User login
PMDD May Get More Validation in DSM-5
MEDELLIN, Columbia – After years of debate and controversy, premenstrual dysphoric disorder could be closer to getting recognized as a full category of mood disorder in the DSM-5, a leading researcher in women’s mental health predicts.
Dr. Meir Steiner, who served as an adviser to the DSM-5’s Mood Disorders Work Group dealing with PMDD, sees the possible repositioning of PMDD within the revised manual as something of a triumph.
The new criteria for PMDD, a severe variant of premenstrual syndrome estimated to affect up to 5% of premenopausal women (Am. J. Psychiatry 2012;AiA:1-11), differ somewhat from those described in the DSM-IV. The order of symptoms likely will be shuffled, with mood swings and irritability now at the top of the list, where "markedly depressed mood" had topped the DSM-IV’s, Dr. Steiner said at the International Congress of Medicine and Women’s Mental Health.
PMDD first appeared as "late luteal phase dysphoric disorder" in Appendix A of the DSM-III-R in 1987, over the objections of some women’s groups and clinicians, who viewed its inclusion as pathologizing the menstrual cycle. The name of the disorder was changed to PMDD for the DSM-IV. Debate over PMDD intensified in 2000, when the Food and Drug Administration approved the rebranding of the selective serotonin reuptake inhibitor (SSRI) fluoxetine under the marketing name Sarafem to treat PMDD. Critics saw in the new indication an example of "disease mongering" benefiting pharmaceutical manufacturers (PLoS Med. 2006;3:e198).
Proponents of PMDD such as Dr. Steiner counter that the menstrual fluctuations in physical and emotional symptoms most women experience would not be considered pathological under the DSM-IV or DSM-5 diagnostic criteria, which require that 5 or more of 11 listed symptoms occur in most menstrual cycles during the luteal phase, begin to improve within a few days after the onset of menses, and are minimal or absent in the week post menses.
Moreover, they say, the criteria can prevent affected women from being incorrectly diagnosed with a depressive or personality disorder.
In the past decade, the debate on PMDD has calmed considerably, thanks in part to evidence from randomized controlled trials using the DSM-IV criteria for PMDD. The European Medicines Agency, which long refused to validate the indication, changed its position in 2010, opening up the possibility for PMDD treatments to be tested and marketed in the European Union.
Research has shown that PMDD can be treated with oral contraceptives containing drospirenone and ovarian suppression with GnRH agonists. It can also be treated with a wide range of SSRIs, which a meta-analysis of 29 randomized controlled trials involving a total of 2,964 patients showed to be effective in alleviating the mood and physical symptoms associated with PMDD (Obstet. Gynecol. 2008;111:1175-82). Unlike in depression, "you can actually accomplish improvement with a few days of treatment" with SSRIs, allowing for intermittent treatment, said Dr. Steiner. He served on an advisory body for the DSM-IV, where PMDD is not listed as a diagnostic category but in an appendix of criteria warranting further study.
In a separate talk at the congress, Dr. Steiner described other women-specific changes that he hopes to see in the DSM-5. He said the DSM-5 should include a category for childbirth-related posttraumatic stress disorder, in which the act of birthing is the triggering trauma.
"There is also no specific category for perinatal bereavement. This should be corrected," said Dr. Steiner, professor emeritus in the departments of psychiatry and behavioural neurosciences and obstetrics and gynecology at McMaster University in Hamilton, Ont.
Additionally, Dr. Steiner said that he would like to see changes to the diagnostic criteria for postpartum depression, and that the DSM-5 should extend the diagnostic window for its onset to 6 months post partum. And finally, he said, the DSM-5 also should identify depression onset during perimenopause as something distinct from depression in other periods of life. "We would like to identify perimenopause as a new window of vulnerability and risk," he said.
Despite his optimism about PMDD and the DSM-5, Dr. Steiner remained critical of the diagnostic manual as a whole, describing the DSM-IV as a "failure" for women. "Right now we’re looking at how we can prevent another failure," he said.
Dr. Steiner disclosed recent financial support from several pharmaceutical companies, including AstraZeneca, Azevan, Bayer Canada, and Servier.
MEDELLIN, Columbia – After years of debate and controversy, premenstrual dysphoric disorder could be closer to getting recognized as a full category of mood disorder in the DSM-5, a leading researcher in women’s mental health predicts.
Dr. Meir Steiner, who served as an adviser to the DSM-5’s Mood Disorders Work Group dealing with PMDD, sees the possible repositioning of PMDD within the revised manual as something of a triumph.
The new criteria for PMDD, a severe variant of premenstrual syndrome estimated to affect up to 5% of premenopausal women (Am. J. Psychiatry 2012;AiA:1-11), differ somewhat from those described in the DSM-IV. The order of symptoms likely will be shuffled, with mood swings and irritability now at the top of the list, where "markedly depressed mood" had topped the DSM-IV’s, Dr. Steiner said at the International Congress of Medicine and Women’s Mental Health.
PMDD first appeared as "late luteal phase dysphoric disorder" in Appendix A of the DSM-III-R in 1987, over the objections of some women’s groups and clinicians, who viewed its inclusion as pathologizing the menstrual cycle. The name of the disorder was changed to PMDD for the DSM-IV. Debate over PMDD intensified in 2000, when the Food and Drug Administration approved the rebranding of the selective serotonin reuptake inhibitor (SSRI) fluoxetine under the marketing name Sarafem to treat PMDD. Critics saw in the new indication an example of "disease mongering" benefiting pharmaceutical manufacturers (PLoS Med. 2006;3:e198).
Proponents of PMDD such as Dr. Steiner counter that the menstrual fluctuations in physical and emotional symptoms most women experience would not be considered pathological under the DSM-IV or DSM-5 diagnostic criteria, which require that 5 or more of 11 listed symptoms occur in most menstrual cycles during the luteal phase, begin to improve within a few days after the onset of menses, and are minimal or absent in the week post menses.
Moreover, they say, the criteria can prevent affected women from being incorrectly diagnosed with a depressive or personality disorder.
In the past decade, the debate on PMDD has calmed considerably, thanks in part to evidence from randomized controlled trials using the DSM-IV criteria for PMDD. The European Medicines Agency, which long refused to validate the indication, changed its position in 2010, opening up the possibility for PMDD treatments to be tested and marketed in the European Union.
Research has shown that PMDD can be treated with oral contraceptives containing drospirenone and ovarian suppression with GnRH agonists. It can also be treated with a wide range of SSRIs, which a meta-analysis of 29 randomized controlled trials involving a total of 2,964 patients showed to be effective in alleviating the mood and physical symptoms associated with PMDD (Obstet. Gynecol. 2008;111:1175-82). Unlike in depression, "you can actually accomplish improvement with a few days of treatment" with SSRIs, allowing for intermittent treatment, said Dr. Steiner. He served on an advisory body for the DSM-IV, where PMDD is not listed as a diagnostic category but in an appendix of criteria warranting further study.
In a separate talk at the congress, Dr. Steiner described other women-specific changes that he hopes to see in the DSM-5. He said the DSM-5 should include a category for childbirth-related posttraumatic stress disorder, in which the act of birthing is the triggering trauma.
"There is also no specific category for perinatal bereavement. This should be corrected," said Dr. Steiner, professor emeritus in the departments of psychiatry and behavioural neurosciences and obstetrics and gynecology at McMaster University in Hamilton, Ont.
Additionally, Dr. Steiner said that he would like to see changes to the diagnostic criteria for postpartum depression, and that the DSM-5 should extend the diagnostic window for its onset to 6 months post partum. And finally, he said, the DSM-5 also should identify depression onset during perimenopause as something distinct from depression in other periods of life. "We would like to identify perimenopause as a new window of vulnerability and risk," he said.
Despite his optimism about PMDD and the DSM-5, Dr. Steiner remained critical of the diagnostic manual as a whole, describing the DSM-IV as a "failure" for women. "Right now we’re looking at how we can prevent another failure," he said.
Dr. Steiner disclosed recent financial support from several pharmaceutical companies, including AstraZeneca, Azevan, Bayer Canada, and Servier.
MEDELLIN, Columbia – After years of debate and controversy, premenstrual dysphoric disorder could be closer to getting recognized as a full category of mood disorder in the DSM-5, a leading researcher in women’s mental health predicts.
Dr. Meir Steiner, who served as an adviser to the DSM-5’s Mood Disorders Work Group dealing with PMDD, sees the possible repositioning of PMDD within the revised manual as something of a triumph.
The new criteria for PMDD, a severe variant of premenstrual syndrome estimated to affect up to 5% of premenopausal women (Am. J. Psychiatry 2012;AiA:1-11), differ somewhat from those described in the DSM-IV. The order of symptoms likely will be shuffled, with mood swings and irritability now at the top of the list, where "markedly depressed mood" had topped the DSM-IV’s, Dr. Steiner said at the International Congress of Medicine and Women’s Mental Health.
PMDD first appeared as "late luteal phase dysphoric disorder" in Appendix A of the DSM-III-R in 1987, over the objections of some women’s groups and clinicians, who viewed its inclusion as pathologizing the menstrual cycle. The name of the disorder was changed to PMDD for the DSM-IV. Debate over PMDD intensified in 2000, when the Food and Drug Administration approved the rebranding of the selective serotonin reuptake inhibitor (SSRI) fluoxetine under the marketing name Sarafem to treat PMDD. Critics saw in the new indication an example of "disease mongering" benefiting pharmaceutical manufacturers (PLoS Med. 2006;3:e198).
Proponents of PMDD such as Dr. Steiner counter that the menstrual fluctuations in physical and emotional symptoms most women experience would not be considered pathological under the DSM-IV or DSM-5 diagnostic criteria, which require that 5 or more of 11 listed symptoms occur in most menstrual cycles during the luteal phase, begin to improve within a few days after the onset of menses, and are minimal or absent in the week post menses.
Moreover, they say, the criteria can prevent affected women from being incorrectly diagnosed with a depressive or personality disorder.
In the past decade, the debate on PMDD has calmed considerably, thanks in part to evidence from randomized controlled trials using the DSM-IV criteria for PMDD. The European Medicines Agency, which long refused to validate the indication, changed its position in 2010, opening up the possibility for PMDD treatments to be tested and marketed in the European Union.
Research has shown that PMDD can be treated with oral contraceptives containing drospirenone and ovarian suppression with GnRH agonists. It can also be treated with a wide range of SSRIs, which a meta-analysis of 29 randomized controlled trials involving a total of 2,964 patients showed to be effective in alleviating the mood and physical symptoms associated with PMDD (Obstet. Gynecol. 2008;111:1175-82). Unlike in depression, "you can actually accomplish improvement with a few days of treatment" with SSRIs, allowing for intermittent treatment, said Dr. Steiner. He served on an advisory body for the DSM-IV, where PMDD is not listed as a diagnostic category but in an appendix of criteria warranting further study.
In a separate talk at the congress, Dr. Steiner described other women-specific changes that he hopes to see in the DSM-5. He said the DSM-5 should include a category for childbirth-related posttraumatic stress disorder, in which the act of birthing is the triggering trauma.
"There is also no specific category for perinatal bereavement. This should be corrected," said Dr. Steiner, professor emeritus in the departments of psychiatry and behavioural neurosciences and obstetrics and gynecology at McMaster University in Hamilton, Ont.
Additionally, Dr. Steiner said that he would like to see changes to the diagnostic criteria for postpartum depression, and that the DSM-5 should extend the diagnostic window for its onset to 6 months post partum. And finally, he said, the DSM-5 also should identify depression onset during perimenopause as something distinct from depression in other periods of life. "We would like to identify perimenopause as a new window of vulnerability and risk," he said.
Despite his optimism about PMDD and the DSM-5, Dr. Steiner remained critical of the diagnostic manual as a whole, describing the DSM-IV as a "failure" for women. "Right now we’re looking at how we can prevent another failure," he said.
Dr. Steiner disclosed recent financial support from several pharmaceutical companies, including AstraZeneca, Azevan, Bayer Canada, and Servier.
EXPERT ANALYSIS FROM THE INTERNATIONAL CONGRESS OF MEDICINE AND WOMEN'S MENTAL HEALTH
AACE Endorses Lower LDL Targets, ApoB Testing
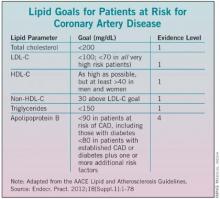
New guidelines on dyslipidemia and the prevention of atherogenesis give official sanction to something endocrinologists have been doing for years: lowering LDL targets to below 70 mg/dL for all very high-risk patients.
They also favor validating the effectiveness of LDL-lowering treatments by looking to apolipoprotein B, a measurement not yet widely used in clinical practice.
Fibrates, niacin, HDL level targets, the use of inflammatory biomarkers, and the frequency of lipid testing are other issues dealt with in the guidelines (Endocr. Pract. Vol. 18 (Suppl 1), March/April 2012, 1-78), which were published this month by the American Association of Clinical Endocrinologists.
AACE’s LDL target represents a departure from the National Cholesterol Education Program’s 2004 guidelines (ATP III), which advise physicians to aim for an LDL level of less than 100 mg/dL for patients with established coronary artery disease or a 10-year risk for a cardiac event of 20% or higher based on Framingham criteria, and to consider an "optional" target of 70 mg/dL for the same patient group. (See chart.) American Heart Association guidelines, similarly, call 70 mg/dL a "reasonable" goal for very high-risk patients.
Dr. Paul S. Jellinger, lead author of the guidelines, said that aiming for an LDL below 70 mg/dL for all patients with CAD, or those with diabetes plus at least one additional risk factor, reflects both a mounting scientific consensus and current practice. For patients at high risk – including people with diabetes and those with two or more risk factors and a 10-year Framingham risk of coronary heart disease – the AACE recommends an LDL target of less than 100 mg/dl.
Though the Food and Drug Administration recently changed statin labeling to reflect a slight hyperglycemia risk associated with their use, the new AACE guideline reinforces that patients with diabetes should be treated "aggressively."
The guideline also recommends that clinicians check the apolipoprotein B levels of patients on LDL-lowering treatments. ApoB is a biomarker that indicates LDL particle number and, indirectly, particle size. "Measuring apoB is the easiest, cheapest, and most consistent way to tell us that we’ve reduced the total number of LDL particles. It is more difficult to lower apoB than to lower LDL," said Dr. Jellinger of the Center for Diabetes and Endocrine Care in Hollywood, Fla., and professor of clinical medicine at the University of Miami.
While non-HDL is also a key measurement, and should be included in all reports, Dr. Jellinger said, "we look at non-HDL as a very good way to start and apoB as a very good final measurement to ascertain that you have reached your pharmacotherapy goal."
The AACE guidelines advise physicians to look at apoB even when LDL levels appear to be well controlled and at goal and consider apoB as a secondary therapeutic target. Recommended apo B levels for patients at risk of CAD, including those with diabetes, are less than 90 mg/dL, and less than 80 mg/dL for patients with established CAD or diabetes and one or more additional risk factors. The guidelines note the evidence here is not as strong as for the other recommendations.
Measurements of homocysteine, uric acid, and plasminogen activator inhibitor-1 are not routinely recommended by the AACE for use in stratifying risk due to the uncertain evidence of benefit achieved by lowering these factors.
But the AACE encourages looking at the inflammatory markers highly sensitive C-reactive protein (hsCRP) and in some patients lipoprotein-associated phospholipase A2 (Lp-PLA2), when risk is uncertain and additional information is needed to decide how aggressively to treat.
The guidelines note that while hsCRP can be useful in stratifying patients with intermediate risk, this marker can be elevated by systemic inflammation and obesity. Lp-PLA2, an enzyme found in arterial plaque, is more specific for vascular sources of inflammation. "With both hsCRP and Lp-PLA2 elevated, it may be appropriate to be more aggressive with treatment in a patient otherwise considered at intermediate risk," Dr. Jellinger said.
Also helpful in cases requiring precise risk stratification are two noninvasive measures of atherosclerosis: carotid intima media thickness and coronary artery calcification (CAC). But since there is no definite evidence that CAC, while strongly correlated with coronary atherosclerosis, independently predicts coronary events, the guidance warns that these are not helpful when performed routinely as a screening measure, which Dr. Jellinger said they often are. Rather, "they should only be used in certain situations as adjuncts to standard CVD risk factors to refine the patients’ risk when considering more aggressive therapy," he said.
The new guidelines recommend raising HDL-C levels as much as possible for patients at risk of CAD, but minimally to greater than 40 mg/dL in both men and women.
While statins remain the cornerstone of therapy, determining the roles fibrates and niacin should play in lowering triglycerides and raising HDL, and in which patients, is more complicated.
Several scientific studies cited in the AACE guidelines support a clinical benefit for the use of fibrates in patients whose triglycerides are greater than 200 and whose HDL is less than 40 mg/dL or 35 mg/dL for primary and secondary prevention of cardiovascular events. There remains uncertain clinical benefit in patients treated with fibrates for lesser triglyceride and HDL abnormalities.
Niacin is also recommended for increasing HDL and reducing LDL-C and triglycerides in certain patients despite results from AIM-HIGH, a randomized controlled trial that was terminated early after it became clear that patients whose LDL levels were very well controlled with simvastatin were seen not to benefit from the addition of niacin.
Dr. Jellinger said that niacin may benefit patients with less well controlled LDLand that the AACE is awaiting the results of another, larger niacin trial (HPS2-THRIVE). "You probably don’t have to start niacin if your LDL is very low. When your LDL is higher, we don’t yet know. It may be suitable," he said.
The guidelines also give more latitude as to when to order lipid testing. More frequent lipid testing may be indicated in the following circumstances: deterioration of diabetes control, the use of a new drug known to affect lipid levels, atherothrombotic disease progression, considerable weight gain, development of a new CAD risk factor and convincing new clinical trial evidence suggesting stricter lipid goals. The AACE’s position "ought to give comfort to physicians who want to order more frequent lipid profiles under certain circumstances," Dr. Jellinger said.
Dr. Jellinger disclosed receiving honoraria from Amylin Pharmaceuticals, Eli Lilly, Merck, and Novo Nordisk, and having served on advisory boards for Novo Nordisk, Merck, Boehringer Ingelheim, and Amylin. Six of the seven task force members reported disclosures of fees, honoraria, or grant support from these and/or other pharmaceutical and/or device manufacturers.
New guidelines on dyslipidemia and the prevention of atherogenesis give official sanction to something endocrinologists have been doing for years: lowering LDL targets to below 70 mg/dL for all very high-risk patients.
They also favor validating the effectiveness of LDL-lowering treatments by looking to apolipoprotein B, a measurement not yet widely used in clinical practice.
Fibrates, niacin, HDL level targets, the use of inflammatory biomarkers, and the frequency of lipid testing are other issues dealt with in the guidelines (Endocr. Pract. Vol. 18 (Suppl 1), March/April 2012, 1-78), which were published this month by the American Association of Clinical Endocrinologists.
AACE’s LDL target represents a departure from the National Cholesterol Education Program’s 2004 guidelines (ATP III), which advise physicians to aim for an LDL level of less than 100 mg/dL for patients with established coronary artery disease or a 10-year risk for a cardiac event of 20% or higher based on Framingham criteria, and to consider an "optional" target of 70 mg/dL for the same patient group. (See chart.) American Heart Association guidelines, similarly, call 70 mg/dL a "reasonable" goal for very high-risk patients.
Dr. Paul S. Jellinger, lead author of the guidelines, said that aiming for an LDL below 70 mg/dL for all patients with CAD, or those with diabetes plus at least one additional risk factor, reflects both a mounting scientific consensus and current practice. For patients at high risk – including people with diabetes and those with two or more risk factors and a 10-year Framingham risk of coronary heart disease – the AACE recommends an LDL target of less than 100 mg/dl.
Though the Food and Drug Administration recently changed statin labeling to reflect a slight hyperglycemia risk associated with their use, the new AACE guideline reinforces that patients with diabetes should be treated "aggressively."
The guideline also recommends that clinicians check the apolipoprotein B levels of patients on LDL-lowering treatments. ApoB is a biomarker that indicates LDL particle number and, indirectly, particle size. "Measuring apoB is the easiest, cheapest, and most consistent way to tell us that we’ve reduced the total number of LDL particles. It is more difficult to lower apoB than to lower LDL," said Dr. Jellinger of the Center for Diabetes and Endocrine Care in Hollywood, Fla., and professor of clinical medicine at the University of Miami.
While non-HDL is also a key measurement, and should be included in all reports, Dr. Jellinger said, "we look at non-HDL as a very good way to start and apoB as a very good final measurement to ascertain that you have reached your pharmacotherapy goal."
The AACE guidelines advise physicians to look at apoB even when LDL levels appear to be well controlled and at goal and consider apoB as a secondary therapeutic target. Recommended apo B levels for patients at risk of CAD, including those with diabetes, are less than 90 mg/dL, and less than 80 mg/dL for patients with established CAD or diabetes and one or more additional risk factors. The guidelines note the evidence here is not as strong as for the other recommendations.
Measurements of homocysteine, uric acid, and plasminogen activator inhibitor-1 are not routinely recommended by the AACE for use in stratifying risk due to the uncertain evidence of benefit achieved by lowering these factors.
But the AACE encourages looking at the inflammatory markers highly sensitive C-reactive protein (hsCRP) and in some patients lipoprotein-associated phospholipase A2 (Lp-PLA2), when risk is uncertain and additional information is needed to decide how aggressively to treat.
The guidelines note that while hsCRP can be useful in stratifying patients with intermediate risk, this marker can be elevated by systemic inflammation and obesity. Lp-PLA2, an enzyme found in arterial plaque, is more specific for vascular sources of inflammation. "With both hsCRP and Lp-PLA2 elevated, it may be appropriate to be more aggressive with treatment in a patient otherwise considered at intermediate risk," Dr. Jellinger said.
Also helpful in cases requiring precise risk stratification are two noninvasive measures of atherosclerosis: carotid intima media thickness and coronary artery calcification (CAC). But since there is no definite evidence that CAC, while strongly correlated with coronary atherosclerosis, independently predicts coronary events, the guidance warns that these are not helpful when performed routinely as a screening measure, which Dr. Jellinger said they often are. Rather, "they should only be used in certain situations as adjuncts to standard CVD risk factors to refine the patients’ risk when considering more aggressive therapy," he said.
The new guidelines recommend raising HDL-C levels as much as possible for patients at risk of CAD, but minimally to greater than 40 mg/dL in both men and women.
While statins remain the cornerstone of therapy, determining the roles fibrates and niacin should play in lowering triglycerides and raising HDL, and in which patients, is more complicated.
Several scientific studies cited in the AACE guidelines support a clinical benefit for the use of fibrates in patients whose triglycerides are greater than 200 and whose HDL is less than 40 mg/dL or 35 mg/dL for primary and secondary prevention of cardiovascular events. There remains uncertain clinical benefit in patients treated with fibrates for lesser triglyceride and HDL abnormalities.
Niacin is also recommended for increasing HDL and reducing LDL-C and triglycerides in certain patients despite results from AIM-HIGH, a randomized controlled trial that was terminated early after it became clear that patients whose LDL levels were very well controlled with simvastatin were seen not to benefit from the addition of niacin.
Dr. Jellinger said that niacin may benefit patients with less well controlled LDLand that the AACE is awaiting the results of another, larger niacin trial (HPS2-THRIVE). "You probably don’t have to start niacin if your LDL is very low. When your LDL is higher, we don’t yet know. It may be suitable," he said.
The guidelines also give more latitude as to when to order lipid testing. More frequent lipid testing may be indicated in the following circumstances: deterioration of diabetes control, the use of a new drug known to affect lipid levels, atherothrombotic disease progression, considerable weight gain, development of a new CAD risk factor and convincing new clinical trial evidence suggesting stricter lipid goals. The AACE’s position "ought to give comfort to physicians who want to order more frequent lipid profiles under certain circumstances," Dr. Jellinger said.
Dr. Jellinger disclosed receiving honoraria from Amylin Pharmaceuticals, Eli Lilly, Merck, and Novo Nordisk, and having served on advisory boards for Novo Nordisk, Merck, Boehringer Ingelheim, and Amylin. Six of the seven task force members reported disclosures of fees, honoraria, or grant support from these and/or other pharmaceutical and/or device manufacturers.
New guidelines on dyslipidemia and the prevention of atherogenesis give official sanction to something endocrinologists have been doing for years: lowering LDL targets to below 70 mg/dL for all very high-risk patients.
They also favor validating the effectiveness of LDL-lowering treatments by looking to apolipoprotein B, a measurement not yet widely used in clinical practice.
Fibrates, niacin, HDL level targets, the use of inflammatory biomarkers, and the frequency of lipid testing are other issues dealt with in the guidelines (Endocr. Pract. Vol. 18 (Suppl 1), March/April 2012, 1-78), which were published this month by the American Association of Clinical Endocrinologists.
AACE’s LDL target represents a departure from the National Cholesterol Education Program’s 2004 guidelines (ATP III), which advise physicians to aim for an LDL level of less than 100 mg/dL for patients with established coronary artery disease or a 10-year risk for a cardiac event of 20% or higher based on Framingham criteria, and to consider an "optional" target of 70 mg/dL for the same patient group. (See chart.) American Heart Association guidelines, similarly, call 70 mg/dL a "reasonable" goal for very high-risk patients.
Dr. Paul S. Jellinger, lead author of the guidelines, said that aiming for an LDL below 70 mg/dL for all patients with CAD, or those with diabetes plus at least one additional risk factor, reflects both a mounting scientific consensus and current practice. For patients at high risk – including people with diabetes and those with two or more risk factors and a 10-year Framingham risk of coronary heart disease – the AACE recommends an LDL target of less than 100 mg/dl.
Though the Food and Drug Administration recently changed statin labeling to reflect a slight hyperglycemia risk associated with their use, the new AACE guideline reinforces that patients with diabetes should be treated "aggressively."
The guideline also recommends that clinicians check the apolipoprotein B levels of patients on LDL-lowering treatments. ApoB is a biomarker that indicates LDL particle number and, indirectly, particle size. "Measuring apoB is the easiest, cheapest, and most consistent way to tell us that we’ve reduced the total number of LDL particles. It is more difficult to lower apoB than to lower LDL," said Dr. Jellinger of the Center for Diabetes and Endocrine Care in Hollywood, Fla., and professor of clinical medicine at the University of Miami.
While non-HDL is also a key measurement, and should be included in all reports, Dr. Jellinger said, "we look at non-HDL as a very good way to start and apoB as a very good final measurement to ascertain that you have reached your pharmacotherapy goal."
The AACE guidelines advise physicians to look at apoB even when LDL levels appear to be well controlled and at goal and consider apoB as a secondary therapeutic target. Recommended apo B levels for patients at risk of CAD, including those with diabetes, are less than 90 mg/dL, and less than 80 mg/dL for patients with established CAD or diabetes and one or more additional risk factors. The guidelines note the evidence here is not as strong as for the other recommendations.
Measurements of homocysteine, uric acid, and plasminogen activator inhibitor-1 are not routinely recommended by the AACE for use in stratifying risk due to the uncertain evidence of benefit achieved by lowering these factors.
But the AACE encourages looking at the inflammatory markers highly sensitive C-reactive protein (hsCRP) and in some patients lipoprotein-associated phospholipase A2 (Lp-PLA2), when risk is uncertain and additional information is needed to decide how aggressively to treat.
The guidelines note that while hsCRP can be useful in stratifying patients with intermediate risk, this marker can be elevated by systemic inflammation and obesity. Lp-PLA2, an enzyme found in arterial plaque, is more specific for vascular sources of inflammation. "With both hsCRP and Lp-PLA2 elevated, it may be appropriate to be more aggressive with treatment in a patient otherwise considered at intermediate risk," Dr. Jellinger said.
Also helpful in cases requiring precise risk stratification are two noninvasive measures of atherosclerosis: carotid intima media thickness and coronary artery calcification (CAC). But since there is no definite evidence that CAC, while strongly correlated with coronary atherosclerosis, independently predicts coronary events, the guidance warns that these are not helpful when performed routinely as a screening measure, which Dr. Jellinger said they often are. Rather, "they should only be used in certain situations as adjuncts to standard CVD risk factors to refine the patients’ risk when considering more aggressive therapy," he said.
The new guidelines recommend raising HDL-C levels as much as possible for patients at risk of CAD, but minimally to greater than 40 mg/dL in both men and women.
While statins remain the cornerstone of therapy, determining the roles fibrates and niacin should play in lowering triglycerides and raising HDL, and in which patients, is more complicated.
Several scientific studies cited in the AACE guidelines support a clinical benefit for the use of fibrates in patients whose triglycerides are greater than 200 and whose HDL is less than 40 mg/dL or 35 mg/dL for primary and secondary prevention of cardiovascular events. There remains uncertain clinical benefit in patients treated with fibrates for lesser triglyceride and HDL abnormalities.
Niacin is also recommended for increasing HDL and reducing LDL-C and triglycerides in certain patients despite results from AIM-HIGH, a randomized controlled trial that was terminated early after it became clear that patients whose LDL levels were very well controlled with simvastatin were seen not to benefit from the addition of niacin.
Dr. Jellinger said that niacin may benefit patients with less well controlled LDLand that the AACE is awaiting the results of another, larger niacin trial (HPS2-THRIVE). "You probably don’t have to start niacin if your LDL is very low. When your LDL is higher, we don’t yet know. It may be suitable," he said.
The guidelines also give more latitude as to when to order lipid testing. More frequent lipid testing may be indicated in the following circumstances: deterioration of diabetes control, the use of a new drug known to affect lipid levels, atherothrombotic disease progression, considerable weight gain, development of a new CAD risk factor and convincing new clinical trial evidence suggesting stricter lipid goals. The AACE’s position "ought to give comfort to physicians who want to order more frequent lipid profiles under certain circumstances," Dr. Jellinger said.
Dr. Jellinger disclosed receiving honoraria from Amylin Pharmaceuticals, Eli Lilly, Merck, and Novo Nordisk, and having served on advisory boards for Novo Nordisk, Merck, Boehringer Ingelheim, and Amylin. Six of the seven task force members reported disclosures of fees, honoraria, or grant support from these and/or other pharmaceutical and/or device manufacturers.
FROM ENDOCRINE PRACTICE
Telestroke Triage Comes of Age
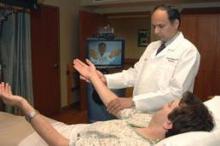
When Dr. Steven R. Levine and Dr. Mark Gorman first proposed that emergency physicians in small rural hospitals link up via bedside video cameras to stroke specialists in large urban hospitals to triage stroke patients, they named the concept "telestroke."
"I never thought it would catch on," Dr. Levine said in a recent interview.
It was 1999, and while thrombolysis with tissue plasminogen activator (TPA) had by then been approved for 3 years, only about 1% of acute ischemic stroke patients in the United States were receiving it, when as many as half could potentially have been eligible.
One important reason for the underuse of TPA was that smaller and more rural hospitals did not have neurologists available to respond to stroke calls. Patients would have to be transported to larger centers to receive intravenous TPA, and by the time they arrived, many would have missed the time window – which published guidelines have now determined to be up to 4.5 hours after a stroke – when TPA can be safely administered.
Dr. Levine, currently professor of neurology and emergency medicine at SUNY Downstate Medical Center in Brooklyn, N.Y., and a leading researcher and proponent of telestroke, envisioned a spoke-and-hub model of care in which smaller hospitals would be linked by video to larger ones with neurologists available, in active shifts, for two-way consults (Stroke 1999;30:464-9).
TPA could be initiated in the emergency departments of the spokes, and higher-risk patients would be transferred to hub hospitals with dedicated stroke centers once a drip was started.
Early adopters included Massachusetts General Hospital, which began building on the spoke-and-hub model and proving the concept in study after study. "We were the first to demonstrate that the NIH stroke scale could be done with equivalent reliability over telemedicine, and to show that a neurologist could interpret information on a laptop computer as effectively as a radiologist could on expensive imaging systems," said Dr. Lee H. Schwamm, professor of neurology at Harvard University and director of the Partners TeleStroke center, a 30-hospital network run out of Massachusetts General and Brigham and Women’s Hospital, both in Boston.
In 2009, on the strength of evidence showing that telestroke programs increased TPA administration rates, Dr. Schwamm and colleagues, including Dr. Levine, drafted a policy statement for the American Heart Association (Stroke 2009;40:2635-60) advocating the creation of telestroke networks wherever resources were available. More recent evidence has shown telestroke to be cost-effective compared with conventional care (Neurology 2011;77:1590-8; Stroke 2012;43:A3077).
Currently there are at least 27 networks up and running in the United States and Canada, along with 14 in Europe, following spoke-and-hub models. Some comprise a handful of hospitals; others, dozens. Many have existed for only a few years.
North American telestroke programs can now boast TPA rates between 10% and 20% of patients admitted for stroke, even in very rural networks, when the U.S. national average is estimated to be about 5%. In Europe, established programs are reporting TPA rates between 12% and 17%, according to a recent meta-analysis (Curr. Opin. Neurol. 2012;25:5-10).
Dr. Charles H. Tegeler, professor of neurology and head of the telestroke program at Wake Forest Baptist Medical Center in Winston-Salem, N.C., said in an interview that his program, which started in 2009 and now incorporates nine network hospitals, has seen TPA administered in 27% of patients who receive a video stroke consultation. And many patients are receiving TPA in hospitals that, before joining the network, did not administer it.
About 45% of patients receiving a telestroke consultation through one of the network hospitals are transferred to Wake Forest Baptist, or another dedicated stroke center of the patient’s choice, for additional evaluation and management. Depending on local resources and expertise, those treated with TPA may be transferred under a treatment model known as "drip and ship." The rest remain at the network hospital for ongoing care, or "drip and keep."
Rayetta Johnson, R.N., stroke program manager for Wake Forest Baptist, said in an interview that an important factor in its success has been achieving close cooperation, through training seminars and other forms of outreach, with hospital emergency department staff at all levels. The program also reached out extensively to local emergency medical services accustomed to rerouting stroke patients to larger hospitals. "Having EMS on board is extremely important," Ms. Johnson said.
Barriers to Adoption
If telestroke programs are cost effective and improve TPA rates dramatically, why aren’t they more widespread?
Part of the problem, experts say, lies with neurologists’ hesitance to join telestroke networks because reimbursement for telestroke is less straightforward than for a traditional stroke call, and many consider it to be inadequate under current Medicare and Medicaid guidelines.
Dr. Ramesh Madhavan, director of telemedicine for Wayne State University, Detroit, and the Michigan Stroke Network, one of the largest telestroke programs in the country with 36 affiliated hospitals, said in an interview that his program’s eight neurologists – including himself – take stroke calls over 12-hour shifts.
"We take shifts because there is no direct reimbursement, and we have to do other things during the course of that time. We have to multitask," Dr. Madhavan said, adding that this can make some neurologists feel burned out.
Dr. Tegeler said that, by contrast, the five vascular neurologists in Wake Forest Baptist’s smaller program have incorporated the telestroke coverage as part of their regular stroke attending call duties. Now, neurologists at some of the network hospitals "may not have to disrupt their office schedule for an hour or more to go over to the hospital to see an acute stroke patient," he said.
The presence of telestroke coverage also was used by one network hospital to help recruit a neurologist to a community where there had not been one, since the local neurologist would "not have to take stroke calls 24-7," Dr. Tegeler said.
In addition to the reimbursement issue, Dr. Levine said that telestroke has been hampered by "fears that the technology won’t work or may break down, or of lawsuits, and also doctors’ fear of something more technologically advanced than some are used to doing." Unlike their counterparts in emergency departments, Dr. Levine said, "neurologists aren’t techies, as a rule." However, telestroke experts are increasingly exploring cheaper and more portable options for videoconferencing, which could prove less imposing in terms of both startup costs to networks and in ease of use.
Most telestroke programs currently employ videoconferencing technology using equipment mounted on a mobile cart or purpose-built robots that can be driven to a patient’s bedside.
The robots, Dr. Schwamm said, "are a luxury. You can put a laptop with a specialized camera on top of a cart and roll it to a patient and get everything you need. Technology should not be where the costs are."
One team of researchers recently demonstrated that iPhones could be used in telestroke networks for patient assessment (J. Stroke Cerebrovasc. Dis. 2011 Oct. 24 [doi:10.1016/j.jstrokecerebrovasdis.2011.09.013]).
Dr. Madhavan said that he has been working with several technology companies on applications that would allow more telestroke consults to be carried out on portable devices using 3G or 4G networks. A migration to mobile platforms might help improve door-to-needle times in addition to reducing costs, he said.
Research Priorities
When Dr. Levine and Dr. Gorman first proposed telestroke networks in 1999, they envisioned them not solely as a way to increase TPA uptake but also as a way to facilitate patient entry into clinical trials of new stroke treatments.
This has yet to happen directly, Dr. Levine noted, but it may soon. "Community hospitals are not used to dealing with experimental medicines, or the logistics of randomization, and working with trial coordinators, IRBs, and rigorous and extensive data collection. But now that the systems are being built and there’s some infrastructure over the last 10 years, hospitals will hopefully start to see what clinical trials can bring them in terms of recruitment, state-of-the-art care while testing the most novel and promising treatments, and financially," he said.
Dr. Schwamm, whose program is among the few actively seeking to design clinical trials using telestroke, said that enrolling patients through telemedicine – and obtaining consent through video – is a challenge that, if met, will help broaden trial populations to reach more people living in rural and resource-poor areas. "The big studies tend to be done at the teaching hospitals, in urban areas. Telestroke can help allow people in the community to be enrolled," he said, leading to better representation of the population at large.
More telestroke programs are collecting at least short-term outcome or discharge data, measured in National Institutes of Health Stroke Scale (NIHSS) scores at admission and discharge. Dr. Madhavan’s team recently reported that, over 4 years, patients receiving intravenous TPA through the Michigan Stroke Network experienced a greater than seven-point reduction in NIHSS score by the end of their hospital stay (Stroke 2012;43:A2991).
Wake Forest Baptist tracks as a surrogate measurement whether patients are discharged to home, rehabilitation, or skilled nursing – and 79% are discharged to home or rehab, Dr. Tegeler said.
A few programs are collecting data on 90-day outcomes, an important measurement in clinical trials. For now, though, outcomes are mainly being analyzed with an aim to improve treatment protocols.
Although several studies have demonstrated that outcomes in the "drip and ship" model are comparable with those of patients admitted and treated at dedicated stroke centers, reducing average door-to-needle times and deciding who benefits from "drip and ship" versus "drip and keep" remain key areas of interest.
At the International Stroke Conference in February, neurologist Dr. Shadi Yaghi presented research on a cohort of 562 patients in the Arkansas SAVES program, a telestroke network that links 22 hospitals to stroke specialists at the University of Arkansas Medical Sciences Center in Little Rock. Patients with a worse outcome at 90 days were more likely to have received postthrombolysis care at a spoke hospital (odds ratio, 15.63; P = .019; 95% CI 1.56-166.67), Dr. Yaghi and his colleagues found (Stroke 2012;43:A2885).
"Our study showed that patients with moderate to severe strokes had better outcomes when transferred to a primary stroke center, likely due to more available resources and treatment options, such as neurointerventional treatment and neurocritical care treatment. Those with mild symptoms did not benefit from the transfer," Dr. Yaghi said in an e-mail interview.
Beyond TPA
In Europe and, in particular, Germany, telestroke has developed along a different course, with an emphasis on building multidisciplinary telestroke units in network hospitals.
Instead of concentrating solely on identifying candidates for TPA, network hospitals have teams of nurses, physiotherapists, occupational therapists, speech and language therapists, and physicians trained through seminars held at stroke centers and grand rounds in the affiliated hospitals. Workup of stroke patients is standardized, and treatment includes TPA for qualified patients under a drip-and-keep model.
Neurologist Dr. Peter Müller-Barna of Klinikum Harlaching, Stadtisches Klinikum in Munich, who is project coordinator of the TEMPiS (Telemedic Pilot Project for Integrative Stroke Care) stroke network in Bavaria, said in an interview that dedicated stroke units have proven "very efficient in reducing mortality and dependency in all stroke patients, with a number needed to treat (NNT) of about 5. In comparison to this, intravenous TPA is less effective; the NNT is about 10. Taken together, building up telestroke units is more expensive, but also by far more effective."
The TEMPiS network is one of the most intensively studied telestroke projects in terms of short- and long-term outcomes (Lancet Neurol. 2006;5:742-8; Neurology 2007;69:898-903). Recently, Dr. Müller-Barna and colleagues showed quality of care in TEMPiS network hospitals to be comparable with that seen at conventional stroke units with neurologists (Stroke 2012;43:A2820).
"In my eyes, the main obstacle for not establishing more telestroke programs in the U.S. is the lack of evidence for the telethrombolysis network approach in terms of improved clinical outcome, in contrast to the integrative stroke network approach with its telestroke units," Dr. Müller-Barna said.
Dr. Schwamm, who has collaborated with Dr. Müller-Barna’s group for research, said that much of the difference in the European and American telestroke models can be attributed to the differences in health care systems overall, with European systems better set up to invest in telestroke networks, keep patients in hospitals, and track long-term outcomes.
"In the U.S. the primary driver is the need to provide acute neurological assessment and make sure TPA-eligible patients have a chance to be treated," Dr. Schwamm said. "But telestroke still has lots of collateral benefits. It’s not only drip and keep and drip and ship but evaluate and triage. There’s a lot of that that goes on in telestroke networks even though that’s not the primary driver."
Dr. Tegeler said that the Wake Forest Baptist program is increasingly looking at telestroke consultations as a pathway to other types of interventions.
"For patients who may have missed the window for TPA, telestroke can help facilitate consideration of eligibility for interventional or intraarterial procedures – there’s a whole host of things you could do with the right patients with the right lesions after the 4.5-hour window."
Dr. Tegeler and colleagues at Wake Forest Baptist have successfully pilot tested the use of telestroke technology for postdischarge follow-up visits, allowing patients who were transferred to the medical center for stroke treatment to be followed up, via videoconferencing, at spoke hospitals closer to their homes.
"There are opportunities to use it in all kinds of ways," he said.
Dr. Levine said that he has served on an advisory board and as a consultant for Genentech, which makes TPA (honorarium donated to stroke research), and has received reimbursement from Genentech for travel to a scientific meeting. He also has served as an expert witness in medical-legal cases involving stroke. Dr. Tegeler disclosed serving on the speakers bureau for Genentech. Dr. Schwamm reported being an advisory board member for lifeIMAGE, a company working on technology for the sharing of brain images in telemedicine. Dr. Madhavan said that he is a consultant for Robolytics, Process Proxy Corp., Great Lakes NeuroTechnologies, and MatrixView Ltd. No other sources had any relevant disclosures.
When Dr. Steven R. Levine and Dr. Mark Gorman first proposed that emergency physicians in small rural hospitals link up via bedside video cameras to stroke specialists in large urban hospitals to triage stroke patients, they named the concept "telestroke."
"I never thought it would catch on," Dr. Levine said in a recent interview.
It was 1999, and while thrombolysis with tissue plasminogen activator (TPA) had by then been approved for 3 years, only about 1% of acute ischemic stroke patients in the United States were receiving it, when as many as half could potentially have been eligible.
One important reason for the underuse of TPA was that smaller and more rural hospitals did not have neurologists available to respond to stroke calls. Patients would have to be transported to larger centers to receive intravenous TPA, and by the time they arrived, many would have missed the time window – which published guidelines have now determined to be up to 4.5 hours after a stroke – when TPA can be safely administered.
Dr. Levine, currently professor of neurology and emergency medicine at SUNY Downstate Medical Center in Brooklyn, N.Y., and a leading researcher and proponent of telestroke, envisioned a spoke-and-hub model of care in which smaller hospitals would be linked by video to larger ones with neurologists available, in active shifts, for two-way consults (Stroke 1999;30:464-9).
TPA could be initiated in the emergency departments of the spokes, and higher-risk patients would be transferred to hub hospitals with dedicated stroke centers once a drip was started.
Early adopters included Massachusetts General Hospital, which began building on the spoke-and-hub model and proving the concept in study after study. "We were the first to demonstrate that the NIH stroke scale could be done with equivalent reliability over telemedicine, and to show that a neurologist could interpret information on a laptop computer as effectively as a radiologist could on expensive imaging systems," said Dr. Lee H. Schwamm, professor of neurology at Harvard University and director of the Partners TeleStroke center, a 30-hospital network run out of Massachusetts General and Brigham and Women’s Hospital, both in Boston.
In 2009, on the strength of evidence showing that telestroke programs increased TPA administration rates, Dr. Schwamm and colleagues, including Dr. Levine, drafted a policy statement for the American Heart Association (Stroke 2009;40:2635-60) advocating the creation of telestroke networks wherever resources were available. More recent evidence has shown telestroke to be cost-effective compared with conventional care (Neurology 2011;77:1590-8; Stroke 2012;43:A3077).
Currently there are at least 27 networks up and running in the United States and Canada, along with 14 in Europe, following spoke-and-hub models. Some comprise a handful of hospitals; others, dozens. Many have existed for only a few years.
North American telestroke programs can now boast TPA rates between 10% and 20% of patients admitted for stroke, even in very rural networks, when the U.S. national average is estimated to be about 5%. In Europe, established programs are reporting TPA rates between 12% and 17%, according to a recent meta-analysis (Curr. Opin. Neurol. 2012;25:5-10).
Dr. Charles H. Tegeler, professor of neurology and head of the telestroke program at Wake Forest Baptist Medical Center in Winston-Salem, N.C., said in an interview that his program, which started in 2009 and now incorporates nine network hospitals, has seen TPA administered in 27% of patients who receive a video stroke consultation. And many patients are receiving TPA in hospitals that, before joining the network, did not administer it.
About 45% of patients receiving a telestroke consultation through one of the network hospitals are transferred to Wake Forest Baptist, or another dedicated stroke center of the patient’s choice, for additional evaluation and management. Depending on local resources and expertise, those treated with TPA may be transferred under a treatment model known as "drip and ship." The rest remain at the network hospital for ongoing care, or "drip and keep."
Rayetta Johnson, R.N., stroke program manager for Wake Forest Baptist, said in an interview that an important factor in its success has been achieving close cooperation, through training seminars and other forms of outreach, with hospital emergency department staff at all levels. The program also reached out extensively to local emergency medical services accustomed to rerouting stroke patients to larger hospitals. "Having EMS on board is extremely important," Ms. Johnson said.
Barriers to Adoption
If telestroke programs are cost effective and improve TPA rates dramatically, why aren’t they more widespread?
Part of the problem, experts say, lies with neurologists’ hesitance to join telestroke networks because reimbursement for telestroke is less straightforward than for a traditional stroke call, and many consider it to be inadequate under current Medicare and Medicaid guidelines.
Dr. Ramesh Madhavan, director of telemedicine for Wayne State University, Detroit, and the Michigan Stroke Network, one of the largest telestroke programs in the country with 36 affiliated hospitals, said in an interview that his program’s eight neurologists – including himself – take stroke calls over 12-hour shifts.
"We take shifts because there is no direct reimbursement, and we have to do other things during the course of that time. We have to multitask," Dr. Madhavan said, adding that this can make some neurologists feel burned out.
Dr. Tegeler said that, by contrast, the five vascular neurologists in Wake Forest Baptist’s smaller program have incorporated the telestroke coverage as part of their regular stroke attending call duties. Now, neurologists at some of the network hospitals "may not have to disrupt their office schedule for an hour or more to go over to the hospital to see an acute stroke patient," he said.
The presence of telestroke coverage also was used by one network hospital to help recruit a neurologist to a community where there had not been one, since the local neurologist would "not have to take stroke calls 24-7," Dr. Tegeler said.
In addition to the reimbursement issue, Dr. Levine said that telestroke has been hampered by "fears that the technology won’t work or may break down, or of lawsuits, and also doctors’ fear of something more technologically advanced than some are used to doing." Unlike their counterparts in emergency departments, Dr. Levine said, "neurologists aren’t techies, as a rule." However, telestroke experts are increasingly exploring cheaper and more portable options for videoconferencing, which could prove less imposing in terms of both startup costs to networks and in ease of use.
Most telestroke programs currently employ videoconferencing technology using equipment mounted on a mobile cart or purpose-built robots that can be driven to a patient’s bedside.
The robots, Dr. Schwamm said, "are a luxury. You can put a laptop with a specialized camera on top of a cart and roll it to a patient and get everything you need. Technology should not be where the costs are."
One team of researchers recently demonstrated that iPhones could be used in telestroke networks for patient assessment (J. Stroke Cerebrovasc. Dis. 2011 Oct. 24 [doi:10.1016/j.jstrokecerebrovasdis.2011.09.013]).
Dr. Madhavan said that he has been working with several technology companies on applications that would allow more telestroke consults to be carried out on portable devices using 3G or 4G networks. A migration to mobile platforms might help improve door-to-needle times in addition to reducing costs, he said.
Research Priorities
When Dr. Levine and Dr. Gorman first proposed telestroke networks in 1999, they envisioned them not solely as a way to increase TPA uptake but also as a way to facilitate patient entry into clinical trials of new stroke treatments.
This has yet to happen directly, Dr. Levine noted, but it may soon. "Community hospitals are not used to dealing with experimental medicines, or the logistics of randomization, and working with trial coordinators, IRBs, and rigorous and extensive data collection. But now that the systems are being built and there’s some infrastructure over the last 10 years, hospitals will hopefully start to see what clinical trials can bring them in terms of recruitment, state-of-the-art care while testing the most novel and promising treatments, and financially," he said.
Dr. Schwamm, whose program is among the few actively seeking to design clinical trials using telestroke, said that enrolling patients through telemedicine – and obtaining consent through video – is a challenge that, if met, will help broaden trial populations to reach more people living in rural and resource-poor areas. "The big studies tend to be done at the teaching hospitals, in urban areas. Telestroke can help allow people in the community to be enrolled," he said, leading to better representation of the population at large.
More telestroke programs are collecting at least short-term outcome or discharge data, measured in National Institutes of Health Stroke Scale (NIHSS) scores at admission and discharge. Dr. Madhavan’s team recently reported that, over 4 years, patients receiving intravenous TPA through the Michigan Stroke Network experienced a greater than seven-point reduction in NIHSS score by the end of their hospital stay (Stroke 2012;43:A2991).
Wake Forest Baptist tracks as a surrogate measurement whether patients are discharged to home, rehabilitation, or skilled nursing – and 79% are discharged to home or rehab, Dr. Tegeler said.
A few programs are collecting data on 90-day outcomes, an important measurement in clinical trials. For now, though, outcomes are mainly being analyzed with an aim to improve treatment protocols.
Although several studies have demonstrated that outcomes in the "drip and ship" model are comparable with those of patients admitted and treated at dedicated stroke centers, reducing average door-to-needle times and deciding who benefits from "drip and ship" versus "drip and keep" remain key areas of interest.
At the International Stroke Conference in February, neurologist Dr. Shadi Yaghi presented research on a cohort of 562 patients in the Arkansas SAVES program, a telestroke network that links 22 hospitals to stroke specialists at the University of Arkansas Medical Sciences Center in Little Rock. Patients with a worse outcome at 90 days were more likely to have received postthrombolysis care at a spoke hospital (odds ratio, 15.63; P = .019; 95% CI 1.56-166.67), Dr. Yaghi and his colleagues found (Stroke 2012;43:A2885).
"Our study showed that patients with moderate to severe strokes had better outcomes when transferred to a primary stroke center, likely due to more available resources and treatment options, such as neurointerventional treatment and neurocritical care treatment. Those with mild symptoms did not benefit from the transfer," Dr. Yaghi said in an e-mail interview.
Beyond TPA
In Europe and, in particular, Germany, telestroke has developed along a different course, with an emphasis on building multidisciplinary telestroke units in network hospitals.
Instead of concentrating solely on identifying candidates for TPA, network hospitals have teams of nurses, physiotherapists, occupational therapists, speech and language therapists, and physicians trained through seminars held at stroke centers and grand rounds in the affiliated hospitals. Workup of stroke patients is standardized, and treatment includes TPA for qualified patients under a drip-and-keep model.
Neurologist Dr. Peter Müller-Barna of Klinikum Harlaching, Stadtisches Klinikum in Munich, who is project coordinator of the TEMPiS (Telemedic Pilot Project for Integrative Stroke Care) stroke network in Bavaria, said in an interview that dedicated stroke units have proven "very efficient in reducing mortality and dependency in all stroke patients, with a number needed to treat (NNT) of about 5. In comparison to this, intravenous TPA is less effective; the NNT is about 10. Taken together, building up telestroke units is more expensive, but also by far more effective."
The TEMPiS network is one of the most intensively studied telestroke projects in terms of short- and long-term outcomes (Lancet Neurol. 2006;5:742-8; Neurology 2007;69:898-903). Recently, Dr. Müller-Barna and colleagues showed quality of care in TEMPiS network hospitals to be comparable with that seen at conventional stroke units with neurologists (Stroke 2012;43:A2820).
"In my eyes, the main obstacle for not establishing more telestroke programs in the U.S. is the lack of evidence for the telethrombolysis network approach in terms of improved clinical outcome, in contrast to the integrative stroke network approach with its telestroke units," Dr. Müller-Barna said.
Dr. Schwamm, who has collaborated with Dr. Müller-Barna’s group for research, said that much of the difference in the European and American telestroke models can be attributed to the differences in health care systems overall, with European systems better set up to invest in telestroke networks, keep patients in hospitals, and track long-term outcomes.
"In the U.S. the primary driver is the need to provide acute neurological assessment and make sure TPA-eligible patients have a chance to be treated," Dr. Schwamm said. "But telestroke still has lots of collateral benefits. It’s not only drip and keep and drip and ship but evaluate and triage. There’s a lot of that that goes on in telestroke networks even though that’s not the primary driver."
Dr. Tegeler said that the Wake Forest Baptist program is increasingly looking at telestroke consultations as a pathway to other types of interventions.
"For patients who may have missed the window for TPA, telestroke can help facilitate consideration of eligibility for interventional or intraarterial procedures – there’s a whole host of things you could do with the right patients with the right lesions after the 4.5-hour window."
Dr. Tegeler and colleagues at Wake Forest Baptist have successfully pilot tested the use of telestroke technology for postdischarge follow-up visits, allowing patients who were transferred to the medical center for stroke treatment to be followed up, via videoconferencing, at spoke hospitals closer to their homes.
"There are opportunities to use it in all kinds of ways," he said.
Dr. Levine said that he has served on an advisory board and as a consultant for Genentech, which makes TPA (honorarium donated to stroke research), and has received reimbursement from Genentech for travel to a scientific meeting. He also has served as an expert witness in medical-legal cases involving stroke. Dr. Tegeler disclosed serving on the speakers bureau for Genentech. Dr. Schwamm reported being an advisory board member for lifeIMAGE, a company working on technology for the sharing of brain images in telemedicine. Dr. Madhavan said that he is a consultant for Robolytics, Process Proxy Corp., Great Lakes NeuroTechnologies, and MatrixView Ltd. No other sources had any relevant disclosures.
When Dr. Steven R. Levine and Dr. Mark Gorman first proposed that emergency physicians in small rural hospitals link up via bedside video cameras to stroke specialists in large urban hospitals to triage stroke patients, they named the concept "telestroke."
"I never thought it would catch on," Dr. Levine said in a recent interview.
It was 1999, and while thrombolysis with tissue plasminogen activator (TPA) had by then been approved for 3 years, only about 1% of acute ischemic stroke patients in the United States were receiving it, when as many as half could potentially have been eligible.
One important reason for the underuse of TPA was that smaller and more rural hospitals did not have neurologists available to respond to stroke calls. Patients would have to be transported to larger centers to receive intravenous TPA, and by the time they arrived, many would have missed the time window – which published guidelines have now determined to be up to 4.5 hours after a stroke – when TPA can be safely administered.
Dr. Levine, currently professor of neurology and emergency medicine at SUNY Downstate Medical Center in Brooklyn, N.Y., and a leading researcher and proponent of telestroke, envisioned a spoke-and-hub model of care in which smaller hospitals would be linked by video to larger ones with neurologists available, in active shifts, for two-way consults (Stroke 1999;30:464-9).
TPA could be initiated in the emergency departments of the spokes, and higher-risk patients would be transferred to hub hospitals with dedicated stroke centers once a drip was started.
Early adopters included Massachusetts General Hospital, which began building on the spoke-and-hub model and proving the concept in study after study. "We were the first to demonstrate that the NIH stroke scale could be done with equivalent reliability over telemedicine, and to show that a neurologist could interpret information on a laptop computer as effectively as a radiologist could on expensive imaging systems," said Dr. Lee H. Schwamm, professor of neurology at Harvard University and director of the Partners TeleStroke center, a 30-hospital network run out of Massachusetts General and Brigham and Women’s Hospital, both in Boston.
In 2009, on the strength of evidence showing that telestroke programs increased TPA administration rates, Dr. Schwamm and colleagues, including Dr. Levine, drafted a policy statement for the American Heart Association (Stroke 2009;40:2635-60) advocating the creation of telestroke networks wherever resources were available. More recent evidence has shown telestroke to be cost-effective compared with conventional care (Neurology 2011;77:1590-8; Stroke 2012;43:A3077).
Currently there are at least 27 networks up and running in the United States and Canada, along with 14 in Europe, following spoke-and-hub models. Some comprise a handful of hospitals; others, dozens. Many have existed for only a few years.
North American telestroke programs can now boast TPA rates between 10% and 20% of patients admitted for stroke, even in very rural networks, when the U.S. national average is estimated to be about 5%. In Europe, established programs are reporting TPA rates between 12% and 17%, according to a recent meta-analysis (Curr. Opin. Neurol. 2012;25:5-10).
Dr. Charles H. Tegeler, professor of neurology and head of the telestroke program at Wake Forest Baptist Medical Center in Winston-Salem, N.C., said in an interview that his program, which started in 2009 and now incorporates nine network hospitals, has seen TPA administered in 27% of patients who receive a video stroke consultation. And many patients are receiving TPA in hospitals that, before joining the network, did not administer it.
About 45% of patients receiving a telestroke consultation through one of the network hospitals are transferred to Wake Forest Baptist, or another dedicated stroke center of the patient’s choice, for additional evaluation and management. Depending on local resources and expertise, those treated with TPA may be transferred under a treatment model known as "drip and ship." The rest remain at the network hospital for ongoing care, or "drip and keep."
Rayetta Johnson, R.N., stroke program manager for Wake Forest Baptist, said in an interview that an important factor in its success has been achieving close cooperation, through training seminars and other forms of outreach, with hospital emergency department staff at all levels. The program also reached out extensively to local emergency medical services accustomed to rerouting stroke patients to larger hospitals. "Having EMS on board is extremely important," Ms. Johnson said.
Barriers to Adoption
If telestroke programs are cost effective and improve TPA rates dramatically, why aren’t they more widespread?
Part of the problem, experts say, lies with neurologists’ hesitance to join telestroke networks because reimbursement for telestroke is less straightforward than for a traditional stroke call, and many consider it to be inadequate under current Medicare and Medicaid guidelines.
Dr. Ramesh Madhavan, director of telemedicine for Wayne State University, Detroit, and the Michigan Stroke Network, one of the largest telestroke programs in the country with 36 affiliated hospitals, said in an interview that his program’s eight neurologists – including himself – take stroke calls over 12-hour shifts.
"We take shifts because there is no direct reimbursement, and we have to do other things during the course of that time. We have to multitask," Dr. Madhavan said, adding that this can make some neurologists feel burned out.
Dr. Tegeler said that, by contrast, the five vascular neurologists in Wake Forest Baptist’s smaller program have incorporated the telestroke coverage as part of their regular stroke attending call duties. Now, neurologists at some of the network hospitals "may not have to disrupt their office schedule for an hour or more to go over to the hospital to see an acute stroke patient," he said.
The presence of telestroke coverage also was used by one network hospital to help recruit a neurologist to a community where there had not been one, since the local neurologist would "not have to take stroke calls 24-7," Dr. Tegeler said.
In addition to the reimbursement issue, Dr. Levine said that telestroke has been hampered by "fears that the technology won’t work or may break down, or of lawsuits, and also doctors’ fear of something more technologically advanced than some are used to doing." Unlike their counterparts in emergency departments, Dr. Levine said, "neurologists aren’t techies, as a rule." However, telestroke experts are increasingly exploring cheaper and more portable options for videoconferencing, which could prove less imposing in terms of both startup costs to networks and in ease of use.
Most telestroke programs currently employ videoconferencing technology using equipment mounted on a mobile cart or purpose-built robots that can be driven to a patient’s bedside.
The robots, Dr. Schwamm said, "are a luxury. You can put a laptop with a specialized camera on top of a cart and roll it to a patient and get everything you need. Technology should not be where the costs are."
One team of researchers recently demonstrated that iPhones could be used in telestroke networks for patient assessment (J. Stroke Cerebrovasc. Dis. 2011 Oct. 24 [doi:10.1016/j.jstrokecerebrovasdis.2011.09.013]).
Dr. Madhavan said that he has been working with several technology companies on applications that would allow more telestroke consults to be carried out on portable devices using 3G or 4G networks. A migration to mobile platforms might help improve door-to-needle times in addition to reducing costs, he said.
Research Priorities
When Dr. Levine and Dr. Gorman first proposed telestroke networks in 1999, they envisioned them not solely as a way to increase TPA uptake but also as a way to facilitate patient entry into clinical trials of new stroke treatments.
This has yet to happen directly, Dr. Levine noted, but it may soon. "Community hospitals are not used to dealing with experimental medicines, or the logistics of randomization, and working with trial coordinators, IRBs, and rigorous and extensive data collection. But now that the systems are being built and there’s some infrastructure over the last 10 years, hospitals will hopefully start to see what clinical trials can bring them in terms of recruitment, state-of-the-art care while testing the most novel and promising treatments, and financially," he said.
Dr. Schwamm, whose program is among the few actively seeking to design clinical trials using telestroke, said that enrolling patients through telemedicine – and obtaining consent through video – is a challenge that, if met, will help broaden trial populations to reach more people living in rural and resource-poor areas. "The big studies tend to be done at the teaching hospitals, in urban areas. Telestroke can help allow people in the community to be enrolled," he said, leading to better representation of the population at large.
More telestroke programs are collecting at least short-term outcome or discharge data, measured in National Institutes of Health Stroke Scale (NIHSS) scores at admission and discharge. Dr. Madhavan’s team recently reported that, over 4 years, patients receiving intravenous TPA through the Michigan Stroke Network experienced a greater than seven-point reduction in NIHSS score by the end of their hospital stay (Stroke 2012;43:A2991).
Wake Forest Baptist tracks as a surrogate measurement whether patients are discharged to home, rehabilitation, or skilled nursing – and 79% are discharged to home or rehab, Dr. Tegeler said.
A few programs are collecting data on 90-day outcomes, an important measurement in clinical trials. For now, though, outcomes are mainly being analyzed with an aim to improve treatment protocols.
Although several studies have demonstrated that outcomes in the "drip and ship" model are comparable with those of patients admitted and treated at dedicated stroke centers, reducing average door-to-needle times and deciding who benefits from "drip and ship" versus "drip and keep" remain key areas of interest.
At the International Stroke Conference in February, neurologist Dr. Shadi Yaghi presented research on a cohort of 562 patients in the Arkansas SAVES program, a telestroke network that links 22 hospitals to stroke specialists at the University of Arkansas Medical Sciences Center in Little Rock. Patients with a worse outcome at 90 days were more likely to have received postthrombolysis care at a spoke hospital (odds ratio, 15.63; P = .019; 95% CI 1.56-166.67), Dr. Yaghi and his colleagues found (Stroke 2012;43:A2885).
"Our study showed that patients with moderate to severe strokes had better outcomes when transferred to a primary stroke center, likely due to more available resources and treatment options, such as neurointerventional treatment and neurocritical care treatment. Those with mild symptoms did not benefit from the transfer," Dr. Yaghi said in an e-mail interview.
Beyond TPA
In Europe and, in particular, Germany, telestroke has developed along a different course, with an emphasis on building multidisciplinary telestroke units in network hospitals.
Instead of concentrating solely on identifying candidates for TPA, network hospitals have teams of nurses, physiotherapists, occupational therapists, speech and language therapists, and physicians trained through seminars held at stroke centers and grand rounds in the affiliated hospitals. Workup of stroke patients is standardized, and treatment includes TPA for qualified patients under a drip-and-keep model.
Neurologist Dr. Peter Müller-Barna of Klinikum Harlaching, Stadtisches Klinikum in Munich, who is project coordinator of the TEMPiS (Telemedic Pilot Project for Integrative Stroke Care) stroke network in Bavaria, said in an interview that dedicated stroke units have proven "very efficient in reducing mortality and dependency in all stroke patients, with a number needed to treat (NNT) of about 5. In comparison to this, intravenous TPA is less effective; the NNT is about 10. Taken together, building up telestroke units is more expensive, but also by far more effective."
The TEMPiS network is one of the most intensively studied telestroke projects in terms of short- and long-term outcomes (Lancet Neurol. 2006;5:742-8; Neurology 2007;69:898-903). Recently, Dr. Müller-Barna and colleagues showed quality of care in TEMPiS network hospitals to be comparable with that seen at conventional stroke units with neurologists (Stroke 2012;43:A2820).
"In my eyes, the main obstacle for not establishing more telestroke programs in the U.S. is the lack of evidence for the telethrombolysis network approach in terms of improved clinical outcome, in contrast to the integrative stroke network approach with its telestroke units," Dr. Müller-Barna said.
Dr. Schwamm, who has collaborated with Dr. Müller-Barna’s group for research, said that much of the difference in the European and American telestroke models can be attributed to the differences in health care systems overall, with European systems better set up to invest in telestroke networks, keep patients in hospitals, and track long-term outcomes.
"In the U.S. the primary driver is the need to provide acute neurological assessment and make sure TPA-eligible patients have a chance to be treated," Dr. Schwamm said. "But telestroke still has lots of collateral benefits. It’s not only drip and keep and drip and ship but evaluate and triage. There’s a lot of that that goes on in telestroke networks even though that’s not the primary driver."
Dr. Tegeler said that the Wake Forest Baptist program is increasingly looking at telestroke consultations as a pathway to other types of interventions.
"For patients who may have missed the window for TPA, telestroke can help facilitate consideration of eligibility for interventional or intraarterial procedures – there’s a whole host of things you could do with the right patients with the right lesions after the 4.5-hour window."
Dr. Tegeler and colleagues at Wake Forest Baptist have successfully pilot tested the use of telestroke technology for postdischarge follow-up visits, allowing patients who were transferred to the medical center for stroke treatment to be followed up, via videoconferencing, at spoke hospitals closer to their homes.
"There are opportunities to use it in all kinds of ways," he said.
Dr. Levine said that he has served on an advisory board and as a consultant for Genentech, which makes TPA (honorarium donated to stroke research), and has received reimbursement from Genentech for travel to a scientific meeting. He also has served as an expert witness in medical-legal cases involving stroke. Dr. Tegeler disclosed serving on the speakers bureau for Genentech. Dr. Schwamm reported being an advisory board member for lifeIMAGE, a company working on technology for the sharing of brain images in telemedicine. Dr. Madhavan said that he is a consultant for Robolytics, Process Proxy Corp., Great Lakes NeuroTechnologies, and MatrixView Ltd. No other sources had any relevant disclosures.
Low PTH Levels Linked to Fracture Risk in Diabetic Women
In postmenopausal women with type 2 diabetes, low levels of parathyroid hormone are associated with an increased risk for vertebral fractures, researchers in Japan have found.
The increase in risk is not associated with poor bone mineral density but rather poor bone formation. Bone quality is measured by both density and turnover, and serum parathyroid hormone (PTH) levels were seen as significantly correlated with lower levels of osteocalcin – a chemical indicator of bone turnover – in both men and women with type 2 diabetes.
However, while the correlation between lower PTH and osteocalcin was seen in diabetics of both sexes, only the diabetic women in the study were seen at elevated fracture risk independent of bone mineral density (J. Clin. Endocrinol. Metab. 2012 Feb. 15 [doi:10.1210/jc.2011-2537]).
The findings help tease apart the complex etiological relationship between type 2 diabetes and fracture risk.
A study by the same group of researchers, led by Dr. Masahiro Yamamoto of the Shimane (Japan) University Faculty of Medicine, established recently that diabetic patients had an increased risk of vertebral fractures independent of bone mineral density (J. Bone Miner. Res. 2009;24:702-9).
For their current study, Dr. Yamamoto and colleagues recruited hospitalized diabetic patients (123 postmenopausal women between the ages of 45 and 80, and 132 men aged 50-82) and nondiabetic controls (189 women and 51 men), measuring PTH and osteocalcin levels and adjusting for age, body mass index, hemoglobin A1c, creatinine, 25-hydroxyvitamin D, and lumbar bone mineral density.
Subjects and controls were divided into subgroups based on mean PTH and osteocalcin levels. Threshold values for women were 34.7 pg/mL for PTH and 18.1 ng/mL for osteocalcin, and for men, 24.1 pg/mL for PTH and 10.7 ng/mL for osteocalcin.
Diabetes patients of both sexes were seen to have lower PTH and osteocalcin levels than controls. Multivariate regression analysis showed that PTH levels of diabetes patients significantly correlated with osteocalcin levels for both men and women.
Postmenopausal diabetic women with lower PTH and osteocalcin levels had a significantly, 4.7-fold higher risk of fracture than did those with higher PTH and osteocalcin.
No increased vertebral fracture risk associated with lower PTH and osteocalcin was seen in diabetic men.
Dr. Yamamoto and colleagues wrote that "in contrast to T2DM women, HbA1c levels in T2DM men were significantly and inversely associated with osteocalcin levels after multivariate adjustment, suggesting that osteocalcin levels in T2DM men could be affected by improvement of glycemic conditions," which could help explain the difference in fracture risk.
The investigators listed several limitations of their study, including its small size and the fact that that it was not population based, allowing for the potential for selection bias. The diabetes cases may have represented people with more severe disease than is usual for Japan, they noted.
Dr. Yamamoto and colleagues also noted that they did not test for deficiencies of magnesium, which is known to modulate PTH secretion. Bone metabolic markers may have been affected by prior fractures, they acknowledged, and the influence of hormones, such as estrogen produced by adipose tissue, was not considered.
The study was funded by the Japanese government. Neither Dr. Yamamoto nor his coauthors disclosed conflicts of interest.
In postmenopausal women with type 2 diabetes, low levels of parathyroid hormone are associated with an increased risk for vertebral fractures, researchers in Japan have found.
The increase in risk is not associated with poor bone mineral density but rather poor bone formation. Bone quality is measured by both density and turnover, and serum parathyroid hormone (PTH) levels were seen as significantly correlated with lower levels of osteocalcin – a chemical indicator of bone turnover – in both men and women with type 2 diabetes.
However, while the correlation between lower PTH and osteocalcin was seen in diabetics of both sexes, only the diabetic women in the study were seen at elevated fracture risk independent of bone mineral density (J. Clin. Endocrinol. Metab. 2012 Feb. 15 [doi:10.1210/jc.2011-2537]).
The findings help tease apart the complex etiological relationship between type 2 diabetes and fracture risk.
A study by the same group of researchers, led by Dr. Masahiro Yamamoto of the Shimane (Japan) University Faculty of Medicine, established recently that diabetic patients had an increased risk of vertebral fractures independent of bone mineral density (J. Bone Miner. Res. 2009;24:702-9).
For their current study, Dr. Yamamoto and colleagues recruited hospitalized diabetic patients (123 postmenopausal women between the ages of 45 and 80, and 132 men aged 50-82) and nondiabetic controls (189 women and 51 men), measuring PTH and osteocalcin levels and adjusting for age, body mass index, hemoglobin A1c, creatinine, 25-hydroxyvitamin D, and lumbar bone mineral density.
Subjects and controls were divided into subgroups based on mean PTH and osteocalcin levels. Threshold values for women were 34.7 pg/mL for PTH and 18.1 ng/mL for osteocalcin, and for men, 24.1 pg/mL for PTH and 10.7 ng/mL for osteocalcin.
Diabetes patients of both sexes were seen to have lower PTH and osteocalcin levels than controls. Multivariate regression analysis showed that PTH levels of diabetes patients significantly correlated with osteocalcin levels for both men and women.
Postmenopausal diabetic women with lower PTH and osteocalcin levels had a significantly, 4.7-fold higher risk of fracture than did those with higher PTH and osteocalcin.
No increased vertebral fracture risk associated with lower PTH and osteocalcin was seen in diabetic men.
Dr. Yamamoto and colleagues wrote that "in contrast to T2DM women, HbA1c levels in T2DM men were significantly and inversely associated with osteocalcin levels after multivariate adjustment, suggesting that osteocalcin levels in T2DM men could be affected by improvement of glycemic conditions," which could help explain the difference in fracture risk.
The investigators listed several limitations of their study, including its small size and the fact that that it was not population based, allowing for the potential for selection bias. The diabetes cases may have represented people with more severe disease than is usual for Japan, they noted.
Dr. Yamamoto and colleagues also noted that they did not test for deficiencies of magnesium, which is known to modulate PTH secretion. Bone metabolic markers may have been affected by prior fractures, they acknowledged, and the influence of hormones, such as estrogen produced by adipose tissue, was not considered.
The study was funded by the Japanese government. Neither Dr. Yamamoto nor his coauthors disclosed conflicts of interest.
In postmenopausal women with type 2 diabetes, low levels of parathyroid hormone are associated with an increased risk for vertebral fractures, researchers in Japan have found.
The increase in risk is not associated with poor bone mineral density but rather poor bone formation. Bone quality is measured by both density and turnover, and serum parathyroid hormone (PTH) levels were seen as significantly correlated with lower levels of osteocalcin – a chemical indicator of bone turnover – in both men and women with type 2 diabetes.
However, while the correlation between lower PTH and osteocalcin was seen in diabetics of both sexes, only the diabetic women in the study were seen at elevated fracture risk independent of bone mineral density (J. Clin. Endocrinol. Metab. 2012 Feb. 15 [doi:10.1210/jc.2011-2537]).
The findings help tease apart the complex etiological relationship between type 2 diabetes and fracture risk.
A study by the same group of researchers, led by Dr. Masahiro Yamamoto of the Shimane (Japan) University Faculty of Medicine, established recently that diabetic patients had an increased risk of vertebral fractures independent of bone mineral density (J. Bone Miner. Res. 2009;24:702-9).
For their current study, Dr. Yamamoto and colleagues recruited hospitalized diabetic patients (123 postmenopausal women between the ages of 45 and 80, and 132 men aged 50-82) and nondiabetic controls (189 women and 51 men), measuring PTH and osteocalcin levels and adjusting for age, body mass index, hemoglobin A1c, creatinine, 25-hydroxyvitamin D, and lumbar bone mineral density.
Subjects and controls were divided into subgroups based on mean PTH and osteocalcin levels. Threshold values for women were 34.7 pg/mL for PTH and 18.1 ng/mL for osteocalcin, and for men, 24.1 pg/mL for PTH and 10.7 ng/mL for osteocalcin.
Diabetes patients of both sexes were seen to have lower PTH and osteocalcin levels than controls. Multivariate regression analysis showed that PTH levels of diabetes patients significantly correlated with osteocalcin levels for both men and women.
Postmenopausal diabetic women with lower PTH and osteocalcin levels had a significantly, 4.7-fold higher risk of fracture than did those with higher PTH and osteocalcin.
No increased vertebral fracture risk associated with lower PTH and osteocalcin was seen in diabetic men.
Dr. Yamamoto and colleagues wrote that "in contrast to T2DM women, HbA1c levels in T2DM men were significantly and inversely associated with osteocalcin levels after multivariate adjustment, suggesting that osteocalcin levels in T2DM men could be affected by improvement of glycemic conditions," which could help explain the difference in fracture risk.
The investigators listed several limitations of their study, including its small size and the fact that that it was not population based, allowing for the potential for selection bias. The diabetes cases may have represented people with more severe disease than is usual for Japan, they noted.
Dr. Yamamoto and colleagues also noted that they did not test for deficiencies of magnesium, which is known to modulate PTH secretion. Bone metabolic markers may have been affected by prior fractures, they acknowledged, and the influence of hormones, such as estrogen produced by adipose tissue, was not considered.
The study was funded by the Japanese government. Neither Dr. Yamamoto nor his coauthors disclosed conflicts of interest.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
Major Finding: Of postmenopausal women with diabetes, those with low parathyroid hormone and osteocalcin levels had a 4.7-fold higher risk for vertebral fracture than did those with higher levels.
Data Source: A Japanese study of 255 hospital-based patients with type 2 diabetes and 240 matched controls.
Disclosures: The study was funded by the Japanese government. Neither Dr. Yamamoto nor his coauthors disclosed conflicts of interest.
Tracking Method Improves Outcomes in CRT
Speckle-tracking echocardiography has been shown to significantly improve clinical outcomes when used in cardiac resynchronization therapy, according to results from a randomized, controlled trial.
The finding adds to the increasing body of evidence that individualized placement of the ventricular pacing lead in CRT – away from scar and on the most delayed segment of contraction – can result in better outcomes.
In research published online March 7 in the Journal of the American College of Cardiology, Dr. Fakhar Z. Khan of Papworth Hospital, Cambridge, U.K., and colleagues, showed that 70% of heart-failure patients treated with CRT guided by speckle-tracking saw improvement at 6 months, compared with 55% treated with unguided CRT (J. Am. Coll. Cardiol. 2012 [(doi:10.1016/j.jacc.2011.12.030]).
Cardiac resynchronization therapy is used to coordinate contractions in people with heart failure who have failed medical therapies. Speckle-tracking echocardiography is an imaging technique that tracks interference patterns and natural acoustic reflections to show tissue deformation and motion.
Because recent evidence has increasingly suggested that the optimal positioning of the left ventricular pacing lead in CRT is at the most delayed site of contraction and away from myocardial scar (J. Am. Coll. Cardiol. 2010;55:566-75; J. Am. Coll. Cardiol. 2010;56:774-81), speckle tracking has been used to help identify the ideal sites for each patient. Conventional CRT, by contrast, places the LV lead at a lateral or postlateral branch of the coronary sinus in all patients.
For their study comparing conventional unguided CRT with guided CRT, Dr. Khan and colleagues randomized 220 men and women scheduled to undergo CRT. In the study group (N = 110), patients were analyzed with two-dimensional speckle-tracking radial strain imaging to determine the ideal LV lead placing.
Controls (N = 110) underwent standard unguided CRT. Both patients and assessors were blinded to group assignment before and after surgery. The primary end point of the study was response at 6 months, defined as a 15% or greater reduction in left ventricular end-systolic volume, or LVESV.
Secondary end points included clinical response (defined as improvement in New York Heart Association functional class of at least 1 level), 2-year all-cause mortality, and heart failure–related hospitalization combined with all-cause mortality at 2 years. A total of seven patients in the intervention group and six in the control group died prior to the intervention or were lost to follow-up.
The results showed that the speckle-tracking group had significantly greater proportion of responders at 6 months than did the control group (70% vs. 55%). The tracking group also saw NYHA functional class improve in 83% of patients, compared with 65% in conventional CRT, also a significant difference. Though there were no significant differences in 2-year all-cause mortality, investigators saw significantly lower rates of the combined end point of all cause mortality and hospitalization.
"The conventional approach to resynchronization has been to direct the LV lead to the lateral and posterior wall based on the benefit shown in early hemodynamic studies and the observation that delayed segments predominate at these sites," Dr. Khan and colleagues wrote in their analysis.
"However, recent data support a more individualized approach to LV lead placement with significant interindividual and intraindividual variation in the optimal LV lead position."
Furthermore, the investigators wrote, "in this randomized study, subgroup analyses confirm previous reports that the greatest clinical response is seen in patients with a concordant LV lead, together with improved survival and a reduction in the combined end point of death and heart failure–related hospitalization."
Dr. Khan and colleagues argued that "an individualized approach to LV lead placement should be considered in all patients undergoing CRT for advanced heart failure."
The investigators noted that newer imaging techniques, such as three-dimensional speckle tracking, might be better than the one they used for their study. Inadequate image quality resulted in 11% of initially recruited patients being excluded before randomization, they said.
They also noted as a limitation of their study that it made no attempt to preselect patients on the basis of dyssynchrony parameters, nor did it consider the extent of total scar burden.
"The presence of both of these parameters would tend to reduce the overall benefit, although this would be distributed in both groups. CRT response may therefore be enhanced by integrating measures of dyssynchrony and total scar burden with a targeted approach to lead placement, and such an approach should be tested in future studies," Dr. Khan and colleagues wrote.
Dr. Khan’s study was funded by the Addenbrooke’s Charitable Trust, Papworth Hospital Research and Development Department, Cambridge Biomedical Research Center, the U.K. National Institute for Health Research. The authors had no relevant disclosures.
The inability to predict individual responses has been the Achilles’ heel of heart failure RCTs, including CRT trials. It is remarkable, however, that for CRT, many more efforts have been devoted to identifying and predicting responders. Initial small single-center studies reported a favorable outlook for several echocardiographic measures of mechanical dyssynchrony in predicting responders. A subsequent large multicenter study, however, the PROSPECT (Predictors of Response to CRT) trial, failed to identify a useful measurement using conventional and tissue Doppler-based methods (Circulation 2008;117:2608-16).
This important study not only identified an important parameter for selecting responders but also provided indirect support for efforts aimed at identifying predictors of CRT response in individual patients. Khan et al. should be commended for the successful completion of this RCT, which is likely to not only stimulate further innovations in imaging modalities and technical approaches but also more clinical trials that incorporate the assessment of the role of surgical approaches in their design.
Jalal K. Ghali, M.D., is in the DMC Cardiovascular Institute in Detroit. These remarks were taken from his editorial accompanying Dr. Kahn’s report (J. Am. Coll. Cardiol. 2012 [doi:10.1016/j.jacc.2012.01.018]). He had no relevant disclosures.
The inability to predict individual responses has been the Achilles’ heel of heart failure RCTs, including CRT trials. It is remarkable, however, that for CRT, many more efforts have been devoted to identifying and predicting responders. Initial small single-center studies reported a favorable outlook for several echocardiographic measures of mechanical dyssynchrony in predicting responders. A subsequent large multicenter study, however, the PROSPECT (Predictors of Response to CRT) trial, failed to identify a useful measurement using conventional and tissue Doppler-based methods (Circulation 2008;117:2608-16).
This important study not only identified an important parameter for selecting responders but also provided indirect support for efforts aimed at identifying predictors of CRT response in individual patients. Khan et al. should be commended for the successful completion of this RCT, which is likely to not only stimulate further innovations in imaging modalities and technical approaches but also more clinical trials that incorporate the assessment of the role of surgical approaches in their design.
Jalal K. Ghali, M.D., is in the DMC Cardiovascular Institute in Detroit. These remarks were taken from his editorial accompanying Dr. Kahn’s report (J. Am. Coll. Cardiol. 2012 [doi:10.1016/j.jacc.2012.01.018]). He had no relevant disclosures.
The inability to predict individual responses has been the Achilles’ heel of heart failure RCTs, including CRT trials. It is remarkable, however, that for CRT, many more efforts have been devoted to identifying and predicting responders. Initial small single-center studies reported a favorable outlook for several echocardiographic measures of mechanical dyssynchrony in predicting responders. A subsequent large multicenter study, however, the PROSPECT (Predictors of Response to CRT) trial, failed to identify a useful measurement using conventional and tissue Doppler-based methods (Circulation 2008;117:2608-16).
This important study not only identified an important parameter for selecting responders but also provided indirect support for efforts aimed at identifying predictors of CRT response in individual patients. Khan et al. should be commended for the successful completion of this RCT, which is likely to not only stimulate further innovations in imaging modalities and technical approaches but also more clinical trials that incorporate the assessment of the role of surgical approaches in their design.
Jalal K. Ghali, M.D., is in the DMC Cardiovascular Institute in Detroit. These remarks were taken from his editorial accompanying Dr. Kahn’s report (J. Am. Coll. Cardiol. 2012 [doi:10.1016/j.jacc.2012.01.018]). He had no relevant disclosures.
Speckle-tracking echocardiography has been shown to significantly improve clinical outcomes when used in cardiac resynchronization therapy, according to results from a randomized, controlled trial.
The finding adds to the increasing body of evidence that individualized placement of the ventricular pacing lead in CRT – away from scar and on the most delayed segment of contraction – can result in better outcomes.
In research published online March 7 in the Journal of the American College of Cardiology, Dr. Fakhar Z. Khan of Papworth Hospital, Cambridge, U.K., and colleagues, showed that 70% of heart-failure patients treated with CRT guided by speckle-tracking saw improvement at 6 months, compared with 55% treated with unguided CRT (J. Am. Coll. Cardiol. 2012 [(doi:10.1016/j.jacc.2011.12.030]).
Cardiac resynchronization therapy is used to coordinate contractions in people with heart failure who have failed medical therapies. Speckle-tracking echocardiography is an imaging technique that tracks interference patterns and natural acoustic reflections to show tissue deformation and motion.
Because recent evidence has increasingly suggested that the optimal positioning of the left ventricular pacing lead in CRT is at the most delayed site of contraction and away from myocardial scar (J. Am. Coll. Cardiol. 2010;55:566-75; J. Am. Coll. Cardiol. 2010;56:774-81), speckle tracking has been used to help identify the ideal sites for each patient. Conventional CRT, by contrast, places the LV lead at a lateral or postlateral branch of the coronary sinus in all patients.
For their study comparing conventional unguided CRT with guided CRT, Dr. Khan and colleagues randomized 220 men and women scheduled to undergo CRT. In the study group (N = 110), patients were analyzed with two-dimensional speckle-tracking radial strain imaging to determine the ideal LV lead placing.
Controls (N = 110) underwent standard unguided CRT. Both patients and assessors were blinded to group assignment before and after surgery. The primary end point of the study was response at 6 months, defined as a 15% or greater reduction in left ventricular end-systolic volume, or LVESV.
Secondary end points included clinical response (defined as improvement in New York Heart Association functional class of at least 1 level), 2-year all-cause mortality, and heart failure–related hospitalization combined with all-cause mortality at 2 years. A total of seven patients in the intervention group and six in the control group died prior to the intervention or were lost to follow-up.
The results showed that the speckle-tracking group had significantly greater proportion of responders at 6 months than did the control group (70% vs. 55%). The tracking group also saw NYHA functional class improve in 83% of patients, compared with 65% in conventional CRT, also a significant difference. Though there were no significant differences in 2-year all-cause mortality, investigators saw significantly lower rates of the combined end point of all cause mortality and hospitalization.
"The conventional approach to resynchronization has been to direct the LV lead to the lateral and posterior wall based on the benefit shown in early hemodynamic studies and the observation that delayed segments predominate at these sites," Dr. Khan and colleagues wrote in their analysis.
"However, recent data support a more individualized approach to LV lead placement with significant interindividual and intraindividual variation in the optimal LV lead position."
Furthermore, the investigators wrote, "in this randomized study, subgroup analyses confirm previous reports that the greatest clinical response is seen in patients with a concordant LV lead, together with improved survival and a reduction in the combined end point of death and heart failure–related hospitalization."
Dr. Khan and colleagues argued that "an individualized approach to LV lead placement should be considered in all patients undergoing CRT for advanced heart failure."
The investigators noted that newer imaging techniques, such as three-dimensional speckle tracking, might be better than the one they used for their study. Inadequate image quality resulted in 11% of initially recruited patients being excluded before randomization, they said.
They also noted as a limitation of their study that it made no attempt to preselect patients on the basis of dyssynchrony parameters, nor did it consider the extent of total scar burden.
"The presence of both of these parameters would tend to reduce the overall benefit, although this would be distributed in both groups. CRT response may therefore be enhanced by integrating measures of dyssynchrony and total scar burden with a targeted approach to lead placement, and such an approach should be tested in future studies," Dr. Khan and colleagues wrote.
Dr. Khan’s study was funded by the Addenbrooke’s Charitable Trust, Papworth Hospital Research and Development Department, Cambridge Biomedical Research Center, the U.K. National Institute for Health Research. The authors had no relevant disclosures.
Speckle-tracking echocardiography has been shown to significantly improve clinical outcomes when used in cardiac resynchronization therapy, according to results from a randomized, controlled trial.
The finding adds to the increasing body of evidence that individualized placement of the ventricular pacing lead in CRT – away from scar and on the most delayed segment of contraction – can result in better outcomes.
In research published online March 7 in the Journal of the American College of Cardiology, Dr. Fakhar Z. Khan of Papworth Hospital, Cambridge, U.K., and colleagues, showed that 70% of heart-failure patients treated with CRT guided by speckle-tracking saw improvement at 6 months, compared with 55% treated with unguided CRT (J. Am. Coll. Cardiol. 2012 [(doi:10.1016/j.jacc.2011.12.030]).
Cardiac resynchronization therapy is used to coordinate contractions in people with heart failure who have failed medical therapies. Speckle-tracking echocardiography is an imaging technique that tracks interference patterns and natural acoustic reflections to show tissue deformation and motion.
Because recent evidence has increasingly suggested that the optimal positioning of the left ventricular pacing lead in CRT is at the most delayed site of contraction and away from myocardial scar (J. Am. Coll. Cardiol. 2010;55:566-75; J. Am. Coll. Cardiol. 2010;56:774-81), speckle tracking has been used to help identify the ideal sites for each patient. Conventional CRT, by contrast, places the LV lead at a lateral or postlateral branch of the coronary sinus in all patients.
For their study comparing conventional unguided CRT with guided CRT, Dr. Khan and colleagues randomized 220 men and women scheduled to undergo CRT. In the study group (N = 110), patients were analyzed with two-dimensional speckle-tracking radial strain imaging to determine the ideal LV lead placing.
Controls (N = 110) underwent standard unguided CRT. Both patients and assessors were blinded to group assignment before and after surgery. The primary end point of the study was response at 6 months, defined as a 15% or greater reduction in left ventricular end-systolic volume, or LVESV.
Secondary end points included clinical response (defined as improvement in New York Heart Association functional class of at least 1 level), 2-year all-cause mortality, and heart failure–related hospitalization combined with all-cause mortality at 2 years. A total of seven patients in the intervention group and six in the control group died prior to the intervention or were lost to follow-up.
The results showed that the speckle-tracking group had significantly greater proportion of responders at 6 months than did the control group (70% vs. 55%). The tracking group also saw NYHA functional class improve in 83% of patients, compared with 65% in conventional CRT, also a significant difference. Though there were no significant differences in 2-year all-cause mortality, investigators saw significantly lower rates of the combined end point of all cause mortality and hospitalization.
"The conventional approach to resynchronization has been to direct the LV lead to the lateral and posterior wall based on the benefit shown in early hemodynamic studies and the observation that delayed segments predominate at these sites," Dr. Khan and colleagues wrote in their analysis.
"However, recent data support a more individualized approach to LV lead placement with significant interindividual and intraindividual variation in the optimal LV lead position."
Furthermore, the investigators wrote, "in this randomized study, subgroup analyses confirm previous reports that the greatest clinical response is seen in patients with a concordant LV lead, together with improved survival and a reduction in the combined end point of death and heart failure–related hospitalization."
Dr. Khan and colleagues argued that "an individualized approach to LV lead placement should be considered in all patients undergoing CRT for advanced heart failure."
The investigators noted that newer imaging techniques, such as three-dimensional speckle tracking, might be better than the one they used for their study. Inadequate image quality resulted in 11% of initially recruited patients being excluded before randomization, they said.
They also noted as a limitation of their study that it made no attempt to preselect patients on the basis of dyssynchrony parameters, nor did it consider the extent of total scar burden.
"The presence of both of these parameters would tend to reduce the overall benefit, although this would be distributed in both groups. CRT response may therefore be enhanced by integrating measures of dyssynchrony and total scar burden with a targeted approach to lead placement, and such an approach should be tested in future studies," Dr. Khan and colleagues wrote.
Dr. Khan’s study was funded by the Addenbrooke’s Charitable Trust, Papworth Hospital Research and Development Department, Cambridge Biomedical Research Center, the U.K. National Institute for Health Research. The authors had no relevant disclosures.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Major Finding: 70% of heart failure patients receiving cardiac resynchronization therapy with guided imaging to determine individualized placement saw significant improvement at 6 months, compared with 55% of those receiving CRT using standard placement protocol.
Data Source: These results came from a two-center randomized controlled trial of 220 patients between April 2009 and July 2010.
Disclosures: The authors had no relevant disclosures.
Pegvisomant Looks Safe for Long-Term Acromegaly Treatment
Interim results from an ongoing, registry-based phase IV surveillance study show that the medication pegvisomant is safe and effective in treating acromegaly, a disorder caused by pituitary tumors.
Among 1,288 people treated for acromegaly with pegvisomant between 2004, when the medication was authorized in Europe, and the end of 2009, serious adverse effects occurred in 159 (12.3%), and pegvisomant-related serious adverse effects in 26 (2%). No deaths were attributed to pegvisomant use, though 15 subjects (1.1% of the cohort) died while on treatment. Injection-site reactions were recorded for 28 subjects (2.2%).
Importantly, pegvisomant, which works by blocking insulinlike growth factor 1 (IGF-1), was not seen to contribute to increases in tumor size. Among the 936 subjects with at least two available MRIs, 3.2% saw MRI-confirmed changes in tumor size during the study period, revealing tumor growth to be uncommon during treatment, and 63.2% of subjects had normal IGF-I levels at a mean dose of 18 mg/day after 5 years of treatment.
Adverse liver effects were rare, with elevated AST or ALT levels (higher than three times the normal upper limit) reported in 30 patients (2.5%) among the 1,178 who had at least one liver enzyme test after starting pegvisomant. However, in all subjects with elevated liver tests and follow-up laboratory results, the increases resolved. There were no reports of liver failure.
The findings, published online ahead of print (J. Clin. Endocrinol. Metab. 2012 [doi:10.1210/jc.2011-2508]), come from ACROSTUDY, a registry of people treated with pegvisomant for acromegaly for a mean of 3.7 years and followed up for an additional 2 years as of December 2009, the cutoff date for the interim analysis. About half (51%) of the subjects were male. Overall, mean age at diagnosis was 42.1 years and mean age at start of treatment was about 50 years. Most subjects (1,131) had been previously treated for their disease, 954 with surgery.
The vast majority of subjects lived in Europe, where pegvisomant is prescribed only for patients in whom other interventions have failed. This means that patients with the worst disease activity and the most comorbidities were likely overincluded, said the study’s authors, led by Dr. A. J. van der Lely of Erasmus University Medical Center in Rotterdam, the Netherlands. Most subjects (n = 1,023) had at least one reported comorbidity before starting pegvisomant, with hypertension and diabetes being the most common.
Dr. van der Lely and his colleagues noted that because the observational trial was conducted in a clinical setting, with fewer clinical visits per year than would be expected in a controlled setting, adverse effects could have been underreported.
The efficacy rate of pegvisomant was seen as lower than reported in earlier clinical trials, with only 63.2% of subjects achieving normal IGF-1 levels on treatment. Dr. van der Lely and his colleagues noted that this, too, could have been related to the administration of pegvisomant in clinical settings, where doses and compliance may have varied. They also noted that stricter criteria were used to assess IGF-1 normalization in this study than in the interventional trials, and that a different assay was used to determine IGF-1 levels. The authors concluded that, "analysis of ACROSTUDY [data] show that pegvisomant can control IGF-1 levels in a majority of patients with acromegaly who do not respond adequately to other therapies."
Dr. van der Lely and six of his coauthors acknowledged receiving ongoing honoraria from Pfizer, the manufacturer of pegvisomant. The remaining five coauthors on the study are Pfizer employees.
Interim results from an ongoing, registry-based phase IV surveillance study show that the medication pegvisomant is safe and effective in treating acromegaly, a disorder caused by pituitary tumors.
Among 1,288 people treated for acromegaly with pegvisomant between 2004, when the medication was authorized in Europe, and the end of 2009, serious adverse effects occurred in 159 (12.3%), and pegvisomant-related serious adverse effects in 26 (2%). No deaths were attributed to pegvisomant use, though 15 subjects (1.1% of the cohort) died while on treatment. Injection-site reactions were recorded for 28 subjects (2.2%).
Importantly, pegvisomant, which works by blocking insulinlike growth factor 1 (IGF-1), was not seen to contribute to increases in tumor size. Among the 936 subjects with at least two available MRIs, 3.2% saw MRI-confirmed changes in tumor size during the study period, revealing tumor growth to be uncommon during treatment, and 63.2% of subjects had normal IGF-I levels at a mean dose of 18 mg/day after 5 years of treatment.
Adverse liver effects were rare, with elevated AST or ALT levels (higher than three times the normal upper limit) reported in 30 patients (2.5%) among the 1,178 who had at least one liver enzyme test after starting pegvisomant. However, in all subjects with elevated liver tests and follow-up laboratory results, the increases resolved. There were no reports of liver failure.
The findings, published online ahead of print (J. Clin. Endocrinol. Metab. 2012 [doi:10.1210/jc.2011-2508]), come from ACROSTUDY, a registry of people treated with pegvisomant for acromegaly for a mean of 3.7 years and followed up for an additional 2 years as of December 2009, the cutoff date for the interim analysis. About half (51%) of the subjects were male. Overall, mean age at diagnosis was 42.1 years and mean age at start of treatment was about 50 years. Most subjects (1,131) had been previously treated for their disease, 954 with surgery.
The vast majority of subjects lived in Europe, where pegvisomant is prescribed only for patients in whom other interventions have failed. This means that patients with the worst disease activity and the most comorbidities were likely overincluded, said the study’s authors, led by Dr. A. J. van der Lely of Erasmus University Medical Center in Rotterdam, the Netherlands. Most subjects (n = 1,023) had at least one reported comorbidity before starting pegvisomant, with hypertension and diabetes being the most common.
Dr. van der Lely and his colleagues noted that because the observational trial was conducted in a clinical setting, with fewer clinical visits per year than would be expected in a controlled setting, adverse effects could have been underreported.
The efficacy rate of pegvisomant was seen as lower than reported in earlier clinical trials, with only 63.2% of subjects achieving normal IGF-1 levels on treatment. Dr. van der Lely and his colleagues noted that this, too, could have been related to the administration of pegvisomant in clinical settings, where doses and compliance may have varied. They also noted that stricter criteria were used to assess IGF-1 normalization in this study than in the interventional trials, and that a different assay was used to determine IGF-1 levels. The authors concluded that, "analysis of ACROSTUDY [data] show that pegvisomant can control IGF-1 levels in a majority of patients with acromegaly who do not respond adequately to other therapies."
Dr. van der Lely and six of his coauthors acknowledged receiving ongoing honoraria from Pfizer, the manufacturer of pegvisomant. The remaining five coauthors on the study are Pfizer employees.
Interim results from an ongoing, registry-based phase IV surveillance study show that the medication pegvisomant is safe and effective in treating acromegaly, a disorder caused by pituitary tumors.
Among 1,288 people treated for acromegaly with pegvisomant between 2004, when the medication was authorized in Europe, and the end of 2009, serious adverse effects occurred in 159 (12.3%), and pegvisomant-related serious adverse effects in 26 (2%). No deaths were attributed to pegvisomant use, though 15 subjects (1.1% of the cohort) died while on treatment. Injection-site reactions were recorded for 28 subjects (2.2%).
Importantly, pegvisomant, which works by blocking insulinlike growth factor 1 (IGF-1), was not seen to contribute to increases in tumor size. Among the 936 subjects with at least two available MRIs, 3.2% saw MRI-confirmed changes in tumor size during the study period, revealing tumor growth to be uncommon during treatment, and 63.2% of subjects had normal IGF-I levels at a mean dose of 18 mg/day after 5 years of treatment.
Adverse liver effects were rare, with elevated AST or ALT levels (higher than three times the normal upper limit) reported in 30 patients (2.5%) among the 1,178 who had at least one liver enzyme test after starting pegvisomant. However, in all subjects with elevated liver tests and follow-up laboratory results, the increases resolved. There were no reports of liver failure.
The findings, published online ahead of print (J. Clin. Endocrinol. Metab. 2012 [doi:10.1210/jc.2011-2508]), come from ACROSTUDY, a registry of people treated with pegvisomant for acromegaly for a mean of 3.7 years and followed up for an additional 2 years as of December 2009, the cutoff date for the interim analysis. About half (51%) of the subjects were male. Overall, mean age at diagnosis was 42.1 years and mean age at start of treatment was about 50 years. Most subjects (1,131) had been previously treated for their disease, 954 with surgery.
The vast majority of subjects lived in Europe, where pegvisomant is prescribed only for patients in whom other interventions have failed. This means that patients with the worst disease activity and the most comorbidities were likely overincluded, said the study’s authors, led by Dr. A. J. van der Lely of Erasmus University Medical Center in Rotterdam, the Netherlands. Most subjects (n = 1,023) had at least one reported comorbidity before starting pegvisomant, with hypertension and diabetes being the most common.
Dr. van der Lely and his colleagues noted that because the observational trial was conducted in a clinical setting, with fewer clinical visits per year than would be expected in a controlled setting, adverse effects could have been underreported.
The efficacy rate of pegvisomant was seen as lower than reported in earlier clinical trials, with only 63.2% of subjects achieving normal IGF-1 levels on treatment. Dr. van der Lely and his colleagues noted that this, too, could have been related to the administration of pegvisomant in clinical settings, where doses and compliance may have varied. They also noted that stricter criteria were used to assess IGF-1 normalization in this study than in the interventional trials, and that a different assay was used to determine IGF-1 levels. The authors concluded that, "analysis of ACROSTUDY [data] show that pegvisomant can control IGF-1 levels in a majority of patients with acromegaly who do not respond adequately to other therapies."
Dr. van der Lely and six of his coauthors acknowledged receiving ongoing honoraria from Pfizer, the manufacturer of pegvisomant. The remaining five coauthors on the study are Pfizer employees.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
Major Finding: Among the 936 subjects with at least two available MRIs, 3.2% saw MRI-confirmed changes in tumor size during the study period, revealing tumor growth to be uncommon during treatment with pegvisomant, and 63.2% of subjects had normal IGF-I levels at a mean dose of 18 mg/day after 5 years of treatment
Data Source: Data are from 1,288 individuals in an ongoing study who have acromegaly and have been treated with pegvisomant.
Disclosures: Dr. van der Lely and six of his coauthors acknowledged receiving ongoing honoraria from Pfizer, the manufacturer of pegvisomant. The remaining five coauthors on the study are Pfizer employees.
Menstrual Patterns Best Indicator of Aging Path
Changes to the menstrual cycle remain the most important clinical indicators of women's reproductive aging, according to new standardized staging criteria.
The changes identified by the most recent Stages of Reproductive Aging Workshop are now defined in 10 stages, whereas the 2001 STRAW criteria had 7. The new criteria, STRAW-10, offer more detail on the characteristics of flow and cycle length at each stage, along with corresponding endocrine changes. The article was published in Menopause, Climacteric, Fertility and Sterility, and the Journal of Clinical Endocrinology and Metabolism.
The late reproductive stage is now subdivided into two stages, −3b and −3a, instead of only stage −3 as before (Menopause 2012 Feb. 16 [doi:10.1097/gme0b013c31824d8f40]). In stage −3b, flow remains regular, whereas in stage −3a there are subtle changes in flow and length of cycle.
The postmenopausal stage +1, identified in the earlier STRAW criteria, also has been subdivided into three lettered stages, with the endocrine changes and duration of each stage described.
Unlike the previous STRAW criteria, the menstrual changes identified in STRAW-10 are relevant to any healthy woman, regardless of ethnicity, age, body mass index, or lifestyle, the researchers said, noting that although factors such as smoking status and BMI may affect the timing of menopause, the bleeding patterns remain reliable indicators of the reproductive stage.
“Despite the availability of blood tests and sonograms, the menstrual cycle remains the single best way to estimate where a woman is along the reproductive path,” said Dr. Margery Gass, a STRAW-10 coauthor, executive director of the North American Menopause Society (NAMS), and editor of Menopause.
“According to STRAW-10, most women (and their clinicians) can use the changes in their menstrual pattern to determine how close they are to menopause: late reproductive phase (subtle changes in cycle length and blood flow), early menopause transition (menstrual period 7 or more days early or late), and late menopause transition (the occurrence of more than 60 days between cycles),” Dr. Gass said in an interview.
STRAW-10 incorporates three biomarkers, anti-Müllerian hormone (AMH), inhibin B, and antral follicle count, which were not mentioned in the original STRAW criteria, along with follicle-stimulating hormone (FSH), which was included in the original criteria.
In developing the new criteria, an international group of 41 researchers, led by epidemiologist Siobán D. Harlow, Ph.D., of the University of Michigan, Ann Arbor, evaluated data from cohort studies of midlife women with the aim to incorporate the scientific findings of the past decade on ovarian aging and its endocrine and clinical indicators.
Although much has been learned since 2001 with regard to biomarkers, Dr. Harlow and colleagues said that the biomarker criteria outlined in STRAW-10 must still be considered “supportive,” in part because more research is needed and because of the invasiveness and expense of testing.
Dr. Gass said that checking hormone levels is not necessary “except in women who have undergone endometrial ablation or hysterectomy, or who have unusual health circumstances.”
Women for whom the STRAW-10 criteria do not apply include those with polycystic ovarian syndrome or hypothalamic amenorrhea. Women with chronic illnesses such as HIV-AIDS, or those undergoing certain types of cancer treatments, also are difficult to assess under STRAW-10, as cycles and hormone levels can change in response to medication.
The new criteria do not use age to determine reproductive staging, but women younger than 40 years who have premature ovarian insufficiency or premature ovarian failure do not fit well under STRAW-10, the researchers noted, as their course of reproductive aging is more variable.
STRAW-10 meetings were funded by the National Institutes of Health, the Department of Health and Human Services through the National Institute on Aging (NIA) and the Office of Research on Women's Health, NAMS, the American Society for Reproductive Medicine (ASRM), the International Menopause Society in Cape Town, South Africa, and the Endocrine Society.
Dr. Gass receives support from NAMS. Dr. Harlow disclosed ties with the NIA, NAMS, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Other coauthors dislcosed ties with the Endocrine Society, ASRM, the NIA, NAMS, and various pharmaceutical companies.
With STRAW-10, most women can use the changes in their menstrual pattern to find how close they are to menopause.
Source DR. GASS
View on the News
STRAW-10 Will Spur More Research
The main changes [in STRAW-10] have to do with the late reproductive stage and the early postmenopausal stage, which has been subdivided to give more detail, and in turn help understand the physiologic changes which occur at these times. In particular, the late reproductive stage has been subdivided so the measurement of the anti-Müllerian hormone (AMH) and follicle count can be used as a measure of decreased fertility, but the menstrual bleeding is still regular and unchanged. Inhibin B also may be low.
The next stage adds a change in menstrual pattern, usually shorter cycles, and also some variability in FSH levels early in the cycle (days 2-5). Unfortunately, there is no standardization for the AMH assay, so a quantitative measurement is not available.
The bottom line is that these definitions will be helpful mostly for research, but will give us only some guidance to counsel patients about their fertility.
In terms of the late reproductive stages, there now are three subdivisions based on stabilization of key hormones at this time (FSH and estradiol). Perimenopause also is better defined and the addition of symptoms with a description of bleeding patterns will be helpful both to researchers and clinicians. This will lead to more research and understanding of the key physiologic events that lead to reproductive decline and menopausal changes. There also is a description of what happens to reproduction with various dysfunctions, such as chemotherapy and in polycystic ovarian syndrome, which can be helpful both clinically and particularly in research.
Changes to the menstrual cycle remain the most important clinical indicators of women's reproductive aging, according to new standardized staging criteria.
The changes identified by the most recent Stages of Reproductive Aging Workshop are now defined in 10 stages, whereas the 2001 STRAW criteria had 7. The new criteria, STRAW-10, offer more detail on the characteristics of flow and cycle length at each stage, along with corresponding endocrine changes. The article was published in Menopause, Climacteric, Fertility and Sterility, and the Journal of Clinical Endocrinology and Metabolism.
The late reproductive stage is now subdivided into two stages, −3b and −3a, instead of only stage −3 as before (Menopause 2012 Feb. 16 [doi:10.1097/gme0b013c31824d8f40]). In stage −3b, flow remains regular, whereas in stage −3a there are subtle changes in flow and length of cycle.
The postmenopausal stage +1, identified in the earlier STRAW criteria, also has been subdivided into three lettered stages, with the endocrine changes and duration of each stage described.
Unlike the previous STRAW criteria, the menstrual changes identified in STRAW-10 are relevant to any healthy woman, regardless of ethnicity, age, body mass index, or lifestyle, the researchers said, noting that although factors such as smoking status and BMI may affect the timing of menopause, the bleeding patterns remain reliable indicators of the reproductive stage.
“Despite the availability of blood tests and sonograms, the menstrual cycle remains the single best way to estimate where a woman is along the reproductive path,” said Dr. Margery Gass, a STRAW-10 coauthor, executive director of the North American Menopause Society (NAMS), and editor of Menopause.
“According to STRAW-10, most women (and their clinicians) can use the changes in their menstrual pattern to determine how close they are to menopause: late reproductive phase (subtle changes in cycle length and blood flow), early menopause transition (menstrual period 7 or more days early or late), and late menopause transition (the occurrence of more than 60 days between cycles),” Dr. Gass said in an interview.
STRAW-10 incorporates three biomarkers, anti-Müllerian hormone (AMH), inhibin B, and antral follicle count, which were not mentioned in the original STRAW criteria, along with follicle-stimulating hormone (FSH), which was included in the original criteria.
In developing the new criteria, an international group of 41 researchers, led by epidemiologist Siobán D. Harlow, Ph.D., of the University of Michigan, Ann Arbor, evaluated data from cohort studies of midlife women with the aim to incorporate the scientific findings of the past decade on ovarian aging and its endocrine and clinical indicators.
Although much has been learned since 2001 with regard to biomarkers, Dr. Harlow and colleagues said that the biomarker criteria outlined in STRAW-10 must still be considered “supportive,” in part because more research is needed and because of the invasiveness and expense of testing.
Dr. Gass said that checking hormone levels is not necessary “except in women who have undergone endometrial ablation or hysterectomy, or who have unusual health circumstances.”
Women for whom the STRAW-10 criteria do not apply include those with polycystic ovarian syndrome or hypothalamic amenorrhea. Women with chronic illnesses such as HIV-AIDS, or those undergoing certain types of cancer treatments, also are difficult to assess under STRAW-10, as cycles and hormone levels can change in response to medication.
The new criteria do not use age to determine reproductive staging, but women younger than 40 years who have premature ovarian insufficiency or premature ovarian failure do not fit well under STRAW-10, the researchers noted, as their course of reproductive aging is more variable.
STRAW-10 meetings were funded by the National Institutes of Health, the Department of Health and Human Services through the National Institute on Aging (NIA) and the Office of Research on Women's Health, NAMS, the American Society for Reproductive Medicine (ASRM), the International Menopause Society in Cape Town, South Africa, and the Endocrine Society.
Dr. Gass receives support from NAMS. Dr. Harlow disclosed ties with the NIA, NAMS, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Other coauthors dislcosed ties with the Endocrine Society, ASRM, the NIA, NAMS, and various pharmaceutical companies.
With STRAW-10, most women can use the changes in their menstrual pattern to find how close they are to menopause.
Source DR. GASS
View on the News
STRAW-10 Will Spur More Research
The main changes [in STRAW-10] have to do with the late reproductive stage and the early postmenopausal stage, which has been subdivided to give more detail, and in turn help understand the physiologic changes which occur at these times. In particular, the late reproductive stage has been subdivided so the measurement of the anti-Müllerian hormone (AMH) and follicle count can be used as a measure of decreased fertility, but the menstrual bleeding is still regular and unchanged. Inhibin B also may be low.
The next stage adds a change in menstrual pattern, usually shorter cycles, and also some variability in FSH levels early in the cycle (days 2-5). Unfortunately, there is no standardization for the AMH assay, so a quantitative measurement is not available.
The bottom line is that these definitions will be helpful mostly for research, but will give us only some guidance to counsel patients about their fertility.
In terms of the late reproductive stages, there now are three subdivisions based on stabilization of key hormones at this time (FSH and estradiol). Perimenopause also is better defined and the addition of symptoms with a description of bleeding patterns will be helpful both to researchers and clinicians. This will lead to more research and understanding of the key physiologic events that lead to reproductive decline and menopausal changes. There also is a description of what happens to reproduction with various dysfunctions, such as chemotherapy and in polycystic ovarian syndrome, which can be helpful both clinically and particularly in research.
Changes to the menstrual cycle remain the most important clinical indicators of women's reproductive aging, according to new standardized staging criteria.
The changes identified by the most recent Stages of Reproductive Aging Workshop are now defined in 10 stages, whereas the 2001 STRAW criteria had 7. The new criteria, STRAW-10, offer more detail on the characteristics of flow and cycle length at each stage, along with corresponding endocrine changes. The article was published in Menopause, Climacteric, Fertility and Sterility, and the Journal of Clinical Endocrinology and Metabolism.
The late reproductive stage is now subdivided into two stages, −3b and −3a, instead of only stage −3 as before (Menopause 2012 Feb. 16 [doi:10.1097/gme0b013c31824d8f40]). In stage −3b, flow remains regular, whereas in stage −3a there are subtle changes in flow and length of cycle.
The postmenopausal stage +1, identified in the earlier STRAW criteria, also has been subdivided into three lettered stages, with the endocrine changes and duration of each stage described.
Unlike the previous STRAW criteria, the menstrual changes identified in STRAW-10 are relevant to any healthy woman, regardless of ethnicity, age, body mass index, or lifestyle, the researchers said, noting that although factors such as smoking status and BMI may affect the timing of menopause, the bleeding patterns remain reliable indicators of the reproductive stage.
“Despite the availability of blood tests and sonograms, the menstrual cycle remains the single best way to estimate where a woman is along the reproductive path,” said Dr. Margery Gass, a STRAW-10 coauthor, executive director of the North American Menopause Society (NAMS), and editor of Menopause.
“According to STRAW-10, most women (and their clinicians) can use the changes in their menstrual pattern to determine how close they are to menopause: late reproductive phase (subtle changes in cycle length and blood flow), early menopause transition (menstrual period 7 or more days early or late), and late menopause transition (the occurrence of more than 60 days between cycles),” Dr. Gass said in an interview.
STRAW-10 incorporates three biomarkers, anti-Müllerian hormone (AMH), inhibin B, and antral follicle count, which were not mentioned in the original STRAW criteria, along with follicle-stimulating hormone (FSH), which was included in the original criteria.
In developing the new criteria, an international group of 41 researchers, led by epidemiologist Siobán D. Harlow, Ph.D., of the University of Michigan, Ann Arbor, evaluated data from cohort studies of midlife women with the aim to incorporate the scientific findings of the past decade on ovarian aging and its endocrine and clinical indicators.
Although much has been learned since 2001 with regard to biomarkers, Dr. Harlow and colleagues said that the biomarker criteria outlined in STRAW-10 must still be considered “supportive,” in part because more research is needed and because of the invasiveness and expense of testing.
Dr. Gass said that checking hormone levels is not necessary “except in women who have undergone endometrial ablation or hysterectomy, or who have unusual health circumstances.”
Women for whom the STRAW-10 criteria do not apply include those with polycystic ovarian syndrome or hypothalamic amenorrhea. Women with chronic illnesses such as HIV-AIDS, or those undergoing certain types of cancer treatments, also are difficult to assess under STRAW-10, as cycles and hormone levels can change in response to medication.
The new criteria do not use age to determine reproductive staging, but women younger than 40 years who have premature ovarian insufficiency or premature ovarian failure do not fit well under STRAW-10, the researchers noted, as their course of reproductive aging is more variable.
STRAW-10 meetings were funded by the National Institutes of Health, the Department of Health and Human Services through the National Institute on Aging (NIA) and the Office of Research on Women's Health, NAMS, the American Society for Reproductive Medicine (ASRM), the International Menopause Society in Cape Town, South Africa, and the Endocrine Society.
Dr. Gass receives support from NAMS. Dr. Harlow disclosed ties with the NIA, NAMS, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Other coauthors dislcosed ties with the Endocrine Society, ASRM, the NIA, NAMS, and various pharmaceutical companies.
With STRAW-10, most women can use the changes in their menstrual pattern to find how close they are to menopause.
Source DR. GASS
View on the News
STRAW-10 Will Spur More Research
The main changes [in STRAW-10] have to do with the late reproductive stage and the early postmenopausal stage, which has been subdivided to give more detail, and in turn help understand the physiologic changes which occur at these times. In particular, the late reproductive stage has been subdivided so the measurement of the anti-Müllerian hormone (AMH) and follicle count can be used as a measure of decreased fertility, but the menstrual bleeding is still regular and unchanged. Inhibin B also may be low.
The next stage adds a change in menstrual pattern, usually shorter cycles, and also some variability in FSH levels early in the cycle (days 2-5). Unfortunately, there is no standardization for the AMH assay, so a quantitative measurement is not available.
The bottom line is that these definitions will be helpful mostly for research, but will give us only some guidance to counsel patients about their fertility.
In terms of the late reproductive stages, there now are three subdivisions based on stabilization of key hormones at this time (FSH and estradiol). Perimenopause also is better defined and the addition of symptoms with a description of bleeding patterns will be helpful both to researchers and clinicians. This will lead to more research and understanding of the key physiologic events that lead to reproductive decline and menopausal changes. There also is a description of what happens to reproduction with various dysfunctions, such as chemotherapy and in polycystic ovarian syndrome, which can be helpful both clinically and particularly in research.
From Menopause
Cancer Deaths in Europe Still Declining
Cancer deaths in Europe are predicted to continue to decline, with age-standardized rates down 10% in men and 7% in women since 2007.
Mortality from breast cancer – the leading cause of cancer death among women in most European countries – will have declined 9% from 2007, researchers predict, with a rate of 14.9 per 100,000 women in 2012. Among women younger than 50 years of age, the drop will be 13% from 2007, representing 6.3 deaths per 100,000 women.
Overall, some 717,398 men and 565,703 women will die of cancer in Europe in 2012, representing an age-standardized rate of 139 per 100,000 men and 85 per 100,000 women. Significant declines in deaths from stomach, lung, prostate, and colorectal cancers, along with leukemia, have been noted in men since 2007, along with deaths from stomach, uterus, colorectal, and breast cancers, as well as leukemia, in women.
The declines are attributable to a drop in smoking, particularly among men, along with improvements in detection and treatment, investigators said.
Mortality trends, however, are not uniform across European countries, nor across cancer types. Lung cancer deaths were seen as increasing by 7% in women from 2007, with an overall rate of 13.44 deaths per 100,000. Markedly high rates among women in the United Kingdom (21.4 per 100,000) are predicted for 2012, an increase over last year. In the United Kingdom and in Poland, where 16.9 per 100,000 women are predicted to die of it in 2012, lung cancer kills more women annually than breast cancer.
Poland still had high, although declining, mortality from a handful of cancers compared with other European countries, and pancreatic cancer mortality was seen as rising slightly in both sexes in Europe overall. The reasons were unclear to the researchers, particularly since one of the major known risk factors for pancreatic cancer – smoking – has declined. Rates of pancreatic cancer have risen from 7.86 in 2007 to 8.01 per 100,000 in men and from 5.24 to 5.38 per 100,000 in women.
The findings, published online in Annals of Oncology (doi:10.1093/annonc/mds024), update Europe-wide declines first reported a year ago, when members of the same research team, led by epidemiologist Dr. Carlo La Vecchia of the Mario Negri Institute for Pharmacological Research, Milan, used mortality and population data from the World Health Organization to determine that cancer deaths had decreased about 7% in women and 6% in men since 2007 (Ann. Oncol. 2011;22:947-56 [doi:10.1093/annonc/mdq774]).
The researchers used data on cancer deaths in the European Union for the period 1970-2007 to calculate death rates for each year and to identify trends, which were used to predict death rates for 2012.
The breast cancer findings, Dr. La Vecchia and colleagues wrote, show that the mortality declines have occurred despite some unfavorable trends in obesity, physical activity, and menstrual and reproductive factors. Also, the fact that the most dramatic declines have been reported in younger women, who are not routinely screened, suggests that improvements in treatment have played the largest role in reducing deaths.
Despite the pancreatic cancer and lung cancer trends for women, the decline in cancer deaths in Europe may have already surpassed a 2003 goal set by the European Commission to see a 15% reduction by 2015, the researchers said.
The research was funded by the Swiss Cancer League and the Italian Association for Cancer Research. One coauthor, Dr. Matteo Malvezzi, was supported by a fellowship from the Italian Foundation for Cancer Research. None of the authors disclosed conflicts of interest.
Cancer deaths in Europe are predicted to continue to decline, with age-standardized rates down 10% in men and 7% in women since 2007.
Mortality from breast cancer – the leading cause of cancer death among women in most European countries – will have declined 9% from 2007, researchers predict, with a rate of 14.9 per 100,000 women in 2012. Among women younger than 50 years of age, the drop will be 13% from 2007, representing 6.3 deaths per 100,000 women.
Overall, some 717,398 men and 565,703 women will die of cancer in Europe in 2012, representing an age-standardized rate of 139 per 100,000 men and 85 per 100,000 women. Significant declines in deaths from stomach, lung, prostate, and colorectal cancers, along with leukemia, have been noted in men since 2007, along with deaths from stomach, uterus, colorectal, and breast cancers, as well as leukemia, in women.
The declines are attributable to a drop in smoking, particularly among men, along with improvements in detection and treatment, investigators said.
Mortality trends, however, are not uniform across European countries, nor across cancer types. Lung cancer deaths were seen as increasing by 7% in women from 2007, with an overall rate of 13.44 deaths per 100,000. Markedly high rates among women in the United Kingdom (21.4 per 100,000) are predicted for 2012, an increase over last year. In the United Kingdom and in Poland, where 16.9 per 100,000 women are predicted to die of it in 2012, lung cancer kills more women annually than breast cancer.
Poland still had high, although declining, mortality from a handful of cancers compared with other European countries, and pancreatic cancer mortality was seen as rising slightly in both sexes in Europe overall. The reasons were unclear to the researchers, particularly since one of the major known risk factors for pancreatic cancer – smoking – has declined. Rates of pancreatic cancer have risen from 7.86 in 2007 to 8.01 per 100,000 in men and from 5.24 to 5.38 per 100,000 in women.
The findings, published online in Annals of Oncology (doi:10.1093/annonc/mds024), update Europe-wide declines first reported a year ago, when members of the same research team, led by epidemiologist Dr. Carlo La Vecchia of the Mario Negri Institute for Pharmacological Research, Milan, used mortality and population data from the World Health Organization to determine that cancer deaths had decreased about 7% in women and 6% in men since 2007 (Ann. Oncol. 2011;22:947-56 [doi:10.1093/annonc/mdq774]).
The researchers used data on cancer deaths in the European Union for the period 1970-2007 to calculate death rates for each year and to identify trends, which were used to predict death rates for 2012.
The breast cancer findings, Dr. La Vecchia and colleagues wrote, show that the mortality declines have occurred despite some unfavorable trends in obesity, physical activity, and menstrual and reproductive factors. Also, the fact that the most dramatic declines have been reported in younger women, who are not routinely screened, suggests that improvements in treatment have played the largest role in reducing deaths.
Despite the pancreatic cancer and lung cancer trends for women, the decline in cancer deaths in Europe may have already surpassed a 2003 goal set by the European Commission to see a 15% reduction by 2015, the researchers said.
The research was funded by the Swiss Cancer League and the Italian Association for Cancer Research. One coauthor, Dr. Matteo Malvezzi, was supported by a fellowship from the Italian Foundation for Cancer Research. None of the authors disclosed conflicts of interest.
Cancer deaths in Europe are predicted to continue to decline, with age-standardized rates down 10% in men and 7% in women since 2007.
Mortality from breast cancer – the leading cause of cancer death among women in most European countries – will have declined 9% from 2007, researchers predict, with a rate of 14.9 per 100,000 women in 2012. Among women younger than 50 years of age, the drop will be 13% from 2007, representing 6.3 deaths per 100,000 women.
Overall, some 717,398 men and 565,703 women will die of cancer in Europe in 2012, representing an age-standardized rate of 139 per 100,000 men and 85 per 100,000 women. Significant declines in deaths from stomach, lung, prostate, and colorectal cancers, along with leukemia, have been noted in men since 2007, along with deaths from stomach, uterus, colorectal, and breast cancers, as well as leukemia, in women.
The declines are attributable to a drop in smoking, particularly among men, along with improvements in detection and treatment, investigators said.
Mortality trends, however, are not uniform across European countries, nor across cancer types. Lung cancer deaths were seen as increasing by 7% in women from 2007, with an overall rate of 13.44 deaths per 100,000. Markedly high rates among women in the United Kingdom (21.4 per 100,000) are predicted for 2012, an increase over last year. In the United Kingdom and in Poland, where 16.9 per 100,000 women are predicted to die of it in 2012, lung cancer kills more women annually than breast cancer.
Poland still had high, although declining, mortality from a handful of cancers compared with other European countries, and pancreatic cancer mortality was seen as rising slightly in both sexes in Europe overall. The reasons were unclear to the researchers, particularly since one of the major known risk factors for pancreatic cancer – smoking – has declined. Rates of pancreatic cancer have risen from 7.86 in 2007 to 8.01 per 100,000 in men and from 5.24 to 5.38 per 100,000 in women.
The findings, published online in Annals of Oncology (doi:10.1093/annonc/mds024), update Europe-wide declines first reported a year ago, when members of the same research team, led by epidemiologist Dr. Carlo La Vecchia of the Mario Negri Institute for Pharmacological Research, Milan, used mortality and population data from the World Health Organization to determine that cancer deaths had decreased about 7% in women and 6% in men since 2007 (Ann. Oncol. 2011;22:947-56 [doi:10.1093/annonc/mdq774]).
The researchers used data on cancer deaths in the European Union for the period 1970-2007 to calculate death rates for each year and to identify trends, which were used to predict death rates for 2012.
The breast cancer findings, Dr. La Vecchia and colleagues wrote, show that the mortality declines have occurred despite some unfavorable trends in obesity, physical activity, and menstrual and reproductive factors. Also, the fact that the most dramatic declines have been reported in younger women, who are not routinely screened, suggests that improvements in treatment have played the largest role in reducing deaths.
Despite the pancreatic cancer and lung cancer trends for women, the decline in cancer deaths in Europe may have already surpassed a 2003 goal set by the European Commission to see a 15% reduction by 2015, the researchers said.
The research was funded by the Swiss Cancer League and the Italian Association for Cancer Research. One coauthor, Dr. Matteo Malvezzi, was supported by a fellowship from the Italian Foundation for Cancer Research. None of the authors disclosed conflicts of interest.
FROM ANNALS OF ONCOLOGY
Major Finding: Cancer death rates in the European Union are predicted to continue their decline. Rates are expected to drop 10% in men and 7% in women between 2007 and 2012.
Data Source: The researchers used data on cancer deaths in the European Union for the period 1970-2007 to calculate death rates for each year and to identify trends, which were used to predict death rates for 2012.
Disclosures: The research was funded by the Swiss Cancer League and the Italian Association for Cancer Research. One coauthor, Dr. Matteo Malvezzi, was supported by a fellowship from the Italian Foundation for Cancer Research. None of the authors disclosed conflicts of interest.
ACOG Releases Guideline for Breast Cancer Management
The American College of Obstetricians and Gynecologists advises that selective serotonin reuptake inhibitors, or SSRIs, can safely be used to treat hot flashes in some women with breast cancer.
And women who are able to become pregnant following breast cancer treatment can do so without increasing the risk of disease recurrence, according to the college.
The advice, published online Feb. 21 in Obstetrics and Gynecology as Practice Bulletin No. 126, is part of the college’s comprehensive new clinical guideline on the gynecological management of women who are being treated for – or who have been treated for – breast cancer (Obstet. Gynecol. 2012;119:666-82).
The guideline, which cites evidence from 166 published sources, covers such diverse issues as vasomotor symptoms; vaginal atrophy; contraception and fertility; uterine evaluation; and the treatment and prevention of bone loss in the context of current treatment regimens, which may include chemotherapy, hormonal treatments, radiation, and surgery.
"More and more women are living with breast cancer, and breast cancer treatments – particularly the hormonal therapies – have gynecological side effects," Dr. Mindy E. Goldman, lead author of the guideline, said in an interview.
"Ob.gyns. need to be aware of how these drugs work and know about any of the gynecological side effects," said Dr. Goldman, who is director of women’s cancer care for the department of obstetrics, gynecology, and reproductive sciences at the University of California, San Francisco.
For managing vasomotor symptoms such as hot flashes, the guideline recommends that – because hormonal therapy is generally contraindicated in women with hormone-positive breast cancer – an SSRI, an SNRI (serotonin norepinephrine reuptake inhibitor), or gabapentin be used instead. For women on tamoxifen, an SNRI is a better choice than an SSRI, because it avoids a potential interaction.
One SNRI, venlafaxine, was shown in randomized clinical trials to provide significant relief at low doses (75 mg) among women who had been treated for breast cancer. Gabapentin, an anticonvulsant used to control neuropathic pain in breast cancer patients, also helps relieve vasomotor symptoms at low doses, and may improve sleep quality. Pregabalin, a drug in the same class as gabapentin, also was seen as helpful, as was clonidine.
None of these medicines is licensed in the United States for the treatment of hot flashes.
Because chemotherapy, ovarian suppression, and aromatase inhibitors contribute to bone loss and increase fracture risk, the guideline recommends that pharmacologic therapy with bisphosphonates be considered for women who have T scores between –1.5 and –2.0, and be strongly considered for women with T scores less than –2.0, or who have a 10-year risk greater than 20% for a major fracture, or a 10-year hip fracture risk greater than 3%. Zoledronic acid was seen as a strong option among the bisphosphonates, and although raloxifene was generally well tolerated, vasomotor symptoms are among its reported adverse effects.
The guideline also recommends annual monitoring of women whose risks of bone loss significantly change as a result of treatment (for example, premenopausal women being treated with aromatase inhibitors). And vitamin D levels should be checked in women with breast cancer.
Up to 40% of women with breast cancer have severe vaginal dryness, and the topical hormonal creams, suppositories, and vaginal rings commonly used to treat vaginal dryness and atrophy have not been shown to be safe in women with breast cancer. Preference should be given to nonhormonal vaginal moisturizers, with hormonal treatments used on a short-term basis when nonhormonal options have failed. Testosterone supplementation, in patches or creams, remains without enough breast safety data to support it.
Contraceptives that are appropriate for women with breast cancer include barrier methods, the copper intrauterine device, and sterilization. Hormonal methods are contraindicated in women with breast cancer and are considered a risk even for women who have been cancer free for 5 or more years.
One exception may be the levonorgestrel-releasing intrauterine system, in which systemic absorption of levonorgestrel is minimal. However, the system has not been thoroughly studied with regard to long-term breast risk and should be considered only on a case-by-case basis, the guideline states.
Pregnancy following breast cancer treatment has not been shown to increase the risk of recurrence or mortality, according to a recent meta-analysis cited in the guideline (Eur. J. Cancer 2011;47:74-83).
But chemotherapy can compromise fertility, and 5-year use of tamoxifen may diminish a woman’s ovarian reserve before she may safely conceive. Therefore, many women of childbearing age will have difficulty becoming pregnant after treatment, and a fertility consultation at diagnosis is recommended so that advance planning can take place.
In vitro fertilization (IVF) with embryo cryopreservation is seen as a strong option for preserving the potential to have a child; however, there is concern that ovarian stimulation in IVF could cause proliferation of breast cancer cells, leading some practitioners to recommend natural cycle (nonstimulated) IVF.
It is not clear whether ovarian suppression during cancer treatment preserves fertility, according to the guideline. Tamoxifen as an ovarian stimulant has been investigated and has shown promise in treating women whose fertility has been compromised as a result of breast cancer treatment. Although the aromatase inhibitor letrozole cannot be used in premenopausal women as a breast cancer treatment, it may be used in combination with gonadotropins as a fertility agent following treatment.
Finally, routine endometrial biopsy and uterine ultrasonography are not recommended for postmenopausal women taking tamoxifen without evidence of vaginal bleeding, as ultrasound has been associated with a significant false-positive rate leading to unnecessary invasive diagnostic procedures.
However, for women on tamoxifen who experience vaginal bleeding, an endometrial evaluation – including biopsy and follow-up of possible uterine structural anomalies – is essential, the guideline states.
The members of the Committee on Practice Bulletins–Gynecology who wrote the guideline reported no relevant conflicts of interest.
The American College of Obstetricians and Gynecologists advises that selective serotonin reuptake inhibitors, or SSRIs, can safely be used to treat hot flashes in some women with breast cancer.
And women who are able to become pregnant following breast cancer treatment can do so without increasing the risk of disease recurrence, according to the college.
The advice, published online Feb. 21 in Obstetrics and Gynecology as Practice Bulletin No. 126, is part of the college’s comprehensive new clinical guideline on the gynecological management of women who are being treated for – or who have been treated for – breast cancer (Obstet. Gynecol. 2012;119:666-82).
The guideline, which cites evidence from 166 published sources, covers such diverse issues as vasomotor symptoms; vaginal atrophy; contraception and fertility; uterine evaluation; and the treatment and prevention of bone loss in the context of current treatment regimens, which may include chemotherapy, hormonal treatments, radiation, and surgery.
"More and more women are living with breast cancer, and breast cancer treatments – particularly the hormonal therapies – have gynecological side effects," Dr. Mindy E. Goldman, lead author of the guideline, said in an interview.
"Ob.gyns. need to be aware of how these drugs work and know about any of the gynecological side effects," said Dr. Goldman, who is director of women’s cancer care for the department of obstetrics, gynecology, and reproductive sciences at the University of California, San Francisco.
For managing vasomotor symptoms such as hot flashes, the guideline recommends that – because hormonal therapy is generally contraindicated in women with hormone-positive breast cancer – an SSRI, an SNRI (serotonin norepinephrine reuptake inhibitor), or gabapentin be used instead. For women on tamoxifen, an SNRI is a better choice than an SSRI, because it avoids a potential interaction.
One SNRI, venlafaxine, was shown in randomized clinical trials to provide significant relief at low doses (75 mg) among women who had been treated for breast cancer. Gabapentin, an anticonvulsant used to control neuropathic pain in breast cancer patients, also helps relieve vasomotor symptoms at low doses, and may improve sleep quality. Pregabalin, a drug in the same class as gabapentin, also was seen as helpful, as was clonidine.
None of these medicines is licensed in the United States for the treatment of hot flashes.
Because chemotherapy, ovarian suppression, and aromatase inhibitors contribute to bone loss and increase fracture risk, the guideline recommends that pharmacologic therapy with bisphosphonates be considered for women who have T scores between –1.5 and –2.0, and be strongly considered for women with T scores less than –2.0, or who have a 10-year risk greater than 20% for a major fracture, or a 10-year hip fracture risk greater than 3%. Zoledronic acid was seen as a strong option among the bisphosphonates, and although raloxifene was generally well tolerated, vasomotor symptoms are among its reported adverse effects.
The guideline also recommends annual monitoring of women whose risks of bone loss significantly change as a result of treatment (for example, premenopausal women being treated with aromatase inhibitors). And vitamin D levels should be checked in women with breast cancer.
Up to 40% of women with breast cancer have severe vaginal dryness, and the topical hormonal creams, suppositories, and vaginal rings commonly used to treat vaginal dryness and atrophy have not been shown to be safe in women with breast cancer. Preference should be given to nonhormonal vaginal moisturizers, with hormonal treatments used on a short-term basis when nonhormonal options have failed. Testosterone supplementation, in patches or creams, remains without enough breast safety data to support it.
Contraceptives that are appropriate for women with breast cancer include barrier methods, the copper intrauterine device, and sterilization. Hormonal methods are contraindicated in women with breast cancer and are considered a risk even for women who have been cancer free for 5 or more years.
One exception may be the levonorgestrel-releasing intrauterine system, in which systemic absorption of levonorgestrel is minimal. However, the system has not been thoroughly studied with regard to long-term breast risk and should be considered only on a case-by-case basis, the guideline states.
Pregnancy following breast cancer treatment has not been shown to increase the risk of recurrence or mortality, according to a recent meta-analysis cited in the guideline (Eur. J. Cancer 2011;47:74-83).
But chemotherapy can compromise fertility, and 5-year use of tamoxifen may diminish a woman’s ovarian reserve before she may safely conceive. Therefore, many women of childbearing age will have difficulty becoming pregnant after treatment, and a fertility consultation at diagnosis is recommended so that advance planning can take place.
In vitro fertilization (IVF) with embryo cryopreservation is seen as a strong option for preserving the potential to have a child; however, there is concern that ovarian stimulation in IVF could cause proliferation of breast cancer cells, leading some practitioners to recommend natural cycle (nonstimulated) IVF.
It is not clear whether ovarian suppression during cancer treatment preserves fertility, according to the guideline. Tamoxifen as an ovarian stimulant has been investigated and has shown promise in treating women whose fertility has been compromised as a result of breast cancer treatment. Although the aromatase inhibitor letrozole cannot be used in premenopausal women as a breast cancer treatment, it may be used in combination with gonadotropins as a fertility agent following treatment.
Finally, routine endometrial biopsy and uterine ultrasonography are not recommended for postmenopausal women taking tamoxifen without evidence of vaginal bleeding, as ultrasound has been associated with a significant false-positive rate leading to unnecessary invasive diagnostic procedures.
However, for women on tamoxifen who experience vaginal bleeding, an endometrial evaluation – including biopsy and follow-up of possible uterine structural anomalies – is essential, the guideline states.
The members of the Committee on Practice Bulletins–Gynecology who wrote the guideline reported no relevant conflicts of interest.
The American College of Obstetricians and Gynecologists advises that selective serotonin reuptake inhibitors, or SSRIs, can safely be used to treat hot flashes in some women with breast cancer.
And women who are able to become pregnant following breast cancer treatment can do so without increasing the risk of disease recurrence, according to the college.
The advice, published online Feb. 21 in Obstetrics and Gynecology as Practice Bulletin No. 126, is part of the college’s comprehensive new clinical guideline on the gynecological management of women who are being treated for – or who have been treated for – breast cancer (Obstet. Gynecol. 2012;119:666-82).
The guideline, which cites evidence from 166 published sources, covers such diverse issues as vasomotor symptoms; vaginal atrophy; contraception and fertility; uterine evaluation; and the treatment and prevention of bone loss in the context of current treatment regimens, which may include chemotherapy, hormonal treatments, radiation, and surgery.
"More and more women are living with breast cancer, and breast cancer treatments – particularly the hormonal therapies – have gynecological side effects," Dr. Mindy E. Goldman, lead author of the guideline, said in an interview.
"Ob.gyns. need to be aware of how these drugs work and know about any of the gynecological side effects," said Dr. Goldman, who is director of women’s cancer care for the department of obstetrics, gynecology, and reproductive sciences at the University of California, San Francisco.
For managing vasomotor symptoms such as hot flashes, the guideline recommends that – because hormonal therapy is generally contraindicated in women with hormone-positive breast cancer – an SSRI, an SNRI (serotonin norepinephrine reuptake inhibitor), or gabapentin be used instead. For women on tamoxifen, an SNRI is a better choice than an SSRI, because it avoids a potential interaction.
One SNRI, venlafaxine, was shown in randomized clinical trials to provide significant relief at low doses (75 mg) among women who had been treated for breast cancer. Gabapentin, an anticonvulsant used to control neuropathic pain in breast cancer patients, also helps relieve vasomotor symptoms at low doses, and may improve sleep quality. Pregabalin, a drug in the same class as gabapentin, also was seen as helpful, as was clonidine.
None of these medicines is licensed in the United States for the treatment of hot flashes.
Because chemotherapy, ovarian suppression, and aromatase inhibitors contribute to bone loss and increase fracture risk, the guideline recommends that pharmacologic therapy with bisphosphonates be considered for women who have T scores between –1.5 and –2.0, and be strongly considered for women with T scores less than –2.0, or who have a 10-year risk greater than 20% for a major fracture, or a 10-year hip fracture risk greater than 3%. Zoledronic acid was seen as a strong option among the bisphosphonates, and although raloxifene was generally well tolerated, vasomotor symptoms are among its reported adverse effects.
The guideline also recommends annual monitoring of women whose risks of bone loss significantly change as a result of treatment (for example, premenopausal women being treated with aromatase inhibitors). And vitamin D levels should be checked in women with breast cancer.
Up to 40% of women with breast cancer have severe vaginal dryness, and the topical hormonal creams, suppositories, and vaginal rings commonly used to treat vaginal dryness and atrophy have not been shown to be safe in women with breast cancer. Preference should be given to nonhormonal vaginal moisturizers, with hormonal treatments used on a short-term basis when nonhormonal options have failed. Testosterone supplementation, in patches or creams, remains without enough breast safety data to support it.
Contraceptives that are appropriate for women with breast cancer include barrier methods, the copper intrauterine device, and sterilization. Hormonal methods are contraindicated in women with breast cancer and are considered a risk even for women who have been cancer free for 5 or more years.
One exception may be the levonorgestrel-releasing intrauterine system, in which systemic absorption of levonorgestrel is minimal. However, the system has not been thoroughly studied with regard to long-term breast risk and should be considered only on a case-by-case basis, the guideline states.
Pregnancy following breast cancer treatment has not been shown to increase the risk of recurrence or mortality, according to a recent meta-analysis cited in the guideline (Eur. J. Cancer 2011;47:74-83).
But chemotherapy can compromise fertility, and 5-year use of tamoxifen may diminish a woman’s ovarian reserve before she may safely conceive. Therefore, many women of childbearing age will have difficulty becoming pregnant after treatment, and a fertility consultation at diagnosis is recommended so that advance planning can take place.
In vitro fertilization (IVF) with embryo cryopreservation is seen as a strong option for preserving the potential to have a child; however, there is concern that ovarian stimulation in IVF could cause proliferation of breast cancer cells, leading some practitioners to recommend natural cycle (nonstimulated) IVF.
It is not clear whether ovarian suppression during cancer treatment preserves fertility, according to the guideline. Tamoxifen as an ovarian stimulant has been investigated and has shown promise in treating women whose fertility has been compromised as a result of breast cancer treatment. Although the aromatase inhibitor letrozole cannot be used in premenopausal women as a breast cancer treatment, it may be used in combination with gonadotropins as a fertility agent following treatment.
Finally, routine endometrial biopsy and uterine ultrasonography are not recommended for postmenopausal women taking tamoxifen without evidence of vaginal bleeding, as ultrasound has been associated with a significant false-positive rate leading to unnecessary invasive diagnostic procedures.
However, for women on tamoxifen who experience vaginal bleeding, an endometrial evaluation – including biopsy and follow-up of possible uterine structural anomalies – is essential, the guideline states.
The members of the Committee on Practice Bulletins–Gynecology who wrote the guideline reported no relevant conflicts of interest.
FROM OBSTETRICS AND GYNECOLOGY
NICE Decision on Erlotinib to Hinge on Cost Comparison With Gefitinib
The cost-effectiveness agency for England and Wales has issued new draft guidance saying that it is not yet able to recommend erlotinib for the first-line treatment of advanced or metastatic EFGR mutation-positive non–small cell lung cancer.
The National Institute for Health and Clinical Excellence stressed that the draft guidance for erlotinib (Tarceva, Roche) was in early stages, however, and that it had asked its manufacturer for more information. Specifically, NICE said it needed cost-effectiveness comparisons with its currently recommended treatment for the same patient group, gefitinib (Iressa, AstraZeneca and Teva).
Both erlotinib and gefitinib are both oral medications that work by interrupting the signaling of endothelial factor growth receptor.
The Scottish Medicines Agency, which advises the National Health Service in Scotland, in January recommend erlotinib for advanced or metastatic EFGR-positive NSCLC. That recommendation was based on clinical evidence from a phase III open-label trial in European patients (N = 173) with advanced or metastatic NSCLC and activating mutations of EGFR. Patients were randomized to receive either erlotinib or platinum-based chemotherapy (ASCO 2011 Abstract 7503).
At the most recent follow-up evaluated by the Scottish agency, median progression-free survival was 9.7 months in the erlotinib group and 5.2 months in the chemotherapy group (hazard ratio, 0.37 [95% confidence interval: 0.27-0.54]; P less than .0001).
The Scottish reviewers also considered evidence from a Chinese study (N = 155) of similar design comparing erlotinib monotherapy with platinum-based chemotherapy (Lancet Oncology 2011;12:735-42). Median progression-free survival was 13.7 months vs. 4.6 months for erlotinib and gemcitabine/carboplatin (HR, 0.164; P less than .0001).
In a news release, NICE did not mention the clinical evidence supporting erlotinib, saying only that it requires "further sensitivity analyses that look at how the cost-effectiveness of erlotinib changes depending on how many patients are able to continue taking erlotinib and gefitinib after 60 days of treatment." Gefitinib is supplied to the NHS at a fixed cost of £12,200 for each patient who is able to continue treatment for 2 months or more. No costs are incurred for patients who take gefitinib for less than 2 months.
Erlotinib is administered as tablets in a standard dose of 150 mg/day. A pack of 30 tablets currently costs £1,631.53; however, erlotinib’s manufacturer has agreed to a discounting scheme that is currently held in confidence by NICE.
The cost-effectiveness agency for England and Wales has issued new draft guidance saying that it is not yet able to recommend erlotinib for the first-line treatment of advanced or metastatic EFGR mutation-positive non–small cell lung cancer.
The National Institute for Health and Clinical Excellence stressed that the draft guidance for erlotinib (Tarceva, Roche) was in early stages, however, and that it had asked its manufacturer for more information. Specifically, NICE said it needed cost-effectiveness comparisons with its currently recommended treatment for the same patient group, gefitinib (Iressa, AstraZeneca and Teva).
Both erlotinib and gefitinib are both oral medications that work by interrupting the signaling of endothelial factor growth receptor.
The Scottish Medicines Agency, which advises the National Health Service in Scotland, in January recommend erlotinib for advanced or metastatic EFGR-positive NSCLC. That recommendation was based on clinical evidence from a phase III open-label trial in European patients (N = 173) with advanced or metastatic NSCLC and activating mutations of EGFR. Patients were randomized to receive either erlotinib or platinum-based chemotherapy (ASCO 2011 Abstract 7503).
At the most recent follow-up evaluated by the Scottish agency, median progression-free survival was 9.7 months in the erlotinib group and 5.2 months in the chemotherapy group (hazard ratio, 0.37 [95% confidence interval: 0.27-0.54]; P less than .0001).
The Scottish reviewers also considered evidence from a Chinese study (N = 155) of similar design comparing erlotinib monotherapy with platinum-based chemotherapy (Lancet Oncology 2011;12:735-42). Median progression-free survival was 13.7 months vs. 4.6 months for erlotinib and gemcitabine/carboplatin (HR, 0.164; P less than .0001).
In a news release, NICE did not mention the clinical evidence supporting erlotinib, saying only that it requires "further sensitivity analyses that look at how the cost-effectiveness of erlotinib changes depending on how many patients are able to continue taking erlotinib and gefitinib after 60 days of treatment." Gefitinib is supplied to the NHS at a fixed cost of £12,200 for each patient who is able to continue treatment for 2 months or more. No costs are incurred for patients who take gefitinib for less than 2 months.
Erlotinib is administered as tablets in a standard dose of 150 mg/day. A pack of 30 tablets currently costs £1,631.53; however, erlotinib’s manufacturer has agreed to a discounting scheme that is currently held in confidence by NICE.
The cost-effectiveness agency for England and Wales has issued new draft guidance saying that it is not yet able to recommend erlotinib for the first-line treatment of advanced or metastatic EFGR mutation-positive non–small cell lung cancer.
The National Institute for Health and Clinical Excellence stressed that the draft guidance for erlotinib (Tarceva, Roche) was in early stages, however, and that it had asked its manufacturer for more information. Specifically, NICE said it needed cost-effectiveness comparisons with its currently recommended treatment for the same patient group, gefitinib (Iressa, AstraZeneca and Teva).
Both erlotinib and gefitinib are both oral medications that work by interrupting the signaling of endothelial factor growth receptor.
The Scottish Medicines Agency, which advises the National Health Service in Scotland, in January recommend erlotinib for advanced or metastatic EFGR-positive NSCLC. That recommendation was based on clinical evidence from a phase III open-label trial in European patients (N = 173) with advanced or metastatic NSCLC and activating mutations of EGFR. Patients were randomized to receive either erlotinib or platinum-based chemotherapy (ASCO 2011 Abstract 7503).
At the most recent follow-up evaluated by the Scottish agency, median progression-free survival was 9.7 months in the erlotinib group and 5.2 months in the chemotherapy group (hazard ratio, 0.37 [95% confidence interval: 0.27-0.54]; P less than .0001).
The Scottish reviewers also considered evidence from a Chinese study (N = 155) of similar design comparing erlotinib monotherapy with platinum-based chemotherapy (Lancet Oncology 2011;12:735-42). Median progression-free survival was 13.7 months vs. 4.6 months for erlotinib and gemcitabine/carboplatin (HR, 0.164; P less than .0001).
In a news release, NICE did not mention the clinical evidence supporting erlotinib, saying only that it requires "further sensitivity analyses that look at how the cost-effectiveness of erlotinib changes depending on how many patients are able to continue taking erlotinib and gefitinib after 60 days of treatment." Gefitinib is supplied to the NHS at a fixed cost of £12,200 for each patient who is able to continue treatment for 2 months or more. No costs are incurred for patients who take gefitinib for less than 2 months.
Erlotinib is administered as tablets in a standard dose of 150 mg/day. A pack of 30 tablets currently costs £1,631.53; however, erlotinib’s manufacturer has agreed to a discounting scheme that is currently held in confidence by NICE.