User login
Eradicating H Pylori Cuts Long-Term Gastric Cancer Risk
Helicobacter pylori (HP) eradication reduced the risk of gastric noncardia adenocarcinoma in five Scandinavian countries, a population-based study in Gastroenterology reported. Risk became virtually similar to the background population from 11 years after treatment onward.
HP infection of the stomach is the main established risk factor for this tumor, but not much was known about the impact of eradication on long-term risk, particularly in Western populations, noted investigators led by Jesper Lagengren, MD, a gastrointestinal surgeon and professor at the Karolinksa Institutet in Stockholm, Sweden. Research with longer follow-up has reported contradictory results.
The study cohort included all adults treated for HP from 1995 to 2019 in Denmark, Finland, Iceland, Norway, and Sweden. Standardized incidence ratios (SIRs) with 95% confidence intervals (CIs) were calculated by comparing the gastric noncardia adenocarcinoma incidence in the study cohort with the incidence in the background population of the same age, sex, calendar period, and country.
The 659,592 treated participants were 54.3% women, 61.5% age 50 or younger, and had no serious comorbidities. They contributed to 5,480,873 person-years at risk with a mean follow-up of 8.3 years. Treatment consisted of a minimum one-week antibiotic regimen with two of amoxicillin, clarithromycin, or metronidazole, in combination with a proton pump inhibitor. This is the recommended regimen in the Nordic countries, where it achieves successful eradication in 90% of infected individuals.
Among these patients, 1311 developed gastric noncardia adenocarcinoma. Over as many as 24 years of follow-up, the SIR in treated HP patients was initially significantly higher than in the background population at 2.27 (95% confidence interval [CI], 2.10-2.44) at 1 to 5 years after treatment. By 6 to 10 years the SIR had dropped to 1.34 (1.21-1.48) and by 11 to 24 years it further fell to 1.11 (.98-1.27). In terms of observed vs expected cases, that translated to 702 vs 310 at 1 to 5 years, 374 vs 270 at 6 to 10 years, and 235 vs 211 from 11 to 24 years.
The results of the Nordic study align with systematic reviews from Asian populations indicating that eradication reduces the risk of gastric cancer, the authors said.
They noted gastric HP infection is the most prevalent bacterial infection worldwide, found in approximately 50% of the global population but with striking geographical variations in prevalence and virulence. The highest prevalence (>80%) and virulence are found in countries with low socioeconomic status and sanitation standards such as regions in Africa and Western Asia.
Gastric adenocarcinoma is the fourth-commonest cause of cancer-related death globally, leading to 660,000 deaths in 2022.
Lagergren and colleagues cited the need for research to delineate high-risk individuals who would benefit rom HP screening and eradication.
This study was supported by the Sjoberg Foundation, Nordic Cancer Union, Stockholm County Council, and Stockholm Cancer Society. The authors had no conflicts of interest to disclose.
Helicobacter pylori (HP) eradication reduced the risk of gastric noncardia adenocarcinoma in five Scandinavian countries, a population-based study in Gastroenterology reported. Risk became virtually similar to the background population from 11 years after treatment onward.
HP infection of the stomach is the main established risk factor for this tumor, but not much was known about the impact of eradication on long-term risk, particularly in Western populations, noted investigators led by Jesper Lagengren, MD, a gastrointestinal surgeon and professor at the Karolinksa Institutet in Stockholm, Sweden. Research with longer follow-up has reported contradictory results.
The study cohort included all adults treated for HP from 1995 to 2019 in Denmark, Finland, Iceland, Norway, and Sweden. Standardized incidence ratios (SIRs) with 95% confidence intervals (CIs) were calculated by comparing the gastric noncardia adenocarcinoma incidence in the study cohort with the incidence in the background population of the same age, sex, calendar period, and country.
The 659,592 treated participants were 54.3% women, 61.5% age 50 or younger, and had no serious comorbidities. They contributed to 5,480,873 person-years at risk with a mean follow-up of 8.3 years. Treatment consisted of a minimum one-week antibiotic regimen with two of amoxicillin, clarithromycin, or metronidazole, in combination with a proton pump inhibitor. This is the recommended regimen in the Nordic countries, where it achieves successful eradication in 90% of infected individuals.
Among these patients, 1311 developed gastric noncardia adenocarcinoma. Over as many as 24 years of follow-up, the SIR in treated HP patients was initially significantly higher than in the background population at 2.27 (95% confidence interval [CI], 2.10-2.44) at 1 to 5 years after treatment. By 6 to 10 years the SIR had dropped to 1.34 (1.21-1.48) and by 11 to 24 years it further fell to 1.11 (.98-1.27). In terms of observed vs expected cases, that translated to 702 vs 310 at 1 to 5 years, 374 vs 270 at 6 to 10 years, and 235 vs 211 from 11 to 24 years.
The results of the Nordic study align with systematic reviews from Asian populations indicating that eradication reduces the risk of gastric cancer, the authors said.
They noted gastric HP infection is the most prevalent bacterial infection worldwide, found in approximately 50% of the global population but with striking geographical variations in prevalence and virulence. The highest prevalence (>80%) and virulence are found in countries with low socioeconomic status and sanitation standards such as regions in Africa and Western Asia.
Gastric adenocarcinoma is the fourth-commonest cause of cancer-related death globally, leading to 660,000 deaths in 2022.
Lagergren and colleagues cited the need for research to delineate high-risk individuals who would benefit rom HP screening and eradication.
This study was supported by the Sjoberg Foundation, Nordic Cancer Union, Stockholm County Council, and Stockholm Cancer Society. The authors had no conflicts of interest to disclose.
Helicobacter pylori (HP) eradication reduced the risk of gastric noncardia adenocarcinoma in five Scandinavian countries, a population-based study in Gastroenterology reported. Risk became virtually similar to the background population from 11 years after treatment onward.
HP infection of the stomach is the main established risk factor for this tumor, but not much was known about the impact of eradication on long-term risk, particularly in Western populations, noted investigators led by Jesper Lagengren, MD, a gastrointestinal surgeon and professor at the Karolinksa Institutet in Stockholm, Sweden. Research with longer follow-up has reported contradictory results.
The study cohort included all adults treated for HP from 1995 to 2019 in Denmark, Finland, Iceland, Norway, and Sweden. Standardized incidence ratios (SIRs) with 95% confidence intervals (CIs) were calculated by comparing the gastric noncardia adenocarcinoma incidence in the study cohort with the incidence in the background population of the same age, sex, calendar period, and country.
The 659,592 treated participants were 54.3% women, 61.5% age 50 or younger, and had no serious comorbidities. They contributed to 5,480,873 person-years at risk with a mean follow-up of 8.3 years. Treatment consisted of a minimum one-week antibiotic regimen with two of amoxicillin, clarithromycin, or metronidazole, in combination with a proton pump inhibitor. This is the recommended regimen in the Nordic countries, where it achieves successful eradication in 90% of infected individuals.
Among these patients, 1311 developed gastric noncardia adenocarcinoma. Over as many as 24 years of follow-up, the SIR in treated HP patients was initially significantly higher than in the background population at 2.27 (95% confidence interval [CI], 2.10-2.44) at 1 to 5 years after treatment. By 6 to 10 years the SIR had dropped to 1.34 (1.21-1.48) and by 11 to 24 years it further fell to 1.11 (.98-1.27). In terms of observed vs expected cases, that translated to 702 vs 310 at 1 to 5 years, 374 vs 270 at 6 to 10 years, and 235 vs 211 from 11 to 24 years.
The results of the Nordic study align with systematic reviews from Asian populations indicating that eradication reduces the risk of gastric cancer, the authors said.
They noted gastric HP infection is the most prevalent bacterial infection worldwide, found in approximately 50% of the global population but with striking geographical variations in prevalence and virulence. The highest prevalence (>80%) and virulence are found in countries with low socioeconomic status and sanitation standards such as regions in Africa and Western Asia.
Gastric adenocarcinoma is the fourth-commonest cause of cancer-related death globally, leading to 660,000 deaths in 2022.
Lagergren and colleagues cited the need for research to delineate high-risk individuals who would benefit rom HP screening and eradication.
This study was supported by the Sjoberg Foundation, Nordic Cancer Union, Stockholm County Council, and Stockholm Cancer Society. The authors had no conflicts of interest to disclose.
FROM GASTROENTEROLOGY
Endoscopic Lifting Agents: AGA Issues New Clinical Practice Update
Published in Clinical Gastroenterology and Hepatology, the commentary reviews available agents and provides clinically relevant commentary on their indications and use — with the caveat that it is not a formal systematic review but rather empirical advice for endoscopists. No formal rating of the quality of evidence or strength of recommendations was performed.
Led by Tobias Zuchelli, MD, a clinical associate professor at Michigan State University and a gastroenterologist at the Henry Ford Health System in Detroit, the expert panel noted that endoscopists are increasingly resecting precancerous lesions and early cancers of the gastrointestinal tract.
“Although new endoscopic procedures have been developed, there had not been much in terms of high-quality guidance on lifting agents,” panelist Amit V. Patel, MD, a professor of medicine at Duke University and director of Endoscopy at Durham Veterans Affairs Medical Center in Durham, North Carolina, told GI & Hepatology News. “With our better understanding and use of techniques, this commentary was timely. It summarizes the available data on the topic and includes our clinical experiences.”
Filling that knowledge gap, the document reviews in detail the timing and methods of agent injection according to procedure type, including the dynamic needle approach, the empirical merits of different agents such as saline (with or without blue contrast) and viscous agents, as well as lift-enhancing assistive devices — for example, the ERBEJET 2 high-pressure water jet, an adjustable hydrosurgical device to facilitate lifting. A chart provides an at-a-glance summary of agents and their pros and cons.
“The feedback from gastroenterologists so far has been quite positive on social media and on GI channels,” Patel said.
Endoscopic resection has evolved from snare polypectomy to endoscopic mucosal resection (EMR) and now, endoscopic submucosal dissection (ESD). The primary benefit of submucosal lifting is the creation of a separating submucosal cushion between the lesion and muscularis propria (MP), which reduces the risk for immediate or delayed perforation of the muscle. Adding a contrast agent also demarcates lesion margins and stains the submucosa, which is fundamental to ESD and allows for assessment of MP injury during EMR.
For decades, homemade solutions were used to lift lesions before removal, with the sentinel agent being normal saline, later mixed with a blue contrast agent, usually indigo carmine or methylene blue. The authors noted that some endoscopists performing ESD start the submucosal injection and incision using a prepackaged viscous solution. “The endoscopist may continue with the viscous fluid or transition to saline or another less expensive solution,” they wrote.
Saline tends to dissipate more quickly than viscous solutions, however. In 2015, the polymer compound SIC-8000 became the first FDA-approved submucosal injection agent. Since then, several other fluids have come on the market, although homemade agents remain available.
Among the update’s recommendations, the fluid selected for EMR should be determined by lesion size, predicted histology, and endoscopist preference. Based on the US Multi-Society Task Force (USMSTF) on Colorectal Cancer, submucosal injection is optional for nonpedunculated colorectal lesions (NPCRLs) of intermediate size (10-19 mm).
Cold snare polypectomy without submucosal injection was later found to be non-inferior to other resection methods utilizing submucosal injection for NPCRLs ≤ 15 mm.
The update noted that the USMSTF considers EMR first-line therapy for most NPCRLs ≥ 20 mm and advocates viscous solutions as preferred, while the use of lifting agents for pedunculated polyps is generally at the discretion of the endoscopist.
For Patel, the main “clinical pearls” in the update are adding a contrast agent to normal saline, using a viscous agent for cold EMR, and manipulating the injection needle first tangentially and then dynamically toward the lumen to maximize separation of the lesion.
In terms of the ideal, an optimal lifting solution would be readily available, inexpensive, and premixed, providing a sustained submucosal cushion. “However, this ideal solution currently does not exist. Injection fluids should, therefore, be selected based on planned resection method, predicted histology, local expertise and preferences, and cost,” the panelists wrote.
Added Patel, “A lot of the agents out there check most of these boxes, but we’re hoping for further development toward the ideal.”
Offering a nonparticipant’s perspective on the overview, Wasseem Skef, MD, a gastroenterologist at UTHealth Houston, found the update very useful. “It always helps to have the literature summarized,” he told GI & Hepatology News. “It’s a pretty balanced review that pulls together the various options but allows people to stick to their preferred practice.”
In his practice, the lifting agent selected depends on the type of resection. “Viscous agents are generally more popular for EMR-type resections,” Skef said. One unanswered question, he noted, is whether adding a hemostatic agent would be superior to a viscous agent alone. “But overall, this is a nice summary of available agents. Gastroenterologists should consider these different options if doing procedures like EMR.”
This review was sponsored by the AGA Institute.
Zuchelli is a consultant for Boston Scientific. Patel consults for Medpace, Renexxion, and Sanofi. Skef reported having no relevant disclosures.
A version of this article appeared on Medscape.com .
Published in Clinical Gastroenterology and Hepatology, the commentary reviews available agents and provides clinically relevant commentary on their indications and use — with the caveat that it is not a formal systematic review but rather empirical advice for endoscopists. No formal rating of the quality of evidence or strength of recommendations was performed.
Led by Tobias Zuchelli, MD, a clinical associate professor at Michigan State University and a gastroenterologist at the Henry Ford Health System in Detroit, the expert panel noted that endoscopists are increasingly resecting precancerous lesions and early cancers of the gastrointestinal tract.
“Although new endoscopic procedures have been developed, there had not been much in terms of high-quality guidance on lifting agents,” panelist Amit V. Patel, MD, a professor of medicine at Duke University and director of Endoscopy at Durham Veterans Affairs Medical Center in Durham, North Carolina, told GI & Hepatology News. “With our better understanding and use of techniques, this commentary was timely. It summarizes the available data on the topic and includes our clinical experiences.”
Filling that knowledge gap, the document reviews in detail the timing and methods of agent injection according to procedure type, including the dynamic needle approach, the empirical merits of different agents such as saline (with or without blue contrast) and viscous agents, as well as lift-enhancing assistive devices — for example, the ERBEJET 2 high-pressure water jet, an adjustable hydrosurgical device to facilitate lifting. A chart provides an at-a-glance summary of agents and their pros and cons.
“The feedback from gastroenterologists so far has been quite positive on social media and on GI channels,” Patel said.
Endoscopic resection has evolved from snare polypectomy to endoscopic mucosal resection (EMR) and now, endoscopic submucosal dissection (ESD). The primary benefit of submucosal lifting is the creation of a separating submucosal cushion between the lesion and muscularis propria (MP), which reduces the risk for immediate or delayed perforation of the muscle. Adding a contrast agent also demarcates lesion margins and stains the submucosa, which is fundamental to ESD and allows for assessment of MP injury during EMR.
For decades, homemade solutions were used to lift lesions before removal, with the sentinel agent being normal saline, later mixed with a blue contrast agent, usually indigo carmine or methylene blue. The authors noted that some endoscopists performing ESD start the submucosal injection and incision using a prepackaged viscous solution. “The endoscopist may continue with the viscous fluid or transition to saline or another less expensive solution,” they wrote.
Saline tends to dissipate more quickly than viscous solutions, however. In 2015, the polymer compound SIC-8000 became the first FDA-approved submucosal injection agent. Since then, several other fluids have come on the market, although homemade agents remain available.
Among the update’s recommendations, the fluid selected for EMR should be determined by lesion size, predicted histology, and endoscopist preference. Based on the US Multi-Society Task Force (USMSTF) on Colorectal Cancer, submucosal injection is optional for nonpedunculated colorectal lesions (NPCRLs) of intermediate size (10-19 mm).
Cold snare polypectomy without submucosal injection was later found to be non-inferior to other resection methods utilizing submucosal injection for NPCRLs ≤ 15 mm.
The update noted that the USMSTF considers EMR first-line therapy for most NPCRLs ≥ 20 mm and advocates viscous solutions as preferred, while the use of lifting agents for pedunculated polyps is generally at the discretion of the endoscopist.
For Patel, the main “clinical pearls” in the update are adding a contrast agent to normal saline, using a viscous agent for cold EMR, and manipulating the injection needle first tangentially and then dynamically toward the lumen to maximize separation of the lesion.
In terms of the ideal, an optimal lifting solution would be readily available, inexpensive, and premixed, providing a sustained submucosal cushion. “However, this ideal solution currently does not exist. Injection fluids should, therefore, be selected based on planned resection method, predicted histology, local expertise and preferences, and cost,” the panelists wrote.
Added Patel, “A lot of the agents out there check most of these boxes, but we’re hoping for further development toward the ideal.”
Offering a nonparticipant’s perspective on the overview, Wasseem Skef, MD, a gastroenterologist at UTHealth Houston, found the update very useful. “It always helps to have the literature summarized,” he told GI & Hepatology News. “It’s a pretty balanced review that pulls together the various options but allows people to stick to their preferred practice.”
In his practice, the lifting agent selected depends on the type of resection. “Viscous agents are generally more popular for EMR-type resections,” Skef said. One unanswered question, he noted, is whether adding a hemostatic agent would be superior to a viscous agent alone. “But overall, this is a nice summary of available agents. Gastroenterologists should consider these different options if doing procedures like EMR.”
This review was sponsored by the AGA Institute.
Zuchelli is a consultant for Boston Scientific. Patel consults for Medpace, Renexxion, and Sanofi. Skef reported having no relevant disclosures.
A version of this article appeared on Medscape.com .
Published in Clinical Gastroenterology and Hepatology, the commentary reviews available agents and provides clinically relevant commentary on their indications and use — with the caveat that it is not a formal systematic review but rather empirical advice for endoscopists. No formal rating of the quality of evidence or strength of recommendations was performed.
Led by Tobias Zuchelli, MD, a clinical associate professor at Michigan State University and a gastroenterologist at the Henry Ford Health System in Detroit, the expert panel noted that endoscopists are increasingly resecting precancerous lesions and early cancers of the gastrointestinal tract.
“Although new endoscopic procedures have been developed, there had not been much in terms of high-quality guidance on lifting agents,” panelist Amit V. Patel, MD, a professor of medicine at Duke University and director of Endoscopy at Durham Veterans Affairs Medical Center in Durham, North Carolina, told GI & Hepatology News. “With our better understanding and use of techniques, this commentary was timely. It summarizes the available data on the topic and includes our clinical experiences.”
Filling that knowledge gap, the document reviews in detail the timing and methods of agent injection according to procedure type, including the dynamic needle approach, the empirical merits of different agents such as saline (with or without blue contrast) and viscous agents, as well as lift-enhancing assistive devices — for example, the ERBEJET 2 high-pressure water jet, an adjustable hydrosurgical device to facilitate lifting. A chart provides an at-a-glance summary of agents and their pros and cons.
“The feedback from gastroenterologists so far has been quite positive on social media and on GI channels,” Patel said.
Endoscopic resection has evolved from snare polypectomy to endoscopic mucosal resection (EMR) and now, endoscopic submucosal dissection (ESD). The primary benefit of submucosal lifting is the creation of a separating submucosal cushion between the lesion and muscularis propria (MP), which reduces the risk for immediate or delayed perforation of the muscle. Adding a contrast agent also demarcates lesion margins and stains the submucosa, which is fundamental to ESD and allows for assessment of MP injury during EMR.
For decades, homemade solutions were used to lift lesions before removal, with the sentinel agent being normal saline, later mixed with a blue contrast agent, usually indigo carmine or methylene blue. The authors noted that some endoscopists performing ESD start the submucosal injection and incision using a prepackaged viscous solution. “The endoscopist may continue with the viscous fluid or transition to saline or another less expensive solution,” they wrote.
Saline tends to dissipate more quickly than viscous solutions, however. In 2015, the polymer compound SIC-8000 became the first FDA-approved submucosal injection agent. Since then, several other fluids have come on the market, although homemade agents remain available.
Among the update’s recommendations, the fluid selected for EMR should be determined by lesion size, predicted histology, and endoscopist preference. Based on the US Multi-Society Task Force (USMSTF) on Colorectal Cancer, submucosal injection is optional for nonpedunculated colorectal lesions (NPCRLs) of intermediate size (10-19 mm).
Cold snare polypectomy without submucosal injection was later found to be non-inferior to other resection methods utilizing submucosal injection for NPCRLs ≤ 15 mm.
The update noted that the USMSTF considers EMR first-line therapy for most NPCRLs ≥ 20 mm and advocates viscous solutions as preferred, while the use of lifting agents for pedunculated polyps is generally at the discretion of the endoscopist.
For Patel, the main “clinical pearls” in the update are adding a contrast agent to normal saline, using a viscous agent for cold EMR, and manipulating the injection needle first tangentially and then dynamically toward the lumen to maximize separation of the lesion.
In terms of the ideal, an optimal lifting solution would be readily available, inexpensive, and premixed, providing a sustained submucosal cushion. “However, this ideal solution currently does not exist. Injection fluids should, therefore, be selected based on planned resection method, predicted histology, local expertise and preferences, and cost,” the panelists wrote.
Added Patel, “A lot of the agents out there check most of these boxes, but we’re hoping for further development toward the ideal.”
Offering a nonparticipant’s perspective on the overview, Wasseem Skef, MD, a gastroenterologist at UTHealth Houston, found the update very useful. “It always helps to have the literature summarized,” he told GI & Hepatology News. “It’s a pretty balanced review that pulls together the various options but allows people to stick to their preferred practice.”
In his practice, the lifting agent selected depends on the type of resection. “Viscous agents are generally more popular for EMR-type resections,” Skef said. One unanswered question, he noted, is whether adding a hemostatic agent would be superior to a viscous agent alone. “But overall, this is a nice summary of available agents. Gastroenterologists should consider these different options if doing procedures like EMR.”
This review was sponsored by the AGA Institute.
Zuchelli is a consultant for Boston Scientific. Patel consults for Medpace, Renexxion, and Sanofi. Skef reported having no relevant disclosures.
A version of this article appeared on Medscape.com .
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
FDA Issues Early Alert for Medtronic pH-Monitoring Capsules
The notice follows two letters sent in June to customers by the devices’ manufacturer Medtronic and its subsidiary Given Imaging Inc., recommending that customers using certain Bravo CF Capsule Delivery Devices (lot numbers below) for esophageal pH monitoring be removed from all sites of use and sale.
All three of the capsule models listed below are thought to pose a potential risk because the capsules fail to attach to the esophagus’s mucosal wall or to detach from the delivery device as intended owing to a misapplication of adhesive during manufacture. The devices transmit pH data to a recorder attached to the waist of the patient, who interacts with the recorder to indicate symptoms, thereby allowing the physician to compare the symptoms with the occurrence of reflux episodes.
Risks associated with the devices include aspiration/inhalation, perforation of the esophagus, obstruction of the airway, hemorrhage/blood loss/bleeding, laceration of the esophagus, a delay in diagnosis, and foreign bodies remaining in the patient.
Medtronic has reported 33 serious injuries but no deaths associated with the devices.
The lot numbers of the three affected units, which should be identified and quarantined immediately are:
- Bravo CF Capsule Delivery Device, 5-pk, Product Number FGS-0635, Unique Device Identifier-Device Identifier (UDI-DI) 07290101369707
- Bravo CF Capsule Delivery Device 5-pk, FGS-0635, UDI-DI 10613994000009
- Bravo CF Capsule Delivery Device 1-pk, FGS-0636, UDI-DI 07290101369714
These lot identifiers can be found on both the 5-pks’ FGS-0635 outer labels and on the 1-pk FGS-036 individual unit. Customers are advised to return all unused affected products to Medtronic for replacement or credit. In addition, they should pass on this notice to all those who need to be aware within their organizations or to any organizations to which the affected products have been distributed.
They are also advised to check the FDA recall website above for updates as it continues to review information about this potentially high-risk device issue.
Healthcare professionals with concerns or reports of adverse events can contact Medtronic at 800-448-3644 or MedWatch: The FDA Safety Information and Adverse Event Reporting Program.
A version of this article appeared on Medscape.com.
The notice follows two letters sent in June to customers by the devices’ manufacturer Medtronic and its subsidiary Given Imaging Inc., recommending that customers using certain Bravo CF Capsule Delivery Devices (lot numbers below) for esophageal pH monitoring be removed from all sites of use and sale.
All three of the capsule models listed below are thought to pose a potential risk because the capsules fail to attach to the esophagus’s mucosal wall or to detach from the delivery device as intended owing to a misapplication of adhesive during manufacture. The devices transmit pH data to a recorder attached to the waist of the patient, who interacts with the recorder to indicate symptoms, thereby allowing the physician to compare the symptoms with the occurrence of reflux episodes.
Risks associated with the devices include aspiration/inhalation, perforation of the esophagus, obstruction of the airway, hemorrhage/blood loss/bleeding, laceration of the esophagus, a delay in diagnosis, and foreign bodies remaining in the patient.
Medtronic has reported 33 serious injuries but no deaths associated with the devices.
The lot numbers of the three affected units, which should be identified and quarantined immediately are:
- Bravo CF Capsule Delivery Device, 5-pk, Product Number FGS-0635, Unique Device Identifier-Device Identifier (UDI-DI) 07290101369707
- Bravo CF Capsule Delivery Device 5-pk, FGS-0635, UDI-DI 10613994000009
- Bravo CF Capsule Delivery Device 1-pk, FGS-0636, UDI-DI 07290101369714
These lot identifiers can be found on both the 5-pks’ FGS-0635 outer labels and on the 1-pk FGS-036 individual unit. Customers are advised to return all unused affected products to Medtronic for replacement or credit. In addition, they should pass on this notice to all those who need to be aware within their organizations or to any organizations to which the affected products have been distributed.
They are also advised to check the FDA recall website above for updates as it continues to review information about this potentially high-risk device issue.
Healthcare professionals with concerns or reports of adverse events can contact Medtronic at 800-448-3644 or MedWatch: The FDA Safety Information and Adverse Event Reporting Program.
A version of this article appeared on Medscape.com.
The notice follows two letters sent in June to customers by the devices’ manufacturer Medtronic and its subsidiary Given Imaging Inc., recommending that customers using certain Bravo CF Capsule Delivery Devices (lot numbers below) for esophageal pH monitoring be removed from all sites of use and sale.
All three of the capsule models listed below are thought to pose a potential risk because the capsules fail to attach to the esophagus’s mucosal wall or to detach from the delivery device as intended owing to a misapplication of adhesive during manufacture. The devices transmit pH data to a recorder attached to the waist of the patient, who interacts with the recorder to indicate symptoms, thereby allowing the physician to compare the symptoms with the occurrence of reflux episodes.
Risks associated with the devices include aspiration/inhalation, perforation of the esophagus, obstruction of the airway, hemorrhage/blood loss/bleeding, laceration of the esophagus, a delay in diagnosis, and foreign bodies remaining in the patient.
Medtronic has reported 33 serious injuries but no deaths associated with the devices.
The lot numbers of the three affected units, which should be identified and quarantined immediately are:
- Bravo CF Capsule Delivery Device, 5-pk, Product Number FGS-0635, Unique Device Identifier-Device Identifier (UDI-DI) 07290101369707
- Bravo CF Capsule Delivery Device 5-pk, FGS-0635, UDI-DI 10613994000009
- Bravo CF Capsule Delivery Device 1-pk, FGS-0636, UDI-DI 07290101369714
These lot identifiers can be found on both the 5-pks’ FGS-0635 outer labels and on the 1-pk FGS-036 individual unit. Customers are advised to return all unused affected products to Medtronic for replacement or credit. In addition, they should pass on this notice to all those who need to be aware within their organizations or to any organizations to which the affected products have been distributed.
They are also advised to check the FDA recall website above for updates as it continues to review information about this potentially high-risk device issue.
Healthcare professionals with concerns or reports of adverse events can contact Medtronic at 800-448-3644 or MedWatch: The FDA Safety Information and Adverse Event Reporting Program.
A version of this article appeared on Medscape.com.
Celiac Blood Test Eliminates Need for Eating Gluten
Think your patient may have celiac disease? The harsh reality is that current diagnostic tests require patients to consume gluten for an accurate diagnosis, which poses challenges for individuals already avoiding gluten.
A more tolerable approach appears to be on the horizon.
“This is a simple and accurate test that can provide a diagnosis within a very short time frame, without the need for patients to continue eating gluten and feeling sick, or to wait months for a gastroscopy,” Olivia Moscatelli, PhD candidate, Tye-Din Lab, Walter and Eliza Hall Institute and University of Melbourne, Parkville, Australia, told GI & Hepatology News.
The study was published in Gastroenterology.
Most Cases Go Undiagnosed
Celiac disease is an autoimmune disorder triggered by gluten found in wheat, rye, and barley. The only available treatment is a strict, life-long gluten-free diet.
The global prevalence of celiac disease is estimated at around 1%-2%, with 50%-80% of cases either undiagnosed or diagnosed late. That’s because the current reliable diagnosis of celiac disease requires the intake of gluten, which may deter people from seeking a diagnosis.
In earlier work, the researchers, working with Robert Anderson, MBChB, BMedSc, PhD, AGAF, now with Novoviah Pharmaceuticals, made the unexpected discovery that interleukin-2 (IL-2) spiked in the blood of people with celiac disease shortly after they ate gluten.
But would this signal be present when no gluten had been consumed?
The team developed and tested a simple whole blood assay measuring IL-2 release (WBAIL- 2) for detecting gluten-specific T cells to aid in diagnosing celiac disease.
They collected blood samples from 181 volunteers — 75 with treated celiac disease on a gluten-free diet, 13 with active untreated celiac disease, 32 with nonceliac gluten sensitivity and 61 healthy controls. The blood samples were mixed with gluten in a test tube for a day to see if the IL-2 signal appeared.
The WBAIL-2 assay demonstrated high accuracy for celiac disease, even in patients following a strict gluten-free diet.
For patients with HLA-DQ2.5+ genetics, sensitivity was 90% and specificity was 95%, with lower sensitivity (56%) for patients with HLA-DQ8+ celiac disease.
The WBAIL-2 assay correlated strongly with the frequency of tetramer-positive gluten-specific CD4+ T cells used to diagnose celiac disease and monitor treatment effectiveness, and with serum IL-2 levels after gluten challenge.
The strength of the IL-2 signal correlated with the severity of a patient’s symptoms, “allowing us to predict how severely a person with celiac disease might react to gluten, without them actually having to eat it,” Moscatelli said in a news release.
“Current diagnostic practice involves a blood-based serology test followed by a confirmatory gastroscopy if positive. Both tests require the patient to eat gluten daily for 6-12 weeks prior for accurate results. We envision the new blood test (IL-2 whole blood assay) will replace the invasive gastroscopy as the confirmatory test following positive serology,” Moscatelli told GI & Hepatology News.
“In people already following a gluten-free diet, we propose they would have this new blood test done on two separate occasions and two positive results would be required for a celiac diagnosis. This would allow a large number of people who previously have been unable to go through the current diagnostic process to receive a diagnosis,” Moscatelli said.
Practice Changing Potential
A blood-based test that can accurately detect celiac disease without the need for a gluten challenge would be “welcome and practice changing,” said Christopher Cao, MD, director, Celiac Disease Program, Division of Gastroenterology, Mount Sinai Health System, New York City.
“A typical ‘gluten challenge’ involves eating the equivalent of 1-2 slices of bread daily for the course of 6 weeks, and this may be incredibly difficult for patients who have already been on a gluten-free diet prior to an official celiac disease diagnosis. Inability to perform a gluten challenge limits the ability to make an accurate celiac disease diagnosis,” Cao told GI & Hepatology News.
“This study shows that gluten-stimulated interleukin release 2 assays may correlate with the presence of pathogenic gluten-specific CD4+ T cell response in celiac disease,” Cao noted.
He cautioned that “further large cohort, multicenter prospective studies are needed to assess generalizability and may be helpful in evaluating the accuracy of WBAIL-2 in non-HLA DQ2.5 genotypes.”
Other considerations prior to implementation may include reproducibility across different laboratories and overall cost effectiveness, Cao said. “Ultimately in clinic, the role of WBAIL-2 will need to be better defined within the algorithm of celiac disease testing,” he added.
The Path Ahead
The researchers plan to test the performance of the IL-2 whole blood assay in a pediatric cohort, as well as in other countries to demonstrate the reproducibility of the test. In these studies, the test will likely be performed alongside the current diagnostic tests (serology and gastroscopy), Moscatelli told GI & Hepatology News.
“There are some validation studies starting in other countries already as many celiac clinicians globally are interested in bringing this test to their clinical practice. I believe the plan is to have this as an approved diagnostic test for celiac disease worldwide,” she said.
Novoviah Pharmaceuticals is managing the commercialization of the test, and the plan is to get it into clinical practice in the next 2 years, Moscatelli said.
The research was supported by Coeliac Australia, Novoviah Pharmaceuticals (who provided the proprietary test for this study), Beck Family Foundation, Butterfield Family, the Veith Foundation. A complete list of author disclosures is available with the original article. Cao had no relevant disclosures.
A version of this article appeared on Medscape.com.
Think your patient may have celiac disease? The harsh reality is that current diagnostic tests require patients to consume gluten for an accurate diagnosis, which poses challenges for individuals already avoiding gluten.
A more tolerable approach appears to be on the horizon.
“This is a simple and accurate test that can provide a diagnosis within a very short time frame, without the need for patients to continue eating gluten and feeling sick, or to wait months for a gastroscopy,” Olivia Moscatelli, PhD candidate, Tye-Din Lab, Walter and Eliza Hall Institute and University of Melbourne, Parkville, Australia, told GI & Hepatology News.
The study was published in Gastroenterology.
Most Cases Go Undiagnosed
Celiac disease is an autoimmune disorder triggered by gluten found in wheat, rye, and barley. The only available treatment is a strict, life-long gluten-free diet.
The global prevalence of celiac disease is estimated at around 1%-2%, with 50%-80% of cases either undiagnosed or diagnosed late. That’s because the current reliable diagnosis of celiac disease requires the intake of gluten, which may deter people from seeking a diagnosis.
In earlier work, the researchers, working with Robert Anderson, MBChB, BMedSc, PhD, AGAF, now with Novoviah Pharmaceuticals, made the unexpected discovery that interleukin-2 (IL-2) spiked in the blood of people with celiac disease shortly after they ate gluten.
But would this signal be present when no gluten had been consumed?
The team developed and tested a simple whole blood assay measuring IL-2 release (WBAIL- 2) for detecting gluten-specific T cells to aid in diagnosing celiac disease.
They collected blood samples from 181 volunteers — 75 with treated celiac disease on a gluten-free diet, 13 with active untreated celiac disease, 32 with nonceliac gluten sensitivity and 61 healthy controls. The blood samples were mixed with gluten in a test tube for a day to see if the IL-2 signal appeared.
The WBAIL-2 assay demonstrated high accuracy for celiac disease, even in patients following a strict gluten-free diet.
For patients with HLA-DQ2.5+ genetics, sensitivity was 90% and specificity was 95%, with lower sensitivity (56%) for patients with HLA-DQ8+ celiac disease.
The WBAIL-2 assay correlated strongly with the frequency of tetramer-positive gluten-specific CD4+ T cells used to diagnose celiac disease and monitor treatment effectiveness, and with serum IL-2 levels after gluten challenge.
The strength of the IL-2 signal correlated with the severity of a patient’s symptoms, “allowing us to predict how severely a person with celiac disease might react to gluten, without them actually having to eat it,” Moscatelli said in a news release.
“Current diagnostic practice involves a blood-based serology test followed by a confirmatory gastroscopy if positive. Both tests require the patient to eat gluten daily for 6-12 weeks prior for accurate results. We envision the new blood test (IL-2 whole blood assay) will replace the invasive gastroscopy as the confirmatory test following positive serology,” Moscatelli told GI & Hepatology News.
“In people already following a gluten-free diet, we propose they would have this new blood test done on two separate occasions and two positive results would be required for a celiac diagnosis. This would allow a large number of people who previously have been unable to go through the current diagnostic process to receive a diagnosis,” Moscatelli said.
Practice Changing Potential
A blood-based test that can accurately detect celiac disease without the need for a gluten challenge would be “welcome and practice changing,” said Christopher Cao, MD, director, Celiac Disease Program, Division of Gastroenterology, Mount Sinai Health System, New York City.
“A typical ‘gluten challenge’ involves eating the equivalent of 1-2 slices of bread daily for the course of 6 weeks, and this may be incredibly difficult for patients who have already been on a gluten-free diet prior to an official celiac disease diagnosis. Inability to perform a gluten challenge limits the ability to make an accurate celiac disease diagnosis,” Cao told GI & Hepatology News.
“This study shows that gluten-stimulated interleukin release 2 assays may correlate with the presence of pathogenic gluten-specific CD4+ T cell response in celiac disease,” Cao noted.
He cautioned that “further large cohort, multicenter prospective studies are needed to assess generalizability and may be helpful in evaluating the accuracy of WBAIL-2 in non-HLA DQ2.5 genotypes.”
Other considerations prior to implementation may include reproducibility across different laboratories and overall cost effectiveness, Cao said. “Ultimately in clinic, the role of WBAIL-2 will need to be better defined within the algorithm of celiac disease testing,” he added.
The Path Ahead
The researchers plan to test the performance of the IL-2 whole blood assay in a pediatric cohort, as well as in other countries to demonstrate the reproducibility of the test. In these studies, the test will likely be performed alongside the current diagnostic tests (serology and gastroscopy), Moscatelli told GI & Hepatology News.
“There are some validation studies starting in other countries already as many celiac clinicians globally are interested in bringing this test to their clinical practice. I believe the plan is to have this as an approved diagnostic test for celiac disease worldwide,” she said.
Novoviah Pharmaceuticals is managing the commercialization of the test, and the plan is to get it into clinical practice in the next 2 years, Moscatelli said.
The research was supported by Coeliac Australia, Novoviah Pharmaceuticals (who provided the proprietary test for this study), Beck Family Foundation, Butterfield Family, the Veith Foundation. A complete list of author disclosures is available with the original article. Cao had no relevant disclosures.
A version of this article appeared on Medscape.com.
Think your patient may have celiac disease? The harsh reality is that current diagnostic tests require patients to consume gluten for an accurate diagnosis, which poses challenges for individuals already avoiding gluten.
A more tolerable approach appears to be on the horizon.
“This is a simple and accurate test that can provide a diagnosis within a very short time frame, without the need for patients to continue eating gluten and feeling sick, or to wait months for a gastroscopy,” Olivia Moscatelli, PhD candidate, Tye-Din Lab, Walter and Eliza Hall Institute and University of Melbourne, Parkville, Australia, told GI & Hepatology News.
The study was published in Gastroenterology.
Most Cases Go Undiagnosed
Celiac disease is an autoimmune disorder triggered by gluten found in wheat, rye, and barley. The only available treatment is a strict, life-long gluten-free diet.
The global prevalence of celiac disease is estimated at around 1%-2%, with 50%-80% of cases either undiagnosed or diagnosed late. That’s because the current reliable diagnosis of celiac disease requires the intake of gluten, which may deter people from seeking a diagnosis.
In earlier work, the researchers, working with Robert Anderson, MBChB, BMedSc, PhD, AGAF, now with Novoviah Pharmaceuticals, made the unexpected discovery that interleukin-2 (IL-2) spiked in the blood of people with celiac disease shortly after they ate gluten.
But would this signal be present when no gluten had been consumed?
The team developed and tested a simple whole blood assay measuring IL-2 release (WBAIL- 2) for detecting gluten-specific T cells to aid in diagnosing celiac disease.
They collected blood samples from 181 volunteers — 75 with treated celiac disease on a gluten-free diet, 13 with active untreated celiac disease, 32 with nonceliac gluten sensitivity and 61 healthy controls. The blood samples were mixed with gluten in a test tube for a day to see if the IL-2 signal appeared.
The WBAIL-2 assay demonstrated high accuracy for celiac disease, even in patients following a strict gluten-free diet.
For patients with HLA-DQ2.5+ genetics, sensitivity was 90% and specificity was 95%, with lower sensitivity (56%) for patients with HLA-DQ8+ celiac disease.
The WBAIL-2 assay correlated strongly with the frequency of tetramer-positive gluten-specific CD4+ T cells used to diagnose celiac disease and monitor treatment effectiveness, and with serum IL-2 levels after gluten challenge.
The strength of the IL-2 signal correlated with the severity of a patient’s symptoms, “allowing us to predict how severely a person with celiac disease might react to gluten, without them actually having to eat it,” Moscatelli said in a news release.
“Current diagnostic practice involves a blood-based serology test followed by a confirmatory gastroscopy if positive. Both tests require the patient to eat gluten daily for 6-12 weeks prior for accurate results. We envision the new blood test (IL-2 whole blood assay) will replace the invasive gastroscopy as the confirmatory test following positive serology,” Moscatelli told GI & Hepatology News.
“In people already following a gluten-free diet, we propose they would have this new blood test done on two separate occasions and two positive results would be required for a celiac diagnosis. This would allow a large number of people who previously have been unable to go through the current diagnostic process to receive a diagnosis,” Moscatelli said.
Practice Changing Potential
A blood-based test that can accurately detect celiac disease without the need for a gluten challenge would be “welcome and practice changing,” said Christopher Cao, MD, director, Celiac Disease Program, Division of Gastroenterology, Mount Sinai Health System, New York City.
“A typical ‘gluten challenge’ involves eating the equivalent of 1-2 slices of bread daily for the course of 6 weeks, and this may be incredibly difficult for patients who have already been on a gluten-free diet prior to an official celiac disease diagnosis. Inability to perform a gluten challenge limits the ability to make an accurate celiac disease diagnosis,” Cao told GI & Hepatology News.
“This study shows that gluten-stimulated interleukin release 2 assays may correlate with the presence of pathogenic gluten-specific CD4+ T cell response in celiac disease,” Cao noted.
He cautioned that “further large cohort, multicenter prospective studies are needed to assess generalizability and may be helpful in evaluating the accuracy of WBAIL-2 in non-HLA DQ2.5 genotypes.”
Other considerations prior to implementation may include reproducibility across different laboratories and overall cost effectiveness, Cao said. “Ultimately in clinic, the role of WBAIL-2 will need to be better defined within the algorithm of celiac disease testing,” he added.
The Path Ahead
The researchers plan to test the performance of the IL-2 whole blood assay in a pediatric cohort, as well as in other countries to demonstrate the reproducibility of the test. In these studies, the test will likely be performed alongside the current diagnostic tests (serology and gastroscopy), Moscatelli told GI & Hepatology News.
“There are some validation studies starting in other countries already as many celiac clinicians globally are interested in bringing this test to their clinical practice. I believe the plan is to have this as an approved diagnostic test for celiac disease worldwide,” she said.
Novoviah Pharmaceuticals is managing the commercialization of the test, and the plan is to get it into clinical practice in the next 2 years, Moscatelli said.
The research was supported by Coeliac Australia, Novoviah Pharmaceuticals (who provided the proprietary test for this study), Beck Family Foundation, Butterfield Family, the Veith Foundation. A complete list of author disclosures is available with the original article. Cao had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM GASTROENTEROLOGY
Journal Highlights: January-April 2025
Esophagus/Motility
Carlson DA, et al. A Standardized Approach to Performing and Interpreting Functional Lumen Imaging Probe Panometry for Esophageal Motility Disorders: The Dallas Consensus. Gastroenterology. 2025 Feb. doi: 10.1053/j.gastro.2025.01.234.
Parkman HP, et al; NIDDK Gastroparesis Clinical Research Consortium. Characterization of Patients with Symptoms of Gastroparesis Having Frequent Emergency Department Visits and Hospitalizations. Clin Gastroenterol Hepatol. 2025 Apr. doi: 10.1016/j.cgh.2025.01.033.
Dellon ES, et al. Long-term Safety and Efficacy of Budesonide Oral Suspension for Eosinophilic Esophagitis: A 4-Year, Phase 3, Open-Label Study. Clin Gastroenterol Hepatol. 2025 Feb. doi: 10.1016/j.cgh.2024.12.024.
Small Bowel
Hård Af Segerstad EM, et al; TEDDY Study Group. Early Dietary Fiber Intake Reduces Celiac Disease Risk in Genetically Prone Children: Insights From the TEDDY Study. Gastroenterology. 2025 Feb. doi: 10.1053/j.gastro.2025.01.241.
Colon
Shaukat A, et al. AGA Clinical Practice Update on Current Role of Blood Tests for Colorectal Cancer Screening: Commentary. Clin Gastroenterol Hepatol. 2025 Apr. doi: 10.1016/j.cgh.2025.04.003.
Bergman D, et al. Cholecystectomy is a Risk Factor for Microscopic Colitis: A Nationwide Population-based Matched Case Control Study. Clin Gastroenterol Hepatol. 2025 Mar. doi: 10.1016/j.cgh.2024.12.032.
Inflammatory Bowel Disease
Ben-Horin S, et al; Israeli IBD Research Nucleus (IIRN). Capsule Endoscopy-Guided Proactive Treat-to-Target Versus Continued Standard Care in Patients With Quiescent Crohn’s Disease: A Randomized Controlled Trial. Gastroenterology. 2025 Mar. doi: 10.1053/j.gastro.2025.02.031.
Pancreas
Guilabert L, et al; ERICA Consortium. Impact of Fluid Therapy in the Emergency Department in Acute Pancreatitis: a posthoc analysis of the WATERFALL Trial. Clin Gastroenterol Hepatol. 2025 Apr. doi: 10.1016/j.cgh.2025.01.038.
Hepatology
Rhee H, et al. Noncontrast Magnetic Resonance Imaging vs Ultrasonography for Hepatocellular Carcinoma Surveillance: A Randomized, Single-Center Trial. Gastroenterology. 2025 Jan. doi: 10.1053/j.gastro.2024.12.035.
Kronsten VT, et al. Hepatic Encephalopathy: When Lactulose and Rifaximin Are Not Working. Gastroenterology. 2025 Jan. doi: 10.1053/j.gastro.2025.01.010.
Edelson JC, et al. Accuracy and Safety of Endoscopic Ultrasound–Guided Liver Biopsy in Patients with Metabolic Dysfunction–Associated Liver Disease. Tech Innov Gastrointest Endosc. 2025 Apr. doi: 10.1016/j.tige.2025.250918.
Miscellaneous
Martin J, et al. Practical and Impactful Tips for Private Industry Collaborations with Gastroenterology Practices. Clin Gastroenterol Hepatol. 2025 Mar. doi: 10.1016/j.cgh.2025.01.021.
Tejada, Natalia et al. Glucagon-like Peptide-1 Receptor Agonists Are Not Associated With Increased Incidence of Pneumonia After Endoscopic Procedures. Tech Innov Gastrointest Endosc. 2025 Apr. doi: 10.1016/j.tige.2025.250925.
Lazaridis KN, et al. Microplastics and Nanoplastics and the Digestive System. Gastro Hep Adv. 2025 May. doi: 10.1016/j.gastha.2025.100694.
Dr. Trieu is assistant professor of medicine, interventional endoscopy, in the Division of Gastroenterology at Washington University in St. Louis School of Medicine, Missouri.
Esophagus/Motility
Carlson DA, et al. A Standardized Approach to Performing and Interpreting Functional Lumen Imaging Probe Panometry for Esophageal Motility Disorders: The Dallas Consensus. Gastroenterology. 2025 Feb. doi: 10.1053/j.gastro.2025.01.234.
Parkman HP, et al; NIDDK Gastroparesis Clinical Research Consortium. Characterization of Patients with Symptoms of Gastroparesis Having Frequent Emergency Department Visits and Hospitalizations. Clin Gastroenterol Hepatol. 2025 Apr. doi: 10.1016/j.cgh.2025.01.033.
Dellon ES, et al. Long-term Safety and Efficacy of Budesonide Oral Suspension for Eosinophilic Esophagitis: A 4-Year, Phase 3, Open-Label Study. Clin Gastroenterol Hepatol. 2025 Feb. doi: 10.1016/j.cgh.2024.12.024.
Small Bowel
Hård Af Segerstad EM, et al; TEDDY Study Group. Early Dietary Fiber Intake Reduces Celiac Disease Risk in Genetically Prone Children: Insights From the TEDDY Study. Gastroenterology. 2025 Feb. doi: 10.1053/j.gastro.2025.01.241.
Colon
Shaukat A, et al. AGA Clinical Practice Update on Current Role of Blood Tests for Colorectal Cancer Screening: Commentary. Clin Gastroenterol Hepatol. 2025 Apr. doi: 10.1016/j.cgh.2025.04.003.
Bergman D, et al. Cholecystectomy is a Risk Factor for Microscopic Colitis: A Nationwide Population-based Matched Case Control Study. Clin Gastroenterol Hepatol. 2025 Mar. doi: 10.1016/j.cgh.2024.12.032.
Inflammatory Bowel Disease
Ben-Horin S, et al; Israeli IBD Research Nucleus (IIRN). Capsule Endoscopy-Guided Proactive Treat-to-Target Versus Continued Standard Care in Patients With Quiescent Crohn’s Disease: A Randomized Controlled Trial. Gastroenterology. 2025 Mar. doi: 10.1053/j.gastro.2025.02.031.
Pancreas
Guilabert L, et al; ERICA Consortium. Impact of Fluid Therapy in the Emergency Department in Acute Pancreatitis: a posthoc analysis of the WATERFALL Trial. Clin Gastroenterol Hepatol. 2025 Apr. doi: 10.1016/j.cgh.2025.01.038.
Hepatology
Rhee H, et al. Noncontrast Magnetic Resonance Imaging vs Ultrasonography for Hepatocellular Carcinoma Surveillance: A Randomized, Single-Center Trial. Gastroenterology. 2025 Jan. doi: 10.1053/j.gastro.2024.12.035.
Kronsten VT, et al. Hepatic Encephalopathy: When Lactulose and Rifaximin Are Not Working. Gastroenterology. 2025 Jan. doi: 10.1053/j.gastro.2025.01.010.
Edelson JC, et al. Accuracy and Safety of Endoscopic Ultrasound–Guided Liver Biopsy in Patients with Metabolic Dysfunction–Associated Liver Disease. Tech Innov Gastrointest Endosc. 2025 Apr. doi: 10.1016/j.tige.2025.250918.
Miscellaneous
Martin J, et al. Practical and Impactful Tips for Private Industry Collaborations with Gastroenterology Practices. Clin Gastroenterol Hepatol. 2025 Mar. doi: 10.1016/j.cgh.2025.01.021.
Tejada, Natalia et al. Glucagon-like Peptide-1 Receptor Agonists Are Not Associated With Increased Incidence of Pneumonia After Endoscopic Procedures. Tech Innov Gastrointest Endosc. 2025 Apr. doi: 10.1016/j.tige.2025.250925.
Lazaridis KN, et al. Microplastics and Nanoplastics and the Digestive System. Gastro Hep Adv. 2025 May. doi: 10.1016/j.gastha.2025.100694.
Dr. Trieu is assistant professor of medicine, interventional endoscopy, in the Division of Gastroenterology at Washington University in St. Louis School of Medicine, Missouri.
Esophagus/Motility
Carlson DA, et al. A Standardized Approach to Performing and Interpreting Functional Lumen Imaging Probe Panometry for Esophageal Motility Disorders: The Dallas Consensus. Gastroenterology. 2025 Feb. doi: 10.1053/j.gastro.2025.01.234.
Parkman HP, et al; NIDDK Gastroparesis Clinical Research Consortium. Characterization of Patients with Symptoms of Gastroparesis Having Frequent Emergency Department Visits and Hospitalizations. Clin Gastroenterol Hepatol. 2025 Apr. doi: 10.1016/j.cgh.2025.01.033.
Dellon ES, et al. Long-term Safety and Efficacy of Budesonide Oral Suspension for Eosinophilic Esophagitis: A 4-Year, Phase 3, Open-Label Study. Clin Gastroenterol Hepatol. 2025 Feb. doi: 10.1016/j.cgh.2024.12.024.
Small Bowel
Hård Af Segerstad EM, et al; TEDDY Study Group. Early Dietary Fiber Intake Reduces Celiac Disease Risk in Genetically Prone Children: Insights From the TEDDY Study. Gastroenterology. 2025 Feb. doi: 10.1053/j.gastro.2025.01.241.
Colon
Shaukat A, et al. AGA Clinical Practice Update on Current Role of Blood Tests for Colorectal Cancer Screening: Commentary. Clin Gastroenterol Hepatol. 2025 Apr. doi: 10.1016/j.cgh.2025.04.003.
Bergman D, et al. Cholecystectomy is a Risk Factor for Microscopic Colitis: A Nationwide Population-based Matched Case Control Study. Clin Gastroenterol Hepatol. 2025 Mar. doi: 10.1016/j.cgh.2024.12.032.
Inflammatory Bowel Disease
Ben-Horin S, et al; Israeli IBD Research Nucleus (IIRN). Capsule Endoscopy-Guided Proactive Treat-to-Target Versus Continued Standard Care in Patients With Quiescent Crohn’s Disease: A Randomized Controlled Trial. Gastroenterology. 2025 Mar. doi: 10.1053/j.gastro.2025.02.031.
Pancreas
Guilabert L, et al; ERICA Consortium. Impact of Fluid Therapy in the Emergency Department in Acute Pancreatitis: a posthoc analysis of the WATERFALL Trial. Clin Gastroenterol Hepatol. 2025 Apr. doi: 10.1016/j.cgh.2025.01.038.
Hepatology
Rhee H, et al. Noncontrast Magnetic Resonance Imaging vs Ultrasonography for Hepatocellular Carcinoma Surveillance: A Randomized, Single-Center Trial. Gastroenterology. 2025 Jan. doi: 10.1053/j.gastro.2024.12.035.
Kronsten VT, et al. Hepatic Encephalopathy: When Lactulose and Rifaximin Are Not Working. Gastroenterology. 2025 Jan. doi: 10.1053/j.gastro.2025.01.010.
Edelson JC, et al. Accuracy and Safety of Endoscopic Ultrasound–Guided Liver Biopsy in Patients with Metabolic Dysfunction–Associated Liver Disease. Tech Innov Gastrointest Endosc. 2025 Apr. doi: 10.1016/j.tige.2025.250918.
Miscellaneous
Martin J, et al. Practical and Impactful Tips for Private Industry Collaborations with Gastroenterology Practices. Clin Gastroenterol Hepatol. 2025 Mar. doi: 10.1016/j.cgh.2025.01.021.
Tejada, Natalia et al. Glucagon-like Peptide-1 Receptor Agonists Are Not Associated With Increased Incidence of Pneumonia After Endoscopic Procedures. Tech Innov Gastrointest Endosc. 2025 Apr. doi: 10.1016/j.tige.2025.250925.
Lazaridis KN, et al. Microplastics and Nanoplastics and the Digestive System. Gastro Hep Adv. 2025 May. doi: 10.1016/j.gastha.2025.100694.
Dr. Trieu is assistant professor of medicine, interventional endoscopy, in the Division of Gastroenterology at Washington University in St. Louis School of Medicine, Missouri.
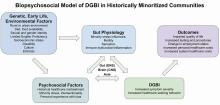
Improving Care for Patients from Historically Minoritized and Marginalized Communities with Disorders of Gut-Brain Interaction
Introduction: Cases
Patient 1: A 57-year-old man with post-prandial distress variant functional dyspepsia (FD) was recommended to start nortriptyline. He previously established primary care with a physician he met at a barbershop health fair in Harlem, who referred him for specialty evaluation. Today, he presents for follow-up and reports he did not take this medication because he heard it is an antidepressant. How would you counsel him?
Patient 2: A 61-year-old woman was previously diagnosed with mixed variant irritable bowel syndrome (IBS-M). Her symptoms have not significantly changed. Her prior workup has been reassuring and consistent with IBS-M. Despite this, the patient pushes to repeat a colonoscopy, fearful that something is being missed or that she is not being offered care because of her undocumented status. How do you respond?
Patient 3: A 36-year-old man is followed for the management of generalized anxiety disorder and functional heartburn. He was started on low-dose amitriptyline with some benefit, but follow-up has been sporadic. On further discussion, he reports financial stressors, time barriers, and difficulty scheduling a meeting with his union representative for work accommodations as he lives in a more rural community. How do you reply?
Patient 4: A 74-year-old man with Parkinson’s disease who uses a wheelchair has functional constipation that is well controlled on his current regimen. He has never undergone colon cancer screening. He occasionally notices blood in his stool, so a colonoscopy was recommended to confirm that his hematochezia reflects functional constipation complicated by hemorrhoids. He is concerned about the bowel preparation required for a colonoscopy given his limited mobility, as his insurance does not cover assistance at home. He does not have family members to help him. How can you assist him?
Social determinants of health, health disparities, and DGBIs
Social determinants of health affect all aspects of patient care, with an increasing body of published work looking at potential disparities in organ-based and structural diseases.1,2,3,4 However, little has been done to explore their influence on disorders of gut-brain interaction or DGBIs.
. As DGBIs cannot be diagnosed with a single laboratory or endoscopic test, the patient history is of the utmost importance and physician-patient rapport is paramount in their treatment. Such rapport may be more difficult to establish in patients coming from historically marginalized and minoritized communities who may be distrustful of healthcare as an institution of (discriminatory) power.
Potential DGBI management pitfalls in historically marginalized or minoritized communities
For racial and ethnic minorities in the United States, disparities in healthcare take on many forms. People from racial and ethnic minority communities are less likely to receive a gastroenterology consultation and those with IBS are more likely to undergo procedures as compared to White patients with IBS.6 Implicit bias may lead to fewer specialist referrals, and specialty care may be limited or unavailable in some areas. Patients may prefer seeing providers in their own community, with whom they share racial or ethnic identities, which could lead to fewer referrals to specialists outside of the community.
Historical discrimination contributes to a lack of trust in healthcare professionals, which may lead patients to favor more objective diagnostics such as endoscopy or view being counseled against invasive procedures as having necessary care denied. Due to a broader cultural stigma surrounding mental illness, patients may be more hesitant to utilize neuromodulators, which have historically been used for psychiatric diagnoses, as it may lead them to conflate their GI illness with mental illness.7,8
Since DGBIs cannot be diagnosed with a single test or managed with a single treatment modality, providing excellent care for patients with DGBIs requires clear communication. For patients with limited English proficiency (LEP), access to high-quality language assistance is the foundation of comprehensive care. Interpreter use (or lack thereof) may limit the ability to obtain a complete and accurate clinical history, which can lead to fewer referrals to specialists and increased reliance on endoscopic evaluations that may not be clinically indicated.
These language barriers affect patients on many levels – in their ability to understand instructions for medication administration, preparation for procedures, and return precautions – which may ultimately lead to poorer responses to therapy or delays in care. LEP alone is broadly associated with fewer referrals for outpatient follow-up, adverse health outcomes and complications, and longer hospital stays.9 These disparities can be mitigated by investing in high-quality interpreter services, providing instructions and forms in multiple languages, and engaging the patient’s family and social supports according to their preferences.
People experiencing poverty (urban and rural) face challenges across multiple domains including access to healthcare, health insurance, stable housing and employment, and more. Many patients seek care at federally qualified health centers, which may face greater difficulties coordinating care with external gastroenterologists.10
Insurance barriers limit access to essential medications, tests, and procedures, and create delays in establishing care with specialists. Significant psychological stress and higher rates of comorbid anxiety and depression contribute to increased IBS severity.11 Financial limitations may limit dietary choices, which can further exacerbate DGBI symptoms. Long work hours with limited flexibility may prohibit them from presenting for regular follow-ups and establishing advanced DGBI care such as with a dietitian or psychologist.
Patients with disabilities face many of the health inequities previously discussed, as well as additional challenges with physical accessibility, transportation, exclusion from education and employment, discrimination, and stigma. Higher prevalence of comorbid mental illness and higher rates of intimate partner violence and interpersonal violence all contribute to DGBI severity and challenges with access to care.12,13 Patients with disabilities may struggle to arrive at appointments, maneuver through the building or exam room, and ultimately follow recommended care plans.
How to approach DGBIs in historically marginalized and minoritized communities
Returning to the patients from the introduction, how would you counsel each of them?
Patient 1: We can discuss with the patient how nortriptyline and other typical antidepressants can and often are used for indications other than depression. These medications modify centrally-mediated pain signaling and many patients with functional dyspepsia experience a significant benefit. It is critical to build on the rapport that was established at the community health outreach event and to explore the patient’s concerns thoroughly.
Patient 2: We would begin by inquiring about her underlying fears associated with her symptoms and seek to understand her goals for repeat intervention. We can review the risks of endoscopy and shift the focus to improving her symptoms. If we can improve her bowel habits or her pain, her desire for further interventions may lessen.
Patient 3: It will be important to work within the realistic time and monetary constraints in this patient’s life. We can validate him and the challenges he is facing, provide positive reinforcement for the progress he has made so far, and avoid disparaging him for the aspects of the treatment plan he has been unable to follow through with. As he reported a benefit from amitriptyline, we can consider increasing his dose as a feasible next step.
Patient 4: We can encourage the patient to discuss with his primary care physician how they may be able to coordinate an inpatient admission for colonoscopy preparation. Given his co-morbidities, this avenue will provide him dedicated support to help him adequately prep to ensure a higher quality examination and limit the need for repeat procedures.
DGBI care in historically marginalized and minoritized communities: A call to action
Understanding cultural differences and existing disparities in care is essential to improving care for patients from historically minoritized communities with DGBIs. Motivational interviewing and shared decision-making, with acknowledgment of social and cultural differences, allow us to work together with patients and their support systems to set and achieve feasible goals.14
To address known health disparities, offices can take steps to ensure the accessibility of language, forms, physical space, providers, and care teams. Providing culturally sensitive care and lowering barriers to care are the first steps to effecting meaningful change for patients with DGBIs from historically minoritized communities.
Dr. Yu is based at Division of Gastroenterology and Hepatology, Boston Medical Center and Boston University, both in Boston, Massachusetts. Dr. Dimino and Dr. Vélez are based at the Division of Gastroenterology, Massachusetts General Hospital and Harvard Medical School, both in Boston, Massachusetts. Dr. Yu, Dr. Dimino, and Dr. Vélez do not have any conflicts of interest for this article.
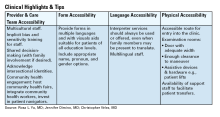
Additional Online Resources
Form Accessibility
- Intake Form Guidance for Providers
- Making Your Clinic Welcoming to LGBTQ Patients
- Transgender data collection in the electronic health record: Current concepts and issues
Language Accessibility
Physical Accessibility
- Access to Medical Care for Individuals with Mobility Disabilities
- Making your medical office accessible
References
1. Zavala VA, et al. Cancer health disparities in racial/ethnic minorities in the United States. Br J Cancer. 2021 Jan. doi: 10.1038/s41416-020-01038-6.
2. Kardashian A, et al. Health disparities in chronic liver disease. Hepatology. 2023 Apr. doi: 10.1002/hep.32743.
3. Nephew LD, Serper M. Racial, Gender, and Socioeconomic Disparities in Liver Transplantation. Liver Transpl. 2021 Jun. doi: 10.1002/lt.25996.
4. Anyane-Yeboa A, et al. The Impact of the Social Determinants of Health on Disparities in Inflammatory Bowel Disease. Clin Gastroenterol Hepatol. 2022 Nov. doi: 10.1016/j.cgh.2022.03.011.
5. Drossman DA. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features and Rome IV. Gastroenterology. 2016 Feb. doi: 10.1053/j.gastro.2016.02.032.
6. Silvernale C, et al. Racial disparity in healthcare utilization among patients with Irritable Bowel Syndrome: results from a multicenter cohort. Neurogastroenterol Motil. 2021 May. doi: 10.1111/nmo.14039.
7. Hearn M, et al. Stigma and irritable bowel syndrome: a taboo subject? Lancet Gastroenterol Hepatol. 2020 Jun. doi: 10.1016/S2468-1253(19)30348-6.
8. Yan XJ, et al. The impact of stigma on medication adherence in patients with functional dyspepsia. Neurogastroenterol Motil. 2021 Feb. doi: 10.1111/nmo.13956.
9. Twersky SE, et al. The Impact of Limited English Proficiency on Healthcare Access and Outcomes in the U.S.: A Scoping Review. Healthcare (Basel). 2024 Jan. doi: 10.3390/healthcare12030364.
10. Bayly JE, et al. Limited English proficiency and reported receipt of colorectal cancer screening among adults 45-75 in 2019 and 2021. Prev Med Rep. 2024 Feb. doi: 10.1016/j.pmedr.2024.102638.
11. Cheng K, et al. Epidemiology of Irritable Bowel Syndrome in a Large Academic Safety-Net Hospital. J Clin Med. 2024 Feb. doi: 10.3390/jcm13051314.
12. Breiding MJ, Armour BS. The association between disability and intimate partner violence in the United States. Ann Epidemiol. 2015 Jun. doi: 10.1016/j.annepidem.2015.03.017.
13. Mitra M, et al. Prevalence and characteristics of sexual violence against men with disabilities. Am J Prev Med. 2016 Mar. doi: 10.1016/j.amepre.2015.07.030.
14. Bahafzallah L, et al. Motivational Interviewing in Ethnic Populations. J Immigr Minor Health. 2020 Aug. doi: 10.1007/s10903-019-00940-3.
Introduction: Cases
Patient 1: A 57-year-old man with post-prandial distress variant functional dyspepsia (FD) was recommended to start nortriptyline. He previously established primary care with a physician he met at a barbershop health fair in Harlem, who referred him for specialty evaluation. Today, he presents for follow-up and reports he did not take this medication because he heard it is an antidepressant. How would you counsel him?
Patient 2: A 61-year-old woman was previously diagnosed with mixed variant irritable bowel syndrome (IBS-M). Her symptoms have not significantly changed. Her prior workup has been reassuring and consistent with IBS-M. Despite this, the patient pushes to repeat a colonoscopy, fearful that something is being missed or that she is not being offered care because of her undocumented status. How do you respond?
Patient 3: A 36-year-old man is followed for the management of generalized anxiety disorder and functional heartburn. He was started on low-dose amitriptyline with some benefit, but follow-up has been sporadic. On further discussion, he reports financial stressors, time barriers, and difficulty scheduling a meeting with his union representative for work accommodations as he lives in a more rural community. How do you reply?
Patient 4: A 74-year-old man with Parkinson’s disease who uses a wheelchair has functional constipation that is well controlled on his current regimen. He has never undergone colon cancer screening. He occasionally notices blood in his stool, so a colonoscopy was recommended to confirm that his hematochezia reflects functional constipation complicated by hemorrhoids. He is concerned about the bowel preparation required for a colonoscopy given his limited mobility, as his insurance does not cover assistance at home. He does not have family members to help him. How can you assist him?
Social determinants of health, health disparities, and DGBIs
Social determinants of health affect all aspects of patient care, with an increasing body of published work looking at potential disparities in organ-based and structural diseases.1,2,3,4 However, little has been done to explore their influence on disorders of gut-brain interaction or DGBIs.
. As DGBIs cannot be diagnosed with a single laboratory or endoscopic test, the patient history is of the utmost importance and physician-patient rapport is paramount in their treatment. Such rapport may be more difficult to establish in patients coming from historically marginalized and minoritized communities who may be distrustful of healthcare as an institution of (discriminatory) power.
Potential DGBI management pitfalls in historically marginalized or minoritized communities
For racial and ethnic minorities in the United States, disparities in healthcare take on many forms. People from racial and ethnic minority communities are less likely to receive a gastroenterology consultation and those with IBS are more likely to undergo procedures as compared to White patients with IBS.6 Implicit bias may lead to fewer specialist referrals, and specialty care may be limited or unavailable in some areas. Patients may prefer seeing providers in their own community, with whom they share racial or ethnic identities, which could lead to fewer referrals to specialists outside of the community.
Historical discrimination contributes to a lack of trust in healthcare professionals, which may lead patients to favor more objective diagnostics such as endoscopy or view being counseled against invasive procedures as having necessary care denied. Due to a broader cultural stigma surrounding mental illness, patients may be more hesitant to utilize neuromodulators, which have historically been used for psychiatric diagnoses, as it may lead them to conflate their GI illness with mental illness.7,8
Since DGBIs cannot be diagnosed with a single test or managed with a single treatment modality, providing excellent care for patients with DGBIs requires clear communication. For patients with limited English proficiency (LEP), access to high-quality language assistance is the foundation of comprehensive care. Interpreter use (or lack thereof) may limit the ability to obtain a complete and accurate clinical history, which can lead to fewer referrals to specialists and increased reliance on endoscopic evaluations that may not be clinically indicated.
These language barriers affect patients on many levels – in their ability to understand instructions for medication administration, preparation for procedures, and return precautions – which may ultimately lead to poorer responses to therapy or delays in care. LEP alone is broadly associated with fewer referrals for outpatient follow-up, adverse health outcomes and complications, and longer hospital stays.9 These disparities can be mitigated by investing in high-quality interpreter services, providing instructions and forms in multiple languages, and engaging the patient’s family and social supports according to their preferences.
People experiencing poverty (urban and rural) face challenges across multiple domains including access to healthcare, health insurance, stable housing and employment, and more. Many patients seek care at federally qualified health centers, which may face greater difficulties coordinating care with external gastroenterologists.10
Insurance barriers limit access to essential medications, tests, and procedures, and create delays in establishing care with specialists. Significant psychological stress and higher rates of comorbid anxiety and depression contribute to increased IBS severity.11 Financial limitations may limit dietary choices, which can further exacerbate DGBI symptoms. Long work hours with limited flexibility may prohibit them from presenting for regular follow-ups and establishing advanced DGBI care such as with a dietitian or psychologist.
Patients with disabilities face many of the health inequities previously discussed, as well as additional challenges with physical accessibility, transportation, exclusion from education and employment, discrimination, and stigma. Higher prevalence of comorbid mental illness and higher rates of intimate partner violence and interpersonal violence all contribute to DGBI severity and challenges with access to care.12,13 Patients with disabilities may struggle to arrive at appointments, maneuver through the building or exam room, and ultimately follow recommended care plans.
How to approach DGBIs in historically marginalized and minoritized communities
Returning to the patients from the introduction, how would you counsel each of them?
Patient 1: We can discuss with the patient how nortriptyline and other typical antidepressants can and often are used for indications other than depression. These medications modify centrally-mediated pain signaling and many patients with functional dyspepsia experience a significant benefit. It is critical to build on the rapport that was established at the community health outreach event and to explore the patient’s concerns thoroughly.
Patient 2: We would begin by inquiring about her underlying fears associated with her symptoms and seek to understand her goals for repeat intervention. We can review the risks of endoscopy and shift the focus to improving her symptoms. If we can improve her bowel habits or her pain, her desire for further interventions may lessen.
Patient 3: It will be important to work within the realistic time and monetary constraints in this patient’s life. We can validate him and the challenges he is facing, provide positive reinforcement for the progress he has made so far, and avoid disparaging him for the aspects of the treatment plan he has been unable to follow through with. As he reported a benefit from amitriptyline, we can consider increasing his dose as a feasible next step.
Patient 4: We can encourage the patient to discuss with his primary care physician how they may be able to coordinate an inpatient admission for colonoscopy preparation. Given his co-morbidities, this avenue will provide him dedicated support to help him adequately prep to ensure a higher quality examination and limit the need for repeat procedures.
DGBI care in historically marginalized and minoritized communities: A call to action
Understanding cultural differences and existing disparities in care is essential to improving care for patients from historically minoritized communities with DGBIs. Motivational interviewing and shared decision-making, with acknowledgment of social and cultural differences, allow us to work together with patients and their support systems to set and achieve feasible goals.14
To address known health disparities, offices can take steps to ensure the accessibility of language, forms, physical space, providers, and care teams. Providing culturally sensitive care and lowering barriers to care are the first steps to effecting meaningful change for patients with DGBIs from historically minoritized communities.
Dr. Yu is based at Division of Gastroenterology and Hepatology, Boston Medical Center and Boston University, both in Boston, Massachusetts. Dr. Dimino and Dr. Vélez are based at the Division of Gastroenterology, Massachusetts General Hospital and Harvard Medical School, both in Boston, Massachusetts. Dr. Yu, Dr. Dimino, and Dr. Vélez do not have any conflicts of interest for this article.
Additional Online Resources
Form Accessibility
- Intake Form Guidance for Providers
- Making Your Clinic Welcoming to LGBTQ Patients
- Transgender data collection in the electronic health record: Current concepts and issues
Language Accessibility
Physical Accessibility
- Access to Medical Care for Individuals with Mobility Disabilities
- Making your medical office accessible
References
1. Zavala VA, et al. Cancer health disparities in racial/ethnic minorities in the United States. Br J Cancer. 2021 Jan. doi: 10.1038/s41416-020-01038-6.
2. Kardashian A, et al. Health disparities in chronic liver disease. Hepatology. 2023 Apr. doi: 10.1002/hep.32743.
3. Nephew LD, Serper M. Racial, Gender, and Socioeconomic Disparities in Liver Transplantation. Liver Transpl. 2021 Jun. doi: 10.1002/lt.25996.
4. Anyane-Yeboa A, et al. The Impact of the Social Determinants of Health on Disparities in Inflammatory Bowel Disease. Clin Gastroenterol Hepatol. 2022 Nov. doi: 10.1016/j.cgh.2022.03.011.
5. Drossman DA. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features and Rome IV. Gastroenterology. 2016 Feb. doi: 10.1053/j.gastro.2016.02.032.
6. Silvernale C, et al. Racial disparity in healthcare utilization among patients with Irritable Bowel Syndrome: results from a multicenter cohort. Neurogastroenterol Motil. 2021 May. doi: 10.1111/nmo.14039.
7. Hearn M, et al. Stigma and irritable bowel syndrome: a taboo subject? Lancet Gastroenterol Hepatol. 2020 Jun. doi: 10.1016/S2468-1253(19)30348-6.
8. Yan XJ, et al. The impact of stigma on medication adherence in patients with functional dyspepsia. Neurogastroenterol Motil. 2021 Feb. doi: 10.1111/nmo.13956.
9. Twersky SE, et al. The Impact of Limited English Proficiency on Healthcare Access and Outcomes in the U.S.: A Scoping Review. Healthcare (Basel). 2024 Jan. doi: 10.3390/healthcare12030364.
10. Bayly JE, et al. Limited English proficiency and reported receipt of colorectal cancer screening among adults 45-75 in 2019 and 2021. Prev Med Rep. 2024 Feb. doi: 10.1016/j.pmedr.2024.102638.
11. Cheng K, et al. Epidemiology of Irritable Bowel Syndrome in a Large Academic Safety-Net Hospital. J Clin Med. 2024 Feb. doi: 10.3390/jcm13051314.
12. Breiding MJ, Armour BS. The association between disability and intimate partner violence in the United States. Ann Epidemiol. 2015 Jun. doi: 10.1016/j.annepidem.2015.03.017.
13. Mitra M, et al. Prevalence and characteristics of sexual violence against men with disabilities. Am J Prev Med. 2016 Mar. doi: 10.1016/j.amepre.2015.07.030.
14. Bahafzallah L, et al. Motivational Interviewing in Ethnic Populations. J Immigr Minor Health. 2020 Aug. doi: 10.1007/s10903-019-00940-3.
Introduction: Cases
Patient 1: A 57-year-old man with post-prandial distress variant functional dyspepsia (FD) was recommended to start nortriptyline. He previously established primary care with a physician he met at a barbershop health fair in Harlem, who referred him for specialty evaluation. Today, he presents for follow-up and reports he did not take this medication because he heard it is an antidepressant. How would you counsel him?
Patient 2: A 61-year-old woman was previously diagnosed with mixed variant irritable bowel syndrome (IBS-M). Her symptoms have not significantly changed. Her prior workup has been reassuring and consistent with IBS-M. Despite this, the patient pushes to repeat a colonoscopy, fearful that something is being missed or that she is not being offered care because of her undocumented status. How do you respond?
Patient 3: A 36-year-old man is followed for the management of generalized anxiety disorder and functional heartburn. He was started on low-dose amitriptyline with some benefit, but follow-up has been sporadic. On further discussion, he reports financial stressors, time barriers, and difficulty scheduling a meeting with his union representative for work accommodations as he lives in a more rural community. How do you reply?
Patient 4: A 74-year-old man with Parkinson’s disease who uses a wheelchair has functional constipation that is well controlled on his current regimen. He has never undergone colon cancer screening. He occasionally notices blood in his stool, so a colonoscopy was recommended to confirm that his hematochezia reflects functional constipation complicated by hemorrhoids. He is concerned about the bowel preparation required for a colonoscopy given his limited mobility, as his insurance does not cover assistance at home. He does not have family members to help him. How can you assist him?
Social determinants of health, health disparities, and DGBIs
Social determinants of health affect all aspects of patient care, with an increasing body of published work looking at potential disparities in organ-based and structural diseases.1,2,3,4 However, little has been done to explore their influence on disorders of gut-brain interaction or DGBIs.
. As DGBIs cannot be diagnosed with a single laboratory or endoscopic test, the patient history is of the utmost importance and physician-patient rapport is paramount in their treatment. Such rapport may be more difficult to establish in patients coming from historically marginalized and minoritized communities who may be distrustful of healthcare as an institution of (discriminatory) power.
Potential DGBI management pitfalls in historically marginalized or minoritized communities
For racial and ethnic minorities in the United States, disparities in healthcare take on many forms. People from racial and ethnic minority communities are less likely to receive a gastroenterology consultation and those with IBS are more likely to undergo procedures as compared to White patients with IBS.6 Implicit bias may lead to fewer specialist referrals, and specialty care may be limited or unavailable in some areas. Patients may prefer seeing providers in their own community, with whom they share racial or ethnic identities, which could lead to fewer referrals to specialists outside of the community.
Historical discrimination contributes to a lack of trust in healthcare professionals, which may lead patients to favor more objective diagnostics such as endoscopy or view being counseled against invasive procedures as having necessary care denied. Due to a broader cultural stigma surrounding mental illness, patients may be more hesitant to utilize neuromodulators, which have historically been used for psychiatric diagnoses, as it may lead them to conflate their GI illness with mental illness.7,8
Since DGBIs cannot be diagnosed with a single test or managed with a single treatment modality, providing excellent care for patients with DGBIs requires clear communication. For patients with limited English proficiency (LEP), access to high-quality language assistance is the foundation of comprehensive care. Interpreter use (or lack thereof) may limit the ability to obtain a complete and accurate clinical history, which can lead to fewer referrals to specialists and increased reliance on endoscopic evaluations that may not be clinically indicated.
These language barriers affect patients on many levels – in their ability to understand instructions for medication administration, preparation for procedures, and return precautions – which may ultimately lead to poorer responses to therapy or delays in care. LEP alone is broadly associated with fewer referrals for outpatient follow-up, adverse health outcomes and complications, and longer hospital stays.9 These disparities can be mitigated by investing in high-quality interpreter services, providing instructions and forms in multiple languages, and engaging the patient’s family and social supports according to their preferences.
People experiencing poverty (urban and rural) face challenges across multiple domains including access to healthcare, health insurance, stable housing and employment, and more. Many patients seek care at federally qualified health centers, which may face greater difficulties coordinating care with external gastroenterologists.10
Insurance barriers limit access to essential medications, tests, and procedures, and create delays in establishing care with specialists. Significant psychological stress and higher rates of comorbid anxiety and depression contribute to increased IBS severity.11 Financial limitations may limit dietary choices, which can further exacerbate DGBI symptoms. Long work hours with limited flexibility may prohibit them from presenting for regular follow-ups and establishing advanced DGBI care such as with a dietitian or psychologist.
Patients with disabilities face many of the health inequities previously discussed, as well as additional challenges with physical accessibility, transportation, exclusion from education and employment, discrimination, and stigma. Higher prevalence of comorbid mental illness and higher rates of intimate partner violence and interpersonal violence all contribute to DGBI severity and challenges with access to care.12,13 Patients with disabilities may struggle to arrive at appointments, maneuver through the building or exam room, and ultimately follow recommended care plans.
How to approach DGBIs in historically marginalized and minoritized communities
Returning to the patients from the introduction, how would you counsel each of them?
Patient 1: We can discuss with the patient how nortriptyline and other typical antidepressants can and often are used for indications other than depression. These medications modify centrally-mediated pain signaling and many patients with functional dyspepsia experience a significant benefit. It is critical to build on the rapport that was established at the community health outreach event and to explore the patient’s concerns thoroughly.
Patient 2: We would begin by inquiring about her underlying fears associated with her symptoms and seek to understand her goals for repeat intervention. We can review the risks of endoscopy and shift the focus to improving her symptoms. If we can improve her bowel habits or her pain, her desire for further interventions may lessen.
Patient 3: It will be important to work within the realistic time and monetary constraints in this patient’s life. We can validate him and the challenges he is facing, provide positive reinforcement for the progress he has made so far, and avoid disparaging him for the aspects of the treatment plan he has been unable to follow through with. As he reported a benefit from amitriptyline, we can consider increasing his dose as a feasible next step.
Patient 4: We can encourage the patient to discuss with his primary care physician how they may be able to coordinate an inpatient admission for colonoscopy preparation. Given his co-morbidities, this avenue will provide him dedicated support to help him adequately prep to ensure a higher quality examination and limit the need for repeat procedures.
DGBI care in historically marginalized and minoritized communities: A call to action
Understanding cultural differences and existing disparities in care is essential to improving care for patients from historically minoritized communities with DGBIs. Motivational interviewing and shared decision-making, with acknowledgment of social and cultural differences, allow us to work together with patients and their support systems to set and achieve feasible goals.14
To address known health disparities, offices can take steps to ensure the accessibility of language, forms, physical space, providers, and care teams. Providing culturally sensitive care and lowering barriers to care are the first steps to effecting meaningful change for patients with DGBIs from historically minoritized communities.
Dr. Yu is based at Division of Gastroenterology and Hepatology, Boston Medical Center and Boston University, both in Boston, Massachusetts. Dr. Dimino and Dr. Vélez are based at the Division of Gastroenterology, Massachusetts General Hospital and Harvard Medical School, both in Boston, Massachusetts. Dr. Yu, Dr. Dimino, and Dr. Vélez do not have any conflicts of interest for this article.
Additional Online Resources
Form Accessibility
- Intake Form Guidance for Providers
- Making Your Clinic Welcoming to LGBTQ Patients
- Transgender data collection in the electronic health record: Current concepts and issues
Language Accessibility
Physical Accessibility
- Access to Medical Care for Individuals with Mobility Disabilities
- Making your medical office accessible
References
1. Zavala VA, et al. Cancer health disparities in racial/ethnic minorities in the United States. Br J Cancer. 2021 Jan. doi: 10.1038/s41416-020-01038-6.
2. Kardashian A, et al. Health disparities in chronic liver disease. Hepatology. 2023 Apr. doi: 10.1002/hep.32743.
3. Nephew LD, Serper M. Racial, Gender, and Socioeconomic Disparities in Liver Transplantation. Liver Transpl. 2021 Jun. doi: 10.1002/lt.25996.
4. Anyane-Yeboa A, et al. The Impact of the Social Determinants of Health on Disparities in Inflammatory Bowel Disease. Clin Gastroenterol Hepatol. 2022 Nov. doi: 10.1016/j.cgh.2022.03.011.
5. Drossman DA. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features and Rome IV. Gastroenterology. 2016 Feb. doi: 10.1053/j.gastro.2016.02.032.
6. Silvernale C, et al. Racial disparity in healthcare utilization among patients with Irritable Bowel Syndrome: results from a multicenter cohort. Neurogastroenterol Motil. 2021 May. doi: 10.1111/nmo.14039.
7. Hearn M, et al. Stigma and irritable bowel syndrome: a taboo subject? Lancet Gastroenterol Hepatol. 2020 Jun. doi: 10.1016/S2468-1253(19)30348-6.
8. Yan XJ, et al. The impact of stigma on medication adherence in patients with functional dyspepsia. Neurogastroenterol Motil. 2021 Feb. doi: 10.1111/nmo.13956.
9. Twersky SE, et al. The Impact of Limited English Proficiency on Healthcare Access and Outcomes in the U.S.: A Scoping Review. Healthcare (Basel). 2024 Jan. doi: 10.3390/healthcare12030364.
10. Bayly JE, et al. Limited English proficiency and reported receipt of colorectal cancer screening among adults 45-75 in 2019 and 2021. Prev Med Rep. 2024 Feb. doi: 10.1016/j.pmedr.2024.102638.
11. Cheng K, et al. Epidemiology of Irritable Bowel Syndrome in a Large Academic Safety-Net Hospital. J Clin Med. 2024 Feb. doi: 10.3390/jcm13051314.
12. Breiding MJ, Armour BS. The association between disability and intimate partner violence in the United States. Ann Epidemiol. 2015 Jun. doi: 10.1016/j.annepidem.2015.03.017.
13. Mitra M, et al. Prevalence and characteristics of sexual violence against men with disabilities. Am J Prev Med. 2016 Mar. doi: 10.1016/j.amepre.2015.07.030.
14. Bahafzallah L, et al. Motivational Interviewing in Ethnic Populations. J Immigr Minor Health. 2020 Aug. doi: 10.1007/s10903-019-00940-3.
Less Invasive Screening May Identify Barrett’s Esophagus Earlier
, a small comparative study in US veterans found.
BE is up to three times more prevalent in veterans than in the general population.
This and other minimally invasive approaches may reduce patient anxiety and increase screening rates, according to investigators led by Katarina B. Greer, MD, MS, of the VA Northeast Ohio Healthcare System and Case Western University in Cleveland. Such screening platforms are expected to open a window on improved prognosis for EAC by offering well-tolerated, office-based testing, the authors wrote in The American Journal of Gastroenterology.
Greer and colleagues compared standard upper endoscopy with EsoCheck (EC), a nonendoscopic esophageal balloon cell-sampling device coupled with EsoGuard (EG), a DNA-based precancer screening assay, with standard upper endoscopy, an FDA-approved minimally invasive alternative.
Sensitivity and specificity of combined EC/EG for esophagogastroduodenoscopy (EGD)-detected BE/EAC were 92.9% (95% CI, 66.1-99.8) and 72.2% (95% CI, 62.1-80.8), respectively. Positive and negative predictive values were 32.5% (95% CI, 18.6-49.1) and 98.6% (95% CI, 92.4-100), respectively.
“With its strong negative predictive power, this screening modality could be a first-line tool available to a greater number of patients,” Greer and associates wrote. “Data from this test support the notion that EC could be performed as a triaging test to increase the yield of diagnostic upper endoscopy 2.5-fold.”
The US rates of EAC have increased more than six-fold in the past four decades and continue to rise. In 2023, 21,560 cases of EAC were diagnosed here. The prognosis for EAC is still poor, with fewer than 22% of patients surviving beyond 5 years.
Current guidelines recommend sedated EGD for patients with chronic gastroesophageal reflux disease (GERD) and additional BE risk factors such as smoking, obesity, and family history. This strategy, however, often fails to detect BE when symptoms are well controlled with over-the-counter or physician-prescribed therapies, Greer and colleagues noted. It also fails to detect BE in individuals without GERD, who comprise 40% of those who develop EAC.
Fewer than 5% of EACs are diagnosed as early-stage lesions caught by surveillance of patients with previously detected BE.
Study Details
The researchers recruited veterans meeting American College of Gastroenterology criteria for endoscopic BE and EAC screening at the Louis Stokes Cleveland Veterans Affairs Medical Center.
Of 782 eligible veterans, 130 (16.6%) entered the study and 124 completed screening. Common reasons for nonparticipation included completion of upper endoscopy outside of the VA healthcare system, lack of interest in joining a research study, and no recommendation for screening from referring gastroenterology or primary care providers. Eligible candidates had gastroesophageal reflux disorder plus three additional risk factors, such as smoking, higher BMI, male sex, age 50 years or older, and family history. The mean number of risk factors was 4.1.
“Available data suggest that family history is the strongest predictor of BE diagnosis, as prevalence of BE among those with family history was 23%,” Greer’s group wrote. “This points to high priority of pursuing screening in patients with family history of the condition, followed by patients who share multiple risk factors.”
All participants completed unsedated EC-guided distal esophageal sampling followed by a sedated EGD on the same day. The prevalence of BE/EAC was 12.9% (n = 14/2), based on standard EGD.
“The study was not powered to prospectively determine EC diagnostic accuracy for subgroups of nondysplastic and dysplastic BE and EAC. These data are reported for this device in development studies but not available for our study population,” the authors wrote. In comparison, they noted, the Cytosponge-TFF3, another nonendoscopic screening device for EAC and BE, exhibited lower sensitivity of 79.5%-87.2%, depending on lesion length, but higher specificity of 92.4%.
Procedural Anxiety
Baseline scores on the short-form six-item Spielberger State-Trait Anxiety Inventory-6 (STAI-6) revealed notable levels of periprocedural anxiety. STAI-6 scores range from 20 to 80, with higher scores indicating more severe anxiety. In the VA study, scores ranged from 20 to 60, and most domains constituting the scores were the same before and after the procedure. Participants did, however, report a statistically significant decrease in sense of worry after EC and reported good tolerability for both EC and EG.
Offering an outsider’s perspective on the study, Joshua Sloan, DO, an esophageal gastroenterologist at University of Minnesota Medical Center in Minneapolis, said that with the acceleration of US rates of EAC, developing a nonendoscopic screening tool to improve identification of Barrett’s and perhaps early EAC is important. “The study by Greer et al helps support the use of nonendoscopic screening with EsoCheck and EsoGuard to identify these conditions,” he told GI & Hepatology News. “It will be interesting to see similar studies in the non-VA population as well. As the study notes, veterans are an enriched population with a higher prevalence of Barrett’s esophagus.”
Ultimately, Sloan added, “the hope is to increase our ability to identify and manage BE before it becomes EAC. Nonendoscopic screening tools have the potential to increase diagnosis and funnel the appropriate patients for endoscopic surveillance.”
The Bottom Line
“Calculations regarding effectiveness of the two-step screening strategy afforded by EC indicate that the burden of screening would be reduced by at least half (53%),” the authors wrote. Since the estimated size of the US screen-eligible population ranges from 19.7 million to 120.1 million, noninvasive tools could significantly decrease EGD procedures. A formal cost effectiveness analysis is being conducted and will be published separately.
This study was funded by a Department of Defense award.
Co-Author Chak reported device patents assigned to Case Western Reserve University and licensed to Lucid Diagnostics. The other authors had no competing interests to declare. Sloan disclosed speaking and/or advisory work for Sanofi-Regeneron, Phathom Pharmaceuticals, and Takeda Pharmaceuticals unrelated to his comments.
A version of this article appeared on Medscape.com.
, a small comparative study in US veterans found.
BE is up to three times more prevalent in veterans than in the general population.
This and other minimally invasive approaches may reduce patient anxiety and increase screening rates, according to investigators led by Katarina B. Greer, MD, MS, of the VA Northeast Ohio Healthcare System and Case Western University in Cleveland. Such screening platforms are expected to open a window on improved prognosis for EAC by offering well-tolerated, office-based testing, the authors wrote in The American Journal of Gastroenterology.
Greer and colleagues compared standard upper endoscopy with EsoCheck (EC), a nonendoscopic esophageal balloon cell-sampling device coupled with EsoGuard (EG), a DNA-based precancer screening assay, with standard upper endoscopy, an FDA-approved minimally invasive alternative.
Sensitivity and specificity of combined EC/EG for esophagogastroduodenoscopy (EGD)-detected BE/EAC were 92.9% (95% CI, 66.1-99.8) and 72.2% (95% CI, 62.1-80.8), respectively. Positive and negative predictive values were 32.5% (95% CI, 18.6-49.1) and 98.6% (95% CI, 92.4-100), respectively.
“With its strong negative predictive power, this screening modality could be a first-line tool available to a greater number of patients,” Greer and associates wrote. “Data from this test support the notion that EC could be performed as a triaging test to increase the yield of diagnostic upper endoscopy 2.5-fold.”
The US rates of EAC have increased more than six-fold in the past four decades and continue to rise. In 2023, 21,560 cases of EAC were diagnosed here. The prognosis for EAC is still poor, with fewer than 22% of patients surviving beyond 5 years.
Current guidelines recommend sedated EGD for patients with chronic gastroesophageal reflux disease (GERD) and additional BE risk factors such as smoking, obesity, and family history. This strategy, however, often fails to detect BE when symptoms are well controlled with over-the-counter or physician-prescribed therapies, Greer and colleagues noted. It also fails to detect BE in individuals without GERD, who comprise 40% of those who develop EAC.
Fewer than 5% of EACs are diagnosed as early-stage lesions caught by surveillance of patients with previously detected BE.
Study Details
The researchers recruited veterans meeting American College of Gastroenterology criteria for endoscopic BE and EAC screening at the Louis Stokes Cleveland Veterans Affairs Medical Center.
Of 782 eligible veterans, 130 (16.6%) entered the study and 124 completed screening. Common reasons for nonparticipation included completion of upper endoscopy outside of the VA healthcare system, lack of interest in joining a research study, and no recommendation for screening from referring gastroenterology or primary care providers. Eligible candidates had gastroesophageal reflux disorder plus three additional risk factors, such as smoking, higher BMI, male sex, age 50 years or older, and family history. The mean number of risk factors was 4.1.
“Available data suggest that family history is the strongest predictor of BE diagnosis, as prevalence of BE among those with family history was 23%,” Greer’s group wrote. “This points to high priority of pursuing screening in patients with family history of the condition, followed by patients who share multiple risk factors.”
All participants completed unsedated EC-guided distal esophageal sampling followed by a sedated EGD on the same day. The prevalence of BE/EAC was 12.9% (n = 14/2), based on standard EGD.
“The study was not powered to prospectively determine EC diagnostic accuracy for subgroups of nondysplastic and dysplastic BE and EAC. These data are reported for this device in development studies but not available for our study population,” the authors wrote. In comparison, they noted, the Cytosponge-TFF3, another nonendoscopic screening device for EAC and BE, exhibited lower sensitivity of 79.5%-87.2%, depending on lesion length, but higher specificity of 92.4%.
Procedural Anxiety
Baseline scores on the short-form six-item Spielberger State-Trait Anxiety Inventory-6 (STAI-6) revealed notable levels of periprocedural anxiety. STAI-6 scores range from 20 to 80, with higher scores indicating more severe anxiety. In the VA study, scores ranged from 20 to 60, and most domains constituting the scores were the same before and after the procedure. Participants did, however, report a statistically significant decrease in sense of worry after EC and reported good tolerability for both EC and EG.
Offering an outsider’s perspective on the study, Joshua Sloan, DO, an esophageal gastroenterologist at University of Minnesota Medical Center in Minneapolis, said that with the acceleration of US rates of EAC, developing a nonendoscopic screening tool to improve identification of Barrett’s and perhaps early EAC is important. “The study by Greer et al helps support the use of nonendoscopic screening with EsoCheck and EsoGuard to identify these conditions,” he told GI & Hepatology News. “It will be interesting to see similar studies in the non-VA population as well. As the study notes, veterans are an enriched population with a higher prevalence of Barrett’s esophagus.”
Ultimately, Sloan added, “the hope is to increase our ability to identify and manage BE before it becomes EAC. Nonendoscopic screening tools have the potential to increase diagnosis and funnel the appropriate patients for endoscopic surveillance.”
The Bottom Line
“Calculations regarding effectiveness of the two-step screening strategy afforded by EC indicate that the burden of screening would be reduced by at least half (53%),” the authors wrote. Since the estimated size of the US screen-eligible population ranges from 19.7 million to 120.1 million, noninvasive tools could significantly decrease EGD procedures. A formal cost effectiveness analysis is being conducted and will be published separately.
This study was funded by a Department of Defense award.
Co-Author Chak reported device patents assigned to Case Western Reserve University and licensed to Lucid Diagnostics. The other authors had no competing interests to declare. Sloan disclosed speaking and/or advisory work for Sanofi-Regeneron, Phathom Pharmaceuticals, and Takeda Pharmaceuticals unrelated to his comments.
A version of this article appeared on Medscape.com.
, a small comparative study in US veterans found.
BE is up to three times more prevalent in veterans than in the general population.
This and other minimally invasive approaches may reduce patient anxiety and increase screening rates, according to investigators led by Katarina B. Greer, MD, MS, of the VA Northeast Ohio Healthcare System and Case Western University in Cleveland. Such screening platforms are expected to open a window on improved prognosis for EAC by offering well-tolerated, office-based testing, the authors wrote in The American Journal of Gastroenterology.
Greer and colleagues compared standard upper endoscopy with EsoCheck (EC), a nonendoscopic esophageal balloon cell-sampling device coupled with EsoGuard (EG), a DNA-based precancer screening assay, with standard upper endoscopy, an FDA-approved minimally invasive alternative.
Sensitivity and specificity of combined EC/EG for esophagogastroduodenoscopy (EGD)-detected BE/EAC were 92.9% (95% CI, 66.1-99.8) and 72.2% (95% CI, 62.1-80.8), respectively. Positive and negative predictive values were 32.5% (95% CI, 18.6-49.1) and 98.6% (95% CI, 92.4-100), respectively.
“With its strong negative predictive power, this screening modality could be a first-line tool available to a greater number of patients,” Greer and associates wrote. “Data from this test support the notion that EC could be performed as a triaging test to increase the yield of diagnostic upper endoscopy 2.5-fold.”
The US rates of EAC have increased more than six-fold in the past four decades and continue to rise. In 2023, 21,560 cases of EAC were diagnosed here. The prognosis for EAC is still poor, with fewer than 22% of patients surviving beyond 5 years.
Current guidelines recommend sedated EGD for patients with chronic gastroesophageal reflux disease (GERD) and additional BE risk factors such as smoking, obesity, and family history. This strategy, however, often fails to detect BE when symptoms are well controlled with over-the-counter or physician-prescribed therapies, Greer and colleagues noted. It also fails to detect BE in individuals without GERD, who comprise 40% of those who develop EAC.
Fewer than 5% of EACs are diagnosed as early-stage lesions caught by surveillance of patients with previously detected BE.
Study Details
The researchers recruited veterans meeting American College of Gastroenterology criteria for endoscopic BE and EAC screening at the Louis Stokes Cleveland Veterans Affairs Medical Center.
Of 782 eligible veterans, 130 (16.6%) entered the study and 124 completed screening. Common reasons for nonparticipation included completion of upper endoscopy outside of the VA healthcare system, lack of interest in joining a research study, and no recommendation for screening from referring gastroenterology or primary care providers. Eligible candidates had gastroesophageal reflux disorder plus three additional risk factors, such as smoking, higher BMI, male sex, age 50 years or older, and family history. The mean number of risk factors was 4.1.
“Available data suggest that family history is the strongest predictor of BE diagnosis, as prevalence of BE among those with family history was 23%,” Greer’s group wrote. “This points to high priority of pursuing screening in patients with family history of the condition, followed by patients who share multiple risk factors.”
All participants completed unsedated EC-guided distal esophageal sampling followed by a sedated EGD on the same day. The prevalence of BE/EAC was 12.9% (n = 14/2), based on standard EGD.
“The study was not powered to prospectively determine EC diagnostic accuracy for subgroups of nondysplastic and dysplastic BE and EAC. These data are reported for this device in development studies but not available for our study population,” the authors wrote. In comparison, they noted, the Cytosponge-TFF3, another nonendoscopic screening device for EAC and BE, exhibited lower sensitivity of 79.5%-87.2%, depending on lesion length, but higher specificity of 92.4%.
Procedural Anxiety
Baseline scores on the short-form six-item Spielberger State-Trait Anxiety Inventory-6 (STAI-6) revealed notable levels of periprocedural anxiety. STAI-6 scores range from 20 to 80, with higher scores indicating more severe anxiety. In the VA study, scores ranged from 20 to 60, and most domains constituting the scores were the same before and after the procedure. Participants did, however, report a statistically significant decrease in sense of worry after EC and reported good tolerability for both EC and EG.
Offering an outsider’s perspective on the study, Joshua Sloan, DO, an esophageal gastroenterologist at University of Minnesota Medical Center in Minneapolis, said that with the acceleration of US rates of EAC, developing a nonendoscopic screening tool to improve identification of Barrett’s and perhaps early EAC is important. “The study by Greer et al helps support the use of nonendoscopic screening with EsoCheck and EsoGuard to identify these conditions,” he told GI & Hepatology News. “It will be interesting to see similar studies in the non-VA population as well. As the study notes, veterans are an enriched population with a higher prevalence of Barrett’s esophagus.”
Ultimately, Sloan added, “the hope is to increase our ability to identify and manage BE before it becomes EAC. Nonendoscopic screening tools have the potential to increase diagnosis and funnel the appropriate patients for endoscopic surveillance.”
The Bottom Line
“Calculations regarding effectiveness of the two-step screening strategy afforded by EC indicate that the burden of screening would be reduced by at least half (53%),” the authors wrote. Since the estimated size of the US screen-eligible population ranges from 19.7 million to 120.1 million, noninvasive tools could significantly decrease EGD procedures. A formal cost effectiveness analysis is being conducted and will be published separately.
This study was funded by a Department of Defense award.
Co-Author Chak reported device patents assigned to Case Western Reserve University and licensed to Lucid Diagnostics. The other authors had no competing interests to declare. Sloan disclosed speaking and/or advisory work for Sanofi-Regeneron, Phathom Pharmaceuticals, and Takeda Pharmaceuticals unrelated to his comments.
A version of this article appeared on Medscape.com.
EoE Prevalence in US Reaches 1 in 700, Costs $1B Annually
, according to recent research.
Although EoE has been considered a rare disease, the chronic condition is becoming more common, and healthcare providers should expect to encounter EoE in clinical settings, the study authors wrote.
“Our last assessment of the prevalence and burden of EoE was more than 10 years ago, and we had a strong suspicion we would continue to see increased numbers of patients with EoE and an increasing cost burden related to the condition in the United States,” said senior author Evan S. Dellon, MD, MPH, AGAF, professor of gastroenterology and hepatology and director of the Center for Esophageal Diseases and Swallowing at the University of North Carolina School of Medicine, Chapel Hill, North Carolina.
“EoE is becoming more common,” Dellon said. “Healthcare providers should expect to see EoE in their practices, including in the primary care setting, emergency departments, allergy practices, GI [gastrointestinal] practices, ENT clinics, and endoscopy suites.”
The study was published in Clinical Gastroenterology and Hepatology.
Estimating EoE Prevalence
Dellon and colleagues analyzed the Merative MarketScan Commercial Claims and Encounters and Medicare Fee-for-Service databases to calculate the annual prevalence of EoE, as well as age- and sex-stratified estimates standardized to the US population. They also calculated healthcare utilization, including medications and endoscopic procedures, to estimate annual EoE-associated costs. Since the EoE billing code was introduced in 2008, the analysis included 2009-2022 MarketScan and 2009-2017 Medicare data.
In the MarketScan database, the research team identified 20,435 EoE cases in 2022, with a mean age of 38 years, 16% younger than 18 years, 62% men, and 41% with a comorbid allergic disease code. The most common symptoms and diagnoses were dysphagia (39%), abdominal pain or dyspepsia (24%), and esophageal stricture (19%). Over time, patients also had previous codes for comorbid allergic diseases (64%), dysphagia (62%), or esophageal stricture (32%).
In the Medicare database, the research team identified 1913 EoE cases in 2017, with a mean age of 73 years, 47% men, 90% non-Hispanic White, and 36% with a comorbid allergic disease. The most common symptoms and diagnoses were dysphagia (49%), abdominal pain or dyspepsia (35%), and esophageal stricture (30%). Over time, patients also had codes for comorbid allergic diseases (64%), dysphagia (65%), or esophageal stricture (42%).
The database numbers translated to EoE prevalences of about 163 cases per 100,000 people in MarketScan in 2022 and 64 cases per 100,000 people in Medicare in 2017. Since 2009, there has been a fivefold increase in prevalence in both databases.
In MarketScan, the prevalence was higher among men than among women, at 204 vs 122 cases per 100,000 people. For both sexes, peak prevalence occurred between ages 40 and 44.
In Medicare, prevalence was also higher among men than among women, at 79 vs 55 cases per 100,000 people. Peak prevalence occurred between ages 65 and 69.
Standardized to the US population, EoE prevalence was 142.5 cases per 100,000 people, extrapolating to 472,380 cases. The overall prevalence was approximately 1 in 700, with rates of 1 in 617 for those younger than 65 years and 1 in 1562 for those aged ≥ 65 years.
“The rapidly increasing prevalence year over year for the entire timeframe of the study was surprising, as were our estimates of the total number of EoE patients in the US, which suggests that EoE is no longer a rare disease and is now seen in about 1 in 700 people,” Dellon said. “This almost triples our prior estimates of 1 in 2000 from 10 years ago, with all trends suggesting that the prevalence will continue to increase.”
Calculating EoE Costs
In terms of procedures, endoscopy with dilation or biopsy was used in about 60%-70% of patients with EoE in both MarketScan and Medicare during the years analyzed. In addition, upper endoscopy with biopsy was coded in 80%-90% of patients, guidewire-based dilation in 11%-17% of patients, and balloon-based dilation in 13%-20% of patients.
In terms of prescription medications, proton pump inhibitors (41%) and topical steroids (26%) were the most common in MarketScan in 2022, as well as in Medicare in 2017, at 32% and 9%, respectively.
When looking at costs by age and sex, the male cohort with the highest costs was aged 10-14 years, estimated at $106.7 million. Among the female cohort, the highest costs were associated with ages 15-19, estimated at $46.5 million.
Overall, total EoE-associated healthcare costs were estimated to be $1.04 billion in 2017, and when adjusted for inflation, the costs were estimated at $1.32 billion in 2024. This is likely an underestimate, the authors wrote, given that EoE prevalence has likely increased for ages 65 or older since 2017 and for all ages since 2022.
“Researching the prevalence and costs is essential to improving patient care by highlighting the growing burden of this recently recognized and growing chronic disease, guiding policy and insurer decisions, and advocating for better access to effective treatments and support for patients,” said Joy Chang, MD, assistant professor of medicine in the Division of Gastroenterology, University of Michigan, Ann Arbor, Michigan.Chang, who wasn’t involved with this study, specializes in eosinophilic GI diseases and researches patient-physician preferences and decision-making in EoE care.
“Clinicians should remain vigilant for symptoms, utilize guideline-based diagnostic approaches, and consider both medical and dietary treatment strategies to optimize patient outcomes and reduce long-term costs,” she said. “Increased awareness and timely intervention can help mitigate the growing impact of this chronic condition.”
The study was supported by a National Institutes of Health grant and used resources from the University of North Carolina Center for Gastrointestinal Biology and Disease. Dellon reported receiving research funding from and having consultant roles with numerous pharmaceutical companies and organizations. Chang reported having no relevant disclosures.
A version of this article appeared on Medscape.com.
, according to recent research.
Although EoE has been considered a rare disease, the chronic condition is becoming more common, and healthcare providers should expect to encounter EoE in clinical settings, the study authors wrote.
“Our last assessment of the prevalence and burden of EoE was more than 10 years ago, and we had a strong suspicion we would continue to see increased numbers of patients with EoE and an increasing cost burden related to the condition in the United States,” said senior author Evan S. Dellon, MD, MPH, AGAF, professor of gastroenterology and hepatology and director of the Center for Esophageal Diseases and Swallowing at the University of North Carolina School of Medicine, Chapel Hill, North Carolina.
“EoE is becoming more common,” Dellon said. “Healthcare providers should expect to see EoE in their practices, including in the primary care setting, emergency departments, allergy practices, GI [gastrointestinal] practices, ENT clinics, and endoscopy suites.”
The study was published in Clinical Gastroenterology and Hepatology.
Estimating EoE Prevalence
Dellon and colleagues analyzed the Merative MarketScan Commercial Claims and Encounters and Medicare Fee-for-Service databases to calculate the annual prevalence of EoE, as well as age- and sex-stratified estimates standardized to the US population. They also calculated healthcare utilization, including medications and endoscopic procedures, to estimate annual EoE-associated costs. Since the EoE billing code was introduced in 2008, the analysis included 2009-2022 MarketScan and 2009-2017 Medicare data.
In the MarketScan database, the research team identified 20,435 EoE cases in 2022, with a mean age of 38 years, 16% younger than 18 years, 62% men, and 41% with a comorbid allergic disease code. The most common symptoms and diagnoses were dysphagia (39%), abdominal pain or dyspepsia (24%), and esophageal stricture (19%). Over time, patients also had previous codes for comorbid allergic diseases (64%), dysphagia (62%), or esophageal stricture (32%).
In the Medicare database, the research team identified 1913 EoE cases in 2017, with a mean age of 73 years, 47% men, 90% non-Hispanic White, and 36% with a comorbid allergic disease. The most common symptoms and diagnoses were dysphagia (49%), abdominal pain or dyspepsia (35%), and esophageal stricture (30%). Over time, patients also had codes for comorbid allergic diseases (64%), dysphagia (65%), or esophageal stricture (42%).
The database numbers translated to EoE prevalences of about 163 cases per 100,000 people in MarketScan in 2022 and 64 cases per 100,000 people in Medicare in 2017. Since 2009, there has been a fivefold increase in prevalence in both databases.
In MarketScan, the prevalence was higher among men than among women, at 204 vs 122 cases per 100,000 people. For both sexes, peak prevalence occurred between ages 40 and 44.
In Medicare, prevalence was also higher among men than among women, at 79 vs 55 cases per 100,000 people. Peak prevalence occurred between ages 65 and 69.
Standardized to the US population, EoE prevalence was 142.5 cases per 100,000 people, extrapolating to 472,380 cases. The overall prevalence was approximately 1 in 700, with rates of 1 in 617 for those younger than 65 years and 1 in 1562 for those aged ≥ 65 years.
“The rapidly increasing prevalence year over year for the entire timeframe of the study was surprising, as were our estimates of the total number of EoE patients in the US, which suggests that EoE is no longer a rare disease and is now seen in about 1 in 700 people,” Dellon said. “This almost triples our prior estimates of 1 in 2000 from 10 years ago, with all trends suggesting that the prevalence will continue to increase.”
Calculating EoE Costs
In terms of procedures, endoscopy with dilation or biopsy was used in about 60%-70% of patients with EoE in both MarketScan and Medicare during the years analyzed. In addition, upper endoscopy with biopsy was coded in 80%-90% of patients, guidewire-based dilation in 11%-17% of patients, and balloon-based dilation in 13%-20% of patients.
In terms of prescription medications, proton pump inhibitors (41%) and topical steroids (26%) were the most common in MarketScan in 2022, as well as in Medicare in 2017, at 32% and 9%, respectively.
When looking at costs by age and sex, the male cohort with the highest costs was aged 10-14 years, estimated at $106.7 million. Among the female cohort, the highest costs were associated with ages 15-19, estimated at $46.5 million.
Overall, total EoE-associated healthcare costs were estimated to be $1.04 billion in 2017, and when adjusted for inflation, the costs were estimated at $1.32 billion in 2024. This is likely an underestimate, the authors wrote, given that EoE prevalence has likely increased for ages 65 or older since 2017 and for all ages since 2022.
“Researching the prevalence and costs is essential to improving patient care by highlighting the growing burden of this recently recognized and growing chronic disease, guiding policy and insurer decisions, and advocating for better access to effective treatments and support for patients,” said Joy Chang, MD, assistant professor of medicine in the Division of Gastroenterology, University of Michigan, Ann Arbor, Michigan.Chang, who wasn’t involved with this study, specializes in eosinophilic GI diseases and researches patient-physician preferences and decision-making in EoE care.
“Clinicians should remain vigilant for symptoms, utilize guideline-based diagnostic approaches, and consider both medical and dietary treatment strategies to optimize patient outcomes and reduce long-term costs,” she said. “Increased awareness and timely intervention can help mitigate the growing impact of this chronic condition.”
The study was supported by a National Institutes of Health grant and used resources from the University of North Carolina Center for Gastrointestinal Biology and Disease. Dellon reported receiving research funding from and having consultant roles with numerous pharmaceutical companies and organizations. Chang reported having no relevant disclosures.
A version of this article appeared on Medscape.com.
, according to recent research.
Although EoE has been considered a rare disease, the chronic condition is becoming more common, and healthcare providers should expect to encounter EoE in clinical settings, the study authors wrote.
“Our last assessment of the prevalence and burden of EoE was more than 10 years ago, and we had a strong suspicion we would continue to see increased numbers of patients with EoE and an increasing cost burden related to the condition in the United States,” said senior author Evan S. Dellon, MD, MPH, AGAF, professor of gastroenterology and hepatology and director of the Center for Esophageal Diseases and Swallowing at the University of North Carolina School of Medicine, Chapel Hill, North Carolina.
“EoE is becoming more common,” Dellon said. “Healthcare providers should expect to see EoE in their practices, including in the primary care setting, emergency departments, allergy practices, GI [gastrointestinal] practices, ENT clinics, and endoscopy suites.”
The study was published in Clinical Gastroenterology and Hepatology.
Estimating EoE Prevalence
Dellon and colleagues analyzed the Merative MarketScan Commercial Claims and Encounters and Medicare Fee-for-Service databases to calculate the annual prevalence of EoE, as well as age- and sex-stratified estimates standardized to the US population. They also calculated healthcare utilization, including medications and endoscopic procedures, to estimate annual EoE-associated costs. Since the EoE billing code was introduced in 2008, the analysis included 2009-2022 MarketScan and 2009-2017 Medicare data.
In the MarketScan database, the research team identified 20,435 EoE cases in 2022, with a mean age of 38 years, 16% younger than 18 years, 62% men, and 41% with a comorbid allergic disease code. The most common symptoms and diagnoses were dysphagia (39%), abdominal pain or dyspepsia (24%), and esophageal stricture (19%). Over time, patients also had previous codes for comorbid allergic diseases (64%), dysphagia (62%), or esophageal stricture (32%).
In the Medicare database, the research team identified 1913 EoE cases in 2017, with a mean age of 73 years, 47% men, 90% non-Hispanic White, and 36% with a comorbid allergic disease. The most common symptoms and diagnoses were dysphagia (49%), abdominal pain or dyspepsia (35%), and esophageal stricture (30%). Over time, patients also had codes for comorbid allergic diseases (64%), dysphagia (65%), or esophageal stricture (42%).
The database numbers translated to EoE prevalences of about 163 cases per 100,000 people in MarketScan in 2022 and 64 cases per 100,000 people in Medicare in 2017. Since 2009, there has been a fivefold increase in prevalence in both databases.
In MarketScan, the prevalence was higher among men than among women, at 204 vs 122 cases per 100,000 people. For both sexes, peak prevalence occurred between ages 40 and 44.
In Medicare, prevalence was also higher among men than among women, at 79 vs 55 cases per 100,000 people. Peak prevalence occurred between ages 65 and 69.
Standardized to the US population, EoE prevalence was 142.5 cases per 100,000 people, extrapolating to 472,380 cases. The overall prevalence was approximately 1 in 700, with rates of 1 in 617 for those younger than 65 years and 1 in 1562 for those aged ≥ 65 years.
“The rapidly increasing prevalence year over year for the entire timeframe of the study was surprising, as were our estimates of the total number of EoE patients in the US, which suggests that EoE is no longer a rare disease and is now seen in about 1 in 700 people,” Dellon said. “This almost triples our prior estimates of 1 in 2000 from 10 years ago, with all trends suggesting that the prevalence will continue to increase.”
Calculating EoE Costs
In terms of procedures, endoscopy with dilation or biopsy was used in about 60%-70% of patients with EoE in both MarketScan and Medicare during the years analyzed. In addition, upper endoscopy with biopsy was coded in 80%-90% of patients, guidewire-based dilation in 11%-17% of patients, and balloon-based dilation in 13%-20% of patients.
In terms of prescription medications, proton pump inhibitors (41%) and topical steroids (26%) were the most common in MarketScan in 2022, as well as in Medicare in 2017, at 32% and 9%, respectively.
When looking at costs by age and sex, the male cohort with the highest costs was aged 10-14 years, estimated at $106.7 million. Among the female cohort, the highest costs were associated with ages 15-19, estimated at $46.5 million.
Overall, total EoE-associated healthcare costs were estimated to be $1.04 billion in 2017, and when adjusted for inflation, the costs were estimated at $1.32 billion in 2024. This is likely an underestimate, the authors wrote, given that EoE prevalence has likely increased for ages 65 or older since 2017 and for all ages since 2022.
“Researching the prevalence and costs is essential to improving patient care by highlighting the growing burden of this recently recognized and growing chronic disease, guiding policy and insurer decisions, and advocating for better access to effective treatments and support for patients,” said Joy Chang, MD, assistant professor of medicine in the Division of Gastroenterology, University of Michigan, Ann Arbor, Michigan.Chang, who wasn’t involved with this study, specializes in eosinophilic GI diseases and researches patient-physician preferences and decision-making in EoE care.
“Clinicians should remain vigilant for symptoms, utilize guideline-based diagnostic approaches, and consider both medical and dietary treatment strategies to optimize patient outcomes and reduce long-term costs,” she said. “Increased awareness and timely intervention can help mitigate the growing impact of this chronic condition.”
The study was supported by a National Institutes of Health grant and used resources from the University of North Carolina Center for Gastrointestinal Biology and Disease. Dellon reported receiving research funding from and having consultant roles with numerous pharmaceutical companies and organizations. Chang reported having no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
AI Algorithm Predicts Transfusion Need, Mortality Risk in Acute GI Bleeds
SAN DIEGO — , researchers reported at Digestive Disease Week® (DDW) 2025.
Acute GI bleeding is the most common cause of digestive disease–related hospitalization, with an estimated 500,000 hospital admissions annually. It’s known that predicting the need for red blood cell transfusion in the first 24 hours may improve resuscitation and decrease both morbidity and mortality.
However, an existing clinical score known as the Rockall Score does not perform well for predicting mortality, Xi (Nicole) Zhang, an MD-PhD student at McGill University, Montreal, Quebec, Canada, told attendees at DDW. With an area under the curve of 0.65-0.75, better prediction is needed, said Zhang, whose coresearchers included Dennis Shung, MD, MHS, PhD, director of Applied Artificial Intelligence at Yale University School of Medicine, New Haven, Connecticut.
“We’d like to predict multiple outcomes in addition to mortality,” said Zhang, who is also a student at the Mila-Quebec Artificial Intelligence Institute.
As a result, the researchers turned to the TFM approach, applying it to ICU patients with acute GI bleeding to predict both the need for transfusion and in-hospital mortality risk. The all-cause mortality rate is up to 11%, according to a 2020 study by James Y. W. Lau, MD, and colleagues. The rebleeding rate of nonvariceal upper GI bleeds is up to 10.4%. Zhang said the rebleeding rate for variceal upper gastrointestinal bleeding is up to 65%.
The AI method the researchers used outperformed a standard deep learning model at predicting the need for transfusion and estimating mortality risk.
Defining the AI Framework
“Probabilistic flow matching is a class of generative artificial intelligence that learns how a simple distribution becomes a more complex distribution with ordinary differential equations,” Zhang told GI & Hepatology News. “For example, if you had a few lines and shapes you could learn how it could become a detailed portrait of a face. In our case, we start with a few blood pressure and heart rate measurements and learn the pattern of blood pressures and heart rates over time, particularly if they reflect clinical deterioration with hemodynamic instability.”
Another way to think about the underlying algorithm, Zhang said, is to think about a river with boats where the river flow determines where the boats end up. “We are trying to direct the boat to the correct dock by adjusting the flow of water in the canal. In this case we are mapping the distribution with the first few data points to the distribution with the entire patient trajectory.”
The information gained, she said, could be helpful in timing endoscopic evaluation or allocating red blood cell products for emergent transfusion.
Study Details
The researchers evaluated a cohort of 2602 patients admitted to the ICU, identified from the publicly available MIMIC-III database. They divided the patients into a training set of 2342 patients and an internal validation set of 260 patients. Input variables were severe liver disease comorbidity, administration of vasopressor medications, mean arterial blood pressure, and heart rate over the first 24 hours.
Excluded was hemoglobin, since the point was to test the trajectory of hemodynamic parameters independent of hemoglobin thresholds used to guide red blood cell transfusion.
The outcome measures were administration of packed red blood cell transfusion within 24 hours and all-cause hospital mortality.
The TFM was more accurate than a standard deep learning model in predicting red blood cell transfusion, with an accuracy of 93.6% vs 43.2%; P ≤ .001. It was also more accurate at predicting all-cause in-hospital mortality, with an accuracy of 89.5% vs 42.5%, P = .01.
The researchers concluded that the TFM approach was able to predict the hemodynamic trajectories of patients with acute GI bleeding defined as deviation and outperformed the baseline from the measured mean arterial pressure and heart rate.
Expert Perspective
“This is an exciting proof-of-concept study that shows generative AI methods may be applied to complex datasets in order to improve on our current predictive models and improve patient care,” said Jeremy Glissen Brown, MD, MSc, an assistant professor of medicine and a practicing gastroenterologist at Duke University who has published research on the use of AI in clinical practice. He reviewed the study for GI & Hepatology News but was not involved in the research.
“Future work will likely look into the implementation of a version of this model on real-time data.” he said. “We are at an exciting inflection point in predictive models within GI and clinical medicine. Predictive models based on deep learning and generative AI hold the promise of improving how we predict and treat disease states, but the excitement being generated with studies such as this needs to be balanced with the trade-offs inherent to the current paradigm of deep learning and generative models compared to more traditional regression-based models. These include many of the same ‘black box’ explainability questions that have risen in the age of convolutional neural networks as well as some method-specific questions due to the continuous and implicit nature of TFM.”
Elaborating on that, Glissen Brown said: “TFM, like many deep learning techniques, raises concerns about explainability that we’ve long seen with convolutional neural networks — the ‘black box’ problem, where it’s difficult to interpret exactly how and why the model arrives at a particular decision. But TFM also introduces unique challenges due to its continuous and implicit formulation. Since it often learns flows without explicitly defining intermediate representations or steps, it can be harder to trace the logic or pathways it uses to connect inputs to outputs. This makes standard interpretability tools less effective and calls for new techniques tailored to these continuous architectures.”
“This approach could have a real clinical impact,” said Robert Hirten, MD, associate professor of medicine and artificial intelligence, Icahn School of Medicine at Mount Sinai, New York City, who also reviewed the study. “Accurately predicting transfusion needs and mortality risk in real time could support earlier, more targeted interventions for high-risk patients. While these findings still need to be validated in prospective studies, it could enhance ICU decision-making and resource allocation.”
“For the practicing gastroenterologist, we envision this system could help them figure out when to perform endoscopy in a patient admitted with acute gastrointestinal bleeding in the ICU at very high risk of exsanguination,” Zhang told GI & Hepatology News.
The approach, the researchers said, will be useful in identifying unique patient characteristics, make possible the identification of high-risk patients and lead to more personalized medicine.
Hirten, Zhang, and Shung had no disclosures. Glissen Brown reported consulting relationships with Medtronic, OdinVision, Doximity, and Olympus. The National Institutes of Health funded this study.
A version of this article appeared on Medscape.com.
SAN DIEGO — , researchers reported at Digestive Disease Week® (DDW) 2025.
Acute GI bleeding is the most common cause of digestive disease–related hospitalization, with an estimated 500,000 hospital admissions annually. It’s known that predicting the need for red blood cell transfusion in the first 24 hours may improve resuscitation and decrease both morbidity and mortality.
However, an existing clinical score known as the Rockall Score does not perform well for predicting mortality, Xi (Nicole) Zhang, an MD-PhD student at McGill University, Montreal, Quebec, Canada, told attendees at DDW. With an area under the curve of 0.65-0.75, better prediction is needed, said Zhang, whose coresearchers included Dennis Shung, MD, MHS, PhD, director of Applied Artificial Intelligence at Yale University School of Medicine, New Haven, Connecticut.
“We’d like to predict multiple outcomes in addition to mortality,” said Zhang, who is also a student at the Mila-Quebec Artificial Intelligence Institute.
As a result, the researchers turned to the TFM approach, applying it to ICU patients with acute GI bleeding to predict both the need for transfusion and in-hospital mortality risk. The all-cause mortality rate is up to 11%, according to a 2020 study by James Y. W. Lau, MD, and colleagues. The rebleeding rate of nonvariceal upper GI bleeds is up to 10.4%. Zhang said the rebleeding rate for variceal upper gastrointestinal bleeding is up to 65%.
The AI method the researchers used outperformed a standard deep learning model at predicting the need for transfusion and estimating mortality risk.
Defining the AI Framework
“Probabilistic flow matching is a class of generative artificial intelligence that learns how a simple distribution becomes a more complex distribution with ordinary differential equations,” Zhang told GI & Hepatology News. “For example, if you had a few lines and shapes you could learn how it could become a detailed portrait of a face. In our case, we start with a few blood pressure and heart rate measurements and learn the pattern of blood pressures and heart rates over time, particularly if they reflect clinical deterioration with hemodynamic instability.”
Another way to think about the underlying algorithm, Zhang said, is to think about a river with boats where the river flow determines where the boats end up. “We are trying to direct the boat to the correct dock by adjusting the flow of water in the canal. In this case we are mapping the distribution with the first few data points to the distribution with the entire patient trajectory.”
The information gained, she said, could be helpful in timing endoscopic evaluation or allocating red blood cell products for emergent transfusion.
Study Details
The researchers evaluated a cohort of 2602 patients admitted to the ICU, identified from the publicly available MIMIC-III database. They divided the patients into a training set of 2342 patients and an internal validation set of 260 patients. Input variables were severe liver disease comorbidity, administration of vasopressor medications, mean arterial blood pressure, and heart rate over the first 24 hours.
Excluded was hemoglobin, since the point was to test the trajectory of hemodynamic parameters independent of hemoglobin thresholds used to guide red blood cell transfusion.
The outcome measures were administration of packed red blood cell transfusion within 24 hours and all-cause hospital mortality.
The TFM was more accurate than a standard deep learning model in predicting red blood cell transfusion, with an accuracy of 93.6% vs 43.2%; P ≤ .001. It was also more accurate at predicting all-cause in-hospital mortality, with an accuracy of 89.5% vs 42.5%, P = .01.
The researchers concluded that the TFM approach was able to predict the hemodynamic trajectories of patients with acute GI bleeding defined as deviation and outperformed the baseline from the measured mean arterial pressure and heart rate.
Expert Perspective
“This is an exciting proof-of-concept study that shows generative AI methods may be applied to complex datasets in order to improve on our current predictive models and improve patient care,” said Jeremy Glissen Brown, MD, MSc, an assistant professor of medicine and a practicing gastroenterologist at Duke University who has published research on the use of AI in clinical practice. He reviewed the study for GI & Hepatology News but was not involved in the research.
“Future work will likely look into the implementation of a version of this model on real-time data.” he said. “We are at an exciting inflection point in predictive models within GI and clinical medicine. Predictive models based on deep learning and generative AI hold the promise of improving how we predict and treat disease states, but the excitement being generated with studies such as this needs to be balanced with the trade-offs inherent to the current paradigm of deep learning and generative models compared to more traditional regression-based models. These include many of the same ‘black box’ explainability questions that have risen in the age of convolutional neural networks as well as some method-specific questions due to the continuous and implicit nature of TFM.”
Elaborating on that, Glissen Brown said: “TFM, like many deep learning techniques, raises concerns about explainability that we’ve long seen with convolutional neural networks — the ‘black box’ problem, where it’s difficult to interpret exactly how and why the model arrives at a particular decision. But TFM also introduces unique challenges due to its continuous and implicit formulation. Since it often learns flows without explicitly defining intermediate representations or steps, it can be harder to trace the logic or pathways it uses to connect inputs to outputs. This makes standard interpretability tools less effective and calls for new techniques tailored to these continuous architectures.”
“This approach could have a real clinical impact,” said Robert Hirten, MD, associate professor of medicine and artificial intelligence, Icahn School of Medicine at Mount Sinai, New York City, who also reviewed the study. “Accurately predicting transfusion needs and mortality risk in real time could support earlier, more targeted interventions for high-risk patients. While these findings still need to be validated in prospective studies, it could enhance ICU decision-making and resource allocation.”
“For the practicing gastroenterologist, we envision this system could help them figure out when to perform endoscopy in a patient admitted with acute gastrointestinal bleeding in the ICU at very high risk of exsanguination,” Zhang told GI & Hepatology News.
The approach, the researchers said, will be useful in identifying unique patient characteristics, make possible the identification of high-risk patients and lead to more personalized medicine.
Hirten, Zhang, and Shung had no disclosures. Glissen Brown reported consulting relationships with Medtronic, OdinVision, Doximity, and Olympus. The National Institutes of Health funded this study.
A version of this article appeared on Medscape.com.
SAN DIEGO — , researchers reported at Digestive Disease Week® (DDW) 2025.
Acute GI bleeding is the most common cause of digestive disease–related hospitalization, with an estimated 500,000 hospital admissions annually. It’s known that predicting the need for red blood cell transfusion in the first 24 hours may improve resuscitation and decrease both morbidity and mortality.
However, an existing clinical score known as the Rockall Score does not perform well for predicting mortality, Xi (Nicole) Zhang, an MD-PhD student at McGill University, Montreal, Quebec, Canada, told attendees at DDW. With an area under the curve of 0.65-0.75, better prediction is needed, said Zhang, whose coresearchers included Dennis Shung, MD, MHS, PhD, director of Applied Artificial Intelligence at Yale University School of Medicine, New Haven, Connecticut.
“We’d like to predict multiple outcomes in addition to mortality,” said Zhang, who is also a student at the Mila-Quebec Artificial Intelligence Institute.
As a result, the researchers turned to the TFM approach, applying it to ICU patients with acute GI bleeding to predict both the need for transfusion and in-hospital mortality risk. The all-cause mortality rate is up to 11%, according to a 2020 study by James Y. W. Lau, MD, and colleagues. The rebleeding rate of nonvariceal upper GI bleeds is up to 10.4%. Zhang said the rebleeding rate for variceal upper gastrointestinal bleeding is up to 65%.
The AI method the researchers used outperformed a standard deep learning model at predicting the need for transfusion and estimating mortality risk.
Defining the AI Framework
“Probabilistic flow matching is a class of generative artificial intelligence that learns how a simple distribution becomes a more complex distribution with ordinary differential equations,” Zhang told GI & Hepatology News. “For example, if you had a few lines and shapes you could learn how it could become a detailed portrait of a face. In our case, we start with a few blood pressure and heart rate measurements and learn the pattern of blood pressures and heart rates over time, particularly if they reflect clinical deterioration with hemodynamic instability.”
Another way to think about the underlying algorithm, Zhang said, is to think about a river with boats where the river flow determines where the boats end up. “We are trying to direct the boat to the correct dock by adjusting the flow of water in the canal. In this case we are mapping the distribution with the first few data points to the distribution with the entire patient trajectory.”
The information gained, she said, could be helpful in timing endoscopic evaluation or allocating red blood cell products for emergent transfusion.
Study Details
The researchers evaluated a cohort of 2602 patients admitted to the ICU, identified from the publicly available MIMIC-III database. They divided the patients into a training set of 2342 patients and an internal validation set of 260 patients. Input variables were severe liver disease comorbidity, administration of vasopressor medications, mean arterial blood pressure, and heart rate over the first 24 hours.
Excluded was hemoglobin, since the point was to test the trajectory of hemodynamic parameters independent of hemoglobin thresholds used to guide red blood cell transfusion.
The outcome measures were administration of packed red blood cell transfusion within 24 hours and all-cause hospital mortality.
The TFM was more accurate than a standard deep learning model in predicting red blood cell transfusion, with an accuracy of 93.6% vs 43.2%; P ≤ .001. It was also more accurate at predicting all-cause in-hospital mortality, with an accuracy of 89.5% vs 42.5%, P = .01.
The researchers concluded that the TFM approach was able to predict the hemodynamic trajectories of patients with acute GI bleeding defined as deviation and outperformed the baseline from the measured mean arterial pressure and heart rate.
Expert Perspective
“This is an exciting proof-of-concept study that shows generative AI methods may be applied to complex datasets in order to improve on our current predictive models and improve patient care,” said Jeremy Glissen Brown, MD, MSc, an assistant professor of medicine and a practicing gastroenterologist at Duke University who has published research on the use of AI in clinical practice. He reviewed the study for GI & Hepatology News but was not involved in the research.
“Future work will likely look into the implementation of a version of this model on real-time data.” he said. “We are at an exciting inflection point in predictive models within GI and clinical medicine. Predictive models based on deep learning and generative AI hold the promise of improving how we predict and treat disease states, but the excitement being generated with studies such as this needs to be balanced with the trade-offs inherent to the current paradigm of deep learning and generative models compared to more traditional regression-based models. These include many of the same ‘black box’ explainability questions that have risen in the age of convolutional neural networks as well as some method-specific questions due to the continuous and implicit nature of TFM.”
Elaborating on that, Glissen Brown said: “TFM, like many deep learning techniques, raises concerns about explainability that we’ve long seen with convolutional neural networks — the ‘black box’ problem, where it’s difficult to interpret exactly how and why the model arrives at a particular decision. But TFM also introduces unique challenges due to its continuous and implicit formulation. Since it often learns flows without explicitly defining intermediate representations or steps, it can be harder to trace the logic or pathways it uses to connect inputs to outputs. This makes standard interpretability tools less effective and calls for new techniques tailored to these continuous architectures.”
“This approach could have a real clinical impact,” said Robert Hirten, MD, associate professor of medicine and artificial intelligence, Icahn School of Medicine at Mount Sinai, New York City, who also reviewed the study. “Accurately predicting transfusion needs and mortality risk in real time could support earlier, more targeted interventions for high-risk patients. While these findings still need to be validated in prospective studies, it could enhance ICU decision-making and resource allocation.”
“For the practicing gastroenterologist, we envision this system could help them figure out when to perform endoscopy in a patient admitted with acute gastrointestinal bleeding in the ICU at very high risk of exsanguination,” Zhang told GI & Hepatology News.
The approach, the researchers said, will be useful in identifying unique patient characteristics, make possible the identification of high-risk patients and lead to more personalized medicine.
Hirten, Zhang, and Shung had no disclosures. Glissen Brown reported consulting relationships with Medtronic, OdinVision, Doximity, and Olympus. The National Institutes of Health funded this study.
A version of this article appeared on Medscape.com.
FROM DDW 2025
Blood Detection Capsule Helpful in Suspected Upper GI Bleeding
SAN DIEGO — , a study found.
Notably, patients with negative capsule results had shorter hospital stays and lower acuity markers, and in more than one third of cases, an esophagogastroduodenoscopy (EGD) was avoided altogether without any observed adverse events or readmissions, the study team found.
“Our study shows that this novel capsule that detects blood in the upper GI tract (PillSense) was highly sensitive and specific (> 90%) for detecting recent or active upper GI blood, influenced clinical management in 80% of cases and allowed about one third of patients to be safely discharged from the emergency department, with close outpatient follow-up,” Linda Lee, MD, AGAF, medical director of endoscopy, Brigham and Women’s Hospital and associate professor of medicine, Harvard Medical School, Boston, told GI & Hepatology News.
The study was presented at Digestive Disease Week® (DDW) 2025.
Real-World Insights
EGD is the gold standard for diagnosing suspected upper GI bleeding, but limited access to timely EGD complicates diagnosis and resource allocation.
Approved by the US Food and Drug Administration, PillSense (EnteraSense) is an ingestible capsule with a reusable receiver that provides a rapid, noninvasive method for detecting upper GI bleeding. The capsule analyzes light absorption to identify blood and transmits the result within 10 minutes.
Lee and colleagues evaluated the real-world impact of this point-of-care device on clinical triage and resource allocation, while assessing its safety profile.
They analyzed data on 43 patients (mean age 60 years; 72% men) with clinical suspicion of upper GI bleeding in whom the device was used. The most common symptoms were symptomatic anemia (70%), melena (67%), and hematemesis (33%).
Sixteen PillSense studies (37%) were positive for blood detection, and 27 (63%) were negative.
Compared to patients with a positive capsule results, those without blood detected by the capsule had shorter hospital stays (mean, 3.8 vs 13.4 days, P = .02), lower GBS scores (mean, 7.93 vs 12.81; P = .005), and fewer units of blood transfused (mean, 1.19 vs 10.94; P = .01) and were less apt to be hemodynamically unstable (5 vs 8 patients; P = .03).
Capsule results influenced clinical management in 80% of cases, leading to avoidance of EGD in 37% and prioritization of urgent EGD in 18% (all had active bleeding on EGD).
Capsule use improved resource allocation in 51% of cases. This included 12 patients who were discharged from the ED, six who were assigned an inpatient bed early, and four who underwent expedited colonoscopy as upper GI bleeding was ruled out, they noted.
Among the eight patients who did not undergo EGD, there were no readmissions within 30 days and no adverse events. There were no capsule-related adverse events.
“Clinicians should consider using this novel capsule PillSense as another data point in the management of suspected upper GI bleed,” Lee told GI & Hepatology News.
“This could include in helping to triage patients for safe discharge from the ED or to more urgent endoscopy, to differentiate between upper vs lower GI bleed and to manage ICU patients with possible rebleeding,” Lee said.
Important Real-World Evidence
Reached for comment, Shahin Ayazi, MD, esophageal surgeon, Director, Allegheny Health Network Chevalier Jackson Esophageal Research Center, Pittsburgh, Pennsylvania, said this study is important for several reasons.
“Prior investigations have established that PillSense possesses a high negative predictive value for detecting upper GI bleeding and have speculated on its utility in triage, decision-making, and potentially avoiding unnecessary endoscopy. This study is important because it substantiates that speculation with clinical data,” Ayazi, who wasn’t involved in the study, told GI & Hepatology News.
“These findings support the capsule’s practical application in patient stratification and clinical workflow, particularly when diagnostic uncertainty is high and endoscopic resources are limited,” Ayazi noted.
In his experience, PillSense is “highly useful as a triage adjunct in the evaluation of suspected upper GI bleeding. It provides direct and objective evidence as to whether blood is currently present in the stomach,” he said.
“In patients whose presentation is ambiguous or whose clinical scores fall into an intermediate risk zone, this binary result can provide clarity that subjective assessment alone may not achieve. This is particularly relevant in settings where the goal is to perform endoscopy within 24 hours, but the volume of consults exceeds procedural capacity,” Ayazi explained.
“In such scenarios, PillSense enables physicians to stratify patients based on objective evidence of active bleeding, helping to prioritize those who require urgent endoscopy and defer or even avoid endoscopic evaluation in those who do not. The result is a more efficient allocation of endoscopic resources without compromising patient safety,” he added.
Ayazi cautioned that the PillSense capsule should not be used as a replacement for clinical evaluation or established risk stratification protocols.
“It is intended for hemodynamically stable patients and has not been validated in cases of active or massive bleeding. Its diagnostic yield depends on the presence of blood in the stomach at the time of capsule transit; intermittent or proximal bleeding that has ceased may not be detected, introducing the potential for false-negative results,” Ayazi told GI & Hepatology News.
“However, in prior studies, the negative predictive value was high, and in the present study, no adverse outcomes were observed in patients who did not undergo endoscopy following a negative PillSense result,” Ayazi noted.
“It must also be understood that PillSense does not localize the source of bleeding or replace endoscopy in patients with a high likelihood of active hemorrhage. It is not designed to detect bleeding from the lower GI tract or distal small bowel. Rather, it serves as an adjunct that can provide immediate clarity when the need for endoscopy is uncertain, and should be interpreted within the broader context of clinical findings, laboratory data, and established risk stratification tools,” he added.
The study had no specific funding. Lee and Ayazi had no relevant disclosures.
A version of this article appeared on Medscape.com.
SAN DIEGO — , a study found.
Notably, patients with negative capsule results had shorter hospital stays and lower acuity markers, and in more than one third of cases, an esophagogastroduodenoscopy (EGD) was avoided altogether without any observed adverse events or readmissions, the study team found.
“Our study shows that this novel capsule that detects blood in the upper GI tract (PillSense) was highly sensitive and specific (> 90%) for detecting recent or active upper GI blood, influenced clinical management in 80% of cases and allowed about one third of patients to be safely discharged from the emergency department, with close outpatient follow-up,” Linda Lee, MD, AGAF, medical director of endoscopy, Brigham and Women’s Hospital and associate professor of medicine, Harvard Medical School, Boston, told GI & Hepatology News.
The study was presented at Digestive Disease Week® (DDW) 2025.
Real-World Insights
EGD is the gold standard for diagnosing suspected upper GI bleeding, but limited access to timely EGD complicates diagnosis and resource allocation.
Approved by the US Food and Drug Administration, PillSense (EnteraSense) is an ingestible capsule with a reusable receiver that provides a rapid, noninvasive method for detecting upper GI bleeding. The capsule analyzes light absorption to identify blood and transmits the result within 10 minutes.
Lee and colleagues evaluated the real-world impact of this point-of-care device on clinical triage and resource allocation, while assessing its safety profile.
They analyzed data on 43 patients (mean age 60 years; 72% men) with clinical suspicion of upper GI bleeding in whom the device was used. The most common symptoms were symptomatic anemia (70%), melena (67%), and hematemesis (33%).
Sixteen PillSense studies (37%) were positive for blood detection, and 27 (63%) were negative.
Compared to patients with a positive capsule results, those without blood detected by the capsule had shorter hospital stays (mean, 3.8 vs 13.4 days, P = .02), lower GBS scores (mean, 7.93 vs 12.81; P = .005), and fewer units of blood transfused (mean, 1.19 vs 10.94; P = .01) and were less apt to be hemodynamically unstable (5 vs 8 patients; P = .03).
Capsule results influenced clinical management in 80% of cases, leading to avoidance of EGD in 37% and prioritization of urgent EGD in 18% (all had active bleeding on EGD).
Capsule use improved resource allocation in 51% of cases. This included 12 patients who were discharged from the ED, six who were assigned an inpatient bed early, and four who underwent expedited colonoscopy as upper GI bleeding was ruled out, they noted.
Among the eight patients who did not undergo EGD, there were no readmissions within 30 days and no adverse events. There were no capsule-related adverse events.
“Clinicians should consider using this novel capsule PillSense as another data point in the management of suspected upper GI bleed,” Lee told GI & Hepatology News.
“This could include in helping to triage patients for safe discharge from the ED or to more urgent endoscopy, to differentiate between upper vs lower GI bleed and to manage ICU patients with possible rebleeding,” Lee said.
Important Real-World Evidence
Reached for comment, Shahin Ayazi, MD, esophageal surgeon, Director, Allegheny Health Network Chevalier Jackson Esophageal Research Center, Pittsburgh, Pennsylvania, said this study is important for several reasons.
“Prior investigations have established that PillSense possesses a high negative predictive value for detecting upper GI bleeding and have speculated on its utility in triage, decision-making, and potentially avoiding unnecessary endoscopy. This study is important because it substantiates that speculation with clinical data,” Ayazi, who wasn’t involved in the study, told GI & Hepatology News.
“These findings support the capsule’s practical application in patient stratification and clinical workflow, particularly when diagnostic uncertainty is high and endoscopic resources are limited,” Ayazi noted.
In his experience, PillSense is “highly useful as a triage adjunct in the evaluation of suspected upper GI bleeding. It provides direct and objective evidence as to whether blood is currently present in the stomach,” he said.
“In patients whose presentation is ambiguous or whose clinical scores fall into an intermediate risk zone, this binary result can provide clarity that subjective assessment alone may not achieve. This is particularly relevant in settings where the goal is to perform endoscopy within 24 hours, but the volume of consults exceeds procedural capacity,” Ayazi explained.
“In such scenarios, PillSense enables physicians to stratify patients based on objective evidence of active bleeding, helping to prioritize those who require urgent endoscopy and defer or even avoid endoscopic evaluation in those who do not. The result is a more efficient allocation of endoscopic resources without compromising patient safety,” he added.
Ayazi cautioned that the PillSense capsule should not be used as a replacement for clinical evaluation or established risk stratification protocols.
“It is intended for hemodynamically stable patients and has not been validated in cases of active or massive bleeding. Its diagnostic yield depends on the presence of blood in the stomach at the time of capsule transit; intermittent or proximal bleeding that has ceased may not be detected, introducing the potential for false-negative results,” Ayazi told GI & Hepatology News.
“However, in prior studies, the negative predictive value was high, and in the present study, no adverse outcomes were observed in patients who did not undergo endoscopy following a negative PillSense result,” Ayazi noted.
“It must also be understood that PillSense does not localize the source of bleeding or replace endoscopy in patients with a high likelihood of active hemorrhage. It is not designed to detect bleeding from the lower GI tract or distal small bowel. Rather, it serves as an adjunct that can provide immediate clarity when the need for endoscopy is uncertain, and should be interpreted within the broader context of clinical findings, laboratory data, and established risk stratification tools,” he added.
The study had no specific funding. Lee and Ayazi had no relevant disclosures.
A version of this article appeared on Medscape.com.
SAN DIEGO — , a study found.
Notably, patients with negative capsule results had shorter hospital stays and lower acuity markers, and in more than one third of cases, an esophagogastroduodenoscopy (EGD) was avoided altogether without any observed adverse events or readmissions, the study team found.
“Our study shows that this novel capsule that detects blood in the upper GI tract (PillSense) was highly sensitive and specific (> 90%) for detecting recent or active upper GI blood, influenced clinical management in 80% of cases and allowed about one third of patients to be safely discharged from the emergency department, with close outpatient follow-up,” Linda Lee, MD, AGAF, medical director of endoscopy, Brigham and Women’s Hospital and associate professor of medicine, Harvard Medical School, Boston, told GI & Hepatology News.
The study was presented at Digestive Disease Week® (DDW) 2025.
Real-World Insights
EGD is the gold standard for diagnosing suspected upper GI bleeding, but limited access to timely EGD complicates diagnosis and resource allocation.
Approved by the US Food and Drug Administration, PillSense (EnteraSense) is an ingestible capsule with a reusable receiver that provides a rapid, noninvasive method for detecting upper GI bleeding. The capsule analyzes light absorption to identify blood and transmits the result within 10 minutes.
Lee and colleagues evaluated the real-world impact of this point-of-care device on clinical triage and resource allocation, while assessing its safety profile.
They analyzed data on 43 patients (mean age 60 years; 72% men) with clinical suspicion of upper GI bleeding in whom the device was used. The most common symptoms were symptomatic anemia (70%), melena (67%), and hematemesis (33%).
Sixteen PillSense studies (37%) were positive for blood detection, and 27 (63%) were negative.
Compared to patients with a positive capsule results, those without blood detected by the capsule had shorter hospital stays (mean, 3.8 vs 13.4 days, P = .02), lower GBS scores (mean, 7.93 vs 12.81; P = .005), and fewer units of blood transfused (mean, 1.19 vs 10.94; P = .01) and were less apt to be hemodynamically unstable (5 vs 8 patients; P = .03).
Capsule results influenced clinical management in 80% of cases, leading to avoidance of EGD in 37% and prioritization of urgent EGD in 18% (all had active bleeding on EGD).
Capsule use improved resource allocation in 51% of cases. This included 12 patients who were discharged from the ED, six who were assigned an inpatient bed early, and four who underwent expedited colonoscopy as upper GI bleeding was ruled out, they noted.
Among the eight patients who did not undergo EGD, there were no readmissions within 30 days and no adverse events. There were no capsule-related adverse events.
“Clinicians should consider using this novel capsule PillSense as another data point in the management of suspected upper GI bleed,” Lee told GI & Hepatology News.
“This could include in helping to triage patients for safe discharge from the ED or to more urgent endoscopy, to differentiate between upper vs lower GI bleed and to manage ICU patients with possible rebleeding,” Lee said.
Important Real-World Evidence
Reached for comment, Shahin Ayazi, MD, esophageal surgeon, Director, Allegheny Health Network Chevalier Jackson Esophageal Research Center, Pittsburgh, Pennsylvania, said this study is important for several reasons.
“Prior investigations have established that PillSense possesses a high negative predictive value for detecting upper GI bleeding and have speculated on its utility in triage, decision-making, and potentially avoiding unnecessary endoscopy. This study is important because it substantiates that speculation with clinical data,” Ayazi, who wasn’t involved in the study, told GI & Hepatology News.
“These findings support the capsule’s practical application in patient stratification and clinical workflow, particularly when diagnostic uncertainty is high and endoscopic resources are limited,” Ayazi noted.
In his experience, PillSense is “highly useful as a triage adjunct in the evaluation of suspected upper GI bleeding. It provides direct and objective evidence as to whether blood is currently present in the stomach,” he said.
“In patients whose presentation is ambiguous or whose clinical scores fall into an intermediate risk zone, this binary result can provide clarity that subjective assessment alone may not achieve. This is particularly relevant in settings where the goal is to perform endoscopy within 24 hours, but the volume of consults exceeds procedural capacity,” Ayazi explained.
“In such scenarios, PillSense enables physicians to stratify patients based on objective evidence of active bleeding, helping to prioritize those who require urgent endoscopy and defer or even avoid endoscopic evaluation in those who do not. The result is a more efficient allocation of endoscopic resources without compromising patient safety,” he added.
Ayazi cautioned that the PillSense capsule should not be used as a replacement for clinical evaluation or established risk stratification protocols.
“It is intended for hemodynamically stable patients and has not been validated in cases of active or massive bleeding. Its diagnostic yield depends on the presence of blood in the stomach at the time of capsule transit; intermittent or proximal bleeding that has ceased may not be detected, introducing the potential for false-negative results,” Ayazi told GI & Hepatology News.
“However, in prior studies, the negative predictive value was high, and in the present study, no adverse outcomes were observed in patients who did not undergo endoscopy following a negative PillSense result,” Ayazi noted.
“It must also be understood that PillSense does not localize the source of bleeding or replace endoscopy in patients with a high likelihood of active hemorrhage. It is not designed to detect bleeding from the lower GI tract or distal small bowel. Rather, it serves as an adjunct that can provide immediate clarity when the need for endoscopy is uncertain, and should be interpreted within the broader context of clinical findings, laboratory data, and established risk stratification tools,” he added.
The study had no specific funding. Lee and Ayazi had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM DDW 2025