User login
Become a Social Media Ambassador
If you are a medical student, a general surgery resident and/or a trainee who’d like to get more involved within the Society for Vascular Surgery, apply to be an SVS Social Media Ambassador. Ambassadors must be members of the SVS and understand the basic concept of social media. Once a part of the program, they are encouraged to be advocates for the SVS and provide honest feedback to the staff as to what they’d like to see on our channels. If you are interested, learn more about the program here and apply today.
If you are a medical student, a general surgery resident and/or a trainee who’d like to get more involved within the Society for Vascular Surgery, apply to be an SVS Social Media Ambassador. Ambassadors must be members of the SVS and understand the basic concept of social media. Once a part of the program, they are encouraged to be advocates for the SVS and provide honest feedback to the staff as to what they’d like to see on our channels. If you are interested, learn more about the program here and apply today.
If you are a medical student, a general surgery resident and/or a trainee who’d like to get more involved within the Society for Vascular Surgery, apply to be an SVS Social Media Ambassador. Ambassadors must be members of the SVS and understand the basic concept of social media. Once a part of the program, they are encouraged to be advocates for the SVS and provide honest feedback to the staff as to what they’d like to see on our channels. If you are interested, learn more about the program here and apply today.
Interested in joining an SVS Task Force?
The Society for Vascular Surgery is establishing a new Health Information Technology Task Force. The SVS Executive Board is seeking members with a desire to explore and advance technology-based health improvement for vascular patients. Volunteers should be interested in, as well as have some experience with, technology-related health improvements, medical informatics, etc. Selected task force members will collaborate with non-member industry technology experts, as well as SVS leadership. If you’d like to be considered, please email a brief statement of interest to [email protected]. Read all the details here.
The Society for Vascular Surgery is establishing a new Health Information Technology Task Force. The SVS Executive Board is seeking members with a desire to explore and advance technology-based health improvement for vascular patients. Volunteers should be interested in, as well as have some experience with, technology-related health improvements, medical informatics, etc. Selected task force members will collaborate with non-member industry technology experts, as well as SVS leadership. If you’d like to be considered, please email a brief statement of interest to [email protected]. Read all the details here.
The Society for Vascular Surgery is establishing a new Health Information Technology Task Force. The SVS Executive Board is seeking members with a desire to explore and advance technology-based health improvement for vascular patients. Volunteers should be interested in, as well as have some experience with, technology-related health improvements, medical informatics, etc. Selected task force members will collaborate with non-member industry technology experts, as well as SVS leadership. If you’d like to be considered, please email a brief statement of interest to [email protected]. Read all the details here.
Dr. Younossi presents at congressional briefing on International NASH Day
AGA member Zobair Younossi, MD, MPH, and three other expert and patient panelists called upon Congress to help educate policymakers and the public on the increasing burden of NASH and to take steps to prevent its increase, including more funding for research.
Dr. Younossi provided congressional staffers with an informative presentation on the prevalence, adverse clinical outcomes, and economic outcomes of nonalcoholic steatohepatitis (NASH). He noted that 6.65 million adults have NASH – 688,000 have advanced NASH. Treatment of these patients carries an economic burden of $222.6 billion in direct costs and $95.4 billion in lifetime direct costs of advanced NASH.
Although NASH was discovered almost 40 years ago, the prevalence has doubled in the last 15 years and challenges to optimize disease diagnosis and management remain. The briefing was sponsored by the Global Liver Institute and its founder and CEO, Donna Cryer, JD, is a NASH patient and liver transplant recipient. Dr. Younossi noted that, between 2015 and 2030, the percentage of nonalcoholic fatty liver disease (NAFLD) that is related to NASH is projected to increase from 20% to 27%.
AGA thanks Dr. Younossi, the other panelists, and the Global Liver Institute for assembling this important educational forum and encouraging Congress to take action.
AGA member Zobair Younossi, MD, MPH, and three other expert and patient panelists called upon Congress to help educate policymakers and the public on the increasing burden of NASH and to take steps to prevent its increase, including more funding for research.
Dr. Younossi provided congressional staffers with an informative presentation on the prevalence, adverse clinical outcomes, and economic outcomes of nonalcoholic steatohepatitis (NASH). He noted that 6.65 million adults have NASH – 688,000 have advanced NASH. Treatment of these patients carries an economic burden of $222.6 billion in direct costs and $95.4 billion in lifetime direct costs of advanced NASH.
Although NASH was discovered almost 40 years ago, the prevalence has doubled in the last 15 years and challenges to optimize disease diagnosis and management remain. The briefing was sponsored by the Global Liver Institute and its founder and CEO, Donna Cryer, JD, is a NASH patient and liver transplant recipient. Dr. Younossi noted that, between 2015 and 2030, the percentage of nonalcoholic fatty liver disease (NAFLD) that is related to NASH is projected to increase from 20% to 27%.
AGA thanks Dr. Younossi, the other panelists, and the Global Liver Institute for assembling this important educational forum and encouraging Congress to take action.
AGA member Zobair Younossi, MD, MPH, and three other expert and patient panelists called upon Congress to help educate policymakers and the public on the increasing burden of NASH and to take steps to prevent its increase, including more funding for research.
Dr. Younossi provided congressional staffers with an informative presentation on the prevalence, adverse clinical outcomes, and economic outcomes of nonalcoholic steatohepatitis (NASH). He noted that 6.65 million adults have NASH – 688,000 have advanced NASH. Treatment of these patients carries an economic burden of $222.6 billion in direct costs and $95.4 billion in lifetime direct costs of advanced NASH.
Although NASH was discovered almost 40 years ago, the prevalence has doubled in the last 15 years and challenges to optimize disease diagnosis and management remain. The briefing was sponsored by the Global Liver Institute and its founder and CEO, Donna Cryer, JD, is a NASH patient and liver transplant recipient. Dr. Younossi noted that, between 2015 and 2030, the percentage of nonalcoholic fatty liver disease (NAFLD) that is related to NASH is projected to increase from 20% to 27%.
AGA thanks Dr. Younossi, the other panelists, and the Global Liver Institute for assembling this important educational forum and encouraging Congress to take action.
AGA journals select new editorial fellows
The AGA journals – Gastroenterology, Clinical Gastroenterology and Hepatology (CGH), and Cellular and Molecular Gastroenterology and Hepatology (CMGH) – are pleased to announce their 2019-2020 editorial fellows.
Gastroenterology
Feng Su, MD
University of Washington, Seattle
@FengSu_MD
Victoria Weis, PhD
Wake Forest School of Medicine, Winston-Salem, N.C.
CGH
Austin Chiang, MD, MPH
Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia
@AustinChiangMD
Jennifer Kolb, MD
University of Colorado Anschutz Medical Campus, Aurora
CMGH
Cambrian Liu, PhD
The Saban Research Institute, Children’s Hospital Los Angeles
Tirthadipa Pradhan-Sundd, PhD
University of Pittsburgh, Pennsylvania
@Tirthadipa
The editorial fellows will be mentored on their respective journals’ editorial processes, including peer review and the publication process from manuscript submission to acceptance. They will participate in discussions and conferences with the boards of editors and work closely with the AGA editorial staff. Additionally, the fellows will participate in AGA’s new reviewer education program and will also be offered the opportunity to contribute content to their respective journals. The newly expanded program builds on the success of the previous 2 years when Gastroenterology had an editorial fellow.
The journals’ board of editors and editorial staff congratulate the fellows and are excited to work with them over the next year.
The AGA journals – Gastroenterology, Clinical Gastroenterology and Hepatology (CGH), and Cellular and Molecular Gastroenterology and Hepatology (CMGH) – are pleased to announce their 2019-2020 editorial fellows.
Gastroenterology
Feng Su, MD
University of Washington, Seattle
@FengSu_MD
Victoria Weis, PhD
Wake Forest School of Medicine, Winston-Salem, N.C.
CGH
Austin Chiang, MD, MPH
Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia
@AustinChiangMD
Jennifer Kolb, MD
University of Colorado Anschutz Medical Campus, Aurora
CMGH
Cambrian Liu, PhD
The Saban Research Institute, Children’s Hospital Los Angeles
Tirthadipa Pradhan-Sundd, PhD
University of Pittsburgh, Pennsylvania
@Tirthadipa
The editorial fellows will be mentored on their respective journals’ editorial processes, including peer review and the publication process from manuscript submission to acceptance. They will participate in discussions and conferences with the boards of editors and work closely with the AGA editorial staff. Additionally, the fellows will participate in AGA’s new reviewer education program and will also be offered the opportunity to contribute content to their respective journals. The newly expanded program builds on the success of the previous 2 years when Gastroenterology had an editorial fellow.
The journals’ board of editors and editorial staff congratulate the fellows and are excited to work with them over the next year.
The AGA journals – Gastroenterology, Clinical Gastroenterology and Hepatology (CGH), and Cellular and Molecular Gastroenterology and Hepatology (CMGH) – are pleased to announce their 2019-2020 editorial fellows.
Gastroenterology
Feng Su, MD
University of Washington, Seattle
@FengSu_MD
Victoria Weis, PhD
Wake Forest School of Medicine, Winston-Salem, N.C.
CGH
Austin Chiang, MD, MPH
Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia
@AustinChiangMD
Jennifer Kolb, MD
University of Colorado Anschutz Medical Campus, Aurora
CMGH
Cambrian Liu, PhD
The Saban Research Institute, Children’s Hospital Los Angeles
Tirthadipa Pradhan-Sundd, PhD
University of Pittsburgh, Pennsylvania
@Tirthadipa
The editorial fellows will be mentored on their respective journals’ editorial processes, including peer review and the publication process from manuscript submission to acceptance. They will participate in discussions and conferences with the boards of editors and work closely with the AGA editorial staff. Additionally, the fellows will participate in AGA’s new reviewer education program and will also be offered the opportunity to contribute content to their respective journals. The newly expanded program builds on the success of the previous 2 years when Gastroenterology had an editorial fellow.
The journals’ board of editors and editorial staff congratulate the fellows and are excited to work with them over the next year.
Inside AGA’s meeting with FDA on fecal microbiota transplantation
AGA’s microbiome leaders recently met with representatives from FDA’s Center for Biologics Evaluation and Research (CBER) to share clinician and researcher perspectives on fecal microbiota transplantation (FMT) and understand CBER’s current thinking on the regulation of FMT for the treatment of Clostridioides difficile (C. difficile) infection. Here are the key takeaways from AGA’s discussion with CBER.
AGA made clear to FDA the needs and concerns of the clinical and research communities regarding FMT. AGA communicated clinician concerns about patient access to whole-stool FMT being restricted or perhaps eliminated once drugs containing live microbials are FDA approved. AGA’s representatives also shared concerns about the narrow inclusion criteria for current clinical trials and whether the new drugs will be as effective as whole-stool FMT for vulnerable populations such as the elderly or immunocompromised, who make up the majority of patients with C. difficile infection but are often excluded from current trials. Finally, AGA emphasized the need to encourage innovation in product development and the importance of performing controlled safety and efficacy studies on products that can be manufactured predictably and reproducibly.
All stakeholders agreed that the AGA FMT National Registry is an important effort to collect short- and long-term data on the safety and efficacy of FMT. AGA will maintain dialogue with CBER regarding data from the registry and lessons learned. Clinicians practicing FMT are strongly encouraged to participate in the FMT National Registry, which will follow short- and long-term outcomes of patients receiving FMT for up to 10 years. The registry is funded by a grant from the National Institute of Allergy and Infectious Diseases at the National Institutes of Health (award number R24 AI118629) and is a partnership of AGA, the Crohn’s &nd Colitis Foundation, the Infectious Diseases Society of America and North American Society for Pediatric Gastroenterology, Hepatology and Nutrition.
CBER is currently working on an update to the enforcement discretion policy on the use of FMT for C. difficile infection not responsive to standard therapies.Agency representatives noted that all comments will be considered as the agency finalizes the guidance. The current enforcement discretion policy has been in place since July 2013 and was most recently updated by CBER in a draft guidance in March 2016. The policy enables clinicians to use FMT for the treatment of C. difficile infection not responsive to standard therapies without having an investigational new drug (IND) application in place.
Human stool will continue to be regulated as a drug and biological product. The agency stated that human stool does not meet the definition of a tissue and FDA does not intend to change how it is currently classified.
CBER is interested in hearing ideas for novel trial designs that may help address the challenges of patient recruitment for clinical trials in C. difficile infection and other indications for FMT. AGA encourages members to share their thoughts on this topic through the AGA Community.
Following AGA’s meeting with CBER, FDA issued a safety alert because of the death of a patient who died from an FMT containing a multi-drug resistant organism. The agency has since issued additional requirements for IND holders on stool donor screening. AGA will continue to engage with FDA on this issue and share updates as they become available with all members.
Meeting participants from AGA membership included:
• Gail A. Hecht, MD, MS, AGAF, immediate past chair, AGA Center for Gut Microbiome Research and Education Scientific Advisory Board
• Colleen R. Kelly, MD, co-chair, AGA FMT National Registry Steering Committee
• Alexander Khoruts, MD, member, AGA Center for Gut Microbiome Research and Education Scientific Advisory Board
• Gary D. Wu, MD, AGAF, basic research councilor, AGA Institute Governing Board, and member, AGA FMT National Registry Steering Committee
Meeting participants from FDA/CBER included:
• Peter Marks, MD, PhD, Director, CBER
• Celia Witten, PhD, MD, Deputy Director, CBER
• Diane Maloney, JD, Associate Director for Policy, CBER
• Julie Tierney, JD, Senior Policy Advisor for Strategic Planning & Legislation, CBER
• Marion Gruber, PhD, Director, Office of Vaccines Research and Review (OVRR), CBER
• Theresa Finn, PhD, Associate Director for Policy, OVRR, CBER
• Doran Fink, MD, PhD, Deputy Director, Clinical, Division of Vaccines and Related Products Applications, OVRR, CBER
• Paul Carlson, PhD, Senior Staff Fellow, OVRR
• Lorrie McNeill, Director, Office of Communication, Outreach and Development, CBER
This meeting took place on May 6, 2019, at the FDA headquarters in Silver Spring, Md.
AGA’s microbiome leaders recently met with representatives from FDA’s Center for Biologics Evaluation and Research (CBER) to share clinician and researcher perspectives on fecal microbiota transplantation (FMT) and understand CBER’s current thinking on the regulation of FMT for the treatment of Clostridioides difficile (C. difficile) infection. Here are the key takeaways from AGA’s discussion with CBER.
AGA made clear to FDA the needs and concerns of the clinical and research communities regarding FMT. AGA communicated clinician concerns about patient access to whole-stool FMT being restricted or perhaps eliminated once drugs containing live microbials are FDA approved. AGA’s representatives also shared concerns about the narrow inclusion criteria for current clinical trials and whether the new drugs will be as effective as whole-stool FMT for vulnerable populations such as the elderly or immunocompromised, who make up the majority of patients with C. difficile infection but are often excluded from current trials. Finally, AGA emphasized the need to encourage innovation in product development and the importance of performing controlled safety and efficacy studies on products that can be manufactured predictably and reproducibly.
All stakeholders agreed that the AGA FMT National Registry is an important effort to collect short- and long-term data on the safety and efficacy of FMT. AGA will maintain dialogue with CBER regarding data from the registry and lessons learned. Clinicians practicing FMT are strongly encouraged to participate in the FMT National Registry, which will follow short- and long-term outcomes of patients receiving FMT for up to 10 years. The registry is funded by a grant from the National Institute of Allergy and Infectious Diseases at the National Institutes of Health (award number R24 AI118629) and is a partnership of AGA, the Crohn’s &nd Colitis Foundation, the Infectious Diseases Society of America and North American Society for Pediatric Gastroenterology, Hepatology and Nutrition.
CBER is currently working on an update to the enforcement discretion policy on the use of FMT for C. difficile infection not responsive to standard therapies.Agency representatives noted that all comments will be considered as the agency finalizes the guidance. The current enforcement discretion policy has been in place since July 2013 and was most recently updated by CBER in a draft guidance in March 2016. The policy enables clinicians to use FMT for the treatment of C. difficile infection not responsive to standard therapies without having an investigational new drug (IND) application in place.
Human stool will continue to be regulated as a drug and biological product. The agency stated that human stool does not meet the definition of a tissue and FDA does not intend to change how it is currently classified.
CBER is interested in hearing ideas for novel trial designs that may help address the challenges of patient recruitment for clinical trials in C. difficile infection and other indications for FMT. AGA encourages members to share their thoughts on this topic through the AGA Community.
Following AGA’s meeting with CBER, FDA issued a safety alert because of the death of a patient who died from an FMT containing a multi-drug resistant organism. The agency has since issued additional requirements for IND holders on stool donor screening. AGA will continue to engage with FDA on this issue and share updates as they become available with all members.
Meeting participants from AGA membership included:
• Gail A. Hecht, MD, MS, AGAF, immediate past chair, AGA Center for Gut Microbiome Research and Education Scientific Advisory Board
• Colleen R. Kelly, MD, co-chair, AGA FMT National Registry Steering Committee
• Alexander Khoruts, MD, member, AGA Center for Gut Microbiome Research and Education Scientific Advisory Board
• Gary D. Wu, MD, AGAF, basic research councilor, AGA Institute Governing Board, and member, AGA FMT National Registry Steering Committee
Meeting participants from FDA/CBER included:
• Peter Marks, MD, PhD, Director, CBER
• Celia Witten, PhD, MD, Deputy Director, CBER
• Diane Maloney, JD, Associate Director for Policy, CBER
• Julie Tierney, JD, Senior Policy Advisor for Strategic Planning & Legislation, CBER
• Marion Gruber, PhD, Director, Office of Vaccines Research and Review (OVRR), CBER
• Theresa Finn, PhD, Associate Director for Policy, OVRR, CBER
• Doran Fink, MD, PhD, Deputy Director, Clinical, Division of Vaccines and Related Products Applications, OVRR, CBER
• Paul Carlson, PhD, Senior Staff Fellow, OVRR
• Lorrie McNeill, Director, Office of Communication, Outreach and Development, CBER
This meeting took place on May 6, 2019, at the FDA headquarters in Silver Spring, Md.
AGA’s microbiome leaders recently met with representatives from FDA’s Center for Biologics Evaluation and Research (CBER) to share clinician and researcher perspectives on fecal microbiota transplantation (FMT) and understand CBER’s current thinking on the regulation of FMT for the treatment of Clostridioides difficile (C. difficile) infection. Here are the key takeaways from AGA’s discussion with CBER.
AGA made clear to FDA the needs and concerns of the clinical and research communities regarding FMT. AGA communicated clinician concerns about patient access to whole-stool FMT being restricted or perhaps eliminated once drugs containing live microbials are FDA approved. AGA’s representatives also shared concerns about the narrow inclusion criteria for current clinical trials and whether the new drugs will be as effective as whole-stool FMT for vulnerable populations such as the elderly or immunocompromised, who make up the majority of patients with C. difficile infection but are often excluded from current trials. Finally, AGA emphasized the need to encourage innovation in product development and the importance of performing controlled safety and efficacy studies on products that can be manufactured predictably and reproducibly.
All stakeholders agreed that the AGA FMT National Registry is an important effort to collect short- and long-term data on the safety and efficacy of FMT. AGA will maintain dialogue with CBER regarding data from the registry and lessons learned. Clinicians practicing FMT are strongly encouraged to participate in the FMT National Registry, which will follow short- and long-term outcomes of patients receiving FMT for up to 10 years. The registry is funded by a grant from the National Institute of Allergy and Infectious Diseases at the National Institutes of Health (award number R24 AI118629) and is a partnership of AGA, the Crohn’s &nd Colitis Foundation, the Infectious Diseases Society of America and North American Society for Pediatric Gastroenterology, Hepatology and Nutrition.
CBER is currently working on an update to the enforcement discretion policy on the use of FMT for C. difficile infection not responsive to standard therapies.Agency representatives noted that all comments will be considered as the agency finalizes the guidance. The current enforcement discretion policy has been in place since July 2013 and was most recently updated by CBER in a draft guidance in March 2016. The policy enables clinicians to use FMT for the treatment of C. difficile infection not responsive to standard therapies without having an investigational new drug (IND) application in place.
Human stool will continue to be regulated as a drug and biological product. The agency stated that human stool does not meet the definition of a tissue and FDA does not intend to change how it is currently classified.
CBER is interested in hearing ideas for novel trial designs that may help address the challenges of patient recruitment for clinical trials in C. difficile infection and other indications for FMT. AGA encourages members to share their thoughts on this topic through the AGA Community.
Following AGA’s meeting with CBER, FDA issued a safety alert because of the death of a patient who died from an FMT containing a multi-drug resistant organism. The agency has since issued additional requirements for IND holders on stool donor screening. AGA will continue to engage with FDA on this issue and share updates as they become available with all members.
Meeting participants from AGA membership included:
• Gail A. Hecht, MD, MS, AGAF, immediate past chair, AGA Center for Gut Microbiome Research and Education Scientific Advisory Board
• Colleen R. Kelly, MD, co-chair, AGA FMT National Registry Steering Committee
• Alexander Khoruts, MD, member, AGA Center for Gut Microbiome Research and Education Scientific Advisory Board
• Gary D. Wu, MD, AGAF, basic research councilor, AGA Institute Governing Board, and member, AGA FMT National Registry Steering Committee
Meeting participants from FDA/CBER included:
• Peter Marks, MD, PhD, Director, CBER
• Celia Witten, PhD, MD, Deputy Director, CBER
• Diane Maloney, JD, Associate Director for Policy, CBER
• Julie Tierney, JD, Senior Policy Advisor for Strategic Planning & Legislation, CBER
• Marion Gruber, PhD, Director, Office of Vaccines Research and Review (OVRR), CBER
• Theresa Finn, PhD, Associate Director for Policy, OVRR, CBER
• Doran Fink, MD, PhD, Deputy Director, Clinical, Division of Vaccines and Related Products Applications, OVRR, CBER
• Paul Carlson, PhD, Senior Staff Fellow, OVRR
• Lorrie McNeill, Director, Office of Communication, Outreach and Development, CBER
This meeting took place on May 6, 2019, at the FDA headquarters in Silver Spring, Md.
Consider a Donation to the SVS PAC
The SVS Political Action Committee has set an aggressive fundraising goal for this current 2020 election cycle, and your contribution is critical to helping the PAC to meet this goal. The SVS PAC is one of the most effective tools for communicating with policymakers on Capitol Hill to make the greatest impact on legislative issues that affect your patients and your practices. Contributions allow the PAC Committee to establish strong relationships with members of Congress and cultivate congressional champions for our legislative and regulatory priorities. Make your donation or enhance your previously made donation to the SVS PAC here. Want to learn more? Click here.
The SVS Political Action Committee has set an aggressive fundraising goal for this current 2020 election cycle, and your contribution is critical to helping the PAC to meet this goal. The SVS PAC is one of the most effective tools for communicating with policymakers on Capitol Hill to make the greatest impact on legislative issues that affect your patients and your practices. Contributions allow the PAC Committee to establish strong relationships with members of Congress and cultivate congressional champions for our legislative and regulatory priorities. Make your donation or enhance your previously made donation to the SVS PAC here. Want to learn more? Click here.
The SVS Political Action Committee has set an aggressive fundraising goal for this current 2020 election cycle, and your contribution is critical to helping the PAC to meet this goal. The SVS PAC is one of the most effective tools for communicating with policymakers on Capitol Hill to make the greatest impact on legislative issues that affect your patients and your practices. Contributions allow the PAC Committee to establish strong relationships with members of Congress and cultivate congressional champions for our legislative and regulatory priorities. Make your donation or enhance your previously made donation to the SVS PAC here. Want to learn more? Click here.
Register for the UCLA / SVS Symposium
Register today for the fourth annual UCLA / SVS Symposium. This year’s event, “A Comprehensive Review and Update of What’s New in Vascular and Endovascular Surgery,” will be held Aug. 24 to 26 at the Beverly Hilton in Beverly Hills, Calif. An internationally recognized faculty will present a comprehensive survey of generally accepted views. These will include basic science, pathogenesis, diagnosis and management of the broad spectrum of vascular disorders. Additionally, all speakers will address important new developments related to their topic that have taken place within the last year. Learn more and register today.
Register today for the fourth annual UCLA / SVS Symposium. This year’s event, “A Comprehensive Review and Update of What’s New in Vascular and Endovascular Surgery,” will be held Aug. 24 to 26 at the Beverly Hilton in Beverly Hills, Calif. An internationally recognized faculty will present a comprehensive survey of generally accepted views. These will include basic science, pathogenesis, diagnosis and management of the broad spectrum of vascular disorders. Additionally, all speakers will address important new developments related to their topic that have taken place within the last year. Learn more and register today.
Register today for the fourth annual UCLA / SVS Symposium. This year’s event, “A Comprehensive Review and Update of What’s New in Vascular and Endovascular Surgery,” will be held Aug. 24 to 26 at the Beverly Hilton in Beverly Hills, Calif. An internationally recognized faculty will present a comprehensive survey of generally accepted views. These will include basic science, pathogenesis, diagnosis and management of the broad spectrum of vascular disorders. Additionally, all speakers will address important new developments related to their topic that have taken place within the last year. Learn more and register today.
CHEST Clinical Perspectives explores the emerging field of precision medicine
For clinicians seeking to provide a pathway to treatment or diagnosis that is individualized to the patient, a recent study found that the issues go beyond awareness or a patient’s degree of comfort – there remains the question of something as simple as: what should we call it?
Clinicians remain uncertain whether to name the new field precision or personalized medicine according the new CHEST Clinical PerspectivesTM white paper, “Precision Medicine: Adoption of Emerging Methods of Evaluation and Therapy.” A survey of leading community clinicians from among CHEST membership found that only 35 % called tailoring medical treatment to the individual characteristics of each patient “precision medicine,” with 24% preferring “personalized” medicine. Thirty-six percent of respondents used the terms interchangeably.
Beyond the communication issues, the study found that most clinicians surveyed did not know enough about precision medicine to adopt it into their practice. Those surveyed reported that they wanted to see more published studies on the effectiveness of the newly available tools before discussing these options with their patients.
The majority of the respondents were general pulmonologists with intensivists and interventional pulmonologists also responding. The study was led by Nichole T. Tanner, MD, MSCR, FCCP, of the Medical University of South Carolina. Dr Tanner will be hosting a webinar to review the conclusions of this paper at 10:00 AM CT on Tuesday, July 30.
More information about CHEST Clinical PerspectivesTM, part of the CHEST Analytics program, can be found at insights.chestnet.org. To suggest a topic to be covered in a future issue, contact Linda Tomczynski, [email protected] or +1 (224) 521-9593. Register today at https://hubs.ly/H0jqCGb0.
For clinicians seeking to provide a pathway to treatment or diagnosis that is individualized to the patient, a recent study found that the issues go beyond awareness or a patient’s degree of comfort – there remains the question of something as simple as: what should we call it?
Clinicians remain uncertain whether to name the new field precision or personalized medicine according the new CHEST Clinical PerspectivesTM white paper, “Precision Medicine: Adoption of Emerging Methods of Evaluation and Therapy.” A survey of leading community clinicians from among CHEST membership found that only 35 % called tailoring medical treatment to the individual characteristics of each patient “precision medicine,” with 24% preferring “personalized” medicine. Thirty-six percent of respondents used the terms interchangeably.
Beyond the communication issues, the study found that most clinicians surveyed did not know enough about precision medicine to adopt it into their practice. Those surveyed reported that they wanted to see more published studies on the effectiveness of the newly available tools before discussing these options with their patients.
The majority of the respondents were general pulmonologists with intensivists and interventional pulmonologists also responding. The study was led by Nichole T. Tanner, MD, MSCR, FCCP, of the Medical University of South Carolina. Dr Tanner will be hosting a webinar to review the conclusions of this paper at 10:00 AM CT on Tuesday, July 30.
More information about CHEST Clinical PerspectivesTM, part of the CHEST Analytics program, can be found at insights.chestnet.org. To suggest a topic to be covered in a future issue, contact Linda Tomczynski, [email protected] or +1 (224) 521-9593. Register today at https://hubs.ly/H0jqCGb0.
For clinicians seeking to provide a pathway to treatment or diagnosis that is individualized to the patient, a recent study found that the issues go beyond awareness or a patient’s degree of comfort – there remains the question of something as simple as: what should we call it?
Clinicians remain uncertain whether to name the new field precision or personalized medicine according the new CHEST Clinical PerspectivesTM white paper, “Precision Medicine: Adoption of Emerging Methods of Evaluation and Therapy.” A survey of leading community clinicians from among CHEST membership found that only 35 % called tailoring medical treatment to the individual characteristics of each patient “precision medicine,” with 24% preferring “personalized” medicine. Thirty-six percent of respondents used the terms interchangeably.
Beyond the communication issues, the study found that most clinicians surveyed did not know enough about precision medicine to adopt it into their practice. Those surveyed reported that they wanted to see more published studies on the effectiveness of the newly available tools before discussing these options with their patients.
The majority of the respondents were general pulmonologists with intensivists and interventional pulmonologists also responding. The study was led by Nichole T. Tanner, MD, MSCR, FCCP, of the Medical University of South Carolina. Dr Tanner will be hosting a webinar to review the conclusions of this paper at 10:00 AM CT on Tuesday, July 30.
More information about CHEST Clinical PerspectivesTM, part of the CHEST Analytics program, can be found at insights.chestnet.org. To suggest a topic to be covered in a future issue, contact Linda Tomczynski, [email protected] or +1 (224) 521-9593. Register today at https://hubs.ly/H0jqCGb0.
Environmental Scan: Drivers of change in health care
CHEST Inspiration is a collection of programmatic initiatives developed by the American College of Chest Physicians leadership and aimed at stimulating and encouraging innovation within the association. One of the components of CHEST Inspiration is the Environmental Scan, a series of articles focusing on the internal and external environmental factors that bear on success currently and in the future. See “Envisioning the Future: the CHEST Environmental Scan,” CHEST Physician, June 2019, p. 44, for an introduction to the series.
Chest physicians are witnessing a revolution within the environment in which they practice. Information technology, changing consumer behavior, and the social imperative to contain costs are coming together to transform health care.
Innovation in the prevention, diagnosis, and treatment of health-related issues is being fueled by the emergence of accessible and affordable technology-based solutions and changes in patient approaches to health care. Consumers and employers are increasingly motivated to look for cost-effective options for health in care delivery and for economical access to innovations.1 Organizations will need to respond with a strategy that aligns with the changing environment and position physicians to lead these trends in the direction of improved patient care.2
Enabling technologies like electronic health records, blockchain, and artificial intelligence will increase connectivity among all the stakeholders in the health-care system. The exponential increase in connectivity means growing engagement of health systems, health plans, patients, and families in all aspects of health care. For health-care providers, these technologies will mean an acceleration of the requirement to generate data in clinical settings and utilize data for clinical decision making. Easily available data on outcomes and, most importantly, cost of treatment will be expected at point of service.3
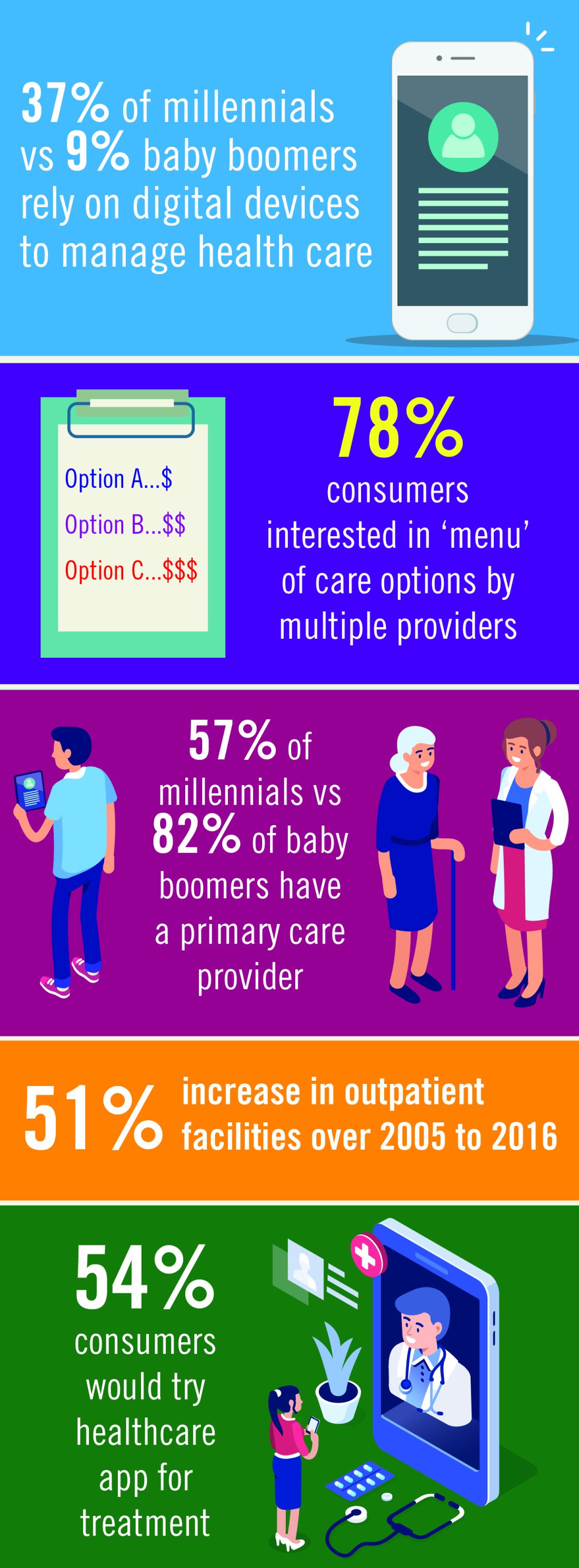
Access to information will continue to empower consumers to take an active role in their own health care. More patients will be comfortable with delivery of some health care via digital devices, apps, and virtual access to treatment. The market will respond with technology that helps consumers navigate health-care systems, explore options, and communicate directly with providers. The use of apps and virtual encounters is expected to transform the role of primary care providers: patients will increasingly utilize nonphysician resources in outpatient settings, bypassing primary care physicians and reaching out to specialty care as needed.4
David A. Schulman, MD, FCCP, Professor of Medicine, Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, Emory University School of Medicine, Atlanta, and Editor in Chief of CHEST Physician, has seen the transformation of patient behavior and attitudes in his own practice.
“In general, they have done far more research about their health problems before seeking my counsel than patients did previously. Many use the internet not just to read about their symptoms and diseases, but also to connect with others having similar issues, sharing experiences, treatments, outcomes, and emotions; in some ways, this is the new ‘crowdsourcing’ of medicine.”
Patients who do their own “research” can present a challenge for physicians. Dr. Schulman noted, “I am often surprised about the misconceptions about disease that derive from information gleaned from a web-based source. One need not look any farther than the groundswell of misinformation being spread about vaccinations to see the potential downside of the pervasive availability of medical ‘facts’ online. Since we are unlikely to convince our patients to avoid the online milieu entirely, our role as health-care providers is to help our patients process and appropriately weigh the information that they receive, potentially partnering with our national societies to help curate such information.”
Dr. Schulman’s approach to the potential of patient misinformation is to initiate almost all discussions with patients with the question “Have your read or seen anything about this condition?” He said, “It is rare for patients to answer negatively. And listening to them speak about their understanding of their disease provides me with invaluable information about how the remainder of our visit should be spent. Do we need to correct misunderstandings? Are there gaps in the explanation that I can fill? Can we move directly into a conversation about treatment options? Can I provide you with some additional resources that might help to further your knowledge about the condition?”
Generational factors will play a big role in health-care demand and delivery. Health-care companies are already building lower cost delivery models to capture the millennial market.4 Cost-saving digital tools and virtual contacts are currently most commonly used by younger patients.5 Physicians need to understand and be a part of this trend, Dr. Schulman argued. “We should embrace telemedicine and mobile applications to collect data from the patients in their day-to-day lives. While insurance coverage of telemedicine is far from universal at the moment, and the reliability of mobile applications is highly variable, we know that a growing number of our patients are already relying on their digital devices to manage their health. In much the same way that we will need to help patient evaluate online information, we should work with our national societies to support the creation of tools that will allow us to collect data in the home environment in a more robust and reliable fashion.”
The proportion of the US population over the age of 65 is increasing yearly.6 Six out of 10 Americans live with a chronic illness, such as heart disease or diabetes. These and other chronic diseases are the leading drivers of the $3.3 billion annual health-care costs.7 Cost containment for these older patients and those with chronic illness will involve a focus on quality and outcomes data, a drive to deliver treatment in lower cost outpatient settings, and an acceleration of the adoption of value-based models currently underway.8
Taken together, these trends will mean a growing digital interface between physician and patient, a more active consumer-patient, and the availability of a vast array of new tools to access and manage health-care data.
- Delivery of procedures and services will trend from physicians to other members of the health-care team and to lower cost, outpatient settings.9
- Health-care systems will ramp up investment in products and services that improve outcomes and cost effectiveness.10
- Increased regulatory requirements and new payment models mean an ever-growing utilization of information technology by providers to fulfill data imperatives.11
- Physicians will have an increased need for tools that prioritize costs and outcomes data at the point of care.12
- Integration of data from new technologies will touch every aspect of health-care delivery with the objective of improving outcomes and, in turn, reducing costs.13
- Changing consumer attitudes toward delivery of care will be based on a growing familiarity of patients with a digital or virtual interface with providers, facility with health-care apps, and preference for a menu of options for health-care delivery.14
Dr. Schulman concluded, “We can no more expect our patients to ignore the full panoply of medical information on the internet and digital tools on their mobile devices than we can tell the tide not to come in. The die is cast; this is the world within which we must ply our trade. By identifying best practices and sharing our successes, we can come through this revolution better for the experience.”
References
1. https://www.modernhealthcare.com/article/20181220/NEWS/181229992/number-of-outpatient-facilities-surges-as-industry-values-more
2. https://www.accenture.com/us-en/insights/health/digital-health-tech-vision-2018
3. https://www.accenture.com/us-en/insights/health/digital-health-primary-care
4. PcW Health Research Institute: Top health industry issues of 2019
5. https://www.accenture.com/us-en/insights/health/digital-health-primary-care
6. https://www.census.gov/newsroom/press-releases/2017/cb17-100.html
7. https://www.cdc.gov/chronicdisease/index.htm
8. https://www2.deloitte.com/us/en/pages/life-sciences-and-health-care/articles/health-care-current-december4-2018.html
9. PcW Health Research Institute Top health industry issues of 2019: The New Health Economy comes of age
10. https://www.accenture.com/us-en/insights/health/digital-health-tech-vision-2018
11. https://www2.deloitte.com/insights/us/en/industry/life-sciences/medtech-research-and-development-innovation.html
12. https://www2.deloitte.com/insights/us/en/industry/health-care/volume-to-value-based-care.html
13. https://www2.deloitte.com/insights/us/en/industry/health-care/volume-to-value-based-care.html
14. https://www.accenture.com/us-en/insights/health/digital-health-primary-care
CHEST Inspiration is a collection of programmatic initiatives developed by the American College of Chest Physicians leadership and aimed at stimulating and encouraging innovation within the association. One of the components of CHEST Inspiration is the Environmental Scan, a series of articles focusing on the internal and external environmental factors that bear on success currently and in the future. See “Envisioning the Future: the CHEST Environmental Scan,” CHEST Physician, June 2019, p. 44, for an introduction to the series.
Chest physicians are witnessing a revolution within the environment in which they practice. Information technology, changing consumer behavior, and the social imperative to contain costs are coming together to transform health care.
Innovation in the prevention, diagnosis, and treatment of health-related issues is being fueled by the emergence of accessible and affordable technology-based solutions and changes in patient approaches to health care. Consumers and employers are increasingly motivated to look for cost-effective options for health in care delivery and for economical access to innovations.1 Organizations will need to respond with a strategy that aligns with the changing environment and position physicians to lead these trends in the direction of improved patient care.2
Enabling technologies like electronic health records, blockchain, and artificial intelligence will increase connectivity among all the stakeholders in the health-care system. The exponential increase in connectivity means growing engagement of health systems, health plans, patients, and families in all aspects of health care. For health-care providers, these technologies will mean an acceleration of the requirement to generate data in clinical settings and utilize data for clinical decision making. Easily available data on outcomes and, most importantly, cost of treatment will be expected at point of service.3
Access to information will continue to empower consumers to take an active role in their own health care. More patients will be comfortable with delivery of some health care via digital devices, apps, and virtual access to treatment. The market will respond with technology that helps consumers navigate health-care systems, explore options, and communicate directly with providers. The use of apps and virtual encounters is expected to transform the role of primary care providers: patients will increasingly utilize nonphysician resources in outpatient settings, bypassing primary care physicians and reaching out to specialty care as needed.4
David A. Schulman, MD, FCCP, Professor of Medicine, Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, Emory University School of Medicine, Atlanta, and Editor in Chief of CHEST Physician, has seen the transformation of patient behavior and attitudes in his own practice.
“In general, they have done far more research about their health problems before seeking my counsel than patients did previously. Many use the internet not just to read about their symptoms and diseases, but also to connect with others having similar issues, sharing experiences, treatments, outcomes, and emotions; in some ways, this is the new ‘crowdsourcing’ of medicine.”
Patients who do their own “research” can present a challenge for physicians. Dr. Schulman noted, “I am often surprised about the misconceptions about disease that derive from information gleaned from a web-based source. One need not look any farther than the groundswell of misinformation being spread about vaccinations to see the potential downside of the pervasive availability of medical ‘facts’ online. Since we are unlikely to convince our patients to avoid the online milieu entirely, our role as health-care providers is to help our patients process and appropriately weigh the information that they receive, potentially partnering with our national societies to help curate such information.”
Dr. Schulman’s approach to the potential of patient misinformation is to initiate almost all discussions with patients with the question “Have your read or seen anything about this condition?” He said, “It is rare for patients to answer negatively. And listening to them speak about their understanding of their disease provides me with invaluable information about how the remainder of our visit should be spent. Do we need to correct misunderstandings? Are there gaps in the explanation that I can fill? Can we move directly into a conversation about treatment options? Can I provide you with some additional resources that might help to further your knowledge about the condition?”
Generational factors will play a big role in health-care demand and delivery. Health-care companies are already building lower cost delivery models to capture the millennial market.4 Cost-saving digital tools and virtual contacts are currently most commonly used by younger patients.5 Physicians need to understand and be a part of this trend, Dr. Schulman argued. “We should embrace telemedicine and mobile applications to collect data from the patients in their day-to-day lives. While insurance coverage of telemedicine is far from universal at the moment, and the reliability of mobile applications is highly variable, we know that a growing number of our patients are already relying on their digital devices to manage their health. In much the same way that we will need to help patient evaluate online information, we should work with our national societies to support the creation of tools that will allow us to collect data in the home environment in a more robust and reliable fashion.”
The proportion of the US population over the age of 65 is increasing yearly.6 Six out of 10 Americans live with a chronic illness, such as heart disease or diabetes. These and other chronic diseases are the leading drivers of the $3.3 billion annual health-care costs.7 Cost containment for these older patients and those with chronic illness will involve a focus on quality and outcomes data, a drive to deliver treatment in lower cost outpatient settings, and an acceleration of the adoption of value-based models currently underway.8
Taken together, these trends will mean a growing digital interface between physician and patient, a more active consumer-patient, and the availability of a vast array of new tools to access and manage health-care data.
- Delivery of procedures and services will trend from physicians to other members of the health-care team and to lower cost, outpatient settings.9
- Health-care systems will ramp up investment in products and services that improve outcomes and cost effectiveness.10
- Increased regulatory requirements and new payment models mean an ever-growing utilization of information technology by providers to fulfill data imperatives.11
- Physicians will have an increased need for tools that prioritize costs and outcomes data at the point of care.12
- Integration of data from new technologies will touch every aspect of health-care delivery with the objective of improving outcomes and, in turn, reducing costs.13
- Changing consumer attitudes toward delivery of care will be based on a growing familiarity of patients with a digital or virtual interface with providers, facility with health-care apps, and preference for a menu of options for health-care delivery.14
Dr. Schulman concluded, “We can no more expect our patients to ignore the full panoply of medical information on the internet and digital tools on their mobile devices than we can tell the tide not to come in. The die is cast; this is the world within which we must ply our trade. By identifying best practices and sharing our successes, we can come through this revolution better for the experience.”

References
1. https://www.modernhealthcare.com/article/20181220/NEWS/181229992/number-of-outpatient-facilities-surges-as-industry-values-more
2. https://www.accenture.com/us-en/insights/health/digital-health-tech-vision-2018
3. https://www.accenture.com/us-en/insights/health/digital-health-primary-care
4. PcW Health Research Institute: Top health industry issues of 2019
5. https://www.accenture.com/us-en/insights/health/digital-health-primary-care
6. https://www.census.gov/newsroom/press-releases/2017/cb17-100.html
7. https://www.cdc.gov/chronicdisease/index.htm
8. https://www2.deloitte.com/us/en/pages/life-sciences-and-health-care/articles/health-care-current-december4-2018.html
9. PcW Health Research Institute Top health industry issues of 2019: The New Health Economy comes of age
10. https://www.accenture.com/us-en/insights/health/digital-health-tech-vision-2018
11. https://www2.deloitte.com/insights/us/en/industry/life-sciences/medtech-research-and-development-innovation.html
12. https://www2.deloitte.com/insights/us/en/industry/health-care/volume-to-value-based-care.html
13. https://www2.deloitte.com/insights/us/en/industry/health-care/volume-to-value-based-care.html
14. https://www.accenture.com/us-en/insights/health/digital-health-primary-care
CHEST Inspiration is a collection of programmatic initiatives developed by the American College of Chest Physicians leadership and aimed at stimulating and encouraging innovation within the association. One of the components of CHEST Inspiration is the Environmental Scan, a series of articles focusing on the internal and external environmental factors that bear on success currently and in the future. See “Envisioning the Future: the CHEST Environmental Scan,” CHEST Physician, June 2019, p. 44, for an introduction to the series.
Chest physicians are witnessing a revolution within the environment in which they practice. Information technology, changing consumer behavior, and the social imperative to contain costs are coming together to transform health care.
Innovation in the prevention, diagnosis, and treatment of health-related issues is being fueled by the emergence of accessible and affordable technology-based solutions and changes in patient approaches to health care. Consumers and employers are increasingly motivated to look for cost-effective options for health in care delivery and for economical access to innovations.1 Organizations will need to respond with a strategy that aligns with the changing environment and position physicians to lead these trends in the direction of improved patient care.2
Enabling technologies like electronic health records, blockchain, and artificial intelligence will increase connectivity among all the stakeholders in the health-care system. The exponential increase in connectivity means growing engagement of health systems, health plans, patients, and families in all aspects of health care. For health-care providers, these technologies will mean an acceleration of the requirement to generate data in clinical settings and utilize data for clinical decision making. Easily available data on outcomes and, most importantly, cost of treatment will be expected at point of service.3
Access to information will continue to empower consumers to take an active role in their own health care. More patients will be comfortable with delivery of some health care via digital devices, apps, and virtual access to treatment. The market will respond with technology that helps consumers navigate health-care systems, explore options, and communicate directly with providers. The use of apps and virtual encounters is expected to transform the role of primary care providers: patients will increasingly utilize nonphysician resources in outpatient settings, bypassing primary care physicians and reaching out to specialty care as needed.4
David A. Schulman, MD, FCCP, Professor of Medicine, Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, Emory University School of Medicine, Atlanta, and Editor in Chief of CHEST Physician, has seen the transformation of patient behavior and attitudes in his own practice.
“In general, they have done far more research about their health problems before seeking my counsel than patients did previously. Many use the internet not just to read about their symptoms and diseases, but also to connect with others having similar issues, sharing experiences, treatments, outcomes, and emotions; in some ways, this is the new ‘crowdsourcing’ of medicine.”
Patients who do their own “research” can present a challenge for physicians. Dr. Schulman noted, “I am often surprised about the misconceptions about disease that derive from information gleaned from a web-based source. One need not look any farther than the groundswell of misinformation being spread about vaccinations to see the potential downside of the pervasive availability of medical ‘facts’ online. Since we are unlikely to convince our patients to avoid the online milieu entirely, our role as health-care providers is to help our patients process and appropriately weigh the information that they receive, potentially partnering with our national societies to help curate such information.”
Dr. Schulman’s approach to the potential of patient misinformation is to initiate almost all discussions with patients with the question “Have your read or seen anything about this condition?” He said, “It is rare for patients to answer negatively. And listening to them speak about their understanding of their disease provides me with invaluable information about how the remainder of our visit should be spent. Do we need to correct misunderstandings? Are there gaps in the explanation that I can fill? Can we move directly into a conversation about treatment options? Can I provide you with some additional resources that might help to further your knowledge about the condition?”
Generational factors will play a big role in health-care demand and delivery. Health-care companies are already building lower cost delivery models to capture the millennial market.4 Cost-saving digital tools and virtual contacts are currently most commonly used by younger patients.5 Physicians need to understand and be a part of this trend, Dr. Schulman argued. “We should embrace telemedicine and mobile applications to collect data from the patients in their day-to-day lives. While insurance coverage of telemedicine is far from universal at the moment, and the reliability of mobile applications is highly variable, we know that a growing number of our patients are already relying on their digital devices to manage their health. In much the same way that we will need to help patient evaluate online information, we should work with our national societies to support the creation of tools that will allow us to collect data in the home environment in a more robust and reliable fashion.”
The proportion of the US population over the age of 65 is increasing yearly.6 Six out of 10 Americans live with a chronic illness, such as heart disease or diabetes. These and other chronic diseases are the leading drivers of the $3.3 billion annual health-care costs.7 Cost containment for these older patients and those with chronic illness will involve a focus on quality and outcomes data, a drive to deliver treatment in lower cost outpatient settings, and an acceleration of the adoption of value-based models currently underway.8
Taken together, these trends will mean a growing digital interface between physician and patient, a more active consumer-patient, and the availability of a vast array of new tools to access and manage health-care data.
- Delivery of procedures and services will trend from physicians to other members of the health-care team and to lower cost, outpatient settings.9
- Health-care systems will ramp up investment in products and services that improve outcomes and cost effectiveness.10
- Increased regulatory requirements and new payment models mean an ever-growing utilization of information technology by providers to fulfill data imperatives.11
- Physicians will have an increased need for tools that prioritize costs and outcomes data at the point of care.12
- Integration of data from new technologies will touch every aspect of health-care delivery with the objective of improving outcomes and, in turn, reducing costs.13
- Changing consumer attitudes toward delivery of care will be based on a growing familiarity of patients with a digital or virtual interface with providers, facility with health-care apps, and preference for a menu of options for health-care delivery.14
Dr. Schulman concluded, “We can no more expect our patients to ignore the full panoply of medical information on the internet and digital tools on their mobile devices than we can tell the tide not to come in. The die is cast; this is the world within which we must ply our trade. By identifying best practices and sharing our successes, we can come through this revolution better for the experience.”

References
1. https://www.modernhealthcare.com/article/20181220/NEWS/181229992/number-of-outpatient-facilities-surges-as-industry-values-more
2. https://www.accenture.com/us-en/insights/health/digital-health-tech-vision-2018
3. https://www.accenture.com/us-en/insights/health/digital-health-primary-care
4. PcW Health Research Institute: Top health industry issues of 2019
5. https://www.accenture.com/us-en/insights/health/digital-health-primary-care
6. https://www.census.gov/newsroom/press-releases/2017/cb17-100.html
7. https://www.cdc.gov/chronicdisease/index.htm
8. https://www2.deloitte.com/us/en/pages/life-sciences-and-health-care/articles/health-care-current-december4-2018.html
9. PcW Health Research Institute Top health industry issues of 2019: The New Health Economy comes of age
10. https://www.accenture.com/us-en/insights/health/digital-health-tech-vision-2018
11. https://www2.deloitte.com/insights/us/en/industry/life-sciences/medtech-research-and-development-innovation.html
12. https://www2.deloitte.com/insights/us/en/industry/health-care/volume-to-value-based-care.html
13. https://www2.deloitte.com/insights/us/en/industry/health-care/volume-to-value-based-care.html
14. https://www.accenture.com/us-en/insights/health/digital-health-primary-care
This month in the journal CHEST®
Editor’s picks
EDITORIAL
The CHEST Editorial Team: Serving Our Contributors and Readers
By Dr. P. J. Mazzone
ORIGINAL RESEARCH
Pulmonary Arterial Histologic Lesions in Patients With COPD With Severe Pulmonary Hypertension
By Dr. V. Bunel, et al.
Pulmonary Edema Following Initiation of Parenteral Prostacyclin Therapy for Pulmonary Arterial Hypertension: A Retrospective Study
By Dr. N. A. Khan, et al.
Lung Allocation Score Thresholds Prioritize Survival After Lung Transplantation
By Dr. S. S. Li, et al.
EVIDENCE-BASED MEDICINE
Chronic Cough and Gastroesophageal Reflux in Children: CHEST Guideline and Expert Panel Report
By Dr. A. B. Chang, et al.
Editor’s picks
Editor’s picks
EDITORIAL
The CHEST Editorial Team: Serving Our Contributors and Readers
By Dr. P. J. Mazzone
ORIGINAL RESEARCH
Pulmonary Arterial Histologic Lesions in Patients With COPD With Severe Pulmonary Hypertension
By Dr. V. Bunel, et al.
Pulmonary Edema Following Initiation of Parenteral Prostacyclin Therapy for Pulmonary Arterial Hypertension: A Retrospective Study
By Dr. N. A. Khan, et al.
Lung Allocation Score Thresholds Prioritize Survival After Lung Transplantation
By Dr. S. S. Li, et al.
EVIDENCE-BASED MEDICINE
Chronic Cough and Gastroesophageal Reflux in Children: CHEST Guideline and Expert Panel Report
By Dr. A. B. Chang, et al.
EDITORIAL
The CHEST Editorial Team: Serving Our Contributors and Readers
By Dr. P. J. Mazzone
ORIGINAL RESEARCH
Pulmonary Arterial Histologic Lesions in Patients With COPD With Severe Pulmonary Hypertension
By Dr. V. Bunel, et al.
Pulmonary Edema Following Initiation of Parenteral Prostacyclin Therapy for Pulmonary Arterial Hypertension: A Retrospective Study
By Dr. N. A. Khan, et al.
Lung Allocation Score Thresholds Prioritize Survival After Lung Transplantation
By Dr. S. S. Li, et al.
EVIDENCE-BASED MEDICINE
Chronic Cough and Gastroesophageal Reflux in Children: CHEST Guideline and Expert Panel Report
By Dr. A. B. Chang, et al.