User login
ERRATUM: Decreasing Hypoglycemia following Insulin Administration for Inpatient Hyperkalemia
A correction has been made to the Figure. A dosage was incorrect in the Orderset 1.1 (1/1/16-3/19/17) box. The figure listed Insulin 19 Units IV x 1 and should have been Insulin 10 Units IV x 1. Below is the corrected figure..

A correction has been made to the Figure. A dosage was incorrect in the Orderset 1.1 (1/1/16-3/19/17) box. The figure listed Insulin 19 Units IV x 1 and should have been Insulin 10 Units IV x 1. Below is the corrected figure..

A correction has been made to the Figure. A dosage was incorrect in the Orderset 1.1 (1/1/16-3/19/17) box. The figure listed Insulin 19 Units IV x 1 and should have been Insulin 10 Units IV x 1. Below is the corrected figure..

Things We Do for No Reason™: Obtaining an Abdominal X-ray to Assess for Constipation in Children

Inspired by the ABIM Foundation’s Choosing Wisely® campaign, the “Things We Do for No Reason™” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent clear-cut conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
CLINICAL SCENARIO
A 5-year old boy is admitted to the hospital for a bowel clean-out after presenting with abdominal pain and having an abdominal x-ray that demonstrated a “moderate stool burden.” After ingestion of the bowel prep, he develops worsening abdominal cramping and diarrhea. Upon reviewing the bowel history with his mother afterward, the team learns that he has had a bowel movement every 1-2 days as usual and has been having soft stools without any straining, pain, or blood present.
BACKGROUND
Functional constipation is a common clinical problem in pediatrics and constitutes a large number of admissions into hospitals and visits to clinics and emergency departments. In the United States, up to 36% of children are affected.Associated healthcare costs for children with constipation are estimated at $5.9 billion per year, which is $3.9 billion more than the general pediatric population without constipation.1 In 2011, American children aged 17 years and younger had more than 270,000 visits to the emergency department for constipation.2 As many as 70% of children who are given a diagnosis of constipation in the emergency department have an abdominal x-ray completed.3 The carcinogenic effects of radiation from radiography are well known. Unnecessary imaging places the child at risk for these effects while adding to the overall cost of medical care.4
WHY AN ABDOMINAL X-RAY MAY SEEM HELPFUL
The overall utilization of diagnostic imaging is increasing in pediatric emergency departments.4 When questioning why this is the case, one should consider the method of problem solving used by most physicians. After formulating initial hypotheses based on available information, prior knowledge, and experience, physicians aim to obtain additional data to confirm or reject each hypothesis.5
A 2017 study surveyed 24 pediatric gastroenterologists after 72 patient encounters and found that the most common cause for obtaining an abdominal x-ray was for evaluation of stool burden (70%).5 Other reasons included assessing the need for a bowel clean-out (35%), diagnosing fecal impaction (27%), finding the cause for abdominal pain (24%), and demonstrating stool burden to a family (14%). This same study found that most of the polled providers used an abdominal x-ray to assess for constipation, and nearly half changed their management based on the findings.
WHY ABDOMINAL X-RAYS ARE NOT HELPFUL
Many systematic reviews and retrospective studies have investigated the efficacy of abdominal x-rays for diagnosing constipation. One retrospective review involving 160 children with defecation complaints assessed the accuracy of different radiologic scoring methods in identifying children with constipation.6 Three pediatric gastroenterologists and 1 pediatric radiologist blindly applied 4 scoring methods: colonic transit time, Leech score, Barr score, and fecal loading. The results showed that all x-ray scoring methods had low sensitivity for diagnosing constipation, variable specificity, and low interobserver reproducibility of scores.6 There was also poor ability to differentiate between patients with constipation and nonretentive fecal incontinence. Fecal loading had the worst performance in differentiating between these 2. Greater than 20% of children with clinically diagnosed constipation had normal Barr and Leech scores.6 Another systematic review also found no evidence for a diagnostic association between clinical symptoms of constipation and fecal loading on abdominal x-rays.7 In this study, the sensitivity and specificity of the x-ray were as low as 61% and 55%, respectively, which indicate poor overall diagnostic accuracy. Abdominal x-rays are subjective, not standardized, and represent a single observation in time. The amount of fecal loading seen on imaging is subject to daily variation depending on the timing of last food intake and timing of last defecation. There is a large variance in interpretation of fecal loading, and any stool seen on an x-ray does not rule out another potential diagnosis causing abdominal pain.
In 2014, the North American Society for Pediatric Gastroenterology, Hepatology, & Nutrition (NASPGHAN) and the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) released joint clinical guidelines that the evidence supports not obtaining an abdominal x-ray to diagnose functional constipation.8 Similarly, the National Institute for Health and Care Excellence (NICE) stated that abdominal x-rays should not be recommended as an additional test for constipation in children.9 These groups advocate for diagnosing functional constipation clinically by using a careful history and physical exam.
WHY ABDOMINAL X-RAYS ARE POTENTIALLY HARMFUL
The primary patient harm associated with abdominal x-rays is radiation exposure. While the amount of radiation in a single x-ray is low, children with constipation tend to have frequent revisits, significantly more than children with other common chronic conditions (eg, asthma and migraine headaches).10
One multicenter retrospective cohort study that included approximately 282,000 children diagnosed with constipation found that children who received an abdominal x-ray were twice as likely to return to the emergency department with a clinically significant alternate diagnosis (0.33% vs 0.17%). The 2 most common missed diagnoses were acute appendicitis and intussusception.3 Another retrospective study that included about 3,700 children also found that x-rays were performed more frequently in children who were misdiagnosed than in those who did not have a significant alternate diagnosis (75% vs 46%).11 In this case, both of these groups had a similar amount of stool on the x-rays as determined by the mean Leech scores. While this study identified an association between abdominal x-ray use and misdiagnoses, a causative effect was not necessarily discovered between the 2. The authors felt that even relatively large amounts of stool on an x-ray should not discount serious causes of abdominal pain or tenderness.11 A third retrospective study determined that children who received an abdominal x-ray and were diagnosed with constipation were significantly more likely to be admitted to the hospital, further raising healthcare costs.12 In this study, having an x-ray reduced the odds of being discharged home by about half. They also found that abdominal x-rays could be avoided if digital rectal exams were performed.12
HOW CONSTIPATION SHOULD BE DIAGNOSED
Functional constipation is a clinical diagnosis based on a thorough collection of history and a complete physical exam in children of all ages, including digital examination of the rectum to assess for fecal impaction, if necessary.
The Rome IV criteria for chronic constipation can be helpful and includes at least 2 of the following features for at least 1 month in infants up to 4 years of age: 2 or fewer stools per week, history of excessive stool retention, history of painful or hard bowel movements, history of large-diameter stools, and presence of a large fecal mass in the rectum.13 In children who are toilet trained, 2 additional criteria may be used: at least 1 episode of fecal incontinence per week after being toilet-trained and history of large-diameter stools that may obstruct the toilet.13
The NASPGHAN and ESPGHAN joint guidelines from 2014 state that, while constipation is based on history and physical exam, a major role of the history and physical exam is to exclude other disorders that also present with difficulty in defecation.8 This can help identify red-flag features or complications and guide further investigation. While evidence did not support routine use of a digital rectal exam in diagnosing constipation, the guidelines stated that a rectal exam (visual and digital) helps to evaluate for anorectal malformations, anal stenosis, rectal tone, distension, erythema, skin tags, anal fissures, or a fecal mass.8
In regard to history, approximately 0.4%-20% of healthy children without constipation have at least 1 clinical feature listed above. Therefore, the use of a single clinical finding to diagnose constipation, such as decreased bowel frequency, can result in an inappropriate diagnosis. Children experience large variations in stool output depending on diet, genetics, and environmental factors.10 The usual pattern of bowel habits in humans range from 3 times daily to every 3 days.14 Importantly, there are times to order an abdominal x-ray for patients with abdominal pain. The NASPGHAN and ESPGHAN joint guidelines recommend obtaining abdominal x-rays to evaluate children who have concerning features, such as previous abdominal surgeries, known genetic conditions or malformations, bilious emesis, or severe abdominal distension.8
RECOMMENDATIONS
- Functional constipation should be diagnosed based purely on a thorough history and physical examination, including a rectal exam
- Abdominal x-rays (ordered for any reason) should not be used to diagnose or assess for functional constipation
CONCLUSIONS
Performing abdominal x-rays to assess for pediatric functional constipation is not beneficial and potentially harmful to patients. Multiple retrospective studies revealed no diagnostic association between clinical symptoms or severity of constipation and findings on abdominal radiography. X-rays have very low sensitivity and specificity for diagnosing constipation. In the pediatric emergency department, abdominal x-rays completed for patients diagnosed with constipation have been associated with missed diagnoses, false reassurance of constipation, more frequent admissions into the hospital, longer hospital stays, higher healthcare costs, and unnecessary radiation exposure. The NICE as well as 2014 NASPGHAN and ESPGHAN clinical guidelines recommend against obtaining x-rays to diagnose constipation. The most effective way to diagnose functional constipation in children is with a thorough collection of history and physical exam. In the introductory case, the boy received an osmotic laxative based on abdominal x-ray findings, which resulted in the adverse effect of diarrhea. This case demonstrates how using abdominal x-rays to assess for constipation can be misleading and emphasizes the importance of collecting a thorough history and physical exam.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason™”? Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason™” topics by emailing [email protected].
1. Afzal NA, Tighe MP, Thomson MA. (2011, June 13). Constipation in children. Ital J Pediatr. 2011;37:28. https://doi.org/10.1186/1824-7288-37-28.
2. Sommers T, Corban C, Sengupta N, et al. Emergency department burden of constipation in the United States from 2006 to 2011. Am J Gastroenterol. 2015;110(4):572-579. https://doi.org/10.1038/ajg.2015.64.
3. Freedman SB, Rodean J, Hall M, et al. (2017). Delayed diagnoses in children with constipation: multicenter retrospective cohort study. J Pediatr. 186, 87-94.e16. https://doi.org/10.1016/j.jpeds.2017.03.061.
4. Reed MH. Imaging utilization commentary: A radiology perspective. Pediatr Radiol. 2008;38 (Suppl 4):S660-S663. https://doi.org/10.1007/s00247-008-0982-y.
5. Beinvogl B, Sabharwal S, McSweeney M, Nurko S. Are we using abdominal radiographs appropriately in the management of pediatric constipation? J Pediatr. 2017;191:179-183. https://doi.org/10.1016/j.jpeds.2017.08.075.
6. Pensabene L, Buonomo C, Fishman L, Chitkara D, Nurko S. Lack of utility of abdominal x-rays in the evaluation of children with constipation: Comparison of different scoring methods. J Pediatr Gastroenterol Nutr. 2010;51(2):155-159. https://doi.org/10.1097/MPG.0b013e3181cb4309.
7. Berger MY, Tabbers MM, Kurver MJ, Boluyt N, Benninga MA. Value of abdominal radiography, colonic transit time, and rectal ultrasound scanning in the diagnosis of idiopathic constipation in children: A systematic review. J Pediatr. 2012;161(1):44–50.e502. https://doi.org/10.1016/j.jpeds.2011.12.045.
8. Tabbers MM, DiLorenzo C, Berger MY, et al. Evaluation and treatment of functional constipation in infants and children: Evidence-based recommendations from ESPGHAN and NASPGHAN. J Pediatr Gastroenterol Nutr. 2014;58(2):258-274. https://doi.org/10.1097/mpg.0000000000000266.
9. Bardisa-Ezcurra L, Ullman R, Gordon J; Guideline Development Group. Diagnosis and management of idiopathic childhood constipation: summary of NICE guidance. BMJ. 2010;340:c2585. https://doi.org/10.1136/bmj.c2585.
10. Rajindrajith S, Manjuri Devanarayana N, Benninga MA. Defecation Disorders in Children: Constipation and Functional Fecal Incontinence. In: Guandalini S, Dhawan A, Branski D. eds. Textbook of Pediatric Gastroenterology, Hepatology and Nutrition: A Comprehensive Guide to Practice (1st ed.). Basingstoke, England: Springer; 2016:247-260.
11. Freedman SB, Thull-Freedman J, Manson D, et al. Pediatric abdominal radiograph use, constipation, and significant misdiagnoses. J Pediatr. 2014;164(1):83-88.e2. https://doi.org/10.1016/j.jpeds.2013.08.074.
12. Chumpitazi CE, Rees CA, Camp EA, Henkel EB, Valdez KL, Chumpitazi BP. Diagnostic approach to constipation impacts pediatric emergency department disposition. Am J Emerg Med. 2017;35(10):1490-1493. https://doi.org/10.1016/j.ajem.2017.04.060.
13. Benninga MA, Nurko S, Faure C, Hyman PE, St. James Roberts I, Schechter NL. Childhood functional GI disorders: Neonate/toddler. Gastroenterology. 2016;150(6):1443-1455. https://doi.org/10.1053/j.gastro.2016.02.016.
14. Walter SA, Kjellström L, Nyhlin H, Talley NJ, Agréus L. Assessment of normal bowel habits in the general adult population: the Popcol study. Scand J Gastroenterol. 2010;45(5):556-566. https://doi.org/10.3109/00365520903551332.

Inspired by the ABIM Foundation’s Choosing Wisely® campaign, the “Things We Do for No Reason™” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent clear-cut conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
CLINICAL SCENARIO
A 5-year old boy is admitted to the hospital for a bowel clean-out after presenting with abdominal pain and having an abdominal x-ray that demonstrated a “moderate stool burden.” After ingestion of the bowel prep, he develops worsening abdominal cramping and diarrhea. Upon reviewing the bowel history with his mother afterward, the team learns that he has had a bowel movement every 1-2 days as usual and has been having soft stools without any straining, pain, or blood present.
BACKGROUND
Functional constipation is a common clinical problem in pediatrics and constitutes a large number of admissions into hospitals and visits to clinics and emergency departments. In the United States, up to 36% of children are affected.Associated healthcare costs for children with constipation are estimated at $5.9 billion per year, which is $3.9 billion more than the general pediatric population without constipation.1 In 2011, American children aged 17 years and younger had more than 270,000 visits to the emergency department for constipation.2 As many as 70% of children who are given a diagnosis of constipation in the emergency department have an abdominal x-ray completed.3 The carcinogenic effects of radiation from radiography are well known. Unnecessary imaging places the child at risk for these effects while adding to the overall cost of medical care.4
WHY AN ABDOMINAL X-RAY MAY SEEM HELPFUL
The overall utilization of diagnostic imaging is increasing in pediatric emergency departments.4 When questioning why this is the case, one should consider the method of problem solving used by most physicians. After formulating initial hypotheses based on available information, prior knowledge, and experience, physicians aim to obtain additional data to confirm or reject each hypothesis.5
A 2017 study surveyed 24 pediatric gastroenterologists after 72 patient encounters and found that the most common cause for obtaining an abdominal x-ray was for evaluation of stool burden (70%).5 Other reasons included assessing the need for a bowel clean-out (35%), diagnosing fecal impaction (27%), finding the cause for abdominal pain (24%), and demonstrating stool burden to a family (14%). This same study found that most of the polled providers used an abdominal x-ray to assess for constipation, and nearly half changed their management based on the findings.
WHY ABDOMINAL X-RAYS ARE NOT HELPFUL
Many systematic reviews and retrospective studies have investigated the efficacy of abdominal x-rays for diagnosing constipation. One retrospective review involving 160 children with defecation complaints assessed the accuracy of different radiologic scoring methods in identifying children with constipation.6 Three pediatric gastroenterologists and 1 pediatric radiologist blindly applied 4 scoring methods: colonic transit time, Leech score, Barr score, and fecal loading. The results showed that all x-ray scoring methods had low sensitivity for diagnosing constipation, variable specificity, and low interobserver reproducibility of scores.6 There was also poor ability to differentiate between patients with constipation and nonretentive fecal incontinence. Fecal loading had the worst performance in differentiating between these 2. Greater than 20% of children with clinically diagnosed constipation had normal Barr and Leech scores.6 Another systematic review also found no evidence for a diagnostic association between clinical symptoms of constipation and fecal loading on abdominal x-rays.7 In this study, the sensitivity and specificity of the x-ray were as low as 61% and 55%, respectively, which indicate poor overall diagnostic accuracy. Abdominal x-rays are subjective, not standardized, and represent a single observation in time. The amount of fecal loading seen on imaging is subject to daily variation depending on the timing of last food intake and timing of last defecation. There is a large variance in interpretation of fecal loading, and any stool seen on an x-ray does not rule out another potential diagnosis causing abdominal pain.
In 2014, the North American Society for Pediatric Gastroenterology, Hepatology, & Nutrition (NASPGHAN) and the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) released joint clinical guidelines that the evidence supports not obtaining an abdominal x-ray to diagnose functional constipation.8 Similarly, the National Institute for Health and Care Excellence (NICE) stated that abdominal x-rays should not be recommended as an additional test for constipation in children.9 These groups advocate for diagnosing functional constipation clinically by using a careful history and physical exam.
WHY ABDOMINAL X-RAYS ARE POTENTIALLY HARMFUL
The primary patient harm associated with abdominal x-rays is radiation exposure. While the amount of radiation in a single x-ray is low, children with constipation tend to have frequent revisits, significantly more than children with other common chronic conditions (eg, asthma and migraine headaches).10
One multicenter retrospective cohort study that included approximately 282,000 children diagnosed with constipation found that children who received an abdominal x-ray were twice as likely to return to the emergency department with a clinically significant alternate diagnosis (0.33% vs 0.17%). The 2 most common missed diagnoses were acute appendicitis and intussusception.3 Another retrospective study that included about 3,700 children also found that x-rays were performed more frequently in children who were misdiagnosed than in those who did not have a significant alternate diagnosis (75% vs 46%).11 In this case, both of these groups had a similar amount of stool on the x-rays as determined by the mean Leech scores. While this study identified an association between abdominal x-ray use and misdiagnoses, a causative effect was not necessarily discovered between the 2. The authors felt that even relatively large amounts of stool on an x-ray should not discount serious causes of abdominal pain or tenderness.11 A third retrospective study determined that children who received an abdominal x-ray and were diagnosed with constipation were significantly more likely to be admitted to the hospital, further raising healthcare costs.12 In this study, having an x-ray reduced the odds of being discharged home by about half. They also found that abdominal x-rays could be avoided if digital rectal exams were performed.12
HOW CONSTIPATION SHOULD BE DIAGNOSED
Functional constipation is a clinical diagnosis based on a thorough collection of history and a complete physical exam in children of all ages, including digital examination of the rectum to assess for fecal impaction, if necessary.
The Rome IV criteria for chronic constipation can be helpful and includes at least 2 of the following features for at least 1 month in infants up to 4 years of age: 2 or fewer stools per week, history of excessive stool retention, history of painful or hard bowel movements, history of large-diameter stools, and presence of a large fecal mass in the rectum.13 In children who are toilet trained, 2 additional criteria may be used: at least 1 episode of fecal incontinence per week after being toilet-trained and history of large-diameter stools that may obstruct the toilet.13
The NASPGHAN and ESPGHAN joint guidelines from 2014 state that, while constipation is based on history and physical exam, a major role of the history and physical exam is to exclude other disorders that also present with difficulty in defecation.8 This can help identify red-flag features or complications and guide further investigation. While evidence did not support routine use of a digital rectal exam in diagnosing constipation, the guidelines stated that a rectal exam (visual and digital) helps to evaluate for anorectal malformations, anal stenosis, rectal tone, distension, erythema, skin tags, anal fissures, or a fecal mass.8
In regard to history, approximately 0.4%-20% of healthy children without constipation have at least 1 clinical feature listed above. Therefore, the use of a single clinical finding to diagnose constipation, such as decreased bowel frequency, can result in an inappropriate diagnosis. Children experience large variations in stool output depending on diet, genetics, and environmental factors.10 The usual pattern of bowel habits in humans range from 3 times daily to every 3 days.14 Importantly, there are times to order an abdominal x-ray for patients with abdominal pain. The NASPGHAN and ESPGHAN joint guidelines recommend obtaining abdominal x-rays to evaluate children who have concerning features, such as previous abdominal surgeries, known genetic conditions or malformations, bilious emesis, or severe abdominal distension.8
RECOMMENDATIONS
- Functional constipation should be diagnosed based purely on a thorough history and physical examination, including a rectal exam
- Abdominal x-rays (ordered for any reason) should not be used to diagnose or assess for functional constipation
CONCLUSIONS
Performing abdominal x-rays to assess for pediatric functional constipation is not beneficial and potentially harmful to patients. Multiple retrospective studies revealed no diagnostic association between clinical symptoms or severity of constipation and findings on abdominal radiography. X-rays have very low sensitivity and specificity for diagnosing constipation. In the pediatric emergency department, abdominal x-rays completed for patients diagnosed with constipation have been associated with missed diagnoses, false reassurance of constipation, more frequent admissions into the hospital, longer hospital stays, higher healthcare costs, and unnecessary radiation exposure. The NICE as well as 2014 NASPGHAN and ESPGHAN clinical guidelines recommend against obtaining x-rays to diagnose constipation. The most effective way to diagnose functional constipation in children is with a thorough collection of history and physical exam. In the introductory case, the boy received an osmotic laxative based on abdominal x-ray findings, which resulted in the adverse effect of diarrhea. This case demonstrates how using abdominal x-rays to assess for constipation can be misleading and emphasizes the importance of collecting a thorough history and physical exam.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason™”? Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason™” topics by emailing [email protected].

Inspired by the ABIM Foundation’s Choosing Wisely® campaign, the “Things We Do for No Reason™” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent clear-cut conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
CLINICAL SCENARIO
A 5-year old boy is admitted to the hospital for a bowel clean-out after presenting with abdominal pain and having an abdominal x-ray that demonstrated a “moderate stool burden.” After ingestion of the bowel prep, he develops worsening abdominal cramping and diarrhea. Upon reviewing the bowel history with his mother afterward, the team learns that he has had a bowel movement every 1-2 days as usual and has been having soft stools without any straining, pain, or blood present.
BACKGROUND
Functional constipation is a common clinical problem in pediatrics and constitutes a large number of admissions into hospitals and visits to clinics and emergency departments. In the United States, up to 36% of children are affected.Associated healthcare costs for children with constipation are estimated at $5.9 billion per year, which is $3.9 billion more than the general pediatric population without constipation.1 In 2011, American children aged 17 years and younger had more than 270,000 visits to the emergency department for constipation.2 As many as 70% of children who are given a diagnosis of constipation in the emergency department have an abdominal x-ray completed.3 The carcinogenic effects of radiation from radiography are well known. Unnecessary imaging places the child at risk for these effects while adding to the overall cost of medical care.4
WHY AN ABDOMINAL X-RAY MAY SEEM HELPFUL
The overall utilization of diagnostic imaging is increasing in pediatric emergency departments.4 When questioning why this is the case, one should consider the method of problem solving used by most physicians. After formulating initial hypotheses based on available information, prior knowledge, and experience, physicians aim to obtain additional data to confirm or reject each hypothesis.5
A 2017 study surveyed 24 pediatric gastroenterologists after 72 patient encounters and found that the most common cause for obtaining an abdominal x-ray was for evaluation of stool burden (70%).5 Other reasons included assessing the need for a bowel clean-out (35%), diagnosing fecal impaction (27%), finding the cause for abdominal pain (24%), and demonstrating stool burden to a family (14%). This same study found that most of the polled providers used an abdominal x-ray to assess for constipation, and nearly half changed their management based on the findings.
WHY ABDOMINAL X-RAYS ARE NOT HELPFUL
Many systematic reviews and retrospective studies have investigated the efficacy of abdominal x-rays for diagnosing constipation. One retrospective review involving 160 children with defecation complaints assessed the accuracy of different radiologic scoring methods in identifying children with constipation.6 Three pediatric gastroenterologists and 1 pediatric radiologist blindly applied 4 scoring methods: colonic transit time, Leech score, Barr score, and fecal loading. The results showed that all x-ray scoring methods had low sensitivity for diagnosing constipation, variable specificity, and low interobserver reproducibility of scores.6 There was also poor ability to differentiate between patients with constipation and nonretentive fecal incontinence. Fecal loading had the worst performance in differentiating between these 2. Greater than 20% of children with clinically diagnosed constipation had normal Barr and Leech scores.6 Another systematic review also found no evidence for a diagnostic association between clinical symptoms of constipation and fecal loading on abdominal x-rays.7 In this study, the sensitivity and specificity of the x-ray were as low as 61% and 55%, respectively, which indicate poor overall diagnostic accuracy. Abdominal x-rays are subjective, not standardized, and represent a single observation in time. The amount of fecal loading seen on imaging is subject to daily variation depending on the timing of last food intake and timing of last defecation. There is a large variance in interpretation of fecal loading, and any stool seen on an x-ray does not rule out another potential diagnosis causing abdominal pain.
In 2014, the North American Society for Pediatric Gastroenterology, Hepatology, & Nutrition (NASPGHAN) and the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) released joint clinical guidelines that the evidence supports not obtaining an abdominal x-ray to diagnose functional constipation.8 Similarly, the National Institute for Health and Care Excellence (NICE) stated that abdominal x-rays should not be recommended as an additional test for constipation in children.9 These groups advocate for diagnosing functional constipation clinically by using a careful history and physical exam.
WHY ABDOMINAL X-RAYS ARE POTENTIALLY HARMFUL
The primary patient harm associated with abdominal x-rays is radiation exposure. While the amount of radiation in a single x-ray is low, children with constipation tend to have frequent revisits, significantly more than children with other common chronic conditions (eg, asthma and migraine headaches).10
One multicenter retrospective cohort study that included approximately 282,000 children diagnosed with constipation found that children who received an abdominal x-ray were twice as likely to return to the emergency department with a clinically significant alternate diagnosis (0.33% vs 0.17%). The 2 most common missed diagnoses were acute appendicitis and intussusception.3 Another retrospective study that included about 3,700 children also found that x-rays were performed more frequently in children who were misdiagnosed than in those who did not have a significant alternate diagnosis (75% vs 46%).11 In this case, both of these groups had a similar amount of stool on the x-rays as determined by the mean Leech scores. While this study identified an association between abdominal x-ray use and misdiagnoses, a causative effect was not necessarily discovered between the 2. The authors felt that even relatively large amounts of stool on an x-ray should not discount serious causes of abdominal pain or tenderness.11 A third retrospective study determined that children who received an abdominal x-ray and were diagnosed with constipation were significantly more likely to be admitted to the hospital, further raising healthcare costs.12 In this study, having an x-ray reduced the odds of being discharged home by about half. They also found that abdominal x-rays could be avoided if digital rectal exams were performed.12
HOW CONSTIPATION SHOULD BE DIAGNOSED
Functional constipation is a clinical diagnosis based on a thorough collection of history and a complete physical exam in children of all ages, including digital examination of the rectum to assess for fecal impaction, if necessary.
The Rome IV criteria for chronic constipation can be helpful and includes at least 2 of the following features for at least 1 month in infants up to 4 years of age: 2 or fewer stools per week, history of excessive stool retention, history of painful or hard bowel movements, history of large-diameter stools, and presence of a large fecal mass in the rectum.13 In children who are toilet trained, 2 additional criteria may be used: at least 1 episode of fecal incontinence per week after being toilet-trained and history of large-diameter stools that may obstruct the toilet.13
The NASPGHAN and ESPGHAN joint guidelines from 2014 state that, while constipation is based on history and physical exam, a major role of the history and physical exam is to exclude other disorders that also present with difficulty in defecation.8 This can help identify red-flag features or complications and guide further investigation. While evidence did not support routine use of a digital rectal exam in diagnosing constipation, the guidelines stated that a rectal exam (visual and digital) helps to evaluate for anorectal malformations, anal stenosis, rectal tone, distension, erythema, skin tags, anal fissures, or a fecal mass.8
In regard to history, approximately 0.4%-20% of healthy children without constipation have at least 1 clinical feature listed above. Therefore, the use of a single clinical finding to diagnose constipation, such as decreased bowel frequency, can result in an inappropriate diagnosis. Children experience large variations in stool output depending on diet, genetics, and environmental factors.10 The usual pattern of bowel habits in humans range from 3 times daily to every 3 days.14 Importantly, there are times to order an abdominal x-ray for patients with abdominal pain. The NASPGHAN and ESPGHAN joint guidelines recommend obtaining abdominal x-rays to evaluate children who have concerning features, such as previous abdominal surgeries, known genetic conditions or malformations, bilious emesis, or severe abdominal distension.8
RECOMMENDATIONS
- Functional constipation should be diagnosed based purely on a thorough history and physical examination, including a rectal exam
- Abdominal x-rays (ordered for any reason) should not be used to diagnose or assess for functional constipation
CONCLUSIONS
Performing abdominal x-rays to assess for pediatric functional constipation is not beneficial and potentially harmful to patients. Multiple retrospective studies revealed no diagnostic association between clinical symptoms or severity of constipation and findings on abdominal radiography. X-rays have very low sensitivity and specificity for diagnosing constipation. In the pediatric emergency department, abdominal x-rays completed for patients diagnosed with constipation have been associated with missed diagnoses, false reassurance of constipation, more frequent admissions into the hospital, longer hospital stays, higher healthcare costs, and unnecessary radiation exposure. The NICE as well as 2014 NASPGHAN and ESPGHAN clinical guidelines recommend against obtaining x-rays to diagnose constipation. The most effective way to diagnose functional constipation in children is with a thorough collection of history and physical exam. In the introductory case, the boy received an osmotic laxative based on abdominal x-ray findings, which resulted in the adverse effect of diarrhea. This case demonstrates how using abdominal x-rays to assess for constipation can be misleading and emphasizes the importance of collecting a thorough history and physical exam.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason™”? Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason™” topics by emailing [email protected].
1. Afzal NA, Tighe MP, Thomson MA. (2011, June 13). Constipation in children. Ital J Pediatr. 2011;37:28. https://doi.org/10.1186/1824-7288-37-28.
2. Sommers T, Corban C, Sengupta N, et al. Emergency department burden of constipation in the United States from 2006 to 2011. Am J Gastroenterol. 2015;110(4):572-579. https://doi.org/10.1038/ajg.2015.64.
3. Freedman SB, Rodean J, Hall M, et al. (2017). Delayed diagnoses in children with constipation: multicenter retrospective cohort study. J Pediatr. 186, 87-94.e16. https://doi.org/10.1016/j.jpeds.2017.03.061.
4. Reed MH. Imaging utilization commentary: A radiology perspective. Pediatr Radiol. 2008;38 (Suppl 4):S660-S663. https://doi.org/10.1007/s00247-008-0982-y.
5. Beinvogl B, Sabharwal S, McSweeney M, Nurko S. Are we using abdominal radiographs appropriately in the management of pediatric constipation? J Pediatr. 2017;191:179-183. https://doi.org/10.1016/j.jpeds.2017.08.075.
6. Pensabene L, Buonomo C, Fishman L, Chitkara D, Nurko S. Lack of utility of abdominal x-rays in the evaluation of children with constipation: Comparison of different scoring methods. J Pediatr Gastroenterol Nutr. 2010;51(2):155-159. https://doi.org/10.1097/MPG.0b013e3181cb4309.
7. Berger MY, Tabbers MM, Kurver MJ, Boluyt N, Benninga MA. Value of abdominal radiography, colonic transit time, and rectal ultrasound scanning in the diagnosis of idiopathic constipation in children: A systematic review. J Pediatr. 2012;161(1):44–50.e502. https://doi.org/10.1016/j.jpeds.2011.12.045.
8. Tabbers MM, DiLorenzo C, Berger MY, et al. Evaluation and treatment of functional constipation in infants and children: Evidence-based recommendations from ESPGHAN and NASPGHAN. J Pediatr Gastroenterol Nutr. 2014;58(2):258-274. https://doi.org/10.1097/mpg.0000000000000266.
9. Bardisa-Ezcurra L, Ullman R, Gordon J; Guideline Development Group. Diagnosis and management of idiopathic childhood constipation: summary of NICE guidance. BMJ. 2010;340:c2585. https://doi.org/10.1136/bmj.c2585.
10. Rajindrajith S, Manjuri Devanarayana N, Benninga MA. Defecation Disorders in Children: Constipation and Functional Fecal Incontinence. In: Guandalini S, Dhawan A, Branski D. eds. Textbook of Pediatric Gastroenterology, Hepatology and Nutrition: A Comprehensive Guide to Practice (1st ed.). Basingstoke, England: Springer; 2016:247-260.
11. Freedman SB, Thull-Freedman J, Manson D, et al. Pediatric abdominal radiograph use, constipation, and significant misdiagnoses. J Pediatr. 2014;164(1):83-88.e2. https://doi.org/10.1016/j.jpeds.2013.08.074.
12. Chumpitazi CE, Rees CA, Camp EA, Henkel EB, Valdez KL, Chumpitazi BP. Diagnostic approach to constipation impacts pediatric emergency department disposition. Am J Emerg Med. 2017;35(10):1490-1493. https://doi.org/10.1016/j.ajem.2017.04.060.
13. Benninga MA, Nurko S, Faure C, Hyman PE, St. James Roberts I, Schechter NL. Childhood functional GI disorders: Neonate/toddler. Gastroenterology. 2016;150(6):1443-1455. https://doi.org/10.1053/j.gastro.2016.02.016.
14. Walter SA, Kjellström L, Nyhlin H, Talley NJ, Agréus L. Assessment of normal bowel habits in the general adult population: the Popcol study. Scand J Gastroenterol. 2010;45(5):556-566. https://doi.org/10.3109/00365520903551332.
1. Afzal NA, Tighe MP, Thomson MA. (2011, June 13). Constipation in children. Ital J Pediatr. 2011;37:28. https://doi.org/10.1186/1824-7288-37-28.
2. Sommers T, Corban C, Sengupta N, et al. Emergency department burden of constipation in the United States from 2006 to 2011. Am J Gastroenterol. 2015;110(4):572-579. https://doi.org/10.1038/ajg.2015.64.
3. Freedman SB, Rodean J, Hall M, et al. (2017). Delayed diagnoses in children with constipation: multicenter retrospective cohort study. J Pediatr. 186, 87-94.e16. https://doi.org/10.1016/j.jpeds.2017.03.061.
4. Reed MH. Imaging utilization commentary: A radiology perspective. Pediatr Radiol. 2008;38 (Suppl 4):S660-S663. https://doi.org/10.1007/s00247-008-0982-y.
5. Beinvogl B, Sabharwal S, McSweeney M, Nurko S. Are we using abdominal radiographs appropriately in the management of pediatric constipation? J Pediatr. 2017;191:179-183. https://doi.org/10.1016/j.jpeds.2017.08.075.
6. Pensabene L, Buonomo C, Fishman L, Chitkara D, Nurko S. Lack of utility of abdominal x-rays in the evaluation of children with constipation: Comparison of different scoring methods. J Pediatr Gastroenterol Nutr. 2010;51(2):155-159. https://doi.org/10.1097/MPG.0b013e3181cb4309.
7. Berger MY, Tabbers MM, Kurver MJ, Boluyt N, Benninga MA. Value of abdominal radiography, colonic transit time, and rectal ultrasound scanning in the diagnosis of idiopathic constipation in children: A systematic review. J Pediatr. 2012;161(1):44–50.e502. https://doi.org/10.1016/j.jpeds.2011.12.045.
8. Tabbers MM, DiLorenzo C, Berger MY, et al. Evaluation and treatment of functional constipation in infants and children: Evidence-based recommendations from ESPGHAN and NASPGHAN. J Pediatr Gastroenterol Nutr. 2014;58(2):258-274. https://doi.org/10.1097/mpg.0000000000000266.
9. Bardisa-Ezcurra L, Ullman R, Gordon J; Guideline Development Group. Diagnosis and management of idiopathic childhood constipation: summary of NICE guidance. BMJ. 2010;340:c2585. https://doi.org/10.1136/bmj.c2585.
10. Rajindrajith S, Manjuri Devanarayana N, Benninga MA. Defecation Disorders in Children: Constipation and Functional Fecal Incontinence. In: Guandalini S, Dhawan A, Branski D. eds. Textbook of Pediatric Gastroenterology, Hepatology and Nutrition: A Comprehensive Guide to Practice (1st ed.). Basingstoke, England: Springer; 2016:247-260.
11. Freedman SB, Thull-Freedman J, Manson D, et al. Pediatric abdominal radiograph use, constipation, and significant misdiagnoses. J Pediatr. 2014;164(1):83-88.e2. https://doi.org/10.1016/j.jpeds.2013.08.074.
12. Chumpitazi CE, Rees CA, Camp EA, Henkel EB, Valdez KL, Chumpitazi BP. Diagnostic approach to constipation impacts pediatric emergency department disposition. Am J Emerg Med. 2017;35(10):1490-1493. https://doi.org/10.1016/j.ajem.2017.04.060.
13. Benninga MA, Nurko S, Faure C, Hyman PE, St. James Roberts I, Schechter NL. Childhood functional GI disorders: Neonate/toddler. Gastroenterology. 2016;150(6):1443-1455. https://doi.org/10.1053/j.gastro.2016.02.016.
14. Walter SA, Kjellström L, Nyhlin H, Talley NJ, Agréus L. Assessment of normal bowel habits in the general adult population: the Popcol study. Scand J Gastroenterol. 2010;45(5):556-566. https://doi.org/10.3109/00365520903551332.
© 2020 Society of Hospital Medicine
A Traumatic Traveler
A 19-year-old man with Duchenne Muscular Dystrophy (DMD) presented to the Emergency Department (ED) for left knee pain after ejection from his motorized wheelchair at a low velocity. In the ED, he developed increasing respiratory distress.
When addressing a new problem in a patient with a chronic condition, it is crucial to first understand the chronic condition and then consider whether the presenting symptoms relate to that condition or stem from an unrelated inciting event.
Patients with DMD are at risk of pulmonary complications relating to their underlying disease. For instance, dysphagia and ineffective cough can predispose them to recurrent aspiration pneumonitis and/or pneumonia, whereas decreased lung compliance (from scoliosis, atelectasis, and/or pulmonary fibrosis) and respiratory muscle weakness can progress to ventilatory failure. In addition, patients with DMD are at risk for pulmonary thromboembolism in the setting of immobility. Patients with DMD may also develop congestive heart failure resulting from myocardial fibrosis and nonischemic cardiomyopathy.
The ejection from his wheelchair signals potential trauma-associated conditions that could explain his respiratory distress. Respiratory complications of blunt thoracic trauma include pulmonary contusion, pneumothorax, flail chest (resulting from fractured ribs), and acute respiratory distress syndrome (ARDS). Lower extremity injury can result in venous thrombosis and pulmonary thromboembolism. While classically associated with long bone fractures, fat embolism syndrome (FES) may rarely occur with rib fractures and soft-tissue trauma. Respiratory compromise may also result from cervical spinal cord injury or severe anemia from trauma-associated hemorrhage.
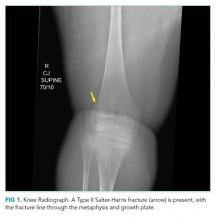
Additional past medical history included growth hormone deficiency, migraine headaches, osteoporosis secondary to chronic steroid use, cardiac fibrosis of the inferolateral wall and septum with a baseline left ventricular ejection fraction of 65%, and atrial fibrillation. His medications included calcium carbonate, vitamin D, omeprazole, lisinopril, metoprolol, prednisone, escitalopram, and testosterone. Physical examination revealed an ill-appearing obese man in respiratory distress. Temperature was 37.3°C, heart rate was 102 beats per minute (bpm), blood pressure was 110/74 mm Hg. His oxygen saturation was 93% with a respiratory rate of 25 breaths per minute while breathing ambient air. His lung sounds were clear, and his heart was without murmur. The left knee was diffusely tender to palpation without specific point tenderness. Strength was 2/5 with flexion and extension at the bilateral knees and hips and 3/5 flexion and extension at the bilateral elbows. He reported this level of weakness was his baseline. Radiographs revealed a minimally displaced Salter Harris II fracture (fracture line through the metaphysis and growth plate) of the left distal femur. His fracture was splinted early in his ED course. During his ED evaluation, the patient had acute worsening of tachycardia to 130 bpm, increased respiratory rate of 34 breaths per minute, and hypoxemia with an oxygen saturation of 83% on ambient air. He was placed on 3 L/min of oxygen via nasal cannula with improvement in his oxygen saturation to 90%. A chest radiograph was unremarkable, without evidence of pneumothorax, effusion, or pneumonia. The patient was admitted to the hospital.
The acute onset of tachypnea, tachycardia, and hypoxia, accompanied by a clear lung exam and normal chest radiograph, increases the likelihood of a pulmonary embolism. Obesity, testosterone therapy, and trauma increase his susceptibility to venous thromboembolism, while a distal femur fracture increases his risk for FES. Acute pulmonary aspiration often presents with initially absent or subtle radiographic findings. An arterial blood gas analysis would determine the presence and extent of an alveolar-arterial (A-a) gradient; a normal A-a gradient is seen in hypoventilation, while an elevated A-a gradient is seen in conditions affecting gas exchange, including pulmonary emboli and alveolar filling processes. His hypoxemia only partially corrects with supplemental oxygen, raising the possibility of capillary or anatomic shunting. Capillary shunting may occur with atelectasis, aspiration/pneumonia and pulmonary edema, whereas anatomic shunting can be intra-cardiac (eg, patent foramen ovale or septic defect) or intrapulmonary (eg, arteriovenous malformations). Patients with pulmonary emboli may also develop right-to-left shunting because of increased pulmonary vascular resistance, although hypoxemia with pulmonary emboli largely relates to ventilation/perfusion mismatch and decreased level of mixed venous blood oxygen (PvO2).
This patient’s complex medical history warrants a broadened differential with consideration of his cardiac history, including myocardial fibrosis and arrhythmia, and the impact of exposure to steroids on his immune and musculoskeletal systems. He has a history of atrial fibrillation, and an electrocardiogram is warranted to determine the underlying rhythm. Prolonged periods of rapid ventricular response may lead to tachycardia-induced cardiomyopathy. Myocardial fibrosis may progress despite use of angiotensin-converting enzyme inhibitors and is associated with systolic and/or diastolic dysfunction, although neither the examination findings provided nor the chest radiograph are suggestive of decompensated heart failure. Chronic exposure to corticosteroids (used in DMD to improve muscle strength and function) may predispose to numerous infectious and metabolic complications. Up to 10%-15% of patients with Pneumocystis jirovecii pneumonia may present with a normal chest radiograph. Acute adrenal insufficiency can present with tachycardia, weakness, and respiratory distress, so recent prednisone dose changes or interruptions should be assessed.
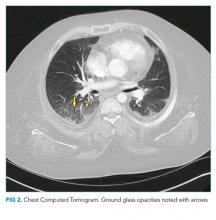
The patient’s respiratory status worsened. In light of his complex medical history, he was transferred to a children’s hospital for a higher level of care with a presumptive diagnosis of aspiration pneumonia. Upon reassessment at the new facility, the patient reported an ongoing and severe headache since his initial injury. NSAIDs had been given prior to transfer. His exam continued to be significant for tachycardia, tachypnea, and hypoxemia. His cardiac and lung examinations were otherwise normal. A comprehensive metabolic panel, procalcitonin, complete blood count with differential, and lactate were normal; his C-reactive protein (CRP) was 46.8 mg/dL (Normal <8 mg/dL). A computed tomography (CT) angiogram of the chest revealed small multifocal nodular ground-glass opacities, especially in the lower lobes, concerning for microatelectasis, multifocal pneumonia, or aspiration pneumonia. After consultation with pediatric pulmonology consultants, antimicrobials were held during the initial phase of work-up.
His headache may reflect a migraine, although further characterization and assessment for the presence and extent of head or neck trauma is warranted. Headache following trauma warrants consideration of cerebral contusion, diffuse axonal injury, intracranial hemorrhage, and carotid or vertebral artery dissection. Screening for concussion should also be performed. Hypoxemia may increase cerebral blood flow and raise intracranial pressure, resulting in headache.
CRP elevation is nonspecific and signals the presence of focal or systemic inflammation and is often elevated to a milder extent in obese patients with DMD. While normal procalcitonin argues against bacterial pneumonia, the precise level can be informative, and serial procalcitonin values may be more helpful than a single value. Although antecedent respiratory symptoms were not mentioned, viral or fungal pneumonia can present insidiously. An occult malignancy may be incidentally discovered when patients present for unrelated issues, although this and other sources of elevated CRP (eg, exacerbation of an autoimmune disease or drug reaction) remain less likely given the acuity of his presentation. Acute pulmonary embolism may be associated with a systemic inflammatory response and elevation in CRP.
In addition to the radiographic differential diagnosis already presented, the appearance of multifocal opacifications with hypoxemia raises the possibility of pulmonary infarcts or noncardiogenic pulmonary edema.
On hospital day 2, the patient continued to complain of “the worst headache of his life” as well as blurry vision and seeing “dark spots.” His headache did not improve with NSAIDs. A noncontrast CT scan of the head was normal. Neurology was consulted. Given his symptoms, history of migraines, stable neurological examination, and normal head CT, he was diagnosed with migraines and administered fluids, prochlorperazine, diphenhydramine, ondansetron, and NSAIDs. His headache continued and he continued to require supplemental oxygen.
The combination of hypoxemia, severe headache, and vision changes remains consistent with systemic emboli caused by thromboembolism or fat embolism. Headache assessment must also involve screening for “red flags,” which include sudden onset, antecedent head trauma, systemic illness (eg, fever or meningismus), focal neurologic findings, papilledema, changes with position or Valsalva, and immunosuppression. Although primary headache syndromes (eg, migraines or tension and cluster headache) may be triggered in the setting of trauma and systemic illness, “the worst headache of my life” is a concerning symptom that warrants urgent attention. While this invokes the possibility of a subarachnoid hemorrhage (SAH), headache severity is nonspecific, and rapid onset (ie, thunderclap headache) would be more suggestive. After 6 hours of symptoms, the sensitivity of head CT for detecting SAH declines, and lumbar puncture would be warranted to evaluate for xanthochromia.
His blurry vision and dark spots require testing of visual acuity and visual fields, as well as fundoscopic examination to assess for embolic phenomena or papilledema. Migraine is classically associated with “positive” or scintillating scotomata, although dark spots may occur. The presence of horizontal diplopia would indicate a cranial nerve VI palsy, which can occur with increased intracranial pressure. Visual-field cuts may also present as blurry vision, and monocular vs binocular deficits signal whether the issue is anterior or involving/posterior to the optic chiasm, respectively. Magnetic resonance imaging (MRI) may reveal the presence or sequelae of cerebral emboli (eg, fat emboli), including vasogenic edema.
Dilated fundus examination revealed Purtscher retinopathy: bilateral cotton-wool spots and larger areas of retinal whitening (Purtscher flecken).
Typical findings of Purtscher retinopathy include Purtscher flecken, cotton-wool spots, retinal hemorrhage, and optic disc edema. Purtscher retinopathy is classically associated with severe head trauma. Without associated head trauma, the term “Purtscher-like retinopathy” is used. Conditions that can cause Purtscher-like retinopathy include pancreatitis, vasculitis, microangiopathy, chronic renal failure, and systemic embolization. The most likely source of systemic embolization remains fat emboli stemming from his femur fracture. Treatment of FES is largely supportive.
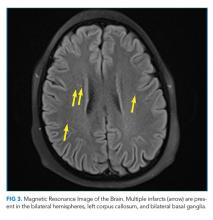
The possibility of fat emboli had been repeatedly raised by the patient’s mother since admission. While providers had considered this a possibility, it was discounted early on because of the minor nature of the patient’s orthopedic trauma, the lack of clear radiographic evidence for pulmonary emboli on chest CT, and the normal head CT. The findings on the ophthalmologist’s fundoscopic examination led the primary team to reconsider FES, along with thromboemboli and pancreatitis. Lipase was normal. MRI of the brain with contrast revealed >20 microinfarcts in the bilateral hemispheres, left corpus callosum, and bilateral basal ganglia. The CT angiogram of the chest was rereviewed; the pediatric radiologists suggested that microinfarcts could explain the patchy small ground glass opacities seen in the lungs. A transthoracic echocardiogram and electrocardiogram were normal. The diagnosis of FES was made, and the patient was started on aspirin and enoxaparin prophylaxis. His headache and respiratory status improved, and he was discharged home with close follow-up.
DISCUSSION
FES is a rare complication associated with long bone fractures and orthopedic manipulation.1,2 The exact mechanism of fat emboli production is unknown, but two theories prevail. The mechanical theory states that an outside mechanical source causes bone marrow contents or adipose tissue contents to be dislodged into the circulation where they travel through the venous circulation to become embedded in the lungs.1,2 These fragments may also migrate to the arterial circulation, through a patent foramen ovale or intrapulmonary shunts, leading to end organ damage.1,2 The biochemical theory suggests that fat emboli in the venous circulation precipitate an inflammatory and prothrombotic cascade that triggers fibrin production, platelet aggregation, and release of free fatty acids into the circulation, predisposing patients to develop multifocal systemic emboli.1
Although the classic triad in FES includes respiratory symptoms, rash, and CNS symptoms, all three findings are only present in 1%-29% of cases.1,2 Respiratory abnormalities, ranging from tachypnea and dyspnea to ARDS and hypoxic respiratory failure, occur in up to 75% of patients with FES.1 Central nervous system (CNS) complications, including headache, confusion, coma, seizures, and death caused by cerebral ischemia, occur in up to 86% of patients.1,2 Petechiae may occur in 20%-60% of patients and are usually located on nondependent regions of the body such as the head, neck, and chest.
Diagnosis of FES is largely clinical and requires a high index of suspicion and elimination of other conditions, including pulmonary thromboembolism, diffuse intravascular coagulation, and sepsis. The CNS complications must be differentiated from CNS infection, stroke, migraine, benign intracranial hypertension, and intracranial hemorrhage. There is no gold standard test for diagnosis. The Gurd and Wilson criteria, modified Gurd criteria, and Schonfeld’s criteria (Table) are commonly used but have not been clinically validated.1,3-5 These use a combination of clinical signs of respiratory distress, neurological symptoms, petechial rash, and various other diagnostic factors. When patients have risk factors, such as trauma, surgery, or predisposing conditions (eg, mobility-limiting neuromuscular disorders) and signs and symptoms potentially consistent with FES, the diagnostic evaluation should include arterial blood gas analysis, complete blood count, chest radiographs, and coagulation studies. Ophthalmological exam to evaluate for cotton-wool spots, brain MRI to detect cerebral emboli, and CT pulmonary angiogram to assess for pulmonary infarcts may help to eliminate other diagnoses and/or confirm FES.
Diagnosis of uncommon conditions that present with nonspecific symptoms, like FES, can be challenging because the symptoms may overlap with many other possible diagnoses. This challenge is further exacerbated in patients with significant medical complexity, as with the patient discussed here. Specifically, this patient had multiple plausible explanations for CNS symptoms and respiratory symptoms. It was ultimately the visual symptoms that began to link his symptoms together into a unifying syndrome and the ophthalmologic examination that prompted confirmatory imaging. It is important to continually revisit and revise the differential diagnosis in patients with medical complexity and avoid the competing temptations to abandon the search for a unifying diagnosis and attribute all symptoms to a patient’s underlying condition.
Treatment of FES is largely supportive with close monitoring of neurological status and providing supplemental oxygen as needed. Corticosteroids have been suggested to help prevent FES in patients with long bone fractures, but there is no evidence to suggest they are helpful once FES is diagnosed.2 There is conflicting evidence for the efficacy of heparin or low-molecular-weight heparin as treatment in FES.2,6 After discussions with consulting physician teams, the patient, and his family, enoxaparin and aspirin were started for this patient in light of his tenuous condition in order to decrease the risk of further embolic complications.
Patients with DMD and other neuromuscular disorders likely have a greater propensity to develop FES even after minor trauma.1,6 This is believed to be caused by patients becoming nonambulatory early in life and receiving substantial corticosteroid therapy, which can lead to osteopenia and fatty replacement of the bone marrow.1,6 This population is also often obese by the second or third decade of life, which contributes to their already increased propensity to fall.1,6
To our knowledge, this patient is 1 of 18 reported cases of FES after trauma in DMD patients. Two-thirds of these cases occurred when an unrestrained patient fell from their wheelchair. The other cases occurred while walking, during physical therapy, and during assisted transfers.6-12 In these cases, FES had a guarded prognosis, with 7 of the 18 patients dying and 1 of the patients remaining in a persistent vegetative state.8,9 While caution is warranted in generalizing these findings, given the small number of reported cases and likely publication bias,education of caregivers and patients on use of restraints and safe transfers is paramount to limit the risk of trauma.
TEACHING POINTS
- FES is a rare condition that most commonly manifests with respiratory, neurological, and cutaneous findings.
- Patients with Duchenne’s Muscular Dystrophy are likely at increased risk for FES even with minor trauma; this makes wheelchair restraints and safe transfers fundamental.
- Patients with medical complexity and their caregivers are key members of the diagnostic team.
1. Fukumoto LE, Fukumoto KD. Fat embolism syndrome. Nurs Clin North Am. 2018;53(3):335-347. https://doi.org/10.1016/j.cnur.2018.04.003.
2. Scarpino M, Lanzo G, Lolli F, Grippo A. From the diagnosis to the therapeutic management: Cerebral fat embolism, a clinical challenge. Int J Gen Med. 2019;2019(12):39-48. https://doi.org/10.2147/IJGM.S177407.
A 19-year-old man with Duchenne Muscular Dystrophy (DMD) presented to the Emergency Department (ED) for left knee pain after ejection from his motorized wheelchair at a low velocity. In the ED, he developed increasing respiratory distress.
When addressing a new problem in a patient with a chronic condition, it is crucial to first understand the chronic condition and then consider whether the presenting symptoms relate to that condition or stem from an unrelated inciting event.
Patients with DMD are at risk of pulmonary complications relating to their underlying disease. For instance, dysphagia and ineffective cough can predispose them to recurrent aspiration pneumonitis and/or pneumonia, whereas decreased lung compliance (from scoliosis, atelectasis, and/or pulmonary fibrosis) and respiratory muscle weakness can progress to ventilatory failure. In addition, patients with DMD are at risk for pulmonary thromboembolism in the setting of immobility. Patients with DMD may also develop congestive heart failure resulting from myocardial fibrosis and nonischemic cardiomyopathy.
The ejection from his wheelchair signals potential trauma-associated conditions that could explain his respiratory distress. Respiratory complications of blunt thoracic trauma include pulmonary contusion, pneumothorax, flail chest (resulting from fractured ribs), and acute respiratory distress syndrome (ARDS). Lower extremity injury can result in venous thrombosis and pulmonary thromboembolism. While classically associated with long bone fractures, fat embolism syndrome (FES) may rarely occur with rib fractures and soft-tissue trauma. Respiratory compromise may also result from cervical spinal cord injury or severe anemia from trauma-associated hemorrhage.
Additional past medical history included growth hormone deficiency, migraine headaches, osteoporosis secondary to chronic steroid use, cardiac fibrosis of the inferolateral wall and septum with a baseline left ventricular ejection fraction of 65%, and atrial fibrillation. His medications included calcium carbonate, vitamin D, omeprazole, lisinopril, metoprolol, prednisone, escitalopram, and testosterone. Physical examination revealed an ill-appearing obese man in respiratory distress. Temperature was 37.3°C, heart rate was 102 beats per minute (bpm), blood pressure was 110/74 mm Hg. His oxygen saturation was 93% with a respiratory rate of 25 breaths per minute while breathing ambient air. His lung sounds were clear, and his heart was without murmur. The left knee was diffusely tender to palpation without specific point tenderness. Strength was 2/5 with flexion and extension at the bilateral knees and hips and 3/5 flexion and extension at the bilateral elbows. He reported this level of weakness was his baseline. Radiographs revealed a minimally displaced Salter Harris II fracture (fracture line through the metaphysis and growth plate) of the left distal femur. His fracture was splinted early in his ED course. During his ED evaluation, the patient had acute worsening of tachycardia to 130 bpm, increased respiratory rate of 34 breaths per minute, and hypoxemia with an oxygen saturation of 83% on ambient air. He was placed on 3 L/min of oxygen via nasal cannula with improvement in his oxygen saturation to 90%. A chest radiograph was unremarkable, without evidence of pneumothorax, effusion, or pneumonia. The patient was admitted to the hospital.
The acute onset of tachypnea, tachycardia, and hypoxia, accompanied by a clear lung exam and normal chest radiograph, increases the likelihood of a pulmonary embolism. Obesity, testosterone therapy, and trauma increase his susceptibility to venous thromboembolism, while a distal femur fracture increases his risk for FES. Acute pulmonary aspiration often presents with initially absent or subtle radiographic findings. An arterial blood gas analysis would determine the presence and extent of an alveolar-arterial (A-a) gradient; a normal A-a gradient is seen in hypoventilation, while an elevated A-a gradient is seen in conditions affecting gas exchange, including pulmonary emboli and alveolar filling processes. His hypoxemia only partially corrects with supplemental oxygen, raising the possibility of capillary or anatomic shunting. Capillary shunting may occur with atelectasis, aspiration/pneumonia and pulmonary edema, whereas anatomic shunting can be intra-cardiac (eg, patent foramen ovale or septic defect) or intrapulmonary (eg, arteriovenous malformations). Patients with pulmonary emboli may also develop right-to-left shunting because of increased pulmonary vascular resistance, although hypoxemia with pulmonary emboli largely relates to ventilation/perfusion mismatch and decreased level of mixed venous blood oxygen (PvO2).
This patient’s complex medical history warrants a broadened differential with consideration of his cardiac history, including myocardial fibrosis and arrhythmia, and the impact of exposure to steroids on his immune and musculoskeletal systems. He has a history of atrial fibrillation, and an electrocardiogram is warranted to determine the underlying rhythm. Prolonged periods of rapid ventricular response may lead to tachycardia-induced cardiomyopathy. Myocardial fibrosis may progress despite use of angiotensin-converting enzyme inhibitors and is associated with systolic and/or diastolic dysfunction, although neither the examination findings provided nor the chest radiograph are suggestive of decompensated heart failure. Chronic exposure to corticosteroids (used in DMD to improve muscle strength and function) may predispose to numerous infectious and metabolic complications. Up to 10%-15% of patients with Pneumocystis jirovecii pneumonia may present with a normal chest radiograph. Acute adrenal insufficiency can present with tachycardia, weakness, and respiratory distress, so recent prednisone dose changes or interruptions should be assessed.
The patient’s respiratory status worsened. In light of his complex medical history, he was transferred to a children’s hospital for a higher level of care with a presumptive diagnosis of aspiration pneumonia. Upon reassessment at the new facility, the patient reported an ongoing and severe headache since his initial injury. NSAIDs had been given prior to transfer. His exam continued to be significant for tachycardia, tachypnea, and hypoxemia. His cardiac and lung examinations were otherwise normal. A comprehensive metabolic panel, procalcitonin, complete blood count with differential, and lactate were normal; his C-reactive protein (CRP) was 46.8 mg/dL (Normal <8 mg/dL). A computed tomography (CT) angiogram of the chest revealed small multifocal nodular ground-glass opacities, especially in the lower lobes, concerning for microatelectasis, multifocal pneumonia, or aspiration pneumonia. After consultation with pediatric pulmonology consultants, antimicrobials were held during the initial phase of work-up.
His headache may reflect a migraine, although further characterization and assessment for the presence and extent of head or neck trauma is warranted. Headache following trauma warrants consideration of cerebral contusion, diffuse axonal injury, intracranial hemorrhage, and carotid or vertebral artery dissection. Screening for concussion should also be performed. Hypoxemia may increase cerebral blood flow and raise intracranial pressure, resulting in headache.
CRP elevation is nonspecific and signals the presence of focal or systemic inflammation and is often elevated to a milder extent in obese patients with DMD. While normal procalcitonin argues against bacterial pneumonia, the precise level can be informative, and serial procalcitonin values may be more helpful than a single value. Although antecedent respiratory symptoms were not mentioned, viral or fungal pneumonia can present insidiously. An occult malignancy may be incidentally discovered when patients present for unrelated issues, although this and other sources of elevated CRP (eg, exacerbation of an autoimmune disease or drug reaction) remain less likely given the acuity of his presentation. Acute pulmonary embolism may be associated with a systemic inflammatory response and elevation in CRP.
In addition to the radiographic differential diagnosis already presented, the appearance of multifocal opacifications with hypoxemia raises the possibility of pulmonary infarcts or noncardiogenic pulmonary edema.
On hospital day 2, the patient continued to complain of “the worst headache of his life” as well as blurry vision and seeing “dark spots.” His headache did not improve with NSAIDs. A noncontrast CT scan of the head was normal. Neurology was consulted. Given his symptoms, history of migraines, stable neurological examination, and normal head CT, he was diagnosed with migraines and administered fluids, prochlorperazine, diphenhydramine, ondansetron, and NSAIDs. His headache continued and he continued to require supplemental oxygen.
The combination of hypoxemia, severe headache, and vision changes remains consistent with systemic emboli caused by thromboembolism or fat embolism. Headache assessment must also involve screening for “red flags,” which include sudden onset, antecedent head trauma, systemic illness (eg, fever or meningismus), focal neurologic findings, papilledema, changes with position or Valsalva, and immunosuppression. Although primary headache syndromes (eg, migraines or tension and cluster headache) may be triggered in the setting of trauma and systemic illness, “the worst headache of my life” is a concerning symptom that warrants urgent attention. While this invokes the possibility of a subarachnoid hemorrhage (SAH), headache severity is nonspecific, and rapid onset (ie, thunderclap headache) would be more suggestive. After 6 hours of symptoms, the sensitivity of head CT for detecting SAH declines, and lumbar puncture would be warranted to evaluate for xanthochromia.
His blurry vision and dark spots require testing of visual acuity and visual fields, as well as fundoscopic examination to assess for embolic phenomena or papilledema. Migraine is classically associated with “positive” or scintillating scotomata, although dark spots may occur. The presence of horizontal diplopia would indicate a cranial nerve VI palsy, which can occur with increased intracranial pressure. Visual-field cuts may also present as blurry vision, and monocular vs binocular deficits signal whether the issue is anterior or involving/posterior to the optic chiasm, respectively. Magnetic resonance imaging (MRI) may reveal the presence or sequelae of cerebral emboli (eg, fat emboli), including vasogenic edema.
Dilated fundus examination revealed Purtscher retinopathy: bilateral cotton-wool spots and larger areas of retinal whitening (Purtscher flecken).
Typical findings of Purtscher retinopathy include Purtscher flecken, cotton-wool spots, retinal hemorrhage, and optic disc edema. Purtscher retinopathy is classically associated with severe head trauma. Without associated head trauma, the term “Purtscher-like retinopathy” is used. Conditions that can cause Purtscher-like retinopathy include pancreatitis, vasculitis, microangiopathy, chronic renal failure, and systemic embolization. The most likely source of systemic embolization remains fat emboli stemming from his femur fracture. Treatment of FES is largely supportive.
The possibility of fat emboli had been repeatedly raised by the patient’s mother since admission. While providers had considered this a possibility, it was discounted early on because of the minor nature of the patient’s orthopedic trauma, the lack of clear radiographic evidence for pulmonary emboli on chest CT, and the normal head CT. The findings on the ophthalmologist’s fundoscopic examination led the primary team to reconsider FES, along with thromboemboli and pancreatitis. Lipase was normal. MRI of the brain with contrast revealed >20 microinfarcts in the bilateral hemispheres, left corpus callosum, and bilateral basal ganglia. The CT angiogram of the chest was rereviewed; the pediatric radiologists suggested that microinfarcts could explain the patchy small ground glass opacities seen in the lungs. A transthoracic echocardiogram and electrocardiogram were normal. The diagnosis of FES was made, and the patient was started on aspirin and enoxaparin prophylaxis. His headache and respiratory status improved, and he was discharged home with close follow-up.
DISCUSSION
FES is a rare complication associated with long bone fractures and orthopedic manipulation.1,2 The exact mechanism of fat emboli production is unknown, but two theories prevail. The mechanical theory states that an outside mechanical source causes bone marrow contents or adipose tissue contents to be dislodged into the circulation where they travel through the venous circulation to become embedded in the lungs.1,2 These fragments may also migrate to the arterial circulation, through a patent foramen ovale or intrapulmonary shunts, leading to end organ damage.1,2 The biochemical theory suggests that fat emboli in the venous circulation precipitate an inflammatory and prothrombotic cascade that triggers fibrin production, platelet aggregation, and release of free fatty acids into the circulation, predisposing patients to develop multifocal systemic emboli.1
Although the classic triad in FES includes respiratory symptoms, rash, and CNS symptoms, all three findings are only present in 1%-29% of cases.1,2 Respiratory abnormalities, ranging from tachypnea and dyspnea to ARDS and hypoxic respiratory failure, occur in up to 75% of patients with FES.1 Central nervous system (CNS) complications, including headache, confusion, coma, seizures, and death caused by cerebral ischemia, occur in up to 86% of patients.1,2 Petechiae may occur in 20%-60% of patients and are usually located on nondependent regions of the body such as the head, neck, and chest.
Diagnosis of FES is largely clinical and requires a high index of suspicion and elimination of other conditions, including pulmonary thromboembolism, diffuse intravascular coagulation, and sepsis. The CNS complications must be differentiated from CNS infection, stroke, migraine, benign intracranial hypertension, and intracranial hemorrhage. There is no gold standard test for diagnosis. The Gurd and Wilson criteria, modified Gurd criteria, and Schonfeld’s criteria (Table) are commonly used but have not been clinically validated.1,3-5 These use a combination of clinical signs of respiratory distress, neurological symptoms, petechial rash, and various other diagnostic factors. When patients have risk factors, such as trauma, surgery, or predisposing conditions (eg, mobility-limiting neuromuscular disorders) and signs and symptoms potentially consistent with FES, the diagnostic evaluation should include arterial blood gas analysis, complete blood count, chest radiographs, and coagulation studies. Ophthalmological exam to evaluate for cotton-wool spots, brain MRI to detect cerebral emboli, and CT pulmonary angiogram to assess for pulmonary infarcts may help to eliminate other diagnoses and/or confirm FES.
Diagnosis of uncommon conditions that present with nonspecific symptoms, like FES, can be challenging because the symptoms may overlap with many other possible diagnoses. This challenge is further exacerbated in patients with significant medical complexity, as with the patient discussed here. Specifically, this patient had multiple plausible explanations for CNS symptoms and respiratory symptoms. It was ultimately the visual symptoms that began to link his symptoms together into a unifying syndrome and the ophthalmologic examination that prompted confirmatory imaging. It is important to continually revisit and revise the differential diagnosis in patients with medical complexity and avoid the competing temptations to abandon the search for a unifying diagnosis and attribute all symptoms to a patient’s underlying condition.
Treatment of FES is largely supportive with close monitoring of neurological status and providing supplemental oxygen as needed. Corticosteroids have been suggested to help prevent FES in patients with long bone fractures, but there is no evidence to suggest they are helpful once FES is diagnosed.2 There is conflicting evidence for the efficacy of heparin or low-molecular-weight heparin as treatment in FES.2,6 After discussions with consulting physician teams, the patient, and his family, enoxaparin and aspirin were started for this patient in light of his tenuous condition in order to decrease the risk of further embolic complications.
Patients with DMD and other neuromuscular disorders likely have a greater propensity to develop FES even after minor trauma.1,6 This is believed to be caused by patients becoming nonambulatory early in life and receiving substantial corticosteroid therapy, which can lead to osteopenia and fatty replacement of the bone marrow.1,6 This population is also often obese by the second or third decade of life, which contributes to their already increased propensity to fall.1,6
To our knowledge, this patient is 1 of 18 reported cases of FES after trauma in DMD patients. Two-thirds of these cases occurred when an unrestrained patient fell from their wheelchair. The other cases occurred while walking, during physical therapy, and during assisted transfers.6-12 In these cases, FES had a guarded prognosis, with 7 of the 18 patients dying and 1 of the patients remaining in a persistent vegetative state.8,9 While caution is warranted in generalizing these findings, given the small number of reported cases and likely publication bias,education of caregivers and patients on use of restraints and safe transfers is paramount to limit the risk of trauma.
TEACHING POINTS
- FES is a rare condition that most commonly manifests with respiratory, neurological, and cutaneous findings.
- Patients with Duchenne’s Muscular Dystrophy are likely at increased risk for FES even with minor trauma; this makes wheelchair restraints and safe transfers fundamental.
- Patients with medical complexity and their caregivers are key members of the diagnostic team.
A 19-year-old man with Duchenne Muscular Dystrophy (DMD) presented to the Emergency Department (ED) for left knee pain after ejection from his motorized wheelchair at a low velocity. In the ED, he developed increasing respiratory distress.
When addressing a new problem in a patient with a chronic condition, it is crucial to first understand the chronic condition and then consider whether the presenting symptoms relate to that condition or stem from an unrelated inciting event.
Patients with DMD are at risk of pulmonary complications relating to their underlying disease. For instance, dysphagia and ineffective cough can predispose them to recurrent aspiration pneumonitis and/or pneumonia, whereas decreased lung compliance (from scoliosis, atelectasis, and/or pulmonary fibrosis) and respiratory muscle weakness can progress to ventilatory failure. In addition, patients with DMD are at risk for pulmonary thromboembolism in the setting of immobility. Patients with DMD may also develop congestive heart failure resulting from myocardial fibrosis and nonischemic cardiomyopathy.
The ejection from his wheelchair signals potential trauma-associated conditions that could explain his respiratory distress. Respiratory complications of blunt thoracic trauma include pulmonary contusion, pneumothorax, flail chest (resulting from fractured ribs), and acute respiratory distress syndrome (ARDS). Lower extremity injury can result in venous thrombosis and pulmonary thromboembolism. While classically associated with long bone fractures, fat embolism syndrome (FES) may rarely occur with rib fractures and soft-tissue trauma. Respiratory compromise may also result from cervical spinal cord injury or severe anemia from trauma-associated hemorrhage.
Additional past medical history included growth hormone deficiency, migraine headaches, osteoporosis secondary to chronic steroid use, cardiac fibrosis of the inferolateral wall and septum with a baseline left ventricular ejection fraction of 65%, and atrial fibrillation. His medications included calcium carbonate, vitamin D, omeprazole, lisinopril, metoprolol, prednisone, escitalopram, and testosterone. Physical examination revealed an ill-appearing obese man in respiratory distress. Temperature was 37.3°C, heart rate was 102 beats per minute (bpm), blood pressure was 110/74 mm Hg. His oxygen saturation was 93% with a respiratory rate of 25 breaths per minute while breathing ambient air. His lung sounds were clear, and his heart was without murmur. The left knee was diffusely tender to palpation without specific point tenderness. Strength was 2/5 with flexion and extension at the bilateral knees and hips and 3/5 flexion and extension at the bilateral elbows. He reported this level of weakness was his baseline. Radiographs revealed a minimally displaced Salter Harris II fracture (fracture line through the metaphysis and growth plate) of the left distal femur. His fracture was splinted early in his ED course. During his ED evaluation, the patient had acute worsening of tachycardia to 130 bpm, increased respiratory rate of 34 breaths per minute, and hypoxemia with an oxygen saturation of 83% on ambient air. He was placed on 3 L/min of oxygen via nasal cannula with improvement in his oxygen saturation to 90%. A chest radiograph was unremarkable, without evidence of pneumothorax, effusion, or pneumonia. The patient was admitted to the hospital.
The acute onset of tachypnea, tachycardia, and hypoxia, accompanied by a clear lung exam and normal chest radiograph, increases the likelihood of a pulmonary embolism. Obesity, testosterone therapy, and trauma increase his susceptibility to venous thromboembolism, while a distal femur fracture increases his risk for FES. Acute pulmonary aspiration often presents with initially absent or subtle radiographic findings. An arterial blood gas analysis would determine the presence and extent of an alveolar-arterial (A-a) gradient; a normal A-a gradient is seen in hypoventilation, while an elevated A-a gradient is seen in conditions affecting gas exchange, including pulmonary emboli and alveolar filling processes. His hypoxemia only partially corrects with supplemental oxygen, raising the possibility of capillary or anatomic shunting. Capillary shunting may occur with atelectasis, aspiration/pneumonia and pulmonary edema, whereas anatomic shunting can be intra-cardiac (eg, patent foramen ovale or septic defect) or intrapulmonary (eg, arteriovenous malformations). Patients with pulmonary emboli may also develop right-to-left shunting because of increased pulmonary vascular resistance, although hypoxemia with pulmonary emboli largely relates to ventilation/perfusion mismatch and decreased level of mixed venous blood oxygen (PvO2).
This patient’s complex medical history warrants a broadened differential with consideration of his cardiac history, including myocardial fibrosis and arrhythmia, and the impact of exposure to steroids on his immune and musculoskeletal systems. He has a history of atrial fibrillation, and an electrocardiogram is warranted to determine the underlying rhythm. Prolonged periods of rapid ventricular response may lead to tachycardia-induced cardiomyopathy. Myocardial fibrosis may progress despite use of angiotensin-converting enzyme inhibitors and is associated with systolic and/or diastolic dysfunction, although neither the examination findings provided nor the chest radiograph are suggestive of decompensated heart failure. Chronic exposure to corticosteroids (used in DMD to improve muscle strength and function) may predispose to numerous infectious and metabolic complications. Up to 10%-15% of patients with Pneumocystis jirovecii pneumonia may present with a normal chest radiograph. Acute adrenal insufficiency can present with tachycardia, weakness, and respiratory distress, so recent prednisone dose changes or interruptions should be assessed.
The patient’s respiratory status worsened. In light of his complex medical history, he was transferred to a children’s hospital for a higher level of care with a presumptive diagnosis of aspiration pneumonia. Upon reassessment at the new facility, the patient reported an ongoing and severe headache since his initial injury. NSAIDs had been given prior to transfer. His exam continued to be significant for tachycardia, tachypnea, and hypoxemia. His cardiac and lung examinations were otherwise normal. A comprehensive metabolic panel, procalcitonin, complete blood count with differential, and lactate were normal; his C-reactive protein (CRP) was 46.8 mg/dL (Normal <8 mg/dL). A computed tomography (CT) angiogram of the chest revealed small multifocal nodular ground-glass opacities, especially in the lower lobes, concerning for microatelectasis, multifocal pneumonia, or aspiration pneumonia. After consultation with pediatric pulmonology consultants, antimicrobials were held during the initial phase of work-up.
His headache may reflect a migraine, although further characterization and assessment for the presence and extent of head or neck trauma is warranted. Headache following trauma warrants consideration of cerebral contusion, diffuse axonal injury, intracranial hemorrhage, and carotid or vertebral artery dissection. Screening for concussion should also be performed. Hypoxemia may increase cerebral blood flow and raise intracranial pressure, resulting in headache.
CRP elevation is nonspecific and signals the presence of focal or systemic inflammation and is often elevated to a milder extent in obese patients with DMD. While normal procalcitonin argues against bacterial pneumonia, the precise level can be informative, and serial procalcitonin values may be more helpful than a single value. Although antecedent respiratory symptoms were not mentioned, viral or fungal pneumonia can present insidiously. An occult malignancy may be incidentally discovered when patients present for unrelated issues, although this and other sources of elevated CRP (eg, exacerbation of an autoimmune disease or drug reaction) remain less likely given the acuity of his presentation. Acute pulmonary embolism may be associated with a systemic inflammatory response and elevation in CRP.
In addition to the radiographic differential diagnosis already presented, the appearance of multifocal opacifications with hypoxemia raises the possibility of pulmonary infarcts or noncardiogenic pulmonary edema.
On hospital day 2, the patient continued to complain of “the worst headache of his life” as well as blurry vision and seeing “dark spots.” His headache did not improve with NSAIDs. A noncontrast CT scan of the head was normal. Neurology was consulted. Given his symptoms, history of migraines, stable neurological examination, and normal head CT, he was diagnosed with migraines and administered fluids, prochlorperazine, diphenhydramine, ondansetron, and NSAIDs. His headache continued and he continued to require supplemental oxygen.
The combination of hypoxemia, severe headache, and vision changes remains consistent with systemic emboli caused by thromboembolism or fat embolism. Headache assessment must also involve screening for “red flags,” which include sudden onset, antecedent head trauma, systemic illness (eg, fever or meningismus), focal neurologic findings, papilledema, changes with position or Valsalva, and immunosuppression. Although primary headache syndromes (eg, migraines or tension and cluster headache) may be triggered in the setting of trauma and systemic illness, “the worst headache of my life” is a concerning symptom that warrants urgent attention. While this invokes the possibility of a subarachnoid hemorrhage (SAH), headache severity is nonspecific, and rapid onset (ie, thunderclap headache) would be more suggestive. After 6 hours of symptoms, the sensitivity of head CT for detecting SAH declines, and lumbar puncture would be warranted to evaluate for xanthochromia.
His blurry vision and dark spots require testing of visual acuity and visual fields, as well as fundoscopic examination to assess for embolic phenomena or papilledema. Migraine is classically associated with “positive” or scintillating scotomata, although dark spots may occur. The presence of horizontal diplopia would indicate a cranial nerve VI palsy, which can occur with increased intracranial pressure. Visual-field cuts may also present as blurry vision, and monocular vs binocular deficits signal whether the issue is anterior or involving/posterior to the optic chiasm, respectively. Magnetic resonance imaging (MRI) may reveal the presence or sequelae of cerebral emboli (eg, fat emboli), including vasogenic edema.
Dilated fundus examination revealed Purtscher retinopathy: bilateral cotton-wool spots and larger areas of retinal whitening (Purtscher flecken).
Typical findings of Purtscher retinopathy include Purtscher flecken, cotton-wool spots, retinal hemorrhage, and optic disc edema. Purtscher retinopathy is classically associated with severe head trauma. Without associated head trauma, the term “Purtscher-like retinopathy” is used. Conditions that can cause Purtscher-like retinopathy include pancreatitis, vasculitis, microangiopathy, chronic renal failure, and systemic embolization. The most likely source of systemic embolization remains fat emboli stemming from his femur fracture. Treatment of FES is largely supportive.
The possibility of fat emboli had been repeatedly raised by the patient’s mother since admission. While providers had considered this a possibility, it was discounted early on because of the minor nature of the patient’s orthopedic trauma, the lack of clear radiographic evidence for pulmonary emboli on chest CT, and the normal head CT. The findings on the ophthalmologist’s fundoscopic examination led the primary team to reconsider FES, along with thromboemboli and pancreatitis. Lipase was normal. MRI of the brain with contrast revealed >20 microinfarcts in the bilateral hemispheres, left corpus callosum, and bilateral basal ganglia. The CT angiogram of the chest was rereviewed; the pediatric radiologists suggested that microinfarcts could explain the patchy small ground glass opacities seen in the lungs. A transthoracic echocardiogram and electrocardiogram were normal. The diagnosis of FES was made, and the patient was started on aspirin and enoxaparin prophylaxis. His headache and respiratory status improved, and he was discharged home with close follow-up.
DISCUSSION
FES is a rare complication associated with long bone fractures and orthopedic manipulation.1,2 The exact mechanism of fat emboli production is unknown, but two theories prevail. The mechanical theory states that an outside mechanical source causes bone marrow contents or adipose tissue contents to be dislodged into the circulation where they travel through the venous circulation to become embedded in the lungs.1,2 These fragments may also migrate to the arterial circulation, through a patent foramen ovale or intrapulmonary shunts, leading to end organ damage.1,2 The biochemical theory suggests that fat emboli in the venous circulation precipitate an inflammatory and prothrombotic cascade that triggers fibrin production, platelet aggregation, and release of free fatty acids into the circulation, predisposing patients to develop multifocal systemic emboli.1
Although the classic triad in FES includes respiratory symptoms, rash, and CNS symptoms, all three findings are only present in 1%-29% of cases.1,2 Respiratory abnormalities, ranging from tachypnea and dyspnea to ARDS and hypoxic respiratory failure, occur in up to 75% of patients with FES.1 Central nervous system (CNS) complications, including headache, confusion, coma, seizures, and death caused by cerebral ischemia, occur in up to 86% of patients.1,2 Petechiae may occur in 20%-60% of patients and are usually located on nondependent regions of the body such as the head, neck, and chest.
Diagnosis of FES is largely clinical and requires a high index of suspicion and elimination of other conditions, including pulmonary thromboembolism, diffuse intravascular coagulation, and sepsis. The CNS complications must be differentiated from CNS infection, stroke, migraine, benign intracranial hypertension, and intracranial hemorrhage. There is no gold standard test for diagnosis. The Gurd and Wilson criteria, modified Gurd criteria, and Schonfeld’s criteria (Table) are commonly used but have not been clinically validated.1,3-5 These use a combination of clinical signs of respiratory distress, neurological symptoms, petechial rash, and various other diagnostic factors. When patients have risk factors, such as trauma, surgery, or predisposing conditions (eg, mobility-limiting neuromuscular disorders) and signs and symptoms potentially consistent with FES, the diagnostic evaluation should include arterial blood gas analysis, complete blood count, chest radiographs, and coagulation studies. Ophthalmological exam to evaluate for cotton-wool spots, brain MRI to detect cerebral emboli, and CT pulmonary angiogram to assess for pulmonary infarcts may help to eliminate other diagnoses and/or confirm FES.
Diagnosis of uncommon conditions that present with nonspecific symptoms, like FES, can be challenging because the symptoms may overlap with many other possible diagnoses. This challenge is further exacerbated in patients with significant medical complexity, as with the patient discussed here. Specifically, this patient had multiple plausible explanations for CNS symptoms and respiratory symptoms. It was ultimately the visual symptoms that began to link his symptoms together into a unifying syndrome and the ophthalmologic examination that prompted confirmatory imaging. It is important to continually revisit and revise the differential diagnosis in patients with medical complexity and avoid the competing temptations to abandon the search for a unifying diagnosis and attribute all symptoms to a patient’s underlying condition.
Treatment of FES is largely supportive with close monitoring of neurological status and providing supplemental oxygen as needed. Corticosteroids have been suggested to help prevent FES in patients with long bone fractures, but there is no evidence to suggest they are helpful once FES is diagnosed.2 There is conflicting evidence for the efficacy of heparin or low-molecular-weight heparin as treatment in FES.2,6 After discussions with consulting physician teams, the patient, and his family, enoxaparin and aspirin were started for this patient in light of his tenuous condition in order to decrease the risk of further embolic complications.
Patients with DMD and other neuromuscular disorders likely have a greater propensity to develop FES even after minor trauma.1,6 This is believed to be caused by patients becoming nonambulatory early in life and receiving substantial corticosteroid therapy, which can lead to osteopenia and fatty replacement of the bone marrow.1,6 This population is also often obese by the second or third decade of life, which contributes to their already increased propensity to fall.1,6
To our knowledge, this patient is 1 of 18 reported cases of FES after trauma in DMD patients. Two-thirds of these cases occurred when an unrestrained patient fell from their wheelchair. The other cases occurred while walking, during physical therapy, and during assisted transfers.6-12 In these cases, FES had a guarded prognosis, with 7 of the 18 patients dying and 1 of the patients remaining in a persistent vegetative state.8,9 While caution is warranted in generalizing these findings, given the small number of reported cases and likely publication bias,education of caregivers and patients on use of restraints and safe transfers is paramount to limit the risk of trauma.
TEACHING POINTS
- FES is a rare condition that most commonly manifests with respiratory, neurological, and cutaneous findings.
- Patients with Duchenne’s Muscular Dystrophy are likely at increased risk for FES even with minor trauma; this makes wheelchair restraints and safe transfers fundamental.
- Patients with medical complexity and their caregivers are key members of the diagnostic team.
1. Fukumoto LE, Fukumoto KD. Fat embolism syndrome. Nurs Clin North Am. 2018;53(3):335-347. https://doi.org/10.1016/j.cnur.2018.04.003.
2. Scarpino M, Lanzo G, Lolli F, Grippo A. From the diagnosis to the therapeutic management: Cerebral fat embolism, a clinical challenge. Int J Gen Med. 2019;2019(12):39-48. https://doi.org/10.2147/IJGM.S177407.
1. Fukumoto LE, Fukumoto KD. Fat embolism syndrome. Nurs Clin North Am. 2018;53(3):335-347. https://doi.org/10.1016/j.cnur.2018.04.003.
2. Scarpino M, Lanzo G, Lolli F, Grippo A. From the diagnosis to the therapeutic management: Cerebral fat embolism, a clinical challenge. Int J Gen Med. 2019;2019(12):39-48. https://doi.org/10.2147/IJGM.S177407.
© 2020 Society of Hospital Medicine
Is Transfer Always the Best Choice?
Some veterans who present to smaller facilities, such as rural hospitals, are transferred to larger facilities for diagnostic or therapeutic procedures. But that access also can mean hardship for rural veterans by taking them far from family and adding costs. Moreover, complex care coordination can cause “triage mismatch” when the patients are at their most vulnerable: “over-triage”—transferring patients unlikely to benefit and “under-triage”—failing to transfer those likely to benefit.
Researchers from VA Iowa City Healthcare System and University of Iowa conducted a study to find out what proportion of VHA transfers were potentially avoidable. Their study included all veterans treated in any of 120 VHA emergency departments (EDs) and transferred to a VHA acute care hospital between January 2012 and December 2014.
Potentially avoidable transfers (PATs) were defined as transfers in which the patient was either discharged from the referral ED or admitted to the referral hospital for < 24 hours, without having an invasive procedure. The researchers chose that definition to identify patients whose transfer might have been avoided if real-time specialty telemedicine were available at the index hospital. (They caution that the definition was not intended to suggest that all PATs were inappropriate.)
Over 3 years, 18,852 patients were transferred. Of the total patients transferred, 36% were transferred from 1 VHA ED to another VHA facility. Of the VHA transfers, 8,639 (46%) were transferred to another VHA ED; the rest were transferred to another VHA facility inpatient unit. The median transfer distance was 81.5 miles. Rural residents were transferred 3 times as often as urban residents.
The good news is that PATs are rare. Only 0.8% of VHA ED visits resulted in transfer, and of those, only one-fourth were deemed potentially avoidable. And while rural veterans were more likely to be transferred, PATs were less prevalent among those transfers (20.8% vs 23.9% for urban veterans).
More than half of VHA transfers were for patients diagnosed with mental health, cardiac, and digestive conditions. The top ICD-9 diagnosis related to VHA ED transfer was suicidal ideation. The diagnostic procedures associated with most PATs were mental health (11% potentially avoidable) and cardiac (21% potentially avoidable).
Their research turned up some unexpected data: For example, smaller EDs did not have a higher prevalence of PATs, suggesting that ED size was not associated with transfer appropriateness. And the proportion of PATs was higher in hospitals with > 50% board-certified emergency physicians.
The researchers say their findings highlight important differences between the VHA health care and civilian health care systems, emphasizing that the resources available within the VHA health system “might be unique” and underlining the need for VHA-specific solutions to health care delivery challenges.
The overall purpose of this study, the researchers say, was to identify areas where novel delivery of specialty care might reduce the need for some VHA transfers. Their analysis provides data for developing targeted intervention, such as ED-based telemedicine or “targeted remote care.”
Patients with mental health conditions—who made up more than one-third of all VHA-to-VHA interfacility transfers, higher than that reported in civilian hospitals—represent a “rich target population” for telehealth, the researchers suggest. They also note that because mental health providers are in critical shortage in most of the US, real-time telemedicine providing psychiatric resources could be an important and timely service.
Nearly half of medical directors of VHA EDs who responded to the VHA Healthcare Analysis and Information Group survey cited the transfer process as “overly burdensome,” and > 65% said administrative processes contribute to delay in transfer. Finding new ways to keep patients local could benefit providers as well.
Some veterans who present to smaller facilities, such as rural hospitals, are transferred to larger facilities for diagnostic or therapeutic procedures. But that access also can mean hardship for rural veterans by taking them far from family and adding costs. Moreover, complex care coordination can cause “triage mismatch” when the patients are at their most vulnerable: “over-triage”—transferring patients unlikely to benefit and “under-triage”—failing to transfer those likely to benefit.
Researchers from VA Iowa City Healthcare System and University of Iowa conducted a study to find out what proportion of VHA transfers were potentially avoidable. Their study included all veterans treated in any of 120 VHA emergency departments (EDs) and transferred to a VHA acute care hospital between January 2012 and December 2014.
Potentially avoidable transfers (PATs) were defined as transfers in which the patient was either discharged from the referral ED or admitted to the referral hospital for < 24 hours, without having an invasive procedure. The researchers chose that definition to identify patients whose transfer might have been avoided if real-time specialty telemedicine were available at the index hospital. (They caution that the definition was not intended to suggest that all PATs were inappropriate.)
Over 3 years, 18,852 patients were transferred. Of the total patients transferred, 36% were transferred from 1 VHA ED to another VHA facility. Of the VHA transfers, 8,639 (46%) were transferred to another VHA ED; the rest were transferred to another VHA facility inpatient unit. The median transfer distance was 81.5 miles. Rural residents were transferred 3 times as often as urban residents.
The good news is that PATs are rare. Only 0.8% of VHA ED visits resulted in transfer, and of those, only one-fourth were deemed potentially avoidable. And while rural veterans were more likely to be transferred, PATs were less prevalent among those transfers (20.8% vs 23.9% for urban veterans).
More than half of VHA transfers were for patients diagnosed with mental health, cardiac, and digestive conditions. The top ICD-9 diagnosis related to VHA ED transfer was suicidal ideation. The diagnostic procedures associated with most PATs were mental health (11% potentially avoidable) and cardiac (21% potentially avoidable).
Their research turned up some unexpected data: For example, smaller EDs did not have a higher prevalence of PATs, suggesting that ED size was not associated with transfer appropriateness. And the proportion of PATs was higher in hospitals with > 50% board-certified emergency physicians.
The researchers say their findings highlight important differences between the VHA health care and civilian health care systems, emphasizing that the resources available within the VHA health system “might be unique” and underlining the need for VHA-specific solutions to health care delivery challenges.
The overall purpose of this study, the researchers say, was to identify areas where novel delivery of specialty care might reduce the need for some VHA transfers. Their analysis provides data for developing targeted intervention, such as ED-based telemedicine or “targeted remote care.”
Patients with mental health conditions—who made up more than one-third of all VHA-to-VHA interfacility transfers, higher than that reported in civilian hospitals—represent a “rich target population” for telehealth, the researchers suggest. They also note that because mental health providers are in critical shortage in most of the US, real-time telemedicine providing psychiatric resources could be an important and timely service.
Nearly half of medical directors of VHA EDs who responded to the VHA Healthcare Analysis and Information Group survey cited the transfer process as “overly burdensome,” and > 65% said administrative processes contribute to delay in transfer. Finding new ways to keep patients local could benefit providers as well.
Some veterans who present to smaller facilities, such as rural hospitals, are transferred to larger facilities for diagnostic or therapeutic procedures. But that access also can mean hardship for rural veterans by taking them far from family and adding costs. Moreover, complex care coordination can cause “triage mismatch” when the patients are at their most vulnerable: “over-triage”—transferring patients unlikely to benefit and “under-triage”—failing to transfer those likely to benefit.
Researchers from VA Iowa City Healthcare System and University of Iowa conducted a study to find out what proportion of VHA transfers were potentially avoidable. Their study included all veterans treated in any of 120 VHA emergency departments (EDs) and transferred to a VHA acute care hospital between January 2012 and December 2014.
Potentially avoidable transfers (PATs) were defined as transfers in which the patient was either discharged from the referral ED or admitted to the referral hospital for < 24 hours, without having an invasive procedure. The researchers chose that definition to identify patients whose transfer might have been avoided if real-time specialty telemedicine were available at the index hospital. (They caution that the definition was not intended to suggest that all PATs were inappropriate.)
Over 3 years, 18,852 patients were transferred. Of the total patients transferred, 36% were transferred from 1 VHA ED to another VHA facility. Of the VHA transfers, 8,639 (46%) were transferred to another VHA ED; the rest were transferred to another VHA facility inpatient unit. The median transfer distance was 81.5 miles. Rural residents were transferred 3 times as often as urban residents.
The good news is that PATs are rare. Only 0.8% of VHA ED visits resulted in transfer, and of those, only one-fourth were deemed potentially avoidable. And while rural veterans were more likely to be transferred, PATs were less prevalent among those transfers (20.8% vs 23.9% for urban veterans).
More than half of VHA transfers were for patients diagnosed with mental health, cardiac, and digestive conditions. The top ICD-9 diagnosis related to VHA ED transfer was suicidal ideation. The diagnostic procedures associated with most PATs were mental health (11% potentially avoidable) and cardiac (21% potentially avoidable).
Their research turned up some unexpected data: For example, smaller EDs did not have a higher prevalence of PATs, suggesting that ED size was not associated with transfer appropriateness. And the proportion of PATs was higher in hospitals with > 50% board-certified emergency physicians.
The researchers say their findings highlight important differences between the VHA health care and civilian health care systems, emphasizing that the resources available within the VHA health system “might be unique” and underlining the need for VHA-specific solutions to health care delivery challenges.
The overall purpose of this study, the researchers say, was to identify areas where novel delivery of specialty care might reduce the need for some VHA transfers. Their analysis provides data for developing targeted intervention, such as ED-based telemedicine or “targeted remote care.”
Patients with mental health conditions—who made up more than one-third of all VHA-to-VHA interfacility transfers, higher than that reported in civilian hospitals—represent a “rich target population” for telehealth, the researchers suggest. They also note that because mental health providers are in critical shortage in most of the US, real-time telemedicine providing psychiatric resources could be an important and timely service.
Nearly half of medical directors of VHA EDs who responded to the VHA Healthcare Analysis and Information Group survey cited the transfer process as “overly burdensome,” and > 65% said administrative processes contribute to delay in transfer. Finding new ways to keep patients local could benefit providers as well.
Leadership & Professional Development: Cultivating Habits for the Hospitalist
“We are what we repeatedly do. Excellence, then, is not an act, but a habit.”
—Will Durant
We are a collection of our habits—the routine, repetitive, subconscious behaviors we perform on a daily basis. Some of these behaviors are positive, others less so. Habits allow us to perform tasks automatically, without the need for active decision making. Amidst a constantly changing clinical environment, cultivating consistent habits can improve our adherence to best practices and free cognitive effort toward more challenging diagnostic or therapeutic tasks.
Establishing habits requires practice and intentionality. First, identify those habits that are desirable in your personal and professional life. Next, find a method to develop the habit. Then, hold yourself accountable as you work to embed the habit. Simple? Not quite.
In “The Power of Habit,” author Charles Duhigg introduces habit loops as a way to successfully develop this practice.1 Habit loops—sequences comprising a cue, routine, and reward—are integral to developing routines that support professional and personal aspects of hospitalist life. Consider a hospitalist seeking to develop a prerounds routine to increase efficiency and limit missed patient information. First, the clinician should identify a cue to start the routine, such as sitting down to log in at a specific workstation. Second, a sequence of actions is “chunked” into a consistent order, such as a review of vital signs, clinical notes, and patient labs. After the routine is completed, the clinician should finish with a reward, such as a cup of coffee after rounds. Want to set up a habit for ensuring learning goals are set with trainees at the beginning of every block? Set a calendar reminder for this on the first day, standardize how you communicate goals, and reward yourself with a team lunch at the end of the rotation. What if it’s a busy first day on service? Doesn’t matter. As Clay Christensen notes in “How Will You Measure Your Life?,” making one commitment to a habit is easier than deciding whether or not to engage in the routine every time new circumstances arise.2 The intentionality that comes with this act ensures consistency in the practice.
As a busy hospitalist, establishing habits for personal and professional development requires cues and rewards. For example, do you want to cement a habit of reading the latest journal articles or carving out time each day to reflect on your work? Then cultivate the routine by creating a cue, such as a dashboard on a wall to visualize how many articles you’ve read this week or whether you’ve paused to reflect on your rotation. Reinforce the routine by creating a reward: a walk outside, time with family, or another activity you enjoy. Pair the same reward with the same routine to strengthen the habit loop.
A few additional tips for cultivating habits: it is useful to pair an existing reliable habit, or “anchor habit,” with a new one, such as a short meditation after brushing your teeth.3 Doing so reinforces behaviors in a positive way. You may use the same principles to lose unwanted habits (eg, checking e-mail excessively) by removing cues, such as turning off notifications or using airplane mode and rewarding yourself when you see the behavior through.
Habits are larger than behaviors; they can impact your personal and professional life in important ways. By actively creating habits that align with your long-term priorities, you can create a safety net if and when change arrives. Understanding the psychology of habits and employing cues and rewards effectively can lead hospitalists to create positive routines that improve their clinical practice and personal lives.
1. Duhigg C. The Power of Habit: Why We Do What We Do in Life and Business. Random House; 2012.
2. Christensen CM. How Will You Measure Your Life? (Harvard Business Review Classics). Harvard Business Review Press; 2017.
3. Fogg B. Tiny Habits w/Dr. BJ Fogg-Behavior Change: Tiny Habits; 2011.
“We are what we repeatedly do. Excellence, then, is not an act, but a habit.”
—Will Durant
We are a collection of our habits—the routine, repetitive, subconscious behaviors we perform on a daily basis. Some of these behaviors are positive, others less so. Habits allow us to perform tasks automatically, without the need for active decision making. Amidst a constantly changing clinical environment, cultivating consistent habits can improve our adherence to best practices and free cognitive effort toward more challenging diagnostic or therapeutic tasks.
Establishing habits requires practice and intentionality. First, identify those habits that are desirable in your personal and professional life. Next, find a method to develop the habit. Then, hold yourself accountable as you work to embed the habit. Simple? Not quite.
In “The Power of Habit,” author Charles Duhigg introduces habit loops as a way to successfully develop this practice.1 Habit loops—sequences comprising a cue, routine, and reward—are integral to developing routines that support professional and personal aspects of hospitalist life. Consider a hospitalist seeking to develop a prerounds routine to increase efficiency and limit missed patient information. First, the clinician should identify a cue to start the routine, such as sitting down to log in at a specific workstation. Second, a sequence of actions is “chunked” into a consistent order, such as a review of vital signs, clinical notes, and patient labs. After the routine is completed, the clinician should finish with a reward, such as a cup of coffee after rounds. Want to set up a habit for ensuring learning goals are set with trainees at the beginning of every block? Set a calendar reminder for this on the first day, standardize how you communicate goals, and reward yourself with a team lunch at the end of the rotation. What if it’s a busy first day on service? Doesn’t matter. As Clay Christensen notes in “How Will You Measure Your Life?,” making one commitment to a habit is easier than deciding whether or not to engage in the routine every time new circumstances arise.2 The intentionality that comes with this act ensures consistency in the practice.
As a busy hospitalist, establishing habits for personal and professional development requires cues and rewards. For example, do you want to cement a habit of reading the latest journal articles or carving out time each day to reflect on your work? Then cultivate the routine by creating a cue, such as a dashboard on a wall to visualize how many articles you’ve read this week or whether you’ve paused to reflect on your rotation. Reinforce the routine by creating a reward: a walk outside, time with family, or another activity you enjoy. Pair the same reward with the same routine to strengthen the habit loop.
A few additional tips for cultivating habits: it is useful to pair an existing reliable habit, or “anchor habit,” with a new one, such as a short meditation after brushing your teeth.3 Doing so reinforces behaviors in a positive way. You may use the same principles to lose unwanted habits (eg, checking e-mail excessively) by removing cues, such as turning off notifications or using airplane mode and rewarding yourself when you see the behavior through.
Habits are larger than behaviors; they can impact your personal and professional life in important ways. By actively creating habits that align with your long-term priorities, you can create a safety net if and when change arrives. Understanding the psychology of habits and employing cues and rewards effectively can lead hospitalists to create positive routines that improve their clinical practice and personal lives.
“We are what we repeatedly do. Excellence, then, is not an act, but a habit.”
—Will Durant
We are a collection of our habits—the routine, repetitive, subconscious behaviors we perform on a daily basis. Some of these behaviors are positive, others less so. Habits allow us to perform tasks automatically, without the need for active decision making. Amidst a constantly changing clinical environment, cultivating consistent habits can improve our adherence to best practices and free cognitive effort toward more challenging diagnostic or therapeutic tasks.
Establishing habits requires practice and intentionality. First, identify those habits that are desirable in your personal and professional life. Next, find a method to develop the habit. Then, hold yourself accountable as you work to embed the habit. Simple? Not quite.
In “The Power of Habit,” author Charles Duhigg introduces habit loops as a way to successfully develop this practice.1 Habit loops—sequences comprising a cue, routine, and reward—are integral to developing routines that support professional and personal aspects of hospitalist life. Consider a hospitalist seeking to develop a prerounds routine to increase efficiency and limit missed patient information. First, the clinician should identify a cue to start the routine, such as sitting down to log in at a specific workstation. Second, a sequence of actions is “chunked” into a consistent order, such as a review of vital signs, clinical notes, and patient labs. After the routine is completed, the clinician should finish with a reward, such as a cup of coffee after rounds. Want to set up a habit for ensuring learning goals are set with trainees at the beginning of every block? Set a calendar reminder for this on the first day, standardize how you communicate goals, and reward yourself with a team lunch at the end of the rotation. What if it’s a busy first day on service? Doesn’t matter. As Clay Christensen notes in “How Will You Measure Your Life?,” making one commitment to a habit is easier than deciding whether or not to engage in the routine every time new circumstances arise.2 The intentionality that comes with this act ensures consistency in the practice.
As a busy hospitalist, establishing habits for personal and professional development requires cues and rewards. For example, do you want to cement a habit of reading the latest journal articles or carving out time each day to reflect on your work? Then cultivate the routine by creating a cue, such as a dashboard on a wall to visualize how many articles you’ve read this week or whether you’ve paused to reflect on your rotation. Reinforce the routine by creating a reward: a walk outside, time with family, or another activity you enjoy. Pair the same reward with the same routine to strengthen the habit loop.
A few additional tips for cultivating habits: it is useful to pair an existing reliable habit, or “anchor habit,” with a new one, such as a short meditation after brushing your teeth.3 Doing so reinforces behaviors in a positive way. You may use the same principles to lose unwanted habits (eg, checking e-mail excessively) by removing cues, such as turning off notifications or using airplane mode and rewarding yourself when you see the behavior through.
Habits are larger than behaviors; they can impact your personal and professional life in important ways. By actively creating habits that align with your long-term priorities, you can create a safety net if and when change arrives. Understanding the psychology of habits and employing cues and rewards effectively can lead hospitalists to create positive routines that improve their clinical practice and personal lives.
1. Duhigg C. The Power of Habit: Why We Do What We Do in Life and Business. Random House; 2012.
2. Christensen CM. How Will You Measure Your Life? (Harvard Business Review Classics). Harvard Business Review Press; 2017.
3. Fogg B. Tiny Habits w/Dr. BJ Fogg-Behavior Change: Tiny Habits; 2011.
1. Duhigg C. The Power of Habit: Why We Do What We Do in Life and Business. Random House; 2012.
2. Christensen CM. How Will You Measure Your Life? (Harvard Business Review Classics). Harvard Business Review Press; 2017.
3. Fogg B. Tiny Habits w/Dr. BJ Fogg-Behavior Change: Tiny Habits; 2011.
© 2020 Society of Hospital Medicine
Fulfilling the Potential of Point-of-Care Ultrasound in Hospital Medicine
The enthusiasm surrounding point-of-care ultrasound (POCUS) is clear and well founded. POCUS is a powerful tool that produces valuable diagnostic information for common and important clinical problems faced by hospitalists, such as pneumonia, soft-tissue infections,1 and myriad other applications. It can inform the evaluation and management of complex clinical problems such as dyspnea.2 Beyond its diagnostic potential, POCUS is well known to improve common procedures performed by adult and pediatric hospitalists by improving success rates and decreasing complications.
Excitement surrounding this technology continues to grow among hospitalists, leading to a proliferation of high-quality educational programs over the last 5 years. Most notable among these offerings has been the more comprehensive training available through the Society of Hospital Medicine (SHM) certificate-based pathway, though many other strong options exist, including institution-based curricula, such as the HealthPartners CHAMP program,3 and pediatric-focused programs. Growth in training is also occurring among medical students and residents. As of a 2012 survey, the majority (51%) of US medical schools had begun to weave ultrasound into their curricula,4 and this growth is also occurring in internal medicine and pediatric residency programs.5
Given the high potential for this technology and the growth in interest, it is an excellent time to pause and review some of the challenges faced by practitioners, hospitalist groups, and educators seeking to optimize POCUS implementation. A deliberate approach to POCUS education, the development of shared standards for high-quality use, and an ongoing dedication to develop specialty-specific practices will largely determine how much of this potential is fulfilled.
The largest challenge is likely to be educational. Educating clinicians to be able to integrate POCUS into practice is a complex, multistep process requiring not only an adequate core of didactic training and access to machines, but also the structured opportunity to develop rudimentary hands-on skills. Such initial training should be followed by continued practice and feedback as developing POCUS users progress toward independent practice. The study by Kumar et al.6 reaffirms that brief didactic lectures and access to machines are necessary, but they are clearly insufficient for learners to be able to use POCUS independently for a wide variety of applications. Their intervention also contrasts markedly with the 20 hours of didactics and 150 supervised scans recommended by the American College of Emergency Physicians prior to independent use for a core of six applications.7
Shared standards for education, use, and oversight will be crucial to fulfilling the potential of POCUS within hospital medicine. Our belief is that much can be learned from the thoughtful approach taken during the development of POCUS as a mainstream tool in emergency medicine in the early 2000s. In this approach, emergency physicians determined a sufficient and achievable standard of training for core POCUS applications, which was widely adopted. Based on completion of this training, physicians who were required to complete credentialing from their hospitals were widely able to achieve it, without any need for external certification. Emergency medicine guidelines further mandated the documentation of examinations and the creation of an exam report, features that improve clinical communication and facilitate quality improvement. Quality assurance processes that reviewed images and clinician interpretations were established as mandatory, which they should be in hospital medicine. Evidence was produced as to which exams physicians could do reliably with this focused training and which they could not. In the context of these thoughtful constructs, lawsuits have been noted to be exceedingly rare; and when they do occur, they have typically been for the failure to use POCUS rather than the converse.8
While many of these precepts deserve replication, others should also be modified to reflect changes in technology, medical education, and medical practice over the last 20 years and to improve upon this base of success. For example, with POCUS training now appearing in many medical school and residency curricula, training paradigms for both residents and attendings will need to accommodate a wider range of incoming skills. Emphasis should continue to be shifted toward competency-based assessments and entrustment and away from a fixed training time or exam number threshold. Important financial aspects have also changed. The cost of practical machines has dropped considerably, and medicine is shifting away from a fee-for-service model. While it remains appropriate that physicians may bill for POCUS examinations, it is likely that improved diagnosis, improved throughput, and a reduction in complications will yield greater value and should be the emphasis of cost/value discussions.9 Finally, while hospitals may impose credentialing, this process can also create a burden not present for most other noninvasive skills and may deter appropriate use. If this approach is chosen by a hospital, requirements should ideally remain modest, and as these skills become more widespread, POCUS should ultimately be built into board examinations and core credentialing.9
Thoughtful and concerted effort will be required by hospitalist leaders, educational innovators, and professional societies in developing POCUS to best serve hospitalists and their patients. This work has already begun. For example, in 2019 SHM offered a position statement outlining important aspects such as current evidence-based applications, training pathways, quality assurance, and program management.10 These recommendations should guide both adult and pediatric hospitalists. The Alliance for Academic Internal Medicine offered a similar position statement for resident training.11 Interest groups are growing in numerous professional societies, which will facilitate collaboration and promote propagation of best practices. High-quality educational tools are continuing to be developed by numerous organizations.
While further development is needed to add the detail, granularity, and practical tools that educational and practice leaders need to assure that POCUS achieves its potential in hospital medicine, the foundation for POCUS use within the specialty is being thoughtfully constructed. As this process proceeds, it will be vital to continue to learn from our emergency medicine colleagues, who have already met similar challenges, while at the same time be able to develop a modern POCUS model optimized for hospital medicine workflow, training, and patient care.
1. Kinnear B, Kelleher M, Chorny V. Clinical practice update: Point-of-care ultrasound for the pediatric hospitalist. J Hosp Med. 2019;15(3):170-172. https://doi.org/10.12788/jhm.3325.
2. Kelleher M, Kinnear B, Olson A. Clinical progress note: Point-of-care ultrasound in the evaluation of the dyspneic adult. J Hosp Med. 2020;15(3):173-175. https://doi.org/10.12788/jhm.3340.
3. Mathews BK, Reierson K, Vuong K, et al. The design and evaluation of the Comprehensive Hospitalist Assessment and Mentorship with Portfolios (CHAMP) Ultrasound Program. J Hosp Med. 2018;13(8):544-550. https://doi.org/10.12788/jhm.2938.
4. Bahner DP, Goldman E, Way D, Royall NA, Liu YT. The state of ultrasound education in U.S. medical schools: Results of a national survey. Acad Med. 2014;89(12):1681-1686. https://doi.org/10.1097/ACM.0000000000000414.
5. Reaume M, Siuba M, Wagner M, Woodwyk A, Melgar TA. Prevalence and Scope of point-of-care ultrasound education in internal medicine, pediatric, and medicine-pediatric residency programs in the United States. J Ultrasound Med. 2019;38(6):1433-1439. https://doi.org/10.1002/jum.14821.
6. Kumar A, Weng Y, Wang L, et al. Portable ultrasound device usage and learning outcomes among internal medicine trainees: a parallel-group randomized trial. J Hosp Med. 2020;15(3):154-159. https://doi.org/10.12788/jhm.3351.
7. Ultrasound Guidelines: Emergency, Point-of-Care and Clinical Ultrasound Guidelines in Medicine. Ann Emerg Med. 2017;69(5):e27-e54. https://doi.org/10.1016/j.annemergmed.2016.08.457.
8. Stolz L, O’Brien KM, Miller ML, Winters-Brown ND, Blaivas M, Adhikari S. A review of lawsuits related to point-of-care emergency ultrasound applications. West J Emerg Med. 2015;16(1):1-4. https://doi.org/10.5811/westjem.2014.11.23592.
9, Soni NJ, Tierney DM, Jensen TP, Lucas BP. Certification of Point-of-Care Ultrasound Competency. J Hosp Med. 2017;12(9):775-776. doi:10.12788/jhm.2812
10. Soni NJ, Schnobrich D, Matthews BK, et al. Point-of-Care Ultrasound for hospitalists: A position statement of the society of hospital medicine. J Hosp Med. 2019;14. https://doi.org/10.12788/jhm.3079.
11. LoPresti CM, Jensen TP, Dversdal RK, Astiz DJ. Point-of-Care Ultrasound for Internal Medicine Residency Training: A position statement from the Alliance of Academic Internal Medicine. Am J Med. 2019 Nov;132(11):1356-1360. https://doi.org/10.1016/j.amjmed.2019.07.019.
The enthusiasm surrounding point-of-care ultrasound (POCUS) is clear and well founded. POCUS is a powerful tool that produces valuable diagnostic information for common and important clinical problems faced by hospitalists, such as pneumonia, soft-tissue infections,1 and myriad other applications. It can inform the evaluation and management of complex clinical problems such as dyspnea.2 Beyond its diagnostic potential, POCUS is well known to improve common procedures performed by adult and pediatric hospitalists by improving success rates and decreasing complications.
Excitement surrounding this technology continues to grow among hospitalists, leading to a proliferation of high-quality educational programs over the last 5 years. Most notable among these offerings has been the more comprehensive training available through the Society of Hospital Medicine (SHM) certificate-based pathway, though many other strong options exist, including institution-based curricula, such as the HealthPartners CHAMP program,3 and pediatric-focused programs. Growth in training is also occurring among medical students and residents. As of a 2012 survey, the majority (51%) of US medical schools had begun to weave ultrasound into their curricula,4 and this growth is also occurring in internal medicine and pediatric residency programs.5
Given the high potential for this technology and the growth in interest, it is an excellent time to pause and review some of the challenges faced by practitioners, hospitalist groups, and educators seeking to optimize POCUS implementation. A deliberate approach to POCUS education, the development of shared standards for high-quality use, and an ongoing dedication to develop specialty-specific practices will largely determine how much of this potential is fulfilled.
The largest challenge is likely to be educational. Educating clinicians to be able to integrate POCUS into practice is a complex, multistep process requiring not only an adequate core of didactic training and access to machines, but also the structured opportunity to develop rudimentary hands-on skills. Such initial training should be followed by continued practice and feedback as developing POCUS users progress toward independent practice. The study by Kumar et al.6 reaffirms that brief didactic lectures and access to machines are necessary, but they are clearly insufficient for learners to be able to use POCUS independently for a wide variety of applications. Their intervention also contrasts markedly with the 20 hours of didactics and 150 supervised scans recommended by the American College of Emergency Physicians prior to independent use for a core of six applications.7
Shared standards for education, use, and oversight will be crucial to fulfilling the potential of POCUS within hospital medicine. Our belief is that much can be learned from the thoughtful approach taken during the development of POCUS as a mainstream tool in emergency medicine in the early 2000s. In this approach, emergency physicians determined a sufficient and achievable standard of training for core POCUS applications, which was widely adopted. Based on completion of this training, physicians who were required to complete credentialing from their hospitals were widely able to achieve it, without any need for external certification. Emergency medicine guidelines further mandated the documentation of examinations and the creation of an exam report, features that improve clinical communication and facilitate quality improvement. Quality assurance processes that reviewed images and clinician interpretations were established as mandatory, which they should be in hospital medicine. Evidence was produced as to which exams physicians could do reliably with this focused training and which they could not. In the context of these thoughtful constructs, lawsuits have been noted to be exceedingly rare; and when they do occur, they have typically been for the failure to use POCUS rather than the converse.8
While many of these precepts deserve replication, others should also be modified to reflect changes in technology, medical education, and medical practice over the last 20 years and to improve upon this base of success. For example, with POCUS training now appearing in many medical school and residency curricula, training paradigms for both residents and attendings will need to accommodate a wider range of incoming skills. Emphasis should continue to be shifted toward competency-based assessments and entrustment and away from a fixed training time or exam number threshold. Important financial aspects have also changed. The cost of practical machines has dropped considerably, and medicine is shifting away from a fee-for-service model. While it remains appropriate that physicians may bill for POCUS examinations, it is likely that improved diagnosis, improved throughput, and a reduction in complications will yield greater value and should be the emphasis of cost/value discussions.9 Finally, while hospitals may impose credentialing, this process can also create a burden not present for most other noninvasive skills and may deter appropriate use. If this approach is chosen by a hospital, requirements should ideally remain modest, and as these skills become more widespread, POCUS should ultimately be built into board examinations and core credentialing.9
Thoughtful and concerted effort will be required by hospitalist leaders, educational innovators, and professional societies in developing POCUS to best serve hospitalists and their patients. This work has already begun. For example, in 2019 SHM offered a position statement outlining important aspects such as current evidence-based applications, training pathways, quality assurance, and program management.10 These recommendations should guide both adult and pediatric hospitalists. The Alliance for Academic Internal Medicine offered a similar position statement for resident training.11 Interest groups are growing in numerous professional societies, which will facilitate collaboration and promote propagation of best practices. High-quality educational tools are continuing to be developed by numerous organizations.
While further development is needed to add the detail, granularity, and practical tools that educational and practice leaders need to assure that POCUS achieves its potential in hospital medicine, the foundation for POCUS use within the specialty is being thoughtfully constructed. As this process proceeds, it will be vital to continue to learn from our emergency medicine colleagues, who have already met similar challenges, while at the same time be able to develop a modern POCUS model optimized for hospital medicine workflow, training, and patient care.
The enthusiasm surrounding point-of-care ultrasound (POCUS) is clear and well founded. POCUS is a powerful tool that produces valuable diagnostic information for common and important clinical problems faced by hospitalists, such as pneumonia, soft-tissue infections,1 and myriad other applications. It can inform the evaluation and management of complex clinical problems such as dyspnea.2 Beyond its diagnostic potential, POCUS is well known to improve common procedures performed by adult and pediatric hospitalists by improving success rates and decreasing complications.
Excitement surrounding this technology continues to grow among hospitalists, leading to a proliferation of high-quality educational programs over the last 5 years. Most notable among these offerings has been the more comprehensive training available through the Society of Hospital Medicine (SHM) certificate-based pathway, though many other strong options exist, including institution-based curricula, such as the HealthPartners CHAMP program,3 and pediatric-focused programs. Growth in training is also occurring among medical students and residents. As of a 2012 survey, the majority (51%) of US medical schools had begun to weave ultrasound into their curricula,4 and this growth is also occurring in internal medicine and pediatric residency programs.5
Given the high potential for this technology and the growth in interest, it is an excellent time to pause and review some of the challenges faced by practitioners, hospitalist groups, and educators seeking to optimize POCUS implementation. A deliberate approach to POCUS education, the development of shared standards for high-quality use, and an ongoing dedication to develop specialty-specific practices will largely determine how much of this potential is fulfilled.
The largest challenge is likely to be educational. Educating clinicians to be able to integrate POCUS into practice is a complex, multistep process requiring not only an adequate core of didactic training and access to machines, but also the structured opportunity to develop rudimentary hands-on skills. Such initial training should be followed by continued practice and feedback as developing POCUS users progress toward independent practice. The study by Kumar et al.6 reaffirms that brief didactic lectures and access to machines are necessary, but they are clearly insufficient for learners to be able to use POCUS independently for a wide variety of applications. Their intervention also contrasts markedly with the 20 hours of didactics and 150 supervised scans recommended by the American College of Emergency Physicians prior to independent use for a core of six applications.7
Shared standards for education, use, and oversight will be crucial to fulfilling the potential of POCUS within hospital medicine. Our belief is that much can be learned from the thoughtful approach taken during the development of POCUS as a mainstream tool in emergency medicine in the early 2000s. In this approach, emergency physicians determined a sufficient and achievable standard of training for core POCUS applications, which was widely adopted. Based on completion of this training, physicians who were required to complete credentialing from their hospitals were widely able to achieve it, without any need for external certification. Emergency medicine guidelines further mandated the documentation of examinations and the creation of an exam report, features that improve clinical communication and facilitate quality improvement. Quality assurance processes that reviewed images and clinician interpretations were established as mandatory, which they should be in hospital medicine. Evidence was produced as to which exams physicians could do reliably with this focused training and which they could not. In the context of these thoughtful constructs, lawsuits have been noted to be exceedingly rare; and when they do occur, they have typically been for the failure to use POCUS rather than the converse.8
While many of these precepts deserve replication, others should also be modified to reflect changes in technology, medical education, and medical practice over the last 20 years and to improve upon this base of success. For example, with POCUS training now appearing in many medical school and residency curricula, training paradigms for both residents and attendings will need to accommodate a wider range of incoming skills. Emphasis should continue to be shifted toward competency-based assessments and entrustment and away from a fixed training time or exam number threshold. Important financial aspects have also changed. The cost of practical machines has dropped considerably, and medicine is shifting away from a fee-for-service model. While it remains appropriate that physicians may bill for POCUS examinations, it is likely that improved diagnosis, improved throughput, and a reduction in complications will yield greater value and should be the emphasis of cost/value discussions.9 Finally, while hospitals may impose credentialing, this process can also create a burden not present for most other noninvasive skills and may deter appropriate use. If this approach is chosen by a hospital, requirements should ideally remain modest, and as these skills become more widespread, POCUS should ultimately be built into board examinations and core credentialing.9
Thoughtful and concerted effort will be required by hospitalist leaders, educational innovators, and professional societies in developing POCUS to best serve hospitalists and their patients. This work has already begun. For example, in 2019 SHM offered a position statement outlining important aspects such as current evidence-based applications, training pathways, quality assurance, and program management.10 These recommendations should guide both adult and pediatric hospitalists. The Alliance for Academic Internal Medicine offered a similar position statement for resident training.11 Interest groups are growing in numerous professional societies, which will facilitate collaboration and promote propagation of best practices. High-quality educational tools are continuing to be developed by numerous organizations.
While further development is needed to add the detail, granularity, and practical tools that educational and practice leaders need to assure that POCUS achieves its potential in hospital medicine, the foundation for POCUS use within the specialty is being thoughtfully constructed. As this process proceeds, it will be vital to continue to learn from our emergency medicine colleagues, who have already met similar challenges, while at the same time be able to develop a modern POCUS model optimized for hospital medicine workflow, training, and patient care.
1. Kinnear B, Kelleher M, Chorny V. Clinical practice update: Point-of-care ultrasound for the pediatric hospitalist. J Hosp Med. 2019;15(3):170-172. https://doi.org/10.12788/jhm.3325.
2. Kelleher M, Kinnear B, Olson A. Clinical progress note: Point-of-care ultrasound in the evaluation of the dyspneic adult. J Hosp Med. 2020;15(3):173-175. https://doi.org/10.12788/jhm.3340.
3. Mathews BK, Reierson K, Vuong K, et al. The design and evaluation of the Comprehensive Hospitalist Assessment and Mentorship with Portfolios (CHAMP) Ultrasound Program. J Hosp Med. 2018;13(8):544-550. https://doi.org/10.12788/jhm.2938.
4. Bahner DP, Goldman E, Way D, Royall NA, Liu YT. The state of ultrasound education in U.S. medical schools: Results of a national survey. Acad Med. 2014;89(12):1681-1686. https://doi.org/10.1097/ACM.0000000000000414.
5. Reaume M, Siuba M, Wagner M, Woodwyk A, Melgar TA. Prevalence and Scope of point-of-care ultrasound education in internal medicine, pediatric, and medicine-pediatric residency programs in the United States. J Ultrasound Med. 2019;38(6):1433-1439. https://doi.org/10.1002/jum.14821.
6. Kumar A, Weng Y, Wang L, et al. Portable ultrasound device usage and learning outcomes among internal medicine trainees: a parallel-group randomized trial. J Hosp Med. 2020;15(3):154-159. https://doi.org/10.12788/jhm.3351.
7. Ultrasound Guidelines: Emergency, Point-of-Care and Clinical Ultrasound Guidelines in Medicine. Ann Emerg Med. 2017;69(5):e27-e54. https://doi.org/10.1016/j.annemergmed.2016.08.457.
8. Stolz L, O’Brien KM, Miller ML, Winters-Brown ND, Blaivas M, Adhikari S. A review of lawsuits related to point-of-care emergency ultrasound applications. West J Emerg Med. 2015;16(1):1-4. https://doi.org/10.5811/westjem.2014.11.23592.
9, Soni NJ, Tierney DM, Jensen TP, Lucas BP. Certification of Point-of-Care Ultrasound Competency. J Hosp Med. 2017;12(9):775-776. doi:10.12788/jhm.2812
10. Soni NJ, Schnobrich D, Matthews BK, et al. Point-of-Care Ultrasound for hospitalists: A position statement of the society of hospital medicine. J Hosp Med. 2019;14. https://doi.org/10.12788/jhm.3079.
11. LoPresti CM, Jensen TP, Dversdal RK, Astiz DJ. Point-of-Care Ultrasound for Internal Medicine Residency Training: A position statement from the Alliance of Academic Internal Medicine. Am J Med. 2019 Nov;132(11):1356-1360. https://doi.org/10.1016/j.amjmed.2019.07.019.
1. Kinnear B, Kelleher M, Chorny V. Clinical practice update: Point-of-care ultrasound for the pediatric hospitalist. J Hosp Med. 2019;15(3):170-172. https://doi.org/10.12788/jhm.3325.
2. Kelleher M, Kinnear B, Olson A. Clinical progress note: Point-of-care ultrasound in the evaluation of the dyspneic adult. J Hosp Med. 2020;15(3):173-175. https://doi.org/10.12788/jhm.3340.
3. Mathews BK, Reierson K, Vuong K, et al. The design and evaluation of the Comprehensive Hospitalist Assessment and Mentorship with Portfolios (CHAMP) Ultrasound Program. J Hosp Med. 2018;13(8):544-550. https://doi.org/10.12788/jhm.2938.
4. Bahner DP, Goldman E, Way D, Royall NA, Liu YT. The state of ultrasound education in U.S. medical schools: Results of a national survey. Acad Med. 2014;89(12):1681-1686. https://doi.org/10.1097/ACM.0000000000000414.
5. Reaume M, Siuba M, Wagner M, Woodwyk A, Melgar TA. Prevalence and Scope of point-of-care ultrasound education in internal medicine, pediatric, and medicine-pediatric residency programs in the United States. J Ultrasound Med. 2019;38(6):1433-1439. https://doi.org/10.1002/jum.14821.
6. Kumar A, Weng Y, Wang L, et al. Portable ultrasound device usage and learning outcomes among internal medicine trainees: a parallel-group randomized trial. J Hosp Med. 2020;15(3):154-159. https://doi.org/10.12788/jhm.3351.
7. Ultrasound Guidelines: Emergency, Point-of-Care and Clinical Ultrasound Guidelines in Medicine. Ann Emerg Med. 2017;69(5):e27-e54. https://doi.org/10.1016/j.annemergmed.2016.08.457.
8. Stolz L, O’Brien KM, Miller ML, Winters-Brown ND, Blaivas M, Adhikari S. A review of lawsuits related to point-of-care emergency ultrasound applications. West J Emerg Med. 2015;16(1):1-4. https://doi.org/10.5811/westjem.2014.11.23592.
9, Soni NJ, Tierney DM, Jensen TP, Lucas BP. Certification of Point-of-Care Ultrasound Competency. J Hosp Med. 2017;12(9):775-776. doi:10.12788/jhm.2812
10. Soni NJ, Schnobrich D, Matthews BK, et al. Point-of-Care Ultrasound for hospitalists: A position statement of the society of hospital medicine. J Hosp Med. 2019;14. https://doi.org/10.12788/jhm.3079.
11. LoPresti CM, Jensen TP, Dversdal RK, Astiz DJ. Point-of-Care Ultrasound for Internal Medicine Residency Training: A position statement from the Alliance of Academic Internal Medicine. Am J Med. 2019 Nov;132(11):1356-1360. https://doi.org/10.1016/j.amjmed.2019.07.019.
© 2020 Society of Hospital Medicine
MISSION Possible, but Incomplete: Pairing Better Access with Better Transitions in Veteran Care
What childhood game better captures communication exchange than “telephone”: as whispers pass from ear to ear, the original message degrades or transforms entirely. In complex healthcare systems, a more perilous version of “telephone” emerges, distinct from the well-worn metaphor: the signal never arrives at all. The primary care provider never even knew the patient was in the hospital; the discharge summary was never received; the patient cannot remember important details; and key medications are missing. In this edition of the Journal, Roman Ayele et al.1 used qualitative methods to explore this transitional black box between community hospitals and Veterans’ Affairs (VA) primary care clinics, illuminating how signal fragmentation may render the increasing use of care services outside the VA system as inversely proportionate to quality.
To understand why, a small amount of historical context is necessary. The VA has increasingly focused on expanding healthcare options to its nine million veterans. On June 6, 2019, the VA Maintaining Internal Systems and Strengthening Integrated Outside Networks (MISSION) Act was passed to consolidate existing programs and lower barriers for Veterans to seek care in non-VA urgent care and subspecialty settings.2 Though this act is not specifically focused on access to community hospitals, patients seeking urgent and subspecialty care are likely to be increasingly hospitalized outside of the VA due to geographic factors affecting point-of-care decisions. Concurrent with this expansion of options is the planned replacement of the VA’s legacy electronic health record, VistA.3 Both transformations indicate the need for the VA to be watchful and to intensify its focus on safe, effective exchanges of information.
Against this backdrop, Ayele et al.3 use stakeholder interviews with veterans and both non-VA and VA clinicians to identify the current lack of standardized practices for transitions of veteran care from community hospitals to VA primary care in Eastern Colorado. The themes most linked to care fragmentation included difficulty in identifying veterans and notifying VA primary care of hospital discharges, transferring medical records, making follow-up appointments, and coordinating prescribing with VA pharmacies. Participants identified incomplete or delayed information exchanges that were further complicated by the inability to confirm transmission across systems. A patchwork of postacute care solutions failed to prevent wasteful, low-value transitional care, including unscheduled primary care walk-ins and ED visits for medication refills. Participants arrived at a simple common solution: develop a clinically trained “VA liaison” to work at the interface between VA primary care and non-VA community hospitals so as to provide a single point of contact to coordinate these transitions. In short, to have someone to pick up the phone.
The strengths of this qualitative study lie in its insights into the current gaps in care transitions through the eyes of key stakeholders. By engaging patients and providers in imagining system changes that are actionable in the near- (clinical VA liaisons) and longer-term (pharmacy and EHR integration), Ayele et al. have provided a helpful starting place in studying and improving the interface between VA and non-VA care. Stakeholders emphasized the importance of a clear access point so that outside providers can easily notify VA clinics, arrange follow-ups, and streamline physician prescribing to avoid dangerous and costly delays in care.4 Though similar issues have been illuminated in prior work on care fragmentation,4 perspective in context is a fundamental strength of qualitative research, and further highlights the urgency of this period in veteran care.
There is the old adage: “if you have seen one VA, you have seen one VA”. This is arguably reflected in how each VA medical center is situated in a different regional and local healthcare delivery context, despite a common national infrastructure. The authors acknowledge limited generalizability but provide a framework for reproducing such work in regional VA systems. A national model for transitioning patients from regional community partners to VA primary care would require further testing, and to be a credible system-wide investment, would necessitate meaningful measurement across multiple sites. Given recent evidence of strong internal VA performance compared to the private sector,5 it is time for the VA to intensify focus on external care transitions. Given its history and continued commitment to funding innovation,6 the VA ought to be up to the task. Yet, as VA hospitalists, we know only too well that the system is increasingly under pressure to apply constrained resources inside and outside its own walls. Sending business elsewhere might not only fail at improving care but also weaken the fragile care delivery infrastructure.7
The idea that access and continuity may be in conflict raises an ethical question in modern practice and shared decision-making: how do we advise patients navigating complicated and imperfect health systems to understand the choices they are making and the risks they are taking when they spread care across systems? How are access and convenience weighed against the troubled movement of information across systems? How great is the risk if their care teams do not hear the same message? Knowing that increased fragmentation disproportionately affects the marginalized and vulnerable, especially those with complex chronic care needs,8 should we advise certain patients to stay in place within a single system?
As hospitalists, we are implied players in this dangerous version of the telephone game at a fascinating time in healthcare. Unlike when we advise patients on the risks and benefits of treatment, we have little evidence to guide our patients on when to stay put and when to leave to get care outside the system, inviting the risk of lost signals, garbled messages, and worst of all, frustrating, duplicative, unsafe care. As we strive for incremental improvements toward sweeping transformations in healthcare, we may for a few more years have to remind each other—and our students—of the incredible value of one more phone call: to make sure the intended message was
Disclaimer
The contents of this publication do not represent the views of the U.S. Department of Veterans Affairs or the United States Government.
1. Ayele RA, Lawrence E, McCreight M, et al. Perspectives of clinicians, staff, and veterans in transitioning veterans from non-VA hospitals to primary care in a single VA healthcare system. J Hosp Med. 2020;15(3):133-139. https://doi.org/10.12788/jhm.3320.
2. US Department of Veterans Affairs: VA Maintaining Internal Systems and Strengthening Integrated Outside Networks (MISSION) Act of 2018. https://missionact.va.gov/ at https://www.congress.gov/115/bills/s2372/BILLS-115s2372enr.pdf. Accessed October 31, 2019.
3. US Department of Veterans Affairs: VA EHR Modernization. ehrm.va.gov. Accessed October 31, 2019.
4. Thorpe JM, Thorpe CT, Schleiden L, et al. Association between dual use of Department of Veterans Affairs and Medicare Part D drug benefits and potentially unsafe prescribing. JAMA Intern Med. 2019;179(11):1584-1586. https://doi.org/10.1001/jamainternmed.2019.2788.
5. Weeks WB, West AN. Veterans Health Administration hospitals outperform non–Veterans health administration hospitals in most health care markets. Ann Intern Med. 2018;170(6):426-428. https://doi.org/10.7326/M18-1540.
6. US Department of Veterans Affairs: VA Innovation Center. https://www.innovation.va.gov/. Accessed October 31, 2019.
7. Shulkin, DL. Implications for veterans’ Health Care: the danger becomes clearer [published online ahead of print July 22, 2019. JAMA Intern Med. 2019. https://doi.org/10.1001/jamainternmed.2019.2996.
8. Englander H, Michaels L, Chan B, Kansagara D. The care transitions innovation (C-TraIn) for socioeconomically disadvantaged adults: results of a cluster randomized controlled trial. J Gen Intern Med. 2014;29(11):1460-1467. https://doi.org/10.1007/s11606-014-2903-0.
What childhood game better captures communication exchange than “telephone”: as whispers pass from ear to ear, the original message degrades or transforms entirely. In complex healthcare systems, a more perilous version of “telephone” emerges, distinct from the well-worn metaphor: the signal never arrives at all. The primary care provider never even knew the patient was in the hospital; the discharge summary was never received; the patient cannot remember important details; and key medications are missing. In this edition of the Journal, Roman Ayele et al.1 used qualitative methods to explore this transitional black box between community hospitals and Veterans’ Affairs (VA) primary care clinics, illuminating how signal fragmentation may render the increasing use of care services outside the VA system as inversely proportionate to quality.
To understand why, a small amount of historical context is necessary. The VA has increasingly focused on expanding healthcare options to its nine million veterans. On June 6, 2019, the VA Maintaining Internal Systems and Strengthening Integrated Outside Networks (MISSION) Act was passed to consolidate existing programs and lower barriers for Veterans to seek care in non-VA urgent care and subspecialty settings.2 Though this act is not specifically focused on access to community hospitals, patients seeking urgent and subspecialty care are likely to be increasingly hospitalized outside of the VA due to geographic factors affecting point-of-care decisions. Concurrent with this expansion of options is the planned replacement of the VA’s legacy electronic health record, VistA.3 Both transformations indicate the need for the VA to be watchful and to intensify its focus on safe, effective exchanges of information.
Against this backdrop, Ayele et al.3 use stakeholder interviews with veterans and both non-VA and VA clinicians to identify the current lack of standardized practices for transitions of veteran care from community hospitals to VA primary care in Eastern Colorado. The themes most linked to care fragmentation included difficulty in identifying veterans and notifying VA primary care of hospital discharges, transferring medical records, making follow-up appointments, and coordinating prescribing with VA pharmacies. Participants identified incomplete or delayed information exchanges that were further complicated by the inability to confirm transmission across systems. A patchwork of postacute care solutions failed to prevent wasteful, low-value transitional care, including unscheduled primary care walk-ins and ED visits for medication refills. Participants arrived at a simple common solution: develop a clinically trained “VA liaison” to work at the interface between VA primary care and non-VA community hospitals so as to provide a single point of contact to coordinate these transitions. In short, to have someone to pick up the phone.
The strengths of this qualitative study lie in its insights into the current gaps in care transitions through the eyes of key stakeholders. By engaging patients and providers in imagining system changes that are actionable in the near- (clinical VA liaisons) and longer-term (pharmacy and EHR integration), Ayele et al. have provided a helpful starting place in studying and improving the interface between VA and non-VA care. Stakeholders emphasized the importance of a clear access point so that outside providers can easily notify VA clinics, arrange follow-ups, and streamline physician prescribing to avoid dangerous and costly delays in care.4 Though similar issues have been illuminated in prior work on care fragmentation,4 perspective in context is a fundamental strength of qualitative research, and further highlights the urgency of this period in veteran care.
There is the old adage: “if you have seen one VA, you have seen one VA”. This is arguably reflected in how each VA medical center is situated in a different regional and local healthcare delivery context, despite a common national infrastructure. The authors acknowledge limited generalizability but provide a framework for reproducing such work in regional VA systems. A national model for transitioning patients from regional community partners to VA primary care would require further testing, and to be a credible system-wide investment, would necessitate meaningful measurement across multiple sites. Given recent evidence of strong internal VA performance compared to the private sector,5 it is time for the VA to intensify focus on external care transitions. Given its history and continued commitment to funding innovation,6 the VA ought to be up to the task. Yet, as VA hospitalists, we know only too well that the system is increasingly under pressure to apply constrained resources inside and outside its own walls. Sending business elsewhere might not only fail at improving care but also weaken the fragile care delivery infrastructure.7
The idea that access and continuity may be in conflict raises an ethical question in modern practice and shared decision-making: how do we advise patients navigating complicated and imperfect health systems to understand the choices they are making and the risks they are taking when they spread care across systems? How are access and convenience weighed against the troubled movement of information across systems? How great is the risk if their care teams do not hear the same message? Knowing that increased fragmentation disproportionately affects the marginalized and vulnerable, especially those with complex chronic care needs,8 should we advise certain patients to stay in place within a single system?
As hospitalists, we are implied players in this dangerous version of the telephone game at a fascinating time in healthcare. Unlike when we advise patients on the risks and benefits of treatment, we have little evidence to guide our patients on when to stay put and when to leave to get care outside the system, inviting the risk of lost signals, garbled messages, and worst of all, frustrating, duplicative, unsafe care. As we strive for incremental improvements toward sweeping transformations in healthcare, we may for a few more years have to remind each other—and our students—of the incredible value of one more phone call: to make sure the intended message was
Disclaimer
The contents of this publication do not represent the views of the U.S. Department of Veterans Affairs or the United States Government.
What childhood game better captures communication exchange than “telephone”: as whispers pass from ear to ear, the original message degrades or transforms entirely. In complex healthcare systems, a more perilous version of “telephone” emerges, distinct from the well-worn metaphor: the signal never arrives at all. The primary care provider never even knew the patient was in the hospital; the discharge summary was never received; the patient cannot remember important details; and key medications are missing. In this edition of the Journal, Roman Ayele et al.1 used qualitative methods to explore this transitional black box between community hospitals and Veterans’ Affairs (VA) primary care clinics, illuminating how signal fragmentation may render the increasing use of care services outside the VA system as inversely proportionate to quality.
To understand why, a small amount of historical context is necessary. The VA has increasingly focused on expanding healthcare options to its nine million veterans. On June 6, 2019, the VA Maintaining Internal Systems and Strengthening Integrated Outside Networks (MISSION) Act was passed to consolidate existing programs and lower barriers for Veterans to seek care in non-VA urgent care and subspecialty settings.2 Though this act is not specifically focused on access to community hospitals, patients seeking urgent and subspecialty care are likely to be increasingly hospitalized outside of the VA due to geographic factors affecting point-of-care decisions. Concurrent with this expansion of options is the planned replacement of the VA’s legacy electronic health record, VistA.3 Both transformations indicate the need for the VA to be watchful and to intensify its focus on safe, effective exchanges of information.
Against this backdrop, Ayele et al.3 use stakeholder interviews with veterans and both non-VA and VA clinicians to identify the current lack of standardized practices for transitions of veteran care from community hospitals to VA primary care in Eastern Colorado. The themes most linked to care fragmentation included difficulty in identifying veterans and notifying VA primary care of hospital discharges, transferring medical records, making follow-up appointments, and coordinating prescribing with VA pharmacies. Participants identified incomplete or delayed information exchanges that were further complicated by the inability to confirm transmission across systems. A patchwork of postacute care solutions failed to prevent wasteful, low-value transitional care, including unscheduled primary care walk-ins and ED visits for medication refills. Participants arrived at a simple common solution: develop a clinically trained “VA liaison” to work at the interface between VA primary care and non-VA community hospitals so as to provide a single point of contact to coordinate these transitions. In short, to have someone to pick up the phone.
The strengths of this qualitative study lie in its insights into the current gaps in care transitions through the eyes of key stakeholders. By engaging patients and providers in imagining system changes that are actionable in the near- (clinical VA liaisons) and longer-term (pharmacy and EHR integration), Ayele et al. have provided a helpful starting place in studying and improving the interface between VA and non-VA care. Stakeholders emphasized the importance of a clear access point so that outside providers can easily notify VA clinics, arrange follow-ups, and streamline physician prescribing to avoid dangerous and costly delays in care.4 Though similar issues have been illuminated in prior work on care fragmentation,4 perspective in context is a fundamental strength of qualitative research, and further highlights the urgency of this period in veteran care.
There is the old adage: “if you have seen one VA, you have seen one VA”. This is arguably reflected in how each VA medical center is situated in a different regional and local healthcare delivery context, despite a common national infrastructure. The authors acknowledge limited generalizability but provide a framework for reproducing such work in regional VA systems. A national model for transitioning patients from regional community partners to VA primary care would require further testing, and to be a credible system-wide investment, would necessitate meaningful measurement across multiple sites. Given recent evidence of strong internal VA performance compared to the private sector,5 it is time for the VA to intensify focus on external care transitions. Given its history and continued commitment to funding innovation,6 the VA ought to be up to the task. Yet, as VA hospitalists, we know only too well that the system is increasingly under pressure to apply constrained resources inside and outside its own walls. Sending business elsewhere might not only fail at improving care but also weaken the fragile care delivery infrastructure.7
The idea that access and continuity may be in conflict raises an ethical question in modern practice and shared decision-making: how do we advise patients navigating complicated and imperfect health systems to understand the choices they are making and the risks they are taking when they spread care across systems? How are access and convenience weighed against the troubled movement of information across systems? How great is the risk if their care teams do not hear the same message? Knowing that increased fragmentation disproportionately affects the marginalized and vulnerable, especially those with complex chronic care needs,8 should we advise certain patients to stay in place within a single system?
As hospitalists, we are implied players in this dangerous version of the telephone game at a fascinating time in healthcare. Unlike when we advise patients on the risks and benefits of treatment, we have little evidence to guide our patients on when to stay put and when to leave to get care outside the system, inviting the risk of lost signals, garbled messages, and worst of all, frustrating, duplicative, unsafe care. As we strive for incremental improvements toward sweeping transformations in healthcare, we may for a few more years have to remind each other—and our students—of the incredible value of one more phone call: to make sure the intended message was
Disclaimer
The contents of this publication do not represent the views of the U.S. Department of Veterans Affairs or the United States Government.
1. Ayele RA, Lawrence E, McCreight M, et al. Perspectives of clinicians, staff, and veterans in transitioning veterans from non-VA hospitals to primary care in a single VA healthcare system. J Hosp Med. 2020;15(3):133-139. https://doi.org/10.12788/jhm.3320.
2. US Department of Veterans Affairs: VA Maintaining Internal Systems and Strengthening Integrated Outside Networks (MISSION) Act of 2018. https://missionact.va.gov/ at https://www.congress.gov/115/bills/s2372/BILLS-115s2372enr.pdf. Accessed October 31, 2019.
3. US Department of Veterans Affairs: VA EHR Modernization. ehrm.va.gov. Accessed October 31, 2019.
4. Thorpe JM, Thorpe CT, Schleiden L, et al. Association between dual use of Department of Veterans Affairs and Medicare Part D drug benefits and potentially unsafe prescribing. JAMA Intern Med. 2019;179(11):1584-1586. https://doi.org/10.1001/jamainternmed.2019.2788.
5. Weeks WB, West AN. Veterans Health Administration hospitals outperform non–Veterans health administration hospitals in most health care markets. Ann Intern Med. 2018;170(6):426-428. https://doi.org/10.7326/M18-1540.
6. US Department of Veterans Affairs: VA Innovation Center. https://www.innovation.va.gov/. Accessed October 31, 2019.
7. Shulkin, DL. Implications for veterans’ Health Care: the danger becomes clearer [published online ahead of print July 22, 2019. JAMA Intern Med. 2019. https://doi.org/10.1001/jamainternmed.2019.2996.
8. Englander H, Michaels L, Chan B, Kansagara D. The care transitions innovation (C-TraIn) for socioeconomically disadvantaged adults: results of a cluster randomized controlled trial. J Gen Intern Med. 2014;29(11):1460-1467. https://doi.org/10.1007/s11606-014-2903-0.
1. Ayele RA, Lawrence E, McCreight M, et al. Perspectives of clinicians, staff, and veterans in transitioning veterans from non-VA hospitals to primary care in a single VA healthcare system. J Hosp Med. 2020;15(3):133-139. https://doi.org/10.12788/jhm.3320.
2. US Department of Veterans Affairs: VA Maintaining Internal Systems and Strengthening Integrated Outside Networks (MISSION) Act of 2018. https://missionact.va.gov/ at https://www.congress.gov/115/bills/s2372/BILLS-115s2372enr.pdf. Accessed October 31, 2019.
3. US Department of Veterans Affairs: VA EHR Modernization. ehrm.va.gov. Accessed October 31, 2019.
4. Thorpe JM, Thorpe CT, Schleiden L, et al. Association between dual use of Department of Veterans Affairs and Medicare Part D drug benefits and potentially unsafe prescribing. JAMA Intern Med. 2019;179(11):1584-1586. https://doi.org/10.1001/jamainternmed.2019.2788.
5. Weeks WB, West AN. Veterans Health Administration hospitals outperform non–Veterans health administration hospitals in most health care markets. Ann Intern Med. 2018;170(6):426-428. https://doi.org/10.7326/M18-1540.
6. US Department of Veterans Affairs: VA Innovation Center. https://www.innovation.va.gov/. Accessed October 31, 2019.
7. Shulkin, DL. Implications for veterans’ Health Care: the danger becomes clearer [published online ahead of print July 22, 2019. JAMA Intern Med. 2019. https://doi.org/10.1001/jamainternmed.2019.2996.
8. Englander H, Michaels L, Chan B, Kansagara D. The care transitions innovation (C-TraIn) for socioeconomically disadvantaged adults: results of a cluster randomized controlled trial. J Gen Intern Med. 2014;29(11):1460-1467. https://doi.org/10.1007/s11606-014-2903-0.
© 2020 Society of Hospital Medicine
Contrasting qSOFA and SIRS Criteria for Early Sepsis Identification in a Veteran Population (FULL)
Sepsis is a major public health concern: 10% of patients with sepsis die, and mortality quadruples with progression to septic shock.1 Systemic inflammatory response syndrome (SIRS) criteria, originally published in 1992, are commonly used to detect sepsis, but as early as 2001, these criteria were recognized as lacking specificity.2 Nonetheless, the use of SIRS criteria has persisted in practice. Sepsis was redefined in Sepsis-3 (2016) to guide earlier and more appropriate identification and treatment, which has been shown to greatly improve patient outcomes.1,3 Key recommendations in Sepsis 3 included eliminating SIRS criteria, defining organ dysfunction by the Sequential Organ Failure Assessment (SOFA) score, and introducing the quick SOFA (qSOFA) score.1
The qSOFA combines 3 clinical variables to provide a rapid, simple bedside score that measures the likelihood of poor outcomes, such as admission to an intensive care unit (ICU) or mortality in adults with suspected infection.1,3 The qSOFA score is intended to aid healthcare professionals in more timely stratification of those patients who need escalated care to prevent deterioration.1 The assessment also has been explored as a screening tool for sepsis in clinical practice; however, limited data exists concerning the comparative utility of qSOFA and SIRS in this capacity, and study results are inconsistent.4-6
The most important attribute of a screening tool is high sensitivity, but high specificity also is desired. The qSOFA could supplant SIRS as a screening tool for sepsis if it maintained similarly high sensitivity but achieved superior specificity. Therefore, our primary objective for this study was to determine the effectiveness of qSOFA as a screening assessment for sepsis in the setting of a general inpatient medicine service by contrasting the sensitivity and specificity of qSOFA with SIRS in predicting sepsis, using a retrospective chart review design.
Methods
Administrative data from the Department of Veterans Affairs (VA) Corporate Data Warehouse were accessed via the VA Informatics and Computing Infrastructure (VINCI) and used to identify VA inpatient admissions and obtain the laboratory and vital sign data necessary to calculate SIRS, qSOFA, and SOFA scores. The data were supplemented by manual review of VA health records to obtain information that was not readily available in administrative records, including septic shock outcomes and laboratory and vital sign data obtained in the ICU. This study was approved by the institutional review board at the University of Iowa and the research and development committee at the Iowa City VA Medical Center (ICVAMC).
Patients
The study population included veterans admitted to the nonsurgical medicine unit at ICVAMC between August 1, 2014 and August 1, 2016 who were transferred to an ICU after admission; direct ICU admissions were not included as the qSOFA has been shown in studies to be more beneficial and offer better predictive validity outside the ICU. Excluding these direct admissions prevented any potential skewing of the data. To control for possible selection bias, veterans also were excluded if they transferred from another facility, were admitted under observation status, or if they had been admitted within the prior 30 days. These patients may have been more critically ill than those who presented directly to our facility and any prior treatment could affect the clinical status of the patient and assessment for sepsis at the time of presentation to the VA. Veterans were further required to have evidence of suspected infection based on manual review of the health record, which was determined by receipt of an antibiotic relevant to the empiric treatment of sepsis within 48 hours of admission.
Sepsis and Septic Shock Assessment Tools
As outlined in the Sepsis-3 guidelines, sepsis was defined as suspected or confirmed infection with an acute change in the SOFA score of ≥ 2 points, which is assumed to be 0 in those not known to have preexisting dysfunction.1 The SOFA score includes variables from the respiratory, coagulation, hepatic, cardiovascular, renal, and central nervous systems.1 Septic shock was defined as vasopressor administration and a serum lactic acid level > 2 mmol/L occurring up to 24 hours apart and within 3 days of the first antibiotic dose administered.
The SIRS assessment includes 4 clinical variables (temperature, heart rate, respiratory rate, and white blood cell count) while qSOFA is comprised of 3 variables (respiratory rate, systolic blood pressure, and altered mental status).1 With both assessments, a score ≥ 2 is considered positive, which indicates increased risk for sepsis in patients with suspected infection.1 In keeping with existing studies, qSOFA and SIRS assessments were scored using maximum values found within 48 hours before and 24 hours after the first administered antibiotic dose.3
Outcomes
The primary outcome variable was the presence of sepsis in adults with evidence of infection within 48 hours of admission. Secondary outcome measures included 30-day mortality and septic shock.
Performance between the SIRS and qSOFA assessments was contrasted using sensitivity, specificity, and positive and negative predictive value measurements. Associations of qSOFA and SIRS with septic shock and 30-day mortality were evaluated using a 2-tailed Fisher’s exact test with a threshold of α = 0.05 to determine statistical significance.
Results
The study sample of 481 veterans had a mean age of 67.4 years, 94% were male, and 91.1% were white (Table 1).
Scores for qSOFA, but not SIRS, were significantly associated with septic shock (Fisher’s exact test; qSOFA: P = .009; SIRS: P = .58) (Table 3).
Discussion
High sensitivity is critical for a sepsis screening tool. To be clinically useful, it has been suggested that biomarkers predicting poor outcomes for sepsis should have a sensitivity of > 80%.4 Although qSOFA demonstrated greater specificity than SIRS in our study (83.6% vs 25.7%), qSOFA showed lower sensitivity (44.7% vs 80.0%), which resulted in a greater potential for false negatives; 55.3% of those with sepsis would go undetected. Therefore, our study does not support qSOFA as a better screening assessment than SIRS for sepsis in the veteran population.
Most studies concur with our findings of low sensitivity and high specificity of qSOFA. In a systematic review and meta-analysis, Serafim and colleagues identified 10 studies published after Sepsis-3 that reported sensitivity or specificity of qSOFA and SIRS for sepsis diagnosis.5 Seven of the 10 studies reported sensitivities and favored SIRS in the diagnosis of sepsis (Relative risk: 1.32; 95% CI: 0.40-2.24; P < .0001; I2 = 100%). The authors noted that substantial heterogeneity among studies, including differences in study design, sample size, and criteria for determination of infection, was an important limitation. In addition, most studies that contrast qSOFA and SIRS center on prognostic value in predicting mortality, rather than as a screening test for a diagnosis of sepsis.
We concluded SIRS was more sensitive and thus superior to qSOFA when used as a screening tool for sepsis but conceded that more prospective and homogenous investigations were necessary. To our knowledge, only 1 published study has deviated from this conclusion and reported comparable sensitivity between SIRS (92%) and qSOFA (90%).6 Our study adds to existing literature as it is the first conducted in a veteran population. Additionally, we performed our investigation in a general medicine population with methods similar to existing literature, including the key study validating clinical criteria for sepsis by Seymour and colleagues.3
Limitations
This study is not without limitations, including potential misclassification of cases if essential data points were not available during data collection via health record review or the data points were not representative of a true change from baseline (eg, the Glasgow Coma Scale score for altered mental status in the qSOFA or the SOFA score for organ dysfunction). Generalizability of the results also may be limited due to our retrospective, single-center design and characteristics typical of a veteran population (eg, older, white males). Additionally, many veterans were excluded from the study if they transferred from another facility. These veterans may have been more critically ill than those who presented directly to our facility, which possibly introduced selection bias.
Conclusion
Our findings do not support use of the qSOFA as a suitable replacement for SIRS as a sepsis screening tool among patients with suspected infection in the general medicine inpatient setting. The clinical concern with SIRS is that unfavorable specificity leads to unnecessary antibiotic exposure among patients who are falsely positive. While qSOFA has demonstrated higher specificity, its use would cause many sepsis cases to go undetected due to the technique’s low sensitivity. Frequent false negative qSOFA results could thus serve to impede, rather than enhance, early recognition and intervention for sepsis.
The ideal sepsis screening tool is rapid and possesses high sensitivity and specificity to promptly identify and manage sepsis and avert unfavorable outcomes such as septic shock and death. While the SIRS criteria do not satisfy these ideal features, its measurement characteristics are more suitable for the application of sepsis screening than the qSOFA and should thus remain the standard tool in this setting. Future prospectively designed studies with more uniform methodologies are necessary to ascertain the most effective approach to identify sepsis for which novel screening approaches with more clinically suitable measurement properties are greatly needed.
Acknowledgements
This research was supported by the Iowa City VA Health Care System, Department of Pharmacy Services. Additional support was provided by the Health Services Research and Development Service, Department of Veterans Affairs.
1. Singer M, Deutchman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-810.
2. Levy MM, Fink MP, Marshall JC, et al; SCCM/ESICM/ACCP/ATS/SIS. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 2003;31(4):1250-1256.
3. Seymour CW, Liu VX, Iwashyna TJ, et al. Assessment of clinical criteria for sepsis: for the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):762-774.
4. Giamorellos-Bourboulis EJ, Tsaganos T, Tsangaris I, et al; Hellenic Sepsis Study Group. Validation of the new Sepsis-3 definitions: proposal for improvement of early risk identification. Clin Microbiol Infect. 2016;23(2):104-109.
5. Serafim R, Gomes JA, Salluh J, Póvoa P. A Comparison of the Quick-SOFA and Systemic Inflammatory Response Syndrome criteria for the diagnosis of sepsis and prediction of mortality: a systematic review and meta-analysis. Chest. 2018;153(3):646-655.
6. Forward E, Konecny P, Burston J, Adhikari S, Doolan H, Jensen T. Predictive validity of qSOFA criteria for sepsis in non-ICU patients. Intensive Care Med. 2017;43(6):945-946.
Sepsis is a major public health concern: 10% of patients with sepsis die, and mortality quadruples with progression to septic shock.1 Systemic inflammatory response syndrome (SIRS) criteria, originally published in 1992, are commonly used to detect sepsis, but as early as 2001, these criteria were recognized as lacking specificity.2 Nonetheless, the use of SIRS criteria has persisted in practice. Sepsis was redefined in Sepsis-3 (2016) to guide earlier and more appropriate identification and treatment, which has been shown to greatly improve patient outcomes.1,3 Key recommendations in Sepsis 3 included eliminating SIRS criteria, defining organ dysfunction by the Sequential Organ Failure Assessment (SOFA) score, and introducing the quick SOFA (qSOFA) score.1
The qSOFA combines 3 clinical variables to provide a rapid, simple bedside score that measures the likelihood of poor outcomes, such as admission to an intensive care unit (ICU) or mortality in adults with suspected infection.1,3 The qSOFA score is intended to aid healthcare professionals in more timely stratification of those patients who need escalated care to prevent deterioration.1 The assessment also has been explored as a screening tool for sepsis in clinical practice; however, limited data exists concerning the comparative utility of qSOFA and SIRS in this capacity, and study results are inconsistent.4-6
The most important attribute of a screening tool is high sensitivity, but high specificity also is desired. The qSOFA could supplant SIRS as a screening tool for sepsis if it maintained similarly high sensitivity but achieved superior specificity. Therefore, our primary objective for this study was to determine the effectiveness of qSOFA as a screening assessment for sepsis in the setting of a general inpatient medicine service by contrasting the sensitivity and specificity of qSOFA with SIRS in predicting sepsis, using a retrospective chart review design.
Methods
Administrative data from the Department of Veterans Affairs (VA) Corporate Data Warehouse were accessed via the VA Informatics and Computing Infrastructure (VINCI) and used to identify VA inpatient admissions and obtain the laboratory and vital sign data necessary to calculate SIRS, qSOFA, and SOFA scores. The data were supplemented by manual review of VA health records to obtain information that was not readily available in administrative records, including septic shock outcomes and laboratory and vital sign data obtained in the ICU. This study was approved by the institutional review board at the University of Iowa and the research and development committee at the Iowa City VA Medical Center (ICVAMC).
Patients
The study population included veterans admitted to the nonsurgical medicine unit at ICVAMC between August 1, 2014 and August 1, 2016 who were transferred to an ICU after admission; direct ICU admissions were not included as the qSOFA has been shown in studies to be more beneficial and offer better predictive validity outside the ICU. Excluding these direct admissions prevented any potential skewing of the data. To control for possible selection bias, veterans also were excluded if they transferred from another facility, were admitted under observation status, or if they had been admitted within the prior 30 days. These patients may have been more critically ill than those who presented directly to our facility and any prior treatment could affect the clinical status of the patient and assessment for sepsis at the time of presentation to the VA. Veterans were further required to have evidence of suspected infection based on manual review of the health record, which was determined by receipt of an antibiotic relevant to the empiric treatment of sepsis within 48 hours of admission.
Sepsis and Septic Shock Assessment Tools
As outlined in the Sepsis-3 guidelines, sepsis was defined as suspected or confirmed infection with an acute change in the SOFA score of ≥ 2 points, which is assumed to be 0 in those not known to have preexisting dysfunction.1 The SOFA score includes variables from the respiratory, coagulation, hepatic, cardiovascular, renal, and central nervous systems.1 Septic shock was defined as vasopressor administration and a serum lactic acid level > 2 mmol/L occurring up to 24 hours apart and within 3 days of the first antibiotic dose administered.
The SIRS assessment includes 4 clinical variables (temperature, heart rate, respiratory rate, and white blood cell count) while qSOFA is comprised of 3 variables (respiratory rate, systolic blood pressure, and altered mental status).1 With both assessments, a score ≥ 2 is considered positive, which indicates increased risk for sepsis in patients with suspected infection.1 In keeping with existing studies, qSOFA and SIRS assessments were scored using maximum values found within 48 hours before and 24 hours after the first administered antibiotic dose.3
Outcomes
The primary outcome variable was the presence of sepsis in adults with evidence of infection within 48 hours of admission. Secondary outcome measures included 30-day mortality and septic shock.
Performance between the SIRS and qSOFA assessments was contrasted using sensitivity, specificity, and positive and negative predictive value measurements. Associations of qSOFA and SIRS with septic shock and 30-day mortality were evaluated using a 2-tailed Fisher’s exact test with a threshold of α = 0.05 to determine statistical significance.
Results
The study sample of 481 veterans had a mean age of 67.4 years, 94% were male, and 91.1% were white (Table 1).
Scores for qSOFA, but not SIRS, were significantly associated with septic shock (Fisher’s exact test; qSOFA: P = .009; SIRS: P = .58) (Table 3).
Discussion
High sensitivity is critical for a sepsis screening tool. To be clinically useful, it has been suggested that biomarkers predicting poor outcomes for sepsis should have a sensitivity of > 80%.4 Although qSOFA demonstrated greater specificity than SIRS in our study (83.6% vs 25.7%), qSOFA showed lower sensitivity (44.7% vs 80.0%), which resulted in a greater potential for false negatives; 55.3% of those with sepsis would go undetected. Therefore, our study does not support qSOFA as a better screening assessment than SIRS for sepsis in the veteran population.
Most studies concur with our findings of low sensitivity and high specificity of qSOFA. In a systematic review and meta-analysis, Serafim and colleagues identified 10 studies published after Sepsis-3 that reported sensitivity or specificity of qSOFA and SIRS for sepsis diagnosis.5 Seven of the 10 studies reported sensitivities and favored SIRS in the diagnosis of sepsis (Relative risk: 1.32; 95% CI: 0.40-2.24; P < .0001; I2 = 100%). The authors noted that substantial heterogeneity among studies, including differences in study design, sample size, and criteria for determination of infection, was an important limitation. In addition, most studies that contrast qSOFA and SIRS center on prognostic value in predicting mortality, rather than as a screening test for a diagnosis of sepsis.
We concluded SIRS was more sensitive and thus superior to qSOFA when used as a screening tool for sepsis but conceded that more prospective and homogenous investigations were necessary. To our knowledge, only 1 published study has deviated from this conclusion and reported comparable sensitivity between SIRS (92%) and qSOFA (90%).6 Our study adds to existing literature as it is the first conducted in a veteran population. Additionally, we performed our investigation in a general medicine population with methods similar to existing literature, including the key study validating clinical criteria for sepsis by Seymour and colleagues.3
Limitations
This study is not without limitations, including potential misclassification of cases if essential data points were not available during data collection via health record review or the data points were not representative of a true change from baseline (eg, the Glasgow Coma Scale score for altered mental status in the qSOFA or the SOFA score for organ dysfunction). Generalizability of the results also may be limited due to our retrospective, single-center design and characteristics typical of a veteran population (eg, older, white males). Additionally, many veterans were excluded from the study if they transferred from another facility. These veterans may have been more critically ill than those who presented directly to our facility, which possibly introduced selection bias.
Conclusion
Our findings do not support use of the qSOFA as a suitable replacement for SIRS as a sepsis screening tool among patients with suspected infection in the general medicine inpatient setting. The clinical concern with SIRS is that unfavorable specificity leads to unnecessary antibiotic exposure among patients who are falsely positive. While qSOFA has demonstrated higher specificity, its use would cause many sepsis cases to go undetected due to the technique’s low sensitivity. Frequent false negative qSOFA results could thus serve to impede, rather than enhance, early recognition and intervention for sepsis.
The ideal sepsis screening tool is rapid and possesses high sensitivity and specificity to promptly identify and manage sepsis and avert unfavorable outcomes such as septic shock and death. While the SIRS criteria do not satisfy these ideal features, its measurement characteristics are more suitable for the application of sepsis screening than the qSOFA and should thus remain the standard tool in this setting. Future prospectively designed studies with more uniform methodologies are necessary to ascertain the most effective approach to identify sepsis for which novel screening approaches with more clinically suitable measurement properties are greatly needed.
Acknowledgements
This research was supported by the Iowa City VA Health Care System, Department of Pharmacy Services. Additional support was provided by the Health Services Research and Development Service, Department of Veterans Affairs.
Sepsis is a major public health concern: 10% of patients with sepsis die, and mortality quadruples with progression to septic shock.1 Systemic inflammatory response syndrome (SIRS) criteria, originally published in 1992, are commonly used to detect sepsis, but as early as 2001, these criteria were recognized as lacking specificity.2 Nonetheless, the use of SIRS criteria has persisted in practice. Sepsis was redefined in Sepsis-3 (2016) to guide earlier and more appropriate identification and treatment, which has been shown to greatly improve patient outcomes.1,3 Key recommendations in Sepsis 3 included eliminating SIRS criteria, defining organ dysfunction by the Sequential Organ Failure Assessment (SOFA) score, and introducing the quick SOFA (qSOFA) score.1
The qSOFA combines 3 clinical variables to provide a rapid, simple bedside score that measures the likelihood of poor outcomes, such as admission to an intensive care unit (ICU) or mortality in adults with suspected infection.1,3 The qSOFA score is intended to aid healthcare professionals in more timely stratification of those patients who need escalated care to prevent deterioration.1 The assessment also has been explored as a screening tool for sepsis in clinical practice; however, limited data exists concerning the comparative utility of qSOFA and SIRS in this capacity, and study results are inconsistent.4-6
The most important attribute of a screening tool is high sensitivity, but high specificity also is desired. The qSOFA could supplant SIRS as a screening tool for sepsis if it maintained similarly high sensitivity but achieved superior specificity. Therefore, our primary objective for this study was to determine the effectiveness of qSOFA as a screening assessment for sepsis in the setting of a general inpatient medicine service by contrasting the sensitivity and specificity of qSOFA with SIRS in predicting sepsis, using a retrospective chart review design.
Methods
Administrative data from the Department of Veterans Affairs (VA) Corporate Data Warehouse were accessed via the VA Informatics and Computing Infrastructure (VINCI) and used to identify VA inpatient admissions and obtain the laboratory and vital sign data necessary to calculate SIRS, qSOFA, and SOFA scores. The data were supplemented by manual review of VA health records to obtain information that was not readily available in administrative records, including septic shock outcomes and laboratory and vital sign data obtained in the ICU. This study was approved by the institutional review board at the University of Iowa and the research and development committee at the Iowa City VA Medical Center (ICVAMC).
Patients
The study population included veterans admitted to the nonsurgical medicine unit at ICVAMC between August 1, 2014 and August 1, 2016 who were transferred to an ICU after admission; direct ICU admissions were not included as the qSOFA has been shown in studies to be more beneficial and offer better predictive validity outside the ICU. Excluding these direct admissions prevented any potential skewing of the data. To control for possible selection bias, veterans also were excluded if they transferred from another facility, were admitted under observation status, or if they had been admitted within the prior 30 days. These patients may have been more critically ill than those who presented directly to our facility and any prior treatment could affect the clinical status of the patient and assessment for sepsis at the time of presentation to the VA. Veterans were further required to have evidence of suspected infection based on manual review of the health record, which was determined by receipt of an antibiotic relevant to the empiric treatment of sepsis within 48 hours of admission.
Sepsis and Septic Shock Assessment Tools
As outlined in the Sepsis-3 guidelines, sepsis was defined as suspected or confirmed infection with an acute change in the SOFA score of ≥ 2 points, which is assumed to be 0 in those not known to have preexisting dysfunction.1 The SOFA score includes variables from the respiratory, coagulation, hepatic, cardiovascular, renal, and central nervous systems.1 Septic shock was defined as vasopressor administration and a serum lactic acid level > 2 mmol/L occurring up to 24 hours apart and within 3 days of the first antibiotic dose administered.
The SIRS assessment includes 4 clinical variables (temperature, heart rate, respiratory rate, and white blood cell count) while qSOFA is comprised of 3 variables (respiratory rate, systolic blood pressure, and altered mental status).1 With both assessments, a score ≥ 2 is considered positive, which indicates increased risk for sepsis in patients with suspected infection.1 In keeping with existing studies, qSOFA and SIRS assessments were scored using maximum values found within 48 hours before and 24 hours after the first administered antibiotic dose.3
Outcomes
The primary outcome variable was the presence of sepsis in adults with evidence of infection within 48 hours of admission. Secondary outcome measures included 30-day mortality and septic shock.
Performance between the SIRS and qSOFA assessments was contrasted using sensitivity, specificity, and positive and negative predictive value measurements. Associations of qSOFA and SIRS with septic shock and 30-day mortality were evaluated using a 2-tailed Fisher’s exact test with a threshold of α = 0.05 to determine statistical significance.
Results
The study sample of 481 veterans had a mean age of 67.4 years, 94% were male, and 91.1% were white (Table 1).
Scores for qSOFA, but not SIRS, were significantly associated with septic shock (Fisher’s exact test; qSOFA: P = .009; SIRS: P = .58) (Table 3).
Discussion
High sensitivity is critical for a sepsis screening tool. To be clinically useful, it has been suggested that biomarkers predicting poor outcomes for sepsis should have a sensitivity of > 80%.4 Although qSOFA demonstrated greater specificity than SIRS in our study (83.6% vs 25.7%), qSOFA showed lower sensitivity (44.7% vs 80.0%), which resulted in a greater potential for false negatives; 55.3% of those with sepsis would go undetected. Therefore, our study does not support qSOFA as a better screening assessment than SIRS for sepsis in the veteran population.
Most studies concur with our findings of low sensitivity and high specificity of qSOFA. In a systematic review and meta-analysis, Serafim and colleagues identified 10 studies published after Sepsis-3 that reported sensitivity or specificity of qSOFA and SIRS for sepsis diagnosis.5 Seven of the 10 studies reported sensitivities and favored SIRS in the diagnosis of sepsis (Relative risk: 1.32; 95% CI: 0.40-2.24; P < .0001; I2 = 100%). The authors noted that substantial heterogeneity among studies, including differences in study design, sample size, and criteria for determination of infection, was an important limitation. In addition, most studies that contrast qSOFA and SIRS center on prognostic value in predicting mortality, rather than as a screening test for a diagnosis of sepsis.
We concluded SIRS was more sensitive and thus superior to qSOFA when used as a screening tool for sepsis but conceded that more prospective and homogenous investigations were necessary. To our knowledge, only 1 published study has deviated from this conclusion and reported comparable sensitivity between SIRS (92%) and qSOFA (90%).6 Our study adds to existing literature as it is the first conducted in a veteran population. Additionally, we performed our investigation in a general medicine population with methods similar to existing literature, including the key study validating clinical criteria for sepsis by Seymour and colleagues.3
Limitations
This study is not without limitations, including potential misclassification of cases if essential data points were not available during data collection via health record review or the data points were not representative of a true change from baseline (eg, the Glasgow Coma Scale score for altered mental status in the qSOFA or the SOFA score for organ dysfunction). Generalizability of the results also may be limited due to our retrospective, single-center design and characteristics typical of a veteran population (eg, older, white males). Additionally, many veterans were excluded from the study if they transferred from another facility. These veterans may have been more critically ill than those who presented directly to our facility, which possibly introduced selection bias.
Conclusion
Our findings do not support use of the qSOFA as a suitable replacement for SIRS as a sepsis screening tool among patients with suspected infection in the general medicine inpatient setting. The clinical concern with SIRS is that unfavorable specificity leads to unnecessary antibiotic exposure among patients who are falsely positive. While qSOFA has demonstrated higher specificity, its use would cause many sepsis cases to go undetected due to the technique’s low sensitivity. Frequent false negative qSOFA results could thus serve to impede, rather than enhance, early recognition and intervention for sepsis.
The ideal sepsis screening tool is rapid and possesses high sensitivity and specificity to promptly identify and manage sepsis and avert unfavorable outcomes such as septic shock and death. While the SIRS criteria do not satisfy these ideal features, its measurement characteristics are more suitable for the application of sepsis screening than the qSOFA and should thus remain the standard tool in this setting. Future prospectively designed studies with more uniform methodologies are necessary to ascertain the most effective approach to identify sepsis for which novel screening approaches with more clinically suitable measurement properties are greatly needed.
Acknowledgements
This research was supported by the Iowa City VA Health Care System, Department of Pharmacy Services. Additional support was provided by the Health Services Research and Development Service, Department of Veterans Affairs.
1. Singer M, Deutchman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-810.
2. Levy MM, Fink MP, Marshall JC, et al; SCCM/ESICM/ACCP/ATS/SIS. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 2003;31(4):1250-1256.
3. Seymour CW, Liu VX, Iwashyna TJ, et al. Assessment of clinical criteria for sepsis: for the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):762-774.
4. Giamorellos-Bourboulis EJ, Tsaganos T, Tsangaris I, et al; Hellenic Sepsis Study Group. Validation of the new Sepsis-3 definitions: proposal for improvement of early risk identification. Clin Microbiol Infect. 2016;23(2):104-109.
5. Serafim R, Gomes JA, Salluh J, Póvoa P. A Comparison of the Quick-SOFA and Systemic Inflammatory Response Syndrome criteria for the diagnosis of sepsis and prediction of mortality: a systematic review and meta-analysis. Chest. 2018;153(3):646-655.
6. Forward E, Konecny P, Burston J, Adhikari S, Doolan H, Jensen T. Predictive validity of qSOFA criteria for sepsis in non-ICU patients. Intensive Care Med. 2017;43(6):945-946.
1. Singer M, Deutchman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-810.
2. Levy MM, Fink MP, Marshall JC, et al; SCCM/ESICM/ACCP/ATS/SIS. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 2003;31(4):1250-1256.
3. Seymour CW, Liu VX, Iwashyna TJ, et al. Assessment of clinical criteria for sepsis: for the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):762-774.
4. Giamorellos-Bourboulis EJ, Tsaganos T, Tsangaris I, et al; Hellenic Sepsis Study Group. Validation of the new Sepsis-3 definitions: proposal for improvement of early risk identification. Clin Microbiol Infect. 2016;23(2):104-109.
5. Serafim R, Gomes JA, Salluh J, Póvoa P. A Comparison of the Quick-SOFA and Systemic Inflammatory Response Syndrome criteria for the diagnosis of sepsis and prediction of mortality: a systematic review and meta-analysis. Chest. 2018;153(3):646-655.
6. Forward E, Konecny P, Burston J, Adhikari S, Doolan H, Jensen T. Predictive validity of qSOFA criteria for sepsis in non-ICU patients. Intensive Care Med. 2017;43(6):945-946.
Clinical Guideline Highlights for the Hospitalist: The GOLD and NICE Guidelines for the Management of COPD
Chronic obstructive pulmonary disease (COPD), projected to be the third leading cause of death by 2020, accounts for 6% of deaths globally.3 Hospitalization for COPD exacerbations is common and impacts patients’ disease trajectory, and mortality, with fewer than half of patients hospitalized for exacerbation surviving 5 years.4 Hospitalization provides an opportunity to optimize care. Due to recent practice-changing evidence, the National Institute for Health and Care Excellence (NICE) and the Global Initiative for Chronic Obstructive Lung Disease (GOLD) published updated guidelines.
KEY RECOMMENDATIONS
These are selected recommendations relevant to adult hospitalists. The GOLD guidelines grade recommendations by evidence strength from category A (randomized control trial data) to category D (expert consensus). The NICE guidelines relay strength of evidence through terminology referring to the presence or absence of a strong recommendation. Recommendations without evidence level specified are NS.
Diagnosis and Classification of COPD Severity
Recommendation 1. In patients with risk factors for and symptoms of COPD, spirometry is required to confirm the diagnosis, defined as a postbronchodilator FEV1/FVC ratio of <0.7 (NS, NICE, GOLD). The Global Lung Function Initiative (GLI) 2012 reference ranges5 are recommended (NS, NICE). Recommendation 2. Severity of airflow obstruction should be assessed according to reduction in the postbronchodilator FEV1 as: Stage I, Mild: FEV1 ≥80%; Stage II, Moderate: FEV1 = 50-79%; Stage III, Severe FEV1 = 30%-49%; Stage IV, FEV1<30% (NS, NICE, GOLD). Recommendation 3. Reversibility testing (aka bronchodilator response) does not indicate long-term response to therapy (NS, NICE, GOLD). Recommendation 4. The combined COPD assessment to classify patient symptoms and disease severity in one of four groups (A, B, C, or D) based on exacerbation history and daily symptom control (NS, GOLD). Use the Medical Research Council dyspnea scale to classify symptoms (strong, NICE).
Pharmacologic COPD Management
Recommendation 5. Short-acting inhaled bronchodilators such as short-acting beta2 agonists (SABAs) or short-acting muscarinic antagonists (SAMAs) improve FEV1 and symptoms. Combining SABA/SAMA is superior to monotherapy (A, GOLD). Recommendation 6. Long-acting bronchodilators, such as long-acting antimuscarinics (LAMAs) or long-acting beta2 agonists (LABAs), improve lung function and dyspnea and reduce exacerbations. Combination therapy (LABA/LAMA) is superior to using a single agent (LABA or LAMA) for improving FEV1 and reducing exacerbations (A, GOLD). Recommendation 7. Triple therapy of inhaled corticosteroid ICS/LAMA/LABA is more effective than the individual components in reducing exacerbations in the case of moderate to severe COPD (A, GOLD). Recommendation 8. Treatment with an ICS increases pneumonia risk (A, GOLD). Discuss these side effects (Strong, NICE). Recommendation 9. Use SABAs and SAMAs as initial treatment for patients with COPD (Strong, NICE). LABAs and LAMAs are preferred over short-acting agents except for patients with mild symptoms (A, GOLD). Recommendation 10. For symptomatic patients on long-acting monotherapy, escalate to combination LABA/LAMA, or if asthmatic features or elevated eosinophils (≥300 cells/µL) are present, combination LABA/ICS (A, GOLD; Strong, NICE). Recommendation 11. Assess and correct patient inhaler technique (NS, GOLD; Strong, NICE).
Nonpharmacologic COPD Management
Oxygen. Recommendation 12. Long-term oxygen supplementation increases survival in patients with resting arterial hypoxemia (PaO2<55 mm Hg) or hypoxemia (PaO2<60 mm Hg) with cor pulmonale (A, GOLD). Recommendation 13. In patients with moderate resting (89%-93%) or exercise-induced arterial desaturation (80%-90%), long-term oxygen does not improve outcomes (A, GOLD).6 Recommendation 14. Consider long-term oxygan after a risk assessment of fall and burn risk. Do not offer oxygen to those who continue to smoke (Strong, NICE).
Tobacco
Pulmonary Rehabilitation. Recommendation 17. Provide rehabilitation to patients with high exacerbation risk and relevant symptoms (A, GOLD). Offer pulmonary rehabilitation to patients with recent hospitalizations and/or severe dyspnea (Strong, NICE).
Immunizations. Recommendation 18. Influenza and pneumococcal vaccinations (PPSV23 as well as PCV13 when age ≥ 65 years) are recommended for patients with COPD (NS, GOLD; Strong, NICE).
Palliative Care. Recommendation 19. For patients with end-stage COPD or poorly controlled symptoms, provide access to palliative care (NS, GOLD; Strong, NICE).
Management of COPD Exacerbations and Patients at high risk for Exacerbations
Recommendation 20. Use SABAs with or without SAMAs as initial bronchodilators to treat acute exacerbations (C, GOLD). Recommendation 21. Systemic corticosteroids for exacerbations improve lung function, oxygenation, and recovery time. Recommend 5 to 7 days of therapy (A, GOLD; Strong, NICE). Recommendation 22. Antibiotics shorten recovery time and reduce treatment failure and rehospitalization. Treatment should be 5 to 7 days (B, GOLD). Consider antibiotics while balancing the severity of symptoms and hospitalization need (Conditional, NICE). Recommendation 23. Noninvasive mechanical ventilation is the preferred mode of ventilation for COPD patients with acute respiratory failure without acute contraindications (A, GOLD). Recommendation 24. Avoid long-term oral corticosteroids therapy (A, GOLD). Recommendation 25. Consider roflumilast for patients with exacerbations despite LABA/ICS or LABA/LAMA/ICS, and seek respiratory medicine consultation (B, GOLD; Strong, NICE). For former smokers with exacerbations despite appropriate therapy, consider azithromycin (B, GOLD; Strong, NICE).
CRITIQUE
GOLD is an International committee of experts who compile the report based on scientific literature review. NICE is an independent organization funded by Department of Health and Social Care in the United Kingdom responsible for evidence-based guidance on healthcare determined by an expert committee through scientific review and a transparent process that details committee formation and framework (GRADE) used and stakeholder input. While both guidelines review current publications, practice-influencing clinical trials of recent publication may be missed.
On the GOLD Science committee, 17/20 members have pharmaceutical relationships, with no mitigation plan provided. The NICE guidelines detail a panel with few industry ties and a mitigation plan for potential conflicts of interest.
These recommendations comprehensively cover outpatient and inpatient COPD management. The GOLD and NICE guidelines are similar with the exception of recommendations surrounding use of oxygen. The NICE guidelines, based on the adverse events documented in the recent Long-Term Oxygen Treatment Trial,6 recommend against oxygen use by patients who smoke because of the risk of fire-related injuries;7 GOLD guidelines do not differentiate oxygen recommendation by patient population.
Differences in the strength of NICE and GOLD recommendations highlight areas for further study. Investigations determining distinct COPD phenotypes will likely influence future guidelines. More discriminative multidimensional prognostication tools are needed to improve precision surrounding prognosis.
1. NICE. Overview. Chronic obstructive pulmonary disease in over 16s: Diagnosis and management, Guidance. https://www.nice.org.uk/guidance/ng115. Accessed November 21, 2019
2. GOLD Reports for Personal Use. Global Initiative for Chronic Obstructive Lung Disease - GOLD. https://goldcopd.org/gold-reports/. Accessed September 17, 2019.
3. Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2095-128. https://doi.org/10.1016/S0140-6736(12)61728-0.
4. Suissa S, Dell’Aniello S, Ernst P. Long-term natural history of chronic obstructive pulmonary disease: Severe exacerbations and mortality. Thorax. 2012;67(11):957-63. https://doi.org/10.1136/thoraxjnl-2011-201518.
5. Quanjer PH, Stanojevic S, Cole TJ, et al. Multi-ethnic reference values for spirometry for the 3–95-yr age range: The global lung function 2012 equations. Eur Respir J. 2012;40(6):1324-43. https://doi.org/10.1183/09031936.00080312.
6. Albert RK, Au DH, Blackford AL, et al. Long-term oxygen treatment trial research group. A randomized trial of long-term oxygen for COPD with moderate desaturation. N Engl J Med. 2016;375(17):1617-27. https://doi.org/10.1056/NEJMoa1604344.
7. National Institute for Health and Care Excellence. Chronic obstructive pulmonary disease in over 16s: diagnosis and management [B} Oxygen therapy in people with stable COPD. https://www.nice.org.uk/guidance/ng115/evidence/b-oxygen-therapy-in-people-with-stable-copd-pdf-6602768751. Accessed November 21, 2019.
Chronic obstructive pulmonary disease (COPD), projected to be the third leading cause of death by 2020, accounts for 6% of deaths globally.3 Hospitalization for COPD exacerbations is common and impacts patients’ disease trajectory, and mortality, with fewer than half of patients hospitalized for exacerbation surviving 5 years.4 Hospitalization provides an opportunity to optimize care. Due to recent practice-changing evidence, the National Institute for Health and Care Excellence (NICE) and the Global Initiative for Chronic Obstructive Lung Disease (GOLD) published updated guidelines.
KEY RECOMMENDATIONS
These are selected recommendations relevant to adult hospitalists. The GOLD guidelines grade recommendations by evidence strength from category A (randomized control trial data) to category D (expert consensus). The NICE guidelines relay strength of evidence through terminology referring to the presence or absence of a strong recommendation. Recommendations without evidence level specified are NS.
Diagnosis and Classification of COPD Severity
Recommendation 1. In patients with risk factors for and symptoms of COPD, spirometry is required to confirm the diagnosis, defined as a postbronchodilator FEV1/FVC ratio of <0.7 (NS, NICE, GOLD). The Global Lung Function Initiative (GLI) 2012 reference ranges5 are recommended (NS, NICE). Recommendation 2. Severity of airflow obstruction should be assessed according to reduction in the postbronchodilator FEV1 as: Stage I, Mild: FEV1 ≥80%; Stage II, Moderate: FEV1 = 50-79%; Stage III, Severe FEV1 = 30%-49%; Stage IV, FEV1<30% (NS, NICE, GOLD). Recommendation 3. Reversibility testing (aka bronchodilator response) does not indicate long-term response to therapy (NS, NICE, GOLD). Recommendation 4. The combined COPD assessment to classify patient symptoms and disease severity in one of four groups (A, B, C, or D) based on exacerbation history and daily symptom control (NS, GOLD). Use the Medical Research Council dyspnea scale to classify symptoms (strong, NICE).
Pharmacologic COPD Management
Recommendation 5. Short-acting inhaled bronchodilators such as short-acting beta2 agonists (SABAs) or short-acting muscarinic antagonists (SAMAs) improve FEV1 and symptoms. Combining SABA/SAMA is superior to monotherapy (A, GOLD). Recommendation 6. Long-acting bronchodilators, such as long-acting antimuscarinics (LAMAs) or long-acting beta2 agonists (LABAs), improve lung function and dyspnea and reduce exacerbations. Combination therapy (LABA/LAMA) is superior to using a single agent (LABA or LAMA) for improving FEV1 and reducing exacerbations (A, GOLD). Recommendation 7. Triple therapy of inhaled corticosteroid ICS/LAMA/LABA is more effective than the individual components in reducing exacerbations in the case of moderate to severe COPD (A, GOLD). Recommendation 8. Treatment with an ICS increases pneumonia risk (A, GOLD). Discuss these side effects (Strong, NICE). Recommendation 9. Use SABAs and SAMAs as initial treatment for patients with COPD (Strong, NICE). LABAs and LAMAs are preferred over short-acting agents except for patients with mild symptoms (A, GOLD). Recommendation 10. For symptomatic patients on long-acting monotherapy, escalate to combination LABA/LAMA, or if asthmatic features or elevated eosinophils (≥300 cells/µL) are present, combination LABA/ICS (A, GOLD; Strong, NICE). Recommendation 11. Assess and correct patient inhaler technique (NS, GOLD; Strong, NICE).
Nonpharmacologic COPD Management
Oxygen. Recommendation 12. Long-term oxygen supplementation increases survival in patients with resting arterial hypoxemia (PaO2<55 mm Hg) or hypoxemia (PaO2<60 mm Hg) with cor pulmonale (A, GOLD). Recommendation 13. In patients with moderate resting (89%-93%) or exercise-induced arterial desaturation (80%-90%), long-term oxygen does not improve outcomes (A, GOLD).6 Recommendation 14. Consider long-term oxygan after a risk assessment of fall and burn risk. Do not offer oxygen to those who continue to smoke (Strong, NICE).
Tobacco
Pulmonary Rehabilitation. Recommendation 17. Provide rehabilitation to patients with high exacerbation risk and relevant symptoms (A, GOLD). Offer pulmonary rehabilitation to patients with recent hospitalizations and/or severe dyspnea (Strong, NICE).
Immunizations. Recommendation 18. Influenza and pneumococcal vaccinations (PPSV23 as well as PCV13 when age ≥ 65 years) are recommended for patients with COPD (NS, GOLD; Strong, NICE).
Palliative Care. Recommendation 19. For patients with end-stage COPD or poorly controlled symptoms, provide access to palliative care (NS, GOLD; Strong, NICE).
Management of COPD Exacerbations and Patients at high risk for Exacerbations
Recommendation 20. Use SABAs with or without SAMAs as initial bronchodilators to treat acute exacerbations (C, GOLD). Recommendation 21. Systemic corticosteroids for exacerbations improve lung function, oxygenation, and recovery time. Recommend 5 to 7 days of therapy (A, GOLD; Strong, NICE). Recommendation 22. Antibiotics shorten recovery time and reduce treatment failure and rehospitalization. Treatment should be 5 to 7 days (B, GOLD). Consider antibiotics while balancing the severity of symptoms and hospitalization need (Conditional, NICE). Recommendation 23. Noninvasive mechanical ventilation is the preferred mode of ventilation for COPD patients with acute respiratory failure without acute contraindications (A, GOLD). Recommendation 24. Avoid long-term oral corticosteroids therapy (A, GOLD). Recommendation 25. Consider roflumilast for patients with exacerbations despite LABA/ICS or LABA/LAMA/ICS, and seek respiratory medicine consultation (B, GOLD; Strong, NICE). For former smokers with exacerbations despite appropriate therapy, consider azithromycin (B, GOLD; Strong, NICE).
CRITIQUE
GOLD is an International committee of experts who compile the report based on scientific literature review. NICE is an independent organization funded by Department of Health and Social Care in the United Kingdom responsible for evidence-based guidance on healthcare determined by an expert committee through scientific review and a transparent process that details committee formation and framework (GRADE) used and stakeholder input. While both guidelines review current publications, practice-influencing clinical trials of recent publication may be missed.
On the GOLD Science committee, 17/20 members have pharmaceutical relationships, with no mitigation plan provided. The NICE guidelines detail a panel with few industry ties and a mitigation plan for potential conflicts of interest.
These recommendations comprehensively cover outpatient and inpatient COPD management. The GOLD and NICE guidelines are similar with the exception of recommendations surrounding use of oxygen. The NICE guidelines, based on the adverse events documented in the recent Long-Term Oxygen Treatment Trial,6 recommend against oxygen use by patients who smoke because of the risk of fire-related injuries;7 GOLD guidelines do not differentiate oxygen recommendation by patient population.
Differences in the strength of NICE and GOLD recommendations highlight areas for further study. Investigations determining distinct COPD phenotypes will likely influence future guidelines. More discriminative multidimensional prognostication tools are needed to improve precision surrounding prognosis.
Chronic obstructive pulmonary disease (COPD), projected to be the third leading cause of death by 2020, accounts for 6% of deaths globally.3 Hospitalization for COPD exacerbations is common and impacts patients’ disease trajectory, and mortality, with fewer than half of patients hospitalized for exacerbation surviving 5 years.4 Hospitalization provides an opportunity to optimize care. Due to recent practice-changing evidence, the National Institute for Health and Care Excellence (NICE) and the Global Initiative for Chronic Obstructive Lung Disease (GOLD) published updated guidelines.
KEY RECOMMENDATIONS
These are selected recommendations relevant to adult hospitalists. The GOLD guidelines grade recommendations by evidence strength from category A (randomized control trial data) to category D (expert consensus). The NICE guidelines relay strength of evidence through terminology referring to the presence or absence of a strong recommendation. Recommendations without evidence level specified are NS.
Diagnosis and Classification of COPD Severity
Recommendation 1. In patients with risk factors for and symptoms of COPD, spirometry is required to confirm the diagnosis, defined as a postbronchodilator FEV1/FVC ratio of <0.7 (NS, NICE, GOLD). The Global Lung Function Initiative (GLI) 2012 reference ranges5 are recommended (NS, NICE). Recommendation 2. Severity of airflow obstruction should be assessed according to reduction in the postbronchodilator FEV1 as: Stage I, Mild: FEV1 ≥80%; Stage II, Moderate: FEV1 = 50-79%; Stage III, Severe FEV1 = 30%-49%; Stage IV, FEV1<30% (NS, NICE, GOLD). Recommendation 3. Reversibility testing (aka bronchodilator response) does not indicate long-term response to therapy (NS, NICE, GOLD). Recommendation 4. The combined COPD assessment to classify patient symptoms and disease severity in one of four groups (A, B, C, or D) based on exacerbation history and daily symptom control (NS, GOLD). Use the Medical Research Council dyspnea scale to classify symptoms (strong, NICE).
Pharmacologic COPD Management
Recommendation 5. Short-acting inhaled bronchodilators such as short-acting beta2 agonists (SABAs) or short-acting muscarinic antagonists (SAMAs) improve FEV1 and symptoms. Combining SABA/SAMA is superior to monotherapy (A, GOLD). Recommendation 6. Long-acting bronchodilators, such as long-acting antimuscarinics (LAMAs) or long-acting beta2 agonists (LABAs), improve lung function and dyspnea and reduce exacerbations. Combination therapy (LABA/LAMA) is superior to using a single agent (LABA or LAMA) for improving FEV1 and reducing exacerbations (A, GOLD). Recommendation 7. Triple therapy of inhaled corticosteroid ICS/LAMA/LABA is more effective than the individual components in reducing exacerbations in the case of moderate to severe COPD (A, GOLD). Recommendation 8. Treatment with an ICS increases pneumonia risk (A, GOLD). Discuss these side effects (Strong, NICE). Recommendation 9. Use SABAs and SAMAs as initial treatment for patients with COPD (Strong, NICE). LABAs and LAMAs are preferred over short-acting agents except for patients with mild symptoms (A, GOLD). Recommendation 10. For symptomatic patients on long-acting monotherapy, escalate to combination LABA/LAMA, or if asthmatic features or elevated eosinophils (≥300 cells/µL) are present, combination LABA/ICS (A, GOLD; Strong, NICE). Recommendation 11. Assess and correct patient inhaler technique (NS, GOLD; Strong, NICE).
Nonpharmacologic COPD Management
Oxygen. Recommendation 12. Long-term oxygen supplementation increases survival in patients with resting arterial hypoxemia (PaO2<55 mm Hg) or hypoxemia (PaO2<60 mm Hg) with cor pulmonale (A, GOLD). Recommendation 13. In patients with moderate resting (89%-93%) or exercise-induced arterial desaturation (80%-90%), long-term oxygen does not improve outcomes (A, GOLD).6 Recommendation 14. Consider long-term oxygan after a risk assessment of fall and burn risk. Do not offer oxygen to those who continue to smoke (Strong, NICE).
Tobacco
Pulmonary Rehabilitation. Recommendation 17. Provide rehabilitation to patients with high exacerbation risk and relevant symptoms (A, GOLD). Offer pulmonary rehabilitation to patients with recent hospitalizations and/or severe dyspnea (Strong, NICE).
Immunizations. Recommendation 18. Influenza and pneumococcal vaccinations (PPSV23 as well as PCV13 when age ≥ 65 years) are recommended for patients with COPD (NS, GOLD; Strong, NICE).
Palliative Care. Recommendation 19. For patients with end-stage COPD or poorly controlled symptoms, provide access to palliative care (NS, GOLD; Strong, NICE).
Management of COPD Exacerbations and Patients at high risk for Exacerbations
Recommendation 20. Use SABAs with or without SAMAs as initial bronchodilators to treat acute exacerbations (C, GOLD). Recommendation 21. Systemic corticosteroids for exacerbations improve lung function, oxygenation, and recovery time. Recommend 5 to 7 days of therapy (A, GOLD; Strong, NICE). Recommendation 22. Antibiotics shorten recovery time and reduce treatment failure and rehospitalization. Treatment should be 5 to 7 days (B, GOLD). Consider antibiotics while balancing the severity of symptoms and hospitalization need (Conditional, NICE). Recommendation 23. Noninvasive mechanical ventilation is the preferred mode of ventilation for COPD patients with acute respiratory failure without acute contraindications (A, GOLD). Recommendation 24. Avoid long-term oral corticosteroids therapy (A, GOLD). Recommendation 25. Consider roflumilast for patients with exacerbations despite LABA/ICS or LABA/LAMA/ICS, and seek respiratory medicine consultation (B, GOLD; Strong, NICE). For former smokers with exacerbations despite appropriate therapy, consider azithromycin (B, GOLD; Strong, NICE).
CRITIQUE
GOLD is an International committee of experts who compile the report based on scientific literature review. NICE is an independent organization funded by Department of Health and Social Care in the United Kingdom responsible for evidence-based guidance on healthcare determined by an expert committee through scientific review and a transparent process that details committee formation and framework (GRADE) used and stakeholder input. While both guidelines review current publications, practice-influencing clinical trials of recent publication may be missed.
On the GOLD Science committee, 17/20 members have pharmaceutical relationships, with no mitigation plan provided. The NICE guidelines detail a panel with few industry ties and a mitigation plan for potential conflicts of interest.
These recommendations comprehensively cover outpatient and inpatient COPD management. The GOLD and NICE guidelines are similar with the exception of recommendations surrounding use of oxygen. The NICE guidelines, based on the adverse events documented in the recent Long-Term Oxygen Treatment Trial,6 recommend against oxygen use by patients who smoke because of the risk of fire-related injuries;7 GOLD guidelines do not differentiate oxygen recommendation by patient population.
Differences in the strength of NICE and GOLD recommendations highlight areas for further study. Investigations determining distinct COPD phenotypes will likely influence future guidelines. More discriminative multidimensional prognostication tools are needed to improve precision surrounding prognosis.
1. NICE. Overview. Chronic obstructive pulmonary disease in over 16s: Diagnosis and management, Guidance. https://www.nice.org.uk/guidance/ng115. Accessed November 21, 2019
2. GOLD Reports for Personal Use. Global Initiative for Chronic Obstructive Lung Disease - GOLD. https://goldcopd.org/gold-reports/. Accessed September 17, 2019.
3. Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2095-128. https://doi.org/10.1016/S0140-6736(12)61728-0.
4. Suissa S, Dell’Aniello S, Ernst P. Long-term natural history of chronic obstructive pulmonary disease: Severe exacerbations and mortality. Thorax. 2012;67(11):957-63. https://doi.org/10.1136/thoraxjnl-2011-201518.
5. Quanjer PH, Stanojevic S, Cole TJ, et al. Multi-ethnic reference values for spirometry for the 3–95-yr age range: The global lung function 2012 equations. Eur Respir J. 2012;40(6):1324-43. https://doi.org/10.1183/09031936.00080312.
6. Albert RK, Au DH, Blackford AL, et al. Long-term oxygen treatment trial research group. A randomized trial of long-term oxygen for COPD with moderate desaturation. N Engl J Med. 2016;375(17):1617-27. https://doi.org/10.1056/NEJMoa1604344.
7. National Institute for Health and Care Excellence. Chronic obstructive pulmonary disease in over 16s: diagnosis and management [B} Oxygen therapy in people with stable COPD. https://www.nice.org.uk/guidance/ng115/evidence/b-oxygen-therapy-in-people-with-stable-copd-pdf-6602768751. Accessed November 21, 2019.
1. NICE. Overview. Chronic obstructive pulmonary disease in over 16s: Diagnosis and management, Guidance. https://www.nice.org.uk/guidance/ng115. Accessed November 21, 2019
2. GOLD Reports for Personal Use. Global Initiative for Chronic Obstructive Lung Disease - GOLD. https://goldcopd.org/gold-reports/. Accessed September 17, 2019.
3. Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2095-128. https://doi.org/10.1016/S0140-6736(12)61728-0.
4. Suissa S, Dell’Aniello S, Ernst P. Long-term natural history of chronic obstructive pulmonary disease: Severe exacerbations and mortality. Thorax. 2012;67(11):957-63. https://doi.org/10.1136/thoraxjnl-2011-201518.
5. Quanjer PH, Stanojevic S, Cole TJ, et al. Multi-ethnic reference values for spirometry for the 3–95-yr age range: The global lung function 2012 equations. Eur Respir J. 2012;40(6):1324-43. https://doi.org/10.1183/09031936.00080312.
6. Albert RK, Au DH, Blackford AL, et al. Long-term oxygen treatment trial research group. A randomized trial of long-term oxygen for COPD with moderate desaturation. N Engl J Med. 2016;375(17):1617-27. https://doi.org/10.1056/NEJMoa1604344.
7. National Institute for Health and Care Excellence. Chronic obstructive pulmonary disease in over 16s: diagnosis and management [B} Oxygen therapy in people with stable COPD. https://www.nice.org.uk/guidance/ng115/evidence/b-oxygen-therapy-in-people-with-stable-copd-pdf-6602768751. Accessed November 21, 2019.
© 2020 Society of Hospital Medicine
Hahnemann’s Closure as a Lesson in Private Equity Healthcare
The recent closure of Hahnemann University Hospital, a 500-bed teaching hospital in downtown Philadelphia, Pennsylvania, offers a case study of a new form of for-profit business involvement in academic medicine —private equity investment. Though the closure of this 171-year-old institution is the result of multiple factors affecting the hospital’s financial health over decades and may not have been avoidable, the hospital’s final years in the hands of a private equity firm led to a closure process that was chaotic, uncoordinated, and fundamentally not aligned with the needs of the patients and trainees that make up the core constituent
Tracing the hospital’s history, much of its financial troubles began over 20 years ago. In 1993, the Allegheny Health, Education, and Research Foundation (AHERF), a nonprofit Pittsburgh-based hospital and physician practice organization, acquired Hahnemann Medical College. Forming the MCP-Hahnemann Medical School, AHERF merged the institution with another acquisition, Medical College of Pennsylvania (MCP),1 formerly known as the Woman’s Medical College of Pennsylvania, one of the first American medical schools devoted to exclusively training female physicians.1,2 This was part of AHERF’s aggressive growth strategy at the time and resulted in the acquisition of 14 hospitals and more than 300 Philadelphia-area primary care physician practices by 1998. This caused about $1.3 billion of debt and over $1 million in losses per day, which led AHERF to file for bankruptcy that year,2 the country’s largest nonprofit healthcare bankruptcy at the time.1 That same year, Tenet Healthcare Corporation, a for-profit healthcare company, bought AHERF’s assets in the Philadelphia region from bankruptcy for $345 million, acquiring eight hospitals, as well as all of AHREF’s physician practices.2 Ultimately, Tenet sold or closed six of the acquired hospitals by 2007, leaving just Hahnemann and St. Christopher’s Hospital for Children,3 while Drexel University, a private, nonprofit university, came forward to salvage AHERF’s educational programs, creating the Drexel University College of Medicine.2 Under the ownership of Tenet, Hahnemann’s financial health declined as its patient population included a growing proportion of those utilizing Medicare, Medicaid, and charity care, which resulted in a negative operating profit margin annually for the final 14 years under Tenet.3,4 In this setting, American Academic Health System, LLC (AAHS) stepped in to purchase Hahnemann and St. Christopher’s from Tenet and, eventually, chose to close Hahnemann.4
That Hahnemann found itself in the hands of a private equity firm was not surprising. Such investment firms’ acquisitions of hospitals and physician practices have become increasingly more common, with the number of these types of deals increasing by 48% and reaching a value of $42.6 billion from 2010 to 2017.5 While for-profit hospitals have been shown to have higher mortality6 and lower patient satisfaction7 than nonprofit hospitals, the relatively new and growing trend of private equity investment in healthcare has not been rigorously evaluated. By nature, these firms use investor capital to acquire assets with the goal of increasing their value and selling them off at a profit after about 3-7 years.5 Thus, healthcare services provided by private equity–owned facilities are valued and supported based on their profitability. Low-profit services, such as primary care and psychiatry, are minimized while more profitable services, such as same-day surgery, are maximized.5 In addition, given that for-profit hospitals tend to invest less in charity care8 and population health9 as compared with nonprofit institutions, private equity–owned hospitals likely follow suit, in contrast to the humanistic values of academic medicine. Ultimately, Hahnemann’s decades-long financial troubles set the stage for a buyout by private equity investors. But this transaction was the death knell for this teaching hospital and eventually proved to be a disadvantage for the community it served.
Purchasing Hahnemann and St Christopher’s from Tenet in early 2018 for $170 million, AAHS—an affiliate of the private equity firm, Paladin Healthcare Capital, LLC, led by investment banker Joel Freedman—entered the Philadelphia healthcare market in partnership with Chicago-based healthcare real estate private equity firm, Harrison Street Real Estate Capital, LLC.4 Paladin had previously invested in smaller hospitals serving underserved communities,4 and as it began its venture with this large teaching hospital, Paladin’s president, Barry Wolfman, stated that the company’s goal was “to return [Hahnemann] to its rightful place in the landscape of healthcare.”3 However, given the real estate firm’s involvement in the deal and the permissive tier of zoning for Hahnemann’s real estate,10 there were suspicions that the purchase of the hospital was a means to acquire and develop the valuable Center City real estate rather than to serve the community.3
Within months of the hospital purchase, AAHS‘s Philadelphia venture proved difficult. Four CEOs came and went as time passed, with some holding their position for only a couple of months.11 About 175 of Hahnemann’s nurses, support staff, and managers were laid off in April of 2019, but the hospital finances did not improve significantly.12 As it became evident that AAHS planned to close the hospital, efforts were made to prevent the closure. Drexel University filed an unsuccessful lawsuit, claiming that it would be a violation of the academic agreement between the university and hospital.13 Once AAHS announced plans for hospital closure, the Pennsylvania Secretary of Health, Rachel Levine, MD, wrote to AAHS leadership ordering a “cease and desist” of any action toward hospital closure.12 Despite this, AAHS began cutting vital hospital services, including trauma and cardiothoracic surgery services, within days of the closure announcement.14 While there is a state law that a hospital cannot be closed with less than 90 days’ notice, AAHS filed for bankruptcy and shut down Hahnemann’s service to the community in about half that time.13 The hospital real estate was separated from the operating businesses and was excluded from the bankruptcy filing,10 which further cemented suspicions that the involved private equity firms looked to profit off the land once the hospital closed.
The immediate and long-term effects of the closure of Hahnemann University Hospital on healthcare and medical education in Philadelphia are yet to be rigorously measured and evaluated. However, the hasty closure of a large inner-city teaching hospital that served as a healthcare safety net for a largely underserved minority population with 50,000 ED visits per year4 is a dangerous disruption to a community. The way that the hospital was closed not only defied regulatory attempts at protecting the community but also defied the values of the healthcare workers working in the hospital. Because AAHS ceased payments to hospital vendors, medical supplies were low during the final weeks at Hahnemann, which didn’t even have enough cups on the wards to provide drinking water for patients.15 Nurses reported feeling shame as they used scissors to cut wash cloths in half to have enough to wash their patients.15 The teaching hospital’s humanistic and social capital was being liquidated quickly. Even after Hahnemann’s 570 graduate medical trainees endured the stressful and chaotic process of being displaced and fortunately taken in by other programs,16 AAHS attempted to auction off Hahnemann’s graduate medical education (GME) slots and their associated government funding to the highest bidder. While a US bankruptcy judge initially approved the sale of those GME slots to a consortium of academic institutions in the Philadelphia area,17 the Center for Medicare & Medicaid Services (CMS) has appealed that decision, which resulted in a current stay on the transaction.17 AAHS treating GME trainee positions as assets to be bought and sold is a dangerous precedent to set, especially since it attempts to bypass CMS’s existing regulated process for redistributing the slots.
While time will reveal the effects of the hospital closure, the most concerning element of this story is that the methods of a private equity firm in closing a large inner-city teaching hospital flouted attempts by regulatory agencies acting to preserve the hospital’s mission to the community. The governor of Pennsylvania, Tom Wolf (D), and mayor of Philadelphia, Jim Kenney (D), issued a joint statement chastising the actions of AAHS: “The situation at Hahnemann University Hospital, caused by CEO Joel Freedman and his team of venture capitalists, is an absolute disgrace and shows a greed-driven lack of care for the community.”18 This chaotic situation inspired Philadelphia Councilperson Helen Gym (D) to propose city legislation requiring 180 days’ notice of a hospital closure, bestowing a strong local means of protecting the city’s people from similar healthcare fiascos in the future.15
At its core, healthcare is a human-to-human interaction with the purpose of improving and maintaining the health and life of the patient. Adding to that the noble efforts in educating students and trainees to provide that public good, academic medicine is a virtuous endeavor. The new and growing phenomenon of private equity in healthcare prioritizes maximizing a return on investment, so the closure of Hahnemann University Hospital in Philadelphia highlights manifestations of the discordance of the missions of private equity and academic medicine and serves as “the canary in the coal mine,” warning teaching hospitals and communities that this disconnect necessitates regulatory policies to protect academic medicine’s service to the community while private equity investment continues to spread in healthcare.
1. Burling, S. Hahnemann University Hospital: 171 years of Philadelphia medical history. The Philadelphia Inquirer. https://www.inquirer.com/health/hahnemann-university-hospital-timeline-history-20190821.html. August 21, 2019. Accessed October 10, 2019.
2. Klasko S and Ekarius J. Collision course: The privatization of graduate medical education at one university. Acad Med. 2007;82(3):238-244. https://doi.org/10.1097/ACM.0b013e3180305fb1.
3. Brubaker H. Tenet will leave Philly, selling Hahnemann, St. Christopher’s to Paladin. The Philadelphia Inquirer. https://www.inquirer.com/philly/business/tenet-leaves-philly-selling-hahnemann-st-christophers-to-paladin-20170901.html. September 1, 2017. Accessed October 10, 2019.
4. Brubaker H. This California banker bet on turning around Philly’s Hahnemann Hospital. He’s running out of time. The Philadelphia Inquirer. https://www.inquirer.com/business/hahnemann-turnaround-closure-california-banker-joel-freedman-20190408.html. April 8, 2019. Accessed October 10, 2019.
5. Gondi S and Song Z. Potential implications of private equity investments in health care delivery. JAMA. 2019;321(11):1047-1048. https://doi.org/10.1001/jama.2019.1077.
6. Devereaux PJ, Choi PT, Lacchetti C, et al. A systematic review and meta-analysis of studies comparing mortality rates of private for-profit and private not-for-profit hospitals. CMAJ. 2002;166(11):1399-1406.
7. Mazurenko O, Collum T, Ferdinand A, and Menachemi N. Predictors of hospital patient satisfaction as measured by HCAHPS: A systematic review. J of Healthc Manag. 2017;62(4):272-283. https://doi.org/10.1097/JHM-D-15-00050.
8. Valdovinos E, Le S, Hsia RY. In California, not-for-profit hospitals spent more operating expenses on charity care than for-profit hospitals spent. Health Affairs. 2015;34(8):1296-1303. https://doi.org/10.1377/hlthaff.2014.1208.
9. Gabriel MH, Atkins D, Liu X, Tregerman R. Examining the relationship between hospital ownership and population health efforts. J Health Organ Manag. 2018 Nov 19;32(8):934-942. https://doi.org/10.1108/JHOM-02-2018-0042.
10. Feldman N. Hospital union wants city to rezone Hahnemann property so it can’t be flipped. WHYY.org. https://whyy.org/articles/hospital-union-wants-city-to-rezone-hahnemann-property-so-it-cant-be-flipped/. August 2, 2019. Accessed October 10, 2019.
11. Brubaker H. New CEO fired at Hahnemann and St. Christopher’s Hospital for Children, two months into the job. The Philadelphia Inquirer. https://www.inquirer.com/business/hahnemann-st-christophers-hospital-ceo-turnover-20190308.html. March 8, 2019. Accessed October 10, 2019.
12. Rush M. Hahnemann University Hospital’s inner turmoil: A timeline of changes, layoffs, and closing. The Philadelphia Inquirer. https://www.inquirer.com/business/health/hahnemann-university-hospital-closing-timeline-20190626.html. July 1, 2019. Accessed October 10, 2019.
13. Brubaker H. Drexel sues to block threatened closure of Hahnemann University Hospital. The Philadelphia Inquirer. https://www.inquirer.com/business/hahnemann-hospital-drexel-freedman-closure-20190624.html. June 24, 2019. Accessed October 10, 2019.
14. Fernandez B, Dunn C. Hahnemann officially closes emergency room to critically ill. Nurses’ union says the hospital lacks basic supplies. The Philadelphia Inquirer. https://www.inquirer.com/news/hahnemann-hospital-emergency-room-closing-turmoil-20190629.html. June 29, 2019. Accessed October 10, 2019.
15. Bate D. Bill to prevent sudden hospital closures (like Hahnemann) moves along in City Council. WHYY.org. https://whyy.org/articles/bill-to-prevent-sudden-hospital-closures-like-hahnemann-moves-along-in-city-council/. November 20, 2019. Accessed October 10, 2019.
16. Aizenberg DJ and Logio LS. The Graduate Medical Education (GME) gold rush: GME slots and funding as a financial asset. Acad Med. 2019. https://doi.org/10.1097/ACM.0000000000003133.
17. Feldman N. Judge puts freeze on sale of Hahnemann residency program – for now. WHYY.org. https://whyy.org/articles/judge-puts-freeze-on-sale-of-hahnemann-residency-program-for-now/. September 16, 2019. Accessed October 11, 2019.
18. Pennsylvania Governor’s Office Press Release: Governor Wolf, Mayor Kenney Joint Statement on Hahnemann University Hospital. https://www.governor.pa.gov/newsroom/governor-wolf-mayor-kenney-joint-statement-on-hahnemann-university-hospital. July 11, 2019. Accessed October 18, 2019.
The recent closure of Hahnemann University Hospital, a 500-bed teaching hospital in downtown Philadelphia, Pennsylvania, offers a case study of a new form of for-profit business involvement in academic medicine —private equity investment. Though the closure of this 171-year-old institution is the result of multiple factors affecting the hospital’s financial health over decades and may not have been avoidable, the hospital’s final years in the hands of a private equity firm led to a closure process that was chaotic, uncoordinated, and fundamentally not aligned with the needs of the patients and trainees that make up the core constituent
Tracing the hospital’s history, much of its financial troubles began over 20 years ago. In 1993, the Allegheny Health, Education, and Research Foundation (AHERF), a nonprofit Pittsburgh-based hospital and physician practice organization, acquired Hahnemann Medical College. Forming the MCP-Hahnemann Medical School, AHERF merged the institution with another acquisition, Medical College of Pennsylvania (MCP),1 formerly known as the Woman’s Medical College of Pennsylvania, one of the first American medical schools devoted to exclusively training female physicians.1,2 This was part of AHERF’s aggressive growth strategy at the time and resulted in the acquisition of 14 hospitals and more than 300 Philadelphia-area primary care physician practices by 1998. This caused about $1.3 billion of debt and over $1 million in losses per day, which led AHERF to file for bankruptcy that year,2 the country’s largest nonprofit healthcare bankruptcy at the time.1 That same year, Tenet Healthcare Corporation, a for-profit healthcare company, bought AHERF’s assets in the Philadelphia region from bankruptcy for $345 million, acquiring eight hospitals, as well as all of AHREF’s physician practices.2 Ultimately, Tenet sold or closed six of the acquired hospitals by 2007, leaving just Hahnemann and St. Christopher’s Hospital for Children,3 while Drexel University, a private, nonprofit university, came forward to salvage AHERF’s educational programs, creating the Drexel University College of Medicine.2 Under the ownership of Tenet, Hahnemann’s financial health declined as its patient population included a growing proportion of those utilizing Medicare, Medicaid, and charity care, which resulted in a negative operating profit margin annually for the final 14 years under Tenet.3,4 In this setting, American Academic Health System, LLC (AAHS) stepped in to purchase Hahnemann and St. Christopher’s from Tenet and, eventually, chose to close Hahnemann.4
That Hahnemann found itself in the hands of a private equity firm was not surprising. Such investment firms’ acquisitions of hospitals and physician practices have become increasingly more common, with the number of these types of deals increasing by 48% and reaching a value of $42.6 billion from 2010 to 2017.5 While for-profit hospitals have been shown to have higher mortality6 and lower patient satisfaction7 than nonprofit hospitals, the relatively new and growing trend of private equity investment in healthcare has not been rigorously evaluated. By nature, these firms use investor capital to acquire assets with the goal of increasing their value and selling them off at a profit after about 3-7 years.5 Thus, healthcare services provided by private equity–owned facilities are valued and supported based on their profitability. Low-profit services, such as primary care and psychiatry, are minimized while more profitable services, such as same-day surgery, are maximized.5 In addition, given that for-profit hospitals tend to invest less in charity care8 and population health9 as compared with nonprofit institutions, private equity–owned hospitals likely follow suit, in contrast to the humanistic values of academic medicine. Ultimately, Hahnemann’s decades-long financial troubles set the stage for a buyout by private equity investors. But this transaction was the death knell for this teaching hospital and eventually proved to be a disadvantage for the community it served.
Purchasing Hahnemann and St Christopher’s from Tenet in early 2018 for $170 million, AAHS—an affiliate of the private equity firm, Paladin Healthcare Capital, LLC, led by investment banker Joel Freedman—entered the Philadelphia healthcare market in partnership with Chicago-based healthcare real estate private equity firm, Harrison Street Real Estate Capital, LLC.4 Paladin had previously invested in smaller hospitals serving underserved communities,4 and as it began its venture with this large teaching hospital, Paladin’s president, Barry Wolfman, stated that the company’s goal was “to return [Hahnemann] to its rightful place in the landscape of healthcare.”3 However, given the real estate firm’s involvement in the deal and the permissive tier of zoning for Hahnemann’s real estate,10 there were suspicions that the purchase of the hospital was a means to acquire and develop the valuable Center City real estate rather than to serve the community.3
Within months of the hospital purchase, AAHS‘s Philadelphia venture proved difficult. Four CEOs came and went as time passed, with some holding their position for only a couple of months.11 About 175 of Hahnemann’s nurses, support staff, and managers were laid off in April of 2019, but the hospital finances did not improve significantly.12 As it became evident that AAHS planned to close the hospital, efforts were made to prevent the closure. Drexel University filed an unsuccessful lawsuit, claiming that it would be a violation of the academic agreement between the university and hospital.13 Once AAHS announced plans for hospital closure, the Pennsylvania Secretary of Health, Rachel Levine, MD, wrote to AAHS leadership ordering a “cease and desist” of any action toward hospital closure.12 Despite this, AAHS began cutting vital hospital services, including trauma and cardiothoracic surgery services, within days of the closure announcement.14 While there is a state law that a hospital cannot be closed with less than 90 days’ notice, AAHS filed for bankruptcy and shut down Hahnemann’s service to the community in about half that time.13 The hospital real estate was separated from the operating businesses and was excluded from the bankruptcy filing,10 which further cemented suspicions that the involved private equity firms looked to profit off the land once the hospital closed.
The immediate and long-term effects of the closure of Hahnemann University Hospital on healthcare and medical education in Philadelphia are yet to be rigorously measured and evaluated. However, the hasty closure of a large inner-city teaching hospital that served as a healthcare safety net for a largely underserved minority population with 50,000 ED visits per year4 is a dangerous disruption to a community. The way that the hospital was closed not only defied regulatory attempts at protecting the community but also defied the values of the healthcare workers working in the hospital. Because AAHS ceased payments to hospital vendors, medical supplies were low during the final weeks at Hahnemann, which didn’t even have enough cups on the wards to provide drinking water for patients.15 Nurses reported feeling shame as they used scissors to cut wash cloths in half to have enough to wash their patients.15 The teaching hospital’s humanistic and social capital was being liquidated quickly. Even after Hahnemann’s 570 graduate medical trainees endured the stressful and chaotic process of being displaced and fortunately taken in by other programs,16 AAHS attempted to auction off Hahnemann’s graduate medical education (GME) slots and their associated government funding to the highest bidder. While a US bankruptcy judge initially approved the sale of those GME slots to a consortium of academic institutions in the Philadelphia area,17 the Center for Medicare & Medicaid Services (CMS) has appealed that decision, which resulted in a current stay on the transaction.17 AAHS treating GME trainee positions as assets to be bought and sold is a dangerous precedent to set, especially since it attempts to bypass CMS’s existing regulated process for redistributing the slots.
While time will reveal the effects of the hospital closure, the most concerning element of this story is that the methods of a private equity firm in closing a large inner-city teaching hospital flouted attempts by regulatory agencies acting to preserve the hospital’s mission to the community. The governor of Pennsylvania, Tom Wolf (D), and mayor of Philadelphia, Jim Kenney (D), issued a joint statement chastising the actions of AAHS: “The situation at Hahnemann University Hospital, caused by CEO Joel Freedman and his team of venture capitalists, is an absolute disgrace and shows a greed-driven lack of care for the community.”18 This chaotic situation inspired Philadelphia Councilperson Helen Gym (D) to propose city legislation requiring 180 days’ notice of a hospital closure, bestowing a strong local means of protecting the city’s people from similar healthcare fiascos in the future.15
At its core, healthcare is a human-to-human interaction with the purpose of improving and maintaining the health and life of the patient. Adding to that the noble efforts in educating students and trainees to provide that public good, academic medicine is a virtuous endeavor. The new and growing phenomenon of private equity in healthcare prioritizes maximizing a return on investment, so the closure of Hahnemann University Hospital in Philadelphia highlights manifestations of the discordance of the missions of private equity and academic medicine and serves as “the canary in the coal mine,” warning teaching hospitals and communities that this disconnect necessitates regulatory policies to protect academic medicine’s service to the community while private equity investment continues to spread in healthcare.
The recent closure of Hahnemann University Hospital, a 500-bed teaching hospital in downtown Philadelphia, Pennsylvania, offers a case study of a new form of for-profit business involvement in academic medicine —private equity investment. Though the closure of this 171-year-old institution is the result of multiple factors affecting the hospital’s financial health over decades and may not have been avoidable, the hospital’s final years in the hands of a private equity firm led to a closure process that was chaotic, uncoordinated, and fundamentally not aligned with the needs of the patients and trainees that make up the core constituent
Tracing the hospital’s history, much of its financial troubles began over 20 years ago. In 1993, the Allegheny Health, Education, and Research Foundation (AHERF), a nonprofit Pittsburgh-based hospital and physician practice organization, acquired Hahnemann Medical College. Forming the MCP-Hahnemann Medical School, AHERF merged the institution with another acquisition, Medical College of Pennsylvania (MCP),1 formerly known as the Woman’s Medical College of Pennsylvania, one of the first American medical schools devoted to exclusively training female physicians.1,2 This was part of AHERF’s aggressive growth strategy at the time and resulted in the acquisition of 14 hospitals and more than 300 Philadelphia-area primary care physician practices by 1998. This caused about $1.3 billion of debt and over $1 million in losses per day, which led AHERF to file for bankruptcy that year,2 the country’s largest nonprofit healthcare bankruptcy at the time.1 That same year, Tenet Healthcare Corporation, a for-profit healthcare company, bought AHERF’s assets in the Philadelphia region from bankruptcy for $345 million, acquiring eight hospitals, as well as all of AHREF’s physician practices.2 Ultimately, Tenet sold or closed six of the acquired hospitals by 2007, leaving just Hahnemann and St. Christopher’s Hospital for Children,3 while Drexel University, a private, nonprofit university, came forward to salvage AHERF’s educational programs, creating the Drexel University College of Medicine.2 Under the ownership of Tenet, Hahnemann’s financial health declined as its patient population included a growing proportion of those utilizing Medicare, Medicaid, and charity care, which resulted in a negative operating profit margin annually for the final 14 years under Tenet.3,4 In this setting, American Academic Health System, LLC (AAHS) stepped in to purchase Hahnemann and St. Christopher’s from Tenet and, eventually, chose to close Hahnemann.4
That Hahnemann found itself in the hands of a private equity firm was not surprising. Such investment firms’ acquisitions of hospitals and physician practices have become increasingly more common, with the number of these types of deals increasing by 48% and reaching a value of $42.6 billion from 2010 to 2017.5 While for-profit hospitals have been shown to have higher mortality6 and lower patient satisfaction7 than nonprofit hospitals, the relatively new and growing trend of private equity investment in healthcare has not been rigorously evaluated. By nature, these firms use investor capital to acquire assets with the goal of increasing their value and selling them off at a profit after about 3-7 years.5 Thus, healthcare services provided by private equity–owned facilities are valued and supported based on their profitability. Low-profit services, such as primary care and psychiatry, are minimized while more profitable services, such as same-day surgery, are maximized.5 In addition, given that for-profit hospitals tend to invest less in charity care8 and population health9 as compared with nonprofit institutions, private equity–owned hospitals likely follow suit, in contrast to the humanistic values of academic medicine. Ultimately, Hahnemann’s decades-long financial troubles set the stage for a buyout by private equity investors. But this transaction was the death knell for this teaching hospital and eventually proved to be a disadvantage for the community it served.
Purchasing Hahnemann and St Christopher’s from Tenet in early 2018 for $170 million, AAHS—an affiliate of the private equity firm, Paladin Healthcare Capital, LLC, led by investment banker Joel Freedman—entered the Philadelphia healthcare market in partnership with Chicago-based healthcare real estate private equity firm, Harrison Street Real Estate Capital, LLC.4 Paladin had previously invested in smaller hospitals serving underserved communities,4 and as it began its venture with this large teaching hospital, Paladin’s president, Barry Wolfman, stated that the company’s goal was “to return [Hahnemann] to its rightful place in the landscape of healthcare.”3 However, given the real estate firm’s involvement in the deal and the permissive tier of zoning for Hahnemann’s real estate,10 there were suspicions that the purchase of the hospital was a means to acquire and develop the valuable Center City real estate rather than to serve the community.3
Within months of the hospital purchase, AAHS‘s Philadelphia venture proved difficult. Four CEOs came and went as time passed, with some holding their position for only a couple of months.11 About 175 of Hahnemann’s nurses, support staff, and managers were laid off in April of 2019, but the hospital finances did not improve significantly.12 As it became evident that AAHS planned to close the hospital, efforts were made to prevent the closure. Drexel University filed an unsuccessful lawsuit, claiming that it would be a violation of the academic agreement between the university and hospital.13 Once AAHS announced plans for hospital closure, the Pennsylvania Secretary of Health, Rachel Levine, MD, wrote to AAHS leadership ordering a “cease and desist” of any action toward hospital closure.12 Despite this, AAHS began cutting vital hospital services, including trauma and cardiothoracic surgery services, within days of the closure announcement.14 While there is a state law that a hospital cannot be closed with less than 90 days’ notice, AAHS filed for bankruptcy and shut down Hahnemann’s service to the community in about half that time.13 The hospital real estate was separated from the operating businesses and was excluded from the bankruptcy filing,10 which further cemented suspicions that the involved private equity firms looked to profit off the land once the hospital closed.
The immediate and long-term effects of the closure of Hahnemann University Hospital on healthcare and medical education in Philadelphia are yet to be rigorously measured and evaluated. However, the hasty closure of a large inner-city teaching hospital that served as a healthcare safety net for a largely underserved minority population with 50,000 ED visits per year4 is a dangerous disruption to a community. The way that the hospital was closed not only defied regulatory attempts at protecting the community but also defied the values of the healthcare workers working in the hospital. Because AAHS ceased payments to hospital vendors, medical supplies were low during the final weeks at Hahnemann, which didn’t even have enough cups on the wards to provide drinking water for patients.15 Nurses reported feeling shame as they used scissors to cut wash cloths in half to have enough to wash their patients.15 The teaching hospital’s humanistic and social capital was being liquidated quickly. Even after Hahnemann’s 570 graduate medical trainees endured the stressful and chaotic process of being displaced and fortunately taken in by other programs,16 AAHS attempted to auction off Hahnemann’s graduate medical education (GME) slots and their associated government funding to the highest bidder. While a US bankruptcy judge initially approved the sale of those GME slots to a consortium of academic institutions in the Philadelphia area,17 the Center for Medicare & Medicaid Services (CMS) has appealed that decision, which resulted in a current stay on the transaction.17 AAHS treating GME trainee positions as assets to be bought and sold is a dangerous precedent to set, especially since it attempts to bypass CMS’s existing regulated process for redistributing the slots.
While time will reveal the effects of the hospital closure, the most concerning element of this story is that the methods of a private equity firm in closing a large inner-city teaching hospital flouted attempts by regulatory agencies acting to preserve the hospital’s mission to the community. The governor of Pennsylvania, Tom Wolf (D), and mayor of Philadelphia, Jim Kenney (D), issued a joint statement chastising the actions of AAHS: “The situation at Hahnemann University Hospital, caused by CEO Joel Freedman and his team of venture capitalists, is an absolute disgrace and shows a greed-driven lack of care for the community.”18 This chaotic situation inspired Philadelphia Councilperson Helen Gym (D) to propose city legislation requiring 180 days’ notice of a hospital closure, bestowing a strong local means of protecting the city’s people from similar healthcare fiascos in the future.15
At its core, healthcare is a human-to-human interaction with the purpose of improving and maintaining the health and life of the patient. Adding to that the noble efforts in educating students and trainees to provide that public good, academic medicine is a virtuous endeavor. The new and growing phenomenon of private equity in healthcare prioritizes maximizing a return on investment, so the closure of Hahnemann University Hospital in Philadelphia highlights manifestations of the discordance of the missions of private equity and academic medicine and serves as “the canary in the coal mine,” warning teaching hospitals and communities that this disconnect necessitates regulatory policies to protect academic medicine’s service to the community while private equity investment continues to spread in healthcare.
1. Burling, S. Hahnemann University Hospital: 171 years of Philadelphia medical history. The Philadelphia Inquirer. https://www.inquirer.com/health/hahnemann-university-hospital-timeline-history-20190821.html. August 21, 2019. Accessed October 10, 2019.
2. Klasko S and Ekarius J. Collision course: The privatization of graduate medical education at one university. Acad Med. 2007;82(3):238-244. https://doi.org/10.1097/ACM.0b013e3180305fb1.
3. Brubaker H. Tenet will leave Philly, selling Hahnemann, St. Christopher’s to Paladin. The Philadelphia Inquirer. https://www.inquirer.com/philly/business/tenet-leaves-philly-selling-hahnemann-st-christophers-to-paladin-20170901.html. September 1, 2017. Accessed October 10, 2019.
4. Brubaker H. This California banker bet on turning around Philly’s Hahnemann Hospital. He’s running out of time. The Philadelphia Inquirer. https://www.inquirer.com/business/hahnemann-turnaround-closure-california-banker-joel-freedman-20190408.html. April 8, 2019. Accessed October 10, 2019.
5. Gondi S and Song Z. Potential implications of private equity investments in health care delivery. JAMA. 2019;321(11):1047-1048. https://doi.org/10.1001/jama.2019.1077.
6. Devereaux PJ, Choi PT, Lacchetti C, et al. A systematic review and meta-analysis of studies comparing mortality rates of private for-profit and private not-for-profit hospitals. CMAJ. 2002;166(11):1399-1406.
7. Mazurenko O, Collum T, Ferdinand A, and Menachemi N. Predictors of hospital patient satisfaction as measured by HCAHPS: A systematic review. J of Healthc Manag. 2017;62(4):272-283. https://doi.org/10.1097/JHM-D-15-00050.
8. Valdovinos E, Le S, Hsia RY. In California, not-for-profit hospitals spent more operating expenses on charity care than for-profit hospitals spent. Health Affairs. 2015;34(8):1296-1303. https://doi.org/10.1377/hlthaff.2014.1208.
9. Gabriel MH, Atkins D, Liu X, Tregerman R. Examining the relationship between hospital ownership and population health efforts. J Health Organ Manag. 2018 Nov 19;32(8):934-942. https://doi.org/10.1108/JHOM-02-2018-0042.
10. Feldman N. Hospital union wants city to rezone Hahnemann property so it can’t be flipped. WHYY.org. https://whyy.org/articles/hospital-union-wants-city-to-rezone-hahnemann-property-so-it-cant-be-flipped/. August 2, 2019. Accessed October 10, 2019.
11. Brubaker H. New CEO fired at Hahnemann and St. Christopher’s Hospital for Children, two months into the job. The Philadelphia Inquirer. https://www.inquirer.com/business/hahnemann-st-christophers-hospital-ceo-turnover-20190308.html. March 8, 2019. Accessed October 10, 2019.
12. Rush M. Hahnemann University Hospital’s inner turmoil: A timeline of changes, layoffs, and closing. The Philadelphia Inquirer. https://www.inquirer.com/business/health/hahnemann-university-hospital-closing-timeline-20190626.html. July 1, 2019. Accessed October 10, 2019.
13. Brubaker H. Drexel sues to block threatened closure of Hahnemann University Hospital. The Philadelphia Inquirer. https://www.inquirer.com/business/hahnemann-hospital-drexel-freedman-closure-20190624.html. June 24, 2019. Accessed October 10, 2019.
14. Fernandez B, Dunn C. Hahnemann officially closes emergency room to critically ill. Nurses’ union says the hospital lacks basic supplies. The Philadelphia Inquirer. https://www.inquirer.com/news/hahnemann-hospital-emergency-room-closing-turmoil-20190629.html. June 29, 2019. Accessed October 10, 2019.
15. Bate D. Bill to prevent sudden hospital closures (like Hahnemann) moves along in City Council. WHYY.org. https://whyy.org/articles/bill-to-prevent-sudden-hospital-closures-like-hahnemann-moves-along-in-city-council/. November 20, 2019. Accessed October 10, 2019.
16. Aizenberg DJ and Logio LS. The Graduate Medical Education (GME) gold rush: GME slots and funding as a financial asset. Acad Med. 2019. https://doi.org/10.1097/ACM.0000000000003133.
17. Feldman N. Judge puts freeze on sale of Hahnemann residency program – for now. WHYY.org. https://whyy.org/articles/judge-puts-freeze-on-sale-of-hahnemann-residency-program-for-now/. September 16, 2019. Accessed October 11, 2019.
18. Pennsylvania Governor’s Office Press Release: Governor Wolf, Mayor Kenney Joint Statement on Hahnemann University Hospital. https://www.governor.pa.gov/newsroom/governor-wolf-mayor-kenney-joint-statement-on-hahnemann-university-hospital. July 11, 2019. Accessed October 18, 2019.
1. Burling, S. Hahnemann University Hospital: 171 years of Philadelphia medical history. The Philadelphia Inquirer. https://www.inquirer.com/health/hahnemann-university-hospital-timeline-history-20190821.html. August 21, 2019. Accessed October 10, 2019.
2. Klasko S and Ekarius J. Collision course: The privatization of graduate medical education at one university. Acad Med. 2007;82(3):238-244. https://doi.org/10.1097/ACM.0b013e3180305fb1.
3. Brubaker H. Tenet will leave Philly, selling Hahnemann, St. Christopher’s to Paladin. The Philadelphia Inquirer. https://www.inquirer.com/philly/business/tenet-leaves-philly-selling-hahnemann-st-christophers-to-paladin-20170901.html. September 1, 2017. Accessed October 10, 2019.
4. Brubaker H. This California banker bet on turning around Philly’s Hahnemann Hospital. He’s running out of time. The Philadelphia Inquirer. https://www.inquirer.com/business/hahnemann-turnaround-closure-california-banker-joel-freedman-20190408.html. April 8, 2019. Accessed October 10, 2019.
5. Gondi S and Song Z. Potential implications of private equity investments in health care delivery. JAMA. 2019;321(11):1047-1048. https://doi.org/10.1001/jama.2019.1077.
6. Devereaux PJ, Choi PT, Lacchetti C, et al. A systematic review and meta-analysis of studies comparing mortality rates of private for-profit and private not-for-profit hospitals. CMAJ. 2002;166(11):1399-1406.
7. Mazurenko O, Collum T, Ferdinand A, and Menachemi N. Predictors of hospital patient satisfaction as measured by HCAHPS: A systematic review. J of Healthc Manag. 2017;62(4):272-283. https://doi.org/10.1097/JHM-D-15-00050.
8. Valdovinos E, Le S, Hsia RY. In California, not-for-profit hospitals spent more operating expenses on charity care than for-profit hospitals spent. Health Affairs. 2015;34(8):1296-1303. https://doi.org/10.1377/hlthaff.2014.1208.
9. Gabriel MH, Atkins D, Liu X, Tregerman R. Examining the relationship between hospital ownership and population health efforts. J Health Organ Manag. 2018 Nov 19;32(8):934-942. https://doi.org/10.1108/JHOM-02-2018-0042.
10. Feldman N. Hospital union wants city to rezone Hahnemann property so it can’t be flipped. WHYY.org. https://whyy.org/articles/hospital-union-wants-city-to-rezone-hahnemann-property-so-it-cant-be-flipped/. August 2, 2019. Accessed October 10, 2019.
11. Brubaker H. New CEO fired at Hahnemann and St. Christopher’s Hospital for Children, two months into the job. The Philadelphia Inquirer. https://www.inquirer.com/business/hahnemann-st-christophers-hospital-ceo-turnover-20190308.html. March 8, 2019. Accessed October 10, 2019.
12. Rush M. Hahnemann University Hospital’s inner turmoil: A timeline of changes, layoffs, and closing. The Philadelphia Inquirer. https://www.inquirer.com/business/health/hahnemann-university-hospital-closing-timeline-20190626.html. July 1, 2019. Accessed October 10, 2019.
13. Brubaker H. Drexel sues to block threatened closure of Hahnemann University Hospital. The Philadelphia Inquirer. https://www.inquirer.com/business/hahnemann-hospital-drexel-freedman-closure-20190624.html. June 24, 2019. Accessed October 10, 2019.
14. Fernandez B, Dunn C. Hahnemann officially closes emergency room to critically ill. Nurses’ union says the hospital lacks basic supplies. The Philadelphia Inquirer. https://www.inquirer.com/news/hahnemann-hospital-emergency-room-closing-turmoil-20190629.html. June 29, 2019. Accessed October 10, 2019.
15. Bate D. Bill to prevent sudden hospital closures (like Hahnemann) moves along in City Council. WHYY.org. https://whyy.org/articles/bill-to-prevent-sudden-hospital-closures-like-hahnemann-moves-along-in-city-council/. November 20, 2019. Accessed October 10, 2019.
16. Aizenberg DJ and Logio LS. The Graduate Medical Education (GME) gold rush: GME slots and funding as a financial asset. Acad Med. 2019. https://doi.org/10.1097/ACM.0000000000003133.
17. Feldman N. Judge puts freeze on sale of Hahnemann residency program – for now. WHYY.org. https://whyy.org/articles/judge-puts-freeze-on-sale-of-hahnemann-residency-program-for-now/. September 16, 2019. Accessed October 11, 2019.
18. Pennsylvania Governor’s Office Press Release: Governor Wolf, Mayor Kenney Joint Statement on Hahnemann University Hospital. https://www.governor.pa.gov/newsroom/governor-wolf-mayor-kenney-joint-statement-on-hahnemann-university-hospital. July 11, 2019. Accessed October 18, 2019.
© 2020 Society of Hospital Medicine