User login
Official Newspaper of the American College of Surgeons
Moderate stenosis in coronary arteries grows severe over time
HOUSTON – Most nongrafted, moderately stenosed coronary arteries progress to severe stenosis or occlusion in the long term, results from a large, long-term study have shown.
“Not uncommonly, patients referred for coronary surgery have one or more coronary arteries with only moderate stenosis,” Joseph F. Sabik III, MD, said at the annual meeting of the Society of Thoracic Surgeons.
“There is controversy as to whether arteries with only moderate stenosis should be grafted during coronary surgery, and if it should be grafted, with what conduit?” For example, the Fractional Flow Reserve-Guided PCI versus Medical Therapy in Stable Coronary Disease study, known as FAME, suggests not intervening on moderate stenosis, since stenting non–ischemia-producing lesions led to worse outcomes (N Engl J Med. 2012 Sep 13;367:991-1001). However, Dr. Sabik, who chairs the department of surgery at University Hospitals Cleveland Medical Center, and his associates recently reported that grafting moderately stenosed coronary arteries during surgical revascularization is not harmful and can be beneficial by improving survival if an internal thoracic artery graft is used (J. Thoracic Cardiovasc Surg. 2016 Mar;151[3]:806-11).
In an effort to determine how grafting moderately stenosed coronary arteries influences native-vessel disease progression, and whether grafting may be protective from late ischemia, Dr. Sabik and his associates evaluated the medical records of 55,567 patients who underwent primary isolated coronary artery bypass graft (CABG) surgery at the Cleveland Clinic from 1972 to 2011. Of the 55,567 patients, 1,902 had a single coronary artery with angiographically moderate stenosis (defined as a narrowing of 50%-69%) and results of at least one postoperative angiogram available. Of these moderately stenosed coronary arteries (MSCAs), 488 were not grafted, 385 were internal thoracic artery (ITA)–grafted, and 1,028 were saphenous vein (SV)–grafted. At follow-up angiograms, information about disease progression was available for 488 nongrafted, 371 ITA-grafted, and 957 SV-grafted MSCAs, and patency information was available for 376 ITA and 1,016 SV grafts to these MSCAs. Grafts were considered patent if they were not occluded. Severe occlusion was defined as a narrowing of more than 70%.
The researchers found that at 1, 5, 10, and 15 years, native-vessel disease progressed from moderate to severe stenosis/occlusion in 32%, 52%, 66%, and 72% of nongrafted MSCAs, respectively; in 55%, 73%, 84%, and 87% of ITA-grafted MSCAs, and in 67%, 82%, 90%, and 92% of SV-grafted MSCAs. After Dr. Sabik and his associates adjusted for patient characteristics, disease progression in MSCAs was significantly higher with ITA and SV grafting, compared with nongrafting (odds ratios, 3.6 and 9.9, respectively). At 1, 5, 10, and 15 years, occlusion in grafts to MSCAs was 8%, 9%, 11%, and 15%, respectively, for ITA grafts and 13%, 32%, 46%, and 56% for SV grafts. At these same time points, protection from myocardial ischemia in ITA-grafted vs. nongrafted MSCAs was 29%, 47%, 59%, and 61%.
“Our opinion is you that shouldn’t ignore moderate lesions,” Dr. Sabik, surgeon-in-chief and vice president for surgical operations for the University Hospitals system, said in an interview at the meeting. “Although it may not help that patient over the next short period of time, over their lifespan it will. What works for intervention doesn’t necessarily mean it’s right for bypass surgery. If you have a vessel that’s only moderately stenosed you should at least consider grafting it, because moderate lesions progress over time. Bypassing it helps people live longer when you use an internal thoracic artery graft, because they are likely to remain patent. You always have to individualize the therapy, but the key is to use your grafts in the best way possible.”
Dr. Sabik disclosed that he has received research grants from Medtronic, Abbott Vascular, and Edwards Lifesciences.
HOUSTON – Most nongrafted, moderately stenosed coronary arteries progress to severe stenosis or occlusion in the long term, results from a large, long-term study have shown.
“Not uncommonly, patients referred for coronary surgery have one or more coronary arteries with only moderate stenosis,” Joseph F. Sabik III, MD, said at the annual meeting of the Society of Thoracic Surgeons.
“There is controversy as to whether arteries with only moderate stenosis should be grafted during coronary surgery, and if it should be grafted, with what conduit?” For example, the Fractional Flow Reserve-Guided PCI versus Medical Therapy in Stable Coronary Disease study, known as FAME, suggests not intervening on moderate stenosis, since stenting non–ischemia-producing lesions led to worse outcomes (N Engl J Med. 2012 Sep 13;367:991-1001). However, Dr. Sabik, who chairs the department of surgery at University Hospitals Cleveland Medical Center, and his associates recently reported that grafting moderately stenosed coronary arteries during surgical revascularization is not harmful and can be beneficial by improving survival if an internal thoracic artery graft is used (J. Thoracic Cardiovasc Surg. 2016 Mar;151[3]:806-11).
In an effort to determine how grafting moderately stenosed coronary arteries influences native-vessel disease progression, and whether grafting may be protective from late ischemia, Dr. Sabik and his associates evaluated the medical records of 55,567 patients who underwent primary isolated coronary artery bypass graft (CABG) surgery at the Cleveland Clinic from 1972 to 2011. Of the 55,567 patients, 1,902 had a single coronary artery with angiographically moderate stenosis (defined as a narrowing of 50%-69%) and results of at least one postoperative angiogram available. Of these moderately stenosed coronary arteries (MSCAs), 488 were not grafted, 385 were internal thoracic artery (ITA)–grafted, and 1,028 were saphenous vein (SV)–grafted. At follow-up angiograms, information about disease progression was available for 488 nongrafted, 371 ITA-grafted, and 957 SV-grafted MSCAs, and patency information was available for 376 ITA and 1,016 SV grafts to these MSCAs. Grafts were considered patent if they were not occluded. Severe occlusion was defined as a narrowing of more than 70%.
The researchers found that at 1, 5, 10, and 15 years, native-vessel disease progressed from moderate to severe stenosis/occlusion in 32%, 52%, 66%, and 72% of nongrafted MSCAs, respectively; in 55%, 73%, 84%, and 87% of ITA-grafted MSCAs, and in 67%, 82%, 90%, and 92% of SV-grafted MSCAs. After Dr. Sabik and his associates adjusted for patient characteristics, disease progression in MSCAs was significantly higher with ITA and SV grafting, compared with nongrafting (odds ratios, 3.6 and 9.9, respectively). At 1, 5, 10, and 15 years, occlusion in grafts to MSCAs was 8%, 9%, 11%, and 15%, respectively, for ITA grafts and 13%, 32%, 46%, and 56% for SV grafts. At these same time points, protection from myocardial ischemia in ITA-grafted vs. nongrafted MSCAs was 29%, 47%, 59%, and 61%.
“Our opinion is you that shouldn’t ignore moderate lesions,” Dr. Sabik, surgeon-in-chief and vice president for surgical operations for the University Hospitals system, said in an interview at the meeting. “Although it may not help that patient over the next short period of time, over their lifespan it will. What works for intervention doesn’t necessarily mean it’s right for bypass surgery. If you have a vessel that’s only moderately stenosed you should at least consider grafting it, because moderate lesions progress over time. Bypassing it helps people live longer when you use an internal thoracic artery graft, because they are likely to remain patent. You always have to individualize the therapy, but the key is to use your grafts in the best way possible.”
Dr. Sabik disclosed that he has received research grants from Medtronic, Abbott Vascular, and Edwards Lifesciences.
HOUSTON – Most nongrafted, moderately stenosed coronary arteries progress to severe stenosis or occlusion in the long term, results from a large, long-term study have shown.
“Not uncommonly, patients referred for coronary surgery have one or more coronary arteries with only moderate stenosis,” Joseph F. Sabik III, MD, said at the annual meeting of the Society of Thoracic Surgeons.
“There is controversy as to whether arteries with only moderate stenosis should be grafted during coronary surgery, and if it should be grafted, with what conduit?” For example, the Fractional Flow Reserve-Guided PCI versus Medical Therapy in Stable Coronary Disease study, known as FAME, suggests not intervening on moderate stenosis, since stenting non–ischemia-producing lesions led to worse outcomes (N Engl J Med. 2012 Sep 13;367:991-1001). However, Dr. Sabik, who chairs the department of surgery at University Hospitals Cleveland Medical Center, and his associates recently reported that grafting moderately stenosed coronary arteries during surgical revascularization is not harmful and can be beneficial by improving survival if an internal thoracic artery graft is used (J. Thoracic Cardiovasc Surg. 2016 Mar;151[3]:806-11).
In an effort to determine how grafting moderately stenosed coronary arteries influences native-vessel disease progression, and whether grafting may be protective from late ischemia, Dr. Sabik and his associates evaluated the medical records of 55,567 patients who underwent primary isolated coronary artery bypass graft (CABG) surgery at the Cleveland Clinic from 1972 to 2011. Of the 55,567 patients, 1,902 had a single coronary artery with angiographically moderate stenosis (defined as a narrowing of 50%-69%) and results of at least one postoperative angiogram available. Of these moderately stenosed coronary arteries (MSCAs), 488 were not grafted, 385 were internal thoracic artery (ITA)–grafted, and 1,028 were saphenous vein (SV)–grafted. At follow-up angiograms, information about disease progression was available for 488 nongrafted, 371 ITA-grafted, and 957 SV-grafted MSCAs, and patency information was available for 376 ITA and 1,016 SV grafts to these MSCAs. Grafts were considered patent if they were not occluded. Severe occlusion was defined as a narrowing of more than 70%.
The researchers found that at 1, 5, 10, and 15 years, native-vessel disease progressed from moderate to severe stenosis/occlusion in 32%, 52%, 66%, and 72% of nongrafted MSCAs, respectively; in 55%, 73%, 84%, and 87% of ITA-grafted MSCAs, and in 67%, 82%, 90%, and 92% of SV-grafted MSCAs. After Dr. Sabik and his associates adjusted for patient characteristics, disease progression in MSCAs was significantly higher with ITA and SV grafting, compared with nongrafting (odds ratios, 3.6 and 9.9, respectively). At 1, 5, 10, and 15 years, occlusion in grafts to MSCAs was 8%, 9%, 11%, and 15%, respectively, for ITA grafts and 13%, 32%, 46%, and 56% for SV grafts. At these same time points, protection from myocardial ischemia in ITA-grafted vs. nongrafted MSCAs was 29%, 47%, 59%, and 61%.
“Our opinion is you that shouldn’t ignore moderate lesions,” Dr. Sabik, surgeon-in-chief and vice president for surgical operations for the University Hospitals system, said in an interview at the meeting. “Although it may not help that patient over the next short period of time, over their lifespan it will. What works for intervention doesn’t necessarily mean it’s right for bypass surgery. If you have a vessel that’s only moderately stenosed you should at least consider grafting it, because moderate lesions progress over time. Bypassing it helps people live longer when you use an internal thoracic artery graft, because they are likely to remain patent. You always have to individualize the therapy, but the key is to use your grafts in the best way possible.”
Dr. Sabik disclosed that he has received research grants from Medtronic, Abbott Vascular, and Edwards Lifesciences.
AT THE STS ANNUAL MEETING
Key clinical point:
Major finding: At 15 years, native-vessel disease progressed from moderate to severe stenosis/occlusion in 72% of nongrafted coronary arteries, in 87% of internal thoracic artery–grafted arteries, and in 92% of saphenous vein–grafted moderately stenosed coronary arteries.
Data source: An analysis of medical records from 55,567 patients who underwent primary isolated CABG at the Cleveland Clinic from 1972 to 2011.
Disclosures: Dr. Sabik disclosed that he has received research grants from Medtronic, Abbott Vascular, and Edwards Lifesciences.
IgG4-related disease can strike any organ system
SNOWMASS, COLO. – Progress in the understanding and treatment of immunoglobulin G4–related disease is occurring “at lightning speed,” John H. Stone, MD, said at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
Eight or nine years ago no one had heard of immunoglobulin G4–related disease (IgG4-RD). Today, because of the broad swathe the disease cuts, it’s a hot research topic in every subspecialty of medicine as well as surgery, pathology, and radiology.
This new understanding of IgG4-RD, he added, is opening the door to novel treatments.
“This is not a new disease. It was there when we were all in medical school, and for hundreds of years before that. But it’s really only in the last decade that we have come to understand that the disease can affect literally every organ system in the body with syndromes that we once thought were isolated organ-specific syndromes but we now recognize are part of a multiorgan disease currently called IgG4-related disease,” the rheumatologist said.
IgG4-RD is an immune-mediated fibroinflammatory condition characterized histopathologically by three hallmark features in involved tissue: obliterative phlebitis, storiform fibrosis, and a dense lymphoplasmacytic infiltrate.
Clinically, IgG4-RD often presents as a mass lesion that can affect any organ.
“I have many patients who’ve undergone modified Whipple procedures because they were thought to have adenocarcinoma of the pancreas,” according to Dr. Stone.
Other common presentations include Riedel’s thyroiditis, autoimmune pancreatitis, sclerosing cholangitis, sialadenitis, dacryoadenitis, periaortitis, an eosinophilic rash, and pseudotumor of the lung, lymph nodes, or orbits.
“Retroperitoneal fibrosis is a common and underappreciated manifestation. It may be the most common subsyndrome associated with IgG4-related disease,” he observed.
Another common presentation involves atopic disease – asthma, allergic rhinitis, eczema, eosinophilia, nasal polyps – developing out of the blue in middle age or later life. This observation led some other investigators to posit that IgG4-RD is a T-helper type 2–driven disease, an assertion debunked by Dr. Stone and coworkers (Allergy. 2014 Feb;69[2]:269-72).
Dr. Stone and his coinvestigators have published the largest series of patients with biopsy-proven IgG4-RD reported to date (Arthritis Rheumatol. 2015 Sep; 67[9]:2466-75). The average age at disease onset was 50 years. Of note, multiorgan involvement was the norm: 24% of patients had two organs involved, and 38% had three or more.
Analysis of this large patient series has led Dr. Stone to a surprising conclusion about the nature of IgG4-RD: “We have greatly overemphasized the importance of IgG4 in this condition,” he asserted.
Indeed, a mere 51% of the patients with clinically active untreated IgG4-RD in his series had an elevated serum IgG level. Dr. Stone characterized IgG4 as “kind of a wimpy antibody” incapable of driving the disease process because it is a noninflammatory immunoglobulin. This has led to speculation that IgG4 functions as what he termed an “antigen sink,” attempting to bind antigen at sites of inflammation.
But while an elevated serum IgG4 is of limited utility for diagnostic purposes, Dr. Stone and coworkers have demonstrated that it is of value as a predictor of relapse. Among patients with a treatment-induced remission, those in the top quartile in terms of baseline pretreatment serum IgG4 were 6.2-fold more likely to relapse (Rheumatology [Oxford]. 2016 Jun;55[6]:1000-8).
“This is a very useful marker for patients who are going to need chronic ongoing therapy. The notion of putting such patients on steroids for months and years is not appealing,” he said.
Levels of circulating plasmablasts as measured by peripheral blood flow cytometry, especially IgG4-positive plasmablasts, have proven much more helpful than serum IgG4 levels as a diagnostic tool, a reliable biomarker of disease activity, and a therapeutic target. Levels of these short-lived CD19+CD38+CD27+ plasmablasts are enormously elevated independent of serum IgG4 in patients with active IgG4-RD.
“One of the questions I’m most often asked is whether IgG4-related disease is a premalignant condition. My answer is no. The plasmablast expansion is oligoclonal, not polyclonal,” Dr. Stone continued.
He described IgG4-RD as “a continuous dance between T cells and B cells.” The latest thinking regarding pathogenesis is that type 2 T follicular helper cells activate B cells, which become memory B cells or plasmablasts. These activated B cells and plasmablasts present antigen to CD4+ cytotoxic T cells at sites of disease. Dr. Stone and his coinvestigators recently identified these CD4+ cytotoxic T cells as a novel population of clonally expanded T cells with SLAMF7 as a surface marker. The cells secrete interferon-gamma, interleukin-1, and transforming growth factor-beta, all of which are capable of driving the intense fibrosis characteristic of IgG4-RD. In addition, these CD4+ cytotoxic T cells secrete granzyme B and perforin, previously thought to be released mainly by natural killer T cells.
Joint American College of Rheumatology/European League Against Rheumatism classification criteria for the disease are expected to be finalized this winter at the Third International Symposium on IgG4-Related Diseases.
Dr. Stone reported receiving IgG4-RD–related research funding from and serving as a consultant to Genentech and Xencor.
SNOWMASS, COLO. – Progress in the understanding and treatment of immunoglobulin G4–related disease is occurring “at lightning speed,” John H. Stone, MD, said at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
Eight or nine years ago no one had heard of immunoglobulin G4–related disease (IgG4-RD). Today, because of the broad swathe the disease cuts, it’s a hot research topic in every subspecialty of medicine as well as surgery, pathology, and radiology.
This new understanding of IgG4-RD, he added, is opening the door to novel treatments.
“This is not a new disease. It was there when we were all in medical school, and for hundreds of years before that. But it’s really only in the last decade that we have come to understand that the disease can affect literally every organ system in the body with syndromes that we once thought were isolated organ-specific syndromes but we now recognize are part of a multiorgan disease currently called IgG4-related disease,” the rheumatologist said.
IgG4-RD is an immune-mediated fibroinflammatory condition characterized histopathologically by three hallmark features in involved tissue: obliterative phlebitis, storiform fibrosis, and a dense lymphoplasmacytic infiltrate.
Clinically, IgG4-RD often presents as a mass lesion that can affect any organ.
“I have many patients who’ve undergone modified Whipple procedures because they were thought to have adenocarcinoma of the pancreas,” according to Dr. Stone.
Other common presentations include Riedel’s thyroiditis, autoimmune pancreatitis, sclerosing cholangitis, sialadenitis, dacryoadenitis, periaortitis, an eosinophilic rash, and pseudotumor of the lung, lymph nodes, or orbits.
“Retroperitoneal fibrosis is a common and underappreciated manifestation. It may be the most common subsyndrome associated with IgG4-related disease,” he observed.
Another common presentation involves atopic disease – asthma, allergic rhinitis, eczema, eosinophilia, nasal polyps – developing out of the blue in middle age or later life. This observation led some other investigators to posit that IgG4-RD is a T-helper type 2–driven disease, an assertion debunked by Dr. Stone and coworkers (Allergy. 2014 Feb;69[2]:269-72).
Dr. Stone and his coinvestigators have published the largest series of patients with biopsy-proven IgG4-RD reported to date (Arthritis Rheumatol. 2015 Sep; 67[9]:2466-75). The average age at disease onset was 50 years. Of note, multiorgan involvement was the norm: 24% of patients had two organs involved, and 38% had three or more.
Analysis of this large patient series has led Dr. Stone to a surprising conclusion about the nature of IgG4-RD: “We have greatly overemphasized the importance of IgG4 in this condition,” he asserted.
Indeed, a mere 51% of the patients with clinically active untreated IgG4-RD in his series had an elevated serum IgG level. Dr. Stone characterized IgG4 as “kind of a wimpy antibody” incapable of driving the disease process because it is a noninflammatory immunoglobulin. This has led to speculation that IgG4 functions as what he termed an “antigen sink,” attempting to bind antigen at sites of inflammation.
But while an elevated serum IgG4 is of limited utility for diagnostic purposes, Dr. Stone and coworkers have demonstrated that it is of value as a predictor of relapse. Among patients with a treatment-induced remission, those in the top quartile in terms of baseline pretreatment serum IgG4 were 6.2-fold more likely to relapse (Rheumatology [Oxford]. 2016 Jun;55[6]:1000-8).
“This is a very useful marker for patients who are going to need chronic ongoing therapy. The notion of putting such patients on steroids for months and years is not appealing,” he said.
Levels of circulating plasmablasts as measured by peripheral blood flow cytometry, especially IgG4-positive plasmablasts, have proven much more helpful than serum IgG4 levels as a diagnostic tool, a reliable biomarker of disease activity, and a therapeutic target. Levels of these short-lived CD19+CD38+CD27+ plasmablasts are enormously elevated independent of serum IgG4 in patients with active IgG4-RD.
“One of the questions I’m most often asked is whether IgG4-related disease is a premalignant condition. My answer is no. The plasmablast expansion is oligoclonal, not polyclonal,” Dr. Stone continued.
He described IgG4-RD as “a continuous dance between T cells and B cells.” The latest thinking regarding pathogenesis is that type 2 T follicular helper cells activate B cells, which become memory B cells or plasmablasts. These activated B cells and plasmablasts present antigen to CD4+ cytotoxic T cells at sites of disease. Dr. Stone and his coinvestigators recently identified these CD4+ cytotoxic T cells as a novel population of clonally expanded T cells with SLAMF7 as a surface marker. The cells secrete interferon-gamma, interleukin-1, and transforming growth factor-beta, all of which are capable of driving the intense fibrosis characteristic of IgG4-RD. In addition, these CD4+ cytotoxic T cells secrete granzyme B and perforin, previously thought to be released mainly by natural killer T cells.
Joint American College of Rheumatology/European League Against Rheumatism classification criteria for the disease are expected to be finalized this winter at the Third International Symposium on IgG4-Related Diseases.
Dr. Stone reported receiving IgG4-RD–related research funding from and serving as a consultant to Genentech and Xencor.
SNOWMASS, COLO. – Progress in the understanding and treatment of immunoglobulin G4–related disease is occurring “at lightning speed,” John H. Stone, MD, said at the Winter Rheumatology Symposium sponsored by the American College of Rheumatology.
Eight or nine years ago no one had heard of immunoglobulin G4–related disease (IgG4-RD). Today, because of the broad swathe the disease cuts, it’s a hot research topic in every subspecialty of medicine as well as surgery, pathology, and radiology.
This new understanding of IgG4-RD, he added, is opening the door to novel treatments.
“This is not a new disease. It was there when we were all in medical school, and for hundreds of years before that. But it’s really only in the last decade that we have come to understand that the disease can affect literally every organ system in the body with syndromes that we once thought were isolated organ-specific syndromes but we now recognize are part of a multiorgan disease currently called IgG4-related disease,” the rheumatologist said.
IgG4-RD is an immune-mediated fibroinflammatory condition characterized histopathologically by three hallmark features in involved tissue: obliterative phlebitis, storiform fibrosis, and a dense lymphoplasmacytic infiltrate.
Clinically, IgG4-RD often presents as a mass lesion that can affect any organ.
“I have many patients who’ve undergone modified Whipple procedures because they were thought to have adenocarcinoma of the pancreas,” according to Dr. Stone.
Other common presentations include Riedel’s thyroiditis, autoimmune pancreatitis, sclerosing cholangitis, sialadenitis, dacryoadenitis, periaortitis, an eosinophilic rash, and pseudotumor of the lung, lymph nodes, or orbits.
“Retroperitoneal fibrosis is a common and underappreciated manifestation. It may be the most common subsyndrome associated with IgG4-related disease,” he observed.
Another common presentation involves atopic disease – asthma, allergic rhinitis, eczema, eosinophilia, nasal polyps – developing out of the blue in middle age or later life. This observation led some other investigators to posit that IgG4-RD is a T-helper type 2–driven disease, an assertion debunked by Dr. Stone and coworkers (Allergy. 2014 Feb;69[2]:269-72).
Dr. Stone and his coinvestigators have published the largest series of patients with biopsy-proven IgG4-RD reported to date (Arthritis Rheumatol. 2015 Sep; 67[9]:2466-75). The average age at disease onset was 50 years. Of note, multiorgan involvement was the norm: 24% of patients had two organs involved, and 38% had three or more.
Analysis of this large patient series has led Dr. Stone to a surprising conclusion about the nature of IgG4-RD: “We have greatly overemphasized the importance of IgG4 in this condition,” he asserted.
Indeed, a mere 51% of the patients with clinically active untreated IgG4-RD in his series had an elevated serum IgG level. Dr. Stone characterized IgG4 as “kind of a wimpy antibody” incapable of driving the disease process because it is a noninflammatory immunoglobulin. This has led to speculation that IgG4 functions as what he termed an “antigen sink,” attempting to bind antigen at sites of inflammation.
But while an elevated serum IgG4 is of limited utility for diagnostic purposes, Dr. Stone and coworkers have demonstrated that it is of value as a predictor of relapse. Among patients with a treatment-induced remission, those in the top quartile in terms of baseline pretreatment serum IgG4 were 6.2-fold more likely to relapse (Rheumatology [Oxford]. 2016 Jun;55[6]:1000-8).
“This is a very useful marker for patients who are going to need chronic ongoing therapy. The notion of putting such patients on steroids for months and years is not appealing,” he said.
Levels of circulating plasmablasts as measured by peripheral blood flow cytometry, especially IgG4-positive plasmablasts, have proven much more helpful than serum IgG4 levels as a diagnostic tool, a reliable biomarker of disease activity, and a therapeutic target. Levels of these short-lived CD19+CD38+CD27+ plasmablasts are enormously elevated independent of serum IgG4 in patients with active IgG4-RD.
“One of the questions I’m most often asked is whether IgG4-related disease is a premalignant condition. My answer is no. The plasmablast expansion is oligoclonal, not polyclonal,” Dr. Stone continued.
He described IgG4-RD as “a continuous dance between T cells and B cells.” The latest thinking regarding pathogenesis is that type 2 T follicular helper cells activate B cells, which become memory B cells or plasmablasts. These activated B cells and plasmablasts present antigen to CD4+ cytotoxic T cells at sites of disease. Dr. Stone and his coinvestigators recently identified these CD4+ cytotoxic T cells as a novel population of clonally expanded T cells with SLAMF7 as a surface marker. The cells secrete interferon-gamma, interleukin-1, and transforming growth factor-beta, all of which are capable of driving the intense fibrosis characteristic of IgG4-RD. In addition, these CD4+ cytotoxic T cells secrete granzyme B and perforin, previously thought to be released mainly by natural killer T cells.
Joint American College of Rheumatology/European League Against Rheumatism classification criteria for the disease are expected to be finalized this winter at the Third International Symposium on IgG4-Related Diseases.
Dr. Stone reported receiving IgG4-RD–related research funding from and serving as a consultant to Genentech and Xencor.
EXPERT ANALYSIS FROM THE WINTER RHEUMATOLOGY SYMPOSIUM
Federal judge blocks merger between Anthem and Cigna
A federal district court judge has blocked health insurer Anthem from acquiring Cigna, ruling the megamerger would violate antitrust laws and stifle competition.
The decision comes weeks after another U.S. district court judge barred a merger between health insurance giants Aetna and Humana.
“This merger would have stifled competition, harming consumers by increasing health insurance prices and slowing innovation aimed at lowering the costs of health care,” Acting Assistant Attorney General Brent Snyder said in a statement.
Anthem intends to appeal the decision, said Joseph R. Swedish, Anthem’s chair, president, and chief executive officer.
“Anthem is significantly disappointed by the decision, as combining Anthem and Cigna would positively impact the health and well-being of millions of Americans – saving them more than $2 billion in medical costs annually,” Mr. Swedish said in a statement.“If not overturned, the consequences of the decision are far reaching and will hurt American consumers by limiting their access to high-quality affordable care, slowing the industry’s shift to value-based care and improved outcomes for patients, and restricting innovation, which is critical to meeting the evolving needs of health care consumers.”
In a statement, a Cigna official said the company intends to carefully review the opinion and evaluate its options in accordance with the merger agreement.
“Cigna remains focused on helping to improve health care by delivering value to our customers and clients and expanding our business around the world,” the statement said.
The DOJ, 11 states, and the District of Columbia sued Anthem and Cigna in July over their proposed $54 billion consolidation in what would have been the largest merger in history.
The DOJ argued the merger would substantially harm competition and negatively impact the entire insurance industry if allowed to proceed. The consolidation would enhance Anthem’s power to profit at the expense of consumers and the doctors and hospitals who provide their medical care, DOJ attorneys said in their complaint.
Anthem and Cigna argued the proposed acquisition was “procompetitive,” and that the merger would result in efficiencies that would directly benefit consumers via greater access to affordable health care. The benefits of the merger outweigh any alleged anticompetitive effects, according to Anthem.
A trial before Judge Amy Berman Jackson of the U.S. District Court for the District of Columbia ran from November through January.
Judge Berman’s opinion is temporarily under seal to allow parties to review for confidentiality.
The ruling is the second victory for the DOJ in as many weeks. In a Jan. 23 decision, Judge John D. Bates of the U.S. District Court for the District of Columbia denied Aetna’s $37 billion plan to purchase Humana, following a month-long trial that began in early December. Judge Bates ruled the consolidation would violate antitrust laws and reduce competition.
[email protected]
On Twitter @legal_med
A federal district court judge has blocked health insurer Anthem from acquiring Cigna, ruling the megamerger would violate antitrust laws and stifle competition.
The decision comes weeks after another U.S. district court judge barred a merger between health insurance giants Aetna and Humana.
“This merger would have stifled competition, harming consumers by increasing health insurance prices and slowing innovation aimed at lowering the costs of health care,” Acting Assistant Attorney General Brent Snyder said in a statement.
Anthem intends to appeal the decision, said Joseph R. Swedish, Anthem’s chair, president, and chief executive officer.
“Anthem is significantly disappointed by the decision, as combining Anthem and Cigna would positively impact the health and well-being of millions of Americans – saving them more than $2 billion in medical costs annually,” Mr. Swedish said in a statement.“If not overturned, the consequences of the decision are far reaching and will hurt American consumers by limiting their access to high-quality affordable care, slowing the industry’s shift to value-based care and improved outcomes for patients, and restricting innovation, which is critical to meeting the evolving needs of health care consumers.”
In a statement, a Cigna official said the company intends to carefully review the opinion and evaluate its options in accordance with the merger agreement.
“Cigna remains focused on helping to improve health care by delivering value to our customers and clients and expanding our business around the world,” the statement said.
The DOJ, 11 states, and the District of Columbia sued Anthem and Cigna in July over their proposed $54 billion consolidation in what would have been the largest merger in history.
The DOJ argued the merger would substantially harm competition and negatively impact the entire insurance industry if allowed to proceed. The consolidation would enhance Anthem’s power to profit at the expense of consumers and the doctors and hospitals who provide their medical care, DOJ attorneys said in their complaint.
Anthem and Cigna argued the proposed acquisition was “procompetitive,” and that the merger would result in efficiencies that would directly benefit consumers via greater access to affordable health care. The benefits of the merger outweigh any alleged anticompetitive effects, according to Anthem.
A trial before Judge Amy Berman Jackson of the U.S. District Court for the District of Columbia ran from November through January.
Judge Berman’s opinion is temporarily under seal to allow parties to review for confidentiality.
The ruling is the second victory for the DOJ in as many weeks. In a Jan. 23 decision, Judge John D. Bates of the U.S. District Court for the District of Columbia denied Aetna’s $37 billion plan to purchase Humana, following a month-long trial that began in early December. Judge Bates ruled the consolidation would violate antitrust laws and reduce competition.
[email protected]
On Twitter @legal_med
A federal district court judge has blocked health insurer Anthem from acquiring Cigna, ruling the megamerger would violate antitrust laws and stifle competition.
The decision comes weeks after another U.S. district court judge barred a merger between health insurance giants Aetna and Humana.
“This merger would have stifled competition, harming consumers by increasing health insurance prices and slowing innovation aimed at lowering the costs of health care,” Acting Assistant Attorney General Brent Snyder said in a statement.
Anthem intends to appeal the decision, said Joseph R. Swedish, Anthem’s chair, president, and chief executive officer.
“Anthem is significantly disappointed by the decision, as combining Anthem and Cigna would positively impact the health and well-being of millions of Americans – saving them more than $2 billion in medical costs annually,” Mr. Swedish said in a statement.“If not overturned, the consequences of the decision are far reaching and will hurt American consumers by limiting their access to high-quality affordable care, slowing the industry’s shift to value-based care and improved outcomes for patients, and restricting innovation, which is critical to meeting the evolving needs of health care consumers.”
In a statement, a Cigna official said the company intends to carefully review the opinion and evaluate its options in accordance with the merger agreement.
“Cigna remains focused on helping to improve health care by delivering value to our customers and clients and expanding our business around the world,” the statement said.
The DOJ, 11 states, and the District of Columbia sued Anthem and Cigna in July over their proposed $54 billion consolidation in what would have been the largest merger in history.
The DOJ argued the merger would substantially harm competition and negatively impact the entire insurance industry if allowed to proceed. The consolidation would enhance Anthem’s power to profit at the expense of consumers and the doctors and hospitals who provide their medical care, DOJ attorneys said in their complaint.
Anthem and Cigna argued the proposed acquisition was “procompetitive,” and that the merger would result in efficiencies that would directly benefit consumers via greater access to affordable health care. The benefits of the merger outweigh any alleged anticompetitive effects, according to Anthem.
A trial before Judge Amy Berman Jackson of the U.S. District Court for the District of Columbia ran from November through January.
Judge Berman’s opinion is temporarily under seal to allow parties to review for confidentiality.
The ruling is the second victory for the DOJ in as many weeks. In a Jan. 23 decision, Judge John D. Bates of the U.S. District Court for the District of Columbia denied Aetna’s $37 billion plan to purchase Humana, following a month-long trial that began in early December. Judge Bates ruled the consolidation would violate antitrust laws and reduce competition.
[email protected]
On Twitter @legal_med
Increase in U.S. drug spending slowed in 2016
Prescription drug spending for those with employer-sponsored insurance increased by 3.8% in 2016, compared with a rise of 5.2% in 2015, according to pharmacy benefits manager Express Scripts.
Commercial plans managed by Express Scripts saw the cost of prescription drugs rise by 2.5% per person, while utilization was up by 1.3%, which adds up to the 3.8% overall increase. That represents a 27% drop from 2015, when drug use rose 2.0% and costs went up by 3.2%, Express Scripts said in its “2016 Drug Trend Report.”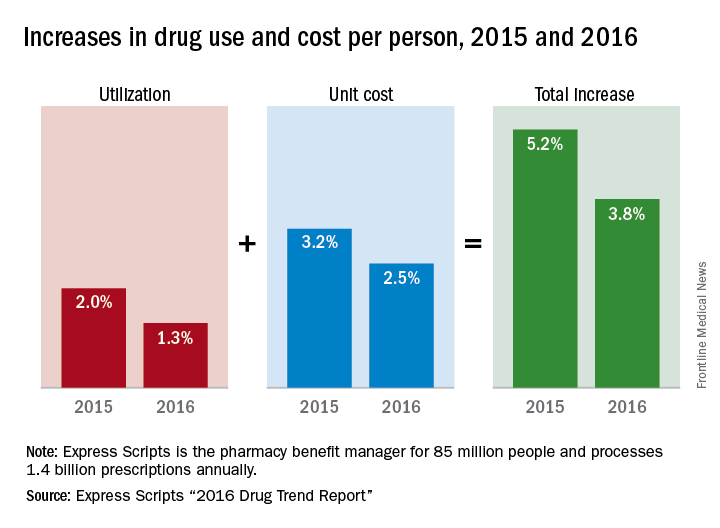
For a second consecutive year, patients of pharmacy plans managed by Express Scripts had a smaller out-of-pocket share of their total pharmacy costs, 14.6%, compared with 14.8% in 2015. That smaller share did represent an increase from $11.25 in 2015 to $11.34 in 2016, however, as the adjusted total cost per prescription rose from $75.85 to $77.84, Express Scripts said.
The company is the largest pharmacy benefits manager in the country, according to its corporate overview, processing 1.4 billion prescriptions annually for 85 million people.
Prescription drug spending for those with employer-sponsored insurance increased by 3.8% in 2016, compared with a rise of 5.2% in 2015, according to pharmacy benefits manager Express Scripts.
Commercial plans managed by Express Scripts saw the cost of prescription drugs rise by 2.5% per person, while utilization was up by 1.3%, which adds up to the 3.8% overall increase. That represents a 27% drop from 2015, when drug use rose 2.0% and costs went up by 3.2%, Express Scripts said in its “2016 Drug Trend Report.”
For a second consecutive year, patients of pharmacy plans managed by Express Scripts had a smaller out-of-pocket share of their total pharmacy costs, 14.6%, compared with 14.8% in 2015. That smaller share did represent an increase from $11.25 in 2015 to $11.34 in 2016, however, as the adjusted total cost per prescription rose from $75.85 to $77.84, Express Scripts said.
The company is the largest pharmacy benefits manager in the country, according to its corporate overview, processing 1.4 billion prescriptions annually for 85 million people.
Prescription drug spending for those with employer-sponsored insurance increased by 3.8% in 2016, compared with a rise of 5.2% in 2015, according to pharmacy benefits manager Express Scripts.
Commercial plans managed by Express Scripts saw the cost of prescription drugs rise by 2.5% per person, while utilization was up by 1.3%, which adds up to the 3.8% overall increase. That represents a 27% drop from 2015, when drug use rose 2.0% and costs went up by 3.2%, Express Scripts said in its “2016 Drug Trend Report.”
For a second consecutive year, patients of pharmacy plans managed by Express Scripts had a smaller out-of-pocket share of their total pharmacy costs, 14.6%, compared with 14.8% in 2015. That smaller share did represent an increase from $11.25 in 2015 to $11.34 in 2016, however, as the adjusted total cost per prescription rose from $75.85 to $77.84, Express Scripts said.
The company is the largest pharmacy benefits manager in the country, according to its corporate overview, processing 1.4 billion prescriptions annually for 85 million people.
Complicated appendicitis outcomes worse with delayed surgery
LAS VEGAS – Contrary to what some recent studies suggest, patients with complicated appendicitis may benefit from immediate surgery, with shorter hospital stays and fewer postoperative complications.
According to findings from a large database review, delaying the surgery for a complicated case is likely to result in worse patient outcomes, Matthew Symer, MD, said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress.
There are no firm guidelines about the timing of surgery for complicated appendicitis, he said. “There is in fact some controversy about the timing of surgery,” with studies coming to conflicting conclusions about the benefits and risks of both immediate and delayed treatment. “We theorized that the potential morbidity of operating at the height of the inflammatory process would be less than the risk of complications associated with delay,” he said.
To investigate the question, Dr. Symer and his colleagues queried the New York Statewide Planning and Research Cooperative Database, which contains information on all hospital admissions with an ICD-9 code on any patient covered by any payer in the state. Each patient has a unique identifier that allows tracking over time and across facilities.
From 2000 to 2013, the investigators identified 38,840 patients who presented with complicated appendicitis, defined as a perforation. Of these, 31,167 had an appendectomy within 1 year of the index admission. These patients were separated into two groups: those who had surgery within 48 hours of the index admission (28,015) and those who had later surgery (3,152).
The delayed surgery group was further parsed into three: those who had surgery during the index admission, but at least 48 hours after admission (51%); those who had an appendectomy at a subsequent urgent admission (23%); and those who had an elective interval appendectomy sometime within that year (26%).
In comparing the early vs. late surgery groups overall, Dr. Symer noticed some significant initial differences. Patients in the early surgery group were significantly younger (48 vs. 53 years), more likely to be male (55% vs. 47%), white (70% vs. 64%), and to have private insurance (53% vs. 45%).
Comorbidities were more common among the delayed surgery group. These included chronic obstructive pulmonary disease, renal failure, coronary artery disease, hypertension, diabetes, and congestive heart failure. Delayed-surgery patients were more likely to be treated at high-volume hospitals (45% vs. 34%).
Abscess was more common among the delayed surgery group (72% vs. 51%). Their median length of stay was significantly longer (9 vs. 5 days).
Delayed-surgery patients experienced significantly more iatrogenic complications (4% vs. 2%), and more urinary and wound complications. Overall, two or more complications occurred in 23% of the delayed surgery group and 14% of the early surgery group. The readmission rate was higher (28% vs. 18%). Significantly more in the delayed group reached the 75th percentile in hospital charges (62% vs. 26%).
In a multivariate regression analysis, patients with delayed surgery were more likely to experience a prolonged length of stay (odds ratio, 6); high hospital charges (OR, 4.8), iatrogenic complications (OR, 1.9), any complications (OR, 1.5) and readmission (OR, 1.5).
These findings were largely recapitulated when Dr. Symer broke the delayed group down into the three subgroups: patients who had surgery late in the index admission, patients who had an urgent later appendectomy, and patients who had a later elective procedure.
“All of these relationships held up, with patients who delayed surgery having worse overall complications, whether iatrogenic or any complications, more readmissions, and a longer stay in the hospital,” Dr. Symer said.
He had no financial disclosures.
[email protected]
On Twitter @Alz_Gal
LAS VEGAS – Contrary to what some recent studies suggest, patients with complicated appendicitis may benefit from immediate surgery, with shorter hospital stays and fewer postoperative complications.
According to findings from a large database review, delaying the surgery for a complicated case is likely to result in worse patient outcomes, Matthew Symer, MD, said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress.
There are no firm guidelines about the timing of surgery for complicated appendicitis, he said. “There is in fact some controversy about the timing of surgery,” with studies coming to conflicting conclusions about the benefits and risks of both immediate and delayed treatment. “We theorized that the potential morbidity of operating at the height of the inflammatory process would be less than the risk of complications associated with delay,” he said.
To investigate the question, Dr. Symer and his colleagues queried the New York Statewide Planning and Research Cooperative Database, which contains information on all hospital admissions with an ICD-9 code on any patient covered by any payer in the state. Each patient has a unique identifier that allows tracking over time and across facilities.
From 2000 to 2013, the investigators identified 38,840 patients who presented with complicated appendicitis, defined as a perforation. Of these, 31,167 had an appendectomy within 1 year of the index admission. These patients were separated into two groups: those who had surgery within 48 hours of the index admission (28,015) and those who had later surgery (3,152).
The delayed surgery group was further parsed into three: those who had surgery during the index admission, but at least 48 hours after admission (51%); those who had an appendectomy at a subsequent urgent admission (23%); and those who had an elective interval appendectomy sometime within that year (26%).
In comparing the early vs. late surgery groups overall, Dr. Symer noticed some significant initial differences. Patients in the early surgery group were significantly younger (48 vs. 53 years), more likely to be male (55% vs. 47%), white (70% vs. 64%), and to have private insurance (53% vs. 45%).
Comorbidities were more common among the delayed surgery group. These included chronic obstructive pulmonary disease, renal failure, coronary artery disease, hypertension, diabetes, and congestive heart failure. Delayed-surgery patients were more likely to be treated at high-volume hospitals (45% vs. 34%).
Abscess was more common among the delayed surgery group (72% vs. 51%). Their median length of stay was significantly longer (9 vs. 5 days).
Delayed-surgery patients experienced significantly more iatrogenic complications (4% vs. 2%), and more urinary and wound complications. Overall, two or more complications occurred in 23% of the delayed surgery group and 14% of the early surgery group. The readmission rate was higher (28% vs. 18%). Significantly more in the delayed group reached the 75th percentile in hospital charges (62% vs. 26%).
In a multivariate regression analysis, patients with delayed surgery were more likely to experience a prolonged length of stay (odds ratio, 6); high hospital charges (OR, 4.8), iatrogenic complications (OR, 1.9), any complications (OR, 1.5) and readmission (OR, 1.5).
These findings were largely recapitulated when Dr. Symer broke the delayed group down into the three subgroups: patients who had surgery late in the index admission, patients who had an urgent later appendectomy, and patients who had a later elective procedure.
“All of these relationships held up, with patients who delayed surgery having worse overall complications, whether iatrogenic or any complications, more readmissions, and a longer stay in the hospital,” Dr. Symer said.
He had no financial disclosures.
[email protected]
On Twitter @Alz_Gal
LAS VEGAS – Contrary to what some recent studies suggest, patients with complicated appendicitis may benefit from immediate surgery, with shorter hospital stays and fewer postoperative complications.
According to findings from a large database review, delaying the surgery for a complicated case is likely to result in worse patient outcomes, Matthew Symer, MD, said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress.
There are no firm guidelines about the timing of surgery for complicated appendicitis, he said. “There is in fact some controversy about the timing of surgery,” with studies coming to conflicting conclusions about the benefits and risks of both immediate and delayed treatment. “We theorized that the potential morbidity of operating at the height of the inflammatory process would be less than the risk of complications associated with delay,” he said.
To investigate the question, Dr. Symer and his colleagues queried the New York Statewide Planning and Research Cooperative Database, which contains information on all hospital admissions with an ICD-9 code on any patient covered by any payer in the state. Each patient has a unique identifier that allows tracking over time and across facilities.
From 2000 to 2013, the investigators identified 38,840 patients who presented with complicated appendicitis, defined as a perforation. Of these, 31,167 had an appendectomy within 1 year of the index admission. These patients were separated into two groups: those who had surgery within 48 hours of the index admission (28,015) and those who had later surgery (3,152).
The delayed surgery group was further parsed into three: those who had surgery during the index admission, but at least 48 hours after admission (51%); those who had an appendectomy at a subsequent urgent admission (23%); and those who had an elective interval appendectomy sometime within that year (26%).
In comparing the early vs. late surgery groups overall, Dr. Symer noticed some significant initial differences. Patients in the early surgery group were significantly younger (48 vs. 53 years), more likely to be male (55% vs. 47%), white (70% vs. 64%), and to have private insurance (53% vs. 45%).
Comorbidities were more common among the delayed surgery group. These included chronic obstructive pulmonary disease, renal failure, coronary artery disease, hypertension, diabetes, and congestive heart failure. Delayed-surgery patients were more likely to be treated at high-volume hospitals (45% vs. 34%).
Abscess was more common among the delayed surgery group (72% vs. 51%). Their median length of stay was significantly longer (9 vs. 5 days).
Delayed-surgery patients experienced significantly more iatrogenic complications (4% vs. 2%), and more urinary and wound complications. Overall, two or more complications occurred in 23% of the delayed surgery group and 14% of the early surgery group. The readmission rate was higher (28% vs. 18%). Significantly more in the delayed group reached the 75th percentile in hospital charges (62% vs. 26%).
In a multivariate regression analysis, patients with delayed surgery were more likely to experience a prolonged length of stay (odds ratio, 6); high hospital charges (OR, 4.8), iatrogenic complications (OR, 1.9), any complications (OR, 1.5) and readmission (OR, 1.5).
These findings were largely recapitulated when Dr. Symer broke the delayed group down into the three subgroups: patients who had surgery late in the index admission, patients who had an urgent later appendectomy, and patients who had a later elective procedure.
“All of these relationships held up, with patients who delayed surgery having worse overall complications, whether iatrogenic or any complications, more readmissions, and a longer stay in the hospital,” Dr. Symer said.
He had no financial disclosures.
[email protected]
On Twitter @Alz_Gal
AT THE ACADEMIC SURGICAL CONGRESS
Key clinical point: Delayed surgery for complicated appendicitis was associated with worse patient outcomes than immediate surgery.
Major finding: Patients with delayed surgery were more likely to experience a prolonged length of stay (odds ratio, 6); high hospital charges (OR, 4.8), iatrogenic complications (OR, 1.9), any complications (OR, 1.5) and readmission (OR, 1.5).
Data source: The database review comprised almost 39,000 patients.
Disclosures: Dr. Symer had no financial disclosures.
Nicardipine okay to use after pediatric cardiac surgery
HOUSTON – The use of nicardipine following cardiac surgery in children appears to be safe and effective, results from a single-center study suggest.
“There has been a traditional hesitation to use calcium channel blockers, particularly in infants, due to underdevelopment of their calcium channels,” study investigator Matthew L. Stone, MD, PhD, said in an interview at the annual meeting of the Society of Thoracic Surgeons.
“Further, these agents have commonly lacked selectivity to the vascular smooth muscles affecting both the blood vessels and the heart. Nicardipine offers a unique advantage over other calcium channel blockers in that it has more direct effects on vascular smooth muscles than it does on the actual myocardium.”
In their study, Dr. Stone, a first-year fellow in the division of cardiothoracic surgery at the University of Virginia Health System, Charlottesville, and his associates noted that nicardipine offers a favorable pharmacokinetic profile with both rapid onset and short half-life. The purpose of the study was to evaluate the use of nicardipine as a first-line agent for treatment of postoperative hypertension and to compare outcomes between children younger than 6 months of age and those older than 6 months. The researchers retrospectively reviewed the medical records of 68 children who received nicardipine for postoperative hypertension after undergoing cardiac surgery at the University of Virginia during 2010-2015. They compared the incidence of adverse postoperative events between 33 children who were younger than 6 months (group 1) and 35 who were older than 6 months (group 2). Major events including stroke or cardiogenic shock were considered failure of therapy.
Dr. Stone and his associates found that all children received nicardipine within a median of 90 minutes following cardiac surgery; 22 (33%) were started on the drug prior to leaving the operating room and most required dosing for less than 24 hours. Clinically significant hypertension that required dose titration or cessation of therapy occurred in 13% of patients, but there were no significant differences between age groups (17% in group 1 vs. 9% in group 2; P = 0.47). “While the incidence of hypotension following nicardipine administration did not reach statistical significance, it’s important to note that going forward, a lower starting dose in infants less than 6 months of age may be most appropriate. This would certainly be an important focus for future prospective study in the development of postoperative blood pressure control protocols,” said Dr. Stone.
No significant adverse events including stroke or cardiogenic shock occurred in either group. In addition, no operative or postoperative factors reviewed were associated with the development of complications during administration of nicardipine. This included cardiopulmonary bypass time, cross-clamp time, ventilator time, nicardipine duration, ICU length of stay, and hospital length of stay.
“Our traditional hesitation to use this class of agents in infants should be reevaluated,” Dr. Stone concluded. “As we move toward standardization and optimization of perioperative care, our study supports the use and prospective clinical study of nicardipine. Additionally, further pharmacologic study of dose-specific responses within myocardial and vascular smooth muscle cells may further optimize this treatment strategy and provide a more reliable standard with which to control blood pressure.
“Our traditional agents such as beta-blockers and nitroprusside have side effects that need to be considered, the most significant of which being myocardial depression and cyanide toxicity. In a limited number of very-high-risk children, we’ve shown that nicardipine may provide an option with less deleterious side effects. It’s a foundation for future study.”
Dr. Stone reported having no financial disclosures.
HOUSTON – The use of nicardipine following cardiac surgery in children appears to be safe and effective, results from a single-center study suggest.
“There has been a traditional hesitation to use calcium channel blockers, particularly in infants, due to underdevelopment of their calcium channels,” study investigator Matthew L. Stone, MD, PhD, said in an interview at the annual meeting of the Society of Thoracic Surgeons.
“Further, these agents have commonly lacked selectivity to the vascular smooth muscles affecting both the blood vessels and the heart. Nicardipine offers a unique advantage over other calcium channel blockers in that it has more direct effects on vascular smooth muscles than it does on the actual myocardium.”
In their study, Dr. Stone, a first-year fellow in the division of cardiothoracic surgery at the University of Virginia Health System, Charlottesville, and his associates noted that nicardipine offers a favorable pharmacokinetic profile with both rapid onset and short half-life. The purpose of the study was to evaluate the use of nicardipine as a first-line agent for treatment of postoperative hypertension and to compare outcomes between children younger than 6 months of age and those older than 6 months. The researchers retrospectively reviewed the medical records of 68 children who received nicardipine for postoperative hypertension after undergoing cardiac surgery at the University of Virginia during 2010-2015. They compared the incidence of adverse postoperative events between 33 children who were younger than 6 months (group 1) and 35 who were older than 6 months (group 2). Major events including stroke or cardiogenic shock were considered failure of therapy.
Dr. Stone and his associates found that all children received nicardipine within a median of 90 minutes following cardiac surgery; 22 (33%) were started on the drug prior to leaving the operating room and most required dosing for less than 24 hours. Clinically significant hypertension that required dose titration or cessation of therapy occurred in 13% of patients, but there were no significant differences between age groups (17% in group 1 vs. 9% in group 2; P = 0.47). “While the incidence of hypotension following nicardipine administration did not reach statistical significance, it’s important to note that going forward, a lower starting dose in infants less than 6 months of age may be most appropriate. This would certainly be an important focus for future prospective study in the development of postoperative blood pressure control protocols,” said Dr. Stone.
No significant adverse events including stroke or cardiogenic shock occurred in either group. In addition, no operative or postoperative factors reviewed were associated with the development of complications during administration of nicardipine. This included cardiopulmonary bypass time, cross-clamp time, ventilator time, nicardipine duration, ICU length of stay, and hospital length of stay.
“Our traditional hesitation to use this class of agents in infants should be reevaluated,” Dr. Stone concluded. “As we move toward standardization and optimization of perioperative care, our study supports the use and prospective clinical study of nicardipine. Additionally, further pharmacologic study of dose-specific responses within myocardial and vascular smooth muscle cells may further optimize this treatment strategy and provide a more reliable standard with which to control blood pressure.
“Our traditional agents such as beta-blockers and nitroprusside have side effects that need to be considered, the most significant of which being myocardial depression and cyanide toxicity. In a limited number of very-high-risk children, we’ve shown that nicardipine may provide an option with less deleterious side effects. It’s a foundation for future study.”
Dr. Stone reported having no financial disclosures.
HOUSTON – The use of nicardipine following cardiac surgery in children appears to be safe and effective, results from a single-center study suggest.
“There has been a traditional hesitation to use calcium channel blockers, particularly in infants, due to underdevelopment of their calcium channels,” study investigator Matthew L. Stone, MD, PhD, said in an interview at the annual meeting of the Society of Thoracic Surgeons.
“Further, these agents have commonly lacked selectivity to the vascular smooth muscles affecting both the blood vessels and the heart. Nicardipine offers a unique advantage over other calcium channel blockers in that it has more direct effects on vascular smooth muscles than it does on the actual myocardium.”
In their study, Dr. Stone, a first-year fellow in the division of cardiothoracic surgery at the University of Virginia Health System, Charlottesville, and his associates noted that nicardipine offers a favorable pharmacokinetic profile with both rapid onset and short half-life. The purpose of the study was to evaluate the use of nicardipine as a first-line agent for treatment of postoperative hypertension and to compare outcomes between children younger than 6 months of age and those older than 6 months. The researchers retrospectively reviewed the medical records of 68 children who received nicardipine for postoperative hypertension after undergoing cardiac surgery at the University of Virginia during 2010-2015. They compared the incidence of adverse postoperative events between 33 children who were younger than 6 months (group 1) and 35 who were older than 6 months (group 2). Major events including stroke or cardiogenic shock were considered failure of therapy.
Dr. Stone and his associates found that all children received nicardipine within a median of 90 minutes following cardiac surgery; 22 (33%) were started on the drug prior to leaving the operating room and most required dosing for less than 24 hours. Clinically significant hypertension that required dose titration or cessation of therapy occurred in 13% of patients, but there were no significant differences between age groups (17% in group 1 vs. 9% in group 2; P = 0.47). “While the incidence of hypotension following nicardipine administration did not reach statistical significance, it’s important to note that going forward, a lower starting dose in infants less than 6 months of age may be most appropriate. This would certainly be an important focus for future prospective study in the development of postoperative blood pressure control protocols,” said Dr. Stone.
No significant adverse events including stroke or cardiogenic shock occurred in either group. In addition, no operative or postoperative factors reviewed were associated with the development of complications during administration of nicardipine. This included cardiopulmonary bypass time, cross-clamp time, ventilator time, nicardipine duration, ICU length of stay, and hospital length of stay.
“Our traditional hesitation to use this class of agents in infants should be reevaluated,” Dr. Stone concluded. “As we move toward standardization and optimization of perioperative care, our study supports the use and prospective clinical study of nicardipine. Additionally, further pharmacologic study of dose-specific responses within myocardial and vascular smooth muscle cells may further optimize this treatment strategy and provide a more reliable standard with which to control blood pressure.
“Our traditional agents such as beta-blockers and nitroprusside have side effects that need to be considered, the most significant of which being myocardial depression and cyanide toxicity. In a limited number of very-high-risk children, we’ve shown that nicardipine may provide an option with less deleterious side effects. It’s a foundation for future study.”
Dr. Stone reported having no financial disclosures.
AT THE STS ANNUAL MEETING
Key clinical point:
Major finding: The incidence of adverse postoperative events was similar between children who were younger than 6 months and those who were older than 6 months (17% vs. 9%, respectively), but no significant adverse events, including stroke and cardiogenic shock, occurred in either group.
Data source: A retrospective review of 68 children who received nicardipine for postoperative hypertension after undergoing cardiac surgery during 2010-2015.
Disclosures: Dr. Stone reported having no financial disclosures.
Four factors signal complicated appendicitis
LAS VEGAS – Four clinical and imaging characteristics can preoperatively identify cases of complicated appendicitis, potentially saving many from long and unnecessary courses of antibiotics.
In a retrospective study, increasing age and days of pain, combined with the size of the appendix and the presence of an appendicolith on imaging were significantly associated with a histopathologic diagnosis of complicated appendicitis, Jonathan Imran, MD, said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress.
“A lot of patients get tagged as complicated but they really aren’t,” said Dr. Imran, a surgical resident at the University of Texas, Dallas. “This definition then guides clinical assessment and has a profound impact on postoperative antibiotics and length of stay. Despite its common use, intraoperative assessment of complicated appendicitis remains subjective.”
On the other hand, the standard of a histopathologic diagnosis isn’t available during surgery to guide postoperative management. Dr. Imran and his colleagues sought to create a risk assessment tool to identify patients at risk of complicated appendicitis on the basis of clinical and imaging findings.
They retrospectively examined 1,066 patients who underwent appendectomy at a single institution from 2011 to 2013. They compared the intraoperative designations of simple and complicated appendicitis with the histopathologic diagnosis.
Of the 827 patients designated as having simple appendicitis during surgery, 763 (93%) were confirmed by histopathology. The remainder had complicated appendicitis on histopathology.
Of the 239 patients designated as having complicated appendicitis during surgery, 143 (60%) were confirmed by histopathology. The remainder actually had simple appendicitis. Of these 96 patients, 60% went on to have prolonged courses of antibiotics that, by definition, were unnecessary.
The team then looked at 30 patient variables in an attempt to construct a prediction tool. Among the significant associations with a complicated presentation were older age, type 2 diabetes, longer duration of pain, less lower left quadrant pain, higher median temperature, higher serum creatinine, longer time from presentation of symptoms to surgery, larger appendix diameter, abscess, and the presence of an appendicolith.
Four of these factors remained significantly associated with complicated appendicitis in a multivariate regression analysis:
• Age (per 10 years) – odds ratio, 1.25.
• Duration of pain (per day) – OR, 1.21.
• Appendix diameter on imaging (per mm) – OR, 1.10.
• Presence of an appendicolith on imaging – OR, 1.65.
These findings are the basis of a preoperative risk assessment score the team is developing, which will be prospectively tested.
“We hope that these predictors, in combination with improved intraoperative grading, could be used to achieve a more timely and accurate diagnosis of complicated appendicitis,” Dr. Imran said.
He had no financial disclosures.
[email protected]
On Twitter @alz_gal
LAS VEGAS – Four clinical and imaging characteristics can preoperatively identify cases of complicated appendicitis, potentially saving many from long and unnecessary courses of antibiotics.
In a retrospective study, increasing age and days of pain, combined with the size of the appendix and the presence of an appendicolith on imaging were significantly associated with a histopathologic diagnosis of complicated appendicitis, Jonathan Imran, MD, said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress.
“A lot of patients get tagged as complicated but they really aren’t,” said Dr. Imran, a surgical resident at the University of Texas, Dallas. “This definition then guides clinical assessment and has a profound impact on postoperative antibiotics and length of stay. Despite its common use, intraoperative assessment of complicated appendicitis remains subjective.”
On the other hand, the standard of a histopathologic diagnosis isn’t available during surgery to guide postoperative management. Dr. Imran and his colleagues sought to create a risk assessment tool to identify patients at risk of complicated appendicitis on the basis of clinical and imaging findings.
They retrospectively examined 1,066 patients who underwent appendectomy at a single institution from 2011 to 2013. They compared the intraoperative designations of simple and complicated appendicitis with the histopathologic diagnosis.
Of the 827 patients designated as having simple appendicitis during surgery, 763 (93%) were confirmed by histopathology. The remainder had complicated appendicitis on histopathology.
Of the 239 patients designated as having complicated appendicitis during surgery, 143 (60%) were confirmed by histopathology. The remainder actually had simple appendicitis. Of these 96 patients, 60% went on to have prolonged courses of antibiotics that, by definition, were unnecessary.
The team then looked at 30 patient variables in an attempt to construct a prediction tool. Among the significant associations with a complicated presentation were older age, type 2 diabetes, longer duration of pain, less lower left quadrant pain, higher median temperature, higher serum creatinine, longer time from presentation of symptoms to surgery, larger appendix diameter, abscess, and the presence of an appendicolith.
Four of these factors remained significantly associated with complicated appendicitis in a multivariate regression analysis:
• Age (per 10 years) – odds ratio, 1.25.
• Duration of pain (per day) – OR, 1.21.
• Appendix diameter on imaging (per mm) – OR, 1.10.
• Presence of an appendicolith on imaging – OR, 1.65.
These findings are the basis of a preoperative risk assessment score the team is developing, which will be prospectively tested.
“We hope that these predictors, in combination with improved intraoperative grading, could be used to achieve a more timely and accurate diagnosis of complicated appendicitis,” Dr. Imran said.
He had no financial disclosures.
[email protected]
On Twitter @alz_gal
LAS VEGAS – Four clinical and imaging characteristics can preoperatively identify cases of complicated appendicitis, potentially saving many from long and unnecessary courses of antibiotics.
In a retrospective study, increasing age and days of pain, combined with the size of the appendix and the presence of an appendicolith on imaging were significantly associated with a histopathologic diagnosis of complicated appendicitis, Jonathan Imran, MD, said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress.
“A lot of patients get tagged as complicated but they really aren’t,” said Dr. Imran, a surgical resident at the University of Texas, Dallas. “This definition then guides clinical assessment and has a profound impact on postoperative antibiotics and length of stay. Despite its common use, intraoperative assessment of complicated appendicitis remains subjective.”
On the other hand, the standard of a histopathologic diagnosis isn’t available during surgery to guide postoperative management. Dr. Imran and his colleagues sought to create a risk assessment tool to identify patients at risk of complicated appendicitis on the basis of clinical and imaging findings.
They retrospectively examined 1,066 patients who underwent appendectomy at a single institution from 2011 to 2013. They compared the intraoperative designations of simple and complicated appendicitis with the histopathologic diagnosis.
Of the 827 patients designated as having simple appendicitis during surgery, 763 (93%) were confirmed by histopathology. The remainder had complicated appendicitis on histopathology.
Of the 239 patients designated as having complicated appendicitis during surgery, 143 (60%) were confirmed by histopathology. The remainder actually had simple appendicitis. Of these 96 patients, 60% went on to have prolonged courses of antibiotics that, by definition, were unnecessary.
The team then looked at 30 patient variables in an attempt to construct a prediction tool. Among the significant associations with a complicated presentation were older age, type 2 diabetes, longer duration of pain, less lower left quadrant pain, higher median temperature, higher serum creatinine, longer time from presentation of symptoms to surgery, larger appendix diameter, abscess, and the presence of an appendicolith.
Four of these factors remained significantly associated with complicated appendicitis in a multivariate regression analysis:
• Age (per 10 years) – odds ratio, 1.25.
• Duration of pain (per day) – OR, 1.21.
• Appendix diameter on imaging (per mm) – OR, 1.10.
• Presence of an appendicolith on imaging – OR, 1.65.
These findings are the basis of a preoperative risk assessment score the team is developing, which will be prospectively tested.
“We hope that these predictors, in combination with improved intraoperative grading, could be used to achieve a more timely and accurate diagnosis of complicated appendicitis,” Dr. Imran said.
He had no financial disclosures.
[email protected]
On Twitter @alz_gal
Key clinical point: Two clinical signs and two imaging findings can help identify patients with complicated appendicitis.
Major finding: Older age, days of pain, appendix diameter, and the presence of an appendicolith significantly predicted a complicated presentation.
Data source: The retrospective study comprised 1,066 patients.
Disclosures: Dr. Imran had no financial disclosures.
Liver transplantation largely effective in critically ill children
The use of advanced critical care in children and infants with liver failure is justified because orthotopic liver transplantation can be performed on the sickest children and achieve acceptable outcomes, results from a large analysis demonstrated.
“Hand in hand with improved care for critically ill children with liver failure, posttransplant critical care has made tremendous strides,” Abbas Rana, MD, wrote in a study published online in the Journal of the American College of Surgeons. “Our recipients have gotten sicker while our postoperative outcomes have improved. The question then becomes, have our operative skills and postoperative critical care management kept up with the abilities to keep sick children with liver failure alive? Just because transplantation is now possible in our sickest children, is it justified?”
To find out, Dr. Rana of the division of abdominal transplantation and hepatobiliary surgery at Baylor College of Medicine, Houston, and colleagues retrospectively analyzed United Network for Organ Sharing data from all orthotopic liver transplantation (OLT) recipients between Sept. 1, 1987, and June 30, 2015. The analysis paired the liver registry data with data collected by the Organ Procurement and Transplantation Network, and was limited to transplant recipients younger than age 18. The researchers followed a total of 13,723 recipients from date of transplant until either death or the date of last known follow-up (J Am Coll Surg. 2016 Dec 25. doi: 10.1016/j.jamcollsurg.2016.12.025).
In another part of the study, the researchers retrospectively reviewed the charts of 354 patients under 18 years of age who underwent OLT between March 1, 2002, and June 30, 2015, at Texas Children’s Hospital, including 65 who were admitted to the ICU at the time of transplantation.
In the analysis of national data, the researchers found that the rates of 1-year survival following OLT in children in the ICU improved from 60% in 1987 to 92% in 2013 (P less than .001). The rates of 1-year survival also improved for children on dialysis at the time of transplant (from 50% in 1995 to 95% in 2013; P less than .001) and for those dependent on a mechanical ventilator at the time of transplant (from 49% in 1994 to 94% in 2013; P less than .001). The significant risk factors were two previous transplants (hazard ratio, 4.2), one previous transplant (HR, 2.5), serum sodium greater than 150 mEq/L (HR, 2.0), dialysis or glomerular filtration rate less than 30 mL/min per 1.73 m2 (HR, 2.0), mechanical ventilator dependence (HR, 1.8), body weight under 6 kg (HR, 1.8), encephalopathy (HR, 1.8), and annual center volume of fewer than five cases (HR, 1.7).
In the experience at Texas Children’s Hospital, the researchers observed “preserved and successful patient survival outcomes” in many markers of acuity. For example, the 10-year survival rates for patients dependent on mechanical ventilation and dialysis were 85% and 96%, respectively, and reached 100% for those requiring therapeutic plasma exchange, molescular adsorbent recirculating system (MARS) liver dialysis, and vasopressors.
“Our collective ability to keep sick children alive with liver failure has improved considerably over the years,” the researchers wrote. “Keeping pace, this analysis demonstrates that the posttransplant outcomes have also improved dramatically. The survival outcomes are comparable to the general population, justifying the use of scarce donors. Although we cannot declare in absolute that no child should be left behind, we can demonstrate acceptable outcomes to date and urge the continual revisiting of our concepts of futility.
“We have learned throughout our experience that almost every child with end-stage liver disease and acute liver failure should be offered liver replacement, as long as the vasoactive medication and mechanical support are not maximized prior to the initiation of the OLT procedure. Every effort should be made to transplant our sickest children.”
This study was supported by the Cade R. Alpard Foundation. The researchers reported having no relevant financial disclosures.
The use of advanced critical care in children and infants with liver failure is justified because orthotopic liver transplantation can be performed on the sickest children and achieve acceptable outcomes, results from a large analysis demonstrated.
“Hand in hand with improved care for critically ill children with liver failure, posttransplant critical care has made tremendous strides,” Abbas Rana, MD, wrote in a study published online in the Journal of the American College of Surgeons. “Our recipients have gotten sicker while our postoperative outcomes have improved. The question then becomes, have our operative skills and postoperative critical care management kept up with the abilities to keep sick children with liver failure alive? Just because transplantation is now possible in our sickest children, is it justified?”
To find out, Dr. Rana of the division of abdominal transplantation and hepatobiliary surgery at Baylor College of Medicine, Houston, and colleagues retrospectively analyzed United Network for Organ Sharing data from all orthotopic liver transplantation (OLT) recipients between Sept. 1, 1987, and June 30, 2015. The analysis paired the liver registry data with data collected by the Organ Procurement and Transplantation Network, and was limited to transplant recipients younger than age 18. The researchers followed a total of 13,723 recipients from date of transplant until either death or the date of last known follow-up (J Am Coll Surg. 2016 Dec 25. doi: 10.1016/j.jamcollsurg.2016.12.025).
In another part of the study, the researchers retrospectively reviewed the charts of 354 patients under 18 years of age who underwent OLT between March 1, 2002, and June 30, 2015, at Texas Children’s Hospital, including 65 who were admitted to the ICU at the time of transplantation.
In the analysis of national data, the researchers found that the rates of 1-year survival following OLT in children in the ICU improved from 60% in 1987 to 92% in 2013 (P less than .001). The rates of 1-year survival also improved for children on dialysis at the time of transplant (from 50% in 1995 to 95% in 2013; P less than .001) and for those dependent on a mechanical ventilator at the time of transplant (from 49% in 1994 to 94% in 2013; P less than .001). The significant risk factors were two previous transplants (hazard ratio, 4.2), one previous transplant (HR, 2.5), serum sodium greater than 150 mEq/L (HR, 2.0), dialysis or glomerular filtration rate less than 30 mL/min per 1.73 m2 (HR, 2.0), mechanical ventilator dependence (HR, 1.8), body weight under 6 kg (HR, 1.8), encephalopathy (HR, 1.8), and annual center volume of fewer than five cases (HR, 1.7).
In the experience at Texas Children’s Hospital, the researchers observed “preserved and successful patient survival outcomes” in many markers of acuity. For example, the 10-year survival rates for patients dependent on mechanical ventilation and dialysis were 85% and 96%, respectively, and reached 100% for those requiring therapeutic plasma exchange, molescular adsorbent recirculating system (MARS) liver dialysis, and vasopressors.
“Our collective ability to keep sick children alive with liver failure has improved considerably over the years,” the researchers wrote. “Keeping pace, this analysis demonstrates that the posttransplant outcomes have also improved dramatically. The survival outcomes are comparable to the general population, justifying the use of scarce donors. Although we cannot declare in absolute that no child should be left behind, we can demonstrate acceptable outcomes to date and urge the continual revisiting of our concepts of futility.
“We have learned throughout our experience that almost every child with end-stage liver disease and acute liver failure should be offered liver replacement, as long as the vasoactive medication and mechanical support are not maximized prior to the initiation of the OLT procedure. Every effort should be made to transplant our sickest children.”
This study was supported by the Cade R. Alpard Foundation. The researchers reported having no relevant financial disclosures.
The use of advanced critical care in children and infants with liver failure is justified because orthotopic liver transplantation can be performed on the sickest children and achieve acceptable outcomes, results from a large analysis demonstrated.
“Hand in hand with improved care for critically ill children with liver failure, posttransplant critical care has made tremendous strides,” Abbas Rana, MD, wrote in a study published online in the Journal of the American College of Surgeons. “Our recipients have gotten sicker while our postoperative outcomes have improved. The question then becomes, have our operative skills and postoperative critical care management kept up with the abilities to keep sick children with liver failure alive? Just because transplantation is now possible in our sickest children, is it justified?”
To find out, Dr. Rana of the division of abdominal transplantation and hepatobiliary surgery at Baylor College of Medicine, Houston, and colleagues retrospectively analyzed United Network for Organ Sharing data from all orthotopic liver transplantation (OLT) recipients between Sept. 1, 1987, and June 30, 2015. The analysis paired the liver registry data with data collected by the Organ Procurement and Transplantation Network, and was limited to transplant recipients younger than age 18. The researchers followed a total of 13,723 recipients from date of transplant until either death or the date of last known follow-up (J Am Coll Surg. 2016 Dec 25. doi: 10.1016/j.jamcollsurg.2016.12.025).
In another part of the study, the researchers retrospectively reviewed the charts of 354 patients under 18 years of age who underwent OLT between March 1, 2002, and June 30, 2015, at Texas Children’s Hospital, including 65 who were admitted to the ICU at the time of transplantation.
In the analysis of national data, the researchers found that the rates of 1-year survival following OLT in children in the ICU improved from 60% in 1987 to 92% in 2013 (P less than .001). The rates of 1-year survival also improved for children on dialysis at the time of transplant (from 50% in 1995 to 95% in 2013; P less than .001) and for those dependent on a mechanical ventilator at the time of transplant (from 49% in 1994 to 94% in 2013; P less than .001). The significant risk factors were two previous transplants (hazard ratio, 4.2), one previous transplant (HR, 2.5), serum sodium greater than 150 mEq/L (HR, 2.0), dialysis or glomerular filtration rate less than 30 mL/min per 1.73 m2 (HR, 2.0), mechanical ventilator dependence (HR, 1.8), body weight under 6 kg (HR, 1.8), encephalopathy (HR, 1.8), and annual center volume of fewer than five cases (HR, 1.7).
In the experience at Texas Children’s Hospital, the researchers observed “preserved and successful patient survival outcomes” in many markers of acuity. For example, the 10-year survival rates for patients dependent on mechanical ventilation and dialysis were 85% and 96%, respectively, and reached 100% for those requiring therapeutic plasma exchange, molescular adsorbent recirculating system (MARS) liver dialysis, and vasopressors.
“Our collective ability to keep sick children alive with liver failure has improved considerably over the years,” the researchers wrote. “Keeping pace, this analysis demonstrates that the posttransplant outcomes have also improved dramatically. The survival outcomes are comparable to the general population, justifying the use of scarce donors. Although we cannot declare in absolute that no child should be left behind, we can demonstrate acceptable outcomes to date and urge the continual revisiting of our concepts of futility.
“We have learned throughout our experience that almost every child with end-stage liver disease and acute liver failure should be offered liver replacement, as long as the vasoactive medication and mechanical support are not maximized prior to the initiation of the OLT procedure. Every effort should be made to transplant our sickest children.”
This study was supported by the Cade R. Alpard Foundation. The researchers reported having no relevant financial disclosures.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF SURGEONS
Key clinical point:
Major finding: The rates of 1-year survival following orthotopic liver transplantation in ICU children improved from 60% in 1987 to 92% in 2013 (P less than .001).
Data source: An analysis of 13,723 patients under the age of 18 years who underwent OLT between Sept. 1, 1987, and June 30, 2015.
Disclosures: The study was supported by the Cade R. Alpard Foundation. The researchers reported having no relevant financial disclosures.
Intrathecal hydromorphone boosts post-op pain control
LAS VEGAS – Intrathecal hydromorphone, administered alone or with lidocaine, effectively controlled pain and decreased postoperative opioid use after colorectal surgery in a retrospective study.
The technique was so effective that 28% of patients required no postoperative opioids at all, Amit Merchea, MD, said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress.
The intrathecal analgesic was part of an enhanced recovery pathway (ERP) for patients undergoing elective colorectal surgery at the Mayo Clinic, Jacksonville, Fla., where Dr. Merchea practices colorectal surgery.
Morphine has been the gold standard for this approach, he said. Dr. Merchea and his colleagues investigated the use of hydromorphone in 601 patients who underwent open or minimally invasive colorectal surgery at the Mayo Clinic from 2012 to 2013.
The patients were a median of 52 years old. The surgical approach was almost evenly split between open and laparoscopic. The median length of hospital stay was 3 days. All received intrathecal hydromorphone either alone (91%) or with a local anesthetic (9%).
Everyone was on the same presurgical and postsurgical pain control regimen, which consisted of celecoxib, gabapentin, and acetaminophen before surgery, followed by nonsteroidal anti-inflammatories and acetaminophen, with oxycodone as needed, after surgery.
Overall, the procedure was well tolerated, with seven cases of pruritus requiring Nubain (nalbuphine), one case of respiratory depression that required naloxone, and one postdural headache that required a patch. The rate of ileus was 16%.
At 4 hours, the median pain score was 3 on a 1- to 10-point scale. At 24 hours, it was a median of 4. By 48 hours, the median pain score was 6. This increase is to be expected as the hydrocodone exists in the intrathecal space for up to 36 hours, Dr. Merchea noted.
The median total oral morphine equivalent (OME) was 24; 170 patients (28%) needed no opioid medications after surgery.
He also presented outcomes by infusion composition. There was no difference in the rate of ileus among those who had hydromorphone alone and those who had it with lidocaine. The length of stay was 3 vs. 3.5 days, respectively. The only significant difference in pain scores was the 48-hour maximum, which was a median of 7 in the combination group and 6 in the hydromorphone-only group.
The combination group, however, required more postoperative opioids (33.8 vs. 22.5 OMEs). Significantly more patients in the hydromorphone-only group were able to go without any postoperative opioids (30% vs. 15%).
Dr. Merchea also broke down the results by hydromorphone dosage, but there were no significant differences in ileus rate, length of stay, or pain scores correlated with dosage. However, those who received higher doses were significantly more likely to need more postoperative opioids than those who had lower doses.
Session moderator Peter Muscarella, MD, of Montefiore Medical Center, New York, asked whether the intrathecal infusion was associated with hypotension. “Some of these procedures with epidural analgesics intraoperatively, we have seen shifts in blood pressure that result in excess fluid administration, sometimes leading to tissue complications.”
Dr. Merchea said hypotension was not an outcome of this trial, but that he has looked at it before. “We have previously reported that epidural analgesia was associated with a 15% occurrence of hypotension, but it had no clinical impact and didn’t warrant giving any additional fluids.”
Dr. Merchea had no relevant financial disclosures.
[email protected]
On Twitter @Alz_Gal
LAS VEGAS – Intrathecal hydromorphone, administered alone or with lidocaine, effectively controlled pain and decreased postoperative opioid use after colorectal surgery in a retrospective study.
The technique was so effective that 28% of patients required no postoperative opioids at all, Amit Merchea, MD, said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress.
The intrathecal analgesic was part of an enhanced recovery pathway (ERP) for patients undergoing elective colorectal surgery at the Mayo Clinic, Jacksonville, Fla., where Dr. Merchea practices colorectal surgery.
Morphine has been the gold standard for this approach, he said. Dr. Merchea and his colleagues investigated the use of hydromorphone in 601 patients who underwent open or minimally invasive colorectal surgery at the Mayo Clinic from 2012 to 2013.
The patients were a median of 52 years old. The surgical approach was almost evenly split between open and laparoscopic. The median length of hospital stay was 3 days. All received intrathecal hydromorphone either alone (91%) or with a local anesthetic (9%).
Everyone was on the same presurgical and postsurgical pain control regimen, which consisted of celecoxib, gabapentin, and acetaminophen before surgery, followed by nonsteroidal anti-inflammatories and acetaminophen, with oxycodone as needed, after surgery.
Overall, the procedure was well tolerated, with seven cases of pruritus requiring Nubain (nalbuphine), one case of respiratory depression that required naloxone, and one postdural headache that required a patch. The rate of ileus was 16%.
At 4 hours, the median pain score was 3 on a 1- to 10-point scale. At 24 hours, it was a median of 4. By 48 hours, the median pain score was 6. This increase is to be expected as the hydrocodone exists in the intrathecal space for up to 36 hours, Dr. Merchea noted.
The median total oral morphine equivalent (OME) was 24; 170 patients (28%) needed no opioid medications after surgery.
He also presented outcomes by infusion composition. There was no difference in the rate of ileus among those who had hydromorphone alone and those who had it with lidocaine. The length of stay was 3 vs. 3.5 days, respectively. The only significant difference in pain scores was the 48-hour maximum, which was a median of 7 in the combination group and 6 in the hydromorphone-only group.
The combination group, however, required more postoperative opioids (33.8 vs. 22.5 OMEs). Significantly more patients in the hydromorphone-only group were able to go without any postoperative opioids (30% vs. 15%).
Dr. Merchea also broke down the results by hydromorphone dosage, but there were no significant differences in ileus rate, length of stay, or pain scores correlated with dosage. However, those who received higher doses were significantly more likely to need more postoperative opioids than those who had lower doses.
Session moderator Peter Muscarella, MD, of Montefiore Medical Center, New York, asked whether the intrathecal infusion was associated with hypotension. “Some of these procedures with epidural analgesics intraoperatively, we have seen shifts in blood pressure that result in excess fluid administration, sometimes leading to tissue complications.”
Dr. Merchea said hypotension was not an outcome of this trial, but that he has looked at it before. “We have previously reported that epidural analgesia was associated with a 15% occurrence of hypotension, but it had no clinical impact and didn’t warrant giving any additional fluids.”
Dr. Merchea had no relevant financial disclosures.
[email protected]
On Twitter @Alz_Gal
LAS VEGAS – Intrathecal hydromorphone, administered alone or with lidocaine, effectively controlled pain and decreased postoperative opioid use after colorectal surgery in a retrospective study.
The technique was so effective that 28% of patients required no postoperative opioids at all, Amit Merchea, MD, said at the Association for Academic Surgery/Society of University Surgeons Academic Surgical Congress.
The intrathecal analgesic was part of an enhanced recovery pathway (ERP) for patients undergoing elective colorectal surgery at the Mayo Clinic, Jacksonville, Fla., where Dr. Merchea practices colorectal surgery.
Morphine has been the gold standard for this approach, he said. Dr. Merchea and his colleagues investigated the use of hydromorphone in 601 patients who underwent open or minimally invasive colorectal surgery at the Mayo Clinic from 2012 to 2013.
The patients were a median of 52 years old. The surgical approach was almost evenly split between open and laparoscopic. The median length of hospital stay was 3 days. All received intrathecal hydromorphone either alone (91%) or with a local anesthetic (9%).
Everyone was on the same presurgical and postsurgical pain control regimen, which consisted of celecoxib, gabapentin, and acetaminophen before surgery, followed by nonsteroidal anti-inflammatories and acetaminophen, with oxycodone as needed, after surgery.
Overall, the procedure was well tolerated, with seven cases of pruritus requiring Nubain (nalbuphine), one case of respiratory depression that required naloxone, and one postdural headache that required a patch. The rate of ileus was 16%.
At 4 hours, the median pain score was 3 on a 1- to 10-point scale. At 24 hours, it was a median of 4. By 48 hours, the median pain score was 6. This increase is to be expected as the hydrocodone exists in the intrathecal space for up to 36 hours, Dr. Merchea noted.
The median total oral morphine equivalent (OME) was 24; 170 patients (28%) needed no opioid medications after surgery.
He also presented outcomes by infusion composition. There was no difference in the rate of ileus among those who had hydromorphone alone and those who had it with lidocaine. The length of stay was 3 vs. 3.5 days, respectively. The only significant difference in pain scores was the 48-hour maximum, which was a median of 7 in the combination group and 6 in the hydromorphone-only group.
The combination group, however, required more postoperative opioids (33.8 vs. 22.5 OMEs). Significantly more patients in the hydromorphone-only group were able to go without any postoperative opioids (30% vs. 15%).
Dr. Merchea also broke down the results by hydromorphone dosage, but there were no significant differences in ileus rate, length of stay, or pain scores correlated with dosage. However, those who received higher doses were significantly more likely to need more postoperative opioids than those who had lower doses.
Session moderator Peter Muscarella, MD, of Montefiore Medical Center, New York, asked whether the intrathecal infusion was associated with hypotension. “Some of these procedures with epidural analgesics intraoperatively, we have seen shifts in blood pressure that result in excess fluid administration, sometimes leading to tissue complications.”
Dr. Merchea said hypotension was not an outcome of this trial, but that he has looked at it before. “We have previously reported that epidural analgesia was associated with a 15% occurrence of hypotension, but it had no clinical impact and didn’t warrant giving any additional fluids.”
Dr. Merchea had no relevant financial disclosures.
[email protected]
On Twitter @Alz_Gal
AT THE ACADEMIC SURGICAL CONGRESS
Key clinical point:
Major finding: About a quarter of patients (28%) needed no postoperative opioids.
Data source: A retrospective study of 601 patients.
Disclosures: Dr. Merchea had no relevant financial disclosures.
HHS pick Price made ‘brazen’ trades while committee was under scrutiny
Health and Human Services secretary nominee Tom Price showed little restraint in his personal stock trading during the 3 years that federal investigators were bearing down on a key House committee on which the Republican congressman served, a review of his financial disclosures shows.
Rep. Price (Ga.) made dozens of health industry stock trades during a 3-year investigation by the Securities and Exchange Commission that focused on the Ways and Means Committee, according to financial disclosure records he filed with the House of Representatives. The investigation was considered the first test of a law passed to ban members of Congress and their staffs from trading stock based on insider information.
Rep. Price, who is a retired orthopedic surgeon, was never a target of the federal investigation, which scrutinized a top Ways and Means staffer, and no charges were brought. But ethics experts say Rep. Price’s personal trading, even during the thick of federal pressure on his committee, shows he was unconcerned about financial investments that could create an appearance of impropriety.
“He should have known better,” Richard Painter, former White House chief ethics attorney under President George W. Bush and a professor at the University of Minnesota Law School said of Rep. Price’s conduct during the SEC inquiry.
As Rep. Price awaits a Senate vote on his confirmation, Senate Democrats and a number of watchdog groups have asked the SEC to investigate whether Rep. Price engaged in insider trading with some of his trades in health care companies. Rep. Price has said he abided by all ethics rules, although he acknowledged to the Senate Finance Committee that he did not consult the House Ethics Committee on trades that have now become controversial.
The SEC’s inquiry began in 2013, as it battled Ways and Means for documents to develop its case.
A few weeks ago, the day before President Donald Trump’s inauguration, the SEC quietly dropped its pursuit of committee documents without explanation, according to federal court records. No charges were brought against the staffer, Brian Sutter, who is now a health care lobbyist. Sutter’s lawyer declined to comment.
Craig Holman, government affairs lobbyist with Public Citizen, described Rep. Price’s volume of stock trades during the SEC inquiry as “brazen,” given the congressman’s access to nonpublic information affecting the companies’ fortunes.
“The public is seeing this and they really don’t like it,” said Holman, whose watchdog group recently filed complaints about Rep. Price’s stock trading with both the SEC and the Office of Congressional Ethics.
Trump administration officials and Rep. Price have dismissed questions that news reports and lawmakers have raised about stock trades coinciding with official actions to help certain companies, saying Rep. Price’s brokers chose the stocks independently and all of his conduct was transparent.
After acknowledging that he asked his broker to buy stock in an Australian drug company, he told the Senate Finance Committee that he did not direct his broker to make other trades.
“To the best of my knowledge, I have not undertaken such actions,” he wrote in response to finance committee questions. “I have abided by and adhered to all ethics and conflict of interest rules applicable to me.”
An analysis of Rep. Price’s trades shows that he bought health stocks in 2007, the first year Congress financial disclosures are posted online. In 2011, the first year Rep. Price sat on the health subcommittee, he traded no health-related stocks, according to his financial disclosures filed with Congress.
That same year, members were facing public criticism because of a book detailing how they could use inside information and a “60 Minutes” investigation focused on how members and staff could legally use inside information to gain from their own stock trades.
In 2012, President Barack Obama signed the Stop Trading on Congressional Knowledge Act to rein in insider trading by members and require more disclosure. Public watchdog groups suggested at the time that the law would curb the practice.
That year, after his 1-year break in health care trades, Rep. Price resumed investing in health care companies.
Along with investments in technology, financial services, and retail stocks, he also bought and sold stock in companies that could be impacted by actions of his subcommittee, which has a role in determining rates the government pays under the Medicare program.
Health care firms spend heavily to influence members of Congress, lobbying on health matters, funding political campaigns, and seeking favor with Medicare officials who decide how much the program will pay for certain drugs and devices. The Food and Drug Administration holds similar power, approving or putting conditions on drug and device use.
Beyond his personal investments in health care companies, Rep. Price has also advocated their interests in letters to officials and proposed laws, government records show.
In 2012, disclosure records show Rep. Price sold stock in several drug firms, including more than $110,000 worth of Amgen stock. Amgen’s stock price had steadily climbed out of a recession-level slump, but Rep. Price’s sale came a few weeks before the company pleaded guilty to illegally marketing an anemia drug.
By 2013, the health subcommittee was at the center of a major conflict between Medicare, which sets Medicare Advantage rates, and the insurance industry. Medicare issued a notice early that year announcing its intention to reduce Medicare Advantage rates by 2.3 percent as part of a major cost-cutting initiative.
That prompted fierce lobbying by the health insurance industry. Members of Congress, including Rep. Price, wrote a letter to Marilyn Tavenner, then acting administrator for the Centers for Medicare & Medicaid Services, protesting the rate cut, saying the decrease would “disadvantage vulnerable beneficiaries with multiple chronic conditions.”
Ultimately, Medicare decided not to cut rates but instead, to increase them. Yet an hour before Medicare announced the change, a Height Securities analyst fired off a “flash” report to 200 clients that touched off a surge of trading.
The analyst’s report said a political deal was hatched on Capitol Hill to prevent the cuts as a condition for moving forward on Tavenner’s confirmation. Medicare officials increased rates by nearly 4 percent, a change that would positively impact the bottom lines of health insurance companies.
The SEC began looking for the leak’s source, and within weeks, FBI agents began interviewing staffers at the Ways and Means Committee, court records show.
They discovered communications between Sutter and a health care lobbyist. The HHS Inspector General also began a probe, and federal prosecutors briefly examined the matter as well.
As the case unfolded, Rep. Price bought more health care-related stocks, according to his financial disclosures. He has testified that his broker directed all of the trades, except for his investments in Innate Immunotherapeutics, an Australian company partly owned by Rep. Chris Collins (R-N.Y.), according to Collins’ disclosures. An HHS spokesman said Monday that Rep. Price held three broker-directed accounts.
Ethics experts have said that Rep. Price should have further distanced himself by placing his assets in a blind trust.
On April 30, 2013, Rep. Price bought $2,093 worth of stocks in Incyte, a company that develops cancer drugs; $2,076 in Onyx Pharmaceuticals, a drug maker that would soon merge with a larger drug firm; and $2,097 in Parexel International, a consultancy that helps drugs and devices win FDA approval, according to the financial disclosure records.
The same day, Rep. Price shed shares of Express Scripts, a drug management firm, and Danaher, which makes products hospitals and doctor’s offices using for testing and diagnostics. In August of that year, he bought a $2,429 stake in Jazz Pharmaceuticals, which makes sleep and cancer drugs.
On May 6, 2014, the SEC served its first subpoena for the Ways and Means Committee documents. The committee launched a vigorous fight, appealing a federal district judge’s ruling that it should comply with the SEC subpoena.
Rep. Price continued his health stock trades, including $1,000 to $15,000 in drug firms Amgen, Biogen, Bristol-Myers Squibb, Eli Lilly, and Pfizer. He also bought stock in Aetna, a major health insurer, and Athenahealth, which sells electronic medical record and medical billing software. In 2016, he also increased his investment in Innate Immunotherapeutics.
The purchase became controversial because both he and Collins bought stock in a private placement at a discounted price.
“You’re asking for trouble if you have access to nonpublic information about the health care industry and you’re buying and selling health care stocks,” Painter said.
Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
Health and Human Services secretary nominee Tom Price showed little restraint in his personal stock trading during the 3 years that federal investigators were bearing down on a key House committee on which the Republican congressman served, a review of his financial disclosures shows.
Rep. Price (Ga.) made dozens of health industry stock trades during a 3-year investigation by the Securities and Exchange Commission that focused on the Ways and Means Committee, according to financial disclosure records he filed with the House of Representatives. The investigation was considered the first test of a law passed to ban members of Congress and their staffs from trading stock based on insider information.
Rep. Price, who is a retired orthopedic surgeon, was never a target of the federal investigation, which scrutinized a top Ways and Means staffer, and no charges were brought. But ethics experts say Rep. Price’s personal trading, even during the thick of federal pressure on his committee, shows he was unconcerned about financial investments that could create an appearance of impropriety.
“He should have known better,” Richard Painter, former White House chief ethics attorney under President George W. Bush and a professor at the University of Minnesota Law School said of Rep. Price’s conduct during the SEC inquiry.
As Rep. Price awaits a Senate vote on his confirmation, Senate Democrats and a number of watchdog groups have asked the SEC to investigate whether Rep. Price engaged in insider trading with some of his trades in health care companies. Rep. Price has said he abided by all ethics rules, although he acknowledged to the Senate Finance Committee that he did not consult the House Ethics Committee on trades that have now become controversial.
The SEC’s inquiry began in 2013, as it battled Ways and Means for documents to develop its case.
A few weeks ago, the day before President Donald Trump’s inauguration, the SEC quietly dropped its pursuit of committee documents without explanation, according to federal court records. No charges were brought against the staffer, Brian Sutter, who is now a health care lobbyist. Sutter’s lawyer declined to comment.
Craig Holman, government affairs lobbyist with Public Citizen, described Rep. Price’s volume of stock trades during the SEC inquiry as “brazen,” given the congressman’s access to nonpublic information affecting the companies’ fortunes.
“The public is seeing this and they really don’t like it,” said Holman, whose watchdog group recently filed complaints about Rep. Price’s stock trading with both the SEC and the Office of Congressional Ethics.
Trump administration officials and Rep. Price have dismissed questions that news reports and lawmakers have raised about stock trades coinciding with official actions to help certain companies, saying Rep. Price’s brokers chose the stocks independently and all of his conduct was transparent.
After acknowledging that he asked his broker to buy stock in an Australian drug company, he told the Senate Finance Committee that he did not direct his broker to make other trades.
“To the best of my knowledge, I have not undertaken such actions,” he wrote in response to finance committee questions. “I have abided by and adhered to all ethics and conflict of interest rules applicable to me.”
An analysis of Rep. Price’s trades shows that he bought health stocks in 2007, the first year Congress financial disclosures are posted online. In 2011, the first year Rep. Price sat on the health subcommittee, he traded no health-related stocks, according to his financial disclosures filed with Congress.
That same year, members were facing public criticism because of a book detailing how they could use inside information and a “60 Minutes” investigation focused on how members and staff could legally use inside information to gain from their own stock trades.
In 2012, President Barack Obama signed the Stop Trading on Congressional Knowledge Act to rein in insider trading by members and require more disclosure. Public watchdog groups suggested at the time that the law would curb the practice.
That year, after his 1-year break in health care trades, Rep. Price resumed investing in health care companies.
Along with investments in technology, financial services, and retail stocks, he also bought and sold stock in companies that could be impacted by actions of his subcommittee, which has a role in determining rates the government pays under the Medicare program.
Health care firms spend heavily to influence members of Congress, lobbying on health matters, funding political campaigns, and seeking favor with Medicare officials who decide how much the program will pay for certain drugs and devices. The Food and Drug Administration holds similar power, approving or putting conditions on drug and device use.
Beyond his personal investments in health care companies, Rep. Price has also advocated their interests in letters to officials and proposed laws, government records show.
In 2012, disclosure records show Rep. Price sold stock in several drug firms, including more than $110,000 worth of Amgen stock. Amgen’s stock price had steadily climbed out of a recession-level slump, but Rep. Price’s sale came a few weeks before the company pleaded guilty to illegally marketing an anemia drug.
By 2013, the health subcommittee was at the center of a major conflict between Medicare, which sets Medicare Advantage rates, and the insurance industry. Medicare issued a notice early that year announcing its intention to reduce Medicare Advantage rates by 2.3 percent as part of a major cost-cutting initiative.
That prompted fierce lobbying by the health insurance industry. Members of Congress, including Rep. Price, wrote a letter to Marilyn Tavenner, then acting administrator for the Centers for Medicare & Medicaid Services, protesting the rate cut, saying the decrease would “disadvantage vulnerable beneficiaries with multiple chronic conditions.”
Ultimately, Medicare decided not to cut rates but instead, to increase them. Yet an hour before Medicare announced the change, a Height Securities analyst fired off a “flash” report to 200 clients that touched off a surge of trading.
The analyst’s report said a political deal was hatched on Capitol Hill to prevent the cuts as a condition for moving forward on Tavenner’s confirmation. Medicare officials increased rates by nearly 4 percent, a change that would positively impact the bottom lines of health insurance companies.
The SEC began looking for the leak’s source, and within weeks, FBI agents began interviewing staffers at the Ways and Means Committee, court records show.
They discovered communications between Sutter and a health care lobbyist. The HHS Inspector General also began a probe, and federal prosecutors briefly examined the matter as well.
As the case unfolded, Rep. Price bought more health care-related stocks, according to his financial disclosures. He has testified that his broker directed all of the trades, except for his investments in Innate Immunotherapeutics, an Australian company partly owned by Rep. Chris Collins (R-N.Y.), according to Collins’ disclosures. An HHS spokesman said Monday that Rep. Price held three broker-directed accounts.
Ethics experts have said that Rep. Price should have further distanced himself by placing his assets in a blind trust.
On April 30, 2013, Rep. Price bought $2,093 worth of stocks in Incyte, a company that develops cancer drugs; $2,076 in Onyx Pharmaceuticals, a drug maker that would soon merge with a larger drug firm; and $2,097 in Parexel International, a consultancy that helps drugs and devices win FDA approval, according to the financial disclosure records.
The same day, Rep. Price shed shares of Express Scripts, a drug management firm, and Danaher, which makes products hospitals and doctor’s offices using for testing and diagnostics. In August of that year, he bought a $2,429 stake in Jazz Pharmaceuticals, which makes sleep and cancer drugs.
On May 6, 2014, the SEC served its first subpoena for the Ways and Means Committee documents. The committee launched a vigorous fight, appealing a federal district judge’s ruling that it should comply with the SEC subpoena.
Rep. Price continued his health stock trades, including $1,000 to $15,000 in drug firms Amgen, Biogen, Bristol-Myers Squibb, Eli Lilly, and Pfizer. He also bought stock in Aetna, a major health insurer, and Athenahealth, which sells electronic medical record and medical billing software. In 2016, he also increased his investment in Innate Immunotherapeutics.
The purchase became controversial because both he and Collins bought stock in a private placement at a discounted price.
“You’re asking for trouble if you have access to nonpublic information about the health care industry and you’re buying and selling health care stocks,” Painter said.
Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
Health and Human Services secretary nominee Tom Price showed little restraint in his personal stock trading during the 3 years that federal investigators were bearing down on a key House committee on which the Republican congressman served, a review of his financial disclosures shows.
Rep. Price (Ga.) made dozens of health industry stock trades during a 3-year investigation by the Securities and Exchange Commission that focused on the Ways and Means Committee, according to financial disclosure records he filed with the House of Representatives. The investigation was considered the first test of a law passed to ban members of Congress and their staffs from trading stock based on insider information.
Rep. Price, who is a retired orthopedic surgeon, was never a target of the federal investigation, which scrutinized a top Ways and Means staffer, and no charges were brought. But ethics experts say Rep. Price’s personal trading, even during the thick of federal pressure on his committee, shows he was unconcerned about financial investments that could create an appearance of impropriety.
“He should have known better,” Richard Painter, former White House chief ethics attorney under President George W. Bush and a professor at the University of Minnesota Law School said of Rep. Price’s conduct during the SEC inquiry.
As Rep. Price awaits a Senate vote on his confirmation, Senate Democrats and a number of watchdog groups have asked the SEC to investigate whether Rep. Price engaged in insider trading with some of his trades in health care companies. Rep. Price has said he abided by all ethics rules, although he acknowledged to the Senate Finance Committee that he did not consult the House Ethics Committee on trades that have now become controversial.
The SEC’s inquiry began in 2013, as it battled Ways and Means for documents to develop its case.
A few weeks ago, the day before President Donald Trump’s inauguration, the SEC quietly dropped its pursuit of committee documents without explanation, according to federal court records. No charges were brought against the staffer, Brian Sutter, who is now a health care lobbyist. Sutter’s lawyer declined to comment.
Craig Holman, government affairs lobbyist with Public Citizen, described Rep. Price’s volume of stock trades during the SEC inquiry as “brazen,” given the congressman’s access to nonpublic information affecting the companies’ fortunes.
“The public is seeing this and they really don’t like it,” said Holman, whose watchdog group recently filed complaints about Rep. Price’s stock trading with both the SEC and the Office of Congressional Ethics.
Trump administration officials and Rep. Price have dismissed questions that news reports and lawmakers have raised about stock trades coinciding with official actions to help certain companies, saying Rep. Price’s brokers chose the stocks independently and all of his conduct was transparent.
After acknowledging that he asked his broker to buy stock in an Australian drug company, he told the Senate Finance Committee that he did not direct his broker to make other trades.
“To the best of my knowledge, I have not undertaken such actions,” he wrote in response to finance committee questions. “I have abided by and adhered to all ethics and conflict of interest rules applicable to me.”
An analysis of Rep. Price’s trades shows that he bought health stocks in 2007, the first year Congress financial disclosures are posted online. In 2011, the first year Rep. Price sat on the health subcommittee, he traded no health-related stocks, according to his financial disclosures filed with Congress.
That same year, members were facing public criticism because of a book detailing how they could use inside information and a “60 Minutes” investigation focused on how members and staff could legally use inside information to gain from their own stock trades.
In 2012, President Barack Obama signed the Stop Trading on Congressional Knowledge Act to rein in insider trading by members and require more disclosure. Public watchdog groups suggested at the time that the law would curb the practice.
That year, after his 1-year break in health care trades, Rep. Price resumed investing in health care companies.
Along with investments in technology, financial services, and retail stocks, he also bought and sold stock in companies that could be impacted by actions of his subcommittee, which has a role in determining rates the government pays under the Medicare program.
Health care firms spend heavily to influence members of Congress, lobbying on health matters, funding political campaigns, and seeking favor with Medicare officials who decide how much the program will pay for certain drugs and devices. The Food and Drug Administration holds similar power, approving or putting conditions on drug and device use.
Beyond his personal investments in health care companies, Rep. Price has also advocated their interests in letters to officials and proposed laws, government records show.
In 2012, disclosure records show Rep. Price sold stock in several drug firms, including more than $110,000 worth of Amgen stock. Amgen’s stock price had steadily climbed out of a recession-level slump, but Rep. Price’s sale came a few weeks before the company pleaded guilty to illegally marketing an anemia drug.
By 2013, the health subcommittee was at the center of a major conflict between Medicare, which sets Medicare Advantage rates, and the insurance industry. Medicare issued a notice early that year announcing its intention to reduce Medicare Advantage rates by 2.3 percent as part of a major cost-cutting initiative.
That prompted fierce lobbying by the health insurance industry. Members of Congress, including Rep. Price, wrote a letter to Marilyn Tavenner, then acting administrator for the Centers for Medicare & Medicaid Services, protesting the rate cut, saying the decrease would “disadvantage vulnerable beneficiaries with multiple chronic conditions.”
Ultimately, Medicare decided not to cut rates but instead, to increase them. Yet an hour before Medicare announced the change, a Height Securities analyst fired off a “flash” report to 200 clients that touched off a surge of trading.
The analyst’s report said a political deal was hatched on Capitol Hill to prevent the cuts as a condition for moving forward on Tavenner’s confirmation. Medicare officials increased rates by nearly 4 percent, a change that would positively impact the bottom lines of health insurance companies.
The SEC began looking for the leak’s source, and within weeks, FBI agents began interviewing staffers at the Ways and Means Committee, court records show.
They discovered communications between Sutter and a health care lobbyist. The HHS Inspector General also began a probe, and federal prosecutors briefly examined the matter as well.
As the case unfolded, Rep. Price bought more health care-related stocks, according to his financial disclosures. He has testified that his broker directed all of the trades, except for his investments in Innate Immunotherapeutics, an Australian company partly owned by Rep. Chris Collins (R-N.Y.), according to Collins’ disclosures. An HHS spokesman said Monday that Rep. Price held three broker-directed accounts.
Ethics experts have said that Rep. Price should have further distanced himself by placing his assets in a blind trust.
On April 30, 2013, Rep. Price bought $2,093 worth of stocks in Incyte, a company that develops cancer drugs; $2,076 in Onyx Pharmaceuticals, a drug maker that would soon merge with a larger drug firm; and $2,097 in Parexel International, a consultancy that helps drugs and devices win FDA approval, according to the financial disclosure records.
The same day, Rep. Price shed shares of Express Scripts, a drug management firm, and Danaher, which makes products hospitals and doctor’s offices using for testing and diagnostics. In August of that year, he bought a $2,429 stake in Jazz Pharmaceuticals, which makes sleep and cancer drugs.
On May 6, 2014, the SEC served its first subpoena for the Ways and Means Committee documents. The committee launched a vigorous fight, appealing a federal district judge’s ruling that it should comply with the SEC subpoena.
Rep. Price continued his health stock trades, including $1,000 to $15,000 in drug firms Amgen, Biogen, Bristol-Myers Squibb, Eli Lilly, and Pfizer. He also bought stock in Aetna, a major health insurer, and Athenahealth, which sells electronic medical record and medical billing software. In 2016, he also increased his investment in Innate Immunotherapeutics.
The purchase became controversial because both he and Collins bought stock in a private placement at a discounted price.
“You’re asking for trouble if you have access to nonpublic information about the health care industry and you’re buying and selling health care stocks,” Painter said.
Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.