User login
Poor-prognosis cancers linked to highest suicide risk in first year
Suicide risk significantly increases within the first year of a cancer diagnosis, with risk varying by type of cancer, according to investigators who conducted a retrospective analysis representing nearly 4.7 million patients.
Risk of suicide in that first year after diagnosis was especially high in pancreatic and lung cancers, while by contrast, breast and prostate cancer did not increase suicide risk, reported the researchers, led by Hesham Hamoda, MD, MPH, of Boston Children’s Hospital/Harvard Medical School, and Ahmad Alfaar, MBBCh, MSc, of Charité–Universitätsmedizin Berlin.
That variation in suicide risk by cancer type suggests that prognosis and 5-year relative survival play a role in increasing suicide rates, according to Dr. Hamoda, Dr. Alfaar, and their coauthors.
“After the diagnosis, it is important that health care providers be vigilant in screening for suicide and ensuring that patients have access to social and emotional support,” they wrote in a report published in Cancer. Their analysis was based on 4,671,989 patients with a diagnosis of cancer in the Surveillance, Epidemiology, and End Results (SEER) database between 2000 and 2014. Out of 1,005,825 of those patients who died within the first year of diagnosis, the cause of death was suicide for 1,585, or 0.16%.
Overall, the risk of suicide increased significantly among cancer patients versus the general population, with an observed-to-expected (O/E) ratio of 2.51 per 10,000 person-years, the investigators found. The risk was highest in the first 6 months, with an O/E mortality of 3.13 versus 1.8 in the latter 6 months.
The highest ratios were seen for pancreatic cancer, with an O/E ratio of 8.01, and lung cancer, with a ratio of 6.05, the researchers found in further analysis.
Significant increases in suicide risk were also seen for colorectal cancer (2.08) and melanoma (1.45), though rates were not significantly different versus the general population for breast (1.23) and prostate (0.99), according to the reported data.
Suicide risk was relatively high for any cancer with distant metastases (5.63), though still significantly higher at 1.65 in persons with localized/regional disease, the data show.
The increased suicide risk persisted more than 1 year after the cancer diagnosis, though not to the degree observed within that first year, they added.
Most patients with suicide as a cause of death were white (90.2%) and male (87%). Nearly 60% were between the ages of 65 and 84 at the time of suicide.
Social support plays an integral role in suicide prevention among cancer patients, the researchers noted.
Previous studies suggest that support programs may decrease suicide risk by making patients better aware of their prognosis, receptive to decreased social stigma, or less likely to have stress related to cost of care, they said.
“Discussing the quality of life after diagnosis, the effectiveness of therapy, and the prognosis of the disease and maintaining a trusting relationship with health care professionals all decrease the likelihood of suicide immediately after a diagnosis of cancer,” they said.
Dr. Hamoda, Dr. Alfaar, and their coauthors reported no conflicts of interest. Funding for the study came in part from the German Academic Exchange Service (Dr. Alfaar).
SOURCE: Saad AM, et al. Cancer 2019 Jan 7. doi: 10.1002/cncr.31876.
Suicide risk significantly increases within the first year of a cancer diagnosis, with risk varying by type of cancer, according to investigators who conducted a retrospective analysis representing nearly 4.7 million patients.
Risk of suicide in that first year after diagnosis was especially high in pancreatic and lung cancers, while by contrast, breast and prostate cancer did not increase suicide risk, reported the researchers, led by Hesham Hamoda, MD, MPH, of Boston Children’s Hospital/Harvard Medical School, and Ahmad Alfaar, MBBCh, MSc, of Charité–Universitätsmedizin Berlin.
That variation in suicide risk by cancer type suggests that prognosis and 5-year relative survival play a role in increasing suicide rates, according to Dr. Hamoda, Dr. Alfaar, and their coauthors.
“After the diagnosis, it is important that health care providers be vigilant in screening for suicide and ensuring that patients have access to social and emotional support,” they wrote in a report published in Cancer. Their analysis was based on 4,671,989 patients with a diagnosis of cancer in the Surveillance, Epidemiology, and End Results (SEER) database between 2000 and 2014. Out of 1,005,825 of those patients who died within the first year of diagnosis, the cause of death was suicide for 1,585, or 0.16%.
Overall, the risk of suicide increased significantly among cancer patients versus the general population, with an observed-to-expected (O/E) ratio of 2.51 per 10,000 person-years, the investigators found. The risk was highest in the first 6 months, with an O/E mortality of 3.13 versus 1.8 in the latter 6 months.
The highest ratios were seen for pancreatic cancer, with an O/E ratio of 8.01, and lung cancer, with a ratio of 6.05, the researchers found in further analysis.
Significant increases in suicide risk were also seen for colorectal cancer (2.08) and melanoma (1.45), though rates were not significantly different versus the general population for breast (1.23) and prostate (0.99), according to the reported data.
Suicide risk was relatively high for any cancer with distant metastases (5.63), though still significantly higher at 1.65 in persons with localized/regional disease, the data show.
The increased suicide risk persisted more than 1 year after the cancer diagnosis, though not to the degree observed within that first year, they added.
Most patients with suicide as a cause of death were white (90.2%) and male (87%). Nearly 60% were between the ages of 65 and 84 at the time of suicide.
Social support plays an integral role in suicide prevention among cancer patients, the researchers noted.
Previous studies suggest that support programs may decrease suicide risk by making patients better aware of their prognosis, receptive to decreased social stigma, or less likely to have stress related to cost of care, they said.
“Discussing the quality of life after diagnosis, the effectiveness of therapy, and the prognosis of the disease and maintaining a trusting relationship with health care professionals all decrease the likelihood of suicide immediately after a diagnosis of cancer,” they said.
Dr. Hamoda, Dr. Alfaar, and their coauthors reported no conflicts of interest. Funding for the study came in part from the German Academic Exchange Service (Dr. Alfaar).
SOURCE: Saad AM, et al. Cancer 2019 Jan 7. doi: 10.1002/cncr.31876.
Suicide risk significantly increases within the first year of a cancer diagnosis, with risk varying by type of cancer, according to investigators who conducted a retrospective analysis representing nearly 4.7 million patients.
Risk of suicide in that first year after diagnosis was especially high in pancreatic and lung cancers, while by contrast, breast and prostate cancer did not increase suicide risk, reported the researchers, led by Hesham Hamoda, MD, MPH, of Boston Children’s Hospital/Harvard Medical School, and Ahmad Alfaar, MBBCh, MSc, of Charité–Universitätsmedizin Berlin.
That variation in suicide risk by cancer type suggests that prognosis and 5-year relative survival play a role in increasing suicide rates, according to Dr. Hamoda, Dr. Alfaar, and their coauthors.
“After the diagnosis, it is important that health care providers be vigilant in screening for suicide and ensuring that patients have access to social and emotional support,” they wrote in a report published in Cancer. Their analysis was based on 4,671,989 patients with a diagnosis of cancer in the Surveillance, Epidemiology, and End Results (SEER) database between 2000 and 2014. Out of 1,005,825 of those patients who died within the first year of diagnosis, the cause of death was suicide for 1,585, or 0.16%.
Overall, the risk of suicide increased significantly among cancer patients versus the general population, with an observed-to-expected (O/E) ratio of 2.51 per 10,000 person-years, the investigators found. The risk was highest in the first 6 months, with an O/E mortality of 3.13 versus 1.8 in the latter 6 months.
The highest ratios were seen for pancreatic cancer, with an O/E ratio of 8.01, and lung cancer, with a ratio of 6.05, the researchers found in further analysis.
Significant increases in suicide risk were also seen for colorectal cancer (2.08) and melanoma (1.45), though rates were not significantly different versus the general population for breast (1.23) and prostate (0.99), according to the reported data.
Suicide risk was relatively high for any cancer with distant metastases (5.63), though still significantly higher at 1.65 in persons with localized/regional disease, the data show.
The increased suicide risk persisted more than 1 year after the cancer diagnosis, though not to the degree observed within that first year, they added.
Most patients with suicide as a cause of death were white (90.2%) and male (87%). Nearly 60% were between the ages of 65 and 84 at the time of suicide.
Social support plays an integral role in suicide prevention among cancer patients, the researchers noted.
Previous studies suggest that support programs may decrease suicide risk by making patients better aware of their prognosis, receptive to decreased social stigma, or less likely to have stress related to cost of care, they said.
“Discussing the quality of life after diagnosis, the effectiveness of therapy, and the prognosis of the disease and maintaining a trusting relationship with health care professionals all decrease the likelihood of suicide immediately after a diagnosis of cancer,” they said.
Dr. Hamoda, Dr. Alfaar, and their coauthors reported no conflicts of interest. Funding for the study came in part from the German Academic Exchange Service (Dr. Alfaar).
SOURCE: Saad AM, et al. Cancer 2019 Jan 7. doi: 10.1002/cncr.31876.
FROM CANCER
Key clinical point: A cancer diagnosis significantly increases risk of suicide in comparison to the general population, particularly for poorer-prognosis cancers.
Major finding: The observed-to-expected mortality ratio was substantially higher for pancreatic cancer (8.01), and lung cancer (6.05), but not significantly increased for breast (1.23) and prostate (0.99).
Study details: A retrospective population-based study of 4,671,989 cancer patients.
Disclosures: The authors reported no conflicts of interest. Funding for the study came in part from the German Academic Exchange Service.
Source: Saad AM et al. Cancer. 2019 Jan 7. doi: 10.1002/cncr.31876.
mRNA-based urine test performs well in bladder cancer surveillance
An mRNA-based assay for surveillance of patients with bladder cancer outperformed standard urine tests on certain measures in a validation study, investigators report.
The mRNA-based urine test (Xpert, Cepheid) had better sensitivity and negative predictive value compared to urine cytology and to UroVysion fluorescence in situ hybridization (FISH) testing, according to F. Johannes P. van Valenberg, of Radboud University Medical Center, Nijmegen, the Netherlands, and his colleagues.
The reported results suggest this mRNA test could partially replace white-light cystoscopy, helping urologists maintain the recommended follow-up schedules for patients with a history of bladder cancer, Dr. van Valenberg and his coauthors wrote. The report is in European Urology.
“If used in the follow-up of non–muscle invasive bladder cancer, a cystoscopy might be waived if the Xpert result is negative,” they wrote.
The negative predictive value for high-grade disease was 98%, suggesting the mRNA-based test could help urologists avoid invasive cystoscopies for intermediate- to high-risk patients, which could reduce costs and patient discomfort, they added.
The prospective, 19-center validation study enrolled 363 individuals with a history of non–muscle invasive bladder cancer who were scheduled for a standard cystoscopy. Voided urine specimens from a total of 259 patients were evaluated with all three methods: the mRNA test, FISH testing, and cytology.
In a comparison of the tests, the mRNA test identified more recurrent cancers correctly, and was more sensitive in detecting low-grade tumors than was FISH (P less than .001) or cytology (P = .021), they reported.
The mRNA test was more sensitive for detection of the most common recurrent tumors, independent of grade.
The sensitivity, specificity, and negative predictive values for mRNA testing were 74%, 80%, and 93%, respectively. By comparison the sensitivity, specificity, and negative predictive values were 51%, 80%, and 88% for FISH and 30%, 90%, and 86% for cytology.
Looking at high-grade disease only, the sensitivity, specificity, and negative predictive values were 83.3%, 75.8%, and 97.6% for mRNA testing, 75.0%, 79.5%, and 96.6% for FISH testing, and 50.0%, 90.7%, and 94.2% for cytology.
Bacillus Calmette-Guérin treatment in the past 90 days did not influence results of the mRNA test, one additional analysis showed.
The Xpert mRNA assay evaluates five mRNA targets: ABL1, ANXA10, UPK1B, CRH, and IGF2, results of which are combined to classify samples as either negative or positive. The test has a “hands-on time” of less than 2 minutes and provides results in 90 minutes, according to Dr. van Valenberg and his coinvestigators.
“Cytology requires a review by a pathologist, is not performed on the same day as a clinic visit, and has associated inter- and intraobserver variability,” the researchers wrote.
Dr. van Valenberg reported no financial disclosures related to the study. Several study coauthors reported employment or financial disclosures related to Cepheid, which provided funding and support for the conduct of the study. Study coauthors provided additional disclosures related to MDxHealth, PHotocure, Bristol-Myers Squibb, Johnson & Johnson, Roche, Bayer, and Astellas, among others.
SOURCE: van Valenberg FJP et al. Eur Urol. 2018 Dec 12. doi: 10.1016/j.eururo.2018.11.055.
An mRNA-based assay for surveillance of patients with bladder cancer outperformed standard urine tests on certain measures in a validation study, investigators report.
The mRNA-based urine test (Xpert, Cepheid) had better sensitivity and negative predictive value compared to urine cytology and to UroVysion fluorescence in situ hybridization (FISH) testing, according to F. Johannes P. van Valenberg, of Radboud University Medical Center, Nijmegen, the Netherlands, and his colleagues.
The reported results suggest this mRNA test could partially replace white-light cystoscopy, helping urologists maintain the recommended follow-up schedules for patients with a history of bladder cancer, Dr. van Valenberg and his coauthors wrote. The report is in European Urology.
“If used in the follow-up of non–muscle invasive bladder cancer, a cystoscopy might be waived if the Xpert result is negative,” they wrote.
The negative predictive value for high-grade disease was 98%, suggesting the mRNA-based test could help urologists avoid invasive cystoscopies for intermediate- to high-risk patients, which could reduce costs and patient discomfort, they added.
The prospective, 19-center validation study enrolled 363 individuals with a history of non–muscle invasive bladder cancer who were scheduled for a standard cystoscopy. Voided urine specimens from a total of 259 patients were evaluated with all three methods: the mRNA test, FISH testing, and cytology.
In a comparison of the tests, the mRNA test identified more recurrent cancers correctly, and was more sensitive in detecting low-grade tumors than was FISH (P less than .001) or cytology (P = .021), they reported.
The mRNA test was more sensitive for detection of the most common recurrent tumors, independent of grade.
The sensitivity, specificity, and negative predictive values for mRNA testing were 74%, 80%, and 93%, respectively. By comparison the sensitivity, specificity, and negative predictive values were 51%, 80%, and 88% for FISH and 30%, 90%, and 86% for cytology.
Looking at high-grade disease only, the sensitivity, specificity, and negative predictive values were 83.3%, 75.8%, and 97.6% for mRNA testing, 75.0%, 79.5%, and 96.6% for FISH testing, and 50.0%, 90.7%, and 94.2% for cytology.
Bacillus Calmette-Guérin treatment in the past 90 days did not influence results of the mRNA test, one additional analysis showed.
The Xpert mRNA assay evaluates five mRNA targets: ABL1, ANXA10, UPK1B, CRH, and IGF2, results of which are combined to classify samples as either negative or positive. The test has a “hands-on time” of less than 2 minutes and provides results in 90 minutes, according to Dr. van Valenberg and his coinvestigators.
“Cytology requires a review by a pathologist, is not performed on the same day as a clinic visit, and has associated inter- and intraobserver variability,” the researchers wrote.
Dr. van Valenberg reported no financial disclosures related to the study. Several study coauthors reported employment or financial disclosures related to Cepheid, which provided funding and support for the conduct of the study. Study coauthors provided additional disclosures related to MDxHealth, PHotocure, Bristol-Myers Squibb, Johnson & Johnson, Roche, Bayer, and Astellas, among others.
SOURCE: van Valenberg FJP et al. Eur Urol. 2018 Dec 12. doi: 10.1016/j.eururo.2018.11.055.
An mRNA-based assay for surveillance of patients with bladder cancer outperformed standard urine tests on certain measures in a validation study, investigators report.
The mRNA-based urine test (Xpert, Cepheid) had better sensitivity and negative predictive value compared to urine cytology and to UroVysion fluorescence in situ hybridization (FISH) testing, according to F. Johannes P. van Valenberg, of Radboud University Medical Center, Nijmegen, the Netherlands, and his colleagues.
The reported results suggest this mRNA test could partially replace white-light cystoscopy, helping urologists maintain the recommended follow-up schedules for patients with a history of bladder cancer, Dr. van Valenberg and his coauthors wrote. The report is in European Urology.
“If used in the follow-up of non–muscle invasive bladder cancer, a cystoscopy might be waived if the Xpert result is negative,” they wrote.
The negative predictive value for high-grade disease was 98%, suggesting the mRNA-based test could help urologists avoid invasive cystoscopies for intermediate- to high-risk patients, which could reduce costs and patient discomfort, they added.
The prospective, 19-center validation study enrolled 363 individuals with a history of non–muscle invasive bladder cancer who were scheduled for a standard cystoscopy. Voided urine specimens from a total of 259 patients were evaluated with all three methods: the mRNA test, FISH testing, and cytology.
In a comparison of the tests, the mRNA test identified more recurrent cancers correctly, and was more sensitive in detecting low-grade tumors than was FISH (P less than .001) or cytology (P = .021), they reported.
The mRNA test was more sensitive for detection of the most common recurrent tumors, independent of grade.
The sensitivity, specificity, and negative predictive values for mRNA testing were 74%, 80%, and 93%, respectively. By comparison the sensitivity, specificity, and negative predictive values were 51%, 80%, and 88% for FISH and 30%, 90%, and 86% for cytology.
Looking at high-grade disease only, the sensitivity, specificity, and negative predictive values were 83.3%, 75.8%, and 97.6% for mRNA testing, 75.0%, 79.5%, and 96.6% for FISH testing, and 50.0%, 90.7%, and 94.2% for cytology.
Bacillus Calmette-Guérin treatment in the past 90 days did not influence results of the mRNA test, one additional analysis showed.
The Xpert mRNA assay evaluates five mRNA targets: ABL1, ANXA10, UPK1B, CRH, and IGF2, results of which are combined to classify samples as either negative or positive. The test has a “hands-on time” of less than 2 minutes and provides results in 90 minutes, according to Dr. van Valenberg and his coinvestigators.
“Cytology requires a review by a pathologist, is not performed on the same day as a clinic visit, and has associated inter- and intraobserver variability,” the researchers wrote.
Dr. van Valenberg reported no financial disclosures related to the study. Several study coauthors reported employment or financial disclosures related to Cepheid, which provided funding and support for the conduct of the study. Study coauthors provided additional disclosures related to MDxHealth, PHotocure, Bristol-Myers Squibb, Johnson & Johnson, Roche, Bayer, and Astellas, among others.
SOURCE: van Valenberg FJP et al. Eur Urol. 2018 Dec 12. doi: 10.1016/j.eururo.2018.11.055.
FROM EUROPEAN UROLOGY
Key clinical point: A noninvasive mRNA-based urine test performed favorably compared with standard urine tests for surveillance of patients with bladder cancer.
Major finding: The mRNA test had a sensitivity of 74% and negative predictive value of 93%, superior to what was observed with fluorescence in situ hybridization (FISH) testing and cytology, according to investigators.
Study details: Prospective validation study including 363 patients with a history of non–muscle invasive bladder cancer.
Disclosures: Funding and trial support came from Cepheid. Several study coauthors reported employment or financial disclosures related to that company. Dr. van Valenberg reported no financial disclosures related to the study, while coauthors provided disclosures related to Bristol-Myers Squibb, Johnson & Johnson, Roche, Bayer, Astellas, and others.
Source: Van Valenberg FJP et al. Eur Urol. 2018 Dec 12. doi: 10.1016/j.eururo.2018.11.055.
‘Optimal’ pazopanib levels linked to lower toxicity in RCC patients
In patients with renal cell carcinoma, trough concentrations of pazopanib in a specific target range were associated with better safety and comparable efficacy, versus higher concentrations of the drug, authors of an exploratory investigation have reported.
Fewer serious toxicities were seen in patients with pazopanib concentrations in the 20- to 50-mcg/mL range, yet overall response rate was similar compared to patients with concentrations over 50 mcg/mL, according to results of the retrospective study.
The findings suggest therapeutic drug monitoring to achieve a specific trough concentration may be useful to optimize pazopanib dosing, according to investigator Tomohiro Terada, PhD, of Shiga University of Medical Science Hospital, Japan, and coinvestigators.
Therapeutic drug monitoring for sunitinib in RCC patients was recently covered by medical insurance in Japan, Dr. Terada and colleagues wrote in Clinical Genitourinary Cancer.
The strategy may be especially suited to adjusting the dose of pazopanib, which is often associated with severe toxicities and has pharmacokinetics that suggest a wide variation between individuals, they added.
The retrospective study by Dr. Terada and colleagues included 27 renal cell carcinoma patients who received pazopanib at doses of 400, 600, or 800 mg daily based on the recommendation of the treating physician, with doses reduced or discontinued based on adverse events or progression.
Trough concentrations of pazopanib 3 months after starting the treatment ranged from a low of 10.6 mcg/mL to a high of 106 mcg/mL, with a median of 49.1 mcg/mL, according to the report.
One-third of patients experienced grade 3 or greater toxicities, including anorexia, hypertension, anemia, and thrombocytopenia, among others. The median pazopanib concentration for those nine patients was 69.3 mcg/mL, compared with 41.2 mcg/mL for those not experiencing serious toxicities (P less than .05).
A concentration over 50.3 mcg/mL predicted grade 3 or greater toxicity, statistical analysis showed, with 8 out of 13 patients with concentrations above that threshold experiencing serious toxicities. By contrast, 1 of 14 patients below that threshold experienced grade 3 or greater toxicities.
No responses were observed in three patients with pazopanib concentrations below 20.5 mcg/mL, a concentration level considered to be subtherapeutic based on previous investigations.
Overall response rates were similar for patients with concentrations in the “optimal” 20.5- and 50.3-mcg/mL range and those with concentrations over 50.3 mcg/mL, according to the investigators.
Among 11 patients in that “optimal” range, partial responses were seen in 5, they reported, while for the 13 patients with high concentrations of the drug, partial responses were seen in 4 patients and a complete response was seen in 1 patient.
Trough concentrations may not predict all types of toxicities, according to the investigators, who said that the results of their small, retrospective analysis should be validated in a large, prospective study.
In particular, there was no significant relationship between trough concentration of pazopanib and development of grade 2 or greater hepatotoxicity, a common and specific side effect of the drug.
“Based on these findings, pazopanib-induced hepatotoxicity may not be related to the pharmacokinetics of pazopanib,” they wrote. “Therefore, pharmacogenomics testing is needed to predict pazopanib-induced hepatotoxicity.”
Dr. Terada and coauthors said they had no conflicts of interest to report.
SOURCE: Noda S et al. Clin Genitourin Cancer. 2018 Dec 7. doi: 10.1016/j.clgc.2018.12.001.
In patients with renal cell carcinoma, trough concentrations of pazopanib in a specific target range were associated with better safety and comparable efficacy, versus higher concentrations of the drug, authors of an exploratory investigation have reported.
Fewer serious toxicities were seen in patients with pazopanib concentrations in the 20- to 50-mcg/mL range, yet overall response rate was similar compared to patients with concentrations over 50 mcg/mL, according to results of the retrospective study.
The findings suggest therapeutic drug monitoring to achieve a specific trough concentration may be useful to optimize pazopanib dosing, according to investigator Tomohiro Terada, PhD, of Shiga University of Medical Science Hospital, Japan, and coinvestigators.
Therapeutic drug monitoring for sunitinib in RCC patients was recently covered by medical insurance in Japan, Dr. Terada and colleagues wrote in Clinical Genitourinary Cancer.
The strategy may be especially suited to adjusting the dose of pazopanib, which is often associated with severe toxicities and has pharmacokinetics that suggest a wide variation between individuals, they added.
The retrospective study by Dr. Terada and colleagues included 27 renal cell carcinoma patients who received pazopanib at doses of 400, 600, or 800 mg daily based on the recommendation of the treating physician, with doses reduced or discontinued based on adverse events or progression.
Trough concentrations of pazopanib 3 months after starting the treatment ranged from a low of 10.6 mcg/mL to a high of 106 mcg/mL, with a median of 49.1 mcg/mL, according to the report.
One-third of patients experienced grade 3 or greater toxicities, including anorexia, hypertension, anemia, and thrombocytopenia, among others. The median pazopanib concentration for those nine patients was 69.3 mcg/mL, compared with 41.2 mcg/mL for those not experiencing serious toxicities (P less than .05).
A concentration over 50.3 mcg/mL predicted grade 3 or greater toxicity, statistical analysis showed, with 8 out of 13 patients with concentrations above that threshold experiencing serious toxicities. By contrast, 1 of 14 patients below that threshold experienced grade 3 or greater toxicities.
No responses were observed in three patients with pazopanib concentrations below 20.5 mcg/mL, a concentration level considered to be subtherapeutic based on previous investigations.
Overall response rates were similar for patients with concentrations in the “optimal” 20.5- and 50.3-mcg/mL range and those with concentrations over 50.3 mcg/mL, according to the investigators.
Among 11 patients in that “optimal” range, partial responses were seen in 5, they reported, while for the 13 patients with high concentrations of the drug, partial responses were seen in 4 patients and a complete response was seen in 1 patient.
Trough concentrations may not predict all types of toxicities, according to the investigators, who said that the results of their small, retrospective analysis should be validated in a large, prospective study.
In particular, there was no significant relationship between trough concentration of pazopanib and development of grade 2 or greater hepatotoxicity, a common and specific side effect of the drug.
“Based on these findings, pazopanib-induced hepatotoxicity may not be related to the pharmacokinetics of pazopanib,” they wrote. “Therefore, pharmacogenomics testing is needed to predict pazopanib-induced hepatotoxicity.”
Dr. Terada and coauthors said they had no conflicts of interest to report.
SOURCE: Noda S et al. Clin Genitourin Cancer. 2018 Dec 7. doi: 10.1016/j.clgc.2018.12.001.
In patients with renal cell carcinoma, trough concentrations of pazopanib in a specific target range were associated with better safety and comparable efficacy, versus higher concentrations of the drug, authors of an exploratory investigation have reported.
Fewer serious toxicities were seen in patients with pazopanib concentrations in the 20- to 50-mcg/mL range, yet overall response rate was similar compared to patients with concentrations over 50 mcg/mL, according to results of the retrospective study.
The findings suggest therapeutic drug monitoring to achieve a specific trough concentration may be useful to optimize pazopanib dosing, according to investigator Tomohiro Terada, PhD, of Shiga University of Medical Science Hospital, Japan, and coinvestigators.
Therapeutic drug monitoring for sunitinib in RCC patients was recently covered by medical insurance in Japan, Dr. Terada and colleagues wrote in Clinical Genitourinary Cancer.
The strategy may be especially suited to adjusting the dose of pazopanib, which is often associated with severe toxicities and has pharmacokinetics that suggest a wide variation between individuals, they added.
The retrospective study by Dr. Terada and colleagues included 27 renal cell carcinoma patients who received pazopanib at doses of 400, 600, or 800 mg daily based on the recommendation of the treating physician, with doses reduced or discontinued based on adverse events or progression.
Trough concentrations of pazopanib 3 months after starting the treatment ranged from a low of 10.6 mcg/mL to a high of 106 mcg/mL, with a median of 49.1 mcg/mL, according to the report.
One-third of patients experienced grade 3 or greater toxicities, including anorexia, hypertension, anemia, and thrombocytopenia, among others. The median pazopanib concentration for those nine patients was 69.3 mcg/mL, compared with 41.2 mcg/mL for those not experiencing serious toxicities (P less than .05).
A concentration over 50.3 mcg/mL predicted grade 3 or greater toxicity, statistical analysis showed, with 8 out of 13 patients with concentrations above that threshold experiencing serious toxicities. By contrast, 1 of 14 patients below that threshold experienced grade 3 or greater toxicities.
No responses were observed in three patients with pazopanib concentrations below 20.5 mcg/mL, a concentration level considered to be subtherapeutic based on previous investigations.
Overall response rates were similar for patients with concentrations in the “optimal” 20.5- and 50.3-mcg/mL range and those with concentrations over 50.3 mcg/mL, according to the investigators.
Among 11 patients in that “optimal” range, partial responses were seen in 5, they reported, while for the 13 patients with high concentrations of the drug, partial responses were seen in 4 patients and a complete response was seen in 1 patient.
Trough concentrations may not predict all types of toxicities, according to the investigators, who said that the results of their small, retrospective analysis should be validated in a large, prospective study.
In particular, there was no significant relationship between trough concentration of pazopanib and development of grade 2 or greater hepatotoxicity, a common and specific side effect of the drug.
“Based on these findings, pazopanib-induced hepatotoxicity may not be related to the pharmacokinetics of pazopanib,” they wrote. “Therefore, pharmacogenomics testing is needed to predict pazopanib-induced hepatotoxicity.”
Dr. Terada and coauthors said they had no conflicts of interest to report.
SOURCE: Noda S et al. Clin Genitourin Cancer. 2018 Dec 7. doi: 10.1016/j.clgc.2018.12.001.
FROM CLINICAL GENITOURINARY CANCER
Key clinical point: Trough concentrations of pazopanib in approximately the 20- to 50-mcg/mL range were associated with less toxicity but a similar overall response rate, versus higher concentrations.
Major finding: The overall response rate was about 46% in patients with pazopanib concentrations in that range, and in patients with higher concentrations. One patient in the “optimal” range experienced a grade 3 or greater toxicity.
Study details: An exploratory analysis including 27 patients with renal cell carcinoma.
Disclosures: The authors reported no conflicts of interest.
Source: Noda S et al. Clin Genitourin Cancer. 2018 Dec 7. doi: 10.1016/j.clgc.2018.12.001.
Cytoreductive nephrectomy may benefit RCC patients with brain mets
While patients with renal cell carcinoma (RCC) brain metastases are traditionally thought of as having a grim prognosis, a subset of patients may experience durable long-term survival with cytoreductive nephrectomy, authors of a retrospective study have reported.
Specifically, patients with brain lesions and no additional metastatic disease sites who underwent cytoreduction had a 2-year survival rate of 52%, according to the researchers’ analysis of data from the Surveillance, Epidemiology, and End Results (SEER) Program.
By contrast, these patients with brain-only metastases who did not undergo the procedure had a 2-year survival of 14%, reported Gennady Bratslavsky, MD, of State University of New York, Syracuse, and his associates.
The findings are at odds with “previously accepted dogma” that surgery is inadvisable in patients with RCC with brain metastases, which is traditionally thought of as a poor-risk feature, according to Dr. Bratslavsky and his colleagues.
“These patients seem to benefit from cytoreductive nephrectomy and may potentially be excellent candidates for clinical trials,” they wrote in Urologic Oncology.
However, patient selection likely drove survival outcomes in this retrospective cohort, the authors acknowledged, adding that they did not intend to argue “for or against” the role of cytoreductive nephrectomy.
In their analysis, they identified 6,403 patients with metastatic RCC and complete information on sites of metastasis; of these, 775 (12.1%) had brain metastases.
Patients with brain metastases generally fared worse than did patients with other, non-brain metastases, with 2-year survival rates of 14.9% and 28.6%, respectively (P less than .0001); however, patients with brain-only metastases had a 2-year survival of 31% overall in this analysis.
Cytoreductive nephrectomy was performed in 40.8% of patients with brain-only metastases, versus 20.8% of patients with synchronous metastases (P less than .0001).
Brain-only metastases patients who underwent cytoreductive nephrectomy had 1-year survival of 67%, 2-year survival of 52%, and median survival of 33 months, the data show. In contrast, brain-only metastases patients who did not undergo the procedure had 1-year survival of 26%, 2-year survival of 14%, and median survival of just 5 months.
Even when the researchers included patients with brain metastases in addition to other sites, cytoreduction was associated with superior outcomes, compared with patients with non-brain metastases who did not undergo cytoreduction, the investigators said. The reported 2-year survival rates in that analysis were 34.1% for brain metastasis and cytoreduction versus 14.4% for non-brain metastasis and no cytoreduction.
While this study was retrospective and was based on limited patient data, these findings nevertheless suggest that patients with RCC and isolated brain metastases might benefit from the procedure, Dr. Bratslavsky and his coauthors said.
“We anticipate that this work will be helpful for patient counseling and for modifying future exclusion criteria in clinical trials,” they concluded.
Dr. Bratslavsky and his coauthors listed no disclosures related to their research.
SOURCE: Daugherty M et al. Urol Oncol. 2018 Dec 5. doi: 10.1016/j.urolonc.2018.10.021.
While patients with renal cell carcinoma (RCC) brain metastases are traditionally thought of as having a grim prognosis, a subset of patients may experience durable long-term survival with cytoreductive nephrectomy, authors of a retrospective study have reported.
Specifically, patients with brain lesions and no additional metastatic disease sites who underwent cytoreduction had a 2-year survival rate of 52%, according to the researchers’ analysis of data from the Surveillance, Epidemiology, and End Results (SEER) Program.
By contrast, these patients with brain-only metastases who did not undergo the procedure had a 2-year survival of 14%, reported Gennady Bratslavsky, MD, of State University of New York, Syracuse, and his associates.
The findings are at odds with “previously accepted dogma” that surgery is inadvisable in patients with RCC with brain metastases, which is traditionally thought of as a poor-risk feature, according to Dr. Bratslavsky and his colleagues.
“These patients seem to benefit from cytoreductive nephrectomy and may potentially be excellent candidates for clinical trials,” they wrote in Urologic Oncology.
However, patient selection likely drove survival outcomes in this retrospective cohort, the authors acknowledged, adding that they did not intend to argue “for or against” the role of cytoreductive nephrectomy.
In their analysis, they identified 6,403 patients with metastatic RCC and complete information on sites of metastasis; of these, 775 (12.1%) had brain metastases.
Patients with brain metastases generally fared worse than did patients with other, non-brain metastases, with 2-year survival rates of 14.9% and 28.6%, respectively (P less than .0001); however, patients with brain-only metastases had a 2-year survival of 31% overall in this analysis.
Cytoreductive nephrectomy was performed in 40.8% of patients with brain-only metastases, versus 20.8% of patients with synchronous metastases (P less than .0001).
Brain-only metastases patients who underwent cytoreductive nephrectomy had 1-year survival of 67%, 2-year survival of 52%, and median survival of 33 months, the data show. In contrast, brain-only metastases patients who did not undergo the procedure had 1-year survival of 26%, 2-year survival of 14%, and median survival of just 5 months.
Even when the researchers included patients with brain metastases in addition to other sites, cytoreduction was associated with superior outcomes, compared with patients with non-brain metastases who did not undergo cytoreduction, the investigators said. The reported 2-year survival rates in that analysis were 34.1% for brain metastasis and cytoreduction versus 14.4% for non-brain metastasis and no cytoreduction.
While this study was retrospective and was based on limited patient data, these findings nevertheless suggest that patients with RCC and isolated brain metastases might benefit from the procedure, Dr. Bratslavsky and his coauthors said.
“We anticipate that this work will be helpful for patient counseling and for modifying future exclusion criteria in clinical trials,” they concluded.
Dr. Bratslavsky and his coauthors listed no disclosures related to their research.
SOURCE: Daugherty M et al. Urol Oncol. 2018 Dec 5. doi: 10.1016/j.urolonc.2018.10.021.
While patients with renal cell carcinoma (RCC) brain metastases are traditionally thought of as having a grim prognosis, a subset of patients may experience durable long-term survival with cytoreductive nephrectomy, authors of a retrospective study have reported.
Specifically, patients with brain lesions and no additional metastatic disease sites who underwent cytoreduction had a 2-year survival rate of 52%, according to the researchers’ analysis of data from the Surveillance, Epidemiology, and End Results (SEER) Program.
By contrast, these patients with brain-only metastases who did not undergo the procedure had a 2-year survival of 14%, reported Gennady Bratslavsky, MD, of State University of New York, Syracuse, and his associates.
The findings are at odds with “previously accepted dogma” that surgery is inadvisable in patients with RCC with brain metastases, which is traditionally thought of as a poor-risk feature, according to Dr. Bratslavsky and his colleagues.
“These patients seem to benefit from cytoreductive nephrectomy and may potentially be excellent candidates for clinical trials,” they wrote in Urologic Oncology.
However, patient selection likely drove survival outcomes in this retrospective cohort, the authors acknowledged, adding that they did not intend to argue “for or against” the role of cytoreductive nephrectomy.
In their analysis, they identified 6,403 patients with metastatic RCC and complete information on sites of metastasis; of these, 775 (12.1%) had brain metastases.
Patients with brain metastases generally fared worse than did patients with other, non-brain metastases, with 2-year survival rates of 14.9% and 28.6%, respectively (P less than .0001); however, patients with brain-only metastases had a 2-year survival of 31% overall in this analysis.
Cytoreductive nephrectomy was performed in 40.8% of patients with brain-only metastases, versus 20.8% of patients with synchronous metastases (P less than .0001).
Brain-only metastases patients who underwent cytoreductive nephrectomy had 1-year survival of 67%, 2-year survival of 52%, and median survival of 33 months, the data show. In contrast, brain-only metastases patients who did not undergo the procedure had 1-year survival of 26%, 2-year survival of 14%, and median survival of just 5 months.
Even when the researchers included patients with brain metastases in addition to other sites, cytoreduction was associated with superior outcomes, compared with patients with non-brain metastases who did not undergo cytoreduction, the investigators said. The reported 2-year survival rates in that analysis were 34.1% for brain metastasis and cytoreduction versus 14.4% for non-brain metastasis and no cytoreduction.
While this study was retrospective and was based on limited patient data, these findings nevertheless suggest that patients with RCC and isolated brain metastases might benefit from the procedure, Dr. Bratslavsky and his coauthors said.
“We anticipate that this work will be helpful for patient counseling and for modifying future exclusion criteria in clinical trials,” they concluded.
Dr. Bratslavsky and his coauthors listed no disclosures related to their research.
SOURCE: Daugherty M et al. Urol Oncol. 2018 Dec 5. doi: 10.1016/j.urolonc.2018.10.021.
FROM UROLOGIC ONCOLOGY
Key clinical point: Renal cell carcinoma patients with brain-only metastases may experience durable long-term survival following cytoreductive nephrectomy.
Major finding: Two-year survival was 52% and 14%, respectively, for patients with brain-only metastases who did and did not undergo the procedure.
Study details: A retrospective analysis of Surveillance, Epidemiology, and End Results (SEER) Program data including 6,403 patients with metastatic RCC and complete data on sites of metastasis.
Disclosures: Dr. Bratslavsky and coauthors listed no disclosures related to their research.
Source: Daugherty M et al. Urol Oncol. 2018 Dec 5.doi: 10.1016/j.urolonc.2018.10.021.
Lipoprotein ratio linked to poor survival in metastatic RCC
The ratio of apolipoproteins B and A1 (apo B/A1) is an independent prognostic factor in patients with metastatic renal cell carcinoma, according to authors of a recent retrospective study.
The apo B/A1 ratio, evaluated prior to cytoreductive nephrectomy, was significantly linked with poor progression-free survival and overall survival, according to Fan Zhang, MD, of the Chinese PLA General Hospital in Beijing and coauthors.
Those findings suggest that patients with metastatic RCC should receive “consistent follow-up” that includes evaluation of that ratio, Dr. Zhang and colleagues said.
“As a novel prognostic factor, the preoperative apo B/A1 ratio can be utilized as a supplement to improve the current prognostic evaluation and treatment decision for patients with metastatic RCC,” they wrote in Urologic Oncology.
Apo B and A1, which are predominant components of low-density lipoprotein (LDL) and high-density lipoprotein (HDL), respectively, have “extensive connections” with cardiovascular disease, diabetes, and Alzheimer disease, the authors said.
The apo B/A1 ratio is a known risk index for cardiovascular disease, and in recent studies, it appeared to have some value in prognosis and prediction of gastric and colorectal cancer, among other neoplasms, they added.
In their retrospective study, Dr. Zhang and colleagues analyzed data on 287 metastatic RCC patients who underwent cytoreductive nephrectomy at the Chinese PLA General Hospital. The median age of the patients was 56 years, and the median apo B/A1 ratio was 0.859.
Significantly poorer progression-free survival was seen in the group of patients with a preoperative apo B/A1 ratio over the cutoff of 0.977 (P less than .0001) compared with those under the cutoff, the investigators reported. Likewise, overall survival was poorer for patients with an apo B/A1 ratio over a cutoff of 0.847 (P = .0005).
The apo B/A1 ratio was higher in patients with Fuhrman grade 3-4 versus grade 1-2 patients (P = .010), though the investigators said no significant differences in the ratio were seen in patients stratified by age, body mass index, fatty liver, number of metastases, among other subgroup analyses.
Multivariate analysis revealed that the two independent prognostic factors for progression-free survival were preoperative apo B/A1 ratio and Fuhrman grade, with hazard ratios of 3.131 and 1.906, respectively (P less than .001 for both), while independent prognostic factors for overall survival also included the apo B/A1 ratio (hazard ratio, 2.173; P less than .001) and Fuhrman grade, along with tumor necrosis and receiving targeted therapy.
This retrospective study was limited by relatively small samples from a single center, and the prognostic value of the apo B/A1 ratio needs to be verified in other studies, investigators said.
“Peripheral blood biomarkers only provide a supplement to the traditional prognostic factors in the prediction of the prognosis for patients with metastatic RCC, and are still unable to replace it,” Dr. Zhang and associates said.
The Beijing Natural Science Foundation supported the study. Dr. Zhang and coauthors had no disclosed conflicts of interest related to the work.
SOURCE: Zhang F et al. Urol Oncol. 2018 Nov 30. doi: 10.1016/j.urolonc.2018.11.010.
The ratio of apolipoproteins B and A1 (apo B/A1) is an independent prognostic factor in patients with metastatic renal cell carcinoma, according to authors of a recent retrospective study.
The apo B/A1 ratio, evaluated prior to cytoreductive nephrectomy, was significantly linked with poor progression-free survival and overall survival, according to Fan Zhang, MD, of the Chinese PLA General Hospital in Beijing and coauthors.
Those findings suggest that patients with metastatic RCC should receive “consistent follow-up” that includes evaluation of that ratio, Dr. Zhang and colleagues said.
“As a novel prognostic factor, the preoperative apo B/A1 ratio can be utilized as a supplement to improve the current prognostic evaluation and treatment decision for patients with metastatic RCC,” they wrote in Urologic Oncology.
Apo B and A1, which are predominant components of low-density lipoprotein (LDL) and high-density lipoprotein (HDL), respectively, have “extensive connections” with cardiovascular disease, diabetes, and Alzheimer disease, the authors said.
The apo B/A1 ratio is a known risk index for cardiovascular disease, and in recent studies, it appeared to have some value in prognosis and prediction of gastric and colorectal cancer, among other neoplasms, they added.
In their retrospective study, Dr. Zhang and colleagues analyzed data on 287 metastatic RCC patients who underwent cytoreductive nephrectomy at the Chinese PLA General Hospital. The median age of the patients was 56 years, and the median apo B/A1 ratio was 0.859.
Significantly poorer progression-free survival was seen in the group of patients with a preoperative apo B/A1 ratio over the cutoff of 0.977 (P less than .0001) compared with those under the cutoff, the investigators reported. Likewise, overall survival was poorer for patients with an apo B/A1 ratio over a cutoff of 0.847 (P = .0005).
The apo B/A1 ratio was higher in patients with Fuhrman grade 3-4 versus grade 1-2 patients (P = .010), though the investigators said no significant differences in the ratio were seen in patients stratified by age, body mass index, fatty liver, number of metastases, among other subgroup analyses.
Multivariate analysis revealed that the two independent prognostic factors for progression-free survival were preoperative apo B/A1 ratio and Fuhrman grade, with hazard ratios of 3.131 and 1.906, respectively (P less than .001 for both), while independent prognostic factors for overall survival also included the apo B/A1 ratio (hazard ratio, 2.173; P less than .001) and Fuhrman grade, along with tumor necrosis and receiving targeted therapy.
This retrospective study was limited by relatively small samples from a single center, and the prognostic value of the apo B/A1 ratio needs to be verified in other studies, investigators said.
“Peripheral blood biomarkers only provide a supplement to the traditional prognostic factors in the prediction of the prognosis for patients with metastatic RCC, and are still unable to replace it,” Dr. Zhang and associates said.
The Beijing Natural Science Foundation supported the study. Dr. Zhang and coauthors had no disclosed conflicts of interest related to the work.
SOURCE: Zhang F et al. Urol Oncol. 2018 Nov 30. doi: 10.1016/j.urolonc.2018.11.010.
The ratio of apolipoproteins B and A1 (apo B/A1) is an independent prognostic factor in patients with metastatic renal cell carcinoma, according to authors of a recent retrospective study.
The apo B/A1 ratio, evaluated prior to cytoreductive nephrectomy, was significantly linked with poor progression-free survival and overall survival, according to Fan Zhang, MD, of the Chinese PLA General Hospital in Beijing and coauthors.
Those findings suggest that patients with metastatic RCC should receive “consistent follow-up” that includes evaluation of that ratio, Dr. Zhang and colleagues said.
“As a novel prognostic factor, the preoperative apo B/A1 ratio can be utilized as a supplement to improve the current prognostic evaluation and treatment decision for patients with metastatic RCC,” they wrote in Urologic Oncology.
Apo B and A1, which are predominant components of low-density lipoprotein (LDL) and high-density lipoprotein (HDL), respectively, have “extensive connections” with cardiovascular disease, diabetes, and Alzheimer disease, the authors said.
The apo B/A1 ratio is a known risk index for cardiovascular disease, and in recent studies, it appeared to have some value in prognosis and prediction of gastric and colorectal cancer, among other neoplasms, they added.
In their retrospective study, Dr. Zhang and colleagues analyzed data on 287 metastatic RCC patients who underwent cytoreductive nephrectomy at the Chinese PLA General Hospital. The median age of the patients was 56 years, and the median apo B/A1 ratio was 0.859.
Significantly poorer progression-free survival was seen in the group of patients with a preoperative apo B/A1 ratio over the cutoff of 0.977 (P less than .0001) compared with those under the cutoff, the investigators reported. Likewise, overall survival was poorer for patients with an apo B/A1 ratio over a cutoff of 0.847 (P = .0005).
The apo B/A1 ratio was higher in patients with Fuhrman grade 3-4 versus grade 1-2 patients (P = .010), though the investigators said no significant differences in the ratio were seen in patients stratified by age, body mass index, fatty liver, number of metastases, among other subgroup analyses.
Multivariate analysis revealed that the two independent prognostic factors for progression-free survival were preoperative apo B/A1 ratio and Fuhrman grade, with hazard ratios of 3.131 and 1.906, respectively (P less than .001 for both), while independent prognostic factors for overall survival also included the apo B/A1 ratio (hazard ratio, 2.173; P less than .001) and Fuhrman grade, along with tumor necrosis and receiving targeted therapy.
This retrospective study was limited by relatively small samples from a single center, and the prognostic value of the apo B/A1 ratio needs to be verified in other studies, investigators said.
“Peripheral blood biomarkers only provide a supplement to the traditional prognostic factors in the prediction of the prognosis for patients with metastatic RCC, and are still unable to replace it,” Dr. Zhang and associates said.
The Beijing Natural Science Foundation supported the study. Dr. Zhang and coauthors had no disclosed conflicts of interest related to the work.
SOURCE: Zhang F et al. Urol Oncol. 2018 Nov 30. doi: 10.1016/j.urolonc.2018.11.010.
FROM UROLOGIC ONCOLOGY
Key clinical point: The ratio of apolipoproteins B and A1 (apo B/A1) was an independent prognostic factor in patients with metastatic renal cell carcinoma (RCC).
Major finding: The preoperative apo B/A1 ratio was independently prognostic for progression-free survival and overall survival, with hazard ratios of 3.131 and 2.173, respectively (P less than .001 for both).
Study details: A retrospective, single-center study including 287 patients with metastatic RCC who underwent cytoreductive nephrectomy.
Disclosures: Beijing Natural Science Foundation supported the study. Dr. Zhang and coauthors had no disclosed conflicts of interest.
Source: Zhang F et al. Urol Oncol. 2018 Nov 30. doi: 10.1016/j.urolonc.2018.11.010.
RCC strongly linked to melanoma, and vice versa
A review of registry data from a major cancer center revealed a strong, bidirectional association between renal cell carcinoma (RCC) and melanoma.
A greater than twofold risk of melanoma in patients with RCC, and a nearly threefold risk of RCC in melanoma patients, were found in the review of the International Tumor Registry Database at The University of Texas MD Anderson Cancer Center.
The incidence of subsequent melanomas or RCCs in this study was in line with other recent reports from cancer registry analyses, reported Kevin B. Kim, MD, and his coinvestigators.
“Clinicians should be more watchful for these secondary cancers in patients with a history of melanoma or RCC,” they wrote. The report is in Cancer Epidemiology.
They found a total of 13,879 patients with melanoma and 7,597 patients with RCC in their review. Of those patients, 89 had both a melanoma and an RCC diagnosis. About 30% were first diagnosed with RCC, 61% were first diagnosed with melanoma, and 9% had both diagnoses around the same time.
Among the RCC-first patients, the standardized incidence ratio for developing a second primary melanoma was 2.31 (95% confidence interval, 1.52-3.37; P less than .001), while for melanoma-first patients, for developing a second primary RCC, it was 2.87 (95% CI, 2.16-3.74; P less than .001).
Those statistics were consistent with other registry reports, according to Dr. Kim and his colleagues, who wrote that the standardized incidence ratios in those studies ranged from 1.28 to 2.5.
In the MD Anderson registry study, nearly one-third of patients with secondary primary melanoma or RCC had a history of additional secondary cancers, according to the researchers. Those diagnoses included nonmelanoma skin cancers, leukemias, prostate cancer, breast cancer, and colon cancer, among others.
That suggested the presence of possible common risk factors that may have included abnormal genetics, though the database lacked the genetic sequencing and family history data to explore that hypothesis further.
“It would be highly desirable to assess germline genetic information on patients and their families, and also somatic gene aberrations in the tumor lesions, in a more systematic way in order to better elucidate the contribution of the genetics in the association between melanoma and RCC,” Dr. Kim and his colleagues said.
They reported that they had no conflicts of interest.
SOURCE: Kim KB et al. Cancer Epidemiol. 2018 Oct 19;57:80-4.
A review of registry data from a major cancer center revealed a strong, bidirectional association between renal cell carcinoma (RCC) and melanoma.
A greater than twofold risk of melanoma in patients with RCC, and a nearly threefold risk of RCC in melanoma patients, were found in the review of the International Tumor Registry Database at The University of Texas MD Anderson Cancer Center.
The incidence of subsequent melanomas or RCCs in this study was in line with other recent reports from cancer registry analyses, reported Kevin B. Kim, MD, and his coinvestigators.
“Clinicians should be more watchful for these secondary cancers in patients with a history of melanoma or RCC,” they wrote. The report is in Cancer Epidemiology.
They found a total of 13,879 patients with melanoma and 7,597 patients with RCC in their review. Of those patients, 89 had both a melanoma and an RCC diagnosis. About 30% were first diagnosed with RCC, 61% were first diagnosed with melanoma, and 9% had both diagnoses around the same time.
Among the RCC-first patients, the standardized incidence ratio for developing a second primary melanoma was 2.31 (95% confidence interval, 1.52-3.37; P less than .001), while for melanoma-first patients, for developing a second primary RCC, it was 2.87 (95% CI, 2.16-3.74; P less than .001).
Those statistics were consistent with other registry reports, according to Dr. Kim and his colleagues, who wrote that the standardized incidence ratios in those studies ranged from 1.28 to 2.5.
In the MD Anderson registry study, nearly one-third of patients with secondary primary melanoma or RCC had a history of additional secondary cancers, according to the researchers. Those diagnoses included nonmelanoma skin cancers, leukemias, prostate cancer, breast cancer, and colon cancer, among others.
That suggested the presence of possible common risk factors that may have included abnormal genetics, though the database lacked the genetic sequencing and family history data to explore that hypothesis further.
“It would be highly desirable to assess germline genetic information on patients and their families, and also somatic gene aberrations in the tumor lesions, in a more systematic way in order to better elucidate the contribution of the genetics in the association between melanoma and RCC,” Dr. Kim and his colleagues said.
They reported that they had no conflicts of interest.
SOURCE: Kim KB et al. Cancer Epidemiol. 2018 Oct 19;57:80-4.
A review of registry data from a major cancer center revealed a strong, bidirectional association between renal cell carcinoma (RCC) and melanoma.
A greater than twofold risk of melanoma in patients with RCC, and a nearly threefold risk of RCC in melanoma patients, were found in the review of the International Tumor Registry Database at The University of Texas MD Anderson Cancer Center.
The incidence of subsequent melanomas or RCCs in this study was in line with other recent reports from cancer registry analyses, reported Kevin B. Kim, MD, and his coinvestigators.
“Clinicians should be more watchful for these secondary cancers in patients with a history of melanoma or RCC,” they wrote. The report is in Cancer Epidemiology.
They found a total of 13,879 patients with melanoma and 7,597 patients with RCC in their review. Of those patients, 89 had both a melanoma and an RCC diagnosis. About 30% were first diagnosed with RCC, 61% were first diagnosed with melanoma, and 9% had both diagnoses around the same time.
Among the RCC-first patients, the standardized incidence ratio for developing a second primary melanoma was 2.31 (95% confidence interval, 1.52-3.37; P less than .001), while for melanoma-first patients, for developing a second primary RCC, it was 2.87 (95% CI, 2.16-3.74; P less than .001).
Those statistics were consistent with other registry reports, according to Dr. Kim and his colleagues, who wrote that the standardized incidence ratios in those studies ranged from 1.28 to 2.5.
In the MD Anderson registry study, nearly one-third of patients with secondary primary melanoma or RCC had a history of additional secondary cancers, according to the researchers. Those diagnoses included nonmelanoma skin cancers, leukemias, prostate cancer, breast cancer, and colon cancer, among others.
That suggested the presence of possible common risk factors that may have included abnormal genetics, though the database lacked the genetic sequencing and family history data to explore that hypothesis further.
“It would be highly desirable to assess germline genetic information on patients and their families, and also somatic gene aberrations in the tumor lesions, in a more systematic way in order to better elucidate the contribution of the genetics in the association between melanoma and RCC,” Dr. Kim and his colleagues said.
They reported that they had no conflicts of interest.
SOURCE: Kim KB et al. Cancer Epidemiol. 2018 Oct 19;57:80-4.
FROM CANCER EPIDEMIOLOGY
Key clinical point: Review of international tumor registry data demonstrated a strong and bidirectional association between renal cell carcinoma (RCC) and melanoma.
Major finding: Standardized incidence ratios were 2.31 for developing a second primary melanoma in patients who first had an RCC diagnosis, and 2.87 for developing a second primary RCC in patients who had melanoma first.
Study details: Analysis of 13,879 patients with melanoma and 7,597 patients with RCC in the International Tumor Registry Database at The University of Texas MD Anderson Cancer Center.
Disclosures: The authors reported that they had no conflicts of interest.
Source: Kim KB et al. Cancer Epidemiol. 2018 Oct 19;57:80-4.
Development, implementation, and evaluation of a prostate cancer supportive care program
Prostate cancer is the most common malignancy diagnosed in Canadian men. An estimated 21,300 Canadian men were diagnosed with the disease in 2017, representing 21% of all new cancer cases.1 There are about 176,000 men living with prostate cancer in Canada.1 In the United States, there were 2,778,630 survivors of prostate cancer as of 2012 and that population is expected to increase by more than 1 million (40%) to 3,922,600 by 2022.2
Although 96% of men diagnosed with prostate cancer now survive longer than 5 years3, many will suffer from treatment-related sequelae that can have a profound effect on quality of life for themselves and their partners.4,5 Impacts include sexual, urinary, and bowel dysfunctions6 owing to treatment of the primary tumor as well as reduced muscle and bone mass, osteoporosis, fatigue, obesity, and glucose intolerance or diabetes7 owing to androgen-deprivation therapy (ADT). Many men also suffer from psychological issues such as depression, anxiety, anger and irritability, sense of isolation, grief, and loss of masculinity.8,9 The psychological impacts also continue well beyond the completion of treatment and can be significant for both patients and their partners.5,8
With posttreatment longevity and the associated complex sequelae, prostate cancer is being viewed increasingly as a chronic disease whose effects must be managed for many years after the completion of primary treatment. Supportive care that “[manages] symptoms and side effects, enables adaptation and coping, optimizes understanding and informed decision-making, and minimizes decrements in functioning”10 is becoming recognized as a critical component of direct oncologic care before, during, and after treatment. Health care professionals, scientists, governments, and patient advocates are increasingly calling for the development of comprehensive supportive care programs improve the quality of life for people diagnosed with cancer. A common model for survivorship care is a general program for all cancer survivors that provides disease- and patient-specific care plans. These care plans outline patients’ prior therapies, potential side effects, recommendations for monitoring (for side effects or relapse of cancer), and advice on how patients can maintain a healthy lifestyle.11 However, there are few survivorship programs for men with prostate cancer and their partners, and the evidence base around best practices for these programs is scant.12 Furthermore, up to 87% of men with a prostate cancer diagnosis report specific and significant unmet supportive care needs,10,13 with sexuality-related and psychological issues10,14 being the areas of greatest concern.
To address the complex supportive care needs of men with prostate cancer in British Columbia, Canada, the Vancouver Prostate Centre (VPC) and Department of Urologic Sciences at the University of British Columbia developed the multidisciplinary Prostate Cancer Supportive Care (PCSC) Program. The program aims to address the challenges of decision-making and coping faced by men with prostate cancer and their partners and family members along the entire disease trajectory. Services are provided at no cost to participants. Here, we outline the guiding principles for the PCSC program and its scope, delivery, and evaluation. We provide information on the more than 1,200 patients who have participated in the program since its inception in January of 2013, the rates of participation across the different program modules, and a selection of patient satisfaction measures. We also discuss successes and limitations and ongoing research and evaluation efforts, providing lessons learned to support the development of other supportive care programs in Canada and internationally.
Program description
Guiding principles
The PCSC Program is a clinical, educational, and research-based program, with 4 guiding principles: it is comprehensive, patient- and partner-centered, evidence-based, and supports new research. The program serves patients, partners, and families along the entire disease trajectory, recognizing that cancer is a family disease, affecting both the individual and social network, and that the psychological stress associated with a diagnosis of prostate cancer is borne heavily by partners. It has been designed, implemented, and refined with the best available evidence and with the intention to undergo consistent and repeated evaluation. Finally, it was designed to provide opportunities for targeted research efforts, supporting the growth of the evidence base in this area.
Patient entry and module descriptions
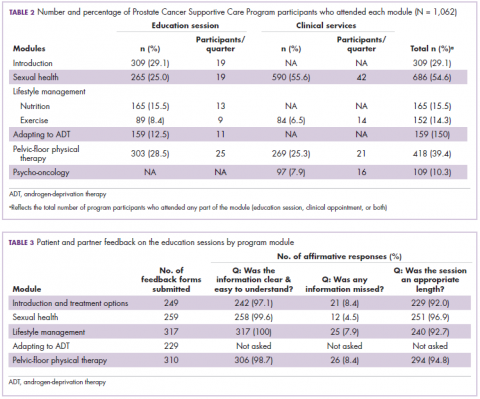
Patients can be referred to the program by a physician or other allied health professional. They may also self-refer, having been made aware of the program through our website, a variety of print materials, or by word of mouth. On referral, the program coordinator collects patients’ basic clinical and demographic data, assesses health literacy and lifestyle factors, and provides them with information on the program modules. As of December 2015, the program consisted of 6 distinct modules, each focusing on different elements of the disease trajectory or on addressing specific physical or mental health concerns. Modules are led by licensed health professionals with experience in oncology. No elements of the program are mandatory, and participants are free to pick and choose the components that are most relevant to them and their partners.
Introduction to prostate cancer and primary treatment options. This is a group-based module that focuses on educating newly diagnosed patients (and those going on or off active surveillance) on the basic biology of prostate cancer, the primary treatment options for localized disease, and the main side effects associated with the treatments. It also includes information about the other services offered by the program and any ongoing research studies. The session is held twice a month in the early evening and is run collectively by a urologist, radiation oncologist, patient representative, and program coordinator. It includes a brief one-on-one discussion between each patient and their partner or family member and the urologist and radiation oncologist to address any remaining questions. A copy of the patient’s biopsy report is on hand for the physician(s). Attendance of this session has been shown to significantly reduce pretreatment distress in both patients and their partners.15
Managing sexual function and intimacy. Sexual intimacy is tied to overall health outcomes, relationship satisfaction, and quality of life.16 Primary therapy for prostate cancer can be associated with substantial side-effects (eg, erectile dysfunction, incontinence, altered libido, penile shortening) that negatively affect sexual intimacy and have an impact on the patient individually as well as the sexual relationship he has with his partner.17
The program’s Sexual Health Service (SHS) provides patients and partners with information on the impact of treatment on sexual health.18 The SHS offers educational sessions led by a sexual rehabilitation nurse and clinical psychologist with a specialization in sexual health. Sessions focus on the impact of prostate cancer treatments on sexual function and therapeutic modalities, promote an understanding of the barriers to sexual adaptation posttreatment, and present options for sexual activity that are not solely dependent on the ability to achieve an erection. Once participants have attended an educational session, they are offered individual consultations with the sexual health nurse every 3 to 6 months for 2 years or longer, depending on the patient’s or couple’s needs. They are referred to the SHS’s sexual medicine physician if further medical intervention is warranted. The sexual health nurse works with the patient and partner to develop an individualized Sexual Health Rehabilitation Action Plan (SHRAP), which assists the couple in sexual adaptation going forward. The SHRAP is a tool devised by the sexual health nurse based on her clinical experience with couples affected by prostate cancer.
Couples who have been evaluated within the SHS are also invited to attend a second workshop on intimacy that is offered quarterly. Workshop participants discuss the impact of sexual changes on relationships, and strategies on how to enhance intimacy and sexual communication are presented. A resource package is provided to each couple to help re-establish and/or strengthen their various dimensions of intimacy.
Lifestyle management. The lifestyle management modules include separate nutrition and physical activity or exercise components. Referral to the smoking cessation program in the Vancouver Coastal Health Authority is made at program registration, if appropriate. The nutrition group-based education session is delivered by a registered dietitian from the British Columbia Cancer Agency who specializes in prostate cancer. The session focuses on evidence-based recommendations on diet after a diagnosis of prostate cancer, the use of dietary supplements, body weight and health, and practical nutrition tips. The exercise session is delivered by an exercise physiologist who specializes in working with cancer patients. It covers the value of exercise in terms of safety, prevention and reduction of treatment side effects (including from ADT), treatment prehabilitation and recovery, advanced cancer management, and long-term survival. A one-on-one exercise counseling clinic is also offered and aims to increase exercise adoption and long-term adherence in line with Canadian Physical Activity guidelines and exercise oncology guidelines,19,20 with follow-up appointments at 3, 6, and 12 months to help patients stay motivated and ensure they are exercising correctly. The individual consultations with the exercise physiologist include physical measures, exercise volume, treatment side effects, and coconstructed goal setting with an individualized formal exercise regimen (exercise prescription).
Adapting to ADT. This is an educational module offered to patients with metastatic prostate cancer who are starting hormone therapy treatments that lower serum testosterone into the castrate range. This program was one of several available through TrueNTH, a portfolio of projects funded by the Movember Foundation, through Prostate Cancer Canada. The session is delivered by a patient facilitator and focuses on strategies to recognize and adapt to the side effects of ADT21 while maintaining a good quality of life and strong intimate relationships with partners.22,23
Pelvic-floor physical therapy for urinary incontinence. This module includes a group-based and individualized education session for patients (either pre- or posttreatment) focused on reducing the effects of surgery and/or radiation therapy on urinary and sexual continence as well as on how to cope with these symptoms and minimize the effect they have on quality of life.24 The session is conducted by a physical therapist who is certified as a pelvic-floor specialist. Supervised pelvic-floor re-education and/or exercise has been shown to successfully reduce the degree of incontinence in this population.25 The module therefore also includes 3 one-on-one clinical appointments for patients who are still experiencing bother from incontinence 12 weeks after a radical prostatectomy or postradiation treatment.
Psycho-oncology. In recognition of the emotional and psychological burden associated with prostate cancer and the important role partners play in facilitating treatment of these psychological and/or psychosocial issues, the program offers appointments with a registered clinical counselor to address acute emotional distress. These are usually 1-hour appointments offered to both patients and partners, either separately or together. Appointments can be attended in person or conducted by telephone. When appropriate, patients are referred for further long-term individual support or couple support with their partners. A group therapy workshop was also initiated in 2016. In this program, men participate in a guided autobiographical life review through a process that focuses on developing a cohesive working group, learning strategic communication skills, and understanding and learning how to manage difficult emotions and transitional life stressors associated with prostate cancer. It also focuses on processing and integrating critical events that contribute to the men’s identity and psychological function and involves the consolidation of the personal learning that occurs. Postgroup referral plans are developed on an individual basis as needed.
Methods
Data
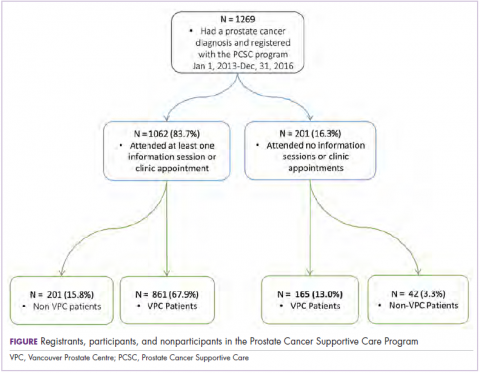
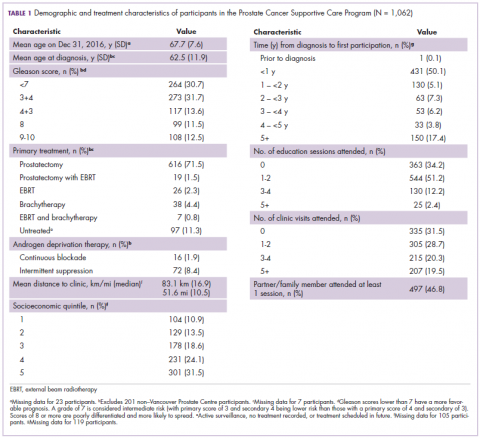
We obtained sociodemographic, diagnostic, and treatment information as well as clinic visit records for all PCSC Program registrants from the electronic medical record held at the VPC. Clinical variables included age at diagnosis, Gleason score, and primary treatment modality (including active surveillance and ADT use). The Gleason score determines the aggressiveness of a patient’s prostate cancer based on biopsy results. A score of 6 or less indicates that the disease is likely to grow slowly. A grade of 7 is considered intermediate risk (with primary score of 3 and secondary 4 being lower risk than those with a primary score of 4 and secondary of 3). A Gleason score of 8 or higher indicates aggressive disease that is poorly differentiated or high grade. Sociodemographic characteristics included age, travel distance to the clinic, and income quintile. We obtained attendance records for the modular education sessions from the program’s database. Patients who did not have any medical visits at the VPC (referred to henceforth as non-VPC patients) did not have a clinic record, so we excluded them from the subset of the analyses that depended on specific clinical variables.
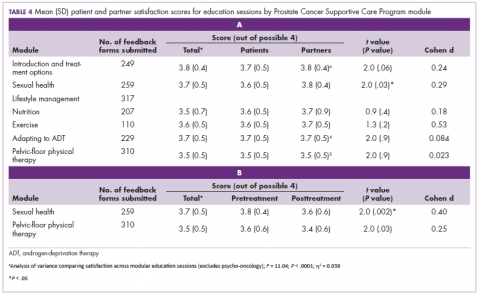
All patients and partners who participate in any PCSC Program education sessions (introduction to prostate cancer, sexual health, nutrition, exercise, ADT, and pelvic-floor physical therapy) are asked to complete voluntary, anonymous feedback forms. These forms assess participant satisfaction using a series of Likert-based and Boolean response items as well as qualitative commentary. They include questions that assess the timing, structure, and content of each session.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Statistical approach
Descriptive statistics were used to analyze participant characteristics, program participation rates, and participant satisfaction. For each module’s education session, we compared the overall satisfaction between patients and partners using t tests. We also compared the level of satisfaction across the different modules using a 1-way analysis of variance. For the sexual health and pelvic-floor physical therapy sessions, we compared satisfaction between participants who attended the education sessions before to those who attended following their primary treatment using t tests. We provide the eta squared (for analyses of variance) and Cohen d (for t tests) to provide an effect size estimate of any significant differences observed.
Results
Participants
From the program’s founding in January of 2013 to December 31, 2016, a total of 1,269 patients registered (an average of 317 patients a year). Of those, 1,026 (80.9%) had at least 1 prostate cancer–related visit at the VPC. The remaining 243 (19.1%) were non-VPC patients (Figure). Overall, 1,062 men (83.4%) who registered with the program went on to attend at least 1 education session or clinic appointment.
Average age among male program participants was 67.7 years, and age at diagnosis was 62.5 years (Table 1). In all, 273 men (31.7%) had Gleason 3+4, and 117 (13.7%) had Gleason 4+3. Most of the participants (76.9%) elected to undergo radical prostatectomy for primary treatment. Ninety-five men (8.9%) received at least some ADT treatment as an adjunct to radiation or to treat recurrent disease. Participants traveled an average of 83.1 km (51.6 miles; median, 6.9 km and 10.5 miles, respectively) to attend the program; 10% of participants traveled further than 112 km (70 mi) to the clinic. One hundred and four (10.9%) and 301 (31.5%) of registrants were in the lowest and highest income quintiles respectively. Four hundred and ninety-seven (46.8%) attended at lesson 1 session or clinic appointment with a partner or family member.
Program participation
Of the 1,062 men who participated in the program, 867 (80.1%) were patients of the VPC, and 205 (19.1%) were non-VPC patients. The education sessions for the introduction to prostate cancer and sexual health modules had the largest numbers of participants (309 and 265, respectively; Table 2); however, pelvic-floor physical therapy had the highest participation rate per quarter (25 patients). The clinical services offered within the sexual health module had the larger number of participants and highest participation rate per quarter (590 total patients, 42/quarter). Timing of program participation was highly variable, ranging from 6 days to 18.5 years after diagnosis (SD, 1,301 days). More than half of participants attended a session or clinic visit within the first year of their diagnosis. A total of 17% of patients who registered did not attend any part of the program.
Satisfaction
Most patients and partners said that they found the information presented at the modular education sessions comprehensive, clear, and easy to understand (Table 3). Although the overall average satisfaction score varied significantly across sessions, ranging from 3.5 (out of a possible 4) for pelvic-floor physical therapy to 3.8 for introduction to prostate cancer (F = 3.8, P < .001), the effect size of this difference was small (η2 = .039; Table 4A). We found no difference in the level of satisfaction between patients and partners, with the exception of the sexual health module, which was rated better by partners than by patients (patients: 3.6, partners: 3.8; t = 2.0; P = .03); however, the effect size of this difference was again small (Cohen d = .29). A total of 86% of patients found the inclusion of their partners at the sessions useful. For both pelvic-floor physical therapy and sexual health, attendees were more satisfied if they attended before treatment initiation rather than after completion (Table 4B).
Discussion
The purpose of this descriptive analysis was to outline a comprehensive, multidisciplinary supportive care program for men with prostate cancer and to present initial data on the population that has used the program and their satisfaction with the services provided. Within the first 5 years of the PCSC Program, 1,269 patients registered to participate. However, nearly 1 in 6 men who registered for the program did not subsequently attend any education sessions or use any clinical services offered, despite the fact that all services were free of charge. It is possible that nonparticipation may be related to men on active surveillance choosing not to engage with the program until they are faced with making a treatment decision, which may not happen until several years after an initial positive biopsy.26 This and other factors that affect a patient’s decision not to participate will be investigated in a future study. There is existing evidence documenting high levels of distress and anxiety for patients and their partners resulting from decision-making around prostate cancer treatment,27,28 and many face both decisional conflict and subsequent regret.15,29 Further work to help patients access the program could include defining a prehabilitation program for which patients can sign up that automatically selects the education sessions and clinical services most relevant to them.
The number of attendees varied across the 6 education sessions, with introduction to prostate cancer and sexual health being the best attended. This is consistent with the literature concerning the specific unmet supportive care and information needs in this population10,13 and with the value that men have placed on taking an active role in the decisions around their prostate cancer treatment.30 It is also possible that attendance varied because modules were introduced in a stepwise fashion and were offered on different schedules. Patients and partners both reported a high degree of satisfaction with all of the modules’ education sessions, reporting that the length, content, and delivery were appropriate.
Since 2013, a wide research portfolio has grown alongside the program. It has acted as a recruitment site for multicenter national studies and has attracted funding for several in-house research projects and evaluations. In addition, the VPC has implemented clinic-wide electronic collection of several patient-reported outcome measures using iPads. Patients have the option of contributing their data to Canadian (PC360o) and Global (TrueNTH Global Registry – Prostate Cancer Outcomes) registries for prostate cancer. The program has also created educational opportunities by supporting postdoctoral fellows. It has also provided a rich environment for urology and radiation oncology residents and fellows to participate in a multidisciplinary supportive care team, ensuring that the next generation of surgeons and oncologists recognize the importance of this approach to care.
Limitations
This is a brief descriptive study that relies on a mixture of anonymized survey and clinical chart data. Because the program’s patient feedback forms are anonymous, we are not able to link satisfaction scores to differences in sociodemographic, clinical, or prognostic factors. We also have not directly measured clinical, psychological, or quality of life outcomes; however, all 3 will be included in future studies of the program. An additional limitation is that not all program modules were offered for the entirety of the study duration and are offered at different frequencies. Thus, some modules have disproportionally higher participation rates than others. Lastly, we are missing clinical information for 16% of our participants who are not patients at the VPC.
The program is offered within an academic and teaching hospital in a major metropolitan center and depends on the work of a large interdisciplinary team. Cancer programs that are not embedded within a similar environment, such as those located in smaller rural communities, may not have access to the specialized clinical professionals who run our program, affecting its direct generalizability to these locations. Other specialists, such as palliative care teams, could be well positioned to provide support in locations that do not have a similar level of resource available. Furthermore, some program elements will be adapted to be delivered using telemedicine technology, which is an additional approach to improving access for patients who are beyond the reach of a tertiary care facility.
Conclusions
There is a growing need to provide consistent and comprehensive supportive care to patients with prostate cancer and their partners and families throughout the disease and treatment journey. The PCSC Program uses a multidisciplinary, evidenced-based, disease-focused approach to support informed treatment decision-making and address the physical, psychological, and psychosocial effects of prostate cancer diagnosis and treatment. We proactively collect data on disease, personal demographic details, and symptoms or quality of life, supporting opportunities to partner with researchers with the goal of further improving quality of life based on evidenced-based practices. Going forward, we will conduct detailed examinations of the costs and benefits (in terms of symptom management and quality of life) of the PCSC Program, further contributing to the development of evidence-based best practices for supportive care for men with prostate cancer and their families.
Acknowledgments
The authors express their gratitude to the urologists and radiation oncologists who referred their patients to the program and participated in delivering education sessions, including Dr Martin Gleave, Dr Peter Black, Dr Alan So, Dr Scott Tyldesley, and Dr Mira Keyes. They also thank Dr Richard Wassersug for his contributions to the initial program design and implementation. They thank the patients and their families for participating, and all of their current or past staff and collaborators. Lastly, they thank the funders of the program: the Specialist Services Committee, the BC Ministry of Health, the Prostate Cancer Foundation of BC, and philanthropic donors. They acknowledge Vancouver Coastal Health Research Institute and the University of British Columbia for their institutional support.
1. Cancer Research UK. Prostate cancer statistics. http://www.cancer.ca/en/cancer-information/cancer-type/prostate/statistics/?region=sk. Published 2015. Accessed June 22, 2017.
2. Siegel R, DeSantis C, Virgo K, et al. Cancer treatment and survivorship statistics, 2012. CA Cancer J Clin. 2012;62(4):220-241.
3. Canadian Cancer Society. Canadian cancer statistics special topic: predictions of the future burden of cancer in Canada. Ottawa, Canada: Public Health Agency of Canada; 2015.
4. Roth AJ, Weinberger MI, Nelson CJ. Prostate cancer: psychosocial implications and management. Future Oncol. 2008;4(4):561-568.
5. Couper J, Bloch S, Love A, Macvean M, Duchesne GM, Kissane D. Psychosocial adjustment of female partners of men with prostate cancer: a review of the literature. Psychooncology 2006;15(11):937-953.
6. Wilt TJ, MacDonald R, Rutks I, Shamliyan TA, Taylor BC, Kane RL. Systematic review: comparative effectiveness and harms of treatments for clinically localized prostate cancer. Ann Intern Med. 2008;148(6):435-448.
7. Galvão DA, Spry NA, Taaffe DR, et al. Changes in muscle, fat and bone mass after 36 weeks of maximal androgen blockade for prostate cancer. BJU Int. 2008;102(1):44-47.
8. Watts S, Leydon G, Birch B, et al. Depression and anxiety in prostate cancer: a systematic review and meta-analysis of prevalence rates. BMJ Open. 2014;4(3):e003901.
9. Zaider T, Manne S, Nelson C, Mulhall J, Kissane D. Loss of masculine identity, marital affection, and sexual bother in men with localized prostate cancer. J Sex Med. 2012;9(10):2724-2732.
10. Ream E, Quennell A, Fincham L, et al. Supportive care needs of men living with prostate cancer in England: a survey. Br J Cancer. 2008;98(12):1903-1909.
11. Howell D, Hack TF, Oliver TK, et al. Models of care for post-treatment follow-up of adult cancer survivors: a systematic review and quality appraisal of the evidence. J Cancer Surviv. 2012;6(4):359-371.
12. Halpern MT, Viswanathan M, Evans TS, Birken SA, Basch E, Mayer DK. Models of cancer survivorship care: overview and summary of current evidence. J Oncol Pract. 2015;11(1):e19-e27.
13. Smith DP, Supramaniam R, King MT, Ward J, Berry M, Armstrong BK. Age, health, and education determine supportive care needs of men younger than 70 years with prostate cancer. J Clin Oncol. 2007;25(18):2560-2566.
14. Northouse LL, Mood DW, Montie JE, et al. Living with prostate cancer: patients’ and spouses’ psychosocial status and quality of life. J Clin Oncol. 2007;25(27):4171-4177.
15. Hedden L, Wassersug R, Mahovlich S, et al. Evaluating an educational intervention to alleviate distress amongst men with newly diagnosed prostate cancer and their partners. BJU Int. 2017;120(5B):E21-E29.
16. Bradley EB, Bissonette EA, Theodorescu D. Determinants of long-term quality of life and voiding function of patients treated with radical prostatectomy or permanent brachytherapy for prostate cancer. BJU Int. 2004;94(7):1003-1009.
17. Ramsey SD, Zeliadt SB, Blough DK, et al. Impact of prostate cancer on sexual relationships: a longitudinal perspective on intimate partners’ experiences. J Sex Med. 2013;10(12):3135-3143.
18. Wittmann D, Carolan M, Given B, et al. Exploring the role of the partner in couples’ sexual recovery after surgery for prostate cancer. Support Care Cancer. 2014;22(9):2509-2515.
19. Schmitz KH, Courneya KS, Matthews C, et al. American college of sports medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42(7):1409-1426.
20. Rock CL, Doyle C, Demark-Wahnefried W, et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin. 2012;62(4):243-274.
21. Elliott S, Latini DM, Walker LM, Wassersug R, Robinson JW; ADT Suvivorship Working Group. Androgen deprivation therapy for prostate cancer: recommendations to improve patient and partner quality of life. J Sex Med. 2010;7(9):2996-3010.
22. Wassersug RJ, Walker LM, Robinson JW. Androgen deprivation therapy: an essential guide for prostate cancer patients and their loved ones. New York, NY: Demos Health; 2014.
23. Wibowo E, Walker LM, Wilyman S, et al. Androgen deprivation therapy educational program: a Canadian True NTH initiative. J Clin Oncol. 2016;34(suppl 3):243.
24. Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer. JAMA. 2000;283(3):354-360.
25. Overgård M, Angelsen A, Lydersen S, Mørkved S. Does physiotherapist-guided pelvic floor muscle training reduce urinary incontinence after radical prostatectomy? A randomised controlled trial. Eur Urol. 2008;54(2):438-448.
26. Godtman RA, Holmberg E, Khatami A, Pihl CG, Stranne J, Hugosson J. Long-term results of active surveillance in the Göteborg randomized, population-based prostate cancer screening trial. Eur Urol. 2016;70(5):760-766.
27. Cohen H, Britten N. Who decides about prostate cancer treatment? A qualitative study. Fam Pract. 2003;20(6):724-729.
28. Denberg TD, Melhado TV, Steiner JF. Patient treatment preferences in localized prostate carcinoma: the influence of emotion, misconception, and anecdote. Cancer. 2006;107(3):620-630.
29. Morris BB, Farnan L, Song L, et al. Treatment decisional regret among men with prostate cancer: racial differences and influential factors in the North Carolina health access and prostate cancer treatment project (HCaP-NC). Cancer. 2015;121(12):2029-2035.
30. Feldman-Stewart D, Capirci C, Brennenstuhl S, et al. Information for decision making by patients with early-stage prostate cancer: a comparison across 9 countries. Med Decis Making. 2011;31(5):754-766.
Prostate cancer is the most common malignancy diagnosed in Canadian men. An estimated 21,300 Canadian men were diagnosed with the disease in 2017, representing 21% of all new cancer cases.1 There are about 176,000 men living with prostate cancer in Canada.1 In the United States, there were 2,778,630 survivors of prostate cancer as of 2012 and that population is expected to increase by more than 1 million (40%) to 3,922,600 by 2022.2
Although 96% of men diagnosed with prostate cancer now survive longer than 5 years3, many will suffer from treatment-related sequelae that can have a profound effect on quality of life for themselves and their partners.4,5 Impacts include sexual, urinary, and bowel dysfunctions6 owing to treatment of the primary tumor as well as reduced muscle and bone mass, osteoporosis, fatigue, obesity, and glucose intolerance or diabetes7 owing to androgen-deprivation therapy (ADT). Many men also suffer from psychological issues such as depression, anxiety, anger and irritability, sense of isolation, grief, and loss of masculinity.8,9 The psychological impacts also continue well beyond the completion of treatment and can be significant for both patients and their partners.5,8
With posttreatment longevity and the associated complex sequelae, prostate cancer is being viewed increasingly as a chronic disease whose effects must be managed for many years after the completion of primary treatment. Supportive care that “[manages] symptoms and side effects, enables adaptation and coping, optimizes understanding and informed decision-making, and minimizes decrements in functioning”10 is becoming recognized as a critical component of direct oncologic care before, during, and after treatment. Health care professionals, scientists, governments, and patient advocates are increasingly calling for the development of comprehensive supportive care programs improve the quality of life for people diagnosed with cancer. A common model for survivorship care is a general program for all cancer survivors that provides disease- and patient-specific care plans. These care plans outline patients’ prior therapies, potential side effects, recommendations for monitoring (for side effects or relapse of cancer), and advice on how patients can maintain a healthy lifestyle.11 However, there are few survivorship programs for men with prostate cancer and their partners, and the evidence base around best practices for these programs is scant.12 Furthermore, up to 87% of men with a prostate cancer diagnosis report specific and significant unmet supportive care needs,10,13 with sexuality-related and psychological issues10,14 being the areas of greatest concern.
To address the complex supportive care needs of men with prostate cancer in British Columbia, Canada, the Vancouver Prostate Centre (VPC) and Department of Urologic Sciences at the University of British Columbia developed the multidisciplinary Prostate Cancer Supportive Care (PCSC) Program. The program aims to address the challenges of decision-making and coping faced by men with prostate cancer and their partners and family members along the entire disease trajectory. Services are provided at no cost to participants. Here, we outline the guiding principles for the PCSC program and its scope, delivery, and evaluation. We provide information on the more than 1,200 patients who have participated in the program since its inception in January of 2013, the rates of participation across the different program modules, and a selection of patient satisfaction measures. We also discuss successes and limitations and ongoing research and evaluation efforts, providing lessons learned to support the development of other supportive care programs in Canada and internationally.
Program description
Guiding principles
The PCSC Program is a clinical, educational, and research-based program, with 4 guiding principles: it is comprehensive, patient- and partner-centered, evidence-based, and supports new research. The program serves patients, partners, and families along the entire disease trajectory, recognizing that cancer is a family disease, affecting both the individual and social network, and that the psychological stress associated with a diagnosis of prostate cancer is borne heavily by partners. It has been designed, implemented, and refined with the best available evidence and with the intention to undergo consistent and repeated evaluation. Finally, it was designed to provide opportunities for targeted research efforts, supporting the growth of the evidence base in this area.
Patient entry and module descriptions
Patients can be referred to the program by a physician or other allied health professional. They may also self-refer, having been made aware of the program through our website, a variety of print materials, or by word of mouth. On referral, the program coordinator collects patients’ basic clinical and demographic data, assesses health literacy and lifestyle factors, and provides them with information on the program modules. As of December 2015, the program consisted of 6 distinct modules, each focusing on different elements of the disease trajectory or on addressing specific physical or mental health concerns. Modules are led by licensed health professionals with experience in oncology. No elements of the program are mandatory, and participants are free to pick and choose the components that are most relevant to them and their partners.
Introduction to prostate cancer and primary treatment options. This is a group-based module that focuses on educating newly diagnosed patients (and those going on or off active surveillance) on the basic biology of prostate cancer, the primary treatment options for localized disease, and the main side effects associated with the treatments. It also includes information about the other services offered by the program and any ongoing research studies. The session is held twice a month in the early evening and is run collectively by a urologist, radiation oncologist, patient representative, and program coordinator. It includes a brief one-on-one discussion between each patient and their partner or family member and the urologist and radiation oncologist to address any remaining questions. A copy of the patient’s biopsy report is on hand for the physician(s). Attendance of this session has been shown to significantly reduce pretreatment distress in both patients and their partners.15
Managing sexual function and intimacy. Sexual intimacy is tied to overall health outcomes, relationship satisfaction, and quality of life.16 Primary therapy for prostate cancer can be associated with substantial side-effects (eg, erectile dysfunction, incontinence, altered libido, penile shortening) that negatively affect sexual intimacy and have an impact on the patient individually as well as the sexual relationship he has with his partner.17
The program’s Sexual Health Service (SHS) provides patients and partners with information on the impact of treatment on sexual health.18 The SHS offers educational sessions led by a sexual rehabilitation nurse and clinical psychologist with a specialization in sexual health. Sessions focus on the impact of prostate cancer treatments on sexual function and therapeutic modalities, promote an understanding of the barriers to sexual adaptation posttreatment, and present options for sexual activity that are not solely dependent on the ability to achieve an erection. Once participants have attended an educational session, they are offered individual consultations with the sexual health nurse every 3 to 6 months for 2 years or longer, depending on the patient’s or couple’s needs. They are referred to the SHS’s sexual medicine physician if further medical intervention is warranted. The sexual health nurse works with the patient and partner to develop an individualized Sexual Health Rehabilitation Action Plan (SHRAP), which assists the couple in sexual adaptation going forward. The SHRAP is a tool devised by the sexual health nurse based on her clinical experience with couples affected by prostate cancer.
Couples who have been evaluated within the SHS are also invited to attend a second workshop on intimacy that is offered quarterly. Workshop participants discuss the impact of sexual changes on relationships, and strategies on how to enhance intimacy and sexual communication are presented. A resource package is provided to each couple to help re-establish and/or strengthen their various dimensions of intimacy.
Lifestyle management. The lifestyle management modules include separate nutrition and physical activity or exercise components. Referral to the smoking cessation program in the Vancouver Coastal Health Authority is made at program registration, if appropriate. The nutrition group-based education session is delivered by a registered dietitian from the British Columbia Cancer Agency who specializes in prostate cancer. The session focuses on evidence-based recommendations on diet after a diagnosis of prostate cancer, the use of dietary supplements, body weight and health, and practical nutrition tips. The exercise session is delivered by an exercise physiologist who specializes in working with cancer patients. It covers the value of exercise in terms of safety, prevention and reduction of treatment side effects (including from ADT), treatment prehabilitation and recovery, advanced cancer management, and long-term survival. A one-on-one exercise counseling clinic is also offered and aims to increase exercise adoption and long-term adherence in line with Canadian Physical Activity guidelines and exercise oncology guidelines,19,20 with follow-up appointments at 3, 6, and 12 months to help patients stay motivated and ensure they are exercising correctly. The individual consultations with the exercise physiologist include physical measures, exercise volume, treatment side effects, and coconstructed goal setting with an individualized formal exercise regimen (exercise prescription).
Adapting to ADT. This is an educational module offered to patients with metastatic prostate cancer who are starting hormone therapy treatments that lower serum testosterone into the castrate range. This program was one of several available through TrueNTH, a portfolio of projects funded by the Movember Foundation, through Prostate Cancer Canada. The session is delivered by a patient facilitator and focuses on strategies to recognize and adapt to the side effects of ADT21 while maintaining a good quality of life and strong intimate relationships with partners.22,23
Pelvic-floor physical therapy for urinary incontinence. This module includes a group-based and individualized education session for patients (either pre- or posttreatment) focused on reducing the effects of surgery and/or radiation therapy on urinary and sexual continence as well as on how to cope with these symptoms and minimize the effect they have on quality of life.24 The session is conducted by a physical therapist who is certified as a pelvic-floor specialist. Supervised pelvic-floor re-education and/or exercise has been shown to successfully reduce the degree of incontinence in this population.25 The module therefore also includes 3 one-on-one clinical appointments for patients who are still experiencing bother from incontinence 12 weeks after a radical prostatectomy or postradiation treatment.
Psycho-oncology. In recognition of the emotional and psychological burden associated with prostate cancer and the important role partners play in facilitating treatment of these psychological and/or psychosocial issues, the program offers appointments with a registered clinical counselor to address acute emotional distress. These are usually 1-hour appointments offered to both patients and partners, either separately or together. Appointments can be attended in person or conducted by telephone. When appropriate, patients are referred for further long-term individual support or couple support with their partners. A group therapy workshop was also initiated in 2016. In this program, men participate in a guided autobiographical life review through a process that focuses on developing a cohesive working group, learning strategic communication skills, and understanding and learning how to manage difficult emotions and transitional life stressors associated with prostate cancer. It also focuses on processing and integrating critical events that contribute to the men’s identity and psychological function and involves the consolidation of the personal learning that occurs. Postgroup referral plans are developed on an individual basis as needed.
Methods
Data
We obtained sociodemographic, diagnostic, and treatment information as well as clinic visit records for all PCSC Program registrants from the electronic medical record held at the VPC. Clinical variables included age at diagnosis, Gleason score, and primary treatment modality (including active surveillance and ADT use). The Gleason score determines the aggressiveness of a patient’s prostate cancer based on biopsy results. A score of 6 or less indicates that the disease is likely to grow slowly. A grade of 7 is considered intermediate risk (with primary score of 3 and secondary 4 being lower risk than those with a primary score of 4 and secondary of 3). A Gleason score of 8 or higher indicates aggressive disease that is poorly differentiated or high grade. Sociodemographic characteristics included age, travel distance to the clinic, and income quintile. We obtained attendance records for the modular education sessions from the program’s database. Patients who did not have any medical visits at the VPC (referred to henceforth as non-VPC patients) did not have a clinic record, so we excluded them from the subset of the analyses that depended on specific clinical variables.
All patients and partners who participate in any PCSC Program education sessions (introduction to prostate cancer, sexual health, nutrition, exercise, ADT, and pelvic-floor physical therapy) are asked to complete voluntary, anonymous feedback forms. These forms assess participant satisfaction using a series of Likert-based and Boolean response items as well as qualitative commentary. They include questions that assess the timing, structure, and content of each session.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Statistical approach
Descriptive statistics were used to analyze participant characteristics, program participation rates, and participant satisfaction. For each module’s education session, we compared the overall satisfaction between patients and partners using t tests. We also compared the level of satisfaction across the different modules using a 1-way analysis of variance. For the sexual health and pelvic-floor physical therapy sessions, we compared satisfaction between participants who attended the education sessions before to those who attended following their primary treatment using t tests. We provide the eta squared (for analyses of variance) and Cohen d (for t tests) to provide an effect size estimate of any significant differences observed.
Results
Participants
From the program’s founding in January of 2013 to December 31, 2016, a total of 1,269 patients registered (an average of 317 patients a year). Of those, 1,026 (80.9%) had at least 1 prostate cancer–related visit at the VPC. The remaining 243 (19.1%) were non-VPC patients (Figure). Overall, 1,062 men (83.4%) who registered with the program went on to attend at least 1 education session or clinic appointment.
Average age among male program participants was 67.7 years, and age at diagnosis was 62.5 years (Table 1). In all, 273 men (31.7%) had Gleason 3+4, and 117 (13.7%) had Gleason 4+3. Most of the participants (76.9%) elected to undergo radical prostatectomy for primary treatment. Ninety-five men (8.9%) received at least some ADT treatment as an adjunct to radiation or to treat recurrent disease. Participants traveled an average of 83.1 km (51.6 miles; median, 6.9 km and 10.5 miles, respectively) to attend the program; 10% of participants traveled further than 112 km (70 mi) to the clinic. One hundred and four (10.9%) and 301 (31.5%) of registrants were in the lowest and highest income quintiles respectively. Four hundred and ninety-seven (46.8%) attended at lesson 1 session or clinic appointment with a partner or family member.
Program participation
Of the 1,062 men who participated in the program, 867 (80.1%) were patients of the VPC, and 205 (19.1%) were non-VPC patients. The education sessions for the introduction to prostate cancer and sexual health modules had the largest numbers of participants (309 and 265, respectively; Table 2); however, pelvic-floor physical therapy had the highest participation rate per quarter (25 patients). The clinical services offered within the sexual health module had the larger number of participants and highest participation rate per quarter (590 total patients, 42/quarter). Timing of program participation was highly variable, ranging from 6 days to 18.5 years after diagnosis (SD, 1,301 days). More than half of participants attended a session or clinic visit within the first year of their diagnosis. A total of 17% of patients who registered did not attend any part of the program.
Satisfaction
Most patients and partners said that they found the information presented at the modular education sessions comprehensive, clear, and easy to understand (Table 3). Although the overall average satisfaction score varied significantly across sessions, ranging from 3.5 (out of a possible 4) for pelvic-floor physical therapy to 3.8 for introduction to prostate cancer (F = 3.8, P < .001), the effect size of this difference was small (η2 = .039; Table 4A). We found no difference in the level of satisfaction between patients and partners, with the exception of the sexual health module, which was rated better by partners than by patients (patients: 3.6, partners: 3.8; t = 2.0; P = .03); however, the effect size of this difference was again small (Cohen d = .29). A total of 86% of patients found the inclusion of their partners at the sessions useful. For both pelvic-floor physical therapy and sexual health, attendees were more satisfied if they attended before treatment initiation rather than after completion (Table 4B).
Discussion
The purpose of this descriptive analysis was to outline a comprehensive, multidisciplinary supportive care program for men with prostate cancer and to present initial data on the population that has used the program and their satisfaction with the services provided. Within the first 5 years of the PCSC Program, 1,269 patients registered to participate. However, nearly 1 in 6 men who registered for the program did not subsequently attend any education sessions or use any clinical services offered, despite the fact that all services were free of charge. It is possible that nonparticipation may be related to men on active surveillance choosing not to engage with the program until they are faced with making a treatment decision, which may not happen until several years after an initial positive biopsy.26 This and other factors that affect a patient’s decision not to participate will be investigated in a future study. There is existing evidence documenting high levels of distress and anxiety for patients and their partners resulting from decision-making around prostate cancer treatment,27,28 and many face both decisional conflict and subsequent regret.15,29 Further work to help patients access the program could include defining a prehabilitation program for which patients can sign up that automatically selects the education sessions and clinical services most relevant to them.
The number of attendees varied across the 6 education sessions, with introduction to prostate cancer and sexual health being the best attended. This is consistent with the literature concerning the specific unmet supportive care and information needs in this population10,13 and with the value that men have placed on taking an active role in the decisions around their prostate cancer treatment.30 It is also possible that attendance varied because modules were introduced in a stepwise fashion and were offered on different schedules. Patients and partners both reported a high degree of satisfaction with all of the modules’ education sessions, reporting that the length, content, and delivery were appropriate.
Since 2013, a wide research portfolio has grown alongside the program. It has acted as a recruitment site for multicenter national studies and has attracted funding for several in-house research projects and evaluations. In addition, the VPC has implemented clinic-wide electronic collection of several patient-reported outcome measures using iPads. Patients have the option of contributing their data to Canadian (PC360o) and Global (TrueNTH Global Registry – Prostate Cancer Outcomes) registries for prostate cancer. The program has also created educational opportunities by supporting postdoctoral fellows. It has also provided a rich environment for urology and radiation oncology residents and fellows to participate in a multidisciplinary supportive care team, ensuring that the next generation of surgeons and oncologists recognize the importance of this approach to care.
Limitations
This is a brief descriptive study that relies on a mixture of anonymized survey and clinical chart data. Because the program’s patient feedback forms are anonymous, we are not able to link satisfaction scores to differences in sociodemographic, clinical, or prognostic factors. We also have not directly measured clinical, psychological, or quality of life outcomes; however, all 3 will be included in future studies of the program. An additional limitation is that not all program modules were offered for the entirety of the study duration and are offered at different frequencies. Thus, some modules have disproportionally higher participation rates than others. Lastly, we are missing clinical information for 16% of our participants who are not patients at the VPC.
The program is offered within an academic and teaching hospital in a major metropolitan center and depends on the work of a large interdisciplinary team. Cancer programs that are not embedded within a similar environment, such as those located in smaller rural communities, may not have access to the specialized clinical professionals who run our program, affecting its direct generalizability to these locations. Other specialists, such as palliative care teams, could be well positioned to provide support in locations that do not have a similar level of resource available. Furthermore, some program elements will be adapted to be delivered using telemedicine technology, which is an additional approach to improving access for patients who are beyond the reach of a tertiary care facility.
Conclusions
There is a growing need to provide consistent and comprehensive supportive care to patients with prostate cancer and their partners and families throughout the disease and treatment journey. The PCSC Program uses a multidisciplinary, evidenced-based, disease-focused approach to support informed treatment decision-making and address the physical, psychological, and psychosocial effects of prostate cancer diagnosis and treatment. We proactively collect data on disease, personal demographic details, and symptoms or quality of life, supporting opportunities to partner with researchers with the goal of further improving quality of life based on evidenced-based practices. Going forward, we will conduct detailed examinations of the costs and benefits (in terms of symptom management and quality of life) of the PCSC Program, further contributing to the development of evidence-based best practices for supportive care for men with prostate cancer and their families.
Acknowledgments
The authors express their gratitude to the urologists and radiation oncologists who referred their patients to the program and participated in delivering education sessions, including Dr Martin Gleave, Dr Peter Black, Dr Alan So, Dr Scott Tyldesley, and Dr Mira Keyes. They also thank Dr Richard Wassersug for his contributions to the initial program design and implementation. They thank the patients and their families for participating, and all of their current or past staff and collaborators. Lastly, they thank the funders of the program: the Specialist Services Committee, the BC Ministry of Health, the Prostate Cancer Foundation of BC, and philanthropic donors. They acknowledge Vancouver Coastal Health Research Institute and the University of British Columbia for their institutional support.
Prostate cancer is the most common malignancy diagnosed in Canadian men. An estimated 21,300 Canadian men were diagnosed with the disease in 2017, representing 21% of all new cancer cases.1 There are about 176,000 men living with prostate cancer in Canada.1 In the United States, there were 2,778,630 survivors of prostate cancer as of 2012 and that population is expected to increase by more than 1 million (40%) to 3,922,600 by 2022.2
Although 96% of men diagnosed with prostate cancer now survive longer than 5 years3, many will suffer from treatment-related sequelae that can have a profound effect on quality of life for themselves and their partners.4,5 Impacts include sexual, urinary, and bowel dysfunctions6 owing to treatment of the primary tumor as well as reduced muscle and bone mass, osteoporosis, fatigue, obesity, and glucose intolerance or diabetes7 owing to androgen-deprivation therapy (ADT). Many men also suffer from psychological issues such as depression, anxiety, anger and irritability, sense of isolation, grief, and loss of masculinity.8,9 The psychological impacts also continue well beyond the completion of treatment and can be significant for both patients and their partners.5,8
With posttreatment longevity and the associated complex sequelae, prostate cancer is being viewed increasingly as a chronic disease whose effects must be managed for many years after the completion of primary treatment. Supportive care that “[manages] symptoms and side effects, enables adaptation and coping, optimizes understanding and informed decision-making, and minimizes decrements in functioning”10 is becoming recognized as a critical component of direct oncologic care before, during, and after treatment. Health care professionals, scientists, governments, and patient advocates are increasingly calling for the development of comprehensive supportive care programs improve the quality of life for people diagnosed with cancer. A common model for survivorship care is a general program for all cancer survivors that provides disease- and patient-specific care plans. These care plans outline patients’ prior therapies, potential side effects, recommendations for monitoring (for side effects or relapse of cancer), and advice on how patients can maintain a healthy lifestyle.11 However, there are few survivorship programs for men with prostate cancer and their partners, and the evidence base around best practices for these programs is scant.12 Furthermore, up to 87% of men with a prostate cancer diagnosis report specific and significant unmet supportive care needs,10,13 with sexuality-related and psychological issues10,14 being the areas of greatest concern.
To address the complex supportive care needs of men with prostate cancer in British Columbia, Canada, the Vancouver Prostate Centre (VPC) and Department of Urologic Sciences at the University of British Columbia developed the multidisciplinary Prostate Cancer Supportive Care (PCSC) Program. The program aims to address the challenges of decision-making and coping faced by men with prostate cancer and their partners and family members along the entire disease trajectory. Services are provided at no cost to participants. Here, we outline the guiding principles for the PCSC program and its scope, delivery, and evaluation. We provide information on the more than 1,200 patients who have participated in the program since its inception in January of 2013, the rates of participation across the different program modules, and a selection of patient satisfaction measures. We also discuss successes and limitations and ongoing research and evaluation efforts, providing lessons learned to support the development of other supportive care programs in Canada and internationally.
Program description
Guiding principles
The PCSC Program is a clinical, educational, and research-based program, with 4 guiding principles: it is comprehensive, patient- and partner-centered, evidence-based, and supports new research. The program serves patients, partners, and families along the entire disease trajectory, recognizing that cancer is a family disease, affecting both the individual and social network, and that the psychological stress associated with a diagnosis of prostate cancer is borne heavily by partners. It has been designed, implemented, and refined with the best available evidence and with the intention to undergo consistent and repeated evaluation. Finally, it was designed to provide opportunities for targeted research efforts, supporting the growth of the evidence base in this area.
Patient entry and module descriptions
Patients can be referred to the program by a physician or other allied health professional. They may also self-refer, having been made aware of the program through our website, a variety of print materials, or by word of mouth. On referral, the program coordinator collects patients’ basic clinical and demographic data, assesses health literacy and lifestyle factors, and provides them with information on the program modules. As of December 2015, the program consisted of 6 distinct modules, each focusing on different elements of the disease trajectory or on addressing specific physical or mental health concerns. Modules are led by licensed health professionals with experience in oncology. No elements of the program are mandatory, and participants are free to pick and choose the components that are most relevant to them and their partners.
Introduction to prostate cancer and primary treatment options. This is a group-based module that focuses on educating newly diagnosed patients (and those going on or off active surveillance) on the basic biology of prostate cancer, the primary treatment options for localized disease, and the main side effects associated with the treatments. It also includes information about the other services offered by the program and any ongoing research studies. The session is held twice a month in the early evening and is run collectively by a urologist, radiation oncologist, patient representative, and program coordinator. It includes a brief one-on-one discussion between each patient and their partner or family member and the urologist and radiation oncologist to address any remaining questions. A copy of the patient’s biopsy report is on hand for the physician(s). Attendance of this session has been shown to significantly reduce pretreatment distress in both patients and their partners.15
Managing sexual function and intimacy. Sexual intimacy is tied to overall health outcomes, relationship satisfaction, and quality of life.16 Primary therapy for prostate cancer can be associated with substantial side-effects (eg, erectile dysfunction, incontinence, altered libido, penile shortening) that negatively affect sexual intimacy and have an impact on the patient individually as well as the sexual relationship he has with his partner.17
The program’s Sexual Health Service (SHS) provides patients and partners with information on the impact of treatment on sexual health.18 The SHS offers educational sessions led by a sexual rehabilitation nurse and clinical psychologist with a specialization in sexual health. Sessions focus on the impact of prostate cancer treatments on sexual function and therapeutic modalities, promote an understanding of the barriers to sexual adaptation posttreatment, and present options for sexual activity that are not solely dependent on the ability to achieve an erection. Once participants have attended an educational session, they are offered individual consultations with the sexual health nurse every 3 to 6 months for 2 years or longer, depending on the patient’s or couple’s needs. They are referred to the SHS’s sexual medicine physician if further medical intervention is warranted. The sexual health nurse works with the patient and partner to develop an individualized Sexual Health Rehabilitation Action Plan (SHRAP), which assists the couple in sexual adaptation going forward. The SHRAP is a tool devised by the sexual health nurse based on her clinical experience with couples affected by prostate cancer.
Couples who have been evaluated within the SHS are also invited to attend a second workshop on intimacy that is offered quarterly. Workshop participants discuss the impact of sexual changes on relationships, and strategies on how to enhance intimacy and sexual communication are presented. A resource package is provided to each couple to help re-establish and/or strengthen their various dimensions of intimacy.
Lifestyle management. The lifestyle management modules include separate nutrition and physical activity or exercise components. Referral to the smoking cessation program in the Vancouver Coastal Health Authority is made at program registration, if appropriate. The nutrition group-based education session is delivered by a registered dietitian from the British Columbia Cancer Agency who specializes in prostate cancer. The session focuses on evidence-based recommendations on diet after a diagnosis of prostate cancer, the use of dietary supplements, body weight and health, and practical nutrition tips. The exercise session is delivered by an exercise physiologist who specializes in working with cancer patients. It covers the value of exercise in terms of safety, prevention and reduction of treatment side effects (including from ADT), treatment prehabilitation and recovery, advanced cancer management, and long-term survival. A one-on-one exercise counseling clinic is also offered and aims to increase exercise adoption and long-term adherence in line with Canadian Physical Activity guidelines and exercise oncology guidelines,19,20 with follow-up appointments at 3, 6, and 12 months to help patients stay motivated and ensure they are exercising correctly. The individual consultations with the exercise physiologist include physical measures, exercise volume, treatment side effects, and coconstructed goal setting with an individualized formal exercise regimen (exercise prescription).
Adapting to ADT. This is an educational module offered to patients with metastatic prostate cancer who are starting hormone therapy treatments that lower serum testosterone into the castrate range. This program was one of several available through TrueNTH, a portfolio of projects funded by the Movember Foundation, through Prostate Cancer Canada. The session is delivered by a patient facilitator and focuses on strategies to recognize and adapt to the side effects of ADT21 while maintaining a good quality of life and strong intimate relationships with partners.22,23
Pelvic-floor physical therapy for urinary incontinence. This module includes a group-based and individualized education session for patients (either pre- or posttreatment) focused on reducing the effects of surgery and/or radiation therapy on urinary and sexual continence as well as on how to cope with these symptoms and minimize the effect they have on quality of life.24 The session is conducted by a physical therapist who is certified as a pelvic-floor specialist. Supervised pelvic-floor re-education and/or exercise has been shown to successfully reduce the degree of incontinence in this population.25 The module therefore also includes 3 one-on-one clinical appointments for patients who are still experiencing bother from incontinence 12 weeks after a radical prostatectomy or postradiation treatment.
Psycho-oncology. In recognition of the emotional and psychological burden associated with prostate cancer and the important role partners play in facilitating treatment of these psychological and/or psychosocial issues, the program offers appointments with a registered clinical counselor to address acute emotional distress. These are usually 1-hour appointments offered to both patients and partners, either separately or together. Appointments can be attended in person or conducted by telephone. When appropriate, patients are referred for further long-term individual support or couple support with their partners. A group therapy workshop was also initiated in 2016. In this program, men participate in a guided autobiographical life review through a process that focuses on developing a cohesive working group, learning strategic communication skills, and understanding and learning how to manage difficult emotions and transitional life stressors associated with prostate cancer. It also focuses on processing and integrating critical events that contribute to the men’s identity and psychological function and involves the consolidation of the personal learning that occurs. Postgroup referral plans are developed on an individual basis as needed.
Methods
Data
We obtained sociodemographic, diagnostic, and treatment information as well as clinic visit records for all PCSC Program registrants from the electronic medical record held at the VPC. Clinical variables included age at diagnosis, Gleason score, and primary treatment modality (including active surveillance and ADT use). The Gleason score determines the aggressiveness of a patient’s prostate cancer based on biopsy results. A score of 6 or less indicates that the disease is likely to grow slowly. A grade of 7 is considered intermediate risk (with primary score of 3 and secondary 4 being lower risk than those with a primary score of 4 and secondary of 3). A Gleason score of 8 or higher indicates aggressive disease that is poorly differentiated or high grade. Sociodemographic characteristics included age, travel distance to the clinic, and income quintile. We obtained attendance records for the modular education sessions from the program’s database. Patients who did not have any medical visits at the VPC (referred to henceforth as non-VPC patients) did not have a clinic record, so we excluded them from the subset of the analyses that depended on specific clinical variables.
All patients and partners who participate in any PCSC Program education sessions (introduction to prostate cancer, sexual health, nutrition, exercise, ADT, and pelvic-floor physical therapy) are asked to complete voluntary, anonymous feedback forms. These forms assess participant satisfaction using a series of Likert-based and Boolean response items as well as qualitative commentary. They include questions that assess the timing, structure, and content of each session.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Statistical approach
Descriptive statistics were used to analyze participant characteristics, program participation rates, and participant satisfaction. For each module’s education session, we compared the overall satisfaction between patients and partners using t tests. We also compared the level of satisfaction across the different modules using a 1-way analysis of variance. For the sexual health and pelvic-floor physical therapy sessions, we compared satisfaction between participants who attended the education sessions before to those who attended following their primary treatment using t tests. We provide the eta squared (for analyses of variance) and Cohen d (for t tests) to provide an effect size estimate of any significant differences observed.
Results
Participants
From the program’s founding in January of 2013 to December 31, 2016, a total of 1,269 patients registered (an average of 317 patients a year). Of those, 1,026 (80.9%) had at least 1 prostate cancer–related visit at the VPC. The remaining 243 (19.1%) were non-VPC patients (Figure). Overall, 1,062 men (83.4%) who registered with the program went on to attend at least 1 education session or clinic appointment.
Average age among male program participants was 67.7 years, and age at diagnosis was 62.5 years (Table 1). In all, 273 men (31.7%) had Gleason 3+4, and 117 (13.7%) had Gleason 4+3. Most of the participants (76.9%) elected to undergo radical prostatectomy for primary treatment. Ninety-five men (8.9%) received at least some ADT treatment as an adjunct to radiation or to treat recurrent disease. Participants traveled an average of 83.1 km (51.6 miles; median, 6.9 km and 10.5 miles, respectively) to attend the program; 10% of participants traveled further than 112 km (70 mi) to the clinic. One hundred and four (10.9%) and 301 (31.5%) of registrants were in the lowest and highest income quintiles respectively. Four hundred and ninety-seven (46.8%) attended at lesson 1 session or clinic appointment with a partner or family member.
Program participation
Of the 1,062 men who participated in the program, 867 (80.1%) were patients of the VPC, and 205 (19.1%) were non-VPC patients. The education sessions for the introduction to prostate cancer and sexual health modules had the largest numbers of participants (309 and 265, respectively; Table 2); however, pelvic-floor physical therapy had the highest participation rate per quarter (25 patients). The clinical services offered within the sexual health module had the larger number of participants and highest participation rate per quarter (590 total patients, 42/quarter). Timing of program participation was highly variable, ranging from 6 days to 18.5 years after diagnosis (SD, 1,301 days). More than half of participants attended a session or clinic visit within the first year of their diagnosis. A total of 17% of patients who registered did not attend any part of the program.
Satisfaction
Most patients and partners said that they found the information presented at the modular education sessions comprehensive, clear, and easy to understand (Table 3). Although the overall average satisfaction score varied significantly across sessions, ranging from 3.5 (out of a possible 4) for pelvic-floor physical therapy to 3.8 for introduction to prostate cancer (F = 3.8, P < .001), the effect size of this difference was small (η2 = .039; Table 4A). We found no difference in the level of satisfaction between patients and partners, with the exception of the sexual health module, which was rated better by partners than by patients (patients: 3.6, partners: 3.8; t = 2.0; P = .03); however, the effect size of this difference was again small (Cohen d = .29). A total of 86% of patients found the inclusion of their partners at the sessions useful. For both pelvic-floor physical therapy and sexual health, attendees were more satisfied if they attended before treatment initiation rather than after completion (Table 4B).
Discussion
The purpose of this descriptive analysis was to outline a comprehensive, multidisciplinary supportive care program for men with prostate cancer and to present initial data on the population that has used the program and their satisfaction with the services provided. Within the first 5 years of the PCSC Program, 1,269 patients registered to participate. However, nearly 1 in 6 men who registered for the program did not subsequently attend any education sessions or use any clinical services offered, despite the fact that all services were free of charge. It is possible that nonparticipation may be related to men on active surveillance choosing not to engage with the program until they are faced with making a treatment decision, which may not happen until several years after an initial positive biopsy.26 This and other factors that affect a patient’s decision not to participate will be investigated in a future study. There is existing evidence documenting high levels of distress and anxiety for patients and their partners resulting from decision-making around prostate cancer treatment,27,28 and many face both decisional conflict and subsequent regret.15,29 Further work to help patients access the program could include defining a prehabilitation program for which patients can sign up that automatically selects the education sessions and clinical services most relevant to them.
The number of attendees varied across the 6 education sessions, with introduction to prostate cancer and sexual health being the best attended. This is consistent with the literature concerning the specific unmet supportive care and information needs in this population10,13 and with the value that men have placed on taking an active role in the decisions around their prostate cancer treatment.30 It is also possible that attendance varied because modules were introduced in a stepwise fashion and were offered on different schedules. Patients and partners both reported a high degree of satisfaction with all of the modules’ education sessions, reporting that the length, content, and delivery were appropriate.
Since 2013, a wide research portfolio has grown alongside the program. It has acted as a recruitment site for multicenter national studies and has attracted funding for several in-house research projects and evaluations. In addition, the VPC has implemented clinic-wide electronic collection of several patient-reported outcome measures using iPads. Patients have the option of contributing their data to Canadian (PC360o) and Global (TrueNTH Global Registry – Prostate Cancer Outcomes) registries for prostate cancer. The program has also created educational opportunities by supporting postdoctoral fellows. It has also provided a rich environment for urology and radiation oncology residents and fellows to participate in a multidisciplinary supportive care team, ensuring that the next generation of surgeons and oncologists recognize the importance of this approach to care.
Limitations
This is a brief descriptive study that relies on a mixture of anonymized survey and clinical chart data. Because the program’s patient feedback forms are anonymous, we are not able to link satisfaction scores to differences in sociodemographic, clinical, or prognostic factors. We also have not directly measured clinical, psychological, or quality of life outcomes; however, all 3 will be included in future studies of the program. An additional limitation is that not all program modules were offered for the entirety of the study duration and are offered at different frequencies. Thus, some modules have disproportionally higher participation rates than others. Lastly, we are missing clinical information for 16% of our participants who are not patients at the VPC.
The program is offered within an academic and teaching hospital in a major metropolitan center and depends on the work of a large interdisciplinary team. Cancer programs that are not embedded within a similar environment, such as those located in smaller rural communities, may not have access to the specialized clinical professionals who run our program, affecting its direct generalizability to these locations. Other specialists, such as palliative care teams, could be well positioned to provide support in locations that do not have a similar level of resource available. Furthermore, some program elements will be adapted to be delivered using telemedicine technology, which is an additional approach to improving access for patients who are beyond the reach of a tertiary care facility.
Conclusions
There is a growing need to provide consistent and comprehensive supportive care to patients with prostate cancer and their partners and families throughout the disease and treatment journey. The PCSC Program uses a multidisciplinary, evidenced-based, disease-focused approach to support informed treatment decision-making and address the physical, psychological, and psychosocial effects of prostate cancer diagnosis and treatment. We proactively collect data on disease, personal demographic details, and symptoms or quality of life, supporting opportunities to partner with researchers with the goal of further improving quality of life based on evidenced-based practices. Going forward, we will conduct detailed examinations of the costs and benefits (in terms of symptom management and quality of life) of the PCSC Program, further contributing to the development of evidence-based best practices for supportive care for men with prostate cancer and their families.
Acknowledgments
The authors express their gratitude to the urologists and radiation oncologists who referred their patients to the program and participated in delivering education sessions, including Dr Martin Gleave, Dr Peter Black, Dr Alan So, Dr Scott Tyldesley, and Dr Mira Keyes. They also thank Dr Richard Wassersug for his contributions to the initial program design and implementation. They thank the patients and their families for participating, and all of their current or past staff and collaborators. Lastly, they thank the funders of the program: the Specialist Services Committee, the BC Ministry of Health, the Prostate Cancer Foundation of BC, and philanthropic donors. They acknowledge Vancouver Coastal Health Research Institute and the University of British Columbia for their institutional support.
1. Cancer Research UK. Prostate cancer statistics. http://www.cancer.ca/en/cancer-information/cancer-type/prostate/statistics/?region=sk. Published 2015. Accessed June 22, 2017.
2. Siegel R, DeSantis C, Virgo K, et al. Cancer treatment and survivorship statistics, 2012. CA Cancer J Clin. 2012;62(4):220-241.
3. Canadian Cancer Society. Canadian cancer statistics special topic: predictions of the future burden of cancer in Canada. Ottawa, Canada: Public Health Agency of Canada; 2015.
4. Roth AJ, Weinberger MI, Nelson CJ. Prostate cancer: psychosocial implications and management. Future Oncol. 2008;4(4):561-568.
5. Couper J, Bloch S, Love A, Macvean M, Duchesne GM, Kissane D. Psychosocial adjustment of female partners of men with prostate cancer: a review of the literature. Psychooncology 2006;15(11):937-953.
6. Wilt TJ, MacDonald R, Rutks I, Shamliyan TA, Taylor BC, Kane RL. Systematic review: comparative effectiveness and harms of treatments for clinically localized prostate cancer. Ann Intern Med. 2008;148(6):435-448.
7. Galvão DA, Spry NA, Taaffe DR, et al. Changes in muscle, fat and bone mass after 36 weeks of maximal androgen blockade for prostate cancer. BJU Int. 2008;102(1):44-47.
8. Watts S, Leydon G, Birch B, et al. Depression and anxiety in prostate cancer: a systematic review and meta-analysis of prevalence rates. BMJ Open. 2014;4(3):e003901.
9. Zaider T, Manne S, Nelson C, Mulhall J, Kissane D. Loss of masculine identity, marital affection, and sexual bother in men with localized prostate cancer. J Sex Med. 2012;9(10):2724-2732.
10. Ream E, Quennell A, Fincham L, et al. Supportive care needs of men living with prostate cancer in England: a survey. Br J Cancer. 2008;98(12):1903-1909.
11. Howell D, Hack TF, Oliver TK, et al. Models of care for post-treatment follow-up of adult cancer survivors: a systematic review and quality appraisal of the evidence. J Cancer Surviv. 2012;6(4):359-371.
12. Halpern MT, Viswanathan M, Evans TS, Birken SA, Basch E, Mayer DK. Models of cancer survivorship care: overview and summary of current evidence. J Oncol Pract. 2015;11(1):e19-e27.
13. Smith DP, Supramaniam R, King MT, Ward J, Berry M, Armstrong BK. Age, health, and education determine supportive care needs of men younger than 70 years with prostate cancer. J Clin Oncol. 2007;25(18):2560-2566.
14. Northouse LL, Mood DW, Montie JE, et al. Living with prostate cancer: patients’ and spouses’ psychosocial status and quality of life. J Clin Oncol. 2007;25(27):4171-4177.
15. Hedden L, Wassersug R, Mahovlich S, et al. Evaluating an educational intervention to alleviate distress amongst men with newly diagnosed prostate cancer and their partners. BJU Int. 2017;120(5B):E21-E29.
16. Bradley EB, Bissonette EA, Theodorescu D. Determinants of long-term quality of life and voiding function of patients treated with radical prostatectomy or permanent brachytherapy for prostate cancer. BJU Int. 2004;94(7):1003-1009.
17. Ramsey SD, Zeliadt SB, Blough DK, et al. Impact of prostate cancer on sexual relationships: a longitudinal perspective on intimate partners’ experiences. J Sex Med. 2013;10(12):3135-3143.
18. Wittmann D, Carolan M, Given B, et al. Exploring the role of the partner in couples’ sexual recovery after surgery for prostate cancer. Support Care Cancer. 2014;22(9):2509-2515.
19. Schmitz KH, Courneya KS, Matthews C, et al. American college of sports medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42(7):1409-1426.
20. Rock CL, Doyle C, Demark-Wahnefried W, et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin. 2012;62(4):243-274.
21. Elliott S, Latini DM, Walker LM, Wassersug R, Robinson JW; ADT Suvivorship Working Group. Androgen deprivation therapy for prostate cancer: recommendations to improve patient and partner quality of life. J Sex Med. 2010;7(9):2996-3010.
22. Wassersug RJ, Walker LM, Robinson JW. Androgen deprivation therapy: an essential guide for prostate cancer patients and their loved ones. New York, NY: Demos Health; 2014.
23. Wibowo E, Walker LM, Wilyman S, et al. Androgen deprivation therapy educational program: a Canadian True NTH initiative. J Clin Oncol. 2016;34(suppl 3):243.
24. Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer. JAMA. 2000;283(3):354-360.
25. Overgård M, Angelsen A, Lydersen S, Mørkved S. Does physiotherapist-guided pelvic floor muscle training reduce urinary incontinence after radical prostatectomy? A randomised controlled trial. Eur Urol. 2008;54(2):438-448.
26. Godtman RA, Holmberg E, Khatami A, Pihl CG, Stranne J, Hugosson J. Long-term results of active surveillance in the Göteborg randomized, population-based prostate cancer screening trial. Eur Urol. 2016;70(5):760-766.
27. Cohen H, Britten N. Who decides about prostate cancer treatment? A qualitative study. Fam Pract. 2003;20(6):724-729.
28. Denberg TD, Melhado TV, Steiner JF. Patient treatment preferences in localized prostate carcinoma: the influence of emotion, misconception, and anecdote. Cancer. 2006;107(3):620-630.
29. Morris BB, Farnan L, Song L, et al. Treatment decisional regret among men with prostate cancer: racial differences and influential factors in the North Carolina health access and prostate cancer treatment project (HCaP-NC). Cancer. 2015;121(12):2029-2035.
30. Feldman-Stewart D, Capirci C, Brennenstuhl S, et al. Information for decision making by patients with early-stage prostate cancer: a comparison across 9 countries. Med Decis Making. 2011;31(5):754-766.
1. Cancer Research UK. Prostate cancer statistics. http://www.cancer.ca/en/cancer-information/cancer-type/prostate/statistics/?region=sk. Published 2015. Accessed June 22, 2017.
2. Siegel R, DeSantis C, Virgo K, et al. Cancer treatment and survivorship statistics, 2012. CA Cancer J Clin. 2012;62(4):220-241.
3. Canadian Cancer Society. Canadian cancer statistics special topic: predictions of the future burden of cancer in Canada. Ottawa, Canada: Public Health Agency of Canada; 2015.
4. Roth AJ, Weinberger MI, Nelson CJ. Prostate cancer: psychosocial implications and management. Future Oncol. 2008;4(4):561-568.
5. Couper J, Bloch S, Love A, Macvean M, Duchesne GM, Kissane D. Psychosocial adjustment of female partners of men with prostate cancer: a review of the literature. Psychooncology 2006;15(11):937-953.
6. Wilt TJ, MacDonald R, Rutks I, Shamliyan TA, Taylor BC, Kane RL. Systematic review: comparative effectiveness and harms of treatments for clinically localized prostate cancer. Ann Intern Med. 2008;148(6):435-448.
7. Galvão DA, Spry NA, Taaffe DR, et al. Changes in muscle, fat and bone mass after 36 weeks of maximal androgen blockade for prostate cancer. BJU Int. 2008;102(1):44-47.
8. Watts S, Leydon G, Birch B, et al. Depression and anxiety in prostate cancer: a systematic review and meta-analysis of prevalence rates. BMJ Open. 2014;4(3):e003901.
9. Zaider T, Manne S, Nelson C, Mulhall J, Kissane D. Loss of masculine identity, marital affection, and sexual bother in men with localized prostate cancer. J Sex Med. 2012;9(10):2724-2732.
10. Ream E, Quennell A, Fincham L, et al. Supportive care needs of men living with prostate cancer in England: a survey. Br J Cancer. 2008;98(12):1903-1909.
11. Howell D, Hack TF, Oliver TK, et al. Models of care for post-treatment follow-up of adult cancer survivors: a systematic review and quality appraisal of the evidence. J Cancer Surviv. 2012;6(4):359-371.
12. Halpern MT, Viswanathan M, Evans TS, Birken SA, Basch E, Mayer DK. Models of cancer survivorship care: overview and summary of current evidence. J Oncol Pract. 2015;11(1):e19-e27.
13. Smith DP, Supramaniam R, King MT, Ward J, Berry M, Armstrong BK. Age, health, and education determine supportive care needs of men younger than 70 years with prostate cancer. J Clin Oncol. 2007;25(18):2560-2566.
14. Northouse LL, Mood DW, Montie JE, et al. Living with prostate cancer: patients’ and spouses’ psychosocial status and quality of life. J Clin Oncol. 2007;25(27):4171-4177.
15. Hedden L, Wassersug R, Mahovlich S, et al. Evaluating an educational intervention to alleviate distress amongst men with newly diagnosed prostate cancer and their partners. BJU Int. 2017;120(5B):E21-E29.
16. Bradley EB, Bissonette EA, Theodorescu D. Determinants of long-term quality of life and voiding function of patients treated with radical prostatectomy or permanent brachytherapy for prostate cancer. BJU Int. 2004;94(7):1003-1009.
17. Ramsey SD, Zeliadt SB, Blough DK, et al. Impact of prostate cancer on sexual relationships: a longitudinal perspective on intimate partners’ experiences. J Sex Med. 2013;10(12):3135-3143.
18. Wittmann D, Carolan M, Given B, et al. Exploring the role of the partner in couples’ sexual recovery after surgery for prostate cancer. Support Care Cancer. 2014;22(9):2509-2515.
19. Schmitz KH, Courneya KS, Matthews C, et al. American college of sports medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42(7):1409-1426.
20. Rock CL, Doyle C, Demark-Wahnefried W, et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin. 2012;62(4):243-274.
21. Elliott S, Latini DM, Walker LM, Wassersug R, Robinson JW; ADT Suvivorship Working Group. Androgen deprivation therapy for prostate cancer: recommendations to improve patient and partner quality of life. J Sex Med. 2010;7(9):2996-3010.
22. Wassersug RJ, Walker LM, Robinson JW. Androgen deprivation therapy: an essential guide for prostate cancer patients and their loved ones. New York, NY: Demos Health; 2014.
23. Wibowo E, Walker LM, Wilyman S, et al. Androgen deprivation therapy educational program: a Canadian True NTH initiative. J Clin Oncol. 2016;34(suppl 3):243.
24. Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer. JAMA. 2000;283(3):354-360.
25. Overgård M, Angelsen A, Lydersen S, Mørkved S. Does physiotherapist-guided pelvic floor muscle training reduce urinary incontinence after radical prostatectomy? A randomised controlled trial. Eur Urol. 2008;54(2):438-448.
26. Godtman RA, Holmberg E, Khatami A, Pihl CG, Stranne J, Hugosson J. Long-term results of active surveillance in the Göteborg randomized, population-based prostate cancer screening trial. Eur Urol. 2016;70(5):760-766.
27. Cohen H, Britten N. Who decides about prostate cancer treatment? A qualitative study. Fam Pract. 2003;20(6):724-729.
28. Denberg TD, Melhado TV, Steiner JF. Patient treatment preferences in localized prostate carcinoma: the influence of emotion, misconception, and anecdote. Cancer. 2006;107(3):620-630.
29. Morris BB, Farnan L, Song L, et al. Treatment decisional regret among men with prostate cancer: racial differences and influential factors in the North Carolina health access and prostate cancer treatment project (HCaP-NC). Cancer. 2015;121(12):2029-2035.
30. Feldman-Stewart D, Capirci C, Brennenstuhl S, et al. Information for decision making by patients with early-stage prostate cancer: a comparison across 9 countries. Med Decis Making. 2011;31(5):754-766.
hTERT expression predicts RCC survival, tumor aggressiveness
Human telomerase reverse transcriptase (hTERT) protein expression is associated with clear cell renal carcinoma (ccRCC) tumor aggressiveness and disease-specific survival (DSS), according to investigators.
Associations between hTERT expression and clinicopathologic features and outcomes were less robust or nonexistent in papillary and chromophobe subtypes, reported Leili Saeednejad Zanjani, MD, of the Oncopathology Research Center at Iran University of Medical Sciences in Tehran, and colleagues.
“Evidence shows that telomerase is expressed in 85% of malignancies, and the level of its activity is higher in advanced and metastatic tumors,” the authors wrote in Pathology.
“A number of clinical studies have been performed to evaluate the association between telomerase activity and clinicopathological parameters in renal cancer showing that telomerase activity level correlates with progression of RCC,” Dr. Zanjani and associates wrote. As none of these specifically evaluated hTERT protein expression, the investigators conducted a study to learn more.
The investigators analyzed hTERT expression level in 176 cases of RCC, requiring that each tumor had three core biopsies because of concerns of heterogeneity. The population consisted of 113 clear cell, 12 type I papillary, 20 type II papillary, and 31 chromophobe subtypes. Patient and clinicopathologic features were compared with survival and hTERT expression. Median follow-up time was 42 months.
Correlations between hTERT expression and disease characteristics were pronounced in cases of ccRCC, compared with other subtypes. In ccRCC, hTERT expression was significantly associated with tumor stage, nucleolar grade, tumor size, microvascular invasion, lymph node invasion, renal pelvis involvement, renal sinus fat involvement, Gerota fascia invasion, and distant metastasis. Survival analysis showed that DSS of ccRCC patients with high hTERT expression was 58 months, compared with 68 months for those with low hTERT expression (P =.012). Other parameters associated with survival were nucleolar grade, tumor stage, and tumor size.
For type I and II papillary subtypes, associations were found between hTERT expression and tumor stage and distant metastasis. In contrast, chromophobe RCC revealed no such relationships. No associations were found between hTERT expression and survival in any of these three latter subtypes, for slightly different reasons; no patients with type I disease died of renal cancer, disallowing creation of a Kaplan-Meier survival curve, whereas type II and chromophobe survival curves revealed insignificant relationships with hTERT expression. Along the same lines, no clinicopathologic characteristics of these subtypes were tied with survival.
“From these findings we are able to conclude that hTERT protein expression may be a novel prognostic indicator of worse outcome in tumor biopsies of patients with ccRCC, if follow up time is more prolonged,” the authors wrote. They noted that “telomerase is an attractive and ideal target for therapy due to overexpression in the majority of malignancies and low or nonexpression in most somatic cells.”
The study was funded by the Iran National Science Foundation. The authors declared no conflicts of interest.
SOURCE: Zanjani LS et al. Pathology. 2018 Nov 19. doi: 10.1016/j.pathol.2018.08.019.
Human telomerase reverse transcriptase (hTERT) protein expression is associated with clear cell renal carcinoma (ccRCC) tumor aggressiveness and disease-specific survival (DSS), according to investigators.
Associations between hTERT expression and clinicopathologic features and outcomes were less robust or nonexistent in papillary and chromophobe subtypes, reported Leili Saeednejad Zanjani, MD, of the Oncopathology Research Center at Iran University of Medical Sciences in Tehran, and colleagues.
“Evidence shows that telomerase is expressed in 85% of malignancies, and the level of its activity is higher in advanced and metastatic tumors,” the authors wrote in Pathology.
“A number of clinical studies have been performed to evaluate the association between telomerase activity and clinicopathological parameters in renal cancer showing that telomerase activity level correlates with progression of RCC,” Dr. Zanjani and associates wrote. As none of these specifically evaluated hTERT protein expression, the investigators conducted a study to learn more.
The investigators analyzed hTERT expression level in 176 cases of RCC, requiring that each tumor had three core biopsies because of concerns of heterogeneity. The population consisted of 113 clear cell, 12 type I papillary, 20 type II papillary, and 31 chromophobe subtypes. Patient and clinicopathologic features were compared with survival and hTERT expression. Median follow-up time was 42 months.
Correlations between hTERT expression and disease characteristics were pronounced in cases of ccRCC, compared with other subtypes. In ccRCC, hTERT expression was significantly associated with tumor stage, nucleolar grade, tumor size, microvascular invasion, lymph node invasion, renal pelvis involvement, renal sinus fat involvement, Gerota fascia invasion, and distant metastasis. Survival analysis showed that DSS of ccRCC patients with high hTERT expression was 58 months, compared with 68 months for those with low hTERT expression (P =.012). Other parameters associated with survival were nucleolar grade, tumor stage, and tumor size.
For type I and II papillary subtypes, associations were found between hTERT expression and tumor stage and distant metastasis. In contrast, chromophobe RCC revealed no such relationships. No associations were found between hTERT expression and survival in any of these three latter subtypes, for slightly different reasons; no patients with type I disease died of renal cancer, disallowing creation of a Kaplan-Meier survival curve, whereas type II and chromophobe survival curves revealed insignificant relationships with hTERT expression. Along the same lines, no clinicopathologic characteristics of these subtypes were tied with survival.
“From these findings we are able to conclude that hTERT protein expression may be a novel prognostic indicator of worse outcome in tumor biopsies of patients with ccRCC, if follow up time is more prolonged,” the authors wrote. They noted that “telomerase is an attractive and ideal target for therapy due to overexpression in the majority of malignancies and low or nonexpression in most somatic cells.”
The study was funded by the Iran National Science Foundation. The authors declared no conflicts of interest.
SOURCE: Zanjani LS et al. Pathology. 2018 Nov 19. doi: 10.1016/j.pathol.2018.08.019.
Human telomerase reverse transcriptase (hTERT) protein expression is associated with clear cell renal carcinoma (ccRCC) tumor aggressiveness and disease-specific survival (DSS), according to investigators.
Associations between hTERT expression and clinicopathologic features and outcomes were less robust or nonexistent in papillary and chromophobe subtypes, reported Leili Saeednejad Zanjani, MD, of the Oncopathology Research Center at Iran University of Medical Sciences in Tehran, and colleagues.
“Evidence shows that telomerase is expressed in 85% of malignancies, and the level of its activity is higher in advanced and metastatic tumors,” the authors wrote in Pathology.
“A number of clinical studies have been performed to evaluate the association between telomerase activity and clinicopathological parameters in renal cancer showing that telomerase activity level correlates with progression of RCC,” Dr. Zanjani and associates wrote. As none of these specifically evaluated hTERT protein expression, the investigators conducted a study to learn more.
The investigators analyzed hTERT expression level in 176 cases of RCC, requiring that each tumor had three core biopsies because of concerns of heterogeneity. The population consisted of 113 clear cell, 12 type I papillary, 20 type II papillary, and 31 chromophobe subtypes. Patient and clinicopathologic features were compared with survival and hTERT expression. Median follow-up time was 42 months.
Correlations between hTERT expression and disease characteristics were pronounced in cases of ccRCC, compared with other subtypes. In ccRCC, hTERT expression was significantly associated with tumor stage, nucleolar grade, tumor size, microvascular invasion, lymph node invasion, renal pelvis involvement, renal sinus fat involvement, Gerota fascia invasion, and distant metastasis. Survival analysis showed that DSS of ccRCC patients with high hTERT expression was 58 months, compared with 68 months for those with low hTERT expression (P =.012). Other parameters associated with survival were nucleolar grade, tumor stage, and tumor size.
For type I and II papillary subtypes, associations were found between hTERT expression and tumor stage and distant metastasis. In contrast, chromophobe RCC revealed no such relationships. No associations were found between hTERT expression and survival in any of these three latter subtypes, for slightly different reasons; no patients with type I disease died of renal cancer, disallowing creation of a Kaplan-Meier survival curve, whereas type II and chromophobe survival curves revealed insignificant relationships with hTERT expression. Along the same lines, no clinicopathologic characteristics of these subtypes were tied with survival.
“From these findings we are able to conclude that hTERT protein expression may be a novel prognostic indicator of worse outcome in tumor biopsies of patients with ccRCC, if follow up time is more prolonged,” the authors wrote. They noted that “telomerase is an attractive and ideal target for therapy due to overexpression in the majority of malignancies and low or nonexpression in most somatic cells.”
The study was funded by the Iran National Science Foundation. The authors declared no conflicts of interest.
SOURCE: Zanjani LS et al. Pathology. 2018 Nov 19. doi: 10.1016/j.pathol.2018.08.019.
FROM PATHOLOGY
Key clinical point: Human telomerase reverse transcriptase (hTERT) protein expression is associated with clear cell renal carcinoma (ccRCC) tumor aggressiveness and disease-specific survival (DSS).
Major finding: DSS of ccRCC patients with high hTERT expression was 58 months, compared with 68 months for those with low hTERT expression (P equal to .012).
Study details: An analysis of hTERT protein expression and disease characteristics in 176 patients with RCC. The subtype population consisted of 113 clear cell, 12 type I papillary, 20 type II papillary, and 31 chromophobe cases.
Disclosures: The study was funded by the Iran National Science Foundation. The authors declared no conflicts of interest.
Source: Zanjani LS et al. Pathology. 2018 Nov 19. doi: 10.1016/j.pathol.2018.08.019.
Age-related gene expression may affect responses to RCC therapy
Older patients with ccRCC may respond better than younger patients to phosphoinositide 3-kinase (PI3K) or checkpoint inhibition because of age-related changes in gene expression, according to investigators.
This possibility was raised by in silico results from a broader study of gene expression patterns in clear cell renal carcinoma (ccRCC) and normal kidney tissues, reported lead author, Lara Feulner, MD, of the department of human genetics at McGill University and Genome Quebec Innovation Centre in Montreal.
“Several factors could contribute to the interindividual diversity among cancer patients,” the investigators wrote in a report published in Urologic Oncology.
“Their disease course could be affected not only by cell-intrinsic factors, but also by age-related changes impacting the vasculature, immune system and stroma. Little is known in this regard about ccRCC, a disease which affects adults across a wide age spectrum. Whether and how aging and comorbidities such as atherosclerosis may affect the biology and therapy of ccRCC has scarcely been considered,” they wrote.
The investigators explored this territory by analyzing datasets from The Cancer Genome Atlas (TCGA) and the Cancer Genomics of the Kidney (CAGEKID) program of the International Cancer Genome Consortium. Using regression, pathway enrichment, and connectivity mapping analyses, they were able to determine associations between age and gene expression, cellular processes, and drug treatment responses, respectively.
The investigators reported that age-related gene expression patterns occurred commonly in both normal and tumor tissues. Associations were reproducible between TCGA and CAGEKID datasets for both classes of tissue (tumor samples, R equal to 0.416, P less than 2.2 x 10-16; normal samples, R equal to 0.403, P less than 2.2 x 10-16). Out of the top 1,000 age-associated genes in tumor samples from each dataset, 383 were commonly downregulated with age and 294 were commonly upregulated with age in both datasets (P less than 2.2 x 10-16).
Among cellular pathways, the investigators found opposite age-relationship patterns. For example, normal tissues upregulated extracellular matrix and cell adhesion pathways with age, whereas tumor tissues downregulated the same pathways. Similar patterns of opposition were found in metabolism and oxidation pathways. Other age-related patterns were noted in some immune pathways, such as upregulation of toll-like receptor and tumor necrosis factor 2 noncanonical NF-kappa-B signaling in tumors, which became more common with age. A closer look showed that upregulation of tumor necrosis factor signaling was more common in female patients, who also downregulated Notch pathways more often than men.
Analysis of treatment responses showed possible relationships with age-dependent gene expression and immunotherapy. Specifically, of 532 genes tied to programmed cell death protein 1 (PD-1) resistance, 69 were among the 383 genes downregulated in older patients with ccRCC (P less than 2.2 x 10-16; 4.05 fold-enrichment), suggesting that older patients may respond better to anti-PD-1 therapy than younger patients. Similarly, connectivity map analysis showed that age-dependent gene expression may improve candidacy of older ccRCC patients for PI3K inhibition.
“We now have evidence that there are notable differences in tumor-associated pathway regulation between younger and older ccRCC patients, which may be therapeutically actionable,” the authors concluded.
The study was funded by the Cancer Research Society operation grant, a Canadian Cancer Society Research Institute Innovation-to-Impact grant, and a Canadian Institutes of Health Research Foundation grant. The authors reported no conflicts of interest.
SOURCE: Feulner et al. Urol Onc. 2018 Nov 23. doi: 10.1016/j.urolonc.2018.11.006.
Older patients with ccRCC may respond better than younger patients to phosphoinositide 3-kinase (PI3K) or checkpoint inhibition because of age-related changes in gene expression, according to investigators.
This possibility was raised by in silico results from a broader study of gene expression patterns in clear cell renal carcinoma (ccRCC) and normal kidney tissues, reported lead author, Lara Feulner, MD, of the department of human genetics at McGill University and Genome Quebec Innovation Centre in Montreal.
“Several factors could contribute to the interindividual diversity among cancer patients,” the investigators wrote in a report published in Urologic Oncology.
“Their disease course could be affected not only by cell-intrinsic factors, but also by age-related changes impacting the vasculature, immune system and stroma. Little is known in this regard about ccRCC, a disease which affects adults across a wide age spectrum. Whether and how aging and comorbidities such as atherosclerosis may affect the biology and therapy of ccRCC has scarcely been considered,” they wrote.
The investigators explored this territory by analyzing datasets from The Cancer Genome Atlas (TCGA) and the Cancer Genomics of the Kidney (CAGEKID) program of the International Cancer Genome Consortium. Using regression, pathway enrichment, and connectivity mapping analyses, they were able to determine associations between age and gene expression, cellular processes, and drug treatment responses, respectively.
The investigators reported that age-related gene expression patterns occurred commonly in both normal and tumor tissues. Associations were reproducible between TCGA and CAGEKID datasets for both classes of tissue (tumor samples, R equal to 0.416, P less than 2.2 x 10-16; normal samples, R equal to 0.403, P less than 2.2 x 10-16). Out of the top 1,000 age-associated genes in tumor samples from each dataset, 383 were commonly downregulated with age and 294 were commonly upregulated with age in both datasets (P less than 2.2 x 10-16).
Among cellular pathways, the investigators found opposite age-relationship patterns. For example, normal tissues upregulated extracellular matrix and cell adhesion pathways with age, whereas tumor tissues downregulated the same pathways. Similar patterns of opposition were found in metabolism and oxidation pathways. Other age-related patterns were noted in some immune pathways, such as upregulation of toll-like receptor and tumor necrosis factor 2 noncanonical NF-kappa-B signaling in tumors, which became more common with age. A closer look showed that upregulation of tumor necrosis factor signaling was more common in female patients, who also downregulated Notch pathways more often than men.
Analysis of treatment responses showed possible relationships with age-dependent gene expression and immunotherapy. Specifically, of 532 genes tied to programmed cell death protein 1 (PD-1) resistance, 69 were among the 383 genes downregulated in older patients with ccRCC (P less than 2.2 x 10-16; 4.05 fold-enrichment), suggesting that older patients may respond better to anti-PD-1 therapy than younger patients. Similarly, connectivity map analysis showed that age-dependent gene expression may improve candidacy of older ccRCC patients for PI3K inhibition.
“We now have evidence that there are notable differences in tumor-associated pathway regulation between younger and older ccRCC patients, which may be therapeutically actionable,” the authors concluded.
The study was funded by the Cancer Research Society operation grant, a Canadian Cancer Society Research Institute Innovation-to-Impact grant, and a Canadian Institutes of Health Research Foundation grant. The authors reported no conflicts of interest.
SOURCE: Feulner et al. Urol Onc. 2018 Nov 23. doi: 10.1016/j.urolonc.2018.11.006.
Older patients with ccRCC may respond better than younger patients to phosphoinositide 3-kinase (PI3K) or checkpoint inhibition because of age-related changes in gene expression, according to investigators.
This possibility was raised by in silico results from a broader study of gene expression patterns in clear cell renal carcinoma (ccRCC) and normal kidney tissues, reported lead author, Lara Feulner, MD, of the department of human genetics at McGill University and Genome Quebec Innovation Centre in Montreal.
“Several factors could contribute to the interindividual diversity among cancer patients,” the investigators wrote in a report published in Urologic Oncology.
“Their disease course could be affected not only by cell-intrinsic factors, but also by age-related changes impacting the vasculature, immune system and stroma. Little is known in this regard about ccRCC, a disease which affects adults across a wide age spectrum. Whether and how aging and comorbidities such as atherosclerosis may affect the biology and therapy of ccRCC has scarcely been considered,” they wrote.
The investigators explored this territory by analyzing datasets from The Cancer Genome Atlas (TCGA) and the Cancer Genomics of the Kidney (CAGEKID) program of the International Cancer Genome Consortium. Using regression, pathway enrichment, and connectivity mapping analyses, they were able to determine associations between age and gene expression, cellular processes, and drug treatment responses, respectively.
The investigators reported that age-related gene expression patterns occurred commonly in both normal and tumor tissues. Associations were reproducible between TCGA and CAGEKID datasets for both classes of tissue (tumor samples, R equal to 0.416, P less than 2.2 x 10-16; normal samples, R equal to 0.403, P less than 2.2 x 10-16). Out of the top 1,000 age-associated genes in tumor samples from each dataset, 383 were commonly downregulated with age and 294 were commonly upregulated with age in both datasets (P less than 2.2 x 10-16).
Among cellular pathways, the investigators found opposite age-relationship patterns. For example, normal tissues upregulated extracellular matrix and cell adhesion pathways with age, whereas tumor tissues downregulated the same pathways. Similar patterns of opposition were found in metabolism and oxidation pathways. Other age-related patterns were noted in some immune pathways, such as upregulation of toll-like receptor and tumor necrosis factor 2 noncanonical NF-kappa-B signaling in tumors, which became more common with age. A closer look showed that upregulation of tumor necrosis factor signaling was more common in female patients, who also downregulated Notch pathways more often than men.
Analysis of treatment responses showed possible relationships with age-dependent gene expression and immunotherapy. Specifically, of 532 genes tied to programmed cell death protein 1 (PD-1) resistance, 69 were among the 383 genes downregulated in older patients with ccRCC (P less than 2.2 x 10-16; 4.05 fold-enrichment), suggesting that older patients may respond better to anti-PD-1 therapy than younger patients. Similarly, connectivity map analysis showed that age-dependent gene expression may improve candidacy of older ccRCC patients for PI3K inhibition.
“We now have evidence that there are notable differences in tumor-associated pathway regulation between younger and older ccRCC patients, which may be therapeutically actionable,” the authors concluded.
The study was funded by the Cancer Research Society operation grant, a Canadian Cancer Society Research Institute Innovation-to-Impact grant, and a Canadian Institutes of Health Research Foundation grant. The authors reported no conflicts of interest.
SOURCE: Feulner et al. Urol Onc. 2018 Nov 23. doi: 10.1016/j.urolonc.2018.11.006.
FROM UROLOGIC ONCOLOGY
Key clinical point: Older patients with ccRCC may respond better than younger patients to phosphoinositide 3-kinase or checkpoint inhibition due to age-related changes in gene expression.
Major finding: Out of the top 1,000 age-associated genes in tumor samples, 383 were commonly downregulated with age and 294 were commonly upregulated with age in two large data sets (P less than 2.2 x 10-16).
Study details: An analysis of data from The Cancer Genome Atlas (TCGA) and the Cancer Genomics of the Kidney (CAGEKID) program of the International Cancer Genome Consortium.
Disclosures: The study was funded by the Cancer Research Society operation grant, a Canadian Cancer Society Research Institute Innovation-to-Impact grant, and a Canadian Institutes of Health Research Foundation grant.
Source: Feulner et al. Urol Oncol. 2018 Nov 23. doi: 10.1016/j.urolonc.2018.11.006.
Clear cell RCC with papillary features is still ccRCC
Clear cell renal cell carcinoma (ccRCC) with predominant papillary features is best classified as a rare morphologic variant of ccRCC, not MiT family translocation or papillary RCC, according to an analysis of 23 tumors.
Areas of papillary pattern made up anywhere from 40% to 100% of the 23 tumors. Even so, cytology and immunohistochemical and genetic testing was most consistent with ccRCC, and tumors were negative for TFE3 protein, which ruled out MiT family translocation RCC, reported Reza Alaghehbandan, MD, and his associates. The report is in Annals of Diagnostic Pathology.
The findings help clarify where ccRCC fits in the diagnostic tree when there’s significant papillary morphology, something that hasn’t been clear until now. The proper classification matters because it carries treatment implications; for instance, ccRCC generally responds well to immunotherapy and targeted therapy, but papillary RCC does not, so doctors often recommend treatment through a clinical trial.
“The presence of mixed morphologic components in renal neoplasms in general and in ccRCCs in particular can be puzzling in routine practice, which can potentially lead to misdiagnosis.” Getting it right is “crucial” to ensure the best treatment, said Dr. Alaghehbandan, of the University of British Columbia, Vancouver, and his associates.
Cytokeratin 7 (CK7) staining was negative in the nonpapillary areas of 20 tumors (87%), and only focally positive in three (13%). In contrast, clear cell papillary RCC is strongly and diffusely positive for CK7.
In papillary areas, Alpha-methyl CoA racemase was positive or focally positive in 17 tumors (73.9%); in nonpapillary areas, it was positive or focally positive in 22 (95.6%). Carbonic anhydrase IX was mainly negative in both nonpapillary and papillary areas, while vimentin and CD10 were positive or focally positive in both.
Patients were a mean of 65.2 years old, and 19 were men. The median tumor size was 6.5 cm. At a median follow-up of 2.5 years, two patients had died of their disease, and two had developed metastasis.
There was no industry funding, and the investigators didn’t have any disclosures.
SOURCE: Alaghehbandan R et al. Ann Diagn Pathol. 2018 Nov 22. doi: 10.1016/j.anndiagpath.2018.11.004.
Clear cell renal cell carcinoma (ccRCC) with predominant papillary features is best classified as a rare morphologic variant of ccRCC, not MiT family translocation or papillary RCC, according to an analysis of 23 tumors.
Areas of papillary pattern made up anywhere from 40% to 100% of the 23 tumors. Even so, cytology and immunohistochemical and genetic testing was most consistent with ccRCC, and tumors were negative for TFE3 protein, which ruled out MiT family translocation RCC, reported Reza Alaghehbandan, MD, and his associates. The report is in Annals of Diagnostic Pathology.
The findings help clarify where ccRCC fits in the diagnostic tree when there’s significant papillary morphology, something that hasn’t been clear until now. The proper classification matters because it carries treatment implications; for instance, ccRCC generally responds well to immunotherapy and targeted therapy, but papillary RCC does not, so doctors often recommend treatment through a clinical trial.
“The presence of mixed morphologic components in renal neoplasms in general and in ccRCCs in particular can be puzzling in routine practice, which can potentially lead to misdiagnosis.” Getting it right is “crucial” to ensure the best treatment, said Dr. Alaghehbandan, of the University of British Columbia, Vancouver, and his associates.
Cytokeratin 7 (CK7) staining was negative in the nonpapillary areas of 20 tumors (87%), and only focally positive in three (13%). In contrast, clear cell papillary RCC is strongly and diffusely positive for CK7.
In papillary areas, Alpha-methyl CoA racemase was positive or focally positive in 17 tumors (73.9%); in nonpapillary areas, it was positive or focally positive in 22 (95.6%). Carbonic anhydrase IX was mainly negative in both nonpapillary and papillary areas, while vimentin and CD10 were positive or focally positive in both.
Patients were a mean of 65.2 years old, and 19 were men. The median tumor size was 6.5 cm. At a median follow-up of 2.5 years, two patients had died of their disease, and two had developed metastasis.
There was no industry funding, and the investigators didn’t have any disclosures.
SOURCE: Alaghehbandan R et al. Ann Diagn Pathol. 2018 Nov 22. doi: 10.1016/j.anndiagpath.2018.11.004.
Clear cell renal cell carcinoma (ccRCC) with predominant papillary features is best classified as a rare morphologic variant of ccRCC, not MiT family translocation or papillary RCC, according to an analysis of 23 tumors.
Areas of papillary pattern made up anywhere from 40% to 100% of the 23 tumors. Even so, cytology and immunohistochemical and genetic testing was most consistent with ccRCC, and tumors were negative for TFE3 protein, which ruled out MiT family translocation RCC, reported Reza Alaghehbandan, MD, and his associates. The report is in Annals of Diagnostic Pathology.
The findings help clarify where ccRCC fits in the diagnostic tree when there’s significant papillary morphology, something that hasn’t been clear until now. The proper classification matters because it carries treatment implications; for instance, ccRCC generally responds well to immunotherapy and targeted therapy, but papillary RCC does not, so doctors often recommend treatment through a clinical trial.
“The presence of mixed morphologic components in renal neoplasms in general and in ccRCCs in particular can be puzzling in routine practice, which can potentially lead to misdiagnosis.” Getting it right is “crucial” to ensure the best treatment, said Dr. Alaghehbandan, of the University of British Columbia, Vancouver, and his associates.
Cytokeratin 7 (CK7) staining was negative in the nonpapillary areas of 20 tumors (87%), and only focally positive in three (13%). In contrast, clear cell papillary RCC is strongly and diffusely positive for CK7.
In papillary areas, Alpha-methyl CoA racemase was positive or focally positive in 17 tumors (73.9%); in nonpapillary areas, it was positive or focally positive in 22 (95.6%). Carbonic anhydrase IX was mainly negative in both nonpapillary and papillary areas, while vimentin and CD10 were positive or focally positive in both.
Patients were a mean of 65.2 years old, and 19 were men. The median tumor size was 6.5 cm. At a median follow-up of 2.5 years, two patients had died of their disease, and two had developed metastasis.
There was no industry funding, and the investigators didn’t have any disclosures.
SOURCE: Alaghehbandan R et al. Ann Diagn Pathol. 2018 Nov 22. doi: 10.1016/j.anndiagpath.2018.11.004.
FROM ANNALS OF DIAGNOSTIC PATHOLOGY
Key clinical point: Clear cell renal cell carcinoma (ccRCC) with predominant papillary features is best classified as a rare morphologic variant of ccRCC, not MiT family translocation or papillary RCC. The distinction has treatment implications.
Major finding: Areas of papillary pattern made up anywhere from 40% to 100% of the 23 tumors. Even so, cytology and immunohistochemical and genetic testing was most consistent with ccRCC, and tumors were negative for TFE3 protein, which ruled out MiT family translocation RCC.
Study details: Analysis of 23 tumors.
Disclosures: There was no industry funding, and the investigators didn’t have any disclosures.
Source: Alaghehbandan R et al. Ann Diagn Pathol. 2018 Nov 22. doi: 10.1016/j.anndiagpath.2018.11.004.