User login
Lumbar Fusion With Polyetheretherketone Rods Use for Patients With Degenerative Disease
Surgical treatment of degenerative lumbar spine disease has been rising steadily in the United States, and an increasing fraction of surgery involves lumbar fusion.1,2 Various techniques are used to accomplish a lumbar fusion, including noninstrumented fusion, anterior lumbar interbody fusion (ALIF), lateral lumbar interbody fusion (XLIF, OLIF), posterior pedicle screw fusion, posterior cortical screw fusion, posterior interbody fusion (TLIF, PLIF), and interspinous process fusion. Rigid, metallic fusion hardware provides high stability and fusion rates, but it likely leads to stress shielding and adjacent segment disease.3 There is interest in less rigid and dynamic stabilization techniques to reduce the risk of adjacent segment disease, such as polyetheretherketone (PEEK) rods, which have been available since 2007. However, literature regarding PEEK rod utility is sparse and of mixed outcomes.3,4 Additional patient reported outcome (PRO) information would be useful to both surgeons and patients. Using institutional data, this review was designed to examine our experience with PEEK rod lumbar fusion and to document PROs.
Methods
The study was approved by the institutional review board at the US Department of Veterans Affairs (VA) Portland Health Care System (VAPHCS) in Oregon with a waiver of authorization. In this retrospective, single center study, data were queried from the senior author’s (DAR) case logs from VA Computerized Patient Record System (CPRS). Electronic medical records, imaging, and PROs of all consecutive patients undergoing lumbar fusion at 1 or 2 levels with PEEK rods for degenerative disease were retrospectively reviewed. Cases of trauma, malignancy, or infection were excluded. From March 2011 through October 2019, 108 patients underwent lumbar fusion with PEEK rods.
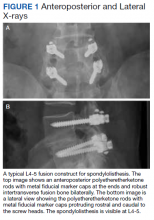
Surgeries were conducted on a Mizuho OSI Jackson Table via bilateral 3 to 4 cm Wiltse incisions using the Medtronic Quadrant retractor system. Medtronic O-Arm images were acquired and delivered to a Medtronic Stealth Station for navigation of the screws. Monopolar coagulation was not used. PEEK pedicle screws were placed and verified with a second O-Arm spin before placing lordotic PEEK rods in the screw heads. No attempt was made to reduce any spondylolisthesis, but distraction was used to open the foramina and indirectly decompress the canal. An interbody device was placed only in treatment of multiply recurrent disc protrusion. After decortication of the transverse processes and facets, intertransverse fusion constructs consisting of calcium hydroxyapatite soaked in autologous bone marrow blood and wrapped in 6-mg bone morphogenetic protein-soaked sponges were placed on the bone. If canal decompression was indicated, a Medtronic Metrx retractor tube was then placed through one of the incisions and decompression carried out. Wounds were closed with absorbable suture. No bracing was used postoperatively. Figure 1 shows a typical single level PEEK rod fusion construct.
Patient pre- and postoperative Short Form-36 (SF-36) physical function (PF) scores and Oswestry Disability Index (ODI) scores had been obtained at routine clinic visits.
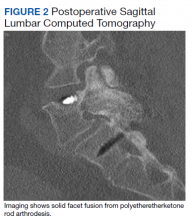
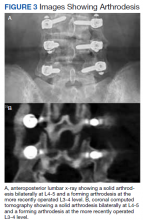
Static radiographs were used to assess the fusion. Dynamic films and/or computed tomography (CT) scans were obtained only when symptomatic pseudarthrosis was suspected. Some patients had abdominal or lumbar CT scans for other indications, and these were reviewed when available. Particular care was taken to assess facet fusion as an indicator of arthrodesis (Figure 2).5
Statistical Analysis
Pre- and postoperative pairwise t tests were completed for patients with a complete data, using SAS 9.2 statistical package. Data are presented as standard deviation (SD) of the mean.
Results
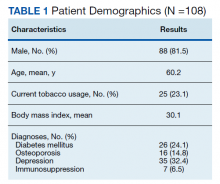
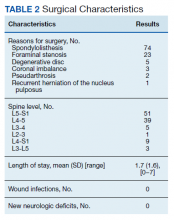
Following application of the inclusion/exclusion criteria, 108 patients had undergone lumbar fusion with PEEK rods. Mean (SD) patient age was 60.2 (10.3) years and 88 patients were male (Table 1). Most surgeries were at L5-S1 and L4-5. There were 97 single-level fusions and 11 bilevel fusions. Seventy-four procedures were for spondylolisthesis, 23 for foraminal stenosis, 5 for degenerative disc disease, 3 for coronal imbalance with foraminal stenosis, 2 for pseudarthrosis after surgery elsewhere, and 1 for multiple recurrent disc herniation (Table 2). Twenty-five patients (23.1%) were current tobacco users and 28 (25.9%) were former smokers, 26 (24.1%) had diabetes mellitus (DM), 16 (14.8%) had low bone density by dual energy X-ray absorptiometry (DEXA) imaging, 35 (32.4%) had depression, and 7 (6.5%) were taking an immunosuppressive agent (chronic steroids, biological response modifiers, or methotrexate). Mean body mass index was 30.1.
Surgical Procedure
Of the 108 patients, the first 18 underwent a procedure with fluoroscopic guidance and the Medtronic FluoroNav and Stealth Systems. The next 90 patients underwent a procedure with O-Arm intraoperative CT scanning and Stealth frameless stereotactic navigation. The mean (SD) length of stay was 1.7 (1.3) days. There were no wound infections and no new neurologic deficits. Mean (SD) follow up time was 30.3 (21.8) months.
Imaging
Final imaging was by radiograph in 73 patients, CT in 31, and magnetic resonance imaging (MRI) in 3 (1 patient had no imaging). Sixty-seven patients (62.0%) had a bilateral arthrodesis, and 15 (13.9%) had at least a unilateral arthrodesis. MRI was not used to assess arthrodesis. Eight patients (7.4%) had no definite arthrodesis. Seventeen patients had inadequate or early imaging from which a fusion determination could not be made. Of 81 patients with > 11 months of follow up, 58 (71.6%) had a bilateral arthrodesis, 12 (14.8%) had a unilateral arthrodesis, 8 (9.9%) had no arthrodesis, and 3 (3.7%) were indeterminate.
No patient had any revision fusion surgery at the index level during follow up. Two patients had adjacent level fusions at 27 and 60 months after the index procedure. One patient had a laminectomy at an adjacent segment at 18 months postfusion, and 1 had a foraminotomy at an adjacent segment 89 months post fusion (Figure 3). Overall, there were 4 (3.7%) adjacent segment surgeries at a mean of 48.5 months after surgery. One patient had a sacro-iliac joint fusion below an L5-S1 fusion 17 months prior for persisting pain after the fusion procedure.
Patient Reported Outcomes
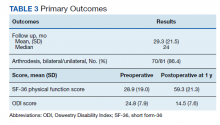
Preoperative SF-36 PF and ODI scores were available for 81 patients (Table 3). Postoperative SF-36 PF scores were obtained at 3 months for 65 of these patients, and at 1 year for 63 patients. Postoperative ODI scores were obtained at 3 months for 65 patients, and at 1 year for 55 patients. Among the 65 patients with completed SF-36 scores at 3 months, a mean increase of 22.4 (95% CI, 17-27; P < .001) was noted, and for the 63 patients at 1 year a mean increase of 30.3 (95% CI, 25-35; P < .001) was noted. Among the 65 patients with completed ODI scores at 3 months, a mean decrease of 6.8 (95% CI, 4.9-8.6; P < .001) was noted, and for the 55 patients with completed ODI scores at 1 year a mean decrease of 10.3 (n = 55; 95% CI, 8.4-12.2; P < .001) was noted.
Cost
We compared the hardware cost of a single level construct consisting of 4 pedicle screws, 4 locking caps, and 2 rods using a PEEK system with that of 2 other titanium construct systems. At VAPHCS, the PEEK system cost was about 71% of the cost of 2 other titanium construct systems and 62% of the cost when compared with Medtronic titanium rods.
Discussion
PEEK is useful for spine and cranial implants. It is inert and fully biocompatible with a modulus of elasticity between that of cortical and cancellous bone, and much lower than that of titanium, and is therefore considered to be semirigid.3,4,6 PEEK rods are intermediate in stiffness between titanium rods (110 Gigapascals) and dynamic devices such as the Zimmer Biomet DYNESYS dynamic stabilization system or the Premia Spine TOPS system.3 Carbon fiber rods and carbon fiber reinforced PEEK implants are other semirigid rod alternatives.7,8 PEEK rods for posterior lumbar fusion surgery were introduced in 2007. Li and colleagues provide a thorough review of the biomechanical properties of PEEK rods.3
PEEK is thought to have several advantages when compared with titanium. These advantages include more physiologic load sharing and reduction in stress shielding, improved durability, reduced risk of failure in osteoporotic bone, less wear debris, no change in bone forming environment, and imaging radiolucency.4,9 Spinal PEEK cages have been reported to allow more uniform radiation dose distribution compared with metal constructs, an advantage that also may pertain to PEEK rods.10 Disadvantages of PEEK rods include an inability to detect rod breakage easily, lack of data on the use in more than minimally unstable clinical situations, and greater expense, although this was not the authors’ observation.3,4,11
Importantly, it has been reported that PEEK rods permit a greater range of motion in all planes when compared with titanium rods.9 Polyetheretherketone rods unload the bone screw interface and increased the anterior column load to a more physiologic 75% when compared with titanium rods.6,9 However, in another biomechanical study that compared titanium rods, PEEK rods, and a dynamic stabilization device, it was reported that anterior load sharing was 55%, 59%, and 75%, respectively.12 This indicated that PEEK rods are closer to metal rods than truly dynamic devices for anterior load sharing. The endurance limit of a PEEK rod construct was similar to that of clinically useful metal systems.9 PEEK rods resulted in no increase in postfatigue motion compared with titanium rods in a biomechanical model.13 Intradiscal pressures at PEEK instrumented segments were similar to uninstrumented segments and greater than those with titanium rod constructs.14 Intradiscal pressures at adjacent segments were highest with dynamic devices, intermediate with semirigid rods, and lowest with rigid constructs; however, stress values at adjacent segments were lower in PEEK than titanium constructs in any direction of motion.15,16
Fusion Rates
The use of PEEK rods in lumbar fusion has been reported previously.3,4,17,18 However, these studies featured small sample sizes, short follow up times, and contradictory results.4 Of 8 outcome reports found in a systematic review, 2 studies reported on procedures designed to create nonfusion outcomes (a third similar trial from 2013 was not included in the systematic review), and 1 study reported only on the condition of PEEK rods removed at subsequent surgery.3,19-21 Reported fusion rates varied from 86 to 100%.
In 42 patients with PEEK rod fusions who were followed for a mean of 31.4 months, 5 patients required adjacent segment surgery and 3 patients were treated for interbody cage migration and nonunion.17 Radiographic fusion rate was 86%. These authors concluded that PEEK rod fusion results were similar to those of other constructs, but not better, or perhaps worse than, metal rods.
Other studies have reported better results with PEEK.11,18,19,22-24 Highsmith and colleagues reported on 3 successful example cases of the use of PEEK rods.11 De Iure and colleagues reported on 30 cases up to 5 levels (mean, 2.9) using autograft bone, with a mean follow up of 18 months.23 Results were reported as satisfactory. Three patients had radiographic nonunions, 1 of which required revision for asymptomatic screw loosening at the cranial end of the construct. Qi and colleagues, reported on 20 patients with PEEK rods compared to 21 patients with titanium alloy rods.24 Both groups had similar clinical outcomes, structural parameters, and 100% fusion rates. Athanasakopoulos and colleagues reported on 52 patients with up to 3 level fusions followed for a mean of 3 years.22 There were significant improvements in PROs: at 1 year 96% had radiographic union. Two patients had screw breakage, 1 of whom required revision to a metal rod construct. Colangeli and colleagues reported on 12 patients treated with PEEK rods compared with 12 who were treated with a dynamic system.18 They reported significant improvements, no complications, and 100% fusion at 6 months. Huang and colleagues reported on 38 patients intended to undergo a nonfusion procedure with 2 years of follow up.19 They reported good outcomes and 1 case of screw loosening. As no fusion was intended, no fusion outcomes were reported. All these studies suggested that longer follow up and more patients would be needed to assess the role of PEEK rods in lumbar fusion.3
Our results show a radiographic fusion rate of 86.4% and a radiographic nonunion rate of 9.9% in patients followed for at least 12 months. There was no clinical need for revision fusion at the index level. In our retrospective review, patients had high levels of smoking, DM, depression, immunosuppression, and obesity, which may negatively influence radiographic fusion rates when compared with other studies with 100% reported fusion rates. There was no instance of construct breakage or screw breakout, indicating that PEEK rods may allow enough flexibility to avoid construct failure under stress as in a fall.
Patient Reported Outcomes
Recent large studies were reviewed to assess the pre- and postoperative patient PROs reported in comparison with our study population (Table 4). In the Swedish Spine Registry analysis of 765 patients with 3 different types of lumbar fusion, the mean preoperative ODI score was 37 and mean SF-36 physical component score (PCS) was 35 for the most similar approach (posterolateral fusion with instrumentation).25 At 1 year postoperation, the mean ODI was 26 and mean SF-36 PCS was 43. In the Spine Patient Outcomes Research Trial (SPORT) spondylolisthesis trial of 3 fusion types, the mean preoperative ODI was 41.2 and mean SF-36 PF score was 31.2 for the most similar approach (posterolateral instrumented fusion with pedicle screws).26 Postoperative ODI scores at 1 year decreased by a mean 20.9 points and mean SF-36 PF scores increased by 29.9.
We report a mean preoperative SF-36 PF score of 28.9, which is lower than the SPORT study score for posterolateral fusion with instrumentation and the Swedish Study score for posterolateral instrumented fusion with pedicle screws. Similarly, our mean ODI score of 24.8 was better than the scores reported in the Swedish and SPORT studies. Our mean SF-36 PF score at 1 year postoperation was 59.3, compared with 58.5 for the SPORT study group and 46.0 in the Swedish study group. Mean ODI score at 1 year postoperatively was 14.5, which is better than the scores reported in the Swedish and SPORT studies.
Minimally clinically important difference (MCID) is a parameter used to gauge the efficacy of spine surgery. The utility of the MCID based upon PROs has been questioned in lumbar fusion surgery, as it has been thought to measure if the patient is “feeling” rather than “doing” better, the latter of which can be better measured by functional performance measures and objective, external socioeconomic anchors such as return to work and health care costs.27 Nevertheless, validated PROs are reported widely in the spine surgery literature. The MCID in the SF-36 is not well established and can depend upon whether the scores are at the extremes or more in the central range and whether there is large variability in the scores.28 Rheumatoid arthritis was estimated to be 7.1 points on the PF scale and 7.2 on the physical component summary (PCS).29 For total knee replacement, it has been estimated to be 10 points on the SF-36 PCS.30 Lumbar surgery was estimated to be 4.9 points for the SF-36 PCS and 12.8 points for the ODI.31 And the SPORT trial it has been estimated that a 30% change in the possible gain (or loss) may be an appropriate criterion.28
With a preoperative mean SF-36 PF of 28.9, a 30% improvement in the available range (70.1) would be 21 points, making our data mean improvement of 30 points above the MCID. With a mean preoperative ODI of 24.6, a 30% improvement in the available range (25.4) would be 7.6 points, making our data mean improvement of 10.3 points better than the MCID. Therefore, our outcome results are comparable with other lumbar fusion outcome studies in terms of degree of disability prior to surgery and amount of improvement from surgery.
Adjacent Segment Disease
The precise factors resulting in adjacent segment disease are not fully defined.3,32 In reviews of lumbar adjacent segment disease, reported rates ranged from 2.5% at 1 year up to 80 to 100% at 10 years, with lower rates with noninstrumented fusions.4,32-34 Annual incidence of symptomatic adjacent segment disease following lumbar fusion ranges from 0.6 to 3.9% per year.32,35,36 Mismatch between lumbar lordosis and pelvic incidence after fusion is thought to lead to higher rates of adjacent segment disease, as can a laminectomy at an adjacent segment.32,36 Percutaneous fusion techniques or use of the Wiltse approach may lower the risk of adjacent segment disease due to avoidance of facet capsule disruption.37,38
Dynamic stabilization techniques do not appear be clearly protective against adjacent segment disease, although biomechanical models suggest that they may do so.33,39,40 A review by Wang and colleagues pooled studies to assess the risk of lumbar adjacent segment disease in spinal fusion to compare to disc arthroplasty and concluded that fusion carried a higher risk of adjacent segment disease.41 Definitive data on other types of motion preservation devices is lacking.3We show 3 adjacent segment fusions and 1 laminectomy have been needed in 108 patients and at a mean of 46 months after the index procedure and over 2.5 years of mean overall follow up. This is a low adjacent segment surgery rate compared to the historical data cited above, and may suggest some advantage for PEEK rods over more rigid constructs.
Strengths and Limitations
Strengths of this study include larger numbers than prior series of PEEK rod use and use in a population with high comorbidities linked to poor results without reduction in good outcomes. PEEK rods as used at the VAPHCS do not result in higher instrumentation costs than all metal constructs.
Study limitations include the retrospective nature with loss of follow up on some patients and incomplete radiographic and PROs in some patients. The use of 100% stereotactic guidance, the avoidance of interbody devices, and the off-label use of bone morphogenetic protein as part of the fusion construct introduce additional variables that may influence comparison to other studies. To avoid unnecessary radiation exposure, flexion extension films or CT scans were not routinely obtained if patients were doing well.42 Additionally, the degree of motion on dynamic views that would differentiate pseudarthrosis from arthrodesis has not been defined.5
Conclusions
The results presented show that lumbar fusion with PEEK rods can be undertaken with short hospitalization times and low complication rates, produce satisfactory clinical improvements, and result in radiographic fusion rates similar to metal constructs. Low rates of hardware failure or need for revision surgery were found. Preliminarily results of low rates of adjacent segment surgery are comparable with previously published metal construct rates. Longer follow up is needed to confirm these findings and to investigate whether semirigid constructs truly offer some protection from adjacent segment disease when compared to all metal constructs.
Acknowledgments
The authors thank Shirley McCartney, PhD, for editorial assistance.
1. Deyo RA, Mirza SK, Martin BI, Kreuter W, Goodman DC, Jarvik JG. Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA. 2010;303(13):1259-1265. doi:10.1001/jama.2010.338
2. Machado GC, Maher CG, Ferreira PH, et al. Trends, complications, and costs for hospital admission and surgery for lumbar spinal stenosis. Spine (Phila Pa 1976). 2017;42(22):1737-1743. doi:10.1097/BRS.0000000000002207
3. Li C, Liu L, Shi JY, Yan KZ, Shen WZ, Yang ZR. Clinical and biomechanical researches of polyetheretherketone (PEEK) rods for semi-rigid lumbar fusion: a systematic review. Neurosurg Rev. 2018;41(2):375-389. doi:10.1007/s10143-016-0763-2
4. Mavrogenis AF, Vottis C, Triantafyllopoulos G, Papagelopoulos PJ, Pneumaticos SG. PEEK rod systems for the spine. Eur J Orthop Surg Traumatol. 2014;24 Suppl 1:S111-S116. doi:10.1007/s00590-014-1421-4
5. Choudhri TF, Mummaneni PV, Dhall SS, et al. Guideline update for the performance of fusion procedures for degenerative disease of the lumbar spine. Part 4: radiographic assessment of fusion status. J Neurosurg Spine. 2014;21(1):23-30. doi:10.3171/2014.4.SPINE14267
6. Ahn YH, Chen WM, Lee KY, Park KW, Lee SJ. Comparison of the load-sharing characteristics between pedicle-based dynamic and rigid rod devices. Biomed Mater. 2008;3(4):044101. doi:10.1088/1748-6041/3/4/044101
7. Ozer AF, Cevik OM, Erbulut DU, et al. A novel modular dynamic stabilization system for the treatment of degenerative spinal pathologies. Turk Neurosurg. 2019;29(1):115-120. doi:10.5137/1019-5149.JTN.23227-18.1
8. Hak DJ, Mauffrey C, Seligson D, Lindeque B. Use of carbon-fiber-reinforced composite implants in orthopedic surgery. Orthopedics. 2014;37(12):825-830. doi:10.3928/01477447-20141124-05
9. Gornet MF, Chan FW, Coleman JC, et al. Biomechanical assessment of a PEEK rod system for semi-rigid fixation of lumbar fusion constructs. J Biomech Eng. 2011;133(8):081009. doi:10.1115/1.4004862
10. Jackson JB 3rd, Crimaldi AJ, Peindl R, Norton HJ, Anderson WE, Patt JC. Effect of polyether ether ketone on therapeutic radiation to the spine: a pilot study. Spine (Phila Pa 1976). 2017;42(1):E1-E7. doi:10.1097/BRS.0000000000001695
11. Highsmith JM, Tumialán LM, Rodts GE Jr. Flexible rods and the case for dynamic stabilization. Neurosurg Focus. 2007;22(1):E11. Published 2007 Jan 15. doi:10.3171/foc.2007.22.1.11
12. Sengupta DK, Bucklen B, McAfee PC, Nichols J, Angara R, Khalil S. The comprehensive biomechanics and load-sharing of semirigid PEEK and semirigid posterior dynamic stabilization systems. Adv Orthop. 2013;2013:745610. doi:10.1155/2013/745610
13. Agarwal A, Ingels M, Kodigudla M, Momeni N, Goel V, Agarwal AK. Adjacent-level hypermobility and instrumented-level fatigue loosening with titanium and PEEK rods for a pedicle screw system: an in vitro study. J Biomech Eng. 2016;138(5):051004. doi:10.1115/1.4032965
14. Chou WK, Chien A, Wang JL. Biomechanical analysis between PEEK and titanium screw-rods spinal construct subjected to fatigue loading. J Spinal Disord Tech. 2015;28(3):E121-E125. doi:10.1097/BSD.0000000000000176
15. Shih KS Hsu CC, Zhou SY, Hou SM. Biomechanical investigation of pedicle screw-based posterior stabilization systems for the treatment of lumbar degenerative disc disease using finite element analyses. Biomed Eng: Appl Basis Commun. 2015;27(06):1550060. doi: 10.4015/S101623721550060X

16. Chang TK, Huang CH, Liu YC, et al. Biomechanical evaluation and comparison of polyetheretherketone rod system to traditional titanium rod fixation on adjacent levels. Formosan J Musculoskeletal Disord. 2013;4(2):42-47. doi: 10.1016/j.fjmd.2013.04.003
17. Ormond DR, Albert L Jr, Das K. Polyetheretherketone (PEEK) rods in lumbar spine degenerative disease: a case series. Clin Spine Surg. 2016;29(7):E371-E375. doi:10.1097/BSD.0b013e318277cb9b
18. Colangeli S, Barbanti Brodàno G, Gasbarrini A, et al. Polyetheretherketone (PEEK) rods: short-term results in lumbar spine degenerative disease. J Neurosurg Sci. 2015;59(2):91-96.
19. Huang W, Chang Z, Song R, Zhou K, Yu X. Non-fusion procedure using PEEK rod systems for lumbar degenerative diseases: clinical experience with a 2-year follow-up. BMC Musculoskelet Disord. 2016;17:53. Published 2016 Feb 1. doi:10.1186/s12891-016-0913-2
20. Wang C-J, Graf H, Wei H-W. Clinical outcomes of the dynamic lumbar pedicle screw-rod stabilization. Neurosurg Q. 2016;26(3):214-218. doi:10.1097/WNQ.0000000000000169
21. Kurtz SM, Lanman TH, Higgs G, et al. Retrieval analysis of PEEK rods for posterior fusion and motion preservation. Eur Spine J. 2013;22(12):2752-2759. doi:10.1007/s00586-013-2920-4
22. Athanasakopoulos M, Mavrogenis AF, Triantafyllopoulos G, Koufos S, Pneumaticos SG. Posterior spinal fusion using pedicle screws. Orthopedics. 2013;36(7):e951-e957. doi:10.3928/01477447-20130624-28
23. De Iure F, Bosco G, Cappuccio M, Paderni S, Amendola L. Posterior lumbar fusion by peek rods in degenerative spine: preliminary report on 30 cases. Eur Spine J. 2012;21 Suppl 1(Suppl 1):S50-S54. doi:10.1007/s00586-012-2219-x
24. Qi L, Li M, Zhang S, Xue J, Si H. Comparative effectiveness of PEEK rods versus titanium alloy rods in lumbar fusion: a preliminary report. Acta Neurochir (Wien). 2013;155(7):1187-1193. doi:10.1007/s00701-013-1772-3
25. Endler P, Ekman P, Möller H, Gerdhem P. Outcomes of posterolateral fusion with and without instrumentation and of interbody fusion for isthmic spondylolisthesis: a prospective study. J Bone Joint Surg Am. 2017;99(9):743-752. doi:10.2106/JBJS.16.00679
26. Abdu WA, Lurie JD, Spratt KF, et al. Degenerative spondylolisthesis: does fusion method influence outcome? Four-year results of the spine patient outcomes research trial. Spine (Phila Pa 1976). 2009;34(21):2351-2360. doi:10.1097/BRS.0b013e3181b8a829
27. Gatchel RJ, Mayer TG, Chou R. What does/should the minimum clinically important difference measure? A reconsideration of its clinical value in evaluating efficacy of lumbar fusion surgery. Clin J Pain. 2012;28(5):387-397. doi:10.1097/AJP.0b013e3182327f20
28. Spratt KF. Patient-level minimal clinically important difference based on clinical judgment and minimally detectable measurement difference: a rationale for the SF-36 physical function scale in the SPORT intervertebral disc herniation cohort. Spine (Phila Pa 1976). 2009;34(16):1722-1731. doi:10.1097/BRS.0b013e3181a8faf2
29. Ward MM, Guthrie LC, Alba MI. Clinically important changes in short form 36 health survey scales for use in rheumatoid arthritis clinical trials: the impact of low responsiveness. Arthritis Care Res (Hoboken). 2014;66(12):1783-1789. doi:10.1002/acr.22392
30. Escobar A, Quintana JM, Bilbao A, Aróstegui I, Lafuente I, Vidaurreta I. Responsiveness and clinically important differences for the WOMAC and SF-36 after total knee replacement. Osteoarthritis Cartilage. 2007;15(3):273-280. doi:10.1016/j.joca.2006.09.001
31. Copay AG, Glassman SD, Subach BR, Berven S, Schuler TC, Carreon LY. Minimum clinically important difference in lumbar spine surgery patients: a choice of methods using the Oswestry Disability Index, Medical Outcomes Study questionnaire Short Form 36, and pain scales. Spine J. 2008;8(6):968-974. doi:10.1016/j.spinee.2007.11.006
32. Radcliff KE, Kepler CK, Jakoi A, et al. Adjacent segment disease in the lumbar spine following different treatment interventions. Spine J. 2013;13(10):1339-1349. doi:10.1016/j.spinee.2013.03.020
33. Epstein NE. Adjacent level disease following lumbar spine surgery: a review. Surg Neurol Int. 2015;6(Suppl 24):S591-S599. Published 2015 Nov 25. doi:10.4103/2152-7806.170432
34. Epstein NE. A review: reduced reoperation rate for multilevel lumbar laminectomies with noninstrumented versus instrumented fusions. Surg Neurol Int. 2016;7(Suppl 13):S337-S346. Published 2016 May 17. doi:10.4103/2152-7806.182546
35. Scemama C, Magrino B, Gillet P, Guigui P. Risk of adjacent-segment disease requiring surgery after short lumbar fusion: results of the French Spine Surgery Society Series. J Neurosurg Spine. 2016;25(1):46-51. doi:10.3171/2015.11.SPINE15700
36. Tempel ZJ, Gandhoke GS, Bolinger BD, et al. The influence of pelvic incidence and lumbar lordosis mismatch on development of symptomatic adjacent level disease following single-level transforaminal lumbar interbody fusion. Neurosurgery. 2017;80(6):880-886. doi:10.1093/neuros/nyw073

37. Cheng YW, Chang PY, Wu JC, et al. Letter to the editor: Pedicle screw-based dynamic stabilization and adjacent-segment disease. J Neurosurg Spine. 2017;26(3):405-406. doi:10.3171/2016.7.SPINE16816
38. Street JT, Andrew Glennie R, Dea N, et al. A comparison of the Wiltse versus midline approaches in degenerative conditions of the lumbar spine. J Neurosurg Spine. 2016;25(3):332-338. doi:10.3171/2016.2.SPINE151018
39. Kuo CH, Huang WC, Wu JC, et al. Radiological adjacent-segment degeneration in L4-5 spondylolisthesis: comparison between dynamic stabilization and minimally invasive transforaminal lumbar interbody fusion. J Neurosurg Spine. 2018;29(3):250-258. doi:10.3171/2018.1.SPINE17993
40. Lee CH, Kim YE, Lee HJ, Kim DG, Kim CH. Biomechanical effects of hybrid stabilization on the risk of proximal adjacent-segment degeneration following lumbar spinal fusion using an interspinous device or a pedicle screw-based dynamic fixator. J Neurosurg Spine. 2017;27(6):643-649. doi:10.3171/2017.3.SPINE161169
41. Wang JC, Arnold PM, Hermsmeyer JT, Norvell DC. Do lumbar motion preserving devices reduce the risk of adjacent segment pathology compared with fusion surgery? A systematic review. Spine (Phila Pa 1976). 2012;37(22 Suppl):S133-S143. doi:10.1097/BRS.0b013e31826cadf2
42. Ross DA. Letter to the editor: steroid use in anterior cervical discectomy and fusion. J Neurosurg Spine. 2016;24(6):998-1000. doi:10.3171/2015.9.SPINE151052
Surgical treatment of degenerative lumbar spine disease has been rising steadily in the United States, and an increasing fraction of surgery involves lumbar fusion.1,2 Various techniques are used to accomplish a lumbar fusion, including noninstrumented fusion, anterior lumbar interbody fusion (ALIF), lateral lumbar interbody fusion (XLIF, OLIF), posterior pedicle screw fusion, posterior cortical screw fusion, posterior interbody fusion (TLIF, PLIF), and interspinous process fusion. Rigid, metallic fusion hardware provides high stability and fusion rates, but it likely leads to stress shielding and adjacent segment disease.3 There is interest in less rigid and dynamic stabilization techniques to reduce the risk of adjacent segment disease, such as polyetheretherketone (PEEK) rods, which have been available since 2007. However, literature regarding PEEK rod utility is sparse and of mixed outcomes.3,4 Additional patient reported outcome (PRO) information would be useful to both surgeons and patients. Using institutional data, this review was designed to examine our experience with PEEK rod lumbar fusion and to document PROs.
Methods
The study was approved by the institutional review board at the US Department of Veterans Affairs (VA) Portland Health Care System (VAPHCS) in Oregon with a waiver of authorization. In this retrospective, single center study, data were queried from the senior author’s (DAR) case logs from VA Computerized Patient Record System (CPRS). Electronic medical records, imaging, and PROs of all consecutive patients undergoing lumbar fusion at 1 or 2 levels with PEEK rods for degenerative disease were retrospectively reviewed. Cases of trauma, malignancy, or infection were excluded. From March 2011 through October 2019, 108 patients underwent lumbar fusion with PEEK rods.
Surgeries were conducted on a Mizuho OSI Jackson Table via bilateral 3 to 4 cm Wiltse incisions using the Medtronic Quadrant retractor system. Medtronic O-Arm images were acquired and delivered to a Medtronic Stealth Station for navigation of the screws. Monopolar coagulation was not used. PEEK pedicle screws were placed and verified with a second O-Arm spin before placing lordotic PEEK rods in the screw heads. No attempt was made to reduce any spondylolisthesis, but distraction was used to open the foramina and indirectly decompress the canal. An interbody device was placed only in treatment of multiply recurrent disc protrusion. After decortication of the transverse processes and facets, intertransverse fusion constructs consisting of calcium hydroxyapatite soaked in autologous bone marrow blood and wrapped in 6-mg bone morphogenetic protein-soaked sponges were placed on the bone. If canal decompression was indicated, a Medtronic Metrx retractor tube was then placed through one of the incisions and decompression carried out. Wounds were closed with absorbable suture. No bracing was used postoperatively. Figure 1 shows a typical single level PEEK rod fusion construct.
Patient pre- and postoperative Short Form-36 (SF-36) physical function (PF) scores and Oswestry Disability Index (ODI) scores had been obtained at routine clinic visits.
Static radiographs were used to assess the fusion. Dynamic films and/or computed tomography (CT) scans were obtained only when symptomatic pseudarthrosis was suspected. Some patients had abdominal or lumbar CT scans for other indications, and these were reviewed when available. Particular care was taken to assess facet fusion as an indicator of arthrodesis (Figure 2).5
Statistical Analysis
Pre- and postoperative pairwise t tests were completed for patients with a complete data, using SAS 9.2 statistical package. Data are presented as standard deviation (SD) of the mean.
Results
Following application of the inclusion/exclusion criteria, 108 patients had undergone lumbar fusion with PEEK rods. Mean (SD) patient age was 60.2 (10.3) years and 88 patients were male (Table 1). Most surgeries were at L5-S1 and L4-5. There were 97 single-level fusions and 11 bilevel fusions. Seventy-four procedures were for spondylolisthesis, 23 for foraminal stenosis, 5 for degenerative disc disease, 3 for coronal imbalance with foraminal stenosis, 2 for pseudarthrosis after surgery elsewhere, and 1 for multiple recurrent disc herniation (Table 2). Twenty-five patients (23.1%) were current tobacco users and 28 (25.9%) were former smokers, 26 (24.1%) had diabetes mellitus (DM), 16 (14.8%) had low bone density by dual energy X-ray absorptiometry (DEXA) imaging, 35 (32.4%) had depression, and 7 (6.5%) were taking an immunosuppressive agent (chronic steroids, biological response modifiers, or methotrexate). Mean body mass index was 30.1.
Surgical Procedure
Of the 108 patients, the first 18 underwent a procedure with fluoroscopic guidance and the Medtronic FluoroNav and Stealth Systems. The next 90 patients underwent a procedure with O-Arm intraoperative CT scanning and Stealth frameless stereotactic navigation. The mean (SD) length of stay was 1.7 (1.3) days. There were no wound infections and no new neurologic deficits. Mean (SD) follow up time was 30.3 (21.8) months.
Imaging
Final imaging was by radiograph in 73 patients, CT in 31, and magnetic resonance imaging (MRI) in 3 (1 patient had no imaging). Sixty-seven patients (62.0%) had a bilateral arthrodesis, and 15 (13.9%) had at least a unilateral arthrodesis. MRI was not used to assess arthrodesis. Eight patients (7.4%) had no definite arthrodesis. Seventeen patients had inadequate or early imaging from which a fusion determination could not be made. Of 81 patients with > 11 months of follow up, 58 (71.6%) had a bilateral arthrodesis, 12 (14.8%) had a unilateral arthrodesis, 8 (9.9%) had no arthrodesis, and 3 (3.7%) were indeterminate.
No patient had any revision fusion surgery at the index level during follow up. Two patients had adjacent level fusions at 27 and 60 months after the index procedure. One patient had a laminectomy at an adjacent segment at 18 months postfusion, and 1 had a foraminotomy at an adjacent segment 89 months post fusion (Figure 3). Overall, there were 4 (3.7%) adjacent segment surgeries at a mean of 48.5 months after surgery. One patient had a sacro-iliac joint fusion below an L5-S1 fusion 17 months prior for persisting pain after the fusion procedure.
Patient Reported Outcomes
Preoperative SF-36 PF and ODI scores were available for 81 patients (Table 3). Postoperative SF-36 PF scores were obtained at 3 months for 65 of these patients, and at 1 year for 63 patients. Postoperative ODI scores were obtained at 3 months for 65 patients, and at 1 year for 55 patients. Among the 65 patients with completed SF-36 scores at 3 months, a mean increase of 22.4 (95% CI, 17-27; P < .001) was noted, and for the 63 patients at 1 year a mean increase of 30.3 (95% CI, 25-35; P < .001) was noted. Among the 65 patients with completed ODI scores at 3 months, a mean decrease of 6.8 (95% CI, 4.9-8.6; P < .001) was noted, and for the 55 patients with completed ODI scores at 1 year a mean decrease of 10.3 (n = 55; 95% CI, 8.4-12.2; P < .001) was noted.
Cost
We compared the hardware cost of a single level construct consisting of 4 pedicle screws, 4 locking caps, and 2 rods using a PEEK system with that of 2 other titanium construct systems. At VAPHCS, the PEEK system cost was about 71% of the cost of 2 other titanium construct systems and 62% of the cost when compared with Medtronic titanium rods.
Discussion
PEEK is useful for spine and cranial implants. It is inert and fully biocompatible with a modulus of elasticity between that of cortical and cancellous bone, and much lower than that of titanium, and is therefore considered to be semirigid.3,4,6 PEEK rods are intermediate in stiffness between titanium rods (110 Gigapascals) and dynamic devices such as the Zimmer Biomet DYNESYS dynamic stabilization system or the Premia Spine TOPS system.3 Carbon fiber rods and carbon fiber reinforced PEEK implants are other semirigid rod alternatives.7,8 PEEK rods for posterior lumbar fusion surgery were introduced in 2007. Li and colleagues provide a thorough review of the biomechanical properties of PEEK rods.3
PEEK is thought to have several advantages when compared with titanium. These advantages include more physiologic load sharing and reduction in stress shielding, improved durability, reduced risk of failure in osteoporotic bone, less wear debris, no change in bone forming environment, and imaging radiolucency.4,9 Spinal PEEK cages have been reported to allow more uniform radiation dose distribution compared with metal constructs, an advantage that also may pertain to PEEK rods.10 Disadvantages of PEEK rods include an inability to detect rod breakage easily, lack of data on the use in more than minimally unstable clinical situations, and greater expense, although this was not the authors’ observation.3,4,11
Importantly, it has been reported that PEEK rods permit a greater range of motion in all planes when compared with titanium rods.9 Polyetheretherketone rods unload the bone screw interface and increased the anterior column load to a more physiologic 75% when compared with titanium rods.6,9 However, in another biomechanical study that compared titanium rods, PEEK rods, and a dynamic stabilization device, it was reported that anterior load sharing was 55%, 59%, and 75%, respectively.12 This indicated that PEEK rods are closer to metal rods than truly dynamic devices for anterior load sharing. The endurance limit of a PEEK rod construct was similar to that of clinically useful metal systems.9 PEEK rods resulted in no increase in postfatigue motion compared with titanium rods in a biomechanical model.13 Intradiscal pressures at PEEK instrumented segments were similar to uninstrumented segments and greater than those with titanium rod constructs.14 Intradiscal pressures at adjacent segments were highest with dynamic devices, intermediate with semirigid rods, and lowest with rigid constructs; however, stress values at adjacent segments were lower in PEEK than titanium constructs in any direction of motion.15,16
Fusion Rates
The use of PEEK rods in lumbar fusion has been reported previously.3,4,17,18 However, these studies featured small sample sizes, short follow up times, and contradictory results.4 Of 8 outcome reports found in a systematic review, 2 studies reported on procedures designed to create nonfusion outcomes (a third similar trial from 2013 was not included in the systematic review), and 1 study reported only on the condition of PEEK rods removed at subsequent surgery.3,19-21 Reported fusion rates varied from 86 to 100%.
In 42 patients with PEEK rod fusions who were followed for a mean of 31.4 months, 5 patients required adjacent segment surgery and 3 patients were treated for interbody cage migration and nonunion.17 Radiographic fusion rate was 86%. These authors concluded that PEEK rod fusion results were similar to those of other constructs, but not better, or perhaps worse than, metal rods.
Other studies have reported better results with PEEK.11,18,19,22-24 Highsmith and colleagues reported on 3 successful example cases of the use of PEEK rods.11 De Iure and colleagues reported on 30 cases up to 5 levels (mean, 2.9) using autograft bone, with a mean follow up of 18 months.23 Results were reported as satisfactory. Three patients had radiographic nonunions, 1 of which required revision for asymptomatic screw loosening at the cranial end of the construct. Qi and colleagues, reported on 20 patients with PEEK rods compared to 21 patients with titanium alloy rods.24 Both groups had similar clinical outcomes, structural parameters, and 100% fusion rates. Athanasakopoulos and colleagues reported on 52 patients with up to 3 level fusions followed for a mean of 3 years.22 There were significant improvements in PROs: at 1 year 96% had radiographic union. Two patients had screw breakage, 1 of whom required revision to a metal rod construct. Colangeli and colleagues reported on 12 patients treated with PEEK rods compared with 12 who were treated with a dynamic system.18 They reported significant improvements, no complications, and 100% fusion at 6 months. Huang and colleagues reported on 38 patients intended to undergo a nonfusion procedure with 2 years of follow up.19 They reported good outcomes and 1 case of screw loosening. As no fusion was intended, no fusion outcomes were reported. All these studies suggested that longer follow up and more patients would be needed to assess the role of PEEK rods in lumbar fusion.3
Our results show a radiographic fusion rate of 86.4% and a radiographic nonunion rate of 9.9% in patients followed for at least 12 months. There was no clinical need for revision fusion at the index level. In our retrospective review, patients had high levels of smoking, DM, depression, immunosuppression, and obesity, which may negatively influence radiographic fusion rates when compared with other studies with 100% reported fusion rates. There was no instance of construct breakage or screw breakout, indicating that PEEK rods may allow enough flexibility to avoid construct failure under stress as in a fall.
Patient Reported Outcomes
Recent large studies were reviewed to assess the pre- and postoperative patient PROs reported in comparison with our study population (Table 4). In the Swedish Spine Registry analysis of 765 patients with 3 different types of lumbar fusion, the mean preoperative ODI score was 37 and mean SF-36 physical component score (PCS) was 35 for the most similar approach (posterolateral fusion with instrumentation).25 At 1 year postoperation, the mean ODI was 26 and mean SF-36 PCS was 43. In the Spine Patient Outcomes Research Trial (SPORT) spondylolisthesis trial of 3 fusion types, the mean preoperative ODI was 41.2 and mean SF-36 PF score was 31.2 for the most similar approach (posterolateral instrumented fusion with pedicle screws).26 Postoperative ODI scores at 1 year decreased by a mean 20.9 points and mean SF-36 PF scores increased by 29.9.
We report a mean preoperative SF-36 PF score of 28.9, which is lower than the SPORT study score for posterolateral fusion with instrumentation and the Swedish Study score for posterolateral instrumented fusion with pedicle screws. Similarly, our mean ODI score of 24.8 was better than the scores reported in the Swedish and SPORT studies. Our mean SF-36 PF score at 1 year postoperation was 59.3, compared with 58.5 for the SPORT study group and 46.0 in the Swedish study group. Mean ODI score at 1 year postoperatively was 14.5, which is better than the scores reported in the Swedish and SPORT studies.
Minimally clinically important difference (MCID) is a parameter used to gauge the efficacy of spine surgery. The utility of the MCID based upon PROs has been questioned in lumbar fusion surgery, as it has been thought to measure if the patient is “feeling” rather than “doing” better, the latter of which can be better measured by functional performance measures and objective, external socioeconomic anchors such as return to work and health care costs.27 Nevertheless, validated PROs are reported widely in the spine surgery literature. The MCID in the SF-36 is not well established and can depend upon whether the scores are at the extremes or more in the central range and whether there is large variability in the scores.28 Rheumatoid arthritis was estimated to be 7.1 points on the PF scale and 7.2 on the physical component summary (PCS).29 For total knee replacement, it has been estimated to be 10 points on the SF-36 PCS.30 Lumbar surgery was estimated to be 4.9 points for the SF-36 PCS and 12.8 points for the ODI.31 And the SPORT trial it has been estimated that a 30% change in the possible gain (or loss) may be an appropriate criterion.28
With a preoperative mean SF-36 PF of 28.9, a 30% improvement in the available range (70.1) would be 21 points, making our data mean improvement of 30 points above the MCID. With a mean preoperative ODI of 24.6, a 30% improvement in the available range (25.4) would be 7.6 points, making our data mean improvement of 10.3 points better than the MCID. Therefore, our outcome results are comparable with other lumbar fusion outcome studies in terms of degree of disability prior to surgery and amount of improvement from surgery.
Adjacent Segment Disease
The precise factors resulting in adjacent segment disease are not fully defined.3,32 In reviews of lumbar adjacent segment disease, reported rates ranged from 2.5% at 1 year up to 80 to 100% at 10 years, with lower rates with noninstrumented fusions.4,32-34 Annual incidence of symptomatic adjacent segment disease following lumbar fusion ranges from 0.6 to 3.9% per year.32,35,36 Mismatch between lumbar lordosis and pelvic incidence after fusion is thought to lead to higher rates of adjacent segment disease, as can a laminectomy at an adjacent segment.32,36 Percutaneous fusion techniques or use of the Wiltse approach may lower the risk of adjacent segment disease due to avoidance of facet capsule disruption.37,38
Dynamic stabilization techniques do not appear be clearly protective against adjacent segment disease, although biomechanical models suggest that they may do so.33,39,40 A review by Wang and colleagues pooled studies to assess the risk of lumbar adjacent segment disease in spinal fusion to compare to disc arthroplasty and concluded that fusion carried a higher risk of adjacent segment disease.41 Definitive data on other types of motion preservation devices is lacking.3We show 3 adjacent segment fusions and 1 laminectomy have been needed in 108 patients and at a mean of 46 months after the index procedure and over 2.5 years of mean overall follow up. This is a low adjacent segment surgery rate compared to the historical data cited above, and may suggest some advantage for PEEK rods over more rigid constructs.
Strengths and Limitations
Strengths of this study include larger numbers than prior series of PEEK rod use and use in a population with high comorbidities linked to poor results without reduction in good outcomes. PEEK rods as used at the VAPHCS do not result in higher instrumentation costs than all metal constructs.
Study limitations include the retrospective nature with loss of follow up on some patients and incomplete radiographic and PROs in some patients. The use of 100% stereotactic guidance, the avoidance of interbody devices, and the off-label use of bone morphogenetic protein as part of the fusion construct introduce additional variables that may influence comparison to other studies. To avoid unnecessary radiation exposure, flexion extension films or CT scans were not routinely obtained if patients were doing well.42 Additionally, the degree of motion on dynamic views that would differentiate pseudarthrosis from arthrodesis has not been defined.5
Conclusions
The results presented show that lumbar fusion with PEEK rods can be undertaken with short hospitalization times and low complication rates, produce satisfactory clinical improvements, and result in radiographic fusion rates similar to metal constructs. Low rates of hardware failure or need for revision surgery were found. Preliminarily results of low rates of adjacent segment surgery are comparable with previously published metal construct rates. Longer follow up is needed to confirm these findings and to investigate whether semirigid constructs truly offer some protection from adjacent segment disease when compared to all metal constructs.
Acknowledgments
The authors thank Shirley McCartney, PhD, for editorial assistance.
Surgical treatment of degenerative lumbar spine disease has been rising steadily in the United States, and an increasing fraction of surgery involves lumbar fusion.1,2 Various techniques are used to accomplish a lumbar fusion, including noninstrumented fusion, anterior lumbar interbody fusion (ALIF), lateral lumbar interbody fusion (XLIF, OLIF), posterior pedicle screw fusion, posterior cortical screw fusion, posterior interbody fusion (TLIF, PLIF), and interspinous process fusion. Rigid, metallic fusion hardware provides high stability and fusion rates, but it likely leads to stress shielding and adjacent segment disease.3 There is interest in less rigid and dynamic stabilization techniques to reduce the risk of adjacent segment disease, such as polyetheretherketone (PEEK) rods, which have been available since 2007. However, literature regarding PEEK rod utility is sparse and of mixed outcomes.3,4 Additional patient reported outcome (PRO) information would be useful to both surgeons and patients. Using institutional data, this review was designed to examine our experience with PEEK rod lumbar fusion and to document PROs.
Methods
The study was approved by the institutional review board at the US Department of Veterans Affairs (VA) Portland Health Care System (VAPHCS) in Oregon with a waiver of authorization. In this retrospective, single center study, data were queried from the senior author’s (DAR) case logs from VA Computerized Patient Record System (CPRS). Electronic medical records, imaging, and PROs of all consecutive patients undergoing lumbar fusion at 1 or 2 levels with PEEK rods for degenerative disease were retrospectively reviewed. Cases of trauma, malignancy, or infection were excluded. From March 2011 through October 2019, 108 patients underwent lumbar fusion with PEEK rods.
Surgeries were conducted on a Mizuho OSI Jackson Table via bilateral 3 to 4 cm Wiltse incisions using the Medtronic Quadrant retractor system. Medtronic O-Arm images were acquired and delivered to a Medtronic Stealth Station for navigation of the screws. Monopolar coagulation was not used. PEEK pedicle screws were placed and verified with a second O-Arm spin before placing lordotic PEEK rods in the screw heads. No attempt was made to reduce any spondylolisthesis, but distraction was used to open the foramina and indirectly decompress the canal. An interbody device was placed only in treatment of multiply recurrent disc protrusion. After decortication of the transverse processes and facets, intertransverse fusion constructs consisting of calcium hydroxyapatite soaked in autologous bone marrow blood and wrapped in 6-mg bone morphogenetic protein-soaked sponges were placed on the bone. If canal decompression was indicated, a Medtronic Metrx retractor tube was then placed through one of the incisions and decompression carried out. Wounds were closed with absorbable suture. No bracing was used postoperatively. Figure 1 shows a typical single level PEEK rod fusion construct.
Patient pre- and postoperative Short Form-36 (SF-36) physical function (PF) scores and Oswestry Disability Index (ODI) scores had been obtained at routine clinic visits.
Static radiographs were used to assess the fusion. Dynamic films and/or computed tomography (CT) scans were obtained only when symptomatic pseudarthrosis was suspected. Some patients had abdominal or lumbar CT scans for other indications, and these were reviewed when available. Particular care was taken to assess facet fusion as an indicator of arthrodesis (Figure 2).5
Statistical Analysis
Pre- and postoperative pairwise t tests were completed for patients with a complete data, using SAS 9.2 statistical package. Data are presented as standard deviation (SD) of the mean.
Results
Following application of the inclusion/exclusion criteria, 108 patients had undergone lumbar fusion with PEEK rods. Mean (SD) patient age was 60.2 (10.3) years and 88 patients were male (Table 1). Most surgeries were at L5-S1 and L4-5. There were 97 single-level fusions and 11 bilevel fusions. Seventy-four procedures were for spondylolisthesis, 23 for foraminal stenosis, 5 for degenerative disc disease, 3 for coronal imbalance with foraminal stenosis, 2 for pseudarthrosis after surgery elsewhere, and 1 for multiple recurrent disc herniation (Table 2). Twenty-five patients (23.1%) were current tobacco users and 28 (25.9%) were former smokers, 26 (24.1%) had diabetes mellitus (DM), 16 (14.8%) had low bone density by dual energy X-ray absorptiometry (DEXA) imaging, 35 (32.4%) had depression, and 7 (6.5%) were taking an immunosuppressive agent (chronic steroids, biological response modifiers, or methotrexate). Mean body mass index was 30.1.
Surgical Procedure
Of the 108 patients, the first 18 underwent a procedure with fluoroscopic guidance and the Medtronic FluoroNav and Stealth Systems. The next 90 patients underwent a procedure with O-Arm intraoperative CT scanning and Stealth frameless stereotactic navigation. The mean (SD) length of stay was 1.7 (1.3) days. There were no wound infections and no new neurologic deficits. Mean (SD) follow up time was 30.3 (21.8) months.
Imaging
Final imaging was by radiograph in 73 patients, CT in 31, and magnetic resonance imaging (MRI) in 3 (1 patient had no imaging). Sixty-seven patients (62.0%) had a bilateral arthrodesis, and 15 (13.9%) had at least a unilateral arthrodesis. MRI was not used to assess arthrodesis. Eight patients (7.4%) had no definite arthrodesis. Seventeen patients had inadequate or early imaging from which a fusion determination could not be made. Of 81 patients with > 11 months of follow up, 58 (71.6%) had a bilateral arthrodesis, 12 (14.8%) had a unilateral arthrodesis, 8 (9.9%) had no arthrodesis, and 3 (3.7%) were indeterminate.
No patient had any revision fusion surgery at the index level during follow up. Two patients had adjacent level fusions at 27 and 60 months after the index procedure. One patient had a laminectomy at an adjacent segment at 18 months postfusion, and 1 had a foraminotomy at an adjacent segment 89 months post fusion (Figure 3). Overall, there were 4 (3.7%) adjacent segment surgeries at a mean of 48.5 months after surgery. One patient had a sacro-iliac joint fusion below an L5-S1 fusion 17 months prior for persisting pain after the fusion procedure.
Patient Reported Outcomes
Preoperative SF-36 PF and ODI scores were available for 81 patients (Table 3). Postoperative SF-36 PF scores were obtained at 3 months for 65 of these patients, and at 1 year for 63 patients. Postoperative ODI scores were obtained at 3 months for 65 patients, and at 1 year for 55 patients. Among the 65 patients with completed SF-36 scores at 3 months, a mean increase of 22.4 (95% CI, 17-27; P < .001) was noted, and for the 63 patients at 1 year a mean increase of 30.3 (95% CI, 25-35; P < .001) was noted. Among the 65 patients with completed ODI scores at 3 months, a mean decrease of 6.8 (95% CI, 4.9-8.6; P < .001) was noted, and for the 55 patients with completed ODI scores at 1 year a mean decrease of 10.3 (n = 55; 95% CI, 8.4-12.2; P < .001) was noted.
Cost
We compared the hardware cost of a single level construct consisting of 4 pedicle screws, 4 locking caps, and 2 rods using a PEEK system with that of 2 other titanium construct systems. At VAPHCS, the PEEK system cost was about 71% of the cost of 2 other titanium construct systems and 62% of the cost when compared with Medtronic titanium rods.
Discussion
PEEK is useful for spine and cranial implants. It is inert and fully biocompatible with a modulus of elasticity between that of cortical and cancellous bone, and much lower than that of titanium, and is therefore considered to be semirigid.3,4,6 PEEK rods are intermediate in stiffness between titanium rods (110 Gigapascals) and dynamic devices such as the Zimmer Biomet DYNESYS dynamic stabilization system or the Premia Spine TOPS system.3 Carbon fiber rods and carbon fiber reinforced PEEK implants are other semirigid rod alternatives.7,8 PEEK rods for posterior lumbar fusion surgery were introduced in 2007. Li and colleagues provide a thorough review of the biomechanical properties of PEEK rods.3
PEEK is thought to have several advantages when compared with titanium. These advantages include more physiologic load sharing and reduction in stress shielding, improved durability, reduced risk of failure in osteoporotic bone, less wear debris, no change in bone forming environment, and imaging radiolucency.4,9 Spinal PEEK cages have been reported to allow more uniform radiation dose distribution compared with metal constructs, an advantage that also may pertain to PEEK rods.10 Disadvantages of PEEK rods include an inability to detect rod breakage easily, lack of data on the use in more than minimally unstable clinical situations, and greater expense, although this was not the authors’ observation.3,4,11
Importantly, it has been reported that PEEK rods permit a greater range of motion in all planes when compared with titanium rods.9 Polyetheretherketone rods unload the bone screw interface and increased the anterior column load to a more physiologic 75% when compared with titanium rods.6,9 However, in another biomechanical study that compared titanium rods, PEEK rods, and a dynamic stabilization device, it was reported that anterior load sharing was 55%, 59%, and 75%, respectively.12 This indicated that PEEK rods are closer to metal rods than truly dynamic devices for anterior load sharing. The endurance limit of a PEEK rod construct was similar to that of clinically useful metal systems.9 PEEK rods resulted in no increase in postfatigue motion compared with titanium rods in a biomechanical model.13 Intradiscal pressures at PEEK instrumented segments were similar to uninstrumented segments and greater than those with titanium rod constructs.14 Intradiscal pressures at adjacent segments were highest with dynamic devices, intermediate with semirigid rods, and lowest with rigid constructs; however, stress values at adjacent segments were lower in PEEK than titanium constructs in any direction of motion.15,16
Fusion Rates
The use of PEEK rods in lumbar fusion has been reported previously.3,4,17,18 However, these studies featured small sample sizes, short follow up times, and contradictory results.4 Of 8 outcome reports found in a systematic review, 2 studies reported on procedures designed to create nonfusion outcomes (a third similar trial from 2013 was not included in the systematic review), and 1 study reported only on the condition of PEEK rods removed at subsequent surgery.3,19-21 Reported fusion rates varied from 86 to 100%.
In 42 patients with PEEK rod fusions who were followed for a mean of 31.4 months, 5 patients required adjacent segment surgery and 3 patients were treated for interbody cage migration and nonunion.17 Radiographic fusion rate was 86%. These authors concluded that PEEK rod fusion results were similar to those of other constructs, but not better, or perhaps worse than, metal rods.
Other studies have reported better results with PEEK.11,18,19,22-24 Highsmith and colleagues reported on 3 successful example cases of the use of PEEK rods.11 De Iure and colleagues reported on 30 cases up to 5 levels (mean, 2.9) using autograft bone, with a mean follow up of 18 months.23 Results were reported as satisfactory. Three patients had radiographic nonunions, 1 of which required revision for asymptomatic screw loosening at the cranial end of the construct. Qi and colleagues, reported on 20 patients with PEEK rods compared to 21 patients with titanium alloy rods.24 Both groups had similar clinical outcomes, structural parameters, and 100% fusion rates. Athanasakopoulos and colleagues reported on 52 patients with up to 3 level fusions followed for a mean of 3 years.22 There were significant improvements in PROs: at 1 year 96% had radiographic union. Two patients had screw breakage, 1 of whom required revision to a metal rod construct. Colangeli and colleagues reported on 12 patients treated with PEEK rods compared with 12 who were treated with a dynamic system.18 They reported significant improvements, no complications, and 100% fusion at 6 months. Huang and colleagues reported on 38 patients intended to undergo a nonfusion procedure with 2 years of follow up.19 They reported good outcomes and 1 case of screw loosening. As no fusion was intended, no fusion outcomes were reported. All these studies suggested that longer follow up and more patients would be needed to assess the role of PEEK rods in lumbar fusion.3
Our results show a radiographic fusion rate of 86.4% and a radiographic nonunion rate of 9.9% in patients followed for at least 12 months. There was no clinical need for revision fusion at the index level. In our retrospective review, patients had high levels of smoking, DM, depression, immunosuppression, and obesity, which may negatively influence radiographic fusion rates when compared with other studies with 100% reported fusion rates. There was no instance of construct breakage or screw breakout, indicating that PEEK rods may allow enough flexibility to avoid construct failure under stress as in a fall.
Patient Reported Outcomes
Recent large studies were reviewed to assess the pre- and postoperative patient PROs reported in comparison with our study population (Table 4). In the Swedish Spine Registry analysis of 765 patients with 3 different types of lumbar fusion, the mean preoperative ODI score was 37 and mean SF-36 physical component score (PCS) was 35 for the most similar approach (posterolateral fusion with instrumentation).25 At 1 year postoperation, the mean ODI was 26 and mean SF-36 PCS was 43. In the Spine Patient Outcomes Research Trial (SPORT) spondylolisthesis trial of 3 fusion types, the mean preoperative ODI was 41.2 and mean SF-36 PF score was 31.2 for the most similar approach (posterolateral instrumented fusion with pedicle screws).26 Postoperative ODI scores at 1 year decreased by a mean 20.9 points and mean SF-36 PF scores increased by 29.9.
We report a mean preoperative SF-36 PF score of 28.9, which is lower than the SPORT study score for posterolateral fusion with instrumentation and the Swedish Study score for posterolateral instrumented fusion with pedicle screws. Similarly, our mean ODI score of 24.8 was better than the scores reported in the Swedish and SPORT studies. Our mean SF-36 PF score at 1 year postoperation was 59.3, compared with 58.5 for the SPORT study group and 46.0 in the Swedish study group. Mean ODI score at 1 year postoperatively was 14.5, which is better than the scores reported in the Swedish and SPORT studies.
Minimally clinically important difference (MCID) is a parameter used to gauge the efficacy of spine surgery. The utility of the MCID based upon PROs has been questioned in lumbar fusion surgery, as it has been thought to measure if the patient is “feeling” rather than “doing” better, the latter of which can be better measured by functional performance measures and objective, external socioeconomic anchors such as return to work and health care costs.27 Nevertheless, validated PROs are reported widely in the spine surgery literature. The MCID in the SF-36 is not well established and can depend upon whether the scores are at the extremes or more in the central range and whether there is large variability in the scores.28 Rheumatoid arthritis was estimated to be 7.1 points on the PF scale and 7.2 on the physical component summary (PCS).29 For total knee replacement, it has been estimated to be 10 points on the SF-36 PCS.30 Lumbar surgery was estimated to be 4.9 points for the SF-36 PCS and 12.8 points for the ODI.31 And the SPORT trial it has been estimated that a 30% change in the possible gain (or loss) may be an appropriate criterion.28
With a preoperative mean SF-36 PF of 28.9, a 30% improvement in the available range (70.1) would be 21 points, making our data mean improvement of 30 points above the MCID. With a mean preoperative ODI of 24.6, a 30% improvement in the available range (25.4) would be 7.6 points, making our data mean improvement of 10.3 points better than the MCID. Therefore, our outcome results are comparable with other lumbar fusion outcome studies in terms of degree of disability prior to surgery and amount of improvement from surgery.
Adjacent Segment Disease
The precise factors resulting in adjacent segment disease are not fully defined.3,32 In reviews of lumbar adjacent segment disease, reported rates ranged from 2.5% at 1 year up to 80 to 100% at 10 years, with lower rates with noninstrumented fusions.4,32-34 Annual incidence of symptomatic adjacent segment disease following lumbar fusion ranges from 0.6 to 3.9% per year.32,35,36 Mismatch between lumbar lordosis and pelvic incidence after fusion is thought to lead to higher rates of adjacent segment disease, as can a laminectomy at an adjacent segment.32,36 Percutaneous fusion techniques or use of the Wiltse approach may lower the risk of adjacent segment disease due to avoidance of facet capsule disruption.37,38
Dynamic stabilization techniques do not appear be clearly protective against adjacent segment disease, although biomechanical models suggest that they may do so.33,39,40 A review by Wang and colleagues pooled studies to assess the risk of lumbar adjacent segment disease in spinal fusion to compare to disc arthroplasty and concluded that fusion carried a higher risk of adjacent segment disease.41 Definitive data on other types of motion preservation devices is lacking.3We show 3 adjacent segment fusions and 1 laminectomy have been needed in 108 patients and at a mean of 46 months after the index procedure and over 2.5 years of mean overall follow up. This is a low adjacent segment surgery rate compared to the historical data cited above, and may suggest some advantage for PEEK rods over more rigid constructs.
Strengths and Limitations
Strengths of this study include larger numbers than prior series of PEEK rod use and use in a population with high comorbidities linked to poor results without reduction in good outcomes. PEEK rods as used at the VAPHCS do not result in higher instrumentation costs than all metal constructs.
Study limitations include the retrospective nature with loss of follow up on some patients and incomplete radiographic and PROs in some patients. The use of 100% stereotactic guidance, the avoidance of interbody devices, and the off-label use of bone morphogenetic protein as part of the fusion construct introduce additional variables that may influence comparison to other studies. To avoid unnecessary radiation exposure, flexion extension films or CT scans were not routinely obtained if patients were doing well.42 Additionally, the degree of motion on dynamic views that would differentiate pseudarthrosis from arthrodesis has not been defined.5
Conclusions
The results presented show that lumbar fusion with PEEK rods can be undertaken with short hospitalization times and low complication rates, produce satisfactory clinical improvements, and result in radiographic fusion rates similar to metal constructs. Low rates of hardware failure or need for revision surgery were found. Preliminarily results of low rates of adjacent segment surgery are comparable with previously published metal construct rates. Longer follow up is needed to confirm these findings and to investigate whether semirigid constructs truly offer some protection from adjacent segment disease when compared to all metal constructs.
Acknowledgments
The authors thank Shirley McCartney, PhD, for editorial assistance.
1. Deyo RA, Mirza SK, Martin BI, Kreuter W, Goodman DC, Jarvik JG. Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA. 2010;303(13):1259-1265. doi:10.1001/jama.2010.338
2. Machado GC, Maher CG, Ferreira PH, et al. Trends, complications, and costs for hospital admission and surgery for lumbar spinal stenosis. Spine (Phila Pa 1976). 2017;42(22):1737-1743. doi:10.1097/BRS.0000000000002207
3. Li C, Liu L, Shi JY, Yan KZ, Shen WZ, Yang ZR. Clinical and biomechanical researches of polyetheretherketone (PEEK) rods for semi-rigid lumbar fusion: a systematic review. Neurosurg Rev. 2018;41(2):375-389. doi:10.1007/s10143-016-0763-2
4. Mavrogenis AF, Vottis C, Triantafyllopoulos G, Papagelopoulos PJ, Pneumaticos SG. PEEK rod systems for the spine. Eur J Orthop Surg Traumatol. 2014;24 Suppl 1:S111-S116. doi:10.1007/s00590-014-1421-4
5. Choudhri TF, Mummaneni PV, Dhall SS, et al. Guideline update for the performance of fusion procedures for degenerative disease of the lumbar spine. Part 4: radiographic assessment of fusion status. J Neurosurg Spine. 2014;21(1):23-30. doi:10.3171/2014.4.SPINE14267
6. Ahn YH, Chen WM, Lee KY, Park KW, Lee SJ. Comparison of the load-sharing characteristics between pedicle-based dynamic and rigid rod devices. Biomed Mater. 2008;3(4):044101. doi:10.1088/1748-6041/3/4/044101
7. Ozer AF, Cevik OM, Erbulut DU, et al. A novel modular dynamic stabilization system for the treatment of degenerative spinal pathologies. Turk Neurosurg. 2019;29(1):115-120. doi:10.5137/1019-5149.JTN.23227-18.1
8. Hak DJ, Mauffrey C, Seligson D, Lindeque B. Use of carbon-fiber-reinforced composite implants in orthopedic surgery. Orthopedics. 2014;37(12):825-830. doi:10.3928/01477447-20141124-05
9. Gornet MF, Chan FW, Coleman JC, et al. Biomechanical assessment of a PEEK rod system for semi-rigid fixation of lumbar fusion constructs. J Biomech Eng. 2011;133(8):081009. doi:10.1115/1.4004862
10. Jackson JB 3rd, Crimaldi AJ, Peindl R, Norton HJ, Anderson WE, Patt JC. Effect of polyether ether ketone on therapeutic radiation to the spine: a pilot study. Spine (Phila Pa 1976). 2017;42(1):E1-E7. doi:10.1097/BRS.0000000000001695
11. Highsmith JM, Tumialán LM, Rodts GE Jr. Flexible rods and the case for dynamic stabilization. Neurosurg Focus. 2007;22(1):E11. Published 2007 Jan 15. doi:10.3171/foc.2007.22.1.11
12. Sengupta DK, Bucklen B, McAfee PC, Nichols J, Angara R, Khalil S. The comprehensive biomechanics and load-sharing of semirigid PEEK and semirigid posterior dynamic stabilization systems. Adv Orthop. 2013;2013:745610. doi:10.1155/2013/745610
13. Agarwal A, Ingels M, Kodigudla M, Momeni N, Goel V, Agarwal AK. Adjacent-level hypermobility and instrumented-level fatigue loosening with titanium and PEEK rods for a pedicle screw system: an in vitro study. J Biomech Eng. 2016;138(5):051004. doi:10.1115/1.4032965
14. Chou WK, Chien A, Wang JL. Biomechanical analysis between PEEK and titanium screw-rods spinal construct subjected to fatigue loading. J Spinal Disord Tech. 2015;28(3):E121-E125. doi:10.1097/BSD.0000000000000176
15. Shih KS Hsu CC, Zhou SY, Hou SM. Biomechanical investigation of pedicle screw-based posterior stabilization systems for the treatment of lumbar degenerative disc disease using finite element analyses. Biomed Eng: Appl Basis Commun. 2015;27(06):1550060. doi: 10.4015/S101623721550060X

16. Chang TK, Huang CH, Liu YC, et al. Biomechanical evaluation and comparison of polyetheretherketone rod system to traditional titanium rod fixation on adjacent levels. Formosan J Musculoskeletal Disord. 2013;4(2):42-47. doi: 10.1016/j.fjmd.2013.04.003
17. Ormond DR, Albert L Jr, Das K. Polyetheretherketone (PEEK) rods in lumbar spine degenerative disease: a case series. Clin Spine Surg. 2016;29(7):E371-E375. doi:10.1097/BSD.0b013e318277cb9b
18. Colangeli S, Barbanti Brodàno G, Gasbarrini A, et al. Polyetheretherketone (PEEK) rods: short-term results in lumbar spine degenerative disease. J Neurosurg Sci. 2015;59(2):91-96.
19. Huang W, Chang Z, Song R, Zhou K, Yu X. Non-fusion procedure using PEEK rod systems for lumbar degenerative diseases: clinical experience with a 2-year follow-up. BMC Musculoskelet Disord. 2016;17:53. Published 2016 Feb 1. doi:10.1186/s12891-016-0913-2
20. Wang C-J, Graf H, Wei H-W. Clinical outcomes of the dynamic lumbar pedicle screw-rod stabilization. Neurosurg Q. 2016;26(3):214-218. doi:10.1097/WNQ.0000000000000169
21. Kurtz SM, Lanman TH, Higgs G, et al. Retrieval analysis of PEEK rods for posterior fusion and motion preservation. Eur Spine J. 2013;22(12):2752-2759. doi:10.1007/s00586-013-2920-4
22. Athanasakopoulos M, Mavrogenis AF, Triantafyllopoulos G, Koufos S, Pneumaticos SG. Posterior spinal fusion using pedicle screws. Orthopedics. 2013;36(7):e951-e957. doi:10.3928/01477447-20130624-28
23. De Iure F, Bosco G, Cappuccio M, Paderni S, Amendola L. Posterior lumbar fusion by peek rods in degenerative spine: preliminary report on 30 cases. Eur Spine J. 2012;21 Suppl 1(Suppl 1):S50-S54. doi:10.1007/s00586-012-2219-x
24. Qi L, Li M, Zhang S, Xue J, Si H. Comparative effectiveness of PEEK rods versus titanium alloy rods in lumbar fusion: a preliminary report. Acta Neurochir (Wien). 2013;155(7):1187-1193. doi:10.1007/s00701-013-1772-3
25. Endler P, Ekman P, Möller H, Gerdhem P. Outcomes of posterolateral fusion with and without instrumentation and of interbody fusion for isthmic spondylolisthesis: a prospective study. J Bone Joint Surg Am. 2017;99(9):743-752. doi:10.2106/JBJS.16.00679
26. Abdu WA, Lurie JD, Spratt KF, et al. Degenerative spondylolisthesis: does fusion method influence outcome? Four-year results of the spine patient outcomes research trial. Spine (Phila Pa 1976). 2009;34(21):2351-2360. doi:10.1097/BRS.0b013e3181b8a829
27. Gatchel RJ, Mayer TG, Chou R. What does/should the minimum clinically important difference measure? A reconsideration of its clinical value in evaluating efficacy of lumbar fusion surgery. Clin J Pain. 2012;28(5):387-397. doi:10.1097/AJP.0b013e3182327f20
28. Spratt KF. Patient-level minimal clinically important difference based on clinical judgment and minimally detectable measurement difference: a rationale for the SF-36 physical function scale in the SPORT intervertebral disc herniation cohort. Spine (Phila Pa 1976). 2009;34(16):1722-1731. doi:10.1097/BRS.0b013e3181a8faf2
29. Ward MM, Guthrie LC, Alba MI. Clinically important changes in short form 36 health survey scales for use in rheumatoid arthritis clinical trials: the impact of low responsiveness. Arthritis Care Res (Hoboken). 2014;66(12):1783-1789. doi:10.1002/acr.22392
30. Escobar A, Quintana JM, Bilbao A, Aróstegui I, Lafuente I, Vidaurreta I. Responsiveness and clinically important differences for the WOMAC and SF-36 after total knee replacement. Osteoarthritis Cartilage. 2007;15(3):273-280. doi:10.1016/j.joca.2006.09.001
31. Copay AG, Glassman SD, Subach BR, Berven S, Schuler TC, Carreon LY. Minimum clinically important difference in lumbar spine surgery patients: a choice of methods using the Oswestry Disability Index, Medical Outcomes Study questionnaire Short Form 36, and pain scales. Spine J. 2008;8(6):968-974. doi:10.1016/j.spinee.2007.11.006
32. Radcliff KE, Kepler CK, Jakoi A, et al. Adjacent segment disease in the lumbar spine following different treatment interventions. Spine J. 2013;13(10):1339-1349. doi:10.1016/j.spinee.2013.03.020
33. Epstein NE. Adjacent level disease following lumbar spine surgery: a review. Surg Neurol Int. 2015;6(Suppl 24):S591-S599. Published 2015 Nov 25. doi:10.4103/2152-7806.170432
34. Epstein NE. A review: reduced reoperation rate for multilevel lumbar laminectomies with noninstrumented versus instrumented fusions. Surg Neurol Int. 2016;7(Suppl 13):S337-S346. Published 2016 May 17. doi:10.4103/2152-7806.182546
35. Scemama C, Magrino B, Gillet P, Guigui P. Risk of adjacent-segment disease requiring surgery after short lumbar fusion: results of the French Spine Surgery Society Series. J Neurosurg Spine. 2016;25(1):46-51. doi:10.3171/2015.11.SPINE15700
36. Tempel ZJ, Gandhoke GS, Bolinger BD, et al. The influence of pelvic incidence and lumbar lordosis mismatch on development of symptomatic adjacent level disease following single-level transforaminal lumbar interbody fusion. Neurosurgery. 2017;80(6):880-886. doi:10.1093/neuros/nyw073

37. Cheng YW, Chang PY, Wu JC, et al. Letter to the editor: Pedicle screw-based dynamic stabilization and adjacent-segment disease. J Neurosurg Spine. 2017;26(3):405-406. doi:10.3171/2016.7.SPINE16816
38. Street JT, Andrew Glennie R, Dea N, et al. A comparison of the Wiltse versus midline approaches in degenerative conditions of the lumbar spine. J Neurosurg Spine. 2016;25(3):332-338. doi:10.3171/2016.2.SPINE151018
39. Kuo CH, Huang WC, Wu JC, et al. Radiological adjacent-segment degeneration in L4-5 spondylolisthesis: comparison between dynamic stabilization and minimally invasive transforaminal lumbar interbody fusion. J Neurosurg Spine. 2018;29(3):250-258. doi:10.3171/2018.1.SPINE17993
40. Lee CH, Kim YE, Lee HJ, Kim DG, Kim CH. Biomechanical effects of hybrid stabilization on the risk of proximal adjacent-segment degeneration following lumbar spinal fusion using an interspinous device or a pedicle screw-based dynamic fixator. J Neurosurg Spine. 2017;27(6):643-649. doi:10.3171/2017.3.SPINE161169
41. Wang JC, Arnold PM, Hermsmeyer JT, Norvell DC. Do lumbar motion preserving devices reduce the risk of adjacent segment pathology compared with fusion surgery? A systematic review. Spine (Phila Pa 1976). 2012;37(22 Suppl):S133-S143. doi:10.1097/BRS.0b013e31826cadf2
42. Ross DA. Letter to the editor: steroid use in anterior cervical discectomy and fusion. J Neurosurg Spine. 2016;24(6):998-1000. doi:10.3171/2015.9.SPINE151052
1. Deyo RA, Mirza SK, Martin BI, Kreuter W, Goodman DC, Jarvik JG. Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA. 2010;303(13):1259-1265. doi:10.1001/jama.2010.338
2. Machado GC, Maher CG, Ferreira PH, et al. Trends, complications, and costs for hospital admission and surgery for lumbar spinal stenosis. Spine (Phila Pa 1976). 2017;42(22):1737-1743. doi:10.1097/BRS.0000000000002207
3. Li C, Liu L, Shi JY, Yan KZ, Shen WZ, Yang ZR. Clinical and biomechanical researches of polyetheretherketone (PEEK) rods for semi-rigid lumbar fusion: a systematic review. Neurosurg Rev. 2018;41(2):375-389. doi:10.1007/s10143-016-0763-2
4. Mavrogenis AF, Vottis C, Triantafyllopoulos G, Papagelopoulos PJ, Pneumaticos SG. PEEK rod systems for the spine. Eur J Orthop Surg Traumatol. 2014;24 Suppl 1:S111-S116. doi:10.1007/s00590-014-1421-4
5. Choudhri TF, Mummaneni PV, Dhall SS, et al. Guideline update for the performance of fusion procedures for degenerative disease of the lumbar spine. Part 4: radiographic assessment of fusion status. J Neurosurg Spine. 2014;21(1):23-30. doi:10.3171/2014.4.SPINE14267
6. Ahn YH, Chen WM, Lee KY, Park KW, Lee SJ. Comparison of the load-sharing characteristics between pedicle-based dynamic and rigid rod devices. Biomed Mater. 2008;3(4):044101. doi:10.1088/1748-6041/3/4/044101
7. Ozer AF, Cevik OM, Erbulut DU, et al. A novel modular dynamic stabilization system for the treatment of degenerative spinal pathologies. Turk Neurosurg. 2019;29(1):115-120. doi:10.5137/1019-5149.JTN.23227-18.1
8. Hak DJ, Mauffrey C, Seligson D, Lindeque B. Use of carbon-fiber-reinforced composite implants in orthopedic surgery. Orthopedics. 2014;37(12):825-830. doi:10.3928/01477447-20141124-05
9. Gornet MF, Chan FW, Coleman JC, et al. Biomechanical assessment of a PEEK rod system for semi-rigid fixation of lumbar fusion constructs. J Biomech Eng. 2011;133(8):081009. doi:10.1115/1.4004862
10. Jackson JB 3rd, Crimaldi AJ, Peindl R, Norton HJ, Anderson WE, Patt JC. Effect of polyether ether ketone on therapeutic radiation to the spine: a pilot study. Spine (Phila Pa 1976). 2017;42(1):E1-E7. doi:10.1097/BRS.0000000000001695
11. Highsmith JM, Tumialán LM, Rodts GE Jr. Flexible rods and the case for dynamic stabilization. Neurosurg Focus. 2007;22(1):E11. Published 2007 Jan 15. doi:10.3171/foc.2007.22.1.11
12. Sengupta DK, Bucklen B, McAfee PC, Nichols J, Angara R, Khalil S. The comprehensive biomechanics and load-sharing of semirigid PEEK and semirigid posterior dynamic stabilization systems. Adv Orthop. 2013;2013:745610. doi:10.1155/2013/745610
13. Agarwal A, Ingels M, Kodigudla M, Momeni N, Goel V, Agarwal AK. Adjacent-level hypermobility and instrumented-level fatigue loosening with titanium and PEEK rods for a pedicle screw system: an in vitro study. J Biomech Eng. 2016;138(5):051004. doi:10.1115/1.4032965
14. Chou WK, Chien A, Wang JL. Biomechanical analysis between PEEK and titanium screw-rods spinal construct subjected to fatigue loading. J Spinal Disord Tech. 2015;28(3):E121-E125. doi:10.1097/BSD.0000000000000176
15. Shih KS Hsu CC, Zhou SY, Hou SM. Biomechanical investigation of pedicle screw-based posterior stabilization systems for the treatment of lumbar degenerative disc disease using finite element analyses. Biomed Eng: Appl Basis Commun. 2015;27(06):1550060. doi: 10.4015/S101623721550060X

16. Chang TK, Huang CH, Liu YC, et al. Biomechanical evaluation and comparison of polyetheretherketone rod system to traditional titanium rod fixation on adjacent levels. Formosan J Musculoskeletal Disord. 2013;4(2):42-47. doi: 10.1016/j.fjmd.2013.04.003
17. Ormond DR, Albert L Jr, Das K. Polyetheretherketone (PEEK) rods in lumbar spine degenerative disease: a case series. Clin Spine Surg. 2016;29(7):E371-E375. doi:10.1097/BSD.0b013e318277cb9b
18. Colangeli S, Barbanti Brodàno G, Gasbarrini A, et al. Polyetheretherketone (PEEK) rods: short-term results in lumbar spine degenerative disease. J Neurosurg Sci. 2015;59(2):91-96.
19. Huang W, Chang Z, Song R, Zhou K, Yu X. Non-fusion procedure using PEEK rod systems for lumbar degenerative diseases: clinical experience with a 2-year follow-up. BMC Musculoskelet Disord. 2016;17:53. Published 2016 Feb 1. doi:10.1186/s12891-016-0913-2
20. Wang C-J, Graf H, Wei H-W. Clinical outcomes of the dynamic lumbar pedicle screw-rod stabilization. Neurosurg Q. 2016;26(3):214-218. doi:10.1097/WNQ.0000000000000169
21. Kurtz SM, Lanman TH, Higgs G, et al. Retrieval analysis of PEEK rods for posterior fusion and motion preservation. Eur Spine J. 2013;22(12):2752-2759. doi:10.1007/s00586-013-2920-4
22. Athanasakopoulos M, Mavrogenis AF, Triantafyllopoulos G, Koufos S, Pneumaticos SG. Posterior spinal fusion using pedicle screws. Orthopedics. 2013;36(7):e951-e957. doi:10.3928/01477447-20130624-28
23. De Iure F, Bosco G, Cappuccio M, Paderni S, Amendola L. Posterior lumbar fusion by peek rods in degenerative spine: preliminary report on 30 cases. Eur Spine J. 2012;21 Suppl 1(Suppl 1):S50-S54. doi:10.1007/s00586-012-2219-x
24. Qi L, Li M, Zhang S, Xue J, Si H. Comparative effectiveness of PEEK rods versus titanium alloy rods in lumbar fusion: a preliminary report. Acta Neurochir (Wien). 2013;155(7):1187-1193. doi:10.1007/s00701-013-1772-3
25. Endler P, Ekman P, Möller H, Gerdhem P. Outcomes of posterolateral fusion with and without instrumentation and of interbody fusion for isthmic spondylolisthesis: a prospective study. J Bone Joint Surg Am. 2017;99(9):743-752. doi:10.2106/JBJS.16.00679
26. Abdu WA, Lurie JD, Spratt KF, et al. Degenerative spondylolisthesis: does fusion method influence outcome? Four-year results of the spine patient outcomes research trial. Spine (Phila Pa 1976). 2009;34(21):2351-2360. doi:10.1097/BRS.0b013e3181b8a829
27. Gatchel RJ, Mayer TG, Chou R. What does/should the minimum clinically important difference measure? A reconsideration of its clinical value in evaluating efficacy of lumbar fusion surgery. Clin J Pain. 2012;28(5):387-397. doi:10.1097/AJP.0b013e3182327f20
28. Spratt KF. Patient-level minimal clinically important difference based on clinical judgment and minimally detectable measurement difference: a rationale for the SF-36 physical function scale in the SPORT intervertebral disc herniation cohort. Spine (Phila Pa 1976). 2009;34(16):1722-1731. doi:10.1097/BRS.0b013e3181a8faf2
29. Ward MM, Guthrie LC, Alba MI. Clinically important changes in short form 36 health survey scales for use in rheumatoid arthritis clinical trials: the impact of low responsiveness. Arthritis Care Res (Hoboken). 2014;66(12):1783-1789. doi:10.1002/acr.22392
30. Escobar A, Quintana JM, Bilbao A, Aróstegui I, Lafuente I, Vidaurreta I. Responsiveness and clinically important differences for the WOMAC and SF-36 after total knee replacement. Osteoarthritis Cartilage. 2007;15(3):273-280. doi:10.1016/j.joca.2006.09.001
31. Copay AG, Glassman SD, Subach BR, Berven S, Schuler TC, Carreon LY. Minimum clinically important difference in lumbar spine surgery patients: a choice of methods using the Oswestry Disability Index, Medical Outcomes Study questionnaire Short Form 36, and pain scales. Spine J. 2008;8(6):968-974. doi:10.1016/j.spinee.2007.11.006
32. Radcliff KE, Kepler CK, Jakoi A, et al. Adjacent segment disease in the lumbar spine following different treatment interventions. Spine J. 2013;13(10):1339-1349. doi:10.1016/j.spinee.2013.03.020
33. Epstein NE. Adjacent level disease following lumbar spine surgery: a review. Surg Neurol Int. 2015;6(Suppl 24):S591-S599. Published 2015 Nov 25. doi:10.4103/2152-7806.170432
34. Epstein NE. A review: reduced reoperation rate for multilevel lumbar laminectomies with noninstrumented versus instrumented fusions. Surg Neurol Int. 2016;7(Suppl 13):S337-S346. Published 2016 May 17. doi:10.4103/2152-7806.182546
35. Scemama C, Magrino B, Gillet P, Guigui P. Risk of adjacent-segment disease requiring surgery after short lumbar fusion: results of the French Spine Surgery Society Series. J Neurosurg Spine. 2016;25(1):46-51. doi:10.3171/2015.11.SPINE15700
36. Tempel ZJ, Gandhoke GS, Bolinger BD, et al. The influence of pelvic incidence and lumbar lordosis mismatch on development of symptomatic adjacent level disease following single-level transforaminal lumbar interbody fusion. Neurosurgery. 2017;80(6):880-886. doi:10.1093/neuros/nyw073

37. Cheng YW, Chang PY, Wu JC, et al. Letter to the editor: Pedicle screw-based dynamic stabilization and adjacent-segment disease. J Neurosurg Spine. 2017;26(3):405-406. doi:10.3171/2016.7.SPINE16816
38. Street JT, Andrew Glennie R, Dea N, et al. A comparison of the Wiltse versus midline approaches in degenerative conditions of the lumbar spine. J Neurosurg Spine. 2016;25(3):332-338. doi:10.3171/2016.2.SPINE151018
39. Kuo CH, Huang WC, Wu JC, et al. Radiological adjacent-segment degeneration in L4-5 spondylolisthesis: comparison between dynamic stabilization and minimally invasive transforaminal lumbar interbody fusion. J Neurosurg Spine. 2018;29(3):250-258. doi:10.3171/2018.1.SPINE17993
40. Lee CH, Kim YE, Lee HJ, Kim DG, Kim CH. Biomechanical effects of hybrid stabilization on the risk of proximal adjacent-segment degeneration following lumbar spinal fusion using an interspinous device or a pedicle screw-based dynamic fixator. J Neurosurg Spine. 2017;27(6):643-649. doi:10.3171/2017.3.SPINE161169
41. Wang JC, Arnold PM, Hermsmeyer JT, Norvell DC. Do lumbar motion preserving devices reduce the risk of adjacent segment pathology compared with fusion surgery? A systematic review. Spine (Phila Pa 1976). 2012;37(22 Suppl):S133-S143. doi:10.1097/BRS.0b013e31826cadf2
42. Ross DA. Letter to the editor: steroid use in anterior cervical discectomy and fusion. J Neurosurg Spine. 2016;24(6):998-1000. doi:10.3171/2015.9.SPINE151052
Neurosurgical Subspecialty Bedside Guide Improves Nursing Confidence
The VA Portland Healthcare System (VAPHCS) is a 277-bed facility that serves more than 85,000 inpatient and 880,000 outpatient visits each year from veterans in Oregon and southwestern Washington. The VAPHCS consists of a main tertiary care VAMC with an acute medical and surgical facility that includes 30 beds serving qualifying veterans. Supported surgical specialties include urology, general surgery, vascular surgery, otolaryngology, orthopedic surgery, ophthalmology, cardiothoracic surgery, transplant surgery, and neurological surgery. Neurosurgical patients account for about 12% to 13% of annual surgical patients. The VAPHCS also is partnered with Oregon Health & Science University in the training of health care professionals, such as physicians and nurses.
The expectation at the VAPHCS is that medical-surgical nurses care for 4 to 5 concurrent patients, often from different surgical services. Caring for patients with different medical and surgical needs, variable ambulatory, swallowing, and elimination functions, and different physician teams can become confusing; even within a single surgical service, postoperative care due to procedure complexity, specificity of care orders, and the real possibility of medical catastrophe can seem overwhelming. Therefore, subspecialty nursing training poses a challenge that requires technical in-service and didactic education and allocation of resources.
Despite systems level subspecialty nursing training, medical emergencies identified at the bedside can be mismanaged.1 Errors in care can be due to an incomplete knowledge of the patient’s procedure and misunderstanding of positioning and activity limitations.
To encourage medical-surgical nurses to become more engaged and confident in subspecialty patient care, the authors developed a bedside neurosurgical nursing guide to allow for independent procedure related education. The comprehensive guide summarized the clinical course for postoperative neurosurgical patients undergoing cranial and spinal surgeries. This guide included appropriate surgery-related images, procedure overviews, management decisions, potential postoperative complications, and wound care directions. The guide was distributed to medical-surgical nurses caring for neurosurgical patients. The authors hypothesized that the guide would enable nurses to better predict adverse outcomes and respond appropriately and would improve confidence in patient care.
Methods
For educational purposes, a bedside neurosurgical nursing guide (text and graphics) was created for the 16 surgical subspecialty nurses at the VAPHCS. The guide detailed the most common cranial and spinal neurosurgical procedures performed at VAPHCS and was written based on a typical postoperative course for each procedure by the chief neurosurgery resident at VAPHCS with collaboration from the attending neurosurgeons (Figure).
A quality improvement (QI) project was undertaken to assess nursing confidence with neurosurgical patients’ care pre- and postfamiliarity with the bedside neurosurgical nursing guide. A literature search revealed no validated survey assessing nursing confidence, so one was created using the Likert scale. Specifically, an anonymous 6-question survey was completed by all 16 surgical nurses prior to familiarization with the guide. Responses were recorded as scores of 1 to 5 for questions 1, 3, and 4, with a response of 1 indicative of no comfort or confidence and a response of 5 indicative of the highest level of comfort or confidence. Responses were recorded as either true or false for questions 2 and 6, and never, occasionally, frequently, or always for question 5.
The guide was made available to nurses for 6 months without encouragement to use it. After 6 months, a 3-week period of familiarization with and education about the availability of the guide was instituted at morning nursing reports; the total availability of the guide to nursing staff was 6 months 3 weeks. After this period the same 6-question survey was distributed, and data were collected.
Survey responses were categorized into 2 groups. Responses to questions 1, 3, and 4 were categorized as group 1, and responses to questions 4 and 5 were categorized as group 2. Responses (never and occasionally) to question 5, were categorized as group 1 and responses (frequently and always) as group 2 (Table). Responses to questions 2 and 6 were grouped 1 for true and 2 for false. Nurses participating in this study ranged in age from 22 to 57 years, education level ranged from registered nurse to a bachelor of science in nursing, and years of experience ranged from < 1 year to 27 years.
Statistics were calculated using chi-square analysis with Yates correction online calculator. For the chi-square analysis, the prefamiliarization data for groups 1 and 2 were used as the expected values, and the postfamiliarization data were used for the observed values. In this manner, differences were discerned between the before and after questionnaire responses. The VAPHCS institutional review board determined that the study was not human research and exempt from review.
Findings
Anonymous survey responses were collected from all 16 surgical subspecialty nurses both prior and after familiarization with the nursing guide.The response rate was 100% with only a few incomplete responses excluded from the analysis. Three questions in the prefamiliarization questionnaire had no appropriate response, and 1 question in the postfamiliarization survey had no appropriate response.
Improvement was statistically significant in responses for questions 1, 3, 5, and 6 (P = .026, .008, .004, and .033, respectively). No significant differences were found for questions 2 and 4 (P = .974 and .116, respectively). It is possible that there was no significant difference in question 2 because prefamiliarization responses were already favorable. Even if nurses did not feel comfortable taking care of neurosurgical patients (as assessed in question 1), they noted confidence improvement by working on the ward and through informal assimilation of knowledge and skill, which would have accumulated naturally over 1 year.
Prior to familiarization with the guide, 7 nurses did not feel confident in assessing the need to contact a physician (question 4). After familiarization with the nursing guide, favorable responses increased from 9 to 14 nurses. Results trended toward but did not reach statistical significance, likely due to the small sample size.
Ultimately, in the 16 surgical subspecialty nurses surveyed, familiarization with the nursing guide was shown to improve comfort in taking care of neurosurgical patients and increase confidence in patient care skills. At the end of the QI project (6 months, 3 weeks), all nurses knew where to locate the bedside neurosurgical nursing guide and were familiar with it and its use. The guide remains accessible to the medical-surgical nurses and continues to be used.
Discussion
Nursing confidence has an undervalued effect on patient care.2 Confidence, or a belief in one’s own ability, varies directly with competence. Systematic quantification of nursing competence has been extensively studied using self-report questionnaires and clinical simulations.2,3 Competency can be quantified and normalized using formal assessment; however, confidence is somewhat intangible. Nursing confidence is a situation-dependent subjective feeling of security and is derived from an internalized assessment of skills that are commensurate with patient needs. Nursing confidence is further influenced by an intuited value within the care team, adequate knowledge of the patient’s condition, and procedures and protocols.4
A similar but less specific definition deconstructs nursing confidence as “significance of a professional network of coworkers” and the “importance of confirmation of professional role and competence.”5 The professional network of coworkers is invaluable as it underlies the essence of patient-centered care. The adaptive leadership framework is integral to the modern delivery of patient care, and via this framework frontline clinical staff, including nurses, are empowered.6,7
The second portion of Haavardsholm and Nåden’s definition, “importance of confirmation of professional role and competence” describes the association of the most easily augmented correlate of confidence: competency.5 Nursing competency is supplemented continuously with in-service training and recertification processes; however, despite this, demands placed on nurses can be technologically advanced and extremely varied. Nursing competency is known to directly correlate with increasing education, as nurses holding a master’s degree have been shown to outperform those with a bachelor of nursing degree.3
Increased formal education as well as increased work experience (> 5 years) are correlated with increased critical thinking ability.4,5 The critical thinking ability of health care providers can be fortified by clinical simulation, which leads to statistically significant improvement in clinical competency.2,3
A literature review of Medline and the National Library of Medicine PubMed online databases for search terms (nurs*, confidence, bedside, guide) was performed but did not result in original research assessing nurse confidence related to bedside guides. In this population, nurses were anonymously compared against their own historical data obviating any effect of education or experience on survey measures.
Nursing Self-Confidence
Evidence suggests that nursing confidence is a complex manifestation of the security felt within the care team and the comfort of one’s own professional abilities.4 Patients’ trust in the team caring for them is based on the confidence exuded by the team.8 In this way, nursing confidence can affect the patient-care team profoundly. Value is maximized when a nurse’s self-confidence engenders patient confidence and trust. Due to the varied patient load and complexity of subspecialty nursing care, it is hypothesized that bedside manuals/guidelines can be used to educate the subspecialty nurses on specific patient-related issues.
Nursing practice competence and confidence is vital to providing care for patients with complex postsurgical health care needs. Patient safety and outcome are paramount. This can be intimidating for newly qualified surgical subspecialty nurses who have not yet had experience with or adequate exposure to patients with complex postsurgical needs. Surgical nursing continuing education places an emphasis on adaptation to ever-changing specialized surgical procedures and postoperative patient care. Nevertheless, it is difficult for surgical subspecialty nurses to learn and retain all the possible complexities of individual cases and to confidently, appropriately, and safely care for patients especially when adverse events arise.
Recognizing that leadership is personal and not dependent on hierarchy, surgical subspecialty nurses may be better suited to specific bedside training and counseling.6,9 A key factor influencing nursing confidence is communication and collaboration with physicians.9 The role of the physician at VA medical facilities is no longer to be a commanding figure with complete medical autonomy; rather, a unified team of specialized practitioners collaboratively facilitate and deliver patient care.
There is no specific research detailing the use of bedside nursing guides in caring for postoperative patients. However, at VAPHCS, nurses created supplemental material regarding postoperative acute care of vascular surgery patients, which was found to be subjectively helpful in elevating nursing confidence. To the authors’ knowledge, no such supplemental information/guide exists for other specialty surgical services.
The surgical nursing guide created here detailed visuals of many common neurosurgical procedures performed at VAPHCS and included a prioritized checklist, which the 16 surgical subspecialty nurses could reference postoperatively. The authors hypothesized that this would enhance the nurses’ ability to efficiently manage specific situations while bridging communication gaps between surgical teams and nurses. The survey results agree with previous reports that suggested that the application of an adaptive leadership framework would empower nurses to deliver excellent patient-centered care, care that can be augmented with subspecialty nursing guides.7,10
Based on these results the authors propose that subspecialty surgical services consider use of a practical nursing guide for all surgical subspecialty nurses to reference, improve familiarity with procedures, and provide guidance to manage adverse events. Since implementing this reader-friendly paradigm within neurosurgical care, a nurse driven expansion has now included other subspecialty services at the VAPHCS with success.
Limitations
Survey responses have inherent bias and sampling error rates. The sample size for this survey was small. Data were grouped for data analysis. Competency and patient outcomes were not measured.
Future Directions
Despite specific surgical specialty postoperative patient care training, an overall lack of confidence can persist. A physician-created neurosurgical nursing guide that detailed the most common neurosurgical procedures, expected postoperative care, and potential emergencies was shown to improve nursing confidence. Collaborative (physician and nursing leaders) QI projects, such as described here; development of specific surgical specialty initiatives designed to improve confidence and quality; and nurse-physician communication and teamwork could lead to improved patient satisfaction and outcome.
The costs associated with developing and using bedside nursing guides are relatively low, and efficiency can be considered high. Competency improvement could be measured by creating a specialty-specific case scenario question bank. Effects on patient satisfaction and outcome could be measured by a patient satisfaction survey. Improvements in beside catastrophe management could be prospectively tracked; for example, rates of mismanagement of mobility status, emergent transfers to the intensive care unit, or poor wound care could be compared pre- and postfamiliarization with a subspecialty guide.
Conclusion
Familiarization with the VAPHCS neurosurgical nursing guide had a positive impact on the confidence of medical-surgical nurses caring for neurosurgical patients. Medical-surgical nurses were more comfortable taking care of neurosurgical patients; they felt the guide helped improve skills and noted improved knowledge regarding involvement of physician oversight. Although objective parameters were not assessed, improvement in nursing confidence in general leads to improved overall nurse-physician communication and patient management. A further study might target objective parameters associated with guide usage, such as changes in the number of emergencies or calls to physicians regarding management.
Acknowledgments
The authors thank Andy Rekito, MS, for illustrative assistance.
1. Pusateri ME, Prior MM, Kiely SC. The role of the non-ICU staff nurse on a medical emergency team: perceptions and understanding. Am J Nurs. 2011;111(5):22-29, quiz 30-31.
2. Bambini D, Washburn J, Perkins R. Outcomes of clinical simulation for novice nursing students: communication, confidence, clinical judgment. Nurs Educ Perspect. 2009;30(2):79-82.
3. Chang MJ, Chang YJ, Kuo SH, Yang YH, Chou FH. Relationships between critical thinking ability and nursing competence in clinical nurses. J Clin Nurs. 2011;20(21-22):3224-3232.
4. Perry P. Concept analysis: confidence/self-confidence. Nurs Forum. 2011;46(4):218-230.
5. Haavardsholm I, Nåden D. The concept of confidence—the nurse’s perception. Eur J Cancer Care (Engl). 2009;18(5):483-491.
6. Bailey DE Jr, Docherty SL, Adams JA, et al. Studying the clinical encounter with the Adaptive Leadership framework. J Healthc Leadersh. 2012;2012(4):83-91.
7. Hall C, McCutcheon H, Deuter K, Matricciani L. Evaluating and improving a model of nursing care delivery: a process of partnership. Collegian. 2012;19(4):203-210.
8. Williams AM, Irurita VF. Therapeutic and non-therapeutic interpersonal interactions: the patient’s perspective. J Clin Nurs. 2004;13(7):806-815.
9. Carryer J. Collaboration between doctors and nurses. J Prim Health Care. 2011;3(1):77-79.
10. Chadwick MM. Creating order out of chaos: a leadership approach. AORN J. 2010;91(1):154-170.
The VA Portland Healthcare System (VAPHCS) is a 277-bed facility that serves more than 85,000 inpatient and 880,000 outpatient visits each year from veterans in Oregon and southwestern Washington. The VAPHCS consists of a main tertiary care VAMC with an acute medical and surgical facility that includes 30 beds serving qualifying veterans. Supported surgical specialties include urology, general surgery, vascular surgery, otolaryngology, orthopedic surgery, ophthalmology, cardiothoracic surgery, transplant surgery, and neurological surgery. Neurosurgical patients account for about 12% to 13% of annual surgical patients. The VAPHCS also is partnered with Oregon Health & Science University in the training of health care professionals, such as physicians and nurses.
The expectation at the VAPHCS is that medical-surgical nurses care for 4 to 5 concurrent patients, often from different surgical services. Caring for patients with different medical and surgical needs, variable ambulatory, swallowing, and elimination functions, and different physician teams can become confusing; even within a single surgical service, postoperative care due to procedure complexity, specificity of care orders, and the real possibility of medical catastrophe can seem overwhelming. Therefore, subspecialty nursing training poses a challenge that requires technical in-service and didactic education and allocation of resources.
Despite systems level subspecialty nursing training, medical emergencies identified at the bedside can be mismanaged.1 Errors in care can be due to an incomplete knowledge of the patient’s procedure and misunderstanding of positioning and activity limitations.
To encourage medical-surgical nurses to become more engaged and confident in subspecialty patient care, the authors developed a bedside neurosurgical nursing guide to allow for independent procedure related education. The comprehensive guide summarized the clinical course for postoperative neurosurgical patients undergoing cranial and spinal surgeries. This guide included appropriate surgery-related images, procedure overviews, management decisions, potential postoperative complications, and wound care directions. The guide was distributed to medical-surgical nurses caring for neurosurgical patients. The authors hypothesized that the guide would enable nurses to better predict adverse outcomes and respond appropriately and would improve confidence in patient care.
Methods
For educational purposes, a bedside neurosurgical nursing guide (text and graphics) was created for the 16 surgical subspecialty nurses at the VAPHCS. The guide detailed the most common cranial and spinal neurosurgical procedures performed at VAPHCS and was written based on a typical postoperative course for each procedure by the chief neurosurgery resident at VAPHCS with collaboration from the attending neurosurgeons (Figure).
A quality improvement (QI) project was undertaken to assess nursing confidence with neurosurgical patients’ care pre- and postfamiliarity with the bedside neurosurgical nursing guide. A literature search revealed no validated survey assessing nursing confidence, so one was created using the Likert scale. Specifically, an anonymous 6-question survey was completed by all 16 surgical nurses prior to familiarization with the guide. Responses were recorded as scores of 1 to 5 for questions 1, 3, and 4, with a response of 1 indicative of no comfort or confidence and a response of 5 indicative of the highest level of comfort or confidence. Responses were recorded as either true or false for questions 2 and 6, and never, occasionally, frequently, or always for question 5.
The guide was made available to nurses for 6 months without encouragement to use it. After 6 months, a 3-week period of familiarization with and education about the availability of the guide was instituted at morning nursing reports; the total availability of the guide to nursing staff was 6 months 3 weeks. After this period the same 6-question survey was distributed, and data were collected.
Survey responses were categorized into 2 groups. Responses to questions 1, 3, and 4 were categorized as group 1, and responses to questions 4 and 5 were categorized as group 2. Responses (never and occasionally) to question 5, were categorized as group 1 and responses (frequently and always) as group 2 (Table). Responses to questions 2 and 6 were grouped 1 for true and 2 for false. Nurses participating in this study ranged in age from 22 to 57 years, education level ranged from registered nurse to a bachelor of science in nursing, and years of experience ranged from < 1 year to 27 years.
Statistics were calculated using chi-square analysis with Yates correction online calculator. For the chi-square analysis, the prefamiliarization data for groups 1 and 2 were used as the expected values, and the postfamiliarization data were used for the observed values. In this manner, differences were discerned between the before and after questionnaire responses. The VAPHCS institutional review board determined that the study was not human research and exempt from review.
Findings
Anonymous survey responses were collected from all 16 surgical subspecialty nurses both prior and after familiarization with the nursing guide.The response rate was 100% with only a few incomplete responses excluded from the analysis. Three questions in the prefamiliarization questionnaire had no appropriate response, and 1 question in the postfamiliarization survey had no appropriate response.
Improvement was statistically significant in responses for questions 1, 3, 5, and 6 (P = .026, .008, .004, and .033, respectively). No significant differences were found for questions 2 and 4 (P = .974 and .116, respectively). It is possible that there was no significant difference in question 2 because prefamiliarization responses were already favorable. Even if nurses did not feel comfortable taking care of neurosurgical patients (as assessed in question 1), they noted confidence improvement by working on the ward and through informal assimilation of knowledge and skill, which would have accumulated naturally over 1 year.
Prior to familiarization with the guide, 7 nurses did not feel confident in assessing the need to contact a physician (question 4). After familiarization with the nursing guide, favorable responses increased from 9 to 14 nurses. Results trended toward but did not reach statistical significance, likely due to the small sample size.
Ultimately, in the 16 surgical subspecialty nurses surveyed, familiarization with the nursing guide was shown to improve comfort in taking care of neurosurgical patients and increase confidence in patient care skills. At the end of the QI project (6 months, 3 weeks), all nurses knew where to locate the bedside neurosurgical nursing guide and were familiar with it and its use. The guide remains accessible to the medical-surgical nurses and continues to be used.
Discussion
Nursing confidence has an undervalued effect on patient care.2 Confidence, or a belief in one’s own ability, varies directly with competence. Systematic quantification of nursing competence has been extensively studied using self-report questionnaires and clinical simulations.2,3 Competency can be quantified and normalized using formal assessment; however, confidence is somewhat intangible. Nursing confidence is a situation-dependent subjective feeling of security and is derived from an internalized assessment of skills that are commensurate with patient needs. Nursing confidence is further influenced by an intuited value within the care team, adequate knowledge of the patient’s condition, and procedures and protocols.4
A similar but less specific definition deconstructs nursing confidence as “significance of a professional network of coworkers” and the “importance of confirmation of professional role and competence.”5 The professional network of coworkers is invaluable as it underlies the essence of patient-centered care. The adaptive leadership framework is integral to the modern delivery of patient care, and via this framework frontline clinical staff, including nurses, are empowered.6,7
The second portion of Haavardsholm and Nåden’s definition, “importance of confirmation of professional role and competence” describes the association of the most easily augmented correlate of confidence: competency.5 Nursing competency is supplemented continuously with in-service training and recertification processes; however, despite this, demands placed on nurses can be technologically advanced and extremely varied. Nursing competency is known to directly correlate with increasing education, as nurses holding a master’s degree have been shown to outperform those with a bachelor of nursing degree.3
Increased formal education as well as increased work experience (> 5 years) are correlated with increased critical thinking ability.4,5 The critical thinking ability of health care providers can be fortified by clinical simulation, which leads to statistically significant improvement in clinical competency.2,3
A literature review of Medline and the National Library of Medicine PubMed online databases for search terms (nurs*, confidence, bedside, guide) was performed but did not result in original research assessing nurse confidence related to bedside guides. In this population, nurses were anonymously compared against their own historical data obviating any effect of education or experience on survey measures.
Nursing Self-Confidence
Evidence suggests that nursing confidence is a complex manifestation of the security felt within the care team and the comfort of one’s own professional abilities.4 Patients’ trust in the team caring for them is based on the confidence exuded by the team.8 In this way, nursing confidence can affect the patient-care team profoundly. Value is maximized when a nurse’s self-confidence engenders patient confidence and trust. Due to the varied patient load and complexity of subspecialty nursing care, it is hypothesized that bedside manuals/guidelines can be used to educate the subspecialty nurses on specific patient-related issues.
Nursing practice competence and confidence is vital to providing care for patients with complex postsurgical health care needs. Patient safety and outcome are paramount. This can be intimidating for newly qualified surgical subspecialty nurses who have not yet had experience with or adequate exposure to patients with complex postsurgical needs. Surgical nursing continuing education places an emphasis on adaptation to ever-changing specialized surgical procedures and postoperative patient care. Nevertheless, it is difficult for surgical subspecialty nurses to learn and retain all the possible complexities of individual cases and to confidently, appropriately, and safely care for patients especially when adverse events arise.
Recognizing that leadership is personal and not dependent on hierarchy, surgical subspecialty nurses may be better suited to specific bedside training and counseling.6,9 A key factor influencing nursing confidence is communication and collaboration with physicians.9 The role of the physician at VA medical facilities is no longer to be a commanding figure with complete medical autonomy; rather, a unified team of specialized practitioners collaboratively facilitate and deliver patient care.
There is no specific research detailing the use of bedside nursing guides in caring for postoperative patients. However, at VAPHCS, nurses created supplemental material regarding postoperative acute care of vascular surgery patients, which was found to be subjectively helpful in elevating nursing confidence. To the authors’ knowledge, no such supplemental information/guide exists for other specialty surgical services.
The surgical nursing guide created here detailed visuals of many common neurosurgical procedures performed at VAPHCS and included a prioritized checklist, which the 16 surgical subspecialty nurses could reference postoperatively. The authors hypothesized that this would enhance the nurses’ ability to efficiently manage specific situations while bridging communication gaps between surgical teams and nurses. The survey results agree with previous reports that suggested that the application of an adaptive leadership framework would empower nurses to deliver excellent patient-centered care, care that can be augmented with subspecialty nursing guides.7,10
Based on these results the authors propose that subspecialty surgical services consider use of a practical nursing guide for all surgical subspecialty nurses to reference, improve familiarity with procedures, and provide guidance to manage adverse events. Since implementing this reader-friendly paradigm within neurosurgical care, a nurse driven expansion has now included other subspecialty services at the VAPHCS with success.
Limitations
Survey responses have inherent bias and sampling error rates. The sample size for this survey was small. Data were grouped for data analysis. Competency and patient outcomes were not measured.
Future Directions
Despite specific surgical specialty postoperative patient care training, an overall lack of confidence can persist. A physician-created neurosurgical nursing guide that detailed the most common neurosurgical procedures, expected postoperative care, and potential emergencies was shown to improve nursing confidence. Collaborative (physician and nursing leaders) QI projects, such as described here; development of specific surgical specialty initiatives designed to improve confidence and quality; and nurse-physician communication and teamwork could lead to improved patient satisfaction and outcome.
The costs associated with developing and using bedside nursing guides are relatively low, and efficiency can be considered high. Competency improvement could be measured by creating a specialty-specific case scenario question bank. Effects on patient satisfaction and outcome could be measured by a patient satisfaction survey. Improvements in beside catastrophe management could be prospectively tracked; for example, rates of mismanagement of mobility status, emergent transfers to the intensive care unit, or poor wound care could be compared pre- and postfamiliarization with a subspecialty guide.
Conclusion
Familiarization with the VAPHCS neurosurgical nursing guide had a positive impact on the confidence of medical-surgical nurses caring for neurosurgical patients. Medical-surgical nurses were more comfortable taking care of neurosurgical patients; they felt the guide helped improve skills and noted improved knowledge regarding involvement of physician oversight. Although objective parameters were not assessed, improvement in nursing confidence in general leads to improved overall nurse-physician communication and patient management. A further study might target objective parameters associated with guide usage, such as changes in the number of emergencies or calls to physicians regarding management.
Acknowledgments
The authors thank Andy Rekito, MS, for illustrative assistance.
The VA Portland Healthcare System (VAPHCS) is a 277-bed facility that serves more than 85,000 inpatient and 880,000 outpatient visits each year from veterans in Oregon and southwestern Washington. The VAPHCS consists of a main tertiary care VAMC with an acute medical and surgical facility that includes 30 beds serving qualifying veterans. Supported surgical specialties include urology, general surgery, vascular surgery, otolaryngology, orthopedic surgery, ophthalmology, cardiothoracic surgery, transplant surgery, and neurological surgery. Neurosurgical patients account for about 12% to 13% of annual surgical patients. The VAPHCS also is partnered with Oregon Health & Science University in the training of health care professionals, such as physicians and nurses.
The expectation at the VAPHCS is that medical-surgical nurses care for 4 to 5 concurrent patients, often from different surgical services. Caring for patients with different medical and surgical needs, variable ambulatory, swallowing, and elimination functions, and different physician teams can become confusing; even within a single surgical service, postoperative care due to procedure complexity, specificity of care orders, and the real possibility of medical catastrophe can seem overwhelming. Therefore, subspecialty nursing training poses a challenge that requires technical in-service and didactic education and allocation of resources.
Despite systems level subspecialty nursing training, medical emergencies identified at the bedside can be mismanaged.1 Errors in care can be due to an incomplete knowledge of the patient’s procedure and misunderstanding of positioning and activity limitations.
To encourage medical-surgical nurses to become more engaged and confident in subspecialty patient care, the authors developed a bedside neurosurgical nursing guide to allow for independent procedure related education. The comprehensive guide summarized the clinical course for postoperative neurosurgical patients undergoing cranial and spinal surgeries. This guide included appropriate surgery-related images, procedure overviews, management decisions, potential postoperative complications, and wound care directions. The guide was distributed to medical-surgical nurses caring for neurosurgical patients. The authors hypothesized that the guide would enable nurses to better predict adverse outcomes and respond appropriately and would improve confidence in patient care.
Methods
For educational purposes, a bedside neurosurgical nursing guide (text and graphics) was created for the 16 surgical subspecialty nurses at the VAPHCS. The guide detailed the most common cranial and spinal neurosurgical procedures performed at VAPHCS and was written based on a typical postoperative course for each procedure by the chief neurosurgery resident at VAPHCS with collaboration from the attending neurosurgeons (Figure).
A quality improvement (QI) project was undertaken to assess nursing confidence with neurosurgical patients’ care pre- and postfamiliarity with the bedside neurosurgical nursing guide. A literature search revealed no validated survey assessing nursing confidence, so one was created using the Likert scale. Specifically, an anonymous 6-question survey was completed by all 16 surgical nurses prior to familiarization with the guide. Responses were recorded as scores of 1 to 5 for questions 1, 3, and 4, with a response of 1 indicative of no comfort or confidence and a response of 5 indicative of the highest level of comfort or confidence. Responses were recorded as either true or false for questions 2 and 6, and never, occasionally, frequently, or always for question 5.
The guide was made available to nurses for 6 months without encouragement to use it. After 6 months, a 3-week period of familiarization with and education about the availability of the guide was instituted at morning nursing reports; the total availability of the guide to nursing staff was 6 months 3 weeks. After this period the same 6-question survey was distributed, and data were collected.
Survey responses were categorized into 2 groups. Responses to questions 1, 3, and 4 were categorized as group 1, and responses to questions 4 and 5 were categorized as group 2. Responses (never and occasionally) to question 5, were categorized as group 1 and responses (frequently and always) as group 2 (Table). Responses to questions 2 and 6 were grouped 1 for true and 2 for false. Nurses participating in this study ranged in age from 22 to 57 years, education level ranged from registered nurse to a bachelor of science in nursing, and years of experience ranged from < 1 year to 27 years.
Statistics were calculated using chi-square analysis with Yates correction online calculator. For the chi-square analysis, the prefamiliarization data for groups 1 and 2 were used as the expected values, and the postfamiliarization data were used for the observed values. In this manner, differences were discerned between the before and after questionnaire responses. The VAPHCS institutional review board determined that the study was not human research and exempt from review.
Findings
Anonymous survey responses were collected from all 16 surgical subspecialty nurses both prior and after familiarization with the nursing guide.The response rate was 100% with only a few incomplete responses excluded from the analysis. Three questions in the prefamiliarization questionnaire had no appropriate response, and 1 question in the postfamiliarization survey had no appropriate response.
Improvement was statistically significant in responses for questions 1, 3, 5, and 6 (P = .026, .008, .004, and .033, respectively). No significant differences were found for questions 2 and 4 (P = .974 and .116, respectively). It is possible that there was no significant difference in question 2 because prefamiliarization responses were already favorable. Even if nurses did not feel comfortable taking care of neurosurgical patients (as assessed in question 1), they noted confidence improvement by working on the ward and through informal assimilation of knowledge and skill, which would have accumulated naturally over 1 year.
Prior to familiarization with the guide, 7 nurses did not feel confident in assessing the need to contact a physician (question 4). After familiarization with the nursing guide, favorable responses increased from 9 to 14 nurses. Results trended toward but did not reach statistical significance, likely due to the small sample size.
Ultimately, in the 16 surgical subspecialty nurses surveyed, familiarization with the nursing guide was shown to improve comfort in taking care of neurosurgical patients and increase confidence in patient care skills. At the end of the QI project (6 months, 3 weeks), all nurses knew where to locate the bedside neurosurgical nursing guide and were familiar with it and its use. The guide remains accessible to the medical-surgical nurses and continues to be used.
Discussion
Nursing confidence has an undervalued effect on patient care.2 Confidence, or a belief in one’s own ability, varies directly with competence. Systematic quantification of nursing competence has been extensively studied using self-report questionnaires and clinical simulations.2,3 Competency can be quantified and normalized using formal assessment; however, confidence is somewhat intangible. Nursing confidence is a situation-dependent subjective feeling of security and is derived from an internalized assessment of skills that are commensurate with patient needs. Nursing confidence is further influenced by an intuited value within the care team, adequate knowledge of the patient’s condition, and procedures and protocols.4
A similar but less specific definition deconstructs nursing confidence as “significance of a professional network of coworkers” and the “importance of confirmation of professional role and competence.”5 The professional network of coworkers is invaluable as it underlies the essence of patient-centered care. The adaptive leadership framework is integral to the modern delivery of patient care, and via this framework frontline clinical staff, including nurses, are empowered.6,7
The second portion of Haavardsholm and Nåden’s definition, “importance of confirmation of professional role and competence” describes the association of the most easily augmented correlate of confidence: competency.5 Nursing competency is supplemented continuously with in-service training and recertification processes; however, despite this, demands placed on nurses can be technologically advanced and extremely varied. Nursing competency is known to directly correlate with increasing education, as nurses holding a master’s degree have been shown to outperform those with a bachelor of nursing degree.3
Increased formal education as well as increased work experience (> 5 years) are correlated with increased critical thinking ability.4,5 The critical thinking ability of health care providers can be fortified by clinical simulation, which leads to statistically significant improvement in clinical competency.2,3
A literature review of Medline and the National Library of Medicine PubMed online databases for search terms (nurs*, confidence, bedside, guide) was performed but did not result in original research assessing nurse confidence related to bedside guides. In this population, nurses were anonymously compared against their own historical data obviating any effect of education or experience on survey measures.
Nursing Self-Confidence
Evidence suggests that nursing confidence is a complex manifestation of the security felt within the care team and the comfort of one’s own professional abilities.4 Patients’ trust in the team caring for them is based on the confidence exuded by the team.8 In this way, nursing confidence can affect the patient-care team profoundly. Value is maximized when a nurse’s self-confidence engenders patient confidence and trust. Due to the varied patient load and complexity of subspecialty nursing care, it is hypothesized that bedside manuals/guidelines can be used to educate the subspecialty nurses on specific patient-related issues.
Nursing practice competence and confidence is vital to providing care for patients with complex postsurgical health care needs. Patient safety and outcome are paramount. This can be intimidating for newly qualified surgical subspecialty nurses who have not yet had experience with or adequate exposure to patients with complex postsurgical needs. Surgical nursing continuing education places an emphasis on adaptation to ever-changing specialized surgical procedures and postoperative patient care. Nevertheless, it is difficult for surgical subspecialty nurses to learn and retain all the possible complexities of individual cases and to confidently, appropriately, and safely care for patients especially when adverse events arise.
Recognizing that leadership is personal and not dependent on hierarchy, surgical subspecialty nurses may be better suited to specific bedside training and counseling.6,9 A key factor influencing nursing confidence is communication and collaboration with physicians.9 The role of the physician at VA medical facilities is no longer to be a commanding figure with complete medical autonomy; rather, a unified team of specialized practitioners collaboratively facilitate and deliver patient care.
There is no specific research detailing the use of bedside nursing guides in caring for postoperative patients. However, at VAPHCS, nurses created supplemental material regarding postoperative acute care of vascular surgery patients, which was found to be subjectively helpful in elevating nursing confidence. To the authors’ knowledge, no such supplemental information/guide exists for other specialty surgical services.
The surgical nursing guide created here detailed visuals of many common neurosurgical procedures performed at VAPHCS and included a prioritized checklist, which the 16 surgical subspecialty nurses could reference postoperatively. The authors hypothesized that this would enhance the nurses’ ability to efficiently manage specific situations while bridging communication gaps between surgical teams and nurses. The survey results agree with previous reports that suggested that the application of an adaptive leadership framework would empower nurses to deliver excellent patient-centered care, care that can be augmented with subspecialty nursing guides.7,10
Based on these results the authors propose that subspecialty surgical services consider use of a practical nursing guide for all surgical subspecialty nurses to reference, improve familiarity with procedures, and provide guidance to manage adverse events. Since implementing this reader-friendly paradigm within neurosurgical care, a nurse driven expansion has now included other subspecialty services at the VAPHCS with success.
Limitations
Survey responses have inherent bias and sampling error rates. The sample size for this survey was small. Data were grouped for data analysis. Competency and patient outcomes were not measured.
Future Directions
Despite specific surgical specialty postoperative patient care training, an overall lack of confidence can persist. A physician-created neurosurgical nursing guide that detailed the most common neurosurgical procedures, expected postoperative care, and potential emergencies was shown to improve nursing confidence. Collaborative (physician and nursing leaders) QI projects, such as described here; development of specific surgical specialty initiatives designed to improve confidence and quality; and nurse-physician communication and teamwork could lead to improved patient satisfaction and outcome.
The costs associated with developing and using bedside nursing guides are relatively low, and efficiency can be considered high. Competency improvement could be measured by creating a specialty-specific case scenario question bank. Effects on patient satisfaction and outcome could be measured by a patient satisfaction survey. Improvements in beside catastrophe management could be prospectively tracked; for example, rates of mismanagement of mobility status, emergent transfers to the intensive care unit, or poor wound care could be compared pre- and postfamiliarization with a subspecialty guide.
Conclusion
Familiarization with the VAPHCS neurosurgical nursing guide had a positive impact on the confidence of medical-surgical nurses caring for neurosurgical patients. Medical-surgical nurses were more comfortable taking care of neurosurgical patients; they felt the guide helped improve skills and noted improved knowledge regarding involvement of physician oversight. Although objective parameters were not assessed, improvement in nursing confidence in general leads to improved overall nurse-physician communication and patient management. A further study might target objective parameters associated with guide usage, such as changes in the number of emergencies or calls to physicians regarding management.
Acknowledgments
The authors thank Andy Rekito, MS, for illustrative assistance.
1. Pusateri ME, Prior MM, Kiely SC. The role of the non-ICU staff nurse on a medical emergency team: perceptions and understanding. Am J Nurs. 2011;111(5):22-29, quiz 30-31.
2. Bambini D, Washburn J, Perkins R. Outcomes of clinical simulation for novice nursing students: communication, confidence, clinical judgment. Nurs Educ Perspect. 2009;30(2):79-82.
3. Chang MJ, Chang YJ, Kuo SH, Yang YH, Chou FH. Relationships between critical thinking ability and nursing competence in clinical nurses. J Clin Nurs. 2011;20(21-22):3224-3232.
4. Perry P. Concept analysis: confidence/self-confidence. Nurs Forum. 2011;46(4):218-230.
5. Haavardsholm I, Nåden D. The concept of confidence—the nurse’s perception. Eur J Cancer Care (Engl). 2009;18(5):483-491.
6. Bailey DE Jr, Docherty SL, Adams JA, et al. Studying the clinical encounter with the Adaptive Leadership framework. J Healthc Leadersh. 2012;2012(4):83-91.
7. Hall C, McCutcheon H, Deuter K, Matricciani L. Evaluating and improving a model of nursing care delivery: a process of partnership. Collegian. 2012;19(4):203-210.
8. Williams AM, Irurita VF. Therapeutic and non-therapeutic interpersonal interactions: the patient’s perspective. J Clin Nurs. 2004;13(7):806-815.
9. Carryer J. Collaboration between doctors and nurses. J Prim Health Care. 2011;3(1):77-79.
10. Chadwick MM. Creating order out of chaos: a leadership approach. AORN J. 2010;91(1):154-170.
1. Pusateri ME, Prior MM, Kiely SC. The role of the non-ICU staff nurse on a medical emergency team: perceptions and understanding. Am J Nurs. 2011;111(5):22-29, quiz 30-31.
2. Bambini D, Washburn J, Perkins R. Outcomes of clinical simulation for novice nursing students: communication, confidence, clinical judgment. Nurs Educ Perspect. 2009;30(2):79-82.
3. Chang MJ, Chang YJ, Kuo SH, Yang YH, Chou FH. Relationships between critical thinking ability and nursing competence in clinical nurses. J Clin Nurs. 2011;20(21-22):3224-3232.
4. Perry P. Concept analysis: confidence/self-confidence. Nurs Forum. 2011;46(4):218-230.
5. Haavardsholm I, Nåden D. The concept of confidence—the nurse’s perception. Eur J Cancer Care (Engl). 2009;18(5):483-491.
6. Bailey DE Jr, Docherty SL, Adams JA, et al. Studying the clinical encounter with the Adaptive Leadership framework. J Healthc Leadersh. 2012;2012(4):83-91.
7. Hall C, McCutcheon H, Deuter K, Matricciani L. Evaluating and improving a model of nursing care delivery: a process of partnership. Collegian. 2012;19(4):203-210.
8. Williams AM, Irurita VF. Therapeutic and non-therapeutic interpersonal interactions: the patient’s perspective. J Clin Nurs. 2004;13(7):806-815.
9. Carryer J. Collaboration between doctors and nurses. J Prim Health Care. 2011;3(1):77-79.
10. Chadwick MM. Creating order out of chaos: a leadership approach. AORN J. 2010;91(1):154-170.
Anterior Cervical Interbody Fusion Using a Polyetheretherketone (PEEK) Cage Device and Local Autograft Bone
Anterior cervical discectomy and fusion (ACDF) has been performed with various techniques and devices for many years. Autologous iliac crest grafts were initially used for the Cloward1,2 and Robinson and Smith3 techniques, but because of iliac crest graft site complications (eg, pain, infection, fracture, dystrophic scarring4,5), the procedure was generally superseded by allograft implants. These implants were then supplemented with anterior locking plate devices. More recently, unitary devices combining a polyetheretherketone (PEEK) spacer with screw or blade fixation have been developed, such as the Zero P (Synthes, Inc.) and the ROI-C cervical cage (LDR). Bone graft is required to fill the cavity of these devices and to promote osseous union. Demineralized bone matrix,6 tricalcium phosphate,7,8 and bone morphogenetic protein (BMP) have been used for these purposes, but they add expense to the procedure and have been associated with several complications (eg, neck swelling, dysphagia associated with BMP).9
Although multiple studies have demonstrated effective fusion rates and good outcomes for both iliac crest autograft and grafting/spacer constructs, the debate over cost and “added value” remains unresolved. One institution, which has published articles reviewing the spine literature and its own data, concluded that iliac crest autograft was the most cost-effective and consistently successful ACDF procedure.5,10
The VA Portland Health Care System (VAPORHCS) has analyzed the use of local autograft sources at the surgical site to circumvent the need to make a second incision at the iliac crest and, theoretically, to decrease risks and expenses associated with iliac crest autograft, allograft bone, and artificial constructs. Given the paucity of data on this method, the case series presented here represents one of a few studies that analyze local autograft for promotion of arthrodesis in a PEEK spacer device.
This article will report on the prospectively collected results of consecutive cases performed by Dr. Ross using a ROI-C cervical cage for 1-level anterior cervical discectomy between August 2011 and November 2014. This study received institutional review board approval.
Methods
Neck disability index (NDI) forms were used to assess the impact of neck pain on patients’ ability to manage in everyday life. The NDI form was completed before surgery and 3 and 9 months after surgery.
Dr. Ross preferred to perform minimally invasive posterior cervical foraminotomy for unilateral radiculopathy. Therefore, all patients with radiculopathy had bilateral symptoms or a symptomatic midline disc protrusion not accessible from a posterior approach. Standard techniques were used to make a left-side approach to the anterior cervical spine except in cases in which a previous right-side approach could be reused. Under the microscope, the anterior longitudinal ligament and annulus were incised, and the anterior contents of the disc space were removed with curettes and pituitary rongeurs. Care was taken to remove all cartilage from beneath the anterior inferior lip of the rostral vertebral body and to remove a few millimeters of the anterior longitudinal ligament from the rostral vertebral body without use of monopolar cautery (Figure 1). A 2 mm Kerrison punch then was used to remove the anterior inferior lip of the rostral vertebral body, and this bone was saved for grafting. No bone wax was used within the disc space.
After all disc space cartilage was removed from the endplates, additional bone was obtained from the uncovertebral joints and posterior vertebral bodies as the decompression proceeded posteriorly. Occasionally, distraction posts were used if the disc space was too narrow for optimal visualization posteriorly. After decompression was achieved, a lordotic ROI-C cervical cage was packed in its lumen with the bone chips and impacted into the disc space under fluoroscopic guidance. The blades were impacted under fluoroscopic guidance as well. The wound was closed with absorbable suture.
Antibiotics were given for no more than 24 hours after surgery. Ketorolac was used for analgesia the night of the surgery, and patients were asked to not use nonsteroidal anti-inflammatory drugs for 3 months after surgery. Lateral radiographs were obtained 3 and 9 months after surgery and every 6 months thereafter until arthrodesis was detected.
Results
Seventy-seven consecutive patients underwent 1-level anterior cervical discectomy (Table 1). Twenty-four procedures were performed for radiculopathy, 52 for myelopathy, and 1 for central cord injury sustained in a fall by a patient with preexisting spinal stenosis. Surgery was performed at C3-C4 (25 cases), C4-C5 (11 cases), C5-C6 (15 cases), and C6-C7 (1 case) for patients with myelopathy. Surgery was performed at C3-C4 (2 cases), C4-C5 (3 cases), C5-C6 (9 cases), and C6-C7 (10 cases) for patients with radiculopathy.
Twenty-eight patients reported presurgery tobacco use. Although all tobacco-using patients agreed to cease use in the perioperative period, at least 9 admitted to resuming tobacco use immediately after surgery. Eighteen patients had diabetes mellitus. In 2 patients, a diagnosis of osteoporosis was made with dual-energy X-ray absorptiometry. One patient was a chronic user of steroids before and after surgery. Mean body mass index (BMI) was 30.6, and 13 patients were morbidly obese (BMI > 34).
In 2 cases, only a single blade was placed. The second blade could not be placed because of broken adjacent screws (1 case) or undetermined reason (1 case).
The mean time for follow-up was 17 months (range 3-34). Four patients were lost to follow-up: 3 after the 1-month postoperative visit and 1 with severe psychiatric problems after hospital discharge.
There were no new neurologic deficits, no wound infections, and no recurrent laryngeal nerve palsies in the 77 patients. Eight months after surgery, 1 patient with radiculopathy underwent foraminotomy at the index level for persisting foraminal stenosis. Two patients whose myelopathic symptoms persisted after surgery returned for minimally invasive posterior laminotomy to remove infolded ligamentum flavum. The presurgery and 3- and 9-month postsurgery NDI scores were available for 52 patients (Table 2). Before surgery the mean NDI score was 24 (range 8-40). Three months postsurgery the mean NDI score was 15 (range 2-27) for patients with myelopathy and 13 (range 2-28) for patients with radiculopathy. The patient with the highest NDI score (28) stated that though all his symptoms were relieved, he had gauged his responses to protect his disability claim. Nine months after surgery, the mean NDI scores were 9.5 (range 5-17) for patients with myelopathy and 6 (range 2-13) for patients with radiculopathy. No NDI score was higher postsurgery than presurgery.
Arthrodesis was defined as bony bridging between the adjacent vertebral bodies and the bone graft within the lumen of the device, anterior to the device, or posterior to the device. In Dr. Ross’ protocol, computed tomography (CT) scans or flexion-extension radiographs were obtained only if pseudarthrosis was suspected to avoid unnecessary radiation exposure. Sixty-six patients had at least the 3-month radiography follow-up available. All 52 patients with 9-month follow-up data achieved complete arthrodesis, as determined by plain film radiography. Bridging ossification was found anterior to the device in all but 9 patients. Trabeculated bone was growing through the lumen of the device in all cases (Figure 2). A broken blade without clinical correlation was noted on imaging for 1 patient.
The total cost of the ROI-C cervical cage (LDR) for VAPORHCS was $3,498, or $1,749 for the PEEK spacer plus $1,749 for 2 metal blades. In comparison, the total cost of a typical anterior locking plate would have been $6,700, or $3,200 for the plate plus $2,000 for 4 screws and $1,500 for an allograft fibular spacer. Demineralized bone matrix (1 mL) as used in cervical arthrodesis by other surgeons at VAPORHCS cost $279, or about $500 including shipping.
DISCUSSION
Anterior cervical discectomy with fusion is a very common and successful surgical procedure for cervical myelopathy, radiculopathy, and degenerative disease that has failed to be corrected with conservative therapy.10 Medicare data documented a 206% increase in 1-level fusion procedures for degenerative spine pathology performed between 1992 and 2005.11 When a procedure is performed so often, it is appropriate to review methods and analyze efficacy, cost, and cost-effectiveness.
According to a 2007 meta-analysis, the fusion rates of 1-level ACDF arthrodesis at 1-year follow-up are 97.1% in patients treated with anterior plates and 92.1% in patients treated with noninstrumented fusion.12 The rate disparity was larger for multiple-level fusion: 50% to 82.5% for instrumented cases12,13 vs 3% to 42% for noninstrumented cases.14-16 Given the higher fusion rates achieved with instrumentation, surgeons have favored its use in ACDF.
Computed Tomography Use
Computed tomography has long been considered the gold standard for assessing arthrodesis outcomes (eg, Siambanes and Mather).17 However, recent data on potential harm caused by CT-related ionizing radiation suggest a need for caution with routine CT use.18,19 For cervical spine CT, Schonfeld and colleagues found that the risk for excess thyroid cancers ranged from 1 to 33 cases per 10,000 CT scans.20 According to another report, “limiting neck CT scanning to a higher risk group would increase the gap between benefit and harm, whereas performing CT routinely on low-risk cases approaches a point where its harm equals or exceeds its benefit.”19 As some have questioned even routinepostoperative use of radiation in patients with unremarkable clinical courses—patients should be spared unnecessary exposure—CT scans or flexion-extensionradiographs were obtained at VAPORHCS only if clinical symptoms or radiographs were suggestive of pseudarthrosis.21 As none of the VAPORHCS patients had those symptoms, none underwent postoperative CT.
For anterior cervical arthrodesis, surgeon preference determines which of many different bone substrates can be used with instrumentation, which impacts the costs. Fusion substrates include structural autografts, structural allografts, morselized autografts, morselized allografts, demineralized allografts, porous ceramics and metals, and BMP. Given these many options, studies comparing the constructs are lacking, especially with regard to the cost of alternative fusion constructs that produce similar outcomes. The Centers for Disease Control and Prevention defines cost-benefit analysis as a “type of economic evaluation that measures both costs and benefits (ie, negative and positive consequences) associated with an intervention in dollar terms.”22 It has been reported that using iliac crest autografts with anterior plate instrumentation is the most cost-effective method, yet alternatives remain in use.5,10
For ACDF, iliac crest bone is an ideal and widely used construct substrate. Structural grafts harvested from the crest provide significant stability due to their bicortical or tricortical configuration with interposed osteoinductive and osteogenic cancellous bone. Few graft complications (eg, graft resorption) and no immunogenic or infectious complications have been reported for iliac crest bone. However, autologous iliac crest increases operative time, and donor-site morbidity has been reported.23,24 A retrospective questionnaire-based investigation by Silber and colleagues, who evaluated iliac crest bone graft site morbidity in 1-level ACDF, found that 26.1% of patients had pain at the iliac crest harvest site, and 15.7% had numbness.24 Other complications, which occurred at lower rates, were bruising, hematoma, pelvic fracture, and poor cosmesis.23,25 In addition, osteoporosis and comorbid conditions have made it a challenge to acquire iliac crest autograft, contributing to the popularity of alternative substrates.25
Allografts
An alternative to autografts, allografts have the advantages of reduced operative time and reduced donor-site morbidity.26 Major historical concerns with allografts have included risk for disease transmission, costs associated with sterilization and serologic screening of grafts, and lack of oversight, leading to human allografts being acquired from dubious sources and ending up in the operating room.27,28 Two major types of allografts are available: mineralized and demineralized.
Arthrodesis rates are inferior for mineralized (structural) allografts with instrumentation than for autografts with instrumentation.29 In addition, smoking and other comorbidities have influenced fusion rates more in allograft than autograft fusions.30-33 However, allografts are being widely used because they avoid the donor-site morbidity associated with autografts and because they are load bearing, can provide structural stability and an osteoconductive matrix, and can be used off the shelf without adding much time to surgery.
Demineralized matrix substrates are commercial osteoconductive and osteoinductive biomaterials approved for filling bone gaps and extending graft when combined with autograft.7,8 Despite their osteoinductive properties, these substrates have had a high degree of product inconsistency, in some cases leading to poor outcomes.34 The lack of randomized studies with these constructs has made the determination of clear indications a challenge.
The initial enthusiasm over use of BMP, another bone-graft substitute for cervical fusion, was curtailed by reports of adverse events (AEs). Effective in anterior lumbar spine fusions, BMP was adapted to off-label use in the cervical spine a few years ago.35 Initial studies by Baskin and colleagues and Bishop and colleagues showed its fusion rates superior to those of allograft.31,32 Both studies reported no significant AEs. However, studies by Dickerman and colleagues and Smucker and colleagues demonstrated increased soft-tissue swelling leading to dysphagia and prolonged hospitalization, which were attributed to higher dosage (no study has identified a precise dose for individual patients).36,37 In addition, the cost of BMP is higher than that of any other bone-graft option for ACDF.3 Osteolysis has also been reported with BMP use.38-40 Carragee and colleagues highlighted the potential carcinogenicity of BMP, but this finding was not corroborated by Lad and colleagues.41,42
Cost Considerations
In addition to surgical effectiveness, spine surgical device costs have come under increased scrutiny.43-45 In 2012, plates were reported to cost (without overhead or profit margin to hospitals) between $1,015 and $3,601, and allograft spacers were estimated to cost between $1,220 and $3,640, cage costs ranged from $1,942 to $4,347, and PEEK spacers cost from $4,930 to $5,246.5 Individual surgeon instrumentation costs varied 10-fold based on the fusion constructs used.5
In a cost-effectiveness review of anterior cervical techniques, cage alone was the least expensive technique, disc arthroplasty or cage/plate/bone substitute groups were the next most expensive, and autograft alone was the most expensive option due to hip graft site morbidity.43 In another study, operative time associated with harvesting an iliac crest graft was equivalent in cost to that of an interbody cage.44 Other studies have compared the costs of various anterior cervical fusion constructs.9,10,45,46 A limitation of these studies is that autologous bone often refers to iliac crest grafts rather than local autograft. Epstein reviewed data from these studies and concluded, “ACDF using dynamic plates and autografts are the most cost effective treatment for anterior cervical discectomy,” citing a cost of $1,015 for this construct.5 Although Epstein demonstrated the cost-effectiveness of autograft in an individual surgeon’s hands, the results also are significant in that the studies identified areas in which improvements can be made at other institutions. The ROI-C cervical cage and local autograft bone cost that the authors report is at the lower end of the range reported by Epstein.5
Device explant rates also can be a concern. Operative waste was well described in a retrospective analysis of 87 ACDF procedures.47 The study found that the cost of explanting devices implanted during the same intraoperative period was equivalent to 9.2% of the cost of permanently implanted constructs. Epstein addressed operative waste by using educational modules to evaluate spine surgeons’ decision making before and after education. After the intervention, the institution noted a marked decline in costs related to explanted devices—from 20% in 2010 (before education) to 5.8% of the total cost of implanted devices in 2010 (after education).5
In the present study, the authors demonstrated that use of local morselized autograft with a PEEK spacer for 1-level ACDF had excellent arthrodesis rates and minimal complications. Of the 52 patients with 9 month postoperative data, all achieved arthrodesis regardless of tobacco use. This method compares favorably with other fusion options in terms of radiographic arthrodesis rates. In addition, it avoids the donor-site morbidity associated with autografts from an iliac site but maintains the benefits of the osteogenic, osteoconductive, and osteoinductive properties of autograft bone. Use of local autograft avoids the costs associated with iliac crest autograft, including increased operating and anesthesia time, additional operating room supplies (drapes, sutures, etc) needed for operating at a second site, and prolonged hospital stay due to pain at the donor site. Use of local autograft also obviates complications at a second surgical site; purchase, storage, and sterilization of allograft; and the neck swelling, possible carcinogenicity, and cost of purchase of BMP. Other than the occasional reuse of distraction posts, this method involves no other expensive explant supplies.
Autografts have osteogenic, osteoconductive, and osteoinductive properties, and autograft fusion rates are generally superior to allograft fusion rates. Bone morphogenetic protein fusion rates may be comparable to autograft fusion rates.9,26,32 Shortcomings of iliac crest autografts include increased operative time, blood loss, and donor-site morbidity. Allografts are osteoconductive and osteoinductive, but their fusion rates are inferior to those of iliac crest autografts. Other shortcomings are infection transmission and immunogenicity risks, higher graft resorption and collapse rates, cost, and previous issues relating to provenance. Bone morphogenetic protein is the most osteoinductive material with fusion rates similar to those of autograft, but its use is associated with neck swelling, dysphagia, osteolysis, potential carcinogenicity, and high cost.9
Conclusion
Overall, use of local autograft with a PEEK spacer has all the advantages of iliac crest autograft along with the benefit of working within the same operative window as the ACDF, thus reducing the infection, bleeding, and pain risks that may be encountered with a second incision. This procedure is effective, inexpensive, and cost-effective compared with alternatives and may be preferable for 1-level ACDF. In a population of patients with high rates of tobacco use, diabetes mellitus, obesity, and other factors that negatively affect fusion rates, local autograft may be a good choice for efficacy and cost savings.
Acknowledgments
The authors thank Shirley McCartney, PhD, for editorial assistance and Andy Rekito, MS, for illustrative assistance.
1. Cloward RB. The anterior approach for removal of ruptured cervical disks. 1958. J Neurosurg Spine. 2007;6(5):496-511.
2. Cloward RB. The anterior approach for removal of ruptured cervical disks. J Neurosurg. 1958;15(6):602-617.
3. Robinson RA, Smith GW. Anterolateral cervical disc removal and interbody fusion for cervical disc syndrome. SAS J. 2010;4(1):34-35.
4. Dimitriou R, Mataliotakis GI, Angoules AG, Kanakaris NK, Giannoudis PV. Complications following autologous bone graft harvesting from the iliac crest and using the RIA: a systematic review. Injury. 2011;42(suppl 2):S3-S15.
5. Epstein NE. Iliac crest autograft versus alternative constructs for anterior cervical spine surgery: pros, cons, and costs. Surg Neurol Int. 2012;3(suppl 3):S143-S156.
6. Gruskin E, Doll BA, Futrell FW, Schmitz JP, Hollinger JO. Demineralized bone matrix in bone repair: history and use. Adv Drug Deliv Rev. 2012;64(12):1063-1077.
7. Becker S, Maissen O, Ponomarev I, Stoll T, Rahn B, Wilke I. Osteopromotion by a beta-tricalcium phosphate/bone marrow hybrid implant for use in spine surgery. Spine (Phila Pa 1976). 2006;31(1):11-17.
8. Muschik M, Ludwig R, Halbhübner S, Bursche K, Stoll T. Beta-tricalcium phosphate as a bone substitute for dorsal spinal fusion in adolescent idiopathic scoliosis: preliminary results of a prospective clinical study. Eur Spine J. 2001;10(suppl 2):S178-S184.
9. Buttermann GR. Prospective nonrandomized comparison of an allograft with bone morphogenic protein versus an iliac-crest autograft in anterior cervical discectomy and fusion. Spine J. 2008;8(3):426-435.
10. Epstein NE. Efficacy and outcomes of dynamic-plated single-level anterior diskectomy/fusion with additional analysis of comparative costs. Surg Neurol Int. 2011;2:9.
11. Wang MC, Kreuter W, Wolfla CE, Maiman DJ, Deyo RA. Trends and variations in cervical spine surgery in the United States: Medicare beneficiaries, 1992 to 2005. Spine (Phila Pa 1976). 2009;34(9):955-961.
12. Fraser JF, Härtl R. Anterior approaches to fusion of the cervical spine: a metaanalysis of fusion rates. J Neurosurg Spine. 2007;6(4):298-303.
13. Nirala AP, Husain M, Vatsal DK. A retrospective study of multiple interbody grafting and long segment strut grafting following multilevel anterior cervical decompression. Br J Neurosurg. 2004;18(3):227-232.
14. Bohlman HH, Emery SE, Goodfellow DB, Jones PK. Robinson anterior cervical discectomy and arthrodesis for cervical radiculopathy. Long-term follow-up of one hundred and twenty-two patients. J Bone Joint Surg Am. 1993;75(9):1298-1307.
15. Cauthen JC, Kinard RE, Vogler JB, et al. Outcome analysis of noninstrumented anterior cervical discectomy and interbody fusion in 348 patients. Spine (Phila Pa 1976). 1998;23(2):188-192.
16. Emery SE, Fisher JR, Bohlman HH. Three-level anterior cervical discectomy and fusion: radiographic and clinical results. Spine (Phila Pa 1976). 1997;22(22):2622-2624.
17. Siambanes D, Mather S. Comparison of plain radiographs and CT scans in instrumented posterior lumbar interbody fusion. Orthopedics. 1998;21(2):165-167.
18. Berrington de González A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169(22):2071-2077.
19. Hikino K, Yamamoto LG. The benefit of neck computed tomography compared with its harm (risk of cancer). J Trauma Acute Care Surg. 2015;78(1):126-131.
20. Schonfeld SJ, Lee C, Berrington de González A. Medical exposure to radiation and thyroid cancer. Clin Oncol (R Coll Radiol). 2011;23(4):244-250.
21. Bartels RH, Beems T, Schutte PJ, Verbeek AL. The rationale of postoperative radiographs after cervical anterior discectomy with stand-alone cage for radicular pain. J Neurosurg Spine. 2010;12(3):275-279.
22. Centers for Disease Control and Prevention. The different types of health assessments. Centers for Disease Control and Prevention website. http://www.cdc.gov/healthyplaces/types_health_assessments.htm. Updated July 25, 2012. Accessed April 8, 2016.
23. Schnee CL, Freese A, Weil RJ, Marcotte PJ. Analysis of harvest morbidity and radiographic outcome using autograft for anterior cervical fusion. Spine (Phila Pa 1976). 1997;22(19):2222-2227.
24. Silber JS, Anderson DG, Daffner SD, et al. Donor site morbidity after anterior iliac crest bone harvest for single-level anterior cervical discectomy and fusion. Spine (Phila Pa 1976). 2003;28(2):134-139.
25. Seiler JG 3rd, Johnson J. Iliac crest autogenous bone grafting: donor site complications. J South Orthop Assoc. 2000;9(2):91-97.
26. Floyd T, Ohnmeiss D. A meta-analysis of autograft versus allograft in anterior cervical fusion. Eur Spine J. 2000;9(5):398-403.
27. Delloye C, Cornu O, Druez V, Barbier O. Bone allografts: what they can offer and what they cannot. J Bone Joint Surg Br. 2007;89(5):574-579.
28. Armour S. Illegal trade in bodies shakes loved ones. USA Today. http://usatoday30.usatoday.com/money/2006-04-26-body-parts-cover-usat_x.htm. Updated April 28, 2006. Accessed April 6, 2016.
29. Wigfield CC, Nelson RJ. Nonautologous interbody fusion materials in cervical spine surgery: how strong is the evidence to justify their use? Spine (Phila Pa 1976). 2001;26(6):687-694.
30. Bärlocher CB, Barth A, Krauss JK, Binggeli R, Seiler RW. Comparative evaluation of microdiscectomy only, autograft fusion, polymethylmethacrylate interposition, and threaded titanium cage fusion for treatment of single-level cervical disc disease: a prospective randomized study in 125 patients. Neurosurg Focus. 2002;12(1):E4.
31. Baskin DS, Ryan P, Sonntag V, Westmark R, Widmayer MA. A prospective, randomized, controlled cervical fusion study using recombinant human bone morphogenetic protein-2 with the CORNERSTONE-SR allograft ring and the ATLANTIS anterior cervical plate. Spine (Phila Pa 1976). 2003;28(12):1219-1224.
32. Bishop RC, Moore KA, Hadley MN. Anterior cervical interbody fusion using autogeneic and allogeneic bone graft substrate: a prospective comparative analysis. J Neurosurg. 1996;85(2):206-210.
33. Martin GJ Jr, Haid RW Jr, MacMillan M, Rodts GE Jr, Berkman R. Anterior cervical discectomy with freeze-dried fibula allograft. Overview of 317 cases and literature review. Spine (Phila Pa 1976). 1999;24(9):852-858.
34. Bae HW, Zhao L, Kanim LE, Wong P, Delamarter RB, Dawson EG. Intervariability and intravariability of bone morphogenetic proteins in commercially available demineralized bone matrix products. Spine (Phila Pa 1976). 2006;31(12):1299-1306.
35. Burkus JK, Gornet MF, Dickman CA, Zdeblick TA. Anterior lumbar interbody fusion using rhBMP-2 with tapered interbody cages. J Spinal Disord Tech. 2002;15(5):337-349.
36. Dickerman RD, Reynolds AS, Morgan BC, Tompkins J, Cattorini J, Bennett M. rh-BMP-2 can be used safely in the cervical spine: dose and containment are the keys! Spine J. 2007;7(4):508-509.
37. Smucker JD, Rhee JM, Singh K, Yoon ST, Heller JG. Increased swelling complications associated with off-label usage of rhBMP-2 in the anterior cervical spine. Spine (Phila Pa 1976). 2006;31(24):2813-2819.
38. Vaidya R, Carp J, Sethi A, Bartol S, Craig J, Les CM. Complications of anterior cervical discectomy and fusion using recombinant human bone morphogenetic protein-2. Eur Spine J. 2007;16(8):1257-1265.
39. Vaidya R, Sethi A, Bartol S, Jacobson M, Coe C, Craig JG. Complications in the use of rhBMP-2 in PEEK cages for interbody spinal fusions. J Spinal Disord Tech. 2008;21(8):557-562.
40. Knox JB, Dai JM 3rd, Orchowski J. Osteolysis in transforaminal lumbar interbody fusion with bone morphogenetic protein-2. Spine (Phila Pa 1976). 2011;36(8):672-676.
41. Carragee EJ, Chu G, Rohatgi R, et al. Cancer risk after use of recombinant bone morphogenetic protein-2 for spinal arthrodesis. J Bone Joint Surg Am. 2013;95(17):1537-1545.
42. Lad SP, Bagley JH, Karikari IO, et al. Cancer after spinal fusion: the role of bone morphogenetic protein. Neurosurgery. 2013;73(3):440-449.
43. Bhadra AK, Raman AS, Casey AT, Crawford RJ. Single-level cervical radiculopathy: clinical outcome and cost-effectiveness of four techniques of anterior cervical discectomy and fusion and disc arthroplasty. Eur Spine J. 2009;18(2):232-237.
44. Castro FP Jr, Holt RT, Majd M, Whitecloud TS 3rd. A cost analysis of two anterior cervical fusion procedures. J Spinal Disord. 2000;13(6):511-514.
45. Kandziora F, Pflugmacher R, Scholz M, et al. Treatment of traumatic cervical spine instability with interbody fusion cages: a prospective controlled study with a 2-year follow-up. Injury. 2005;36(suppl 2):B27-B35.
46. Vaidya R, Weir R, Sethi A, Meisterling S, Hakeos W, Wybo CD. Interbody fusion with allograft and rhBMP-2 leads to consistent fusion but early subsidence. J Bone Joint Surg Br. 2007;89(3):342-345.
47. Epstein NE, Schwall GS, Hood DC. The incidence and cost of devices explanted during single-level anterior diskectomy/fusions. Surg Neurol Int. 2011;2:23.
Anterior cervical discectomy and fusion (ACDF) has been performed with various techniques and devices for many years. Autologous iliac crest grafts were initially used for the Cloward1,2 and Robinson and Smith3 techniques, but because of iliac crest graft site complications (eg, pain, infection, fracture, dystrophic scarring4,5), the procedure was generally superseded by allograft implants. These implants were then supplemented with anterior locking plate devices. More recently, unitary devices combining a polyetheretherketone (PEEK) spacer with screw or blade fixation have been developed, such as the Zero P (Synthes, Inc.) and the ROI-C cervical cage (LDR). Bone graft is required to fill the cavity of these devices and to promote osseous union. Demineralized bone matrix,6 tricalcium phosphate,7,8 and bone morphogenetic protein (BMP) have been used for these purposes, but they add expense to the procedure and have been associated with several complications (eg, neck swelling, dysphagia associated with BMP).9
Although multiple studies have demonstrated effective fusion rates and good outcomes for both iliac crest autograft and grafting/spacer constructs, the debate over cost and “added value” remains unresolved. One institution, which has published articles reviewing the spine literature and its own data, concluded that iliac crest autograft was the most cost-effective and consistently successful ACDF procedure.5,10
The VA Portland Health Care System (VAPORHCS) has analyzed the use of local autograft sources at the surgical site to circumvent the need to make a second incision at the iliac crest and, theoretically, to decrease risks and expenses associated with iliac crest autograft, allograft bone, and artificial constructs. Given the paucity of data on this method, the case series presented here represents one of a few studies that analyze local autograft for promotion of arthrodesis in a PEEK spacer device.
This article will report on the prospectively collected results of consecutive cases performed by Dr. Ross using a ROI-C cervical cage for 1-level anterior cervical discectomy between August 2011 and November 2014. This study received institutional review board approval.
Methods
Neck disability index (NDI) forms were used to assess the impact of neck pain on patients’ ability to manage in everyday life. The NDI form was completed before surgery and 3 and 9 months after surgery.
Dr. Ross preferred to perform minimally invasive posterior cervical foraminotomy for unilateral radiculopathy. Therefore, all patients with radiculopathy had bilateral symptoms or a symptomatic midline disc protrusion not accessible from a posterior approach. Standard techniques were used to make a left-side approach to the anterior cervical spine except in cases in which a previous right-side approach could be reused. Under the microscope, the anterior longitudinal ligament and annulus were incised, and the anterior contents of the disc space were removed with curettes and pituitary rongeurs. Care was taken to remove all cartilage from beneath the anterior inferior lip of the rostral vertebral body and to remove a few millimeters of the anterior longitudinal ligament from the rostral vertebral body without use of monopolar cautery (Figure 1). A 2 mm Kerrison punch then was used to remove the anterior inferior lip of the rostral vertebral body, and this bone was saved for grafting. No bone wax was used within the disc space.
After all disc space cartilage was removed from the endplates, additional bone was obtained from the uncovertebral joints and posterior vertebral bodies as the decompression proceeded posteriorly. Occasionally, distraction posts were used if the disc space was too narrow for optimal visualization posteriorly. After decompression was achieved, a lordotic ROI-C cervical cage was packed in its lumen with the bone chips and impacted into the disc space under fluoroscopic guidance. The blades were impacted under fluoroscopic guidance as well. The wound was closed with absorbable suture.
Antibiotics were given for no more than 24 hours after surgery. Ketorolac was used for analgesia the night of the surgery, and patients were asked to not use nonsteroidal anti-inflammatory drugs for 3 months after surgery. Lateral radiographs were obtained 3 and 9 months after surgery and every 6 months thereafter until arthrodesis was detected.
Results
Seventy-seven consecutive patients underwent 1-level anterior cervical discectomy (Table 1). Twenty-four procedures were performed for radiculopathy, 52 for myelopathy, and 1 for central cord injury sustained in a fall by a patient with preexisting spinal stenosis. Surgery was performed at C3-C4 (25 cases), C4-C5 (11 cases), C5-C6 (15 cases), and C6-C7 (1 case) for patients with myelopathy. Surgery was performed at C3-C4 (2 cases), C4-C5 (3 cases), C5-C6 (9 cases), and C6-C7 (10 cases) for patients with radiculopathy.
Twenty-eight patients reported presurgery tobacco use. Although all tobacco-using patients agreed to cease use in the perioperative period, at least 9 admitted to resuming tobacco use immediately after surgery. Eighteen patients had diabetes mellitus. In 2 patients, a diagnosis of osteoporosis was made with dual-energy X-ray absorptiometry. One patient was a chronic user of steroids before and after surgery. Mean body mass index (BMI) was 30.6, and 13 patients were morbidly obese (BMI > 34).
In 2 cases, only a single blade was placed. The second blade could not be placed because of broken adjacent screws (1 case) or undetermined reason (1 case).
The mean time for follow-up was 17 months (range 3-34). Four patients were lost to follow-up: 3 after the 1-month postoperative visit and 1 with severe psychiatric problems after hospital discharge.
There were no new neurologic deficits, no wound infections, and no recurrent laryngeal nerve palsies in the 77 patients. Eight months after surgery, 1 patient with radiculopathy underwent foraminotomy at the index level for persisting foraminal stenosis. Two patients whose myelopathic symptoms persisted after surgery returned for minimally invasive posterior laminotomy to remove infolded ligamentum flavum. The presurgery and 3- and 9-month postsurgery NDI scores were available for 52 patients (Table 2). Before surgery the mean NDI score was 24 (range 8-40). Three months postsurgery the mean NDI score was 15 (range 2-27) for patients with myelopathy and 13 (range 2-28) for patients with radiculopathy. The patient with the highest NDI score (28) stated that though all his symptoms were relieved, he had gauged his responses to protect his disability claim. Nine months after surgery, the mean NDI scores were 9.5 (range 5-17) for patients with myelopathy and 6 (range 2-13) for patients with radiculopathy. No NDI score was higher postsurgery than presurgery.
Arthrodesis was defined as bony bridging between the adjacent vertebral bodies and the bone graft within the lumen of the device, anterior to the device, or posterior to the device. In Dr. Ross’ protocol, computed tomography (CT) scans or flexion-extension radiographs were obtained only if pseudarthrosis was suspected to avoid unnecessary radiation exposure. Sixty-six patients had at least the 3-month radiography follow-up available. All 52 patients with 9-month follow-up data achieved complete arthrodesis, as determined by plain film radiography. Bridging ossification was found anterior to the device in all but 9 patients. Trabeculated bone was growing through the lumen of the device in all cases (Figure 2). A broken blade without clinical correlation was noted on imaging for 1 patient.
The total cost of the ROI-C cervical cage (LDR) for VAPORHCS was $3,498, or $1,749 for the PEEK spacer plus $1,749 for 2 metal blades. In comparison, the total cost of a typical anterior locking plate would have been $6,700, or $3,200 for the plate plus $2,000 for 4 screws and $1,500 for an allograft fibular spacer. Demineralized bone matrix (1 mL) as used in cervical arthrodesis by other surgeons at VAPORHCS cost $279, or about $500 including shipping.
DISCUSSION
Anterior cervical discectomy with fusion is a very common and successful surgical procedure for cervical myelopathy, radiculopathy, and degenerative disease that has failed to be corrected with conservative therapy.10 Medicare data documented a 206% increase in 1-level fusion procedures for degenerative spine pathology performed between 1992 and 2005.11 When a procedure is performed so often, it is appropriate to review methods and analyze efficacy, cost, and cost-effectiveness.
According to a 2007 meta-analysis, the fusion rates of 1-level ACDF arthrodesis at 1-year follow-up are 97.1% in patients treated with anterior plates and 92.1% in patients treated with noninstrumented fusion.12 The rate disparity was larger for multiple-level fusion: 50% to 82.5% for instrumented cases12,13 vs 3% to 42% for noninstrumented cases.14-16 Given the higher fusion rates achieved with instrumentation, surgeons have favored its use in ACDF.
Computed Tomography Use
Computed tomography has long been considered the gold standard for assessing arthrodesis outcomes (eg, Siambanes and Mather).17 However, recent data on potential harm caused by CT-related ionizing radiation suggest a need for caution with routine CT use.18,19 For cervical spine CT, Schonfeld and colleagues found that the risk for excess thyroid cancers ranged from 1 to 33 cases per 10,000 CT scans.20 According to another report, “limiting neck CT scanning to a higher risk group would increase the gap between benefit and harm, whereas performing CT routinely on low-risk cases approaches a point where its harm equals or exceeds its benefit.”19 As some have questioned even routinepostoperative use of radiation in patients with unremarkable clinical courses—patients should be spared unnecessary exposure—CT scans or flexion-extensionradiographs were obtained at VAPORHCS only if clinical symptoms or radiographs were suggestive of pseudarthrosis.21 As none of the VAPORHCS patients had those symptoms, none underwent postoperative CT.
For anterior cervical arthrodesis, surgeon preference determines which of many different bone substrates can be used with instrumentation, which impacts the costs. Fusion substrates include structural autografts, structural allografts, morselized autografts, morselized allografts, demineralized allografts, porous ceramics and metals, and BMP. Given these many options, studies comparing the constructs are lacking, especially with regard to the cost of alternative fusion constructs that produce similar outcomes. The Centers for Disease Control and Prevention defines cost-benefit analysis as a “type of economic evaluation that measures both costs and benefits (ie, negative and positive consequences) associated with an intervention in dollar terms.”22 It has been reported that using iliac crest autografts with anterior plate instrumentation is the most cost-effective method, yet alternatives remain in use.5,10
For ACDF, iliac crest bone is an ideal and widely used construct substrate. Structural grafts harvested from the crest provide significant stability due to their bicortical or tricortical configuration with interposed osteoinductive and osteogenic cancellous bone. Few graft complications (eg, graft resorption) and no immunogenic or infectious complications have been reported for iliac crest bone. However, autologous iliac crest increases operative time, and donor-site morbidity has been reported.23,24 A retrospective questionnaire-based investigation by Silber and colleagues, who evaluated iliac crest bone graft site morbidity in 1-level ACDF, found that 26.1% of patients had pain at the iliac crest harvest site, and 15.7% had numbness.24 Other complications, which occurred at lower rates, were bruising, hematoma, pelvic fracture, and poor cosmesis.23,25 In addition, osteoporosis and comorbid conditions have made it a challenge to acquire iliac crest autograft, contributing to the popularity of alternative substrates.25
Allografts
An alternative to autografts, allografts have the advantages of reduced operative time and reduced donor-site morbidity.26 Major historical concerns with allografts have included risk for disease transmission, costs associated with sterilization and serologic screening of grafts, and lack of oversight, leading to human allografts being acquired from dubious sources and ending up in the operating room.27,28 Two major types of allografts are available: mineralized and demineralized.
Arthrodesis rates are inferior for mineralized (structural) allografts with instrumentation than for autografts with instrumentation.29 In addition, smoking and other comorbidities have influenced fusion rates more in allograft than autograft fusions.30-33 However, allografts are being widely used because they avoid the donor-site morbidity associated with autografts and because they are load bearing, can provide structural stability and an osteoconductive matrix, and can be used off the shelf without adding much time to surgery.
Demineralized matrix substrates are commercial osteoconductive and osteoinductive biomaterials approved for filling bone gaps and extending graft when combined with autograft.7,8 Despite their osteoinductive properties, these substrates have had a high degree of product inconsistency, in some cases leading to poor outcomes.34 The lack of randomized studies with these constructs has made the determination of clear indications a challenge.
The initial enthusiasm over use of BMP, another bone-graft substitute for cervical fusion, was curtailed by reports of adverse events (AEs). Effective in anterior lumbar spine fusions, BMP was adapted to off-label use in the cervical spine a few years ago.35 Initial studies by Baskin and colleagues and Bishop and colleagues showed its fusion rates superior to those of allograft.31,32 Both studies reported no significant AEs. However, studies by Dickerman and colleagues and Smucker and colleagues demonstrated increased soft-tissue swelling leading to dysphagia and prolonged hospitalization, which were attributed to higher dosage (no study has identified a precise dose for individual patients).36,37 In addition, the cost of BMP is higher than that of any other bone-graft option for ACDF.3 Osteolysis has also been reported with BMP use.38-40 Carragee and colleagues highlighted the potential carcinogenicity of BMP, but this finding was not corroborated by Lad and colleagues.41,42
Cost Considerations
In addition to surgical effectiveness, spine surgical device costs have come under increased scrutiny.43-45 In 2012, plates were reported to cost (without overhead or profit margin to hospitals) between $1,015 and $3,601, and allograft spacers were estimated to cost between $1,220 and $3,640, cage costs ranged from $1,942 to $4,347, and PEEK spacers cost from $4,930 to $5,246.5 Individual surgeon instrumentation costs varied 10-fold based on the fusion constructs used.5
In a cost-effectiveness review of anterior cervical techniques, cage alone was the least expensive technique, disc arthroplasty or cage/plate/bone substitute groups were the next most expensive, and autograft alone was the most expensive option due to hip graft site morbidity.43 In another study, operative time associated with harvesting an iliac crest graft was equivalent in cost to that of an interbody cage.44 Other studies have compared the costs of various anterior cervical fusion constructs.9,10,45,46 A limitation of these studies is that autologous bone often refers to iliac crest grafts rather than local autograft. Epstein reviewed data from these studies and concluded, “ACDF using dynamic plates and autografts are the most cost effective treatment for anterior cervical discectomy,” citing a cost of $1,015 for this construct.5 Although Epstein demonstrated the cost-effectiveness of autograft in an individual surgeon’s hands, the results also are significant in that the studies identified areas in which improvements can be made at other institutions. The ROI-C cervical cage and local autograft bone cost that the authors report is at the lower end of the range reported by Epstein.5
Device explant rates also can be a concern. Operative waste was well described in a retrospective analysis of 87 ACDF procedures.47 The study found that the cost of explanting devices implanted during the same intraoperative period was equivalent to 9.2% of the cost of permanently implanted constructs. Epstein addressed operative waste by using educational modules to evaluate spine surgeons’ decision making before and after education. After the intervention, the institution noted a marked decline in costs related to explanted devices—from 20% in 2010 (before education) to 5.8% of the total cost of implanted devices in 2010 (after education).5
In the present study, the authors demonstrated that use of local morselized autograft with a PEEK spacer for 1-level ACDF had excellent arthrodesis rates and minimal complications. Of the 52 patients with 9 month postoperative data, all achieved arthrodesis regardless of tobacco use. This method compares favorably with other fusion options in terms of radiographic arthrodesis rates. In addition, it avoids the donor-site morbidity associated with autografts from an iliac site but maintains the benefits of the osteogenic, osteoconductive, and osteoinductive properties of autograft bone. Use of local autograft avoids the costs associated with iliac crest autograft, including increased operating and anesthesia time, additional operating room supplies (drapes, sutures, etc) needed for operating at a second site, and prolonged hospital stay due to pain at the donor site. Use of local autograft also obviates complications at a second surgical site; purchase, storage, and sterilization of allograft; and the neck swelling, possible carcinogenicity, and cost of purchase of BMP. Other than the occasional reuse of distraction posts, this method involves no other expensive explant supplies.
Autografts have osteogenic, osteoconductive, and osteoinductive properties, and autograft fusion rates are generally superior to allograft fusion rates. Bone morphogenetic protein fusion rates may be comparable to autograft fusion rates.9,26,32 Shortcomings of iliac crest autografts include increased operative time, blood loss, and donor-site morbidity. Allografts are osteoconductive and osteoinductive, but their fusion rates are inferior to those of iliac crest autografts. Other shortcomings are infection transmission and immunogenicity risks, higher graft resorption and collapse rates, cost, and previous issues relating to provenance. Bone morphogenetic protein is the most osteoinductive material with fusion rates similar to those of autograft, but its use is associated with neck swelling, dysphagia, osteolysis, potential carcinogenicity, and high cost.9
Conclusion
Overall, use of local autograft with a PEEK spacer has all the advantages of iliac crest autograft along with the benefit of working within the same operative window as the ACDF, thus reducing the infection, bleeding, and pain risks that may be encountered with a second incision. This procedure is effective, inexpensive, and cost-effective compared with alternatives and may be preferable for 1-level ACDF. In a population of patients with high rates of tobacco use, diabetes mellitus, obesity, and other factors that negatively affect fusion rates, local autograft may be a good choice for efficacy and cost savings.
Acknowledgments
The authors thank Shirley McCartney, PhD, for editorial assistance and Andy Rekito, MS, for illustrative assistance.
Anterior cervical discectomy and fusion (ACDF) has been performed with various techniques and devices for many years. Autologous iliac crest grafts were initially used for the Cloward1,2 and Robinson and Smith3 techniques, but because of iliac crest graft site complications (eg, pain, infection, fracture, dystrophic scarring4,5), the procedure was generally superseded by allograft implants. These implants were then supplemented with anterior locking plate devices. More recently, unitary devices combining a polyetheretherketone (PEEK) spacer with screw or blade fixation have been developed, such as the Zero P (Synthes, Inc.) and the ROI-C cervical cage (LDR). Bone graft is required to fill the cavity of these devices and to promote osseous union. Demineralized bone matrix,6 tricalcium phosphate,7,8 and bone morphogenetic protein (BMP) have been used for these purposes, but they add expense to the procedure and have been associated with several complications (eg, neck swelling, dysphagia associated with BMP).9
Although multiple studies have demonstrated effective fusion rates and good outcomes for both iliac crest autograft and grafting/spacer constructs, the debate over cost and “added value” remains unresolved. One institution, which has published articles reviewing the spine literature and its own data, concluded that iliac crest autograft was the most cost-effective and consistently successful ACDF procedure.5,10
The VA Portland Health Care System (VAPORHCS) has analyzed the use of local autograft sources at the surgical site to circumvent the need to make a second incision at the iliac crest and, theoretically, to decrease risks and expenses associated with iliac crest autograft, allograft bone, and artificial constructs. Given the paucity of data on this method, the case series presented here represents one of a few studies that analyze local autograft for promotion of arthrodesis in a PEEK spacer device.
This article will report on the prospectively collected results of consecutive cases performed by Dr. Ross using a ROI-C cervical cage for 1-level anterior cervical discectomy between August 2011 and November 2014. This study received institutional review board approval.
Methods
Neck disability index (NDI) forms were used to assess the impact of neck pain on patients’ ability to manage in everyday life. The NDI form was completed before surgery and 3 and 9 months after surgery.
Dr. Ross preferred to perform minimally invasive posterior cervical foraminotomy for unilateral radiculopathy. Therefore, all patients with radiculopathy had bilateral symptoms or a symptomatic midline disc protrusion not accessible from a posterior approach. Standard techniques were used to make a left-side approach to the anterior cervical spine except in cases in which a previous right-side approach could be reused. Under the microscope, the anterior longitudinal ligament and annulus were incised, and the anterior contents of the disc space were removed with curettes and pituitary rongeurs. Care was taken to remove all cartilage from beneath the anterior inferior lip of the rostral vertebral body and to remove a few millimeters of the anterior longitudinal ligament from the rostral vertebral body without use of monopolar cautery (Figure 1). A 2 mm Kerrison punch then was used to remove the anterior inferior lip of the rostral vertebral body, and this bone was saved for grafting. No bone wax was used within the disc space.
After all disc space cartilage was removed from the endplates, additional bone was obtained from the uncovertebral joints and posterior vertebral bodies as the decompression proceeded posteriorly. Occasionally, distraction posts were used if the disc space was too narrow for optimal visualization posteriorly. After decompression was achieved, a lordotic ROI-C cervical cage was packed in its lumen with the bone chips and impacted into the disc space under fluoroscopic guidance. The blades were impacted under fluoroscopic guidance as well. The wound was closed with absorbable suture.
Antibiotics were given for no more than 24 hours after surgery. Ketorolac was used for analgesia the night of the surgery, and patients were asked to not use nonsteroidal anti-inflammatory drugs for 3 months after surgery. Lateral radiographs were obtained 3 and 9 months after surgery and every 6 months thereafter until arthrodesis was detected.
Results
Seventy-seven consecutive patients underwent 1-level anterior cervical discectomy (Table 1). Twenty-four procedures were performed for radiculopathy, 52 for myelopathy, and 1 for central cord injury sustained in a fall by a patient with preexisting spinal stenosis. Surgery was performed at C3-C4 (25 cases), C4-C5 (11 cases), C5-C6 (15 cases), and C6-C7 (1 case) for patients with myelopathy. Surgery was performed at C3-C4 (2 cases), C4-C5 (3 cases), C5-C6 (9 cases), and C6-C7 (10 cases) for patients with radiculopathy.
Twenty-eight patients reported presurgery tobacco use. Although all tobacco-using patients agreed to cease use in the perioperative period, at least 9 admitted to resuming tobacco use immediately after surgery. Eighteen patients had diabetes mellitus. In 2 patients, a diagnosis of osteoporosis was made with dual-energy X-ray absorptiometry. One patient was a chronic user of steroids before and after surgery. Mean body mass index (BMI) was 30.6, and 13 patients were morbidly obese (BMI > 34).
In 2 cases, only a single blade was placed. The second blade could not be placed because of broken adjacent screws (1 case) or undetermined reason (1 case).
The mean time for follow-up was 17 months (range 3-34). Four patients were lost to follow-up: 3 after the 1-month postoperative visit and 1 with severe psychiatric problems after hospital discharge.
There were no new neurologic deficits, no wound infections, and no recurrent laryngeal nerve palsies in the 77 patients. Eight months after surgery, 1 patient with radiculopathy underwent foraminotomy at the index level for persisting foraminal stenosis. Two patients whose myelopathic symptoms persisted after surgery returned for minimally invasive posterior laminotomy to remove infolded ligamentum flavum. The presurgery and 3- and 9-month postsurgery NDI scores were available for 52 patients (Table 2). Before surgery the mean NDI score was 24 (range 8-40). Three months postsurgery the mean NDI score was 15 (range 2-27) for patients with myelopathy and 13 (range 2-28) for patients with radiculopathy. The patient with the highest NDI score (28) stated that though all his symptoms were relieved, he had gauged his responses to protect his disability claim. Nine months after surgery, the mean NDI scores were 9.5 (range 5-17) for patients with myelopathy and 6 (range 2-13) for patients with radiculopathy. No NDI score was higher postsurgery than presurgery.
Arthrodesis was defined as bony bridging between the adjacent vertebral bodies and the bone graft within the lumen of the device, anterior to the device, or posterior to the device. In Dr. Ross’ protocol, computed tomography (CT) scans or flexion-extension radiographs were obtained only if pseudarthrosis was suspected to avoid unnecessary radiation exposure. Sixty-six patients had at least the 3-month radiography follow-up available. All 52 patients with 9-month follow-up data achieved complete arthrodesis, as determined by plain film radiography. Bridging ossification was found anterior to the device in all but 9 patients. Trabeculated bone was growing through the lumen of the device in all cases (Figure 2). A broken blade without clinical correlation was noted on imaging for 1 patient.
The total cost of the ROI-C cervical cage (LDR) for VAPORHCS was $3,498, or $1,749 for the PEEK spacer plus $1,749 for 2 metal blades. In comparison, the total cost of a typical anterior locking plate would have been $6,700, or $3,200 for the plate plus $2,000 for 4 screws and $1,500 for an allograft fibular spacer. Demineralized bone matrix (1 mL) as used in cervical arthrodesis by other surgeons at VAPORHCS cost $279, or about $500 including shipping.
DISCUSSION
Anterior cervical discectomy with fusion is a very common and successful surgical procedure for cervical myelopathy, radiculopathy, and degenerative disease that has failed to be corrected with conservative therapy.10 Medicare data documented a 206% increase in 1-level fusion procedures for degenerative spine pathology performed between 1992 and 2005.11 When a procedure is performed so often, it is appropriate to review methods and analyze efficacy, cost, and cost-effectiveness.
According to a 2007 meta-analysis, the fusion rates of 1-level ACDF arthrodesis at 1-year follow-up are 97.1% in patients treated with anterior plates and 92.1% in patients treated with noninstrumented fusion.12 The rate disparity was larger for multiple-level fusion: 50% to 82.5% for instrumented cases12,13 vs 3% to 42% for noninstrumented cases.14-16 Given the higher fusion rates achieved with instrumentation, surgeons have favored its use in ACDF.
Computed Tomography Use
Computed tomography has long been considered the gold standard for assessing arthrodesis outcomes (eg, Siambanes and Mather).17 However, recent data on potential harm caused by CT-related ionizing radiation suggest a need for caution with routine CT use.18,19 For cervical spine CT, Schonfeld and colleagues found that the risk for excess thyroid cancers ranged from 1 to 33 cases per 10,000 CT scans.20 According to another report, “limiting neck CT scanning to a higher risk group would increase the gap between benefit and harm, whereas performing CT routinely on low-risk cases approaches a point where its harm equals or exceeds its benefit.”19 As some have questioned even routinepostoperative use of radiation in patients with unremarkable clinical courses—patients should be spared unnecessary exposure—CT scans or flexion-extensionradiographs were obtained at VAPORHCS only if clinical symptoms or radiographs were suggestive of pseudarthrosis.21 As none of the VAPORHCS patients had those symptoms, none underwent postoperative CT.
For anterior cervical arthrodesis, surgeon preference determines which of many different bone substrates can be used with instrumentation, which impacts the costs. Fusion substrates include structural autografts, structural allografts, morselized autografts, morselized allografts, demineralized allografts, porous ceramics and metals, and BMP. Given these many options, studies comparing the constructs are lacking, especially with regard to the cost of alternative fusion constructs that produce similar outcomes. The Centers for Disease Control and Prevention defines cost-benefit analysis as a “type of economic evaluation that measures both costs and benefits (ie, negative and positive consequences) associated with an intervention in dollar terms.”22 It has been reported that using iliac crest autografts with anterior plate instrumentation is the most cost-effective method, yet alternatives remain in use.5,10
For ACDF, iliac crest bone is an ideal and widely used construct substrate. Structural grafts harvested from the crest provide significant stability due to their bicortical or tricortical configuration with interposed osteoinductive and osteogenic cancellous bone. Few graft complications (eg, graft resorption) and no immunogenic or infectious complications have been reported for iliac crest bone. However, autologous iliac crest increases operative time, and donor-site morbidity has been reported.23,24 A retrospective questionnaire-based investigation by Silber and colleagues, who evaluated iliac crest bone graft site morbidity in 1-level ACDF, found that 26.1% of patients had pain at the iliac crest harvest site, and 15.7% had numbness.24 Other complications, which occurred at lower rates, were bruising, hematoma, pelvic fracture, and poor cosmesis.23,25 In addition, osteoporosis and comorbid conditions have made it a challenge to acquire iliac crest autograft, contributing to the popularity of alternative substrates.25
Allografts
An alternative to autografts, allografts have the advantages of reduced operative time and reduced donor-site morbidity.26 Major historical concerns with allografts have included risk for disease transmission, costs associated with sterilization and serologic screening of grafts, and lack of oversight, leading to human allografts being acquired from dubious sources and ending up in the operating room.27,28 Two major types of allografts are available: mineralized and demineralized.
Arthrodesis rates are inferior for mineralized (structural) allografts with instrumentation than for autografts with instrumentation.29 In addition, smoking and other comorbidities have influenced fusion rates more in allograft than autograft fusions.30-33 However, allografts are being widely used because they avoid the donor-site morbidity associated with autografts and because they are load bearing, can provide structural stability and an osteoconductive matrix, and can be used off the shelf without adding much time to surgery.
Demineralized matrix substrates are commercial osteoconductive and osteoinductive biomaterials approved for filling bone gaps and extending graft when combined with autograft.7,8 Despite their osteoinductive properties, these substrates have had a high degree of product inconsistency, in some cases leading to poor outcomes.34 The lack of randomized studies with these constructs has made the determination of clear indications a challenge.
The initial enthusiasm over use of BMP, another bone-graft substitute for cervical fusion, was curtailed by reports of adverse events (AEs). Effective in anterior lumbar spine fusions, BMP was adapted to off-label use in the cervical spine a few years ago.35 Initial studies by Baskin and colleagues and Bishop and colleagues showed its fusion rates superior to those of allograft.31,32 Both studies reported no significant AEs. However, studies by Dickerman and colleagues and Smucker and colleagues demonstrated increased soft-tissue swelling leading to dysphagia and prolonged hospitalization, which were attributed to higher dosage (no study has identified a precise dose for individual patients).36,37 In addition, the cost of BMP is higher than that of any other bone-graft option for ACDF.3 Osteolysis has also been reported with BMP use.38-40 Carragee and colleagues highlighted the potential carcinogenicity of BMP, but this finding was not corroborated by Lad and colleagues.41,42
Cost Considerations
In addition to surgical effectiveness, spine surgical device costs have come under increased scrutiny.43-45 In 2012, plates were reported to cost (without overhead or profit margin to hospitals) between $1,015 and $3,601, and allograft spacers were estimated to cost between $1,220 and $3,640, cage costs ranged from $1,942 to $4,347, and PEEK spacers cost from $4,930 to $5,246.5 Individual surgeon instrumentation costs varied 10-fold based on the fusion constructs used.5
In a cost-effectiveness review of anterior cervical techniques, cage alone was the least expensive technique, disc arthroplasty or cage/plate/bone substitute groups were the next most expensive, and autograft alone was the most expensive option due to hip graft site morbidity.43 In another study, operative time associated with harvesting an iliac crest graft was equivalent in cost to that of an interbody cage.44 Other studies have compared the costs of various anterior cervical fusion constructs.9,10,45,46 A limitation of these studies is that autologous bone often refers to iliac crest grafts rather than local autograft. Epstein reviewed data from these studies and concluded, “ACDF using dynamic plates and autografts are the most cost effective treatment for anterior cervical discectomy,” citing a cost of $1,015 for this construct.5 Although Epstein demonstrated the cost-effectiveness of autograft in an individual surgeon’s hands, the results also are significant in that the studies identified areas in which improvements can be made at other institutions. The ROI-C cervical cage and local autograft bone cost that the authors report is at the lower end of the range reported by Epstein.5
Device explant rates also can be a concern. Operative waste was well described in a retrospective analysis of 87 ACDF procedures.47 The study found that the cost of explanting devices implanted during the same intraoperative period was equivalent to 9.2% of the cost of permanently implanted constructs. Epstein addressed operative waste by using educational modules to evaluate spine surgeons’ decision making before and after education. After the intervention, the institution noted a marked decline in costs related to explanted devices—from 20% in 2010 (before education) to 5.8% of the total cost of implanted devices in 2010 (after education).5
In the present study, the authors demonstrated that use of local morselized autograft with a PEEK spacer for 1-level ACDF had excellent arthrodesis rates and minimal complications. Of the 52 patients with 9 month postoperative data, all achieved arthrodesis regardless of tobacco use. This method compares favorably with other fusion options in terms of radiographic arthrodesis rates. In addition, it avoids the donor-site morbidity associated with autografts from an iliac site but maintains the benefits of the osteogenic, osteoconductive, and osteoinductive properties of autograft bone. Use of local autograft avoids the costs associated with iliac crest autograft, including increased operating and anesthesia time, additional operating room supplies (drapes, sutures, etc) needed for operating at a second site, and prolonged hospital stay due to pain at the donor site. Use of local autograft also obviates complications at a second surgical site; purchase, storage, and sterilization of allograft; and the neck swelling, possible carcinogenicity, and cost of purchase of BMP. Other than the occasional reuse of distraction posts, this method involves no other expensive explant supplies.
Autografts have osteogenic, osteoconductive, and osteoinductive properties, and autograft fusion rates are generally superior to allograft fusion rates. Bone morphogenetic protein fusion rates may be comparable to autograft fusion rates.9,26,32 Shortcomings of iliac crest autografts include increased operative time, blood loss, and donor-site morbidity. Allografts are osteoconductive and osteoinductive, but their fusion rates are inferior to those of iliac crest autografts. Other shortcomings are infection transmission and immunogenicity risks, higher graft resorption and collapse rates, cost, and previous issues relating to provenance. Bone morphogenetic protein is the most osteoinductive material with fusion rates similar to those of autograft, but its use is associated with neck swelling, dysphagia, osteolysis, potential carcinogenicity, and high cost.9
Conclusion
Overall, use of local autograft with a PEEK spacer has all the advantages of iliac crest autograft along with the benefit of working within the same operative window as the ACDF, thus reducing the infection, bleeding, and pain risks that may be encountered with a second incision. This procedure is effective, inexpensive, and cost-effective compared with alternatives and may be preferable for 1-level ACDF. In a population of patients with high rates of tobacco use, diabetes mellitus, obesity, and other factors that negatively affect fusion rates, local autograft may be a good choice for efficacy and cost savings.
Acknowledgments
The authors thank Shirley McCartney, PhD, for editorial assistance and Andy Rekito, MS, for illustrative assistance.
1. Cloward RB. The anterior approach for removal of ruptured cervical disks. 1958. J Neurosurg Spine. 2007;6(5):496-511.
2. Cloward RB. The anterior approach for removal of ruptured cervical disks. J Neurosurg. 1958;15(6):602-617.
3. Robinson RA, Smith GW. Anterolateral cervical disc removal and interbody fusion for cervical disc syndrome. SAS J. 2010;4(1):34-35.
4. Dimitriou R, Mataliotakis GI, Angoules AG, Kanakaris NK, Giannoudis PV. Complications following autologous bone graft harvesting from the iliac crest and using the RIA: a systematic review. Injury. 2011;42(suppl 2):S3-S15.
5. Epstein NE. Iliac crest autograft versus alternative constructs for anterior cervical spine surgery: pros, cons, and costs. Surg Neurol Int. 2012;3(suppl 3):S143-S156.
6. Gruskin E, Doll BA, Futrell FW, Schmitz JP, Hollinger JO. Demineralized bone matrix in bone repair: history and use. Adv Drug Deliv Rev. 2012;64(12):1063-1077.
7. Becker S, Maissen O, Ponomarev I, Stoll T, Rahn B, Wilke I. Osteopromotion by a beta-tricalcium phosphate/bone marrow hybrid implant for use in spine surgery. Spine (Phila Pa 1976). 2006;31(1):11-17.
8. Muschik M, Ludwig R, Halbhübner S, Bursche K, Stoll T. Beta-tricalcium phosphate as a bone substitute for dorsal spinal fusion in adolescent idiopathic scoliosis: preliminary results of a prospective clinical study. Eur Spine J. 2001;10(suppl 2):S178-S184.
9. Buttermann GR. Prospective nonrandomized comparison of an allograft with bone morphogenic protein versus an iliac-crest autograft in anterior cervical discectomy and fusion. Spine J. 2008;8(3):426-435.
10. Epstein NE. Efficacy and outcomes of dynamic-plated single-level anterior diskectomy/fusion with additional analysis of comparative costs. Surg Neurol Int. 2011;2:9.
11. Wang MC, Kreuter W, Wolfla CE, Maiman DJ, Deyo RA. Trends and variations in cervical spine surgery in the United States: Medicare beneficiaries, 1992 to 2005. Spine (Phila Pa 1976). 2009;34(9):955-961.
12. Fraser JF, Härtl R. Anterior approaches to fusion of the cervical spine: a metaanalysis of fusion rates. J Neurosurg Spine. 2007;6(4):298-303.
13. Nirala AP, Husain M, Vatsal DK. A retrospective study of multiple interbody grafting and long segment strut grafting following multilevel anterior cervical decompression. Br J Neurosurg. 2004;18(3):227-232.
14. Bohlman HH, Emery SE, Goodfellow DB, Jones PK. Robinson anterior cervical discectomy and arthrodesis for cervical radiculopathy. Long-term follow-up of one hundred and twenty-two patients. J Bone Joint Surg Am. 1993;75(9):1298-1307.
15. Cauthen JC, Kinard RE, Vogler JB, et al. Outcome analysis of noninstrumented anterior cervical discectomy and interbody fusion in 348 patients. Spine (Phila Pa 1976). 1998;23(2):188-192.
16. Emery SE, Fisher JR, Bohlman HH. Three-level anterior cervical discectomy and fusion: radiographic and clinical results. Spine (Phila Pa 1976). 1997;22(22):2622-2624.
17. Siambanes D, Mather S. Comparison of plain radiographs and CT scans in instrumented posterior lumbar interbody fusion. Orthopedics. 1998;21(2):165-167.
18. Berrington de González A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169(22):2071-2077.
19. Hikino K, Yamamoto LG. The benefit of neck computed tomography compared with its harm (risk of cancer). J Trauma Acute Care Surg. 2015;78(1):126-131.
20. Schonfeld SJ, Lee C, Berrington de González A. Medical exposure to radiation and thyroid cancer. Clin Oncol (R Coll Radiol). 2011;23(4):244-250.
21. Bartels RH, Beems T, Schutte PJ, Verbeek AL. The rationale of postoperative radiographs after cervical anterior discectomy with stand-alone cage for radicular pain. J Neurosurg Spine. 2010;12(3):275-279.
22. Centers for Disease Control and Prevention. The different types of health assessments. Centers for Disease Control and Prevention website. http://www.cdc.gov/healthyplaces/types_health_assessments.htm. Updated July 25, 2012. Accessed April 8, 2016.
23. Schnee CL, Freese A, Weil RJ, Marcotte PJ. Analysis of harvest morbidity and radiographic outcome using autograft for anterior cervical fusion. Spine (Phila Pa 1976). 1997;22(19):2222-2227.
24. Silber JS, Anderson DG, Daffner SD, et al. Donor site morbidity after anterior iliac crest bone harvest for single-level anterior cervical discectomy and fusion. Spine (Phila Pa 1976). 2003;28(2):134-139.
25. Seiler JG 3rd, Johnson J. Iliac crest autogenous bone grafting: donor site complications. J South Orthop Assoc. 2000;9(2):91-97.
26. Floyd T, Ohnmeiss D. A meta-analysis of autograft versus allograft in anterior cervical fusion. Eur Spine J. 2000;9(5):398-403.
27. Delloye C, Cornu O, Druez V, Barbier O. Bone allografts: what they can offer and what they cannot. J Bone Joint Surg Br. 2007;89(5):574-579.
28. Armour S. Illegal trade in bodies shakes loved ones. USA Today. http://usatoday30.usatoday.com/money/2006-04-26-body-parts-cover-usat_x.htm. Updated April 28, 2006. Accessed April 6, 2016.
29. Wigfield CC, Nelson RJ. Nonautologous interbody fusion materials in cervical spine surgery: how strong is the evidence to justify their use? Spine (Phila Pa 1976). 2001;26(6):687-694.
30. Bärlocher CB, Barth A, Krauss JK, Binggeli R, Seiler RW. Comparative evaluation of microdiscectomy only, autograft fusion, polymethylmethacrylate interposition, and threaded titanium cage fusion for treatment of single-level cervical disc disease: a prospective randomized study in 125 patients. Neurosurg Focus. 2002;12(1):E4.
31. Baskin DS, Ryan P, Sonntag V, Westmark R, Widmayer MA. A prospective, randomized, controlled cervical fusion study using recombinant human bone morphogenetic protein-2 with the CORNERSTONE-SR allograft ring and the ATLANTIS anterior cervical plate. Spine (Phila Pa 1976). 2003;28(12):1219-1224.
32. Bishop RC, Moore KA, Hadley MN. Anterior cervical interbody fusion using autogeneic and allogeneic bone graft substrate: a prospective comparative analysis. J Neurosurg. 1996;85(2):206-210.
33. Martin GJ Jr, Haid RW Jr, MacMillan M, Rodts GE Jr, Berkman R. Anterior cervical discectomy with freeze-dried fibula allograft. Overview of 317 cases and literature review. Spine (Phila Pa 1976). 1999;24(9):852-858.
34. Bae HW, Zhao L, Kanim LE, Wong P, Delamarter RB, Dawson EG. Intervariability and intravariability of bone morphogenetic proteins in commercially available demineralized bone matrix products. Spine (Phila Pa 1976). 2006;31(12):1299-1306.
35. Burkus JK, Gornet MF, Dickman CA, Zdeblick TA. Anterior lumbar interbody fusion using rhBMP-2 with tapered interbody cages. J Spinal Disord Tech. 2002;15(5):337-349.
36. Dickerman RD, Reynolds AS, Morgan BC, Tompkins J, Cattorini J, Bennett M. rh-BMP-2 can be used safely in the cervical spine: dose and containment are the keys! Spine J. 2007;7(4):508-509.
37. Smucker JD, Rhee JM, Singh K, Yoon ST, Heller JG. Increased swelling complications associated with off-label usage of rhBMP-2 in the anterior cervical spine. Spine (Phila Pa 1976). 2006;31(24):2813-2819.
38. Vaidya R, Carp J, Sethi A, Bartol S, Craig J, Les CM. Complications of anterior cervical discectomy and fusion using recombinant human bone morphogenetic protein-2. Eur Spine J. 2007;16(8):1257-1265.
39. Vaidya R, Sethi A, Bartol S, Jacobson M, Coe C, Craig JG. Complications in the use of rhBMP-2 in PEEK cages for interbody spinal fusions. J Spinal Disord Tech. 2008;21(8):557-562.
40. Knox JB, Dai JM 3rd, Orchowski J. Osteolysis in transforaminal lumbar interbody fusion with bone morphogenetic protein-2. Spine (Phila Pa 1976). 2011;36(8):672-676.
41. Carragee EJ, Chu G, Rohatgi R, et al. Cancer risk after use of recombinant bone morphogenetic protein-2 for spinal arthrodesis. J Bone Joint Surg Am. 2013;95(17):1537-1545.
42. Lad SP, Bagley JH, Karikari IO, et al. Cancer after spinal fusion: the role of bone morphogenetic protein. Neurosurgery. 2013;73(3):440-449.
43. Bhadra AK, Raman AS, Casey AT, Crawford RJ. Single-level cervical radiculopathy: clinical outcome and cost-effectiveness of four techniques of anterior cervical discectomy and fusion and disc arthroplasty. Eur Spine J. 2009;18(2):232-237.
44. Castro FP Jr, Holt RT, Majd M, Whitecloud TS 3rd. A cost analysis of two anterior cervical fusion procedures. J Spinal Disord. 2000;13(6):511-514.
45. Kandziora F, Pflugmacher R, Scholz M, et al. Treatment of traumatic cervical spine instability with interbody fusion cages: a prospective controlled study with a 2-year follow-up. Injury. 2005;36(suppl 2):B27-B35.
46. Vaidya R, Weir R, Sethi A, Meisterling S, Hakeos W, Wybo CD. Interbody fusion with allograft and rhBMP-2 leads to consistent fusion but early subsidence. J Bone Joint Surg Br. 2007;89(3):342-345.
47. Epstein NE, Schwall GS, Hood DC. The incidence and cost of devices explanted during single-level anterior diskectomy/fusions. Surg Neurol Int. 2011;2:23.
1. Cloward RB. The anterior approach for removal of ruptured cervical disks. 1958. J Neurosurg Spine. 2007;6(5):496-511.
2. Cloward RB. The anterior approach for removal of ruptured cervical disks. J Neurosurg. 1958;15(6):602-617.
3. Robinson RA, Smith GW. Anterolateral cervical disc removal and interbody fusion for cervical disc syndrome. SAS J. 2010;4(1):34-35.
4. Dimitriou R, Mataliotakis GI, Angoules AG, Kanakaris NK, Giannoudis PV. Complications following autologous bone graft harvesting from the iliac crest and using the RIA: a systematic review. Injury. 2011;42(suppl 2):S3-S15.
5. Epstein NE. Iliac crest autograft versus alternative constructs for anterior cervical spine surgery: pros, cons, and costs. Surg Neurol Int. 2012;3(suppl 3):S143-S156.
6. Gruskin E, Doll BA, Futrell FW, Schmitz JP, Hollinger JO. Demineralized bone matrix in bone repair: history and use. Adv Drug Deliv Rev. 2012;64(12):1063-1077.
7. Becker S, Maissen O, Ponomarev I, Stoll T, Rahn B, Wilke I. Osteopromotion by a beta-tricalcium phosphate/bone marrow hybrid implant for use in spine surgery. Spine (Phila Pa 1976). 2006;31(1):11-17.
8. Muschik M, Ludwig R, Halbhübner S, Bursche K, Stoll T. Beta-tricalcium phosphate as a bone substitute for dorsal spinal fusion in adolescent idiopathic scoliosis: preliminary results of a prospective clinical study. Eur Spine J. 2001;10(suppl 2):S178-S184.
9. Buttermann GR. Prospective nonrandomized comparison of an allograft with bone morphogenic protein versus an iliac-crest autograft in anterior cervical discectomy and fusion. Spine J. 2008;8(3):426-435.
10. Epstein NE. Efficacy and outcomes of dynamic-plated single-level anterior diskectomy/fusion with additional analysis of comparative costs. Surg Neurol Int. 2011;2:9.
11. Wang MC, Kreuter W, Wolfla CE, Maiman DJ, Deyo RA. Trends and variations in cervical spine surgery in the United States: Medicare beneficiaries, 1992 to 2005. Spine (Phila Pa 1976). 2009;34(9):955-961.
12. Fraser JF, Härtl R. Anterior approaches to fusion of the cervical spine: a metaanalysis of fusion rates. J Neurosurg Spine. 2007;6(4):298-303.
13. Nirala AP, Husain M, Vatsal DK. A retrospective study of multiple interbody grafting and long segment strut grafting following multilevel anterior cervical decompression. Br J Neurosurg. 2004;18(3):227-232.
14. Bohlman HH, Emery SE, Goodfellow DB, Jones PK. Robinson anterior cervical discectomy and arthrodesis for cervical radiculopathy. Long-term follow-up of one hundred and twenty-two patients. J Bone Joint Surg Am. 1993;75(9):1298-1307.
15. Cauthen JC, Kinard RE, Vogler JB, et al. Outcome analysis of noninstrumented anterior cervical discectomy and interbody fusion in 348 patients. Spine (Phila Pa 1976). 1998;23(2):188-192.
16. Emery SE, Fisher JR, Bohlman HH. Three-level anterior cervical discectomy and fusion: radiographic and clinical results. Spine (Phila Pa 1976). 1997;22(22):2622-2624.
17. Siambanes D, Mather S. Comparison of plain radiographs and CT scans in instrumented posterior lumbar interbody fusion. Orthopedics. 1998;21(2):165-167.
18. Berrington de González A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169(22):2071-2077.
19. Hikino K, Yamamoto LG. The benefit of neck computed tomography compared with its harm (risk of cancer). J Trauma Acute Care Surg. 2015;78(1):126-131.
20. Schonfeld SJ, Lee C, Berrington de González A. Medical exposure to radiation and thyroid cancer. Clin Oncol (R Coll Radiol). 2011;23(4):244-250.
21. Bartels RH, Beems T, Schutte PJ, Verbeek AL. The rationale of postoperative radiographs after cervical anterior discectomy with stand-alone cage for radicular pain. J Neurosurg Spine. 2010;12(3):275-279.
22. Centers for Disease Control and Prevention. The different types of health assessments. Centers for Disease Control and Prevention website. http://www.cdc.gov/healthyplaces/types_health_assessments.htm. Updated July 25, 2012. Accessed April 8, 2016.
23. Schnee CL, Freese A, Weil RJ, Marcotte PJ. Analysis of harvest morbidity and radiographic outcome using autograft for anterior cervical fusion. Spine (Phila Pa 1976). 1997;22(19):2222-2227.
24. Silber JS, Anderson DG, Daffner SD, et al. Donor site morbidity after anterior iliac crest bone harvest for single-level anterior cervical discectomy and fusion. Spine (Phila Pa 1976). 2003;28(2):134-139.
25. Seiler JG 3rd, Johnson J. Iliac crest autogenous bone grafting: donor site complications. J South Orthop Assoc. 2000;9(2):91-97.
26. Floyd T, Ohnmeiss D. A meta-analysis of autograft versus allograft in anterior cervical fusion. Eur Spine J. 2000;9(5):398-403.
27. Delloye C, Cornu O, Druez V, Barbier O. Bone allografts: what they can offer and what they cannot. J Bone Joint Surg Br. 2007;89(5):574-579.
28. Armour S. Illegal trade in bodies shakes loved ones. USA Today. http://usatoday30.usatoday.com/money/2006-04-26-body-parts-cover-usat_x.htm. Updated April 28, 2006. Accessed April 6, 2016.
29. Wigfield CC, Nelson RJ. Nonautologous interbody fusion materials in cervical spine surgery: how strong is the evidence to justify their use? Spine (Phila Pa 1976). 2001;26(6):687-694.
30. Bärlocher CB, Barth A, Krauss JK, Binggeli R, Seiler RW. Comparative evaluation of microdiscectomy only, autograft fusion, polymethylmethacrylate interposition, and threaded titanium cage fusion for treatment of single-level cervical disc disease: a prospective randomized study in 125 patients. Neurosurg Focus. 2002;12(1):E4.
31. Baskin DS, Ryan P, Sonntag V, Westmark R, Widmayer MA. A prospective, randomized, controlled cervical fusion study using recombinant human bone morphogenetic protein-2 with the CORNERSTONE-SR allograft ring and the ATLANTIS anterior cervical plate. Spine (Phila Pa 1976). 2003;28(12):1219-1224.
32. Bishop RC, Moore KA, Hadley MN. Anterior cervical interbody fusion using autogeneic and allogeneic bone graft substrate: a prospective comparative analysis. J Neurosurg. 1996;85(2):206-210.
33. Martin GJ Jr, Haid RW Jr, MacMillan M, Rodts GE Jr, Berkman R. Anterior cervical discectomy with freeze-dried fibula allograft. Overview of 317 cases and literature review. Spine (Phila Pa 1976). 1999;24(9):852-858.
34. Bae HW, Zhao L, Kanim LE, Wong P, Delamarter RB, Dawson EG. Intervariability and intravariability of bone morphogenetic proteins in commercially available demineralized bone matrix products. Spine (Phila Pa 1976). 2006;31(12):1299-1306.
35. Burkus JK, Gornet MF, Dickman CA, Zdeblick TA. Anterior lumbar interbody fusion using rhBMP-2 with tapered interbody cages. J Spinal Disord Tech. 2002;15(5):337-349.
36. Dickerman RD, Reynolds AS, Morgan BC, Tompkins J, Cattorini J, Bennett M. rh-BMP-2 can be used safely in the cervical spine: dose and containment are the keys! Spine J. 2007;7(4):508-509.
37. Smucker JD, Rhee JM, Singh K, Yoon ST, Heller JG. Increased swelling complications associated with off-label usage of rhBMP-2 in the anterior cervical spine. Spine (Phila Pa 1976). 2006;31(24):2813-2819.
38. Vaidya R, Carp J, Sethi A, Bartol S, Craig J, Les CM. Complications of anterior cervical discectomy and fusion using recombinant human bone morphogenetic protein-2. Eur Spine J. 2007;16(8):1257-1265.
39. Vaidya R, Sethi A, Bartol S, Jacobson M, Coe C, Craig JG. Complications in the use of rhBMP-2 in PEEK cages for interbody spinal fusions. J Spinal Disord Tech. 2008;21(8):557-562.
40. Knox JB, Dai JM 3rd, Orchowski J. Osteolysis in transforaminal lumbar interbody fusion with bone morphogenetic protein-2. Spine (Phila Pa 1976). 2011;36(8):672-676.
41. Carragee EJ, Chu G, Rohatgi R, et al. Cancer risk after use of recombinant bone morphogenetic protein-2 for spinal arthrodesis. J Bone Joint Surg Am. 2013;95(17):1537-1545.
42. Lad SP, Bagley JH, Karikari IO, et al. Cancer after spinal fusion: the role of bone morphogenetic protein. Neurosurgery. 2013;73(3):440-449.
43. Bhadra AK, Raman AS, Casey AT, Crawford RJ. Single-level cervical radiculopathy: clinical outcome and cost-effectiveness of four techniques of anterior cervical discectomy and fusion and disc arthroplasty. Eur Spine J. 2009;18(2):232-237.
44. Castro FP Jr, Holt RT, Majd M, Whitecloud TS 3rd. A cost analysis of two anterior cervical fusion procedures. J Spinal Disord. 2000;13(6):511-514.
45. Kandziora F, Pflugmacher R, Scholz M, et al. Treatment of traumatic cervical spine instability with interbody fusion cages: a prospective controlled study with a 2-year follow-up. Injury. 2005;36(suppl 2):B27-B35.
46. Vaidya R, Weir R, Sethi A, Meisterling S, Hakeos W, Wybo CD. Interbody fusion with allograft and rhBMP-2 leads to consistent fusion but early subsidence. J Bone Joint Surg Br. 2007;89(3):342-345.
47. Epstein NE, Schwall GS, Hood DC. The incidence and cost of devices explanted during single-level anterior diskectomy/fusions. Surg Neurol Int. 2011;2:23.