User login
Pediatric atopic dermatitis and neuropsychiatric disorders: What is the link?
Key clinical point: Atopic dermatitis (AD) did not increase the incidence risk for most neuropsychiatric disorders in a pediatric cohort.
Major finding: The risks for attention deficit hyperactivity disorder (hazard ratio [HR] 1.02; 95% CI 0.97-1.06), autism (HR 1.02; 95% CI 0.98-1.06), anxiety (HR 1.01; 95% CI 0.99-1.03), and bipolar disorder (HR 1.08; 95% CI 0.85-1.36) were comparable in the AD and non-AD groups. Participants with vs without AD were less likely to develop depression (HR 0.93; 95% CI 0.91-0.95) or schizophrenia (HR 0.72; 95% CI 0.54-0.95) but more likely to develop obsessive compulsive disorder (HR 1.26; 95% CI 1.16-1.37). However, the risks varied with disease severity and patient’s age.
Study details: Findings are from a retrospective population-based cohort study including 409,431 children with AD and 1,809,029 matched children without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. One author declared being an employee of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Wan J et al. Atopic dermatitis and risk of major neuropsychiatric disorders in children: A population-based cohort study. J Eur Acad Dermatol Venereol. 2022 (Aug 26). Doi: 10.1111/jdv.18564
Key clinical point: Atopic dermatitis (AD) did not increase the incidence risk for most neuropsychiatric disorders in a pediatric cohort.
Major finding: The risks for attention deficit hyperactivity disorder (hazard ratio [HR] 1.02; 95% CI 0.97-1.06), autism (HR 1.02; 95% CI 0.98-1.06), anxiety (HR 1.01; 95% CI 0.99-1.03), and bipolar disorder (HR 1.08; 95% CI 0.85-1.36) were comparable in the AD and non-AD groups. Participants with vs without AD were less likely to develop depression (HR 0.93; 95% CI 0.91-0.95) or schizophrenia (HR 0.72; 95% CI 0.54-0.95) but more likely to develop obsessive compulsive disorder (HR 1.26; 95% CI 1.16-1.37). However, the risks varied with disease severity and patient’s age.
Study details: Findings are from a retrospective population-based cohort study including 409,431 children with AD and 1,809,029 matched children without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. One author declared being an employee of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Wan J et al. Atopic dermatitis and risk of major neuropsychiatric disorders in children: A population-based cohort study. J Eur Acad Dermatol Venereol. 2022 (Aug 26). Doi: 10.1111/jdv.18564
Key clinical point: Atopic dermatitis (AD) did not increase the incidence risk for most neuropsychiatric disorders in a pediatric cohort.
Major finding: The risks for attention deficit hyperactivity disorder (hazard ratio [HR] 1.02; 95% CI 0.97-1.06), autism (HR 1.02; 95% CI 0.98-1.06), anxiety (HR 1.01; 95% CI 0.99-1.03), and bipolar disorder (HR 1.08; 95% CI 0.85-1.36) were comparable in the AD and non-AD groups. Participants with vs without AD were less likely to develop depression (HR 0.93; 95% CI 0.91-0.95) or schizophrenia (HR 0.72; 95% CI 0.54-0.95) but more likely to develop obsessive compulsive disorder (HR 1.26; 95% CI 1.16-1.37). However, the risks varied with disease severity and patient’s age.
Study details: Findings are from a retrospective population-based cohort study including 409,431 children with AD and 1,809,029 matched children without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. One author declared being an employee of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Wan J et al. Atopic dermatitis and risk of major neuropsychiatric disorders in children: A population-based cohort study. J Eur Acad Dermatol Venereol. 2022 (Aug 26). Doi: 10.1111/jdv.18564
Early initiation of emollient reduces risk for atopic dermatitis in high risk infants
Key clinical point: Initiation of daily application of a specialized emollient from the first to the eighth week of life reduced the risk for atopic dermatitis (AD) incidence for 12 months in infants with high risk for AD.
Major finding: At 12 months, the cumulative incidence of AD was significantly lower in the emollient vs standard routine skin care group (32.8% vs 46.4%; relative risk 0.707; P = .036). The rate of patient-reported skin infections was similar between both the treatment groups during the 8-week intervention period (5.0% vs 5.7%).
Study details: Findings are from the STOP AD trial including 321 newborn infants at high risk for AD who were randomly assigned to receive twice-daily emollient for the first 8 weeks of life or standard routine skin care.
Disclosures: This study was supported by The City of Dublin Skin and Cancer Hospital Charity and the Skin Research Institute of Singapore. Some authors declared being managing directors, employees, shareholders, or consultants or receiving research funding, speaker fees, or consulting fees from several sources.
Source: Ní Chaoimh C, Lad D, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high risk infants - the STOP AD randomised controlled trial. Allergy. 2022 (Aug 23). Doi: 10.1111/all.15491
Key clinical point: Initiation of daily application of a specialized emollient from the first to the eighth week of life reduced the risk for atopic dermatitis (AD) incidence for 12 months in infants with high risk for AD.
Major finding: At 12 months, the cumulative incidence of AD was significantly lower in the emollient vs standard routine skin care group (32.8% vs 46.4%; relative risk 0.707; P = .036). The rate of patient-reported skin infections was similar between both the treatment groups during the 8-week intervention period (5.0% vs 5.7%).
Study details: Findings are from the STOP AD trial including 321 newborn infants at high risk for AD who were randomly assigned to receive twice-daily emollient for the first 8 weeks of life or standard routine skin care.
Disclosures: This study was supported by The City of Dublin Skin and Cancer Hospital Charity and the Skin Research Institute of Singapore. Some authors declared being managing directors, employees, shareholders, or consultants or receiving research funding, speaker fees, or consulting fees from several sources.
Source: Ní Chaoimh C, Lad D, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high risk infants - the STOP AD randomised controlled trial. Allergy. 2022 (Aug 23). Doi: 10.1111/all.15491
Key clinical point: Initiation of daily application of a specialized emollient from the first to the eighth week of life reduced the risk for atopic dermatitis (AD) incidence for 12 months in infants with high risk for AD.
Major finding: At 12 months, the cumulative incidence of AD was significantly lower in the emollient vs standard routine skin care group (32.8% vs 46.4%; relative risk 0.707; P = .036). The rate of patient-reported skin infections was similar between both the treatment groups during the 8-week intervention period (5.0% vs 5.7%).
Study details: Findings are from the STOP AD trial including 321 newborn infants at high risk for AD who were randomly assigned to receive twice-daily emollient for the first 8 weeks of life or standard routine skin care.
Disclosures: This study was supported by The City of Dublin Skin and Cancer Hospital Charity and the Skin Research Institute of Singapore. Some authors declared being managing directors, employees, shareholders, or consultants or receiving research funding, speaker fees, or consulting fees from several sources.
Source: Ní Chaoimh C, Lad D, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high risk infants - the STOP AD randomised controlled trial. Allergy. 2022 (Aug 23). Doi: 10.1111/all.15491
Moderate-to-severe atopic dermatitis: Astegolimab fails to reduce disease severity in phase 2 trial
Key clinical point: Astegolimab, despite being well-tolerated, did not lessen the severity of the disease in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: At week 16, the adjusted mean percent changes in the mean Eczema Area and Severity Index score were comparable in the astegolimab (−51.47%) and placebo (−58.24%) groups (
Study details: Findings are from a phase 2 trial including 65 adult patients with moderate-to-severe AD and inadequate response to topical medications who were randomly assigned to receive 490 mg astegolimab or placebo every 4 weeks for 16 weeks and were further followed-up for 8 weeks.
Disclosures: This study was supported by Genentech, Inc. Six authors declared serving as employees of Genentech, Inc., a member of the Roche group, and owning stocks in Roche. The other authors reported ties with several sources, including Roche.
Source: Maurer M et al. Phase 2 randomized clinical trial of astegolimab in patients with moderate to severe atopic dermatitis. J Allergy Clin Immunol. 2022 (Aug 27). Doi: 10.1016/j.jaci.2022.08.015
Key clinical point: Astegolimab, despite being well-tolerated, did not lessen the severity of the disease in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: At week 16, the adjusted mean percent changes in the mean Eczema Area and Severity Index score were comparable in the astegolimab (−51.47%) and placebo (−58.24%) groups (
Study details: Findings are from a phase 2 trial including 65 adult patients with moderate-to-severe AD and inadequate response to topical medications who were randomly assigned to receive 490 mg astegolimab or placebo every 4 weeks for 16 weeks and were further followed-up for 8 weeks.
Disclosures: This study was supported by Genentech, Inc. Six authors declared serving as employees of Genentech, Inc., a member of the Roche group, and owning stocks in Roche. The other authors reported ties with several sources, including Roche.
Source: Maurer M et al. Phase 2 randomized clinical trial of astegolimab in patients with moderate to severe atopic dermatitis. J Allergy Clin Immunol. 2022 (Aug 27). Doi: 10.1016/j.jaci.2022.08.015
Key clinical point: Astegolimab, despite being well-tolerated, did not lessen the severity of the disease in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: At week 16, the adjusted mean percent changes in the mean Eczema Area and Severity Index score were comparable in the astegolimab (−51.47%) and placebo (−58.24%) groups (
Study details: Findings are from a phase 2 trial including 65 adult patients with moderate-to-severe AD and inadequate response to topical medications who were randomly assigned to receive 490 mg astegolimab or placebo every 4 weeks for 16 weeks and were further followed-up for 8 weeks.
Disclosures: This study was supported by Genentech, Inc. Six authors declared serving as employees of Genentech, Inc., a member of the Roche group, and owning stocks in Roche. The other authors reported ties with several sources, including Roche.
Source: Maurer M et al. Phase 2 randomized clinical trial of astegolimab in patients with moderate to severe atopic dermatitis. J Allergy Clin Immunol. 2022 (Aug 27). Doi: 10.1016/j.jaci.2022.08.015
EHA cream shows promise in children with mild-to-moderate atopic dermatitis
Key clinical point: A cream containing 1% ectoine and 0.1% hyaluronic acid (EHA) demonstrated superior efficacy than a vehicle cream and was well-tolerated in children aged 2-18 years with mild-to-moderate atopic dermatitis (AD).
Major finding: At week 4, patients receiving EHA cream achieved a significantly higher clinical improvement in SCORing AD (mean difference [MD] −6.62; P < .001) and Investigator’s Global Assessment scores (MD −0.69; P < .001) than those receiving vehicle cream. Mild adverse events like skin erythema, pruritus, and burning skin were reported by 23.5% of patients receiving EHA cream and 5.7% of patients receiving vehicle cream.
Study details: Findings are from an observer-blind, multicenter clinical trial including 57 children aged 2-18 years with mild-to-moderate AD who were randomly assigned to receive EHA or vehicle cream twice daily for 4 weeks.
Disclosures: This study was funded by BODERM SA. The authors declared no conflicts of interest.
Source: Alexopoulos A et al. A randomized, observer-blind, vehicle-control, multi-center clinical investigation for assessing the efficacy and tolerability of a 1% ectoine and hyaluronic acid 0.1%-containing medical device in pediatric patients with mild-to-moderate atopic dermatitis. Pediatr Dermatol. 2022 (Aug 29). Doi: 10.1111/pde.15117
Key clinical point: A cream containing 1% ectoine and 0.1% hyaluronic acid (EHA) demonstrated superior efficacy than a vehicle cream and was well-tolerated in children aged 2-18 years with mild-to-moderate atopic dermatitis (AD).
Major finding: At week 4, patients receiving EHA cream achieved a significantly higher clinical improvement in SCORing AD (mean difference [MD] −6.62; P < .001) and Investigator’s Global Assessment scores (MD −0.69; P < .001) than those receiving vehicle cream. Mild adverse events like skin erythema, pruritus, and burning skin were reported by 23.5% of patients receiving EHA cream and 5.7% of patients receiving vehicle cream.
Study details: Findings are from an observer-blind, multicenter clinical trial including 57 children aged 2-18 years with mild-to-moderate AD who were randomly assigned to receive EHA or vehicle cream twice daily for 4 weeks.
Disclosures: This study was funded by BODERM SA. The authors declared no conflicts of interest.
Source: Alexopoulos A et al. A randomized, observer-blind, vehicle-control, multi-center clinical investigation for assessing the efficacy and tolerability of a 1% ectoine and hyaluronic acid 0.1%-containing medical device in pediatric patients with mild-to-moderate atopic dermatitis. Pediatr Dermatol. 2022 (Aug 29). Doi: 10.1111/pde.15117
Key clinical point: A cream containing 1% ectoine and 0.1% hyaluronic acid (EHA) demonstrated superior efficacy than a vehicle cream and was well-tolerated in children aged 2-18 years with mild-to-moderate atopic dermatitis (AD).
Major finding: At week 4, patients receiving EHA cream achieved a significantly higher clinical improvement in SCORing AD (mean difference [MD] −6.62; P < .001) and Investigator’s Global Assessment scores (MD −0.69; P < .001) than those receiving vehicle cream. Mild adverse events like skin erythema, pruritus, and burning skin were reported by 23.5% of patients receiving EHA cream and 5.7% of patients receiving vehicle cream.
Study details: Findings are from an observer-blind, multicenter clinical trial including 57 children aged 2-18 years with mild-to-moderate AD who were randomly assigned to receive EHA or vehicle cream twice daily for 4 weeks.
Disclosures: This study was funded by BODERM SA. The authors declared no conflicts of interest.
Source: Alexopoulos A et al. A randomized, observer-blind, vehicle-control, multi-center clinical investigation for assessing the efficacy and tolerability of a 1% ectoine and hyaluronic acid 0.1%-containing medical device in pediatric patients with mild-to-moderate atopic dermatitis. Pediatr Dermatol. 2022 (Aug 29). Doi: 10.1111/pde.15117
Rapid itch reduction with ruxolitinib in mild-to-moderate atopic dermatitis
Key clinical point: Ruxolitinib cream demonstrated rapid and sustained improvement in itch in patients with mild-to-moderate atopic dermatitis (AD).
Major finding: A significantly higher proportion of patients receiving ruxolitinib (0.75% or 1.5%) vs vehicle cream achieved ≥2-point reduction in itch numerical rating scale (NRS2) score as rapidly as within 12 hours (16.3% and 13.1% vs 6.9%; P < .05), with further improvements at week 8 (58.3% and 65.1% vs 29.4%; P < .0001). In patients receiving ruxolitinib (0.75% or 1.5%) vs vehicle cream, time to achieve itch NRS2 score was shorter (5 and 4 vs 17 days).
Study details: Findings are from a pooled analysis of two phase 3 trials, TRuE-AD1 and TRuE-AD2, including 1249 patients with mild-to-moderate AD who were randomly assigned to receive ruxolitinib (0.75% or 1.5%) or vehicle cream twice daily for 8 weeks.
Disclosures: This study was funded by Incyte Corporation. Three authors declared being employees and shareholders of Incyte Corporation. The other authors declared serving as scientific advisors, investigators, or consultants or receiving research grants and honoraria from several sources.
Source: Blauvelt A et al. Rapid pruritus reduction with ruxolitinib cream treatment in patients with atopic dermatitis. J Eur Acad Dermatol Venereol. 2022 (Sep 6). Doi: 10.1111/jdv.18571
Key clinical point: Ruxolitinib cream demonstrated rapid and sustained improvement in itch in patients with mild-to-moderate atopic dermatitis (AD).
Major finding: A significantly higher proportion of patients receiving ruxolitinib (0.75% or 1.5%) vs vehicle cream achieved ≥2-point reduction in itch numerical rating scale (NRS2) score as rapidly as within 12 hours (16.3% and 13.1% vs 6.9%; P < .05), with further improvements at week 8 (58.3% and 65.1% vs 29.4%; P < .0001). In patients receiving ruxolitinib (0.75% or 1.5%) vs vehicle cream, time to achieve itch NRS2 score was shorter (5 and 4 vs 17 days).
Study details: Findings are from a pooled analysis of two phase 3 trials, TRuE-AD1 and TRuE-AD2, including 1249 patients with mild-to-moderate AD who were randomly assigned to receive ruxolitinib (0.75% or 1.5%) or vehicle cream twice daily for 8 weeks.
Disclosures: This study was funded by Incyte Corporation. Three authors declared being employees and shareholders of Incyte Corporation. The other authors declared serving as scientific advisors, investigators, or consultants or receiving research grants and honoraria from several sources.
Source: Blauvelt A et al. Rapid pruritus reduction with ruxolitinib cream treatment in patients with atopic dermatitis. J Eur Acad Dermatol Venereol. 2022 (Sep 6). Doi: 10.1111/jdv.18571
Key clinical point: Ruxolitinib cream demonstrated rapid and sustained improvement in itch in patients with mild-to-moderate atopic dermatitis (AD).
Major finding: A significantly higher proportion of patients receiving ruxolitinib (0.75% or 1.5%) vs vehicle cream achieved ≥2-point reduction in itch numerical rating scale (NRS2) score as rapidly as within 12 hours (16.3% and 13.1% vs 6.9%; P < .05), with further improvements at week 8 (58.3% and 65.1% vs 29.4%; P < .0001). In patients receiving ruxolitinib (0.75% or 1.5%) vs vehicle cream, time to achieve itch NRS2 score was shorter (5 and 4 vs 17 days).
Study details: Findings are from a pooled analysis of two phase 3 trials, TRuE-AD1 and TRuE-AD2, including 1249 patients with mild-to-moderate AD who were randomly assigned to receive ruxolitinib (0.75% or 1.5%) or vehicle cream twice daily for 8 weeks.
Disclosures: This study was funded by Incyte Corporation. Three authors declared being employees and shareholders of Incyte Corporation. The other authors declared serving as scientific advisors, investigators, or consultants or receiving research grants and honoraria from several sources.
Source: Blauvelt A et al. Rapid pruritus reduction with ruxolitinib cream treatment in patients with atopic dermatitis. J Eur Acad Dermatol Venereol. 2022 (Sep 6). Doi: 10.1111/jdv.18571
Brepocitinib shows potential against mild-to-moderate atopic dermatitis in phase 2 trial
Key clinical point: Topical brepocitinib cream showed significant efficacy in reducing disease severity and was well-tolerated in patients with mild-to-moderate atopic dermatitis (AD).
Major finding: The reduction in the mean Eczema Area and Severity Index at week 6 was significantly higher with 1% brepocitinib cream once daily (QD) vs vehicle QD (−70.1% vs −44.4%) and 1% brepocitinib cream twice daily (BID) vs vehicle BID (−75.0% vs −47.6%; both P < .05). No serious adverse events or deaths were reported.
Study details: Findings are from a double-blind, dose-ranging, phase 2 study including 292 patients with mild-to-moderate AD who were randomly assigned to receive brepocitinib (0.1% QD, 0.3% QD or BID, 1.0% QD or BID, or 3.0% QD) or vehicle (QD or BID).
Disclosures: This study was sponsored by Pfizer Inc. Nine authors declared being shareholders and current or former employees of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Landis MN et al. Efficacy and safety of topical brepocitinib for the treatment of mild-to-moderate atopic dermatitis: A phase IIb, randomised, double-blind, vehicle-controlled, dose-ranging, and parallel-group study. Br J Dermatol. 2022 (Aug 20). Doi: 10.1111/bjd.21826
Key clinical point: Topical brepocitinib cream showed significant efficacy in reducing disease severity and was well-tolerated in patients with mild-to-moderate atopic dermatitis (AD).
Major finding: The reduction in the mean Eczema Area and Severity Index at week 6 was significantly higher with 1% brepocitinib cream once daily (QD) vs vehicle QD (−70.1% vs −44.4%) and 1% brepocitinib cream twice daily (BID) vs vehicle BID (−75.0% vs −47.6%; both P < .05). No serious adverse events or deaths were reported.
Study details: Findings are from a double-blind, dose-ranging, phase 2 study including 292 patients with mild-to-moderate AD who were randomly assigned to receive brepocitinib (0.1% QD, 0.3% QD or BID, 1.0% QD or BID, or 3.0% QD) or vehicle (QD or BID).
Disclosures: This study was sponsored by Pfizer Inc. Nine authors declared being shareholders and current or former employees of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Landis MN et al. Efficacy and safety of topical brepocitinib for the treatment of mild-to-moderate atopic dermatitis: A phase IIb, randomised, double-blind, vehicle-controlled, dose-ranging, and parallel-group study. Br J Dermatol. 2022 (Aug 20). Doi: 10.1111/bjd.21826
Key clinical point: Topical brepocitinib cream showed significant efficacy in reducing disease severity and was well-tolerated in patients with mild-to-moderate atopic dermatitis (AD).
Major finding: The reduction in the mean Eczema Area and Severity Index at week 6 was significantly higher with 1% brepocitinib cream once daily (QD) vs vehicle QD (−70.1% vs −44.4%) and 1% brepocitinib cream twice daily (BID) vs vehicle BID (−75.0% vs −47.6%; both P < .05). No serious adverse events or deaths were reported.
Study details: Findings are from a double-blind, dose-ranging, phase 2 study including 292 patients with mild-to-moderate AD who were randomly assigned to receive brepocitinib (0.1% QD, 0.3% QD or BID, 1.0% QD or BID, or 3.0% QD) or vehicle (QD or BID).
Disclosures: This study was sponsored by Pfizer Inc. Nine authors declared being shareholders and current or former employees of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Landis MN et al. Efficacy and safety of topical brepocitinib for the treatment of mild-to-moderate atopic dermatitis: A phase IIb, randomised, double-blind, vehicle-controlled, dose-ranging, and parallel-group study. Br J Dermatol. 2022 (Aug 20). Doi: 10.1111/bjd.21826
Meta-analysis finds no increased VTE risk in AD patients receiving JAK inhibitors
Key clinical point: The results of this meta-analysis do not demonstrate an elevated risk for incident venous thromboembolism (VTE) in patients with atopic dermatitis (AD), particularly among those receiving treatment with Janus kinase (JAK) inhibitors.
Major finding: The risk for incident VTE was similar among participants with vs without AD (pooled hazard ratio 0.95; 95% CI 0.62-1.45). Among patients with AD who received JAK inhibitors vs placebo /dupilumab, 0.05% vs 0.03% reported VTE (Mantel-Haenszel risk difference 0; 95% CI 0-0).
Study details: Findings are from a meta-analysis of two cohort studies including 458,206 participants with (n = 229,103) and without AD (n = 229,103) and 15 randomized controlled trials including 8787 patients with AD who received an interventional treatment with JAK inhibitors or a control treatment with dupilumab or placebo.
Disclosures: This study did not report any source of funding. The authors declared no conflicts of interest.
Source: Chen TL et al. Association of risk of incident venous thromboembolism with atopic dermatitis and treatment with Janus kinase inhibitors: A systematic review and meta-analysis. JAMA Dermatol. 2022;e223516 (Aug 24). Doi: 10.1001/jamadermatol.2022.3516
Key clinical point: The results of this meta-analysis do not demonstrate an elevated risk for incident venous thromboembolism (VTE) in patients with atopic dermatitis (AD), particularly among those receiving treatment with Janus kinase (JAK) inhibitors.
Major finding: The risk for incident VTE was similar among participants with vs without AD (pooled hazard ratio 0.95; 95% CI 0.62-1.45). Among patients with AD who received JAK inhibitors vs placebo /dupilumab, 0.05% vs 0.03% reported VTE (Mantel-Haenszel risk difference 0; 95% CI 0-0).
Study details: Findings are from a meta-analysis of two cohort studies including 458,206 participants with (n = 229,103) and without AD (n = 229,103) and 15 randomized controlled trials including 8787 patients with AD who received an interventional treatment with JAK inhibitors or a control treatment with dupilumab or placebo.
Disclosures: This study did not report any source of funding. The authors declared no conflicts of interest.
Source: Chen TL et al. Association of risk of incident venous thromboembolism with atopic dermatitis and treatment with Janus kinase inhibitors: A systematic review and meta-analysis. JAMA Dermatol. 2022;e223516 (Aug 24). Doi: 10.1001/jamadermatol.2022.3516
Key clinical point: The results of this meta-analysis do not demonstrate an elevated risk for incident venous thromboembolism (VTE) in patients with atopic dermatitis (AD), particularly among those receiving treatment with Janus kinase (JAK) inhibitors.
Major finding: The risk for incident VTE was similar among participants with vs without AD (pooled hazard ratio 0.95; 95% CI 0.62-1.45). Among patients with AD who received JAK inhibitors vs placebo /dupilumab, 0.05% vs 0.03% reported VTE (Mantel-Haenszel risk difference 0; 95% CI 0-0).
Study details: Findings are from a meta-analysis of two cohort studies including 458,206 participants with (n = 229,103) and without AD (n = 229,103) and 15 randomized controlled trials including 8787 patients with AD who received an interventional treatment with JAK inhibitors or a control treatment with dupilumab or placebo.
Disclosures: This study did not report any source of funding. The authors declared no conflicts of interest.
Source: Chen TL et al. Association of risk of incident venous thromboembolism with atopic dermatitis and treatment with Janus kinase inhibitors: A systematic review and meta-analysis. JAMA Dermatol. 2022;e223516 (Aug 24). Doi: 10.1001/jamadermatol.2022.3516
Dupilumab shows good drug survival in moderate-to-severe atopic dermatitis
Key clinical point: Dupilumab demonstrated good overall drug survival for up to 3 years in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: Dupilumab showed good overall drug survival rates at 1-year (90.3%), 2-year (85.9%), and 3-year (78.6%). The use of immunosuppressant drugs at baseline was associated with shorter drug survival owing to ineffectiveness (hazard ratio [HR] 2.64; 95% CI 1.10-6.37) and adverse events (HR 2.69; 95% CI 1.32-5.48).
Study details: Findings are from an analysis of the BioDay registry data of 715 adult patients with moderate-to-severe AD who received dupilumab and were followed-up for ≥4 weeks.
Disclosures: The BioDay registry was sponsored by Sanofi Genzyme. The authors declared receiving grants, personal fees, speaking fees, financial support or nonfinancial support from several sources.
Source: Spekhorst LS et al. Dupilumab drug survival and associated predictors in patients with moderate to severe atopic dermatitis: Long-term results from the daily practice BioDay registry. JAMA Dermatol. 2022;e223014 (Aug 10). Doi: 10.1001/jamadermatol.2022.3014
Key clinical point: Dupilumab demonstrated good overall drug survival for up to 3 years in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: Dupilumab showed good overall drug survival rates at 1-year (90.3%), 2-year (85.9%), and 3-year (78.6%). The use of immunosuppressant drugs at baseline was associated with shorter drug survival owing to ineffectiveness (hazard ratio [HR] 2.64; 95% CI 1.10-6.37) and adverse events (HR 2.69; 95% CI 1.32-5.48).
Study details: Findings are from an analysis of the BioDay registry data of 715 adult patients with moderate-to-severe AD who received dupilumab and were followed-up for ≥4 weeks.
Disclosures: The BioDay registry was sponsored by Sanofi Genzyme. The authors declared receiving grants, personal fees, speaking fees, financial support or nonfinancial support from several sources.
Source: Spekhorst LS et al. Dupilumab drug survival and associated predictors in patients with moderate to severe atopic dermatitis: Long-term results from the daily practice BioDay registry. JAMA Dermatol. 2022;e223014 (Aug 10). Doi: 10.1001/jamadermatol.2022.3014
Key clinical point: Dupilumab demonstrated good overall drug survival for up to 3 years in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: Dupilumab showed good overall drug survival rates at 1-year (90.3%), 2-year (85.9%), and 3-year (78.6%). The use of immunosuppressant drugs at baseline was associated with shorter drug survival owing to ineffectiveness (hazard ratio [HR] 2.64; 95% CI 1.10-6.37) and adverse events (HR 2.69; 95% CI 1.32-5.48).
Study details: Findings are from an analysis of the BioDay registry data of 715 adult patients with moderate-to-severe AD who received dupilumab and were followed-up for ≥4 weeks.
Disclosures: The BioDay registry was sponsored by Sanofi Genzyme. The authors declared receiving grants, personal fees, speaking fees, financial support or nonfinancial support from several sources.
Source: Spekhorst LS et al. Dupilumab drug survival and associated predictors in patients with moderate to severe atopic dermatitis: Long-term results from the daily practice BioDay registry. JAMA Dermatol. 2022;e223014 (Aug 10). Doi: 10.1001/jamadermatol.2022.3014
Open Clinical Trials for Psoriasis
The psoriasis clinical trials listed below are all phase 3 and recruiting participants as of July 19, 2022. For additional information on the study design, eligibility criteria, and contacts/locations, visit ClinicalTrials.gov.
GENERALIZED PUSTULAR PSORIASIS
Long-Term Safety and Efficacy of Imsidolimab (ANB019) in Subjects With Generalized Pustular Psoriasis (GEMINI2)
ClinicalTrials.gov Identifier: NCT05366855
An Expanded Access Trial in Japan to Provide Spesolimab to People With a Flare-up in Generalized Pustular Psoriasis Who Have no Other Treatment Options
ClinicalTrials.gov Identifier: NCT05200247
An Expanded Access Program in China to Provide Spesolimab to People With a Flare-up in Generalized Pustular Psoriasis Who Have No Other Treatment Options
ClinicalTrials.gov Identifier: NCT05239039
Study to Evaluate the Efficacy and Safety of Imsidolimab (ANB019) in the Treatment of Subjects With GPP (GEMINI1)
ClinicalTrials.gov Identifier: NCT05352893
NAIL PSORIASIS
Efficacy and Safety Study of Tildrakizumab in the Treatment of Nail Psoriasis
ClinicalTrials.gov Identifier: NCT03897075
PALMOPLANTAR PUSTULOSIS
Phase 3, Randomized Study of Apremilast in Japanese Participants With Palmoplantar Pustulosis (PPP)
ClinicalTrials.gov Identifier: NCT05174065
PLAQUE PSORIASIS
A Long-term Extension Study of Apremilast (CC-10004) in Pediatric Subjects From 6 Through 17 Years of Age With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04175613
A Phase III Efficacy and Safety Study of Hemay005 in Subjects With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04839328
A Study of Subcutaneous Risankizumab Injection for Pediatric Participants With Moderate to Severe Plaque Psoriasis to Assess Change in Disease Symptoms
ClinicalTrials.gov Identifier: NCT04435600
A Study to Evaluate the Drug Levels, Efficacy and Safety of Deucravacitinib in Adolescent Participants With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04772079
Investigator Initiated Trial to Study Duobrii® Lotion in the Treatment of Mild Plaque Psoriasis in Adults
ClinicalTrials.gov Identifier: NCT05203315
Comparative Study of BAT2206 With Stelara® in Patients With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04728360
A Study to Evaluate the Efficacy and Safety of Bimekizumab in Adult Korean Study Participants With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT05020249
Comparing Efficacy and Safety of Bmab 1200 and Stelara in Patients With Moderate to Severe Chronic Plaque Psoriasis (STELLAR-2)
ClinicalTrials.gov Identifier: NCT05335356
A Study to Evaluate the Efficacy, Safety, and Drug Concentration of Certolizumab Pegol (CZP) in Children and Adolescent Study Participants With Moderate to Severe Chronic Plaque Psoriasis (PSO)(CIMcare)
ClinicalTrials.gov Identifier: NCT04123795
A Study of Tildrakizumab in Pediatric Subjects With Chronic Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT03997786
Tapinarof for the Treatment of Plaque Psoriasis in Pediatric Subjects
ClinicalTrials.gov Identifier: NCT05172726
A Study to Evaluate the Efficacy, Safety, and Pharmacokinetics of Subcutaneously Administered Guselkumab for the Treatment of Chronic Plaque Psoriasis in Pediatric Participants (PROTOSTAR)
ClinicalTrials.gov Identifier: NCT03451851
PSORIATIC ARTHRITIS
Efficacy of Secukinumab Compared to Ustekinumab in Adults With Active Psoriatic Arthritis and Failure of TNFα-Inhibitor Treatment (AgAIN)
ClinicalTrials.gov Identifier: NCT04632927
A Long-term Extension Study of Ustekinumab in Pediatric Participants (UNITED)
ClinicalTrials.gov Identifier: NCT05092269
A Study of Ustekinumab or Guselkumab in Pediatric Participants With Active Juvenile Psoriatic Arthritis (PSUMMIT-Jr)
ClinicalTrials.gov Identifier: NCT05083182
Comparative Study of BAT2506 With Simponi® in Participants With Active Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT05046431
Long Term Evaluation of Safety and Efficacy of Tildrakizumab in Patients With Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04991116
To Evaluate the Efficacy and Safety of SHR0302 Tablet in Subjects of Active Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04957550
PSORIATIC ARTHRITIS (continued)
Guselkumab in Active Psoriatic Arthritis Participants With Inadequate Response/Intolerance to One Prior Anti-TNF Alpha Agent (SOLSTICE)
ClinicalTrials.gov Identifier: NCT04936308
A Study to Determine the Efficacy and Safety of Deucravacitinib Compared With Placebo in Participants With Active Psoriatic Arthritis (PsA) Who Are Naïve to Biologic Disease-modifying Anti-rheumatic Drugs
ClinicalTrials.gov Identifier: NCT04908202
A Study to Determine the Efficacy and Safety of Deucravacitinib Compared With Placebo in Participants With Active Psoriatic Arthritis (PsA) Who Are Naïve to Biologic Disease Modifying Anti-rheumatic Drugs or Had Previously Received TNFα Inhibitor Treatment
ClinicalTrials.gov Identifier: NCT04908189
A Study of Guselkumab in Participants With Active Psoriatic Arthritis (APEX) ClinicalTrials.gov Identifier: NCT04882098 Apremilast Pediatric Study in Children With Active Juvenile Psoriatic Arthritis (PEAPOD)
ClinicalTrials.gov Identifier: NCT04804553
Impact of Tapering Immunosuppressants on Maintaining Minimal Disease Activity in Adult Subjects With Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04610476
A Study of Ixekizumab (LY2439821) in Children With Juvenile Idiopathic Arthritis Categories of Enthesitis-related Arthritis (Including Juvenile Onset Ankylosing Spondylitis) and Juvenile Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04527380
Efficacy and Safety of Tildrakizumab Compared to Placebo in Subjects With Active Psoriatic Arthritis I (INSPIRE 1)
ClinicalTrials.gov Identifier: NCT04314544
Efficacy and Safety of Tildrakizumab Compared to Placebo in Anti- TNF naïve Subjects With Active Psoriatic Arthritis II (INSPIRE 2)
ClinicalTrials.gov Identifier: NCT04314531
The psoriasis clinical trials listed below are all phase 3 and recruiting participants as of July 19, 2022. For additional information on the study design, eligibility criteria, and contacts/locations, visit ClinicalTrials.gov.
GENERALIZED PUSTULAR PSORIASIS
Long-Term Safety and Efficacy of Imsidolimab (ANB019) in Subjects With Generalized Pustular Psoriasis (GEMINI2)
ClinicalTrials.gov Identifier: NCT05366855
An Expanded Access Trial in Japan to Provide Spesolimab to People With a Flare-up in Generalized Pustular Psoriasis Who Have no Other Treatment Options
ClinicalTrials.gov Identifier: NCT05200247
An Expanded Access Program in China to Provide Spesolimab to People With a Flare-up in Generalized Pustular Psoriasis Who Have No Other Treatment Options
ClinicalTrials.gov Identifier: NCT05239039
Study to Evaluate the Efficacy and Safety of Imsidolimab (ANB019) in the Treatment of Subjects With GPP (GEMINI1)
ClinicalTrials.gov Identifier: NCT05352893
NAIL PSORIASIS
Efficacy and Safety Study of Tildrakizumab in the Treatment of Nail Psoriasis
ClinicalTrials.gov Identifier: NCT03897075
PALMOPLANTAR PUSTULOSIS
Phase 3, Randomized Study of Apremilast in Japanese Participants With Palmoplantar Pustulosis (PPP)
ClinicalTrials.gov Identifier: NCT05174065
PLAQUE PSORIASIS
A Long-term Extension Study of Apremilast (CC-10004) in Pediatric Subjects From 6 Through 17 Years of Age With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04175613
A Phase III Efficacy and Safety Study of Hemay005 in Subjects With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04839328
A Study of Subcutaneous Risankizumab Injection for Pediatric Participants With Moderate to Severe Plaque Psoriasis to Assess Change in Disease Symptoms
ClinicalTrials.gov Identifier: NCT04435600
A Study to Evaluate the Drug Levels, Efficacy and Safety of Deucravacitinib in Adolescent Participants With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04772079
Investigator Initiated Trial to Study Duobrii® Lotion in the Treatment of Mild Plaque Psoriasis in Adults
ClinicalTrials.gov Identifier: NCT05203315
Comparative Study of BAT2206 With Stelara® in Patients With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04728360
A Study to Evaluate the Efficacy and Safety of Bimekizumab in Adult Korean Study Participants With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT05020249
Comparing Efficacy and Safety of Bmab 1200 and Stelara in Patients With Moderate to Severe Chronic Plaque Psoriasis (STELLAR-2)
ClinicalTrials.gov Identifier: NCT05335356
A Study to Evaluate the Efficacy, Safety, and Drug Concentration of Certolizumab Pegol (CZP) in Children and Adolescent Study Participants With Moderate to Severe Chronic Plaque Psoriasis (PSO)(CIMcare)
ClinicalTrials.gov Identifier: NCT04123795
A Study of Tildrakizumab in Pediatric Subjects With Chronic Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT03997786
Tapinarof for the Treatment of Plaque Psoriasis in Pediatric Subjects
ClinicalTrials.gov Identifier: NCT05172726
A Study to Evaluate the Efficacy, Safety, and Pharmacokinetics of Subcutaneously Administered Guselkumab for the Treatment of Chronic Plaque Psoriasis in Pediatric Participants (PROTOSTAR)
ClinicalTrials.gov Identifier: NCT03451851
PSORIATIC ARTHRITIS
Efficacy of Secukinumab Compared to Ustekinumab in Adults With Active Psoriatic Arthritis and Failure of TNFα-Inhibitor Treatment (AgAIN)
ClinicalTrials.gov Identifier: NCT04632927
A Long-term Extension Study of Ustekinumab in Pediatric Participants (UNITED)
ClinicalTrials.gov Identifier: NCT05092269
A Study of Ustekinumab or Guselkumab in Pediatric Participants With Active Juvenile Psoriatic Arthritis (PSUMMIT-Jr)
ClinicalTrials.gov Identifier: NCT05083182
Comparative Study of BAT2506 With Simponi® in Participants With Active Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT05046431
Long Term Evaluation of Safety and Efficacy of Tildrakizumab in Patients With Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04991116
To Evaluate the Efficacy and Safety of SHR0302 Tablet in Subjects of Active Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04957550
PSORIATIC ARTHRITIS (continued)
Guselkumab in Active Psoriatic Arthritis Participants With Inadequate Response/Intolerance to One Prior Anti-TNF Alpha Agent (SOLSTICE)
ClinicalTrials.gov Identifier: NCT04936308
A Study to Determine the Efficacy and Safety of Deucravacitinib Compared With Placebo in Participants With Active Psoriatic Arthritis (PsA) Who Are Naïve to Biologic Disease-modifying Anti-rheumatic Drugs
ClinicalTrials.gov Identifier: NCT04908202
A Study to Determine the Efficacy and Safety of Deucravacitinib Compared With Placebo in Participants With Active Psoriatic Arthritis (PsA) Who Are Naïve to Biologic Disease Modifying Anti-rheumatic Drugs or Had Previously Received TNFα Inhibitor Treatment
ClinicalTrials.gov Identifier: NCT04908189
A Study of Guselkumab in Participants With Active Psoriatic Arthritis (APEX) ClinicalTrials.gov Identifier: NCT04882098 Apremilast Pediatric Study in Children With Active Juvenile Psoriatic Arthritis (PEAPOD)
ClinicalTrials.gov Identifier: NCT04804553
Impact of Tapering Immunosuppressants on Maintaining Minimal Disease Activity in Adult Subjects With Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04610476
A Study of Ixekizumab (LY2439821) in Children With Juvenile Idiopathic Arthritis Categories of Enthesitis-related Arthritis (Including Juvenile Onset Ankylosing Spondylitis) and Juvenile Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04527380
Efficacy and Safety of Tildrakizumab Compared to Placebo in Subjects With Active Psoriatic Arthritis I (INSPIRE 1)
ClinicalTrials.gov Identifier: NCT04314544
Efficacy and Safety of Tildrakizumab Compared to Placebo in Anti- TNF naïve Subjects With Active Psoriatic Arthritis II (INSPIRE 2)
ClinicalTrials.gov Identifier: NCT04314531
The psoriasis clinical trials listed below are all phase 3 and recruiting participants as of July 19, 2022. For additional information on the study design, eligibility criteria, and contacts/locations, visit ClinicalTrials.gov.
GENERALIZED PUSTULAR PSORIASIS
Long-Term Safety and Efficacy of Imsidolimab (ANB019) in Subjects With Generalized Pustular Psoriasis (GEMINI2)
ClinicalTrials.gov Identifier: NCT05366855
An Expanded Access Trial in Japan to Provide Spesolimab to People With a Flare-up in Generalized Pustular Psoriasis Who Have no Other Treatment Options
ClinicalTrials.gov Identifier: NCT05200247
An Expanded Access Program in China to Provide Spesolimab to People With a Flare-up in Generalized Pustular Psoriasis Who Have No Other Treatment Options
ClinicalTrials.gov Identifier: NCT05239039
Study to Evaluate the Efficacy and Safety of Imsidolimab (ANB019) in the Treatment of Subjects With GPP (GEMINI1)
ClinicalTrials.gov Identifier: NCT05352893
NAIL PSORIASIS
Efficacy and Safety Study of Tildrakizumab in the Treatment of Nail Psoriasis
ClinicalTrials.gov Identifier: NCT03897075
PALMOPLANTAR PUSTULOSIS
Phase 3, Randomized Study of Apremilast in Japanese Participants With Palmoplantar Pustulosis (PPP)
ClinicalTrials.gov Identifier: NCT05174065
PLAQUE PSORIASIS
A Long-term Extension Study of Apremilast (CC-10004) in Pediatric Subjects From 6 Through 17 Years of Age With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04175613
A Phase III Efficacy and Safety Study of Hemay005 in Subjects With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04839328
A Study of Subcutaneous Risankizumab Injection for Pediatric Participants With Moderate to Severe Plaque Psoriasis to Assess Change in Disease Symptoms
ClinicalTrials.gov Identifier: NCT04435600
A Study to Evaluate the Drug Levels, Efficacy and Safety of Deucravacitinib in Adolescent Participants With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04772079
Investigator Initiated Trial to Study Duobrii® Lotion in the Treatment of Mild Plaque Psoriasis in Adults
ClinicalTrials.gov Identifier: NCT05203315
Comparative Study of BAT2206 With Stelara® in Patients With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT04728360
A Study to Evaluate the Efficacy and Safety of Bimekizumab in Adult Korean Study Participants With Moderate to Severe Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT05020249
Comparing Efficacy and Safety of Bmab 1200 and Stelara in Patients With Moderate to Severe Chronic Plaque Psoriasis (STELLAR-2)
ClinicalTrials.gov Identifier: NCT05335356
A Study to Evaluate the Efficacy, Safety, and Drug Concentration of Certolizumab Pegol (CZP) in Children and Adolescent Study Participants With Moderate to Severe Chronic Plaque Psoriasis (PSO)(CIMcare)
ClinicalTrials.gov Identifier: NCT04123795
A Study of Tildrakizumab in Pediatric Subjects With Chronic Plaque Psoriasis
ClinicalTrials.gov Identifier: NCT03997786
Tapinarof for the Treatment of Plaque Psoriasis in Pediatric Subjects
ClinicalTrials.gov Identifier: NCT05172726
A Study to Evaluate the Efficacy, Safety, and Pharmacokinetics of Subcutaneously Administered Guselkumab for the Treatment of Chronic Plaque Psoriasis in Pediatric Participants (PROTOSTAR)
ClinicalTrials.gov Identifier: NCT03451851
PSORIATIC ARTHRITIS
Efficacy of Secukinumab Compared to Ustekinumab in Adults With Active Psoriatic Arthritis and Failure of TNFα-Inhibitor Treatment (AgAIN)
ClinicalTrials.gov Identifier: NCT04632927
A Long-term Extension Study of Ustekinumab in Pediatric Participants (UNITED)
ClinicalTrials.gov Identifier: NCT05092269
A Study of Ustekinumab or Guselkumab in Pediatric Participants With Active Juvenile Psoriatic Arthritis (PSUMMIT-Jr)
ClinicalTrials.gov Identifier: NCT05083182
Comparative Study of BAT2506 With Simponi® in Participants With Active Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT05046431
Long Term Evaluation of Safety and Efficacy of Tildrakizumab in Patients With Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04991116
To Evaluate the Efficacy and Safety of SHR0302 Tablet in Subjects of Active Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04957550
PSORIATIC ARTHRITIS (continued)
Guselkumab in Active Psoriatic Arthritis Participants With Inadequate Response/Intolerance to One Prior Anti-TNF Alpha Agent (SOLSTICE)
ClinicalTrials.gov Identifier: NCT04936308
A Study to Determine the Efficacy and Safety of Deucravacitinib Compared With Placebo in Participants With Active Psoriatic Arthritis (PsA) Who Are Naïve to Biologic Disease-modifying Anti-rheumatic Drugs
ClinicalTrials.gov Identifier: NCT04908202
A Study to Determine the Efficacy and Safety of Deucravacitinib Compared With Placebo in Participants With Active Psoriatic Arthritis (PsA) Who Are Naïve to Biologic Disease Modifying Anti-rheumatic Drugs or Had Previously Received TNFα Inhibitor Treatment
ClinicalTrials.gov Identifier: NCT04908189
A Study of Guselkumab in Participants With Active Psoriatic Arthritis (APEX) ClinicalTrials.gov Identifier: NCT04882098 Apremilast Pediatric Study in Children With Active Juvenile Psoriatic Arthritis (PEAPOD)
ClinicalTrials.gov Identifier: NCT04804553
Impact of Tapering Immunosuppressants on Maintaining Minimal Disease Activity in Adult Subjects With Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04610476
A Study of Ixekizumab (LY2439821) in Children With Juvenile Idiopathic Arthritis Categories of Enthesitis-related Arthritis (Including Juvenile Onset Ankylosing Spondylitis) and Juvenile Psoriatic Arthritis
ClinicalTrials.gov Identifier: NCT04527380
Efficacy and Safety of Tildrakizumab Compared to Placebo in Subjects With Active Psoriatic Arthritis I (INSPIRE 1)
ClinicalTrials.gov Identifier: NCT04314544
Efficacy and Safety of Tildrakizumab Compared to Placebo in Anti- TNF naïve Subjects With Active Psoriatic Arthritis II (INSPIRE 2)
ClinicalTrials.gov Identifier: NCT04314531
Paresthesias along forearm
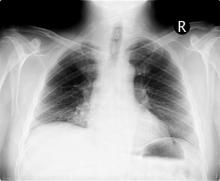
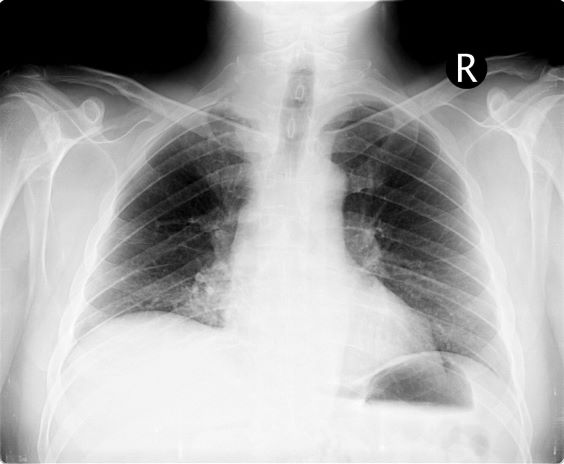
On the basis of this presentation, and the findings from the chest x-ray (as shown), the likely diagnosis is non–small cell lung cancer (NSCLC), Pancoast tumor, also known as superior sulcus tumor. Pancoast tumors are rare, representing about 3%-5% of all lung cancers, and invade the structures in the apex of the chest, including the first thoracic ribs or periosteum, the lower nerve roots of the bronchial plexus, the sympathetic chain and stellate ganglion, or the subclavian vessels. The majority of Pancoast tumors are non–small cell carcinomas.
Because of their pulmonary location, Pancoast tumors are characterized by several distinct symptoms. As seen in this case, patients often present with shoulder pain that worsens over time, especially with invasion of the chest wall and brachial plexus. The pain may radiate to the neck; axilla; anterior chest wall; and medial aspect of the arm, forearm, and wrist. If Pancoast tumors infiltrate the ulnar nerve, patients may present with weakness and muscle atrophy of the intrinsic muscles of the hand. In addition, invasion of the sympathetic chain and of the inferior cervical ganglion can cause Horner syndrome (ptosis, miosis, enophthalmos, and anhidrosis). Lastly, upper-arm edema may develop, signaling invasion and potentially occlusion of the subclavian vein.
During workup, CT-guided core biopsy is the first-line diagnostic test for Pancoast tumors. CT of the chest can confirm the presence of an apical mass and its position in relation to other structures of the thoracic inlet. MRI can further assess suspected brachial plexus, subclavian vessels, spine, and neural foramina invasion, specifying the extent of the disease and of the amount of nerve-root involvement.
For resectable Pancoast tumors, the National Comprehensive Cancer Network recommends chemoradiation, followed by surgical resection and chemotherapy. Preoperative chemoradiation together with surgical resection has shown a 2-year survival between 50% and 70%. Depending on biomarker status (certain EGFR mutations or programmed death ligand 1 levels ≥ 1%), the addition of either atezolizumab or osimertinib is advised. However, the positioning of Pancoast tumors can pose a surgical challenge, and if the lesion remains unresectable after preoperative concurrent chemoradiation, then consolidation immunotherapy with durvalumab is recommended.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
On the basis of this presentation, and the findings from the chest x-ray (as shown), the likely diagnosis is non–small cell lung cancer (NSCLC), Pancoast tumor, also known as superior sulcus tumor. Pancoast tumors are rare, representing about 3%-5% of all lung cancers, and invade the structures in the apex of the chest, including the first thoracic ribs or periosteum, the lower nerve roots of the bronchial plexus, the sympathetic chain and stellate ganglion, or the subclavian vessels. The majority of Pancoast tumors are non–small cell carcinomas.
Because of their pulmonary location, Pancoast tumors are characterized by several distinct symptoms. As seen in this case, patients often present with shoulder pain that worsens over time, especially with invasion of the chest wall and brachial plexus. The pain may radiate to the neck; axilla; anterior chest wall; and medial aspect of the arm, forearm, and wrist. If Pancoast tumors infiltrate the ulnar nerve, patients may present with weakness and muscle atrophy of the intrinsic muscles of the hand. In addition, invasion of the sympathetic chain and of the inferior cervical ganglion can cause Horner syndrome (ptosis, miosis, enophthalmos, and anhidrosis). Lastly, upper-arm edema may develop, signaling invasion and potentially occlusion of the subclavian vein.
During workup, CT-guided core biopsy is the first-line diagnostic test for Pancoast tumors. CT of the chest can confirm the presence of an apical mass and its position in relation to other structures of the thoracic inlet. MRI can further assess suspected brachial plexus, subclavian vessels, spine, and neural foramina invasion, specifying the extent of the disease and of the amount of nerve-root involvement.
For resectable Pancoast tumors, the National Comprehensive Cancer Network recommends chemoradiation, followed by surgical resection and chemotherapy. Preoperative chemoradiation together with surgical resection has shown a 2-year survival between 50% and 70%. Depending on biomarker status (certain EGFR mutations or programmed death ligand 1 levels ≥ 1%), the addition of either atezolizumab or osimertinib is advised. However, the positioning of Pancoast tumors can pose a surgical challenge, and if the lesion remains unresectable after preoperative concurrent chemoradiation, then consolidation immunotherapy with durvalumab is recommended.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
On the basis of this presentation, and the findings from the chest x-ray (as shown), the likely diagnosis is non–small cell lung cancer (NSCLC), Pancoast tumor, also known as superior sulcus tumor. Pancoast tumors are rare, representing about 3%-5% of all lung cancers, and invade the structures in the apex of the chest, including the first thoracic ribs or periosteum, the lower nerve roots of the bronchial plexus, the sympathetic chain and stellate ganglion, or the subclavian vessels. The majority of Pancoast tumors are non–small cell carcinomas.
Because of their pulmonary location, Pancoast tumors are characterized by several distinct symptoms. As seen in this case, patients often present with shoulder pain that worsens over time, especially with invasion of the chest wall and brachial plexus. The pain may radiate to the neck; axilla; anterior chest wall; and medial aspect of the arm, forearm, and wrist. If Pancoast tumors infiltrate the ulnar nerve, patients may present with weakness and muscle atrophy of the intrinsic muscles of the hand. In addition, invasion of the sympathetic chain and of the inferior cervical ganglion can cause Horner syndrome (ptosis, miosis, enophthalmos, and anhidrosis). Lastly, upper-arm edema may develop, signaling invasion and potentially occlusion of the subclavian vein.
During workup, CT-guided core biopsy is the first-line diagnostic test for Pancoast tumors. CT of the chest can confirm the presence of an apical mass and its position in relation to other structures of the thoracic inlet. MRI can further assess suspected brachial plexus, subclavian vessels, spine, and neural foramina invasion, specifying the extent of the disease and of the amount of nerve-root involvement.
For resectable Pancoast tumors, the National Comprehensive Cancer Network recommends chemoradiation, followed by surgical resection and chemotherapy. Preoperative chemoradiation together with surgical resection has shown a 2-year survival between 50% and 70%. Depending on biomarker status (certain EGFR mutations or programmed death ligand 1 levels ≥ 1%), the addition of either atezolizumab or osimertinib is advised. However, the positioning of Pancoast tumors can pose a surgical challenge, and if the lesion remains unresectable after preoperative concurrent chemoradiation, then consolidation immunotherapy with durvalumab is recommended.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 54-year-old man presents with shoulder pain and paresthesias along the medial side of the forearm. The patient has a 50–pack-year history of smoking. He reports that the pain began about 6 weeks ago, at which point he scheduled an orthopedic consultation. Physical examination is also notable for facial flushing. Breathing is normal, with no shortness of breath. Chest radiography reveals asymmetry of the apices (right apex is more opaque than the left). Invasion of the ribs is also seen.