User login
Cortical thinning in adolescence ‘definitively’ tied to subsequent psychosis
Subtle differences in brain morphometric features present in adolescence were associated with the subsequent development of psychosis in what is believed to be the largest neuroimaging investigation to date involving people at clinical high risk (CHR).
Investigators found widespread lower cortical thickness (CT) in individuals at CHR, consistent with previously reported CT differences in individuals with an established psychotic disorder.
“This is the first study to definitively show that there are subtle, widespread structural brain differences in high-risk youth before they develop psychosis,” study investigator Maria Jalbrzikowski, PhD, assistant professor of psychiatry, University of Pittsburgh, said in an interview.
The findings also suggest that there are developmental periods during which certain brain abnormalities may be more apparent, “highlighting the need to consider developmental period when developing objective, biological risk factors for early intervention in psychosis,” Dr. Jalbrzikowski said.
The study was published online May 5 in JAMA Psychiatry.
‘Sobering’ results
The findings are based on pooled structural MRI scans from 3,169 individuals recruited at 31 international sites participating in the Enhancing Neuro Imaging Genetics Through Meta-Analysis (ENIGMA) Clinical High Risk for Psychosis Working Group.
Forty-five percent of the participants were female; the mean age was 21 years (range, 9.5 to 39.9 years).
The cohort included 1,792 individuals at CHR for psychosis and 1,377 healthy control persons. Using longitudinal clinical information, the researchers identified 253 individuals at CHR who went on to develop a psychotic disorder (CHR-PS+) and 1,234 at CHR who did not develop a psychotic disorder (CHR-PS-). For the remaining 305 individuals at CHR, follow-up data were unavailable.
Compared with healthy control persons, individuals at CHR had widespread lower CT measures but not lower surface area or subcortical volume. Lower CT measures in the fusiform, superior temporal, and paracentral regions were associated with conversion to psychosis.
The pattern of differences in cortical thickness in those in the CHR-PS+ group mirrored patterns seen in people with schizophrenia and in people with 22q11.2 microdeletion syndrome who developed a psychotic disorder.
The researchers note that although all individuals experience cortical thinning as they move into adulthood, in their study, cortical thinning was already present in participants aged 12 to 16 years who developed psychosis.
“We don’t yet know exactly what this means, but adolescence is a critical time in a child’s life – it’s a time of opportunity to take risks and explore, but also a period of vulnerability,” Dr. Jalbrzikowski said in a news release.
“We could be seeing the result of something that happened even earlier in brain development but only begins to influence behavior during this developmental stage,” she noted.
This analysis represents the largest-ever pooling of brain scans in children and young adults who were determined by psychiatric assessment to be at high risk of developing psychosis.
“These results were, in a sense, sobering. On the one hand, our dataset includes 600% more high-risk youth who developed psychosis than any existing study, allowing us to see statistically significant results in brain structure.”
“But the variance between whether or not a high-risk youth develops psychosis is so small that it would be impossible to see a difference at the individual level,” Dr. Jalbrzikowski said.
More work is needed in order for the findings to be translated into clinical care, she added.
Definitive findings
Commenting on the findings for an interview, Russell Margolis, MD, clinical director, Johns Hopkins Schizophrenia Center, said that “it’s not so much that the findings are novel but rather that they’re fairly definitive in that this is by far the largest study of its kind looking at this particular question, and that gives it power. The problem with imaging studies has often been inconsistent results from study to study because of small sample size.”
“In order to see these differences in a robust way, you need a large sample size, meaning that for any one individual, this kind of structural imaging is not going to add much to the prediction of whether someone will eventually develop a schizophrenia-like illness,” said Dr. Margolis, professor of psychiatry and behavioral sciences, Johns Hopkins University, Baltimore.
From a clinical standpoint,
“The predominant hypothesis in the field is that early developmental abnormalities are the root cause of schizophrenia and related disorders, and this study is consistent with that, particularly the age-related differences, which are suggestive of neurodevelopmental abnormalities preceding the development of overt symptoms, which many other findings have also suggested,” Dr. Margolis said.
“Abnormalities in cortical thickness could be from a number of different neurobiological processes, and research into those processes are worth investigating,” he added.
The researchers received support from numerous funders, all of which are listed in the original article, along with author disclosures for ENIGMA working group members. Dr. Margolis has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Subtle differences in brain morphometric features present in adolescence were associated with the subsequent development of psychosis in what is believed to be the largest neuroimaging investigation to date involving people at clinical high risk (CHR).
Investigators found widespread lower cortical thickness (CT) in individuals at CHR, consistent with previously reported CT differences in individuals with an established psychotic disorder.
“This is the first study to definitively show that there are subtle, widespread structural brain differences in high-risk youth before they develop psychosis,” study investigator Maria Jalbrzikowski, PhD, assistant professor of psychiatry, University of Pittsburgh, said in an interview.
The findings also suggest that there are developmental periods during which certain brain abnormalities may be more apparent, “highlighting the need to consider developmental period when developing objective, biological risk factors for early intervention in psychosis,” Dr. Jalbrzikowski said.
The study was published online May 5 in JAMA Psychiatry.
‘Sobering’ results
The findings are based on pooled structural MRI scans from 3,169 individuals recruited at 31 international sites participating in the Enhancing Neuro Imaging Genetics Through Meta-Analysis (ENIGMA) Clinical High Risk for Psychosis Working Group.
Forty-five percent of the participants were female; the mean age was 21 years (range, 9.5 to 39.9 years).
The cohort included 1,792 individuals at CHR for psychosis and 1,377 healthy control persons. Using longitudinal clinical information, the researchers identified 253 individuals at CHR who went on to develop a psychotic disorder (CHR-PS+) and 1,234 at CHR who did not develop a psychotic disorder (CHR-PS-). For the remaining 305 individuals at CHR, follow-up data were unavailable.
Compared with healthy control persons, individuals at CHR had widespread lower CT measures but not lower surface area or subcortical volume. Lower CT measures in the fusiform, superior temporal, and paracentral regions were associated with conversion to psychosis.
The pattern of differences in cortical thickness in those in the CHR-PS+ group mirrored patterns seen in people with schizophrenia and in people with 22q11.2 microdeletion syndrome who developed a psychotic disorder.
The researchers note that although all individuals experience cortical thinning as they move into adulthood, in their study, cortical thinning was already present in participants aged 12 to 16 years who developed psychosis.
“We don’t yet know exactly what this means, but adolescence is a critical time in a child’s life – it’s a time of opportunity to take risks and explore, but also a period of vulnerability,” Dr. Jalbrzikowski said in a news release.
“We could be seeing the result of something that happened even earlier in brain development but only begins to influence behavior during this developmental stage,” she noted.
This analysis represents the largest-ever pooling of brain scans in children and young adults who were determined by psychiatric assessment to be at high risk of developing psychosis.
“These results were, in a sense, sobering. On the one hand, our dataset includes 600% more high-risk youth who developed psychosis than any existing study, allowing us to see statistically significant results in brain structure.”
“But the variance between whether or not a high-risk youth develops psychosis is so small that it would be impossible to see a difference at the individual level,” Dr. Jalbrzikowski said.
More work is needed in order for the findings to be translated into clinical care, she added.
Definitive findings
Commenting on the findings for an interview, Russell Margolis, MD, clinical director, Johns Hopkins Schizophrenia Center, said that “it’s not so much that the findings are novel but rather that they’re fairly definitive in that this is by far the largest study of its kind looking at this particular question, and that gives it power. The problem with imaging studies has often been inconsistent results from study to study because of small sample size.”
“In order to see these differences in a robust way, you need a large sample size, meaning that for any one individual, this kind of structural imaging is not going to add much to the prediction of whether someone will eventually develop a schizophrenia-like illness,” said Dr. Margolis, professor of psychiatry and behavioral sciences, Johns Hopkins University, Baltimore.
From a clinical standpoint,
“The predominant hypothesis in the field is that early developmental abnormalities are the root cause of schizophrenia and related disorders, and this study is consistent with that, particularly the age-related differences, which are suggestive of neurodevelopmental abnormalities preceding the development of overt symptoms, which many other findings have also suggested,” Dr. Margolis said.
“Abnormalities in cortical thickness could be from a number of different neurobiological processes, and research into those processes are worth investigating,” he added.
The researchers received support from numerous funders, all of which are listed in the original article, along with author disclosures for ENIGMA working group members. Dr. Margolis has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Subtle differences in brain morphometric features present in adolescence were associated with the subsequent development of psychosis in what is believed to be the largest neuroimaging investigation to date involving people at clinical high risk (CHR).
Investigators found widespread lower cortical thickness (CT) in individuals at CHR, consistent with previously reported CT differences in individuals with an established psychotic disorder.
“This is the first study to definitively show that there are subtle, widespread structural brain differences in high-risk youth before they develop psychosis,” study investigator Maria Jalbrzikowski, PhD, assistant professor of psychiatry, University of Pittsburgh, said in an interview.
The findings also suggest that there are developmental periods during which certain brain abnormalities may be more apparent, “highlighting the need to consider developmental period when developing objective, biological risk factors for early intervention in psychosis,” Dr. Jalbrzikowski said.
The study was published online May 5 in JAMA Psychiatry.
‘Sobering’ results
The findings are based on pooled structural MRI scans from 3,169 individuals recruited at 31 international sites participating in the Enhancing Neuro Imaging Genetics Through Meta-Analysis (ENIGMA) Clinical High Risk for Psychosis Working Group.
Forty-five percent of the participants were female; the mean age was 21 years (range, 9.5 to 39.9 years).
The cohort included 1,792 individuals at CHR for psychosis and 1,377 healthy control persons. Using longitudinal clinical information, the researchers identified 253 individuals at CHR who went on to develop a psychotic disorder (CHR-PS+) and 1,234 at CHR who did not develop a psychotic disorder (CHR-PS-). For the remaining 305 individuals at CHR, follow-up data were unavailable.
Compared with healthy control persons, individuals at CHR had widespread lower CT measures but not lower surface area or subcortical volume. Lower CT measures in the fusiform, superior temporal, and paracentral regions were associated with conversion to psychosis.
The pattern of differences in cortical thickness in those in the CHR-PS+ group mirrored patterns seen in people with schizophrenia and in people with 22q11.2 microdeletion syndrome who developed a psychotic disorder.
The researchers note that although all individuals experience cortical thinning as they move into adulthood, in their study, cortical thinning was already present in participants aged 12 to 16 years who developed psychosis.
“We don’t yet know exactly what this means, but adolescence is a critical time in a child’s life – it’s a time of opportunity to take risks and explore, but also a period of vulnerability,” Dr. Jalbrzikowski said in a news release.
“We could be seeing the result of something that happened even earlier in brain development but only begins to influence behavior during this developmental stage,” she noted.
This analysis represents the largest-ever pooling of brain scans in children and young adults who were determined by psychiatric assessment to be at high risk of developing psychosis.
“These results were, in a sense, sobering. On the one hand, our dataset includes 600% more high-risk youth who developed psychosis than any existing study, allowing us to see statistically significant results in brain structure.”
“But the variance between whether or not a high-risk youth develops psychosis is so small that it would be impossible to see a difference at the individual level,” Dr. Jalbrzikowski said.
More work is needed in order for the findings to be translated into clinical care, she added.
Definitive findings
Commenting on the findings for an interview, Russell Margolis, MD, clinical director, Johns Hopkins Schizophrenia Center, said that “it’s not so much that the findings are novel but rather that they’re fairly definitive in that this is by far the largest study of its kind looking at this particular question, and that gives it power. The problem with imaging studies has often been inconsistent results from study to study because of small sample size.”
“In order to see these differences in a robust way, you need a large sample size, meaning that for any one individual, this kind of structural imaging is not going to add much to the prediction of whether someone will eventually develop a schizophrenia-like illness,” said Dr. Margolis, professor of psychiatry and behavioral sciences, Johns Hopkins University, Baltimore.
From a clinical standpoint,
“The predominant hypothesis in the field is that early developmental abnormalities are the root cause of schizophrenia and related disorders, and this study is consistent with that, particularly the age-related differences, which are suggestive of neurodevelopmental abnormalities preceding the development of overt symptoms, which many other findings have also suggested,” Dr. Margolis said.
“Abnormalities in cortical thickness could be from a number of different neurobiological processes, and research into those processes are worth investigating,” he added.
The researchers received support from numerous funders, all of which are listed in the original article, along with author disclosures for ENIGMA working group members. Dr. Margolis has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Schizophrenia meds a key contributor to cognitive impairment
Anticholinergic medication burden from antipsychotics, antidepressants, and other psychotropics has a cumulative effect of worsening cognitive function in patients with schizophrenia, new research indicates.
“The link between long-term use of anticholinergic medications and cognitive impairment is well-known and growing,” lead researcher Yash Joshi, MD, department of psychiatry, University of California, San Diego, said in an interview.
“While this association is relevant for everyone, it is particularly important for those living with schizophrenia, who often struggle with cognitive difficulties conferred by the illness itself,” said Dr. Joshi.
“Brain health in schizophrenia is a game of inches, and even small negative effects on cognitive functioning through anticholinergic medication burden may have large impacts on patients’ lives,” he added.
The study was published online May 14 in the American Journal of Psychiatry.
‘Striking’ results
Dr. Joshi and colleagues set out to comprehensively characterize how the cumulative anticholinergic burden from different classes of medications affect cognition in patients with schizophrenia.
They assessed medical records, including all prescribed medications, for 1,120 adults with a diagnosis of schizophrenia or schizoaffective disorder.
For each participant, prescribed medications were rated and summed using a modified anticholinergic cognitive burden (ACB) scale. Cognitive functioning was assessed by performance on domains of the Penn Computerized Neurocognitive Battery (PCNB).
The investigators found that 63% of participants had an ACB score of at least 3, which is “striking,” said Dr. Joshi, given that previous studies have shown that an ACB score of 3 in a healthy, older adult is associated with cognitive dysfunction and a 50% increased risk of developing dementia.
About one-quarter of participants had an ACB score of 6 or higher.
Yet, these high ACB scores are not hard to achieve in routine psychiatric care, the researchers note.
For example, a patient taking olanzapine daily to ease symptoms of psychosis would have an ACB score of 3; if hydroxyzine was added for anxiety or insomnia, the patient’s ACB score would rise to 6, they point out.
Lightening the load
Antipsychotics contributed more than half of the anticholinergic burden, while traditional anticholinergics, antidepressants, mood stabilizers, and benzodiazepines accounted for the remainder.
“It is easy even for well-meaning clinicians to inadvertently contribute to anticholinergic medication burden through routine and appropriate care. The unique finding here is that this burden comes from medications we don’t usually think of as typical anticholinergic agents,” senior author Gregory Light, PhD, with University of California, San Diego, said in a news release.
Anticholinergic medication burden was significantly associated with generalized impairments in cognitive functioning across all cognitive domains on the PCNB with comparable magnitude and after controlling for multiple proxies of functioning or disease severity.
Higher anticholinergic medication burden was associated with worse cognitive performance. The PCNB global cognitive averages for none, low, average, high, and very high anticholinergic burdens were, respectively (in z values), -0.51, -0.70, -0.85, -0.96, and -1.15.
The results suggest “total cumulative anticholinergic burden – rather than anticholinergic burden attributable to a specific antipsychotic or psychotropic medication class – is a key contributor to cognitive impairment in schizophrenia,” the researchers write.
“The results imply that if it is clinically safe and practical,” said Dr. Joshi.
“This may be accomplished by reducing overall polypharmacy or transitioning to equivalent medications with lower overall anticholinergic burden. While ‘traditional’ anticholinergic medications should always be scrutinized, all medications should be carefully evaluated to understand whether they contribute to cumulative anticholinergic medication burden,” he added.
Confirmatory findings
Commenting on the study for this news organization, Jessica Gannon, MD, assistant professor of psychiatry, University of Pittsburgh, said the author’s findings “aren’t surprising, but the work that they did was pretty comprehensive [and] further fleshed out some of our concerns about the impact of anticholinergics on cognitive function in patients with schizophrenia.”
“We certainly have to use some of these medications for patients, like antipsychotics that do have some anticholinergic burden associated with them. We don’t really have other options,” Dr. Gannon said.
“But certainly I think this calls us to be better stewards of medication in general. And when we prescribe for comorbid conditions, like depression and anxiety, we should be careful in our prescribing practices, try not to prescribe an anticholinergic medication, and, if they have been prescribed, to deprescribe them,” Dr. Gannon added.
The study was supported by grants from the National Institute of Mental Health; the Sidney R. Baer, Jr. Foundation; the Brain and Behavior Research Foundation; the VISN-22 Mental Illness Research, Education, and Clinical Center; and the Department of Veterans Affairs. Dr. Joshi and Dr. Gannon have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Anticholinergic medication burden from antipsychotics, antidepressants, and other psychotropics has a cumulative effect of worsening cognitive function in patients with schizophrenia, new research indicates.
“The link between long-term use of anticholinergic medications and cognitive impairment is well-known and growing,” lead researcher Yash Joshi, MD, department of psychiatry, University of California, San Diego, said in an interview.
“While this association is relevant for everyone, it is particularly important for those living with schizophrenia, who often struggle with cognitive difficulties conferred by the illness itself,” said Dr. Joshi.
“Brain health in schizophrenia is a game of inches, and even small negative effects on cognitive functioning through anticholinergic medication burden may have large impacts on patients’ lives,” he added.
The study was published online May 14 in the American Journal of Psychiatry.
‘Striking’ results
Dr. Joshi and colleagues set out to comprehensively characterize how the cumulative anticholinergic burden from different classes of medications affect cognition in patients with schizophrenia.
They assessed medical records, including all prescribed medications, for 1,120 adults with a diagnosis of schizophrenia or schizoaffective disorder.
For each participant, prescribed medications were rated and summed using a modified anticholinergic cognitive burden (ACB) scale. Cognitive functioning was assessed by performance on domains of the Penn Computerized Neurocognitive Battery (PCNB).
The investigators found that 63% of participants had an ACB score of at least 3, which is “striking,” said Dr. Joshi, given that previous studies have shown that an ACB score of 3 in a healthy, older adult is associated with cognitive dysfunction and a 50% increased risk of developing dementia.
About one-quarter of participants had an ACB score of 6 or higher.
Yet, these high ACB scores are not hard to achieve in routine psychiatric care, the researchers note.
For example, a patient taking olanzapine daily to ease symptoms of psychosis would have an ACB score of 3; if hydroxyzine was added for anxiety or insomnia, the patient’s ACB score would rise to 6, they point out.
Lightening the load
Antipsychotics contributed more than half of the anticholinergic burden, while traditional anticholinergics, antidepressants, mood stabilizers, and benzodiazepines accounted for the remainder.
“It is easy even for well-meaning clinicians to inadvertently contribute to anticholinergic medication burden through routine and appropriate care. The unique finding here is that this burden comes from medications we don’t usually think of as typical anticholinergic agents,” senior author Gregory Light, PhD, with University of California, San Diego, said in a news release.
Anticholinergic medication burden was significantly associated with generalized impairments in cognitive functioning across all cognitive domains on the PCNB with comparable magnitude and after controlling for multiple proxies of functioning or disease severity.
Higher anticholinergic medication burden was associated with worse cognitive performance. The PCNB global cognitive averages for none, low, average, high, and very high anticholinergic burdens were, respectively (in z values), -0.51, -0.70, -0.85, -0.96, and -1.15.
The results suggest “total cumulative anticholinergic burden – rather than anticholinergic burden attributable to a specific antipsychotic or psychotropic medication class – is a key contributor to cognitive impairment in schizophrenia,” the researchers write.
“The results imply that if it is clinically safe and practical,” said Dr. Joshi.
“This may be accomplished by reducing overall polypharmacy or transitioning to equivalent medications with lower overall anticholinergic burden. While ‘traditional’ anticholinergic medications should always be scrutinized, all medications should be carefully evaluated to understand whether they contribute to cumulative anticholinergic medication burden,” he added.
Confirmatory findings
Commenting on the study for this news organization, Jessica Gannon, MD, assistant professor of psychiatry, University of Pittsburgh, said the author’s findings “aren’t surprising, but the work that they did was pretty comprehensive [and] further fleshed out some of our concerns about the impact of anticholinergics on cognitive function in patients with schizophrenia.”
“We certainly have to use some of these medications for patients, like antipsychotics that do have some anticholinergic burden associated with them. We don’t really have other options,” Dr. Gannon said.
“But certainly I think this calls us to be better stewards of medication in general. And when we prescribe for comorbid conditions, like depression and anxiety, we should be careful in our prescribing practices, try not to prescribe an anticholinergic medication, and, if they have been prescribed, to deprescribe them,” Dr. Gannon added.
The study was supported by grants from the National Institute of Mental Health; the Sidney R. Baer, Jr. Foundation; the Brain and Behavior Research Foundation; the VISN-22 Mental Illness Research, Education, and Clinical Center; and the Department of Veterans Affairs. Dr. Joshi and Dr. Gannon have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Anticholinergic medication burden from antipsychotics, antidepressants, and other psychotropics has a cumulative effect of worsening cognitive function in patients with schizophrenia, new research indicates.
“The link between long-term use of anticholinergic medications and cognitive impairment is well-known and growing,” lead researcher Yash Joshi, MD, department of psychiatry, University of California, San Diego, said in an interview.
“While this association is relevant for everyone, it is particularly important for those living with schizophrenia, who often struggle with cognitive difficulties conferred by the illness itself,” said Dr. Joshi.
“Brain health in schizophrenia is a game of inches, and even small negative effects on cognitive functioning through anticholinergic medication burden may have large impacts on patients’ lives,” he added.
The study was published online May 14 in the American Journal of Psychiatry.
‘Striking’ results
Dr. Joshi and colleagues set out to comprehensively characterize how the cumulative anticholinergic burden from different classes of medications affect cognition in patients with schizophrenia.
They assessed medical records, including all prescribed medications, for 1,120 adults with a diagnosis of schizophrenia or schizoaffective disorder.
For each participant, prescribed medications were rated and summed using a modified anticholinergic cognitive burden (ACB) scale. Cognitive functioning was assessed by performance on domains of the Penn Computerized Neurocognitive Battery (PCNB).
The investigators found that 63% of participants had an ACB score of at least 3, which is “striking,” said Dr. Joshi, given that previous studies have shown that an ACB score of 3 in a healthy, older adult is associated with cognitive dysfunction and a 50% increased risk of developing dementia.
About one-quarter of participants had an ACB score of 6 or higher.
Yet, these high ACB scores are not hard to achieve in routine psychiatric care, the researchers note.
For example, a patient taking olanzapine daily to ease symptoms of psychosis would have an ACB score of 3; if hydroxyzine was added for anxiety or insomnia, the patient’s ACB score would rise to 6, they point out.
Lightening the load
Antipsychotics contributed more than half of the anticholinergic burden, while traditional anticholinergics, antidepressants, mood stabilizers, and benzodiazepines accounted for the remainder.
“It is easy even for well-meaning clinicians to inadvertently contribute to anticholinergic medication burden through routine and appropriate care. The unique finding here is that this burden comes from medications we don’t usually think of as typical anticholinergic agents,” senior author Gregory Light, PhD, with University of California, San Diego, said in a news release.
Anticholinergic medication burden was significantly associated with generalized impairments in cognitive functioning across all cognitive domains on the PCNB with comparable magnitude and after controlling for multiple proxies of functioning or disease severity.
Higher anticholinergic medication burden was associated with worse cognitive performance. The PCNB global cognitive averages for none, low, average, high, and very high anticholinergic burdens were, respectively (in z values), -0.51, -0.70, -0.85, -0.96, and -1.15.
The results suggest “total cumulative anticholinergic burden – rather than anticholinergic burden attributable to a specific antipsychotic or psychotropic medication class – is a key contributor to cognitive impairment in schizophrenia,” the researchers write.
“The results imply that if it is clinically safe and practical,” said Dr. Joshi.
“This may be accomplished by reducing overall polypharmacy or transitioning to equivalent medications with lower overall anticholinergic burden. While ‘traditional’ anticholinergic medications should always be scrutinized, all medications should be carefully evaluated to understand whether they contribute to cumulative anticholinergic medication burden,” he added.
Confirmatory findings
Commenting on the study for this news organization, Jessica Gannon, MD, assistant professor of psychiatry, University of Pittsburgh, said the author’s findings “aren’t surprising, but the work that they did was pretty comprehensive [and] further fleshed out some of our concerns about the impact of anticholinergics on cognitive function in patients with schizophrenia.”
“We certainly have to use some of these medications for patients, like antipsychotics that do have some anticholinergic burden associated with them. We don’t really have other options,” Dr. Gannon said.
“But certainly I think this calls us to be better stewards of medication in general. And when we prescribe for comorbid conditions, like depression and anxiety, we should be careful in our prescribing practices, try not to prescribe an anticholinergic medication, and, if they have been prescribed, to deprescribe them,” Dr. Gannon added.
The study was supported by grants from the National Institute of Mental Health; the Sidney R. Baer, Jr. Foundation; the Brain and Behavior Research Foundation; the VISN-22 Mental Illness Research, Education, and Clinical Center; and the Department of Veterans Affairs. Dr. Joshi and Dr. Gannon have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Once-weekly oral antipsychotic for schizophrenia on the horizon
A novel, ultra–long-acting oral formulation of the antipsychotic risperidone (Risperdal) only needs to be taken once weekly and appears to be safe and effective, results of a new phase 2 study suggest.
The new formulation, LYN-005 (Lyndra Therapeutics), quickly reached therapeutic levels in patients, provided sustained exposure to risperidone active moiety over 7 days, and reduced peak drug exposure.
“This novel formulation has the potential to improve treatment adherence and quality of life in patients with schizophrenia or schizoaffective disorder,” study investigator David Walling, PhD, chief clinical officer for the Collaborative NeuroScience Network, Long Beach, Calif., said in an interview.
The findings were presented at the 2021 American Society of Clinical Psychopharmacology annual meeting.
Adherence is key
About 50% of patients don’t take medications as prescribed, creating a significant relapse risk, Dr. Walling noted.
“Here we have the possibility of having a once-weekly oral medication, which means patients don’t have to struggle with the issue of taking the medication daily. Right now, all we have on the market for long-acting medications for schizophrenia are injectables, where the patient has to go get a shot every month or every 2 weeks in order to have the medication in their system for a longer period of time,” he added.
The study included 32 clinically stable patients with a primary diagnosis of schizophrenia or schizoaffective disorder.
Patients received immediate-release (IR) risperidone at 2 mg or 4 mg, based on their current antipsychotic dose, for 13 days.
They were then randomly assigned 3:1 to receive either IR risperidone-matched placebo and LYN-005 at 14 mg or 28 mg risperidone (12 patients per group), or to LYN-005 matched placebo and IR risperidone, 2 mg or 4 mg, (4 patients per group) for 3 weeks.
LYN-05 was administered once weekly for a total of three doses. IR risperidone was administered once daily.
The study’s primary endpoints were pharmacokinetics after LYN-005 and IR risperidone and the incidence of adverse events.
Following LYN-005 administration, systemic exposure to risperidone active moiety (risperidone and 9-hydroxyrisperidone combined) increased with the increasing dose. Peak concentration occurred within the first 3 days of dosing and peak exposures from LYN-005 were lower than with IR risperidone.
“Steady state was achieved around day 15. It didn’t take 3 weeks of dosing for patients to achieve steady state. We achieved that around day 15,” Dr. Walling said.
LYN-005 was well tolerated in the 85% of study participants who received all three doses.
Adverse events occurred in 18 (75%) patients who received LYN-005. Of these, 10 were with the 14-mg dose, and 8 with the 28-mg dose.
The most common AEs were gastrointestinal, which occurred in 13 (54%) patients receiving LYN-005, with a higher incidence in the 28-mg group than in the 14 mg group.
Additionally, nine patients had abdominal pain, discomfort, or tenderness, and five patients (21%) had nausea.
Overall, the incidence of adverse events was higher for LYN-005, compared with IR risperidone, but they were judged to be mild and transitory, with fewer AEs reported with subsequent LYN-005 dosing. After the first dose, 58% of patients reported an AE; this dropped to 18% after the third dose.
An important development
Commenting on the findings, Ira D. Glick, MD, professor emeritus, Stanford (Calif.) University, said: “The major problem with schizophrenia is getting adherence.”
“The better the adherence, the better the outcome, the worst the adherence, the worse the outcome, so being able to take a preparation less often is a very important advance in the field,” said Dr. Glick, who was not involved in the research
Long-acting preparations for chronic mental illness represent a significant advance, he said.
“The future of the treatment of schizophrenia is long-acting injectables. That is the trend,” Dr. Glick explained. “The oral once-a-week preparation may be useful in a group of patients who are phobic about needles, but with the injections, you can be sure that the patient has received their medication. With the oral, there is more of a chance to be noncompliant.
“That said, having an effective, longer-acting oral for those who refuse injections is helpful. to ensure adherence to treatment. We know treatment works, we know it is going to save lives, and that’s what this ultra–long-acting formulation is promoting.”
Dr. Glick has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A novel, ultra–long-acting oral formulation of the antipsychotic risperidone (Risperdal) only needs to be taken once weekly and appears to be safe and effective, results of a new phase 2 study suggest.
The new formulation, LYN-005 (Lyndra Therapeutics), quickly reached therapeutic levels in patients, provided sustained exposure to risperidone active moiety over 7 days, and reduced peak drug exposure.
“This novel formulation has the potential to improve treatment adherence and quality of life in patients with schizophrenia or schizoaffective disorder,” study investigator David Walling, PhD, chief clinical officer for the Collaborative NeuroScience Network, Long Beach, Calif., said in an interview.
The findings were presented at the 2021 American Society of Clinical Psychopharmacology annual meeting.
Adherence is key
About 50% of patients don’t take medications as prescribed, creating a significant relapse risk, Dr. Walling noted.
“Here we have the possibility of having a once-weekly oral medication, which means patients don’t have to struggle with the issue of taking the medication daily. Right now, all we have on the market for long-acting medications for schizophrenia are injectables, where the patient has to go get a shot every month or every 2 weeks in order to have the medication in their system for a longer period of time,” he added.
The study included 32 clinically stable patients with a primary diagnosis of schizophrenia or schizoaffective disorder.
Patients received immediate-release (IR) risperidone at 2 mg or 4 mg, based on their current antipsychotic dose, for 13 days.
They were then randomly assigned 3:1 to receive either IR risperidone-matched placebo and LYN-005 at 14 mg or 28 mg risperidone (12 patients per group), or to LYN-005 matched placebo and IR risperidone, 2 mg or 4 mg, (4 patients per group) for 3 weeks.
LYN-05 was administered once weekly for a total of three doses. IR risperidone was administered once daily.
The study’s primary endpoints were pharmacokinetics after LYN-005 and IR risperidone and the incidence of adverse events.
Following LYN-005 administration, systemic exposure to risperidone active moiety (risperidone and 9-hydroxyrisperidone combined) increased with the increasing dose. Peak concentration occurred within the first 3 days of dosing and peak exposures from LYN-005 were lower than with IR risperidone.
“Steady state was achieved around day 15. It didn’t take 3 weeks of dosing for patients to achieve steady state. We achieved that around day 15,” Dr. Walling said.
LYN-005 was well tolerated in the 85% of study participants who received all three doses.
Adverse events occurred in 18 (75%) patients who received LYN-005. Of these, 10 were with the 14-mg dose, and 8 with the 28-mg dose.
The most common AEs were gastrointestinal, which occurred in 13 (54%) patients receiving LYN-005, with a higher incidence in the 28-mg group than in the 14 mg group.
Additionally, nine patients had abdominal pain, discomfort, or tenderness, and five patients (21%) had nausea.
Overall, the incidence of adverse events was higher for LYN-005, compared with IR risperidone, but they were judged to be mild and transitory, with fewer AEs reported with subsequent LYN-005 dosing. After the first dose, 58% of patients reported an AE; this dropped to 18% after the third dose.
An important development
Commenting on the findings, Ira D. Glick, MD, professor emeritus, Stanford (Calif.) University, said: “The major problem with schizophrenia is getting adherence.”
“The better the adherence, the better the outcome, the worst the adherence, the worse the outcome, so being able to take a preparation less often is a very important advance in the field,” said Dr. Glick, who was not involved in the research
Long-acting preparations for chronic mental illness represent a significant advance, he said.
“The future of the treatment of schizophrenia is long-acting injectables. That is the trend,” Dr. Glick explained. “The oral once-a-week preparation may be useful in a group of patients who are phobic about needles, but with the injections, you can be sure that the patient has received their medication. With the oral, there is more of a chance to be noncompliant.
“That said, having an effective, longer-acting oral for those who refuse injections is helpful. to ensure adherence to treatment. We know treatment works, we know it is going to save lives, and that’s what this ultra–long-acting formulation is promoting.”
Dr. Glick has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A novel, ultra–long-acting oral formulation of the antipsychotic risperidone (Risperdal) only needs to be taken once weekly and appears to be safe and effective, results of a new phase 2 study suggest.
The new formulation, LYN-005 (Lyndra Therapeutics), quickly reached therapeutic levels in patients, provided sustained exposure to risperidone active moiety over 7 days, and reduced peak drug exposure.
“This novel formulation has the potential to improve treatment adherence and quality of life in patients with schizophrenia or schizoaffective disorder,” study investigator David Walling, PhD, chief clinical officer for the Collaborative NeuroScience Network, Long Beach, Calif., said in an interview.
The findings were presented at the 2021 American Society of Clinical Psychopharmacology annual meeting.
Adherence is key
About 50% of patients don’t take medications as prescribed, creating a significant relapse risk, Dr. Walling noted.
“Here we have the possibility of having a once-weekly oral medication, which means patients don’t have to struggle with the issue of taking the medication daily. Right now, all we have on the market for long-acting medications for schizophrenia are injectables, where the patient has to go get a shot every month or every 2 weeks in order to have the medication in their system for a longer period of time,” he added.
The study included 32 clinically stable patients with a primary diagnosis of schizophrenia or schizoaffective disorder.
Patients received immediate-release (IR) risperidone at 2 mg or 4 mg, based on their current antipsychotic dose, for 13 days.
They were then randomly assigned 3:1 to receive either IR risperidone-matched placebo and LYN-005 at 14 mg or 28 mg risperidone (12 patients per group), or to LYN-005 matched placebo and IR risperidone, 2 mg or 4 mg, (4 patients per group) for 3 weeks.
LYN-05 was administered once weekly for a total of three doses. IR risperidone was administered once daily.
The study’s primary endpoints were pharmacokinetics after LYN-005 and IR risperidone and the incidence of adverse events.
Following LYN-005 administration, systemic exposure to risperidone active moiety (risperidone and 9-hydroxyrisperidone combined) increased with the increasing dose. Peak concentration occurred within the first 3 days of dosing and peak exposures from LYN-005 were lower than with IR risperidone.
“Steady state was achieved around day 15. It didn’t take 3 weeks of dosing for patients to achieve steady state. We achieved that around day 15,” Dr. Walling said.
LYN-005 was well tolerated in the 85% of study participants who received all three doses.
Adverse events occurred in 18 (75%) patients who received LYN-005. Of these, 10 were with the 14-mg dose, and 8 with the 28-mg dose.
The most common AEs were gastrointestinal, which occurred in 13 (54%) patients receiving LYN-005, with a higher incidence in the 28-mg group than in the 14 mg group.
Additionally, nine patients had abdominal pain, discomfort, or tenderness, and five patients (21%) had nausea.
Overall, the incidence of adverse events was higher for LYN-005, compared with IR risperidone, but they were judged to be mild and transitory, with fewer AEs reported with subsequent LYN-005 dosing. After the first dose, 58% of patients reported an AE; this dropped to 18% after the third dose.
An important development
Commenting on the findings, Ira D. Glick, MD, professor emeritus, Stanford (Calif.) University, said: “The major problem with schizophrenia is getting adherence.”
“The better the adherence, the better the outcome, the worst the adherence, the worse the outcome, so being able to take a preparation less often is a very important advance in the field,” said Dr. Glick, who was not involved in the research
Long-acting preparations for chronic mental illness represent a significant advance, he said.
“The future of the treatment of schizophrenia is long-acting injectables. That is the trend,” Dr. Glick explained. “The oral once-a-week preparation may be useful in a group of patients who are phobic about needles, but with the injections, you can be sure that the patient has received their medication. With the oral, there is more of a chance to be noncompliant.
“That said, having an effective, longer-acting oral for those who refuse injections is helpful. to ensure adherence to treatment. We know treatment works, we know it is going to save lives, and that’s what this ultra–long-acting formulation is promoting.”
Dr. Glick has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Emotional support animals help lick symptoms of depression, anxiety in serious mental illness
Use of emotional support animals (ESAs) yields quantifiable reductions in depression, anxiety, and loneliness for patients with serious mental illness (SMI) who live alone, early research suggests.
Investigators followed 11 community-dwelling adults with SMI who were paired with a shelter dog or cat for 1 year. Participants’ depression, anxiety, and loneliness were assessed at baseline and 12 months after receipt of their ESAs.
At regular home visits during the study, participants also underwent saliva testing before playing with their pets and after 10 minutes of enjoyable pet interaction to assess levels of oxytocin – a biomarker associated with bonding – as well as cortisol and alpha amylase, which are markers of stress.
Significant reductions in measures of anxiety, depression, and loneliness were found between baseline and 12 months for all participants. Moreover, there was a pattern of an increase in levels of oxytocin and a decrease in levels of cortisol after 10 minutes of ESA interaction, but the degree of change did not reach statistical significance.
“Although this was a small pilot study and the findings are correlational, rather than causal, we can nevertheless say from the self-report of this group of participants and from the data collected that having an emotional support animal was beneficial to their mental health,” lead author Janet Hoy-Gerlach, PhD, professor of social work, University of Toledo (Ohio), said in an interview.
“We feel this data is a strong justification for additional study, and we hope that it will be a catalyst for future research with larger samples and more rigorous methodology,” said Dr. Hoy-Gerlach, author of “Human-Animal Interaction: A Social Work Guide,” published by NASW Press in 2017.
The study was published online May 20 in Human Animal Interaction Bulletin.
Everyday interactions
An ESA is a “companion animal (pet) who helps to reduce disability-related impairment for a particular person through the animal’s presence and everyday interactions,” the authors wrote.
Unlike service animals, which perform specific functions, ESAs “provide benefits that fall along the same dimensions as the benefits of pets – physical, social, emotional, and psychological – and there is research supporting the role that animals can play in each of these arenas,” Dr. Hoy-Gerlach said.
ESAs require no special training. All that is needed is a letter from a medical or mental health professional “that the individual meets the definition of ‘disability’ under the Fair Housing Act and a companion animal is a needed disability-ameliorating accommodation and should be allowed in buildings that don’t ordinarily permit pets,” she noted.
There is currently no peer-reviewed research that focuses explicitly on the impact of ESAs in individuals with SMI. To investigate, the researchers turned to the Hope and Recovery Pet Program (HARP) – a community partnership of the University of Toledo, the Toledo Humane Society, and ProMedica, a large regional nonprofit Toledo-based health care system – that pairs community-living individuals who have depression and/or anxiety with shelter animals that require adoption. The program pays for pet food, supplies, and veterinary care for those unable to afford these.
Participants (n = 11; mean age, 53.67 years; 78% women) were recruited from the HARP program. Participants were required to be psychiatrically stable, have stable housing, live alone, be at risk for social isolation, have low income, be sober, and have no history of violence. Their primary diagnoses were major depressive disorder, bipolar disorder, and schizoaffective disorder (63%, 18%, and 18%, respectively).
Six participants adopted a cat, and five adopted a dog.
Prior to ESA adoption and at 12 months, participants completed the Beck Depression Inventory (BDI), the Beck Anxiety Inventory (BAI), and the UCLA Loneliness Scale Version 3.
Prior to ESA adoption and at 1, 3, 6, 9, and 12 months, saliva samples were collected from participants by researchers at the beginning of a home visit and then after 10 minutes of “focused pleasant interaction” with the ESA. The saliva was tested for oxytocin, alpha amylase, and cortisol.
Motivation, comfort, calm
The researchers found statistically significant decreases in UCLA Loneliness Scale scores from pre-ESA (mean [SD],59.20 [9.47]) to 12 months (49.90 [13.66], P = .004). The eta-squared statistic (.62) indicated a large effect size.
For 18 of the 20 items on the loneliness scale, mean values were lower after the intervention than before the intervention. Of these, four were statistically significant.
A statistically significant decrease in BDI total scores was also seen from pre-ESA to 12 months (21.09 [8.43] to 14.64 [7.03], respectively; P = .03). The eta-squared statistic (.41) indicated a large effect size.
Of the 21 items on the BDI scale, the mean value was lower for 19 after the intervention. Of these, five were statistically significant.
Similarly, a statistically significant decrease in BAI score was found from pre-ESA to 12 months (23.55 [9.81] to 17.73 [11.79], P = .049). The eta-squared statistic (.36) indicated a large effect size, although there were no statistically significant changes in individual item scores.
The researchers found “observable patterns” of decreases in cortisol and increases in oxytocin after the 10-minute enjoyable ESA interactions. The highest oxytocin increase occurred at 12 months; however, these improvements did not reach statistical significance.
Participants offered open-ended statements about the positive impact of their ESA on their mental health, Dr. Hoy-Gerlach said. “For example, they described feeling motivated to take better care of themselves because their ESA needed them. Some described feeling ‘comforted,’ distracted from symptoms, soothed, and calmed.
“There is definitely a place for ESAs, especially with mental health post pandemic, when we need all the resources that we can for those who can benefit,” she added.
Postpandemic mental health
Commenting on the study for this news organization, Christine Crawford, MD, MPH, assistant professor of psychiatry, Boston University, observed that ESAs “are not on the radar for a lot of clinicians, and a lot of clinicians don’t know about the science [supporting their use] or what an emotional support pet entails.
“ besides traditional forms of treatment, such as medication and therapy. Even a little relief is important, and having an emotional support pet is a good option,” said Dr. Crawford, associate medical director of the National Alliance on Mental Illness. She was not involved with the study.
The Kenneth A. Scott Charitable Trust provided grant funding. Dr. Hoy-Gerlach, her coauthors, and Dr. Crawford have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of emotional support animals (ESAs) yields quantifiable reductions in depression, anxiety, and loneliness for patients with serious mental illness (SMI) who live alone, early research suggests.
Investigators followed 11 community-dwelling adults with SMI who were paired with a shelter dog or cat for 1 year. Participants’ depression, anxiety, and loneliness were assessed at baseline and 12 months after receipt of their ESAs.
At regular home visits during the study, participants also underwent saliva testing before playing with their pets and after 10 minutes of enjoyable pet interaction to assess levels of oxytocin – a biomarker associated with bonding – as well as cortisol and alpha amylase, which are markers of stress.
Significant reductions in measures of anxiety, depression, and loneliness were found between baseline and 12 months for all participants. Moreover, there was a pattern of an increase in levels of oxytocin and a decrease in levels of cortisol after 10 minutes of ESA interaction, but the degree of change did not reach statistical significance.
“Although this was a small pilot study and the findings are correlational, rather than causal, we can nevertheless say from the self-report of this group of participants and from the data collected that having an emotional support animal was beneficial to their mental health,” lead author Janet Hoy-Gerlach, PhD, professor of social work, University of Toledo (Ohio), said in an interview.
“We feel this data is a strong justification for additional study, and we hope that it will be a catalyst for future research with larger samples and more rigorous methodology,” said Dr. Hoy-Gerlach, author of “Human-Animal Interaction: A Social Work Guide,” published by NASW Press in 2017.
The study was published online May 20 in Human Animal Interaction Bulletin.
Everyday interactions
An ESA is a “companion animal (pet) who helps to reduce disability-related impairment for a particular person through the animal’s presence and everyday interactions,” the authors wrote.
Unlike service animals, which perform specific functions, ESAs “provide benefits that fall along the same dimensions as the benefits of pets – physical, social, emotional, and psychological – and there is research supporting the role that animals can play in each of these arenas,” Dr. Hoy-Gerlach said.
ESAs require no special training. All that is needed is a letter from a medical or mental health professional “that the individual meets the definition of ‘disability’ under the Fair Housing Act and a companion animal is a needed disability-ameliorating accommodation and should be allowed in buildings that don’t ordinarily permit pets,” she noted.
There is currently no peer-reviewed research that focuses explicitly on the impact of ESAs in individuals with SMI. To investigate, the researchers turned to the Hope and Recovery Pet Program (HARP) – a community partnership of the University of Toledo, the Toledo Humane Society, and ProMedica, a large regional nonprofit Toledo-based health care system – that pairs community-living individuals who have depression and/or anxiety with shelter animals that require adoption. The program pays for pet food, supplies, and veterinary care for those unable to afford these.
Participants (n = 11; mean age, 53.67 years; 78% women) were recruited from the HARP program. Participants were required to be psychiatrically stable, have stable housing, live alone, be at risk for social isolation, have low income, be sober, and have no history of violence. Their primary diagnoses were major depressive disorder, bipolar disorder, and schizoaffective disorder (63%, 18%, and 18%, respectively).
Six participants adopted a cat, and five adopted a dog.
Prior to ESA adoption and at 12 months, participants completed the Beck Depression Inventory (BDI), the Beck Anxiety Inventory (BAI), and the UCLA Loneliness Scale Version 3.
Prior to ESA adoption and at 1, 3, 6, 9, and 12 months, saliva samples were collected from participants by researchers at the beginning of a home visit and then after 10 minutes of “focused pleasant interaction” with the ESA. The saliva was tested for oxytocin, alpha amylase, and cortisol.
Motivation, comfort, calm
The researchers found statistically significant decreases in UCLA Loneliness Scale scores from pre-ESA (mean [SD],59.20 [9.47]) to 12 months (49.90 [13.66], P = .004). The eta-squared statistic (.62) indicated a large effect size.
For 18 of the 20 items on the loneliness scale, mean values were lower after the intervention than before the intervention. Of these, four were statistically significant.
A statistically significant decrease in BDI total scores was also seen from pre-ESA to 12 months (21.09 [8.43] to 14.64 [7.03], respectively; P = .03). The eta-squared statistic (.41) indicated a large effect size.
Of the 21 items on the BDI scale, the mean value was lower for 19 after the intervention. Of these, five were statistically significant.
Similarly, a statistically significant decrease in BAI score was found from pre-ESA to 12 months (23.55 [9.81] to 17.73 [11.79], P = .049). The eta-squared statistic (.36) indicated a large effect size, although there were no statistically significant changes in individual item scores.
The researchers found “observable patterns” of decreases in cortisol and increases in oxytocin after the 10-minute enjoyable ESA interactions. The highest oxytocin increase occurred at 12 months; however, these improvements did not reach statistical significance.
Participants offered open-ended statements about the positive impact of their ESA on their mental health, Dr. Hoy-Gerlach said. “For example, they described feeling motivated to take better care of themselves because their ESA needed them. Some described feeling ‘comforted,’ distracted from symptoms, soothed, and calmed.
“There is definitely a place for ESAs, especially with mental health post pandemic, when we need all the resources that we can for those who can benefit,” she added.
Postpandemic mental health
Commenting on the study for this news organization, Christine Crawford, MD, MPH, assistant professor of psychiatry, Boston University, observed that ESAs “are not on the radar for a lot of clinicians, and a lot of clinicians don’t know about the science [supporting their use] or what an emotional support pet entails.
“ besides traditional forms of treatment, such as medication and therapy. Even a little relief is important, and having an emotional support pet is a good option,” said Dr. Crawford, associate medical director of the National Alliance on Mental Illness. She was not involved with the study.
The Kenneth A. Scott Charitable Trust provided grant funding. Dr. Hoy-Gerlach, her coauthors, and Dr. Crawford have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of emotional support animals (ESAs) yields quantifiable reductions in depression, anxiety, and loneliness for patients with serious mental illness (SMI) who live alone, early research suggests.
Investigators followed 11 community-dwelling adults with SMI who were paired with a shelter dog or cat for 1 year. Participants’ depression, anxiety, and loneliness were assessed at baseline and 12 months after receipt of their ESAs.
At regular home visits during the study, participants also underwent saliva testing before playing with their pets and after 10 minutes of enjoyable pet interaction to assess levels of oxytocin – a biomarker associated with bonding – as well as cortisol and alpha amylase, which are markers of stress.
Significant reductions in measures of anxiety, depression, and loneliness were found between baseline and 12 months for all participants. Moreover, there was a pattern of an increase in levels of oxytocin and a decrease in levels of cortisol after 10 minutes of ESA interaction, but the degree of change did not reach statistical significance.
“Although this was a small pilot study and the findings are correlational, rather than causal, we can nevertheless say from the self-report of this group of participants and from the data collected that having an emotional support animal was beneficial to their mental health,” lead author Janet Hoy-Gerlach, PhD, professor of social work, University of Toledo (Ohio), said in an interview.
“We feel this data is a strong justification for additional study, and we hope that it will be a catalyst for future research with larger samples and more rigorous methodology,” said Dr. Hoy-Gerlach, author of “Human-Animal Interaction: A Social Work Guide,” published by NASW Press in 2017.
The study was published online May 20 in Human Animal Interaction Bulletin.
Everyday interactions
An ESA is a “companion animal (pet) who helps to reduce disability-related impairment for a particular person through the animal’s presence and everyday interactions,” the authors wrote.
Unlike service animals, which perform specific functions, ESAs “provide benefits that fall along the same dimensions as the benefits of pets – physical, social, emotional, and psychological – and there is research supporting the role that animals can play in each of these arenas,” Dr. Hoy-Gerlach said.
ESAs require no special training. All that is needed is a letter from a medical or mental health professional “that the individual meets the definition of ‘disability’ under the Fair Housing Act and a companion animal is a needed disability-ameliorating accommodation and should be allowed in buildings that don’t ordinarily permit pets,” she noted.
There is currently no peer-reviewed research that focuses explicitly on the impact of ESAs in individuals with SMI. To investigate, the researchers turned to the Hope and Recovery Pet Program (HARP) – a community partnership of the University of Toledo, the Toledo Humane Society, and ProMedica, a large regional nonprofit Toledo-based health care system – that pairs community-living individuals who have depression and/or anxiety with shelter animals that require adoption. The program pays for pet food, supplies, and veterinary care for those unable to afford these.
Participants (n = 11; mean age, 53.67 years; 78% women) were recruited from the HARP program. Participants were required to be psychiatrically stable, have stable housing, live alone, be at risk for social isolation, have low income, be sober, and have no history of violence. Their primary diagnoses were major depressive disorder, bipolar disorder, and schizoaffective disorder (63%, 18%, and 18%, respectively).
Six participants adopted a cat, and five adopted a dog.
Prior to ESA adoption and at 12 months, participants completed the Beck Depression Inventory (BDI), the Beck Anxiety Inventory (BAI), and the UCLA Loneliness Scale Version 3.
Prior to ESA adoption and at 1, 3, 6, 9, and 12 months, saliva samples were collected from participants by researchers at the beginning of a home visit and then after 10 minutes of “focused pleasant interaction” with the ESA. The saliva was tested for oxytocin, alpha amylase, and cortisol.
Motivation, comfort, calm
The researchers found statistically significant decreases in UCLA Loneliness Scale scores from pre-ESA (mean [SD],59.20 [9.47]) to 12 months (49.90 [13.66], P = .004). The eta-squared statistic (.62) indicated a large effect size.
For 18 of the 20 items on the loneliness scale, mean values were lower after the intervention than before the intervention. Of these, four were statistically significant.
A statistically significant decrease in BDI total scores was also seen from pre-ESA to 12 months (21.09 [8.43] to 14.64 [7.03], respectively; P = .03). The eta-squared statistic (.41) indicated a large effect size.
Of the 21 items on the BDI scale, the mean value was lower for 19 after the intervention. Of these, five were statistically significant.
Similarly, a statistically significant decrease in BAI score was found from pre-ESA to 12 months (23.55 [9.81] to 17.73 [11.79], P = .049). The eta-squared statistic (.36) indicated a large effect size, although there were no statistically significant changes in individual item scores.
The researchers found “observable patterns” of decreases in cortisol and increases in oxytocin after the 10-minute enjoyable ESA interactions. The highest oxytocin increase occurred at 12 months; however, these improvements did not reach statistical significance.
Participants offered open-ended statements about the positive impact of their ESA on their mental health, Dr. Hoy-Gerlach said. “For example, they described feeling motivated to take better care of themselves because their ESA needed them. Some described feeling ‘comforted,’ distracted from symptoms, soothed, and calmed.
“There is definitely a place for ESAs, especially with mental health post pandemic, when we need all the resources that we can for those who can benefit,” she added.
Postpandemic mental health
Commenting on the study for this news organization, Christine Crawford, MD, MPH, assistant professor of psychiatry, Boston University, observed that ESAs “are not on the radar for a lot of clinicians, and a lot of clinicians don’t know about the science [supporting their use] or what an emotional support pet entails.
“ besides traditional forms of treatment, such as medication and therapy. Even a little relief is important, and having an emotional support pet is a good option,” said Dr. Crawford, associate medical director of the National Alliance on Mental Illness. She was not involved with the study.
The Kenneth A. Scott Charitable Trust provided grant funding. Dr. Hoy-Gerlach, her coauthors, and Dr. Crawford have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA okays new drug option for schizophrenia, bipolar I disorder
The U.S. Food and Drug Administration has approved a once-daily oral medication, which is a combination of olanzapine and samidorphan (Lybalvi, Alkermes), for the treatment of schizophrenia and bipolar I disorder.
The drug is approved for the treatment of adults with schizophrenia and for adults with bipolar I disorder as a maintenance monotherapy or to treat acute manic or mixed episodes, as either monotherapy or an adjunct to lithium or valproate.
An atypical antipsychotic, the drug is a combination of olanzapine, an established antipsychotic medication, and samidorphan, a new chemical entity.
“Schizophrenia and bipolar I disorder are complex, chronic diseases, and there remains a persistent need for new medications with proven efficacy and safety. Olanzapine, a highly efficacious atypical antipsychotic, is associated with significant side effects, including weight gain that may impact patients’ treatment experiences and limit its use. With the efficacy of olanzapine and evidence of less weight gain in patients with schizophrenia, Lybalvi brings a welcome new addition to our medication arsenal,” René S. Kahn, MD, PhD, Esther and Joseph Klingenstein professor & chair, department of psychiatry and Behavioral Health System at the Icahn School of Medicine at Mount Sinai, New York, said in a company press release.
In a clinical development program, the drug demonstrated antipsychotic efficacy, safety, and tolerability, including significantly less weight gain than olanzapine in patients with schizophrenia in the ENLIGHTEN-2 study.
The FDA approved Lybalvi under the 505(b)(2) regulatory pathway based on data from 27 clinical studies, including 18 studies evaluating Lybalvi and nine studies evaluating samidorphan alone and the FDA’s findings of the safety and effectiveness of olanzapine in the treatment of bipolar I disorder and schizophrenia. Data suggest that olanzapine-associated weight gain is disease independent, the company reports.
“People living with schizophrenia or bipolar I disorder must evaluate both efficacy and tolerability when making treatment decisions,” Paul Gionfriddo, president and CEO of Mental Health America, said in the same company press release. “We are grateful that companies like Alkermes are driven to continue developing new treatment options in psychiatry that seek to address unmet needs of our community, and we applaud the FDA for considering the experiences of individuals living with these conditions.”
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved a once-daily oral medication, which is a combination of olanzapine and samidorphan (Lybalvi, Alkermes), for the treatment of schizophrenia and bipolar I disorder.
The drug is approved for the treatment of adults with schizophrenia and for adults with bipolar I disorder as a maintenance monotherapy or to treat acute manic or mixed episodes, as either monotherapy or an adjunct to lithium or valproate.
An atypical antipsychotic, the drug is a combination of olanzapine, an established antipsychotic medication, and samidorphan, a new chemical entity.
“Schizophrenia and bipolar I disorder are complex, chronic diseases, and there remains a persistent need for new medications with proven efficacy and safety. Olanzapine, a highly efficacious atypical antipsychotic, is associated with significant side effects, including weight gain that may impact patients’ treatment experiences and limit its use. With the efficacy of olanzapine and evidence of less weight gain in patients with schizophrenia, Lybalvi brings a welcome new addition to our medication arsenal,” René S. Kahn, MD, PhD, Esther and Joseph Klingenstein professor & chair, department of psychiatry and Behavioral Health System at the Icahn School of Medicine at Mount Sinai, New York, said in a company press release.
In a clinical development program, the drug demonstrated antipsychotic efficacy, safety, and tolerability, including significantly less weight gain than olanzapine in patients with schizophrenia in the ENLIGHTEN-2 study.
The FDA approved Lybalvi under the 505(b)(2) regulatory pathway based on data from 27 clinical studies, including 18 studies evaluating Lybalvi and nine studies evaluating samidorphan alone and the FDA’s findings of the safety and effectiveness of olanzapine in the treatment of bipolar I disorder and schizophrenia. Data suggest that olanzapine-associated weight gain is disease independent, the company reports.
“People living with schizophrenia or bipolar I disorder must evaluate both efficacy and tolerability when making treatment decisions,” Paul Gionfriddo, president and CEO of Mental Health America, said in the same company press release. “We are grateful that companies like Alkermes are driven to continue developing new treatment options in psychiatry that seek to address unmet needs of our community, and we applaud the FDA for considering the experiences of individuals living with these conditions.”
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved a once-daily oral medication, which is a combination of olanzapine and samidorphan (Lybalvi, Alkermes), for the treatment of schizophrenia and bipolar I disorder.
The drug is approved for the treatment of adults with schizophrenia and for adults with bipolar I disorder as a maintenance monotherapy or to treat acute manic or mixed episodes, as either monotherapy or an adjunct to lithium or valproate.
An atypical antipsychotic, the drug is a combination of olanzapine, an established antipsychotic medication, and samidorphan, a new chemical entity.
“Schizophrenia and bipolar I disorder are complex, chronic diseases, and there remains a persistent need for new medications with proven efficacy and safety. Olanzapine, a highly efficacious atypical antipsychotic, is associated with significant side effects, including weight gain that may impact patients’ treatment experiences and limit its use. With the efficacy of olanzapine and evidence of less weight gain in patients with schizophrenia, Lybalvi brings a welcome new addition to our medication arsenal,” René S. Kahn, MD, PhD, Esther and Joseph Klingenstein professor & chair, department of psychiatry and Behavioral Health System at the Icahn School of Medicine at Mount Sinai, New York, said in a company press release.
In a clinical development program, the drug demonstrated antipsychotic efficacy, safety, and tolerability, including significantly less weight gain than olanzapine in patients with schizophrenia in the ENLIGHTEN-2 study.
The FDA approved Lybalvi under the 505(b)(2) regulatory pathway based on data from 27 clinical studies, including 18 studies evaluating Lybalvi and nine studies evaluating samidorphan alone and the FDA’s findings of the safety and effectiveness of olanzapine in the treatment of bipolar I disorder and schizophrenia. Data suggest that olanzapine-associated weight gain is disease independent, the company reports.
“People living with schizophrenia or bipolar I disorder must evaluate both efficacy and tolerability when making treatment decisions,” Paul Gionfriddo, president and CEO of Mental Health America, said in the same company press release. “We are grateful that companies like Alkermes are driven to continue developing new treatment options in psychiatry that seek to address unmet needs of our community, and we applaud the FDA for considering the experiences of individuals living with these conditions.”
A version of this article first appeared on Medscape.com.
Ketamine for acute catatonia: A case report
Ms. C, age 44, who has major depressive disorder (MDD), anxiety, obsessive-compulsive disorder (OCD) (religious subtype), and has experienced multiple episodes of treatment-resistant catatonia, is brought to the emergency department (ED) by her parents. She has immobility, mutism, rigidity, and decreased oral intake that she has experienced for 1 day.
The night before, Ms. C had been stressed about an upcoming job interview. She cancelled the interview and went to her bedroom. Later that night her parents found her lying on the floor, immobile.
Before the onset of her psychiatric symptoms, Ms. C had been high functioning. She had been an athlete in college and had a career as a school psychologist. The Sidebar summarizes Ms. C’s psychiatric history, which includes similar complex episodes and multiple hospitalizations. She also has a history of hypothyroidism.
SIDEBAR
In 2013, Ms. C experienced severe social stress from both her work as a psychologist and a divorce. She sold all of her possessions and was living in motels and hotels searching for the “truth of God.” In February 2016, she was hospitalized after refusing to eat and self-discontinuing all medications, including her thyroid medications. She was then placed under the conservatorship of her parents.
In July 2017, Ms. C was hospitalized again for refusing to eat or take her medications; this time she also exhibited selective mutism. Catatonia was suspected and she was started on oral lorazepam, 2 mg 3 times a day. Duloxetine and ziprasidone were also trialed but were stopped due to noncompliance and adverse effects. Ms. C showed little improvement on these regimens. In the hospital, IV lorazepam, 4 mg, was trialed with good effect, and she began to respond to questioning. She was transitioned to oral lorazepam, 4 mg 5 times per day, and mirtazapine, 15 mg/d. With this regimen, Ms. C became progressively more interactive; however, she still refused to eat. Throughout her hospitalization, multiple medications were prescribed, including divalproex sodium, memantine, zolpidem, olanzapine, and dextroamphetamine/levoamphetamine, all of which were not effective in stimulating her appetite. Due to malnutrition, Ms. C was placed on total parenteral nutrition. During this time, the highest dose of IM lorazepam was 20 mg/d in divided doses.
Some improvement with ECT
Four months into her hospitalization, Ms. C’s lorazepam was titrated down to 4 mg 4 times a day, and she underwent a trial of electroconvulsive therapy (ECT). Following the fourth ECT session, she displayed significant improvement. Ms. C engaged with her clinicians, displayed bright mood and affect, began eating again, and was able to recount her depressive symptoms following her divorce. At this time, she received a total of 8 ECT treatments and was started on fluoxetine. At the end of January 2018, after 19 days of hospitalization, she was transitioned to a partial hospitalization program (PHP) on a regimen of lorazepam, 2 mg 3 times daily; fluoxetine, 40 mg/d; midodrine, 10 mg 3 times daily; fludrocortisone; and levothyroxine. Her discharge diagnosis was major depressive disorder with psychotic features and catatonia.
Between her first hospitalization and her current presentation to the emergency department (ED), Ms. C presented several times to the ED with similar symptoms of decreased speech, movement, and oral intake. In February 2018, she was hospitalized and responded after 4 sessions of ECT. She returned to work as a substitute teacher and was stable for >1 year on a regimen of lorazepam, olanzapine, and risperidone. In June 2019, her symptoms returned. She was hospitalized and required a nasogastric tube to address malnutrition. She was eventually stabilized on a regimen of risperidone and lorazepam, which she continued as an outpatient until she was hospitalized again in August 2019. During this hospitalization, Ms. C failed to respond to risperidone or lorazepam, up to 2 mg 3 times a day. After several changes to her regimen, she began to respond to olanzapine, 30 mg/d; mirtazapine, 15 mg/d; and lorazepam, 2 mg 3 times a day.
Throughout her hospitalizations, once she became verbal, Ms. C demonstrated hyper-religiosity. She would ask to read the Bible, and state that her purpose was to find the truth of God. As an outpatient, she would compulsively go to church in the middle of the night and read the Bible for hours. A preliminary diagnosis of obsessive-compulsive disorder was made based on her scrupulosity, and mirtazapine was cross-titrated to fluvoxamine prior to discharge.
Shortly after discharge, she was readmitted to a PHP, and did well on fluvoxamine, 100 mg twice a day; olanzapine, 5 mg every night; levothyroxine, 100 mcg/d; and oral lorazepam, 1 mg 4 times a day. Ms. C displayed full mood, appropriate affect, and began working part-time as a substitute teacher. She had begun to interview for full-time jobs before her most recent ED presentation.
In the ED, the psychiatry team evaluates Ms. C. She displays a similar pattern of mutism, immobility, and rigidity as she did upon her initial presentation. Her father reports that she had been compliant with her medications but had not taken them the previous night. Ms. C screens positive for catatonia on the Bush-Francis Catatonia Rating Scale (BFCRS). Her severity score of 10/69 indicates a mild presentation. She is diagnosed with catatonia and is administered IV
Because Ms. C has been hospitalized many times for similar presentations, the treatment team decides to initiate a trial of IV ketamine.
Catatonia can manifest in many different ways in patients with psychiatric illness. If left untreated, it is associated with a high rate of mortality.1 Catatonia often is described along a continuum from retarded/stuporous to excited, and presentations can vary substantially. The physiologic and psychological mechanisms of catatonia are poorly understood.
Traditionally, most patients respond well to low-dose benzodiazepines, with electroconvulsive therapy as a second-line intervention for refractory and malignant cases. However, these interventions are not always successful or readily available.
Continue to: Research into the anesthetic ketamine...
Research into the anesthetic ketamine is gradually expanding, and the use of this agent for treating various psychiatric illnesses, including both unipolar and bipolar depression, has been increasing.2 Empiric evidence suggests ketamine is effective for certain psychiatric disorders, but the mechanism of action remains unclear. Although the evidence base is small, additional cases demonstrating the effectiveness of ketamine in the treatment of acute catatonia might make it a therapeutic option for use by psychiatrists and emergency medicine clinicians.
In this article, we discuss ketamine’s possible role in the treatment of catatonia, possible adverse effects, dosing strategies, and theories about ketamine’s mechanism of action.
Ketamine’s utility in psychiatry
Ketamine is a rapid-acting anesthetic that acts primarily by antagonizing N-methyl-
Previously, ketamine had been thought to induce a catatonic state, which was supported by a neurophysiologic model of catatonia that suggested the condition was caused in part by glutamate hypoactivity at the NMDA receptor.9 However, recent studies have shown that the NMDA receptor antagonists
Ketamine originally was used for sedation, and much of its safety and risk profile has been developed from decades of administration as an anesthetic. Studies have found that ketamine has a large therapeutic window in children and adults.15,16 Moreover, it does not depress the respiratory system. As an anesthetic, ketamine has a rapid onset and a quick resolution, with its sedative and disorienting effects resolving within 30 to 120 minutes.17 Ketamine’s rapid onset of action extends beyond its sedating effects. Trials with the intranasal spray esketamine for treatment-resistant depression have demonstrated an onset antidepressant effects within 2 days.18 This is much faster than that of traditional antidepressants, such as selective serotonin reuptake inhibitors.18 Based on these features, ketamine has the potential to be a useful medication in the emergency psychiatric setting, particularly for acute presentations such as catatonia.
Continue to: Beware of the potential risks
Beware of the potential risks
Although ketamine may be clinically useful, it also carries some risks. Adverse effects associated with ketamine include sedation, dissociation, hallucinations, elevated blood pressure, nausea, increased heart rate, vomiting, dizziness, fatigue, blurred vision, itching, and emesis. Clinicians also should be aware that some patients may use illicit ketamine, either as self-treatment to control depressive symptoms or for recreational purposes. When misused/abused, long-term use of ketamine can cause neurologic damage.19 Studies also have reported rare occurrences of recurrent hallucinations even after discontinuation of ketamine.20 Animal studies have demonstrated addiction and cognitive deficits with repeated use of ketamine in rodents.21 This research has led to concerns that chronic use of ketamine to treat illnesses such as depression might lead to similar long-term adverse outcomes.
Dosing
As a sedative, IV ketamine dosing is generally 1 to 2 mg/kg, and IM ketamine dosing is 3 to 5 mg/kg.16 As an antidepressant, small clinical trials have suggested that the preferred dose of IV ketamine may be 0.5 to 1 mg/kg, with dose-dependent increases in dissociation and blood pressure.21 Studies have also demonstrated that once-daily IV ketamine, 0.5 mg/kg administered over 40 minutes, led to greater improvements in patients with MDD than placebo, whereas once-daily IV ketamine, 0.2 mg/kg, did not.20
CASE CONTINUED
The team begins to treat Ms. C with IV ketamine. Ketamine, 0.2 mg/kg, is used to calculate the initial dose, and a total of 10 mg is administered over 10 minutes. Fifteen minutes after administration, Ms. C is able to move around in her bed, make eye contact, and nod to questions. She has purposeful movements, such as examining her IV line, scratching her head, and repositioning herself in the bed. After a few more minutes, she makes eye contact with her father, and nods to him during conversation. She is able to make a few noises but does not speak.
Later that day, Ms. C is discharged home (in a wheelchair) with her parents, on a medication regimen of fluvoxamine, 100 mg/d; lorazepam, 1 mg 4 times a day; and olanzapine, 5 mg/d. She is scheduled for an outpatient follow-up appointment 5 days later. Her parents are given instructions and several precautions to ensure that Ms. C receives proper nutrition until her appointment. That evening, Ms. C is able to eat voluntarily.
Five days later, Ms. C visits the outpatient psychiatric clinic and is verbal and ambulatory. Her father reports that she has become more verbal. During her follow-up interview, she is observed to be more subdued and less verbal than her baseline, but is vocal and able to voice her understanding of the treatment plan.
Continue to: After 3 months of being stable...
After 3 months of being stable on her outpatient regimen, Ms. C’s catatonic symptoms return, including refusing to eat and mutism. She is administered IV lorazepam, 4 mg, with no response and is admitted to the hospital for placement of a nasogastric feeding tube to address malnutrition. After several days, Ms. C responds to lorazepam, 4 mg every 6 hours. Six days later, after she begins eating and taking her medications voluntarily and the nasogastric tube is removed, Ms. C is discharged to home.
Findings need to be replicated in larger studies
Although some research has indicated that ketamine may be pro-catatonic, Ms. C’s improvement after receiving ketamine suggests that perhaps the situation is more complex.12,22 The exact mechanisms underlying catatonia remain uncertain. Carroll et al9 described 4 theories, and only 1 of them involved glutamate. Additionally, ketamine’s mechanism of action may extend beyond NMDA antagonism. In our case, Ms. C’s low BFCRS score during her most recent visit to the ED suggests she may have had a milder or less typical form of catatonia compared with her previous presentations (Sidebar). However, Ms. C’s clinical improvement after receiving ketamine is noteworthy.
A review of the literature yielded only 1 other case report that described using ketamine to treat catatonia.23 Iserson et al23 reported that their patient’s catatonic symptoms resolved after a total of 12.5 mg of ketamine was administered in 0.03 mg/kg boluses every 3 minutes. Compared with our own protocol, ketamine was administered at a much slower rate in this case, although both total doses of ketamine were comparable and well below the dose used for sedation. Additionally, in Iserson et al,23 lorazepam was not administered before ketamine because lorazepam was not readily available in the treatment setting. In our case, Ms. C may have had a delayed response to the IV lorazepam she received an hour before the ketamine dose; however, she exhibited a distinct clinical improvement 10 to 15 minutes after IV ketamine was administered. Nevertheless, both cases demonstrated rapid resolution of catatonic symptoms following administration of ketamine.
The marked improvement after the ketamine infusion allowed Ms. C to be discharged from the ED the same day, which was never possible after her previous catatonic episodes. Five days after discharge, she was walking, eating, talking, and able to attend to her activities of daily living without any change to her other medications. Moreover, these effects outlasted the duration of ketamine. Ms. C remained stable for 5 months until she destabilized in June 2020. At that time, she did not respond to lorazepam in the ED, needed to be hospitalized, and required a nasogastric feeding tube. Ketamine was not trialed during this presentation, so it remains to be seen if the patient’s response to ketamine was an isolated incident, or whether it could potentially spare her from future hospitalizations.
Bottom Line
In our case report, a woman with a long history of catatonia responded to a single infusion of IV ketamine, and the beneficial effects lasted for months. More research evaluating the efficacy of ketamine is needed to determine if this agent has a place in the treatment of catatonia.
Continue to: Related Resources
Related Resources
- Dubovsky SL, Dubovsky AN. Catatonia: How to identify and treat it. Current Psychiatry. 2018;17(8):16-26.
- Iserson KV, Durga D. Catatonia-like syndrome treated with low-dose ketamine. J Emerg Med. 2020;58(5):771-774.
Drug Brand Names
Amantadine • Gocovri
Dextroamphetamine sulfate/levoamphetamine sulfate • Evekeo
Divalproex sodium • Depakote
Duloxetine • Cymbalta
Esketamine • Spravato
Fluoxetine • Prozac
Fludrocortisone • Florinef
Fluvoxamine • Luvox
Ketamine • Ketalar
Levothyroxine • Synthroid
Lorazepam • Ativan
Memantine • Namenda
Mirtazapine • Remeron
Olanzapine • Zyprexa
Risperidone • Risperdal
Ziprasidone • Geodon
Zolpidem • Ambien
1. Rasmussen SA, Mazurek MF, Rosebush PI. Catatonia: our current understanding of its diagnosis, treatment and pathophysiology. World J Psychiatry. 2016;6(4):391-398.
2. Grady SE, Marsh TA, Tenhouse A, et al. Ketamine for the treatment of major depressive disorder and bipolar depression: a review of the literature. Mental Health Clin. 2017;7(1):16-23.
3. KETALAR (ketamine hydrochloride) injection. (n.d.). Accessed April 29, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/016812s043lbl.pdf
4. Williams NR, Schatzberg AF. NMDA antagonist treatment of depression. Curr Opin Neurobiol. 2016;36:112-117.
5. Parashchanka A, Schelfout S, Coppens M. Role of novel drugs in sedation outside the operating room: dexmedetomidine, ketamine and remifentanil. Curr Opin Anaesthesiol. 2014;27(4):442-447.
6. Radvansky BM, Puri S, Sifonios AN, et al. Ketamine—a narrative review of its uses in medicine. Am J Ther. 2016;23(6):e1414-e1426. doi: 10.1097/MJT.0000000000000257
7. O’Brien SL, Pangarkar S, Prager J. The use of ketamine in neuropathic pain. Current Physical Medicine and Rehabilitation Reports. 2014;2(2):128-145.
8. Swainson J, Thomas RK, Archer S, et al. Esketamine for treatment resistant depression. Expert Rev Neurother. 2019;19(10):899-911.
9. Carroll BT. The universal field hypothesis of catatonia and neuroleptic malignant syndrome. CNS Spectr. 2000;5(7):26-33.
10. Carroll BT, Goforth HW, Thomas C, et al. Review of adjunctive glutamate antagonist therapy in the treatment of catatonic syndromes. J Neuropsychiatry Clin Neurosci. 2007;19(4):406-412.
11. Northoff G, Eckert J, Fritze J. Glutamatergic dysfunction in catatonia? Successful treatment of three acute akinetic catatonic patients with the NMDA antagonist amantadine. J Neurol Neurosurg Psychiatry. 1997;62(4):404-406.
12. Denysenko L, Sica N, Penders TM, et al. Catatonia in the medically ill: etiology, diagnosis, and treatment. The Academy of Consultation-Liaison Psychiatry Evidence-Based Medicine Subcommittee Monograph. Ann Clin Psychiatry. 2018;30(2):140-155.
13. Gideons ES, Kavalali ET, Monteggia LM. Mechanisms underlying differential effectiveness of memantine and ketamine in rapid antidepressant responses. Proc Natl Acad Sci U S A. 2014;111(23):8649-8654.
14. de Bartolomeis A, Sarappa C, Buonaguro EF, et al. Different effects of the NMDA receptor antagonists ketamine, MK-801, and memantine on postsynaptic density transcripts and their topography: role of Homer signaling, and implications for novel antipsychotic and pro-cognitive targets in psychosis. Prog Neuropsychopharmacol Biol Psychiatry. 2013;46:1-12.
15. Green SM, Johnson NE. Ketamine sedation for pediatric procedures: part 2, review and implications. Ann Emerg Med. 1990;19(9):1033-1046.
16. Kurdi MS, Theerth KA, Deva RS. Ketamine: current applications in anesthesia, pain, and critical care. Anesth Essays Res. 2014;8(3):283-290.
17. Majidi S, Parna A, Zamani M, et al. Onset and effect duration of intrabuccal space and intramuscular ketamine in pediatrics. Adv Biomed Res. 2018;7:91.
18. Bahr R, Lopez A, Rey JA. Intranasal esketamine (SpravatoTM) for use in treatment-resistant depression in conjunction with an oral antidepressant. P T. 2019;44(6):340-342,344-346,375.
19. Strong CE, Kabbaj M. On the safety of repeated ketamine infusions for the treatment of depression: effects of sex and developmental periods. Neurobiol Stress. 2018;9:166-175.
20. Su TP, Chen MH, Li CT, et al. Dose-related effects of adjunctive ketamine in Taiwanese patients with treatment-resistant depression. Neuropsychopharmacology. 2017;42(13):2482-2492.
21. Fava M, Freeman MP, Flynn M, et al. Double-blind, placebo-controlled, dose-ranging trial of intravenous ketamine as adjunctive therapy in treatment-resistant depression (TRD). Mol Psychiatry. 2020;25(7):1592-1603.
22. Wong DH, Jenkins LC. An experimental study of the mechanism of action of ketamine on the central nervous system. Can Anaesth Soc J. 1974;21(1):57-67.
23. Iserson KV, Durga D. Catatonia-like syndrome treated with low-dose ketamine. J Emerg Med. 2020;58(5):771-774.
Ms. C, age 44, who has major depressive disorder (MDD), anxiety, obsessive-compulsive disorder (OCD) (religious subtype), and has experienced multiple episodes of treatment-resistant catatonia, is brought to the emergency department (ED) by her parents. She has immobility, mutism, rigidity, and decreased oral intake that she has experienced for 1 day.
The night before, Ms. C had been stressed about an upcoming job interview. She cancelled the interview and went to her bedroom. Later that night her parents found her lying on the floor, immobile.
Before the onset of her psychiatric symptoms, Ms. C had been high functioning. She had been an athlete in college and had a career as a school psychologist. The Sidebar summarizes Ms. C’s psychiatric history, which includes similar complex episodes and multiple hospitalizations. She also has a history of hypothyroidism.
SIDEBAR
In 2013, Ms. C experienced severe social stress from both her work as a psychologist and a divorce. She sold all of her possessions and was living in motels and hotels searching for the “truth of God.” In February 2016, she was hospitalized after refusing to eat and self-discontinuing all medications, including her thyroid medications. She was then placed under the conservatorship of her parents.
In July 2017, Ms. C was hospitalized again for refusing to eat or take her medications; this time she also exhibited selective mutism. Catatonia was suspected and she was started on oral lorazepam, 2 mg 3 times a day. Duloxetine and ziprasidone were also trialed but were stopped due to noncompliance and adverse effects. Ms. C showed little improvement on these regimens. In the hospital, IV lorazepam, 4 mg, was trialed with good effect, and she began to respond to questioning. She was transitioned to oral lorazepam, 4 mg 5 times per day, and mirtazapine, 15 mg/d. With this regimen, Ms. C became progressively more interactive; however, she still refused to eat. Throughout her hospitalization, multiple medications were prescribed, including divalproex sodium, memantine, zolpidem, olanzapine, and dextroamphetamine/levoamphetamine, all of which were not effective in stimulating her appetite. Due to malnutrition, Ms. C was placed on total parenteral nutrition. During this time, the highest dose of IM lorazepam was 20 mg/d in divided doses.
Some improvement with ECT
Four months into her hospitalization, Ms. C’s lorazepam was titrated down to 4 mg 4 times a day, and she underwent a trial of electroconvulsive therapy (ECT). Following the fourth ECT session, she displayed significant improvement. Ms. C engaged with her clinicians, displayed bright mood and affect, began eating again, and was able to recount her depressive symptoms following her divorce. At this time, she received a total of 8 ECT treatments and was started on fluoxetine. At the end of January 2018, after 19 days of hospitalization, she was transitioned to a partial hospitalization program (PHP) on a regimen of lorazepam, 2 mg 3 times daily; fluoxetine, 40 mg/d; midodrine, 10 mg 3 times daily; fludrocortisone; and levothyroxine. Her discharge diagnosis was major depressive disorder with psychotic features and catatonia.
Between her first hospitalization and her current presentation to the emergency department (ED), Ms. C presented several times to the ED with similar symptoms of decreased speech, movement, and oral intake. In February 2018, she was hospitalized and responded after 4 sessions of ECT. She returned to work as a substitute teacher and was stable for >1 year on a regimen of lorazepam, olanzapine, and risperidone. In June 2019, her symptoms returned. She was hospitalized and required a nasogastric tube to address malnutrition. She was eventually stabilized on a regimen of risperidone and lorazepam, which she continued as an outpatient until she was hospitalized again in August 2019. During this hospitalization, Ms. C failed to respond to risperidone or lorazepam, up to 2 mg 3 times a day. After several changes to her regimen, she began to respond to olanzapine, 30 mg/d; mirtazapine, 15 mg/d; and lorazepam, 2 mg 3 times a day.
Throughout her hospitalizations, once she became verbal, Ms. C demonstrated hyper-religiosity. She would ask to read the Bible, and state that her purpose was to find the truth of God. As an outpatient, she would compulsively go to church in the middle of the night and read the Bible for hours. A preliminary diagnosis of obsessive-compulsive disorder was made based on her scrupulosity, and mirtazapine was cross-titrated to fluvoxamine prior to discharge.
Shortly after discharge, she was readmitted to a PHP, and did well on fluvoxamine, 100 mg twice a day; olanzapine, 5 mg every night; levothyroxine, 100 mcg/d; and oral lorazepam, 1 mg 4 times a day. Ms. C displayed full mood, appropriate affect, and began working part-time as a substitute teacher. She had begun to interview for full-time jobs before her most recent ED presentation.
In the ED, the psychiatry team evaluates Ms. C. She displays a similar pattern of mutism, immobility, and rigidity as she did upon her initial presentation. Her father reports that she had been compliant with her medications but had not taken them the previous night. Ms. C screens positive for catatonia on the Bush-Francis Catatonia Rating Scale (BFCRS). Her severity score of 10/69 indicates a mild presentation. She is diagnosed with catatonia and is administered IV
Because Ms. C has been hospitalized many times for similar presentations, the treatment team decides to initiate a trial of IV ketamine.
Catatonia can manifest in many different ways in patients with psychiatric illness. If left untreated, it is associated with a high rate of mortality.1 Catatonia often is described along a continuum from retarded/stuporous to excited, and presentations can vary substantially. The physiologic and psychological mechanisms of catatonia are poorly understood.
Traditionally, most patients respond well to low-dose benzodiazepines, with electroconvulsive therapy as a second-line intervention for refractory and malignant cases. However, these interventions are not always successful or readily available.
Continue to: Research into the anesthetic ketamine...
Research into the anesthetic ketamine is gradually expanding, and the use of this agent for treating various psychiatric illnesses, including both unipolar and bipolar depression, has been increasing.2 Empiric evidence suggests ketamine is effective for certain psychiatric disorders, but the mechanism of action remains unclear. Although the evidence base is small, additional cases demonstrating the effectiveness of ketamine in the treatment of acute catatonia might make it a therapeutic option for use by psychiatrists and emergency medicine clinicians.
In this article, we discuss ketamine’s possible role in the treatment of catatonia, possible adverse effects, dosing strategies, and theories about ketamine’s mechanism of action.
Ketamine’s utility in psychiatry
Ketamine is a rapid-acting anesthetic that acts primarily by antagonizing N-methyl-
Previously, ketamine had been thought to induce a catatonic state, which was supported by a neurophysiologic model of catatonia that suggested the condition was caused in part by glutamate hypoactivity at the NMDA receptor.9 However, recent studies have shown that the NMDA receptor antagonists
Ketamine originally was used for sedation, and much of its safety and risk profile has been developed from decades of administration as an anesthetic. Studies have found that ketamine has a large therapeutic window in children and adults.15,16 Moreover, it does not depress the respiratory system. As an anesthetic, ketamine has a rapid onset and a quick resolution, with its sedative and disorienting effects resolving within 30 to 120 minutes.17 Ketamine’s rapid onset of action extends beyond its sedating effects. Trials with the intranasal spray esketamine for treatment-resistant depression have demonstrated an onset antidepressant effects within 2 days.18 This is much faster than that of traditional antidepressants, such as selective serotonin reuptake inhibitors.18 Based on these features, ketamine has the potential to be a useful medication in the emergency psychiatric setting, particularly for acute presentations such as catatonia.
Continue to: Beware of the potential risks
Beware of the potential risks
Although ketamine may be clinically useful, it also carries some risks. Adverse effects associated with ketamine include sedation, dissociation, hallucinations, elevated blood pressure, nausea, increased heart rate, vomiting, dizziness, fatigue, blurred vision, itching, and emesis. Clinicians also should be aware that some patients may use illicit ketamine, either as self-treatment to control depressive symptoms or for recreational purposes. When misused/abused, long-term use of ketamine can cause neurologic damage.19 Studies also have reported rare occurrences of recurrent hallucinations even after discontinuation of ketamine.20 Animal studies have demonstrated addiction and cognitive deficits with repeated use of ketamine in rodents.21 This research has led to concerns that chronic use of ketamine to treat illnesses such as depression might lead to similar long-term adverse outcomes.
Dosing
As a sedative, IV ketamine dosing is generally 1 to 2 mg/kg, and IM ketamine dosing is 3 to 5 mg/kg.16 As an antidepressant, small clinical trials have suggested that the preferred dose of IV ketamine may be 0.5 to 1 mg/kg, with dose-dependent increases in dissociation and blood pressure.21 Studies have also demonstrated that once-daily IV ketamine, 0.5 mg/kg administered over 40 minutes, led to greater improvements in patients with MDD than placebo, whereas once-daily IV ketamine, 0.2 mg/kg, did not.20
CASE CONTINUED
The team begins to treat Ms. C with IV ketamine. Ketamine, 0.2 mg/kg, is used to calculate the initial dose, and a total of 10 mg is administered over 10 minutes. Fifteen minutes after administration, Ms. C is able to move around in her bed, make eye contact, and nod to questions. She has purposeful movements, such as examining her IV line, scratching her head, and repositioning herself in the bed. After a few more minutes, she makes eye contact with her father, and nods to him during conversation. She is able to make a few noises but does not speak.
Later that day, Ms. C is discharged home (in a wheelchair) with her parents, on a medication regimen of fluvoxamine, 100 mg/d; lorazepam, 1 mg 4 times a day; and olanzapine, 5 mg/d. She is scheduled for an outpatient follow-up appointment 5 days later. Her parents are given instructions and several precautions to ensure that Ms. C receives proper nutrition until her appointment. That evening, Ms. C is able to eat voluntarily.
Five days later, Ms. C visits the outpatient psychiatric clinic and is verbal and ambulatory. Her father reports that she has become more verbal. During her follow-up interview, she is observed to be more subdued and less verbal than her baseline, but is vocal and able to voice her understanding of the treatment plan.
Continue to: After 3 months of being stable...
After 3 months of being stable on her outpatient regimen, Ms. C’s catatonic symptoms return, including refusing to eat and mutism. She is administered IV lorazepam, 4 mg, with no response and is admitted to the hospital for placement of a nasogastric feeding tube to address malnutrition. After several days, Ms. C responds to lorazepam, 4 mg every 6 hours. Six days later, after she begins eating and taking her medications voluntarily and the nasogastric tube is removed, Ms. C is discharged to home.
Findings need to be replicated in larger studies
Although some research has indicated that ketamine may be pro-catatonic, Ms. C’s improvement after receiving ketamine suggests that perhaps the situation is more complex.12,22 The exact mechanisms underlying catatonia remain uncertain. Carroll et al9 described 4 theories, and only 1 of them involved glutamate. Additionally, ketamine’s mechanism of action may extend beyond NMDA antagonism. In our case, Ms. C’s low BFCRS score during her most recent visit to the ED suggests she may have had a milder or less typical form of catatonia compared with her previous presentations (Sidebar). However, Ms. C’s clinical improvement after receiving ketamine is noteworthy.
A review of the literature yielded only 1 other case report that described using ketamine to treat catatonia.23 Iserson et al23 reported that their patient’s catatonic symptoms resolved after a total of 12.5 mg of ketamine was administered in 0.03 mg/kg boluses every 3 minutes. Compared with our own protocol, ketamine was administered at a much slower rate in this case, although both total doses of ketamine were comparable and well below the dose used for sedation. Additionally, in Iserson et al,23 lorazepam was not administered before ketamine because lorazepam was not readily available in the treatment setting. In our case, Ms. C may have had a delayed response to the IV lorazepam she received an hour before the ketamine dose; however, she exhibited a distinct clinical improvement 10 to 15 minutes after IV ketamine was administered. Nevertheless, both cases demonstrated rapid resolution of catatonic symptoms following administration of ketamine.
The marked improvement after the ketamine infusion allowed Ms. C to be discharged from the ED the same day, which was never possible after her previous catatonic episodes. Five days after discharge, she was walking, eating, talking, and able to attend to her activities of daily living without any change to her other medications. Moreover, these effects outlasted the duration of ketamine. Ms. C remained stable for 5 months until she destabilized in June 2020. At that time, she did not respond to lorazepam in the ED, needed to be hospitalized, and required a nasogastric feeding tube. Ketamine was not trialed during this presentation, so it remains to be seen if the patient’s response to ketamine was an isolated incident, or whether it could potentially spare her from future hospitalizations.
Bottom Line
In our case report, a woman with a long history of catatonia responded to a single infusion of IV ketamine, and the beneficial effects lasted for months. More research evaluating the efficacy of ketamine is needed to determine if this agent has a place in the treatment of catatonia.
Continue to: Related Resources
Related Resources
- Dubovsky SL, Dubovsky AN. Catatonia: How to identify and treat it. Current Psychiatry. 2018;17(8):16-26.
- Iserson KV, Durga D. Catatonia-like syndrome treated with low-dose ketamine. J Emerg Med. 2020;58(5):771-774.
Drug Brand Names
Amantadine • Gocovri
Dextroamphetamine sulfate/levoamphetamine sulfate • Evekeo
Divalproex sodium • Depakote
Duloxetine • Cymbalta
Esketamine • Spravato
Fluoxetine • Prozac
Fludrocortisone • Florinef
Fluvoxamine • Luvox
Ketamine • Ketalar
Levothyroxine • Synthroid
Lorazepam • Ativan
Memantine • Namenda
Mirtazapine • Remeron
Olanzapine • Zyprexa
Risperidone • Risperdal
Ziprasidone • Geodon
Zolpidem • Ambien
Ms. C, age 44, who has major depressive disorder (MDD), anxiety, obsessive-compulsive disorder (OCD) (religious subtype), and has experienced multiple episodes of treatment-resistant catatonia, is brought to the emergency department (ED) by her parents. She has immobility, mutism, rigidity, and decreased oral intake that she has experienced for 1 day.
The night before, Ms. C had been stressed about an upcoming job interview. She cancelled the interview and went to her bedroom. Later that night her parents found her lying on the floor, immobile.
Before the onset of her psychiatric symptoms, Ms. C had been high functioning. She had been an athlete in college and had a career as a school psychologist. The Sidebar summarizes Ms. C’s psychiatric history, which includes similar complex episodes and multiple hospitalizations. She also has a history of hypothyroidism.
SIDEBAR
In 2013, Ms. C experienced severe social stress from both her work as a psychologist and a divorce. She sold all of her possessions and was living in motels and hotels searching for the “truth of God.” In February 2016, she was hospitalized after refusing to eat and self-discontinuing all medications, including her thyroid medications. She was then placed under the conservatorship of her parents.
In July 2017, Ms. C was hospitalized again for refusing to eat or take her medications; this time she also exhibited selective mutism. Catatonia was suspected and she was started on oral lorazepam, 2 mg 3 times a day. Duloxetine and ziprasidone were also trialed but were stopped due to noncompliance and adverse effects. Ms. C showed little improvement on these regimens. In the hospital, IV lorazepam, 4 mg, was trialed with good effect, and she began to respond to questioning. She was transitioned to oral lorazepam, 4 mg 5 times per day, and mirtazapine, 15 mg/d. With this regimen, Ms. C became progressively more interactive; however, she still refused to eat. Throughout her hospitalization, multiple medications were prescribed, including divalproex sodium, memantine, zolpidem, olanzapine, and dextroamphetamine/levoamphetamine, all of which were not effective in stimulating her appetite. Due to malnutrition, Ms. C was placed on total parenteral nutrition. During this time, the highest dose of IM lorazepam was 20 mg/d in divided doses.
Some improvement with ECT
Four months into her hospitalization, Ms. C’s lorazepam was titrated down to 4 mg 4 times a day, and she underwent a trial of electroconvulsive therapy (ECT). Following the fourth ECT session, she displayed significant improvement. Ms. C engaged with her clinicians, displayed bright mood and affect, began eating again, and was able to recount her depressive symptoms following her divorce. At this time, she received a total of 8 ECT treatments and was started on fluoxetine. At the end of January 2018, after 19 days of hospitalization, she was transitioned to a partial hospitalization program (PHP) on a regimen of lorazepam, 2 mg 3 times daily; fluoxetine, 40 mg/d; midodrine, 10 mg 3 times daily; fludrocortisone; and levothyroxine. Her discharge diagnosis was major depressive disorder with psychotic features and catatonia.
Between her first hospitalization and her current presentation to the emergency department (ED), Ms. C presented several times to the ED with similar symptoms of decreased speech, movement, and oral intake. In February 2018, she was hospitalized and responded after 4 sessions of ECT. She returned to work as a substitute teacher and was stable for >1 year on a regimen of lorazepam, olanzapine, and risperidone. In June 2019, her symptoms returned. She was hospitalized and required a nasogastric tube to address malnutrition. She was eventually stabilized on a regimen of risperidone and lorazepam, which she continued as an outpatient until she was hospitalized again in August 2019. During this hospitalization, Ms. C failed to respond to risperidone or lorazepam, up to 2 mg 3 times a day. After several changes to her regimen, she began to respond to olanzapine, 30 mg/d; mirtazapine, 15 mg/d; and lorazepam, 2 mg 3 times a day.
Throughout her hospitalizations, once she became verbal, Ms. C demonstrated hyper-religiosity. She would ask to read the Bible, and state that her purpose was to find the truth of God. As an outpatient, she would compulsively go to church in the middle of the night and read the Bible for hours. A preliminary diagnosis of obsessive-compulsive disorder was made based on her scrupulosity, and mirtazapine was cross-titrated to fluvoxamine prior to discharge.
Shortly after discharge, she was readmitted to a PHP, and did well on fluvoxamine, 100 mg twice a day; olanzapine, 5 mg every night; levothyroxine, 100 mcg/d; and oral lorazepam, 1 mg 4 times a day. Ms. C displayed full mood, appropriate affect, and began working part-time as a substitute teacher. She had begun to interview for full-time jobs before her most recent ED presentation.
In the ED, the psychiatry team evaluates Ms. C. She displays a similar pattern of mutism, immobility, and rigidity as she did upon her initial presentation. Her father reports that she had been compliant with her medications but had not taken them the previous night. Ms. C screens positive for catatonia on the Bush-Francis Catatonia Rating Scale (BFCRS). Her severity score of 10/69 indicates a mild presentation. She is diagnosed with catatonia and is administered IV
Because Ms. C has been hospitalized many times for similar presentations, the treatment team decides to initiate a trial of IV ketamine.
Catatonia can manifest in many different ways in patients with psychiatric illness. If left untreated, it is associated with a high rate of mortality.1 Catatonia often is described along a continuum from retarded/stuporous to excited, and presentations can vary substantially. The physiologic and psychological mechanisms of catatonia are poorly understood.
Traditionally, most patients respond well to low-dose benzodiazepines, with electroconvulsive therapy as a second-line intervention for refractory and malignant cases. However, these interventions are not always successful or readily available.
Continue to: Research into the anesthetic ketamine...
Research into the anesthetic ketamine is gradually expanding, and the use of this agent for treating various psychiatric illnesses, including both unipolar and bipolar depression, has been increasing.2 Empiric evidence suggests ketamine is effective for certain psychiatric disorders, but the mechanism of action remains unclear. Although the evidence base is small, additional cases demonstrating the effectiveness of ketamine in the treatment of acute catatonia might make it a therapeutic option for use by psychiatrists and emergency medicine clinicians.
In this article, we discuss ketamine’s possible role in the treatment of catatonia, possible adverse effects, dosing strategies, and theories about ketamine’s mechanism of action.
Ketamine’s utility in psychiatry
Ketamine is a rapid-acting anesthetic that acts primarily by antagonizing N-methyl-
Previously, ketamine had been thought to induce a catatonic state, which was supported by a neurophysiologic model of catatonia that suggested the condition was caused in part by glutamate hypoactivity at the NMDA receptor.9 However, recent studies have shown that the NMDA receptor antagonists
Ketamine originally was used for sedation, and much of its safety and risk profile has been developed from decades of administration as an anesthetic. Studies have found that ketamine has a large therapeutic window in children and adults.15,16 Moreover, it does not depress the respiratory system. As an anesthetic, ketamine has a rapid onset and a quick resolution, with its sedative and disorienting effects resolving within 30 to 120 minutes.17 Ketamine’s rapid onset of action extends beyond its sedating effects. Trials with the intranasal spray esketamine for treatment-resistant depression have demonstrated an onset antidepressant effects within 2 days.18 This is much faster than that of traditional antidepressants, such as selective serotonin reuptake inhibitors.18 Based on these features, ketamine has the potential to be a useful medication in the emergency psychiatric setting, particularly for acute presentations such as catatonia.
Continue to: Beware of the potential risks
Beware of the potential risks
Although ketamine may be clinically useful, it also carries some risks. Adverse effects associated with ketamine include sedation, dissociation, hallucinations, elevated blood pressure, nausea, increased heart rate, vomiting, dizziness, fatigue, blurred vision, itching, and emesis. Clinicians also should be aware that some patients may use illicit ketamine, either as self-treatment to control depressive symptoms or for recreational purposes. When misused/abused, long-term use of ketamine can cause neurologic damage.19 Studies also have reported rare occurrences of recurrent hallucinations even after discontinuation of ketamine.20 Animal studies have demonstrated addiction and cognitive deficits with repeated use of ketamine in rodents.21 This research has led to concerns that chronic use of ketamine to treat illnesses such as depression might lead to similar long-term adverse outcomes.
Dosing
As a sedative, IV ketamine dosing is generally 1 to 2 mg/kg, and IM ketamine dosing is 3 to 5 mg/kg.16 As an antidepressant, small clinical trials have suggested that the preferred dose of IV ketamine may be 0.5 to 1 mg/kg, with dose-dependent increases in dissociation and blood pressure.21 Studies have also demonstrated that once-daily IV ketamine, 0.5 mg/kg administered over 40 minutes, led to greater improvements in patients with MDD than placebo, whereas once-daily IV ketamine, 0.2 mg/kg, did not.20
CASE CONTINUED
The team begins to treat Ms. C with IV ketamine. Ketamine, 0.2 mg/kg, is used to calculate the initial dose, and a total of 10 mg is administered over 10 minutes. Fifteen minutes after administration, Ms. C is able to move around in her bed, make eye contact, and nod to questions. She has purposeful movements, such as examining her IV line, scratching her head, and repositioning herself in the bed. After a few more minutes, she makes eye contact with her father, and nods to him during conversation. She is able to make a few noises but does not speak.
Later that day, Ms. C is discharged home (in a wheelchair) with her parents, on a medication regimen of fluvoxamine, 100 mg/d; lorazepam, 1 mg 4 times a day; and olanzapine, 5 mg/d. She is scheduled for an outpatient follow-up appointment 5 days later. Her parents are given instructions and several precautions to ensure that Ms. C receives proper nutrition until her appointment. That evening, Ms. C is able to eat voluntarily.
Five days later, Ms. C visits the outpatient psychiatric clinic and is verbal and ambulatory. Her father reports that she has become more verbal. During her follow-up interview, she is observed to be more subdued and less verbal than her baseline, but is vocal and able to voice her understanding of the treatment plan.
Continue to: After 3 months of being stable...
After 3 months of being stable on her outpatient regimen, Ms. C’s catatonic symptoms return, including refusing to eat and mutism. She is administered IV lorazepam, 4 mg, with no response and is admitted to the hospital for placement of a nasogastric feeding tube to address malnutrition. After several days, Ms. C responds to lorazepam, 4 mg every 6 hours. Six days later, after she begins eating and taking her medications voluntarily and the nasogastric tube is removed, Ms. C is discharged to home.
Findings need to be replicated in larger studies
Although some research has indicated that ketamine may be pro-catatonic, Ms. C’s improvement after receiving ketamine suggests that perhaps the situation is more complex.12,22 The exact mechanisms underlying catatonia remain uncertain. Carroll et al9 described 4 theories, and only 1 of them involved glutamate. Additionally, ketamine’s mechanism of action may extend beyond NMDA antagonism. In our case, Ms. C’s low BFCRS score during her most recent visit to the ED suggests she may have had a milder or less typical form of catatonia compared with her previous presentations (Sidebar). However, Ms. C’s clinical improvement after receiving ketamine is noteworthy.
A review of the literature yielded only 1 other case report that described using ketamine to treat catatonia.23 Iserson et al23 reported that their patient’s catatonic symptoms resolved after a total of 12.5 mg of ketamine was administered in 0.03 mg/kg boluses every 3 minutes. Compared with our own protocol, ketamine was administered at a much slower rate in this case, although both total doses of ketamine were comparable and well below the dose used for sedation. Additionally, in Iserson et al,23 lorazepam was not administered before ketamine because lorazepam was not readily available in the treatment setting. In our case, Ms. C may have had a delayed response to the IV lorazepam she received an hour before the ketamine dose; however, she exhibited a distinct clinical improvement 10 to 15 minutes after IV ketamine was administered. Nevertheless, both cases demonstrated rapid resolution of catatonic symptoms following administration of ketamine.
The marked improvement after the ketamine infusion allowed Ms. C to be discharged from the ED the same day, which was never possible after her previous catatonic episodes. Five days after discharge, she was walking, eating, talking, and able to attend to her activities of daily living without any change to her other medications. Moreover, these effects outlasted the duration of ketamine. Ms. C remained stable for 5 months until she destabilized in June 2020. At that time, she did not respond to lorazepam in the ED, needed to be hospitalized, and required a nasogastric feeding tube. Ketamine was not trialed during this presentation, so it remains to be seen if the patient’s response to ketamine was an isolated incident, or whether it could potentially spare her from future hospitalizations.
Bottom Line
In our case report, a woman with a long history of catatonia responded to a single infusion of IV ketamine, and the beneficial effects lasted for months. More research evaluating the efficacy of ketamine is needed to determine if this agent has a place in the treatment of catatonia.
Continue to: Related Resources
Related Resources
- Dubovsky SL, Dubovsky AN. Catatonia: How to identify and treat it. Current Psychiatry. 2018;17(8):16-26.
- Iserson KV, Durga D. Catatonia-like syndrome treated with low-dose ketamine. J Emerg Med. 2020;58(5):771-774.
Drug Brand Names
Amantadine • Gocovri
Dextroamphetamine sulfate/levoamphetamine sulfate • Evekeo
Divalproex sodium • Depakote
Duloxetine • Cymbalta
Esketamine • Spravato
Fluoxetine • Prozac
Fludrocortisone • Florinef
Fluvoxamine • Luvox
Ketamine • Ketalar
Levothyroxine • Synthroid
Lorazepam • Ativan
Memantine • Namenda
Mirtazapine • Remeron
Olanzapine • Zyprexa
Risperidone • Risperdal
Ziprasidone • Geodon
Zolpidem • Ambien
1. Rasmussen SA, Mazurek MF, Rosebush PI. Catatonia: our current understanding of its diagnosis, treatment and pathophysiology. World J Psychiatry. 2016;6(4):391-398.
2. Grady SE, Marsh TA, Tenhouse A, et al. Ketamine for the treatment of major depressive disorder and bipolar depression: a review of the literature. Mental Health Clin. 2017;7(1):16-23.
3. KETALAR (ketamine hydrochloride) injection. (n.d.). Accessed April 29, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/016812s043lbl.pdf
4. Williams NR, Schatzberg AF. NMDA antagonist treatment of depression. Curr Opin Neurobiol. 2016;36:112-117.
5. Parashchanka A, Schelfout S, Coppens M. Role of novel drugs in sedation outside the operating room: dexmedetomidine, ketamine and remifentanil. Curr Opin Anaesthesiol. 2014;27(4):442-447.
6. Radvansky BM, Puri S, Sifonios AN, et al. Ketamine—a narrative review of its uses in medicine. Am J Ther. 2016;23(6):e1414-e1426. doi: 10.1097/MJT.0000000000000257
7. O’Brien SL, Pangarkar S, Prager J. The use of ketamine in neuropathic pain. Current Physical Medicine and Rehabilitation Reports. 2014;2(2):128-145.
8. Swainson J, Thomas RK, Archer S, et al. Esketamine for treatment resistant depression. Expert Rev Neurother. 2019;19(10):899-911.
9. Carroll BT. The universal field hypothesis of catatonia and neuroleptic malignant syndrome. CNS Spectr. 2000;5(7):26-33.
10. Carroll BT, Goforth HW, Thomas C, et al. Review of adjunctive glutamate antagonist therapy in the treatment of catatonic syndromes. J Neuropsychiatry Clin Neurosci. 2007;19(4):406-412.
11. Northoff G, Eckert J, Fritze J. Glutamatergic dysfunction in catatonia? Successful treatment of three acute akinetic catatonic patients with the NMDA antagonist amantadine. J Neurol Neurosurg Psychiatry. 1997;62(4):404-406.
12. Denysenko L, Sica N, Penders TM, et al. Catatonia in the medically ill: etiology, diagnosis, and treatment. The Academy of Consultation-Liaison Psychiatry Evidence-Based Medicine Subcommittee Monograph. Ann Clin Psychiatry. 2018;30(2):140-155.
13. Gideons ES, Kavalali ET, Monteggia LM. Mechanisms underlying differential effectiveness of memantine and ketamine in rapid antidepressant responses. Proc Natl Acad Sci U S A. 2014;111(23):8649-8654.
14. de Bartolomeis A, Sarappa C, Buonaguro EF, et al. Different effects of the NMDA receptor antagonists ketamine, MK-801, and memantine on postsynaptic density transcripts and their topography: role of Homer signaling, and implications for novel antipsychotic and pro-cognitive targets in psychosis. Prog Neuropsychopharmacol Biol Psychiatry. 2013;46:1-12.
15. Green SM, Johnson NE. Ketamine sedation for pediatric procedures: part 2, review and implications. Ann Emerg Med. 1990;19(9):1033-1046.
16. Kurdi MS, Theerth KA, Deva RS. Ketamine: current applications in anesthesia, pain, and critical care. Anesth Essays Res. 2014;8(3):283-290.
17. Majidi S, Parna A, Zamani M, et al. Onset and effect duration of intrabuccal space and intramuscular ketamine in pediatrics. Adv Biomed Res. 2018;7:91.
18. Bahr R, Lopez A, Rey JA. Intranasal esketamine (SpravatoTM) for use in treatment-resistant depression in conjunction with an oral antidepressant. P T. 2019;44(6):340-342,344-346,375.
19. Strong CE, Kabbaj M. On the safety of repeated ketamine infusions for the treatment of depression: effects of sex and developmental periods. Neurobiol Stress. 2018;9:166-175.
20. Su TP, Chen MH, Li CT, et al. Dose-related effects of adjunctive ketamine in Taiwanese patients with treatment-resistant depression. Neuropsychopharmacology. 2017;42(13):2482-2492.
21. Fava M, Freeman MP, Flynn M, et al. Double-blind, placebo-controlled, dose-ranging trial of intravenous ketamine as adjunctive therapy in treatment-resistant depression (TRD). Mol Psychiatry. 2020;25(7):1592-1603.
22. Wong DH, Jenkins LC. An experimental study of the mechanism of action of ketamine on the central nervous system. Can Anaesth Soc J. 1974;21(1):57-67.
23. Iserson KV, Durga D. Catatonia-like syndrome treated with low-dose ketamine. J Emerg Med. 2020;58(5):771-774.
1. Rasmussen SA, Mazurek MF, Rosebush PI. Catatonia: our current understanding of its diagnosis, treatment and pathophysiology. World J Psychiatry. 2016;6(4):391-398.
2. Grady SE, Marsh TA, Tenhouse A, et al. Ketamine for the treatment of major depressive disorder and bipolar depression: a review of the literature. Mental Health Clin. 2017;7(1):16-23.
3. KETALAR (ketamine hydrochloride) injection. (n.d.). Accessed April 29, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/016812s043lbl.pdf
4. Williams NR, Schatzberg AF. NMDA antagonist treatment of depression. Curr Opin Neurobiol. 2016;36:112-117.
5. Parashchanka A, Schelfout S, Coppens M. Role of novel drugs in sedation outside the operating room: dexmedetomidine, ketamine and remifentanil. Curr Opin Anaesthesiol. 2014;27(4):442-447.
6. Radvansky BM, Puri S, Sifonios AN, et al. Ketamine—a narrative review of its uses in medicine. Am J Ther. 2016;23(6):e1414-e1426. doi: 10.1097/MJT.0000000000000257
7. O’Brien SL, Pangarkar S, Prager J. The use of ketamine in neuropathic pain. Current Physical Medicine and Rehabilitation Reports. 2014;2(2):128-145.
8. Swainson J, Thomas RK, Archer S, et al. Esketamine for treatment resistant depression. Expert Rev Neurother. 2019;19(10):899-911.
9. Carroll BT. The universal field hypothesis of catatonia and neuroleptic malignant syndrome. CNS Spectr. 2000;5(7):26-33.
10. Carroll BT, Goforth HW, Thomas C, et al. Review of adjunctive glutamate antagonist therapy in the treatment of catatonic syndromes. J Neuropsychiatry Clin Neurosci. 2007;19(4):406-412.
11. Northoff G, Eckert J, Fritze J. Glutamatergic dysfunction in catatonia? Successful treatment of three acute akinetic catatonic patients with the NMDA antagonist amantadine. J Neurol Neurosurg Psychiatry. 1997;62(4):404-406.
12. Denysenko L, Sica N, Penders TM, et al. Catatonia in the medically ill: etiology, diagnosis, and treatment. The Academy of Consultation-Liaison Psychiatry Evidence-Based Medicine Subcommittee Monograph. Ann Clin Psychiatry. 2018;30(2):140-155.
13. Gideons ES, Kavalali ET, Monteggia LM. Mechanisms underlying differential effectiveness of memantine and ketamine in rapid antidepressant responses. Proc Natl Acad Sci U S A. 2014;111(23):8649-8654.
14. de Bartolomeis A, Sarappa C, Buonaguro EF, et al. Different effects of the NMDA receptor antagonists ketamine, MK-801, and memantine on postsynaptic density transcripts and their topography: role of Homer signaling, and implications for novel antipsychotic and pro-cognitive targets in psychosis. Prog Neuropsychopharmacol Biol Psychiatry. 2013;46:1-12.
15. Green SM, Johnson NE. Ketamine sedation for pediatric procedures: part 2, review and implications. Ann Emerg Med. 1990;19(9):1033-1046.
16. Kurdi MS, Theerth KA, Deva RS. Ketamine: current applications in anesthesia, pain, and critical care. Anesth Essays Res. 2014;8(3):283-290.
17. Majidi S, Parna A, Zamani M, et al. Onset and effect duration of intrabuccal space and intramuscular ketamine in pediatrics. Adv Biomed Res. 2018;7:91.
18. Bahr R, Lopez A, Rey JA. Intranasal esketamine (SpravatoTM) for use in treatment-resistant depression in conjunction with an oral antidepressant. P T. 2019;44(6):340-342,344-346,375.
19. Strong CE, Kabbaj M. On the safety of repeated ketamine infusions for the treatment of depression: effects of sex and developmental periods. Neurobiol Stress. 2018;9:166-175.
20. Su TP, Chen MH, Li CT, et al. Dose-related effects of adjunctive ketamine in Taiwanese patients with treatment-resistant depression. Neuropsychopharmacology. 2017;42(13):2482-2492.
21. Fava M, Freeman MP, Flynn M, et al. Double-blind, placebo-controlled, dose-ranging trial of intravenous ketamine as adjunctive therapy in treatment-resistant depression (TRD). Mol Psychiatry. 2020;25(7):1592-1603.
22. Wong DH, Jenkins LC. An experimental study of the mechanism of action of ketamine on the central nervous system. Can Anaesth Soc J. 1974;21(1):57-67.
23. Iserson KV, Durga D. Catatonia-like syndrome treated with low-dose ketamine. J Emerg Med. 2020;58(5):771-774.
Efficacy and safety of high-dose antipsychotic therapy
Mr. K, age 21, is admitted to the psychiatry unit with agitation, disorganized behavior, and paranoia. Upon presentation, he has no known medical history or current medications. He is diagnosed with schizophrenia and subsequently tolerates but does not respond to adequate durations of treatment with fluphenazine, 20 mg/d; aripiprazole, 30 mg/d; and risperidone, 6 mg/d. Medication adherence is verified, but Mr. K is reluctant to try a fourth antipsychotic. The treatment team suspects that Mr. K may be a cytochrome P450 (CYP) 2D6 ultra-rapid metabolizer, so they obtain a serum risperidone level. The serum risperidone concentration is subtherapeutic (10 ng/mL). What should be considered next?
Several factors must be considered when a patient with psychosis does not experience significant symptomatic improvement with an adequate antipsychotic trial. This article focuses on high-dose second-generation antipsychotic (SGA) therapy in adults with psychosis. “High-dose” antipsychotic therapy is dosing that exceeds the standard maximum dosage for a given antipsychotic. Existing evidence on the use of high-dose SGAs consists of open-label studies and case reports, as well as a handful of randomized controlled trials (RCTs) with small sample sizes and high dropout rates. In some studies, the use of concomitant interventions (eg, duplicate antipsychotic therapy) limit the interpretation of data. High-dose first-generation antipsychotic therapy is discouraged because of a heightened risk of extrapyramidal symptoms (EPS).
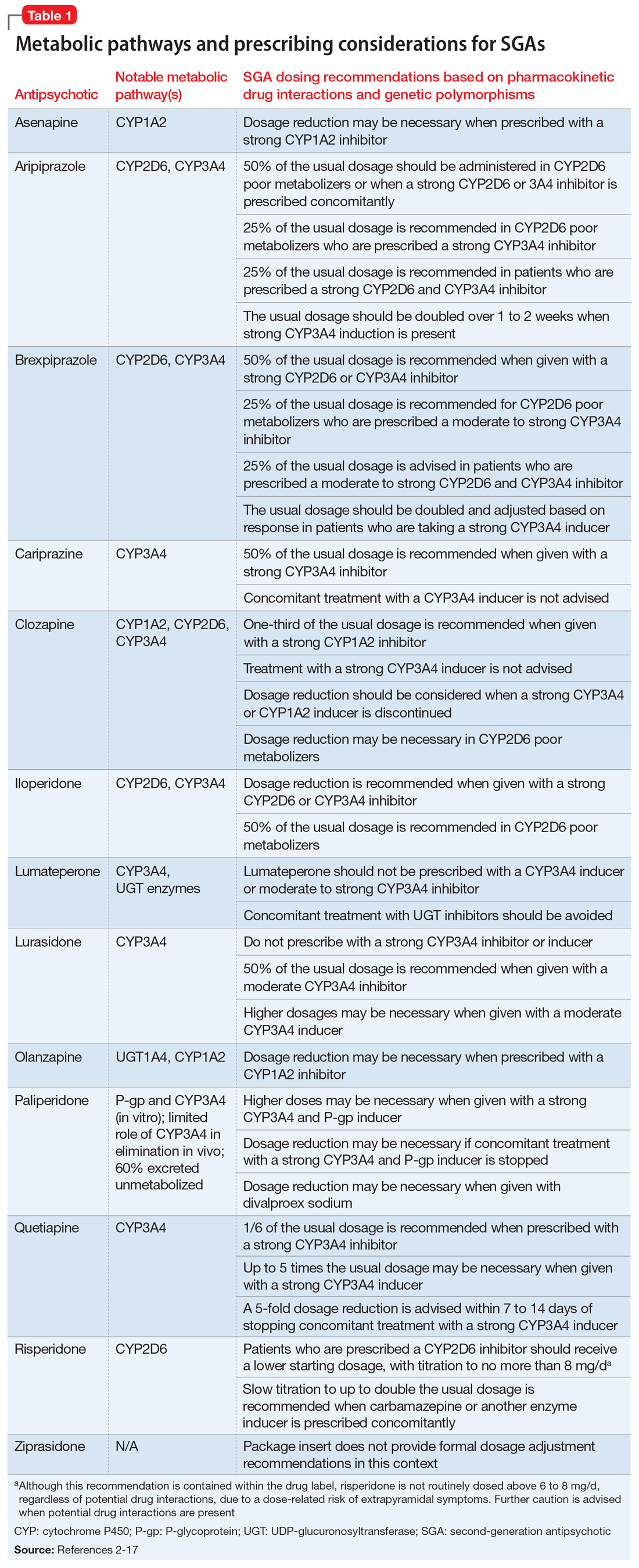
Steps to take before increasing the dose
When considering prescribing high-dose antipsychotic therapy, first confirm that the patient has been adherent to the current medication regimen. Also, screen for factors that might impair drug absorption, such as bariatric surgery or noncompliance with administration precautions.1 For example, administration of lurasidone with less than 350 calories may considerably decrease absorption.2 Dosage requirements may vary based on ethnicity, gender, CYP polymorphisms, and pharmacokinetic drug interactions (Table 12-17).1,18,19 Causes of inadequate efficacy should be addressed before considering the use of high-dose antipsychotic therapy.1 Under certain circumstances, serum drug concentrations may be used to guide antipsychotic dosing (Table 22-17). Inadequate response despite a therapeutic serum concentration may indicate pharmacodynamic failure.1 Inadequate response in the context of subtherapeutic serum concentrations, good medication adherence, and compliance to administration precautions may be indicative of a genetic polymorphism or drug interaction.1 Changes in antipsychotic dosing or selection may be warranted, depending on associated risks and benefits.
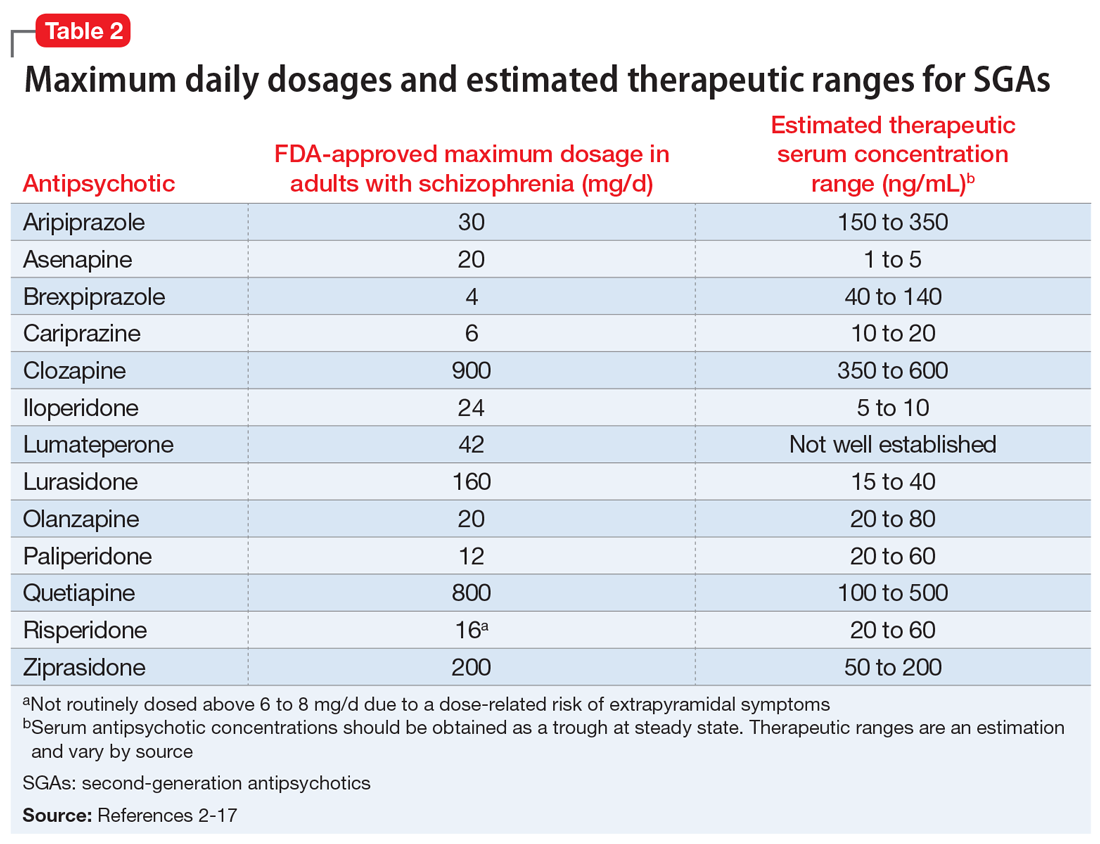
SGAs and high-dose administration
The SGA with the greatest evidence for high-dose administration is olanzapine, which is similar in structure and receptor pharmacology to clozapine.20,21 The use of high-dose olanzapine is controversial. High-dose olanzapine has been compared to clozapine in patients with treatment-resistant schizophrenia (TRS) and schizoaffective disorder. Meltzer et al22 reported similar efficacy with clozapine, 300 to 900 mg/d, and olanzapine, 25 to 45 mg/d. In this study, high-dose olanzapine caused more weight gain when compared to clozapine. Olanzapine dosages of up to 100 mg/d have been prescribed for TRS; however, this is not common practice.23 A study comparing 10, 20, and 40 mg/d in patients with non-TRS or schizoaffective disorder showed no advantage with higher dosages.24
There is limited data on high-dose treatment with other SGAs.17 Orthostasis may limit iloperidone’s safety at high doses, and single doses of asenapine should not exceed 10 mg.25 Limited sublingual surface area and saliva saturation result in decreased bioavailability with higher asenapine doses.25,26 In a small RCT of patients with stable schizophrenia or schizoaffective disorder, aripiprazole was relatively well-tolerated up to 75 mg/d, whereas akathisia and tachycardia occurred with 90 mg/d.27 Case reports have documented successful treatment with aripiprazole, 60 to 75 mg/d; however, dizziness and worsening psychosis, agitation, and confusion have been observed.28-31
There is a paucity of data on high-dose risperidone and paliperidone, possibly due to their potent dopamine-2 (D2) receptor antagonism and dose-related risk of EPS.1 At risperidone dosages >6 mg/d, the balance between D2 and serotonin-2A (5-HT2A) receptor potency is lost, which increases the potential for EPS.32 In one RCT, long-acting injectable (LAI) risperidone, up to 100 mg biweekly, was well-tolerated but no more effective for TRS than 50 mg biweekly.33 A case report suggested improvement of TRS in a patient administered risperidone LAI, 75 mg vs 37.5 mg biweekly, but it is unclear if a 50-mg dosage was tried.34 Another case report documented improvement in schizophrenia symptoms with risperidone LAI, 125 mg biweekly; however, anticholinergic therapy was required for EPS.35
Dose-dependent adverse effects, including EPS, sedation, anticholinergic effects, orthostasis, hyperprolactinemia, and QTc prolongation, may limit the safety of high-dose antipsychotic therapy.1,20,36 Two studies showed no correlation between QTc prolongation and ziprasidone dosages of up to 320 mg/d for psychosis.37,38 QTc prolongation was more likely at higher ziprasidone concentrations.37 Higher concentrations, but not higher dosages, also trended toward improvement in positive symptoms, and concentrations >100 ng/mL were associated with more negative symptoms.37 A case report described improvement in positive symptoms of schizoaffective disorder with ziprasidone, 320 mg/d, but activation, hostility, and depression worsened.39
Continue to: Compared with other antipsychotics...
Compared with other antipsychotics, high-dose clozapine and quetiapine may be less likely to cause EPS due to lower D2 receptor occupancies.40 Nevertheless, increased activity at other postsynaptic receptors may lead to constipation, metabolic effects, and sedation.1,41,42 Case reports suggest efficacy with quetiapine, 1,200 to 2,400 mg/d, vs lower dosages for patients with TRS.43,44 However, RCTs of quetiapine, 600 and 800 mg/d vs 1,200 mg/d, have not demonstrated an efficacy advantage with high-dose treatment in patients with schizophrenia or schizoaffective disorder.41,45 High-dose quetiapine has also resulted in photopsia, cardiotoxicity, orthostasis, dysphagia, and sedation.43,46,47
Proceed with caution
In light of safety concerns and a lack of high-quality evidence for high-dose antipsychotic therapy, alternative solutions for inadequate response to treatment should be considered. Underlying causes of poor response should be addressed, and alternative antipsychotics should be utilized, when appropriate. A clozapine trial remains first-line for TRS. Olanzapine may be the best-supported high-dose antipsychotic alternative when clozapine is not an option. High antipsychotic dosages are not well-studied in patients with genetic polymorphisms or unavoidable drug interactions. Serum antipsychotic concentrations may facilitate dosing in these patients.
If high-dose antipsychotic therapy is deemed necessary, its ongoing appropriateness should be continually re-evaluated. Higher antipsychotic dosages and D2 receptor occupancies may be required to manage acute psychosis, but efficacy may be maintained and adverse effects limited with the use of lower dosages during maintenance treatment.48,49 Long-term treatment with high-dose antipsychotic therapy should be avoided, when possible. If high-dose antipsychotic therapy is prescribed, the rationale should be well-documented. Dosage adjustments should not be made until steady state is reached on a given dosage. Electrocardiograms should be obtained at baseline, steady state, and routinely thereafter.3,20 Tolerability should be assessed regularly, and screening for drug interactions should be conducted when new medications are initiated.
Case CONTINUED
Because Mr. K’s serum risperidone level is subtherapeutic (10 ng/mL), his risperidone dosage is cautiously titrated to 10 mg/d, divided (serum concentration: 22 ng/mL). Mr. K develops mild orthostasis but denies other adverse effects. His psychotic symptoms resolve, and he is discharged with education on nonpharmacologic management of orthostasis. The rationale for high-dose risperidone is relayed to his outpatient psychiatrist, as well as a recommendation to monitor Mr. K closely for continued efficacy and tolerability.
Related Resource
- Barnes TRE, Drake R, Paton C, et al. Evidence-based guidelines for the pharmacological treatment of schizophrenia: updated recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2020;34(1):3-78.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Brexpiprazole • Rexulti
Cariprazine • Vraylar
Clozapine • Clozaril
Iloperidone • Fanapt
Lumateperone • Caplyta
Lurasidone • Latuda
Olanzapine • Zyprexa
Paliperidone • Invega
Quetiapine • Seroquel
Risperidone • Risperdal
Risperidone long-acting injection • Risperdal Consta
Ziprasidone • Geodon
1. Morrissette DA, Stahl SM. Treating the violence patient with psychosis or impulsivity utilizing antipsychotic polypharmacy and high-dose monotherapy. CNS Spectrums. 2014;19(5):439-448.
2. Latuda [package insert]. Marlborough, MA: Sunovion Pharmaceuticals Inc.; 2019.
3. Taylor D, Paton C, Kapur S. The Maudsley prescribing guidelines in psychiatry. 12th ed. Wiley Blackwell; 2015.
4. Vyas P, Hwang BJ, Brasic JR. An evaluation of lumateperone tosylate for the treatment of schizophrenia. Expert Opin Pharmacother. 2020;21(2):139-145.
5. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62.
6. Saphris [package insert]. Irvine, CA: Allergan USA, Inc; 2017.
7. Abilify [package insert]. Tokyo, Japan: Otsuka America Pharmaceutical, Inc.; 2014.
8. Rexulti [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc.; 2020.
9. Vraylar [package insert]. Madison, NJ: Allergan USA, Inc.; 2019.
10. Clozaril [package insert]. Rosemont, PA: Novartis Pharmaceuticals Corporation; 2017.
11. Fanapt [package insert]. Washington, DC: Vanda Pharmaceuticals Inc.; 2016.
12. Caplyta [package insert]. New York, NY: Intra-Cellular Therapies, Inc.; 2019.
13. Zyprexa [package insert]. Indianapolis, IN: Lilly USA, LLC.; 2020.
14. Invega [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2019.
15. Seroquel [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2016.
16. Risperdal [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2020.
17. Geodon [package insert]. New York, NY: Pfizer Inc.; 2020.
18. Chaudhry IB, Neelam K, Duddu V, et al. Ethnicity and psychopharmacology. J Psychopharmacol. 2008;22(6):673-680.
19. Seeman MV. Men and women respond differently to antipsychotic drugs. Neuropharmacology. 2020;163:107631. doi: 10.1016/j.neuropharm.2019.05.008
20. Barnes TRE, Drake R, Paton C, et al. Evidence-based guidelines for the pharmacological treatment of schizophrenia: updated recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2020;34(1):3-78.
21. Citrome L, McEvoy JP, Todtenkopf MS, et al. A commentary on the efficacy of olanzapine for the treatment of schizophrenia: the past, present, and future. Neuropsych Dis Treat. 2019;15:2559-2569.
22. Meltzer HY, Bobo WV, Ajanta R, et al. A randomized, double-blind comparison of clozapine and high-dose olanzapine in treatment-resistant patients with schizophrenia. J Clin Psychiatry. 2008;69(2):274-285.
23. Batail JM, Langree B, Robert G, et al. Use of very-high-dose olanzapine in treatment-resistant schizophrenia. Schizophr Res. 2014;159(2-3):411-414.
24. Kinon BJ, Volavka J, Stauffer V, et al. Standard and higher dose of olanzapine in patients with schizophrenia or schizoaffective disorder. J Clin Psychopharmacol. 2008;28(4):392-400.
25. Stahl SM. Stahl’s essential psychopharmacology prescriber’s guide. 6th ed. Cambridge University Press; 2017.
26. Bartlett JA, van der Voort Maarschalk K. Understanding the oral mucosal absorption and resulting clinical pharmacokinetics of asenapine. AAPS PharmSciTech. 2012;13(4):1110-1115.
27. Auby P, Saha A, Ali M, et al. Safety and tolerability of aripiprazole at doses higher than 30 mg. Eur Neuropsychopharm. 2002;12(3):288.
28. Chavez B, Poveda R. Efficacy with high-dose aripiprazole after olanzapine-related metabolic disturbances. Ann Pharmacother. 2006;40(12):2265-2268.
29. Duggal HS, Mendhekar DN. High-dose aripiprazole in treatment-resistant schizophrenia. J Clin Psychiatry. 2006;67(4):674-675.
30. Thone J. Worsened agitation and confusion in schizophrenia subsequent to high-dose aripiprazole. J Neuropsychiatry Clin Neurosci. 2007;19(4):481-482.
31. Saatcioglu O, Gumus S, Kamberyan K, et al. Efficacy of high-dose aripiprazole for treatment-resistant schizoaffective disorder: a case report. Psychopharmacol Bull. 2010;43(4):70-72.
32. Thomson SR, Chogtu B, Bhattacharjee D, et al. Extrapyramidal symptoms probably related to risperidone treatment: a case series. Ann Neurosci. 2017;24(3):155-163.
33. Meltzer HY, Lindenmayer JP, Kwentus J, et al. A six month randomized controlled trial of long acting injectable risperidone 50 and 100 mg in treatment resistant schizophrenia. Schizophr Res. 2014;154(1-3):14-22.
34. Hou Y, Lai C. The response of psychotic symptoms in a patient with resistant schizophrenia under treatment of high-dose risperidone long-acting injection. J Neuropsychiatry Clin Neurosci. 2014;26(3):E16-E17. doi: 10.1176/appi.neuropsych.13070150
35. Albrecht A, Morena PG, Baumann P, et al. High dose of depot risperidone in a nonresponder schizophrenic patient. J Clin Psychopharmacol. 2004;24(6):673-674.
36. Mace S, Taylor D. Reducing the rates of prescribing high-dose antipsychotics and polypharmacy on psychiatric inpatient and intensive care units: results of a 6-year quality improvement programme. Ther Adv Psychopharmacol. 2015;5(1):4-12.
37. Goff DC, McEvoy JP, Citrome L, et al. High-dose oral ziprasidone versus conventional dosing in schizophrenia patients with residual symptoms. J Clin Psychopharmacol. 2013;33:485-490.
38. Levy WO, Robichaux-Keene NR, Nunez C. No significant QTc interval changes with high-dose ziprasidone: a case series. J Psychiatr Pract. 2004;10(4):227-232.
39. Kaushik S, Maccabee N, Kaushik S, et al. Activation induced by high-dose ziprasidone: a case report. J Clin Psychiatry. 2009;70(9):1326-1327.
40. Seeman P. Targeting the dopamine D2 receptor in schizophrenia. Expert Opin Ther Targets. 2006;10(4):515-531.
41. Honer WG, MacEwan W, Gendron A, et al. A randomized, double-blind, placebo-controlled study of safety and tolerability of high-dose quetiapine in patients with persistent symptoms of schizophrenia or schizoaffective disorder. J Clin Psychiatry. 2012;73(1):13-20.
42. Sokolski KN, Brown BJ, Meldon M. Urinary retention following repeated high-dose quetiapine. Ann Pharmacother. 2004;38(5):899-890.
43. Chandrappa P, Ho L. Case reports of patients with treatment-resistant schizophrenia and related psychotic disorders intolerant to clozapine responding to high doses of quetiapine. Ther Adv Psychopharmacol. 2012;2(5):207-209.
44. Pierre JM, Wirshing DA, Wirshing WC, et al. High-dose quetiapine in treatment refractory schizophrenia. Schizophr Res. 2005;73:373-375.
45. Lindenmyer JP, Citrome L, Khan A, et al. A randomized, double-blind parallel-group, fixed-dose, clinical trial of quetiapine at 600 vs. 1200 mg/d for patients with treatment-resistant schizophrenia or schizoaffective disorder. J Clin Psychopharmacol. 2011;31(2):160-168.
46. Hazra M, Culo S, Mamo D. High-dose quetiapine and photopsia. J Clin Psychopharmacol. 2006;26(5):546-547.
47. Smolders DME, Smolders WAP. Case report and review of the literature: cardiomyopathy in a young woman on high-dose quetiapine. Cardiovasc Toxicol. 2017;17(4):478-481.
48. Takeuchi H, Suzuki T, Bies RR, et al. Dose reduction of risperidone and olanzapine and estimated D2 receptor occupancy in stable patients with schizophrenia: findings from an open-label, randomized, controlled study. J Clin Psychiatry. 2014;75(11):1209-1214.
49. Kumar V, Rao NP, Narasimha V, et al. Antipsychotic dose in maintenance treatment of schizophrenia: a retrospective study. Psychiatry Res. 2016;245:311-316.
Mr. K, age 21, is admitted to the psychiatry unit with agitation, disorganized behavior, and paranoia. Upon presentation, he has no known medical history or current medications. He is diagnosed with schizophrenia and subsequently tolerates but does not respond to adequate durations of treatment with fluphenazine, 20 mg/d; aripiprazole, 30 mg/d; and risperidone, 6 mg/d. Medication adherence is verified, but Mr. K is reluctant to try a fourth antipsychotic. The treatment team suspects that Mr. K may be a cytochrome P450 (CYP) 2D6 ultra-rapid metabolizer, so they obtain a serum risperidone level. The serum risperidone concentration is subtherapeutic (10 ng/mL). What should be considered next?
Several factors must be considered when a patient with psychosis does not experience significant symptomatic improvement with an adequate antipsychotic trial. This article focuses on high-dose second-generation antipsychotic (SGA) therapy in adults with psychosis. “High-dose” antipsychotic therapy is dosing that exceeds the standard maximum dosage for a given antipsychotic. Existing evidence on the use of high-dose SGAs consists of open-label studies and case reports, as well as a handful of randomized controlled trials (RCTs) with small sample sizes and high dropout rates. In some studies, the use of concomitant interventions (eg, duplicate antipsychotic therapy) limit the interpretation of data. High-dose first-generation antipsychotic therapy is discouraged because of a heightened risk of extrapyramidal symptoms (EPS).

Steps to take before increasing the dose
When considering prescribing high-dose antipsychotic therapy, first confirm that the patient has been adherent to the current medication regimen. Also, screen for factors that might impair drug absorption, such as bariatric surgery or noncompliance with administration precautions.1 For example, administration of lurasidone with less than 350 calories may considerably decrease absorption.2 Dosage requirements may vary based on ethnicity, gender, CYP polymorphisms, and pharmacokinetic drug interactions (Table 12-17).1,18,19 Causes of inadequate efficacy should be addressed before considering the use of high-dose antipsychotic therapy.1 Under certain circumstances, serum drug concentrations may be used to guide antipsychotic dosing (Table 22-17). Inadequate response despite a therapeutic serum concentration may indicate pharmacodynamic failure.1 Inadequate response in the context of subtherapeutic serum concentrations, good medication adherence, and compliance to administration precautions may be indicative of a genetic polymorphism or drug interaction.1 Changes in antipsychotic dosing or selection may be warranted, depending on associated risks and benefits.

SGAs and high-dose administration
The SGA with the greatest evidence for high-dose administration is olanzapine, which is similar in structure and receptor pharmacology to clozapine.20,21 The use of high-dose olanzapine is controversial. High-dose olanzapine has been compared to clozapine in patients with treatment-resistant schizophrenia (TRS) and schizoaffective disorder. Meltzer et al22 reported similar efficacy with clozapine, 300 to 900 mg/d, and olanzapine, 25 to 45 mg/d. In this study, high-dose olanzapine caused more weight gain when compared to clozapine. Olanzapine dosages of up to 100 mg/d have been prescribed for TRS; however, this is not common practice.23 A study comparing 10, 20, and 40 mg/d in patients with non-TRS or schizoaffective disorder showed no advantage with higher dosages.24
There is limited data on high-dose treatment with other SGAs.17 Orthostasis may limit iloperidone’s safety at high doses, and single doses of asenapine should not exceed 10 mg.25 Limited sublingual surface area and saliva saturation result in decreased bioavailability with higher asenapine doses.25,26 In a small RCT of patients with stable schizophrenia or schizoaffective disorder, aripiprazole was relatively well-tolerated up to 75 mg/d, whereas akathisia and tachycardia occurred with 90 mg/d.27 Case reports have documented successful treatment with aripiprazole, 60 to 75 mg/d; however, dizziness and worsening psychosis, agitation, and confusion have been observed.28-31
There is a paucity of data on high-dose risperidone and paliperidone, possibly due to their potent dopamine-2 (D2) receptor antagonism and dose-related risk of EPS.1 At risperidone dosages >6 mg/d, the balance between D2 and serotonin-2A (5-HT2A) receptor potency is lost, which increases the potential for EPS.32 In one RCT, long-acting injectable (LAI) risperidone, up to 100 mg biweekly, was well-tolerated but no more effective for TRS than 50 mg biweekly.33 A case report suggested improvement of TRS in a patient administered risperidone LAI, 75 mg vs 37.5 mg biweekly, but it is unclear if a 50-mg dosage was tried.34 Another case report documented improvement in schizophrenia symptoms with risperidone LAI, 125 mg biweekly; however, anticholinergic therapy was required for EPS.35
Dose-dependent adverse effects, including EPS, sedation, anticholinergic effects, orthostasis, hyperprolactinemia, and QTc prolongation, may limit the safety of high-dose antipsychotic therapy.1,20,36 Two studies showed no correlation between QTc prolongation and ziprasidone dosages of up to 320 mg/d for psychosis.37,38 QTc prolongation was more likely at higher ziprasidone concentrations.37 Higher concentrations, but not higher dosages, also trended toward improvement in positive symptoms, and concentrations >100 ng/mL were associated with more negative symptoms.37 A case report described improvement in positive symptoms of schizoaffective disorder with ziprasidone, 320 mg/d, but activation, hostility, and depression worsened.39
Continue to: Compared with other antipsychotics...
Compared with other antipsychotics, high-dose clozapine and quetiapine may be less likely to cause EPS due to lower D2 receptor occupancies.40 Nevertheless, increased activity at other postsynaptic receptors may lead to constipation, metabolic effects, and sedation.1,41,42 Case reports suggest efficacy with quetiapine, 1,200 to 2,400 mg/d, vs lower dosages for patients with TRS.43,44 However, RCTs of quetiapine, 600 and 800 mg/d vs 1,200 mg/d, have not demonstrated an efficacy advantage with high-dose treatment in patients with schizophrenia or schizoaffective disorder.41,45 High-dose quetiapine has also resulted in photopsia, cardiotoxicity, orthostasis, dysphagia, and sedation.43,46,47
Proceed with caution
In light of safety concerns and a lack of high-quality evidence for high-dose antipsychotic therapy, alternative solutions for inadequate response to treatment should be considered. Underlying causes of poor response should be addressed, and alternative antipsychotics should be utilized, when appropriate. A clozapine trial remains first-line for TRS. Olanzapine may be the best-supported high-dose antipsychotic alternative when clozapine is not an option. High antipsychotic dosages are not well-studied in patients with genetic polymorphisms or unavoidable drug interactions. Serum antipsychotic concentrations may facilitate dosing in these patients.
If high-dose antipsychotic therapy is deemed necessary, its ongoing appropriateness should be continually re-evaluated. Higher antipsychotic dosages and D2 receptor occupancies may be required to manage acute psychosis, but efficacy may be maintained and adverse effects limited with the use of lower dosages during maintenance treatment.48,49 Long-term treatment with high-dose antipsychotic therapy should be avoided, when possible. If high-dose antipsychotic therapy is prescribed, the rationale should be well-documented. Dosage adjustments should not be made until steady state is reached on a given dosage. Electrocardiograms should be obtained at baseline, steady state, and routinely thereafter.3,20 Tolerability should be assessed regularly, and screening for drug interactions should be conducted when new medications are initiated.
Case CONTINUED
Because Mr. K’s serum risperidone level is subtherapeutic (10 ng/mL), his risperidone dosage is cautiously titrated to 10 mg/d, divided (serum concentration: 22 ng/mL). Mr. K develops mild orthostasis but denies other adverse effects. His psychotic symptoms resolve, and he is discharged with education on nonpharmacologic management of orthostasis. The rationale for high-dose risperidone is relayed to his outpatient psychiatrist, as well as a recommendation to monitor Mr. K closely for continued efficacy and tolerability.
Related Resource
- Barnes TRE, Drake R, Paton C, et al. Evidence-based guidelines for the pharmacological treatment of schizophrenia: updated recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2020;34(1):3-78.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Brexpiprazole • Rexulti
Cariprazine • Vraylar
Clozapine • Clozaril
Iloperidone • Fanapt
Lumateperone • Caplyta
Lurasidone • Latuda
Olanzapine • Zyprexa
Paliperidone • Invega
Quetiapine • Seroquel
Risperidone • Risperdal
Risperidone long-acting injection • Risperdal Consta
Ziprasidone • Geodon
Mr. K, age 21, is admitted to the psychiatry unit with agitation, disorganized behavior, and paranoia. Upon presentation, he has no known medical history or current medications. He is diagnosed with schizophrenia and subsequently tolerates but does not respond to adequate durations of treatment with fluphenazine, 20 mg/d; aripiprazole, 30 mg/d; and risperidone, 6 mg/d. Medication adherence is verified, but Mr. K is reluctant to try a fourth antipsychotic. The treatment team suspects that Mr. K may be a cytochrome P450 (CYP) 2D6 ultra-rapid metabolizer, so they obtain a serum risperidone level. The serum risperidone concentration is subtherapeutic (10 ng/mL). What should be considered next?
Several factors must be considered when a patient with psychosis does not experience significant symptomatic improvement with an adequate antipsychotic trial. This article focuses on high-dose second-generation antipsychotic (SGA) therapy in adults with psychosis. “High-dose” antipsychotic therapy is dosing that exceeds the standard maximum dosage for a given antipsychotic. Existing evidence on the use of high-dose SGAs consists of open-label studies and case reports, as well as a handful of randomized controlled trials (RCTs) with small sample sizes and high dropout rates. In some studies, the use of concomitant interventions (eg, duplicate antipsychotic therapy) limit the interpretation of data. High-dose first-generation antipsychotic therapy is discouraged because of a heightened risk of extrapyramidal symptoms (EPS).

Steps to take before increasing the dose
When considering prescribing high-dose antipsychotic therapy, first confirm that the patient has been adherent to the current medication regimen. Also, screen for factors that might impair drug absorption, such as bariatric surgery or noncompliance with administration precautions.1 For example, administration of lurasidone with less than 350 calories may considerably decrease absorption.2 Dosage requirements may vary based on ethnicity, gender, CYP polymorphisms, and pharmacokinetic drug interactions (Table 12-17).1,18,19 Causes of inadequate efficacy should be addressed before considering the use of high-dose antipsychotic therapy.1 Under certain circumstances, serum drug concentrations may be used to guide antipsychotic dosing (Table 22-17). Inadequate response despite a therapeutic serum concentration may indicate pharmacodynamic failure.1 Inadequate response in the context of subtherapeutic serum concentrations, good medication adherence, and compliance to administration precautions may be indicative of a genetic polymorphism or drug interaction.1 Changes in antipsychotic dosing or selection may be warranted, depending on associated risks and benefits.

SGAs and high-dose administration
The SGA with the greatest evidence for high-dose administration is olanzapine, which is similar in structure and receptor pharmacology to clozapine.20,21 The use of high-dose olanzapine is controversial. High-dose olanzapine has been compared to clozapine in patients with treatment-resistant schizophrenia (TRS) and schizoaffective disorder. Meltzer et al22 reported similar efficacy with clozapine, 300 to 900 mg/d, and olanzapine, 25 to 45 mg/d. In this study, high-dose olanzapine caused more weight gain when compared to clozapine. Olanzapine dosages of up to 100 mg/d have been prescribed for TRS; however, this is not common practice.23 A study comparing 10, 20, and 40 mg/d in patients with non-TRS or schizoaffective disorder showed no advantage with higher dosages.24
There is limited data on high-dose treatment with other SGAs.17 Orthostasis may limit iloperidone’s safety at high doses, and single doses of asenapine should not exceed 10 mg.25 Limited sublingual surface area and saliva saturation result in decreased bioavailability with higher asenapine doses.25,26 In a small RCT of patients with stable schizophrenia or schizoaffective disorder, aripiprazole was relatively well-tolerated up to 75 mg/d, whereas akathisia and tachycardia occurred with 90 mg/d.27 Case reports have documented successful treatment with aripiprazole, 60 to 75 mg/d; however, dizziness and worsening psychosis, agitation, and confusion have been observed.28-31
There is a paucity of data on high-dose risperidone and paliperidone, possibly due to their potent dopamine-2 (D2) receptor antagonism and dose-related risk of EPS.1 At risperidone dosages >6 mg/d, the balance between D2 and serotonin-2A (5-HT2A) receptor potency is lost, which increases the potential for EPS.32 In one RCT, long-acting injectable (LAI) risperidone, up to 100 mg biweekly, was well-tolerated but no more effective for TRS than 50 mg biweekly.33 A case report suggested improvement of TRS in a patient administered risperidone LAI, 75 mg vs 37.5 mg biweekly, but it is unclear if a 50-mg dosage was tried.34 Another case report documented improvement in schizophrenia symptoms with risperidone LAI, 125 mg biweekly; however, anticholinergic therapy was required for EPS.35
Dose-dependent adverse effects, including EPS, sedation, anticholinergic effects, orthostasis, hyperprolactinemia, and QTc prolongation, may limit the safety of high-dose antipsychotic therapy.1,20,36 Two studies showed no correlation between QTc prolongation and ziprasidone dosages of up to 320 mg/d for psychosis.37,38 QTc prolongation was more likely at higher ziprasidone concentrations.37 Higher concentrations, but not higher dosages, also trended toward improvement in positive symptoms, and concentrations >100 ng/mL were associated with more negative symptoms.37 A case report described improvement in positive symptoms of schizoaffective disorder with ziprasidone, 320 mg/d, but activation, hostility, and depression worsened.39
Continue to: Compared with other antipsychotics...
Compared with other antipsychotics, high-dose clozapine and quetiapine may be less likely to cause EPS due to lower D2 receptor occupancies.40 Nevertheless, increased activity at other postsynaptic receptors may lead to constipation, metabolic effects, and sedation.1,41,42 Case reports suggest efficacy with quetiapine, 1,200 to 2,400 mg/d, vs lower dosages for patients with TRS.43,44 However, RCTs of quetiapine, 600 and 800 mg/d vs 1,200 mg/d, have not demonstrated an efficacy advantage with high-dose treatment in patients with schizophrenia or schizoaffective disorder.41,45 High-dose quetiapine has also resulted in photopsia, cardiotoxicity, orthostasis, dysphagia, and sedation.43,46,47
Proceed with caution
In light of safety concerns and a lack of high-quality evidence for high-dose antipsychotic therapy, alternative solutions for inadequate response to treatment should be considered. Underlying causes of poor response should be addressed, and alternative antipsychotics should be utilized, when appropriate. A clozapine trial remains first-line for TRS. Olanzapine may be the best-supported high-dose antipsychotic alternative when clozapine is not an option. High antipsychotic dosages are not well-studied in patients with genetic polymorphisms or unavoidable drug interactions. Serum antipsychotic concentrations may facilitate dosing in these patients.
If high-dose antipsychotic therapy is deemed necessary, its ongoing appropriateness should be continually re-evaluated. Higher antipsychotic dosages and D2 receptor occupancies may be required to manage acute psychosis, but efficacy may be maintained and adverse effects limited with the use of lower dosages during maintenance treatment.48,49 Long-term treatment with high-dose antipsychotic therapy should be avoided, when possible. If high-dose antipsychotic therapy is prescribed, the rationale should be well-documented. Dosage adjustments should not be made until steady state is reached on a given dosage. Electrocardiograms should be obtained at baseline, steady state, and routinely thereafter.3,20 Tolerability should be assessed regularly, and screening for drug interactions should be conducted when new medications are initiated.
Case CONTINUED
Because Mr. K’s serum risperidone level is subtherapeutic (10 ng/mL), his risperidone dosage is cautiously titrated to 10 mg/d, divided (serum concentration: 22 ng/mL). Mr. K develops mild orthostasis but denies other adverse effects. His psychotic symptoms resolve, and he is discharged with education on nonpharmacologic management of orthostasis. The rationale for high-dose risperidone is relayed to his outpatient psychiatrist, as well as a recommendation to monitor Mr. K closely for continued efficacy and tolerability.
Related Resource
- Barnes TRE, Drake R, Paton C, et al. Evidence-based guidelines for the pharmacological treatment of schizophrenia: updated recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2020;34(1):3-78.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Brexpiprazole • Rexulti
Cariprazine • Vraylar
Clozapine • Clozaril
Iloperidone • Fanapt
Lumateperone • Caplyta
Lurasidone • Latuda
Olanzapine • Zyprexa
Paliperidone • Invega
Quetiapine • Seroquel
Risperidone • Risperdal
Risperidone long-acting injection • Risperdal Consta
Ziprasidone • Geodon
1. Morrissette DA, Stahl SM. Treating the violence patient with psychosis or impulsivity utilizing antipsychotic polypharmacy and high-dose monotherapy. CNS Spectrums. 2014;19(5):439-448.
2. Latuda [package insert]. Marlborough, MA: Sunovion Pharmaceuticals Inc.; 2019.
3. Taylor D, Paton C, Kapur S. The Maudsley prescribing guidelines in psychiatry. 12th ed. Wiley Blackwell; 2015.
4. Vyas P, Hwang BJ, Brasic JR. An evaluation of lumateperone tosylate for the treatment of schizophrenia. Expert Opin Pharmacother. 2020;21(2):139-145.
5. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62.
6. Saphris [package insert]. Irvine, CA: Allergan USA, Inc; 2017.
7. Abilify [package insert]. Tokyo, Japan: Otsuka America Pharmaceutical, Inc.; 2014.
8. Rexulti [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc.; 2020.
9. Vraylar [package insert]. Madison, NJ: Allergan USA, Inc.; 2019.
10. Clozaril [package insert]. Rosemont, PA: Novartis Pharmaceuticals Corporation; 2017.
11. Fanapt [package insert]. Washington, DC: Vanda Pharmaceuticals Inc.; 2016.
12. Caplyta [package insert]. New York, NY: Intra-Cellular Therapies, Inc.; 2019.
13. Zyprexa [package insert]. Indianapolis, IN: Lilly USA, LLC.; 2020.
14. Invega [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2019.
15. Seroquel [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2016.
16. Risperdal [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2020.
17. Geodon [package insert]. New York, NY: Pfizer Inc.; 2020.
18. Chaudhry IB, Neelam K, Duddu V, et al. Ethnicity and psychopharmacology. J Psychopharmacol. 2008;22(6):673-680.
19. Seeman MV. Men and women respond differently to antipsychotic drugs. Neuropharmacology. 2020;163:107631. doi: 10.1016/j.neuropharm.2019.05.008
20. Barnes TRE, Drake R, Paton C, et al. Evidence-based guidelines for the pharmacological treatment of schizophrenia: updated recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2020;34(1):3-78.
21. Citrome L, McEvoy JP, Todtenkopf MS, et al. A commentary on the efficacy of olanzapine for the treatment of schizophrenia: the past, present, and future. Neuropsych Dis Treat. 2019;15:2559-2569.
22. Meltzer HY, Bobo WV, Ajanta R, et al. A randomized, double-blind comparison of clozapine and high-dose olanzapine in treatment-resistant patients with schizophrenia. J Clin Psychiatry. 2008;69(2):274-285.
23. Batail JM, Langree B, Robert G, et al. Use of very-high-dose olanzapine in treatment-resistant schizophrenia. Schizophr Res. 2014;159(2-3):411-414.
24. Kinon BJ, Volavka J, Stauffer V, et al. Standard and higher dose of olanzapine in patients with schizophrenia or schizoaffective disorder. J Clin Psychopharmacol. 2008;28(4):392-400.
25. Stahl SM. Stahl’s essential psychopharmacology prescriber’s guide. 6th ed. Cambridge University Press; 2017.
26. Bartlett JA, van der Voort Maarschalk K. Understanding the oral mucosal absorption and resulting clinical pharmacokinetics of asenapine. AAPS PharmSciTech. 2012;13(4):1110-1115.
27. Auby P, Saha A, Ali M, et al. Safety and tolerability of aripiprazole at doses higher than 30 mg. Eur Neuropsychopharm. 2002;12(3):288.
28. Chavez B, Poveda R. Efficacy with high-dose aripiprazole after olanzapine-related metabolic disturbances. Ann Pharmacother. 2006;40(12):2265-2268.
29. Duggal HS, Mendhekar DN. High-dose aripiprazole in treatment-resistant schizophrenia. J Clin Psychiatry. 2006;67(4):674-675.
30. Thone J. Worsened agitation and confusion in schizophrenia subsequent to high-dose aripiprazole. J Neuropsychiatry Clin Neurosci. 2007;19(4):481-482.
31. Saatcioglu O, Gumus S, Kamberyan K, et al. Efficacy of high-dose aripiprazole for treatment-resistant schizoaffective disorder: a case report. Psychopharmacol Bull. 2010;43(4):70-72.
32. Thomson SR, Chogtu B, Bhattacharjee D, et al. Extrapyramidal symptoms probably related to risperidone treatment: a case series. Ann Neurosci. 2017;24(3):155-163.
33. Meltzer HY, Lindenmayer JP, Kwentus J, et al. A six month randomized controlled trial of long acting injectable risperidone 50 and 100 mg in treatment resistant schizophrenia. Schizophr Res. 2014;154(1-3):14-22.
34. Hou Y, Lai C. The response of psychotic symptoms in a patient with resistant schizophrenia under treatment of high-dose risperidone long-acting injection. J Neuropsychiatry Clin Neurosci. 2014;26(3):E16-E17. doi: 10.1176/appi.neuropsych.13070150
35. Albrecht A, Morena PG, Baumann P, et al. High dose of depot risperidone in a nonresponder schizophrenic patient. J Clin Psychopharmacol. 2004;24(6):673-674.
36. Mace S, Taylor D. Reducing the rates of prescribing high-dose antipsychotics and polypharmacy on psychiatric inpatient and intensive care units: results of a 6-year quality improvement programme. Ther Adv Psychopharmacol. 2015;5(1):4-12.
37. Goff DC, McEvoy JP, Citrome L, et al. High-dose oral ziprasidone versus conventional dosing in schizophrenia patients with residual symptoms. J Clin Psychopharmacol. 2013;33:485-490.
38. Levy WO, Robichaux-Keene NR, Nunez C. No significant QTc interval changes with high-dose ziprasidone: a case series. J Psychiatr Pract. 2004;10(4):227-232.
39. Kaushik S, Maccabee N, Kaushik S, et al. Activation induced by high-dose ziprasidone: a case report. J Clin Psychiatry. 2009;70(9):1326-1327.
40. Seeman P. Targeting the dopamine D2 receptor in schizophrenia. Expert Opin Ther Targets. 2006;10(4):515-531.
41. Honer WG, MacEwan W, Gendron A, et al. A randomized, double-blind, placebo-controlled study of safety and tolerability of high-dose quetiapine in patients with persistent symptoms of schizophrenia or schizoaffective disorder. J Clin Psychiatry. 2012;73(1):13-20.
42. Sokolski KN, Brown BJ, Meldon M. Urinary retention following repeated high-dose quetiapine. Ann Pharmacother. 2004;38(5):899-890.
43. Chandrappa P, Ho L. Case reports of patients with treatment-resistant schizophrenia and related psychotic disorders intolerant to clozapine responding to high doses of quetiapine. Ther Adv Psychopharmacol. 2012;2(5):207-209.
44. Pierre JM, Wirshing DA, Wirshing WC, et al. High-dose quetiapine in treatment refractory schizophrenia. Schizophr Res. 2005;73:373-375.
45. Lindenmyer JP, Citrome L, Khan A, et al. A randomized, double-blind parallel-group, fixed-dose, clinical trial of quetiapine at 600 vs. 1200 mg/d for patients with treatment-resistant schizophrenia or schizoaffective disorder. J Clin Psychopharmacol. 2011;31(2):160-168.
46. Hazra M, Culo S, Mamo D. High-dose quetiapine and photopsia. J Clin Psychopharmacol. 2006;26(5):546-547.
47. Smolders DME, Smolders WAP. Case report and review of the literature: cardiomyopathy in a young woman on high-dose quetiapine. Cardiovasc Toxicol. 2017;17(4):478-481.
48. Takeuchi H, Suzuki T, Bies RR, et al. Dose reduction of risperidone and olanzapine and estimated D2 receptor occupancy in stable patients with schizophrenia: findings from an open-label, randomized, controlled study. J Clin Psychiatry. 2014;75(11):1209-1214.
49. Kumar V, Rao NP, Narasimha V, et al. Antipsychotic dose in maintenance treatment of schizophrenia: a retrospective study. Psychiatry Res. 2016;245:311-316.
1. Morrissette DA, Stahl SM. Treating the violence patient with psychosis or impulsivity utilizing antipsychotic polypharmacy and high-dose monotherapy. CNS Spectrums. 2014;19(5):439-448.
2. Latuda [package insert]. Marlborough, MA: Sunovion Pharmaceuticals Inc.; 2019.
3. Taylor D, Paton C, Kapur S. The Maudsley prescribing guidelines in psychiatry. 12th ed. Wiley Blackwell; 2015.
4. Vyas P, Hwang BJ, Brasic JR. An evaluation of lumateperone tosylate for the treatment of schizophrenia. Expert Opin Pharmacother. 2020;21(2):139-145.
5. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62.
6. Saphris [package insert]. Irvine, CA: Allergan USA, Inc; 2017.
7. Abilify [package insert]. Tokyo, Japan: Otsuka America Pharmaceutical, Inc.; 2014.
8. Rexulti [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc.; 2020.
9. Vraylar [package insert]. Madison, NJ: Allergan USA, Inc.; 2019.
10. Clozaril [package insert]. Rosemont, PA: Novartis Pharmaceuticals Corporation; 2017.
11. Fanapt [package insert]. Washington, DC: Vanda Pharmaceuticals Inc.; 2016.
12. Caplyta [package insert]. New York, NY: Intra-Cellular Therapies, Inc.; 2019.
13. Zyprexa [package insert]. Indianapolis, IN: Lilly USA, LLC.; 2020.
14. Invega [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2019.
15. Seroquel [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2016.
16. Risperdal [package insert]. Titusville, NJ: Janssen Pharmaceutical Companies; 2020.
17. Geodon [package insert]. New York, NY: Pfizer Inc.; 2020.
18. Chaudhry IB, Neelam K, Duddu V, et al. Ethnicity and psychopharmacology. J Psychopharmacol. 2008;22(6):673-680.
19. Seeman MV. Men and women respond differently to antipsychotic drugs. Neuropharmacology. 2020;163:107631. doi: 10.1016/j.neuropharm.2019.05.008
20. Barnes TRE, Drake R, Paton C, et al. Evidence-based guidelines for the pharmacological treatment of schizophrenia: updated recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2020;34(1):3-78.
21. Citrome L, McEvoy JP, Todtenkopf MS, et al. A commentary on the efficacy of olanzapine for the treatment of schizophrenia: the past, present, and future. Neuropsych Dis Treat. 2019;15:2559-2569.
22. Meltzer HY, Bobo WV, Ajanta R, et al. A randomized, double-blind comparison of clozapine and high-dose olanzapine in treatment-resistant patients with schizophrenia. J Clin Psychiatry. 2008;69(2):274-285.
23. Batail JM, Langree B, Robert G, et al. Use of very-high-dose olanzapine in treatment-resistant schizophrenia. Schizophr Res. 2014;159(2-3):411-414.
24. Kinon BJ, Volavka J, Stauffer V, et al. Standard and higher dose of olanzapine in patients with schizophrenia or schizoaffective disorder. J Clin Psychopharmacol. 2008;28(4):392-400.
25. Stahl SM. Stahl’s essential psychopharmacology prescriber’s guide. 6th ed. Cambridge University Press; 2017.
26. Bartlett JA, van der Voort Maarschalk K. Understanding the oral mucosal absorption and resulting clinical pharmacokinetics of asenapine. AAPS PharmSciTech. 2012;13(4):1110-1115.
27. Auby P, Saha A, Ali M, et al. Safety and tolerability of aripiprazole at doses higher than 30 mg. Eur Neuropsychopharm. 2002;12(3):288.
28. Chavez B, Poveda R. Efficacy with high-dose aripiprazole after olanzapine-related metabolic disturbances. Ann Pharmacother. 2006;40(12):2265-2268.
29. Duggal HS, Mendhekar DN. High-dose aripiprazole in treatment-resistant schizophrenia. J Clin Psychiatry. 2006;67(4):674-675.
30. Thone J. Worsened agitation and confusion in schizophrenia subsequent to high-dose aripiprazole. J Neuropsychiatry Clin Neurosci. 2007;19(4):481-482.
31. Saatcioglu O, Gumus S, Kamberyan K, et al. Efficacy of high-dose aripiprazole for treatment-resistant schizoaffective disorder: a case report. Psychopharmacol Bull. 2010;43(4):70-72.
32. Thomson SR, Chogtu B, Bhattacharjee D, et al. Extrapyramidal symptoms probably related to risperidone treatment: a case series. Ann Neurosci. 2017;24(3):155-163.
33. Meltzer HY, Lindenmayer JP, Kwentus J, et al. A six month randomized controlled trial of long acting injectable risperidone 50 and 100 mg in treatment resistant schizophrenia. Schizophr Res. 2014;154(1-3):14-22.
34. Hou Y, Lai C. The response of psychotic symptoms in a patient with resistant schizophrenia under treatment of high-dose risperidone long-acting injection. J Neuropsychiatry Clin Neurosci. 2014;26(3):E16-E17. doi: 10.1176/appi.neuropsych.13070150
35. Albrecht A, Morena PG, Baumann P, et al. High dose of depot risperidone in a nonresponder schizophrenic patient. J Clin Psychopharmacol. 2004;24(6):673-674.
36. Mace S, Taylor D. Reducing the rates of prescribing high-dose antipsychotics and polypharmacy on psychiatric inpatient and intensive care units: results of a 6-year quality improvement programme. Ther Adv Psychopharmacol. 2015;5(1):4-12.
37. Goff DC, McEvoy JP, Citrome L, et al. High-dose oral ziprasidone versus conventional dosing in schizophrenia patients with residual symptoms. J Clin Psychopharmacol. 2013;33:485-490.
38. Levy WO, Robichaux-Keene NR, Nunez C. No significant QTc interval changes with high-dose ziprasidone: a case series. J Psychiatr Pract. 2004;10(4):227-232.
39. Kaushik S, Maccabee N, Kaushik S, et al. Activation induced by high-dose ziprasidone: a case report. J Clin Psychiatry. 2009;70(9):1326-1327.
40. Seeman P. Targeting the dopamine D2 receptor in schizophrenia. Expert Opin Ther Targets. 2006;10(4):515-531.
41. Honer WG, MacEwan W, Gendron A, et al. A randomized, double-blind, placebo-controlled study of safety and tolerability of high-dose quetiapine in patients with persistent symptoms of schizophrenia or schizoaffective disorder. J Clin Psychiatry. 2012;73(1):13-20.
42. Sokolski KN, Brown BJ, Meldon M. Urinary retention following repeated high-dose quetiapine. Ann Pharmacother. 2004;38(5):899-890.
43. Chandrappa P, Ho L. Case reports of patients with treatment-resistant schizophrenia and related psychotic disorders intolerant to clozapine responding to high doses of quetiapine. Ther Adv Psychopharmacol. 2012;2(5):207-209.
44. Pierre JM, Wirshing DA, Wirshing WC, et al. High-dose quetiapine in treatment refractory schizophrenia. Schizophr Res. 2005;73:373-375.
45. Lindenmyer JP, Citrome L, Khan A, et al. A randomized, double-blind parallel-group, fixed-dose, clinical trial of quetiapine at 600 vs. 1200 mg/d for patients with treatment-resistant schizophrenia or schizoaffective disorder. J Clin Psychopharmacol. 2011;31(2):160-168.
46. Hazra M, Culo S, Mamo D. High-dose quetiapine and photopsia. J Clin Psychopharmacol. 2006;26(5):546-547.
47. Smolders DME, Smolders WAP. Case report and review of the literature: cardiomyopathy in a young woman on high-dose quetiapine. Cardiovasc Toxicol. 2017;17(4):478-481.
48. Takeuchi H, Suzuki T, Bies RR, et al. Dose reduction of risperidone and olanzapine and estimated D2 receptor occupancy in stable patients with schizophrenia: findings from an open-label, randomized, controlled study. J Clin Psychiatry. 2014;75(11):1209-1214.
49. Kumar V, Rao NP, Narasimha V, et al. Antipsychotic dose in maintenance treatment of schizophrenia: a retrospective study. Psychiatry Res. 2016;245:311-316.
FDA fast tracks testing of schizophrenia drug for impaired cognition
The U.S. Food and Drug Administration has granted breakthrough therapy designation for Boehringer Ingelheim’s experimental agent for the treatment of cognitive impairment associated with schizophrenia (CIAS).
The drug, known as BI 425809, is a novel glycine transporter-1 inhibitor.
The company announced it will start the CONNEX phase 3 clinical trial program to assess the safety and efficacy of the drug for improving cognition for adults with schizophrenia.
The breakthrough therapy designation and the initiation of phase 3 testing are based on results from a phase 2 clinical trial published in The Lancet Psychiatry.
In the phase 2 trial, oral BI 425809, taken once daily, improved cognition after 12 weeks for patients with schizophrenia; doses of 10 mg and 25 mg showed the largest separation from placebo.
Impairment of cognitive function is a major burden for people with schizophrenia, and no pharmacologic treatments are currently approved for CIAS.
“Cognition is a fundamental aspect of everyday life, including problem solving, memory, and attention, which is why finding solutions for cognitive impairment is a key area of Boehringer Ingelheim mental health research,” Vikas Mohan Sharma, MS, with Boehringer Ingelheim, said in a news release.
“This breakthrough therapy designation further highlights the urgent need for novel treatments for people living with schizophrenia. By combining traditional treatment approaches with new and innovative technologies, we are developing targeted therapies that will help to ease the burden of mental health conditions and enable people living with these conditions to create more meaningful connections to their lives, loved ones, and society,” said Mr. Sharma.
The CONNEX clinical trial program is composed of three clinical trials – CONNEX-1, CONNEX-2, and CONNEX-3. All are phase 3 randomized, double-blind, placebo-controlled parallel group trials that will examine the efficacy and safety of BI 425809 taken once daily over a 26-week period for patients with schizophrenia.
The CONNEX trial program will use VeraSci’s Pathway electronic clinical outcome assessment platform, including VeraSci’s Virtual Reality Functional Capacity Assessment Tool (VRFCAT), which simulates key instrumental activities of daily living in a realistic interactive virtual environment, VeraSci explains in a news release announcing the partnership with Boehringer Ingelheim.
The VRFCAT is sensitive to functional capacity deficits and has been accepted into the FDA’s Clinical Outcome Assessment Qualification Program.
The CONNEX trials will also utilize speech biomarker technology from Aural Analytics, which will provide a “richer picture of trial participants’ cognition alongside more conventional clinical outcome measures,” Boehringer Ingelheim says.
“Several of the symptoms of schizophrenia are generated by cognitive and emotional processes that can be identified through disruptions in the outward flow of speech. Using innovative speech analytics may help to objectively assess the downstream consequences of these disruptions,” said Daniel Jones, Aural Analytics co-founder and CEO.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has granted breakthrough therapy designation for Boehringer Ingelheim’s experimental agent for the treatment of cognitive impairment associated with schizophrenia (CIAS).
The drug, known as BI 425809, is a novel glycine transporter-1 inhibitor.
The company announced it will start the CONNEX phase 3 clinical trial program to assess the safety and efficacy of the drug for improving cognition for adults with schizophrenia.
The breakthrough therapy designation and the initiation of phase 3 testing are based on results from a phase 2 clinical trial published in The Lancet Psychiatry.
In the phase 2 trial, oral BI 425809, taken once daily, improved cognition after 12 weeks for patients with schizophrenia; doses of 10 mg and 25 mg showed the largest separation from placebo.
Impairment of cognitive function is a major burden for people with schizophrenia, and no pharmacologic treatments are currently approved for CIAS.
“Cognition is a fundamental aspect of everyday life, including problem solving, memory, and attention, which is why finding solutions for cognitive impairment is a key area of Boehringer Ingelheim mental health research,” Vikas Mohan Sharma, MS, with Boehringer Ingelheim, said in a news release.
“This breakthrough therapy designation further highlights the urgent need for novel treatments for people living with schizophrenia. By combining traditional treatment approaches with new and innovative technologies, we are developing targeted therapies that will help to ease the burden of mental health conditions and enable people living with these conditions to create more meaningful connections to their lives, loved ones, and society,” said Mr. Sharma.
The CONNEX clinical trial program is composed of three clinical trials – CONNEX-1, CONNEX-2, and CONNEX-3. All are phase 3 randomized, double-blind, placebo-controlled parallel group trials that will examine the efficacy and safety of BI 425809 taken once daily over a 26-week period for patients with schizophrenia.
The CONNEX trial program will use VeraSci’s Pathway electronic clinical outcome assessment platform, including VeraSci’s Virtual Reality Functional Capacity Assessment Tool (VRFCAT), which simulates key instrumental activities of daily living in a realistic interactive virtual environment, VeraSci explains in a news release announcing the partnership with Boehringer Ingelheim.
The VRFCAT is sensitive to functional capacity deficits and has been accepted into the FDA’s Clinical Outcome Assessment Qualification Program.
The CONNEX trials will also utilize speech biomarker technology from Aural Analytics, which will provide a “richer picture of trial participants’ cognition alongside more conventional clinical outcome measures,” Boehringer Ingelheim says.
“Several of the symptoms of schizophrenia are generated by cognitive and emotional processes that can be identified through disruptions in the outward flow of speech. Using innovative speech analytics may help to objectively assess the downstream consequences of these disruptions,” said Daniel Jones, Aural Analytics co-founder and CEO.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has granted breakthrough therapy designation for Boehringer Ingelheim’s experimental agent for the treatment of cognitive impairment associated with schizophrenia (CIAS).
The drug, known as BI 425809, is a novel glycine transporter-1 inhibitor.
The company announced it will start the CONNEX phase 3 clinical trial program to assess the safety and efficacy of the drug for improving cognition for adults with schizophrenia.
The breakthrough therapy designation and the initiation of phase 3 testing are based on results from a phase 2 clinical trial published in The Lancet Psychiatry.
In the phase 2 trial, oral BI 425809, taken once daily, improved cognition after 12 weeks for patients with schizophrenia; doses of 10 mg and 25 mg showed the largest separation from placebo.
Impairment of cognitive function is a major burden for people with schizophrenia, and no pharmacologic treatments are currently approved for CIAS.
“Cognition is a fundamental aspect of everyday life, including problem solving, memory, and attention, which is why finding solutions for cognitive impairment is a key area of Boehringer Ingelheim mental health research,” Vikas Mohan Sharma, MS, with Boehringer Ingelheim, said in a news release.
“This breakthrough therapy designation further highlights the urgent need for novel treatments for people living with schizophrenia. By combining traditional treatment approaches with new and innovative technologies, we are developing targeted therapies that will help to ease the burden of mental health conditions and enable people living with these conditions to create more meaningful connections to their lives, loved ones, and society,” said Mr. Sharma.
The CONNEX clinical trial program is composed of three clinical trials – CONNEX-1, CONNEX-2, and CONNEX-3. All are phase 3 randomized, double-blind, placebo-controlled parallel group trials that will examine the efficacy and safety of BI 425809 taken once daily over a 26-week period for patients with schizophrenia.
The CONNEX trial program will use VeraSci’s Pathway electronic clinical outcome assessment platform, including VeraSci’s Virtual Reality Functional Capacity Assessment Tool (VRFCAT), which simulates key instrumental activities of daily living in a realistic interactive virtual environment, VeraSci explains in a news release announcing the partnership with Boehringer Ingelheim.
The VRFCAT is sensitive to functional capacity deficits and has been accepted into the FDA’s Clinical Outcome Assessment Qualification Program.
The CONNEX trials will also utilize speech biomarker technology from Aural Analytics, which will provide a “richer picture of trial participants’ cognition alongside more conventional clinical outcome measures,” Boehringer Ingelheim says.
“Several of the symptoms of schizophrenia are generated by cognitive and emotional processes that can be identified through disruptions in the outward flow of speech. Using innovative speech analytics may help to objectively assess the downstream consequences of these disruptions,” said Daniel Jones, Aural Analytics co-founder and CEO.
A version of this article first appeared on Medscape.com.
APA, AMA, others move to stop insurer from overturning mental health claims ruling
The American Psychiatric Association has joined with the American Medical Association and other medical societies to oppose United Behavioral Health’s (UBH) request that a court throw out a ruling that found the insurer unfairly denied tens of thousands of claims for mental health and substance use disorder services.
Wit v. United Behavioral Health, in litigation since 2014, is being closely watched by clinicians, patients, providers, and attorneys.
Reena Kapoor, MD, chair of the APA’s Committee on Judicial Action, said in an interview that the APA is hopeful that “whatever the court says about UBH should be applicable to all insurance companies that are providing employer-sponsored health benefits.”
In a friend of the court (amicus curiae) brief, the APA, AMA, the California Medical Association, Southern California Psychiatric Society, Northern California Psychiatric Society, Orange County Psychiatric Society, Central California Psychiatric Society, and San Diego Psychiatric Society argue that “despite the availability of professionally developed, evidence-based guidelines embodying generally accepted standards of care for mental health and substance use disorders, managed care organizations commonly base coverage decisions on internally developed ‘level of care guidelines’ that are inappropriately restrictive.”
The guidelines “may lead to denial of coverage for treatment that is recommended by a patient’s physician and even cut off coverage when treatment is already being delivered,” said the groups.
The U.S. Department of Labor also filed a brief in support of the plaintiffs who are suing UBH. Those individuals suffered injury when they were denied coverage, said the federal agency, which regulates employer-sponsored insurance plans.
California Attorney General Rob Bonta also made an amicus filing supporting the plaintiffs.
“When insurers limit access to this critical care, they leave Californians who need it feeling as if they have no other option than to try to cope alone,” said Mr. Bonta in a statement.
‘Discrimination must end’
Mr. Bonta said he agreed with a 2019 ruling by the U.S. District Court for the Northern District of California that UBH had violated its fiduciary duties by wrongfully using its internally developed coverage determination guidelines and level of care guidelines to deny care.
The court also found that UBH’s medically necessary criteria meant that only “acute” episodes would be covered. Instead, said the court last November, chronic and comorbid conditions should always be treated, according to Maureen Gammon and Kathleen Rosenow of Willis Towers Watson, a risk advisor.
In November, the same Northern California District Court ruled on the remedies it would require of United, including that the insurer reprocess more than 67,000 claims. UBH was also barred indefinitely from using any of its guidelines to make coverage determinations. Instead, it was ordered to make determinations “consistent with generally accepted standards of care,” and consistent with state laws.
The District Court denied a request by UBH to put a hold on the claims reprocessing until it appealed the overall case. But the Ninth Circuit Court of Appeals in February granted that request.
Then, in March, United appealed the District Court’s overall ruling, claiming that the plaintiffs had not proven harm.
The U.S. Chamber of Commerce has filed a brief in support of United, agreeing with its arguments.
However, the APA and other clinician groups said there is no question of harm.
“Failure to provide appropriate levels of care for treatment of mental illness and substance use disorders leads to relapse, overdose, transmission of infectious diseases, and death,” said APA CEO and Medical Director Saul Levin, MD, MPA, in a statement.
APA President Vivian Pender, MD, said guidelines that “are overly focused on stabilizing acute symptoms of mental health and substance use disorders” are not treating the underlying disease. “When the injury is physical, insurers treat the underlying disease and not just the symptoms. Discrimination against patients with mental illness must end,” she said.
No court has ever recognized the type of claims reprocessing ordered by the District Court judge, said attorneys Nathaniel Cohen and Joseph Laska of Manatt, Phelps & Phillips, in an analysis of the case.
Mr. Cohen and Mr. Laska write. “Practitioners, health plans, and health insurers would be wise to track UBH’s long-awaited appeal to the Ninth Circuit.”
This article first appeared on Medscape.com.
The American Psychiatric Association has joined with the American Medical Association and other medical societies to oppose United Behavioral Health’s (UBH) request that a court throw out a ruling that found the insurer unfairly denied tens of thousands of claims for mental health and substance use disorder services.
Wit v. United Behavioral Health, in litigation since 2014, is being closely watched by clinicians, patients, providers, and attorneys.
Reena Kapoor, MD, chair of the APA’s Committee on Judicial Action, said in an interview that the APA is hopeful that “whatever the court says about UBH should be applicable to all insurance companies that are providing employer-sponsored health benefits.”
In a friend of the court (amicus curiae) brief, the APA, AMA, the California Medical Association, Southern California Psychiatric Society, Northern California Psychiatric Society, Orange County Psychiatric Society, Central California Psychiatric Society, and San Diego Psychiatric Society argue that “despite the availability of professionally developed, evidence-based guidelines embodying generally accepted standards of care for mental health and substance use disorders, managed care organizations commonly base coverage decisions on internally developed ‘level of care guidelines’ that are inappropriately restrictive.”
The guidelines “may lead to denial of coverage for treatment that is recommended by a patient’s physician and even cut off coverage when treatment is already being delivered,” said the groups.
The U.S. Department of Labor also filed a brief in support of the plaintiffs who are suing UBH. Those individuals suffered injury when they were denied coverage, said the federal agency, which regulates employer-sponsored insurance plans.
California Attorney General Rob Bonta also made an amicus filing supporting the plaintiffs.
“When insurers limit access to this critical care, they leave Californians who need it feeling as if they have no other option than to try to cope alone,” said Mr. Bonta in a statement.
‘Discrimination must end’
Mr. Bonta said he agreed with a 2019 ruling by the U.S. District Court for the Northern District of California that UBH had violated its fiduciary duties by wrongfully using its internally developed coverage determination guidelines and level of care guidelines to deny care.
The court also found that UBH’s medically necessary criteria meant that only “acute” episodes would be covered. Instead, said the court last November, chronic and comorbid conditions should always be treated, according to Maureen Gammon and Kathleen Rosenow of Willis Towers Watson, a risk advisor.
In November, the same Northern California District Court ruled on the remedies it would require of United, including that the insurer reprocess more than 67,000 claims. UBH was also barred indefinitely from using any of its guidelines to make coverage determinations. Instead, it was ordered to make determinations “consistent with generally accepted standards of care,” and consistent with state laws.
The District Court denied a request by UBH to put a hold on the claims reprocessing until it appealed the overall case. But the Ninth Circuit Court of Appeals in February granted that request.
Then, in March, United appealed the District Court’s overall ruling, claiming that the plaintiffs had not proven harm.
The U.S. Chamber of Commerce has filed a brief in support of United, agreeing with its arguments.
However, the APA and other clinician groups said there is no question of harm.
“Failure to provide appropriate levels of care for treatment of mental illness and substance use disorders leads to relapse, overdose, transmission of infectious diseases, and death,” said APA CEO and Medical Director Saul Levin, MD, MPA, in a statement.
APA President Vivian Pender, MD, said guidelines that “are overly focused on stabilizing acute symptoms of mental health and substance use disorders” are not treating the underlying disease. “When the injury is physical, insurers treat the underlying disease and not just the symptoms. Discrimination against patients with mental illness must end,” she said.
No court has ever recognized the type of claims reprocessing ordered by the District Court judge, said attorneys Nathaniel Cohen and Joseph Laska of Manatt, Phelps & Phillips, in an analysis of the case.
Mr. Cohen and Mr. Laska write. “Practitioners, health plans, and health insurers would be wise to track UBH’s long-awaited appeal to the Ninth Circuit.”
This article first appeared on Medscape.com.
The American Psychiatric Association has joined with the American Medical Association and other medical societies to oppose United Behavioral Health’s (UBH) request that a court throw out a ruling that found the insurer unfairly denied tens of thousands of claims for mental health and substance use disorder services.
Wit v. United Behavioral Health, in litigation since 2014, is being closely watched by clinicians, patients, providers, and attorneys.
Reena Kapoor, MD, chair of the APA’s Committee on Judicial Action, said in an interview that the APA is hopeful that “whatever the court says about UBH should be applicable to all insurance companies that are providing employer-sponsored health benefits.”
In a friend of the court (amicus curiae) brief, the APA, AMA, the California Medical Association, Southern California Psychiatric Society, Northern California Psychiatric Society, Orange County Psychiatric Society, Central California Psychiatric Society, and San Diego Psychiatric Society argue that “despite the availability of professionally developed, evidence-based guidelines embodying generally accepted standards of care for mental health and substance use disorders, managed care organizations commonly base coverage decisions on internally developed ‘level of care guidelines’ that are inappropriately restrictive.”
The guidelines “may lead to denial of coverage for treatment that is recommended by a patient’s physician and even cut off coverage when treatment is already being delivered,” said the groups.
The U.S. Department of Labor also filed a brief in support of the plaintiffs who are suing UBH. Those individuals suffered injury when they were denied coverage, said the federal agency, which regulates employer-sponsored insurance plans.
California Attorney General Rob Bonta also made an amicus filing supporting the plaintiffs.
“When insurers limit access to this critical care, they leave Californians who need it feeling as if they have no other option than to try to cope alone,” said Mr. Bonta in a statement.
‘Discrimination must end’
Mr. Bonta said he agreed with a 2019 ruling by the U.S. District Court for the Northern District of California that UBH had violated its fiduciary duties by wrongfully using its internally developed coverage determination guidelines and level of care guidelines to deny care.
The court also found that UBH’s medically necessary criteria meant that only “acute” episodes would be covered. Instead, said the court last November, chronic and comorbid conditions should always be treated, according to Maureen Gammon and Kathleen Rosenow of Willis Towers Watson, a risk advisor.
In November, the same Northern California District Court ruled on the remedies it would require of United, including that the insurer reprocess more than 67,000 claims. UBH was also barred indefinitely from using any of its guidelines to make coverage determinations. Instead, it was ordered to make determinations “consistent with generally accepted standards of care,” and consistent with state laws.
The District Court denied a request by UBH to put a hold on the claims reprocessing until it appealed the overall case. But the Ninth Circuit Court of Appeals in February granted that request.
Then, in March, United appealed the District Court’s overall ruling, claiming that the plaintiffs had not proven harm.
The U.S. Chamber of Commerce has filed a brief in support of United, agreeing with its arguments.
However, the APA and other clinician groups said there is no question of harm.
“Failure to provide appropriate levels of care for treatment of mental illness and substance use disorders leads to relapse, overdose, transmission of infectious diseases, and death,” said APA CEO and Medical Director Saul Levin, MD, MPA, in a statement.
APA President Vivian Pender, MD, said guidelines that “are overly focused on stabilizing acute symptoms of mental health and substance use disorders” are not treating the underlying disease. “When the injury is physical, insurers treat the underlying disease and not just the symptoms. Discrimination against patients with mental illness must end,” she said.
No court has ever recognized the type of claims reprocessing ordered by the District Court judge, said attorneys Nathaniel Cohen and Joseph Laska of Manatt, Phelps & Phillips, in an analysis of the case.
Mr. Cohen and Mr. Laska write. “Practitioners, health plans, and health insurers would be wise to track UBH’s long-awaited appeal to the Ninth Circuit.”
This article first appeared on Medscape.com.
Psychosis, depression tied to neurodegeneration in Parkinson’s
Depression and psychosis are significantly associated with neuronal loss and gliosis – but not with Lewy body scores – in Parkinson’s disease, data from analyses of the brains of 175 patients suggest.
Previous research has suggested a link between neuronal loss and depression in Parkinson’s disease (PD) but the impact of Lewy bodies has not been well studied, Nicole Mercado Fischer, MPH, of Johns Hopkins University, Baltimore, and colleagues wrote.
Evaluating Lewy body scores and neuronal loss/gliosis in the substantia nigra pars compacta (SN) and locus coeruleus (LC) could increase understanding of pathophysiology in PD, they said.
In a study published in the American Journal of Geriatric Psychiatry, the researchers analyzed the brains of 175 individuals with a primary diagnosis of PD.
A total of 98 participants had diagnoses of psychosis, 88 had depression, and 55 had anxiety. The average age of onset for PD was 62.4 years; 67.4% of the subjects were male, and 97.8% were White. The mean duration of illness was 16 years, and the average age at death was 78 years.
Psychosis was significantly associated with severe neuronal loss and gliosis in both the LC and SN (P = .048 and P = .042, respectively). Depression was significantly associated with severe neuronal loss in the SN (P = .042) but not in the LC. Anxiety was not associated with severe neuronal loss in either brain region. These results remained significant after a multivariate analysis, the researchers noted. However, Lewy body scores were not associated with any neuropsychiatric symptom, and severity of neuronal loss and gliosis was not correlated with Lewy body scores.
The study findings were limited by several factors, including the retrospective design and inability to collect pathology data for all patients, the researchers noted. Also, in some cases, the collection of clinical data and observation of brain tissue pathology took place years apart, and the researchers did not assess medication records.
However, the results were strengthened by the large sample size and “further support the notion that in vivo clinical symptoms of PD are either not caused by Lewy body pathology or that the relationship is confounded by the time of autopsy,” they said. and eventually by using new functional imaging techniques in vivo.”
The researchers had no financial conflicts to disclose. Two coauthors were supported in part by the National Institutes of Health.
Depression and psychosis are significantly associated with neuronal loss and gliosis – but not with Lewy body scores – in Parkinson’s disease, data from analyses of the brains of 175 patients suggest.
Previous research has suggested a link between neuronal loss and depression in Parkinson’s disease (PD) but the impact of Lewy bodies has not been well studied, Nicole Mercado Fischer, MPH, of Johns Hopkins University, Baltimore, and colleagues wrote.
Evaluating Lewy body scores and neuronal loss/gliosis in the substantia nigra pars compacta (SN) and locus coeruleus (LC) could increase understanding of pathophysiology in PD, they said.
In a study published in the American Journal of Geriatric Psychiatry, the researchers analyzed the brains of 175 individuals with a primary diagnosis of PD.
A total of 98 participants had diagnoses of psychosis, 88 had depression, and 55 had anxiety. The average age of onset for PD was 62.4 years; 67.4% of the subjects were male, and 97.8% were White. The mean duration of illness was 16 years, and the average age at death was 78 years.
Psychosis was significantly associated with severe neuronal loss and gliosis in both the LC and SN (P = .048 and P = .042, respectively). Depression was significantly associated with severe neuronal loss in the SN (P = .042) but not in the LC. Anxiety was not associated with severe neuronal loss in either brain region. These results remained significant after a multivariate analysis, the researchers noted. However, Lewy body scores were not associated with any neuropsychiatric symptom, and severity of neuronal loss and gliosis was not correlated with Lewy body scores.
The study findings were limited by several factors, including the retrospective design and inability to collect pathology data for all patients, the researchers noted. Also, in some cases, the collection of clinical data and observation of brain tissue pathology took place years apart, and the researchers did not assess medication records.
However, the results were strengthened by the large sample size and “further support the notion that in vivo clinical symptoms of PD are either not caused by Lewy body pathology or that the relationship is confounded by the time of autopsy,” they said. and eventually by using new functional imaging techniques in vivo.”
The researchers had no financial conflicts to disclose. Two coauthors were supported in part by the National Institutes of Health.
Depression and psychosis are significantly associated with neuronal loss and gliosis – but not with Lewy body scores – in Parkinson’s disease, data from analyses of the brains of 175 patients suggest.
Previous research has suggested a link between neuronal loss and depression in Parkinson’s disease (PD) but the impact of Lewy bodies has not been well studied, Nicole Mercado Fischer, MPH, of Johns Hopkins University, Baltimore, and colleagues wrote.
Evaluating Lewy body scores and neuronal loss/gliosis in the substantia nigra pars compacta (SN) and locus coeruleus (LC) could increase understanding of pathophysiology in PD, they said.
In a study published in the American Journal of Geriatric Psychiatry, the researchers analyzed the brains of 175 individuals with a primary diagnosis of PD.
A total of 98 participants had diagnoses of psychosis, 88 had depression, and 55 had anxiety. The average age of onset for PD was 62.4 years; 67.4% of the subjects were male, and 97.8% were White. The mean duration of illness was 16 years, and the average age at death was 78 years.
Psychosis was significantly associated with severe neuronal loss and gliosis in both the LC and SN (P = .048 and P = .042, respectively). Depression was significantly associated with severe neuronal loss in the SN (P = .042) but not in the LC. Anxiety was not associated with severe neuronal loss in either brain region. These results remained significant after a multivariate analysis, the researchers noted. However, Lewy body scores were not associated with any neuropsychiatric symptom, and severity of neuronal loss and gliosis was not correlated with Lewy body scores.
The study findings were limited by several factors, including the retrospective design and inability to collect pathology data for all patients, the researchers noted. Also, in some cases, the collection of clinical data and observation of brain tissue pathology took place years apart, and the researchers did not assess medication records.
However, the results were strengthened by the large sample size and “further support the notion that in vivo clinical symptoms of PD are either not caused by Lewy body pathology or that the relationship is confounded by the time of autopsy,” they said. and eventually by using new functional imaging techniques in vivo.”
The researchers had no financial conflicts to disclose. Two coauthors were supported in part by the National Institutes of Health.
FROM THE AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY











