User login
Olaparib + paclitaxel for metastatic gastric cancer
Adding olaparib to second-line paclitaxel therapy for recurrent or metastatic gastric adenocarcinoma improved overall survival but not progression-free survival in a phase II clinical trial, investigators reported online Aug. 17 in Journal of Clinical Oncology.
The greatest survival benefit occurred in patients whose cancers had low expression of the ataxia telangiectasia mutated (ATM) gene, suggesting that ATM status may predict response to this treatment, said Dr. Yung-Jue Bang Seoul (Korea) National University, and his associates.
Low ATM expression is thought to reflect a deficiency in DNA repair. Tumors with low ATM levels were particularly sensitive to olaparib in previous studies, so Dr. Bang and his associates compared outcomes with paclitaxel plus olaparib (62 patients) against paclitaxel plus placebo (62 patients) in this prospective double-blind study conducted at multiple medical centers in Korea.
ATM status was determined by testing archived tumor samples. Fourteen percent of the study population had low ATM levels.
Compared with paclitaxel plus placebo, paclitaxel plus olaparib produced a statistically significant and clinically meaningful improvement in overall survival. After a median of 8.4 months of follow-up (range, 0.3-26.2 months), median overall survival was 13.1 months with the combination therapy vs. 8.3 months with paclitaxel plus placebo, for a hazard ratio of 0.56.
In the subgroup of patients with low ATM, median overall survival was not reached by patients receiving combined therapy and was 8.2 months for those receiving paclitaxel alone, for an HR of 0.35.
However, the primary end point of this study wasn’t overall survival but progression-free survival, and adding olaparib to paclitaxel produced a numerical but not a statistically significant improvement in progression-free survival. Median progression-free survival was 3.91 months with combined therapy vs. 3.55 months with paclitaxel alone, for an HR of 0.80. In the subgroup of patients with low ATM, median progression-free survival was 5.29 months with combined therapy vs. 3.68 months with paclitaxel alone, for an HR of 0.74.
“The difference between PFS and OS [outcomes] probably results from the relatively small sample size and the exploratory nature of this phase II trial, which was powered to determine whether olaparib/paclitaxel was sufficiently active to warrant assessment in a phase III trial. The sample size was calculated to detect a promising, but not definitive, benefit. The progression-free survival HRs of 0.80 and 0.74 … are considered quite promising for further exploration in a phase III trial,” Dr. Bang and his associates said (J Clin Oncol. 2015 Aug 17. doi: 10.1200/JCO.2014.60.0320]).
The combination therapy was “generally well tolerated,” and there were no unexpected safety findings. The most common adverse event in both study groups was neutropenia. Rates of grade 3 or higher adverse events were similar between the two study groups: 75.4% with combined therapy and 74.2% with paclitaxel plus placebo. The rate of serious adverse events was higher with paclitaxel alone (37.1%) than with the addition of olaparib (27.9%). No serious adverse events were judged to be related to olaparib.
“Overall, our results provide strong evidence of a treatment effect for olaparib/paclitaxel in this setting, and the clinical signals justify further investigation of this combination in a phase III trial,” they added.
Such a phase III trial is now under way (NCT01924533).
The primary end point of this trial, progression-free survival, likely was difficult to determine in some patients because the study included patients who had nonmeasurable disease and peritoneal spread. This is notably more difficult to evaluate for treatment response or disease progression than other metastatic sites.
Nevertheless the authors should be congratulated for their effort to introduce novel agents into the treatment of advanced gastric cancer.
Dr. Barbara Burtness is in the section of medical oncology in the department of internal medicine at Yale University, New Haven. Her disclosures of potential conflicts of interest are available at www.jco.org. Dr. Burtness reported consulting or advisory roles with VentiRx, Merck, Novartis, MedImmune. Amgen, Bayer, Boehringer Ingelheim and research funding from Genentech and Merck. She made these remarks in an editorial accompanying Dr. Bang’s report (J Clin Oncol. 2015 Aug 17. doi: 10.1200/JCO.2015.62.8487).
The primary end point of this trial, progression-free survival, likely was difficult to determine in some patients because the study included patients who had nonmeasurable disease and peritoneal spread. This is notably more difficult to evaluate for treatment response or disease progression than other metastatic sites.
Nevertheless the authors should be congratulated for their effort to introduce novel agents into the treatment of advanced gastric cancer.
Dr. Barbara Burtness is in the section of medical oncology in the department of internal medicine at Yale University, New Haven. Her disclosures of potential conflicts of interest are available at www.jco.org. Dr. Burtness reported consulting or advisory roles with VentiRx, Merck, Novartis, MedImmune. Amgen, Bayer, Boehringer Ingelheim and research funding from Genentech and Merck. She made these remarks in an editorial accompanying Dr. Bang’s report (J Clin Oncol. 2015 Aug 17. doi: 10.1200/JCO.2015.62.8487).
The primary end point of this trial, progression-free survival, likely was difficult to determine in some patients because the study included patients who had nonmeasurable disease and peritoneal spread. This is notably more difficult to evaluate for treatment response or disease progression than other metastatic sites.
Nevertheless the authors should be congratulated for their effort to introduce novel agents into the treatment of advanced gastric cancer.
Dr. Barbara Burtness is in the section of medical oncology in the department of internal medicine at Yale University, New Haven. Her disclosures of potential conflicts of interest are available at www.jco.org. Dr. Burtness reported consulting or advisory roles with VentiRx, Merck, Novartis, MedImmune. Amgen, Bayer, Boehringer Ingelheim and research funding from Genentech and Merck. She made these remarks in an editorial accompanying Dr. Bang’s report (J Clin Oncol. 2015 Aug 17. doi: 10.1200/JCO.2015.62.8487).
Adding olaparib to second-line paclitaxel therapy for recurrent or metastatic gastric adenocarcinoma improved overall survival but not progression-free survival in a phase II clinical trial, investigators reported online Aug. 17 in Journal of Clinical Oncology.
The greatest survival benefit occurred in patients whose cancers had low expression of the ataxia telangiectasia mutated (ATM) gene, suggesting that ATM status may predict response to this treatment, said Dr. Yung-Jue Bang Seoul (Korea) National University, and his associates.
Low ATM expression is thought to reflect a deficiency in DNA repair. Tumors with low ATM levels were particularly sensitive to olaparib in previous studies, so Dr. Bang and his associates compared outcomes with paclitaxel plus olaparib (62 patients) against paclitaxel plus placebo (62 patients) in this prospective double-blind study conducted at multiple medical centers in Korea.
ATM status was determined by testing archived tumor samples. Fourteen percent of the study population had low ATM levels.
Compared with paclitaxel plus placebo, paclitaxel plus olaparib produced a statistically significant and clinically meaningful improvement in overall survival. After a median of 8.4 months of follow-up (range, 0.3-26.2 months), median overall survival was 13.1 months with the combination therapy vs. 8.3 months with paclitaxel plus placebo, for a hazard ratio of 0.56.
In the subgroup of patients with low ATM, median overall survival was not reached by patients receiving combined therapy and was 8.2 months for those receiving paclitaxel alone, for an HR of 0.35.
However, the primary end point of this study wasn’t overall survival but progression-free survival, and adding olaparib to paclitaxel produced a numerical but not a statistically significant improvement in progression-free survival. Median progression-free survival was 3.91 months with combined therapy vs. 3.55 months with paclitaxel alone, for an HR of 0.80. In the subgroup of patients with low ATM, median progression-free survival was 5.29 months with combined therapy vs. 3.68 months with paclitaxel alone, for an HR of 0.74.
“The difference between PFS and OS [outcomes] probably results from the relatively small sample size and the exploratory nature of this phase II trial, which was powered to determine whether olaparib/paclitaxel was sufficiently active to warrant assessment in a phase III trial. The sample size was calculated to detect a promising, but not definitive, benefit. The progression-free survival HRs of 0.80 and 0.74 … are considered quite promising for further exploration in a phase III trial,” Dr. Bang and his associates said (J Clin Oncol. 2015 Aug 17. doi: 10.1200/JCO.2014.60.0320]).
The combination therapy was “generally well tolerated,” and there were no unexpected safety findings. The most common adverse event in both study groups was neutropenia. Rates of grade 3 or higher adverse events were similar between the two study groups: 75.4% with combined therapy and 74.2% with paclitaxel plus placebo. The rate of serious adverse events was higher with paclitaxel alone (37.1%) than with the addition of olaparib (27.9%). No serious adverse events were judged to be related to olaparib.
“Overall, our results provide strong evidence of a treatment effect for olaparib/paclitaxel in this setting, and the clinical signals justify further investigation of this combination in a phase III trial,” they added.
Such a phase III trial is now under way (NCT01924533).
Adding olaparib to second-line paclitaxel therapy for recurrent or metastatic gastric adenocarcinoma improved overall survival but not progression-free survival in a phase II clinical trial, investigators reported online Aug. 17 in Journal of Clinical Oncology.
The greatest survival benefit occurred in patients whose cancers had low expression of the ataxia telangiectasia mutated (ATM) gene, suggesting that ATM status may predict response to this treatment, said Dr. Yung-Jue Bang Seoul (Korea) National University, and his associates.
Low ATM expression is thought to reflect a deficiency in DNA repair. Tumors with low ATM levels were particularly sensitive to olaparib in previous studies, so Dr. Bang and his associates compared outcomes with paclitaxel plus olaparib (62 patients) against paclitaxel plus placebo (62 patients) in this prospective double-blind study conducted at multiple medical centers in Korea.
ATM status was determined by testing archived tumor samples. Fourteen percent of the study population had low ATM levels.
Compared with paclitaxel plus placebo, paclitaxel plus olaparib produced a statistically significant and clinically meaningful improvement in overall survival. After a median of 8.4 months of follow-up (range, 0.3-26.2 months), median overall survival was 13.1 months with the combination therapy vs. 8.3 months with paclitaxel plus placebo, for a hazard ratio of 0.56.
In the subgroup of patients with low ATM, median overall survival was not reached by patients receiving combined therapy and was 8.2 months for those receiving paclitaxel alone, for an HR of 0.35.
However, the primary end point of this study wasn’t overall survival but progression-free survival, and adding olaparib to paclitaxel produced a numerical but not a statistically significant improvement in progression-free survival. Median progression-free survival was 3.91 months with combined therapy vs. 3.55 months with paclitaxel alone, for an HR of 0.80. In the subgroup of patients with low ATM, median progression-free survival was 5.29 months with combined therapy vs. 3.68 months with paclitaxel alone, for an HR of 0.74.
“The difference between PFS and OS [outcomes] probably results from the relatively small sample size and the exploratory nature of this phase II trial, which was powered to determine whether olaparib/paclitaxel was sufficiently active to warrant assessment in a phase III trial. The sample size was calculated to detect a promising, but not definitive, benefit. The progression-free survival HRs of 0.80 and 0.74 … are considered quite promising for further exploration in a phase III trial,” Dr. Bang and his associates said (J Clin Oncol. 2015 Aug 17. doi: 10.1200/JCO.2014.60.0320]).
The combination therapy was “generally well tolerated,” and there were no unexpected safety findings. The most common adverse event in both study groups was neutropenia. Rates of grade 3 or higher adverse events were similar between the two study groups: 75.4% with combined therapy and 74.2% with paclitaxel plus placebo. The rate of serious adverse events was higher with paclitaxel alone (37.1%) than with the addition of olaparib (27.9%). No serious adverse events were judged to be related to olaparib.
“Overall, our results provide strong evidence of a treatment effect for olaparib/paclitaxel in this setting, and the clinical signals justify further investigation of this combination in a phase III trial,” they added.
Such a phase III trial is now under way (NCT01924533).
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Adding olaparib to paclitaxel improved overall survival in a phase II study of metastatic or recurrent gastric adenocarcinoma.
Major finding: After a median of 8.4 months of follow-up (range, 0.3-26.2 months), median overall survival was 13.1 months with the combination therapy vs. 8.3 months with paclitaxel plus placebo, for an HR of 0.56.
Data source: A multicenter, prospective, randomized, double-blind, placebo-controlled phase II study comparing second-line paclitaxel alone to paclitaxel plus olaparib in 124 Korean patients followed for a median of 8.4 months.
Disclosures: This study was sponsored by AstraZeneca, maker of olaparib. Dr. Bang and his associates also reported other financial ties to AstraZeneca.
High troponin T level doubles CVD risk
An abnormal troponin T level of 14 ng/L or higher, as measured using a high-sensitivity assay, doubles the risk of cardiovascular events and death among patients who have stable ischemic heart disease and type 2 diabetes, according to a report published online Aug. 13 in the New England Journal of Medicine.
Moreover, an increase of more than 25% in troponin T level during the course of 1 year predicts a worse outcome than do stable or decreasing troponin T levels in this patient population, significantly increasing the rates of death from cardiovascular causes, MI, or stroke. These findings “raise the possibility that serial measurements of troponin concentration may improve its prognostic value, and that persistently elevated and increasing troponin concentrations may be the best predictor of adverse outcomes,” said Dr. Brendan M. Everett of the divisions of cardiovascular medicine and preventive medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston.
These findings have particular significance because the prevalence of elevated troponin T was fully 40% in this study, noted Dr. Everett and his associates.
To examine the possible relationship between elevated troponin T and adverse cardiovascular outcomes in patients who had both stable ischemic heart disease and diabetes, the investigators performed a post hoc analysis of data gathered in the Bypass Angioplasty Revascularization Investigation in Type 2 Diabetes (BARI 2D) clinical trial, which compared outcomes between patients who underwent preventive percutaneous coronary intervention or CABG plus intensive medical therapy against those who received intensive medical therapy alone.
For their ancillary study, Dr. Everett and his colleagues focused on 2,285 of these participants who had high-sensitivity assays to measure very low cardiac troponin levels in plasma samples and who were followed for a mean of 5 years. At baseline, 897 (40%) of these patients had troponin T levels of 14 ng/L or higher, the current cutoff point for both men and women.
The 5-year incidence of the composite outcome of death from cardiovascular causes, MI, or stroke was 27.1% in patients with elevated troponin T at baseline, compared with 12.9% in those with normal troponin T. The between-groups differences in each of the individual components of this composite outcome were of similar magnitude, as were the between-group differences in the secondary outcomes of death from any cause and heart failure (N Engl J Med. 2015 Aug 13 [doi:10.1056/NEJMoa1415921]).
This association remained robust after the data were adjusted to account for numerous potentially confounding factors such as traditional CV risk factors, a history of MI, a history of heart failure, the severity of diabetes, glomerular filtration rate, ECG abnormalities, the number of coronary lesions, and the presence of an abnormal ejection fraction.
In a further analysis that divided patients into five groups according to their troponin T levels, adverse event rates were substantially higher than average only in the highest two categories: 14.0-23.0 ng/L and 23.0 ng/L and higher.
In another analysis, the investigators assessed outcomes in a subgroup of 1,984 participants who underwent troponin T testing both at baseline and 1 year later. Patients whose levels increased by more than 25% during that year – about 7% of this subgroup – showed significantly increased risk of all adverse outcomes, compared with patients whose troponin T levels either remained stable or decreased.
Prompt revascularization via percutaneous coronary intervention or coronary artery bypass surgery did not lower the risk of any adverse outcomes in patients who had elevated troponin T. It is therefore crucial that the troponin T assay, already in widespread use, not be used to justify revascularization procedures, the researchers wrote. At least on the basis of this study’s findings, such procedures appear to offer little benefit, they noted.
The National Heart, Lung, and Blood Institute, the National Institute of Diabetes and Digestive and Kidney Diseases, and Roche Diagnostics supported the study. Dr. Everett reported ties with Roche, Novartis, and Genzyme; his associates reported ties to numerous industry sources.
The findings of this study add to the accumulating data that suggest cardiac troponin testing may become routine for risk stratification across the entire spectrum of ischemic heart disease.
However, it is important to note that interpreting elevations in troponin T is a challenge in patients who have kidney impairment, because the problem may impair renal clearance of troponin. In addition, kidney disease may be a concomitant risk factor for ongoing subclinical thrombosis, which could be one of the pathological mechanisms underlying troponin elevation in patients with stable ischemic heart disease.
Chiara Melloni, M.D., and Matthew T. Roe, M.D., are at Duke Clinical Research Institute, Durham, N.C. Their financial disclosures are available at NEJM.org. Dr. Melloni and Dr. Roe made these remarks in an editorial accompanying Dr. Everett’s report (N Engl J Med. 2015 Aug 13 [doi:10.1056/NEJMe1506298]).
The findings of this study add to the accumulating data that suggest cardiac troponin testing may become routine for risk stratification across the entire spectrum of ischemic heart disease.
However, it is important to note that interpreting elevations in troponin T is a challenge in patients who have kidney impairment, because the problem may impair renal clearance of troponin. In addition, kidney disease may be a concomitant risk factor for ongoing subclinical thrombosis, which could be one of the pathological mechanisms underlying troponin elevation in patients with stable ischemic heart disease.
Chiara Melloni, M.D., and Matthew T. Roe, M.D., are at Duke Clinical Research Institute, Durham, N.C. Their financial disclosures are available at NEJM.org. Dr. Melloni and Dr. Roe made these remarks in an editorial accompanying Dr. Everett’s report (N Engl J Med. 2015 Aug 13 [doi:10.1056/NEJMe1506298]).
The findings of this study add to the accumulating data that suggest cardiac troponin testing may become routine for risk stratification across the entire spectrum of ischemic heart disease.
However, it is important to note that interpreting elevations in troponin T is a challenge in patients who have kidney impairment, because the problem may impair renal clearance of troponin. In addition, kidney disease may be a concomitant risk factor for ongoing subclinical thrombosis, which could be one of the pathological mechanisms underlying troponin elevation in patients with stable ischemic heart disease.
Chiara Melloni, M.D., and Matthew T. Roe, M.D., are at Duke Clinical Research Institute, Durham, N.C. Their financial disclosures are available at NEJM.org. Dr. Melloni and Dr. Roe made these remarks in an editorial accompanying Dr. Everett’s report (N Engl J Med. 2015 Aug 13 [doi:10.1056/NEJMe1506298]).
An abnormal troponin T level of 14 ng/L or higher, as measured using a high-sensitivity assay, doubles the risk of cardiovascular events and death among patients who have stable ischemic heart disease and type 2 diabetes, according to a report published online Aug. 13 in the New England Journal of Medicine.
Moreover, an increase of more than 25% in troponin T level during the course of 1 year predicts a worse outcome than do stable or decreasing troponin T levels in this patient population, significantly increasing the rates of death from cardiovascular causes, MI, or stroke. These findings “raise the possibility that serial measurements of troponin concentration may improve its prognostic value, and that persistently elevated and increasing troponin concentrations may be the best predictor of adverse outcomes,” said Dr. Brendan M. Everett of the divisions of cardiovascular medicine and preventive medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston.
These findings have particular significance because the prevalence of elevated troponin T was fully 40% in this study, noted Dr. Everett and his associates.
To examine the possible relationship between elevated troponin T and adverse cardiovascular outcomes in patients who had both stable ischemic heart disease and diabetes, the investigators performed a post hoc analysis of data gathered in the Bypass Angioplasty Revascularization Investigation in Type 2 Diabetes (BARI 2D) clinical trial, which compared outcomes between patients who underwent preventive percutaneous coronary intervention or CABG plus intensive medical therapy against those who received intensive medical therapy alone.
For their ancillary study, Dr. Everett and his colleagues focused on 2,285 of these participants who had high-sensitivity assays to measure very low cardiac troponin levels in plasma samples and who were followed for a mean of 5 years. At baseline, 897 (40%) of these patients had troponin T levels of 14 ng/L or higher, the current cutoff point for both men and women.
The 5-year incidence of the composite outcome of death from cardiovascular causes, MI, or stroke was 27.1% in patients with elevated troponin T at baseline, compared with 12.9% in those with normal troponin T. The between-groups differences in each of the individual components of this composite outcome were of similar magnitude, as were the between-group differences in the secondary outcomes of death from any cause and heart failure (N Engl J Med. 2015 Aug 13 [doi:10.1056/NEJMoa1415921]).
This association remained robust after the data were adjusted to account for numerous potentially confounding factors such as traditional CV risk factors, a history of MI, a history of heart failure, the severity of diabetes, glomerular filtration rate, ECG abnormalities, the number of coronary lesions, and the presence of an abnormal ejection fraction.
In a further analysis that divided patients into five groups according to their troponin T levels, adverse event rates were substantially higher than average only in the highest two categories: 14.0-23.0 ng/L and 23.0 ng/L and higher.
In another analysis, the investigators assessed outcomes in a subgroup of 1,984 participants who underwent troponin T testing both at baseline and 1 year later. Patients whose levels increased by more than 25% during that year – about 7% of this subgroup – showed significantly increased risk of all adverse outcomes, compared with patients whose troponin T levels either remained stable or decreased.
Prompt revascularization via percutaneous coronary intervention or coronary artery bypass surgery did not lower the risk of any adverse outcomes in patients who had elevated troponin T. It is therefore crucial that the troponin T assay, already in widespread use, not be used to justify revascularization procedures, the researchers wrote. At least on the basis of this study’s findings, such procedures appear to offer little benefit, they noted.
The National Heart, Lung, and Blood Institute, the National Institute of Diabetes and Digestive and Kidney Diseases, and Roche Diagnostics supported the study. Dr. Everett reported ties with Roche, Novartis, and Genzyme; his associates reported ties to numerous industry sources.
An abnormal troponin T level of 14 ng/L or higher, as measured using a high-sensitivity assay, doubles the risk of cardiovascular events and death among patients who have stable ischemic heart disease and type 2 diabetes, according to a report published online Aug. 13 in the New England Journal of Medicine.
Moreover, an increase of more than 25% in troponin T level during the course of 1 year predicts a worse outcome than do stable or decreasing troponin T levels in this patient population, significantly increasing the rates of death from cardiovascular causes, MI, or stroke. These findings “raise the possibility that serial measurements of troponin concentration may improve its prognostic value, and that persistently elevated and increasing troponin concentrations may be the best predictor of adverse outcomes,” said Dr. Brendan M. Everett of the divisions of cardiovascular medicine and preventive medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston.
These findings have particular significance because the prevalence of elevated troponin T was fully 40% in this study, noted Dr. Everett and his associates.
To examine the possible relationship between elevated troponin T and adverse cardiovascular outcomes in patients who had both stable ischemic heart disease and diabetes, the investigators performed a post hoc analysis of data gathered in the Bypass Angioplasty Revascularization Investigation in Type 2 Diabetes (BARI 2D) clinical trial, which compared outcomes between patients who underwent preventive percutaneous coronary intervention or CABG plus intensive medical therapy against those who received intensive medical therapy alone.
For their ancillary study, Dr. Everett and his colleagues focused on 2,285 of these participants who had high-sensitivity assays to measure very low cardiac troponin levels in plasma samples and who were followed for a mean of 5 years. At baseline, 897 (40%) of these patients had troponin T levels of 14 ng/L or higher, the current cutoff point for both men and women.
The 5-year incidence of the composite outcome of death from cardiovascular causes, MI, or stroke was 27.1% in patients with elevated troponin T at baseline, compared with 12.9% in those with normal troponin T. The between-groups differences in each of the individual components of this composite outcome were of similar magnitude, as were the between-group differences in the secondary outcomes of death from any cause and heart failure (N Engl J Med. 2015 Aug 13 [doi:10.1056/NEJMoa1415921]).
This association remained robust after the data were adjusted to account for numerous potentially confounding factors such as traditional CV risk factors, a history of MI, a history of heart failure, the severity of diabetes, glomerular filtration rate, ECG abnormalities, the number of coronary lesions, and the presence of an abnormal ejection fraction.
In a further analysis that divided patients into five groups according to their troponin T levels, adverse event rates were substantially higher than average only in the highest two categories: 14.0-23.0 ng/L and 23.0 ng/L and higher.
In another analysis, the investigators assessed outcomes in a subgroup of 1,984 participants who underwent troponin T testing both at baseline and 1 year later. Patients whose levels increased by more than 25% during that year – about 7% of this subgroup – showed significantly increased risk of all adverse outcomes, compared with patients whose troponin T levels either remained stable or decreased.
Prompt revascularization via percutaneous coronary intervention or coronary artery bypass surgery did not lower the risk of any adverse outcomes in patients who had elevated troponin T. It is therefore crucial that the troponin T assay, already in widespread use, not be used to justify revascularization procedures, the researchers wrote. At least on the basis of this study’s findings, such procedures appear to offer little benefit, they noted.
The National Heart, Lung, and Blood Institute, the National Institute of Diabetes and Digestive and Kidney Diseases, and Roche Diagnostics supported the study. Dr. Everett reported ties with Roche, Novartis, and Genzyme; his associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Troponin T levels of 14 ng/L or higher double the risk of CVD events and death in patients with stable ischemic heart disease and concomitant type 2 diabetes.
Major finding: The 5-year incidence of the composite outcome of death from cardiovascular causes, MI, or stroke was 27.1% in patients with elevated troponin T at baseline, compared with 12.9% in those with normal troponin T.
Data source: A post hoc analysis of data in the Bypass Angioplasty Revascularization Type 2 Diabetes (BARI 2D) trial of 2,285 participants who were followed for 5 years.
Disclosures: The National Heart, Lung, and Blood Institute, the National Institute of Diabetes and Digestive and Kidney Diseases, and Roche Diagnostics supported the study. Dr. Everett reported ties with Roche, Novartis, and Genzyme; his associates reported ties to numerous industry sources.
Testosterone Therapy Didn’t Boost Atherosclerosis Over 3 Years
For older men with low or low-normal testosterone levels, using testosterone gel doesn’t appear to worsen two key indicators of cardiovascular health – carotid artery intima-media thickness and coronary artery calcium deposition – according to results of a randomized trial published online Aug. 11 in JAMA.
Testosterone sales for “low T” have risen substantially in the past decade, even though the agent’s long-term effects on the cardiovascular health of older men are not yet known and even though there are concerns that testosterone supplementation may raise the risk of CVD events.
The Testosterone’s Effects on Atherosclerosis Progression in Aging Men (TEAAM) study was a randomized, placebo-controlled, double-blind clinical trial conducted at three U.S. medical centers to investigate this issue, said Dr. Shehzad Basaria, director of the Research Program in Men’s Health, Aging, and Metabolism, Boston Claude D. Pepper Older Americans Independence Center, Brigham and Women’s Hospital, Boston, and his associates.
Study participants were 306 community-dwelling men aged 60 and older enrolled during a 5-year period and randomly assigned to use either 1% testosterone gel (155 men) or placebo gel (151 men) every day for 3 years to treat low or low-normal testosterone levels. The mean age was 68 years. A total of 42% of the participants had hypertension, 15% had diabetes, 15% had known coronary artery disease, 27% were obese, and 43% were taking statin therapy. As expected, free testosterone levels increased in the testosterone group but did not change significantly in the placebo group.
The coprimary endpoint of rate of change in intima-media thickness, which was measured every 6 months, differed by just 0.0002 mm/year between the two study groups, a nonsignificant difference. The difference in the final measure of intima-media thickness taken at the conclusion of treatment, the other coprimary endpoint, was −10.8 Agatston units/year. Neither difference was significant. Similarly, the yearly rate of change in coronary artery calcium did not differ between the two groups, and neither did the final measure of coronary artery calcium at the conclusion of treatment. These findings persisted in several sensitivity analyses and across all subgroups of patients, such as men who were obese, men who had preexisting coronary artery disease, and men who had diabetes, the investigators said (JAMA. 2015 Aug 11. doi: 10.1001/jama.2015.8881). Notably, however, the two study groups also showed no significant differences in scores on the International Index of Erectile Function, either in the total score or in any of the domains of sexual function: erectile function, orgasmic function, sexual desire, intercourse satisfaction, or overall sexual satisfaction. Overall health-related quality of life also was no different between men who took active testosterone and men who took placebo, and there were no differences in the individual domains of physical function, bodily pain, vitality, role limitations due to physical problems, general health perceptions, emotional well-being, social function, or role limitations due to emotional problems.
There were few major cardiovascular disease events, and the numbers and types of events did not differ between the two study groups. Three men in the testosterone group and two in the placebo group had MIs, five in the testosterone group and two in the placebo group underwent coronary revascularization, three in the testosterone group and none in the placebo group had a stroke, and one in the testosterone group and none in the placebo group had a fatal cardiovascular disease (CVD) event. However, the investigators emphasized that this trial was not designed to assess testosterone’s effect on actual CVD events, and a substantially larger study population with many more events would be needed to examine this issue adequately. “These findings should not be interpreted as establishing cardiovascular safety of testosterone use in older men,” Dr. Basaria and his associates noted.
It is also important to note that this study assessed only two measured of cardiovascular health. Testosterone may well affect other measures, such as plaque stability, clot formation, salt retention, water retention, inflammation, “or other mechanisms that might render some men susceptible to early CVD events,” they said.
In addition, testosterone levels decreased over time in the men assigned to active testosterone gel. The researchers could not determine whether this was due to declining treatment adherence, changes in testosterone metabolism over time, progressive suppression of endogenous testosterone production because of the exogenous supplementation, or other factors.
This clinical trial was supported by Solvay Pharmaceuticals and AbbVie, which provided the testosterone and placebo gels used in the study; the Aurora Foundation; the Kronos Longevity Research Institute; the Boston Claude D. Pepper Older Americans Independence Center; and Boston University’s Clinical and Translational Science Institute. Dr. Basaria reported serving as a consultant to Eli Lilly; his associates reported ties to AbbVie, Regenron, Sanofi, Eli Lilly, Function Promoting Therapies, and Hoffman-LaRoche.
For older men with low or low-normal testosterone levels, using testosterone gel doesn’t appear to worsen two key indicators of cardiovascular health – carotid artery intima-media thickness and coronary artery calcium deposition – according to results of a randomized trial published online Aug. 11 in JAMA.
Testosterone sales for “low T” have risen substantially in the past decade, even though the agent’s long-term effects on the cardiovascular health of older men are not yet known and even though there are concerns that testosterone supplementation may raise the risk of CVD events.
The Testosterone’s Effects on Atherosclerosis Progression in Aging Men (TEAAM) study was a randomized, placebo-controlled, double-blind clinical trial conducted at three U.S. medical centers to investigate this issue, said Dr. Shehzad Basaria, director of the Research Program in Men’s Health, Aging, and Metabolism, Boston Claude D. Pepper Older Americans Independence Center, Brigham and Women’s Hospital, Boston, and his associates.
Study participants were 306 community-dwelling men aged 60 and older enrolled during a 5-year period and randomly assigned to use either 1% testosterone gel (155 men) or placebo gel (151 men) every day for 3 years to treat low or low-normal testosterone levels. The mean age was 68 years. A total of 42% of the participants had hypertension, 15% had diabetes, 15% had known coronary artery disease, 27% were obese, and 43% were taking statin therapy. As expected, free testosterone levels increased in the testosterone group but did not change significantly in the placebo group.
The coprimary endpoint of rate of change in intima-media thickness, which was measured every 6 months, differed by just 0.0002 mm/year between the two study groups, a nonsignificant difference. The difference in the final measure of intima-media thickness taken at the conclusion of treatment, the other coprimary endpoint, was −10.8 Agatston units/year. Neither difference was significant. Similarly, the yearly rate of change in coronary artery calcium did not differ between the two groups, and neither did the final measure of coronary artery calcium at the conclusion of treatment. These findings persisted in several sensitivity analyses and across all subgroups of patients, such as men who were obese, men who had preexisting coronary artery disease, and men who had diabetes, the investigators said (JAMA. 2015 Aug 11. doi: 10.1001/jama.2015.8881). Notably, however, the two study groups also showed no significant differences in scores on the International Index of Erectile Function, either in the total score or in any of the domains of sexual function: erectile function, orgasmic function, sexual desire, intercourse satisfaction, or overall sexual satisfaction. Overall health-related quality of life also was no different between men who took active testosterone and men who took placebo, and there were no differences in the individual domains of physical function, bodily pain, vitality, role limitations due to physical problems, general health perceptions, emotional well-being, social function, or role limitations due to emotional problems.
There were few major cardiovascular disease events, and the numbers and types of events did not differ between the two study groups. Three men in the testosterone group and two in the placebo group had MIs, five in the testosterone group and two in the placebo group underwent coronary revascularization, three in the testosterone group and none in the placebo group had a stroke, and one in the testosterone group and none in the placebo group had a fatal cardiovascular disease (CVD) event. However, the investigators emphasized that this trial was not designed to assess testosterone’s effect on actual CVD events, and a substantially larger study population with many more events would be needed to examine this issue adequately. “These findings should not be interpreted as establishing cardiovascular safety of testosterone use in older men,” Dr. Basaria and his associates noted.
It is also important to note that this study assessed only two measured of cardiovascular health. Testosterone may well affect other measures, such as plaque stability, clot formation, salt retention, water retention, inflammation, “or other mechanisms that might render some men susceptible to early CVD events,” they said.
In addition, testosterone levels decreased over time in the men assigned to active testosterone gel. The researchers could not determine whether this was due to declining treatment adherence, changes in testosterone metabolism over time, progressive suppression of endogenous testosterone production because of the exogenous supplementation, or other factors.
This clinical trial was supported by Solvay Pharmaceuticals and AbbVie, which provided the testosterone and placebo gels used in the study; the Aurora Foundation; the Kronos Longevity Research Institute; the Boston Claude D. Pepper Older Americans Independence Center; and Boston University’s Clinical and Translational Science Institute. Dr. Basaria reported serving as a consultant to Eli Lilly; his associates reported ties to AbbVie, Regenron, Sanofi, Eli Lilly, Function Promoting Therapies, and Hoffman-LaRoche.
For older men with low or low-normal testosterone levels, using testosterone gel doesn’t appear to worsen two key indicators of cardiovascular health – carotid artery intima-media thickness and coronary artery calcium deposition – according to results of a randomized trial published online Aug. 11 in JAMA.
Testosterone sales for “low T” have risen substantially in the past decade, even though the agent’s long-term effects on the cardiovascular health of older men are not yet known and even though there are concerns that testosterone supplementation may raise the risk of CVD events.
The Testosterone’s Effects on Atherosclerosis Progression in Aging Men (TEAAM) study was a randomized, placebo-controlled, double-blind clinical trial conducted at three U.S. medical centers to investigate this issue, said Dr. Shehzad Basaria, director of the Research Program in Men’s Health, Aging, and Metabolism, Boston Claude D. Pepper Older Americans Independence Center, Brigham and Women’s Hospital, Boston, and his associates.
Study participants were 306 community-dwelling men aged 60 and older enrolled during a 5-year period and randomly assigned to use either 1% testosterone gel (155 men) or placebo gel (151 men) every day for 3 years to treat low or low-normal testosterone levels. The mean age was 68 years. A total of 42% of the participants had hypertension, 15% had diabetes, 15% had known coronary artery disease, 27% were obese, and 43% were taking statin therapy. As expected, free testosterone levels increased in the testosterone group but did not change significantly in the placebo group.
The coprimary endpoint of rate of change in intima-media thickness, which was measured every 6 months, differed by just 0.0002 mm/year between the two study groups, a nonsignificant difference. The difference in the final measure of intima-media thickness taken at the conclusion of treatment, the other coprimary endpoint, was −10.8 Agatston units/year. Neither difference was significant. Similarly, the yearly rate of change in coronary artery calcium did not differ between the two groups, and neither did the final measure of coronary artery calcium at the conclusion of treatment. These findings persisted in several sensitivity analyses and across all subgroups of patients, such as men who were obese, men who had preexisting coronary artery disease, and men who had diabetes, the investigators said (JAMA. 2015 Aug 11. doi: 10.1001/jama.2015.8881). Notably, however, the two study groups also showed no significant differences in scores on the International Index of Erectile Function, either in the total score or in any of the domains of sexual function: erectile function, orgasmic function, sexual desire, intercourse satisfaction, or overall sexual satisfaction. Overall health-related quality of life also was no different between men who took active testosterone and men who took placebo, and there were no differences in the individual domains of physical function, bodily pain, vitality, role limitations due to physical problems, general health perceptions, emotional well-being, social function, or role limitations due to emotional problems.
There were few major cardiovascular disease events, and the numbers and types of events did not differ between the two study groups. Three men in the testosterone group and two in the placebo group had MIs, five in the testosterone group and two in the placebo group underwent coronary revascularization, three in the testosterone group and none in the placebo group had a stroke, and one in the testosterone group and none in the placebo group had a fatal cardiovascular disease (CVD) event. However, the investigators emphasized that this trial was not designed to assess testosterone’s effect on actual CVD events, and a substantially larger study population with many more events would be needed to examine this issue adequately. “These findings should not be interpreted as establishing cardiovascular safety of testosterone use in older men,” Dr. Basaria and his associates noted.
It is also important to note that this study assessed only two measured of cardiovascular health. Testosterone may well affect other measures, such as plaque stability, clot formation, salt retention, water retention, inflammation, “or other mechanisms that might render some men susceptible to early CVD events,” they said.
In addition, testosterone levels decreased over time in the men assigned to active testosterone gel. The researchers could not determine whether this was due to declining treatment adherence, changes in testosterone metabolism over time, progressive suppression of endogenous testosterone production because of the exogenous supplementation, or other factors.
This clinical trial was supported by Solvay Pharmaceuticals and AbbVie, which provided the testosterone and placebo gels used in the study; the Aurora Foundation; the Kronos Longevity Research Institute; the Boston Claude D. Pepper Older Americans Independence Center; and Boston University’s Clinical and Translational Science Institute. Dr. Basaria reported serving as a consultant to Eli Lilly; his associates reported ties to AbbVie, Regenron, Sanofi, Eli Lilly, Function Promoting Therapies, and Hoffman-LaRoche.
FROM JAMA
Intermittent hypoxemia linked to disability at 18 months
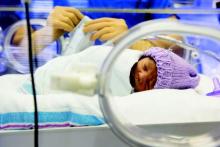
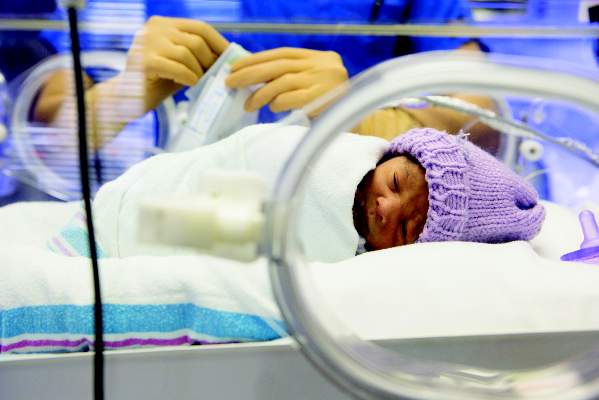
Intermittent episodes of hypoxemia lasting more than 1 minute during the first 2-3 months after extremely preterm birth are associated with severe disability at age 18 months, according to a report published online Aug. 11 in JAMA.
Almost all extremely preterm infants experience intermittent hypoxemia during their NICU stay, but the relationship between this hypoxemia and later neurodevelopmental problems is uncertain. To examine this issue, researchers performed a post-hoc analysis of data gathered in the multicenter Canadian Oxygen Trial, which compared the effects of lower (85%-89%) vs. higher (91%-95%) oxygen saturation targets in preterm infants. The post-hoc study focused on 1,019 infants born at 23-27 weeks’ gestation who survived to 36 weeks’ postmenstrual age, said Dr. Christian F. Poets of the department of neonatology at Tuebingen (Germany) University Hospital and his associates.
The investigators assessed whether hypoxemia correlated with the composite outcome of death after 36 weeks’ postmenstrual age or disability – motor impairment, cognitive or language delay, severe hearing loss requiring hearing aids or cochlear implants, bilateral blindness, or severe retinopathy of prematurity – assessed at a corrected age of 18 months. These adverse outcomes occurred in 43% of the study population.
The mean percentage of time with hypoxemia ranged from 0.4% in the lowest decile to 13.5% in the highest. The probability of each adverse outcome increased significantly as the percentage of time in hypoxemia increased. For the primary outcome of late death or disability, the relative risk was 1.53 among infants in the highest vs. the lowest decile. Stated another way, hypoxemic episodes were associated with death or severe disability in 56.5% of infants in the highest decile of hypoxemia exposure, compared with only 36.9% of those in the lowest decile. Each individual adverse outcome also followed this pattern of risk, the investigators said (JAMA. 2015;314(6):595-603. doi: 10.1001/jama.2015.8841). Only prolonged episodes of hypoxemia lasting more than 1 minute were found to correlate with adverse outcomes; shorter episodes did not. The risk of adverse outcomes increased as age at the time of hypoxemia episodes increased, so that the greatest risk was observed in infants whose hypoxemia occurred 9-10 weeks after birth.
The post-hoc design of this study can only generate hypotheses for future research rather than determine definitive conclusions, so it is not yet known whether prolonged intermittent hypoxemia contributes to neurodevelopmental impairment or is just a feature that denotes which infants are destined to develop impairments. If future studies determine that hypoxemia is a cause of adverse outcomes, it may be a preventable cause – perhaps with caffeine therapy, specific ventilator strategies, or doxapram administration, Dr. Poets and his associates said.
This study was funded exclusively by the Canadian Institutes of Health Research. Dr. Poets reported receiving research grants from Chiesi Farmaceurici SpA and Masimo.
Interventions to minimize hypoxemic episodes in this patient population are not yet warranted. Any such strategies, including the use of doxapram or high-dose caffeine therapy suggested by Dr. Poets and his associates, must first be evaluated in rigorous randomized clinical trials with long-term developmental outcome as the primary endpoint.
It is critical to minimize the exposure of these high-risk patients to therapies that may be useless or even harmful. Neonatal intensive care is littered with examples of treatments that were introduced based on observational data, only to be proven disastrous when tested properly in RCTs.
For now, well-tested treatments to improve developmental outcomes in extremely preterm infants, such as magnesium sulfate before birth, regular doses of caffeine, or developmental care interventions after discharge, should be maximized.
Dr. Lex W. Doyle is with the department of obstetrics and gynecology at Royal Women’s Hospital in Parkville, Australia. He reported having no relevant financial conflicts of interest. Dr. Doyle made these remarks in an accompanying editorial (JAMA. 2015;314(6):568-9. doi: 10.1001/jama.2015.9136).
Interventions to minimize hypoxemic episodes in this patient population are not yet warranted. Any such strategies, including the use of doxapram or high-dose caffeine therapy suggested by Dr. Poets and his associates, must first be evaluated in rigorous randomized clinical trials with long-term developmental outcome as the primary endpoint.
It is critical to minimize the exposure of these high-risk patients to therapies that may be useless or even harmful. Neonatal intensive care is littered with examples of treatments that were introduced based on observational data, only to be proven disastrous when tested properly in RCTs.
For now, well-tested treatments to improve developmental outcomes in extremely preterm infants, such as magnesium sulfate before birth, regular doses of caffeine, or developmental care interventions after discharge, should be maximized.
Dr. Lex W. Doyle is with the department of obstetrics and gynecology at Royal Women’s Hospital in Parkville, Australia. He reported having no relevant financial conflicts of interest. Dr. Doyle made these remarks in an accompanying editorial (JAMA. 2015;314(6):568-9. doi: 10.1001/jama.2015.9136).
Interventions to minimize hypoxemic episodes in this patient population are not yet warranted. Any such strategies, including the use of doxapram or high-dose caffeine therapy suggested by Dr. Poets and his associates, must first be evaluated in rigorous randomized clinical trials with long-term developmental outcome as the primary endpoint.
It is critical to minimize the exposure of these high-risk patients to therapies that may be useless or even harmful. Neonatal intensive care is littered with examples of treatments that were introduced based on observational data, only to be proven disastrous when tested properly in RCTs.
For now, well-tested treatments to improve developmental outcomes in extremely preterm infants, such as magnesium sulfate before birth, regular doses of caffeine, or developmental care interventions after discharge, should be maximized.
Dr. Lex W. Doyle is with the department of obstetrics and gynecology at Royal Women’s Hospital in Parkville, Australia. He reported having no relevant financial conflicts of interest. Dr. Doyle made these remarks in an accompanying editorial (JAMA. 2015;314(6):568-9. doi: 10.1001/jama.2015.9136).
Intermittent episodes of hypoxemia lasting more than 1 minute during the first 2-3 months after extremely preterm birth are associated with severe disability at age 18 months, according to a report published online Aug. 11 in JAMA.
Almost all extremely preterm infants experience intermittent hypoxemia during their NICU stay, but the relationship between this hypoxemia and later neurodevelopmental problems is uncertain. To examine this issue, researchers performed a post-hoc analysis of data gathered in the multicenter Canadian Oxygen Trial, which compared the effects of lower (85%-89%) vs. higher (91%-95%) oxygen saturation targets in preterm infants. The post-hoc study focused on 1,019 infants born at 23-27 weeks’ gestation who survived to 36 weeks’ postmenstrual age, said Dr. Christian F. Poets of the department of neonatology at Tuebingen (Germany) University Hospital and his associates.
The investigators assessed whether hypoxemia correlated with the composite outcome of death after 36 weeks’ postmenstrual age or disability – motor impairment, cognitive or language delay, severe hearing loss requiring hearing aids or cochlear implants, bilateral blindness, or severe retinopathy of prematurity – assessed at a corrected age of 18 months. These adverse outcomes occurred in 43% of the study population.
The mean percentage of time with hypoxemia ranged from 0.4% in the lowest decile to 13.5% in the highest. The probability of each adverse outcome increased significantly as the percentage of time in hypoxemia increased. For the primary outcome of late death or disability, the relative risk was 1.53 among infants in the highest vs. the lowest decile. Stated another way, hypoxemic episodes were associated with death or severe disability in 56.5% of infants in the highest decile of hypoxemia exposure, compared with only 36.9% of those in the lowest decile. Each individual adverse outcome also followed this pattern of risk, the investigators said (JAMA. 2015;314(6):595-603. doi: 10.1001/jama.2015.8841). Only prolonged episodes of hypoxemia lasting more than 1 minute were found to correlate with adverse outcomes; shorter episodes did not. The risk of adverse outcomes increased as age at the time of hypoxemia episodes increased, so that the greatest risk was observed in infants whose hypoxemia occurred 9-10 weeks after birth.
The post-hoc design of this study can only generate hypotheses for future research rather than determine definitive conclusions, so it is not yet known whether prolonged intermittent hypoxemia contributes to neurodevelopmental impairment or is just a feature that denotes which infants are destined to develop impairments. If future studies determine that hypoxemia is a cause of adverse outcomes, it may be a preventable cause – perhaps with caffeine therapy, specific ventilator strategies, or doxapram administration, Dr. Poets and his associates said.
This study was funded exclusively by the Canadian Institutes of Health Research. Dr. Poets reported receiving research grants from Chiesi Farmaceurici SpA and Masimo.
Intermittent episodes of hypoxemia lasting more than 1 minute during the first 2-3 months after extremely preterm birth are associated with severe disability at age 18 months, according to a report published online Aug. 11 in JAMA.
Almost all extremely preterm infants experience intermittent hypoxemia during their NICU stay, but the relationship between this hypoxemia and later neurodevelopmental problems is uncertain. To examine this issue, researchers performed a post-hoc analysis of data gathered in the multicenter Canadian Oxygen Trial, which compared the effects of lower (85%-89%) vs. higher (91%-95%) oxygen saturation targets in preterm infants. The post-hoc study focused on 1,019 infants born at 23-27 weeks’ gestation who survived to 36 weeks’ postmenstrual age, said Dr. Christian F. Poets of the department of neonatology at Tuebingen (Germany) University Hospital and his associates.
The investigators assessed whether hypoxemia correlated with the composite outcome of death after 36 weeks’ postmenstrual age or disability – motor impairment, cognitive or language delay, severe hearing loss requiring hearing aids or cochlear implants, bilateral blindness, or severe retinopathy of prematurity – assessed at a corrected age of 18 months. These adverse outcomes occurred in 43% of the study population.
The mean percentage of time with hypoxemia ranged from 0.4% in the lowest decile to 13.5% in the highest. The probability of each adverse outcome increased significantly as the percentage of time in hypoxemia increased. For the primary outcome of late death or disability, the relative risk was 1.53 among infants in the highest vs. the lowest decile. Stated another way, hypoxemic episodes were associated with death or severe disability in 56.5% of infants in the highest decile of hypoxemia exposure, compared with only 36.9% of those in the lowest decile. Each individual adverse outcome also followed this pattern of risk, the investigators said (JAMA. 2015;314(6):595-603. doi: 10.1001/jama.2015.8841). Only prolonged episodes of hypoxemia lasting more than 1 minute were found to correlate with adverse outcomes; shorter episodes did not. The risk of adverse outcomes increased as age at the time of hypoxemia episodes increased, so that the greatest risk was observed in infants whose hypoxemia occurred 9-10 weeks after birth.
The post-hoc design of this study can only generate hypotheses for future research rather than determine definitive conclusions, so it is not yet known whether prolonged intermittent hypoxemia contributes to neurodevelopmental impairment or is just a feature that denotes which infants are destined to develop impairments. If future studies determine that hypoxemia is a cause of adverse outcomes, it may be a preventable cause – perhaps with caffeine therapy, specific ventilator strategies, or doxapram administration, Dr. Poets and his associates said.
This study was funded exclusively by the Canadian Institutes of Health Research. Dr. Poets reported receiving research grants from Chiesi Farmaceurici SpA and Masimo.
FROM JAMA
Key clinical point: Intermittent episodes of hypoxemia in extremely preterm infants correlate with disability at age 18 months.
Major finding: Hypoxemic episodes were associated with death or severe disability in 56.5% of infants in the highest decile of hypoxemia exposure, vs. only 36.9% of those in the lowest decile.
Data source: A post-hoc analysis of data for 1,019 extremely preterm infants participating in the Canadian Oxygen Trial whose oxygen saturation in the NICU was closely monitored and recorded.
Disclosures: This study was funded exclusively by the Canadian Institutes of Health Research. Dr. Poets reported receiving research grants from Chiesi Farmaceurici SpA and Masimo.
Testosterone therapy didn’t boost atherosclerosis over 3 years
For older men with low or low-normal testosterone levels, using testosterone gel doesn’t appear to worsen two key indicators of cardiovascular health – carotid artery intima-media thickness and coronary artery calcium deposition – according to results of a randomized trial published online Aug. 11 in JAMA.
Testosterone sales for “low T” have risen substantially in the past decade, even though the agent’s long-term effects on the cardiovascular health of older men are not yet known and even though there are concerns that testosterone supplementation may raise the risk of CVD events.
The Testosterone’s Effects on Atherosclerosis Progression in Aging Men (TEAAM) study was a randomized, placebo-controlled, double-blind clinical trial conducted at three U.S. medical centers to investigate this issue, said Dr. Shehzad Basaria, director of the Research Program in Men’s Health, Aging, and Metabolism, Boston Claude D. Pepper Older Americans Independence Center, Brigham and Women’s Hospital, Boston, and his associates.
Study participants were 306 community-dwelling men aged 60 and older enrolled during a 5-year period and randomly assigned to use either 1% testosterone gel (155 men) or placebo gel (151 men) every day for 3 years to treat low or low-normal testosterone levels. The mean age was 68 years. A total of 42% of the participants had hypertension, 15% had diabetes, 15% had known coronary artery disease, 27% were obese, and 43% were taking statin therapy. As expected, free testosterone levels increased in the testosterone group but did not change significantly in the placebo group.
The coprimary endpoint of rate of change in intima-media thickness, which was measured every 6 months, differed by just 0.0002 mm/year between the two study groups, a nonsignificant difference. The difference in the final measure of intima-media thickness taken at the conclusion of treatment, the other coprimary endpoint, was −10.8 Agatston units/year. Neither difference was significant. Similarly, the yearly rate of change in coronary artery calcium did not differ between the two groups, and neither did the final measure of coronary artery calcium at the conclusion of treatment. These findings persisted in several sensitivity analyses and across all subgroups of patients, such as men who were obese, men who had preexisting coronary artery disease, and men who had diabetes, the investigators said (JAMA. 2015 Aug 11. doi: 10.1001/jama.2015.8881). Notably, however, the two study groups also showed no significant differences in scores on the International Index of Erectile Function, either in the total score or in any of the domains of sexual function: erectile function, orgasmic function, sexual desire, intercourse satisfaction, or overall sexual satisfaction. Overall health-related quality of life also was no different between men who took active testosterone and men who took placebo, and there were no differences in the individual domains of physical function, bodily pain, vitality, role limitations due to physical problems, general health perceptions, emotional well-being, social function, or role limitations due to emotional problems.
There were few major cardiovascular disease events, and the numbers and types of events did not differ between the two study groups. Three men in the testosterone group and two in the placebo group had MIs, five in the testosterone group and two in the placebo group underwent coronary revascularization, three in the testosterone group and none in the placebo group had a stroke, and one in the testosterone group and none in the placebo group had a fatal cardiovascular disease (CVD) event. However, the investigators emphasized that this trial was not designed to assess testosterone’s effect on actual CVD events, and a substantially larger study population with many more events would be needed to examine this issue adequately. “These findings should not be interpreted as establishing cardiovascular safety of testosterone use in older men,” Dr. Basaria and his associates noted.
It is also important to note that this study assessed only two measured of cardiovascular health. Testosterone may well affect other measures, such as plaque stability, clot formation, salt retention, water retention, inflammation, “or other mechanisms that might render some men susceptible to early CVD events,” they said.
In addition, testosterone levels decreased over time in the men assigned to active testosterone gel. The researchers could not determine whether this was due to declining treatment adherence, changes in testosterone metabolism over time, progressive suppression of endogenous testosterone production because of the exogenous supplementation, or other factors.
This clinical trial was supported by Solvay Pharmaceuticals and AbbVie, which provided the testosterone and placebo gels used in the study; the Aurora Foundation; the Kronos Longevity Research Institute; the Boston Claude D. Pepper Older Americans Independence Center; and Boston University’s Clinical and Translational Science Institute. Dr. Basaria reported serving as a consultant to Eli Lilly; his associates reported ties to AbbVie, Regenron, Sanofi, Eli Lilly, Function Promoting Therapies, and Hoffman-LaRoche.
For older men with low or low-normal testosterone levels, using testosterone gel doesn’t appear to worsen two key indicators of cardiovascular health – carotid artery intima-media thickness and coronary artery calcium deposition – according to results of a randomized trial published online Aug. 11 in JAMA.
Testosterone sales for “low T” have risen substantially in the past decade, even though the agent’s long-term effects on the cardiovascular health of older men are not yet known and even though there are concerns that testosterone supplementation may raise the risk of CVD events.
The Testosterone’s Effects on Atherosclerosis Progression in Aging Men (TEAAM) study was a randomized, placebo-controlled, double-blind clinical trial conducted at three U.S. medical centers to investigate this issue, said Dr. Shehzad Basaria, director of the Research Program in Men’s Health, Aging, and Metabolism, Boston Claude D. Pepper Older Americans Independence Center, Brigham and Women’s Hospital, Boston, and his associates.
Study participants were 306 community-dwelling men aged 60 and older enrolled during a 5-year period and randomly assigned to use either 1% testosterone gel (155 men) or placebo gel (151 men) every day for 3 years to treat low or low-normal testosterone levels. The mean age was 68 years. A total of 42% of the participants had hypertension, 15% had diabetes, 15% had known coronary artery disease, 27% were obese, and 43% were taking statin therapy. As expected, free testosterone levels increased in the testosterone group but did not change significantly in the placebo group.
The coprimary endpoint of rate of change in intima-media thickness, which was measured every 6 months, differed by just 0.0002 mm/year between the two study groups, a nonsignificant difference. The difference in the final measure of intima-media thickness taken at the conclusion of treatment, the other coprimary endpoint, was −10.8 Agatston units/year. Neither difference was significant. Similarly, the yearly rate of change in coronary artery calcium did not differ between the two groups, and neither did the final measure of coronary artery calcium at the conclusion of treatment. These findings persisted in several sensitivity analyses and across all subgroups of patients, such as men who were obese, men who had preexisting coronary artery disease, and men who had diabetes, the investigators said (JAMA. 2015 Aug 11. doi: 10.1001/jama.2015.8881). Notably, however, the two study groups also showed no significant differences in scores on the International Index of Erectile Function, either in the total score or in any of the domains of sexual function: erectile function, orgasmic function, sexual desire, intercourse satisfaction, or overall sexual satisfaction. Overall health-related quality of life also was no different between men who took active testosterone and men who took placebo, and there were no differences in the individual domains of physical function, bodily pain, vitality, role limitations due to physical problems, general health perceptions, emotional well-being, social function, or role limitations due to emotional problems.
There were few major cardiovascular disease events, and the numbers and types of events did not differ between the two study groups. Three men in the testosterone group and two in the placebo group had MIs, five in the testosterone group and two in the placebo group underwent coronary revascularization, three in the testosterone group and none in the placebo group had a stroke, and one in the testosterone group and none in the placebo group had a fatal cardiovascular disease (CVD) event. However, the investigators emphasized that this trial was not designed to assess testosterone’s effect on actual CVD events, and a substantially larger study population with many more events would be needed to examine this issue adequately. “These findings should not be interpreted as establishing cardiovascular safety of testosterone use in older men,” Dr. Basaria and his associates noted.
It is also important to note that this study assessed only two measured of cardiovascular health. Testosterone may well affect other measures, such as plaque stability, clot formation, salt retention, water retention, inflammation, “or other mechanisms that might render some men susceptible to early CVD events,” they said.
In addition, testosterone levels decreased over time in the men assigned to active testosterone gel. The researchers could not determine whether this was due to declining treatment adherence, changes in testosterone metabolism over time, progressive suppression of endogenous testosterone production because of the exogenous supplementation, or other factors.
This clinical trial was supported by Solvay Pharmaceuticals and AbbVie, which provided the testosterone and placebo gels used in the study; the Aurora Foundation; the Kronos Longevity Research Institute; the Boston Claude D. Pepper Older Americans Independence Center; and Boston University’s Clinical and Translational Science Institute. Dr. Basaria reported serving as a consultant to Eli Lilly; his associates reported ties to AbbVie, Regenron, Sanofi, Eli Lilly, Function Promoting Therapies, and Hoffman-LaRoche.
For older men with low or low-normal testosterone levels, using testosterone gel doesn’t appear to worsen two key indicators of cardiovascular health – carotid artery intima-media thickness and coronary artery calcium deposition – according to results of a randomized trial published online Aug. 11 in JAMA.
Testosterone sales for “low T” have risen substantially in the past decade, even though the agent’s long-term effects on the cardiovascular health of older men are not yet known and even though there are concerns that testosterone supplementation may raise the risk of CVD events.
The Testosterone’s Effects on Atherosclerosis Progression in Aging Men (TEAAM) study was a randomized, placebo-controlled, double-blind clinical trial conducted at three U.S. medical centers to investigate this issue, said Dr. Shehzad Basaria, director of the Research Program in Men’s Health, Aging, and Metabolism, Boston Claude D. Pepper Older Americans Independence Center, Brigham and Women’s Hospital, Boston, and his associates.
Study participants were 306 community-dwelling men aged 60 and older enrolled during a 5-year period and randomly assigned to use either 1% testosterone gel (155 men) or placebo gel (151 men) every day for 3 years to treat low or low-normal testosterone levels. The mean age was 68 years. A total of 42% of the participants had hypertension, 15% had diabetes, 15% had known coronary artery disease, 27% were obese, and 43% were taking statin therapy. As expected, free testosterone levels increased in the testosterone group but did not change significantly in the placebo group.
The coprimary endpoint of rate of change in intima-media thickness, which was measured every 6 months, differed by just 0.0002 mm/year between the two study groups, a nonsignificant difference. The difference in the final measure of intima-media thickness taken at the conclusion of treatment, the other coprimary endpoint, was −10.8 Agatston units/year. Neither difference was significant. Similarly, the yearly rate of change in coronary artery calcium did not differ between the two groups, and neither did the final measure of coronary artery calcium at the conclusion of treatment. These findings persisted in several sensitivity analyses and across all subgroups of patients, such as men who were obese, men who had preexisting coronary artery disease, and men who had diabetes, the investigators said (JAMA. 2015 Aug 11. doi: 10.1001/jama.2015.8881). Notably, however, the two study groups also showed no significant differences in scores on the International Index of Erectile Function, either in the total score or in any of the domains of sexual function: erectile function, orgasmic function, sexual desire, intercourse satisfaction, or overall sexual satisfaction. Overall health-related quality of life also was no different between men who took active testosterone and men who took placebo, and there were no differences in the individual domains of physical function, bodily pain, vitality, role limitations due to physical problems, general health perceptions, emotional well-being, social function, or role limitations due to emotional problems.
There were few major cardiovascular disease events, and the numbers and types of events did not differ between the two study groups. Three men in the testosterone group and two in the placebo group had MIs, five in the testosterone group and two in the placebo group underwent coronary revascularization, three in the testosterone group and none in the placebo group had a stroke, and one in the testosterone group and none in the placebo group had a fatal cardiovascular disease (CVD) event. However, the investigators emphasized that this trial was not designed to assess testosterone’s effect on actual CVD events, and a substantially larger study population with many more events would be needed to examine this issue adequately. “These findings should not be interpreted as establishing cardiovascular safety of testosterone use in older men,” Dr. Basaria and his associates noted.
It is also important to note that this study assessed only two measured of cardiovascular health. Testosterone may well affect other measures, such as plaque stability, clot formation, salt retention, water retention, inflammation, “or other mechanisms that might render some men susceptible to early CVD events,” they said.
In addition, testosterone levels decreased over time in the men assigned to active testosterone gel. The researchers could not determine whether this was due to declining treatment adherence, changes in testosterone metabolism over time, progressive suppression of endogenous testosterone production because of the exogenous supplementation, or other factors.
This clinical trial was supported by Solvay Pharmaceuticals and AbbVie, which provided the testosterone and placebo gels used in the study; the Aurora Foundation; the Kronos Longevity Research Institute; the Boston Claude D. Pepper Older Americans Independence Center; and Boston University’s Clinical and Translational Science Institute. Dr. Basaria reported serving as a consultant to Eli Lilly; his associates reported ties to AbbVie, Regenron, Sanofi, Eli Lilly, Function Promoting Therapies, and Hoffman-LaRoche.
FROM JAMA
Key clinical point: For older men who used testosterone gel, 3 years of treatment did not worsen carotid artery intima-media thickness or coronary artery calcium deposition.
Major finding: The rate of change in intima-media thickness, which was measured every 6 months, was not significantly different between the two study groups, nor was the final measure of intima-media thickness taken at the conclusion of treatment.
Data source: A randomized, placebo-controlled, double-blind trial in 306 men aged 60 and older who were followed for 3 years.
Disclosures: This clinical trial was supported by Solvay Pharmaceuticals and AbbVie, which provided the testosterone and placebo gels used in the study; the Aurora Foundation; the Kronos Longevity Research Institute; the Boston Claude D. Pepper Older Americans Independence Center; and Boston University’s Clinical and Translational Science Institute. Dr. Basaria reported serving as a consultant to Eli Lilly; his associates reported ties to AbbVie, Regenron, Sanofi, Eli Lilly, Function Promoting Therapies, and Hoffman-LaRoche.
CSF markers differentiate Parkinson’s, predict related dementia
Certain cerebrospinal fluid biomarkers may help differentiate Parkinson’s disease from other causes of parkinsonism and also predict which patients will later develop Parkinson’s-related dementia, according to a report published online Aug. 10 in JAMA Neurology.
In a prospective, population-based, longitudinal study, Dr. David C. Bäckström of Umeå (Sweden) University and his coinvestigators examined several biomarkers in cerebrospinal fluid (CSF) samples from 128 patients with new-onset, treatment-naive parkinsonism who resided in a defined geographic area of Sweden. These patients agreed to CSF collection by lumbar puncture at baseline, and some agreed to serial sampling. All were followed yearly for 5-9 years, undergoing periodic comprehensive neuropsychological testing. At their most recent follow-up visit, 104 of these participants had been diagnosed as having Parkinson’s disease, 11 as having multiple system atrophy, and 13 as having progressive supranuclear palsy. For reference, CSF samples were also obtained from 30 demographically similar but neurologically healthy control subjects.
High levels of neurofilament light chain protein (NFL) together with low levels of amyloid-beta1-42 distinguished progressive supranuclear palsy from PD at baseline as well as 1 year later, after patients had been receiving dopaminergic therapy for months. Even elevated NFL alone was useful for differentiating progressive supranuclear palsy from PD: A baseline NFL exceeding 2,020 ng/L had a sensitivity of 75% and a specificity of 83% in distinguishing the two disorders. If measured over time, the tendency of NFL to gradually increase in progressive supranuclear palsy was even more indicative: At 1 year, an NFL exceeding 2,916 ng/L had a sensitivity of 89% and a specificity of 93% for identifying progressive supranuclear palsy. However, CSF samples at baseline or could not distinguish multiple system atrophy from Parkinson’s disease.
In addition, an early pattern of high NFL, low amyloid-beta1-42, and high heart fatty-acid-binding protein (HFABP) predicted the later onset of PD-related dementia with 90% sensitivity and 71% specificity. Patients whose baseline NFL exceeded 1,100 ng/L, whose amyloid-beta1-42 was less than 626 ng/L, and whose HFABP exceeded 500 ng/L were, respectively, at 2.6, 2.8, and 2.8 times higher risk of future PD-related dementia than were other patients with parkinsonism. And low amyloid-beta1-42 alone predicted PD-related dementia for up to 9 years after initial presentation of parkinsonism, Dr. Bäckström and his associates said (JAMA Neurol. 2015 Aug 10. doi:10.1001/jamaneurol.2015.1449).
Taken together, these findings suggest that CSF testing can be clinically useful to differentiate PD from other disorders and as a presymptomatic screening tool for PD-related dementia, they added.
In contrast, other CSF biomarkers examined in this study, including total tau, phosphorylated tau, and alpha-synuclein, were not helpful in identifying PD or predicting PD-related dementia.
The Swedish Medical Research Council, Erling Persson Foundation, Parkinson Foundation in Sweden, Umeå University, Västerbotten County Council, King Gustaf V and Queen Victoria Freemason Foundation, Swedish Parkinson Foundation, Kempe Foundation, and Swedish Parkinson’s Disease Association funded the study. Dr. Bäckström reported having no conflicts of interest. One of his associates reported ties to numerous pharmaceutical companies.
Certain cerebrospinal fluid biomarkers may help differentiate Parkinson’s disease from other causes of parkinsonism and also predict which patients will later develop Parkinson’s-related dementia, according to a report published online Aug. 10 in JAMA Neurology.
In a prospective, population-based, longitudinal study, Dr. David C. Bäckström of Umeå (Sweden) University and his coinvestigators examined several biomarkers in cerebrospinal fluid (CSF) samples from 128 patients with new-onset, treatment-naive parkinsonism who resided in a defined geographic area of Sweden. These patients agreed to CSF collection by lumbar puncture at baseline, and some agreed to serial sampling. All were followed yearly for 5-9 years, undergoing periodic comprehensive neuropsychological testing. At their most recent follow-up visit, 104 of these participants had been diagnosed as having Parkinson’s disease, 11 as having multiple system atrophy, and 13 as having progressive supranuclear palsy. For reference, CSF samples were also obtained from 30 demographically similar but neurologically healthy control subjects.
High levels of neurofilament light chain protein (NFL) together with low levels of amyloid-beta1-42 distinguished progressive supranuclear palsy from PD at baseline as well as 1 year later, after patients had been receiving dopaminergic therapy for months. Even elevated NFL alone was useful for differentiating progressive supranuclear palsy from PD: A baseline NFL exceeding 2,020 ng/L had a sensitivity of 75% and a specificity of 83% in distinguishing the two disorders. If measured over time, the tendency of NFL to gradually increase in progressive supranuclear palsy was even more indicative: At 1 year, an NFL exceeding 2,916 ng/L had a sensitivity of 89% and a specificity of 93% for identifying progressive supranuclear palsy. However, CSF samples at baseline or could not distinguish multiple system atrophy from Parkinson’s disease.
In addition, an early pattern of high NFL, low amyloid-beta1-42, and high heart fatty-acid-binding protein (HFABP) predicted the later onset of PD-related dementia with 90% sensitivity and 71% specificity. Patients whose baseline NFL exceeded 1,100 ng/L, whose amyloid-beta1-42 was less than 626 ng/L, and whose HFABP exceeded 500 ng/L were, respectively, at 2.6, 2.8, and 2.8 times higher risk of future PD-related dementia than were other patients with parkinsonism. And low amyloid-beta1-42 alone predicted PD-related dementia for up to 9 years after initial presentation of parkinsonism, Dr. Bäckström and his associates said (JAMA Neurol. 2015 Aug 10. doi:10.1001/jamaneurol.2015.1449).
Taken together, these findings suggest that CSF testing can be clinically useful to differentiate PD from other disorders and as a presymptomatic screening tool for PD-related dementia, they added.
In contrast, other CSF biomarkers examined in this study, including total tau, phosphorylated tau, and alpha-synuclein, were not helpful in identifying PD or predicting PD-related dementia.
The Swedish Medical Research Council, Erling Persson Foundation, Parkinson Foundation in Sweden, Umeå University, Västerbotten County Council, King Gustaf V and Queen Victoria Freemason Foundation, Swedish Parkinson Foundation, Kempe Foundation, and Swedish Parkinson’s Disease Association funded the study. Dr. Bäckström reported having no conflicts of interest. One of his associates reported ties to numerous pharmaceutical companies.
Certain cerebrospinal fluid biomarkers may help differentiate Parkinson’s disease from other causes of parkinsonism and also predict which patients will later develop Parkinson’s-related dementia, according to a report published online Aug. 10 in JAMA Neurology.
In a prospective, population-based, longitudinal study, Dr. David C. Bäckström of Umeå (Sweden) University and his coinvestigators examined several biomarkers in cerebrospinal fluid (CSF) samples from 128 patients with new-onset, treatment-naive parkinsonism who resided in a defined geographic area of Sweden. These patients agreed to CSF collection by lumbar puncture at baseline, and some agreed to serial sampling. All were followed yearly for 5-9 years, undergoing periodic comprehensive neuropsychological testing. At their most recent follow-up visit, 104 of these participants had been diagnosed as having Parkinson’s disease, 11 as having multiple system atrophy, and 13 as having progressive supranuclear palsy. For reference, CSF samples were also obtained from 30 demographically similar but neurologically healthy control subjects.
High levels of neurofilament light chain protein (NFL) together with low levels of amyloid-beta1-42 distinguished progressive supranuclear palsy from PD at baseline as well as 1 year later, after patients had been receiving dopaminergic therapy for months. Even elevated NFL alone was useful for differentiating progressive supranuclear palsy from PD: A baseline NFL exceeding 2,020 ng/L had a sensitivity of 75% and a specificity of 83% in distinguishing the two disorders. If measured over time, the tendency of NFL to gradually increase in progressive supranuclear palsy was even more indicative: At 1 year, an NFL exceeding 2,916 ng/L had a sensitivity of 89% and a specificity of 93% for identifying progressive supranuclear palsy. However, CSF samples at baseline or could not distinguish multiple system atrophy from Parkinson’s disease.
In addition, an early pattern of high NFL, low amyloid-beta1-42, and high heart fatty-acid-binding protein (HFABP) predicted the later onset of PD-related dementia with 90% sensitivity and 71% specificity. Patients whose baseline NFL exceeded 1,100 ng/L, whose amyloid-beta1-42 was less than 626 ng/L, and whose HFABP exceeded 500 ng/L were, respectively, at 2.6, 2.8, and 2.8 times higher risk of future PD-related dementia than were other patients with parkinsonism. And low amyloid-beta1-42 alone predicted PD-related dementia for up to 9 years after initial presentation of parkinsonism, Dr. Bäckström and his associates said (JAMA Neurol. 2015 Aug 10. doi:10.1001/jamaneurol.2015.1449).
Taken together, these findings suggest that CSF testing can be clinically useful to differentiate PD from other disorders and as a presymptomatic screening tool for PD-related dementia, they added.
In contrast, other CSF biomarkers examined in this study, including total tau, phosphorylated tau, and alpha-synuclein, were not helpful in identifying PD or predicting PD-related dementia.
The Swedish Medical Research Council, Erling Persson Foundation, Parkinson Foundation in Sweden, Umeå University, Västerbotten County Council, King Gustaf V and Queen Victoria Freemason Foundation, Swedish Parkinson Foundation, Kempe Foundation, and Swedish Parkinson’s Disease Association funded the study. Dr. Bäckström reported having no conflicts of interest. One of his associates reported ties to numerous pharmaceutical companies.
FROM JAMA NEUROLOGY
Key clinical point: CSF markers may help differentiate Parkinson’s disease from other sources of parkinsonism.
Major finding: A pattern of high NFL, low amyloid-beta1-42, and high HFABP levels in CSF predicted the later onset of Parkinson’s disease-related dementia with 90% sensitivity and 71% specificity.
Data source: A prospective, population-based, longitudinal study involving 128 Swedish patients with treatment-naive parkinsonism who were followed for 5-9 years.
Disclosures: The Swedish Medical Research Council, Erling Persson Foundation, Parkinson Foundation in Sweden, Umeå University, Västerbotten County Council, King Gustaf V and Queen Victoria Freemason Foundation, Swedish Parkinson Foundation, Kempe Foundation, and Swedish Parkinson’s Disease Association funded the study. Dr. Bäckström reported having no conflicts of interest. One of his associates reported ties to numerous pharmaceutical companies.
Integrating behavioral with primary care benefits children, adolescents
Integrating care for behavioral/mental problems into primary health care is beneficial for children and adolescents, as it has been shown to be for adults, according to a report published online Aug. 10 in JAMA Pediatrics.
A meta-analysis of 79 randomized clinical trials involving nearly 25,000 patients established the clinical superiority of collaborative care in the primary care setting over usual care for adults with depression or anxiety, but until now there hasn’t been a similar attempt to compare these two approaches in the pediatric population.
“We conducted a systematic review and meta-analysis of the literature to address the empirical question: Does integrated behavioral health and primary medical care for children and adolescents lead to improved behavioral health outcomes, compared with usual care?” said Joan Rosenbaum Asarnow, Ph.D., of the Semel Institute for Neuroscience and Human Behavior, department of psychiatry and biobehavioral sciences, University of California, Los Angeles, and her associates.
The investigators included 31 randomized clinical trials in their analysis, with a total population of 13,129 patients aged 1-21 years: the sample sizes of the individual trials ranged from 28 to 3,111 participants. These studies compared usual primary care against integrated primary care that included cognitive-behavioral therapy, parenting education, interpersonal psychotherapy, medication management, substance-use treatment, peer counseling, and motivational interviewing. The most common behavioral/mental disorders that these children and adolescents sought treatment for were depression, anxiety, somatic concerns, attention deficit–hyperactivity disorder (ADHD), substance use, and the catch-all category of “behavior problems.”
Integrated behavioral/mental health care within the primary care setting consistently showed a small-to-medium but significant advantage over usual care. In particular, collaborative care involving a team of primary care physicians, care managers, and mental health specialists was very beneficial, “with a 73% probability that a randomly selected youth would experience better outcomes after receiving collaborative care than a randomly selected youth receiving usual care,” Dr. Asarnow and her associates wrote (JAMA Pediatr. 2015 Aug 10 doi: 101001/jamapediatrics.2015.1141).
The Patient Protection and Affordable Care Act incentivizes this integrated approach, in part by identifying behavioral health treatment as an “essential health benefit” and by increasing insurance coverage for behavioral/mental problems (Adm Policy Ment Health. 2013;40[4]:258-63). This meta-analysis documents the benefits of such an integrated approach, which enhances confidence that “we are on a course that will yield improvements in the lives of youth and families,” the investigators added.
This study was supported in part by the National Institute of Mental Health and the Society for Clinical Child and Adolescent Psychology. Dr. Asarnow and her associates reported having no relevant financial disclosures.
Integrating care for behavioral/mental problems into primary health care is beneficial for children and adolescents, as it has been shown to be for adults, according to a report published online Aug. 10 in JAMA Pediatrics.
A meta-analysis of 79 randomized clinical trials involving nearly 25,000 patients established the clinical superiority of collaborative care in the primary care setting over usual care for adults with depression or anxiety, but until now there hasn’t been a similar attempt to compare these two approaches in the pediatric population.
“We conducted a systematic review and meta-analysis of the literature to address the empirical question: Does integrated behavioral health and primary medical care for children and adolescents lead to improved behavioral health outcomes, compared with usual care?” said Joan Rosenbaum Asarnow, Ph.D., of the Semel Institute for Neuroscience and Human Behavior, department of psychiatry and biobehavioral sciences, University of California, Los Angeles, and her associates.
The investigators included 31 randomized clinical trials in their analysis, with a total population of 13,129 patients aged 1-21 years: the sample sizes of the individual trials ranged from 28 to 3,111 participants. These studies compared usual primary care against integrated primary care that included cognitive-behavioral therapy, parenting education, interpersonal psychotherapy, medication management, substance-use treatment, peer counseling, and motivational interviewing. The most common behavioral/mental disorders that these children and adolescents sought treatment for were depression, anxiety, somatic concerns, attention deficit–hyperactivity disorder (ADHD), substance use, and the catch-all category of “behavior problems.”
Integrated behavioral/mental health care within the primary care setting consistently showed a small-to-medium but significant advantage over usual care. In particular, collaborative care involving a team of primary care physicians, care managers, and mental health specialists was very beneficial, “with a 73% probability that a randomly selected youth would experience better outcomes after receiving collaborative care than a randomly selected youth receiving usual care,” Dr. Asarnow and her associates wrote (JAMA Pediatr. 2015 Aug 10 doi: 101001/jamapediatrics.2015.1141).
The Patient Protection and Affordable Care Act incentivizes this integrated approach, in part by identifying behavioral health treatment as an “essential health benefit” and by increasing insurance coverage for behavioral/mental problems (Adm Policy Ment Health. 2013;40[4]:258-63). This meta-analysis documents the benefits of such an integrated approach, which enhances confidence that “we are on a course that will yield improvements in the lives of youth and families,” the investigators added.
This study was supported in part by the National Institute of Mental Health and the Society for Clinical Child and Adolescent Psychology. Dr. Asarnow and her associates reported having no relevant financial disclosures.
Integrating care for behavioral/mental problems into primary health care is beneficial for children and adolescents, as it has been shown to be for adults, according to a report published online Aug. 10 in JAMA Pediatrics.
A meta-analysis of 79 randomized clinical trials involving nearly 25,000 patients established the clinical superiority of collaborative care in the primary care setting over usual care for adults with depression or anxiety, but until now there hasn’t been a similar attempt to compare these two approaches in the pediatric population.
“We conducted a systematic review and meta-analysis of the literature to address the empirical question: Does integrated behavioral health and primary medical care for children and adolescents lead to improved behavioral health outcomes, compared with usual care?” said Joan Rosenbaum Asarnow, Ph.D., of the Semel Institute for Neuroscience and Human Behavior, department of psychiatry and biobehavioral sciences, University of California, Los Angeles, and her associates.
The investigators included 31 randomized clinical trials in their analysis, with a total population of 13,129 patients aged 1-21 years: the sample sizes of the individual trials ranged from 28 to 3,111 participants. These studies compared usual primary care against integrated primary care that included cognitive-behavioral therapy, parenting education, interpersonal psychotherapy, medication management, substance-use treatment, peer counseling, and motivational interviewing. The most common behavioral/mental disorders that these children and adolescents sought treatment for were depression, anxiety, somatic concerns, attention deficit–hyperactivity disorder (ADHD), substance use, and the catch-all category of “behavior problems.”
Integrated behavioral/mental health care within the primary care setting consistently showed a small-to-medium but significant advantage over usual care. In particular, collaborative care involving a team of primary care physicians, care managers, and mental health specialists was very beneficial, “with a 73% probability that a randomly selected youth would experience better outcomes after receiving collaborative care than a randomly selected youth receiving usual care,” Dr. Asarnow and her associates wrote (JAMA Pediatr. 2015 Aug 10 doi: 101001/jamapediatrics.2015.1141).
The Patient Protection and Affordable Care Act incentivizes this integrated approach, in part by identifying behavioral health treatment as an “essential health benefit” and by increasing insurance coverage for behavioral/mental problems (Adm Policy Ment Health. 2013;40[4]:258-63). This meta-analysis documents the benefits of such an integrated approach, which enhances confidence that “we are on a course that will yield improvements in the lives of youth and families,” the investigators added.
This study was supported in part by the National Institute of Mental Health and the Society for Clinical Child and Adolescent Psychology. Dr. Asarnow and her associates reported having no relevant financial disclosures.
FROM JAMA PEDIATRICS
Key clinical point: Integrating care for behavioral/mental problems into primary care benefits pediatric patients, as it does adults.
Major finding: There was a 73% probability that a randomly selected youth would show better behavioral/mental outcomes after receiving collaborative care than a randomly selected youth receiving usual care.
Data source: A systematic review and meta-analysis of 31 randomized clinical trials comparing integrated behavioral and primary health care against usual care in 13,129 children and adolescents.
Disclosures: This study was supported in part by the National Institute of Mental Health and the Society for Clinical Child and Adolescent Psychology. Dr. Asarnow and her associates reported having no relevant financial disclosures.
Limited resection inferior for elderly with early NSCLC
Limited resection is inferior to lobectomy for older patients with early-stage invasive NSCLC, yielding lower overall survival and cancer-specific survival, according to a study published online Aug. 3 in Journal of Clinical Oncology.
Limited resection – wedge resection or segmentectomy – is increasingly chosen over lobectomy for patients older than 65 because it is thought to yield equivalent survival among patients who are already near the end of their lives and to cut down on perioperative and postoperative complications. Moreover, the number of these surgeries is expected to increase substantially when the recently released U.S. Preventive Services Task Force recommendations for lung cancer screening are fully implemented, said Dr. Rajwanth R. Veluswamy of the division of hematology and medical oncology, Mount Sinai University, New York, and his associates.
However, the evidence supporting the equivalency of limited resection to lobectomy is scant and not of high quality. To examine the issue more closely, the investigators analyzed survival outcomes for 3,147 patients aged 65 and older who were included in the nationally representative population-based Surveillance, Epidemiology, and End Results (SEER) database. These patients had stage 1A NSCLC of 2 cm or less in diameter and were treated surgically in 1998 through 2009.
Limited resection was found to be inferior to lobectomy regarding overall survival (HR, 1.21) and lung cancer–specific survival (HR, 1.66) among patients with invasive adenocarcinoma. Limited resection also was inferior to lobectomy regarding overall survival (HR, 1.21) and lung cancer–specific survival (HR, 1.41) among patients with squamous cell carcinoma, Dr. Veluswamy and his associates said (J Clin Oncol. 2015 Aug. 3. doi:10.1200/JCO.2014.60.6624).
“Our findings should help decide the best treatment for older patients by balancing the potential short- and long-term risks of limited resection versus lobectomy,” they said.
The investigators added that they focused on patients older than age 65 because that is the age group most frequently affected by invasive NSCLC and most likely to be considered for limited resection. These results cannot be extrapolated to younger patients because they have longer life expectancies and thus higher risk of recurrence after “limited” treatment.
The U.S. Agency for Healthcare Research and Quality supported the study. Dr. Veluswamy had no disclosures; his associates reported ties to Pfizer, Otsuka, Teva Neuroscience, EHE International, United BioSource, Ethicon, Covidien, Genentech, IMS Health, Merck, Bristol-Myers Squibb, Quintiles, and Sanofi.
Limited resection is inferior to lobectomy for older patients with early-stage invasive NSCLC, yielding lower overall survival and cancer-specific survival, according to a study published online Aug. 3 in Journal of Clinical Oncology.
Limited resection – wedge resection or segmentectomy – is increasingly chosen over lobectomy for patients older than 65 because it is thought to yield equivalent survival among patients who are already near the end of their lives and to cut down on perioperative and postoperative complications. Moreover, the number of these surgeries is expected to increase substantially when the recently released U.S. Preventive Services Task Force recommendations for lung cancer screening are fully implemented, said Dr. Rajwanth R. Veluswamy of the division of hematology and medical oncology, Mount Sinai University, New York, and his associates.
However, the evidence supporting the equivalency of limited resection to lobectomy is scant and not of high quality. To examine the issue more closely, the investigators analyzed survival outcomes for 3,147 patients aged 65 and older who were included in the nationally representative population-based Surveillance, Epidemiology, and End Results (SEER) database. These patients had stage 1A NSCLC of 2 cm or less in diameter and were treated surgically in 1998 through 2009.
Limited resection was found to be inferior to lobectomy regarding overall survival (HR, 1.21) and lung cancer–specific survival (HR, 1.66) among patients with invasive adenocarcinoma. Limited resection also was inferior to lobectomy regarding overall survival (HR, 1.21) and lung cancer–specific survival (HR, 1.41) among patients with squamous cell carcinoma, Dr. Veluswamy and his associates said (J Clin Oncol. 2015 Aug. 3. doi:10.1200/JCO.2014.60.6624).
“Our findings should help decide the best treatment for older patients by balancing the potential short- and long-term risks of limited resection versus lobectomy,” they said.
The investigators added that they focused on patients older than age 65 because that is the age group most frequently affected by invasive NSCLC and most likely to be considered for limited resection. These results cannot be extrapolated to younger patients because they have longer life expectancies and thus higher risk of recurrence after “limited” treatment.
The U.S. Agency for Healthcare Research and Quality supported the study. Dr. Veluswamy had no disclosures; his associates reported ties to Pfizer, Otsuka, Teva Neuroscience, EHE International, United BioSource, Ethicon, Covidien, Genentech, IMS Health, Merck, Bristol-Myers Squibb, Quintiles, and Sanofi.
Limited resection is inferior to lobectomy for older patients with early-stage invasive NSCLC, yielding lower overall survival and cancer-specific survival, according to a study published online Aug. 3 in Journal of Clinical Oncology.
Limited resection – wedge resection or segmentectomy – is increasingly chosen over lobectomy for patients older than 65 because it is thought to yield equivalent survival among patients who are already near the end of their lives and to cut down on perioperative and postoperative complications. Moreover, the number of these surgeries is expected to increase substantially when the recently released U.S. Preventive Services Task Force recommendations for lung cancer screening are fully implemented, said Dr. Rajwanth R. Veluswamy of the division of hematology and medical oncology, Mount Sinai University, New York, and his associates.
However, the evidence supporting the equivalency of limited resection to lobectomy is scant and not of high quality. To examine the issue more closely, the investigators analyzed survival outcomes for 3,147 patients aged 65 and older who were included in the nationally representative population-based Surveillance, Epidemiology, and End Results (SEER) database. These patients had stage 1A NSCLC of 2 cm or less in diameter and were treated surgically in 1998 through 2009.
Limited resection was found to be inferior to lobectomy regarding overall survival (HR, 1.21) and lung cancer–specific survival (HR, 1.66) among patients with invasive adenocarcinoma. Limited resection also was inferior to lobectomy regarding overall survival (HR, 1.21) and lung cancer–specific survival (HR, 1.41) among patients with squamous cell carcinoma, Dr. Veluswamy and his associates said (J Clin Oncol. 2015 Aug. 3. doi:10.1200/JCO.2014.60.6624).
“Our findings should help decide the best treatment for older patients by balancing the potential short- and long-term risks of limited resection versus lobectomy,” they said.
The investigators added that they focused on patients older than age 65 because that is the age group most frequently affected by invasive NSCLC and most likely to be considered for limited resection. These results cannot be extrapolated to younger patients because they have longer life expectancies and thus higher risk of recurrence after “limited” treatment.
The U.S. Agency for Healthcare Research and Quality supported the study. Dr. Veluswamy had no disclosures; his associates reported ties to Pfizer, Otsuka, Teva Neuroscience, EHE International, United BioSource, Ethicon, Covidien, Genentech, IMS Health, Merck, Bristol-Myers Squibb, Quintiles, and Sanofi.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Limited resection is inferior to lobectomy for older patients with early-stage invasive NSCLC.
Major finding: Limited resection was found to be inferior to lobectomy regarding overall survival (HR, 1.21) and lung cancer–specific survival (HR, 1.66) among patients with invasive adenocarcinoma. Limited resection also was inferior to lobectomy regarding overall survival (HR, 1.21) and lung cancer–specific survival (HR, 1.41) among patients with squamous cell carcinoma, .
Data source: An analysis of population-based data of survival outcomes in 3,147 older patients with early-stage invasive NSCLC treated surgically in 1998-2009.
Disclosures: The U.S. Agency for Healthcare Research and Quality supported the study. Dr. Veluswamy had no disclosures; his associates reported ties to Pfizer, Otsuka, Teva Neuroscience, EHE International, United BioSource, Ethicon, Covidien, Genentech, IMS Health, Merck, Bristol-Myers Squibb, Quintiles, and Sanofi.
Treatment of choice for NLPHL? Involved-field RT alone
Involved-field radiotherapy alone should be the treatment of choice for stage 1A nodular lymphocyte-predominant Hodgkin lymphoma because it yields similar survival outcomes but fewer toxic effects than other therapies, investigators reported online Aug. 3 in Journal of Clinical Oncology.
Few studies have examined the long-term outcomes of patients who have this generally indolent malignancy, even though late treatment-associated effects are their major cause of death, said Dr. Dennis A. Eichenauer and his associates at University Hospital Cologne and University Hospital Münster, both in Germany.
To assess long-term outcomes in nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL), the investigators analyzed data for 256 patients who participated in seven prospective randomized clinical trials comparing various treatments during a 21-year period, who were followed for a median of 91 months. The study participants’ median age at diagnosis was 39 years (range, 16-75 years), and all achieved remission with therapy. They were categorized by treatment type: 108 received involved-field radiotherapy (IF-RT) alone; 49 received extended-field radiotherapy (EF-RT) alone; 72 received combined modality treatment involving various combinations of doxorubicin, bleomycin, vinblastine, dacarbazine, IF-RT, EF-RT, and/or rituximab; and 27 received rituximab alone.
At 8 years, progression-free survival was 91.9% with IF-RT, 84.3% with EF-RT, and 88.5% with combined modalities, which are nonsignificant differences. Overall survival was 99.0%, 95.7%, and 98.6%, respectively, which also are nonsignificant differences. Rituximab alone was markedly less effective than the other treatments, with a 4-year progression-free survival of only 81.0%. “Compared with patients who received IF-RT, patients treated with rituximab had a hazard ratio of 4.99 for relapse,” Dr. Eichenauer and his associates wrote (J Clin Oncol. 2015 Aug 3 [doi: 10.1200/JCO. 2014.60.4363]).
Only 3.7% of patients treated with IF-RT developed a second malignancy during follow-up, compared with 11.1% of those treated with combined modalities, 6.1% of those treated with EF-RT, and 7.4% of those treated with rituximab. The rate of relapse was markedly higher with rituximab than with the other treatments.
The main study findings are twofold. First, IF-RT was at least as effective as other treatments in controlling NLPHL, was less toxic acutely, and carried similar or reduced risks of late adverse effects such as relapse and second malignancies. It should be considered the first-line treatment of choice. Second, rituximab alone should not be used routinely in these patients because it yields poorer survival outcomes and a higher relapse rate. “However, it might represent an option for individual patients, such as young women with abdominal disease, to avoid gonadotoxic effects of radiotherapy,” the investigators said.
Involved-field radiotherapy alone should be the treatment of choice for stage 1A nodular lymphocyte-predominant Hodgkin lymphoma because it yields similar survival outcomes but fewer toxic effects than other therapies, investigators reported online Aug. 3 in Journal of Clinical Oncology.
Few studies have examined the long-term outcomes of patients who have this generally indolent malignancy, even though late treatment-associated effects are their major cause of death, said Dr. Dennis A. Eichenauer and his associates at University Hospital Cologne and University Hospital Münster, both in Germany.
To assess long-term outcomes in nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL), the investigators analyzed data for 256 patients who participated in seven prospective randomized clinical trials comparing various treatments during a 21-year period, who were followed for a median of 91 months. The study participants’ median age at diagnosis was 39 years (range, 16-75 years), and all achieved remission with therapy. They were categorized by treatment type: 108 received involved-field radiotherapy (IF-RT) alone; 49 received extended-field radiotherapy (EF-RT) alone; 72 received combined modality treatment involving various combinations of doxorubicin, bleomycin, vinblastine, dacarbazine, IF-RT, EF-RT, and/or rituximab; and 27 received rituximab alone.
At 8 years, progression-free survival was 91.9% with IF-RT, 84.3% with EF-RT, and 88.5% with combined modalities, which are nonsignificant differences. Overall survival was 99.0%, 95.7%, and 98.6%, respectively, which also are nonsignificant differences. Rituximab alone was markedly less effective than the other treatments, with a 4-year progression-free survival of only 81.0%. “Compared with patients who received IF-RT, patients treated with rituximab had a hazard ratio of 4.99 for relapse,” Dr. Eichenauer and his associates wrote (J Clin Oncol. 2015 Aug 3 [doi: 10.1200/JCO. 2014.60.4363]).
Only 3.7% of patients treated with IF-RT developed a second malignancy during follow-up, compared with 11.1% of those treated with combined modalities, 6.1% of those treated with EF-RT, and 7.4% of those treated with rituximab. The rate of relapse was markedly higher with rituximab than with the other treatments.
The main study findings are twofold. First, IF-RT was at least as effective as other treatments in controlling NLPHL, was less toxic acutely, and carried similar or reduced risks of late adverse effects such as relapse and second malignancies. It should be considered the first-line treatment of choice. Second, rituximab alone should not be used routinely in these patients because it yields poorer survival outcomes and a higher relapse rate. “However, it might represent an option for individual patients, such as young women with abdominal disease, to avoid gonadotoxic effects of radiotherapy,” the investigators said.
Involved-field radiotherapy alone should be the treatment of choice for stage 1A nodular lymphocyte-predominant Hodgkin lymphoma because it yields similar survival outcomes but fewer toxic effects than other therapies, investigators reported online Aug. 3 in Journal of Clinical Oncology.
Few studies have examined the long-term outcomes of patients who have this generally indolent malignancy, even though late treatment-associated effects are their major cause of death, said Dr. Dennis A. Eichenauer and his associates at University Hospital Cologne and University Hospital Münster, both in Germany.
To assess long-term outcomes in nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL), the investigators analyzed data for 256 patients who participated in seven prospective randomized clinical trials comparing various treatments during a 21-year period, who were followed for a median of 91 months. The study participants’ median age at diagnosis was 39 years (range, 16-75 years), and all achieved remission with therapy. They were categorized by treatment type: 108 received involved-field radiotherapy (IF-RT) alone; 49 received extended-field radiotherapy (EF-RT) alone; 72 received combined modality treatment involving various combinations of doxorubicin, bleomycin, vinblastine, dacarbazine, IF-RT, EF-RT, and/or rituximab; and 27 received rituximab alone.
At 8 years, progression-free survival was 91.9% with IF-RT, 84.3% with EF-RT, and 88.5% with combined modalities, which are nonsignificant differences. Overall survival was 99.0%, 95.7%, and 98.6%, respectively, which also are nonsignificant differences. Rituximab alone was markedly less effective than the other treatments, with a 4-year progression-free survival of only 81.0%. “Compared with patients who received IF-RT, patients treated with rituximab had a hazard ratio of 4.99 for relapse,” Dr. Eichenauer and his associates wrote (J Clin Oncol. 2015 Aug 3 [doi: 10.1200/JCO. 2014.60.4363]).
Only 3.7% of patients treated with IF-RT developed a second malignancy during follow-up, compared with 11.1% of those treated with combined modalities, 6.1% of those treated with EF-RT, and 7.4% of those treated with rituximab. The rate of relapse was markedly higher with rituximab than with the other treatments.
The main study findings are twofold. First, IF-RT was at least as effective as other treatments in controlling NLPHL, was less toxic acutely, and carried similar or reduced risks of late adverse effects such as relapse and second malignancies. It should be considered the first-line treatment of choice. Second, rituximab alone should not be used routinely in these patients because it yields poorer survival outcomes and a higher relapse rate. “However, it might represent an option for individual patients, such as young women with abdominal disease, to avoid gonadotoxic effects of radiotherapy,” the investigators said.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Involved-field radiotherapy alone yields similar survival but fewer toxic effects than do other treatments for stage 1A nodular lymphocyte–predominant Hodgkin lymphoma.
Major finding: Progression-free survival was 91.9% with IF-RT, 84.3% with EF-RT, and 88.5% with combined modalities, and overall survival was 99.0%, 95.7%, and 98.6%, respectively.
Data source: A retrospective analysis of outcomes of seven German prospective clinical trials involving 256 patients followed for a median of 91 months.
Disclosures: This study was supported by University Hospital Cologne and University Hospital Münster. Dr. Eichenauer reported having no relevant financial disclosures; his associates reported ties to Amgen, Takeda, Novartis, Gilead, and Takeda/Millennium.
Intraperitoneal chemo still underused for ovarian cancer
Intraperitoneal chemotherapy provides a clear survival benefit in stage III ovarian cancer in real-world practice as it did in clinical trials, yet it is still underused, according to a study published online in Journal of Clinical Oncology.
The National Cancer Institute issued an unusual Clinical Announcement in 2006 encouraging the use of combined intraperitoneal/intravenous (IP/IV) chemotherapy in this setting, after clinical trials showed it substantially improved overall survival, compared with IV chemotherapy alone.
But some clinicians may be reluctant to use the combined approach, believing that other regimens such as dose-dense paclitaxel yield comparable survival with fewer toxicities. Others cite barriers such as increased toxicity with intraperitoneal treatment, the lack of a standardized regimen, the inconvenience because inpatient administration is required, and patient preference for other approaches, said Dr. Alexi Wright of Dana-Farber/Brigham and Women’s Cancer Center, Boston, and her associates.
To assess the uptake of intraperitoneal therapy over time and its use in real-world practice, Dr. Wright and her associates analyzed data from two cohorts treated at six elite comprehensive cancer centers: a cohort of 823 women treated during a 9-year period that spanned before and after the 2006 Clinical Announcement, and a propensity-matched cohort of 498 women treated outside of clinical trials and after 2006. They found that the use of IP/IV increased from 0% to 33% between 2003 and 2006, increased again to nearly 50% between 2007 and 2008, but plateaued after that.
Three-year overall survival was 81% with IP/IV chemotherapy, significantly higher than the 71% survival for IV chemotherapy. The relative risk of death in the IP/IV group compared with the IV group was 0.68. Thus, the magnitude of benefit in this real-world population matches that reported in clinical trials, the investigators said (J Clin Oncol. 2015 Aug 3. doi:10.1200/JCO.2015.61.4776).
Yet only 41% of patients eligible for IP/IV therapy in this study received it. Use of the treatment varied dramatically across the six cancer centers, from 4% to 67% of patients, even though sociodemographic and disease characteristics were relatively uniform. And IP/IV regimens varied widely, with 43% of the women who received it given modified regimens to reduce its toxicity.
The findings show that cancer treatment may be influenced by local culture and by leaders’ endorsement of particular treatments or trial results. Women with ovarian cancer may receive substantially different treatment depending on where they seek care. “Additional interventions may be required to ensure that IP/IV chemotherapy decision-making is more uniform and patient-centered,” Dr. Wright and her associates said.
The National Cancer Institute supported the study. Dr. Wright reported having no financial conflicts. Her associates reported ties to McKesson, Genentech, Boehringer Ingelheim, AstraZeneca, AbbVie, Sanofi, Novartis, ImmunoGen, Endocyte, Gradalis, Amgen, Janssen, Oxigene, Roche, CSL Behring, Medtronic, and Eisai.
Intraperitoneal chemotherapy provides a clear survival benefit in stage III ovarian cancer in real-world practice as it did in clinical trials, yet it is still underused, according to a study published online in Journal of Clinical Oncology.
The National Cancer Institute issued an unusual Clinical Announcement in 2006 encouraging the use of combined intraperitoneal/intravenous (IP/IV) chemotherapy in this setting, after clinical trials showed it substantially improved overall survival, compared with IV chemotherapy alone.
But some clinicians may be reluctant to use the combined approach, believing that other regimens such as dose-dense paclitaxel yield comparable survival with fewer toxicities. Others cite barriers such as increased toxicity with intraperitoneal treatment, the lack of a standardized regimen, the inconvenience because inpatient administration is required, and patient preference for other approaches, said Dr. Alexi Wright of Dana-Farber/Brigham and Women’s Cancer Center, Boston, and her associates.
To assess the uptake of intraperitoneal therapy over time and its use in real-world practice, Dr. Wright and her associates analyzed data from two cohorts treated at six elite comprehensive cancer centers: a cohort of 823 women treated during a 9-year period that spanned before and after the 2006 Clinical Announcement, and a propensity-matched cohort of 498 women treated outside of clinical trials and after 2006. They found that the use of IP/IV increased from 0% to 33% between 2003 and 2006, increased again to nearly 50% between 2007 and 2008, but plateaued after that.
Three-year overall survival was 81% with IP/IV chemotherapy, significantly higher than the 71% survival for IV chemotherapy. The relative risk of death in the IP/IV group compared with the IV group was 0.68. Thus, the magnitude of benefit in this real-world population matches that reported in clinical trials, the investigators said (J Clin Oncol. 2015 Aug 3. doi:10.1200/JCO.2015.61.4776).
Yet only 41% of patients eligible for IP/IV therapy in this study received it. Use of the treatment varied dramatically across the six cancer centers, from 4% to 67% of patients, even though sociodemographic and disease characteristics were relatively uniform. And IP/IV regimens varied widely, with 43% of the women who received it given modified regimens to reduce its toxicity.
The findings show that cancer treatment may be influenced by local culture and by leaders’ endorsement of particular treatments or trial results. Women with ovarian cancer may receive substantially different treatment depending on where they seek care. “Additional interventions may be required to ensure that IP/IV chemotherapy decision-making is more uniform and patient-centered,” Dr. Wright and her associates said.
The National Cancer Institute supported the study. Dr. Wright reported having no financial conflicts. Her associates reported ties to McKesson, Genentech, Boehringer Ingelheim, AstraZeneca, AbbVie, Sanofi, Novartis, ImmunoGen, Endocyte, Gradalis, Amgen, Janssen, Oxigene, Roche, CSL Behring, Medtronic, and Eisai.
Intraperitoneal chemotherapy provides a clear survival benefit in stage III ovarian cancer in real-world practice as it did in clinical trials, yet it is still underused, according to a study published online in Journal of Clinical Oncology.
The National Cancer Institute issued an unusual Clinical Announcement in 2006 encouraging the use of combined intraperitoneal/intravenous (IP/IV) chemotherapy in this setting, after clinical trials showed it substantially improved overall survival, compared with IV chemotherapy alone.
But some clinicians may be reluctant to use the combined approach, believing that other regimens such as dose-dense paclitaxel yield comparable survival with fewer toxicities. Others cite barriers such as increased toxicity with intraperitoneal treatment, the lack of a standardized regimen, the inconvenience because inpatient administration is required, and patient preference for other approaches, said Dr. Alexi Wright of Dana-Farber/Brigham and Women’s Cancer Center, Boston, and her associates.
To assess the uptake of intraperitoneal therapy over time and its use in real-world practice, Dr. Wright and her associates analyzed data from two cohorts treated at six elite comprehensive cancer centers: a cohort of 823 women treated during a 9-year period that spanned before and after the 2006 Clinical Announcement, and a propensity-matched cohort of 498 women treated outside of clinical trials and after 2006. They found that the use of IP/IV increased from 0% to 33% between 2003 and 2006, increased again to nearly 50% between 2007 and 2008, but plateaued after that.
Three-year overall survival was 81% with IP/IV chemotherapy, significantly higher than the 71% survival for IV chemotherapy. The relative risk of death in the IP/IV group compared with the IV group was 0.68. Thus, the magnitude of benefit in this real-world population matches that reported in clinical trials, the investigators said (J Clin Oncol. 2015 Aug 3. doi:10.1200/JCO.2015.61.4776).
Yet only 41% of patients eligible for IP/IV therapy in this study received it. Use of the treatment varied dramatically across the six cancer centers, from 4% to 67% of patients, even though sociodemographic and disease characteristics were relatively uniform. And IP/IV regimens varied widely, with 43% of the women who received it given modified regimens to reduce its toxicity.
The findings show that cancer treatment may be influenced by local culture and by leaders’ endorsement of particular treatments or trial results. Women with ovarian cancer may receive substantially different treatment depending on where they seek care. “Additional interventions may be required to ensure that IP/IV chemotherapy decision-making is more uniform and patient-centered,” Dr. Wright and her associates said.
The National Cancer Institute supported the study. Dr. Wright reported having no financial conflicts. Her associates reported ties to McKesson, Genentech, Boehringer Ingelheim, AstraZeneca, AbbVie, Sanofi, Novartis, ImmunoGen, Endocyte, Gradalis, Amgen, Janssen, Oxigene, Roche, CSL Behring, Medtronic, and Eisai.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Intraperitoneal chemotherapy provides a clear survival benefit in stage III ovarian cancer but is still underused in real-world practice.
Major finding: The relative risk of death in the IP/IV group compared with the IV group was 0.68, yet only 41% of eligible patients received IP/IV.
Data source: Two prospective cohort studies assessing time trends in intraperitoneal chemotherapy among 823 women treated at six elite cancer centers.
Disclosures: The National Cancer Institute supported the study. Dr. Wright reported having no financial conflicts. Her associates reported ties to McKesson, Genentech, Boehringer Ingelheim, AstraZeneca, AbbVie, Sanofi, Novartis, ImmunoGen, Endocyte, Gradalis, Amgen, Janssen, Oxigene, Roche, CSL Behring, Medtronic, and Eisai.