User login
ASH 2018 coming attractions look at the big picture
In the closest thing the medical world has to movie trailers, the American Society of Hematology held a press conference offering
Shorter R-CHOP regimen for DLBCL
Under the heading “Big Trials, Big Results” will be data from the FLYER trial, a phase 3, randomized, deescalation trial in 592 patients aged 18-60 years with favorable-prognosis diffuse large B-cell lymphoma. The investigators report that both progression-free survival and overall survival with four cycles of R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone) were noninferior to those for patients treated with six cycles of R-CHOP (abstract 781).
Ibrutinib mastery in CLL
Also on the program are results of a study showing that ibrutinib (Imbruvica), either alone or in combination with rituximab, is associated with superior progression-free survival than bendamustine and rituximab in older patients with chronic lymphocytic leukemia (CLL).
The trial, the Alliance North American Intergroup Study A041202 (abstract 6) is the first major trial to pit ibrutinib against the modern standard of immunochemotherapy rather than the older standard of chlorambucil, Dr. Brodsky noted.
Anemia support in beta-thalassemia, MDS
In nonmalignant disease, investigators in the randomized, phase 3 BELIEVE trial are reporting results of their study showing that the first-in-class erythroid maturation agent luspatercept was associated with significant reductions in the need for RBC transfusion in adults with transfusion-dependent beta-thalassemia.
The investigators report that the experimental agent was “generally well tolerated” (abstract 163).
“Beyond a proof of principle, [this is] certainly a very exciting advancement in this group of patients who otherwise had very few treatment options,” said Alexis A. Thompson, MD, associate director of equity and minority health at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago, and the current ASH president.
Dr. Thompson also highlighted the MEDALIST trial (abstract 1), a phase 3, randomized study showing that luspatercept significantly reduced transfusion burden, compared with placebo, in patients with anemia caused by very low–, low-, or intermediate-risk myelodysplastic syndrome with ring sideroblasts who require RBC transfusions.
“This group of patients were individuals who were refractory or were not responders or did not tolerate erythropoietic stimulating agents and therefore were requiring regular transfusion,” Dr. Thompson said.
Worth the wait
The late-breaking abstract program was stretched from the usual six abstracts to seven this year because of the unusually high quality of the science, Dr. Brodsky said.
Among these star attractions are results of a phase 3, randomized study of daratumumab (Darzalex) plus lenalidomide and dexamethasone versus lenalidomide-dexamethasone alone for patients with newly diagnosed multiple myeloma who are ineligible for transplant.
The investigators found that adding daratumumab reduced the risk of disease progression or death by close to 50%, supporting the combination as a new standard of care in these patients, according to Thierry Facon, MD, from the Hospital Claude Huriez in Lille, France, and colleagues (abstract LBA-2).
Two other late-breakers deal with CLL. The first, a randomized, phase 3 study of ibrutinib-based therapy versus standard fludarabine, cyclophosphamide, and rituximab chemoimmunotherapy in younger patients with untreated CLL, found that ibrutinib and rituximab provided significantly better progression-free survival and overall survival (abstract LBA-4).
“These findings have immediate practice-changing implications and establish ibrutinib-based therapy as the most efficacious first-line therapy for patients with CLL,” wrote Tait D. Shanafelt, MD, from Stanford (Calif.) University, and colleagues.
On a less positive note, Australian researchers report their discovery of a recurrent mutation in BCL2 that confers resistance to venetoclax (Venclexta) in patients with progressive CLL (abstract LBA-7).
“This mutation provides new insights into the pathobiology of venetoclax resistance and provides a potential biomarker of impending clinical relapse,” wrote Piers Blombery, MBBS, from the University of Melbourne, and colleagues.
Finally, investigators from children’s hospitals in the United States and Europe report promising findings on the safety and efficacy of emapalumab for the treatment of patients with the rare genetic disorder primary hemophagocytic lymphohistiocytosis (HLH).
The drug, newly approved by the Food and Drug Administration, was able to control HLH’s hyperinflammatory activity, and allowed a substantial proportion of patients to survive to hematopoietic stem cell transplantation, the investigators said (abstract LBA-6).
In the closest thing the medical world has to movie trailers, the American Society of Hematology held a press conference offering
Shorter R-CHOP regimen for DLBCL
Under the heading “Big Trials, Big Results” will be data from the FLYER trial, a phase 3, randomized, deescalation trial in 592 patients aged 18-60 years with favorable-prognosis diffuse large B-cell lymphoma. The investigators report that both progression-free survival and overall survival with four cycles of R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone) were noninferior to those for patients treated with six cycles of R-CHOP (abstract 781).
Ibrutinib mastery in CLL
Also on the program are results of a study showing that ibrutinib (Imbruvica), either alone or in combination with rituximab, is associated with superior progression-free survival than bendamustine and rituximab in older patients with chronic lymphocytic leukemia (CLL).
The trial, the Alliance North American Intergroup Study A041202 (abstract 6) is the first major trial to pit ibrutinib against the modern standard of immunochemotherapy rather than the older standard of chlorambucil, Dr. Brodsky noted.
Anemia support in beta-thalassemia, MDS
In nonmalignant disease, investigators in the randomized, phase 3 BELIEVE trial are reporting results of their study showing that the first-in-class erythroid maturation agent luspatercept was associated with significant reductions in the need for RBC transfusion in adults with transfusion-dependent beta-thalassemia.
The investigators report that the experimental agent was “generally well tolerated” (abstract 163).
“Beyond a proof of principle, [this is] certainly a very exciting advancement in this group of patients who otherwise had very few treatment options,” said Alexis A. Thompson, MD, associate director of equity and minority health at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago, and the current ASH president.
Dr. Thompson also highlighted the MEDALIST trial (abstract 1), a phase 3, randomized study showing that luspatercept significantly reduced transfusion burden, compared with placebo, in patients with anemia caused by very low–, low-, or intermediate-risk myelodysplastic syndrome with ring sideroblasts who require RBC transfusions.
“This group of patients were individuals who were refractory or were not responders or did not tolerate erythropoietic stimulating agents and therefore were requiring regular transfusion,” Dr. Thompson said.
Worth the wait
The late-breaking abstract program was stretched from the usual six abstracts to seven this year because of the unusually high quality of the science, Dr. Brodsky said.
Among these star attractions are results of a phase 3, randomized study of daratumumab (Darzalex) plus lenalidomide and dexamethasone versus lenalidomide-dexamethasone alone for patients with newly diagnosed multiple myeloma who are ineligible for transplant.
The investigators found that adding daratumumab reduced the risk of disease progression or death by close to 50%, supporting the combination as a new standard of care in these patients, according to Thierry Facon, MD, from the Hospital Claude Huriez in Lille, France, and colleagues (abstract LBA-2).
Two other late-breakers deal with CLL. The first, a randomized, phase 3 study of ibrutinib-based therapy versus standard fludarabine, cyclophosphamide, and rituximab chemoimmunotherapy in younger patients with untreated CLL, found that ibrutinib and rituximab provided significantly better progression-free survival and overall survival (abstract LBA-4).
“These findings have immediate practice-changing implications and establish ibrutinib-based therapy as the most efficacious first-line therapy for patients with CLL,” wrote Tait D. Shanafelt, MD, from Stanford (Calif.) University, and colleagues.
On a less positive note, Australian researchers report their discovery of a recurrent mutation in BCL2 that confers resistance to venetoclax (Venclexta) in patients with progressive CLL (abstract LBA-7).
“This mutation provides new insights into the pathobiology of venetoclax resistance and provides a potential biomarker of impending clinical relapse,” wrote Piers Blombery, MBBS, from the University of Melbourne, and colleagues.
Finally, investigators from children’s hospitals in the United States and Europe report promising findings on the safety and efficacy of emapalumab for the treatment of patients with the rare genetic disorder primary hemophagocytic lymphohistiocytosis (HLH).
The drug, newly approved by the Food and Drug Administration, was able to control HLH’s hyperinflammatory activity, and allowed a substantial proportion of patients to survive to hematopoietic stem cell transplantation, the investigators said (abstract LBA-6).
In the closest thing the medical world has to movie trailers, the American Society of Hematology held a press conference offering
Shorter R-CHOP regimen for DLBCL
Under the heading “Big Trials, Big Results” will be data from the FLYER trial, a phase 3, randomized, deescalation trial in 592 patients aged 18-60 years with favorable-prognosis diffuse large B-cell lymphoma. The investigators report that both progression-free survival and overall survival with four cycles of R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone) were noninferior to those for patients treated with six cycles of R-CHOP (abstract 781).
Ibrutinib mastery in CLL
Also on the program are results of a study showing that ibrutinib (Imbruvica), either alone or in combination with rituximab, is associated with superior progression-free survival than bendamustine and rituximab in older patients with chronic lymphocytic leukemia (CLL).
The trial, the Alliance North American Intergroup Study A041202 (abstract 6) is the first major trial to pit ibrutinib against the modern standard of immunochemotherapy rather than the older standard of chlorambucil, Dr. Brodsky noted.
Anemia support in beta-thalassemia, MDS
In nonmalignant disease, investigators in the randomized, phase 3 BELIEVE trial are reporting results of their study showing that the first-in-class erythroid maturation agent luspatercept was associated with significant reductions in the need for RBC transfusion in adults with transfusion-dependent beta-thalassemia.
The investigators report that the experimental agent was “generally well tolerated” (abstract 163).
“Beyond a proof of principle, [this is] certainly a very exciting advancement in this group of patients who otherwise had very few treatment options,” said Alexis A. Thompson, MD, associate director of equity and minority health at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago, and the current ASH president.
Dr. Thompson also highlighted the MEDALIST trial (abstract 1), a phase 3, randomized study showing that luspatercept significantly reduced transfusion burden, compared with placebo, in patients with anemia caused by very low–, low-, or intermediate-risk myelodysplastic syndrome with ring sideroblasts who require RBC transfusions.
“This group of patients were individuals who were refractory or were not responders or did not tolerate erythropoietic stimulating agents and therefore were requiring regular transfusion,” Dr. Thompson said.
Worth the wait
The late-breaking abstract program was stretched from the usual six abstracts to seven this year because of the unusually high quality of the science, Dr. Brodsky said.
Among these star attractions are results of a phase 3, randomized study of daratumumab (Darzalex) plus lenalidomide and dexamethasone versus lenalidomide-dexamethasone alone for patients with newly diagnosed multiple myeloma who are ineligible for transplant.
The investigators found that adding daratumumab reduced the risk of disease progression or death by close to 50%, supporting the combination as a new standard of care in these patients, according to Thierry Facon, MD, from the Hospital Claude Huriez in Lille, France, and colleagues (abstract LBA-2).
Two other late-breakers deal with CLL. The first, a randomized, phase 3 study of ibrutinib-based therapy versus standard fludarabine, cyclophosphamide, and rituximab chemoimmunotherapy in younger patients with untreated CLL, found that ibrutinib and rituximab provided significantly better progression-free survival and overall survival (abstract LBA-4).
“These findings have immediate practice-changing implications and establish ibrutinib-based therapy as the most efficacious first-line therapy for patients with CLL,” wrote Tait D. Shanafelt, MD, from Stanford (Calif.) University, and colleagues.
On a less positive note, Australian researchers report their discovery of a recurrent mutation in BCL2 that confers resistance to venetoclax (Venclexta) in patients with progressive CLL (abstract LBA-7).
“This mutation provides new insights into the pathobiology of venetoclax resistance and provides a potential biomarker of impending clinical relapse,” wrote Piers Blombery, MBBS, from the University of Melbourne, and colleagues.
Finally, investigators from children’s hospitals in the United States and Europe report promising findings on the safety and efficacy of emapalumab for the treatment of patients with the rare genetic disorder primary hemophagocytic lymphohistiocytosis (HLH).
The drug, newly approved by the Food and Drug Administration, was able to control HLH’s hyperinflammatory activity, and allowed a substantial proportion of patients to survive to hematopoietic stem cell transplantation, the investigators said (abstract LBA-6).
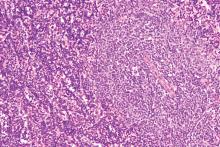
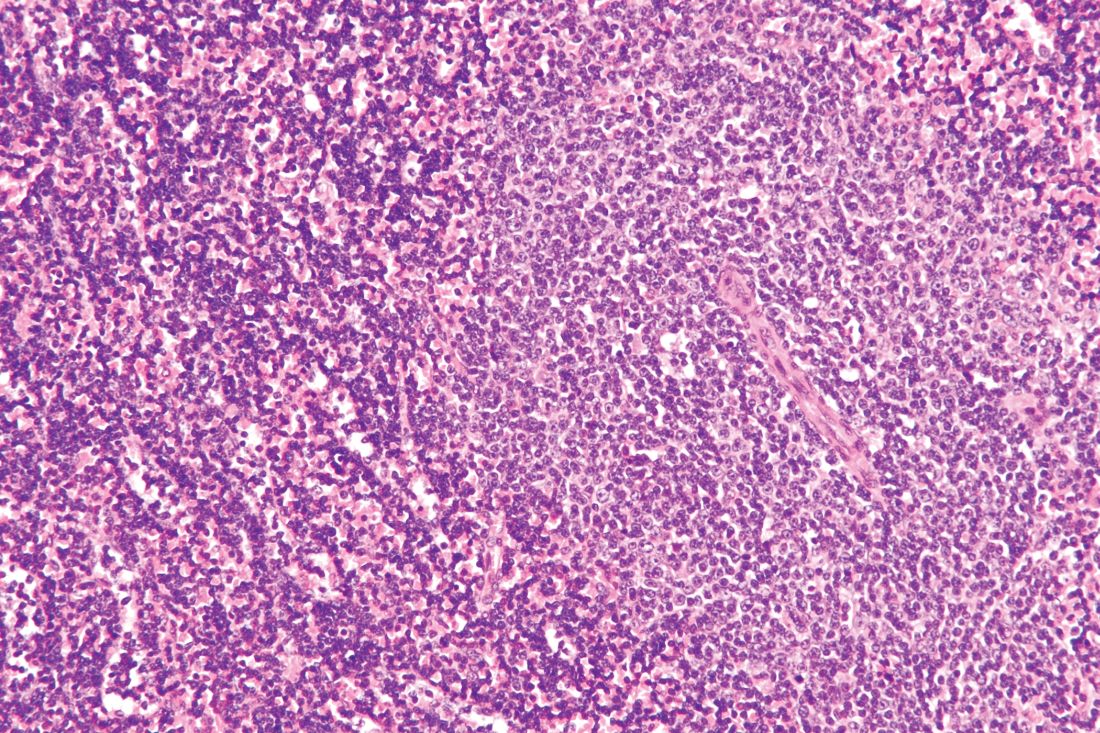
Consider treatment, testing when CLL symptoms emerge
CHICAGO – said Paul M. Barr, MD.
He described a patient who had been observed for 7 years when he began to complain of increasing fatigue and lost work time. A complete blood count (CBC) showed thrombocytopenia.
The recently updated International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines state that assessments before treatment in this type of patient should include history and physical, evaluation of infectious disease status, and routine laboratory testing – including CBC and differential, chemistry, serum immunoglobulin, and direct antiglobulin test.
“Bone marrow biopsies and [computed tomography] scans are listed as ‘not necessarily required,’ ” Dr. Barr, medical director of the Clinical Trials Office for Wilmot Cancer Institute at the University of Rochester (N.Y.) said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
He added that he opts for CT scans prior to therapy “to understand the patient’s disease burden and potentially to compare to later” and that bone marrow biopsy is “still very reasonable” for understanding the source of a patient’s cytopenias.
“Is it marrow failure or [immune thrombocytopenia]? Could the patient have [myelodysplastic syndrome]? All important considerations,” he said.
Positron emission tomography (PET) scans, however, are only considered when there is concern about transformation, he noted.
Predictive tests that should be conducted before initiating therapy, and that could help in guiding therapy decisions, include TP53 mutation testing and immunoglobulin heavy chain variable region gene (IGHV) mutational status testing (although this doesn’t need to be repeated if it was done at diagnosis because mutational status doesn’t change). Another helpful test is molecular cytogenetics using fluorescence in situ hybridization (FISH) for del(13q), del(17p), and trisomy 12 in peripheral blood lymphocytes. This should be repeated even it was done at diagnosis because patients can acquire additional molecular aberrations over time, Dr. Barr said.
Among the data that justify this advice for predictive testing are studies showing the curative potential of fludarabine/cyclophosphamide/rituximab (FCR) in mutated IGHV CLL, the progression-free survival (PFS) benefits of ibrutinib for patients with del(17p), and the activity of idelalisib in relapsed/refractory CLL patients, including those with TP53 dysfunction.
“The IGHV mutation status is useful to know what to expect from chemoimmunotherapy over time,” Dr. Barr said, explaining that several analyses demonstrate that patients with mutated IGHV genes (patients with low-risk disease) respond exceptionally well to chemoimmunotherapy, especially FCR.
In fact, studies, including a 2016 study by Philip A. Thompson and his colleagues and another by Kirsten Fischer and her colleagues, show that nearly 60% of patients with IGHV mutation remain in remission 10 years after FCR treatment, he said. However, the same is not necessarily true for bendamustine/rituximab (BR); the CLL10 study showed a significantly greater PFS with FCR, compared with that seen with BR.
Unmutated patients in that study had lower PFS, but the outcomes were still better with FCR than with BR, he said.
Studies of novel agents, including ibrutinib and idelalisib, suggest they may have particular benefit in higher-risk patients.
Ibrutinib was shown in a phase 2 study to be of benefit regardless of IGHV status, and this was replicated in the first-line RESONATE 2 study, which compared ibrutinib with chlorambucil and showed it had better PFS than that seen in unmutated patients treated with FCR in other studies, said Dr. Barr, the first author on that study.
“So you can see how the treatment paradigms are starting to evolve. It does look like ... comparing across trials, that ibrutinib leads to better remission durations, compared to chemoimmunotherapy, so far,” he said.
Ibrutinib has also been shown to be of benefit for patients with del(17p). A single-arm phase 2 study showed 79% PFS in relapsed, high-risk patients, which is much better than has been seen with chemoimmunotherapy, he noted.
“Venetoclax is also a very good option for this patient population in the relapse setting,” he said, adding that the PFS with venetoclax has been shown to be very similar to that with ibrutinib.
Similarly, idelalisib has been shown to provide comparable benefit in relapsed/refractory CLL patients, with and without del(17p)/TP53 mutation, he said.
Dr. Barr is a consultant for Pharmacyclics, AbbVie, Celgene, Gilead, Infinity, Novartis, and Seattle Genetics and has received research funding from Pharmacyclics and AbbVie.
CHICAGO – said Paul M. Barr, MD.
He described a patient who had been observed for 7 years when he began to complain of increasing fatigue and lost work time. A complete blood count (CBC) showed thrombocytopenia.
The recently updated International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines state that assessments before treatment in this type of patient should include history and physical, evaluation of infectious disease status, and routine laboratory testing – including CBC and differential, chemistry, serum immunoglobulin, and direct antiglobulin test.
“Bone marrow biopsies and [computed tomography] scans are listed as ‘not necessarily required,’ ” Dr. Barr, medical director of the Clinical Trials Office for Wilmot Cancer Institute at the University of Rochester (N.Y.) said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
He added that he opts for CT scans prior to therapy “to understand the patient’s disease burden and potentially to compare to later” and that bone marrow biopsy is “still very reasonable” for understanding the source of a patient’s cytopenias.
“Is it marrow failure or [immune thrombocytopenia]? Could the patient have [myelodysplastic syndrome]? All important considerations,” he said.
Positron emission tomography (PET) scans, however, are only considered when there is concern about transformation, he noted.
Predictive tests that should be conducted before initiating therapy, and that could help in guiding therapy decisions, include TP53 mutation testing and immunoglobulin heavy chain variable region gene (IGHV) mutational status testing (although this doesn’t need to be repeated if it was done at diagnosis because mutational status doesn’t change). Another helpful test is molecular cytogenetics using fluorescence in situ hybridization (FISH) for del(13q), del(17p), and trisomy 12 in peripheral blood lymphocytes. This should be repeated even it was done at diagnosis because patients can acquire additional molecular aberrations over time, Dr. Barr said.
Among the data that justify this advice for predictive testing are studies showing the curative potential of fludarabine/cyclophosphamide/rituximab (FCR) in mutated IGHV CLL, the progression-free survival (PFS) benefits of ibrutinib for patients with del(17p), and the activity of idelalisib in relapsed/refractory CLL patients, including those with TP53 dysfunction.
“The IGHV mutation status is useful to know what to expect from chemoimmunotherapy over time,” Dr. Barr said, explaining that several analyses demonstrate that patients with mutated IGHV genes (patients with low-risk disease) respond exceptionally well to chemoimmunotherapy, especially FCR.
In fact, studies, including a 2016 study by Philip A. Thompson and his colleagues and another by Kirsten Fischer and her colleagues, show that nearly 60% of patients with IGHV mutation remain in remission 10 years after FCR treatment, he said. However, the same is not necessarily true for bendamustine/rituximab (BR); the CLL10 study showed a significantly greater PFS with FCR, compared with that seen with BR.
Unmutated patients in that study had lower PFS, but the outcomes were still better with FCR than with BR, he said.
Studies of novel agents, including ibrutinib and idelalisib, suggest they may have particular benefit in higher-risk patients.
Ibrutinib was shown in a phase 2 study to be of benefit regardless of IGHV status, and this was replicated in the first-line RESONATE 2 study, which compared ibrutinib with chlorambucil and showed it had better PFS than that seen in unmutated patients treated with FCR in other studies, said Dr. Barr, the first author on that study.
“So you can see how the treatment paradigms are starting to evolve. It does look like ... comparing across trials, that ibrutinib leads to better remission durations, compared to chemoimmunotherapy, so far,” he said.
Ibrutinib has also been shown to be of benefit for patients with del(17p). A single-arm phase 2 study showed 79% PFS in relapsed, high-risk patients, which is much better than has been seen with chemoimmunotherapy, he noted.
“Venetoclax is also a very good option for this patient population in the relapse setting,” he said, adding that the PFS with venetoclax has been shown to be very similar to that with ibrutinib.
Similarly, idelalisib has been shown to provide comparable benefit in relapsed/refractory CLL patients, with and without del(17p)/TP53 mutation, he said.
Dr. Barr is a consultant for Pharmacyclics, AbbVie, Celgene, Gilead, Infinity, Novartis, and Seattle Genetics and has received research funding from Pharmacyclics and AbbVie.
CHICAGO – said Paul M. Barr, MD.
He described a patient who had been observed for 7 years when he began to complain of increasing fatigue and lost work time. A complete blood count (CBC) showed thrombocytopenia.
The recently updated International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines state that assessments before treatment in this type of patient should include history and physical, evaluation of infectious disease status, and routine laboratory testing – including CBC and differential, chemistry, serum immunoglobulin, and direct antiglobulin test.
“Bone marrow biopsies and [computed tomography] scans are listed as ‘not necessarily required,’ ” Dr. Barr, medical director of the Clinical Trials Office for Wilmot Cancer Institute at the University of Rochester (N.Y.) said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
He added that he opts for CT scans prior to therapy “to understand the patient’s disease burden and potentially to compare to later” and that bone marrow biopsy is “still very reasonable” for understanding the source of a patient’s cytopenias.
“Is it marrow failure or [immune thrombocytopenia]? Could the patient have [myelodysplastic syndrome]? All important considerations,” he said.
Positron emission tomography (PET) scans, however, are only considered when there is concern about transformation, he noted.
Predictive tests that should be conducted before initiating therapy, and that could help in guiding therapy decisions, include TP53 mutation testing and immunoglobulin heavy chain variable region gene (IGHV) mutational status testing (although this doesn’t need to be repeated if it was done at diagnosis because mutational status doesn’t change). Another helpful test is molecular cytogenetics using fluorescence in situ hybridization (FISH) for del(13q), del(17p), and trisomy 12 in peripheral blood lymphocytes. This should be repeated even it was done at diagnosis because patients can acquire additional molecular aberrations over time, Dr. Barr said.
Among the data that justify this advice for predictive testing are studies showing the curative potential of fludarabine/cyclophosphamide/rituximab (FCR) in mutated IGHV CLL, the progression-free survival (PFS) benefits of ibrutinib for patients with del(17p), and the activity of idelalisib in relapsed/refractory CLL patients, including those with TP53 dysfunction.
“The IGHV mutation status is useful to know what to expect from chemoimmunotherapy over time,” Dr. Barr said, explaining that several analyses demonstrate that patients with mutated IGHV genes (patients with low-risk disease) respond exceptionally well to chemoimmunotherapy, especially FCR.
In fact, studies, including a 2016 study by Philip A. Thompson and his colleagues and another by Kirsten Fischer and her colleagues, show that nearly 60% of patients with IGHV mutation remain in remission 10 years after FCR treatment, he said. However, the same is not necessarily true for bendamustine/rituximab (BR); the CLL10 study showed a significantly greater PFS with FCR, compared with that seen with BR.
Unmutated patients in that study had lower PFS, but the outcomes were still better with FCR than with BR, he said.
Studies of novel agents, including ibrutinib and idelalisib, suggest they may have particular benefit in higher-risk patients.
Ibrutinib was shown in a phase 2 study to be of benefit regardless of IGHV status, and this was replicated in the first-line RESONATE 2 study, which compared ibrutinib with chlorambucil and showed it had better PFS than that seen in unmutated patients treated with FCR in other studies, said Dr. Barr, the first author on that study.
“So you can see how the treatment paradigms are starting to evolve. It does look like ... comparing across trials, that ibrutinib leads to better remission durations, compared to chemoimmunotherapy, so far,” he said.
Ibrutinib has also been shown to be of benefit for patients with del(17p). A single-arm phase 2 study showed 79% PFS in relapsed, high-risk patients, which is much better than has been seen with chemoimmunotherapy, he noted.
“Venetoclax is also a very good option for this patient population in the relapse setting,” he said, adding that the PFS with venetoclax has been shown to be very similar to that with ibrutinib.
Similarly, idelalisib has been shown to provide comparable benefit in relapsed/refractory CLL patients, with and without del(17p)/TP53 mutation, he said.
Dr. Barr is a consultant for Pharmacyclics, AbbVie, Celgene, Gilead, Infinity, Novartis, and Seattle Genetics and has received research funding from Pharmacyclics and AbbVie.
EXPERT ANALYSIS FROM MHM 2018
CLL: The initial work-up
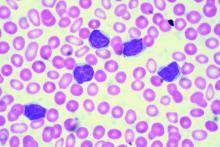
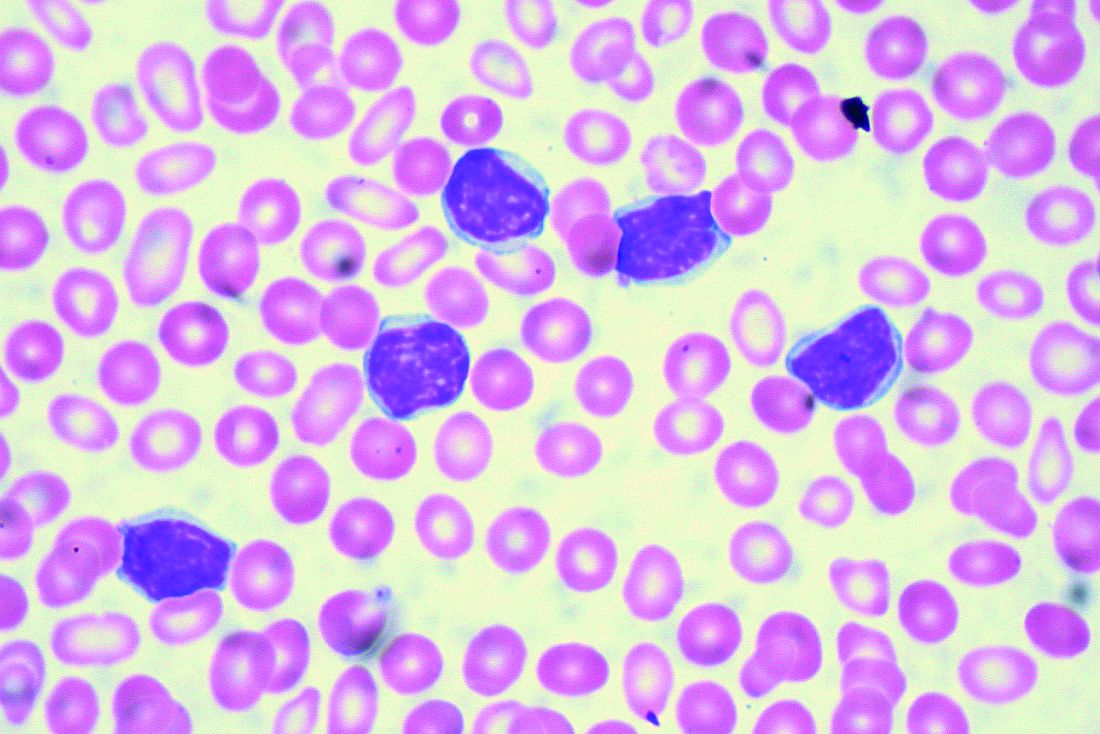
CHICAGO – A 50-year old otherwise healthy man was found on routine history and physical to have lymphocytosis and was referred for additional work-up. He denied recent infection, had no lymphadenopathy, organomegaly, or rash or other concerning skin lesions. A complete blood count showed a white cell count of 23 x 109/Land absolute lymphocyte count of 19 x 109/L and normal hemoglobin and platelets.
Based on recently updated International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines, additional work-up for this patient might include peripheral smear and flow cytometry, according to Paul Barr, MD.
“A peripheral smear is still useful in this day and age just to ensure that a patient has a typical look under the microscope. We expect to see small mature lymphocytes, smudge cells, and perhaps a smaller number of prolymphocytes. But to mark CLL based on flow cytometry we need to see greater than 5 x 109/L clonal B lymphocytes in the peripheral blood sustained over time,” Dr. Barr, medical director of the clinical trials office for Wilmot Cancer Institute at the University of Rochester (N.Y.), said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
A nuance in the iwCLL guidelines is that CLL also can be defined by a cytopenia caused by a typical marrow infiltrate, regardless of the number of circulating B lymphocytes, he noted.
Immunophenotype
As for CLL immunophenotype, the cells are CD5- and CD23-positive, and additional B cell markers like CD20 are “often dim, and – understandably – the cells are light-chain restricted,” he said.
A subtle difference between the World Health Organization classification of CLL and the iwCLL guidelines is that, by the former, patients can have “a somewhat atypical immunophenotype.”
“So our pathologists, I like to think, use a commonsense approach where, if there are very minor differences, they will still call it CLL, but not if there are major differences in the immunophenotype,” Dr. Barr said.
Patients with lymphadenopathy, without the critical threshold of circulating B lymphocytes, are considered to have small lymphocytic lymphoma (SLL).
“In this day and age we treat CLL and SLL relatively similarly, however, monoclonal B lymphocytosis (MBL) is the precursor lesion to CLL where we see less than 5 x 109/L of circulating B lymphocytes and an absence of adenopathy and disease-related cytopenias,” he noted.
Staging
It is still common practice to stage patients given the prognostic value of staging and given that treatment is provided in advanced disease, Dr. Barr said.
“This is simple, easy to apply, applicable worldwide, and only requires laboratory testing and a physical exam,” he said.
The stages include:
- Stage 0: Lymphocytosis, peripheral lymphocyte count greater than 15,000/mcL and greater than 40% lymphocytes in bone marrow (low-risk disease status).
- Stage I: Stage 0 disease plus enlarged lymph nodes (intermediate-risk disease status).
- Stage II: Stage 0-I disease with splenomegaly and/or hepatomegaly (intermediate-risk disease status).
- Stage III: Stage 0-II disease with hemoglobin less than 11g/dL or hematocrit less than 33% (high-risk disease status).
- Stage IV: Stage 0-III disease with platelet count less than 100,000/mcL (high-risk disease status).
Prognostic testing
Once a patient is diagnosed with CLL, as was the case with the 50-year-old patient Dr. Barr described, a number of tests can be considered to assess prognosis.
There’s no “perfect answer” when it comes to which tests are considered a reasonable standard of care, he noted.
“I would typically perform [immunoglobulin variable heavy-chain gene] mutation testing, a [fluorescence in situ hybridization] panel, and TP53 mutation testing,” he said.
Scoring systems such as CLL-IPI, which combine prognostic factors to divide patients into various risk categories, can be useful.
For example, such systems may identify high-risk patients who might be appropriate candidates for clinical trials, or low-risk patients who could be expected to do well over time despite having advanced stage disease, he explained.
“I do think it’s a useful process to go through to understand a patient’s risk over time,” he added.
However, treatment for CLL still is not based on molecular aberrations/prognostic features. In fact, the treatment indications according to the updated iwCLL guidelines remain exactly the same, he said.
Therefore, the case of the 50-year-old man described earlier would be observed as long as he remained asymptomatic.
Dr. Barr is a consultant for Pharmacyclics, AbbVie, Celgene, Gilead, Infinity, Novartis, and Seattle Genetics and has received research funding from Pharmacyclics and AbbVie.
CHICAGO – A 50-year old otherwise healthy man was found on routine history and physical to have lymphocytosis and was referred for additional work-up. He denied recent infection, had no lymphadenopathy, organomegaly, or rash or other concerning skin lesions. A complete blood count showed a white cell count of 23 x 109/Land absolute lymphocyte count of 19 x 109/L and normal hemoglobin and platelets.
Based on recently updated International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines, additional work-up for this patient might include peripheral smear and flow cytometry, according to Paul Barr, MD.
“A peripheral smear is still useful in this day and age just to ensure that a patient has a typical look under the microscope. We expect to see small mature lymphocytes, smudge cells, and perhaps a smaller number of prolymphocytes. But to mark CLL based on flow cytometry we need to see greater than 5 x 109/L clonal B lymphocytes in the peripheral blood sustained over time,” Dr. Barr, medical director of the clinical trials office for Wilmot Cancer Institute at the University of Rochester (N.Y.), said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
A nuance in the iwCLL guidelines is that CLL also can be defined by a cytopenia caused by a typical marrow infiltrate, regardless of the number of circulating B lymphocytes, he noted.
Immunophenotype
As for CLL immunophenotype, the cells are CD5- and CD23-positive, and additional B cell markers like CD20 are “often dim, and – understandably – the cells are light-chain restricted,” he said.
A subtle difference between the World Health Organization classification of CLL and the iwCLL guidelines is that, by the former, patients can have “a somewhat atypical immunophenotype.”
“So our pathologists, I like to think, use a commonsense approach where, if there are very minor differences, they will still call it CLL, but not if there are major differences in the immunophenotype,” Dr. Barr said.
Patients with lymphadenopathy, without the critical threshold of circulating B lymphocytes, are considered to have small lymphocytic lymphoma (SLL).
“In this day and age we treat CLL and SLL relatively similarly, however, monoclonal B lymphocytosis (MBL) is the precursor lesion to CLL where we see less than 5 x 109/L of circulating B lymphocytes and an absence of adenopathy and disease-related cytopenias,” he noted.
Staging
It is still common practice to stage patients given the prognostic value of staging and given that treatment is provided in advanced disease, Dr. Barr said.
“This is simple, easy to apply, applicable worldwide, and only requires laboratory testing and a physical exam,” he said.
The stages include:
- Stage 0: Lymphocytosis, peripheral lymphocyte count greater than 15,000/mcL and greater than 40% lymphocytes in bone marrow (low-risk disease status).
- Stage I: Stage 0 disease plus enlarged lymph nodes (intermediate-risk disease status).
- Stage II: Stage 0-I disease with splenomegaly and/or hepatomegaly (intermediate-risk disease status).
- Stage III: Stage 0-II disease with hemoglobin less than 11g/dL or hematocrit less than 33% (high-risk disease status).
- Stage IV: Stage 0-III disease with platelet count less than 100,000/mcL (high-risk disease status).
Prognostic testing
Once a patient is diagnosed with CLL, as was the case with the 50-year-old patient Dr. Barr described, a number of tests can be considered to assess prognosis.
There’s no “perfect answer” when it comes to which tests are considered a reasonable standard of care, he noted.
“I would typically perform [immunoglobulin variable heavy-chain gene] mutation testing, a [fluorescence in situ hybridization] panel, and TP53 mutation testing,” he said.
Scoring systems such as CLL-IPI, which combine prognostic factors to divide patients into various risk categories, can be useful.
For example, such systems may identify high-risk patients who might be appropriate candidates for clinical trials, or low-risk patients who could be expected to do well over time despite having advanced stage disease, he explained.
“I do think it’s a useful process to go through to understand a patient’s risk over time,” he added.
However, treatment for CLL still is not based on molecular aberrations/prognostic features. In fact, the treatment indications according to the updated iwCLL guidelines remain exactly the same, he said.
Therefore, the case of the 50-year-old man described earlier would be observed as long as he remained asymptomatic.
Dr. Barr is a consultant for Pharmacyclics, AbbVie, Celgene, Gilead, Infinity, Novartis, and Seattle Genetics and has received research funding from Pharmacyclics and AbbVie.
CHICAGO – A 50-year old otherwise healthy man was found on routine history and physical to have lymphocytosis and was referred for additional work-up. He denied recent infection, had no lymphadenopathy, organomegaly, or rash or other concerning skin lesions. A complete blood count showed a white cell count of 23 x 109/Land absolute lymphocyte count of 19 x 109/L and normal hemoglobin and platelets.
Based on recently updated International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines, additional work-up for this patient might include peripheral smear and flow cytometry, according to Paul Barr, MD.
“A peripheral smear is still useful in this day and age just to ensure that a patient has a typical look under the microscope. We expect to see small mature lymphocytes, smudge cells, and perhaps a smaller number of prolymphocytes. But to mark CLL based on flow cytometry we need to see greater than 5 x 109/L clonal B lymphocytes in the peripheral blood sustained over time,” Dr. Barr, medical director of the clinical trials office for Wilmot Cancer Institute at the University of Rochester (N.Y.), said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
A nuance in the iwCLL guidelines is that CLL also can be defined by a cytopenia caused by a typical marrow infiltrate, regardless of the number of circulating B lymphocytes, he noted.
Immunophenotype
As for CLL immunophenotype, the cells are CD5- and CD23-positive, and additional B cell markers like CD20 are “often dim, and – understandably – the cells are light-chain restricted,” he said.
A subtle difference between the World Health Organization classification of CLL and the iwCLL guidelines is that, by the former, patients can have “a somewhat atypical immunophenotype.”
“So our pathologists, I like to think, use a commonsense approach where, if there are very minor differences, they will still call it CLL, but not if there are major differences in the immunophenotype,” Dr. Barr said.
Patients with lymphadenopathy, without the critical threshold of circulating B lymphocytes, are considered to have small lymphocytic lymphoma (SLL).
“In this day and age we treat CLL and SLL relatively similarly, however, monoclonal B lymphocytosis (MBL) is the precursor lesion to CLL where we see less than 5 x 109/L of circulating B lymphocytes and an absence of adenopathy and disease-related cytopenias,” he noted.
Staging
It is still common practice to stage patients given the prognostic value of staging and given that treatment is provided in advanced disease, Dr. Barr said.
“This is simple, easy to apply, applicable worldwide, and only requires laboratory testing and a physical exam,” he said.
The stages include:
- Stage 0: Lymphocytosis, peripheral lymphocyte count greater than 15,000/mcL and greater than 40% lymphocytes in bone marrow (low-risk disease status).
- Stage I: Stage 0 disease plus enlarged lymph nodes (intermediate-risk disease status).
- Stage II: Stage 0-I disease with splenomegaly and/or hepatomegaly (intermediate-risk disease status).
- Stage III: Stage 0-II disease with hemoglobin less than 11g/dL or hematocrit less than 33% (high-risk disease status).
- Stage IV: Stage 0-III disease with platelet count less than 100,000/mcL (high-risk disease status).
Prognostic testing
Once a patient is diagnosed with CLL, as was the case with the 50-year-old patient Dr. Barr described, a number of tests can be considered to assess prognosis.
There’s no “perfect answer” when it comes to which tests are considered a reasonable standard of care, he noted.
“I would typically perform [immunoglobulin variable heavy-chain gene] mutation testing, a [fluorescence in situ hybridization] panel, and TP53 mutation testing,” he said.
Scoring systems such as CLL-IPI, which combine prognostic factors to divide patients into various risk categories, can be useful.
For example, such systems may identify high-risk patients who might be appropriate candidates for clinical trials, or low-risk patients who could be expected to do well over time despite having advanced stage disease, he explained.
“I do think it’s a useful process to go through to understand a patient’s risk over time,” he added.
However, treatment for CLL still is not based on molecular aberrations/prognostic features. In fact, the treatment indications according to the updated iwCLL guidelines remain exactly the same, he said.
Therefore, the case of the 50-year-old man described earlier would be observed as long as he remained asymptomatic.
Dr. Barr is a consultant for Pharmacyclics, AbbVie, Celgene, Gilead, Infinity, Novartis, and Seattle Genetics and has received research funding from Pharmacyclics and AbbVie.
EXPERT ANALYSIS FROM MHM 2018
Variant not linked to CLL in Southeast Europe
DUBROVNIK, CROATIA – New research suggests there is no association between the PTPN22 R620W polymorphism and chronic lymphocytic leukemia (CLL) or autoimmune hematologic disorders in patients from the Republic of Macedonia.
Past studies have shown an association between the PTPN22 R620W variant and both CLL and autoimmune diseases in patients from Northwest Europe. However, a new study of Macedonian patients suggests there is no association between the variant and CLL, autoimmune hemolytic anemia (AIHA), or idiopathic thrombocytopenic purpura (ITP) for patients from Southeast Europe.
Irina Panovska-Stavridis, PhD, of Ss. Cyril and Methodius University in Skopje, Republic of Macedonia, and her colleagues presented this finding at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
“A lot of data from the literature suggests [the PTPN22 R620W variant ] has a role in developing multiple immune diseases, but it is validated just in patients from Northwest Europe,” Dr. Panovska-Stavridis noted.
She and her colleagues decided to assess the frequency of the PTPN22 R620W variant (C1858T, rs2476601) in individuals from Southeast Europe, particularly the Republic of Macedonia.
The researchers evaluated 320 patients – 168 with CLL, 66 with AIHA, and 86 with ITP – and 182 age- and sex-matched control subjects with no history of malignant or autoimmune disease.
The team found a similar frequency of the minor T allele and genotype distribution in control subjects and patients. For example, minor T allele was 0.107 in CLL, 0.067 in AIHA, 0.036 in ITP, and 0.05 in controls. Similarly, the frequency of the CC genotype was 0.809 in CLL, 0.166 in AIHA, 0.023 in ITP, and 0.901 in controls.
Dr. Panovska-Stavridis said these results suggest the PTPN22 R620W variant is not a risk factor for the development of CLL, AIHA, or ITP in patients from Southeast Europe.
She also said the results suggest the influence of the variant on lymphocytic homeostasis is affected by certain genetic and environmental factors, and the development of CLL and autoimmune diseases is influenced by race/ethnicity-based variations in the germline composition of the IGHV locus in correlation with environmental factors.
Dr. Panovska-Stavridis did not declare any conflicts of interest.
The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Associates, which is owned by the parent company of this news organization.
DUBROVNIK, CROATIA – New research suggests there is no association between the PTPN22 R620W polymorphism and chronic lymphocytic leukemia (CLL) or autoimmune hematologic disorders in patients from the Republic of Macedonia.
Past studies have shown an association between the PTPN22 R620W variant and both CLL and autoimmune diseases in patients from Northwest Europe. However, a new study of Macedonian patients suggests there is no association between the variant and CLL, autoimmune hemolytic anemia (AIHA), or idiopathic thrombocytopenic purpura (ITP) for patients from Southeast Europe.
Irina Panovska-Stavridis, PhD, of Ss. Cyril and Methodius University in Skopje, Republic of Macedonia, and her colleagues presented this finding at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
“A lot of data from the literature suggests [the PTPN22 R620W variant ] has a role in developing multiple immune diseases, but it is validated just in patients from Northwest Europe,” Dr. Panovska-Stavridis noted.
She and her colleagues decided to assess the frequency of the PTPN22 R620W variant (C1858T, rs2476601) in individuals from Southeast Europe, particularly the Republic of Macedonia.
The researchers evaluated 320 patients – 168 with CLL, 66 with AIHA, and 86 with ITP – and 182 age- and sex-matched control subjects with no history of malignant or autoimmune disease.
The team found a similar frequency of the minor T allele and genotype distribution in control subjects and patients. For example, minor T allele was 0.107 in CLL, 0.067 in AIHA, 0.036 in ITP, and 0.05 in controls. Similarly, the frequency of the CC genotype was 0.809 in CLL, 0.166 in AIHA, 0.023 in ITP, and 0.901 in controls.
Dr. Panovska-Stavridis said these results suggest the PTPN22 R620W variant is not a risk factor for the development of CLL, AIHA, or ITP in patients from Southeast Europe.
She also said the results suggest the influence of the variant on lymphocytic homeostasis is affected by certain genetic and environmental factors, and the development of CLL and autoimmune diseases is influenced by race/ethnicity-based variations in the germline composition of the IGHV locus in correlation with environmental factors.
Dr. Panovska-Stavridis did not declare any conflicts of interest.
The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Associates, which is owned by the parent company of this news organization.
DUBROVNIK, CROATIA – New research suggests there is no association between the PTPN22 R620W polymorphism and chronic lymphocytic leukemia (CLL) or autoimmune hematologic disorders in patients from the Republic of Macedonia.
Past studies have shown an association between the PTPN22 R620W variant and both CLL and autoimmune diseases in patients from Northwest Europe. However, a new study of Macedonian patients suggests there is no association between the variant and CLL, autoimmune hemolytic anemia (AIHA), or idiopathic thrombocytopenic purpura (ITP) for patients from Southeast Europe.
Irina Panovska-Stavridis, PhD, of Ss. Cyril and Methodius University in Skopje, Republic of Macedonia, and her colleagues presented this finding at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
“A lot of data from the literature suggests [the PTPN22 R620W variant ] has a role in developing multiple immune diseases, but it is validated just in patients from Northwest Europe,” Dr. Panovska-Stavridis noted.
She and her colleagues decided to assess the frequency of the PTPN22 R620W variant (C1858T, rs2476601) in individuals from Southeast Europe, particularly the Republic of Macedonia.
The researchers evaluated 320 patients – 168 with CLL, 66 with AIHA, and 86 with ITP – and 182 age- and sex-matched control subjects with no history of malignant or autoimmune disease.
The team found a similar frequency of the minor T allele and genotype distribution in control subjects and patients. For example, minor T allele was 0.107 in CLL, 0.067 in AIHA, 0.036 in ITP, and 0.05 in controls. Similarly, the frequency of the CC genotype was 0.809 in CLL, 0.166 in AIHA, 0.023 in ITP, and 0.901 in controls.
Dr. Panovska-Stavridis said these results suggest the PTPN22 R620W variant is not a risk factor for the development of CLL, AIHA, or ITP in patients from Southeast Europe.
She also said the results suggest the influence of the variant on lymphocytic homeostasis is affected by certain genetic and environmental factors, and the development of CLL and autoimmune diseases is influenced by race/ethnicity-based variations in the germline composition of the IGHV locus in correlation with environmental factors.
Dr. Panovska-Stavridis did not declare any conflicts of interest.
The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Associates, which is owned by the parent company of this news organization.
REPORTING FROM LEUKEMIA AND LYMPHOMA 2018
Key clinical point:
Major finding: The frequency of minor T allele was 0.107 in patients with CLL, 0.067 in patients with autoimmune hemolytic anemia, 0.036 in patients with idiopathic thrombocytopenic purpura, and 0.05 in controls.
Study details: An analysis of the frequency of the PTPN22 R620W variant in 320 individuals from the Republic of Macedonia.
Disclosures: Dr. Panovska-Stavridis did not declare any conflicts of interest.
Sandoz halts pursuit of U.S. approval for rituximab biosimilar
.
Sandoz, a division of Novartis, was seeking Food and Drug Administration approval of GP2013 for all the same indications as the reference product – B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
GP2013 already is approved in the European Union and elsewhere.
The FDA had accepted the biologics license application (BLA) for GP2013 in September 2017. In May 2018, the agency issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in a statement.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by results from the ASSIST-FL trial, in which researchers compared GP2013 with the reference product (Lancet Haematol. 2017 Aug;4[8]:e350-61).
The phase 3 trial included adults with previously untreated, advanced-stage follicular lymphoma. Patients received eight cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% in the GP2013 arm and 88% in the rituximab arm.
.
Sandoz, a division of Novartis, was seeking Food and Drug Administration approval of GP2013 for all the same indications as the reference product – B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
GP2013 already is approved in the European Union and elsewhere.
The FDA had accepted the biologics license application (BLA) for GP2013 in September 2017. In May 2018, the agency issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in a statement.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by results from the ASSIST-FL trial, in which researchers compared GP2013 with the reference product (Lancet Haematol. 2017 Aug;4[8]:e350-61).
The phase 3 trial included adults with previously untreated, advanced-stage follicular lymphoma. Patients received eight cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% in the GP2013 arm and 88% in the rituximab arm.
.
Sandoz, a division of Novartis, was seeking Food and Drug Administration approval of GP2013 for all the same indications as the reference product – B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
GP2013 already is approved in the European Union and elsewhere.
The FDA had accepted the biologics license application (BLA) for GP2013 in September 2017. In May 2018, the agency issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in a statement.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by results from the ASSIST-FL trial, in which researchers compared GP2013 with the reference product (Lancet Haematol. 2017 Aug;4[8]:e350-61).
The phase 3 trial included adults with previously untreated, advanced-stage follicular lymphoma. Patients received eight cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% in the GP2013 arm and 88% in the rituximab arm.
Ibrutinib discontinuation harms survival in CLL
Discontinuing ibrutinib therapy because of disease progression was associated with worse survival, according to a real-world study of ibrutinib dosing in chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma patients.
Researchers at the University of Rochester Wilmot Cancer Institute in New York, who performed the single-center study, also found that optimal dosing early on in treatment has a significant impact on disease progression.
“Treating physicians need to be aware of these outcomes when initiating therapy on patients with high-risk CLL or lymphoma, as well as those with significant comorbidities or immune deficiencies,” AnnaLynn M. Williams. MS, and her colleagues reported in Clinical Lymphoma, Myeloma and Leukemia.
The researchers examined the impact of ibrutinib discontinuation and dose adherence on overall and progression-free survival in 170 patients with non-Hodgkin lymphoma and CLL treated with the drug at the Wilmot Cancer Institute between Jan. 1, 2014, and Dec. 1, 2016.
The study comprised 115 patients with CLL, 23 patients with Waldenstrom macroglobulinemia, 21 patients with mantle cell lymphoma, and 11 patients with other non-Hodgkin lymphomas. The median age of patients who started ibrutinib was 68 years, and the median treatment duration was 14.3 months. About a third of patients were taking ibrutinib as a first-line treatment.
Overall, 51 patients (30%) permanently discontinued ibrutinib during the study period, with more than half of the discontinuations stemming from adverse events or comorbidities. About 35% of the discontinuations were due to disease progression.
Median overall survival after discontinuation due to disease progression was 1.7 months. When patients discontinued for other reasons, median overall survival was not reached, compared with stopping for disease progression (P = .0008).
The researchers reported that among patients who discontinued for nonprogression reasons, 67% were alive after 1 year. Among CLL patients, 80% were alive after 1 year.
Among 20 patients who had a dose adherence of less than 80% in the first 8 weeks, the researchers found worse progression-free survival (P = .002) and overall survival (P = .021). Among CLL patients only, progression-free survival was significantly worse (P = .043) but overall survival was not (P = .816).
The study also included five patients who reduced their ibrutinib dose in the first 8 weeks – down to 280 mg in two patients, 140 mg in two patients, and 420 mg in one patient. Again, the researchers observed worse progression-free survival (P = .004) and overall survival (P = .014), compared with patients who maintained their dosing level.
Interrupting ibrutinib dosing had an impact on survival but not as much as discontinuation. Among 10 patients who interrupted therapy for more than a week and then restarted, progression-free survival was worse, compared with those who stayed on treatment continuously (P = .047), but overall survival was not significantly worse (P = .577).
“This would suggest that the ideal treatment strategy would be to recommend initiation of therapy at standard dosing and interruption as needed as directed in the [Food and Drug Administration] label,” the researchers wrote.
The study was funded by the National Cancer Institute and the Cadregari Endowment Fund. The researchers reported having no conflicts of interest.
SOURCE: Williams AM et al. Clin Lymphoma Myeloma Leuk. 2018 Oct 12. doi: 10.1016/j.clml.2018.10.005.
Discontinuing ibrutinib therapy because of disease progression was associated with worse survival, according to a real-world study of ibrutinib dosing in chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma patients.
Researchers at the University of Rochester Wilmot Cancer Institute in New York, who performed the single-center study, also found that optimal dosing early on in treatment has a significant impact on disease progression.
“Treating physicians need to be aware of these outcomes when initiating therapy on patients with high-risk CLL or lymphoma, as well as those with significant comorbidities or immune deficiencies,” AnnaLynn M. Williams. MS, and her colleagues reported in Clinical Lymphoma, Myeloma and Leukemia.
The researchers examined the impact of ibrutinib discontinuation and dose adherence on overall and progression-free survival in 170 patients with non-Hodgkin lymphoma and CLL treated with the drug at the Wilmot Cancer Institute between Jan. 1, 2014, and Dec. 1, 2016.
The study comprised 115 patients with CLL, 23 patients with Waldenstrom macroglobulinemia, 21 patients with mantle cell lymphoma, and 11 patients with other non-Hodgkin lymphomas. The median age of patients who started ibrutinib was 68 years, and the median treatment duration was 14.3 months. About a third of patients were taking ibrutinib as a first-line treatment.
Overall, 51 patients (30%) permanently discontinued ibrutinib during the study period, with more than half of the discontinuations stemming from adverse events or comorbidities. About 35% of the discontinuations were due to disease progression.
Median overall survival after discontinuation due to disease progression was 1.7 months. When patients discontinued for other reasons, median overall survival was not reached, compared with stopping for disease progression (P = .0008).
The researchers reported that among patients who discontinued for nonprogression reasons, 67% were alive after 1 year. Among CLL patients, 80% were alive after 1 year.
Among 20 patients who had a dose adherence of less than 80% in the first 8 weeks, the researchers found worse progression-free survival (P = .002) and overall survival (P = .021). Among CLL patients only, progression-free survival was significantly worse (P = .043) but overall survival was not (P = .816).
The study also included five patients who reduced their ibrutinib dose in the first 8 weeks – down to 280 mg in two patients, 140 mg in two patients, and 420 mg in one patient. Again, the researchers observed worse progression-free survival (P = .004) and overall survival (P = .014), compared with patients who maintained their dosing level.
Interrupting ibrutinib dosing had an impact on survival but not as much as discontinuation. Among 10 patients who interrupted therapy for more than a week and then restarted, progression-free survival was worse, compared with those who stayed on treatment continuously (P = .047), but overall survival was not significantly worse (P = .577).
“This would suggest that the ideal treatment strategy would be to recommend initiation of therapy at standard dosing and interruption as needed as directed in the [Food and Drug Administration] label,” the researchers wrote.
The study was funded by the National Cancer Institute and the Cadregari Endowment Fund. The researchers reported having no conflicts of interest.
SOURCE: Williams AM et al. Clin Lymphoma Myeloma Leuk. 2018 Oct 12. doi: 10.1016/j.clml.2018.10.005.
Discontinuing ibrutinib therapy because of disease progression was associated with worse survival, according to a real-world study of ibrutinib dosing in chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma patients.
Researchers at the University of Rochester Wilmot Cancer Institute in New York, who performed the single-center study, also found that optimal dosing early on in treatment has a significant impact on disease progression.
“Treating physicians need to be aware of these outcomes when initiating therapy on patients with high-risk CLL or lymphoma, as well as those with significant comorbidities or immune deficiencies,” AnnaLynn M. Williams. MS, and her colleagues reported in Clinical Lymphoma, Myeloma and Leukemia.
The researchers examined the impact of ibrutinib discontinuation and dose adherence on overall and progression-free survival in 170 patients with non-Hodgkin lymphoma and CLL treated with the drug at the Wilmot Cancer Institute between Jan. 1, 2014, and Dec. 1, 2016.
The study comprised 115 patients with CLL, 23 patients with Waldenstrom macroglobulinemia, 21 patients with mantle cell lymphoma, and 11 patients with other non-Hodgkin lymphomas. The median age of patients who started ibrutinib was 68 years, and the median treatment duration was 14.3 months. About a third of patients were taking ibrutinib as a first-line treatment.
Overall, 51 patients (30%) permanently discontinued ibrutinib during the study period, with more than half of the discontinuations stemming from adverse events or comorbidities. About 35% of the discontinuations were due to disease progression.
Median overall survival after discontinuation due to disease progression was 1.7 months. When patients discontinued for other reasons, median overall survival was not reached, compared with stopping for disease progression (P = .0008).
The researchers reported that among patients who discontinued for nonprogression reasons, 67% were alive after 1 year. Among CLL patients, 80% were alive after 1 year.
Among 20 patients who had a dose adherence of less than 80% in the first 8 weeks, the researchers found worse progression-free survival (P = .002) and overall survival (P = .021). Among CLL patients only, progression-free survival was significantly worse (P = .043) but overall survival was not (P = .816).
The study also included five patients who reduced their ibrutinib dose in the first 8 weeks – down to 280 mg in two patients, 140 mg in two patients, and 420 mg in one patient. Again, the researchers observed worse progression-free survival (P = .004) and overall survival (P = .014), compared with patients who maintained their dosing level.
Interrupting ibrutinib dosing had an impact on survival but not as much as discontinuation. Among 10 patients who interrupted therapy for more than a week and then restarted, progression-free survival was worse, compared with those who stayed on treatment continuously (P = .047), but overall survival was not significantly worse (P = .577).
“This would suggest that the ideal treatment strategy would be to recommend initiation of therapy at standard dosing and interruption as needed as directed in the [Food and Drug Administration] label,” the researchers wrote.
The study was funded by the National Cancer Institute and the Cadregari Endowment Fund. The researchers reported having no conflicts of interest.
SOURCE: Williams AM et al. Clin Lymphoma Myeloma Leuk. 2018 Oct 12. doi: 10.1016/j.clml.2018.10.005.
FROM CLINICAL LYMPHOMA, MYELOMA AND LEUKEMIA
Key clinical point:
Major finding: Median overall survival after discontinuation of ibrutinib due to disease progression was 1.7 months.
Study details: A single-institution study of 170 patients with CLL or non-Hodgkin lymphoma who were taking ibrutinib.
Disclosures: The study was funded by the National Cancer Institute and the Cadregari Endowment Fund. The researchers reported having no conflicts of interest.
Source: Williams AM et al. Clin Lymphoma Myeloma Leuk. 2018 Oct 12. doi: 10.1016/j.clml.2018.10.005.
Ibrutinib plus obinutuzumab gets priority review in CLL/SLL
The Food and Drug Administration has granted priority review to an anti-CD20, chemotherapy-free combination – ibrutinib plus obinutuzumab – for the frontline treatment of chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL).
The agency will review the combination in previously untreated adults.
Ibrutinib (Imbruvica) is already approved as a single agent for adults with CLL/SLL for all lines of therapy and in combination with bendamustine and rituximab. Obinutuzumab (Gazyva) has been approved for patients with previously untreated CLL, in combination with chlorambucil.
The current application, which is sponsored by Janssen and Pharmacyclics, is based on results from the phase 3 iLLUMINATE trial. Preliminary results announced by Janssen and Pharmacyclics showed that ibrutinib plus obinutuzumab had statistically significant better progression-free survival, compared with chlorambucil plus obinutuzumab, as assessed by an independent review committee.
Complete results from the trial will be presented at an upcoming medical meeting, according to the sponsors.
The Food and Drug Administration has granted priority review to an anti-CD20, chemotherapy-free combination – ibrutinib plus obinutuzumab – for the frontline treatment of chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL).
The agency will review the combination in previously untreated adults.
Ibrutinib (Imbruvica) is already approved as a single agent for adults with CLL/SLL for all lines of therapy and in combination with bendamustine and rituximab. Obinutuzumab (Gazyva) has been approved for patients with previously untreated CLL, in combination with chlorambucil.
The current application, which is sponsored by Janssen and Pharmacyclics, is based on results from the phase 3 iLLUMINATE trial. Preliminary results announced by Janssen and Pharmacyclics showed that ibrutinib plus obinutuzumab had statistically significant better progression-free survival, compared with chlorambucil plus obinutuzumab, as assessed by an independent review committee.
Complete results from the trial will be presented at an upcoming medical meeting, according to the sponsors.
The Food and Drug Administration has granted priority review to an anti-CD20, chemotherapy-free combination – ibrutinib plus obinutuzumab – for the frontline treatment of chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL).
The agency will review the combination in previously untreated adults.
Ibrutinib (Imbruvica) is already approved as a single agent for adults with CLL/SLL for all lines of therapy and in combination with bendamustine and rituximab. Obinutuzumab (Gazyva) has been approved for patients with previously untreated CLL, in combination with chlorambucil.
The current application, which is sponsored by Janssen and Pharmacyclics, is based on results from the phase 3 iLLUMINATE trial. Preliminary results announced by Janssen and Pharmacyclics showed that ibrutinib plus obinutuzumab had statistically significant better progression-free survival, compared with chlorambucil plus obinutuzumab, as assessed by an independent review committee.
Complete results from the trial will be presented at an upcoming medical meeting, according to the sponsors.
Some mutation testing can be useful at CLL diagnosis
CHICAGO – A number of mutation tests – including immunoglobulin heavy chain gene (IgVH), fluorescence in situ hybridization (FISH), and TP53 – provide useful prognostic information at the time of chronic lymphocytic leukemia (CLL) diagnosis, according to Paul M. Barr, MD.
“It’s understood that IgVH mutation status is certainly prognostic,” Dr. Barr, associate professor of hematology/oncology at the University of Rochester (N.Y.), said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
The B-cell receptor of the CLL cells uses IgVH genes that may or may not have undergone somatic mutations, with unmutated being defined as 98% or more sequence homology to germline.
“This is indicative of stronger signaling through the B-cell receptor and, as we all know, predicts for an inferior prognosis,” he explained, citing a study that demonstrated superior survival rates with mutated IgVH genes (Blood. 1999;94[6]:1840-7).
“It’s also well understood and accepted that we should perform a FISH panel; we should look for interphase cytogenetics based on FISH in our patients,” Dr. Barr said. “Having said that, we, as medical oncologists, do not do a very good job of using this testing appropriately. Patterns of care studies have suggested that a significant number of patients don’t get FISH testing at diagnosis or before first-line therapy.”
In fact, a typical interphase FISH panel identifies cytogenetic lesions, including del(17p), del(11q), del(13q), and trisomy 12 in more than 80% of CLL cases, with del(13q) being the most common.
Another marker that can be assessed in CLL patients and has maintained prognostic value across multiple analyses is serum beta-2 microglobulin, Dr. Barr noted.
TP53 sequencing is valuable as well and has been associated with outcomes similar to those seen in patients with del(17p), he said, citing data from a study that found similarly poor outcomes with TP53 mutations or deletions and del(17p), even when minor subclones are identified using next-generation sequencing (Blood. 2014;123:2139-47).
“One of the primary reasons for this is that the two aberrations go together. Most often, if you have del(17p) you’re also going to find a TP53 mutation, but in about 30% of patients or so, only one allele is affected, so this is why it’s still important to test for TP53 mutations when you’re looking for a 17p deletion,” he said.
Numerous other recurrent mutations in CLL have been associated with poor overall survival and/or progression-free survival, including SF3B1, ATM, NOTCH1, POT1, BIRC3, and NFKBIE.
“The gut instinct is that maybe we should start testing for all of these mutations now, but I would caution everybody that we still need further validation before we can apply these to the majority of patients,” Dr. Barr said. “We still don’t know exactly what to do with all of these mutations – when and how often we should test for them, if the novel agents are truly better – so while, again, they can predict for inferior outcomes, I would say these are not yet standard of care to be tested in all patients.”
It is likely, though, that new prognostic systems will evolve as more is learned about how to use these molecular aberrations. Attempts are already being made to incorporate novel mutations into a prognostic system. Dr. Barr pointed to a report that looked at the integration of mutations and cytogenetic lesions to improve the accuracy of survival prediction in CLL (Blood. 2013;121:1403-12).
“But this still requires prospective testing, especially in patients getting the novel agents,” he said.
Conventional karyotyping also has potential, though a limited role in this setting, he said, noting that it can be reliably performed with stimulation of CLL cells.
“We also know additional aberrations are prognostic and that a complex karyotype predicts for a very poor outcome,” he said. The International Workshop on CLL (iwCLL) guidelines, which were recently updated for the first time in a decade, state that further validation is needed.
“I think it’s potentially useful in a very young patient you are considering taking to transplant, but again, I agree with the stance that this is not something that should be performed in every patient across the board,” he said.
The tests currently recommended by iwCLL before CLL treatment include IgVH mutation status; FISH for del(13q), del(11q), del(17p), and trisomy 12 in peripheral blood lymphocytes; and TP53.
“Some folks... don’t check a lot of these markers at diagnosis, but wait for patients to require therapy, and that’s a reasonable way to practice,” Dr. Barr said, noting, however, that he prefers knowing patients’ risk up front – especially for those patients he will see just once before they are “managed closer to home for the majority of their course.
“But if you [wait], then knowing what to repeat later is important,” he added. Namely, the FISH and TP53 tests are worth repeating as patients can acquire additional molecular aberrations over time.
Dr. Barr reported serving as a consultant for Pharmacyclics, AbbVie, Celgene, Gilead Sciences, Infinity Pharmaceuticals, Novartis, and Seattle Genetics. He also reported receiving research funding from Pharmacyclics and AbbVie.
CHICAGO – A number of mutation tests – including immunoglobulin heavy chain gene (IgVH), fluorescence in situ hybridization (FISH), and TP53 – provide useful prognostic information at the time of chronic lymphocytic leukemia (CLL) diagnosis, according to Paul M. Barr, MD.
“It’s understood that IgVH mutation status is certainly prognostic,” Dr. Barr, associate professor of hematology/oncology at the University of Rochester (N.Y.), said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
The B-cell receptor of the CLL cells uses IgVH genes that may or may not have undergone somatic mutations, with unmutated being defined as 98% or more sequence homology to germline.
“This is indicative of stronger signaling through the B-cell receptor and, as we all know, predicts for an inferior prognosis,” he explained, citing a study that demonstrated superior survival rates with mutated IgVH genes (Blood. 1999;94[6]:1840-7).
“It’s also well understood and accepted that we should perform a FISH panel; we should look for interphase cytogenetics based on FISH in our patients,” Dr. Barr said. “Having said that, we, as medical oncologists, do not do a very good job of using this testing appropriately. Patterns of care studies have suggested that a significant number of patients don’t get FISH testing at diagnosis or before first-line therapy.”
In fact, a typical interphase FISH panel identifies cytogenetic lesions, including del(17p), del(11q), del(13q), and trisomy 12 in more than 80% of CLL cases, with del(13q) being the most common.
Another marker that can be assessed in CLL patients and has maintained prognostic value across multiple analyses is serum beta-2 microglobulin, Dr. Barr noted.
TP53 sequencing is valuable as well and has been associated with outcomes similar to those seen in patients with del(17p), he said, citing data from a study that found similarly poor outcomes with TP53 mutations or deletions and del(17p), even when minor subclones are identified using next-generation sequencing (Blood. 2014;123:2139-47).
“One of the primary reasons for this is that the two aberrations go together. Most often, if you have del(17p) you’re also going to find a TP53 mutation, but in about 30% of patients or so, only one allele is affected, so this is why it’s still important to test for TP53 mutations when you’re looking for a 17p deletion,” he said.
Numerous other recurrent mutations in CLL have been associated with poor overall survival and/or progression-free survival, including SF3B1, ATM, NOTCH1, POT1, BIRC3, and NFKBIE.
“The gut instinct is that maybe we should start testing for all of these mutations now, but I would caution everybody that we still need further validation before we can apply these to the majority of patients,” Dr. Barr said. “We still don’t know exactly what to do with all of these mutations – when and how often we should test for them, if the novel agents are truly better – so while, again, they can predict for inferior outcomes, I would say these are not yet standard of care to be tested in all patients.”
It is likely, though, that new prognostic systems will evolve as more is learned about how to use these molecular aberrations. Attempts are already being made to incorporate novel mutations into a prognostic system. Dr. Barr pointed to a report that looked at the integration of mutations and cytogenetic lesions to improve the accuracy of survival prediction in CLL (Blood. 2013;121:1403-12).
“But this still requires prospective testing, especially in patients getting the novel agents,” he said.
Conventional karyotyping also has potential, though a limited role in this setting, he said, noting that it can be reliably performed with stimulation of CLL cells.
“We also know additional aberrations are prognostic and that a complex karyotype predicts for a very poor outcome,” he said. The International Workshop on CLL (iwCLL) guidelines, which were recently updated for the first time in a decade, state that further validation is needed.
“I think it’s potentially useful in a very young patient you are considering taking to transplant, but again, I agree with the stance that this is not something that should be performed in every patient across the board,” he said.
The tests currently recommended by iwCLL before CLL treatment include IgVH mutation status; FISH for del(13q), del(11q), del(17p), and trisomy 12 in peripheral blood lymphocytes; and TP53.
“Some folks... don’t check a lot of these markers at diagnosis, but wait for patients to require therapy, and that’s a reasonable way to practice,” Dr. Barr said, noting, however, that he prefers knowing patients’ risk up front – especially for those patients he will see just once before they are “managed closer to home for the majority of their course.
“But if you [wait], then knowing what to repeat later is important,” he added. Namely, the FISH and TP53 tests are worth repeating as patients can acquire additional molecular aberrations over time.
Dr. Barr reported serving as a consultant for Pharmacyclics, AbbVie, Celgene, Gilead Sciences, Infinity Pharmaceuticals, Novartis, and Seattle Genetics. He also reported receiving research funding from Pharmacyclics and AbbVie.
CHICAGO – A number of mutation tests – including immunoglobulin heavy chain gene (IgVH), fluorescence in situ hybridization (FISH), and TP53 – provide useful prognostic information at the time of chronic lymphocytic leukemia (CLL) diagnosis, according to Paul M. Barr, MD.
“It’s understood that IgVH mutation status is certainly prognostic,” Dr. Barr, associate professor of hematology/oncology at the University of Rochester (N.Y.), said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
The B-cell receptor of the CLL cells uses IgVH genes that may or may not have undergone somatic mutations, with unmutated being defined as 98% or more sequence homology to germline.
“This is indicative of stronger signaling through the B-cell receptor and, as we all know, predicts for an inferior prognosis,” he explained, citing a study that demonstrated superior survival rates with mutated IgVH genes (Blood. 1999;94[6]:1840-7).
“It’s also well understood and accepted that we should perform a FISH panel; we should look for interphase cytogenetics based on FISH in our patients,” Dr. Barr said. “Having said that, we, as medical oncologists, do not do a very good job of using this testing appropriately. Patterns of care studies have suggested that a significant number of patients don’t get FISH testing at diagnosis or before first-line therapy.”
In fact, a typical interphase FISH panel identifies cytogenetic lesions, including del(17p), del(11q), del(13q), and trisomy 12 in more than 80% of CLL cases, with del(13q) being the most common.
Another marker that can be assessed in CLL patients and has maintained prognostic value across multiple analyses is serum beta-2 microglobulin, Dr. Barr noted.
TP53 sequencing is valuable as well and has been associated with outcomes similar to those seen in patients with del(17p), he said, citing data from a study that found similarly poor outcomes with TP53 mutations or deletions and del(17p), even when minor subclones are identified using next-generation sequencing (Blood. 2014;123:2139-47).
“One of the primary reasons for this is that the two aberrations go together. Most often, if you have del(17p) you’re also going to find a TP53 mutation, but in about 30% of patients or so, only one allele is affected, so this is why it’s still important to test for TP53 mutations when you’re looking for a 17p deletion,” he said.
Numerous other recurrent mutations in CLL have been associated with poor overall survival and/or progression-free survival, including SF3B1, ATM, NOTCH1, POT1, BIRC3, and NFKBIE.
“The gut instinct is that maybe we should start testing for all of these mutations now, but I would caution everybody that we still need further validation before we can apply these to the majority of patients,” Dr. Barr said. “We still don’t know exactly what to do with all of these mutations – when and how often we should test for them, if the novel agents are truly better – so while, again, they can predict for inferior outcomes, I would say these are not yet standard of care to be tested in all patients.”
It is likely, though, that new prognostic systems will evolve as more is learned about how to use these molecular aberrations. Attempts are already being made to incorporate novel mutations into a prognostic system. Dr. Barr pointed to a report that looked at the integration of mutations and cytogenetic lesions to improve the accuracy of survival prediction in CLL (Blood. 2013;121:1403-12).
“But this still requires prospective testing, especially in patients getting the novel agents,” he said.
Conventional karyotyping also has potential, though a limited role in this setting, he said, noting that it can be reliably performed with stimulation of CLL cells.
“We also know additional aberrations are prognostic and that a complex karyotype predicts for a very poor outcome,” he said. The International Workshop on CLL (iwCLL) guidelines, which were recently updated for the first time in a decade, state that further validation is needed.
“I think it’s potentially useful in a very young patient you are considering taking to transplant, but again, I agree with the stance that this is not something that should be performed in every patient across the board,” he said.
The tests currently recommended by iwCLL before CLL treatment include IgVH mutation status; FISH for del(13q), del(11q), del(17p), and trisomy 12 in peripheral blood lymphocytes; and TP53.
“Some folks... don’t check a lot of these markers at diagnosis, but wait for patients to require therapy, and that’s a reasonable way to practice,” Dr. Barr said, noting, however, that he prefers knowing patients’ risk up front – especially for those patients he will see just once before they are “managed closer to home for the majority of their course.
“But if you [wait], then knowing what to repeat later is important,” he added. Namely, the FISH and TP53 tests are worth repeating as patients can acquire additional molecular aberrations over time.
Dr. Barr reported serving as a consultant for Pharmacyclics, AbbVie, Celgene, Gilead Sciences, Infinity Pharmaceuticals, Novartis, and Seattle Genetics. He also reported receiving research funding from Pharmacyclics and AbbVie.
EXPERT ANALYSIS FROM MHM 2018
Novel options for treating hairy cell leukemia
NEW YORK – Ibrutinib, and now moxetumomab pasudotox, are two novel therapies that can be tried in patients with previously treated hairy cell leukemia, although data and experience with them are so far limited in this rare disease, experts said during a panel discussion at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
Since there are so few patients, data on the BTK inhibitor ibrutinib in hairy cell leukemia is largely “anecdotal,” said Andrew D. Zelenetz, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York.
The anti-CD22 monoclonal antibody moxetumomab pasudotox – approved for hairy cell leukemia in September – isn’t yet on the formulary at Memorial Sloan Kettering, Dr. Zelenetz added in a panel discussion of treatment options for a patient previously treated with purine analogueues and vemurafenib.
Between the two agents, moxetumomab pasudotox has more robust data in this disease, said John N. Allan, MD, of Weill Cornell Medicine, New York.
“I think if you can get access to the drug, that’s probably the best answer,” Dr. Allan said in the case discussion.
Hairy cell leukemia is an indolent B-cell lymphoma that makes up just 2% of all lymphoid leukemias, according to NCCN guidelines.
It is a chronic disease that requires long-term management, according to Dr. Allan.
First-line treatment is usually a purine analogue, either cladribine or pentostatin, and multiple treatments are possible as long as responses of greater than 2 years are achieved, he told attendees at the NCCN conference.
For relapses more than 2 years after first-line treatment, patients can be retreated with the same purine analogue, with or without rituximab, or can be switched to the alternative purine analogue, he said.
Vemurafenib, the BRAF inhibitor, is “surprisingly” effective in 90% of classic hairy cell leukemia patients with the BRAF V600E mutation, Dr. Allan added, though only about 40% of patients achieve complete response.
In discussing therapy options for a hairy cell leukemia patient previously treated with purine analogues and vemurafenib, Dr. Allan noted that the data behind ibrutinib includes case reports and early clinical investigations.
Several phase 1 studies with small numbers of patients show response rates “in the 50% range,” he said.
“This is an option,” he said. “It’s in the guidelines, and it’s something to consider.”
Moxetumomab pasudotox was recently approved for intravenous use in adults with relapsed or refractory hairy cell leukemia who have had at least two previous systemic treatments, including a purine nucleoside analogue. The CD22-directed cytotoxin is the first of its kind for treating patients with hairy cell leukemia, according to the Food and Drug Administration.
In a single-arm, open-label clinical trial including 80 patients with hairy cell leukemia who had previous treatment in line with that indication, 75% had a partial or complete response, of whom 30% had a durable complete response (CR), defined as maintaining hematologic remission for at least 180 days following CR.
Following the FDA’s approval of moxetumomab pasudotox, the NCCN updated its hairy cell leukemia clinical practice guidelines to include the drug as a category 2A recommendation for relapsed/refractory treatment. Other category 2A options in that setting include ibrutinib, vemurafenib with or without rituximab, or a clinical trial.
Along with that, NCCN guideline authors added a full page on special considerations for use of moxetumomab pasudotox. That includes advice on monitoring for capillary leak syndrome and hemolytic uremic syndrome, along with guidance on capillary leak syndrome grading and management by grade.
Dr. Zelenetz reported financial disclosures related to Adaptive Biotechnology, Amgen, AstraZeneca, Celgene, Genentech, Gilead, Hoffman La Roche, MEI Pharma, MorphoSys AG, Novartis, Pfizer, Pharmacyclics, Roche, and Verastem Oncology. Dr. Allan reported disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis, and Verastem Oncology.
NEW YORK – Ibrutinib, and now moxetumomab pasudotox, are two novel therapies that can be tried in patients with previously treated hairy cell leukemia, although data and experience with them are so far limited in this rare disease, experts said during a panel discussion at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
Since there are so few patients, data on the BTK inhibitor ibrutinib in hairy cell leukemia is largely “anecdotal,” said Andrew D. Zelenetz, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York.
The anti-CD22 monoclonal antibody moxetumomab pasudotox – approved for hairy cell leukemia in September – isn’t yet on the formulary at Memorial Sloan Kettering, Dr. Zelenetz added in a panel discussion of treatment options for a patient previously treated with purine analogueues and vemurafenib.
Between the two agents, moxetumomab pasudotox has more robust data in this disease, said John N. Allan, MD, of Weill Cornell Medicine, New York.
“I think if you can get access to the drug, that’s probably the best answer,” Dr. Allan said in the case discussion.
Hairy cell leukemia is an indolent B-cell lymphoma that makes up just 2% of all lymphoid leukemias, according to NCCN guidelines.
It is a chronic disease that requires long-term management, according to Dr. Allan.
First-line treatment is usually a purine analogue, either cladribine or pentostatin, and multiple treatments are possible as long as responses of greater than 2 years are achieved, he told attendees at the NCCN conference.
For relapses more than 2 years after first-line treatment, patients can be retreated with the same purine analogue, with or without rituximab, or can be switched to the alternative purine analogue, he said.
Vemurafenib, the BRAF inhibitor, is “surprisingly” effective in 90% of classic hairy cell leukemia patients with the BRAF V600E mutation, Dr. Allan added, though only about 40% of patients achieve complete response.
In discussing therapy options for a hairy cell leukemia patient previously treated with purine analogues and vemurafenib, Dr. Allan noted that the data behind ibrutinib includes case reports and early clinical investigations.
Several phase 1 studies with small numbers of patients show response rates “in the 50% range,” he said.
“This is an option,” he said. “It’s in the guidelines, and it’s something to consider.”
Moxetumomab pasudotox was recently approved for intravenous use in adults with relapsed or refractory hairy cell leukemia who have had at least two previous systemic treatments, including a purine nucleoside analogue. The CD22-directed cytotoxin is the first of its kind for treating patients with hairy cell leukemia, according to the Food and Drug Administration.
In a single-arm, open-label clinical trial including 80 patients with hairy cell leukemia who had previous treatment in line with that indication, 75% had a partial or complete response, of whom 30% had a durable complete response (CR), defined as maintaining hematologic remission for at least 180 days following CR.
Following the FDA’s approval of moxetumomab pasudotox, the NCCN updated its hairy cell leukemia clinical practice guidelines to include the drug as a category 2A recommendation for relapsed/refractory treatment. Other category 2A options in that setting include ibrutinib, vemurafenib with or without rituximab, or a clinical trial.
Along with that, NCCN guideline authors added a full page on special considerations for use of moxetumomab pasudotox. That includes advice on monitoring for capillary leak syndrome and hemolytic uremic syndrome, along with guidance on capillary leak syndrome grading and management by grade.
Dr. Zelenetz reported financial disclosures related to Adaptive Biotechnology, Amgen, AstraZeneca, Celgene, Genentech, Gilead, Hoffman La Roche, MEI Pharma, MorphoSys AG, Novartis, Pfizer, Pharmacyclics, Roche, and Verastem Oncology. Dr. Allan reported disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis, and Verastem Oncology.
NEW YORK – Ibrutinib, and now moxetumomab pasudotox, are two novel therapies that can be tried in patients with previously treated hairy cell leukemia, although data and experience with them are so far limited in this rare disease, experts said during a panel discussion at the National Comprehensive Cancer Network Hematologic Malignancies Annual Congress.
Since there are so few patients, data on the BTK inhibitor ibrutinib in hairy cell leukemia is largely “anecdotal,” said Andrew D. Zelenetz, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York.
The anti-CD22 monoclonal antibody moxetumomab pasudotox – approved for hairy cell leukemia in September – isn’t yet on the formulary at Memorial Sloan Kettering, Dr. Zelenetz added in a panel discussion of treatment options for a patient previously treated with purine analogueues and vemurafenib.
Between the two agents, moxetumomab pasudotox has more robust data in this disease, said John N. Allan, MD, of Weill Cornell Medicine, New York.
“I think if you can get access to the drug, that’s probably the best answer,” Dr. Allan said in the case discussion.
Hairy cell leukemia is an indolent B-cell lymphoma that makes up just 2% of all lymphoid leukemias, according to NCCN guidelines.
It is a chronic disease that requires long-term management, according to Dr. Allan.
First-line treatment is usually a purine analogue, either cladribine or pentostatin, and multiple treatments are possible as long as responses of greater than 2 years are achieved, he told attendees at the NCCN conference.
For relapses more than 2 years after first-line treatment, patients can be retreated with the same purine analogue, with or without rituximab, or can be switched to the alternative purine analogue, he said.
Vemurafenib, the BRAF inhibitor, is “surprisingly” effective in 90% of classic hairy cell leukemia patients with the BRAF V600E mutation, Dr. Allan added, though only about 40% of patients achieve complete response.
In discussing therapy options for a hairy cell leukemia patient previously treated with purine analogues and vemurafenib, Dr. Allan noted that the data behind ibrutinib includes case reports and early clinical investigations.
Several phase 1 studies with small numbers of patients show response rates “in the 50% range,” he said.
“This is an option,” he said. “It’s in the guidelines, and it’s something to consider.”
Moxetumomab pasudotox was recently approved for intravenous use in adults with relapsed or refractory hairy cell leukemia who have had at least two previous systemic treatments, including a purine nucleoside analogue. The CD22-directed cytotoxin is the first of its kind for treating patients with hairy cell leukemia, according to the Food and Drug Administration.
In a single-arm, open-label clinical trial including 80 patients with hairy cell leukemia who had previous treatment in line with that indication, 75% had a partial or complete response, of whom 30% had a durable complete response (CR), defined as maintaining hematologic remission for at least 180 days following CR.
Following the FDA’s approval of moxetumomab pasudotox, the NCCN updated its hairy cell leukemia clinical practice guidelines to include the drug as a category 2A recommendation for relapsed/refractory treatment. Other category 2A options in that setting include ibrutinib, vemurafenib with or without rituximab, or a clinical trial.
Along with that, NCCN guideline authors added a full page on special considerations for use of moxetumomab pasudotox. That includes advice on monitoring for capillary leak syndrome and hemolytic uremic syndrome, along with guidance on capillary leak syndrome grading and management by grade.
Dr. Zelenetz reported financial disclosures related to Adaptive Biotechnology, Amgen, AstraZeneca, Celgene, Genentech, Gilead, Hoffman La Roche, MEI Pharma, MorphoSys AG, Novartis, Pfizer, Pharmacyclics, Roche, and Verastem Oncology. Dr. Allan reported disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis, and Verastem Oncology.
EXPERT ANALYSIS FROM NCCN HEMATOLOGIC MALIGNANCIES
Real-world clues for optimal sequencing of CLL novel agents
NEW YORK – Although optimal sequencing strategies in chronic lymphocytic leukemia are still unclear, real-world data suggest an alternate kinase inhibitor or venetoclax is the best approach for a patient who has received ibrutinib or idelalisib, according to John N. Allan, MD, of Cornell University, New York.
“I think for the most part, there’s enough evidence,” Dr. Allan said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
“If you had one to two lines of therapy, it still favors the novel agents rather than the chemotherapy arms in all these studies,” said Dr. Allan, referring to some of the pivotal trials supporting approval of novel agents in chronic lymphocytic leukemia (CLL). “The earlier we get to these drugs, I believe, the better.”
While venetoclax after ibrutinib is supported by multiple studies, “vice versa is unknown, but there’s seemingly no reason to think it wouldn’t work – different mechanisms of actions, different pathways,” Dr. Allan said.
What is clear, he added, is that retreating those patients with chemoimmunotherapy is not optimal.
In support of that, he cited a multicenter retrospective analysis, which is believed to be the largest real-world experience to date of novel agents in CLL looking at post–kinase inhibitor salvage strategies (Ann Oncol. 2017 May 1;28[5]:1050-6).
Using an alternate kinase inhibitor or venetoclax resulted in superior progression-free survival versus chemoimmunotherapy at the time of initial kinase inhibitor failure in that study, which looked at treatment strategies and outcomes for 683 patients.
Ibrutinib appeared to be superior to idelalisib as a first kinase inhibitor, with significantly better progression-free survival in both frontline and relapsed/refractory settings, and in both complex karyotype and del17p patients, according to the report. Additionally, the response rate to venetoclax seemed superior to that of idelalisib in patients who discontinued ibrutinib because of progression or toxicity.
All of that supports the need for trials to test various sequencing strategies and establish clear treatment algorithms, according to Dr. Allan. “Optimal sequencing is unknown, but real-world data gives us some idea.”
For relapsed/refractory patients, ibrutinib, idelalisib, and venetoclax all have lengthened responses, improved survival, and are approved by the Food and Drug Administration, he added, noting that the toxicity profiles vary and must be understood when dosing and prescribing these agents.
More novel treatments are on the way. On Sept. 24, just days after Dr. Allan’s NCCN presentation, the FDA granted approval to duvelisib for adults with relapsed or refractory CLL or small lymphocytic lymphoma following two or more previous lines of therapy.
Dr. Allan reported financial disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis Pharmaceuticals, and Verastem Oncology.
NEW YORK – Although optimal sequencing strategies in chronic lymphocytic leukemia are still unclear, real-world data suggest an alternate kinase inhibitor or venetoclax is the best approach for a patient who has received ibrutinib or idelalisib, according to John N. Allan, MD, of Cornell University, New York.
“I think for the most part, there’s enough evidence,” Dr. Allan said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
“If you had one to two lines of therapy, it still favors the novel agents rather than the chemotherapy arms in all these studies,” said Dr. Allan, referring to some of the pivotal trials supporting approval of novel agents in chronic lymphocytic leukemia (CLL). “The earlier we get to these drugs, I believe, the better.”
While venetoclax after ibrutinib is supported by multiple studies, “vice versa is unknown, but there’s seemingly no reason to think it wouldn’t work – different mechanisms of actions, different pathways,” Dr. Allan said.
What is clear, he added, is that retreating those patients with chemoimmunotherapy is not optimal.
In support of that, he cited a multicenter retrospective analysis, which is believed to be the largest real-world experience to date of novel agents in CLL looking at post–kinase inhibitor salvage strategies (Ann Oncol. 2017 May 1;28[5]:1050-6).
Using an alternate kinase inhibitor or venetoclax resulted in superior progression-free survival versus chemoimmunotherapy at the time of initial kinase inhibitor failure in that study, which looked at treatment strategies and outcomes for 683 patients.
Ibrutinib appeared to be superior to idelalisib as a first kinase inhibitor, with significantly better progression-free survival in both frontline and relapsed/refractory settings, and in both complex karyotype and del17p patients, according to the report. Additionally, the response rate to venetoclax seemed superior to that of idelalisib in patients who discontinued ibrutinib because of progression or toxicity.
All of that supports the need for trials to test various sequencing strategies and establish clear treatment algorithms, according to Dr. Allan. “Optimal sequencing is unknown, but real-world data gives us some idea.”
For relapsed/refractory patients, ibrutinib, idelalisib, and venetoclax all have lengthened responses, improved survival, and are approved by the Food and Drug Administration, he added, noting that the toxicity profiles vary and must be understood when dosing and prescribing these agents.
More novel treatments are on the way. On Sept. 24, just days after Dr. Allan’s NCCN presentation, the FDA granted approval to duvelisib for adults with relapsed or refractory CLL or small lymphocytic lymphoma following two or more previous lines of therapy.
Dr. Allan reported financial disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis Pharmaceuticals, and Verastem Oncology.
NEW YORK – Although optimal sequencing strategies in chronic lymphocytic leukemia are still unclear, real-world data suggest an alternate kinase inhibitor or venetoclax is the best approach for a patient who has received ibrutinib or idelalisib, according to John N. Allan, MD, of Cornell University, New York.
“I think for the most part, there’s enough evidence,” Dr. Allan said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
“If you had one to two lines of therapy, it still favors the novel agents rather than the chemotherapy arms in all these studies,” said Dr. Allan, referring to some of the pivotal trials supporting approval of novel agents in chronic lymphocytic leukemia (CLL). “The earlier we get to these drugs, I believe, the better.”
While venetoclax after ibrutinib is supported by multiple studies, “vice versa is unknown, but there’s seemingly no reason to think it wouldn’t work – different mechanisms of actions, different pathways,” Dr. Allan said.
What is clear, he added, is that retreating those patients with chemoimmunotherapy is not optimal.
In support of that, he cited a multicenter retrospective analysis, which is believed to be the largest real-world experience to date of novel agents in CLL looking at post–kinase inhibitor salvage strategies (Ann Oncol. 2017 May 1;28[5]:1050-6).
Using an alternate kinase inhibitor or venetoclax resulted in superior progression-free survival versus chemoimmunotherapy at the time of initial kinase inhibitor failure in that study, which looked at treatment strategies and outcomes for 683 patients.
Ibrutinib appeared to be superior to idelalisib as a first kinase inhibitor, with significantly better progression-free survival in both frontline and relapsed/refractory settings, and in both complex karyotype and del17p patients, according to the report. Additionally, the response rate to venetoclax seemed superior to that of idelalisib in patients who discontinued ibrutinib because of progression or toxicity.
All of that supports the need for trials to test various sequencing strategies and establish clear treatment algorithms, according to Dr. Allan. “Optimal sequencing is unknown, but real-world data gives us some idea.”
For relapsed/refractory patients, ibrutinib, idelalisib, and venetoclax all have lengthened responses, improved survival, and are approved by the Food and Drug Administration, he added, noting that the toxicity profiles vary and must be understood when dosing and prescribing these agents.
More novel treatments are on the way. On Sept. 24, just days after Dr. Allan’s NCCN presentation, the FDA granted approval to duvelisib for adults with relapsed or refractory CLL or small lymphocytic lymphoma following two or more previous lines of therapy.
Dr. Allan reported financial disclosures related to AbbVie, Acerta Pharma, Genentech, Pharmacyclics, Sunesis Pharmaceuticals, and Verastem Oncology.
EXPERT ANALYSIS FROM NCCN HEMATOLOGIC MALIGNANCIES CONGRESS