User login
Official Newspaper of the American College of Surgeons
DOJ charges 412 in massive health care fraud bust
The U.S. Department of Justice (DOJ) has charged more than 400 health professionals with fraud for allegedly bilking $1.3 billion from the government through false billings to the Medicare, Medicaid, and TRICARE programs.
In all, 412 people, including 56 doctors, were charged across 41 federal districts for their participation in the alleged schemes, a large portion of which involved unnecessarily prescribing and distributing opioids to patients, according to a July 13 announcement by the DOJ. The agency called the enforcement the largest health care fraud action in DOJ history.
Defendants were charged in more than 20 states, including Florida, Michigan, Texas, California, Illinois, and Louisiana, where the federal government operates Medicare Fraud Strike Forces.
Southern Florida had the highest number of defendants, with 77 health professionals charged with a combined $141 million in false billings for alleged home health care, mental health services, and pharmacy fraud. In one case, an owner and operator of a Florida addiction treatment center is accused of actively recruiting addicted patients to move to South Florida so that coconspirators could bill for treatment and testing. In return, the coconspirators offered kickbacks to patients in the form of gift cards, free airline travel, trips to casinos and strip clubs, and drugs, according to the DOJ.
Health care fraud is not only a criminal act that costs billions of taxpayer dollars, it is an affront to all Americans who rely on national health care programs, Tom Price, MD, Secretary of Health and Human Services, said in a statement. “The United States is home to the world’s best medical professionals, but their ability to provide affordable, high-quality care to their patients is jeopardized every time a criminal commits health care fraud,” Dr. Price said.
[email protected]
On Twitter @legal_med
The U.S. Department of Justice (DOJ) has charged more than 400 health professionals with fraud for allegedly bilking $1.3 billion from the government through false billings to the Medicare, Medicaid, and TRICARE programs.
In all, 412 people, including 56 doctors, were charged across 41 federal districts for their participation in the alleged schemes, a large portion of which involved unnecessarily prescribing and distributing opioids to patients, according to a July 13 announcement by the DOJ. The agency called the enforcement the largest health care fraud action in DOJ history.
Defendants were charged in more than 20 states, including Florida, Michigan, Texas, California, Illinois, and Louisiana, where the federal government operates Medicare Fraud Strike Forces.
Southern Florida had the highest number of defendants, with 77 health professionals charged with a combined $141 million in false billings for alleged home health care, mental health services, and pharmacy fraud. In one case, an owner and operator of a Florida addiction treatment center is accused of actively recruiting addicted patients to move to South Florida so that coconspirators could bill for treatment and testing. In return, the coconspirators offered kickbacks to patients in the form of gift cards, free airline travel, trips to casinos and strip clubs, and drugs, according to the DOJ.
Health care fraud is not only a criminal act that costs billions of taxpayer dollars, it is an affront to all Americans who rely on national health care programs, Tom Price, MD, Secretary of Health and Human Services, said in a statement. “The United States is home to the world’s best medical professionals, but their ability to provide affordable, high-quality care to their patients is jeopardized every time a criminal commits health care fraud,” Dr. Price said.
[email protected]
On Twitter @legal_med
The U.S. Department of Justice (DOJ) has charged more than 400 health professionals with fraud for allegedly bilking $1.3 billion from the government through false billings to the Medicare, Medicaid, and TRICARE programs.
In all, 412 people, including 56 doctors, were charged across 41 federal districts for their participation in the alleged schemes, a large portion of which involved unnecessarily prescribing and distributing opioids to patients, according to a July 13 announcement by the DOJ. The agency called the enforcement the largest health care fraud action in DOJ history.
Defendants were charged in more than 20 states, including Florida, Michigan, Texas, California, Illinois, and Louisiana, where the federal government operates Medicare Fraud Strike Forces.
Southern Florida had the highest number of defendants, with 77 health professionals charged with a combined $141 million in false billings for alleged home health care, mental health services, and pharmacy fraud. In one case, an owner and operator of a Florida addiction treatment center is accused of actively recruiting addicted patients to move to South Florida so that coconspirators could bill for treatment and testing. In return, the coconspirators offered kickbacks to patients in the form of gift cards, free airline travel, trips to casinos and strip clubs, and drugs, according to the DOJ.
Health care fraud is not only a criminal act that costs billions of taxpayer dollars, it is an affront to all Americans who rely on national health care programs, Tom Price, MD, Secretary of Health and Human Services, said in a statement. “The United States is home to the world’s best medical professionals, but their ability to provide affordable, high-quality care to their patients is jeopardized every time a criminal commits health care fraud,” Dr. Price said.
[email protected]
On Twitter @legal_med
Senate GOP tweaks health care reform proposal, but it still lacks support
in the upper chamber of Congress.
The update, released July 13, includes a number of provisions to sweeten the pot for conservatives, but it does little to address the concerns of the moderates in the party, particularly of those who are worried about cuts to the Medicaid program.
The GOP leadership also is putting an additional $45 billion into the fight against opioid addiction. Plus, the revised bill includes a provision to allow tax credits to be used to purchase plans that offer only catastrophic coverage.
What the new revision doesn’t do is alter the cuts in the Medicaid program, leaving unchanged the plan to phase out the expansion of Medicaid in 2021 and complete it by 2024; allowing state governors to choose between block grants and per capita allotments to help pay for their Medicaid populations; and allowing states to institute a work requirement for nonpregnant, nondisabled, and nonelderly individuals eligible to receive Medicaid coverage.
Despite the changes, the bill still faces an uphill climb.
Sen. Rand Paul (R-Ky.), Sen. Susan Collins (R-Maine), and Sen. Rob Portman (R-Ohio) have publicly stated that they are opposed to the bill. With only a 52-seat majority and no Democrats expected to vote in favor of the bill, Senate Majority Leader Mitch McConnell (R-Ky.) needs at least 50 votes under budget reconciliation rules to pass the bill. (Vice President Mike Pence would be the tie-breaking vote if the GOP can get to 50 votes.) There are other moderate Republican senators who have opposed previous iterations of the bill, and it remains to be seen if the current tweaks will swing their votes.
The increased funding for the opioid crisis could draw some of the opposing GOP moderates. But, as Julius Hobson, a health care lobbyist with the Washington-based firm Polsinelli, noted, there is “not enough money to deal with” the opioid crisis.
The bill also has provisions allowing insurers to offer catastrophic coverage policies with minimal coverage alongside more comprehensive policies, an effort to bring in healthy younger individuals who may not want more comprehensive coverage. But those provisions have sparked pushback from state insurance directors, America’s Health Insurance Plans, and the Blue Cross Blue Shield Association.
“I can’t see how this bill got better in terms of health care access,” added Mr. Hobson, a former lobbyist with the American Medical Association.
AMA President David Barbe, MD, said that the tweaks do not do enough.
“The revised bill does not address the key concerns of physicians and patients regarding proposed Medicaid cuts and inadequate subsidies that will result in millions of Americans losing health insurance coverage,” Dr. Barbe said in a statement.
“The additional funding to address the opioid epidemic is a positive step; however, those suffering from substance use disorder have other health care needs that are not likely to be addressed if they lose coverage through a rollback of the Medicaid expansion,” he added. “While stabilizing the individual market is an initial step, more bipartisan collaboration is needed in the months ahead to improve the delivery and financing of health care.”
in the upper chamber of Congress.
The update, released July 13, includes a number of provisions to sweeten the pot for conservatives, but it does little to address the concerns of the moderates in the party, particularly of those who are worried about cuts to the Medicaid program.
The GOP leadership also is putting an additional $45 billion into the fight against opioid addiction. Plus, the revised bill includes a provision to allow tax credits to be used to purchase plans that offer only catastrophic coverage.
What the new revision doesn’t do is alter the cuts in the Medicaid program, leaving unchanged the plan to phase out the expansion of Medicaid in 2021 and complete it by 2024; allowing state governors to choose between block grants and per capita allotments to help pay for their Medicaid populations; and allowing states to institute a work requirement for nonpregnant, nondisabled, and nonelderly individuals eligible to receive Medicaid coverage.
Despite the changes, the bill still faces an uphill climb.
Sen. Rand Paul (R-Ky.), Sen. Susan Collins (R-Maine), and Sen. Rob Portman (R-Ohio) have publicly stated that they are opposed to the bill. With only a 52-seat majority and no Democrats expected to vote in favor of the bill, Senate Majority Leader Mitch McConnell (R-Ky.) needs at least 50 votes under budget reconciliation rules to pass the bill. (Vice President Mike Pence would be the tie-breaking vote if the GOP can get to 50 votes.) There are other moderate Republican senators who have opposed previous iterations of the bill, and it remains to be seen if the current tweaks will swing their votes.
The increased funding for the opioid crisis could draw some of the opposing GOP moderates. But, as Julius Hobson, a health care lobbyist with the Washington-based firm Polsinelli, noted, there is “not enough money to deal with” the opioid crisis.
The bill also has provisions allowing insurers to offer catastrophic coverage policies with minimal coverage alongside more comprehensive policies, an effort to bring in healthy younger individuals who may not want more comprehensive coverage. But those provisions have sparked pushback from state insurance directors, America’s Health Insurance Plans, and the Blue Cross Blue Shield Association.
“I can’t see how this bill got better in terms of health care access,” added Mr. Hobson, a former lobbyist with the American Medical Association.
AMA President David Barbe, MD, said that the tweaks do not do enough.
“The revised bill does not address the key concerns of physicians and patients regarding proposed Medicaid cuts and inadequate subsidies that will result in millions of Americans losing health insurance coverage,” Dr. Barbe said in a statement.
“The additional funding to address the opioid epidemic is a positive step; however, those suffering from substance use disorder have other health care needs that are not likely to be addressed if they lose coverage through a rollback of the Medicaid expansion,” he added. “While stabilizing the individual market is an initial step, more bipartisan collaboration is needed in the months ahead to improve the delivery and financing of health care.”
in the upper chamber of Congress.
The update, released July 13, includes a number of provisions to sweeten the pot for conservatives, but it does little to address the concerns of the moderates in the party, particularly of those who are worried about cuts to the Medicaid program.
The GOP leadership also is putting an additional $45 billion into the fight against opioid addiction. Plus, the revised bill includes a provision to allow tax credits to be used to purchase plans that offer only catastrophic coverage.
What the new revision doesn’t do is alter the cuts in the Medicaid program, leaving unchanged the plan to phase out the expansion of Medicaid in 2021 and complete it by 2024; allowing state governors to choose between block grants and per capita allotments to help pay for their Medicaid populations; and allowing states to institute a work requirement for nonpregnant, nondisabled, and nonelderly individuals eligible to receive Medicaid coverage.
Despite the changes, the bill still faces an uphill climb.
Sen. Rand Paul (R-Ky.), Sen. Susan Collins (R-Maine), and Sen. Rob Portman (R-Ohio) have publicly stated that they are opposed to the bill. With only a 52-seat majority and no Democrats expected to vote in favor of the bill, Senate Majority Leader Mitch McConnell (R-Ky.) needs at least 50 votes under budget reconciliation rules to pass the bill. (Vice President Mike Pence would be the tie-breaking vote if the GOP can get to 50 votes.) There are other moderate Republican senators who have opposed previous iterations of the bill, and it remains to be seen if the current tweaks will swing their votes.
The increased funding for the opioid crisis could draw some of the opposing GOP moderates. But, as Julius Hobson, a health care lobbyist with the Washington-based firm Polsinelli, noted, there is “not enough money to deal with” the opioid crisis.
The bill also has provisions allowing insurers to offer catastrophic coverage policies with minimal coverage alongside more comprehensive policies, an effort to bring in healthy younger individuals who may not want more comprehensive coverage. But those provisions have sparked pushback from state insurance directors, America’s Health Insurance Plans, and the Blue Cross Blue Shield Association.
“I can’t see how this bill got better in terms of health care access,” added Mr. Hobson, a former lobbyist with the American Medical Association.
AMA President David Barbe, MD, said that the tweaks do not do enough.
“The revised bill does not address the key concerns of physicians and patients regarding proposed Medicaid cuts and inadequate subsidies that will result in millions of Americans losing health insurance coverage,” Dr. Barbe said in a statement.
“The additional funding to address the opioid epidemic is a positive step; however, those suffering from substance use disorder have other health care needs that are not likely to be addressed if they lose coverage through a rollback of the Medicaid expansion,” he added. “While stabilizing the individual market is an initial step, more bipartisan collaboration is needed in the months ahead to improve the delivery and financing of health care.”
Physician liability in opioid deaths
Question: Regarding opioid deaths, which of the following is incorrect?
A. The term refers to accidental or intentional deaths caused mostly by heroin.
B. They are reaching epidemic proportions.
C. May form the basis for a wrongful death lawsuit.
D. May lead to loss of medical license.
E. The physician may face prosecution for homicide.
Answer: A. Opioids are a class of drugs that include the illegal drug heroin, as well as prescription drugs such as fentanyl, oxycodone, hydrocodone, codeine, and morphine. To be sure, opioid deaths occur in addicts from the deliberate or accidental use of heroin; but other opioids, especially painkillers, are also widely implicated. In addition, deaths have resulted from the careless, negligent, reckless, or wanton conduct of doctors who prescribe them without the proper indications or in inappropriate amounts, and then fail to provide careful follow-up.
Physicians may face both civil and criminal liabilities in such a situation. One remedy sought in wrongful death is a civil action, i.e., a malpractice lawsuit against the negligent doctor. The plaintiff is asserting that by violating community professional standards, the physician’s substandard conduct breached his duty of due care and was a proximate cause of the patient’s death. The evidentiary proof that is required to sustain such an allegation is “more probable than not” or “preponderance of evidence,” and expert medical testimony is typically necessary to establish the requisite standard of care and causation. Where there is gross negligence, i.e., egregious conduct that was reckless, the jury may award punitive damages.
Not infrequently, the wayward doctor faces triple liability: a civil lawsuit, state medical board action, and criminal prosecution for homicide. Given the publicity over soaring opioid death rates, one can expect aggressive prosecution of dealers and doctors alike.
This was not the first such case in Oklahoma. In 2014, a 71-year-old pain management doctor pleaded guilty to eight counts of second-degree murder in connection with several drug overdose deaths and will serve 8 years in prison. The doctor had reportedly prescribed more controlled drugs than any other physician in the state of Oklahoma. These drugs included hydrocodone, oxycodone, alprazolam, diazepam (Valium), and carisoprodol (Soma) – as many as 600 pills at a time. He allegedly accepted only cash payment for the office visits, and a review of his patient files revealed inadequate assessment of patient complaints or physical findings to justify the prescriptions.
Other states have been equally aggressive in prosecuting doctors over opioid deaths from reckless prescribing habits.
For the first time, New York in 2014 convicted a doctor of manslaughter in the overdose deaths of patients from oxycodone and alprazolam (Xanax). Some of the patients were prescribed as many as 500-800 pills over a 5- to 6-week period. The defendant, an anesthesiologist and pain management specialist, allegedly saw upward of 90 patients a day in his Queens weekend storefront clinic, charging them on a per-prescription basis. In his defense, he claimed that he was simply trying to help suffering people who misused medications and who misled him (“tough patients and good liars”).
Likewise, a Los Angeles–area doctor was recently convicted of second-degree murder for prescribing painkillers that killed three patients, and he was sentenced to 30 years to life in prison.
According to the Centers for Disease Control and Prevention, both drug overdose and opioid-involved deaths continue to increase in the United States.2 The majority of drug overdose deaths (more than 6 out of 10) involve an opioid, and the number has quadrupled since 1999.2 It has been estimated that more than 18,000 overdose deaths in 2014 involved prescription painkillers, while an additional 10,000 fatalities were attributed to heroin and 5,000 to fentanyl and other synthetic opioids. Overdose deaths exceed motor vehicle accidents as the leading cause of injury-related deaths. About 90 Americans die every day from an opioid overdose, and opioids have been forecast to kill 500,000 Americans over the next decade.
The CDC acknowledges that prescription opioids are a driving factor, noting that since 1999, the amount sold in the United States has nearly quadrupled, yet there has not been an overall change in the amount of pain that Americans report.
States such as Missouri, faced with the skyrocketing cost of treating the opioid epidemic, have sued the drug manufacturers, blaming them for their “campaign of fraud and deception.” At the same time, doctors have been deemed the “biggest culprit” for the opioid addiction epidemic, and one author has pointedly asserted that “by refusing to accept their inability to separate pain relief from addiction, physicians have long suffered the sin of hubris – and their patients have paid the price.”3
The U.S. Surgeon General recently took the historic step of writing to all American doctors asking for their help. And the American Medical Association has developed an educational module explaining the epidemic and how opioid misuse is linked to heroin addiction. The module also outlines risk-reducing steps when using opioids for pain relief.4
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. Some of the materials here have appeared in previous columns by the author. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Available at www.foxnews.om/health/2017/06/26/oklahoma-doctor-charged-in-opioid-deaths-5-patients.html. Accessed June 28, 2017.
2. Available at www.cdc.gov/drugoverdose/epidemic/index.html. Accessed June 28, 2017.
3. Available at www.thedailybeast.com/the-doctors-who-started-the-opioid-epidemic. Accessed June 27, 2017.
4. https://www.end-opioid-epidemic.org. Accessed July 5, 2017.
Question: Regarding opioid deaths, which of the following is incorrect?
A. The term refers to accidental or intentional deaths caused mostly by heroin.
B. They are reaching epidemic proportions.
C. May form the basis for a wrongful death lawsuit.
D. May lead to loss of medical license.
E. The physician may face prosecution for homicide.
Answer: A. Opioids are a class of drugs that include the illegal drug heroin, as well as prescription drugs such as fentanyl, oxycodone, hydrocodone, codeine, and morphine. To be sure, opioid deaths occur in addicts from the deliberate or accidental use of heroin; but other opioids, especially painkillers, are also widely implicated. In addition, deaths have resulted from the careless, negligent, reckless, or wanton conduct of doctors who prescribe them without the proper indications or in inappropriate amounts, and then fail to provide careful follow-up.
Physicians may face both civil and criminal liabilities in such a situation. One remedy sought in wrongful death is a civil action, i.e., a malpractice lawsuit against the negligent doctor. The plaintiff is asserting that by violating community professional standards, the physician’s substandard conduct breached his duty of due care and was a proximate cause of the patient’s death. The evidentiary proof that is required to sustain such an allegation is “more probable than not” or “preponderance of evidence,” and expert medical testimony is typically necessary to establish the requisite standard of care and causation. Where there is gross negligence, i.e., egregious conduct that was reckless, the jury may award punitive damages.
Not infrequently, the wayward doctor faces triple liability: a civil lawsuit, state medical board action, and criminal prosecution for homicide. Given the publicity over soaring opioid death rates, one can expect aggressive prosecution of dealers and doctors alike.
This was not the first such case in Oklahoma. In 2014, a 71-year-old pain management doctor pleaded guilty to eight counts of second-degree murder in connection with several drug overdose deaths and will serve 8 years in prison. The doctor had reportedly prescribed more controlled drugs than any other physician in the state of Oklahoma. These drugs included hydrocodone, oxycodone, alprazolam, diazepam (Valium), and carisoprodol (Soma) – as many as 600 pills at a time. He allegedly accepted only cash payment for the office visits, and a review of his patient files revealed inadequate assessment of patient complaints or physical findings to justify the prescriptions.
Other states have been equally aggressive in prosecuting doctors over opioid deaths from reckless prescribing habits.
For the first time, New York in 2014 convicted a doctor of manslaughter in the overdose deaths of patients from oxycodone and alprazolam (Xanax). Some of the patients were prescribed as many as 500-800 pills over a 5- to 6-week period. The defendant, an anesthesiologist and pain management specialist, allegedly saw upward of 90 patients a day in his Queens weekend storefront clinic, charging them on a per-prescription basis. In his defense, he claimed that he was simply trying to help suffering people who misused medications and who misled him (“tough patients and good liars”).
Likewise, a Los Angeles–area doctor was recently convicted of second-degree murder for prescribing painkillers that killed three patients, and he was sentenced to 30 years to life in prison.
According to the Centers for Disease Control and Prevention, both drug overdose and opioid-involved deaths continue to increase in the United States.2 The majority of drug overdose deaths (more than 6 out of 10) involve an opioid, and the number has quadrupled since 1999.2 It has been estimated that more than 18,000 overdose deaths in 2014 involved prescription painkillers, while an additional 10,000 fatalities were attributed to heroin and 5,000 to fentanyl and other synthetic opioids. Overdose deaths exceed motor vehicle accidents as the leading cause of injury-related deaths. About 90 Americans die every day from an opioid overdose, and opioids have been forecast to kill 500,000 Americans over the next decade.
The CDC acknowledges that prescription opioids are a driving factor, noting that since 1999, the amount sold in the United States has nearly quadrupled, yet there has not been an overall change in the amount of pain that Americans report.
States such as Missouri, faced with the skyrocketing cost of treating the opioid epidemic, have sued the drug manufacturers, blaming them for their “campaign of fraud and deception.” At the same time, doctors have been deemed the “biggest culprit” for the opioid addiction epidemic, and one author has pointedly asserted that “by refusing to accept their inability to separate pain relief from addiction, physicians have long suffered the sin of hubris – and their patients have paid the price.”3
The U.S. Surgeon General recently took the historic step of writing to all American doctors asking for their help. And the American Medical Association has developed an educational module explaining the epidemic and how opioid misuse is linked to heroin addiction. The module also outlines risk-reducing steps when using opioids for pain relief.4
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. Some of the materials here have appeared in previous columns by the author. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Available at www.foxnews.om/health/2017/06/26/oklahoma-doctor-charged-in-opioid-deaths-5-patients.html. Accessed June 28, 2017.
2. Available at www.cdc.gov/drugoverdose/epidemic/index.html. Accessed June 28, 2017.
3. Available at www.thedailybeast.com/the-doctors-who-started-the-opioid-epidemic. Accessed June 27, 2017.
4. https://www.end-opioid-epidemic.org. Accessed July 5, 2017.
Question: Regarding opioid deaths, which of the following is incorrect?
A. The term refers to accidental or intentional deaths caused mostly by heroin.
B. They are reaching epidemic proportions.
C. May form the basis for a wrongful death lawsuit.
D. May lead to loss of medical license.
E. The physician may face prosecution for homicide.
Answer: A. Opioids are a class of drugs that include the illegal drug heroin, as well as prescription drugs such as fentanyl, oxycodone, hydrocodone, codeine, and morphine. To be sure, opioid deaths occur in addicts from the deliberate or accidental use of heroin; but other opioids, especially painkillers, are also widely implicated. In addition, deaths have resulted from the careless, negligent, reckless, or wanton conduct of doctors who prescribe them without the proper indications or in inappropriate amounts, and then fail to provide careful follow-up.
Physicians may face both civil and criminal liabilities in such a situation. One remedy sought in wrongful death is a civil action, i.e., a malpractice lawsuit against the negligent doctor. The plaintiff is asserting that by violating community professional standards, the physician’s substandard conduct breached his duty of due care and was a proximate cause of the patient’s death. The evidentiary proof that is required to sustain such an allegation is “more probable than not” or “preponderance of evidence,” and expert medical testimony is typically necessary to establish the requisite standard of care and causation. Where there is gross negligence, i.e., egregious conduct that was reckless, the jury may award punitive damages.
Not infrequently, the wayward doctor faces triple liability: a civil lawsuit, state medical board action, and criminal prosecution for homicide. Given the publicity over soaring opioid death rates, one can expect aggressive prosecution of dealers and doctors alike.
This was not the first such case in Oklahoma. In 2014, a 71-year-old pain management doctor pleaded guilty to eight counts of second-degree murder in connection with several drug overdose deaths and will serve 8 years in prison. The doctor had reportedly prescribed more controlled drugs than any other physician in the state of Oklahoma. These drugs included hydrocodone, oxycodone, alprazolam, diazepam (Valium), and carisoprodol (Soma) – as many as 600 pills at a time. He allegedly accepted only cash payment for the office visits, and a review of his patient files revealed inadequate assessment of patient complaints or physical findings to justify the prescriptions.
Other states have been equally aggressive in prosecuting doctors over opioid deaths from reckless prescribing habits.
For the first time, New York in 2014 convicted a doctor of manslaughter in the overdose deaths of patients from oxycodone and alprazolam (Xanax). Some of the patients were prescribed as many as 500-800 pills over a 5- to 6-week period. The defendant, an anesthesiologist and pain management specialist, allegedly saw upward of 90 patients a day in his Queens weekend storefront clinic, charging them on a per-prescription basis. In his defense, he claimed that he was simply trying to help suffering people who misused medications and who misled him (“tough patients and good liars”).
Likewise, a Los Angeles–area doctor was recently convicted of second-degree murder for prescribing painkillers that killed three patients, and he was sentenced to 30 years to life in prison.
According to the Centers for Disease Control and Prevention, both drug overdose and opioid-involved deaths continue to increase in the United States.2 The majority of drug overdose deaths (more than 6 out of 10) involve an opioid, and the number has quadrupled since 1999.2 It has been estimated that more than 18,000 overdose deaths in 2014 involved prescription painkillers, while an additional 10,000 fatalities were attributed to heroin and 5,000 to fentanyl and other synthetic opioids. Overdose deaths exceed motor vehicle accidents as the leading cause of injury-related deaths. About 90 Americans die every day from an opioid overdose, and opioids have been forecast to kill 500,000 Americans over the next decade.
The CDC acknowledges that prescription opioids are a driving factor, noting that since 1999, the amount sold in the United States has nearly quadrupled, yet there has not been an overall change in the amount of pain that Americans report.
States such as Missouri, faced with the skyrocketing cost of treating the opioid epidemic, have sued the drug manufacturers, blaming them for their “campaign of fraud and deception.” At the same time, doctors have been deemed the “biggest culprit” for the opioid addiction epidemic, and one author has pointedly asserted that “by refusing to accept their inability to separate pain relief from addiction, physicians have long suffered the sin of hubris – and their patients have paid the price.”3
The U.S. Surgeon General recently took the historic step of writing to all American doctors asking for their help. And the American Medical Association has developed an educational module explaining the epidemic and how opioid misuse is linked to heroin addiction. The module also outlines risk-reducing steps when using opioids for pain relief.4
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. Some of the materials here have appeared in previous columns by the author. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Available at www.foxnews.om/health/2017/06/26/oklahoma-doctor-charged-in-opioid-deaths-5-patients.html. Accessed June 28, 2017.
2. Available at www.cdc.gov/drugoverdose/epidemic/index.html. Accessed June 28, 2017.
3. Available at www.thedailybeast.com/the-doctors-who-started-the-opioid-epidemic. Accessed June 27, 2017.
4. https://www.end-opioid-epidemic.org. Accessed July 5, 2017.
ABS wants feedback on 10-year exam alternatives
Many surgeons let out a collective cheer recently when the American Board of Surgery announced that, as of 2018, they will no longer have to take a high-stakes, pass-fail exam every 10 years to maintain certification.
The Board also announced, effective immediately, that it’s extending its continuing medical education (CME) reporting cycle from 3 to 5 years and reducing the number of required self-assessment CMEs – lectures and articles with a short quiz at the end – by a third. Surgeons now have to report 150 credits over 5 years, 50 from self-assessment CMEs. Under the old system, it was 90 credits over 3 years, 60 of which had to include self-assessment.
Alternative evaluation options
What’s on the minds of many, though, is what the alternatives to the 10-year exam will be. “That’s the $64,000 question,” said Emery Chen, MD, FACS, a general surgeon in private practice in Lancaster, Calif.
ABS is considering models that have worked well for other medical boards and hopes to roll out the alternative pathways at its winter meeting in January 2018, according to ABS Executive Director Frank Lewis, MD, FACS.
“I think it will probably be less stressful. People worry about the high-stakes exam. They don’t know what’s going to be on it, and they worry that failing could result in losing their staff privileges. We don’t think that’s particularly useful. The purpose is not to either pass or fail you but for you to learn the material,” Dr. Lewis said.
Meanwhile, surgeons who want to take the 10-year exam will still have the option.
Among the many options, surgeons could be given the two dozen journal articles deemed by experts to be the most cutting-edge for a given period, and quizzed on the material, with a chance to be re-quizzed as needed. There could be CME mini-courses or open-book exams on the areas most relevant to a surgeon’s practice and opportunities to relearn what might have been forgotten. Questions could be pushed out by smart phone to assess surgeons’ knowledge, with follow-up review material for incorrect answers and additional questions until the material is aced.
“Everything’s on the table,” said ABS at-large Board Member Tyler G. Hughes, MD, FACS, clinical faculty member at Kansas University, Salina.
A changing world
Like the country’s many other medical boards, ABS has been under pressure to make its MOC process less burdensome and more useful. A major problem is that general surgery isn’t very general anymore; it covers everything from transplants and bariatrics to trauma, endocrine, and vascular operations and more. Surgeons who have specialized have chafed at being examined every 10 years on areas that are no longer part of their practice and at questions about rare diseases such as multiple endocrine neoplasia I and II.
“They remember that one question that kind of stuck in their craw,” said Carol Scott-Conner, MD, FACS, emeritus professor and former head of surgery at the University of Iowa, Iowa City. At one point in her career, Dr. Scott-Conner helped write questions for the exam. “It wasn’t designed to be tricky, but, if you were not practicing in all these fields,” it was tough. “What you’re tested on should be relevant to your practice and help you get better at what you are doing. I think it’s a good change,” she said.
ABS has taken note. “There are a whole variety of things targeted at ways of maintaining longitudinal learning and providing some demonstration of it. We haven’t settled on any one of those. In fact, we may have more than one. We aim to get a good deal of feedback and see what our diplomates feel would be most helpful for them. It’s going to require a lot more detail, but hopefully it’s going to be more effective and more useful.” Dr. Lewis said.
The group will be at national and regional medical meetings this summer and fall to ask surgeons, in person, what they want their MOC system to be. There might be surveys as well, and ABS plans to be at the ACS Clinical Congress in October to solicit input.
The Board is looking to help surgeons with multiple certificates, whether from ABS or other boards, as well. “If we can create a more flexible assessment program that helps them maintain all their certifications, that’s what we want to do. We are looking at options to make it easier for them,” said ABS Director of Communications and Public Affairs Christine Shiffer, who noted that ABS will still likely require a 12-month case log every 10 years, even if surgeons opt out of the 10-year exam.
Will the new system have a negative impact on patient outcomes? After all, even specialized surgeons take night call sometimes. “ABS is responsible to surgeons but also the American public. If we say somebody is certified, it has to mean something. People would argue that there’s no real evidence that the recertification process improves surgical competence. Time will tell,” Dr. Scott-Conner said.
Dr. Lewis said he doesn’t know if the changes will save any money on MOC but noted that, instead of one $1,600 fee every 10 years, there’ll be an option to spread payments out.
Dr. Hughes is on the editorial advisory board of this publication.
Many surgeons let out a collective cheer recently when the American Board of Surgery announced that, as of 2018, they will no longer have to take a high-stakes, pass-fail exam every 10 years to maintain certification.
The Board also announced, effective immediately, that it’s extending its continuing medical education (CME) reporting cycle from 3 to 5 years and reducing the number of required self-assessment CMEs – lectures and articles with a short quiz at the end – by a third. Surgeons now have to report 150 credits over 5 years, 50 from self-assessment CMEs. Under the old system, it was 90 credits over 3 years, 60 of which had to include self-assessment.
Alternative evaluation options
What’s on the minds of many, though, is what the alternatives to the 10-year exam will be. “That’s the $64,000 question,” said Emery Chen, MD, FACS, a general surgeon in private practice in Lancaster, Calif.
ABS is considering models that have worked well for other medical boards and hopes to roll out the alternative pathways at its winter meeting in January 2018, according to ABS Executive Director Frank Lewis, MD, FACS.
“I think it will probably be less stressful. People worry about the high-stakes exam. They don’t know what’s going to be on it, and they worry that failing could result in losing their staff privileges. We don’t think that’s particularly useful. The purpose is not to either pass or fail you but for you to learn the material,” Dr. Lewis said.
Meanwhile, surgeons who want to take the 10-year exam will still have the option.
Among the many options, surgeons could be given the two dozen journal articles deemed by experts to be the most cutting-edge for a given period, and quizzed on the material, with a chance to be re-quizzed as needed. There could be CME mini-courses or open-book exams on the areas most relevant to a surgeon’s practice and opportunities to relearn what might have been forgotten. Questions could be pushed out by smart phone to assess surgeons’ knowledge, with follow-up review material for incorrect answers and additional questions until the material is aced.
“Everything’s on the table,” said ABS at-large Board Member Tyler G. Hughes, MD, FACS, clinical faculty member at Kansas University, Salina.
A changing world
Like the country’s many other medical boards, ABS has been under pressure to make its MOC process less burdensome and more useful. A major problem is that general surgery isn’t very general anymore; it covers everything from transplants and bariatrics to trauma, endocrine, and vascular operations and more. Surgeons who have specialized have chafed at being examined every 10 years on areas that are no longer part of their practice and at questions about rare diseases such as multiple endocrine neoplasia I and II.
“They remember that one question that kind of stuck in their craw,” said Carol Scott-Conner, MD, FACS, emeritus professor and former head of surgery at the University of Iowa, Iowa City. At one point in her career, Dr. Scott-Conner helped write questions for the exam. “It wasn’t designed to be tricky, but, if you were not practicing in all these fields,” it was tough. “What you’re tested on should be relevant to your practice and help you get better at what you are doing. I think it’s a good change,” she said.
ABS has taken note. “There are a whole variety of things targeted at ways of maintaining longitudinal learning and providing some demonstration of it. We haven’t settled on any one of those. In fact, we may have more than one. We aim to get a good deal of feedback and see what our diplomates feel would be most helpful for them. It’s going to require a lot more detail, but hopefully it’s going to be more effective and more useful.” Dr. Lewis said.
The group will be at national and regional medical meetings this summer and fall to ask surgeons, in person, what they want their MOC system to be. There might be surveys as well, and ABS plans to be at the ACS Clinical Congress in October to solicit input.
The Board is looking to help surgeons with multiple certificates, whether from ABS or other boards, as well. “If we can create a more flexible assessment program that helps them maintain all their certifications, that’s what we want to do. We are looking at options to make it easier for them,” said ABS Director of Communications and Public Affairs Christine Shiffer, who noted that ABS will still likely require a 12-month case log every 10 years, even if surgeons opt out of the 10-year exam.
Will the new system have a negative impact on patient outcomes? After all, even specialized surgeons take night call sometimes. “ABS is responsible to surgeons but also the American public. If we say somebody is certified, it has to mean something. People would argue that there’s no real evidence that the recertification process improves surgical competence. Time will tell,” Dr. Scott-Conner said.
Dr. Lewis said he doesn’t know if the changes will save any money on MOC but noted that, instead of one $1,600 fee every 10 years, there’ll be an option to spread payments out.
Dr. Hughes is on the editorial advisory board of this publication.
Many surgeons let out a collective cheer recently when the American Board of Surgery announced that, as of 2018, they will no longer have to take a high-stakes, pass-fail exam every 10 years to maintain certification.
The Board also announced, effective immediately, that it’s extending its continuing medical education (CME) reporting cycle from 3 to 5 years and reducing the number of required self-assessment CMEs – lectures and articles with a short quiz at the end – by a third. Surgeons now have to report 150 credits over 5 years, 50 from self-assessment CMEs. Under the old system, it was 90 credits over 3 years, 60 of which had to include self-assessment.
Alternative evaluation options
What’s on the minds of many, though, is what the alternatives to the 10-year exam will be. “That’s the $64,000 question,” said Emery Chen, MD, FACS, a general surgeon in private practice in Lancaster, Calif.
ABS is considering models that have worked well for other medical boards and hopes to roll out the alternative pathways at its winter meeting in January 2018, according to ABS Executive Director Frank Lewis, MD, FACS.
“I think it will probably be less stressful. People worry about the high-stakes exam. They don’t know what’s going to be on it, and they worry that failing could result in losing their staff privileges. We don’t think that’s particularly useful. The purpose is not to either pass or fail you but for you to learn the material,” Dr. Lewis said.
Meanwhile, surgeons who want to take the 10-year exam will still have the option.
Among the many options, surgeons could be given the two dozen journal articles deemed by experts to be the most cutting-edge for a given period, and quizzed on the material, with a chance to be re-quizzed as needed. There could be CME mini-courses or open-book exams on the areas most relevant to a surgeon’s practice and opportunities to relearn what might have been forgotten. Questions could be pushed out by smart phone to assess surgeons’ knowledge, with follow-up review material for incorrect answers and additional questions until the material is aced.
“Everything’s on the table,” said ABS at-large Board Member Tyler G. Hughes, MD, FACS, clinical faculty member at Kansas University, Salina.
A changing world
Like the country’s many other medical boards, ABS has been under pressure to make its MOC process less burdensome and more useful. A major problem is that general surgery isn’t very general anymore; it covers everything from transplants and bariatrics to trauma, endocrine, and vascular operations and more. Surgeons who have specialized have chafed at being examined every 10 years on areas that are no longer part of their practice and at questions about rare diseases such as multiple endocrine neoplasia I and II.
“They remember that one question that kind of stuck in their craw,” said Carol Scott-Conner, MD, FACS, emeritus professor and former head of surgery at the University of Iowa, Iowa City. At one point in her career, Dr. Scott-Conner helped write questions for the exam. “It wasn’t designed to be tricky, but, if you were not practicing in all these fields,” it was tough. “What you’re tested on should be relevant to your practice and help you get better at what you are doing. I think it’s a good change,” she said.
ABS has taken note. “There are a whole variety of things targeted at ways of maintaining longitudinal learning and providing some demonstration of it. We haven’t settled on any one of those. In fact, we may have more than one. We aim to get a good deal of feedback and see what our diplomates feel would be most helpful for them. It’s going to require a lot more detail, but hopefully it’s going to be more effective and more useful.” Dr. Lewis said.
The group will be at national and regional medical meetings this summer and fall to ask surgeons, in person, what they want their MOC system to be. There might be surveys as well, and ABS plans to be at the ACS Clinical Congress in October to solicit input.
The Board is looking to help surgeons with multiple certificates, whether from ABS or other boards, as well. “If we can create a more flexible assessment program that helps them maintain all their certifications, that’s what we want to do. We are looking at options to make it easier for them,” said ABS Director of Communications and Public Affairs Christine Shiffer, who noted that ABS will still likely require a 12-month case log every 10 years, even if surgeons opt out of the 10-year exam.
Will the new system have a negative impact on patient outcomes? After all, even specialized surgeons take night call sometimes. “ABS is responsible to surgeons but also the American public. If we say somebody is certified, it has to mean something. People would argue that there’s no real evidence that the recertification process improves surgical competence. Time will tell,” Dr. Scott-Conner said.
Dr. Lewis said he doesn’t know if the changes will save any money on MOC but noted that, instead of one $1,600 fee every 10 years, there’ll be an option to spread payments out.
Dr. Hughes is on the editorial advisory board of this publication.
Clinical trial: Mesh Type in Ventral Hernia Repair
The Mesh Type in Ventral Hernia Repair trial is an interventional study currently recruiting patients scheduled for open ventral hernia repair.
Half of ventral hernia repairs utilize synthetic mesh, while the other half use biologic mesh. There is currently little solid evidence that one mesh type is better than the other, although the study investigators hypothesize that biologic mesh is superior to synthetic.
Patients will be included in the trial if they are scheduled for open ventral hernia repair at LBJ General Hospital in Houston and are at least 18 years old. Patients will be excluded if they have an active infection, are unlikely to survive the next 2 years, are individuals in whom a prosthetic would not normally be placed, or are unlikely to follow up.
The primary endpoint goal is zero complications 1 year after the operation. Secondary endpoint goals include patient-centered outcomes, cost, Dindo-Clavien complications (grades I-IV), and to be complication free 3 years after the operation.
Recruitment began on March 27, 2017, and the study is expected to include 50 people. The primary study endpoint will be completed March 31, 2019, with the full study being completed March 31, 2022.
Find more information at the study page on Clinicaltrials.gov.
The Mesh Type in Ventral Hernia Repair trial is an interventional study currently recruiting patients scheduled for open ventral hernia repair.
Half of ventral hernia repairs utilize synthetic mesh, while the other half use biologic mesh. There is currently little solid evidence that one mesh type is better than the other, although the study investigators hypothesize that biologic mesh is superior to synthetic.
Patients will be included in the trial if they are scheduled for open ventral hernia repair at LBJ General Hospital in Houston and are at least 18 years old. Patients will be excluded if they have an active infection, are unlikely to survive the next 2 years, are individuals in whom a prosthetic would not normally be placed, or are unlikely to follow up.
The primary endpoint goal is zero complications 1 year after the operation. Secondary endpoint goals include patient-centered outcomes, cost, Dindo-Clavien complications (grades I-IV), and to be complication free 3 years after the operation.
Recruitment began on March 27, 2017, and the study is expected to include 50 people. The primary study endpoint will be completed March 31, 2019, with the full study being completed March 31, 2022.
Find more information at the study page on Clinicaltrials.gov.
The Mesh Type in Ventral Hernia Repair trial is an interventional study currently recruiting patients scheduled for open ventral hernia repair.
Half of ventral hernia repairs utilize synthetic mesh, while the other half use biologic mesh. There is currently little solid evidence that one mesh type is better than the other, although the study investigators hypothesize that biologic mesh is superior to synthetic.
Patients will be included in the trial if they are scheduled for open ventral hernia repair at LBJ General Hospital in Houston and are at least 18 years old. Patients will be excluded if they have an active infection, are unlikely to survive the next 2 years, are individuals in whom a prosthetic would not normally be placed, or are unlikely to follow up.
The primary endpoint goal is zero complications 1 year after the operation. Secondary endpoint goals include patient-centered outcomes, cost, Dindo-Clavien complications (grades I-IV), and to be complication free 3 years after the operation.
Recruitment began on March 27, 2017, and the study is expected to include 50 people. The primary study endpoint will be completed March 31, 2019, with the full study being completed March 31, 2022.
Find more information at the study page on Clinicaltrials.gov.
SUMMARY FROM CLINICALTRIALS.GOV
Nearly half of patients who stop taking opioids for 6 months resume use later
SAN DIEGO – A new study of medical records offers insights into the persistence of opioid use: Most patients who were prescribed opioid painkillers did not go back for a refill right away, but nearly half of patients who stopped taking the drugs for at least 6 months ended up using them again over a 3-year period.
“This key finding indicates that programs that address opioid use need to focus on long-term support and education to ensure that individuals do not become long-term users,” psychiatrist and lead author Shareh Ghani, MD, vice president and medical director of Magellan Health Services, San Francisco, said in an interview.
Researchers also found that opioid use appeared linked to three unexpected conditions – lipid disorders, hypertension, and sleep-wake disorders – and found that more than half of those who had at least two prescriptions for high-dose opioids kept taking the drugs over 18 months after an initial 90-day period.
Dr. Ghani and his colleague, Gowri Shetty, MPH, analyzed medical and pharmacy data from 2009-2012 for 2.5 million people. The participants, aged 20-64 years, came from across the United States and were part of a commercial health plan.
The researchers found that 21% had received one prescription for an opioid. Users considered at risk for persistent use – more than one prescription over 3 years – were more likely than were nonusers to have these characteristics: spondylosis and other back problems (odds ratio, 5.3), substance-related and addictive disorders (OR, 4.6), sleep-wake disorders (OR, 2.2), depressive disorders (OR, 1.7), headaches (OR, 2.1), and anxiety disorders (OR, 1.5.) The P values for all of those characteristics were less than .001.
They also found that patients who received certain kinds of treatment were at higher risk, compared with nonusers: those who were treated for substance abuse treatment (OR, 4.5), in emergency departments (OR, 3.2), with anesthesia (OR, 4.2), for mental health issues (OR, 2.3), and with surgery (OR, 2.0). The P values for all of those characteristics also were less than .001.
“The unexpected findings were the presence of lipid disorders, hypertension, and sleep-wake disorders. These diagnoses were not found in other literature,” Dr. Ghani said in an interview. “These conditions, however, are related to others that are known. For instance, a person with knee joint pain who is overweight – a known risk factor – may also have hypertension and lipid disorders.”
The researchers also discovered that 80% of patients who received an opioid prescription did not get a refill. Of those who had at least two prescriptions and a stable dose over an initial 90 days, 14% went on to have more prescriptions and a boost in dosage over 18 months, while 12% stayed the same and almost 74% took less.
But the situation was different for those with at least two prescriptions and a high dose (more than 120 mg) over an initial 90 days: 56% of them stayed at that level over 18 months.
The researchers also found that 48% of those who had stopped using opioids for at least 6 months went on to use them again. This high rate “suggests that physicians and patients need to be aware of the high risk of dependence and addiction for some individuals,” Dr. Ghani said. “Studying prescription fill behaviors and the persistence of prescription opioid users helps identify individuals at high risk for persistent use and may provide a better understanding of how to target interventions for inappropriate opioid use.”
The study has limitations. It does not indicate whether patients became substance abusers, nor does it provide details about opioids obtained illegally. Still, “we do know from literature and clinical experience that staying on prescription opioids may lead to dependence, escalation of dose, and increased risk of developing addictions that can lead to using street drugs like heroin,” Dr. Ghani said.
Magellan funded the study. Dr. Ghani reported no additional disclosures.
SAN DIEGO – A new study of medical records offers insights into the persistence of opioid use: Most patients who were prescribed opioid painkillers did not go back for a refill right away, but nearly half of patients who stopped taking the drugs for at least 6 months ended up using them again over a 3-year period.
“This key finding indicates that programs that address opioid use need to focus on long-term support and education to ensure that individuals do not become long-term users,” psychiatrist and lead author Shareh Ghani, MD, vice president and medical director of Magellan Health Services, San Francisco, said in an interview.
Researchers also found that opioid use appeared linked to three unexpected conditions – lipid disorders, hypertension, and sleep-wake disorders – and found that more than half of those who had at least two prescriptions for high-dose opioids kept taking the drugs over 18 months after an initial 90-day period.
Dr. Ghani and his colleague, Gowri Shetty, MPH, analyzed medical and pharmacy data from 2009-2012 for 2.5 million people. The participants, aged 20-64 years, came from across the United States and were part of a commercial health plan.
The researchers found that 21% had received one prescription for an opioid. Users considered at risk for persistent use – more than one prescription over 3 years – were more likely than were nonusers to have these characteristics: spondylosis and other back problems (odds ratio, 5.3), substance-related and addictive disorders (OR, 4.6), sleep-wake disorders (OR, 2.2), depressive disorders (OR, 1.7), headaches (OR, 2.1), and anxiety disorders (OR, 1.5.) The P values for all of those characteristics were less than .001.
They also found that patients who received certain kinds of treatment were at higher risk, compared with nonusers: those who were treated for substance abuse treatment (OR, 4.5), in emergency departments (OR, 3.2), with anesthesia (OR, 4.2), for mental health issues (OR, 2.3), and with surgery (OR, 2.0). The P values for all of those characteristics also were less than .001.
“The unexpected findings were the presence of lipid disorders, hypertension, and sleep-wake disorders. These diagnoses were not found in other literature,” Dr. Ghani said in an interview. “These conditions, however, are related to others that are known. For instance, a person with knee joint pain who is overweight – a known risk factor – may also have hypertension and lipid disorders.”
The researchers also discovered that 80% of patients who received an opioid prescription did not get a refill. Of those who had at least two prescriptions and a stable dose over an initial 90 days, 14% went on to have more prescriptions and a boost in dosage over 18 months, while 12% stayed the same and almost 74% took less.
But the situation was different for those with at least two prescriptions and a high dose (more than 120 mg) over an initial 90 days: 56% of them stayed at that level over 18 months.
The researchers also found that 48% of those who had stopped using opioids for at least 6 months went on to use them again. This high rate “suggests that physicians and patients need to be aware of the high risk of dependence and addiction for some individuals,” Dr. Ghani said. “Studying prescription fill behaviors and the persistence of prescription opioid users helps identify individuals at high risk for persistent use and may provide a better understanding of how to target interventions for inappropriate opioid use.”
The study has limitations. It does not indicate whether patients became substance abusers, nor does it provide details about opioids obtained illegally. Still, “we do know from literature and clinical experience that staying on prescription opioids may lead to dependence, escalation of dose, and increased risk of developing addictions that can lead to using street drugs like heroin,” Dr. Ghani said.
Magellan funded the study. Dr. Ghani reported no additional disclosures.
SAN DIEGO – A new study of medical records offers insights into the persistence of opioid use: Most patients who were prescribed opioid painkillers did not go back for a refill right away, but nearly half of patients who stopped taking the drugs for at least 6 months ended up using them again over a 3-year period.
“This key finding indicates that programs that address opioid use need to focus on long-term support and education to ensure that individuals do not become long-term users,” psychiatrist and lead author Shareh Ghani, MD, vice president and medical director of Magellan Health Services, San Francisco, said in an interview.
Researchers also found that opioid use appeared linked to three unexpected conditions – lipid disorders, hypertension, and sleep-wake disorders – and found that more than half of those who had at least two prescriptions for high-dose opioids kept taking the drugs over 18 months after an initial 90-day period.
Dr. Ghani and his colleague, Gowri Shetty, MPH, analyzed medical and pharmacy data from 2009-2012 for 2.5 million people. The participants, aged 20-64 years, came from across the United States and were part of a commercial health plan.
The researchers found that 21% had received one prescription for an opioid. Users considered at risk for persistent use – more than one prescription over 3 years – were more likely than were nonusers to have these characteristics: spondylosis and other back problems (odds ratio, 5.3), substance-related and addictive disorders (OR, 4.6), sleep-wake disorders (OR, 2.2), depressive disorders (OR, 1.7), headaches (OR, 2.1), and anxiety disorders (OR, 1.5.) The P values for all of those characteristics were less than .001.
They also found that patients who received certain kinds of treatment were at higher risk, compared with nonusers: those who were treated for substance abuse treatment (OR, 4.5), in emergency departments (OR, 3.2), with anesthesia (OR, 4.2), for mental health issues (OR, 2.3), and with surgery (OR, 2.0). The P values for all of those characteristics also were less than .001.
“The unexpected findings were the presence of lipid disorders, hypertension, and sleep-wake disorders. These diagnoses were not found in other literature,” Dr. Ghani said in an interview. “These conditions, however, are related to others that are known. For instance, a person with knee joint pain who is overweight – a known risk factor – may also have hypertension and lipid disorders.”
The researchers also discovered that 80% of patients who received an opioid prescription did not get a refill. Of those who had at least two prescriptions and a stable dose over an initial 90 days, 14% went on to have more prescriptions and a boost in dosage over 18 months, while 12% stayed the same and almost 74% took less.
But the situation was different for those with at least two prescriptions and a high dose (more than 120 mg) over an initial 90 days: 56% of them stayed at that level over 18 months.
The researchers also found that 48% of those who had stopped using opioids for at least 6 months went on to use them again. This high rate “suggests that physicians and patients need to be aware of the high risk of dependence and addiction for some individuals,” Dr. Ghani said. “Studying prescription fill behaviors and the persistence of prescription opioid users helps identify individuals at high risk for persistent use and may provide a better understanding of how to target interventions for inappropriate opioid use.”
The study has limitations. It does not indicate whether patients became substance abusers, nor does it provide details about opioids obtained illegally. Still, “we do know from literature and clinical experience that staying on prescription opioids may lead to dependence, escalation of dose, and increased risk of developing addictions that can lead to using street drugs like heroin,” Dr. Ghani said.
Magellan funded the study. Dr. Ghani reported no additional disclosures.
AT APA
Key clinical point:
Major finding: Forty-eight percent of patients who had stopped using opioids for at least 6 months went on to use them again.
Data source: An analysis of medical and pharmacy data from 2009-2012 for 2.5 million people aged 20-64 who were part of a commercial health plan.
Disclosures: Dr. Ghani is vice president and medical director of Magellan Health Services, which funded the study.
Federal exchanges attract fewer insurers for 2018
The number of health plans submitting applications to offer coverage on the federal insurance exchanges in 2018 was down 38% from last year, officials at the Centers for Medicare & Medicaid announced July 10. At this year’s initial filing deadline for fiscal year 2018, 141 qualified health plans had submitted applications to offer coverage in the 39 states that use healthcare.gov, the federally facilitated exchange platform, compared with 227 last year and 281 the year before, CMS officials said in a statement.
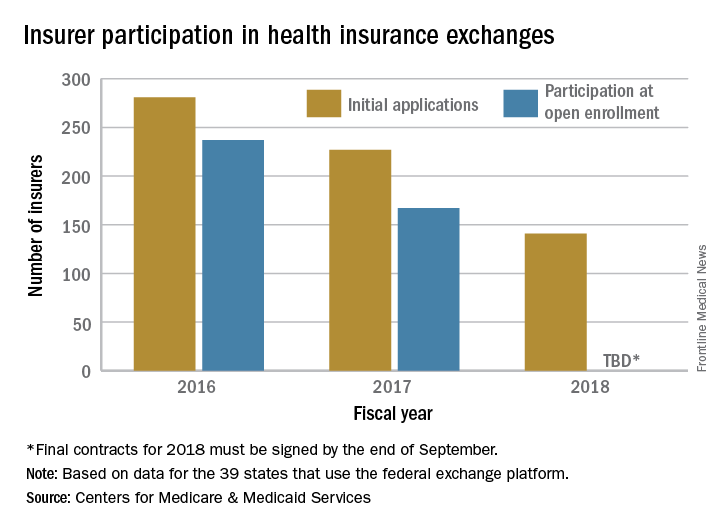
“This is further proof that the Affordable Care Act is failing,” CMS Administrator Seema Verma said in the statement. “Insurers continue to flee the Exchanges, causing Americans to lose their choice for health insurance or lose their coverage all together. These numbers are clear; the status quo is not working. The American people deserve health care choices and access to quality, affordable health care coverage.”
The number of health plans submitting applications to offer coverage on the federal insurance exchanges in 2018 was down 38% from last year, officials at the Centers for Medicare & Medicaid announced July 10. At this year’s initial filing deadline for fiscal year 2018, 141 qualified health plans had submitted applications to offer coverage in the 39 states that use healthcare.gov, the federally facilitated exchange platform, compared with 227 last year and 281 the year before, CMS officials said in a statement.

“This is further proof that the Affordable Care Act is failing,” CMS Administrator Seema Verma said in the statement. “Insurers continue to flee the Exchanges, causing Americans to lose their choice for health insurance or lose their coverage all together. These numbers are clear; the status quo is not working. The American people deserve health care choices and access to quality, affordable health care coverage.”
The number of health plans submitting applications to offer coverage on the federal insurance exchanges in 2018 was down 38% from last year, officials at the Centers for Medicare & Medicaid announced July 10. At this year’s initial filing deadline for fiscal year 2018, 141 qualified health plans had submitted applications to offer coverage in the 39 states that use healthcare.gov, the federally facilitated exchange platform, compared with 227 last year and 281 the year before, CMS officials said in a statement.

“This is further proof that the Affordable Care Act is failing,” CMS Administrator Seema Verma said in the statement. “Insurers continue to flee the Exchanges, causing Americans to lose their choice for health insurance or lose their coverage all together. These numbers are clear; the status quo is not working. The American people deserve health care choices and access to quality, affordable health care coverage.”
New federal health IT leadership, same goals
WASHINGTON – Although the leadership at the Office of the National Coordinator for Health Information Technology is new, the focus of the federal office – reducing physician burden and improving interoperability of electronic heath records – remains the same.
“One priority is on the whole question of burden of [EHR] usability,” said Don Rucker, MD, the new national coordinator, at a July 11 press briefing. “The other is interoperability. We’ve obviously spent a lot of money collectively in the country on these systems, and there’s a widespread dissatisfaction with the level of interoperability.”
“We are looking at documentation and the whole quality framework around value-based purchasing,” he said. “For a lot of practices now, this has become a challenge that we just have to think about what’s the win. At some point, the expense of complying with the quality measures is a much greater expense than the innate value of the quality measures. ”
EHRs “have become symbolic of physician administrative burden, but by no means are they the whole cause,” John Flemming, MD, ONC deputy assistant secretary for health technology reform, said at the briefing. “The physician, particularly in an independent practice, must manage the practice. So he or she is the CEO. They are also on the assembly line, seeing patients. Now with EHRs, they have to be the data input person as well. It’s time consuming.”
Dr. Fleming is a family physician from Louisiana and a former Republican member of congress.
Dr. Rucker acknowledged that reducing the burden of EHRs has been discussed for quite a long time now. He recalled beginning working with them in his private practice back in 1988 and figured, based on the quick rate of technological innovation demonstrated in Silicon Valley, the issues would be solved by 1992 or 1993 at the latest.
“Right now, [EHRs] are really about documentation, about billing, but that is a funny kind of beast,” he said. “Every other industry uses their enterprise computer software to do automation, to become more efficient. We are the only business that I am aware of to have used computers to become less efficient. ... I think part of what we are trying to do ... is let some of these newer technologies that will actually reduce costs, reduce variance, have those technologies have an entrée into some of these data collections that are out there.”
WASHINGTON – Although the leadership at the Office of the National Coordinator for Health Information Technology is new, the focus of the federal office – reducing physician burden and improving interoperability of electronic heath records – remains the same.
“One priority is on the whole question of burden of [EHR] usability,” said Don Rucker, MD, the new national coordinator, at a July 11 press briefing. “The other is interoperability. We’ve obviously spent a lot of money collectively in the country on these systems, and there’s a widespread dissatisfaction with the level of interoperability.”
“We are looking at documentation and the whole quality framework around value-based purchasing,” he said. “For a lot of practices now, this has become a challenge that we just have to think about what’s the win. At some point, the expense of complying with the quality measures is a much greater expense than the innate value of the quality measures. ”
EHRs “have become symbolic of physician administrative burden, but by no means are they the whole cause,” John Flemming, MD, ONC deputy assistant secretary for health technology reform, said at the briefing. “The physician, particularly in an independent practice, must manage the practice. So he or she is the CEO. They are also on the assembly line, seeing patients. Now with EHRs, they have to be the data input person as well. It’s time consuming.”
Dr. Fleming is a family physician from Louisiana and a former Republican member of congress.
Dr. Rucker acknowledged that reducing the burden of EHRs has been discussed for quite a long time now. He recalled beginning working with them in his private practice back in 1988 and figured, based on the quick rate of technological innovation demonstrated in Silicon Valley, the issues would be solved by 1992 or 1993 at the latest.
“Right now, [EHRs] are really about documentation, about billing, but that is a funny kind of beast,” he said. “Every other industry uses their enterprise computer software to do automation, to become more efficient. We are the only business that I am aware of to have used computers to become less efficient. ... I think part of what we are trying to do ... is let some of these newer technologies that will actually reduce costs, reduce variance, have those technologies have an entrée into some of these data collections that are out there.”
WASHINGTON – Although the leadership at the Office of the National Coordinator for Health Information Technology is new, the focus of the federal office – reducing physician burden and improving interoperability of electronic heath records – remains the same.
“One priority is on the whole question of burden of [EHR] usability,” said Don Rucker, MD, the new national coordinator, at a July 11 press briefing. “The other is interoperability. We’ve obviously spent a lot of money collectively in the country on these systems, and there’s a widespread dissatisfaction with the level of interoperability.”
“We are looking at documentation and the whole quality framework around value-based purchasing,” he said. “For a lot of practices now, this has become a challenge that we just have to think about what’s the win. At some point, the expense of complying with the quality measures is a much greater expense than the innate value of the quality measures. ”
EHRs “have become symbolic of physician administrative burden, but by no means are they the whole cause,” John Flemming, MD, ONC deputy assistant secretary for health technology reform, said at the briefing. “The physician, particularly in an independent practice, must manage the practice. So he or she is the CEO. They are also on the assembly line, seeing patients. Now with EHRs, they have to be the data input person as well. It’s time consuming.”
Dr. Fleming is a family physician from Louisiana and a former Republican member of congress.
Dr. Rucker acknowledged that reducing the burden of EHRs has been discussed for quite a long time now. He recalled beginning working with them in his private practice back in 1988 and figured, based on the quick rate of technological innovation demonstrated in Silicon Valley, the issues would be solved by 1992 or 1993 at the latest.
“Right now, [EHRs] are really about documentation, about billing, but that is a funny kind of beast,” he said. “Every other industry uses their enterprise computer software to do automation, to become more efficient. We are the only business that I am aware of to have used computers to become less efficient. ... I think part of what we are trying to do ... is let some of these newer technologies that will actually reduce costs, reduce variance, have those technologies have an entrée into some of these data collections that are out there.”
Point/Counterpoint: Is intraoperative drain placement essential during pancreatectomy?
Yes, placing drains is essential.
It’s important to look at the evidence in the literature, starting with a randomized controlled trial of 179 patients from 2001 that showed no reduction in death or complication rate associated with use of surgical intraperitoneal closed suction drainage (Ann. Surg. 2001;234:487-94). Interestingly, even though this study showed there was no benefit to using drains, they address the use of closed suction drainage. The investigators were not claiming all drains are unnecessary.
Why drain placement? The argument for drains includes evacuation of blood, pancreatic juice, bile, and chyle. In addition, assessing drainage can act as a warning sign for anastomotic leak or hemorrhage, so patients can potentially avoid additional interventions.
A study from University of Tokyo researchers found three drains were more effective than one. In addition, the investigators argued against early removal, pointing out that the risk for infection associated with drains only increased after day 10. The researchers were not only in favor of drains but in favor of multiple drains (World J Surg. 2016;40:1226-35).
A multicenter, randomized prospective trial conducted in the United States compared 68 patients with drains to 69 others without during pancreaticoduodenectomy. They reported an increase in the frequency and severity of complications when drains were omitted, including the number of grade 2 or greater complications. Furthermore, the safety monitoring board stopped the study early because mortality among patients in the drain group was 3%, compared with 12% in the no-drain group. (Ann. Surg. 2014:259:605-12).
Another set of researchers in Germany conducted a prospective, randomized study that favored omission of drains. However, a closer look at demographics shows that about one-fourth of participants had chronic pancreatitis, which is associated with a low risk of fistula (Ann Surg. 2016;263:440-9). In addition, they found no significant difference in fistula rates between patients who underwent pancreaticojejunostomy or pancreaticoduodenectomy. Interestingly, the authors noted that surgeons were reluctant to omit drains in many situations, even in a clinical trial context.
Furthermore, a systematic review of nine studies with nearly 3,000 patients suggests it is still necessary to place abdominal drains during pancreatic resection, researchers at the Medical College of Xi’an Jiaotong University in China reported (World J Gastroenterol. 2015;21:5719-34). The authors cited a significant increase in morbidity among patients in whom drains were omitted (odds ratio, 2.39).
Perhaps the cleverest way to address this controversy with drains during pancreatic resection comes from researchers at the University of Pennsylvania. They looked at surgical risk factors to gain a more nuanced view. They used the Fistula Risk Score (FRS) to stratify patients and re-examined the outcomes of the multicenter U.S. study I cited earlier that was stopped early because of differences in mortality rates. The University of Pennsylvania research found that FRS correlated well with outcomes, suggesting its use as a mitigation strategy in patients at moderate to high risk for developing clinically relevant postoperative pancreatic fistula (J Gastrointest Surg. 2015;19:21-30). In other words, they suggest routine prophylactic drainage for patients at moderate to high risk but also suggest that there may be no need for prophylactic drainage for negligible to low risk patients.
I would like to conclude that drains appear essential in many cases, including for moderate to higher risk patients. We need to tailor our practices as surgeons regarding placement of drains during pancreatectomy or pancreaticoduodenectomy. Our goal remains to create opportunities for prolonged survival and better quality of life for our patients.
Dr. Dervenis is head of the Department of Surgical Oncology at the Metropolitan Hospital in Athens, Greece. He is also chair of the hepato-pancreato-biliary surgical unit. He reported no disclosures.
No, drain placement is not always necessary.
Drain placement is not essential during all pancreatectomies. The practice is so historically entrenched in our specialty that I need you to check your dogma at the door and take a look at the data with an open mind.
There are many, many retrospective studies that examine drainage versus no drainage. All of these studies have shown the same thing: There is no difference in many outcomes, including mortality. There may be a difference in terms of complicated, postoperative fistulae, which are difficult to manage in patients who have drains.
Another prospective randomized trial from Germany compared the reintervention rate, an endpoint I like, among 439 patients (Ann Surg. 2016;264:528-37). There was no statistically significant difference between the drain or no drain groups, further suggesting that drains are not essential in all cases.
We also need to take a closer look at the study Dr. Dervenis cited (Ann Surg. 2014:259:605-12). It’s true this trial was stopped early because of a higher mortality rate of 12% in the no drain group, but the difference was not statistically significant (P = .097). There was no difference in the fistula rate.
When you look at these data, there are a couple of conclusions you can reach. One conclusion is that a trial of 130 people designed for 750 found something uniquely different that had never been reported in any retrospective series, including one by the study’s senior author that demonstrated drains are essential (HPB [Oxford]. 2011;13:503-10). Another conclusion is that drains are not needed, and there is a very good chance this is a false positive finding. Look at the reasons the 10 participants died. I’m not sure a drain would have helped some of the patients without them, and some of the patients who had drains still died.
A study looking at practice at my institution, MSKCC, shows that drains are still used about half the time, indicating they are not essential. (Ann. Surg. 2013 Dec;258[6]:1051-8). Drains were more commonly used for pancreaticoduodenectomy and when the surgeon thought there might be a problem, such as a soft gland, difficult time in the OR, or a small pancreatic duct.
If you look at these data, this pans out in every retrospective study I’ve seen comparing drain versus no drain – the patients with drains tend to have higher morbidity. Among the 1,122 resection patients in the MSKCC study, those without operative drains had significantly lower rates of grade 3 complications and overall morbidity and fewer readmissions and lower rates of grade 3 or higher pancreatic fistula. Mortality and reintervention rates were no different.
I also looked at our most recent data, between 2010 and 2015. Now we are using intraoperative drains even less often. They are not considered essential at this point.
There are multiple retrospective and well-designed prospective, randomized trials that show no benefit to routine drainage following pancreatectomy. Acceptance of randomized clinical data is slow, particularly when it flies in the face of what you were taught by your mentors over the past 40 or 50 years. I encourage you to look at these data carefully and utilize them in your practice.
Dr. Allen is associate director for clinical programs at David M. Rubenstein Center for Pancreatic Cancer Research and the Murray F. Brennan chair in surgery at Memorial Sloan-Kettering Cancer Center in New York City. He reported no disclosures.
Yes, placing drains is essential.
It’s important to look at the evidence in the literature, starting with a randomized controlled trial of 179 patients from 2001 that showed no reduction in death or complication rate associated with use of surgical intraperitoneal closed suction drainage (Ann. Surg. 2001;234:487-94). Interestingly, even though this study showed there was no benefit to using drains, they address the use of closed suction drainage. The investigators were not claiming all drains are unnecessary.
Why drain placement? The argument for drains includes evacuation of blood, pancreatic juice, bile, and chyle. In addition, assessing drainage can act as a warning sign for anastomotic leak or hemorrhage, so patients can potentially avoid additional interventions.
A study from University of Tokyo researchers found three drains were more effective than one. In addition, the investigators argued against early removal, pointing out that the risk for infection associated with drains only increased after day 10. The researchers were not only in favor of drains but in favor of multiple drains (World J Surg. 2016;40:1226-35).
A multicenter, randomized prospective trial conducted in the United States compared 68 patients with drains to 69 others without during pancreaticoduodenectomy. They reported an increase in the frequency and severity of complications when drains were omitted, including the number of grade 2 or greater complications. Furthermore, the safety monitoring board stopped the study early because mortality among patients in the drain group was 3%, compared with 12% in the no-drain group. (Ann. Surg. 2014:259:605-12).
Another set of researchers in Germany conducted a prospective, randomized study that favored omission of drains. However, a closer look at demographics shows that about one-fourth of participants had chronic pancreatitis, which is associated with a low risk of fistula (Ann Surg. 2016;263:440-9). In addition, they found no significant difference in fistula rates between patients who underwent pancreaticojejunostomy or pancreaticoduodenectomy. Interestingly, the authors noted that surgeons were reluctant to omit drains in many situations, even in a clinical trial context.
Furthermore, a systematic review of nine studies with nearly 3,000 patients suggests it is still necessary to place abdominal drains during pancreatic resection, researchers at the Medical College of Xi’an Jiaotong University in China reported (World J Gastroenterol. 2015;21:5719-34). The authors cited a significant increase in morbidity among patients in whom drains were omitted (odds ratio, 2.39).
Perhaps the cleverest way to address this controversy with drains during pancreatic resection comes from researchers at the University of Pennsylvania. They looked at surgical risk factors to gain a more nuanced view. They used the Fistula Risk Score (FRS) to stratify patients and re-examined the outcomes of the multicenter U.S. study I cited earlier that was stopped early because of differences in mortality rates. The University of Pennsylvania research found that FRS correlated well with outcomes, suggesting its use as a mitigation strategy in patients at moderate to high risk for developing clinically relevant postoperative pancreatic fistula (J Gastrointest Surg. 2015;19:21-30). In other words, they suggest routine prophylactic drainage for patients at moderate to high risk but also suggest that there may be no need for prophylactic drainage for negligible to low risk patients.
I would like to conclude that drains appear essential in many cases, including for moderate to higher risk patients. We need to tailor our practices as surgeons regarding placement of drains during pancreatectomy or pancreaticoduodenectomy. Our goal remains to create opportunities for prolonged survival and better quality of life for our patients.
Dr. Dervenis is head of the Department of Surgical Oncology at the Metropolitan Hospital in Athens, Greece. He is also chair of the hepato-pancreato-biliary surgical unit. He reported no disclosures.
No, drain placement is not always necessary.
Drain placement is not essential during all pancreatectomies. The practice is so historically entrenched in our specialty that I need you to check your dogma at the door and take a look at the data with an open mind.
There are many, many retrospective studies that examine drainage versus no drainage. All of these studies have shown the same thing: There is no difference in many outcomes, including mortality. There may be a difference in terms of complicated, postoperative fistulae, which are difficult to manage in patients who have drains.
Another prospective randomized trial from Germany compared the reintervention rate, an endpoint I like, among 439 patients (Ann Surg. 2016;264:528-37). There was no statistically significant difference between the drain or no drain groups, further suggesting that drains are not essential in all cases.
We also need to take a closer look at the study Dr. Dervenis cited (Ann Surg. 2014:259:605-12). It’s true this trial was stopped early because of a higher mortality rate of 12% in the no drain group, but the difference was not statistically significant (P = .097). There was no difference in the fistula rate.
When you look at these data, there are a couple of conclusions you can reach. One conclusion is that a trial of 130 people designed for 750 found something uniquely different that had never been reported in any retrospective series, including one by the study’s senior author that demonstrated drains are essential (HPB [Oxford]. 2011;13:503-10). Another conclusion is that drains are not needed, and there is a very good chance this is a false positive finding. Look at the reasons the 10 participants died. I’m not sure a drain would have helped some of the patients without them, and some of the patients who had drains still died.
A study looking at practice at my institution, MSKCC, shows that drains are still used about half the time, indicating they are not essential. (Ann. Surg. 2013 Dec;258[6]:1051-8). Drains were more commonly used for pancreaticoduodenectomy and when the surgeon thought there might be a problem, such as a soft gland, difficult time in the OR, or a small pancreatic duct.
If you look at these data, this pans out in every retrospective study I’ve seen comparing drain versus no drain – the patients with drains tend to have higher morbidity. Among the 1,122 resection patients in the MSKCC study, those without operative drains had significantly lower rates of grade 3 complications and overall morbidity and fewer readmissions and lower rates of grade 3 or higher pancreatic fistula. Mortality and reintervention rates were no different.
I also looked at our most recent data, between 2010 and 2015. Now we are using intraoperative drains even less often. They are not considered essential at this point.
There are multiple retrospective and well-designed prospective, randomized trials that show no benefit to routine drainage following pancreatectomy. Acceptance of randomized clinical data is slow, particularly when it flies in the face of what you were taught by your mentors over the past 40 or 50 years. I encourage you to look at these data carefully and utilize them in your practice.
Dr. Allen is associate director for clinical programs at David M. Rubenstein Center for Pancreatic Cancer Research and the Murray F. Brennan chair in surgery at Memorial Sloan-Kettering Cancer Center in New York City. He reported no disclosures.
Yes, placing drains is essential.
It’s important to look at the evidence in the literature, starting with a randomized controlled trial of 179 patients from 2001 that showed no reduction in death or complication rate associated with use of surgical intraperitoneal closed suction drainage (Ann. Surg. 2001;234:487-94). Interestingly, even though this study showed there was no benefit to using drains, they address the use of closed suction drainage. The investigators were not claiming all drains are unnecessary.
Why drain placement? The argument for drains includes evacuation of blood, pancreatic juice, bile, and chyle. In addition, assessing drainage can act as a warning sign for anastomotic leak or hemorrhage, so patients can potentially avoid additional interventions.
A study from University of Tokyo researchers found three drains were more effective than one. In addition, the investigators argued against early removal, pointing out that the risk for infection associated with drains only increased after day 10. The researchers were not only in favor of drains but in favor of multiple drains (World J Surg. 2016;40:1226-35).
A multicenter, randomized prospective trial conducted in the United States compared 68 patients with drains to 69 others without during pancreaticoduodenectomy. They reported an increase in the frequency and severity of complications when drains were omitted, including the number of grade 2 or greater complications. Furthermore, the safety monitoring board stopped the study early because mortality among patients in the drain group was 3%, compared with 12% in the no-drain group. (Ann. Surg. 2014:259:605-12).
Another set of researchers in Germany conducted a prospective, randomized study that favored omission of drains. However, a closer look at demographics shows that about one-fourth of participants had chronic pancreatitis, which is associated with a low risk of fistula (Ann Surg. 2016;263:440-9). In addition, they found no significant difference in fistula rates between patients who underwent pancreaticojejunostomy or pancreaticoduodenectomy. Interestingly, the authors noted that surgeons were reluctant to omit drains in many situations, even in a clinical trial context.
Furthermore, a systematic review of nine studies with nearly 3,000 patients suggests it is still necessary to place abdominal drains during pancreatic resection, researchers at the Medical College of Xi’an Jiaotong University in China reported (World J Gastroenterol. 2015;21:5719-34). The authors cited a significant increase in morbidity among patients in whom drains were omitted (odds ratio, 2.39).
Perhaps the cleverest way to address this controversy with drains during pancreatic resection comes from researchers at the University of Pennsylvania. They looked at surgical risk factors to gain a more nuanced view. They used the Fistula Risk Score (FRS) to stratify patients and re-examined the outcomes of the multicenter U.S. study I cited earlier that was stopped early because of differences in mortality rates. The University of Pennsylvania research found that FRS correlated well with outcomes, suggesting its use as a mitigation strategy in patients at moderate to high risk for developing clinically relevant postoperative pancreatic fistula (J Gastrointest Surg. 2015;19:21-30). In other words, they suggest routine prophylactic drainage for patients at moderate to high risk but also suggest that there may be no need for prophylactic drainage for negligible to low risk patients.
I would like to conclude that drains appear essential in many cases, including for moderate to higher risk patients. We need to tailor our practices as surgeons regarding placement of drains during pancreatectomy or pancreaticoduodenectomy. Our goal remains to create opportunities for prolonged survival and better quality of life for our patients.
Dr. Dervenis is head of the Department of Surgical Oncology at the Metropolitan Hospital in Athens, Greece. He is also chair of the hepato-pancreato-biliary surgical unit. He reported no disclosures.
No, drain placement is not always necessary.
Drain placement is not essential during all pancreatectomies. The practice is so historically entrenched in our specialty that I need you to check your dogma at the door and take a look at the data with an open mind.
There are many, many retrospective studies that examine drainage versus no drainage. All of these studies have shown the same thing: There is no difference in many outcomes, including mortality. There may be a difference in terms of complicated, postoperative fistulae, which are difficult to manage in patients who have drains.
Another prospective randomized trial from Germany compared the reintervention rate, an endpoint I like, among 439 patients (Ann Surg. 2016;264:528-37). There was no statistically significant difference between the drain or no drain groups, further suggesting that drains are not essential in all cases.
We also need to take a closer look at the study Dr. Dervenis cited (Ann Surg. 2014:259:605-12). It’s true this trial was stopped early because of a higher mortality rate of 12% in the no drain group, but the difference was not statistically significant (P = .097). There was no difference in the fistula rate.
When you look at these data, there are a couple of conclusions you can reach. One conclusion is that a trial of 130 people designed for 750 found something uniquely different that had never been reported in any retrospective series, including one by the study’s senior author that demonstrated drains are essential (HPB [Oxford]. 2011;13:503-10). Another conclusion is that drains are not needed, and there is a very good chance this is a false positive finding. Look at the reasons the 10 participants died. I’m not sure a drain would have helped some of the patients without them, and some of the patients who had drains still died.
A study looking at practice at my institution, MSKCC, shows that drains are still used about half the time, indicating they are not essential. (Ann. Surg. 2013 Dec;258[6]:1051-8). Drains were more commonly used for pancreaticoduodenectomy and when the surgeon thought there might be a problem, such as a soft gland, difficult time in the OR, or a small pancreatic duct.
If you look at these data, this pans out in every retrospective study I’ve seen comparing drain versus no drain – the patients with drains tend to have higher morbidity. Among the 1,122 resection patients in the MSKCC study, those without operative drains had significantly lower rates of grade 3 complications and overall morbidity and fewer readmissions and lower rates of grade 3 or higher pancreatic fistula. Mortality and reintervention rates were no different.
I also looked at our most recent data, between 2010 and 2015. Now we are using intraoperative drains even less often. They are not considered essential at this point.
There are multiple retrospective and well-designed prospective, randomized trials that show no benefit to routine drainage following pancreatectomy. Acceptance of randomized clinical data is slow, particularly when it flies in the face of what you were taught by your mentors over the past 40 or 50 years. I encourage you to look at these data carefully and utilize them in your practice.
Dr. Allen is associate director for clinical programs at David M. Rubenstein Center for Pancreatic Cancer Research and the Murray F. Brennan chair in surgery at Memorial Sloan-Kettering Cancer Center in New York City. He reported no disclosures.
Temporary tissue expanders optimize radiotherapy after mastectomy
LAS VEGAS – Radiation oncologists at the University of Texas MD Anderson Cancer Center, Houston, were able to complete 98% of their radiotherapy plans when women received temporary tissue expanders, instead of immediate reconstructions, at the time of skin-sparing mastectomy, in a series of 384 women, most with stage 2-3 breast cancer.
The expanders – saline-filled bags commonly used in plastic surgery to create new skin – were kept in place but deflated for radiotherapy, which allowed for optimal access to treatment fields; the final reconstruction, successful in 90% of women, came a median of 7 months following radiation.
“The shape and volume of the reconstruction” – and the need to avoid damaging the new breast – “got in the way of putting radiation where we wanted it to be. We ended up having bad radiotherapy plans, patients not getting skin-sparing mastectomies, and high probabilities of radiation complications to the reconstruction,” said investigator Eric Strom, MD, professor of radiation oncology at MD Anderson.
Radiologists and plastic and oncologic surgeons collaborated to try tissue expanders instead. “We wanted the advantage of skin-sparing mastectomy without the disadvantages” of immediate reconstruction, Dr. Strom said at the American Society of Breast Surgeons annual meeting.
With the new approach, “radiotherapy is superior. We don’t have to compromise our plans. I can put radiation everywhere it needs to be, without frying the heart” and almost completely avoiding the lungs, he said.
The 5-year rates of locoregional control, disease-free survival, and overall survival were 99.2%, 86.1%, and 92.4%, respectively, which “is extraordinary” in patients with stage 2-3 breast cancer, and likely due at least in part to optimal radiotherapy, he said.
Tissue expanders also keep the skin envelope open so it’s able to receive a graft at final reconstruction; abdominal skin doesn’t have to brought up to recreate the breast.
“This approach lessens negative interactions between breast reconstruction and [radiotherapy] and offers patients what they most desire: a high probability of freedom from cancer and optimal final aesthetic outcome,” said Zeina Ayoub, MD, a radiation oncology fellow at Anderson who presented the findings.
The median age of the women was 44 years, and almost all were node positive. Radiation was delivered to the chest wall and regional lymphatics, including the internal mammary chain.
Fifty women (13.0%) required explantation after radiation but before reconstruction, most commonly because of cellulitis; even so, more than half went on to final reconstruction.
Abdominal autologous reconstruction was the most common type, followed by latissimus dorsi–based reconstruction, and exchange of the tissue expander with an implant.
Dr. Ayoub and Dr. Strom had no relevant disclosures.
LAS VEGAS – Radiation oncologists at the University of Texas MD Anderson Cancer Center, Houston, were able to complete 98% of their radiotherapy plans when women received temporary tissue expanders, instead of immediate reconstructions, at the time of skin-sparing mastectomy, in a series of 384 women, most with stage 2-3 breast cancer.
The expanders – saline-filled bags commonly used in plastic surgery to create new skin – were kept in place but deflated for radiotherapy, which allowed for optimal access to treatment fields; the final reconstruction, successful in 90% of women, came a median of 7 months following radiation.
“The shape and volume of the reconstruction” – and the need to avoid damaging the new breast – “got in the way of putting radiation where we wanted it to be. We ended up having bad radiotherapy plans, patients not getting skin-sparing mastectomies, and high probabilities of radiation complications to the reconstruction,” said investigator Eric Strom, MD, professor of radiation oncology at MD Anderson.
Radiologists and plastic and oncologic surgeons collaborated to try tissue expanders instead. “We wanted the advantage of skin-sparing mastectomy without the disadvantages” of immediate reconstruction, Dr. Strom said at the American Society of Breast Surgeons annual meeting.
With the new approach, “radiotherapy is superior. We don’t have to compromise our plans. I can put radiation everywhere it needs to be, without frying the heart” and almost completely avoiding the lungs, he said.
The 5-year rates of locoregional control, disease-free survival, and overall survival were 99.2%, 86.1%, and 92.4%, respectively, which “is extraordinary” in patients with stage 2-3 breast cancer, and likely due at least in part to optimal radiotherapy, he said.
Tissue expanders also keep the skin envelope open so it’s able to receive a graft at final reconstruction; abdominal skin doesn’t have to brought up to recreate the breast.
“This approach lessens negative interactions between breast reconstruction and [radiotherapy] and offers patients what they most desire: a high probability of freedom from cancer and optimal final aesthetic outcome,” said Zeina Ayoub, MD, a radiation oncology fellow at Anderson who presented the findings.
The median age of the women was 44 years, and almost all were node positive. Radiation was delivered to the chest wall and regional lymphatics, including the internal mammary chain.
Fifty women (13.0%) required explantation after radiation but before reconstruction, most commonly because of cellulitis; even so, more than half went on to final reconstruction.
Abdominal autologous reconstruction was the most common type, followed by latissimus dorsi–based reconstruction, and exchange of the tissue expander with an implant.
Dr. Ayoub and Dr. Strom had no relevant disclosures.
LAS VEGAS – Radiation oncologists at the University of Texas MD Anderson Cancer Center, Houston, were able to complete 98% of their radiotherapy plans when women received temporary tissue expanders, instead of immediate reconstructions, at the time of skin-sparing mastectomy, in a series of 384 women, most with stage 2-3 breast cancer.
The expanders – saline-filled bags commonly used in plastic surgery to create new skin – were kept in place but deflated for radiotherapy, which allowed for optimal access to treatment fields; the final reconstruction, successful in 90% of women, came a median of 7 months following radiation.
“The shape and volume of the reconstruction” – and the need to avoid damaging the new breast – “got in the way of putting radiation where we wanted it to be. We ended up having bad radiotherapy plans, patients not getting skin-sparing mastectomies, and high probabilities of radiation complications to the reconstruction,” said investigator Eric Strom, MD, professor of radiation oncology at MD Anderson.
Radiologists and plastic and oncologic surgeons collaborated to try tissue expanders instead. “We wanted the advantage of skin-sparing mastectomy without the disadvantages” of immediate reconstruction, Dr. Strom said at the American Society of Breast Surgeons annual meeting.
With the new approach, “radiotherapy is superior. We don’t have to compromise our plans. I can put radiation everywhere it needs to be, without frying the heart” and almost completely avoiding the lungs, he said.
The 5-year rates of locoregional control, disease-free survival, and overall survival were 99.2%, 86.1%, and 92.4%, respectively, which “is extraordinary” in patients with stage 2-3 breast cancer, and likely due at least in part to optimal radiotherapy, he said.
Tissue expanders also keep the skin envelope open so it’s able to receive a graft at final reconstruction; abdominal skin doesn’t have to brought up to recreate the breast.
“This approach lessens negative interactions between breast reconstruction and [radiotherapy] and offers patients what they most desire: a high probability of freedom from cancer and optimal final aesthetic outcome,” said Zeina Ayoub, MD, a radiation oncology fellow at Anderson who presented the findings.
The median age of the women was 44 years, and almost all were node positive. Radiation was delivered to the chest wall and regional lymphatics, including the internal mammary chain.
Fifty women (13.0%) required explantation after radiation but before reconstruction, most commonly because of cellulitis; even so, more than half went on to final reconstruction.
Abdominal autologous reconstruction was the most common type, followed by latissimus dorsi–based reconstruction, and exchange of the tissue expander with an implant.
Dr. Ayoub and Dr. Strom had no relevant disclosures.
AT ASBS 2017
Key clinical point:
Major finding: The 5-year rates of locoregional control, disease-free survival, and overall survival were 99.2%, 86.1%, and 92.4%, respectively, likely due at least in part to optimal radiotherapy.
Data source: Review of 384 patients.
Disclosures: The investigators said they had no relevant disclosures.