User login
Families as Care Partners: Implementing the Better Together Initiative Across a Large Health System
From the Institute for Patient- and Family-Centered Care, Bethesda, MD (Ms. Dokken and Ms. Johnson), and Northwell Health, New Hyde Park, NY (Dr. Barden, Ms. Tuomey, and Ms. Giammarinaro).
Abstract
Objective: To describe the growth of Better Together: Partnering with Families, a campaign launched in 2014 to eliminate restrictive hospital visiting policies and to put in place policies that recognize families as partners in care, and to discuss the processes involved in implementing the initiative in a large, integrated health system.
Methods: Descriptive report.
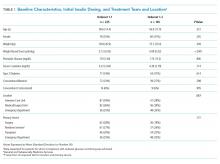
Results: In June 2014, the Institute for Patient- and Family-Centered Care (IPFCC) launched the Better Together campaign to emphasize the importance of family presence and participation to the quality, experience, safety, and outcomes of care. Since then, this initiative has expanded in both the United States and Canada. With support from 2 funders in the United States, special attention was focused on acute care hospitals across New York State. Nearly 50 hospitals participated in 2 separate but related projects. Fifteen of the hospitals are part of Northwell Health, New York State’s largest health system. Over a 10-month period, these hospitals made significant progress in changing policy, practice, and communication to support family presence.
Conclusion: The Better Together initiative was implemented across a health system with strong support from leadership and the involvement of patient and family advisors. An intervention offering structured training, coaching, and resources, like IPFCC’s Better Together initiative, can facilitate the change process.
Keywords: family presence; visiting policies; patient-centered care; family-centered care; patient experience.
The presence of families at the bedside of patients is often restricted by hospital visiting hours. Hospitals that maintain these restrictive policies cite concerns about negative impacts on security, infection control, privacy, and staff workload. But there are no data to support these concerns, and the experience of hospitals that have successfully changed policy and practice to welcome families demonstrates the potential positive impacts of less restrictive policies on patient care and outcomes.1 For example, hospitalization can lead to reduced cognitive function in elderly patients. Family members would recognize the changes and could provide valuable information to hospital staff, potentially improving outcomes.2
In June 2014, the Institute for Patient- and Family-Centered Care (IPFCC) launched the campaign Better Together: Partnering with Families.3 The campaign is is grounded in patient- and family- centered care, an approach to care that supports partnerships among health care providers, patients, and families, and, among other core principles, advocates that patients define their “families” and how they will participate in care and decision-making.
Emphasizing the importance of family presence and participation to quality and safety, the Better Together campaign seeks to eliminate restrictive visiting policies and calls upon hospitals to include families as members of the care team and to welcome them 24 hours a day, 7 days a week, according to patient preference. As part of the campaign, IPFCC developed an extensive toolkit of resources that is available to hospitals and other organizations at no cost. The resources include sample policies; profiles of hospitals that have implemented family presence policies; educational materials for staff, patients, and families; and a template for hospital websites. This article, a follow-up to an article published in the January 2015 issue of JCOM,1 discusses the growth of the Better Together initiative as well as the processes involved in implementing the initiative across a large health system.
Growth of the Initiative
Since its launch in 2014, the Better Together initiative has continued to expand in the United States and Canada. In Canada, under the leadership of the Canadian Foundation for Healthcare Improvement (CFHI), more than 50 organizations have made a commitment to the Better Together program and family presence.4 Utilizing and adapting IPFCC’s Toolkit, CFHI developed a change package of free resources for Canadian organizations.5 Some of the materials, including the Pocket Guide for Families (Manuel des Familles), were translated into French.6
With support from 2 funders in the United States, the United Hospital Fund and the New York State Health (NYSHealth) Foundation, through a subcontract with the New York Public Interest Research Group (NYPIRG), IPFCC has been able to focus on hospitals in New York City, including public hospitals, and, more broadly, acute care hospitals across New York State. Nearly 50 hospitals participated in these 2 separate but related projects.
Education and Support for New York City Hospitals
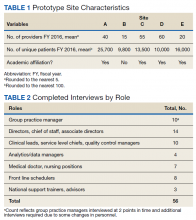
Supported by the United Hospital Fund, an 18-month project that focused specifically on New York City hospitals was completed in June 2017. The project began with a 1-day intensive training event with representatives of 21 hospitals. Eighteen of those hospitals were eligible to participate in follow-up consultation provided by IPFCC, and 14 participated in some kind of follow-up. NYC Health + Hospitals (H+H), the system of public hospitals in NYC, participated most fully in these activities.
The outcomes of the Better Together initiative in New York City are summarized in the report Sick, Scared, & Separated From Loved Ones,2 which is based on a pre/post review of hospital visitation/family presence policies and website communications. According to the report, hospitals that participated in the IPFCC training and consultation program performed better, as a group, with respect to improved policy and website scores on post review than those that did not. Of the 10 hospitals whose scores improved during the review period, 8 had participated in the IPFCC training and 1 hospital was part of a hospital network that did so. (Six of these hospitals are part of the H+H public hospital system.) Those 9 hospitals saw an average increase in scores of 4.9 points (out of a possible 11).
A Learning Community for Hospitals in New York State
With support from the NYSHealth Foundation, IPFCC again collaborated with NYPIRG and New Yorkers for Patient & Family Empowerment on a 2-year initiative, completed in November 2019, that involved 26 hospitals: 15 from Northwell Health, New York State’s largest health system, and 11 hospitals from health systems throughout the state (Greater Hudson Valley Health System, now Garnet Health; Mohawk Valley Health System; Rochester Regional Health; and University of Vermont Health Network). An update of the report Sick, Scared, & Separated From Loved Onescompared pre/post reviews of policies and website communications regarding hospital visitation/family presence.7 Its findings confirm that hospitals that participated in the Better Together Learning Community improved both their policy and website scores to a greater degree than hospitals that did not participate and that a planned intervention can help facilitate change.
During the survey period, 28 out of 40 hospitals’ website navigability scores improved. Of those, hospitals that did not participate in the Better Together Learning Community saw an average increase in scores of 1.2 points, out of a possible 11, while the participating hospitals saw an average increase of 2.7 points, with the top 5 largest increases in scores belonging to hospitals that participated in the Better Together Learning Community.7
The Northwell Health Experience
Northwell Health is a large integrated health care organization comprising more than 69,000 employees, 23 hospitals, and more than 750 medical practices, located geographically across New York State. Embracing patient- and family-centered care, Northwell is dedicated to improving the quality, experience, and safety of care for patients and their families. Welcoming and including patients, families, and care partners as members of the health care team has always been a core element of Northwell’s organizational goal of providing world-class patient care and experience.
Four years ago, the organization reorganized and formalized a system-wide Patient & Family Partnership Council (PFPC).8 Representatives on the PFPC include a Northwell patient experience leader and patient/family co-chair from local councils that have been established in nearly all 23 hospitals as well as service lines. Modeling partnership, the PFPC is grounded in listening to the “voice” of patients and families and promoting collaboration, with the goal of driving change across varied aspects and experiences of health care delivery.
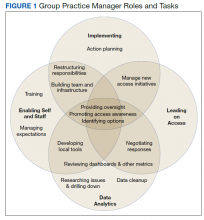
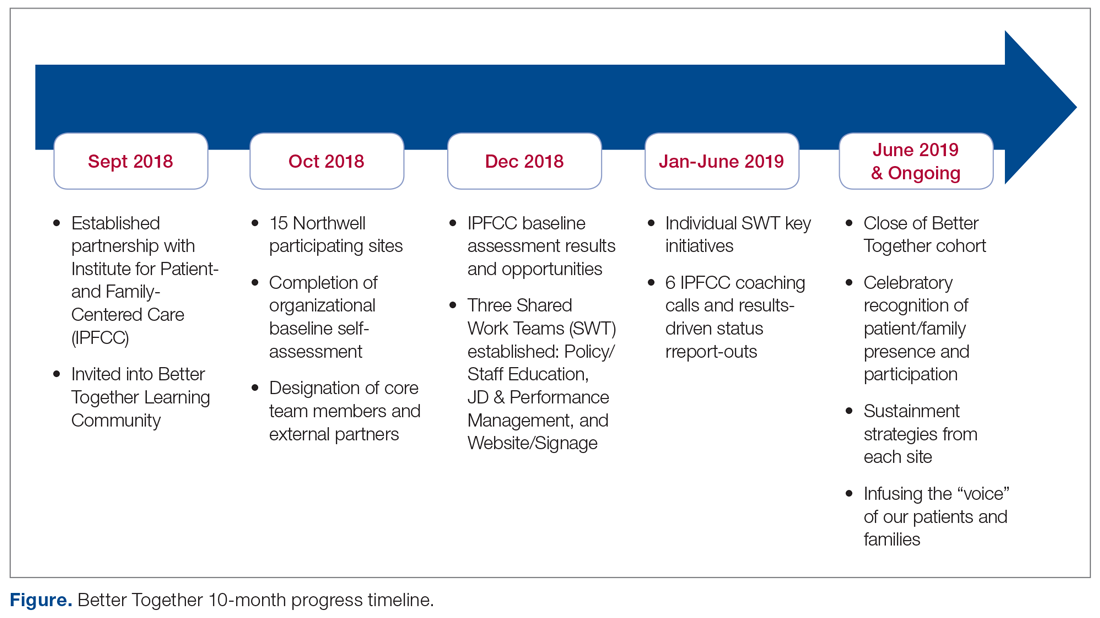
Through the Office of Patient and Customer Experience (OPCE), a partnership with IPFCC and the Better Together Learning Community for Hospitals in New York State was initiated as a fundamental next step in Northwell’s journey to enhance system-wide family presence and participation. Results from Better Together’s Organizational Self-Assessment Tool and process identified opportunities to influence 3 distinct areas: policy/staff education, position descriptions/performance management, and website/signage. Over a 10-month period (September 2018 through June 2019), 15 Northwell hospitals implemened significant patient- and family-centered improvements through multifaceted shared work teams (SWT) that partnered around the common goal of supporting the patient and family experience (Figure). Northwell’s SWT structure allowed teams to meet individually on specific tasks, led by a dedicated staff member of the OPCE to ensure progress, support, and accountability. Six monthly coaching calls or report-out meetings were attended by participating teams, where feedback and recommendations shared by IPFCC were discussed in order to maintain momentum and results.
Policy/Staff Education
The policy/staff education SWT focused on appraising and updating existing policies to ensure alignment with key patient- and family-centered concepts and Better Together principles (Table 1). By establishing representation on the System Policy and Procedure Committee, OPCE enabled patients and families to have a voice at the decision-making table. OPCE leaders presented the ideology and scope of the transformation to this committee. After reviewing all system-wide policies, 4 were identified as key opportunities for revision. One overarching policy titled “Visitation Guidelines” was reviewed and updated to reflect Northwell’s mission of patient- and family-centered care, retiring the reference to “families” as “visitors” in definitions, incorporating language of inclusion and partnership, and citing other related policies. The policy was vetted through a multilayer process of review and stakeholder feedback and was ultimately approved at a system

Three additional related policies were also updated to reflect core principles of inclusion and partnership. These included system policies focused on discharge planning; identification of health care proxy, agent, support person and caregiver; and standards of behavior not conducive in a health care setting. As a result of this work, OPCE was invited to remain an active member of the System Policy and Procedure Committee, adding meaningful new perspectives to the clinical and administrative policy management process. Once policies were updated and approved, the SWT focused on educating leaders and teams. Using a diversified strategy, education was provided through various modes, including weekly system-wide internal communication channels, patient experience huddle messages, yearly mandatory topics training, and the incorporation of essential concepts in existing educational courses (classroom and e-learning modalities).
Position Descriptions/Performance Management
The position descriptions/performance management SWT focused its efforts on incorporating patient- and family-centered concepts and language into position descriptions and the performance appraisal process (Table 2). Due to the complex nature of this work, the process required collaboration from key subject matter experts in human resources, talent management, corporate compensation, and labor management. In 2019, Northwell began an initiative focused on streamlining and standardizing job titles, roles, and developmental pathways across the system. The overarching goal was to create system-wide consistency and standardization. The SWT was successful in advising the leaders overseeing this job architecture initiative on the importance of including language of patient- and family-centered care, like partnership and collaboration, and of highlighting the critical role of family members as part of the care team in subsequent documents.

Northwell has 6 behavioral expectations, standards to which all team members are held accountable: Patient/Customer Focus, Teamwork, Execution, Organizational Awareness, Enable Change, and Develop Self. As a result of the SWT’s work, Patient/Customer Focus was revised to include “families” as essential care partners, demonstrating Northwell’s ongoing commitment to honoring the role of families as members of the care team. It also ensures that all employees are aligned around this priority, as these expectations are utilized to support areas such as recognition and performance. Collaborating with talent management and organizational development, the SWT reviewed yearly performance management and new-hire evaluations. In doing so, they identified an opportunity to refresh the anchored qualitative rating scales to include behavioral demonstrations of patient- and family-centered care, collaboration, respect, and partnership with family members.
Website/Signage
Websites make an important first impression on patients and families looking for information to best prepare for a hospital experience. Therefore, the website/signage SWT worked to redesign hospital websites, enhance digital signage, and perform a baseline assessment of physical signage across facilities. Initial feedback on Northwell’s websites identified opportunities to include more patient- and family-centered, care-partner-infused language; improve navigation; and streamline click levels for easier access. Content for the websites was carefully crafted in collaboration with Northwell’s internal web team, utilizing IPFCC’s best practice standards as a framework and guide.
Next, a multidisciplinary website shared-governance team was established by the OPCE to ensure that key stakeholders were represented and had the opportunity to review and make recommendations for appropriate language and messaging about family presence and participation. This 13-person team was comprised of patient/family partners, patient-experience culture leaders, quality, compliance, human resources, policy, a chief nursing officer, a medical director, and representation from the Institute for Nursing. After careful review and consideration from Northwell’s family partners and teams, all participating hospital websites were enhanced as of June 2019 to include prominent 1-click access from homepages to information for “patients, families and visitors,” as well as “your care partners” information on the important role of families and care partners.
Along with refreshing websites, another step in Northwell’s work to strengthen messaging about family presence and participation was to partner and collaborate with the system’s digital web team as well as local facility councils to understand the capacity to adjust digital signage across facilities. Opportunities were found to make simple yet effective enhancements to the language and imagery of digital signage upon entry, creating a warmer and more welcoming first impression for patients and families. With patient and family partner feedback, the team designed digital signage with inclusive messaging and images that would circulate appropriately based on the facility. Signage specifically welcomes families and refers to them as members of patients’ care teams.
Northwell’s website/signage SWT also directed a 2-phase physical signage assessment to determine ongoing opportunities to alter signs in areas that particularly impact patients and families, such as emergency departments, main lobbies, cafeterias, surgical waiting areas, and intensive care units. Each hospital’s local PFPC did a “walk-about”9 to make enhancements to physical signage, such as removing paper and overcrowded signs, adjusting negative language, ensuring alignment with brand guidelines, and including language that welcomed families. As a result of the team’s efforts around signage, collaboration began with the health system’s signage committee to help standardize signage terminology to reflect family inclusiveness, and to implement the recommendation for a standardized signage shared-governance team to ensure accountability and a patient- and family-centered structure.
Sustainment
Since implementing Better Together, Northwell has been able to infuse a more patient- and family-centered emphasis into its overall patient experience message of “Every role, every person, every moment matters.” As a strategic tool aimed at encouraging leaders, clinicians, and staff to pause and reflect about the “heart” of their work, patient and family stories are now included at the beginning of meetings, forums, and team huddles. Elements of the initiative have been integrated in current Patient and Family Partnership sustainment plans at participating hospitals. Some highlights include continued integration of patient/family partners on committees and councils that impact areas such as way finding, signage, recruitment, new-hire orientation, and community outreach; focus on enhancing partner retention and development programs; and inclusion of patient- and family-centered care and Better Together principles in ongoing leadership meetings.
Factors Contributing to Success
Health care is a complex, regulated, and often bureaucratic world that can be very difficult for patients and families to navigate. The system’s partnership with the Better Together Learning Community for Hospitals in New York State enhanced its efforts to improve family presence and participation and created powerful synergy. The success of this partnership was based on a number of important factors:
A solid foundation of support, structure, and accountability. The OPCE initiated the IPFCC Better Together partnership and established a synergistic collaboration inclusive of leadership, frontline teams, multiple departments, and patient and family partners. As a major strategic component of Northwell’s mission to deliver high-quality, patient- and family-centered care, OPCE was instrumental in connecting key areas and stakeholders and mobilizing the recommendations coming from patients and families.
A visible commitment of leadership at all levels. Partnering with leadership across Northwell’s system required a delineated vision, clear purpose and ownership, and comprehensive implementation and sustainment strategies. The existing format of Northwell’s PFPC provided the structure and framework needed for engaged patient and family input; the OPCE motivated and organized key areas of involvement and led communication efforts across the organization. The IPFCC coaching calls provided the underlying guidance and accountability needed to sustain momentum. As leadership and frontline teams became aware of the vision, they understood the larger connection to the system’s purpose, which ultimately created a clear path for positive change.
Meaningful involvement and input of patient and family partners. Throughout this project, Northwell’s patient/family partners were involved through the PFPC and local councils. For example, patient/family partners attended every IPFCC coaching call; members had a central voice in every decision made within each SWT; and local PFPCs actively participated in physical signage “walk-abouts” across facilities, making key recommendations for improvement. This multifaceted, supportive collaboration created a rejuvenated and purposeful focus for all council members involved. Some of their reactions include, “…I am so happy to be able to help other families in crisis, so that they don’t have to be alone, like I was,” and “I feel how important the patient and family’s voice is … it’s truly a partnership between patients, families, and staff.”
Regular access to IPFCC as a best practice coach and expert resource. Throughout the 10-month process, IPFCC’s Better Together Learning Community for Hospitals in New York State provided ongoing learning interventions for members of the SWT; multiple and varied resources from the Better Together toolkit for adaptation; and opportunities to share and reinforce new, learned expertise with colleagues within the Northwell Health system and beyond through IPFCC’s free online learning community, PFCC.Connect.
Conclusion
Family presence and participation are important to the quality, experience, safety, and outcomes of care. IPFCC’s campaign, Better Together: Partnering with Families, encourages hospitals to change restrictive visiting policies and, instead, to welcome families and caregivers 24 hours a day.
Two projects within Better Together involving almost 50 acute care hospitals in New York State confirm that change in policy, practice, and communication is particularly effective when implemented with strong support from leadership. An intervention like the Better Together Learning Community, offering structured training, coaching, and resources, can facilitate the change process.
Corresponding author: IPFCC, Deborah L. Dokken, 6917 Arlington Rd., Ste. 309, Bethesda, MD 20814; [email protected].
Funding disclosures: None.
1. Dokken DL, Kaufman J, Johnson BJ et al. Changing hospital visiting policies: from families as “visitors” to families as partners. J Clin Outcomes Manag. 2015; 22:29-36.
2. New York Public Interest Research Group and New Yorkers for Patient & Family Empowerment. Sick, scared and separated from loved ones. third edition: A pathway to improvement in New York City. New York: NYPIRG: 2018. www.nypirg.org/pubs/201801/NYPIRG_SICK_SCARED_FINAL.pdf. Accessed December 12, 2019.
3. Institute for Patient- and Family-Centered Care. Better Together: Partnering with Families. www.ipfcc.org/bestpractices/better-together.html. Accessed December 12, 2019.
4. Canadian Foundation for Healthcare Improvement. Better Together. www.cfhi-fcass.ca/WhatWeDo/better-together. Accessed December 12, 2019.
5. Canadian Foundation for Healthcare Improvement. Better Together: A change package to support the adoption of family presence and participation in acute care hospitals and accelerate healthcare improvement. www.cfhi-fcass.ca/sf-docs/default-source/patient-engagement/better-together-change-package.pdf?sfvrsn=9656d044_4. Accessed December 12, 2019.
6. Canadian Foundation for Healthcare Improvement. L’Objectif santé: main dans la main avec les familles. www.cfhi-fcass.ca/sf-docs/default-source/patient-engagement/families-pocket-screen_fr.pdf. Accessed December 12, 2019.
7. New York Public Interest Research Group and New Yorkers for Patient & Family Empowerment. Sick, scared and separated from loved ones. fourth edition: A pathway to improvement in New York. New York: NYPIRG: 2019. www.nypirg.org/pubs/201911/Sick_Scared_Separated_2019_web_FINAL.pdf. Accessed December 12, 2019.
8. Northwell Health. Patient and Family Partnership Councils. www.northwell.edu/about/commitment-to-excellence/patient-and-customer-experience/care-delivery-hospitality. Accessed December 12, 2019.
9 . Institute for Patient- and Family-Centered Care. How to conduct a “walk-about” from the patient and family perspective. www.ipfcc.org/resources/How_To_Conduct_A_Walk-About.pdf. Accessed December 12, 2019.
From the Institute for Patient- and Family-Centered Care, Bethesda, MD (Ms. Dokken and Ms. Johnson), and Northwell Health, New Hyde Park, NY (Dr. Barden, Ms. Tuomey, and Ms. Giammarinaro).
Abstract
Objective: To describe the growth of Better Together: Partnering with Families, a campaign launched in 2014 to eliminate restrictive hospital visiting policies and to put in place policies that recognize families as partners in care, and to discuss the processes involved in implementing the initiative in a large, integrated health system.
Methods: Descriptive report.
Results: In June 2014, the Institute for Patient- and Family-Centered Care (IPFCC) launched the Better Together campaign to emphasize the importance of family presence and participation to the quality, experience, safety, and outcomes of care. Since then, this initiative has expanded in both the United States and Canada. With support from 2 funders in the United States, special attention was focused on acute care hospitals across New York State. Nearly 50 hospitals participated in 2 separate but related projects. Fifteen of the hospitals are part of Northwell Health, New York State’s largest health system. Over a 10-month period, these hospitals made significant progress in changing policy, practice, and communication to support family presence.
Conclusion: The Better Together initiative was implemented across a health system with strong support from leadership and the involvement of patient and family advisors. An intervention offering structured training, coaching, and resources, like IPFCC’s Better Together initiative, can facilitate the change process.
Keywords: family presence; visiting policies; patient-centered care; family-centered care; patient experience.
The presence of families at the bedside of patients is often restricted by hospital visiting hours. Hospitals that maintain these restrictive policies cite concerns about negative impacts on security, infection control, privacy, and staff workload. But there are no data to support these concerns, and the experience of hospitals that have successfully changed policy and practice to welcome families demonstrates the potential positive impacts of less restrictive policies on patient care and outcomes.1 For example, hospitalization can lead to reduced cognitive function in elderly patients. Family members would recognize the changes and could provide valuable information to hospital staff, potentially improving outcomes.2
In June 2014, the Institute for Patient- and Family-Centered Care (IPFCC) launched the campaign Better Together: Partnering with Families.3 The campaign is is grounded in patient- and family- centered care, an approach to care that supports partnerships among health care providers, patients, and families, and, among other core principles, advocates that patients define their “families” and how they will participate in care and decision-making.
Emphasizing the importance of family presence and participation to quality and safety, the Better Together campaign seeks to eliminate restrictive visiting policies and calls upon hospitals to include families as members of the care team and to welcome them 24 hours a day, 7 days a week, according to patient preference. As part of the campaign, IPFCC developed an extensive toolkit of resources that is available to hospitals and other organizations at no cost. The resources include sample policies; profiles of hospitals that have implemented family presence policies; educational materials for staff, patients, and families; and a template for hospital websites. This article, a follow-up to an article published in the January 2015 issue of JCOM,1 discusses the growth of the Better Together initiative as well as the processes involved in implementing the initiative across a large health system.
Growth of the Initiative
Since its launch in 2014, the Better Together initiative has continued to expand in the United States and Canada. In Canada, under the leadership of the Canadian Foundation for Healthcare Improvement (CFHI), more than 50 organizations have made a commitment to the Better Together program and family presence.4 Utilizing and adapting IPFCC’s Toolkit, CFHI developed a change package of free resources for Canadian organizations.5 Some of the materials, including the Pocket Guide for Families (Manuel des Familles), were translated into French.6
With support from 2 funders in the United States, the United Hospital Fund and the New York State Health (NYSHealth) Foundation, through a subcontract with the New York Public Interest Research Group (NYPIRG), IPFCC has been able to focus on hospitals in New York City, including public hospitals, and, more broadly, acute care hospitals across New York State. Nearly 50 hospitals participated in these 2 separate but related projects.
Education and Support for New York City Hospitals
Supported by the United Hospital Fund, an 18-month project that focused specifically on New York City hospitals was completed in June 2017. The project began with a 1-day intensive training event with representatives of 21 hospitals. Eighteen of those hospitals were eligible to participate in follow-up consultation provided by IPFCC, and 14 participated in some kind of follow-up. NYC Health + Hospitals (H+H), the system of public hospitals in NYC, participated most fully in these activities.
The outcomes of the Better Together initiative in New York City are summarized in the report Sick, Scared, & Separated From Loved Ones,2 which is based on a pre/post review of hospital visitation/family presence policies and website communications. According to the report, hospitals that participated in the IPFCC training and consultation program performed better, as a group, with respect to improved policy and website scores on post review than those that did not. Of the 10 hospitals whose scores improved during the review period, 8 had participated in the IPFCC training and 1 hospital was part of a hospital network that did so. (Six of these hospitals are part of the H+H public hospital system.) Those 9 hospitals saw an average increase in scores of 4.9 points (out of a possible 11).
A Learning Community for Hospitals in New York State
With support from the NYSHealth Foundation, IPFCC again collaborated with NYPIRG and New Yorkers for Patient & Family Empowerment on a 2-year initiative, completed in November 2019, that involved 26 hospitals: 15 from Northwell Health, New York State’s largest health system, and 11 hospitals from health systems throughout the state (Greater Hudson Valley Health System, now Garnet Health; Mohawk Valley Health System; Rochester Regional Health; and University of Vermont Health Network). An update of the report Sick, Scared, & Separated From Loved Onescompared pre/post reviews of policies and website communications regarding hospital visitation/family presence.7 Its findings confirm that hospitals that participated in the Better Together Learning Community improved both their policy and website scores to a greater degree than hospitals that did not participate and that a planned intervention can help facilitate change.
During the survey period, 28 out of 40 hospitals’ website navigability scores improved. Of those, hospitals that did not participate in the Better Together Learning Community saw an average increase in scores of 1.2 points, out of a possible 11, while the participating hospitals saw an average increase of 2.7 points, with the top 5 largest increases in scores belonging to hospitals that participated in the Better Together Learning Community.7
The Northwell Health Experience
Northwell Health is a large integrated health care organization comprising more than 69,000 employees, 23 hospitals, and more than 750 medical practices, located geographically across New York State. Embracing patient- and family-centered care, Northwell is dedicated to improving the quality, experience, and safety of care for patients and their families. Welcoming and including patients, families, and care partners as members of the health care team has always been a core element of Northwell’s organizational goal of providing world-class patient care and experience.
Four years ago, the organization reorganized and formalized a system-wide Patient & Family Partnership Council (PFPC).8 Representatives on the PFPC include a Northwell patient experience leader and patient/family co-chair from local councils that have been established in nearly all 23 hospitals as well as service lines. Modeling partnership, the PFPC is grounded in listening to the “voice” of patients and families and promoting collaboration, with the goal of driving change across varied aspects and experiences of health care delivery.
Through the Office of Patient and Customer Experience (OPCE), a partnership with IPFCC and the Better Together Learning Community for Hospitals in New York State was initiated as a fundamental next step in Northwell’s journey to enhance system-wide family presence and participation. Results from Better Together’s Organizational Self-Assessment Tool and process identified opportunities to influence 3 distinct areas: policy/staff education, position descriptions/performance management, and website/signage. Over a 10-month period (September 2018 through June 2019), 15 Northwell hospitals implemened significant patient- and family-centered improvements through multifaceted shared work teams (SWT) that partnered around the common goal of supporting the patient and family experience (Figure). Northwell’s SWT structure allowed teams to meet individually on specific tasks, led by a dedicated staff member of the OPCE to ensure progress, support, and accountability. Six monthly coaching calls or report-out meetings were attended by participating teams, where feedback and recommendations shared by IPFCC were discussed in order to maintain momentum and results.

Policy/Staff Education
The policy/staff education SWT focused on appraising and updating existing policies to ensure alignment with key patient- and family-centered concepts and Better Together principles (Table 1). By establishing representation on the System Policy and Procedure Committee, OPCE enabled patients and families to have a voice at the decision-making table. OPCE leaders presented the ideology and scope of the transformation to this committee. After reviewing all system-wide policies, 4 were identified as key opportunities for revision. One overarching policy titled “Visitation Guidelines” was reviewed and updated to reflect Northwell’s mission of patient- and family-centered care, retiring the reference to “families” as “visitors” in definitions, incorporating language of inclusion and partnership, and citing other related policies. The policy was vetted through a multilayer process of review and stakeholder feedback and was ultimately approved at a system

Three additional related policies were also updated to reflect core principles of inclusion and partnership. These included system policies focused on discharge planning; identification of health care proxy, agent, support person and caregiver; and standards of behavior not conducive in a health care setting. As a result of this work, OPCE was invited to remain an active member of the System Policy and Procedure Committee, adding meaningful new perspectives to the clinical and administrative policy management process. Once policies were updated and approved, the SWT focused on educating leaders and teams. Using a diversified strategy, education was provided through various modes, including weekly system-wide internal communication channels, patient experience huddle messages, yearly mandatory topics training, and the incorporation of essential concepts in existing educational courses (classroom and e-learning modalities).
Position Descriptions/Performance Management
The position descriptions/performance management SWT focused its efforts on incorporating patient- and family-centered concepts and language into position descriptions and the performance appraisal process (Table 2). Due to the complex nature of this work, the process required collaboration from key subject matter experts in human resources, talent management, corporate compensation, and labor management. In 2019, Northwell began an initiative focused on streamlining and standardizing job titles, roles, and developmental pathways across the system. The overarching goal was to create system-wide consistency and standardization. The SWT was successful in advising the leaders overseeing this job architecture initiative on the importance of including language of patient- and family-centered care, like partnership and collaboration, and of highlighting the critical role of family members as part of the care team in subsequent documents.

Northwell has 6 behavioral expectations, standards to which all team members are held accountable: Patient/Customer Focus, Teamwork, Execution, Organizational Awareness, Enable Change, and Develop Self. As a result of the SWT’s work, Patient/Customer Focus was revised to include “families” as essential care partners, demonstrating Northwell’s ongoing commitment to honoring the role of families as members of the care team. It also ensures that all employees are aligned around this priority, as these expectations are utilized to support areas such as recognition and performance. Collaborating with talent management and organizational development, the SWT reviewed yearly performance management and new-hire evaluations. In doing so, they identified an opportunity to refresh the anchored qualitative rating scales to include behavioral demonstrations of patient- and family-centered care, collaboration, respect, and partnership with family members.
Website/Signage
Websites make an important first impression on patients and families looking for information to best prepare for a hospital experience. Therefore, the website/signage SWT worked to redesign hospital websites, enhance digital signage, and perform a baseline assessment of physical signage across facilities. Initial feedback on Northwell’s websites identified opportunities to include more patient- and family-centered, care-partner-infused language; improve navigation; and streamline click levels for easier access. Content for the websites was carefully crafted in collaboration with Northwell’s internal web team, utilizing IPFCC’s best practice standards as a framework and guide.
Next, a multidisciplinary website shared-governance team was established by the OPCE to ensure that key stakeholders were represented and had the opportunity to review and make recommendations for appropriate language and messaging about family presence and participation. This 13-person team was comprised of patient/family partners, patient-experience culture leaders, quality, compliance, human resources, policy, a chief nursing officer, a medical director, and representation from the Institute for Nursing. After careful review and consideration from Northwell’s family partners and teams, all participating hospital websites were enhanced as of June 2019 to include prominent 1-click access from homepages to information for “patients, families and visitors,” as well as “your care partners” information on the important role of families and care partners.
Along with refreshing websites, another step in Northwell’s work to strengthen messaging about family presence and participation was to partner and collaborate with the system’s digital web team as well as local facility councils to understand the capacity to adjust digital signage across facilities. Opportunities were found to make simple yet effective enhancements to the language and imagery of digital signage upon entry, creating a warmer and more welcoming first impression for patients and families. With patient and family partner feedback, the team designed digital signage with inclusive messaging and images that would circulate appropriately based on the facility. Signage specifically welcomes families and refers to them as members of patients’ care teams.
Northwell’s website/signage SWT also directed a 2-phase physical signage assessment to determine ongoing opportunities to alter signs in areas that particularly impact patients and families, such as emergency departments, main lobbies, cafeterias, surgical waiting areas, and intensive care units. Each hospital’s local PFPC did a “walk-about”9 to make enhancements to physical signage, such as removing paper and overcrowded signs, adjusting negative language, ensuring alignment with brand guidelines, and including language that welcomed families. As a result of the team’s efforts around signage, collaboration began with the health system’s signage committee to help standardize signage terminology to reflect family inclusiveness, and to implement the recommendation for a standardized signage shared-governance team to ensure accountability and a patient- and family-centered structure.
Sustainment
Since implementing Better Together, Northwell has been able to infuse a more patient- and family-centered emphasis into its overall patient experience message of “Every role, every person, every moment matters.” As a strategic tool aimed at encouraging leaders, clinicians, and staff to pause and reflect about the “heart” of their work, patient and family stories are now included at the beginning of meetings, forums, and team huddles. Elements of the initiative have been integrated in current Patient and Family Partnership sustainment plans at participating hospitals. Some highlights include continued integration of patient/family partners on committees and councils that impact areas such as way finding, signage, recruitment, new-hire orientation, and community outreach; focus on enhancing partner retention and development programs; and inclusion of patient- and family-centered care and Better Together principles in ongoing leadership meetings.
Factors Contributing to Success
Health care is a complex, regulated, and often bureaucratic world that can be very difficult for patients and families to navigate. The system’s partnership with the Better Together Learning Community for Hospitals in New York State enhanced its efforts to improve family presence and participation and created powerful synergy. The success of this partnership was based on a number of important factors:
A solid foundation of support, structure, and accountability. The OPCE initiated the IPFCC Better Together partnership and established a synergistic collaboration inclusive of leadership, frontline teams, multiple departments, and patient and family partners. As a major strategic component of Northwell’s mission to deliver high-quality, patient- and family-centered care, OPCE was instrumental in connecting key areas and stakeholders and mobilizing the recommendations coming from patients and families.
A visible commitment of leadership at all levels. Partnering with leadership across Northwell’s system required a delineated vision, clear purpose and ownership, and comprehensive implementation and sustainment strategies. The existing format of Northwell’s PFPC provided the structure and framework needed for engaged patient and family input; the OPCE motivated and organized key areas of involvement and led communication efforts across the organization. The IPFCC coaching calls provided the underlying guidance and accountability needed to sustain momentum. As leadership and frontline teams became aware of the vision, they understood the larger connection to the system’s purpose, which ultimately created a clear path for positive change.
Meaningful involvement and input of patient and family partners. Throughout this project, Northwell’s patient/family partners were involved through the PFPC and local councils. For example, patient/family partners attended every IPFCC coaching call; members had a central voice in every decision made within each SWT; and local PFPCs actively participated in physical signage “walk-abouts” across facilities, making key recommendations for improvement. This multifaceted, supportive collaboration created a rejuvenated and purposeful focus for all council members involved. Some of their reactions include, “…I am so happy to be able to help other families in crisis, so that they don’t have to be alone, like I was,” and “I feel how important the patient and family’s voice is … it’s truly a partnership between patients, families, and staff.”
Regular access to IPFCC as a best practice coach and expert resource. Throughout the 10-month process, IPFCC’s Better Together Learning Community for Hospitals in New York State provided ongoing learning interventions for members of the SWT; multiple and varied resources from the Better Together toolkit for adaptation; and opportunities to share and reinforce new, learned expertise with colleagues within the Northwell Health system and beyond through IPFCC’s free online learning community, PFCC.Connect.
Conclusion
Family presence and participation are important to the quality, experience, safety, and outcomes of care. IPFCC’s campaign, Better Together: Partnering with Families, encourages hospitals to change restrictive visiting policies and, instead, to welcome families and caregivers 24 hours a day.
Two projects within Better Together involving almost 50 acute care hospitals in New York State confirm that change in policy, practice, and communication is particularly effective when implemented with strong support from leadership. An intervention like the Better Together Learning Community, offering structured training, coaching, and resources, can facilitate the change process.
Corresponding author: IPFCC, Deborah L. Dokken, 6917 Arlington Rd., Ste. 309, Bethesda, MD 20814; [email protected].
Funding disclosures: None.
From the Institute for Patient- and Family-Centered Care, Bethesda, MD (Ms. Dokken and Ms. Johnson), and Northwell Health, New Hyde Park, NY (Dr. Barden, Ms. Tuomey, and Ms. Giammarinaro).
Abstract
Objective: To describe the growth of Better Together: Partnering with Families, a campaign launched in 2014 to eliminate restrictive hospital visiting policies and to put in place policies that recognize families as partners in care, and to discuss the processes involved in implementing the initiative in a large, integrated health system.
Methods: Descriptive report.
Results: In June 2014, the Institute for Patient- and Family-Centered Care (IPFCC) launched the Better Together campaign to emphasize the importance of family presence and participation to the quality, experience, safety, and outcomes of care. Since then, this initiative has expanded in both the United States and Canada. With support from 2 funders in the United States, special attention was focused on acute care hospitals across New York State. Nearly 50 hospitals participated in 2 separate but related projects. Fifteen of the hospitals are part of Northwell Health, New York State’s largest health system. Over a 10-month period, these hospitals made significant progress in changing policy, practice, and communication to support family presence.
Conclusion: The Better Together initiative was implemented across a health system with strong support from leadership and the involvement of patient and family advisors. An intervention offering structured training, coaching, and resources, like IPFCC’s Better Together initiative, can facilitate the change process.
Keywords: family presence; visiting policies; patient-centered care; family-centered care; patient experience.
The presence of families at the bedside of patients is often restricted by hospital visiting hours. Hospitals that maintain these restrictive policies cite concerns about negative impacts on security, infection control, privacy, and staff workload. But there are no data to support these concerns, and the experience of hospitals that have successfully changed policy and practice to welcome families demonstrates the potential positive impacts of less restrictive policies on patient care and outcomes.1 For example, hospitalization can lead to reduced cognitive function in elderly patients. Family members would recognize the changes and could provide valuable information to hospital staff, potentially improving outcomes.2
In June 2014, the Institute for Patient- and Family-Centered Care (IPFCC) launched the campaign Better Together: Partnering with Families.3 The campaign is is grounded in patient- and family- centered care, an approach to care that supports partnerships among health care providers, patients, and families, and, among other core principles, advocates that patients define their “families” and how they will participate in care and decision-making.
Emphasizing the importance of family presence and participation to quality and safety, the Better Together campaign seeks to eliminate restrictive visiting policies and calls upon hospitals to include families as members of the care team and to welcome them 24 hours a day, 7 days a week, according to patient preference. As part of the campaign, IPFCC developed an extensive toolkit of resources that is available to hospitals and other organizations at no cost. The resources include sample policies; profiles of hospitals that have implemented family presence policies; educational materials for staff, patients, and families; and a template for hospital websites. This article, a follow-up to an article published in the January 2015 issue of JCOM,1 discusses the growth of the Better Together initiative as well as the processes involved in implementing the initiative across a large health system.
Growth of the Initiative
Since its launch in 2014, the Better Together initiative has continued to expand in the United States and Canada. In Canada, under the leadership of the Canadian Foundation for Healthcare Improvement (CFHI), more than 50 organizations have made a commitment to the Better Together program and family presence.4 Utilizing and adapting IPFCC’s Toolkit, CFHI developed a change package of free resources for Canadian organizations.5 Some of the materials, including the Pocket Guide for Families (Manuel des Familles), were translated into French.6
With support from 2 funders in the United States, the United Hospital Fund and the New York State Health (NYSHealth) Foundation, through a subcontract with the New York Public Interest Research Group (NYPIRG), IPFCC has been able to focus on hospitals in New York City, including public hospitals, and, more broadly, acute care hospitals across New York State. Nearly 50 hospitals participated in these 2 separate but related projects.
Education and Support for New York City Hospitals
Supported by the United Hospital Fund, an 18-month project that focused specifically on New York City hospitals was completed in June 2017. The project began with a 1-day intensive training event with representatives of 21 hospitals. Eighteen of those hospitals were eligible to participate in follow-up consultation provided by IPFCC, and 14 participated in some kind of follow-up. NYC Health + Hospitals (H+H), the system of public hospitals in NYC, participated most fully in these activities.
The outcomes of the Better Together initiative in New York City are summarized in the report Sick, Scared, & Separated From Loved Ones,2 which is based on a pre/post review of hospital visitation/family presence policies and website communications. According to the report, hospitals that participated in the IPFCC training and consultation program performed better, as a group, with respect to improved policy and website scores on post review than those that did not. Of the 10 hospitals whose scores improved during the review period, 8 had participated in the IPFCC training and 1 hospital was part of a hospital network that did so. (Six of these hospitals are part of the H+H public hospital system.) Those 9 hospitals saw an average increase in scores of 4.9 points (out of a possible 11).
A Learning Community for Hospitals in New York State
With support from the NYSHealth Foundation, IPFCC again collaborated with NYPIRG and New Yorkers for Patient & Family Empowerment on a 2-year initiative, completed in November 2019, that involved 26 hospitals: 15 from Northwell Health, New York State’s largest health system, and 11 hospitals from health systems throughout the state (Greater Hudson Valley Health System, now Garnet Health; Mohawk Valley Health System; Rochester Regional Health; and University of Vermont Health Network). An update of the report Sick, Scared, & Separated From Loved Onescompared pre/post reviews of policies and website communications regarding hospital visitation/family presence.7 Its findings confirm that hospitals that participated in the Better Together Learning Community improved both their policy and website scores to a greater degree than hospitals that did not participate and that a planned intervention can help facilitate change.
During the survey period, 28 out of 40 hospitals’ website navigability scores improved. Of those, hospitals that did not participate in the Better Together Learning Community saw an average increase in scores of 1.2 points, out of a possible 11, while the participating hospitals saw an average increase of 2.7 points, with the top 5 largest increases in scores belonging to hospitals that participated in the Better Together Learning Community.7
The Northwell Health Experience
Northwell Health is a large integrated health care organization comprising more than 69,000 employees, 23 hospitals, and more than 750 medical practices, located geographically across New York State. Embracing patient- and family-centered care, Northwell is dedicated to improving the quality, experience, and safety of care for patients and their families. Welcoming and including patients, families, and care partners as members of the health care team has always been a core element of Northwell’s organizational goal of providing world-class patient care and experience.
Four years ago, the organization reorganized and formalized a system-wide Patient & Family Partnership Council (PFPC).8 Representatives on the PFPC include a Northwell patient experience leader and patient/family co-chair from local councils that have been established in nearly all 23 hospitals as well as service lines. Modeling partnership, the PFPC is grounded in listening to the “voice” of patients and families and promoting collaboration, with the goal of driving change across varied aspects and experiences of health care delivery.
Through the Office of Patient and Customer Experience (OPCE), a partnership with IPFCC and the Better Together Learning Community for Hospitals in New York State was initiated as a fundamental next step in Northwell’s journey to enhance system-wide family presence and participation. Results from Better Together’s Organizational Self-Assessment Tool and process identified opportunities to influence 3 distinct areas: policy/staff education, position descriptions/performance management, and website/signage. Over a 10-month period (September 2018 through June 2019), 15 Northwell hospitals implemened significant patient- and family-centered improvements through multifaceted shared work teams (SWT) that partnered around the common goal of supporting the patient and family experience (Figure). Northwell’s SWT structure allowed teams to meet individually on specific tasks, led by a dedicated staff member of the OPCE to ensure progress, support, and accountability. Six monthly coaching calls or report-out meetings were attended by participating teams, where feedback and recommendations shared by IPFCC were discussed in order to maintain momentum and results.

Policy/Staff Education
The policy/staff education SWT focused on appraising and updating existing policies to ensure alignment with key patient- and family-centered concepts and Better Together principles (Table 1). By establishing representation on the System Policy and Procedure Committee, OPCE enabled patients and families to have a voice at the decision-making table. OPCE leaders presented the ideology and scope of the transformation to this committee. After reviewing all system-wide policies, 4 were identified as key opportunities for revision. One overarching policy titled “Visitation Guidelines” was reviewed and updated to reflect Northwell’s mission of patient- and family-centered care, retiring the reference to “families” as “visitors” in definitions, incorporating language of inclusion and partnership, and citing other related policies. The policy was vetted through a multilayer process of review and stakeholder feedback and was ultimately approved at a system

Three additional related policies were also updated to reflect core principles of inclusion and partnership. These included system policies focused on discharge planning; identification of health care proxy, agent, support person and caregiver; and standards of behavior not conducive in a health care setting. As a result of this work, OPCE was invited to remain an active member of the System Policy and Procedure Committee, adding meaningful new perspectives to the clinical and administrative policy management process. Once policies were updated and approved, the SWT focused on educating leaders and teams. Using a diversified strategy, education was provided through various modes, including weekly system-wide internal communication channels, patient experience huddle messages, yearly mandatory topics training, and the incorporation of essential concepts in existing educational courses (classroom and e-learning modalities).
Position Descriptions/Performance Management
The position descriptions/performance management SWT focused its efforts on incorporating patient- and family-centered concepts and language into position descriptions and the performance appraisal process (Table 2). Due to the complex nature of this work, the process required collaboration from key subject matter experts in human resources, talent management, corporate compensation, and labor management. In 2019, Northwell began an initiative focused on streamlining and standardizing job titles, roles, and developmental pathways across the system. The overarching goal was to create system-wide consistency and standardization. The SWT was successful in advising the leaders overseeing this job architecture initiative on the importance of including language of patient- and family-centered care, like partnership and collaboration, and of highlighting the critical role of family members as part of the care team in subsequent documents.

Northwell has 6 behavioral expectations, standards to which all team members are held accountable: Patient/Customer Focus, Teamwork, Execution, Organizational Awareness, Enable Change, and Develop Self. As a result of the SWT’s work, Patient/Customer Focus was revised to include “families” as essential care partners, demonstrating Northwell’s ongoing commitment to honoring the role of families as members of the care team. It also ensures that all employees are aligned around this priority, as these expectations are utilized to support areas such as recognition and performance. Collaborating with talent management and organizational development, the SWT reviewed yearly performance management and new-hire evaluations. In doing so, they identified an opportunity to refresh the anchored qualitative rating scales to include behavioral demonstrations of patient- and family-centered care, collaboration, respect, and partnership with family members.
Website/Signage
Websites make an important first impression on patients and families looking for information to best prepare for a hospital experience. Therefore, the website/signage SWT worked to redesign hospital websites, enhance digital signage, and perform a baseline assessment of physical signage across facilities. Initial feedback on Northwell’s websites identified opportunities to include more patient- and family-centered, care-partner-infused language; improve navigation; and streamline click levels for easier access. Content for the websites was carefully crafted in collaboration with Northwell’s internal web team, utilizing IPFCC’s best practice standards as a framework and guide.
Next, a multidisciplinary website shared-governance team was established by the OPCE to ensure that key stakeholders were represented and had the opportunity to review and make recommendations for appropriate language and messaging about family presence and participation. This 13-person team was comprised of patient/family partners, patient-experience culture leaders, quality, compliance, human resources, policy, a chief nursing officer, a medical director, and representation from the Institute for Nursing. After careful review and consideration from Northwell’s family partners and teams, all participating hospital websites were enhanced as of June 2019 to include prominent 1-click access from homepages to information for “patients, families and visitors,” as well as “your care partners” information on the important role of families and care partners.
Along with refreshing websites, another step in Northwell’s work to strengthen messaging about family presence and participation was to partner and collaborate with the system’s digital web team as well as local facility councils to understand the capacity to adjust digital signage across facilities. Opportunities were found to make simple yet effective enhancements to the language and imagery of digital signage upon entry, creating a warmer and more welcoming first impression for patients and families. With patient and family partner feedback, the team designed digital signage with inclusive messaging and images that would circulate appropriately based on the facility. Signage specifically welcomes families and refers to them as members of patients’ care teams.
Northwell’s website/signage SWT also directed a 2-phase physical signage assessment to determine ongoing opportunities to alter signs in areas that particularly impact patients and families, such as emergency departments, main lobbies, cafeterias, surgical waiting areas, and intensive care units. Each hospital’s local PFPC did a “walk-about”9 to make enhancements to physical signage, such as removing paper and overcrowded signs, adjusting negative language, ensuring alignment with brand guidelines, and including language that welcomed families. As a result of the team’s efforts around signage, collaboration began with the health system’s signage committee to help standardize signage terminology to reflect family inclusiveness, and to implement the recommendation for a standardized signage shared-governance team to ensure accountability and a patient- and family-centered structure.
Sustainment
Since implementing Better Together, Northwell has been able to infuse a more patient- and family-centered emphasis into its overall patient experience message of “Every role, every person, every moment matters.” As a strategic tool aimed at encouraging leaders, clinicians, and staff to pause and reflect about the “heart” of their work, patient and family stories are now included at the beginning of meetings, forums, and team huddles. Elements of the initiative have been integrated in current Patient and Family Partnership sustainment plans at participating hospitals. Some highlights include continued integration of patient/family partners on committees and councils that impact areas such as way finding, signage, recruitment, new-hire orientation, and community outreach; focus on enhancing partner retention and development programs; and inclusion of patient- and family-centered care and Better Together principles in ongoing leadership meetings.
Factors Contributing to Success
Health care is a complex, regulated, and often bureaucratic world that can be very difficult for patients and families to navigate. The system’s partnership with the Better Together Learning Community for Hospitals in New York State enhanced its efforts to improve family presence and participation and created powerful synergy. The success of this partnership was based on a number of important factors:
A solid foundation of support, structure, and accountability. The OPCE initiated the IPFCC Better Together partnership and established a synergistic collaboration inclusive of leadership, frontline teams, multiple departments, and patient and family partners. As a major strategic component of Northwell’s mission to deliver high-quality, patient- and family-centered care, OPCE was instrumental in connecting key areas and stakeholders and mobilizing the recommendations coming from patients and families.
A visible commitment of leadership at all levels. Partnering with leadership across Northwell’s system required a delineated vision, clear purpose and ownership, and comprehensive implementation and sustainment strategies. The existing format of Northwell’s PFPC provided the structure and framework needed for engaged patient and family input; the OPCE motivated and organized key areas of involvement and led communication efforts across the organization. The IPFCC coaching calls provided the underlying guidance and accountability needed to sustain momentum. As leadership and frontline teams became aware of the vision, they understood the larger connection to the system’s purpose, which ultimately created a clear path for positive change.
Meaningful involvement and input of patient and family partners. Throughout this project, Northwell’s patient/family partners were involved through the PFPC and local councils. For example, patient/family partners attended every IPFCC coaching call; members had a central voice in every decision made within each SWT; and local PFPCs actively participated in physical signage “walk-abouts” across facilities, making key recommendations for improvement. This multifaceted, supportive collaboration created a rejuvenated and purposeful focus for all council members involved. Some of their reactions include, “…I am so happy to be able to help other families in crisis, so that they don’t have to be alone, like I was,” and “I feel how important the patient and family’s voice is … it’s truly a partnership between patients, families, and staff.”
Regular access to IPFCC as a best practice coach and expert resource. Throughout the 10-month process, IPFCC’s Better Together Learning Community for Hospitals in New York State provided ongoing learning interventions for members of the SWT; multiple and varied resources from the Better Together toolkit for adaptation; and opportunities to share and reinforce new, learned expertise with colleagues within the Northwell Health system and beyond through IPFCC’s free online learning community, PFCC.Connect.
Conclusion
Family presence and participation are important to the quality, experience, safety, and outcomes of care. IPFCC’s campaign, Better Together: Partnering with Families, encourages hospitals to change restrictive visiting policies and, instead, to welcome families and caregivers 24 hours a day.
Two projects within Better Together involving almost 50 acute care hospitals in New York State confirm that change in policy, practice, and communication is particularly effective when implemented with strong support from leadership. An intervention like the Better Together Learning Community, offering structured training, coaching, and resources, can facilitate the change process.
Corresponding author: IPFCC, Deborah L. Dokken, 6917 Arlington Rd., Ste. 309, Bethesda, MD 20814; [email protected].
Funding disclosures: None.
1. Dokken DL, Kaufman J, Johnson BJ et al. Changing hospital visiting policies: from families as “visitors” to families as partners. J Clin Outcomes Manag. 2015; 22:29-36.
2. New York Public Interest Research Group and New Yorkers for Patient & Family Empowerment. Sick, scared and separated from loved ones. third edition: A pathway to improvement in New York City. New York: NYPIRG: 2018. www.nypirg.org/pubs/201801/NYPIRG_SICK_SCARED_FINAL.pdf. Accessed December 12, 2019.
3. Institute for Patient- and Family-Centered Care. Better Together: Partnering with Families. www.ipfcc.org/bestpractices/better-together.html. Accessed December 12, 2019.
4. Canadian Foundation for Healthcare Improvement. Better Together. www.cfhi-fcass.ca/WhatWeDo/better-together. Accessed December 12, 2019.
5. Canadian Foundation for Healthcare Improvement. Better Together: A change package to support the adoption of family presence and participation in acute care hospitals and accelerate healthcare improvement. www.cfhi-fcass.ca/sf-docs/default-source/patient-engagement/better-together-change-package.pdf?sfvrsn=9656d044_4. Accessed December 12, 2019.
6. Canadian Foundation for Healthcare Improvement. L’Objectif santé: main dans la main avec les familles. www.cfhi-fcass.ca/sf-docs/default-source/patient-engagement/families-pocket-screen_fr.pdf. Accessed December 12, 2019.
7. New York Public Interest Research Group and New Yorkers for Patient & Family Empowerment. Sick, scared and separated from loved ones. fourth edition: A pathway to improvement in New York. New York: NYPIRG: 2019. www.nypirg.org/pubs/201911/Sick_Scared_Separated_2019_web_FINAL.pdf. Accessed December 12, 2019.
8. Northwell Health. Patient and Family Partnership Councils. www.northwell.edu/about/commitment-to-excellence/patient-and-customer-experience/care-delivery-hospitality. Accessed December 12, 2019.
9 . Institute for Patient- and Family-Centered Care. How to conduct a “walk-about” from the patient and family perspective. www.ipfcc.org/resources/How_To_Conduct_A_Walk-About.pdf. Accessed December 12, 2019.
1. Dokken DL, Kaufman J, Johnson BJ et al. Changing hospital visiting policies: from families as “visitors” to families as partners. J Clin Outcomes Manag. 2015; 22:29-36.
2. New York Public Interest Research Group and New Yorkers for Patient & Family Empowerment. Sick, scared and separated from loved ones. third edition: A pathway to improvement in New York City. New York: NYPIRG: 2018. www.nypirg.org/pubs/201801/NYPIRG_SICK_SCARED_FINAL.pdf. Accessed December 12, 2019.
3. Institute for Patient- and Family-Centered Care. Better Together: Partnering with Families. www.ipfcc.org/bestpractices/better-together.html. Accessed December 12, 2019.
4. Canadian Foundation for Healthcare Improvement. Better Together. www.cfhi-fcass.ca/WhatWeDo/better-together. Accessed December 12, 2019.
5. Canadian Foundation for Healthcare Improvement. Better Together: A change package to support the adoption of family presence and participation in acute care hospitals and accelerate healthcare improvement. www.cfhi-fcass.ca/sf-docs/default-source/patient-engagement/better-together-change-package.pdf?sfvrsn=9656d044_4. Accessed December 12, 2019.
6. Canadian Foundation for Healthcare Improvement. L’Objectif santé: main dans la main avec les familles. www.cfhi-fcass.ca/sf-docs/default-source/patient-engagement/families-pocket-screen_fr.pdf. Accessed December 12, 2019.
7. New York Public Interest Research Group and New Yorkers for Patient & Family Empowerment. Sick, scared and separated from loved ones. fourth edition: A pathway to improvement in New York. New York: NYPIRG: 2019. www.nypirg.org/pubs/201911/Sick_Scared_Separated_2019_web_FINAL.pdf. Accessed December 12, 2019.
8. Northwell Health. Patient and Family Partnership Councils. www.northwell.edu/about/commitment-to-excellence/patient-and-customer-experience/care-delivery-hospitality. Accessed December 12, 2019.
9 . Institute for Patient- and Family-Centered Care. How to conduct a “walk-about” from the patient and family perspective. www.ipfcc.org/resources/How_To_Conduct_A_Walk-About.pdf. Accessed December 12, 2019.
When Horses and Zebras Coexist: Achieving Diagnostic Excellence in the Age of High-Value Care
Safe, timely, and efficient diagnosis is fundamental for high-quality, effective healthcare. Why is diagnosis so important? First, it informs the two other main areas of medical decision-making: treatment and prognosis. These are the means by which physicians can actually change health outcomes for patients, as well as ensure that patients and their families have a realistic and accurate understanding of what the future holds with respect to their health. Second, patients and families tend to feel a sense of closure from having a name and an explanation for symptoms, even in the absence of specific treatment. Proper labeling allows patients and families to connect with others with the same diagnosis, who are best positioned to offer empathy by virtue of their similar experiences.
Despite the fundamental role of diagnosis, diagnostic error is pervasive in medicine, with unacceptable levels of resultant harm.1 In 2015, the Institute of Medicine published a landmark report, “Improving Diagnosis in Health Care,” bringing the problem to the forefront of the minds of healthcare professionals and the general public alike. According to the report, “improving the diagnostic process…represents a moral, professional, and public health imperative.”1 We must do more than avoid diagnostic error, however—we must aim to achieve diagnostic excellence. Not getting it wrong is not enough.
There are real challenges to achieving diagnostic safety, let alone excellence. The “churn” of modern hospital medicine does not reward deep diagnostic thought, nor does it often encourage reflection or collaboration, important components of being able to achieve diagnostic excellence.2 Furthermore, despite their years of training, physicians often have difficulty applying probabilistic reasoning and appropriately incorporating diagnostic information in the best evidence-based manner.3,4 In addition, there are no validated measures of diagnostic performance in practice. It is telling that many hospitalists, despite a professed interest in complex diagnosis, would rather be assigned to care for a patient with cellulitis than a patient with a complicated differential diagnosis.
Given these challenges, how can the modern healthcare ecosystem be changed to achieve diagnostic excellence? In this month’s issue of Journal of Hospital Medicine, Singer and colleagues describe a pilot project of a proposed solution to the problem.5 Aptly named, the Socrates Project is an intervention that makes available a team of “diagnosticians” that can be consulted for assistance with challenging diagnostic cases. The physicians on the team volunteer their time, allowing for deep diagnostic evaluation that is not limited by one’s daily workload, thus overcoming one of the major hurdles to achieving diagnostic excellence. The described program also focuses on harnessing the power of teamwork, which is especially relevant given recent descriptions of the effectiveness of collective intelligence in improving diagnostic performance.6 Importantly, the authors recognize that their intervention will not achieve a diagnosis in every case for which they are consulted; rather, they hope that their thorough evaluation will uncover additional potential diagnostic avenues for the referring team to pursue, with a goal to “improve patient care by providing…ideas to reduce—or at least manage—diagnostic uncertainty.”
Programs of this nature are exciting for hospitalists. Hospital medicine is, perhaps, a place in modern medicine where diagnostic excellence has a natural home. Patients admitted to the hospital are acutely (and often severely) ill, and hospitalists are tasked with rapidly identifying the cause of their illness in order to initiate appropriate treatment and accurately inform prognosis. Hospitalists, as generalists, take a broad approach to challenging cases, and they tend to practice in well-resourced environments with nearly every diagnostic modality at their disposal. Many hospitalists would envy participating in a program such as the Socrates Project.
While Singer et al.’s innovation—and the institutional support thereof—should be lauded, some discussion must be had about how to assess the effectiveness of such a program. The authors acknowledge the need for evaluation of both the diagnostic process and the outcomes that process achieves. Measuring diagnostic performance is challenging, however, and while there is substantial progress being made in this area, recent efforts tend to focus on identifying diagnostic errors rather than measuring diagnostic excellence. Moreover, even if a program does improve diagnostic performance, how should we evaluate for unintended consequences of its implementation? In the age of high-value care, how can we ensure that efforts to do a better job of spotting proverbial zebras do not come at the cost of harming too many horses?7
Hospitalists are well primed to answer this question. The juxtaposition of Singer et al.’s article with the Journal of Hospital Medicine’s long-running series on Choosing Wisely®: Things We Do for No Reason™ provides a natural synergy to begin crafting a framework to evaluate unintended consequences of a program in diagnostic excellence. More diagnosis is not the goal; more appropriate diagnosis is what is needed. A clinical program aimed at achieving diagnostic excellence should not employ low-value, wasteful strategies that do not add substantively to the diagnostic process but should instead seek to improve the overall efficiency of even complicated diagnostic odysseys. Avoiding waste throughout will allow for allocation of diagnostic resources where they are needed. In turn, hospitalists can do a better job of correctly identifying both horses and zebras for what they are. While a given hospitalization for a diagnostically complex patient may be relatively expensive, better diagnosis during an index hospitalization is likely to lead to decreased downstream costs, such as those related to readmissions and further testing, as well as better health outcomes.
The Socrates Project, along with similar programs at other institutions, are exciting innovations. These programs are not only likely to be good for patients but are also good for hospitalists. The field of hospital medicine should leverage its collective expertise in clinical medicine, systems of care, and high-value care to become a home for diagnostic excellence.
1. National Academies of Sciences, Engineering, and Medicine. Improving Diagnosis in Health Care. Washington, DC: The National Academies Press; 2015. https://doi.org/10.17226/21794
2. Olson A, Rencic J, Cosby K, et al. Competencies for improving diagnosis: an interprofessional framework for education and training in health care. Diagnosis. 2019;6(4):335-341. https://doi.org/10.1515/dx-2018-0107.
3. Baduashvili A, Guyatt G, Evans AT. ROC anatomy—getting the most out of your diagnostic test. J Gen Intern Med. 2019;34(9):1892-1898. https://doi.org/10.1007/s11606-019-05125-0.
4. Manrai AK, Bhatia G, Strymish J, Kohane IS, Jain SH. Medicine’s uncomfortable relationship with math: calculating positive predictive value. JAMA Intern Med. 2014;174(6):991-993. https://doi.org/10.1001/jamainternmed.2014.1059.
5. Singer BD, Goodwin AM, Patel AA, Vaughan DE. The Socrates Project for difficult diagnosis at Northwestern Medicine. J Hosp Med. 2020;15(2):116-118. https://doi.org/ 10.12788/jhm.3335.
6. Barnett ML, Boddupalli D, Nundy S, Bates DW. Comparative accuracy of diagnosis by collective intelligence of multiple physicians vs individual physicians. JAMA Netw Open. 2019;2(3):e190096. https://doi.org/10.1001/jamanetworkopen.2019.0096.
7. Dhaliwal G. Bringing high-value care to the inpatient teaching service. JAMA Intern Med. 2014;174(7):1021-1022. https://doi.org/10.1001/jamainternmed.2014.2012.
Safe, timely, and efficient diagnosis is fundamental for high-quality, effective healthcare. Why is diagnosis so important? First, it informs the two other main areas of medical decision-making: treatment and prognosis. These are the means by which physicians can actually change health outcomes for patients, as well as ensure that patients and their families have a realistic and accurate understanding of what the future holds with respect to their health. Second, patients and families tend to feel a sense of closure from having a name and an explanation for symptoms, even in the absence of specific treatment. Proper labeling allows patients and families to connect with others with the same diagnosis, who are best positioned to offer empathy by virtue of their similar experiences.
Despite the fundamental role of diagnosis, diagnostic error is pervasive in medicine, with unacceptable levels of resultant harm.1 In 2015, the Institute of Medicine published a landmark report, “Improving Diagnosis in Health Care,” bringing the problem to the forefront of the minds of healthcare professionals and the general public alike. According to the report, “improving the diagnostic process…represents a moral, professional, and public health imperative.”1 We must do more than avoid diagnostic error, however—we must aim to achieve diagnostic excellence. Not getting it wrong is not enough.
There are real challenges to achieving diagnostic safety, let alone excellence. The “churn” of modern hospital medicine does not reward deep diagnostic thought, nor does it often encourage reflection or collaboration, important components of being able to achieve diagnostic excellence.2 Furthermore, despite their years of training, physicians often have difficulty applying probabilistic reasoning and appropriately incorporating diagnostic information in the best evidence-based manner.3,4 In addition, there are no validated measures of diagnostic performance in practice. It is telling that many hospitalists, despite a professed interest in complex diagnosis, would rather be assigned to care for a patient with cellulitis than a patient with a complicated differential diagnosis.
Given these challenges, how can the modern healthcare ecosystem be changed to achieve diagnostic excellence? In this month’s issue of Journal of Hospital Medicine, Singer and colleagues describe a pilot project of a proposed solution to the problem.5 Aptly named, the Socrates Project is an intervention that makes available a team of “diagnosticians” that can be consulted for assistance with challenging diagnostic cases. The physicians on the team volunteer their time, allowing for deep diagnostic evaluation that is not limited by one’s daily workload, thus overcoming one of the major hurdles to achieving diagnostic excellence. The described program also focuses on harnessing the power of teamwork, which is especially relevant given recent descriptions of the effectiveness of collective intelligence in improving diagnostic performance.6 Importantly, the authors recognize that their intervention will not achieve a diagnosis in every case for which they are consulted; rather, they hope that their thorough evaluation will uncover additional potential diagnostic avenues for the referring team to pursue, with a goal to “improve patient care by providing…ideas to reduce—or at least manage—diagnostic uncertainty.”
Programs of this nature are exciting for hospitalists. Hospital medicine is, perhaps, a place in modern medicine where diagnostic excellence has a natural home. Patients admitted to the hospital are acutely (and often severely) ill, and hospitalists are tasked with rapidly identifying the cause of their illness in order to initiate appropriate treatment and accurately inform prognosis. Hospitalists, as generalists, take a broad approach to challenging cases, and they tend to practice in well-resourced environments with nearly every diagnostic modality at their disposal. Many hospitalists would envy participating in a program such as the Socrates Project.
While Singer et al.’s innovation—and the institutional support thereof—should be lauded, some discussion must be had about how to assess the effectiveness of such a program. The authors acknowledge the need for evaluation of both the diagnostic process and the outcomes that process achieves. Measuring diagnostic performance is challenging, however, and while there is substantial progress being made in this area, recent efforts tend to focus on identifying diagnostic errors rather than measuring diagnostic excellence. Moreover, even if a program does improve diagnostic performance, how should we evaluate for unintended consequences of its implementation? In the age of high-value care, how can we ensure that efforts to do a better job of spotting proverbial zebras do not come at the cost of harming too many horses?7
Hospitalists are well primed to answer this question. The juxtaposition of Singer et al.’s article with the Journal of Hospital Medicine’s long-running series on Choosing Wisely®: Things We Do for No Reason™ provides a natural synergy to begin crafting a framework to evaluate unintended consequences of a program in diagnostic excellence. More diagnosis is not the goal; more appropriate diagnosis is what is needed. A clinical program aimed at achieving diagnostic excellence should not employ low-value, wasteful strategies that do not add substantively to the diagnostic process but should instead seek to improve the overall efficiency of even complicated diagnostic odysseys. Avoiding waste throughout will allow for allocation of diagnostic resources where they are needed. In turn, hospitalists can do a better job of correctly identifying both horses and zebras for what they are. While a given hospitalization for a diagnostically complex patient may be relatively expensive, better diagnosis during an index hospitalization is likely to lead to decreased downstream costs, such as those related to readmissions and further testing, as well as better health outcomes.
The Socrates Project, along with similar programs at other institutions, are exciting innovations. These programs are not only likely to be good for patients but are also good for hospitalists. The field of hospital medicine should leverage its collective expertise in clinical medicine, systems of care, and high-value care to become a home for diagnostic excellence.
Safe, timely, and efficient diagnosis is fundamental for high-quality, effective healthcare. Why is diagnosis so important? First, it informs the two other main areas of medical decision-making: treatment and prognosis. These are the means by which physicians can actually change health outcomes for patients, as well as ensure that patients and their families have a realistic and accurate understanding of what the future holds with respect to their health. Second, patients and families tend to feel a sense of closure from having a name and an explanation for symptoms, even in the absence of specific treatment. Proper labeling allows patients and families to connect with others with the same diagnosis, who are best positioned to offer empathy by virtue of their similar experiences.
Despite the fundamental role of diagnosis, diagnostic error is pervasive in medicine, with unacceptable levels of resultant harm.1 In 2015, the Institute of Medicine published a landmark report, “Improving Diagnosis in Health Care,” bringing the problem to the forefront of the minds of healthcare professionals and the general public alike. According to the report, “improving the diagnostic process…represents a moral, professional, and public health imperative.”1 We must do more than avoid diagnostic error, however—we must aim to achieve diagnostic excellence. Not getting it wrong is not enough.
There are real challenges to achieving diagnostic safety, let alone excellence. The “churn” of modern hospital medicine does not reward deep diagnostic thought, nor does it often encourage reflection or collaboration, important components of being able to achieve diagnostic excellence.2 Furthermore, despite their years of training, physicians often have difficulty applying probabilistic reasoning and appropriately incorporating diagnostic information in the best evidence-based manner.3,4 In addition, there are no validated measures of diagnostic performance in practice. It is telling that many hospitalists, despite a professed interest in complex diagnosis, would rather be assigned to care for a patient with cellulitis than a patient with a complicated differential diagnosis.
Given these challenges, how can the modern healthcare ecosystem be changed to achieve diagnostic excellence? In this month’s issue of Journal of Hospital Medicine, Singer and colleagues describe a pilot project of a proposed solution to the problem.5 Aptly named, the Socrates Project is an intervention that makes available a team of “diagnosticians” that can be consulted for assistance with challenging diagnostic cases. The physicians on the team volunteer their time, allowing for deep diagnostic evaluation that is not limited by one’s daily workload, thus overcoming one of the major hurdles to achieving diagnostic excellence. The described program also focuses on harnessing the power of teamwork, which is especially relevant given recent descriptions of the effectiveness of collective intelligence in improving diagnostic performance.6 Importantly, the authors recognize that their intervention will not achieve a diagnosis in every case for which they are consulted; rather, they hope that their thorough evaluation will uncover additional potential diagnostic avenues for the referring team to pursue, with a goal to “improve patient care by providing…ideas to reduce—or at least manage—diagnostic uncertainty.”
Programs of this nature are exciting for hospitalists. Hospital medicine is, perhaps, a place in modern medicine where diagnostic excellence has a natural home. Patients admitted to the hospital are acutely (and often severely) ill, and hospitalists are tasked with rapidly identifying the cause of their illness in order to initiate appropriate treatment and accurately inform prognosis. Hospitalists, as generalists, take a broad approach to challenging cases, and they tend to practice in well-resourced environments with nearly every diagnostic modality at their disposal. Many hospitalists would envy participating in a program such as the Socrates Project.
While Singer et al.’s innovation—and the institutional support thereof—should be lauded, some discussion must be had about how to assess the effectiveness of such a program. The authors acknowledge the need for evaluation of both the diagnostic process and the outcomes that process achieves. Measuring diagnostic performance is challenging, however, and while there is substantial progress being made in this area, recent efforts tend to focus on identifying diagnostic errors rather than measuring diagnostic excellence. Moreover, even if a program does improve diagnostic performance, how should we evaluate for unintended consequences of its implementation? In the age of high-value care, how can we ensure that efforts to do a better job of spotting proverbial zebras do not come at the cost of harming too many horses?7
Hospitalists are well primed to answer this question. The juxtaposition of Singer et al.’s article with the Journal of Hospital Medicine’s long-running series on Choosing Wisely®: Things We Do for No Reason™ provides a natural synergy to begin crafting a framework to evaluate unintended consequences of a program in diagnostic excellence. More diagnosis is not the goal; more appropriate diagnosis is what is needed. A clinical program aimed at achieving diagnostic excellence should not employ low-value, wasteful strategies that do not add substantively to the diagnostic process but should instead seek to improve the overall efficiency of even complicated diagnostic odysseys. Avoiding waste throughout will allow for allocation of diagnostic resources where they are needed. In turn, hospitalists can do a better job of correctly identifying both horses and zebras for what they are. While a given hospitalization for a diagnostically complex patient may be relatively expensive, better diagnosis during an index hospitalization is likely to lead to decreased downstream costs, such as those related to readmissions and further testing, as well as better health outcomes.
The Socrates Project, along with similar programs at other institutions, are exciting innovations. These programs are not only likely to be good for patients but are also good for hospitalists. The field of hospital medicine should leverage its collective expertise in clinical medicine, systems of care, and high-value care to become a home for diagnostic excellence.
1. National Academies of Sciences, Engineering, and Medicine. Improving Diagnosis in Health Care. Washington, DC: The National Academies Press; 2015. https://doi.org/10.17226/21794
2. Olson A, Rencic J, Cosby K, et al. Competencies for improving diagnosis: an interprofessional framework for education and training in health care. Diagnosis. 2019;6(4):335-341. https://doi.org/10.1515/dx-2018-0107.
3. Baduashvili A, Guyatt G, Evans AT. ROC anatomy—getting the most out of your diagnostic test. J Gen Intern Med. 2019;34(9):1892-1898. https://doi.org/10.1007/s11606-019-05125-0.
4. Manrai AK, Bhatia G, Strymish J, Kohane IS, Jain SH. Medicine’s uncomfortable relationship with math: calculating positive predictive value. JAMA Intern Med. 2014;174(6):991-993. https://doi.org/10.1001/jamainternmed.2014.1059.
5. Singer BD, Goodwin AM, Patel AA, Vaughan DE. The Socrates Project for difficult diagnosis at Northwestern Medicine. J Hosp Med. 2020;15(2):116-118. https://doi.org/ 10.12788/jhm.3335.
6. Barnett ML, Boddupalli D, Nundy S, Bates DW. Comparative accuracy of diagnosis by collective intelligence of multiple physicians vs individual physicians. JAMA Netw Open. 2019;2(3):e190096. https://doi.org/10.1001/jamanetworkopen.2019.0096.
7. Dhaliwal G. Bringing high-value care to the inpatient teaching service. JAMA Intern Med. 2014;174(7):1021-1022. https://doi.org/10.1001/jamainternmed.2014.2012.
1. National Academies of Sciences, Engineering, and Medicine. Improving Diagnosis in Health Care. Washington, DC: The National Academies Press; 2015. https://doi.org/10.17226/21794
2. Olson A, Rencic J, Cosby K, et al. Competencies for improving diagnosis: an interprofessional framework for education and training in health care. Diagnosis. 2019;6(4):335-341. https://doi.org/10.1515/dx-2018-0107.
3. Baduashvili A, Guyatt G, Evans AT. ROC anatomy—getting the most out of your diagnostic test. J Gen Intern Med. 2019;34(9):1892-1898. https://doi.org/10.1007/s11606-019-05125-0.
4. Manrai AK, Bhatia G, Strymish J, Kohane IS, Jain SH. Medicine’s uncomfortable relationship with math: calculating positive predictive value. JAMA Intern Med. 2014;174(6):991-993. https://doi.org/10.1001/jamainternmed.2014.1059.
5. Singer BD, Goodwin AM, Patel AA, Vaughan DE. The Socrates Project for difficult diagnosis at Northwestern Medicine. J Hosp Med. 2020;15(2):116-118. https://doi.org/ 10.12788/jhm.3335.
6. Barnett ML, Boddupalli D, Nundy S, Bates DW. Comparative accuracy of diagnosis by collective intelligence of multiple physicians vs individual physicians. JAMA Netw Open. 2019;2(3):e190096. https://doi.org/10.1001/jamanetworkopen.2019.0096.
7. Dhaliwal G. Bringing high-value care to the inpatient teaching service. JAMA Intern Med. 2014;174(7):1021-1022. https://doi.org/10.1001/jamainternmed.2014.2012.
© 2020 Society of Hospital Medicine
Leadership & Professional Development: A Letter to the Future Teaching Physician
“No one cares how much you know, until they know how much you care.”
—Theodore Roosevelt (attributed)
Like many early career clinician-educators, you are likely embarking on your teaching role with excitement and trepidation. Excitement accompanies the opportunity to develop the next generation of physicians. Trepidation arises from a fear of insufficient knowledge. This concern is understandable but misplaced: great teachers are great because of their emotional intelligence, not their medical intelligence. These five principles will help you establish an optimal learning environment.
Small-Talk before Med-Talk. “What do you like to do outside of the hospital?” “Where is your favorite place to eat?” These questions indicate that your interest in learners transcends clinical work. Leaders who are more relationship- than task-oriented achieve greater group cohesion and more team learning. Exemplary inpatient attending physicians use learners’ first names and get to know them on a personal level to signal that they care as much about the person as they do about the performance.1
Be Available. Medical educators balance supervision and autonomy while trainees engage in high-stakes decisions. The best teachers get this right by signaling “I have faith in you” and “I’m always available.” Clinician-educator Kimberly Manning, MD portrayed this balance in a recent Twitter thread. The resident called: “I am sorry to bother you.” Dr. Manning responded, “Never be sorry.” The resident was concerned about a patient with new abdominal pain but reassured Dr. Manning that she did not need to return to the hospital. She returned anyway. She assessed the patient and had nothing to add to the resident’s outstanding management. As the patient recovered from his operation for a perforated ulcer, Dr. Manning reflected, “On a perfect Saturday afternoon, I chose to return to the hospital. To make not one decision or write one single order. But instead to stand beside my resident and intentionally affirm her.”
Build from the Ground Up. Asking questions is the teacher’s core procedure. Strive to master the true Socratic method of starting with an elemental inquiry and then leading a conversation that poses questions of increasing difficulty until you reach the limits of the learner’s understanding. This method reinforces their hard-earned knowledge and sets the stage for growth. “What would be your first test to evaluate tachycardia?” Once the correct answer is firmly in hand, explore the margin of their knowledge. “Which regular, narrow complex tachycardias stop with adenosine?”
Never Judge. Never endorse an incorrect response—but do not disparage it either. A trainee must learn that their answer was wrong but should not feel defeated or embarrassed. Use judgment regarding whether constructive feedback should be delivered in public or in private.
I recall answering a question incorrectly in medical school. The attending responded, “How many years did you take off before starting third year?” I had not taken any time off. The attending was a phenomenal clinician but a lousy teacher. A master teacher would have accessed a foothold and built my knowledge without judgment.
Remain Humble. One of the most liberating phrases you will deploy as a teacher is “I don’t know.” Its utterance demonstrates the honesty and humility you hope to instill in learners. Be on the lookout for the many times your trainees will know more than you.
Recently my team evaluated a patient with blunted facial expression, bradykinesia, and a resting hand tremor. I disclosed to my team: “I don’t know the key maneuvers to distinguish the Parkinson plus syndromes from Parkinson disease.” The medical student had spent one year studying patients with neurodegenerative diseases (I learned this during the “small-talk before med-talk” phase). I invited him to demonstrate the neurologic exam, which he did admirably. That day I did not know the subject well, and we all learned because I freely admitted it.
Being a physician is the greatest job in the world. If you leverage your EQ (emotional quotient) as much as your IQ (intelligence quotient), your learners will conclude the same.
1. Houchens N, Harrod M, Moody S, Fowler KE, Saint S. Techniques and behaviors associated with exemplary inpatient general medicine teaching: an exploratory qualitative study. J Hosp Med. 2017;12(7):503-509. https://doi.org/10.12788/jhm.2763.
“No one cares how much you know, until they know how much you care.”
—Theodore Roosevelt (attributed)
Like many early career clinician-educators, you are likely embarking on your teaching role with excitement and trepidation. Excitement accompanies the opportunity to develop the next generation of physicians. Trepidation arises from a fear of insufficient knowledge. This concern is understandable but misplaced: great teachers are great because of their emotional intelligence, not their medical intelligence. These five principles will help you establish an optimal learning environment.
Small-Talk before Med-Talk. “What do you like to do outside of the hospital?” “Where is your favorite place to eat?” These questions indicate that your interest in learners transcends clinical work. Leaders who are more relationship- than task-oriented achieve greater group cohesion and more team learning. Exemplary inpatient attending physicians use learners’ first names and get to know them on a personal level to signal that they care as much about the person as they do about the performance.1
Be Available. Medical educators balance supervision and autonomy while trainees engage in high-stakes decisions. The best teachers get this right by signaling “I have faith in you” and “I’m always available.” Clinician-educator Kimberly Manning, MD portrayed this balance in a recent Twitter thread. The resident called: “I am sorry to bother you.” Dr. Manning responded, “Never be sorry.” The resident was concerned about a patient with new abdominal pain but reassured Dr. Manning that she did not need to return to the hospital. She returned anyway. She assessed the patient and had nothing to add to the resident’s outstanding management. As the patient recovered from his operation for a perforated ulcer, Dr. Manning reflected, “On a perfect Saturday afternoon, I chose to return to the hospital. To make not one decision or write one single order. But instead to stand beside my resident and intentionally affirm her.”
Build from the Ground Up. Asking questions is the teacher’s core procedure. Strive to master the true Socratic method of starting with an elemental inquiry and then leading a conversation that poses questions of increasing difficulty until you reach the limits of the learner’s understanding. This method reinforces their hard-earned knowledge and sets the stage for growth. “What would be your first test to evaluate tachycardia?” Once the correct answer is firmly in hand, explore the margin of their knowledge. “Which regular, narrow complex tachycardias stop with adenosine?”
Never Judge. Never endorse an incorrect response—but do not disparage it either. A trainee must learn that their answer was wrong but should not feel defeated or embarrassed. Use judgment regarding whether constructive feedback should be delivered in public or in private.
I recall answering a question incorrectly in medical school. The attending responded, “How many years did you take off before starting third year?” I had not taken any time off. The attending was a phenomenal clinician but a lousy teacher. A master teacher would have accessed a foothold and built my knowledge without judgment.
Remain Humble. One of the most liberating phrases you will deploy as a teacher is “I don’t know.” Its utterance demonstrates the honesty and humility you hope to instill in learners. Be on the lookout for the many times your trainees will know more than you.
Recently my team evaluated a patient with blunted facial expression, bradykinesia, and a resting hand tremor. I disclosed to my team: “I don’t know the key maneuvers to distinguish the Parkinson plus syndromes from Parkinson disease.” The medical student had spent one year studying patients with neurodegenerative diseases (I learned this during the “small-talk before med-talk” phase). I invited him to demonstrate the neurologic exam, which he did admirably. That day I did not know the subject well, and we all learned because I freely admitted it.
Being a physician is the greatest job in the world. If you leverage your EQ (emotional quotient) as much as your IQ (intelligence quotient), your learners will conclude the same.
“No one cares how much you know, until they know how much you care.”
—Theodore Roosevelt (attributed)
Like many early career clinician-educators, you are likely embarking on your teaching role with excitement and trepidation. Excitement accompanies the opportunity to develop the next generation of physicians. Trepidation arises from a fear of insufficient knowledge. This concern is understandable but misplaced: great teachers are great because of their emotional intelligence, not their medical intelligence. These five principles will help you establish an optimal learning environment.
Small-Talk before Med-Talk. “What do you like to do outside of the hospital?” “Where is your favorite place to eat?” These questions indicate that your interest in learners transcends clinical work. Leaders who are more relationship- than task-oriented achieve greater group cohesion and more team learning. Exemplary inpatient attending physicians use learners’ first names and get to know them on a personal level to signal that they care as much about the person as they do about the performance.1
Be Available. Medical educators balance supervision and autonomy while trainees engage in high-stakes decisions. The best teachers get this right by signaling “I have faith in you” and “I’m always available.” Clinician-educator Kimberly Manning, MD portrayed this balance in a recent Twitter thread. The resident called: “I am sorry to bother you.” Dr. Manning responded, “Never be sorry.” The resident was concerned about a patient with new abdominal pain but reassured Dr. Manning that she did not need to return to the hospital. She returned anyway. She assessed the patient and had nothing to add to the resident’s outstanding management. As the patient recovered from his operation for a perforated ulcer, Dr. Manning reflected, “On a perfect Saturday afternoon, I chose to return to the hospital. To make not one decision or write one single order. But instead to stand beside my resident and intentionally affirm her.”
Build from the Ground Up. Asking questions is the teacher’s core procedure. Strive to master the true Socratic method of starting with an elemental inquiry and then leading a conversation that poses questions of increasing difficulty until you reach the limits of the learner’s understanding. This method reinforces their hard-earned knowledge and sets the stage for growth. “What would be your first test to evaluate tachycardia?” Once the correct answer is firmly in hand, explore the margin of their knowledge. “Which regular, narrow complex tachycardias stop with adenosine?”
Never Judge. Never endorse an incorrect response—but do not disparage it either. A trainee must learn that their answer was wrong but should not feel defeated or embarrassed. Use judgment regarding whether constructive feedback should be delivered in public or in private.
I recall answering a question incorrectly in medical school. The attending responded, “How many years did you take off before starting third year?” I had not taken any time off. The attending was a phenomenal clinician but a lousy teacher. A master teacher would have accessed a foothold and built my knowledge without judgment.
Remain Humble. One of the most liberating phrases you will deploy as a teacher is “I don’t know.” Its utterance demonstrates the honesty and humility you hope to instill in learners. Be on the lookout for the many times your trainees will know more than you.
Recently my team evaluated a patient with blunted facial expression, bradykinesia, and a resting hand tremor. I disclosed to my team: “I don’t know the key maneuvers to distinguish the Parkinson plus syndromes from Parkinson disease.” The medical student had spent one year studying patients with neurodegenerative diseases (I learned this during the “small-talk before med-talk” phase). I invited him to demonstrate the neurologic exam, which he did admirably. That day I did not know the subject well, and we all learned because I freely admitted it.
Being a physician is the greatest job in the world. If you leverage your EQ (emotional quotient) as much as your IQ (intelligence quotient), your learners will conclude the same.
1. Houchens N, Harrod M, Moody S, Fowler KE, Saint S. Techniques and behaviors associated with exemplary inpatient general medicine teaching: an exploratory qualitative study. J Hosp Med. 2017;12(7):503-509. https://doi.org/10.12788/jhm.2763.
1. Houchens N, Harrod M, Moody S, Fowler KE, Saint S. Techniques and behaviors associated with exemplary inpatient general medicine teaching: an exploratory qualitative study. J Hosp Med. 2017;12(7):503-509. https://doi.org/10.12788/jhm.2763.
© 2020 Society of Hospital Medicine
Decreasing Hypoglycemia following Insulin Administration for Inpatient Hyperkalemia
Hyperkalemia (serum potassium ≥5.1 mEq/L), if left untreated, may result in cardiac arrhythmias, severe muscle weakness, or paralysis.1,2 Insulin administration can rapidly correct hyperkalemia by shifting serum potassiufm intracellularly.3 Treatment of hyperkalemia with insulin may lead to hypoglycemia, which, when severe, can cause confusion, seizures, loss of consciousness, and death. The use of regular and short-acting insulins to correct hyperkalemia quickly in hospitalized patients results in the greatest risk of hypoglycemia within three hours of treatment.4 Nonetheless, monitoring blood glucose levels within six hours of postinsulin administration is not a standard part of hyperkalemia treatment guidelines,3 leaving the rates of hypoglycemia in this setting poorly characterized.
Without standardized blood glucose measurement protocols, retrospective studies have reported posttreatment hypoglycemia rates of 8.7%-17.5% among all patients with hyperkalemia,5,6 and 13% among patients with end-stage renal disease.4 These estimates likely underestimate the true hypoglycemia rates as they measure blood glucose sporadically and are often outside the three-hour window of highest risk after insulin administration.
At the University of California, San Francisco Medical Center (UCSFMC), we faced similar issues in measuring the true hypoglycemia rates associated with hyperkalemia treatment. In December 2015, a 12-month retrospective review revealed a 12% hypoglycemia rate among patients treated with insulin for hyperkalemia. This review was limited by the inclusion of only patients treated for hyperkalemia using the standard orderset supplied with the electronic health record system (EHR; EPIC Systems, Verona, Wisconsin) and the absence of specific orders for glucose monitoring. As a result, more than 40% of these inpatients had no documented glucose within six hours of postinsulin administration.
We subsequently designed and implemented an adult inpatient hyperkalemia treatment orderset aimed at reducing iatrogenic hypoglycemia by promoting appropriate insulin use and blood glucose monitoring during the treatment of hyperkalemia. Through rapid improvement cycles, we iteratively revised the orderset to optimally mitigate the risk of hypoglycemia that was associated with insulin use. We describe implementation and outcomes of weight-based insulin dosing,7 automated alerts to identify patients at greatest risk for hypoglycemia, and clinical decision support based on the preinsulin blood glucose level. We report the rates of iatrogenic hypoglycemia after the implementation of these order-set changes.
METHODS
Design Overview
EHR data were extracted from Epic Clarity. We analyzed data following Orderset 1.1 implementation (January 1, 2016-March 19, 2017) when hypoglycemia rates were reliably quantifiable and following orderset revision 1.2 (March 20, 2017-September 30, 2017) to evaluate the impact of the orderset intervention. The data collection was approved by the Institutional Review Board at the University of California, San Francisco.
Additionally, we explored the frequency in which providers ordered insulin through the hyperkalemia orderset for each version of the orderset via two-month baseline reviews. Investigation for Orderset 1.1 was from January 1, 2017 to February 28, 2017 and for Orderset 1.2 was from August 1, 2017 to September 30, 2017. Insulin ordering frequency through the hyperkalemia orderset was defined as ordering insulin through the adult inpatient hyperkalemia orderset versus ordering insulin in and outside of the hyperkalemia orderset.
Last, we measured the nursing point of care testing (POCT) blood glucose measurement compliance with the hyperkalemia orderset. Nursing utilization acceptance of the hyperkalemia orderset was defined as adequate POCT blood glucose levels monitored in comparison to all insulin treatments via the hyperkalemia orderset.
Setting and Participants
We evaluated nonobstetric adult inpatients admitted to UCSF Medical Center between January 2016 and September 2017. All medical and surgical wards and intensive care units were included in the analysis.
Intervention
In June 2012, an EHR developed by Epic Systems was introduced at UCSFMC. In January 2016, we designed a new EHR-based hyperkalemia treatment orderset (Orderset 1.1), which added standard POCT blood glucose checks before and at one, two, four, and six hours after insulin injection (Appendix 1). In March 2017, a newly designed orderset (Orderset 1.2) replaced the previous hyperkalemia treatment orderset (Appendix 2). Orderset 1.2 included three updates. First, providers were now presented the option of ordering insulin as a
CORRECTED FIGURE PER ERRATUM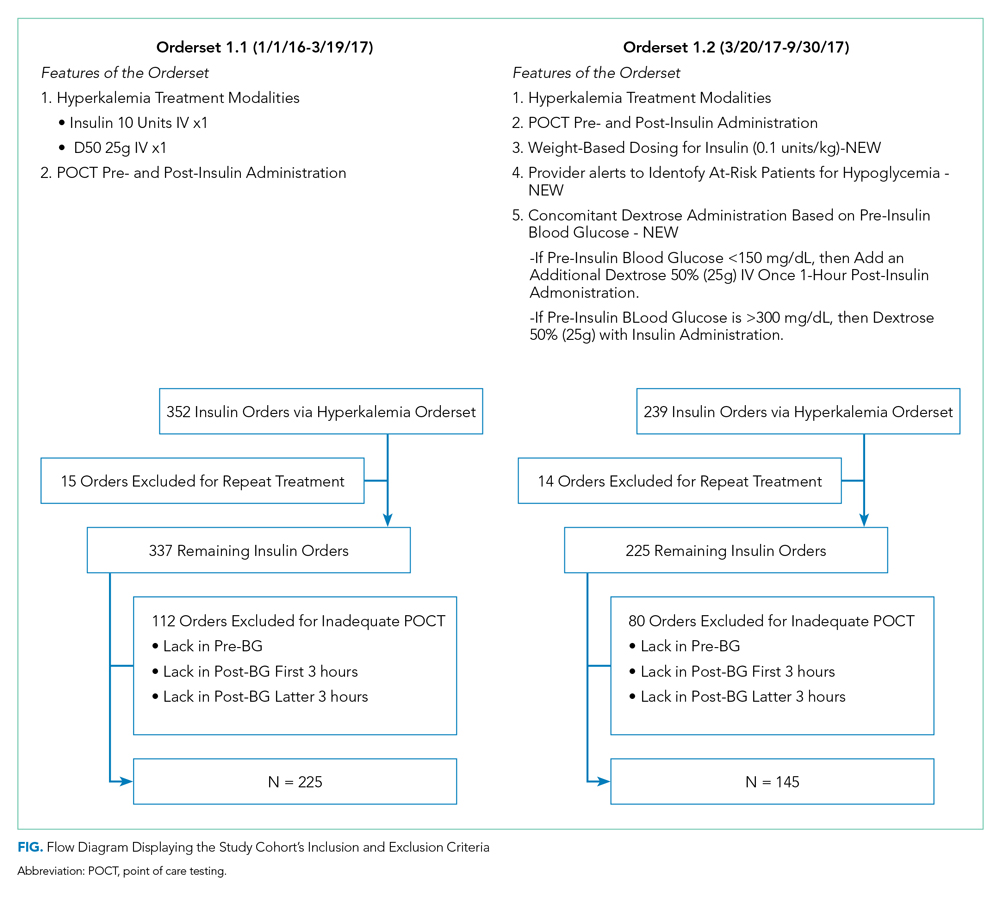
Inclusion and exclusion criteria are shown in the Figure. All patients who had insulin ordered via a hyperkalemia orderset were included in an intention-to-treat analysis. A further analysis was performed for patients for whom orderset compliance was achieved (ie, insulin ordered through the ordersets with adequate blood glucose monitoring). These patients were required to have a POCT blood glucose check preinsulin administration and postinsulin administration as follows: (1) between 30 to 180 minutes (0.5 to three hours) after insulin administration, and (2) between 180 and 360 minutes (three to six hours) after insulin administration. For patients receiving repeated insulin treatments for hyperkalemia within six hours, the first treatment data points were excluded to prevent duplication.
Outcomes
We extracted data on all nonobstetric adult patients admitted to UCSFMC between January 1, 2016 and March 19, 2017 (Orderset 1.1) and between March 20, 2017 and September 30, 2017 (Orderset 1.2).
We measured unique insulin administrations given that each insulin injection poses a risk of iatrogenic hypoglycemia. Hypoglycemia was defined as glucose <70 mg/dL and severe hypoglycemia was defined as glucose <40 mg/dL. Covariates included time and date of insulin administration; blood glucose levels before and at one, two, four, and six hours after insulin injection (if available); sex; weight; dose of insulin given for hyperkalemia treatment; creatinine; known diagnosis of diabetes; concomitant use of albuterol; and concomitant use of corticosteroids. Hyperglycemia was defined as glucose >180 mg/dL. We collected potassium levels pre- and postinsulin treatment. The responsible team’s discipline and the location of the patient (eg, medical/surgical unit, intensive care unit, emergency department) where the insulin orderset was used were recorded.
Statistical Analysis
Statistical analysis for our data included the χ2 test for categorical data and Student t test for continuous data. The bivariate analysis identified potential risk factors and protective factors for hypoglycemia, and logistic regression was used to determine independent predictors of hypoglycemia. Through bivariate analyses, any factor with a P value below .05 was included in the multivariable analyses to investigate a significant contribution to hypoglycemia outcomes. Analyses for hypoglycemia and severe hypoglycemia rates, potassium levels pre- and postinsulin treatment, and hyperglycemia rates were done for both the intention-to-treat group and the group with all criteria met. All analyses were rendered utilizing Stata version 14 (Stata Corp LLC, College Station, Texas).
RESULTS
Baseline patient characteristics, initial insulin dosing, the treatment team, and the location are shown in Table 1. With the implementation of weight-based dosing, a lower dose of insulin was administered with Orderset 1.2 compared with Orderset 1.1.
Orderset adherence rates for Orderset 1.1 and 1.2 were as follows: Acute Care Floor 65% (70%), Intensive Care Unit 63% (66%), and Emergency Department 60% (55%). A two-month audit of orderset usage and compliance revealed that 73% (70 of 96) of insulin treatments were ordered through Orderset 1.1, and 77% (71 of 92) of insulin treatments were ordered through Orderset 1.2. The distribution of orderset usage across location and primary service are shown in Table 1.
The patient distribution is shown in the Figure. In the Orderset 1.1 period, there were 352 total insulin treatments utilizing the newly revised UCSFMC adult inpatient hyperkalemia orderset that were used for the intention-to-treat analysis, and there were 225 patients for whom compliance with orderset monitoring was achieved. Notably, 112 treatments were excluded for the lack of adequate blood glucose monitoring. In the Orderset 1.2 period, there were 239 total insulin treatments utilizing the newly revised UCSFMC adult inpatient hyperkalemia orderset that were used for the intention-to-treat analysis, and there were 145 patients for whom compliance with orderset monitoring was achieved. During this phase, 80 treatments were excluded for inadequate blood glucose monitoring.
Predictors of hypoglycemia following the implementation of Orderset 1.1 are shown in Table 2, and the logistic regression model of these risks is shown in Appendix Table 1. Female gender, weight-based dosing of insulin exceeding 0.14 units/kg, preinsulin blood glucose less than 140 mg/dL, and serum creatinine greater than 2.5 mg/dl were associated with an increased risk of hypoglycemia. A known diagnosis of Type 2 diabetes, concomitant albuterol within one hour of insulin administration, and corticosteroid administration within two hours of insulin administration were associated with a decreased risk of hypoglycemia.
The rates of hypoglycemia (<70 mg/dl) and severe hypoglycemia (<40 mg/dl) are shown in Table 3. During the Orderset 1.1 period,
During the Orderset 1.2 period, for patients with all criteria met, 14 of 145 (10%) had hypoglycemia, and three of 145 (2%) had severe hypoglycemia. Ten of 14 (72%) of these hypoglycemic events occurred in the first three hours, with the remaining four hypoglycemic events (28%) occurring in the last three hours.
An intention-to-treat analysis for hyperglycemia, defined as glucose >180 mg/dl, revealed that during the Orderset 1.1 period, 80 of 352 (23%) had hyperglycemia before insulin administration, and 38 of 352 (11%) had hyperglycemia after insulin administration. During the Orderset 1.2 period, 52 of 239 (22%) had hyperglycemia before insulin administration, and 15 of 239 (6%) had hyperglycemia after insulin administration. Results can be found in Table 3.
Pre- and posttreatment potassium levels are shown in Table 3. An intention-to-treat analysis for potassium reduction postinsulin administration revealed that during the Orderset 1.1 period, there was an absolute reduction of 0.73 mmol/L, while during the Orderset 1.2 period, there was an absolute reduction of 0.95 mmol/L.
DISCUSSION
Treatment of hyperkalemia with insulin may result in significant iatrogenic hypoglycemia. Prior studies have likely underestimated the incidence of hyperkalemia treatment-associated hypoglycemia as glucose levels are rarely checked within three hours of insulin administration.8 In our study, which was designed to ensure appropriate blood glucose measurement, 21% of insulin treatments for hyperkalemia resulted in hypoglycemia, with 92% of hypoglycemic events occurring within the first three hours.
For the Orderset 1.1 period, patient risk factors identified for iatrogenic hypoglycemia postinsulin administration were female sex, doses of regular insulin greater than 0.14 units/kg, preinsulin blood glucose less than 140 mg/dL, and serum creatinine greater than 2.5 mg/dL. These results are consistent with studies suggesting that preinsulin blood glucose levels less than 140 mg/dL and the standard 10 units of insulin for hyperkalemia treatment may increase the risk of hypoglycemia.4,7,9
To decrease the risk of iatrogenic hypoglycemia, we redesigned our hyperkalemia insulin orderset to address the strongest predictors of hypoglycemia (doses of regular insulin greater than 0.14 units/kg and preinsulin blood glucose less than 140 mg/dL). The main changes were weight-based insulin dosing (based on previously published data)10 and adjustment of glucose administration based on the patient’s glucose levels.11 Following these changes, the rates of both hypoglycemia and severe hypoglycemia were statistically significantly reduced. In addition, of the 14 hypoglycemia events identified after the introduction of Orderset 1.2, five could have been prevented (36%) had the protocol been strictly followed. These five hypoglycemia events occurred later than one-hour postinsulin administration in patients with blood sugars < 150 mg/dL prior to insulin administration. In each of these cases, Orderset 1.2 called for an additional dextrose 50% (50 mL) IV bolus, which likely would have prevented the subsequently recorded hypoglycemia. In other words, our orderset indicated that these patients received an additional bolus of dextrose. However, they did not receive their glucose at the appropriate time, contributing to the hypoglycemia events. The orderset did not include a best practice alert (BPA) to remind providers about the extra dextrose bolus. In the future, we plan to add this BPA.
The hypoglycemia rate identified by Orderset 1.1 was 21% and the hypoglycemia rate identified by the Orderset 1.2 was 10%. The severe hypoglycemia rate identified by Orderset 1.1 was 5% and the severe hypoglycemia rate identified by Orderset 1.2 was 2%. The hypoglycemia and severe hypoglycemia rates significantly decreased after the introduction of Orderset 1.2. To mimic a real-world clinical setting, where monitoring of blood glucose is not always achieved multiple times within a six-hour timeframe of postinsulin treatment for hyperkalemia, we conducted an intention-to-treat analysis. Even when including patients for whom full blood glucose monitoring was not achieved, the introduction of Orderset 1.2 was associated with a significant decrease in the hypoglycemia rate.
To demonstrate whether weight-based dosing of insulin was as effective as a standard dose for hyperkalemia treatment, we compared the impact of Orderset 1.1, which only had the option for single standard doses of insulin, with the impact of Orderset 1.2, which included weight-based dosing options. With the introduction of Orderset 1.2, there was a significant decrease in serum potassium, indicating that weight-based dosing options may not only prevent hypoglycemia but may potentially provide more effective hyperkalemia treatment.
We also compared the rate of hyperglycemia (a glucose >180 mg/dl) pre- and posttreatment (Table 3). Although not statistically significant, the rate of hyperglycemia decreased from 11% to 6%, suggesting a trend toward decreased hyperglycemia with orderset usage.
As orderset usage for hyperkalemia management only occurred approximately 75% of the time, likely, forcing the use of these ordersets would further reduce the incidence of treatment-associated hypoglycemia. To encourage the use of ordersets for hyperkalemia management, our medical center has largely restricted insulin ordering so that it can only be done through ordersets with the proper precautions in place, regardless of the indication. Furthermore, adherence to all the blood glucose monitoring orders embedded in the ordersets remained suboptimal irrespective of managing the service or clinical setting
Finally, development and implementation of these hyperkalemia treatment ordersets required an experienced multidisciplinary team, including pharmacists, nurses, hospitalists, endocrinologists, and EHR system programmers.12,13 We, therefore, encourage interprofessional collaboration for any institutions seeking to establish innovative clinical protocols.
This analysis was limited to the insulin administration meeting our inclusion criteria. Thus, we could not identify a true hypoglycemia rate for treatments that were not followed by adequate blood glucose monitoring postinsulin administration, or for insulin administration ordered outside of the hyperkalemia ordersets.
CONCLUSION
The use of a comprehensive EHR orderset for the treatment of hyperkalemia with predefined times for blood glucose monitoring, weight-based insulin dosing, and prompts to warn providers of an individual patient’s risk for hypoglycemia may significantly reduce the incidence of iatrogenic hypoglycemia.
1. Acker CG, Johnson JP, Palevsky PM, Greenberg A. Hyperkalemia in hospitalized patients: causes, adequacy of treatment, and results of an attempt to improve physician compliance with published therapy guidelines. Arch Intern Med. 1998;158(8):917-924. https://doi.org/10.1001/archinte.158.8.917.
2. Fordjour KN, Walton T, Doran JJ. Management of hyperkalemia in hospitalized patients. Am J Med Sci. 2014;347(2):93-100. https://doi.org/10.1097/MAJ.0b013e318279b105.
3. Part-10-Special-Circumstances-of-Resuscitation.pdf. https://eccguidelines.heart.org/wp-content/themes/eccstaging/dompdf-master/pdffiles/part-10-special-circumstances-of-resuscitation.pdf. Accessed December 16, 2017.
4. Apel J, Reutrakul S, Baldwin D. Hypoglycemia in the treatment of hyperkalemia with insulin in patients with end-stage renal disease. Clin Kidney J. 2014;7(3):248-250. https://doi.org/10.1093/ckj/sfu026.
5. Schafers S, Naunheim R, Vijayan A, Tobin G. Incidence of hypoglycemia following insulin-based acute stabilization of hyperkalemia treatment. J Hosp Med. 2012;7(3):239-242. https://doi.org/10.1002/jhm.977.
6. Boughton CK, Dixon D, Goble E, et al. Preventing hypoglycemia following treatment of hyperkalemia in hospitalized patients. J Hosp Med. 2019;14:E1-E4. https://doi.org/10.12788/jhm.3145.
7. Wheeler DT, Schafers SJ, Horwedel TA, Deal EN, Tobin GS. Weight-based insulin dosing for acute hyperkalemia results in less hypoglycemia. J Hosp Med. 2016;11(5):355-357. https://doi.org/10.1002/jhm.2545.
8. Coca A, Valencia AL, Bustamante J, Mendiluce A, Floege J. Hypoglycemia following intravenous insulin plus glucose for hyperkalemia in patients with impaired renal function. PLoS ONE. 2017;12(2):e0172961. https://doi.org/10.1371/journal.pone.0172961.
9. LaRue HA, Peksa GD, Shah SC. A comparison of insulin doses for the treatment of hyperkalemia in patients with renal insufficiency. Pharmacotherapy. 2017;37(12):1516-1522. https://doi.org/10.1002/phar.2038.
10. Brown K, Setji TL, Hale SL, et al. Assessing the impact of an order panel utilizing weight-based insulin and standardized monitoring of blood glucose for patients with hyperkalemia. Am J Med Qual. 2018;33(6):598-603. https://doi.org/10.1177/1062860618764610.
11. Farina N, Anderson C. Impact of dextrose dose on hypoglycemia development following treatment of hyperkalemia. Ther Adv Drug Saf. 2018;9(6):323-329. https://doi.org/10.1177/2042098618768725.
12. Neinstein A, MacMaster HW, Sullivan MM, Rushakoff R. A detailed description of the implementation of inpatient insulin orders with a commercial electronic health record system. J Diabetes Sci Technol. 2014;8(4):641-651. https://doi.org/10.1177/1932296814536290.
13. MacMaster HW, Gonzalez S, Maruoka A, et al. Development and implementation of a subcutaneous Insulin pen label bar code scanning protocol to prevent wrong-patient insulin pen errors. Jt Comm J Qual Patient Saf. 2019;45(5):380-386. https://doi.org/10.1016/j.jcjq.2018.08.006.
Hyperkalemia (serum potassium ≥5.1 mEq/L), if left untreated, may result in cardiac arrhythmias, severe muscle weakness, or paralysis.1,2 Insulin administration can rapidly correct hyperkalemia by shifting serum potassiufm intracellularly.3 Treatment of hyperkalemia with insulin may lead to hypoglycemia, which, when severe, can cause confusion, seizures, loss of consciousness, and death. The use of regular and short-acting insulins to correct hyperkalemia quickly in hospitalized patients results in the greatest risk of hypoglycemia within three hours of treatment.4 Nonetheless, monitoring blood glucose levels within six hours of postinsulin administration is not a standard part of hyperkalemia treatment guidelines,3 leaving the rates of hypoglycemia in this setting poorly characterized.
Without standardized blood glucose measurement protocols, retrospective studies have reported posttreatment hypoglycemia rates of 8.7%-17.5% among all patients with hyperkalemia,5,6 and 13% among patients with end-stage renal disease.4 These estimates likely underestimate the true hypoglycemia rates as they measure blood glucose sporadically and are often outside the three-hour window of highest risk after insulin administration.
At the University of California, San Francisco Medical Center (UCSFMC), we faced similar issues in measuring the true hypoglycemia rates associated with hyperkalemia treatment. In December 2015, a 12-month retrospective review revealed a 12% hypoglycemia rate among patients treated with insulin for hyperkalemia. This review was limited by the inclusion of only patients treated for hyperkalemia using the standard orderset supplied with the electronic health record system (EHR; EPIC Systems, Verona, Wisconsin) and the absence of specific orders for glucose monitoring. As a result, more than 40% of these inpatients had no documented glucose within six hours of postinsulin administration.
We subsequently designed and implemented an adult inpatient hyperkalemia treatment orderset aimed at reducing iatrogenic hypoglycemia by promoting appropriate insulin use and blood glucose monitoring during the treatment of hyperkalemia. Through rapid improvement cycles, we iteratively revised the orderset to optimally mitigate the risk of hypoglycemia that was associated with insulin use. We describe implementation and outcomes of weight-based insulin dosing,7 automated alerts to identify patients at greatest risk for hypoglycemia, and clinical decision support based on the preinsulin blood glucose level. We report the rates of iatrogenic hypoglycemia after the implementation of these order-set changes.
METHODS
Design Overview
EHR data were extracted from Epic Clarity. We analyzed data following Orderset 1.1 implementation (January 1, 2016-March 19, 2017) when hypoglycemia rates were reliably quantifiable and following orderset revision 1.2 (March 20, 2017-September 30, 2017) to evaluate the impact of the orderset intervention. The data collection was approved by the Institutional Review Board at the University of California, San Francisco.
Additionally, we explored the frequency in which providers ordered insulin through the hyperkalemia orderset for each version of the orderset via two-month baseline reviews. Investigation for Orderset 1.1 was from January 1, 2017 to February 28, 2017 and for Orderset 1.2 was from August 1, 2017 to September 30, 2017. Insulin ordering frequency through the hyperkalemia orderset was defined as ordering insulin through the adult inpatient hyperkalemia orderset versus ordering insulin in and outside of the hyperkalemia orderset.
Last, we measured the nursing point of care testing (POCT) blood glucose measurement compliance with the hyperkalemia orderset. Nursing utilization acceptance of the hyperkalemia orderset was defined as adequate POCT blood glucose levels monitored in comparison to all insulin treatments via the hyperkalemia orderset.
Setting and Participants
We evaluated nonobstetric adult inpatients admitted to UCSF Medical Center between January 2016 and September 2017. All medical and surgical wards and intensive care units were included in the analysis.
Intervention
In June 2012, an EHR developed by Epic Systems was introduced at UCSFMC. In January 2016, we designed a new EHR-based hyperkalemia treatment orderset (Orderset 1.1), which added standard POCT blood glucose checks before and at one, two, four, and six hours after insulin injection (Appendix 1). In March 2017, a newly designed orderset (Orderset 1.2) replaced the previous hyperkalemia treatment orderset (Appendix 2). Orderset 1.2 included three updates. First, providers were now presented the option of ordering insulin as a
CORRECTED FIGURE PER ERRATUM
Inclusion and exclusion criteria are shown in the Figure. All patients who had insulin ordered via a hyperkalemia orderset were included in an intention-to-treat analysis. A further analysis was performed for patients for whom orderset compliance was achieved (ie, insulin ordered through the ordersets with adequate blood glucose monitoring). These patients were required to have a POCT blood glucose check preinsulin administration and postinsulin administration as follows: (1) between 30 to 180 minutes (0.5 to three hours) after insulin administration, and (2) between 180 and 360 minutes (three to six hours) after insulin administration. For patients receiving repeated insulin treatments for hyperkalemia within six hours, the first treatment data points were excluded to prevent duplication.
Outcomes
We extracted data on all nonobstetric adult patients admitted to UCSFMC between January 1, 2016 and March 19, 2017 (Orderset 1.1) and between March 20, 2017 and September 30, 2017 (Orderset 1.2).
We measured unique insulin administrations given that each insulin injection poses a risk of iatrogenic hypoglycemia. Hypoglycemia was defined as glucose <70 mg/dL and severe hypoglycemia was defined as glucose <40 mg/dL. Covariates included time and date of insulin administration; blood glucose levels before and at one, two, four, and six hours after insulin injection (if available); sex; weight; dose of insulin given for hyperkalemia treatment; creatinine; known diagnosis of diabetes; concomitant use of albuterol; and concomitant use of corticosteroids. Hyperglycemia was defined as glucose >180 mg/dL. We collected potassium levels pre- and postinsulin treatment. The responsible team’s discipline and the location of the patient (eg, medical/surgical unit, intensive care unit, emergency department) where the insulin orderset was used were recorded.
Statistical Analysis
Statistical analysis for our data included the χ2 test for categorical data and Student t test for continuous data. The bivariate analysis identified potential risk factors and protective factors for hypoglycemia, and logistic regression was used to determine independent predictors of hypoglycemia. Through bivariate analyses, any factor with a P value below .05 was included in the multivariable analyses to investigate a significant contribution to hypoglycemia outcomes. Analyses for hypoglycemia and severe hypoglycemia rates, potassium levels pre- and postinsulin treatment, and hyperglycemia rates were done for both the intention-to-treat group and the group with all criteria met. All analyses were rendered utilizing Stata version 14 (Stata Corp LLC, College Station, Texas).
RESULTS
Baseline patient characteristics, initial insulin dosing, the treatment team, and the location are shown in Table 1. With the implementation of weight-based dosing, a lower dose of insulin was administered with Orderset 1.2 compared with Orderset 1.1.
Orderset adherence rates for Orderset 1.1 and 1.2 were as follows: Acute Care Floor 65% (70%), Intensive Care Unit 63% (66%), and Emergency Department 60% (55%). A two-month audit of orderset usage and compliance revealed that 73% (70 of 96) of insulin treatments were ordered through Orderset 1.1, and 77% (71 of 92) of insulin treatments were ordered through Orderset 1.2. The distribution of orderset usage across location and primary service are shown in Table 1.
The patient distribution is shown in the Figure. In the Orderset 1.1 period, there were 352 total insulin treatments utilizing the newly revised UCSFMC adult inpatient hyperkalemia orderset that were used for the intention-to-treat analysis, and there were 225 patients for whom compliance with orderset monitoring was achieved. Notably, 112 treatments were excluded for the lack of adequate blood glucose monitoring. In the Orderset 1.2 period, there were 239 total insulin treatments utilizing the newly revised UCSFMC adult inpatient hyperkalemia orderset that were used for the intention-to-treat analysis, and there were 145 patients for whom compliance with orderset monitoring was achieved. During this phase, 80 treatments were excluded for inadequate blood glucose monitoring.
Predictors of hypoglycemia following the implementation of Orderset 1.1 are shown in Table 2, and the logistic regression model of these risks is shown in Appendix Table 1. Female gender, weight-based dosing of insulin exceeding 0.14 units/kg, preinsulin blood glucose less than 140 mg/dL, and serum creatinine greater than 2.5 mg/dl were associated with an increased risk of hypoglycemia. A known diagnosis of Type 2 diabetes, concomitant albuterol within one hour of insulin administration, and corticosteroid administration within two hours of insulin administration were associated with a decreased risk of hypoglycemia.
The rates of hypoglycemia (<70 mg/dl) and severe hypoglycemia (<40 mg/dl) are shown in Table 3. During the Orderset 1.1 period,
During the Orderset 1.2 period, for patients with all criteria met, 14 of 145 (10%) had hypoglycemia, and three of 145 (2%) had severe hypoglycemia. Ten of 14 (72%) of these hypoglycemic events occurred in the first three hours, with the remaining four hypoglycemic events (28%) occurring in the last three hours.
An intention-to-treat analysis for hyperglycemia, defined as glucose >180 mg/dl, revealed that during the Orderset 1.1 period, 80 of 352 (23%) had hyperglycemia before insulin administration, and 38 of 352 (11%) had hyperglycemia after insulin administration. During the Orderset 1.2 period, 52 of 239 (22%) had hyperglycemia before insulin administration, and 15 of 239 (6%) had hyperglycemia after insulin administration. Results can be found in Table 3.
Pre- and posttreatment potassium levels are shown in Table 3. An intention-to-treat analysis for potassium reduction postinsulin administration revealed that during the Orderset 1.1 period, there was an absolute reduction of 0.73 mmol/L, while during the Orderset 1.2 period, there was an absolute reduction of 0.95 mmol/L.
DISCUSSION
Treatment of hyperkalemia with insulin may result in significant iatrogenic hypoglycemia. Prior studies have likely underestimated the incidence of hyperkalemia treatment-associated hypoglycemia as glucose levels are rarely checked within three hours of insulin administration.8 In our study, which was designed to ensure appropriate blood glucose measurement, 21% of insulin treatments for hyperkalemia resulted in hypoglycemia, with 92% of hypoglycemic events occurring within the first three hours.
For the Orderset 1.1 period, patient risk factors identified for iatrogenic hypoglycemia postinsulin administration were female sex, doses of regular insulin greater than 0.14 units/kg, preinsulin blood glucose less than 140 mg/dL, and serum creatinine greater than 2.5 mg/dL. These results are consistent with studies suggesting that preinsulin blood glucose levels less than 140 mg/dL and the standard 10 units of insulin for hyperkalemia treatment may increase the risk of hypoglycemia.4,7,9
To decrease the risk of iatrogenic hypoglycemia, we redesigned our hyperkalemia insulin orderset to address the strongest predictors of hypoglycemia (doses of regular insulin greater than 0.14 units/kg and preinsulin blood glucose less than 140 mg/dL). The main changes were weight-based insulin dosing (based on previously published data)10 and adjustment of glucose administration based on the patient’s glucose levels.11 Following these changes, the rates of both hypoglycemia and severe hypoglycemia were statistically significantly reduced. In addition, of the 14 hypoglycemia events identified after the introduction of Orderset 1.2, five could have been prevented (36%) had the protocol been strictly followed. These five hypoglycemia events occurred later than one-hour postinsulin administration in patients with blood sugars < 150 mg/dL prior to insulin administration. In each of these cases, Orderset 1.2 called for an additional dextrose 50% (50 mL) IV bolus, which likely would have prevented the subsequently recorded hypoglycemia. In other words, our orderset indicated that these patients received an additional bolus of dextrose. However, they did not receive their glucose at the appropriate time, contributing to the hypoglycemia events. The orderset did not include a best practice alert (BPA) to remind providers about the extra dextrose bolus. In the future, we plan to add this BPA.
The hypoglycemia rate identified by Orderset 1.1 was 21% and the hypoglycemia rate identified by the Orderset 1.2 was 10%. The severe hypoglycemia rate identified by Orderset 1.1 was 5% and the severe hypoglycemia rate identified by Orderset 1.2 was 2%. The hypoglycemia and severe hypoglycemia rates significantly decreased after the introduction of Orderset 1.2. To mimic a real-world clinical setting, where monitoring of blood glucose is not always achieved multiple times within a six-hour timeframe of postinsulin treatment for hyperkalemia, we conducted an intention-to-treat analysis. Even when including patients for whom full blood glucose monitoring was not achieved, the introduction of Orderset 1.2 was associated with a significant decrease in the hypoglycemia rate.
To demonstrate whether weight-based dosing of insulin was as effective as a standard dose for hyperkalemia treatment, we compared the impact of Orderset 1.1, which only had the option for single standard doses of insulin, with the impact of Orderset 1.2, which included weight-based dosing options. With the introduction of Orderset 1.2, there was a significant decrease in serum potassium, indicating that weight-based dosing options may not only prevent hypoglycemia but may potentially provide more effective hyperkalemia treatment.
We also compared the rate of hyperglycemia (a glucose >180 mg/dl) pre- and posttreatment (Table 3). Although not statistically significant, the rate of hyperglycemia decreased from 11% to 6%, suggesting a trend toward decreased hyperglycemia with orderset usage.
As orderset usage for hyperkalemia management only occurred approximately 75% of the time, likely, forcing the use of these ordersets would further reduce the incidence of treatment-associated hypoglycemia. To encourage the use of ordersets for hyperkalemia management, our medical center has largely restricted insulin ordering so that it can only be done through ordersets with the proper precautions in place, regardless of the indication. Furthermore, adherence to all the blood glucose monitoring orders embedded in the ordersets remained suboptimal irrespective of managing the service or clinical setting
Finally, development and implementation of these hyperkalemia treatment ordersets required an experienced multidisciplinary team, including pharmacists, nurses, hospitalists, endocrinologists, and EHR system programmers.12,13 We, therefore, encourage interprofessional collaboration for any institutions seeking to establish innovative clinical protocols.
This analysis was limited to the insulin administration meeting our inclusion criteria. Thus, we could not identify a true hypoglycemia rate for treatments that were not followed by adequate blood glucose monitoring postinsulin administration, or for insulin administration ordered outside of the hyperkalemia ordersets.
CONCLUSION
The use of a comprehensive EHR orderset for the treatment of hyperkalemia with predefined times for blood glucose monitoring, weight-based insulin dosing, and prompts to warn providers of an individual patient’s risk for hypoglycemia may significantly reduce the incidence of iatrogenic hypoglycemia.
Hyperkalemia (serum potassium ≥5.1 mEq/L), if left untreated, may result in cardiac arrhythmias, severe muscle weakness, or paralysis.1,2 Insulin administration can rapidly correct hyperkalemia by shifting serum potassiufm intracellularly.3 Treatment of hyperkalemia with insulin may lead to hypoglycemia, which, when severe, can cause confusion, seizures, loss of consciousness, and death. The use of regular and short-acting insulins to correct hyperkalemia quickly in hospitalized patients results in the greatest risk of hypoglycemia within three hours of treatment.4 Nonetheless, monitoring blood glucose levels within six hours of postinsulin administration is not a standard part of hyperkalemia treatment guidelines,3 leaving the rates of hypoglycemia in this setting poorly characterized.
Without standardized blood glucose measurement protocols, retrospective studies have reported posttreatment hypoglycemia rates of 8.7%-17.5% among all patients with hyperkalemia,5,6 and 13% among patients with end-stage renal disease.4 These estimates likely underestimate the true hypoglycemia rates as they measure blood glucose sporadically and are often outside the three-hour window of highest risk after insulin administration.
At the University of California, San Francisco Medical Center (UCSFMC), we faced similar issues in measuring the true hypoglycemia rates associated with hyperkalemia treatment. In December 2015, a 12-month retrospective review revealed a 12% hypoglycemia rate among patients treated with insulin for hyperkalemia. This review was limited by the inclusion of only patients treated for hyperkalemia using the standard orderset supplied with the electronic health record system (EHR; EPIC Systems, Verona, Wisconsin) and the absence of specific orders for glucose monitoring. As a result, more than 40% of these inpatients had no documented glucose within six hours of postinsulin administration.
We subsequently designed and implemented an adult inpatient hyperkalemia treatment orderset aimed at reducing iatrogenic hypoglycemia by promoting appropriate insulin use and blood glucose monitoring during the treatment of hyperkalemia. Through rapid improvement cycles, we iteratively revised the orderset to optimally mitigate the risk of hypoglycemia that was associated with insulin use. We describe implementation and outcomes of weight-based insulin dosing,7 automated alerts to identify patients at greatest risk for hypoglycemia, and clinical decision support based on the preinsulin blood glucose level. We report the rates of iatrogenic hypoglycemia after the implementation of these order-set changes.
METHODS
Design Overview
EHR data were extracted from Epic Clarity. We analyzed data following Orderset 1.1 implementation (January 1, 2016-March 19, 2017) when hypoglycemia rates were reliably quantifiable and following orderset revision 1.2 (March 20, 2017-September 30, 2017) to evaluate the impact of the orderset intervention. The data collection was approved by the Institutional Review Board at the University of California, San Francisco.
Additionally, we explored the frequency in which providers ordered insulin through the hyperkalemia orderset for each version of the orderset via two-month baseline reviews. Investigation for Orderset 1.1 was from January 1, 2017 to February 28, 2017 and for Orderset 1.2 was from August 1, 2017 to September 30, 2017. Insulin ordering frequency through the hyperkalemia orderset was defined as ordering insulin through the adult inpatient hyperkalemia orderset versus ordering insulin in and outside of the hyperkalemia orderset.
Last, we measured the nursing point of care testing (POCT) blood glucose measurement compliance with the hyperkalemia orderset. Nursing utilization acceptance of the hyperkalemia orderset was defined as adequate POCT blood glucose levels monitored in comparison to all insulin treatments via the hyperkalemia orderset.
Setting and Participants
We evaluated nonobstetric adult inpatients admitted to UCSF Medical Center between January 2016 and September 2017. All medical and surgical wards and intensive care units were included in the analysis.
Intervention
In June 2012, an EHR developed by Epic Systems was introduced at UCSFMC. In January 2016, we designed a new EHR-based hyperkalemia treatment orderset (Orderset 1.1), which added standard POCT blood glucose checks before and at one, two, four, and six hours after insulin injection (Appendix 1). In March 2017, a newly designed orderset (Orderset 1.2) replaced the previous hyperkalemia treatment orderset (Appendix 2). Orderset 1.2 included three updates. First, providers were now presented the option of ordering insulin as a
CORRECTED FIGURE PER ERRATUM
Inclusion and exclusion criteria are shown in the Figure. All patients who had insulin ordered via a hyperkalemia orderset were included in an intention-to-treat analysis. A further analysis was performed for patients for whom orderset compliance was achieved (ie, insulin ordered through the ordersets with adequate blood glucose monitoring). These patients were required to have a POCT blood glucose check preinsulin administration and postinsulin administration as follows: (1) between 30 to 180 minutes (0.5 to three hours) after insulin administration, and (2) between 180 and 360 minutes (three to six hours) after insulin administration. For patients receiving repeated insulin treatments for hyperkalemia within six hours, the first treatment data points were excluded to prevent duplication.
Outcomes
We extracted data on all nonobstetric adult patients admitted to UCSFMC between January 1, 2016 and March 19, 2017 (Orderset 1.1) and between March 20, 2017 and September 30, 2017 (Orderset 1.2).
We measured unique insulin administrations given that each insulin injection poses a risk of iatrogenic hypoglycemia. Hypoglycemia was defined as glucose <70 mg/dL and severe hypoglycemia was defined as glucose <40 mg/dL. Covariates included time and date of insulin administration; blood glucose levels before and at one, two, four, and six hours after insulin injection (if available); sex; weight; dose of insulin given for hyperkalemia treatment; creatinine; known diagnosis of diabetes; concomitant use of albuterol; and concomitant use of corticosteroids. Hyperglycemia was defined as glucose >180 mg/dL. We collected potassium levels pre- and postinsulin treatment. The responsible team’s discipline and the location of the patient (eg, medical/surgical unit, intensive care unit, emergency department) where the insulin orderset was used were recorded.
Statistical Analysis
Statistical analysis for our data included the χ2 test for categorical data and Student t test for continuous data. The bivariate analysis identified potential risk factors and protective factors for hypoglycemia, and logistic regression was used to determine independent predictors of hypoglycemia. Through bivariate analyses, any factor with a P value below .05 was included in the multivariable analyses to investigate a significant contribution to hypoglycemia outcomes. Analyses for hypoglycemia and severe hypoglycemia rates, potassium levels pre- and postinsulin treatment, and hyperglycemia rates were done for both the intention-to-treat group and the group with all criteria met. All analyses were rendered utilizing Stata version 14 (Stata Corp LLC, College Station, Texas).
RESULTS
Baseline patient characteristics, initial insulin dosing, the treatment team, and the location are shown in Table 1. With the implementation of weight-based dosing, a lower dose of insulin was administered with Orderset 1.2 compared with Orderset 1.1.
Orderset adherence rates for Orderset 1.1 and 1.2 were as follows: Acute Care Floor 65% (70%), Intensive Care Unit 63% (66%), and Emergency Department 60% (55%). A two-month audit of orderset usage and compliance revealed that 73% (70 of 96) of insulin treatments were ordered through Orderset 1.1, and 77% (71 of 92) of insulin treatments were ordered through Orderset 1.2. The distribution of orderset usage across location and primary service are shown in Table 1.
The patient distribution is shown in the Figure. In the Orderset 1.1 period, there were 352 total insulin treatments utilizing the newly revised UCSFMC adult inpatient hyperkalemia orderset that were used for the intention-to-treat analysis, and there were 225 patients for whom compliance with orderset monitoring was achieved. Notably, 112 treatments were excluded for the lack of adequate blood glucose monitoring. In the Orderset 1.2 period, there were 239 total insulin treatments utilizing the newly revised UCSFMC adult inpatient hyperkalemia orderset that were used for the intention-to-treat analysis, and there were 145 patients for whom compliance with orderset monitoring was achieved. During this phase, 80 treatments were excluded for inadequate blood glucose monitoring.
Predictors of hypoglycemia following the implementation of Orderset 1.1 are shown in Table 2, and the logistic regression model of these risks is shown in Appendix Table 1. Female gender, weight-based dosing of insulin exceeding 0.14 units/kg, preinsulin blood glucose less than 140 mg/dL, and serum creatinine greater than 2.5 mg/dl were associated with an increased risk of hypoglycemia. A known diagnosis of Type 2 diabetes, concomitant albuterol within one hour of insulin administration, and corticosteroid administration within two hours of insulin administration were associated with a decreased risk of hypoglycemia.
The rates of hypoglycemia (<70 mg/dl) and severe hypoglycemia (<40 mg/dl) are shown in Table 3. During the Orderset 1.1 period,
During the Orderset 1.2 period, for patients with all criteria met, 14 of 145 (10%) had hypoglycemia, and three of 145 (2%) had severe hypoglycemia. Ten of 14 (72%) of these hypoglycemic events occurred in the first three hours, with the remaining four hypoglycemic events (28%) occurring in the last three hours.
An intention-to-treat analysis for hyperglycemia, defined as glucose >180 mg/dl, revealed that during the Orderset 1.1 period, 80 of 352 (23%) had hyperglycemia before insulin administration, and 38 of 352 (11%) had hyperglycemia after insulin administration. During the Orderset 1.2 period, 52 of 239 (22%) had hyperglycemia before insulin administration, and 15 of 239 (6%) had hyperglycemia after insulin administration. Results can be found in Table 3.
Pre- and posttreatment potassium levels are shown in Table 3. An intention-to-treat analysis for potassium reduction postinsulin administration revealed that during the Orderset 1.1 period, there was an absolute reduction of 0.73 mmol/L, while during the Orderset 1.2 period, there was an absolute reduction of 0.95 mmol/L.
DISCUSSION
Treatment of hyperkalemia with insulin may result in significant iatrogenic hypoglycemia. Prior studies have likely underestimated the incidence of hyperkalemia treatment-associated hypoglycemia as glucose levels are rarely checked within three hours of insulin administration.8 In our study, which was designed to ensure appropriate blood glucose measurement, 21% of insulin treatments for hyperkalemia resulted in hypoglycemia, with 92% of hypoglycemic events occurring within the first three hours.
For the Orderset 1.1 period, patient risk factors identified for iatrogenic hypoglycemia postinsulin administration were female sex, doses of regular insulin greater than 0.14 units/kg, preinsulin blood glucose less than 140 mg/dL, and serum creatinine greater than 2.5 mg/dL. These results are consistent with studies suggesting that preinsulin blood glucose levels less than 140 mg/dL and the standard 10 units of insulin for hyperkalemia treatment may increase the risk of hypoglycemia.4,7,9
To decrease the risk of iatrogenic hypoglycemia, we redesigned our hyperkalemia insulin orderset to address the strongest predictors of hypoglycemia (doses of regular insulin greater than 0.14 units/kg and preinsulin blood glucose less than 140 mg/dL). The main changes were weight-based insulin dosing (based on previously published data)10 and adjustment of glucose administration based on the patient’s glucose levels.11 Following these changes, the rates of both hypoglycemia and severe hypoglycemia were statistically significantly reduced. In addition, of the 14 hypoglycemia events identified after the introduction of Orderset 1.2, five could have been prevented (36%) had the protocol been strictly followed. These five hypoglycemia events occurred later than one-hour postinsulin administration in patients with blood sugars < 150 mg/dL prior to insulin administration. In each of these cases, Orderset 1.2 called for an additional dextrose 50% (50 mL) IV bolus, which likely would have prevented the subsequently recorded hypoglycemia. In other words, our orderset indicated that these patients received an additional bolus of dextrose. However, they did not receive their glucose at the appropriate time, contributing to the hypoglycemia events. The orderset did not include a best practice alert (BPA) to remind providers about the extra dextrose bolus. In the future, we plan to add this BPA.
The hypoglycemia rate identified by Orderset 1.1 was 21% and the hypoglycemia rate identified by the Orderset 1.2 was 10%. The severe hypoglycemia rate identified by Orderset 1.1 was 5% and the severe hypoglycemia rate identified by Orderset 1.2 was 2%. The hypoglycemia and severe hypoglycemia rates significantly decreased after the introduction of Orderset 1.2. To mimic a real-world clinical setting, where monitoring of blood glucose is not always achieved multiple times within a six-hour timeframe of postinsulin treatment for hyperkalemia, we conducted an intention-to-treat analysis. Even when including patients for whom full blood glucose monitoring was not achieved, the introduction of Orderset 1.2 was associated with a significant decrease in the hypoglycemia rate.
To demonstrate whether weight-based dosing of insulin was as effective as a standard dose for hyperkalemia treatment, we compared the impact of Orderset 1.1, which only had the option for single standard doses of insulin, with the impact of Orderset 1.2, which included weight-based dosing options. With the introduction of Orderset 1.2, there was a significant decrease in serum potassium, indicating that weight-based dosing options may not only prevent hypoglycemia but may potentially provide more effective hyperkalemia treatment.
We also compared the rate of hyperglycemia (a glucose >180 mg/dl) pre- and posttreatment (Table 3). Although not statistically significant, the rate of hyperglycemia decreased from 11% to 6%, suggesting a trend toward decreased hyperglycemia with orderset usage.
As orderset usage for hyperkalemia management only occurred approximately 75% of the time, likely, forcing the use of these ordersets would further reduce the incidence of treatment-associated hypoglycemia. To encourage the use of ordersets for hyperkalemia management, our medical center has largely restricted insulin ordering so that it can only be done through ordersets with the proper precautions in place, regardless of the indication. Furthermore, adherence to all the blood glucose monitoring orders embedded in the ordersets remained suboptimal irrespective of managing the service or clinical setting
Finally, development and implementation of these hyperkalemia treatment ordersets required an experienced multidisciplinary team, including pharmacists, nurses, hospitalists, endocrinologists, and EHR system programmers.12,13 We, therefore, encourage interprofessional collaboration for any institutions seeking to establish innovative clinical protocols.
This analysis was limited to the insulin administration meeting our inclusion criteria. Thus, we could not identify a true hypoglycemia rate for treatments that were not followed by adequate blood glucose monitoring postinsulin administration, or for insulin administration ordered outside of the hyperkalemia ordersets.
CONCLUSION
The use of a comprehensive EHR orderset for the treatment of hyperkalemia with predefined times for blood glucose monitoring, weight-based insulin dosing, and prompts to warn providers of an individual patient’s risk for hypoglycemia may significantly reduce the incidence of iatrogenic hypoglycemia.
1. Acker CG, Johnson JP, Palevsky PM, Greenberg A. Hyperkalemia in hospitalized patients: causes, adequacy of treatment, and results of an attempt to improve physician compliance with published therapy guidelines. Arch Intern Med. 1998;158(8):917-924. https://doi.org/10.1001/archinte.158.8.917.
2. Fordjour KN, Walton T, Doran JJ. Management of hyperkalemia in hospitalized patients. Am J Med Sci. 2014;347(2):93-100. https://doi.org/10.1097/MAJ.0b013e318279b105.
3. Part-10-Special-Circumstances-of-Resuscitation.pdf. https://eccguidelines.heart.org/wp-content/themes/eccstaging/dompdf-master/pdffiles/part-10-special-circumstances-of-resuscitation.pdf. Accessed December 16, 2017.
4. Apel J, Reutrakul S, Baldwin D. Hypoglycemia in the treatment of hyperkalemia with insulin in patients with end-stage renal disease. Clin Kidney J. 2014;7(3):248-250. https://doi.org/10.1093/ckj/sfu026.
5. Schafers S, Naunheim R, Vijayan A, Tobin G. Incidence of hypoglycemia following insulin-based acute stabilization of hyperkalemia treatment. J Hosp Med. 2012;7(3):239-242. https://doi.org/10.1002/jhm.977.
6. Boughton CK, Dixon D, Goble E, et al. Preventing hypoglycemia following treatment of hyperkalemia in hospitalized patients. J Hosp Med. 2019;14:E1-E4. https://doi.org/10.12788/jhm.3145.
7. Wheeler DT, Schafers SJ, Horwedel TA, Deal EN, Tobin GS. Weight-based insulin dosing for acute hyperkalemia results in less hypoglycemia. J Hosp Med. 2016;11(5):355-357. https://doi.org/10.1002/jhm.2545.
8. Coca A, Valencia AL, Bustamante J, Mendiluce A, Floege J. Hypoglycemia following intravenous insulin plus glucose for hyperkalemia in patients with impaired renal function. PLoS ONE. 2017;12(2):e0172961. https://doi.org/10.1371/journal.pone.0172961.
9. LaRue HA, Peksa GD, Shah SC. A comparison of insulin doses for the treatment of hyperkalemia in patients with renal insufficiency. Pharmacotherapy. 2017;37(12):1516-1522. https://doi.org/10.1002/phar.2038.
10. Brown K, Setji TL, Hale SL, et al. Assessing the impact of an order panel utilizing weight-based insulin and standardized monitoring of blood glucose for patients with hyperkalemia. Am J Med Qual. 2018;33(6):598-603. https://doi.org/10.1177/1062860618764610.
11. Farina N, Anderson C. Impact of dextrose dose on hypoglycemia development following treatment of hyperkalemia. Ther Adv Drug Saf. 2018;9(6):323-329. https://doi.org/10.1177/2042098618768725.
12. Neinstein A, MacMaster HW, Sullivan MM, Rushakoff R. A detailed description of the implementation of inpatient insulin orders with a commercial electronic health record system. J Diabetes Sci Technol. 2014;8(4):641-651. https://doi.org/10.1177/1932296814536290.
13. MacMaster HW, Gonzalez S, Maruoka A, et al. Development and implementation of a subcutaneous Insulin pen label bar code scanning protocol to prevent wrong-patient insulin pen errors. Jt Comm J Qual Patient Saf. 2019;45(5):380-386. https://doi.org/10.1016/j.jcjq.2018.08.006.
1. Acker CG, Johnson JP, Palevsky PM, Greenberg A. Hyperkalemia in hospitalized patients: causes, adequacy of treatment, and results of an attempt to improve physician compliance with published therapy guidelines. Arch Intern Med. 1998;158(8):917-924. https://doi.org/10.1001/archinte.158.8.917.
2. Fordjour KN, Walton T, Doran JJ. Management of hyperkalemia in hospitalized patients. Am J Med Sci. 2014;347(2):93-100. https://doi.org/10.1097/MAJ.0b013e318279b105.
3. Part-10-Special-Circumstances-of-Resuscitation.pdf. https://eccguidelines.heart.org/wp-content/themes/eccstaging/dompdf-master/pdffiles/part-10-special-circumstances-of-resuscitation.pdf. Accessed December 16, 2017.
4. Apel J, Reutrakul S, Baldwin D. Hypoglycemia in the treatment of hyperkalemia with insulin in patients with end-stage renal disease. Clin Kidney J. 2014;7(3):248-250. https://doi.org/10.1093/ckj/sfu026.
5. Schafers S, Naunheim R, Vijayan A, Tobin G. Incidence of hypoglycemia following insulin-based acute stabilization of hyperkalemia treatment. J Hosp Med. 2012;7(3):239-242. https://doi.org/10.1002/jhm.977.
6. Boughton CK, Dixon D, Goble E, et al. Preventing hypoglycemia following treatment of hyperkalemia in hospitalized patients. J Hosp Med. 2019;14:E1-E4. https://doi.org/10.12788/jhm.3145.
7. Wheeler DT, Schafers SJ, Horwedel TA, Deal EN, Tobin GS. Weight-based insulin dosing for acute hyperkalemia results in less hypoglycemia. J Hosp Med. 2016;11(5):355-357. https://doi.org/10.1002/jhm.2545.
8. Coca A, Valencia AL, Bustamante J, Mendiluce A, Floege J. Hypoglycemia following intravenous insulin plus glucose for hyperkalemia in patients with impaired renal function. PLoS ONE. 2017;12(2):e0172961. https://doi.org/10.1371/journal.pone.0172961.
9. LaRue HA, Peksa GD, Shah SC. A comparison of insulin doses for the treatment of hyperkalemia in patients with renal insufficiency. Pharmacotherapy. 2017;37(12):1516-1522. https://doi.org/10.1002/phar.2038.
10. Brown K, Setji TL, Hale SL, et al. Assessing the impact of an order panel utilizing weight-based insulin and standardized monitoring of blood glucose for patients with hyperkalemia. Am J Med Qual. 2018;33(6):598-603. https://doi.org/10.1177/1062860618764610.
11. Farina N, Anderson C. Impact of dextrose dose on hypoglycemia development following treatment of hyperkalemia. Ther Adv Drug Saf. 2018;9(6):323-329. https://doi.org/10.1177/2042098618768725.
12. Neinstein A, MacMaster HW, Sullivan MM, Rushakoff R. A detailed description of the implementation of inpatient insulin orders with a commercial electronic health record system. J Diabetes Sci Technol. 2014;8(4):641-651. https://doi.org/10.1177/1932296814536290.
13. MacMaster HW, Gonzalez S, Maruoka A, et al. Development and implementation of a subcutaneous Insulin pen label bar code scanning protocol to prevent wrong-patient insulin pen errors. Jt Comm J Qual Patient Saf. 2019;45(5):380-386. https://doi.org/10.1016/j.jcjq.2018.08.006.
© 2020 Society of Hospital Medicine
The Group Practice Manager in the VHA: A View From the Field
The Veterans Health Administration (VHA) provides care for 9 million veterans at 1,255 health care sites linked to one of 170 local medical systems.1 Recognizing that providing timely care requires effective access management, the US Congress mandated training of VHA staff to manage and improve access to care but did not provide additional local funds for new positions.2 In response, the VHA created the group practice manager (GPM), a new position responsible for improving clinical practice management and unifying access improvement across leadership levels, professions, and services within each local medical system.
In May 2015, the VHA began hiring and training GPMs to spearhead management of access to services. The US Department of Veterans Affairs (VA) Office of Veteran Access to Care spearheaded GPM training, including face-to-face sessions, national calls, webinars, and educational materials. Five local medical systems were selected by the VA Office of Veteran Access to Care to implement the GPM role to allow for an early evaluation of the program that would inform a subsequent nationwide rollout. Implementation of the GPM role remained in the hands of local medical systems.
Longer wait times are shown to impact patient health.3,4 Open access scheduling and other patient-centered access management interventions have been shown to improve availability of primary care appointments.5 However, little empirical evidence exists regarding the managers who focus on clinic access interventions. While the nonpeer-reviewed literature includes references to such roles, including GPMs, the empirical literature has focused on external practice faciliators,6-8 “mid-level managers,”9 and clinic staff.10 We found no peer-reviewed articles on the needs and experiences of practice managers who are focused on improving access. The purpose of this study was to examine GPM prototype sites to both enhance subsequent nationwide implementation and to advance empirical literature on managing patient access within health care.
Methods
In 2015, the VA identified 5 prototype sites representing diverse geographic locations, size, and complexity for the implementation of the GPM role (Table 1). These sites self-identified as having clinical practice management experience. GPMs attended 4 training sessions between February and August 2015.
Data Collection
Participants from each prototype site included GPMs, national trainers, clinic leaders, and frontline staff. Table 2 includes the roles and sample size. Participants were recruited through purposive sampling followed by snowball sampling until thematic saturation was reached (the point at which subsequent data fail to produce new findings across sites and roles of interest).
Guided by the Consolidated Framework for Implementation Research (CFIR), the research team developed semistructured interview guides tailored to participants’ roles to elicit rich descriptions regarding overall impressions, practice management strategies, goals, activities, relationship to clinic roles, data analytics usage, challenges, barriers, and facilitators.11 These guides included open-ended questions and structured prompts utilizing participant language for follow-up probes to minimize interviewing bias (eAppendix:
Data Analysis
Data were analyzed using iterative deductive and inductive content analysis.12 Deductive content analysis consisted of identifying quotes that fit within preidentified categories (ie, perceptions of national effort, organizational structure for GPM, challenges, facilitators, metrics and tools, and mobilizing access culture) developed by the interdisciplinary research team. Further content analysis entailed open-coding and iteratively revisiting and reconciling codes associated within each preidentified category as new codes emerged. The team analyzed the resulting codes to inductively and iteratively identify and stabilize themes regarding the GPM role: roles and tasks, GPM characteristics, issues, and challenges. Through this process we moved coded data to reconciled descriptions suited to addressing the purposes of this study. Dedoose 7.0.23 software was used for qualitative data management and analysis.
Results
The study identified participants’ overall impressions of the GPM initiative and key themes within 4 major domains regarding implementing the GPM role: roles and tasks (implementing clinic practice management, leading patient access, supporting data analytics, and enabling self and staff); GPM characteristics (familiarity with clinical services, knowledge of VHA systems, ability to analyze patient data, communication skills, and the ability to work with others); and issues, and challenges (technical, social, and structural).
Overall Impressions
Interviewees perceived the GPM initiative as a consolidation of existing distributed responsibilities into one role that directly reported to local top-level management with indirect reporting to national leaders. Many of the sites reported that they had designated or planned to designate a role resembling the GPM prior to the initiative. “There are staff who’ve been doing some of this work all along,” a GPM noted. “We just didn’t have them grouped together. They weren’t necessarily all working in the same type of service under the same type of structure.”
Whether the GPM position was new or not, participants referenced the importance and challenges of engaging the local facility in recognizing the agency associated with the GPM position. According to national support, the staff are trying to get the facility to understand “why the group practice manager is so important… we’ve got to embed that standard position in the system.”
While the GPM was recognized as the hub of access management, respondents recognized that transformation regarding access involved many players. “We have to create [an] orchestrated team inside each facility,” an advisor argued.
Respondents discussed how the initiative allows local facilities to appoint a specific person with a specific title and role who helps facilitate, organize, and legitimize an access focus at their sites. One GPM interviewee noted how the initiative helped refocus some of their previously less centralized efforts. “We’ve always looked at productivity; we’ve always looked at access; we’ve always looked at efficiency. I think the bigger difference is now there are individuals identified in the clinics, as practice managers as well…I interact with them. They interact with individual clinic staff, and it’s more of a group process than a single individual.”
The value of having tools available and being able to track and manage patient care as a specific example of the positive impact of this new role was noted by participants. A GPM noted that many health care providers will be happy to have tools to better manage their services and a process “that flows from a service level all the way up to executive management, where there is a common interest in making those things happen—I think that’s going to be a tremendous help.”
Participants expressed concern that the national GPM rollout would be a one-size-fits-all approach. These respondents emphasized the need to have the flexibility to customize their activities to meet their unique site and patient needs.
GPM Roles and Tasks
Participants described 4 primary roles that the GPM was expected to fill: implementing clinic practice management, leading patient access, supporting data analytics, and enabling self and staff. Some activities overlapped in that they served to support multiple role areas (Figure 1).
Implementing clinic practice management. In the early stages of the initiative, the GPM’s primary role was to prepare the facility to implement a standardized set of clinic practice management (CPM) team processes. Part of standardizing the CPM process was defining the scope and tasks of the GPM, which requires significant planning for the implementation. “My big job is to finalize what we think group practice management is going to look [like] here,” a GPM reported.
Each prototype site had latitude to interpret the GPM initiative in a way that would work in their context within given VHA boundaries and ongoing initiatives. To achieve the high-level vision and purpose, the GPM first had to develop action plans that accounted for the operating environment of the facility. According to one GPM, VA national officials are “constantly” asking for action plans, which required significant time by specific deadlines. “They want an action plan [and to] clean up all your consults, [and to] clean up all your recall reminders.”
Leading on improving access efforts. Participants saw the GPM as the central staff member responsible for providing oversight of any activities and people involved in improving access. “I ensure everybody is doing what they’re supposed to do,” one GPM reported. When the GPM sees areas that are not being addressed, the individual tries to develop a process or training to “close those gaps.”
GPMs promoted an awareness of their goals, changes in process, and new tools accompanying the initiative. However, other access initiatives were occurring simultaneously creating confusion for health care providers and patients; thus GPMs found they were managing a wide array of related initiatives.
GPMs have to negotiate with leaders across the VHA facility, many of whom operated at a higher leadership level and had different priorities, to address access problems.
“I’m a lieutenant as a GPM in a clinic, a GPM noted. “How is the lieutenant going to talk to a major or a colonel in the clinic and say your clinic has problems. How[‘s] that lieutenant...going to do that? With people skills!”
Managing expectations about the speed and to what extent a problem could be resolved was an important part of the GPM leadership role. “I see myself as managing expectations both up to the leadership and down to the frontline,” a GPM explained. “I find myself talking to leadership [about] our progress. But at the same time, we have to say, ‘not everything can be fixed overnight.’”
Providing leadership on access-related issues included developing a range of options for addressing patient access problems. One analytics manager recounted how the GPM role led to evaluating how physical space limited efficiency in clinic flow. The first step was identifying possible additional rooms to improve clinic flow. This required working with the space committee to “get someone to look at our overarching space and find someplace else for them to sit” to avoid adding to congestion in the clinic area.
Supporting data analytics.
GPMs routinely immersed themselves in the data to understand access issues. GPMs worked with clinic leaders to identify the underlying causes and various solutions. The GPMs maintained access through administrative scrubbing of the data and finding “smart ways to get patients scheduled,” a GPM explained. “I don’t think our facility would have taken care of as many veterans in the time frame as we did....We’ve cleared over 4,000 consults that were older than 90 days. We have cleared thousands and thousands of weekly reminders.”
GPMs expressed the need for aggregated (ie, dashboard) and standardized information to efficiently address access issues. “I would like to have some more standardization on what’s being reviewed; it seems to change frequently, and so [to] be able to track and trend and have something given to me to review,” one health care provider requested. On the other hand, participants also described a need for decision support tools that would lead to action aligned with best practices. “Instead of a dashboard or something that’s just measuring their performance, it’s more something that they can look at and take action” a national support staff advisor suggested.
Enabling self and staff. GPMs felt they were most effective if they enabled themselves and stakeholders through training and by cultivating relationships and team building. Figure 2 illustrates the various stakeholders GPMs reported engaging with. The GPMS should be building relationships, bridging relationships, developing trust, and then providing a higher level of hands-on management. However, “that doesn’t really happen right now, day to day,” one member of leadership reported.
Key topics in GPM leadership training included both soft skills (change management, culture change, and negotiation skills) and crucial analytic/technical training (understanding each metric and dashboard available, data analytics, and supply/demand balancing techniques). The GPMs not only wanted to understand metrics as part of their training, but also want to know what to do about them.
An “operationalization” training approach (discerning the meaning of data, data-based decision making, and determining action from multiple options) inspired by real-life situations was preferred by participants. Other effective learning structures included job aids in the form of templated Gantt charts, process maps to guide GPMs through implementation of new processes, formalized peer learning (accumulated field insights shared during training courses), and informal peer sharing of direct experiences during calls.
GPMs also emphasized training for frontline clinical and support staff, including schedulers. VHA schedulers typically have less education and experience higher turnover rates than do other clinic staff, yet they carry out complex and critical tasks. Providing training and ensuring that any materials developed for training and education were appropriate to the level of education of schedulers was an important task for GPMs. “If they don’t understand all of the scheduling principles and potential,” one GPM explained, “we will not be maximizing the utilization of our parts.”
GPMs also provided informal education to clinicians. Participants noted GPMs have to avoid appearing to overstep their positions or presuming more knowledge and expertise than clinicians. They “have to be able to teach a physician without being overbearing, in a way a physician will accept it as advisement,” one program leader reported.
GPM knowledge, skills, and abilities. GPMs presented a complex range of knowledge, skills, and abilities, including clinical, administrative, analytics, and people skills. All interviewees reported that their prior education and experience did not sufficiently train them for the GPM role. GPMs identified a willingness to learn quickly as a critical characteristic. Many GPMs tended to have a formal education in health administration or business (eg, MBAs); others had administrative experience (eg, administrative assistance to executive leadership) or clinical training (eg, physician assistant). Detailed clinical knowledge was not expected, but clinical familiarity was helpful.
Some interviewees also mentioned previous experience and familiarity with the VHA system specifically as an advantage. This was especially true for VA outpatient flows, clinic flows, and understanding what an outpatient is in a VA context. Interviewees noted the importance of GPMs needing to be able to analyze patient demand metrics and underlying data in order to determine supply of providers and then to allocate adequate resources to complement providers. Forecasting skills were referenced as a key point. “They need to be able to be assured that they can recruit more providers if needed,” a national support staff advisor noted.
Given the importance of developing effective relationships, communication skills were mentioned by most participants and underscored as critical to establishing trust between GPMs and others as the initiative was being implemented. Interviewees indicated that relationship building was further enhanced when GPMs possessed the ability to “work with” rather than command clinicians and staff; navigate politics; and were respectful of other people’s knowledge, skills, abilities, and status. “They have to work with the nursing staff and teach them,” a leader described, “so that people understand that we are going to a different place to achieve our primary objectives and goals.”
Issues and Challenges
Participants identified several technical, social, and structural challenges and barriers to successfully implementing the GPM role.
Technical challenges. Recurring themes across all phases of data analytics were GPMs’ capability to challenge data use and use large volumes of information from multiple data sources (entering and accessing data; “drilling down” from summaries; generating reports; and analyzing and interpreting resulting metrics). Interviewees reported that information assessment and analytic support were not consistent. One GPM had a data analyst pulling reports needed to support clinical units while other GPMs trained staff to pull data. Even with support, some GPMs had issues due to limited information technology (IT) skills or access privileges leading to inefficiencies and delays. “Whenever I need anything from a programmer, I have to go through, you know, the IT gods in the sky,” one GPM remarked. “That usually takes a few months or more.”
Social challenges. Instituting the GPM role was a cultural change, and interviewees reported needing to address resistance to CPM model efforts. Resistance to change “is particularly hard in the VA just because it has a unique culture,” one leader noted. “There is a comfort in the legacy way of doing things.” The GPM initiative was introduced during a time when other national level initiatives were being implemented throughout the VHA. Fulfilling requests for information for these initiatives became the responsibility of the GPM and their team, which diverted attention from the mandate to improve access. Furthermore, GPMs were often considered the “change communicators” to clinics putting them in the role of “bad messenger,” which degraded trust and made it difficult to partner with clinicians.
Efforts to work through change management and build relationships included general program awareness presentation to internal stakeholders; including key stakeholders in GPM committees; pre-emptive conversations with unit chiefs; creating awareness of the GPM activities and progress through formal and informal update meetings; and identifying successes regarding access.
Structural challenges. The GPM role did not have direct supervision over clinical and administrative leaders, making it challenging to enact change. GPMs reported that “they do not always have authority over the area that they are being asked to manage,” which made their work difficult, requiring strong negotiation skills and political savvy to affect change. However, as the clinic staff and providers saw how the GPM could support and positively impact their practice, these challenges began to subside.
Discussion
This study provides empirical evidence regarding the implementation of a new access management strategy for health care systems focused on improving timeliness of care. First, the GPM position was seen as critical at each facility, as a single point person, to help local system leaders respond effectively to both national mandates and local context. Second, requiring the GPMs to report to the medical center director or chief of staff was important for integrating access perspectives across service lines and to facilitate a strong GPM role in strategic planning. Third, the intentional flexibility of the access management initiative, beyond the nationally specified aspects of the GPM role, was key for allowing individual sites to adapt to unique local challenges, resources, and population demands. Fourth, the initiative provided GPMs with opportunities to learn important skills and support the acquisition, utilization, and communication of a tremendous range of data toward responsive action.
According to our respondents, the GPM role demands functioning across a broad set of responsibilities; understanding the big picture as well as the complex underlying variables; engaging facility leaders, clinical and administrative staff; and prioritizing competing national and local demands. Consistent with previous findings, effective GPMs must possess a complex set of skills (interpersonal, analytic, and leadership) and the ability to create a supporting team.13
In practice, improving access at individual sites of care (VA medical centers and community-based outpatient clinics) poses significant challenges, especially in the early stages, even with the assistance of a GPM. For example, some respondents reported being overwhelmed by the volume of available data and dashboards, and responding to current requests for data analysis and dissemination sometimes impeded long range planning. Multiple national access-related initiatives and local pressures also generated excessive and potentially conflicting demands. Thus, while the creation of a GPM position seemed to be essential for the pilot sites to improve local access and timeliness to care, success also requires ongoing national and facility-wide communication, education, and support. Ongoing data analysis training and support will be critical to ensuring the sustainability of the position. Last, recruiting GPMs with the needed complex skill set may prove to be challenging, and it will be important to provide resources to identify, attract, and retain well-qualified GPMs.
Limitations and Future Work
This study was based on a small initial sample of pilot sites of varying sizes and, as such, may not reflect the experience of all VHA GPMs. In addition, the use of snowball sampling, while facilitating identification of key stakeholders, may introduce bias in participant sampling. Nonetheless, the results from this study provide findings that informed the national VHA GPM initiative and can inform further studies of practice management roles outside of the VA.
Further study of the VHA GPM implementation and similar roles in other health care systems is needed. As the GPM position is fully implemented across the VHA, large scale evaluation is needed to gain a more representative picture and allow for comparison of the GPM role at various types of facilities (eg, size, rurality, complexity, ranking based on access performance metrics).
Conclusion
Improving access to care is a central goal for health care systems. The incorporation of the GPM role is an innovative approach to improve access management strategies. Early study of prototype sites provided VHA leadership with valuable insights used to influence further rollout of this initiative. Based on our findings, national and local support are important to ongoing success. National access mandates, training, and resources should focus on ensuring sufficient GPM authority, enabling GPMs to use data, and ensuring GPMs engage with frontline clinical and administrative staff to improve veteran access to care.
1. US Department of Veterans Affairs. Veterans Health Administration. https://www.va.gov/health. Updated October 25, 2019. Accessed January 8, 2020.
2. Veterans Access, Choice, and Accountability Act of 2014. 38 CFR § 17.1500 (2014).
3. Fahmy N, Aprikian A, Al-Otaibi M, et al. Impact of treatment delay in patients with bladder cancer managed with partial cystectomy in Quebec: a population-based study. Can Urol Assoc J. 2009;3(2):131-135.
4. Hill CJ, Joonas K. The impact of unacceptable wait time on health care patients’ attitudes and actions. Health Mark Q. 2005;23(2):69-87.
5. Ansell D, Crispo JAG, Simard B, Bjerre LM. Interventions to reduce wait times for primary care appointments: a systematic review. BMC Health Serv Res. 2017;17(1):295.
6. Kotecha J, Han H, Green M, Russell G, Martin MI, Birtwhistle R. The role of the practice facilitators in Ontario primary healthcare quality improvement. BMC Fam Pract. 2015;16:93.
7. Taylor EF, Machta RM, Meyers DS, Genevro J, Peikes DN. Enhancing the primary care team to provide redesigned care: the roles of practice facilitators and care managers. Ann Fam Med. 2013;11(1):80-83.
8. Liddy C, Laferriere D, Baskerville B, Dahrouge S, Knox L, Hogg W. An overview of practice facilitation programs in Canada: current perspectives and future directions. Healthc Policy. 2013;8(3):58-67.
9. Birken SA, Lee SY, Weiner BJ, Chin MH, Schaefer CT. Improving the effectiveness of health care innovation implementation: middle managers as change agents. Med Care Res Rev. 2013;70(1):29-45.
10. Ahluwalia S, Offredy M. A qualitative study of the impact of the implementation of advanced access in primary healthcare on the working lives of general practice staff. BMC Fam Pract. 2005;6:39.
11. Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 2009;4:50.
12. Elo S, Kyngäs H. The qualitative content analysis process. J Adv Nurs. 2008;62(1):107-115.
13. Stefl ME. Common competencies for all healthcare managers: the Healthcare Leadership Alliance model. J Healthc Manag. 2008;53(6):360-374.
The Veterans Health Administration (VHA) provides care for 9 million veterans at 1,255 health care sites linked to one of 170 local medical systems.1 Recognizing that providing timely care requires effective access management, the US Congress mandated training of VHA staff to manage and improve access to care but did not provide additional local funds for new positions.2 In response, the VHA created the group practice manager (GPM), a new position responsible for improving clinical practice management and unifying access improvement across leadership levels, professions, and services within each local medical system.
In May 2015, the VHA began hiring and training GPMs to spearhead management of access to services. The US Department of Veterans Affairs (VA) Office of Veteran Access to Care spearheaded GPM training, including face-to-face sessions, national calls, webinars, and educational materials. Five local medical systems were selected by the VA Office of Veteran Access to Care to implement the GPM role to allow for an early evaluation of the program that would inform a subsequent nationwide rollout. Implementation of the GPM role remained in the hands of local medical systems.
Longer wait times are shown to impact patient health.3,4 Open access scheduling and other patient-centered access management interventions have been shown to improve availability of primary care appointments.5 However, little empirical evidence exists regarding the managers who focus on clinic access interventions. While the nonpeer-reviewed literature includes references to such roles, including GPMs, the empirical literature has focused on external practice faciliators,6-8 “mid-level managers,”9 and clinic staff.10 We found no peer-reviewed articles on the needs and experiences of practice managers who are focused on improving access. The purpose of this study was to examine GPM prototype sites to both enhance subsequent nationwide implementation and to advance empirical literature on managing patient access within health care.
Methods
In 2015, the VA identified 5 prototype sites representing diverse geographic locations, size, and complexity for the implementation of the GPM role (Table 1). These sites self-identified as having clinical practice management experience. GPMs attended 4 training sessions between February and August 2015.
Data Collection
Participants from each prototype site included GPMs, national trainers, clinic leaders, and frontline staff. Table 2 includes the roles and sample size. Participants were recruited through purposive sampling followed by snowball sampling until thematic saturation was reached (the point at which subsequent data fail to produce new findings across sites and roles of interest).
Guided by the Consolidated Framework for Implementation Research (CFIR), the research team developed semistructured interview guides tailored to participants’ roles to elicit rich descriptions regarding overall impressions, practice management strategies, goals, activities, relationship to clinic roles, data analytics usage, challenges, barriers, and facilitators.11 These guides included open-ended questions and structured prompts utilizing participant language for follow-up probes to minimize interviewing bias (eAppendix:
Data Analysis
Data were analyzed using iterative deductive and inductive content analysis.12 Deductive content analysis consisted of identifying quotes that fit within preidentified categories (ie, perceptions of national effort, organizational structure for GPM, challenges, facilitators, metrics and tools, and mobilizing access culture) developed by the interdisciplinary research team. Further content analysis entailed open-coding and iteratively revisiting and reconciling codes associated within each preidentified category as new codes emerged. The team analyzed the resulting codes to inductively and iteratively identify and stabilize themes regarding the GPM role: roles and tasks, GPM characteristics, issues, and challenges. Through this process we moved coded data to reconciled descriptions suited to addressing the purposes of this study. Dedoose 7.0.23 software was used for qualitative data management and analysis.
Results
The study identified participants’ overall impressions of the GPM initiative and key themes within 4 major domains regarding implementing the GPM role: roles and tasks (implementing clinic practice management, leading patient access, supporting data analytics, and enabling self and staff); GPM characteristics (familiarity with clinical services, knowledge of VHA systems, ability to analyze patient data, communication skills, and the ability to work with others); and issues, and challenges (technical, social, and structural).
Overall Impressions
Interviewees perceived the GPM initiative as a consolidation of existing distributed responsibilities into one role that directly reported to local top-level management with indirect reporting to national leaders. Many of the sites reported that they had designated or planned to designate a role resembling the GPM prior to the initiative. “There are staff who’ve been doing some of this work all along,” a GPM noted. “We just didn’t have them grouped together. They weren’t necessarily all working in the same type of service under the same type of structure.”
Whether the GPM position was new or not, participants referenced the importance and challenges of engaging the local facility in recognizing the agency associated with the GPM position. According to national support, the staff are trying to get the facility to understand “why the group practice manager is so important… we’ve got to embed that standard position in the system.”
While the GPM was recognized as the hub of access management, respondents recognized that transformation regarding access involved many players. “We have to create [an] orchestrated team inside each facility,” an advisor argued.
Respondents discussed how the initiative allows local facilities to appoint a specific person with a specific title and role who helps facilitate, organize, and legitimize an access focus at their sites. One GPM interviewee noted how the initiative helped refocus some of their previously less centralized efforts. “We’ve always looked at productivity; we’ve always looked at access; we’ve always looked at efficiency. I think the bigger difference is now there are individuals identified in the clinics, as practice managers as well…I interact with them. They interact with individual clinic staff, and it’s more of a group process than a single individual.”
The value of having tools available and being able to track and manage patient care as a specific example of the positive impact of this new role was noted by participants. A GPM noted that many health care providers will be happy to have tools to better manage their services and a process “that flows from a service level all the way up to executive management, where there is a common interest in making those things happen—I think that’s going to be a tremendous help.”
Participants expressed concern that the national GPM rollout would be a one-size-fits-all approach. These respondents emphasized the need to have the flexibility to customize their activities to meet their unique site and patient needs.
GPM Roles and Tasks
Participants described 4 primary roles that the GPM was expected to fill: implementing clinic practice management, leading patient access, supporting data analytics, and enabling self and staff. Some activities overlapped in that they served to support multiple role areas (Figure 1).
Implementing clinic practice management. In the early stages of the initiative, the GPM’s primary role was to prepare the facility to implement a standardized set of clinic practice management (CPM) team processes. Part of standardizing the CPM process was defining the scope and tasks of the GPM, which requires significant planning for the implementation. “My big job is to finalize what we think group practice management is going to look [like] here,” a GPM reported.
Each prototype site had latitude to interpret the GPM initiative in a way that would work in their context within given VHA boundaries and ongoing initiatives. To achieve the high-level vision and purpose, the GPM first had to develop action plans that accounted for the operating environment of the facility. According to one GPM, VA national officials are “constantly” asking for action plans, which required significant time by specific deadlines. “They want an action plan [and to] clean up all your consults, [and to] clean up all your recall reminders.”
Leading on improving access efforts. Participants saw the GPM as the central staff member responsible for providing oversight of any activities and people involved in improving access. “I ensure everybody is doing what they’re supposed to do,” one GPM reported. When the GPM sees areas that are not being addressed, the individual tries to develop a process or training to “close those gaps.”
GPMs promoted an awareness of their goals, changes in process, and new tools accompanying the initiative. However, other access initiatives were occurring simultaneously creating confusion for health care providers and patients; thus GPMs found they were managing a wide array of related initiatives.
GPMs have to negotiate with leaders across the VHA facility, many of whom operated at a higher leadership level and had different priorities, to address access problems.
“I’m a lieutenant as a GPM in a clinic, a GPM noted. “How is the lieutenant going to talk to a major or a colonel in the clinic and say your clinic has problems. How[‘s] that lieutenant...going to do that? With people skills!”
Managing expectations about the speed and to what extent a problem could be resolved was an important part of the GPM leadership role. “I see myself as managing expectations both up to the leadership and down to the frontline,” a GPM explained. “I find myself talking to leadership [about] our progress. But at the same time, we have to say, ‘not everything can be fixed overnight.’”
Providing leadership on access-related issues included developing a range of options for addressing patient access problems. One analytics manager recounted how the GPM role led to evaluating how physical space limited efficiency in clinic flow. The first step was identifying possible additional rooms to improve clinic flow. This required working with the space committee to “get someone to look at our overarching space and find someplace else for them to sit” to avoid adding to congestion in the clinic area.
Supporting data analytics.
GPMs routinely immersed themselves in the data to understand access issues. GPMs worked with clinic leaders to identify the underlying causes and various solutions. The GPMs maintained access through administrative scrubbing of the data and finding “smart ways to get patients scheduled,” a GPM explained. “I don’t think our facility would have taken care of as many veterans in the time frame as we did....We’ve cleared over 4,000 consults that were older than 90 days. We have cleared thousands and thousands of weekly reminders.”
GPMs expressed the need for aggregated (ie, dashboard) and standardized information to efficiently address access issues. “I would like to have some more standardization on what’s being reviewed; it seems to change frequently, and so [to] be able to track and trend and have something given to me to review,” one health care provider requested. On the other hand, participants also described a need for decision support tools that would lead to action aligned with best practices. “Instead of a dashboard or something that’s just measuring their performance, it’s more something that they can look at and take action” a national support staff advisor suggested.
Enabling self and staff. GPMs felt they were most effective if they enabled themselves and stakeholders through training and by cultivating relationships and team building. Figure 2 illustrates the various stakeholders GPMs reported engaging with. The GPMS should be building relationships, bridging relationships, developing trust, and then providing a higher level of hands-on management. However, “that doesn’t really happen right now, day to day,” one member of leadership reported.
Key topics in GPM leadership training included both soft skills (change management, culture change, and negotiation skills) and crucial analytic/technical training (understanding each metric and dashboard available, data analytics, and supply/demand balancing techniques). The GPMs not only wanted to understand metrics as part of their training, but also want to know what to do about them.
An “operationalization” training approach (discerning the meaning of data, data-based decision making, and determining action from multiple options) inspired by real-life situations was preferred by participants. Other effective learning structures included job aids in the form of templated Gantt charts, process maps to guide GPMs through implementation of new processes, formalized peer learning (accumulated field insights shared during training courses), and informal peer sharing of direct experiences during calls.
GPMs also emphasized training for frontline clinical and support staff, including schedulers. VHA schedulers typically have less education and experience higher turnover rates than do other clinic staff, yet they carry out complex and critical tasks. Providing training and ensuring that any materials developed for training and education were appropriate to the level of education of schedulers was an important task for GPMs. “If they don’t understand all of the scheduling principles and potential,” one GPM explained, “we will not be maximizing the utilization of our parts.”
GPMs also provided informal education to clinicians. Participants noted GPMs have to avoid appearing to overstep their positions or presuming more knowledge and expertise than clinicians. They “have to be able to teach a physician without being overbearing, in a way a physician will accept it as advisement,” one program leader reported.
GPM knowledge, skills, and abilities. GPMs presented a complex range of knowledge, skills, and abilities, including clinical, administrative, analytics, and people skills. All interviewees reported that their prior education and experience did not sufficiently train them for the GPM role. GPMs identified a willingness to learn quickly as a critical characteristic. Many GPMs tended to have a formal education in health administration or business (eg, MBAs); others had administrative experience (eg, administrative assistance to executive leadership) or clinical training (eg, physician assistant). Detailed clinical knowledge was not expected, but clinical familiarity was helpful.
Some interviewees also mentioned previous experience and familiarity with the VHA system specifically as an advantage. This was especially true for VA outpatient flows, clinic flows, and understanding what an outpatient is in a VA context. Interviewees noted the importance of GPMs needing to be able to analyze patient demand metrics and underlying data in order to determine supply of providers and then to allocate adequate resources to complement providers. Forecasting skills were referenced as a key point. “They need to be able to be assured that they can recruit more providers if needed,” a national support staff advisor noted.
Given the importance of developing effective relationships, communication skills were mentioned by most participants and underscored as critical to establishing trust between GPMs and others as the initiative was being implemented. Interviewees indicated that relationship building was further enhanced when GPMs possessed the ability to “work with” rather than command clinicians and staff; navigate politics; and were respectful of other people’s knowledge, skills, abilities, and status. “They have to work with the nursing staff and teach them,” a leader described, “so that people understand that we are going to a different place to achieve our primary objectives and goals.”
Issues and Challenges
Participants identified several technical, social, and structural challenges and barriers to successfully implementing the GPM role.
Technical challenges. Recurring themes across all phases of data analytics were GPMs’ capability to challenge data use and use large volumes of information from multiple data sources (entering and accessing data; “drilling down” from summaries; generating reports; and analyzing and interpreting resulting metrics). Interviewees reported that information assessment and analytic support were not consistent. One GPM had a data analyst pulling reports needed to support clinical units while other GPMs trained staff to pull data. Even with support, some GPMs had issues due to limited information technology (IT) skills or access privileges leading to inefficiencies and delays. “Whenever I need anything from a programmer, I have to go through, you know, the IT gods in the sky,” one GPM remarked. “That usually takes a few months or more.”
Social challenges. Instituting the GPM role was a cultural change, and interviewees reported needing to address resistance to CPM model efforts. Resistance to change “is particularly hard in the VA just because it has a unique culture,” one leader noted. “There is a comfort in the legacy way of doing things.” The GPM initiative was introduced during a time when other national level initiatives were being implemented throughout the VHA. Fulfilling requests for information for these initiatives became the responsibility of the GPM and their team, which diverted attention from the mandate to improve access. Furthermore, GPMs were often considered the “change communicators” to clinics putting them in the role of “bad messenger,” which degraded trust and made it difficult to partner with clinicians.
Efforts to work through change management and build relationships included general program awareness presentation to internal stakeholders; including key stakeholders in GPM committees; pre-emptive conversations with unit chiefs; creating awareness of the GPM activities and progress through formal and informal update meetings; and identifying successes regarding access.
Structural challenges. The GPM role did not have direct supervision over clinical and administrative leaders, making it challenging to enact change. GPMs reported that “they do not always have authority over the area that they are being asked to manage,” which made their work difficult, requiring strong negotiation skills and political savvy to affect change. However, as the clinic staff and providers saw how the GPM could support and positively impact their practice, these challenges began to subside.
Discussion
This study provides empirical evidence regarding the implementation of a new access management strategy for health care systems focused on improving timeliness of care. First, the GPM position was seen as critical at each facility, as a single point person, to help local system leaders respond effectively to both national mandates and local context. Second, requiring the GPMs to report to the medical center director or chief of staff was important for integrating access perspectives across service lines and to facilitate a strong GPM role in strategic planning. Third, the intentional flexibility of the access management initiative, beyond the nationally specified aspects of the GPM role, was key for allowing individual sites to adapt to unique local challenges, resources, and population demands. Fourth, the initiative provided GPMs with opportunities to learn important skills and support the acquisition, utilization, and communication of a tremendous range of data toward responsive action.
According to our respondents, the GPM role demands functioning across a broad set of responsibilities; understanding the big picture as well as the complex underlying variables; engaging facility leaders, clinical and administrative staff; and prioritizing competing national and local demands. Consistent with previous findings, effective GPMs must possess a complex set of skills (interpersonal, analytic, and leadership) and the ability to create a supporting team.13
In practice, improving access at individual sites of care (VA medical centers and community-based outpatient clinics) poses significant challenges, especially in the early stages, even with the assistance of a GPM. For example, some respondents reported being overwhelmed by the volume of available data and dashboards, and responding to current requests for data analysis and dissemination sometimes impeded long range planning. Multiple national access-related initiatives and local pressures also generated excessive and potentially conflicting demands. Thus, while the creation of a GPM position seemed to be essential for the pilot sites to improve local access and timeliness to care, success also requires ongoing national and facility-wide communication, education, and support. Ongoing data analysis training and support will be critical to ensuring the sustainability of the position. Last, recruiting GPMs with the needed complex skill set may prove to be challenging, and it will be important to provide resources to identify, attract, and retain well-qualified GPMs.
Limitations and Future Work
This study was based on a small initial sample of pilot sites of varying sizes and, as such, may not reflect the experience of all VHA GPMs. In addition, the use of snowball sampling, while facilitating identification of key stakeholders, may introduce bias in participant sampling. Nonetheless, the results from this study provide findings that informed the national VHA GPM initiative and can inform further studies of practice management roles outside of the VA.
Further study of the VHA GPM implementation and similar roles in other health care systems is needed. As the GPM position is fully implemented across the VHA, large scale evaluation is needed to gain a more representative picture and allow for comparison of the GPM role at various types of facilities (eg, size, rurality, complexity, ranking based on access performance metrics).
Conclusion
Improving access to care is a central goal for health care systems. The incorporation of the GPM role is an innovative approach to improve access management strategies. Early study of prototype sites provided VHA leadership with valuable insights used to influence further rollout of this initiative. Based on our findings, national and local support are important to ongoing success. National access mandates, training, and resources should focus on ensuring sufficient GPM authority, enabling GPMs to use data, and ensuring GPMs engage with frontline clinical and administrative staff to improve veteran access to care.
The Veterans Health Administration (VHA) provides care for 9 million veterans at 1,255 health care sites linked to one of 170 local medical systems.1 Recognizing that providing timely care requires effective access management, the US Congress mandated training of VHA staff to manage and improve access to care but did not provide additional local funds for new positions.2 In response, the VHA created the group practice manager (GPM), a new position responsible for improving clinical practice management and unifying access improvement across leadership levels, professions, and services within each local medical system.
In May 2015, the VHA began hiring and training GPMs to spearhead management of access to services. The US Department of Veterans Affairs (VA) Office of Veteran Access to Care spearheaded GPM training, including face-to-face sessions, national calls, webinars, and educational materials. Five local medical systems were selected by the VA Office of Veteran Access to Care to implement the GPM role to allow for an early evaluation of the program that would inform a subsequent nationwide rollout. Implementation of the GPM role remained in the hands of local medical systems.
Longer wait times are shown to impact patient health.3,4 Open access scheduling and other patient-centered access management interventions have been shown to improve availability of primary care appointments.5 However, little empirical evidence exists regarding the managers who focus on clinic access interventions. While the nonpeer-reviewed literature includes references to such roles, including GPMs, the empirical literature has focused on external practice faciliators,6-8 “mid-level managers,”9 and clinic staff.10 We found no peer-reviewed articles on the needs and experiences of practice managers who are focused on improving access. The purpose of this study was to examine GPM prototype sites to both enhance subsequent nationwide implementation and to advance empirical literature on managing patient access within health care.
Methods
In 2015, the VA identified 5 prototype sites representing diverse geographic locations, size, and complexity for the implementation of the GPM role (Table 1). These sites self-identified as having clinical practice management experience. GPMs attended 4 training sessions between February and August 2015.
Data Collection
Participants from each prototype site included GPMs, national trainers, clinic leaders, and frontline staff. Table 2 includes the roles and sample size. Participants were recruited through purposive sampling followed by snowball sampling until thematic saturation was reached (the point at which subsequent data fail to produce new findings across sites and roles of interest).
Guided by the Consolidated Framework for Implementation Research (CFIR), the research team developed semistructured interview guides tailored to participants’ roles to elicit rich descriptions regarding overall impressions, practice management strategies, goals, activities, relationship to clinic roles, data analytics usage, challenges, barriers, and facilitators.11 These guides included open-ended questions and structured prompts utilizing participant language for follow-up probes to minimize interviewing bias (eAppendix:
Data Analysis
Data were analyzed using iterative deductive and inductive content analysis.12 Deductive content analysis consisted of identifying quotes that fit within preidentified categories (ie, perceptions of national effort, organizational structure for GPM, challenges, facilitators, metrics and tools, and mobilizing access culture) developed by the interdisciplinary research team. Further content analysis entailed open-coding and iteratively revisiting and reconciling codes associated within each preidentified category as new codes emerged. The team analyzed the resulting codes to inductively and iteratively identify and stabilize themes regarding the GPM role: roles and tasks, GPM characteristics, issues, and challenges. Through this process we moved coded data to reconciled descriptions suited to addressing the purposes of this study. Dedoose 7.0.23 software was used for qualitative data management and analysis.
Results
The study identified participants’ overall impressions of the GPM initiative and key themes within 4 major domains regarding implementing the GPM role: roles and tasks (implementing clinic practice management, leading patient access, supporting data analytics, and enabling self and staff); GPM characteristics (familiarity with clinical services, knowledge of VHA systems, ability to analyze patient data, communication skills, and the ability to work with others); and issues, and challenges (technical, social, and structural).
Overall Impressions
Interviewees perceived the GPM initiative as a consolidation of existing distributed responsibilities into one role that directly reported to local top-level management with indirect reporting to national leaders. Many of the sites reported that they had designated or planned to designate a role resembling the GPM prior to the initiative. “There are staff who’ve been doing some of this work all along,” a GPM noted. “We just didn’t have them grouped together. They weren’t necessarily all working in the same type of service under the same type of structure.”
Whether the GPM position was new or not, participants referenced the importance and challenges of engaging the local facility in recognizing the agency associated with the GPM position. According to national support, the staff are trying to get the facility to understand “why the group practice manager is so important… we’ve got to embed that standard position in the system.”
While the GPM was recognized as the hub of access management, respondents recognized that transformation regarding access involved many players. “We have to create [an] orchestrated team inside each facility,” an advisor argued.
Respondents discussed how the initiative allows local facilities to appoint a specific person with a specific title and role who helps facilitate, organize, and legitimize an access focus at their sites. One GPM interviewee noted how the initiative helped refocus some of their previously less centralized efforts. “We’ve always looked at productivity; we’ve always looked at access; we’ve always looked at efficiency. I think the bigger difference is now there are individuals identified in the clinics, as practice managers as well…I interact with them. They interact with individual clinic staff, and it’s more of a group process than a single individual.”
The value of having tools available and being able to track and manage patient care as a specific example of the positive impact of this new role was noted by participants. A GPM noted that many health care providers will be happy to have tools to better manage their services and a process “that flows from a service level all the way up to executive management, where there is a common interest in making those things happen—I think that’s going to be a tremendous help.”
Participants expressed concern that the national GPM rollout would be a one-size-fits-all approach. These respondents emphasized the need to have the flexibility to customize their activities to meet their unique site and patient needs.
GPM Roles and Tasks
Participants described 4 primary roles that the GPM was expected to fill: implementing clinic practice management, leading patient access, supporting data analytics, and enabling self and staff. Some activities overlapped in that they served to support multiple role areas (Figure 1).
Implementing clinic practice management. In the early stages of the initiative, the GPM’s primary role was to prepare the facility to implement a standardized set of clinic practice management (CPM) team processes. Part of standardizing the CPM process was defining the scope and tasks of the GPM, which requires significant planning for the implementation. “My big job is to finalize what we think group practice management is going to look [like] here,” a GPM reported.
Each prototype site had latitude to interpret the GPM initiative in a way that would work in their context within given VHA boundaries and ongoing initiatives. To achieve the high-level vision and purpose, the GPM first had to develop action plans that accounted for the operating environment of the facility. According to one GPM, VA national officials are “constantly” asking for action plans, which required significant time by specific deadlines. “They want an action plan [and to] clean up all your consults, [and to] clean up all your recall reminders.”
Leading on improving access efforts. Participants saw the GPM as the central staff member responsible for providing oversight of any activities and people involved in improving access. “I ensure everybody is doing what they’re supposed to do,” one GPM reported. When the GPM sees areas that are not being addressed, the individual tries to develop a process or training to “close those gaps.”
GPMs promoted an awareness of their goals, changes in process, and new tools accompanying the initiative. However, other access initiatives were occurring simultaneously creating confusion for health care providers and patients; thus GPMs found they were managing a wide array of related initiatives.
GPMs have to negotiate with leaders across the VHA facility, many of whom operated at a higher leadership level and had different priorities, to address access problems.
“I’m a lieutenant as a GPM in a clinic, a GPM noted. “How is the lieutenant going to talk to a major or a colonel in the clinic and say your clinic has problems. How[‘s] that lieutenant...going to do that? With people skills!”
Managing expectations about the speed and to what extent a problem could be resolved was an important part of the GPM leadership role. “I see myself as managing expectations both up to the leadership and down to the frontline,” a GPM explained. “I find myself talking to leadership [about] our progress. But at the same time, we have to say, ‘not everything can be fixed overnight.’”
Providing leadership on access-related issues included developing a range of options for addressing patient access problems. One analytics manager recounted how the GPM role led to evaluating how physical space limited efficiency in clinic flow. The first step was identifying possible additional rooms to improve clinic flow. This required working with the space committee to “get someone to look at our overarching space and find someplace else for them to sit” to avoid adding to congestion in the clinic area.
Supporting data analytics.
GPMs routinely immersed themselves in the data to understand access issues. GPMs worked with clinic leaders to identify the underlying causes and various solutions. The GPMs maintained access through administrative scrubbing of the data and finding “smart ways to get patients scheduled,” a GPM explained. “I don’t think our facility would have taken care of as many veterans in the time frame as we did....We’ve cleared over 4,000 consults that were older than 90 days. We have cleared thousands and thousands of weekly reminders.”
GPMs expressed the need for aggregated (ie, dashboard) and standardized information to efficiently address access issues. “I would like to have some more standardization on what’s being reviewed; it seems to change frequently, and so [to] be able to track and trend and have something given to me to review,” one health care provider requested. On the other hand, participants also described a need for decision support tools that would lead to action aligned with best practices. “Instead of a dashboard or something that’s just measuring their performance, it’s more something that they can look at and take action” a national support staff advisor suggested.
Enabling self and staff. GPMs felt they were most effective if they enabled themselves and stakeholders through training and by cultivating relationships and team building. Figure 2 illustrates the various stakeholders GPMs reported engaging with. The GPMS should be building relationships, bridging relationships, developing trust, and then providing a higher level of hands-on management. However, “that doesn’t really happen right now, day to day,” one member of leadership reported.
Key topics in GPM leadership training included both soft skills (change management, culture change, and negotiation skills) and crucial analytic/technical training (understanding each metric and dashboard available, data analytics, and supply/demand balancing techniques). The GPMs not only wanted to understand metrics as part of their training, but also want to know what to do about them.
An “operationalization” training approach (discerning the meaning of data, data-based decision making, and determining action from multiple options) inspired by real-life situations was preferred by participants. Other effective learning structures included job aids in the form of templated Gantt charts, process maps to guide GPMs through implementation of new processes, formalized peer learning (accumulated field insights shared during training courses), and informal peer sharing of direct experiences during calls.
GPMs also emphasized training for frontline clinical and support staff, including schedulers. VHA schedulers typically have less education and experience higher turnover rates than do other clinic staff, yet they carry out complex and critical tasks. Providing training and ensuring that any materials developed for training and education were appropriate to the level of education of schedulers was an important task for GPMs. “If they don’t understand all of the scheduling principles and potential,” one GPM explained, “we will not be maximizing the utilization of our parts.”
GPMs also provided informal education to clinicians. Participants noted GPMs have to avoid appearing to overstep their positions or presuming more knowledge and expertise than clinicians. They “have to be able to teach a physician without being overbearing, in a way a physician will accept it as advisement,” one program leader reported.
GPM knowledge, skills, and abilities. GPMs presented a complex range of knowledge, skills, and abilities, including clinical, administrative, analytics, and people skills. All interviewees reported that their prior education and experience did not sufficiently train them for the GPM role. GPMs identified a willingness to learn quickly as a critical characteristic. Many GPMs tended to have a formal education in health administration or business (eg, MBAs); others had administrative experience (eg, administrative assistance to executive leadership) or clinical training (eg, physician assistant). Detailed clinical knowledge was not expected, but clinical familiarity was helpful.
Some interviewees also mentioned previous experience and familiarity with the VHA system specifically as an advantage. This was especially true for VA outpatient flows, clinic flows, and understanding what an outpatient is in a VA context. Interviewees noted the importance of GPMs needing to be able to analyze patient demand metrics and underlying data in order to determine supply of providers and then to allocate adequate resources to complement providers. Forecasting skills were referenced as a key point. “They need to be able to be assured that they can recruit more providers if needed,” a national support staff advisor noted.
Given the importance of developing effective relationships, communication skills were mentioned by most participants and underscored as critical to establishing trust between GPMs and others as the initiative was being implemented. Interviewees indicated that relationship building was further enhanced when GPMs possessed the ability to “work with” rather than command clinicians and staff; navigate politics; and were respectful of other people’s knowledge, skills, abilities, and status. “They have to work with the nursing staff and teach them,” a leader described, “so that people understand that we are going to a different place to achieve our primary objectives and goals.”
Issues and Challenges
Participants identified several technical, social, and structural challenges and barriers to successfully implementing the GPM role.
Technical challenges. Recurring themes across all phases of data analytics were GPMs’ capability to challenge data use and use large volumes of information from multiple data sources (entering and accessing data; “drilling down” from summaries; generating reports; and analyzing and interpreting resulting metrics). Interviewees reported that information assessment and analytic support were not consistent. One GPM had a data analyst pulling reports needed to support clinical units while other GPMs trained staff to pull data. Even with support, some GPMs had issues due to limited information technology (IT) skills or access privileges leading to inefficiencies and delays. “Whenever I need anything from a programmer, I have to go through, you know, the IT gods in the sky,” one GPM remarked. “That usually takes a few months or more.”
Social challenges. Instituting the GPM role was a cultural change, and interviewees reported needing to address resistance to CPM model efforts. Resistance to change “is particularly hard in the VA just because it has a unique culture,” one leader noted. “There is a comfort in the legacy way of doing things.” The GPM initiative was introduced during a time when other national level initiatives were being implemented throughout the VHA. Fulfilling requests for information for these initiatives became the responsibility of the GPM and their team, which diverted attention from the mandate to improve access. Furthermore, GPMs were often considered the “change communicators” to clinics putting them in the role of “bad messenger,” which degraded trust and made it difficult to partner with clinicians.
Efforts to work through change management and build relationships included general program awareness presentation to internal stakeholders; including key stakeholders in GPM committees; pre-emptive conversations with unit chiefs; creating awareness of the GPM activities and progress through formal and informal update meetings; and identifying successes regarding access.
Structural challenges. The GPM role did not have direct supervision over clinical and administrative leaders, making it challenging to enact change. GPMs reported that “they do not always have authority over the area that they are being asked to manage,” which made their work difficult, requiring strong negotiation skills and political savvy to affect change. However, as the clinic staff and providers saw how the GPM could support and positively impact their practice, these challenges began to subside.
Discussion
This study provides empirical evidence regarding the implementation of a new access management strategy for health care systems focused on improving timeliness of care. First, the GPM position was seen as critical at each facility, as a single point person, to help local system leaders respond effectively to both national mandates and local context. Second, requiring the GPMs to report to the medical center director or chief of staff was important for integrating access perspectives across service lines and to facilitate a strong GPM role in strategic planning. Third, the intentional flexibility of the access management initiative, beyond the nationally specified aspects of the GPM role, was key for allowing individual sites to adapt to unique local challenges, resources, and population demands. Fourth, the initiative provided GPMs with opportunities to learn important skills and support the acquisition, utilization, and communication of a tremendous range of data toward responsive action.
According to our respondents, the GPM role demands functioning across a broad set of responsibilities; understanding the big picture as well as the complex underlying variables; engaging facility leaders, clinical and administrative staff; and prioritizing competing national and local demands. Consistent with previous findings, effective GPMs must possess a complex set of skills (interpersonal, analytic, and leadership) and the ability to create a supporting team.13
In practice, improving access at individual sites of care (VA medical centers and community-based outpatient clinics) poses significant challenges, especially in the early stages, even with the assistance of a GPM. For example, some respondents reported being overwhelmed by the volume of available data and dashboards, and responding to current requests for data analysis and dissemination sometimes impeded long range planning. Multiple national access-related initiatives and local pressures also generated excessive and potentially conflicting demands. Thus, while the creation of a GPM position seemed to be essential for the pilot sites to improve local access and timeliness to care, success also requires ongoing national and facility-wide communication, education, and support. Ongoing data analysis training and support will be critical to ensuring the sustainability of the position. Last, recruiting GPMs with the needed complex skill set may prove to be challenging, and it will be important to provide resources to identify, attract, and retain well-qualified GPMs.
Limitations and Future Work
This study was based on a small initial sample of pilot sites of varying sizes and, as such, may not reflect the experience of all VHA GPMs. In addition, the use of snowball sampling, while facilitating identification of key stakeholders, may introduce bias in participant sampling. Nonetheless, the results from this study provide findings that informed the national VHA GPM initiative and can inform further studies of practice management roles outside of the VA.
Further study of the VHA GPM implementation and similar roles in other health care systems is needed. As the GPM position is fully implemented across the VHA, large scale evaluation is needed to gain a more representative picture and allow for comparison of the GPM role at various types of facilities (eg, size, rurality, complexity, ranking based on access performance metrics).
Conclusion
Improving access to care is a central goal for health care systems. The incorporation of the GPM role is an innovative approach to improve access management strategies. Early study of prototype sites provided VHA leadership with valuable insights used to influence further rollout of this initiative. Based on our findings, national and local support are important to ongoing success. National access mandates, training, and resources should focus on ensuring sufficient GPM authority, enabling GPMs to use data, and ensuring GPMs engage with frontline clinical and administrative staff to improve veteran access to care.
1. US Department of Veterans Affairs. Veterans Health Administration. https://www.va.gov/health. Updated October 25, 2019. Accessed January 8, 2020.
2. Veterans Access, Choice, and Accountability Act of 2014. 38 CFR § 17.1500 (2014).
3. Fahmy N, Aprikian A, Al-Otaibi M, et al. Impact of treatment delay in patients with bladder cancer managed with partial cystectomy in Quebec: a population-based study. Can Urol Assoc J. 2009;3(2):131-135.
4. Hill CJ, Joonas K. The impact of unacceptable wait time on health care patients’ attitudes and actions. Health Mark Q. 2005;23(2):69-87.
5. Ansell D, Crispo JAG, Simard B, Bjerre LM. Interventions to reduce wait times for primary care appointments: a systematic review. BMC Health Serv Res. 2017;17(1):295.
6. Kotecha J, Han H, Green M, Russell G, Martin MI, Birtwhistle R. The role of the practice facilitators in Ontario primary healthcare quality improvement. BMC Fam Pract. 2015;16:93.
7. Taylor EF, Machta RM, Meyers DS, Genevro J, Peikes DN. Enhancing the primary care team to provide redesigned care: the roles of practice facilitators and care managers. Ann Fam Med. 2013;11(1):80-83.
8. Liddy C, Laferriere D, Baskerville B, Dahrouge S, Knox L, Hogg W. An overview of practice facilitation programs in Canada: current perspectives and future directions. Healthc Policy. 2013;8(3):58-67.
9. Birken SA, Lee SY, Weiner BJ, Chin MH, Schaefer CT. Improving the effectiveness of health care innovation implementation: middle managers as change agents. Med Care Res Rev. 2013;70(1):29-45.
10. Ahluwalia S, Offredy M. A qualitative study of the impact of the implementation of advanced access in primary healthcare on the working lives of general practice staff. BMC Fam Pract. 2005;6:39.
11. Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 2009;4:50.
12. Elo S, Kyngäs H. The qualitative content analysis process. J Adv Nurs. 2008;62(1):107-115.
13. Stefl ME. Common competencies for all healthcare managers: the Healthcare Leadership Alliance model. J Healthc Manag. 2008;53(6):360-374.
1. US Department of Veterans Affairs. Veterans Health Administration. https://www.va.gov/health. Updated October 25, 2019. Accessed January 8, 2020.
2. Veterans Access, Choice, and Accountability Act of 2014. 38 CFR § 17.1500 (2014).
3. Fahmy N, Aprikian A, Al-Otaibi M, et al. Impact of treatment delay in patients with bladder cancer managed with partial cystectomy in Quebec: a population-based study. Can Urol Assoc J. 2009;3(2):131-135.
4. Hill CJ, Joonas K. The impact of unacceptable wait time on health care patients’ attitudes and actions. Health Mark Q. 2005;23(2):69-87.
5. Ansell D, Crispo JAG, Simard B, Bjerre LM. Interventions to reduce wait times for primary care appointments: a systematic review. BMC Health Serv Res. 2017;17(1):295.
6. Kotecha J, Han H, Green M, Russell G, Martin MI, Birtwhistle R. The role of the practice facilitators in Ontario primary healthcare quality improvement. BMC Fam Pract. 2015;16:93.
7. Taylor EF, Machta RM, Meyers DS, Genevro J, Peikes DN. Enhancing the primary care team to provide redesigned care: the roles of practice facilitators and care managers. Ann Fam Med. 2013;11(1):80-83.
8. Liddy C, Laferriere D, Baskerville B, Dahrouge S, Knox L, Hogg W. An overview of practice facilitation programs in Canada: current perspectives and future directions. Healthc Policy. 2013;8(3):58-67.
9. Birken SA, Lee SY, Weiner BJ, Chin MH, Schaefer CT. Improving the effectiveness of health care innovation implementation: middle managers as change agents. Med Care Res Rev. 2013;70(1):29-45.
10. Ahluwalia S, Offredy M. A qualitative study of the impact of the implementation of advanced access in primary healthcare on the working lives of general practice staff. BMC Fam Pract. 2005;6:39.
11. Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 2009;4:50.
12. Elo S, Kyngäs H. The qualitative content analysis process. J Adv Nurs. 2008;62(1):107-115.
13. Stefl ME. Common competencies for all healthcare managers: the Healthcare Leadership Alliance model. J Healthc Manag. 2008;53(6):360-374.
Delayed hospital admission after hip fracture raises mortality risk
a retrospective, observational study suggests.
Among 867 elderly patients who underwent hip fracture surgery at a university hospital in China and who were available for follow-up, the proportion hospitalized on the day of injury was 25.4%, and the proportion hospitalized on days 1, 2, and 7 after injury were 54.7%, 66.3%, and 12.6%, respectively, reported Wei He, MD, of the Second Affiliated Hospital of Zhejiang University, Hangzhou, China, and colleagues in the World Journal of Emergency Medicine.
The mean time from admission to surgery was 5.2 days. Mortality rates at 1 year, 3 months, and 1 month after surgery were 10.5%, 5.4%, and 3.3%, respectively. Hospitalization at 7 or more days after injury was an independent risk factor for 1-year mortality (odds ratio, 1.76), the authors found.
Although the influence of surgical delay on mortality and morbidity among hip fracture patients has been widely studied, most data focus on surgery timing among hospitalized patients and fail to consider preadmission waiting time, they noted.
The current study aimed to assess outcomes based on “actual preadmission waiting time” through an analysis of data and surgical outcomes from a hospital electronic medical record system and from postoperative telephone interviews. Study subjects were patients aged over 65 years who underwent hip fracture surgery between Jan. 1, 2014, and Dec. 31, 2017. The mean age was 81.4 years, 74.7% of the patients were women, 67.1% had femoral neck fracture, and 56.1% had hip replacement surgery.
The findings, though limited by the retrospective nature of the study and the single-center design, suggest that, under the current conditions in China, admission delay may increase 1-year mortality, they wrote, concluding that “[i]n addition to early surgery highlighted in the guidelines, we also advocate early admission.”
The authors reported having no disclosures.
SOURCE: He W et al. World J Emerg Med. 2020;11(1):27-32.
a retrospective, observational study suggests.
Among 867 elderly patients who underwent hip fracture surgery at a university hospital in China and who were available for follow-up, the proportion hospitalized on the day of injury was 25.4%, and the proportion hospitalized on days 1, 2, and 7 after injury were 54.7%, 66.3%, and 12.6%, respectively, reported Wei He, MD, of the Second Affiliated Hospital of Zhejiang University, Hangzhou, China, and colleagues in the World Journal of Emergency Medicine.
The mean time from admission to surgery was 5.2 days. Mortality rates at 1 year, 3 months, and 1 month after surgery were 10.5%, 5.4%, and 3.3%, respectively. Hospitalization at 7 or more days after injury was an independent risk factor for 1-year mortality (odds ratio, 1.76), the authors found.
Although the influence of surgical delay on mortality and morbidity among hip fracture patients has been widely studied, most data focus on surgery timing among hospitalized patients and fail to consider preadmission waiting time, they noted.
The current study aimed to assess outcomes based on “actual preadmission waiting time” through an analysis of data and surgical outcomes from a hospital electronic medical record system and from postoperative telephone interviews. Study subjects were patients aged over 65 years who underwent hip fracture surgery between Jan. 1, 2014, and Dec. 31, 2017. The mean age was 81.4 years, 74.7% of the patients were women, 67.1% had femoral neck fracture, and 56.1% had hip replacement surgery.
The findings, though limited by the retrospective nature of the study and the single-center design, suggest that, under the current conditions in China, admission delay may increase 1-year mortality, they wrote, concluding that “[i]n addition to early surgery highlighted in the guidelines, we also advocate early admission.”
The authors reported having no disclosures.
SOURCE: He W et al. World J Emerg Med. 2020;11(1):27-32.
a retrospective, observational study suggests.
Among 867 elderly patients who underwent hip fracture surgery at a university hospital in China and who were available for follow-up, the proportion hospitalized on the day of injury was 25.4%, and the proportion hospitalized on days 1, 2, and 7 after injury were 54.7%, 66.3%, and 12.6%, respectively, reported Wei He, MD, of the Second Affiliated Hospital of Zhejiang University, Hangzhou, China, and colleagues in the World Journal of Emergency Medicine.
The mean time from admission to surgery was 5.2 days. Mortality rates at 1 year, 3 months, and 1 month after surgery were 10.5%, 5.4%, and 3.3%, respectively. Hospitalization at 7 or more days after injury was an independent risk factor for 1-year mortality (odds ratio, 1.76), the authors found.
Although the influence of surgical delay on mortality and morbidity among hip fracture patients has been widely studied, most data focus on surgery timing among hospitalized patients and fail to consider preadmission waiting time, they noted.
The current study aimed to assess outcomes based on “actual preadmission waiting time” through an analysis of data and surgical outcomes from a hospital electronic medical record system and from postoperative telephone interviews. Study subjects were patients aged over 65 years who underwent hip fracture surgery between Jan. 1, 2014, and Dec. 31, 2017. The mean age was 81.4 years, 74.7% of the patients were women, 67.1% had femoral neck fracture, and 56.1% had hip replacement surgery.
The findings, though limited by the retrospective nature of the study and the single-center design, suggest that, under the current conditions in China, admission delay may increase 1-year mortality, they wrote, concluding that “[i]n addition to early surgery highlighted in the guidelines, we also advocate early admission.”
The authors reported having no disclosures.
SOURCE: He W et al. World J Emerg Med. 2020;11(1):27-32.
FROM THE WORLD JOURNAL OF EMERGENCY MEDICINE
SimLEARN Musculoskeletal Training for VHA Primary Care Providers and Health Professions Educators
Diseases of the musculoskeletal (MSK) system are common, accounting for some of the most frequent visits to primary care clinics.1-3 In addition, care for patients with chronic MSK diseases represents a substantial economic burden.4-6
In response to this clinical training need, the Veterans Health Administration (VHA) developed a portfolio of educational experiences for VHA health care providers and trainees, including both the Salt Lake City and National MSK “mini-residencies.”17-19 These programs have educated more than 800 individuals. Early observations show a progressive increase in the number of joint injections performed at participant’s VHA clinics as well as a reduction in unnecessary magnetic resonance imaging orders of the knee.20,21 These findings may be interpreted as markers for improved access to care for veterans as well as cost savings for the health care system.
The success of these early initiatives was recognized by the medical leadership of the VHA Simulation Learning, Education and Research Network (SimLEARN), who requested the Mini-Residency course directors to implement a similar educational program at the National Simulation Center in Orlando, Florida. SimLEARN was created to promote best practices in learning and education and provides a high-tech immersive environment for the development and delivery of simulation-based training curricula to facilitate workforce development.22 This article describes the initial experience of the VHA SimLEARN MSK continuing professional development (CPD) training programs, including curriculum design and educational impact on early learners, and how this informed additional CPD needs to continue advancing MSK education and care.
Methods
The initial vision was inspired by the national MSK Mini-Residency initiative for PCPs, which involved 13 US Department of Veterans Affairs (VA) medical centers; its development, dissemination, and validity evidence for assessment methods have been previously described.17,18,23 SimLEARN leadership attended a Mini-Residency, observing the educational experience and identifying learning objectives most aligned with national goals. The director and codirector of the MSK Mini-Residency (MJB, AMB) then worked with SimLEARN using its educational platform and train-the-trainer model to create a condensed 2-day course, centered on primary care evaluation and management of shoulder and knee pain. The course also included elements supporting educational leaders in providing similar trainings at their local facility (Table 1).
Curriculum was introduced through didactics and reinforced in hands-on sessions enhanced by peer-teaching, arthrocentesis task trainers, and simulated patient experiences. At the end of day 1, participants engaged in critical reflection, reviewing knowledge and skills they had acquired.
On day 2, each participant was evaluated using an observed structured clinical examination (OSCE) for the shoulder, followed by an observed structured teaching experience (OSTE). Given the complexity of the physical examination and the greater potential for appropriate interpretation of clinical findings to influence best practice care, the shoulder was emphasized for these experiences. Time constraints of a 2-day program based on SimLEARN format requirements prevented including an additional OSCE for the knee. At the conclusion of the course, faculty and participants discussed strategies for bringing this educational experience to learners at their local facilities as well as for avoiding potential barriers to implementation. The course was accredited through the VHA Employee Education System (EES), and participants received 16 hours of CPD credit.
Participants
Opportunity to attend was communicated through national, regional, and local VHA organizational networks. Participants self-registered online through the VHA Talent Management System, the main learning resource for VHA employee education, and registration was open to both PCPs and clinician educators. Class size was limited to 10 to facilitate detailed faculty observation during skill acquisition experiences, simulations, and assessment exercises.
Program Evaluation
A standard process for evaluating and measuring learning objectives was performed through VHA EES. Self-assessment surveys and OSCEs were used to assess the activity.
Self-assessment surveys were administered at the beginning and end of the program. Content was adapted from that used in the national MSK Mini-Residency initiative and revised by experts in survey design.18,24,25 Pre- and postcourse surveys asked participants to rate how important it was for them to be competent in evaluating shoulder and knee pain and in performing related joint injections, as well as to rate their level of confidence in their ability to evaluate and manage these conditions. The survey used 5 construct-specific response options distributed equally on a visual scale. Participants’ learning goals were collected on the precourse survey.
Participants’ competence in performing and interpreting a systematic and thorough physical examination of the shoulder and in suggesting a reasonable plan of management were assessed using a single-station OSCE. This tool, which presented learners with a simulated case depicting rotator cuff pathology, has been described in multiple educational settings, and validity evidence supporting its use has been published.18,19,23 Course faculty conducted the OSCE, one as the simulated patient, the other as the rater. Immediately following the examination, both faculty conducted a debriefing session with each participant. The OSCE was scored using the validated checklist for specific elements of the shoulder exam, followed by a structured sequence of questions exploring participants’ interpretation of findings, diagnostic impressions, and recommendations for initial management. Scores for participants’ differential diagnosis were based on the completeness and specificity of diagnoses given; scores for management plans were based on appropriateness and accuracy of both the primary and secondary approach to treatment or further diagnostic efforts. A global rating (range 1 to 9) was assigned, independent of scores in other domains.
Following the OSCE, participants rotated through a 3-cycle OSTE where they practiced the roles of simulated patient, learner, and educator. Faculty observed each OSTE and led focused debriefing sessions immediately following each rotation to facilitate participants’ critical reflection of their involvement in these elements of the course. This exercise was formative without quantitative assessment of performance.
Statistical Analysis
Pre- and postsurvey data were analyzed using a paired Student t test. Comparisons between multiple variables (eg, OSCE scores by years of experience or level of credentials) were analyzed using analysis of variance. Relationships between variables were analyzed with a Pearson correlation. All statistical analyses were conducted using IBM SPSS, Version 24 (Armonk, NY).
This project was reviewed by the institutional review board of the University of Utah and the Salt Lake City VA and was determined to be exempt from review because the work did not meet the definition of research with human subjects and was considered a quality improvement study.
Results
Twenty-four participants completed the program over 3 course offerings between February and May 2016, and all completed pre- and postcourse self-assessment surveys (Table 2). Self-ratings of the importance of competence in shoulder and knee MSK skills remained high before and after the course, and confidence improved significantly across all learning objectives. Despite the emphasis on the evaluation and management of shoulder pain, participants’ self-confidence still improved significantly with the knee—though these improvements were generally smaller in scale compared with those of the shoulder.
Overall OSCE scores and scores by domain were not found to be statistically different based on either years of experience or by level of credential or specialty (advanced practice registered nurse/physician assistant, PCP, or specialty care physician)(Table 3). However, there was a trend toward higher performance among the specialty care physician group, and a trend toward lower performance among participants with less than 3 years’ experience.
Discussion
Building on the foundation of other successful innovations in MSK education, the first year of the SimLEARN National MSK Training Program demonstrated the feasibility of a 2-day centralized national course as a method to increase participants’ confidence and competence in evaluating and managing MSK problems, and to disseminate a portable curriculum to a range of clinician educators. Although this course focused on developing competence for shoulder skills, including an OSCE on day 2, self-perceived improvements in participants’ ability to evaluate and manage knee pain were observed. Future program refinement and follow-up of participants’ experience and needs may lead to increased time allocated to the knee exam as well as objective measures of competence for knee skills.
In comparing our findings to the work that others have previously described, we looked for reports of CPD programs in 2 contexts: those that focused on acquisition of MSK skills relevant to clinical practice, and those designed as clinician educator or faculty development initiatives. Although there are few reports of MSK-themed CPD experiences designed specifically for nurses and allied health professionals, a recent effort to survey members of these disciplines in the United Kingdom was an important contribution to a systematic needs assessment.26-28 Increased support from leadership, mostly in terms of time allowance and budgetary support, was identified as an important driver to facilitate participation in MSK CPD experiences. Through SimLEARN, the VHA is investing in CPD, providing the MSK Training Programs and other courses at no cost to its employees.
Most published reports on physician education have not evaluated content knowledge or physical examination skills with measures for which validity evidence has been published.19,29,30 One notable exception is the 2000 Canadian Viscosupplementation Injector Preceptor experience, in which Bellamy and colleagues examined patient outcomes in evaluating their program.31
Our experience is congruent with the work of Macedo and colleagues and Sturpe and colleagues, who described the effectiveness and acceptability of an OSTE for faculty development.32,33 These studies emphasize debriefing, a critical element in faculty development identified by Steinert and colleagues in a 2006 best evidence medical education (BEME) review.34 The shoulder OSTE was one of the most well-received elements of our course, and each debrief was critical to facilitating rich discussions between educators and practitioners playing the role of teacher or student during this simulated experience, gaining insight into each other’s perspectives.
This program has several significant strengths: First, this is the most recent step in the development of a portfolio of innovative MSK CPD programs that were envisioned through a systematic process involving projections of cost-effectiveness, local pilot testing, and national expansion.17,18,35 Second, the SimLEARN program uses assessment tools for which validity evidence has been published, made available for reflective critique by educational scholars.19,23 This supports a national consortium of MSK educators, advancing clinical teaching and educational scholarship, and creating opportunities for interprofessional collaboration in congruence with the vision expressed in the 2010 Institute of Medicine report, “Redesigning Continuing Education in the Health Professions,” as well as the 2016 update of the BEME recommendations for faculty development.36,37
Our experience with the SimLEARN National MSK Training Program demonstrates need for 2 distinct courses: (1) the MSK Clinician—serving PCPs seeking to develop their skills in evaluating and managing patients with MSK problems; and (2), the MSK Master Educator—for those with preexisting content expertise who would value the introduction to a national curriculum and connections with other MSK master educators. Both of these are now offered regularly through SimLEARN for VHA and US Department of Defense employees. The MSK Clinician program establishes competence in systematically evaluating and managing shoulder and knee MSK problems in an educational setting and prepares participants for subsequent clinical experiences where they can perform related procedures if desired, under appropriate supervision. The Master Educator program introduces partici pants to the clinician curriculum and provides the opportunity to develop an individualized plan for implementation of an MSK educational program at their home institutions. Participants are selected through a competitive application process, and funding for travel to attend the Master Educator program is provided by SimLEARN for participants who are accepted. Additionally, the Master Educator program serves as a repository for potential future SimLEARN MSK Clinician course faculty.
Limitations
The small number of participants may limit the validity of our conclusions. Although we included an OSCE to measure competence in performing and interpreting the shoulder exam, the durability of these skills is not known. Periodic postcourse OSCEs could help determine this and refresh and preserve accuracy in the performance of specific maneuvers. Second, although this experience was rated highly by participants, we do not know the impact of the program on their daily work or career trajectory. Sustained follow-up of learners, perhaps developed on the model of the Long-Term Career Outcome Study, may increase the value of this experience for future participants.38 This program appealed to a diverse pool of learners, with a broad range of precourse expertise and varied expectations of how course experiences would impact their future work and career development. Some clinical educator attendees came from tertiary care facilities affiliated with academic medical centers, held specialist or subspecialist credentials, and had formal responsibilities as leaders in HPE. Other clinical practitioner participants were solitary PCPs, often in rural or home-based settings; although they may have been eager to apply new knowledge and skills in patient care, they neither anticipated nor desired any role as an educator.
Conclusion
The initial SimLEARN MSK Training Program provides PCPs and clinician educators with rich learning experiences, increasing confidence in addressing MSK problems and competence in performing and interpreting a systematic physical examination of the shoulder. The success of this program has created new opportunities for practitioners seeking to strengthen clinical skills and for leaders in health professions education looking to disseminate similar trainings and connect with a national group of educators.
Acknowledgments
The authors gratefully acknowledge the faculty and staff at the Veterans Health Administration SimLEARN National Simulation Center, the faculty of the Salt Lake City Musculoskeletal Mini-Residency program, the supportive leadership of the George E. Wahlen Salt Lake City Veterans Affairs Medical Center, and the efforts of Danielle Blake for logistical support and data entry.
1. Helmick CG, Felson DT, Lawrence RC, et al; National Arthritis Data Workgroup. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum. 2008;58(1):15-25.
2. Lawrence RC, Felson DT, Helmick CG, et al; National Arthritis Data Workgroup. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58(1):26-35.
3. Sacks JJ, Luo YH, Helmick CG. Prevalence of specific types of arthritis and other rheumatic conditions in the ambulatory health care system in the United States, 2001-2005. Arthritis Care Res (Hoboken). 2010;62(4):460-464.
4. Gupta S, Hawker GA, Laporte A, Croxford R, Coyte PC. The economic burden of disabling hip and knee osteoarthritis (OA) from the perspective of individuals living with this condition. Rheumatology (Oxford). 2005;44(12):1531-1537.
5. Gore M, Tai KS, Sadosky A, Leslie D, Stacey BR. Clinical comorbidities, treatment patterns, and direct medical costs of patients with osteoarthritis in usual care: a retrospective claims database analysis. J Med Econ. 2011;14(4):497-507.
6. Rabenda V, Manette C, Lemmens R, Mariani AM, Struvay N, Reginster JY. Direct and indirect costs attributable to osteoarthritis in active subjects. J Rheumatol. 2006;33(6):1152-1158.
7. Day CS, Yeh AC. Evidence of educational inadequacies in region-specific musculoskeletal medicine. Clin Orthop Relat Res. 2008;466(10):2542-2547.
8. Glazier RH, Dalby DM, Badley EM, Hawker GA, Bell MJ, Buchbinder R. Determinants of physician confidence in the primary care management of musculoskeletal disorders. J Rheumatol. 1996;23(2):351-356.
9. Haywood BL, Porter SL, Grana WA. Assessment of musculoskeletal knowledge in primary care residents. Am J Orthop (Belle Mead NJ). 2006;35(6):273-275.
10. Monrad SU, Zeller JL, Craig CL, Diponio LA. Musculoskeletal education in US medical schools: lessons from the past and suggestions for the future. Curr Rev Musculoskelet Med. 2011;4(3):91-98.
11. O’Dunn-Orto A, Hartling L, Campbell S, Oswald AE. Teaching musculoskeletal clinical skills to medical trainees and physicians: a Best Evidence in Medical Education systematic review of strategies and their effectiveness: BEME Guide No. 18. Med Teach. 2012;34(2):93-102.
12. Wilcox T, Oyler J, Harada C, Utset T. Musculoskeletal exam and joint injection training for internal medicine residents. J Gen Intern Med. 2006;21(5):521-523.
13. Petron DJ, Greis PE, Aoki SK, et al. Use of knee magnetic resonance imaging by primary care physicians in patients aged 40 years and older. Sports Health. 2010;2(5):385-390.
14. Roberts TT, Singer N, Hushmendy S, et al. MRI for the evaluation of knee pain: comparison of ordering practices of primary care physicians and orthopaedic surgeons. J Bone Joint Surg Am. 2015;97(9):709-714.
15. Wylie JD, Crim JR, Working ZM, Schmidt RL, Burks RT. Physician provider type influences utilization and diagnostic utility of magnetic resonance imaging of the knee. J Bone Joint Surg Am. 2015;97(1):56-62.
16. Smith M, Saunders R, Stuckhardt L, McGinnis JM, eds. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America. Washington, DC; 2013.
17. Battistone MJ, Barker AM, Lawrence P, Grotzke MP, Cannon GW. Mini-residency in musculoskeletal care: an interprofessional, mixed-methods educational initiative for primary care providers. Arthritis Care Res (Hoboken). 2016;68(2):275-279.
18. Battistone MJ, Barker AM, Grotzke MP, Beck JP, Lawrence P, Cannon GW. “Mini-residency” in musculoskeletal care: a national continuing professional development program for primary care providers. J Gen Intern Med. 2016;31(11):1301-1307.
19. Battistone MJ, Barker AM, Grotzke MP, et al. Effectiveness of an interprofessional and multidisciplinary musculoskeletal training program. J Grad Med Educ. 2016;8(3):398-404.
20. Battistone MJ, Barker AM, Lawrence P, Grotzke M, Cannon GW. Two-year impact of a continuing professional education program to train primary care providers to perform arthrocentesis. Presented at: 2017 ACR/ARHP Annual Meeting [Abstract 909]. https://acrabstracts.org/abstract/two-year-impact-of-a-continuing-professional-education-program-to-train-primary-care-providers-to-perform-arthrocentesis. Accessed November 14, 2019.
21. Call MR, Barker AM, Lawrence P, Cannon GW, Battistone MJ. Impact of a musculoskeltal “mini-residency” continuing professional education program on knee mri orders by primary care providers. Presented at: 2015 ACR/ARHP Annual Meeting [Abstract 1011]. https://acrabstracts.org/abstract/impact-of-a-musculoskeletal-aeoemini-residencyae%ef%bf%bd-continuing-professional-education-program-on-knee-mri-orders-by-primary-care-providers. Accessed November 14, 2019.
22. US Department of Veterans Affairs. VHA SimLEARN. https://www.simlearn.va.gov/SIMLEARN/about_us.asp. Updated January 24, 2019. Accessed November 13, 2019.
23. Battistone MJ, Barker AM, Beck JP, Tashjian RZ, Cannon GW. Validity evidence for two objective structured clinical examination stations to evaluate core skills of the shoulder and knee assessment. BMC Med Educ. 2017;17(1):13.
24. Artino AR Jr, La Rochelle JS, Dezee KJ, Gehlbach H. Developing questionnaires for educational research: AMEE Guide No. 87. Med Teach. 2014;36(6):463-474.
25. Gehlbach H, Artino AR Jr. The survey checklist (Manifesto). Acad Med. 2018;93(3):360-366.
26. Haywood H, Pain H, Ryan S, Adams J. The continuing professional development for nurses and allied health professionals working within musculoskeletal services: a national UK survey. Musculoskeletal Care. 2013;11(2):63-70.
27. Haywood H, Pain H, Ryan S, Adams J. Continuing professional development: issues raised by nurses and allied health professionals working in musculoskeletal settings. Musculoskeletal Care. 2013;11(3):136-144.
28. Warburton L. Continuing professional development in musculoskeletal domains. Musculoskeletal Care. 2012;10(3):125-126.
29. Stansfield RB, Diponio L, Craig C, et al. Assessing musculoskeletal examination skills and diagnostic reasoning of 4th year medical students using a novel objective structured clinical exam. BMC Med Educ. 2016;16(1):268.
30. Hose MK, Fontanesi J, Woytowitz M, Jarrin D, Quan A. Competency based clinical shoulder examination training improves physical exam, confidence, and knowledge in common shoulder conditions. J Gen Intern Med. 2017;32(11):1261-1265.
31. Bellamy N, Goldstein LD, Tekanoff RA. Continuing medical education-driven skills acquisition and impact on improved patient outcomes in family practice setting. J Contin Educ Health Prof. 2000;20(1):52-61.
32. Macedo L, Sturpe DA, Haines ST, Layson-Wolf C, Tofade TS, McPherson ML. An objective structured teaching exercise (OSTE) for preceptor development. Curr Pharm Teach Learn. 2015;7(5):627-634.
33. Sturpe DA, Schaivone KA. A primer for objective structured teaching exercises. Am J Pharm Educ. 2014;78(5):104.
34. Steinert Y, Mann K, Centeno A, et al. A systematic review of faculty development initiatives designed to improve teaching effectiveness in medical education: BEME Guide No. 8. Med Teach. 2006;28(6):497-526.
35. Nelson SD, Nelson RE, Cannon GW, et al. Cost-effectiveness of training rural providers to identify and treat patients at risk for fragility fractures. Osteoporos Int. 2014;25(12):2701-2707.
36. Steinert Y, Mann K, Anderson B, et al. A systematic review of faculty development initiatives designed to enhance teaching effectiveness: A 10-year update: BEME Guide No. 40. Med Teach. 2016;38(8):769-786.
37. Institute of Medicine. Redesigning Continuing Education in the Health Professions. Washington, DC: National Academies Press; 2010.
38. Durning SJ, Dong T, LaRochelle JL, et al. The long-term career outcome study: lessons learned and implications for educational practice. Mil Med. 2015;180(suppl 4):164-170.
Diseases of the musculoskeletal (MSK) system are common, accounting for some of the most frequent visits to primary care clinics.1-3 In addition, care for patients with chronic MSK diseases represents a substantial economic burden.4-6
In response to this clinical training need, the Veterans Health Administration (VHA) developed a portfolio of educational experiences for VHA health care providers and trainees, including both the Salt Lake City and National MSK “mini-residencies.”17-19 These programs have educated more than 800 individuals. Early observations show a progressive increase in the number of joint injections performed at participant’s VHA clinics as well as a reduction in unnecessary magnetic resonance imaging orders of the knee.20,21 These findings may be interpreted as markers for improved access to care for veterans as well as cost savings for the health care system.
The success of these early initiatives was recognized by the medical leadership of the VHA Simulation Learning, Education and Research Network (SimLEARN), who requested the Mini-Residency course directors to implement a similar educational program at the National Simulation Center in Orlando, Florida. SimLEARN was created to promote best practices in learning and education and provides a high-tech immersive environment for the development and delivery of simulation-based training curricula to facilitate workforce development.22 This article describes the initial experience of the VHA SimLEARN MSK continuing professional development (CPD) training programs, including curriculum design and educational impact on early learners, and how this informed additional CPD needs to continue advancing MSK education and care.
Methods
The initial vision was inspired by the national MSK Mini-Residency initiative for PCPs, which involved 13 US Department of Veterans Affairs (VA) medical centers; its development, dissemination, and validity evidence for assessment methods have been previously described.17,18,23 SimLEARN leadership attended a Mini-Residency, observing the educational experience and identifying learning objectives most aligned with national goals. The director and codirector of the MSK Mini-Residency (MJB, AMB) then worked with SimLEARN using its educational platform and train-the-trainer model to create a condensed 2-day course, centered on primary care evaluation and management of shoulder and knee pain. The course also included elements supporting educational leaders in providing similar trainings at their local facility (Table 1).
Curriculum was introduced through didactics and reinforced in hands-on sessions enhanced by peer-teaching, arthrocentesis task trainers, and simulated patient experiences. At the end of day 1, participants engaged in critical reflection, reviewing knowledge and skills they had acquired.
On day 2, each participant was evaluated using an observed structured clinical examination (OSCE) for the shoulder, followed by an observed structured teaching experience (OSTE). Given the complexity of the physical examination and the greater potential for appropriate interpretation of clinical findings to influence best practice care, the shoulder was emphasized for these experiences. Time constraints of a 2-day program based on SimLEARN format requirements prevented including an additional OSCE for the knee. At the conclusion of the course, faculty and participants discussed strategies for bringing this educational experience to learners at their local facilities as well as for avoiding potential barriers to implementation. The course was accredited through the VHA Employee Education System (EES), and participants received 16 hours of CPD credit.
Participants
Opportunity to attend was communicated through national, regional, and local VHA organizational networks. Participants self-registered online through the VHA Talent Management System, the main learning resource for VHA employee education, and registration was open to both PCPs and clinician educators. Class size was limited to 10 to facilitate detailed faculty observation during skill acquisition experiences, simulations, and assessment exercises.
Program Evaluation
A standard process for evaluating and measuring learning objectives was performed through VHA EES. Self-assessment surveys and OSCEs were used to assess the activity.
Self-assessment surveys were administered at the beginning and end of the program. Content was adapted from that used in the national MSK Mini-Residency initiative and revised by experts in survey design.18,24,25 Pre- and postcourse surveys asked participants to rate how important it was for them to be competent in evaluating shoulder and knee pain and in performing related joint injections, as well as to rate their level of confidence in their ability to evaluate and manage these conditions. The survey used 5 construct-specific response options distributed equally on a visual scale. Participants’ learning goals were collected on the precourse survey.
Participants’ competence in performing and interpreting a systematic and thorough physical examination of the shoulder and in suggesting a reasonable plan of management were assessed using a single-station OSCE. This tool, which presented learners with a simulated case depicting rotator cuff pathology, has been described in multiple educational settings, and validity evidence supporting its use has been published.18,19,23 Course faculty conducted the OSCE, one as the simulated patient, the other as the rater. Immediately following the examination, both faculty conducted a debriefing session with each participant. The OSCE was scored using the validated checklist for specific elements of the shoulder exam, followed by a structured sequence of questions exploring participants’ interpretation of findings, diagnostic impressions, and recommendations for initial management. Scores for participants’ differential diagnosis were based on the completeness and specificity of diagnoses given; scores for management plans were based on appropriateness and accuracy of both the primary and secondary approach to treatment or further diagnostic efforts. A global rating (range 1 to 9) was assigned, independent of scores in other domains.
Following the OSCE, participants rotated through a 3-cycle OSTE where they practiced the roles of simulated patient, learner, and educator. Faculty observed each OSTE and led focused debriefing sessions immediately following each rotation to facilitate participants’ critical reflection of their involvement in these elements of the course. This exercise was formative without quantitative assessment of performance.
Statistical Analysis
Pre- and postsurvey data were analyzed using a paired Student t test. Comparisons between multiple variables (eg, OSCE scores by years of experience or level of credentials) were analyzed using analysis of variance. Relationships between variables were analyzed with a Pearson correlation. All statistical analyses were conducted using IBM SPSS, Version 24 (Armonk, NY).
This project was reviewed by the institutional review board of the University of Utah and the Salt Lake City VA and was determined to be exempt from review because the work did not meet the definition of research with human subjects and was considered a quality improvement study.
Results
Twenty-four participants completed the program over 3 course offerings between February and May 2016, and all completed pre- and postcourse self-assessment surveys (Table 2). Self-ratings of the importance of competence in shoulder and knee MSK skills remained high before and after the course, and confidence improved significantly across all learning objectives. Despite the emphasis on the evaluation and management of shoulder pain, participants’ self-confidence still improved significantly with the knee—though these improvements were generally smaller in scale compared with those of the shoulder.
Overall OSCE scores and scores by domain were not found to be statistically different based on either years of experience or by level of credential or specialty (advanced practice registered nurse/physician assistant, PCP, or specialty care physician)(Table 3). However, there was a trend toward higher performance among the specialty care physician group, and a trend toward lower performance among participants with less than 3 years’ experience.
Discussion
Building on the foundation of other successful innovations in MSK education, the first year of the SimLEARN National MSK Training Program demonstrated the feasibility of a 2-day centralized national course as a method to increase participants’ confidence and competence in evaluating and managing MSK problems, and to disseminate a portable curriculum to a range of clinician educators. Although this course focused on developing competence for shoulder skills, including an OSCE on day 2, self-perceived improvements in participants’ ability to evaluate and manage knee pain were observed. Future program refinement and follow-up of participants’ experience and needs may lead to increased time allocated to the knee exam as well as objective measures of competence for knee skills.
In comparing our findings to the work that others have previously described, we looked for reports of CPD programs in 2 contexts: those that focused on acquisition of MSK skills relevant to clinical practice, and those designed as clinician educator or faculty development initiatives. Although there are few reports of MSK-themed CPD experiences designed specifically for nurses and allied health professionals, a recent effort to survey members of these disciplines in the United Kingdom was an important contribution to a systematic needs assessment.26-28 Increased support from leadership, mostly in terms of time allowance and budgetary support, was identified as an important driver to facilitate participation in MSK CPD experiences. Through SimLEARN, the VHA is investing in CPD, providing the MSK Training Programs and other courses at no cost to its employees.
Most published reports on physician education have not evaluated content knowledge or physical examination skills with measures for which validity evidence has been published.19,29,30 One notable exception is the 2000 Canadian Viscosupplementation Injector Preceptor experience, in which Bellamy and colleagues examined patient outcomes in evaluating their program.31
Our experience is congruent with the work of Macedo and colleagues and Sturpe and colleagues, who described the effectiveness and acceptability of an OSTE for faculty development.32,33 These studies emphasize debriefing, a critical element in faculty development identified by Steinert and colleagues in a 2006 best evidence medical education (BEME) review.34 The shoulder OSTE was one of the most well-received elements of our course, and each debrief was critical to facilitating rich discussions between educators and practitioners playing the role of teacher or student during this simulated experience, gaining insight into each other’s perspectives.
This program has several significant strengths: First, this is the most recent step in the development of a portfolio of innovative MSK CPD programs that were envisioned through a systematic process involving projections of cost-effectiveness, local pilot testing, and national expansion.17,18,35 Second, the SimLEARN program uses assessment tools for which validity evidence has been published, made available for reflective critique by educational scholars.19,23 This supports a national consortium of MSK educators, advancing clinical teaching and educational scholarship, and creating opportunities for interprofessional collaboration in congruence with the vision expressed in the 2010 Institute of Medicine report, “Redesigning Continuing Education in the Health Professions,” as well as the 2016 update of the BEME recommendations for faculty development.36,37
Our experience with the SimLEARN National MSK Training Program demonstrates need for 2 distinct courses: (1) the MSK Clinician—serving PCPs seeking to develop their skills in evaluating and managing patients with MSK problems; and (2), the MSK Master Educator—for those with preexisting content expertise who would value the introduction to a national curriculum and connections with other MSK master educators. Both of these are now offered regularly through SimLEARN for VHA and US Department of Defense employees. The MSK Clinician program establishes competence in systematically evaluating and managing shoulder and knee MSK problems in an educational setting and prepares participants for subsequent clinical experiences where they can perform related procedures if desired, under appropriate supervision. The Master Educator program introduces partici pants to the clinician curriculum and provides the opportunity to develop an individualized plan for implementation of an MSK educational program at their home institutions. Participants are selected through a competitive application process, and funding for travel to attend the Master Educator program is provided by SimLEARN for participants who are accepted. Additionally, the Master Educator program serves as a repository for potential future SimLEARN MSK Clinician course faculty.
Limitations
The small number of participants may limit the validity of our conclusions. Although we included an OSCE to measure competence in performing and interpreting the shoulder exam, the durability of these skills is not known. Periodic postcourse OSCEs could help determine this and refresh and preserve accuracy in the performance of specific maneuvers. Second, although this experience was rated highly by participants, we do not know the impact of the program on their daily work or career trajectory. Sustained follow-up of learners, perhaps developed on the model of the Long-Term Career Outcome Study, may increase the value of this experience for future participants.38 This program appealed to a diverse pool of learners, with a broad range of precourse expertise and varied expectations of how course experiences would impact their future work and career development. Some clinical educator attendees came from tertiary care facilities affiliated with academic medical centers, held specialist or subspecialist credentials, and had formal responsibilities as leaders in HPE. Other clinical practitioner participants were solitary PCPs, often in rural or home-based settings; although they may have been eager to apply new knowledge and skills in patient care, they neither anticipated nor desired any role as an educator.
Conclusion
The initial SimLEARN MSK Training Program provides PCPs and clinician educators with rich learning experiences, increasing confidence in addressing MSK problems and competence in performing and interpreting a systematic physical examination of the shoulder. The success of this program has created new opportunities for practitioners seeking to strengthen clinical skills and for leaders in health professions education looking to disseminate similar trainings and connect with a national group of educators.
Acknowledgments
The authors gratefully acknowledge the faculty and staff at the Veterans Health Administration SimLEARN National Simulation Center, the faculty of the Salt Lake City Musculoskeletal Mini-Residency program, the supportive leadership of the George E. Wahlen Salt Lake City Veterans Affairs Medical Center, and the efforts of Danielle Blake for logistical support and data entry.
Diseases of the musculoskeletal (MSK) system are common, accounting for some of the most frequent visits to primary care clinics.1-3 In addition, care for patients with chronic MSK diseases represents a substantial economic burden.4-6
In response to this clinical training need, the Veterans Health Administration (VHA) developed a portfolio of educational experiences for VHA health care providers and trainees, including both the Salt Lake City and National MSK “mini-residencies.”17-19 These programs have educated more than 800 individuals. Early observations show a progressive increase in the number of joint injections performed at participant’s VHA clinics as well as a reduction in unnecessary magnetic resonance imaging orders of the knee.20,21 These findings may be interpreted as markers for improved access to care for veterans as well as cost savings for the health care system.
The success of these early initiatives was recognized by the medical leadership of the VHA Simulation Learning, Education and Research Network (SimLEARN), who requested the Mini-Residency course directors to implement a similar educational program at the National Simulation Center in Orlando, Florida. SimLEARN was created to promote best practices in learning and education and provides a high-tech immersive environment for the development and delivery of simulation-based training curricula to facilitate workforce development.22 This article describes the initial experience of the VHA SimLEARN MSK continuing professional development (CPD) training programs, including curriculum design and educational impact on early learners, and how this informed additional CPD needs to continue advancing MSK education and care.
Methods
The initial vision was inspired by the national MSK Mini-Residency initiative for PCPs, which involved 13 US Department of Veterans Affairs (VA) medical centers; its development, dissemination, and validity evidence for assessment methods have been previously described.17,18,23 SimLEARN leadership attended a Mini-Residency, observing the educational experience and identifying learning objectives most aligned with national goals. The director and codirector of the MSK Mini-Residency (MJB, AMB) then worked with SimLEARN using its educational platform and train-the-trainer model to create a condensed 2-day course, centered on primary care evaluation and management of shoulder and knee pain. The course also included elements supporting educational leaders in providing similar trainings at their local facility (Table 1).
Curriculum was introduced through didactics and reinforced in hands-on sessions enhanced by peer-teaching, arthrocentesis task trainers, and simulated patient experiences. At the end of day 1, participants engaged in critical reflection, reviewing knowledge and skills they had acquired.
On day 2, each participant was evaluated using an observed structured clinical examination (OSCE) for the shoulder, followed by an observed structured teaching experience (OSTE). Given the complexity of the physical examination and the greater potential for appropriate interpretation of clinical findings to influence best practice care, the shoulder was emphasized for these experiences. Time constraints of a 2-day program based on SimLEARN format requirements prevented including an additional OSCE for the knee. At the conclusion of the course, faculty and participants discussed strategies for bringing this educational experience to learners at their local facilities as well as for avoiding potential barriers to implementation. The course was accredited through the VHA Employee Education System (EES), and participants received 16 hours of CPD credit.
Participants
Opportunity to attend was communicated through national, regional, and local VHA organizational networks. Participants self-registered online through the VHA Talent Management System, the main learning resource for VHA employee education, and registration was open to both PCPs and clinician educators. Class size was limited to 10 to facilitate detailed faculty observation during skill acquisition experiences, simulations, and assessment exercises.
Program Evaluation
A standard process for evaluating and measuring learning objectives was performed through VHA EES. Self-assessment surveys and OSCEs were used to assess the activity.
Self-assessment surveys were administered at the beginning and end of the program. Content was adapted from that used in the national MSK Mini-Residency initiative and revised by experts in survey design.18,24,25 Pre- and postcourse surveys asked participants to rate how important it was for them to be competent in evaluating shoulder and knee pain and in performing related joint injections, as well as to rate their level of confidence in their ability to evaluate and manage these conditions. The survey used 5 construct-specific response options distributed equally on a visual scale. Participants’ learning goals were collected on the precourse survey.
Participants’ competence in performing and interpreting a systematic and thorough physical examination of the shoulder and in suggesting a reasonable plan of management were assessed using a single-station OSCE. This tool, which presented learners with a simulated case depicting rotator cuff pathology, has been described in multiple educational settings, and validity evidence supporting its use has been published.18,19,23 Course faculty conducted the OSCE, one as the simulated patient, the other as the rater. Immediately following the examination, both faculty conducted a debriefing session with each participant. The OSCE was scored using the validated checklist for specific elements of the shoulder exam, followed by a structured sequence of questions exploring participants’ interpretation of findings, diagnostic impressions, and recommendations for initial management. Scores for participants’ differential diagnosis were based on the completeness and specificity of diagnoses given; scores for management plans were based on appropriateness and accuracy of both the primary and secondary approach to treatment or further diagnostic efforts. A global rating (range 1 to 9) was assigned, independent of scores in other domains.
Following the OSCE, participants rotated through a 3-cycle OSTE where they practiced the roles of simulated patient, learner, and educator. Faculty observed each OSTE and led focused debriefing sessions immediately following each rotation to facilitate participants’ critical reflection of their involvement in these elements of the course. This exercise was formative without quantitative assessment of performance.
Statistical Analysis
Pre- and postsurvey data were analyzed using a paired Student t test. Comparisons between multiple variables (eg, OSCE scores by years of experience or level of credentials) were analyzed using analysis of variance. Relationships between variables were analyzed with a Pearson correlation. All statistical analyses were conducted using IBM SPSS, Version 24 (Armonk, NY).
This project was reviewed by the institutional review board of the University of Utah and the Salt Lake City VA and was determined to be exempt from review because the work did not meet the definition of research with human subjects and was considered a quality improvement study.
Results
Twenty-four participants completed the program over 3 course offerings between February and May 2016, and all completed pre- and postcourse self-assessment surveys (Table 2). Self-ratings of the importance of competence in shoulder and knee MSK skills remained high before and after the course, and confidence improved significantly across all learning objectives. Despite the emphasis on the evaluation and management of shoulder pain, participants’ self-confidence still improved significantly with the knee—though these improvements were generally smaller in scale compared with those of the shoulder.
Overall OSCE scores and scores by domain were not found to be statistically different based on either years of experience or by level of credential or specialty (advanced practice registered nurse/physician assistant, PCP, or specialty care physician)(Table 3). However, there was a trend toward higher performance among the specialty care physician group, and a trend toward lower performance among participants with less than 3 years’ experience.
Discussion
Building on the foundation of other successful innovations in MSK education, the first year of the SimLEARN National MSK Training Program demonstrated the feasibility of a 2-day centralized national course as a method to increase participants’ confidence and competence in evaluating and managing MSK problems, and to disseminate a portable curriculum to a range of clinician educators. Although this course focused on developing competence for shoulder skills, including an OSCE on day 2, self-perceived improvements in participants’ ability to evaluate and manage knee pain were observed. Future program refinement and follow-up of participants’ experience and needs may lead to increased time allocated to the knee exam as well as objective measures of competence for knee skills.
In comparing our findings to the work that others have previously described, we looked for reports of CPD programs in 2 contexts: those that focused on acquisition of MSK skills relevant to clinical practice, and those designed as clinician educator or faculty development initiatives. Although there are few reports of MSK-themed CPD experiences designed specifically for nurses and allied health professionals, a recent effort to survey members of these disciplines in the United Kingdom was an important contribution to a systematic needs assessment.26-28 Increased support from leadership, mostly in terms of time allowance and budgetary support, was identified as an important driver to facilitate participation in MSK CPD experiences. Through SimLEARN, the VHA is investing in CPD, providing the MSK Training Programs and other courses at no cost to its employees.
Most published reports on physician education have not evaluated content knowledge or physical examination skills with measures for which validity evidence has been published.19,29,30 One notable exception is the 2000 Canadian Viscosupplementation Injector Preceptor experience, in which Bellamy and colleagues examined patient outcomes in evaluating their program.31
Our experience is congruent with the work of Macedo and colleagues and Sturpe and colleagues, who described the effectiveness and acceptability of an OSTE for faculty development.32,33 These studies emphasize debriefing, a critical element in faculty development identified by Steinert and colleagues in a 2006 best evidence medical education (BEME) review.34 The shoulder OSTE was one of the most well-received elements of our course, and each debrief was critical to facilitating rich discussions between educators and practitioners playing the role of teacher or student during this simulated experience, gaining insight into each other’s perspectives.
This program has several significant strengths: First, this is the most recent step in the development of a portfolio of innovative MSK CPD programs that were envisioned through a systematic process involving projections of cost-effectiveness, local pilot testing, and national expansion.17,18,35 Second, the SimLEARN program uses assessment tools for which validity evidence has been published, made available for reflective critique by educational scholars.19,23 This supports a national consortium of MSK educators, advancing clinical teaching and educational scholarship, and creating opportunities for interprofessional collaboration in congruence with the vision expressed in the 2010 Institute of Medicine report, “Redesigning Continuing Education in the Health Professions,” as well as the 2016 update of the BEME recommendations for faculty development.36,37
Our experience with the SimLEARN National MSK Training Program demonstrates need for 2 distinct courses: (1) the MSK Clinician—serving PCPs seeking to develop their skills in evaluating and managing patients with MSK problems; and (2), the MSK Master Educator—for those with preexisting content expertise who would value the introduction to a national curriculum and connections with other MSK master educators. Both of these are now offered regularly through SimLEARN for VHA and US Department of Defense employees. The MSK Clinician program establishes competence in systematically evaluating and managing shoulder and knee MSK problems in an educational setting and prepares participants for subsequent clinical experiences where they can perform related procedures if desired, under appropriate supervision. The Master Educator program introduces partici pants to the clinician curriculum and provides the opportunity to develop an individualized plan for implementation of an MSK educational program at their home institutions. Participants are selected through a competitive application process, and funding for travel to attend the Master Educator program is provided by SimLEARN for participants who are accepted. Additionally, the Master Educator program serves as a repository for potential future SimLEARN MSK Clinician course faculty.
Limitations
The small number of participants may limit the validity of our conclusions. Although we included an OSCE to measure competence in performing and interpreting the shoulder exam, the durability of these skills is not known. Periodic postcourse OSCEs could help determine this and refresh and preserve accuracy in the performance of specific maneuvers. Second, although this experience was rated highly by participants, we do not know the impact of the program on their daily work or career trajectory. Sustained follow-up of learners, perhaps developed on the model of the Long-Term Career Outcome Study, may increase the value of this experience for future participants.38 This program appealed to a diverse pool of learners, with a broad range of precourse expertise and varied expectations of how course experiences would impact their future work and career development. Some clinical educator attendees came from tertiary care facilities affiliated with academic medical centers, held specialist or subspecialist credentials, and had formal responsibilities as leaders in HPE. Other clinical practitioner participants were solitary PCPs, often in rural or home-based settings; although they may have been eager to apply new knowledge and skills in patient care, they neither anticipated nor desired any role as an educator.
Conclusion
The initial SimLEARN MSK Training Program provides PCPs and clinician educators with rich learning experiences, increasing confidence in addressing MSK problems and competence in performing and interpreting a systematic physical examination of the shoulder. The success of this program has created new opportunities for practitioners seeking to strengthen clinical skills and for leaders in health professions education looking to disseminate similar trainings and connect with a national group of educators.
Acknowledgments
The authors gratefully acknowledge the faculty and staff at the Veterans Health Administration SimLEARN National Simulation Center, the faculty of the Salt Lake City Musculoskeletal Mini-Residency program, the supportive leadership of the George E. Wahlen Salt Lake City Veterans Affairs Medical Center, and the efforts of Danielle Blake for logistical support and data entry.
1. Helmick CG, Felson DT, Lawrence RC, et al; National Arthritis Data Workgroup. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum. 2008;58(1):15-25.
2. Lawrence RC, Felson DT, Helmick CG, et al; National Arthritis Data Workgroup. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58(1):26-35.
3. Sacks JJ, Luo YH, Helmick CG. Prevalence of specific types of arthritis and other rheumatic conditions in the ambulatory health care system in the United States, 2001-2005. Arthritis Care Res (Hoboken). 2010;62(4):460-464.
4. Gupta S, Hawker GA, Laporte A, Croxford R, Coyte PC. The economic burden of disabling hip and knee osteoarthritis (OA) from the perspective of individuals living with this condition. Rheumatology (Oxford). 2005;44(12):1531-1537.
5. Gore M, Tai KS, Sadosky A, Leslie D, Stacey BR. Clinical comorbidities, treatment patterns, and direct medical costs of patients with osteoarthritis in usual care: a retrospective claims database analysis. J Med Econ. 2011;14(4):497-507.
6. Rabenda V, Manette C, Lemmens R, Mariani AM, Struvay N, Reginster JY. Direct and indirect costs attributable to osteoarthritis in active subjects. J Rheumatol. 2006;33(6):1152-1158.
7. Day CS, Yeh AC. Evidence of educational inadequacies in region-specific musculoskeletal medicine. Clin Orthop Relat Res. 2008;466(10):2542-2547.
8. Glazier RH, Dalby DM, Badley EM, Hawker GA, Bell MJ, Buchbinder R. Determinants of physician confidence in the primary care management of musculoskeletal disorders. J Rheumatol. 1996;23(2):351-356.
9. Haywood BL, Porter SL, Grana WA. Assessment of musculoskeletal knowledge in primary care residents. Am J Orthop (Belle Mead NJ). 2006;35(6):273-275.
10. Monrad SU, Zeller JL, Craig CL, Diponio LA. Musculoskeletal education in US medical schools: lessons from the past and suggestions for the future. Curr Rev Musculoskelet Med. 2011;4(3):91-98.
11. O’Dunn-Orto A, Hartling L, Campbell S, Oswald AE. Teaching musculoskeletal clinical skills to medical trainees and physicians: a Best Evidence in Medical Education systematic review of strategies and their effectiveness: BEME Guide No. 18. Med Teach. 2012;34(2):93-102.
12. Wilcox T, Oyler J, Harada C, Utset T. Musculoskeletal exam and joint injection training for internal medicine residents. J Gen Intern Med. 2006;21(5):521-523.
13. Petron DJ, Greis PE, Aoki SK, et al. Use of knee magnetic resonance imaging by primary care physicians in patients aged 40 years and older. Sports Health. 2010;2(5):385-390.
14. Roberts TT, Singer N, Hushmendy S, et al. MRI for the evaluation of knee pain: comparison of ordering practices of primary care physicians and orthopaedic surgeons. J Bone Joint Surg Am. 2015;97(9):709-714.
15. Wylie JD, Crim JR, Working ZM, Schmidt RL, Burks RT. Physician provider type influences utilization and diagnostic utility of magnetic resonance imaging of the knee. J Bone Joint Surg Am. 2015;97(1):56-62.
16. Smith M, Saunders R, Stuckhardt L, McGinnis JM, eds. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America. Washington, DC; 2013.
17. Battistone MJ, Barker AM, Lawrence P, Grotzke MP, Cannon GW. Mini-residency in musculoskeletal care: an interprofessional, mixed-methods educational initiative for primary care providers. Arthritis Care Res (Hoboken). 2016;68(2):275-279.
18. Battistone MJ, Barker AM, Grotzke MP, Beck JP, Lawrence P, Cannon GW. “Mini-residency” in musculoskeletal care: a national continuing professional development program for primary care providers. J Gen Intern Med. 2016;31(11):1301-1307.
19. Battistone MJ, Barker AM, Grotzke MP, et al. Effectiveness of an interprofessional and multidisciplinary musculoskeletal training program. J Grad Med Educ. 2016;8(3):398-404.
20. Battistone MJ, Barker AM, Lawrence P, Grotzke M, Cannon GW. Two-year impact of a continuing professional education program to train primary care providers to perform arthrocentesis. Presented at: 2017 ACR/ARHP Annual Meeting [Abstract 909]. https://acrabstracts.org/abstract/two-year-impact-of-a-continuing-professional-education-program-to-train-primary-care-providers-to-perform-arthrocentesis. Accessed November 14, 2019.
21. Call MR, Barker AM, Lawrence P, Cannon GW, Battistone MJ. Impact of a musculoskeltal “mini-residency” continuing professional education program on knee mri orders by primary care providers. Presented at: 2015 ACR/ARHP Annual Meeting [Abstract 1011]. https://acrabstracts.org/abstract/impact-of-a-musculoskeletal-aeoemini-residencyae%ef%bf%bd-continuing-professional-education-program-on-knee-mri-orders-by-primary-care-providers. Accessed November 14, 2019.
22. US Department of Veterans Affairs. VHA SimLEARN. https://www.simlearn.va.gov/SIMLEARN/about_us.asp. Updated January 24, 2019. Accessed November 13, 2019.
23. Battistone MJ, Barker AM, Beck JP, Tashjian RZ, Cannon GW. Validity evidence for two objective structured clinical examination stations to evaluate core skills of the shoulder and knee assessment. BMC Med Educ. 2017;17(1):13.
24. Artino AR Jr, La Rochelle JS, Dezee KJ, Gehlbach H. Developing questionnaires for educational research: AMEE Guide No. 87. Med Teach. 2014;36(6):463-474.
25. Gehlbach H, Artino AR Jr. The survey checklist (Manifesto). Acad Med. 2018;93(3):360-366.
26. Haywood H, Pain H, Ryan S, Adams J. The continuing professional development for nurses and allied health professionals working within musculoskeletal services: a national UK survey. Musculoskeletal Care. 2013;11(2):63-70.
27. Haywood H, Pain H, Ryan S, Adams J. Continuing professional development: issues raised by nurses and allied health professionals working in musculoskeletal settings. Musculoskeletal Care. 2013;11(3):136-144.
28. Warburton L. Continuing professional development in musculoskeletal domains. Musculoskeletal Care. 2012;10(3):125-126.
29. Stansfield RB, Diponio L, Craig C, et al. Assessing musculoskeletal examination skills and diagnostic reasoning of 4th year medical students using a novel objective structured clinical exam. BMC Med Educ. 2016;16(1):268.
30. Hose MK, Fontanesi J, Woytowitz M, Jarrin D, Quan A. Competency based clinical shoulder examination training improves physical exam, confidence, and knowledge in common shoulder conditions. J Gen Intern Med. 2017;32(11):1261-1265.
31. Bellamy N, Goldstein LD, Tekanoff RA. Continuing medical education-driven skills acquisition and impact on improved patient outcomes in family practice setting. J Contin Educ Health Prof. 2000;20(1):52-61.
32. Macedo L, Sturpe DA, Haines ST, Layson-Wolf C, Tofade TS, McPherson ML. An objective structured teaching exercise (OSTE) for preceptor development. Curr Pharm Teach Learn. 2015;7(5):627-634.
33. Sturpe DA, Schaivone KA. A primer for objective structured teaching exercises. Am J Pharm Educ. 2014;78(5):104.
34. Steinert Y, Mann K, Centeno A, et al. A systematic review of faculty development initiatives designed to improve teaching effectiveness in medical education: BEME Guide No. 8. Med Teach. 2006;28(6):497-526.
35. Nelson SD, Nelson RE, Cannon GW, et al. Cost-effectiveness of training rural providers to identify and treat patients at risk for fragility fractures. Osteoporos Int. 2014;25(12):2701-2707.
36. Steinert Y, Mann K, Anderson B, et al. A systematic review of faculty development initiatives designed to enhance teaching effectiveness: A 10-year update: BEME Guide No. 40. Med Teach. 2016;38(8):769-786.
37. Institute of Medicine. Redesigning Continuing Education in the Health Professions. Washington, DC: National Academies Press; 2010.
38. Durning SJ, Dong T, LaRochelle JL, et al. The long-term career outcome study: lessons learned and implications for educational practice. Mil Med. 2015;180(suppl 4):164-170.
1. Helmick CG, Felson DT, Lawrence RC, et al; National Arthritis Data Workgroup. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum. 2008;58(1):15-25.
2. Lawrence RC, Felson DT, Helmick CG, et al; National Arthritis Data Workgroup. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58(1):26-35.
3. Sacks JJ, Luo YH, Helmick CG. Prevalence of specific types of arthritis and other rheumatic conditions in the ambulatory health care system in the United States, 2001-2005. Arthritis Care Res (Hoboken). 2010;62(4):460-464.
4. Gupta S, Hawker GA, Laporte A, Croxford R, Coyte PC. The economic burden of disabling hip and knee osteoarthritis (OA) from the perspective of individuals living with this condition. Rheumatology (Oxford). 2005;44(12):1531-1537.
5. Gore M, Tai KS, Sadosky A, Leslie D, Stacey BR. Clinical comorbidities, treatment patterns, and direct medical costs of patients with osteoarthritis in usual care: a retrospective claims database analysis. J Med Econ. 2011;14(4):497-507.
6. Rabenda V, Manette C, Lemmens R, Mariani AM, Struvay N, Reginster JY. Direct and indirect costs attributable to osteoarthritis in active subjects. J Rheumatol. 2006;33(6):1152-1158.
7. Day CS, Yeh AC. Evidence of educational inadequacies in region-specific musculoskeletal medicine. Clin Orthop Relat Res. 2008;466(10):2542-2547.
8. Glazier RH, Dalby DM, Badley EM, Hawker GA, Bell MJ, Buchbinder R. Determinants of physician confidence in the primary care management of musculoskeletal disorders. J Rheumatol. 1996;23(2):351-356.
9. Haywood BL, Porter SL, Grana WA. Assessment of musculoskeletal knowledge in primary care residents. Am J Orthop (Belle Mead NJ). 2006;35(6):273-275.
10. Monrad SU, Zeller JL, Craig CL, Diponio LA. Musculoskeletal education in US medical schools: lessons from the past and suggestions for the future. Curr Rev Musculoskelet Med. 2011;4(3):91-98.
11. O’Dunn-Orto A, Hartling L, Campbell S, Oswald AE. Teaching musculoskeletal clinical skills to medical trainees and physicians: a Best Evidence in Medical Education systematic review of strategies and their effectiveness: BEME Guide No. 18. Med Teach. 2012;34(2):93-102.
12. Wilcox T, Oyler J, Harada C, Utset T. Musculoskeletal exam and joint injection training for internal medicine residents. J Gen Intern Med. 2006;21(5):521-523.
13. Petron DJ, Greis PE, Aoki SK, et al. Use of knee magnetic resonance imaging by primary care physicians in patients aged 40 years and older. Sports Health. 2010;2(5):385-390.
14. Roberts TT, Singer N, Hushmendy S, et al. MRI for the evaluation of knee pain: comparison of ordering practices of primary care physicians and orthopaedic surgeons. J Bone Joint Surg Am. 2015;97(9):709-714.
15. Wylie JD, Crim JR, Working ZM, Schmidt RL, Burks RT. Physician provider type influences utilization and diagnostic utility of magnetic resonance imaging of the knee. J Bone Joint Surg Am. 2015;97(1):56-62.
16. Smith M, Saunders R, Stuckhardt L, McGinnis JM, eds. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America. Washington, DC; 2013.
17. Battistone MJ, Barker AM, Lawrence P, Grotzke MP, Cannon GW. Mini-residency in musculoskeletal care: an interprofessional, mixed-methods educational initiative for primary care providers. Arthritis Care Res (Hoboken). 2016;68(2):275-279.
18. Battistone MJ, Barker AM, Grotzke MP, Beck JP, Lawrence P, Cannon GW. “Mini-residency” in musculoskeletal care: a national continuing professional development program for primary care providers. J Gen Intern Med. 2016;31(11):1301-1307.
19. Battistone MJ, Barker AM, Grotzke MP, et al. Effectiveness of an interprofessional and multidisciplinary musculoskeletal training program. J Grad Med Educ. 2016;8(3):398-404.
20. Battistone MJ, Barker AM, Lawrence P, Grotzke M, Cannon GW. Two-year impact of a continuing professional education program to train primary care providers to perform arthrocentesis. Presented at: 2017 ACR/ARHP Annual Meeting [Abstract 909]. https://acrabstracts.org/abstract/two-year-impact-of-a-continuing-professional-education-program-to-train-primary-care-providers-to-perform-arthrocentesis. Accessed November 14, 2019.
21. Call MR, Barker AM, Lawrence P, Cannon GW, Battistone MJ. Impact of a musculoskeltal “mini-residency” continuing professional education program on knee mri orders by primary care providers. Presented at: 2015 ACR/ARHP Annual Meeting [Abstract 1011]. https://acrabstracts.org/abstract/impact-of-a-musculoskeletal-aeoemini-residencyae%ef%bf%bd-continuing-professional-education-program-on-knee-mri-orders-by-primary-care-providers. Accessed November 14, 2019.
22. US Department of Veterans Affairs. VHA SimLEARN. https://www.simlearn.va.gov/SIMLEARN/about_us.asp. Updated January 24, 2019. Accessed November 13, 2019.
23. Battistone MJ, Barker AM, Beck JP, Tashjian RZ, Cannon GW. Validity evidence for two objective structured clinical examination stations to evaluate core skills of the shoulder and knee assessment. BMC Med Educ. 2017;17(1):13.
24. Artino AR Jr, La Rochelle JS, Dezee KJ, Gehlbach H. Developing questionnaires for educational research: AMEE Guide No. 87. Med Teach. 2014;36(6):463-474.
25. Gehlbach H, Artino AR Jr. The survey checklist (Manifesto). Acad Med. 2018;93(3):360-366.
26. Haywood H, Pain H, Ryan S, Adams J. The continuing professional development for nurses and allied health professionals working within musculoskeletal services: a national UK survey. Musculoskeletal Care. 2013;11(2):63-70.
27. Haywood H, Pain H, Ryan S, Adams J. Continuing professional development: issues raised by nurses and allied health professionals working in musculoskeletal settings. Musculoskeletal Care. 2013;11(3):136-144.
28. Warburton L. Continuing professional development in musculoskeletal domains. Musculoskeletal Care. 2012;10(3):125-126.
29. Stansfield RB, Diponio L, Craig C, et al. Assessing musculoskeletal examination skills and diagnostic reasoning of 4th year medical students using a novel objective structured clinical exam. BMC Med Educ. 2016;16(1):268.
30. Hose MK, Fontanesi J, Woytowitz M, Jarrin D, Quan A. Competency based clinical shoulder examination training improves physical exam, confidence, and knowledge in common shoulder conditions. J Gen Intern Med. 2017;32(11):1261-1265.
31. Bellamy N, Goldstein LD, Tekanoff RA. Continuing medical education-driven skills acquisition and impact on improved patient outcomes in family practice setting. J Contin Educ Health Prof. 2000;20(1):52-61.
32. Macedo L, Sturpe DA, Haines ST, Layson-Wolf C, Tofade TS, McPherson ML. An objective structured teaching exercise (OSTE) for preceptor development. Curr Pharm Teach Learn. 2015;7(5):627-634.
33. Sturpe DA, Schaivone KA. A primer for objective structured teaching exercises. Am J Pharm Educ. 2014;78(5):104.
34. Steinert Y, Mann K, Centeno A, et al. A systematic review of faculty development initiatives designed to improve teaching effectiveness in medical education: BEME Guide No. 8. Med Teach. 2006;28(6):497-526.
35. Nelson SD, Nelson RE, Cannon GW, et al. Cost-effectiveness of training rural providers to identify and treat patients at risk for fragility fractures. Osteoporos Int. 2014;25(12):2701-2707.
36. Steinert Y, Mann K, Anderson B, et al. A systematic review of faculty development initiatives designed to enhance teaching effectiveness: A 10-year update: BEME Guide No. 40. Med Teach. 2016;38(8):769-786.
37. Institute of Medicine. Redesigning Continuing Education in the Health Professions. Washington, DC: National Academies Press; 2010.
38. Durning SJ, Dong T, LaRochelle JL, et al. The long-term career outcome study: lessons learned and implications for educational practice. Mil Med. 2015;180(suppl 4):164-170.
Shining a Light to Reduce Hospital Falls
Fall prevention strategies for hospitalized older adults include environmental factors such as adequate room lighting and patient-specific factors such as medications. In 2008, the Centers for Medicare & Medicaid Services (CMS) implemented a regulatory “shining of the light” on hospital-acquired falls by eliminating hospital payment for fall-related injuries. Shorr et al. found that implementation of the CMS Hospital-Acquired Conditions Initiative was associated with only a modest decline in falls and injurious falls over the first seven years, with the greatest reduction occurring in urban, teaching hospitals.1 These disappointing findings were mitigated only by the finding that the prevalence of physical restraints decreased over the seven years of observation from 1.6% to 0.6%, suggesting that the modest reductions in falls did not occur at the expense of further restricting the mobility of hospitalized older adults. Shorr et al. concluded that falls may be largely attributable to individual patient risk and may not be prevented through health system quality and safety programs such as those that have achieved successes in never-events, including wrong-side surgery and catheter-associated blood stream infections.2 The authors expressed concern that hospital leaders remain in the dark regarding proven fall prevention strategies. They question whether hospital-acquired falls are preventable without restricting the mobility of older adults most at risk for falls.
Hoff et al. found in their 2011 literature review of the first three years following implementation of the 2008 CMS hospital payment polices limited evidence-based approaches to address falls as a spotlighted avoidable hospital-acquired condition.3 Swartzell et al. reported that at some level, every patient admitted to an acute care hospital is at risk for falls. “Patients sick enough to be in the hospital have underlying disease, are receiving physiologically altering medications and treatments, and are likely experiencing pain, fatigue, anxiety, sleep disturbance, and other symptoms that interfere with cognitive and physical functioning. The key to preventing falls among hospitalized patients may lie in addressing how the hospital environment creates risk.”4
In 2017, Avanecean et al. published a systematic review of randomized control trials on fall prevention in hospitals.5 Three of five studies demonstrated 20%-30% reductions in fall rates, whereas two studies showed no difference in fall rates among control and intervention groups. In the three studies that demonstrated reduced fall rates, standardized fall risk assessments were used to identify patient-specific risks for falls. Individualized care plans addressed gait and balance disorders, delirium and cognitive deficits, vision and hearing impairments, and toileting needs. For example, physical therapists provided instruction on the safe use of walkers for those with gait and balance disorders. Patients with delirium and cognitive deficits received some form of staff alert of unsupervised transfers out of bed, ranging from bed alarms to customized rubber socks that contained pressure alarms. All three successful intervention studies included patient-centered care plans for toileting.
None of the three studies that measured the secondary outcome of fall-related injuries demonstrated impact of interventions, although the rates of injurious falls were low in both the control and intervention groups (2%-5%).3-5
Since the 2008 CMS policies eliminated hospital payments for complications of falls, patient-centered models of fall risk reduction were widely implemented. The Systems Addressing Frail Elder (SAFE) Care, designed by Ansryan et al. includes nursing, social work, pharmacist, and medical provider assessments.6 Team huddles occur daily to establish individualized care plans, although as Shorr et al. highlight, without report of outcomes.2 Nurses Improving Care for Healthsystem Elders (NICHE) is an New York University-based nursing education and consultation program that has extended to 566 healthcare organizations.7 Factors that promote the adoption of organizational interventions such as NICHE have been identified.8 The findings that NICHE is adopted more in larger, urban healthcare systems are consistent with the findings reported by Shorr et al. that fall rate reductions were greater in such hospital settings. Patient-centered care, although time-consuming, may promote staff satisfaction and is associated with reductions in other hospital-acquired conditions such as delirium.9
Patient-engaged video surveillance systems are recent technological solutions to reduce falls. One staff monitors multiple patients for behaviors that risk falls such as unsupervised transfers out of bed. Staff can speak to a patient through the monitoring system to request the patient to wait for assistance, while the unit staff are alerted to the fall risk. Bedside caregivers can activate virtual privacy screens during personal patient care.
Shorr et al. appropriately call for studies to further illuminate strategies to reduce hospital-acquired falls. A multihospital report of fall rates before and after the implementation of SAFE Care and NICHE would have sufficient scale to address the impact of these patient-centered interventions on injurious falls. Similarly, patient-engaged video surveillance systems need validation from clinical trials.
1. Shorr RI, Staggs VS, Waters TM, et al. Impact of the hospital-acquired conditions initiative on falls and physical restraints: a longitudinal study. J Hosp Med. 2019;14:E31-E36. https://doi.org/10.12788/jhm.3295.
2. Austin JM, Demski R, Callender T, et al. From board to bedside: how the application of financial structures to safety and quality can drive accountability in a large health care system. Jt Comm J Qual Patient Saf. 2017;43(4):166-175. https://doi.org/10.1016/j.jcjq.2017.01.001.
3. Hoff TJ, Soerensen C. No payment for preventable complications: reviewing the early literature for content, guidance, and impressions. Qual Manag Health Care. 2011;20(1):62-75. https://doi.org/10.1097/QMH.0b013e31820311d2.
4. Swartzell KL, Fulton JS, Friesth BM. Relationship between occurrence of falls and fall-risk scores in an acute care setting using the Hendrich II fall risk model. Medsurg Nurs. 2013;22(3):180-187.
5. Avanecean D, Calliste D, Contreras T, Lim Y, Fitzpatrick A. Effectiveness of patient-centered interventions on falls in the acute care setting compared to usual care: a systematic review. JBI Database System Rev Implement Rep. 2017;15(12): 3006-3048. https://doi.org/10.11124/JBISRIR-2016-003331.
6. Ansryan LZ1, Aronow HU, Borenstein JE, et al. Systems addressing frail elder care: description of a successful model. J Nurs Adm. 2018;48(1):11-17. https://doi.org/10.1097/NNA.0000000000000564.
7. Boltz M1, Capezuti E, Bowar-Ferres S, et al. Changes in the geriatric care environment associated with NICHE (Nurses Improving Care for HealthSystem Elders). Geriatr Nurs. 2008;29(3):176-185. https://doi.org/10.1016/j.gerinurse.2008.02.002.
8. Stimpfel AW1, Gilmartin MJ. Factors predicting adoption of the nurses improving care of healthsystem elders program. Nurs Res. 2019;68(1):13-21. https://doi.org/10.1097/NNR.0000000000000327.
9.
Fall prevention strategies for hospitalized older adults include environmental factors such as adequate room lighting and patient-specific factors such as medications. In 2008, the Centers for Medicare & Medicaid Services (CMS) implemented a regulatory “shining of the light” on hospital-acquired falls by eliminating hospital payment for fall-related injuries. Shorr et al. found that implementation of the CMS Hospital-Acquired Conditions Initiative was associated with only a modest decline in falls and injurious falls over the first seven years, with the greatest reduction occurring in urban, teaching hospitals.1 These disappointing findings were mitigated only by the finding that the prevalence of physical restraints decreased over the seven years of observation from 1.6% to 0.6%, suggesting that the modest reductions in falls did not occur at the expense of further restricting the mobility of hospitalized older adults. Shorr et al. concluded that falls may be largely attributable to individual patient risk and may not be prevented through health system quality and safety programs such as those that have achieved successes in never-events, including wrong-side surgery and catheter-associated blood stream infections.2 The authors expressed concern that hospital leaders remain in the dark regarding proven fall prevention strategies. They question whether hospital-acquired falls are preventable without restricting the mobility of older adults most at risk for falls.
Hoff et al. found in their 2011 literature review of the first three years following implementation of the 2008 CMS hospital payment polices limited evidence-based approaches to address falls as a spotlighted avoidable hospital-acquired condition.3 Swartzell et al. reported that at some level, every patient admitted to an acute care hospital is at risk for falls. “Patients sick enough to be in the hospital have underlying disease, are receiving physiologically altering medications and treatments, and are likely experiencing pain, fatigue, anxiety, sleep disturbance, and other symptoms that interfere with cognitive and physical functioning. The key to preventing falls among hospitalized patients may lie in addressing how the hospital environment creates risk.”4
In 2017, Avanecean et al. published a systematic review of randomized control trials on fall prevention in hospitals.5 Three of five studies demonstrated 20%-30% reductions in fall rates, whereas two studies showed no difference in fall rates among control and intervention groups. In the three studies that demonstrated reduced fall rates, standardized fall risk assessments were used to identify patient-specific risks for falls. Individualized care plans addressed gait and balance disorders, delirium and cognitive deficits, vision and hearing impairments, and toileting needs. For example, physical therapists provided instruction on the safe use of walkers for those with gait and balance disorders. Patients with delirium and cognitive deficits received some form of staff alert of unsupervised transfers out of bed, ranging from bed alarms to customized rubber socks that contained pressure alarms. All three successful intervention studies included patient-centered care plans for toileting.
None of the three studies that measured the secondary outcome of fall-related injuries demonstrated impact of interventions, although the rates of injurious falls were low in both the control and intervention groups (2%-5%).3-5
Since the 2008 CMS policies eliminated hospital payments for complications of falls, patient-centered models of fall risk reduction were widely implemented. The Systems Addressing Frail Elder (SAFE) Care, designed by Ansryan et al. includes nursing, social work, pharmacist, and medical provider assessments.6 Team huddles occur daily to establish individualized care plans, although as Shorr et al. highlight, without report of outcomes.2 Nurses Improving Care for Healthsystem Elders (NICHE) is an New York University-based nursing education and consultation program that has extended to 566 healthcare organizations.7 Factors that promote the adoption of organizational interventions such as NICHE have been identified.8 The findings that NICHE is adopted more in larger, urban healthcare systems are consistent with the findings reported by Shorr et al. that fall rate reductions were greater in such hospital settings. Patient-centered care, although time-consuming, may promote staff satisfaction and is associated with reductions in other hospital-acquired conditions such as delirium.9
Patient-engaged video surveillance systems are recent technological solutions to reduce falls. One staff monitors multiple patients for behaviors that risk falls such as unsupervised transfers out of bed. Staff can speak to a patient through the monitoring system to request the patient to wait for assistance, while the unit staff are alerted to the fall risk. Bedside caregivers can activate virtual privacy screens during personal patient care.
Shorr et al. appropriately call for studies to further illuminate strategies to reduce hospital-acquired falls. A multihospital report of fall rates before and after the implementation of SAFE Care and NICHE would have sufficient scale to address the impact of these patient-centered interventions on injurious falls. Similarly, patient-engaged video surveillance systems need validation from clinical trials.
Fall prevention strategies for hospitalized older adults include environmental factors such as adequate room lighting and patient-specific factors such as medications. In 2008, the Centers for Medicare & Medicaid Services (CMS) implemented a regulatory “shining of the light” on hospital-acquired falls by eliminating hospital payment for fall-related injuries. Shorr et al. found that implementation of the CMS Hospital-Acquired Conditions Initiative was associated with only a modest decline in falls and injurious falls over the first seven years, with the greatest reduction occurring in urban, teaching hospitals.1 These disappointing findings were mitigated only by the finding that the prevalence of physical restraints decreased over the seven years of observation from 1.6% to 0.6%, suggesting that the modest reductions in falls did not occur at the expense of further restricting the mobility of hospitalized older adults. Shorr et al. concluded that falls may be largely attributable to individual patient risk and may not be prevented through health system quality and safety programs such as those that have achieved successes in never-events, including wrong-side surgery and catheter-associated blood stream infections.2 The authors expressed concern that hospital leaders remain in the dark regarding proven fall prevention strategies. They question whether hospital-acquired falls are preventable without restricting the mobility of older adults most at risk for falls.
Hoff et al. found in their 2011 literature review of the first three years following implementation of the 2008 CMS hospital payment polices limited evidence-based approaches to address falls as a spotlighted avoidable hospital-acquired condition.3 Swartzell et al. reported that at some level, every patient admitted to an acute care hospital is at risk for falls. “Patients sick enough to be in the hospital have underlying disease, are receiving physiologically altering medications and treatments, and are likely experiencing pain, fatigue, anxiety, sleep disturbance, and other symptoms that interfere with cognitive and physical functioning. The key to preventing falls among hospitalized patients may lie in addressing how the hospital environment creates risk.”4
In 2017, Avanecean et al. published a systematic review of randomized control trials on fall prevention in hospitals.5 Three of five studies demonstrated 20%-30% reductions in fall rates, whereas two studies showed no difference in fall rates among control and intervention groups. In the three studies that demonstrated reduced fall rates, standardized fall risk assessments were used to identify patient-specific risks for falls. Individualized care plans addressed gait and balance disorders, delirium and cognitive deficits, vision and hearing impairments, and toileting needs. For example, physical therapists provided instruction on the safe use of walkers for those with gait and balance disorders. Patients with delirium and cognitive deficits received some form of staff alert of unsupervised transfers out of bed, ranging from bed alarms to customized rubber socks that contained pressure alarms. All three successful intervention studies included patient-centered care plans for toileting.
None of the three studies that measured the secondary outcome of fall-related injuries demonstrated impact of interventions, although the rates of injurious falls were low in both the control and intervention groups (2%-5%).3-5
Since the 2008 CMS policies eliminated hospital payments for complications of falls, patient-centered models of fall risk reduction were widely implemented. The Systems Addressing Frail Elder (SAFE) Care, designed by Ansryan et al. includes nursing, social work, pharmacist, and medical provider assessments.6 Team huddles occur daily to establish individualized care plans, although as Shorr et al. highlight, without report of outcomes.2 Nurses Improving Care for Healthsystem Elders (NICHE) is an New York University-based nursing education and consultation program that has extended to 566 healthcare organizations.7 Factors that promote the adoption of organizational interventions such as NICHE have been identified.8 The findings that NICHE is adopted more in larger, urban healthcare systems are consistent with the findings reported by Shorr et al. that fall rate reductions were greater in such hospital settings. Patient-centered care, although time-consuming, may promote staff satisfaction and is associated with reductions in other hospital-acquired conditions such as delirium.9
Patient-engaged video surveillance systems are recent technological solutions to reduce falls. One staff monitors multiple patients for behaviors that risk falls such as unsupervised transfers out of bed. Staff can speak to a patient through the monitoring system to request the patient to wait for assistance, while the unit staff are alerted to the fall risk. Bedside caregivers can activate virtual privacy screens during personal patient care.
Shorr et al. appropriately call for studies to further illuminate strategies to reduce hospital-acquired falls. A multihospital report of fall rates before and after the implementation of SAFE Care and NICHE would have sufficient scale to address the impact of these patient-centered interventions on injurious falls. Similarly, patient-engaged video surveillance systems need validation from clinical trials.
1. Shorr RI, Staggs VS, Waters TM, et al. Impact of the hospital-acquired conditions initiative on falls and physical restraints: a longitudinal study. J Hosp Med. 2019;14:E31-E36. https://doi.org/10.12788/jhm.3295.
2. Austin JM, Demski R, Callender T, et al. From board to bedside: how the application of financial structures to safety and quality can drive accountability in a large health care system. Jt Comm J Qual Patient Saf. 2017;43(4):166-175. https://doi.org/10.1016/j.jcjq.2017.01.001.
3. Hoff TJ, Soerensen C. No payment for preventable complications: reviewing the early literature for content, guidance, and impressions. Qual Manag Health Care. 2011;20(1):62-75. https://doi.org/10.1097/QMH.0b013e31820311d2.
4. Swartzell KL, Fulton JS, Friesth BM. Relationship between occurrence of falls and fall-risk scores in an acute care setting using the Hendrich II fall risk model. Medsurg Nurs. 2013;22(3):180-187.
5. Avanecean D, Calliste D, Contreras T, Lim Y, Fitzpatrick A. Effectiveness of patient-centered interventions on falls in the acute care setting compared to usual care: a systematic review. JBI Database System Rev Implement Rep. 2017;15(12): 3006-3048. https://doi.org/10.11124/JBISRIR-2016-003331.
6. Ansryan LZ1, Aronow HU, Borenstein JE, et al. Systems addressing frail elder care: description of a successful model. J Nurs Adm. 2018;48(1):11-17. https://doi.org/10.1097/NNA.0000000000000564.
7. Boltz M1, Capezuti E, Bowar-Ferres S, et al. Changes in the geriatric care environment associated with NICHE (Nurses Improving Care for HealthSystem Elders). Geriatr Nurs. 2008;29(3):176-185. https://doi.org/10.1016/j.gerinurse.2008.02.002.
8. Stimpfel AW1, Gilmartin MJ. Factors predicting adoption of the nurses improving care of healthsystem elders program. Nurs Res. 2019;68(1):13-21. https://doi.org/10.1097/NNR.0000000000000327.
9.
1. Shorr RI, Staggs VS, Waters TM, et al. Impact of the hospital-acquired conditions initiative on falls and physical restraints: a longitudinal study. J Hosp Med. 2019;14:E31-E36. https://doi.org/10.12788/jhm.3295.
2. Austin JM, Demski R, Callender T, et al. From board to bedside: how the application of financial structures to safety and quality can drive accountability in a large health care system. Jt Comm J Qual Patient Saf. 2017;43(4):166-175. https://doi.org/10.1016/j.jcjq.2017.01.001.
3. Hoff TJ, Soerensen C. No payment for preventable complications: reviewing the early literature for content, guidance, and impressions. Qual Manag Health Care. 2011;20(1):62-75. https://doi.org/10.1097/QMH.0b013e31820311d2.
4. Swartzell KL, Fulton JS, Friesth BM. Relationship between occurrence of falls and fall-risk scores in an acute care setting using the Hendrich II fall risk model. Medsurg Nurs. 2013;22(3):180-187.
5. Avanecean D, Calliste D, Contreras T, Lim Y, Fitzpatrick A. Effectiveness of patient-centered interventions on falls in the acute care setting compared to usual care: a systematic review. JBI Database System Rev Implement Rep. 2017;15(12): 3006-3048. https://doi.org/10.11124/JBISRIR-2016-003331.
6. Ansryan LZ1, Aronow HU, Borenstein JE, et al. Systems addressing frail elder care: description of a successful model. J Nurs Adm. 2018;48(1):11-17. https://doi.org/10.1097/NNA.0000000000000564.
7. Boltz M1, Capezuti E, Bowar-Ferres S, et al. Changes in the geriatric care environment associated with NICHE (Nurses Improving Care for HealthSystem Elders). Geriatr Nurs. 2008;29(3):176-185. https://doi.org/10.1016/j.gerinurse.2008.02.002.
8. Stimpfel AW1, Gilmartin MJ. Factors predicting adoption of the nurses improving care of healthsystem elders program. Nurs Res. 2019;68(1):13-21. https://doi.org/10.1097/NNR.0000000000000327.
9.
© 2020 Society of Hospital Medicine DOI 10.12788/jhm.3345
Worry Loves Company, but Unnecessary Consultations May Harm the Patients We Comanage
“Never worry alone” is a common mantra that most of us have heard throughout medical training. The premise is simple and well meaning. If a patient has an issue that concerns you, ask someone for help. As a student, this can be a resident; as a resident, this can be an attending. However, for hospitalists, the answer is often a subspecialty consultation. Asking for help never seems to be wrong, but what happens when our worry delays appropriate care with unnecessary consultations? In this month’s issue of the Journal of Hospital Medicine, authors Bellas et al. have investigated this issue through the lens of subspecialty preoperative consultation for patients admitted to a hospitalist comanagement service with a fragility hip fracture requiring surgery.1
Morbidity and mortality for patients who experience hip fractures are high, and time to appropriate surgery is one of the few modifiable risk factors that may reduce morbidity and mortality.2,3 Bellas et al. conducted a retrospective cohort study to test the association between preoperative subspecialty consultation and multiple clinically relevant outcomes in patients admitted with an acute hip fracture.1 All patients were comanaged by a hospitalist and orthopedic surgery, and “consultation” was defined as any preoperative subspecialty consultation requested by the hospitalist. Outcome measures included time to surgery, length of stay, readmission rate, perioperative complications, and 30-day mortality. In total, 36% (177/491) of patients who underwent surgery received a subspecialty preoperative consultation. Unsurprisingly, these patients were older with higher rates of comorbidity. After controlling for age and Charlson Comorbidity Index, preoperative consultation was associated with dramatic delays and increased rates of time to surgery >24 hours (adjusted odds ratio, 4.2; 95% CI: 2.8-6.6). The authors classified 90% of consultations as appropriate, either because of an active condition (eg, acute coronary syndrome) or because admitting physicians documented a perception that patients were at increased risk. However, 73% of consultations had only minor recommendations, such as ordering an ECG or changing the dose of an existing medication, and only 37% of the time did consultations lead to an identifiable change in management as a result of the consultation.
Although striking, integrating these findings into clinical practice is complex. As a retrospective study, patients who received consultations were obviously different from those who did not. The authors attempted to adjust for this but used only age and Charlson Comorbidity Index. Other factors that are both associated with consultations and known to increase mortality—such as frailty and functional status—were not included in their adjustment. Such unmeasured confounders possibly explain at least some, if not all, of the findings that consultations were associated with a doubling of the likelihood of 30-day mortality. In addition, although the authors assessed the appropriateness of consultation and degree of recommendations, their methods for this deserve scrutiny. Two independent providers adjudicated the consultations with excellent agreement (kappa 0.96 for indication, 0.95 for degree of recommendation), but this reliability assessment was done on previously extracted chart data, probably inflating their agreement statistics. Finally, the adjudication of consultant recommendations into minor, moderate, and major categories may oversimplify the outcome of each consultation. For example, all medication recommendations, regardless of type, were considered as minor, and recommendations were considered as major only if they resulted in invasive testing or procedures. This approach may underrepresent the impact of consultations as in clinical practice not all high-impact recommendations result in invasive testing or procedures. Despite these important limitations, Bellas et al. present a compelling case for preoperative consultation being associated with delays in surgery.
How then should this study change practice? The authors’ findings tell two separate but intertwined stories. The first is that preoperative consultation leads to delays in surgery. As patients who received preoperative consultation were obviously sicker, and because delays caused by consultation may lead to increased morbidity and mortality, perhaps the solution is to simply fix the delays. However, this approach ignores the more compelling story the authors tell. More important than the delays was the surprising lack of impact of preoperative consultations. Bellas et al. found that the majority of consultations resulted in only minor recommendations, and more importantly, hospitalists rarely changed treatment as a result. Although patients who received consultations were more ill, consultation rarely changed their care or decreased the risk posed by surgery. Bellas et al. found that only patients with active medical conditions had consultations, which resulted in moderate or major recommendations. These findings highlight an opportunity to better identify patients for whom consultation might be helpful and to prevent delays by avoiding consultation for those unlikely to benefit. There have been several efforts in the orthopedic literature to use guidelines for preoperative cardiac testing to guide cardiology consultation.4,5,6 One study using this approach reported findings that were extremely similar to those reported by Bellas et al. in that 71% of preoperative cardiology consultations in their institution did not meet the guideline criteria for invasive cardiac testing.7 The primary difference between the findings of Bellas et al. and the studies in the orthopedic literature is the presence of the comanaging hospitalist. As more and more patients receive hospitalist comanagement prior to inpatient surgery, it is well within the scope of the hospitalist to differentiate chronic risk factors from active or decompensated medical disease requiring a subspecialist. This is in fact much of the value that a hospitalist adds. Avoiding consultation for patients with only elevated chronic risk factors is an important first step in avoiding unnecessary delays to surgery and an opportunity for hospitalists to improve the care of the patients they comanage.
The goal of teaching trainees to “never worry alone” is to harness the feelings of uncertainty that all providers face to improve patient care. Knowing when to worry is a valuable lesson, but as with all skills, it should be applied thoughtfully and informed by evidence. Appreciating the risks that surgery poses is quintessential to safe perioperative care, but equally important is understanding that inappropriate consultations can create risks from needless delays and testing. Only in balancing these two concerns, and appreciating when it is appropriate to worry, can we provide the highest quality of care to our patients.
1. Bellas N, Stohler S, Staff I, et al. Impact of preoperative consults and hospitalist comanagement in hip fracture patients. J Hosp Med. 2020;15(1):16-21. https:doi.org/jhm.3264.
2. Goldacre MJ, Roberts SE, Yeates D. Mortality after admission to hospital with fractured neck of femur: database study. BMJ 2002;325(7369):868-869. https://doi.org/10.1136/bmj.325.7369.868.
3. Shiga T, Wajima Z, Ohe Y. Is operative delay associated with increased mortality of hip fracture patients? Systematic review, meta-analysis, and meta-regression. Can J Anaesth. 2008;55(3):146-154. https://doi.org/10.1007/BF03016088.
4. Cluett J, Caplan J, Yu W. Preoperative cardiac evaluation of patients with acute hip fracture. Am J Orthop. 2008;37(1):32-36.
5. Smeets SJ, Poeze M, Verbruggen JP. Preoperative cardiac evaluation of geriatric patients with hip fracture. Injury. 2012;43(12):2146-2151. https://doi.org/10.1016/j.injury.2012.08.007.
6. Siu CW, Sun NC, Lau TW, Yiu KH, Leung F, Tse HF. Preoperative cardiac risk assessment in geriatric patients with hip fractures: an orthopedic surgeons’ perspective. Osteoporos Int. 2010;21(Suppl 4):S587-S591. https://doi.org/10.1007/s00198-010-1393-0.
7. Stitgen A, Poludnianyk K, Dulaney-Cripe E, Markert R, Prayson M. Adherence to preoperative cardiac clearance guidelines in hip fracture patients. J Orthop Trauma 2015;29(11):500-503. https://doi.org/10.1097/BOT.0000000000000381.
“Never worry alone” is a common mantra that most of us have heard throughout medical training. The premise is simple and well meaning. If a patient has an issue that concerns you, ask someone for help. As a student, this can be a resident; as a resident, this can be an attending. However, for hospitalists, the answer is often a subspecialty consultation. Asking for help never seems to be wrong, but what happens when our worry delays appropriate care with unnecessary consultations? In this month’s issue of the Journal of Hospital Medicine, authors Bellas et al. have investigated this issue through the lens of subspecialty preoperative consultation for patients admitted to a hospitalist comanagement service with a fragility hip fracture requiring surgery.1
Morbidity and mortality for patients who experience hip fractures are high, and time to appropriate surgery is one of the few modifiable risk factors that may reduce morbidity and mortality.2,3 Bellas et al. conducted a retrospective cohort study to test the association between preoperative subspecialty consultation and multiple clinically relevant outcomes in patients admitted with an acute hip fracture.1 All patients were comanaged by a hospitalist and orthopedic surgery, and “consultation” was defined as any preoperative subspecialty consultation requested by the hospitalist. Outcome measures included time to surgery, length of stay, readmission rate, perioperative complications, and 30-day mortality. In total, 36% (177/491) of patients who underwent surgery received a subspecialty preoperative consultation. Unsurprisingly, these patients were older with higher rates of comorbidity. After controlling for age and Charlson Comorbidity Index, preoperative consultation was associated with dramatic delays and increased rates of time to surgery >24 hours (adjusted odds ratio, 4.2; 95% CI: 2.8-6.6). The authors classified 90% of consultations as appropriate, either because of an active condition (eg, acute coronary syndrome) or because admitting physicians documented a perception that patients were at increased risk. However, 73% of consultations had only minor recommendations, such as ordering an ECG or changing the dose of an existing medication, and only 37% of the time did consultations lead to an identifiable change in management as a result of the consultation.
Although striking, integrating these findings into clinical practice is complex. As a retrospective study, patients who received consultations were obviously different from those who did not. The authors attempted to adjust for this but used only age and Charlson Comorbidity Index. Other factors that are both associated with consultations and known to increase mortality—such as frailty and functional status—were not included in their adjustment. Such unmeasured confounders possibly explain at least some, if not all, of the findings that consultations were associated with a doubling of the likelihood of 30-day mortality. In addition, although the authors assessed the appropriateness of consultation and degree of recommendations, their methods for this deserve scrutiny. Two independent providers adjudicated the consultations with excellent agreement (kappa 0.96 for indication, 0.95 for degree of recommendation), but this reliability assessment was done on previously extracted chart data, probably inflating their agreement statistics. Finally, the adjudication of consultant recommendations into minor, moderate, and major categories may oversimplify the outcome of each consultation. For example, all medication recommendations, regardless of type, were considered as minor, and recommendations were considered as major only if they resulted in invasive testing or procedures. This approach may underrepresent the impact of consultations as in clinical practice not all high-impact recommendations result in invasive testing or procedures. Despite these important limitations, Bellas et al. present a compelling case for preoperative consultation being associated with delays in surgery.
How then should this study change practice? The authors’ findings tell two separate but intertwined stories. The first is that preoperative consultation leads to delays in surgery. As patients who received preoperative consultation were obviously sicker, and because delays caused by consultation may lead to increased morbidity and mortality, perhaps the solution is to simply fix the delays. However, this approach ignores the more compelling story the authors tell. More important than the delays was the surprising lack of impact of preoperative consultations. Bellas et al. found that the majority of consultations resulted in only minor recommendations, and more importantly, hospitalists rarely changed treatment as a result. Although patients who received consultations were more ill, consultation rarely changed their care or decreased the risk posed by surgery. Bellas et al. found that only patients with active medical conditions had consultations, which resulted in moderate or major recommendations. These findings highlight an opportunity to better identify patients for whom consultation might be helpful and to prevent delays by avoiding consultation for those unlikely to benefit. There have been several efforts in the orthopedic literature to use guidelines for preoperative cardiac testing to guide cardiology consultation.4,5,6 One study using this approach reported findings that were extremely similar to those reported by Bellas et al. in that 71% of preoperative cardiology consultations in their institution did not meet the guideline criteria for invasive cardiac testing.7 The primary difference between the findings of Bellas et al. and the studies in the orthopedic literature is the presence of the comanaging hospitalist. As more and more patients receive hospitalist comanagement prior to inpatient surgery, it is well within the scope of the hospitalist to differentiate chronic risk factors from active or decompensated medical disease requiring a subspecialist. This is in fact much of the value that a hospitalist adds. Avoiding consultation for patients with only elevated chronic risk factors is an important first step in avoiding unnecessary delays to surgery and an opportunity for hospitalists to improve the care of the patients they comanage.
The goal of teaching trainees to “never worry alone” is to harness the feelings of uncertainty that all providers face to improve patient care. Knowing when to worry is a valuable lesson, but as with all skills, it should be applied thoughtfully and informed by evidence. Appreciating the risks that surgery poses is quintessential to safe perioperative care, but equally important is understanding that inappropriate consultations can create risks from needless delays and testing. Only in balancing these two concerns, and appreciating when it is appropriate to worry, can we provide the highest quality of care to our patients.
“Never worry alone” is a common mantra that most of us have heard throughout medical training. The premise is simple and well meaning. If a patient has an issue that concerns you, ask someone for help. As a student, this can be a resident; as a resident, this can be an attending. However, for hospitalists, the answer is often a subspecialty consultation. Asking for help never seems to be wrong, but what happens when our worry delays appropriate care with unnecessary consultations? In this month’s issue of the Journal of Hospital Medicine, authors Bellas et al. have investigated this issue through the lens of subspecialty preoperative consultation for patients admitted to a hospitalist comanagement service with a fragility hip fracture requiring surgery.1
Morbidity and mortality for patients who experience hip fractures are high, and time to appropriate surgery is one of the few modifiable risk factors that may reduce morbidity and mortality.2,3 Bellas et al. conducted a retrospective cohort study to test the association between preoperative subspecialty consultation and multiple clinically relevant outcomes in patients admitted with an acute hip fracture.1 All patients were comanaged by a hospitalist and orthopedic surgery, and “consultation” was defined as any preoperative subspecialty consultation requested by the hospitalist. Outcome measures included time to surgery, length of stay, readmission rate, perioperative complications, and 30-day mortality. In total, 36% (177/491) of patients who underwent surgery received a subspecialty preoperative consultation. Unsurprisingly, these patients were older with higher rates of comorbidity. After controlling for age and Charlson Comorbidity Index, preoperative consultation was associated with dramatic delays and increased rates of time to surgery >24 hours (adjusted odds ratio, 4.2; 95% CI: 2.8-6.6). The authors classified 90% of consultations as appropriate, either because of an active condition (eg, acute coronary syndrome) or because admitting physicians documented a perception that patients were at increased risk. However, 73% of consultations had only minor recommendations, such as ordering an ECG or changing the dose of an existing medication, and only 37% of the time did consultations lead to an identifiable change in management as a result of the consultation.
Although striking, integrating these findings into clinical practice is complex. As a retrospective study, patients who received consultations were obviously different from those who did not. The authors attempted to adjust for this but used only age and Charlson Comorbidity Index. Other factors that are both associated with consultations and known to increase mortality—such as frailty and functional status—were not included in their adjustment. Such unmeasured confounders possibly explain at least some, if not all, of the findings that consultations were associated with a doubling of the likelihood of 30-day mortality. In addition, although the authors assessed the appropriateness of consultation and degree of recommendations, their methods for this deserve scrutiny. Two independent providers adjudicated the consultations with excellent agreement (kappa 0.96 for indication, 0.95 for degree of recommendation), but this reliability assessment was done on previously extracted chart data, probably inflating their agreement statistics. Finally, the adjudication of consultant recommendations into minor, moderate, and major categories may oversimplify the outcome of each consultation. For example, all medication recommendations, regardless of type, were considered as minor, and recommendations were considered as major only if they resulted in invasive testing or procedures. This approach may underrepresent the impact of consultations as in clinical practice not all high-impact recommendations result in invasive testing or procedures. Despite these important limitations, Bellas et al. present a compelling case for preoperative consultation being associated with delays in surgery.
How then should this study change practice? The authors’ findings tell two separate but intertwined stories. The first is that preoperative consultation leads to delays in surgery. As patients who received preoperative consultation were obviously sicker, and because delays caused by consultation may lead to increased morbidity and mortality, perhaps the solution is to simply fix the delays. However, this approach ignores the more compelling story the authors tell. More important than the delays was the surprising lack of impact of preoperative consultations. Bellas et al. found that the majority of consultations resulted in only minor recommendations, and more importantly, hospitalists rarely changed treatment as a result. Although patients who received consultations were more ill, consultation rarely changed their care or decreased the risk posed by surgery. Bellas et al. found that only patients with active medical conditions had consultations, which resulted in moderate or major recommendations. These findings highlight an opportunity to better identify patients for whom consultation might be helpful and to prevent delays by avoiding consultation for those unlikely to benefit. There have been several efforts in the orthopedic literature to use guidelines for preoperative cardiac testing to guide cardiology consultation.4,5,6 One study using this approach reported findings that were extremely similar to those reported by Bellas et al. in that 71% of preoperative cardiology consultations in their institution did not meet the guideline criteria for invasive cardiac testing.7 The primary difference between the findings of Bellas et al. and the studies in the orthopedic literature is the presence of the comanaging hospitalist. As more and more patients receive hospitalist comanagement prior to inpatient surgery, it is well within the scope of the hospitalist to differentiate chronic risk factors from active or decompensated medical disease requiring a subspecialist. This is in fact much of the value that a hospitalist adds. Avoiding consultation for patients with only elevated chronic risk factors is an important first step in avoiding unnecessary delays to surgery and an opportunity for hospitalists to improve the care of the patients they comanage.
The goal of teaching trainees to “never worry alone” is to harness the feelings of uncertainty that all providers face to improve patient care. Knowing when to worry is a valuable lesson, but as with all skills, it should be applied thoughtfully and informed by evidence. Appreciating the risks that surgery poses is quintessential to safe perioperative care, but equally important is understanding that inappropriate consultations can create risks from needless delays and testing. Only in balancing these two concerns, and appreciating when it is appropriate to worry, can we provide the highest quality of care to our patients.
1. Bellas N, Stohler S, Staff I, et al. Impact of preoperative consults and hospitalist comanagement in hip fracture patients. J Hosp Med. 2020;15(1):16-21. https:doi.org/jhm.3264.
2. Goldacre MJ, Roberts SE, Yeates D. Mortality after admission to hospital with fractured neck of femur: database study. BMJ 2002;325(7369):868-869. https://doi.org/10.1136/bmj.325.7369.868.
3. Shiga T, Wajima Z, Ohe Y. Is operative delay associated with increased mortality of hip fracture patients? Systematic review, meta-analysis, and meta-regression. Can J Anaesth. 2008;55(3):146-154. https://doi.org/10.1007/BF03016088.
4. Cluett J, Caplan J, Yu W. Preoperative cardiac evaluation of patients with acute hip fracture. Am J Orthop. 2008;37(1):32-36.
5. Smeets SJ, Poeze M, Verbruggen JP. Preoperative cardiac evaluation of geriatric patients with hip fracture. Injury. 2012;43(12):2146-2151. https://doi.org/10.1016/j.injury.2012.08.007.
6. Siu CW, Sun NC, Lau TW, Yiu KH, Leung F, Tse HF. Preoperative cardiac risk assessment in geriatric patients with hip fractures: an orthopedic surgeons’ perspective. Osteoporos Int. 2010;21(Suppl 4):S587-S591. https://doi.org/10.1007/s00198-010-1393-0.
7. Stitgen A, Poludnianyk K, Dulaney-Cripe E, Markert R, Prayson M. Adherence to preoperative cardiac clearance guidelines in hip fracture patients. J Orthop Trauma 2015;29(11):500-503. https://doi.org/10.1097/BOT.0000000000000381.
1. Bellas N, Stohler S, Staff I, et al. Impact of preoperative consults and hospitalist comanagement in hip fracture patients. J Hosp Med. 2020;15(1):16-21. https:doi.org/jhm.3264.
2. Goldacre MJ, Roberts SE, Yeates D. Mortality after admission to hospital with fractured neck of femur: database study. BMJ 2002;325(7369):868-869. https://doi.org/10.1136/bmj.325.7369.868.
3. Shiga T, Wajima Z, Ohe Y. Is operative delay associated with increased mortality of hip fracture patients? Systematic review, meta-analysis, and meta-regression. Can J Anaesth. 2008;55(3):146-154. https://doi.org/10.1007/BF03016088.
4. Cluett J, Caplan J, Yu W. Preoperative cardiac evaluation of patients with acute hip fracture. Am J Orthop. 2008;37(1):32-36.
5. Smeets SJ, Poeze M, Verbruggen JP. Preoperative cardiac evaluation of geriatric patients with hip fracture. Injury. 2012;43(12):2146-2151. https://doi.org/10.1016/j.injury.2012.08.007.
6. Siu CW, Sun NC, Lau TW, Yiu KH, Leung F, Tse HF. Preoperative cardiac risk assessment in geriatric patients with hip fractures: an orthopedic surgeons’ perspective. Osteoporos Int. 2010;21(Suppl 4):S587-S591. https://doi.org/10.1007/s00198-010-1393-0.
7. Stitgen A, Poludnianyk K, Dulaney-Cripe E, Markert R, Prayson M. Adherence to preoperative cardiac clearance guidelines in hip fracture patients. J Orthop Trauma 2015;29(11):500-503. https://doi.org/10.1097/BOT.0000000000000381.
© 2020 Society of Hospital Medicine
Quantifying the Risks of Hospitalization—Is It Really as Safe as We Believe?
Even though I could not remember her name, I remembered her story, and I would bet that my colleagues did as well. She was someone that we had all cared for at one time or another. She frequently presented to the hospital with chest pain or shortness of breath attributable to a combination of longstanding congestive heart failure, chronic obstructive pulmonary disease, and cocaine abuse. But most tragic of all, she was homeless, which meant that she was frequently hospitalized not only for medical complaints but also for a night’s shelter and a bite of food. Even though she often refused medical treatment and social workers’ efforts to stabilize her housing situation, the staff in the emergency room and observation unit all knew her by name and greeted her like an old friend. And then one day she stopped showing up to the hospital. Sitting in the emergency department (ED), I overheard that she was found outside of a storefront and had passed away. Saddened by her death, which was not unexpected given her medical issues, I still wondered if we had done right by her during the hundreds of times that she had come to our hospital. Clinicians at busy safety-net hospitals face these questions every day, and it would seem beyond doubt that our duty is to address both medical and nonmedical determinants of health of everyone that walks through our door. But is this in fact the right thing to do? Is it possible that we unwittingly expose these vulnerable patients to risks from hospitalization alone?
In this month’s Journal of Hospital Medicine, Sekijima et al. sought to quantify precisely the risks of hospitalization, particularly among the subset of patients whose “severity” of medical problems alone might not have warranted hospital admission, a scenario known colloquially as a “social” admission.1 In real time, an inhouse triage physician classified patients as being admitted with or without “definite medical acuity.” Investigators retrospectively identified adverse events and illness acuity using standardized instruments, the Institute for Healthcare Improvement Global Trigger Tool and Emergency Severity Index, respectively. Despite the acknowledged differences in the patient population and the inherent subjectivity within the designation process, Sekijima et al. found no statistically significant difference in the percentage of admissions with an adverse event nor in the rate of adverse events per 1,000 patient days. Falls, oversedation/hypotension and code/arrest/rapid response activation were the most frequently encountered adverse events.
Delving deeper into the origin of admissions without definite medical acuity, the authors identified homelessness, lack of outpatient social support, substance use disorder, and lack of outpatient medical support as the most common reasons for “nonmedical” admissions. As healthcare providers, we recognize that these factors are generally long-term, chronic socioeconomic determinants of health. Despite our objective knowledge that we are limited in our ability to fix these problems on a short-term basis, the authors’ observations reflect our compulsion to try and help in any way possible. Patients admitted without definite medical acuity were more vulnerable and had higher rates of public insurance and housing insecurity. However, they were less acutely ill, as indicated by lower Emergency Severity Index scores. These factors were not associated with statistically significant differences in either 48-hour ED readmission or 30-day hospital readmission rates.
The process of appropriately triaging patients to an inpatient setting is challenging because of wide variability in both patients and ED providers. Hospitalists are increasingly recognized as an additional resource to assist in the triage process, as we are uniquely in a position to view the patient’s clinical presentation within the context of their anticipated clinical trajectory, promote effective utilization of inpatient bed availability, and anticipate potential barriers to discharge. Graduate medical education now identifies the triage process as a specific milestone within the transitions of care competency, as it requires mastery of interpersonal communication, professionalism, systems-based thinking, and patient-centered care.2 However, many institutions lack a dedicated faculty member to perform the triage role. Our institution recently examined the feasibility of instituting a daily “huddle” between the admitting hospitalist and the ED to facilitate interdepartmental communication to create care plans in patient triage and to promote patient throughput. Available admission beds are valuable commodities, and one challenge is that the ED makes disposition decisions without knowledge of the number of available beds in the hospital. The goal of the huddle was to quickly discuss all patients potentially requiring admission prior to the final disposition decision and to address any modifiable factors to potentially prevent a “social” admission with social work early in the day. Further work is in progress to determine if introducing flexibility within existing provider roles can improve the triage process in a measurable and efficient manner.
Many challenges remain as we balance the medical needs of patients with any potential social drivers that necessitate admission to the inpatient hospital setting. From an ED perspective, social support and community follow-up were “universally considered powerful influences on admission,” and other factors such as time of day, clinical volume, and the four-hour waiting time target also played a significant role in the decision to admit.3 Hunter et al. found that admissions with moderate to low acuity may be shorter or less costly,4 which presents an interesting question of cost-effectiveness as an avenue for further study. As clinicians, we are intuitively aware of the subjective risk of hospitalization itself, and this work provides new objective evidence that hospitalization confers specific and quantifiable risks. Though we can undoubtedly use this knowledge to guide internal decisions about admissions and discharges, do we also have an obligation to inform our patients about these risks in real time? Ultimately, hospitalization itself might be viewed as a “procedure” or intervention that has inherent risks for all who receive it, regardless of the individual patient or hospital characteristics. As hospitalists, we should continue to strive to reduce these risks, but we should also initiate a conversation about the risks and benefits of hospitalization similarly to how we discuss other procedures with patients and their families.
1. Sekijima A, Sunga C, Bann M. Adverse events experienced by patients hospitalized without definite medical acuity: A retrospective cohort study. J Hosp Med. 2020;15(1):42-45. https://doi.org/10.12788/jhm.3235.
2. Wang ES, Velásquez ST, Smith CJ, et al. Triaging inpatient admissions : An opportunity for resident education. J Gen Intern Med. 2019;34(5):754-757. https://doi.org/10.1007/s11606-019-04882-2.
3. Pope I, Burn H, Ismail SA, et al. A qualitative study exploring the factors influencing admission to hospital from the emergency department. BMJ Open. 2017;7(8):e011543. https://doi.org/10.1136/bmjopen-2016-011543.
4. Lewis Hunter AE, Spatz ES, Bernstein SL, Rosenthal MS. Factors influencing hospital admission of non-critically ill patients presenting to the emergency department: a cross-sectional study. J Gen Intern Med. 2016;31(1):37-44. https://doi.org/10.1007/s11606-015-3438-8.
Even though I could not remember her name, I remembered her story, and I would bet that my colleagues did as well. She was someone that we had all cared for at one time or another. She frequently presented to the hospital with chest pain or shortness of breath attributable to a combination of longstanding congestive heart failure, chronic obstructive pulmonary disease, and cocaine abuse. But most tragic of all, she was homeless, which meant that she was frequently hospitalized not only for medical complaints but also for a night’s shelter and a bite of food. Even though she often refused medical treatment and social workers’ efforts to stabilize her housing situation, the staff in the emergency room and observation unit all knew her by name and greeted her like an old friend. And then one day she stopped showing up to the hospital. Sitting in the emergency department (ED), I overheard that she was found outside of a storefront and had passed away. Saddened by her death, which was not unexpected given her medical issues, I still wondered if we had done right by her during the hundreds of times that she had come to our hospital. Clinicians at busy safety-net hospitals face these questions every day, and it would seem beyond doubt that our duty is to address both medical and nonmedical determinants of health of everyone that walks through our door. But is this in fact the right thing to do? Is it possible that we unwittingly expose these vulnerable patients to risks from hospitalization alone?
In this month’s Journal of Hospital Medicine, Sekijima et al. sought to quantify precisely the risks of hospitalization, particularly among the subset of patients whose “severity” of medical problems alone might not have warranted hospital admission, a scenario known colloquially as a “social” admission.1 In real time, an inhouse triage physician classified patients as being admitted with or without “definite medical acuity.” Investigators retrospectively identified adverse events and illness acuity using standardized instruments, the Institute for Healthcare Improvement Global Trigger Tool and Emergency Severity Index, respectively. Despite the acknowledged differences in the patient population and the inherent subjectivity within the designation process, Sekijima et al. found no statistically significant difference in the percentage of admissions with an adverse event nor in the rate of adverse events per 1,000 patient days. Falls, oversedation/hypotension and code/arrest/rapid response activation were the most frequently encountered adverse events.
Delving deeper into the origin of admissions without definite medical acuity, the authors identified homelessness, lack of outpatient social support, substance use disorder, and lack of outpatient medical support as the most common reasons for “nonmedical” admissions. As healthcare providers, we recognize that these factors are generally long-term, chronic socioeconomic determinants of health. Despite our objective knowledge that we are limited in our ability to fix these problems on a short-term basis, the authors’ observations reflect our compulsion to try and help in any way possible. Patients admitted without definite medical acuity were more vulnerable and had higher rates of public insurance and housing insecurity. However, they were less acutely ill, as indicated by lower Emergency Severity Index scores. These factors were not associated with statistically significant differences in either 48-hour ED readmission or 30-day hospital readmission rates.
The process of appropriately triaging patients to an inpatient setting is challenging because of wide variability in both patients and ED providers. Hospitalists are increasingly recognized as an additional resource to assist in the triage process, as we are uniquely in a position to view the patient’s clinical presentation within the context of their anticipated clinical trajectory, promote effective utilization of inpatient bed availability, and anticipate potential barriers to discharge. Graduate medical education now identifies the triage process as a specific milestone within the transitions of care competency, as it requires mastery of interpersonal communication, professionalism, systems-based thinking, and patient-centered care.2 However, many institutions lack a dedicated faculty member to perform the triage role. Our institution recently examined the feasibility of instituting a daily “huddle” between the admitting hospitalist and the ED to facilitate interdepartmental communication to create care plans in patient triage and to promote patient throughput. Available admission beds are valuable commodities, and one challenge is that the ED makes disposition decisions without knowledge of the number of available beds in the hospital. The goal of the huddle was to quickly discuss all patients potentially requiring admission prior to the final disposition decision and to address any modifiable factors to potentially prevent a “social” admission with social work early in the day. Further work is in progress to determine if introducing flexibility within existing provider roles can improve the triage process in a measurable and efficient manner.
Many challenges remain as we balance the medical needs of patients with any potential social drivers that necessitate admission to the inpatient hospital setting. From an ED perspective, social support and community follow-up were “universally considered powerful influences on admission,” and other factors such as time of day, clinical volume, and the four-hour waiting time target also played a significant role in the decision to admit.3 Hunter et al. found that admissions with moderate to low acuity may be shorter or less costly,4 which presents an interesting question of cost-effectiveness as an avenue for further study. As clinicians, we are intuitively aware of the subjective risk of hospitalization itself, and this work provides new objective evidence that hospitalization confers specific and quantifiable risks. Though we can undoubtedly use this knowledge to guide internal decisions about admissions and discharges, do we also have an obligation to inform our patients about these risks in real time? Ultimately, hospitalization itself might be viewed as a “procedure” or intervention that has inherent risks for all who receive it, regardless of the individual patient or hospital characteristics. As hospitalists, we should continue to strive to reduce these risks, but we should also initiate a conversation about the risks and benefits of hospitalization similarly to how we discuss other procedures with patients and their families.
Even though I could not remember her name, I remembered her story, and I would bet that my colleagues did as well. She was someone that we had all cared for at one time or another. She frequently presented to the hospital with chest pain or shortness of breath attributable to a combination of longstanding congestive heart failure, chronic obstructive pulmonary disease, and cocaine abuse. But most tragic of all, she was homeless, which meant that she was frequently hospitalized not only for medical complaints but also for a night’s shelter and a bite of food. Even though she often refused medical treatment and social workers’ efforts to stabilize her housing situation, the staff in the emergency room and observation unit all knew her by name and greeted her like an old friend. And then one day she stopped showing up to the hospital. Sitting in the emergency department (ED), I overheard that she was found outside of a storefront and had passed away. Saddened by her death, which was not unexpected given her medical issues, I still wondered if we had done right by her during the hundreds of times that she had come to our hospital. Clinicians at busy safety-net hospitals face these questions every day, and it would seem beyond doubt that our duty is to address both medical and nonmedical determinants of health of everyone that walks through our door. But is this in fact the right thing to do? Is it possible that we unwittingly expose these vulnerable patients to risks from hospitalization alone?
In this month’s Journal of Hospital Medicine, Sekijima et al. sought to quantify precisely the risks of hospitalization, particularly among the subset of patients whose “severity” of medical problems alone might not have warranted hospital admission, a scenario known colloquially as a “social” admission.1 In real time, an inhouse triage physician classified patients as being admitted with or without “definite medical acuity.” Investigators retrospectively identified adverse events and illness acuity using standardized instruments, the Institute for Healthcare Improvement Global Trigger Tool and Emergency Severity Index, respectively. Despite the acknowledged differences in the patient population and the inherent subjectivity within the designation process, Sekijima et al. found no statistically significant difference in the percentage of admissions with an adverse event nor in the rate of adverse events per 1,000 patient days. Falls, oversedation/hypotension and code/arrest/rapid response activation were the most frequently encountered adverse events.
Delving deeper into the origin of admissions without definite medical acuity, the authors identified homelessness, lack of outpatient social support, substance use disorder, and lack of outpatient medical support as the most common reasons for “nonmedical” admissions. As healthcare providers, we recognize that these factors are generally long-term, chronic socioeconomic determinants of health. Despite our objective knowledge that we are limited in our ability to fix these problems on a short-term basis, the authors’ observations reflect our compulsion to try and help in any way possible. Patients admitted without definite medical acuity were more vulnerable and had higher rates of public insurance and housing insecurity. However, they were less acutely ill, as indicated by lower Emergency Severity Index scores. These factors were not associated with statistically significant differences in either 48-hour ED readmission or 30-day hospital readmission rates.
The process of appropriately triaging patients to an inpatient setting is challenging because of wide variability in both patients and ED providers. Hospitalists are increasingly recognized as an additional resource to assist in the triage process, as we are uniquely in a position to view the patient’s clinical presentation within the context of their anticipated clinical trajectory, promote effective utilization of inpatient bed availability, and anticipate potential barriers to discharge. Graduate medical education now identifies the triage process as a specific milestone within the transitions of care competency, as it requires mastery of interpersonal communication, professionalism, systems-based thinking, and patient-centered care.2 However, many institutions lack a dedicated faculty member to perform the triage role. Our institution recently examined the feasibility of instituting a daily “huddle” between the admitting hospitalist and the ED to facilitate interdepartmental communication to create care plans in patient triage and to promote patient throughput. Available admission beds are valuable commodities, and one challenge is that the ED makes disposition decisions without knowledge of the number of available beds in the hospital. The goal of the huddle was to quickly discuss all patients potentially requiring admission prior to the final disposition decision and to address any modifiable factors to potentially prevent a “social” admission with social work early in the day. Further work is in progress to determine if introducing flexibility within existing provider roles can improve the triage process in a measurable and efficient manner.
Many challenges remain as we balance the medical needs of patients with any potential social drivers that necessitate admission to the inpatient hospital setting. From an ED perspective, social support and community follow-up were “universally considered powerful influences on admission,” and other factors such as time of day, clinical volume, and the four-hour waiting time target also played a significant role in the decision to admit.3 Hunter et al. found that admissions with moderate to low acuity may be shorter or less costly,4 which presents an interesting question of cost-effectiveness as an avenue for further study. As clinicians, we are intuitively aware of the subjective risk of hospitalization itself, and this work provides new objective evidence that hospitalization confers specific and quantifiable risks. Though we can undoubtedly use this knowledge to guide internal decisions about admissions and discharges, do we also have an obligation to inform our patients about these risks in real time? Ultimately, hospitalization itself might be viewed as a “procedure” or intervention that has inherent risks for all who receive it, regardless of the individual patient or hospital characteristics. As hospitalists, we should continue to strive to reduce these risks, but we should also initiate a conversation about the risks and benefits of hospitalization similarly to how we discuss other procedures with patients and their families.
1. Sekijima A, Sunga C, Bann M. Adverse events experienced by patients hospitalized without definite medical acuity: A retrospective cohort study. J Hosp Med. 2020;15(1):42-45. https://doi.org/10.12788/jhm.3235.
2. Wang ES, Velásquez ST, Smith CJ, et al. Triaging inpatient admissions : An opportunity for resident education. J Gen Intern Med. 2019;34(5):754-757. https://doi.org/10.1007/s11606-019-04882-2.
3. Pope I, Burn H, Ismail SA, et al. A qualitative study exploring the factors influencing admission to hospital from the emergency department. BMJ Open. 2017;7(8):e011543. https://doi.org/10.1136/bmjopen-2016-011543.
4. Lewis Hunter AE, Spatz ES, Bernstein SL, Rosenthal MS. Factors influencing hospital admission of non-critically ill patients presenting to the emergency department: a cross-sectional study. J Gen Intern Med. 2016;31(1):37-44. https://doi.org/10.1007/s11606-015-3438-8.
1. Sekijima A, Sunga C, Bann M. Adverse events experienced by patients hospitalized without definite medical acuity: A retrospective cohort study. J Hosp Med. 2020;15(1):42-45. https://doi.org/10.12788/jhm.3235.
2. Wang ES, Velásquez ST, Smith CJ, et al. Triaging inpatient admissions : An opportunity for resident education. J Gen Intern Med. 2019;34(5):754-757. https://doi.org/10.1007/s11606-019-04882-2.
3. Pope I, Burn H, Ismail SA, et al. A qualitative study exploring the factors influencing admission to hospital from the emergency department. BMJ Open. 2017;7(8):e011543. https://doi.org/10.1136/bmjopen-2016-011543.
4. Lewis Hunter AE, Spatz ES, Bernstein SL, Rosenthal MS. Factors influencing hospital admission of non-critically ill patients presenting to the emergency department: a cross-sectional study. J Gen Intern Med. 2016;31(1):37-44. https://doi.org/10.1007/s11606-015-3438-8.
© 2020 Society of Hospital Medicine