lymphoma—and Kaposi sarcoma-associated herpesvirus (also known as human herpesvirus-8, or HHV8).
“These viruses are driving these cancers, and it begs the question of whether there’s something we can do to target these viruses within these cancer cells in a way that’s therapeutic,” she said.
Compared with the non-HIV population, patients with AIDS-NHL “are much more likely to present with advanced stage, extranodal disease and central nervous system involvement,” she said.
HAART Benefits
It’s become clear that HIV control via HAART has benefits in terms of higher tolerance of chemotherapy doses—“we’re able to use more full or traditional dose regimens”—and perhaps cancer suppression too, she said. A 2013 meta-analysis “favored concurrent therapy with chemotherapy [and HAART]. This has become our recommended standard of care for virtually all cases, except the very rare ones where you cannot find a regimen that is compatible from a PK [pharmacokinetics] standpoint.”
Reid also noted that the HAART era has changed the role of CD4 counts in AIDS-NHLs. “While CD4 count still has some predictive value, its impact on mortality appears attenuated,” she said.
EPOCH Treatment
With regard to treatment, she emphasized the importance of HAART: “We would recommend concurrent HAART whenever possible with chemotherapy, or start it immediately afterward.”
Aggressive infection prophylaxis also is recommended through granulocyte colony-stimulating factor and agents to target threats from pneumocystis jiroveci pneumonia, gram negative rods, and varicella-zoster virus. “I’ve moved away from fungal prophylaxis over the years, only dealing with it if there’s a known fungal infection,” she said.
As for treatment of AIDS-NHL, Reid Suggested that research supports the EPOCH regimen --etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin. However, “we still need strategies for refractory and relapsed disease,” she said.
Reid noted that she has started to see more plasmablastic cases, although her experience is anecdotal. Plasmablastic lymphoma is much more common in the HIV-positive setting, she said.
Lifespans are poor for these patients, with many failing to live for a year. But research hints that the prognosis in AIDS-NHL patients on HAART may actually be better than in the non-HIV population, she said.
A trial published in September 2020, in fact, reports that 87% of 15 patients with AIDS-associated plasmablastic lymphoma survived for at least one year on the EPOCH regimen. Overall, the study found that “people with a collection of HIV-associated lymphomas were doing well overall with the EPOCH backbone,” Reid explained.
Reid reported no relevant disclosures.
Prognoses for patients with AIDS-associated, non-Hodgkin lymphomas (AIDS-NHLs) have improved dramatically as HIV/AIDS has become easier to treat, and “we’re actually seeing patients with long-term remissions that are translating to cure,” a hematologist told colleagues at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO).
“Even those with low CD4 counts initially have more chance of survival compared to the historic patients in the pre-HAART [highly active antiretroviral therapy] era,” explained Erin Reid, MD, MS, of the University of California at San Diego Moores Cancer Center. “They’re seeing complete-response rates and overall-survival rates that are nearly matching what we’re seeing in the non-HIV lymphoma cases. And aggressive infection prophylaxis has seemed to mitigate some of the infectious complications.”
Still, Reid said, a severe form of AIDS-NHL continues to have very poor outcomes, although specific regimens appear to be brightening the picture somewhat.
According to Dr. Reid, AIDS-NHLs are the most common malignancy in the HIV-positive population, and patients with these cancers are more likely to have aggressive lymphomas. These patients are also more likely to have lymphomas associated with Epstein-Barr virus—40 to 80%, depending on the subtype of lymphoma—and Kaposi sarcoma-associated herpesvirus (also known as human herpesvirus-8, or HHV8).
“These viruses are driving these cancers, and it begs the question of whether there’s something we can do to target these viruses within these cancer cells in a way that’s therapeutic,” she said.
Compared with the non-HIV population, patients with AIDS-NHL “are much more likely to present with advanced stage, extranodal disease and central nervous system involvement,” she said.
HAART Benefits
It’s become clear that HIV control via HAART has benefits in terms of higher tolerance of chemotherapy doses—“we’re able to use more full or traditional dose regimens”—and perhaps cancer suppression too, she said. A 2013 meta-analysis “favored concurrent therapy with chemotherapy [and HAART]. This has become our recommended standard of care for virtually all cases, except the very rare ones where you cannot find a regimen that is compatible from a PK [pharmacokinetics] standpoint.”
Reid also noted that the HAART era has changed the role of CD4 counts in AIDS-NHLs. “While CD4 count still has some predictive value, its impact on mortality appears attenuated,” she said.
EPOCH Treatment
With regard to treatment, she emphasized the importance of HAART: “We would recommend concurrent HAART whenever possible with chemotherapy, or start it immediately afterward.”
Aggressive infection prophylaxis also is recommended through granulocyte colony-stimulating factor and agents to target threats from pneumocystis jiroveci pneumonia, gram negative rods, and varicella-zoster virus. “I’ve moved away from fungal prophylaxis over the years, only dealing with it if there’s a known fungal infection,” she said.
As for treatment of AIDS-NHL, Reid Suggested that research supports the EPOCH regimen --etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin. However, “we still need strategies for refractory and relapsed disease,” she said.
Reid noted that she has started to see more plasmablastic cases, although her experience is anecdotal. Plasmablastic lymphoma is much more common in the HIV-positive setting, she said.
Lifespans are poor for these patients, with many failing to live for a year. But research hints that the prognosis in AIDS-NHL patients on HAART may actually be better than in the non-HIV population, she said.
A trial published in September 2020, in fact, reports that 87% of 15 patients with AIDS-associated plasmablastic lymphoma survived for at least one year on the EPOCH regimen. Overall, the study found that “people with a collection of HIV-associated lymphomas were doing well overall with the EPOCH backbone,” Reid explained.
Reid reported no relevant disclosures.
Prognoses for patients with AIDS-associated, non-Hodgkin lymphomas (AIDS-NHLs) have improved dramatically as HIV/AIDS has become easier to treat, and “we’re actually seeing patients with long-term remissions that are translating to cure,” a hematologist told colleagues at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO).
“Even those with low CD4 counts initially have more chance of survival compared to the historic patients in the pre-HAART [highly active antiretroviral therapy] era,” explained Erin Reid, MD, MS, of the University of California at San Diego Moores Cancer Center. “They’re seeing complete-response rates and overall-survival rates that are nearly matching what we’re seeing in the non-HIV lymphoma cases. And aggressive infection prophylaxis has seemed to mitigate some of the infectious complications.”
Still, Reid said, a severe form of AIDS-NHL continues to have very poor outcomes, although specific regimens appear to be brightening the picture somewhat.
According to Dr. Reid, AIDS-NHLs are the most common malignancy in the HIV-positive population, and patients with these cancers are more likely to have aggressive lymphomas. These patients are also more likely to have lymphomas associated with Epstein-Barr virus—40 to 80%, depending on the subtype of lymphoma—and Kaposi sarcoma-associated herpesvirus (also known as human herpesvirus-8, or HHV8).
“These viruses are driving these cancers, and it begs the question of whether there’s something we can do to target these viruses within these cancer cells in a way that’s therapeutic,” she said.
Compared with the non-HIV population, patients with AIDS-NHL “are much more likely to present with advanced stage, extranodal disease and central nervous system involvement,” she said.
HAART Benefits
It’s become clear that HIV control via HAART has benefits in terms of higher tolerance of chemotherapy doses—“we’re able to use more full or traditional dose regimens”—and perhaps cancer suppression too, she said. A 2013 meta-analysis “favored concurrent therapy with chemotherapy [and HAART]. This has become our recommended standard of care for virtually all cases, except the very rare ones where you cannot find a regimen that is compatible from a PK [pharmacokinetics] standpoint.”
Reid also noted that the HAART era has changed the role of CD4 counts in AIDS-NHLs. “While CD4 count still has some predictive value, its impact on mortality appears attenuated,” she said.
EPOCH Treatment
With regard to treatment, she emphasized the importance of HAART: “We would recommend concurrent HAART whenever possible with chemotherapy, or start it immediately afterward.”
Aggressive infection prophylaxis also is recommended through granulocyte colony-stimulating factor and agents to target threats from pneumocystis jiroveci pneumonia, gram negative rods, and varicella-zoster virus. “I’ve moved away from fungal prophylaxis over the years, only dealing with it if there’s a known fungal infection,” she said.
As for treatment of AIDS-NHL, Reid Suggested that research supports the EPOCH regimen --etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin. However, “we still need strategies for refractory and relapsed disease,” she said.
Reid noted that she has started to see more plasmablastic cases, although her experience is anecdotal. Plasmablastic lymphoma is much more common in the HIV-positive setting, she said.
Lifespans are poor for these patients, with many failing to live for a year. But research hints that the prognosis in AIDS-NHL patients on HAART may actually be better than in the non-HIV population, she said.
A trial published in September 2020, in fact, reports that 87% of 15 patients with AIDS-associated plasmablastic lymphoma survived for at least one year on the EPOCH regimen. Overall, the study found that “people with a collection of HIV-associated lymphomas were doing well overall with the EPOCH backbone,” Reid explained.
Reid reported no relevant disclosures.
One Year In, Mission Act Transforms Community Care for Veterans
One year into the implementation of the Mission Act, the US Department of Veterans Affairs (VA) continues to expand a wide-ranging third-party-administered program to boost the access of veterans to the civilian medical system, a VA official told colleagues at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO). “VA has given to the third-party administrators what they do well, which is payment of claims, and VA has taken back what we think we can do better—scheduling, care coordination, and customer service,” said Elizabeth Brill, MD, MBA, chief medical officer and senior adviser to the acting assistant undersecretary for Health for Community Care.
The 2 third-party contractors that run the Community Care Network (CCN) are Optum and TriWest. Both companies now proces patients at all VA medical facilities in the continental US. Optum serves all the states that are entirely (or mainly) in the Central and Eastern Time Zones—except for Texas—plus Puerto Rico, Washington D.C., and the US Virgin Islands. TriWest serves Texas, Hawaii, and the states that are entirely (or mainly) in the Mountain and Pacific Time Zones. The VA has not yet assigned any contractor for Alaska, Guam, American Samoa, and the Northern Mariana Islands.
“We have tried to consolidate as much as possible into the Community Care Network, pulling in a lot of services that were not previously covered, but it’s still not covering 100% of all services that veterans need outside VA medical centers,” said Brill.
Although the VA continues to rely on Veterans Care Agreements, in which health care providers contract directly with the VA, “the main focus of community care is the Community Care Network.” Said Brill. In a pair of regions—encompassing the Midwest, Northeast, and a few other states—90% of private health services are now provided through the network, she said.
One benefit of the new system is a better experience for the health care providers who work with Optum and TriWest. These administrators are responsible for finding providers and providing them with credentials based on appropriate criteria, Brill said. “In prior days, VA paid community providers directly, and some of you may be familiar with the delays that occurred in the system,” she said. “In the new system, we have third-party administrators who pay the providers quickly, and then VA pays them.”
Urgent care services are now available through CCN in most of the continental US and will be expanded to Texas, the Mountain region and the West by the end of September, she said. “We’ve been very pleased to see the response.” Meanwhile, flu shots for enrolled veterans are now available through the system via 60,000 locations.
There are requirements for private health care providers: They must meet new training requirements and submit claims within 180 days. Patients also must meet standards to get community care. For example, patients are eligible for access if the VA can’t serve them within a 30-minute drive time for primary care and mental health care and a 60-minute drive time for specialty care. Similarly, veterans are eligible if they cannot get an appointment within 20 days for primary and mental health care and 28 days for specialty care.
However, Brill insisted, the Mission Act is “not a move toward privatization. Internal VA care is just as important as external VA care,” she said. “This just gives them more choice.”
Brill also noted that there’s more to the Mission Act than expanded access. For example, an expansion of the Program of Comprehensive Assistance for Family Caregivers Program “will start kicking in this fall and the winter,” she said. It will include families of veterans from all eras of service.
The act also is designed to improve infrastructure, although the coronavirus pandemic may disrupt timing, she said. And, she added, it will strengthen the VA’s ability to recruit and keep health care providers through projects like a new scholarship program and more access to medical education debt reduction.
One year into the implementation of the Mission Act, the US Department of Veterans Affairs (VA) continues to expand a wide-ranging third-party-administered program to boost the access of veterans to the civilian medical system, a VA official told colleagues at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO). “VA has given to the third-party administrators what they do well, which is payment of claims, and VA has taken back what we think we can do better—scheduling, care coordination, and customer service,” said Elizabeth Brill, MD, MBA, chief medical officer and senior adviser to the acting assistant undersecretary for Health for Community Care.
The 2 third-party contractors that run the Community Care Network (CCN) are Optum and TriWest. Both companies now proces patients at all VA medical facilities in the continental US. Optum serves all the states that are entirely (or mainly) in the Central and Eastern Time Zones—except for Texas—plus Puerto Rico, Washington D.C., and the US Virgin Islands. TriWest serves Texas, Hawaii, and the states that are entirely (or mainly) in the Mountain and Pacific Time Zones. The VA has not yet assigned any contractor for Alaska, Guam, American Samoa, and the Northern Mariana Islands.
“We have tried to consolidate as much as possible into the Community Care Network, pulling in a lot of services that were not previously covered, but it’s still not covering 100% of all services that veterans need outside VA medical centers,” said Brill.
Although the VA continues to rely on Veterans Care Agreements, in which health care providers contract directly with the VA, “the main focus of community care is the Community Care Network.” Said Brill. In a pair of regions—encompassing the Midwest, Northeast, and a few other states—90% of private health services are now provided through the network, she said.
One benefit of the new system is a better experience for the health care providers who work with Optum and TriWest. These administrators are responsible for finding providers and providing them with credentials based on appropriate criteria, Brill said. “In prior days, VA paid community providers directly, and some of you may be familiar with the delays that occurred in the system,” she said. “In the new system, we have third-party administrators who pay the providers quickly, and then VA pays them.”
Urgent care services are now available through CCN in most of the continental US and will be expanded to Texas, the Mountain region and the West by the end of September, she said. “We’ve been very pleased to see the response.” Meanwhile, flu shots for enrolled veterans are now available through the system via 60,000 locations.
There are requirements for private health care providers: They must meet new training requirements and submit claims within 180 days. Patients also must meet standards to get community care. For example, patients are eligible for access if the VA can’t serve them within a 30-minute drive time for primary care and mental health care and a 60-minute drive time for specialty care. Similarly, veterans are eligible if they cannot get an appointment within 20 days for primary and mental health care and 28 days for specialty care.
However, Brill insisted, the Mission Act is “not a move toward privatization. Internal VA care is just as important as external VA care,” she said. “This just gives them more choice.”
Brill also noted that there’s more to the Mission Act than expanded access. For example, an expansion of the Program of Comprehensive Assistance for Family Caregivers Program “will start kicking in this fall and the winter,” she said. It will include families of veterans from all eras of service.
The act also is designed to improve infrastructure, although the coronavirus pandemic may disrupt timing, she said. And, she added, it will strengthen the VA’s ability to recruit and keep health care providers through projects like a new scholarship program and more access to medical education debt reduction.
One year into the implementation of the Mission Act, the US Department of Veterans Affairs (VA) continues to expand a wide-ranging third-party-administered program to boost the access of veterans to the civilian medical system, a VA official told colleagues at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO). “VA has given to the third-party administrators what they do well, which is payment of claims, and VA has taken back what we think we can do better—scheduling, care coordination, and customer service,” said Elizabeth Brill, MD, MBA, chief medical officer and senior adviser to the acting assistant undersecretary for Health for Community Care.
The 2 third-party contractors that run the Community Care Network (CCN) are Optum and TriWest. Both companies now proces patients at all VA medical facilities in the continental US. Optum serves all the states that are entirely (or mainly) in the Central and Eastern Time Zones—except for Texas—plus Puerto Rico, Washington D.C., and the US Virgin Islands. TriWest serves Texas, Hawaii, and the states that are entirely (or mainly) in the Mountain and Pacific Time Zones. The VA has not yet assigned any contractor for Alaska, Guam, American Samoa, and the Northern Mariana Islands.
“We have tried to consolidate as much as possible into the Community Care Network, pulling in a lot of services that were not previously covered, but it’s still not covering 100% of all services that veterans need outside VA medical centers,” said Brill.
Although the VA continues to rely on Veterans Care Agreements, in which health care providers contract directly with the VA, “the main focus of community care is the Community Care Network.” Said Brill. In a pair of regions—encompassing the Midwest, Northeast, and a few other states—90% of private health services are now provided through the network, she said.
One benefit of the new system is a better experience for the health care providers who work with Optum and TriWest. These administrators are responsible for finding providers and providing them with credentials based on appropriate criteria, Brill said. “In prior days, VA paid community providers directly, and some of you may be familiar with the delays that occurred in the system,” she said. “In the new system, we have third-party administrators who pay the providers quickly, and then VA pays them.”
Urgent care services are now available through CCN in most of the continental US and will be expanded to Texas, the Mountain region and the West by the end of September, she said. “We’ve been very pleased to see the response.” Meanwhile, flu shots for enrolled veterans are now available through the system via 60,000 locations.
There are requirements for private health care providers: They must meet new training requirements and submit claims within 180 days. Patients also must meet standards to get community care. For example, patients are eligible for access if the VA can’t serve them within a 30-minute drive time for primary care and mental health care and a 60-minute drive time for specialty care. Similarly, veterans are eligible if they cannot get an appointment within 20 days for primary and mental health care and 28 days for specialty care.
However, Brill insisted, the Mission Act is “not a move toward privatization. Internal VA care is just as important as external VA care,” she said. “This just gives them more choice.”
Brill also noted that there’s more to the Mission Act than expanded access. For example, an expansion of the Program of Comprehensive Assistance for Family Caregivers Program “will start kicking in this fall and the winter,” she said. It will include families of veterans from all eras of service.
The act also is designed to improve infrastructure, although the coronavirus pandemic may disrupt timing, she said. And, she added, it will strengthen the VA’s ability to recruit and keep health care providers through projects like a new scholarship program and more access to medical education debt reduction.
HPV-Mediated Head, Neck Cancers Predicted to Rise for Decades
Human papilloma virus (HPV)-mediated squamous cell carcinoma of the head and neck is on the rise, and the lack of herd immunity in young people will ensure growth for many years to come. “We’re really looking at another 30 to 40 years of HPV and oropharynx cancer growth,” said head and neck cancer surgeon Joseph Califano, MD, deputy director of the Moores Cancer Center at the University of California at San Diego, at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO).
Califano highlighted a 2019 study that estimated the number of diagnoses of oropharynx cancer cases in the US will grow by half to 30,000 by 2030, with the wide majority (about 25,000) in men. In 2016, the annual number of oropharynx cancer cases was 20,124. “The exponential increase in oropharynx cancer incidence in young white US men has ebbed, and modest increases are occurring/anticipated in cohorts born after 1955,” the study authors wrote.
“Currently in the United States, we don't have adequate vaccine efficiency to provide herd immunity, particularly for young boys,” said Califano. He added that although HPV vaccinations may create herd immunity in 5 to 10 years, the cancers associated with HPV can take decades to develop so a dip in rates won’t come for many years.
HPV-associated head and neck squamous cell cancer (HNSCC) affects people at a younger age when compared with other head and neck cancers—a decade or 2 earlier, according to Califano. Many patients are nonsmokers and nondrinkers, he said, and tumors may be painless and asymptomatic.
It’s also becoming clear that the HPV-associated HNSCC can strike across a widespread area of the oropharynx, including the palatine and lingual tonsils, the nasal cavity, nasopharynx, and hypopharynx (the lower part of the voice box), he said. “It has an even larger footprint than we originally supposed when we realized HPV was a dominant mechanism for development of oropharyngeal cancer,” said Califano.
Describing the extent of these cancers as an “epidemic,” Califono said a turning point in the understanding of HPV’s role in oropharynx cancers came in a “definitive” 2001 study that reported that HPV-positive patients were much more likely to develop oropharynx cancer (adjusted odds ratio, 14.4). Later research found that HPV-associated oropharynx cancers were more common than HPV-associated cervical cancer. Higher lifetime numbers of vaginal sex and oral sex partners are linked to higher risk of HPV-mediated HNSCC, he said, as is prolonged daily marijuana use.
Califano emphasized the importance of counseling patients about sexual behaviors linked to the cancers, although it’s also important to consider that “the majority of patients don’t have these risk factors.”
“The diagnosis is not an indication of infidelity or promiscuity,” he added, recalling that he saw at least one marriage dissolve because of “misunderstandings” regarding how the cancer is caused.
There are multiple treatment options. Early-stage oropharynx cancers can be treated with primary excision and staging neck dissection or radiotherapy. Multimodality therapy is appropriate for late-stage cancer and can include concurrent chemotherapy and radiation, primary excision, and treatment with concurrent cisplatinum, depending on the case. Also, “patients do really benefit if they’re enrolled in clinical trials.”
The good news is that HPV-positivity is associated with improved survival in oropharynx cancer, he said. He highlighted a 2019 study that said radiotherapy and cisplatin improve survival in HPV-positive oropharynx cancer patients. “This has become the de-facto standard of care for locally advanced, low-risk HPV-positive oropharynx cancer,” he said.
Califano reported no relevant disclosures.
Human papilloma virus (HPV)-mediated squamous cell carcinoma of the head and neck is on the rise, and the lack of herd immunity in young people will ensure growth for many years to come. “We’re really looking at another 30 to 40 years of HPV and oropharynx cancer growth,” said head and neck cancer surgeon Joseph Califano, MD, deputy director of the Moores Cancer Center at the University of California at San Diego, at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO).
Califano highlighted a 2019 study that estimated the number of diagnoses of oropharynx cancer cases in the US will grow by half to 30,000 by 2030, with the wide majority (about 25,000) in men. In 2016, the annual number of oropharynx cancer cases was 20,124. “The exponential increase in oropharynx cancer incidence in young white US men has ebbed, and modest increases are occurring/anticipated in cohorts born after 1955,” the study authors wrote.
“Currently in the United States, we don't have adequate vaccine efficiency to provide herd immunity, particularly for young boys,” said Califano. He added that although HPV vaccinations may create herd immunity in 5 to 10 years, the cancers associated with HPV can take decades to develop so a dip in rates won’t come for many years.
HPV-associated head and neck squamous cell cancer (HNSCC) affects people at a younger age when compared with other head and neck cancers—a decade or 2 earlier, according to Califano. Many patients are nonsmokers and nondrinkers, he said, and tumors may be painless and asymptomatic.
It’s also becoming clear that the HPV-associated HNSCC can strike across a widespread area of the oropharynx, including the palatine and lingual tonsils, the nasal cavity, nasopharynx, and hypopharynx (the lower part of the voice box), he said. “It has an even larger footprint than we originally supposed when we realized HPV was a dominant mechanism for development of oropharyngeal cancer,” said Califano.
Describing the extent of these cancers as an “epidemic,” Califono said a turning point in the understanding of HPV’s role in oropharynx cancers came in a “definitive” 2001 study that reported that HPV-positive patients were much more likely to develop oropharynx cancer (adjusted odds ratio, 14.4). Later research found that HPV-associated oropharynx cancers were more common than HPV-associated cervical cancer. Higher lifetime numbers of vaginal sex and oral sex partners are linked to higher risk of HPV-mediated HNSCC, he said, as is prolonged daily marijuana use.
Califano emphasized the importance of counseling patients about sexual behaviors linked to the cancers, although it’s also important to consider that “the majority of patients don’t have these risk factors.”
“The diagnosis is not an indication of infidelity or promiscuity,” he added, recalling that he saw at least one marriage dissolve because of “misunderstandings” regarding how the cancer is caused.
There are multiple treatment options. Early-stage oropharynx cancers can be treated with primary excision and staging neck dissection or radiotherapy. Multimodality therapy is appropriate for late-stage cancer and can include concurrent chemotherapy and radiation, primary excision, and treatment with concurrent cisplatinum, depending on the case. Also, “patients do really benefit if they’re enrolled in clinical trials.”
The good news is that HPV-positivity is associated with improved survival in oropharynx cancer, he said. He highlighted a 2019 study that said radiotherapy and cisplatin improve survival in HPV-positive oropharynx cancer patients. “This has become the de-facto standard of care for locally advanced, low-risk HPV-positive oropharynx cancer,” he said.
Califano reported no relevant disclosures.
Human papilloma virus (HPV)-mediated squamous cell carcinoma of the head and neck is on the rise, and the lack of herd immunity in young people will ensure growth for many years to come. “We’re really looking at another 30 to 40 years of HPV and oropharynx cancer growth,” said head and neck cancer surgeon Joseph Califano, MD, deputy director of the Moores Cancer Center at the University of California at San Diego, at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO).
Califano highlighted a 2019 study that estimated the number of diagnoses of oropharynx cancer cases in the US will grow by half to 30,000 by 2030, with the wide majority (about 25,000) in men. In 2016, the annual number of oropharynx cancer cases was 20,124. “The exponential increase in oropharynx cancer incidence in young white US men has ebbed, and modest increases are occurring/anticipated in cohorts born after 1955,” the study authors wrote.
“Currently in the United States, we don't have adequate vaccine efficiency to provide herd immunity, particularly for young boys,” said Califano. He added that although HPV vaccinations may create herd immunity in 5 to 10 years, the cancers associated with HPV can take decades to develop so a dip in rates won’t come for many years.
HPV-associated head and neck squamous cell cancer (HNSCC) affects people at a younger age when compared with other head and neck cancers—a decade or 2 earlier, according to Califano. Many patients are nonsmokers and nondrinkers, he said, and tumors may be painless and asymptomatic.
It’s also becoming clear that the HPV-associated HNSCC can strike across a widespread area of the oropharynx, including the palatine and lingual tonsils, the nasal cavity, nasopharynx, and hypopharynx (the lower part of the voice box), he said. “It has an even larger footprint than we originally supposed when we realized HPV was a dominant mechanism for development of oropharyngeal cancer,” said Califano.
Describing the extent of these cancers as an “epidemic,” Califono said a turning point in the understanding of HPV’s role in oropharynx cancers came in a “definitive” 2001 study that reported that HPV-positive patients were much more likely to develop oropharynx cancer (adjusted odds ratio, 14.4). Later research found that HPV-associated oropharynx cancers were more common than HPV-associated cervical cancer. Higher lifetime numbers of vaginal sex and oral sex partners are linked to higher risk of HPV-mediated HNSCC, he said, as is prolonged daily marijuana use.
Califano emphasized the importance of counseling patients about sexual behaviors linked to the cancers, although it’s also important to consider that “the majority of patients don’t have these risk factors.”
“The diagnosis is not an indication of infidelity or promiscuity,” he added, recalling that he saw at least one marriage dissolve because of “misunderstandings” regarding how the cancer is caused.
There are multiple treatment options. Early-stage oropharynx cancers can be treated with primary excision and staging neck dissection or radiotherapy. Multimodality therapy is appropriate for late-stage cancer and can include concurrent chemotherapy and radiation, primary excision, and treatment with concurrent cisplatinum, depending on the case. Also, “patients do really benefit if they’re enrolled in clinical trials.”
The good news is that HPV-positivity is associated with improved survival in oropharynx cancer, he said. He highlighted a 2019 study that said radiotherapy and cisplatin improve survival in HPV-positive oropharynx cancer patients. “This has become the de-facto standard of care for locally advanced, low-risk HPV-positive oropharynx cancer,” he said.
Califano reported no relevant disclosures.
COVID-19 and Blood Clots: Inside the Battle to Save Patients
Abnormal coagulation is a hallmark of COVID-19. Now, as we’re learning more about the high risk of thrombosis, physicians need to prescribe prophylaxis routinely in the hospital, stay alert, and act immediately when signs of trouble appear. “We must have a low suspicion for diagnosis and treatment of thrombosis,” said hematologist-oncologist Thomas DeLoughery, MD, professor of medicine at Oregon Health & Science University in Portland in a presentation at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO).
Still, research is sparse, and there are disagreements about the best strategies to protect patients, said DeLoughery. Physicians recognized coagulation problems early on during the course of the COVID-19 pandemic, he said, and they’re very common. According to DeLoughery, most patients have abnormal coagulation, very high D-dimer test results, and very high fibrinogen levels—even to the extraordinary level of 1,500 mg/dL, he said. And unlike in typical patients with septic shock, patients with thrombosis have a higher risk than bleeding.
A high D-dimer level is a major prognostic indicator of thrombosis and bad outcomes. “It’s representative of widespread coagulation activation, and it can be a sign of pulmonary thrombosis and local thrombosis happening at the site of the COVID infection,” he said.
DeLoughery highlighted an April 2020 study that found that “patients with D‐dimer levels ≥ 2.0 µg/mL had a higher incidence of mortality when compared with those who with D‐dimer levels < 2.0 µg/mL (12/67 vs 1/267; P < .001; hazard ratio, 51.5; 95% CI, 12.9‐206.7).”
Research also suggests that “there's something about getting COVID and going to the intensive care unit (ICU) that dramatically raises the risk of thrombosis,” he said, and the risk goes up over time in the ICU. Venous thrombosis isn’t the only risk. Relatively young patients with COVID have suffered from arterial thrombosis, even though they have minimal to no respiratory symptoms and no cardiovascular risk factors.
As for treatments, DeLoughery noted that thrombosis can occur despite standard prophylaxis, and patients may show resistance to heparin and, therefore, need massive doses. Still, there’s consensus that every patient with COVID-19 in the hospital should get thromboprophylaxis with low-molecular-weight heparin (LMWH), he said, and unfractionated heparin is appropriate for those with renal failure.
“The problem is everything else is controversial,” he said. For example, hematologists are split evenly on whether heparin dosing should be increased beyond standard protocol for patients in the ICU with 1.5 to 3 times normal D-dimers levels. He agreed with this approach but notes that some centers set their D-dimer triggers higher—at 3 to 6 times the normal level.
“The problem is that there’s limited data,” he said. “We have lots of observational studies suggesting benefits from higher doses, but we have no randomized trial data, and the observational studies are not uniform in their recommendations.”
What about outpatient prophylaxis? It appears that risk of thrombosis is < 1% percent when patients are out of the hospital, he said. “This is very reassuring that once the patient gets better, their prothrombotic drive goes away.”
Dr. DeLoughery highlighted the protocol at Oregon Health & Science University:
- Prophylaxis. Everyone with COVID-19 admitted to the hospital receives enoxaparin 40 mg daily. If the patient’s body mass index > 40, it should be increased to twice daily. For patients with renal failure, use unfractionated heparin 5000 u twice daily or enoxaparin 30 mg daily.
- In the ICU. Screen for deep vein thrombosis at admission and every 4 to 5 days thereafter. Increase enoxaparin to 40 mg twice daily, and to 1 mg/kg twice daily if signs of thrombosis develop, such as sudden deterioration, respiratory failure, the patient is too unstable to get a computed tomography, or with D-dimer > 3.0 µg/mL. “People’s thresholds for initiating empiric therapy differ, but this is an option,” he said.
For outpatient patients who are likely to be immobile for a month, 40 mg enoxaparin or 10 mg rivaroxaban are appropriate. “We’re not as aggressive as we used to be about outpatient prophylaxis,” he said.
Moving forward, he said, “this is an area where we really need clinical trials. There's just so much uncertainty.”
DeLoughery reported no disclosures.
Abnormal coagulation is a hallmark of COVID-19. Now, as we’re learning more about the high risk of thrombosis, physicians need to prescribe prophylaxis routinely in the hospital, stay alert, and act immediately when signs of trouble appear. “We must have a low suspicion for diagnosis and treatment of thrombosis,” said hematologist-oncologist Thomas DeLoughery, MD, professor of medicine at Oregon Health & Science University in Portland in a presentation at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO).
Still, research is sparse, and there are disagreements about the best strategies to protect patients, said DeLoughery. Physicians recognized coagulation problems early on during the course of the COVID-19 pandemic, he said, and they’re very common. According to DeLoughery, most patients have abnormal coagulation, very high D-dimer test results, and very high fibrinogen levels—even to the extraordinary level of 1,500 mg/dL, he said. And unlike in typical patients with septic shock, patients with thrombosis have a higher risk than bleeding.
A high D-dimer level is a major prognostic indicator of thrombosis and bad outcomes. “It’s representative of widespread coagulation activation, and it can be a sign of pulmonary thrombosis and local thrombosis happening at the site of the COVID infection,” he said.
DeLoughery highlighted an April 2020 study that found that “patients with D‐dimer levels ≥ 2.0 µg/mL had a higher incidence of mortality when compared with those who with D‐dimer levels < 2.0 µg/mL (12/67 vs 1/267; P < .001; hazard ratio, 51.5; 95% CI, 12.9‐206.7).”
Research also suggests that “there's something about getting COVID and going to the intensive care unit (ICU) that dramatically raises the risk of thrombosis,” he said, and the risk goes up over time in the ICU. Venous thrombosis isn’t the only risk. Relatively young patients with COVID have suffered from arterial thrombosis, even though they have minimal to no respiratory symptoms and no cardiovascular risk factors.
As for treatments, DeLoughery noted that thrombosis can occur despite standard prophylaxis, and patients may show resistance to heparin and, therefore, need massive doses. Still, there’s consensus that every patient with COVID-19 in the hospital should get thromboprophylaxis with low-molecular-weight heparin (LMWH), he said, and unfractionated heparin is appropriate for those with renal failure.
“The problem is everything else is controversial,” he said. For example, hematologists are split evenly on whether heparin dosing should be increased beyond standard protocol for patients in the ICU with 1.5 to 3 times normal D-dimers levels. He agreed with this approach but notes that some centers set their D-dimer triggers higher—at 3 to 6 times the normal level.
“The problem is that there’s limited data,” he said. “We have lots of observational studies suggesting benefits from higher doses, but we have no randomized trial data, and the observational studies are not uniform in their recommendations.”
What about outpatient prophylaxis? It appears that risk of thrombosis is < 1% percent when patients are out of the hospital, he said. “This is very reassuring that once the patient gets better, their prothrombotic drive goes away.”
Dr. DeLoughery highlighted the protocol at Oregon Health & Science University:
- Prophylaxis. Everyone with COVID-19 admitted to the hospital receives enoxaparin 40 mg daily. If the patient’s body mass index > 40, it should be increased to twice daily. For patients with renal failure, use unfractionated heparin 5000 u twice daily or enoxaparin 30 mg daily.
- In the ICU. Screen for deep vein thrombosis at admission and every 4 to 5 days thereafter. Increase enoxaparin to 40 mg twice daily, and to 1 mg/kg twice daily if signs of thrombosis develop, such as sudden deterioration, respiratory failure, the patient is too unstable to get a computed tomography, or with D-dimer > 3.0 µg/mL. “People’s thresholds for initiating empiric therapy differ, but this is an option,” he said.
For outpatient patients who are likely to be immobile for a month, 40 mg enoxaparin or 10 mg rivaroxaban are appropriate. “We’re not as aggressive as we used to be about outpatient prophylaxis,” he said.
Moving forward, he said, “this is an area where we really need clinical trials. There's just so much uncertainty.”
DeLoughery reported no disclosures.
Abnormal coagulation is a hallmark of COVID-19. Now, as we’re learning more about the high risk of thrombosis, physicians need to prescribe prophylaxis routinely in the hospital, stay alert, and act immediately when signs of trouble appear. “We must have a low suspicion for diagnosis and treatment of thrombosis,” said hematologist-oncologist Thomas DeLoughery, MD, professor of medicine at Oregon Health & Science University in Portland in a presentation at the virtual 2020 annual meeting of the Association of VA Hematology/Oncology (AVAHO).
Still, research is sparse, and there are disagreements about the best strategies to protect patients, said DeLoughery. Physicians recognized coagulation problems early on during the course of the COVID-19 pandemic, he said, and they’re very common. According to DeLoughery, most patients have abnormal coagulation, very high D-dimer test results, and very high fibrinogen levels—even to the extraordinary level of 1,500 mg/dL, he said. And unlike in typical patients with septic shock, patients with thrombosis have a higher risk than bleeding.
A high D-dimer level is a major prognostic indicator of thrombosis and bad outcomes. “It’s representative of widespread coagulation activation, and it can be a sign of pulmonary thrombosis and local thrombosis happening at the site of the COVID infection,” he said.
DeLoughery highlighted an April 2020 study that found that “patients with D‐dimer levels ≥ 2.0 µg/mL had a higher incidence of mortality when compared with those who with D‐dimer levels < 2.0 µg/mL (12/67 vs 1/267; P < .001; hazard ratio, 51.5; 95% CI, 12.9‐206.7).”
Research also suggests that “there's something about getting COVID and going to the intensive care unit (ICU) that dramatically raises the risk of thrombosis,” he said, and the risk goes up over time in the ICU. Venous thrombosis isn’t the only risk. Relatively young patients with COVID have suffered from arterial thrombosis, even though they have minimal to no respiratory symptoms and no cardiovascular risk factors.
As for treatments, DeLoughery noted that thrombosis can occur despite standard prophylaxis, and patients may show resistance to heparin and, therefore, need massive doses. Still, there’s consensus that every patient with COVID-19 in the hospital should get thromboprophylaxis with low-molecular-weight heparin (LMWH), he said, and unfractionated heparin is appropriate for those with renal failure.
“The problem is everything else is controversial,” he said. For example, hematologists are split evenly on whether heparin dosing should be increased beyond standard protocol for patients in the ICU with 1.5 to 3 times normal D-dimers levels. He agreed with this approach but notes that some centers set their D-dimer triggers higher—at 3 to 6 times the normal level.
“The problem is that there’s limited data,” he said. “We have lots of observational studies suggesting benefits from higher doses, but we have no randomized trial data, and the observational studies are not uniform in their recommendations.”
What about outpatient prophylaxis? It appears that risk of thrombosis is < 1% percent when patients are out of the hospital, he said. “This is very reassuring that once the patient gets better, their prothrombotic drive goes away.”
Dr. DeLoughery highlighted the protocol at Oregon Health & Science University:
- Prophylaxis. Everyone with COVID-19 admitted to the hospital receives enoxaparin 40 mg daily. If the patient’s body mass index > 40, it should be increased to twice daily. For patients with renal failure, use unfractionated heparin 5000 u twice daily or enoxaparin 30 mg daily.
- In the ICU. Screen for deep vein thrombosis at admission and every 4 to 5 days thereafter. Increase enoxaparin to 40 mg twice daily, and to 1 mg/kg twice daily if signs of thrombosis develop, such as sudden deterioration, respiratory failure, the patient is too unstable to get a computed tomography, or with D-dimer > 3.0 µg/mL. “People’s thresholds for initiating empiric therapy differ, but this is an option,” he said.
For outpatient patients who are likely to be immobile for a month, 40 mg enoxaparin or 10 mg rivaroxaban are appropriate. “We’re not as aggressive as we used to be about outpatient prophylaxis,” he said.
Moving forward, he said, “this is an area where we really need clinical trials. There's just so much uncertainty.”
DeLoughery reported no disclosures.
VA Looks to Increase Real-World Impact of Clinical Research
The US Department of Veterans Affairs (VA) is embracing clinical trials with a focus on oncology, and patients will benefit from new priorities and programs, VA officials reported at the Association of VA Hematology/Oncology (AVAHO) virtual meeting. “The whole model is one that is far more proactive,” said Carolyn Clancy, MD, Under Secretary for Health for Discovery, Education, and Affiliate Networks.
According to Clancy, the department’s top research priority is to increase veteran access to high-quality clinical trials. “Priority number 2 is increasing the real-world impact of VA research,” she said. “Our commitment to veterans and the taxpayers is to reverse and shorten the [research-to-implementation] timeline. And the third priority is to put VA data to work for veterans, not just through people who work in VA and Veterans Health Administration, but through other researchers who can have access to them.”
To meet these goals, VA is engaging in multiple research programs and collaborations. Rachel B. Ramoni, DMD, ScD, the VA chief research and development officer, highlighted a number of the projects in a separate AVAHO meeting presentation, including:
- The National Cancer Institute and VA Interagency Group to Accelerate Trials Enrollment (NAVIGATE), an interagency collaboration between the VA and the National Cancer Institute (NCI). This program established a network of sites to help enrolled veterans take part in NCI-supported clinical trials. “It really got up and running in 2018, and I’m proud to say that over 250 veterans have been enrolled, and enrollment exceeds that at non-NAVIGATE sites,” Ramoni reported. “Clearly, the additional support that these sites are getting is really helping to achieve the outcome of getting more veterans access to these trials.” However, she said, some areas of the nation aren’t yet covered by the program.
- The Precision Oncology Program for Cancer of the Prostate (POPCaP), established through a partnership with the Prostate Cancer Foundation. The foundation provided a $50 million investment. “This program ensures that veterans, no matter where they are, get best-in-class prostate cancer care,” Ramoni explained. “The initial focus was ensuring that men get sequencing if they have metastatic prostate cancer, and that they get access to clinical trials. The really distinguishing factor about POPCaP is that it has built a vibrant community of clinicians, researchers and program offices. The whole is much greater than the sum of its parts.” More POPCaP hubs are in development, she said.
- PATCH (Prostate Cancer Analysis for Therapy Choice), a program funded by the VA and the Prostate Cancer Foundation. “The whole purpose of PATCH is to create this network of sites to systematically go through different clinical trials that are biomarker-driven,” Ramoni said. “One of the great things about PATCH is that it’s leveraging the genetics databases to help proactively identify men who might qualify for these trials and to find them wherever they might be across the system so they have access to these trials.” She also praised the program’s commitment to collaboration and mentorship. “If you’re new to putting together clinical trials concepts or to submitting merit proposals to VA for funding, PATCH is a great place to get into a community that’s supportive and wants to help you succeed.”
- The VA Phenomics Library. This library, based at the Boston VA Medical Center, focuses on improving the analysis of “messy” electronic health record data, Ramoni noted. “There are automated algorithms that go through and help you clean up that data to make sense of it,” she said. “The problem is that it’s really been an every-person-for-himself-or-herself system. Each researcher who needed these phenotypes was creating his or her own.” The Phenomics Library will promote sharing “so there’s not going to be as much wasted time duplicating effort,” she said. “By the end of fiscal year 2021, we will have over 1,000 curated phenotypes in there. We hope that will be a great resource for the oncology community as well as many other communities.”
- Access to Clinical Trials (ACT) for Veterans. “This program, which began a couple of years ago, has really succeeded,” Ramoni said. “We were focusing on decreasing the time it takes to start up multi-site industry trials. When we got started with ACT, it was taking over 200 days to get started. And now, just a couple years later, we are well under 100 days, which is within industry standards.” Also, she said, the VA established a Partnered Research Program office, “which serves to interact with our industry partners and really guide them through the VA system, which can be complex if you’re approaching it for the first time.”
In a separate presentation, Krissa Caroff, MS, CPC, program manager of the Partnered Research Program, said it had quickened the process of implementing clinical trials by tackling roadblocks such as the need for multiple master agreements to be signed. Central coordination has been key, she said, “and we are working closely to ensure that we when have a multisite trial, all the VA sites are utilizing the same single IRB [institutional review board]. We’ve also identified the critical information that we need to collect from industry in order for us to evaluate a trial.”
What’s next? “We really are going to be focusing on oncology trials,” Ramoni insisted. “This is a high priority for us.” She added: “Please share your feedback and experiences with us. And also please communicate amongst your colleagues within your organization to explain how we’re standardizing things within VA.”
The speakers reported no relevant disclosures.
The US Department of Veterans Affairs (VA) is embracing clinical trials with a focus on oncology, and patients will benefit from new priorities and programs, VA officials reported at the Association of VA Hematology/Oncology (AVAHO) virtual meeting. “The whole model is one that is far more proactive,” said Carolyn Clancy, MD, Under Secretary for Health for Discovery, Education, and Affiliate Networks.
According to Clancy, the department’s top research priority is to increase veteran access to high-quality clinical trials. “Priority number 2 is increasing the real-world impact of VA research,” she said. “Our commitment to veterans and the taxpayers is to reverse and shorten the [research-to-implementation] timeline. And the third priority is to put VA data to work for veterans, not just through people who work in VA and Veterans Health Administration, but through other researchers who can have access to them.”
To meet these goals, VA is engaging in multiple research programs and collaborations. Rachel B. Ramoni, DMD, ScD, the VA chief research and development officer, highlighted a number of the projects in a separate AVAHO meeting presentation, including:
- The National Cancer Institute and VA Interagency Group to Accelerate Trials Enrollment (NAVIGATE), an interagency collaboration between the VA and the National Cancer Institute (NCI). This program established a network of sites to help enrolled veterans take part in NCI-supported clinical trials. “It really got up and running in 2018, and I’m proud to say that over 250 veterans have been enrolled, and enrollment exceeds that at non-NAVIGATE sites,” Ramoni reported. “Clearly, the additional support that these sites are getting is really helping to achieve the outcome of getting more veterans access to these trials.” However, she said, some areas of the nation aren’t yet covered by the program.
- The Precision Oncology Program for Cancer of the Prostate (POPCaP), established through a partnership with the Prostate Cancer Foundation. The foundation provided a $50 million investment. “This program ensures that veterans, no matter where they are, get best-in-class prostate cancer care,” Ramoni explained. “The initial focus was ensuring that men get sequencing if they have metastatic prostate cancer, and that they get access to clinical trials. The really distinguishing factor about POPCaP is that it has built a vibrant community of clinicians, researchers and program offices. The whole is much greater than the sum of its parts.” More POPCaP hubs are in development, she said.
- PATCH (Prostate Cancer Analysis for Therapy Choice), a program funded by the VA and the Prostate Cancer Foundation. “The whole purpose of PATCH is to create this network of sites to systematically go through different clinical trials that are biomarker-driven,” Ramoni said. “One of the great things about PATCH is that it’s leveraging the genetics databases to help proactively identify men who might qualify for these trials and to find them wherever they might be across the system so they have access to these trials.” She also praised the program’s commitment to collaboration and mentorship. “If you’re new to putting together clinical trials concepts or to submitting merit proposals to VA for funding, PATCH is a great place to get into a community that’s supportive and wants to help you succeed.”
- The VA Phenomics Library. This library, based at the Boston VA Medical Center, focuses on improving the analysis of “messy” electronic health record data, Ramoni noted. “There are automated algorithms that go through and help you clean up that data to make sense of it,” she said. “The problem is that it’s really been an every-person-for-himself-or-herself system. Each researcher who needed these phenotypes was creating his or her own.” The Phenomics Library will promote sharing “so there’s not going to be as much wasted time duplicating effort,” she said. “By the end of fiscal year 2021, we will have over 1,000 curated phenotypes in there. We hope that will be a great resource for the oncology community as well as many other communities.”
- Access to Clinical Trials (ACT) for Veterans. “This program, which began a couple of years ago, has really succeeded,” Ramoni said. “We were focusing on decreasing the time it takes to start up multi-site industry trials. When we got started with ACT, it was taking over 200 days to get started. And now, just a couple years later, we are well under 100 days, which is within industry standards.” Also, she said, the VA established a Partnered Research Program office, “which serves to interact with our industry partners and really guide them through the VA system, which can be complex if you’re approaching it for the first time.”
In a separate presentation, Krissa Caroff, MS, CPC, program manager of the Partnered Research Program, said it had quickened the process of implementing clinical trials by tackling roadblocks such as the need for multiple master agreements to be signed. Central coordination has been key, she said, “and we are working closely to ensure that we when have a multisite trial, all the VA sites are utilizing the same single IRB [institutional review board]. We’ve also identified the critical information that we need to collect from industry in order for us to evaluate a trial.”
What’s next? “We really are going to be focusing on oncology trials,” Ramoni insisted. “This is a high priority for us.” She added: “Please share your feedback and experiences with us. And also please communicate amongst your colleagues within your organization to explain how we’re standardizing things within VA.”
The speakers reported no relevant disclosures.
The US Department of Veterans Affairs (VA) is embracing clinical trials with a focus on oncology, and patients will benefit from new priorities and programs, VA officials reported at the Association of VA Hematology/Oncology (AVAHO) virtual meeting. “The whole model is one that is far more proactive,” said Carolyn Clancy, MD, Under Secretary for Health for Discovery, Education, and Affiliate Networks.
According to Clancy, the department’s top research priority is to increase veteran access to high-quality clinical trials. “Priority number 2 is increasing the real-world impact of VA research,” she said. “Our commitment to veterans and the taxpayers is to reverse and shorten the [research-to-implementation] timeline. And the third priority is to put VA data to work for veterans, not just through people who work in VA and Veterans Health Administration, but through other researchers who can have access to them.”
To meet these goals, VA is engaging in multiple research programs and collaborations. Rachel B. Ramoni, DMD, ScD, the VA chief research and development officer, highlighted a number of the projects in a separate AVAHO meeting presentation, including:
- The National Cancer Institute and VA Interagency Group to Accelerate Trials Enrollment (NAVIGATE), an interagency collaboration between the VA and the National Cancer Institute (NCI). This program established a network of sites to help enrolled veterans take part in NCI-supported clinical trials. “It really got up and running in 2018, and I’m proud to say that over 250 veterans have been enrolled, and enrollment exceeds that at non-NAVIGATE sites,” Ramoni reported. “Clearly, the additional support that these sites are getting is really helping to achieve the outcome of getting more veterans access to these trials.” However, she said, some areas of the nation aren’t yet covered by the program.
- The Precision Oncology Program for Cancer of the Prostate (POPCaP), established through a partnership with the Prostate Cancer Foundation. The foundation provided a $50 million investment. “This program ensures that veterans, no matter where they are, get best-in-class prostate cancer care,” Ramoni explained. “The initial focus was ensuring that men get sequencing if they have metastatic prostate cancer, and that they get access to clinical trials. The really distinguishing factor about POPCaP is that it has built a vibrant community of clinicians, researchers and program offices. The whole is much greater than the sum of its parts.” More POPCaP hubs are in development, she said.
- PATCH (Prostate Cancer Analysis for Therapy Choice), a program funded by the VA and the Prostate Cancer Foundation. “The whole purpose of PATCH is to create this network of sites to systematically go through different clinical trials that are biomarker-driven,” Ramoni said. “One of the great things about PATCH is that it’s leveraging the genetics databases to help proactively identify men who might qualify for these trials and to find them wherever they might be across the system so they have access to these trials.” She also praised the program’s commitment to collaboration and mentorship. “If you’re new to putting together clinical trials concepts or to submitting merit proposals to VA for funding, PATCH is a great place to get into a community that’s supportive and wants to help you succeed.”
- The VA Phenomics Library. This library, based at the Boston VA Medical Center, focuses on improving the analysis of “messy” electronic health record data, Ramoni noted. “There are automated algorithms that go through and help you clean up that data to make sense of it,” she said. “The problem is that it’s really been an every-person-for-himself-or-herself system. Each researcher who needed these phenotypes was creating his or her own.” The Phenomics Library will promote sharing “so there’s not going to be as much wasted time duplicating effort,” she said. “By the end of fiscal year 2021, we will have over 1,000 curated phenotypes in there. We hope that will be a great resource for the oncology community as well as many other communities.”
- Access to Clinical Trials (ACT) for Veterans. “This program, which began a couple of years ago, has really succeeded,” Ramoni said. “We were focusing on decreasing the time it takes to start up multi-site industry trials. When we got started with ACT, it was taking over 200 days to get started. And now, just a couple years later, we are well under 100 days, which is within industry standards.” Also, she said, the VA established a Partnered Research Program office, “which serves to interact with our industry partners and really guide them through the VA system, which can be complex if you’re approaching it for the first time.”
In a separate presentation, Krissa Caroff, MS, CPC, program manager of the Partnered Research Program, said it had quickened the process of implementing clinical trials by tackling roadblocks such as the need for multiple master agreements to be signed. Central coordination has been key, she said, “and we are working closely to ensure that we when have a multisite trial, all the VA sites are utilizing the same single IRB [institutional review board]. We’ve also identified the critical information that we need to collect from industry in order for us to evaluate a trial.”
What’s next? “We really are going to be focusing on oncology trials,” Ramoni insisted. “This is a high priority for us.” She added: “Please share your feedback and experiences with us. And also please communicate amongst your colleagues within your organization to explain how we’re standardizing things within VA.”
The speakers reported no relevant disclosures.