User login
Trial matches pediatric cancer patients to targeted therapies
Researchers have found they can screen pediatric cancer patients for genetic alterations and match those patients to appropriate targeted therapies.
Thus far, 24% of the patients screened have been matched and assigned to a treatment, and 10% have been enrolled on treatment protocols.
The patients were screened and matched as part of the National Cancer Institute–Children’s Oncology Group Pediatric MATCH (Molecular Analysis for Therapy Choice) trial.
Results from this trial are scheduled to be presented at the annual meeting of the American Society of Clinical Oncology.
Donald Williams Parsons, MD, PhD, of Baylor College of Medicine in Houston, Tex., presented some results at a press briefing in advance of the meeting. “[T]he last 10 years have been an incredible time in terms of learning more about the genetics and underlying molecular basis of both adult and pediatric cancers,” Dr. Parsons said.
He pointed out, however, that it is not yet known if this information will be useful in guiding the treatment of pediatric cancers. Specifically, how many pediatric patients can be matched to targeted therapies, and how effective will those therapies be?
The Pediatric MATCH trial (NCT03155620) was developed to answer these questions. Researchers plan to enroll at least 1,000 patients in this trial. Patients are eligible if they are 1-21 years of age and have refractory or recurrent solid tumors, non-Hodgkin lymphomas, or histiocytic disorders.
After patients are enrolled in the trial, their tumor samples undergo DNA and RNA sequencing, and the results are used to match each patient to a targeted therapy. At present, the trial can match patients to one of 10 drugs:
- larotrectinib (targeting NTRK fusions).
- erdafitinib (targeting FGFR1/2/3/4).
- tazemetostat (targeting EZH2 or members of the SWI/SNF complex).
- LY3023414 (targeting the PI3K/MTOR pathway).
- selumetinib (targeting the MAPK pathway).
- ensartinib (targeting ALK or ROS1).
- vemurafenib (targeting BRAF V600 mutations).
- olaparib (targeting defects in DNA damage repair).
- palbociclib (targeting alterations in cell cycle genes).
- ulixertinib (targeting MAPK pathway mutations).
Early results
From July 2017 through December 2018, 422 patients were enrolled in the trial. The patients had more than 60 different diagnoses, including brain tumors, sarcomas, neuroblastoma, renal and liver cancers, and other malignancies.
The researchers received tumor samples from 390 patients, attempted sequencing of 370 samples (95%), and completed sequencing of 357 samples (92%).
A treatment target was found in 112 (29%) patients, 95 (24%) of those patients were assigned to a treatment, and 39 (10%) were enrolled in a protocol. The median turnaround time from sample receipt to treatment assignment was 15 days.
“In addition to the sequencing being successful, the patients are being matched to the different treatments,” Dr. Parsons said. He added that the study is ongoing, so more of the matched and assigned patients will be enrolled in protocols in the future.
Dr. Parsons also presented results by tumor type. A targetable alteration was identified in 26% (67/255) of all non–central nervous system solid tumors, 13% (10/75) of osteosarcomas, 50% (18/36) of rhabdomyosarcomas, 21% (7/33) of Ewing sarcomas, 25% (9/36) of other sarcomas, 19% (5/26) of renal cancers, 16% (3/19) of carcinomas, 44% (8/18) of neuroblastomas, 43% (3/7) of liver cancers, and 29% (4/14) of “other” tumors.
Drilling down further, Dr. Parsons presented details on specific alterations in one cancer type: astrocytomas. Targetable alterations were found in 74% (29/39) of astrocytomas. This includes NF1 mutations (18%), BRAF V600E (15%), FGFR1 fusions/mutations (10%), BRAF fusions (10%), PIK3CA mutations (8%), NRAS/KRAS mutations (5%), and other alterations.
“Pretty remarkably, in this one diagnosis, there are patients who have been matched to nine of the ten different treatment arms,” Dr. Parsons said. “This study is allowing us to evaluate targeted therapies – specific types of investigational drugs – in patients with many different cancer types, some common, some very rare. So, hopefully, we can study these agents and identify signals of activity where some of these drugs may work for our patients.”
The Pediatric MATCH trial is sponsored by the National Cancer Institute. Dr. Parsons has patents, royalties, and other intellectual property related to genes discovered through sequencing of several adult cancer types.
SOURCE: Parsons DW et al. ASCO 2019, Abstract 10011.
Researchers have found they can screen pediatric cancer patients for genetic alterations and match those patients to appropriate targeted therapies.
Thus far, 24% of the patients screened have been matched and assigned to a treatment, and 10% have been enrolled on treatment protocols.
The patients were screened and matched as part of the National Cancer Institute–Children’s Oncology Group Pediatric MATCH (Molecular Analysis for Therapy Choice) trial.
Results from this trial are scheduled to be presented at the annual meeting of the American Society of Clinical Oncology.
Donald Williams Parsons, MD, PhD, of Baylor College of Medicine in Houston, Tex., presented some results at a press briefing in advance of the meeting. “[T]he last 10 years have been an incredible time in terms of learning more about the genetics and underlying molecular basis of both adult and pediatric cancers,” Dr. Parsons said.
He pointed out, however, that it is not yet known if this information will be useful in guiding the treatment of pediatric cancers. Specifically, how many pediatric patients can be matched to targeted therapies, and how effective will those therapies be?
The Pediatric MATCH trial (NCT03155620) was developed to answer these questions. Researchers plan to enroll at least 1,000 patients in this trial. Patients are eligible if they are 1-21 years of age and have refractory or recurrent solid tumors, non-Hodgkin lymphomas, or histiocytic disorders.
After patients are enrolled in the trial, their tumor samples undergo DNA and RNA sequencing, and the results are used to match each patient to a targeted therapy. At present, the trial can match patients to one of 10 drugs:
- larotrectinib (targeting NTRK fusions).
- erdafitinib (targeting FGFR1/2/3/4).
- tazemetostat (targeting EZH2 or members of the SWI/SNF complex).
- LY3023414 (targeting the PI3K/MTOR pathway).
- selumetinib (targeting the MAPK pathway).
- ensartinib (targeting ALK or ROS1).
- vemurafenib (targeting BRAF V600 mutations).
- olaparib (targeting defects in DNA damage repair).
- palbociclib (targeting alterations in cell cycle genes).
- ulixertinib (targeting MAPK pathway mutations).
Early results
From July 2017 through December 2018, 422 patients were enrolled in the trial. The patients had more than 60 different diagnoses, including brain tumors, sarcomas, neuroblastoma, renal and liver cancers, and other malignancies.
The researchers received tumor samples from 390 patients, attempted sequencing of 370 samples (95%), and completed sequencing of 357 samples (92%).
A treatment target was found in 112 (29%) patients, 95 (24%) of those patients were assigned to a treatment, and 39 (10%) were enrolled in a protocol. The median turnaround time from sample receipt to treatment assignment was 15 days.
“In addition to the sequencing being successful, the patients are being matched to the different treatments,” Dr. Parsons said. He added that the study is ongoing, so more of the matched and assigned patients will be enrolled in protocols in the future.
Dr. Parsons also presented results by tumor type. A targetable alteration was identified in 26% (67/255) of all non–central nervous system solid tumors, 13% (10/75) of osteosarcomas, 50% (18/36) of rhabdomyosarcomas, 21% (7/33) of Ewing sarcomas, 25% (9/36) of other sarcomas, 19% (5/26) of renal cancers, 16% (3/19) of carcinomas, 44% (8/18) of neuroblastomas, 43% (3/7) of liver cancers, and 29% (4/14) of “other” tumors.
Drilling down further, Dr. Parsons presented details on specific alterations in one cancer type: astrocytomas. Targetable alterations were found in 74% (29/39) of astrocytomas. This includes NF1 mutations (18%), BRAF V600E (15%), FGFR1 fusions/mutations (10%), BRAF fusions (10%), PIK3CA mutations (8%), NRAS/KRAS mutations (5%), and other alterations.
“Pretty remarkably, in this one diagnosis, there are patients who have been matched to nine of the ten different treatment arms,” Dr. Parsons said. “This study is allowing us to evaluate targeted therapies – specific types of investigational drugs – in patients with many different cancer types, some common, some very rare. So, hopefully, we can study these agents and identify signals of activity where some of these drugs may work for our patients.”
The Pediatric MATCH trial is sponsored by the National Cancer Institute. Dr. Parsons has patents, royalties, and other intellectual property related to genes discovered through sequencing of several adult cancer types.
SOURCE: Parsons DW et al. ASCO 2019, Abstract 10011.
Researchers have found they can screen pediatric cancer patients for genetic alterations and match those patients to appropriate targeted therapies.
Thus far, 24% of the patients screened have been matched and assigned to a treatment, and 10% have been enrolled on treatment protocols.
The patients were screened and matched as part of the National Cancer Institute–Children’s Oncology Group Pediatric MATCH (Molecular Analysis for Therapy Choice) trial.
Results from this trial are scheduled to be presented at the annual meeting of the American Society of Clinical Oncology.
Donald Williams Parsons, MD, PhD, of Baylor College of Medicine in Houston, Tex., presented some results at a press briefing in advance of the meeting. “[T]he last 10 years have been an incredible time in terms of learning more about the genetics and underlying molecular basis of both adult and pediatric cancers,” Dr. Parsons said.
He pointed out, however, that it is not yet known if this information will be useful in guiding the treatment of pediatric cancers. Specifically, how many pediatric patients can be matched to targeted therapies, and how effective will those therapies be?
The Pediatric MATCH trial (NCT03155620) was developed to answer these questions. Researchers plan to enroll at least 1,000 patients in this trial. Patients are eligible if they are 1-21 years of age and have refractory or recurrent solid tumors, non-Hodgkin lymphomas, or histiocytic disorders.
After patients are enrolled in the trial, their tumor samples undergo DNA and RNA sequencing, and the results are used to match each patient to a targeted therapy. At present, the trial can match patients to one of 10 drugs:
- larotrectinib (targeting NTRK fusions).
- erdafitinib (targeting FGFR1/2/3/4).
- tazemetostat (targeting EZH2 or members of the SWI/SNF complex).
- LY3023414 (targeting the PI3K/MTOR pathway).
- selumetinib (targeting the MAPK pathway).
- ensartinib (targeting ALK or ROS1).
- vemurafenib (targeting BRAF V600 mutations).
- olaparib (targeting defects in DNA damage repair).
- palbociclib (targeting alterations in cell cycle genes).
- ulixertinib (targeting MAPK pathway mutations).
Early results
From July 2017 through December 2018, 422 patients were enrolled in the trial. The patients had more than 60 different diagnoses, including brain tumors, sarcomas, neuroblastoma, renal and liver cancers, and other malignancies.
The researchers received tumor samples from 390 patients, attempted sequencing of 370 samples (95%), and completed sequencing of 357 samples (92%).
A treatment target was found in 112 (29%) patients, 95 (24%) of those patients were assigned to a treatment, and 39 (10%) were enrolled in a protocol. The median turnaround time from sample receipt to treatment assignment was 15 days.
“In addition to the sequencing being successful, the patients are being matched to the different treatments,” Dr. Parsons said. He added that the study is ongoing, so more of the matched and assigned patients will be enrolled in protocols in the future.
Dr. Parsons also presented results by tumor type. A targetable alteration was identified in 26% (67/255) of all non–central nervous system solid tumors, 13% (10/75) of osteosarcomas, 50% (18/36) of rhabdomyosarcomas, 21% (7/33) of Ewing sarcomas, 25% (9/36) of other sarcomas, 19% (5/26) of renal cancers, 16% (3/19) of carcinomas, 44% (8/18) of neuroblastomas, 43% (3/7) of liver cancers, and 29% (4/14) of “other” tumors.
Drilling down further, Dr. Parsons presented details on specific alterations in one cancer type: astrocytomas. Targetable alterations were found in 74% (29/39) of astrocytomas. This includes NF1 mutations (18%), BRAF V600E (15%), FGFR1 fusions/mutations (10%), BRAF fusions (10%), PIK3CA mutations (8%), NRAS/KRAS mutations (5%), and other alterations.
“Pretty remarkably, in this one diagnosis, there are patients who have been matched to nine of the ten different treatment arms,” Dr. Parsons said. “This study is allowing us to evaluate targeted therapies – specific types of investigational drugs – in patients with many different cancer types, some common, some very rare. So, hopefully, we can study these agents and identify signals of activity where some of these drugs may work for our patients.”
The Pediatric MATCH trial is sponsored by the National Cancer Institute. Dr. Parsons has patents, royalties, and other intellectual property related to genes discovered through sequencing of several adult cancer types.
SOURCE: Parsons DW et al. ASCO 2019, Abstract 10011.
REPORTING FROM ASCO 2019
Equal access to care for black men lowers risk of prostate cancer-specific mortality
Black men with newly diagnosed nonmetastatic prostate cancer had no significant difference in prostate cancer–specific mortality compared with white men when treated with a standardized approach and follow-up or at a health care system with standardized access, according to recent research published in JAMA Oncology.
Robert T. Dess, MD, from the department of radiation oncology at the University of Michigan, Ann Arbor, and colleagues examined data from 296,273 patients in the Surveillance, Epidemiology, and End Results (SEER) cohort, 3,972 patients from the Veterans Affairs (VA) health system, and 5,854 patients in four randomized controlled trials (RCTs) from the National Cancer Institute–sponsored Radiation Therapy Oncology Group between January 1992 and December 2013. Of these, 52,840 patients (17.8%) in the SEER cohort, 1,513 patients (38.1%) in the VA cohort, and 1,129 (19.3%) in the RCT cohort were black men. The mean age across all cohorts was 64.9 years, and the median follow-up was 75 months in the SEER cohort, 97 months in the VA cohort, and 104 months in the RCT cohort.
After adjustment for age in the SEER cohort, black men had a 30% increased risk of prostate cancer–specific mortality (PCSM) compared with white men (subdistribution hazard ratio, 1.30; 95% CI, 1.23-1.37; P less than .001). However, after the researchers performed inverse probability weighting (IPW) to adjust for race-based imbalances such as access to care and standardized treatment, there was a 0.5% increased risk over 10 years (sHR, 1.09; 95% CI, 1.04-1.15; P less than .001) after diagnosis.
In the VA cohort, IPW yielded no significant differences between black men and white men (sHR, 0.85; 95% CI, 0.56-1.30; P = .46), and there was a significantly lower risk for black men in the RCT cohort after IPW (sHR, 0.81; 95% CI, 0.66-0.99; P = .04). With regard to other outcomes, other-cause mortality was significantly higher for black men in the SEER cohort (sHR, 1.30; 95% CI, 1.27-1.34; P less than .001) and in the RCT cohort (sHR, 1.17; 95% CI, 1.06-1.29; P = .002) after IPW.
“Black race remains associated with many factors that negatively affect outcomes, and disparities persist at the population level,” Dr. Dess and colleagues wrote in their study. “Continued efforts are needed to address this clear racial health inequity driven by modifiable nonbiological risk factors.”
The researchers said some residual confounding may exist, but noted the strengths of the study included a diverse cohort with a large range of treatment approaches.
This study was funded in part by the Prostate Cancer Foundation, a grant from the Prostate Cancer National Institutes of Health Specialized Programs of Research Excellence, two grants from the Department of Defense, a grant from NIH, and a grant from the NIH Cancer Center. One or more of the authors reported relationships in the form of grants, personal fees, and consulting fees with numerous companies. The other authors report no relevant conflicts of interest.
SOURCE: Dess RT et al. JAMA Oncol. 2019 May 23. doi: 10.1001/jamaoncol.2019.0826.
The results by Dess et al. show that inequality in prostate cancer mortality is the result of socioeconomic barriers that reduce access to care rather than a biological predisposition, Channing J. Paller, MD; Lin Wang, MSc, MMed; and Otis W. Brawley, MD, wrote in an editorial (JAMA Oncol 2019 May 23. doi: 10.1001/jamaoncol.2019.0812).
As registries collect more socioeconomic data from patients, including information on insurance status and health care system where treatment occurred, adjusting for confounders to study the effects of prevention, diagnostic and treatment strategies in addition to “open-minded analyses will help to mitigate some of the prevailing biases” about racial differences in medicine, they said.
While profiling by population can be a useful tool, tracking by geographical location may be a better way to categorize patients, such as glucose-6-phosphate dehydrogenase deficiency that came about as protection from malaria in the case of Mediterranean, African, and Asian populations.
“Although it is still true that men of African origin have a higher incidence of prostate cancer and higher mortality rates, the causes of these differences are complex and may involve exposure to prostate cancer risk factors, genomic differences, and other biology-based factors,” the authors wrote. “Research to determine the relative contributions of these factors should continue; but in the meantime, we as health care professionals are likely to have the greatest effect on improved outcomes for African American patients with prostate cancer by ensuring that they get the same care as white patients, not just in clinical trials but throughout the national health care system.”
In addition, comorbid disease associated with prostate cancer is a significant cause of mortality among African American and white patients, and efforts to treat cancer should also consider cardiovascular disease, diabetes and other comorbidities.
“It is an unsettling fact that there is not equal treatment in the United States. African Americans, other minorities, and the poor in general often experience disparate quality of care or no care at all,” they said. “Although race does not matter biologically, race still matters.”
Dr. Paller and Dr. Brawley are from Johns Hopkins School of Medicine; Lin Wang and Dr. Brawley are from the Johns Hopkins Bloomberg School of Public Health in Baltimore. These comments summarize their editorial in response to Dess et al. Dr. Brawley reports receiving grants from the National Cancer Institute and Bloomberg Philanthropies. The other authors report no relevant conflicts of interest.
The results by Dess et al. show that inequality in prostate cancer mortality is the result of socioeconomic barriers that reduce access to care rather than a biological predisposition, Channing J. Paller, MD; Lin Wang, MSc, MMed; and Otis W. Brawley, MD, wrote in an editorial (JAMA Oncol 2019 May 23. doi: 10.1001/jamaoncol.2019.0812).
As registries collect more socioeconomic data from patients, including information on insurance status and health care system where treatment occurred, adjusting for confounders to study the effects of prevention, diagnostic and treatment strategies in addition to “open-minded analyses will help to mitigate some of the prevailing biases” about racial differences in medicine, they said.
While profiling by population can be a useful tool, tracking by geographical location may be a better way to categorize patients, such as glucose-6-phosphate dehydrogenase deficiency that came about as protection from malaria in the case of Mediterranean, African, and Asian populations.
“Although it is still true that men of African origin have a higher incidence of prostate cancer and higher mortality rates, the causes of these differences are complex and may involve exposure to prostate cancer risk factors, genomic differences, and other biology-based factors,” the authors wrote. “Research to determine the relative contributions of these factors should continue; but in the meantime, we as health care professionals are likely to have the greatest effect on improved outcomes for African American patients with prostate cancer by ensuring that they get the same care as white patients, not just in clinical trials but throughout the national health care system.”
In addition, comorbid disease associated with prostate cancer is a significant cause of mortality among African American and white patients, and efforts to treat cancer should also consider cardiovascular disease, diabetes and other comorbidities.
“It is an unsettling fact that there is not equal treatment in the United States. African Americans, other minorities, and the poor in general often experience disparate quality of care or no care at all,” they said. “Although race does not matter biologically, race still matters.”
Dr. Paller and Dr. Brawley are from Johns Hopkins School of Medicine; Lin Wang and Dr. Brawley are from the Johns Hopkins Bloomberg School of Public Health in Baltimore. These comments summarize their editorial in response to Dess et al. Dr. Brawley reports receiving grants from the National Cancer Institute and Bloomberg Philanthropies. The other authors report no relevant conflicts of interest.
The results by Dess et al. show that inequality in prostate cancer mortality is the result of socioeconomic barriers that reduce access to care rather than a biological predisposition, Channing J. Paller, MD; Lin Wang, MSc, MMed; and Otis W. Brawley, MD, wrote in an editorial (JAMA Oncol 2019 May 23. doi: 10.1001/jamaoncol.2019.0812).
As registries collect more socioeconomic data from patients, including information on insurance status and health care system where treatment occurred, adjusting for confounders to study the effects of prevention, diagnostic and treatment strategies in addition to “open-minded analyses will help to mitigate some of the prevailing biases” about racial differences in medicine, they said.
While profiling by population can be a useful tool, tracking by geographical location may be a better way to categorize patients, such as glucose-6-phosphate dehydrogenase deficiency that came about as protection from malaria in the case of Mediterranean, African, and Asian populations.
“Although it is still true that men of African origin have a higher incidence of prostate cancer and higher mortality rates, the causes of these differences are complex and may involve exposure to prostate cancer risk factors, genomic differences, and other biology-based factors,” the authors wrote. “Research to determine the relative contributions of these factors should continue; but in the meantime, we as health care professionals are likely to have the greatest effect on improved outcomes for African American patients with prostate cancer by ensuring that they get the same care as white patients, not just in clinical trials but throughout the national health care system.”
In addition, comorbid disease associated with prostate cancer is a significant cause of mortality among African American and white patients, and efforts to treat cancer should also consider cardiovascular disease, diabetes and other comorbidities.
“It is an unsettling fact that there is not equal treatment in the United States. African Americans, other minorities, and the poor in general often experience disparate quality of care or no care at all,” they said. “Although race does not matter biologically, race still matters.”
Dr. Paller and Dr. Brawley are from Johns Hopkins School of Medicine; Lin Wang and Dr. Brawley are from the Johns Hopkins Bloomberg School of Public Health in Baltimore. These comments summarize their editorial in response to Dess et al. Dr. Brawley reports receiving grants from the National Cancer Institute and Bloomberg Philanthropies. The other authors report no relevant conflicts of interest.
Black men with newly diagnosed nonmetastatic prostate cancer had no significant difference in prostate cancer–specific mortality compared with white men when treated with a standardized approach and follow-up or at a health care system with standardized access, according to recent research published in JAMA Oncology.
Robert T. Dess, MD, from the department of radiation oncology at the University of Michigan, Ann Arbor, and colleagues examined data from 296,273 patients in the Surveillance, Epidemiology, and End Results (SEER) cohort, 3,972 patients from the Veterans Affairs (VA) health system, and 5,854 patients in four randomized controlled trials (RCTs) from the National Cancer Institute–sponsored Radiation Therapy Oncology Group between January 1992 and December 2013. Of these, 52,840 patients (17.8%) in the SEER cohort, 1,513 patients (38.1%) in the VA cohort, and 1,129 (19.3%) in the RCT cohort were black men. The mean age across all cohorts was 64.9 years, and the median follow-up was 75 months in the SEER cohort, 97 months in the VA cohort, and 104 months in the RCT cohort.
After adjustment for age in the SEER cohort, black men had a 30% increased risk of prostate cancer–specific mortality (PCSM) compared with white men (subdistribution hazard ratio, 1.30; 95% CI, 1.23-1.37; P less than .001). However, after the researchers performed inverse probability weighting (IPW) to adjust for race-based imbalances such as access to care and standardized treatment, there was a 0.5% increased risk over 10 years (sHR, 1.09; 95% CI, 1.04-1.15; P less than .001) after diagnosis.
In the VA cohort, IPW yielded no significant differences between black men and white men (sHR, 0.85; 95% CI, 0.56-1.30; P = .46), and there was a significantly lower risk for black men in the RCT cohort after IPW (sHR, 0.81; 95% CI, 0.66-0.99; P = .04). With regard to other outcomes, other-cause mortality was significantly higher for black men in the SEER cohort (sHR, 1.30; 95% CI, 1.27-1.34; P less than .001) and in the RCT cohort (sHR, 1.17; 95% CI, 1.06-1.29; P = .002) after IPW.
“Black race remains associated with many factors that negatively affect outcomes, and disparities persist at the population level,” Dr. Dess and colleagues wrote in their study. “Continued efforts are needed to address this clear racial health inequity driven by modifiable nonbiological risk factors.”
The researchers said some residual confounding may exist, but noted the strengths of the study included a diverse cohort with a large range of treatment approaches.
This study was funded in part by the Prostate Cancer Foundation, a grant from the Prostate Cancer National Institutes of Health Specialized Programs of Research Excellence, two grants from the Department of Defense, a grant from NIH, and a grant from the NIH Cancer Center. One or more of the authors reported relationships in the form of grants, personal fees, and consulting fees with numerous companies. The other authors report no relevant conflicts of interest.
SOURCE: Dess RT et al. JAMA Oncol. 2019 May 23. doi: 10.1001/jamaoncol.2019.0826.
Black men with newly diagnosed nonmetastatic prostate cancer had no significant difference in prostate cancer–specific mortality compared with white men when treated with a standardized approach and follow-up or at a health care system with standardized access, according to recent research published in JAMA Oncology.
Robert T. Dess, MD, from the department of radiation oncology at the University of Michigan, Ann Arbor, and colleagues examined data from 296,273 patients in the Surveillance, Epidemiology, and End Results (SEER) cohort, 3,972 patients from the Veterans Affairs (VA) health system, and 5,854 patients in four randomized controlled trials (RCTs) from the National Cancer Institute–sponsored Radiation Therapy Oncology Group between January 1992 and December 2013. Of these, 52,840 patients (17.8%) in the SEER cohort, 1,513 patients (38.1%) in the VA cohort, and 1,129 (19.3%) in the RCT cohort were black men. The mean age across all cohorts was 64.9 years, and the median follow-up was 75 months in the SEER cohort, 97 months in the VA cohort, and 104 months in the RCT cohort.
After adjustment for age in the SEER cohort, black men had a 30% increased risk of prostate cancer–specific mortality (PCSM) compared with white men (subdistribution hazard ratio, 1.30; 95% CI, 1.23-1.37; P less than .001). However, after the researchers performed inverse probability weighting (IPW) to adjust for race-based imbalances such as access to care and standardized treatment, there was a 0.5% increased risk over 10 years (sHR, 1.09; 95% CI, 1.04-1.15; P less than .001) after diagnosis.
In the VA cohort, IPW yielded no significant differences between black men and white men (sHR, 0.85; 95% CI, 0.56-1.30; P = .46), and there was a significantly lower risk for black men in the RCT cohort after IPW (sHR, 0.81; 95% CI, 0.66-0.99; P = .04). With regard to other outcomes, other-cause mortality was significantly higher for black men in the SEER cohort (sHR, 1.30; 95% CI, 1.27-1.34; P less than .001) and in the RCT cohort (sHR, 1.17; 95% CI, 1.06-1.29; P = .002) after IPW.
“Black race remains associated with many factors that negatively affect outcomes, and disparities persist at the population level,” Dr. Dess and colleagues wrote in their study. “Continued efforts are needed to address this clear racial health inequity driven by modifiable nonbiological risk factors.”
The researchers said some residual confounding may exist, but noted the strengths of the study included a diverse cohort with a large range of treatment approaches.
This study was funded in part by the Prostate Cancer Foundation, a grant from the Prostate Cancer National Institutes of Health Specialized Programs of Research Excellence, two grants from the Department of Defense, a grant from NIH, and a grant from the NIH Cancer Center. One or more of the authors reported relationships in the form of grants, personal fees, and consulting fees with numerous companies. The other authors report no relevant conflicts of interest.
SOURCE: Dess RT et al. JAMA Oncol. 2019 May 23. doi: 10.1001/jamaoncol.2019.0826.
FROM JAMA ONCOLOGY
Enzalutamide in castration-sensitive prostate cancer and ctDNA to monitor early colorectal cancer
In this edition of “How I will treat my next patient,” I take a look at two recent trials – a late-breaking abstract presented at the annual meeting of the American Urological Association on the value of enzalutamide in hormone-sensitive metastatic prostate cancer (mHSPC) patients and a recent publication in JAMA Oncology about the potential for circulating tumor DNA (ctDNA) testing to inform our management of early stage colorectal cancer (CRC).
ARCHES trial
The ARCHES trial was reported as a late-breaking abstract at AUA 2019. ARCHES was a double-blind, placebo-controlled trial in 1,150 men with mHSPC. Patients were required to have been free from radiographic disease progression or a rising prostate-specific antigen (PSA) level for at least 3 months on androgen deprivation therapy (ADT) or at least 6 months after prior docetaxel chemotherapy.
Patients were randomly assigned to receive either ADT plus enzalutamide (an androgen receptor signaling inhibitor) or ADT plus placebo. The coprimary endpoints of the trial were radiographic progression-free survival (rPFS) and death within 24 weeks.
ADT plus enzalutamide had dramatically better PSA-related endpoints (as expected) and produced significantly better rPFS (median PFS, not reached versus 19.5 months; 61% relative prolongation of rPFS) than placebo plus ADT.
Overall survival data were unreported and may be confounded by all patients being offered enzalutamide at progression. Despite the known adverse effects of enzalutamide from prior studies, enzalutamide-related adverse effects in ARCHES were no worse than placebo (about 85% in both study arms). Formal quality-of-life analyses are yet to be reported.
What this means in practice
It is no surprise that enzalutamide, a potent drug in castration-resistant prostate cancer, would beat placebo. It joins docetaxel and abiraterone in helping to delay the time until castration-resistant disease develops – a meaningful goal. The authors commented that baseline PSA level did not predict benefit from enzalutamide – again, no surprise given that other published trials have suggested that baseline PSA is more likely prognostic than predictive.
It is always prudent to wait for a formal manuscript, but this abstract suggests that men with mHSPC have yet another option for treatment with modest toxicity and broad applicability in practice.
ctDNA in early colorectal cancer
In JAMA Oncology, Yuxuan Wang, MD, PhD, and colleagues summarized their experience with 58 patients with stages I-III colorectal cancer (CRC) who had curative-intent surgical resection at four Swedish hospitals.
The patients had levels of ctDNA monitored every 3 months post operatively. Prediction of the development of metastatic disease using ctDNA was compared to conventional surveillance testing (carcinoembryonic antigen [CEA] blood tests and computed tomographic scanning) per guidelines from the National Comprehensive Cancer Network.
Among the 45 patients with no elevation of ctDNA, there were no recurrences at median follow-up of 49 months. In contrast, 10 of 13 patients (77%) with elevated ctDNA levels during follow-up developed metastatic disease. CEA levels were less sensitive, detecting just 63% of recurrences.
Among the three patients with false positive ctDNA levels, all three fell to undetectable levels with continued follow-up. One of the 18 patients who received adjuvant post-operative chemotherapy had the ctDNA levels fall to undetectable with chemotherapy and that patient remained relapse-free at 37 months.
What this means in practice
The results of this study are remarkably concordant with recently published work in the Journal of Clinical Oncology by Emil Christensen, PhD, and colleagues, that involved patients with localized bladder cancer and illustrate the predictive value of ctDNA over traditional risk factors and conventional surveillance monitoring.
Monitoring ctDNA remains a promising research tool that should not be used for clinical decision making at the present time. However, its potential to help us personalize treatment selection, surveillance intensity, and to select patients who may be spared costly, toxic, and anxiety-provoking treatment and monitoring could be practice changing in the near future.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I will treat my next patient,” I take a look at two recent trials – a late-breaking abstract presented at the annual meeting of the American Urological Association on the value of enzalutamide in hormone-sensitive metastatic prostate cancer (mHSPC) patients and a recent publication in JAMA Oncology about the potential for circulating tumor DNA (ctDNA) testing to inform our management of early stage colorectal cancer (CRC).
ARCHES trial
The ARCHES trial was reported as a late-breaking abstract at AUA 2019. ARCHES was a double-blind, placebo-controlled trial in 1,150 men with mHSPC. Patients were required to have been free from radiographic disease progression or a rising prostate-specific antigen (PSA) level for at least 3 months on androgen deprivation therapy (ADT) or at least 6 months after prior docetaxel chemotherapy.
Patients were randomly assigned to receive either ADT plus enzalutamide (an androgen receptor signaling inhibitor) or ADT plus placebo. The coprimary endpoints of the trial were radiographic progression-free survival (rPFS) and death within 24 weeks.
ADT plus enzalutamide had dramatically better PSA-related endpoints (as expected) and produced significantly better rPFS (median PFS, not reached versus 19.5 months; 61% relative prolongation of rPFS) than placebo plus ADT.
Overall survival data were unreported and may be confounded by all patients being offered enzalutamide at progression. Despite the known adverse effects of enzalutamide from prior studies, enzalutamide-related adverse effects in ARCHES were no worse than placebo (about 85% in both study arms). Formal quality-of-life analyses are yet to be reported.
What this means in practice
It is no surprise that enzalutamide, a potent drug in castration-resistant prostate cancer, would beat placebo. It joins docetaxel and abiraterone in helping to delay the time until castration-resistant disease develops – a meaningful goal. The authors commented that baseline PSA level did not predict benefit from enzalutamide – again, no surprise given that other published trials have suggested that baseline PSA is more likely prognostic than predictive.
It is always prudent to wait for a formal manuscript, but this abstract suggests that men with mHSPC have yet another option for treatment with modest toxicity and broad applicability in practice.
ctDNA in early colorectal cancer
In JAMA Oncology, Yuxuan Wang, MD, PhD, and colleagues summarized their experience with 58 patients with stages I-III colorectal cancer (CRC) who had curative-intent surgical resection at four Swedish hospitals.
The patients had levels of ctDNA monitored every 3 months post operatively. Prediction of the development of metastatic disease using ctDNA was compared to conventional surveillance testing (carcinoembryonic antigen [CEA] blood tests and computed tomographic scanning) per guidelines from the National Comprehensive Cancer Network.
Among the 45 patients with no elevation of ctDNA, there were no recurrences at median follow-up of 49 months. In contrast, 10 of 13 patients (77%) with elevated ctDNA levels during follow-up developed metastatic disease. CEA levels were less sensitive, detecting just 63% of recurrences.
Among the three patients with false positive ctDNA levels, all three fell to undetectable levels with continued follow-up. One of the 18 patients who received adjuvant post-operative chemotherapy had the ctDNA levels fall to undetectable with chemotherapy and that patient remained relapse-free at 37 months.
What this means in practice
The results of this study are remarkably concordant with recently published work in the Journal of Clinical Oncology by Emil Christensen, PhD, and colleagues, that involved patients with localized bladder cancer and illustrate the predictive value of ctDNA over traditional risk factors and conventional surveillance monitoring.
Monitoring ctDNA remains a promising research tool that should not be used for clinical decision making at the present time. However, its potential to help us personalize treatment selection, surveillance intensity, and to select patients who may be spared costly, toxic, and anxiety-provoking treatment and monitoring could be practice changing in the near future.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I will treat my next patient,” I take a look at two recent trials – a late-breaking abstract presented at the annual meeting of the American Urological Association on the value of enzalutamide in hormone-sensitive metastatic prostate cancer (mHSPC) patients and a recent publication in JAMA Oncology about the potential for circulating tumor DNA (ctDNA) testing to inform our management of early stage colorectal cancer (CRC).
ARCHES trial
The ARCHES trial was reported as a late-breaking abstract at AUA 2019. ARCHES was a double-blind, placebo-controlled trial in 1,150 men with mHSPC. Patients were required to have been free from radiographic disease progression or a rising prostate-specific antigen (PSA) level for at least 3 months on androgen deprivation therapy (ADT) or at least 6 months after prior docetaxel chemotherapy.
Patients were randomly assigned to receive either ADT plus enzalutamide (an androgen receptor signaling inhibitor) or ADT plus placebo. The coprimary endpoints of the trial were radiographic progression-free survival (rPFS) and death within 24 weeks.
ADT plus enzalutamide had dramatically better PSA-related endpoints (as expected) and produced significantly better rPFS (median PFS, not reached versus 19.5 months; 61% relative prolongation of rPFS) than placebo plus ADT.
Overall survival data were unreported and may be confounded by all patients being offered enzalutamide at progression. Despite the known adverse effects of enzalutamide from prior studies, enzalutamide-related adverse effects in ARCHES were no worse than placebo (about 85% in both study arms). Formal quality-of-life analyses are yet to be reported.
What this means in practice
It is no surprise that enzalutamide, a potent drug in castration-resistant prostate cancer, would beat placebo. It joins docetaxel and abiraterone in helping to delay the time until castration-resistant disease develops – a meaningful goal. The authors commented that baseline PSA level did not predict benefit from enzalutamide – again, no surprise given that other published trials have suggested that baseline PSA is more likely prognostic than predictive.
It is always prudent to wait for a formal manuscript, but this abstract suggests that men with mHSPC have yet another option for treatment with modest toxicity and broad applicability in practice.
ctDNA in early colorectal cancer
In JAMA Oncology, Yuxuan Wang, MD, PhD, and colleagues summarized their experience with 58 patients with stages I-III colorectal cancer (CRC) who had curative-intent surgical resection at four Swedish hospitals.
The patients had levels of ctDNA monitored every 3 months post operatively. Prediction of the development of metastatic disease using ctDNA was compared to conventional surveillance testing (carcinoembryonic antigen [CEA] blood tests and computed tomographic scanning) per guidelines from the National Comprehensive Cancer Network.
Among the 45 patients with no elevation of ctDNA, there were no recurrences at median follow-up of 49 months. In contrast, 10 of 13 patients (77%) with elevated ctDNA levels during follow-up developed metastatic disease. CEA levels were less sensitive, detecting just 63% of recurrences.
Among the three patients with false positive ctDNA levels, all three fell to undetectable levels with continued follow-up. One of the 18 patients who received adjuvant post-operative chemotherapy had the ctDNA levels fall to undetectable with chemotherapy and that patient remained relapse-free at 37 months.
What this means in practice
The results of this study are remarkably concordant with recently published work in the Journal of Clinical Oncology by Emil Christensen, PhD, and colleagues, that involved patients with localized bladder cancer and illustrate the predictive value of ctDNA over traditional risk factors and conventional surveillance monitoring.
Monitoring ctDNA remains a promising research tool that should not be used for clinical decision making at the present time. However, its potential to help us personalize treatment selection, surveillance intensity, and to select patients who may be spared costly, toxic, and anxiety-provoking treatment and monitoring could be practice changing in the near future.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
ARCHES: Enzalutamide improves mHSPC survival regardless of baseline PSA
For men with metastatic hormone-sensitive prostate cancer (mHSPC), enzalutamide with androgen deprivation therapy (ADT) offers longer radiographic progression-free survival (rPFS) compared with placebo and ADT, regardless of baseline prostate-specific antigen level (PSA), based on results from the phase 3 ARCHES trial.
Although initial PSA level was not predictive of response, treatment with enzalutamide and ADT improved PSA-related efficacy measures, reported lead author Arnulf Stenzl, MD, of the University of Tübingen, Germany, and colleagues. The adverse effect profile of enzalutamide was similar to previous experiences with castrate-resistant patients, they noted in their abstract, which was presented at the annual meeting of the American Urological Association.
ARCHES was a double-blind, placebo-controlled trial involving 1,150 patients with mHSPC, divided approximately 1:1 to receive either enzalutamide with ADT (n = 574) or placebo with ADT (n = 576). Eligibility required that patients had not exhibited radiographic disease progression or rising PSA levels for up to 3 months of ADT, or up to 6 months with prior docetaxel. The primary endpoints were death within 24 weeks of stopping treatment and rPFS. Treatment was continued until unacceptable toxicity or disease progression. PSA levels were measured at baseline and at follow-up, which was an average of 14.4 months.
Median baseline PSA level across both cohorts was 5.21 ng/mL, with most patients having received prior ADT (91%). Baseline PSA was not predictive of response, “suggesting the limitation of baseline PSA as a predictive factor in this population in which most [patients] received prior ADT,” the investigators wrote. Although PSA levels were not predictive, enzalutamide did have a greater impact on PSA-related efficacy endpoints; compared with the placebo group, patients in the enzalutamide arm were significantly more likely to have PSA reductions from baseline of at least 50% (92.9% vs. 56.8%) and at least 90% (72.8% vs. 30.0%). In further support of the efficacy of enzalutamide, median rPFS in the enzalutamide group was significantly better than in the placebo group (not reached vs. 19.4 months). Adverse events were comparable between enzalutamide (85.1%) and placebo (85.9%) cohorts.
Astellas Pharma and Medivation funded the study. The investigators reported no conflicts of interest.
SOURCE: Stenzl et al. AUA 2019. Abstract LBA-10.
For men with metastatic hormone-sensitive prostate cancer (mHSPC), enzalutamide with androgen deprivation therapy (ADT) offers longer radiographic progression-free survival (rPFS) compared with placebo and ADT, regardless of baseline prostate-specific antigen level (PSA), based on results from the phase 3 ARCHES trial.
Although initial PSA level was not predictive of response, treatment with enzalutamide and ADT improved PSA-related efficacy measures, reported lead author Arnulf Stenzl, MD, of the University of Tübingen, Germany, and colleagues. The adverse effect profile of enzalutamide was similar to previous experiences with castrate-resistant patients, they noted in their abstract, which was presented at the annual meeting of the American Urological Association.
ARCHES was a double-blind, placebo-controlled trial involving 1,150 patients with mHSPC, divided approximately 1:1 to receive either enzalutamide with ADT (n = 574) or placebo with ADT (n = 576). Eligibility required that patients had not exhibited radiographic disease progression or rising PSA levels for up to 3 months of ADT, or up to 6 months with prior docetaxel. The primary endpoints were death within 24 weeks of stopping treatment and rPFS. Treatment was continued until unacceptable toxicity or disease progression. PSA levels were measured at baseline and at follow-up, which was an average of 14.4 months.
Median baseline PSA level across both cohorts was 5.21 ng/mL, with most patients having received prior ADT (91%). Baseline PSA was not predictive of response, “suggesting the limitation of baseline PSA as a predictive factor in this population in which most [patients] received prior ADT,” the investigators wrote. Although PSA levels were not predictive, enzalutamide did have a greater impact on PSA-related efficacy endpoints; compared with the placebo group, patients in the enzalutamide arm were significantly more likely to have PSA reductions from baseline of at least 50% (92.9% vs. 56.8%) and at least 90% (72.8% vs. 30.0%). In further support of the efficacy of enzalutamide, median rPFS in the enzalutamide group was significantly better than in the placebo group (not reached vs. 19.4 months). Adverse events were comparable between enzalutamide (85.1%) and placebo (85.9%) cohorts.
Astellas Pharma and Medivation funded the study. The investigators reported no conflicts of interest.
SOURCE: Stenzl et al. AUA 2019. Abstract LBA-10.
For men with metastatic hormone-sensitive prostate cancer (mHSPC), enzalutamide with androgen deprivation therapy (ADT) offers longer radiographic progression-free survival (rPFS) compared with placebo and ADT, regardless of baseline prostate-specific antigen level (PSA), based on results from the phase 3 ARCHES trial.
Although initial PSA level was not predictive of response, treatment with enzalutamide and ADT improved PSA-related efficacy measures, reported lead author Arnulf Stenzl, MD, of the University of Tübingen, Germany, and colleagues. The adverse effect profile of enzalutamide was similar to previous experiences with castrate-resistant patients, they noted in their abstract, which was presented at the annual meeting of the American Urological Association.
ARCHES was a double-blind, placebo-controlled trial involving 1,150 patients with mHSPC, divided approximately 1:1 to receive either enzalutamide with ADT (n = 574) or placebo with ADT (n = 576). Eligibility required that patients had not exhibited radiographic disease progression or rising PSA levels for up to 3 months of ADT, or up to 6 months with prior docetaxel. The primary endpoints were death within 24 weeks of stopping treatment and rPFS. Treatment was continued until unacceptable toxicity or disease progression. PSA levels were measured at baseline and at follow-up, which was an average of 14.4 months.
Median baseline PSA level across both cohorts was 5.21 ng/mL, with most patients having received prior ADT (91%). Baseline PSA was not predictive of response, “suggesting the limitation of baseline PSA as a predictive factor in this population in which most [patients] received prior ADT,” the investigators wrote. Although PSA levels were not predictive, enzalutamide did have a greater impact on PSA-related efficacy endpoints; compared with the placebo group, patients in the enzalutamide arm were significantly more likely to have PSA reductions from baseline of at least 50% (92.9% vs. 56.8%) and at least 90% (72.8% vs. 30.0%). In further support of the efficacy of enzalutamide, median rPFS in the enzalutamide group was significantly better than in the placebo group (not reached vs. 19.4 months). Adverse events were comparable between enzalutamide (85.1%) and placebo (85.9%) cohorts.
Astellas Pharma and Medivation funded the study. The investigators reported no conflicts of interest.
SOURCE: Stenzl et al. AUA 2019. Abstract LBA-10.
FROM AUA 2019
Key clinical point: For men with metastatic hormone-sensitive prostate cancer (mHSPC), enzalutamide with androgen deprivation therapy offers longer radiographic progression-free survival (rPFS) compared with placebo and androgen deprivation therapy, regardless of baseline prostate-specific antigen level (PSA).
Major finding: A greater percentage of patients treated with enzalutamide and androgen deprivation therapy had a prostate-specific antigen reduction from baseline of at least 90%, compared with those who received placebo and androgen deprivation therapy (72.8% vs. 30.0%).
Study details: ARCHES was a double-blind, placebo-controlled, phase 3 study involving 1,150 men with metastatic hormone-sensitive prostate cancer.
Disclosures: The study was funded by Astellas Pharma and Medivation. The investigators reported no conflicts of interest.
Source: Stenzl et al. AUA 2019. Abstract LBA-10.
Management of Castration-Resistant Prostate Cancer
Prostate cancer is the most common malignancy in men, with an estimated 165,000 new prostate cancer diagnoses and 29,000 prostate cancer deaths occurring in the United States in 2018.1 Due to the widespread use of screening prostate-specific antigen (PSA), prostate cancer has been mainly diagnosed when the tumor is confined to the prostate. Despite definitive treatment of localized prostate cancer, some men develop systemic disease, either biochemical failure, as defined by rising PSA level, or metastatic disease.1 Several factors have been demonstrated to predict risk of relapse, including higher pretreatment PSA, higher Gleason score, and a greater anatomic extent of disease.2 In addition, the incidence of de novo metastatic prostate cancer was recently noted to be increasing. This may be due to changes in the United States Preventive Services Task Force prostate cancer screening guidelines in 2012, which recommended against screening for prostate cancer in men of any age. The updated 2018 guidelines recommend a discussion of the risks versus benefits of screening for prostate cancer for all men aged 55 to 69 years,recommend against screening for men older than 70 years, and do not have recommendations for high-risk subgroups.3
Androgen deprivation therapy (ADT) has been the cornerstone of therapy since 1941 for men with hormone-sensitive systemic disease, both in biochemically relapsed and metastatic disease.4,5 While more than 90% of patients respond to initial ADT, castration resistance is inevitable in some men.6,7 Prostate cancer will become castration-resistant typically after 18 to 24 months of ADT, with the majority of patients developing castration-resistant prostate cancer (CRPC) within 5 years of initiation of ADT.8
Pathogenesis
CRPC (previously called androgen independent prostate cancer) is defined as progression of disease despite serum total testosterone levels less than 50 ng/dL. CRPC is characterized by a rising PSA level and/or radiographic progression. One mechanism of castration resistance is genetic modification of the androgen receptor (AR), including increased expression of the wild-type AR.9 Alternatively, mutations of the steroid-binding domain may play a role in the development of castration resistance by allowing the AR to become activated by non-androgen steroid hormones or even paradoxically by antiandrogens. Studies suggest, however, that AR mutations may be seen in only 10% of prostate cancers that have developed castration resistance.10 The AR-V7 splice variant of the AR lacks an androgen binding site altogether, and may play an important role in castration resistance. In one study, the presence of this splice variant in circulating prostate cancer tumor cells predicted resistance to enzalutamide and abiraterone as well as poor outcomes.11 Intratumoral androgen synthesis also may play a role in the development of CRPC.12,13
CRPC can be broadly categorized into 2 categories, metastatic (mCRPC) and nonmetastatic (nmCRPC; Figure). The exact proportion of patients entering CRPC at a nonmetastatic stage (M0) is largely unknown.14 In one study of patients at the time of diagnosis of CRPC, ≥ 84% of patients were shown to have metastases.8 In this article, we review key aspects of management of CRPC, including selection of first- and second-line therapy, and briefly discuss upcoming clinical trials.
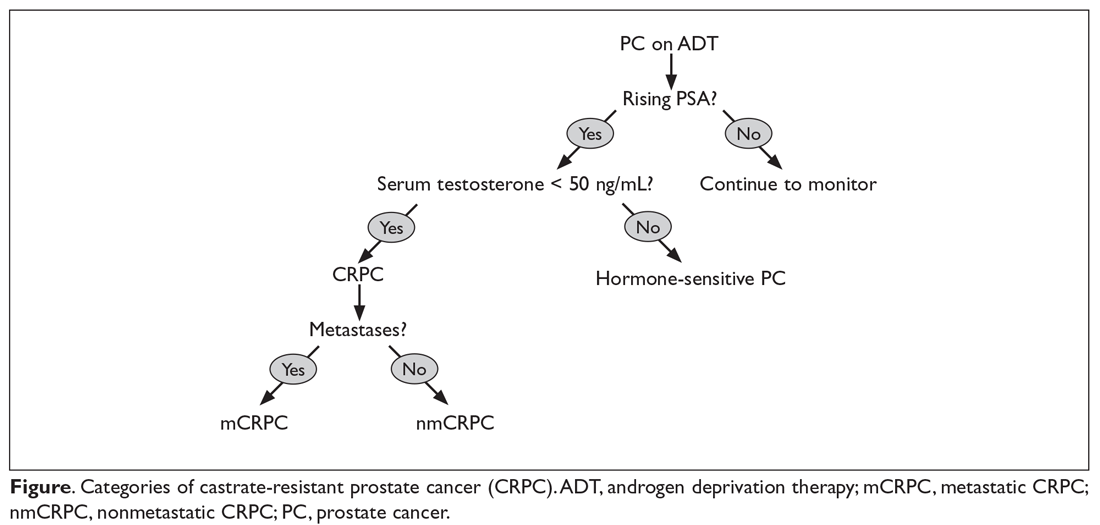
Treatment of Nonmetastatic CRPC (M0 Disease)
Early identification of M0 CRPC is important because patients with nonmetastatic CRPC are at risk for metastasis, as demonstrated by Smith and colleagues.15 In this study that evaluated data from patients with nmCRPC in the placebo group (n = 331) of a randomized controlled trial, at 2 years 46% had developed ≥ 1 bone metastasis, 20% had died, and the median bone metastasis-free survival (MFS) was 25 months.15
Rapid PSA doubling time (PSADT) is linked to shorter time to metastasis in this group of patients. Patients with a PSADT of < 10 months had a risk for bone metastasis 12 times greater and a risk for death 4 times greater than patients with a PSADT of ≥ 10 months.16 Accordingly, observation should be reserved for those patients with a PSADT of ≥ 10 months.
Options for secondary hormonal therapy in those with a PSADT of ≤ 10 months include a first-generation antiandrogen (bicalutamide, flutamide, nilutamide), ketoconazole with hydrocortisone, and more recently second-generation antiandrogens (apalutamide or enzalutamide).
Bicalutamide competitively inhibits dihydrotestosterone and testosterone binding to the AR and is generally well-tolerated; it is given in conjunction with a GnRH agonist/castration.17 The use of other first-generation antiandrogens is limited mainly due to their toxicity profile. When compared to flutamide in a randomized, double-blinded control study, bicalutamide had significantly improved time to treatment failure.18 Due to promiscuous binding to AR, withdrawal of first-generation antiandrogen therapy has been associated with a biochemical response in a small proportion of patients, with response typically seen after 5 to 7 half-lives of the drug have elapsed.19
Although traditionally used as an antifungal agent, ketoconazole also inhibits androgen synthesis in the adrenal glands and acts as a direct cytotoxin to cancer cells.20 Ketoconazole (with hydrocortisone) has been considered as a treatment option, typically at the time of antiandrogen withdrawal. However, ketoconazole offers no survival benefit, and with the approval of abiraterone in M1 CRPC, its use has declined significantly.21 Additionally, ketoconazole poses a risk for severe hepatotoxicity and QT prolongation, and has significant interactions with numerous drugs, thereby limiting its use. Given the typically short duration of response to first-generation antiandrogens, the second-generation antiandrogens were developed and are associated with a significantly greater progression-free survival (PFS) in M0 CRPC.22,23
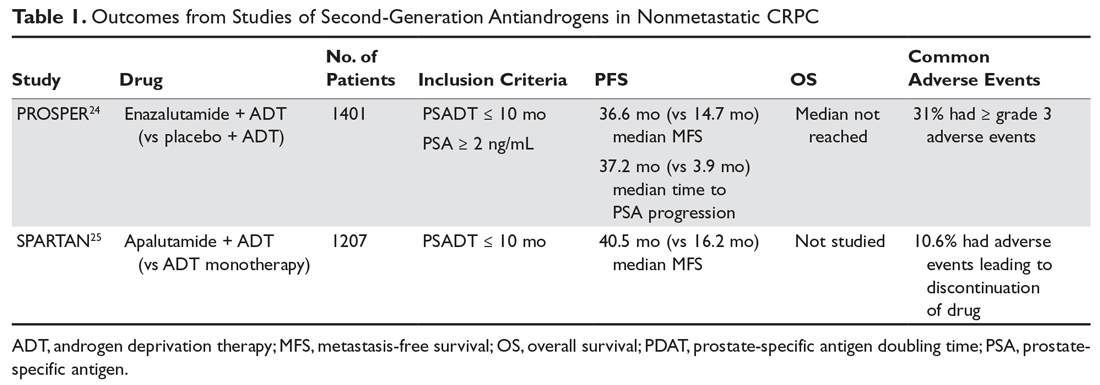
The second-generation antiandrogens enzalutamide and apalutamide not only competitively bind to the AR, inhibiting formation of the androgen/AR complex, but they also inhibit androgen/AR complex nuclear translocation and binding to nuclear DNA. In the PROSPER trial, enzalutamide significantly increased radiographic PFS and improved quality of life compared to placebo in chemotherapy-naive patients (Table 1).24 Apalutamide significantly increased MFS as well as PFS and time to PSA progression compared to placebo in the phase 3 SPARTAN trial.25 Apalutamide is generally well tolerated, with hypertension and rash being the most common severe adverse effects. Apalutamide also has less potential for central nervous system toxicities than enzalutamide. The recent approval of these agents is likely to change responses to subsequent treatments, especially in the metastatic setting.
Treatment of Metastatic CRPC (M1 Disease)
As with M0 CRPC, ADT should be continued in patients with mCRPC to maintain castration levels of testosterone while initiating additional treatments. Several drugs for the treatment of mCRPC have been approved by the US Food and Drug Administration (FDA) since 2010, including abiraterone with prednisone (or methylprednisolone), enzalutamide (but not apalutamide), radium-223, sipuleucel-T, and cabazitaxel (Table 2).
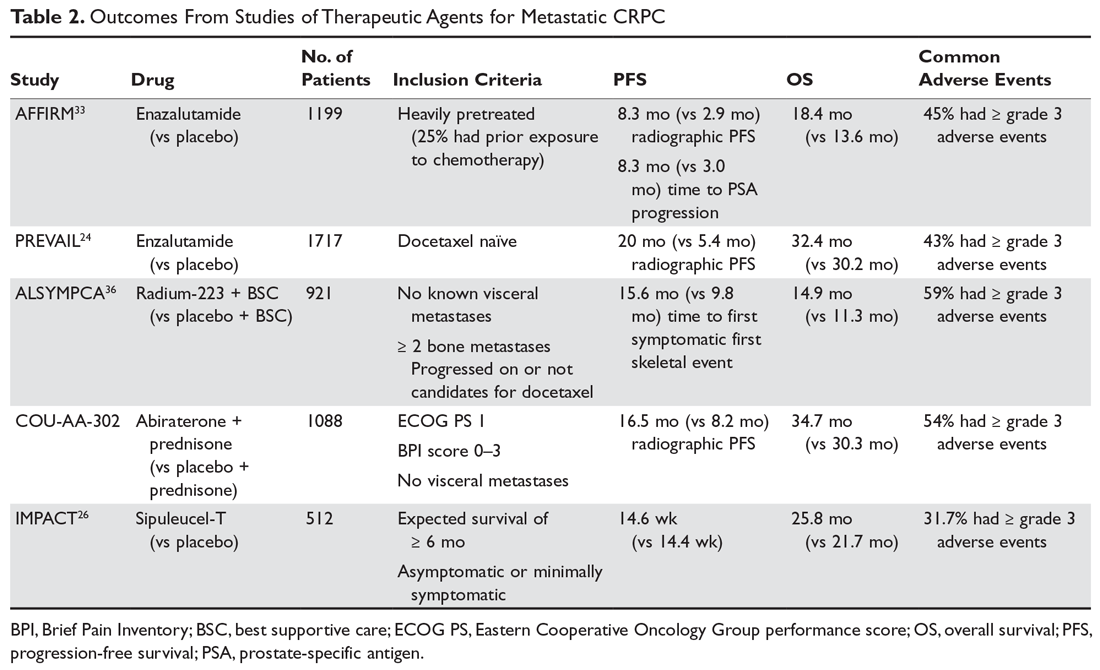
Given the availability of numerous treatment options for men with mCRPC, the sequencing of treatments should be based on careful consideration of the efficacy and adverse effect profiles of each drug as well as the anatomic and molecular characteristics of the cancer, comorbidities, and patient preference. If there is no evidence of visceral disease and the patient has an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1 with an estimated life expectancy of greater than 6 months and is minimally symptomatic, then treatment with either oral targeted agents or immunotherapy with sipuleucel-T is considered appropriate.
Sipuleucel-T is an autologous dendritic cell vaccination designed to enhance T-cell–mediated response to prostatic acid phosphatase (PAP). The treatments are prepared from leukapheresed host mononuclear cells that are then exposed to PAP fused to granulocyte-macrophage colony-stimulating factor. The activated dendritic cells are then infused back into the host once every 2 weeks for a total of 3 treatments. The main side effects of this treatment include chills, fever, and headache, but it is generally well-tolerated and has demonstrated a survival benefit.26
Both enzalutamide and abiraterone (abiraterone given with physiologic-dose steroid replacement) confer a survival benefit in chemotherapy-naive patients with M1 CRPC. Per the PREVAIL study, enzalutamide (when compared to placebo) offers a median improvement in overall survival (OS) by about 2 months and in radiographic PFS by about 14.6 months.24 Abiraterone blocks the synthesis of androgens via inhibition of CYP17 in the testes and adrenal glands. Abiraterone also confers an overall survival advantage for patients with M1 CRPC who are chemotherapy-naïve, with an estimated 25% decrease in the risk of death (hazard ratio, 0.75, P = 0.009) when compared to prednisone.27
In patients with symptomatic M1 CRPC who have visceral disease or rapidly progressive disease and who are candidates for chemotherapy, docetaxel is frequently used and is given concurrently with steroids. Docetaxel has been given for up to 10 cycles in clinical trials (assuming no progression of disease or dose-limiting toxicities were observed), and at least 6 cycles of treatment are recommended. When compared to mitoxantrone plus prednisone in the TAX 327 phase 3 trial, docetaxel plus prednisone offered a significant OS benefit of about 3 months (19.2 months versus 16.3 months).28 For patients who are not candidates for docetaxel (eg, due to preexisting peripheral neuropathy), cabazitaxel should be considered. OS is similar for mCRPC with docetaxel versus cabazitaxel when given in the first-line setting.29 Additionally, cabazitaxel dosed at 20 mg/m2 is noninferior to cabazitaxel dosed at 25 mg/m2, and the lower dose is associated with lower rates of peripheral neuropathy.30 Cabazitaxel should also be considered for mCRPC that has progressed following treatment with docetaxel, with improved OS and PFS when compared to treatment with mitoxantrone and prednisone in this setting, as shown in the TROPIC study.31 Mitoxantrone given with prednisone has been shown to improve quality of life, but it is associated with significant cardiac toxicity. Additionally, mitoxantrone does not improve disease-free survival or OS in chemotherapy-naive patients32 or in patients who have progressed on docetaxel, and therefore should not be given to patients prior to a taxane chemotherapy unless the patient absolutely cannot tolerate docetaxel or cabazitaxel.
Once a patient’s prostate cancer progresses following treatment with a taxane, the sequence in which to administer subsequent therapies should involve careful consideration of previous treatments and duration of response to each of these treatments. Both enzalutamide and abiraterone are FDA-approved for use following treatment with chemotherapy. Per the AFFIRM trial, heavily pre-treated patients (including those who have received docetaxel) have a median 5-month OS benefit with enzalutamide compared to placebo.33 Another study of M1 CRPC patients who had previously received docetaxel demonstrated an OS benefit with abiraterone (versus placebo),34 but this regimen has limited benefit in patients who have previously received both docetaxel and enzalutamide.35 A rechallenge with docetaxel therapy also can be considered if the patient’s disease responded to docetaxel in the metastatic hormone-sensitive setting.
If the patient’s metastases are limited to bone (ie, no visceral disease), then radiotherapy with radium-223 should be considered. Radium-223 is an alpha-emitting calcium-mimetic radioactive compound that tracks to bone to delay the onset of symptoms from bone metastases.36 Radium-223 also confers a median OS benefit of about 3 months.37 However, this treatment is often limited by preexisting cytopenias.
Diethylstilbestrol (1 mg daily) competes with androgens for AR binding and is also cytotoxic to androgen-sensitive and insensitive prostate cancer cells. While its efficacy is similar to bicalutamide in terms of PSA response rate and median response duration, diethylstilbestrol is associated with significantly more cardiovascular toxicity, including stroke, pulmonary embolism, and heart failure, and its use is therefore limited.38 The glucocorticoids—prednisone (5 mg orally twice daily), dexamethasone (0.5 mg daily), and hydrocortisone (40 mg daily)—inhibit pituitary synthesis of adrenocorticotropic hormone, resulting in decreased adrenal androgen synthesis. Data suggest that among the glucocorticoids, dexamethasone monotherapy may produce superior response rates compared to prednisone monotherapy.39 While the glucocorticoids do produce a PSA response, prolong time to disease progression, and can provide symptomatic relief (eg, from bone pain), they have not been shown to confer a survival benefit and therefore are not commonly used as monotherapy.
Future of CRPC Treatment
Patients with CRPC should be considered for clinical trials when available. These patients’ tumors should be assessed with next-generation sequencing for analysis of microsatellite instability (MSI) or mismatch repair (MMR) as well as the presence of other potentially targetable mutations, as this information may bring into consideration additional investigational as well as FDA-approved treatment options. As of May 2017, immunotherapy with pembrolizumab is approved for patients whose prostate cancer is deficient in MMR or has a high MSI burden based on a study of 12 solid tumor types (including prostate cancer) with deficient MMR.40 Additionally, for patients whose tumor has ≥ 1% programmed death ligand 1 (PD-L1) expression, pembrolizumab has a 17% overall response rate and confers stability of disease in 35%, with a median response duration of 13.5 months.41 Cabozantinib is a mesenchymal epithelial transition (MET) kinase and vascular endothelial growth factor receptor (VEGF-R) inhibitor. When used in heavily pre-treated patients with mCRPC, it showed a radiographic PFS benefit but no survival benefit over prednisone monotherapy.42 One study showed that for patients whose mCRPC had a homozygous deletion and/or a deleterious mutation in the homologous recombination repair genes BRCA1/2, ATM, and CHEK2 or the Fanconi anemia genes, the response rate to the poly ADP ribose polymerase (PARP) inhibitor olaparib was 88%, with a 100% response rate in those with BRCA2 mutations.43 Furthermore, mutations in these DNA repair genes predict increased sensitivity to platinum-based chemotherapy.
Supportive Care
Zoledronic acid or denosumab are FDA approved for men with CRPC and bone metastasis based on the ability of these agents to delay skeletal-related events, including pathologic fracture and spinal cord compression.46 Bisphosphonates, however, do not decrease the incidence of bone metastases.47 And while denosumab does delay the time to first bone metastasis in nmCRPC (particularly in patients with a PSADT of ≤ 6 months), it does not improve OS.48 Other supportive measures include exercise and nutrition. Moderate aerobic exercise for 150 minutes in addition to 2 or 3 strength training sessions per week is recommended by the American College of Sport Medicine to combat cancer-related fatigue.49 There are currently no dietary changes that are routinely recommended to improve the outcome of prostate cancer, but a study noted a shorter biochemical failure–free survival in men with prostate cancer who were obese and consumed a diet high in saturated fat.50
Conclusion
Prostate cancer affects more men in the United States than any other cancer. Once a patient is started on hormone therapy, in all likelihood their prostate cancer will become castration-resistant. Once prostate cancer has developed hormone resistance, there are a host of further treatment options available, including further hormone therapy, chemotherapy, immunotherapy, radiation therapy, bone-targeting agents, and clinical trials. Determining the appropriate sequence in which to use these therapies requires knowledge of the natural history of CRPC, the indications for changing therapies, the mechanism of action and adverse event profile of each treatment, and the optimal time to enroll in a clinical trial.
1. Pound CR, Partin AW, Epstein JI, Walsh PC. Prostate-specific antigen after anatomic radical retropubic prostatectomy. Patterns of recurrence and cancer control. Urol Clin North Am. 1997;24:395-406.
2. Caire AA, Sun L, Ode O, et al. Delayed prostate-specific antigen recurrence after radical prostatectomy: how to identify and what are their clinical outcomes? Urology. 2009;74:643-647.
3. US Preventive Services Task Force, Grossman DC, Curry SJ, Owens DK, et al. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:1901-1913.
4. Huggins C, Hodges CV. Studies on prostatic cancer. I: The effects of castration, of estrogen, and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res. 1941;1:293-297.
5. Huggins C, Stevens RE Jr, Hodges CV. Studies on prostatic cancer. II: The effects of castration on advanced carcinoma of the prostate gland. Arch Surg. 1941;43:209-223.
6. Pomerantz M, Kantoff P. Clinical progression to castration recurrent prostate cancer. In: Tindall DJ, James M, eds. Androgen Action in Prostate Cancer. New York: Springer; 2009:57-72.
7. Kirby M, Hirst C, Crawford ED. Characterising the castration-resistant prostate cancer population: a systematic review. Int J Clin Pract. 2011;65:1180-1192.
8. Sharifi N, Dahut WL, Steinberg SM, et al. A retrospective study of the time to clinical endpoints for advanced prostate cancer. BJU Int. 2005;96:985-989.
9. Chen CD, Welsbie DS, Tran C, et al. Molecular determinants of resistance to antiandrogen therapy. Nat Med. 2004;10:33-39.
10. Taplin ME, Rajeshkumar B, Halabi S, et al. Androgen receptor mutations in androgen-independent prostate cancer: Cancer and Leukemia Group B Study 9663. J Clin Oncol. 2003;21:2673-2678.
11. Antonarakis ES, Lu C, Luber B, et al. Clinical significance of androgen receptor splice variant-7 (AR-V7) mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line abiraterone and enzalutamide. J Clin Oncol. 2017;35:2149-2156.
12. Kahn B, Collazo J, Kyprianou N. Androgen receptor as a driver of therapeutic resistance in advanced prostate cancer. Int J Biol Sci. 2014;10:588-595.
13. Logothetis CJ, Gallick GE, Maity SN, et al. Molecular classification of prostate cancer progression: foundation for marker-driven treatment of prostate cancer. Cancer Discov. 2013;3:849-861
14. Tombal B. Non-metastatic CRPC and asymptomatic metastatic CRPC: which treatment for which patient? Ann Oncol. 2012;23(suppl 10):251-258
15. Smith MR, Cook R, Lee KA, et al. Disease and host characteristics as predictors of time to first bone metastasis and death in men with progressive castration-resistant non-metastatic prostate cancer. Cancer. 2011;117:2077-2085.
16. Metwalli AR, Rosner IL, Cullen J, et al. Elevated alkaline phosphatase velocity strongly predicts overall survival and the risk of bone metastases in castrate-resistant prostate cancer. Urol Oncol. 2014;32:761-768
17. Akaza H, Yamaguchi A, Matsuda T, et al. Superior anti-tumor efficacy of bicalutamide 80mg in combination with luteinizing hormone-releasing hormone (LHRH) against versus LHRH agonist monotherapy as first line treatment for advanced prostate cancer: Interim results of a randomized study in Japanese patients. J Clin Oncol. 2004;34:20-28
18. Schellhammer P, Patterson AL, Sharifi R, et al. A controlled trial of bicalutamide versus flutamide, each in combination with luteinizing hormone-releasing hormone analogue therapy, in patients with advanced prostate cancer. Urology. 1995;45(5):745-752.
19. Sartor AO, Tangen CM, Hussain MH, et al. Antiandrogen withdrawal in castrate-refractory prostate cancer: a Southwest Oncology Group Trial (SWOG 9426). Cancer. 2008;112:2393-2400.
20. Eichenberger T, Trachtenberg J, Toor P, et al. Ketoconazole: a possible direct cytotoxic effect on prostate carcinoma cells. J Urol. 1989;141:190-191.
21. Small EJ, Halabi S, Dawson NA, et al. Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583). J Clin Oncol. 2004;22:1025-1033.
22. Penson DF, Armstrong AJ, Concepcion R, et al. Enzalutamide versus bicalutamide in castration resistant prostate cancer: the STRIVE trial. J Clin Oncol. 2016;34:2098-2106.
23. Shore ND, Chowdhury S, Villers A, et al. Efficacy and safety of enzalutamide versus bicalutamide for patients with metastatic prostate cancer (TERRAIN): a randomized, double-blind, phase 2 study. Lancet Oncol. 2016;199:147-154.
24. Beer TM, Armstrong AJ, Rathkopf D, et al. Enzalutamide in men with chemotherapy-naïve castration-resistant prostate cancer: extended analysis of the phase 3 PREVAIL study. Eur Urol. 2017;71:151-154.
25. Smith MR, Saad F, Chowdhury S; SPARTAN Investigators, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408-1418.
26. Kantoff PW, Higano CS, Shore ND, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411-422.
27. Ryan CJ, Smith MR, de Bono JS, et al. Randomized phase 3 trial of abiraterone acetate in men with metastatic castration-resistant prostate cancer and no prior chemotherapy. N Engl J Med. 2013;368:138-148
28. Berthold DR, Pond GR, Soban F, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: updated survival in the TAX 327 study. J Clin Oncol. 2008;26:242-245.
29. Oudard S, Fizazi K, Sengelov L, et al. Cabazitaxel versus docetaxel as first-line therapy for patients with metastatic castration-resistant prostate cancer: a randomized phase III trial—FIRSTANA. J Clin Oncol. 2017;35:3189-3197.
30. Eisenberger M, Hardy-Bessard A-C, Kim CS, et al. Phase III study comparing a reduced dose of cabazitaxel (20 mg/m2) and the currently approved dose (25 mg/m2) in postdocetaxel patients with metastatic castration-resistant prostate cancer—PROSELICA. J Clin Oncol. 2017;35:3198-3206.
31. de Bono JS, Oudard S, Ozguroglu M, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376(9747):P1147-1154.
32. Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004;351:1502-1512.
33. Fizazi K, Scher HI, Miller K, et al. Effect of enzalutamide on time to first skeletal-related event, pain, and quality of life in men with castration-resistant prostate cancer: results from the randomised, phase 3 AFFIRM trial. Lancet Oncol. 2014;15:1147-1156.
34. de Bono JS, Logothetis CJ, Molina A, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995-2005.
35. Loriot Y, Bianchini D, Ileana E, et al. Antitumor activity of abiraterone acetate against metastatic castration-resistant prostate cancer progressing after docetaxel and enzalutamide. Ann Oncol. 2013;24:1807-1812.
36. Sartor O, Coleman R, Nilsson S, et al. Effect of radium-223 dichloride on symptomatic skeletal events in patients with castration-resistant prostate cancer and bone metastases: results from a phase 3, double blind, randomized trial. Lancet Oncol. 2014;15:738-746.
37. Parker C, Nilsson S, Heinrich D, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013; 369:213-223.
38. Turo R, Smolski M, Esler R, et al. Diethylstilboestrol for the treatment of prostate cancer: past, present and future. Scand J Urol. 2014;48:4-14.
39. Venkitaraman R, Lorente D, Murthy V. A randomized phase 2 trial of dexamethasone versus prednisolone in castration-resistant prostate cancer. Eur Urol. 2015 67:673-679.
40. Le DT, Durham JN, Smith KN, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357(6349):409-413.
41. Hansen AR, Massard C, Ott PA, et al. Pembrolizumab for advanced prostate adenocarcinoma: findings of the KEYNOTE-028 study. Ann Oncol. 2018;29:1807-1813.
42. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
43. Mateo J, Carreira S, Sandhu S, et al. DNA-Repair defects and olaparib in metastatic prostate cancer. N Engl J Med. 2015;373:1697-1708.
44. Kentepozidis N, Soultati A, Giassas S, et al. Paclitaxel in combination with carboplatin as salvage treatment in patients with castration-resistant prostate cancer: a Hellenic oncology research group multicenter phase II study. Cancer Chemother Pharmacol. 2012;70:161-168.
45. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
46. Saad F, Gleason DM, Murray R, et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst. 2002;94:1458-1468.
47. Wirth M, Tammela T, Cicalese V, et al. Prevention of bone metastases in patients with high-risk nonmetastatic prostate cancer treated with zoledronic acid: efficacy and safety results of the Zometa European Study (ZEUS). Eur Urol. 2015;67:482-491.
48. Smith MR, Saad F, Coleman R, et al. Denosumab and bone-metastases-free survival in men with castration-resistant prostate cancer: results of a phase 3, randomized, placebo-controlled trial. Lancet. 2012;379(9810):39-46.
49. Schmitz KH, Courneya KS, Matthews C, et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42:1409-1426.
50. Strom SS, Yamamura Y, Flores-Sandoval FN, et al. Prostate cancer in Mexican-Americans: identification of risk factors. Prostate. 2008;68:563-570.
Prostate cancer is the most common malignancy in men, with an estimated 165,000 new prostate cancer diagnoses and 29,000 prostate cancer deaths occurring in the United States in 2018.1 Due to the widespread use of screening prostate-specific antigen (PSA), prostate cancer has been mainly diagnosed when the tumor is confined to the prostate. Despite definitive treatment of localized prostate cancer, some men develop systemic disease, either biochemical failure, as defined by rising PSA level, or metastatic disease.1 Several factors have been demonstrated to predict risk of relapse, including higher pretreatment PSA, higher Gleason score, and a greater anatomic extent of disease.2 In addition, the incidence of de novo metastatic prostate cancer was recently noted to be increasing. This may be due to changes in the United States Preventive Services Task Force prostate cancer screening guidelines in 2012, which recommended against screening for prostate cancer in men of any age. The updated 2018 guidelines recommend a discussion of the risks versus benefits of screening for prostate cancer for all men aged 55 to 69 years,recommend against screening for men older than 70 years, and do not have recommendations for high-risk subgroups.3
Androgen deprivation therapy (ADT) has been the cornerstone of therapy since 1941 for men with hormone-sensitive systemic disease, both in biochemically relapsed and metastatic disease.4,5 While more than 90% of patients respond to initial ADT, castration resistance is inevitable in some men.6,7 Prostate cancer will become castration-resistant typically after 18 to 24 months of ADT, with the majority of patients developing castration-resistant prostate cancer (CRPC) within 5 years of initiation of ADT.8
Pathogenesis
CRPC (previously called androgen independent prostate cancer) is defined as progression of disease despite serum total testosterone levels less than 50 ng/dL. CRPC is characterized by a rising PSA level and/or radiographic progression. One mechanism of castration resistance is genetic modification of the androgen receptor (AR), including increased expression of the wild-type AR.9 Alternatively, mutations of the steroid-binding domain may play a role in the development of castration resistance by allowing the AR to become activated by non-androgen steroid hormones or even paradoxically by antiandrogens. Studies suggest, however, that AR mutations may be seen in only 10% of prostate cancers that have developed castration resistance.10 The AR-V7 splice variant of the AR lacks an androgen binding site altogether, and may play an important role in castration resistance. In one study, the presence of this splice variant in circulating prostate cancer tumor cells predicted resistance to enzalutamide and abiraterone as well as poor outcomes.11 Intratumoral androgen synthesis also may play a role in the development of CRPC.12,13
CRPC can be broadly categorized into 2 categories, metastatic (mCRPC) and nonmetastatic (nmCRPC; Figure). The exact proportion of patients entering CRPC at a nonmetastatic stage (M0) is largely unknown.14 In one study of patients at the time of diagnosis of CRPC, ≥ 84% of patients were shown to have metastases.8 In this article, we review key aspects of management of CRPC, including selection of first- and second-line therapy, and briefly discuss upcoming clinical trials.

Treatment of Nonmetastatic CRPC (M0 Disease)
Early identification of M0 CRPC is important because patients with nonmetastatic CRPC are at risk for metastasis, as demonstrated by Smith and colleagues.15 In this study that evaluated data from patients with nmCRPC in the placebo group (n = 331) of a randomized controlled trial, at 2 years 46% had developed ≥ 1 bone metastasis, 20% had died, and the median bone metastasis-free survival (MFS) was 25 months.15
Rapid PSA doubling time (PSADT) is linked to shorter time to metastasis in this group of patients. Patients with a PSADT of < 10 months had a risk for bone metastasis 12 times greater and a risk for death 4 times greater than patients with a PSADT of ≥ 10 months.16 Accordingly, observation should be reserved for those patients with a PSADT of ≥ 10 months.
Options for secondary hormonal therapy in those with a PSADT of ≤ 10 months include a first-generation antiandrogen (bicalutamide, flutamide, nilutamide), ketoconazole with hydrocortisone, and more recently second-generation antiandrogens (apalutamide or enzalutamide).
Bicalutamide competitively inhibits dihydrotestosterone and testosterone binding to the AR and is generally well-tolerated; it is given in conjunction with a GnRH agonist/castration.17 The use of other first-generation antiandrogens is limited mainly due to their toxicity profile. When compared to flutamide in a randomized, double-blinded control study, bicalutamide had significantly improved time to treatment failure.18 Due to promiscuous binding to AR, withdrawal of first-generation antiandrogen therapy has been associated with a biochemical response in a small proportion of patients, with response typically seen after 5 to 7 half-lives of the drug have elapsed.19
Although traditionally used as an antifungal agent, ketoconazole also inhibits androgen synthesis in the adrenal glands and acts as a direct cytotoxin to cancer cells.20 Ketoconazole (with hydrocortisone) has been considered as a treatment option, typically at the time of antiandrogen withdrawal. However, ketoconazole offers no survival benefit, and with the approval of abiraterone in M1 CRPC, its use has declined significantly.21 Additionally, ketoconazole poses a risk for severe hepatotoxicity and QT prolongation, and has significant interactions with numerous drugs, thereby limiting its use. Given the typically short duration of response to first-generation antiandrogens, the second-generation antiandrogens were developed and are associated with a significantly greater progression-free survival (PFS) in M0 CRPC.22,23
The second-generation antiandrogens enzalutamide and apalutamide not only competitively bind to the AR, inhibiting formation of the androgen/AR complex, but they also inhibit androgen/AR complex nuclear translocation and binding to nuclear DNA. In the PROSPER trial, enzalutamide significantly increased radiographic PFS and improved quality of life compared to placebo in chemotherapy-naive patients (Table 1).24 Apalutamide significantly increased MFS as well as PFS and time to PSA progression compared to placebo in the phase 3 SPARTAN trial.25 Apalutamide is generally well tolerated, with hypertension and rash being the most common severe adverse effects. Apalutamide also has less potential for central nervous system toxicities than enzalutamide. The recent approval of these agents is likely to change responses to subsequent treatments, especially in the metastatic setting.

Treatment of Metastatic CRPC (M1 Disease)
As with M0 CRPC, ADT should be continued in patients with mCRPC to maintain castration levels of testosterone while initiating additional treatments. Several drugs for the treatment of mCRPC have been approved by the US Food and Drug Administration (FDA) since 2010, including abiraterone with prednisone (or methylprednisolone), enzalutamide (but not apalutamide), radium-223, sipuleucel-T, and cabazitaxel (Table 2).

Given the availability of numerous treatment options for men with mCRPC, the sequencing of treatments should be based on careful consideration of the efficacy and adverse effect profiles of each drug as well as the anatomic and molecular characteristics of the cancer, comorbidities, and patient preference. If there is no evidence of visceral disease and the patient has an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1 with an estimated life expectancy of greater than 6 months and is minimally symptomatic, then treatment with either oral targeted agents or immunotherapy with sipuleucel-T is considered appropriate.
Sipuleucel-T is an autologous dendritic cell vaccination designed to enhance T-cell–mediated response to prostatic acid phosphatase (PAP). The treatments are prepared from leukapheresed host mononuclear cells that are then exposed to PAP fused to granulocyte-macrophage colony-stimulating factor. The activated dendritic cells are then infused back into the host once every 2 weeks for a total of 3 treatments. The main side effects of this treatment include chills, fever, and headache, but it is generally well-tolerated and has demonstrated a survival benefit.26
Both enzalutamide and abiraterone (abiraterone given with physiologic-dose steroid replacement) confer a survival benefit in chemotherapy-naive patients with M1 CRPC. Per the PREVAIL study, enzalutamide (when compared to placebo) offers a median improvement in overall survival (OS) by about 2 months and in radiographic PFS by about 14.6 months.24 Abiraterone blocks the synthesis of androgens via inhibition of CYP17 in the testes and adrenal glands. Abiraterone also confers an overall survival advantage for patients with M1 CRPC who are chemotherapy-naïve, with an estimated 25% decrease in the risk of death (hazard ratio, 0.75, P = 0.009) when compared to prednisone.27
In patients with symptomatic M1 CRPC who have visceral disease or rapidly progressive disease and who are candidates for chemotherapy, docetaxel is frequently used and is given concurrently with steroids. Docetaxel has been given for up to 10 cycles in clinical trials (assuming no progression of disease or dose-limiting toxicities were observed), and at least 6 cycles of treatment are recommended. When compared to mitoxantrone plus prednisone in the TAX 327 phase 3 trial, docetaxel plus prednisone offered a significant OS benefit of about 3 months (19.2 months versus 16.3 months).28 For patients who are not candidates for docetaxel (eg, due to preexisting peripheral neuropathy), cabazitaxel should be considered. OS is similar for mCRPC with docetaxel versus cabazitaxel when given in the first-line setting.29 Additionally, cabazitaxel dosed at 20 mg/m2 is noninferior to cabazitaxel dosed at 25 mg/m2, and the lower dose is associated with lower rates of peripheral neuropathy.30 Cabazitaxel should also be considered for mCRPC that has progressed following treatment with docetaxel, with improved OS and PFS when compared to treatment with mitoxantrone and prednisone in this setting, as shown in the TROPIC study.31 Mitoxantrone given with prednisone has been shown to improve quality of life, but it is associated with significant cardiac toxicity. Additionally, mitoxantrone does not improve disease-free survival or OS in chemotherapy-naive patients32 or in patients who have progressed on docetaxel, and therefore should not be given to patients prior to a taxane chemotherapy unless the patient absolutely cannot tolerate docetaxel or cabazitaxel.
Once a patient’s prostate cancer progresses following treatment with a taxane, the sequence in which to administer subsequent therapies should involve careful consideration of previous treatments and duration of response to each of these treatments. Both enzalutamide and abiraterone are FDA-approved for use following treatment with chemotherapy. Per the AFFIRM trial, heavily pre-treated patients (including those who have received docetaxel) have a median 5-month OS benefit with enzalutamide compared to placebo.33 Another study of M1 CRPC patients who had previously received docetaxel demonstrated an OS benefit with abiraterone (versus placebo),34 but this regimen has limited benefit in patients who have previously received both docetaxel and enzalutamide.35 A rechallenge with docetaxel therapy also can be considered if the patient’s disease responded to docetaxel in the metastatic hormone-sensitive setting.
If the patient’s metastases are limited to bone (ie, no visceral disease), then radiotherapy with radium-223 should be considered. Radium-223 is an alpha-emitting calcium-mimetic radioactive compound that tracks to bone to delay the onset of symptoms from bone metastases.36 Radium-223 also confers a median OS benefit of about 3 months.37 However, this treatment is often limited by preexisting cytopenias.
Diethylstilbestrol (1 mg daily) competes with androgens for AR binding and is also cytotoxic to androgen-sensitive and insensitive prostate cancer cells. While its efficacy is similar to bicalutamide in terms of PSA response rate and median response duration, diethylstilbestrol is associated with significantly more cardiovascular toxicity, including stroke, pulmonary embolism, and heart failure, and its use is therefore limited.38 The glucocorticoids—prednisone (5 mg orally twice daily), dexamethasone (0.5 mg daily), and hydrocortisone (40 mg daily)—inhibit pituitary synthesis of adrenocorticotropic hormone, resulting in decreased adrenal androgen synthesis. Data suggest that among the glucocorticoids, dexamethasone monotherapy may produce superior response rates compared to prednisone monotherapy.39 While the glucocorticoids do produce a PSA response, prolong time to disease progression, and can provide symptomatic relief (eg, from bone pain), they have not been shown to confer a survival benefit and therefore are not commonly used as monotherapy.
Future of CRPC Treatment
Patients with CRPC should be considered for clinical trials when available. These patients’ tumors should be assessed with next-generation sequencing for analysis of microsatellite instability (MSI) or mismatch repair (MMR) as well as the presence of other potentially targetable mutations, as this information may bring into consideration additional investigational as well as FDA-approved treatment options. As of May 2017, immunotherapy with pembrolizumab is approved for patients whose prostate cancer is deficient in MMR or has a high MSI burden based on a study of 12 solid tumor types (including prostate cancer) with deficient MMR.40 Additionally, for patients whose tumor has ≥ 1% programmed death ligand 1 (PD-L1) expression, pembrolizumab has a 17% overall response rate and confers stability of disease in 35%, with a median response duration of 13.5 months.41 Cabozantinib is a mesenchymal epithelial transition (MET) kinase and vascular endothelial growth factor receptor (VEGF-R) inhibitor. When used in heavily pre-treated patients with mCRPC, it showed a radiographic PFS benefit but no survival benefit over prednisone monotherapy.42 One study showed that for patients whose mCRPC had a homozygous deletion and/or a deleterious mutation in the homologous recombination repair genes BRCA1/2, ATM, and CHEK2 or the Fanconi anemia genes, the response rate to the poly ADP ribose polymerase (PARP) inhibitor olaparib was 88%, with a 100% response rate in those with BRCA2 mutations.43 Furthermore, mutations in these DNA repair genes predict increased sensitivity to platinum-based chemotherapy.
Supportive Care
Zoledronic acid or denosumab are FDA approved for men with CRPC and bone metastasis based on the ability of these agents to delay skeletal-related events, including pathologic fracture and spinal cord compression.46 Bisphosphonates, however, do not decrease the incidence of bone metastases.47 And while denosumab does delay the time to first bone metastasis in nmCRPC (particularly in patients with a PSADT of ≤ 6 months), it does not improve OS.48 Other supportive measures include exercise and nutrition. Moderate aerobic exercise for 150 minutes in addition to 2 or 3 strength training sessions per week is recommended by the American College of Sport Medicine to combat cancer-related fatigue.49 There are currently no dietary changes that are routinely recommended to improve the outcome of prostate cancer, but a study noted a shorter biochemical failure–free survival in men with prostate cancer who were obese and consumed a diet high in saturated fat.50
Conclusion
Prostate cancer affects more men in the United States than any other cancer. Once a patient is started on hormone therapy, in all likelihood their prostate cancer will become castration-resistant. Once prostate cancer has developed hormone resistance, there are a host of further treatment options available, including further hormone therapy, chemotherapy, immunotherapy, radiation therapy, bone-targeting agents, and clinical trials. Determining the appropriate sequence in which to use these therapies requires knowledge of the natural history of CRPC, the indications for changing therapies, the mechanism of action and adverse event profile of each treatment, and the optimal time to enroll in a clinical trial.
Prostate cancer is the most common malignancy in men, with an estimated 165,000 new prostate cancer diagnoses and 29,000 prostate cancer deaths occurring in the United States in 2018.1 Due to the widespread use of screening prostate-specific antigen (PSA), prostate cancer has been mainly diagnosed when the tumor is confined to the prostate. Despite definitive treatment of localized prostate cancer, some men develop systemic disease, either biochemical failure, as defined by rising PSA level, or metastatic disease.1 Several factors have been demonstrated to predict risk of relapse, including higher pretreatment PSA, higher Gleason score, and a greater anatomic extent of disease.2 In addition, the incidence of de novo metastatic prostate cancer was recently noted to be increasing. This may be due to changes in the United States Preventive Services Task Force prostate cancer screening guidelines in 2012, which recommended against screening for prostate cancer in men of any age. The updated 2018 guidelines recommend a discussion of the risks versus benefits of screening for prostate cancer for all men aged 55 to 69 years,recommend against screening for men older than 70 years, and do not have recommendations for high-risk subgroups.3
Androgen deprivation therapy (ADT) has been the cornerstone of therapy since 1941 for men with hormone-sensitive systemic disease, both in biochemically relapsed and metastatic disease.4,5 While more than 90% of patients respond to initial ADT, castration resistance is inevitable in some men.6,7 Prostate cancer will become castration-resistant typically after 18 to 24 months of ADT, with the majority of patients developing castration-resistant prostate cancer (CRPC) within 5 years of initiation of ADT.8
Pathogenesis
CRPC (previously called androgen independent prostate cancer) is defined as progression of disease despite serum total testosterone levels less than 50 ng/dL. CRPC is characterized by a rising PSA level and/or radiographic progression. One mechanism of castration resistance is genetic modification of the androgen receptor (AR), including increased expression of the wild-type AR.9 Alternatively, mutations of the steroid-binding domain may play a role in the development of castration resistance by allowing the AR to become activated by non-androgen steroid hormones or even paradoxically by antiandrogens. Studies suggest, however, that AR mutations may be seen in only 10% of prostate cancers that have developed castration resistance.10 The AR-V7 splice variant of the AR lacks an androgen binding site altogether, and may play an important role in castration resistance. In one study, the presence of this splice variant in circulating prostate cancer tumor cells predicted resistance to enzalutamide and abiraterone as well as poor outcomes.11 Intratumoral androgen synthesis also may play a role in the development of CRPC.12,13
CRPC can be broadly categorized into 2 categories, metastatic (mCRPC) and nonmetastatic (nmCRPC; Figure). The exact proportion of patients entering CRPC at a nonmetastatic stage (M0) is largely unknown.14 In one study of patients at the time of diagnosis of CRPC, ≥ 84% of patients were shown to have metastases.8 In this article, we review key aspects of management of CRPC, including selection of first- and second-line therapy, and briefly discuss upcoming clinical trials.

Treatment of Nonmetastatic CRPC (M0 Disease)
Early identification of M0 CRPC is important because patients with nonmetastatic CRPC are at risk for metastasis, as demonstrated by Smith and colleagues.15 In this study that evaluated data from patients with nmCRPC in the placebo group (n = 331) of a randomized controlled trial, at 2 years 46% had developed ≥ 1 bone metastasis, 20% had died, and the median bone metastasis-free survival (MFS) was 25 months.15
Rapid PSA doubling time (PSADT) is linked to shorter time to metastasis in this group of patients. Patients with a PSADT of < 10 months had a risk for bone metastasis 12 times greater and a risk for death 4 times greater than patients with a PSADT of ≥ 10 months.16 Accordingly, observation should be reserved for those patients with a PSADT of ≥ 10 months.
Options for secondary hormonal therapy in those with a PSADT of ≤ 10 months include a first-generation antiandrogen (bicalutamide, flutamide, nilutamide), ketoconazole with hydrocortisone, and more recently second-generation antiandrogens (apalutamide or enzalutamide).
Bicalutamide competitively inhibits dihydrotestosterone and testosterone binding to the AR and is generally well-tolerated; it is given in conjunction with a GnRH agonist/castration.17 The use of other first-generation antiandrogens is limited mainly due to their toxicity profile. When compared to flutamide in a randomized, double-blinded control study, bicalutamide had significantly improved time to treatment failure.18 Due to promiscuous binding to AR, withdrawal of first-generation antiandrogen therapy has been associated with a biochemical response in a small proportion of patients, with response typically seen after 5 to 7 half-lives of the drug have elapsed.19
Although traditionally used as an antifungal agent, ketoconazole also inhibits androgen synthesis in the adrenal glands and acts as a direct cytotoxin to cancer cells.20 Ketoconazole (with hydrocortisone) has been considered as a treatment option, typically at the time of antiandrogen withdrawal. However, ketoconazole offers no survival benefit, and with the approval of abiraterone in M1 CRPC, its use has declined significantly.21 Additionally, ketoconazole poses a risk for severe hepatotoxicity and QT prolongation, and has significant interactions with numerous drugs, thereby limiting its use. Given the typically short duration of response to first-generation antiandrogens, the second-generation antiandrogens were developed and are associated with a significantly greater progression-free survival (PFS) in M0 CRPC.22,23
The second-generation antiandrogens enzalutamide and apalutamide not only competitively bind to the AR, inhibiting formation of the androgen/AR complex, but they also inhibit androgen/AR complex nuclear translocation and binding to nuclear DNA. In the PROSPER trial, enzalutamide significantly increased radiographic PFS and improved quality of life compared to placebo in chemotherapy-naive patients (Table 1).24 Apalutamide significantly increased MFS as well as PFS and time to PSA progression compared to placebo in the phase 3 SPARTAN trial.25 Apalutamide is generally well tolerated, with hypertension and rash being the most common severe adverse effects. Apalutamide also has less potential for central nervous system toxicities than enzalutamide. The recent approval of these agents is likely to change responses to subsequent treatments, especially in the metastatic setting.

Treatment of Metastatic CRPC (M1 Disease)
As with M0 CRPC, ADT should be continued in patients with mCRPC to maintain castration levels of testosterone while initiating additional treatments. Several drugs for the treatment of mCRPC have been approved by the US Food and Drug Administration (FDA) since 2010, including abiraterone with prednisone (or methylprednisolone), enzalutamide (but not apalutamide), radium-223, sipuleucel-T, and cabazitaxel (Table 2).

Given the availability of numerous treatment options for men with mCRPC, the sequencing of treatments should be based on careful consideration of the efficacy and adverse effect profiles of each drug as well as the anatomic and molecular characteristics of the cancer, comorbidities, and patient preference. If there is no evidence of visceral disease and the patient has an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1 with an estimated life expectancy of greater than 6 months and is minimally symptomatic, then treatment with either oral targeted agents or immunotherapy with sipuleucel-T is considered appropriate.
Sipuleucel-T is an autologous dendritic cell vaccination designed to enhance T-cell–mediated response to prostatic acid phosphatase (PAP). The treatments are prepared from leukapheresed host mononuclear cells that are then exposed to PAP fused to granulocyte-macrophage colony-stimulating factor. The activated dendritic cells are then infused back into the host once every 2 weeks for a total of 3 treatments. The main side effects of this treatment include chills, fever, and headache, but it is generally well-tolerated and has demonstrated a survival benefit.26
Both enzalutamide and abiraterone (abiraterone given with physiologic-dose steroid replacement) confer a survival benefit in chemotherapy-naive patients with M1 CRPC. Per the PREVAIL study, enzalutamide (when compared to placebo) offers a median improvement in overall survival (OS) by about 2 months and in radiographic PFS by about 14.6 months.24 Abiraterone blocks the synthesis of androgens via inhibition of CYP17 in the testes and adrenal glands. Abiraterone also confers an overall survival advantage for patients with M1 CRPC who are chemotherapy-naïve, with an estimated 25% decrease in the risk of death (hazard ratio, 0.75, P = 0.009) when compared to prednisone.27
In patients with symptomatic M1 CRPC who have visceral disease or rapidly progressive disease and who are candidates for chemotherapy, docetaxel is frequently used and is given concurrently with steroids. Docetaxel has been given for up to 10 cycles in clinical trials (assuming no progression of disease or dose-limiting toxicities were observed), and at least 6 cycles of treatment are recommended. When compared to mitoxantrone plus prednisone in the TAX 327 phase 3 trial, docetaxel plus prednisone offered a significant OS benefit of about 3 months (19.2 months versus 16.3 months).28 For patients who are not candidates for docetaxel (eg, due to preexisting peripheral neuropathy), cabazitaxel should be considered. OS is similar for mCRPC with docetaxel versus cabazitaxel when given in the first-line setting.29 Additionally, cabazitaxel dosed at 20 mg/m2 is noninferior to cabazitaxel dosed at 25 mg/m2, and the lower dose is associated with lower rates of peripheral neuropathy.30 Cabazitaxel should also be considered for mCRPC that has progressed following treatment with docetaxel, with improved OS and PFS when compared to treatment with mitoxantrone and prednisone in this setting, as shown in the TROPIC study.31 Mitoxantrone given with prednisone has been shown to improve quality of life, but it is associated with significant cardiac toxicity. Additionally, mitoxantrone does not improve disease-free survival or OS in chemotherapy-naive patients32 or in patients who have progressed on docetaxel, and therefore should not be given to patients prior to a taxane chemotherapy unless the patient absolutely cannot tolerate docetaxel or cabazitaxel.
Once a patient’s prostate cancer progresses following treatment with a taxane, the sequence in which to administer subsequent therapies should involve careful consideration of previous treatments and duration of response to each of these treatments. Both enzalutamide and abiraterone are FDA-approved for use following treatment with chemotherapy. Per the AFFIRM trial, heavily pre-treated patients (including those who have received docetaxel) have a median 5-month OS benefit with enzalutamide compared to placebo.33 Another study of M1 CRPC patients who had previously received docetaxel demonstrated an OS benefit with abiraterone (versus placebo),34 but this regimen has limited benefit in patients who have previously received both docetaxel and enzalutamide.35 A rechallenge with docetaxel therapy also can be considered if the patient’s disease responded to docetaxel in the metastatic hormone-sensitive setting.
If the patient’s metastases are limited to bone (ie, no visceral disease), then radiotherapy with radium-223 should be considered. Radium-223 is an alpha-emitting calcium-mimetic radioactive compound that tracks to bone to delay the onset of symptoms from bone metastases.36 Radium-223 also confers a median OS benefit of about 3 months.37 However, this treatment is often limited by preexisting cytopenias.
Diethylstilbestrol (1 mg daily) competes with androgens for AR binding and is also cytotoxic to androgen-sensitive and insensitive prostate cancer cells. While its efficacy is similar to bicalutamide in terms of PSA response rate and median response duration, diethylstilbestrol is associated with significantly more cardiovascular toxicity, including stroke, pulmonary embolism, and heart failure, and its use is therefore limited.38 The glucocorticoids—prednisone (5 mg orally twice daily), dexamethasone (0.5 mg daily), and hydrocortisone (40 mg daily)—inhibit pituitary synthesis of adrenocorticotropic hormone, resulting in decreased adrenal androgen synthesis. Data suggest that among the glucocorticoids, dexamethasone monotherapy may produce superior response rates compared to prednisone monotherapy.39 While the glucocorticoids do produce a PSA response, prolong time to disease progression, and can provide symptomatic relief (eg, from bone pain), they have not been shown to confer a survival benefit and therefore are not commonly used as monotherapy.
Future of CRPC Treatment
Patients with CRPC should be considered for clinical trials when available. These patients’ tumors should be assessed with next-generation sequencing for analysis of microsatellite instability (MSI) or mismatch repair (MMR) as well as the presence of other potentially targetable mutations, as this information may bring into consideration additional investigational as well as FDA-approved treatment options. As of May 2017, immunotherapy with pembrolizumab is approved for patients whose prostate cancer is deficient in MMR or has a high MSI burden based on a study of 12 solid tumor types (including prostate cancer) with deficient MMR.40 Additionally, for patients whose tumor has ≥ 1% programmed death ligand 1 (PD-L1) expression, pembrolizumab has a 17% overall response rate and confers stability of disease in 35%, with a median response duration of 13.5 months.41 Cabozantinib is a mesenchymal epithelial transition (MET) kinase and vascular endothelial growth factor receptor (VEGF-R) inhibitor. When used in heavily pre-treated patients with mCRPC, it showed a radiographic PFS benefit but no survival benefit over prednisone monotherapy.42 One study showed that for patients whose mCRPC had a homozygous deletion and/or a deleterious mutation in the homologous recombination repair genes BRCA1/2, ATM, and CHEK2 or the Fanconi anemia genes, the response rate to the poly ADP ribose polymerase (PARP) inhibitor olaparib was 88%, with a 100% response rate in those with BRCA2 mutations.43 Furthermore, mutations in these DNA repair genes predict increased sensitivity to platinum-based chemotherapy.
Supportive Care
Zoledronic acid or denosumab are FDA approved for men with CRPC and bone metastasis based on the ability of these agents to delay skeletal-related events, including pathologic fracture and spinal cord compression.46 Bisphosphonates, however, do not decrease the incidence of bone metastases.47 And while denosumab does delay the time to first bone metastasis in nmCRPC (particularly in patients with a PSADT of ≤ 6 months), it does not improve OS.48 Other supportive measures include exercise and nutrition. Moderate aerobic exercise for 150 minutes in addition to 2 or 3 strength training sessions per week is recommended by the American College of Sport Medicine to combat cancer-related fatigue.49 There are currently no dietary changes that are routinely recommended to improve the outcome of prostate cancer, but a study noted a shorter biochemical failure–free survival in men with prostate cancer who were obese and consumed a diet high in saturated fat.50
Conclusion
Prostate cancer affects more men in the United States than any other cancer. Once a patient is started on hormone therapy, in all likelihood their prostate cancer will become castration-resistant. Once prostate cancer has developed hormone resistance, there are a host of further treatment options available, including further hormone therapy, chemotherapy, immunotherapy, radiation therapy, bone-targeting agents, and clinical trials. Determining the appropriate sequence in which to use these therapies requires knowledge of the natural history of CRPC, the indications for changing therapies, the mechanism of action and adverse event profile of each treatment, and the optimal time to enroll in a clinical trial.
1. Pound CR, Partin AW, Epstein JI, Walsh PC. Prostate-specific antigen after anatomic radical retropubic prostatectomy. Patterns of recurrence and cancer control. Urol Clin North Am. 1997;24:395-406.
2. Caire AA, Sun L, Ode O, et al. Delayed prostate-specific antigen recurrence after radical prostatectomy: how to identify and what are their clinical outcomes? Urology. 2009;74:643-647.
3. US Preventive Services Task Force, Grossman DC, Curry SJ, Owens DK, et al. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:1901-1913.
4. Huggins C, Hodges CV. Studies on prostatic cancer. I: The effects of castration, of estrogen, and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res. 1941;1:293-297.
5. Huggins C, Stevens RE Jr, Hodges CV. Studies on prostatic cancer. II: The effects of castration on advanced carcinoma of the prostate gland. Arch Surg. 1941;43:209-223.
6. Pomerantz M, Kantoff P. Clinical progression to castration recurrent prostate cancer. In: Tindall DJ, James M, eds. Androgen Action in Prostate Cancer. New York: Springer; 2009:57-72.
7. Kirby M, Hirst C, Crawford ED. Characterising the castration-resistant prostate cancer population: a systematic review. Int J Clin Pract. 2011;65:1180-1192.
8. Sharifi N, Dahut WL, Steinberg SM, et al. A retrospective study of the time to clinical endpoints for advanced prostate cancer. BJU Int. 2005;96:985-989.
9. Chen CD, Welsbie DS, Tran C, et al. Molecular determinants of resistance to antiandrogen therapy. Nat Med. 2004;10:33-39.
10. Taplin ME, Rajeshkumar B, Halabi S, et al. Androgen receptor mutations in androgen-independent prostate cancer: Cancer and Leukemia Group B Study 9663. J Clin Oncol. 2003;21:2673-2678.
11. Antonarakis ES, Lu C, Luber B, et al. Clinical significance of androgen receptor splice variant-7 (AR-V7) mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line abiraterone and enzalutamide. J Clin Oncol. 2017;35:2149-2156.
12. Kahn B, Collazo J, Kyprianou N. Androgen receptor as a driver of therapeutic resistance in advanced prostate cancer. Int J Biol Sci. 2014;10:588-595.
13. Logothetis CJ, Gallick GE, Maity SN, et al. Molecular classification of prostate cancer progression: foundation for marker-driven treatment of prostate cancer. Cancer Discov. 2013;3:849-861
14. Tombal B. Non-metastatic CRPC and asymptomatic metastatic CRPC: which treatment for which patient? Ann Oncol. 2012;23(suppl 10):251-258
15. Smith MR, Cook R, Lee KA, et al. Disease and host characteristics as predictors of time to first bone metastasis and death in men with progressive castration-resistant non-metastatic prostate cancer. Cancer. 2011;117:2077-2085.
16. Metwalli AR, Rosner IL, Cullen J, et al. Elevated alkaline phosphatase velocity strongly predicts overall survival and the risk of bone metastases in castrate-resistant prostate cancer. Urol Oncol. 2014;32:761-768
17. Akaza H, Yamaguchi A, Matsuda T, et al. Superior anti-tumor efficacy of bicalutamide 80mg in combination with luteinizing hormone-releasing hormone (LHRH) against versus LHRH agonist monotherapy as first line treatment for advanced prostate cancer: Interim results of a randomized study in Japanese patients. J Clin Oncol. 2004;34:20-28
18. Schellhammer P, Patterson AL, Sharifi R, et al. A controlled trial of bicalutamide versus flutamide, each in combination with luteinizing hormone-releasing hormone analogue therapy, in patients with advanced prostate cancer. Urology. 1995;45(5):745-752.
19. Sartor AO, Tangen CM, Hussain MH, et al. Antiandrogen withdrawal in castrate-refractory prostate cancer: a Southwest Oncology Group Trial (SWOG 9426). Cancer. 2008;112:2393-2400.
20. Eichenberger T, Trachtenberg J, Toor P, et al. Ketoconazole: a possible direct cytotoxic effect on prostate carcinoma cells. J Urol. 1989;141:190-191.
21. Small EJ, Halabi S, Dawson NA, et al. Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583). J Clin Oncol. 2004;22:1025-1033.
22. Penson DF, Armstrong AJ, Concepcion R, et al. Enzalutamide versus bicalutamide in castration resistant prostate cancer: the STRIVE trial. J Clin Oncol. 2016;34:2098-2106.
23. Shore ND, Chowdhury S, Villers A, et al. Efficacy and safety of enzalutamide versus bicalutamide for patients with metastatic prostate cancer (TERRAIN): a randomized, double-blind, phase 2 study. Lancet Oncol. 2016;199:147-154.
24. Beer TM, Armstrong AJ, Rathkopf D, et al. Enzalutamide in men with chemotherapy-naïve castration-resistant prostate cancer: extended analysis of the phase 3 PREVAIL study. Eur Urol. 2017;71:151-154.
25. Smith MR, Saad F, Chowdhury S; SPARTAN Investigators, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408-1418.
26. Kantoff PW, Higano CS, Shore ND, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411-422.
27. Ryan CJ, Smith MR, de Bono JS, et al. Randomized phase 3 trial of abiraterone acetate in men with metastatic castration-resistant prostate cancer and no prior chemotherapy. N Engl J Med. 2013;368:138-148
28. Berthold DR, Pond GR, Soban F, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: updated survival in the TAX 327 study. J Clin Oncol. 2008;26:242-245.
29. Oudard S, Fizazi K, Sengelov L, et al. Cabazitaxel versus docetaxel as first-line therapy for patients with metastatic castration-resistant prostate cancer: a randomized phase III trial—FIRSTANA. J Clin Oncol. 2017;35:3189-3197.
30. Eisenberger M, Hardy-Bessard A-C, Kim CS, et al. Phase III study comparing a reduced dose of cabazitaxel (20 mg/m2) and the currently approved dose (25 mg/m2) in postdocetaxel patients with metastatic castration-resistant prostate cancer—PROSELICA. J Clin Oncol. 2017;35:3198-3206.
31. de Bono JS, Oudard S, Ozguroglu M, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376(9747):P1147-1154.
32. Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004;351:1502-1512.
33. Fizazi K, Scher HI, Miller K, et al. Effect of enzalutamide on time to first skeletal-related event, pain, and quality of life in men with castration-resistant prostate cancer: results from the randomised, phase 3 AFFIRM trial. Lancet Oncol. 2014;15:1147-1156.
34. de Bono JS, Logothetis CJ, Molina A, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995-2005.
35. Loriot Y, Bianchini D, Ileana E, et al. Antitumor activity of abiraterone acetate against metastatic castration-resistant prostate cancer progressing after docetaxel and enzalutamide. Ann Oncol. 2013;24:1807-1812.
36. Sartor O, Coleman R, Nilsson S, et al. Effect of radium-223 dichloride on symptomatic skeletal events in patients with castration-resistant prostate cancer and bone metastases: results from a phase 3, double blind, randomized trial. Lancet Oncol. 2014;15:738-746.
37. Parker C, Nilsson S, Heinrich D, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013; 369:213-223.
38. Turo R, Smolski M, Esler R, et al. Diethylstilboestrol for the treatment of prostate cancer: past, present and future. Scand J Urol. 2014;48:4-14.
39. Venkitaraman R, Lorente D, Murthy V. A randomized phase 2 trial of dexamethasone versus prednisolone in castration-resistant prostate cancer. Eur Urol. 2015 67:673-679.
40. Le DT, Durham JN, Smith KN, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357(6349):409-413.
41. Hansen AR, Massard C, Ott PA, et al. Pembrolizumab for advanced prostate adenocarcinoma: findings of the KEYNOTE-028 study. Ann Oncol. 2018;29:1807-1813.
42. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
43. Mateo J, Carreira S, Sandhu S, et al. DNA-Repair defects and olaparib in metastatic prostate cancer. N Engl J Med. 2015;373:1697-1708.
44. Kentepozidis N, Soultati A, Giassas S, et al. Paclitaxel in combination with carboplatin as salvage treatment in patients with castration-resistant prostate cancer: a Hellenic oncology research group multicenter phase II study. Cancer Chemother Pharmacol. 2012;70:161-168.
45. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
46. Saad F, Gleason DM, Murray R, et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst. 2002;94:1458-1468.
47. Wirth M, Tammela T, Cicalese V, et al. Prevention of bone metastases in patients with high-risk nonmetastatic prostate cancer treated with zoledronic acid: efficacy and safety results of the Zometa European Study (ZEUS). Eur Urol. 2015;67:482-491.
48. Smith MR, Saad F, Coleman R, et al. Denosumab and bone-metastases-free survival in men with castration-resistant prostate cancer: results of a phase 3, randomized, placebo-controlled trial. Lancet. 2012;379(9810):39-46.
49. Schmitz KH, Courneya KS, Matthews C, et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42:1409-1426.
50. Strom SS, Yamamura Y, Flores-Sandoval FN, et al. Prostate cancer in Mexican-Americans: identification of risk factors. Prostate. 2008;68:563-570.
1. Pound CR, Partin AW, Epstein JI, Walsh PC. Prostate-specific antigen after anatomic radical retropubic prostatectomy. Patterns of recurrence and cancer control. Urol Clin North Am. 1997;24:395-406.
2. Caire AA, Sun L, Ode O, et al. Delayed prostate-specific antigen recurrence after radical prostatectomy: how to identify and what are their clinical outcomes? Urology. 2009;74:643-647.
3. US Preventive Services Task Force, Grossman DC, Curry SJ, Owens DK, et al. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:1901-1913.
4. Huggins C, Hodges CV. Studies on prostatic cancer. I: The effects of castration, of estrogen, and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res. 1941;1:293-297.
5. Huggins C, Stevens RE Jr, Hodges CV. Studies on prostatic cancer. II: The effects of castration on advanced carcinoma of the prostate gland. Arch Surg. 1941;43:209-223.
6. Pomerantz M, Kantoff P. Clinical progression to castration recurrent prostate cancer. In: Tindall DJ, James M, eds. Androgen Action in Prostate Cancer. New York: Springer; 2009:57-72.
7. Kirby M, Hirst C, Crawford ED. Characterising the castration-resistant prostate cancer population: a systematic review. Int J Clin Pract. 2011;65:1180-1192.
8. Sharifi N, Dahut WL, Steinberg SM, et al. A retrospective study of the time to clinical endpoints for advanced prostate cancer. BJU Int. 2005;96:985-989.
9. Chen CD, Welsbie DS, Tran C, et al. Molecular determinants of resistance to antiandrogen therapy. Nat Med. 2004;10:33-39.
10. Taplin ME, Rajeshkumar B, Halabi S, et al. Androgen receptor mutations in androgen-independent prostate cancer: Cancer and Leukemia Group B Study 9663. J Clin Oncol. 2003;21:2673-2678.
11. Antonarakis ES, Lu C, Luber B, et al. Clinical significance of androgen receptor splice variant-7 (AR-V7) mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line abiraterone and enzalutamide. J Clin Oncol. 2017;35:2149-2156.
12. Kahn B, Collazo J, Kyprianou N. Androgen receptor as a driver of therapeutic resistance in advanced prostate cancer. Int J Biol Sci. 2014;10:588-595.
13. Logothetis CJ, Gallick GE, Maity SN, et al. Molecular classification of prostate cancer progression: foundation for marker-driven treatment of prostate cancer. Cancer Discov. 2013;3:849-861
14. Tombal B. Non-metastatic CRPC and asymptomatic metastatic CRPC: which treatment for which patient? Ann Oncol. 2012;23(suppl 10):251-258
15. Smith MR, Cook R, Lee KA, et al. Disease and host characteristics as predictors of time to first bone metastasis and death in men with progressive castration-resistant non-metastatic prostate cancer. Cancer. 2011;117:2077-2085.
16. Metwalli AR, Rosner IL, Cullen J, et al. Elevated alkaline phosphatase velocity strongly predicts overall survival and the risk of bone metastases in castrate-resistant prostate cancer. Urol Oncol. 2014;32:761-768
17. Akaza H, Yamaguchi A, Matsuda T, et al. Superior anti-tumor efficacy of bicalutamide 80mg in combination with luteinizing hormone-releasing hormone (LHRH) against versus LHRH agonist monotherapy as first line treatment for advanced prostate cancer: Interim results of a randomized study in Japanese patients. J Clin Oncol. 2004;34:20-28
18. Schellhammer P, Patterson AL, Sharifi R, et al. A controlled trial of bicalutamide versus flutamide, each in combination with luteinizing hormone-releasing hormone analogue therapy, in patients with advanced prostate cancer. Urology. 1995;45(5):745-752.
19. Sartor AO, Tangen CM, Hussain MH, et al. Antiandrogen withdrawal in castrate-refractory prostate cancer: a Southwest Oncology Group Trial (SWOG 9426). Cancer. 2008;112:2393-2400.
20. Eichenberger T, Trachtenberg J, Toor P, et al. Ketoconazole: a possible direct cytotoxic effect on prostate carcinoma cells. J Urol. 1989;141:190-191.
21. Small EJ, Halabi S, Dawson NA, et al. Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583). J Clin Oncol. 2004;22:1025-1033.
22. Penson DF, Armstrong AJ, Concepcion R, et al. Enzalutamide versus bicalutamide in castration resistant prostate cancer: the STRIVE trial. J Clin Oncol. 2016;34:2098-2106.
23. Shore ND, Chowdhury S, Villers A, et al. Efficacy and safety of enzalutamide versus bicalutamide for patients with metastatic prostate cancer (TERRAIN): a randomized, double-blind, phase 2 study. Lancet Oncol. 2016;199:147-154.
24. Beer TM, Armstrong AJ, Rathkopf D, et al. Enzalutamide in men with chemotherapy-naïve castration-resistant prostate cancer: extended analysis of the phase 3 PREVAIL study. Eur Urol. 2017;71:151-154.
25. Smith MR, Saad F, Chowdhury S; SPARTAN Investigators, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408-1418.
26. Kantoff PW, Higano CS, Shore ND, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411-422.
27. Ryan CJ, Smith MR, de Bono JS, et al. Randomized phase 3 trial of abiraterone acetate in men with metastatic castration-resistant prostate cancer and no prior chemotherapy. N Engl J Med. 2013;368:138-148
28. Berthold DR, Pond GR, Soban F, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: updated survival in the TAX 327 study. J Clin Oncol. 2008;26:242-245.
29. Oudard S, Fizazi K, Sengelov L, et al. Cabazitaxel versus docetaxel as first-line therapy for patients with metastatic castration-resistant prostate cancer: a randomized phase III trial—FIRSTANA. J Clin Oncol. 2017;35:3189-3197.
30. Eisenberger M, Hardy-Bessard A-C, Kim CS, et al. Phase III study comparing a reduced dose of cabazitaxel (20 mg/m2) and the currently approved dose (25 mg/m2) in postdocetaxel patients with metastatic castration-resistant prostate cancer—PROSELICA. J Clin Oncol. 2017;35:3198-3206.
31. de Bono JS, Oudard S, Ozguroglu M, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376(9747):P1147-1154.
32. Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004;351:1502-1512.
33. Fizazi K, Scher HI, Miller K, et al. Effect of enzalutamide on time to first skeletal-related event, pain, and quality of life in men with castration-resistant prostate cancer: results from the randomised, phase 3 AFFIRM trial. Lancet Oncol. 2014;15:1147-1156.
34. de Bono JS, Logothetis CJ, Molina A, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995-2005.
35. Loriot Y, Bianchini D, Ileana E, et al. Antitumor activity of abiraterone acetate against metastatic castration-resistant prostate cancer progressing after docetaxel and enzalutamide. Ann Oncol. 2013;24:1807-1812.
36. Sartor O, Coleman R, Nilsson S, et al. Effect of radium-223 dichloride on symptomatic skeletal events in patients with castration-resistant prostate cancer and bone metastases: results from a phase 3, double blind, randomized trial. Lancet Oncol. 2014;15:738-746.
37. Parker C, Nilsson S, Heinrich D, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013; 369:213-223.
38. Turo R, Smolski M, Esler R, et al. Diethylstilboestrol for the treatment of prostate cancer: past, present and future. Scand J Urol. 2014;48:4-14.
39. Venkitaraman R, Lorente D, Murthy V. A randomized phase 2 trial of dexamethasone versus prednisolone in castration-resistant prostate cancer. Eur Urol. 2015 67:673-679.
40. Le DT, Durham JN, Smith KN, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357(6349):409-413.
41. Hansen AR, Massard C, Ott PA, et al. Pembrolizumab for advanced prostate adenocarcinoma: findings of the KEYNOTE-028 study. Ann Oncol. 2018;29:1807-1813.
42. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
43. Mateo J, Carreira S, Sandhu S, et al. DNA-Repair defects and olaparib in metastatic prostate cancer. N Engl J Med. 2015;373:1697-1708.
44. Kentepozidis N, Soultati A, Giassas S, et al. Paclitaxel in combination with carboplatin as salvage treatment in patients with castration-resistant prostate cancer: a Hellenic oncology research group multicenter phase II study. Cancer Chemother Pharmacol. 2012;70:161-168.
45. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
46. Saad F, Gleason DM, Murray R, et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst. 2002;94:1458-1468.
47. Wirth M, Tammela T, Cicalese V, et al. Prevention of bone metastases in patients with high-risk nonmetastatic prostate cancer treated with zoledronic acid: efficacy and safety results of the Zometa European Study (ZEUS). Eur Urol. 2015;67:482-491.
48. Smith MR, Saad F, Coleman R, et al. Denosumab and bone-metastases-free survival in men with castration-resistant prostate cancer: results of a phase 3, randomized, placebo-controlled trial. Lancet. 2012;379(9810):39-46.
49. Schmitz KH, Courneya KS, Matthews C, et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42:1409-1426.
50. Strom SS, Yamamura Y, Flores-Sandoval FN, et al. Prostate cancer in Mexican-Americans: identification of risk factors. Prostate. 2008;68:563-570.
Pembrolizumab/lenvatinib active against urothelial carcinoma
SAN FRANCISCO – A combination of a targeted therapy and an immune checkpoint inhibitor showed promising activity against advanced urothelial cancer in early data from a phase 1b/2 study.
In a cohort of 20 patients with urothelial carcinoma who were enrolled in a larger clinical trial testing the combination of the tyrosine kinase inhibitor (TKI) lenvatinib (Lenvima) and the checkpoint inhibitor pembrolizumab (Keytruda) against urinary tract and other solid malignancies, 5 had an objective response to the combination, including one complete and four partial responses, for an objective response rate of 25%, reported Nicholas J. Vogelzang, MD, from Comprehensive Cancers Centers of Nevada in Las Vegas.
“This response rate warrants further investigation. The lenvatinib plus pembrolizumab combination will be studied in a phase 3 trial in urothelial carcinoma,” he said at the American Society of Clinical Oncology (ASCO) - Society for Immunotherapy of Cancer (SITC): Clinical Immuno-Oncology Symposium.
Dr. Vogelzang noted that urothelial carcinomas account for more than 90% of all bladder cancers. Pembrolizumab monotherapy is approved for treatment of patients with urothelial carcinoma who are ineligible for cisplatin and whose tumors have a combined positive score (CPS) for programmed death-ligand 1 (PD-L1) of 10 or greater or who are ineligible for platinum-based chemotherapy regimens and, in the second line, for advanced or metastatic urothelial carcinoma.
Lenvatinib, a multikinase inhibitor, is approved as monotherapy for radioiodine-refractory differentiated thyroid cancer, unresectable hepatocellular carcinoma, and in combination with everolimus for advanced renal cell carcinoma (RCC) after one year of antiangiogenic therapy.
Dr. Vogelzang reported results of the urothelial cancer cohort from a multicohort study testing the combination.
Twenty patients with histologically confirmed metastatic urothelial cancer were enrolled. The patients all had no more than two prior systemic regimens, good performance status, and a life expectancy of at least 12 weeks. The patients received oral lenvatinib 20 mg daily and pembrolizumab 200 mg intravenously every 21 days. The median patient age was 72 years. The cohort included 14 men and six women.
The objective response rate (ORR) at 24 weeks, the primary endpoint, was 25%, comprising one complete and four partial responses. Nine patients had stable disease, two had disease progression, and four were not evaluable for efficacy. The results were identical according to immune-related Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 and modified RECIST version 1.1. Of the 16 evaluable patients, 12 experienced tumor-size reductions from baseline.
“Although there were five objective responses, there were an additional seven patients or more who had minor regressions of disease – clearly an active regimen,” Dr. Vogelzang said. Four of the patients, including one with a PD-L1–positive tumor and three with PD-L1–negative tumors were still alive, with the longest survival past 80 weeks since the start of therapy. The majority of patients, however, had no objective response or disease progression within about 20 weeks.
After a median follow-up of 11.7 months, the median progression-free survival (PFS) was 5.4 months, and the 12-month PFS rate was 26%.
In all, 18 of the 20 patients (90%) experienced a treatment-related adverse event of any grade, 10 had grade 3 or 4 events, and 6 had serious adverse events including one death from gastrointestinal hemorrhage that Dr. Vogelzang said appeared to be related to lenvatinib. A total of four patients (20%) had a treatment-related event leading to withdrawal or discontinuation, seven had a dose reduction, and 12 had an interruption in therapy, primarily of lenvatinib. The most common toxicities were proteinuria, diarrhea, hypertension, fatigue, hypothyroidism, decreased appetite with nausea, pancreatitis with increased lipase, skin rash, vomiting, and dry mouth.
In addition to the planned phase 3 trial of the combination in urothelial carcinoma, lenvatinib/pembrolizumab is also being studied for the treatment of RCC.
The study was supported by Eisai and Merck Sharp & Dohme. Dr. Vogelzang disclosed financial relationships with Caris Life Sciences, Pfizer, Up to Date, AstraZeneca, MedImmune, and other companies. Five coauthors are employees of Merck or Esai.
SOURCE: Vogelzang NJ et al. ASCO-SITC, Abstract 11.
SAN FRANCISCO – A combination of a targeted therapy and an immune checkpoint inhibitor showed promising activity against advanced urothelial cancer in early data from a phase 1b/2 study.
In a cohort of 20 patients with urothelial carcinoma who were enrolled in a larger clinical trial testing the combination of the tyrosine kinase inhibitor (TKI) lenvatinib (Lenvima) and the checkpoint inhibitor pembrolizumab (Keytruda) against urinary tract and other solid malignancies, 5 had an objective response to the combination, including one complete and four partial responses, for an objective response rate of 25%, reported Nicholas J. Vogelzang, MD, from Comprehensive Cancers Centers of Nevada in Las Vegas.
“This response rate warrants further investigation. The lenvatinib plus pembrolizumab combination will be studied in a phase 3 trial in urothelial carcinoma,” he said at the American Society of Clinical Oncology (ASCO) - Society for Immunotherapy of Cancer (SITC): Clinical Immuno-Oncology Symposium.
Dr. Vogelzang noted that urothelial carcinomas account for more than 90% of all bladder cancers. Pembrolizumab monotherapy is approved for treatment of patients with urothelial carcinoma who are ineligible for cisplatin and whose tumors have a combined positive score (CPS) for programmed death-ligand 1 (PD-L1) of 10 or greater or who are ineligible for platinum-based chemotherapy regimens and, in the second line, for advanced or metastatic urothelial carcinoma.
Lenvatinib, a multikinase inhibitor, is approved as monotherapy for radioiodine-refractory differentiated thyroid cancer, unresectable hepatocellular carcinoma, and in combination with everolimus for advanced renal cell carcinoma (RCC) after one year of antiangiogenic therapy.
Dr. Vogelzang reported results of the urothelial cancer cohort from a multicohort study testing the combination.
Twenty patients with histologically confirmed metastatic urothelial cancer were enrolled. The patients all had no more than two prior systemic regimens, good performance status, and a life expectancy of at least 12 weeks. The patients received oral lenvatinib 20 mg daily and pembrolizumab 200 mg intravenously every 21 days. The median patient age was 72 years. The cohort included 14 men and six women.
The objective response rate (ORR) at 24 weeks, the primary endpoint, was 25%, comprising one complete and four partial responses. Nine patients had stable disease, two had disease progression, and four were not evaluable for efficacy. The results were identical according to immune-related Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 and modified RECIST version 1.1. Of the 16 evaluable patients, 12 experienced tumor-size reductions from baseline.
“Although there were five objective responses, there were an additional seven patients or more who had minor regressions of disease – clearly an active regimen,” Dr. Vogelzang said. Four of the patients, including one with a PD-L1–positive tumor and three with PD-L1–negative tumors were still alive, with the longest survival past 80 weeks since the start of therapy. The majority of patients, however, had no objective response or disease progression within about 20 weeks.
After a median follow-up of 11.7 months, the median progression-free survival (PFS) was 5.4 months, and the 12-month PFS rate was 26%.
In all, 18 of the 20 patients (90%) experienced a treatment-related adverse event of any grade, 10 had grade 3 or 4 events, and 6 had serious adverse events including one death from gastrointestinal hemorrhage that Dr. Vogelzang said appeared to be related to lenvatinib. A total of four patients (20%) had a treatment-related event leading to withdrawal or discontinuation, seven had a dose reduction, and 12 had an interruption in therapy, primarily of lenvatinib. The most common toxicities were proteinuria, diarrhea, hypertension, fatigue, hypothyroidism, decreased appetite with nausea, pancreatitis with increased lipase, skin rash, vomiting, and dry mouth.
In addition to the planned phase 3 trial of the combination in urothelial carcinoma, lenvatinib/pembrolizumab is also being studied for the treatment of RCC.
The study was supported by Eisai and Merck Sharp & Dohme. Dr. Vogelzang disclosed financial relationships with Caris Life Sciences, Pfizer, Up to Date, AstraZeneca, MedImmune, and other companies. Five coauthors are employees of Merck or Esai.
SOURCE: Vogelzang NJ et al. ASCO-SITC, Abstract 11.
SAN FRANCISCO – A combination of a targeted therapy and an immune checkpoint inhibitor showed promising activity against advanced urothelial cancer in early data from a phase 1b/2 study.
In a cohort of 20 patients with urothelial carcinoma who were enrolled in a larger clinical trial testing the combination of the tyrosine kinase inhibitor (TKI) lenvatinib (Lenvima) and the checkpoint inhibitor pembrolizumab (Keytruda) against urinary tract and other solid malignancies, 5 had an objective response to the combination, including one complete and four partial responses, for an objective response rate of 25%, reported Nicholas J. Vogelzang, MD, from Comprehensive Cancers Centers of Nevada in Las Vegas.
“This response rate warrants further investigation. The lenvatinib plus pembrolizumab combination will be studied in a phase 3 trial in urothelial carcinoma,” he said at the American Society of Clinical Oncology (ASCO) - Society for Immunotherapy of Cancer (SITC): Clinical Immuno-Oncology Symposium.
Dr. Vogelzang noted that urothelial carcinomas account for more than 90% of all bladder cancers. Pembrolizumab monotherapy is approved for treatment of patients with urothelial carcinoma who are ineligible for cisplatin and whose tumors have a combined positive score (CPS) for programmed death-ligand 1 (PD-L1) of 10 or greater or who are ineligible for platinum-based chemotherapy regimens and, in the second line, for advanced or metastatic urothelial carcinoma.
Lenvatinib, a multikinase inhibitor, is approved as monotherapy for radioiodine-refractory differentiated thyroid cancer, unresectable hepatocellular carcinoma, and in combination with everolimus for advanced renal cell carcinoma (RCC) after one year of antiangiogenic therapy.
Dr. Vogelzang reported results of the urothelial cancer cohort from a multicohort study testing the combination.
Twenty patients with histologically confirmed metastatic urothelial cancer were enrolled. The patients all had no more than two prior systemic regimens, good performance status, and a life expectancy of at least 12 weeks. The patients received oral lenvatinib 20 mg daily and pembrolizumab 200 mg intravenously every 21 days. The median patient age was 72 years. The cohort included 14 men and six women.
The objective response rate (ORR) at 24 weeks, the primary endpoint, was 25%, comprising one complete and four partial responses. Nine patients had stable disease, two had disease progression, and four were not evaluable for efficacy. The results were identical according to immune-related Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 and modified RECIST version 1.1. Of the 16 evaluable patients, 12 experienced tumor-size reductions from baseline.
“Although there were five objective responses, there were an additional seven patients or more who had minor regressions of disease – clearly an active regimen,” Dr. Vogelzang said. Four of the patients, including one with a PD-L1–positive tumor and three with PD-L1–negative tumors were still alive, with the longest survival past 80 weeks since the start of therapy. The majority of patients, however, had no objective response or disease progression within about 20 weeks.
After a median follow-up of 11.7 months, the median progression-free survival (PFS) was 5.4 months, and the 12-month PFS rate was 26%.
In all, 18 of the 20 patients (90%) experienced a treatment-related adverse event of any grade, 10 had grade 3 or 4 events, and 6 had serious adverse events including one death from gastrointestinal hemorrhage that Dr. Vogelzang said appeared to be related to lenvatinib. A total of four patients (20%) had a treatment-related event leading to withdrawal or discontinuation, seven had a dose reduction, and 12 had an interruption in therapy, primarily of lenvatinib. The most common toxicities were proteinuria, diarrhea, hypertension, fatigue, hypothyroidism, decreased appetite with nausea, pancreatitis with increased lipase, skin rash, vomiting, and dry mouth.
In addition to the planned phase 3 trial of the combination in urothelial carcinoma, lenvatinib/pembrolizumab is also being studied for the treatment of RCC.
The study was supported by Eisai and Merck Sharp & Dohme. Dr. Vogelzang disclosed financial relationships with Caris Life Sciences, Pfizer, Up to Date, AstraZeneca, MedImmune, and other companies. Five coauthors are employees of Merck or Esai.
SOURCE: Vogelzang NJ et al. ASCO-SITC, Abstract 11.
REPORTING FROM ASCO-SITC
SEER data: Abiraterone acetate may up mortality risk in prostate cancer patients with CVD
Prostate cancer patients with cardiovascular disease (CVD) who are treated with abiraterone acetate (Zytiga) have an increased risk of death within 6 months of starting therapy, compared with those without preexisting CVD, according to an analysis of Surveillance, Epidemiology, and End Results (SEER)–Medicare linked data.
Of 2,845 patients diagnosed with prostate cancer between 1991 and 2013 and treated with abiraterone acetate (AA) between 2011 and 2014, 1,924 (67.6%) had at least one serious preexisting CVD condition. Mortality within 6 months of treatment initiation in those with preexisting CVD ranged from 21.4% to 25.6%, depending on the type of condition, compared with 15.8% among those with no preexisting CVD, Grace Lu-Yao, PhD, reported during a press conference highlighting data to be presented at the upcoming American Association for Cancer Research annual meeting in Atlanta.
An additional analysis of health care utilization showed that AA treatment was associated with risks for all patients, regardless of CVD; among patients without chemotherapy and without CVD, the hospitalization rate increased by 53%, and in those with preexisting CVD the rate increased from 34% to 55%, depending on the cardiovascular condition, said Dr. Lu-Yao, associate director for population science at the Sidney Kimmel Cancer Center at Jefferson, Philadelphia.
Since patients with preexisting CVD are frequently excluded from clinical trials of AA, its effects in this population are uncertain. However, these data – though limited by the retrospective nature of the study – provide evidence that a significant proportion of patients treated in the real world differ from those in clinical trials, and therefore that the trial findings may not apply to patients who are excluded, said Dr. Lu-Yao, professor and vice chair in the department of medical Oncology at the Sidney Kimmel Medical College.
AA, which was initially approved in 2011 for use in combination with prednisone in patients with metastatic castration-resistant prostate cancer who were previously treated with docetaxel, received additional approval in 2018 for use in combination with prednisone for metastatic high-risk castration-sensitive prostate cancer.
The potential for expanded use of AA further underscores the need for improved understanding of its effects in the real-world setting, she noted.
This study was funded by a Pennsylvania CURE Program award and the National Cancer Institute. Dr. Lu-Yao has no direct conflicts to declare except that her spouse, who has no involvement with this study, is an officer of Sun Pharmaceutical Industries Inc.
SOURCE: Lu-Yao G et al. AACR 2019, Abstract preview.
Prostate cancer patients with cardiovascular disease (CVD) who are treated with abiraterone acetate (Zytiga) have an increased risk of death within 6 months of starting therapy, compared with those without preexisting CVD, according to an analysis of Surveillance, Epidemiology, and End Results (SEER)–Medicare linked data.
Of 2,845 patients diagnosed with prostate cancer between 1991 and 2013 and treated with abiraterone acetate (AA) between 2011 and 2014, 1,924 (67.6%) had at least one serious preexisting CVD condition. Mortality within 6 months of treatment initiation in those with preexisting CVD ranged from 21.4% to 25.6%, depending on the type of condition, compared with 15.8% among those with no preexisting CVD, Grace Lu-Yao, PhD, reported during a press conference highlighting data to be presented at the upcoming American Association for Cancer Research annual meeting in Atlanta.
An additional analysis of health care utilization showed that AA treatment was associated with risks for all patients, regardless of CVD; among patients without chemotherapy and without CVD, the hospitalization rate increased by 53%, and in those with preexisting CVD the rate increased from 34% to 55%, depending on the cardiovascular condition, said Dr. Lu-Yao, associate director for population science at the Sidney Kimmel Cancer Center at Jefferson, Philadelphia.
Since patients with preexisting CVD are frequently excluded from clinical trials of AA, its effects in this population are uncertain. However, these data – though limited by the retrospective nature of the study – provide evidence that a significant proportion of patients treated in the real world differ from those in clinical trials, and therefore that the trial findings may not apply to patients who are excluded, said Dr. Lu-Yao, professor and vice chair in the department of medical Oncology at the Sidney Kimmel Medical College.
AA, which was initially approved in 2011 for use in combination with prednisone in patients with metastatic castration-resistant prostate cancer who were previously treated with docetaxel, received additional approval in 2018 for use in combination with prednisone for metastatic high-risk castration-sensitive prostate cancer.
The potential for expanded use of AA further underscores the need for improved understanding of its effects in the real-world setting, she noted.
This study was funded by a Pennsylvania CURE Program award and the National Cancer Institute. Dr. Lu-Yao has no direct conflicts to declare except that her spouse, who has no involvement with this study, is an officer of Sun Pharmaceutical Industries Inc.
SOURCE: Lu-Yao G et al. AACR 2019, Abstract preview.
Prostate cancer patients with cardiovascular disease (CVD) who are treated with abiraterone acetate (Zytiga) have an increased risk of death within 6 months of starting therapy, compared with those without preexisting CVD, according to an analysis of Surveillance, Epidemiology, and End Results (SEER)–Medicare linked data.
Of 2,845 patients diagnosed with prostate cancer between 1991 and 2013 and treated with abiraterone acetate (AA) between 2011 and 2014, 1,924 (67.6%) had at least one serious preexisting CVD condition. Mortality within 6 months of treatment initiation in those with preexisting CVD ranged from 21.4% to 25.6%, depending on the type of condition, compared with 15.8% among those with no preexisting CVD, Grace Lu-Yao, PhD, reported during a press conference highlighting data to be presented at the upcoming American Association for Cancer Research annual meeting in Atlanta.
An additional analysis of health care utilization showed that AA treatment was associated with risks for all patients, regardless of CVD; among patients without chemotherapy and without CVD, the hospitalization rate increased by 53%, and in those with preexisting CVD the rate increased from 34% to 55%, depending on the cardiovascular condition, said Dr. Lu-Yao, associate director for population science at the Sidney Kimmel Cancer Center at Jefferson, Philadelphia.
Since patients with preexisting CVD are frequently excluded from clinical trials of AA, its effects in this population are uncertain. However, these data – though limited by the retrospective nature of the study – provide evidence that a significant proportion of patients treated in the real world differ from those in clinical trials, and therefore that the trial findings may not apply to patients who are excluded, said Dr. Lu-Yao, professor and vice chair in the department of medical Oncology at the Sidney Kimmel Medical College.
AA, which was initially approved in 2011 for use in combination with prednisone in patients with metastatic castration-resistant prostate cancer who were previously treated with docetaxel, received additional approval in 2018 for use in combination with prednisone for metastatic high-risk castration-sensitive prostate cancer.
The potential for expanded use of AA further underscores the need for improved understanding of its effects in the real-world setting, she noted.
This study was funded by a Pennsylvania CURE Program award and the National Cancer Institute. Dr. Lu-Yao has no direct conflicts to declare except that her spouse, who has no involvement with this study, is an officer of Sun Pharmaceutical Industries Inc.
SOURCE: Lu-Yao G et al. AACR 2019, Abstract preview.
Sperm counts largely stable after adjuvant treatment of clinical stage I testicular cancer
Adjuvant treatments appear to have no significant detrimental long-term effects on sperm count in men being treated for clinical stage I testicular cancer, results of a recent investigation suggest.
Sperm number and concentration were largely stable over time in patients who received a round of chemotherapy or radiation to lymph nodes following orchiectomy, according to results of the 182-patient study.
Investigators said they still offer sperm banking before orchiectomy, since some patients will have low sperm counts prior to orchiectomy that persist after the procedure.
Moreover, the type of testicular cancer and the potential need for other postorchiectomy treatments are often “unknown factors” that underscore the importance of sperm banking, said the researchers, led by Kristina Weibring, MD, of Karolinska University Hospital, Stockholm.
“Assisted reproductive measures may be necessary for these patients regardless of any treatment given,” the researchers noted. The report is in Annals of Oncology.
The lack of effect on sperm counts in this study stands in contrast to previous studies, which clearly show the detrimental effects of multiple chemotherapy cycles on sperm recovery, the investigators said.
Their study comprised 182 patients 18-50 years of age with clinical stage I testicular cancer who underwent unilateral orchiectomy. Depending on tumor characteristics, the patients then received one cycle of adjuvant carboplatin, one cycle of a bleomycin, etoposide, and cisplatin (BEP) regimen, surveillance, or adjuvant radiotherapy to the infradiaphragmal para-aortic and ipsilateral iliac lymph nodes. Sperm samples were obtained at 6, 12, 24, 36, and 60 months after the completion of treatment.
While there was a transient drop in the radiation-treated patients at the 6-month evaluation, mean total sperm number otherwise increased over time in all groups, according to the investigators’ report.
Similarly, mean sperm concentration significantly increased from baseline to 12 months post treatment in the surveillance, BEP, and carboplatin groups, with a nonsignificant decrease in the radiotherapy group, they said in the report.
There were generally no significant differences in sperm count or concentration for the treatments, compared with surveillance, beyond a significant decrease in mean sperm count for radiation versus surveillance, they added.
There were likewise no significant changes in sperm measures for seminoma and nonseminoma patients at any point over the 5 years of evaluation, reported data show.
“With the results of this study, we can now inform our patients that adjuvant chemotherapy does not seem to affect the testicular function,” Dr. Weibring and her colleagues concluded.
The authors reported that they had no conflicts of interest related to the study, which was supported by the Swedish Cancer Society, among other sources.
SOURCE: Weibring K et al. Ann Oncol. 2019 Feb 25. doi: 10.1093/annonc/mdz017/5348526.
Adjuvant treatments appear to have no significant detrimental long-term effects on sperm count in men being treated for clinical stage I testicular cancer, results of a recent investigation suggest.
Sperm number and concentration were largely stable over time in patients who received a round of chemotherapy or radiation to lymph nodes following orchiectomy, according to results of the 182-patient study.
Investigators said they still offer sperm banking before orchiectomy, since some patients will have low sperm counts prior to orchiectomy that persist after the procedure.
Moreover, the type of testicular cancer and the potential need for other postorchiectomy treatments are often “unknown factors” that underscore the importance of sperm banking, said the researchers, led by Kristina Weibring, MD, of Karolinska University Hospital, Stockholm.
“Assisted reproductive measures may be necessary for these patients regardless of any treatment given,” the researchers noted. The report is in Annals of Oncology.
The lack of effect on sperm counts in this study stands in contrast to previous studies, which clearly show the detrimental effects of multiple chemotherapy cycles on sperm recovery, the investigators said.
Their study comprised 182 patients 18-50 years of age with clinical stage I testicular cancer who underwent unilateral orchiectomy. Depending on tumor characteristics, the patients then received one cycle of adjuvant carboplatin, one cycle of a bleomycin, etoposide, and cisplatin (BEP) regimen, surveillance, or adjuvant radiotherapy to the infradiaphragmal para-aortic and ipsilateral iliac lymph nodes. Sperm samples were obtained at 6, 12, 24, 36, and 60 months after the completion of treatment.
While there was a transient drop in the radiation-treated patients at the 6-month evaluation, mean total sperm number otherwise increased over time in all groups, according to the investigators’ report.
Similarly, mean sperm concentration significantly increased from baseline to 12 months post treatment in the surveillance, BEP, and carboplatin groups, with a nonsignificant decrease in the radiotherapy group, they said in the report.
There were generally no significant differences in sperm count or concentration for the treatments, compared with surveillance, beyond a significant decrease in mean sperm count for radiation versus surveillance, they added.
There were likewise no significant changes in sperm measures for seminoma and nonseminoma patients at any point over the 5 years of evaluation, reported data show.
“With the results of this study, we can now inform our patients that adjuvant chemotherapy does not seem to affect the testicular function,” Dr. Weibring and her colleagues concluded.
The authors reported that they had no conflicts of interest related to the study, which was supported by the Swedish Cancer Society, among other sources.
SOURCE: Weibring K et al. Ann Oncol. 2019 Feb 25. doi: 10.1093/annonc/mdz017/5348526.
Adjuvant treatments appear to have no significant detrimental long-term effects on sperm count in men being treated for clinical stage I testicular cancer, results of a recent investigation suggest.
Sperm number and concentration were largely stable over time in patients who received a round of chemotherapy or radiation to lymph nodes following orchiectomy, according to results of the 182-patient study.
Investigators said they still offer sperm banking before orchiectomy, since some patients will have low sperm counts prior to orchiectomy that persist after the procedure.
Moreover, the type of testicular cancer and the potential need for other postorchiectomy treatments are often “unknown factors” that underscore the importance of sperm banking, said the researchers, led by Kristina Weibring, MD, of Karolinska University Hospital, Stockholm.
“Assisted reproductive measures may be necessary for these patients regardless of any treatment given,” the researchers noted. The report is in Annals of Oncology.
The lack of effect on sperm counts in this study stands in contrast to previous studies, which clearly show the detrimental effects of multiple chemotherapy cycles on sperm recovery, the investigators said.
Their study comprised 182 patients 18-50 years of age with clinical stage I testicular cancer who underwent unilateral orchiectomy. Depending on tumor characteristics, the patients then received one cycle of adjuvant carboplatin, one cycle of a bleomycin, etoposide, and cisplatin (BEP) regimen, surveillance, or adjuvant radiotherapy to the infradiaphragmal para-aortic and ipsilateral iliac lymph nodes. Sperm samples were obtained at 6, 12, 24, 36, and 60 months after the completion of treatment.
While there was a transient drop in the radiation-treated patients at the 6-month evaluation, mean total sperm number otherwise increased over time in all groups, according to the investigators’ report.
Similarly, mean sperm concentration significantly increased from baseline to 12 months post treatment in the surveillance, BEP, and carboplatin groups, with a nonsignificant decrease in the radiotherapy group, they said in the report.
There were generally no significant differences in sperm count or concentration for the treatments, compared with surveillance, beyond a significant decrease in mean sperm count for radiation versus surveillance, they added.
There were likewise no significant changes in sperm measures for seminoma and nonseminoma patients at any point over the 5 years of evaluation, reported data show.
“With the results of this study, we can now inform our patients that adjuvant chemotherapy does not seem to affect the testicular function,” Dr. Weibring and her colleagues concluded.
The authors reported that they had no conflicts of interest related to the study, which was supported by the Swedish Cancer Society, among other sources.
SOURCE: Weibring K et al. Ann Oncol. 2019 Feb 25. doi: 10.1093/annonc/mdz017/5348526.
FROM ANNALS OF ONCOLOGY
Enzalutamide boosts ADT benefit in metastatic hormone-sensitive prostate cancer
SAN FRANCISCO – Dual targeting of the androgen signaling axis in metastatic hormone-sensitive prostate cancer improves outcomes and is well tolerated, according to results of the phase 3 ARCHES trial reported at the 2019 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology, ASTRO, and the Society of Urologic Oncology.
“Androgen-deprivation therapy [ADT] has been the mainstay of therapy for men who present with metastatic disease for many years. But generally, progression to castration-resistant diseases is observed within approximately 3 years,” said lead investigator Andrew J. Armstrong, MD, a professor of medicine in medical oncology at Duke University, Durham, N.C.
“Thus the advent of improving outcomes with the [additional] use of early docetaxel, abiraterone, and now radiation to the primary in the face of oligometastatic disease has been shown to improve survival,” he added. “However, the most recent studies have really not studied the sequential use of docetaxel and have largely excluded patients who had had prior docetaxel chemotherapy in the hormone-sensitive setting.”
The investigators in the ARCHES trial (NCT02677896) randomized 1,150 men with metastatic hormone-sensitive prostate cancer to enzalutamide (Xtandi) plus ADT or placebo plus ADT.
With a median follow-up of 14.4 months, radiographic progression-free survival – the trial’s primary endpoint – was prolonged in the enzalutamide arm. Risk of progression or death was reduced by a relative 61% with enzalutamide versus placebo. There were also significant relative reductions in risks of other important outcomes such as time to prostate-specific antigen (PSA) progression (81% reduction), time to new antineoplastic therapy (72% reduction), and time to castration resistance (72% reduction).
Meanwhile, the enzalutamide group had somewhat more fatigue, hot flashes, and hypertension, but most of these events were grade 1 or 2. Rates of adverse events leading to treatment withdrawal were similar and low.
“Following the success of this study, all patients in the placebo group will be offered participation in an open-label extension protocol where they’ll be offered enzalutamide and the opportunity to receive a clinically beneficial treatment,” Dr. Armstrong reported.
Ready for prime time?
The ARCHES findings are probably not yet sufficient to be implemented in real-world practice, according to invited discussant Ian D. Davis, MBBS, PhD, a professor at Monash University, Melbourne.
The condition studied in the trial – metastatic hormone-sensitive prostate cancer – certainly needs treatment, but it is unclear whether radiographic progression-free survival is a meaningful endpoint, he said. “We know in the CRPC [castration-resistant prostate cancer] setting for enzalutamide in the PREVAIL study, radiographic progression-free survival was correlated with overall survival, but we haven’t actually proven that yet in the CSPC [castration-sensitive prostate cancer] setting.”
Toxicity was acceptable, but information about the efficacy of subsequent treatment is lacking. “This is where progression-free survival 2 [PFS2] information would be useful if we had it,” Dr. Davis noted. Cost-effectiveness in terms of overall survival is likewise still unknown.
“Cautiously, and I’m conscious of my conflict of interest here because I’m doing a similar trial, I’d say that the ARCHES trial should probably not yet change practice,” he said. However, he qualified that statement by pointing to recently announced results from the phase 3 TITAN trial (NCT02489318) of apalutamide (Erleada), showing significant radiographic progression-free survival and overall survival benefit in this patient population. “So we await those data with interest.”
The first interim analysis of the enzalutamide trial he is cochairing, ANZUP ENZAMET (NCT02446405), is imminent, according to Dr. Davis. “The differences here are the primary endpoint of ENZAMET is overall survival and there was an early amendment to allow concurrent docetaxel, so a significant proportion of patients on this study will have received concurrent docetaxel with enzalutamide or ADT,” he noted.
Study details
Men enrolled in ARCHES were allowed to have received prior ADT for up to 3 months or, if they had received docetaxel, for up to 6 months, Dr. Armstrong noted at the symposium.
About two-thirds each had high disease volume and had distant metastasis at their initial diagnosis. Overall, median duration of prior ADT was 1.6 months.
Radiographic progression-free survival was not reached with enzalutamide versus 19.45 months with placebo (hazard ratio, 0.39; P less than .0001). Corresponding 12-month rates were 84% and 64%. Findings were essentially the same across diverse subgroups, including among the 18% of patients who had received prior docetaxel (HR, 0.53).
Median time to PSA progression – typically one of the first indications of castration resistance – was not reached in either group, but the 12-month rate was 91% with enzalutamide versus 63% with placebo (HR, 0.19; P less than .0001), Dr. Armstrong reported. Median time to castration resistance was not reached with enzalutamide versus 13.9 months with placebo (HR, 0.28; P less than .0001).
There also were significant differences in favor of enzalutamide on the rate of achievement of undetectable PSA (68.1% vs. 17.6%; P less than .0001), the objective response rate (83.1% vs. 63.7%; P less than .0001), and the time to initiation of new antineoplastic therapy (not reached vs. 30.19 months; HR, 0.28; P less than .0001).
Quality of life was very high in both groups at study baseline and remained similarly so during follow-up. An interim analysis showed overall survival had not been reached in either group, although there was a nonsignificant trend favoring enzalutamide.
Safety was much the same, with the enzalutamide and placebo groups having similar rates of grade 3 or worse adverse events (24.3% vs. 25.6%), as well as similar rates of adverse events leading to treatment withdrawal (7.2% vs. 5.2%) and death (2.4% vs. 1.7%).
In terms of adverse events of special interest, the groups were comparable on rates of grade 3 or 4 convulsion, ischemic heart disease, falls, and fractures.
Dr. Armstrong disclosed that he has a consulting or advisory role with, receives research funding (institutional) from, and receives travel, accommodations, and/or expenses from Astellas – among other disclosures. The trial was sponsored by Astellas.
SOURCE: Armstrong AJ et al. GUCS 2019, Abstract 687.
SAN FRANCISCO – Dual targeting of the androgen signaling axis in metastatic hormone-sensitive prostate cancer improves outcomes and is well tolerated, according to results of the phase 3 ARCHES trial reported at the 2019 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology, ASTRO, and the Society of Urologic Oncology.
“Androgen-deprivation therapy [ADT] has been the mainstay of therapy for men who present with metastatic disease for many years. But generally, progression to castration-resistant diseases is observed within approximately 3 years,” said lead investigator Andrew J. Armstrong, MD, a professor of medicine in medical oncology at Duke University, Durham, N.C.
“Thus the advent of improving outcomes with the [additional] use of early docetaxel, abiraterone, and now radiation to the primary in the face of oligometastatic disease has been shown to improve survival,” he added. “However, the most recent studies have really not studied the sequential use of docetaxel and have largely excluded patients who had had prior docetaxel chemotherapy in the hormone-sensitive setting.”
The investigators in the ARCHES trial (NCT02677896) randomized 1,150 men with metastatic hormone-sensitive prostate cancer to enzalutamide (Xtandi) plus ADT or placebo plus ADT.
With a median follow-up of 14.4 months, radiographic progression-free survival – the trial’s primary endpoint – was prolonged in the enzalutamide arm. Risk of progression or death was reduced by a relative 61% with enzalutamide versus placebo. There were also significant relative reductions in risks of other important outcomes such as time to prostate-specific antigen (PSA) progression (81% reduction), time to new antineoplastic therapy (72% reduction), and time to castration resistance (72% reduction).
Meanwhile, the enzalutamide group had somewhat more fatigue, hot flashes, and hypertension, but most of these events were grade 1 or 2. Rates of adverse events leading to treatment withdrawal were similar and low.
“Following the success of this study, all patients in the placebo group will be offered participation in an open-label extension protocol where they’ll be offered enzalutamide and the opportunity to receive a clinically beneficial treatment,” Dr. Armstrong reported.
Ready for prime time?
The ARCHES findings are probably not yet sufficient to be implemented in real-world practice, according to invited discussant Ian D. Davis, MBBS, PhD, a professor at Monash University, Melbourne.
The condition studied in the trial – metastatic hormone-sensitive prostate cancer – certainly needs treatment, but it is unclear whether radiographic progression-free survival is a meaningful endpoint, he said. “We know in the CRPC [castration-resistant prostate cancer] setting for enzalutamide in the PREVAIL study, radiographic progression-free survival was correlated with overall survival, but we haven’t actually proven that yet in the CSPC [castration-sensitive prostate cancer] setting.”
Toxicity was acceptable, but information about the efficacy of subsequent treatment is lacking. “This is where progression-free survival 2 [PFS2] information would be useful if we had it,” Dr. Davis noted. Cost-effectiveness in terms of overall survival is likewise still unknown.
“Cautiously, and I’m conscious of my conflict of interest here because I’m doing a similar trial, I’d say that the ARCHES trial should probably not yet change practice,” he said. However, he qualified that statement by pointing to recently announced results from the phase 3 TITAN trial (NCT02489318) of apalutamide (Erleada), showing significant radiographic progression-free survival and overall survival benefit in this patient population. “So we await those data with interest.”
The first interim analysis of the enzalutamide trial he is cochairing, ANZUP ENZAMET (NCT02446405), is imminent, according to Dr. Davis. “The differences here are the primary endpoint of ENZAMET is overall survival and there was an early amendment to allow concurrent docetaxel, so a significant proportion of patients on this study will have received concurrent docetaxel with enzalutamide or ADT,” he noted.
Study details
Men enrolled in ARCHES were allowed to have received prior ADT for up to 3 months or, if they had received docetaxel, for up to 6 months, Dr. Armstrong noted at the symposium.
About two-thirds each had high disease volume and had distant metastasis at their initial diagnosis. Overall, median duration of prior ADT was 1.6 months.
Radiographic progression-free survival was not reached with enzalutamide versus 19.45 months with placebo (hazard ratio, 0.39; P less than .0001). Corresponding 12-month rates were 84% and 64%. Findings were essentially the same across diverse subgroups, including among the 18% of patients who had received prior docetaxel (HR, 0.53).
Median time to PSA progression – typically one of the first indications of castration resistance – was not reached in either group, but the 12-month rate was 91% with enzalutamide versus 63% with placebo (HR, 0.19; P less than .0001), Dr. Armstrong reported. Median time to castration resistance was not reached with enzalutamide versus 13.9 months with placebo (HR, 0.28; P less than .0001).
There also were significant differences in favor of enzalutamide on the rate of achievement of undetectable PSA (68.1% vs. 17.6%; P less than .0001), the objective response rate (83.1% vs. 63.7%; P less than .0001), and the time to initiation of new antineoplastic therapy (not reached vs. 30.19 months; HR, 0.28; P less than .0001).
Quality of life was very high in both groups at study baseline and remained similarly so during follow-up. An interim analysis showed overall survival had not been reached in either group, although there was a nonsignificant trend favoring enzalutamide.
Safety was much the same, with the enzalutamide and placebo groups having similar rates of grade 3 or worse adverse events (24.3% vs. 25.6%), as well as similar rates of adverse events leading to treatment withdrawal (7.2% vs. 5.2%) and death (2.4% vs. 1.7%).
In terms of adverse events of special interest, the groups were comparable on rates of grade 3 or 4 convulsion, ischemic heart disease, falls, and fractures.
Dr. Armstrong disclosed that he has a consulting or advisory role with, receives research funding (institutional) from, and receives travel, accommodations, and/or expenses from Astellas – among other disclosures. The trial was sponsored by Astellas.
SOURCE: Armstrong AJ et al. GUCS 2019, Abstract 687.
SAN FRANCISCO – Dual targeting of the androgen signaling axis in metastatic hormone-sensitive prostate cancer improves outcomes and is well tolerated, according to results of the phase 3 ARCHES trial reported at the 2019 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology, ASTRO, and the Society of Urologic Oncology.
“Androgen-deprivation therapy [ADT] has been the mainstay of therapy for men who present with metastatic disease for many years. But generally, progression to castration-resistant diseases is observed within approximately 3 years,” said lead investigator Andrew J. Armstrong, MD, a professor of medicine in medical oncology at Duke University, Durham, N.C.
“Thus the advent of improving outcomes with the [additional] use of early docetaxel, abiraterone, and now radiation to the primary in the face of oligometastatic disease has been shown to improve survival,” he added. “However, the most recent studies have really not studied the sequential use of docetaxel and have largely excluded patients who had had prior docetaxel chemotherapy in the hormone-sensitive setting.”
The investigators in the ARCHES trial (NCT02677896) randomized 1,150 men with metastatic hormone-sensitive prostate cancer to enzalutamide (Xtandi) plus ADT or placebo plus ADT.
With a median follow-up of 14.4 months, radiographic progression-free survival – the trial’s primary endpoint – was prolonged in the enzalutamide arm. Risk of progression or death was reduced by a relative 61% with enzalutamide versus placebo. There were also significant relative reductions in risks of other important outcomes such as time to prostate-specific antigen (PSA) progression (81% reduction), time to new antineoplastic therapy (72% reduction), and time to castration resistance (72% reduction).
Meanwhile, the enzalutamide group had somewhat more fatigue, hot flashes, and hypertension, but most of these events were grade 1 or 2. Rates of adverse events leading to treatment withdrawal were similar and low.
“Following the success of this study, all patients in the placebo group will be offered participation in an open-label extension protocol where they’ll be offered enzalutamide and the opportunity to receive a clinically beneficial treatment,” Dr. Armstrong reported.
Ready for prime time?
The ARCHES findings are probably not yet sufficient to be implemented in real-world practice, according to invited discussant Ian D. Davis, MBBS, PhD, a professor at Monash University, Melbourne.
The condition studied in the trial – metastatic hormone-sensitive prostate cancer – certainly needs treatment, but it is unclear whether radiographic progression-free survival is a meaningful endpoint, he said. “We know in the CRPC [castration-resistant prostate cancer] setting for enzalutamide in the PREVAIL study, radiographic progression-free survival was correlated with overall survival, but we haven’t actually proven that yet in the CSPC [castration-sensitive prostate cancer] setting.”
Toxicity was acceptable, but information about the efficacy of subsequent treatment is lacking. “This is where progression-free survival 2 [PFS2] information would be useful if we had it,” Dr. Davis noted. Cost-effectiveness in terms of overall survival is likewise still unknown.
“Cautiously, and I’m conscious of my conflict of interest here because I’m doing a similar trial, I’d say that the ARCHES trial should probably not yet change practice,” he said. However, he qualified that statement by pointing to recently announced results from the phase 3 TITAN trial (NCT02489318) of apalutamide (Erleada), showing significant radiographic progression-free survival and overall survival benefit in this patient population. “So we await those data with interest.”
The first interim analysis of the enzalutamide trial he is cochairing, ANZUP ENZAMET (NCT02446405), is imminent, according to Dr. Davis. “The differences here are the primary endpoint of ENZAMET is overall survival and there was an early amendment to allow concurrent docetaxel, so a significant proportion of patients on this study will have received concurrent docetaxel with enzalutamide or ADT,” he noted.
Study details
Men enrolled in ARCHES were allowed to have received prior ADT for up to 3 months or, if they had received docetaxel, for up to 6 months, Dr. Armstrong noted at the symposium.
About two-thirds each had high disease volume and had distant metastasis at their initial diagnosis. Overall, median duration of prior ADT was 1.6 months.
Radiographic progression-free survival was not reached with enzalutamide versus 19.45 months with placebo (hazard ratio, 0.39; P less than .0001). Corresponding 12-month rates were 84% and 64%. Findings were essentially the same across diverse subgroups, including among the 18% of patients who had received prior docetaxel (HR, 0.53).
Median time to PSA progression – typically one of the first indications of castration resistance – was not reached in either group, but the 12-month rate was 91% with enzalutamide versus 63% with placebo (HR, 0.19; P less than .0001), Dr. Armstrong reported. Median time to castration resistance was not reached with enzalutamide versus 13.9 months with placebo (HR, 0.28; P less than .0001).
There also were significant differences in favor of enzalutamide on the rate of achievement of undetectable PSA (68.1% vs. 17.6%; P less than .0001), the objective response rate (83.1% vs. 63.7%; P less than .0001), and the time to initiation of new antineoplastic therapy (not reached vs. 30.19 months; HR, 0.28; P less than .0001).
Quality of life was very high in both groups at study baseline and remained similarly so during follow-up. An interim analysis showed overall survival had not been reached in either group, although there was a nonsignificant trend favoring enzalutamide.
Safety was much the same, with the enzalutamide and placebo groups having similar rates of grade 3 or worse adverse events (24.3% vs. 25.6%), as well as similar rates of adverse events leading to treatment withdrawal (7.2% vs. 5.2%) and death (2.4% vs. 1.7%).
In terms of adverse events of special interest, the groups were comparable on rates of grade 3 or 4 convulsion, ischemic heart disease, falls, and fractures.
Dr. Armstrong disclosed that he has a consulting or advisory role with, receives research funding (institutional) from, and receives travel, accommodations, and/or expenses from Astellas – among other disclosures. The trial was sponsored by Astellas.
SOURCE: Armstrong AJ et al. GUCS 2019, Abstract 687.
REPORTING FROM GUCS 2019
Model shows nivolumab plus ipilimumab cost effective in mRCC
First-line treatment with nivolumab plus ipilimumab may be cost effective when compared with sunitinib for patients with intermediate- and poor-risk metastatic renal cell carcinoma (mRCC), according to a cost-effectiveness analysis.
“A Markov model was developed to estimate the costs and effectiveness of treatment of mRCC,” XiaoMin Wan, PhD, of the Capital Medical University in Beijing and his colleagues wrote in JAMA Oncology.
The researchers used data from an economic model that extrapolated findings from a phase 3 randomized study of 1,096 patients with intermediate- and poor-risk mRCC treated with first-line sunitinib or nivolumab plus ipilimumab.
“We assumed that the first-line treatments continued until disease progression or unacceptable toxic effects,” the team wrote.
Several measures were estimated using the model, including quality-adjusted life-years (QALYs), lifetime costs, and life-years. The team set a willingness-to-pay threshold of $100,000-$150,000 per QALY. In addition, Dr. Wan and his colleagues completed a sensitivity analysis to investigate how the results changed across different ranges of drug cost.
After analysis, the researchers found that first-line therapy with nivolumab plus ipilimumab was estimated to cost $108,363 per QALY gained. The incremental QALYs added using the combination was 0.96 years versus sunitinib, at the same cost per QALY.
With respect to the sensitivity analysis, the likelihood of the combination being cost effective, compared with sunitinib, was calculated to be 42.5% and 80.2% at the minimum and maximum of the willingness-to-pay threshold, respectively.
“The results of subgroup analyses showed that nivolumab plus ipilimumab was most cost effective for patients with 1% or greater programmed cell death 1 ligand 1 (PD-L1) expression,” they added.
The researchers acknowledged a key limitation of the study was that the analysis used data from only a single randomized trial. Consequently, Dr. Wan and his colleagues reported that any bias contained within that trial will also be present in this analysis.
The study was supported by funding from the National Natural Science Foundation of China and the Health and Family Planning Commission of Hunan province. The authors reported no conflicts of interest.
SOURCE: Wan X et al. JAMA Oncol. 2019 Feb 21. doi: 10.1001/jamaoncol.2018.7086.
First-line treatment with nivolumab plus ipilimumab may be cost effective when compared with sunitinib for patients with intermediate- and poor-risk metastatic renal cell carcinoma (mRCC), according to a cost-effectiveness analysis.
“A Markov model was developed to estimate the costs and effectiveness of treatment of mRCC,” XiaoMin Wan, PhD, of the Capital Medical University in Beijing and his colleagues wrote in JAMA Oncology.
The researchers used data from an economic model that extrapolated findings from a phase 3 randomized study of 1,096 patients with intermediate- and poor-risk mRCC treated with first-line sunitinib or nivolumab plus ipilimumab.
“We assumed that the first-line treatments continued until disease progression or unacceptable toxic effects,” the team wrote.
Several measures were estimated using the model, including quality-adjusted life-years (QALYs), lifetime costs, and life-years. The team set a willingness-to-pay threshold of $100,000-$150,000 per QALY. In addition, Dr. Wan and his colleagues completed a sensitivity analysis to investigate how the results changed across different ranges of drug cost.
After analysis, the researchers found that first-line therapy with nivolumab plus ipilimumab was estimated to cost $108,363 per QALY gained. The incremental QALYs added using the combination was 0.96 years versus sunitinib, at the same cost per QALY.
With respect to the sensitivity analysis, the likelihood of the combination being cost effective, compared with sunitinib, was calculated to be 42.5% and 80.2% at the minimum and maximum of the willingness-to-pay threshold, respectively.
“The results of subgroup analyses showed that nivolumab plus ipilimumab was most cost effective for patients with 1% or greater programmed cell death 1 ligand 1 (PD-L1) expression,” they added.
The researchers acknowledged a key limitation of the study was that the analysis used data from only a single randomized trial. Consequently, Dr. Wan and his colleagues reported that any bias contained within that trial will also be present in this analysis.
The study was supported by funding from the National Natural Science Foundation of China and the Health and Family Planning Commission of Hunan province. The authors reported no conflicts of interest.
SOURCE: Wan X et al. JAMA Oncol. 2019 Feb 21. doi: 10.1001/jamaoncol.2018.7086.
First-line treatment with nivolumab plus ipilimumab may be cost effective when compared with sunitinib for patients with intermediate- and poor-risk metastatic renal cell carcinoma (mRCC), according to a cost-effectiveness analysis.
“A Markov model was developed to estimate the costs and effectiveness of treatment of mRCC,” XiaoMin Wan, PhD, of the Capital Medical University in Beijing and his colleagues wrote in JAMA Oncology.
The researchers used data from an economic model that extrapolated findings from a phase 3 randomized study of 1,096 patients with intermediate- and poor-risk mRCC treated with first-line sunitinib or nivolumab plus ipilimumab.
“We assumed that the first-line treatments continued until disease progression or unacceptable toxic effects,” the team wrote.
Several measures were estimated using the model, including quality-adjusted life-years (QALYs), lifetime costs, and life-years. The team set a willingness-to-pay threshold of $100,000-$150,000 per QALY. In addition, Dr. Wan and his colleagues completed a sensitivity analysis to investigate how the results changed across different ranges of drug cost.
After analysis, the researchers found that first-line therapy with nivolumab plus ipilimumab was estimated to cost $108,363 per QALY gained. The incremental QALYs added using the combination was 0.96 years versus sunitinib, at the same cost per QALY.
With respect to the sensitivity analysis, the likelihood of the combination being cost effective, compared with sunitinib, was calculated to be 42.5% and 80.2% at the minimum and maximum of the willingness-to-pay threshold, respectively.
“The results of subgroup analyses showed that nivolumab plus ipilimumab was most cost effective for patients with 1% or greater programmed cell death 1 ligand 1 (PD-L1) expression,” they added.
The researchers acknowledged a key limitation of the study was that the analysis used data from only a single randomized trial. Consequently, Dr. Wan and his colleagues reported that any bias contained within that trial will also be present in this analysis.
The study was supported by funding from the National Natural Science Foundation of China and the Health and Family Planning Commission of Hunan province. The authors reported no conflicts of interest.
SOURCE: Wan X et al. JAMA Oncol. 2019 Feb 21. doi: 10.1001/jamaoncol.2018.7086.
REPORTING FROM JAMA ONCOLOGY







