User login
Olanzapine Weight Gain May Start in the Brain
Olanzapine apparently alters the way the brain anticipates food and experiences the reward of eating – changes that might predispose patients taking the drug to rapid weight gain, a study has shown.
Healthy, normal-weight volunteers who took the drug for a week gained a mean of 1 kg – a significant change from baseline. Brain imaging showed altered activity in areas involved with anticipation, reward response, and satiety, Dr. Jose Mathews and his colleagues reported in the August issue of Archives of General Psychiatry (doi:10.1001/archgenpsychiatry.2012.934).
"Interestingly, subjects’ perception of hunger did not change significantly after olanzapine treatment nor did their perception of the pleasantness," of morning food intake, wrote Dr. Mathews of Washington University in St Louis and his coauthors. "These findings contrast to the actual behavior of the subjects ... as they consumed significantly more breakfast and gained weight after treatment with olanzapine."
The study comprised 19 young people (mean age, 27 years) with a mean body mass index of 25.78 kg/m2. On the first night, they took 5 mg of olanzapine. They took 10 mg each night for the next 6 nights.
Each night, all of the volunteers stayed at the investigation site. Every morning, they had a functional magnetic resonance imaging session during which they saw images of chocolate milk, tomato juice, or water. After identifying which drink they preferred, they received a small portion through a tube, then rated their choices (very pleasant, pleasant, neutral, unpleasant, and very unpleasant) on a 5-point Likert scale.
After the scan, they consumed a breakfast of the liquid they had chosen and were instructed to drink until they were no longer hungry. Then all were rescanned.
A significant increase was found in the mean amount of breakfast consumed, from about 25 ounces at baseline to about 32 ounces by day 7. The subjects gained a mean of 1.1 kg over the course of the 1-week study.
They also showed a significant increase in the disinhibited eating subscale of the Three-Factor Eating Questionnaire. However, no changes were found in their feeling of hunger or in how pleasant they perceived their drink to be.
Brain imaging, however, showed definite changes in regions involved with anticipation and reward, the authors said.
"In the caudate and putamen, activation to the experience of rewarding taste increased after olanzapine treatment while the response to [water] decreased," the investigators wrote. The drug also affected the inferior frontal cortex, striatum, and anterior cingulate. "All these regions showed enhanced responses to cues predicting rewarding liquids after olanzapine, while there was a decrease in activation elicited by the picture of the [water]."
They also observed decreased activity in the lateral orbital frontal cortex, a region involved in satiety. "These fMRI changes suggested an enhanced anticipatory desire for food, an enhanced reward experience of [eating], and a compromised satiety-related mechanism. This pattern of change after treatment with olanzapine provides a plausible set of mechanisms that may contribute to the weight gain commonly associated with this medication."
The investigators cited several limitations of the study. The absence of a placebo control group limited their ability to show conclusively that the observed changes were tied to olanzapine treatment. Also, the subjects’ unmedicated scan always came before the scan with olanzapine, which means "it is possible that habituation effects could be confounded with the olanzapine effects."
Despite these limitations, Dr. Mathews and his colleagues said they think their findings might "pave the way for targeted treatments that may help dial down the enhanced reward value of food while strengthening the inhibitory circuits that control food intake."
The study was sponsored by the National Alliance for Research on Schizophrenia and Depression, and the National Institutes of Health. Dr. Mathews has been on the speakers bureaus of Janssen Pharmaceuticals and Bristol-Myers Squibb, but had no current financial disclosures.
Olanzapine apparently alters the way the brain anticipates food and experiences the reward of eating – changes that might predispose patients taking the drug to rapid weight gain, a study has shown.
Healthy, normal-weight volunteers who took the drug for a week gained a mean of 1 kg – a significant change from baseline. Brain imaging showed altered activity in areas involved with anticipation, reward response, and satiety, Dr. Jose Mathews and his colleagues reported in the August issue of Archives of General Psychiatry (doi:10.1001/archgenpsychiatry.2012.934).
"Interestingly, subjects’ perception of hunger did not change significantly after olanzapine treatment nor did their perception of the pleasantness," of morning food intake, wrote Dr. Mathews of Washington University in St Louis and his coauthors. "These findings contrast to the actual behavior of the subjects ... as they consumed significantly more breakfast and gained weight after treatment with olanzapine."
The study comprised 19 young people (mean age, 27 years) with a mean body mass index of 25.78 kg/m2. On the first night, they took 5 mg of olanzapine. They took 10 mg each night for the next 6 nights.
Each night, all of the volunteers stayed at the investigation site. Every morning, they had a functional magnetic resonance imaging session during which they saw images of chocolate milk, tomato juice, or water. After identifying which drink they preferred, they received a small portion through a tube, then rated their choices (very pleasant, pleasant, neutral, unpleasant, and very unpleasant) on a 5-point Likert scale.
After the scan, they consumed a breakfast of the liquid they had chosen and were instructed to drink until they were no longer hungry. Then all were rescanned.
A significant increase was found in the mean amount of breakfast consumed, from about 25 ounces at baseline to about 32 ounces by day 7. The subjects gained a mean of 1.1 kg over the course of the 1-week study.
They also showed a significant increase in the disinhibited eating subscale of the Three-Factor Eating Questionnaire. However, no changes were found in their feeling of hunger or in how pleasant they perceived their drink to be.
Brain imaging, however, showed definite changes in regions involved with anticipation and reward, the authors said.
"In the caudate and putamen, activation to the experience of rewarding taste increased after olanzapine treatment while the response to [water] decreased," the investigators wrote. The drug also affected the inferior frontal cortex, striatum, and anterior cingulate. "All these regions showed enhanced responses to cues predicting rewarding liquids after olanzapine, while there was a decrease in activation elicited by the picture of the [water]."
They also observed decreased activity in the lateral orbital frontal cortex, a region involved in satiety. "These fMRI changes suggested an enhanced anticipatory desire for food, an enhanced reward experience of [eating], and a compromised satiety-related mechanism. This pattern of change after treatment with olanzapine provides a plausible set of mechanisms that may contribute to the weight gain commonly associated with this medication."
The investigators cited several limitations of the study. The absence of a placebo control group limited their ability to show conclusively that the observed changes were tied to olanzapine treatment. Also, the subjects’ unmedicated scan always came before the scan with olanzapine, which means "it is possible that habituation effects could be confounded with the olanzapine effects."
Despite these limitations, Dr. Mathews and his colleagues said they think their findings might "pave the way for targeted treatments that may help dial down the enhanced reward value of food while strengthening the inhibitory circuits that control food intake."
The study was sponsored by the National Alliance for Research on Schizophrenia and Depression, and the National Institutes of Health. Dr. Mathews has been on the speakers bureaus of Janssen Pharmaceuticals and Bristol-Myers Squibb, but had no current financial disclosures.
Olanzapine apparently alters the way the brain anticipates food and experiences the reward of eating – changes that might predispose patients taking the drug to rapid weight gain, a study has shown.
Healthy, normal-weight volunteers who took the drug for a week gained a mean of 1 kg – a significant change from baseline. Brain imaging showed altered activity in areas involved with anticipation, reward response, and satiety, Dr. Jose Mathews and his colleagues reported in the August issue of Archives of General Psychiatry (doi:10.1001/archgenpsychiatry.2012.934).
"Interestingly, subjects’ perception of hunger did not change significantly after olanzapine treatment nor did their perception of the pleasantness," of morning food intake, wrote Dr. Mathews of Washington University in St Louis and his coauthors. "These findings contrast to the actual behavior of the subjects ... as they consumed significantly more breakfast and gained weight after treatment with olanzapine."
The study comprised 19 young people (mean age, 27 years) with a mean body mass index of 25.78 kg/m2. On the first night, they took 5 mg of olanzapine. They took 10 mg each night for the next 6 nights.
Each night, all of the volunteers stayed at the investigation site. Every morning, they had a functional magnetic resonance imaging session during which they saw images of chocolate milk, tomato juice, or water. After identifying which drink they preferred, they received a small portion through a tube, then rated their choices (very pleasant, pleasant, neutral, unpleasant, and very unpleasant) on a 5-point Likert scale.
After the scan, they consumed a breakfast of the liquid they had chosen and were instructed to drink until they were no longer hungry. Then all were rescanned.
A significant increase was found in the mean amount of breakfast consumed, from about 25 ounces at baseline to about 32 ounces by day 7. The subjects gained a mean of 1.1 kg over the course of the 1-week study.
They also showed a significant increase in the disinhibited eating subscale of the Three-Factor Eating Questionnaire. However, no changes were found in their feeling of hunger or in how pleasant they perceived their drink to be.
Brain imaging, however, showed definite changes in regions involved with anticipation and reward, the authors said.
"In the caudate and putamen, activation to the experience of rewarding taste increased after olanzapine treatment while the response to [water] decreased," the investigators wrote. The drug also affected the inferior frontal cortex, striatum, and anterior cingulate. "All these regions showed enhanced responses to cues predicting rewarding liquids after olanzapine, while there was a decrease in activation elicited by the picture of the [water]."
They also observed decreased activity in the lateral orbital frontal cortex, a region involved in satiety. "These fMRI changes suggested an enhanced anticipatory desire for food, an enhanced reward experience of [eating], and a compromised satiety-related mechanism. This pattern of change after treatment with olanzapine provides a plausible set of mechanisms that may contribute to the weight gain commonly associated with this medication."
The investigators cited several limitations of the study. The absence of a placebo control group limited their ability to show conclusively that the observed changes were tied to olanzapine treatment. Also, the subjects’ unmedicated scan always came before the scan with olanzapine, which means "it is possible that habituation effects could be confounded with the olanzapine effects."
Despite these limitations, Dr. Mathews and his colleagues said they think their findings might "pave the way for targeted treatments that may help dial down the enhanced reward value of food while strengthening the inhibitory circuits that control food intake."
The study was sponsored by the National Alliance for Research on Schizophrenia and Depression, and the National Institutes of Health. Dr. Mathews has been on the speakers bureaus of Janssen Pharmaceuticals and Bristol-Myers Squibb, but had no current financial disclosures.
FROM ARCHIVES OF GENERAL PSYCHIATRY
Major Finding: Normal-weight subjects who took olanzapine for 1 week gained a mean of 1 kg, and showed functional changes in brain regions involved with anticipation, reward, and satiety.
Data Source: The open-label observational study involved 19 healthy young adults.
Disclosures: The study was sponsored by the National Alliance for Research on Schizophrenia and Depression, and the National Institutes of Health. Dr. Mathews has been on the speakers bureaus of Janssen Pharmaceuticals and Bristol-Myers Squibb, but had no current financial disclosures.
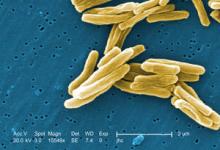
Resistance to Second-Line TB Drugs Rises
Nearly 44% of multidrug-resistant tuberculosis cases tested in eight countries were also resistant to at least one second-line tuberculosis drug, according to results of an international prospective cohort study.
Extensively drug-resistant (XDR) strains were unexpectedly prevalent as well, particularly in South Korea and Russia, reported Tracy Dalton, Ph.D., of the Centers for Disease Control and Prevention, Atlanta, and colleagues. The report was published in the Aug. 30 online issue of The Lancet.
These XDR isolates were detected in 6.7% of patients overall, with prevalence in South Korea (15%) and Russia (11%) exceeding the current World Health Organization global estimate (9.4%). The risk of XDR disease was four times greater in previously treated patients, and previous treatment with second-line drugs was consistently the strongest risk factor for resistance to these drugs (Lancet 2012 Aug. 30 [http://dx.doi.org/10.1016/S0140-6736(12)60734-X]).
Multidrug-resistant (MDR) tuberculosis is resistant to at least rifampicin and isoniazid, and accounts for 3.6%-4.8% of new tuberculosis cases worldwide. XDR tuberculosis is resistant to at least rifampicin, isoniazid, and one or more of the second-line antituberculosis drugs. XDR tuberculosis has been reported in 77 countries.
While the numbers varied between nations, investigators with the international Preserving Effective TB Treatment Study (PETTS) saw a concerning pattern: The prevalence of drug-resistant strains correlated with the time that the second-line drugs had become available through the Green Light Committee, a World Health Organization program designed to increase access to second-line antituberculosis agents.
"[Second-line drugs] had been available for 10 years or less in Thailand (7 years), the Philippines (9 years), and Peru (10 years), and these countries had the lowest rates of resistance," wrote Dr. Dalton of the Centers for Disease Control and Prevention. "By contrast, South Korea and Russia had the longest histories of availability (more than 20 years) and the highest rates of resistance."
PETTS was launched in 2003 to determine the risk factors for and frequency of acquired resistance to second-line therapies in people with MDR tuberculosis. In 2005, in light of burgeoning numbers, the program was modified to include data on people with XDR tuberculosis.
The current report focused on eight countries: Estonia, Latvia, Peru, the Philippines, Russia, South Africa, South Korea, and Thailand. Samples from patients with MDR tuberculosis were obtained from large clinical centers in each country during 2005-2008. Inclusion criteria were at least 30 days of treatment with a second-line antituberculosis drug, with sputum collection within 30 days before or after the initiation of therapy.
Of 1,540 isolates tested, 1,278 (83%) were MDR. Most of those patients (94%) had a history of tuberculosis, and of those, 71% had experienced it at least once before the tested case.
Of the entire group, 93% had received first-line therapy, but only 15% had received second-line drugs. South Africa had the lowest rate of second-line treatment (3%), while South Korea had the highest (54%).
The overall prevalence of resistance to any second-line drug was 43.7%, but the rate varied among the countries, from 33% in Thailand to 62% in Latvia.
Overall, 20% of isolates were resistant to at least one second-line injectable drug, ranging from 2% in the Philippines to 47% in Latvia. The Philippines also had the lowest prevalence of resistance to all injectables (0.3%), while South Africa had the highest (26%).
The overall resistance rate to at least one second-line oral drug was 27%. Resistance to at least one oral drug ranged from 13% in Estonia to 38% in Latvia; however, other countries also had a high prevalence, including South Korea (36%), the Philippines (32%), Russia (26%), and South Africa (22%).
A total of 6.7% of the isolates were XDR, with the highest prevalence in South Korea (15%) and the lowest in the Philippines (0.8%).
Prior treatment for MDR strains with the second-line drugs was the strongest risk factor for XDR tuberculosis, with a relative risk of 4.75 for injectables and 4 for oral medications.
Although countries with Green Light projects did have more cases, the risks ratios for different resistance types did not reflect the actual numbers, the authors noted.
Resistance to fluoroquinolones and second-line injectable drugs – but not to other oral second-line drugs – was significantly less prevalent in countries that had Green Light Committee approved projects. "This difference was due to the very low prevalence of resistance to second-line drugs in the Philippines, which had the largest Green Light Committee project," the authors said.
The individualized numbers should be useful to national disease management efforts, the researchers added. "Our country-specific results can be extrapolated to guide in-country policy for laboratory capacity and for designing effective treatment recommendations."
PETTS data collection continues, they noted. "The effect of the Green Light Committee initiative in combating acquired resistance to second-line drugs, the timing of acquired resistance, and the role of specific genetic mutations in different regions of the world are also being assessed."
The U.S. Agency for International Development, the Centers for Disease Control and Prevention, the National Institutes of Health, and the Korean Ministry of Health and Welfare sponsored the study. The authors declared no financial conflicts.
To be truly effective, tuberculosis treatment guidelines should reflect every country’s unique patterns of transmissibility and drug resistance, Sven Hoffner, Ph.D., wrote in an accompanying editorial (Lancet 2012 Aug. 30 [http://dx.doi.org/10.1016/ S0140-6736(12)61069-1]).
"Dalton and colleagues’ study increases awareness of the clinical and public health issues caused by resistant M. tuberculosis and reveals differences in prevalence and risk factors between countries and settings," wrote Dr. Hoffner of the Swedish Institute for Communicable Disease Control. "Hopefully, these findings will contribute to the identification of the tools needed for optimum control of multidrug-resistant tuberculosis in specific epidemiological settings."
While prior tuberculosis treatment remains the biggest risk factor for resistant infections, other risks vary widely among countries, depending not only on local sociodemographic factors, but also on a global inability to quantify strains that have become extensively drug resistant.
"These results show that extensively drug-resistant tuberculosis is high. Nevertheless, the information remains insufficient to give a clear view of the worldwide distributions and true magnitude of XDR tuberculosis, even more so for the most severely resistant cases," Dr. Hoffner noted. "Updated information on multidrug-resistant tuberculosis and investigation of the trends are urgently needed, especially since the true scale of the burden of multi- and extensively-resistant tuberculosis might be underestimated and seems to be rapidly increasing."
Dr. Hoffner is chief microbiologist at the Swedish Institute for Communicable Disease Control, Solna. He had no financial disclosures.
To be truly effective, tuberculosis treatment guidelines should reflect every country’s unique patterns of transmissibility and drug resistance, Sven Hoffner, Ph.D., wrote in an accompanying editorial (Lancet 2012 Aug. 30 [http://dx.doi.org/10.1016/ S0140-6736(12)61069-1]).
"Dalton and colleagues’ study increases awareness of the clinical and public health issues caused by resistant M. tuberculosis and reveals differences in prevalence and risk factors between countries and settings," wrote Dr. Hoffner of the Swedish Institute for Communicable Disease Control. "Hopefully, these findings will contribute to the identification of the tools needed for optimum control of multidrug-resistant tuberculosis in specific epidemiological settings."
While prior tuberculosis treatment remains the biggest risk factor for resistant infections, other risks vary widely among countries, depending not only on local sociodemographic factors, but also on a global inability to quantify strains that have become extensively drug resistant.
"These results show that extensively drug-resistant tuberculosis is high. Nevertheless, the information remains insufficient to give a clear view of the worldwide distributions and true magnitude of XDR tuberculosis, even more so for the most severely resistant cases," Dr. Hoffner noted. "Updated information on multidrug-resistant tuberculosis and investigation of the trends are urgently needed, especially since the true scale of the burden of multi- and extensively-resistant tuberculosis might be underestimated and seems to be rapidly increasing."
Dr. Hoffner is chief microbiologist at the Swedish Institute for Communicable Disease Control, Solna. He had no financial disclosures.
To be truly effective, tuberculosis treatment guidelines should reflect every country’s unique patterns of transmissibility and drug resistance, Sven Hoffner, Ph.D., wrote in an accompanying editorial (Lancet 2012 Aug. 30 [http://dx.doi.org/10.1016/ S0140-6736(12)61069-1]).
"Dalton and colleagues’ study increases awareness of the clinical and public health issues caused by resistant M. tuberculosis and reveals differences in prevalence and risk factors between countries and settings," wrote Dr. Hoffner of the Swedish Institute for Communicable Disease Control. "Hopefully, these findings will contribute to the identification of the tools needed for optimum control of multidrug-resistant tuberculosis in specific epidemiological settings."
While prior tuberculosis treatment remains the biggest risk factor for resistant infections, other risks vary widely among countries, depending not only on local sociodemographic factors, but also on a global inability to quantify strains that have become extensively drug resistant.
"These results show that extensively drug-resistant tuberculosis is high. Nevertheless, the information remains insufficient to give a clear view of the worldwide distributions and true magnitude of XDR tuberculosis, even more so for the most severely resistant cases," Dr. Hoffner noted. "Updated information on multidrug-resistant tuberculosis and investigation of the trends are urgently needed, especially since the true scale of the burden of multi- and extensively-resistant tuberculosis might be underestimated and seems to be rapidly increasing."
Dr. Hoffner is chief microbiologist at the Swedish Institute for Communicable Disease Control, Solna. He had no financial disclosures.
Nearly 44% of multidrug-resistant tuberculosis cases tested in eight countries were also resistant to at least one second-line tuberculosis drug, according to results of an international prospective cohort study.
Extensively drug-resistant (XDR) strains were unexpectedly prevalent as well, particularly in South Korea and Russia, reported Tracy Dalton, Ph.D., of the Centers for Disease Control and Prevention, Atlanta, and colleagues. The report was published in the Aug. 30 online issue of The Lancet.
These XDR isolates were detected in 6.7% of patients overall, with prevalence in South Korea (15%) and Russia (11%) exceeding the current World Health Organization global estimate (9.4%). The risk of XDR disease was four times greater in previously treated patients, and previous treatment with second-line drugs was consistently the strongest risk factor for resistance to these drugs (Lancet 2012 Aug. 30 [http://dx.doi.org/10.1016/S0140-6736(12)60734-X]).
Multidrug-resistant (MDR) tuberculosis is resistant to at least rifampicin and isoniazid, and accounts for 3.6%-4.8% of new tuberculosis cases worldwide. XDR tuberculosis is resistant to at least rifampicin, isoniazid, and one or more of the second-line antituberculosis drugs. XDR tuberculosis has been reported in 77 countries.
While the numbers varied between nations, investigators with the international Preserving Effective TB Treatment Study (PETTS) saw a concerning pattern: The prevalence of drug-resistant strains correlated with the time that the second-line drugs had become available through the Green Light Committee, a World Health Organization program designed to increase access to second-line antituberculosis agents.
"[Second-line drugs] had been available for 10 years or less in Thailand (7 years), the Philippines (9 years), and Peru (10 years), and these countries had the lowest rates of resistance," wrote Dr. Dalton of the Centers for Disease Control and Prevention. "By contrast, South Korea and Russia had the longest histories of availability (more than 20 years) and the highest rates of resistance."
PETTS was launched in 2003 to determine the risk factors for and frequency of acquired resistance to second-line therapies in people with MDR tuberculosis. In 2005, in light of burgeoning numbers, the program was modified to include data on people with XDR tuberculosis.
The current report focused on eight countries: Estonia, Latvia, Peru, the Philippines, Russia, South Africa, South Korea, and Thailand. Samples from patients with MDR tuberculosis were obtained from large clinical centers in each country during 2005-2008. Inclusion criteria were at least 30 days of treatment with a second-line antituberculosis drug, with sputum collection within 30 days before or after the initiation of therapy.
Of 1,540 isolates tested, 1,278 (83%) were MDR. Most of those patients (94%) had a history of tuberculosis, and of those, 71% had experienced it at least once before the tested case.
Of the entire group, 93% had received first-line therapy, but only 15% had received second-line drugs. South Africa had the lowest rate of second-line treatment (3%), while South Korea had the highest (54%).
The overall prevalence of resistance to any second-line drug was 43.7%, but the rate varied among the countries, from 33% in Thailand to 62% in Latvia.
Overall, 20% of isolates were resistant to at least one second-line injectable drug, ranging from 2% in the Philippines to 47% in Latvia. The Philippines also had the lowest prevalence of resistance to all injectables (0.3%), while South Africa had the highest (26%).
The overall resistance rate to at least one second-line oral drug was 27%. Resistance to at least one oral drug ranged from 13% in Estonia to 38% in Latvia; however, other countries also had a high prevalence, including South Korea (36%), the Philippines (32%), Russia (26%), and South Africa (22%).
A total of 6.7% of the isolates were XDR, with the highest prevalence in South Korea (15%) and the lowest in the Philippines (0.8%).
Prior treatment for MDR strains with the second-line drugs was the strongest risk factor for XDR tuberculosis, with a relative risk of 4.75 for injectables and 4 for oral medications.
Although countries with Green Light projects did have more cases, the risks ratios for different resistance types did not reflect the actual numbers, the authors noted.
Resistance to fluoroquinolones and second-line injectable drugs – but not to other oral second-line drugs – was significantly less prevalent in countries that had Green Light Committee approved projects. "This difference was due to the very low prevalence of resistance to second-line drugs in the Philippines, which had the largest Green Light Committee project," the authors said.
The individualized numbers should be useful to national disease management efforts, the researchers added. "Our country-specific results can be extrapolated to guide in-country policy for laboratory capacity and for designing effective treatment recommendations."
PETTS data collection continues, they noted. "The effect of the Green Light Committee initiative in combating acquired resistance to second-line drugs, the timing of acquired resistance, and the role of specific genetic mutations in different regions of the world are also being assessed."
The U.S. Agency for International Development, the Centers for Disease Control and Prevention, the National Institutes of Health, and the Korean Ministry of Health and Welfare sponsored the study. The authors declared no financial conflicts.
Nearly 44% of multidrug-resistant tuberculosis cases tested in eight countries were also resistant to at least one second-line tuberculosis drug, according to results of an international prospective cohort study.
Extensively drug-resistant (XDR) strains were unexpectedly prevalent as well, particularly in South Korea and Russia, reported Tracy Dalton, Ph.D., of the Centers for Disease Control and Prevention, Atlanta, and colleagues. The report was published in the Aug. 30 online issue of The Lancet.
These XDR isolates were detected in 6.7% of patients overall, with prevalence in South Korea (15%) and Russia (11%) exceeding the current World Health Organization global estimate (9.4%). The risk of XDR disease was four times greater in previously treated patients, and previous treatment with second-line drugs was consistently the strongest risk factor for resistance to these drugs (Lancet 2012 Aug. 30 [http://dx.doi.org/10.1016/S0140-6736(12)60734-X]).
Multidrug-resistant (MDR) tuberculosis is resistant to at least rifampicin and isoniazid, and accounts for 3.6%-4.8% of new tuberculosis cases worldwide. XDR tuberculosis is resistant to at least rifampicin, isoniazid, and one or more of the second-line antituberculosis drugs. XDR tuberculosis has been reported in 77 countries.
While the numbers varied between nations, investigators with the international Preserving Effective TB Treatment Study (PETTS) saw a concerning pattern: The prevalence of drug-resistant strains correlated with the time that the second-line drugs had become available through the Green Light Committee, a World Health Organization program designed to increase access to second-line antituberculosis agents.
"[Second-line drugs] had been available for 10 years or less in Thailand (7 years), the Philippines (9 years), and Peru (10 years), and these countries had the lowest rates of resistance," wrote Dr. Dalton of the Centers for Disease Control and Prevention. "By contrast, South Korea and Russia had the longest histories of availability (more than 20 years) and the highest rates of resistance."
PETTS was launched in 2003 to determine the risk factors for and frequency of acquired resistance to second-line therapies in people with MDR tuberculosis. In 2005, in light of burgeoning numbers, the program was modified to include data on people with XDR tuberculosis.
The current report focused on eight countries: Estonia, Latvia, Peru, the Philippines, Russia, South Africa, South Korea, and Thailand. Samples from patients with MDR tuberculosis were obtained from large clinical centers in each country during 2005-2008. Inclusion criteria were at least 30 days of treatment with a second-line antituberculosis drug, with sputum collection within 30 days before or after the initiation of therapy.
Of 1,540 isolates tested, 1,278 (83%) were MDR. Most of those patients (94%) had a history of tuberculosis, and of those, 71% had experienced it at least once before the tested case.
Of the entire group, 93% had received first-line therapy, but only 15% had received second-line drugs. South Africa had the lowest rate of second-line treatment (3%), while South Korea had the highest (54%).
The overall prevalence of resistance to any second-line drug was 43.7%, but the rate varied among the countries, from 33% in Thailand to 62% in Latvia.
Overall, 20% of isolates were resistant to at least one second-line injectable drug, ranging from 2% in the Philippines to 47% in Latvia. The Philippines also had the lowest prevalence of resistance to all injectables (0.3%), while South Africa had the highest (26%).
The overall resistance rate to at least one second-line oral drug was 27%. Resistance to at least one oral drug ranged from 13% in Estonia to 38% in Latvia; however, other countries also had a high prevalence, including South Korea (36%), the Philippines (32%), Russia (26%), and South Africa (22%).
A total of 6.7% of the isolates were XDR, with the highest prevalence in South Korea (15%) and the lowest in the Philippines (0.8%).
Prior treatment for MDR strains with the second-line drugs was the strongest risk factor for XDR tuberculosis, with a relative risk of 4.75 for injectables and 4 for oral medications.
Although countries with Green Light projects did have more cases, the risks ratios for different resistance types did not reflect the actual numbers, the authors noted.
Resistance to fluoroquinolones and second-line injectable drugs – but not to other oral second-line drugs – was significantly less prevalent in countries that had Green Light Committee approved projects. "This difference was due to the very low prevalence of resistance to second-line drugs in the Philippines, which had the largest Green Light Committee project," the authors said.
The individualized numbers should be useful to national disease management efforts, the researchers added. "Our country-specific results can be extrapolated to guide in-country policy for laboratory capacity and for designing effective treatment recommendations."
PETTS data collection continues, they noted. "The effect of the Green Light Committee initiative in combating acquired resistance to second-line drugs, the timing of acquired resistance, and the role of specific genetic mutations in different regions of the world are also being assessed."
The U.S. Agency for International Development, the Centers for Disease Control and Prevention, the National Institutes of Health, and the Korean Ministry of Health and Welfare sponsored the study. The authors declared no financial conflicts.
FROM THE LANCET
Major Finding: In eight countries, 44% patients with multidrug-resistant tuberculosis were resistant to at least one second-line drug, while 6.7% of patients had extensively drug-resistant tuberculosis.
Data Source: The Preserving Effective Tuberculosis Treatment Study is an ongoing international observational study that examines the risks and timing of the development of multidrug-resistant tuberculosis isolates.
Disclosures: The U.S. Agency for International Development, the Centers for Disease Control and Prevention, the National Institutes of Health, and the Korean Ministry of Health and Welfare sponsored this study. The authors declared no financial conflicts.
West Nile Cases, Deaths Continue to Rise
The number of West Nile cases continues to increase, with 1,590 confirmed cases and 66 deaths so far, officials from the Centers for Disease Control and Prevention announced Aug. 29.
The number represents a 40% jump from the Aug. 22 case update, Dr. Lyle Petersen said during a CDC press briefing, with 629 more cases of neuroinvasive disease and an additional 21 deaths.
As of now, 48 states have reported the virus in people, birds, or mosquitoes; 43 states have reported at least one human case, said Dr. Petersen, director of the CDC Division of Vector-Borne Infectious Diseases. The case load is the highest for the first 3 weeks of August since the disease first appeared in the United States in 1999.
"We can’t accurately predict how many human cases will be reported," Dr. Petersen said. "But we think this may come close to, or exceed, the outbreaks of 2002 and 2003."
The majority of the cases (56%; 889) have been the serious, neuroinvasive disease, Dr. Petersen said.
More than 70% of the cases have occurred in South Dakota, Mississippi, Oklahoma, Louisiana, Michigan, and Texas. Texas continues to bear the brunt of the epidemic, accounting for more than 50%.
Since last week, Texas has reported an additional 197 cases, bringing that state’s total to 783, said Dr. David Lakey, commissioner of the Texas Department of State Health Services. The state now claims 416 neuroinvasive cases – an increase of 93 over last week. There have been 10 additional deaths, bringing the total count to 31. The majority of patients with neuroinvasive disease (95%) have been hospitalized, and 40% treated in intensive care units.
Those numbers are likely to increase for at least a few more weeks, Dr. Lakey said during the briefing. "As I look at the data, I am not convinced the disease has peaked."
Hardest hit has been Dallas County, with a total of 309 cases, 152 of those neuroinvasive, and 12 deaths.
Hurricane Isaac is not likely to have an effect on transmission, Dr. Petersen said. There was an uptick in West Nile after Hurricane Katrina, but data suggest that was mostly due to increased outdoor exposure due to damaged houses and the recovery efforts.
Although other states are reporting a far lower incidence, the virus is there and people are still at risk.
"This virus is endemic now throughout the entire U.S., and it circulates every year. It’s just a matter of how much. There is a risk everywhere. It may be lower in some states, but it is still not zero," Dr. Peterson said.
Currently, there are four approved West Nile vaccines for horses, but none for humans, and there probably will never be, he added. "We know we can create an effective vaccine, and in fact, there have been a few successful phase I and II trials. The problem is that we are dealing with a relatively low-incidence disease with widely dispersed cases, so it’s fairly daunting to produce a phase III trial that shows efficacy."
West Nile’s unpredictable seasonal transmission also makes it nearly impossible to get the numbers necessary for such a study. The same issue affects treatment research. "A number of treatment trials have been started, but none has been completed because of low patient enrollment."
As public health officials, neither Dr. Petersen nor Dr. Lakey have any financial disclosures.
The number of West Nile cases continues to increase, with 1,590 confirmed cases and 66 deaths so far, officials from the Centers for Disease Control and Prevention announced Aug. 29.
The number represents a 40% jump from the Aug. 22 case update, Dr. Lyle Petersen said during a CDC press briefing, with 629 more cases of neuroinvasive disease and an additional 21 deaths.
As of now, 48 states have reported the virus in people, birds, or mosquitoes; 43 states have reported at least one human case, said Dr. Petersen, director of the CDC Division of Vector-Borne Infectious Diseases. The case load is the highest for the first 3 weeks of August since the disease first appeared in the United States in 1999.
"We can’t accurately predict how many human cases will be reported," Dr. Petersen said. "But we think this may come close to, or exceed, the outbreaks of 2002 and 2003."
The majority of the cases (56%; 889) have been the serious, neuroinvasive disease, Dr. Petersen said.
More than 70% of the cases have occurred in South Dakota, Mississippi, Oklahoma, Louisiana, Michigan, and Texas. Texas continues to bear the brunt of the epidemic, accounting for more than 50%.
Since last week, Texas has reported an additional 197 cases, bringing that state’s total to 783, said Dr. David Lakey, commissioner of the Texas Department of State Health Services. The state now claims 416 neuroinvasive cases – an increase of 93 over last week. There have been 10 additional deaths, bringing the total count to 31. The majority of patients with neuroinvasive disease (95%) have been hospitalized, and 40% treated in intensive care units.
Those numbers are likely to increase for at least a few more weeks, Dr. Lakey said during the briefing. "As I look at the data, I am not convinced the disease has peaked."
Hardest hit has been Dallas County, with a total of 309 cases, 152 of those neuroinvasive, and 12 deaths.
Hurricane Isaac is not likely to have an effect on transmission, Dr. Petersen said. There was an uptick in West Nile after Hurricane Katrina, but data suggest that was mostly due to increased outdoor exposure due to damaged houses and the recovery efforts.
Although other states are reporting a far lower incidence, the virus is there and people are still at risk.
"This virus is endemic now throughout the entire U.S., and it circulates every year. It’s just a matter of how much. There is a risk everywhere. It may be lower in some states, but it is still not zero," Dr. Peterson said.
Currently, there are four approved West Nile vaccines for horses, but none for humans, and there probably will never be, he added. "We know we can create an effective vaccine, and in fact, there have been a few successful phase I and II trials. The problem is that we are dealing with a relatively low-incidence disease with widely dispersed cases, so it’s fairly daunting to produce a phase III trial that shows efficacy."
West Nile’s unpredictable seasonal transmission also makes it nearly impossible to get the numbers necessary for such a study. The same issue affects treatment research. "A number of treatment trials have been started, but none has been completed because of low patient enrollment."
As public health officials, neither Dr. Petersen nor Dr. Lakey have any financial disclosures.
The number of West Nile cases continues to increase, with 1,590 confirmed cases and 66 deaths so far, officials from the Centers for Disease Control and Prevention announced Aug. 29.
The number represents a 40% jump from the Aug. 22 case update, Dr. Lyle Petersen said during a CDC press briefing, with 629 more cases of neuroinvasive disease and an additional 21 deaths.
As of now, 48 states have reported the virus in people, birds, or mosquitoes; 43 states have reported at least one human case, said Dr. Petersen, director of the CDC Division of Vector-Borne Infectious Diseases. The case load is the highest for the first 3 weeks of August since the disease first appeared in the United States in 1999.
"We can’t accurately predict how many human cases will be reported," Dr. Petersen said. "But we think this may come close to, or exceed, the outbreaks of 2002 and 2003."
The majority of the cases (56%; 889) have been the serious, neuroinvasive disease, Dr. Petersen said.
More than 70% of the cases have occurred in South Dakota, Mississippi, Oklahoma, Louisiana, Michigan, and Texas. Texas continues to bear the brunt of the epidemic, accounting for more than 50%.
Since last week, Texas has reported an additional 197 cases, bringing that state’s total to 783, said Dr. David Lakey, commissioner of the Texas Department of State Health Services. The state now claims 416 neuroinvasive cases – an increase of 93 over last week. There have been 10 additional deaths, bringing the total count to 31. The majority of patients with neuroinvasive disease (95%) have been hospitalized, and 40% treated in intensive care units.
Those numbers are likely to increase for at least a few more weeks, Dr. Lakey said during the briefing. "As I look at the data, I am not convinced the disease has peaked."
Hardest hit has been Dallas County, with a total of 309 cases, 152 of those neuroinvasive, and 12 deaths.
Hurricane Isaac is not likely to have an effect on transmission, Dr. Petersen said. There was an uptick in West Nile after Hurricane Katrina, but data suggest that was mostly due to increased outdoor exposure due to damaged houses and the recovery efforts.
Although other states are reporting a far lower incidence, the virus is there and people are still at risk.
"This virus is endemic now throughout the entire U.S., and it circulates every year. It’s just a matter of how much. There is a risk everywhere. It may be lower in some states, but it is still not zero," Dr. Peterson said.
Currently, there are four approved West Nile vaccines for horses, but none for humans, and there probably will never be, he added. "We know we can create an effective vaccine, and in fact, there have been a few successful phase I and II trials. The problem is that we are dealing with a relatively low-incidence disease with widely dispersed cases, so it’s fairly daunting to produce a phase III trial that shows efficacy."
West Nile’s unpredictable seasonal transmission also makes it nearly impossible to get the numbers necessary for such a study. The same issue affects treatment research. "A number of treatment trials have been started, but none has been completed because of low patient enrollment."
As public health officials, neither Dr. Petersen nor Dr. Lakey have any financial disclosures.
Equally Low Thrombosis Seen With Zotarolimus-, Sirolimus-Eluting Stents
Rates of stent thrombosis were low and similar between patients implanted with zotarolimus-eluting stents and those with sirolimus-eluting stents in a large, randomized controlled trial.
Over a 3-year period, thrombosis occurred in less than 2% of each group. Furthermore, there were no significant between-group differences in overall mortality, myocardial infarction, or cardiac death, Dr. Edoardo Camenzind and colleagues reported in the August 27 online issue of the Lancet. The study results were simultaneously presented at the annual congress of the European Society of Cardiology.
The 1.4% rate of stent thrombosis noted with the zotarolimus stent matched the expected rate of 1.5%, but the rate with the sirolimus stent was lower than anticipated, with an expected rate of 2.5% and an observed rate of 1.8%, wrote Dr. Camenzind of the University of Geneva and his coauthors (Lancet Aug. 27, 2012 [doi: 10.1016/S0140-6736(12)61336-1]).
But the Patient Related Outcomes With Endeavor Versus Cypher Stenting Trial (PROTECT) did show some time-related differences between the two devices, finding more thromboses in the first year with the zotarolimus stent (Z-ES, Endeavor), but more in years 2 and 3 with the sirolimus stent (S-ES, Cypher).
"Interestingly, the pattern of events over time was distributed differentially, with both devices having the same incidence of stent thrombosis, but C-ES having fewer late, but more very late, stent thrombosis than Z-ES," the authors noted. "The higher incidence of late stent thrombosis in the Z-ES group was driven by an increased incidence of definite stent thrombosis."
The study enrolled 8,709 patients who underwent an elective, unplanned, or emergency stenting of coronary arteries. They were a mean of 63 years old. The most common reason for stent placement was acute myocardial infarction (26%). Other reasons for stenting included unstable or stable angina or silent ischemia.
At discharge, 96% were on dual antiplatelet therapy. This number had dropped to 88% at 1 year, 37% at 2 years, and 30% by 3 years.
At the end of follow-up, definite or probable stent thrombosis had occurred in 61 (1.4%) of the Z-ES group and in 75 (1.8%) of the S-ES group – not a significant difference. Nor were there significant differences in any of the main secondary end points of total death and nonfatal MI, or cardiac death and nonfatal MI.
The study revealed a distinct time-associated pattern of thrombosis. In the first month after the procedure, definite or probable thrombosis occurred in 31 of the Z-ES group and 26 of the S-ES group. From days 31 to 360, the numbers were 17 and 5, respectively. During the last 2 years of follow-up, there were 13 definite or probable thromboses in the Z-ES group and 44 in the S-ES group.
Over the entire study period, the Z-ES was associated with a lower incidence of definite stent thrombosis than the S-ES (0.7% vs. 1.2%), but the S-ES had a lower incidence of target vessel revascularization (7.1% vs. 8.2%). None of these differences were significant, however.
Those findings also showed time-related differences. During the first year of the study, significantly more target vessel revascularizations occurred in the Z-ES group (5.8% vs. 3.4%), but that difference was reversed in years 2-3 (4.0% S-ES vs. 2.9% Z-ES).
Although slight, the differences observed in PROTECT are still important, the investigators said: "Given the seriousness of clinical manifestation of stent thrombosis, typically death or myocardial infarction, any reduction has clinical relevance. Furthermore, hundreds of thousands of patients worldwide have been implanted with these devices."
Follow-up on these patients will continue for another 2 years. This "will show whether curves of definite and definite or probable stent thrombosis further diverge and will translate into differences in clinical safety outcomes," Dr. Camenzind and his associates said.
The study was funded by Medtronic, maker of the Endeavor stent. Dr. Camenzind had no financial disclosures but several of his coauthors noted receiving financial remuneration from Medtronic and from other pharmaceutical companies and device manufacturers.
Results of the PROTECT study are academic now, but perhaps still interesting to physicians caring for the millions of patients who still carry the zotarolimus- and sirolimus-eluting stents, Dr. Robert A. Byrne and his colleagues wrote in an accompanying editorial.
"Since the study began in 2007, a significant shift has occurred in interventional cardiology practice, such that both study devices have largely fallen out of use worldwide, being superseded by new devices with increased efficacy and decreased rates of stent thrombosis," wrote Dr. Byrne. "This shift undoubtedly limits the immediate clinical relevance of these findings."
Dr. Byrne and his coauthors also said that PROTECT investigators problematically searched too hard for any interesting tidbits in their secondary analyses.
"The authors report a negative clinical trial with no differences in the primary endpoint. This finding is supported by an absence of difference in the main secondary endpoints. For this reason, the value of all additional analyses presented in the article is restricted, and to highlight differences in the temporal distribution of stent thrombosis between the two devices is inadvisable. This analysis was not prespecified and the probability of a chance finding is high."
The time-driven conclusions can be seeing as a never-ending division problem, they suggested.
"If one notes that stent thrombosis beyond 1 year is higher with [the sirolimus-eluting stent] ... then one must also observe that the number of patients with stent thrombosis up to 1 year is higher with [the zotarolimus-eluting stent]. To take the argument further, one must then question whether it is preferable for a patient to suffer stent thrombosis during the first year or after it: reductio ad absurdum" – arguing on and on that the finding is true because a false result follows from its denial, they wrote.
Dr. Byrne is an interventional cardiologist at Deutsches Herzzentrum, Munich. He had no financial disclosures, but a coauthor disclosed that he has received lecture fees from Medtronic, as well as other device and pharmaceutical companies.
Results of the PROTECT study are academic now, but perhaps still interesting to physicians caring for the millions of patients who still carry the zotarolimus- and sirolimus-eluting stents, Dr. Robert A. Byrne and his colleagues wrote in an accompanying editorial.
"Since the study began in 2007, a significant shift has occurred in interventional cardiology practice, such that both study devices have largely fallen out of use worldwide, being superseded by new devices with increased efficacy and decreased rates of stent thrombosis," wrote Dr. Byrne. "This shift undoubtedly limits the immediate clinical relevance of these findings."
Dr. Byrne and his coauthors also said that PROTECT investigators problematically searched too hard for any interesting tidbits in their secondary analyses.
"The authors report a negative clinical trial with no differences in the primary endpoint. This finding is supported by an absence of difference in the main secondary endpoints. For this reason, the value of all additional analyses presented in the article is restricted, and to highlight differences in the temporal distribution of stent thrombosis between the two devices is inadvisable. This analysis was not prespecified and the probability of a chance finding is high."
The time-driven conclusions can be seeing as a never-ending division problem, they suggested.
"If one notes that stent thrombosis beyond 1 year is higher with [the sirolimus-eluting stent] ... then one must also observe that the number of patients with stent thrombosis up to 1 year is higher with [the zotarolimus-eluting stent]. To take the argument further, one must then question whether it is preferable for a patient to suffer stent thrombosis during the first year or after it: reductio ad absurdum" – arguing on and on that the finding is true because a false result follows from its denial, they wrote.
Dr. Byrne is an interventional cardiologist at Deutsches Herzzentrum, Munich. He had no financial disclosures, but a coauthor disclosed that he has received lecture fees from Medtronic, as well as other device and pharmaceutical companies.
Results of the PROTECT study are academic now, but perhaps still interesting to physicians caring for the millions of patients who still carry the zotarolimus- and sirolimus-eluting stents, Dr. Robert A. Byrne and his colleagues wrote in an accompanying editorial.
"Since the study began in 2007, a significant shift has occurred in interventional cardiology practice, such that both study devices have largely fallen out of use worldwide, being superseded by new devices with increased efficacy and decreased rates of stent thrombosis," wrote Dr. Byrne. "This shift undoubtedly limits the immediate clinical relevance of these findings."
Dr. Byrne and his coauthors also said that PROTECT investigators problematically searched too hard for any interesting tidbits in their secondary analyses.
"The authors report a negative clinical trial with no differences in the primary endpoint. This finding is supported by an absence of difference in the main secondary endpoints. For this reason, the value of all additional analyses presented in the article is restricted, and to highlight differences in the temporal distribution of stent thrombosis between the two devices is inadvisable. This analysis was not prespecified and the probability of a chance finding is high."
The time-driven conclusions can be seeing as a never-ending division problem, they suggested.
"If one notes that stent thrombosis beyond 1 year is higher with [the sirolimus-eluting stent] ... then one must also observe that the number of patients with stent thrombosis up to 1 year is higher with [the zotarolimus-eluting stent]. To take the argument further, one must then question whether it is preferable for a patient to suffer stent thrombosis during the first year or after it: reductio ad absurdum" – arguing on and on that the finding is true because a false result follows from its denial, they wrote.
Dr. Byrne is an interventional cardiologist at Deutsches Herzzentrum, Munich. He had no financial disclosures, but a coauthor disclosed that he has received lecture fees from Medtronic, as well as other device and pharmaceutical companies.
Rates of stent thrombosis were low and similar between patients implanted with zotarolimus-eluting stents and those with sirolimus-eluting stents in a large, randomized controlled trial.
Over a 3-year period, thrombosis occurred in less than 2% of each group. Furthermore, there were no significant between-group differences in overall mortality, myocardial infarction, or cardiac death, Dr. Edoardo Camenzind and colleagues reported in the August 27 online issue of the Lancet. The study results were simultaneously presented at the annual congress of the European Society of Cardiology.
The 1.4% rate of stent thrombosis noted with the zotarolimus stent matched the expected rate of 1.5%, but the rate with the sirolimus stent was lower than anticipated, with an expected rate of 2.5% and an observed rate of 1.8%, wrote Dr. Camenzind of the University of Geneva and his coauthors (Lancet Aug. 27, 2012 [doi: 10.1016/S0140-6736(12)61336-1]).
But the Patient Related Outcomes With Endeavor Versus Cypher Stenting Trial (PROTECT) did show some time-related differences between the two devices, finding more thromboses in the first year with the zotarolimus stent (Z-ES, Endeavor), but more in years 2 and 3 with the sirolimus stent (S-ES, Cypher).
"Interestingly, the pattern of events over time was distributed differentially, with both devices having the same incidence of stent thrombosis, but C-ES having fewer late, but more very late, stent thrombosis than Z-ES," the authors noted. "The higher incidence of late stent thrombosis in the Z-ES group was driven by an increased incidence of definite stent thrombosis."
The study enrolled 8,709 patients who underwent an elective, unplanned, or emergency stenting of coronary arteries. They were a mean of 63 years old. The most common reason for stent placement was acute myocardial infarction (26%). Other reasons for stenting included unstable or stable angina or silent ischemia.
At discharge, 96% were on dual antiplatelet therapy. This number had dropped to 88% at 1 year, 37% at 2 years, and 30% by 3 years.
At the end of follow-up, definite or probable stent thrombosis had occurred in 61 (1.4%) of the Z-ES group and in 75 (1.8%) of the S-ES group – not a significant difference. Nor were there significant differences in any of the main secondary end points of total death and nonfatal MI, or cardiac death and nonfatal MI.
The study revealed a distinct time-associated pattern of thrombosis. In the first month after the procedure, definite or probable thrombosis occurred in 31 of the Z-ES group and 26 of the S-ES group. From days 31 to 360, the numbers were 17 and 5, respectively. During the last 2 years of follow-up, there were 13 definite or probable thromboses in the Z-ES group and 44 in the S-ES group.
Over the entire study period, the Z-ES was associated with a lower incidence of definite stent thrombosis than the S-ES (0.7% vs. 1.2%), but the S-ES had a lower incidence of target vessel revascularization (7.1% vs. 8.2%). None of these differences were significant, however.
Those findings also showed time-related differences. During the first year of the study, significantly more target vessel revascularizations occurred in the Z-ES group (5.8% vs. 3.4%), but that difference was reversed in years 2-3 (4.0% S-ES vs. 2.9% Z-ES).
Although slight, the differences observed in PROTECT are still important, the investigators said: "Given the seriousness of clinical manifestation of stent thrombosis, typically death or myocardial infarction, any reduction has clinical relevance. Furthermore, hundreds of thousands of patients worldwide have been implanted with these devices."
Follow-up on these patients will continue for another 2 years. This "will show whether curves of definite and definite or probable stent thrombosis further diverge and will translate into differences in clinical safety outcomes," Dr. Camenzind and his associates said.
The study was funded by Medtronic, maker of the Endeavor stent. Dr. Camenzind had no financial disclosures but several of his coauthors noted receiving financial remuneration from Medtronic and from other pharmaceutical companies and device manufacturers.
Rates of stent thrombosis were low and similar between patients implanted with zotarolimus-eluting stents and those with sirolimus-eluting stents in a large, randomized controlled trial.
Over a 3-year period, thrombosis occurred in less than 2% of each group. Furthermore, there were no significant between-group differences in overall mortality, myocardial infarction, or cardiac death, Dr. Edoardo Camenzind and colleagues reported in the August 27 online issue of the Lancet. The study results were simultaneously presented at the annual congress of the European Society of Cardiology.
The 1.4% rate of stent thrombosis noted with the zotarolimus stent matched the expected rate of 1.5%, but the rate with the sirolimus stent was lower than anticipated, with an expected rate of 2.5% and an observed rate of 1.8%, wrote Dr. Camenzind of the University of Geneva and his coauthors (Lancet Aug. 27, 2012 [doi: 10.1016/S0140-6736(12)61336-1]).
But the Patient Related Outcomes With Endeavor Versus Cypher Stenting Trial (PROTECT) did show some time-related differences between the two devices, finding more thromboses in the first year with the zotarolimus stent (Z-ES, Endeavor), but more in years 2 and 3 with the sirolimus stent (S-ES, Cypher).
"Interestingly, the pattern of events over time was distributed differentially, with both devices having the same incidence of stent thrombosis, but C-ES having fewer late, but more very late, stent thrombosis than Z-ES," the authors noted. "The higher incidence of late stent thrombosis in the Z-ES group was driven by an increased incidence of definite stent thrombosis."
The study enrolled 8,709 patients who underwent an elective, unplanned, or emergency stenting of coronary arteries. They were a mean of 63 years old. The most common reason for stent placement was acute myocardial infarction (26%). Other reasons for stenting included unstable or stable angina or silent ischemia.
At discharge, 96% were on dual antiplatelet therapy. This number had dropped to 88% at 1 year, 37% at 2 years, and 30% by 3 years.
At the end of follow-up, definite or probable stent thrombosis had occurred in 61 (1.4%) of the Z-ES group and in 75 (1.8%) of the S-ES group – not a significant difference. Nor were there significant differences in any of the main secondary end points of total death and nonfatal MI, or cardiac death and nonfatal MI.
The study revealed a distinct time-associated pattern of thrombosis. In the first month after the procedure, definite or probable thrombosis occurred in 31 of the Z-ES group and 26 of the S-ES group. From days 31 to 360, the numbers were 17 and 5, respectively. During the last 2 years of follow-up, there were 13 definite or probable thromboses in the Z-ES group and 44 in the S-ES group.
Over the entire study period, the Z-ES was associated with a lower incidence of definite stent thrombosis than the S-ES (0.7% vs. 1.2%), but the S-ES had a lower incidence of target vessel revascularization (7.1% vs. 8.2%). None of these differences were significant, however.
Those findings also showed time-related differences. During the first year of the study, significantly more target vessel revascularizations occurred in the Z-ES group (5.8% vs. 3.4%), but that difference was reversed in years 2-3 (4.0% S-ES vs. 2.9% Z-ES).
Although slight, the differences observed in PROTECT are still important, the investigators said: "Given the seriousness of clinical manifestation of stent thrombosis, typically death or myocardial infarction, any reduction has clinical relevance. Furthermore, hundreds of thousands of patients worldwide have been implanted with these devices."
Follow-up on these patients will continue for another 2 years. This "will show whether curves of definite and definite or probable stent thrombosis further diverge and will translate into differences in clinical safety outcomes," Dr. Camenzind and his associates said.
The study was funded by Medtronic, maker of the Endeavor stent. Dr. Camenzind had no financial disclosures but several of his coauthors noted receiving financial remuneration from Medtronic and from other pharmaceutical companies and device manufacturers.
FROM THE LANCET
Major Finding: Over 3 years, the incidence of stent thrombosis was 1.4% with a zotarolimus-eluting device and 1.8% with a sirolumus-eluting stent.
Data Source: PROTECT was a randomized controlled trial involving 8,709 patients implanted with drug-eluting stents and followed for 3 years.
Disclosures: The study was funded by Medtronic. Dr. Camenzind had no financial disclosures, but several of his coauthors noted receiving financial remuneration from Medtronic and other pharmaceutical companies and device manufacturers.
Alzheimer's Drug Misses Primary End Points in Trials
Eli Lilly's experimental Alzheimer's drug solanezumab failed to have statistically significant benefits in the primary end points of two phase III trials but showed possible early cognitive benefits in secondary analyses, the company announced Aug. 24.
The two 18-month trials, called EXPEDITION 1 and 2, randomized more than 2,050 patients with mild-to-moderate Alzheimer’s disease to the drug or placebo.
Two secondary analyses of pooled data from the trials found significant slowing of cognitive decline that was driven mainly by patients with the mildest disease, Dr. William Thies said in an interview.
"This is consistent with the idea that treatment is best started well before a patient becomes demented," said Dr. Thies, medical director of the Alzheimer’s Association.
The studies’ main contribution may be scientific rather than clinical, he said. "This finding [of slowed decline] in a large human trial shows that if you lower amyloid, you can at least partly affect disease course. This is very important. It’s a piece we have not had before."
Dr. Richard Caselli, a neurologist at the Mayo Clinic in Scottsdale, Ariz., was less enthusiastic.
"The planned secondary analyses of pooled data are certainly of scientific interest, but was this statistically significant effect clinically significant?"
He also suggested that cost could hamper the drug’s real-life impact, should it come to market. "Another monoclonal antibody, rituximab, costs roughly $60,000 per treatment cycle per patient. It seems reasonable to question the potential cost for a drug with clinically minuscule effects, given for a disease that affects 5.4 million Americans.
"Clearly we need an effective therapy, but it must be clinically significant and it must be affordable. Unfortunately, we still have a long way to go to meet this goal."
The Alzheimer’s Disease Cooperative Study is conducting an independent review of the data from the trials, and will present its findings on Oct. 8 at the annual meeting of the American Neurological Association, according to Eli Lilly.
Neither Dr. Thies nor Dr. Caselli had any relevant financial disclosures.
Eli Lilly's experimental Alzheimer's drug solanezumab failed to have statistically significant benefits in the primary end points of two phase III trials but showed possible early cognitive benefits in secondary analyses, the company announced Aug. 24.
The two 18-month trials, called EXPEDITION 1 and 2, randomized more than 2,050 patients with mild-to-moderate Alzheimer’s disease to the drug or placebo.
Two secondary analyses of pooled data from the trials found significant slowing of cognitive decline that was driven mainly by patients with the mildest disease, Dr. William Thies said in an interview.
"This is consistent with the idea that treatment is best started well before a patient becomes demented," said Dr. Thies, medical director of the Alzheimer’s Association.
The studies’ main contribution may be scientific rather than clinical, he said. "This finding [of slowed decline] in a large human trial shows that if you lower amyloid, you can at least partly affect disease course. This is very important. It’s a piece we have not had before."
Dr. Richard Caselli, a neurologist at the Mayo Clinic in Scottsdale, Ariz., was less enthusiastic.
"The planned secondary analyses of pooled data are certainly of scientific interest, but was this statistically significant effect clinically significant?"
He also suggested that cost could hamper the drug’s real-life impact, should it come to market. "Another monoclonal antibody, rituximab, costs roughly $60,000 per treatment cycle per patient. It seems reasonable to question the potential cost for a drug with clinically minuscule effects, given for a disease that affects 5.4 million Americans.
"Clearly we need an effective therapy, but it must be clinically significant and it must be affordable. Unfortunately, we still have a long way to go to meet this goal."
The Alzheimer’s Disease Cooperative Study is conducting an independent review of the data from the trials, and will present its findings on Oct. 8 at the annual meeting of the American Neurological Association, according to Eli Lilly.
Neither Dr. Thies nor Dr. Caselli had any relevant financial disclosures.
Eli Lilly's experimental Alzheimer's drug solanezumab failed to have statistically significant benefits in the primary end points of two phase III trials but showed possible early cognitive benefits in secondary analyses, the company announced Aug. 24.
The two 18-month trials, called EXPEDITION 1 and 2, randomized more than 2,050 patients with mild-to-moderate Alzheimer’s disease to the drug or placebo.
Two secondary analyses of pooled data from the trials found significant slowing of cognitive decline that was driven mainly by patients with the mildest disease, Dr. William Thies said in an interview.
"This is consistent with the idea that treatment is best started well before a patient becomes demented," said Dr. Thies, medical director of the Alzheimer’s Association.
The studies’ main contribution may be scientific rather than clinical, he said. "This finding [of slowed decline] in a large human trial shows that if you lower amyloid, you can at least partly affect disease course. This is very important. It’s a piece we have not had before."
Dr. Richard Caselli, a neurologist at the Mayo Clinic in Scottsdale, Ariz., was less enthusiastic.
"The planned secondary analyses of pooled data are certainly of scientific interest, but was this statistically significant effect clinically significant?"
He also suggested that cost could hamper the drug’s real-life impact, should it come to market. "Another monoclonal antibody, rituximab, costs roughly $60,000 per treatment cycle per patient. It seems reasonable to question the potential cost for a drug with clinically minuscule effects, given for a disease that affects 5.4 million Americans.
"Clearly we need an effective therapy, but it must be clinically significant and it must be affordable. Unfortunately, we still have a long way to go to meet this goal."
The Alzheimer’s Disease Cooperative Study is conducting an independent review of the data from the trials, and will present its findings on Oct. 8 at the annual meeting of the American Neurological Association, according to Eli Lilly.
Neither Dr. Thies nor Dr. Caselli had any relevant financial disclosures.
CDC: 'Worst Season Ever' for West Nile Virus
West Nile virus has never struck the U.S. as hard as it is striking now.
The number of confirmed cases now reaches 1,118 – including 41 deaths, all from the neuroinvasive form of the disease. The number has risen dramatically in recent weeks, leading federal health officials to call the outbreak the worst since the virus arrived in the United States in 1999.
"This is the highest number of cases and deaths that has ever been reported in the period of the first 3 weeks of August," said Dr. Lyle R. Petersen, director of the CDC’s Division of Vector-Borne Infectious Diseases. By the third week in July, only 25 cases had been identified, he added.
Human cases occurred in 38 states; 47 states have found the virus in birds, mosquitoes, or in humans, Dr. Petersen said during a press briefing.
Most troubling is that 56% of the cases (629) have been the more aggressive neuroinvasive disease, he said. The remaining 44% were the noninvasive type characterized by fever and myalgia.
Neuroinvasive cases are somewhat easy to track, since most people who develop that form end up in a hospital. The number of fever-related cases is almost certainly a vast underestimate.
"We think that only 2%-3% of all people with [nonneuroinvasive disease] actually get reported. The reasons are very simple. Many people with milder disease don’t go to the doctor or the doctor does not recognize it as a case. There’s no specific treatment for it, and even during an outbreak, routine testing is not recommended," Dr. Petersen said.
Most of the cases (75%) have occurred in Texas, Mississippi, Louisiana, and Oklahoma, with 50% in Texas alone. "People are understandably very concerned, especially in the hardest-hit areas," Dr. Petersen said.
The case load is particularly high in the city of Dallas and its northern suburbs, said Dr. David Lakey, commissioner of Texas Department of State Health Services.
"We are talking about a disease that’s impacting the lives of hundreds – even thousands – of people," Dr. Lakey said at the briefing. "As of right now we have confirmed 586 cases and 21 deaths. ... I know of at least four more," that will be included in the next Texas case update.
From 2003 to 2011, there were 10 deaths. "So now we have many more cases" than in all the previous years of West Nile combined. "This will certainly be our worst season ever." Cases will probably continue to rise as the West Nile season wears on, Dr. Petersen said.
"The peak usually occurs in mid-August, but it takes a couple of weeks before people become sick, go to the doctor, and get diagnosed and reported. So we expect many more cases to occur and the risk of infection will be with us through the end of September."
A mild winter, early spring, and exceptionally hot summer have probably contributed to the high West Nile activity. "It’s a complicated cycle within the interaction of birds, mosquitoes, and people," Dr Petersen said. "Hot weather seems to promote major outbreaks. The biggest outbreaks in Europe and Africa have occurred during periods of abnormally hot weather. And we know from lab experience that heat can increase transmissibility through mosquitoes."
The CDC is investigating whether the virus has mutated into a more dangerous form, but so far, Dr. Petersen said, there is no evidence to support this.
The blood donor screening program has identified West Nile-infected blood in 26 states. "All donors are now screened for West Nile virus, and by deferring those, we have taken an important step in protecting the blood supply," he said.
West Nile virus has never struck the U.S. as hard as it is striking now.
The number of confirmed cases now reaches 1,118 – including 41 deaths, all from the neuroinvasive form of the disease. The number has risen dramatically in recent weeks, leading federal health officials to call the outbreak the worst since the virus arrived in the United States in 1999.
"This is the highest number of cases and deaths that has ever been reported in the period of the first 3 weeks of August," said Dr. Lyle R. Petersen, director of the CDC’s Division of Vector-Borne Infectious Diseases. By the third week in July, only 25 cases had been identified, he added.
Human cases occurred in 38 states; 47 states have found the virus in birds, mosquitoes, or in humans, Dr. Petersen said during a press briefing.
Most troubling is that 56% of the cases (629) have been the more aggressive neuroinvasive disease, he said. The remaining 44% were the noninvasive type characterized by fever and myalgia.
Neuroinvasive cases are somewhat easy to track, since most people who develop that form end up in a hospital. The number of fever-related cases is almost certainly a vast underestimate.
"We think that only 2%-3% of all people with [nonneuroinvasive disease] actually get reported. The reasons are very simple. Many people with milder disease don’t go to the doctor or the doctor does not recognize it as a case. There’s no specific treatment for it, and even during an outbreak, routine testing is not recommended," Dr. Petersen said.
Most of the cases (75%) have occurred in Texas, Mississippi, Louisiana, and Oklahoma, with 50% in Texas alone. "People are understandably very concerned, especially in the hardest-hit areas," Dr. Petersen said.
The case load is particularly high in the city of Dallas and its northern suburbs, said Dr. David Lakey, commissioner of Texas Department of State Health Services.
"We are talking about a disease that’s impacting the lives of hundreds – even thousands – of people," Dr. Lakey said at the briefing. "As of right now we have confirmed 586 cases and 21 deaths. ... I know of at least four more," that will be included in the next Texas case update.
From 2003 to 2011, there were 10 deaths. "So now we have many more cases" than in all the previous years of West Nile combined. "This will certainly be our worst season ever." Cases will probably continue to rise as the West Nile season wears on, Dr. Petersen said.
"The peak usually occurs in mid-August, but it takes a couple of weeks before people become sick, go to the doctor, and get diagnosed and reported. So we expect many more cases to occur and the risk of infection will be with us through the end of September."
A mild winter, early spring, and exceptionally hot summer have probably contributed to the high West Nile activity. "It’s a complicated cycle within the interaction of birds, mosquitoes, and people," Dr Petersen said. "Hot weather seems to promote major outbreaks. The biggest outbreaks in Europe and Africa have occurred during periods of abnormally hot weather. And we know from lab experience that heat can increase transmissibility through mosquitoes."
The CDC is investigating whether the virus has mutated into a more dangerous form, but so far, Dr. Petersen said, there is no evidence to support this.
The blood donor screening program has identified West Nile-infected blood in 26 states. "All donors are now screened for West Nile virus, and by deferring those, we have taken an important step in protecting the blood supply," he said.
West Nile virus has never struck the U.S. as hard as it is striking now.
The number of confirmed cases now reaches 1,118 – including 41 deaths, all from the neuroinvasive form of the disease. The number has risen dramatically in recent weeks, leading federal health officials to call the outbreak the worst since the virus arrived in the United States in 1999.
"This is the highest number of cases and deaths that has ever been reported in the period of the first 3 weeks of August," said Dr. Lyle R. Petersen, director of the CDC’s Division of Vector-Borne Infectious Diseases. By the third week in July, only 25 cases had been identified, he added.
Human cases occurred in 38 states; 47 states have found the virus in birds, mosquitoes, or in humans, Dr. Petersen said during a press briefing.
Most troubling is that 56% of the cases (629) have been the more aggressive neuroinvasive disease, he said. The remaining 44% were the noninvasive type characterized by fever and myalgia.
Neuroinvasive cases are somewhat easy to track, since most people who develop that form end up in a hospital. The number of fever-related cases is almost certainly a vast underestimate.
"We think that only 2%-3% of all people with [nonneuroinvasive disease] actually get reported. The reasons are very simple. Many people with milder disease don’t go to the doctor or the doctor does not recognize it as a case. There’s no specific treatment for it, and even during an outbreak, routine testing is not recommended," Dr. Petersen said.
Most of the cases (75%) have occurred in Texas, Mississippi, Louisiana, and Oklahoma, with 50% in Texas alone. "People are understandably very concerned, especially in the hardest-hit areas," Dr. Petersen said.
The case load is particularly high in the city of Dallas and its northern suburbs, said Dr. David Lakey, commissioner of Texas Department of State Health Services.
"We are talking about a disease that’s impacting the lives of hundreds – even thousands – of people," Dr. Lakey said at the briefing. "As of right now we have confirmed 586 cases and 21 deaths. ... I know of at least four more," that will be included in the next Texas case update.
From 2003 to 2011, there were 10 deaths. "So now we have many more cases" than in all the previous years of West Nile combined. "This will certainly be our worst season ever." Cases will probably continue to rise as the West Nile season wears on, Dr. Petersen said.
"The peak usually occurs in mid-August, but it takes a couple of weeks before people become sick, go to the doctor, and get diagnosed and reported. So we expect many more cases to occur and the risk of infection will be with us through the end of September."
A mild winter, early spring, and exceptionally hot summer have probably contributed to the high West Nile activity. "It’s a complicated cycle within the interaction of birds, mosquitoes, and people," Dr Petersen said. "Hot weather seems to promote major outbreaks. The biggest outbreaks in Europe and Africa have occurred during periods of abnormally hot weather. And we know from lab experience that heat can increase transmissibility through mosquitoes."
The CDC is investigating whether the virus has mutated into a more dangerous form, but so far, Dr. Petersen said, there is no evidence to support this.
The blood donor screening program has identified West Nile-infected blood in 26 states. "All donors are now screened for West Nile virus, and by deferring those, we have taken an important step in protecting the blood supply," he said.
Mycobacterial Infections: The Link to Ink
Tattoo ink contaminated with Mycobacterium chelonae caused skin infections in 19 people in one New York county last year, underscoring the need for regulatory oversight of tattooing and the inks used in tattooing to ensure public safety.
"Since tattooing has become more popular over time, the incidence of cutaneous mycobacterial infections may be underestimated, given the lack of both routine testing and mandated reporting," wrote Dr. Byron Kennedy and his colleagues. And, "although contamination in tattoo parlors has been implicated in previous outbreaks, our investigation of this outbreak shows that premixed ink contaminated before distribution poses a risk to public health," they reported Aug. 22 in the New England Journal of Medicine (2012 [doi: 10.1056/NEJMoa1205114]).
Investigators at the Monroe County (N.Y.) Department of Public Health tracked the infections to a specific tattoo parlor – and even to a single tattoo artist using a new brand of premixed ink.
The Centers for Disease Control and Prevention (CDC) found the contaminated ink in an investigation that eventually led to a voluntary withdrawal of the product, the investigators noted.
In January, public health officers received a report of a male patient with a persistent granulomatous rash on a recently tattooed area of his arm, wrote Dr. Kennedy, deputy director of the Monroe County public health department, and his colleagues. The patient had received a number of tattoos in his town of Rochester, and had never experienced any reaction. His primary care physician had unsuccessfully treated him with glucocorticoids, so he was referred to a dermatologist. Histopathology showed infection with M. chelonae.
Health department investigators found similar reactions in patients who received tattoos from the same artist at the parlor. During May-December 2011, the artist had been using a new brand of premixed, hand-blended, diluted gray ink as a wash to produce shadow effects. The product contained pigment, distilled water, witch hazel, and glycerin. It was packaged in 20%, 60%, and 90% dilutions.
The artist provided a list of clients who had received tattoos during the time the suspect ink was in use.
Investigators contacted all tattoo parlors in the county to inquire about the type of ink used, and to find out if any clients reported adverse events. No other parlors reported using the ink, and none had received reports of rashes from either patrons or local health care workers.
The tattoo parlor and artist associated with the infection appeared to follow best-practice techniques by using sterile needles, disposable gloves, and ink poured into single-use containers, as well as by providing after-care instructions. The artist did not dilute any inks at the parlor, and there appeared to be no contamination from tap water.
The Food and Drug Administration inspected the facility that manufactured and distributed the premixed ink, and collected packaging materials and samples of the ink and its ingredients. The samples were sent to the CDC for culture.
The investigation identified 19 subjects who developed a similar erythematous rash after receiving tattoos from the same artist at the same location. Clinical results were available for 17. Twelve showed sparse lymphohistiocytic infiltrates in the upper dermis. Granulomas were present in five and acid-fast bacteria in two of the patients. DNA sequencing identified M. chelonae as the causative agent. The organism was also found in an unopened bottle of the 30% dilution ink.
Antibiotic sensitivity testing was performed on two samples. The first culture showed sensitivity to clarithromycin, doxycycline, and linezolid; had intermediate resistance to ciprofloxacin; and was resistant to cefoxitin. The second culture was sensitive to clarithromycin and doxycycline, had intermediate resistance to linezolid, and was resistant to ciprofloxacin and cefoxitin. Most (18) of the patients received effective treatment and improved.
The investigators collected the rest of the premixed ink from the tattoo parlor, and the CDC issued a national alert about the outbreak with the intent of discovering additional cases.
Three states – Washington, Iowa, and Colorado – reported clusters of cutaneous M. chelonae infections associated with tattoos, the CDC noted Aug. 22 in a companion article published in Morbidity and Mortality Weekly Report.
Two clusters occurred in Washington, Brenden Bedard of the Monroe County public health department and his colleagues wrote in MMWR. Cluster 1 included three confirmed and 24 possible cases, all in people tattooed with ink from the same company. Samples from the ink company did not grow the organism, but the company reported that it had received complaints of long-lasting skin reactions.
Cluster 2 included two confirmed and two possible cases, both associated with the same ink, but from a different company. A sample from an unopened ink bottle grew M. chelonae. Investigators could not identify the source of contamination at either company.
The Iowa Department of Public Health reported one confirmed case. M. chelonae recovered from the ink was indistinguishable from the Washington strain, but unrelated to the New York strain.
One case was confirmed in Colorado. Artists at the tattoo parlor in question reported diluting ink and rinsing needles with distilled or reverse-osmosis water. The organism was not recovered from any sources at the manufacturing company or the tattoo parlor; however, the ink used was labeled as drawing ink, not tattoo ink.
The investigators had no relevant financial disclosures.
*Correction, 11/8/2012: An earlier version of this story misstated the location of the patient mentioned in the photo caption. The photo was taken by Dr. Matthew Mahlberg.
State and local offices have long provided safety information about tattoo techniques, materials, and adverse events. But it may be time for the Food and Drug Administration to reinforce that message, Ms. Pamela M. LeBlanc and her colleagues wrote in an accompanying editorial (N. Engl. J. Med. 2012 [doi: 10.1056/NEJMp1206063]).
"In recent months, ... reported outbreaks of nontuberculous mycobaterial infections associated with contaminated tattoo ink have raised questions about the adequacy of prevention efforts implemented at the tattoo-parlor level alone," they wrote. The FDA is "reaching out to health care providers, public health officials, consumers, and the tattoo industry to improve awareness, diagnosis, and reporting (through the MedWatch program) in order to develop more effective measures for tattoo ink–related public health problems."
Since tattoo inks are considered to be in the cosmetic realm, the FDA has little control as long as the inks are marketed in accordance with intended use. Consumers should understand the inherent risks of tattooing, and patronize establishments that use techniques and inks that pose little risk for infection.
However, they wrote, "even if a person receives a tattoo at a tattoo parlor that maintains the highest standards of hygienic practice, there remains a risk of infection from the use of contaminated ink."
The MedWatch system is the most effective way to keep the FDA informed of any problems.
"The FDA encourages health care providers, public health officials, consumers, and tattoo artists to use MedWatch to report to the FDA any tattoo-related infections," as well as other adverse events related to tattooing, they noted. "The agency will continue to collaborate with other public health partners in investigating reported adverse events, identifying root causes, and taking the actions necessary to prevent future illnesses."
Ms. LeBlanc is a consumer safety officer at the FDA. She and her coauthors had no conflicts of interest to disclose.
State and local offices have long provided safety information about tattoo techniques, materials, and adverse events. But it may be time for the Food and Drug Administration to reinforce that message, Ms. Pamela M. LeBlanc and her colleagues wrote in an accompanying editorial (N. Engl. J. Med. 2012 [doi: 10.1056/NEJMp1206063]).
"In recent months, ... reported outbreaks of nontuberculous mycobaterial infections associated with contaminated tattoo ink have raised questions about the adequacy of prevention efforts implemented at the tattoo-parlor level alone," they wrote. The FDA is "reaching out to health care providers, public health officials, consumers, and the tattoo industry to improve awareness, diagnosis, and reporting (through the MedWatch program) in order to develop more effective measures for tattoo ink–related public health problems."
Since tattoo inks are considered to be in the cosmetic realm, the FDA has little control as long as the inks are marketed in accordance with intended use. Consumers should understand the inherent risks of tattooing, and patronize establishments that use techniques and inks that pose little risk for infection.
However, they wrote, "even if a person receives a tattoo at a tattoo parlor that maintains the highest standards of hygienic practice, there remains a risk of infection from the use of contaminated ink."
The MedWatch system is the most effective way to keep the FDA informed of any problems.
"The FDA encourages health care providers, public health officials, consumers, and tattoo artists to use MedWatch to report to the FDA any tattoo-related infections," as well as other adverse events related to tattooing, they noted. "The agency will continue to collaborate with other public health partners in investigating reported adverse events, identifying root causes, and taking the actions necessary to prevent future illnesses."
Ms. LeBlanc is a consumer safety officer at the FDA. She and her coauthors had no conflicts of interest to disclose.
State and local offices have long provided safety information about tattoo techniques, materials, and adverse events. But it may be time for the Food and Drug Administration to reinforce that message, Ms. Pamela M. LeBlanc and her colleagues wrote in an accompanying editorial (N. Engl. J. Med. 2012 [doi: 10.1056/NEJMp1206063]).
"In recent months, ... reported outbreaks of nontuberculous mycobaterial infections associated with contaminated tattoo ink have raised questions about the adequacy of prevention efforts implemented at the tattoo-parlor level alone," they wrote. The FDA is "reaching out to health care providers, public health officials, consumers, and the tattoo industry to improve awareness, diagnosis, and reporting (through the MedWatch program) in order to develop more effective measures for tattoo ink–related public health problems."
Since tattoo inks are considered to be in the cosmetic realm, the FDA has little control as long as the inks are marketed in accordance with intended use. Consumers should understand the inherent risks of tattooing, and patronize establishments that use techniques and inks that pose little risk for infection.
However, they wrote, "even if a person receives a tattoo at a tattoo parlor that maintains the highest standards of hygienic practice, there remains a risk of infection from the use of contaminated ink."
The MedWatch system is the most effective way to keep the FDA informed of any problems.
"The FDA encourages health care providers, public health officials, consumers, and tattoo artists to use MedWatch to report to the FDA any tattoo-related infections," as well as other adverse events related to tattooing, they noted. "The agency will continue to collaborate with other public health partners in investigating reported adverse events, identifying root causes, and taking the actions necessary to prevent future illnesses."
Ms. LeBlanc is a consumer safety officer at the FDA. She and her coauthors had no conflicts of interest to disclose.
Tattoo ink contaminated with Mycobacterium chelonae caused skin infections in 19 people in one New York county last year, underscoring the need for regulatory oversight of tattooing and the inks used in tattooing to ensure public safety.
"Since tattooing has become more popular over time, the incidence of cutaneous mycobacterial infections may be underestimated, given the lack of both routine testing and mandated reporting," wrote Dr. Byron Kennedy and his colleagues. And, "although contamination in tattoo parlors has been implicated in previous outbreaks, our investigation of this outbreak shows that premixed ink contaminated before distribution poses a risk to public health," they reported Aug. 22 in the New England Journal of Medicine (2012 [doi: 10.1056/NEJMoa1205114]).
Investigators at the Monroe County (N.Y.) Department of Public Health tracked the infections to a specific tattoo parlor – and even to a single tattoo artist using a new brand of premixed ink.
The Centers for Disease Control and Prevention (CDC) found the contaminated ink in an investigation that eventually led to a voluntary withdrawal of the product, the investigators noted.
In January, public health officers received a report of a male patient with a persistent granulomatous rash on a recently tattooed area of his arm, wrote Dr. Kennedy, deputy director of the Monroe County public health department, and his colleagues. The patient had received a number of tattoos in his town of Rochester, and had never experienced any reaction. His primary care physician had unsuccessfully treated him with glucocorticoids, so he was referred to a dermatologist. Histopathology showed infection with M. chelonae.
Health department investigators found similar reactions in patients who received tattoos from the same artist at the parlor. During May-December 2011, the artist had been using a new brand of premixed, hand-blended, diluted gray ink as a wash to produce shadow effects. The product contained pigment, distilled water, witch hazel, and glycerin. It was packaged in 20%, 60%, and 90% dilutions.
The artist provided a list of clients who had received tattoos during the time the suspect ink was in use.
Investigators contacted all tattoo parlors in the county to inquire about the type of ink used, and to find out if any clients reported adverse events. No other parlors reported using the ink, and none had received reports of rashes from either patrons or local health care workers.
The tattoo parlor and artist associated with the infection appeared to follow best-practice techniques by using sterile needles, disposable gloves, and ink poured into single-use containers, as well as by providing after-care instructions. The artist did not dilute any inks at the parlor, and there appeared to be no contamination from tap water.
The Food and Drug Administration inspected the facility that manufactured and distributed the premixed ink, and collected packaging materials and samples of the ink and its ingredients. The samples were sent to the CDC for culture.
The investigation identified 19 subjects who developed a similar erythematous rash after receiving tattoos from the same artist at the same location. Clinical results were available for 17. Twelve showed sparse lymphohistiocytic infiltrates in the upper dermis. Granulomas were present in five and acid-fast bacteria in two of the patients. DNA sequencing identified M. chelonae as the causative agent. The organism was also found in an unopened bottle of the 30% dilution ink.
Antibiotic sensitivity testing was performed on two samples. The first culture showed sensitivity to clarithromycin, doxycycline, and linezolid; had intermediate resistance to ciprofloxacin; and was resistant to cefoxitin. The second culture was sensitive to clarithromycin and doxycycline, had intermediate resistance to linezolid, and was resistant to ciprofloxacin and cefoxitin. Most (18) of the patients received effective treatment and improved.
The investigators collected the rest of the premixed ink from the tattoo parlor, and the CDC issued a national alert about the outbreak with the intent of discovering additional cases.
Three states – Washington, Iowa, and Colorado – reported clusters of cutaneous M. chelonae infections associated with tattoos, the CDC noted Aug. 22 in a companion article published in Morbidity and Mortality Weekly Report.
Two clusters occurred in Washington, Brenden Bedard of the Monroe County public health department and his colleagues wrote in MMWR. Cluster 1 included three confirmed and 24 possible cases, all in people tattooed with ink from the same company. Samples from the ink company did not grow the organism, but the company reported that it had received complaints of long-lasting skin reactions.
Cluster 2 included two confirmed and two possible cases, both associated with the same ink, but from a different company. A sample from an unopened ink bottle grew M. chelonae. Investigators could not identify the source of contamination at either company.
The Iowa Department of Public Health reported one confirmed case. M. chelonae recovered from the ink was indistinguishable from the Washington strain, but unrelated to the New York strain.
One case was confirmed in Colorado. Artists at the tattoo parlor in question reported diluting ink and rinsing needles with distilled or reverse-osmosis water. The organism was not recovered from any sources at the manufacturing company or the tattoo parlor; however, the ink used was labeled as drawing ink, not tattoo ink.
The investigators had no relevant financial disclosures.
*Correction, 11/8/2012: An earlier version of this story misstated the location of the patient mentioned in the photo caption. The photo was taken by Dr. Matthew Mahlberg.
Tattoo ink contaminated with Mycobacterium chelonae caused skin infections in 19 people in one New York county last year, underscoring the need for regulatory oversight of tattooing and the inks used in tattooing to ensure public safety.
"Since tattooing has become more popular over time, the incidence of cutaneous mycobacterial infections may be underestimated, given the lack of both routine testing and mandated reporting," wrote Dr. Byron Kennedy and his colleagues. And, "although contamination in tattoo parlors has been implicated in previous outbreaks, our investigation of this outbreak shows that premixed ink contaminated before distribution poses a risk to public health," they reported Aug. 22 in the New England Journal of Medicine (2012 [doi: 10.1056/NEJMoa1205114]).
Investigators at the Monroe County (N.Y.) Department of Public Health tracked the infections to a specific tattoo parlor – and even to a single tattoo artist using a new brand of premixed ink.
The Centers for Disease Control and Prevention (CDC) found the contaminated ink in an investigation that eventually led to a voluntary withdrawal of the product, the investigators noted.
In January, public health officers received a report of a male patient with a persistent granulomatous rash on a recently tattooed area of his arm, wrote Dr. Kennedy, deputy director of the Monroe County public health department, and his colleagues. The patient had received a number of tattoos in his town of Rochester, and had never experienced any reaction. His primary care physician had unsuccessfully treated him with glucocorticoids, so he was referred to a dermatologist. Histopathology showed infection with M. chelonae.
Health department investigators found similar reactions in patients who received tattoos from the same artist at the parlor. During May-December 2011, the artist had been using a new brand of premixed, hand-blended, diluted gray ink as a wash to produce shadow effects. The product contained pigment, distilled water, witch hazel, and glycerin. It was packaged in 20%, 60%, and 90% dilutions.
The artist provided a list of clients who had received tattoos during the time the suspect ink was in use.
Investigators contacted all tattoo parlors in the county to inquire about the type of ink used, and to find out if any clients reported adverse events. No other parlors reported using the ink, and none had received reports of rashes from either patrons or local health care workers.
The tattoo parlor and artist associated with the infection appeared to follow best-practice techniques by using sterile needles, disposable gloves, and ink poured into single-use containers, as well as by providing after-care instructions. The artist did not dilute any inks at the parlor, and there appeared to be no contamination from tap water.
The Food and Drug Administration inspected the facility that manufactured and distributed the premixed ink, and collected packaging materials and samples of the ink and its ingredients. The samples were sent to the CDC for culture.
The investigation identified 19 subjects who developed a similar erythematous rash after receiving tattoos from the same artist at the same location. Clinical results were available for 17. Twelve showed sparse lymphohistiocytic infiltrates in the upper dermis. Granulomas were present in five and acid-fast bacteria in two of the patients. DNA sequencing identified M. chelonae as the causative agent. The organism was also found in an unopened bottle of the 30% dilution ink.
Antibiotic sensitivity testing was performed on two samples. The first culture showed sensitivity to clarithromycin, doxycycline, and linezolid; had intermediate resistance to ciprofloxacin; and was resistant to cefoxitin. The second culture was sensitive to clarithromycin and doxycycline, had intermediate resistance to linezolid, and was resistant to ciprofloxacin and cefoxitin. Most (18) of the patients received effective treatment and improved.
The investigators collected the rest of the premixed ink from the tattoo parlor, and the CDC issued a national alert about the outbreak with the intent of discovering additional cases.
Three states – Washington, Iowa, and Colorado – reported clusters of cutaneous M. chelonae infections associated with tattoos, the CDC noted Aug. 22 in a companion article published in Morbidity and Mortality Weekly Report.
Two clusters occurred in Washington, Brenden Bedard of the Monroe County public health department and his colleagues wrote in MMWR. Cluster 1 included three confirmed and 24 possible cases, all in people tattooed with ink from the same company. Samples from the ink company did not grow the organism, but the company reported that it had received complaints of long-lasting skin reactions.
Cluster 2 included two confirmed and two possible cases, both associated with the same ink, but from a different company. A sample from an unopened ink bottle grew M. chelonae. Investigators could not identify the source of contamination at either company.
The Iowa Department of Public Health reported one confirmed case. M. chelonae recovered from the ink was indistinguishable from the Washington strain, but unrelated to the New York strain.
One case was confirmed in Colorado. Artists at the tattoo parlor in question reported diluting ink and rinsing needles with distilled or reverse-osmosis water. The organism was not recovered from any sources at the manufacturing company or the tattoo parlor; however, the ink used was labeled as drawing ink, not tattoo ink.
The investigators had no relevant financial disclosures.
*Correction, 11/8/2012: An earlier version of this story misstated the location of the patient mentioned in the photo caption. The photo was taken by Dr. Matthew Mahlberg.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: After receiving tattoos from the same artist, at the same location, 19 patients developed a similar erythematous rash that was linked to a new brand of tattoo ink.
Data Source: A local and federal public health investigation identified consumers with the infections.
Disclosures: Dr. Kennedy had no financial disclosures. Individual author disclosures are available with the full text of the article at NEJM.org.
Thought Disorder May Be Predictive of Psychosis
Thought disorder can precede psychosis by years and, in some cases, help predict its outcome.
A prospective study of patients with new-onset psychosis found high rates of thought disorder, regardless of the patients’ underlying mental illness. After 20 years of follow-up, thought disorder had remitted significantly more in bipolar patients than in patients with schizophrenia or schizoaffective disorder.
"Positive symptoms seemed to fade away over time and were less prominent [by 20 years’ follow-up] in all groups," Dr. James A. Wilcox and his colleagues reported in the August issue of Comprehensive Psychiatry. "By contrast, the more negative symptoms seemed to persist in schizophrenia and were less relenting overall."
Dr. Wilcox of the University of Arizona and his coauthors followed 188 patients with new-onset psychosis for 20 years, In all, 68 had been diagnosed with schizophrenia, 60 with schizoaffective disorder, and 60 with bipolar disorder in the manic state. All of the patients provided a detailed personal history and were evaluated with the Brief Psychiatric Rating Scale (BPRS) and the Scale for the Assessment of Thought, Language, and Communication (TLC). Patients were reinterviewed at 10 and 20 years after initial presentation (Compr. Psychiatry 2012;53:674-8).
The initial BPRS evaluation indicated that all patients were severely or extremely ill, with a mean score of 86 for those with schizophrenia, 81 for those with schizoaffective disorder, and 78 for those with bipolar disorder. The baseline mean TLC scores were 33, 32, and 29, respectively.
At the 10-year follow-up, the authors were able to reassess 86% of the cohort. "We found that levels of thought disorder remained significantly higher in patients with schizophrenia than in all other groups," they noted. In contrast, "levels of thought disorder dropped considerably in patients with schizoaffective disorder and very significantly in subjects with bipolar disorder."
The mean TLC score at that evaluation was 23 for those with schizophrenia, 20 for those with schizoaffective disorder, and only 3 for those with bipolar disorder. The mean BPRS scores reflected a similar trend: They remained high for the schizophrenia and schizoaffective groups (70 and 68, respectively), and were significantly lower for those with bipolar disorder (38).
The authors evaluated 79% of the cohort at 20 years. Both the schizophrenia and schizoaffective groups still had "prominent" rates of thought disorder. The mean TLC score was 19 for those with schizophrenia and 18 for those with schizoaffective disorder. In contrast, the bipolar group had a mean TLC score of 5.
The BPRS showed a similar pattern, with still-elevated mean scores in the schizophrenia and schizoaffective groups (65 and 63, respectively), compared with a mean score of 23 for the bipolar group.
The negative thought disorders of poverty of thought and poverty of content were highly predictive of later BPRS scores, and of the long-term outcomes of employment and hospitalization.
"We feel comfortable reporting that the symptoms normally assessed as ‘negative thought disorder’ are strong predictors of poor clinical outcome over the life span of many people," the authors noted.
Limitations of the study included the fairly small number of subjects enrolled and the inability of the investigators to ensure that interviewers were blind to the diagnoses.
Still, the findings show that gathering data early in the course of illness can help doctors understand the evolution of psychotic symptoms over time. "We urge continued study of unique symptoms such as thought disorder to help guide and understand the developing features of severe psychiatric disease states," the investigators wrote.
The National Institute of Mental Health and the CHOPE Endowment paid for the study. The authors had no relevant disclosures.
Thought disorder can precede psychosis by years and, in some cases, help predict its outcome.
A prospective study of patients with new-onset psychosis found high rates of thought disorder, regardless of the patients’ underlying mental illness. After 20 years of follow-up, thought disorder had remitted significantly more in bipolar patients than in patients with schizophrenia or schizoaffective disorder.
"Positive symptoms seemed to fade away over time and were less prominent [by 20 years’ follow-up] in all groups," Dr. James A. Wilcox and his colleagues reported in the August issue of Comprehensive Psychiatry. "By contrast, the more negative symptoms seemed to persist in schizophrenia and were less relenting overall."
Dr. Wilcox of the University of Arizona and his coauthors followed 188 patients with new-onset psychosis for 20 years, In all, 68 had been diagnosed with schizophrenia, 60 with schizoaffective disorder, and 60 with bipolar disorder in the manic state. All of the patients provided a detailed personal history and were evaluated with the Brief Psychiatric Rating Scale (BPRS) and the Scale for the Assessment of Thought, Language, and Communication (TLC). Patients were reinterviewed at 10 and 20 years after initial presentation (Compr. Psychiatry 2012;53:674-8).
The initial BPRS evaluation indicated that all patients were severely or extremely ill, with a mean score of 86 for those with schizophrenia, 81 for those with schizoaffective disorder, and 78 for those with bipolar disorder. The baseline mean TLC scores were 33, 32, and 29, respectively.
At the 10-year follow-up, the authors were able to reassess 86% of the cohort. "We found that levels of thought disorder remained significantly higher in patients with schizophrenia than in all other groups," they noted. In contrast, "levels of thought disorder dropped considerably in patients with schizoaffective disorder and very significantly in subjects with bipolar disorder."
The mean TLC score at that evaluation was 23 for those with schizophrenia, 20 for those with schizoaffective disorder, and only 3 for those with bipolar disorder. The mean BPRS scores reflected a similar trend: They remained high for the schizophrenia and schizoaffective groups (70 and 68, respectively), and were significantly lower for those with bipolar disorder (38).
The authors evaluated 79% of the cohort at 20 years. Both the schizophrenia and schizoaffective groups still had "prominent" rates of thought disorder. The mean TLC score was 19 for those with schizophrenia and 18 for those with schizoaffective disorder. In contrast, the bipolar group had a mean TLC score of 5.
The BPRS showed a similar pattern, with still-elevated mean scores in the schizophrenia and schizoaffective groups (65 and 63, respectively), compared with a mean score of 23 for the bipolar group.
The negative thought disorders of poverty of thought and poverty of content were highly predictive of later BPRS scores, and of the long-term outcomes of employment and hospitalization.
"We feel comfortable reporting that the symptoms normally assessed as ‘negative thought disorder’ are strong predictors of poor clinical outcome over the life span of many people," the authors noted.
Limitations of the study included the fairly small number of subjects enrolled and the inability of the investigators to ensure that interviewers were blind to the diagnoses.
Still, the findings show that gathering data early in the course of illness can help doctors understand the evolution of psychotic symptoms over time. "We urge continued study of unique symptoms such as thought disorder to help guide and understand the developing features of severe psychiatric disease states," the investigators wrote.
The National Institute of Mental Health and the CHOPE Endowment paid for the study. The authors had no relevant disclosures.
Thought disorder can precede psychosis by years and, in some cases, help predict its outcome.
A prospective study of patients with new-onset psychosis found high rates of thought disorder, regardless of the patients’ underlying mental illness. After 20 years of follow-up, thought disorder had remitted significantly more in bipolar patients than in patients with schizophrenia or schizoaffective disorder.
"Positive symptoms seemed to fade away over time and were less prominent [by 20 years’ follow-up] in all groups," Dr. James A. Wilcox and his colleagues reported in the August issue of Comprehensive Psychiatry. "By contrast, the more negative symptoms seemed to persist in schizophrenia and were less relenting overall."
Dr. Wilcox of the University of Arizona and his coauthors followed 188 patients with new-onset psychosis for 20 years, In all, 68 had been diagnosed with schizophrenia, 60 with schizoaffective disorder, and 60 with bipolar disorder in the manic state. All of the patients provided a detailed personal history and were evaluated with the Brief Psychiatric Rating Scale (BPRS) and the Scale for the Assessment of Thought, Language, and Communication (TLC). Patients were reinterviewed at 10 and 20 years after initial presentation (Compr. Psychiatry 2012;53:674-8).
The initial BPRS evaluation indicated that all patients were severely or extremely ill, with a mean score of 86 for those with schizophrenia, 81 for those with schizoaffective disorder, and 78 for those with bipolar disorder. The baseline mean TLC scores were 33, 32, and 29, respectively.
At the 10-year follow-up, the authors were able to reassess 86% of the cohort. "We found that levels of thought disorder remained significantly higher in patients with schizophrenia than in all other groups," they noted. In contrast, "levels of thought disorder dropped considerably in patients with schizoaffective disorder and very significantly in subjects with bipolar disorder."
The mean TLC score at that evaluation was 23 for those with schizophrenia, 20 for those with schizoaffective disorder, and only 3 for those with bipolar disorder. The mean BPRS scores reflected a similar trend: They remained high for the schizophrenia and schizoaffective groups (70 and 68, respectively), and were significantly lower for those with bipolar disorder (38).
The authors evaluated 79% of the cohort at 20 years. Both the schizophrenia and schizoaffective groups still had "prominent" rates of thought disorder. The mean TLC score was 19 for those with schizophrenia and 18 for those with schizoaffective disorder. In contrast, the bipolar group had a mean TLC score of 5.
The BPRS showed a similar pattern, with still-elevated mean scores in the schizophrenia and schizoaffective groups (65 and 63, respectively), compared with a mean score of 23 for the bipolar group.
The negative thought disorders of poverty of thought and poverty of content were highly predictive of later BPRS scores, and of the long-term outcomes of employment and hospitalization.
"We feel comfortable reporting that the symptoms normally assessed as ‘negative thought disorder’ are strong predictors of poor clinical outcome over the life span of many people," the authors noted.
Limitations of the study included the fairly small number of subjects enrolled and the inability of the investigators to ensure that interviewers were blind to the diagnoses.
Still, the findings show that gathering data early in the course of illness can help doctors understand the evolution of psychotic symptoms over time. "We urge continued study of unique symptoms such as thought disorder to help guide and understand the developing features of severe psychiatric disease states," the investigators wrote.
The National Institute of Mental Health and the CHOPE Endowment paid for the study. The authors had no relevant disclosures.
FROM COMPREHENSIVE PSYCHIATRY
Major Finding: At 10 years’ follow-up, the mean score on the Thought, Language, and Communication (TLC) scale was 23 for schizophrenia patients, 20 for those with schizoaffective disorder, and 3 for those with bipolar disorder. At 20 years’ follow-up, when 79% of the cohort was assessed, the TLC scores were 19, 18, and 5, respectively.
Data Source: The prospective study followed 188 patients with new-onset psychosis for up to 20 years.
Disclosures: The National Institute of Mental Health and the CHOPE Endowment paid for the study. The authors had no relevant disclosures.
Skin Cancer Drug Steps Into New Alzheimer's Study
The first human trial of bexarotene, the skin cancer drug shown to clear beta-amyloid brain plaques in mice with Alzheimer’s-like pathology, will commence sometime in September.
The phase Ib trial will randomize 12 healthy subjects to placebo or bexarotene (Targretin), Gary E. Landreth, Ph.D., said in an interview.
Researchers will obtain hourly samples of cerebrospinal fluid and blood for 36 hours after administration of the drug, looking for signs that bexarotene increases natural clearance of the beta-amyloid protein, said Dr. Landreth of Case Western Reserve University, Cleveland.
"If our hypothesis is correct, the drug should accelerate the synthesis and steady-state levels of apolipoprotein E," a protein that helps break down soluble beta-amyloid, he said. Hungry microglia are bexarotene’s second punch; in mice, the drug activated these cells to scavenge beta-amyloid plaques.
If these two events also occur in humans, Dr. Landreth said, "we will have a reason to go forward" into phase II trials.
Media stories about Dr. Landreth’s research sparked a firestorm of dialogue on Alzheimer’s Internet forums and message boards this spring. Families discussed traveling to Canada for bexarotene, and begging their family doctors for a prescription.
Two opinion pieces in the Aug. 9 issue of the New England Journal of Medicine tackled the thorny problem physicians may face when desperate families come looking for answers.
"Writing an off-label prescription is completely permissible and would fulfill family members’ desire to try anything to help their loved ones," wrote Dr. Steven D. Pearson of Massachusetts General Hospital’s Institute for Technology Assessment, Boston, and his colleagues. But in the case of bexarotene, they asked, "is it the right thing to do?"
In the second editorial, Frank M. LaFerla, Ph.D., of the University of California, Irvine, warned against overenthusiasm: "Only a well-designed and carefully executed clinical trial will reveal whether this class of drug lives up to its promise. Until such trials are performed, it would be a mistake to offer this treatment to Alzheimer’s patients."
Dr. Landreth agrees. Although relatively benign, bexarotene can cause hypothyroidism and dyslipidemia in cancer patients. Although volunteers in the upcoming trial will almost certainly experience neither of these, "there is no guarantee at all that it would be safe in an Alzheimer’s population," he said. "I have absolutely no idea what could happen when this drug interacts with a diseased brain. If you remove the plaque, then what happens? Do you end up with holes in the brain? This is the question that keeps me up at night."
Dr. Landreth and his colleague, Paige Cramer, hold a U.S. provisional patent application for bexarotene as a potential therapy for Alzheimer’s disease and have founded ReXceptor Inc., which has licensing options from the university to use bexarotene to treat Alzheimer’s disease.
The first human trial of bexarotene, the skin cancer drug shown to clear beta-amyloid brain plaques in mice with Alzheimer’s-like pathology, will commence sometime in September.
The phase Ib trial will randomize 12 healthy subjects to placebo or bexarotene (Targretin), Gary E. Landreth, Ph.D., said in an interview.
Researchers will obtain hourly samples of cerebrospinal fluid and blood for 36 hours after administration of the drug, looking for signs that bexarotene increases natural clearance of the beta-amyloid protein, said Dr. Landreth of Case Western Reserve University, Cleveland.
"If our hypothesis is correct, the drug should accelerate the synthesis and steady-state levels of apolipoprotein E," a protein that helps break down soluble beta-amyloid, he said. Hungry microglia are bexarotene’s second punch; in mice, the drug activated these cells to scavenge beta-amyloid plaques.
If these two events also occur in humans, Dr. Landreth said, "we will have a reason to go forward" into phase II trials.
Media stories about Dr. Landreth’s research sparked a firestorm of dialogue on Alzheimer’s Internet forums and message boards this spring. Families discussed traveling to Canada for bexarotene, and begging their family doctors for a prescription.
Two opinion pieces in the Aug. 9 issue of the New England Journal of Medicine tackled the thorny problem physicians may face when desperate families come looking for answers.
"Writing an off-label prescription is completely permissible and would fulfill family members’ desire to try anything to help their loved ones," wrote Dr. Steven D. Pearson of Massachusetts General Hospital’s Institute for Technology Assessment, Boston, and his colleagues. But in the case of bexarotene, they asked, "is it the right thing to do?"
In the second editorial, Frank M. LaFerla, Ph.D., of the University of California, Irvine, warned against overenthusiasm: "Only a well-designed and carefully executed clinical trial will reveal whether this class of drug lives up to its promise. Until such trials are performed, it would be a mistake to offer this treatment to Alzheimer’s patients."
Dr. Landreth agrees. Although relatively benign, bexarotene can cause hypothyroidism and dyslipidemia in cancer patients. Although volunteers in the upcoming trial will almost certainly experience neither of these, "there is no guarantee at all that it would be safe in an Alzheimer’s population," he said. "I have absolutely no idea what could happen when this drug interacts with a diseased brain. If you remove the plaque, then what happens? Do you end up with holes in the brain? This is the question that keeps me up at night."
Dr. Landreth and his colleague, Paige Cramer, hold a U.S. provisional patent application for bexarotene as a potential therapy for Alzheimer’s disease and have founded ReXceptor Inc., which has licensing options from the university to use bexarotene to treat Alzheimer’s disease.
The first human trial of bexarotene, the skin cancer drug shown to clear beta-amyloid brain plaques in mice with Alzheimer’s-like pathology, will commence sometime in September.
The phase Ib trial will randomize 12 healthy subjects to placebo or bexarotene (Targretin), Gary E. Landreth, Ph.D., said in an interview.
Researchers will obtain hourly samples of cerebrospinal fluid and blood for 36 hours after administration of the drug, looking for signs that bexarotene increases natural clearance of the beta-amyloid protein, said Dr. Landreth of Case Western Reserve University, Cleveland.
"If our hypothesis is correct, the drug should accelerate the synthesis and steady-state levels of apolipoprotein E," a protein that helps break down soluble beta-amyloid, he said. Hungry microglia are bexarotene’s second punch; in mice, the drug activated these cells to scavenge beta-amyloid plaques.
If these two events also occur in humans, Dr. Landreth said, "we will have a reason to go forward" into phase II trials.
Media stories about Dr. Landreth’s research sparked a firestorm of dialogue on Alzheimer’s Internet forums and message boards this spring. Families discussed traveling to Canada for bexarotene, and begging their family doctors for a prescription.
Two opinion pieces in the Aug. 9 issue of the New England Journal of Medicine tackled the thorny problem physicians may face when desperate families come looking for answers.
"Writing an off-label prescription is completely permissible and would fulfill family members’ desire to try anything to help their loved ones," wrote Dr. Steven D. Pearson of Massachusetts General Hospital’s Institute for Technology Assessment, Boston, and his colleagues. But in the case of bexarotene, they asked, "is it the right thing to do?"
In the second editorial, Frank M. LaFerla, Ph.D., of the University of California, Irvine, warned against overenthusiasm: "Only a well-designed and carefully executed clinical trial will reveal whether this class of drug lives up to its promise. Until such trials are performed, it would be a mistake to offer this treatment to Alzheimer’s patients."
Dr. Landreth agrees. Although relatively benign, bexarotene can cause hypothyroidism and dyslipidemia in cancer patients. Although volunteers in the upcoming trial will almost certainly experience neither of these, "there is no guarantee at all that it would be safe in an Alzheimer’s population," he said. "I have absolutely no idea what could happen when this drug interacts with a diseased brain. If you remove the plaque, then what happens? Do you end up with holes in the brain? This is the question that keeps me up at night."
Dr. Landreth and his colleague, Paige Cramer, hold a U.S. provisional patent application for bexarotene as a potential therapy for Alzheimer’s disease and have founded ReXceptor Inc., which has licensing options from the university to use bexarotene to treat Alzheimer’s disease.
Combination Therapy Yields Mixed Results for PTSD, Substance Abuse
A program of cognitive-behavioral therapy combined with substance abuse treatment decreased the symptoms of posttraumatic stress disorder but had little effect on drug use.
That finding, however, can be construed as a positive, Katherine L. Mills, Ph.D., and her colleagues reported in the Aug. 15 issue of JAMA.
"It’s important to note that most participants randomized to receive [the dual therapy] continued to use substances throughout the study," wrote Dr. Mills and her coauthors. "These findings challenge the widely held view that patients need to be abstinent before any trauma work, let alone prolonged exposure therapy, is commenced."
The study randomized 103 patients with concurrent PTSD and substance abuse to one of two arms: a dual treatment of cognitive-behavioral therapy plus substance abuse treatment, or substance abuse treatment alone, said Dr. Mills of the Australian National Drug and Alcohol Research Centre, Sydney (JAMA 2012;308:690-9).
The active arm employed the Concurrent Treatment of PTSD and Substance Use Disorders Using Prolonged Exposure (COPE) program, which consists of 13 90-minute sessions. Components included motivational enhancement and cognitive-behavioral therapy for substance abuse, education about the interaction of PTSD and substance abuse, in vivo and imaginary exposure, and a final session in which patients constructed an after-care plan.
Usual treatment was the patient’s choice of counseling, inpatient detoxification, residential rehabilitation, and pharmacotherapies. All of the participants were offered 9 months to complete their treatment program.
The patients were a mean of 33 years old. Most (77%) were unemployed, and 35% had been in prison. The most commonly abused drug was heroin, followed by marijuana, amphetamines, benzodiazepines, alcohol, cocaine, opiates, and hallucinogens. Most (93%) had undergone prior substance abuse treatment.
All had experienced multiple traumas, including physical assault (92%), being threatened or held captive (89%), witnessing injury or death (77%), sexual assault (78%), accident or disaster (66%), torture (24%), or combat (2%).
The mean age at first trauma was 8 years. About half of the group had experienced a childhood sexual assault; half also had attempted suicide at least once.
There were compliance problems for the group randomized to dual therapy, the investigators noted. Only 10 patients attended all 13 of the sessions.
By the end of the 9-month follow-up period, both groups had experienced significant reductions in PTSD symptom severity, although the intervention group had significantly greater improvements than did the control group.
Most of the patients continued to use substances at the end of the follow-up period (82% active, 73% control); there was no significant between-group difference. Both groups experienced significant decreases in drug abuse severity, but again, this was not significantly different between the groups. Both groups also experienced similar, significant decreases in anxiety and depression, without a between-group difference.
Seven patients attempted suicide during the study – two from the treatment group and five from the control group. "Although it is possible that these attempts were related to participation in the study, all seven individuals ... elected to remain involved with the study," Dr. Mills and her associates noted.
The investigators also suggested that the final outcomes might actually improve with time. "It’s important to note that studies examining the temporal sequencing of changes in PTSD and substance dependent symptoms have shown that improvements in PTSD symptoms are associated with subsequent improvements in substance dependence, but the reciprocal relationship is not observed," they wrote. "These findings highlight the importance of treating PTSD to improve substance dependence outcomes for individuals with this comorbidity."
The study was funded by the Australian National Health and Medical Research Council. Neither Dr. Mills nor any coauthor had any relevant financial disclosures.
This study is the first published randomized controlled trial to assess the efficacy of prolonged cognitive-behavioral therapy for PTSD and comorbid substance abuse, Lisa M. Najavits, Ph.D., wrote in an accompanying editorial (JAMA 2012;308:714-6).
Although the study was well designed, its results highlight this clinically thorny presentation, she said. "The majority of patients still had PTSD and moderate depression, and nearly half still had evidence of substance dependence, despite receiving treatment and close monitoring in this trial and, for 65% of the patients, other treatments for PTSD prior to this trial."
Allowing a 9-month compliance period was probably an ill-advised aspect of the study, resulting in "a lack of consistent timing for end-of-treatment outcome assessment. ... Even with the time extension, the dropout rate was considered high by the investigators, with patients attending a median of only 5 of 13 sessions," she said.
The trial’s limited results highlight the need for more research, Dr. Najavits said. In light of its ambiguous results, "exposure-based therapy for co-occurring PTSD and substance abuse cannot be widely recommended based on the results of this trial alone."
Dr. Najavits is professor of psychiatry at Boston University, a lecturer at Harvard Medical School, a clinical psychologist at the Veterans Affairs Boston Healthcare System, and clinical associate at McLean Hospital in Belmont, Mass. She had no financial disclosures.
This study is the first published randomized controlled trial to assess the efficacy of prolonged cognitive-behavioral therapy for PTSD and comorbid substance abuse, Lisa M. Najavits, Ph.D., wrote in an accompanying editorial (JAMA 2012;308:714-6).
Although the study was well designed, its results highlight this clinically thorny presentation, she said. "The majority of patients still had PTSD and moderate depression, and nearly half still had evidence of substance dependence, despite receiving treatment and close monitoring in this trial and, for 65% of the patients, other treatments for PTSD prior to this trial."
Allowing a 9-month compliance period was probably an ill-advised aspect of the study, resulting in "a lack of consistent timing for end-of-treatment outcome assessment. ... Even with the time extension, the dropout rate was considered high by the investigators, with patients attending a median of only 5 of 13 sessions," she said.
The trial’s limited results highlight the need for more research, Dr. Najavits said. In light of its ambiguous results, "exposure-based therapy for co-occurring PTSD and substance abuse cannot be widely recommended based on the results of this trial alone."
Dr. Najavits is professor of psychiatry at Boston University, a lecturer at Harvard Medical School, a clinical psychologist at the Veterans Affairs Boston Healthcare System, and clinical associate at McLean Hospital in Belmont, Mass. She had no financial disclosures.
This study is the first published randomized controlled trial to assess the efficacy of prolonged cognitive-behavioral therapy for PTSD and comorbid substance abuse, Lisa M. Najavits, Ph.D., wrote in an accompanying editorial (JAMA 2012;308:714-6).
Although the study was well designed, its results highlight this clinically thorny presentation, she said. "The majority of patients still had PTSD and moderate depression, and nearly half still had evidence of substance dependence, despite receiving treatment and close monitoring in this trial and, for 65% of the patients, other treatments for PTSD prior to this trial."
Allowing a 9-month compliance period was probably an ill-advised aspect of the study, resulting in "a lack of consistent timing for end-of-treatment outcome assessment. ... Even with the time extension, the dropout rate was considered high by the investigators, with patients attending a median of only 5 of 13 sessions," she said.
The trial’s limited results highlight the need for more research, Dr. Najavits said. In light of its ambiguous results, "exposure-based therapy for co-occurring PTSD and substance abuse cannot be widely recommended based on the results of this trial alone."
Dr. Najavits is professor of psychiatry at Boston University, a lecturer at Harvard Medical School, a clinical psychologist at the Veterans Affairs Boston Healthcare System, and clinical associate at McLean Hospital in Belmont, Mass. She had no financial disclosures.
A program of cognitive-behavioral therapy combined with substance abuse treatment decreased the symptoms of posttraumatic stress disorder but had little effect on drug use.
That finding, however, can be construed as a positive, Katherine L. Mills, Ph.D., and her colleagues reported in the Aug. 15 issue of JAMA.
"It’s important to note that most participants randomized to receive [the dual therapy] continued to use substances throughout the study," wrote Dr. Mills and her coauthors. "These findings challenge the widely held view that patients need to be abstinent before any trauma work, let alone prolonged exposure therapy, is commenced."
The study randomized 103 patients with concurrent PTSD and substance abuse to one of two arms: a dual treatment of cognitive-behavioral therapy plus substance abuse treatment, or substance abuse treatment alone, said Dr. Mills of the Australian National Drug and Alcohol Research Centre, Sydney (JAMA 2012;308:690-9).
The active arm employed the Concurrent Treatment of PTSD and Substance Use Disorders Using Prolonged Exposure (COPE) program, which consists of 13 90-minute sessions. Components included motivational enhancement and cognitive-behavioral therapy for substance abuse, education about the interaction of PTSD and substance abuse, in vivo and imaginary exposure, and a final session in which patients constructed an after-care plan.
Usual treatment was the patient’s choice of counseling, inpatient detoxification, residential rehabilitation, and pharmacotherapies. All of the participants were offered 9 months to complete their treatment program.
The patients were a mean of 33 years old. Most (77%) were unemployed, and 35% had been in prison. The most commonly abused drug was heroin, followed by marijuana, amphetamines, benzodiazepines, alcohol, cocaine, opiates, and hallucinogens. Most (93%) had undergone prior substance abuse treatment.
All had experienced multiple traumas, including physical assault (92%), being threatened or held captive (89%), witnessing injury or death (77%), sexual assault (78%), accident or disaster (66%), torture (24%), or combat (2%).
The mean age at first trauma was 8 years. About half of the group had experienced a childhood sexual assault; half also had attempted suicide at least once.
There were compliance problems for the group randomized to dual therapy, the investigators noted. Only 10 patients attended all 13 of the sessions.
By the end of the 9-month follow-up period, both groups had experienced significant reductions in PTSD symptom severity, although the intervention group had significantly greater improvements than did the control group.
Most of the patients continued to use substances at the end of the follow-up period (82% active, 73% control); there was no significant between-group difference. Both groups experienced significant decreases in drug abuse severity, but again, this was not significantly different between the groups. Both groups also experienced similar, significant decreases in anxiety and depression, without a between-group difference.
Seven patients attempted suicide during the study – two from the treatment group and five from the control group. "Although it is possible that these attempts were related to participation in the study, all seven individuals ... elected to remain involved with the study," Dr. Mills and her associates noted.
The investigators also suggested that the final outcomes might actually improve with time. "It’s important to note that studies examining the temporal sequencing of changes in PTSD and substance dependent symptoms have shown that improvements in PTSD symptoms are associated with subsequent improvements in substance dependence, but the reciprocal relationship is not observed," they wrote. "These findings highlight the importance of treating PTSD to improve substance dependence outcomes for individuals with this comorbidity."
The study was funded by the Australian National Health and Medical Research Council. Neither Dr. Mills nor any coauthor had any relevant financial disclosures.
A program of cognitive-behavioral therapy combined with substance abuse treatment decreased the symptoms of posttraumatic stress disorder but had little effect on drug use.
That finding, however, can be construed as a positive, Katherine L. Mills, Ph.D., and her colleagues reported in the Aug. 15 issue of JAMA.
"It’s important to note that most participants randomized to receive [the dual therapy] continued to use substances throughout the study," wrote Dr. Mills and her coauthors. "These findings challenge the widely held view that patients need to be abstinent before any trauma work, let alone prolonged exposure therapy, is commenced."
The study randomized 103 patients with concurrent PTSD and substance abuse to one of two arms: a dual treatment of cognitive-behavioral therapy plus substance abuse treatment, or substance abuse treatment alone, said Dr. Mills of the Australian National Drug and Alcohol Research Centre, Sydney (JAMA 2012;308:690-9).
The active arm employed the Concurrent Treatment of PTSD and Substance Use Disorders Using Prolonged Exposure (COPE) program, which consists of 13 90-minute sessions. Components included motivational enhancement and cognitive-behavioral therapy for substance abuse, education about the interaction of PTSD and substance abuse, in vivo and imaginary exposure, and a final session in which patients constructed an after-care plan.
Usual treatment was the patient’s choice of counseling, inpatient detoxification, residential rehabilitation, and pharmacotherapies. All of the participants were offered 9 months to complete their treatment program.
The patients were a mean of 33 years old. Most (77%) were unemployed, and 35% had been in prison. The most commonly abused drug was heroin, followed by marijuana, amphetamines, benzodiazepines, alcohol, cocaine, opiates, and hallucinogens. Most (93%) had undergone prior substance abuse treatment.
All had experienced multiple traumas, including physical assault (92%), being threatened or held captive (89%), witnessing injury or death (77%), sexual assault (78%), accident or disaster (66%), torture (24%), or combat (2%).
The mean age at first trauma was 8 years. About half of the group had experienced a childhood sexual assault; half also had attempted suicide at least once.
There were compliance problems for the group randomized to dual therapy, the investigators noted. Only 10 patients attended all 13 of the sessions.
By the end of the 9-month follow-up period, both groups had experienced significant reductions in PTSD symptom severity, although the intervention group had significantly greater improvements than did the control group.
Most of the patients continued to use substances at the end of the follow-up period (82% active, 73% control); there was no significant between-group difference. Both groups experienced significant decreases in drug abuse severity, but again, this was not significantly different between the groups. Both groups also experienced similar, significant decreases in anxiety and depression, without a between-group difference.
Seven patients attempted suicide during the study – two from the treatment group and five from the control group. "Although it is possible that these attempts were related to participation in the study, all seven individuals ... elected to remain involved with the study," Dr. Mills and her associates noted.
The investigators also suggested that the final outcomes might actually improve with time. "It’s important to note that studies examining the temporal sequencing of changes in PTSD and substance dependent symptoms have shown that improvements in PTSD symptoms are associated with subsequent improvements in substance dependence, but the reciprocal relationship is not observed," they wrote. "These findings highlight the importance of treating PTSD to improve substance dependence outcomes for individuals with this comorbidity."
The study was funded by the Australian National Health and Medical Research Council. Neither Dr. Mills nor any coauthor had any relevant financial disclosures.
FROM JAMA
Major Finding: A combination of cognitive-behavioral therapy and substance abuse treatment significant reduced PTSD symptoms but did not significantly reduce substance use at the end of the follow-up period (82% active, 73% control).
Data Source: The randomized, controlled trial included 103 patients with co-occurring PTSD and substance abuse.
Disclosures: The study was funded by the Australian National Health and Medical Research Council. Neither Dr. Mills nor any coauthor had any relevant financial disclosures.