User login
A closer look at migraine aura
Migraine aura sometimes accompanies or precedes migraine pain, but the phenomenon is difficult to treat and poorly understood. However, some evidence points to potential neurological mechanisms, and migraine aura is associated with cardiovascular disease risk.
Andrea Harriott, MD, PhD, said at the Stowe Headache Symposium sponsored by the Headache Cooperative of New England, which was conducted virtually. Dr. Harriott is assistant professor of neurology at Massachusetts General Hospital in Boston.
Somewhere between 20% and 40% of patients with migraine experience aura. It is most often visual, though it can also include sensory, aphasic, and motor symptoms. Visual aura usually begins as a flickering zigzag pattern in the central visual field that moves slowly toward the periphery and often leaves a scotoma. Typical duration is 15-30 minutes. Aura symptoms are more common in females.
Research in the 1940s conducted by the Brazilian researcher Aristides de Azevedo Pacheco Leão, PhD, then at Harvard Medical School, Boston, showed evidence of CSD in rabbits after electrical or mechanical stimulation. He observed a wave of vasodilation and increased blood flow over the cortex that spread over nearly the entire dorsolateral cortex within 3-6 minutes.
In the 1940s and 1950s, researchers sketched on paper the visual disturbance over 10 minutes, tracking the expanding spectrum across the visual field, from the center toward the periphery. The resulting scotoma advanced across the visual cortex at a rate very similar to that of the cortical spreading observed by Dr. Leão, “potentially linking this electrical event that was described with the aura event of migraine,” said Dr. Harriott. Those researchers hypothesized that the aura was produced by a strong excitation phase, followed by a wave of total inhibition.
More recent functional magnetic resonance imaging studies have also shown that CSD-like disturbances occur when patients experience migraine aura. In one study, researchers observed an initial increase and then a decrease in the blood oxygenation level dependent (BOLD) signal, which spread slowly across the visual cortex and correlated with the aura event. “This study was really important in confirming that a CSD-like phenomenon was likely the underlying perturbation that produced the visual aura of migraine,” said Dr. Harriott.
Despite the evidence that CSD causes migraine aura, its connection to migraine pain hasn’t been firmly established. But Dr. Harriott presented some evidence linking the two. Migraine aura is usually followed by pain, and aura precedes migraine attacks 78%-93% of the time. Cephalic allodynia occurs in migraine about 70% to 80% of the time, and migraine with aura is more often associated with severe cutaneous allodynia than is migraine without aura. Finally, migraine patients with comorbidities have more severe disability, and more frequent cutaneous allodynia and aura than does the general migraine population (40% vs. 29%).
All of that suggests that activation of trigeminal nociceptors is involved with migraine aura, according to Dr. Harriott. Preclinical studies have also suggested links between CSD and activation of trigeminal nociceptors, with both immunohistochemical and electrophysiological lines of evidence. “These data suggest that spreading depression actually activates trigeminal nociceptors that we know are involved in signal pain in the head and neck, and that we know are involved in cephalic allodynia as well,” Dr. Harriott said.
The evidence impressed Allan Purdy, MD, professor of medicine at Dalhousie University, Halifax, N.S., who was the discussant for the presentation. “It’s an excellent case that CSD is a remarkably good correlate for aura,” he said during the session.
Along with potential impacts on migraine pain, aura is also associated with cardiovascular risk. “This is really important to know about in our clinical population,” said Dr. Harriott.
Meta-analyses of case control and cohort studies have shown associations between migraine aura and vascular disorders such as ischemic stroke. One meta-analysis showed about a twofold increased risk associated with migraine compared with the nonmigraine population. This difference was driven by migraine with aura (relative risk [RR], 2.25; 95% confidence interval [CI], 1.53-3.33) rather than migraine without aura (RR, 1.24; 95% CI, 0.86-1.79). Migraine generally is associated with greater risk of myocardial infarction (adjusted hazard ratio, 1.33; 95% CI, 1.08-1.64), and that association may be stronger in the aura phenotype.
There doesn’t appear to be evidence that traditional risk factors for heart disease – such as hypertension, diabetes, or high cholesterol – play a role in the association between aura and heart disease. One possibility is that variables like platelet activation, hypercoagulable state, or genetic susceptibility could be responsible.
The risks associated with migraine aura should be noted, but with a caveat, according to Dr. Purdy. “Even though the relative risk is high, the absolute risk is still relatively low, and patients with migraine with aura, who smoke or are female and over 45, those are the cases where the worry comes in.”
Dr. Harriott and Dr. Purdy have nothing to disclose.
Migraine aura sometimes accompanies or precedes migraine pain, but the phenomenon is difficult to treat and poorly understood. However, some evidence points to potential neurological mechanisms, and migraine aura is associated with cardiovascular disease risk.
Andrea Harriott, MD, PhD, said at the Stowe Headache Symposium sponsored by the Headache Cooperative of New England, which was conducted virtually. Dr. Harriott is assistant professor of neurology at Massachusetts General Hospital in Boston.
Somewhere between 20% and 40% of patients with migraine experience aura. It is most often visual, though it can also include sensory, aphasic, and motor symptoms. Visual aura usually begins as a flickering zigzag pattern in the central visual field that moves slowly toward the periphery and often leaves a scotoma. Typical duration is 15-30 minutes. Aura symptoms are more common in females.
Research in the 1940s conducted by the Brazilian researcher Aristides de Azevedo Pacheco Leão, PhD, then at Harvard Medical School, Boston, showed evidence of CSD in rabbits after electrical or mechanical stimulation. He observed a wave of vasodilation and increased blood flow over the cortex that spread over nearly the entire dorsolateral cortex within 3-6 minutes.
In the 1940s and 1950s, researchers sketched on paper the visual disturbance over 10 minutes, tracking the expanding spectrum across the visual field, from the center toward the periphery. The resulting scotoma advanced across the visual cortex at a rate very similar to that of the cortical spreading observed by Dr. Leão, “potentially linking this electrical event that was described with the aura event of migraine,” said Dr. Harriott. Those researchers hypothesized that the aura was produced by a strong excitation phase, followed by a wave of total inhibition.
More recent functional magnetic resonance imaging studies have also shown that CSD-like disturbances occur when patients experience migraine aura. In one study, researchers observed an initial increase and then a decrease in the blood oxygenation level dependent (BOLD) signal, which spread slowly across the visual cortex and correlated with the aura event. “This study was really important in confirming that a CSD-like phenomenon was likely the underlying perturbation that produced the visual aura of migraine,” said Dr. Harriott.
Despite the evidence that CSD causes migraine aura, its connection to migraine pain hasn’t been firmly established. But Dr. Harriott presented some evidence linking the two. Migraine aura is usually followed by pain, and aura precedes migraine attacks 78%-93% of the time. Cephalic allodynia occurs in migraine about 70% to 80% of the time, and migraine with aura is more often associated with severe cutaneous allodynia than is migraine without aura. Finally, migraine patients with comorbidities have more severe disability, and more frequent cutaneous allodynia and aura than does the general migraine population (40% vs. 29%).
All of that suggests that activation of trigeminal nociceptors is involved with migraine aura, according to Dr. Harriott. Preclinical studies have also suggested links between CSD and activation of trigeminal nociceptors, with both immunohistochemical and electrophysiological lines of evidence. “These data suggest that spreading depression actually activates trigeminal nociceptors that we know are involved in signal pain in the head and neck, and that we know are involved in cephalic allodynia as well,” Dr. Harriott said.
The evidence impressed Allan Purdy, MD, professor of medicine at Dalhousie University, Halifax, N.S., who was the discussant for the presentation. “It’s an excellent case that CSD is a remarkably good correlate for aura,” he said during the session.
Along with potential impacts on migraine pain, aura is also associated with cardiovascular risk. “This is really important to know about in our clinical population,” said Dr. Harriott.
Meta-analyses of case control and cohort studies have shown associations between migraine aura and vascular disorders such as ischemic stroke. One meta-analysis showed about a twofold increased risk associated with migraine compared with the nonmigraine population. This difference was driven by migraine with aura (relative risk [RR], 2.25; 95% confidence interval [CI], 1.53-3.33) rather than migraine without aura (RR, 1.24; 95% CI, 0.86-1.79). Migraine generally is associated with greater risk of myocardial infarction (adjusted hazard ratio, 1.33; 95% CI, 1.08-1.64), and that association may be stronger in the aura phenotype.
There doesn’t appear to be evidence that traditional risk factors for heart disease – such as hypertension, diabetes, or high cholesterol – play a role in the association between aura and heart disease. One possibility is that variables like platelet activation, hypercoagulable state, or genetic susceptibility could be responsible.
The risks associated with migraine aura should be noted, but with a caveat, according to Dr. Purdy. “Even though the relative risk is high, the absolute risk is still relatively low, and patients with migraine with aura, who smoke or are female and over 45, those are the cases where the worry comes in.”
Dr. Harriott and Dr. Purdy have nothing to disclose.
Migraine aura sometimes accompanies or precedes migraine pain, but the phenomenon is difficult to treat and poorly understood. However, some evidence points to potential neurological mechanisms, and migraine aura is associated with cardiovascular disease risk.
Andrea Harriott, MD, PhD, said at the Stowe Headache Symposium sponsored by the Headache Cooperative of New England, which was conducted virtually. Dr. Harriott is assistant professor of neurology at Massachusetts General Hospital in Boston.
Somewhere between 20% and 40% of patients with migraine experience aura. It is most often visual, though it can also include sensory, aphasic, and motor symptoms. Visual aura usually begins as a flickering zigzag pattern in the central visual field that moves slowly toward the periphery and often leaves a scotoma. Typical duration is 15-30 minutes. Aura symptoms are more common in females.
Research in the 1940s conducted by the Brazilian researcher Aristides de Azevedo Pacheco Leão, PhD, then at Harvard Medical School, Boston, showed evidence of CSD in rabbits after electrical or mechanical stimulation. He observed a wave of vasodilation and increased blood flow over the cortex that spread over nearly the entire dorsolateral cortex within 3-6 minutes.
In the 1940s and 1950s, researchers sketched on paper the visual disturbance over 10 minutes, tracking the expanding spectrum across the visual field, from the center toward the periphery. The resulting scotoma advanced across the visual cortex at a rate very similar to that of the cortical spreading observed by Dr. Leão, “potentially linking this electrical event that was described with the aura event of migraine,” said Dr. Harriott. Those researchers hypothesized that the aura was produced by a strong excitation phase, followed by a wave of total inhibition.
More recent functional magnetic resonance imaging studies have also shown that CSD-like disturbances occur when patients experience migraine aura. In one study, researchers observed an initial increase and then a decrease in the blood oxygenation level dependent (BOLD) signal, which spread slowly across the visual cortex and correlated with the aura event. “This study was really important in confirming that a CSD-like phenomenon was likely the underlying perturbation that produced the visual aura of migraine,” said Dr. Harriott.
Despite the evidence that CSD causes migraine aura, its connection to migraine pain hasn’t been firmly established. But Dr. Harriott presented some evidence linking the two. Migraine aura is usually followed by pain, and aura precedes migraine attacks 78%-93% of the time. Cephalic allodynia occurs in migraine about 70% to 80% of the time, and migraine with aura is more often associated with severe cutaneous allodynia than is migraine without aura. Finally, migraine patients with comorbidities have more severe disability, and more frequent cutaneous allodynia and aura than does the general migraine population (40% vs. 29%).
All of that suggests that activation of trigeminal nociceptors is involved with migraine aura, according to Dr. Harriott. Preclinical studies have also suggested links between CSD and activation of trigeminal nociceptors, with both immunohistochemical and electrophysiological lines of evidence. “These data suggest that spreading depression actually activates trigeminal nociceptors that we know are involved in signal pain in the head and neck, and that we know are involved in cephalic allodynia as well,” Dr. Harriott said.
The evidence impressed Allan Purdy, MD, professor of medicine at Dalhousie University, Halifax, N.S., who was the discussant for the presentation. “It’s an excellent case that CSD is a remarkably good correlate for aura,” he said during the session.
Along with potential impacts on migraine pain, aura is also associated with cardiovascular risk. “This is really important to know about in our clinical population,” said Dr. Harriott.
Meta-analyses of case control and cohort studies have shown associations between migraine aura and vascular disorders such as ischemic stroke. One meta-analysis showed about a twofold increased risk associated with migraine compared with the nonmigraine population. This difference was driven by migraine with aura (relative risk [RR], 2.25; 95% confidence interval [CI], 1.53-3.33) rather than migraine without aura (RR, 1.24; 95% CI, 0.86-1.79). Migraine generally is associated with greater risk of myocardial infarction (adjusted hazard ratio, 1.33; 95% CI, 1.08-1.64), and that association may be stronger in the aura phenotype.
There doesn’t appear to be evidence that traditional risk factors for heart disease – such as hypertension, diabetes, or high cholesterol – play a role in the association between aura and heart disease. One possibility is that variables like platelet activation, hypercoagulable state, or genetic susceptibility could be responsible.
The risks associated with migraine aura should be noted, but with a caveat, according to Dr. Purdy. “Even though the relative risk is high, the absolute risk is still relatively low, and patients with migraine with aura, who smoke or are female and over 45, those are the cases where the worry comes in.”
Dr. Harriott and Dr. Purdy have nothing to disclose.
FROM HCNE STOWE 2020
HIT-6 may help track meaningful change in chronic migraine
, recent research suggests.
Using data from the phase 3 PROMISE-2 study, which evaluated intravenous eptinezumab in doses of 100 mg or 300 mg, or placebo every 12 weeks in 1,072 participants for the prevention of chronic migraine, Carrie R. Houts, PhD, director of psychometrics at the Vector Psychometric Group, in Chapel Hill, N.C., and colleagues determined that their finding of 6-point improvement of HIT-6 total score was consistent with other studies. However, they pointed out that little research has been done in evaluating how item-specific scores of HIT-6 impact individuals with chronic migraine. HIT-6 item scores examine whether individuals with headaches experience severe pain, limit their daily activities, have a desire to lie down, feel too tired to do daily activities, felt “fed up or irritated” because of headaches, and feel their headaches limit concentration on work or daily activities.
“The item-specific responder definitions give clinicians and researchers the ability to evaluate and track the impact of headache on specific item-level areas of patients’ lives. These responder definitions provide practical and easily interpreted results that can be used to evaluate treatment benefits over time and to improve clinician-patients communication focus on improvements in key aspects of functioning in individuals with chronic migraine,” Dr. Houts and colleagues wrote in their study, published in the October issue of Headache.
The 6-point value and the 1-2 category improvement values in item-specific scores, they suggested, could be used as a benchmark to help other clinicians and researchers detect meaningful change in individual patients with chronic migraine. Although the user guide for HIT-6 highlights a 5-point change in the total score as clinically meaningful, the authors of the guide do not provide evidence for why the 5-point value signifies clinically meaningful change, they said.
Determining thresholds of clinically meaningful change
In their study, Dr. Houts and colleagues used distribution-based methods to gauge responder values for the HIT-6 total score, while item-specific HIT-6 analyses were measured with Patients’ Global Impression of Change (PGIC), reduction in migraine frequency through monthly migraine days (MMDs), and EuroQol 5 dimensions 5 levels visual analog scale (EQ-5D-5L VAS). The researchers also used HIT-6 values from a literature review and from analyses in PROMISE-2 to calculate “a final chronic migraine-specific responder definition value” between baseline and 12 weeks. Participants in the PROMISE-2 study were mostly women (88.2%) and white (91.0%) with a mean age of 40.5 years.
The literature search revealed responder thresholds for the HIT-6 total score in a range between a decrease of 3 points and 8 points. Within PROMISE-2, the HIT-6 total score responder threshold was found to be between –2.6 and –2.2, which the researchers rounded down to a decrease of 3 points. When taking both sets of responder thresholds into account, the researchers calculated the median responder value as –5.5, which was rounded down to a decrease in 6 points in the HIT-6 total score. “[The estimate] appears most appropriate for discriminating between individuals with chronic migraine who have experienced meaningful change over time and those who have not,” Dr. Houts and colleagues said.
For item-specific HIT-6 scores, the mean score changes were –1 points for categories involving severe pain, limiting activities, lying down, and –2 points for categories involving feeling tired, being fed up or irritated, and limiting concentration.
“Taken together, the current chronic migraine-specific results are consistent with values derived from general headache/migraine samples and suggest that a decrease of 6 points or more on the HIT-6 total score would be considered meaningful to chronic migraine patients,” Dr. Houts and colleagues said. “This would translate to approximately a 4-category change on a single item, change on 2 items of approximately 2 and 3 categories, or a 1-category change on 3 or 4 of the 6 items, depending on the initial category.”
The researchers cautioned that the values outlined in the study “should not be used to determine clinically meaningful difference between treatment groups” and that “future work, similar to that reported here, will identify a chronic migraine-specific clinically meaningful difference between treatment groups value.”
A better measure of chronic migraine?
In an interview, J. D. Bartleson Jr., MD, a retired neurologist with the Mayo Clinic in Rochester, Minn., questioned why HIT-6 criteria was used in the initial PROMISE-2 study. “There is not a lot of difference between the significant and insignificant categories. Chronic migraine may be better measured with pain severity and number of headache days per month,” he said.
,“It may be appropriate to use just 1 or 2 symptoms for evaluating a given patient’s headache burden,” in terms of clinical application of the study for neurologists, Dr. Bartleson said. He emphasized that more research is needed.
This study was funded by H. Lundbeck A/S, which also provided funding of medical writing and editorial support for the manuscript. Three authors report being employees of Vector Psychometric Group at the time of the study, and the company received funding from H. Lundbeck A/S for their time conducting study-related research. Three other authors report relationships with pharmaceutical companies, medical societies, government agencies, and industry related to the study in the form of consultancies, advisory board memberships, honoraria, research support, stock or stock options, and employment. Dr. Bartleson reports no relevant conflicts of interest.
, recent research suggests.
Using data from the phase 3 PROMISE-2 study, which evaluated intravenous eptinezumab in doses of 100 mg or 300 mg, or placebo every 12 weeks in 1,072 participants for the prevention of chronic migraine, Carrie R. Houts, PhD, director of psychometrics at the Vector Psychometric Group, in Chapel Hill, N.C., and colleagues determined that their finding of 6-point improvement of HIT-6 total score was consistent with other studies. However, they pointed out that little research has been done in evaluating how item-specific scores of HIT-6 impact individuals with chronic migraine. HIT-6 item scores examine whether individuals with headaches experience severe pain, limit their daily activities, have a desire to lie down, feel too tired to do daily activities, felt “fed up or irritated” because of headaches, and feel their headaches limit concentration on work or daily activities.
“The item-specific responder definitions give clinicians and researchers the ability to evaluate and track the impact of headache on specific item-level areas of patients’ lives. These responder definitions provide practical and easily interpreted results that can be used to evaluate treatment benefits over time and to improve clinician-patients communication focus on improvements in key aspects of functioning in individuals with chronic migraine,” Dr. Houts and colleagues wrote in their study, published in the October issue of Headache.
The 6-point value and the 1-2 category improvement values in item-specific scores, they suggested, could be used as a benchmark to help other clinicians and researchers detect meaningful change in individual patients with chronic migraine. Although the user guide for HIT-6 highlights a 5-point change in the total score as clinically meaningful, the authors of the guide do not provide evidence for why the 5-point value signifies clinically meaningful change, they said.
Determining thresholds of clinically meaningful change
In their study, Dr. Houts and colleagues used distribution-based methods to gauge responder values for the HIT-6 total score, while item-specific HIT-6 analyses were measured with Patients’ Global Impression of Change (PGIC), reduction in migraine frequency through monthly migraine days (MMDs), and EuroQol 5 dimensions 5 levels visual analog scale (EQ-5D-5L VAS). The researchers also used HIT-6 values from a literature review and from analyses in PROMISE-2 to calculate “a final chronic migraine-specific responder definition value” between baseline and 12 weeks. Participants in the PROMISE-2 study were mostly women (88.2%) and white (91.0%) with a mean age of 40.5 years.
The literature search revealed responder thresholds for the HIT-6 total score in a range between a decrease of 3 points and 8 points. Within PROMISE-2, the HIT-6 total score responder threshold was found to be between –2.6 and –2.2, which the researchers rounded down to a decrease of 3 points. When taking both sets of responder thresholds into account, the researchers calculated the median responder value as –5.5, which was rounded down to a decrease in 6 points in the HIT-6 total score. “[The estimate] appears most appropriate for discriminating between individuals with chronic migraine who have experienced meaningful change over time and those who have not,” Dr. Houts and colleagues said.
For item-specific HIT-6 scores, the mean score changes were –1 points for categories involving severe pain, limiting activities, lying down, and –2 points for categories involving feeling tired, being fed up or irritated, and limiting concentration.
“Taken together, the current chronic migraine-specific results are consistent with values derived from general headache/migraine samples and suggest that a decrease of 6 points or more on the HIT-6 total score would be considered meaningful to chronic migraine patients,” Dr. Houts and colleagues said. “This would translate to approximately a 4-category change on a single item, change on 2 items of approximately 2 and 3 categories, or a 1-category change on 3 or 4 of the 6 items, depending on the initial category.”
The researchers cautioned that the values outlined in the study “should not be used to determine clinically meaningful difference between treatment groups” and that “future work, similar to that reported here, will identify a chronic migraine-specific clinically meaningful difference between treatment groups value.”
A better measure of chronic migraine?
In an interview, J. D. Bartleson Jr., MD, a retired neurologist with the Mayo Clinic in Rochester, Minn., questioned why HIT-6 criteria was used in the initial PROMISE-2 study. “There is not a lot of difference between the significant and insignificant categories. Chronic migraine may be better measured with pain severity and number of headache days per month,” he said.
,“It may be appropriate to use just 1 or 2 symptoms for evaluating a given patient’s headache burden,” in terms of clinical application of the study for neurologists, Dr. Bartleson said. He emphasized that more research is needed.
This study was funded by H. Lundbeck A/S, which also provided funding of medical writing and editorial support for the manuscript. Three authors report being employees of Vector Psychometric Group at the time of the study, and the company received funding from H. Lundbeck A/S for their time conducting study-related research. Three other authors report relationships with pharmaceutical companies, medical societies, government agencies, and industry related to the study in the form of consultancies, advisory board memberships, honoraria, research support, stock or stock options, and employment. Dr. Bartleson reports no relevant conflicts of interest.
, recent research suggests.
Using data from the phase 3 PROMISE-2 study, which evaluated intravenous eptinezumab in doses of 100 mg or 300 mg, or placebo every 12 weeks in 1,072 participants for the prevention of chronic migraine, Carrie R. Houts, PhD, director of psychometrics at the Vector Psychometric Group, in Chapel Hill, N.C., and colleagues determined that their finding of 6-point improvement of HIT-6 total score was consistent with other studies. However, they pointed out that little research has been done in evaluating how item-specific scores of HIT-6 impact individuals with chronic migraine. HIT-6 item scores examine whether individuals with headaches experience severe pain, limit their daily activities, have a desire to lie down, feel too tired to do daily activities, felt “fed up or irritated” because of headaches, and feel their headaches limit concentration on work or daily activities.
“The item-specific responder definitions give clinicians and researchers the ability to evaluate and track the impact of headache on specific item-level areas of patients’ lives. These responder definitions provide practical and easily interpreted results that can be used to evaluate treatment benefits over time and to improve clinician-patients communication focus on improvements in key aspects of functioning in individuals with chronic migraine,” Dr. Houts and colleagues wrote in their study, published in the October issue of Headache.
The 6-point value and the 1-2 category improvement values in item-specific scores, they suggested, could be used as a benchmark to help other clinicians and researchers detect meaningful change in individual patients with chronic migraine. Although the user guide for HIT-6 highlights a 5-point change in the total score as clinically meaningful, the authors of the guide do not provide evidence for why the 5-point value signifies clinically meaningful change, they said.
Determining thresholds of clinically meaningful change
In their study, Dr. Houts and colleagues used distribution-based methods to gauge responder values for the HIT-6 total score, while item-specific HIT-6 analyses were measured with Patients’ Global Impression of Change (PGIC), reduction in migraine frequency through monthly migraine days (MMDs), and EuroQol 5 dimensions 5 levels visual analog scale (EQ-5D-5L VAS). The researchers also used HIT-6 values from a literature review and from analyses in PROMISE-2 to calculate “a final chronic migraine-specific responder definition value” between baseline and 12 weeks. Participants in the PROMISE-2 study were mostly women (88.2%) and white (91.0%) with a mean age of 40.5 years.
The literature search revealed responder thresholds for the HIT-6 total score in a range between a decrease of 3 points and 8 points. Within PROMISE-2, the HIT-6 total score responder threshold was found to be between –2.6 and –2.2, which the researchers rounded down to a decrease of 3 points. When taking both sets of responder thresholds into account, the researchers calculated the median responder value as –5.5, which was rounded down to a decrease in 6 points in the HIT-6 total score. “[The estimate] appears most appropriate for discriminating between individuals with chronic migraine who have experienced meaningful change over time and those who have not,” Dr. Houts and colleagues said.
For item-specific HIT-6 scores, the mean score changes were –1 points for categories involving severe pain, limiting activities, lying down, and –2 points for categories involving feeling tired, being fed up or irritated, and limiting concentration.
“Taken together, the current chronic migraine-specific results are consistent with values derived from general headache/migraine samples and suggest that a decrease of 6 points or more on the HIT-6 total score would be considered meaningful to chronic migraine patients,” Dr. Houts and colleagues said. “This would translate to approximately a 4-category change on a single item, change on 2 items of approximately 2 and 3 categories, or a 1-category change on 3 or 4 of the 6 items, depending on the initial category.”
The researchers cautioned that the values outlined in the study “should not be used to determine clinically meaningful difference between treatment groups” and that “future work, similar to that reported here, will identify a chronic migraine-specific clinically meaningful difference between treatment groups value.”
A better measure of chronic migraine?
In an interview, J. D. Bartleson Jr., MD, a retired neurologist with the Mayo Clinic in Rochester, Minn., questioned why HIT-6 criteria was used in the initial PROMISE-2 study. “There is not a lot of difference between the significant and insignificant categories. Chronic migraine may be better measured with pain severity and number of headache days per month,” he said.
,“It may be appropriate to use just 1 or 2 symptoms for evaluating a given patient’s headache burden,” in terms of clinical application of the study for neurologists, Dr. Bartleson said. He emphasized that more research is needed.
This study was funded by H. Lundbeck A/S, which also provided funding of medical writing and editorial support for the manuscript. Three authors report being employees of Vector Psychometric Group at the time of the study, and the company received funding from H. Lundbeck A/S for their time conducting study-related research. Three other authors report relationships with pharmaceutical companies, medical societies, government agencies, and industry related to the study in the form of consultancies, advisory board memberships, honoraria, research support, stock or stock options, and employment. Dr. Bartleson reports no relevant conflicts of interest.
FROM HEADACHE
Migraine nerve stimulation device now available over the counter
The Food and Drug Administration has cleared Cefaly Dual (Cefaly Technology) which was previously available only by prescription.
Most migraines involve the trigeminal nerve, which can be accessed through the skin on the forehead. Cefaly Dual stimulates the trigeminal nerve using a reusable self-adhesive electrode placed on the forehead.
The device has two settings, ACUTE and PREVENT. In the ACUTE setting, the individual wears the device for 60 minutes at headache onset or during a migraine attack. In the PREVENT setting, the individual wears the device for 20 minutes daily to help prevent future episodes.
At the start of a session, the wearer may feel a slight tingling sensation, which gradually increases and spreads throughout the forehead and the front part of the head. After about 14 minutes, the intensity stabilizes and remains constant until the treatment session is over, according to the company. The device automatically shuts off at the end of each session. It can be used as a stand-alone option or with existing treatment, the company noted.
“For millions of people across the U.S., living with migraine pain and coping with debilitating symptoms are daily realities. It is our mission to provide consumers with increased access to an effective and safe dual modality migraine treatment that is scientifically proven to reduce the number of monthly migraine days by almost half,” Jennifer Trainor McDermott, CEO of Cefaly Technology, said in a news release.
The FDA’s over-the-counter clearance of Cefaly Dual was based on several randomized, controlled clinical trials supporting the efficacy and safety of the device, the company said.
An earlier version of the Cefaly device was approved in the United States in March 2014 to help prevent migraine headache in adults aged 18 or older. The next-generation Cefaly Dual device is “small and sleek in comparison to its older model, which uses bands along the sides to create room for batteries. The newest device is palm-sized, more portable, and uses a battery that is rechargeable via USB,” the company said.
Last spring, the company announced a buyback program where customers in the United States may return their original device and receive a discount of the purchase of the Cefaly Dual device.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has cleared Cefaly Dual (Cefaly Technology) which was previously available only by prescription.
Most migraines involve the trigeminal nerve, which can be accessed through the skin on the forehead. Cefaly Dual stimulates the trigeminal nerve using a reusable self-adhesive electrode placed on the forehead.
The device has two settings, ACUTE and PREVENT. In the ACUTE setting, the individual wears the device for 60 minutes at headache onset or during a migraine attack. In the PREVENT setting, the individual wears the device for 20 minutes daily to help prevent future episodes.
At the start of a session, the wearer may feel a slight tingling sensation, which gradually increases and spreads throughout the forehead and the front part of the head. After about 14 minutes, the intensity stabilizes and remains constant until the treatment session is over, according to the company. The device automatically shuts off at the end of each session. It can be used as a stand-alone option or with existing treatment, the company noted.
“For millions of people across the U.S., living with migraine pain and coping with debilitating symptoms are daily realities. It is our mission to provide consumers with increased access to an effective and safe dual modality migraine treatment that is scientifically proven to reduce the number of monthly migraine days by almost half,” Jennifer Trainor McDermott, CEO of Cefaly Technology, said in a news release.
The FDA’s over-the-counter clearance of Cefaly Dual was based on several randomized, controlled clinical trials supporting the efficacy and safety of the device, the company said.
An earlier version of the Cefaly device was approved in the United States in March 2014 to help prevent migraine headache in adults aged 18 or older. The next-generation Cefaly Dual device is “small and sleek in comparison to its older model, which uses bands along the sides to create room for batteries. The newest device is palm-sized, more portable, and uses a battery that is rechargeable via USB,” the company said.
Last spring, the company announced a buyback program where customers in the United States may return their original device and receive a discount of the purchase of the Cefaly Dual device.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has cleared Cefaly Dual (Cefaly Technology) which was previously available only by prescription.
Most migraines involve the trigeminal nerve, which can be accessed through the skin on the forehead. Cefaly Dual stimulates the trigeminal nerve using a reusable self-adhesive electrode placed on the forehead.
The device has two settings, ACUTE and PREVENT. In the ACUTE setting, the individual wears the device for 60 minutes at headache onset or during a migraine attack. In the PREVENT setting, the individual wears the device for 20 minutes daily to help prevent future episodes.
At the start of a session, the wearer may feel a slight tingling sensation, which gradually increases and spreads throughout the forehead and the front part of the head. After about 14 minutes, the intensity stabilizes and remains constant until the treatment session is over, according to the company. The device automatically shuts off at the end of each session. It can be used as a stand-alone option or with existing treatment, the company noted.
“For millions of people across the U.S., living with migraine pain and coping with debilitating symptoms are daily realities. It is our mission to provide consumers with increased access to an effective and safe dual modality migraine treatment that is scientifically proven to reduce the number of monthly migraine days by almost half,” Jennifer Trainor McDermott, CEO of Cefaly Technology, said in a news release.
The FDA’s over-the-counter clearance of Cefaly Dual was based on several randomized, controlled clinical trials supporting the efficacy and safety of the device, the company said.
An earlier version of the Cefaly device was approved in the United States in March 2014 to help prevent migraine headache in adults aged 18 or older. The next-generation Cefaly Dual device is “small and sleek in comparison to its older model, which uses bands along the sides to create room for batteries. The newest device is palm-sized, more portable, and uses a battery that is rechargeable via USB,” the company said.
Last spring, the company announced a buyback program where customers in the United States may return their original device and receive a discount of the purchase of the Cefaly Dual device.
A version of this article originally appeared on Medscape.com.
Choroid plexuses may play a role in migraine
according to a new study. The molecule could be a novel biomarker for the study of the mechanisms that underlie migraine. The work also suggests that the barrier between blood and CSF, sometimes described as leaky, is in fact selectively permeable.
The findings complement recent PET and dynamic contrast-enhanced MRI studies that have shown no sign of damage to the blood brain barrier (BBB) in migraine. Instead, there may be heightened transport of some molecules from blood to the CSF, evidenced by greater increases in fibrinogen levels in CSF than albumin. sVCAM1 might influence BBB or blood-CSF barrier permeability, possibly as a protective measure against fibrinogen, according to Michael Harrington, MD, scientific director of neuroscience at the Huntington Medical Research Institutes, Pasadena, Calif., who presented the findings in a poster at the virtual annual meeting of the American Headache Society.
BBB disruption?
The BBB is a well-known structure that regulates what molecules enter the brain, but the blood-CSF barrier, while lesser known, is also important. It comprises choroid plexus epithelial cells that oversee selective exchange of waste products, ions, and nutrients. Acute inflammation or chronic effects from conditions like stroke, multiple sclerosis, and Alzheimer’s disease can alter the function of this barrier.
No other capillary biomarkers were different between controls and patients with migraine – only sVCAM1. “My data supports a highly selective transport change from blood to CSF, which I propose is less likely to come from brain capillaries than choroid plexuses, especially since choroid plexuses produce the bulk of the CSF. It’s a work in progress, but based on this likelihood of choroid plexus involvement, I am accumulating more data that support the choroid plexuses as the primary source of change in migraine,” said Dr. Harrington in an interview.
“The most important finding of the study is that the blood brain barrier is not compromised in people with migraine,” said Rami Burstein, PhD, professor of neuroscience at Harvard Medical School, Boston, who was asked to comment on the findings. “Most unwanted adverse events are caused by drug action inside the brain, and thus, peripherally acting drugs become more favorable as they usually have fewer side effects. Given that the headache aspect of migraine could be intercepted outside the brain, the fact that the BBB is not compromised is a very good news,” Dr. Burstein added.
Dr. Harrington’s team recruited 74 subjects: 14 nonmigraine controls, 16 who were experiencing migraine illness (ictal), 27 not experiencing migraine illness (interictal), and 17 with chronic migraine. The CSF/serum quotient for albumen was higher in the 60 migraineurs than in the 14 controls (5.6 g/L vs. 4.1 g/L; P = .04), as was the CSF/serum quotient for fibrinogen (161.5 g/L vs. 86.1 g/L; P = .007). CSF levels of plasminogen were also higher in patients with migraine (240.7 ng/mL vs. 186.2 ng/mL; P = .03).
When the researchers compared ictal to interictal subjects, they found no difference in fibrinogen or albumen. That suggested that these values are generally increased in migraine patients compared with controls, rather than spiking during attacks. They also divided subjects by annual frequency, including groups experiencing fewer than 24 migraines per year, 24-180 attacks per year, and more than 180 attacks per year. The quotient for fibrinogen increased in migraineurs in general, compared with controls, but then decreased as the frequency of migraine went up (198.6 g/L, 167.0 g/L, and 121.6 g/L, respectively; P = .004).
CSF levels of sVCAM1 were 4.7 ng/mL in controls, 4.5 in the group with fewer than 24 migraines per year, 5.5 in the 24-180 group, and 7.1 in the group with more than 180 (P = .004).
Implications for therapy
The research, though at a very early stage, could have implications for therapies. Most drugs that treat migraine remain something of a mystery because researchers don’t know for sure where they act. In the brain? Systemically? The question of permeability of various molecules through both barriers could lend insight into what’s happening. “That’s why there is interest in barrier transport, and we’re showing there is a selective change of transport in migraineurs,” said Dr. Harrington.
As for more general therapeutic implications, “I can only speculate, but clearly there is baseline altered transport, probably in the choroid plexuses of these people,” said Dr. Harrington. He added that in time researchers might test drugs to see if they alter sVCAM1 levels or even develop novel drug candidates to act directly on it.
But he also sounded a note of caution because of the exploratory nature of the study. “These are all really early speculations.”
The study was funded by NIH, the Sunstar Foundation, Wyngs Foundation, and the Higgins Family. Dr. Harrington has no relevant disclosures.
SOURCE: Harrington M et al. AHS 2020, Abstract 842752.
according to a new study. The molecule could be a novel biomarker for the study of the mechanisms that underlie migraine. The work also suggests that the barrier between blood and CSF, sometimes described as leaky, is in fact selectively permeable.
The findings complement recent PET and dynamic contrast-enhanced MRI studies that have shown no sign of damage to the blood brain barrier (BBB) in migraine. Instead, there may be heightened transport of some molecules from blood to the CSF, evidenced by greater increases in fibrinogen levels in CSF than albumin. sVCAM1 might influence BBB or blood-CSF barrier permeability, possibly as a protective measure against fibrinogen, according to Michael Harrington, MD, scientific director of neuroscience at the Huntington Medical Research Institutes, Pasadena, Calif., who presented the findings in a poster at the virtual annual meeting of the American Headache Society.
BBB disruption?
The BBB is a well-known structure that regulates what molecules enter the brain, but the blood-CSF barrier, while lesser known, is also important. It comprises choroid plexus epithelial cells that oversee selective exchange of waste products, ions, and nutrients. Acute inflammation or chronic effects from conditions like stroke, multiple sclerosis, and Alzheimer’s disease can alter the function of this barrier.
No other capillary biomarkers were different between controls and patients with migraine – only sVCAM1. “My data supports a highly selective transport change from blood to CSF, which I propose is less likely to come from brain capillaries than choroid plexuses, especially since choroid plexuses produce the bulk of the CSF. It’s a work in progress, but based on this likelihood of choroid plexus involvement, I am accumulating more data that support the choroid plexuses as the primary source of change in migraine,” said Dr. Harrington in an interview.
“The most important finding of the study is that the blood brain barrier is not compromised in people with migraine,” said Rami Burstein, PhD, professor of neuroscience at Harvard Medical School, Boston, who was asked to comment on the findings. “Most unwanted adverse events are caused by drug action inside the brain, and thus, peripherally acting drugs become more favorable as they usually have fewer side effects. Given that the headache aspect of migraine could be intercepted outside the brain, the fact that the BBB is not compromised is a very good news,” Dr. Burstein added.
Dr. Harrington’s team recruited 74 subjects: 14 nonmigraine controls, 16 who were experiencing migraine illness (ictal), 27 not experiencing migraine illness (interictal), and 17 with chronic migraine. The CSF/serum quotient for albumen was higher in the 60 migraineurs than in the 14 controls (5.6 g/L vs. 4.1 g/L; P = .04), as was the CSF/serum quotient for fibrinogen (161.5 g/L vs. 86.1 g/L; P = .007). CSF levels of plasminogen were also higher in patients with migraine (240.7 ng/mL vs. 186.2 ng/mL; P = .03).
When the researchers compared ictal to interictal subjects, they found no difference in fibrinogen or albumen. That suggested that these values are generally increased in migraine patients compared with controls, rather than spiking during attacks. They also divided subjects by annual frequency, including groups experiencing fewer than 24 migraines per year, 24-180 attacks per year, and more than 180 attacks per year. The quotient for fibrinogen increased in migraineurs in general, compared with controls, but then decreased as the frequency of migraine went up (198.6 g/L, 167.0 g/L, and 121.6 g/L, respectively; P = .004).
CSF levels of sVCAM1 were 4.7 ng/mL in controls, 4.5 in the group with fewer than 24 migraines per year, 5.5 in the 24-180 group, and 7.1 in the group with more than 180 (P = .004).
Implications for therapy
The research, though at a very early stage, could have implications for therapies. Most drugs that treat migraine remain something of a mystery because researchers don’t know for sure where they act. In the brain? Systemically? The question of permeability of various molecules through both barriers could lend insight into what’s happening. “That’s why there is interest in barrier transport, and we’re showing there is a selective change of transport in migraineurs,” said Dr. Harrington.
As for more general therapeutic implications, “I can only speculate, but clearly there is baseline altered transport, probably in the choroid plexuses of these people,” said Dr. Harrington. He added that in time researchers might test drugs to see if they alter sVCAM1 levels or even develop novel drug candidates to act directly on it.
But he also sounded a note of caution because of the exploratory nature of the study. “These are all really early speculations.”
The study was funded by NIH, the Sunstar Foundation, Wyngs Foundation, and the Higgins Family. Dr. Harrington has no relevant disclosures.
SOURCE: Harrington M et al. AHS 2020, Abstract 842752.
according to a new study. The molecule could be a novel biomarker for the study of the mechanisms that underlie migraine. The work also suggests that the barrier between blood and CSF, sometimes described as leaky, is in fact selectively permeable.
The findings complement recent PET and dynamic contrast-enhanced MRI studies that have shown no sign of damage to the blood brain barrier (BBB) in migraine. Instead, there may be heightened transport of some molecules from blood to the CSF, evidenced by greater increases in fibrinogen levels in CSF than albumin. sVCAM1 might influence BBB or blood-CSF barrier permeability, possibly as a protective measure against fibrinogen, according to Michael Harrington, MD, scientific director of neuroscience at the Huntington Medical Research Institutes, Pasadena, Calif., who presented the findings in a poster at the virtual annual meeting of the American Headache Society.
BBB disruption?
The BBB is a well-known structure that regulates what molecules enter the brain, but the blood-CSF barrier, while lesser known, is also important. It comprises choroid plexus epithelial cells that oversee selective exchange of waste products, ions, and nutrients. Acute inflammation or chronic effects from conditions like stroke, multiple sclerosis, and Alzheimer’s disease can alter the function of this barrier.
No other capillary biomarkers were different between controls and patients with migraine – only sVCAM1. “My data supports a highly selective transport change from blood to CSF, which I propose is less likely to come from brain capillaries than choroid plexuses, especially since choroid plexuses produce the bulk of the CSF. It’s a work in progress, but based on this likelihood of choroid plexus involvement, I am accumulating more data that support the choroid plexuses as the primary source of change in migraine,” said Dr. Harrington in an interview.
“The most important finding of the study is that the blood brain barrier is not compromised in people with migraine,” said Rami Burstein, PhD, professor of neuroscience at Harvard Medical School, Boston, who was asked to comment on the findings. “Most unwanted adverse events are caused by drug action inside the brain, and thus, peripherally acting drugs become more favorable as they usually have fewer side effects. Given that the headache aspect of migraine could be intercepted outside the brain, the fact that the BBB is not compromised is a very good news,” Dr. Burstein added.
Dr. Harrington’s team recruited 74 subjects: 14 nonmigraine controls, 16 who were experiencing migraine illness (ictal), 27 not experiencing migraine illness (interictal), and 17 with chronic migraine. The CSF/serum quotient for albumen was higher in the 60 migraineurs than in the 14 controls (5.6 g/L vs. 4.1 g/L; P = .04), as was the CSF/serum quotient for fibrinogen (161.5 g/L vs. 86.1 g/L; P = .007). CSF levels of plasminogen were also higher in patients with migraine (240.7 ng/mL vs. 186.2 ng/mL; P = .03).
When the researchers compared ictal to interictal subjects, they found no difference in fibrinogen or albumen. That suggested that these values are generally increased in migraine patients compared with controls, rather than spiking during attacks. They also divided subjects by annual frequency, including groups experiencing fewer than 24 migraines per year, 24-180 attacks per year, and more than 180 attacks per year. The quotient for fibrinogen increased in migraineurs in general, compared with controls, but then decreased as the frequency of migraine went up (198.6 g/L, 167.0 g/L, and 121.6 g/L, respectively; P = .004).
CSF levels of sVCAM1 were 4.7 ng/mL in controls, 4.5 in the group with fewer than 24 migraines per year, 5.5 in the 24-180 group, and 7.1 in the group with more than 180 (P = .004).
Implications for therapy
The research, though at a very early stage, could have implications for therapies. Most drugs that treat migraine remain something of a mystery because researchers don’t know for sure where they act. In the brain? Systemically? The question of permeability of various molecules through both barriers could lend insight into what’s happening. “That’s why there is interest in barrier transport, and we’re showing there is a selective change of transport in migraineurs,” said Dr. Harrington.
As for more general therapeutic implications, “I can only speculate, but clearly there is baseline altered transport, probably in the choroid plexuses of these people,” said Dr. Harrington. He added that in time researchers might test drugs to see if they alter sVCAM1 levels or even develop novel drug candidates to act directly on it.
But he also sounded a note of caution because of the exploratory nature of the study. “These are all really early speculations.”
The study was funded by NIH, the Sunstar Foundation, Wyngs Foundation, and the Higgins Family. Dr. Harrington has no relevant disclosures.
SOURCE: Harrington M et al. AHS 2020, Abstract 842752.
FROM AHS 2020
Multiple traits more common in difficult-to-treat patients with migraine
Overall, insufficient responders—patients less likely to get relief shortly after acute treatment—are “more medically and psychosocially complex,” wrote the authors of the study, which appeared in the July/August issue of Headache.
Common characteristics of insufficient responders
The researchers, led by Louise Lombard, M Nutr, of Eli Lilly and Company, analyzed data from a 2014 cross-sectional survey. They tracked 583 patients with migraine, including 200 (34%) who were considered insufficient responders because they failed to achieve freedom from pain within 2 hours of acute treatment in at least four of five attacks.
The insufficient and sufficient responder groups were similar in age (mean = 40 for both) and gender (80% and 75% female, respectively, P = .170) and race (72% and 77% white, P = .279).
However, insufficient responders were clearly more affected by headaches, multiple treatments, and other burdens. Compared with those who had better responses to treatment, they were more likely to have four or more migraine headache days per month (46% vs. 31%), rebound or medication-overuse headaches (16% vs. 7%) and chronic migraine (12% vs. 5%, all P < .05).
They were also more likely have comorbid depression (38% vs. 22%) and psychological conditions other than depression and anxiety (8% vs. 4%, all P < .05).
As for treatment, insufficient response was higher in patients who waited until the appearance of pain to take medication (odds ratio = 1.83, 95% confidence interval [CI] 1.15–2.92, P = .011, after adjustment for covariates). And insufficient responders were more likely to have been prescribed at least three unique preventive regimens (12% vs. 6%), to take over-the-counter medications (50% vs. 38%) and to take opioid painkillers (16% vs. 8%, all P < .05).
The authors, who caution that the study does not prove cause and effect, wrote that insufficient responders “may benefit from education on how and when to use current treatments.”
Managing insufficient responders
Neurology Reviews editor-in-chief Alan M. Rapoport, MD, said the study “confirms a lot of what we knew.” Dr, Rapoport, who was not involved in the study, is clinical professor of neurology at the University of California, Los Angeles.
“As expected, the insufficient responders used more opioids and over-the-counter medications, which is not the ideal way to treat migraine,” he said. “That probably caused them to have medication-overuse headache, which might have caused them to respond poorly to even the best treatment regimen. They also had more severe symptoms, more comorbidities, and a poorer quality of life. They also had more impairment and greater impact on work, with more of them unemployed.”
The insufficient responders also “took medication at the time or after the pain began, rather than before it when they thought the attack was beginning due to premonitory symptoms,” he said.
Dr. Rapoport also noted a surprising and unusual finding: Patients who did not report sensitivity to light as their most bothersome symptom were more likely to be insufficient responders (OR = 2.3, 95% CI [1.21–4.37], P = .011). “In all recent migraine studies,” he said, “the majority of patients selected photophobia as their most bothersome symptom.”
In the big picture, he said, the study suggests that “a third triptan does not seem to work better than the first two, patients with medication-overuse headache and chronic migraine and those not on preventive medication do not respond that well to acute care treatment, and the same is true when depression is present.”
No study funding was reported. Four study authors reported ties with Eli Lilly, and two reported employment by Adelphi Real World, which provided the survey results..
SOURCE: Lombard L et al. Headache. 2020;60(7):1325-39. doi: 10.1111/head.13835.
Overall, insufficient responders—patients less likely to get relief shortly after acute treatment—are “more medically and psychosocially complex,” wrote the authors of the study, which appeared in the July/August issue of Headache.
Common characteristics of insufficient responders
The researchers, led by Louise Lombard, M Nutr, of Eli Lilly and Company, analyzed data from a 2014 cross-sectional survey. They tracked 583 patients with migraine, including 200 (34%) who were considered insufficient responders because they failed to achieve freedom from pain within 2 hours of acute treatment in at least four of five attacks.
The insufficient and sufficient responder groups were similar in age (mean = 40 for both) and gender (80% and 75% female, respectively, P = .170) and race (72% and 77% white, P = .279).
However, insufficient responders were clearly more affected by headaches, multiple treatments, and other burdens. Compared with those who had better responses to treatment, they were more likely to have four or more migraine headache days per month (46% vs. 31%), rebound or medication-overuse headaches (16% vs. 7%) and chronic migraine (12% vs. 5%, all P < .05).
They were also more likely have comorbid depression (38% vs. 22%) and psychological conditions other than depression and anxiety (8% vs. 4%, all P < .05).
As for treatment, insufficient response was higher in patients who waited until the appearance of pain to take medication (odds ratio = 1.83, 95% confidence interval [CI] 1.15–2.92, P = .011, after adjustment for covariates). And insufficient responders were more likely to have been prescribed at least three unique preventive regimens (12% vs. 6%), to take over-the-counter medications (50% vs. 38%) and to take opioid painkillers (16% vs. 8%, all P < .05).
The authors, who caution that the study does not prove cause and effect, wrote that insufficient responders “may benefit from education on how and when to use current treatments.”
Managing insufficient responders
Neurology Reviews editor-in-chief Alan M. Rapoport, MD, said the study “confirms a lot of what we knew.” Dr, Rapoport, who was not involved in the study, is clinical professor of neurology at the University of California, Los Angeles.
“As expected, the insufficient responders used more opioids and over-the-counter medications, which is not the ideal way to treat migraine,” he said. “That probably caused them to have medication-overuse headache, which might have caused them to respond poorly to even the best treatment regimen. They also had more severe symptoms, more comorbidities, and a poorer quality of life. They also had more impairment and greater impact on work, with more of them unemployed.”
The insufficient responders also “took medication at the time or after the pain began, rather than before it when they thought the attack was beginning due to premonitory symptoms,” he said.
Dr. Rapoport also noted a surprising and unusual finding: Patients who did not report sensitivity to light as their most bothersome symptom were more likely to be insufficient responders (OR = 2.3, 95% CI [1.21–4.37], P = .011). “In all recent migraine studies,” he said, “the majority of patients selected photophobia as their most bothersome symptom.”
In the big picture, he said, the study suggests that “a third triptan does not seem to work better than the first two, patients with medication-overuse headache and chronic migraine and those not on preventive medication do not respond that well to acute care treatment, and the same is true when depression is present.”
No study funding was reported. Four study authors reported ties with Eli Lilly, and two reported employment by Adelphi Real World, which provided the survey results..
SOURCE: Lombard L et al. Headache. 2020;60(7):1325-39. doi: 10.1111/head.13835.
Overall, insufficient responders—patients less likely to get relief shortly after acute treatment—are “more medically and psychosocially complex,” wrote the authors of the study, which appeared in the July/August issue of Headache.
Common characteristics of insufficient responders
The researchers, led by Louise Lombard, M Nutr, of Eli Lilly and Company, analyzed data from a 2014 cross-sectional survey. They tracked 583 patients with migraine, including 200 (34%) who were considered insufficient responders because they failed to achieve freedom from pain within 2 hours of acute treatment in at least four of five attacks.
The insufficient and sufficient responder groups were similar in age (mean = 40 for both) and gender (80% and 75% female, respectively, P = .170) and race (72% and 77% white, P = .279).
However, insufficient responders were clearly more affected by headaches, multiple treatments, and other burdens. Compared with those who had better responses to treatment, they were more likely to have four or more migraine headache days per month (46% vs. 31%), rebound or medication-overuse headaches (16% vs. 7%) and chronic migraine (12% vs. 5%, all P < .05).
They were also more likely have comorbid depression (38% vs. 22%) and psychological conditions other than depression and anxiety (8% vs. 4%, all P < .05).
As for treatment, insufficient response was higher in patients who waited until the appearance of pain to take medication (odds ratio = 1.83, 95% confidence interval [CI] 1.15–2.92, P = .011, after adjustment for covariates). And insufficient responders were more likely to have been prescribed at least three unique preventive regimens (12% vs. 6%), to take over-the-counter medications (50% vs. 38%) and to take opioid painkillers (16% vs. 8%, all P < .05).
The authors, who caution that the study does not prove cause and effect, wrote that insufficient responders “may benefit from education on how and when to use current treatments.”
Managing insufficient responders
Neurology Reviews editor-in-chief Alan M. Rapoport, MD, said the study “confirms a lot of what we knew.” Dr, Rapoport, who was not involved in the study, is clinical professor of neurology at the University of California, Los Angeles.
“As expected, the insufficient responders used more opioids and over-the-counter medications, which is not the ideal way to treat migraine,” he said. “That probably caused them to have medication-overuse headache, which might have caused them to respond poorly to even the best treatment regimen. They also had more severe symptoms, more comorbidities, and a poorer quality of life. They also had more impairment and greater impact on work, with more of them unemployed.”
The insufficient responders also “took medication at the time or after the pain began, rather than before it when they thought the attack was beginning due to premonitory symptoms,” he said.
Dr. Rapoport also noted a surprising and unusual finding: Patients who did not report sensitivity to light as their most bothersome symptom were more likely to be insufficient responders (OR = 2.3, 95% CI [1.21–4.37], P = .011). “In all recent migraine studies,” he said, “the majority of patients selected photophobia as their most bothersome symptom.”
In the big picture, he said, the study suggests that “a third triptan does not seem to work better than the first two, patients with medication-overuse headache and chronic migraine and those not on preventive medication do not respond that well to acute care treatment, and the same is true when depression is present.”
No study funding was reported. Four study authors reported ties with Eli Lilly, and two reported employment by Adelphi Real World, which provided the survey results..
SOURCE: Lombard L et al. Headache. 2020;60(7):1325-39. doi: 10.1111/head.13835.
FROM HEADACHE
Positive phase 3 top-line results for migraine prevention drug
AbbVie, the company developing the drug, has announced.
Top-line results from the ADVANCE trial, which evaluated atogepant 10, 30, and 60 mg, showed all three doses were associated with a significant reduction from baseline in mean monthly migraine days, compared with placebo.
There were also significant improvements in all six secondary endpoints with the two higher doses.
Data from the ADVANCE trial and a previous phase 2/3 trial will be the basis for regulatory submissions in the United States and other countries, AbbVie reported.
Decreased migraine days
The phase 3, multicenter, randomized, double-blind, placebo-controlled, parallel-group trial was designed to evaluate the efficacy, safety, and tolerability of oral atogepant for the prevention of migraine in those who experienced 4-14 migraine days per month.
A total of 910 patients were randomized to one of four treatment groups: 10 mg, 30 mg, or 60 mg of atogepant once daily or placebo. Efficacy analyses were based on the modified intent-to-treat population of 873 patients.
The primary endpoint was change from baseline in mean monthly migraine days during the 12-week treatment period. All atogepant dose groups met the primary endpoint and demonstrated significantly greater decreases in mean monthly migraine days, compared with placebo.
Mean monthly migraine days were reduced by 3.69 days with the 10-mg dose, 3.86 days with the 30-mg dose, and 4.2 days with the 60-mg dose of atogepant, compared with a reduction of 2.48 migraine days in the placebo group (P < .0001, all dose groups vs. placebo).
A key secondary endpoint measured the proportion of patients who achieved at least a 50% reduction in mean monthly migraine days over 12 weeks. This outcome occurred in 55.6% of the 10-mg atogepant group, 58.7% of the 30-mg group, and 60.8% of the 60-mg group, compared with 29% of the placebo group (P < .0001, all dose groups vs. placebo).
Significant improvements
Additional secondary endpoints measured during the 12-week treatment period included change from baseline in mean monthly headache days, mean monthly acute–medication use days, mean monthly performance of daily activities and physical impairment domain scores on the Activity Impairment in Migraine-Diary, and change from baseline in the Migraine-Specific Quality of Life Questionnaire Role Function-Restrictive domain score at week 12. Treatment with the 30-mg and 60-mg doses resulted in significant improvements in all secondary endpoints, and treatment with the 10-mg dose resulted in significant improvements in four of the six secondary endpoints.
No new safety risks were observed when compared with the safety profile of atogepant observed in previous trials, AbbVie said. Serious adverse events occurred in 0.9% of patients in the atogepant 10-mg group versus 0.9% of patients in the placebo group. No patients in the atogepant 30-mg or 60-mg groups experienced a serious adverse event. The most common adverse events (reported in at least 5% of patients and at least one atogepant group and at a rate greater than placebo), across all doses versus placebo, were constipation (6.9%-7.7% vs. 0.5%), nausea (4.4%-6.1% vs. 1.8%), and upper respiratory tract infection (3.9%-5.7% vs. 4.5%).
Most cases of constipation, nausea, and upper respiratory tract infection were mild or moderate in severity and did not lead to discontinuation. There were no hepatic safety issues identified in the trial.
Full data results will be presented at an upcoming medical congress and/or published in a peer-reviewed journal, the company said.
A version of this article originally appeared on Medscape.com.
AbbVie, the company developing the drug, has announced.
Top-line results from the ADVANCE trial, which evaluated atogepant 10, 30, and 60 mg, showed all three doses were associated with a significant reduction from baseline in mean monthly migraine days, compared with placebo.
There were also significant improvements in all six secondary endpoints with the two higher doses.
Data from the ADVANCE trial and a previous phase 2/3 trial will be the basis for regulatory submissions in the United States and other countries, AbbVie reported.
Decreased migraine days
The phase 3, multicenter, randomized, double-blind, placebo-controlled, parallel-group trial was designed to evaluate the efficacy, safety, and tolerability of oral atogepant for the prevention of migraine in those who experienced 4-14 migraine days per month.
A total of 910 patients were randomized to one of four treatment groups: 10 mg, 30 mg, or 60 mg of atogepant once daily or placebo. Efficacy analyses were based on the modified intent-to-treat population of 873 patients.
The primary endpoint was change from baseline in mean monthly migraine days during the 12-week treatment period. All atogepant dose groups met the primary endpoint and demonstrated significantly greater decreases in mean monthly migraine days, compared with placebo.
Mean monthly migraine days were reduced by 3.69 days with the 10-mg dose, 3.86 days with the 30-mg dose, and 4.2 days with the 60-mg dose of atogepant, compared with a reduction of 2.48 migraine days in the placebo group (P < .0001, all dose groups vs. placebo).
A key secondary endpoint measured the proportion of patients who achieved at least a 50% reduction in mean monthly migraine days over 12 weeks. This outcome occurred in 55.6% of the 10-mg atogepant group, 58.7% of the 30-mg group, and 60.8% of the 60-mg group, compared with 29% of the placebo group (P < .0001, all dose groups vs. placebo).
Significant improvements
Additional secondary endpoints measured during the 12-week treatment period included change from baseline in mean monthly headache days, mean monthly acute–medication use days, mean monthly performance of daily activities and physical impairment domain scores on the Activity Impairment in Migraine-Diary, and change from baseline in the Migraine-Specific Quality of Life Questionnaire Role Function-Restrictive domain score at week 12. Treatment with the 30-mg and 60-mg doses resulted in significant improvements in all secondary endpoints, and treatment with the 10-mg dose resulted in significant improvements in four of the six secondary endpoints.
No new safety risks were observed when compared with the safety profile of atogepant observed in previous trials, AbbVie said. Serious adverse events occurred in 0.9% of patients in the atogepant 10-mg group versus 0.9% of patients in the placebo group. No patients in the atogepant 30-mg or 60-mg groups experienced a serious adverse event. The most common adverse events (reported in at least 5% of patients and at least one atogepant group and at a rate greater than placebo), across all doses versus placebo, were constipation (6.9%-7.7% vs. 0.5%), nausea (4.4%-6.1% vs. 1.8%), and upper respiratory tract infection (3.9%-5.7% vs. 4.5%).
Most cases of constipation, nausea, and upper respiratory tract infection were mild or moderate in severity and did not lead to discontinuation. There were no hepatic safety issues identified in the trial.
Full data results will be presented at an upcoming medical congress and/or published in a peer-reviewed journal, the company said.
A version of this article originally appeared on Medscape.com.
AbbVie, the company developing the drug, has announced.
Top-line results from the ADVANCE trial, which evaluated atogepant 10, 30, and 60 mg, showed all three doses were associated with a significant reduction from baseline in mean monthly migraine days, compared with placebo.
There were also significant improvements in all six secondary endpoints with the two higher doses.
Data from the ADVANCE trial and a previous phase 2/3 trial will be the basis for regulatory submissions in the United States and other countries, AbbVie reported.
Decreased migraine days
The phase 3, multicenter, randomized, double-blind, placebo-controlled, parallel-group trial was designed to evaluate the efficacy, safety, and tolerability of oral atogepant for the prevention of migraine in those who experienced 4-14 migraine days per month.
A total of 910 patients were randomized to one of four treatment groups: 10 mg, 30 mg, or 60 mg of atogepant once daily or placebo. Efficacy analyses were based on the modified intent-to-treat population of 873 patients.
The primary endpoint was change from baseline in mean monthly migraine days during the 12-week treatment period. All atogepant dose groups met the primary endpoint and demonstrated significantly greater decreases in mean monthly migraine days, compared with placebo.
Mean monthly migraine days were reduced by 3.69 days with the 10-mg dose, 3.86 days with the 30-mg dose, and 4.2 days with the 60-mg dose of atogepant, compared with a reduction of 2.48 migraine days in the placebo group (P < .0001, all dose groups vs. placebo).
A key secondary endpoint measured the proportion of patients who achieved at least a 50% reduction in mean monthly migraine days over 12 weeks. This outcome occurred in 55.6% of the 10-mg atogepant group, 58.7% of the 30-mg group, and 60.8% of the 60-mg group, compared with 29% of the placebo group (P < .0001, all dose groups vs. placebo).
Significant improvements
Additional secondary endpoints measured during the 12-week treatment period included change from baseline in mean monthly headache days, mean monthly acute–medication use days, mean monthly performance of daily activities and physical impairment domain scores on the Activity Impairment in Migraine-Diary, and change from baseline in the Migraine-Specific Quality of Life Questionnaire Role Function-Restrictive domain score at week 12. Treatment with the 30-mg and 60-mg doses resulted in significant improvements in all secondary endpoints, and treatment with the 10-mg dose resulted in significant improvements in four of the six secondary endpoints.
No new safety risks were observed when compared with the safety profile of atogepant observed in previous trials, AbbVie said. Serious adverse events occurred in 0.9% of patients in the atogepant 10-mg group versus 0.9% of patients in the placebo group. No patients in the atogepant 30-mg or 60-mg groups experienced a serious adverse event. The most common adverse events (reported in at least 5% of patients and at least one atogepant group and at a rate greater than placebo), across all doses versus placebo, were constipation (6.9%-7.7% vs. 0.5%), nausea (4.4%-6.1% vs. 1.8%), and upper respiratory tract infection (3.9%-5.7% vs. 4.5%).
Most cases of constipation, nausea, and upper respiratory tract infection were mild or moderate in severity and did not lead to discontinuation. There were no hepatic safety issues identified in the trial.
Full data results will be presented at an upcoming medical congress and/or published in a peer-reviewed journal, the company said.
A version of this article originally appeared on Medscape.com.
Migraine headache pearls
A 25-year-old woman presents to discuss treatment of her headaches. They occur two or three times a month and last for 4-6 hours. The headaches are disabling, have a pounding quality behind the patient’s right eye, and worsen with exercise. The patient’s neurologic exam is normal.
She has tried oral sumatriptan and naproxen, but neither drug provided her with any relief from the headaches. What treatment would you recommend?
A. Topiramate
B. Beta-blocker
C. Lasmiditan
D. Metoclopramide plus sumatriptan
E. Ubrogepant
It is common to see patients with migraine headaches and to see patients with migraines who have not responded to previous migraine therapies.
For this patient, I would try choice D first, giving metoclopramide with oral sumatriptan to see if it can improve response to sumatriptan. The two new classes of drugs for acute migraine therapy, the gepants and ditans, certainly have a role in patients unresponsive or intolerant of triptans/NSAIDS, but I would try several tricks with these less expensive medications first before entering into prior authorization hell trying to get a gepant or ditan.
When a patient has already used a triptan but experienced no benefit from it, often the next medication a patient tries is a different triptan. Dahlof reviewed four trials that looked at the efficacy of switching sumatriptan nonresponders to a different triptan and found that lack of response to sumatriptan did not predict lack of response to an alternative triptan.1 Unfortunately, acquiring insurance coverage for an alternate triptan can be difficult.
Other treatment options are nasal or injectable formulations of sumatriptan. Both of these are more costly than oral sumatriptan, and injectable sumatriptan has more side effects than oral triptans.
Combining treatment with metoclopramide can be helpful. In a study by Schulman and Dermott looking at patients who had previously been triptan nonresponders, 63% of those who took metoclopramide with sumatriptan had meaningful pain relief, compared with 31% of those who received sumatriptan and placebo.2
In a different study, Tfelt-Hansen et al. compared treatment with the combination of lysine acetylsalicylate plus metoclopramide versus treatment with 100 mg of sumatriptan.3 There was no difference in outcomes between the two treatment groups, with the lysine acetylsalicylate plus metoclopramide patients having a 57% success rate for first treated migraine compared with 53% of the sumatriptan-treated patients.
Treating with the combination of naproxen plus sumatriptan is superior to treating with either medication alone. Brandes et al. reported on two studies involving the use of the sumatriptan/naproxen combination, compared with using sumatriptan, naproxen, or placebo.4 In both, taking the sumatriptan/naproxen combination was superior to taking sumatriptan, naproxen, or placebo (P < .001).
In a study of patients with poor prior response to triptans, Mathew et al. found that the sumatriptan/naproxen combination was superior to placebo for both 2- and 24-hour headache relief (P < .001).5
Pearl
Try several options before abandoning triptans in previous triptan nonresponders, including trying a different triptan, adding metoclopramide, orcombining with an NSAID.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Dahlöf CG. Infrequent or nonresponse to oral sumatriptan does not predict response to other triptans – review of four trials. Cephalalgia. 2006 Feb;26(2):98-106.
2. Schulman EA, Dermott KF. Sumatriptan plus metoclopramide in triptan-nonresponsive migraineurs. Headache. 2003 Jul-Aug;43(7):729-33.
3. Tfelt-Hansen P et al. The effectiveness of combined oral lysine acetylsalicylate and metoclopramide compared with oral sumatriptan for migraine. Lancet. 1995 Oct 7;346(8980):923-6.
4. Brandes JL et al. Sumatriptan‐naproxen for acute treatment of migraine: A randomized trial. JAMA. 2007;297:1443‐54.
5. Mathew NT, Landy S, Stark S, et al. Fixed‐dose sumatriptan and naproxen in poor responders to triptans with a short half‐life. Headache. 2009;49:971‐82.
A 25-year-old woman presents to discuss treatment of her headaches. They occur two or three times a month and last for 4-6 hours. The headaches are disabling, have a pounding quality behind the patient’s right eye, and worsen with exercise. The patient’s neurologic exam is normal.
She has tried oral sumatriptan and naproxen, but neither drug provided her with any relief from the headaches. What treatment would you recommend?
A. Topiramate
B. Beta-blocker
C. Lasmiditan
D. Metoclopramide plus sumatriptan
E. Ubrogepant
It is common to see patients with migraine headaches and to see patients with migraines who have not responded to previous migraine therapies.
For this patient, I would try choice D first, giving metoclopramide with oral sumatriptan to see if it can improve response to sumatriptan. The two new classes of drugs for acute migraine therapy, the gepants and ditans, certainly have a role in patients unresponsive or intolerant of triptans/NSAIDS, but I would try several tricks with these less expensive medications first before entering into prior authorization hell trying to get a gepant or ditan.
When a patient has already used a triptan but experienced no benefit from it, often the next medication a patient tries is a different triptan. Dahlof reviewed four trials that looked at the efficacy of switching sumatriptan nonresponders to a different triptan and found that lack of response to sumatriptan did not predict lack of response to an alternative triptan.1 Unfortunately, acquiring insurance coverage for an alternate triptan can be difficult.
Other treatment options are nasal or injectable formulations of sumatriptan. Both of these are more costly than oral sumatriptan, and injectable sumatriptan has more side effects than oral triptans.
Combining treatment with metoclopramide can be helpful. In a study by Schulman and Dermott looking at patients who had previously been triptan nonresponders, 63% of those who took metoclopramide with sumatriptan had meaningful pain relief, compared with 31% of those who received sumatriptan and placebo.2
In a different study, Tfelt-Hansen et al. compared treatment with the combination of lysine acetylsalicylate plus metoclopramide versus treatment with 100 mg of sumatriptan.3 There was no difference in outcomes between the two treatment groups, with the lysine acetylsalicylate plus metoclopramide patients having a 57% success rate for first treated migraine compared with 53% of the sumatriptan-treated patients.
Treating with the combination of naproxen plus sumatriptan is superior to treating with either medication alone. Brandes et al. reported on two studies involving the use of the sumatriptan/naproxen combination, compared with using sumatriptan, naproxen, or placebo.4 In both, taking the sumatriptan/naproxen combination was superior to taking sumatriptan, naproxen, or placebo (P < .001).
In a study of patients with poor prior response to triptans, Mathew et al. found that the sumatriptan/naproxen combination was superior to placebo for both 2- and 24-hour headache relief (P < .001).5
Pearl
Try several options before abandoning triptans in previous triptan nonresponders, including trying a different triptan, adding metoclopramide, orcombining with an NSAID.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Dahlöf CG. Infrequent or nonresponse to oral sumatriptan does not predict response to other triptans – review of four trials. Cephalalgia. 2006 Feb;26(2):98-106.
2. Schulman EA, Dermott KF. Sumatriptan plus metoclopramide in triptan-nonresponsive migraineurs. Headache. 2003 Jul-Aug;43(7):729-33.
3. Tfelt-Hansen P et al. The effectiveness of combined oral lysine acetylsalicylate and metoclopramide compared with oral sumatriptan for migraine. Lancet. 1995 Oct 7;346(8980):923-6.
4. Brandes JL et al. Sumatriptan‐naproxen for acute treatment of migraine: A randomized trial. JAMA. 2007;297:1443‐54.
5. Mathew NT, Landy S, Stark S, et al. Fixed‐dose sumatriptan and naproxen in poor responders to triptans with a short half‐life. Headache. 2009;49:971‐82.
A 25-year-old woman presents to discuss treatment of her headaches. They occur two or three times a month and last for 4-6 hours. The headaches are disabling, have a pounding quality behind the patient’s right eye, and worsen with exercise. The patient’s neurologic exam is normal.
She has tried oral sumatriptan and naproxen, but neither drug provided her with any relief from the headaches. What treatment would you recommend?
A. Topiramate
B. Beta-blocker
C. Lasmiditan
D. Metoclopramide plus sumatriptan
E. Ubrogepant
It is common to see patients with migraine headaches and to see patients with migraines who have not responded to previous migraine therapies.
For this patient, I would try choice D first, giving metoclopramide with oral sumatriptan to see if it can improve response to sumatriptan. The two new classes of drugs for acute migraine therapy, the gepants and ditans, certainly have a role in patients unresponsive or intolerant of triptans/NSAIDS, but I would try several tricks with these less expensive medications first before entering into prior authorization hell trying to get a gepant or ditan.
When a patient has already used a triptan but experienced no benefit from it, often the next medication a patient tries is a different triptan. Dahlof reviewed four trials that looked at the efficacy of switching sumatriptan nonresponders to a different triptan and found that lack of response to sumatriptan did not predict lack of response to an alternative triptan.1 Unfortunately, acquiring insurance coverage for an alternate triptan can be difficult.
Other treatment options are nasal or injectable formulations of sumatriptan. Both of these are more costly than oral sumatriptan, and injectable sumatriptan has more side effects than oral triptans.
Combining treatment with metoclopramide can be helpful. In a study by Schulman and Dermott looking at patients who had previously been triptan nonresponders, 63% of those who took metoclopramide with sumatriptan had meaningful pain relief, compared with 31% of those who received sumatriptan and placebo.2
In a different study, Tfelt-Hansen et al. compared treatment with the combination of lysine acetylsalicylate plus metoclopramide versus treatment with 100 mg of sumatriptan.3 There was no difference in outcomes between the two treatment groups, with the lysine acetylsalicylate plus metoclopramide patients having a 57% success rate for first treated migraine compared with 53% of the sumatriptan-treated patients.
Treating with the combination of naproxen plus sumatriptan is superior to treating with either medication alone. Brandes et al. reported on two studies involving the use of the sumatriptan/naproxen combination, compared with using sumatriptan, naproxen, or placebo.4 In both, taking the sumatriptan/naproxen combination was superior to taking sumatriptan, naproxen, or placebo (P < .001).
In a study of patients with poor prior response to triptans, Mathew et al. found that the sumatriptan/naproxen combination was superior to placebo for both 2- and 24-hour headache relief (P < .001).5
Pearl
Try several options before abandoning triptans in previous triptan nonresponders, including trying a different triptan, adding metoclopramide, orcombining with an NSAID.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Dahlöf CG. Infrequent or nonresponse to oral sumatriptan does not predict response to other triptans – review of four trials. Cephalalgia. 2006 Feb;26(2):98-106.
2. Schulman EA, Dermott KF. Sumatriptan plus metoclopramide in triptan-nonresponsive migraineurs. Headache. 2003 Jul-Aug;43(7):729-33.
3. Tfelt-Hansen P et al. The effectiveness of combined oral lysine acetylsalicylate and metoclopramide compared with oral sumatriptan for migraine. Lancet. 1995 Oct 7;346(8980):923-6.
4. Brandes JL et al. Sumatriptan‐naproxen for acute treatment of migraine: A randomized trial. JAMA. 2007;297:1443‐54.
5. Mathew NT, Landy S, Stark S, et al. Fixed‐dose sumatriptan and naproxen in poor responders to triptans with a short half‐life. Headache. 2009;49:971‐82.
Migraine is often a deciding factor in pregnancy planning
new research shows. Results from a multicenter study of more than 600 women showed that, among participants with migraine, those who were younger, had menstrual migraine, or had chronic migraine were more likely to decide to not become pregnant.
Although women with migraine who avoided pregnancy believed their migraines would worsen during pregnancy or make their pregnancy difficult, previous observational research indicates that migraine often improves during pregnancy.
“Women who avoided pregnancy due to migraine were most concerned that migraine would make raising a child difficult, that the migraine medications they take would have a negative impact on their child’s development, and that their migraine pattern would worsen during or just after pregnancy,” said study investigator Ryotaro Ishii, MD, PhD, a visiting scientist at Mayo Clinic in Phoenix, Arizona.
The findings were presented at the virtual annual meeting of the American Headache Society.
Plans for the future
There is a paucity of research on the effects of migraine on pregnancy planning, the researchers noted. The few studies that have investigated this issue have focused on women’s previous family planning decisions and experience rather than on plans for the future, the researchers noted.
To evaluate how migraine in women influences pregnancy planning, the investigators analyzed data from the American Registry for Migraine Research (ARMR). The registry, which was established by the American Migraine Foundation, collects clinical data about individuals with migraine and other headache disorders from multiple centers.
Participants eligible for the current analysis were women who had been diagnosed with migraine on the basis of the International Classification of Headache Disorders–3 criteria. All completed the ARMR questionnaire between February 2016 and September 2019. The investigators excluded patients with trigeminal autonomic cephalalgia, secondary headache, painful cranial neuropathies, other facial pain, and other headaches.
They identified 895 eligible women with migraine. Of these, 607 completed the pregnancy question. Among those participants, 121 women (19.9%) reported that migraine was a factor in their decision to not become pregnant. Of this group, 70 (11.5%) reported that migraine was a “significant” factor in deciding to not have children, and 8.4% said it was “somewhat” of a factor. The remainder of the cohort (479) reported that migraine had no influence on their pregnancy plans.
There were no between-group differences by race, marital status, employment, or income. This finding suggests that sociodemographic differences “have less impact on pregnancy planning than migraine-specific characteristics like headache frequency and experience with having migraine attacks triggered by menstruation,” Dr. Ishii said.
“Substantial burden”
Not surprisingly, women who avoided pregnancy had fewer children than the rest of the sample. About 60% of those who made the decision to not become pregnant had no children, and 72% had not been pregnant since they began experiencing migraine.
Compared with women who reported that migraine had no influence on their pregnancy plans, those who avoided pregnancy were more likely to have chronic migraine at 81.8% versus 70.2%. They were also more likely to have menstrual migraine at 4.1% versus 1%. In addition, women who decided to not have children because of migraine were significantly younger at an average age of 37.5 versus 47.2 years.
The number of days with headache per 3-month interval was 53.9 among women who avoided pregnancy versus 42.5 among the other women. The Migraine Disability Assessment score was also higher for women who avoided pregnancy (132.5) than for it was the other women (91.7), indicating more severe disability.
In addition, more of the women who avoided pregnancy had a history of depression (48.8%) compared with the other women (37.7%). The average score on the Patient Health Questionnaire–4 was higher among women who avoided pregnancy (4.0) than among other women (3.1), which indicates greater anxiety or depression. Among women who avoided pregnancy, 72.5% believed their migraine would worsen during pregnancy, and 68.3% believed that migraine would make pregnancy very difficult.
“Clinicians need to recognize that migraine often has a substantial burden on multiple aspects of life, including one’s plans for having children,” Dr. Ishii said.
“Clinicians should educate their patients who are considering pregnancy about the most likely course of migraine during pregnancy, migraine treatment during pregnancy, and the potential impacts of migraine and its treatment on pregnancy outcomes,” he added.
More education needed
Commenting on the study, Susan Hutchinson, MD, director of the Orange County Migraine and Headache Center, Irvine, California, said that not knowing how pregnancy is going to affect patients’ migraines can be “very scary” for women. In addition, patients often wonder what migraine treatments they can safely take once they do become pregnant, said Dr. Hutchinson, who was not involved in the research.
She noted that advantages of the ARMR data are that they are derived from a multicenter study and that migraine diagnoses were made by a headache specialist. A potential limitation of the study is that the population may not reflect outcomes of the millions of women who have migraine and become pregnant but never see a specialist.
“These findings show that more education is needed,” Dr. Hutchinson said.
Most women, especially those who have migraine without aura, note improvement with migraine during pregnancy, primarily because of the high, steady levels of estradiol, especially in the second and third trimesters, she said. In light of this, neurologists should reassure women that migraine is not a contraindication to pregnancy, she added.
There is also a need for additional research to assess how past experience with migraine and pregnancy influences a woman’s comfort level with additional pregnancies. Studies as to which treatments are safest for acute and preventive treatment of migraine during prepregnancy, pregnancy, and lactation are also needed, Dr. Hutchinson noted.
“If women knew they had treatment options that were evidence-based, they might be much more comfortable contemplating a pregnancy,” she said.
Dr. Ishii and Dr. Hutchinson have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research shows. Results from a multicenter study of more than 600 women showed that, among participants with migraine, those who were younger, had menstrual migraine, or had chronic migraine were more likely to decide to not become pregnant.
Although women with migraine who avoided pregnancy believed their migraines would worsen during pregnancy or make their pregnancy difficult, previous observational research indicates that migraine often improves during pregnancy.
“Women who avoided pregnancy due to migraine were most concerned that migraine would make raising a child difficult, that the migraine medications they take would have a negative impact on their child’s development, and that their migraine pattern would worsen during or just after pregnancy,” said study investigator Ryotaro Ishii, MD, PhD, a visiting scientist at Mayo Clinic in Phoenix, Arizona.
The findings were presented at the virtual annual meeting of the American Headache Society.
Plans for the future
There is a paucity of research on the effects of migraine on pregnancy planning, the researchers noted. The few studies that have investigated this issue have focused on women’s previous family planning decisions and experience rather than on plans for the future, the researchers noted.
To evaluate how migraine in women influences pregnancy planning, the investigators analyzed data from the American Registry for Migraine Research (ARMR). The registry, which was established by the American Migraine Foundation, collects clinical data about individuals with migraine and other headache disorders from multiple centers.
Participants eligible for the current analysis were women who had been diagnosed with migraine on the basis of the International Classification of Headache Disorders–3 criteria. All completed the ARMR questionnaire between February 2016 and September 2019. The investigators excluded patients with trigeminal autonomic cephalalgia, secondary headache, painful cranial neuropathies, other facial pain, and other headaches.
They identified 895 eligible women with migraine. Of these, 607 completed the pregnancy question. Among those participants, 121 women (19.9%) reported that migraine was a factor in their decision to not become pregnant. Of this group, 70 (11.5%) reported that migraine was a “significant” factor in deciding to not have children, and 8.4% said it was “somewhat” of a factor. The remainder of the cohort (479) reported that migraine had no influence on their pregnancy plans.
There were no between-group differences by race, marital status, employment, or income. This finding suggests that sociodemographic differences “have less impact on pregnancy planning than migraine-specific characteristics like headache frequency and experience with having migraine attacks triggered by menstruation,” Dr. Ishii said.
“Substantial burden”
Not surprisingly, women who avoided pregnancy had fewer children than the rest of the sample. About 60% of those who made the decision to not become pregnant had no children, and 72% had not been pregnant since they began experiencing migraine.
Compared with women who reported that migraine had no influence on their pregnancy plans, those who avoided pregnancy were more likely to have chronic migraine at 81.8% versus 70.2%. They were also more likely to have menstrual migraine at 4.1% versus 1%. In addition, women who decided to not have children because of migraine were significantly younger at an average age of 37.5 versus 47.2 years.
The number of days with headache per 3-month interval was 53.9 among women who avoided pregnancy versus 42.5 among the other women. The Migraine Disability Assessment score was also higher for women who avoided pregnancy (132.5) than for it was the other women (91.7), indicating more severe disability.
In addition, more of the women who avoided pregnancy had a history of depression (48.8%) compared with the other women (37.7%). The average score on the Patient Health Questionnaire–4 was higher among women who avoided pregnancy (4.0) than among other women (3.1), which indicates greater anxiety or depression. Among women who avoided pregnancy, 72.5% believed their migraine would worsen during pregnancy, and 68.3% believed that migraine would make pregnancy very difficult.
“Clinicians need to recognize that migraine often has a substantial burden on multiple aspects of life, including one’s plans for having children,” Dr. Ishii said.
“Clinicians should educate their patients who are considering pregnancy about the most likely course of migraine during pregnancy, migraine treatment during pregnancy, and the potential impacts of migraine and its treatment on pregnancy outcomes,” he added.
More education needed
Commenting on the study, Susan Hutchinson, MD, director of the Orange County Migraine and Headache Center, Irvine, California, said that not knowing how pregnancy is going to affect patients’ migraines can be “very scary” for women. In addition, patients often wonder what migraine treatments they can safely take once they do become pregnant, said Dr. Hutchinson, who was not involved in the research.
She noted that advantages of the ARMR data are that they are derived from a multicenter study and that migraine diagnoses were made by a headache specialist. A potential limitation of the study is that the population may not reflect outcomes of the millions of women who have migraine and become pregnant but never see a specialist.
“These findings show that more education is needed,” Dr. Hutchinson said.
Most women, especially those who have migraine without aura, note improvement with migraine during pregnancy, primarily because of the high, steady levels of estradiol, especially in the second and third trimesters, she said. In light of this, neurologists should reassure women that migraine is not a contraindication to pregnancy, she added.
There is also a need for additional research to assess how past experience with migraine and pregnancy influences a woman’s comfort level with additional pregnancies. Studies as to which treatments are safest for acute and preventive treatment of migraine during prepregnancy, pregnancy, and lactation are also needed, Dr. Hutchinson noted.
“If women knew they had treatment options that were evidence-based, they might be much more comfortable contemplating a pregnancy,” she said.
Dr. Ishii and Dr. Hutchinson have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research shows. Results from a multicenter study of more than 600 women showed that, among participants with migraine, those who were younger, had menstrual migraine, or had chronic migraine were more likely to decide to not become pregnant.
Although women with migraine who avoided pregnancy believed their migraines would worsen during pregnancy or make their pregnancy difficult, previous observational research indicates that migraine often improves during pregnancy.
“Women who avoided pregnancy due to migraine were most concerned that migraine would make raising a child difficult, that the migraine medications they take would have a negative impact on their child’s development, and that their migraine pattern would worsen during or just after pregnancy,” said study investigator Ryotaro Ishii, MD, PhD, a visiting scientist at Mayo Clinic in Phoenix, Arizona.
The findings were presented at the virtual annual meeting of the American Headache Society.
Plans for the future
There is a paucity of research on the effects of migraine on pregnancy planning, the researchers noted. The few studies that have investigated this issue have focused on women’s previous family planning decisions and experience rather than on plans for the future, the researchers noted.
To evaluate how migraine in women influences pregnancy planning, the investigators analyzed data from the American Registry for Migraine Research (ARMR). The registry, which was established by the American Migraine Foundation, collects clinical data about individuals with migraine and other headache disorders from multiple centers.
Participants eligible for the current analysis were women who had been diagnosed with migraine on the basis of the International Classification of Headache Disorders–3 criteria. All completed the ARMR questionnaire between February 2016 and September 2019. The investigators excluded patients with trigeminal autonomic cephalalgia, secondary headache, painful cranial neuropathies, other facial pain, and other headaches.
They identified 895 eligible women with migraine. Of these, 607 completed the pregnancy question. Among those participants, 121 women (19.9%) reported that migraine was a factor in their decision to not become pregnant. Of this group, 70 (11.5%) reported that migraine was a “significant” factor in deciding to not have children, and 8.4% said it was “somewhat” of a factor. The remainder of the cohort (479) reported that migraine had no influence on their pregnancy plans.
There were no between-group differences by race, marital status, employment, or income. This finding suggests that sociodemographic differences “have less impact on pregnancy planning than migraine-specific characteristics like headache frequency and experience with having migraine attacks triggered by menstruation,” Dr. Ishii said.
“Substantial burden”
Not surprisingly, women who avoided pregnancy had fewer children than the rest of the sample. About 60% of those who made the decision to not become pregnant had no children, and 72% had not been pregnant since they began experiencing migraine.
Compared with women who reported that migraine had no influence on their pregnancy plans, those who avoided pregnancy were more likely to have chronic migraine at 81.8% versus 70.2%. They were also more likely to have menstrual migraine at 4.1% versus 1%. In addition, women who decided to not have children because of migraine were significantly younger at an average age of 37.5 versus 47.2 years.
The number of days with headache per 3-month interval was 53.9 among women who avoided pregnancy versus 42.5 among the other women. The Migraine Disability Assessment score was also higher for women who avoided pregnancy (132.5) than for it was the other women (91.7), indicating more severe disability.
In addition, more of the women who avoided pregnancy had a history of depression (48.8%) compared with the other women (37.7%). The average score on the Patient Health Questionnaire–4 was higher among women who avoided pregnancy (4.0) than among other women (3.1), which indicates greater anxiety or depression. Among women who avoided pregnancy, 72.5% believed their migraine would worsen during pregnancy, and 68.3% believed that migraine would make pregnancy very difficult.
“Clinicians need to recognize that migraine often has a substantial burden on multiple aspects of life, including one’s plans for having children,” Dr. Ishii said.
“Clinicians should educate their patients who are considering pregnancy about the most likely course of migraine during pregnancy, migraine treatment during pregnancy, and the potential impacts of migraine and its treatment on pregnancy outcomes,” he added.
More education needed
Commenting on the study, Susan Hutchinson, MD, director of the Orange County Migraine and Headache Center, Irvine, California, said that not knowing how pregnancy is going to affect patients’ migraines can be “very scary” for women. In addition, patients often wonder what migraine treatments they can safely take once they do become pregnant, said Dr. Hutchinson, who was not involved in the research.
She noted that advantages of the ARMR data are that they are derived from a multicenter study and that migraine diagnoses were made by a headache specialist. A potential limitation of the study is that the population may not reflect outcomes of the millions of women who have migraine and become pregnant but never see a specialist.
“These findings show that more education is needed,” Dr. Hutchinson said.
Most women, especially those who have migraine without aura, note improvement with migraine during pregnancy, primarily because of the high, steady levels of estradiol, especially in the second and third trimesters, she said. In light of this, neurologists should reassure women that migraine is not a contraindication to pregnancy, she added.
There is also a need for additional research to assess how past experience with migraine and pregnancy influences a woman’s comfort level with additional pregnancies. Studies as to which treatments are safest for acute and preventive treatment of migraine during prepregnancy, pregnancy, and lactation are also needed, Dr. Hutchinson noted.
“If women knew they had treatment options that were evidence-based, they might be much more comfortable contemplating a pregnancy,” she said.
Dr. Ishii and Dr. Hutchinson have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AHS 2020
Visualization tool aids migraine management
The tool is still in the prototype stage, but it could eventually synthesize patient responses to an integrated questionnaire and produce a chart illustrating where the patient stands with respect to a range of modifiable risk factors, including depression, medication overuse, insomnia, and body mass index, among others.
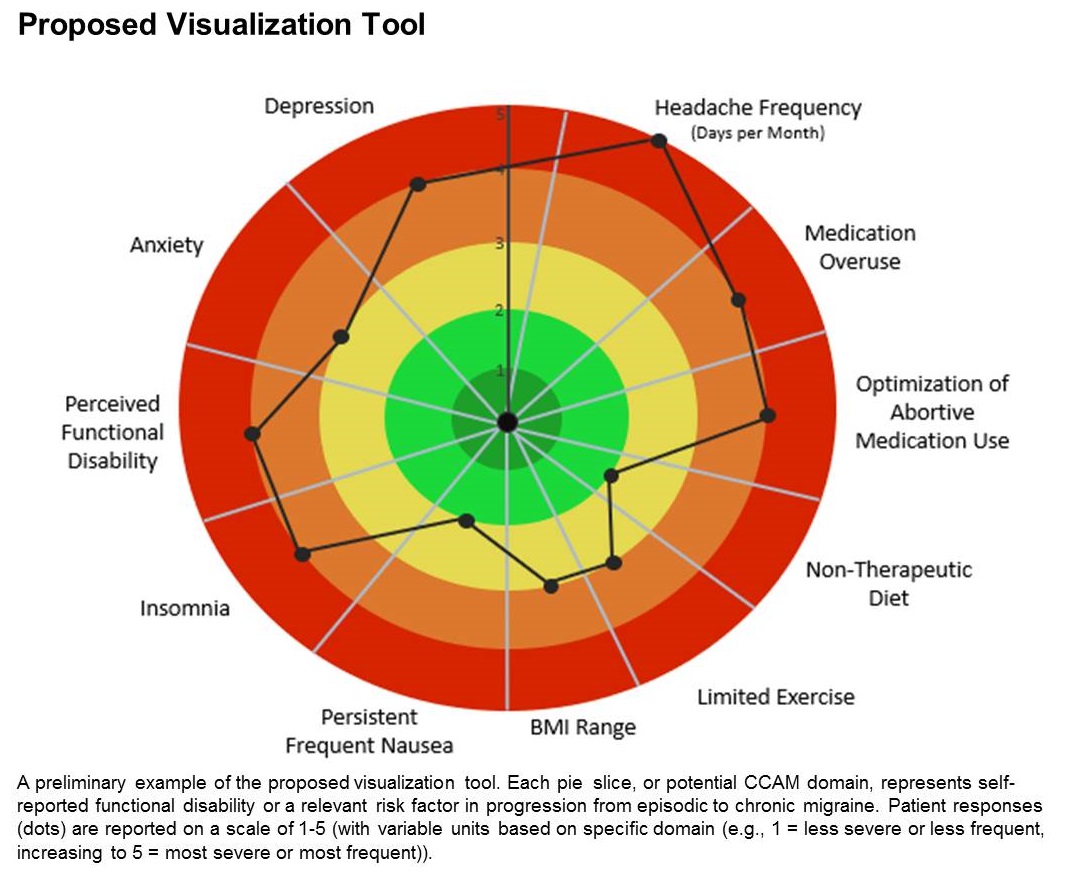
A few such tools exist for other conditions, such as stroke and risk of developing chronic diseases. Existing migraine visualization models focus only on individual risk factors, but they are capable of much more. “Visualization tools can effectively communicate a huge amount of clinical information,” said lead author Ami Cuneo, MD, who is a headache fellow at the University of Washington, Seattle, in an interview. Dr. Cuneo presented a poster describing the concept at the virtual annual meeting of the American Headache Society.
A picture is worth a thousand words
Dr. Cuneo’s background is well suited to the effort: Before entering medicine, she was a documentary producer. “I have a lot of interest in the patient story and history,” she added. She also believes that the tool could improve patient-provider relationships. In rushed sessions, patients may not feel heard. Patients gain a therapeutic benefit from the belief that their provider is listening to them and listening to their story. Visualization tools could promote that if the provider can quickly identify key elements of the patient’s condition. “A lot of headache patients can have a complex picture,” said Dr. Cuneo.
Physicians must see patients in short appointment periods, making it difficult to communicate all of the risk factors and behavioral characteristics that can contribute to risk of progression. “If you have a patient and you’re able to look at a visualization tool quickly and say: ‘Okay, my patient really is having insomnia and sleep issues,’ you can focus the session talking about sleep, cognitive-behavioral therapy for insomnia, and all the things we can help patients with,” said Dr. Cuneo.
The prototype visualization tool uses a color-coded wheel divided into pie slices, each representing a clinical characteristic or modifiable risk factor. In the proposed tool presented in the poster, these included depression, anxiety, functional disability, insomnia, nausea, headache frequency, medication overuse, optimization of abortive medication use, nontherapeutic diet, limited exercise, and body mass index range. The circle also contains colored concentric circles, ranging from red to green, and a small filled circle represents the patient’s status in each category as ranked using the integrated questionnaire. A line connects the circles in each pie, revealing the patient’s overall status.
The visual cue allows both the physician and patient to quickly assess these factors and see them in relationship to one another. Verbally communicating each factor is time consuming and harder for the patient to take in, according to Dr. Cuneo. “The provider can just look at it and see the areas to focus questions on to try to improve care. So it’s a way I’m hopeful that we can help target visits and improve patient-provider communication without extending visit time.”
A key challenge for the project will be choosing and consolidating scales so that the patient isn’t burdened with too many questions in advance of the appointment. The team will draw from existing scales and then create their own and validate it. “The questions will have to be vetted with patients through focus groups, and then the software platform [will have to be developed] so that patients can complete the survey online. Then we have to test it to see if providers and patients feel this is something that’s helpful in the clinical practice,” said Dr. Cuneo.
Will it change behavior?
If successful, the tool would be a welcome addition, according to Andrew Charles, MD, who was asked to comment on the work. “Epidemiological studies have identified these risk factors, but we haven’t had a way of operationalizing a strategy to reduce them systematically, so having some sort of tool that visualizes not just one but multiple risk factors is something I think could be helpful to address those factors more aggressively. The real question would be, if you put it in the hands of practitioners and patients, will they really be able to easily implement it and will it change behavior,” said Dr. Charles, who is a professor of neurology and director of the Goldberg Migraine Program at the University of California, Los Angeles.
The study received no funding. Dr. Cuneo and Dr. Charles have no relevant financial disclosures.
SOURCE; Cuneo A et al. AHS 2020, Abstract 273715.
The tool is still in the prototype stage, but it could eventually synthesize patient responses to an integrated questionnaire and produce a chart illustrating where the patient stands with respect to a range of modifiable risk factors, including depression, medication overuse, insomnia, and body mass index, among others.

A few such tools exist for other conditions, such as stroke and risk of developing chronic diseases. Existing migraine visualization models focus only on individual risk factors, but they are capable of much more. “Visualization tools can effectively communicate a huge amount of clinical information,” said lead author Ami Cuneo, MD, who is a headache fellow at the University of Washington, Seattle, in an interview. Dr. Cuneo presented a poster describing the concept at the virtual annual meeting of the American Headache Society.
A picture is worth a thousand words
Dr. Cuneo’s background is well suited to the effort: Before entering medicine, she was a documentary producer. “I have a lot of interest in the patient story and history,” she added. She also believes that the tool could improve patient-provider relationships. In rushed sessions, patients may not feel heard. Patients gain a therapeutic benefit from the belief that their provider is listening to them and listening to their story. Visualization tools could promote that if the provider can quickly identify key elements of the patient’s condition. “A lot of headache patients can have a complex picture,” said Dr. Cuneo.
Physicians must see patients in short appointment periods, making it difficult to communicate all of the risk factors and behavioral characteristics that can contribute to risk of progression. “If you have a patient and you’re able to look at a visualization tool quickly and say: ‘Okay, my patient really is having insomnia and sleep issues,’ you can focus the session talking about sleep, cognitive-behavioral therapy for insomnia, and all the things we can help patients with,” said Dr. Cuneo.
The prototype visualization tool uses a color-coded wheel divided into pie slices, each representing a clinical characteristic or modifiable risk factor. In the proposed tool presented in the poster, these included depression, anxiety, functional disability, insomnia, nausea, headache frequency, medication overuse, optimization of abortive medication use, nontherapeutic diet, limited exercise, and body mass index range. The circle also contains colored concentric circles, ranging from red to green, and a small filled circle represents the patient’s status in each category as ranked using the integrated questionnaire. A line connects the circles in each pie, revealing the patient’s overall status.
The visual cue allows both the physician and patient to quickly assess these factors and see them in relationship to one another. Verbally communicating each factor is time consuming and harder for the patient to take in, according to Dr. Cuneo. “The provider can just look at it and see the areas to focus questions on to try to improve care. So it’s a way I’m hopeful that we can help target visits and improve patient-provider communication without extending visit time.”
A key challenge for the project will be choosing and consolidating scales so that the patient isn’t burdened with too many questions in advance of the appointment. The team will draw from existing scales and then create their own and validate it. “The questions will have to be vetted with patients through focus groups, and then the software platform [will have to be developed] so that patients can complete the survey online. Then we have to test it to see if providers and patients feel this is something that’s helpful in the clinical practice,” said Dr. Cuneo.
Will it change behavior?
If successful, the tool would be a welcome addition, according to Andrew Charles, MD, who was asked to comment on the work. “Epidemiological studies have identified these risk factors, but we haven’t had a way of operationalizing a strategy to reduce them systematically, so having some sort of tool that visualizes not just one but multiple risk factors is something I think could be helpful to address those factors more aggressively. The real question would be, if you put it in the hands of practitioners and patients, will they really be able to easily implement it and will it change behavior,” said Dr. Charles, who is a professor of neurology and director of the Goldberg Migraine Program at the University of California, Los Angeles.
The study received no funding. Dr. Cuneo and Dr. Charles have no relevant financial disclosures.
SOURCE; Cuneo A et al. AHS 2020, Abstract 273715.
The tool is still in the prototype stage, but it could eventually synthesize patient responses to an integrated questionnaire and produce a chart illustrating where the patient stands with respect to a range of modifiable risk factors, including depression, medication overuse, insomnia, and body mass index, among others.

A few such tools exist for other conditions, such as stroke and risk of developing chronic diseases. Existing migraine visualization models focus only on individual risk factors, but they are capable of much more. “Visualization tools can effectively communicate a huge amount of clinical information,” said lead author Ami Cuneo, MD, who is a headache fellow at the University of Washington, Seattle, in an interview. Dr. Cuneo presented a poster describing the concept at the virtual annual meeting of the American Headache Society.
A picture is worth a thousand words
Dr. Cuneo’s background is well suited to the effort: Before entering medicine, she was a documentary producer. “I have a lot of interest in the patient story and history,” she added. She also believes that the tool could improve patient-provider relationships. In rushed sessions, patients may not feel heard. Patients gain a therapeutic benefit from the belief that their provider is listening to them and listening to their story. Visualization tools could promote that if the provider can quickly identify key elements of the patient’s condition. “A lot of headache patients can have a complex picture,” said Dr. Cuneo.
Physicians must see patients in short appointment periods, making it difficult to communicate all of the risk factors and behavioral characteristics that can contribute to risk of progression. “If you have a patient and you’re able to look at a visualization tool quickly and say: ‘Okay, my patient really is having insomnia and sleep issues,’ you can focus the session talking about sleep, cognitive-behavioral therapy for insomnia, and all the things we can help patients with,” said Dr. Cuneo.
The prototype visualization tool uses a color-coded wheel divided into pie slices, each representing a clinical characteristic or modifiable risk factor. In the proposed tool presented in the poster, these included depression, anxiety, functional disability, insomnia, nausea, headache frequency, medication overuse, optimization of abortive medication use, nontherapeutic diet, limited exercise, and body mass index range. The circle also contains colored concentric circles, ranging from red to green, and a small filled circle represents the patient’s status in each category as ranked using the integrated questionnaire. A line connects the circles in each pie, revealing the patient’s overall status.
The visual cue allows both the physician and patient to quickly assess these factors and see them in relationship to one another. Verbally communicating each factor is time consuming and harder for the patient to take in, according to Dr. Cuneo. “The provider can just look at it and see the areas to focus questions on to try to improve care. So it’s a way I’m hopeful that we can help target visits and improve patient-provider communication without extending visit time.”
A key challenge for the project will be choosing and consolidating scales so that the patient isn’t burdened with too many questions in advance of the appointment. The team will draw from existing scales and then create their own and validate it. “The questions will have to be vetted with patients through focus groups, and then the software platform [will have to be developed] so that patients can complete the survey online. Then we have to test it to see if providers and patients feel this is something that’s helpful in the clinical practice,” said Dr. Cuneo.
Will it change behavior?
If successful, the tool would be a welcome addition, according to Andrew Charles, MD, who was asked to comment on the work. “Epidemiological studies have identified these risk factors, but we haven’t had a way of operationalizing a strategy to reduce them systematically, so having some sort of tool that visualizes not just one but multiple risk factors is something I think could be helpful to address those factors more aggressively. The real question would be, if you put it in the hands of practitioners and patients, will they really be able to easily implement it and will it change behavior,” said Dr. Charles, who is a professor of neurology and director of the Goldberg Migraine Program at the University of California, Los Angeles.
The study received no funding. Dr. Cuneo and Dr. Charles have no relevant financial disclosures.
SOURCE; Cuneo A et al. AHS 2020, Abstract 273715.
FROM AHS 2020
Consistent effects for galcanezumab in cluster headache
new research suggests. A post hoc analysis of patients from the phase 3 CGAL study who also entered the open-label CGAR extension study was conducted. Results showed that the majority of participants whose scores on the Patient Global Impression of Improvement (PGI-I) showed improvement 1 month after the initial dose of galcanezumab in the CGAL study also showed improvement after treatment for subsequent cluster bouts during the CGAR study.
“There was good agreement between PGI-I between the two [cluster headache] periods,” noted the investigators, led by Brian Plato, DO, a neurologist at Norton Neuroscience Institute in Louisville, Ky.
The findings were presented at the virtual annual meeting of the American Headache Society.
Two cluster periods
Galcanezumab was approved by the Food and Drug Administration in 2019 for the treatment of episodic cluster headache in adults.
In cluster headache, attacks of recurrent, unilateral headaches with cranial autonomic symptoms last for weeks or months and are followed by periods of remission. Most studies of therapies for cluster headache examine only one cluster period. Few data about the consistency of treatment response throughout consecutive cluster periods are available, the investigators noted.
The current analysis was undertaken to examine the consistency of galcanezumab’s effect in episodic cluster headache during two cluster periods. Patients eligible for inclusion in the analysis had completed the double-blind phase of the CGAL study and had entered the open-label CGAR study.
CGAL was a phase 3, multicenter, randomized, double-blind study in which patients with episodic cluster headache were assigned to receive galcanezumab 300 mg per month or placebo. Patients who completed the double-blind and washout phases of this study were eligible for enrollment into CGAR, a phase 3b, single-arm safety study. The investigators determined the dose of galcanezumab in accordance with each patient’s symptoms and clinical response.
Response agreement
In both studies, the PGI-I was administered 1 month after the initial dose of galcanezumab. Only patients who were in an active cluster bout on entry into CGAR and who had valid PGI-I results 1 month after the first dose in CGAL and CGAR were included in the analysis.
PGI-I responses ranged from 1, signifying very much better, to 7, signifying very much worse. The investigators summarized the proportions of patients who reported each level of PGI-I score in CGAR and analyzed the results by dichotomizing PGI-I scores at both time points in two ways.
Fifty patients entered CGAR (78% men; mean age, 46.8 years). Of this group, Dr. Plato and colleagues included 39 in their analysis. Of the 17 patients who had a PGI-I score of 1 or 2 in CGAL, 12 (70.6%) had a score in the same range in CGAR. All four participants who had a score of 3 or higher in CGAL had a score in the same range in CGAR. Eighteen participants had a PGI-I score of 1, 2, or 3 in CGAL. Of this group, 15 patients (83.3%) had a score in the same range in CGAR. Of the three patients who had a score above 3 in CGAL, two (66.7%) had a score in the same range in CGAR.
The results indicate that most patients whose PGI-I score improved in one cluster bout, such as in CGAL, also improved in a subsequent bout, such as in CGAR, the investigators noted.
‘Encouraging’ results
Commenting on the study, Brian E. McGeeney, MD, a neurologist at the John R. Graham Headache Center, Brigham and Women’s Faulkner Hospital, Boston, noted that the PGI-I is an “easy-to-understand” outcome that has been widely used in headache medicine.
“Patient-assessed outcomes have become increasingly important and are an important complement to other outcomes,” said Dr. McGeeney, who was not involved in the research. However, a disadvantage is that “it is entirely subjective and may or may not reflect a change on other outcome measures that reflect the disorder itself,” he said.
“It can be difficult to demonstrate how much usefulness a treatment has with the helpful but simple outcome measures that are seen in CGAL and CGAR,” Dr. McGeeney added. “This is due to the nature of cluster headache and not to any methodological shortcomings of those studies.”
He said this is a core problem in general with cluster headache studies, “of which there are very few.”
In addition, CGAR only included episodic cluster headache, and the study period was relatively short; and CGAL only explored one cluster period per patient, Dr. McGeeney noted.
The current research attempts to provide insight that was previously unavailable, he said. “Many headache medicine clinical trial results reflect only one episode, and in general, we infer repeated usefulness – although it is not demonstrated in clinical trials,” said Dr. McGeeney.
“In this recent presentation, the authors attempt to go further and demonstrate some consistency across multiple cluster periods. The results are encouraging and what one might expect,” he said. However, “the small numbers and ad hoc nature preclude much inference from this study alone.”
Dr. Plato has received honoraria for speaking from Allergan, Amgen/Novartis, and Eli Lilly. He has also received research grants and support from Electrocore and Teva. Dr. McGeeney has consulted for Upsher-Smith and Theranica.
A version of this article originally appeared on Medscape.com.
new research suggests. A post hoc analysis of patients from the phase 3 CGAL study who also entered the open-label CGAR extension study was conducted. Results showed that the majority of participants whose scores on the Patient Global Impression of Improvement (PGI-I) showed improvement 1 month after the initial dose of galcanezumab in the CGAL study also showed improvement after treatment for subsequent cluster bouts during the CGAR study.
“There was good agreement between PGI-I between the two [cluster headache] periods,” noted the investigators, led by Brian Plato, DO, a neurologist at Norton Neuroscience Institute in Louisville, Ky.
The findings were presented at the virtual annual meeting of the American Headache Society.
Two cluster periods
Galcanezumab was approved by the Food and Drug Administration in 2019 for the treatment of episodic cluster headache in adults.
In cluster headache, attacks of recurrent, unilateral headaches with cranial autonomic symptoms last for weeks or months and are followed by periods of remission. Most studies of therapies for cluster headache examine only one cluster period. Few data about the consistency of treatment response throughout consecutive cluster periods are available, the investigators noted.
The current analysis was undertaken to examine the consistency of galcanezumab’s effect in episodic cluster headache during two cluster periods. Patients eligible for inclusion in the analysis had completed the double-blind phase of the CGAL study and had entered the open-label CGAR study.
CGAL was a phase 3, multicenter, randomized, double-blind study in which patients with episodic cluster headache were assigned to receive galcanezumab 300 mg per month or placebo. Patients who completed the double-blind and washout phases of this study were eligible for enrollment into CGAR, a phase 3b, single-arm safety study. The investigators determined the dose of galcanezumab in accordance with each patient’s symptoms and clinical response.
Response agreement
In both studies, the PGI-I was administered 1 month after the initial dose of galcanezumab. Only patients who were in an active cluster bout on entry into CGAR and who had valid PGI-I results 1 month after the first dose in CGAL and CGAR were included in the analysis.
PGI-I responses ranged from 1, signifying very much better, to 7, signifying very much worse. The investigators summarized the proportions of patients who reported each level of PGI-I score in CGAR and analyzed the results by dichotomizing PGI-I scores at both time points in two ways.
Fifty patients entered CGAR (78% men; mean age, 46.8 years). Of this group, Dr. Plato and colleagues included 39 in their analysis. Of the 17 patients who had a PGI-I score of 1 or 2 in CGAL, 12 (70.6%) had a score in the same range in CGAR. All four participants who had a score of 3 or higher in CGAL had a score in the same range in CGAR. Eighteen participants had a PGI-I score of 1, 2, or 3 in CGAL. Of this group, 15 patients (83.3%) had a score in the same range in CGAR. Of the three patients who had a score above 3 in CGAL, two (66.7%) had a score in the same range in CGAR.
The results indicate that most patients whose PGI-I score improved in one cluster bout, such as in CGAL, also improved in a subsequent bout, such as in CGAR, the investigators noted.
‘Encouraging’ results
Commenting on the study, Brian E. McGeeney, MD, a neurologist at the John R. Graham Headache Center, Brigham and Women’s Faulkner Hospital, Boston, noted that the PGI-I is an “easy-to-understand” outcome that has been widely used in headache medicine.
“Patient-assessed outcomes have become increasingly important and are an important complement to other outcomes,” said Dr. McGeeney, who was not involved in the research. However, a disadvantage is that “it is entirely subjective and may or may not reflect a change on other outcome measures that reflect the disorder itself,” he said.
“It can be difficult to demonstrate how much usefulness a treatment has with the helpful but simple outcome measures that are seen in CGAL and CGAR,” Dr. McGeeney added. “This is due to the nature of cluster headache and not to any methodological shortcomings of those studies.”
He said this is a core problem in general with cluster headache studies, “of which there are very few.”
In addition, CGAR only included episodic cluster headache, and the study period was relatively short; and CGAL only explored one cluster period per patient, Dr. McGeeney noted.
The current research attempts to provide insight that was previously unavailable, he said. “Many headache medicine clinical trial results reflect only one episode, and in general, we infer repeated usefulness – although it is not demonstrated in clinical trials,” said Dr. McGeeney.
“In this recent presentation, the authors attempt to go further and demonstrate some consistency across multiple cluster periods. The results are encouraging and what one might expect,” he said. However, “the small numbers and ad hoc nature preclude much inference from this study alone.”
Dr. Plato has received honoraria for speaking from Allergan, Amgen/Novartis, and Eli Lilly. He has also received research grants and support from Electrocore and Teva. Dr. McGeeney has consulted for Upsher-Smith and Theranica.
A version of this article originally appeared on Medscape.com.
new research suggests. A post hoc analysis of patients from the phase 3 CGAL study who also entered the open-label CGAR extension study was conducted. Results showed that the majority of participants whose scores on the Patient Global Impression of Improvement (PGI-I) showed improvement 1 month after the initial dose of galcanezumab in the CGAL study also showed improvement after treatment for subsequent cluster bouts during the CGAR study.
“There was good agreement between PGI-I between the two [cluster headache] periods,” noted the investigators, led by Brian Plato, DO, a neurologist at Norton Neuroscience Institute in Louisville, Ky.
The findings were presented at the virtual annual meeting of the American Headache Society.
Two cluster periods
Galcanezumab was approved by the Food and Drug Administration in 2019 for the treatment of episodic cluster headache in adults.
In cluster headache, attacks of recurrent, unilateral headaches with cranial autonomic symptoms last for weeks or months and are followed by periods of remission. Most studies of therapies for cluster headache examine only one cluster period. Few data about the consistency of treatment response throughout consecutive cluster periods are available, the investigators noted.
The current analysis was undertaken to examine the consistency of galcanezumab’s effect in episodic cluster headache during two cluster periods. Patients eligible for inclusion in the analysis had completed the double-blind phase of the CGAL study and had entered the open-label CGAR study.
CGAL was a phase 3, multicenter, randomized, double-blind study in which patients with episodic cluster headache were assigned to receive galcanezumab 300 mg per month or placebo. Patients who completed the double-blind and washout phases of this study were eligible for enrollment into CGAR, a phase 3b, single-arm safety study. The investigators determined the dose of galcanezumab in accordance with each patient’s symptoms and clinical response.
Response agreement
In both studies, the PGI-I was administered 1 month after the initial dose of galcanezumab. Only patients who were in an active cluster bout on entry into CGAR and who had valid PGI-I results 1 month after the first dose in CGAL and CGAR were included in the analysis.
PGI-I responses ranged from 1, signifying very much better, to 7, signifying very much worse. The investigators summarized the proportions of patients who reported each level of PGI-I score in CGAR and analyzed the results by dichotomizing PGI-I scores at both time points in two ways.
Fifty patients entered CGAR (78% men; mean age, 46.8 years). Of this group, Dr. Plato and colleagues included 39 in their analysis. Of the 17 patients who had a PGI-I score of 1 or 2 in CGAL, 12 (70.6%) had a score in the same range in CGAR. All four participants who had a score of 3 or higher in CGAL had a score in the same range in CGAR. Eighteen participants had a PGI-I score of 1, 2, or 3 in CGAL. Of this group, 15 patients (83.3%) had a score in the same range in CGAR. Of the three patients who had a score above 3 in CGAL, two (66.7%) had a score in the same range in CGAR.
The results indicate that most patients whose PGI-I score improved in one cluster bout, such as in CGAL, also improved in a subsequent bout, such as in CGAR, the investigators noted.
‘Encouraging’ results
Commenting on the study, Brian E. McGeeney, MD, a neurologist at the John R. Graham Headache Center, Brigham and Women’s Faulkner Hospital, Boston, noted that the PGI-I is an “easy-to-understand” outcome that has been widely used in headache medicine.
“Patient-assessed outcomes have become increasingly important and are an important complement to other outcomes,” said Dr. McGeeney, who was not involved in the research. However, a disadvantage is that “it is entirely subjective and may or may not reflect a change on other outcome measures that reflect the disorder itself,” he said.
“It can be difficult to demonstrate how much usefulness a treatment has with the helpful but simple outcome measures that are seen in CGAL and CGAR,” Dr. McGeeney added. “This is due to the nature of cluster headache and not to any methodological shortcomings of those studies.”
He said this is a core problem in general with cluster headache studies, “of which there are very few.”
In addition, CGAR only included episodic cluster headache, and the study period was relatively short; and CGAL only explored one cluster period per patient, Dr. McGeeney noted.
The current research attempts to provide insight that was previously unavailable, he said. “Many headache medicine clinical trial results reflect only one episode, and in general, we infer repeated usefulness – although it is not demonstrated in clinical trials,” said Dr. McGeeney.
“In this recent presentation, the authors attempt to go further and demonstrate some consistency across multiple cluster periods. The results are encouraging and what one might expect,” he said. However, “the small numbers and ad hoc nature preclude much inference from this study alone.”
Dr. Plato has received honoraria for speaking from Allergan, Amgen/Novartis, and Eli Lilly. He has also received research grants and support from Electrocore and Teva. Dr. McGeeney has consulted for Upsher-Smith and Theranica.
A version of this article originally appeared on Medscape.com.
FROM AHS 2020




