User login
UPDATE: MENOPAUSE
Much has changed in the management of menopausal women. The Women’s Health Initiative (WHI) and other trials shed light on the risk-benefit ratio of hormone therapy (HT) and significantly altered patterns of usage. A new fracture risk-assessment tool devised by the World Health Organization is now available for widespread use; it continues to be refined so that it can be applied to specific populations with greater accuracy. And the management of low bone mass and osteoporosis has evolved so that we can determine with greater precision exactly who merits our attention.
This year, the Update on Menopause describes:
- a reanalysis of WHI data, focusing on the relationship between hormone therapy and the risk of coronary artery disease (CAD)
- a study from Finland that explores the risk of endometrial cancer associated with various progestin regimens in women who are taking estrogen and who have an intact uterus
- guidance from the North American Menopause Society and the National Osteoporosis Foundation on who, how, and when to evaluate for a likelihood of fracture
- insight into the benefits of and need for vitamin D among menopausal women
- information on a new selective estrogen-receptor modulator under development.
Hormone therapy and CAD: Is the glass half full…or half empty?
Toh S, Hernández-Díaz S, Logan R, Rossouw JE, Hernán MA. Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear? A randomized trial. Ann Intern Med. 2010;152(4):211–217.
North American Menopause Society. Position Statement: Estrogen and progestogens use in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(2):242–255. DOI: 10.1097/gme.0b013e3181d0f6b9. http://www.menopause.org/PSht10.pdf. Accessed April 1, 2010.
When estrogen therapy is initiated within 10 years of menopause, it may reduce the risk of CAD, according to data from the WHI randomized trial and observational data.
The picture isn’t as clear in regard to estrogen-progestin HT. In a just-published study, Toh and colleagues reassessed data from the WHI trial of continuous oral conjugated equine estrogen plus medroxyprogesterone acetate versus placebo. They also compared the WHI findings with those of the large observational Nurses’ Health Study (NHS). Here are some of their findings:
- participants became less consistent in taking study medication over time—a finding of many long-term studies. This trend prompted Toh and colleagues to adjust their analysis for adherence
- among women who used HT within 10 years after the menopausal transition, the hazard ratio (HR) for CAD was 0.64 (95% confidence interval [CI], 0.21, 1.99) in the WHI and 0.68 (95% CI, 0.24, 1.91) in the NHS. Both hazard ratios suggest that the risk of CAD is lower in Ht users than in nonusers—although the difference is not significant
- when investigators pooled the WHI and NHS findings, the hazard ratio for CAD associated with combination HT was 0.66 (95% CI, 0.31, 1.42). Note that, as the number of participants increases, the confidence limits narrow.
Toh and colleagues concluded that their analysis demonstrated no diminished risk of CAD with HT use. My reading of these data is different: Combination HT does not increase the risk of CAD in women who have been postmenopausal for less than 10 years.
Focus on risk was unbalanced
The question of whether combination HT reduces the risk of CAD in younger women is somewhat moot. I am not aware of any ObGyn in the United States who uses HT to prevent CAD, and the great majority of symptomatic women who consider initiating HT have been menopausal for less than a decade. For these reasons, I find the conclusions drawn by Toh and colleagues a bit mystifying—and the title they chose for their study may be misleading:
Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear?
Nevertheless, fear that HT might increase the risk of CAD is common among symptomatic menopausal women and their physicians. What this important analysis can offer is reassurance to symptomatic women who have been menopausal for less than 10 years: namely, that HT—estrogen alone or estrogen plus progestin—does not increase the risk of myocardial infarction or death from CAD.
You should counsel symptomatic women who have been menopausal less than 10 years that short-term use of estrogen therapy or estrogen-progestin therapy does not appear to increase their risk of CAD.
In women taking estrogen, sequential progestin therapy raises the risk of endometrial cancer
Jaakkola S, Lyytinen H, Pukkala E, Ylikorkala O. Endometrial cancer in postmenopausal women using estradiol-progestin therapy. Obstet Gynecol. 2009;114(6):1197–1204.
Because unopposed estrogen raises the risk of endometrial hyperplasia and adenocarcinoma, we prescribe progestational therapy when a menopausal woman who has an intact uterus decides to use estrogen.
Some ObGyns prescribe continuous progestin in this setting; others, sequential progestin (e.g., continuous estrogen along with 14 days of progestin each month). Still others prescribe “long-cycle” sequential regimens comprising continuous estrogen plus 2 weeks of progestin every 3 months. However, the long-term endometrial safety of these regimens has not been adequately explored.
In a national cohort study in Finland, Jaakkola and coworkers followed more than 200,000 menopausal women who used estrogen plus progestin (E+P) HT from 1994 to 2006. The incidence of endometrial cancer in these women was compared with that of all women in Finland. Overall, 1,400 newly diagnosed cases of endometrial cancer were identified.
Investigators found that, compared with the general population:
- E+P users overall had a 54% greater risk of endometrioid endometrial adenocarcinoma, the tumor most closely associated with use of unopposed estrogen
- monthly sequential E+P users had a 69% (5 years’ use) and 156% (10 years’ use) greater risk of this tumor
- at 5 and 10 years of use, long-cycle sequential HT was associated with a substantially higher risk of endometrial adenocarcinoma than was monthly sequential HT
- the endometrial safety profiles of transdermal and oral sequential HT were comparable; so were those of medroxyprogesterone acetate and norethindrone acetate
- in contrast to sequential HT regimens, continuous E+P was associated with a lower risk of endometrial cancer than that found in the overall population (76% risk reduction after 3 to 5 years of use).
These important findings clarify the safety profiles of long-term continuous E+P, which is protective, and sequential E+P, which substantially raises the risk of endometrial adenocarcinoma.
The authors estimate that, for every 1,000 women who use E+P for 10 years, eight additional cases of cancer will be diagnosed if that therapy is sequential and monthly; on the other hand, three or four fewer cases will be diagnosed if HT is continuous.
Women who use E+P hormone therapy should take the progestin continuously rather than sequentially.
Any woman who uses sequential progestin over the long term should undergo regular endometrial monitoring via transvaginal ultrasonography, endometrial biopsy, or both.
Guidance on who to assess for osteoporosis—and when to treat them
The North American Menopause Society. NAMS continuing medical education activity: Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(1):23–24.
National Osteoporosis Foundation. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Washington, D.C.: NOF: 2008. http://www.nof.org/professionals/NOF_Clinicians_Guide.pdf. Accessed April 1, 2010.
Kaunitz AM, McClung MR, Feldman RG, Wysocki S. Postmenopausal osteoporosis: fracture risk and prevention. OBG Management. 2009;21(10)(suppl):S1–6.
The North American Menopause Society (NAMS) has updated its position statement on the management of osteoporosis in postmenopausal women, incorporating many recommendations from guidelines issued in 2008 by the National Osteoporosis Foundation. You may find the recommendations on bone mineral density (BMD) assessment and use of medications to prevent fractures of particular interest.
BMD assessment should focus on women at high risk of fracture
In the United States, many women at low risk of fracture begin BMD assessment in their 50s via dual-energy x-ray absorptiometry (DXA) imaging of the lumbar spine and hip (FIGURE). As a result, many of these menopausal women are given a diagnosis of low bone mass (often termed “osteopenia” when in fact their DXA findings do not meet the criteria for osteoporosis) and are given a prescription for a bisphosphonate, often of indefinite duration.
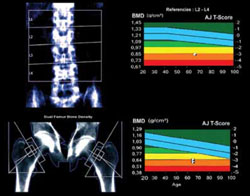
FIGURE When DXA imaging reveals low BMD
DXA scan of the lumbar spine and both hips showing osteoporosis in a 69-year-old woman. The colored graphs show the range of bone mineral density (BMD) across eight decades. The blue bars represent normal bone density; the white squares within the orange and red bars denote this patient’s low BMD. NAMS’s latest position statement clarifies exactly which women should be assessed, concluding that BMD measurement is appropriate in postmenopausal women who are not only at least 50 years old, but who also have one or more of the following risk factors for fracture:
- a history of fracture after menopause
- body mass index (BMI) <21 kg/m2 or weight <127 lb
- parental history of hip fracture
- current smoking, rheumatoid arthritis, or excessive alcohol intake (i.e., three or more drinks per day).
In the absence of these risk factors, BMD assessment should begin at 65 years of age.
Once initiated, treatment is usually long-term
The NAMS statement indicates that drug therapy—using bisphosphonates as first-line agents—is appropriate in postmenopausal women who have any of the following:
- a history of osteoporotic hip or vertebral fracture
- DXA-defined T-score lower than –2.5, indicating osteoporosis
- T-score from –1.0 to –2.5 (low bone mass) plus either a FRAX score that indicates a 10-year risk of hip fracture of at least 3% or a 10-year overall risk of osteoporotic fracture of at least 20%.
The World Health Organization (WHO) Fracture Risk Algorithm (FRAX) was developed to calculate the 10-year risk of hip fracture and the 10-year overall risk of osteoporotic fracture (online at http://www.shef.ac.uk/FRAX/). (For a description of two menopausal cases in which this Web site was used to determine appropriate clinical management, see the article by Kaunitz and colleagues cited above. Also, be aware that use of the FRAX Web site is inappropriate for making clinical decisions about women who are already using prescription antifracture therapy.)
NAMS points out that 1) treatment should usually span a period of years and 2) the risk of fracture after discontinuation of treatment has not been adequately studied.
Lasofoxifene seems unlikely to offer net benefits greater than what women obtain from existing therapies
Clinicians and menopausal women would welcome any agent that can prevent osteoporotic fracture with minimal adverse outcomes. Lasofoxifene appears to fulfill the first half of this equation, but its potential risks and questionable long-term effects cast doubt on its overall utility.
In a manufacturer-sponsored international trial, investigators randomized 8,556 women (mean age, 67 years) who met BMD criteria for osteoporosis to the selective estrogen-receptor modulator (SERM) lasofoxifene (0.25 mg daily or 0.5 mg daily) or placebo for 5 years. Women who received 0.5 mg daily of lasofoxifene had a substantially lower risk of vertebral (HR, 0.58) and nonvertebral (HR, 0.76) fracture than did women who received placebo.1
This dosage of lasofoxifene was also associated with a lower risk of estrogen-receptor–positive breast cancer (HR, 0.19), CAD events (HR, 0.68), and stroke (HR, 0.64), but a twofold higher risk of venous thromboembolic events overall and more than fourfold higher risk of pulmonary embolism. The incidence of endometrial cancer and endometrial hyperplasia was low (fewer than three women in each group), but endometrial polyps and hypertrophy were substantially more common among women who received either dosage of lasofoxifene.1
At 3 years, questionable benefit
Although these findings indicate that lasofoxifene lowers the risk of radiologically confirmed vertebral fracture, data submitted to the FDA reveal that the risk of clinical vertebral fracture was not reduced at 3 years.
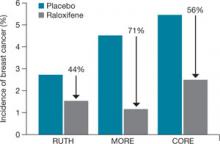
Both raloxifene and lasofoxifene are associated with a heightened risk of venous thromboembolic events. Although lasofoxifene did not raise the risk of endometrial neoplasia or hyperplasia in the trial just described, the rates of other endometrial outcomes suggest that this agent has a proliferative effect on the endometrium.1
The reduced risk of estrogen-receptor–positive breast cancer and CAD events is intriguing. However, as an editorial writer points out, a clinician would need to treat 492 women for 1 year to prevent one major CAD event.2
Lasofoxifene does not seem to offer any clinically important benefit over existing SERMs. Moreover, alendronate, a generic bisphosphonate proven to prevent clinical vertebral and nonvertebral fracture, often fills the bill for the prevention of osteoporotic fracture in menopausal women.
References
1. Cummings SR, Ensrud K, Delmas PF, et al. Lasofoxifene in postmenopausal women with osteoporosis. N Engl J Med. 2010;362(8):686-696.
2. Becker C. Another selective estrogen-receptor modulator for osteoporosis. N Engl J Med. 2010;362(8):752-754.
BMD reassessment should be minimal
BMD assessment by means of DXA imaging to monitor the effects of therapy is appropriate after 1 or 2 years of treatment. Thereafter, repeat measurement is of little value in women whose BMD has stabilized or increased on therapy. A follow-up DXA scan is of limited use in predicting the effectiveness of antiresorptive therapy in lowering the risk of fracture. Moreover, changes in BMD can lag behind actual therapeutic benefits (i.e., fracture prevention).
In menopausal women who are not taking prescription antifracture therapy, the follow-up measurement of BMD is not useful until 2 to 5 years after initial testing. Although BMD may be lost rapidly in the initial years after menopause (or after discontinuation of HT), subsequently it plateaus or declines slowly.
If a woman is not using prescription antifracture therapy and is within 2 or 3 years of menopause (or if she has discontinued menopausal HT in the past 2 to 3 years), retesting in 2 years is prudent. However, if the same woman were 5 or more years post-menopausal and had not recently discontinued HT, follow-up BMD assessment can be deferred for 3 to 5 years.
This updated guidance from NAMS emphasizes that BMD need not be assessed in women younger than 65 years unless they have specific risk factors. FRAX evaluation also makes it possible to estimate the 10-year risk of fracture in women who have low bone mass but who do not meet criteria for osteoporosis. FRAX evaluation indicates that prescription therapy is rarely required for women in their 50s or 60s who have low bone mass (but not osteoporosis). For women in their 70s or 80s who have low bone mass (but not osteoporosis), however, FRAX evaluation often leads to a recommendation to initiate prescription antifracture treatment.
Many postmenopausal women who are in their 50s or 60s and who have T-scores of –1.0 to –2.5 are given bisphosphonates, despite being at low risk of fracture. Adherence to the NAMS guidelines will help prevent unnecessary assessment and treatment. In particular, the NAMS recommendations for follow-up BMD assessment—i.e., one-time evaluation 1 to 2 years after initiating therapy and no further assessment in women found to have stable BMD at the first follow-up DXA—should simplify clinical management in this setting.
Hold off on ordering DXA testing until women meet criteria for BMD assessment. In women who do not have osteoporosis, limit use of bisphosphonates to those who have an elevated 10-year risk of fracture, as assessed using the FRAX tool.
Our menopausal patients should be taking more vitamin D supplements
Bischoff-Ferrari H. Vitamin D: what is an adequate vitamin D level and how much supplementation is necessary? Best Pract Res Clin Rheumatol. 2009;23(6):789.
Bischoff-Ferrari HA, Dawson-Hughes B, Staehelin HB, et al. Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ. 2009;339:b3692.
Stewart JW, Alekel DL, Ritland LM, et al. Serum 25-hydroxyvitamin D is related to indicators of overall physical fitness in healthy postmenopausal women. Menopause. 2009;16(6):1093–1101.
Office of Dietary Supplements. Dietary Supplement Fact Sheet: Vitamin D. National Institutes of Health Web site. http://dietary-supplements.info.nih.gov/factsheets/vitamind.asp. Updated November 13, 2009. Accessed April 2, 2010.
We have long recognized the important role vitamin D plays in promoting calcium absorption from the gut and maintaining adequate serum calcium and phosphate concentrations to enable normal bone mineralization. Now, studies reveal that the vitamin also helps prevent falls and promotes overall fitness in menopausal women. It has also become clear that traditional targets for vitamin D supplementation are inadequate.
Two recent meta-analyses of double-blind, randomized trials concluded that vitamin D reduces the risk of falls in a dose-dependent manner. Dr. Heike Bischoff-Ferrari, a Swiss scientist and a leading vitamin D researcher, points out that 1) a minimum of 700 to 1,000 IU of vitamin D supplementation daily is appropriate in menopausal women and 2) a higher amount is indicated for those who are obese or deficient in vitamin D.
Compare the current recommended dietary allowance (RDA) for adults 51 to 70 years old: 400 IU daily. The federal Food and Nutrition Board is expected to update the vitamin D RDA this spring. The 2010 NAMS statement on osteoporosis recommends a daily vitamin D intake of 800 to 1,000 IU for menopausal women.
Ask your patient to add up the aggregate daily amount of vitamin D she ingests with her multivitamin and calcium and vitamin D supplements. If it is less than 800 IU, have her purchase over-the-counter vitamin D supplements (available in 400, 1,000, and 2,000 IU capsules). Obese patients and those known to be deficient may need to ingest higher daily amounts of vitamin D.
Much has changed in the management of menopausal women. The Women’s Health Initiative (WHI) and other trials shed light on the risk-benefit ratio of hormone therapy (HT) and significantly altered patterns of usage. A new fracture risk-assessment tool devised by the World Health Organization is now available for widespread use; it continues to be refined so that it can be applied to specific populations with greater accuracy. And the management of low bone mass and osteoporosis has evolved so that we can determine with greater precision exactly who merits our attention.
This year, the Update on Menopause describes:
- a reanalysis of WHI data, focusing on the relationship between hormone therapy and the risk of coronary artery disease (CAD)
- a study from Finland that explores the risk of endometrial cancer associated with various progestin regimens in women who are taking estrogen and who have an intact uterus
- guidance from the North American Menopause Society and the National Osteoporosis Foundation on who, how, and when to evaluate for a likelihood of fracture
- insight into the benefits of and need for vitamin D among menopausal women
- information on a new selective estrogen-receptor modulator under development.
Hormone therapy and CAD: Is the glass half full…or half empty?
Toh S, Hernández-Díaz S, Logan R, Rossouw JE, Hernán MA. Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear? A randomized trial. Ann Intern Med. 2010;152(4):211–217.
North American Menopause Society. Position Statement: Estrogen and progestogens use in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(2):242–255. DOI: 10.1097/gme.0b013e3181d0f6b9. http://www.menopause.org/PSht10.pdf. Accessed April 1, 2010.
When estrogen therapy is initiated within 10 years of menopause, it may reduce the risk of CAD, according to data from the WHI randomized trial and observational data.
The picture isn’t as clear in regard to estrogen-progestin HT. In a just-published study, Toh and colleagues reassessed data from the WHI trial of continuous oral conjugated equine estrogen plus medroxyprogesterone acetate versus placebo. They also compared the WHI findings with those of the large observational Nurses’ Health Study (NHS). Here are some of their findings:
- participants became less consistent in taking study medication over time—a finding of many long-term studies. This trend prompted Toh and colleagues to adjust their analysis for adherence
- among women who used HT within 10 years after the menopausal transition, the hazard ratio (HR) for CAD was 0.64 (95% confidence interval [CI], 0.21, 1.99) in the WHI and 0.68 (95% CI, 0.24, 1.91) in the NHS. Both hazard ratios suggest that the risk of CAD is lower in Ht users than in nonusers—although the difference is not significant
- when investigators pooled the WHI and NHS findings, the hazard ratio for CAD associated with combination HT was 0.66 (95% CI, 0.31, 1.42). Note that, as the number of participants increases, the confidence limits narrow.
Toh and colleagues concluded that their analysis demonstrated no diminished risk of CAD with HT use. My reading of these data is different: Combination HT does not increase the risk of CAD in women who have been postmenopausal for less than 10 years.
Focus on risk was unbalanced
The question of whether combination HT reduces the risk of CAD in younger women is somewhat moot. I am not aware of any ObGyn in the United States who uses HT to prevent CAD, and the great majority of symptomatic women who consider initiating HT have been menopausal for less than a decade. For these reasons, I find the conclusions drawn by Toh and colleagues a bit mystifying—and the title they chose for their study may be misleading:
Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear?
Nevertheless, fear that HT might increase the risk of CAD is common among symptomatic menopausal women and their physicians. What this important analysis can offer is reassurance to symptomatic women who have been menopausal for less than 10 years: namely, that HT—estrogen alone or estrogen plus progestin—does not increase the risk of myocardial infarction or death from CAD.
You should counsel symptomatic women who have been menopausal less than 10 years that short-term use of estrogen therapy or estrogen-progestin therapy does not appear to increase their risk of CAD.
In women taking estrogen, sequential progestin therapy raises the risk of endometrial cancer
Jaakkola S, Lyytinen H, Pukkala E, Ylikorkala O. Endometrial cancer in postmenopausal women using estradiol-progestin therapy. Obstet Gynecol. 2009;114(6):1197–1204.
Because unopposed estrogen raises the risk of endometrial hyperplasia and adenocarcinoma, we prescribe progestational therapy when a menopausal woman who has an intact uterus decides to use estrogen.
Some ObGyns prescribe continuous progestin in this setting; others, sequential progestin (e.g., continuous estrogen along with 14 days of progestin each month). Still others prescribe “long-cycle” sequential regimens comprising continuous estrogen plus 2 weeks of progestin every 3 months. However, the long-term endometrial safety of these regimens has not been adequately explored.
In a national cohort study in Finland, Jaakkola and coworkers followed more than 200,000 menopausal women who used estrogen plus progestin (E+P) HT from 1994 to 2006. The incidence of endometrial cancer in these women was compared with that of all women in Finland. Overall, 1,400 newly diagnosed cases of endometrial cancer were identified.
Investigators found that, compared with the general population:
- E+P users overall had a 54% greater risk of endometrioid endometrial adenocarcinoma, the tumor most closely associated with use of unopposed estrogen
- monthly sequential E+P users had a 69% (5 years’ use) and 156% (10 years’ use) greater risk of this tumor
- at 5 and 10 years of use, long-cycle sequential HT was associated with a substantially higher risk of endometrial adenocarcinoma than was monthly sequential HT
- the endometrial safety profiles of transdermal and oral sequential HT were comparable; so were those of medroxyprogesterone acetate and norethindrone acetate
- in contrast to sequential HT regimens, continuous E+P was associated with a lower risk of endometrial cancer than that found in the overall population (76% risk reduction after 3 to 5 years of use).
These important findings clarify the safety profiles of long-term continuous E+P, which is protective, and sequential E+P, which substantially raises the risk of endometrial adenocarcinoma.
The authors estimate that, for every 1,000 women who use E+P for 10 years, eight additional cases of cancer will be diagnosed if that therapy is sequential and monthly; on the other hand, three or four fewer cases will be diagnosed if HT is continuous.
Women who use E+P hormone therapy should take the progestin continuously rather than sequentially.
Any woman who uses sequential progestin over the long term should undergo regular endometrial monitoring via transvaginal ultrasonography, endometrial biopsy, or both.
Guidance on who to assess for osteoporosis—and when to treat them
The North American Menopause Society. NAMS continuing medical education activity: Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(1):23–24.
National Osteoporosis Foundation. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Washington, D.C.: NOF: 2008. http://www.nof.org/professionals/NOF_Clinicians_Guide.pdf. Accessed April 1, 2010.
Kaunitz AM, McClung MR, Feldman RG, Wysocki S. Postmenopausal osteoporosis: fracture risk and prevention. OBG Management. 2009;21(10)(suppl):S1–6.
The North American Menopause Society (NAMS) has updated its position statement on the management of osteoporosis in postmenopausal women, incorporating many recommendations from guidelines issued in 2008 by the National Osteoporosis Foundation. You may find the recommendations on bone mineral density (BMD) assessment and use of medications to prevent fractures of particular interest.
BMD assessment should focus on women at high risk of fracture
In the United States, many women at low risk of fracture begin BMD assessment in their 50s via dual-energy x-ray absorptiometry (DXA) imaging of the lumbar spine and hip (FIGURE). As a result, many of these menopausal women are given a diagnosis of low bone mass (often termed “osteopenia” when in fact their DXA findings do not meet the criteria for osteoporosis) and are given a prescription for a bisphosphonate, often of indefinite duration.

FIGURE When DXA imaging reveals low BMD
DXA scan of the lumbar spine and both hips showing osteoporosis in a 69-year-old woman. The colored graphs show the range of bone mineral density (BMD) across eight decades. The blue bars represent normal bone density; the white squares within the orange and red bars denote this patient’s low BMD. NAMS’s latest position statement clarifies exactly which women should be assessed, concluding that BMD measurement is appropriate in postmenopausal women who are not only at least 50 years old, but who also have one or more of the following risk factors for fracture:
- a history of fracture after menopause
- body mass index (BMI) <21 kg/m2 or weight <127 lb
- parental history of hip fracture
- current smoking, rheumatoid arthritis, or excessive alcohol intake (i.e., three or more drinks per day).
In the absence of these risk factors, BMD assessment should begin at 65 years of age.
Once initiated, treatment is usually long-term
The NAMS statement indicates that drug therapy—using bisphosphonates as first-line agents—is appropriate in postmenopausal women who have any of the following:
- a history of osteoporotic hip or vertebral fracture
- DXA-defined T-score lower than –2.5, indicating osteoporosis
- T-score from –1.0 to –2.5 (low bone mass) plus either a FRAX score that indicates a 10-year risk of hip fracture of at least 3% or a 10-year overall risk of osteoporotic fracture of at least 20%.
The World Health Organization (WHO) Fracture Risk Algorithm (FRAX) was developed to calculate the 10-year risk of hip fracture and the 10-year overall risk of osteoporotic fracture (online at http://www.shef.ac.uk/FRAX/). (For a description of two menopausal cases in which this Web site was used to determine appropriate clinical management, see the article by Kaunitz and colleagues cited above. Also, be aware that use of the FRAX Web site is inappropriate for making clinical decisions about women who are already using prescription antifracture therapy.)
NAMS points out that 1) treatment should usually span a period of years and 2) the risk of fracture after discontinuation of treatment has not been adequately studied.
Lasofoxifene seems unlikely to offer net benefits greater than what women obtain from existing therapies
Clinicians and menopausal women would welcome any agent that can prevent osteoporotic fracture with minimal adverse outcomes. Lasofoxifene appears to fulfill the first half of this equation, but its potential risks and questionable long-term effects cast doubt on its overall utility.
In a manufacturer-sponsored international trial, investigators randomized 8,556 women (mean age, 67 years) who met BMD criteria for osteoporosis to the selective estrogen-receptor modulator (SERM) lasofoxifene (0.25 mg daily or 0.5 mg daily) or placebo for 5 years. Women who received 0.5 mg daily of lasofoxifene had a substantially lower risk of vertebral (HR, 0.58) and nonvertebral (HR, 0.76) fracture than did women who received placebo.1
This dosage of lasofoxifene was also associated with a lower risk of estrogen-receptor–positive breast cancer (HR, 0.19), CAD events (HR, 0.68), and stroke (HR, 0.64), but a twofold higher risk of venous thromboembolic events overall and more than fourfold higher risk of pulmonary embolism. The incidence of endometrial cancer and endometrial hyperplasia was low (fewer than three women in each group), but endometrial polyps and hypertrophy were substantially more common among women who received either dosage of lasofoxifene.1
At 3 years, questionable benefit
Although these findings indicate that lasofoxifene lowers the risk of radiologically confirmed vertebral fracture, data submitted to the FDA reveal that the risk of clinical vertebral fracture was not reduced at 3 years.
Both raloxifene and lasofoxifene are associated with a heightened risk of venous thromboembolic events. Although lasofoxifene did not raise the risk of endometrial neoplasia or hyperplasia in the trial just described, the rates of other endometrial outcomes suggest that this agent has a proliferative effect on the endometrium.1
The reduced risk of estrogen-receptor–positive breast cancer and CAD events is intriguing. However, as an editorial writer points out, a clinician would need to treat 492 women for 1 year to prevent one major CAD event.2
Lasofoxifene does not seem to offer any clinically important benefit over existing SERMs. Moreover, alendronate, a generic bisphosphonate proven to prevent clinical vertebral and nonvertebral fracture, often fills the bill for the prevention of osteoporotic fracture in menopausal women.
References
1. Cummings SR, Ensrud K, Delmas PF, et al. Lasofoxifene in postmenopausal women with osteoporosis. N Engl J Med. 2010;362(8):686-696.
2. Becker C. Another selective estrogen-receptor modulator for osteoporosis. N Engl J Med. 2010;362(8):752-754.
BMD reassessment should be minimal
BMD assessment by means of DXA imaging to monitor the effects of therapy is appropriate after 1 or 2 years of treatment. Thereafter, repeat measurement is of little value in women whose BMD has stabilized or increased on therapy. A follow-up DXA scan is of limited use in predicting the effectiveness of antiresorptive therapy in lowering the risk of fracture. Moreover, changes in BMD can lag behind actual therapeutic benefits (i.e., fracture prevention).
In menopausal women who are not taking prescription antifracture therapy, the follow-up measurement of BMD is not useful until 2 to 5 years after initial testing. Although BMD may be lost rapidly in the initial years after menopause (or after discontinuation of HT), subsequently it plateaus or declines slowly.
If a woman is not using prescription antifracture therapy and is within 2 or 3 years of menopause (or if she has discontinued menopausal HT in the past 2 to 3 years), retesting in 2 years is prudent. However, if the same woman were 5 or more years post-menopausal and had not recently discontinued HT, follow-up BMD assessment can be deferred for 3 to 5 years.
This updated guidance from NAMS emphasizes that BMD need not be assessed in women younger than 65 years unless they have specific risk factors. FRAX evaluation also makes it possible to estimate the 10-year risk of fracture in women who have low bone mass but who do not meet criteria for osteoporosis. FRAX evaluation indicates that prescription therapy is rarely required for women in their 50s or 60s who have low bone mass (but not osteoporosis). For women in their 70s or 80s who have low bone mass (but not osteoporosis), however, FRAX evaluation often leads to a recommendation to initiate prescription antifracture treatment.
Many postmenopausal women who are in their 50s or 60s and who have T-scores of –1.0 to –2.5 are given bisphosphonates, despite being at low risk of fracture. Adherence to the NAMS guidelines will help prevent unnecessary assessment and treatment. In particular, the NAMS recommendations for follow-up BMD assessment—i.e., one-time evaluation 1 to 2 years after initiating therapy and no further assessment in women found to have stable BMD at the first follow-up DXA—should simplify clinical management in this setting.
Hold off on ordering DXA testing until women meet criteria for BMD assessment. In women who do not have osteoporosis, limit use of bisphosphonates to those who have an elevated 10-year risk of fracture, as assessed using the FRAX tool.
Our menopausal patients should be taking more vitamin D supplements
Bischoff-Ferrari H. Vitamin D: what is an adequate vitamin D level and how much supplementation is necessary? Best Pract Res Clin Rheumatol. 2009;23(6):789.
Bischoff-Ferrari HA, Dawson-Hughes B, Staehelin HB, et al. Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ. 2009;339:b3692.
Stewart JW, Alekel DL, Ritland LM, et al. Serum 25-hydroxyvitamin D is related to indicators of overall physical fitness in healthy postmenopausal women. Menopause. 2009;16(6):1093–1101.
Office of Dietary Supplements. Dietary Supplement Fact Sheet: Vitamin D. National Institutes of Health Web site. http://dietary-supplements.info.nih.gov/factsheets/vitamind.asp. Updated November 13, 2009. Accessed April 2, 2010.
We have long recognized the important role vitamin D plays in promoting calcium absorption from the gut and maintaining adequate serum calcium and phosphate concentrations to enable normal bone mineralization. Now, studies reveal that the vitamin also helps prevent falls and promotes overall fitness in menopausal women. It has also become clear that traditional targets for vitamin D supplementation are inadequate.
Two recent meta-analyses of double-blind, randomized trials concluded that vitamin D reduces the risk of falls in a dose-dependent manner. Dr. Heike Bischoff-Ferrari, a Swiss scientist and a leading vitamin D researcher, points out that 1) a minimum of 700 to 1,000 IU of vitamin D supplementation daily is appropriate in menopausal women and 2) a higher amount is indicated for those who are obese or deficient in vitamin D.
Compare the current recommended dietary allowance (RDA) for adults 51 to 70 years old: 400 IU daily. The federal Food and Nutrition Board is expected to update the vitamin D RDA this spring. The 2010 NAMS statement on osteoporosis recommends a daily vitamin D intake of 800 to 1,000 IU for menopausal women.
Ask your patient to add up the aggregate daily amount of vitamin D she ingests with her multivitamin and calcium and vitamin D supplements. If it is less than 800 IU, have her purchase over-the-counter vitamin D supplements (available in 400, 1,000, and 2,000 IU capsules). Obese patients and those known to be deficient may need to ingest higher daily amounts of vitamin D.
Much has changed in the management of menopausal women. The Women’s Health Initiative (WHI) and other trials shed light on the risk-benefit ratio of hormone therapy (HT) and significantly altered patterns of usage. A new fracture risk-assessment tool devised by the World Health Organization is now available for widespread use; it continues to be refined so that it can be applied to specific populations with greater accuracy. And the management of low bone mass and osteoporosis has evolved so that we can determine with greater precision exactly who merits our attention.
This year, the Update on Menopause describes:
- a reanalysis of WHI data, focusing on the relationship between hormone therapy and the risk of coronary artery disease (CAD)
- a study from Finland that explores the risk of endometrial cancer associated with various progestin regimens in women who are taking estrogen and who have an intact uterus
- guidance from the North American Menopause Society and the National Osteoporosis Foundation on who, how, and when to evaluate for a likelihood of fracture
- insight into the benefits of and need for vitamin D among menopausal women
- information on a new selective estrogen-receptor modulator under development.
Hormone therapy and CAD: Is the glass half full…or half empty?
Toh S, Hernández-Díaz S, Logan R, Rossouw JE, Hernán MA. Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear? A randomized trial. Ann Intern Med. 2010;152(4):211–217.
North American Menopause Society. Position Statement: Estrogen and progestogens use in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(2):242–255. DOI: 10.1097/gme.0b013e3181d0f6b9. http://www.menopause.org/PSht10.pdf. Accessed April 1, 2010.
When estrogen therapy is initiated within 10 years of menopause, it may reduce the risk of CAD, according to data from the WHI randomized trial and observational data.
The picture isn’t as clear in regard to estrogen-progestin HT. In a just-published study, Toh and colleagues reassessed data from the WHI trial of continuous oral conjugated equine estrogen plus medroxyprogesterone acetate versus placebo. They also compared the WHI findings with those of the large observational Nurses’ Health Study (NHS). Here are some of their findings:
- participants became less consistent in taking study medication over time—a finding of many long-term studies. This trend prompted Toh and colleagues to adjust their analysis for adherence
- among women who used HT within 10 years after the menopausal transition, the hazard ratio (HR) for CAD was 0.64 (95% confidence interval [CI], 0.21, 1.99) in the WHI and 0.68 (95% CI, 0.24, 1.91) in the NHS. Both hazard ratios suggest that the risk of CAD is lower in Ht users than in nonusers—although the difference is not significant
- when investigators pooled the WHI and NHS findings, the hazard ratio for CAD associated with combination HT was 0.66 (95% CI, 0.31, 1.42). Note that, as the number of participants increases, the confidence limits narrow.
Toh and colleagues concluded that their analysis demonstrated no diminished risk of CAD with HT use. My reading of these data is different: Combination HT does not increase the risk of CAD in women who have been postmenopausal for less than 10 years.
Focus on risk was unbalanced
The question of whether combination HT reduces the risk of CAD in younger women is somewhat moot. I am not aware of any ObGyn in the United States who uses HT to prevent CAD, and the great majority of symptomatic women who consider initiating HT have been menopausal for less than a decade. For these reasons, I find the conclusions drawn by Toh and colleagues a bit mystifying—and the title they chose for their study may be misleading:
Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear?
Nevertheless, fear that HT might increase the risk of CAD is common among symptomatic menopausal women and their physicians. What this important analysis can offer is reassurance to symptomatic women who have been menopausal for less than 10 years: namely, that HT—estrogen alone or estrogen plus progestin—does not increase the risk of myocardial infarction or death from CAD.
You should counsel symptomatic women who have been menopausal less than 10 years that short-term use of estrogen therapy or estrogen-progestin therapy does not appear to increase their risk of CAD.
In women taking estrogen, sequential progestin therapy raises the risk of endometrial cancer
Jaakkola S, Lyytinen H, Pukkala E, Ylikorkala O. Endometrial cancer in postmenopausal women using estradiol-progestin therapy. Obstet Gynecol. 2009;114(6):1197–1204.
Because unopposed estrogen raises the risk of endometrial hyperplasia and adenocarcinoma, we prescribe progestational therapy when a menopausal woman who has an intact uterus decides to use estrogen.
Some ObGyns prescribe continuous progestin in this setting; others, sequential progestin (e.g., continuous estrogen along with 14 days of progestin each month). Still others prescribe “long-cycle” sequential regimens comprising continuous estrogen plus 2 weeks of progestin every 3 months. However, the long-term endometrial safety of these regimens has not been adequately explored.
In a national cohort study in Finland, Jaakkola and coworkers followed more than 200,000 menopausal women who used estrogen plus progestin (E+P) HT from 1994 to 2006. The incidence of endometrial cancer in these women was compared with that of all women in Finland. Overall, 1,400 newly diagnosed cases of endometrial cancer were identified.
Investigators found that, compared with the general population:
- E+P users overall had a 54% greater risk of endometrioid endometrial adenocarcinoma, the tumor most closely associated with use of unopposed estrogen
- monthly sequential E+P users had a 69% (5 years’ use) and 156% (10 years’ use) greater risk of this tumor
- at 5 and 10 years of use, long-cycle sequential HT was associated with a substantially higher risk of endometrial adenocarcinoma than was monthly sequential HT
- the endometrial safety profiles of transdermal and oral sequential HT were comparable; so were those of medroxyprogesterone acetate and norethindrone acetate
- in contrast to sequential HT regimens, continuous E+P was associated with a lower risk of endometrial cancer than that found in the overall population (76% risk reduction after 3 to 5 years of use).
These important findings clarify the safety profiles of long-term continuous E+P, which is protective, and sequential E+P, which substantially raises the risk of endometrial adenocarcinoma.
The authors estimate that, for every 1,000 women who use E+P for 10 years, eight additional cases of cancer will be diagnosed if that therapy is sequential and monthly; on the other hand, three or four fewer cases will be diagnosed if HT is continuous.
Women who use E+P hormone therapy should take the progestin continuously rather than sequentially.
Any woman who uses sequential progestin over the long term should undergo regular endometrial monitoring via transvaginal ultrasonography, endometrial biopsy, or both.
Guidance on who to assess for osteoporosis—and when to treat them
The North American Menopause Society. NAMS continuing medical education activity: Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(1):23–24.
National Osteoporosis Foundation. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Washington, D.C.: NOF: 2008. http://www.nof.org/professionals/NOF_Clinicians_Guide.pdf. Accessed April 1, 2010.
Kaunitz AM, McClung MR, Feldman RG, Wysocki S. Postmenopausal osteoporosis: fracture risk and prevention. OBG Management. 2009;21(10)(suppl):S1–6.
The North American Menopause Society (NAMS) has updated its position statement on the management of osteoporosis in postmenopausal women, incorporating many recommendations from guidelines issued in 2008 by the National Osteoporosis Foundation. You may find the recommendations on bone mineral density (BMD) assessment and use of medications to prevent fractures of particular interest.
BMD assessment should focus on women at high risk of fracture
In the United States, many women at low risk of fracture begin BMD assessment in their 50s via dual-energy x-ray absorptiometry (DXA) imaging of the lumbar spine and hip (FIGURE). As a result, many of these menopausal women are given a diagnosis of low bone mass (often termed “osteopenia” when in fact their DXA findings do not meet the criteria for osteoporosis) and are given a prescription for a bisphosphonate, often of indefinite duration.

FIGURE When DXA imaging reveals low BMD
DXA scan of the lumbar spine and both hips showing osteoporosis in a 69-year-old woman. The colored graphs show the range of bone mineral density (BMD) across eight decades. The blue bars represent normal bone density; the white squares within the orange and red bars denote this patient’s low BMD. NAMS’s latest position statement clarifies exactly which women should be assessed, concluding that BMD measurement is appropriate in postmenopausal women who are not only at least 50 years old, but who also have one or more of the following risk factors for fracture:
- a history of fracture after menopause
- body mass index (BMI) <21 kg/m2 or weight <127 lb
- parental history of hip fracture
- current smoking, rheumatoid arthritis, or excessive alcohol intake (i.e., three or more drinks per day).
In the absence of these risk factors, BMD assessment should begin at 65 years of age.
Once initiated, treatment is usually long-term
The NAMS statement indicates that drug therapy—using bisphosphonates as first-line agents—is appropriate in postmenopausal women who have any of the following:
- a history of osteoporotic hip or vertebral fracture
- DXA-defined T-score lower than –2.5, indicating osteoporosis
- T-score from –1.0 to –2.5 (low bone mass) plus either a FRAX score that indicates a 10-year risk of hip fracture of at least 3% or a 10-year overall risk of osteoporotic fracture of at least 20%.
The World Health Organization (WHO) Fracture Risk Algorithm (FRAX) was developed to calculate the 10-year risk of hip fracture and the 10-year overall risk of osteoporotic fracture (online at http://www.shef.ac.uk/FRAX/). (For a description of two menopausal cases in which this Web site was used to determine appropriate clinical management, see the article by Kaunitz and colleagues cited above. Also, be aware that use of the FRAX Web site is inappropriate for making clinical decisions about women who are already using prescription antifracture therapy.)
NAMS points out that 1) treatment should usually span a period of years and 2) the risk of fracture after discontinuation of treatment has not been adequately studied.
Lasofoxifene seems unlikely to offer net benefits greater than what women obtain from existing therapies
Clinicians and menopausal women would welcome any agent that can prevent osteoporotic fracture with minimal adverse outcomes. Lasofoxifene appears to fulfill the first half of this equation, but its potential risks and questionable long-term effects cast doubt on its overall utility.
In a manufacturer-sponsored international trial, investigators randomized 8,556 women (mean age, 67 years) who met BMD criteria for osteoporosis to the selective estrogen-receptor modulator (SERM) lasofoxifene (0.25 mg daily or 0.5 mg daily) or placebo for 5 years. Women who received 0.5 mg daily of lasofoxifene had a substantially lower risk of vertebral (HR, 0.58) and nonvertebral (HR, 0.76) fracture than did women who received placebo.1
This dosage of lasofoxifene was also associated with a lower risk of estrogen-receptor–positive breast cancer (HR, 0.19), CAD events (HR, 0.68), and stroke (HR, 0.64), but a twofold higher risk of venous thromboembolic events overall and more than fourfold higher risk of pulmonary embolism. The incidence of endometrial cancer and endometrial hyperplasia was low (fewer than three women in each group), but endometrial polyps and hypertrophy were substantially more common among women who received either dosage of lasofoxifene.1
At 3 years, questionable benefit
Although these findings indicate that lasofoxifene lowers the risk of radiologically confirmed vertebral fracture, data submitted to the FDA reveal that the risk of clinical vertebral fracture was not reduced at 3 years.
Both raloxifene and lasofoxifene are associated with a heightened risk of venous thromboembolic events. Although lasofoxifene did not raise the risk of endometrial neoplasia or hyperplasia in the trial just described, the rates of other endometrial outcomes suggest that this agent has a proliferative effect on the endometrium.1
The reduced risk of estrogen-receptor–positive breast cancer and CAD events is intriguing. However, as an editorial writer points out, a clinician would need to treat 492 women for 1 year to prevent one major CAD event.2
Lasofoxifene does not seem to offer any clinically important benefit over existing SERMs. Moreover, alendronate, a generic bisphosphonate proven to prevent clinical vertebral and nonvertebral fracture, often fills the bill for the prevention of osteoporotic fracture in menopausal women.
References
1. Cummings SR, Ensrud K, Delmas PF, et al. Lasofoxifene in postmenopausal women with osteoporosis. N Engl J Med. 2010;362(8):686-696.
2. Becker C. Another selective estrogen-receptor modulator for osteoporosis. N Engl J Med. 2010;362(8):752-754.
BMD reassessment should be minimal
BMD assessment by means of DXA imaging to monitor the effects of therapy is appropriate after 1 or 2 years of treatment. Thereafter, repeat measurement is of little value in women whose BMD has stabilized or increased on therapy. A follow-up DXA scan is of limited use in predicting the effectiveness of antiresorptive therapy in lowering the risk of fracture. Moreover, changes in BMD can lag behind actual therapeutic benefits (i.e., fracture prevention).
In menopausal women who are not taking prescription antifracture therapy, the follow-up measurement of BMD is not useful until 2 to 5 years after initial testing. Although BMD may be lost rapidly in the initial years after menopause (or after discontinuation of HT), subsequently it plateaus or declines slowly.
If a woman is not using prescription antifracture therapy and is within 2 or 3 years of menopause (or if she has discontinued menopausal HT in the past 2 to 3 years), retesting in 2 years is prudent. However, if the same woman were 5 or more years post-menopausal and had not recently discontinued HT, follow-up BMD assessment can be deferred for 3 to 5 years.
This updated guidance from NAMS emphasizes that BMD need not be assessed in women younger than 65 years unless they have specific risk factors. FRAX evaluation also makes it possible to estimate the 10-year risk of fracture in women who have low bone mass but who do not meet criteria for osteoporosis. FRAX evaluation indicates that prescription therapy is rarely required for women in their 50s or 60s who have low bone mass (but not osteoporosis). For women in their 70s or 80s who have low bone mass (but not osteoporosis), however, FRAX evaluation often leads to a recommendation to initiate prescription antifracture treatment.
Many postmenopausal women who are in their 50s or 60s and who have T-scores of –1.0 to –2.5 are given bisphosphonates, despite being at low risk of fracture. Adherence to the NAMS guidelines will help prevent unnecessary assessment and treatment. In particular, the NAMS recommendations for follow-up BMD assessment—i.e., one-time evaluation 1 to 2 years after initiating therapy and no further assessment in women found to have stable BMD at the first follow-up DXA—should simplify clinical management in this setting.
Hold off on ordering DXA testing until women meet criteria for BMD assessment. In women who do not have osteoporosis, limit use of bisphosphonates to those who have an elevated 10-year risk of fracture, as assessed using the FRAX tool.
Our menopausal patients should be taking more vitamin D supplements
Bischoff-Ferrari H. Vitamin D: what is an adequate vitamin D level and how much supplementation is necessary? Best Pract Res Clin Rheumatol. 2009;23(6):789.
Bischoff-Ferrari HA, Dawson-Hughes B, Staehelin HB, et al. Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ. 2009;339:b3692.
Stewart JW, Alekel DL, Ritland LM, et al. Serum 25-hydroxyvitamin D is related to indicators of overall physical fitness in healthy postmenopausal women. Menopause. 2009;16(6):1093–1101.
Office of Dietary Supplements. Dietary Supplement Fact Sheet: Vitamin D. National Institutes of Health Web site. http://dietary-supplements.info.nih.gov/factsheets/vitamind.asp. Updated November 13, 2009. Accessed April 2, 2010.
We have long recognized the important role vitamin D plays in promoting calcium absorption from the gut and maintaining adequate serum calcium and phosphate concentrations to enable normal bone mineralization. Now, studies reveal that the vitamin also helps prevent falls and promotes overall fitness in menopausal women. It has also become clear that traditional targets for vitamin D supplementation are inadequate.
Two recent meta-analyses of double-blind, randomized trials concluded that vitamin D reduces the risk of falls in a dose-dependent manner. Dr. Heike Bischoff-Ferrari, a Swiss scientist and a leading vitamin D researcher, points out that 1) a minimum of 700 to 1,000 IU of vitamin D supplementation daily is appropriate in menopausal women and 2) a higher amount is indicated for those who are obese or deficient in vitamin D.
Compare the current recommended dietary allowance (RDA) for adults 51 to 70 years old: 400 IU daily. The federal Food and Nutrition Board is expected to update the vitamin D RDA this spring. The 2010 NAMS statement on osteoporosis recommends a daily vitamin D intake of 800 to 1,000 IU for menopausal women.
Ask your patient to add up the aggregate daily amount of vitamin D she ingests with her multivitamin and calcium and vitamin D supplements. If it is less than 800 IU, have her purchase over-the-counter vitamin D supplements (available in 400, 1,000, and 2,000 IU capsules). Obese patients and those known to be deficient may need to ingest higher daily amounts of vitamin D.
Hormone Therapy Associated With Need for Cataract Surgery
Major Finding: Women who had ever used hormone therapy had a 14% higher risk for cataract extraction and current HT users had an 18% higher risk, compared with women who never used HT.
Data Source: A 98-month Swedish prospective study of 30,861 postmenopausal women.
Disclosures: Dr. Lindblad and her associates reported having no conflicts of interest related to the study, which was funded by Swedish government agencies and research foundations.
Women who used hormone therapy were more likely to need cataract surgery, a risk potentiated by drinking alcohol, a large Swedish prospective study found.
In a 98-month study of 30,861 postmenopausal women, those who had ever used hormone therapy (HT) had a 14% higher risk for cataract extraction and current HT users had an 18% higher risk, compared with women who had never used HT, in a multivariate adjusted analysis, Dr. Birgitta Ejdervik Lindblad and her associates reported. The study appears in the journal Ophthalmology (doi:10.1016/j.ophtha.2009.07.046).
Cataract extraction was even more likely in women who were using HT and drank alcohol. Among current HT users, any alcohol consumption was associated with a 29% higher risk for cataract extraction, and those who drank more than one alcoholic drink per day had a 42% higher risk, compared with women who were neither using HT nor drinking alcohol. (One drink was defined as about 13 g of alcohol, roughly equal to one glass of wine, bottle of beer, or drink of liquor.) Drinking alcohol has been associated with increased levels of plasma estrogen in postmenopausal women in prior studies.
Investigators collected data from women in the Swedish Mammography Cohort who completed questionnaires in September 1997 about hormone status, use of hormone therapy, and lifestyle factors. The researchers followed them through October 2005 and compared their names with those on Swedish registers of cataract surgeries, which identified 4,324 women who underwent cataract surgery during the study period.
Among women aged 65 years or older, the risk for cataract surgery was 73% higher in those using hormone therapy, compared with women who never used HT, after the researchers adjusted for the effects of alcohol consumption, smoking, diabetes, hypertension, steroid or vitamin use, body mass index, and education level.
Longer use of HT was associated with a higher risk for cataract extraction in a linear fashion, added Dr. Lindblad of the Karolinska Institute, Stockholm. Current users of hormone therapy reported a longer duration of HT (a mean of 6 years) compared with past users (4 years). Women who used HT for more than 10 years had a 20% higher risk of cataract extraction, compared with women who never used HT.
Dr. Lindblad and her associates advised caution in comparing their study with those conducted outside of Sweden because HT preparations and clinical practices vary between countries. Hormone therapy with estrogen alone is more common in the United States. And U.S. versions of HT for postmenopausal symptoms like hot flushes most commonly use conjugated estrogens alone or in combination with progesteronelike progestins, while in Sweden the predominant hormone therapy is a combination of estradiol with testosteronelike progestins. The different chemistries might have different effects on the body.
Major Finding: Women who had ever used hormone therapy had a 14% higher risk for cataract extraction and current HT users had an 18% higher risk, compared with women who never used HT.
Data Source: A 98-month Swedish prospective study of 30,861 postmenopausal women.
Disclosures: Dr. Lindblad and her associates reported having no conflicts of interest related to the study, which was funded by Swedish government agencies and research foundations.
Women who used hormone therapy were more likely to need cataract surgery, a risk potentiated by drinking alcohol, a large Swedish prospective study found.
In a 98-month study of 30,861 postmenopausal women, those who had ever used hormone therapy (HT) had a 14% higher risk for cataract extraction and current HT users had an 18% higher risk, compared with women who had never used HT, in a multivariate adjusted analysis, Dr. Birgitta Ejdervik Lindblad and her associates reported. The study appears in the journal Ophthalmology (doi:10.1016/j.ophtha.2009.07.046).
Cataract extraction was even more likely in women who were using HT and drank alcohol. Among current HT users, any alcohol consumption was associated with a 29% higher risk for cataract extraction, and those who drank more than one alcoholic drink per day had a 42% higher risk, compared with women who were neither using HT nor drinking alcohol. (One drink was defined as about 13 g of alcohol, roughly equal to one glass of wine, bottle of beer, or drink of liquor.) Drinking alcohol has been associated with increased levels of plasma estrogen in postmenopausal women in prior studies.
Investigators collected data from women in the Swedish Mammography Cohort who completed questionnaires in September 1997 about hormone status, use of hormone therapy, and lifestyle factors. The researchers followed them through October 2005 and compared their names with those on Swedish registers of cataract surgeries, which identified 4,324 women who underwent cataract surgery during the study period.
Among women aged 65 years or older, the risk for cataract surgery was 73% higher in those using hormone therapy, compared with women who never used HT, after the researchers adjusted for the effects of alcohol consumption, smoking, diabetes, hypertension, steroid or vitamin use, body mass index, and education level.
Longer use of HT was associated with a higher risk for cataract extraction in a linear fashion, added Dr. Lindblad of the Karolinska Institute, Stockholm. Current users of hormone therapy reported a longer duration of HT (a mean of 6 years) compared with past users (4 years). Women who used HT for more than 10 years had a 20% higher risk of cataract extraction, compared with women who never used HT.
Dr. Lindblad and her associates advised caution in comparing their study with those conducted outside of Sweden because HT preparations and clinical practices vary between countries. Hormone therapy with estrogen alone is more common in the United States. And U.S. versions of HT for postmenopausal symptoms like hot flushes most commonly use conjugated estrogens alone or in combination with progesteronelike progestins, while in Sweden the predominant hormone therapy is a combination of estradiol with testosteronelike progestins. The different chemistries might have different effects on the body.
Major Finding: Women who had ever used hormone therapy had a 14% higher risk for cataract extraction and current HT users had an 18% higher risk, compared with women who never used HT.
Data Source: A 98-month Swedish prospective study of 30,861 postmenopausal women.
Disclosures: Dr. Lindblad and her associates reported having no conflicts of interest related to the study, which was funded by Swedish government agencies and research foundations.
Women who used hormone therapy were more likely to need cataract surgery, a risk potentiated by drinking alcohol, a large Swedish prospective study found.
In a 98-month study of 30,861 postmenopausal women, those who had ever used hormone therapy (HT) had a 14% higher risk for cataract extraction and current HT users had an 18% higher risk, compared with women who had never used HT, in a multivariate adjusted analysis, Dr. Birgitta Ejdervik Lindblad and her associates reported. The study appears in the journal Ophthalmology (doi:10.1016/j.ophtha.2009.07.046).
Cataract extraction was even more likely in women who were using HT and drank alcohol. Among current HT users, any alcohol consumption was associated with a 29% higher risk for cataract extraction, and those who drank more than one alcoholic drink per day had a 42% higher risk, compared with women who were neither using HT nor drinking alcohol. (One drink was defined as about 13 g of alcohol, roughly equal to one glass of wine, bottle of beer, or drink of liquor.) Drinking alcohol has been associated with increased levels of plasma estrogen in postmenopausal women in prior studies.
Investigators collected data from women in the Swedish Mammography Cohort who completed questionnaires in September 1997 about hormone status, use of hormone therapy, and lifestyle factors. The researchers followed them through October 2005 and compared their names with those on Swedish registers of cataract surgeries, which identified 4,324 women who underwent cataract surgery during the study period.
Among women aged 65 years or older, the risk for cataract surgery was 73% higher in those using hormone therapy, compared with women who never used HT, after the researchers adjusted for the effects of alcohol consumption, smoking, diabetes, hypertension, steroid or vitamin use, body mass index, and education level.
Longer use of HT was associated with a higher risk for cataract extraction in a linear fashion, added Dr. Lindblad of the Karolinska Institute, Stockholm. Current users of hormone therapy reported a longer duration of HT (a mean of 6 years) compared with past users (4 years). Women who used HT for more than 10 years had a 20% higher risk of cataract extraction, compared with women who never used HT.
Dr. Lindblad and her associates advised caution in comparing their study with those conducted outside of Sweden because HT preparations and clinical practices vary between countries. Hormone therapy with estrogen alone is more common in the United States. And U.S. versions of HT for postmenopausal symptoms like hot flushes most commonly use conjugated estrogens alone or in combination with progesteronelike progestins, while in Sweden the predominant hormone therapy is a combination of estradiol with testosteronelike progestins. The different chemistries might have different effects on the body.
Aerobic Exercise May Cut Menopausal Symptoms
Major Finding: Six months of 70-minute aerobic exercise sessions three days per week significantly reduced the severity of menopausal symptoms while improving physical fitness.
Data Source: An uncontrolled study in 65 postmenopausal women.
Disclosures: None reported.
Postmenopausal women improved their physical fitness and reported reductions in the severity of menopausal symptoms after 12–24 weeks of aerobic exercise in three 70-minute sessions per week.
The 65 women (mean age, 50.1 years) rated the severity of menopausal symptoms on the self-administered Menopause Rating Scale questionnaire at baseline, 12 weeks, and 24 weeks in the uncontrolled study. The program of aerobic and calisthenic exercise aimed for 75%–80% of maximal heart rate according to the Karvonen method and consisted of 10 minutes of warm-up exercises; 40 minutes of aerobics; 15 minutes of exercise targeting the abdomen, hip, and leg muscles; and 5 minutes for cooldown and stretching.
Participants reported significant decreases in the severity of hot flushes, night sweats, cardiac symptoms, muscle and joint pain, sleeping disorder symptoms, depressive mood, irritability, anxiety, exhaustion, sexual problems, and urinary symptoms between the start and end of the study, Dr. Selma Karacan of Selcuk University in Konya, Turkey, reported (Sci. Sports 2009 [doi:10.1016/j.scispo.2009.07.004]).
Some of the symptoms showed improvement by 12 weeks and further significant improvements by 24 weeks, including vasomotor symptoms, muscle and joint pain, psychological symptoms, and sexual problems. The women reported no significant change in vaginal dryness.
Significant improvements also were seen in resting heart rate, systolic and diastolic blood pressures, flexibility, aerobic power, and the ability to perform sit-ups, push-ups, and right or left hand grips. Body weight, body mass index, body fat percentage, and fat weight decreased significantly, with no change in lean body mass values.
The findings support results from previous observational studies of physically active postmenopausal women compared with age-matched, sedentary control women. No randomized controlled trials have looked at the efficacy of exercise in managing hot flushes.
The current study suggests that a high level of cardiorespiratory fitness may help reduce menopausal symptoms, Dr. Karacan concluded.
Major Finding: Six months of 70-minute aerobic exercise sessions three days per week significantly reduced the severity of menopausal symptoms while improving physical fitness.
Data Source: An uncontrolled study in 65 postmenopausal women.
Disclosures: None reported.
Postmenopausal women improved their physical fitness and reported reductions in the severity of menopausal symptoms after 12–24 weeks of aerobic exercise in three 70-minute sessions per week.
The 65 women (mean age, 50.1 years) rated the severity of menopausal symptoms on the self-administered Menopause Rating Scale questionnaire at baseline, 12 weeks, and 24 weeks in the uncontrolled study. The program of aerobic and calisthenic exercise aimed for 75%–80% of maximal heart rate according to the Karvonen method and consisted of 10 minutes of warm-up exercises; 40 minutes of aerobics; 15 minutes of exercise targeting the abdomen, hip, and leg muscles; and 5 minutes for cooldown and stretching.
Participants reported significant decreases in the severity of hot flushes, night sweats, cardiac symptoms, muscle and joint pain, sleeping disorder symptoms, depressive mood, irritability, anxiety, exhaustion, sexual problems, and urinary symptoms between the start and end of the study, Dr. Selma Karacan of Selcuk University in Konya, Turkey, reported (Sci. Sports 2009 [doi:10.1016/j.scispo.2009.07.004]).
Some of the symptoms showed improvement by 12 weeks and further significant improvements by 24 weeks, including vasomotor symptoms, muscle and joint pain, psychological symptoms, and sexual problems. The women reported no significant change in vaginal dryness.
Significant improvements also were seen in resting heart rate, systolic and diastolic blood pressures, flexibility, aerobic power, and the ability to perform sit-ups, push-ups, and right or left hand grips. Body weight, body mass index, body fat percentage, and fat weight decreased significantly, with no change in lean body mass values.
The findings support results from previous observational studies of physically active postmenopausal women compared with age-matched, sedentary control women. No randomized controlled trials have looked at the efficacy of exercise in managing hot flushes.
The current study suggests that a high level of cardiorespiratory fitness may help reduce menopausal symptoms, Dr. Karacan concluded.
Major Finding: Six months of 70-minute aerobic exercise sessions three days per week significantly reduced the severity of menopausal symptoms while improving physical fitness.
Data Source: An uncontrolled study in 65 postmenopausal women.
Disclosures: None reported.
Postmenopausal women improved their physical fitness and reported reductions in the severity of menopausal symptoms after 12–24 weeks of aerobic exercise in three 70-minute sessions per week.
The 65 women (mean age, 50.1 years) rated the severity of menopausal symptoms on the self-administered Menopause Rating Scale questionnaire at baseline, 12 weeks, and 24 weeks in the uncontrolled study. The program of aerobic and calisthenic exercise aimed for 75%–80% of maximal heart rate according to the Karvonen method and consisted of 10 minutes of warm-up exercises; 40 minutes of aerobics; 15 minutes of exercise targeting the abdomen, hip, and leg muscles; and 5 minutes for cooldown and stretching.
Participants reported significant decreases in the severity of hot flushes, night sweats, cardiac symptoms, muscle and joint pain, sleeping disorder symptoms, depressive mood, irritability, anxiety, exhaustion, sexual problems, and urinary symptoms between the start and end of the study, Dr. Selma Karacan of Selcuk University in Konya, Turkey, reported (Sci. Sports 2009 [doi:10.1016/j.scispo.2009.07.004]).
Some of the symptoms showed improvement by 12 weeks and further significant improvements by 24 weeks, including vasomotor symptoms, muscle and joint pain, psychological symptoms, and sexual problems. The women reported no significant change in vaginal dryness.
Significant improvements also were seen in resting heart rate, systolic and diastolic blood pressures, flexibility, aerobic power, and the ability to perform sit-ups, push-ups, and right or left hand grips. Body weight, body mass index, body fat percentage, and fat weight decreased significantly, with no change in lean body mass values.
The findings support results from previous observational studies of physically active postmenopausal women compared with age-matched, sedentary control women. No randomized controlled trials have looked at the efficacy of exercise in managing hot flushes.
The current study suggests that a high level of cardiorespiratory fitness may help reduce menopausal symptoms, Dr. Karacan concluded.
Remove the ovaries at hysterectomy? Here’s the lowdown on risks and benefits
CASE: Hysterectomy candidate asks about her ovaries
A 51-year-old premenopausal woman complains of severe menorrhagia that often causes her to miss work. Although she is taking an iron supplement, her hemoglobin level often drops below 10 g/dL. She has already been identified as having fibroids, with a uterine size of 14 weeks. You order ultra-sonography, which reveals an enlarged uterus with multiple fibroids and normal endometrial thickness, but no intracavitary lesions.
After you describe the treatment options, including uterine artery embolization, the patient requests a hysterectomy as a reasonably low-risk means of cure. During informed consent, she asks whether she should have her ovaries removed during the surgery. Further discussion reveals that her father died of a myocardial infarction when he was 64 years old, but there is no family or personal history of ovarian or breast cancer.
How do you advise this patient, based on her history and recent findings from medical research?
Many gynecologists have been trained to recommend bilateral oophorectomy for women older than 45 or 50 years who request a hysterectomy for benign disease. In these women, oophorectomy is recommended to prevent ovarian cancer and avert the potential for other ovarian pathology that might require later surgery.
In the United States, 78% of women 45 to 64 years old and 55% of women overall undergo bilateral oophorectomy at the time of hysterectomy.1 These percentages mean that almost 300,000 women undergo bilateral oophorectomy each year.1
Hysterectomy alone can sometimes lead to early ovarian failure, but this phenomenon is infrequent. A prospective study of premenopausal women found that, after 5 years of follow-up, 20% of women who underwent simple hysterectomy reached menopause, compared with 7% of matched women who did not undergo hysterectomy.2
In this article, I explore the risks and benefits associated with bilateral oophorectomy and present an algorithm to aid in deciding whether the patient should keep her ovaries—and when oophorectomy might be a better option ( FIGURE ).
Among the hazards associated with bilateral oophorectomy are:
- an increased risk of death from coronary artery disease (CAD), lung cancer, all cancers (except ovarian), and all causes3,4
- an increased risk of osteoporosis and hip fracture5
- when performed before the onset of menopause, an increased risk of parkinsonism, cognitive impairment, dementia, anxiety, and depression.6-8
Benefits include a reduced risk of ovarian cancer, particularly among women who have a BRCA gene mutation or strong family history of ovarian or breast cancer.
Although ovarian cancer causes 15,000 deaths each year in the United States, that figure pales when compared with heart disease, which accounts for 350,000 deaths. In addition, hip fracture may cause approximately 66,000 deaths each year, and dementia attributable to bilateral oophorectomy may affect 100,000 to 200,000 women.9 Reoperation for adnexal pathology or pain after hysterectomy is rare, occurring in only 2.8% of women. Therefore, the benefits of oophorectomy are often outweighed by the risks of CAD, hip fracture, and neurologic conditions.
The assumption that medical treatment will ameliorate the risks associated with oophorectomy is unrealistic. Estrogen may mitigate some risks, but many women avoid hormone therapy. This avoidance can be especially problematic in young women.
In the Nurses’ Health Study, a separate analysis focused on women who had never used postmenopausal hormone therapy.4 In this analysis, all women who underwent bilateral oophorectomy had a greater risk of stroke (HR, 1.85; 95% CI, 1.09, 3.16) and lung cancer (HR, 2.09; 95% CI, 1.01, 4.33) than did women who retained their ovaries. Among women who underwent oophorectomy before age 50 and who did not take estrogen, the risk of coronary artery disease (CAD) was higher (HR, 1.98; 95% CI, 1.18, 3.32), as was the risk of death from all causes (HR, 1.40; 95% CI, 1.01, 1.96), compared with women who retained their ovaries.
Despite estrogen’s proven benefit among oophorectomized women, usage rates continue to decline. In the 6 months after publication of Women’s Health Initiative findings on estrogen–progestin therapy, the continuation rate of estrogen therapy decreased from 12.6% to 9.1%, and new starts also declined significantly.47
Among women who had a diagnosis of osteoporosis and who began treatment with estrogen, estrogen plus progestin, a bisphosphonate, or raloxifene, medication continuation rates were less than 25% at 12 months.48 Moreover, only 18% of women started on a statin to reduce the risk of cardiovascular disease were still taking the drug after 1 year.49
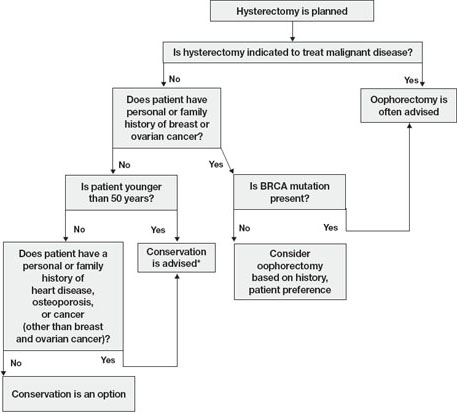
FIGURE Conservation vs oophorectomy: A guide to decision-making
* Estrogen replacement is recommended for women younger than 45 years who opt for oophorectomy
Ovarian cancer is a real, but relatively low, risk
In 2008, an estimated 21,650 new cases of ovarian cancer were diagnosed (age at diagnosis: mean, 63 years), and 15,520 women died from the disease.10 Because we lack a reliable screening test to detect early-stage ovarian cancer in the general population, most women are given a diagnosis when disease is advanced and the 5-year survival rate is 15% to 25%.
There is agreement that women who are known to have a BRCA mutation, which increases the risk of ovarian and breast cancer, should strongly consider oophorectomy once childbearing is complete.11 In the general population, however, the outlook is different.
In the United States, the lifetime risk of ovarian cancer is 1.4% overall. Among white women who have had three or more term pregnancies and who have used an oral contraceptive for at least 4 years, the lifetime risk of ovarian cancer drops to 0.3%.12
Need for reoperation is very low
The percentage of women who require reoperation after ovarian conservation—2.8%—may surprise you.13 That figure is lower than once thought because many studies were performed before asymptomatic, benign ovarian cysts were determined to be a fairly common phenomenon in postmenopausal women (prevalence, 6.6%). These cysts do not undergo transformation to cancer and, therefore, do not need to be removed.14
In addition, studies indicate that only 0.1% to 0.75% of women who retain their ovaries at the time of hysterectomy develop ovarian cancer.15,16 Therefore, the rationale of performing oophorectomy to avoid future surgery appears to be unfounded.
CAD risk rises sharply after oophorectomy
A recent systematic review found mixed evidence concerning the risk of CAD following bilateral salpingo-oophorectomy.17 In observational studies, however, earlier age of surgical or natural menopause has been associated with a higher risk of cardiovascular mortality.18-20 Early reports from the Nurses’ Health Study found that the risk of myocardial infarction doubled among women who underwent oophorectomy and never used estrogen, compared with age-matched premenopausal women (relative risk [RR], 2.2; 95% confidence interval [CI], 1.2, 4.2).3 Even after age 50, the risk of a first myocardial infarction is increased among oophorectomized women, compared with women who retain their ovaries (RR, 1.4; 95% CI, 1.0–2.0).21
A study by researchers from the Mayo Clinic, who examined all causes listed on the death certificate, found a significant association between bilateral oophorectomy before the age of 45 years and cardiovascular mortality (hazard ratio [HR], 1.44; 95% CI, 1.01–2.05).22 This risk was significantly increased among women who were not treated with estrogen through at least age 45, compared with estrogen-treated women.
Oophorectomy may impair bone health
After menopause, ovaries continue to produce significant amounts of the androgens testosterone and androstenedione, which are converted to estrone peripherally by skin, muscle, and fat cells.23,24 The levels of these hormones remain consistent and have been documented to age 80.25
Both estrogens and androgens inhibit bone resorption, and androgens also stimulate bone formation.26 Low levels of androgens and estrogens are linked to lower bone density and a higher risk of hip and vertebral fracture in postmenopausal women.27-29
Postmenopausal women who have been oophorectomized may have an even greater risk of osteoporosis. Over 16 years of follow-up, 340 women who had undergone oophorectomy at a median age of 62 years had 54% more osteoporotic fractures than women who had intact ovaries.5 Two other studies found no association between oophorectomy and bone loss or fracture risk, however.30,31
Hip fracture is a well-documented cause of increased morbidity and mortality in older women. One study found that, before hip fracture, 28% of patients were housebound; 1 year after hip fracture, the percentage was 46%.32 Women older than 60 who underwent oophorectomy had a doubled risk of mortality after low-trauma hip fracture, compared with women who had intact ovaries (odds ratio [OR], 2.18; 95% CI, 2.03–2.32).5
Loss of ovaries may affect mental health and sexuality
In a premenopausal woman, oophorectomy causes a sudden loss of estrogen and often triggers hot flashes, mood changes, sleep disturbances, headaches, and a decline in feelings of well-being.33,34 Over time, vaginal dryness, painful intercourse, loss of libido, bladder dysfunction, and depression may occur.35,36
Evidence suggests that, in women, sexual desire, sexual sensation, and orgasmic response are influenced by androgens. After elective oophorectomy, declines in sexual desire have been reported.37-39
Mental health and sexuality may rebound over time, however. One study found less improvement in mental health measures and body image 6 months after hysterectomy among women who were oophorectomized, compared with those who retained their ovaries. After 2 years, improvement levels were similar between groups.40
Cognitive function may suffer
Analysis of data from the Mayo Clinic Cohort Study of Oophorectomy and Aging found that bilateral oophorectomy before the onset of menopause increased the risk of parkinsonism, cognitive impairment or dementia, and anxiety or depression, particularly if estrogen was not replaced.6-8 These risks increased with younger age at oophorectomy.
The Women’s Health Initiative found an increased risk of dementia or mild cognitive impairment in women who were treated by estrogen alone or estrogen plus progestin after age 65.41-44
These disparate conclusions suggest that estrogen may have a protective effect on the brain if it is given right after natural menopause or premenopausal oophorectomy, but deleterious effects if it is started years later.45
Other studies of endogenous estrogen and cognitive function are few and yield inconsistent results.
Ovarian conservation boosts long-term survival
When there is no family history of ovarian cancer, ovarian conservation appears to maximize survival among healthy women 40 to 65 years old who undergo hysterectomy for benign disease.46 Among healthy women hysterectomized before the age of 55, calculations suggest that 8.6% more would be alive at age 80 if their ovaries were conserved than if they were removed.46
A study from the Mayo Clinic found that all-cause mortality was significantly higher among women who underwent prophylactic bilateral oophorectomy before the age of 45 than it was among women in the control group (HR, 1.67; 95% CI, 1.16–2.40); it was particularly high in women who did not receive estrogen treatment before age 45 (HR, 1.93; 95% CI, 1.25–2.96).22
In a recent study, investigators used the Nurses’ Health Study database to explore the long-term health outcomes of 29,380 women who underwent hysterectomy.4 Of these women, 13,035 (44.4%) had their ovaries conserved, and 16,345 (55.6%) underwent bilateral oophorectomy. Follow-up was 24 years. Oophorectomy was associated with an increased risk of nonfatal CAD among all women (HR, 1.17; 95% CI, 1.02, 1.35), especially those who underwent the procedure before age 45 (HR, 1.26; 95% CI, 1.04, 1.54). Oophorectomy was associated with a markedly reduced risk of ovarian cancer but an increased risk of lung cancer (HR, 1.26; 95% CI, 1.02–1.56).
In regard to fatal events, oophorectomy increased the risk of death from all causes (HR, 1.12; 95% CI, 1.03, 1.21). Specifically, there was an increased risk of death from CAD (HR, 1.28; 95% CI, 1.00, 1.64), lung cancer (HR, 1.31; 95% CI, 1.02, 1.68), and all cancers (HR, 1.17; 95% CI, 1.04, 1.32). There was no overall difference in the risk of death from stroke, breast cancer, and colorectal cancer between women who underwent oophorectomy and those who retained their ovaries.
During the 24 years of follow-up, 37 women died from ovarian cancer, accounting for 1.2% of all deaths. At no age did oophorectomy show a survival benefit.
How this evidence should inform your practice
It is unfortunate that the entire body of evidence on the risks and benefits of bilateral salpingo-oophorectomy consists of observational studies, which have significant inherent limitations. Although the Nurses’ Health Study was the largest prospective study to examine the effect of oophorectomy on women’s health, and involved the longest follow-up, the study was observational, and oophorectomy and ovarian conservation were self-selected. Nevertheless, recent data suggest that a more detailed informed-consent process is warranted than the process in place. Informed consent should cover the risks and benefits of both oophorectomy and ovarian conservation.
Prophylactic oophorectomy is recommended only if a preponderance of the evidence establishes that it clearly benefits the patient. The studies described in this article suggest that bilateral oophorectomy does harm more often than it does good. Therefore, a cautious approach to oophorectomy at the time of hysterectomy is advised.
CASE RESOLVED
After you describe the risks and benefits of oophorectomy, and address the patient’s concerns about her family history of heart disease, she decides to keep her ovaries.
1. Healthcare Cost and Utilization Project (HCUP), 1988–2001: A Federal–State–Industry Partnership in Health Data. Rockville, Md: Agency for Healthcare Research and Quality; July 2003.
2. Farquhar CM, Sadler L, Harvey SA, Stewart AW. The association of hysterectomy and menopause: a prospective cohort study. BJOG. 2005;112:956-962.
3. Colditz G, Willett W, Stampfer M, Rosner B, Speizer F, Hennekens C. Menopause and the risk of coronary heart disease in women. N Engl J Med. 1987;316:1105-1110.
4. Parker WH, Broder MS, Chang E, et al. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstet Gynecol. 2009;113:1027-1037.
5. Melton LJ 3rd, Khosla S, Malkasian GD, Achenbach SJ, Oberg AL, Riggs BL. Fracture risk after bilateral oophorectomy in elderly women. J Bone Miner Res. 2003;18:900-905.
6. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology. 2007;69:1074.-
7. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of parkinsonism in women who underwent oophorectomy before menopause. Neurology. 2008;70:200-209.
8. Rocca WA, Grossardt BR, Geda YE, et al. Long-term risk of depressive and anxiety symptoms after early bilateral oophorectomy. Menopause. 2008;15:1050-1059.
9. Bennett DA. Editorial comment on “Prevalence of dementia in the United States: the aging, demographics, and memory study” by Plassman et al. Neuroepidemiology. 2007;29:133-135.
10. Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71-96.
11. Armstrong K, Schwartz J, Randall T, Rubin S, Weber B. Hormone replacement therapy and life expectancy after prophylactic oophorectomy in women with BRCA1/2 mutations: a decision analysis. J Clin Oncol. 2004;22:1045-1054.
12. Hartge P, Whittemore AS, Itnyre J, McGowan L, Cramer D. Rates and risks of ovarian cancer in subgroups of white women in the United States. The Collaborative Ovarian Cancer Group. Obstet Gynecol. 1994;84:760-764.
13. Dekel A, Efrat Z, Orvieto R, et al. The residual ovary syndrome: a 20-year experience. Eur J Obstet Gynecol Reprod Biol. 1996;68:159-164.
14. Bailey CL, Ueland FR, Land GL, et al. The malignant potential of small cystic ovarian tumors in women over 50 years of age. Gynecol Oncol. 1998;69:3-7.
15. Naylor AC. Hysterectomy—analysis of 2,901 personally performed procedures. S Afr Med J. 1984;65:242-245.
16. Christ JE, Lotze EC. The residual ovary syndrome. Obstet Gynecol. 1975;46:551-556.
17. Jacoby VL, Grady D, Sawaya GF. Oophorectomy as a risk factor for coronary heart disease. Am J Obstet Gynecol. 2009;200:140.e1-140.e9.
18. van der Schouw YT, van der Graaf Y, Steyerberg EW, Eijkemans JC, Banga JD. Age at menopause as a risk factor for cardiovascular mortality. Lancet. 1996;347:714-718.
19. Løkkegaard E, Jovanovic Z, Heitmann BL, et al. The association between early menopause and risk of ischaemic heart disease: influence of hormone therapy. Maturitas. 2006;53:226-233.
20. de Kleijn MJJ, van der Schouw YT, Verbeek ALM, et al. Endogenous estrogen exposure and cardiovascular mortality risk in postmenopausal women. Am J Epidemiol. 2002;155:339.-
21. Falkeborn M, Schairer C, Naessen T, Persson I. Risk of myocardial infarction after oophorectomy and hysterectomy. J Clin Epidemiol. 2000;53:832-837.
22. Rocca WA, Grossardt BR, de Andrade M, Malkasian GD, Melton LJ, 3rd. Survival patterns after oophorectomy in premenopausal women: a population-based cohort study. Lancet Oncol. 2006;7:821-828.
23. Judd H, Judd G, Lucas W, Yen S. Endocrine function of the postmenopausal ovary: concentration of androgens and estrogens in ovarian and peripheral vein blood. J Clin Endocrinol Metab. 1974;39:1020-1024.
24. Fogle R, Stanczyk F, Zhang X, Paulson R. Ovarian androgen production in postmenopausal women. J Clin Endocrinol Metab. 2007;92:3040-3043.
25. Meldrum D, Davidson B, Tataryn I, Judd H. Changes in circulating steroids with aging in postmenopausal women. Obstet Gynecol. 1981;57:624-628.
26. Raisz LG, Wilta B, Artis A, et al. Comparison of the effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab. 1996;81:37-43.
27. Johansson C, Mellstrom D, Milsom I. Reproductive factors as predictors of bone density and fractures in women at age 70. Maturitas. 1993;17:39-50.
28. Davidson BJ, Ross RK, Paganini-Hill A, et al. Total and free estrogens and androgens in postmenopausal women with hip fractures. J Clin Endocrinol Metab. 1982;54:115-120.
29. Cummings SR, Browner WS, Bauer D, et al. Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N Engl J Med. 1998;339:733-738.
30. Kritz-Silverstein D, von Mühlen D, Barrett-Connor E. Hysterectomy and oophorectomy are unrelated to bone loss in older women. Maturitas. 2004;47:61-69.
31. Antoniucci DM, Sellmeyer DE, Cauley JA, et al. Postmenopausal bilateral oophorectomy is not associated with increased fracture risk in older women. J Bone Miner Res. 2005;20:741-747.
32. Keene GS, Parker MJ, Pryor GA. Mortality and morbidity after hip fractures. BMJ. 1993;307:1248-1250.
33. Nathorst-Boos J, von Schoultz B, Carlstrom K. Elective ovarian removal and estrogen replacement therapy—effects on sexual life, psychological well-being and androgen status. J Psychosom Obstet Gynaecol. 1993;14:283-293.
34. Nieman L. Management of surgically hypogonadal patients unable to take sex hormone replacement therapy. Endocrinol Metab Clin North Am. 2003;32:325-336.
35. Shifren J. Androgen deficiency in the oophorectomized woman. Fertil Steril. 2002;77 Suppl 4:s60-s62.
36. Sherwin B, Gelfan M. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med. 1978;49:397-409.
37. Shifren JL, Avis NE. Surgical menopause: effects on psychological well-being and sexuality. Menopause. 2007;14:586.-
38. Elit L, Esplen MJ, Butler K, Narod S. Quality of life and psychosexual adjustment after prophylactic oophorectomy for a family history of ovarian cancer. Fam Cancer. 2001;1:149-156.
39. Sherwin BB, Gelfan MM. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med 1978;49:397-409.
40. Teplin V, Vittinghoff E, Lin F, Learman L, Richter H, Kuppermann M. Oophorectomy in premenopausal women: health-related quality of life and sexual functioning. Obstet Gynecol. 2007;109:347-354.
41. Shumaker SA, Legault C, Rapp SR, et al. WHIMS Investigators Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2651-2662.
42. Rapp SR, Espeland MA, Shumaker SA, et al. WHIMS Investigators. Effect of estrogen plus progestin on global cognitive function in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2663-2672.
43. Espeland MA, Rapp SR, Shumaker SA, et al. Conjugated equine estrogens and global cognitive function in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2959-2968.
44. Shumaker SA, Legault C, Kuller L, et al. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2947-2958.
45. Siegfried T. Neuroscience: it’s all in the timing. Nature. 2007;445:359-361.
46. Parker W, Broder M, Liu Z, Shoupe D, Farquhar C, Berek J. Ovarian conservation at the time of hysterectomy for benign disease. Obstet Gynecol. 2005;106:219-226.
47. Buist DS, Newton KM, Miglioretti DL, et al. Hormone therapy prescribing patterns in the United States. Obstet Gynecol. 2004;104:1042-1050.
48. McCombs JS, Thiebaud P, McLaughlin-Miley C, Shi J. Compliance with drug therapies for the treatment and prevention of osteoporosis. Maturitas. 2004;48:271-287.
49. Huser MA, Evans TS, Berger V. Medication adherence trends with statins. Adv Ther. 2005;22:163-171.
CASE: Hysterectomy candidate asks about her ovaries
A 51-year-old premenopausal woman complains of severe menorrhagia that often causes her to miss work. Although she is taking an iron supplement, her hemoglobin level often drops below 10 g/dL. She has already been identified as having fibroids, with a uterine size of 14 weeks. You order ultra-sonography, which reveals an enlarged uterus with multiple fibroids and normal endometrial thickness, but no intracavitary lesions.
After you describe the treatment options, including uterine artery embolization, the patient requests a hysterectomy as a reasonably low-risk means of cure. During informed consent, she asks whether she should have her ovaries removed during the surgery. Further discussion reveals that her father died of a myocardial infarction when he was 64 years old, but there is no family or personal history of ovarian or breast cancer.
How do you advise this patient, based on her history and recent findings from medical research?
Many gynecologists have been trained to recommend bilateral oophorectomy for women older than 45 or 50 years who request a hysterectomy for benign disease. In these women, oophorectomy is recommended to prevent ovarian cancer and avert the potential for other ovarian pathology that might require later surgery.
In the United States, 78% of women 45 to 64 years old and 55% of women overall undergo bilateral oophorectomy at the time of hysterectomy.1 These percentages mean that almost 300,000 women undergo bilateral oophorectomy each year.1
Hysterectomy alone can sometimes lead to early ovarian failure, but this phenomenon is infrequent. A prospective study of premenopausal women found that, after 5 years of follow-up, 20% of women who underwent simple hysterectomy reached menopause, compared with 7% of matched women who did not undergo hysterectomy.2
In this article, I explore the risks and benefits associated with bilateral oophorectomy and present an algorithm to aid in deciding whether the patient should keep her ovaries—and when oophorectomy might be a better option ( FIGURE ).
Among the hazards associated with bilateral oophorectomy are:
- an increased risk of death from coronary artery disease (CAD), lung cancer, all cancers (except ovarian), and all causes3,4
- an increased risk of osteoporosis and hip fracture5
- when performed before the onset of menopause, an increased risk of parkinsonism, cognitive impairment, dementia, anxiety, and depression.6-8
Benefits include a reduced risk of ovarian cancer, particularly among women who have a BRCA gene mutation or strong family history of ovarian or breast cancer.
Although ovarian cancer causes 15,000 deaths each year in the United States, that figure pales when compared with heart disease, which accounts for 350,000 deaths. In addition, hip fracture may cause approximately 66,000 deaths each year, and dementia attributable to bilateral oophorectomy may affect 100,000 to 200,000 women.9 Reoperation for adnexal pathology or pain after hysterectomy is rare, occurring in only 2.8% of women. Therefore, the benefits of oophorectomy are often outweighed by the risks of CAD, hip fracture, and neurologic conditions.
The assumption that medical treatment will ameliorate the risks associated with oophorectomy is unrealistic. Estrogen may mitigate some risks, but many women avoid hormone therapy. This avoidance can be especially problematic in young women.
In the Nurses’ Health Study, a separate analysis focused on women who had never used postmenopausal hormone therapy.4 In this analysis, all women who underwent bilateral oophorectomy had a greater risk of stroke (HR, 1.85; 95% CI, 1.09, 3.16) and lung cancer (HR, 2.09; 95% CI, 1.01, 4.33) than did women who retained their ovaries. Among women who underwent oophorectomy before age 50 and who did not take estrogen, the risk of coronary artery disease (CAD) was higher (HR, 1.98; 95% CI, 1.18, 3.32), as was the risk of death from all causes (HR, 1.40; 95% CI, 1.01, 1.96), compared with women who retained their ovaries.
Despite estrogen’s proven benefit among oophorectomized women, usage rates continue to decline. In the 6 months after publication of Women’s Health Initiative findings on estrogen–progestin therapy, the continuation rate of estrogen therapy decreased from 12.6% to 9.1%, and new starts also declined significantly.47
Among women who had a diagnosis of osteoporosis and who began treatment with estrogen, estrogen plus progestin, a bisphosphonate, or raloxifene, medication continuation rates were less than 25% at 12 months.48 Moreover, only 18% of women started on a statin to reduce the risk of cardiovascular disease were still taking the drug after 1 year.49

FIGURE Conservation vs oophorectomy: A guide to decision-making
* Estrogen replacement is recommended for women younger than 45 years who opt for oophorectomy
Ovarian cancer is a real, but relatively low, risk
In 2008, an estimated 21,650 new cases of ovarian cancer were diagnosed (age at diagnosis: mean, 63 years), and 15,520 women died from the disease.10 Because we lack a reliable screening test to detect early-stage ovarian cancer in the general population, most women are given a diagnosis when disease is advanced and the 5-year survival rate is 15% to 25%.
There is agreement that women who are known to have a BRCA mutation, which increases the risk of ovarian and breast cancer, should strongly consider oophorectomy once childbearing is complete.11 In the general population, however, the outlook is different.
In the United States, the lifetime risk of ovarian cancer is 1.4% overall. Among white women who have had three or more term pregnancies and who have used an oral contraceptive for at least 4 years, the lifetime risk of ovarian cancer drops to 0.3%.12
Need for reoperation is very low
The percentage of women who require reoperation after ovarian conservation—2.8%—may surprise you.13 That figure is lower than once thought because many studies were performed before asymptomatic, benign ovarian cysts were determined to be a fairly common phenomenon in postmenopausal women (prevalence, 6.6%). These cysts do not undergo transformation to cancer and, therefore, do not need to be removed.14
In addition, studies indicate that only 0.1% to 0.75% of women who retain their ovaries at the time of hysterectomy develop ovarian cancer.15,16 Therefore, the rationale of performing oophorectomy to avoid future surgery appears to be unfounded.
CAD risk rises sharply after oophorectomy
A recent systematic review found mixed evidence concerning the risk of CAD following bilateral salpingo-oophorectomy.17 In observational studies, however, earlier age of surgical or natural menopause has been associated with a higher risk of cardiovascular mortality.18-20 Early reports from the Nurses’ Health Study found that the risk of myocardial infarction doubled among women who underwent oophorectomy and never used estrogen, compared with age-matched premenopausal women (relative risk [RR], 2.2; 95% confidence interval [CI], 1.2, 4.2).3 Even after age 50, the risk of a first myocardial infarction is increased among oophorectomized women, compared with women who retain their ovaries (RR, 1.4; 95% CI, 1.0–2.0).21
A study by researchers from the Mayo Clinic, who examined all causes listed on the death certificate, found a significant association between bilateral oophorectomy before the age of 45 years and cardiovascular mortality (hazard ratio [HR], 1.44; 95% CI, 1.01–2.05).22 This risk was significantly increased among women who were not treated with estrogen through at least age 45, compared with estrogen-treated women.
Oophorectomy may impair bone health
After menopause, ovaries continue to produce significant amounts of the androgens testosterone and androstenedione, which are converted to estrone peripherally by skin, muscle, and fat cells.23,24 The levels of these hormones remain consistent and have been documented to age 80.25
Both estrogens and androgens inhibit bone resorption, and androgens also stimulate bone formation.26 Low levels of androgens and estrogens are linked to lower bone density and a higher risk of hip and vertebral fracture in postmenopausal women.27-29
Postmenopausal women who have been oophorectomized may have an even greater risk of osteoporosis. Over 16 years of follow-up, 340 women who had undergone oophorectomy at a median age of 62 years had 54% more osteoporotic fractures than women who had intact ovaries.5 Two other studies found no association between oophorectomy and bone loss or fracture risk, however.30,31
Hip fracture is a well-documented cause of increased morbidity and mortality in older women. One study found that, before hip fracture, 28% of patients were housebound; 1 year after hip fracture, the percentage was 46%.32 Women older than 60 who underwent oophorectomy had a doubled risk of mortality after low-trauma hip fracture, compared with women who had intact ovaries (odds ratio [OR], 2.18; 95% CI, 2.03–2.32).5
Loss of ovaries may affect mental health and sexuality
In a premenopausal woman, oophorectomy causes a sudden loss of estrogen and often triggers hot flashes, mood changes, sleep disturbances, headaches, and a decline in feelings of well-being.33,34 Over time, vaginal dryness, painful intercourse, loss of libido, bladder dysfunction, and depression may occur.35,36
Evidence suggests that, in women, sexual desire, sexual sensation, and orgasmic response are influenced by androgens. After elective oophorectomy, declines in sexual desire have been reported.37-39
Mental health and sexuality may rebound over time, however. One study found less improvement in mental health measures and body image 6 months after hysterectomy among women who were oophorectomized, compared with those who retained their ovaries. After 2 years, improvement levels were similar between groups.40
Cognitive function may suffer
Analysis of data from the Mayo Clinic Cohort Study of Oophorectomy and Aging found that bilateral oophorectomy before the onset of menopause increased the risk of parkinsonism, cognitive impairment or dementia, and anxiety or depression, particularly if estrogen was not replaced.6-8 These risks increased with younger age at oophorectomy.
The Women’s Health Initiative found an increased risk of dementia or mild cognitive impairment in women who were treated by estrogen alone or estrogen plus progestin after age 65.41-44
These disparate conclusions suggest that estrogen may have a protective effect on the brain if it is given right after natural menopause or premenopausal oophorectomy, but deleterious effects if it is started years later.45
Other studies of endogenous estrogen and cognitive function are few and yield inconsistent results.
Ovarian conservation boosts long-term survival
When there is no family history of ovarian cancer, ovarian conservation appears to maximize survival among healthy women 40 to 65 years old who undergo hysterectomy for benign disease.46 Among healthy women hysterectomized before the age of 55, calculations suggest that 8.6% more would be alive at age 80 if their ovaries were conserved than if they were removed.46
A study from the Mayo Clinic found that all-cause mortality was significantly higher among women who underwent prophylactic bilateral oophorectomy before the age of 45 than it was among women in the control group (HR, 1.67; 95% CI, 1.16–2.40); it was particularly high in women who did not receive estrogen treatment before age 45 (HR, 1.93; 95% CI, 1.25–2.96).22
In a recent study, investigators used the Nurses’ Health Study database to explore the long-term health outcomes of 29,380 women who underwent hysterectomy.4 Of these women, 13,035 (44.4%) had their ovaries conserved, and 16,345 (55.6%) underwent bilateral oophorectomy. Follow-up was 24 years. Oophorectomy was associated with an increased risk of nonfatal CAD among all women (HR, 1.17; 95% CI, 1.02, 1.35), especially those who underwent the procedure before age 45 (HR, 1.26; 95% CI, 1.04, 1.54). Oophorectomy was associated with a markedly reduced risk of ovarian cancer but an increased risk of lung cancer (HR, 1.26; 95% CI, 1.02–1.56).
In regard to fatal events, oophorectomy increased the risk of death from all causes (HR, 1.12; 95% CI, 1.03, 1.21). Specifically, there was an increased risk of death from CAD (HR, 1.28; 95% CI, 1.00, 1.64), lung cancer (HR, 1.31; 95% CI, 1.02, 1.68), and all cancers (HR, 1.17; 95% CI, 1.04, 1.32). There was no overall difference in the risk of death from stroke, breast cancer, and colorectal cancer between women who underwent oophorectomy and those who retained their ovaries.
During the 24 years of follow-up, 37 women died from ovarian cancer, accounting for 1.2% of all deaths. At no age did oophorectomy show a survival benefit.
How this evidence should inform your practice
It is unfortunate that the entire body of evidence on the risks and benefits of bilateral salpingo-oophorectomy consists of observational studies, which have significant inherent limitations. Although the Nurses’ Health Study was the largest prospective study to examine the effect of oophorectomy on women’s health, and involved the longest follow-up, the study was observational, and oophorectomy and ovarian conservation were self-selected. Nevertheless, recent data suggest that a more detailed informed-consent process is warranted than the process in place. Informed consent should cover the risks and benefits of both oophorectomy and ovarian conservation.
Prophylactic oophorectomy is recommended only if a preponderance of the evidence establishes that it clearly benefits the patient. The studies described in this article suggest that bilateral oophorectomy does harm more often than it does good. Therefore, a cautious approach to oophorectomy at the time of hysterectomy is advised.
CASE RESOLVED
After you describe the risks and benefits of oophorectomy, and address the patient’s concerns about her family history of heart disease, she decides to keep her ovaries.
CASE: Hysterectomy candidate asks about her ovaries
A 51-year-old premenopausal woman complains of severe menorrhagia that often causes her to miss work. Although she is taking an iron supplement, her hemoglobin level often drops below 10 g/dL. She has already been identified as having fibroids, with a uterine size of 14 weeks. You order ultra-sonography, which reveals an enlarged uterus with multiple fibroids and normal endometrial thickness, but no intracavitary lesions.
After you describe the treatment options, including uterine artery embolization, the patient requests a hysterectomy as a reasonably low-risk means of cure. During informed consent, she asks whether she should have her ovaries removed during the surgery. Further discussion reveals that her father died of a myocardial infarction when he was 64 years old, but there is no family or personal history of ovarian or breast cancer.
How do you advise this patient, based on her history and recent findings from medical research?
Many gynecologists have been trained to recommend bilateral oophorectomy for women older than 45 or 50 years who request a hysterectomy for benign disease. In these women, oophorectomy is recommended to prevent ovarian cancer and avert the potential for other ovarian pathology that might require later surgery.
In the United States, 78% of women 45 to 64 years old and 55% of women overall undergo bilateral oophorectomy at the time of hysterectomy.1 These percentages mean that almost 300,000 women undergo bilateral oophorectomy each year.1
Hysterectomy alone can sometimes lead to early ovarian failure, but this phenomenon is infrequent. A prospective study of premenopausal women found that, after 5 years of follow-up, 20% of women who underwent simple hysterectomy reached menopause, compared with 7% of matched women who did not undergo hysterectomy.2
In this article, I explore the risks and benefits associated with bilateral oophorectomy and present an algorithm to aid in deciding whether the patient should keep her ovaries—and when oophorectomy might be a better option ( FIGURE ).
Among the hazards associated with bilateral oophorectomy are:
- an increased risk of death from coronary artery disease (CAD), lung cancer, all cancers (except ovarian), and all causes3,4
- an increased risk of osteoporosis and hip fracture5
- when performed before the onset of menopause, an increased risk of parkinsonism, cognitive impairment, dementia, anxiety, and depression.6-8
Benefits include a reduced risk of ovarian cancer, particularly among women who have a BRCA gene mutation or strong family history of ovarian or breast cancer.
Although ovarian cancer causes 15,000 deaths each year in the United States, that figure pales when compared with heart disease, which accounts for 350,000 deaths. In addition, hip fracture may cause approximately 66,000 deaths each year, and dementia attributable to bilateral oophorectomy may affect 100,000 to 200,000 women.9 Reoperation for adnexal pathology or pain after hysterectomy is rare, occurring in only 2.8% of women. Therefore, the benefits of oophorectomy are often outweighed by the risks of CAD, hip fracture, and neurologic conditions.
The assumption that medical treatment will ameliorate the risks associated with oophorectomy is unrealistic. Estrogen may mitigate some risks, but many women avoid hormone therapy. This avoidance can be especially problematic in young women.
In the Nurses’ Health Study, a separate analysis focused on women who had never used postmenopausal hormone therapy.4 In this analysis, all women who underwent bilateral oophorectomy had a greater risk of stroke (HR, 1.85; 95% CI, 1.09, 3.16) and lung cancer (HR, 2.09; 95% CI, 1.01, 4.33) than did women who retained their ovaries. Among women who underwent oophorectomy before age 50 and who did not take estrogen, the risk of coronary artery disease (CAD) was higher (HR, 1.98; 95% CI, 1.18, 3.32), as was the risk of death from all causes (HR, 1.40; 95% CI, 1.01, 1.96), compared with women who retained their ovaries.
Despite estrogen’s proven benefit among oophorectomized women, usage rates continue to decline. In the 6 months after publication of Women’s Health Initiative findings on estrogen–progestin therapy, the continuation rate of estrogen therapy decreased from 12.6% to 9.1%, and new starts also declined significantly.47
Among women who had a diagnosis of osteoporosis and who began treatment with estrogen, estrogen plus progestin, a bisphosphonate, or raloxifene, medication continuation rates were less than 25% at 12 months.48 Moreover, only 18% of women started on a statin to reduce the risk of cardiovascular disease were still taking the drug after 1 year.49

FIGURE Conservation vs oophorectomy: A guide to decision-making
* Estrogen replacement is recommended for women younger than 45 years who opt for oophorectomy
Ovarian cancer is a real, but relatively low, risk
In 2008, an estimated 21,650 new cases of ovarian cancer were diagnosed (age at diagnosis: mean, 63 years), and 15,520 women died from the disease.10 Because we lack a reliable screening test to detect early-stage ovarian cancer in the general population, most women are given a diagnosis when disease is advanced and the 5-year survival rate is 15% to 25%.
There is agreement that women who are known to have a BRCA mutation, which increases the risk of ovarian and breast cancer, should strongly consider oophorectomy once childbearing is complete.11 In the general population, however, the outlook is different.
In the United States, the lifetime risk of ovarian cancer is 1.4% overall. Among white women who have had three or more term pregnancies and who have used an oral contraceptive for at least 4 years, the lifetime risk of ovarian cancer drops to 0.3%.12
Need for reoperation is very low
The percentage of women who require reoperation after ovarian conservation—2.8%—may surprise you.13 That figure is lower than once thought because many studies were performed before asymptomatic, benign ovarian cysts were determined to be a fairly common phenomenon in postmenopausal women (prevalence, 6.6%). These cysts do not undergo transformation to cancer and, therefore, do not need to be removed.14
In addition, studies indicate that only 0.1% to 0.75% of women who retain their ovaries at the time of hysterectomy develop ovarian cancer.15,16 Therefore, the rationale of performing oophorectomy to avoid future surgery appears to be unfounded.
CAD risk rises sharply after oophorectomy
A recent systematic review found mixed evidence concerning the risk of CAD following bilateral salpingo-oophorectomy.17 In observational studies, however, earlier age of surgical or natural menopause has been associated with a higher risk of cardiovascular mortality.18-20 Early reports from the Nurses’ Health Study found that the risk of myocardial infarction doubled among women who underwent oophorectomy and never used estrogen, compared with age-matched premenopausal women (relative risk [RR], 2.2; 95% confidence interval [CI], 1.2, 4.2).3 Even after age 50, the risk of a first myocardial infarction is increased among oophorectomized women, compared with women who retain their ovaries (RR, 1.4; 95% CI, 1.0–2.0).21
A study by researchers from the Mayo Clinic, who examined all causes listed on the death certificate, found a significant association between bilateral oophorectomy before the age of 45 years and cardiovascular mortality (hazard ratio [HR], 1.44; 95% CI, 1.01–2.05).22 This risk was significantly increased among women who were not treated with estrogen through at least age 45, compared with estrogen-treated women.
Oophorectomy may impair bone health
After menopause, ovaries continue to produce significant amounts of the androgens testosterone and androstenedione, which are converted to estrone peripherally by skin, muscle, and fat cells.23,24 The levels of these hormones remain consistent and have been documented to age 80.25
Both estrogens and androgens inhibit bone resorption, and androgens also stimulate bone formation.26 Low levels of androgens and estrogens are linked to lower bone density and a higher risk of hip and vertebral fracture in postmenopausal women.27-29
Postmenopausal women who have been oophorectomized may have an even greater risk of osteoporosis. Over 16 years of follow-up, 340 women who had undergone oophorectomy at a median age of 62 years had 54% more osteoporotic fractures than women who had intact ovaries.5 Two other studies found no association between oophorectomy and bone loss or fracture risk, however.30,31
Hip fracture is a well-documented cause of increased morbidity and mortality in older women. One study found that, before hip fracture, 28% of patients were housebound; 1 year after hip fracture, the percentage was 46%.32 Women older than 60 who underwent oophorectomy had a doubled risk of mortality after low-trauma hip fracture, compared with women who had intact ovaries (odds ratio [OR], 2.18; 95% CI, 2.03–2.32).5
Loss of ovaries may affect mental health and sexuality
In a premenopausal woman, oophorectomy causes a sudden loss of estrogen and often triggers hot flashes, mood changes, sleep disturbances, headaches, and a decline in feelings of well-being.33,34 Over time, vaginal dryness, painful intercourse, loss of libido, bladder dysfunction, and depression may occur.35,36
Evidence suggests that, in women, sexual desire, sexual sensation, and orgasmic response are influenced by androgens. After elective oophorectomy, declines in sexual desire have been reported.37-39
Mental health and sexuality may rebound over time, however. One study found less improvement in mental health measures and body image 6 months after hysterectomy among women who were oophorectomized, compared with those who retained their ovaries. After 2 years, improvement levels were similar between groups.40
Cognitive function may suffer
Analysis of data from the Mayo Clinic Cohort Study of Oophorectomy and Aging found that bilateral oophorectomy before the onset of menopause increased the risk of parkinsonism, cognitive impairment or dementia, and anxiety or depression, particularly if estrogen was not replaced.6-8 These risks increased with younger age at oophorectomy.
The Women’s Health Initiative found an increased risk of dementia or mild cognitive impairment in women who were treated by estrogen alone or estrogen plus progestin after age 65.41-44
These disparate conclusions suggest that estrogen may have a protective effect on the brain if it is given right after natural menopause or premenopausal oophorectomy, but deleterious effects if it is started years later.45
Other studies of endogenous estrogen and cognitive function are few and yield inconsistent results.
Ovarian conservation boosts long-term survival
When there is no family history of ovarian cancer, ovarian conservation appears to maximize survival among healthy women 40 to 65 years old who undergo hysterectomy for benign disease.46 Among healthy women hysterectomized before the age of 55, calculations suggest that 8.6% more would be alive at age 80 if their ovaries were conserved than if they were removed.46
A study from the Mayo Clinic found that all-cause mortality was significantly higher among women who underwent prophylactic bilateral oophorectomy before the age of 45 than it was among women in the control group (HR, 1.67; 95% CI, 1.16–2.40); it was particularly high in women who did not receive estrogen treatment before age 45 (HR, 1.93; 95% CI, 1.25–2.96).22
In a recent study, investigators used the Nurses’ Health Study database to explore the long-term health outcomes of 29,380 women who underwent hysterectomy.4 Of these women, 13,035 (44.4%) had their ovaries conserved, and 16,345 (55.6%) underwent bilateral oophorectomy. Follow-up was 24 years. Oophorectomy was associated with an increased risk of nonfatal CAD among all women (HR, 1.17; 95% CI, 1.02, 1.35), especially those who underwent the procedure before age 45 (HR, 1.26; 95% CI, 1.04, 1.54). Oophorectomy was associated with a markedly reduced risk of ovarian cancer but an increased risk of lung cancer (HR, 1.26; 95% CI, 1.02–1.56).
In regard to fatal events, oophorectomy increased the risk of death from all causes (HR, 1.12; 95% CI, 1.03, 1.21). Specifically, there was an increased risk of death from CAD (HR, 1.28; 95% CI, 1.00, 1.64), lung cancer (HR, 1.31; 95% CI, 1.02, 1.68), and all cancers (HR, 1.17; 95% CI, 1.04, 1.32). There was no overall difference in the risk of death from stroke, breast cancer, and colorectal cancer between women who underwent oophorectomy and those who retained their ovaries.
During the 24 years of follow-up, 37 women died from ovarian cancer, accounting for 1.2% of all deaths. At no age did oophorectomy show a survival benefit.
How this evidence should inform your practice
It is unfortunate that the entire body of evidence on the risks and benefits of bilateral salpingo-oophorectomy consists of observational studies, which have significant inherent limitations. Although the Nurses’ Health Study was the largest prospective study to examine the effect of oophorectomy on women’s health, and involved the longest follow-up, the study was observational, and oophorectomy and ovarian conservation were self-selected. Nevertheless, recent data suggest that a more detailed informed-consent process is warranted than the process in place. Informed consent should cover the risks and benefits of both oophorectomy and ovarian conservation.
Prophylactic oophorectomy is recommended only if a preponderance of the evidence establishes that it clearly benefits the patient. The studies described in this article suggest that bilateral oophorectomy does harm more often than it does good. Therefore, a cautious approach to oophorectomy at the time of hysterectomy is advised.
CASE RESOLVED
After you describe the risks and benefits of oophorectomy, and address the patient’s concerns about her family history of heart disease, she decides to keep her ovaries.
1. Healthcare Cost and Utilization Project (HCUP), 1988–2001: A Federal–State–Industry Partnership in Health Data. Rockville, Md: Agency for Healthcare Research and Quality; July 2003.
2. Farquhar CM, Sadler L, Harvey SA, Stewart AW. The association of hysterectomy and menopause: a prospective cohort study. BJOG. 2005;112:956-962.
3. Colditz G, Willett W, Stampfer M, Rosner B, Speizer F, Hennekens C. Menopause and the risk of coronary heart disease in women. N Engl J Med. 1987;316:1105-1110.
4. Parker WH, Broder MS, Chang E, et al. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstet Gynecol. 2009;113:1027-1037.
5. Melton LJ 3rd, Khosla S, Malkasian GD, Achenbach SJ, Oberg AL, Riggs BL. Fracture risk after bilateral oophorectomy in elderly women. J Bone Miner Res. 2003;18:900-905.
6. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology. 2007;69:1074.-
7. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of parkinsonism in women who underwent oophorectomy before menopause. Neurology. 2008;70:200-209.
8. Rocca WA, Grossardt BR, Geda YE, et al. Long-term risk of depressive and anxiety symptoms after early bilateral oophorectomy. Menopause. 2008;15:1050-1059.
9. Bennett DA. Editorial comment on “Prevalence of dementia in the United States: the aging, demographics, and memory study” by Plassman et al. Neuroepidemiology. 2007;29:133-135.
10. Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71-96.
11. Armstrong K, Schwartz J, Randall T, Rubin S, Weber B. Hormone replacement therapy and life expectancy after prophylactic oophorectomy in women with BRCA1/2 mutations: a decision analysis. J Clin Oncol. 2004;22:1045-1054.
12. Hartge P, Whittemore AS, Itnyre J, McGowan L, Cramer D. Rates and risks of ovarian cancer in subgroups of white women in the United States. The Collaborative Ovarian Cancer Group. Obstet Gynecol. 1994;84:760-764.
13. Dekel A, Efrat Z, Orvieto R, et al. The residual ovary syndrome: a 20-year experience. Eur J Obstet Gynecol Reprod Biol. 1996;68:159-164.
14. Bailey CL, Ueland FR, Land GL, et al. The malignant potential of small cystic ovarian tumors in women over 50 years of age. Gynecol Oncol. 1998;69:3-7.
15. Naylor AC. Hysterectomy—analysis of 2,901 personally performed procedures. S Afr Med J. 1984;65:242-245.
16. Christ JE, Lotze EC. The residual ovary syndrome. Obstet Gynecol. 1975;46:551-556.
17. Jacoby VL, Grady D, Sawaya GF. Oophorectomy as a risk factor for coronary heart disease. Am J Obstet Gynecol. 2009;200:140.e1-140.e9.
18. van der Schouw YT, van der Graaf Y, Steyerberg EW, Eijkemans JC, Banga JD. Age at menopause as a risk factor for cardiovascular mortality. Lancet. 1996;347:714-718.
19. Løkkegaard E, Jovanovic Z, Heitmann BL, et al. The association between early menopause and risk of ischaemic heart disease: influence of hormone therapy. Maturitas. 2006;53:226-233.
20. de Kleijn MJJ, van der Schouw YT, Verbeek ALM, et al. Endogenous estrogen exposure and cardiovascular mortality risk in postmenopausal women. Am J Epidemiol. 2002;155:339.-
21. Falkeborn M, Schairer C, Naessen T, Persson I. Risk of myocardial infarction after oophorectomy and hysterectomy. J Clin Epidemiol. 2000;53:832-837.
22. Rocca WA, Grossardt BR, de Andrade M, Malkasian GD, Melton LJ, 3rd. Survival patterns after oophorectomy in premenopausal women: a population-based cohort study. Lancet Oncol. 2006;7:821-828.
23. Judd H, Judd G, Lucas W, Yen S. Endocrine function of the postmenopausal ovary: concentration of androgens and estrogens in ovarian and peripheral vein blood. J Clin Endocrinol Metab. 1974;39:1020-1024.
24. Fogle R, Stanczyk F, Zhang X, Paulson R. Ovarian androgen production in postmenopausal women. J Clin Endocrinol Metab. 2007;92:3040-3043.
25. Meldrum D, Davidson B, Tataryn I, Judd H. Changes in circulating steroids with aging in postmenopausal women. Obstet Gynecol. 1981;57:624-628.
26. Raisz LG, Wilta B, Artis A, et al. Comparison of the effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab. 1996;81:37-43.
27. Johansson C, Mellstrom D, Milsom I. Reproductive factors as predictors of bone density and fractures in women at age 70. Maturitas. 1993;17:39-50.
28. Davidson BJ, Ross RK, Paganini-Hill A, et al. Total and free estrogens and androgens in postmenopausal women with hip fractures. J Clin Endocrinol Metab. 1982;54:115-120.
29. Cummings SR, Browner WS, Bauer D, et al. Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N Engl J Med. 1998;339:733-738.
30. Kritz-Silverstein D, von Mühlen D, Barrett-Connor E. Hysterectomy and oophorectomy are unrelated to bone loss in older women. Maturitas. 2004;47:61-69.
31. Antoniucci DM, Sellmeyer DE, Cauley JA, et al. Postmenopausal bilateral oophorectomy is not associated with increased fracture risk in older women. J Bone Miner Res. 2005;20:741-747.
32. Keene GS, Parker MJ, Pryor GA. Mortality and morbidity after hip fractures. BMJ. 1993;307:1248-1250.
33. Nathorst-Boos J, von Schoultz B, Carlstrom K. Elective ovarian removal and estrogen replacement therapy—effects on sexual life, psychological well-being and androgen status. J Psychosom Obstet Gynaecol. 1993;14:283-293.
34. Nieman L. Management of surgically hypogonadal patients unable to take sex hormone replacement therapy. Endocrinol Metab Clin North Am. 2003;32:325-336.
35. Shifren J. Androgen deficiency in the oophorectomized woman. Fertil Steril. 2002;77 Suppl 4:s60-s62.
36. Sherwin B, Gelfan M. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med. 1978;49:397-409.
37. Shifren JL, Avis NE. Surgical menopause: effects on psychological well-being and sexuality. Menopause. 2007;14:586.-
38. Elit L, Esplen MJ, Butler K, Narod S. Quality of life and psychosexual adjustment after prophylactic oophorectomy for a family history of ovarian cancer. Fam Cancer. 2001;1:149-156.
39. Sherwin BB, Gelfan MM. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med 1978;49:397-409.
40. Teplin V, Vittinghoff E, Lin F, Learman L, Richter H, Kuppermann M. Oophorectomy in premenopausal women: health-related quality of life and sexual functioning. Obstet Gynecol. 2007;109:347-354.
41. Shumaker SA, Legault C, Rapp SR, et al. WHIMS Investigators Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2651-2662.
42. Rapp SR, Espeland MA, Shumaker SA, et al. WHIMS Investigators. Effect of estrogen plus progestin on global cognitive function in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2663-2672.
43. Espeland MA, Rapp SR, Shumaker SA, et al. Conjugated equine estrogens and global cognitive function in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2959-2968.
44. Shumaker SA, Legault C, Kuller L, et al. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2947-2958.
45. Siegfried T. Neuroscience: it’s all in the timing. Nature. 2007;445:359-361.
46. Parker W, Broder M, Liu Z, Shoupe D, Farquhar C, Berek J. Ovarian conservation at the time of hysterectomy for benign disease. Obstet Gynecol. 2005;106:219-226.
47. Buist DS, Newton KM, Miglioretti DL, et al. Hormone therapy prescribing patterns in the United States. Obstet Gynecol. 2004;104:1042-1050.
48. McCombs JS, Thiebaud P, McLaughlin-Miley C, Shi J. Compliance with drug therapies for the treatment and prevention of osteoporosis. Maturitas. 2004;48:271-287.
49. Huser MA, Evans TS, Berger V. Medication adherence trends with statins. Adv Ther. 2005;22:163-171.
1. Healthcare Cost and Utilization Project (HCUP), 1988–2001: A Federal–State–Industry Partnership in Health Data. Rockville, Md: Agency for Healthcare Research and Quality; July 2003.
2. Farquhar CM, Sadler L, Harvey SA, Stewart AW. The association of hysterectomy and menopause: a prospective cohort study. BJOG. 2005;112:956-962.
3. Colditz G, Willett W, Stampfer M, Rosner B, Speizer F, Hennekens C. Menopause and the risk of coronary heart disease in women. N Engl J Med. 1987;316:1105-1110.
4. Parker WH, Broder MS, Chang E, et al. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstet Gynecol. 2009;113:1027-1037.
5. Melton LJ 3rd, Khosla S, Malkasian GD, Achenbach SJ, Oberg AL, Riggs BL. Fracture risk after bilateral oophorectomy in elderly women. J Bone Miner Res. 2003;18:900-905.
6. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology. 2007;69:1074.-
7. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of parkinsonism in women who underwent oophorectomy before menopause. Neurology. 2008;70:200-209.
8. Rocca WA, Grossardt BR, Geda YE, et al. Long-term risk of depressive and anxiety symptoms after early bilateral oophorectomy. Menopause. 2008;15:1050-1059.
9. Bennett DA. Editorial comment on “Prevalence of dementia in the United States: the aging, demographics, and memory study” by Plassman et al. Neuroepidemiology. 2007;29:133-135.
10. Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71-96.
11. Armstrong K, Schwartz J, Randall T, Rubin S, Weber B. Hormone replacement therapy and life expectancy after prophylactic oophorectomy in women with BRCA1/2 mutations: a decision analysis. J Clin Oncol. 2004;22:1045-1054.
12. Hartge P, Whittemore AS, Itnyre J, McGowan L, Cramer D. Rates and risks of ovarian cancer in subgroups of white women in the United States. The Collaborative Ovarian Cancer Group. Obstet Gynecol. 1994;84:760-764.
13. Dekel A, Efrat Z, Orvieto R, et al. The residual ovary syndrome: a 20-year experience. Eur J Obstet Gynecol Reprod Biol. 1996;68:159-164.
14. Bailey CL, Ueland FR, Land GL, et al. The malignant potential of small cystic ovarian tumors in women over 50 years of age. Gynecol Oncol. 1998;69:3-7.
15. Naylor AC. Hysterectomy—analysis of 2,901 personally performed procedures. S Afr Med J. 1984;65:242-245.
16. Christ JE, Lotze EC. The residual ovary syndrome. Obstet Gynecol. 1975;46:551-556.
17. Jacoby VL, Grady D, Sawaya GF. Oophorectomy as a risk factor for coronary heart disease. Am J Obstet Gynecol. 2009;200:140.e1-140.e9.
18. van der Schouw YT, van der Graaf Y, Steyerberg EW, Eijkemans JC, Banga JD. Age at menopause as a risk factor for cardiovascular mortality. Lancet. 1996;347:714-718.
19. Løkkegaard E, Jovanovic Z, Heitmann BL, et al. The association between early menopause and risk of ischaemic heart disease: influence of hormone therapy. Maturitas. 2006;53:226-233.
20. de Kleijn MJJ, van der Schouw YT, Verbeek ALM, et al. Endogenous estrogen exposure and cardiovascular mortality risk in postmenopausal women. Am J Epidemiol. 2002;155:339.-
21. Falkeborn M, Schairer C, Naessen T, Persson I. Risk of myocardial infarction after oophorectomy and hysterectomy. J Clin Epidemiol. 2000;53:832-837.
22. Rocca WA, Grossardt BR, de Andrade M, Malkasian GD, Melton LJ, 3rd. Survival patterns after oophorectomy in premenopausal women: a population-based cohort study. Lancet Oncol. 2006;7:821-828.
23. Judd H, Judd G, Lucas W, Yen S. Endocrine function of the postmenopausal ovary: concentration of androgens and estrogens in ovarian and peripheral vein blood. J Clin Endocrinol Metab. 1974;39:1020-1024.
24. Fogle R, Stanczyk F, Zhang X, Paulson R. Ovarian androgen production in postmenopausal women. J Clin Endocrinol Metab. 2007;92:3040-3043.
25. Meldrum D, Davidson B, Tataryn I, Judd H. Changes in circulating steroids with aging in postmenopausal women. Obstet Gynecol. 1981;57:624-628.
26. Raisz LG, Wilta B, Artis A, et al. Comparison of the effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab. 1996;81:37-43.
27. Johansson C, Mellstrom D, Milsom I. Reproductive factors as predictors of bone density and fractures in women at age 70. Maturitas. 1993;17:39-50.
28. Davidson BJ, Ross RK, Paganini-Hill A, et al. Total and free estrogens and androgens in postmenopausal women with hip fractures. J Clin Endocrinol Metab. 1982;54:115-120.
29. Cummings SR, Browner WS, Bauer D, et al. Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N Engl J Med. 1998;339:733-738.
30. Kritz-Silverstein D, von Mühlen D, Barrett-Connor E. Hysterectomy and oophorectomy are unrelated to bone loss in older women. Maturitas. 2004;47:61-69.
31. Antoniucci DM, Sellmeyer DE, Cauley JA, et al. Postmenopausal bilateral oophorectomy is not associated with increased fracture risk in older women. J Bone Miner Res. 2005;20:741-747.
32. Keene GS, Parker MJ, Pryor GA. Mortality and morbidity after hip fractures. BMJ. 1993;307:1248-1250.
33. Nathorst-Boos J, von Schoultz B, Carlstrom K. Elective ovarian removal and estrogen replacement therapy—effects on sexual life, psychological well-being and androgen status. J Psychosom Obstet Gynaecol. 1993;14:283-293.
34. Nieman L. Management of surgically hypogonadal patients unable to take sex hormone replacement therapy. Endocrinol Metab Clin North Am. 2003;32:325-336.
35. Shifren J. Androgen deficiency in the oophorectomized woman. Fertil Steril. 2002;77 Suppl 4:s60-s62.
36. Sherwin B, Gelfan M. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med. 1978;49:397-409.
37. Shifren JL, Avis NE. Surgical menopause: effects on psychological well-being and sexuality. Menopause. 2007;14:586.-
38. Elit L, Esplen MJ, Butler K, Narod S. Quality of life and psychosexual adjustment after prophylactic oophorectomy for a family history of ovarian cancer. Fam Cancer. 2001;1:149-156.
39. Sherwin BB, Gelfan MM. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med 1978;49:397-409.
40. Teplin V, Vittinghoff E, Lin F, Learman L, Richter H, Kuppermann M. Oophorectomy in premenopausal women: health-related quality of life and sexual functioning. Obstet Gynecol. 2007;109:347-354.
41. Shumaker SA, Legault C, Rapp SR, et al. WHIMS Investigators Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2651-2662.
42. Rapp SR, Espeland MA, Shumaker SA, et al. WHIMS Investigators. Effect of estrogen plus progestin on global cognitive function in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2663-2672.
43. Espeland MA, Rapp SR, Shumaker SA, et al. Conjugated equine estrogens and global cognitive function in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2959-2968.
44. Shumaker SA, Legault C, Kuller L, et al. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2947-2958.
45. Siegfried T. Neuroscience: it’s all in the timing. Nature. 2007;445:359-361.
46. Parker W, Broder M, Liu Z, Shoupe D, Farquhar C, Berek J. Ovarian conservation at the time of hysterectomy for benign disease. Obstet Gynecol. 2005;106:219-226.
47. Buist DS, Newton KM, Miglioretti DL, et al. Hormone therapy prescribing patterns in the United States. Obstet Gynecol. 2004;104:1042-1050.
48. McCombs JS, Thiebaud P, McLaughlin-Miley C, Shi J. Compliance with drug therapies for the treatment and prevention of osteoporosis. Maturitas. 2004;48:271-287.
49. Huser MA, Evans TS, Berger V. Medication adherence trends with statins. Adv Ther. 2005;22:163-171.
PELVIC FLOOR DYSFUNCTION
The authors report no financial relationships relevant to this article.
Overactive bladder (OAB)—urinary urgency, with or without incontinence, usually with frequency and nocturia1—is a common problem among women who seek care from an ObGyn. In fact, the condition is estimated to carry a health-care cost in excess of $12 billion annually in the United States.2
A recent community-based survey in Norway estimated the prevalence of urinary incontinence there to be 27% in women between the ages of 65 and 69 years and 35% to 40% in those 80 years or older.3 A population-based study in the United States suggested an even higher rate of urinary incontinence here: greater than 50% in women 60 years or older, with 1) urge urinary incontinence (UUI) predominating4 and 2) the prevalence particularly high among older women who are homebound or who live in a long-term care facility.5
OAB can undermine quality of life in several ways: social isolation, anxiety, poor sleep, higher risk of fracture after a fall,6 reduced ability to function, and poor self-perception. Despite these harmful effects, many women delay seeking care for OAB because they are embarrassed to talk about it with their physician.
Treatment by generalists is feasible—but there is a catch
It’s possible to treat most patients with OAB without referral to a specialist. Two common concerns, however, may set up a roadblock to successful management: the adverse effects associated with some agents and suboptimal control of symptoms.
In this Update, we review recent findings about 1) the potential that anticholinergic therapy has for impairing cognitive function in the older population of women and 2) the important role that concomitant behavioral therapy plays in the long-term success of, and patients’ satisfaction with, treatment of OAB.
Behavioral therapy for OAB: Is it worth all the effort?
Burgio KL, Locher JL, Goode PS. Combined behavioral and drug therapy for urge incontinence in older women. J Am Geriatr Soc. 2000;48:370–374.
The authors of this article followed a randomized clinical trial of older women that compared behavioral and drug therapy for OAB. In the trial, biofeedback-assisted behavioral training (comprising anorectal biofeedback, urge strategies, pelvic muscle biofeedback, and practitioner-directed review with optimization) was compared with treatment with oxybutynin, between 2.5 and 15 mg/day. Both biofeedback-assisted behavioral therapy and the drug regimen were found effective, although neither treatment provided an entirely satisfactory result for all patients. (For a brief description of what constitutes behavioral treatment, see “6 tenets of behavioral therapy for urge urinary incontinence.”)
Second phase of the trial. To determine if treatment satisfaction could be enhanced, the investigators performed a modified crossover study to determine whether combination therapy—biofeedback-assisted behavioral training plus oxybutynin—added any benefit over treatment with behavioral therapy or drug therapy alone. Eligibility was determined by age (55 years or older), demonstrated UUI for at least 3 months, and incomplete dryness or incomplete satisfaction with the outcome of 8 weeks of single-intervention treatment (with either treatment) during the initial phase of the trial.
This subgroup was offered an additional 8 weeks of combination therapy. The primary outcome measure was a reduction in the frequency of episodes of incontinence episodes as recorded by subjects in a bladder diary.
Of 197 women who participated in the original randomized clinical trial, 35—27 who completed drug therapy and 8 who completed behavioral treatment—elected to receive combination therapy. Those 35 subjects did not differ in any of the multiple baseline variables; mean age was 69.3 years (standard deviation [SD], ±7.9 years).
Among subjects originally assigned to behavioral therapy alone, overall reduction in incontinence increased from a mean of 57.5% to a mean of 88.5% after combined therapy (P=.034). Subjects originally assigned to drug therapy alone demonstrated an improvement from 72.7% reduction in incontinence to a mean 84.3% overall reduction with combined therapy (P=.001).
These data suggest that combined therapy can be more effective than behavioral therapy or drug therapy alone. The impact of this study is limited, however, by the relatively low percentage (12.7%) of patients who had received behavioral therapy and chose to add drug therapy, compared with the 41.5% who moved from drug therapy alone to add behavioral therapy.
Furthermore, subjects were self-selected: They chose to continue with an additional 8 weeks of therapy after their initial suboptimal outcome. It is possible that some subjects who were neither totally continent nor completely satisfied with initial therapy chose not to continue with the crossover segment of the trial because it posed too great a burden or because they were discouraged with the initial degree of improvement.
Generalizing these results to all older women with UUI is difficult. The authors point out, however, that, in practice, patients may be more likely than not to choose combination therapy in the hope of shortening the duration of medical therapy. Although it isn’t known whether providing combination therapy from the outset would have yielded better outcomes than either single therapy did, the authors hypothesize that initial combination therapy may result in greater improvement because patients have a high level of motivation and expectation of improvement at the beginning of treatment.
Importance of this article. The investigators demonstrated that a combination of behavioral and drug therapies can provide increased effectiveness in patients for whom each treatment alone led to suboptimal satisfaction. Furthermore, by targeting women older than 55 years, the investigators were able to demonstrate this effectiveness in a group for whom pelvic-floor training may be more difficult than it is for younger women.
It will be interesting to see if future research will 1) validate these findings and 2) determine whether combined therapy can reduce the duration of drug therapy in this older population through behavioral modification and pelvic floor reeducation.
Fluid management
This first-step therapy can involve providing a handout to the patient that details techniques she can use to monitor and control her fluid intake in a manner that addresses her problem. Among such steps:
- avoiding caffeine and artificial sweeteners
- tracking her diet to identify any other bladder irritants
- limiting fluids before times she is more likely to be incontinent—during a long drive, for example, or, in the case of nocturia, after the evening meal.
Scheduled voiding
With scheduled, or prompted, voiding, the patient empties her bladder at a set interval—usually, every 1.5 to 2 hours. If nocturia, or the more severe enuresis, is a problem, the patient can be prompted by an alarm clock or (if she is institutionalized) by nursing staff. Combining scheduled voiding with fluid management principles helps the patient avoid reaching a bladder volume at which an episode of incontinence becomes more likely.
Bladder training
This is a modification of scheduled voiding that attempts to establish a normal voiding interval in patients who have significant frequency but a small voided volume. It imposes a regimented voiding schedule that gradually (over 7 to 10 days) extends the duration between voids.
Pelvic floor-muscle exercises
The focus here is on using pelvic-floor muscles to prevent incontinence. The muscles are strengthened by having the patient perform Kegel exercises (named for Arnold H. Kegel, MD, who, in 1948, recognized the role of pelvic floor-muscle rehabilitation in the treatment of incontinence). The exercises involve simultaneous 1) contraction of the pelvic and periurethral musculature and 2) relaxation of other muscles, including abdominal muscles, which can increase pressure on the bladder.
Once the patient learns to perform Kegel exercises, she can use them to suppress urgency: Instead of hurrying to the bathroom when urgency arises, she is encouraged to sit down, relax, and contract the pelvic-floor muscles repeatedly until the urge to void diminishes. Once it does, the patient proceeds to the toilet to void normally.
Pelvic exam
By self-exam, the patient can identify and familiarize herself with her purposeful contractions of the pelvic-floor musculature and thereby strengthen those muscles with effective exercise.
Biofeedback
Direct feedback about contractions of the pelvic-floor muscles—by a display of data on a gauge or computer monitor, gathered using an intravaginal or anorectal sensor or probe—allows a patient who is exercising those muscles to better target her efforts and maximize their effectiveness.
Combining behavioral therapy and an anticholinergic medication for urge urinary incontinence may yield a superior result after either modality alone has been disappointing by the patient’s account of success.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
Does oxybutynin for UUI further erode cognition in elderly women who are cognitively impaired?
Lackner TE, Wyman JF, McCarthy TC, Monigold M, Davey C. Randomized, placebo-controlled trial of the cognitive effect, safety, and tolerability of oral extended-release oxybutynin in cognitively impaired nursing home residents with urge urinary incontinence. J Am Geriatr Soc. 2008;56:862–870.
Although anticholinergic therapy is modestly effective against UUI in nursing home residents, past studies have suggested that such treatment can impair, or further impair, cognition in this population—a concern that may lead to underuse. This double-blinded, randomized, placebo-controlled trial compared short-term oral extended-release oxybutynin with placebo.
Consequently, the authors sought to determine the cognitive effect, safety, and tolerability of 5 mg/day oral extended-release oxybutynin (the most commonly prescribed dosage) in cognitively impaired older nursing home residents who have UUI.
Subjects were eligible if they:
- were 65 years or older
- had UUI
- lived in a nursing home longer than 3 months
- had cognitive impairment.
Women already being treated for urinary incontinence, those who had an indwelling Foley catheter or urinary retention, and those who were bed-bound or incommunicative were excluded.
Fifty women, mean age 88.6 years (SD, ±6.2), from 12 nursing home facilities, agreed to participate. They were further stratified based on the score of a Mini-Mental State Exam (MMSE): 13 had severe cognitive impairment (MMSE score, 5–10) and 37 had mild or moderate impairment (score, 11–23).
Subjects were randomized to 4 weeks’ treatment with either 5 mg/day oral extended-release oxybutynin or one placebo tablet daily. A nurse practitioner who was blinded to randomization collected all data. The Confusion Assessment Method (CAM) algorithm, MMSE, and Severe Impairment Battery (SIB) were used to assess cognitive decline. The Brief Agitation Rating Scale (BARS) assessed agitation.
No baseline differences were noted with regard to: age; demographic, functional, and neuropsychiatric characteristics; clinical factors predisposing to delirium; and serum anticholinergic activity. Adherence was similar in the treatment (97%) and placebo (97.4%) groups.
Finding: Cognitive impairment. Treatment and placebo groups in the baseline mild-or-moderate stratum (by MMSE) showed equivalent mean changes in CAM scores at all time points. Because of the small sample size, however, CAM score equivalence could not be definitively determined for the groups in the severe impairment stratum. Evaluation of mean MMSE and BARS scores showed no significant changes between groups.
Finding: Tolerability. Excellent tolerability was noted in the treatment group: 96% of subjects completed the trial (compared with 92% of the placebo group). No difference in the rate of adverse events was noted between treatment and placebo groups; of adverse events recorded, 90% were judged “mild” by the investigators. Constipation and dry mouth were most common.
Finding: Falls. More than half—54%—of subjects in both groups experienced at least one fall during the trial or during the preceding or following 3 months. Despite this, no difference in the rate of falls between the treatment and placebo groups was noted. Furthermore, regression analysis revealed no treatment or period effect on falls per month across the time of observation.
Conclusions. Treatment with 5 mg/day oral extended-release oxybutynin in older patients with some cognitive impairment is well tolerated, the study’s findings suggest, with minimal risk of further cognitive decline or delirium over the short term. The potential that long-term therapy has to harm cognitive function remains, however; data on long-term treatment are needed to illuminate that area.
The authors also address the importance of dosing, especially over time, and discuss the lower potential of newer-generation anticholinergics to produce cognitive impairment.
A limited number of articles in the medical literature address anticholinergics in an older population, specifically, and only a few of those evaluated the effects of the drugs on cognitive function. By investigating patients who had an existing cognitive impairment, the authors of this article were able to target a cohort at risk of further cognitive impairment from medication use—thereby giving further weight to their findings of no significant effect.
Main strengths and limitations of the study. The investigators used validated, standardized cognitive tests that were administered by a uniform blinded evaluator in a randomized, controlled trial. The study was limited, however, because patients were evaluated only over a relatively short period (1 month) and because the efficacy of therapy was not addressed.
Further studies of anticholinergic medications, using the same rigorous scientific approach that these investigators applied, are needed to address 1) the long-term efficacy of oxybutynin and similar agents and 2) the cognitive effects of long-term treatment in this older population.
Further impairment is unlikely over the short term when a cognitively impaired nursing home patient who has urge urinary incontinence is treated with 5 mg/day oral extended-release oxybutynin.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
1. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Am J Obstet Gynecol. 2002;187:116-126.
2. Hu TW, Wagner TH, Bentkover JD, et al. Estimated economic costs of overactive bladder in the United States. Urology. 2003;61:1123-1128.
3. Hannestad YS, Rortveit G, Sandvik H, Hunskaar S. A community-based epidemiological survey of female urinary incontinence: the Norwegian EPINCONT study. Epidemiology of Incontinence in the County of Nord-Trøndelag. J Clin Epidemiol. 2000;53:1150-1157.
4. Melville JL, Katon W, Delaney K, Newton K. Urinary incontinence in US women: a population-based study. Arch Intern Med. 2005;165:537-542.
5. Fantl JA, Newman DK, Colling J, et al. Managing Acute and Chronic Urinary Incontinence. Clinical Practice Guideline. Quick Reference Guide for Clinicians, No. 2, 1996 Update. Rockville, MD: US Department of Health and Human Services, Public Health Service, Agency for Health Care Policy and Research. AHCPR Pub. No. 96-0686. January 1996. Available at: http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=hstat6.chapter.32554. Accessed September 11, 2009.
6. Brown JS, Vittinghoff E, Wyman JF, et al. Urinary incontinence: does it increase risk for falls and fractures? Study of Osteoporotic Fractures Research Group. J Am Geriatr Soc. 2000;48:721-725.
The authors report no financial relationships relevant to this article.
Overactive bladder (OAB)—urinary urgency, with or without incontinence, usually with frequency and nocturia1—is a common problem among women who seek care from an ObGyn. In fact, the condition is estimated to carry a health-care cost in excess of $12 billion annually in the United States.2
A recent community-based survey in Norway estimated the prevalence of urinary incontinence there to be 27% in women between the ages of 65 and 69 years and 35% to 40% in those 80 years or older.3 A population-based study in the United States suggested an even higher rate of urinary incontinence here: greater than 50% in women 60 years or older, with 1) urge urinary incontinence (UUI) predominating4 and 2) the prevalence particularly high among older women who are homebound or who live in a long-term care facility.5
OAB can undermine quality of life in several ways: social isolation, anxiety, poor sleep, higher risk of fracture after a fall,6 reduced ability to function, and poor self-perception. Despite these harmful effects, many women delay seeking care for OAB because they are embarrassed to talk about it with their physician.
Treatment by generalists is feasible—but there is a catch
It’s possible to treat most patients with OAB without referral to a specialist. Two common concerns, however, may set up a roadblock to successful management: the adverse effects associated with some agents and suboptimal control of symptoms.
In this Update, we review recent findings about 1) the potential that anticholinergic therapy has for impairing cognitive function in the older population of women and 2) the important role that concomitant behavioral therapy plays in the long-term success of, and patients’ satisfaction with, treatment of OAB.
Behavioral therapy for OAB: Is it worth all the effort?
Burgio KL, Locher JL, Goode PS. Combined behavioral and drug therapy for urge incontinence in older women. J Am Geriatr Soc. 2000;48:370–374.
The authors of this article followed a randomized clinical trial of older women that compared behavioral and drug therapy for OAB. In the trial, biofeedback-assisted behavioral training (comprising anorectal biofeedback, urge strategies, pelvic muscle biofeedback, and practitioner-directed review with optimization) was compared with treatment with oxybutynin, between 2.5 and 15 mg/day. Both biofeedback-assisted behavioral therapy and the drug regimen were found effective, although neither treatment provided an entirely satisfactory result for all patients. (For a brief description of what constitutes behavioral treatment, see “6 tenets of behavioral therapy for urge urinary incontinence.”)
Second phase of the trial. To determine if treatment satisfaction could be enhanced, the investigators performed a modified crossover study to determine whether combination therapy—biofeedback-assisted behavioral training plus oxybutynin—added any benefit over treatment with behavioral therapy or drug therapy alone. Eligibility was determined by age (55 years or older), demonstrated UUI for at least 3 months, and incomplete dryness or incomplete satisfaction with the outcome of 8 weeks of single-intervention treatment (with either treatment) during the initial phase of the trial.
This subgroup was offered an additional 8 weeks of combination therapy. The primary outcome measure was a reduction in the frequency of episodes of incontinence episodes as recorded by subjects in a bladder diary.
Of 197 women who participated in the original randomized clinical trial, 35—27 who completed drug therapy and 8 who completed behavioral treatment—elected to receive combination therapy. Those 35 subjects did not differ in any of the multiple baseline variables; mean age was 69.3 years (standard deviation [SD], ±7.9 years).
Among subjects originally assigned to behavioral therapy alone, overall reduction in incontinence increased from a mean of 57.5% to a mean of 88.5% after combined therapy (P=.034). Subjects originally assigned to drug therapy alone demonstrated an improvement from 72.7% reduction in incontinence to a mean 84.3% overall reduction with combined therapy (P=.001).
These data suggest that combined therapy can be more effective than behavioral therapy or drug therapy alone. The impact of this study is limited, however, by the relatively low percentage (12.7%) of patients who had received behavioral therapy and chose to add drug therapy, compared with the 41.5% who moved from drug therapy alone to add behavioral therapy.
Furthermore, subjects were self-selected: They chose to continue with an additional 8 weeks of therapy after their initial suboptimal outcome. It is possible that some subjects who were neither totally continent nor completely satisfied with initial therapy chose not to continue with the crossover segment of the trial because it posed too great a burden or because they were discouraged with the initial degree of improvement.
Generalizing these results to all older women with UUI is difficult. The authors point out, however, that, in practice, patients may be more likely than not to choose combination therapy in the hope of shortening the duration of medical therapy. Although it isn’t known whether providing combination therapy from the outset would have yielded better outcomes than either single therapy did, the authors hypothesize that initial combination therapy may result in greater improvement because patients have a high level of motivation and expectation of improvement at the beginning of treatment.
Importance of this article. The investigators demonstrated that a combination of behavioral and drug therapies can provide increased effectiveness in patients for whom each treatment alone led to suboptimal satisfaction. Furthermore, by targeting women older than 55 years, the investigators were able to demonstrate this effectiveness in a group for whom pelvic-floor training may be more difficult than it is for younger women.
It will be interesting to see if future research will 1) validate these findings and 2) determine whether combined therapy can reduce the duration of drug therapy in this older population through behavioral modification and pelvic floor reeducation.
Fluid management
This first-step therapy can involve providing a handout to the patient that details techniques she can use to monitor and control her fluid intake in a manner that addresses her problem. Among such steps:
- avoiding caffeine and artificial sweeteners
- tracking her diet to identify any other bladder irritants
- limiting fluids before times she is more likely to be incontinent—during a long drive, for example, or, in the case of nocturia, after the evening meal.
Scheduled voiding
With scheduled, or prompted, voiding, the patient empties her bladder at a set interval—usually, every 1.5 to 2 hours. If nocturia, or the more severe enuresis, is a problem, the patient can be prompted by an alarm clock or (if she is institutionalized) by nursing staff. Combining scheduled voiding with fluid management principles helps the patient avoid reaching a bladder volume at which an episode of incontinence becomes more likely.
Bladder training
This is a modification of scheduled voiding that attempts to establish a normal voiding interval in patients who have significant frequency but a small voided volume. It imposes a regimented voiding schedule that gradually (over 7 to 10 days) extends the duration between voids.
Pelvic floor-muscle exercises
The focus here is on using pelvic-floor muscles to prevent incontinence. The muscles are strengthened by having the patient perform Kegel exercises (named for Arnold H. Kegel, MD, who, in 1948, recognized the role of pelvic floor-muscle rehabilitation in the treatment of incontinence). The exercises involve simultaneous 1) contraction of the pelvic and periurethral musculature and 2) relaxation of other muscles, including abdominal muscles, which can increase pressure on the bladder.
Once the patient learns to perform Kegel exercises, she can use them to suppress urgency: Instead of hurrying to the bathroom when urgency arises, she is encouraged to sit down, relax, and contract the pelvic-floor muscles repeatedly until the urge to void diminishes. Once it does, the patient proceeds to the toilet to void normally.
Pelvic exam
By self-exam, the patient can identify and familiarize herself with her purposeful contractions of the pelvic-floor musculature and thereby strengthen those muscles with effective exercise.
Biofeedback
Direct feedback about contractions of the pelvic-floor muscles—by a display of data on a gauge or computer monitor, gathered using an intravaginal or anorectal sensor or probe—allows a patient who is exercising those muscles to better target her efforts and maximize their effectiveness.
Combining behavioral therapy and an anticholinergic medication for urge urinary incontinence may yield a superior result after either modality alone has been disappointing by the patient’s account of success.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
Does oxybutynin for UUI further erode cognition in elderly women who are cognitively impaired?
Lackner TE, Wyman JF, McCarthy TC, Monigold M, Davey C. Randomized, placebo-controlled trial of the cognitive effect, safety, and tolerability of oral extended-release oxybutynin in cognitively impaired nursing home residents with urge urinary incontinence. J Am Geriatr Soc. 2008;56:862–870.
Although anticholinergic therapy is modestly effective against UUI in nursing home residents, past studies have suggested that such treatment can impair, or further impair, cognition in this population—a concern that may lead to underuse. This double-blinded, randomized, placebo-controlled trial compared short-term oral extended-release oxybutynin with placebo.
Consequently, the authors sought to determine the cognitive effect, safety, and tolerability of 5 mg/day oral extended-release oxybutynin (the most commonly prescribed dosage) in cognitively impaired older nursing home residents who have UUI.
Subjects were eligible if they:
- were 65 years or older
- had UUI
- lived in a nursing home longer than 3 months
- had cognitive impairment.
Women already being treated for urinary incontinence, those who had an indwelling Foley catheter or urinary retention, and those who were bed-bound or incommunicative were excluded.
Fifty women, mean age 88.6 years (SD, ±6.2), from 12 nursing home facilities, agreed to participate. They were further stratified based on the score of a Mini-Mental State Exam (MMSE): 13 had severe cognitive impairment (MMSE score, 5–10) and 37 had mild or moderate impairment (score, 11–23).
Subjects were randomized to 4 weeks’ treatment with either 5 mg/day oral extended-release oxybutynin or one placebo tablet daily. A nurse practitioner who was blinded to randomization collected all data. The Confusion Assessment Method (CAM) algorithm, MMSE, and Severe Impairment Battery (SIB) were used to assess cognitive decline. The Brief Agitation Rating Scale (BARS) assessed agitation.
No baseline differences were noted with regard to: age; demographic, functional, and neuropsychiatric characteristics; clinical factors predisposing to delirium; and serum anticholinergic activity. Adherence was similar in the treatment (97%) and placebo (97.4%) groups.
Finding: Cognitive impairment. Treatment and placebo groups in the baseline mild-or-moderate stratum (by MMSE) showed equivalent mean changes in CAM scores at all time points. Because of the small sample size, however, CAM score equivalence could not be definitively determined for the groups in the severe impairment stratum. Evaluation of mean MMSE and BARS scores showed no significant changes between groups.
Finding: Tolerability. Excellent tolerability was noted in the treatment group: 96% of subjects completed the trial (compared with 92% of the placebo group). No difference in the rate of adverse events was noted between treatment and placebo groups; of adverse events recorded, 90% were judged “mild” by the investigators. Constipation and dry mouth were most common.
Finding: Falls. More than half—54%—of subjects in both groups experienced at least one fall during the trial or during the preceding or following 3 months. Despite this, no difference in the rate of falls between the treatment and placebo groups was noted. Furthermore, regression analysis revealed no treatment or period effect on falls per month across the time of observation.
Conclusions. Treatment with 5 mg/day oral extended-release oxybutynin in older patients with some cognitive impairment is well tolerated, the study’s findings suggest, with minimal risk of further cognitive decline or delirium over the short term. The potential that long-term therapy has to harm cognitive function remains, however; data on long-term treatment are needed to illuminate that area.
The authors also address the importance of dosing, especially over time, and discuss the lower potential of newer-generation anticholinergics to produce cognitive impairment.
A limited number of articles in the medical literature address anticholinergics in an older population, specifically, and only a few of those evaluated the effects of the drugs on cognitive function. By investigating patients who had an existing cognitive impairment, the authors of this article were able to target a cohort at risk of further cognitive impairment from medication use—thereby giving further weight to their findings of no significant effect.
Main strengths and limitations of the study. The investigators used validated, standardized cognitive tests that were administered by a uniform blinded evaluator in a randomized, controlled trial. The study was limited, however, because patients were evaluated only over a relatively short period (1 month) and because the efficacy of therapy was not addressed.
Further studies of anticholinergic medications, using the same rigorous scientific approach that these investigators applied, are needed to address 1) the long-term efficacy of oxybutynin and similar agents and 2) the cognitive effects of long-term treatment in this older population.
Further impairment is unlikely over the short term when a cognitively impaired nursing home patient who has urge urinary incontinence is treated with 5 mg/day oral extended-release oxybutynin.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
The authors report no financial relationships relevant to this article.
Overactive bladder (OAB)—urinary urgency, with or without incontinence, usually with frequency and nocturia1—is a common problem among women who seek care from an ObGyn. In fact, the condition is estimated to carry a health-care cost in excess of $12 billion annually in the United States.2
A recent community-based survey in Norway estimated the prevalence of urinary incontinence there to be 27% in women between the ages of 65 and 69 years and 35% to 40% in those 80 years or older.3 A population-based study in the United States suggested an even higher rate of urinary incontinence here: greater than 50% in women 60 years or older, with 1) urge urinary incontinence (UUI) predominating4 and 2) the prevalence particularly high among older women who are homebound or who live in a long-term care facility.5
OAB can undermine quality of life in several ways: social isolation, anxiety, poor sleep, higher risk of fracture after a fall,6 reduced ability to function, and poor self-perception. Despite these harmful effects, many women delay seeking care for OAB because they are embarrassed to talk about it with their physician.
Treatment by generalists is feasible—but there is a catch
It’s possible to treat most patients with OAB without referral to a specialist. Two common concerns, however, may set up a roadblock to successful management: the adverse effects associated with some agents and suboptimal control of symptoms.
In this Update, we review recent findings about 1) the potential that anticholinergic therapy has for impairing cognitive function in the older population of women and 2) the important role that concomitant behavioral therapy plays in the long-term success of, and patients’ satisfaction with, treatment of OAB.
Behavioral therapy for OAB: Is it worth all the effort?
Burgio KL, Locher JL, Goode PS. Combined behavioral and drug therapy for urge incontinence in older women. J Am Geriatr Soc. 2000;48:370–374.
The authors of this article followed a randomized clinical trial of older women that compared behavioral and drug therapy for OAB. In the trial, biofeedback-assisted behavioral training (comprising anorectal biofeedback, urge strategies, pelvic muscle biofeedback, and practitioner-directed review with optimization) was compared with treatment with oxybutynin, between 2.5 and 15 mg/day. Both biofeedback-assisted behavioral therapy and the drug regimen were found effective, although neither treatment provided an entirely satisfactory result for all patients. (For a brief description of what constitutes behavioral treatment, see “6 tenets of behavioral therapy for urge urinary incontinence.”)
Second phase of the trial. To determine if treatment satisfaction could be enhanced, the investigators performed a modified crossover study to determine whether combination therapy—biofeedback-assisted behavioral training plus oxybutynin—added any benefit over treatment with behavioral therapy or drug therapy alone. Eligibility was determined by age (55 years or older), demonstrated UUI for at least 3 months, and incomplete dryness or incomplete satisfaction with the outcome of 8 weeks of single-intervention treatment (with either treatment) during the initial phase of the trial.
This subgroup was offered an additional 8 weeks of combination therapy. The primary outcome measure was a reduction in the frequency of episodes of incontinence episodes as recorded by subjects in a bladder diary.
Of 197 women who participated in the original randomized clinical trial, 35—27 who completed drug therapy and 8 who completed behavioral treatment—elected to receive combination therapy. Those 35 subjects did not differ in any of the multiple baseline variables; mean age was 69.3 years (standard deviation [SD], ±7.9 years).
Among subjects originally assigned to behavioral therapy alone, overall reduction in incontinence increased from a mean of 57.5% to a mean of 88.5% after combined therapy (P=.034). Subjects originally assigned to drug therapy alone demonstrated an improvement from 72.7% reduction in incontinence to a mean 84.3% overall reduction with combined therapy (P=.001).
These data suggest that combined therapy can be more effective than behavioral therapy or drug therapy alone. The impact of this study is limited, however, by the relatively low percentage (12.7%) of patients who had received behavioral therapy and chose to add drug therapy, compared with the 41.5% who moved from drug therapy alone to add behavioral therapy.
Furthermore, subjects were self-selected: They chose to continue with an additional 8 weeks of therapy after their initial suboptimal outcome. It is possible that some subjects who were neither totally continent nor completely satisfied with initial therapy chose not to continue with the crossover segment of the trial because it posed too great a burden or because they were discouraged with the initial degree of improvement.
Generalizing these results to all older women with UUI is difficult. The authors point out, however, that, in practice, patients may be more likely than not to choose combination therapy in the hope of shortening the duration of medical therapy. Although it isn’t known whether providing combination therapy from the outset would have yielded better outcomes than either single therapy did, the authors hypothesize that initial combination therapy may result in greater improvement because patients have a high level of motivation and expectation of improvement at the beginning of treatment.
Importance of this article. The investigators demonstrated that a combination of behavioral and drug therapies can provide increased effectiveness in patients for whom each treatment alone led to suboptimal satisfaction. Furthermore, by targeting women older than 55 years, the investigators were able to demonstrate this effectiveness in a group for whom pelvic-floor training may be more difficult than it is for younger women.
It will be interesting to see if future research will 1) validate these findings and 2) determine whether combined therapy can reduce the duration of drug therapy in this older population through behavioral modification and pelvic floor reeducation.
Fluid management
This first-step therapy can involve providing a handout to the patient that details techniques she can use to monitor and control her fluid intake in a manner that addresses her problem. Among such steps:
- avoiding caffeine and artificial sweeteners
- tracking her diet to identify any other bladder irritants
- limiting fluids before times she is more likely to be incontinent—during a long drive, for example, or, in the case of nocturia, after the evening meal.
Scheduled voiding
With scheduled, or prompted, voiding, the patient empties her bladder at a set interval—usually, every 1.5 to 2 hours. If nocturia, or the more severe enuresis, is a problem, the patient can be prompted by an alarm clock or (if she is institutionalized) by nursing staff. Combining scheduled voiding with fluid management principles helps the patient avoid reaching a bladder volume at which an episode of incontinence becomes more likely.
Bladder training
This is a modification of scheduled voiding that attempts to establish a normal voiding interval in patients who have significant frequency but a small voided volume. It imposes a regimented voiding schedule that gradually (over 7 to 10 days) extends the duration between voids.
Pelvic floor-muscle exercises
The focus here is on using pelvic-floor muscles to prevent incontinence. The muscles are strengthened by having the patient perform Kegel exercises (named for Arnold H. Kegel, MD, who, in 1948, recognized the role of pelvic floor-muscle rehabilitation in the treatment of incontinence). The exercises involve simultaneous 1) contraction of the pelvic and periurethral musculature and 2) relaxation of other muscles, including abdominal muscles, which can increase pressure on the bladder.
Once the patient learns to perform Kegel exercises, she can use them to suppress urgency: Instead of hurrying to the bathroom when urgency arises, she is encouraged to sit down, relax, and contract the pelvic-floor muscles repeatedly until the urge to void diminishes. Once it does, the patient proceeds to the toilet to void normally.
Pelvic exam
By self-exam, the patient can identify and familiarize herself with her purposeful contractions of the pelvic-floor musculature and thereby strengthen those muscles with effective exercise.
Biofeedback
Direct feedback about contractions of the pelvic-floor muscles—by a display of data on a gauge or computer monitor, gathered using an intravaginal or anorectal sensor or probe—allows a patient who is exercising those muscles to better target her efforts and maximize their effectiveness.
Combining behavioral therapy and an anticholinergic medication for urge urinary incontinence may yield a superior result after either modality alone has been disappointing by the patient’s account of success.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
Does oxybutynin for UUI further erode cognition in elderly women who are cognitively impaired?
Lackner TE, Wyman JF, McCarthy TC, Monigold M, Davey C. Randomized, placebo-controlled trial of the cognitive effect, safety, and tolerability of oral extended-release oxybutynin in cognitively impaired nursing home residents with urge urinary incontinence. J Am Geriatr Soc. 2008;56:862–870.
Although anticholinergic therapy is modestly effective against UUI in nursing home residents, past studies have suggested that such treatment can impair, or further impair, cognition in this population—a concern that may lead to underuse. This double-blinded, randomized, placebo-controlled trial compared short-term oral extended-release oxybutynin with placebo.
Consequently, the authors sought to determine the cognitive effect, safety, and tolerability of 5 mg/day oral extended-release oxybutynin (the most commonly prescribed dosage) in cognitively impaired older nursing home residents who have UUI.
Subjects were eligible if they:
- were 65 years or older
- had UUI
- lived in a nursing home longer than 3 months
- had cognitive impairment.
Women already being treated for urinary incontinence, those who had an indwelling Foley catheter or urinary retention, and those who were bed-bound or incommunicative were excluded.
Fifty women, mean age 88.6 years (SD, ±6.2), from 12 nursing home facilities, agreed to participate. They were further stratified based on the score of a Mini-Mental State Exam (MMSE): 13 had severe cognitive impairment (MMSE score, 5–10) and 37 had mild or moderate impairment (score, 11–23).
Subjects were randomized to 4 weeks’ treatment with either 5 mg/day oral extended-release oxybutynin or one placebo tablet daily. A nurse practitioner who was blinded to randomization collected all data. The Confusion Assessment Method (CAM) algorithm, MMSE, and Severe Impairment Battery (SIB) were used to assess cognitive decline. The Brief Agitation Rating Scale (BARS) assessed agitation.
No baseline differences were noted with regard to: age; demographic, functional, and neuropsychiatric characteristics; clinical factors predisposing to delirium; and serum anticholinergic activity. Adherence was similar in the treatment (97%) and placebo (97.4%) groups.
Finding: Cognitive impairment. Treatment and placebo groups in the baseline mild-or-moderate stratum (by MMSE) showed equivalent mean changes in CAM scores at all time points. Because of the small sample size, however, CAM score equivalence could not be definitively determined for the groups in the severe impairment stratum. Evaluation of mean MMSE and BARS scores showed no significant changes between groups.
Finding: Tolerability. Excellent tolerability was noted in the treatment group: 96% of subjects completed the trial (compared with 92% of the placebo group). No difference in the rate of adverse events was noted between treatment and placebo groups; of adverse events recorded, 90% were judged “mild” by the investigators. Constipation and dry mouth were most common.
Finding: Falls. More than half—54%—of subjects in both groups experienced at least one fall during the trial or during the preceding or following 3 months. Despite this, no difference in the rate of falls between the treatment and placebo groups was noted. Furthermore, regression analysis revealed no treatment or period effect on falls per month across the time of observation.
Conclusions. Treatment with 5 mg/day oral extended-release oxybutynin in older patients with some cognitive impairment is well tolerated, the study’s findings suggest, with minimal risk of further cognitive decline or delirium over the short term. The potential that long-term therapy has to harm cognitive function remains, however; data on long-term treatment are needed to illuminate that area.
The authors also address the importance of dosing, especially over time, and discuss the lower potential of newer-generation anticholinergics to produce cognitive impairment.
A limited number of articles in the medical literature address anticholinergics in an older population, specifically, and only a few of those evaluated the effects of the drugs on cognitive function. By investigating patients who had an existing cognitive impairment, the authors of this article were able to target a cohort at risk of further cognitive impairment from medication use—thereby giving further weight to their findings of no significant effect.
Main strengths and limitations of the study. The investigators used validated, standardized cognitive tests that were administered by a uniform blinded evaluator in a randomized, controlled trial. The study was limited, however, because patients were evaluated only over a relatively short period (1 month) and because the efficacy of therapy was not addressed.
Further studies of anticholinergic medications, using the same rigorous scientific approach that these investigators applied, are needed to address 1) the long-term efficacy of oxybutynin and similar agents and 2) the cognitive effects of long-term treatment in this older population.
Further impairment is unlikely over the short term when a cognitively impaired nursing home patient who has urge urinary incontinence is treated with 5 mg/day oral extended-release oxybutynin.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
1. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Am J Obstet Gynecol. 2002;187:116-126.
2. Hu TW, Wagner TH, Bentkover JD, et al. Estimated economic costs of overactive bladder in the United States. Urology. 2003;61:1123-1128.
3. Hannestad YS, Rortveit G, Sandvik H, Hunskaar S. A community-based epidemiological survey of female urinary incontinence: the Norwegian EPINCONT study. Epidemiology of Incontinence in the County of Nord-Trøndelag. J Clin Epidemiol. 2000;53:1150-1157.
4. Melville JL, Katon W, Delaney K, Newton K. Urinary incontinence in US women: a population-based study. Arch Intern Med. 2005;165:537-542.
5. Fantl JA, Newman DK, Colling J, et al. Managing Acute and Chronic Urinary Incontinence. Clinical Practice Guideline. Quick Reference Guide for Clinicians, No. 2, 1996 Update. Rockville, MD: US Department of Health and Human Services, Public Health Service, Agency for Health Care Policy and Research. AHCPR Pub. No. 96-0686. January 1996. Available at: http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=hstat6.chapter.32554. Accessed September 11, 2009.
6. Brown JS, Vittinghoff E, Wyman JF, et al. Urinary incontinence: does it increase risk for falls and fractures? Study of Osteoporotic Fractures Research Group. J Am Geriatr Soc. 2000;48:721-725.
1. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Am J Obstet Gynecol. 2002;187:116-126.
2. Hu TW, Wagner TH, Bentkover JD, et al. Estimated economic costs of overactive bladder in the United States. Urology. 2003;61:1123-1128.
3. Hannestad YS, Rortveit G, Sandvik H, Hunskaar S. A community-based epidemiological survey of female urinary incontinence: the Norwegian EPINCONT study. Epidemiology of Incontinence in the County of Nord-Trøndelag. J Clin Epidemiol. 2000;53:1150-1157.
4. Melville JL, Katon W, Delaney K, Newton K. Urinary incontinence in US women: a population-based study. Arch Intern Med. 2005;165:537-542.
5. Fantl JA, Newman DK, Colling J, et al. Managing Acute and Chronic Urinary Incontinence. Clinical Practice Guideline. Quick Reference Guide for Clinicians, No. 2, 1996 Update. Rockville, MD: US Department of Health and Human Services, Public Health Service, Agency for Health Care Policy and Research. AHCPR Pub. No. 96-0686. January 1996. Available at: http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=hstat6.chapter.32554. Accessed September 11, 2009.
6. Brown JS, Vittinghoff E, Wyman JF, et al. Urinary incontinence: does it increase risk for falls and fractures? Study of Osteoporotic Fractures Research Group. J Am Geriatr Soc. 2000;48:721-725.
Does cognitive function decline during the menopausal transition?
These important findings from SWAN confirm the memory problems reported by many women during the menopausal transition. They also build on the findings of clinical trials of HT in younger menopausal women, which explored the effect of HT on cardiovascular health, to underscore the critical role that timing of such therapy can play.1,2
Other cohort studies of middle-aged women in the United States have found the menopausal transition to be associated with undesirable changes in mood3,4; some experts have described the endocrine shifts that accompany menopause as “hormonal chaos.”5
Details of the study
Participants were 42 to 52 years old and had an intact uterus and at least one ovary at entry. They were followed for 4 years, with delineation of the menopausal stage (i.e., premenopause, early and late perimenopause, and menopause) and assessment of hormone use prior to the final menstrual period and after menopause. The outcome was longitudinal performance in three cognitive domains:
- processing speed—assessed using the Symbol Digit Modalities Test. Premenopausal, early perimenopausal, and postmenopausal women improved with repeated administration of this test, but late perimenopausal women did not. Prior use of HT improved the score, whereas late use of HT reduced it.
- verbal memory—evaluated via the East Boston Memory Test. Test scores increased during premenopause and postmenopause but not during early or late perimenopause. Prior use of HT improved the test score, but late use reduced the score.
- working memory—assessed using the Digit Span Backward test. This domain did not vary by stage of menopause.
Participants who initiated menopausal HT or oral contraceptives prior to the last menstrual period were excluded from the analysis during use of HT. They were allowed to reenter the study, however, once HT or oral contraceptives were discontinued.
As the cognitive component of SWAN began, the mean age of participants was 50 years, and 8% were premenopausal, 49% were early perimenopausal, 12% were late perimenopausal, and 27% were postmenopausal. In addition, 4% were both postmenopausal and current users of HT.
Because perimenopausal HT should include a dosage of progestin adequate to suppress ovulation in order to prevent iatrogenic irregular uterine bleeding, women who are healthy, lean, nonsmoking, and still having menstrual periods can safely use a conventional oral contraceptive. Options for other symptomatic perimenopausal women include a continuous oral menopausal regimen formulated with 5 μg of ethinyl estradiol and 1 mg of norethindrone acetate (Femhrt 1/5) or 1 mg of estradiol and 0.5 mg of norethindrone acetate (Activella 1/0.5 or generic).—ANDREW M. KAUNITZ, MD
1. Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465-1477.
2. Manson JE, Allison MA, Rossouw JE, et al. For the WHI and WHI-CACS Investigators. Estrogen therapy and coronary-artery calcification. N Engl J Med. 2007;356:2591-2602.
3. Freeman EW, Sammel MD, Lin H, et al. Symptoms associated with the menopausal transition and reproductive hormones in midlife women. Obstet Gynecol. 2007;110(2 Pt 1):230-240.
4. Woods NF, Smith-DiJulio K, Percival DB, Tao EY, Mariella A, Mitchell S. Depressed mood during the menopausal transition and early postmenopause: observations from the Seattle Midlife Women’s Health Study. Menopause. 2008;15:223-232.
5. Berga SL. Disordered folliculogenesis during the menopausal transition: explaining chaos. Menopause. 2009;16:11-12.
These important findings from SWAN confirm the memory problems reported by many women during the menopausal transition. They also build on the findings of clinical trials of HT in younger menopausal women, which explored the effect of HT on cardiovascular health, to underscore the critical role that timing of such therapy can play.1,2
Other cohort studies of middle-aged women in the United States have found the menopausal transition to be associated with undesirable changes in mood3,4; some experts have described the endocrine shifts that accompany menopause as “hormonal chaos.”5
Details of the study
Participants were 42 to 52 years old and had an intact uterus and at least one ovary at entry. They were followed for 4 years, with delineation of the menopausal stage (i.e., premenopause, early and late perimenopause, and menopause) and assessment of hormone use prior to the final menstrual period and after menopause. The outcome was longitudinal performance in three cognitive domains:
- processing speed—assessed using the Symbol Digit Modalities Test. Premenopausal, early perimenopausal, and postmenopausal women improved with repeated administration of this test, but late perimenopausal women did not. Prior use of HT improved the score, whereas late use of HT reduced it.
- verbal memory—evaluated via the East Boston Memory Test. Test scores increased during premenopause and postmenopause but not during early or late perimenopause. Prior use of HT improved the test score, but late use reduced the score.
- working memory—assessed using the Digit Span Backward test. This domain did not vary by stage of menopause.
Participants who initiated menopausal HT or oral contraceptives prior to the last menstrual period were excluded from the analysis during use of HT. They were allowed to reenter the study, however, once HT or oral contraceptives were discontinued.
As the cognitive component of SWAN began, the mean age of participants was 50 years, and 8% were premenopausal, 49% were early perimenopausal, 12% were late perimenopausal, and 27% were postmenopausal. In addition, 4% were both postmenopausal and current users of HT.
Because perimenopausal HT should include a dosage of progestin adequate to suppress ovulation in order to prevent iatrogenic irregular uterine bleeding, women who are healthy, lean, nonsmoking, and still having menstrual periods can safely use a conventional oral contraceptive. Options for other symptomatic perimenopausal women include a continuous oral menopausal regimen formulated with 5 μg of ethinyl estradiol and 1 mg of norethindrone acetate (Femhrt 1/5) or 1 mg of estradiol and 0.5 mg of norethindrone acetate (Activella 1/0.5 or generic).—ANDREW M. KAUNITZ, MD
These important findings from SWAN confirm the memory problems reported by many women during the menopausal transition. They also build on the findings of clinical trials of HT in younger menopausal women, which explored the effect of HT on cardiovascular health, to underscore the critical role that timing of such therapy can play.1,2
Other cohort studies of middle-aged women in the United States have found the menopausal transition to be associated with undesirable changes in mood3,4; some experts have described the endocrine shifts that accompany menopause as “hormonal chaos.”5
Details of the study
Participants were 42 to 52 years old and had an intact uterus and at least one ovary at entry. They were followed for 4 years, with delineation of the menopausal stage (i.e., premenopause, early and late perimenopause, and menopause) and assessment of hormone use prior to the final menstrual period and after menopause. The outcome was longitudinal performance in three cognitive domains:
- processing speed—assessed using the Symbol Digit Modalities Test. Premenopausal, early perimenopausal, and postmenopausal women improved with repeated administration of this test, but late perimenopausal women did not. Prior use of HT improved the score, whereas late use of HT reduced it.
- verbal memory—evaluated via the East Boston Memory Test. Test scores increased during premenopause and postmenopause but not during early or late perimenopause. Prior use of HT improved the test score, but late use reduced the score.
- working memory—assessed using the Digit Span Backward test. This domain did not vary by stage of menopause.
Participants who initiated menopausal HT or oral contraceptives prior to the last menstrual period were excluded from the analysis during use of HT. They were allowed to reenter the study, however, once HT or oral contraceptives were discontinued.
As the cognitive component of SWAN began, the mean age of participants was 50 years, and 8% were premenopausal, 49% were early perimenopausal, 12% were late perimenopausal, and 27% were postmenopausal. In addition, 4% were both postmenopausal and current users of HT.
Because perimenopausal HT should include a dosage of progestin adequate to suppress ovulation in order to prevent iatrogenic irregular uterine bleeding, women who are healthy, lean, nonsmoking, and still having menstrual periods can safely use a conventional oral contraceptive. Options for other symptomatic perimenopausal women include a continuous oral menopausal regimen formulated with 5 μg of ethinyl estradiol and 1 mg of norethindrone acetate (Femhrt 1/5) or 1 mg of estradiol and 0.5 mg of norethindrone acetate (Activella 1/0.5 or generic).—ANDREW M. KAUNITZ, MD
1. Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465-1477.
2. Manson JE, Allison MA, Rossouw JE, et al. For the WHI and WHI-CACS Investigators. Estrogen therapy and coronary-artery calcification. N Engl J Med. 2007;356:2591-2602.
3. Freeman EW, Sammel MD, Lin H, et al. Symptoms associated with the menopausal transition and reproductive hormones in midlife women. Obstet Gynecol. 2007;110(2 Pt 1):230-240.
4. Woods NF, Smith-DiJulio K, Percival DB, Tao EY, Mariella A, Mitchell S. Depressed mood during the menopausal transition and early postmenopause: observations from the Seattle Midlife Women’s Health Study. Menopause. 2008;15:223-232.
5. Berga SL. Disordered folliculogenesis during the menopausal transition: explaining chaos. Menopause. 2009;16:11-12.
1. Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465-1477.
2. Manson JE, Allison MA, Rossouw JE, et al. For the WHI and WHI-CACS Investigators. Estrogen therapy and coronary-artery calcification. N Engl J Med. 2007;356:2591-2602.
3. Freeman EW, Sammel MD, Lin H, et al. Symptoms associated with the menopausal transition and reproductive hormones in midlife women. Obstet Gynecol. 2007;110(2 Pt 1):230-240.
4. Woods NF, Smith-DiJulio K, Percival DB, Tao EY, Mariella A, Mitchell S. Depressed mood during the menopausal transition and early postmenopause: observations from the Seattle Midlife Women’s Health Study. Menopause. 2008;15:223-232.
5. Berga SL. Disordered folliculogenesis during the menopausal transition: explaining chaos. Menopause. 2009;16:11-12.
The case for chemoprevention as a tool to avert breast cancer
The author reports that he is a consultant to Eli Lilly, Pfizer, and Wyeth, and a speaker for Eli Lilly and Wyeth.
CASE 1: Premenopausal woman
at high risk of breast cancer
R. J. is a 43-year-old, nulliparous woman who reached menarche at age 11. She has undergone two breast biopsies, the most recent of which revealed ductal hyperplasia with marked atypia.
R. J.’s sister had breast cancer at 49 years of age; her mother had breast cancer at 66 years. Because of R. J.’s family history, she underwent testing for a BRCA mutation. The result was negative.
R. J. has come to your office today to find out if she can do anything to reduce her risk of breast cancer. What options can you offer?
The most common method of “prevention” of breast cancer involves early detection and assessment of abnormalities through frequent surveillance with mammography. Some women who have dense breasts, a history of breast biopsy, or other risk factors for breast cancer may benefit from intensive surveillance with both mammography and ultrasonography—and, in some cases, magnetic resonance imaging.
More aggressive options include:
- the use of a chemopreventive agent such as tamoxifen or raloxifene
- in rare cases—usually when a BRCA mutation is present—prophylactic mastectomy.
Before it is possible to determine the optimal approach for a particular woman, it is necessary to conduct an individualized assessment of her risk—that is, to estimate the probability that she will develop breast cancer over a defined period of time. Such an estimate is also useful for designing prevention trials in high-risk subsets of the population. (Prevention trials differ from therapeutic clinical trials in that asymptomatic healthy women are exposed to potentially toxic interventions for prolonged periods to reduce their risk of breast cancer.)
This article describes chemopreventive options for women at high risk, based on individualized risk assessment using the Gail model.
(Editor’s note: For additional discussion of the important role ObGyns play in the fight against breast cancer, see Editor in Chief Dr. Robert L. Barbieri’s Editorial.)
You can estimate the likelihood that a woman like your patient may develop breast cancer using various individual risk factors ( TABLE 1 ), but estimates for combinations of risk factors are preferable. The Gail model takes into account some nongenetic factors, such as parity and age at menarche, but also genetic factors, such as family history. The model calculates a woman’s individualized breast cancer probability and yields a numerical risk (a percentage) that she will develop invasive breast cancer over the next 5 years; it also yields an estimate of her risk of developing the malignancy over the remainder of her life.1,2
A Gail-model 5-year estimate of 1.66% or higher denotes a high risk of developing breast cancer. That benchmark was the one employed in the Breast Cancer Prevention Trial (BCPT), conducted as part of the National Surgical Adjuvant Breast and Bowel Project (NSABP).3
TABLE 1
What are the risk factors for breast cancer?
And what degree of relative risk do they confer?
| Relative risk | ||
|---|---|---|
| <2 | 2–4 | >4 |
| • Age 25–34 years at first live birth • Early menarche • Late menopause • Benign proliferative disease • Postmenopausal obesity • Alcohol use • Hormone replacement therapy | • Age >35 years at first live birth • First-degree relative with breast cancer • Nulliparity • Radiation exposure • Personal history of breast cancer | • Gene mutation (BRCA 1 or 2) • Lobular carcinoma in situ • Ductal carcinoma in situ • Atypical hyperplasia |
| Adapted from Bilimoria and Morrow23 | ||
Weaknesses of the Gail model
The Gail model’s approach to estimating risk has some limitations. The model uses the number of prior breast biopsies in its assessment—but the relative risk associated with prior biopsy is smaller for women older than 50 years than it is for younger women.
Furthermore, data on which Gail bases its estimates were collected in the late 1970s and early 1980s. Since then, the increasing ease of breast histopathologic assessment—through fine-needle aspiration and outpatient core-needle biopsy—has confused the issue of just what constitutes a breast “biopsy.” (Most patients surveyed consider it to be any histologic sampling of the breast.)
As a result, the 1.66% cutoff becomes somewhat difficult to interpret in light of current practice.
Consider the following example. A 50-year-old nulliparous Caucasian woman reached menarche when she was 11 years old, has never had a biopsy, and has no first-degree relatives with breast cancer. According to the Gail model, her risk of developing breast cancer is 1.2% over the next 5 years and 10.8% in her lifetime. Therefore, she is not considered at high risk. If she were to give a history of three previous breast biopsies, however, none of them showing hyperplasia, her 5-year risk would rise to 1.8% and push her over the line into the high-risk category.
Compare her situation to that of R. J., the nulliparous woman described in Case 1. R. J. also reached menarche at 11 years, but she has had two breast biopsies (one of which showed atypical hyperplasia) and has two first-degree relatives who have had breast cancer. Her Gail score shows a 5-year risk of breast cancer of 13.5% (the norm for a 43-year-old woman is 0.8%), and a lifetime risk of 69.2%. Clearly, she has a high risk of breast cancer.
How do we improve an imperfect science?
We need to identify objective findings that are patient-specific but highly correlative with the development of breast cancer. Patient-specific biomarkers have been proposed, such as ultrasensitive measurement of the serum estradiol level in postmenopausal women. In the Multiple Outcomes of Raloxifene Evaluation, also known as the MORE trial, women who experienced the greatest reduction in the rate of breast cancer during treatment with raloxifene were a subgroup who had the highest baseline level of serum estradiol—although, overall, all patients had an estradiol level well within the postmenopausal range (≤20 pmol/L).4,5
How tamoxifen became a chemopreventive agent
Tamoxifen inhibits mammary tumors in mice and rats and suppresses hormone-dependent breast cancer cell lines in vitro.6 Clinical data from the Early Breast Cancer Trialists’ Collaborative Group yielded additional motivation for prevention trials with tamoxifen: Besides reducing the rate of recurrent breast cancer, tamoxifen reduced the risk of contralateral new-onset breast cancer by 47% after 5 years of adjuvant treatment.7 Preclinical findings in vitro and in animal models, coupled with clinical data and evidence of tamoxifen’s favorable effects on skeleton remodeling and lipid levels, led to a series of chemoprevention trials in the United States and Europe using tamoxifen.
In the aforementioned BCPT, launched in 1992, 13,388 women 35 years and older who were deemed to be at high risk of developing breast cancer were enrolled at numerous sites throughout the United States and Canada.3 The Gail model was used to select women for the trial—only those who had a 5-year risk of 1.66% or higher were included. Participants were randomly assigned to receive tamoxifen 20 mg or placebo daily for 5 years. The trial was terminated early because of the dramatic reduction in new-onset breast cancer with tamoxifen, compared with placebo.
The overall incidence of breast cancer in the tamoxifen group was 3.4 cases for every 1,000 women, compared with 6.8 cases for every 1,000 women receiving placebo.3 Overall, the reduction in invasive breast cancer with tamoxifen was 49% (P<.00001). When broken down by age group, the reduction was:
- 44% in women 35 to 49 years old
- 51% in women 50 to 59 years old
- 55% in women 60 years and older.
Even noninvasive breast cancer was reduced with tamoxifen
Tamoxifen decreased the incidence of noninvasive breast cancer (ductal carcinoma in situ [DCIS]) by 50%. Expanded use of mammography has increased the detection of DCIS. Most DCIS lesions appear to be estrogen-receptor positive.8
In addition, tamoxifen reduced breast cancer risk in women who had a history of lobular carcinoma in situ (LCIS), a precancer, by 56%, and it reduced the risk of breast cancer in women who had a history of atypical hyperplasia by 86%. Overall, tamoxifen reduced the occurrence of estrogen-positive tumors by 69%, but had no impact on the incidence of estrogen-receptor–negative tumors.
The BCPT was stopped 14 months before planned because the Data and Safety Monitoring Board felt it was unethical to continue to allow one half of such high-risk participants to take placebo in light of the dramatic reduction in both invasive and noninvasive breast cancer among women who took tamoxifen.
In postmenopausal women, tamoxifen increases some risks
Two secondary endpoints of the BCPT are worthy of consideration:
- The overall relative risk (RR) of endometrial cancer associated with tamoxifen therapy in healthy women was 2.53 (95% confidence interval [CI], 1.35, 4.97). However, further analysis by age yielded a RR of 4.01 in women who were older than 50 years (95% CI, 1.70, 10.90), compared with a RR of 1.21 in women 49 years and younger (95% CI, 0.41, 3.60).
- The same age distinction held true for deep venous thrombosis (DVT) and pulmonary embolus, with no statistically significant increases in either in women 49 years and younger, but a RR of 1.71 and 3.19, respectively, in women 50 years and older. It is unclear whether the trial was sufficiently powered for this particular secondary endpoint.
These findings suggest that serious adverse events do not occur at the same magnitude in women younger than 50 years that they do in women 50 and older. The difference in the risk–benefit profile between younger and older women has significant clinical implications for the care of perimenopausal patients.
Risk of other malignancies was not affected by tamoxifen
Overall, invasive cancers other than those of the breast and uterus occurred at the same rate in the tamoxifen and placebo groups of the BCPT. The RR of death from any cause was 0.81 (95% CI, 0.56–1.16). There was a slight increase in the risk of myocardial infarction (RR, 1.11; 95% CI, 0.65–1.92) and a slight decrease in the risk of severe angina (RR, 0.93; 95% CI, 0.40–2.14) in tamoxifen users, although neither of these risks was statistically significant.
The overall RR of fracture of the hip, spine, or radius was 0.81 (95% CI, 0.63–1.05). There was a statistically significant increase in the number of women who had cataracts who then underwent cataract surgery in the tamoxifen group (RR, 1.57; 95% CI, 1.16–2.14).
Tamoxifen is approved as a preventive for high-risk women only
Based on the results of the BCPT, the US Food and Drug Administration (FDA) approved tamoxifen in October 1998 for the primary prevention of breast cancer in women who are at high risk of the disease. The FDA recommends that use of tamoxifen be limited to women at high risk because of the potentially serious side effects seen in clinical trials, including the BCPT.
The FDA did not define “high risk,” but it did recommend that the decision to use tamoxifen as chemopreventive therapy be based on thorough evaluation of the patient’s personal, family, and medical histories; her age; and her understanding of the risks and benefits of treatment.
The FDA also required the following language in the package insert:
- You should not take tamoxifen to reduce the risk of breast cancer unless you are at high risk of breast cancer. Certain conditions put women at high risk, and it is possible to calculate this risk for any woman. Breast cancer risk-assessment tools to help calculate your risk of breast cancer have been developed and are available to your health-care professional. You should discuss your risk with your healthcare professional.
CASE 1 RESOLVED
You determine that R. J. is an excellent candidate for tamoxifen by virtue of her significant risk of breast cancer. You are able to reassure her that, as the BCPT demonstrated, tamoxifen should not increase the risk of uterine cancer, DVT, or pulmonary embolism in a woman her age.
Raloxifene
CASE 2: Patient worries about breasts and bones
S. T. is a 58-year-old Caucasian mother of two whose own mother had breast cancer when she was 74 years old, and whose older sister was given a diagnosis of the malignancy 4 years ago.
S. T. had her first period when she was 11 years old, delivered her first child when she was 31, and entered menopause when she was 52. She is 5 ft 5 in tall and weighs 144 lb.
Her main reason for visiting you today is a breast Mammotome biopsy that showed ductal hyperplasia with atypia. She has been tested for a BRCA mutation, but the result was negative. Her Gail-model score is a 9.7% risk of developing breast cancer over the next 5 years, and a lifetime risk of 44.2%.
She also asks about osteoporosis prevention, given that a dual-energy x-ray absorptiometry (DXA) scan 1 year ago yielded a T-score of –1.3 for her hip and –1.1 for her spine. Her World Health Organization FRAX 10-year risk of hip fracture is 0.7%, and her risk of major osteoporotic fracture is 8.6%.
How do you respond to her concerns?
This patient has a high risk of invasive breast cancer but does not meet criteria for pharmacotherapy for osteoporosis prevention. A good option for her would be raloxifene, a selective estrogen-receptor modulator (SERM) that has been shown to reduce the risk of breast cancer as well as osteoporosis. S. T. would benefit from it on the basis of its breast benefit alone.
The genesis of a drug with multiple benefits
Raloxifene is a benzothiophene derivative, unlike the triphenylethylene family from which tamoxifen is derived. Like tamoxifen, raloxifene was originally investigated as a treatment for advanced breast cancer.
Preclinical studies indicated that raloxifene had an antiproliferative effect on both estrogen-receptor–positive mammary tumors and estrogen-receptor–positive human breast cancer cell lines.9 In the 1980s, however, a small, phase-II trial revealed that raloxifene had no further antitumor effects in postmenopausal women with advanced breast cancer in whom tamoxifen had failed.10 After information surfaced about the neoplastic effect of tamoxifen on the uteri of postmenopausal women, interest in raloxifene revived.11
Raloxifene has estrogen-agonistic activity on bone remodeling and lipid metabolism and was approved by the FDA for prevention of osteoporosis in postmenopausal women in December 1997. Its indication was extended to treatment of osteoporosis 2 years later.
Raloxifene appears to have no effect on the endometrium of postmenopausal women, compared with placebo. In a 12-month comparative trial, there was no difference in endometrial thickness, endoluminal masses, proliferation, or hyperplasia between the raloxifene and placebo groups.12 This finding corroborates earlier evidence that raloxifene does not cause endometrial hyperplasia or cancer and is not associated with vaginal bleeding or increased endometrial thickness, as measured by transvaginal ultrasonography.
A big difference between raloxifene and tamoxifen, therefore, is their varying effect on the uterus of postmenopausal women.
Additional clinical trials confirm anticancer action of raloxifene
Preclinical data in animal models suggested that, like tamoxifen, raloxifene has potent antiestrogenic effects on breast tissue.9 The MORE trial involved 7,705 postmenopausal women up to 80 years old who had established osteoporosis.13 In that trial, participants were randomized to raloxifene or placebo. Bone mineral density (BMD) and fracture incidence were the primary endpoints; breast cancer was a secondary endpoint.
Over the 4 years of the trial, raloxifene significantly reduced the incidence of all invasive breast cancers by 72%, compared with placebo (RR, 0.28; 95% CI, 0.17–0.46). Raloxifene also significantly reduced the incidence of invasive estrogen-receptor–positive tumors by 84%, compared with placebo (RR, 0.16; 95% CI, 0.09–0.30), but had no effect on estrogen-receptor–negative tumors. The incidence of vaginal bleeding, breast pain, and endometrial cancer in the raloxifene group did not differ significantly from that of the placebo group.
Like tamoxifen, raloxifene appeared to be associated with an increased risk of thromboembolic disease, including DVT and pulmonary embolism, which developed in 1.1% of women taking raloxifene, compared with 0.5% of women in the placebo group (P=.003).
In a 4-year continuation of the MORE trial, known as the Continuing Outcomes Relevant to Evista, or CORE, trial, 5,231 women were randomized to continue raloxifene or placebo.14 Over the 8 years of the combined trials, the incidence of invasive breast cancer was reduced by 66% in the raloxifene group (RR, 0.34; 95% CI, 0.22–0.50). The 8-year data are extremely clinically relevant, in that raloxifene has no time limit, whereas tamoxifen is usually prescribed for no longer than 5 years.
Raloxifene is not approved for use in premenopausal women. SERM compounds, which are structurally similar to clomiphene citrate, seem to have different effects in premenopausal and postmenopausal women, as evidenced by tamoxifen’s differing effects by age in the BCPT.
Other investigations of raloxifene confirm its value in high-risk women
To compare the clinical safety and efficacy of tamoxifen and raloxifene in reducing the risk of breast cancer among healthy women, the Study of Tamoxifen and Raloxifene (STAR) was initiated in 1999.15 In that trial, 19,747 postmenopausal women older than 35 years were blindly assigned to raloxifene 60 mg or tamoxifen 20 mg daily.
Baseline characteristics of subjects in STAR are summarized in TABLE 2 . Mean age was 58.5 years. All women had a 5-year risk of developing breast cancer that exceeded 1.66%, according to the Gail model. The average Gail score was 4.03% (standard deviation, ±2.17%). Because it would have been unethical to subject high-risk women to a placebo group in light of the findings of the BCPT, there was no placebo control.
TABLE 2
Baseline characteristics of women
in the Study of Tamoxifen and Raloxifene (STAR) trial
| Characteristic | Value |
|---|---|
| Age (mean) | 58.5 years |
| Caucasian | 93% |
| Hysterectomy | 51% |
| At least one first-degree relative with breast cancer | 71% |
| Lobular carcinoma in situ | 9% |
| Atypical hyperplasia | 23% |
| 5-year risk of invasive breast cancer (mean)* | 4.03% |
| *As estimated with the Gail model Risk Calculator. | |
Here are noteworthy findings of the STAR trial:
- 163 cases of invasive breast cancer occurred in the tamoxifen group, compared with 168 among women taking raloxifene (RR, 1.02; 95% CI, 0.82–1.28).
- 36 cases of uterine cancer occurred in the tamoxifen group, compared with 23 among women taking raloxifene (RR, 0.62; 95% CI, 0.35–1.08). Earlier studies had shown a marked difference in the rate of uterine cancer between these agents. Although the difference here is not statistically significant, uterine cancer was not an endpoint of the study; nor was the study powered to explore this difference.
- The number of hysterectomies among women who were diagnosed with endometrial hyperplasia with or without atypia was, proportionally, significantly higher among women taking tamoxifen ( TABLE 3 ).
- No difference between groups was found for other invasive cancers, ischemic heart events, or stroke.
- Thromboembolic events occurred less frequently in the raloxifene group (RR, 0.70; 95% CI, 0.54–0.91). However, both raloxifene and tamoxifen have consistently been associated with a twofold to threefold increase in the risk of thromboembolic events, compared with placebo.
- Vasomotor symptoms and leg cramps increased in frequency and severity among women in both groups of the trial. These symptoms appear to be less common and less severe among women who are older and more remote from the onset of menopause.
TABLE 3
Relative risk of hysterectomy and uterine hyperplasia during STAR
| Characteristic | Women who took tamoxifen | Women who took raloxifene | Relative risk (95% confidence interval) |
|---|---|---|---|
| Hysterectomy during study | 246 | 92 | 0.37 (0.28, 0.47) |
| Hyperplasia • with atypia • without atypia | 100 15 85 | 17 2 15 | 0.17 (0.09, 0.28) 0.13 (0.01, 0.56) 0.17 (0.09, 0.30) |
What is raloxifene’s effect on the heart?
The Raloxifene Use for The Heart (RUTH) trial explored the primary endpoints of coronary artery disease (CAD) and breast cancer in more than 10,000 women who had CAD or multiple risk factors for it.16 This study began prior to the Women’s Health Initiative, at a time when hormone replacement therapy was widely believed to reduce CAD.
In the double-blinded, randomized, placebo-controlled RUTH trial, raloxifene had no significant effect on primary coronary events (533 vs 553; hazard ratio [HR], 0.95; 95% CI, 0.84–1.07). Even in this population, however, there was a 44% reduction in invasive breast cancer (40 vs 70 events; HR, 0.56; 95% CI, 0.38–0.83).
Based on these results, the FDA approved raloxifene for the “reduction in risk of invasive breast cancer in postmenopausal women at high risk for breast cancer,” as well as for the “reduction in risk of invasive breast cancer in postmenopausal women with osteoporosis” ( FIGURE ).
FIGURE How raloxifene reduced invasive breast cancer in three trials
Raloxifene significantly reduced the risk of cancer, compared with placebo, in the Raloxifene Use for The Heart (RUTH), Multiple Outcomes of Raloxifene Evaluation (MORE), and Continuing Outcomes Relevant to Evista (CORE) trials.
CASE 2 RESOLVED
S. T. begins taking raloxifene 60 mg daily to lower her risk of invasive breast cancer. Although she temporarily experienced hot flashes after initiating the drug, they are only mildly bothersome, and she continues raloxifene therapy. She says she is grateful that there is an agent that can help her reduce the likelihood that she will develop breast cancer, and protection of her BMD is an added benefit.
CASE 3: At risk for both breast cancer and bone fracture
A. N., 63, is a nulliparous Caucasian woman who weighs 134 lb and stands 5 ft 4 in tall. She reached menarche when she was 12 years old and entered menopause at 49.
Although A. N. has never had a breast abnormality, her 59-year-old sister was just given a diagnosis of breast cancer. Her Gail score reveals that she has a 3.1% risk of developing breast cancer over the next 5 years.
In addition to her concerns about breast cancer, A. N. is worried about hip fracture—because her mother suffered one after menopause and because her T-score is –1.9 at the hip and –2.1 at the spine. A. N. has used steroids off and on for much of her life for asthma. Her FRAX score indicates that she has a 2.8% risk of hip fracture and a 25% risk of major osteoporotic fracture over the next 10 years.
What do you offer her?
Because of new FRAX criteria, this osteopenic woman is now a candidate for medication to reduce her risk of major osteoporotic fracture, and raloxifene is a good option. Her Gail score of 3.1% also makes her a good candidate for breast cancer risk reduction with raloxifene.
CASE 3 RESOLVED
Because A. N. needs an agent that benefits both breast and bone, you prescribe raloxifene. The drug should significantly reduce her risk of both invasive breast cancer and bone fracture, without increasing her risk of endometrial hyperplasia and cancer, both of which are associated with tamoxifen in her age group.
Aromatase inhibitors
A fairly new class of drugs being explored for their ability to reduce the risk of breast cancer is aromatase inhibitors. Substantial evidence suggests that estrogens facilitate the development of breast cancer in animals and in women, although the precise mechanism remains unknown.17 The most commonly held theory is that estrogen stimulates proliferation of breast cells and thereby increases the risk of genetic mutation that could lead to cancer.
Aromatase inhibitors block peripheral conversion of androstenedione to estrogens. In premenopausal women, the primary site of this action is in the ovary. In postmenopausal women, this conversion occurs primarily in extraovarian sites, including the adrenal glands, adipose tissue, liver, muscle, and skin.
Aromatase inhibitors may be more effective than SERMs in preventing breast cancer because of their dual role: blocking both the initiation and promotion of breast cancer.18 These agents reduce levels of the genotoxic metabolites of estradiol by lowering estradiol concentration in tissue. At the same time, aromatase inhibitors also block tumor promotion by lowering tissue levels of estrogen and preventing cell proliferation.
The main drawback of these agents—besides the fact that they are not FDA-approved for reducing risk—is their antiestrogenic effect on bone and lipid metabolism. They also induce vasomotor symptoms.
Studies of third-generation aromatase inhibitors in the prevention of breast cancer are under way in high-risk women. These agents include anastrozole, exemestane, and letrozole.
1. Gail MH, Brinton LA, Byar DP, et al. Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl Cancer Inst. 1989;81:1879-1886.
2. Breast Cancer Assessment Tool. Available at: www.cancer.gov/bcrisktool/Default.aspx. Accessed June 5, 2009.
3. Fisher B, Costantino JP, Wickerham DL, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 study. J Natl Cancer Inst. 1998;90:1371-1388.
4. Ruffin MT, 4th, August DA, Kelloff GJ, Boone CW, Weber BL, Brenner DE. Selection criteria for breast cancer chemoprevention subjects. J Cell Biochem Suppl. 1993;17G:234-241.
5. Cummings SR, Duong T, Kenyon E, Cauley JA, Whitehead M, Krueger KA. For the Multiple Outcomes of Raloxifene Evaluation (MORE) Trial. Serum estradiol level and risk of breast cancer during treatment with raloxifene. JAMA. 2002;287:216-220.
6. Jordan VC, Allen KE. Evaluation of the antitumor activity of the non-steroidal antioestrogen monohydroxytamoxifen in the DMBA-induced rat mammary carcinoma mode. Eur J Cancer. 1980;16:239-251.
7. Early Breast Cancer Trialists’ Collaborative Group. Effects of adjuvant tamoxifen and of cytotoxic therapy on mortality in early breast cancer. An overview of 61 randomized trials among 28,896 women. N Engl J Med. 1988;319:1681-1692.
8. Bur ME, Zimarowski MJ, Schnitt SJ, Baker S, Lew R. Estrogen receptor immunohistochemistry in carcinoma in situ of the breast. Cancer. 1992;69:1174-1181.
9. Hol T, Cox MB, Bryant HU, Draper MW. Selective estrogen receptor modulators and postmenopausal women’s health. J Womens Health. 1997;6:523-531.
10. Buzdar AU, Marcus C, Holmes F, Hug V, Hortobagyi G. Phase II evaluation of LY156758 in metastatic breast cancer. Oncology. 1988;45:344-345.
11. Neven P, De Muylder X, Van Belle Y, Vanderick G, De Muylder E. Hysteroscopic follow-up during tamoxifen treatment. Eur J Obstet Gynecol Reprod Biol. 1990;35:235-238.
12. Goldstein SR, Scheele WH, Rajagopalan SK, Wilkie JL, Walsh BW, Parsons AK. A 12-month comparative study of raloxifene, estrogen, and placebo on the postmenopausal endometrium. Obstet Gynecol. 2000;95:95-103.
13. Cauley JA, Norton L, Lippman ME, et al. Continued breast cancer risk reduction in postmenopausal women treated with raloxifene: 4-year results from the MORE trial. Breast Cancer Res Treat. 2001;65:125-134.
14. Martino S, Cauley JA, Barrett-Connor E, et al. For the CORE Investigators. Continuing outcomes relevant to Evista: breast cancer incidence in postmenopausal osteoporotic women in a randomized trial of raloxifene. J Natl Cancer Inst. 2004;96:1751-1761.
15. Vogel VG, Costantino JP, Wickerham DL, et al. For the National Surgical Adjuvant Breast and Bowel Project (NSABP). Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 Trial. JAMA. 2006;21:2727-2741.
16. Barrett-Connor E, Mosca L, Collins P, et al. For the Raloxifene Use for The Heart (RUTH) Trial Investigators. Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women. N Engl J Med. 2006;355:125-137.
17. Santen RJ, Yue W, Naftolin F, Mor G, Berstein L. The potential of aromatase inhibitors in breast cancer prevention. Endocr Relat Cancer. 1999;6:235-243.
18. Goss PE, Strasser K. Aromatase inhibitors in the treatment and prevention of breast cancer. J Clin Oncol. 2001;19:881-894.
19. Bryant HU, Dere WH. Selective estrogen receptor modulators: an alternative to hormone replacement therapy. Proc Soc Exp Biol Med. 1998;217:45-52.
20. Grady D, Gebretsadik T, Kerlikowske K, Ernster V, Petitti D. Hormone replacement therapy and endometrial cancer risk: a meta-analysis. Obstet Gynecol. 1995;85:304-313.
21. Miller BA, Feuer EJ, Hankey BF. The significance of the rising incidence of breast cancer in the United States. In: DeVita VT, Hellman S, Rosenberg SA, eds. Important Advances in Oncology. Philadelphia: Lippincott; 1994:193-207.
22. Spicer DV, Pike MC. Risk factors in breast cancer. In: Roses DF, ed. Breast Cancer. New York: Churchill Livingston; 1944.
23. Bilimoria MM, Morrow M. The woman at increased risk for breast cancer: evaluation and management strategies. CA Cancer J Clin. 1995;45:263-278.
The author reports that he is a consultant to Eli Lilly, Pfizer, and Wyeth, and a speaker for Eli Lilly and Wyeth.
CASE 1: Premenopausal woman
at high risk of breast cancer
R. J. is a 43-year-old, nulliparous woman who reached menarche at age 11. She has undergone two breast biopsies, the most recent of which revealed ductal hyperplasia with marked atypia.
R. J.’s sister had breast cancer at 49 years of age; her mother had breast cancer at 66 years. Because of R. J.’s family history, she underwent testing for a BRCA mutation. The result was negative.
R. J. has come to your office today to find out if she can do anything to reduce her risk of breast cancer. What options can you offer?
The most common method of “prevention” of breast cancer involves early detection and assessment of abnormalities through frequent surveillance with mammography. Some women who have dense breasts, a history of breast biopsy, or other risk factors for breast cancer may benefit from intensive surveillance with both mammography and ultrasonography—and, in some cases, magnetic resonance imaging.
More aggressive options include:
- the use of a chemopreventive agent such as tamoxifen or raloxifene
- in rare cases—usually when a BRCA mutation is present—prophylactic mastectomy.
Before it is possible to determine the optimal approach for a particular woman, it is necessary to conduct an individualized assessment of her risk—that is, to estimate the probability that she will develop breast cancer over a defined period of time. Such an estimate is also useful for designing prevention trials in high-risk subsets of the population. (Prevention trials differ from therapeutic clinical trials in that asymptomatic healthy women are exposed to potentially toxic interventions for prolonged periods to reduce their risk of breast cancer.)
This article describes chemopreventive options for women at high risk, based on individualized risk assessment using the Gail model.
(Editor’s note: For additional discussion of the important role ObGyns play in the fight against breast cancer, see Editor in Chief Dr. Robert L. Barbieri’s Editorial.)
You can estimate the likelihood that a woman like your patient may develop breast cancer using various individual risk factors ( TABLE 1 ), but estimates for combinations of risk factors are preferable. The Gail model takes into account some nongenetic factors, such as parity and age at menarche, but also genetic factors, such as family history. The model calculates a woman’s individualized breast cancer probability and yields a numerical risk (a percentage) that she will develop invasive breast cancer over the next 5 years; it also yields an estimate of her risk of developing the malignancy over the remainder of her life.1,2
A Gail-model 5-year estimate of 1.66% or higher denotes a high risk of developing breast cancer. That benchmark was the one employed in the Breast Cancer Prevention Trial (BCPT), conducted as part of the National Surgical Adjuvant Breast and Bowel Project (NSABP).3
TABLE 1
What are the risk factors for breast cancer?
And what degree of relative risk do they confer?
| Relative risk | ||
|---|---|---|
| <2 | 2–4 | >4 |
| • Age 25–34 years at first live birth • Early menarche • Late menopause • Benign proliferative disease • Postmenopausal obesity • Alcohol use • Hormone replacement therapy | • Age >35 years at first live birth • First-degree relative with breast cancer • Nulliparity • Radiation exposure • Personal history of breast cancer | • Gene mutation (BRCA 1 or 2) • Lobular carcinoma in situ • Ductal carcinoma in situ • Atypical hyperplasia |
| Adapted from Bilimoria and Morrow23 | ||
Weaknesses of the Gail model
The Gail model’s approach to estimating risk has some limitations. The model uses the number of prior breast biopsies in its assessment—but the relative risk associated with prior biopsy is smaller for women older than 50 years than it is for younger women.
Furthermore, data on which Gail bases its estimates were collected in the late 1970s and early 1980s. Since then, the increasing ease of breast histopathologic assessment—through fine-needle aspiration and outpatient core-needle biopsy—has confused the issue of just what constitutes a breast “biopsy.” (Most patients surveyed consider it to be any histologic sampling of the breast.)
As a result, the 1.66% cutoff becomes somewhat difficult to interpret in light of current practice.
Consider the following example. A 50-year-old nulliparous Caucasian woman reached menarche when she was 11 years old, has never had a biopsy, and has no first-degree relatives with breast cancer. According to the Gail model, her risk of developing breast cancer is 1.2% over the next 5 years and 10.8% in her lifetime. Therefore, she is not considered at high risk. If she were to give a history of three previous breast biopsies, however, none of them showing hyperplasia, her 5-year risk would rise to 1.8% and push her over the line into the high-risk category.
Compare her situation to that of R. J., the nulliparous woman described in Case 1. R. J. also reached menarche at 11 years, but she has had two breast biopsies (one of which showed atypical hyperplasia) and has two first-degree relatives who have had breast cancer. Her Gail score shows a 5-year risk of breast cancer of 13.5% (the norm for a 43-year-old woman is 0.8%), and a lifetime risk of 69.2%. Clearly, she has a high risk of breast cancer.
How do we improve an imperfect science?
We need to identify objective findings that are patient-specific but highly correlative with the development of breast cancer. Patient-specific biomarkers have been proposed, such as ultrasensitive measurement of the serum estradiol level in postmenopausal women. In the Multiple Outcomes of Raloxifene Evaluation, also known as the MORE trial, women who experienced the greatest reduction in the rate of breast cancer during treatment with raloxifene were a subgroup who had the highest baseline level of serum estradiol—although, overall, all patients had an estradiol level well within the postmenopausal range (≤20 pmol/L).4,5
How tamoxifen became a chemopreventive agent
Tamoxifen inhibits mammary tumors in mice and rats and suppresses hormone-dependent breast cancer cell lines in vitro.6 Clinical data from the Early Breast Cancer Trialists’ Collaborative Group yielded additional motivation for prevention trials with tamoxifen: Besides reducing the rate of recurrent breast cancer, tamoxifen reduced the risk of contralateral new-onset breast cancer by 47% after 5 years of adjuvant treatment.7 Preclinical findings in vitro and in animal models, coupled with clinical data and evidence of tamoxifen’s favorable effects on skeleton remodeling and lipid levels, led to a series of chemoprevention trials in the United States and Europe using tamoxifen.
In the aforementioned BCPT, launched in 1992, 13,388 women 35 years and older who were deemed to be at high risk of developing breast cancer were enrolled at numerous sites throughout the United States and Canada.3 The Gail model was used to select women for the trial—only those who had a 5-year risk of 1.66% or higher were included. Participants were randomly assigned to receive tamoxifen 20 mg or placebo daily for 5 years. The trial was terminated early because of the dramatic reduction in new-onset breast cancer with tamoxifen, compared with placebo.
The overall incidence of breast cancer in the tamoxifen group was 3.4 cases for every 1,000 women, compared with 6.8 cases for every 1,000 women receiving placebo.3 Overall, the reduction in invasive breast cancer with tamoxifen was 49% (P<.00001). When broken down by age group, the reduction was:
- 44% in women 35 to 49 years old
- 51% in women 50 to 59 years old
- 55% in women 60 years and older.
Even noninvasive breast cancer was reduced with tamoxifen
Tamoxifen decreased the incidence of noninvasive breast cancer (ductal carcinoma in situ [DCIS]) by 50%. Expanded use of mammography has increased the detection of DCIS. Most DCIS lesions appear to be estrogen-receptor positive.8
In addition, tamoxifen reduced breast cancer risk in women who had a history of lobular carcinoma in situ (LCIS), a precancer, by 56%, and it reduced the risk of breast cancer in women who had a history of atypical hyperplasia by 86%. Overall, tamoxifen reduced the occurrence of estrogen-positive tumors by 69%, but had no impact on the incidence of estrogen-receptor–negative tumors.
The BCPT was stopped 14 months before planned because the Data and Safety Monitoring Board felt it was unethical to continue to allow one half of such high-risk participants to take placebo in light of the dramatic reduction in both invasive and noninvasive breast cancer among women who took tamoxifen.
In postmenopausal women, tamoxifen increases some risks
Two secondary endpoints of the BCPT are worthy of consideration:
- The overall relative risk (RR) of endometrial cancer associated with tamoxifen therapy in healthy women was 2.53 (95% confidence interval [CI], 1.35, 4.97). However, further analysis by age yielded a RR of 4.01 in women who were older than 50 years (95% CI, 1.70, 10.90), compared with a RR of 1.21 in women 49 years and younger (95% CI, 0.41, 3.60).
- The same age distinction held true for deep venous thrombosis (DVT) and pulmonary embolus, with no statistically significant increases in either in women 49 years and younger, but a RR of 1.71 and 3.19, respectively, in women 50 years and older. It is unclear whether the trial was sufficiently powered for this particular secondary endpoint.
These findings suggest that serious adverse events do not occur at the same magnitude in women younger than 50 years that they do in women 50 and older. The difference in the risk–benefit profile between younger and older women has significant clinical implications for the care of perimenopausal patients.
Risk of other malignancies was not affected by tamoxifen
Overall, invasive cancers other than those of the breast and uterus occurred at the same rate in the tamoxifen and placebo groups of the BCPT. The RR of death from any cause was 0.81 (95% CI, 0.56–1.16). There was a slight increase in the risk of myocardial infarction (RR, 1.11; 95% CI, 0.65–1.92) and a slight decrease in the risk of severe angina (RR, 0.93; 95% CI, 0.40–2.14) in tamoxifen users, although neither of these risks was statistically significant.
The overall RR of fracture of the hip, spine, or radius was 0.81 (95% CI, 0.63–1.05). There was a statistically significant increase in the number of women who had cataracts who then underwent cataract surgery in the tamoxifen group (RR, 1.57; 95% CI, 1.16–2.14).
Tamoxifen is approved as a preventive for high-risk women only
Based on the results of the BCPT, the US Food and Drug Administration (FDA) approved tamoxifen in October 1998 for the primary prevention of breast cancer in women who are at high risk of the disease. The FDA recommends that use of tamoxifen be limited to women at high risk because of the potentially serious side effects seen in clinical trials, including the BCPT.
The FDA did not define “high risk,” but it did recommend that the decision to use tamoxifen as chemopreventive therapy be based on thorough evaluation of the patient’s personal, family, and medical histories; her age; and her understanding of the risks and benefits of treatment.
The FDA also required the following language in the package insert:
- You should not take tamoxifen to reduce the risk of breast cancer unless you are at high risk of breast cancer. Certain conditions put women at high risk, and it is possible to calculate this risk for any woman. Breast cancer risk-assessment tools to help calculate your risk of breast cancer have been developed and are available to your health-care professional. You should discuss your risk with your healthcare professional.
CASE 1 RESOLVED
You determine that R. J. is an excellent candidate for tamoxifen by virtue of her significant risk of breast cancer. You are able to reassure her that, as the BCPT demonstrated, tamoxifen should not increase the risk of uterine cancer, DVT, or pulmonary embolism in a woman her age.
Raloxifene
CASE 2: Patient worries about breasts and bones
S. T. is a 58-year-old Caucasian mother of two whose own mother had breast cancer when she was 74 years old, and whose older sister was given a diagnosis of the malignancy 4 years ago.
S. T. had her first period when she was 11 years old, delivered her first child when she was 31, and entered menopause when she was 52. She is 5 ft 5 in tall and weighs 144 lb.
Her main reason for visiting you today is a breast Mammotome biopsy that showed ductal hyperplasia with atypia. She has been tested for a BRCA mutation, but the result was negative. Her Gail-model score is a 9.7% risk of developing breast cancer over the next 5 years, and a lifetime risk of 44.2%.
She also asks about osteoporosis prevention, given that a dual-energy x-ray absorptiometry (DXA) scan 1 year ago yielded a T-score of –1.3 for her hip and –1.1 for her spine. Her World Health Organization FRAX 10-year risk of hip fracture is 0.7%, and her risk of major osteoporotic fracture is 8.6%.
How do you respond to her concerns?
This patient has a high risk of invasive breast cancer but does not meet criteria for pharmacotherapy for osteoporosis prevention. A good option for her would be raloxifene, a selective estrogen-receptor modulator (SERM) that has been shown to reduce the risk of breast cancer as well as osteoporosis. S. T. would benefit from it on the basis of its breast benefit alone.
The genesis of a drug with multiple benefits
Raloxifene is a benzothiophene derivative, unlike the triphenylethylene family from which tamoxifen is derived. Like tamoxifen, raloxifene was originally investigated as a treatment for advanced breast cancer.
Preclinical studies indicated that raloxifene had an antiproliferative effect on both estrogen-receptor–positive mammary tumors and estrogen-receptor–positive human breast cancer cell lines.9 In the 1980s, however, a small, phase-II trial revealed that raloxifene had no further antitumor effects in postmenopausal women with advanced breast cancer in whom tamoxifen had failed.10 After information surfaced about the neoplastic effect of tamoxifen on the uteri of postmenopausal women, interest in raloxifene revived.11
Raloxifene has estrogen-agonistic activity on bone remodeling and lipid metabolism and was approved by the FDA for prevention of osteoporosis in postmenopausal women in December 1997. Its indication was extended to treatment of osteoporosis 2 years later.
Raloxifene appears to have no effect on the endometrium of postmenopausal women, compared with placebo. In a 12-month comparative trial, there was no difference in endometrial thickness, endoluminal masses, proliferation, or hyperplasia between the raloxifene and placebo groups.12 This finding corroborates earlier evidence that raloxifene does not cause endometrial hyperplasia or cancer and is not associated with vaginal bleeding or increased endometrial thickness, as measured by transvaginal ultrasonography.
A big difference between raloxifene and tamoxifen, therefore, is their varying effect on the uterus of postmenopausal women.
Additional clinical trials confirm anticancer action of raloxifene
Preclinical data in animal models suggested that, like tamoxifen, raloxifene has potent antiestrogenic effects on breast tissue.9 The MORE trial involved 7,705 postmenopausal women up to 80 years old who had established osteoporosis.13 In that trial, participants were randomized to raloxifene or placebo. Bone mineral density (BMD) and fracture incidence were the primary endpoints; breast cancer was a secondary endpoint.
Over the 4 years of the trial, raloxifene significantly reduced the incidence of all invasive breast cancers by 72%, compared with placebo (RR, 0.28; 95% CI, 0.17–0.46). Raloxifene also significantly reduced the incidence of invasive estrogen-receptor–positive tumors by 84%, compared with placebo (RR, 0.16; 95% CI, 0.09–0.30), but had no effect on estrogen-receptor–negative tumors. The incidence of vaginal bleeding, breast pain, and endometrial cancer in the raloxifene group did not differ significantly from that of the placebo group.
Like tamoxifen, raloxifene appeared to be associated with an increased risk of thromboembolic disease, including DVT and pulmonary embolism, which developed in 1.1% of women taking raloxifene, compared with 0.5% of women in the placebo group (P=.003).
In a 4-year continuation of the MORE trial, known as the Continuing Outcomes Relevant to Evista, or CORE, trial, 5,231 women were randomized to continue raloxifene or placebo.14 Over the 8 years of the combined trials, the incidence of invasive breast cancer was reduced by 66% in the raloxifene group (RR, 0.34; 95% CI, 0.22–0.50). The 8-year data are extremely clinically relevant, in that raloxifene has no time limit, whereas tamoxifen is usually prescribed for no longer than 5 years.
Raloxifene is not approved for use in premenopausal women. SERM compounds, which are structurally similar to clomiphene citrate, seem to have different effects in premenopausal and postmenopausal women, as evidenced by tamoxifen’s differing effects by age in the BCPT.
Other investigations of raloxifene confirm its value in high-risk women
To compare the clinical safety and efficacy of tamoxifen and raloxifene in reducing the risk of breast cancer among healthy women, the Study of Tamoxifen and Raloxifene (STAR) was initiated in 1999.15 In that trial, 19,747 postmenopausal women older than 35 years were blindly assigned to raloxifene 60 mg or tamoxifen 20 mg daily.
Baseline characteristics of subjects in STAR are summarized in TABLE 2 . Mean age was 58.5 years. All women had a 5-year risk of developing breast cancer that exceeded 1.66%, according to the Gail model. The average Gail score was 4.03% (standard deviation, ±2.17%). Because it would have been unethical to subject high-risk women to a placebo group in light of the findings of the BCPT, there was no placebo control.
TABLE 2
Baseline characteristics of women
in the Study of Tamoxifen and Raloxifene (STAR) trial
| Characteristic | Value |
|---|---|
| Age (mean) | 58.5 years |
| Caucasian | 93% |
| Hysterectomy | 51% |
| At least one first-degree relative with breast cancer | 71% |
| Lobular carcinoma in situ | 9% |
| Atypical hyperplasia | 23% |
| 5-year risk of invasive breast cancer (mean)* | 4.03% |
| *As estimated with the Gail model Risk Calculator. | |
Here are noteworthy findings of the STAR trial:
- 163 cases of invasive breast cancer occurred in the tamoxifen group, compared with 168 among women taking raloxifene (RR, 1.02; 95% CI, 0.82–1.28).
- 36 cases of uterine cancer occurred in the tamoxifen group, compared with 23 among women taking raloxifene (RR, 0.62; 95% CI, 0.35–1.08). Earlier studies had shown a marked difference in the rate of uterine cancer between these agents. Although the difference here is not statistically significant, uterine cancer was not an endpoint of the study; nor was the study powered to explore this difference.
- The number of hysterectomies among women who were diagnosed with endometrial hyperplasia with or without atypia was, proportionally, significantly higher among women taking tamoxifen ( TABLE 3 ).
- No difference between groups was found for other invasive cancers, ischemic heart events, or stroke.
- Thromboembolic events occurred less frequently in the raloxifene group (RR, 0.70; 95% CI, 0.54–0.91). However, both raloxifene and tamoxifen have consistently been associated with a twofold to threefold increase in the risk of thromboembolic events, compared with placebo.
- Vasomotor symptoms and leg cramps increased in frequency and severity among women in both groups of the trial. These symptoms appear to be less common and less severe among women who are older and more remote from the onset of menopause.
TABLE 3
Relative risk of hysterectomy and uterine hyperplasia during STAR
| Characteristic | Women who took tamoxifen | Women who took raloxifene | Relative risk (95% confidence interval) |
|---|---|---|---|
| Hysterectomy during study | 246 | 92 | 0.37 (0.28, 0.47) |
| Hyperplasia • with atypia • without atypia | 100 15 85 | 17 2 15 | 0.17 (0.09, 0.28) 0.13 (0.01, 0.56) 0.17 (0.09, 0.30) |
What is raloxifene’s effect on the heart?
The Raloxifene Use for The Heart (RUTH) trial explored the primary endpoints of coronary artery disease (CAD) and breast cancer in more than 10,000 women who had CAD or multiple risk factors for it.16 This study began prior to the Women’s Health Initiative, at a time when hormone replacement therapy was widely believed to reduce CAD.
In the double-blinded, randomized, placebo-controlled RUTH trial, raloxifene had no significant effect on primary coronary events (533 vs 553; hazard ratio [HR], 0.95; 95% CI, 0.84–1.07). Even in this population, however, there was a 44% reduction in invasive breast cancer (40 vs 70 events; HR, 0.56; 95% CI, 0.38–0.83).
Based on these results, the FDA approved raloxifene for the “reduction in risk of invasive breast cancer in postmenopausal women at high risk for breast cancer,” as well as for the “reduction in risk of invasive breast cancer in postmenopausal women with osteoporosis” ( FIGURE ).
FIGURE How raloxifene reduced invasive breast cancer in three trials
Raloxifene significantly reduced the risk of cancer, compared with placebo, in the Raloxifene Use for The Heart (RUTH), Multiple Outcomes of Raloxifene Evaluation (MORE), and Continuing Outcomes Relevant to Evista (CORE) trials.
CASE 2 RESOLVED
S. T. begins taking raloxifene 60 mg daily to lower her risk of invasive breast cancer. Although she temporarily experienced hot flashes after initiating the drug, they are only mildly bothersome, and she continues raloxifene therapy. She says she is grateful that there is an agent that can help her reduce the likelihood that she will develop breast cancer, and protection of her BMD is an added benefit.
CASE 3: At risk for both breast cancer and bone fracture
A. N., 63, is a nulliparous Caucasian woman who weighs 134 lb and stands 5 ft 4 in tall. She reached menarche when she was 12 years old and entered menopause at 49.
Although A. N. has never had a breast abnormality, her 59-year-old sister was just given a diagnosis of breast cancer. Her Gail score reveals that she has a 3.1% risk of developing breast cancer over the next 5 years.
In addition to her concerns about breast cancer, A. N. is worried about hip fracture—because her mother suffered one after menopause and because her T-score is –1.9 at the hip and –2.1 at the spine. A. N. has used steroids off and on for much of her life for asthma. Her FRAX score indicates that she has a 2.8% risk of hip fracture and a 25% risk of major osteoporotic fracture over the next 10 years.
What do you offer her?
Because of new FRAX criteria, this osteopenic woman is now a candidate for medication to reduce her risk of major osteoporotic fracture, and raloxifene is a good option. Her Gail score of 3.1% also makes her a good candidate for breast cancer risk reduction with raloxifene.
CASE 3 RESOLVED
Because A. N. needs an agent that benefits both breast and bone, you prescribe raloxifene. The drug should significantly reduce her risk of both invasive breast cancer and bone fracture, without increasing her risk of endometrial hyperplasia and cancer, both of which are associated with tamoxifen in her age group.
Aromatase inhibitors
A fairly new class of drugs being explored for their ability to reduce the risk of breast cancer is aromatase inhibitors. Substantial evidence suggests that estrogens facilitate the development of breast cancer in animals and in women, although the precise mechanism remains unknown.17 The most commonly held theory is that estrogen stimulates proliferation of breast cells and thereby increases the risk of genetic mutation that could lead to cancer.
Aromatase inhibitors block peripheral conversion of androstenedione to estrogens. In premenopausal women, the primary site of this action is in the ovary. In postmenopausal women, this conversion occurs primarily in extraovarian sites, including the adrenal glands, adipose tissue, liver, muscle, and skin.
Aromatase inhibitors may be more effective than SERMs in preventing breast cancer because of their dual role: blocking both the initiation and promotion of breast cancer.18 These agents reduce levels of the genotoxic metabolites of estradiol by lowering estradiol concentration in tissue. At the same time, aromatase inhibitors also block tumor promotion by lowering tissue levels of estrogen and preventing cell proliferation.
The main drawback of these agents—besides the fact that they are not FDA-approved for reducing risk—is their antiestrogenic effect on bone and lipid metabolism. They also induce vasomotor symptoms.
Studies of third-generation aromatase inhibitors in the prevention of breast cancer are under way in high-risk women. These agents include anastrozole, exemestane, and letrozole.
The author reports that he is a consultant to Eli Lilly, Pfizer, and Wyeth, and a speaker for Eli Lilly and Wyeth.
CASE 1: Premenopausal woman
at high risk of breast cancer
R. J. is a 43-year-old, nulliparous woman who reached menarche at age 11. She has undergone two breast biopsies, the most recent of which revealed ductal hyperplasia with marked atypia.
R. J.’s sister had breast cancer at 49 years of age; her mother had breast cancer at 66 years. Because of R. J.’s family history, she underwent testing for a BRCA mutation. The result was negative.
R. J. has come to your office today to find out if she can do anything to reduce her risk of breast cancer. What options can you offer?
The most common method of “prevention” of breast cancer involves early detection and assessment of abnormalities through frequent surveillance with mammography. Some women who have dense breasts, a history of breast biopsy, or other risk factors for breast cancer may benefit from intensive surveillance with both mammography and ultrasonography—and, in some cases, magnetic resonance imaging.
More aggressive options include:
- the use of a chemopreventive agent such as tamoxifen or raloxifene
- in rare cases—usually when a BRCA mutation is present—prophylactic mastectomy.
Before it is possible to determine the optimal approach for a particular woman, it is necessary to conduct an individualized assessment of her risk—that is, to estimate the probability that she will develop breast cancer over a defined period of time. Such an estimate is also useful for designing prevention trials in high-risk subsets of the population. (Prevention trials differ from therapeutic clinical trials in that asymptomatic healthy women are exposed to potentially toxic interventions for prolonged periods to reduce their risk of breast cancer.)
This article describes chemopreventive options for women at high risk, based on individualized risk assessment using the Gail model.
(Editor’s note: For additional discussion of the important role ObGyns play in the fight against breast cancer, see Editor in Chief Dr. Robert L. Barbieri’s Editorial.)
You can estimate the likelihood that a woman like your patient may develop breast cancer using various individual risk factors ( TABLE 1 ), but estimates for combinations of risk factors are preferable. The Gail model takes into account some nongenetic factors, such as parity and age at menarche, but also genetic factors, such as family history. The model calculates a woman’s individualized breast cancer probability and yields a numerical risk (a percentage) that she will develop invasive breast cancer over the next 5 years; it also yields an estimate of her risk of developing the malignancy over the remainder of her life.1,2
A Gail-model 5-year estimate of 1.66% or higher denotes a high risk of developing breast cancer. That benchmark was the one employed in the Breast Cancer Prevention Trial (BCPT), conducted as part of the National Surgical Adjuvant Breast and Bowel Project (NSABP).3
TABLE 1
What are the risk factors for breast cancer?
And what degree of relative risk do they confer?
| Relative risk | ||
|---|---|---|
| <2 | 2–4 | >4 |
| • Age 25–34 years at first live birth • Early menarche • Late menopause • Benign proliferative disease • Postmenopausal obesity • Alcohol use • Hormone replacement therapy | • Age >35 years at first live birth • First-degree relative with breast cancer • Nulliparity • Radiation exposure • Personal history of breast cancer | • Gene mutation (BRCA 1 or 2) • Lobular carcinoma in situ • Ductal carcinoma in situ • Atypical hyperplasia |
| Adapted from Bilimoria and Morrow23 | ||
Weaknesses of the Gail model
The Gail model’s approach to estimating risk has some limitations. The model uses the number of prior breast biopsies in its assessment—but the relative risk associated with prior biopsy is smaller for women older than 50 years than it is for younger women.
Furthermore, data on which Gail bases its estimates were collected in the late 1970s and early 1980s. Since then, the increasing ease of breast histopathologic assessment—through fine-needle aspiration and outpatient core-needle biopsy—has confused the issue of just what constitutes a breast “biopsy.” (Most patients surveyed consider it to be any histologic sampling of the breast.)
As a result, the 1.66% cutoff becomes somewhat difficult to interpret in light of current practice.
Consider the following example. A 50-year-old nulliparous Caucasian woman reached menarche when she was 11 years old, has never had a biopsy, and has no first-degree relatives with breast cancer. According to the Gail model, her risk of developing breast cancer is 1.2% over the next 5 years and 10.8% in her lifetime. Therefore, she is not considered at high risk. If she were to give a history of three previous breast biopsies, however, none of them showing hyperplasia, her 5-year risk would rise to 1.8% and push her over the line into the high-risk category.
Compare her situation to that of R. J., the nulliparous woman described in Case 1. R. J. also reached menarche at 11 years, but she has had two breast biopsies (one of which showed atypical hyperplasia) and has two first-degree relatives who have had breast cancer. Her Gail score shows a 5-year risk of breast cancer of 13.5% (the norm for a 43-year-old woman is 0.8%), and a lifetime risk of 69.2%. Clearly, she has a high risk of breast cancer.
How do we improve an imperfect science?
We need to identify objective findings that are patient-specific but highly correlative with the development of breast cancer. Patient-specific biomarkers have been proposed, such as ultrasensitive measurement of the serum estradiol level in postmenopausal women. In the Multiple Outcomes of Raloxifene Evaluation, also known as the MORE trial, women who experienced the greatest reduction in the rate of breast cancer during treatment with raloxifene were a subgroup who had the highest baseline level of serum estradiol—although, overall, all patients had an estradiol level well within the postmenopausal range (≤20 pmol/L).4,5
How tamoxifen became a chemopreventive agent
Tamoxifen inhibits mammary tumors in mice and rats and suppresses hormone-dependent breast cancer cell lines in vitro.6 Clinical data from the Early Breast Cancer Trialists’ Collaborative Group yielded additional motivation for prevention trials with tamoxifen: Besides reducing the rate of recurrent breast cancer, tamoxifen reduced the risk of contralateral new-onset breast cancer by 47% after 5 years of adjuvant treatment.7 Preclinical findings in vitro and in animal models, coupled with clinical data and evidence of tamoxifen’s favorable effects on skeleton remodeling and lipid levels, led to a series of chemoprevention trials in the United States and Europe using tamoxifen.
In the aforementioned BCPT, launched in 1992, 13,388 women 35 years and older who were deemed to be at high risk of developing breast cancer were enrolled at numerous sites throughout the United States and Canada.3 The Gail model was used to select women for the trial—only those who had a 5-year risk of 1.66% or higher were included. Participants were randomly assigned to receive tamoxifen 20 mg or placebo daily for 5 years. The trial was terminated early because of the dramatic reduction in new-onset breast cancer with tamoxifen, compared with placebo.
The overall incidence of breast cancer in the tamoxifen group was 3.4 cases for every 1,000 women, compared with 6.8 cases for every 1,000 women receiving placebo.3 Overall, the reduction in invasive breast cancer with tamoxifen was 49% (P<.00001). When broken down by age group, the reduction was:
- 44% in women 35 to 49 years old
- 51% in women 50 to 59 years old
- 55% in women 60 years and older.
Even noninvasive breast cancer was reduced with tamoxifen
Tamoxifen decreased the incidence of noninvasive breast cancer (ductal carcinoma in situ [DCIS]) by 50%. Expanded use of mammography has increased the detection of DCIS. Most DCIS lesions appear to be estrogen-receptor positive.8
In addition, tamoxifen reduced breast cancer risk in women who had a history of lobular carcinoma in situ (LCIS), a precancer, by 56%, and it reduced the risk of breast cancer in women who had a history of atypical hyperplasia by 86%. Overall, tamoxifen reduced the occurrence of estrogen-positive tumors by 69%, but had no impact on the incidence of estrogen-receptor–negative tumors.
The BCPT was stopped 14 months before planned because the Data and Safety Monitoring Board felt it was unethical to continue to allow one half of such high-risk participants to take placebo in light of the dramatic reduction in both invasive and noninvasive breast cancer among women who took tamoxifen.
In postmenopausal women, tamoxifen increases some risks
Two secondary endpoints of the BCPT are worthy of consideration:
- The overall relative risk (RR) of endometrial cancer associated with tamoxifen therapy in healthy women was 2.53 (95% confidence interval [CI], 1.35, 4.97). However, further analysis by age yielded a RR of 4.01 in women who were older than 50 years (95% CI, 1.70, 10.90), compared with a RR of 1.21 in women 49 years and younger (95% CI, 0.41, 3.60).
- The same age distinction held true for deep venous thrombosis (DVT) and pulmonary embolus, with no statistically significant increases in either in women 49 years and younger, but a RR of 1.71 and 3.19, respectively, in women 50 years and older. It is unclear whether the trial was sufficiently powered for this particular secondary endpoint.
These findings suggest that serious adverse events do not occur at the same magnitude in women younger than 50 years that they do in women 50 and older. The difference in the risk–benefit profile between younger and older women has significant clinical implications for the care of perimenopausal patients.
Risk of other malignancies was not affected by tamoxifen
Overall, invasive cancers other than those of the breast and uterus occurred at the same rate in the tamoxifen and placebo groups of the BCPT. The RR of death from any cause was 0.81 (95% CI, 0.56–1.16). There was a slight increase in the risk of myocardial infarction (RR, 1.11; 95% CI, 0.65–1.92) and a slight decrease in the risk of severe angina (RR, 0.93; 95% CI, 0.40–2.14) in tamoxifen users, although neither of these risks was statistically significant.
The overall RR of fracture of the hip, spine, or radius was 0.81 (95% CI, 0.63–1.05). There was a statistically significant increase in the number of women who had cataracts who then underwent cataract surgery in the tamoxifen group (RR, 1.57; 95% CI, 1.16–2.14).
Tamoxifen is approved as a preventive for high-risk women only
Based on the results of the BCPT, the US Food and Drug Administration (FDA) approved tamoxifen in October 1998 for the primary prevention of breast cancer in women who are at high risk of the disease. The FDA recommends that use of tamoxifen be limited to women at high risk because of the potentially serious side effects seen in clinical trials, including the BCPT.
The FDA did not define “high risk,” but it did recommend that the decision to use tamoxifen as chemopreventive therapy be based on thorough evaluation of the patient’s personal, family, and medical histories; her age; and her understanding of the risks and benefits of treatment.
The FDA also required the following language in the package insert:
- You should not take tamoxifen to reduce the risk of breast cancer unless you are at high risk of breast cancer. Certain conditions put women at high risk, and it is possible to calculate this risk for any woman. Breast cancer risk-assessment tools to help calculate your risk of breast cancer have been developed and are available to your health-care professional. You should discuss your risk with your healthcare professional.
CASE 1 RESOLVED
You determine that R. J. is an excellent candidate for tamoxifen by virtue of her significant risk of breast cancer. You are able to reassure her that, as the BCPT demonstrated, tamoxifen should not increase the risk of uterine cancer, DVT, or pulmonary embolism in a woman her age.
Raloxifene
CASE 2: Patient worries about breasts and bones
S. T. is a 58-year-old Caucasian mother of two whose own mother had breast cancer when she was 74 years old, and whose older sister was given a diagnosis of the malignancy 4 years ago.
S. T. had her first period when she was 11 years old, delivered her first child when she was 31, and entered menopause when she was 52. She is 5 ft 5 in tall and weighs 144 lb.
Her main reason for visiting you today is a breast Mammotome biopsy that showed ductal hyperplasia with atypia. She has been tested for a BRCA mutation, but the result was negative. Her Gail-model score is a 9.7% risk of developing breast cancer over the next 5 years, and a lifetime risk of 44.2%.
She also asks about osteoporosis prevention, given that a dual-energy x-ray absorptiometry (DXA) scan 1 year ago yielded a T-score of –1.3 for her hip and –1.1 for her spine. Her World Health Organization FRAX 10-year risk of hip fracture is 0.7%, and her risk of major osteoporotic fracture is 8.6%.
How do you respond to her concerns?
This patient has a high risk of invasive breast cancer but does not meet criteria for pharmacotherapy for osteoporosis prevention. A good option for her would be raloxifene, a selective estrogen-receptor modulator (SERM) that has been shown to reduce the risk of breast cancer as well as osteoporosis. S. T. would benefit from it on the basis of its breast benefit alone.
The genesis of a drug with multiple benefits
Raloxifene is a benzothiophene derivative, unlike the triphenylethylene family from which tamoxifen is derived. Like tamoxifen, raloxifene was originally investigated as a treatment for advanced breast cancer.
Preclinical studies indicated that raloxifene had an antiproliferative effect on both estrogen-receptor–positive mammary tumors and estrogen-receptor–positive human breast cancer cell lines.9 In the 1980s, however, a small, phase-II trial revealed that raloxifene had no further antitumor effects in postmenopausal women with advanced breast cancer in whom tamoxifen had failed.10 After information surfaced about the neoplastic effect of tamoxifen on the uteri of postmenopausal women, interest in raloxifene revived.11
Raloxifene has estrogen-agonistic activity on bone remodeling and lipid metabolism and was approved by the FDA for prevention of osteoporosis in postmenopausal women in December 1997. Its indication was extended to treatment of osteoporosis 2 years later.
Raloxifene appears to have no effect on the endometrium of postmenopausal women, compared with placebo. In a 12-month comparative trial, there was no difference in endometrial thickness, endoluminal masses, proliferation, or hyperplasia between the raloxifene and placebo groups.12 This finding corroborates earlier evidence that raloxifene does not cause endometrial hyperplasia or cancer and is not associated with vaginal bleeding or increased endometrial thickness, as measured by transvaginal ultrasonography.
A big difference between raloxifene and tamoxifen, therefore, is their varying effect on the uterus of postmenopausal women.
Additional clinical trials confirm anticancer action of raloxifene
Preclinical data in animal models suggested that, like tamoxifen, raloxifene has potent antiestrogenic effects on breast tissue.9 The MORE trial involved 7,705 postmenopausal women up to 80 years old who had established osteoporosis.13 In that trial, participants were randomized to raloxifene or placebo. Bone mineral density (BMD) and fracture incidence were the primary endpoints; breast cancer was a secondary endpoint.
Over the 4 years of the trial, raloxifene significantly reduced the incidence of all invasive breast cancers by 72%, compared with placebo (RR, 0.28; 95% CI, 0.17–0.46). Raloxifene also significantly reduced the incidence of invasive estrogen-receptor–positive tumors by 84%, compared with placebo (RR, 0.16; 95% CI, 0.09–0.30), but had no effect on estrogen-receptor–negative tumors. The incidence of vaginal bleeding, breast pain, and endometrial cancer in the raloxifene group did not differ significantly from that of the placebo group.
Like tamoxifen, raloxifene appeared to be associated with an increased risk of thromboembolic disease, including DVT and pulmonary embolism, which developed in 1.1% of women taking raloxifene, compared with 0.5% of women in the placebo group (P=.003).
In a 4-year continuation of the MORE trial, known as the Continuing Outcomes Relevant to Evista, or CORE, trial, 5,231 women were randomized to continue raloxifene or placebo.14 Over the 8 years of the combined trials, the incidence of invasive breast cancer was reduced by 66% in the raloxifene group (RR, 0.34; 95% CI, 0.22–0.50). The 8-year data are extremely clinically relevant, in that raloxifene has no time limit, whereas tamoxifen is usually prescribed for no longer than 5 years.
Raloxifene is not approved for use in premenopausal women. SERM compounds, which are structurally similar to clomiphene citrate, seem to have different effects in premenopausal and postmenopausal women, as evidenced by tamoxifen’s differing effects by age in the BCPT.
Other investigations of raloxifene confirm its value in high-risk women
To compare the clinical safety and efficacy of tamoxifen and raloxifene in reducing the risk of breast cancer among healthy women, the Study of Tamoxifen and Raloxifene (STAR) was initiated in 1999.15 In that trial, 19,747 postmenopausal women older than 35 years were blindly assigned to raloxifene 60 mg or tamoxifen 20 mg daily.
Baseline characteristics of subjects in STAR are summarized in TABLE 2 . Mean age was 58.5 years. All women had a 5-year risk of developing breast cancer that exceeded 1.66%, according to the Gail model. The average Gail score was 4.03% (standard deviation, ±2.17%). Because it would have been unethical to subject high-risk women to a placebo group in light of the findings of the BCPT, there was no placebo control.
TABLE 2
Baseline characteristics of women
in the Study of Tamoxifen and Raloxifene (STAR) trial
| Characteristic | Value |
|---|---|
| Age (mean) | 58.5 years |
| Caucasian | 93% |
| Hysterectomy | 51% |
| At least one first-degree relative with breast cancer | 71% |
| Lobular carcinoma in situ | 9% |
| Atypical hyperplasia | 23% |
| 5-year risk of invasive breast cancer (mean)* | 4.03% |
| *As estimated with the Gail model Risk Calculator. | |
Here are noteworthy findings of the STAR trial:
- 163 cases of invasive breast cancer occurred in the tamoxifen group, compared with 168 among women taking raloxifene (RR, 1.02; 95% CI, 0.82–1.28).
- 36 cases of uterine cancer occurred in the tamoxifen group, compared with 23 among women taking raloxifene (RR, 0.62; 95% CI, 0.35–1.08). Earlier studies had shown a marked difference in the rate of uterine cancer between these agents. Although the difference here is not statistically significant, uterine cancer was not an endpoint of the study; nor was the study powered to explore this difference.
- The number of hysterectomies among women who were diagnosed with endometrial hyperplasia with or without atypia was, proportionally, significantly higher among women taking tamoxifen ( TABLE 3 ).
- No difference between groups was found for other invasive cancers, ischemic heart events, or stroke.
- Thromboembolic events occurred less frequently in the raloxifene group (RR, 0.70; 95% CI, 0.54–0.91). However, both raloxifene and tamoxifen have consistently been associated with a twofold to threefold increase in the risk of thromboembolic events, compared with placebo.
- Vasomotor symptoms and leg cramps increased in frequency and severity among women in both groups of the trial. These symptoms appear to be less common and less severe among women who are older and more remote from the onset of menopause.
TABLE 3
Relative risk of hysterectomy and uterine hyperplasia during STAR
| Characteristic | Women who took tamoxifen | Women who took raloxifene | Relative risk (95% confidence interval) |
|---|---|---|---|
| Hysterectomy during study | 246 | 92 | 0.37 (0.28, 0.47) |
| Hyperplasia • with atypia • without atypia | 100 15 85 | 17 2 15 | 0.17 (0.09, 0.28) 0.13 (0.01, 0.56) 0.17 (0.09, 0.30) |
What is raloxifene’s effect on the heart?
The Raloxifene Use for The Heart (RUTH) trial explored the primary endpoints of coronary artery disease (CAD) and breast cancer in more than 10,000 women who had CAD or multiple risk factors for it.16 This study began prior to the Women’s Health Initiative, at a time when hormone replacement therapy was widely believed to reduce CAD.
In the double-blinded, randomized, placebo-controlled RUTH trial, raloxifene had no significant effect on primary coronary events (533 vs 553; hazard ratio [HR], 0.95; 95% CI, 0.84–1.07). Even in this population, however, there was a 44% reduction in invasive breast cancer (40 vs 70 events; HR, 0.56; 95% CI, 0.38–0.83).
Based on these results, the FDA approved raloxifene for the “reduction in risk of invasive breast cancer in postmenopausal women at high risk for breast cancer,” as well as for the “reduction in risk of invasive breast cancer in postmenopausal women with osteoporosis” ( FIGURE ).
FIGURE How raloxifene reduced invasive breast cancer in three trials
Raloxifene significantly reduced the risk of cancer, compared with placebo, in the Raloxifene Use for The Heart (RUTH), Multiple Outcomes of Raloxifene Evaluation (MORE), and Continuing Outcomes Relevant to Evista (CORE) trials.
CASE 2 RESOLVED
S. T. begins taking raloxifene 60 mg daily to lower her risk of invasive breast cancer. Although she temporarily experienced hot flashes after initiating the drug, they are only mildly bothersome, and she continues raloxifene therapy. She says she is grateful that there is an agent that can help her reduce the likelihood that she will develop breast cancer, and protection of her BMD is an added benefit.
CASE 3: At risk for both breast cancer and bone fracture
A. N., 63, is a nulliparous Caucasian woman who weighs 134 lb and stands 5 ft 4 in tall. She reached menarche when she was 12 years old and entered menopause at 49.
Although A. N. has never had a breast abnormality, her 59-year-old sister was just given a diagnosis of breast cancer. Her Gail score reveals that she has a 3.1% risk of developing breast cancer over the next 5 years.
In addition to her concerns about breast cancer, A. N. is worried about hip fracture—because her mother suffered one after menopause and because her T-score is –1.9 at the hip and –2.1 at the spine. A. N. has used steroids off and on for much of her life for asthma. Her FRAX score indicates that she has a 2.8% risk of hip fracture and a 25% risk of major osteoporotic fracture over the next 10 years.
What do you offer her?
Because of new FRAX criteria, this osteopenic woman is now a candidate for medication to reduce her risk of major osteoporotic fracture, and raloxifene is a good option. Her Gail score of 3.1% also makes her a good candidate for breast cancer risk reduction with raloxifene.
CASE 3 RESOLVED
Because A. N. needs an agent that benefits both breast and bone, you prescribe raloxifene. The drug should significantly reduce her risk of both invasive breast cancer and bone fracture, without increasing her risk of endometrial hyperplasia and cancer, both of which are associated with tamoxifen in her age group.
Aromatase inhibitors
A fairly new class of drugs being explored for their ability to reduce the risk of breast cancer is aromatase inhibitors. Substantial evidence suggests that estrogens facilitate the development of breast cancer in animals and in women, although the precise mechanism remains unknown.17 The most commonly held theory is that estrogen stimulates proliferation of breast cells and thereby increases the risk of genetic mutation that could lead to cancer.
Aromatase inhibitors block peripheral conversion of androstenedione to estrogens. In premenopausal women, the primary site of this action is in the ovary. In postmenopausal women, this conversion occurs primarily in extraovarian sites, including the adrenal glands, adipose tissue, liver, muscle, and skin.
Aromatase inhibitors may be more effective than SERMs in preventing breast cancer because of their dual role: blocking both the initiation and promotion of breast cancer.18 These agents reduce levels of the genotoxic metabolites of estradiol by lowering estradiol concentration in tissue. At the same time, aromatase inhibitors also block tumor promotion by lowering tissue levels of estrogen and preventing cell proliferation.
The main drawback of these agents—besides the fact that they are not FDA-approved for reducing risk—is their antiestrogenic effect on bone and lipid metabolism. They also induce vasomotor symptoms.
Studies of third-generation aromatase inhibitors in the prevention of breast cancer are under way in high-risk women. These agents include anastrozole, exemestane, and letrozole.
1. Gail MH, Brinton LA, Byar DP, et al. Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl Cancer Inst. 1989;81:1879-1886.
2. Breast Cancer Assessment Tool. Available at: www.cancer.gov/bcrisktool/Default.aspx. Accessed June 5, 2009.
3. Fisher B, Costantino JP, Wickerham DL, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 study. J Natl Cancer Inst. 1998;90:1371-1388.
4. Ruffin MT, 4th, August DA, Kelloff GJ, Boone CW, Weber BL, Brenner DE. Selection criteria for breast cancer chemoprevention subjects. J Cell Biochem Suppl. 1993;17G:234-241.
5. Cummings SR, Duong T, Kenyon E, Cauley JA, Whitehead M, Krueger KA. For the Multiple Outcomes of Raloxifene Evaluation (MORE) Trial. Serum estradiol level and risk of breast cancer during treatment with raloxifene. JAMA. 2002;287:216-220.
6. Jordan VC, Allen KE. Evaluation of the antitumor activity of the non-steroidal antioestrogen monohydroxytamoxifen in the DMBA-induced rat mammary carcinoma mode. Eur J Cancer. 1980;16:239-251.
7. Early Breast Cancer Trialists’ Collaborative Group. Effects of adjuvant tamoxifen and of cytotoxic therapy on mortality in early breast cancer. An overview of 61 randomized trials among 28,896 women. N Engl J Med. 1988;319:1681-1692.
8. Bur ME, Zimarowski MJ, Schnitt SJ, Baker S, Lew R. Estrogen receptor immunohistochemistry in carcinoma in situ of the breast. Cancer. 1992;69:1174-1181.
9. Hol T, Cox MB, Bryant HU, Draper MW. Selective estrogen receptor modulators and postmenopausal women’s health. J Womens Health. 1997;6:523-531.
10. Buzdar AU, Marcus C, Holmes F, Hug V, Hortobagyi G. Phase II evaluation of LY156758 in metastatic breast cancer. Oncology. 1988;45:344-345.
11. Neven P, De Muylder X, Van Belle Y, Vanderick G, De Muylder E. Hysteroscopic follow-up during tamoxifen treatment. Eur J Obstet Gynecol Reprod Biol. 1990;35:235-238.
12. Goldstein SR, Scheele WH, Rajagopalan SK, Wilkie JL, Walsh BW, Parsons AK. A 12-month comparative study of raloxifene, estrogen, and placebo on the postmenopausal endometrium. Obstet Gynecol. 2000;95:95-103.
13. Cauley JA, Norton L, Lippman ME, et al. Continued breast cancer risk reduction in postmenopausal women treated with raloxifene: 4-year results from the MORE trial. Breast Cancer Res Treat. 2001;65:125-134.
14. Martino S, Cauley JA, Barrett-Connor E, et al. For the CORE Investigators. Continuing outcomes relevant to Evista: breast cancer incidence in postmenopausal osteoporotic women in a randomized trial of raloxifene. J Natl Cancer Inst. 2004;96:1751-1761.
15. Vogel VG, Costantino JP, Wickerham DL, et al. For the National Surgical Adjuvant Breast and Bowel Project (NSABP). Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 Trial. JAMA. 2006;21:2727-2741.
16. Barrett-Connor E, Mosca L, Collins P, et al. For the Raloxifene Use for The Heart (RUTH) Trial Investigators. Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women. N Engl J Med. 2006;355:125-137.
17. Santen RJ, Yue W, Naftolin F, Mor G, Berstein L. The potential of aromatase inhibitors in breast cancer prevention. Endocr Relat Cancer. 1999;6:235-243.
18. Goss PE, Strasser K. Aromatase inhibitors in the treatment and prevention of breast cancer. J Clin Oncol. 2001;19:881-894.
19. Bryant HU, Dere WH. Selective estrogen receptor modulators: an alternative to hormone replacement therapy. Proc Soc Exp Biol Med. 1998;217:45-52.
20. Grady D, Gebretsadik T, Kerlikowske K, Ernster V, Petitti D. Hormone replacement therapy and endometrial cancer risk: a meta-analysis. Obstet Gynecol. 1995;85:304-313.
21. Miller BA, Feuer EJ, Hankey BF. The significance of the rising incidence of breast cancer in the United States. In: DeVita VT, Hellman S, Rosenberg SA, eds. Important Advances in Oncology. Philadelphia: Lippincott; 1994:193-207.
22. Spicer DV, Pike MC. Risk factors in breast cancer. In: Roses DF, ed. Breast Cancer. New York: Churchill Livingston; 1944.
23. Bilimoria MM, Morrow M. The woman at increased risk for breast cancer: evaluation and management strategies. CA Cancer J Clin. 1995;45:263-278.
1. Gail MH, Brinton LA, Byar DP, et al. Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl Cancer Inst. 1989;81:1879-1886.
2. Breast Cancer Assessment Tool. Available at: www.cancer.gov/bcrisktool/Default.aspx. Accessed June 5, 2009.
3. Fisher B, Costantino JP, Wickerham DL, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 study. J Natl Cancer Inst. 1998;90:1371-1388.
4. Ruffin MT, 4th, August DA, Kelloff GJ, Boone CW, Weber BL, Brenner DE. Selection criteria for breast cancer chemoprevention subjects. J Cell Biochem Suppl. 1993;17G:234-241.
5. Cummings SR, Duong T, Kenyon E, Cauley JA, Whitehead M, Krueger KA. For the Multiple Outcomes of Raloxifene Evaluation (MORE) Trial. Serum estradiol level and risk of breast cancer during treatment with raloxifene. JAMA. 2002;287:216-220.
6. Jordan VC, Allen KE. Evaluation of the antitumor activity of the non-steroidal antioestrogen monohydroxytamoxifen in the DMBA-induced rat mammary carcinoma mode. Eur J Cancer. 1980;16:239-251.
7. Early Breast Cancer Trialists’ Collaborative Group. Effects of adjuvant tamoxifen and of cytotoxic therapy on mortality in early breast cancer. An overview of 61 randomized trials among 28,896 women. N Engl J Med. 1988;319:1681-1692.
8. Bur ME, Zimarowski MJ, Schnitt SJ, Baker S, Lew R. Estrogen receptor immunohistochemistry in carcinoma in situ of the breast. Cancer. 1992;69:1174-1181.
9. Hol T, Cox MB, Bryant HU, Draper MW. Selective estrogen receptor modulators and postmenopausal women’s health. J Womens Health. 1997;6:523-531.
10. Buzdar AU, Marcus C, Holmes F, Hug V, Hortobagyi G. Phase II evaluation of LY156758 in metastatic breast cancer. Oncology. 1988;45:344-345.
11. Neven P, De Muylder X, Van Belle Y, Vanderick G, De Muylder E. Hysteroscopic follow-up during tamoxifen treatment. Eur J Obstet Gynecol Reprod Biol. 1990;35:235-238.
12. Goldstein SR, Scheele WH, Rajagopalan SK, Wilkie JL, Walsh BW, Parsons AK. A 12-month comparative study of raloxifene, estrogen, and placebo on the postmenopausal endometrium. Obstet Gynecol. 2000;95:95-103.
13. Cauley JA, Norton L, Lippman ME, et al. Continued breast cancer risk reduction in postmenopausal women treated with raloxifene: 4-year results from the MORE trial. Breast Cancer Res Treat. 2001;65:125-134.
14. Martino S, Cauley JA, Barrett-Connor E, et al. For the CORE Investigators. Continuing outcomes relevant to Evista: breast cancer incidence in postmenopausal osteoporotic women in a randomized trial of raloxifene. J Natl Cancer Inst. 2004;96:1751-1761.
15. Vogel VG, Costantino JP, Wickerham DL, et al. For the National Surgical Adjuvant Breast and Bowel Project (NSABP). Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 Trial. JAMA. 2006;21:2727-2741.
16. Barrett-Connor E, Mosca L, Collins P, et al. For the Raloxifene Use for The Heart (RUTH) Trial Investigators. Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women. N Engl J Med. 2006;355:125-137.
17. Santen RJ, Yue W, Naftolin F, Mor G, Berstein L. The potential of aromatase inhibitors in breast cancer prevention. Endocr Relat Cancer. 1999;6:235-243.
18. Goss PE, Strasser K. Aromatase inhibitors in the treatment and prevention of breast cancer. J Clin Oncol. 2001;19:881-894.
19. Bryant HU, Dere WH. Selective estrogen receptor modulators: an alternative to hormone replacement therapy. Proc Soc Exp Biol Med. 1998;217:45-52.
20. Grady D, Gebretsadik T, Kerlikowske K, Ernster V, Petitti D. Hormone replacement therapy and endometrial cancer risk: a meta-analysis. Obstet Gynecol. 1995;85:304-313.
21. Miller BA, Feuer EJ, Hankey BF. The significance of the rising incidence of breast cancer in the United States. In: DeVita VT, Hellman S, Rosenberg SA, eds. Important Advances in Oncology. Philadelphia: Lippincott; 1994:193-207.
22. Spicer DV, Pike MC. Risk factors in breast cancer. In: Roses DF, ed. Breast Cancer. New York: Churchill Livingston; 1944.
23. Bilimoria MM, Morrow M. The woman at increased risk for breast cancer: evaluation and management strategies. CA Cancer J Clin. 1995;45:263-278.
Is ovarian Ca screening effective in postmenopausal women?
Ovarian cancer is relatively rare, but late diagnosis leads to a higher death rate than the death rate observed with other gynecologic malignancies. Detection at an early stage (I or II) is associated with higher survival than detection at a later stage (III or IV).
Earlier studies have suggested that ovarian cancer screening with the serum biomarker CA 125 and transvaginal ultrasonography (TVS) may help the clinician diagnose ovarian cancer at an earlier stage.
British study finds screening to be effective
In the United Kingdom Collaborative Trial of Ovarian Cancer Screening, postmenopausal women 50 to 74 years old were randomized to one of the following:
- no screening (n=101,359)
- annual assessment of CA 125 using a proprietary risk-of-cancer algorithm, with TVS as a second-line test (multimodal screening, or MMS) (n=50,078)
- annual TVS in a 2:1:1 ratio (n=48,230). (A scan was considered abnormal when one or both ovaries had complex morphology or a simple cyst exceeded 60 cm3 in size.)
The mean age at screening was 60.6 years, and 96.5% of participants were white.
Women who underwent MMS were triaged according to their estimated risk of developing ovarian cancer, based on the CA 125 level and age-specific risk estimates. Women in the group with the lowest risk of ovarian cancer continued annual assessment of CA 125, whereas those at highest risk underwent repeat measurement of CA 125, followed by TVS if the repeat assay suggested elevated risk. If TVS findings were also abnormal, the patient underwent clinical evaluation.
In the MMS and TVS groups, 8.7% and 12.0% of subjects, respectively, underwent clinical assessment, and surgery was performed in 0.2% and 1.8%, respectively. A primary ovarian or tubal cancer was diagnosed in 42 and 45 women in the MMS and TVS groups, respectively. In addition, 8 (MMS) and 20 (TVS) borderline malignancies were identified.
Overall, 48.3% of invasive cancers were diagnosed during stage I or II, with no difference in the distribution of stages between MMS and TVS groups. The positive predictive value (PPV) of MMS and TVS for primary invasive epithelial and tubal cancers was 35.1% and 2.8%, respectively. The ratio of surgery to case of invasive ovarian cancer was 2.9:1 (MMS) and 35:1 (TVS).
In the US study, women underwent both CA 125 assessment and TVS
The US study involved four annual rounds of screening in women 55 to 74 years old, who underwent both CA 125 measurement and TVS imaging in each round. A CA 125 level of 35 U/mL or above, or ovarian volume greater than 10 cm3 (or detection of an ovarian cyst with complex morphology) was considered abnormal.
Of 34,621 women randomized to screening, 30,630 underwent at least one screen during the four rounds. Almost two thirds of participants were 55 to 64 years old, and almost 89% were non-Hispanic white.
The percentage of women who had at least one positive screen decreased over the four rounds of screening, from 5.8% in year 1 to 4.9%, 4.6%, and 4.5% in years 2 through 4, respectively. In each round of screening, TVS was more likely to be positive than was CA 125 measurement (e.g., 4.6% vs 1.4% in the first round).
Of the 28,746 women who underwent the initial (prevalence) screen, 1,675 (5.8%) had positive findings, 566 (1.97% of those who were screened) underwent surgery, and 27 neoplasms were detected (0.06% of those who were screened), including 18 ovarian or primary peritoneal invasive cancers. Nine borderline tumors were also identified.
The PPV of a positive screen in the first round was 1.1%. Of the 18 invasive cancers identified in this round, 16.7% were diagnosed during stage I or II, and 83.4% were identified during stage III or IV. The ratio of surgery to cases of invasive ovarian cancer was 31.4:1.
Why did the trials have different findings?
The findings of the UK study suggest that an MMS strategy of CA 125 assessment, followed selectively by TVS, can detect early-stage ovarian cancer with an acceptable PPV and ratio of surgery to case of invasive cancer.
In contrast, the US findings are discouraging because of the low PPV, the large percentage of malignancies detected at an advanced stage, and the high ratio of surgery to cases of invasive cancer.
Although the studies had different primary outcomes—ovarian and tubal cancers in the UK and ovarian and primary peritoneal cancers in the US—a majority of invasive malignancies detected in both studies were ovarian.
In the US study, the poor performance of screening may be due, in part, to universal rather than selective use of TVS; that modality generates substantially more false positives than does CA 125. The US study also defined an ovarian abnormality more broadly (volume greater than 10 cm3) than the British study did (volume greater than 60 cm3), which may have lowered the PPV in the US trial.
As investigators in the UK continue to follow participants and report their findings, we will learn more about the value of MMS, including its impact on cancer mortality, possibly as soon as 2014.
Until we have further data on the value of ovarian cancer screening in postmenopausal women, we should continue the current practice of screening only symptomatic or very-high-risk women for ovarian cancer.—ANDREW M. KAUNITZ, MD
Ovarian cancer is relatively rare, but late diagnosis leads to a higher death rate than the death rate observed with other gynecologic malignancies. Detection at an early stage (I or II) is associated with higher survival than detection at a later stage (III or IV).
Earlier studies have suggested that ovarian cancer screening with the serum biomarker CA 125 and transvaginal ultrasonography (TVS) may help the clinician diagnose ovarian cancer at an earlier stage.
British study finds screening to be effective
In the United Kingdom Collaborative Trial of Ovarian Cancer Screening, postmenopausal women 50 to 74 years old were randomized to one of the following:
- no screening (n=101,359)
- annual assessment of CA 125 using a proprietary risk-of-cancer algorithm, with TVS as a second-line test (multimodal screening, or MMS) (n=50,078)
- annual TVS in a 2:1:1 ratio (n=48,230). (A scan was considered abnormal when one or both ovaries had complex morphology or a simple cyst exceeded 60 cm3 in size.)
The mean age at screening was 60.6 years, and 96.5% of participants were white.
Women who underwent MMS were triaged according to their estimated risk of developing ovarian cancer, based on the CA 125 level and age-specific risk estimates. Women in the group with the lowest risk of ovarian cancer continued annual assessment of CA 125, whereas those at highest risk underwent repeat measurement of CA 125, followed by TVS if the repeat assay suggested elevated risk. If TVS findings were also abnormal, the patient underwent clinical evaluation.
In the MMS and TVS groups, 8.7% and 12.0% of subjects, respectively, underwent clinical assessment, and surgery was performed in 0.2% and 1.8%, respectively. A primary ovarian or tubal cancer was diagnosed in 42 and 45 women in the MMS and TVS groups, respectively. In addition, 8 (MMS) and 20 (TVS) borderline malignancies were identified.
Overall, 48.3% of invasive cancers were diagnosed during stage I or II, with no difference in the distribution of stages between MMS and TVS groups. The positive predictive value (PPV) of MMS and TVS for primary invasive epithelial and tubal cancers was 35.1% and 2.8%, respectively. The ratio of surgery to case of invasive ovarian cancer was 2.9:1 (MMS) and 35:1 (TVS).
In the US study, women underwent both CA 125 assessment and TVS
The US study involved four annual rounds of screening in women 55 to 74 years old, who underwent both CA 125 measurement and TVS imaging in each round. A CA 125 level of 35 U/mL or above, or ovarian volume greater than 10 cm3 (or detection of an ovarian cyst with complex morphology) was considered abnormal.
Of 34,621 women randomized to screening, 30,630 underwent at least one screen during the four rounds. Almost two thirds of participants were 55 to 64 years old, and almost 89% were non-Hispanic white.
The percentage of women who had at least one positive screen decreased over the four rounds of screening, from 5.8% in year 1 to 4.9%, 4.6%, and 4.5% in years 2 through 4, respectively. In each round of screening, TVS was more likely to be positive than was CA 125 measurement (e.g., 4.6% vs 1.4% in the first round).
Of the 28,746 women who underwent the initial (prevalence) screen, 1,675 (5.8%) had positive findings, 566 (1.97% of those who were screened) underwent surgery, and 27 neoplasms were detected (0.06% of those who were screened), including 18 ovarian or primary peritoneal invasive cancers. Nine borderline tumors were also identified.
The PPV of a positive screen in the first round was 1.1%. Of the 18 invasive cancers identified in this round, 16.7% were diagnosed during stage I or II, and 83.4% were identified during stage III or IV. The ratio of surgery to cases of invasive ovarian cancer was 31.4:1.
Why did the trials have different findings?
The findings of the UK study suggest that an MMS strategy of CA 125 assessment, followed selectively by TVS, can detect early-stage ovarian cancer with an acceptable PPV and ratio of surgery to case of invasive cancer.
In contrast, the US findings are discouraging because of the low PPV, the large percentage of malignancies detected at an advanced stage, and the high ratio of surgery to cases of invasive cancer.
Although the studies had different primary outcomes—ovarian and tubal cancers in the UK and ovarian and primary peritoneal cancers in the US—a majority of invasive malignancies detected in both studies were ovarian.
In the US study, the poor performance of screening may be due, in part, to universal rather than selective use of TVS; that modality generates substantially more false positives than does CA 125. The US study also defined an ovarian abnormality more broadly (volume greater than 10 cm3) than the British study did (volume greater than 60 cm3), which may have lowered the PPV in the US trial.
As investigators in the UK continue to follow participants and report their findings, we will learn more about the value of MMS, including its impact on cancer mortality, possibly as soon as 2014.
Until we have further data on the value of ovarian cancer screening in postmenopausal women, we should continue the current practice of screening only symptomatic or very-high-risk women for ovarian cancer.—ANDREW M. KAUNITZ, MD
Ovarian cancer is relatively rare, but late diagnosis leads to a higher death rate than the death rate observed with other gynecologic malignancies. Detection at an early stage (I or II) is associated with higher survival than detection at a later stage (III or IV).
Earlier studies have suggested that ovarian cancer screening with the serum biomarker CA 125 and transvaginal ultrasonography (TVS) may help the clinician diagnose ovarian cancer at an earlier stage.
British study finds screening to be effective
In the United Kingdom Collaborative Trial of Ovarian Cancer Screening, postmenopausal women 50 to 74 years old were randomized to one of the following:
- no screening (n=101,359)
- annual assessment of CA 125 using a proprietary risk-of-cancer algorithm, with TVS as a second-line test (multimodal screening, or MMS) (n=50,078)
- annual TVS in a 2:1:1 ratio (n=48,230). (A scan was considered abnormal when one or both ovaries had complex morphology or a simple cyst exceeded 60 cm3 in size.)
The mean age at screening was 60.6 years, and 96.5% of participants were white.
Women who underwent MMS were triaged according to their estimated risk of developing ovarian cancer, based on the CA 125 level and age-specific risk estimates. Women in the group with the lowest risk of ovarian cancer continued annual assessment of CA 125, whereas those at highest risk underwent repeat measurement of CA 125, followed by TVS if the repeat assay suggested elevated risk. If TVS findings were also abnormal, the patient underwent clinical evaluation.
In the MMS and TVS groups, 8.7% and 12.0% of subjects, respectively, underwent clinical assessment, and surgery was performed in 0.2% and 1.8%, respectively. A primary ovarian or tubal cancer was diagnosed in 42 and 45 women in the MMS and TVS groups, respectively. In addition, 8 (MMS) and 20 (TVS) borderline malignancies were identified.
Overall, 48.3% of invasive cancers were diagnosed during stage I or II, with no difference in the distribution of stages between MMS and TVS groups. The positive predictive value (PPV) of MMS and TVS for primary invasive epithelial and tubal cancers was 35.1% and 2.8%, respectively. The ratio of surgery to case of invasive ovarian cancer was 2.9:1 (MMS) and 35:1 (TVS).
In the US study, women underwent both CA 125 assessment and TVS
The US study involved four annual rounds of screening in women 55 to 74 years old, who underwent both CA 125 measurement and TVS imaging in each round. A CA 125 level of 35 U/mL or above, or ovarian volume greater than 10 cm3 (or detection of an ovarian cyst with complex morphology) was considered abnormal.
Of 34,621 women randomized to screening, 30,630 underwent at least one screen during the four rounds. Almost two thirds of participants were 55 to 64 years old, and almost 89% were non-Hispanic white.
The percentage of women who had at least one positive screen decreased over the four rounds of screening, from 5.8% in year 1 to 4.9%, 4.6%, and 4.5% in years 2 through 4, respectively. In each round of screening, TVS was more likely to be positive than was CA 125 measurement (e.g., 4.6% vs 1.4% in the first round).
Of the 28,746 women who underwent the initial (prevalence) screen, 1,675 (5.8%) had positive findings, 566 (1.97% of those who were screened) underwent surgery, and 27 neoplasms were detected (0.06% of those who were screened), including 18 ovarian or primary peritoneal invasive cancers. Nine borderline tumors were also identified.
The PPV of a positive screen in the first round was 1.1%. Of the 18 invasive cancers identified in this round, 16.7% were diagnosed during stage I or II, and 83.4% were identified during stage III or IV. The ratio of surgery to cases of invasive ovarian cancer was 31.4:1.
Why did the trials have different findings?
The findings of the UK study suggest that an MMS strategy of CA 125 assessment, followed selectively by TVS, can detect early-stage ovarian cancer with an acceptable PPV and ratio of surgery to case of invasive cancer.
In contrast, the US findings are discouraging because of the low PPV, the large percentage of malignancies detected at an advanced stage, and the high ratio of surgery to cases of invasive cancer.
Although the studies had different primary outcomes—ovarian and tubal cancers in the UK and ovarian and primary peritoneal cancers in the US—a majority of invasive malignancies detected in both studies were ovarian.
In the US study, the poor performance of screening may be due, in part, to universal rather than selective use of TVS; that modality generates substantially more false positives than does CA 125. The US study also defined an ovarian abnormality more broadly (volume greater than 10 cm3) than the British study did (volume greater than 60 cm3), which may have lowered the PPV in the US trial.
As investigators in the UK continue to follow participants and report their findings, we will learn more about the value of MMS, including its impact on cancer mortality, possibly as soon as 2014.
Until we have further data on the value of ovarian cancer screening in postmenopausal women, we should continue the current practice of screening only symptomatic or very-high-risk women for ovarian cancer.—ANDREW M. KAUNITZ, MD
UPDATE: MENOPAUSE
Baby Boomers have transformed attitudes toward many aspects of aging. Menopause is no exception. Once a taboo topic, menopause is now openly discussed among women who seek information about vasomotor symptoms, hormones and their alternatives, and ways to maintain health as they move past midlife. ObGyns are treating more and more of these women, and fielding their many questions.
In this Update, I examine recent data on three important aspects of menopause:
- how to reduce the risk of cardiovascular disease among women who enter menopause surgically, through oophorectomy
- what to offer women who ask for nonhormonal relief from vasomotor symptoms
- a new drug on the horizon to combat osteoporosis.
Bilateral oophorectomy raises young women’s risk of cardiovascular death
Rivera CM, Grossardt BR, Rhodes DJ, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16:15–23.
Parker WH, Manson JE. Oophorectomy and cardiovascular mortality: is there a link? Menopause. 2009;16:1–2.
Cardiovascular mortality does not increase among women who undergo unilateral oophorectomy, but it does rise among women who undergo bilateral oophorectomy before 45 years of age. However, among women who initiate estrogen therapy at the time of bilateral oophorectomy and continue that therapy through at least 45 years of age, no excess cardiovascular mortality occurs.
Those are the findings of a unique retrospective cohort study performed by investigators from the Mayo Clinic. In the study, investigators reviewed the death certificates of more than 2,300 women who underwent unilateral or bilateral oophorectomy for benign disease before menopause in Olmstead County, Minnesota, from 1950 to 1987. They also followed a similar number of age-matched women for several decades.
These results support the findings of other studies that have observed that menopausal hormone therapy is associated with a lower incidence of cardiovascular death in “young” menopausal women, including those in their 50s or within one decade of the onset of menopause.1,2
More than 500,000 women undergo bilateral oophorectomy each year in the United States, usually in association with hysterectomy for benign disease.
Induced menopause merits special attention
Spontaneous menopause is physiologic. In contrast, induced menopause (whether associated with surgery, radiation therapy, or chemotherapy) and premature ovarian failure are pathologic conditions.3 Unless they are managed appropriately, induced menopause and premature ovarian failure raise the risk of cardiovascular disease.
Since the initial findings of the Women’s Health Initiative trial of estrogen–progestin therapy were published in 2002, many women and clinicians have become wary about the use of hormone therapy, even among young women who have no ovarian function and who lack a contraindication to hormone therapy.4 Unless hormonal management is contraindicated, it is recommended in this setting.
In addition, Parker and Manson recommend that gynecologic surgeons who routinely perform bilateral oophorectomy at the time of hysterectomy for benign disease in premenopausal women who do not have an elevated risk of ovarian cancer should consider updating their therapeutic recommendations and, whenever possible, preserving the ovaries.
Interest in nonhormonal therapies for hot flashes remains high
Bair YA, Gold EB, Zhang G, et al. Use of complementary and alternative medicine during the menopausal transition: longitudinal results from the Study of Women’s Health Across the Nation. Menopause. 2008;15:32–43.
Butt DA, Lock M, Lewis JE, Ross S, Moineddin R. Gabapentin for the treatment of menopausal hot flashes: a randomized trial. Menopause. 2008;15:310–318.
More than three quarters of women use some type of complementary and alternative medicine (CAM) during the menopausal transition. So found a survey conducted as part of the Study of Women’s Health Across the Nation (SWAN), which involved more than 2,000 premenopausal and perimenopausal women.
More than one third of all US women use one or more forms of CAM, spending more than $600 million a year.
In the SWAN survey, Japanese and white women were significantly more likely to report use of CAM than were Chinese, African-American, and Hispanic women during menopause.
Some women use both CAM and hormones
A notable finding of this report from SWAN is that concomitant use of menopausal hormone therapy and CAM is common among symptomatic women, even though herbal therapies have not been proved to be more effective than placebo in the treatment of vasomotor symptoms.5
Given the high prevalence of use of CAM, ObGyns should recognize that symptomatic patients especially bothered by vasomotor symptoms may seek relief with both CAM and a prescription medication. For this reason, it is wise to ask perimenopausal and postmenopausal women to list all the remedies they use—both prescription and over the counter.
Gabapentin eases symptoms in some women
Along with the antidepressants paroxetine and venlafaxine, gabapentin is the most widely studied prescription agent used in the nonhormonal treatment of menopausal vasomotor symptoms.
Canadian investigators performed a double-blind, placebo-controlled trial involving 197 symptomatic, spontaneously menopausal women (age range: 45 to 65 years). Participants were randomized to gabapentin (300 mg) or placebo, given three times daily for 4 weeks.
Ten of the 99 women who were randomized to gabapentin and six of the 98 women who were randomized to placebo dropped out of the study because of adverse effects. Hot-flash scores decreased by 51% among women taking gabapentin and by 26% among women in the placebo arm (p<.001). Women who used gabapentin were significantly more likely to report dizziness, drowsiness, and unsteadiness than those who received placebo.
Despite its proven efficacy, gabapentin does not appear to equal estrogen in its ability to alleviate vasomotor symptoms. Further, gabapentin’s side-effect profile and the need for thrice-daily administration also limit its appeal. Nevertheless, symptomatic women who are interested in nonhormonal treatment may wish to consider this option.
Anti-osteoporosis arsenal may gain a new weapon
Cummings S, McClung M, Christiansen C, et al. A phase III study of the effects of denosumab on vertebral, nonvertebral, and hip fracture in women with osteoporosis: results from the FREEDOM Trial. Paper presented at: Annual Meeting of the American Society for Bone and Mineral Research; September 16, 2008; Montreal, Canada.
Miller PD, Bolognese MA, Lewiecki EM, et al; AMG Bone Loss Study Group. Effect of denosumab on bone density and turnover in postmenopausal women with low bone mass after long-term continued, discontinued, and restarting of therapy: a randomized, blinded phase 2 clinical trial. Bone. 2008;43:222–229.
In a 3-year study in which more than 7,800 women 60 to 90 years old were randomized to denosumab (60 mg) or placebo, denosumab reduced the risk of vertebral fracture by 68% and the risk of hip fracture by 40%.
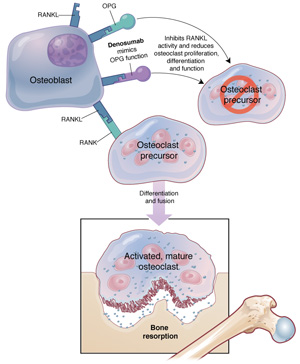
FIGURE A new agent to inhibit bone resorption
RankL is a protein that controls osteoclast maturation, activity, and survival. Denosumab mimics the endogenous decoy receptor OPG to inhibit RankL and reduce osteoclast proliferation, differentiation, and function. Denosumab is a monoclonal antibody to RankL, a mediator of osteoclast function ( FIGURE ). In contrast to alendronate, which enhances trabecular bone density (e.g., that found in the spine), denosumab increases cortical (as in long bones) as well as trabecular bone density.6 If the agent obtains approval by the US Food and Drug Administration, it will be an important new approach to the treatment of osteoporosis in menopausal women.7
The drug is administered every 6 months by subcutaneous injection.
Like estrogen, denosumab has rapidly reversible effects
The trial by Miller and associates found that the effects of denosumab are rapidly reversible after discontinuation of therapy, in contrast to alendronate. As Goldstein noted in “Update on Osteoporosis” in the November 2008 issue of OBG Management, clinicians are still exploring the relationships between alendronate and osteonecrosis of the jaw and proximal femoral-shaft fracture.7 Only further research and surveillance will clarify the clinical pros and cons of denosumab’s swift reversibility.
1. Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465-1477.
2. Manson JE, Allison MA, Rossouw JE, et al. WHI and WHICACS Investigators. Estrogen therapy and coronary-artery calcification. N Engl J Med. 2007;356:2591-2602.
3. Nelson LM. Clinical practice. Primary ovarian insufficiency. N Engl J Med. 2009;360:606-614.
4. Rossouw JE, Anderson GL, Prentice RL, et al. Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
5. Kaunitz AM. Effective herbal treatment of vasomotor symptoms—are we any closer? Menopause. 2009; Jan 21 [Epub ahead of print].
6. McClung MR, Lewiecki EM, Cohen SB, et al. Denosumab in postmenopausal women with low bone mineral density. N Engl J Med. 2006;354:821-832.
7. Goldstein SR. Update on osteoporosis. OBG Management. 2008;20(11):41-46.
Baby Boomers have transformed attitudes toward many aspects of aging. Menopause is no exception. Once a taboo topic, menopause is now openly discussed among women who seek information about vasomotor symptoms, hormones and their alternatives, and ways to maintain health as they move past midlife. ObGyns are treating more and more of these women, and fielding their many questions.
In this Update, I examine recent data on three important aspects of menopause:
- how to reduce the risk of cardiovascular disease among women who enter menopause surgically, through oophorectomy
- what to offer women who ask for nonhormonal relief from vasomotor symptoms
- a new drug on the horizon to combat osteoporosis.
Bilateral oophorectomy raises young women’s risk of cardiovascular death
Rivera CM, Grossardt BR, Rhodes DJ, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16:15–23.
Parker WH, Manson JE. Oophorectomy and cardiovascular mortality: is there a link? Menopause. 2009;16:1–2.
Cardiovascular mortality does not increase among women who undergo unilateral oophorectomy, but it does rise among women who undergo bilateral oophorectomy before 45 years of age. However, among women who initiate estrogen therapy at the time of bilateral oophorectomy and continue that therapy through at least 45 years of age, no excess cardiovascular mortality occurs.
Those are the findings of a unique retrospective cohort study performed by investigators from the Mayo Clinic. In the study, investigators reviewed the death certificates of more than 2,300 women who underwent unilateral or bilateral oophorectomy for benign disease before menopause in Olmstead County, Minnesota, from 1950 to 1987. They also followed a similar number of age-matched women for several decades.
These results support the findings of other studies that have observed that menopausal hormone therapy is associated with a lower incidence of cardiovascular death in “young” menopausal women, including those in their 50s or within one decade of the onset of menopause.1,2
More than 500,000 women undergo bilateral oophorectomy each year in the United States, usually in association with hysterectomy for benign disease.
Induced menopause merits special attention
Spontaneous menopause is physiologic. In contrast, induced menopause (whether associated with surgery, radiation therapy, or chemotherapy) and premature ovarian failure are pathologic conditions.3 Unless they are managed appropriately, induced menopause and premature ovarian failure raise the risk of cardiovascular disease.
Since the initial findings of the Women’s Health Initiative trial of estrogen–progestin therapy were published in 2002, many women and clinicians have become wary about the use of hormone therapy, even among young women who have no ovarian function and who lack a contraindication to hormone therapy.4 Unless hormonal management is contraindicated, it is recommended in this setting.
In addition, Parker and Manson recommend that gynecologic surgeons who routinely perform bilateral oophorectomy at the time of hysterectomy for benign disease in premenopausal women who do not have an elevated risk of ovarian cancer should consider updating their therapeutic recommendations and, whenever possible, preserving the ovaries.
Interest in nonhormonal therapies for hot flashes remains high
Bair YA, Gold EB, Zhang G, et al. Use of complementary and alternative medicine during the menopausal transition: longitudinal results from the Study of Women’s Health Across the Nation. Menopause. 2008;15:32–43.
Butt DA, Lock M, Lewis JE, Ross S, Moineddin R. Gabapentin for the treatment of menopausal hot flashes: a randomized trial. Menopause. 2008;15:310–318.
More than three quarters of women use some type of complementary and alternative medicine (CAM) during the menopausal transition. So found a survey conducted as part of the Study of Women’s Health Across the Nation (SWAN), which involved more than 2,000 premenopausal and perimenopausal women.
More than one third of all US women use one or more forms of CAM, spending more than $600 million a year.
In the SWAN survey, Japanese and white women were significantly more likely to report use of CAM than were Chinese, African-American, and Hispanic women during menopause.
Some women use both CAM and hormones
A notable finding of this report from SWAN is that concomitant use of menopausal hormone therapy and CAM is common among symptomatic women, even though herbal therapies have not been proved to be more effective than placebo in the treatment of vasomotor symptoms.5
Given the high prevalence of use of CAM, ObGyns should recognize that symptomatic patients especially bothered by vasomotor symptoms may seek relief with both CAM and a prescription medication. For this reason, it is wise to ask perimenopausal and postmenopausal women to list all the remedies they use—both prescription and over the counter.
Gabapentin eases symptoms in some women
Along with the antidepressants paroxetine and venlafaxine, gabapentin is the most widely studied prescription agent used in the nonhormonal treatment of menopausal vasomotor symptoms.
Canadian investigators performed a double-blind, placebo-controlled trial involving 197 symptomatic, spontaneously menopausal women (age range: 45 to 65 years). Participants were randomized to gabapentin (300 mg) or placebo, given three times daily for 4 weeks.
Ten of the 99 women who were randomized to gabapentin and six of the 98 women who were randomized to placebo dropped out of the study because of adverse effects. Hot-flash scores decreased by 51% among women taking gabapentin and by 26% among women in the placebo arm (p<.001). Women who used gabapentin were significantly more likely to report dizziness, drowsiness, and unsteadiness than those who received placebo.
Despite its proven efficacy, gabapentin does not appear to equal estrogen in its ability to alleviate vasomotor symptoms. Further, gabapentin’s side-effect profile and the need for thrice-daily administration also limit its appeal. Nevertheless, symptomatic women who are interested in nonhormonal treatment may wish to consider this option.
Anti-osteoporosis arsenal may gain a new weapon
Cummings S, McClung M, Christiansen C, et al. A phase III study of the effects of denosumab on vertebral, nonvertebral, and hip fracture in women with osteoporosis: results from the FREEDOM Trial. Paper presented at: Annual Meeting of the American Society for Bone and Mineral Research; September 16, 2008; Montreal, Canada.
Miller PD, Bolognese MA, Lewiecki EM, et al; AMG Bone Loss Study Group. Effect of denosumab on bone density and turnover in postmenopausal women with low bone mass after long-term continued, discontinued, and restarting of therapy: a randomized, blinded phase 2 clinical trial. Bone. 2008;43:222–229.
In a 3-year study in which more than 7,800 women 60 to 90 years old were randomized to denosumab (60 mg) or placebo, denosumab reduced the risk of vertebral fracture by 68% and the risk of hip fracture by 40%.

FIGURE A new agent to inhibit bone resorption
RankL is a protein that controls osteoclast maturation, activity, and survival. Denosumab mimics the endogenous decoy receptor OPG to inhibit RankL and reduce osteoclast proliferation, differentiation, and function. Denosumab is a monoclonal antibody to RankL, a mediator of osteoclast function ( FIGURE ). In contrast to alendronate, which enhances trabecular bone density (e.g., that found in the spine), denosumab increases cortical (as in long bones) as well as trabecular bone density.6 If the agent obtains approval by the US Food and Drug Administration, it will be an important new approach to the treatment of osteoporosis in menopausal women.7
The drug is administered every 6 months by subcutaneous injection.
Like estrogen, denosumab has rapidly reversible effects
The trial by Miller and associates found that the effects of denosumab are rapidly reversible after discontinuation of therapy, in contrast to alendronate. As Goldstein noted in “Update on Osteoporosis” in the November 2008 issue of OBG Management, clinicians are still exploring the relationships between alendronate and osteonecrosis of the jaw and proximal femoral-shaft fracture.7 Only further research and surveillance will clarify the clinical pros and cons of denosumab’s swift reversibility.
Baby Boomers have transformed attitudes toward many aspects of aging. Menopause is no exception. Once a taboo topic, menopause is now openly discussed among women who seek information about vasomotor symptoms, hormones and their alternatives, and ways to maintain health as they move past midlife. ObGyns are treating more and more of these women, and fielding their many questions.
In this Update, I examine recent data on three important aspects of menopause:
- how to reduce the risk of cardiovascular disease among women who enter menopause surgically, through oophorectomy
- what to offer women who ask for nonhormonal relief from vasomotor symptoms
- a new drug on the horizon to combat osteoporosis.
Bilateral oophorectomy raises young women’s risk of cardiovascular death
Rivera CM, Grossardt BR, Rhodes DJ, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16:15–23.
Parker WH, Manson JE. Oophorectomy and cardiovascular mortality: is there a link? Menopause. 2009;16:1–2.
Cardiovascular mortality does not increase among women who undergo unilateral oophorectomy, but it does rise among women who undergo bilateral oophorectomy before 45 years of age. However, among women who initiate estrogen therapy at the time of bilateral oophorectomy and continue that therapy through at least 45 years of age, no excess cardiovascular mortality occurs.
Those are the findings of a unique retrospective cohort study performed by investigators from the Mayo Clinic. In the study, investigators reviewed the death certificates of more than 2,300 women who underwent unilateral or bilateral oophorectomy for benign disease before menopause in Olmstead County, Minnesota, from 1950 to 1987. They also followed a similar number of age-matched women for several decades.
These results support the findings of other studies that have observed that menopausal hormone therapy is associated with a lower incidence of cardiovascular death in “young” menopausal women, including those in their 50s or within one decade of the onset of menopause.1,2
More than 500,000 women undergo bilateral oophorectomy each year in the United States, usually in association with hysterectomy for benign disease.
Induced menopause merits special attention
Spontaneous menopause is physiologic. In contrast, induced menopause (whether associated with surgery, radiation therapy, or chemotherapy) and premature ovarian failure are pathologic conditions.3 Unless they are managed appropriately, induced menopause and premature ovarian failure raise the risk of cardiovascular disease.
Since the initial findings of the Women’s Health Initiative trial of estrogen–progestin therapy were published in 2002, many women and clinicians have become wary about the use of hormone therapy, even among young women who have no ovarian function and who lack a contraindication to hormone therapy.4 Unless hormonal management is contraindicated, it is recommended in this setting.
In addition, Parker and Manson recommend that gynecologic surgeons who routinely perform bilateral oophorectomy at the time of hysterectomy for benign disease in premenopausal women who do not have an elevated risk of ovarian cancer should consider updating their therapeutic recommendations and, whenever possible, preserving the ovaries.
Interest in nonhormonal therapies for hot flashes remains high
Bair YA, Gold EB, Zhang G, et al. Use of complementary and alternative medicine during the menopausal transition: longitudinal results from the Study of Women’s Health Across the Nation. Menopause. 2008;15:32–43.
Butt DA, Lock M, Lewis JE, Ross S, Moineddin R. Gabapentin for the treatment of menopausal hot flashes: a randomized trial. Menopause. 2008;15:310–318.
More than three quarters of women use some type of complementary and alternative medicine (CAM) during the menopausal transition. So found a survey conducted as part of the Study of Women’s Health Across the Nation (SWAN), which involved more than 2,000 premenopausal and perimenopausal women.
More than one third of all US women use one or more forms of CAM, spending more than $600 million a year.
In the SWAN survey, Japanese and white women were significantly more likely to report use of CAM than were Chinese, African-American, and Hispanic women during menopause.
Some women use both CAM and hormones
A notable finding of this report from SWAN is that concomitant use of menopausal hormone therapy and CAM is common among symptomatic women, even though herbal therapies have not been proved to be more effective than placebo in the treatment of vasomotor symptoms.5
Given the high prevalence of use of CAM, ObGyns should recognize that symptomatic patients especially bothered by vasomotor symptoms may seek relief with both CAM and a prescription medication. For this reason, it is wise to ask perimenopausal and postmenopausal women to list all the remedies they use—both prescription and over the counter.
Gabapentin eases symptoms in some women
Along with the antidepressants paroxetine and venlafaxine, gabapentin is the most widely studied prescription agent used in the nonhormonal treatment of menopausal vasomotor symptoms.
Canadian investigators performed a double-blind, placebo-controlled trial involving 197 symptomatic, spontaneously menopausal women (age range: 45 to 65 years). Participants were randomized to gabapentin (300 mg) or placebo, given three times daily for 4 weeks.
Ten of the 99 women who were randomized to gabapentin and six of the 98 women who were randomized to placebo dropped out of the study because of adverse effects. Hot-flash scores decreased by 51% among women taking gabapentin and by 26% among women in the placebo arm (p<.001). Women who used gabapentin were significantly more likely to report dizziness, drowsiness, and unsteadiness than those who received placebo.
Despite its proven efficacy, gabapentin does not appear to equal estrogen in its ability to alleviate vasomotor symptoms. Further, gabapentin’s side-effect profile and the need for thrice-daily administration also limit its appeal. Nevertheless, symptomatic women who are interested in nonhormonal treatment may wish to consider this option.
Anti-osteoporosis arsenal may gain a new weapon
Cummings S, McClung M, Christiansen C, et al. A phase III study of the effects of denosumab on vertebral, nonvertebral, and hip fracture in women with osteoporosis: results from the FREEDOM Trial. Paper presented at: Annual Meeting of the American Society for Bone and Mineral Research; September 16, 2008; Montreal, Canada.
Miller PD, Bolognese MA, Lewiecki EM, et al; AMG Bone Loss Study Group. Effect of denosumab on bone density and turnover in postmenopausal women with low bone mass after long-term continued, discontinued, and restarting of therapy: a randomized, blinded phase 2 clinical trial. Bone. 2008;43:222–229.
In a 3-year study in which more than 7,800 women 60 to 90 years old were randomized to denosumab (60 mg) or placebo, denosumab reduced the risk of vertebral fracture by 68% and the risk of hip fracture by 40%.

FIGURE A new agent to inhibit bone resorption
RankL is a protein that controls osteoclast maturation, activity, and survival. Denosumab mimics the endogenous decoy receptor OPG to inhibit RankL and reduce osteoclast proliferation, differentiation, and function. Denosumab is a monoclonal antibody to RankL, a mediator of osteoclast function ( FIGURE ). In contrast to alendronate, which enhances trabecular bone density (e.g., that found in the spine), denosumab increases cortical (as in long bones) as well as trabecular bone density.6 If the agent obtains approval by the US Food and Drug Administration, it will be an important new approach to the treatment of osteoporosis in menopausal women.7
The drug is administered every 6 months by subcutaneous injection.
Like estrogen, denosumab has rapidly reversible effects
The trial by Miller and associates found that the effects of denosumab are rapidly reversible after discontinuation of therapy, in contrast to alendronate. As Goldstein noted in “Update on Osteoporosis” in the November 2008 issue of OBG Management, clinicians are still exploring the relationships between alendronate and osteonecrosis of the jaw and proximal femoral-shaft fracture.7 Only further research and surveillance will clarify the clinical pros and cons of denosumab’s swift reversibility.
1. Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465-1477.
2. Manson JE, Allison MA, Rossouw JE, et al. WHI and WHICACS Investigators. Estrogen therapy and coronary-artery calcification. N Engl J Med. 2007;356:2591-2602.
3. Nelson LM. Clinical practice. Primary ovarian insufficiency. N Engl J Med. 2009;360:606-614.
4. Rossouw JE, Anderson GL, Prentice RL, et al. Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
5. Kaunitz AM. Effective herbal treatment of vasomotor symptoms—are we any closer? Menopause. 2009; Jan 21 [Epub ahead of print].
6. McClung MR, Lewiecki EM, Cohen SB, et al. Denosumab in postmenopausal women with low bone mineral density. N Engl J Med. 2006;354:821-832.
7. Goldstein SR. Update on osteoporosis. OBG Management. 2008;20(11):41-46.
1. Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465-1477.
2. Manson JE, Allison MA, Rossouw JE, et al. WHI and WHICACS Investigators. Estrogen therapy and coronary-artery calcification. N Engl J Med. 2007;356:2591-2602.
3. Nelson LM. Clinical practice. Primary ovarian insufficiency. N Engl J Med. 2009;360:606-614.
4. Rossouw JE, Anderson GL, Prentice RL, et al. Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
5. Kaunitz AM. Effective herbal treatment of vasomotor symptoms—are we any closer? Menopause. 2009; Jan 21 [Epub ahead of print].
6. McClung MR, Lewiecki EM, Cohen SB, et al. Denosumab in postmenopausal women with low bone mineral density. N Engl J Med. 2006;354:821-832.
7. Goldstein SR. Update on osteoporosis. OBG Management. 2008;20(11):41-46.
Higher Stroke Risk Tied to Early Menopause
SAN DIEGO — Women who reach menopause before the age of 42 years are twice as likely to suffer a stroke in later life as women who reach menopause after age 42, according to a new analysis of data from the Framingham Heart Study presented at the International Stroke Conference.
The study involved prospectively collected data from 1,430 women who were followed for an average of 22 years, said Lynda Lisabeth, Ph.D., of the University of Michigan, Ann Arbor. All participants were stroke-free at 60 years of age, experienced natural menopause, and had never taken estrogen before menopause. The use of self-reported data on the age of menopause was a limitation of the study, Dr. Lisabeth acknowledged.
In all, the women had 234 ischemic strokes at an average age of 80 years. The unadjusted rate of strokes was 23% among women who reached menopause before the age of 42 years, 16% among women who reached menopause between the ages of 42 and 54 years, and 11% among women who reached menopause at age 55 years or older.
After adjustment for age, systolic blood pressure, atrial fibrillation, diabetes, current smoking, cardiovascular disease, and estrogen use after menopause, the investigators determined that the age of menopause was an independent predictor of ischemic stroke.
Compared with women who reached menopause before age 42 years, women who reached menopause between ages 42 and 54 years were half as likely to experience a stroke, and those who reached menopause at age 55 or older were 69% less likely to experience a stroke.
In other words, women who reached menopause before age 42 years were 2.03-fold more likely to have a stroke than the other women. This difference was statistically significant.
The study showed that 4%–5% of strokes in women can be attributed to menopause before age 42, Dr. Lisabeth said.
About 1%–2% of women reach menopause at or before age 40 years, which is referred to as “premature ovarian failure.” The etiology of this condition remains unknown, but investigators are certain that it's different than natural menopause. About 3%–10% of women experience “early” menopause, defined as natural menopause before age 45 years.
Several possible mechanisms could account for the increased rate of stroke, Dr. Lisabeth said. Estrogen may play a role, since estrogen deficiency is thought to promote cardiovascular disease through functional or structural changes in arteries. Androgens and sex hormone-binding globulin are also risk factors for cardiovascular disease.
Additional studies with measures of endogenous hormones would be needed to unravel the relationship between the hormonal changes of menopause and ischemic stroke, she said.
The study was supported by the National Heart, Lung, and Blood Institute and the National Institute of Neurological Disorders and Stroke. Dr. Lisabeth said that she had no conflicts of interest.
SAN DIEGO — Women who reach menopause before the age of 42 years are twice as likely to suffer a stroke in later life as women who reach menopause after age 42, according to a new analysis of data from the Framingham Heart Study presented at the International Stroke Conference.
The study involved prospectively collected data from 1,430 women who were followed for an average of 22 years, said Lynda Lisabeth, Ph.D., of the University of Michigan, Ann Arbor. All participants were stroke-free at 60 years of age, experienced natural menopause, and had never taken estrogen before menopause. The use of self-reported data on the age of menopause was a limitation of the study, Dr. Lisabeth acknowledged.
In all, the women had 234 ischemic strokes at an average age of 80 years. The unadjusted rate of strokes was 23% among women who reached menopause before the age of 42 years, 16% among women who reached menopause between the ages of 42 and 54 years, and 11% among women who reached menopause at age 55 years or older.
After adjustment for age, systolic blood pressure, atrial fibrillation, diabetes, current smoking, cardiovascular disease, and estrogen use after menopause, the investigators determined that the age of menopause was an independent predictor of ischemic stroke.
Compared with women who reached menopause before age 42 years, women who reached menopause between ages 42 and 54 years were half as likely to experience a stroke, and those who reached menopause at age 55 or older were 69% less likely to experience a stroke.
In other words, women who reached menopause before age 42 years were 2.03-fold more likely to have a stroke than the other women. This difference was statistically significant.
The study showed that 4%–5% of strokes in women can be attributed to menopause before age 42, Dr. Lisabeth said.
About 1%–2% of women reach menopause at or before age 40 years, which is referred to as “premature ovarian failure.” The etiology of this condition remains unknown, but investigators are certain that it's different than natural menopause. About 3%–10% of women experience “early” menopause, defined as natural menopause before age 45 years.
Several possible mechanisms could account for the increased rate of stroke, Dr. Lisabeth said. Estrogen may play a role, since estrogen deficiency is thought to promote cardiovascular disease through functional or structural changes in arteries. Androgens and sex hormone-binding globulin are also risk factors for cardiovascular disease.
Additional studies with measures of endogenous hormones would be needed to unravel the relationship between the hormonal changes of menopause and ischemic stroke, she said.
The study was supported by the National Heart, Lung, and Blood Institute and the National Institute of Neurological Disorders and Stroke. Dr. Lisabeth said that she had no conflicts of interest.
SAN DIEGO — Women who reach menopause before the age of 42 years are twice as likely to suffer a stroke in later life as women who reach menopause after age 42, according to a new analysis of data from the Framingham Heart Study presented at the International Stroke Conference.
The study involved prospectively collected data from 1,430 women who were followed for an average of 22 years, said Lynda Lisabeth, Ph.D., of the University of Michigan, Ann Arbor. All participants were stroke-free at 60 years of age, experienced natural menopause, and had never taken estrogen before menopause. The use of self-reported data on the age of menopause was a limitation of the study, Dr. Lisabeth acknowledged.
In all, the women had 234 ischemic strokes at an average age of 80 years. The unadjusted rate of strokes was 23% among women who reached menopause before the age of 42 years, 16% among women who reached menopause between the ages of 42 and 54 years, and 11% among women who reached menopause at age 55 years or older.
After adjustment for age, systolic blood pressure, atrial fibrillation, diabetes, current smoking, cardiovascular disease, and estrogen use after menopause, the investigators determined that the age of menopause was an independent predictor of ischemic stroke.
Compared with women who reached menopause before age 42 years, women who reached menopause between ages 42 and 54 years were half as likely to experience a stroke, and those who reached menopause at age 55 or older were 69% less likely to experience a stroke.
In other words, women who reached menopause before age 42 years were 2.03-fold more likely to have a stroke than the other women. This difference was statistically significant.
The study showed that 4%–5% of strokes in women can be attributed to menopause before age 42, Dr. Lisabeth said.
About 1%–2% of women reach menopause at or before age 40 years, which is referred to as “premature ovarian failure.” The etiology of this condition remains unknown, but investigators are certain that it's different than natural menopause. About 3%–10% of women experience “early” menopause, defined as natural menopause before age 45 years.
Several possible mechanisms could account for the increased rate of stroke, Dr. Lisabeth said. Estrogen may play a role, since estrogen deficiency is thought to promote cardiovascular disease through functional or structural changes in arteries. Androgens and sex hormone-binding globulin are also risk factors for cardiovascular disease.
Additional studies with measures of endogenous hormones would be needed to unravel the relationship between the hormonal changes of menopause and ischemic stroke, she said.
The study was supported by the National Heart, Lung, and Blood Institute and the National Institute of Neurological Disorders and Stroke. Dr. Lisabeth said that she had no conflicts of interest.