User login
Official Newspaper of the American College of Surgeons
HHS issues guidance on ransomware attacks
The U.S. Department of Health & Human Services’ Office of Civil Rights has issued new guidance to help physicians and their practices combat a ransomware attack.
Ransomware – a type of malicious software designed to block access to a computer system until a sum of money is paid – is becoming a bigger problem for U.S. businesses in general. Daily ransomware attacks against all types of computer systems increased 300% in early 2016 to 4,000, from 1,000 daily attacks in 2015, according to the Department of Justice.
The HHS fact sheet offers information on how HIPAA compliance can help protect and recover infected systems; how to detect if systems are infected; and what to do if a system becomes infected, including what is reportable.
There are “measures known to be effective to prevent the introduction of ransomware and to recover from a ransomware attack,” according to HHS.
The U.S. Department of Health & Human Services’ Office of Civil Rights has issued new guidance to help physicians and their practices combat a ransomware attack.
Ransomware – a type of malicious software designed to block access to a computer system until a sum of money is paid – is becoming a bigger problem for U.S. businesses in general. Daily ransomware attacks against all types of computer systems increased 300% in early 2016 to 4,000, from 1,000 daily attacks in 2015, according to the Department of Justice.
The HHS fact sheet offers information on how HIPAA compliance can help protect and recover infected systems; how to detect if systems are infected; and what to do if a system becomes infected, including what is reportable.
There are “measures known to be effective to prevent the introduction of ransomware and to recover from a ransomware attack,” according to HHS.
The U.S. Department of Health & Human Services’ Office of Civil Rights has issued new guidance to help physicians and their practices combat a ransomware attack.
Ransomware – a type of malicious software designed to block access to a computer system until a sum of money is paid – is becoming a bigger problem for U.S. businesses in general. Daily ransomware attacks against all types of computer systems increased 300% in early 2016 to 4,000, from 1,000 daily attacks in 2015, according to the Department of Justice.
The HHS fact sheet offers information on how HIPAA compliance can help protect and recover infected systems; how to detect if systems are infected; and what to do if a system becomes infected, including what is reportable.
There are “measures known to be effective to prevent the introduction of ransomware and to recover from a ransomware attack,” according to HHS.
Medicaid expansion linked to lower uninsured rates
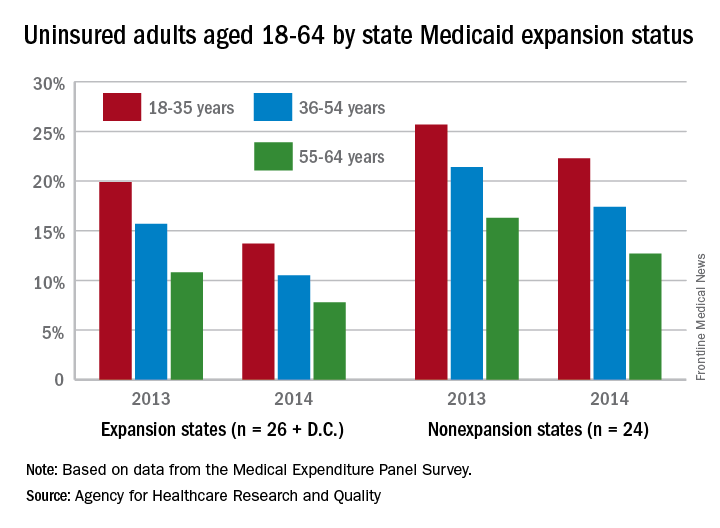
The overall uninsured rate for nonelderly adults took a significant drop from 18.8% in 2013 to 14.4% in 2014, with rates by age lower and declines generally larger among states that expanded Medicaid coverage, according to the Agency for Healthcare Research and Quality.
Among adults aged 18-35 years, states (and the District of Columbia) that expanded Medicaid had a larger drop in the percentage who were uninsured for the entire calendar year, going from 19.9% in 2013 to 13.7% in 2014 (6.2 percentage points), than did states that did not expand Medicaid, which dropped from 25.7% to 22.3% (3.4 percentage points), the AHRQ reported.
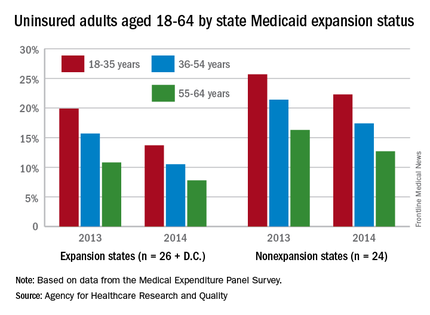
For adults aged 36-54 years, the situation was similar: The uninsured rate in states that expanded Medicaid went from 15.7% in 2013 to 10.5% in 2014, or 5.2 percentage points, while the rate dropped from 21.4% to 17.4%, or 4 percentage points, in states that did not expand Medicaid, the report noted.
Uninsured rates were lower among adults aged 56-64 in states that did expand Medicaid, but the absolute decrease was actually larger among states that did not expand it. The rate in nonexpanding states decreased by 3.6 percentage points, 16.3% to 12.7%, while expanding states saw a drop from 10.8% to 7.8%, 3 percentage points, according to data from the Medical Expenditure Panel Survey.
The overall uninsured rate for nonelderly adults took a significant drop from 18.8% in 2013 to 14.4% in 2014, with rates by age lower and declines generally larger among states that expanded Medicaid coverage, according to the Agency for Healthcare Research and Quality.
Among adults aged 18-35 years, states (and the District of Columbia) that expanded Medicaid had a larger drop in the percentage who were uninsured for the entire calendar year, going from 19.9% in 2013 to 13.7% in 2014 (6.2 percentage points), than did states that did not expand Medicaid, which dropped from 25.7% to 22.3% (3.4 percentage points), the AHRQ reported.

For adults aged 36-54 years, the situation was similar: The uninsured rate in states that expanded Medicaid went from 15.7% in 2013 to 10.5% in 2014, or 5.2 percentage points, while the rate dropped from 21.4% to 17.4%, or 4 percentage points, in states that did not expand Medicaid, the report noted.
Uninsured rates were lower among adults aged 56-64 in states that did expand Medicaid, but the absolute decrease was actually larger among states that did not expand it. The rate in nonexpanding states decreased by 3.6 percentage points, 16.3% to 12.7%, while expanding states saw a drop from 10.8% to 7.8%, 3 percentage points, according to data from the Medical Expenditure Panel Survey.
The overall uninsured rate for nonelderly adults took a significant drop from 18.8% in 2013 to 14.4% in 2014, with rates by age lower and declines generally larger among states that expanded Medicaid coverage, according to the Agency for Healthcare Research and Quality.
Among adults aged 18-35 years, states (and the District of Columbia) that expanded Medicaid had a larger drop in the percentage who were uninsured for the entire calendar year, going from 19.9% in 2013 to 13.7% in 2014 (6.2 percentage points), than did states that did not expand Medicaid, which dropped from 25.7% to 22.3% (3.4 percentage points), the AHRQ reported.

For adults aged 36-54 years, the situation was similar: The uninsured rate in states that expanded Medicaid went from 15.7% in 2013 to 10.5% in 2014, or 5.2 percentage points, while the rate dropped from 21.4% to 17.4%, or 4 percentage points, in states that did not expand Medicaid, the report noted.
Uninsured rates were lower among adults aged 56-64 in states that did expand Medicaid, but the absolute decrease was actually larger among states that did not expand it. The rate in nonexpanding states decreased by 3.6 percentage points, 16.3% to 12.7%, while expanding states saw a drop from 10.8% to 7.8%, 3 percentage points, according to data from the Medical Expenditure Panel Survey.
New antibiotics targeting MDR pathogens are expensive, but not impressive
The U.S. Food and Drug Administration has approved a number of new antibiotics targeting multidrug-resistant bacteria in the past 5 years, but the new drugs have not led to a substantial improvement in patient outcomes when compared with existing antibiotics, according to a recent analysis in the Annals of Internal Medicine.
The eight new antibiotics approved by the FDA between January 2010 and December 2015 were ceftaroline, fidaxomicin, bedaquiline, dalbavancin, tedizolid, oritavancin, ceftolozane/tazobactam, and ceftazidime/avibactam. Of those eight drugs, only three showed in vitro activity against the so-called ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumonia, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species). Only one drug, fidaxomicin, demonstrated in vitro activity against an urgent-threat pathogen from the Centers for Disease Control and Prevention, Clostridium difficile. Bedaquiline was the only new antibiotic specifically indicated for a disease from a multidrug-resistant pathogen, although the investigators said most of the drugs demonstrated in vitro activity against gram-positive drug-resistant pathogens.
Importantly, the authors noted that in vitro activity does not necessarily reflect benefits on actual patient clinical outcomes, as exemplified by such drugs as tigecycline and doripenem.
The researchers found what they called “important deficiencies in the clinical trials leading to approval of these new antibiotic products.” Most pivotal trial designs were primarily noninferiority trials, and the antibiotics were not studied to evaluate whether they have substantial benefits in efficacy over what is currently available, they noted. Additionally, none of the trials evaluated direct patient outcomes as primary end points, and some drugs did not have confirmatory evidence from a second independent trial or did not have any confirmatory trials.
Researchers also examined the prices of a single dose of the new antibiotics. The prices ranged from $1,195 to $4,183 (4-14 days of ceftolozane/tazobactam for acute pyelonephritis and intra-abdominal infections) to $69,702 (24 weeks of bedaquiline) – quite a premium for antibiotics showing unclear evidence of additional benefit.
“As antibiotic innovation continues to move forward, greater attention needs to be paid to incentives for developing high-quality new products with demonstrated superiority to existing products on outcomes in patients with multidrug-resistant disease, replacing the current focus on quantity and presumed future benefits,” researchers concluded.
Read the full study in the Annals of Internal Medicine (doi: 10.7326/M16-0291).
The U.S. Food and Drug Administration has approved a number of new antibiotics targeting multidrug-resistant bacteria in the past 5 years, but the new drugs have not led to a substantial improvement in patient outcomes when compared with existing antibiotics, according to a recent analysis in the Annals of Internal Medicine.
The eight new antibiotics approved by the FDA between January 2010 and December 2015 were ceftaroline, fidaxomicin, bedaquiline, dalbavancin, tedizolid, oritavancin, ceftolozane/tazobactam, and ceftazidime/avibactam. Of those eight drugs, only three showed in vitro activity against the so-called ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumonia, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species). Only one drug, fidaxomicin, demonstrated in vitro activity against an urgent-threat pathogen from the Centers for Disease Control and Prevention, Clostridium difficile. Bedaquiline was the only new antibiotic specifically indicated for a disease from a multidrug-resistant pathogen, although the investigators said most of the drugs demonstrated in vitro activity against gram-positive drug-resistant pathogens.
Importantly, the authors noted that in vitro activity does not necessarily reflect benefits on actual patient clinical outcomes, as exemplified by such drugs as tigecycline and doripenem.
The researchers found what they called “important deficiencies in the clinical trials leading to approval of these new antibiotic products.” Most pivotal trial designs were primarily noninferiority trials, and the antibiotics were not studied to evaluate whether they have substantial benefits in efficacy over what is currently available, they noted. Additionally, none of the trials evaluated direct patient outcomes as primary end points, and some drugs did not have confirmatory evidence from a second independent trial or did not have any confirmatory trials.
Researchers also examined the prices of a single dose of the new antibiotics. The prices ranged from $1,195 to $4,183 (4-14 days of ceftolozane/tazobactam for acute pyelonephritis and intra-abdominal infections) to $69,702 (24 weeks of bedaquiline) – quite a premium for antibiotics showing unclear evidence of additional benefit.
“As antibiotic innovation continues to move forward, greater attention needs to be paid to incentives for developing high-quality new products with demonstrated superiority to existing products on outcomes in patients with multidrug-resistant disease, replacing the current focus on quantity and presumed future benefits,” researchers concluded.
Read the full study in the Annals of Internal Medicine (doi: 10.7326/M16-0291).
The U.S. Food and Drug Administration has approved a number of new antibiotics targeting multidrug-resistant bacteria in the past 5 years, but the new drugs have not led to a substantial improvement in patient outcomes when compared with existing antibiotics, according to a recent analysis in the Annals of Internal Medicine.
The eight new antibiotics approved by the FDA between January 2010 and December 2015 were ceftaroline, fidaxomicin, bedaquiline, dalbavancin, tedizolid, oritavancin, ceftolozane/tazobactam, and ceftazidime/avibactam. Of those eight drugs, only three showed in vitro activity against the so-called ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumonia, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species). Only one drug, fidaxomicin, demonstrated in vitro activity against an urgent-threat pathogen from the Centers for Disease Control and Prevention, Clostridium difficile. Bedaquiline was the only new antibiotic specifically indicated for a disease from a multidrug-resistant pathogen, although the investigators said most of the drugs demonstrated in vitro activity against gram-positive drug-resistant pathogens.
Importantly, the authors noted that in vitro activity does not necessarily reflect benefits on actual patient clinical outcomes, as exemplified by such drugs as tigecycline and doripenem.
The researchers found what they called “important deficiencies in the clinical trials leading to approval of these new antibiotic products.” Most pivotal trial designs were primarily noninferiority trials, and the antibiotics were not studied to evaluate whether they have substantial benefits in efficacy over what is currently available, they noted. Additionally, none of the trials evaluated direct patient outcomes as primary end points, and some drugs did not have confirmatory evidence from a second independent trial or did not have any confirmatory trials.
Researchers also examined the prices of a single dose of the new antibiotics. The prices ranged from $1,195 to $4,183 (4-14 days of ceftolozane/tazobactam for acute pyelonephritis and intra-abdominal infections) to $69,702 (24 weeks of bedaquiline) – quite a premium for antibiotics showing unclear evidence of additional benefit.
“As antibiotic innovation continues to move forward, greater attention needs to be paid to incentives for developing high-quality new products with demonstrated superiority to existing products on outcomes in patients with multidrug-resistant disease, replacing the current focus on quantity and presumed future benefits,” researchers concluded.
Read the full study in the Annals of Internal Medicine (doi: 10.7326/M16-0291).
FROM ANNALS OF INTERNAL MEDICINE
Retiring Baby Boomers leave fewer workers to pay for Medicare
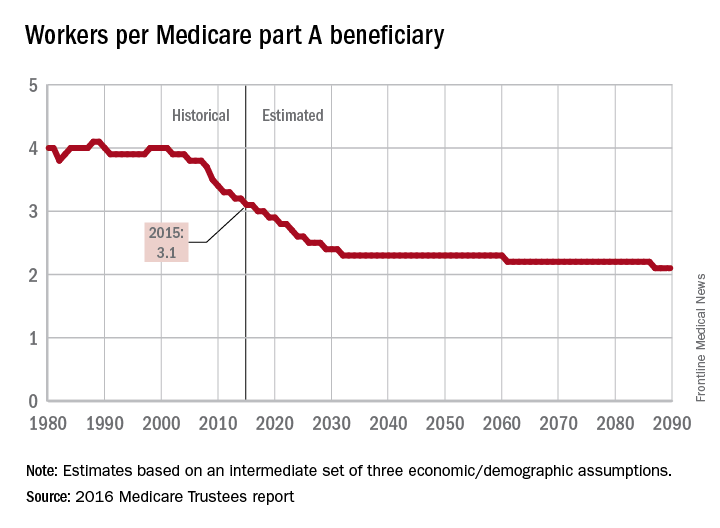
The influx of aging Baby Boomers into the ranks of the retired will reduce the ratio of workers available to pay for each Medicare part A beneficiary by 40% from 2000 to 2030, according to the 2016 report of the Medicare Trustees.
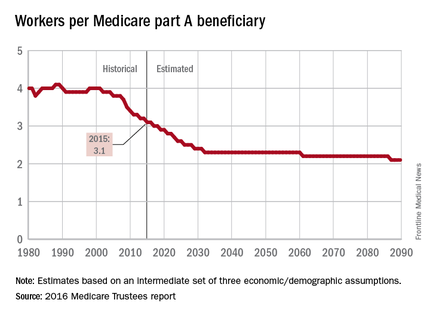
In 2015, there were 3.1 workers for each part A beneficiary, putting the United States in the middle of a projected drop from 4.0 workers per beneficiary in 2000 down to 2.4 in 2030. The Boomer-induced drop will largely be over by then, but the decline will continue until there are about 2.1 workers for each part A beneficiary by 2090, the report said.
“This reduction implies an increase in the [Medicare part A] cost rate of about 50% by 2090, relative to its current level, solely due to this demographic factor,” the trustees noted.
The projections are done using three sets – low-cost, intermediate, and high-cost – of economic and demographic assumptions. The figures presented here are from the intermediate assumption.
The influx of aging Baby Boomers into the ranks of the retired will reduce the ratio of workers available to pay for each Medicare part A beneficiary by 40% from 2000 to 2030, according to the 2016 report of the Medicare Trustees.

In 2015, there were 3.1 workers for each part A beneficiary, putting the United States in the middle of a projected drop from 4.0 workers per beneficiary in 2000 down to 2.4 in 2030. The Boomer-induced drop will largely be over by then, but the decline will continue until there are about 2.1 workers for each part A beneficiary by 2090, the report said.
“This reduction implies an increase in the [Medicare part A] cost rate of about 50% by 2090, relative to its current level, solely due to this demographic factor,” the trustees noted.
The projections are done using three sets – low-cost, intermediate, and high-cost – of economic and demographic assumptions. The figures presented here are from the intermediate assumption.
The influx of aging Baby Boomers into the ranks of the retired will reduce the ratio of workers available to pay for each Medicare part A beneficiary by 40% from 2000 to 2030, according to the 2016 report of the Medicare Trustees.

In 2015, there were 3.1 workers for each part A beneficiary, putting the United States in the middle of a projected drop from 4.0 workers per beneficiary in 2000 down to 2.4 in 2030. The Boomer-induced drop will largely be over by then, but the decline will continue until there are about 2.1 workers for each part A beneficiary by 2090, the report said.
“This reduction implies an increase in the [Medicare part A] cost rate of about 50% by 2090, relative to its current level, solely due to this demographic factor,” the trustees noted.
The projections are done using three sets – low-cost, intermediate, and high-cost – of economic and demographic assumptions. The figures presented here are from the intermediate assumption.
Focused-ultrasound device approved to treat essential tremor
A device that uses transcranial focused ultrasound to destroy the MRI-identified pathologic area of the thalamus in essential tremor patients became the first of its kind to receive Food and Drug Administration approval for use in patients whose condition has been refractory to medications such as beta-blockers or anticonvulsant drugs.
In patients who have undergone evaluation by CT and MRI to determine their suitability for the procedure, the Exablate Neuro device delivers focused ultrasound at incrementally higher energies until patients achieve a reduction of tremor. Patients are awake and responsive during the entire treatment.
“As with other treatments for essential tremor, this new device is not a cure but could help patients enjoy a better quality of life,” said Carlos Peña, PhD, director of the division of neurological and physical medicine devices in the FDA’s Center for Devices and Radiological Health, in the agency’s announcement of the approval. The device “could help them to avoid more extensive surgical treatments,” such as thalamotomy or a deep brain stimulation device, Dr. Peña said.
The clinical data used to support the approval included a double-blind, controlled trial of 76 patients with essential tremor who had not responded to medication therapy.
The 56 patients who were randomly selected to receive the Exablate Neuro treatment showed nearly a 50% improvement in their tremors and motor function 3 months after treatment, compared with their baseline score. The 20 patients who received a sham control treatment had no improvement, and some experienced a slight worsening after the sham procedure before they crossed over into the treatment group at the 3-month time point.
At 12 months post procedure, the treatment group retained a 40% improvement in those scores, compared with baseline.
Adverse events reported by patients treated with the device were similar to those reported by patients who have undergone thalamotomy. Events included numbness/tingling of the fingers, headache, imbalance/unsteadiness, loss of control of body movements (ataxia), or gait disturbance.
Other side effects identified as possibly related to treatment with MR-guided focused ultrasound treatments include tissue damage in an area other than the treatment area, hemorrhage in the treated area requiring emergency treatment, skin burns with ulceration of the skin, skin retraction, and scar formation and blood clots.
InSightec manufacturers the Exablate Neuro device.
A device that uses transcranial focused ultrasound to destroy the MRI-identified pathologic area of the thalamus in essential tremor patients became the first of its kind to receive Food and Drug Administration approval for use in patients whose condition has been refractory to medications such as beta-blockers or anticonvulsant drugs.
In patients who have undergone evaluation by CT and MRI to determine their suitability for the procedure, the Exablate Neuro device delivers focused ultrasound at incrementally higher energies until patients achieve a reduction of tremor. Patients are awake and responsive during the entire treatment.
“As with other treatments for essential tremor, this new device is not a cure but could help patients enjoy a better quality of life,” said Carlos Peña, PhD, director of the division of neurological and physical medicine devices in the FDA’s Center for Devices and Radiological Health, in the agency’s announcement of the approval. The device “could help them to avoid more extensive surgical treatments,” such as thalamotomy or a deep brain stimulation device, Dr. Peña said.
The clinical data used to support the approval included a double-blind, controlled trial of 76 patients with essential tremor who had not responded to medication therapy.
The 56 patients who were randomly selected to receive the Exablate Neuro treatment showed nearly a 50% improvement in their tremors and motor function 3 months after treatment, compared with their baseline score. The 20 patients who received a sham control treatment had no improvement, and some experienced a slight worsening after the sham procedure before they crossed over into the treatment group at the 3-month time point.
At 12 months post procedure, the treatment group retained a 40% improvement in those scores, compared with baseline.
Adverse events reported by patients treated with the device were similar to those reported by patients who have undergone thalamotomy. Events included numbness/tingling of the fingers, headache, imbalance/unsteadiness, loss of control of body movements (ataxia), or gait disturbance.
Other side effects identified as possibly related to treatment with MR-guided focused ultrasound treatments include tissue damage in an area other than the treatment area, hemorrhage in the treated area requiring emergency treatment, skin burns with ulceration of the skin, skin retraction, and scar formation and blood clots.
InSightec manufacturers the Exablate Neuro device.
A device that uses transcranial focused ultrasound to destroy the MRI-identified pathologic area of the thalamus in essential tremor patients became the first of its kind to receive Food and Drug Administration approval for use in patients whose condition has been refractory to medications such as beta-blockers or anticonvulsant drugs.
In patients who have undergone evaluation by CT and MRI to determine their suitability for the procedure, the Exablate Neuro device delivers focused ultrasound at incrementally higher energies until patients achieve a reduction of tremor. Patients are awake and responsive during the entire treatment.
“As with other treatments for essential tremor, this new device is not a cure but could help patients enjoy a better quality of life,” said Carlos Peña, PhD, director of the division of neurological and physical medicine devices in the FDA’s Center for Devices and Radiological Health, in the agency’s announcement of the approval. The device “could help them to avoid more extensive surgical treatments,” such as thalamotomy or a deep brain stimulation device, Dr. Peña said.
The clinical data used to support the approval included a double-blind, controlled trial of 76 patients with essential tremor who had not responded to medication therapy.
The 56 patients who were randomly selected to receive the Exablate Neuro treatment showed nearly a 50% improvement in their tremors and motor function 3 months after treatment, compared with their baseline score. The 20 patients who received a sham control treatment had no improvement, and some experienced a slight worsening after the sham procedure before they crossed over into the treatment group at the 3-month time point.
At 12 months post procedure, the treatment group retained a 40% improvement in those scores, compared with baseline.
Adverse events reported by patients treated with the device were similar to those reported by patients who have undergone thalamotomy. Events included numbness/tingling of the fingers, headache, imbalance/unsteadiness, loss of control of body movements (ataxia), or gait disturbance.
Other side effects identified as possibly related to treatment with MR-guided focused ultrasound treatments include tissue damage in an area other than the treatment area, hemorrhage in the treated area requiring emergency treatment, skin burns with ulceration of the skin, skin retraction, and scar formation and blood clots.
InSightec manufacturers the Exablate Neuro device.
Endobariatrics: Coming to a clinic near you
SAN DIEGO – Device companies are working hard to bring obesity management to the endoscopy suite.
The field is called endobariatrics, and its goal is to fill the gap between surgery and pharmacotherapy. Drugs and lifestyle counseling don’t work too well, but a lot of people don’t want to go under the knife, so something is needed in the middle. Endobariatrics has the potential to be a boon for both obese patients and gastroenterology practices.
Several new investigational devices and approaches were showcased at the annual Digestive Disease Week; some “are beginning to approach the kind of results we see with surgical techniques,” said Steven Edmundowicz, MD, medical director of the University of Colorado Digestive Health Center, Aurora.
“We are seeing a tremendous amount of development in this space, but it’s early, and we have to be cautious,” he said. There have already been a few disappointments, including the EndoBarrier, a fluoropolymer liner anchored in the duodenal bulb and unfurled down the duodenum to block food absorption. A key U.S. trial was recently halted due to liver abscesses.
Dr. Edmundowicz reviewed the latest developments presented at DDW.
Self-assembling magnets for dual-path enteral anastomoses
The goal of the GI Windows system is to create a partial jejunoileal, side to side bypass without surgery. A 28-mm magnet ring is introduced to the ileum by colonoscopy, and a second ring to the jejunum by endoscopy. The rings snap together and tissue caught between them dies from pressure necrosis, leaving patients with a jejunoileal communication. Once food reaches that point, it either diverts through the anastomosis or continues past it down the digestive track. The magnets pass after the anastomosis forms in a week or so.
In a 6-month feasibility study from the Czech Republic, 10 obese patients lost 28.3% of their excess weight without diet restrictions. Those with diabetes had a mean hemoglobin A1c drop of 1.8%, and normalization of fasting blood glucose levels. The procedure took just over an hour and a half after the first five cases.
“I am very excited about [this]; I really want to see where the data are going,” Dr. Edmundowicz said.
Duodenal mucosal resurfacing
The idea of the Revita System (Fractyl) is to ablate “diabetic mucosa” in the duodenum so that normal mucosa can replace it. Saline is injected endoscopically under a portion of the duodenal mucosa to lift it off the muscularis; once isolated, the mucosa is destroyed – some in the audience thought “cooked” was a better word – by exposure to a hot water balloon catheter threaded into the lumen.
Thirty-nine overweight or obese type 2 diabetics had a 1.2% improvement at 6 months from a baseline hemoglobin A1c of 9.6% in a series from Santiago, Chile. Weight loss was modest in the trial; the system is being developed for type 2 diabetics.
There is some histologic support for the notion of a diabetic mucosa with both structural and hormonal aberrations, but it’s unclear if it’s a sign or cause of sickness. Even so, “the mucosa regenerates” and won’t be diabetic “for a while” after the procedure, said investigator Manoel Galvao Neto, MD, of the Gastro Obeso Center, São Paulo.
Gastric balloons
Inflating a balloon in the stomach to make people feel full isn’t new, but the notion of putting the balloon into a capsule that patients can swallow and inflating it through a tether is a more recent notion.
The Obalon (Obalon Therapeutics) is one such device. In an unblinded, sham-controlled trial with 336 obese patients, subjects who got the 250-mL, nitrogen-filled Obalons – most received three – lost about 3% more of their total body weight at 24 weeks than those who did not. Although swallowed, Obalon is removed endoscopically. Meanwhile, 34 obese patients who swallowed the 550-mL, fluid-filled Elipse balloon (Allurion) had a total body weight loss of 9.5% and mean excess weight loss of 37.2% at 4 months, by which time Elipse deflates on its own and passes without endoscopic retrieval.
“This is a very promising approach. I am very excited about digested balloons,” said Dr. Edmundowicz, an investigator in the Obalon study.
Endoscopic sleeve gastroplasty
Endoscopic sleeve gastroplasty duplicates sleeve gastrectomy with stitches placed endoscopically to seal off the greater curvature of the stomach; functionally, patients are left with a narrow sleeve of a stomach. In a multicenter series presented at DDW, 242 patients had a mean total body weight loss of 19.8% at 18 months, with a low incidence of complications. “Weight loss appears to be continuing,” Dr. Edmundowicz said. Investigators used the Apollo OverStitch (Apollo Endosurgery) to place the sutures.
Aspiration therapy
With Food and Drug Administration approval on June 14, AspireAssist (Aspire Bariatrics) is probably the best known of the newer approaches. Patients drain a portion of their meals through an endoscopically placed percutaneous gastrostomy tube a half hour or so after eating. It takes 5-10 minutes. The agency is eager to keep it out of the hands of bulimics.
One-year data were reported at DDW; 111 obese AspireAssist subjects lost a mean of 37.2% of their excess weight versus 13% in 60 patients randomized to lifestyle counseling alone.
“It may not be aesthetically pleasing, but it certainly works. It’s a viable technology,” said Dr. Edmundowicz, who was an investigator.
The studies were funded by companies developing the devices and techniques. Dr. Edmundowicz has stock options, or is a consultant or researcher, Aspire, Obalon, GI Dynamics, Elira, and other firms.
SAN DIEGO – Device companies are working hard to bring obesity management to the endoscopy suite.
The field is called endobariatrics, and its goal is to fill the gap between surgery and pharmacotherapy. Drugs and lifestyle counseling don’t work too well, but a lot of people don’t want to go under the knife, so something is needed in the middle. Endobariatrics has the potential to be a boon for both obese patients and gastroenterology practices.
Several new investigational devices and approaches were showcased at the annual Digestive Disease Week; some “are beginning to approach the kind of results we see with surgical techniques,” said Steven Edmundowicz, MD, medical director of the University of Colorado Digestive Health Center, Aurora.
“We are seeing a tremendous amount of development in this space, but it’s early, and we have to be cautious,” he said. There have already been a few disappointments, including the EndoBarrier, a fluoropolymer liner anchored in the duodenal bulb and unfurled down the duodenum to block food absorption. A key U.S. trial was recently halted due to liver abscesses.
Dr. Edmundowicz reviewed the latest developments presented at DDW.
Self-assembling magnets for dual-path enteral anastomoses
The goal of the GI Windows system is to create a partial jejunoileal, side to side bypass without surgery. A 28-mm magnet ring is introduced to the ileum by colonoscopy, and a second ring to the jejunum by endoscopy. The rings snap together and tissue caught between them dies from pressure necrosis, leaving patients with a jejunoileal communication. Once food reaches that point, it either diverts through the anastomosis or continues past it down the digestive track. The magnets pass after the anastomosis forms in a week or so.
In a 6-month feasibility study from the Czech Republic, 10 obese patients lost 28.3% of their excess weight without diet restrictions. Those with diabetes had a mean hemoglobin A1c drop of 1.8%, and normalization of fasting blood glucose levels. The procedure took just over an hour and a half after the first five cases.
“I am very excited about [this]; I really want to see where the data are going,” Dr. Edmundowicz said.
Duodenal mucosal resurfacing
The idea of the Revita System (Fractyl) is to ablate “diabetic mucosa” in the duodenum so that normal mucosa can replace it. Saline is injected endoscopically under a portion of the duodenal mucosa to lift it off the muscularis; once isolated, the mucosa is destroyed – some in the audience thought “cooked” was a better word – by exposure to a hot water balloon catheter threaded into the lumen.
Thirty-nine overweight or obese type 2 diabetics had a 1.2% improvement at 6 months from a baseline hemoglobin A1c of 9.6% in a series from Santiago, Chile. Weight loss was modest in the trial; the system is being developed for type 2 diabetics.
There is some histologic support for the notion of a diabetic mucosa with both structural and hormonal aberrations, but it’s unclear if it’s a sign or cause of sickness. Even so, “the mucosa regenerates” and won’t be diabetic “for a while” after the procedure, said investigator Manoel Galvao Neto, MD, of the Gastro Obeso Center, São Paulo.
Gastric balloons
Inflating a balloon in the stomach to make people feel full isn’t new, but the notion of putting the balloon into a capsule that patients can swallow and inflating it through a tether is a more recent notion.
The Obalon (Obalon Therapeutics) is one such device. In an unblinded, sham-controlled trial with 336 obese patients, subjects who got the 250-mL, nitrogen-filled Obalons – most received three – lost about 3% more of their total body weight at 24 weeks than those who did not. Although swallowed, Obalon is removed endoscopically. Meanwhile, 34 obese patients who swallowed the 550-mL, fluid-filled Elipse balloon (Allurion) had a total body weight loss of 9.5% and mean excess weight loss of 37.2% at 4 months, by which time Elipse deflates on its own and passes without endoscopic retrieval.
“This is a very promising approach. I am very excited about digested balloons,” said Dr. Edmundowicz, an investigator in the Obalon study.
Endoscopic sleeve gastroplasty
Endoscopic sleeve gastroplasty duplicates sleeve gastrectomy with stitches placed endoscopically to seal off the greater curvature of the stomach; functionally, patients are left with a narrow sleeve of a stomach. In a multicenter series presented at DDW, 242 patients had a mean total body weight loss of 19.8% at 18 months, with a low incidence of complications. “Weight loss appears to be continuing,” Dr. Edmundowicz said. Investigators used the Apollo OverStitch (Apollo Endosurgery) to place the sutures.
Aspiration therapy
With Food and Drug Administration approval on June 14, AspireAssist (Aspire Bariatrics) is probably the best known of the newer approaches. Patients drain a portion of their meals through an endoscopically placed percutaneous gastrostomy tube a half hour or so after eating. It takes 5-10 minutes. The agency is eager to keep it out of the hands of bulimics.
One-year data were reported at DDW; 111 obese AspireAssist subjects lost a mean of 37.2% of their excess weight versus 13% in 60 patients randomized to lifestyle counseling alone.
“It may not be aesthetically pleasing, but it certainly works. It’s a viable technology,” said Dr. Edmundowicz, who was an investigator.
The studies were funded by companies developing the devices and techniques. Dr. Edmundowicz has stock options, or is a consultant or researcher, Aspire, Obalon, GI Dynamics, Elira, and other firms.
SAN DIEGO – Device companies are working hard to bring obesity management to the endoscopy suite.
The field is called endobariatrics, and its goal is to fill the gap between surgery and pharmacotherapy. Drugs and lifestyle counseling don’t work too well, but a lot of people don’t want to go under the knife, so something is needed in the middle. Endobariatrics has the potential to be a boon for both obese patients and gastroenterology practices.
Several new investigational devices and approaches were showcased at the annual Digestive Disease Week; some “are beginning to approach the kind of results we see with surgical techniques,” said Steven Edmundowicz, MD, medical director of the University of Colorado Digestive Health Center, Aurora.
“We are seeing a tremendous amount of development in this space, but it’s early, and we have to be cautious,” he said. There have already been a few disappointments, including the EndoBarrier, a fluoropolymer liner anchored in the duodenal bulb and unfurled down the duodenum to block food absorption. A key U.S. trial was recently halted due to liver abscesses.
Dr. Edmundowicz reviewed the latest developments presented at DDW.
Self-assembling magnets for dual-path enteral anastomoses
The goal of the GI Windows system is to create a partial jejunoileal, side to side bypass without surgery. A 28-mm magnet ring is introduced to the ileum by colonoscopy, and a second ring to the jejunum by endoscopy. The rings snap together and tissue caught between them dies from pressure necrosis, leaving patients with a jejunoileal communication. Once food reaches that point, it either diverts through the anastomosis or continues past it down the digestive track. The magnets pass after the anastomosis forms in a week or so.
In a 6-month feasibility study from the Czech Republic, 10 obese patients lost 28.3% of their excess weight without diet restrictions. Those with diabetes had a mean hemoglobin A1c drop of 1.8%, and normalization of fasting blood glucose levels. The procedure took just over an hour and a half after the first five cases.
“I am very excited about [this]; I really want to see where the data are going,” Dr. Edmundowicz said.
Duodenal mucosal resurfacing
The idea of the Revita System (Fractyl) is to ablate “diabetic mucosa” in the duodenum so that normal mucosa can replace it. Saline is injected endoscopically under a portion of the duodenal mucosa to lift it off the muscularis; once isolated, the mucosa is destroyed – some in the audience thought “cooked” was a better word – by exposure to a hot water balloon catheter threaded into the lumen.
Thirty-nine overweight or obese type 2 diabetics had a 1.2% improvement at 6 months from a baseline hemoglobin A1c of 9.6% in a series from Santiago, Chile. Weight loss was modest in the trial; the system is being developed for type 2 diabetics.
There is some histologic support for the notion of a diabetic mucosa with both structural and hormonal aberrations, but it’s unclear if it’s a sign or cause of sickness. Even so, “the mucosa regenerates” and won’t be diabetic “for a while” after the procedure, said investigator Manoel Galvao Neto, MD, of the Gastro Obeso Center, São Paulo.
Gastric balloons
Inflating a balloon in the stomach to make people feel full isn’t new, but the notion of putting the balloon into a capsule that patients can swallow and inflating it through a tether is a more recent notion.
The Obalon (Obalon Therapeutics) is one such device. In an unblinded, sham-controlled trial with 336 obese patients, subjects who got the 250-mL, nitrogen-filled Obalons – most received three – lost about 3% more of their total body weight at 24 weeks than those who did not. Although swallowed, Obalon is removed endoscopically. Meanwhile, 34 obese patients who swallowed the 550-mL, fluid-filled Elipse balloon (Allurion) had a total body weight loss of 9.5% and mean excess weight loss of 37.2% at 4 months, by which time Elipse deflates on its own and passes without endoscopic retrieval.
“This is a very promising approach. I am very excited about digested balloons,” said Dr. Edmundowicz, an investigator in the Obalon study.
Endoscopic sleeve gastroplasty
Endoscopic sleeve gastroplasty duplicates sleeve gastrectomy with stitches placed endoscopically to seal off the greater curvature of the stomach; functionally, patients are left with a narrow sleeve of a stomach. In a multicenter series presented at DDW, 242 patients had a mean total body weight loss of 19.8% at 18 months, with a low incidence of complications. “Weight loss appears to be continuing,” Dr. Edmundowicz said. Investigators used the Apollo OverStitch (Apollo Endosurgery) to place the sutures.
Aspiration therapy
With Food and Drug Administration approval on June 14, AspireAssist (Aspire Bariatrics) is probably the best known of the newer approaches. Patients drain a portion of their meals through an endoscopically placed percutaneous gastrostomy tube a half hour or so after eating. It takes 5-10 minutes. The agency is eager to keep it out of the hands of bulimics.
One-year data were reported at DDW; 111 obese AspireAssist subjects lost a mean of 37.2% of their excess weight versus 13% in 60 patients randomized to lifestyle counseling alone.
“It may not be aesthetically pleasing, but it certainly works. It’s a viable technology,” said Dr. Edmundowicz, who was an investigator.
The studies were funded by companies developing the devices and techniques. Dr. Edmundowicz has stock options, or is a consultant or researcher, Aspire, Obalon, GI Dynamics, Elira, and other firms.
AT DDW® 2016
Safety of sentinel node dissection alone holds up a decade out
CHICAGO – Women with clinical early-stage breast cancer and a positive sentinel lymph node who receive breast-conserving therapy can safely skip an axillary lymph node dissection (ALND), and therefore avoid its associated morbidity, confirms long-term follow-up of the Z0011 trial conducted by the American College of Surgeons Oncology Group and the Alliance for Clinical Trials in Oncology.
The phase III trial enrolled 891 women with clinical T1-2,N0,M0 disease who underwent lumpectomy and were found to have sentinel node involvement. They were randomized to ALND or no further surgery, followed by whole-breast radiation therapy and, in most cases, systemic adjuvant therapy.
The trial was closed early because of low rates of accrual and events. Results at 6.3 years of follow-up showed that compared with peers who had an ALND, the women who skipped this surgery did not have inferior 5-year rates of locoregional recurrence or overall survival (JAMA. 2011;305:569-75).
“The study, however, like most breast cancer studies, contained mostly postmenopausal women with hormone receptor–positive tumors who are known to have late recurrences, and it was criticized for short follow-up,” commented first author Armando E. Giuliano, MD, executive vice chair of surgical oncology in the department of surgery, and associate director of surgical oncology in the Samuel Oschin Comprehensive Cancer Institute, Los Angeles.
In an update of the findings, now with a median follow-up of 9.3 years, the groups were statistically indistinguishable with respect to 10-year rates of the same outcomes, he reported at the annual meeting of the American Society of Clinical Oncology.
“This study... shows that sentinel node biopsy alone provides excellent 10-year locoregional control and survival comparable to completion axillary lymph node dissection for these patients, even with long-term follow-up,” he maintained. “Routine use of axillary lymph node dissection should be abandoned.”
“This was designed as a noninferiority trial, and I would suggest that based on the data we have seen, even if they had hit their target accrual, the outcomes would not be different,” said invited discussant Elizabeth A. Mittendorf, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston. “Clearly, even before today’s presentation, Z0011 has been identified as a practice-changing trial, as evidenced by the NCCN guidelines.”
In fact, a study last year showed that among patients in the general U.S. population meeting the trial’s enrollment criteria, the use of sentinel lymph node dissection alone has increased significantly since the Z0011 results were first reported (J Am Coll Surg. 2015;221:71-81).
“However, I would highlight that we have also seen an increase in omission of axillary lymph node dissection for patients who do not meet the Z0011 criteria to include those not planned for radiotherapy, those receiving APBI [accelerated partial breast irradiation], and those undergoing mastectomy,” she added. “I highlight these examples specifically because it’s been suggested that one of the reasons the patients on the trial have outstanding regional control is because of the radiation administered as part of their breast-conserving therapy.”
“We will obtain additional data on the locoregional management of these early-stage patients with clinically node-negative breast cancer,” Dr. Mittendorf predicted, pointing to the similar POSNOC trial (which is comparing systemic therapy with versus without axillary treatment) and SOUND trial (which is comparing sentinel node dissection versus no axillary surgery).
In Z0011, all women had tumor in sentinel nodes detected with hematoxylin and eosin staining. Those with sentinel node tumor detected only by immunohistochemistry were excluded, as were those who had matted nodes, three or more involved sentinel nodes, or planned third-field (nodal) irradiation.
Overall, 27.4% of the patients in the ALND group had additional positive nodes removed beyond their sentinel nodes. “There is no reason to suspect that women with sentinel node biopsy [only] had fewer involved nodes than the women treated with axillary lymph node dissection,” Dr. Giuliano commented; thus, a similar share of the former group likely had residual axillary disease that went unresected.
The updated findings showed that the women received ALND and the women who did not were statistically indistinguishable with respect to the 10-year rate of locoregional recurrence (6.2% and 5.3%). Of note, only a single regional recurrence was seen after the initial 5 years of follow-up, and it occurred in the group that did not have ALND.
The groups treated with and without ALND were also statistically indistinguishable with respect to 10-year rates of disease-free survival (78.2% and 80.2%), locoregional recurrence–free survival (81.2% and 83.0%), and overall survival (83.6% and 86.3%).
In multivariate analysis, omission of ALND did not significantly predict locoregional recurrence or overall survival, reported Dr. Giuliano. Additionally, stratified analysis showed that the lack of difference in overall survival between study groups was the same whether tumors had hormone receptors or not.
In a related analysis of radiation protocol deviations in a subset of women from the trial, 11% did not receive any radiation therapy, while 18.9% received third-field radiation, with equal distribution of the latter between study groups (J Clin Oncol. 2014;32:3600-6). Omission of radiation was associated with an increased risk of local recurrence and death, but it did not affect nodal recurrences. Receipt of third-field radiation did not influence survival.
CHICAGO – Women with clinical early-stage breast cancer and a positive sentinel lymph node who receive breast-conserving therapy can safely skip an axillary lymph node dissection (ALND), and therefore avoid its associated morbidity, confirms long-term follow-up of the Z0011 trial conducted by the American College of Surgeons Oncology Group and the Alliance for Clinical Trials in Oncology.
The phase III trial enrolled 891 women with clinical T1-2,N0,M0 disease who underwent lumpectomy and were found to have sentinel node involvement. They were randomized to ALND or no further surgery, followed by whole-breast radiation therapy and, in most cases, systemic adjuvant therapy.
The trial was closed early because of low rates of accrual and events. Results at 6.3 years of follow-up showed that compared with peers who had an ALND, the women who skipped this surgery did not have inferior 5-year rates of locoregional recurrence or overall survival (JAMA. 2011;305:569-75).
“The study, however, like most breast cancer studies, contained mostly postmenopausal women with hormone receptor–positive tumors who are known to have late recurrences, and it was criticized for short follow-up,” commented first author Armando E. Giuliano, MD, executive vice chair of surgical oncology in the department of surgery, and associate director of surgical oncology in the Samuel Oschin Comprehensive Cancer Institute, Los Angeles.
In an update of the findings, now with a median follow-up of 9.3 years, the groups were statistically indistinguishable with respect to 10-year rates of the same outcomes, he reported at the annual meeting of the American Society of Clinical Oncology.
“This study... shows that sentinel node biopsy alone provides excellent 10-year locoregional control and survival comparable to completion axillary lymph node dissection for these patients, even with long-term follow-up,” he maintained. “Routine use of axillary lymph node dissection should be abandoned.”
“This was designed as a noninferiority trial, and I would suggest that based on the data we have seen, even if they had hit their target accrual, the outcomes would not be different,” said invited discussant Elizabeth A. Mittendorf, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston. “Clearly, even before today’s presentation, Z0011 has been identified as a practice-changing trial, as evidenced by the NCCN guidelines.”
In fact, a study last year showed that among patients in the general U.S. population meeting the trial’s enrollment criteria, the use of sentinel lymph node dissection alone has increased significantly since the Z0011 results were first reported (J Am Coll Surg. 2015;221:71-81).
“However, I would highlight that we have also seen an increase in omission of axillary lymph node dissection for patients who do not meet the Z0011 criteria to include those not planned for radiotherapy, those receiving APBI [accelerated partial breast irradiation], and those undergoing mastectomy,” she added. “I highlight these examples specifically because it’s been suggested that one of the reasons the patients on the trial have outstanding regional control is because of the radiation administered as part of their breast-conserving therapy.”
“We will obtain additional data on the locoregional management of these early-stage patients with clinically node-negative breast cancer,” Dr. Mittendorf predicted, pointing to the similar POSNOC trial (which is comparing systemic therapy with versus without axillary treatment) and SOUND trial (which is comparing sentinel node dissection versus no axillary surgery).
In Z0011, all women had tumor in sentinel nodes detected with hematoxylin and eosin staining. Those with sentinel node tumor detected only by immunohistochemistry were excluded, as were those who had matted nodes, three or more involved sentinel nodes, or planned third-field (nodal) irradiation.
Overall, 27.4% of the patients in the ALND group had additional positive nodes removed beyond their sentinel nodes. “There is no reason to suspect that women with sentinel node biopsy [only] had fewer involved nodes than the women treated with axillary lymph node dissection,” Dr. Giuliano commented; thus, a similar share of the former group likely had residual axillary disease that went unresected.
The updated findings showed that the women received ALND and the women who did not were statistically indistinguishable with respect to the 10-year rate of locoregional recurrence (6.2% and 5.3%). Of note, only a single regional recurrence was seen after the initial 5 years of follow-up, and it occurred in the group that did not have ALND.
The groups treated with and without ALND were also statistically indistinguishable with respect to 10-year rates of disease-free survival (78.2% and 80.2%), locoregional recurrence–free survival (81.2% and 83.0%), and overall survival (83.6% and 86.3%).
In multivariate analysis, omission of ALND did not significantly predict locoregional recurrence or overall survival, reported Dr. Giuliano. Additionally, stratified analysis showed that the lack of difference in overall survival between study groups was the same whether tumors had hormone receptors or not.
In a related analysis of radiation protocol deviations in a subset of women from the trial, 11% did not receive any radiation therapy, while 18.9% received third-field radiation, with equal distribution of the latter between study groups (J Clin Oncol. 2014;32:3600-6). Omission of radiation was associated with an increased risk of local recurrence and death, but it did not affect nodal recurrences. Receipt of third-field radiation did not influence survival.
CHICAGO – Women with clinical early-stage breast cancer and a positive sentinel lymph node who receive breast-conserving therapy can safely skip an axillary lymph node dissection (ALND), and therefore avoid its associated morbidity, confirms long-term follow-up of the Z0011 trial conducted by the American College of Surgeons Oncology Group and the Alliance for Clinical Trials in Oncology.
The phase III trial enrolled 891 women with clinical T1-2,N0,M0 disease who underwent lumpectomy and were found to have sentinel node involvement. They were randomized to ALND or no further surgery, followed by whole-breast radiation therapy and, in most cases, systemic adjuvant therapy.
The trial was closed early because of low rates of accrual and events. Results at 6.3 years of follow-up showed that compared with peers who had an ALND, the women who skipped this surgery did not have inferior 5-year rates of locoregional recurrence or overall survival (JAMA. 2011;305:569-75).
“The study, however, like most breast cancer studies, contained mostly postmenopausal women with hormone receptor–positive tumors who are known to have late recurrences, and it was criticized for short follow-up,” commented first author Armando E. Giuliano, MD, executive vice chair of surgical oncology in the department of surgery, and associate director of surgical oncology in the Samuel Oschin Comprehensive Cancer Institute, Los Angeles.
In an update of the findings, now with a median follow-up of 9.3 years, the groups were statistically indistinguishable with respect to 10-year rates of the same outcomes, he reported at the annual meeting of the American Society of Clinical Oncology.
“This study... shows that sentinel node biopsy alone provides excellent 10-year locoregional control and survival comparable to completion axillary lymph node dissection for these patients, even with long-term follow-up,” he maintained. “Routine use of axillary lymph node dissection should be abandoned.”
“This was designed as a noninferiority trial, and I would suggest that based on the data we have seen, even if they had hit their target accrual, the outcomes would not be different,” said invited discussant Elizabeth A. Mittendorf, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston. “Clearly, even before today’s presentation, Z0011 has been identified as a practice-changing trial, as evidenced by the NCCN guidelines.”
In fact, a study last year showed that among patients in the general U.S. population meeting the trial’s enrollment criteria, the use of sentinel lymph node dissection alone has increased significantly since the Z0011 results were first reported (J Am Coll Surg. 2015;221:71-81).
“However, I would highlight that we have also seen an increase in omission of axillary lymph node dissection for patients who do not meet the Z0011 criteria to include those not planned for radiotherapy, those receiving APBI [accelerated partial breast irradiation], and those undergoing mastectomy,” she added. “I highlight these examples specifically because it’s been suggested that one of the reasons the patients on the trial have outstanding regional control is because of the radiation administered as part of their breast-conserving therapy.”
“We will obtain additional data on the locoregional management of these early-stage patients with clinically node-negative breast cancer,” Dr. Mittendorf predicted, pointing to the similar POSNOC trial (which is comparing systemic therapy with versus without axillary treatment) and SOUND trial (which is comparing sentinel node dissection versus no axillary surgery).
In Z0011, all women had tumor in sentinel nodes detected with hematoxylin and eosin staining. Those with sentinel node tumor detected only by immunohistochemistry were excluded, as were those who had matted nodes, three or more involved sentinel nodes, or planned third-field (nodal) irradiation.
Overall, 27.4% of the patients in the ALND group had additional positive nodes removed beyond their sentinel nodes. “There is no reason to suspect that women with sentinel node biopsy [only] had fewer involved nodes than the women treated with axillary lymph node dissection,” Dr. Giuliano commented; thus, a similar share of the former group likely had residual axillary disease that went unresected.
The updated findings showed that the women received ALND and the women who did not were statistically indistinguishable with respect to the 10-year rate of locoregional recurrence (6.2% and 5.3%). Of note, only a single regional recurrence was seen after the initial 5 years of follow-up, and it occurred in the group that did not have ALND.
The groups treated with and without ALND were also statistically indistinguishable with respect to 10-year rates of disease-free survival (78.2% and 80.2%), locoregional recurrence–free survival (81.2% and 83.0%), and overall survival (83.6% and 86.3%).
In multivariate analysis, omission of ALND did not significantly predict locoregional recurrence or overall survival, reported Dr. Giuliano. Additionally, stratified analysis showed that the lack of difference in overall survival between study groups was the same whether tumors had hormone receptors or not.
In a related analysis of radiation protocol deviations in a subset of women from the trial, 11% did not receive any radiation therapy, while 18.9% received third-field radiation, with equal distribution of the latter between study groups (J Clin Oncol. 2014;32:3600-6). Omission of radiation was associated with an increased risk of local recurrence and death, but it did not affect nodal recurrences. Receipt of third-field radiation did not influence survival.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: In women with clinical early-stage breast cancer who have a positive sentinel node and undergo breast-conserving therapy, skipping ALND does not compromise outcomes.
Major finding: Women treated with and without ALND were statistically indistinguishable with respect to 10-year rates of locoregional recurrence (6.2% and 5.3%), disease-free survival (78.2% and 80.2%), and overall survival (83.6% and 86.3%).
Data source: A randomized phase III trial among 891 women with clinical T1-2,N0,M0 breast cancer and positive sentinel nodes treated with breast-conserving therapy and usually adjuvant systemic therapy (ACOSOG Z0011).
Disclosures: Dr. Giuliano disclosed that he had no relevant conflicts of interest.
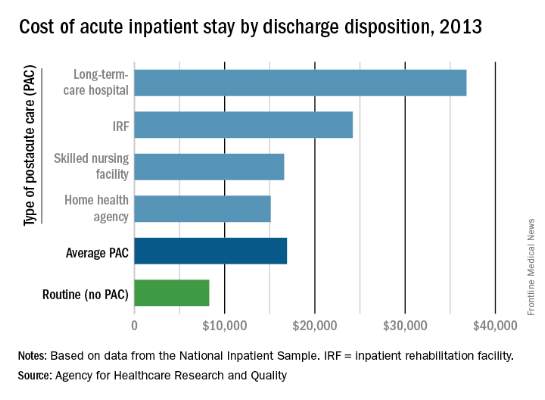
Hospital costs higher for patients discharged to postacute care
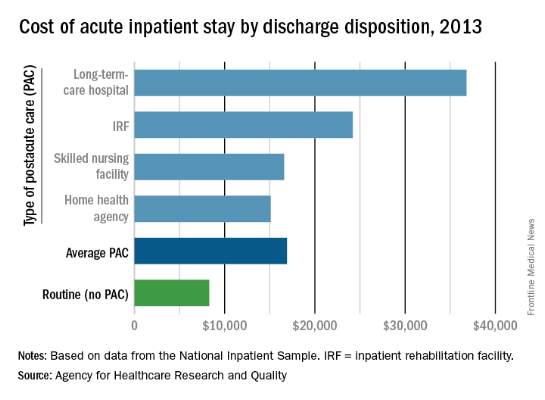
The average cost of U.S. hospital stays for injury or illness in patients discharged to postacute care is more than double that of visits with routine discharges, according to the Agency for Healthcare Research and Quality.
For patients who were discharged from hospitals to PAC, the average cost of an inpatient visit in 2013 was $16,900, compared with $8,300 for patients with routine discharges. The inpatient visits with PAC were almost twice as long as those with routine discharge – 7.0 days vs. 3.6 days – and patients with PAC-discharge visits were much older – 69.5% were aged 65 years or older, compared with 22.4% of visits with routine discharges, the AHRQ reported.
The AHRQ used data from the 2013 National Inpatient Sample (NIS) to estimates discharges to PAC for all types of payers and describe these discharges from the perspective of payers, patients, hospitals, conditions/procedures, and geographic regions.
The cost of stays varied considerably among the various PAC settings in 2013. Inpatient stays with discharge to home health agencies had the lowest average cost at $15,100, with skilled nursing facilities next at $16,600, followed by inpatient rehabilitation facilities at $24,200 and long-term-care hospitals at $36,800. Length of stays by PAC setting showed the same trend: those with discharge to home health agencies were shortest (6.2 days) and those with discharge to long-term-care hospitals were longest (13.5 days), the AHRQ said in the report.
Inpatient stays with discharge to PAC made up 22.3% of all hospital discharges in 2013, with the bulk being discharges to home health agencies (50.1%) and skilled nursing facilities (40.5%). Discharges to inpatient rehabilitation facilities made up 7.2% of all PAC visits, while those to long-term-care hospitals were just 2.2%, the data from the NIS show.
The average cost of U.S. hospital stays for injury or illness in patients discharged to postacute care is more than double that of visits with routine discharges, according to the Agency for Healthcare Research and Quality.
For patients who were discharged from hospitals to PAC, the average cost of an inpatient visit in 2013 was $16,900, compared with $8,300 for patients with routine discharges. The inpatient visits with PAC were almost twice as long as those with routine discharge – 7.0 days vs. 3.6 days – and patients with PAC-discharge visits were much older – 69.5% were aged 65 years or older, compared with 22.4% of visits with routine discharges, the AHRQ reported.
The AHRQ used data from the 2013 National Inpatient Sample (NIS) to estimates discharges to PAC for all types of payers and describe these discharges from the perspective of payers, patients, hospitals, conditions/procedures, and geographic regions.
The cost of stays varied considerably among the various PAC settings in 2013. Inpatient stays with discharge to home health agencies had the lowest average cost at $15,100, with skilled nursing facilities next at $16,600, followed by inpatient rehabilitation facilities at $24,200 and long-term-care hospitals at $36,800. Length of stays by PAC setting showed the same trend: those with discharge to home health agencies were shortest (6.2 days) and those with discharge to long-term-care hospitals were longest (13.5 days), the AHRQ said in the report.
Inpatient stays with discharge to PAC made up 22.3% of all hospital discharges in 2013, with the bulk being discharges to home health agencies (50.1%) and skilled nursing facilities (40.5%). Discharges to inpatient rehabilitation facilities made up 7.2% of all PAC visits, while those to long-term-care hospitals were just 2.2%, the data from the NIS show.
The average cost of U.S. hospital stays for injury or illness in patients discharged to postacute care is more than double that of visits with routine discharges, according to the Agency for Healthcare Research and Quality.
For patients who were discharged from hospitals to PAC, the average cost of an inpatient visit in 2013 was $16,900, compared with $8,300 for patients with routine discharges. The inpatient visits with PAC were almost twice as long as those with routine discharge – 7.0 days vs. 3.6 days – and patients with PAC-discharge visits were much older – 69.5% were aged 65 years or older, compared with 22.4% of visits with routine discharges, the AHRQ reported.
The AHRQ used data from the 2013 National Inpatient Sample (NIS) to estimates discharges to PAC for all types of payers and describe these discharges from the perspective of payers, patients, hospitals, conditions/procedures, and geographic regions.
The cost of stays varied considerably among the various PAC settings in 2013. Inpatient stays with discharge to home health agencies had the lowest average cost at $15,100, with skilled nursing facilities next at $16,600, followed by inpatient rehabilitation facilities at $24,200 and long-term-care hospitals at $36,800. Length of stays by PAC setting showed the same trend: those with discharge to home health agencies were shortest (6.2 days) and those with discharge to long-term-care hospitals were longest (13.5 days), the AHRQ said in the report.
Inpatient stays with discharge to PAC made up 22.3% of all hospital discharges in 2013, with the bulk being discharges to home health agencies (50.1%) and skilled nursing facilities (40.5%). Discharges to inpatient rehabilitation facilities made up 7.2% of all PAC visits, while those to long-term-care hospitals were just 2.2%, the data from the NIS show.
Hospital costs higher for patients discharged to postacute care
The average cost of U.S. hospital stays for injury or illness in patients discharged to postacute care is more than double that of visits with routine discharges, according to the Agency for Healthcare Research and Quality.
For patients who were discharged from hospitals to PAC, the average cost of an inpatient visit in 2013 was $16,900, compared with $8,300 for patients with routine discharges. The inpatient visits with PAC were almost twice as long as those with routine discharge – 7.0 days vs. 3.6 days – and patients with PAC-discharge visits were much older – 69.5% were aged 65 years or older, compared with 22.4% of visits with routine discharges, the AHRQ reported.
The AHRQ used data from the 2013 National Inpatient Sample (NIS) to estimates discharges to PAC for all types of payers and describe these discharges from the perspective of payers, patients, hospitals, conditions/procedures, and geographic regions.
The cost of stays varied considerably among the various PAC settings in 2013. Inpatient stays with discharge to home health agencies had the lowest average cost at $15,100, with skilled nursing facilities next at $16,600, followed by inpatient rehabilitation facilities at $24,200 and long-term-care hospitals at $36,800. Length of stays by PAC setting showed the same trend: those with discharge to home health agencies were shortest (6.2 days) and those with discharge to long-term-care hospitals were longest (13.5 days), the AHRQ said in the report.
Inpatient stays with discharge to PAC made up 22.3% of all hospital discharges in 2013, with the bulk being discharges to home health agencies (50.1%) and skilled nursing facilities (40.5%). Discharges to inpatient rehabilitation facilities made up 7.2% of all PAC visits, while those to long-term-care hospitals were just 2.2%, the data from the NIS show.
The average cost of U.S. hospital stays for injury or illness in patients discharged to postacute care is more than double that of visits with routine discharges, according to the Agency for Healthcare Research and Quality.
For patients who were discharged from hospitals to PAC, the average cost of an inpatient visit in 2013 was $16,900, compared with $8,300 for patients with routine discharges. The inpatient visits with PAC were almost twice as long as those with routine discharge – 7.0 days vs. 3.6 days – and patients with PAC-discharge visits were much older – 69.5% were aged 65 years or older, compared with 22.4% of visits with routine discharges, the AHRQ reported.
The AHRQ used data from the 2013 National Inpatient Sample (NIS) to estimates discharges to PAC for all types of payers and describe these discharges from the perspective of payers, patients, hospitals, conditions/procedures, and geographic regions.
The cost of stays varied considerably among the various PAC settings in 2013. Inpatient stays with discharge to home health agencies had the lowest average cost at $15,100, with skilled nursing facilities next at $16,600, followed by inpatient rehabilitation facilities at $24,200 and long-term-care hospitals at $36,800. Length of stays by PAC setting showed the same trend: those with discharge to home health agencies were shortest (6.2 days) and those with discharge to long-term-care hospitals were longest (13.5 days), the AHRQ said in the report.
Inpatient stays with discharge to PAC made up 22.3% of all hospital discharges in 2013, with the bulk being discharges to home health agencies (50.1%) and skilled nursing facilities (40.5%). Discharges to inpatient rehabilitation facilities made up 7.2% of all PAC visits, while those to long-term-care hospitals were just 2.2%, the data from the NIS show.
The average cost of U.S. hospital stays for injury or illness in patients discharged to postacute care is more than double that of visits with routine discharges, according to the Agency for Healthcare Research and Quality.
For patients who were discharged from hospitals to PAC, the average cost of an inpatient visit in 2013 was $16,900, compared with $8,300 for patients with routine discharges. The inpatient visits with PAC were almost twice as long as those with routine discharge – 7.0 days vs. 3.6 days – and patients with PAC-discharge visits were much older – 69.5% were aged 65 years or older, compared with 22.4% of visits with routine discharges, the AHRQ reported.
The AHRQ used data from the 2013 National Inpatient Sample (NIS) to estimates discharges to PAC for all types of payers and describe these discharges from the perspective of payers, patients, hospitals, conditions/procedures, and geographic regions.
The cost of stays varied considerably among the various PAC settings in 2013. Inpatient stays with discharge to home health agencies had the lowest average cost at $15,100, with skilled nursing facilities next at $16,600, followed by inpatient rehabilitation facilities at $24,200 and long-term-care hospitals at $36,800. Length of stays by PAC setting showed the same trend: those with discharge to home health agencies were shortest (6.2 days) and those with discharge to long-term-care hospitals were longest (13.5 days), the AHRQ said in the report.
Inpatient stays with discharge to PAC made up 22.3% of all hospital discharges in 2013, with the bulk being discharges to home health agencies (50.1%) and skilled nursing facilities (40.5%). Discharges to inpatient rehabilitation facilities made up 7.2% of all PAC visits, while those to long-term-care hospitals were just 2.2%, the data from the NIS show.
Medical errors and the law
Question: A patient was admitted with heart failure, developed deep vein thrombosis, and was started on warfarin and Lovenox as “bridge” therapy. On day 4, the patient achieved anticoagulation with a prothrombin time of 29.8 and international normalized ratio (INR) of 2.86, but continued to receive both warfarin and Lovenox for a total of 13 days. Both medications were dispensed and administered for 2 days when the PT was greater than 50; the supratherapeutic coagulation profile result was overlooked. Medications held on day 14 (PT, 68; INR, 8.35). The patient developed a right subdural hematoma and was transferred to a tertiary care facility for neurosurgery consult.
Given these facts, which of the following statements is best?
A. The hospital is under a legal obligation to disclose the error.
B. The doctor should be sympathetic and apologize for the injury, but not admit fault.
C. All jurisdictions have so-called “apology statutes,” which encourage error disclosure in return for immunity.
D. This is a case of medication, not medical, error.
E. Silence is golden.
Answer: B. A recent publication concluded, “If medical error were a disease, it would rank as the third leading cause of death in the United States.”1 This is the latest follow through on the original landmark report from the Institute of Medicine in 2000, which drew the public’s attention to the fact that medical errors were responsible for between 44,000 and 98,000 annual fatalities in the United States.
A medical error denotes a preventable adverse event, which in turn can be described as an injury caused by medical mismanagement rather than the underlying condition of the patient. It is more formally defined as “the failure of a planned action to be completed as intended or the use of a wrong plan to achieve an aim.”2 The term is not synonymous with medical negligence, which is a legal term of art encompassing four separate elements: duty, breach, causation, and damages.
The most common type of medical error is a medication error, which is any preventable event that may cause or lead to inappropriate medication use or patient harm while the medication is in the control of the health professional, patient, or consumer.
Medication errors account for 6.3%-30% of all malpractice claims, and a 1999 Texas case is an example.3 A 43-year-old Hispanic man with chest pain was prescribed the anti-angina drug Isordil (isosorbide dinitrate) by his cardiologist, to be taken four times a day in doses of 20 mg. The pharmacist misread the order as Plendil (felodipine), a calcium channel blocker for treatment of hypertension. This exceeded the drug’s top dose, and the patient suffered a heart attack and died several days later.
The cardiologist’s illegible prescription was the sole reason for the error, and his overall quality of care was not at issue. The jury returned a verdict for the plaintiff, awarding $450,000 to his estate: $225,000 from the cardiologist, and $225,000 from the pharmacist.
Many, but not all, jurisdictions now require some form of reporting of medical errors occurring in a hospital setting. States such as California and Florida mandate disclosure to patients. Pennsylvania actually requires hospitals to issue a written disclosure within 7 days of a serious event.
Most states have enacted “apology statutes” to encourage open discussions with patients and their families about adverse results. The apologies may cover expressions of regret, sympathy, and compassion, and they are barred from being presented to the jury should a trial ensue. However, an acknowledgment of fault remains admissible into evidence.
Typical is California’s Evidence Code 1160(a), which provides that only “the portions of statements or benevolent gestures expressing sympathy” are inadmissible against a treating physician. On the other hand, some states have chosen to exclude all disclosures, including admissions of fault. An example is Colorado’s Apology Statute (Colo. Rev. Stat. Ann. 13-25-135), which provides that “any and all statements, affirmations, gestures, or conduct expressing apology, fault, sympathy, commiseration, condolence, compassion, or a general sense of benevolence ... shall be inadmissible as evidence of an admission of liability or as evidence of an admission against interest.”
Ohio is one of only a few states whose apology statutes fail to clearly distinguish between the admissibility of a physician’s statement of sympathy and one acknowledging fault. R.C. 2317.43, enacted by the Ohio General Assembly in 2004, renders inadmissible “statements, affirmations, gestures, or conduct expressing apology, sympathy, commiseration, condolence, compassion, or a general sense of benevolence.”
In Davis v. Wooster Orthopaedics & Sports Medicine,4 the plaintiff alleged that Dr. Michael Knapic, an orthopedic surgeon, negligently performed a lumbar microdiscectomy, severing the patient’s common iliac artery. The surgeon allegedly then said to the patient’s husband, “It’s my fault. I take full responsibility.”
In a wrongful-death action, the plaintiff argued that the statute did not prohibit the use of statements of fault, responsibility, or liability as compared to statements of sympathy or condolence.
Ohio’s Ninth Appellate District concluded that the intent behind the apology statute was to protect pure expressions of sympathy but not admissions of fault. The court held that Dr. Knapic’s statements constituted an admission of liability that could be admitted into evidence. The jury awarded damages of $3 million.
More recently, the Ohio Supreme Court ruled that Dr. Randall Smith’s alleged confession regarding accidentally sectioning his patient’s common bile duct was properly excluded from evidence, even though the incident took place before the Ohio law went into effect.5
The patient had to be readmitted within 3 weeks for obstructive jaundice. After the doctor informed her that she would have to undergo additional surgery, she became very emotional. He reportedly took her hand, saying, “I take full responsibility for this.”
The Ohio Supreme Court ruled, “The trial court had determined that Dr. Smith was faced with a distressed patient who was upset and made a statement that was designed to comfort his patient. This is precisely the type of evidence that R.C. 2317.43 was designed to exclude as evidence of liability in a medical-malpractice case.”
Do disclosures and apologies work?
Both claim frequency and severity have diminished following the adoption of a humanistic risk management policy at the Lexington Veterans Affairs Medical Center since 1987.6 The protocol includes early injury review, steadfast maintenance of the relationship between the hospital and the patient, proactive disclosure to patients who have been injured because of accidents or medical negligence, and fair compensation for injuries.
Other institutions such as the University of Michigan have adopted “disclosure and offer” in place of “denial and defend” policies, but these have yet to achieve widespread use.
Many health care providers continue to heed the traditional legal advice to say and admit nothing, believing that it is unsettled whether an apology will influence any decision to sue the doctor. They argue that the odds of a lawsuit are low to begin with.
In the oft-cited Harvard study, there was only one malpractice claim for every 7.6 adverse events caused by negligence among the 3.7% of hospitalized patients who suffered significant iatrogenic injuries, typically from errors or negligence.7
Notwithstanding the controversy, the AMA has properly taken the moral high ground: It asserts that error disclosure is the right thing to do.
References
2. Institute of Medicine: To Err is Human: Building a Safer Health System. National Academy Press, Washington, 2000.
3. Estate of Velasquez v. Albertsons, Inc. et al., Civ. No. A-103-042 (Ector Cnty, TX 1999).
4. Davis v. Wooster Orthopaedics & Sports Medicine, Inc., 193 Ohio App.3d 581 (2011).
5. Estate of Johnson v. Randall Smith, Inc., 131 Ohio St.3d 1543 (2013).
6. N Engl J Med. 2010 Apr 15;362(15):1353-6.
7. N Engl J Med. 1991 Jul 25;325(4):245-51.
Dr. Tan is emeritus professor of medicine and a former adjunct professor of law at the University of Hawaii. This article is meant to be educational and does not constitute medical, ethical or legal advice. It is adapted from the author’s book, “Medical Malpractice: Understanding the Law, Managing the Risk” (2006). For additional information, readers may contact the author at [email protected].
Question: A patient was admitted with heart failure, developed deep vein thrombosis, and was started on warfarin and Lovenox as “bridge” therapy. On day 4, the patient achieved anticoagulation with a prothrombin time of 29.8 and international normalized ratio (INR) of 2.86, but continued to receive both warfarin and Lovenox for a total of 13 days. Both medications were dispensed and administered for 2 days when the PT was greater than 50; the supratherapeutic coagulation profile result was overlooked. Medications held on day 14 (PT, 68; INR, 8.35). The patient developed a right subdural hematoma and was transferred to a tertiary care facility for neurosurgery consult.
Given these facts, which of the following statements is best?
A. The hospital is under a legal obligation to disclose the error.
B. The doctor should be sympathetic and apologize for the injury, but not admit fault.
C. All jurisdictions have so-called “apology statutes,” which encourage error disclosure in return for immunity.
D. This is a case of medication, not medical, error.
E. Silence is golden.
Answer: B. A recent publication concluded, “If medical error were a disease, it would rank as the third leading cause of death in the United States.”1 This is the latest follow through on the original landmark report from the Institute of Medicine in 2000, which drew the public’s attention to the fact that medical errors were responsible for between 44,000 and 98,000 annual fatalities in the United States.
A medical error denotes a preventable adverse event, which in turn can be described as an injury caused by medical mismanagement rather than the underlying condition of the patient. It is more formally defined as “the failure of a planned action to be completed as intended or the use of a wrong plan to achieve an aim.”2 The term is not synonymous with medical negligence, which is a legal term of art encompassing four separate elements: duty, breach, causation, and damages.
The most common type of medical error is a medication error, which is any preventable event that may cause or lead to inappropriate medication use or patient harm while the medication is in the control of the health professional, patient, or consumer.
Medication errors account for 6.3%-30% of all malpractice claims, and a 1999 Texas case is an example.3 A 43-year-old Hispanic man with chest pain was prescribed the anti-angina drug Isordil (isosorbide dinitrate) by his cardiologist, to be taken four times a day in doses of 20 mg. The pharmacist misread the order as Plendil (felodipine), a calcium channel blocker for treatment of hypertension. This exceeded the drug’s top dose, and the patient suffered a heart attack and died several days later.
The cardiologist’s illegible prescription was the sole reason for the error, and his overall quality of care was not at issue. The jury returned a verdict for the plaintiff, awarding $450,000 to his estate: $225,000 from the cardiologist, and $225,000 from the pharmacist.
Many, but not all, jurisdictions now require some form of reporting of medical errors occurring in a hospital setting. States such as California and Florida mandate disclosure to patients. Pennsylvania actually requires hospitals to issue a written disclosure within 7 days of a serious event.
Most states have enacted “apology statutes” to encourage open discussions with patients and their families about adverse results. The apologies may cover expressions of regret, sympathy, and compassion, and they are barred from being presented to the jury should a trial ensue. However, an acknowledgment of fault remains admissible into evidence.
Typical is California’s Evidence Code 1160(a), which provides that only “the portions of statements or benevolent gestures expressing sympathy” are inadmissible against a treating physician. On the other hand, some states have chosen to exclude all disclosures, including admissions of fault. An example is Colorado’s Apology Statute (Colo. Rev. Stat. Ann. 13-25-135), which provides that “any and all statements, affirmations, gestures, or conduct expressing apology, fault, sympathy, commiseration, condolence, compassion, or a general sense of benevolence ... shall be inadmissible as evidence of an admission of liability or as evidence of an admission against interest.”
Ohio is one of only a few states whose apology statutes fail to clearly distinguish between the admissibility of a physician’s statement of sympathy and one acknowledging fault. R.C. 2317.43, enacted by the Ohio General Assembly in 2004, renders inadmissible “statements, affirmations, gestures, or conduct expressing apology, sympathy, commiseration, condolence, compassion, or a general sense of benevolence.”
In Davis v. Wooster Orthopaedics & Sports Medicine,4 the plaintiff alleged that Dr. Michael Knapic, an orthopedic surgeon, negligently performed a lumbar microdiscectomy, severing the patient’s common iliac artery. The surgeon allegedly then said to the patient’s husband, “It’s my fault. I take full responsibility.”
In a wrongful-death action, the plaintiff argued that the statute did not prohibit the use of statements of fault, responsibility, or liability as compared to statements of sympathy or condolence.
Ohio’s Ninth Appellate District concluded that the intent behind the apology statute was to protect pure expressions of sympathy but not admissions of fault. The court held that Dr. Knapic’s statements constituted an admission of liability that could be admitted into evidence. The jury awarded damages of $3 million.
More recently, the Ohio Supreme Court ruled that Dr. Randall Smith’s alleged confession regarding accidentally sectioning his patient’s common bile duct was properly excluded from evidence, even though the incident took place before the Ohio law went into effect.5
The patient had to be readmitted within 3 weeks for obstructive jaundice. After the doctor informed her that she would have to undergo additional surgery, she became very emotional. He reportedly took her hand, saying, “I take full responsibility for this.”
The Ohio Supreme Court ruled, “The trial court had determined that Dr. Smith was faced with a distressed patient who was upset and made a statement that was designed to comfort his patient. This is precisely the type of evidence that R.C. 2317.43 was designed to exclude as evidence of liability in a medical-malpractice case.”
Do disclosures and apologies work?
Both claim frequency and severity have diminished following the adoption of a humanistic risk management policy at the Lexington Veterans Affairs Medical Center since 1987.6 The protocol includes early injury review, steadfast maintenance of the relationship between the hospital and the patient, proactive disclosure to patients who have been injured because of accidents or medical negligence, and fair compensation for injuries.
Other institutions such as the University of Michigan have adopted “disclosure and offer” in place of “denial and defend” policies, but these have yet to achieve widespread use.
Many health care providers continue to heed the traditional legal advice to say and admit nothing, believing that it is unsettled whether an apology will influence any decision to sue the doctor. They argue that the odds of a lawsuit are low to begin with.
In the oft-cited Harvard study, there was only one malpractice claim for every 7.6 adverse events caused by negligence among the 3.7% of hospitalized patients who suffered significant iatrogenic injuries, typically from errors or negligence.7
Notwithstanding the controversy, the AMA has properly taken the moral high ground: It asserts that error disclosure is the right thing to do.
References
2. Institute of Medicine: To Err is Human: Building a Safer Health System. National Academy Press, Washington, 2000.
3. Estate of Velasquez v. Albertsons, Inc. et al., Civ. No. A-103-042 (Ector Cnty, TX 1999).
4. Davis v. Wooster Orthopaedics & Sports Medicine, Inc., 193 Ohio App.3d 581 (2011).
5. Estate of Johnson v. Randall Smith, Inc., 131 Ohio St.3d 1543 (2013).
6. N Engl J Med. 2010 Apr 15;362(15):1353-6.
7. N Engl J Med. 1991 Jul 25;325(4):245-51.
Dr. Tan is emeritus professor of medicine and a former adjunct professor of law at the University of Hawaii. This article is meant to be educational and does not constitute medical, ethical or legal advice. It is adapted from the author’s book, “Medical Malpractice: Understanding the Law, Managing the Risk” (2006). For additional information, readers may contact the author at [email protected].
Question: A patient was admitted with heart failure, developed deep vein thrombosis, and was started on warfarin and Lovenox as “bridge” therapy. On day 4, the patient achieved anticoagulation with a prothrombin time of 29.8 and international normalized ratio (INR) of 2.86, but continued to receive both warfarin and Lovenox for a total of 13 days. Both medications were dispensed and administered for 2 days when the PT was greater than 50; the supratherapeutic coagulation profile result was overlooked. Medications held on day 14 (PT, 68; INR, 8.35). The patient developed a right subdural hematoma and was transferred to a tertiary care facility for neurosurgery consult.
Given these facts, which of the following statements is best?
A. The hospital is under a legal obligation to disclose the error.
B. The doctor should be sympathetic and apologize for the injury, but not admit fault.
C. All jurisdictions have so-called “apology statutes,” which encourage error disclosure in return for immunity.
D. This is a case of medication, not medical, error.
E. Silence is golden.
Answer: B. A recent publication concluded, “If medical error were a disease, it would rank as the third leading cause of death in the United States.”1 This is the latest follow through on the original landmark report from the Institute of Medicine in 2000, which drew the public’s attention to the fact that medical errors were responsible for between 44,000 and 98,000 annual fatalities in the United States.
A medical error denotes a preventable adverse event, which in turn can be described as an injury caused by medical mismanagement rather than the underlying condition of the patient. It is more formally defined as “the failure of a planned action to be completed as intended or the use of a wrong plan to achieve an aim.”2 The term is not synonymous with medical negligence, which is a legal term of art encompassing four separate elements: duty, breach, causation, and damages.
The most common type of medical error is a medication error, which is any preventable event that may cause or lead to inappropriate medication use or patient harm while the medication is in the control of the health professional, patient, or consumer.
Medication errors account for 6.3%-30% of all malpractice claims, and a 1999 Texas case is an example.3 A 43-year-old Hispanic man with chest pain was prescribed the anti-angina drug Isordil (isosorbide dinitrate) by his cardiologist, to be taken four times a day in doses of 20 mg. The pharmacist misread the order as Plendil (felodipine), a calcium channel blocker for treatment of hypertension. This exceeded the drug’s top dose, and the patient suffered a heart attack and died several days later.
The cardiologist’s illegible prescription was the sole reason for the error, and his overall quality of care was not at issue. The jury returned a verdict for the plaintiff, awarding $450,000 to his estate: $225,000 from the cardiologist, and $225,000 from the pharmacist.
Many, but not all, jurisdictions now require some form of reporting of medical errors occurring in a hospital setting. States such as California and Florida mandate disclosure to patients. Pennsylvania actually requires hospitals to issue a written disclosure within 7 days of a serious event.
Most states have enacted “apology statutes” to encourage open discussions with patients and their families about adverse results. The apologies may cover expressions of regret, sympathy, and compassion, and they are barred from being presented to the jury should a trial ensue. However, an acknowledgment of fault remains admissible into evidence.
Typical is California’s Evidence Code 1160(a), which provides that only “the portions of statements or benevolent gestures expressing sympathy” are inadmissible against a treating physician. On the other hand, some states have chosen to exclude all disclosures, including admissions of fault. An example is Colorado’s Apology Statute (Colo. Rev. Stat. Ann. 13-25-135), which provides that “any and all statements, affirmations, gestures, or conduct expressing apology, fault, sympathy, commiseration, condolence, compassion, or a general sense of benevolence ... shall be inadmissible as evidence of an admission of liability or as evidence of an admission against interest.”
Ohio is one of only a few states whose apology statutes fail to clearly distinguish between the admissibility of a physician’s statement of sympathy and one acknowledging fault. R.C. 2317.43, enacted by the Ohio General Assembly in 2004, renders inadmissible “statements, affirmations, gestures, or conduct expressing apology, sympathy, commiseration, condolence, compassion, or a general sense of benevolence.”
In Davis v. Wooster Orthopaedics & Sports Medicine,4 the plaintiff alleged that Dr. Michael Knapic, an orthopedic surgeon, negligently performed a lumbar microdiscectomy, severing the patient’s common iliac artery. The surgeon allegedly then said to the patient’s husband, “It’s my fault. I take full responsibility.”
In a wrongful-death action, the plaintiff argued that the statute did not prohibit the use of statements of fault, responsibility, or liability as compared to statements of sympathy or condolence.
Ohio’s Ninth Appellate District concluded that the intent behind the apology statute was to protect pure expressions of sympathy but not admissions of fault. The court held that Dr. Knapic’s statements constituted an admission of liability that could be admitted into evidence. The jury awarded damages of $3 million.
More recently, the Ohio Supreme Court ruled that Dr. Randall Smith’s alleged confession regarding accidentally sectioning his patient’s common bile duct was properly excluded from evidence, even though the incident took place before the Ohio law went into effect.5
The patient had to be readmitted within 3 weeks for obstructive jaundice. After the doctor informed her that she would have to undergo additional surgery, she became very emotional. He reportedly took her hand, saying, “I take full responsibility for this.”
The Ohio Supreme Court ruled, “The trial court had determined that Dr. Smith was faced with a distressed patient who was upset and made a statement that was designed to comfort his patient. This is precisely the type of evidence that R.C. 2317.43 was designed to exclude as evidence of liability in a medical-malpractice case.”
Do disclosures and apologies work?
Both claim frequency and severity have diminished following the adoption of a humanistic risk management policy at the Lexington Veterans Affairs Medical Center since 1987.6 The protocol includes early injury review, steadfast maintenance of the relationship between the hospital and the patient, proactive disclosure to patients who have been injured because of accidents or medical negligence, and fair compensation for injuries.
Other institutions such as the University of Michigan have adopted “disclosure and offer” in place of “denial and defend” policies, but these have yet to achieve widespread use.
Many health care providers continue to heed the traditional legal advice to say and admit nothing, believing that it is unsettled whether an apology will influence any decision to sue the doctor. They argue that the odds of a lawsuit are low to begin with.
In the oft-cited Harvard study, there was only one malpractice claim for every 7.6 adverse events caused by negligence among the 3.7% of hospitalized patients who suffered significant iatrogenic injuries, typically from errors or negligence.7
Notwithstanding the controversy, the AMA has properly taken the moral high ground: It asserts that error disclosure is the right thing to do.
References
2. Institute of Medicine: To Err is Human: Building a Safer Health System. National Academy Press, Washington, 2000.
3. Estate of Velasquez v. Albertsons, Inc. et al., Civ. No. A-103-042 (Ector Cnty, TX 1999).
4. Davis v. Wooster Orthopaedics & Sports Medicine, Inc., 193 Ohio App.3d 581 (2011).
5. Estate of Johnson v. Randall Smith, Inc., 131 Ohio St.3d 1543 (2013).
6. N Engl J Med. 2010 Apr 15;362(15):1353-6.
7. N Engl J Med. 1991 Jul 25;325(4):245-51.
Dr. Tan is emeritus professor of medicine and a former adjunct professor of law at the University of Hawaii. This article is meant to be educational and does not constitute medical, ethical or legal advice. It is adapted from the author’s book, “Medical Malpractice: Understanding the Law, Managing the Risk” (2006). For additional information, readers may contact the author at [email protected].