User login
A toddler presents with patchy hair loss
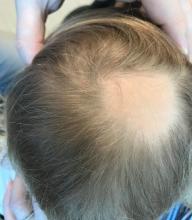
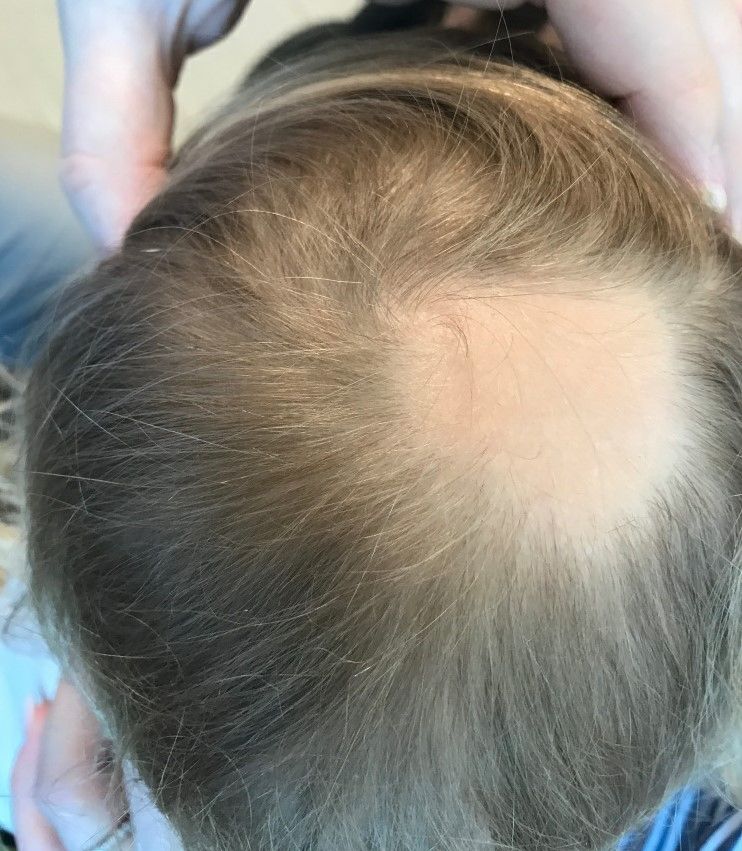
Given the history of sudden hair loss, with the exam revealing a well-circumscribed patch of focal alopecia without cutaneous inflammation, hairs with a narrow base and broad distal shaft, the diagnosis is alopecia areata (AA).
Alopecia areata (AA) is a nonscarring alopecia, within a set of diseases characterized by the preservation of hair follicles and therefore the potential for future hair regrowth.1 AA is believed to be caused by a breakdown of the immune-privileged nature of hair follicles, resulting in T-lymphocytes targeting the hair follicle directly, shifting follicles to early catagen or telogen phase, but sparing follicular stem cells, thereby allowing the follicle to regenerate in the future.1-3 Risk factors include family history of AA, thyroid disorders, as well as iron and vitamin D deficiency.4,5 It characteristically presents with focal, well-demarcated patches of hair loss in the scalp, typically with background skin normal to slightly pink.3,6 Exam can show “exclamation point” hairs consisting of hairs that are narrow at their base and wide at the distal end.3,7 Patients may also exhibit eyebrow and eyelash loss as well as nail changes including nail pitting and splitting.8 Diagnosis is typically made clinically but is supported by a positive hair pull test, where hairs are pulled from the periphery of an alopecic lesion; the presence of greater than 10% of hairs plucked from the scalp indicates a positive result.9,10
What’s the differential diagnosis?
The differential diagnosis of AA includes other nonscarring alopecias such as trichotillomania and telogen effluvium. Other possible diagnoses include lichen planopilaris and tinea capitis.
Trichotillomania results in irregularly bordered hair loss and broken hairs of different lengths because of an internal urge to remove one’s hair, resulting in nonscarring alopecia. It can be associated with obsessive-compulsive disorder, anxiety, or other body-altering behaviors like skin picking and nail biting (characterized as body-focused repetitive behavior disorders). Treatments include reassurance and education, behavior modification, or systemic therapy including tricyclic antidepressants or SSRIs. Toddlers can engage in hair pulling behavior and trichotillomania can be difficult to differentiate from AA. However, the absence of broken hairs of varying lengths makes trichotillomania less likely in this patient.
Telogen effluvium is another form of nonscarring alopecia that presents as diffuse hair thinning across the entire scalp in response to acute psychological or physiological stress, hormonal changes, certain medications, systemic illness, or nutritional deficiency. The timing between the triggering event and hair loss can vary from weeks to months. Diagnosis requires detailed history-taking and may include evaluation for endocrinologic hair thinning (e.g. thyroid function tests) to identify reversible causes. Treatment involves directing therapy to the underlying etiology and most cases of telogen effluvium are self-limited. The presence of a well-circumscribed patch of hair loss in this patient makes AA more likely.
Lichen planopilaris (LPP) is a scarring, irreversible alopecia caused by T-lymphocytes attacking follicular hair stem cells. It is characterized by hair loss, pruritus, burning pain, scalp scaling, and multifocal scarring. Exam shows patches of alopecia with loss of follicular ostia centrally and perifollicular scale and erythema at the borders. Diagnosis is aided by biopsy of the affected scalp. Treatment of LPP requires the use of potent and superpotent topical corticosteroids and intralesional corticosteroids to decrease scalp inflammation and prevent further progression. The presence of follicular ostia and absence of perifollicular scale in this patient makes LPP highly unlikely.
Tinea capitis is a fungal infection of the scalp caused by dermatophytes including Trychophyton tonsurans and Microsporum canis. It presents with patches of alopecia with overlying scale and broken hairs and can have associated cervical and occipital lymphadenopathy. Diagnosis can involve skin scraping and KOH prep to visualize branching hyphae as well as fungal culture to identify the causative organism. Because dermatophytes in tinea capitis invade hair follicles, topical antifungals are ineffective because of their lack of penetration. Therefore, systemic antifungals including oral terbinafine and griseofulvin are considered first-line agents for treatment.
What’s the management plan?
The diagnosis of AA is usually a clinical one, though assessment of alternative diagnoses is appropriate dependent on signs and symptoms. Workup of AA can include thyroid studies because of the association with autoimmune thyroid disease, though studies suggest limited screening benefits in children.11 Given its variable and unpredictable course, management can include “watchful waiting” because of its potential for spontaneous remission.6 For limited patchy loss, active treatment with mid to superpotent topical steroids or intralesional triamcinolone acetonide in older children and adolescents is reasonable.12 Other treatment options include topical or low-dose oral minoxidil and immunotherapy with diphenylcyclopropenone or squaric acid (inducing an allergic contact dermatitis).12 Management of therapies for more extensive AA is evolving, with ongoing studies of oral JAK-inhibitors and biologic agents.12,13
Our patient was started on topical fluocinonide 0.05% solution and achieved good disease control and hair regrowth over the course of 3 months.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology at the university and Rady Children’s Hospital. They had no disclosures.
References
1. Bernardez C et al. Actas Dermosifiliogr. 2015;106(3):158-67.
2. Rajabi F et al. Br J Dermatol. 2018;179(5):1033-48.
3. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):1-12.
4. Lee S et al. J Am Acad Dermatol. 2019;80(2):466-77 e16.
5. MacLean KJ and Tidman MJ. Practitioner. 2013;257(1764):29-32, 3.
6. Pratt CH et al. Nat Rev Dis Primers. 2017;3:17011.
7. Gilhar A et al. N Engl J Med. 2012;366(16):1515-25.
8. Wyrwich KW et al. Am J Clin Dermatol. 2020;21(5):725-32.
9. Spano F and Donovan JC. Can Fam Physician. 2015;61(9):751-5.
10. Mounsey AL and Reed SW. Am Fam Physician. 2009;80(4):356-62.
11. Hordinsky MK. J Investig Dermatol Symp Proc. 2015;17(2):44-6.
12. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):15-24.
13. Zhou C et al. Clin Rev Allergy Immunol. 2021;61(3):403-23.
Given the history of sudden hair loss, with the exam revealing a well-circumscribed patch of focal alopecia without cutaneous inflammation, hairs with a narrow base and broad distal shaft, the diagnosis is alopecia areata (AA).
Alopecia areata (AA) is a nonscarring alopecia, within a set of diseases characterized by the preservation of hair follicles and therefore the potential for future hair regrowth.1 AA is believed to be caused by a breakdown of the immune-privileged nature of hair follicles, resulting in T-lymphocytes targeting the hair follicle directly, shifting follicles to early catagen or telogen phase, but sparing follicular stem cells, thereby allowing the follicle to regenerate in the future.1-3 Risk factors include family history of AA, thyroid disorders, as well as iron and vitamin D deficiency.4,5 It characteristically presents with focal, well-demarcated patches of hair loss in the scalp, typically with background skin normal to slightly pink.3,6 Exam can show “exclamation point” hairs consisting of hairs that are narrow at their base and wide at the distal end.3,7 Patients may also exhibit eyebrow and eyelash loss as well as nail changes including nail pitting and splitting.8 Diagnosis is typically made clinically but is supported by a positive hair pull test, where hairs are pulled from the periphery of an alopecic lesion; the presence of greater than 10% of hairs plucked from the scalp indicates a positive result.9,10
What’s the differential diagnosis?
The differential diagnosis of AA includes other nonscarring alopecias such as trichotillomania and telogen effluvium. Other possible diagnoses include lichen planopilaris and tinea capitis.
Trichotillomania results in irregularly bordered hair loss and broken hairs of different lengths because of an internal urge to remove one’s hair, resulting in nonscarring alopecia. It can be associated with obsessive-compulsive disorder, anxiety, or other body-altering behaviors like skin picking and nail biting (characterized as body-focused repetitive behavior disorders). Treatments include reassurance and education, behavior modification, or systemic therapy including tricyclic antidepressants or SSRIs. Toddlers can engage in hair pulling behavior and trichotillomania can be difficult to differentiate from AA. However, the absence of broken hairs of varying lengths makes trichotillomania less likely in this patient.
Telogen effluvium is another form of nonscarring alopecia that presents as diffuse hair thinning across the entire scalp in response to acute psychological or physiological stress, hormonal changes, certain medications, systemic illness, or nutritional deficiency. The timing between the triggering event and hair loss can vary from weeks to months. Diagnosis requires detailed history-taking and may include evaluation for endocrinologic hair thinning (e.g. thyroid function tests) to identify reversible causes. Treatment involves directing therapy to the underlying etiology and most cases of telogen effluvium are self-limited. The presence of a well-circumscribed patch of hair loss in this patient makes AA more likely.
Lichen planopilaris (LPP) is a scarring, irreversible alopecia caused by T-lymphocytes attacking follicular hair stem cells. It is characterized by hair loss, pruritus, burning pain, scalp scaling, and multifocal scarring. Exam shows patches of alopecia with loss of follicular ostia centrally and perifollicular scale and erythema at the borders. Diagnosis is aided by biopsy of the affected scalp. Treatment of LPP requires the use of potent and superpotent topical corticosteroids and intralesional corticosteroids to decrease scalp inflammation and prevent further progression. The presence of follicular ostia and absence of perifollicular scale in this patient makes LPP highly unlikely.
Tinea capitis is a fungal infection of the scalp caused by dermatophytes including Trychophyton tonsurans and Microsporum canis. It presents with patches of alopecia with overlying scale and broken hairs and can have associated cervical and occipital lymphadenopathy. Diagnosis can involve skin scraping and KOH prep to visualize branching hyphae as well as fungal culture to identify the causative organism. Because dermatophytes in tinea capitis invade hair follicles, topical antifungals are ineffective because of their lack of penetration. Therefore, systemic antifungals including oral terbinafine and griseofulvin are considered first-line agents for treatment.
What’s the management plan?
The diagnosis of AA is usually a clinical one, though assessment of alternative diagnoses is appropriate dependent on signs and symptoms. Workup of AA can include thyroid studies because of the association with autoimmune thyroid disease, though studies suggest limited screening benefits in children.11 Given its variable and unpredictable course, management can include “watchful waiting” because of its potential for spontaneous remission.6 For limited patchy loss, active treatment with mid to superpotent topical steroids or intralesional triamcinolone acetonide in older children and adolescents is reasonable.12 Other treatment options include topical or low-dose oral minoxidil and immunotherapy with diphenylcyclopropenone or squaric acid (inducing an allergic contact dermatitis).12 Management of therapies for more extensive AA is evolving, with ongoing studies of oral JAK-inhibitors and biologic agents.12,13
Our patient was started on topical fluocinonide 0.05% solution and achieved good disease control and hair regrowth over the course of 3 months.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology at the university and Rady Children’s Hospital. They had no disclosures.
References
1. Bernardez C et al. Actas Dermosifiliogr. 2015;106(3):158-67.
2. Rajabi F et al. Br J Dermatol. 2018;179(5):1033-48.
3. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):1-12.
4. Lee S et al. J Am Acad Dermatol. 2019;80(2):466-77 e16.
5. MacLean KJ and Tidman MJ. Practitioner. 2013;257(1764):29-32, 3.
6. Pratt CH et al. Nat Rev Dis Primers. 2017;3:17011.
7. Gilhar A et al. N Engl J Med. 2012;366(16):1515-25.
8. Wyrwich KW et al. Am J Clin Dermatol. 2020;21(5):725-32.
9. Spano F and Donovan JC. Can Fam Physician. 2015;61(9):751-5.
10. Mounsey AL and Reed SW. Am Fam Physician. 2009;80(4):356-62.
11. Hordinsky MK. J Investig Dermatol Symp Proc. 2015;17(2):44-6.
12. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):15-24.
13. Zhou C et al. Clin Rev Allergy Immunol. 2021;61(3):403-23.
Given the history of sudden hair loss, with the exam revealing a well-circumscribed patch of focal alopecia without cutaneous inflammation, hairs with a narrow base and broad distal shaft, the diagnosis is alopecia areata (AA).
Alopecia areata (AA) is a nonscarring alopecia, within a set of diseases characterized by the preservation of hair follicles and therefore the potential for future hair regrowth.1 AA is believed to be caused by a breakdown of the immune-privileged nature of hair follicles, resulting in T-lymphocytes targeting the hair follicle directly, shifting follicles to early catagen or telogen phase, but sparing follicular stem cells, thereby allowing the follicle to regenerate in the future.1-3 Risk factors include family history of AA, thyroid disorders, as well as iron and vitamin D deficiency.4,5 It characteristically presents with focal, well-demarcated patches of hair loss in the scalp, typically with background skin normal to slightly pink.3,6 Exam can show “exclamation point” hairs consisting of hairs that are narrow at their base and wide at the distal end.3,7 Patients may also exhibit eyebrow and eyelash loss as well as nail changes including nail pitting and splitting.8 Diagnosis is typically made clinically but is supported by a positive hair pull test, where hairs are pulled from the periphery of an alopecic lesion; the presence of greater than 10% of hairs plucked from the scalp indicates a positive result.9,10
What’s the differential diagnosis?
The differential diagnosis of AA includes other nonscarring alopecias such as trichotillomania and telogen effluvium. Other possible diagnoses include lichen planopilaris and tinea capitis.
Trichotillomania results in irregularly bordered hair loss and broken hairs of different lengths because of an internal urge to remove one’s hair, resulting in nonscarring alopecia. It can be associated with obsessive-compulsive disorder, anxiety, or other body-altering behaviors like skin picking and nail biting (characterized as body-focused repetitive behavior disorders). Treatments include reassurance and education, behavior modification, or systemic therapy including tricyclic antidepressants or SSRIs. Toddlers can engage in hair pulling behavior and trichotillomania can be difficult to differentiate from AA. However, the absence of broken hairs of varying lengths makes trichotillomania less likely in this patient.
Telogen effluvium is another form of nonscarring alopecia that presents as diffuse hair thinning across the entire scalp in response to acute psychological or physiological stress, hormonal changes, certain medications, systemic illness, or nutritional deficiency. The timing between the triggering event and hair loss can vary from weeks to months. Diagnosis requires detailed history-taking and may include evaluation for endocrinologic hair thinning (e.g. thyroid function tests) to identify reversible causes. Treatment involves directing therapy to the underlying etiology and most cases of telogen effluvium are self-limited. The presence of a well-circumscribed patch of hair loss in this patient makes AA more likely.
Lichen planopilaris (LPP) is a scarring, irreversible alopecia caused by T-lymphocytes attacking follicular hair stem cells. It is characterized by hair loss, pruritus, burning pain, scalp scaling, and multifocal scarring. Exam shows patches of alopecia with loss of follicular ostia centrally and perifollicular scale and erythema at the borders. Diagnosis is aided by biopsy of the affected scalp. Treatment of LPP requires the use of potent and superpotent topical corticosteroids and intralesional corticosteroids to decrease scalp inflammation and prevent further progression. The presence of follicular ostia and absence of perifollicular scale in this patient makes LPP highly unlikely.
Tinea capitis is a fungal infection of the scalp caused by dermatophytes including Trychophyton tonsurans and Microsporum canis. It presents with patches of alopecia with overlying scale and broken hairs and can have associated cervical and occipital lymphadenopathy. Diagnosis can involve skin scraping and KOH prep to visualize branching hyphae as well as fungal culture to identify the causative organism. Because dermatophytes in tinea capitis invade hair follicles, topical antifungals are ineffective because of their lack of penetration. Therefore, systemic antifungals including oral terbinafine and griseofulvin are considered first-line agents for treatment.
What’s the management plan?
The diagnosis of AA is usually a clinical one, though assessment of alternative diagnoses is appropriate dependent on signs and symptoms. Workup of AA can include thyroid studies because of the association with autoimmune thyroid disease, though studies suggest limited screening benefits in children.11 Given its variable and unpredictable course, management can include “watchful waiting” because of its potential for spontaneous remission.6 For limited patchy loss, active treatment with mid to superpotent topical steroids or intralesional triamcinolone acetonide in older children and adolescents is reasonable.12 Other treatment options include topical or low-dose oral minoxidil and immunotherapy with diphenylcyclopropenone or squaric acid (inducing an allergic contact dermatitis).12 Management of therapies for more extensive AA is evolving, with ongoing studies of oral JAK-inhibitors and biologic agents.12,13
Our patient was started on topical fluocinonide 0.05% solution and achieved good disease control and hair regrowth over the course of 3 months.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology at the university and Rady Children’s Hospital. They had no disclosures.
References
1. Bernardez C et al. Actas Dermosifiliogr. 2015;106(3):158-67.
2. Rajabi F et al. Br J Dermatol. 2018;179(5):1033-48.
3. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):1-12.
4. Lee S et al. J Am Acad Dermatol. 2019;80(2):466-77 e16.
5. MacLean KJ and Tidman MJ. Practitioner. 2013;257(1764):29-32, 3.
6. Pratt CH et al. Nat Rev Dis Primers. 2017;3:17011.
7. Gilhar A et al. N Engl J Med. 2012;366(16):1515-25.
8. Wyrwich KW et al. Am J Clin Dermatol. 2020;21(5):725-32.
9. Spano F and Donovan JC. Can Fam Physician. 2015;61(9):751-5.
10. Mounsey AL and Reed SW. Am Fam Physician. 2009;80(4):356-62.
11. Hordinsky MK. J Investig Dermatol Symp Proc. 2015;17(2):44-6.
12. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):15-24.
13. Zhou C et al. Clin Rev Allergy Immunol. 2021;61(3):403-23.
Examination findings of the scalp demonstrate a well-circumscribed alopecic patch on the vertex scalp without erythema or scale. Closer inspection of the patch with magnification or 'dermoscopy' reveals hair follicle ostia and hairs that are broader distally and narrower at their base. Nails and rest of the skin exam are unremarkable.
Religious fundamentalism and later-life anxiety
I was a resident, young and naive, when I bumped into my neighbor in the hospital hallway as he walked out of a psychiatrist’s office.
“Why are you here?” I asked, thinking that my neighbor, a theology professor, had some professional reason to be meeting with a psychiatrist, perhaps some type of community project. As the question escaped from my lips, however, I had an instant sense of regret and made a “note to self” in bold, all capital letters with a few exclamation points: Don’t ever ask friends or neighbors why they are visiting a psychiatrist.
Fast-forward a number of decades, and I received an email from that neighbor. Charles Marsh, is now a professor of religious studies at the University of Virginia, Charlottesville, director of the Lived Theology Project, and author of several books. He sent me a link to an article he’d written about his treatment for an anxiety disorder and let me know he was working on a book on the topic. I later received the galleys for his manuscript, Evangelical Anxiety: A Memoir, which was released last month by HarperOne.
Professor Marsh opens his story as he’s sitting with his family in church, listening to his pastor’s sermon. It is a quiet April day, and as they are throughout this memoir, his descriptions are so vivid that the reader is sitting next to him in his familiar pew, there in that church on that Sunday, seeing what he sees, smelling what he smells, and feeling what he feels. The pastor confers a wish on his congregants: He’d like them all to have a nervous breakdown in their youth. He goes on to say that if Martin Luther had lived in the days of Prozac, his inner torment would have been quelled, and there would have been no Protestant reformation. Professor Marsh then treats us to the first of many humorous moments – he rushes home and swallows a tablet of Ativan.
Professor Marsh focuses on a single dividing point for his life, a day in the fall of 1981. He was resting on his bed in his dorm room at Harvard Divinity School at the ripe age of 23 years, 6 months, and 3 days (but who’s counting), when all of who he was changed. He described what he went through that night:
It was then that a high pandemonium ripped away everything protecting me from the world outside. I was no longer a person alone in his room. In an instant, I could hear all things inside my body in their deepest repercussions. My heart and its soft aortic murmur, my breath’s every exhalation and inhalation, the downward silences, the laborious intake – would this one be the last? How much noise the body makes when amped up on fear! I could hear the hiss of molecules colliding. And outside in the yellow night, the compressors harrumphing atop the nearby physics building, the sound of car engines and slamming doors. All these things I heard as tormenting assault, a soundscape I could not mute. I’d become a thought thinking about thinking itself and nothing else, metaphysics’ ancient curse. A cogitation cycling through every autonomous body function, placing on each a question mark like flowers for the dead.
This moment in time – this “breakdown,” as Professor Marsh repeatedly refers to it – bifurcated his life. He went from being a person who lived “disguised to myself as unaghast and free” to someone who could no longer find escape in his reading, who struggled in his own skin and his own mind, and who, for lack of a better description, was tortured. The “breakdown” passed, and Professor Marsh diagnosed himself with generalized anxiety disorder.
That night, he did not go to an emergency department nor did he seek help from services that were available to Harvard students. There was no psychiatrist, no therapy, no medication. It was, for him, with his fundamentalist Christian background, a religious event of sorts.
I counted it all joy if I should suffer. My sorrow, my soul’s sin-sickness, was not unintelligible – it was a kind of blessing, something that might draw me, like a medieval saint, to the suffering of my Lord, something that would testify wordlessly to my heroic exertion to attain purity. And, at least during those late days of autumn 1981, the heavens above and the earth below, spirit and flesh, felt miraculously aligned. Though suffering, this was the life I had craved.
Charles Marsh grew up as a Baptist pastor’s son in the Deep South during a time when the civil rights movement came to a head, and life was marked by fear and change. The memoir is not simply about one man’s struggle with an anxiety disorder, but a beautifully written account of life as an evangelical Christian during a tumultuous time of racial tensions and horrible violence. He details his life as a lonely only child in a God-fearing world cast in dark shadows, one where he struggled to belong and called out to his mother in the nights. Inside this world, Professor Marsh searched for his own religious identity, with the pride of being a high school “Jesus freak,” running alongside his repressed and frustrated sexual longings.
It was a world of good and bad, of heaven and hell, only the two became so confused as he talked about his existence full of fears: The windows were barred; violence and fear were central in his Alabama hometown, “the epicenter of white terrorism,” and then later when his family moved to Mississippi. He feared the barking dogs that guarded the houses, the bullies who tormented him, and the bullying in which, he too, joined in. He feared the switch-wielding adults – his mother, his principals, his coaches, and his youth pastor, all set on “breaking the will of the child,” a term he explains to be a Christian concept in which the child’s own will is broken so that he will be submissive to his parents and to God.
Professor Marsh wanted so much to be good. And we’re not sure he even knew what that was as he battled his desire to conform and belong, and his ever-present sexual impulses. Even as an adult, he was certain his mother would know if he had premarital sex and he would have to kill himself. Sex outside of marriage was the one unpardonable sin.
He suffered in silence and shame. It was not until a few years later that he entered psychotherapy as a doctoral student. When he moved to Baltimore, he again looked for a therapist and eventually found himself with a psychiatrist who was training to be a psychoanalyst in the hospital where I was a resident. This psychoanalysis proved to be transformative and healing, but first, Professor Marsh needed to reconcile his treatment with his religious beliefs, as therapy and fundamental religion travel different roads.
Analysis and faith traverse similar terrain – they understand how language and narrative heal. They may see each other as strangers or competitors, but they need not. Like prayer, the analytic dialogue slows down to ponder, to meander, to piece together, to redeem; both inspire the mind toward hope under the influence of an empathetic listener. Neither needs the other to effectuate its truths, but they follow parallel tracks into the mysteries of being human, where all truth is God’s truth. It’s more than fine that they neither merge nor collide.
He goes on to describe how powerful the process was for him and his healing.
Analysis is the space where one feels – where I felt in an embodied way, in the unhurried hours over months and years – a trust in the beautiful interplay between the center and the extremes. My body and mind would not be raised in resurrected splendor in the course of the treatment. I wish to emphasize the point. It was tempting to think that it would, that I would undergo a miraculous transformation. If not resurrected splendor, then surely I would take on the “new man.” Instead, I received the gift of mortal life: the freedom to be imperfect, to have fears and face them, to accept brokenness, to let go of the will to control all outcomes.
Professor Marsh’s use of language is extraordinary; he has a gift for metaphors and descriptions, and he carries the reader alongside him on a splendid journey. It has to be said, however, that he assumes a lot: He is a sophisticated scholar who mentions religious leaders, philosophers, historical characters, and the occasional rock song, with no patience for those who don’t follow his quick transitions and impressive vocabulary; I could have read this book with a dictionary beside me (but I didn’t).
It’s an illuminating journey, often sad and disturbing, sometimes funny and endearing, and ultimately uplifting. In our skeptical world where psychiatrists are so are often undone, it is refreshing to read a memoir where the psychiatrist is the good guy and the patient emerges healed and whole.
Dr. Miller, is a coauthor of Committed: The Battle Over Involuntary Psychiatric Care (Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins in Baltimore. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
I was a resident, young and naive, when I bumped into my neighbor in the hospital hallway as he walked out of a psychiatrist’s office.
“Why are you here?” I asked, thinking that my neighbor, a theology professor, had some professional reason to be meeting with a psychiatrist, perhaps some type of community project. As the question escaped from my lips, however, I had an instant sense of regret and made a “note to self” in bold, all capital letters with a few exclamation points: Don’t ever ask friends or neighbors why they are visiting a psychiatrist.
Fast-forward a number of decades, and I received an email from that neighbor. Charles Marsh, is now a professor of religious studies at the University of Virginia, Charlottesville, director of the Lived Theology Project, and author of several books. He sent me a link to an article he’d written about his treatment for an anxiety disorder and let me know he was working on a book on the topic. I later received the galleys for his manuscript, Evangelical Anxiety: A Memoir, which was released last month by HarperOne.
Professor Marsh opens his story as he’s sitting with his family in church, listening to his pastor’s sermon. It is a quiet April day, and as they are throughout this memoir, his descriptions are so vivid that the reader is sitting next to him in his familiar pew, there in that church on that Sunday, seeing what he sees, smelling what he smells, and feeling what he feels. The pastor confers a wish on his congregants: He’d like them all to have a nervous breakdown in their youth. He goes on to say that if Martin Luther had lived in the days of Prozac, his inner torment would have been quelled, and there would have been no Protestant reformation. Professor Marsh then treats us to the first of many humorous moments – he rushes home and swallows a tablet of Ativan.
Professor Marsh focuses on a single dividing point for his life, a day in the fall of 1981. He was resting on his bed in his dorm room at Harvard Divinity School at the ripe age of 23 years, 6 months, and 3 days (but who’s counting), when all of who he was changed. He described what he went through that night:
It was then that a high pandemonium ripped away everything protecting me from the world outside. I was no longer a person alone in his room. In an instant, I could hear all things inside my body in their deepest repercussions. My heart and its soft aortic murmur, my breath’s every exhalation and inhalation, the downward silences, the laborious intake – would this one be the last? How much noise the body makes when amped up on fear! I could hear the hiss of molecules colliding. And outside in the yellow night, the compressors harrumphing atop the nearby physics building, the sound of car engines and slamming doors. All these things I heard as tormenting assault, a soundscape I could not mute. I’d become a thought thinking about thinking itself and nothing else, metaphysics’ ancient curse. A cogitation cycling through every autonomous body function, placing on each a question mark like flowers for the dead.
This moment in time – this “breakdown,” as Professor Marsh repeatedly refers to it – bifurcated his life. He went from being a person who lived “disguised to myself as unaghast and free” to someone who could no longer find escape in his reading, who struggled in his own skin and his own mind, and who, for lack of a better description, was tortured. The “breakdown” passed, and Professor Marsh diagnosed himself with generalized anxiety disorder.
That night, he did not go to an emergency department nor did he seek help from services that were available to Harvard students. There was no psychiatrist, no therapy, no medication. It was, for him, with his fundamentalist Christian background, a religious event of sorts.
I counted it all joy if I should suffer. My sorrow, my soul’s sin-sickness, was not unintelligible – it was a kind of blessing, something that might draw me, like a medieval saint, to the suffering of my Lord, something that would testify wordlessly to my heroic exertion to attain purity. And, at least during those late days of autumn 1981, the heavens above and the earth below, spirit and flesh, felt miraculously aligned. Though suffering, this was the life I had craved.
Charles Marsh grew up as a Baptist pastor’s son in the Deep South during a time when the civil rights movement came to a head, and life was marked by fear and change. The memoir is not simply about one man’s struggle with an anxiety disorder, but a beautifully written account of life as an evangelical Christian during a tumultuous time of racial tensions and horrible violence. He details his life as a lonely only child in a God-fearing world cast in dark shadows, one where he struggled to belong and called out to his mother in the nights. Inside this world, Professor Marsh searched for his own religious identity, with the pride of being a high school “Jesus freak,” running alongside his repressed and frustrated sexual longings.
It was a world of good and bad, of heaven and hell, only the two became so confused as he talked about his existence full of fears: The windows were barred; violence and fear were central in his Alabama hometown, “the epicenter of white terrorism,” and then later when his family moved to Mississippi. He feared the barking dogs that guarded the houses, the bullies who tormented him, and the bullying in which, he too, joined in. He feared the switch-wielding adults – his mother, his principals, his coaches, and his youth pastor, all set on “breaking the will of the child,” a term he explains to be a Christian concept in which the child’s own will is broken so that he will be submissive to his parents and to God.
Professor Marsh wanted so much to be good. And we’re not sure he even knew what that was as he battled his desire to conform and belong, and his ever-present sexual impulses. Even as an adult, he was certain his mother would know if he had premarital sex and he would have to kill himself. Sex outside of marriage was the one unpardonable sin.
He suffered in silence and shame. It was not until a few years later that he entered psychotherapy as a doctoral student. When he moved to Baltimore, he again looked for a therapist and eventually found himself with a psychiatrist who was training to be a psychoanalyst in the hospital where I was a resident. This psychoanalysis proved to be transformative and healing, but first, Professor Marsh needed to reconcile his treatment with his religious beliefs, as therapy and fundamental religion travel different roads.
Analysis and faith traverse similar terrain – they understand how language and narrative heal. They may see each other as strangers or competitors, but they need not. Like prayer, the analytic dialogue slows down to ponder, to meander, to piece together, to redeem; both inspire the mind toward hope under the influence of an empathetic listener. Neither needs the other to effectuate its truths, but they follow parallel tracks into the mysteries of being human, where all truth is God’s truth. It’s more than fine that they neither merge nor collide.
He goes on to describe how powerful the process was for him and his healing.
Analysis is the space where one feels – where I felt in an embodied way, in the unhurried hours over months and years – a trust in the beautiful interplay between the center and the extremes. My body and mind would not be raised in resurrected splendor in the course of the treatment. I wish to emphasize the point. It was tempting to think that it would, that I would undergo a miraculous transformation. If not resurrected splendor, then surely I would take on the “new man.” Instead, I received the gift of mortal life: the freedom to be imperfect, to have fears and face them, to accept brokenness, to let go of the will to control all outcomes.
Professor Marsh’s use of language is extraordinary; he has a gift for metaphors and descriptions, and he carries the reader alongside him on a splendid journey. It has to be said, however, that he assumes a lot: He is a sophisticated scholar who mentions religious leaders, philosophers, historical characters, and the occasional rock song, with no patience for those who don’t follow his quick transitions and impressive vocabulary; I could have read this book with a dictionary beside me (but I didn’t).
It’s an illuminating journey, often sad and disturbing, sometimes funny and endearing, and ultimately uplifting. In our skeptical world where psychiatrists are so are often undone, it is refreshing to read a memoir where the psychiatrist is the good guy and the patient emerges healed and whole.
Dr. Miller, is a coauthor of Committed: The Battle Over Involuntary Psychiatric Care (Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins in Baltimore. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
I was a resident, young and naive, when I bumped into my neighbor in the hospital hallway as he walked out of a psychiatrist’s office.
“Why are you here?” I asked, thinking that my neighbor, a theology professor, had some professional reason to be meeting with a psychiatrist, perhaps some type of community project. As the question escaped from my lips, however, I had an instant sense of regret and made a “note to self” in bold, all capital letters with a few exclamation points: Don’t ever ask friends or neighbors why they are visiting a psychiatrist.
Fast-forward a number of decades, and I received an email from that neighbor. Charles Marsh, is now a professor of religious studies at the University of Virginia, Charlottesville, director of the Lived Theology Project, and author of several books. He sent me a link to an article he’d written about his treatment for an anxiety disorder and let me know he was working on a book on the topic. I later received the galleys for his manuscript, Evangelical Anxiety: A Memoir, which was released last month by HarperOne.
Professor Marsh opens his story as he’s sitting with his family in church, listening to his pastor’s sermon. It is a quiet April day, and as they are throughout this memoir, his descriptions are so vivid that the reader is sitting next to him in his familiar pew, there in that church on that Sunday, seeing what he sees, smelling what he smells, and feeling what he feels. The pastor confers a wish on his congregants: He’d like them all to have a nervous breakdown in their youth. He goes on to say that if Martin Luther had lived in the days of Prozac, his inner torment would have been quelled, and there would have been no Protestant reformation. Professor Marsh then treats us to the first of many humorous moments – he rushes home and swallows a tablet of Ativan.
Professor Marsh focuses on a single dividing point for his life, a day in the fall of 1981. He was resting on his bed in his dorm room at Harvard Divinity School at the ripe age of 23 years, 6 months, and 3 days (but who’s counting), when all of who he was changed. He described what he went through that night:
It was then that a high pandemonium ripped away everything protecting me from the world outside. I was no longer a person alone in his room. In an instant, I could hear all things inside my body in their deepest repercussions. My heart and its soft aortic murmur, my breath’s every exhalation and inhalation, the downward silences, the laborious intake – would this one be the last? How much noise the body makes when amped up on fear! I could hear the hiss of molecules colliding. And outside in the yellow night, the compressors harrumphing atop the nearby physics building, the sound of car engines and slamming doors. All these things I heard as tormenting assault, a soundscape I could not mute. I’d become a thought thinking about thinking itself and nothing else, metaphysics’ ancient curse. A cogitation cycling through every autonomous body function, placing on each a question mark like flowers for the dead.
This moment in time – this “breakdown,” as Professor Marsh repeatedly refers to it – bifurcated his life. He went from being a person who lived “disguised to myself as unaghast and free” to someone who could no longer find escape in his reading, who struggled in his own skin and his own mind, and who, for lack of a better description, was tortured. The “breakdown” passed, and Professor Marsh diagnosed himself with generalized anxiety disorder.
That night, he did not go to an emergency department nor did he seek help from services that were available to Harvard students. There was no psychiatrist, no therapy, no medication. It was, for him, with his fundamentalist Christian background, a religious event of sorts.
I counted it all joy if I should suffer. My sorrow, my soul’s sin-sickness, was not unintelligible – it was a kind of blessing, something that might draw me, like a medieval saint, to the suffering of my Lord, something that would testify wordlessly to my heroic exertion to attain purity. And, at least during those late days of autumn 1981, the heavens above and the earth below, spirit and flesh, felt miraculously aligned. Though suffering, this was the life I had craved.
Charles Marsh grew up as a Baptist pastor’s son in the Deep South during a time when the civil rights movement came to a head, and life was marked by fear and change. The memoir is not simply about one man’s struggle with an anxiety disorder, but a beautifully written account of life as an evangelical Christian during a tumultuous time of racial tensions and horrible violence. He details his life as a lonely only child in a God-fearing world cast in dark shadows, one where he struggled to belong and called out to his mother in the nights. Inside this world, Professor Marsh searched for his own religious identity, with the pride of being a high school “Jesus freak,” running alongside his repressed and frustrated sexual longings.
It was a world of good and bad, of heaven and hell, only the two became so confused as he talked about his existence full of fears: The windows were barred; violence and fear were central in his Alabama hometown, “the epicenter of white terrorism,” and then later when his family moved to Mississippi. He feared the barking dogs that guarded the houses, the bullies who tormented him, and the bullying in which, he too, joined in. He feared the switch-wielding adults – his mother, his principals, his coaches, and his youth pastor, all set on “breaking the will of the child,” a term he explains to be a Christian concept in which the child’s own will is broken so that he will be submissive to his parents and to God.
Professor Marsh wanted so much to be good. And we’re not sure he even knew what that was as he battled his desire to conform and belong, and his ever-present sexual impulses. Even as an adult, he was certain his mother would know if he had premarital sex and he would have to kill himself. Sex outside of marriage was the one unpardonable sin.
He suffered in silence and shame. It was not until a few years later that he entered psychotherapy as a doctoral student. When he moved to Baltimore, he again looked for a therapist and eventually found himself with a psychiatrist who was training to be a psychoanalyst in the hospital where I was a resident. This psychoanalysis proved to be transformative and healing, but first, Professor Marsh needed to reconcile his treatment with his religious beliefs, as therapy and fundamental religion travel different roads.
Analysis and faith traverse similar terrain – they understand how language and narrative heal. They may see each other as strangers or competitors, but they need not. Like prayer, the analytic dialogue slows down to ponder, to meander, to piece together, to redeem; both inspire the mind toward hope under the influence of an empathetic listener. Neither needs the other to effectuate its truths, but they follow parallel tracks into the mysteries of being human, where all truth is God’s truth. It’s more than fine that they neither merge nor collide.
He goes on to describe how powerful the process was for him and his healing.
Analysis is the space where one feels – where I felt in an embodied way, in the unhurried hours over months and years – a trust in the beautiful interplay between the center and the extremes. My body and mind would not be raised in resurrected splendor in the course of the treatment. I wish to emphasize the point. It was tempting to think that it would, that I would undergo a miraculous transformation. If not resurrected splendor, then surely I would take on the “new man.” Instead, I received the gift of mortal life: the freedom to be imperfect, to have fears and face them, to accept brokenness, to let go of the will to control all outcomes.
Professor Marsh’s use of language is extraordinary; he has a gift for metaphors and descriptions, and he carries the reader alongside him on a splendid journey. It has to be said, however, that he assumes a lot: He is a sophisticated scholar who mentions religious leaders, philosophers, historical characters, and the occasional rock song, with no patience for those who don’t follow his quick transitions and impressive vocabulary; I could have read this book with a dictionary beside me (but I didn’t).
It’s an illuminating journey, often sad and disturbing, sometimes funny and endearing, and ultimately uplifting. In our skeptical world where psychiatrists are so are often undone, it is refreshing to read a memoir where the psychiatrist is the good guy and the patient emerges healed and whole.
Dr. Miller, is a coauthor of Committed: The Battle Over Involuntary Psychiatric Care (Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins in Baltimore. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
Who’s in charge here? Rheumatologists battle local Medicare policies threatening access, practice sustainability
Rheumatologists who administer medications in their office for Medicare patients, specifically those that are infused, have in recent years encountered problems providing certain medication formulations as well as coding and billing for their administration. In attempting to resolve these issues, rheumatologists and their professional organizations have found themselves caught in a morass of Medicare agency “ping-pong,” where it is unclear who the decision makers are.
The private health care insurers that process medical claims for Medicare beneficiaries, called A/B Medicare Administrative Contractors or more commonly known as MACs, are the operational intermediary between the Centers for Medicare & Medicaid Services’ fee-for-service program and the physicians enrolled in it. The country is divided into 12 sections, each with a MAC that has jurisdiction over that area. Among other things, the MACs establish local coverage and payment policies based on their understanding of CMS’ rules, regulations, and the Medicare statute, and therein lies the problem: When a physician has a question on a policy or decision that was made by a MAC, it is very difficult to determine the origins of the issue and who can address the problem. It’s a lot of “running in circles” between the MACs and CMS headquarters, hoping that someone will take the time to listen to your concern, but more importantly, work toward resolving the problem.
Who can address problems?
Meaningful, solutions-driven engagement with the MACs and CMS has become frustrating for physicians and advocacy organizations attempting to address a host of problems. The two issues alluded to above include the Self-Administered Drug Exclusion List (SAD List), which excludes certain Part B medication formulations from coverage under certain conditions and the “down coding” of certain infusion administration codes when specific drugs are delivered. These problems are compounded by the curtailment of physician stakeholder input via Contractor Advisory Committees (CACs). Each state has its own CAC, but the CAC meetings have been restructured as a result of the 21st Century Cures Act, and ultimately eradicated the involvement of these physician advisers in policy development at the local level.
This has left many of rheumatology representatives to the CACs demoralized and generally unhappy about certain decisions being made without their input. There is also inconsistency in terms of coverage and payment policies throughout the country. For example, in one MAC jurisdiction, a certain medication may be on the SAD List and excluded from Part B coverage, meaning beneficiary access is only available through Part D (and assuming they can afford it), while in an adjacent MAC jurisdiction, both formulations are covered.
The Coalition of State Rheumatology Organizations, along with the American College of Rheumatology and other specialty groups, is attempting to address these issues from many different angles. There is not enough space to explain the nuances of local coverage policy development, but the timeline below highlights the long and winding road that we have travelled to resolve these issues.
- February 2021: CSRO meets with CMS’ Coverage and Analysis Group (CAG) to raise concerns about ustekinumab (Stelara) and its inclusion on the SAD List.
- April 2021: CSRO follows up with CMS’ CAG on SAD List concerns in a letter.
- May 2021: Most MACs issue or revise local coverage articles, or “billing and coding” articles, that down code the administration of certain biologic medications, with some expanding the list of biologic medications subject to the policy, prompting a strong response from CSRO.
- September 2021: CSRO meets with multijurisdictional MAC Contract Medical Director (CMD) work group to discuss down coding, SAD List, and physician/CAC engagement.
- October 2021: At the suggestion of the CMDs, CSRO re-engages with CMS’ CAG to raise concerns about down-coding policies and physician/CAC engagement, and continue the SAD List discussion.
- November 2021: CSRO is connected with CMS’ “payment ombudsman” on down coding and the SAD List.
- January 2022: CSRO signs on to multispecialty coalition effort aimed at improving local coverage and payment policy and restoring the importance of the CAC.
- February 2022: CSRO participates in CMS CAG meeting with multispecialty coalition, raising concerns about the down-coding and SAD List policies.
- March/April 2022: Through its coalition partner, the Alliance of Specialty Medicine, CSRO meets with the principal deputy CMS administrator and raises awareness to these issues.
- May 2022: CSRO participates in follow-up discussion with CMS’ CAG as part of multispecialty coalition, reiterating concerns about the down coding and SAD List policies. With the assistance of the CMS’ Office of the Administrator, CSRO meets with CMS’ Center for Program Integrity to seek a “pause” in down-coding policies for certain biologic medications.
- June 2022: CMS notifies CSRO of a “temporary pause” in medical review while the agency reviews various manuals and policies to determine the appropriate steps forward. To assist the agency, CSRO works with practices to develop a resource that CMS can use to establish criteria for determining when a medication warrants use of complex drug administration codes. CSRO re-engages with multijurisdictional MAC CMD workgroup to continue discussions on SAD List.
- July 2022: CSRO meets with new multijurisdictional MAC CMD workgroup focused on improving the process for developing local coverage and payment policy.
Our dialogue with CMS leadership and staff continues. In the most recent communication, staff in the CMS administrator’s office informed us that the issue is complicated and crosses several different parts of the agency, and they are still determining next steps.
The rheumatology community’s journey toward solving the challenges facing practices and patients is emblematic of the communication problem between provider groups and the CMS-MAC establishment. While we understand this is how bureaucracy works, it is not to the benefit of Medicare beneficiaries to have a system that is so difficult to navigate, even by the best of the regulatory gurus. This is not an indictment of any specific group but a call to action on the part of the government and their contractors to create a clear, transparent path to getting answers when we have a problem.
And by the way, we do have another meeting with yet another CMS “center” regarding the SAD List in August.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is president of the CSRO, past chair of the Alliance for Safe Biologic Medicines, and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Rheumatologists who administer medications in their office for Medicare patients, specifically those that are infused, have in recent years encountered problems providing certain medication formulations as well as coding and billing for their administration. In attempting to resolve these issues, rheumatologists and their professional organizations have found themselves caught in a morass of Medicare agency “ping-pong,” where it is unclear who the decision makers are.
The private health care insurers that process medical claims for Medicare beneficiaries, called A/B Medicare Administrative Contractors or more commonly known as MACs, are the operational intermediary between the Centers for Medicare & Medicaid Services’ fee-for-service program and the physicians enrolled in it. The country is divided into 12 sections, each with a MAC that has jurisdiction over that area. Among other things, the MACs establish local coverage and payment policies based on their understanding of CMS’ rules, regulations, and the Medicare statute, and therein lies the problem: When a physician has a question on a policy or decision that was made by a MAC, it is very difficult to determine the origins of the issue and who can address the problem. It’s a lot of “running in circles” between the MACs and CMS headquarters, hoping that someone will take the time to listen to your concern, but more importantly, work toward resolving the problem.
Who can address problems?
Meaningful, solutions-driven engagement with the MACs and CMS has become frustrating for physicians and advocacy organizations attempting to address a host of problems. The two issues alluded to above include the Self-Administered Drug Exclusion List (SAD List), which excludes certain Part B medication formulations from coverage under certain conditions and the “down coding” of certain infusion administration codes when specific drugs are delivered. These problems are compounded by the curtailment of physician stakeholder input via Contractor Advisory Committees (CACs). Each state has its own CAC, but the CAC meetings have been restructured as a result of the 21st Century Cures Act, and ultimately eradicated the involvement of these physician advisers in policy development at the local level.
This has left many of rheumatology representatives to the CACs demoralized and generally unhappy about certain decisions being made without their input. There is also inconsistency in terms of coverage and payment policies throughout the country. For example, in one MAC jurisdiction, a certain medication may be on the SAD List and excluded from Part B coverage, meaning beneficiary access is only available through Part D (and assuming they can afford it), while in an adjacent MAC jurisdiction, both formulations are covered.
The Coalition of State Rheumatology Organizations, along with the American College of Rheumatology and other specialty groups, is attempting to address these issues from many different angles. There is not enough space to explain the nuances of local coverage policy development, but the timeline below highlights the long and winding road that we have travelled to resolve these issues.
- February 2021: CSRO meets with CMS’ Coverage and Analysis Group (CAG) to raise concerns about ustekinumab (Stelara) and its inclusion on the SAD List.
- April 2021: CSRO follows up with CMS’ CAG on SAD List concerns in a letter.
- May 2021: Most MACs issue or revise local coverage articles, or “billing and coding” articles, that down code the administration of certain biologic medications, with some expanding the list of biologic medications subject to the policy, prompting a strong response from CSRO.
- September 2021: CSRO meets with multijurisdictional MAC Contract Medical Director (CMD) work group to discuss down coding, SAD List, and physician/CAC engagement.
- October 2021: At the suggestion of the CMDs, CSRO re-engages with CMS’ CAG to raise concerns about down-coding policies and physician/CAC engagement, and continue the SAD List discussion.
- November 2021: CSRO is connected with CMS’ “payment ombudsman” on down coding and the SAD List.
- January 2022: CSRO signs on to multispecialty coalition effort aimed at improving local coverage and payment policy and restoring the importance of the CAC.
- February 2022: CSRO participates in CMS CAG meeting with multispecialty coalition, raising concerns about the down-coding and SAD List policies.
- March/April 2022: Through its coalition partner, the Alliance of Specialty Medicine, CSRO meets with the principal deputy CMS administrator and raises awareness to these issues.
- May 2022: CSRO participates in follow-up discussion with CMS’ CAG as part of multispecialty coalition, reiterating concerns about the down coding and SAD List policies. With the assistance of the CMS’ Office of the Administrator, CSRO meets with CMS’ Center for Program Integrity to seek a “pause” in down-coding policies for certain biologic medications.
- June 2022: CMS notifies CSRO of a “temporary pause” in medical review while the agency reviews various manuals and policies to determine the appropriate steps forward. To assist the agency, CSRO works with practices to develop a resource that CMS can use to establish criteria for determining when a medication warrants use of complex drug administration codes. CSRO re-engages with multijurisdictional MAC CMD workgroup to continue discussions on SAD List.
- July 2022: CSRO meets with new multijurisdictional MAC CMD workgroup focused on improving the process for developing local coverage and payment policy.
Our dialogue with CMS leadership and staff continues. In the most recent communication, staff in the CMS administrator’s office informed us that the issue is complicated and crosses several different parts of the agency, and they are still determining next steps.
The rheumatology community’s journey toward solving the challenges facing practices and patients is emblematic of the communication problem between provider groups and the CMS-MAC establishment. While we understand this is how bureaucracy works, it is not to the benefit of Medicare beneficiaries to have a system that is so difficult to navigate, even by the best of the regulatory gurus. This is not an indictment of any specific group but a call to action on the part of the government and their contractors to create a clear, transparent path to getting answers when we have a problem.
And by the way, we do have another meeting with yet another CMS “center” regarding the SAD List in August.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is president of the CSRO, past chair of the Alliance for Safe Biologic Medicines, and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Rheumatologists who administer medications in their office for Medicare patients, specifically those that are infused, have in recent years encountered problems providing certain medication formulations as well as coding and billing for their administration. In attempting to resolve these issues, rheumatologists and their professional organizations have found themselves caught in a morass of Medicare agency “ping-pong,” where it is unclear who the decision makers are.
The private health care insurers that process medical claims for Medicare beneficiaries, called A/B Medicare Administrative Contractors or more commonly known as MACs, are the operational intermediary between the Centers for Medicare & Medicaid Services’ fee-for-service program and the physicians enrolled in it. The country is divided into 12 sections, each with a MAC that has jurisdiction over that area. Among other things, the MACs establish local coverage and payment policies based on their understanding of CMS’ rules, regulations, and the Medicare statute, and therein lies the problem: When a physician has a question on a policy or decision that was made by a MAC, it is very difficult to determine the origins of the issue and who can address the problem. It’s a lot of “running in circles” between the MACs and CMS headquarters, hoping that someone will take the time to listen to your concern, but more importantly, work toward resolving the problem.
Who can address problems?
Meaningful, solutions-driven engagement with the MACs and CMS has become frustrating for physicians and advocacy organizations attempting to address a host of problems. The two issues alluded to above include the Self-Administered Drug Exclusion List (SAD List), which excludes certain Part B medication formulations from coverage under certain conditions and the “down coding” of certain infusion administration codes when specific drugs are delivered. These problems are compounded by the curtailment of physician stakeholder input via Contractor Advisory Committees (CACs). Each state has its own CAC, but the CAC meetings have been restructured as a result of the 21st Century Cures Act, and ultimately eradicated the involvement of these physician advisers in policy development at the local level.
This has left many of rheumatology representatives to the CACs demoralized and generally unhappy about certain decisions being made without their input. There is also inconsistency in terms of coverage and payment policies throughout the country. For example, in one MAC jurisdiction, a certain medication may be on the SAD List and excluded from Part B coverage, meaning beneficiary access is only available through Part D (and assuming they can afford it), while in an adjacent MAC jurisdiction, both formulations are covered.
The Coalition of State Rheumatology Organizations, along with the American College of Rheumatology and other specialty groups, is attempting to address these issues from many different angles. There is not enough space to explain the nuances of local coverage policy development, but the timeline below highlights the long and winding road that we have travelled to resolve these issues.
- February 2021: CSRO meets with CMS’ Coverage and Analysis Group (CAG) to raise concerns about ustekinumab (Stelara) and its inclusion on the SAD List.
- April 2021: CSRO follows up with CMS’ CAG on SAD List concerns in a letter.
- May 2021: Most MACs issue or revise local coverage articles, or “billing and coding” articles, that down code the administration of certain biologic medications, with some expanding the list of biologic medications subject to the policy, prompting a strong response from CSRO.
- September 2021: CSRO meets with multijurisdictional MAC Contract Medical Director (CMD) work group to discuss down coding, SAD List, and physician/CAC engagement.
- October 2021: At the suggestion of the CMDs, CSRO re-engages with CMS’ CAG to raise concerns about down-coding policies and physician/CAC engagement, and continue the SAD List discussion.
- November 2021: CSRO is connected with CMS’ “payment ombudsman” on down coding and the SAD List.
- January 2022: CSRO signs on to multispecialty coalition effort aimed at improving local coverage and payment policy and restoring the importance of the CAC.
- February 2022: CSRO participates in CMS CAG meeting with multispecialty coalition, raising concerns about the down-coding and SAD List policies.
- March/April 2022: Through its coalition partner, the Alliance of Specialty Medicine, CSRO meets with the principal deputy CMS administrator and raises awareness to these issues.
- May 2022: CSRO participates in follow-up discussion with CMS’ CAG as part of multispecialty coalition, reiterating concerns about the down coding and SAD List policies. With the assistance of the CMS’ Office of the Administrator, CSRO meets with CMS’ Center for Program Integrity to seek a “pause” in down-coding policies for certain biologic medications.
- June 2022: CMS notifies CSRO of a “temporary pause” in medical review while the agency reviews various manuals and policies to determine the appropriate steps forward. To assist the agency, CSRO works with practices to develop a resource that CMS can use to establish criteria for determining when a medication warrants use of complex drug administration codes. CSRO re-engages with multijurisdictional MAC CMD workgroup to continue discussions on SAD List.
- July 2022: CSRO meets with new multijurisdictional MAC CMD workgroup focused on improving the process for developing local coverage and payment policy.
Our dialogue with CMS leadership and staff continues. In the most recent communication, staff in the CMS administrator’s office informed us that the issue is complicated and crosses several different parts of the agency, and they are still determining next steps.
The rheumatology community’s journey toward solving the challenges facing practices and patients is emblematic of the communication problem between provider groups and the CMS-MAC establishment. While we understand this is how bureaucracy works, it is not to the benefit of Medicare beneficiaries to have a system that is so difficult to navigate, even by the best of the regulatory gurus. This is not an indictment of any specific group but a call to action on the part of the government and their contractors to create a clear, transparent path to getting answers when we have a problem.
And by the way, we do have another meeting with yet another CMS “center” regarding the SAD List in August.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is president of the CSRO, past chair of the Alliance for Safe Biologic Medicines, and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
What are your weaknesses?
In a video posted to TikTok by the comedian Will Flanary, MD, better known to his followers as Dr. Glaucomflecken, he imitates a neurosurgical residency interview. With glasses perched on the bridge of his nose, Dr. Glaucomflecken poses as the attending, asking: “What are your weaknesses?”
The residency applicant answers without hesitation: “My physiological need for sleep.” “What are your strengths?” The resident replies with the hard, steely stare of the determined and uninitiated: “My desire to eliminate my physiological need for sleep.”
If you follow Dr. Glaucomflecken on Twitter, you might know the skit I’m referencing. For many physicians and physicians-in-training, what makes the satire successful is its reflection of reality.
Many things have changed in medicine since his time, but the tired trope of the sleepless surgeon hangs on. Undaunted, I spent my second and third year of medical school accumulating accolades, conducting research, and connecting with mentors with the singular goal of joining the surgical ranks.
Midway through my third year, I completed a month-long surgical subinternship designed to give students a taste of what life would look like as an intern. I loved the operating room; it felt like the difference between being on dry land and being underwater. There were fewer distractions – your patient in the spotlight while everything else receded to the shadows.
However, as the month wore on, something stronger took hold. I couldn’t keep my eyes open in the darkened operating rooms and had to decline stools, fearing that I would fall asleep if I sat down.
On early morning prerounds, it’s 4:50 a.m. when I glance at the clock and pull back the curtain, already apologizing. My patient rolls over, flashing a wry smile. “Do you ever go home?” I’ve seen residents respond to this exact question in various ways. I live here. Yes. No. Soon. Not enough. My partner doesn’t think so.
There are days and, yes, years when we are led to believe this is what we live for: to be constantly available to our patients. It feels like a hollow victory when the patient, 2 days out from a total colectomy, begins to worry about your personal life. I ask her how she slept (not enough), any fevers (no), vomiting (no), urinating (I pause – she has a catheter).
My favorite part of these early morning rounds is the pause in my scripted litany of questions to listen to heart and lungs. It never fails to feel sacred: Patients become so quiet and still that I can’t help but think they have faith in me. Without prompting, she slides the back of her hospital gown forward like a curtain, already taking deep breaths so I can hear her lungs.
I look outside. The streetlights are still on, and from the seventh-floor window, I can watch staff making their way through the sliding double-doors, just beyond the yellowed pools of streetlight. I smile. I love medicine. I’m so tired.
For many in medicine, we are treated, and thus behave, as though our ability to manipulate physiology should also apply within the borders of our bodies: commanding less sleep, food, or bathroom breaks.
It places health care workers solidly in the realm of superhuman, living beyond one’s corporeal needs. The pandemic only heightened this misappropriation – adding hero and setting out a pedestal for health care workers to make their ungainly ascent. This kind of unsolicited admiration implicitly implies inhumanness, an otherness.
What would it look like if we started treating ourselves less like physicians and more like patients? I wish I was offering a solution, but really this is just a story.
To students rising through the ranks of medical training, identify what it is you need early and often. I can count on one hand how many physicians I’ve seen take a lunch break – even 10 minutes. Embrace hard work and self-preservation equally. My hope is that if enough of us take this path, it just might become a matter of course.
Dr. Meffert is a resident in the department of emergency medicine, MedStar Georgetown University Hospital, Washington Hospital Center, Washington. Dr. Meffert disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a video posted to TikTok by the comedian Will Flanary, MD, better known to his followers as Dr. Glaucomflecken, he imitates a neurosurgical residency interview. With glasses perched on the bridge of his nose, Dr. Glaucomflecken poses as the attending, asking: “What are your weaknesses?”
The residency applicant answers without hesitation: “My physiological need for sleep.” “What are your strengths?” The resident replies with the hard, steely stare of the determined and uninitiated: “My desire to eliminate my physiological need for sleep.”
If you follow Dr. Glaucomflecken on Twitter, you might know the skit I’m referencing. For many physicians and physicians-in-training, what makes the satire successful is its reflection of reality.
Many things have changed in medicine since his time, but the tired trope of the sleepless surgeon hangs on. Undaunted, I spent my second and third year of medical school accumulating accolades, conducting research, and connecting with mentors with the singular goal of joining the surgical ranks.
Midway through my third year, I completed a month-long surgical subinternship designed to give students a taste of what life would look like as an intern. I loved the operating room; it felt like the difference between being on dry land and being underwater. There were fewer distractions – your patient in the spotlight while everything else receded to the shadows.
However, as the month wore on, something stronger took hold. I couldn’t keep my eyes open in the darkened operating rooms and had to decline stools, fearing that I would fall asleep if I sat down.
On early morning prerounds, it’s 4:50 a.m. when I glance at the clock and pull back the curtain, already apologizing. My patient rolls over, flashing a wry smile. “Do you ever go home?” I’ve seen residents respond to this exact question in various ways. I live here. Yes. No. Soon. Not enough. My partner doesn’t think so.
There are days and, yes, years when we are led to believe this is what we live for: to be constantly available to our patients. It feels like a hollow victory when the patient, 2 days out from a total colectomy, begins to worry about your personal life. I ask her how she slept (not enough), any fevers (no), vomiting (no), urinating (I pause – she has a catheter).
My favorite part of these early morning rounds is the pause in my scripted litany of questions to listen to heart and lungs. It never fails to feel sacred: Patients become so quiet and still that I can’t help but think they have faith in me. Without prompting, she slides the back of her hospital gown forward like a curtain, already taking deep breaths so I can hear her lungs.
I look outside. The streetlights are still on, and from the seventh-floor window, I can watch staff making their way through the sliding double-doors, just beyond the yellowed pools of streetlight. I smile. I love medicine. I’m so tired.
For many in medicine, we are treated, and thus behave, as though our ability to manipulate physiology should also apply within the borders of our bodies: commanding less sleep, food, or bathroom breaks.
It places health care workers solidly in the realm of superhuman, living beyond one’s corporeal needs. The pandemic only heightened this misappropriation – adding hero and setting out a pedestal for health care workers to make their ungainly ascent. This kind of unsolicited admiration implicitly implies inhumanness, an otherness.
What would it look like if we started treating ourselves less like physicians and more like patients? I wish I was offering a solution, but really this is just a story.
To students rising through the ranks of medical training, identify what it is you need early and often. I can count on one hand how many physicians I’ve seen take a lunch break – even 10 minutes. Embrace hard work and self-preservation equally. My hope is that if enough of us take this path, it just might become a matter of course.
Dr. Meffert is a resident in the department of emergency medicine, MedStar Georgetown University Hospital, Washington Hospital Center, Washington. Dr. Meffert disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a video posted to TikTok by the comedian Will Flanary, MD, better known to his followers as Dr. Glaucomflecken, he imitates a neurosurgical residency interview. With glasses perched on the bridge of his nose, Dr. Glaucomflecken poses as the attending, asking: “What are your weaknesses?”
The residency applicant answers without hesitation: “My physiological need for sleep.” “What are your strengths?” The resident replies with the hard, steely stare of the determined and uninitiated: “My desire to eliminate my physiological need for sleep.”
If you follow Dr. Glaucomflecken on Twitter, you might know the skit I’m referencing. For many physicians and physicians-in-training, what makes the satire successful is its reflection of reality.
Many things have changed in medicine since his time, but the tired trope of the sleepless surgeon hangs on. Undaunted, I spent my second and third year of medical school accumulating accolades, conducting research, and connecting with mentors with the singular goal of joining the surgical ranks.
Midway through my third year, I completed a month-long surgical subinternship designed to give students a taste of what life would look like as an intern. I loved the operating room; it felt like the difference between being on dry land and being underwater. There were fewer distractions – your patient in the spotlight while everything else receded to the shadows.
However, as the month wore on, something stronger took hold. I couldn’t keep my eyes open in the darkened operating rooms and had to decline stools, fearing that I would fall asleep if I sat down.
On early morning prerounds, it’s 4:50 a.m. when I glance at the clock and pull back the curtain, already apologizing. My patient rolls over, flashing a wry smile. “Do you ever go home?” I’ve seen residents respond to this exact question in various ways. I live here. Yes. No. Soon. Not enough. My partner doesn’t think so.
There are days and, yes, years when we are led to believe this is what we live for: to be constantly available to our patients. It feels like a hollow victory when the patient, 2 days out from a total colectomy, begins to worry about your personal life. I ask her how she slept (not enough), any fevers (no), vomiting (no), urinating (I pause – she has a catheter).
My favorite part of these early morning rounds is the pause in my scripted litany of questions to listen to heart and lungs. It never fails to feel sacred: Patients become so quiet and still that I can’t help but think they have faith in me. Without prompting, she slides the back of her hospital gown forward like a curtain, already taking deep breaths so I can hear her lungs.
I look outside. The streetlights are still on, and from the seventh-floor window, I can watch staff making their way through the sliding double-doors, just beyond the yellowed pools of streetlight. I smile. I love medicine. I’m so tired.
For many in medicine, we are treated, and thus behave, as though our ability to manipulate physiology should also apply within the borders of our bodies: commanding less sleep, food, or bathroom breaks.
It places health care workers solidly in the realm of superhuman, living beyond one’s corporeal needs. The pandemic only heightened this misappropriation – adding hero and setting out a pedestal for health care workers to make their ungainly ascent. This kind of unsolicited admiration implicitly implies inhumanness, an otherness.
What would it look like if we started treating ourselves less like physicians and more like patients? I wish I was offering a solution, but really this is just a story.
To students rising through the ranks of medical training, identify what it is you need early and often. I can count on one hand how many physicians I’ve seen take a lunch break – even 10 minutes. Embrace hard work and self-preservation equally. My hope is that if enough of us take this path, it just might become a matter of course.
Dr. Meffert is a resident in the department of emergency medicine, MedStar Georgetown University Hospital, Washington Hospital Center, Washington. Dr. Meffert disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘I shall harm’
I was quite sure I had multiple sclerosis when I was a medical student. I first noticed symptoms during my neurology rotation. All the signs were there: Fatigue that was getting worse in the North Carolina heat (Uhthoff sign!). A tingle running down my neck (Lhermitte sign!). Blurry vision late at night while studying pathways in Lange Neurology. “Didn’t cousin Amy have MS?” I asked my Mom. I started researching which medical specialties didn’t require dexterity. My left eyelid began twitching and didn’t stop until I rotated to ob.gyn.
Fortunately, it was not multiple sclerosis I had, but rather nosophobia, also known as Medical Student’s Disease. The combination of intense study of symptoms, spotty knowledge of diagnoses, and grade anxiety makes nosophobia common in med students. Despite its name, it doesn’t afflict only doctors. Patients often come to us convinced they have a disease but without reason. So unshakable is their belief that multiple visits are often required to disabuse them of their self-diagnosis. I sometimes have to remind myself to appear concerned even when a “melanoma” is a freckle so small I can barely see it with a dermatoscope. Or a “genital wart” is a hair follicle that looks exactly like the hundreds on the patient’s scrotum. Tougher though, are the treatment-avoiders: patients whose imagined side effects lead them to stop or refuse treatment.
I recently saw a middle-aged man with erythroderma so severe he looked like a ghillie suit of scale. He had a lifelong history of atopic dermatitis and a 2-year history of avoiding treatments. At some point, he tried all the usual remedies: cyclosporine, methotrexate, azathioprine, light therapy, boxes of topicals. The last treatment had been dupilumab, which he tried for a few weeks. “Why did you stop that one?” I asked. The injections were making him go blind, he explained. “Not blurry? Blind?” I asked. Yes, he could not see at all after each injection. Perhaps he might have dry eyes or keratitis? Sure. But blindness? It seemed an unreasonable concern. Further discussion revealed that intolerance to medication side effects was why he had stopped all his other treatments.
Nocebo, from the Latin “I shall harm,” is the dark counterpart to the placebo. Patients experience imagined, or even real, adverse effects because they believe the treatment is causing them harm. This is true even though that treatment might not be having any unwanted physiologic effect. Statins are a good example. Studies have shown that most patient-reported side effects of statins are in fact nocebo effects rather than a result of pharmacologic causes.
Yet, many patients on statins report muscle pain or other concerns as unbearable. As a consequence, some patients who might have benefited from statins might be missing out on the protective gains. as compared with bad outcomes that occurred from not taking action. It’s frustrating when there’s a standard of care treatment, but our patient can’t get past their fear of harm to try it.
Despite my recommendations, my eczema patient insisted on continuing his nontreatment rather than take any risks with treatments for now. There are ways I might help, but I expect it will require additional visits to build trust. Today, the best I can do is to understand and respect him. At least he doesn’t think he has a genital wart – I’m not sure how I’d treat it if he did.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
I was quite sure I had multiple sclerosis when I was a medical student. I first noticed symptoms during my neurology rotation. All the signs were there: Fatigue that was getting worse in the North Carolina heat (Uhthoff sign!). A tingle running down my neck (Lhermitte sign!). Blurry vision late at night while studying pathways in Lange Neurology. “Didn’t cousin Amy have MS?” I asked my Mom. I started researching which medical specialties didn’t require dexterity. My left eyelid began twitching and didn’t stop until I rotated to ob.gyn.
Fortunately, it was not multiple sclerosis I had, but rather nosophobia, also known as Medical Student’s Disease. The combination of intense study of symptoms, spotty knowledge of diagnoses, and grade anxiety makes nosophobia common in med students. Despite its name, it doesn’t afflict only doctors. Patients often come to us convinced they have a disease but without reason. So unshakable is their belief that multiple visits are often required to disabuse them of their self-diagnosis. I sometimes have to remind myself to appear concerned even when a “melanoma” is a freckle so small I can barely see it with a dermatoscope. Or a “genital wart” is a hair follicle that looks exactly like the hundreds on the patient’s scrotum. Tougher though, are the treatment-avoiders: patients whose imagined side effects lead them to stop or refuse treatment.
I recently saw a middle-aged man with erythroderma so severe he looked like a ghillie suit of scale. He had a lifelong history of atopic dermatitis and a 2-year history of avoiding treatments. At some point, he tried all the usual remedies: cyclosporine, methotrexate, azathioprine, light therapy, boxes of topicals. The last treatment had been dupilumab, which he tried for a few weeks. “Why did you stop that one?” I asked. The injections were making him go blind, he explained. “Not blurry? Blind?” I asked. Yes, he could not see at all after each injection. Perhaps he might have dry eyes or keratitis? Sure. But blindness? It seemed an unreasonable concern. Further discussion revealed that intolerance to medication side effects was why he had stopped all his other treatments.
Nocebo, from the Latin “I shall harm,” is the dark counterpart to the placebo. Patients experience imagined, or even real, adverse effects because they believe the treatment is causing them harm. This is true even though that treatment might not be having any unwanted physiologic effect. Statins are a good example. Studies have shown that most patient-reported side effects of statins are in fact nocebo effects rather than a result of pharmacologic causes.
Yet, many patients on statins report muscle pain or other concerns as unbearable. As a consequence, some patients who might have benefited from statins might be missing out on the protective gains. as compared with bad outcomes that occurred from not taking action. It’s frustrating when there’s a standard of care treatment, but our patient can’t get past their fear of harm to try it.
Despite my recommendations, my eczema patient insisted on continuing his nontreatment rather than take any risks with treatments for now. There are ways I might help, but I expect it will require additional visits to build trust. Today, the best I can do is to understand and respect him. At least he doesn’t think he has a genital wart – I’m not sure how I’d treat it if he did.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
I was quite sure I had multiple sclerosis when I was a medical student. I first noticed symptoms during my neurology rotation. All the signs were there: Fatigue that was getting worse in the North Carolina heat (Uhthoff sign!). A tingle running down my neck (Lhermitte sign!). Blurry vision late at night while studying pathways in Lange Neurology. “Didn’t cousin Amy have MS?” I asked my Mom. I started researching which medical specialties didn’t require dexterity. My left eyelid began twitching and didn’t stop until I rotated to ob.gyn.
Fortunately, it was not multiple sclerosis I had, but rather nosophobia, also known as Medical Student’s Disease. The combination of intense study of symptoms, spotty knowledge of diagnoses, and grade anxiety makes nosophobia common in med students. Despite its name, it doesn’t afflict only doctors. Patients often come to us convinced they have a disease but without reason. So unshakable is their belief that multiple visits are often required to disabuse them of their self-diagnosis. I sometimes have to remind myself to appear concerned even when a “melanoma” is a freckle so small I can barely see it with a dermatoscope. Or a “genital wart” is a hair follicle that looks exactly like the hundreds on the patient’s scrotum. Tougher though, are the treatment-avoiders: patients whose imagined side effects lead them to stop or refuse treatment.
I recently saw a middle-aged man with erythroderma so severe he looked like a ghillie suit of scale. He had a lifelong history of atopic dermatitis and a 2-year history of avoiding treatments. At some point, he tried all the usual remedies: cyclosporine, methotrexate, azathioprine, light therapy, boxes of topicals. The last treatment had been dupilumab, which he tried for a few weeks. “Why did you stop that one?” I asked. The injections were making him go blind, he explained. “Not blurry? Blind?” I asked. Yes, he could not see at all after each injection. Perhaps he might have dry eyes or keratitis? Sure. But blindness? It seemed an unreasonable concern. Further discussion revealed that intolerance to medication side effects was why he had stopped all his other treatments.
Nocebo, from the Latin “I shall harm,” is the dark counterpart to the placebo. Patients experience imagined, or even real, adverse effects because they believe the treatment is causing them harm. This is true even though that treatment might not be having any unwanted physiologic effect. Statins are a good example. Studies have shown that most patient-reported side effects of statins are in fact nocebo effects rather than a result of pharmacologic causes.
Yet, many patients on statins report muscle pain or other concerns as unbearable. As a consequence, some patients who might have benefited from statins might be missing out on the protective gains. as compared with bad outcomes that occurred from not taking action. It’s frustrating when there’s a standard of care treatment, but our patient can’t get past their fear of harm to try it.
Despite my recommendations, my eczema patient insisted on continuing his nontreatment rather than take any risks with treatments for now. There are ways I might help, but I expect it will require additional visits to build trust. Today, the best I can do is to understand and respect him. At least he doesn’t think he has a genital wart – I’m not sure how I’d treat it if he did.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
When and for whom is telehealth OCD treatment appropriate?
It is no secret that the COVID-19 pandemic resulted in widespread disruptions in health care services. While providers and resources were limited and many patients were apprehensive to present to health care settings out of concern of disease contraction, telehealth services did offer some relief.
Compared to other specialty care services, mental health care providers were well equipped to handle the expansion of telehealth services, as extensive treatment literature provides strong support for the use of psychotherapeutic interventions over telehealth mediums.1 This holds true in the context of obsessive-compulsive disorder (OCD), where an impressive literature supports the use of telehealth delivery for the gold-standard psychotherapeutic, exposure and response prevention (ERP).2,3,4
Through ERP, patients work with a clinician to systematically expose themselves to anxiety-providing triggers while actively resisting compulsive behaviors to learn the distress does go away with time and/or the distress is within their ability to cope. This intervention is conceptually similar to repeatedly watching a scary film, by which continued exposure results in less pronounced emotional reaction with subsequent viewings.
Fortunately for patients and providers, the expansion of telehealth ERP across different treatment settings has had many unintended benefits, including increased access to care, lower no-show rates due to the ease of attending appointments, and the ability to offer higher levels of care, including intensive outpatient programs, over telehealth mediums. Anecdotally, our clinic has been able to increase patient reach by providing telehealth ERP to those who historically would not have been able to access care due to geography. Even for those living within driving distance to our clinic, the ease of joining a video visit for a 45-minute appointment far outweighs driving into the clinic, in many circumstances. With these benefits, the delivery of ERP over telehealth appears likely to stay, although OCD providers delivering ERP will need to consider when and for whom this medium may not be appropriate.
To this end, we recently conducted a study examining ERP providers perceptions of telehealth and in-person ERP, patient characteristics best suited for telehealth services, and provider ability to identify and address factors that adversely impact the course of treatment (e.g., substance use, limited symptom insight, distractions during ERP, etc.).5 Providers reported lower feasibility ratings for telehealth compared to in-person ERP for younger patients (aged under 13 years), and patients with more severe OCD presentations. Providers also reported more difficulty identifying and addressing ERP interfering factors over telehealth relative to in-person. The findings from our research do not necessarily speak to the effectiveness of telehealth ERP, which has repeatedly been documented in treatment literature, but rather our findings highlight that ERP providers endorse reservations about the feasibility of ERP for certain OCD patient profiles, and that telehealth ERP may not be appropriate for all patients with OCD.
Mental health care providers, including those delivering ERP, should consider when telehealth is and is not appropriate. Importantly, telehealth offers a limited field of view compared to in-person, and providers can only observe what is captured by the camera. In the context of telehealth ERP, patients may engage in subtle avoidant behaviors that are more difficult to observe, which may prevent them from experiencing necessary anxiety during exposure practice. Many providers may have firsthand experience with this, or patients who appear distracted over telehealth mediums because of environmental factors that can be controlled for during in-person services.
As telehealth treatment options appear increasingly likely to stay, ERP providers and intervention researchers should continue identifying patient characteristics that are more and less appropriate for telehealth settings in order to maximize treatment outcomes. Providers should share concerns with patients when delivering telehealth ERP and work to address interfering factors impacting the course of treatment. In circumstances where this is not possible, such as when the patients age or symptom severity prevents effective telehealth ERP, or when treatment progress stalls, providers should speak with patients to determine if it would be beneficial to transition to in-person services.
Both in-person and telehealth ERP are fundamentally the same, however it does appear that subtle differences across these modalities may have differential impacts on treatment outcomes for certain OCD patient presentations. , however appropriate caution should be exhibited, and providers should use clinical judgment when offering telehealth services.
Dr. Wiese is a clinical psychologist in the department of psychiatry and behavioral sciences at Baylor College of Medicine, Houston, Texas. He is primarily focused on conducting research on OCD and related disorders and providing empirically supported treatments to individuals diagnosed with these conditions.
References
1. Fernandez E et al. Live psychotherapy by video versus in‐person: A meta‐analysis of efficacy and its relationship to types and targets of treatment. Clin Psychol Psychother. 2021 Nov;28(6):1535-49. doi: 10.1002/cpp.2594.
2. Storch EA et al. Preliminary investigation of web-camera delivered cognitive-behavioral therapy for youth with obsessive-compulsive disorder. Psychiatry Res. 2011 Oct 30;189(3):407-12. doi: 10.1016/j.psychres.2011.05.047.
3. Fletcher TL et al. A pilot open trial of video telehealth-delivered exposure and response prevention for obsessive-compulsive disorder in rural Veterans. Mil Psychol. 2021 Oct 28;34(1):83-90. doi: 10.1080/08995605.2021.1970983.
4. Wootton BM. Remote cognitive–behavior therapy for obsessive–compulsive symptoms: a meta-analysis. Clin Psychol Rev. 2016 Feb;43:103-13. doi: 10.1016/j.cpr.2015.10.001.
5. Wiese AD et al. Provider perceptions of telehealth and in-person exposure and response prevention for obsessive–compulsive disorder. Psychiatry Res. 2022 Jul;313:114610. doi: 10.1016/j.psychres.2022.114610.
It is no secret that the COVID-19 pandemic resulted in widespread disruptions in health care services. While providers and resources were limited and many patients were apprehensive to present to health care settings out of concern of disease contraction, telehealth services did offer some relief.
Compared to other specialty care services, mental health care providers were well equipped to handle the expansion of telehealth services, as extensive treatment literature provides strong support for the use of psychotherapeutic interventions over telehealth mediums.1 This holds true in the context of obsessive-compulsive disorder (OCD), where an impressive literature supports the use of telehealth delivery for the gold-standard psychotherapeutic, exposure and response prevention (ERP).2,3,4
Through ERP, patients work with a clinician to systematically expose themselves to anxiety-providing triggers while actively resisting compulsive behaviors to learn the distress does go away with time and/or the distress is within their ability to cope. This intervention is conceptually similar to repeatedly watching a scary film, by which continued exposure results in less pronounced emotional reaction with subsequent viewings.
Fortunately for patients and providers, the expansion of telehealth ERP across different treatment settings has had many unintended benefits, including increased access to care, lower no-show rates due to the ease of attending appointments, and the ability to offer higher levels of care, including intensive outpatient programs, over telehealth mediums. Anecdotally, our clinic has been able to increase patient reach by providing telehealth ERP to those who historically would not have been able to access care due to geography. Even for those living within driving distance to our clinic, the ease of joining a video visit for a 45-minute appointment far outweighs driving into the clinic, in many circumstances. With these benefits, the delivery of ERP over telehealth appears likely to stay, although OCD providers delivering ERP will need to consider when and for whom this medium may not be appropriate.
To this end, we recently conducted a study examining ERP providers perceptions of telehealth and in-person ERP, patient characteristics best suited for telehealth services, and provider ability to identify and address factors that adversely impact the course of treatment (e.g., substance use, limited symptom insight, distractions during ERP, etc.).5 Providers reported lower feasibility ratings for telehealth compared to in-person ERP for younger patients (aged under 13 years), and patients with more severe OCD presentations. Providers also reported more difficulty identifying and addressing ERP interfering factors over telehealth relative to in-person. The findings from our research do not necessarily speak to the effectiveness of telehealth ERP, which has repeatedly been documented in treatment literature, but rather our findings highlight that ERP providers endorse reservations about the feasibility of ERP for certain OCD patient profiles, and that telehealth ERP may not be appropriate for all patients with OCD.
Mental health care providers, including those delivering ERP, should consider when telehealth is and is not appropriate. Importantly, telehealth offers a limited field of view compared to in-person, and providers can only observe what is captured by the camera. In the context of telehealth ERP, patients may engage in subtle avoidant behaviors that are more difficult to observe, which may prevent them from experiencing necessary anxiety during exposure practice. Many providers may have firsthand experience with this, or patients who appear distracted over telehealth mediums because of environmental factors that can be controlled for during in-person services.
As telehealth treatment options appear increasingly likely to stay, ERP providers and intervention researchers should continue identifying patient characteristics that are more and less appropriate for telehealth settings in order to maximize treatment outcomes. Providers should share concerns with patients when delivering telehealth ERP and work to address interfering factors impacting the course of treatment. In circumstances where this is not possible, such as when the patients age or symptom severity prevents effective telehealth ERP, or when treatment progress stalls, providers should speak with patients to determine if it would be beneficial to transition to in-person services.
Both in-person and telehealth ERP are fundamentally the same, however it does appear that subtle differences across these modalities may have differential impacts on treatment outcomes for certain OCD patient presentations. , however appropriate caution should be exhibited, and providers should use clinical judgment when offering telehealth services.
Dr. Wiese is a clinical psychologist in the department of psychiatry and behavioral sciences at Baylor College of Medicine, Houston, Texas. He is primarily focused on conducting research on OCD and related disorders and providing empirically supported treatments to individuals diagnosed with these conditions.
References
1. Fernandez E et al. Live psychotherapy by video versus in‐person: A meta‐analysis of efficacy and its relationship to types and targets of treatment. Clin Psychol Psychother. 2021 Nov;28(6):1535-49. doi: 10.1002/cpp.2594.
2. Storch EA et al. Preliminary investigation of web-camera delivered cognitive-behavioral therapy for youth with obsessive-compulsive disorder. Psychiatry Res. 2011 Oct 30;189(3):407-12. doi: 10.1016/j.psychres.2011.05.047.
3. Fletcher TL et al. A pilot open trial of video telehealth-delivered exposure and response prevention for obsessive-compulsive disorder in rural Veterans. Mil Psychol. 2021 Oct 28;34(1):83-90. doi: 10.1080/08995605.2021.1970983.
4. Wootton BM. Remote cognitive–behavior therapy for obsessive–compulsive symptoms: a meta-analysis. Clin Psychol Rev. 2016 Feb;43:103-13. doi: 10.1016/j.cpr.2015.10.001.
5. Wiese AD et al. Provider perceptions of telehealth and in-person exposure and response prevention for obsessive–compulsive disorder. Psychiatry Res. 2022 Jul;313:114610. doi: 10.1016/j.psychres.2022.114610.
It is no secret that the COVID-19 pandemic resulted in widespread disruptions in health care services. While providers and resources were limited and many patients were apprehensive to present to health care settings out of concern of disease contraction, telehealth services did offer some relief.
Compared to other specialty care services, mental health care providers were well equipped to handle the expansion of telehealth services, as extensive treatment literature provides strong support for the use of psychotherapeutic interventions over telehealth mediums.1 This holds true in the context of obsessive-compulsive disorder (OCD), where an impressive literature supports the use of telehealth delivery for the gold-standard psychotherapeutic, exposure and response prevention (ERP).2,3,4
Through ERP, patients work with a clinician to systematically expose themselves to anxiety-providing triggers while actively resisting compulsive behaviors to learn the distress does go away with time and/or the distress is within their ability to cope. This intervention is conceptually similar to repeatedly watching a scary film, by which continued exposure results in less pronounced emotional reaction with subsequent viewings.
Fortunately for patients and providers, the expansion of telehealth ERP across different treatment settings has had many unintended benefits, including increased access to care, lower no-show rates due to the ease of attending appointments, and the ability to offer higher levels of care, including intensive outpatient programs, over telehealth mediums. Anecdotally, our clinic has been able to increase patient reach by providing telehealth ERP to those who historically would not have been able to access care due to geography. Even for those living within driving distance to our clinic, the ease of joining a video visit for a 45-minute appointment far outweighs driving into the clinic, in many circumstances. With these benefits, the delivery of ERP over telehealth appears likely to stay, although OCD providers delivering ERP will need to consider when and for whom this medium may not be appropriate.
To this end, we recently conducted a study examining ERP providers perceptions of telehealth and in-person ERP, patient characteristics best suited for telehealth services, and provider ability to identify and address factors that adversely impact the course of treatment (e.g., substance use, limited symptom insight, distractions during ERP, etc.).5 Providers reported lower feasibility ratings for telehealth compared to in-person ERP for younger patients (aged under 13 years), and patients with more severe OCD presentations. Providers also reported more difficulty identifying and addressing ERP interfering factors over telehealth relative to in-person. The findings from our research do not necessarily speak to the effectiveness of telehealth ERP, which has repeatedly been documented in treatment literature, but rather our findings highlight that ERP providers endorse reservations about the feasibility of ERP for certain OCD patient profiles, and that telehealth ERP may not be appropriate for all patients with OCD.
Mental health care providers, including those delivering ERP, should consider when telehealth is and is not appropriate. Importantly, telehealth offers a limited field of view compared to in-person, and providers can only observe what is captured by the camera. In the context of telehealth ERP, patients may engage in subtle avoidant behaviors that are more difficult to observe, which may prevent them from experiencing necessary anxiety during exposure practice. Many providers may have firsthand experience with this, or patients who appear distracted over telehealth mediums because of environmental factors that can be controlled for during in-person services.
As telehealth treatment options appear increasingly likely to stay, ERP providers and intervention researchers should continue identifying patient characteristics that are more and less appropriate for telehealth settings in order to maximize treatment outcomes. Providers should share concerns with patients when delivering telehealth ERP and work to address interfering factors impacting the course of treatment. In circumstances where this is not possible, such as when the patients age or symptom severity prevents effective telehealth ERP, or when treatment progress stalls, providers should speak with patients to determine if it would be beneficial to transition to in-person services.
Both in-person and telehealth ERP are fundamentally the same, however it does appear that subtle differences across these modalities may have differential impacts on treatment outcomes for certain OCD patient presentations. , however appropriate caution should be exhibited, and providers should use clinical judgment when offering telehealth services.
Dr. Wiese is a clinical psychologist in the department of psychiatry and behavioral sciences at Baylor College of Medicine, Houston, Texas. He is primarily focused on conducting research on OCD and related disorders and providing empirically supported treatments to individuals diagnosed with these conditions.
References
1. Fernandez E et al. Live psychotherapy by video versus in‐person: A meta‐analysis of efficacy and its relationship to types and targets of treatment. Clin Psychol Psychother. 2021 Nov;28(6):1535-49. doi: 10.1002/cpp.2594.
2. Storch EA et al. Preliminary investigation of web-camera delivered cognitive-behavioral therapy for youth with obsessive-compulsive disorder. Psychiatry Res. 2011 Oct 30;189(3):407-12. doi: 10.1016/j.psychres.2011.05.047.
3. Fletcher TL et al. A pilot open trial of video telehealth-delivered exposure and response prevention for obsessive-compulsive disorder in rural Veterans. Mil Psychol. 2021 Oct 28;34(1):83-90. doi: 10.1080/08995605.2021.1970983.
4. Wootton BM. Remote cognitive–behavior therapy for obsessive–compulsive symptoms: a meta-analysis. Clin Psychol Rev. 2016 Feb;43:103-13. doi: 10.1016/j.cpr.2015.10.001.
5. Wiese AD et al. Provider perceptions of telehealth and in-person exposure and response prevention for obsessive–compulsive disorder. Psychiatry Res. 2022 Jul;313:114610. doi: 10.1016/j.psychres.2022.114610.
Pediatric obesity treatment options: Beyond lifestyle modification
Pediatric obesity is a serious problem, not only in the United States but worldwide. Unfortunately, the ongoing COVID-19 pandemic has worsened the epidemic of childhood obesity. Solutions for treating the millions of children and adolescents with obesity are desperately needed because prevention efforts over the past several decades have not been sufficient in slowing the steady rise in obesity prevalence.
Lifestyle modification, including dietary changes, increases in activity, and behavioral modification, are the cornerstone of any obesity treatment, but they alone are not powerful enough to treat obesity by itself in the vast majority of children and adolescents. This is because obesity is not a lifestyle choice; rather, it is a disease, and a disease that has a tremendous amount of biology driving individuals toward weight gain and the propensity toward weight regain if weight is lost.
Fortunately, the tools to treat the underlying biology driving obesity are becoming safer, more effective, and more widely used every year. The two most effective biology-based treatments for pediatric obesity are antiobesity medications and bariatric surgery. These two treatments, when accompanied by lifestyle modification, have the potential to reduce not only body weight but also treat many other risk factors, such as prediabetes, diabetes, high blood pressure, poor cholesterol, liver disease, and sleep apnea, as well as others.
Rise in antiobesity medications
Antiobesity medications are developing at a rapid pace. Seven medications have been approved by the Food and Drug Administration for adults, and three medications (phentermine, orlistat, and liraglutide) are now approved for children and adolescents.
The number of antiobesity medications for use in children and adolescents is expected to expand to five, with semaglutide and phentermine-topiramate (Qsymia) both completing trials in adolescents in 2022. Each of these medications works by treating the biology that drives weight gain, whether it is decreasing impulsivity, reducing hunger and appetite hormone pathways, or improving energy regulation pathways. Weight loss at 1 year for currently FDA-approved medications in adolescents ranges from 3% to 6% on average, depending on the medications. The newer medications already FDA approved in adults that will soon, hopefully, be available in pediatrics result in 10%-16% weight loss on average.
A common parent and patient question regarding antiobesity medications is: “If I start an antiobesity medication, how long will I need to be on it?” The simple answer is: “Probably for the rest of your life.”
This can be a shock to hear, but obesity treatment is very similar to that of hypertension or diabetes. Using high blood pressure as an example: If a patient has high blood pressure (for example, 160/90 mm Hg), they will be prescribed a medication to treat it. Once blood pressure comes down to near-normal levels (for example, 120/80 mm Hg), a dose will be maintained, not removed, because that is the biological mediator keeping the blood pressure low. Removal of the medication would result in blood pressure going back to homeostasis (160/90 mm Hg in our example) in a short period of time).
The same can be said for obesity. For example, if a 16-year-old girl is prescribed liraglutide, a glucagonlike peptide–1 receptor agonist, and loses 10% of her body weight at 1 year, that is great success. Why would we remove the medication that is treating the underlying biology causing successful weight loss?
In short, we would not want to do that. Even if our example patient only maintained that 10% initial weight loss, that would be very successful, just like someone maintaining their low blood pressure. As medications begin to develop at a rapid pace and become more available to pediatric patients, the messaging and conversation around anti-obesity medications must continue to focus on obesity being a biological disease and not a behavior for treatment to be effective and not stigmatized.
Bariatric surgery most effective treatment for pediatric obesity
Currently, the most effective treatment for pediatric obesity is bariatric surgery. The two most commonly used surgical procedures today are the sleeve gastrectomy and gastric bypass. Sleeve gastrectomy works by removing 75%-85% of the stomach and creating a new stomach, called a “sleeve.” Gastric bypass works by separating the stomach into two parts and connecting one part of the new stomach into the intestine.
Both surgeries are very effective at treating obesity in adolescents, with an average weight loss of 30%-35%. Surgery is not just a restrictive means of controlling body weight; it also changes key hormones for appetite and satiety that signal the brain. In fact, many of the same biological signals that are changed by surgery are the same signals being targeted by antiobesity medications. Long-term outcome of bariatric surgery in adolescents, provided by Teen-LABS, show it to be safe and maybe even more effective than in adults for treating diabetes and hypertension, with similar weight loss.
Does treatment outweigh the potential risks?
Although obesity surgery and antiobesity medications are more successful at treating obesity in children and adolescents than lifestyle medications, they do have some risks. Surgery, depending on the type of surgery, can cause nutritional deficiencies, reduce body mineral density, and is a life-changing medical procedure. Antiobesity medications, depending on the type, can cause nausea and vomiting and increase heart rate – and because they are relatively new, we do not fully understand the long-term impact of continued use past 1 year.
However, an important question to ask is: “Do the risks of obesity surgery and antiobesity medications outweigh the risk of having lifelong obesity?” The answer to me and many of my colleagues is: “Yes!” Although there are risks associated with the two best treatments for pediatric obesity, those risks under proper supervision of a medical professional far outweigh the risks of not properly treating obesity and allowing it to persist and get worse over many years to come. Obesity is a disease deeply rooted in biology, and we must use biology-based treatments to tackle this problem in children and adolescents, who deserve the best care and treatments possible.
Dr. Ryder is assistant professor of pediatrics and associate director of research, Center for Pediatric Obesity Medicine, at the University of Minnesota, Minneapolis. She reported receiving donations for clinical trials from Boehringer Ingelheim. A version of this article first appeared on Medscape.com.
Pediatric obesity is a serious problem, not only in the United States but worldwide. Unfortunately, the ongoing COVID-19 pandemic has worsened the epidemic of childhood obesity. Solutions for treating the millions of children and adolescents with obesity are desperately needed because prevention efforts over the past several decades have not been sufficient in slowing the steady rise in obesity prevalence.
Lifestyle modification, including dietary changes, increases in activity, and behavioral modification, are the cornerstone of any obesity treatment, but they alone are not powerful enough to treat obesity by itself in the vast majority of children and adolescents. This is because obesity is not a lifestyle choice; rather, it is a disease, and a disease that has a tremendous amount of biology driving individuals toward weight gain and the propensity toward weight regain if weight is lost.
Fortunately, the tools to treat the underlying biology driving obesity are becoming safer, more effective, and more widely used every year. The two most effective biology-based treatments for pediatric obesity are antiobesity medications and bariatric surgery. These two treatments, when accompanied by lifestyle modification, have the potential to reduce not only body weight but also treat many other risk factors, such as prediabetes, diabetes, high blood pressure, poor cholesterol, liver disease, and sleep apnea, as well as others.
Rise in antiobesity medications
Antiobesity medications are developing at a rapid pace. Seven medications have been approved by the Food and Drug Administration for adults, and three medications (phentermine, orlistat, and liraglutide) are now approved for children and adolescents.
The number of antiobesity medications for use in children and adolescents is expected to expand to five, with semaglutide and phentermine-topiramate (Qsymia) both completing trials in adolescents in 2022. Each of these medications works by treating the biology that drives weight gain, whether it is decreasing impulsivity, reducing hunger and appetite hormone pathways, or improving energy regulation pathways. Weight loss at 1 year for currently FDA-approved medications in adolescents ranges from 3% to 6% on average, depending on the medications. The newer medications already FDA approved in adults that will soon, hopefully, be available in pediatrics result in 10%-16% weight loss on average.
A common parent and patient question regarding antiobesity medications is: “If I start an antiobesity medication, how long will I need to be on it?” The simple answer is: “Probably for the rest of your life.”
This can be a shock to hear, but obesity treatment is very similar to that of hypertension or diabetes. Using high blood pressure as an example: If a patient has high blood pressure (for example, 160/90 mm Hg), they will be prescribed a medication to treat it. Once blood pressure comes down to near-normal levels (for example, 120/80 mm Hg), a dose will be maintained, not removed, because that is the biological mediator keeping the blood pressure low. Removal of the medication would result in blood pressure going back to homeostasis (160/90 mm Hg in our example) in a short period of time).
The same can be said for obesity. For example, if a 16-year-old girl is prescribed liraglutide, a glucagonlike peptide–1 receptor agonist, and loses 10% of her body weight at 1 year, that is great success. Why would we remove the medication that is treating the underlying biology causing successful weight loss?
In short, we would not want to do that. Even if our example patient only maintained that 10% initial weight loss, that would be very successful, just like someone maintaining their low blood pressure. As medications begin to develop at a rapid pace and become more available to pediatric patients, the messaging and conversation around anti-obesity medications must continue to focus on obesity being a biological disease and not a behavior for treatment to be effective and not stigmatized.
Bariatric surgery most effective treatment for pediatric obesity
Currently, the most effective treatment for pediatric obesity is bariatric surgery. The two most commonly used surgical procedures today are the sleeve gastrectomy and gastric bypass. Sleeve gastrectomy works by removing 75%-85% of the stomach and creating a new stomach, called a “sleeve.” Gastric bypass works by separating the stomach into two parts and connecting one part of the new stomach into the intestine.
Both surgeries are very effective at treating obesity in adolescents, with an average weight loss of 30%-35%. Surgery is not just a restrictive means of controlling body weight; it also changes key hormones for appetite and satiety that signal the brain. In fact, many of the same biological signals that are changed by surgery are the same signals being targeted by antiobesity medications. Long-term outcome of bariatric surgery in adolescents, provided by Teen-LABS, show it to be safe and maybe even more effective than in adults for treating diabetes and hypertension, with similar weight loss.
Does treatment outweigh the potential risks?
Although obesity surgery and antiobesity medications are more successful at treating obesity in children and adolescents than lifestyle medications, they do have some risks. Surgery, depending on the type of surgery, can cause nutritional deficiencies, reduce body mineral density, and is a life-changing medical procedure. Antiobesity medications, depending on the type, can cause nausea and vomiting and increase heart rate – and because they are relatively new, we do not fully understand the long-term impact of continued use past 1 year.
However, an important question to ask is: “Do the risks of obesity surgery and antiobesity medications outweigh the risk of having lifelong obesity?” The answer to me and many of my colleagues is: “Yes!” Although there are risks associated with the two best treatments for pediatric obesity, those risks under proper supervision of a medical professional far outweigh the risks of not properly treating obesity and allowing it to persist and get worse over many years to come. Obesity is a disease deeply rooted in biology, and we must use biology-based treatments to tackle this problem in children and adolescents, who deserve the best care and treatments possible.
Dr. Ryder is assistant professor of pediatrics and associate director of research, Center for Pediatric Obesity Medicine, at the University of Minnesota, Minneapolis. She reported receiving donations for clinical trials from Boehringer Ingelheim. A version of this article first appeared on Medscape.com.
Pediatric obesity is a serious problem, not only in the United States but worldwide. Unfortunately, the ongoing COVID-19 pandemic has worsened the epidemic of childhood obesity. Solutions for treating the millions of children and adolescents with obesity are desperately needed because prevention efforts over the past several decades have not been sufficient in slowing the steady rise in obesity prevalence.
Lifestyle modification, including dietary changes, increases in activity, and behavioral modification, are the cornerstone of any obesity treatment, but they alone are not powerful enough to treat obesity by itself in the vast majority of children and adolescents. This is because obesity is not a lifestyle choice; rather, it is a disease, and a disease that has a tremendous amount of biology driving individuals toward weight gain and the propensity toward weight regain if weight is lost.
Fortunately, the tools to treat the underlying biology driving obesity are becoming safer, more effective, and more widely used every year. The two most effective biology-based treatments for pediatric obesity are antiobesity medications and bariatric surgery. These two treatments, when accompanied by lifestyle modification, have the potential to reduce not only body weight but also treat many other risk factors, such as prediabetes, diabetes, high blood pressure, poor cholesterol, liver disease, and sleep apnea, as well as others.
Rise in antiobesity medications
Antiobesity medications are developing at a rapid pace. Seven medications have been approved by the Food and Drug Administration for adults, and three medications (phentermine, orlistat, and liraglutide) are now approved for children and adolescents.
The number of antiobesity medications for use in children and adolescents is expected to expand to five, with semaglutide and phentermine-topiramate (Qsymia) both completing trials in adolescents in 2022. Each of these medications works by treating the biology that drives weight gain, whether it is decreasing impulsivity, reducing hunger and appetite hormone pathways, or improving energy regulation pathways. Weight loss at 1 year for currently FDA-approved medications in adolescents ranges from 3% to 6% on average, depending on the medications. The newer medications already FDA approved in adults that will soon, hopefully, be available in pediatrics result in 10%-16% weight loss on average.
A common parent and patient question regarding antiobesity medications is: “If I start an antiobesity medication, how long will I need to be on it?” The simple answer is: “Probably for the rest of your life.”
This can be a shock to hear, but obesity treatment is very similar to that of hypertension or diabetes. Using high blood pressure as an example: If a patient has high blood pressure (for example, 160/90 mm Hg), they will be prescribed a medication to treat it. Once blood pressure comes down to near-normal levels (for example, 120/80 mm Hg), a dose will be maintained, not removed, because that is the biological mediator keeping the blood pressure low. Removal of the medication would result in blood pressure going back to homeostasis (160/90 mm Hg in our example) in a short period of time).
The same can be said for obesity. For example, if a 16-year-old girl is prescribed liraglutide, a glucagonlike peptide–1 receptor agonist, and loses 10% of her body weight at 1 year, that is great success. Why would we remove the medication that is treating the underlying biology causing successful weight loss?
In short, we would not want to do that. Even if our example patient only maintained that 10% initial weight loss, that would be very successful, just like someone maintaining their low blood pressure. As medications begin to develop at a rapid pace and become more available to pediatric patients, the messaging and conversation around anti-obesity medications must continue to focus on obesity being a biological disease and not a behavior for treatment to be effective and not stigmatized.
Bariatric surgery most effective treatment for pediatric obesity
Currently, the most effective treatment for pediatric obesity is bariatric surgery. The two most commonly used surgical procedures today are the sleeve gastrectomy and gastric bypass. Sleeve gastrectomy works by removing 75%-85% of the stomach and creating a new stomach, called a “sleeve.” Gastric bypass works by separating the stomach into two parts and connecting one part of the new stomach into the intestine.
Both surgeries are very effective at treating obesity in adolescents, with an average weight loss of 30%-35%. Surgery is not just a restrictive means of controlling body weight; it also changes key hormones for appetite and satiety that signal the brain. In fact, many of the same biological signals that are changed by surgery are the same signals being targeted by antiobesity medications. Long-term outcome of bariatric surgery in adolescents, provided by Teen-LABS, show it to be safe and maybe even more effective than in adults for treating diabetes and hypertension, with similar weight loss.
Does treatment outweigh the potential risks?
Although obesity surgery and antiobesity medications are more successful at treating obesity in children and adolescents than lifestyle medications, they do have some risks. Surgery, depending on the type of surgery, can cause nutritional deficiencies, reduce body mineral density, and is a life-changing medical procedure. Antiobesity medications, depending on the type, can cause nausea and vomiting and increase heart rate – and because they are relatively new, we do not fully understand the long-term impact of continued use past 1 year.
However, an important question to ask is: “Do the risks of obesity surgery and antiobesity medications outweigh the risk of having lifelong obesity?” The answer to me and many of my colleagues is: “Yes!” Although there are risks associated with the two best treatments for pediatric obesity, those risks under proper supervision of a medical professional far outweigh the risks of not properly treating obesity and allowing it to persist and get worse over many years to come. Obesity is a disease deeply rooted in biology, and we must use biology-based treatments to tackle this problem in children and adolescents, who deserve the best care and treatments possible.
Dr. Ryder is assistant professor of pediatrics and associate director of research, Center for Pediatric Obesity Medicine, at the University of Minnesota, Minneapolis. She reported receiving donations for clinical trials from Boehringer Ingelheim. A version of this article first appeared on Medscape.com.
Meet Argireline, the neurotoxinlike cosmeceutical
Acetyl hexapeptide-8 (or -3), better known by its brand name, Argireline (Lubrizol; Wickliffe, Ohio), is a synthetic peptide gaining popularity in cosmeceutical products for its antiaging benefits. Argireline was developed by the company Lipotec in 2001. Media, beauty bloggers, and product claims have likened this product to a “Botox [or other neurotoxin] alternative,” or “Botox mimicker.”
Mechanism of action
Understanding how Argireline works requires a brief refresher on the mechanism of action of botulinum neurotoxin (BoNT). BoNT relaxes facial muscles and smooths expression lines by inhibiting acetylcholine release at the neuromuscular junction.1 More specifically, the various serotypes of BoNT are single-chain polypeptides that target members of the SNARE complex: SNAP-25, syntaxin, and Vamp. The proteins within the SNARE complex are involved in the docking and fusion of presynaptic vesicles to the presynaptic membrane, necessary steps for acetylcholine release into the neuromuscular junction and muscle contraction. By blocking the action of the SNARE complex proteins, BoNT inhibits release of acetylcholine in the neuromuscular junction and prevents muscle contraction.
Argireline is a synthetic peptide with the sequence Ac-EEMQRR-NH2.2 It is patterned after the N-terminal domain of SNAP-25, one of the members of the SNARE complex targeted by BoNT, and functions to interfere with the assembly of the SNARE complex. In this manner, Argireline would theoretically inhibit fusion of presynaptic vesicles and release of acetylcholine into the neuromuscular junction, thus impeding muscle movement. For this reason, it has been likened to topical Botox. Unlike Botox and other neurotoxins, Argireline was developed for topical application rather than injection.
Preclinical studies
In vitro work done 20 years ago demonstrated that Argireline can prevent assembly of the SNARE complex and inhibit neurotransmitter release with a potency similar to that of BoNT A (Botox).2
In 2013, Wang et al. evaluated the histologic effects of Argireline in aged mouse skin induced by D-galactose. For 6 weeks, Argireline was applied twice daily, and histological changes were assessed using hematoxylin and eosin (H&E) and picrosirius–polarization (PSP) stains. The researchers found elevated levels of type I collagen (P < .01) and reduced type III collagen (P < .05) with the Argireline treatment. These results demonstrated that Argireline could histologically enhance collagen in a manner consistent with skin rejuvenation.3
Clinical studies
In 2002, Blanes et al. assessed the antiwrinkle activity of Argireline by measuring skin topography from silicone implants in the lateral periorbital region of an oil/water (O/W) emulsion containing 10% of the acetyl-hexapeptide in 10 healthy women volunteers. The hexapeptide emulsion was applied twice daily in one lateral periorbital area, and the emulsion vehicle alone was applied twice daily on the contralateral side. Over 30 days of treatment, wrinkle depth was found to have decreased by 30%. The investigators also found that Argireline significantly hindered neurotransmitter release in vitro as robustly as BoNT A, though with notably lower efficacy. No toxicity or irritation was associated with this treatment.2 However, it should be noted that this small study conducted 2 decades ago evaluated only silicone implants with confocal microscopy to evaluate wrinkle depth. There was no subjective clinical assessment of dynamic facial wrinkles. As such, their study is an insufficient basis for drawing conclusions that Argireline is a BoNT mimic. Botox and other types of BoNT affect dynamic facial wrinkles mostly (i.e., wrinkles created by moving muscles of facial expression). This study primarily considers static wrinkles on periorbital skin. While static wrinkles may result from longstanding dynamic wrinkles, BoNT mainly targets dynamic wrinkles, again not comparing apples to apples.
At the same time that Wang et al. conducted their experiment on the skin of aged mice as noted above, they performed a multicenter clinical trial in 60 human subjects who received a randomized treatment of Argireline or placebo in a ratio of 3:1 to assess its safety and efficacy. For 4 weeks, the test product or placebo was applied to periorbital wrinkles twice daily. The researchers found the total antiwrinkle efficacy in the Argireline group to be 48.9% based on the subjective evaluation, compared with 0% in the placebo group. The objective evaluation indicated that all parameters of roughness were diminished in the Argireline group (P < .01), with no reduction observed in the placebo group (P < .05).4 There was a little more to appreciate from this study compared with the one reported by Blanes et al., insofar as subjective evaluations and objective evaluations with silica replicas were done. However, this study was not blinded, so the 48.9% wrinkle reduction in the Argireline group vs. 0% in the control group seems suspicious. Additionally, there was a greater focus on static rather than dynamic wrinkles.
In 2017, Raikou et al. conducted a prospective, randomized controlled study to assess the effects of acetyl hexapeptide-3 (Argireline) and tripeptide-10 citrulline in 24 healthy female volunteers (aged 30-60 years) and determine if there was any synergistic action between the peptides. Subjects were randomized to receive a combination of the peptides, tripeptide-10 citrulline only, acetyl hexapeptide-3 only, or neither peptide for 60 days. The researchers found a significant reduction in transepidermal water loss (TEWL) in the Argireline group, compared with the placebo group.5 The result of this study makes me question if the decrease in depth of the wrinkles measured in the former studies is really just a measure of increased skin hydration from the Argireline, rather than a neurotoxic effect of Argireline.
Formulation and penetration: Can Argireline get through your skin?
One of the fundamental questions regarding Argireline is whether it can penetrate through the stratum corneum and find its target – the facial muscles – where it is intended to function. Argireline is a charged, hydrophilic, and large–molecular weight peptide, and each of these factors impairs penetration through the stratum corneum. Therefore, studies assessing penetration are particularly important.
In 2015, Kraeling et al. conducted an in vitro evaluation of the skin penetration of acetyl hexapeptide-8 in hairless guinea pig and human cadaver skin. An oil-in-water (O/W) emulsion containing 10% acetyl hexapeptide-8 was applied (2 mg/cm2) and penetration was quantified in skin layers via hydrophilic interaction liquid chromatography with tandem mass spectrometry. Most of the acetyl hexapeptide-8 was found to have been washed from human cadaver, as well as guinea pig, skin. Less than 1% of the peptide penetrated the guinea pig or human skin. Of this small amount that penetrated the skin, most stayed in the stratum corneum of guinea pigs (0.54%) and human cadavers (0.22%). The levels of acetyl hexapeptide-8 declined further with each layer of tape stripping removal. Epidermal levels of the peptide in tested skin were similar at 0.01%, and none of the peptide was found in the dermis.6 These results indicate negligible penetration by this highly touted peptide ingredient.
Some studies have shown that altering the formulation of acetyl hexapeptide-8 can enhance penetration. Hoppel et al. demonstrated that formulations of the peptide, especially in a water-oil-water (W/O/W emulsion [as compared with O/W and W/O emulsions] can increase penetration into the stratum corneum in porcine skin.7 Notably, this is still very superficial relative to the dermis and muscles. Irrespective of formulation, studies have shown that Argireline barely penetrates the stratum corneum, let alone the dermis. Therefore, I would give pause to attributing any clinical impact or benefit of Argireline to its neurotoxinlike effects measured in vitro.
Conclusion
Despite the growing popularity of this ingredient in cosmeceuticals and the praise it gets in media for acting as a topical neurotoxin, there are no rigorous clinical trials or data demonstrating its efficacy in suppressing dynamic facial wrinkles like BoNT does. Most importantly, without penetration into the stratum corneum and deeper layers of the skin, it seems unlikely that Argireline’s clinical benefit derives from a neurotoxiclike mechanism of action. It seems more likely that the Argireline-containing product enhances hydration or imparts some other quality to the skin surface. While there is certainly great appeal for a neurotoxinlike product without injections, I do not believe this ingredient will replace injections of BoNT in the foreseeable future, or at least until scientists can figure out how to enable these products to penetrate into the deeper layers of the skin.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Dr. Goldman has no relevant disclosures. Write to her at [email protected] or message her on Instagram @DrChloeGoldman.
References
1. Reddy BY et al. Exp Dermatol. 2012 Aug;21(8):569-75.
2. Blanes-Mira C et al. Int J Cosmet Sci. 2002 Oct;24(5):303-10.
3. Wang Y et al. J Cosmet Laser Ther. 2013 Aug;15(4):237-41.
4. Wang Y et al. J Cosmet Laser Ther. 2013;14(2):147-53.
5. Raikou V et al. J Cosmet Dermatol. 2017 Jun;16(2):271-8.
6. Kraeling ME et al. Cutan Ocul Toxicol. 2015 Mar;34(1):46-52.
7. Hoppel M et al. Eur J Pharm Sci. 2015 Feb 20;68:27-35.
Acetyl hexapeptide-8 (or -3), better known by its brand name, Argireline (Lubrizol; Wickliffe, Ohio), is a synthetic peptide gaining popularity in cosmeceutical products for its antiaging benefits. Argireline was developed by the company Lipotec in 2001. Media, beauty bloggers, and product claims have likened this product to a “Botox [or other neurotoxin] alternative,” or “Botox mimicker.”
Mechanism of action
Understanding how Argireline works requires a brief refresher on the mechanism of action of botulinum neurotoxin (BoNT). BoNT relaxes facial muscles and smooths expression lines by inhibiting acetylcholine release at the neuromuscular junction.1 More specifically, the various serotypes of BoNT are single-chain polypeptides that target members of the SNARE complex: SNAP-25, syntaxin, and Vamp. The proteins within the SNARE complex are involved in the docking and fusion of presynaptic vesicles to the presynaptic membrane, necessary steps for acetylcholine release into the neuromuscular junction and muscle contraction. By blocking the action of the SNARE complex proteins, BoNT inhibits release of acetylcholine in the neuromuscular junction and prevents muscle contraction.
Argireline is a synthetic peptide with the sequence Ac-EEMQRR-NH2.2 It is patterned after the N-terminal domain of SNAP-25, one of the members of the SNARE complex targeted by BoNT, and functions to interfere with the assembly of the SNARE complex. In this manner, Argireline would theoretically inhibit fusion of presynaptic vesicles and release of acetylcholine into the neuromuscular junction, thus impeding muscle movement. For this reason, it has been likened to topical Botox. Unlike Botox and other neurotoxins, Argireline was developed for topical application rather than injection.
Preclinical studies
In vitro work done 20 years ago demonstrated that Argireline can prevent assembly of the SNARE complex and inhibit neurotransmitter release with a potency similar to that of BoNT A (Botox).2
In 2013, Wang et al. evaluated the histologic effects of Argireline in aged mouse skin induced by D-galactose. For 6 weeks, Argireline was applied twice daily, and histological changes were assessed using hematoxylin and eosin (H&E) and picrosirius–polarization (PSP) stains. The researchers found elevated levels of type I collagen (P < .01) and reduced type III collagen (P < .05) with the Argireline treatment. These results demonstrated that Argireline could histologically enhance collagen in a manner consistent with skin rejuvenation.3
Clinical studies
In 2002, Blanes et al. assessed the antiwrinkle activity of Argireline by measuring skin topography from silicone implants in the lateral periorbital region of an oil/water (O/W) emulsion containing 10% of the acetyl-hexapeptide in 10 healthy women volunteers. The hexapeptide emulsion was applied twice daily in one lateral periorbital area, and the emulsion vehicle alone was applied twice daily on the contralateral side. Over 30 days of treatment, wrinkle depth was found to have decreased by 30%. The investigators also found that Argireline significantly hindered neurotransmitter release in vitro as robustly as BoNT A, though with notably lower efficacy. No toxicity or irritation was associated with this treatment.2 However, it should be noted that this small study conducted 2 decades ago evaluated only silicone implants with confocal microscopy to evaluate wrinkle depth. There was no subjective clinical assessment of dynamic facial wrinkles. As such, their study is an insufficient basis for drawing conclusions that Argireline is a BoNT mimic. Botox and other types of BoNT affect dynamic facial wrinkles mostly (i.e., wrinkles created by moving muscles of facial expression). This study primarily considers static wrinkles on periorbital skin. While static wrinkles may result from longstanding dynamic wrinkles, BoNT mainly targets dynamic wrinkles, again not comparing apples to apples.
At the same time that Wang et al. conducted their experiment on the skin of aged mice as noted above, they performed a multicenter clinical trial in 60 human subjects who received a randomized treatment of Argireline or placebo in a ratio of 3:1 to assess its safety and efficacy. For 4 weeks, the test product or placebo was applied to periorbital wrinkles twice daily. The researchers found the total antiwrinkle efficacy in the Argireline group to be 48.9% based on the subjective evaluation, compared with 0% in the placebo group. The objective evaluation indicated that all parameters of roughness were diminished in the Argireline group (P < .01), with no reduction observed in the placebo group (P < .05).4 There was a little more to appreciate from this study compared with the one reported by Blanes et al., insofar as subjective evaluations and objective evaluations with silica replicas were done. However, this study was not blinded, so the 48.9% wrinkle reduction in the Argireline group vs. 0% in the control group seems suspicious. Additionally, there was a greater focus on static rather than dynamic wrinkles.
In 2017, Raikou et al. conducted a prospective, randomized controlled study to assess the effects of acetyl hexapeptide-3 (Argireline) and tripeptide-10 citrulline in 24 healthy female volunteers (aged 30-60 years) and determine if there was any synergistic action between the peptides. Subjects were randomized to receive a combination of the peptides, tripeptide-10 citrulline only, acetyl hexapeptide-3 only, or neither peptide for 60 days. The researchers found a significant reduction in transepidermal water loss (TEWL) in the Argireline group, compared with the placebo group.5 The result of this study makes me question if the decrease in depth of the wrinkles measured in the former studies is really just a measure of increased skin hydration from the Argireline, rather than a neurotoxic effect of Argireline.
Formulation and penetration: Can Argireline get through your skin?
One of the fundamental questions regarding Argireline is whether it can penetrate through the stratum corneum and find its target – the facial muscles – where it is intended to function. Argireline is a charged, hydrophilic, and large–molecular weight peptide, and each of these factors impairs penetration through the stratum corneum. Therefore, studies assessing penetration are particularly important.
In 2015, Kraeling et al. conducted an in vitro evaluation of the skin penetration of acetyl hexapeptide-8 in hairless guinea pig and human cadaver skin. An oil-in-water (O/W) emulsion containing 10% acetyl hexapeptide-8 was applied (2 mg/cm2) and penetration was quantified in skin layers via hydrophilic interaction liquid chromatography with tandem mass spectrometry. Most of the acetyl hexapeptide-8 was found to have been washed from human cadaver, as well as guinea pig, skin. Less than 1% of the peptide penetrated the guinea pig or human skin. Of this small amount that penetrated the skin, most stayed in the stratum corneum of guinea pigs (0.54%) and human cadavers (0.22%). The levels of acetyl hexapeptide-8 declined further with each layer of tape stripping removal. Epidermal levels of the peptide in tested skin were similar at 0.01%, and none of the peptide was found in the dermis.6 These results indicate negligible penetration by this highly touted peptide ingredient.
Some studies have shown that altering the formulation of acetyl hexapeptide-8 can enhance penetration. Hoppel et al. demonstrated that formulations of the peptide, especially in a water-oil-water (W/O/W emulsion [as compared with O/W and W/O emulsions] can increase penetration into the stratum corneum in porcine skin.7 Notably, this is still very superficial relative to the dermis and muscles. Irrespective of formulation, studies have shown that Argireline barely penetrates the stratum corneum, let alone the dermis. Therefore, I would give pause to attributing any clinical impact or benefit of Argireline to its neurotoxinlike effects measured in vitro.
Conclusion
Despite the growing popularity of this ingredient in cosmeceuticals and the praise it gets in media for acting as a topical neurotoxin, there are no rigorous clinical trials or data demonstrating its efficacy in suppressing dynamic facial wrinkles like BoNT does. Most importantly, without penetration into the stratum corneum and deeper layers of the skin, it seems unlikely that Argireline’s clinical benefit derives from a neurotoxiclike mechanism of action. It seems more likely that the Argireline-containing product enhances hydration or imparts some other quality to the skin surface. While there is certainly great appeal for a neurotoxinlike product without injections, I do not believe this ingredient will replace injections of BoNT in the foreseeable future, or at least until scientists can figure out how to enable these products to penetrate into the deeper layers of the skin.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Dr. Goldman has no relevant disclosures. Write to her at [email protected] or message her on Instagram @DrChloeGoldman.
References
1. Reddy BY et al. Exp Dermatol. 2012 Aug;21(8):569-75.
2. Blanes-Mira C et al. Int J Cosmet Sci. 2002 Oct;24(5):303-10.
3. Wang Y et al. J Cosmet Laser Ther. 2013 Aug;15(4):237-41.
4. Wang Y et al. J Cosmet Laser Ther. 2013;14(2):147-53.
5. Raikou V et al. J Cosmet Dermatol. 2017 Jun;16(2):271-8.
6. Kraeling ME et al. Cutan Ocul Toxicol. 2015 Mar;34(1):46-52.
7. Hoppel M et al. Eur J Pharm Sci. 2015 Feb 20;68:27-35.
Acetyl hexapeptide-8 (or -3), better known by its brand name, Argireline (Lubrizol; Wickliffe, Ohio), is a synthetic peptide gaining popularity in cosmeceutical products for its antiaging benefits. Argireline was developed by the company Lipotec in 2001. Media, beauty bloggers, and product claims have likened this product to a “Botox [or other neurotoxin] alternative,” or “Botox mimicker.”
Mechanism of action
Understanding how Argireline works requires a brief refresher on the mechanism of action of botulinum neurotoxin (BoNT). BoNT relaxes facial muscles and smooths expression lines by inhibiting acetylcholine release at the neuromuscular junction.1 More specifically, the various serotypes of BoNT are single-chain polypeptides that target members of the SNARE complex: SNAP-25, syntaxin, and Vamp. The proteins within the SNARE complex are involved in the docking and fusion of presynaptic vesicles to the presynaptic membrane, necessary steps for acetylcholine release into the neuromuscular junction and muscle contraction. By blocking the action of the SNARE complex proteins, BoNT inhibits release of acetylcholine in the neuromuscular junction and prevents muscle contraction.
Argireline is a synthetic peptide with the sequence Ac-EEMQRR-NH2.2 It is patterned after the N-terminal domain of SNAP-25, one of the members of the SNARE complex targeted by BoNT, and functions to interfere with the assembly of the SNARE complex. In this manner, Argireline would theoretically inhibit fusion of presynaptic vesicles and release of acetylcholine into the neuromuscular junction, thus impeding muscle movement. For this reason, it has been likened to topical Botox. Unlike Botox and other neurotoxins, Argireline was developed for topical application rather than injection.
Preclinical studies
In vitro work done 20 years ago demonstrated that Argireline can prevent assembly of the SNARE complex and inhibit neurotransmitter release with a potency similar to that of BoNT A (Botox).2
In 2013, Wang et al. evaluated the histologic effects of Argireline in aged mouse skin induced by D-galactose. For 6 weeks, Argireline was applied twice daily, and histological changes were assessed using hematoxylin and eosin (H&E) and picrosirius–polarization (PSP) stains. The researchers found elevated levels of type I collagen (P < .01) and reduced type III collagen (P < .05) with the Argireline treatment. These results demonstrated that Argireline could histologically enhance collagen in a manner consistent with skin rejuvenation.3
Clinical studies
In 2002, Blanes et al. assessed the antiwrinkle activity of Argireline by measuring skin topography from silicone implants in the lateral periorbital region of an oil/water (O/W) emulsion containing 10% of the acetyl-hexapeptide in 10 healthy women volunteers. The hexapeptide emulsion was applied twice daily in one lateral periorbital area, and the emulsion vehicle alone was applied twice daily on the contralateral side. Over 30 days of treatment, wrinkle depth was found to have decreased by 30%. The investigators also found that Argireline significantly hindered neurotransmitter release in vitro as robustly as BoNT A, though with notably lower efficacy. No toxicity or irritation was associated with this treatment.2 However, it should be noted that this small study conducted 2 decades ago evaluated only silicone implants with confocal microscopy to evaluate wrinkle depth. There was no subjective clinical assessment of dynamic facial wrinkles. As such, their study is an insufficient basis for drawing conclusions that Argireline is a BoNT mimic. Botox and other types of BoNT affect dynamic facial wrinkles mostly (i.e., wrinkles created by moving muscles of facial expression). This study primarily considers static wrinkles on periorbital skin. While static wrinkles may result from longstanding dynamic wrinkles, BoNT mainly targets dynamic wrinkles, again not comparing apples to apples.
At the same time that Wang et al. conducted their experiment on the skin of aged mice as noted above, they performed a multicenter clinical trial in 60 human subjects who received a randomized treatment of Argireline or placebo in a ratio of 3:1 to assess its safety and efficacy. For 4 weeks, the test product or placebo was applied to periorbital wrinkles twice daily. The researchers found the total antiwrinkle efficacy in the Argireline group to be 48.9% based on the subjective evaluation, compared with 0% in the placebo group. The objective evaluation indicated that all parameters of roughness were diminished in the Argireline group (P < .01), with no reduction observed in the placebo group (P < .05).4 There was a little more to appreciate from this study compared with the one reported by Blanes et al., insofar as subjective evaluations and objective evaluations with silica replicas were done. However, this study was not blinded, so the 48.9% wrinkle reduction in the Argireline group vs. 0% in the control group seems suspicious. Additionally, there was a greater focus on static rather than dynamic wrinkles.
In 2017, Raikou et al. conducted a prospective, randomized controlled study to assess the effects of acetyl hexapeptide-3 (Argireline) and tripeptide-10 citrulline in 24 healthy female volunteers (aged 30-60 years) and determine if there was any synergistic action between the peptides. Subjects were randomized to receive a combination of the peptides, tripeptide-10 citrulline only, acetyl hexapeptide-3 only, or neither peptide for 60 days. The researchers found a significant reduction in transepidermal water loss (TEWL) in the Argireline group, compared with the placebo group.5 The result of this study makes me question if the decrease in depth of the wrinkles measured in the former studies is really just a measure of increased skin hydration from the Argireline, rather than a neurotoxic effect of Argireline.
Formulation and penetration: Can Argireline get through your skin?
One of the fundamental questions regarding Argireline is whether it can penetrate through the stratum corneum and find its target – the facial muscles – where it is intended to function. Argireline is a charged, hydrophilic, and large–molecular weight peptide, and each of these factors impairs penetration through the stratum corneum. Therefore, studies assessing penetration are particularly important.
In 2015, Kraeling et al. conducted an in vitro evaluation of the skin penetration of acetyl hexapeptide-8 in hairless guinea pig and human cadaver skin. An oil-in-water (O/W) emulsion containing 10% acetyl hexapeptide-8 was applied (2 mg/cm2) and penetration was quantified in skin layers via hydrophilic interaction liquid chromatography with tandem mass spectrometry. Most of the acetyl hexapeptide-8 was found to have been washed from human cadaver, as well as guinea pig, skin. Less than 1% of the peptide penetrated the guinea pig or human skin. Of this small amount that penetrated the skin, most stayed in the stratum corneum of guinea pigs (0.54%) and human cadavers (0.22%). The levels of acetyl hexapeptide-8 declined further with each layer of tape stripping removal. Epidermal levels of the peptide in tested skin were similar at 0.01%, and none of the peptide was found in the dermis.6 These results indicate negligible penetration by this highly touted peptide ingredient.
Some studies have shown that altering the formulation of acetyl hexapeptide-8 can enhance penetration. Hoppel et al. demonstrated that formulations of the peptide, especially in a water-oil-water (W/O/W emulsion [as compared with O/W and W/O emulsions] can increase penetration into the stratum corneum in porcine skin.7 Notably, this is still very superficial relative to the dermis and muscles. Irrespective of formulation, studies have shown that Argireline barely penetrates the stratum corneum, let alone the dermis. Therefore, I would give pause to attributing any clinical impact or benefit of Argireline to its neurotoxinlike effects measured in vitro.
Conclusion
Despite the growing popularity of this ingredient in cosmeceuticals and the praise it gets in media for acting as a topical neurotoxin, there are no rigorous clinical trials or data demonstrating its efficacy in suppressing dynamic facial wrinkles like BoNT does. Most importantly, without penetration into the stratum corneum and deeper layers of the skin, it seems unlikely that Argireline’s clinical benefit derives from a neurotoxiclike mechanism of action. It seems more likely that the Argireline-containing product enhances hydration or imparts some other quality to the skin surface. While there is certainly great appeal for a neurotoxinlike product without injections, I do not believe this ingredient will replace injections of BoNT in the foreseeable future, or at least until scientists can figure out how to enable these products to penetrate into the deeper layers of the skin.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Dr. Goldman has no relevant disclosures. Write to her at [email protected] or message her on Instagram @DrChloeGoldman.
References
1. Reddy BY et al. Exp Dermatol. 2012 Aug;21(8):569-75.
2. Blanes-Mira C et al. Int J Cosmet Sci. 2002 Oct;24(5):303-10.
3. Wang Y et al. J Cosmet Laser Ther. 2013 Aug;15(4):237-41.
4. Wang Y et al. J Cosmet Laser Ther. 2013;14(2):147-53.
5. Raikou V et al. J Cosmet Dermatol. 2017 Jun;16(2):271-8.
6. Kraeling ME et al. Cutan Ocul Toxicol. 2015 Mar;34(1):46-52.
7. Hoppel M et al. Eur J Pharm Sci. 2015 Feb 20;68:27-35.
The shell game
The shell game is probably the world’s oldest confidence game, going back 2,000-3,000 years. You still see people getting lured into it in cities today.
It’s also played, albeit legally, in physician offices and pharmacies around the country.
You know, the ones piled up in your sample cabinet. Get a month free of Whateveritscalled to try and/or have your copay reduced to $0 or something reasonably low. All, of course, subject to terms and conditions in the small print and that of your insurance carrier. Your mileage may vary.
In my experience these things often don’t work as well as advertised. Sometimes it’s because the patient doesn’t understand how to use them, other times because their pharmacy doesn’t want to have anything to do with the cards, and still other times because their insurance has some exclusion against them.
But they do sometimes work ... until they don’t. Sometimes, out of the blue, they’ll stop. Maybe there was an insurance change, or the pharmacy policy changed, or the manufacturer’s program changed, or the offer expired. I usually don’t know and rarely find out.
So the shell game begins. The patients call multiple pharmacies, trying to find one that may be able to honor the coupons. Then they call my office and ask me to switch the script. So I do. They they go to the pharmacy. Sometimes they can get the script, sometimes not. If not, they move to another pharmacy and call my office to switch it again. Wash, rinse, repeat.
Other times it’s more complicated. They want a new card for each try, so they show up at my office asking for one (usually they can be downloaded online so some try that. Others do both). Like pebbles under a shell, the prescriptions and cards get switched from pharmacy to pharmacy.
This isn’t exclusive to manufacturers’ copay cards. I see the same phenomenon with GoodRx and similar cost-saving programs. People move scripts around to find the best deal. It may be helping them, but it certainly doesn’t help me and my staff as we try to keep up, or the badly overworked pharmacy staff.
I’ve even had some patients take the audacious step of asking me to write a script in the name of their spouses so they can double their supply. Obviously, such requests are refused. I’m not going to play that game.
I understand new drugs are costly, and often outside the reach of many patients today. I know the manufacturers are trying to get their products tried, or even make them more affordable for those who need it.
But, in many cases, this becomes (unintentionally or intentionally; I’m not sure) a shell game. Legal, perhaps, but still equally frustrating for all of us who are trying to keep up with it.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
The shell game is probably the world’s oldest confidence game, going back 2,000-3,000 years. You still see people getting lured into it in cities today.
It’s also played, albeit legally, in physician offices and pharmacies around the country.
You know, the ones piled up in your sample cabinet. Get a month free of Whateveritscalled to try and/or have your copay reduced to $0 or something reasonably low. All, of course, subject to terms and conditions in the small print and that of your insurance carrier. Your mileage may vary.
In my experience these things often don’t work as well as advertised. Sometimes it’s because the patient doesn’t understand how to use them, other times because their pharmacy doesn’t want to have anything to do with the cards, and still other times because their insurance has some exclusion against them.
But they do sometimes work ... until they don’t. Sometimes, out of the blue, they’ll stop. Maybe there was an insurance change, or the pharmacy policy changed, or the manufacturer’s program changed, or the offer expired. I usually don’t know and rarely find out.
So the shell game begins. The patients call multiple pharmacies, trying to find one that may be able to honor the coupons. Then they call my office and ask me to switch the script. So I do. They they go to the pharmacy. Sometimes they can get the script, sometimes not. If not, they move to another pharmacy and call my office to switch it again. Wash, rinse, repeat.
Other times it’s more complicated. They want a new card for each try, so they show up at my office asking for one (usually they can be downloaded online so some try that. Others do both). Like pebbles under a shell, the prescriptions and cards get switched from pharmacy to pharmacy.
This isn’t exclusive to manufacturers’ copay cards. I see the same phenomenon with GoodRx and similar cost-saving programs. People move scripts around to find the best deal. It may be helping them, but it certainly doesn’t help me and my staff as we try to keep up, or the badly overworked pharmacy staff.
I’ve even had some patients take the audacious step of asking me to write a script in the name of their spouses so they can double their supply. Obviously, such requests are refused. I’m not going to play that game.
I understand new drugs are costly, and often outside the reach of many patients today. I know the manufacturers are trying to get their products tried, or even make them more affordable for those who need it.
But, in many cases, this becomes (unintentionally or intentionally; I’m not sure) a shell game. Legal, perhaps, but still equally frustrating for all of us who are trying to keep up with it.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
The shell game is probably the world’s oldest confidence game, going back 2,000-3,000 years. You still see people getting lured into it in cities today.
It’s also played, albeit legally, in physician offices and pharmacies around the country.
You know, the ones piled up in your sample cabinet. Get a month free of Whateveritscalled to try and/or have your copay reduced to $0 or something reasonably low. All, of course, subject to terms and conditions in the small print and that of your insurance carrier. Your mileage may vary.
In my experience these things often don’t work as well as advertised. Sometimes it’s because the patient doesn’t understand how to use them, other times because their pharmacy doesn’t want to have anything to do with the cards, and still other times because their insurance has some exclusion against them.
But they do sometimes work ... until they don’t. Sometimes, out of the blue, they’ll stop. Maybe there was an insurance change, or the pharmacy policy changed, or the manufacturer’s program changed, or the offer expired. I usually don’t know and rarely find out.
So the shell game begins. The patients call multiple pharmacies, trying to find one that may be able to honor the coupons. Then they call my office and ask me to switch the script. So I do. They they go to the pharmacy. Sometimes they can get the script, sometimes not. If not, they move to another pharmacy and call my office to switch it again. Wash, rinse, repeat.
Other times it’s more complicated. They want a new card for each try, so they show up at my office asking for one (usually they can be downloaded online so some try that. Others do both). Like pebbles under a shell, the prescriptions and cards get switched from pharmacy to pharmacy.
This isn’t exclusive to manufacturers’ copay cards. I see the same phenomenon with GoodRx and similar cost-saving programs. People move scripts around to find the best deal. It may be helping them, but it certainly doesn’t help me and my staff as we try to keep up, or the badly overworked pharmacy staff.
I’ve even had some patients take the audacious step of asking me to write a script in the name of their spouses so they can double their supply. Obviously, such requests are refused. I’m not going to play that game.
I understand new drugs are costly, and often outside the reach of many patients today. I know the manufacturers are trying to get their products tried, or even make them more affordable for those who need it.
But, in many cases, this becomes (unintentionally or intentionally; I’m not sure) a shell game. Legal, perhaps, but still equally frustrating for all of us who are trying to keep up with it.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Our role in preventing postpartum depression
Tragic, embarrassing, criminal ... Choose your own adjective. The maternal mortality rate in this country is the worst of any developed nation in the world. And the numbers are getting worse with an increase of 14% over the previous year. One-third of these deaths occur weeks or months after the delivery.
In a recent issue of Harvard Public Health, researchers at the T.H. Chan School of Public Health discuss some of the possible remedies for what they describe as a crisis. While some of the solutions they list will require major restructuring of how we deliver health care to mothers, others could take advantage of our current systems by employing a slight shift in emphasis. And here is where those of us on the frontline of care delivery can make a difference.
The researchers point out that “More than 90% of maternal deaths could be prevented if women had access to quality care.” They also observe that most mothers have a single postpartum check with the ob.gyn. facility that delivered the baby and then are often left to navigate the health system because transfer to their primary care and/or mental health professional is haphazard or lacking in follow-up.
As I read through the article it struck me that as pediatricians we could and should be playing a larger role in this critical postpartum period when so many women seem to be falling through the cracks in our health care nonsystem. This is not a great “Ah-ha” moment for which I deserve any credit. In 2010 the American Academy of Pediatrics recommended that mothers be screened for depression at the 1-, 2-, and 4-month visits using either a validated 10-question screening instrument or a more direct 2-question tool (Pediatrics 2010;126[5]:1032-9). However, a periodic survey of AAP members 3 years later revealed that less than a third of the respondents were screening regularly for postpartum depression. In 2019 the academy reemphasized the important role that pediatric primary care givers can play in the detection and early management of the condition.
The reasons for the disappointing response include the list of usual suspects of inadequate training, workload demands, reimbursement, liability concerns, and the difficulty in finding and establishing effective referral networks. Unfortunately, these factors continue to exist, and many cases have multiplied in the wake of the pandemic.
In some states, educational outreach, funding, and changes in the reimbursement structure have resulted in improved outcomes. Not all of us are fortunate enough to live in a state that has made postpartum depression detection and management a priority. However, simply making it our own professional priority can save lives, ease suffering, and improve postpartum outcomes. Here I am talking about first caring and then inquiring about a mother’s mental health. Asking how much sleep she is getting. And then spending the time to give personalized advice on feeding and sleep schedules. Even, if this means ignoring half of the topics on the recommended health maintenance. It doesn’t take but a few minutes to convince yourself that the baby is healthy, and you know that 90% of them are.
However, a new mother who is sleep deprived and already has one foot on the spiral staircase down into postpartum depression represents an emergency. And, you should have the skills to turn it around. But, you have to care about the problem and make it your own priority – high enough on the list to make a follow-up appointment or call in a week instead of waiting a month or 2 until the next visit.
Unfortunately, even with your best efforts there are some families who need services beyond the scope of your practice. Making the necessary referrals can be frustrating and time consuming but not dropping ball until it lands in the appropriate place may save a life.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Tragic, embarrassing, criminal ... Choose your own adjective. The maternal mortality rate in this country is the worst of any developed nation in the world. And the numbers are getting worse with an increase of 14% over the previous year. One-third of these deaths occur weeks or months after the delivery.
In a recent issue of Harvard Public Health, researchers at the T.H. Chan School of Public Health discuss some of the possible remedies for what they describe as a crisis. While some of the solutions they list will require major restructuring of how we deliver health care to mothers, others could take advantage of our current systems by employing a slight shift in emphasis. And here is where those of us on the frontline of care delivery can make a difference.
The researchers point out that “More than 90% of maternal deaths could be prevented if women had access to quality care.” They also observe that most mothers have a single postpartum check with the ob.gyn. facility that delivered the baby and then are often left to navigate the health system because transfer to their primary care and/or mental health professional is haphazard or lacking in follow-up.
As I read through the article it struck me that as pediatricians we could and should be playing a larger role in this critical postpartum period when so many women seem to be falling through the cracks in our health care nonsystem. This is not a great “Ah-ha” moment for which I deserve any credit. In 2010 the American Academy of Pediatrics recommended that mothers be screened for depression at the 1-, 2-, and 4-month visits using either a validated 10-question screening instrument or a more direct 2-question tool (Pediatrics 2010;126[5]:1032-9). However, a periodic survey of AAP members 3 years later revealed that less than a third of the respondents were screening regularly for postpartum depression. In 2019 the academy reemphasized the important role that pediatric primary care givers can play in the detection and early management of the condition.
The reasons for the disappointing response include the list of usual suspects of inadequate training, workload demands, reimbursement, liability concerns, and the difficulty in finding and establishing effective referral networks. Unfortunately, these factors continue to exist, and many cases have multiplied in the wake of the pandemic.
In some states, educational outreach, funding, and changes in the reimbursement structure have resulted in improved outcomes. Not all of us are fortunate enough to live in a state that has made postpartum depression detection and management a priority. However, simply making it our own professional priority can save lives, ease suffering, and improve postpartum outcomes. Here I am talking about first caring and then inquiring about a mother’s mental health. Asking how much sleep she is getting. And then spending the time to give personalized advice on feeding and sleep schedules. Even, if this means ignoring half of the topics on the recommended health maintenance. It doesn’t take but a few minutes to convince yourself that the baby is healthy, and you know that 90% of them are.
However, a new mother who is sleep deprived and already has one foot on the spiral staircase down into postpartum depression represents an emergency. And, you should have the skills to turn it around. But, you have to care about the problem and make it your own priority – high enough on the list to make a follow-up appointment or call in a week instead of waiting a month or 2 until the next visit.
Unfortunately, even with your best efforts there are some families who need services beyond the scope of your practice. Making the necessary referrals can be frustrating and time consuming but not dropping ball until it lands in the appropriate place may save a life.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Tragic, embarrassing, criminal ... Choose your own adjective. The maternal mortality rate in this country is the worst of any developed nation in the world. And the numbers are getting worse with an increase of 14% over the previous year. One-third of these deaths occur weeks or months after the delivery.
In a recent issue of Harvard Public Health, researchers at the T.H. Chan School of Public Health discuss some of the possible remedies for what they describe as a crisis. While some of the solutions they list will require major restructuring of how we deliver health care to mothers, others could take advantage of our current systems by employing a slight shift in emphasis. And here is where those of us on the frontline of care delivery can make a difference.
The researchers point out that “More than 90% of maternal deaths could be prevented if women had access to quality care.” They also observe that most mothers have a single postpartum check with the ob.gyn. facility that delivered the baby and then are often left to navigate the health system because transfer to their primary care and/or mental health professional is haphazard or lacking in follow-up.
As I read through the article it struck me that as pediatricians we could and should be playing a larger role in this critical postpartum period when so many women seem to be falling through the cracks in our health care nonsystem. This is not a great “Ah-ha” moment for which I deserve any credit. In 2010 the American Academy of Pediatrics recommended that mothers be screened for depression at the 1-, 2-, and 4-month visits using either a validated 10-question screening instrument or a more direct 2-question tool (Pediatrics 2010;126[5]:1032-9). However, a periodic survey of AAP members 3 years later revealed that less than a third of the respondents were screening regularly for postpartum depression. In 2019 the academy reemphasized the important role that pediatric primary care givers can play in the detection and early management of the condition.
The reasons for the disappointing response include the list of usual suspects of inadequate training, workload demands, reimbursement, liability concerns, and the difficulty in finding and establishing effective referral networks. Unfortunately, these factors continue to exist, and many cases have multiplied in the wake of the pandemic.
In some states, educational outreach, funding, and changes in the reimbursement structure have resulted in improved outcomes. Not all of us are fortunate enough to live in a state that has made postpartum depression detection and management a priority. However, simply making it our own professional priority can save lives, ease suffering, and improve postpartum outcomes. Here I am talking about first caring and then inquiring about a mother’s mental health. Asking how much sleep she is getting. And then spending the time to give personalized advice on feeding and sleep schedules. Even, if this means ignoring half of the topics on the recommended health maintenance. It doesn’t take but a few minutes to convince yourself that the baby is healthy, and you know that 90% of them are.
However, a new mother who is sleep deprived and already has one foot on the spiral staircase down into postpartum depression represents an emergency. And, you should have the skills to turn it around. But, you have to care about the problem and make it your own priority – high enough on the list to make a follow-up appointment or call in a week instead of waiting a month or 2 until the next visit.
Unfortunately, even with your best efforts there are some families who need services beyond the scope of your practice. Making the necessary referrals can be frustrating and time consuming but not dropping ball until it lands in the appropriate place may save a life.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].