User login
Ozempic Curbs Hunger – And Not Just for Food
This transcript has been edited for clarity.
If you’ve been paying attention only to the headlines, when you think of “Ozempic” you’ll think of a few things: a blockbuster weight loss drug or the tip of the spear of a completely new industry — why not? A drug so popular that the people it was invented for (those with diabetes) can’t even get it.
Ozempic and other GLP-1 receptor agonists are undeniable game changers. Insofar as obesity is the number-one public health risk in the United States, antiobesity drugs hold immense promise even if all they do is reduce obesity.
In 2023, an article in Scientific Reports presented data suggesting that people on Ozempic might be reducing their alcohol intake, not just their total calories.
A 2024 article in Molecular Psychiatry found that the drug might positively impact cannabis use disorder. An article from Brain Sciences suggests that the drug reduces compulsive shopping.
A picture is starting to form, a picture that suggests these drugs curb hunger both literally and figuratively. That GLP-1 receptor agonists like Ozempic and Mounjaro are fundamentally anticonsumption drugs. In a society that — some would argue — is plagued by overconsumption, these drugs might be just what the doctor ordered.
If only they could stop people from smoking.
Oh, wait — they can.
At least it seems they can, based on a new study appearing in Annals of Internal Medicine.
Before we get too excited, this is not a randomized trial. There actually was a small randomized trial of exenatide (Byetta), which is in the same class as Ozempic but probably a bit less potent, with promising results for smoking cessation. 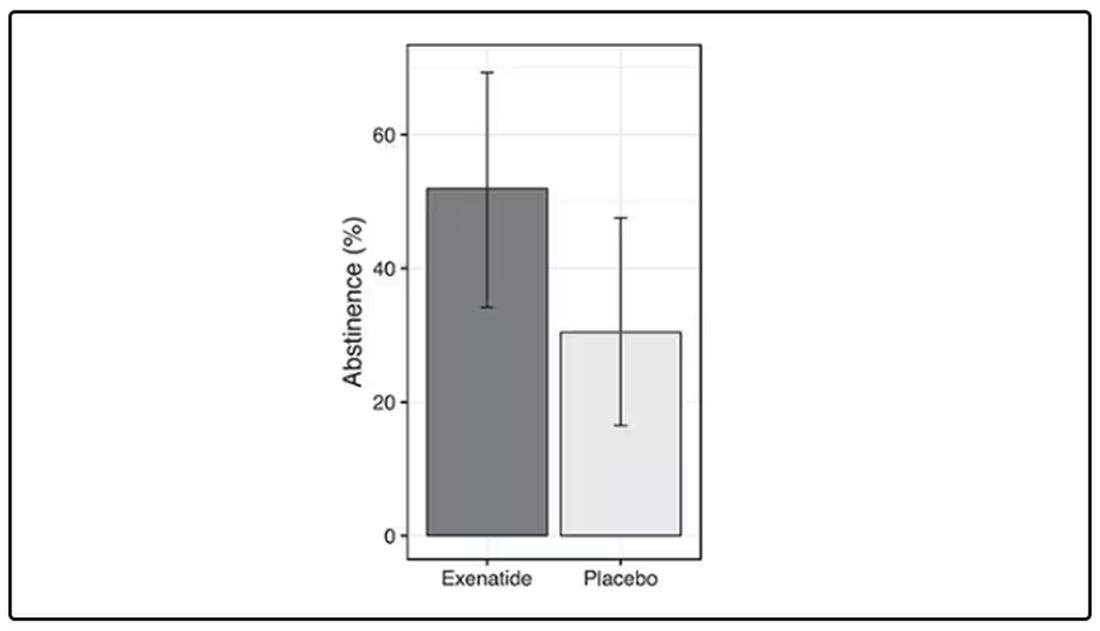
But Byetta is the weaker drug in this class; the market leader is Ozempic. So how can you figure out whether Ozempic can reduce smoking without doing a huge and expensive randomized trial? You can do what Nora Volkow and colleagues from the National Institute on Drug Abuse did: a target trial emulation study.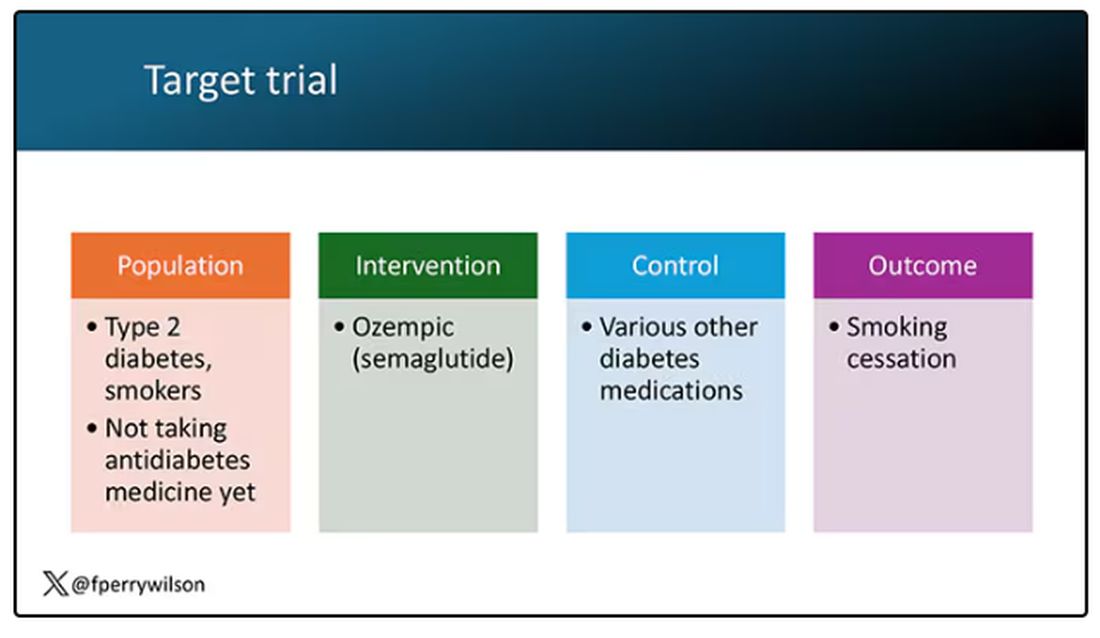
A target trial emulation study is more or less what it sounds like. First, you decide what your dream randomized controlled trial would be and you plan it all out in great detail. You define the population you would recruit, with all the relevant inclusion and exclusion criteria. You define the intervention and the control, and you define the outcome.
But you don’t actually do the trial. You could if someone would lend you $10-$50 million, but assuming you don’t have that lying around, you do the next best thing, which is to dig into a medical record database to find all the people who would be eligible for your imaginary trial. And you analyze them.
The authors wanted to study the effect of Ozempic on smoking among people with diabetes; that’s why all the comparator agents are antidiabetes drugs. They figured out whether these folks were smoking on the basis of a medical record diagnosis of tobacco use disorder before they started one of the drugs of interest. This code is fairly specific: If a patient has it, you can be pretty sure they are smoking. But it’s not very sensitive; not every smoker has this diagnostic code. This is an age-old limitation of using EHR data instead of asking patients, but it’s part of the tradeoff for not having to spend $50 million.
After applying all those inclusion and exclusion criteria, they have a defined population who could be in their dream trial. And, as luck would have it, some of those people really were treated with Ozempic and some really were treated with those other agents. Although decisions about what to prescribe were not randomized, the authors account for this confounding-by-indication using propensity-score matching. You can find a little explainer on propensity-score matching in an earlier column here. 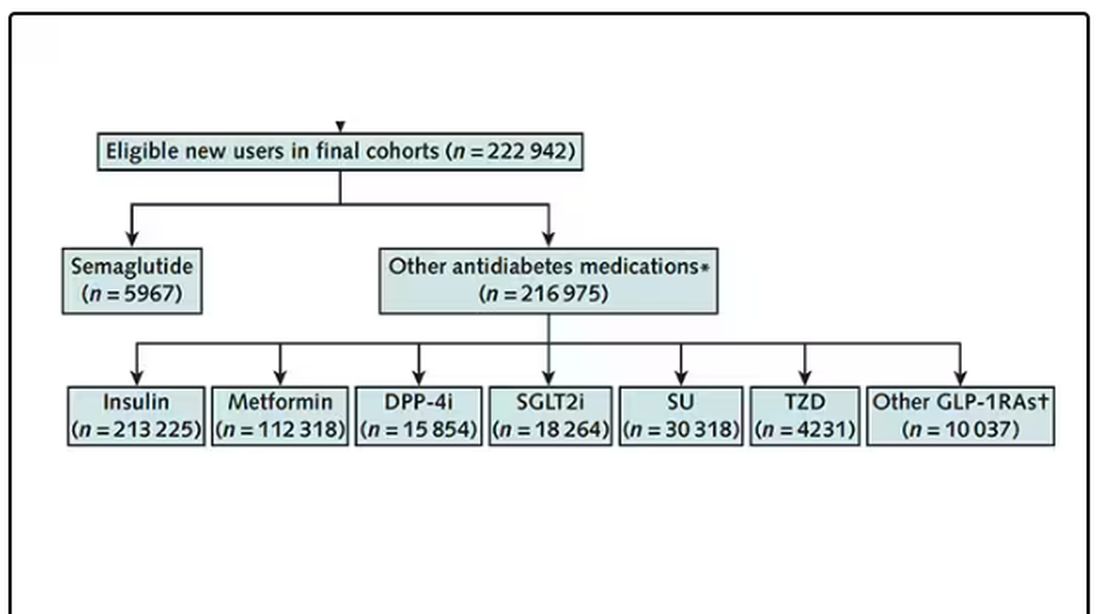
It’s easy enough, using the EHR, to figure out who has diabetes and who got which drug. But how do you know who quit smoking? Remember, everyone had a diagnosis code for tobacco use disorder prior to starting Ozempic or a comparator drug. The authors decided that if the patient had a medical visit where someone again coded tobacco-use disorder, they were still smoking. If someone prescribed smoking cessation meds like a nicotine patch or varenicline, they were obviously still smoking. If someone billed for tobacco-cessation counseling, the patient is still smoking. We’ll get back to the implications of this outcome definition in a minute.
Let’s talk about the results, which are pretty intriguing. 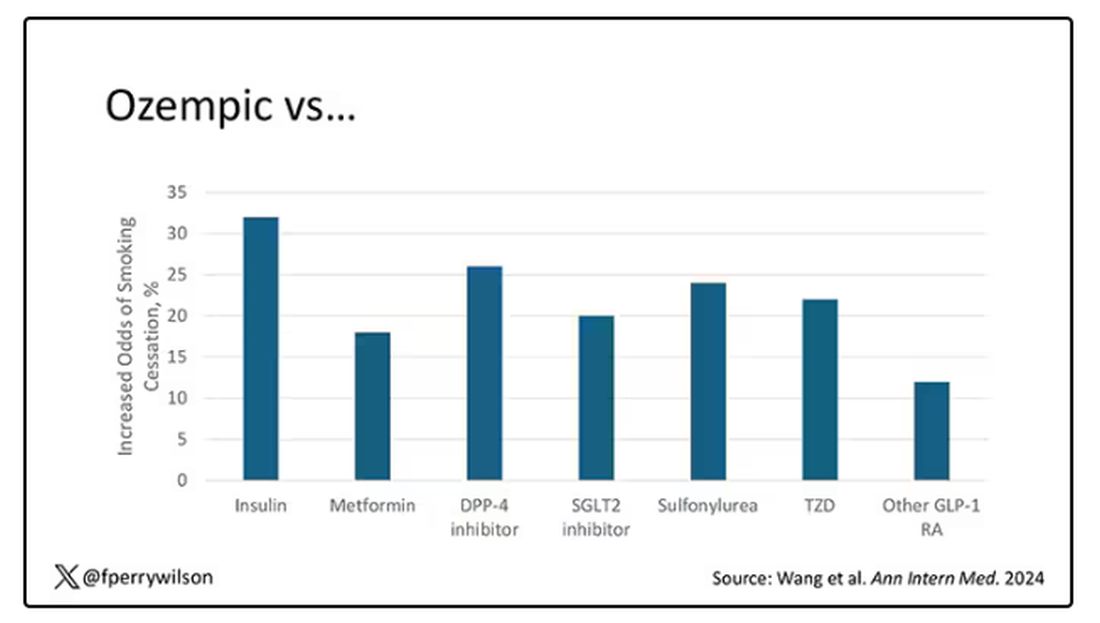
When Ozempic is compared with insulin among smokers with diabetes, those on Ozempic were about 30% more likely to quit smoking. They were about 18% more likely to quit smoking than those who took metformin. They were even slightly more likely to quit smoking than those on other GLP-1 receptor antagonists, though I should note that Mounjaro, which is probably the more potent GLP-1 drug in terms of weight loss, was not among the comparators.
This is pretty impressive for a drug that was not designed to be a smoking cessation drug. It speaks to this emerging idea that these drugs do more than curb appetite by slowing down gastric emptying or something. They work in the brain, modulating some of the reward circuitry that keeps us locked into our bad habits.
There are, of course, some caveats. As I pointed out, this study captured the idea of “still smoking” through the use of administrative codes in the EHR and prescription of smoking cessation aids. You could see similar results if taking Ozempic makes people less likely to address their smoking at all; maybe they shut down the doctor before they even talk about it, or there is too much to discuss during these visits to even get to the subject of smoking. You could also see results like this if people taking Ozempic had fewer visits overall, but the authors showed that that, at least, was not the case.
I’m inclined to believe that this effect is real, simply because we keep seeing signals from multiple sources. If that turns out to be the case, these new “weight loss” drugs may prove to be much more than that; they may turn out to be the drugs that can finally save us from ourselves.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
If you’ve been paying attention only to the headlines, when you think of “Ozempic” you’ll think of a few things: a blockbuster weight loss drug or the tip of the spear of a completely new industry — why not? A drug so popular that the people it was invented for (those with diabetes) can’t even get it.
Ozempic and other GLP-1 receptor agonists are undeniable game changers. Insofar as obesity is the number-one public health risk in the United States, antiobesity drugs hold immense promise even if all they do is reduce obesity.
In 2023, an article in Scientific Reports presented data suggesting that people on Ozempic might be reducing their alcohol intake, not just their total calories.
A 2024 article in Molecular Psychiatry found that the drug might positively impact cannabis use disorder. An article from Brain Sciences suggests that the drug reduces compulsive shopping.
A picture is starting to form, a picture that suggests these drugs curb hunger both literally and figuratively. That GLP-1 receptor agonists like Ozempic and Mounjaro are fundamentally anticonsumption drugs. In a society that — some would argue — is plagued by overconsumption, these drugs might be just what the doctor ordered.
If only they could stop people from smoking.
Oh, wait — they can.
At least it seems they can, based on a new study appearing in Annals of Internal Medicine.
Before we get too excited, this is not a randomized trial. There actually was a small randomized trial of exenatide (Byetta), which is in the same class as Ozempic but probably a bit less potent, with promising results for smoking cessation. 
But Byetta is the weaker drug in this class; the market leader is Ozempic. So how can you figure out whether Ozempic can reduce smoking without doing a huge and expensive randomized trial? You can do what Nora Volkow and colleagues from the National Institute on Drug Abuse did: a target trial emulation study.
A target trial emulation study is more or less what it sounds like. First, you decide what your dream randomized controlled trial would be and you plan it all out in great detail. You define the population you would recruit, with all the relevant inclusion and exclusion criteria. You define the intervention and the control, and you define the outcome.
But you don’t actually do the trial. You could if someone would lend you $10-$50 million, but assuming you don’t have that lying around, you do the next best thing, which is to dig into a medical record database to find all the people who would be eligible for your imaginary trial. And you analyze them.
The authors wanted to study the effect of Ozempic on smoking among people with diabetes; that’s why all the comparator agents are antidiabetes drugs. They figured out whether these folks were smoking on the basis of a medical record diagnosis of tobacco use disorder before they started one of the drugs of interest. This code is fairly specific: If a patient has it, you can be pretty sure they are smoking. But it’s not very sensitive; not every smoker has this diagnostic code. This is an age-old limitation of using EHR data instead of asking patients, but it’s part of the tradeoff for not having to spend $50 million.
After applying all those inclusion and exclusion criteria, they have a defined population who could be in their dream trial. And, as luck would have it, some of those people really were treated with Ozempic and some really were treated with those other agents. Although decisions about what to prescribe were not randomized, the authors account for this confounding-by-indication using propensity-score matching. You can find a little explainer on propensity-score matching in an earlier column here. 
It’s easy enough, using the EHR, to figure out who has diabetes and who got which drug. But how do you know who quit smoking? Remember, everyone had a diagnosis code for tobacco use disorder prior to starting Ozempic or a comparator drug. The authors decided that if the patient had a medical visit where someone again coded tobacco-use disorder, they were still smoking. If someone prescribed smoking cessation meds like a nicotine patch or varenicline, they were obviously still smoking. If someone billed for tobacco-cessation counseling, the patient is still smoking. We’ll get back to the implications of this outcome definition in a minute.
Let’s talk about the results, which are pretty intriguing. 
When Ozempic is compared with insulin among smokers with diabetes, those on Ozempic were about 30% more likely to quit smoking. They were about 18% more likely to quit smoking than those who took metformin. They were even slightly more likely to quit smoking than those on other GLP-1 receptor antagonists, though I should note that Mounjaro, which is probably the more potent GLP-1 drug in terms of weight loss, was not among the comparators.
This is pretty impressive for a drug that was not designed to be a smoking cessation drug. It speaks to this emerging idea that these drugs do more than curb appetite by slowing down gastric emptying or something. They work in the brain, modulating some of the reward circuitry that keeps us locked into our bad habits.
There are, of course, some caveats. As I pointed out, this study captured the idea of “still smoking” through the use of administrative codes in the EHR and prescription of smoking cessation aids. You could see similar results if taking Ozempic makes people less likely to address their smoking at all; maybe they shut down the doctor before they even talk about it, or there is too much to discuss during these visits to even get to the subject of smoking. You could also see results like this if people taking Ozempic had fewer visits overall, but the authors showed that that, at least, was not the case.
I’m inclined to believe that this effect is real, simply because we keep seeing signals from multiple sources. If that turns out to be the case, these new “weight loss” drugs may prove to be much more than that; they may turn out to be the drugs that can finally save us from ourselves.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
If you’ve been paying attention only to the headlines, when you think of “Ozempic” you’ll think of a few things: a blockbuster weight loss drug or the tip of the spear of a completely new industry — why not? A drug so popular that the people it was invented for (those with diabetes) can’t even get it.
Ozempic and other GLP-1 receptor agonists are undeniable game changers. Insofar as obesity is the number-one public health risk in the United States, antiobesity drugs hold immense promise even if all they do is reduce obesity.
In 2023, an article in Scientific Reports presented data suggesting that people on Ozempic might be reducing their alcohol intake, not just their total calories.
A 2024 article in Molecular Psychiatry found that the drug might positively impact cannabis use disorder. An article from Brain Sciences suggests that the drug reduces compulsive shopping.
A picture is starting to form, a picture that suggests these drugs curb hunger both literally and figuratively. That GLP-1 receptor agonists like Ozempic and Mounjaro are fundamentally anticonsumption drugs. In a society that — some would argue — is plagued by overconsumption, these drugs might be just what the doctor ordered.
If only they could stop people from smoking.
Oh, wait — they can.
At least it seems they can, based on a new study appearing in Annals of Internal Medicine.
Before we get too excited, this is not a randomized trial. There actually was a small randomized trial of exenatide (Byetta), which is in the same class as Ozempic but probably a bit less potent, with promising results for smoking cessation. 
But Byetta is the weaker drug in this class; the market leader is Ozempic. So how can you figure out whether Ozempic can reduce smoking without doing a huge and expensive randomized trial? You can do what Nora Volkow and colleagues from the National Institute on Drug Abuse did: a target trial emulation study.
A target trial emulation study is more or less what it sounds like. First, you decide what your dream randomized controlled trial would be and you plan it all out in great detail. You define the population you would recruit, with all the relevant inclusion and exclusion criteria. You define the intervention and the control, and you define the outcome.
But you don’t actually do the trial. You could if someone would lend you $10-$50 million, but assuming you don’t have that lying around, you do the next best thing, which is to dig into a medical record database to find all the people who would be eligible for your imaginary trial. And you analyze them.
The authors wanted to study the effect of Ozempic on smoking among people with diabetes; that’s why all the comparator agents are antidiabetes drugs. They figured out whether these folks were smoking on the basis of a medical record diagnosis of tobacco use disorder before they started one of the drugs of interest. This code is fairly specific: If a patient has it, you can be pretty sure they are smoking. But it’s not very sensitive; not every smoker has this diagnostic code. This is an age-old limitation of using EHR data instead of asking patients, but it’s part of the tradeoff for not having to spend $50 million.
After applying all those inclusion and exclusion criteria, they have a defined population who could be in their dream trial. And, as luck would have it, some of those people really were treated with Ozempic and some really were treated with those other agents. Although decisions about what to prescribe were not randomized, the authors account for this confounding-by-indication using propensity-score matching. You can find a little explainer on propensity-score matching in an earlier column here. 
It’s easy enough, using the EHR, to figure out who has diabetes and who got which drug. But how do you know who quit smoking? Remember, everyone had a diagnosis code for tobacco use disorder prior to starting Ozempic or a comparator drug. The authors decided that if the patient had a medical visit where someone again coded tobacco-use disorder, they were still smoking. If someone prescribed smoking cessation meds like a nicotine patch or varenicline, they were obviously still smoking. If someone billed for tobacco-cessation counseling, the patient is still smoking. We’ll get back to the implications of this outcome definition in a minute.
Let’s talk about the results, which are pretty intriguing. 
When Ozempic is compared with insulin among smokers with diabetes, those on Ozempic were about 30% more likely to quit smoking. They were about 18% more likely to quit smoking than those who took metformin. They were even slightly more likely to quit smoking than those on other GLP-1 receptor antagonists, though I should note that Mounjaro, which is probably the more potent GLP-1 drug in terms of weight loss, was not among the comparators.
This is pretty impressive for a drug that was not designed to be a smoking cessation drug. It speaks to this emerging idea that these drugs do more than curb appetite by slowing down gastric emptying or something. They work in the brain, modulating some of the reward circuitry that keeps us locked into our bad habits.
There are, of course, some caveats. As I pointed out, this study captured the idea of “still smoking” through the use of administrative codes in the EHR and prescription of smoking cessation aids. You could see similar results if taking Ozempic makes people less likely to address their smoking at all; maybe they shut down the doctor before they even talk about it, or there is too much to discuss during these visits to even get to the subject of smoking. You could also see results like this if people taking Ozempic had fewer visits overall, but the authors showed that that, at least, was not the case.
I’m inclined to believe that this effect is real, simply because we keep seeing signals from multiple sources. If that turns out to be the case, these new “weight loss” drugs may prove to be much more than that; they may turn out to be the drugs that can finally save us from ourselves.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Lancet Commission Aims for a Clearer Definition of Obesity
Although obesity affects more than 1 billion people worldwide, according to a global analysis published in The Lancet, it still lacks a clear “identity” in research, social perception, and the healthcare sector. This lack of clarity hinders accurate diagnoses and treatments, while also perpetuating stigma and prejudice. Specialists argue that obesity is a chronic disease rather than just a condition that leads to other diseases.
At the latest International Congress on Obesity held in São Paulo, Brazil from June 26 to 29, The Lancet Commission on the Definition and Diagnosis of Clinical Obesity announced that it is conducting a global study to create a clear definition for obesity. This condition is often wrongly associated solely with individual choices. Ricardo Cohen, MD, PhD, coordinator of the Obesity and Diabetes Specialized Center at the Oswaldo Cruz German Hospital, São Paulo, and a key researcher in the study, made the announcement. “The current definition of obesity is too broad and ineffective for our needs,” said Dr. Cohen.
Dr. Cohen highlighted several challenges stemming from the lack of a precise definition, including confusion between prevention and treatment strategies, inadequate access to evidence-based treatments, and misconceptions about obesity and its reversibility. He also pointed out the limited understanding of the metabolic and biological complexity of the disease. “Society is comfortable with the current scenario because people are commonly blamed for their obesity. This is evident in the acceptance of so-called ‘magic solutions,’ such as fad diets, and the idea that obesity is merely a result of overeating and underexercising,” he said, noting the mental health damage that this perception can cause.
The difficulty in defining obesity stems from its common classification as a risk factor rather than a disease, said Dr. Cohen. Obesity meets the criteria to be considered a disease, such as well-defined pathophysiologic and etiologic mechanisms. In this way, obesity resembles diabetes and depressive disorders, which are classified as diseases based on the same criteria. This inconsistency, maintained by societal perceptions and the healthcare sector, creates confusion. Many professionals still lack a clear understanding of obesity as a disease.
This confusion perpetuates stigma and ignores the unique metabolic function in individuals. As a result, treatments often focus on preventing secondary diseases like diabetes and hypertension rather than on addressing obesity itself. Dr. Cohen recounted the case of a patient with fatigue, knee pain, and osteolysis who couldn’t perform daily activities but did not receive the necessary care. “If he had diabetes, he could have access to treatment because diabetes is recognized as a disease and needs to be treated. But since obesity is not recognized as such, he was sent home.”
To address these challenges, The Lancet Commission’s study, which is expected to be published this year, aims to establish clear diagnostic criteria for adults and children. Drawing inspiration from medical disciplines with well-established diagnostic criteria, such as rheumatology and psychiatry, the research has defined 18 criteria for adults and 14 for children.
The study also redefines treatment outcomes, sets standards for clinical remission of obesity, and proposes clear recommendations for clinical practice and public health policies. The ultimate goal, according to Dr. Cohen, is to transform the global treatment spectrum of obesity and improve access to necessary care.
“Our plan is to recognize obesity as a disease so that health policies, societal attitudes, and treatments will address it more effectively. This approach will also help reduce the harm caused by stigma and prejudice,” concluded Dr. Cohen.
This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Although obesity affects more than 1 billion people worldwide, according to a global analysis published in The Lancet, it still lacks a clear “identity” in research, social perception, and the healthcare sector. This lack of clarity hinders accurate diagnoses and treatments, while also perpetuating stigma and prejudice. Specialists argue that obesity is a chronic disease rather than just a condition that leads to other diseases.
At the latest International Congress on Obesity held in São Paulo, Brazil from June 26 to 29, The Lancet Commission on the Definition and Diagnosis of Clinical Obesity announced that it is conducting a global study to create a clear definition for obesity. This condition is often wrongly associated solely with individual choices. Ricardo Cohen, MD, PhD, coordinator of the Obesity and Diabetes Specialized Center at the Oswaldo Cruz German Hospital, São Paulo, and a key researcher in the study, made the announcement. “The current definition of obesity is too broad and ineffective for our needs,” said Dr. Cohen.
Dr. Cohen highlighted several challenges stemming from the lack of a precise definition, including confusion between prevention and treatment strategies, inadequate access to evidence-based treatments, and misconceptions about obesity and its reversibility. He also pointed out the limited understanding of the metabolic and biological complexity of the disease. “Society is comfortable with the current scenario because people are commonly blamed for their obesity. This is evident in the acceptance of so-called ‘magic solutions,’ such as fad diets, and the idea that obesity is merely a result of overeating and underexercising,” he said, noting the mental health damage that this perception can cause.
The difficulty in defining obesity stems from its common classification as a risk factor rather than a disease, said Dr. Cohen. Obesity meets the criteria to be considered a disease, such as well-defined pathophysiologic and etiologic mechanisms. In this way, obesity resembles diabetes and depressive disorders, which are classified as diseases based on the same criteria. This inconsistency, maintained by societal perceptions and the healthcare sector, creates confusion. Many professionals still lack a clear understanding of obesity as a disease.
This confusion perpetuates stigma and ignores the unique metabolic function in individuals. As a result, treatments often focus on preventing secondary diseases like diabetes and hypertension rather than on addressing obesity itself. Dr. Cohen recounted the case of a patient with fatigue, knee pain, and osteolysis who couldn’t perform daily activities but did not receive the necessary care. “If he had diabetes, he could have access to treatment because diabetes is recognized as a disease and needs to be treated. But since obesity is not recognized as such, he was sent home.”
To address these challenges, The Lancet Commission’s study, which is expected to be published this year, aims to establish clear diagnostic criteria for adults and children. Drawing inspiration from medical disciplines with well-established diagnostic criteria, such as rheumatology and psychiatry, the research has defined 18 criteria for adults and 14 for children.
The study also redefines treatment outcomes, sets standards for clinical remission of obesity, and proposes clear recommendations for clinical practice and public health policies. The ultimate goal, according to Dr. Cohen, is to transform the global treatment spectrum of obesity and improve access to necessary care.
“Our plan is to recognize obesity as a disease so that health policies, societal attitudes, and treatments will address it more effectively. This approach will also help reduce the harm caused by stigma and prejudice,” concluded Dr. Cohen.
This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Although obesity affects more than 1 billion people worldwide, according to a global analysis published in The Lancet, it still lacks a clear “identity” in research, social perception, and the healthcare sector. This lack of clarity hinders accurate diagnoses and treatments, while also perpetuating stigma and prejudice. Specialists argue that obesity is a chronic disease rather than just a condition that leads to other diseases.
At the latest International Congress on Obesity held in São Paulo, Brazil from June 26 to 29, The Lancet Commission on the Definition and Diagnosis of Clinical Obesity announced that it is conducting a global study to create a clear definition for obesity. This condition is often wrongly associated solely with individual choices. Ricardo Cohen, MD, PhD, coordinator of the Obesity and Diabetes Specialized Center at the Oswaldo Cruz German Hospital, São Paulo, and a key researcher in the study, made the announcement. “The current definition of obesity is too broad and ineffective for our needs,” said Dr. Cohen.
Dr. Cohen highlighted several challenges stemming from the lack of a precise definition, including confusion between prevention and treatment strategies, inadequate access to evidence-based treatments, and misconceptions about obesity and its reversibility. He also pointed out the limited understanding of the metabolic and biological complexity of the disease. “Society is comfortable with the current scenario because people are commonly blamed for their obesity. This is evident in the acceptance of so-called ‘magic solutions,’ such as fad diets, and the idea that obesity is merely a result of overeating and underexercising,” he said, noting the mental health damage that this perception can cause.
The difficulty in defining obesity stems from its common classification as a risk factor rather than a disease, said Dr. Cohen. Obesity meets the criteria to be considered a disease, such as well-defined pathophysiologic and etiologic mechanisms. In this way, obesity resembles diabetes and depressive disorders, which are classified as diseases based on the same criteria. This inconsistency, maintained by societal perceptions and the healthcare sector, creates confusion. Many professionals still lack a clear understanding of obesity as a disease.
This confusion perpetuates stigma and ignores the unique metabolic function in individuals. As a result, treatments often focus on preventing secondary diseases like diabetes and hypertension rather than on addressing obesity itself. Dr. Cohen recounted the case of a patient with fatigue, knee pain, and osteolysis who couldn’t perform daily activities but did not receive the necessary care. “If he had diabetes, he could have access to treatment because diabetes is recognized as a disease and needs to be treated. But since obesity is not recognized as such, he was sent home.”
To address these challenges, The Lancet Commission’s study, which is expected to be published this year, aims to establish clear diagnostic criteria for adults and children. Drawing inspiration from medical disciplines with well-established diagnostic criteria, such as rheumatology and psychiatry, the research has defined 18 criteria for adults and 14 for children.
The study also redefines treatment outcomes, sets standards for clinical remission of obesity, and proposes clear recommendations for clinical practice and public health policies. The ultimate goal, according to Dr. Cohen, is to transform the global treatment spectrum of obesity and improve access to necessary care.
“Our plan is to recognize obesity as a disease so that health policies, societal attitudes, and treatments will address it more effectively. This approach will also help reduce the harm caused by stigma and prejudice,” concluded Dr. Cohen.
This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Lipedema: Current Diagnostic and Treatment Evidence
Lipedema affects about 11% of cisgender women, according to the Brazilian Society of Angiology and Vascular Surgery. Yet the condition remains wrapped in uncertainties. Despite significant advancements in understanding its physiology, diagnosis, and treatment, more clarity is needed as awareness and diagnoses increase.
At the latest International Congress on Obesity (ICO) in São Paulo, Brazil, Philipp Scherer, PhD, director of the Touchstone Diabetes Center, discussed the complexities of lipedema. “It is an extremely frustrating condition for someone like me, who has spent a lifetime studying functional and dysfunctional adipose tissue. We are trying to understand the physiology of this pathology, but it is challenging, and so far, we have not been able to find a concrete answer,” he noted.
Lipedema is characterized by the abnormal accumulation of subcutaneous adipose tissue, especially in the lower limbs, and almost exclusively affects cisgender women. The reason for this gender disparity is unclear. It could be an intrinsic characteristic of the disease or a result from clinicians’ lack of familiarity with lipedema, which often leads to misdiagnosis as obesity. This misdiagnosis results in fewer men seeking treatment.
Research has predominantly focused on women, and evidence suggests that hormones play a crucial role in the disease’s pathophysiology. Lipedema typically manifests during periods of hormonal changes, such as puberty, pregnancy, menopause, and hormone replacement therapies, reinforcing the idea that hormones significantly influence the condition’s development and progression.
Main Symptoms
Jonathan Kartt, CEO of the Lipedema Foundation, emphasized that intense pain in the areas of adipose tissue accumulation is a hallmark symptom of lipedema, setting it apart from obesity. Pain levels can vary widely among patients, ranging from moderate to severe, with unbearable peaks on certain days. Mr. Kartt stressed the importance of recognizing and addressing this often underestimated symptom.
Lipedema is characterized by a bilateral, symmetrical increase in mass compared with the rest of the body. This is commonly distinguished by the “cuff sign,” a separation between normal tissue in the feet and abnormal tissue from the ankle upward. Other frequent symptoms include a feeling of heaviness, discomfort, fatigue, frequent bruising, and tiredness. A notable sign is the presence of subcutaneous nodules with a texture similar to that of rice grains, which are crucial for differentiating lipedema from other conditions. Palpation during anamnesis is essential to identify these nodules and confirm the diagnosis.
“It is crucial to investigate the family history for genetic predisposition. Additionally, it is fundamental to ask whether, even with weight loss, the affected areas retain accumulated fat. Hormonal changes, pain symptoms, and impact on quality of life should also be carefully evaluated,” advised Mr. Kartt.
Diagnostic Tools
André Murad, MD, a clinical consultant at the Instituto Lipedema Brazil, has been exploring new diagnostic approaches for lipedema beyond traditional anamnesis. During his presentation at the ICO, he shared studies on the efficacy of imaging exams such as ultrasound, tomography, and MRI in diagnosing the characteristic lipedema-associated increase in subcutaneous tissue.
He also discussed lymphangiography and lymphoscintigraphy, highlighting the use of magnetic resonance lymphangiography to evaluate dilated lymphatic vessels often observed in patients with lipedema. “By injecting contrast into the feet, this technique allows the evaluation of vessels, which are usually dilated, indicating characteristic lymphatic system overload in lipedema. Lymphoscintigraphy is crucial for detecting associated lymphedema, revealing delayed lymphatic flow and asymmetry between limbs in cases of lipedema without lymphedema,” he explained.
Despite the various diagnostic options, Dr. Murad highlighted two highly effective studies. A Brazilian study used ultrasound to establish a cutoff point of 11.7 mm in the pretibial subcutaneous tissue thickness, achieving 96% specificity for diagnosis. Another study emphasized the value of dual-energy x-ray absorptiometry (DXA), which demonstrated 95% sensitivity. This method assesses fat distribution by correlating the amount present in the legs with the total body, providing a cost-effective and accessible option for specialists.
“DXA allows for a precise mathematical evaluation of fat distribution relative to the total body. A ratio of 0.38 in the leg-to-body relationship is a significant indicator of high suspicion of lipedema,” highlighted Dr. Murad. “In clinical practice, many patients self-diagnose with lipedema, but the clinical exam often reveals no disproportion, with the leg-to-body ratio below 0.38 being common in these cases,” he added.
Treatment Approaches
Treatments for lipedema are still evolving, with considerable debate about the best approach. While some specialists advocate exclusively for conservative treatment, others recommend combining these methods with surgical interventions, depending on the stage of the disease. The relative novelty of lipedema and the scarcity of robust, long-term studies contribute to the uncertainty around treatment efficacy.
Conservative treatment typically includes compression, lymphatic drainage techniques, and pressure therapy. An active lifestyle and a healthy diet are also recommended. Although these measures do not prevent the accumulation of adipose tissue, they help reduce inflammation and improve quality of life. “Even though the causes of lipedema are not fully known, lifestyle management is essential for controlling symptoms, starting with an anti-inflammatory diet,” emphasized Dr. Murad.
Because insulin promotes lipogenesis, a diet that avoids spikes in glycemic and insulin levels is advisable. Insulin resistance can exacerbate edema formation, so a Mediterranean diet may be beneficial. This diet limits fast-absorbing carbohydrates, such as added sugar, refined grains, and ultraprocessed foods, while promoting complex carbohydrates from whole grains and legumes.
Dr. Murad also presented a study evaluating the potential benefits of a low-carbohydrate, high-fat diet for patients with lipedema. The study demonstrated weight loss, reduced body fat, controlled leg volume, and, notably, pain relief.
For more advanced stages of lipedema, plastic surgery is often considered when conservative approaches do not yield satisfactory results. Some specialists advocate for surgery as an effective way to remove diseased adipose cells and reduce excess fat accumulation, which can improve physical appearance and associated pain. There is a growing consensus that surgical intervention should be performed early, ideally in stage I of IV, to maximize efficacy and prevent disease progression.
Fábio Masato Kamamoto, MD, a plastic surgeon and director of the Instituto Lipedema Brazil, shared insights into surgical treatments for lipedema. He discussed techniques from liposuction to advanced skin retraction and dermolipectomy, crucial for addressing more advanced stages of the condition. “It’s a complex process that demands precision to protect the lymphatic system, especially considering the characteristic nodules of lipedema,” he noted.
Dr. Kamamoto discussed a former patient with stage III lipedema. In the initial stage, he performed liposuction, removing 8 L of fat and 3.4 kg of skin. After 6 months, a follow-up procedure resulted in a total removal of 15 kg. Complementary procedures, such as microneedling, were performed to stimulate collagen production and reduce skin sagging. In addition to cosmetic improvements, the procedure also removed the distinctive lipedema nodules, which Mr. Kartt described as feeling like “rice grains.” Removing these nodules significantly alleviates pain, according to Dr. Kamamoto.
The benefits of surgical treatment for lipedema can be long lasting. Dr. Kamamoto noted that fat tends not to reaccumulate in treated areas, with patients often experiencing lower weight, reduced edema, and decreased pain over time. “While we hope that patients do not regain weight, the benefits of surgery persist even if weight is regained. Therefore, combining conservative and surgical treatments remains a valid and effective approach,” he concluded.
Dr. Scherer highlighted that despite various approaches, there is still no definitive “magic signature” that fully explains lipedema. This lack of clarity directly affects the effectiveness of diagnoses and treatments. He expressed hope that future integration of data from different studies and approaches will lead to the identification of a clinically useful molecular signature. “The true cause of lipedema remains unknown, requiring more speculation, hypothesis formulation, and testing for significant discoveries. This situation is frustrating, as the disease affects many women who lack a clear diagnosis that differentiates them from patients with obesity, as well as evidence-based recommendations,” he concluded.
This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Lipedema affects about 11% of cisgender women, according to the Brazilian Society of Angiology and Vascular Surgery. Yet the condition remains wrapped in uncertainties. Despite significant advancements in understanding its physiology, diagnosis, and treatment, more clarity is needed as awareness and diagnoses increase.
At the latest International Congress on Obesity (ICO) in São Paulo, Brazil, Philipp Scherer, PhD, director of the Touchstone Diabetes Center, discussed the complexities of lipedema. “It is an extremely frustrating condition for someone like me, who has spent a lifetime studying functional and dysfunctional adipose tissue. We are trying to understand the physiology of this pathology, but it is challenging, and so far, we have not been able to find a concrete answer,” he noted.
Lipedema is characterized by the abnormal accumulation of subcutaneous adipose tissue, especially in the lower limbs, and almost exclusively affects cisgender women. The reason for this gender disparity is unclear. It could be an intrinsic characteristic of the disease or a result from clinicians’ lack of familiarity with lipedema, which often leads to misdiagnosis as obesity. This misdiagnosis results in fewer men seeking treatment.
Research has predominantly focused on women, and evidence suggests that hormones play a crucial role in the disease’s pathophysiology. Lipedema typically manifests during periods of hormonal changes, such as puberty, pregnancy, menopause, and hormone replacement therapies, reinforcing the idea that hormones significantly influence the condition’s development and progression.
Main Symptoms
Jonathan Kartt, CEO of the Lipedema Foundation, emphasized that intense pain in the areas of adipose tissue accumulation is a hallmark symptom of lipedema, setting it apart from obesity. Pain levels can vary widely among patients, ranging from moderate to severe, with unbearable peaks on certain days. Mr. Kartt stressed the importance of recognizing and addressing this often underestimated symptom.
Lipedema is characterized by a bilateral, symmetrical increase in mass compared with the rest of the body. This is commonly distinguished by the “cuff sign,” a separation between normal tissue in the feet and abnormal tissue from the ankle upward. Other frequent symptoms include a feeling of heaviness, discomfort, fatigue, frequent bruising, and tiredness. A notable sign is the presence of subcutaneous nodules with a texture similar to that of rice grains, which are crucial for differentiating lipedema from other conditions. Palpation during anamnesis is essential to identify these nodules and confirm the diagnosis.
“It is crucial to investigate the family history for genetic predisposition. Additionally, it is fundamental to ask whether, even with weight loss, the affected areas retain accumulated fat. Hormonal changes, pain symptoms, and impact on quality of life should also be carefully evaluated,” advised Mr. Kartt.
Diagnostic Tools
André Murad, MD, a clinical consultant at the Instituto Lipedema Brazil, has been exploring new diagnostic approaches for lipedema beyond traditional anamnesis. During his presentation at the ICO, he shared studies on the efficacy of imaging exams such as ultrasound, tomography, and MRI in diagnosing the characteristic lipedema-associated increase in subcutaneous tissue.
He also discussed lymphangiography and lymphoscintigraphy, highlighting the use of magnetic resonance lymphangiography to evaluate dilated lymphatic vessels often observed in patients with lipedema. “By injecting contrast into the feet, this technique allows the evaluation of vessels, which are usually dilated, indicating characteristic lymphatic system overload in lipedema. Lymphoscintigraphy is crucial for detecting associated lymphedema, revealing delayed lymphatic flow and asymmetry between limbs in cases of lipedema without lymphedema,” he explained.
Despite the various diagnostic options, Dr. Murad highlighted two highly effective studies. A Brazilian study used ultrasound to establish a cutoff point of 11.7 mm in the pretibial subcutaneous tissue thickness, achieving 96% specificity for diagnosis. Another study emphasized the value of dual-energy x-ray absorptiometry (DXA), which demonstrated 95% sensitivity. This method assesses fat distribution by correlating the amount present in the legs with the total body, providing a cost-effective and accessible option for specialists.
“DXA allows for a precise mathematical evaluation of fat distribution relative to the total body. A ratio of 0.38 in the leg-to-body relationship is a significant indicator of high suspicion of lipedema,” highlighted Dr. Murad. “In clinical practice, many patients self-diagnose with lipedema, but the clinical exam often reveals no disproportion, with the leg-to-body ratio below 0.38 being common in these cases,” he added.
Treatment Approaches
Treatments for lipedema are still evolving, with considerable debate about the best approach. While some specialists advocate exclusively for conservative treatment, others recommend combining these methods with surgical interventions, depending on the stage of the disease. The relative novelty of lipedema and the scarcity of robust, long-term studies contribute to the uncertainty around treatment efficacy.
Conservative treatment typically includes compression, lymphatic drainage techniques, and pressure therapy. An active lifestyle and a healthy diet are also recommended. Although these measures do not prevent the accumulation of adipose tissue, they help reduce inflammation and improve quality of life. “Even though the causes of lipedema are not fully known, lifestyle management is essential for controlling symptoms, starting with an anti-inflammatory diet,” emphasized Dr. Murad.
Because insulin promotes lipogenesis, a diet that avoids spikes in glycemic and insulin levels is advisable. Insulin resistance can exacerbate edema formation, so a Mediterranean diet may be beneficial. This diet limits fast-absorbing carbohydrates, such as added sugar, refined grains, and ultraprocessed foods, while promoting complex carbohydrates from whole grains and legumes.
Dr. Murad also presented a study evaluating the potential benefits of a low-carbohydrate, high-fat diet for patients with lipedema. The study demonstrated weight loss, reduced body fat, controlled leg volume, and, notably, pain relief.
For more advanced stages of lipedema, plastic surgery is often considered when conservative approaches do not yield satisfactory results. Some specialists advocate for surgery as an effective way to remove diseased adipose cells and reduce excess fat accumulation, which can improve physical appearance and associated pain. There is a growing consensus that surgical intervention should be performed early, ideally in stage I of IV, to maximize efficacy and prevent disease progression.
Fábio Masato Kamamoto, MD, a plastic surgeon and director of the Instituto Lipedema Brazil, shared insights into surgical treatments for lipedema. He discussed techniques from liposuction to advanced skin retraction and dermolipectomy, crucial for addressing more advanced stages of the condition. “It’s a complex process that demands precision to protect the lymphatic system, especially considering the characteristic nodules of lipedema,” he noted.
Dr. Kamamoto discussed a former patient with stage III lipedema. In the initial stage, he performed liposuction, removing 8 L of fat and 3.4 kg of skin. After 6 months, a follow-up procedure resulted in a total removal of 15 kg. Complementary procedures, such as microneedling, were performed to stimulate collagen production and reduce skin sagging. In addition to cosmetic improvements, the procedure also removed the distinctive lipedema nodules, which Mr. Kartt described as feeling like “rice grains.” Removing these nodules significantly alleviates pain, according to Dr. Kamamoto.
The benefits of surgical treatment for lipedema can be long lasting. Dr. Kamamoto noted that fat tends not to reaccumulate in treated areas, with patients often experiencing lower weight, reduced edema, and decreased pain over time. “While we hope that patients do not regain weight, the benefits of surgery persist even if weight is regained. Therefore, combining conservative and surgical treatments remains a valid and effective approach,” he concluded.
Dr. Scherer highlighted that despite various approaches, there is still no definitive “magic signature” that fully explains lipedema. This lack of clarity directly affects the effectiveness of diagnoses and treatments. He expressed hope that future integration of data from different studies and approaches will lead to the identification of a clinically useful molecular signature. “The true cause of lipedema remains unknown, requiring more speculation, hypothesis formulation, and testing for significant discoveries. This situation is frustrating, as the disease affects many women who lack a clear diagnosis that differentiates them from patients with obesity, as well as evidence-based recommendations,” he concluded.
This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Lipedema affects about 11% of cisgender women, according to the Brazilian Society of Angiology and Vascular Surgery. Yet the condition remains wrapped in uncertainties. Despite significant advancements in understanding its physiology, diagnosis, and treatment, more clarity is needed as awareness and diagnoses increase.
At the latest International Congress on Obesity (ICO) in São Paulo, Brazil, Philipp Scherer, PhD, director of the Touchstone Diabetes Center, discussed the complexities of lipedema. “It is an extremely frustrating condition for someone like me, who has spent a lifetime studying functional and dysfunctional adipose tissue. We are trying to understand the physiology of this pathology, but it is challenging, and so far, we have not been able to find a concrete answer,” he noted.
Lipedema is characterized by the abnormal accumulation of subcutaneous adipose tissue, especially in the lower limbs, and almost exclusively affects cisgender women. The reason for this gender disparity is unclear. It could be an intrinsic characteristic of the disease or a result from clinicians’ lack of familiarity with lipedema, which often leads to misdiagnosis as obesity. This misdiagnosis results in fewer men seeking treatment.
Research has predominantly focused on women, and evidence suggests that hormones play a crucial role in the disease’s pathophysiology. Lipedema typically manifests during periods of hormonal changes, such as puberty, pregnancy, menopause, and hormone replacement therapies, reinforcing the idea that hormones significantly influence the condition’s development and progression.
Main Symptoms
Jonathan Kartt, CEO of the Lipedema Foundation, emphasized that intense pain in the areas of adipose tissue accumulation is a hallmark symptom of lipedema, setting it apart from obesity. Pain levels can vary widely among patients, ranging from moderate to severe, with unbearable peaks on certain days. Mr. Kartt stressed the importance of recognizing and addressing this often underestimated symptom.
Lipedema is characterized by a bilateral, symmetrical increase in mass compared with the rest of the body. This is commonly distinguished by the “cuff sign,” a separation between normal tissue in the feet and abnormal tissue from the ankle upward. Other frequent symptoms include a feeling of heaviness, discomfort, fatigue, frequent bruising, and tiredness. A notable sign is the presence of subcutaneous nodules with a texture similar to that of rice grains, which are crucial for differentiating lipedema from other conditions. Palpation during anamnesis is essential to identify these nodules and confirm the diagnosis.
“It is crucial to investigate the family history for genetic predisposition. Additionally, it is fundamental to ask whether, even with weight loss, the affected areas retain accumulated fat. Hormonal changes, pain symptoms, and impact on quality of life should also be carefully evaluated,” advised Mr. Kartt.
Diagnostic Tools
André Murad, MD, a clinical consultant at the Instituto Lipedema Brazil, has been exploring new diagnostic approaches for lipedema beyond traditional anamnesis. During his presentation at the ICO, he shared studies on the efficacy of imaging exams such as ultrasound, tomography, and MRI in diagnosing the characteristic lipedema-associated increase in subcutaneous tissue.
He also discussed lymphangiography and lymphoscintigraphy, highlighting the use of magnetic resonance lymphangiography to evaluate dilated lymphatic vessels often observed in patients with lipedema. “By injecting contrast into the feet, this technique allows the evaluation of vessels, which are usually dilated, indicating characteristic lymphatic system overload in lipedema. Lymphoscintigraphy is crucial for detecting associated lymphedema, revealing delayed lymphatic flow and asymmetry between limbs in cases of lipedema without lymphedema,” he explained.
Despite the various diagnostic options, Dr. Murad highlighted two highly effective studies. A Brazilian study used ultrasound to establish a cutoff point of 11.7 mm in the pretibial subcutaneous tissue thickness, achieving 96% specificity for diagnosis. Another study emphasized the value of dual-energy x-ray absorptiometry (DXA), which demonstrated 95% sensitivity. This method assesses fat distribution by correlating the amount present in the legs with the total body, providing a cost-effective and accessible option for specialists.
“DXA allows for a precise mathematical evaluation of fat distribution relative to the total body. A ratio of 0.38 in the leg-to-body relationship is a significant indicator of high suspicion of lipedema,” highlighted Dr. Murad. “In clinical practice, many patients self-diagnose with lipedema, but the clinical exam often reveals no disproportion, with the leg-to-body ratio below 0.38 being common in these cases,” he added.
Treatment Approaches
Treatments for lipedema are still evolving, with considerable debate about the best approach. While some specialists advocate exclusively for conservative treatment, others recommend combining these methods with surgical interventions, depending on the stage of the disease. The relative novelty of lipedema and the scarcity of robust, long-term studies contribute to the uncertainty around treatment efficacy.
Conservative treatment typically includes compression, lymphatic drainage techniques, and pressure therapy. An active lifestyle and a healthy diet are also recommended. Although these measures do not prevent the accumulation of adipose tissue, they help reduce inflammation and improve quality of life. “Even though the causes of lipedema are not fully known, lifestyle management is essential for controlling symptoms, starting with an anti-inflammatory diet,” emphasized Dr. Murad.
Because insulin promotes lipogenesis, a diet that avoids spikes in glycemic and insulin levels is advisable. Insulin resistance can exacerbate edema formation, so a Mediterranean diet may be beneficial. This diet limits fast-absorbing carbohydrates, such as added sugar, refined grains, and ultraprocessed foods, while promoting complex carbohydrates from whole grains and legumes.
Dr. Murad also presented a study evaluating the potential benefits of a low-carbohydrate, high-fat diet for patients with lipedema. The study demonstrated weight loss, reduced body fat, controlled leg volume, and, notably, pain relief.
For more advanced stages of lipedema, plastic surgery is often considered when conservative approaches do not yield satisfactory results. Some specialists advocate for surgery as an effective way to remove diseased adipose cells and reduce excess fat accumulation, which can improve physical appearance and associated pain. There is a growing consensus that surgical intervention should be performed early, ideally in stage I of IV, to maximize efficacy and prevent disease progression.
Fábio Masato Kamamoto, MD, a plastic surgeon and director of the Instituto Lipedema Brazil, shared insights into surgical treatments for lipedema. He discussed techniques from liposuction to advanced skin retraction and dermolipectomy, crucial for addressing more advanced stages of the condition. “It’s a complex process that demands precision to protect the lymphatic system, especially considering the characteristic nodules of lipedema,” he noted.
Dr. Kamamoto discussed a former patient with stage III lipedema. In the initial stage, he performed liposuction, removing 8 L of fat and 3.4 kg of skin. After 6 months, a follow-up procedure resulted in a total removal of 15 kg. Complementary procedures, such as microneedling, were performed to stimulate collagen production and reduce skin sagging. In addition to cosmetic improvements, the procedure also removed the distinctive lipedema nodules, which Mr. Kartt described as feeling like “rice grains.” Removing these nodules significantly alleviates pain, according to Dr. Kamamoto.
The benefits of surgical treatment for lipedema can be long lasting. Dr. Kamamoto noted that fat tends not to reaccumulate in treated areas, with patients often experiencing lower weight, reduced edema, and decreased pain over time. “While we hope that patients do not regain weight, the benefits of surgery persist even if weight is regained. Therefore, combining conservative and surgical treatments remains a valid and effective approach,” he concluded.
Dr. Scherer highlighted that despite various approaches, there is still no definitive “magic signature” that fully explains lipedema. This lack of clarity directly affects the effectiveness of diagnoses and treatments. He expressed hope that future integration of data from different studies and approaches will lead to the identification of a clinically useful molecular signature. “The true cause of lipedema remains unknown, requiring more speculation, hypothesis formulation, and testing for significant discoveries. This situation is frustrating, as the disease affects many women who lack a clear diagnosis that differentiates them from patients with obesity, as well as evidence-based recommendations,” he concluded.
This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
New Study Says Your Sedentary Lifestyle Is Killing You
TOPLINE:
METHODOLOGY:
- Researchers evaluated the association between PA and ST with the risk for mortality in 5836 middle-aged and older Australian adults (mean age, 56.4 years; 45% men) from the Australian Diabetes, Obesity and Lifestyle Study.
- The Physical Activity and Sitting Time Balance Index (PASTBI) was calculated by dividing the total duration of daily PA by the duration of daily ST.
- Participants were categorized into quartiles on the basis of their PASTBI score, ranging from low PA/high ST to high PA/low ST.
- The primary outcome was all-cause mortality.
TAKEAWAY:
- During a median follow-up time of 14.3 years, 885 (15%) all-cause deaths were reported.
- The risk for all-cause mortality was 47% higher in participants with lower engagement in PA and higher ST (low PASTBI) than those with higher engagement in PA and lower ST (high PASTBI; adjusted hazard ratio, 1.47; 95% confidence interval, 1.21-1.79).
IN PRACTICE:
“The utility of the PASTBI in identifying relationships with mortality risk further highlights the importance of achieving a healthier balance in the dual health behaviors of PA [physical activity] and ST [sitting time],” the authors wrote.
SOURCE:
The study was led by Roslin Botlero, MBBS, MPH, PhD, of the School of Public Health and Preventive Medicine at Monash University in Melbourne, Australia. It was published online in the American Journal of Preventive Medicine.
LIMITATIONS:
The study relied on self-reported data for PA and ST, which may have introduced recall or reporting bias. The generalizability of the findings is restricted to a specific set of self-reported questionnaires. Even after adjustment for several potential confounders, other unmeasured or unknown confounders may have influenced the association between PASTBI and all-cause mortality.
DISCLOSURES:
The Australian Diabetes, Obesity and Lifestyle Study was sponsored by the National Health and Medical Research Council, the Australian Government Department of Health and Aged Care, and others. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers evaluated the association between PA and ST with the risk for mortality in 5836 middle-aged and older Australian adults (mean age, 56.4 years; 45% men) from the Australian Diabetes, Obesity and Lifestyle Study.
- The Physical Activity and Sitting Time Balance Index (PASTBI) was calculated by dividing the total duration of daily PA by the duration of daily ST.
- Participants were categorized into quartiles on the basis of their PASTBI score, ranging from low PA/high ST to high PA/low ST.
- The primary outcome was all-cause mortality.
TAKEAWAY:
- During a median follow-up time of 14.3 years, 885 (15%) all-cause deaths were reported.
- The risk for all-cause mortality was 47% higher in participants with lower engagement in PA and higher ST (low PASTBI) than those with higher engagement in PA and lower ST (high PASTBI; adjusted hazard ratio, 1.47; 95% confidence interval, 1.21-1.79).
IN PRACTICE:
“The utility of the PASTBI in identifying relationships with mortality risk further highlights the importance of achieving a healthier balance in the dual health behaviors of PA [physical activity] and ST [sitting time],” the authors wrote.
SOURCE:
The study was led by Roslin Botlero, MBBS, MPH, PhD, of the School of Public Health and Preventive Medicine at Monash University in Melbourne, Australia. It was published online in the American Journal of Preventive Medicine.
LIMITATIONS:
The study relied on self-reported data for PA and ST, which may have introduced recall or reporting bias. The generalizability of the findings is restricted to a specific set of self-reported questionnaires. Even after adjustment for several potential confounders, other unmeasured or unknown confounders may have influenced the association between PASTBI and all-cause mortality.
DISCLOSURES:
The Australian Diabetes, Obesity and Lifestyle Study was sponsored by the National Health and Medical Research Council, the Australian Government Department of Health and Aged Care, and others. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers evaluated the association between PA and ST with the risk for mortality in 5836 middle-aged and older Australian adults (mean age, 56.4 years; 45% men) from the Australian Diabetes, Obesity and Lifestyle Study.
- The Physical Activity and Sitting Time Balance Index (PASTBI) was calculated by dividing the total duration of daily PA by the duration of daily ST.
- Participants were categorized into quartiles on the basis of their PASTBI score, ranging from low PA/high ST to high PA/low ST.
- The primary outcome was all-cause mortality.
TAKEAWAY:
- During a median follow-up time of 14.3 years, 885 (15%) all-cause deaths were reported.
- The risk for all-cause mortality was 47% higher in participants with lower engagement in PA and higher ST (low PASTBI) than those with higher engagement in PA and lower ST (high PASTBI; adjusted hazard ratio, 1.47; 95% confidence interval, 1.21-1.79).
IN PRACTICE:
“The utility of the PASTBI in identifying relationships with mortality risk further highlights the importance of achieving a healthier balance in the dual health behaviors of PA [physical activity] and ST [sitting time],” the authors wrote.
SOURCE:
The study was led by Roslin Botlero, MBBS, MPH, PhD, of the School of Public Health and Preventive Medicine at Monash University in Melbourne, Australia. It was published online in the American Journal of Preventive Medicine.
LIMITATIONS:
The study relied on self-reported data for PA and ST, which may have introduced recall or reporting bias. The generalizability of the findings is restricted to a specific set of self-reported questionnaires. Even after adjustment for several potential confounders, other unmeasured or unknown confounders may have influenced the association between PASTBI and all-cause mortality.
DISCLOSURES:
The Australian Diabetes, Obesity and Lifestyle Study was sponsored by the National Health and Medical Research Council, the Australian Government Department of Health and Aged Care, and others. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
‘Psychological Weight’ Crucial in Patients With Obesity
Increasingly recognized as a multifactorial disease, obesity demands an approach that involves multiple healthcare professionals. For psychologist Andréa Levy, coordinator and founder of the nongovernmental organization Obesity Brazil, addressing the patient’s “psychological weight” is crucial.
In an interview with this news organization, Ms. Levy, who was one of the speakers at the International Congress on Obesity in 2024, emphasized the importance of integrating emotional and behavioral aspects into treatment, because these factors often influence eating habits and weight gain.
She also highlighted the essential collaboration between endocrinologists, nutritionists, psychiatrists, and psychologists, who must work together to provide comprehensive and effective care to patients.
How do psychological factors affect the treatment of obesity?
Psychological factors are important triggers for weight gain. As the degree of obesity increases, so does the predisposition to mental health problems such as anxiety, mood disorders, personality disorders, and eating disorders. Understanding these factors is important because accurate psychodiagnosis is essential for effective disease treatment.
Without a proper diagnosis, the treatment may be incomplete and omit relevant factors. For example, a person with undiagnosed depression who is starting treatment for weight loss may feel discouraged and low on energy. He or she may wrongly attribute these symptoms to the diet or surgery. Similarly, someone undergoing bariatric surgery may confuse malnutrition symptoms with depression, resulting in inadequate treatment with antidepressants and possible iatrogenic complications.
Furthermore, psychotherapy and psychological follow-up are essential to help the individual organize better and understand the treatment and the disease itself. This is especially important in stigmatized diseases and those subject to prejudice such as obesity, where understanding and acceptance are often challenging, which affects treatment adherence.
Is the collaboration between psychologist and psychiatrist always necessary?
Often, it is necessary to have the support of both a psychologist and a psychiatrist. The process generally begins with a good psychodiagnosis. Initially, there may not be a case that requires treatment, but it is important to perform this evaluation to rule out any issues.
The follow-up, unlike weekly psychotherapy, can be monthly or at an interval agreed on with the patient. It is crucial to help him or her navigate the various stages of obesity treatment. For example, the patient may be going through a period of mourning or separation, or a happier moment, such as the beginning of a relationship or the birth of a child in the family. These moments affect eating habits and need to be well managed.
Depending on the degree of the pathology, such as depression, severe binge-eating disorder, or personality disorders, the psychologist works in conjunction with the psychiatrist. When we talk about obesity, we are possibly also talking about a psychiatric population because it is a disease that, besides being highly recurrent, involves many other factors, such as the gaze of others, difficulty with dressing, body pains, mobility, and relationships. Therefore, having this disease alone is already a trigger for disorders such as depression.
What is the main evidence regarding the psychological follow-up of patients with obesity?
Several studies have investigated the relationship between obesity and mental health. Research indicates that the greater the obesity, the higher the likelihood of a positive diagnosis for a psychiatric disorder. Additionally, there is evidence of the benefits of psychological treatment for patients with obesity.
A study published in the Journal of Clinical Endocrinology and Metabolism addressed the impact of cognitive-behavioral therapy (CBT), which helps patients manage goals and treat maladaptive behaviors such as binge-eating disorders. CBT has a modest effect on weight loss, but its integration as part of a lifestyle modification amplifies the results of this loss.
Recent research also shows that weight loss through bariatric surgery offers significant psychological benefits. In the past, it was believed that this procedure could cause depression and other severe psychiatric disorders, but it is now more than proven that weight loss, when done properly and without misconduct or malnutrition, improves psychological and psychiatric issues.
How does psychological follow-up affect the use of medication during obesity treatment?
Many people who take medications, such as corticosteroids for chronic pain or psychiatric medications, may experience weight gain. It is essential to discuss these issues with the psychiatrist because if the patient already has a predisposition to weight gain, medication X should be chosen instead of medication Y, or the dosage should be adjusted. The psychiatrist needs to understand obesity to medicate correctly. Other types of medication, such as chemotherapeutics, may also cause weight gain, often resulting in more abdominal obesity.
There is also lipedema, a hormone-dependent disease that is different from obesity. In this disease, the person gains weight mainly in the legs and arms. In this case, bariatric surgery may result in weight loss only in specific areas, causing disproportionality and difficulty in understanding for the patient. Therefore, when treating obesity, it is important to analyze the patient from all angles: psychological, physiologic, and physical, considering the diversity of the body, its functioning, and hormonal reactions.
Although psychologists do not prescribe medications, they often explain their functioning to the patient. For example, if a patient is taking a glucagon-like peptide 1 analog and experiences initial nausea, he or she may stop using the treatment because the wrong dose had been started. In this case, the psychologist can explain how the medication works and encourage the patient to discuss adjustments with the doctor, avoiding premature discontinuation.
How has the mental health follow-up of patients with obesity evolved over the years?
I started working with people with obesity 25 years ago, when I myself underwent bariatric surgery. At that time, surgeons were used to “solving” the problem and sending the person home. Often, the patient did not even return for surgical follow-up because, in theory, the problem was solved.
Over time, I believe that surgeons learned to talk to the patient, understanding that there is a whole process that even involves creating a bond with the individual who underwent the surgical procedure. Within this process, the importance of the mental health of patients was recognized, and how common it is to confuse a degree of malnutrition with a mental disorder.
Even though I am not a nutritionist, I need to know the difference between a case of malnutrition and depression. So, it is a whole set of factors that needs to be worked on like an orchestra. It is not necessary for this work to be done in the same physical space, but dialogue is important.
Of course, there are things that the patient will only share with the psychologist or with the surgeon, but there are also pieces of information that need to be shared for positive management. I have had patients who were afraid to go back to the nutritionist because they did not lose weight. If they are afraid, it is because the professional is guiding them incorrectly.
What tips would you give to clinicians regarding the psychological approach to people with obesity?
Accessibility is crucial. When someone tells me they are dealing with obesity and depression, I usually ask, “Did you know you have two chronic diseases?” It is essential to explain these concepts because the patient may often think they are free after a successful diet and weight loss, which is not true because of the high relapse associated with obesity. Depression and anxiety follow similar patterns. If the same person wears prescription glasses, I interact by saying, “Did you know you have three chronic diseases?” This question often causes surprise. “I hadn’t thought of that.”
It is essential to use accessible language for the patient to understand the functioning of the disease. More important than choosing a treatment approach is understanding the pathophysiology of obesity and its psychological impact. This avoids a one-size-fits-all approach for all patients.
For example, the impact on someone who developed obesity in childhood after suffering physical, moral, or sexual abuse will probably be deeper than on someone in a healthy family who gained weight after becoming sedentary. Each life story requires a personalized approach.
Sometimes, a patient with mild obesity (grade 1) may not seem to need specific interventions at first glance, but it is crucial to listen to his or her story. Similarly, patients with severe obesity (grades 3 or 4) who resist surgery are entitled to other treatment options, and this is perfectly valid. Therefore, it is always important to ask, “Who is this person? What does obesity represent in their story?” Then propose the most appropriate treatment.
Ms. Levy reported having no relevant financial relationships.
This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Increasingly recognized as a multifactorial disease, obesity demands an approach that involves multiple healthcare professionals. For psychologist Andréa Levy, coordinator and founder of the nongovernmental organization Obesity Brazil, addressing the patient’s “psychological weight” is crucial.
In an interview with this news organization, Ms. Levy, who was one of the speakers at the International Congress on Obesity in 2024, emphasized the importance of integrating emotional and behavioral aspects into treatment, because these factors often influence eating habits and weight gain.
She also highlighted the essential collaboration between endocrinologists, nutritionists, psychiatrists, and psychologists, who must work together to provide comprehensive and effective care to patients.
How do psychological factors affect the treatment of obesity?
Psychological factors are important triggers for weight gain. As the degree of obesity increases, so does the predisposition to mental health problems such as anxiety, mood disorders, personality disorders, and eating disorders. Understanding these factors is important because accurate psychodiagnosis is essential for effective disease treatment.
Without a proper diagnosis, the treatment may be incomplete and omit relevant factors. For example, a person with undiagnosed depression who is starting treatment for weight loss may feel discouraged and low on energy. He or she may wrongly attribute these symptoms to the diet or surgery. Similarly, someone undergoing bariatric surgery may confuse malnutrition symptoms with depression, resulting in inadequate treatment with antidepressants and possible iatrogenic complications.
Furthermore, psychotherapy and psychological follow-up are essential to help the individual organize better and understand the treatment and the disease itself. This is especially important in stigmatized diseases and those subject to prejudice such as obesity, where understanding and acceptance are often challenging, which affects treatment adherence.
Is the collaboration between psychologist and psychiatrist always necessary?
Often, it is necessary to have the support of both a psychologist and a psychiatrist. The process generally begins with a good psychodiagnosis. Initially, there may not be a case that requires treatment, but it is important to perform this evaluation to rule out any issues.
The follow-up, unlike weekly psychotherapy, can be monthly or at an interval agreed on with the patient. It is crucial to help him or her navigate the various stages of obesity treatment. For example, the patient may be going through a period of mourning or separation, or a happier moment, such as the beginning of a relationship or the birth of a child in the family. These moments affect eating habits and need to be well managed.
Depending on the degree of the pathology, such as depression, severe binge-eating disorder, or personality disorders, the psychologist works in conjunction with the psychiatrist. When we talk about obesity, we are possibly also talking about a psychiatric population because it is a disease that, besides being highly recurrent, involves many other factors, such as the gaze of others, difficulty with dressing, body pains, mobility, and relationships. Therefore, having this disease alone is already a trigger for disorders such as depression.
What is the main evidence regarding the psychological follow-up of patients with obesity?
Several studies have investigated the relationship between obesity and mental health. Research indicates that the greater the obesity, the higher the likelihood of a positive diagnosis for a psychiatric disorder. Additionally, there is evidence of the benefits of psychological treatment for patients with obesity.
A study published in the Journal of Clinical Endocrinology and Metabolism addressed the impact of cognitive-behavioral therapy (CBT), which helps patients manage goals and treat maladaptive behaviors such as binge-eating disorders. CBT has a modest effect on weight loss, but its integration as part of a lifestyle modification amplifies the results of this loss.
Recent research also shows that weight loss through bariatric surgery offers significant psychological benefits. In the past, it was believed that this procedure could cause depression and other severe psychiatric disorders, but it is now more than proven that weight loss, when done properly and without misconduct or malnutrition, improves psychological and psychiatric issues.
How does psychological follow-up affect the use of medication during obesity treatment?
Many people who take medications, such as corticosteroids for chronic pain or psychiatric medications, may experience weight gain. It is essential to discuss these issues with the psychiatrist because if the patient already has a predisposition to weight gain, medication X should be chosen instead of medication Y, or the dosage should be adjusted. The psychiatrist needs to understand obesity to medicate correctly. Other types of medication, such as chemotherapeutics, may also cause weight gain, often resulting in more abdominal obesity.
There is also lipedema, a hormone-dependent disease that is different from obesity. In this disease, the person gains weight mainly in the legs and arms. In this case, bariatric surgery may result in weight loss only in specific areas, causing disproportionality and difficulty in understanding for the patient. Therefore, when treating obesity, it is important to analyze the patient from all angles: psychological, physiologic, and physical, considering the diversity of the body, its functioning, and hormonal reactions.
Although psychologists do not prescribe medications, they often explain their functioning to the patient. For example, if a patient is taking a glucagon-like peptide 1 analog and experiences initial nausea, he or she may stop using the treatment because the wrong dose had been started. In this case, the psychologist can explain how the medication works and encourage the patient to discuss adjustments with the doctor, avoiding premature discontinuation.
How has the mental health follow-up of patients with obesity evolved over the years?
I started working with people with obesity 25 years ago, when I myself underwent bariatric surgery. At that time, surgeons were used to “solving” the problem and sending the person home. Often, the patient did not even return for surgical follow-up because, in theory, the problem was solved.
Over time, I believe that surgeons learned to talk to the patient, understanding that there is a whole process that even involves creating a bond with the individual who underwent the surgical procedure. Within this process, the importance of the mental health of patients was recognized, and how common it is to confuse a degree of malnutrition with a mental disorder.
Even though I am not a nutritionist, I need to know the difference between a case of malnutrition and depression. So, it is a whole set of factors that needs to be worked on like an orchestra. It is not necessary for this work to be done in the same physical space, but dialogue is important.
Of course, there are things that the patient will only share with the psychologist or with the surgeon, but there are also pieces of information that need to be shared for positive management. I have had patients who were afraid to go back to the nutritionist because they did not lose weight. If they are afraid, it is because the professional is guiding them incorrectly.
What tips would you give to clinicians regarding the psychological approach to people with obesity?
Accessibility is crucial. When someone tells me they are dealing with obesity and depression, I usually ask, “Did you know you have two chronic diseases?” It is essential to explain these concepts because the patient may often think they are free after a successful diet and weight loss, which is not true because of the high relapse associated with obesity. Depression and anxiety follow similar patterns. If the same person wears prescription glasses, I interact by saying, “Did you know you have three chronic diseases?” This question often causes surprise. “I hadn’t thought of that.”
It is essential to use accessible language for the patient to understand the functioning of the disease. More important than choosing a treatment approach is understanding the pathophysiology of obesity and its psychological impact. This avoids a one-size-fits-all approach for all patients.
For example, the impact on someone who developed obesity in childhood after suffering physical, moral, or sexual abuse will probably be deeper than on someone in a healthy family who gained weight after becoming sedentary. Each life story requires a personalized approach.
Sometimes, a patient with mild obesity (grade 1) may not seem to need specific interventions at first glance, but it is crucial to listen to his or her story. Similarly, patients with severe obesity (grades 3 or 4) who resist surgery are entitled to other treatment options, and this is perfectly valid. Therefore, it is always important to ask, “Who is this person? What does obesity represent in their story?” Then propose the most appropriate treatment.
Ms. Levy reported having no relevant financial relationships.
This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Increasingly recognized as a multifactorial disease, obesity demands an approach that involves multiple healthcare professionals. For psychologist Andréa Levy, coordinator and founder of the nongovernmental organization Obesity Brazil, addressing the patient’s “psychological weight” is crucial.
In an interview with this news organization, Ms. Levy, who was one of the speakers at the International Congress on Obesity in 2024, emphasized the importance of integrating emotional and behavioral aspects into treatment, because these factors often influence eating habits and weight gain.
She also highlighted the essential collaboration between endocrinologists, nutritionists, psychiatrists, and psychologists, who must work together to provide comprehensive and effective care to patients.
How do psychological factors affect the treatment of obesity?
Psychological factors are important triggers for weight gain. As the degree of obesity increases, so does the predisposition to mental health problems such as anxiety, mood disorders, personality disorders, and eating disorders. Understanding these factors is important because accurate psychodiagnosis is essential for effective disease treatment.
Without a proper diagnosis, the treatment may be incomplete and omit relevant factors. For example, a person with undiagnosed depression who is starting treatment for weight loss may feel discouraged and low on energy. He or she may wrongly attribute these symptoms to the diet or surgery. Similarly, someone undergoing bariatric surgery may confuse malnutrition symptoms with depression, resulting in inadequate treatment with antidepressants and possible iatrogenic complications.
Furthermore, psychotherapy and psychological follow-up are essential to help the individual organize better and understand the treatment and the disease itself. This is especially important in stigmatized diseases and those subject to prejudice such as obesity, where understanding and acceptance are often challenging, which affects treatment adherence.
Is the collaboration between psychologist and psychiatrist always necessary?
Often, it is necessary to have the support of both a psychologist and a psychiatrist. The process generally begins with a good psychodiagnosis. Initially, there may not be a case that requires treatment, but it is important to perform this evaluation to rule out any issues.
The follow-up, unlike weekly psychotherapy, can be monthly or at an interval agreed on with the patient. It is crucial to help him or her navigate the various stages of obesity treatment. For example, the patient may be going through a period of mourning or separation, or a happier moment, such as the beginning of a relationship or the birth of a child in the family. These moments affect eating habits and need to be well managed.
Depending on the degree of the pathology, such as depression, severe binge-eating disorder, or personality disorders, the psychologist works in conjunction with the psychiatrist. When we talk about obesity, we are possibly also talking about a psychiatric population because it is a disease that, besides being highly recurrent, involves many other factors, such as the gaze of others, difficulty with dressing, body pains, mobility, and relationships. Therefore, having this disease alone is already a trigger for disorders such as depression.
What is the main evidence regarding the psychological follow-up of patients with obesity?
Several studies have investigated the relationship between obesity and mental health. Research indicates that the greater the obesity, the higher the likelihood of a positive diagnosis for a psychiatric disorder. Additionally, there is evidence of the benefits of psychological treatment for patients with obesity.
A study published in the Journal of Clinical Endocrinology and Metabolism addressed the impact of cognitive-behavioral therapy (CBT), which helps patients manage goals and treat maladaptive behaviors such as binge-eating disorders. CBT has a modest effect on weight loss, but its integration as part of a lifestyle modification amplifies the results of this loss.
Recent research also shows that weight loss through bariatric surgery offers significant psychological benefits. In the past, it was believed that this procedure could cause depression and other severe psychiatric disorders, but it is now more than proven that weight loss, when done properly and without misconduct or malnutrition, improves psychological and psychiatric issues.
How does psychological follow-up affect the use of medication during obesity treatment?
Many people who take medications, such as corticosteroids for chronic pain or psychiatric medications, may experience weight gain. It is essential to discuss these issues with the psychiatrist because if the patient already has a predisposition to weight gain, medication X should be chosen instead of medication Y, or the dosage should be adjusted. The psychiatrist needs to understand obesity to medicate correctly. Other types of medication, such as chemotherapeutics, may also cause weight gain, often resulting in more abdominal obesity.
There is also lipedema, a hormone-dependent disease that is different from obesity. In this disease, the person gains weight mainly in the legs and arms. In this case, bariatric surgery may result in weight loss only in specific areas, causing disproportionality and difficulty in understanding for the patient. Therefore, when treating obesity, it is important to analyze the patient from all angles: psychological, physiologic, and physical, considering the diversity of the body, its functioning, and hormonal reactions.
Although psychologists do not prescribe medications, they often explain their functioning to the patient. For example, if a patient is taking a glucagon-like peptide 1 analog and experiences initial nausea, he or she may stop using the treatment because the wrong dose had been started. In this case, the psychologist can explain how the medication works and encourage the patient to discuss adjustments with the doctor, avoiding premature discontinuation.
How has the mental health follow-up of patients with obesity evolved over the years?
I started working with people with obesity 25 years ago, when I myself underwent bariatric surgery. At that time, surgeons were used to “solving” the problem and sending the person home. Often, the patient did not even return for surgical follow-up because, in theory, the problem was solved.
Over time, I believe that surgeons learned to talk to the patient, understanding that there is a whole process that even involves creating a bond with the individual who underwent the surgical procedure. Within this process, the importance of the mental health of patients was recognized, and how common it is to confuse a degree of malnutrition with a mental disorder.
Even though I am not a nutritionist, I need to know the difference between a case of malnutrition and depression. So, it is a whole set of factors that needs to be worked on like an orchestra. It is not necessary for this work to be done in the same physical space, but dialogue is important.
Of course, there are things that the patient will only share with the psychologist or with the surgeon, but there are also pieces of information that need to be shared for positive management. I have had patients who were afraid to go back to the nutritionist because they did not lose weight. If they are afraid, it is because the professional is guiding them incorrectly.
What tips would you give to clinicians regarding the psychological approach to people with obesity?
Accessibility is crucial. When someone tells me they are dealing with obesity and depression, I usually ask, “Did you know you have two chronic diseases?” It is essential to explain these concepts because the patient may often think they are free after a successful diet and weight loss, which is not true because of the high relapse associated with obesity. Depression and anxiety follow similar patterns. If the same person wears prescription glasses, I interact by saying, “Did you know you have three chronic diseases?” This question often causes surprise. “I hadn’t thought of that.”
It is essential to use accessible language for the patient to understand the functioning of the disease. More important than choosing a treatment approach is understanding the pathophysiology of obesity and its psychological impact. This avoids a one-size-fits-all approach for all patients.
For example, the impact on someone who developed obesity in childhood after suffering physical, moral, or sexual abuse will probably be deeper than on someone in a healthy family who gained weight after becoming sedentary. Each life story requires a personalized approach.
Sometimes, a patient with mild obesity (grade 1) may not seem to need specific interventions at first glance, but it is crucial to listen to his or her story. Similarly, patients with severe obesity (grades 3 or 4) who resist surgery are entitled to other treatment options, and this is perfectly valid. Therefore, it is always important to ask, “Who is this person? What does obesity represent in their story?” Then propose the most appropriate treatment.
Ms. Levy reported having no relevant financial relationships.
This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Promising New Data Support GLP-1s for Dementia Prevention
PHILADELPHIA –
In the phase 2b ELAD clinical trial, adults with early-stage Alzheimer’s disease taking the GLP-1 receptor agonist liraglutide exhibited slower decline in memory and thinking and experienced less brain atrophy over 12 months, compared with placebo.
“The slower loss of brain volume suggests liraglutide protects the brain, much like statins protect the heart,” study chief Paul Edison, MD, PhD, with Imperial College London, London, England, said in a statement.
“While further research is needed, liraglutide may work through various mechanisms, such as reducing inflammation in the brain, lowering insulin resistance and the toxic effects of Alzheimer’s biomarkers amyloid beta and tau, and improving how the brain’s nerve cells communicate,” Dr. Edison said.
He presented the study results at the 2024 Alzheimer’s Association International Conference (AAIC).
Brain Benefits
Liraglutide has previously demonstrated promising neuroprotective effects in animal models of Alzheimer’s disease and epidemiologic studies.
In ELAD, 204 patients with mild to moderate Alzheimer’s disease were randomly allocated (1:1) to a daily subcutaneous injection of up to 1.8 mg of liraglutide or placebo for 12 months; 80 patients in the liraglutide group and 89 in the placebo group completed the study.
Brain MRI was performed at baseline and at 12 months, along with neuropsychometric evaluation and 18F-fludeoxyglucose PET.
The study’s primary endpoint — change in the cerebral glucose metabolic rate in the cortical regions of the brain (hippocampus, medial temporal lobe, and posterior cingulate) — was not met.
However, patients taking liraglutide experienced a significant slowing of cognitive decline, compared with placebo group (P = .01), which was a key secondary outcome, calculated as a composite score of 18 different tests of memory, comprehension, language, and spatial orientation.
Although the study was not powered to assess cognitive changes, adults taking liraglutide had an 18% slower decline in cognitive function over 12 months, compared with those on placebo, Dr. Edison reported.
In addition, patients treated with liraglutide had nearly 50% less volume loss in several areas of the brain involved in memory, language, and decision-making, including frontal, temporal, parietal, and total gray matter, as measured by MRI.
Liraglutide daily subcutaneous injections were safe and well tolerated in patients with Alzheimer’s disease, Dr. Edison reported. There were 25 serious side effects — 18 in the placebo group and 7 in the liraglutide group — and most were considered unlikely to be related to the study treatment. There were no deaths.
Promising, Preliminary
This study shows a positive effect of liraglutide on the brain in terms of “slowing down of brain atrophy and slowing down the rate of cognitive decline,” said Howard Fillit, MD, founding executive director of the Alzheimer’s Drug Discovery Foundation, who wasn’t involved in the study.
Heather Snyder, PhD, vice-president of medical and scientific relations at the Alzheimer’s Association, said it’s “interesting” to see slowing of brain volume loss and some cognitive benefit “especially as the study was not powered necessarily to see some of those changes. The fact that they did see these changes in this small study provides a window into what may happen, but we certainly need larger phase 3 studies.”
In a statement from the UK nonprofit Science Media Centre, Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, called the data “promising.”
“There are clear links from strong data in the field between vascular risk factors including diabetes and obesity being associated with increased risk of dementia. The GLP-1 drug should help reduce these risk factors as well as potentially directly protecting brain cells,” Dr. Spires-Jones said.
However, she said “more research in bigger trials is needed to confirm whether this type of treatment will be effective in people with Alzheimer’s disease.”
Stephen Evans, MSc, emeritus professor, London School of Hygiene and Tropical Medicine, noted that the repurposing of drugs is “an important avenue of research but there is a lot of uncertainty here.”
He cautioned that the “50% brain volume change may not translate to important cognitive effects, and reporting only on those who completed the full 52 weeks of treatment could bring bias into the results. It sounds like it is worth pursuing a larger trial, but these results cannot demonstrate that liraglutide can protect against dementia.”
The ongoing phase 3 EVOKE trial is investigating the effects of the GLP-1 receptor agonist semaglutide in early Alzheimer’s disease.
Funding for the study was provided by Alzheimer’s Society UK, Alzheimer’s Drug Discovery Foundation, Novo Nordisk, John and Lucille Van Geest Foundation, and the National Institute for Health and Care Research Biomedical Research Centre. Dr. Edison, Dr. Fillit, Dr. Snyder, Mr. Evans, and Dr. Spires-Jones had no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
PHILADELPHIA –
In the phase 2b ELAD clinical trial, adults with early-stage Alzheimer’s disease taking the GLP-1 receptor agonist liraglutide exhibited slower decline in memory and thinking and experienced less brain atrophy over 12 months, compared with placebo.
“The slower loss of brain volume suggests liraglutide protects the brain, much like statins protect the heart,” study chief Paul Edison, MD, PhD, with Imperial College London, London, England, said in a statement.
“While further research is needed, liraglutide may work through various mechanisms, such as reducing inflammation in the brain, lowering insulin resistance and the toxic effects of Alzheimer’s biomarkers amyloid beta and tau, and improving how the brain’s nerve cells communicate,” Dr. Edison said.
He presented the study results at the 2024 Alzheimer’s Association International Conference (AAIC).
Brain Benefits
Liraglutide has previously demonstrated promising neuroprotective effects in animal models of Alzheimer’s disease and epidemiologic studies.
In ELAD, 204 patients with mild to moderate Alzheimer’s disease were randomly allocated (1:1) to a daily subcutaneous injection of up to 1.8 mg of liraglutide or placebo for 12 months; 80 patients in the liraglutide group and 89 in the placebo group completed the study.
Brain MRI was performed at baseline and at 12 months, along with neuropsychometric evaluation and 18F-fludeoxyglucose PET.
The study’s primary endpoint — change in the cerebral glucose metabolic rate in the cortical regions of the brain (hippocampus, medial temporal lobe, and posterior cingulate) — was not met.
However, patients taking liraglutide experienced a significant slowing of cognitive decline, compared with placebo group (P = .01), which was a key secondary outcome, calculated as a composite score of 18 different tests of memory, comprehension, language, and spatial orientation.
Although the study was not powered to assess cognitive changes, adults taking liraglutide had an 18% slower decline in cognitive function over 12 months, compared with those on placebo, Dr. Edison reported.
In addition, patients treated with liraglutide had nearly 50% less volume loss in several areas of the brain involved in memory, language, and decision-making, including frontal, temporal, parietal, and total gray matter, as measured by MRI.
Liraglutide daily subcutaneous injections were safe and well tolerated in patients with Alzheimer’s disease, Dr. Edison reported. There were 25 serious side effects — 18 in the placebo group and 7 in the liraglutide group — and most were considered unlikely to be related to the study treatment. There were no deaths.
Promising, Preliminary
This study shows a positive effect of liraglutide on the brain in terms of “slowing down of brain atrophy and slowing down the rate of cognitive decline,” said Howard Fillit, MD, founding executive director of the Alzheimer’s Drug Discovery Foundation, who wasn’t involved in the study.
Heather Snyder, PhD, vice-president of medical and scientific relations at the Alzheimer’s Association, said it’s “interesting” to see slowing of brain volume loss and some cognitive benefit “especially as the study was not powered necessarily to see some of those changes. The fact that they did see these changes in this small study provides a window into what may happen, but we certainly need larger phase 3 studies.”
In a statement from the UK nonprofit Science Media Centre, Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, called the data “promising.”
“There are clear links from strong data in the field between vascular risk factors including diabetes and obesity being associated with increased risk of dementia. The GLP-1 drug should help reduce these risk factors as well as potentially directly protecting brain cells,” Dr. Spires-Jones said.
However, she said “more research in bigger trials is needed to confirm whether this type of treatment will be effective in people with Alzheimer’s disease.”
Stephen Evans, MSc, emeritus professor, London School of Hygiene and Tropical Medicine, noted that the repurposing of drugs is “an important avenue of research but there is a lot of uncertainty here.”
He cautioned that the “50% brain volume change may not translate to important cognitive effects, and reporting only on those who completed the full 52 weeks of treatment could bring bias into the results. It sounds like it is worth pursuing a larger trial, but these results cannot demonstrate that liraglutide can protect against dementia.”
The ongoing phase 3 EVOKE trial is investigating the effects of the GLP-1 receptor agonist semaglutide in early Alzheimer’s disease.
Funding for the study was provided by Alzheimer’s Society UK, Alzheimer’s Drug Discovery Foundation, Novo Nordisk, John and Lucille Van Geest Foundation, and the National Institute for Health and Care Research Biomedical Research Centre. Dr. Edison, Dr. Fillit, Dr. Snyder, Mr. Evans, and Dr. Spires-Jones had no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
PHILADELPHIA –
In the phase 2b ELAD clinical trial, adults with early-stage Alzheimer’s disease taking the GLP-1 receptor agonist liraglutide exhibited slower decline in memory and thinking and experienced less brain atrophy over 12 months, compared with placebo.
“The slower loss of brain volume suggests liraglutide protects the brain, much like statins protect the heart,” study chief Paul Edison, MD, PhD, with Imperial College London, London, England, said in a statement.
“While further research is needed, liraglutide may work through various mechanisms, such as reducing inflammation in the brain, lowering insulin resistance and the toxic effects of Alzheimer’s biomarkers amyloid beta and tau, and improving how the brain’s nerve cells communicate,” Dr. Edison said.
He presented the study results at the 2024 Alzheimer’s Association International Conference (AAIC).
Brain Benefits
Liraglutide has previously demonstrated promising neuroprotective effects in animal models of Alzheimer’s disease and epidemiologic studies.
In ELAD, 204 patients with mild to moderate Alzheimer’s disease were randomly allocated (1:1) to a daily subcutaneous injection of up to 1.8 mg of liraglutide or placebo for 12 months; 80 patients in the liraglutide group and 89 in the placebo group completed the study.
Brain MRI was performed at baseline and at 12 months, along with neuropsychometric evaluation and 18F-fludeoxyglucose PET.
The study’s primary endpoint — change in the cerebral glucose metabolic rate in the cortical regions of the brain (hippocampus, medial temporal lobe, and posterior cingulate) — was not met.
However, patients taking liraglutide experienced a significant slowing of cognitive decline, compared with placebo group (P = .01), which was a key secondary outcome, calculated as a composite score of 18 different tests of memory, comprehension, language, and spatial orientation.
Although the study was not powered to assess cognitive changes, adults taking liraglutide had an 18% slower decline in cognitive function over 12 months, compared with those on placebo, Dr. Edison reported.
In addition, patients treated with liraglutide had nearly 50% less volume loss in several areas of the brain involved in memory, language, and decision-making, including frontal, temporal, parietal, and total gray matter, as measured by MRI.
Liraglutide daily subcutaneous injections were safe and well tolerated in patients with Alzheimer’s disease, Dr. Edison reported. There were 25 serious side effects — 18 in the placebo group and 7 in the liraglutide group — and most were considered unlikely to be related to the study treatment. There were no deaths.
Promising, Preliminary
This study shows a positive effect of liraglutide on the brain in terms of “slowing down of brain atrophy and slowing down the rate of cognitive decline,” said Howard Fillit, MD, founding executive director of the Alzheimer’s Drug Discovery Foundation, who wasn’t involved in the study.
Heather Snyder, PhD, vice-president of medical and scientific relations at the Alzheimer’s Association, said it’s “interesting” to see slowing of brain volume loss and some cognitive benefit “especially as the study was not powered necessarily to see some of those changes. The fact that they did see these changes in this small study provides a window into what may happen, but we certainly need larger phase 3 studies.”
In a statement from the UK nonprofit Science Media Centre, Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, called the data “promising.”
“There are clear links from strong data in the field between vascular risk factors including diabetes and obesity being associated with increased risk of dementia. The GLP-1 drug should help reduce these risk factors as well as potentially directly protecting brain cells,” Dr. Spires-Jones said.
However, she said “more research in bigger trials is needed to confirm whether this type of treatment will be effective in people with Alzheimer’s disease.”
Stephen Evans, MSc, emeritus professor, London School of Hygiene and Tropical Medicine, noted that the repurposing of drugs is “an important avenue of research but there is a lot of uncertainty here.”
He cautioned that the “50% brain volume change may not translate to important cognitive effects, and reporting only on those who completed the full 52 weeks of treatment could bring bias into the results. It sounds like it is worth pursuing a larger trial, but these results cannot demonstrate that liraglutide can protect against dementia.”
The ongoing phase 3 EVOKE trial is investigating the effects of the GLP-1 receptor agonist semaglutide in early Alzheimer’s disease.
Funding for the study was provided by Alzheimer’s Society UK, Alzheimer’s Drug Discovery Foundation, Novo Nordisk, John and Lucille Van Geest Foundation, and the National Institute for Health and Care Research Biomedical Research Centre. Dr. Edison, Dr. Fillit, Dr. Snyder, Mr. Evans, and Dr. Spires-Jones had no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM AAIC 2024
Could Medium-Chain Fatty Acids Reduce Diabetes Risk?
TOPLINE:
Higher levels of some serum medium-chain fatty acids found in coconut oil, palm kernel oil, and milk products are associated with a reduced risk for type 2 diabetes (T2D). This inverse relationship is more pronounced in individuals with a high genetic risk or physical inactivity.
METHODOLOGY:
- Studies reporting a link between dietary medium-chain fatty acids and a reduced risk for T2D have been based on food intake questionnaires, but serum samples are likely to be a more precise and objective basis for understanding metabolic relationships.
- To assess the association between medium-chain fatty acids and T2D risk, the researchers conducted a nested case-control study within the prospective China Cardiometabolic Disease and Cancer Cohort Study.
- They included 1707 individuals who developed diabetes during a median follow-up of 3.03 years and added a propensity-matched normoglycemic control group for a total of 3414 individuals (mean age, 57.56 years; 59.4% women), all with normal glucose regulation at baseline.
- Researchers investigated associations of baseline levels of five serum medium-chain fatty acids — octanoic acid, nonanoic acid, decanoic acid, undecanoic acid, and lauric acid — between individuals with T2D and control participants and stratified by risk factors, including diabetes genetic susceptibility.
- The genetic risk scores were calculated as a weighted sum of 86 T2D-associated single nucleotide polymorphisms.
TAKEAWAY:
- In an inverse association, each standard deviation increase in the baseline serum levels of octanoic acid and nonanoic acid decreased the odds of T2D by 10% and 16%, respectively (odds ratio [OR], 0.90; 95% CI, 0.82-0.98 and OR, 0.84; 95% CI, 0.74-0.95, respectively; all P < .05).
- , with significant interactions observed for octanoic, nonanoic, and decanoic acids (P for interaction = .042, .034, and .037, respectively).
- Moreover, the negative relationship between octanoic acid and the risk for diabetes was stronger in those with a high genetic risk, with a significant interaction (P for interaction = .003).
- No significant associations were observed between the levels of decanoic, undecanoic, and lauric acids and the overall risk for incident diabetes.
IN PRACTICE:
“Our findings generally support the protective effect of MCFAs [medium-chain fatty acids] but also emphasize the personalized approaches in improving serum MCFA profiles for T2D prevention, which could be tailored according to individuals’ genetic and lifestyle profiles,” the authors wrote.
SOURCE:
The study was led by Xiaojing Jia, MD, and Hong Lin, PhD, of the Shanghai Institute of Endocrine and Metabolic Diseases, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. It was published online in The Journal of Clinical Endocrinology & Metabolism.
LIMITATIONS:
The study’s follow-up duration of 3 years was short, which may have compromised the statistical power of the analysis. The long-term effects of medium-chain fatty acids on the risk for diabetes may not be captured as they were assessed only at baseline. The study population was limited to Chinese adults older than 40 years, which may affect the generalizability of the findings to other ethnicities and age groups.
DISCLOSURES:
The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Higher levels of some serum medium-chain fatty acids found in coconut oil, palm kernel oil, and milk products are associated with a reduced risk for type 2 diabetes (T2D). This inverse relationship is more pronounced in individuals with a high genetic risk or physical inactivity.
METHODOLOGY:
- Studies reporting a link between dietary medium-chain fatty acids and a reduced risk for T2D have been based on food intake questionnaires, but serum samples are likely to be a more precise and objective basis for understanding metabolic relationships.
- To assess the association between medium-chain fatty acids and T2D risk, the researchers conducted a nested case-control study within the prospective China Cardiometabolic Disease and Cancer Cohort Study.
- They included 1707 individuals who developed diabetes during a median follow-up of 3.03 years and added a propensity-matched normoglycemic control group for a total of 3414 individuals (mean age, 57.56 years; 59.4% women), all with normal glucose regulation at baseline.
- Researchers investigated associations of baseline levels of five serum medium-chain fatty acids — octanoic acid, nonanoic acid, decanoic acid, undecanoic acid, and lauric acid — between individuals with T2D and control participants and stratified by risk factors, including diabetes genetic susceptibility.
- The genetic risk scores were calculated as a weighted sum of 86 T2D-associated single nucleotide polymorphisms.
TAKEAWAY:
- In an inverse association, each standard deviation increase in the baseline serum levels of octanoic acid and nonanoic acid decreased the odds of T2D by 10% and 16%, respectively (odds ratio [OR], 0.90; 95% CI, 0.82-0.98 and OR, 0.84; 95% CI, 0.74-0.95, respectively; all P < .05).
- , with significant interactions observed for octanoic, nonanoic, and decanoic acids (P for interaction = .042, .034, and .037, respectively).
- Moreover, the negative relationship between octanoic acid and the risk for diabetes was stronger in those with a high genetic risk, with a significant interaction (P for interaction = .003).
- No significant associations were observed between the levels of decanoic, undecanoic, and lauric acids and the overall risk for incident diabetes.
IN PRACTICE:
“Our findings generally support the protective effect of MCFAs [medium-chain fatty acids] but also emphasize the personalized approaches in improving serum MCFA profiles for T2D prevention, which could be tailored according to individuals’ genetic and lifestyle profiles,” the authors wrote.
SOURCE:
The study was led by Xiaojing Jia, MD, and Hong Lin, PhD, of the Shanghai Institute of Endocrine and Metabolic Diseases, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. It was published online in The Journal of Clinical Endocrinology & Metabolism.
LIMITATIONS:
The study’s follow-up duration of 3 years was short, which may have compromised the statistical power of the analysis. The long-term effects of medium-chain fatty acids on the risk for diabetes may not be captured as they were assessed only at baseline. The study population was limited to Chinese adults older than 40 years, which may affect the generalizability of the findings to other ethnicities and age groups.
DISCLOSURES:
The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Higher levels of some serum medium-chain fatty acids found in coconut oil, palm kernel oil, and milk products are associated with a reduced risk for type 2 diabetes (T2D). This inverse relationship is more pronounced in individuals with a high genetic risk or physical inactivity.
METHODOLOGY:
- Studies reporting a link between dietary medium-chain fatty acids and a reduced risk for T2D have been based on food intake questionnaires, but serum samples are likely to be a more precise and objective basis for understanding metabolic relationships.
- To assess the association between medium-chain fatty acids and T2D risk, the researchers conducted a nested case-control study within the prospective China Cardiometabolic Disease and Cancer Cohort Study.
- They included 1707 individuals who developed diabetes during a median follow-up of 3.03 years and added a propensity-matched normoglycemic control group for a total of 3414 individuals (mean age, 57.56 years; 59.4% women), all with normal glucose regulation at baseline.
- Researchers investigated associations of baseline levels of five serum medium-chain fatty acids — octanoic acid, nonanoic acid, decanoic acid, undecanoic acid, and lauric acid — between individuals with T2D and control participants and stratified by risk factors, including diabetes genetic susceptibility.
- The genetic risk scores were calculated as a weighted sum of 86 T2D-associated single nucleotide polymorphisms.
TAKEAWAY:
- In an inverse association, each standard deviation increase in the baseline serum levels of octanoic acid and nonanoic acid decreased the odds of T2D by 10% and 16%, respectively (odds ratio [OR], 0.90; 95% CI, 0.82-0.98 and OR, 0.84; 95% CI, 0.74-0.95, respectively; all P < .05).
- , with significant interactions observed for octanoic, nonanoic, and decanoic acids (P for interaction = .042, .034, and .037, respectively).
- Moreover, the negative relationship between octanoic acid and the risk for diabetes was stronger in those with a high genetic risk, with a significant interaction (P for interaction = .003).
- No significant associations were observed between the levels of decanoic, undecanoic, and lauric acids and the overall risk for incident diabetes.
IN PRACTICE:
“Our findings generally support the protective effect of MCFAs [medium-chain fatty acids] but also emphasize the personalized approaches in improving serum MCFA profiles for T2D prevention, which could be tailored according to individuals’ genetic and lifestyle profiles,” the authors wrote.
SOURCE:
The study was led by Xiaojing Jia, MD, and Hong Lin, PhD, of the Shanghai Institute of Endocrine and Metabolic Diseases, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. It was published online in The Journal of Clinical Endocrinology & Metabolism.
LIMITATIONS:
The study’s follow-up duration of 3 years was short, which may have compromised the statistical power of the analysis. The long-term effects of medium-chain fatty acids on the risk for diabetes may not be captured as they were assessed only at baseline. The study population was limited to Chinese adults older than 40 years, which may affect the generalizability of the findings to other ethnicities and age groups.
DISCLOSURES:
The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Compounded Semaglutide Overdoses Tied to Hospitalizations
Patients are overdosing on compounded semaglutide due to errors in measuring and self-administering the drug and due to clinicians miscalculating doses that may differ from US Food and Drug Administration (FDA)–approved products.
The FDA published an alert on July 26 after receiving reports of dosing errors involving compounded semaglutide injectable products dispensed in multidose vials. Adverse events included gastrointestinal effects, fainting, dehydration, headache, gallstones, and acute pancreatitis. Some patients required hospitalization.
Why the Risks?
FDA-approved semaglutide injectable products are dosed in milligrams, have standard concentrations, and are currently only available in prefilled pens.
Compounded semaglutide products may differ from approved products in ways that contribute to potential errors — for example, in multidose vials and prefilled syringes. In addition, product concentrations may vary depending on the compounder, and even a single compounder may offer multiple concentrations of semaglutide.
Instructions for a compounded drug, if provided, may tell users to administer semaglutide injections in “units,” the volume of which may vary depending on the concentration — rather than in milligrams. In some instances, patients received syringes significantly larger than the prescribed volume.
Common Errors
The FDA has received reports related to patients mistakenly taking more than the prescribed dose from a multidose vial — sometimes 5-20 times more than the intended dose.
Several reports described clinicians incorrectly calculating the intended dose when converting from milligrams to units or milliliters. In one case, a patient couldn’t get clarity on dosing instructions from the telemedicine provider who prescribed the compounded semaglutide, leading the patient to search online for medical advice. This resulted in the patient taking five times the intended dose.
In another example, one clinician prescribed 20 units instead of two units, affecting three patients who, after receiving 10 times the intended dose, experienced nausea and vomiting.
Another clinician, who also takes semaglutide himself, tried to recalculate his own dose in units and ended up self-administering a dose 10 times higher than intended.
The FDA previously warned about potential risks from the use of compounded drugs during a shortage as is the case with semaglutide. While compounded drugs can “sometimes” be helpful, according to the agency, “compounded drugs pose a higher risk to patients than FDA-approved drugs because compounded drugs do not undergo FDA premarket review for safety, effectiveness, or quality.”
Patients are overdosing on compounded semaglutide due to errors in measuring and self-administering the drug and due to clinicians miscalculating doses that may differ from US Food and Drug Administration (FDA)–approved products.
The FDA published an alert on July 26 after receiving reports of dosing errors involving compounded semaglutide injectable products dispensed in multidose vials. Adverse events included gastrointestinal effects, fainting, dehydration, headache, gallstones, and acute pancreatitis. Some patients required hospitalization.
Why the Risks?
FDA-approved semaglutide injectable products are dosed in milligrams, have standard concentrations, and are currently only available in prefilled pens.
Compounded semaglutide products may differ from approved products in ways that contribute to potential errors — for example, in multidose vials and prefilled syringes. In addition, product concentrations may vary depending on the compounder, and even a single compounder may offer multiple concentrations of semaglutide.
Instructions for a compounded drug, if provided, may tell users to administer semaglutide injections in “units,” the volume of which may vary depending on the concentration — rather than in milligrams. In some instances, patients received syringes significantly larger than the prescribed volume.
Common Errors
The FDA has received reports related to patients mistakenly taking more than the prescribed dose from a multidose vial — sometimes 5-20 times more than the intended dose.
Several reports described clinicians incorrectly calculating the intended dose when converting from milligrams to units or milliliters. In one case, a patient couldn’t get clarity on dosing instructions from the telemedicine provider who prescribed the compounded semaglutide, leading the patient to search online for medical advice. This resulted in the patient taking five times the intended dose.
In another example, one clinician prescribed 20 units instead of two units, affecting three patients who, after receiving 10 times the intended dose, experienced nausea and vomiting.
Another clinician, who also takes semaglutide himself, tried to recalculate his own dose in units and ended up self-administering a dose 10 times higher than intended.
The FDA previously warned about potential risks from the use of compounded drugs during a shortage as is the case with semaglutide. While compounded drugs can “sometimes” be helpful, according to the agency, “compounded drugs pose a higher risk to patients than FDA-approved drugs because compounded drugs do not undergo FDA premarket review for safety, effectiveness, or quality.”
Patients are overdosing on compounded semaglutide due to errors in measuring and self-administering the drug and due to clinicians miscalculating doses that may differ from US Food and Drug Administration (FDA)–approved products.
The FDA published an alert on July 26 after receiving reports of dosing errors involving compounded semaglutide injectable products dispensed in multidose vials. Adverse events included gastrointestinal effects, fainting, dehydration, headache, gallstones, and acute pancreatitis. Some patients required hospitalization.
Why the Risks?
FDA-approved semaglutide injectable products are dosed in milligrams, have standard concentrations, and are currently only available in prefilled pens.
Compounded semaglutide products may differ from approved products in ways that contribute to potential errors — for example, in multidose vials and prefilled syringes. In addition, product concentrations may vary depending on the compounder, and even a single compounder may offer multiple concentrations of semaglutide.
Instructions for a compounded drug, if provided, may tell users to administer semaglutide injections in “units,” the volume of which may vary depending on the concentration — rather than in milligrams. In some instances, patients received syringes significantly larger than the prescribed volume.
Common Errors
The FDA has received reports related to patients mistakenly taking more than the prescribed dose from a multidose vial — sometimes 5-20 times more than the intended dose.
Several reports described clinicians incorrectly calculating the intended dose when converting from milligrams to units or milliliters. In one case, a patient couldn’t get clarity on dosing instructions from the telemedicine provider who prescribed the compounded semaglutide, leading the patient to search online for medical advice. This resulted in the patient taking five times the intended dose.
In another example, one clinician prescribed 20 units instead of two units, affecting three patients who, after receiving 10 times the intended dose, experienced nausea and vomiting.
Another clinician, who also takes semaglutide himself, tried to recalculate his own dose in units and ended up self-administering a dose 10 times higher than intended.
The FDA previously warned about potential risks from the use of compounded drugs during a shortage as is the case with semaglutide. While compounded drugs can “sometimes” be helpful, according to the agency, “compounded drugs pose a higher risk to patients than FDA-approved drugs because compounded drugs do not undergo FDA premarket review for safety, effectiveness, or quality.”
What Time of Day Is Best to Eat to Reduce Diabetes Risk?
TOPLINE:
Higher energy intake and glycemic load in the late morning are associated with a lower risk for type 2 diabetes (T2D) in Hispanic/Latino adults.
METHODOLOGY:
- Glucose tolerance peaks in the morning and declines in the afternoon and evening in individuals without diabetes.
- Researchers conducted a prospective cohort study enrolling 8868 Hispanic/Latino adults (mean age, 38.7 years; 51.5% women) without diabetes across four US communities between 2008 and 2011, with a second clinic examination conducted between 2014 and 2017.
- Meal timing was categorized into five periods: Early morning (6:00-8:59 AM), late morning (9:00-11:59 AM), afternoon (12:00-5:59 PM), evening (6:00-11:59 PM), and night (0:00-5:59 AM).
- Participants’ energy intake and glycemic load for each period were assessed at baseline using two 24-hour dietary recalls.
- Incident diabetes was identified through annual follow-up calls or at the second clinic examination.
TAKEAWAY:
- Each 100-kcal increment in energy intake and 10-unit increment in glycemic load in the late morning was associated with a 6% and 7% lower risk for T2D, respectively (both P = .001), independent of total energy intake, diet quality, and other confounders.
- No such association was found between energy intake and glycemic load in early morning, afternoon, evening, or night meal timings and the risk for diabetes.
- Substituting 100 kcal of energy intake from the early morning, afternoon, or evening with late-morning equivalents was associated with a 5% lower risk for diabetes (all P < .05).
- Similarly, substituting 10 units of energy-adjusted glycemic load from the early morning, afternoon, or evening with late-morning equivalents yielded a 7%-9% lower risk for diabetes (all P < .05).
IN PRACTICE:
“Our findings further enhance the existing literature by demonstrating the potential long-term promise of eating in alignment with the diurnal rhythm of glucose tolerance for diabetes prevention,” the authors wrote.
SOURCE:
The study was led by Jin Dai, PhD, Fielding School of Public Health, University of California, Los Angeles. It was published online in Diabetes Care.
LIMITATIONS:
The study’s reliance on only two 24-hour self-reported dietary recalls may have introduced measurement error. Diabetes was self-reported, which may have led to outcome misclassification. The study’s relatively short follow-up time may have introduced reverse causation bias. As most patients had T2D, the findings predominately apply to this diabetes subtype.
DISCLOSURES:
The study was supported by grants from the National Heart, Lung, and Blood Institute. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Higher energy intake and glycemic load in the late morning are associated with a lower risk for type 2 diabetes (T2D) in Hispanic/Latino adults.
METHODOLOGY:
- Glucose tolerance peaks in the morning and declines in the afternoon and evening in individuals without diabetes.
- Researchers conducted a prospective cohort study enrolling 8868 Hispanic/Latino adults (mean age, 38.7 years; 51.5% women) without diabetes across four US communities between 2008 and 2011, with a second clinic examination conducted between 2014 and 2017.
- Meal timing was categorized into five periods: Early morning (6:00-8:59 AM), late morning (9:00-11:59 AM), afternoon (12:00-5:59 PM), evening (6:00-11:59 PM), and night (0:00-5:59 AM).
- Participants’ energy intake and glycemic load for each period were assessed at baseline using two 24-hour dietary recalls.
- Incident diabetes was identified through annual follow-up calls or at the second clinic examination.
TAKEAWAY:
- Each 100-kcal increment in energy intake and 10-unit increment in glycemic load in the late morning was associated with a 6% and 7% lower risk for T2D, respectively (both P = .001), independent of total energy intake, diet quality, and other confounders.
- No such association was found between energy intake and glycemic load in early morning, afternoon, evening, or night meal timings and the risk for diabetes.
- Substituting 100 kcal of energy intake from the early morning, afternoon, or evening with late-morning equivalents was associated with a 5% lower risk for diabetes (all P < .05).
- Similarly, substituting 10 units of energy-adjusted glycemic load from the early morning, afternoon, or evening with late-morning equivalents yielded a 7%-9% lower risk for diabetes (all P < .05).
IN PRACTICE:
“Our findings further enhance the existing literature by demonstrating the potential long-term promise of eating in alignment with the diurnal rhythm of glucose tolerance for diabetes prevention,” the authors wrote.
SOURCE:
The study was led by Jin Dai, PhD, Fielding School of Public Health, University of California, Los Angeles. It was published online in Diabetes Care.
LIMITATIONS:
The study’s reliance on only two 24-hour self-reported dietary recalls may have introduced measurement error. Diabetes was self-reported, which may have led to outcome misclassification. The study’s relatively short follow-up time may have introduced reverse causation bias. As most patients had T2D, the findings predominately apply to this diabetes subtype.
DISCLOSURES:
The study was supported by grants from the National Heart, Lung, and Blood Institute. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Higher energy intake and glycemic load in the late morning are associated with a lower risk for type 2 diabetes (T2D) in Hispanic/Latino adults.
METHODOLOGY:
- Glucose tolerance peaks in the morning and declines in the afternoon and evening in individuals without diabetes.
- Researchers conducted a prospective cohort study enrolling 8868 Hispanic/Latino adults (mean age, 38.7 years; 51.5% women) without diabetes across four US communities between 2008 and 2011, with a second clinic examination conducted between 2014 and 2017.
- Meal timing was categorized into five periods: Early morning (6:00-8:59 AM), late morning (9:00-11:59 AM), afternoon (12:00-5:59 PM), evening (6:00-11:59 PM), and night (0:00-5:59 AM).
- Participants’ energy intake and glycemic load for each period were assessed at baseline using two 24-hour dietary recalls.
- Incident diabetes was identified through annual follow-up calls or at the second clinic examination.
TAKEAWAY:
- Each 100-kcal increment in energy intake and 10-unit increment in glycemic load in the late morning was associated with a 6% and 7% lower risk for T2D, respectively (both P = .001), independent of total energy intake, diet quality, and other confounders.
- No such association was found between energy intake and glycemic load in early morning, afternoon, evening, or night meal timings and the risk for diabetes.
- Substituting 100 kcal of energy intake from the early morning, afternoon, or evening with late-morning equivalents was associated with a 5% lower risk for diabetes (all P < .05).
- Similarly, substituting 10 units of energy-adjusted glycemic load from the early morning, afternoon, or evening with late-morning equivalents yielded a 7%-9% lower risk for diabetes (all P < .05).
IN PRACTICE:
“Our findings further enhance the existing literature by demonstrating the potential long-term promise of eating in alignment with the diurnal rhythm of glucose tolerance for diabetes prevention,” the authors wrote.
SOURCE:
The study was led by Jin Dai, PhD, Fielding School of Public Health, University of California, Los Angeles. It was published online in Diabetes Care.
LIMITATIONS:
The study’s reliance on only two 24-hour self-reported dietary recalls may have introduced measurement error. Diabetes was self-reported, which may have led to outcome misclassification. The study’s relatively short follow-up time may have introduced reverse causation bias. As most patients had T2D, the findings predominately apply to this diabetes subtype.
DISCLOSURES:
The study was supported by grants from the National Heart, Lung, and Blood Institute. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
A Guide to Eating Healthy While Working in Healthcare
Eat as fast as you can whenever you can.
That was the med student mindset around food, as Catherine Harmon Toomer, MD, discovered during her school years. “Without a good system in place to counter that,” she explains, “unhealthy eating can get out of control, and that’s what happened to me.”
After med school, things got worse for Dr. Toomer. By her second year in practice as a family medicine physician, she’d gained a lot of weight and had been diagnosed with type 2 diabetes and cardiomyopathy. At 36, she went into congestive heart failure and was told she likely had 5 years to live.
A moment she described as “a huge wake-up call.”
Dr. Toomer is far from alone in her struggles to balance working in medicine and eating healthfully.
internist and cofounder of ChefMD and founder of Chef Clinic.
There is also the culture of medicine, which Dr. Toomer said looks down on self-care. “Even with break times, patient needs come before our own.” So, you sit down to eat, and there’s an emergency. Your clinic closes for lunch, but the phones still ring, and patients continue to email questions. Charting is also so time-consuming that “everything else gets put on the back burner.”
Sticking to a nutritious diet in this context can feel hopeless. But it isn’t. Really. Here are some doctor-tested, real-life ways you can nourish yourself while getting it all done.
Something Is Always Better Than Nothing
Sure, you might not be able to eat a balanced lunch or dinner while at work, conceded Amy Margulies, RD, LDN, owner of The Rebellious RD. But try to focus on the bigger picture and take small steps.
First, make sure you eat something, Ms. Margulies advised. “Skipping meals can lead to overeating later and negatively impact energy levels and concentration.”
Lisa Andrews, MEd, RD, LD, owner of Sound Bites Nutrition, recalled one of her patients, a gastrointestinal surgeon with reactive hypoglycemia and fatigue. “She was experiencing energy crashes mid-afternoon,” she said. It was only after starting to eat every 4-5 hours that her patient felt better.
Of course, this is easier said than done. “When you are running from one patient to the other and trying to keep on time with your schedule, there is very little time for eating and no time at all for cooking or even heating up food,” recalled Hélène Bertrand, MD, author of Low Back Pain: 3 Steps to Relief in 2 Minutes.
But during her 55 years as a family medicine physician, Dr. Bertrand found ways to improve (if not perfect) the situation. She lunched on nuts or seeds during the day or grabbed a 95% cacao chocolate bar — higher in antioxidants and lower in sugar than a candy bar.
If you don’t have time for breakfast, try drinking a complete protein shake while driving to work, Dr. Toomer recommended. “It’s not ideal, but it’s better than nothing.” Similarly, if the only way you’ll eat a high-protein, lower-carb snack like hummus is with potato chips, go for it, she said.
Basically, don’t be type A striving for perfection. Take good enough when you can and balance the rest when you have time.
Torpedo Temptation
From free treats in the break room to always-present pizza for residents, high-fat, high-sugar, low-nutrient fare is a constant temptation. “I worked with a physician who would bring a balanced lunch to work every day, then find whatever sweet was around for his afternoon treat,” recalled Ms. Margulies.“The cookies, cakes, and donuts were starting to add up — and stopping at one wasn’t working for him.”
What did work was Ms. Margulies’ suggestion to bring a single serving of dark chocolate and fruit to savor during a longer break. “Bringing your favorite treats in appropriate portions can help you stick with your plan throughout the day,” she explained, and you’ll have an easier time resisting what’s in the break room. “When you desire a treat, tell yourself you have what you need and don’t need to indulge in the ‘free food’ just because it’s there. You have power over your choices.”
How about tricking yourself into perceiving cherry tomatoes as treats? That might be unusual, but one of Dr. La Puma’s physician patients did just that, displaying the produce in a candy dish on his office counter. Not only did this strategy help remind him to snack healthfully, it also prompted his patients to ask about eating better, he said.
Preparation Is Still Underrated
Many people find meal prepping intimidating. But it doesn’t need to be complicated. For instance, try purchasing precut veggies, cooked chicken breasts, or other healthy convenience options. You can then combine them in packable containers to prep a few meals at a time. For less busy weeks, consider cooking the protein yourself and whipping up basic sauces (like pesto and vinaigrette) to jazz up your meals.
“I worked with a resident who was gaining weight each month,” recalled Ms. Margulies. “She would skip lunch, grab a random snack, then wait until she got home to eat anything she could find.”
Encouraged by Ms. Margulies, she prepared and portioned one or two balanced dinners each week, which she’d later reheat. She also bought fresh and dried fruit and high-protein snacks, keeping single servings in her car to eat on the way home.
Similarly, Jess DeGore, RD, LDN, CDCES, CHWC, a diabetes educator and owner of Dietitian Jess Nutrition, recalled an ob.gyn. client who constantly skipped meals and relied on vending machine snacks. To combat her resulting energy crashes, she followed Ms. DeGore’s advice to prep workday lunches (like quinoa salads) over the weekend and bring fruit and nut snacks to work.
Automate as Much as You Can
If healthy is already on hand, you’ll eat healthy, said Ms. Andrews. Build up a snack stash focusing on fiber and protein. Tote a lunch bag with a cooler pack if needed. Some suggestions:
- Oatmeal packets
- Individual Greek yogurt cups or drinkable yogurts
- Protein bars
- Protein shakes
- Fresh fruit
- Fresh veggie sticks
- Nuts, dried chickpeas, or edamame
- Trail mix
- Single servings of hummus, nut butter, or guacamole
- Dried seaweed snacks
- Whole grain crackers
- Hard-boiled eggs
- String cheese
- Peanut butter sandwich
- 95% cacao chocolate bar
Try a Meal Delivery Service
Meal delivery services can be pricey, but potentially worth the expense. By bringing meals or having them sent to your office, you won’t have to find time to go to the cafeteria and stand in line, noted Janese S. Laster, MD, an internal medicine, gastroenterology, obesity medicine, and nutrition physician and founder of Gut Theory Total Digestive Care. Instead, “you’ll have something to warm up and eat while writing notes or in between patients,” she said. Plus, “you won’t have an excuse to skip meals.”
Hydration Yes, Junk Drinks No
The following can be filed in the Doctors-Know-It-But-Don’t-Always-Do-It section: “Hunger can be mistaken for thirst,” said Ms. Margulies. “Staying hydrated will help you better assess whether you’re hungry or thirsty.” Choose water over soda or energy drinks, she added, to hydrate your body without unnecessary extra sugars, sugar substitutes, calories, caffeine, or sodium — all of which can affect how you feel.
Advocate for Your Health
Convincing your institution to make changes might be difficult or even impossible, but consider asking your workplace to implement initiatives like these to boost provider nutrition, suggested Jabe Brown, BHSc (Nat), founder of Melbourne Functional Medicine:
- Establish protected break times when doctors can step away from their duties to eat
- Add more nutritious cafeteria options, like salads, whole grains, and lean proteins
- Overhaul vending machine offerings
- Offer educational workshops on nutrition
Be Tenacious About Good Eating
For Dr. Toomer, that meant taking several years off from work to improve her health. After losing more than 100 pounds, she founded TOTAL Weight Care Institute to help other healthcare professionals follow in her footsteps.
For you, the path toward a healthier diet might be gradual — grabbing a more nutritious snack, spending an extra hour per week on food shopping or prep, remembering a water bottle. Whatever it looks like, make realistic lifestyle tweaks that work for you.
Maybe even try that apple-a-day thing.
A version of this article first appeared on Medscape.com.
Eat as fast as you can whenever you can.
That was the med student mindset around food, as Catherine Harmon Toomer, MD, discovered during her school years. “Without a good system in place to counter that,” she explains, “unhealthy eating can get out of control, and that’s what happened to me.”
After med school, things got worse for Dr. Toomer. By her second year in practice as a family medicine physician, she’d gained a lot of weight and had been diagnosed with type 2 diabetes and cardiomyopathy. At 36, she went into congestive heart failure and was told she likely had 5 years to live.
A moment she described as “a huge wake-up call.”
Dr. Toomer is far from alone in her struggles to balance working in medicine and eating healthfully.
internist and cofounder of ChefMD and founder of Chef Clinic.
There is also the culture of medicine, which Dr. Toomer said looks down on self-care. “Even with break times, patient needs come before our own.” So, you sit down to eat, and there’s an emergency. Your clinic closes for lunch, but the phones still ring, and patients continue to email questions. Charting is also so time-consuming that “everything else gets put on the back burner.”
Sticking to a nutritious diet in this context can feel hopeless. But it isn’t. Really. Here are some doctor-tested, real-life ways you can nourish yourself while getting it all done.
Something Is Always Better Than Nothing
Sure, you might not be able to eat a balanced lunch or dinner while at work, conceded Amy Margulies, RD, LDN, owner of The Rebellious RD. But try to focus on the bigger picture and take small steps.
First, make sure you eat something, Ms. Margulies advised. “Skipping meals can lead to overeating later and negatively impact energy levels and concentration.”
Lisa Andrews, MEd, RD, LD, owner of Sound Bites Nutrition, recalled one of her patients, a gastrointestinal surgeon with reactive hypoglycemia and fatigue. “She was experiencing energy crashes mid-afternoon,” she said. It was only after starting to eat every 4-5 hours that her patient felt better.
Of course, this is easier said than done. “When you are running from one patient to the other and trying to keep on time with your schedule, there is very little time for eating and no time at all for cooking or even heating up food,” recalled Hélène Bertrand, MD, author of Low Back Pain: 3 Steps to Relief in 2 Minutes.
But during her 55 years as a family medicine physician, Dr. Bertrand found ways to improve (if not perfect) the situation. She lunched on nuts or seeds during the day or grabbed a 95% cacao chocolate bar — higher in antioxidants and lower in sugar than a candy bar.
If you don’t have time for breakfast, try drinking a complete protein shake while driving to work, Dr. Toomer recommended. “It’s not ideal, but it’s better than nothing.” Similarly, if the only way you’ll eat a high-protein, lower-carb snack like hummus is with potato chips, go for it, she said.
Basically, don’t be type A striving for perfection. Take good enough when you can and balance the rest when you have time.
Torpedo Temptation
From free treats in the break room to always-present pizza for residents, high-fat, high-sugar, low-nutrient fare is a constant temptation. “I worked with a physician who would bring a balanced lunch to work every day, then find whatever sweet was around for his afternoon treat,” recalled Ms. Margulies.“The cookies, cakes, and donuts were starting to add up — and stopping at one wasn’t working for him.”
What did work was Ms. Margulies’ suggestion to bring a single serving of dark chocolate and fruit to savor during a longer break. “Bringing your favorite treats in appropriate portions can help you stick with your plan throughout the day,” she explained, and you’ll have an easier time resisting what’s in the break room. “When you desire a treat, tell yourself you have what you need and don’t need to indulge in the ‘free food’ just because it’s there. You have power over your choices.”
How about tricking yourself into perceiving cherry tomatoes as treats? That might be unusual, but one of Dr. La Puma’s physician patients did just that, displaying the produce in a candy dish on his office counter. Not only did this strategy help remind him to snack healthfully, it also prompted his patients to ask about eating better, he said.
Preparation Is Still Underrated
Many people find meal prepping intimidating. But it doesn’t need to be complicated. For instance, try purchasing precut veggies, cooked chicken breasts, or other healthy convenience options. You can then combine them in packable containers to prep a few meals at a time. For less busy weeks, consider cooking the protein yourself and whipping up basic sauces (like pesto and vinaigrette) to jazz up your meals.
“I worked with a resident who was gaining weight each month,” recalled Ms. Margulies. “She would skip lunch, grab a random snack, then wait until she got home to eat anything she could find.”
Encouraged by Ms. Margulies, she prepared and portioned one or two balanced dinners each week, which she’d later reheat. She also bought fresh and dried fruit and high-protein snacks, keeping single servings in her car to eat on the way home.
Similarly, Jess DeGore, RD, LDN, CDCES, CHWC, a diabetes educator and owner of Dietitian Jess Nutrition, recalled an ob.gyn. client who constantly skipped meals and relied on vending machine snacks. To combat her resulting energy crashes, she followed Ms. DeGore’s advice to prep workday lunches (like quinoa salads) over the weekend and bring fruit and nut snacks to work.
Automate as Much as You Can
If healthy is already on hand, you’ll eat healthy, said Ms. Andrews. Build up a snack stash focusing on fiber and protein. Tote a lunch bag with a cooler pack if needed. Some suggestions:
- Oatmeal packets
- Individual Greek yogurt cups or drinkable yogurts
- Protein bars
- Protein shakes
- Fresh fruit
- Fresh veggie sticks
- Nuts, dried chickpeas, or edamame
- Trail mix
- Single servings of hummus, nut butter, or guacamole
- Dried seaweed snacks
- Whole grain crackers
- Hard-boiled eggs
- String cheese
- Peanut butter sandwich
- 95% cacao chocolate bar
Try a Meal Delivery Service
Meal delivery services can be pricey, but potentially worth the expense. By bringing meals or having them sent to your office, you won’t have to find time to go to the cafeteria and stand in line, noted Janese S. Laster, MD, an internal medicine, gastroenterology, obesity medicine, and nutrition physician and founder of Gut Theory Total Digestive Care. Instead, “you’ll have something to warm up and eat while writing notes or in between patients,” she said. Plus, “you won’t have an excuse to skip meals.”
Hydration Yes, Junk Drinks No
The following can be filed in the Doctors-Know-It-But-Don’t-Always-Do-It section: “Hunger can be mistaken for thirst,” said Ms. Margulies. “Staying hydrated will help you better assess whether you’re hungry or thirsty.” Choose water over soda or energy drinks, she added, to hydrate your body without unnecessary extra sugars, sugar substitutes, calories, caffeine, or sodium — all of which can affect how you feel.
Advocate for Your Health
Convincing your institution to make changes might be difficult or even impossible, but consider asking your workplace to implement initiatives like these to boost provider nutrition, suggested Jabe Brown, BHSc (Nat), founder of Melbourne Functional Medicine:
- Establish protected break times when doctors can step away from their duties to eat
- Add more nutritious cafeteria options, like salads, whole grains, and lean proteins
- Overhaul vending machine offerings
- Offer educational workshops on nutrition
Be Tenacious About Good Eating
For Dr. Toomer, that meant taking several years off from work to improve her health. After losing more than 100 pounds, she founded TOTAL Weight Care Institute to help other healthcare professionals follow in her footsteps.
For you, the path toward a healthier diet might be gradual — grabbing a more nutritious snack, spending an extra hour per week on food shopping or prep, remembering a water bottle. Whatever it looks like, make realistic lifestyle tweaks that work for you.
Maybe even try that apple-a-day thing.
A version of this article first appeared on Medscape.com.
Eat as fast as you can whenever you can.
That was the med student mindset around food, as Catherine Harmon Toomer, MD, discovered during her school years. “Without a good system in place to counter that,” she explains, “unhealthy eating can get out of control, and that’s what happened to me.”
After med school, things got worse for Dr. Toomer. By her second year in practice as a family medicine physician, she’d gained a lot of weight and had been diagnosed with type 2 diabetes and cardiomyopathy. At 36, she went into congestive heart failure and was told she likely had 5 years to live.
A moment she described as “a huge wake-up call.”
Dr. Toomer is far from alone in her struggles to balance working in medicine and eating healthfully.
internist and cofounder of ChefMD and founder of Chef Clinic.
There is also the culture of medicine, which Dr. Toomer said looks down on self-care. “Even with break times, patient needs come before our own.” So, you sit down to eat, and there’s an emergency. Your clinic closes for lunch, but the phones still ring, and patients continue to email questions. Charting is also so time-consuming that “everything else gets put on the back burner.”
Sticking to a nutritious diet in this context can feel hopeless. But it isn’t. Really. Here are some doctor-tested, real-life ways you can nourish yourself while getting it all done.
Something Is Always Better Than Nothing
Sure, you might not be able to eat a balanced lunch or dinner while at work, conceded Amy Margulies, RD, LDN, owner of The Rebellious RD. But try to focus on the bigger picture and take small steps.
First, make sure you eat something, Ms. Margulies advised. “Skipping meals can lead to overeating later and negatively impact energy levels and concentration.”
Lisa Andrews, MEd, RD, LD, owner of Sound Bites Nutrition, recalled one of her patients, a gastrointestinal surgeon with reactive hypoglycemia and fatigue. “She was experiencing energy crashes mid-afternoon,” she said. It was only after starting to eat every 4-5 hours that her patient felt better.
Of course, this is easier said than done. “When you are running from one patient to the other and trying to keep on time with your schedule, there is very little time for eating and no time at all for cooking or even heating up food,” recalled Hélène Bertrand, MD, author of Low Back Pain: 3 Steps to Relief in 2 Minutes.
But during her 55 years as a family medicine physician, Dr. Bertrand found ways to improve (if not perfect) the situation. She lunched on nuts or seeds during the day or grabbed a 95% cacao chocolate bar — higher in antioxidants and lower in sugar than a candy bar.
If you don’t have time for breakfast, try drinking a complete protein shake while driving to work, Dr. Toomer recommended. “It’s not ideal, but it’s better than nothing.” Similarly, if the only way you’ll eat a high-protein, lower-carb snack like hummus is with potato chips, go for it, she said.
Basically, don’t be type A striving for perfection. Take good enough when you can and balance the rest when you have time.
Torpedo Temptation
From free treats in the break room to always-present pizza for residents, high-fat, high-sugar, low-nutrient fare is a constant temptation. “I worked with a physician who would bring a balanced lunch to work every day, then find whatever sweet was around for his afternoon treat,” recalled Ms. Margulies.“The cookies, cakes, and donuts were starting to add up — and stopping at one wasn’t working for him.”
What did work was Ms. Margulies’ suggestion to bring a single serving of dark chocolate and fruit to savor during a longer break. “Bringing your favorite treats in appropriate portions can help you stick with your plan throughout the day,” she explained, and you’ll have an easier time resisting what’s in the break room. “When you desire a treat, tell yourself you have what you need and don’t need to indulge in the ‘free food’ just because it’s there. You have power over your choices.”
How about tricking yourself into perceiving cherry tomatoes as treats? That might be unusual, but one of Dr. La Puma’s physician patients did just that, displaying the produce in a candy dish on his office counter. Not only did this strategy help remind him to snack healthfully, it also prompted his patients to ask about eating better, he said.
Preparation Is Still Underrated
Many people find meal prepping intimidating. But it doesn’t need to be complicated. For instance, try purchasing precut veggies, cooked chicken breasts, or other healthy convenience options. You can then combine them in packable containers to prep a few meals at a time. For less busy weeks, consider cooking the protein yourself and whipping up basic sauces (like pesto and vinaigrette) to jazz up your meals.
“I worked with a resident who was gaining weight each month,” recalled Ms. Margulies. “She would skip lunch, grab a random snack, then wait until she got home to eat anything she could find.”
Encouraged by Ms. Margulies, she prepared and portioned one or two balanced dinners each week, which she’d later reheat. She also bought fresh and dried fruit and high-protein snacks, keeping single servings in her car to eat on the way home.
Similarly, Jess DeGore, RD, LDN, CDCES, CHWC, a diabetes educator and owner of Dietitian Jess Nutrition, recalled an ob.gyn. client who constantly skipped meals and relied on vending machine snacks. To combat her resulting energy crashes, she followed Ms. DeGore’s advice to prep workday lunches (like quinoa salads) over the weekend and bring fruit and nut snacks to work.
Automate as Much as You Can
If healthy is already on hand, you’ll eat healthy, said Ms. Andrews. Build up a snack stash focusing on fiber and protein. Tote a lunch bag with a cooler pack if needed. Some suggestions:
- Oatmeal packets
- Individual Greek yogurt cups or drinkable yogurts
- Protein bars
- Protein shakes
- Fresh fruit
- Fresh veggie sticks
- Nuts, dried chickpeas, or edamame
- Trail mix
- Single servings of hummus, nut butter, or guacamole
- Dried seaweed snacks
- Whole grain crackers
- Hard-boiled eggs
- String cheese
- Peanut butter sandwich
- 95% cacao chocolate bar
Try a Meal Delivery Service
Meal delivery services can be pricey, but potentially worth the expense. By bringing meals or having them sent to your office, you won’t have to find time to go to the cafeteria and stand in line, noted Janese S. Laster, MD, an internal medicine, gastroenterology, obesity medicine, and nutrition physician and founder of Gut Theory Total Digestive Care. Instead, “you’ll have something to warm up and eat while writing notes or in between patients,” she said. Plus, “you won’t have an excuse to skip meals.”
Hydration Yes, Junk Drinks No
The following can be filed in the Doctors-Know-It-But-Don’t-Always-Do-It section: “Hunger can be mistaken for thirst,” said Ms. Margulies. “Staying hydrated will help you better assess whether you’re hungry or thirsty.” Choose water over soda or energy drinks, she added, to hydrate your body without unnecessary extra sugars, sugar substitutes, calories, caffeine, or sodium — all of which can affect how you feel.
Advocate for Your Health
Convincing your institution to make changes might be difficult or even impossible, but consider asking your workplace to implement initiatives like these to boost provider nutrition, suggested Jabe Brown, BHSc (Nat), founder of Melbourne Functional Medicine:
- Establish protected break times when doctors can step away from their duties to eat
- Add more nutritious cafeteria options, like salads, whole grains, and lean proteins
- Overhaul vending machine offerings
- Offer educational workshops on nutrition
Be Tenacious About Good Eating
For Dr. Toomer, that meant taking several years off from work to improve her health. After losing more than 100 pounds, she founded TOTAL Weight Care Institute to help other healthcare professionals follow in her footsteps.
For you, the path toward a healthier diet might be gradual — grabbing a more nutritious snack, spending an extra hour per week on food shopping or prep, remembering a water bottle. Whatever it looks like, make realistic lifestyle tweaks that work for you.
Maybe even try that apple-a-day thing.
A version of this article first appeared on Medscape.com.