User login
Are psychiatric disorders a ‘canary in a coal mine’ for Alzheimer’s disease?
, according to findings from a review of 1,500 patients with Alzheimer’s disease from a single-center population.
“Could psychosis symptoms be the proverbial canary in a coal mine?” Emily Eijansantos, a medical student at the University of California, San Francisco, said in reporting results of the chart review at the 2021 annual meeting of the American Academy of Neurology. “Previously in this cohort it was found that neurodevelopmental factors as well as chronic insults such as autoimmunity and seizure were also associated with an early age of onset in Alzheimer’s disease.”
The link between depression and autoimmunity, and anxiety and seizure “beg more questions about underlying pathophysiology,” she said. The study included 750 patients with early-onset Alzheimer’s disease and a similar number of late-onset patients from the UCSF Memory and Aging Center.
An inverse correlation between psychiatric disorders and age of Alzheimer’s onset
In the total study population, 43.5% (n = 652) had a previous diagnosis of depression and 32.3% (n = 485) had been diagnosed with anxiety. That, Ms. Eijansantos said, falls into similar ranges that other studies have reported.
“When we look at individual psychiatric disorders, we find that those with depression, anxiety, or PTSD are younger on average,” she said. “Patients with depression and anxiety are more [likely] female and have less vascular risk factors, and we observed an association between depression and autoimmunity, anxiety, and seizures.”
Specifically, patients with a history of depression were 2.2 years younger, on average, at the age of onset than patients without such history (P = .01); those with anxiety were 3 years younger on average (P = .01); and those with PTSD were 6.8 years younger on average, although only 1% (n = 15) of study subjects had PTSD, making for a small sample to study. These age-of-onset disparities didn’t appear among patients with previously diagnosed bipolar disorder (BPD) or schizophrenia.
Ms. Eijansantos noted that there were no differences in education attained or apolipoprotein-E gene status between the patients with and without a history of psychosis, and, within the subgroups of individual psychiatric disorders, there were no differences between patients with past and current or formal and informal diagnoses.
“When we split the cohort into quintiles based on age of Alzheimer’s disease onset, we find an inverse correlation between the amount of depression, anxiety, and PTSD endorsed and their ages of onset,” Ms. Eijansantos said. For example, the youngest quintile had a greater than 50% rate of depression while the oldest quintile had a depression rate around 36%. A similar spread was found with anxiety: a rate around 46% in the youngest quantile versus around 25% in the oldest, whereas rates of PTSD, BPD, and schizophrenia were similar across the five age-of-onset groups.
Patients with a history of multiple psychiatric disorders had an even younger age of onset. “We see that those with two psychiatric disorder are younger than those with one, and those with three psychiatric disorders are younger still,” Ms. Eijansantos said. “And we find that the Alzheimer’s disease age-at-onset reduction doubles with each additional psychiatric disorder.” Multiple disorders also adversely impacted survival, she said.
Because they found no difference between patients with past versus active symptoms and informal versus formal diagnosis, Ms. Eijansantos explained that they further studied the National Alzheimer’s Coordinating Center cohort of 8,267 patients with Alzheimer’s disease and found a similar relationship between psychiatric history and age of onset among patients with depression or anxiety or both. This cohort also documented symptom severity, she noted. “So when we look at depression and anxiety we find similar reductions in the Alzheimer’s disease age of onset with each increasing degree of symptom severity,” she said.
“Does this mean that psychiatric disease is a risk factor for Alzheimr’s disease?” Ms. Eijansantos said. “We can’t answer that with this study because it was only designed to see if the psychiatric factors modulate the age of onset in those that have Alzheimer’s disease, but taken together we believe that these results fit the framework that there are pathophysiological and profound differences between earlier and later presentations of Alzheimer’s disease.”
She pointed to reports that early-onset Alzheimer’s disease is associated with more aggressive tau pathology and that depression is associated with tau. However, the evidence supporting a link between amyloid and psychiatric disease is less certain, she said.
Preliminary and speculative findings
Senior study author Zachary Miller, MD, an assistant professor in the UCSF Memory and Aging Center, explained the significance of the study findings of potential links between depression and autoimmunity, and anxiety and seizure. “There may be distinct underlying pathophysiological mechanisms in patients with Alzheimer’s disease who have symptoms of depression versus anxiety,” he said, acknowledging the findings “are quite preliminary and our interpretations quite speculative.”
The findings raise the question that the symptomatic presentation of greater amounts of depression in early-onset Alzheimer’s disease may be moderated by an underlying neuroinflammatory insult, he said. “If so, depression symptomatology could then be seen as a possible clinical marker of this inflammatory response and possibly be used in testing clinical endpoints for future intervention trials,” Dr. Miller said. “Similarly, if neuronal hyperexcitability in Alzheimer’s disease manifests itself as either seizure and/or anxiety, this would have significant impact for therapeutic monitoring and treatment.”
He said a multicenter study of Alzheimer’s disease cohorts would validate the findings. “At the same time, we are also interested in looking deeper into these findings, investigating the potential cognitive and neuroanatomical correlates associated with these conditions,” Dr. Miller said.
Clinical phenotyping may provide more insight into the relationship between psychosis and age of Alzheimer’s disease onset, said Vijay K. Ramanan, MD, PhD, an assistant professor of neurology at Mayo Clinic in Rochester, Minn.
“Less typical presentations of Alzheimer’s disease, such as posterior cortical atrophy or dysexecutive Alzheimer’s disease, are associated with younger age of onset and are sometimes misdiagnosed as having pure psychiatric disease,” he said. “It is also possible that, in some cases with psychiatric disease, a younger age of onset of cognitive symptoms is charted, even though there are fundamentally two distinct processes at play – a psychiatric disease and a separate neurodegenerative disease – each having independent but additive impacts on cognition.”
Dr. Ramanan added, “This work is also a good reminder to be on the lookout for neuropsychiatric symptoms, treat where indicated, and be open to the possibility that psychiatric symptoms and Alzheimer’s disease can coexist.”
Ms. Eijansantos, Dr. Miller, and Dr. Ramanan have no relevant financial relationships to disclose.
, according to findings from a review of 1,500 patients with Alzheimer’s disease from a single-center population.
“Could psychosis symptoms be the proverbial canary in a coal mine?” Emily Eijansantos, a medical student at the University of California, San Francisco, said in reporting results of the chart review at the 2021 annual meeting of the American Academy of Neurology. “Previously in this cohort it was found that neurodevelopmental factors as well as chronic insults such as autoimmunity and seizure were also associated with an early age of onset in Alzheimer’s disease.”
The link between depression and autoimmunity, and anxiety and seizure “beg more questions about underlying pathophysiology,” she said. The study included 750 patients with early-onset Alzheimer’s disease and a similar number of late-onset patients from the UCSF Memory and Aging Center.
An inverse correlation between psychiatric disorders and age of Alzheimer’s onset
In the total study population, 43.5% (n = 652) had a previous diagnosis of depression and 32.3% (n = 485) had been diagnosed with anxiety. That, Ms. Eijansantos said, falls into similar ranges that other studies have reported.
“When we look at individual psychiatric disorders, we find that those with depression, anxiety, or PTSD are younger on average,” she said. “Patients with depression and anxiety are more [likely] female and have less vascular risk factors, and we observed an association between depression and autoimmunity, anxiety, and seizures.”
Specifically, patients with a history of depression were 2.2 years younger, on average, at the age of onset than patients without such history (P = .01); those with anxiety were 3 years younger on average (P = .01); and those with PTSD were 6.8 years younger on average, although only 1% (n = 15) of study subjects had PTSD, making for a small sample to study. These age-of-onset disparities didn’t appear among patients with previously diagnosed bipolar disorder (BPD) or schizophrenia.
Ms. Eijansantos noted that there were no differences in education attained or apolipoprotein-E gene status between the patients with and without a history of psychosis, and, within the subgroups of individual psychiatric disorders, there were no differences between patients with past and current or formal and informal diagnoses.
“When we split the cohort into quintiles based on age of Alzheimer’s disease onset, we find an inverse correlation between the amount of depression, anxiety, and PTSD endorsed and their ages of onset,” Ms. Eijansantos said. For example, the youngest quintile had a greater than 50% rate of depression while the oldest quintile had a depression rate around 36%. A similar spread was found with anxiety: a rate around 46% in the youngest quantile versus around 25% in the oldest, whereas rates of PTSD, BPD, and schizophrenia were similar across the five age-of-onset groups.
Patients with a history of multiple psychiatric disorders had an even younger age of onset. “We see that those with two psychiatric disorder are younger than those with one, and those with three psychiatric disorders are younger still,” Ms. Eijansantos said. “And we find that the Alzheimer’s disease age-at-onset reduction doubles with each additional psychiatric disorder.” Multiple disorders also adversely impacted survival, she said.
Because they found no difference between patients with past versus active symptoms and informal versus formal diagnosis, Ms. Eijansantos explained that they further studied the National Alzheimer’s Coordinating Center cohort of 8,267 patients with Alzheimer’s disease and found a similar relationship between psychiatric history and age of onset among patients with depression or anxiety or both. This cohort also documented symptom severity, she noted. “So when we look at depression and anxiety we find similar reductions in the Alzheimer’s disease age of onset with each increasing degree of symptom severity,” she said.
“Does this mean that psychiatric disease is a risk factor for Alzheimr’s disease?” Ms. Eijansantos said. “We can’t answer that with this study because it was only designed to see if the psychiatric factors modulate the age of onset in those that have Alzheimer’s disease, but taken together we believe that these results fit the framework that there are pathophysiological and profound differences between earlier and later presentations of Alzheimer’s disease.”
She pointed to reports that early-onset Alzheimer’s disease is associated with more aggressive tau pathology and that depression is associated with tau. However, the evidence supporting a link between amyloid and psychiatric disease is less certain, she said.
Preliminary and speculative findings
Senior study author Zachary Miller, MD, an assistant professor in the UCSF Memory and Aging Center, explained the significance of the study findings of potential links between depression and autoimmunity, and anxiety and seizure. “There may be distinct underlying pathophysiological mechanisms in patients with Alzheimer’s disease who have symptoms of depression versus anxiety,” he said, acknowledging the findings “are quite preliminary and our interpretations quite speculative.”
The findings raise the question that the symptomatic presentation of greater amounts of depression in early-onset Alzheimer’s disease may be moderated by an underlying neuroinflammatory insult, he said. “If so, depression symptomatology could then be seen as a possible clinical marker of this inflammatory response and possibly be used in testing clinical endpoints for future intervention trials,” Dr. Miller said. “Similarly, if neuronal hyperexcitability in Alzheimer’s disease manifests itself as either seizure and/or anxiety, this would have significant impact for therapeutic monitoring and treatment.”
He said a multicenter study of Alzheimer’s disease cohorts would validate the findings. “At the same time, we are also interested in looking deeper into these findings, investigating the potential cognitive and neuroanatomical correlates associated with these conditions,” Dr. Miller said.
Clinical phenotyping may provide more insight into the relationship between psychosis and age of Alzheimer’s disease onset, said Vijay K. Ramanan, MD, PhD, an assistant professor of neurology at Mayo Clinic in Rochester, Minn.
“Less typical presentations of Alzheimer’s disease, such as posterior cortical atrophy or dysexecutive Alzheimer’s disease, are associated with younger age of onset and are sometimes misdiagnosed as having pure psychiatric disease,” he said. “It is also possible that, in some cases with psychiatric disease, a younger age of onset of cognitive symptoms is charted, even though there are fundamentally two distinct processes at play – a psychiatric disease and a separate neurodegenerative disease – each having independent but additive impacts on cognition.”
Dr. Ramanan added, “This work is also a good reminder to be on the lookout for neuropsychiatric symptoms, treat where indicated, and be open to the possibility that psychiatric symptoms and Alzheimer’s disease can coexist.”
Ms. Eijansantos, Dr. Miller, and Dr. Ramanan have no relevant financial relationships to disclose.
, according to findings from a review of 1,500 patients with Alzheimer’s disease from a single-center population.
“Could psychosis symptoms be the proverbial canary in a coal mine?” Emily Eijansantos, a medical student at the University of California, San Francisco, said in reporting results of the chart review at the 2021 annual meeting of the American Academy of Neurology. “Previously in this cohort it was found that neurodevelopmental factors as well as chronic insults such as autoimmunity and seizure were also associated with an early age of onset in Alzheimer’s disease.”
The link between depression and autoimmunity, and anxiety and seizure “beg more questions about underlying pathophysiology,” she said. The study included 750 patients with early-onset Alzheimer’s disease and a similar number of late-onset patients from the UCSF Memory and Aging Center.
An inverse correlation between psychiatric disorders and age of Alzheimer’s onset
In the total study population, 43.5% (n = 652) had a previous diagnosis of depression and 32.3% (n = 485) had been diagnosed with anxiety. That, Ms. Eijansantos said, falls into similar ranges that other studies have reported.
“When we look at individual psychiatric disorders, we find that those with depression, anxiety, or PTSD are younger on average,” she said. “Patients with depression and anxiety are more [likely] female and have less vascular risk factors, and we observed an association between depression and autoimmunity, anxiety, and seizures.”
Specifically, patients with a history of depression were 2.2 years younger, on average, at the age of onset than patients without such history (P = .01); those with anxiety were 3 years younger on average (P = .01); and those with PTSD were 6.8 years younger on average, although only 1% (n = 15) of study subjects had PTSD, making for a small sample to study. These age-of-onset disparities didn’t appear among patients with previously diagnosed bipolar disorder (BPD) or schizophrenia.
Ms. Eijansantos noted that there were no differences in education attained or apolipoprotein-E gene status between the patients with and without a history of psychosis, and, within the subgroups of individual psychiatric disorders, there were no differences between patients with past and current or formal and informal diagnoses.
“When we split the cohort into quintiles based on age of Alzheimer’s disease onset, we find an inverse correlation between the amount of depression, anxiety, and PTSD endorsed and their ages of onset,” Ms. Eijansantos said. For example, the youngest quintile had a greater than 50% rate of depression while the oldest quintile had a depression rate around 36%. A similar spread was found with anxiety: a rate around 46% in the youngest quantile versus around 25% in the oldest, whereas rates of PTSD, BPD, and schizophrenia were similar across the five age-of-onset groups.
Patients with a history of multiple psychiatric disorders had an even younger age of onset. “We see that those with two psychiatric disorder are younger than those with one, and those with three psychiatric disorders are younger still,” Ms. Eijansantos said. “And we find that the Alzheimer’s disease age-at-onset reduction doubles with each additional psychiatric disorder.” Multiple disorders also adversely impacted survival, she said.
Because they found no difference between patients with past versus active symptoms and informal versus formal diagnosis, Ms. Eijansantos explained that they further studied the National Alzheimer’s Coordinating Center cohort of 8,267 patients with Alzheimer’s disease and found a similar relationship between psychiatric history and age of onset among patients with depression or anxiety or both. This cohort also documented symptom severity, she noted. “So when we look at depression and anxiety we find similar reductions in the Alzheimer’s disease age of onset with each increasing degree of symptom severity,” she said.
“Does this mean that psychiatric disease is a risk factor for Alzheimr’s disease?” Ms. Eijansantos said. “We can’t answer that with this study because it was only designed to see if the psychiatric factors modulate the age of onset in those that have Alzheimer’s disease, but taken together we believe that these results fit the framework that there are pathophysiological and profound differences between earlier and later presentations of Alzheimer’s disease.”
She pointed to reports that early-onset Alzheimer’s disease is associated with more aggressive tau pathology and that depression is associated with tau. However, the evidence supporting a link between amyloid and psychiatric disease is less certain, she said.
Preliminary and speculative findings
Senior study author Zachary Miller, MD, an assistant professor in the UCSF Memory and Aging Center, explained the significance of the study findings of potential links between depression and autoimmunity, and anxiety and seizure. “There may be distinct underlying pathophysiological mechanisms in patients with Alzheimer’s disease who have symptoms of depression versus anxiety,” he said, acknowledging the findings “are quite preliminary and our interpretations quite speculative.”
The findings raise the question that the symptomatic presentation of greater amounts of depression in early-onset Alzheimer’s disease may be moderated by an underlying neuroinflammatory insult, he said. “If so, depression symptomatology could then be seen as a possible clinical marker of this inflammatory response and possibly be used in testing clinical endpoints for future intervention trials,” Dr. Miller said. “Similarly, if neuronal hyperexcitability in Alzheimer’s disease manifests itself as either seizure and/or anxiety, this would have significant impact for therapeutic monitoring and treatment.”
He said a multicenter study of Alzheimer’s disease cohorts would validate the findings. “At the same time, we are also interested in looking deeper into these findings, investigating the potential cognitive and neuroanatomical correlates associated with these conditions,” Dr. Miller said.
Clinical phenotyping may provide more insight into the relationship between psychosis and age of Alzheimer’s disease onset, said Vijay K. Ramanan, MD, PhD, an assistant professor of neurology at Mayo Clinic in Rochester, Minn.
“Less typical presentations of Alzheimer’s disease, such as posterior cortical atrophy or dysexecutive Alzheimer’s disease, are associated with younger age of onset and are sometimes misdiagnosed as having pure psychiatric disease,” he said. “It is also possible that, in some cases with psychiatric disease, a younger age of onset of cognitive symptoms is charted, even though there are fundamentally two distinct processes at play – a psychiatric disease and a separate neurodegenerative disease – each having independent but additive impacts on cognition.”
Dr. Ramanan added, “This work is also a good reminder to be on the lookout for neuropsychiatric symptoms, treat where indicated, and be open to the possibility that psychiatric symptoms and Alzheimer’s disease can coexist.”
Ms. Eijansantos, Dr. Miller, and Dr. Ramanan have no relevant financial relationships to disclose.
FROM AAN 2021
Cortical surface changes linked to sensorimotor abnormalities in schizophrenia
Schizophrenia patients with parkinsonism show unique neurodevelopmental signatures on imaging that involve the sensorimotor system, according to MRI data from 73 adult schizophrenia patients.
Although sensorimotor abnormalities are common in patients with schizophrenia, the neurobiology of parkinsonism in particular is not well understood. Aberrant neurodevelopment is considered a potential mechanism of action for the emergence of such abnormalities, wrote Robert Christian Wolf, MD, of Heidelberg (Germany) University, and colleagues.
In a multimodal MRI study published in Schizophrenia Research, the investigators identified 38 adults with schizophrenia and parkinsonism (SZ-P), 35 schizophrenia patients without parkinsonism (SZ-nonP), and 20 healthy controls.
Parkinsonism was defined as scores of 4 or higher on the Simpson-Angus Scale, while non-Parkinsonism schizophrenia patients had scores of 1 or less.
The researchers examined cortical and subcortical gray-matter volume, as well as three cortical surface markers related to neurodevelopment: cortical thickness (CTh), complexity of cortical folding (CCF), and sulcus depth.
Overall, the SZ-P patients showed increased CCF in the left supplementary motor cortex (SMC) and decreased left postcentral sulcus depth, compared with SZ-nonP patients (P < .05). The left SMC also showed increased CCF, compared with healthy controls – but that difference was not significant.
Both SZ-P and SZ-nonP patients showed higher levels of activity in the left SMC, compared with controls, and activity was higher in SZ-nonP patients, compared with SZ-P patients. In addition, Dr. Wolf and colleagues reported.
“Overall, the data support the notion that cortical features of distinct neurodevelopmental origin, particularly cortical folding indices such as CCF and sulcus depth, contribute to the pathogenesis of parkinsonism in SZ,” the researchers said.
The study findings were limited by several factors, including the cross-sectional design, the challenges of using the potential restraint inherent in the Simpson-Angus Scale to diagnose parkinsonism, the inability to gauge the impact of lifetime exposure to antipsychotics, and the inability to identify changes in brain stem nuclei, the researchers noted. However, the results suggest the impact of cortical development on parkinsonism in schizophrenia,.
“Cortical surface changes in the sensorimotor system suggest abnormal neurodevelopmental processes that are associated with increased risk for intrinsic sensorimotor abnormalities in SZ and related psychotic disorders,” they concluded.
The study was supported by the German Research Foundation and the German Federal Ministry of Education and Research. The researchers disclosed no financial conflicts.
Schizophrenia patients with parkinsonism show unique neurodevelopmental signatures on imaging that involve the sensorimotor system, according to MRI data from 73 adult schizophrenia patients.
Although sensorimotor abnormalities are common in patients with schizophrenia, the neurobiology of parkinsonism in particular is not well understood. Aberrant neurodevelopment is considered a potential mechanism of action for the emergence of such abnormalities, wrote Robert Christian Wolf, MD, of Heidelberg (Germany) University, and colleagues.
In a multimodal MRI study published in Schizophrenia Research, the investigators identified 38 adults with schizophrenia and parkinsonism (SZ-P), 35 schizophrenia patients without parkinsonism (SZ-nonP), and 20 healthy controls.
Parkinsonism was defined as scores of 4 or higher on the Simpson-Angus Scale, while non-Parkinsonism schizophrenia patients had scores of 1 or less.
The researchers examined cortical and subcortical gray-matter volume, as well as three cortical surface markers related to neurodevelopment: cortical thickness (CTh), complexity of cortical folding (CCF), and sulcus depth.
Overall, the SZ-P patients showed increased CCF in the left supplementary motor cortex (SMC) and decreased left postcentral sulcus depth, compared with SZ-nonP patients (P < .05). The left SMC also showed increased CCF, compared with healthy controls – but that difference was not significant.
Both SZ-P and SZ-nonP patients showed higher levels of activity in the left SMC, compared with controls, and activity was higher in SZ-nonP patients, compared with SZ-P patients. In addition, Dr. Wolf and colleagues reported.
“Overall, the data support the notion that cortical features of distinct neurodevelopmental origin, particularly cortical folding indices such as CCF and sulcus depth, contribute to the pathogenesis of parkinsonism in SZ,” the researchers said.
The study findings were limited by several factors, including the cross-sectional design, the challenges of using the potential restraint inherent in the Simpson-Angus Scale to diagnose parkinsonism, the inability to gauge the impact of lifetime exposure to antipsychotics, and the inability to identify changes in brain stem nuclei, the researchers noted. However, the results suggest the impact of cortical development on parkinsonism in schizophrenia,.
“Cortical surface changes in the sensorimotor system suggest abnormal neurodevelopmental processes that are associated with increased risk for intrinsic sensorimotor abnormalities in SZ and related psychotic disorders,” they concluded.
The study was supported by the German Research Foundation and the German Federal Ministry of Education and Research. The researchers disclosed no financial conflicts.
Schizophrenia patients with parkinsonism show unique neurodevelopmental signatures on imaging that involve the sensorimotor system, according to MRI data from 73 adult schizophrenia patients.
Although sensorimotor abnormalities are common in patients with schizophrenia, the neurobiology of parkinsonism in particular is not well understood. Aberrant neurodevelopment is considered a potential mechanism of action for the emergence of such abnormalities, wrote Robert Christian Wolf, MD, of Heidelberg (Germany) University, and colleagues.
In a multimodal MRI study published in Schizophrenia Research, the investigators identified 38 adults with schizophrenia and parkinsonism (SZ-P), 35 schizophrenia patients without parkinsonism (SZ-nonP), and 20 healthy controls.
Parkinsonism was defined as scores of 4 or higher on the Simpson-Angus Scale, while non-Parkinsonism schizophrenia patients had scores of 1 or less.
The researchers examined cortical and subcortical gray-matter volume, as well as three cortical surface markers related to neurodevelopment: cortical thickness (CTh), complexity of cortical folding (CCF), and sulcus depth.
Overall, the SZ-P patients showed increased CCF in the left supplementary motor cortex (SMC) and decreased left postcentral sulcus depth, compared with SZ-nonP patients (P < .05). The left SMC also showed increased CCF, compared with healthy controls – but that difference was not significant.
Both SZ-P and SZ-nonP patients showed higher levels of activity in the left SMC, compared with controls, and activity was higher in SZ-nonP patients, compared with SZ-P patients. In addition, Dr. Wolf and colleagues reported.
“Overall, the data support the notion that cortical features of distinct neurodevelopmental origin, particularly cortical folding indices such as CCF and sulcus depth, contribute to the pathogenesis of parkinsonism in SZ,” the researchers said.
The study findings were limited by several factors, including the cross-sectional design, the challenges of using the potential restraint inherent in the Simpson-Angus Scale to diagnose parkinsonism, the inability to gauge the impact of lifetime exposure to antipsychotics, and the inability to identify changes in brain stem nuclei, the researchers noted. However, the results suggest the impact of cortical development on parkinsonism in schizophrenia,.
“Cortical surface changes in the sensorimotor system suggest abnormal neurodevelopmental processes that are associated with increased risk for intrinsic sensorimotor abnormalities in SZ and related psychotic disorders,” they concluded.
The study was supported by the German Research Foundation and the German Federal Ministry of Education and Research. The researchers disclosed no financial conflicts.
FROM SCHIZOPHRENIA RESEARCH
Antipsychotics protective against COVID-19?
Antipsychotics may protect against SARS-CoV-2 infection or lead to a milder course of illness, new research suggests.
“Counterintuitively,” the investigators noted, vulnerable people with severe mental illness “on antipsychotic treatment showed a lower risk of SARS-CoV-2 infection and a likely better COVID-19 prognosis.”
“These are very interesting findings that reflect a clinical reality where we see few patients with severe COVID-19, despite the presence of various risk factors,” study investigator Manuel Canal-Rivero, PhD, clinical psychologist, Virgen del Rocio University Hospital, Sevilla, Spain, said in a news release.
“The number of COVID-19 patients is lower than expected among this group of people and in cases where a proven infection does occur, the evolution is benign and does not reach a life-threatening clinical situation. These data as a whole seem to point to the protective effect of the medication,” Dr. Canal-Rivero added.
The study was published online as a letter to the editor February 19, 2021, in Schizophrenia Research.
A ‘striking’ finding
The researchers assessed the prevalence and prognosis of COVID-19 in 698 patients with serious mental disorders (SMDs) receiving treatment with long-acting injectable antipsychotic medication. The non-SMD population included the catchment area population of 557,576 individuals.
From February to November 2020, 4.1% of the non-SMD population were infected with SARS-CoV-2 versus just 1.3% of the SMD population (9 of 698 patients). All but one patient with SMD had asymptomatic illness (8 of 9, 89%). Accurate information on asymptomatic illness in the non-SMD population was not available.
There were also fewer hospital admissions in the SMD population (0% vs. 8.5%), ICU admissions (0% vs. 0.9%) and deaths because of COVID-19 (0% vs. 1.1%), although the differences were not statistically significant.
In related research, the same investigators found that many of the genes whose expression is altered by SARS-CoV-2 infection are significantly down-regulated by antipsychotic drugs.
“In a striking way, we have shown how antipsychotics reduce the activation of genes involved in many of the inflammatory and immunological pathways associated with the severity of COVID-19 infection,” Benedicto Crespo-Facorro, MD, PhD, University of Sevilla, who led the study, said in the news release.
“Although this finding requires replication, the discovery could be very significant because the treatment of COVID-19 with drugs originally indicated for unrelated clinical situations, that is to say drug repositioning, has been shown to be an interesting source of effective treatments for COVID-19 patients,” he added.
Antiviral properties?
In a comment, Samoon Ahmad, MD, professor, department of psychiatry, New York University, said the findings are “fascinating” and should be explored further.
While the findings on long-acting injectable antipsychotic treatment “seem counterintuitive at first, they are in line with other studies,” said Dr. Ahmad, who heads the inpatient psychiatry unit at Bellevue Hospital Center and is founder of the Integrative Center for Wellness in New York.
“ and antidepressant medications appear to activate key cellular proteins that the SARS-CoV-2 virus uses for replication,” explained Dr. Ahmad, who was not associated with the study.
For example, as reported by this news organization, a preliminary study published in 2020 showed that early treatment with the antidepressant fluvoxamine prevented clinical deterioration in adult outpatients with confirmed COVID-19.
The antipsychotic aripiprazole has also shown potential to treat severe COVID-19 infection.
“Consequently, there appears to be a possible explanation as to why these drugs afford patients with severe mental disorders increased protection against the SARS-CoV-2 virus,” Dr. Ahmad said in an interview.
However, he cautioned, there are several factors at play that could influence the results. Therefore, more research is needed before drawing any firm conclusions.
“Still, the possibility that psychiatric medications may have antiviral properties is a tremendous development and I really hope that additional studies confirm the preliminary findings,” Dr. Ahmad said.
The study had no specific funding. The authors and Dr. Ahmad disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Antipsychotics may protect against SARS-CoV-2 infection or lead to a milder course of illness, new research suggests.
“Counterintuitively,” the investigators noted, vulnerable people with severe mental illness “on antipsychotic treatment showed a lower risk of SARS-CoV-2 infection and a likely better COVID-19 prognosis.”
“These are very interesting findings that reflect a clinical reality where we see few patients with severe COVID-19, despite the presence of various risk factors,” study investigator Manuel Canal-Rivero, PhD, clinical psychologist, Virgen del Rocio University Hospital, Sevilla, Spain, said in a news release.
“The number of COVID-19 patients is lower than expected among this group of people and in cases where a proven infection does occur, the evolution is benign and does not reach a life-threatening clinical situation. These data as a whole seem to point to the protective effect of the medication,” Dr. Canal-Rivero added.
The study was published online as a letter to the editor February 19, 2021, in Schizophrenia Research.
A ‘striking’ finding
The researchers assessed the prevalence and prognosis of COVID-19 in 698 patients with serious mental disorders (SMDs) receiving treatment with long-acting injectable antipsychotic medication. The non-SMD population included the catchment area population of 557,576 individuals.
From February to November 2020, 4.1% of the non-SMD population were infected with SARS-CoV-2 versus just 1.3% of the SMD population (9 of 698 patients). All but one patient with SMD had asymptomatic illness (8 of 9, 89%). Accurate information on asymptomatic illness in the non-SMD population was not available.
There were also fewer hospital admissions in the SMD population (0% vs. 8.5%), ICU admissions (0% vs. 0.9%) and deaths because of COVID-19 (0% vs. 1.1%), although the differences were not statistically significant.
In related research, the same investigators found that many of the genes whose expression is altered by SARS-CoV-2 infection are significantly down-regulated by antipsychotic drugs.
“In a striking way, we have shown how antipsychotics reduce the activation of genes involved in many of the inflammatory and immunological pathways associated with the severity of COVID-19 infection,” Benedicto Crespo-Facorro, MD, PhD, University of Sevilla, who led the study, said in the news release.
“Although this finding requires replication, the discovery could be very significant because the treatment of COVID-19 with drugs originally indicated for unrelated clinical situations, that is to say drug repositioning, has been shown to be an interesting source of effective treatments for COVID-19 patients,” he added.
Antiviral properties?
In a comment, Samoon Ahmad, MD, professor, department of psychiatry, New York University, said the findings are “fascinating” and should be explored further.
While the findings on long-acting injectable antipsychotic treatment “seem counterintuitive at first, they are in line with other studies,” said Dr. Ahmad, who heads the inpatient psychiatry unit at Bellevue Hospital Center and is founder of the Integrative Center for Wellness in New York.
“ and antidepressant medications appear to activate key cellular proteins that the SARS-CoV-2 virus uses for replication,” explained Dr. Ahmad, who was not associated with the study.
For example, as reported by this news organization, a preliminary study published in 2020 showed that early treatment with the antidepressant fluvoxamine prevented clinical deterioration in adult outpatients with confirmed COVID-19.
The antipsychotic aripiprazole has also shown potential to treat severe COVID-19 infection.
“Consequently, there appears to be a possible explanation as to why these drugs afford patients with severe mental disorders increased protection against the SARS-CoV-2 virus,” Dr. Ahmad said in an interview.
However, he cautioned, there are several factors at play that could influence the results. Therefore, more research is needed before drawing any firm conclusions.
“Still, the possibility that psychiatric medications may have antiviral properties is a tremendous development and I really hope that additional studies confirm the preliminary findings,” Dr. Ahmad said.
The study had no specific funding. The authors and Dr. Ahmad disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Antipsychotics may protect against SARS-CoV-2 infection or lead to a milder course of illness, new research suggests.
“Counterintuitively,” the investigators noted, vulnerable people with severe mental illness “on antipsychotic treatment showed a lower risk of SARS-CoV-2 infection and a likely better COVID-19 prognosis.”
“These are very interesting findings that reflect a clinical reality where we see few patients with severe COVID-19, despite the presence of various risk factors,” study investigator Manuel Canal-Rivero, PhD, clinical psychologist, Virgen del Rocio University Hospital, Sevilla, Spain, said in a news release.
“The number of COVID-19 patients is lower than expected among this group of people and in cases where a proven infection does occur, the evolution is benign and does not reach a life-threatening clinical situation. These data as a whole seem to point to the protective effect of the medication,” Dr. Canal-Rivero added.
The study was published online as a letter to the editor February 19, 2021, in Schizophrenia Research.
A ‘striking’ finding
The researchers assessed the prevalence and prognosis of COVID-19 in 698 patients with serious mental disorders (SMDs) receiving treatment with long-acting injectable antipsychotic medication. The non-SMD population included the catchment area population of 557,576 individuals.
From February to November 2020, 4.1% of the non-SMD population were infected with SARS-CoV-2 versus just 1.3% of the SMD population (9 of 698 patients). All but one patient with SMD had asymptomatic illness (8 of 9, 89%). Accurate information on asymptomatic illness in the non-SMD population was not available.
There were also fewer hospital admissions in the SMD population (0% vs. 8.5%), ICU admissions (0% vs. 0.9%) and deaths because of COVID-19 (0% vs. 1.1%), although the differences were not statistically significant.
In related research, the same investigators found that many of the genes whose expression is altered by SARS-CoV-2 infection are significantly down-regulated by antipsychotic drugs.
“In a striking way, we have shown how antipsychotics reduce the activation of genes involved in many of the inflammatory and immunological pathways associated with the severity of COVID-19 infection,” Benedicto Crespo-Facorro, MD, PhD, University of Sevilla, who led the study, said in the news release.
“Although this finding requires replication, the discovery could be very significant because the treatment of COVID-19 with drugs originally indicated for unrelated clinical situations, that is to say drug repositioning, has been shown to be an interesting source of effective treatments for COVID-19 patients,” he added.
Antiviral properties?
In a comment, Samoon Ahmad, MD, professor, department of psychiatry, New York University, said the findings are “fascinating” and should be explored further.
While the findings on long-acting injectable antipsychotic treatment “seem counterintuitive at first, they are in line with other studies,” said Dr. Ahmad, who heads the inpatient psychiatry unit at Bellevue Hospital Center and is founder of the Integrative Center for Wellness in New York.
“ and antidepressant medications appear to activate key cellular proteins that the SARS-CoV-2 virus uses for replication,” explained Dr. Ahmad, who was not associated with the study.
For example, as reported by this news organization, a preliminary study published in 2020 showed that early treatment with the antidepressant fluvoxamine prevented clinical deterioration in adult outpatients with confirmed COVID-19.
The antipsychotic aripiprazole has also shown potential to treat severe COVID-19 infection.
“Consequently, there appears to be a possible explanation as to why these drugs afford patients with severe mental disorders increased protection against the SARS-CoV-2 virus,” Dr. Ahmad said in an interview.
However, he cautioned, there are several factors at play that could influence the results. Therefore, more research is needed before drawing any firm conclusions.
“Still, the possibility that psychiatric medications may have antiviral properties is a tremendous development and I really hope that additional studies confirm the preliminary findings,” Dr. Ahmad said.
The study had no specific funding. The authors and Dr. Ahmad disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Contradictions abound in ‘The End of Mental Illness’
Daniel G. Amen, MD, is an American psychiatrist well-known for his eponymous clinics, television appearances, and series of books on mental health. One of his latest books, “The End of Mental Illness,” summarizes many of his views on the causes of and treatments for mental illnesses.
Dr. Amen’s approaches – such as his advocacy for the widespread use of single photon emission computed tomography (SPECT) imaging – are somewhat controversial and at times fall outside the mainstream of current psychiatric thought. So does “The End of Mental Illness” contain anything of value to the average practicing psychiatrist? (It should be noted that I listened to this as an audiobook and took notes as I listened. This does limit my ability to directly quote portions of the text, but I believe my notes are reliable.)
He begins the book by pointing out that the term “mental illness” might be better replaced with the term “brain illness.” With this shift in terminology, Dr. Amen introduces a theme that recurs throughout the book: That mental illnesses ultimately stem from various ways in which the brain can be harmed. While the suggested change in terminology might help reduce the stigma associated with psychiatric illnesses, Dr. Amen is surprisingly timid about implementing this term in his own book. He repeatedly refers to “brain health/mental health” issues instead of discarding the “mental” term altogether. Even his BRIGHT MINDS acronym for risk factors for mental illnesses includes the term “mind” instead of “brain.”
Continuing the theme of challenging terminology, Dr. Amen goes on to decry the weaknesses of the DSM system of nosology. This is a valid point, because under the current system, the same patient may receive differing diagnoses depending on which provider is seen and how certain symptoms are interpreted. Yet, here again, Dr. Amen does not seem to adhere to his own advice: He uses DSM terminology throughout the book, speaking of depression, anxiety, bipolar disorder, and ADHD. An oddity (which, admittedly, could have been the audiobook reader’s mistake rather than an error in the original text) is that the DSM is referred to as the “Diagnostic and Structural Manual” rather than the Diagnostic and Statistical Manual. He criticizes the DSM for its imprecision, pointing out the variety of symptom combinations that can produce the same diagnoses and how similar symptoms may overlap between differing diagnoses. Yet, his descriptions of common SPECT patterns (his preferred tool to assist in diagnosis) make it clear that here, too, there is a lot of overlap. As an example, ADHD was associated with at least three of the imaging patterns he described. It is also somewhat ironic how Dr. Amen obliquely criticizes the American Psychiatric Association for profiting from the use of the DSM, when SPECT imaging is expensive and profits his own organization.
Dr. Amen repeatedly asserts that psychiatry is unique among medical specialties for making diagnoses based on symptom clusters rather than direct visualization of the affected organ. Yet, psychiatry is not, in fact, unique in making diagnoses in this way. Some examples of diagnoses based on symptom clusters from other medical specialties are systemic lupus erythematosus, fibromyalgia, and chronic fatigue syndrome. Although he asserts that SPECT imaging better demonstrates the root cause of mental illnesses, it is unclear from his book whether this is actually the case.
The descriptions for the ways in which Dr. Amen uses SPECT (which, admittedly, are vague and presumably simplified for a general audience) suggest that he has made observations correlating specific imaging patterns with certain emotional/behavioral outcomes. However, the imaging patterns he describes in the book can be interpreted to represent multiple different mental conditions, making it clear that SPECT is not a laserlike diagnostic tool that produces a single, indisputable diagnosis. Accuracy with SPECT seems especially questionable in light of two case examples he shares where brain imaging was interpreted as representing illness, but the patients were not demonstrating any signs of mental dysfunction. In one case, Dr. Amen opined that the patient’s vibrant spiritual life “overrode” the sick brain, but if this is true,
Patient testimonials are provided, asserting that SPECT imaging helped them know “exactly” what treatment would help them. One cannot help but wonder whether part of the benefit of SPECT imaging is a placebo effect, boosting the confidence of patients that the treatment they are receiving is personalized and scientifically sound. A similar trend is currently seen more broadly in psychiatry with the widespread promotion of pharmacogenetic testing. Such testing may bolster patient confidence in their medication, but its value in improving patient outcomes has not been established.1
Dr. Amen outlines a brief history of mental health care, including differing approaches and therapies from the time of Sigmund Freud up to the present. His outline is somewhat critical of the perceived shortcomings of his psychiatric forebears, yet this seems entirely unnecessary. All scientific disciplines must start somewhere and build from limited knowledge to greater. Is it necessary to belittle Freud for not being able to do SPECT imaging in the 1800s?
Interestingly, Dr. Amen leaves cognitive-behavioral therapy (CBT), a landmark, evidence-based form of psychotherapy, out of his overview of the history of psychiatry. He does go on to mention CBT as part of the treatment offerings of the Amen Clinics, which could leave the lay reader with the incorrect impression that CBT is a treatment unique to Amen Clinics. Similarly, at one point Dr. Amen writes about “what I call automatic negative thoughts.” This phrasing could confuse readers who might not know that automatic thoughts are a concept endemic to CBT.
Dr. Amen writes repeatedly about the Amen Clinics 4 Circles, four key areas of life that can contribute to mental health. These areas are biological, psychological, social, and spiritual. While Amen Clinics may have come up with the term “4 Circles,” the biopsychosocial model of understanding illness was developed by George Engel, MD, in 1977, and current discussions of this model frequently incorporate a spiritual dimension as well.2
Dr. Amen’s writing at times mischaracterizes psychotropic medications in unhelpful ways. He speaks of psychotropic medications generally as being addictive. While this is certainly true for stimulants and benzodiazepines, most would agree that this does not apply to many other commonly used medications in psychiatry, including selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants, antipsychotics, and mood stabilizers. He also paints with a broad brush when he states that anxiety medications can cause dementia. A concerning link has been demonstrated between benzodiazepine use and dementia,3 but SSRIs (which are considered first-line medications for anxiety) are not known to cause dementia and may actually delay progression from mild cognitive impairment to Alzheimer’s dementia.4 His mention of medication use affecting a patient’s insurability could have the unfortunate effect of scaring away suffering individuals from seeking help. The one category of psychiatric medication he does not seem concerned about is psychostimulants, which is odd – given the addictive, cardiovascular, and other risks associated with that medication class.
In contrast to his skepticism regarding many psychotropic medications, Dr. Amen expresses significant enthusiasm regarding nutraceutical use. While there has been research in this area supporting a role for some nutraceutical interventions, there is still a need for more rigorous studies.5 To support his endorsement of natural remedies, Dr. Amen mentions that Hippocrates recommended herbs and spices for many health conditions. But Hippocrates lived more than 2,000 years ago, and the state of medicine has advanced significantly since then.
Dr. Amen also mentions that 80% of the developing world relies upon natural or herbal remedies as the primary source of medicine. While he frames this statement as supporting his endorsement of such remedies, it could conversely be said that this is evidence of the need to make pharmacological interventions more widely available in the developing world.
Much of “The End of Mental Illness” is dedicated to reviewing specific risk factors that could cause harm to a person’s mental well-being. One example is head trauma. Dr. Amen documents at least one instance in which he was convinced that his patient had experienced head trauma, and questioned the patient again and again about possible brain injuries. One must wonder whether the positive results of such focused, repetitive questioning might be evidence of confirmation bias, as a search to confirm the preexisting belief of head trauma could lead to overlooking alternative explanations for a patient’s symptoms.
Another risk factor dwelt upon is exposure to toxins. One toxin Dr. Amen rightly recommends avoiding is tobacco smoke. Yet, his approach to advocate for a tobacco-free lifestyle is somewhat problematic. He lists chemicals contained in tobacco smoke, and then names unpleasant items that share those ingredients, such as paint. This smacks of the same sloppy logic manifested in social media memes decrying the use of vaccines by listing their ingredients alongside scary-sounding products that contain identical ingredients (for example, vaccines contain formaldehyde, which is used to embalm dead bodies!). This is analogous to saying that water is bad for you because it contains hydrogen, which is also an ingredient in atomic bombs.
Dr. Amen makes the blanket recommendation to avoid products containing “chemicals.” This is a difficult recommendation to interpret, since literally all matter is made of chemicals. It seems that Dr. Amen is leaning into the vague idea of a “chemical” as something artificially created in a lab, which must, therefore, be dangerous.
Along these lines, Dr. Amen suggests that if a person doesn’t know what is in a specific food item, it should not be eaten. Although this sounds reasonable on the surface, if people were told the names of the proteins and chemical compounds that make up many naturally occurring plants or meats, they would likely not recognize many of them. Dr. Amen dedicates space to list seemingly benign exposures – such as eating nonorganic produce, using two or more beauty products each day, or touching grocery store receipts – as possible “toxins.” By contrast, there is a certain irony in the absence of any mention of the risks associated with radiation from the SPECT imaging he staunchly advocates for. One potential risk of the book listing so many “toxins” to avoid is that patients could waste valuable time and energy eliminating exposures that pose little or no risk, rather than focusing efforts on well-established treatments.
In light of the observations and critiques offered above, one might come away with the impression that I would not recommend “The End of Mental Illness.” However, although one can nitpick details in the book, some of its bigger ideas make it worth commending to readers. Dr. Amen rightfully emphasizes the need for psychiatrists and patients to think more broadly about mental health issues beyond the use of pills. He justifiably criticizes the “15-minute med check” model of practice and the idea that medications are the end-all, be-all of treatment. He demonstrates an appropriate appreciation for the serious risks of reliance on benzodiazepines.6 Dr. Amen points out important contributions from Viktor Frankl, MD, to the field of psychiatry, which may go overlooked today. He also helpfully points out that bipolar disorder may often be misdiagnosed (although he attributes the misdiagnosis to traumatic brain injury, whereas other psychiatrists might say the misdiagnosis is due to borderline personality disorder).
Much of what Dr. Amen writes is sensible, and psychiatrists would do well to adopt the following steps he advocates for: Taking a comprehensive biopsychosocial-spiritual approach to the assessment and treatment of patients; thinking broadly in their differential diagnoses and not forgetting their medical training; understanding that medication alone is often not sufficient to make lasting, positive change in a person’s life; paying attention to healthy habits such as diet, exercise, sleep, and social activity; and knowing that CBT is a valuable tool that can change lives.
There is much to appreciate in “The End of Mental Illness,” especially the overarching idea that psychiatry isn’t just a symptom checklist and a prescription pad. Rather, achieving mental well-being often requires broader thinking and sustained lifestyle changes.
Although I did not agree with everything in the book, it did cause me to think and reflect on my own practice. I read “The End of Mental Illness” with colleagues in my department, and it stimulated a lively discussion. Isn’t that ultimately what a psychiatrist would want from a book like this – the opportunity to reflect, discuss, and potentially improve one’s own practice?
Dr. Weber is physician lead in the department of psychiatry at Intermountain Healthcare Budge Clinic, Logan (Utah) Psychiatry. He disclosed no relevant financial relationships.
References
1. JAMA Netw Open. 2020;3(12). doi: 10.1001/jamanetworkopen.2020.27909.
2. Curr Opin Psychiatry. 2014;27:358-63.
3. BMJ 2014. doi: 10.1136/bmj.g5205.
4. Am J Psychiatry. 2018 Mar 1;175:232-41.
5. Am J Psychiatry. 2016 Jun 1;173:575-87.
6. Current Psychiatry. 2018 Feb;17(2):22-7.
Daniel G. Amen, MD, is an American psychiatrist well-known for his eponymous clinics, television appearances, and series of books on mental health. One of his latest books, “The End of Mental Illness,” summarizes many of his views on the causes of and treatments for mental illnesses.
Dr. Amen’s approaches – such as his advocacy for the widespread use of single photon emission computed tomography (SPECT) imaging – are somewhat controversial and at times fall outside the mainstream of current psychiatric thought. So does “The End of Mental Illness” contain anything of value to the average practicing psychiatrist? (It should be noted that I listened to this as an audiobook and took notes as I listened. This does limit my ability to directly quote portions of the text, but I believe my notes are reliable.)
He begins the book by pointing out that the term “mental illness” might be better replaced with the term “brain illness.” With this shift in terminology, Dr. Amen introduces a theme that recurs throughout the book: That mental illnesses ultimately stem from various ways in which the brain can be harmed. While the suggested change in terminology might help reduce the stigma associated with psychiatric illnesses, Dr. Amen is surprisingly timid about implementing this term in his own book. He repeatedly refers to “brain health/mental health” issues instead of discarding the “mental” term altogether. Even his BRIGHT MINDS acronym for risk factors for mental illnesses includes the term “mind” instead of “brain.”
Continuing the theme of challenging terminology, Dr. Amen goes on to decry the weaknesses of the DSM system of nosology. This is a valid point, because under the current system, the same patient may receive differing diagnoses depending on which provider is seen and how certain symptoms are interpreted. Yet, here again, Dr. Amen does not seem to adhere to his own advice: He uses DSM terminology throughout the book, speaking of depression, anxiety, bipolar disorder, and ADHD. An oddity (which, admittedly, could have been the audiobook reader’s mistake rather than an error in the original text) is that the DSM is referred to as the “Diagnostic and Structural Manual” rather than the Diagnostic and Statistical Manual. He criticizes the DSM for its imprecision, pointing out the variety of symptom combinations that can produce the same diagnoses and how similar symptoms may overlap between differing diagnoses. Yet, his descriptions of common SPECT patterns (his preferred tool to assist in diagnosis) make it clear that here, too, there is a lot of overlap. As an example, ADHD was associated with at least three of the imaging patterns he described. It is also somewhat ironic how Dr. Amen obliquely criticizes the American Psychiatric Association for profiting from the use of the DSM, when SPECT imaging is expensive and profits his own organization.
Dr. Amen repeatedly asserts that psychiatry is unique among medical specialties for making diagnoses based on symptom clusters rather than direct visualization of the affected organ. Yet, psychiatry is not, in fact, unique in making diagnoses in this way. Some examples of diagnoses based on symptom clusters from other medical specialties are systemic lupus erythematosus, fibromyalgia, and chronic fatigue syndrome. Although he asserts that SPECT imaging better demonstrates the root cause of mental illnesses, it is unclear from his book whether this is actually the case.
The descriptions for the ways in which Dr. Amen uses SPECT (which, admittedly, are vague and presumably simplified for a general audience) suggest that he has made observations correlating specific imaging patterns with certain emotional/behavioral outcomes. However, the imaging patterns he describes in the book can be interpreted to represent multiple different mental conditions, making it clear that SPECT is not a laserlike diagnostic tool that produces a single, indisputable diagnosis. Accuracy with SPECT seems especially questionable in light of two case examples he shares where brain imaging was interpreted as representing illness, but the patients were not demonstrating any signs of mental dysfunction. In one case, Dr. Amen opined that the patient’s vibrant spiritual life “overrode” the sick brain, but if this is true,
Patient testimonials are provided, asserting that SPECT imaging helped them know “exactly” what treatment would help them. One cannot help but wonder whether part of the benefit of SPECT imaging is a placebo effect, boosting the confidence of patients that the treatment they are receiving is personalized and scientifically sound. A similar trend is currently seen more broadly in psychiatry with the widespread promotion of pharmacogenetic testing. Such testing may bolster patient confidence in their medication, but its value in improving patient outcomes has not been established.1
Dr. Amen outlines a brief history of mental health care, including differing approaches and therapies from the time of Sigmund Freud up to the present. His outline is somewhat critical of the perceived shortcomings of his psychiatric forebears, yet this seems entirely unnecessary. All scientific disciplines must start somewhere and build from limited knowledge to greater. Is it necessary to belittle Freud for not being able to do SPECT imaging in the 1800s?
Interestingly, Dr. Amen leaves cognitive-behavioral therapy (CBT), a landmark, evidence-based form of psychotherapy, out of his overview of the history of psychiatry. He does go on to mention CBT as part of the treatment offerings of the Amen Clinics, which could leave the lay reader with the incorrect impression that CBT is a treatment unique to Amen Clinics. Similarly, at one point Dr. Amen writes about “what I call automatic negative thoughts.” This phrasing could confuse readers who might not know that automatic thoughts are a concept endemic to CBT.
Dr. Amen writes repeatedly about the Amen Clinics 4 Circles, four key areas of life that can contribute to mental health. These areas are biological, psychological, social, and spiritual. While Amen Clinics may have come up with the term “4 Circles,” the biopsychosocial model of understanding illness was developed by George Engel, MD, in 1977, and current discussions of this model frequently incorporate a spiritual dimension as well.2
Dr. Amen’s writing at times mischaracterizes psychotropic medications in unhelpful ways. He speaks of psychotropic medications generally as being addictive. While this is certainly true for stimulants and benzodiazepines, most would agree that this does not apply to many other commonly used medications in psychiatry, including selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants, antipsychotics, and mood stabilizers. He also paints with a broad brush when he states that anxiety medications can cause dementia. A concerning link has been demonstrated between benzodiazepine use and dementia,3 but SSRIs (which are considered first-line medications for anxiety) are not known to cause dementia and may actually delay progression from mild cognitive impairment to Alzheimer’s dementia.4 His mention of medication use affecting a patient’s insurability could have the unfortunate effect of scaring away suffering individuals from seeking help. The one category of psychiatric medication he does not seem concerned about is psychostimulants, which is odd – given the addictive, cardiovascular, and other risks associated with that medication class.
In contrast to his skepticism regarding many psychotropic medications, Dr. Amen expresses significant enthusiasm regarding nutraceutical use. While there has been research in this area supporting a role for some nutraceutical interventions, there is still a need for more rigorous studies.5 To support his endorsement of natural remedies, Dr. Amen mentions that Hippocrates recommended herbs and spices for many health conditions. But Hippocrates lived more than 2,000 years ago, and the state of medicine has advanced significantly since then.
Dr. Amen also mentions that 80% of the developing world relies upon natural or herbal remedies as the primary source of medicine. While he frames this statement as supporting his endorsement of such remedies, it could conversely be said that this is evidence of the need to make pharmacological interventions more widely available in the developing world.
Much of “The End of Mental Illness” is dedicated to reviewing specific risk factors that could cause harm to a person’s mental well-being. One example is head trauma. Dr. Amen documents at least one instance in which he was convinced that his patient had experienced head trauma, and questioned the patient again and again about possible brain injuries. One must wonder whether the positive results of such focused, repetitive questioning might be evidence of confirmation bias, as a search to confirm the preexisting belief of head trauma could lead to overlooking alternative explanations for a patient’s symptoms.
Another risk factor dwelt upon is exposure to toxins. One toxin Dr. Amen rightly recommends avoiding is tobacco smoke. Yet, his approach to advocate for a tobacco-free lifestyle is somewhat problematic. He lists chemicals contained in tobacco smoke, and then names unpleasant items that share those ingredients, such as paint. This smacks of the same sloppy logic manifested in social media memes decrying the use of vaccines by listing their ingredients alongside scary-sounding products that contain identical ingredients (for example, vaccines contain formaldehyde, which is used to embalm dead bodies!). This is analogous to saying that water is bad for you because it contains hydrogen, which is also an ingredient in atomic bombs.
Dr. Amen makes the blanket recommendation to avoid products containing “chemicals.” This is a difficult recommendation to interpret, since literally all matter is made of chemicals. It seems that Dr. Amen is leaning into the vague idea of a “chemical” as something artificially created in a lab, which must, therefore, be dangerous.
Along these lines, Dr. Amen suggests that if a person doesn’t know what is in a specific food item, it should not be eaten. Although this sounds reasonable on the surface, if people were told the names of the proteins and chemical compounds that make up many naturally occurring plants or meats, they would likely not recognize many of them. Dr. Amen dedicates space to list seemingly benign exposures – such as eating nonorganic produce, using two or more beauty products each day, or touching grocery store receipts – as possible “toxins.” By contrast, there is a certain irony in the absence of any mention of the risks associated with radiation from the SPECT imaging he staunchly advocates for. One potential risk of the book listing so many “toxins” to avoid is that patients could waste valuable time and energy eliminating exposures that pose little or no risk, rather than focusing efforts on well-established treatments.
In light of the observations and critiques offered above, one might come away with the impression that I would not recommend “The End of Mental Illness.” However, although one can nitpick details in the book, some of its bigger ideas make it worth commending to readers. Dr. Amen rightfully emphasizes the need for psychiatrists and patients to think more broadly about mental health issues beyond the use of pills. He justifiably criticizes the “15-minute med check” model of practice and the idea that medications are the end-all, be-all of treatment. He demonstrates an appropriate appreciation for the serious risks of reliance on benzodiazepines.6 Dr. Amen points out important contributions from Viktor Frankl, MD, to the field of psychiatry, which may go overlooked today. He also helpfully points out that bipolar disorder may often be misdiagnosed (although he attributes the misdiagnosis to traumatic brain injury, whereas other psychiatrists might say the misdiagnosis is due to borderline personality disorder).
Much of what Dr. Amen writes is sensible, and psychiatrists would do well to adopt the following steps he advocates for: Taking a comprehensive biopsychosocial-spiritual approach to the assessment and treatment of patients; thinking broadly in their differential diagnoses and not forgetting their medical training; understanding that medication alone is often not sufficient to make lasting, positive change in a person’s life; paying attention to healthy habits such as diet, exercise, sleep, and social activity; and knowing that CBT is a valuable tool that can change lives.
There is much to appreciate in “The End of Mental Illness,” especially the overarching idea that psychiatry isn’t just a symptom checklist and a prescription pad. Rather, achieving mental well-being often requires broader thinking and sustained lifestyle changes.
Although I did not agree with everything in the book, it did cause me to think and reflect on my own practice. I read “The End of Mental Illness” with colleagues in my department, and it stimulated a lively discussion. Isn’t that ultimately what a psychiatrist would want from a book like this – the opportunity to reflect, discuss, and potentially improve one’s own practice?
Dr. Weber is physician lead in the department of psychiatry at Intermountain Healthcare Budge Clinic, Logan (Utah) Psychiatry. He disclosed no relevant financial relationships.
References
1. JAMA Netw Open. 2020;3(12). doi: 10.1001/jamanetworkopen.2020.27909.
2. Curr Opin Psychiatry. 2014;27:358-63.
3. BMJ 2014. doi: 10.1136/bmj.g5205.
4. Am J Psychiatry. 2018 Mar 1;175:232-41.
5. Am J Psychiatry. 2016 Jun 1;173:575-87.
6. Current Psychiatry. 2018 Feb;17(2):22-7.
Daniel G. Amen, MD, is an American psychiatrist well-known for his eponymous clinics, television appearances, and series of books on mental health. One of his latest books, “The End of Mental Illness,” summarizes many of his views on the causes of and treatments for mental illnesses.
Dr. Amen’s approaches – such as his advocacy for the widespread use of single photon emission computed tomography (SPECT) imaging – are somewhat controversial and at times fall outside the mainstream of current psychiatric thought. So does “The End of Mental Illness” contain anything of value to the average practicing psychiatrist? (It should be noted that I listened to this as an audiobook and took notes as I listened. This does limit my ability to directly quote portions of the text, but I believe my notes are reliable.)
He begins the book by pointing out that the term “mental illness” might be better replaced with the term “brain illness.” With this shift in terminology, Dr. Amen introduces a theme that recurs throughout the book: That mental illnesses ultimately stem from various ways in which the brain can be harmed. While the suggested change in terminology might help reduce the stigma associated with psychiatric illnesses, Dr. Amen is surprisingly timid about implementing this term in his own book. He repeatedly refers to “brain health/mental health” issues instead of discarding the “mental” term altogether. Even his BRIGHT MINDS acronym for risk factors for mental illnesses includes the term “mind” instead of “brain.”
Continuing the theme of challenging terminology, Dr. Amen goes on to decry the weaknesses of the DSM system of nosology. This is a valid point, because under the current system, the same patient may receive differing diagnoses depending on which provider is seen and how certain symptoms are interpreted. Yet, here again, Dr. Amen does not seem to adhere to his own advice: He uses DSM terminology throughout the book, speaking of depression, anxiety, bipolar disorder, and ADHD. An oddity (which, admittedly, could have been the audiobook reader’s mistake rather than an error in the original text) is that the DSM is referred to as the “Diagnostic and Structural Manual” rather than the Diagnostic and Statistical Manual. He criticizes the DSM for its imprecision, pointing out the variety of symptom combinations that can produce the same diagnoses and how similar symptoms may overlap between differing diagnoses. Yet, his descriptions of common SPECT patterns (his preferred tool to assist in diagnosis) make it clear that here, too, there is a lot of overlap. As an example, ADHD was associated with at least three of the imaging patterns he described. It is also somewhat ironic how Dr. Amen obliquely criticizes the American Psychiatric Association for profiting from the use of the DSM, when SPECT imaging is expensive and profits his own organization.
Dr. Amen repeatedly asserts that psychiatry is unique among medical specialties for making diagnoses based on symptom clusters rather than direct visualization of the affected organ. Yet, psychiatry is not, in fact, unique in making diagnoses in this way. Some examples of diagnoses based on symptom clusters from other medical specialties are systemic lupus erythematosus, fibromyalgia, and chronic fatigue syndrome. Although he asserts that SPECT imaging better demonstrates the root cause of mental illnesses, it is unclear from his book whether this is actually the case.
The descriptions for the ways in which Dr. Amen uses SPECT (which, admittedly, are vague and presumably simplified for a general audience) suggest that he has made observations correlating specific imaging patterns with certain emotional/behavioral outcomes. However, the imaging patterns he describes in the book can be interpreted to represent multiple different mental conditions, making it clear that SPECT is not a laserlike diagnostic tool that produces a single, indisputable diagnosis. Accuracy with SPECT seems especially questionable in light of two case examples he shares where brain imaging was interpreted as representing illness, but the patients were not demonstrating any signs of mental dysfunction. In one case, Dr. Amen opined that the patient’s vibrant spiritual life “overrode” the sick brain, but if this is true,
Patient testimonials are provided, asserting that SPECT imaging helped them know “exactly” what treatment would help them. One cannot help but wonder whether part of the benefit of SPECT imaging is a placebo effect, boosting the confidence of patients that the treatment they are receiving is personalized and scientifically sound. A similar trend is currently seen more broadly in psychiatry with the widespread promotion of pharmacogenetic testing. Such testing may bolster patient confidence in their medication, but its value in improving patient outcomes has not been established.1
Dr. Amen outlines a brief history of mental health care, including differing approaches and therapies from the time of Sigmund Freud up to the present. His outline is somewhat critical of the perceived shortcomings of his psychiatric forebears, yet this seems entirely unnecessary. All scientific disciplines must start somewhere and build from limited knowledge to greater. Is it necessary to belittle Freud for not being able to do SPECT imaging in the 1800s?
Interestingly, Dr. Amen leaves cognitive-behavioral therapy (CBT), a landmark, evidence-based form of psychotherapy, out of his overview of the history of psychiatry. He does go on to mention CBT as part of the treatment offerings of the Amen Clinics, which could leave the lay reader with the incorrect impression that CBT is a treatment unique to Amen Clinics. Similarly, at one point Dr. Amen writes about “what I call automatic negative thoughts.” This phrasing could confuse readers who might not know that automatic thoughts are a concept endemic to CBT.
Dr. Amen writes repeatedly about the Amen Clinics 4 Circles, four key areas of life that can contribute to mental health. These areas are biological, psychological, social, and spiritual. While Amen Clinics may have come up with the term “4 Circles,” the biopsychosocial model of understanding illness was developed by George Engel, MD, in 1977, and current discussions of this model frequently incorporate a spiritual dimension as well.2
Dr. Amen’s writing at times mischaracterizes psychotropic medications in unhelpful ways. He speaks of psychotropic medications generally as being addictive. While this is certainly true for stimulants and benzodiazepines, most would agree that this does not apply to many other commonly used medications in psychiatry, including selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants, antipsychotics, and mood stabilizers. He also paints with a broad brush when he states that anxiety medications can cause dementia. A concerning link has been demonstrated between benzodiazepine use and dementia,3 but SSRIs (which are considered first-line medications for anxiety) are not known to cause dementia and may actually delay progression from mild cognitive impairment to Alzheimer’s dementia.4 His mention of medication use affecting a patient’s insurability could have the unfortunate effect of scaring away suffering individuals from seeking help. The one category of psychiatric medication he does not seem concerned about is psychostimulants, which is odd – given the addictive, cardiovascular, and other risks associated with that medication class.
In contrast to his skepticism regarding many psychotropic medications, Dr. Amen expresses significant enthusiasm regarding nutraceutical use. While there has been research in this area supporting a role for some nutraceutical interventions, there is still a need for more rigorous studies.5 To support his endorsement of natural remedies, Dr. Amen mentions that Hippocrates recommended herbs and spices for many health conditions. But Hippocrates lived more than 2,000 years ago, and the state of medicine has advanced significantly since then.
Dr. Amen also mentions that 80% of the developing world relies upon natural or herbal remedies as the primary source of medicine. While he frames this statement as supporting his endorsement of such remedies, it could conversely be said that this is evidence of the need to make pharmacological interventions more widely available in the developing world.
Much of “The End of Mental Illness” is dedicated to reviewing specific risk factors that could cause harm to a person’s mental well-being. One example is head trauma. Dr. Amen documents at least one instance in which he was convinced that his patient had experienced head trauma, and questioned the patient again and again about possible brain injuries. One must wonder whether the positive results of such focused, repetitive questioning might be evidence of confirmation bias, as a search to confirm the preexisting belief of head trauma could lead to overlooking alternative explanations for a patient’s symptoms.
Another risk factor dwelt upon is exposure to toxins. One toxin Dr. Amen rightly recommends avoiding is tobacco smoke. Yet, his approach to advocate for a tobacco-free lifestyle is somewhat problematic. He lists chemicals contained in tobacco smoke, and then names unpleasant items that share those ingredients, such as paint. This smacks of the same sloppy logic manifested in social media memes decrying the use of vaccines by listing their ingredients alongside scary-sounding products that contain identical ingredients (for example, vaccines contain formaldehyde, which is used to embalm dead bodies!). This is analogous to saying that water is bad for you because it contains hydrogen, which is also an ingredient in atomic bombs.
Dr. Amen makes the blanket recommendation to avoid products containing “chemicals.” This is a difficult recommendation to interpret, since literally all matter is made of chemicals. It seems that Dr. Amen is leaning into the vague idea of a “chemical” as something artificially created in a lab, which must, therefore, be dangerous.
Along these lines, Dr. Amen suggests that if a person doesn’t know what is in a specific food item, it should not be eaten. Although this sounds reasonable on the surface, if people were told the names of the proteins and chemical compounds that make up many naturally occurring plants or meats, they would likely not recognize many of them. Dr. Amen dedicates space to list seemingly benign exposures – such as eating nonorganic produce, using two or more beauty products each day, or touching grocery store receipts – as possible “toxins.” By contrast, there is a certain irony in the absence of any mention of the risks associated with radiation from the SPECT imaging he staunchly advocates for. One potential risk of the book listing so many “toxins” to avoid is that patients could waste valuable time and energy eliminating exposures that pose little or no risk, rather than focusing efforts on well-established treatments.
In light of the observations and critiques offered above, one might come away with the impression that I would not recommend “The End of Mental Illness.” However, although one can nitpick details in the book, some of its bigger ideas make it worth commending to readers. Dr. Amen rightfully emphasizes the need for psychiatrists and patients to think more broadly about mental health issues beyond the use of pills. He justifiably criticizes the “15-minute med check” model of practice and the idea that medications are the end-all, be-all of treatment. He demonstrates an appropriate appreciation for the serious risks of reliance on benzodiazepines.6 Dr. Amen points out important contributions from Viktor Frankl, MD, to the field of psychiatry, which may go overlooked today. He also helpfully points out that bipolar disorder may often be misdiagnosed (although he attributes the misdiagnosis to traumatic brain injury, whereas other psychiatrists might say the misdiagnosis is due to borderline personality disorder).
Much of what Dr. Amen writes is sensible, and psychiatrists would do well to adopt the following steps he advocates for: Taking a comprehensive biopsychosocial-spiritual approach to the assessment and treatment of patients; thinking broadly in their differential diagnoses and not forgetting their medical training; understanding that medication alone is often not sufficient to make lasting, positive change in a person’s life; paying attention to healthy habits such as diet, exercise, sleep, and social activity; and knowing that CBT is a valuable tool that can change lives.
There is much to appreciate in “The End of Mental Illness,” especially the overarching idea that psychiatry isn’t just a symptom checklist and a prescription pad. Rather, achieving mental well-being often requires broader thinking and sustained lifestyle changes.
Although I did not agree with everything in the book, it did cause me to think and reflect on my own practice. I read “The End of Mental Illness” with colleagues in my department, and it stimulated a lively discussion. Isn’t that ultimately what a psychiatrist would want from a book like this – the opportunity to reflect, discuss, and potentially improve one’s own practice?
Dr. Weber is physician lead in the department of psychiatry at Intermountain Healthcare Budge Clinic, Logan (Utah) Psychiatry. He disclosed no relevant financial relationships.
References
1. JAMA Netw Open. 2020;3(12). doi: 10.1001/jamanetworkopen.2020.27909.
2. Curr Opin Psychiatry. 2014;27:358-63.
3. BMJ 2014. doi: 10.1136/bmj.g5205.
4. Am J Psychiatry. 2018 Mar 1;175:232-41.
5. Am J Psychiatry. 2016 Jun 1;173:575-87.
6. Current Psychiatry. 2018 Feb;17(2):22-7.
Steroid-induced psychiatric symptoms: What you need to know
Ms. N, age 30, presents to the emergency department for altered mental status, insomnia, and behavioral changes, which she has experienced for 1 week. On evaluation, she grabs a clinician’s hand and details her business ideas and life story with no prompting. Ms. N’s mental status examination is significant for hyperverbal speech with increased rate and volume; tangential thought process; and bright, expanded affect.
One week earlier, Ms. N was hospitalized for sudden-onset chest pain, weakness, and dizziness. She received 45 minutes of cardiopulmonary resuscitation prior to presentation and was found to have a ST-segment elevation myocardial infarction that required emergent left anterior descending coronary artery and right coronary artery percutaneous coronary intervention to place drug-eluting stents. Her recovery was complicated by acute cardiogenic shock, pulmonary edema, and hypoxic respiratory failure. Subsequently, she was intubated, admitted to the ICU, and received high-dose corticosteroids, including IV methylprednisolone, 40 mg every 12 hours, which was tapered prior to discharge. Her husband reports that since Ms. N came home, she has been more talkative and irritable, ruminating about past events, unable to sleep (<1 hour/night), and crying frequently. She has also been endorsing visual and auditory hallucinations, with increased praying and listening to religious music.
The frequent clinical use of steroids necessitates an understanding of these medications’ various adverse effects. The manifestations of steroid-induced psychiatric symptoms are broad and can involve affective, behavioral, and cognitive domains. While the current mechanism is unknown, this phenomenon may be related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity, as well as effects on brain regions such as the hippocampus and amygdala. The best interventions for steroid-induced psychiatric symptoms are awareness and early diagnosis. There are no FDA-approved treatments for steroid-induced psychiatric symptoms; initial measures should include tapering or discontinuing corticosteroids.
In this article, we review the literature on the incidence, characteristics, differential diagnoses, proposed mechanism, risk factors, and proposed treatments of steroid-induced psychiatric symptoms.
A wide range of presentations
Steroid use has increased over the past 2 decades, with 10% of medical and surgical inpatients and 1% to 3% of the general population taking long-term glucocorticoids.1 Even with topical application, steroid therapy is often systemically absorbed, and thus may lead to steroid-induced psychiatric symptoms. The incidence of steroid-induced psychiatric symptoms is difficult to assess because there can be a wide range of reactions that are dose- and time-related. Three reviews of a total of 122 cases reports found that an estimated 5% of patients treated with steroids experience severe psychiatric reactions.1-3
Steroid-induced psychopathology can include mood, behavioral, and/or cognitive impairments. Mania/hypomania is the most common overall psychiatric symptom; the most common mood manifestations are anxiety and depression.4,5 Other possible steroid-induced symptoms include psychosis, dementia, panic disorder, delirium, suicidal thinking and behavior, aggressive behavior, insomnia, agitation, depersonalization, and euphoria.5 The most common cognitive impairment is verbal or declarative memory deficit; others include distractibility and deficits in attention and psychomotor speed.5 These psychiatric symptoms can have a rapid onset, possibly within hours of starting steroids.1 However, studies have reported a median time to onset of 11.5 days; 39% of cases had onset during the first week and 62% within 2 weeks.3,6 After reducing or stopping the steroid, it may take days to weeks before symptoms start to subside.2
What to consider in the differential Dx
Psychiatric symptoms that are induced by steroids can mimic metabolic, neurologic, or toxic disorders. Other factors to consider include drug withdrawal/intoxication, infections, and paraneoplastic syndromes.4,5 Although there is no reported correlation between the location of neurologic lesions and the development of specific psychiatric symptoms, manic symptoms appear most commonly with lesions in the right frontal lobe. 4 Other factors to note include the presence of new-onset psychiatric illnesses such as bipolar, mood, or thought disorders,4 as well as psychosocial stressors that might be contributing to the patient’s presentation.5
Continue to: Proposed mechanisms
Proposed mechanisms
Although the exact mechanism by which steroids induce psychiatric symptoms is unknown, several mechanisms have been proposed. One hypothesis is that steroid-induced psychopathology is related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity.4,5,7 This may explain why many patients with major depressive disorder have elevated cortisol production and/or lack of suppression of cortisol secretion during a dexamethasone stimulation test, and why approximately one-half of patients with Cushing’s disease experience depressive symptoms.8 This is also likely why antipsychotics, which typically reduce cortisol, are efficacious treatments for some steroid-induced psychiatric symptoms.9
Cognitive impairments from steroid use may be related to these agents’ effects on certain brain regions. One such area is the hippocampus, an important mediator in the creation and maintenance of episodic and declarative memories.5,8,9 Acute glucocorticoid use is associated with decreased activity in the left hippocampus, reduced hippocampal glucose metabolism, and reduced cerebral blood flow in the posterior medial temporal lobe.10 Long-term glucocorticoid exposure is associated with smaller hippocampal volume and lower levels of temporal lobe N-acetylaspartate, a marker of neuronal viability.10 Because working memory depends on the prefrontal cortex and declarative memory relies on the hippocampus, deficits in these functions can be attributed to the effect of prolonged glucocorticoid exposure on glucocorticoid or mineralocorticoid receptors in the hippocampus, reduction of hippocampal volume, or elevated glutamate accumulation in that area.11 In addition, high cortisol levels inhibit brain-derived neurotrophic factor, which plays a crucial role in maintaining neural architecture in key brain regions such as the hippocampus and prefrontal cortex.11 There is also a correlation between the duration of prednisone treatment and atrophy of the right amygdala, which is an important regulator of mood and anxiety.11 Both the hippocampus and amygdala have dense collections of glucocorticoid receptors. This may explain why patients who receive high-dose corticosteroids can have reversible atrophy in the hypothalamus and amygdala, leading to deficits in emotional learning and the stress response.
Factors that increase risk
Several factors can increase the risk of steroid-induced psychopathology. The most significant is the dose; higher doses are more likely to produce psychiatric symptoms.1,5 Concurrent use of drugs that increase circulating levels of corticosteroids, such as inhibitors of the cytochrome P450 (CYP) enzyme (eg, clarithromycin), also increases the likelihood of developing psychiatric symptoms.1,5 Risk is also increased in patients with liver or renal dysfunction.1 Cerebral spinal fluid/serum albumin ratio, a marker of blood-brain barrier damage, and low serum complement levels were also reported to be independent risk factors,12 with the thought that increased permeability of the blood-brain barrier may allow hydrophobic steroid molecules to more easily penetrate the CNS, leading to increased neuropsychiatric effects. Hypoalbuminemia is another reported risk factor, perhaps because lower levels of serum albumin are related to higher levels of free and active glucocorticoids, which are normally inactive when bound to albumin.13 There also appears to be an increased prevalence of steroid-induced psychopathology in women, perhaps due to greater propensity in women to seek medical care or a higher prevalence of women with medical disorders that are treated with steroids.5 A previous history of psychiatric disorders may not increase risk.5
Several methods for reducing risk have been proposed, including using a divided-dosing regimens that may lower peak steroid plasma concentrations.13,14 However, the best prevention of steroid-induced psychiatric symptoms are awareness, early diagnosis, and intervention. Studies have suggested that N-methyl-
Treatment options
There are no FDA-approved medications for managing steroid-induced psychiatric symptoms.1,16 Treatment is based on evidence from case reports and a few small case series (Table2-5,17,18).

Continue to: When possible, initial treatment...
When possible, initial treatment should include discontinuing or tapering corticosteroids to <40 mg/d of prednisone-equivalent.1,4,10,18 Most studies have reported rapid reversal of deficits in declarative memory and of hippocampal volume loss once corticosteroids were tapered and discontinued.1,18 One study reported that >90% of patients recovered within 6 weeks, with patients with delirium recovering more quickly (mean: 5.4 days) than those with depression, mania, or psychosis (mean: 19.3 days).3 Another found that the vast majority (92%) of patients treated only with a steroid taper achieved clinical recovery, and 84% recovered with administration of antipsychotics without a steroid taper.3 In this study, all patients who received electroconvulsive therapy (ECT) recovered, as did those who received a steroid taper plus lithium or antipsychotics. Steroid tapering regimens are especially important for patients who have received long-term glucocorticoid treatment. Patients need to be closely monitored for signs of new or increased depression, delirium, or confusion during the taper. If these symptoms occur, the patient should be checked for adrenocortical insufficiency, which can be resolved by re-administering or increasing the dosage of the glucocorticoid.10
Mania. The treatment of mania/hypomania includes mood stabilizers (valproate, lithium, lamotrigine) and antipsychotics (quetiapine, olanzapine, haloperidol).2,4,5,10,14,18 Valproate has been reported to be an effective prophylactic of corticosteroid-induced mania,2 perhaps because it dampens neuronal hyperexcitability by attenuating NMDA receptors, blocking voltage-dependent sodium channels, and inhibiting the synthesis of cortical GABAergic steroids. Starting valproate while continuing corticosteroids (if necessary) may help lessen mania.2 Benzodiazepines also may be useful on a short-term basis.
Depression. Steroid-induced depression may be treated with sertraline or other first-line antidepressants.5,14 Consider ECT for patients with severe depression. Support for the use of antipsychotic medications stems from studies that reported steroids’ role in disrupting dopamine and 5HT2 activity. Lithium also has been used successfully to manage and prevent glucocorticoid-associated affective disorder.10,18 It can be used alone or in combination with selective serotonin reuptake inhibitors to alleviate depressive symptoms.10 Tricyclic antidepressants are generally avoided because their anticholinergic effects can exacerbate or worsen delirium.18 In general, ECT is an effective treatment for persistent and/or unresponsive steroid-induced depression,2,10 but may be difficult to use in patients with serious medical illnesses.
Agitation. Medications that have been proposed for treating steroid-induced agitation include benzodiazepines, haloperidol, and second-generation antipsychotics.5,17
Other considerations. Clinicians, patients, and families should discuss in detail the risks of steroid-induced psychiatric symptoms so an early diagnosis and appropriate intervention can be implemented. Before starting steroids, it is important to review the patient’s current medication list to ensure that steroid treatment is indicated, and to check for potential drug–drug interactions. In addition, the medical condition that is being treated with steroids also needs to be carefully reviewed, because certain illnesses are associated with the development of psychiatric symptoms. 5,10
Continue to: Young children...
Young children (age <6) and older adults appear to be at greater risk for cognitive and memory disturbances from steroid use.10 In addition, patients have individual levels of susceptibility to steroid-induced psychiatric symptoms that can vary over time. The risk for adverse effects may be elevated based on response to previous courses of glucocorticoid treatment.10 While gender, age, dosage, and duration of treatment influence risk, it is not possible to predict which patients will experience psychiatric effects during a given course of glucocorticoid therapy. Therefore, all patients should be considered to have the potential of developing such effects, and should be monitored during glucocorticoid treatment and withdrawal.
Goals for future research
To help reduce the severity of and cost associated with steroid-induced psychiatric symptoms,5,14 future studies should focus on controlled trials of preventative strategies. In particular, recent advances in genetic mapping may help identify involvement of certain genes or polymorphisms.5 Because current guidelines for the prevention and treatment of steroid-induced psychiatric symptoms are not evidence-based, controlled clinical trials are needed to elucidate the optimal management of such symptoms. There is much interplay between many of the proposed mechanisms of steroid-induced psychiatric symptoms, and future studies can help uncover a deeper understanding of the intricacies of this phenomenon.
CASE CONTINUED
Mrs. N is admitted for altered mental status. Medical workup includes MRI of the brain, MRI of the neck, cardiac echocardiogram, and EEG. There is no evidence of acute structural pathology. She is started on olanzapine, 10 mg/d at bedtime for manic and psychotic symptoms, and is discharged after 5 days. After 1 month, the outpatient psychiatrist gradually decreases and discontinues olanzapine as Mrs. N steadily returns to baseline. One year after discharge, Mrs. N continues to report resolution of her manic and psychotic symptoms.
Bottom Line
Steroids can induce a wide range of psychiatric symptoms, including mania/ hypomania, anxiety, and depression. Initial treatment typically includes tapering or discontinuing the steroid when possible. Other proposed treatments include certain antipsychotics, antidepressants, and other psychotropics, but the supporting evidence is largely anecdotal or based on case studies. Additional research is needed to elucidate the mechanism and treatment recommendations.
Related Resources
- Janes M, Kuster S, Goldson TM, et al. Steroid-induced psychosis. Proc (Bayl Univ Med Cent). 2019;32(4):614-615.
- Mayo Clinic. Prednisone and other corticosteroids. https://www.mayoclinic.org/steroids/art-20045692
Drug Brand Names
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Methylprednisolone injection • Solu-Medrol
Olanzapine • Zyprexa
Paroxetine • Paxil
Phenytoin • Dilantin
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Valproate • Depakote
1. Dubovsky AN, Arvikar S, Stern TA, et al. The neuropsychiatric complications of glucocorticoid use: steroid psychosis revisited. Psychosomatics. 2012;53(2):103-115.
2. Roxanas MG, Hunt GE. Rapid reversal of corticosteroid-induced mania with sodium valproate: a case series of 20 patients. Psychosomatics. 2012;53(6):575-581.
3. Lewis DA, Smith RE. Steroid‐induced psychiatric syndromes. A report of 14 cases and a review of the literature. J Affect Disord. 1983;5(4):319-332.
4. Warren KN, Katakam J, Espiridion ED. Acute-onset mania in a patient with non-small cell lung cancer. Cureus. 2019;11(8):e5436.
5. Kenna HA, Poon AW, de los Angeles CP, et al. Psychiatric complications of treatment with corticosteroids: review with case report. Psychiatry Clin Neurosci. 2011;65(6):549-560.
6. Ling MH, Perry PJ, Tsuang MT. Side effects of corticosteroid therapy. Psychiatric aspects. Arch Gen. Psychiatry. 1981;38(4):471-477.
7. Ularntinon S, Tzuang D, Dahl G, et al. Concurrent treatment of steroid-related mood and psychotic symptoms with risperidone. Pediatrics. 2010;125(5):e1241-e1245.
8. Pokladinkova J, Meyboom RH, Vlcek J, et al. Intranasally administered corticosteroids and neuropsychiatric disturbances: a review of the international pharmacovigilance programme of the World Health Organization. Ann Allergy Asthma Immunol. 2008;101(1):67-73.
9. Walker EF, Trotman HD, Pearce BD, et al. Cortisol levels and risk for psychosis: initial findings from the North American prodrome longitudinal study. Biol Psychiatry. 2013;74(6):410-417.
10. Wolkowitz OM, Reus UI. Treatment of depression with antiglucocorticoid drugs. Psychosom Med. 1999;61(5):698-711.
11. Judd LL, Schettler PJ, Brown ES, et al. Adverse consequences of glucocorticoid medication: psychological, cognitive, and behavioral effects. Am J Psychiatry. 2014;171(10):1045-1051.
12. Appenzeller S, Cendes F, Costallat LT. Acute psychosis in systemic lupus erythematosus. Rheumatol Int. 2008;28(3):237-243.
13. Glynne-Jones R, Vernon CC, Bell G. Is steroid psychosis preventable by divided doses? Lancet. 1986;2(8520):1404.
14. Ismail MF, Lavelle C, Cassidy EM. Steroid-induced mental disorders in cancer patients: a systematic review. Future Oncol. 2017;13(29):2719-2731.
15. Magariños AM, McEwen BS. Stress-induced atrophy of apical dendrites of hippocampal CA3c neurons: involvement of glucocorticoid secretion and excitatory amino acid receptors. Neuroscience. 1995;69(1):89-98.
16. Brown BS, Stuard G, Liggin JDM, et al. Effect of phenytoin on mood and declarative memory during prescription corticosteroid therapy. Biol Psychiatry. 2005;57(5):543-548.
17. Desai S, Khanani S, Shad MU, et al. Attenutation of amygdala atrophy with lamotrigine in patients receiving corticosteroid therapy. J Clin Psychopharmacol. 2009;29(3):284-287.
18. Gable M, Depry D. Sustained corticosteroid-induced mania and psychosis despite cessation: a case study and brief literature review. Int J Psychiatry Med. 2015;50(4):398-404.
Ms. N, age 30, presents to the emergency department for altered mental status, insomnia, and behavioral changes, which she has experienced for 1 week. On evaluation, she grabs a clinician’s hand and details her business ideas and life story with no prompting. Ms. N’s mental status examination is significant for hyperverbal speech with increased rate and volume; tangential thought process; and bright, expanded affect.
One week earlier, Ms. N was hospitalized for sudden-onset chest pain, weakness, and dizziness. She received 45 minutes of cardiopulmonary resuscitation prior to presentation and was found to have a ST-segment elevation myocardial infarction that required emergent left anterior descending coronary artery and right coronary artery percutaneous coronary intervention to place drug-eluting stents. Her recovery was complicated by acute cardiogenic shock, pulmonary edema, and hypoxic respiratory failure. Subsequently, she was intubated, admitted to the ICU, and received high-dose corticosteroids, including IV methylprednisolone, 40 mg every 12 hours, which was tapered prior to discharge. Her husband reports that since Ms. N came home, she has been more talkative and irritable, ruminating about past events, unable to sleep (<1 hour/night), and crying frequently. She has also been endorsing visual and auditory hallucinations, with increased praying and listening to religious music.
The frequent clinical use of steroids necessitates an understanding of these medications’ various adverse effects. The manifestations of steroid-induced psychiatric symptoms are broad and can involve affective, behavioral, and cognitive domains. While the current mechanism is unknown, this phenomenon may be related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity, as well as effects on brain regions such as the hippocampus and amygdala. The best interventions for steroid-induced psychiatric symptoms are awareness and early diagnosis. There are no FDA-approved treatments for steroid-induced psychiatric symptoms; initial measures should include tapering or discontinuing corticosteroids.
In this article, we review the literature on the incidence, characteristics, differential diagnoses, proposed mechanism, risk factors, and proposed treatments of steroid-induced psychiatric symptoms.
A wide range of presentations
Steroid use has increased over the past 2 decades, with 10% of medical and surgical inpatients and 1% to 3% of the general population taking long-term glucocorticoids.1 Even with topical application, steroid therapy is often systemically absorbed, and thus may lead to steroid-induced psychiatric symptoms. The incidence of steroid-induced psychiatric symptoms is difficult to assess because there can be a wide range of reactions that are dose- and time-related. Three reviews of a total of 122 cases reports found that an estimated 5% of patients treated with steroids experience severe psychiatric reactions.1-3
Steroid-induced psychopathology can include mood, behavioral, and/or cognitive impairments. Mania/hypomania is the most common overall psychiatric symptom; the most common mood manifestations are anxiety and depression.4,5 Other possible steroid-induced symptoms include psychosis, dementia, panic disorder, delirium, suicidal thinking and behavior, aggressive behavior, insomnia, agitation, depersonalization, and euphoria.5 The most common cognitive impairment is verbal or declarative memory deficit; others include distractibility and deficits in attention and psychomotor speed.5 These psychiatric symptoms can have a rapid onset, possibly within hours of starting steroids.1 However, studies have reported a median time to onset of 11.5 days; 39% of cases had onset during the first week and 62% within 2 weeks.3,6 After reducing or stopping the steroid, it may take days to weeks before symptoms start to subside.2
What to consider in the differential Dx
Psychiatric symptoms that are induced by steroids can mimic metabolic, neurologic, or toxic disorders. Other factors to consider include drug withdrawal/intoxication, infections, and paraneoplastic syndromes.4,5 Although there is no reported correlation between the location of neurologic lesions and the development of specific psychiatric symptoms, manic symptoms appear most commonly with lesions in the right frontal lobe. 4 Other factors to note include the presence of new-onset psychiatric illnesses such as bipolar, mood, or thought disorders,4 as well as psychosocial stressors that might be contributing to the patient’s presentation.5
Continue to: Proposed mechanisms
Proposed mechanisms
Although the exact mechanism by which steroids induce psychiatric symptoms is unknown, several mechanisms have been proposed. One hypothesis is that steroid-induced psychopathology is related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity.4,5,7 This may explain why many patients with major depressive disorder have elevated cortisol production and/or lack of suppression of cortisol secretion during a dexamethasone stimulation test, and why approximately one-half of patients with Cushing’s disease experience depressive symptoms.8 This is also likely why antipsychotics, which typically reduce cortisol, are efficacious treatments for some steroid-induced psychiatric symptoms.9
Cognitive impairments from steroid use may be related to these agents’ effects on certain brain regions. One such area is the hippocampus, an important mediator in the creation and maintenance of episodic and declarative memories.5,8,9 Acute glucocorticoid use is associated with decreased activity in the left hippocampus, reduced hippocampal glucose metabolism, and reduced cerebral blood flow in the posterior medial temporal lobe.10 Long-term glucocorticoid exposure is associated with smaller hippocampal volume and lower levels of temporal lobe N-acetylaspartate, a marker of neuronal viability.10 Because working memory depends on the prefrontal cortex and declarative memory relies on the hippocampus, deficits in these functions can be attributed to the effect of prolonged glucocorticoid exposure on glucocorticoid or mineralocorticoid receptors in the hippocampus, reduction of hippocampal volume, or elevated glutamate accumulation in that area.11 In addition, high cortisol levels inhibit brain-derived neurotrophic factor, which plays a crucial role in maintaining neural architecture in key brain regions such as the hippocampus and prefrontal cortex.11 There is also a correlation between the duration of prednisone treatment and atrophy of the right amygdala, which is an important regulator of mood and anxiety.11 Both the hippocampus and amygdala have dense collections of glucocorticoid receptors. This may explain why patients who receive high-dose corticosteroids can have reversible atrophy in the hypothalamus and amygdala, leading to deficits in emotional learning and the stress response.
Factors that increase risk
Several factors can increase the risk of steroid-induced psychopathology. The most significant is the dose; higher doses are more likely to produce psychiatric symptoms.1,5 Concurrent use of drugs that increase circulating levels of corticosteroids, such as inhibitors of the cytochrome P450 (CYP) enzyme (eg, clarithromycin), also increases the likelihood of developing psychiatric symptoms.1,5 Risk is also increased in patients with liver or renal dysfunction.1 Cerebral spinal fluid/serum albumin ratio, a marker of blood-brain barrier damage, and low serum complement levels were also reported to be independent risk factors,12 with the thought that increased permeability of the blood-brain barrier may allow hydrophobic steroid molecules to more easily penetrate the CNS, leading to increased neuropsychiatric effects. Hypoalbuminemia is another reported risk factor, perhaps because lower levels of serum albumin are related to higher levels of free and active glucocorticoids, which are normally inactive when bound to albumin.13 There also appears to be an increased prevalence of steroid-induced psychopathology in women, perhaps due to greater propensity in women to seek medical care or a higher prevalence of women with medical disorders that are treated with steroids.5 A previous history of psychiatric disorders may not increase risk.5
Several methods for reducing risk have been proposed, including using a divided-dosing regimens that may lower peak steroid plasma concentrations.13,14 However, the best prevention of steroid-induced psychiatric symptoms are awareness, early diagnosis, and intervention. Studies have suggested that N-methyl-
Treatment options
There are no FDA-approved medications for managing steroid-induced psychiatric symptoms.1,16 Treatment is based on evidence from case reports and a few small case series (Table2-5,17,18).

Continue to: When possible, initial treatment...
When possible, initial treatment should include discontinuing or tapering corticosteroids to <40 mg/d of prednisone-equivalent.1,4,10,18 Most studies have reported rapid reversal of deficits in declarative memory and of hippocampal volume loss once corticosteroids were tapered and discontinued.1,18 One study reported that >90% of patients recovered within 6 weeks, with patients with delirium recovering more quickly (mean: 5.4 days) than those with depression, mania, or psychosis (mean: 19.3 days).3 Another found that the vast majority (92%) of patients treated only with a steroid taper achieved clinical recovery, and 84% recovered with administration of antipsychotics without a steroid taper.3 In this study, all patients who received electroconvulsive therapy (ECT) recovered, as did those who received a steroid taper plus lithium or antipsychotics. Steroid tapering regimens are especially important for patients who have received long-term glucocorticoid treatment. Patients need to be closely monitored for signs of new or increased depression, delirium, or confusion during the taper. If these symptoms occur, the patient should be checked for adrenocortical insufficiency, which can be resolved by re-administering or increasing the dosage of the glucocorticoid.10
Mania. The treatment of mania/hypomania includes mood stabilizers (valproate, lithium, lamotrigine) and antipsychotics (quetiapine, olanzapine, haloperidol).2,4,5,10,14,18 Valproate has been reported to be an effective prophylactic of corticosteroid-induced mania,2 perhaps because it dampens neuronal hyperexcitability by attenuating NMDA receptors, blocking voltage-dependent sodium channels, and inhibiting the synthesis of cortical GABAergic steroids. Starting valproate while continuing corticosteroids (if necessary) may help lessen mania.2 Benzodiazepines also may be useful on a short-term basis.
Depression. Steroid-induced depression may be treated with sertraline or other first-line antidepressants.5,14 Consider ECT for patients with severe depression. Support for the use of antipsychotic medications stems from studies that reported steroids’ role in disrupting dopamine and 5HT2 activity. Lithium also has been used successfully to manage and prevent glucocorticoid-associated affective disorder.10,18 It can be used alone or in combination with selective serotonin reuptake inhibitors to alleviate depressive symptoms.10 Tricyclic antidepressants are generally avoided because their anticholinergic effects can exacerbate or worsen delirium.18 In general, ECT is an effective treatment for persistent and/or unresponsive steroid-induced depression,2,10 but may be difficult to use in patients with serious medical illnesses.
Agitation. Medications that have been proposed for treating steroid-induced agitation include benzodiazepines, haloperidol, and second-generation antipsychotics.5,17
Other considerations. Clinicians, patients, and families should discuss in detail the risks of steroid-induced psychiatric symptoms so an early diagnosis and appropriate intervention can be implemented. Before starting steroids, it is important to review the patient’s current medication list to ensure that steroid treatment is indicated, and to check for potential drug–drug interactions. In addition, the medical condition that is being treated with steroids also needs to be carefully reviewed, because certain illnesses are associated with the development of psychiatric symptoms. 5,10
Continue to: Young children...
Young children (age <6) and older adults appear to be at greater risk for cognitive and memory disturbances from steroid use.10 In addition, patients have individual levels of susceptibility to steroid-induced psychiatric symptoms that can vary over time. The risk for adverse effects may be elevated based on response to previous courses of glucocorticoid treatment.10 While gender, age, dosage, and duration of treatment influence risk, it is not possible to predict which patients will experience psychiatric effects during a given course of glucocorticoid therapy. Therefore, all patients should be considered to have the potential of developing such effects, and should be monitored during glucocorticoid treatment and withdrawal.
Goals for future research
To help reduce the severity of and cost associated with steroid-induced psychiatric symptoms,5,14 future studies should focus on controlled trials of preventative strategies. In particular, recent advances in genetic mapping may help identify involvement of certain genes or polymorphisms.5 Because current guidelines for the prevention and treatment of steroid-induced psychiatric symptoms are not evidence-based, controlled clinical trials are needed to elucidate the optimal management of such symptoms. There is much interplay between many of the proposed mechanisms of steroid-induced psychiatric symptoms, and future studies can help uncover a deeper understanding of the intricacies of this phenomenon.
CASE CONTINUED
Mrs. N is admitted for altered mental status. Medical workup includes MRI of the brain, MRI of the neck, cardiac echocardiogram, and EEG. There is no evidence of acute structural pathology. She is started on olanzapine, 10 mg/d at bedtime for manic and psychotic symptoms, and is discharged after 5 days. After 1 month, the outpatient psychiatrist gradually decreases and discontinues olanzapine as Mrs. N steadily returns to baseline. One year after discharge, Mrs. N continues to report resolution of her manic and psychotic symptoms.
Bottom Line
Steroids can induce a wide range of psychiatric symptoms, including mania/ hypomania, anxiety, and depression. Initial treatment typically includes tapering or discontinuing the steroid when possible. Other proposed treatments include certain antipsychotics, antidepressants, and other psychotropics, but the supporting evidence is largely anecdotal or based on case studies. Additional research is needed to elucidate the mechanism and treatment recommendations.
Related Resources
- Janes M, Kuster S, Goldson TM, et al. Steroid-induced psychosis. Proc (Bayl Univ Med Cent). 2019;32(4):614-615.
- Mayo Clinic. Prednisone and other corticosteroids. https://www.mayoclinic.org/steroids/art-20045692
Drug Brand Names
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Methylprednisolone injection • Solu-Medrol
Olanzapine • Zyprexa
Paroxetine • Paxil
Phenytoin • Dilantin
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Valproate • Depakote
Ms. N, age 30, presents to the emergency department for altered mental status, insomnia, and behavioral changes, which she has experienced for 1 week. On evaluation, she grabs a clinician’s hand and details her business ideas and life story with no prompting. Ms. N’s mental status examination is significant for hyperverbal speech with increased rate and volume; tangential thought process; and bright, expanded affect.
One week earlier, Ms. N was hospitalized for sudden-onset chest pain, weakness, and dizziness. She received 45 minutes of cardiopulmonary resuscitation prior to presentation and was found to have a ST-segment elevation myocardial infarction that required emergent left anterior descending coronary artery and right coronary artery percutaneous coronary intervention to place drug-eluting stents. Her recovery was complicated by acute cardiogenic shock, pulmonary edema, and hypoxic respiratory failure. Subsequently, she was intubated, admitted to the ICU, and received high-dose corticosteroids, including IV methylprednisolone, 40 mg every 12 hours, which was tapered prior to discharge. Her husband reports that since Ms. N came home, she has been more talkative and irritable, ruminating about past events, unable to sleep (<1 hour/night), and crying frequently. She has also been endorsing visual and auditory hallucinations, with increased praying and listening to religious music.
The frequent clinical use of steroids necessitates an understanding of these medications’ various adverse effects. The manifestations of steroid-induced psychiatric symptoms are broad and can involve affective, behavioral, and cognitive domains. While the current mechanism is unknown, this phenomenon may be related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity, as well as effects on brain regions such as the hippocampus and amygdala. The best interventions for steroid-induced psychiatric symptoms are awareness and early diagnosis. There are no FDA-approved treatments for steroid-induced psychiatric symptoms; initial measures should include tapering or discontinuing corticosteroids.
In this article, we review the literature on the incidence, characteristics, differential diagnoses, proposed mechanism, risk factors, and proposed treatments of steroid-induced psychiatric symptoms.
A wide range of presentations
Steroid use has increased over the past 2 decades, with 10% of medical and surgical inpatients and 1% to 3% of the general population taking long-term glucocorticoids.1 Even with topical application, steroid therapy is often systemically absorbed, and thus may lead to steroid-induced psychiatric symptoms. The incidence of steroid-induced psychiatric symptoms is difficult to assess because there can be a wide range of reactions that are dose- and time-related. Three reviews of a total of 122 cases reports found that an estimated 5% of patients treated with steroids experience severe psychiatric reactions.1-3
Steroid-induced psychopathology can include mood, behavioral, and/or cognitive impairments. Mania/hypomania is the most common overall psychiatric symptom; the most common mood manifestations are anxiety and depression.4,5 Other possible steroid-induced symptoms include psychosis, dementia, panic disorder, delirium, suicidal thinking and behavior, aggressive behavior, insomnia, agitation, depersonalization, and euphoria.5 The most common cognitive impairment is verbal or declarative memory deficit; others include distractibility and deficits in attention and psychomotor speed.5 These psychiatric symptoms can have a rapid onset, possibly within hours of starting steroids.1 However, studies have reported a median time to onset of 11.5 days; 39% of cases had onset during the first week and 62% within 2 weeks.3,6 After reducing or stopping the steroid, it may take days to weeks before symptoms start to subside.2
What to consider in the differential Dx
Psychiatric symptoms that are induced by steroids can mimic metabolic, neurologic, or toxic disorders. Other factors to consider include drug withdrawal/intoxication, infections, and paraneoplastic syndromes.4,5 Although there is no reported correlation between the location of neurologic lesions and the development of specific psychiatric symptoms, manic symptoms appear most commonly with lesions in the right frontal lobe. 4 Other factors to note include the presence of new-onset psychiatric illnesses such as bipolar, mood, or thought disorders,4 as well as psychosocial stressors that might be contributing to the patient’s presentation.5
Continue to: Proposed mechanisms
Proposed mechanisms
Although the exact mechanism by which steroids induce psychiatric symptoms is unknown, several mechanisms have been proposed. One hypothesis is that steroid-induced psychopathology is related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity.4,5,7 This may explain why many patients with major depressive disorder have elevated cortisol production and/or lack of suppression of cortisol secretion during a dexamethasone stimulation test, and why approximately one-half of patients with Cushing’s disease experience depressive symptoms.8 This is also likely why antipsychotics, which typically reduce cortisol, are efficacious treatments for some steroid-induced psychiatric symptoms.9
Cognitive impairments from steroid use may be related to these agents’ effects on certain brain regions. One such area is the hippocampus, an important mediator in the creation and maintenance of episodic and declarative memories.5,8,9 Acute glucocorticoid use is associated with decreased activity in the left hippocampus, reduced hippocampal glucose metabolism, and reduced cerebral blood flow in the posterior medial temporal lobe.10 Long-term glucocorticoid exposure is associated with smaller hippocampal volume and lower levels of temporal lobe N-acetylaspartate, a marker of neuronal viability.10 Because working memory depends on the prefrontal cortex and declarative memory relies on the hippocampus, deficits in these functions can be attributed to the effect of prolonged glucocorticoid exposure on glucocorticoid or mineralocorticoid receptors in the hippocampus, reduction of hippocampal volume, or elevated glutamate accumulation in that area.11 In addition, high cortisol levels inhibit brain-derived neurotrophic factor, which plays a crucial role in maintaining neural architecture in key brain regions such as the hippocampus and prefrontal cortex.11 There is also a correlation between the duration of prednisone treatment and atrophy of the right amygdala, which is an important regulator of mood and anxiety.11 Both the hippocampus and amygdala have dense collections of glucocorticoid receptors. This may explain why patients who receive high-dose corticosteroids can have reversible atrophy in the hypothalamus and amygdala, leading to deficits in emotional learning and the stress response.
Factors that increase risk
Several factors can increase the risk of steroid-induced psychopathology. The most significant is the dose; higher doses are more likely to produce psychiatric symptoms.1,5 Concurrent use of drugs that increase circulating levels of corticosteroids, such as inhibitors of the cytochrome P450 (CYP) enzyme (eg, clarithromycin), also increases the likelihood of developing psychiatric symptoms.1,5 Risk is also increased in patients with liver or renal dysfunction.1 Cerebral spinal fluid/serum albumin ratio, a marker of blood-brain barrier damage, and low serum complement levels were also reported to be independent risk factors,12 with the thought that increased permeability of the blood-brain barrier may allow hydrophobic steroid molecules to more easily penetrate the CNS, leading to increased neuropsychiatric effects. Hypoalbuminemia is another reported risk factor, perhaps because lower levels of serum albumin are related to higher levels of free and active glucocorticoids, which are normally inactive when bound to albumin.13 There also appears to be an increased prevalence of steroid-induced psychopathology in women, perhaps due to greater propensity in women to seek medical care or a higher prevalence of women with medical disorders that are treated with steroids.5 A previous history of psychiatric disorders may not increase risk.5
Several methods for reducing risk have been proposed, including using a divided-dosing regimens that may lower peak steroid plasma concentrations.13,14 However, the best prevention of steroid-induced psychiatric symptoms are awareness, early diagnosis, and intervention. Studies have suggested that N-methyl-
Treatment options
There are no FDA-approved medications for managing steroid-induced psychiatric symptoms.1,16 Treatment is based on evidence from case reports and a few small case series (Table2-5,17,18).

Continue to: When possible, initial treatment...
When possible, initial treatment should include discontinuing or tapering corticosteroids to <40 mg/d of prednisone-equivalent.1,4,10,18 Most studies have reported rapid reversal of deficits in declarative memory and of hippocampal volume loss once corticosteroids were tapered and discontinued.1,18 One study reported that >90% of patients recovered within 6 weeks, with patients with delirium recovering more quickly (mean: 5.4 days) than those with depression, mania, or psychosis (mean: 19.3 days).3 Another found that the vast majority (92%) of patients treated only with a steroid taper achieved clinical recovery, and 84% recovered with administration of antipsychotics without a steroid taper.3 In this study, all patients who received electroconvulsive therapy (ECT) recovered, as did those who received a steroid taper plus lithium or antipsychotics. Steroid tapering regimens are especially important for patients who have received long-term glucocorticoid treatment. Patients need to be closely monitored for signs of new or increased depression, delirium, or confusion during the taper. If these symptoms occur, the patient should be checked for adrenocortical insufficiency, which can be resolved by re-administering or increasing the dosage of the glucocorticoid.10
Mania. The treatment of mania/hypomania includes mood stabilizers (valproate, lithium, lamotrigine) and antipsychotics (quetiapine, olanzapine, haloperidol).2,4,5,10,14,18 Valproate has been reported to be an effective prophylactic of corticosteroid-induced mania,2 perhaps because it dampens neuronal hyperexcitability by attenuating NMDA receptors, blocking voltage-dependent sodium channels, and inhibiting the synthesis of cortical GABAergic steroids. Starting valproate while continuing corticosteroids (if necessary) may help lessen mania.2 Benzodiazepines also may be useful on a short-term basis.
Depression. Steroid-induced depression may be treated with sertraline or other first-line antidepressants.5,14 Consider ECT for patients with severe depression. Support for the use of antipsychotic medications stems from studies that reported steroids’ role in disrupting dopamine and 5HT2 activity. Lithium also has been used successfully to manage and prevent glucocorticoid-associated affective disorder.10,18 It can be used alone or in combination with selective serotonin reuptake inhibitors to alleviate depressive symptoms.10 Tricyclic antidepressants are generally avoided because their anticholinergic effects can exacerbate or worsen delirium.18 In general, ECT is an effective treatment for persistent and/or unresponsive steroid-induced depression,2,10 but may be difficult to use in patients with serious medical illnesses.
Agitation. Medications that have been proposed for treating steroid-induced agitation include benzodiazepines, haloperidol, and second-generation antipsychotics.5,17
Other considerations. Clinicians, patients, and families should discuss in detail the risks of steroid-induced psychiatric symptoms so an early diagnosis and appropriate intervention can be implemented. Before starting steroids, it is important to review the patient’s current medication list to ensure that steroid treatment is indicated, and to check for potential drug–drug interactions. In addition, the medical condition that is being treated with steroids also needs to be carefully reviewed, because certain illnesses are associated with the development of psychiatric symptoms. 5,10
Continue to: Young children...
Young children (age <6) and older adults appear to be at greater risk for cognitive and memory disturbances from steroid use.10 In addition, patients have individual levels of susceptibility to steroid-induced psychiatric symptoms that can vary over time. The risk for adverse effects may be elevated based on response to previous courses of glucocorticoid treatment.10 While gender, age, dosage, and duration of treatment influence risk, it is not possible to predict which patients will experience psychiatric effects during a given course of glucocorticoid therapy. Therefore, all patients should be considered to have the potential of developing such effects, and should be monitored during glucocorticoid treatment and withdrawal.
Goals for future research
To help reduce the severity of and cost associated with steroid-induced psychiatric symptoms,5,14 future studies should focus on controlled trials of preventative strategies. In particular, recent advances in genetic mapping may help identify involvement of certain genes or polymorphisms.5 Because current guidelines for the prevention and treatment of steroid-induced psychiatric symptoms are not evidence-based, controlled clinical trials are needed to elucidate the optimal management of such symptoms. There is much interplay between many of the proposed mechanisms of steroid-induced psychiatric symptoms, and future studies can help uncover a deeper understanding of the intricacies of this phenomenon.
CASE CONTINUED
Mrs. N is admitted for altered mental status. Medical workup includes MRI of the brain, MRI of the neck, cardiac echocardiogram, and EEG. There is no evidence of acute structural pathology. She is started on olanzapine, 10 mg/d at bedtime for manic and psychotic symptoms, and is discharged after 5 days. After 1 month, the outpatient psychiatrist gradually decreases and discontinues olanzapine as Mrs. N steadily returns to baseline. One year after discharge, Mrs. N continues to report resolution of her manic and psychotic symptoms.
Bottom Line
Steroids can induce a wide range of psychiatric symptoms, including mania/ hypomania, anxiety, and depression. Initial treatment typically includes tapering or discontinuing the steroid when possible. Other proposed treatments include certain antipsychotics, antidepressants, and other psychotropics, but the supporting evidence is largely anecdotal or based on case studies. Additional research is needed to elucidate the mechanism and treatment recommendations.
Related Resources
- Janes M, Kuster S, Goldson TM, et al. Steroid-induced psychosis. Proc (Bayl Univ Med Cent). 2019;32(4):614-615.
- Mayo Clinic. Prednisone and other corticosteroids. https://www.mayoclinic.org/steroids/art-20045692
Drug Brand Names
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Methylprednisolone injection • Solu-Medrol
Olanzapine • Zyprexa
Paroxetine • Paxil
Phenytoin • Dilantin
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Valproate • Depakote
1. Dubovsky AN, Arvikar S, Stern TA, et al. The neuropsychiatric complications of glucocorticoid use: steroid psychosis revisited. Psychosomatics. 2012;53(2):103-115.
2. Roxanas MG, Hunt GE. Rapid reversal of corticosteroid-induced mania with sodium valproate: a case series of 20 patients. Psychosomatics. 2012;53(6):575-581.
3. Lewis DA, Smith RE. Steroid‐induced psychiatric syndromes. A report of 14 cases and a review of the literature. J Affect Disord. 1983;5(4):319-332.
4. Warren KN, Katakam J, Espiridion ED. Acute-onset mania in a patient with non-small cell lung cancer. Cureus. 2019;11(8):e5436.
5. Kenna HA, Poon AW, de los Angeles CP, et al. Psychiatric complications of treatment with corticosteroids: review with case report. Psychiatry Clin Neurosci. 2011;65(6):549-560.
6. Ling MH, Perry PJ, Tsuang MT. Side effects of corticosteroid therapy. Psychiatric aspects. Arch Gen. Psychiatry. 1981;38(4):471-477.
7. Ularntinon S, Tzuang D, Dahl G, et al. Concurrent treatment of steroid-related mood and psychotic symptoms with risperidone. Pediatrics. 2010;125(5):e1241-e1245.
8. Pokladinkova J, Meyboom RH, Vlcek J, et al. Intranasally administered corticosteroids and neuropsychiatric disturbances: a review of the international pharmacovigilance programme of the World Health Organization. Ann Allergy Asthma Immunol. 2008;101(1):67-73.
9. Walker EF, Trotman HD, Pearce BD, et al. Cortisol levels and risk for psychosis: initial findings from the North American prodrome longitudinal study. Biol Psychiatry. 2013;74(6):410-417.
10. Wolkowitz OM, Reus UI. Treatment of depression with antiglucocorticoid drugs. Psychosom Med. 1999;61(5):698-711.
11. Judd LL, Schettler PJ, Brown ES, et al. Adverse consequences of glucocorticoid medication: psychological, cognitive, and behavioral effects. Am J Psychiatry. 2014;171(10):1045-1051.
12. Appenzeller S, Cendes F, Costallat LT. Acute psychosis in systemic lupus erythematosus. Rheumatol Int. 2008;28(3):237-243.
13. Glynne-Jones R, Vernon CC, Bell G. Is steroid psychosis preventable by divided doses? Lancet. 1986;2(8520):1404.
14. Ismail MF, Lavelle C, Cassidy EM. Steroid-induced mental disorders in cancer patients: a systematic review. Future Oncol. 2017;13(29):2719-2731.
15. Magariños AM, McEwen BS. Stress-induced atrophy of apical dendrites of hippocampal CA3c neurons: involvement of glucocorticoid secretion and excitatory amino acid receptors. Neuroscience. 1995;69(1):89-98.
16. Brown BS, Stuard G, Liggin JDM, et al. Effect of phenytoin on mood and declarative memory during prescription corticosteroid therapy. Biol Psychiatry. 2005;57(5):543-548.
17. Desai S, Khanani S, Shad MU, et al. Attenutation of amygdala atrophy with lamotrigine in patients receiving corticosteroid therapy. J Clin Psychopharmacol. 2009;29(3):284-287.
18. Gable M, Depry D. Sustained corticosteroid-induced mania and psychosis despite cessation: a case study and brief literature review. Int J Psychiatry Med. 2015;50(4):398-404.
1. Dubovsky AN, Arvikar S, Stern TA, et al. The neuropsychiatric complications of glucocorticoid use: steroid psychosis revisited. Psychosomatics. 2012;53(2):103-115.
2. Roxanas MG, Hunt GE. Rapid reversal of corticosteroid-induced mania with sodium valproate: a case series of 20 patients. Psychosomatics. 2012;53(6):575-581.
3. Lewis DA, Smith RE. Steroid‐induced psychiatric syndromes. A report of 14 cases and a review of the literature. J Affect Disord. 1983;5(4):319-332.
4. Warren KN, Katakam J, Espiridion ED. Acute-onset mania in a patient with non-small cell lung cancer. Cureus. 2019;11(8):e5436.
5. Kenna HA, Poon AW, de los Angeles CP, et al. Psychiatric complications of treatment with corticosteroids: review with case report. Psychiatry Clin Neurosci. 2011;65(6):549-560.
6. Ling MH, Perry PJ, Tsuang MT. Side effects of corticosteroid therapy. Psychiatric aspects. Arch Gen. Psychiatry. 1981;38(4):471-477.
7. Ularntinon S, Tzuang D, Dahl G, et al. Concurrent treatment of steroid-related mood and psychotic symptoms with risperidone. Pediatrics. 2010;125(5):e1241-e1245.
8. Pokladinkova J, Meyboom RH, Vlcek J, et al. Intranasally administered corticosteroids and neuropsychiatric disturbances: a review of the international pharmacovigilance programme of the World Health Organization. Ann Allergy Asthma Immunol. 2008;101(1):67-73.
9. Walker EF, Trotman HD, Pearce BD, et al. Cortisol levels and risk for psychosis: initial findings from the North American prodrome longitudinal study. Biol Psychiatry. 2013;74(6):410-417.
10. Wolkowitz OM, Reus UI. Treatment of depression with antiglucocorticoid drugs. Psychosom Med. 1999;61(5):698-711.
11. Judd LL, Schettler PJ, Brown ES, et al. Adverse consequences of glucocorticoid medication: psychological, cognitive, and behavioral effects. Am J Psychiatry. 2014;171(10):1045-1051.
12. Appenzeller S, Cendes F, Costallat LT. Acute psychosis in systemic lupus erythematosus. Rheumatol Int. 2008;28(3):237-243.
13. Glynne-Jones R, Vernon CC, Bell G. Is steroid psychosis preventable by divided doses? Lancet. 1986;2(8520):1404.
14. Ismail MF, Lavelle C, Cassidy EM. Steroid-induced mental disorders in cancer patients: a systematic review. Future Oncol. 2017;13(29):2719-2731.
15. Magariños AM, McEwen BS. Stress-induced atrophy of apical dendrites of hippocampal CA3c neurons: involvement of glucocorticoid secretion and excitatory amino acid receptors. Neuroscience. 1995;69(1):89-98.
16. Brown BS, Stuard G, Liggin JDM, et al. Effect of phenytoin on mood and declarative memory during prescription corticosteroid therapy. Biol Psychiatry. 2005;57(5):543-548.
17. Desai S, Khanani S, Shad MU, et al. Attenutation of amygdala atrophy with lamotrigine in patients receiving corticosteroid therapy. J Clin Psychopharmacol. 2009;29(3):284-287.
18. Gable M, Depry D. Sustained corticosteroid-induced mania and psychosis despite cessation: a case study and brief literature review. Int J Psychiatry Med. 2015;50(4):398-404.
High-dose lumateperone: A case report
Lumateperone is a novel antipsychotic that possesses a variety of unique receptor affinities. The recommended dose of lumateperone is 42 mg/d. In clinical trials, reductions in Positive and Negative Syndrome Scale scores observed with lumateperone, 28 mg/d and 84 mg/d, failed to separate from placebo.1 However, in these trials, safety profiles were similar for all 3 doses.
Despite the popular understanding of lumateperone’s “unexplained narrow therapeutic window,”2 we report the case of a patient with schizophrenia who responded well to lumateperone, 84 mg/d, without adverse effects or EKG changes.
Case report. Mr. W, age 26, has treatment-resistant schizophrenia (paranoid type). He failed to achieve remission on fluphenazine (10 to 25 mg/d), perphenazine (4 to 24 mg/d), risperidone (started at 4 mg/d and increased to 8 mg/d), and olanzapine (15, 20, and 25 mg/d). None of these medications eliminated his auditory or visual hallucinations. His response was most robust to perphenazine, as he reported a 50% reduction in the frequency of auditory hallucinations and a near-complete resolution of visual hallucinations (once or twice per week), but he never achieved full remission.
We started lumateperone, 42 mg/d, without a cross-taper. After 4 weeks of partial response, the patient escalated his dose to 84 mg/d on his own. At a follow-up visit 3.5 weeks after this self-directed dose increase, Mr. W reported a complete resolution of his auditory and visual hallucinations.
Six months later, Mr. W continued to receive lumateperone, 84 mg/d, without extrapyramidal symptoms, tardive dyskinesia, or other adverse effects. His QTc showed no significant change (410 ms vs 412 ms).
Although some studies indicate a possible “therapeutic window” for lumateperone dosing, clinicians should not deprive patients who partially respond to the recommended 42 mg/d dose of the opportunity for additional benefit through dose escalation. Due to the vagaries of psychiatric pathology, and unique profiles of metabolism and receptor sensitivity, there will always be patients who may require higher-than-recommended doses of lumateperone, as with all other agents.
1. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952-961. doi: 10.1016/j.biopsych.2015.08.026
2. Kantrowitz JT. The potential role of lumateperone—something borrowed? something new? JAMA Psychiatry. 2020;77(4):343-344. doi:10.1001/jamapsychiatry.2019.4265
Lumateperone is a novel antipsychotic that possesses a variety of unique receptor affinities. The recommended dose of lumateperone is 42 mg/d. In clinical trials, reductions in Positive and Negative Syndrome Scale scores observed with lumateperone, 28 mg/d and 84 mg/d, failed to separate from placebo.1 However, in these trials, safety profiles were similar for all 3 doses.
Despite the popular understanding of lumateperone’s “unexplained narrow therapeutic window,”2 we report the case of a patient with schizophrenia who responded well to lumateperone, 84 mg/d, without adverse effects or EKG changes.
Case report. Mr. W, age 26, has treatment-resistant schizophrenia (paranoid type). He failed to achieve remission on fluphenazine (10 to 25 mg/d), perphenazine (4 to 24 mg/d), risperidone (started at 4 mg/d and increased to 8 mg/d), and olanzapine (15, 20, and 25 mg/d). None of these medications eliminated his auditory or visual hallucinations. His response was most robust to perphenazine, as he reported a 50% reduction in the frequency of auditory hallucinations and a near-complete resolution of visual hallucinations (once or twice per week), but he never achieved full remission.
We started lumateperone, 42 mg/d, without a cross-taper. After 4 weeks of partial response, the patient escalated his dose to 84 mg/d on his own. At a follow-up visit 3.5 weeks after this self-directed dose increase, Mr. W reported a complete resolution of his auditory and visual hallucinations.
Six months later, Mr. W continued to receive lumateperone, 84 mg/d, without extrapyramidal symptoms, tardive dyskinesia, or other adverse effects. His QTc showed no significant change (410 ms vs 412 ms).
Although some studies indicate a possible “therapeutic window” for lumateperone dosing, clinicians should not deprive patients who partially respond to the recommended 42 mg/d dose of the opportunity for additional benefit through dose escalation. Due to the vagaries of psychiatric pathology, and unique profiles of metabolism and receptor sensitivity, there will always be patients who may require higher-than-recommended doses of lumateperone, as with all other agents.
Lumateperone is a novel antipsychotic that possesses a variety of unique receptor affinities. The recommended dose of lumateperone is 42 mg/d. In clinical trials, reductions in Positive and Negative Syndrome Scale scores observed with lumateperone, 28 mg/d and 84 mg/d, failed to separate from placebo.1 However, in these trials, safety profiles were similar for all 3 doses.
Despite the popular understanding of lumateperone’s “unexplained narrow therapeutic window,”2 we report the case of a patient with schizophrenia who responded well to lumateperone, 84 mg/d, without adverse effects or EKG changes.
Case report. Mr. W, age 26, has treatment-resistant schizophrenia (paranoid type). He failed to achieve remission on fluphenazine (10 to 25 mg/d), perphenazine (4 to 24 mg/d), risperidone (started at 4 mg/d and increased to 8 mg/d), and olanzapine (15, 20, and 25 mg/d). None of these medications eliminated his auditory or visual hallucinations. His response was most robust to perphenazine, as he reported a 50% reduction in the frequency of auditory hallucinations and a near-complete resolution of visual hallucinations (once or twice per week), but he never achieved full remission.
We started lumateperone, 42 mg/d, without a cross-taper. After 4 weeks of partial response, the patient escalated his dose to 84 mg/d on his own. At a follow-up visit 3.5 weeks after this self-directed dose increase, Mr. W reported a complete resolution of his auditory and visual hallucinations.
Six months later, Mr. W continued to receive lumateperone, 84 mg/d, without extrapyramidal symptoms, tardive dyskinesia, or other adverse effects. His QTc showed no significant change (410 ms vs 412 ms).
Although some studies indicate a possible “therapeutic window” for lumateperone dosing, clinicians should not deprive patients who partially respond to the recommended 42 mg/d dose of the opportunity for additional benefit through dose escalation. Due to the vagaries of psychiatric pathology, and unique profiles of metabolism and receptor sensitivity, there will always be patients who may require higher-than-recommended doses of lumateperone, as with all other agents.
1. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952-961. doi: 10.1016/j.biopsych.2015.08.026
2. Kantrowitz JT. The potential role of lumateperone—something borrowed? something new? JAMA Psychiatry. 2020;77(4):343-344. doi:10.1001/jamapsychiatry.2019.4265
1. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952-961. doi: 10.1016/j.biopsych.2015.08.026
2. Kantrowitz JT. The potential role of lumateperone—something borrowed? something new? JAMA Psychiatry. 2020;77(4):343-344. doi:10.1001/jamapsychiatry.2019.4265
Switching antipsychotics: A guide to dose equivalents
Chlorpromazine (CPZ), a low-potency first-generation antipsychotic (FGA), was the first medication approved for the management of schizophrenia. Since its approval, some psychiatrists have prescribed subsequent antipsychotics based on CPZ’s efficacy and dosing. Comparing dosages of newer antipsychotics using a CPZ equivalent as a baseline remains a relevant method of determining which agent to prescribe, and at what dose.1,2
Psychiatrists frequently care for patients who are treatment-refractory or older adults with poor medication tolerance and age-related medical illness. Quick access to the comparative potency of different antipsychotics can help guide titration to the approximate equivalent dose of CPZ when initiating a medication, switching from 1 antipsychotic to another, or augmenting or combining antipsychotics. Fortunately, many authors, such as Woods2and Davis,3 have codified the dosing ratio equivalences of FGAs and second-generation antipsychotics (SGAs) using CPZ, 100 mg. To help psychiatrists use CPZ dosages as a point of comparison for prescribing other antipsychotics, the Table1,2,4 (page 14) lists dose equivalents for oral FGAs and SGAs based on CPZ, 100 mg. (For information on dose equivalents for injectable antipsychotics, see “Second-generation long-acting injectable antipsychotics: A practical guide,”
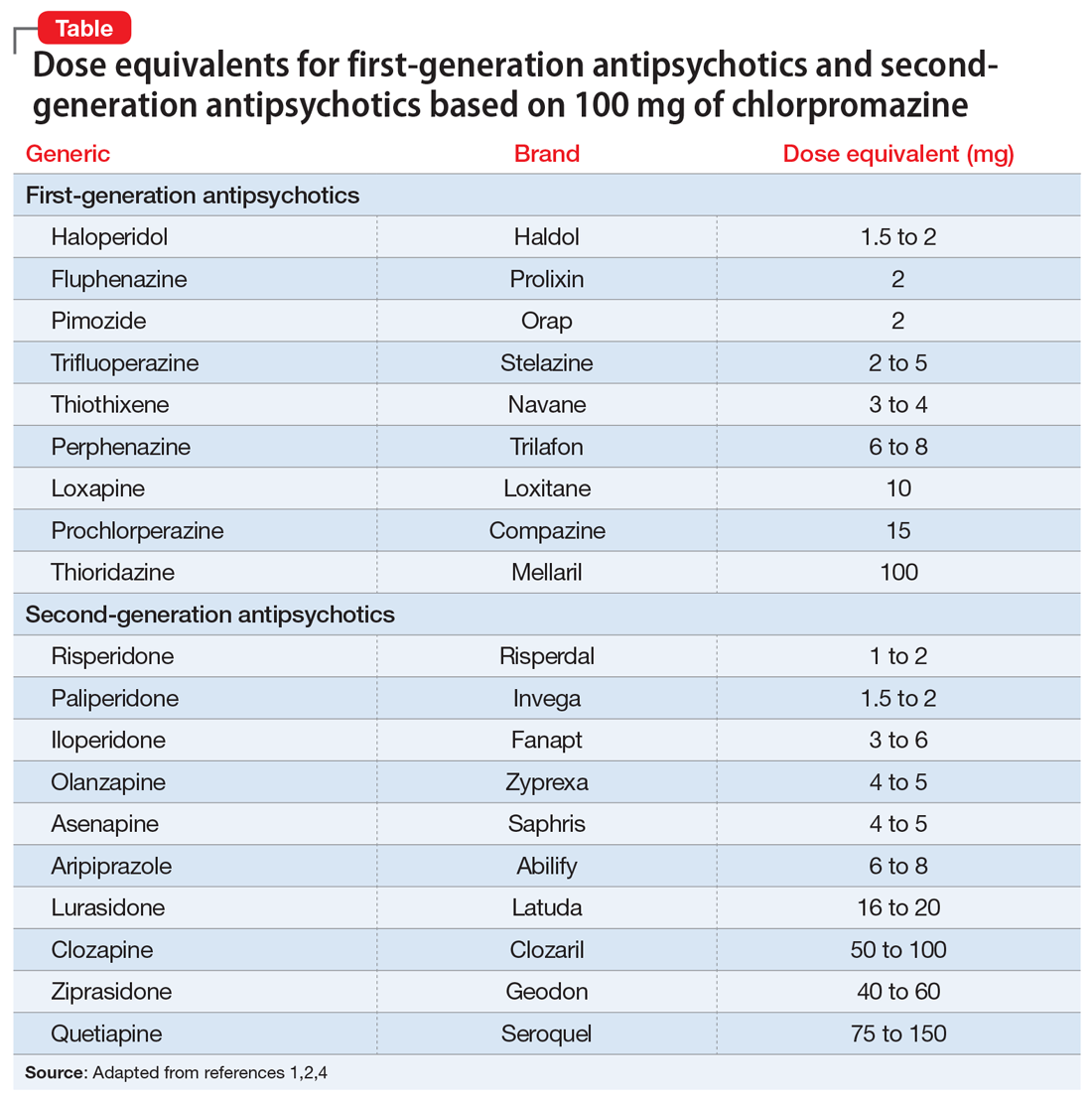
While this information cannot replace a psychiatrist’s clinical judgment, it can serve as a clinically useful prescribing tool. In addition to providing this Table, we discuss what you should consider when using these equivalents to switch antipsychotics and estimate the ultimate dose target for effective management of psychotic disorders.
A few caveats
Bioactive equivalent dosages should be targeted as a rough guide when switching from one FGA or SGA to another. Common indications for switching antipsychotics include an inadequate therapeutic response after a medication trial of an adequate dose and duration; relapse of psychosis despite medication adherence; intolerable adverse effects; cost; a new-onset, contraindicating medical illness; and lapses in medication compliance that necessitate a change to IM formulations.5 Keep in mind that medication changes should be tailored to the patient’s specific clinical characteristics.
Several other clinical and pharmacologic variabilities should be kept in mind when switching antipsychotics using CPZ dosage equivalents5,6:
- The therapeutic CPZ equivalent doses may be less precise for SGAs than for FGAs because the equivalents are largely based on dopaminergic blockade instead of cholinergic, serotonergic, or histaminergic systems
- For some antipsychotics, the relationship between dose and potency is nonlinear. For example, as the dosage of haloperidol increases, its relative antipsychotic potency decreases
- Differences in half-lives between 2 agents can add complexity to calculating the dosage equivalent
- Regardless of comparative dosing, before initiating a new antipsychotic, psychiatrists should read the dosing instructions in the FDA-approved package insert, and exercise caution before titrating a new medication to the maximum recommended dose.
1. Danivas V, Venkatasubramanian G. Current perspectives on chlorpromazine equivalents: comparing apples and oranges! Indian J Psychiatry. 2013;55(2):207-208.
2. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663-667.
3. Davis JM. Dose equivalence of the anti-psychotic drugs. J Psych Res. 1974;11:65-69.
4. Psychiatric pharmacy essentials: antipsychotic dose equivalents. College of Psychiatric and Neurologic Pharmacists. Accessed February 2, 2021. https://cpnp.org/guideline/essentials/antipsychotic-dose-equivalents
5. Guidelines for antipsychotic medication switches. Humber NHS. Last Reviewed September 2012. Accessed February 2, 2021. https://www.psychdb.com/_media/meds/antipsychotics/nhs_guidelines_antipsychotic_switch.pdf
6. Bobo WV. Switching antipsychotics: why, when, and how? Psychiatric Times. Published March 14, 2013. Accessed February 2, 2021. https://www.psychiatrictimes.com/view/switching-antipsychotics-why-when-and-how
Chlorpromazine (CPZ), a low-potency first-generation antipsychotic (FGA), was the first medication approved for the management of schizophrenia. Since its approval, some psychiatrists have prescribed subsequent antipsychotics based on CPZ’s efficacy and dosing. Comparing dosages of newer antipsychotics using a CPZ equivalent as a baseline remains a relevant method of determining which agent to prescribe, and at what dose.1,2
Psychiatrists frequently care for patients who are treatment-refractory or older adults with poor medication tolerance and age-related medical illness. Quick access to the comparative potency of different antipsychotics can help guide titration to the approximate equivalent dose of CPZ when initiating a medication, switching from 1 antipsychotic to another, or augmenting or combining antipsychotics. Fortunately, many authors, such as Woods2and Davis,3 have codified the dosing ratio equivalences of FGAs and second-generation antipsychotics (SGAs) using CPZ, 100 mg. To help psychiatrists use CPZ dosages as a point of comparison for prescribing other antipsychotics, the Table1,2,4 (page 14) lists dose equivalents for oral FGAs and SGAs based on CPZ, 100 mg. (For information on dose equivalents for injectable antipsychotics, see “Second-generation long-acting injectable antipsychotics: A practical guide,”

While this information cannot replace a psychiatrist’s clinical judgment, it can serve as a clinically useful prescribing tool. In addition to providing this Table, we discuss what you should consider when using these equivalents to switch antipsychotics and estimate the ultimate dose target for effective management of psychotic disorders.
A few caveats
Bioactive equivalent dosages should be targeted as a rough guide when switching from one FGA or SGA to another. Common indications for switching antipsychotics include an inadequate therapeutic response after a medication trial of an adequate dose and duration; relapse of psychosis despite medication adherence; intolerable adverse effects; cost; a new-onset, contraindicating medical illness; and lapses in medication compliance that necessitate a change to IM formulations.5 Keep in mind that medication changes should be tailored to the patient’s specific clinical characteristics.
Several other clinical and pharmacologic variabilities should be kept in mind when switching antipsychotics using CPZ dosage equivalents5,6:
- The therapeutic CPZ equivalent doses may be less precise for SGAs than for FGAs because the equivalents are largely based on dopaminergic blockade instead of cholinergic, serotonergic, or histaminergic systems
- For some antipsychotics, the relationship between dose and potency is nonlinear. For example, as the dosage of haloperidol increases, its relative antipsychotic potency decreases
- Differences in half-lives between 2 agents can add complexity to calculating the dosage equivalent
- Regardless of comparative dosing, before initiating a new antipsychotic, psychiatrists should read the dosing instructions in the FDA-approved package insert, and exercise caution before titrating a new medication to the maximum recommended dose.
Chlorpromazine (CPZ), a low-potency first-generation antipsychotic (FGA), was the first medication approved for the management of schizophrenia. Since its approval, some psychiatrists have prescribed subsequent antipsychotics based on CPZ’s efficacy and dosing. Comparing dosages of newer antipsychotics using a CPZ equivalent as a baseline remains a relevant method of determining which agent to prescribe, and at what dose.1,2
Psychiatrists frequently care for patients who are treatment-refractory or older adults with poor medication tolerance and age-related medical illness. Quick access to the comparative potency of different antipsychotics can help guide titration to the approximate equivalent dose of CPZ when initiating a medication, switching from 1 antipsychotic to another, or augmenting or combining antipsychotics. Fortunately, many authors, such as Woods2and Davis,3 have codified the dosing ratio equivalences of FGAs and second-generation antipsychotics (SGAs) using CPZ, 100 mg. To help psychiatrists use CPZ dosages as a point of comparison for prescribing other antipsychotics, the Table1,2,4 (page 14) lists dose equivalents for oral FGAs and SGAs based on CPZ, 100 mg. (For information on dose equivalents for injectable antipsychotics, see “Second-generation long-acting injectable antipsychotics: A practical guide,”

While this information cannot replace a psychiatrist’s clinical judgment, it can serve as a clinically useful prescribing tool. In addition to providing this Table, we discuss what you should consider when using these equivalents to switch antipsychotics and estimate the ultimate dose target for effective management of psychotic disorders.
A few caveats
Bioactive equivalent dosages should be targeted as a rough guide when switching from one FGA or SGA to another. Common indications for switching antipsychotics include an inadequate therapeutic response after a medication trial of an adequate dose and duration; relapse of psychosis despite medication adherence; intolerable adverse effects; cost; a new-onset, contraindicating medical illness; and lapses in medication compliance that necessitate a change to IM formulations.5 Keep in mind that medication changes should be tailored to the patient’s specific clinical characteristics.
Several other clinical and pharmacologic variabilities should be kept in mind when switching antipsychotics using CPZ dosage equivalents5,6:
- The therapeutic CPZ equivalent doses may be less precise for SGAs than for FGAs because the equivalents are largely based on dopaminergic blockade instead of cholinergic, serotonergic, or histaminergic systems
- For some antipsychotics, the relationship between dose and potency is nonlinear. For example, as the dosage of haloperidol increases, its relative antipsychotic potency decreases
- Differences in half-lives between 2 agents can add complexity to calculating the dosage equivalent
- Regardless of comparative dosing, before initiating a new antipsychotic, psychiatrists should read the dosing instructions in the FDA-approved package insert, and exercise caution before titrating a new medication to the maximum recommended dose.
1. Danivas V, Venkatasubramanian G. Current perspectives on chlorpromazine equivalents: comparing apples and oranges! Indian J Psychiatry. 2013;55(2):207-208.
2. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663-667.
3. Davis JM. Dose equivalence of the anti-psychotic drugs. J Psych Res. 1974;11:65-69.
4. Psychiatric pharmacy essentials: antipsychotic dose equivalents. College of Psychiatric and Neurologic Pharmacists. Accessed February 2, 2021. https://cpnp.org/guideline/essentials/antipsychotic-dose-equivalents
5. Guidelines for antipsychotic medication switches. Humber NHS. Last Reviewed September 2012. Accessed February 2, 2021. https://www.psychdb.com/_media/meds/antipsychotics/nhs_guidelines_antipsychotic_switch.pdf
6. Bobo WV. Switching antipsychotics: why, when, and how? Psychiatric Times. Published March 14, 2013. Accessed February 2, 2021. https://www.psychiatrictimes.com/view/switching-antipsychotics-why-when-and-how
1. Danivas V, Venkatasubramanian G. Current perspectives on chlorpromazine equivalents: comparing apples and oranges! Indian J Psychiatry. 2013;55(2):207-208.
2. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663-667.
3. Davis JM. Dose equivalence of the anti-psychotic drugs. J Psych Res. 1974;11:65-69.
4. Psychiatric pharmacy essentials: antipsychotic dose equivalents. College of Psychiatric and Neurologic Pharmacists. Accessed February 2, 2021. https://cpnp.org/guideline/essentials/antipsychotic-dose-equivalents
5. Guidelines for antipsychotic medication switches. Humber NHS. Last Reviewed September 2012. Accessed February 2, 2021. https://www.psychdb.com/_media/meds/antipsychotics/nhs_guidelines_antipsychotic_switch.pdf
6. Bobo WV. Switching antipsychotics: why, when, and how? Psychiatric Times. Published March 14, 2013. Accessed February 2, 2021. https://www.psychiatrictimes.com/view/switching-antipsychotics-why-when-and-how
Discovery of schizophrenia gene could advance research, therapies
A new genetic mutation in schizophrenia that blocks neuron communication in the brain may lead to novel treatment strategies and improve understanding of the mechanics of this disease.
The discovery of this new gene, PCDHA3, could enhance the development of genetic-risk calculators “that may help us understand vulnerability to schizophrenia in high-risk individuals and identify individuals with schizophrenia who have a greater risk for poor outcomes,” said Todd Lencz, PhD, a professor at the Feinstein Institutes for Medical Research in New York, and lead author of this research. Dr. Lencz and associates reported on this new finding in the journal Neuron.
Schizophrenia affects 20 million people worldwide. Previous research has identified the important role genes play in the disease, but isolating individual genes to better understand schizophrenia has proven to be a challenge. This is a very heterogeneous disorder, with many hundreds if not thousands of genes involved, Dr. Lencz explained in an interview. “It is very different from single-gene disorders like Huntington disease, for example. For this reason, we need very large sample sizes to find any one gene that seems to be common to many cases in a sample.”
Study focused on homogeneous population
To enhance the power of finding rare variants in a heterogeneous disease with large numbers of genes, Dr. Lencz and colleagues chose a homogeneous “founder” population, a cohort of Ashkenazi Jews, to examine genomes from schizophrenia patients and controls. “As we have reported in prior work over the last decade, the 10 million or so Ashkenazi Jews living worldwide today all are descended from just a few hundred people who lived approximately 750 years ago, and moved into Central and Eastern Europe,” said Dr. Lencz. The study included 786 cases of schizophrenia and 463 controls from this Ashkenazi population. This is considered to be an extremely small sample for a genetic study. However, because this population evolved from a few hundred individuals to a massive explosion in a historically short period of time, it had enhanced statistical power, said Dr. Lencz.
“We showed that just a few thousand Ashkenazi Jewish cases would have the statistical power of a regular population that was 5-10 times larger, from a genetic discovery perspective,” he added.
Search for ultrarare variants
The investigators used whole-genome sequencing to conduct their analysis, using public databases to filter out any variants that had been previously observed in healthy individuals worldwide. “We were looking for ultrarare variants that might have a very powerful effect on the disease,” Dr. Lencz said. Such individual mutations are very rarely seen in the general population.
Because of the disease’s ultraheterogeneity, it’s extremely unusual to find a recurrent, ultrarare variant. “In some ways, the genetics of schizophrenia is so complex that every patient worldwide is unique in the genetics that led to his or her disorder.” The goal was to find individual mutations that might be observed multiple times across the schizophrenia group, Dr. Lencz said.
Rare gene found in five cases
Dr. Lencz and colleagues accomplished this with their unique Ashkenazi Jewish population. “We identified one particular mutation that was repeatedly observed in our cases that has not been observed in healthy individuals that we’re aware of,” he said. The PCDHA3 mutation was identified in 3 out of the 786 schizophrenia cases.
In another dataset, they examined from the Schizophrenia Exome Sequencing Meta-analysis (SCHEMA) consortium, they found it two additional times, bringing the total to five cases. SCHEMA is a large international consortium of genetics studies in schizophrenia that contains thousands of cases and controls, some of which are Ashkenazi Jewish cases.
“Importantly, the mutation was not observed in any controls, in either our Ashkenazi dataset, the SCHEMA dataset, or more than 100,000 other controls reported in several publicly available genetics databases,” Dr. Lencz said.
How the gene leads to schizophrenia
PCDHA3 derives from the protocadherin gene family, which generates a unique bar code that enables neurons to recognize and communicate with other neurons. This communication creates a scaffolding of sorts that enables normal brain function. Dr. Lencz and colleagues discovered that the PCDHA3 variant blocks this normal protocadherin function.
Among the 786 cases, the investigators found several other genes in the broad cadherin family that had implications in schizophrenia development.
Much of the genetics of schizophrenia in recent years has focused on the synapse as the point of abnormality underlying the disorder. “We think our paper demonstrates in multiple ways the synaptic scaffolding role the cadherins superfamily of genes play in schizophrenia pathophysiology. The discovery of the PCDHA3 variant adds a level of detail and resolution to this process, pointing researchers toward a specific aspect of synaptic formation that may be aberrant. “So the hope is we’re not just learning about these five individuals and their synapses. This result is perhaps telling us to look very carefully at this aspect of synaptic formation.”
Implications for clinical practice
Dr. Lencz and colleagues plan to expand upon and enhance their existing Ashkenazi sample to take advantage of the founder effect in this population. “Of course, there are many large-scale efforts to recruit ethnically diverse patients with schizophrenia to study around the world. We encourage that. Our expectation is that the biology is not in any way unique to Ashkenazi individuals. This is just the approach we took to enhance our power,” he said.
The PCDHA3 discovery won’t have an immediate impact on clinical practice. In the longer term, “we are aware of certain pharmacologic approaches that might be able to manipulate the cadherins. That would be a worthy focus for future research,” Dr. Lencz said.
Additional studies will be critical to see how current medications in schizophrenia treatment could mitigate and improve any changes caused by this genetic mutation, noted Anthony T. Ng, MD, who was not involved with the study. More specifically, studies would help assess the impact of a schizophrenia patient with this mutation in areas of functioning, “so that psychosocial and rehabilitation treatment models of schizophrenia can provide more targeted treatment,” said Dr. Ng, medical director of community services and director of neuromodulation services at Northern Light Acadia Hospital in Bangor, Maine.
The work of Dr. Lencz and associates is significant in that “it started to identify a very specific genetic change that can help focus treatment of schizophrenia,” Dr. Ng said.
Neither Dr. Lencz nor his associates had any conflicts of interest. Dr. Ng had no disclosures.
A new genetic mutation in schizophrenia that blocks neuron communication in the brain may lead to novel treatment strategies and improve understanding of the mechanics of this disease.
The discovery of this new gene, PCDHA3, could enhance the development of genetic-risk calculators “that may help us understand vulnerability to schizophrenia in high-risk individuals and identify individuals with schizophrenia who have a greater risk for poor outcomes,” said Todd Lencz, PhD, a professor at the Feinstein Institutes for Medical Research in New York, and lead author of this research. Dr. Lencz and associates reported on this new finding in the journal Neuron.
Schizophrenia affects 20 million people worldwide. Previous research has identified the important role genes play in the disease, but isolating individual genes to better understand schizophrenia has proven to be a challenge. This is a very heterogeneous disorder, with many hundreds if not thousands of genes involved, Dr. Lencz explained in an interview. “It is very different from single-gene disorders like Huntington disease, for example. For this reason, we need very large sample sizes to find any one gene that seems to be common to many cases in a sample.”
Study focused on homogeneous population
To enhance the power of finding rare variants in a heterogeneous disease with large numbers of genes, Dr. Lencz and colleagues chose a homogeneous “founder” population, a cohort of Ashkenazi Jews, to examine genomes from schizophrenia patients and controls. “As we have reported in prior work over the last decade, the 10 million or so Ashkenazi Jews living worldwide today all are descended from just a few hundred people who lived approximately 750 years ago, and moved into Central and Eastern Europe,” said Dr. Lencz. The study included 786 cases of schizophrenia and 463 controls from this Ashkenazi population. This is considered to be an extremely small sample for a genetic study. However, because this population evolved from a few hundred individuals to a massive explosion in a historically short period of time, it had enhanced statistical power, said Dr. Lencz.
“We showed that just a few thousand Ashkenazi Jewish cases would have the statistical power of a regular population that was 5-10 times larger, from a genetic discovery perspective,” he added.
Search for ultrarare variants
The investigators used whole-genome sequencing to conduct their analysis, using public databases to filter out any variants that had been previously observed in healthy individuals worldwide. “We were looking for ultrarare variants that might have a very powerful effect on the disease,” Dr. Lencz said. Such individual mutations are very rarely seen in the general population.
Because of the disease’s ultraheterogeneity, it’s extremely unusual to find a recurrent, ultrarare variant. “In some ways, the genetics of schizophrenia is so complex that every patient worldwide is unique in the genetics that led to his or her disorder.” The goal was to find individual mutations that might be observed multiple times across the schizophrenia group, Dr. Lencz said.
Rare gene found in five cases
Dr. Lencz and colleagues accomplished this with their unique Ashkenazi Jewish population. “We identified one particular mutation that was repeatedly observed in our cases that has not been observed in healthy individuals that we’re aware of,” he said. The PCDHA3 mutation was identified in 3 out of the 786 schizophrenia cases.
In another dataset, they examined from the Schizophrenia Exome Sequencing Meta-analysis (SCHEMA) consortium, they found it two additional times, bringing the total to five cases. SCHEMA is a large international consortium of genetics studies in schizophrenia that contains thousands of cases and controls, some of which are Ashkenazi Jewish cases.
“Importantly, the mutation was not observed in any controls, in either our Ashkenazi dataset, the SCHEMA dataset, or more than 100,000 other controls reported in several publicly available genetics databases,” Dr. Lencz said.
How the gene leads to schizophrenia
PCDHA3 derives from the protocadherin gene family, which generates a unique bar code that enables neurons to recognize and communicate with other neurons. This communication creates a scaffolding of sorts that enables normal brain function. Dr. Lencz and colleagues discovered that the PCDHA3 variant blocks this normal protocadherin function.
Among the 786 cases, the investigators found several other genes in the broad cadherin family that had implications in schizophrenia development.
Much of the genetics of schizophrenia in recent years has focused on the synapse as the point of abnormality underlying the disorder. “We think our paper demonstrates in multiple ways the synaptic scaffolding role the cadherins superfamily of genes play in schizophrenia pathophysiology. The discovery of the PCDHA3 variant adds a level of detail and resolution to this process, pointing researchers toward a specific aspect of synaptic formation that may be aberrant. “So the hope is we’re not just learning about these five individuals and their synapses. This result is perhaps telling us to look very carefully at this aspect of synaptic formation.”
Implications for clinical practice
Dr. Lencz and colleagues plan to expand upon and enhance their existing Ashkenazi sample to take advantage of the founder effect in this population. “Of course, there are many large-scale efforts to recruit ethnically diverse patients with schizophrenia to study around the world. We encourage that. Our expectation is that the biology is not in any way unique to Ashkenazi individuals. This is just the approach we took to enhance our power,” he said.
The PCDHA3 discovery won’t have an immediate impact on clinical practice. In the longer term, “we are aware of certain pharmacologic approaches that might be able to manipulate the cadherins. That would be a worthy focus for future research,” Dr. Lencz said.
Additional studies will be critical to see how current medications in schizophrenia treatment could mitigate and improve any changes caused by this genetic mutation, noted Anthony T. Ng, MD, who was not involved with the study. More specifically, studies would help assess the impact of a schizophrenia patient with this mutation in areas of functioning, “so that psychosocial and rehabilitation treatment models of schizophrenia can provide more targeted treatment,” said Dr. Ng, medical director of community services and director of neuromodulation services at Northern Light Acadia Hospital in Bangor, Maine.
The work of Dr. Lencz and associates is significant in that “it started to identify a very specific genetic change that can help focus treatment of schizophrenia,” Dr. Ng said.
Neither Dr. Lencz nor his associates had any conflicts of interest. Dr. Ng had no disclosures.
A new genetic mutation in schizophrenia that blocks neuron communication in the brain may lead to novel treatment strategies and improve understanding of the mechanics of this disease.
The discovery of this new gene, PCDHA3, could enhance the development of genetic-risk calculators “that may help us understand vulnerability to schizophrenia in high-risk individuals and identify individuals with schizophrenia who have a greater risk for poor outcomes,” said Todd Lencz, PhD, a professor at the Feinstein Institutes for Medical Research in New York, and lead author of this research. Dr. Lencz and associates reported on this new finding in the journal Neuron.
Schizophrenia affects 20 million people worldwide. Previous research has identified the important role genes play in the disease, but isolating individual genes to better understand schizophrenia has proven to be a challenge. This is a very heterogeneous disorder, with many hundreds if not thousands of genes involved, Dr. Lencz explained in an interview. “It is very different from single-gene disorders like Huntington disease, for example. For this reason, we need very large sample sizes to find any one gene that seems to be common to many cases in a sample.”
Study focused on homogeneous population
To enhance the power of finding rare variants in a heterogeneous disease with large numbers of genes, Dr. Lencz and colleagues chose a homogeneous “founder” population, a cohort of Ashkenazi Jews, to examine genomes from schizophrenia patients and controls. “As we have reported in prior work over the last decade, the 10 million or so Ashkenazi Jews living worldwide today all are descended from just a few hundred people who lived approximately 750 years ago, and moved into Central and Eastern Europe,” said Dr. Lencz. The study included 786 cases of schizophrenia and 463 controls from this Ashkenazi population. This is considered to be an extremely small sample for a genetic study. However, because this population evolved from a few hundred individuals to a massive explosion in a historically short period of time, it had enhanced statistical power, said Dr. Lencz.
“We showed that just a few thousand Ashkenazi Jewish cases would have the statistical power of a regular population that was 5-10 times larger, from a genetic discovery perspective,” he added.
Search for ultrarare variants
The investigators used whole-genome sequencing to conduct their analysis, using public databases to filter out any variants that had been previously observed in healthy individuals worldwide. “We were looking for ultrarare variants that might have a very powerful effect on the disease,” Dr. Lencz said. Such individual mutations are very rarely seen in the general population.
Because of the disease’s ultraheterogeneity, it’s extremely unusual to find a recurrent, ultrarare variant. “In some ways, the genetics of schizophrenia is so complex that every patient worldwide is unique in the genetics that led to his or her disorder.” The goal was to find individual mutations that might be observed multiple times across the schizophrenia group, Dr. Lencz said.
Rare gene found in five cases
Dr. Lencz and colleagues accomplished this with their unique Ashkenazi Jewish population. “We identified one particular mutation that was repeatedly observed in our cases that has not been observed in healthy individuals that we’re aware of,” he said. The PCDHA3 mutation was identified in 3 out of the 786 schizophrenia cases.
In another dataset, they examined from the Schizophrenia Exome Sequencing Meta-analysis (SCHEMA) consortium, they found it two additional times, bringing the total to five cases. SCHEMA is a large international consortium of genetics studies in schizophrenia that contains thousands of cases and controls, some of which are Ashkenazi Jewish cases.
“Importantly, the mutation was not observed in any controls, in either our Ashkenazi dataset, the SCHEMA dataset, or more than 100,000 other controls reported in several publicly available genetics databases,” Dr. Lencz said.
How the gene leads to schizophrenia
PCDHA3 derives from the protocadherin gene family, which generates a unique bar code that enables neurons to recognize and communicate with other neurons. This communication creates a scaffolding of sorts that enables normal brain function. Dr. Lencz and colleagues discovered that the PCDHA3 variant blocks this normal protocadherin function.
Among the 786 cases, the investigators found several other genes in the broad cadherin family that had implications in schizophrenia development.
Much of the genetics of schizophrenia in recent years has focused on the synapse as the point of abnormality underlying the disorder. “We think our paper demonstrates in multiple ways the synaptic scaffolding role the cadherins superfamily of genes play in schizophrenia pathophysiology. The discovery of the PCDHA3 variant adds a level of detail and resolution to this process, pointing researchers toward a specific aspect of synaptic formation that may be aberrant. “So the hope is we’re not just learning about these five individuals and their synapses. This result is perhaps telling us to look very carefully at this aspect of synaptic formation.”
Implications for clinical practice
Dr. Lencz and colleagues plan to expand upon and enhance their existing Ashkenazi sample to take advantage of the founder effect in this population. “Of course, there are many large-scale efforts to recruit ethnically diverse patients with schizophrenia to study around the world. We encourage that. Our expectation is that the biology is not in any way unique to Ashkenazi individuals. This is just the approach we took to enhance our power,” he said.
The PCDHA3 discovery won’t have an immediate impact on clinical practice. In the longer term, “we are aware of certain pharmacologic approaches that might be able to manipulate the cadherins. That would be a worthy focus for future research,” Dr. Lencz said.
Additional studies will be critical to see how current medications in schizophrenia treatment could mitigate and improve any changes caused by this genetic mutation, noted Anthony T. Ng, MD, who was not involved with the study. More specifically, studies would help assess the impact of a schizophrenia patient with this mutation in areas of functioning, “so that psychosocial and rehabilitation treatment models of schizophrenia can provide more targeted treatment,” said Dr. Ng, medical director of community services and director of neuromodulation services at Northern Light Acadia Hospital in Bangor, Maine.
The work of Dr. Lencz and associates is significant in that “it started to identify a very specific genetic change that can help focus treatment of schizophrenia,” Dr. Ng said.
Neither Dr. Lencz nor his associates had any conflicts of interest. Dr. Ng had no disclosures.
FROM NEURON
Blood pressure meds tied to increased schizophrenia risk
ACE inhibitors may be associated with an increased risk for schizophrenia and may affect psychiatric symptoms, new research suggests.
Investigators found individuals who carry a genetic variant associated with lower levels of the ACE gene and protein have increased liability to schizophrenia, suggesting that drugs that lower ACE levels or activity may do the same.
“Our findings warrant further investigation into the role of ACE in schizophrenia and closer monitoring by clinicians of individuals, especially those with schizophrenia, who may be on medication that lower ACE activity, such as ACE inhibitors,” Sonia Shah, PhD, Institute for Biomedical Sciences, University of Queensland, Brisbane, Australia, said in an interview.
The study was published online March 10, 2021, in JAMA Psychiatry.
Antihypertensives and mental illness
Hypertension is common in patients with psychiatric disorders and observational studies have reported associations between antihypertensive medication and these disorders, although the findings have been mixed.
Dr. Shah and colleagues estimated the potential of different antihypertensive drug classes on schizophrenia, bipolar disorder, and major depressive disorder.
In a two-sample Mendelian randomization study, they evaluated ties between a single-nucleotide variant and drug-target gene expression derived from expression quantitative trait loci data in blood (sample 1) and the SNV disease association from published case-control, genomewide association studies (sample 2).
The analyses included 40,675 patients with schizophrenia and 64,643 controls; 20,352 with bipolar disorder and 31,358 controls; and 135,458 with major depressive disorder and 344,901 controls.
The major finding was that a one standard deviation–lower expression of the ACE gene in blood was associated with lower systolic blood pressure of 4.0 mm Hg (95% confidence interval, 2.7-5.3), but also an increased risk of schizophrenia (odds ratio, 1.75; 95% CI, 1.28-2.38).
Could ACE inhibitors worsen symptoms or trigger episodes?
In their article, the researchers noted that, in most patients, onset of schizophrenia occurs in late adolescence or early adult life, ruling out ACE inhibitor treatment as a potential causal factor for most cases.
“However, if lower ACE levels play a causal role for schizophrenia risk, it would be reasonable to hypothesize that further lowering of ACE activity in existing patients could worsen symptoms or trigger a new episode,” they wrote.
Dr. Shah emphasized that evidence from genetic analyses alone is “not sufficient to justify changes in prescription guidelines.”
“Patients should not stop taking these medications if they are effective at controlling their blood pressure and they don’t suffer any adverse effects. But it would be reasonable to encourage greater pharmacovigilance,” she said in an interview.
“One way in which we are hoping to follow up these findings,” said Dr. Shah, “is to access electronic health record data for millions of individuals to investigate if there is evidence of increased rates of psychotic episodes in individuals who use ACE inhibitors, compared to other classes of blood pressure–lowering medication.”
Caution warranted
Reached for comment, Timothy Sullivan, MD, chair of psychiatry and behavioral sciences at Staten Island University Hospital in New York, noted that this is an “extremely complicated” study and urged caution in interpreting the results.
“Since most people develop schizophrenia earlier in life, before they usually develop problems with blood pressure, it’s not so much that these drugs might cause schizophrenia,” Dr. Sullivan said.
“But because of their effects on this particular gene, there’s a possibility that they might worsen symptoms or in somebody with borderline risk might cause them to develop symptoms later in life. This may apply to a relatively small number of people who develop symptoms of schizophrenia in their 40s and beyond,” he added.
That’s where “pharmacovigilance” comes into play, Dr. Sullivan said. “In other words, that they otherwise wouldn’t experience?”
Support for the study was provided by the National Health and Medical Research Council (Australia) and U.S. National Institute for Mental Health. Dr. Shah and Dr. Sullivan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ACE inhibitors may be associated with an increased risk for schizophrenia and may affect psychiatric symptoms, new research suggests.
Investigators found individuals who carry a genetic variant associated with lower levels of the ACE gene and protein have increased liability to schizophrenia, suggesting that drugs that lower ACE levels or activity may do the same.
“Our findings warrant further investigation into the role of ACE in schizophrenia and closer monitoring by clinicians of individuals, especially those with schizophrenia, who may be on medication that lower ACE activity, such as ACE inhibitors,” Sonia Shah, PhD, Institute for Biomedical Sciences, University of Queensland, Brisbane, Australia, said in an interview.
The study was published online March 10, 2021, in JAMA Psychiatry.
Antihypertensives and mental illness
Hypertension is common in patients with psychiatric disorders and observational studies have reported associations between antihypertensive medication and these disorders, although the findings have been mixed.
Dr. Shah and colleagues estimated the potential of different antihypertensive drug classes on schizophrenia, bipolar disorder, and major depressive disorder.
In a two-sample Mendelian randomization study, they evaluated ties between a single-nucleotide variant and drug-target gene expression derived from expression quantitative trait loci data in blood (sample 1) and the SNV disease association from published case-control, genomewide association studies (sample 2).
The analyses included 40,675 patients with schizophrenia and 64,643 controls; 20,352 with bipolar disorder and 31,358 controls; and 135,458 with major depressive disorder and 344,901 controls.
The major finding was that a one standard deviation–lower expression of the ACE gene in blood was associated with lower systolic blood pressure of 4.0 mm Hg (95% confidence interval, 2.7-5.3), but also an increased risk of schizophrenia (odds ratio, 1.75; 95% CI, 1.28-2.38).
Could ACE inhibitors worsen symptoms or trigger episodes?
In their article, the researchers noted that, in most patients, onset of schizophrenia occurs in late adolescence or early adult life, ruling out ACE inhibitor treatment as a potential causal factor for most cases.
“However, if lower ACE levels play a causal role for schizophrenia risk, it would be reasonable to hypothesize that further lowering of ACE activity in existing patients could worsen symptoms or trigger a new episode,” they wrote.
Dr. Shah emphasized that evidence from genetic analyses alone is “not sufficient to justify changes in prescription guidelines.”
“Patients should not stop taking these medications if they are effective at controlling their blood pressure and they don’t suffer any adverse effects. But it would be reasonable to encourage greater pharmacovigilance,” she said in an interview.
“One way in which we are hoping to follow up these findings,” said Dr. Shah, “is to access electronic health record data for millions of individuals to investigate if there is evidence of increased rates of psychotic episodes in individuals who use ACE inhibitors, compared to other classes of blood pressure–lowering medication.”
Caution warranted
Reached for comment, Timothy Sullivan, MD, chair of psychiatry and behavioral sciences at Staten Island University Hospital in New York, noted that this is an “extremely complicated” study and urged caution in interpreting the results.
“Since most people develop schizophrenia earlier in life, before they usually develop problems with blood pressure, it’s not so much that these drugs might cause schizophrenia,” Dr. Sullivan said.
“But because of their effects on this particular gene, there’s a possibility that they might worsen symptoms or in somebody with borderline risk might cause them to develop symptoms later in life. This may apply to a relatively small number of people who develop symptoms of schizophrenia in their 40s and beyond,” he added.
That’s where “pharmacovigilance” comes into play, Dr. Sullivan said. “In other words, that they otherwise wouldn’t experience?”
Support for the study was provided by the National Health and Medical Research Council (Australia) and U.S. National Institute for Mental Health. Dr. Shah and Dr. Sullivan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ACE inhibitors may be associated with an increased risk for schizophrenia and may affect psychiatric symptoms, new research suggests.
Investigators found individuals who carry a genetic variant associated with lower levels of the ACE gene and protein have increased liability to schizophrenia, suggesting that drugs that lower ACE levels or activity may do the same.
“Our findings warrant further investigation into the role of ACE in schizophrenia and closer monitoring by clinicians of individuals, especially those with schizophrenia, who may be on medication that lower ACE activity, such as ACE inhibitors,” Sonia Shah, PhD, Institute for Biomedical Sciences, University of Queensland, Brisbane, Australia, said in an interview.
The study was published online March 10, 2021, in JAMA Psychiatry.
Antihypertensives and mental illness
Hypertension is common in patients with psychiatric disorders and observational studies have reported associations between antihypertensive medication and these disorders, although the findings have been mixed.
Dr. Shah and colleagues estimated the potential of different antihypertensive drug classes on schizophrenia, bipolar disorder, and major depressive disorder.
In a two-sample Mendelian randomization study, they evaluated ties between a single-nucleotide variant and drug-target gene expression derived from expression quantitative trait loci data in blood (sample 1) and the SNV disease association from published case-control, genomewide association studies (sample 2).
The analyses included 40,675 patients with schizophrenia and 64,643 controls; 20,352 with bipolar disorder and 31,358 controls; and 135,458 with major depressive disorder and 344,901 controls.
The major finding was that a one standard deviation–lower expression of the ACE gene in blood was associated with lower systolic blood pressure of 4.0 mm Hg (95% confidence interval, 2.7-5.3), but also an increased risk of schizophrenia (odds ratio, 1.75; 95% CI, 1.28-2.38).
Could ACE inhibitors worsen symptoms or trigger episodes?
In their article, the researchers noted that, in most patients, onset of schizophrenia occurs in late adolescence or early adult life, ruling out ACE inhibitor treatment as a potential causal factor for most cases.
“However, if lower ACE levels play a causal role for schizophrenia risk, it would be reasonable to hypothesize that further lowering of ACE activity in existing patients could worsen symptoms or trigger a new episode,” they wrote.
Dr. Shah emphasized that evidence from genetic analyses alone is “not sufficient to justify changes in prescription guidelines.”
“Patients should not stop taking these medications if they are effective at controlling their blood pressure and they don’t suffer any adverse effects. But it would be reasonable to encourage greater pharmacovigilance,” she said in an interview.
“One way in which we are hoping to follow up these findings,” said Dr. Shah, “is to access electronic health record data for millions of individuals to investigate if there is evidence of increased rates of psychotic episodes in individuals who use ACE inhibitors, compared to other classes of blood pressure–lowering medication.”
Caution warranted
Reached for comment, Timothy Sullivan, MD, chair of psychiatry and behavioral sciences at Staten Island University Hospital in New York, noted that this is an “extremely complicated” study and urged caution in interpreting the results.
“Since most people develop schizophrenia earlier in life, before they usually develop problems with blood pressure, it’s not so much that these drugs might cause schizophrenia,” Dr. Sullivan said.
“But because of their effects on this particular gene, there’s a possibility that they might worsen symptoms or in somebody with borderline risk might cause them to develop symptoms later in life. This may apply to a relatively small number of people who develop symptoms of schizophrenia in their 40s and beyond,” he added.
That’s where “pharmacovigilance” comes into play, Dr. Sullivan said. “In other words, that they otherwise wouldn’t experience?”
Support for the study was provided by the National Health and Medical Research Council (Australia) and U.S. National Institute for Mental Health. Dr. Shah and Dr. Sullivan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘Landmark’ schizophrenia drug in the wings?
A novel therapy that combines a muscarinic receptor agonist with an anticholinergic agent is associated with a greater reduction in psychosis symptoms, compared with placebo, new research shows.
In a randomized, phase 2 trial composed of nearly 200 participants, xanomeline-trospium (KarXT) was generally well tolerated and had none of the common side effects linked to current antipsychotics, including weight gain and extrapyramidal symptoms such as dystonia, parkinsonism, and tardive dyskinesia.
“The results showing robust therapeutic efficacy of a non–dopamine targeting antipsychotic drug is an important milestone in the advance of the therapeutics of schizophrenia and other psychotic disorders,” coinvestigator Jeffrey A. Lieberman, MD, professor and chairman in the department of psychiatry, Columbia University, New York, said in an interview.
If approved, the new agent will be a “landmark” drug, Dr. Lieberman added.
The study was published in the Feb. 25, 2021, issue of the New England Journal of Medicine.
Long journey
The journey to develop an effective schizophrenia drug that reduces psychosis symptoms without onerous side effects has been a long one full of excitement and disappointment.
First-generation antipsychotics, dating back to the 1950s, targeted the postsynaptic dopamine-2 (D2) receptor. At the time, it was a “breakthrough” similar in scope to insulin for diabetes or antibiotics for infections, said Dr. Lieberman.
That was followed by development of numerous “me too” drugs with the same mechanism of action. However, these drugs had significant side effects, especially neurologic adverse events such as parkinsonism.
In 1989, second-generation antipsychotics were introduced, beginning with clozapine. They still targeted the D2 receptor but were “kinder and gentler,” Dr. Lieberman noted. “They didn’t bind to [the receptor] with such affinity that it shut things down completely, so had fewer neurologic side effects.”
However, these agents had other adverse consequences, such as weight gain and other metabolic effects including hyperglycemia and hyperlipidemia.
Many have poor functional status and quality of life despite lifelong treatment with current antipsychotic agents.
“The pharmaceutical industry, biotech industry, and academic psychiatric community have been desperately trying to find novel strategies for antipsychotic drug development and asking, ‘Is D2 the only holy grail or are there other ways of treating psychotic symptoms of schizophrenia?’ ” Dr. Lieberman said.
Enter KarXT – a novel combination of xanomeline with trospium.
An ‘ingenious’ combination
Xanomeline, an oral muscarinic cholinergic receptor agonist, does not have direct effects on the dopamine receptor. Evidence suggests the muscarinic cholinergic system is involved in the pathophysiology of schizophrenia.
However, there may be dose-dependent adverse events with the medication, such as nausea, vomiting, diarrhea, sweating, and hypersalivation from stimulation of peripheral muscarinic cholinergic receptors.
That’s where trospium chloride, an oral panmuscarinic receptor antagonist approved for treating overactive bladder, comes in. It does not reach detectable levels in the cerebrospinal fluid and should avoid adverse central nervous system effects.
Dr. Lieberman said the idea of the drug combination is “ingenious.”
The new phase 2, multisite study included adult patients with a validated diagnosis of schizophrenia who were hospitalized with an acute exacerbation of psychosis, and who were free of antipsychotic medication for at least 2 weeks.
Participants were required to have a baseline Positive and Negative Syndrome Scale (PANSS) total score of 80 points or more.
In addition to seven positive symptom items, including delusions, hallucinations, and conceptual disorganization, the PANSS has seven negative symptom items. These include restricted emotional expression, paucity of speech, and diminished interest, social drive, and activity. Each item is scored from 1 to 7, with higher scores indicating more severe symptoms.
Patients also had to have a score on the Clinical Global Impression–Severity (CGI-S) scale of 4 or higher. Scores on the CGI-S range from 1 to 7, with higher scores indicating greater severity of illness.
The modified intention-to-treat analysis included patients randomly assigned to receive oral xanomeline-trospium (n = 83) or placebo (n = 87).
The dosing schedule was flexible, starting with 50 mg of xanomeline and 20 mg of trospium twice daily. The schedule increased to a maximum of 125 mg of xanomeline and 30 mg of trospium twice daily, with the option of lowering the dose if there were unacceptable side effects.
Mean scores at baseline for the treatment and placebo groups were 97.7 versus 96.6 for the PANSS total score, 26.4 versus 26.3 for the positive subscore, 22.6 versus 22.8 for the negative subscore, and 5.0 versus 4.9 in the CGI-S scale.
‘Impressively robust’ effect size
The primary endpoint was change in the PANSS total score at 5 weeks. Results showed a change of –17.4 points in the treatment group and –5.9 points in the placebo group (least-squares mean difference, –11.6 points; 95% confidence interval, –16.1 to –7.1; P < .001).
The effect size, which was almost 0.8 (0.75), was “impressively robust,” said Dr. Lieberman, adding that a moderate effect size in this patient population might be in the order of 0.4 or 0.5.
“That gives hope that this drug may not just be as effective as other antipsychotics, albeit acting in a novel way and in a way that has a less of side effect burden, but that it may actually have some elements of superior efficacy,” he said.
There were significant benefits on some secondary outcomes, including change in the PANSS positive symptom subscore (–5.6 points in the treatment group vs. –2.4 points in the placebo group; least-squares mean difference, –3.2 points; 95% CI, –4.8 to –1.7; P < .001).
The active treatment also came out on top for CGI-S scores (P < .001), and PANSS negative symptom subscore (P < .001).
Because participants were hospitalized with an acute exacerbation of positive symptoms at time of study, it is difficult to determine “definitive efficacy” for negative symptoms, Dr. Lieberman noted. Negative symptoms may have improved simply because positive symptoms got better, he said.
Although the study included adults only, “there is nothing in the KarXT clinical profile that suggests it would be problematic for younger people,” Dr. Lieberman noted. This could include teenagers with first-episode psychosis.
Safety profile
Adverse events (AEs) were reported in 54% of the treatment group and 43% of the placebo group. AEs that were more common in the active treatment group included constipation (17% vs. 3%), nausea (17% vs. 4%), dry mouth (9% vs. 1%), dyspepsia (9% vs. 4%), and vomiting (9% vs. 4%). All AEs were rated as mild or moderate in severity and none resulted in discontinuation of treatment.
Rates of nausea, vomiting, and dry mouth were highest early in the trial and lower at the end, whereas constipation remained constant throughout the study.
Persistent constipation could affect the drug’s “utility” in elderly patients with cognitive issues but may be more of a “minor nuisance,” compared with other antipsychotics for those with schizophrenia, said Dr. Lieberman. He added that constipation might be mitigated with an over-the-counter treatment such as Metamucil. Importantly, there was no difference between groups in extrapyramidal symptoms.
In addition, participants receiving the active treatment did not have greater weight gain, which was about 3% versus 4% in the placebo group. The mean change in weight was 1.5 kg (3.3 lb) and 1.1 kg (2.4 lb), respectively.
Dr. Lieberman praised the manufacturer for undertaking the study.
“In an era when Big Pharma has retreated to a considerable degree from psychotropic drug development, it’s commendable that some companies have stayed the course and are succeeding in drug development,” he said.
Exciting mechanism
Commenting on the findings in an interview, Thomas Sedlak, MD, PhD, director of the Schizophrenia and Psychosis Consult Clinic and assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, called some aspects of the study “exciting.”
What’s especially important is that the novel agent “acts by a different mechanism than what we have been using for maybe 60 years,” said Dr. Sedlak, who was not involved with the research.
“There has been a lot of research in the last few decades into drugs that might act by alternate mechanisms of action, but nothing’s really gotten to market for schizophrenia,” he noted.
Another interesting aspect of the study is that it examined a combination drug that aims to maximize effects on the brain while minimizing periphery effects, Dr. Sedlak said.
In addition, he called the PANSS results “respectable.”
“It’s interesting that you get a big change in PANSS total score, but it’s not as big in the positive symptom score as you might have expected, and it’s not a huge drop in negative symptoms either,” said Dr. Sedlak.
It’s possible, he said, that the drug is targeting PANSS elements not captured in the main positive or negative symptom items; for example, anxiety, depression, guilt, attention, impulse control, disorientation, and judgment.
Dr. Sedlak noted that, while the new results are exciting, the enthusiasm may not be long-lasting. “When a new drug comes out, there’s often a lot of excitement about it, but once you start using it, expectations may deflate..
“More flexible ratios” of the two components of the drug might be useful to treat individual patients, he added.
In addition, using the components by themselves might be preferable for some clinicians. “I think a lot of physicians prefer using two separate drugs so they can alter the dose of each one independently and not be stuck with the ratio the manufacturer is providing,” Dr. Sedlak said.
However, he acknowledged that studying different choices of ratios would require much larger trials.
The study was supported by Karuna Therapeutics and the Wellcome Trust. Dr. Lieberman reported serving on an advisory board for Karuna Therapeutics and Intra-Cellular Therapies. Dr. Sedlak disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A novel therapy that combines a muscarinic receptor agonist with an anticholinergic agent is associated with a greater reduction in psychosis symptoms, compared with placebo, new research shows.
In a randomized, phase 2 trial composed of nearly 200 participants, xanomeline-trospium (KarXT) was generally well tolerated and had none of the common side effects linked to current antipsychotics, including weight gain and extrapyramidal symptoms such as dystonia, parkinsonism, and tardive dyskinesia.
“The results showing robust therapeutic efficacy of a non–dopamine targeting antipsychotic drug is an important milestone in the advance of the therapeutics of schizophrenia and other psychotic disorders,” coinvestigator Jeffrey A. Lieberman, MD, professor and chairman in the department of psychiatry, Columbia University, New York, said in an interview.
If approved, the new agent will be a “landmark” drug, Dr. Lieberman added.
The study was published in the Feb. 25, 2021, issue of the New England Journal of Medicine.
Long journey
The journey to develop an effective schizophrenia drug that reduces psychosis symptoms without onerous side effects has been a long one full of excitement and disappointment.
First-generation antipsychotics, dating back to the 1950s, targeted the postsynaptic dopamine-2 (D2) receptor. At the time, it was a “breakthrough” similar in scope to insulin for diabetes or antibiotics for infections, said Dr. Lieberman.
That was followed by development of numerous “me too” drugs with the same mechanism of action. However, these drugs had significant side effects, especially neurologic adverse events such as parkinsonism.
In 1989, second-generation antipsychotics were introduced, beginning with clozapine. They still targeted the D2 receptor but were “kinder and gentler,” Dr. Lieberman noted. “They didn’t bind to [the receptor] with such affinity that it shut things down completely, so had fewer neurologic side effects.”
However, these agents had other adverse consequences, such as weight gain and other metabolic effects including hyperglycemia and hyperlipidemia.
Many have poor functional status and quality of life despite lifelong treatment with current antipsychotic agents.
“The pharmaceutical industry, biotech industry, and academic psychiatric community have been desperately trying to find novel strategies for antipsychotic drug development and asking, ‘Is D2 the only holy grail or are there other ways of treating psychotic symptoms of schizophrenia?’ ” Dr. Lieberman said.
Enter KarXT – a novel combination of xanomeline with trospium.
An ‘ingenious’ combination
Xanomeline, an oral muscarinic cholinergic receptor agonist, does not have direct effects on the dopamine receptor. Evidence suggests the muscarinic cholinergic system is involved in the pathophysiology of schizophrenia.
However, there may be dose-dependent adverse events with the medication, such as nausea, vomiting, diarrhea, sweating, and hypersalivation from stimulation of peripheral muscarinic cholinergic receptors.
That’s where trospium chloride, an oral panmuscarinic receptor antagonist approved for treating overactive bladder, comes in. It does not reach detectable levels in the cerebrospinal fluid and should avoid adverse central nervous system effects.
Dr. Lieberman said the idea of the drug combination is “ingenious.”
The new phase 2, multisite study included adult patients with a validated diagnosis of schizophrenia who were hospitalized with an acute exacerbation of psychosis, and who were free of antipsychotic medication for at least 2 weeks.
Participants were required to have a baseline Positive and Negative Syndrome Scale (PANSS) total score of 80 points or more.
In addition to seven positive symptom items, including delusions, hallucinations, and conceptual disorganization, the PANSS has seven negative symptom items. These include restricted emotional expression, paucity of speech, and diminished interest, social drive, and activity. Each item is scored from 1 to 7, with higher scores indicating more severe symptoms.
Patients also had to have a score on the Clinical Global Impression–Severity (CGI-S) scale of 4 or higher. Scores on the CGI-S range from 1 to 7, with higher scores indicating greater severity of illness.
The modified intention-to-treat analysis included patients randomly assigned to receive oral xanomeline-trospium (n = 83) or placebo (n = 87).
The dosing schedule was flexible, starting with 50 mg of xanomeline and 20 mg of trospium twice daily. The schedule increased to a maximum of 125 mg of xanomeline and 30 mg of trospium twice daily, with the option of lowering the dose if there were unacceptable side effects.
Mean scores at baseline for the treatment and placebo groups were 97.7 versus 96.6 for the PANSS total score, 26.4 versus 26.3 for the positive subscore, 22.6 versus 22.8 for the negative subscore, and 5.0 versus 4.9 in the CGI-S scale.
‘Impressively robust’ effect size
The primary endpoint was change in the PANSS total score at 5 weeks. Results showed a change of –17.4 points in the treatment group and –5.9 points in the placebo group (least-squares mean difference, –11.6 points; 95% confidence interval, –16.1 to –7.1; P < .001).
The effect size, which was almost 0.8 (0.75), was “impressively robust,” said Dr. Lieberman, adding that a moderate effect size in this patient population might be in the order of 0.4 or 0.5.
“That gives hope that this drug may not just be as effective as other antipsychotics, albeit acting in a novel way and in a way that has a less of side effect burden, but that it may actually have some elements of superior efficacy,” he said.
There were significant benefits on some secondary outcomes, including change in the PANSS positive symptom subscore (–5.6 points in the treatment group vs. –2.4 points in the placebo group; least-squares mean difference, –3.2 points; 95% CI, –4.8 to –1.7; P < .001).
The active treatment also came out on top for CGI-S scores (P < .001), and PANSS negative symptom subscore (P < .001).
Because participants were hospitalized with an acute exacerbation of positive symptoms at time of study, it is difficult to determine “definitive efficacy” for negative symptoms, Dr. Lieberman noted. Negative symptoms may have improved simply because positive symptoms got better, he said.
Although the study included adults only, “there is nothing in the KarXT clinical profile that suggests it would be problematic for younger people,” Dr. Lieberman noted. This could include teenagers with first-episode psychosis.
Safety profile
Adverse events (AEs) were reported in 54% of the treatment group and 43% of the placebo group. AEs that were more common in the active treatment group included constipation (17% vs. 3%), nausea (17% vs. 4%), dry mouth (9% vs. 1%), dyspepsia (9% vs. 4%), and vomiting (9% vs. 4%). All AEs were rated as mild or moderate in severity and none resulted in discontinuation of treatment.
Rates of nausea, vomiting, and dry mouth were highest early in the trial and lower at the end, whereas constipation remained constant throughout the study.
Persistent constipation could affect the drug’s “utility” in elderly patients with cognitive issues but may be more of a “minor nuisance,” compared with other antipsychotics for those with schizophrenia, said Dr. Lieberman. He added that constipation might be mitigated with an over-the-counter treatment such as Metamucil. Importantly, there was no difference between groups in extrapyramidal symptoms.
In addition, participants receiving the active treatment did not have greater weight gain, which was about 3% versus 4% in the placebo group. The mean change in weight was 1.5 kg (3.3 lb) and 1.1 kg (2.4 lb), respectively.
Dr. Lieberman praised the manufacturer for undertaking the study.
“In an era when Big Pharma has retreated to a considerable degree from psychotropic drug development, it’s commendable that some companies have stayed the course and are succeeding in drug development,” he said.
Exciting mechanism
Commenting on the findings in an interview, Thomas Sedlak, MD, PhD, director of the Schizophrenia and Psychosis Consult Clinic and assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, called some aspects of the study “exciting.”
What’s especially important is that the novel agent “acts by a different mechanism than what we have been using for maybe 60 years,” said Dr. Sedlak, who was not involved with the research.
“There has been a lot of research in the last few decades into drugs that might act by alternate mechanisms of action, but nothing’s really gotten to market for schizophrenia,” he noted.
Another interesting aspect of the study is that it examined a combination drug that aims to maximize effects on the brain while minimizing periphery effects, Dr. Sedlak said.
In addition, he called the PANSS results “respectable.”
“It’s interesting that you get a big change in PANSS total score, but it’s not as big in the positive symptom score as you might have expected, and it’s not a huge drop in negative symptoms either,” said Dr. Sedlak.
It’s possible, he said, that the drug is targeting PANSS elements not captured in the main positive or negative symptom items; for example, anxiety, depression, guilt, attention, impulse control, disorientation, and judgment.
Dr. Sedlak noted that, while the new results are exciting, the enthusiasm may not be long-lasting. “When a new drug comes out, there’s often a lot of excitement about it, but once you start using it, expectations may deflate..
“More flexible ratios” of the two components of the drug might be useful to treat individual patients, he added.
In addition, using the components by themselves might be preferable for some clinicians. “I think a lot of physicians prefer using two separate drugs so they can alter the dose of each one independently and not be stuck with the ratio the manufacturer is providing,” Dr. Sedlak said.
However, he acknowledged that studying different choices of ratios would require much larger trials.
The study was supported by Karuna Therapeutics and the Wellcome Trust. Dr. Lieberman reported serving on an advisory board for Karuna Therapeutics and Intra-Cellular Therapies. Dr. Sedlak disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A novel therapy that combines a muscarinic receptor agonist with an anticholinergic agent is associated with a greater reduction in psychosis symptoms, compared with placebo, new research shows.
In a randomized, phase 2 trial composed of nearly 200 participants, xanomeline-trospium (KarXT) was generally well tolerated and had none of the common side effects linked to current antipsychotics, including weight gain and extrapyramidal symptoms such as dystonia, parkinsonism, and tardive dyskinesia.
“The results showing robust therapeutic efficacy of a non–dopamine targeting antipsychotic drug is an important milestone in the advance of the therapeutics of schizophrenia and other psychotic disorders,” coinvestigator Jeffrey A. Lieberman, MD, professor and chairman in the department of psychiatry, Columbia University, New York, said in an interview.
If approved, the new agent will be a “landmark” drug, Dr. Lieberman added.
The study was published in the Feb. 25, 2021, issue of the New England Journal of Medicine.
Long journey
The journey to develop an effective schizophrenia drug that reduces psychosis symptoms without onerous side effects has been a long one full of excitement and disappointment.
First-generation antipsychotics, dating back to the 1950s, targeted the postsynaptic dopamine-2 (D2) receptor. At the time, it was a “breakthrough” similar in scope to insulin for diabetes or antibiotics for infections, said Dr. Lieberman.
That was followed by development of numerous “me too” drugs with the same mechanism of action. However, these drugs had significant side effects, especially neurologic adverse events such as parkinsonism.
In 1989, second-generation antipsychotics were introduced, beginning with clozapine. They still targeted the D2 receptor but were “kinder and gentler,” Dr. Lieberman noted. “They didn’t bind to [the receptor] with such affinity that it shut things down completely, so had fewer neurologic side effects.”
However, these agents had other adverse consequences, such as weight gain and other metabolic effects including hyperglycemia and hyperlipidemia.
Many have poor functional status and quality of life despite lifelong treatment with current antipsychotic agents.
“The pharmaceutical industry, biotech industry, and academic psychiatric community have been desperately trying to find novel strategies for antipsychotic drug development and asking, ‘Is D2 the only holy grail or are there other ways of treating psychotic symptoms of schizophrenia?’ ” Dr. Lieberman said.
Enter KarXT – a novel combination of xanomeline with trospium.
An ‘ingenious’ combination
Xanomeline, an oral muscarinic cholinergic receptor agonist, does not have direct effects on the dopamine receptor. Evidence suggests the muscarinic cholinergic system is involved in the pathophysiology of schizophrenia.
However, there may be dose-dependent adverse events with the medication, such as nausea, vomiting, diarrhea, sweating, and hypersalivation from stimulation of peripheral muscarinic cholinergic receptors.
That’s where trospium chloride, an oral panmuscarinic receptor antagonist approved for treating overactive bladder, comes in. It does not reach detectable levels in the cerebrospinal fluid and should avoid adverse central nervous system effects.
Dr. Lieberman said the idea of the drug combination is “ingenious.”
The new phase 2, multisite study included adult patients with a validated diagnosis of schizophrenia who were hospitalized with an acute exacerbation of psychosis, and who were free of antipsychotic medication for at least 2 weeks.
Participants were required to have a baseline Positive and Negative Syndrome Scale (PANSS) total score of 80 points or more.
In addition to seven positive symptom items, including delusions, hallucinations, and conceptual disorganization, the PANSS has seven negative symptom items. These include restricted emotional expression, paucity of speech, and diminished interest, social drive, and activity. Each item is scored from 1 to 7, with higher scores indicating more severe symptoms.
Patients also had to have a score on the Clinical Global Impression–Severity (CGI-S) scale of 4 or higher. Scores on the CGI-S range from 1 to 7, with higher scores indicating greater severity of illness.
The modified intention-to-treat analysis included patients randomly assigned to receive oral xanomeline-trospium (n = 83) or placebo (n = 87).
The dosing schedule was flexible, starting with 50 mg of xanomeline and 20 mg of trospium twice daily. The schedule increased to a maximum of 125 mg of xanomeline and 30 mg of trospium twice daily, with the option of lowering the dose if there were unacceptable side effects.
Mean scores at baseline for the treatment and placebo groups were 97.7 versus 96.6 for the PANSS total score, 26.4 versus 26.3 for the positive subscore, 22.6 versus 22.8 for the negative subscore, and 5.0 versus 4.9 in the CGI-S scale.
‘Impressively robust’ effect size
The primary endpoint was change in the PANSS total score at 5 weeks. Results showed a change of –17.4 points in the treatment group and –5.9 points in the placebo group (least-squares mean difference, –11.6 points; 95% confidence interval, –16.1 to –7.1; P < .001).
The effect size, which was almost 0.8 (0.75), was “impressively robust,” said Dr. Lieberman, adding that a moderate effect size in this patient population might be in the order of 0.4 or 0.5.
“That gives hope that this drug may not just be as effective as other antipsychotics, albeit acting in a novel way and in a way that has a less of side effect burden, but that it may actually have some elements of superior efficacy,” he said.
There were significant benefits on some secondary outcomes, including change in the PANSS positive symptom subscore (–5.6 points in the treatment group vs. –2.4 points in the placebo group; least-squares mean difference, –3.2 points; 95% CI, –4.8 to –1.7; P < .001).
The active treatment also came out on top for CGI-S scores (P < .001), and PANSS negative symptom subscore (P < .001).
Because participants were hospitalized with an acute exacerbation of positive symptoms at time of study, it is difficult to determine “definitive efficacy” for negative symptoms, Dr. Lieberman noted. Negative symptoms may have improved simply because positive symptoms got better, he said.
Although the study included adults only, “there is nothing in the KarXT clinical profile that suggests it would be problematic for younger people,” Dr. Lieberman noted. This could include teenagers with first-episode psychosis.
Safety profile
Adverse events (AEs) were reported in 54% of the treatment group and 43% of the placebo group. AEs that were more common in the active treatment group included constipation (17% vs. 3%), nausea (17% vs. 4%), dry mouth (9% vs. 1%), dyspepsia (9% vs. 4%), and vomiting (9% vs. 4%). All AEs were rated as mild or moderate in severity and none resulted in discontinuation of treatment.
Rates of nausea, vomiting, and dry mouth were highest early in the trial and lower at the end, whereas constipation remained constant throughout the study.
Persistent constipation could affect the drug’s “utility” in elderly patients with cognitive issues but may be more of a “minor nuisance,” compared with other antipsychotics for those with schizophrenia, said Dr. Lieberman. He added that constipation might be mitigated with an over-the-counter treatment such as Metamucil. Importantly, there was no difference between groups in extrapyramidal symptoms.
In addition, participants receiving the active treatment did not have greater weight gain, which was about 3% versus 4% in the placebo group. The mean change in weight was 1.5 kg (3.3 lb) and 1.1 kg (2.4 lb), respectively.
Dr. Lieberman praised the manufacturer for undertaking the study.
“In an era when Big Pharma has retreated to a considerable degree from psychotropic drug development, it’s commendable that some companies have stayed the course and are succeeding in drug development,” he said.
Exciting mechanism
Commenting on the findings in an interview, Thomas Sedlak, MD, PhD, director of the Schizophrenia and Psychosis Consult Clinic and assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, called some aspects of the study “exciting.”
What’s especially important is that the novel agent “acts by a different mechanism than what we have been using for maybe 60 years,” said Dr. Sedlak, who was not involved with the research.
“There has been a lot of research in the last few decades into drugs that might act by alternate mechanisms of action, but nothing’s really gotten to market for schizophrenia,” he noted.
Another interesting aspect of the study is that it examined a combination drug that aims to maximize effects on the brain while minimizing periphery effects, Dr. Sedlak said.
In addition, he called the PANSS results “respectable.”
“It’s interesting that you get a big change in PANSS total score, but it’s not as big in the positive symptom score as you might have expected, and it’s not a huge drop in negative symptoms either,” said Dr. Sedlak.
It’s possible, he said, that the drug is targeting PANSS elements not captured in the main positive or negative symptom items; for example, anxiety, depression, guilt, attention, impulse control, disorientation, and judgment.
Dr. Sedlak noted that, while the new results are exciting, the enthusiasm may not be long-lasting. “When a new drug comes out, there’s often a lot of excitement about it, but once you start using it, expectations may deflate..
“More flexible ratios” of the two components of the drug might be useful to treat individual patients, he added.
In addition, using the components by themselves might be preferable for some clinicians. “I think a lot of physicians prefer using two separate drugs so they can alter the dose of each one independently and not be stuck with the ratio the manufacturer is providing,” Dr. Sedlak said.
However, he acknowledged that studying different choices of ratios would require much larger trials.
The study was supported by Karuna Therapeutics and the Wellcome Trust. Dr. Lieberman reported serving on an advisory board for Karuna Therapeutics and Intra-Cellular Therapies. Dr. Sedlak disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
















