User login
Medical Tourism
David Dupray, a 60-year-old uninsured coffee shop owner from Bar Harbor, Maine, had been having left leg pain on ambulation for four years. His cardiologist recommended stent placement for left iliac artery stenosis. The estimated bill: approximately $35,000.
Unable to afford the procedure, Dupray began searching the Web for affordable medical care overseas. His physician suggested Thailand. Within days, Dupray had an appointment with a cardiologist halfway around the world—at Bumrungrad Hospital in Bangkok, Thailand. Dupray spent two days in the hotel-like hospital, had three stents placed in his leg arteries, and completed a cardiac stress test. The total bill: $18,000.
“I will never go to a hospital in the U.S.,” says Dupray, who represents a growing number of Americans searching for affordable healthcare in the global marketplace.
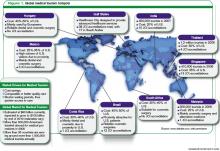
With rising U.S. healthcare costs and millions of Americans uninsured or underinsured, more American patients are seeking affordable, high-quality medical care abroad—known as “medical tourism.” In 2007, an estimated 750,000 Americans traveled abroad for medical care; the number is expected to increase to 6 million by the end of this year.1 On the flip side, only a little more than 400,000 nonresidents visited the U.S. in 2008 for the latest medical care.1 Globally, the medical tourism industry is estimated to grow into a $21 billion-a-year industry by 2012, with much of the growth expected from Western patients traveling overseas for affordable care.2
“As hospitalists, we have been seeing increasing numbers of patients going overseas for urgent and elective procedures, as it is a general perception the medical treatment overseas is less expensive,” says Joseph Ming Wah Li, MD, SFHM, director of hospital medicine at Beth Israel Deaconess Medical Center in Boston and SHM president-elect.
Physicians in U.S. hospitals encounter potential medical tourists all the time. Some are uninsured or underinsured. Some have insurance carriers that limit or exclude coverage for certain procedures and treatments. Even those with insurance sometimes struggle to pay deductibles, copays, and their costs after insurance has paid its part. Others are uncomfortable with the language barriers and cultural differences of U.S. hospitals and physicians.
Medical tourism also lures patients who are citizens of countries (e.g. Canada, the United Kingdom) that offer universal healthcare, Dr. Li says.3 For example, more expatriates from India and Malaysia are traveling to their native countries for medical care, as they receive affordable and quicker medical care while visiting family.
Hospitalists routinely care for patients requiring essential cardiac or orthopedic surgeries—conditions that are common in the medical tourism trade. With medical tourism growing in scope and popularity, it is essential that hospitalists are prepared to discuss with their patients the pros and cons of traveling for medical care. Hospitalists should be able to:
- Identify patients who might benefit from medical tourism;
- Know where and how to look for an accredited overseas facility; and
- Explain to patients the potential travel risks and complications, including insurance coverage and legal restrictions in destination countries.
A basic understanding of the industry and the issues can help guide your patients through medical decisions and help you care for those who have returned from a medical trip.
Big Menu, Discount Prices
Medical tourism offers a wide range of medical services performed in hospitals on nearly every continent, with a wide range of costs for certain procedures (Table 1, p. 26). Most surgeries cost 50% to 90% less than the average cost of the same surgery at a U.S. hospital. Many in the medical tourism industry say these types of savings have brought once-unaffordable surgery within the reach of most Americans, regardless of insurance status. For example, cardiac bypass surgery on average costs $144,000 in the U.S.; it costs about $8,500 in India.
The reasons the costs are so much less at overseas hospitals, as compared with U.S. costs, are many:
- Lower wages for providers;
- Less expensive medical devices and pharmaceutical products;
- Less involvement by third-party payors; and
- Lower malpractice premiums.4
For example, the annual liability insurance premium for a surgeon in India is $4,000; the average cost of a New York City surgeon’s liability insurance premium is $100,000.5
Brazil, Costa Rica, and Mexico are attractive destinations for cosmetic and dental surgeries; Singapore, Malaysia, Thailand, and India have emerged as hubs for cardiac and orthopedic surgeries (Table 1, above).6
The growth in medical tourism led the Joint Commission in 1999 to launch Joint Commission International (JCI), which ensures that offshore hospitals provide the highest-quality care to international patients (see “Ultra-Affordable Prices and No Decline in Quality of Care,” p. 28). JCI has accredited 120 overseas hospitals that meet these standards.
“Overseas hospitals are always keen in partnering with U.S. hospitals,” Dr. Li says. Collaborations, such as Johns Hopkins International Medical Center’s partnership with International Medical Clinic Singapore, and Partners Harvard Medical International’s affiliation with Wockhardt Hospitals in Mumbai, India, have helped facilitate the accreditation process and alleviate U.S. patient concerns.
Overseas hospitals not only offer greatly discounted rates than insurer-negotiated U.S. prices, but many of the international hospitals also report quality scores equal to or better than the average U.S. hospital’s.7
Before You Book a Trip …
How do patients find overseas facilities? It’s as easy as a click of the mouse.
“Our survey shows 75 percent of patients located offshore hospitals through the Internet,” says Renee-Marie Stephano, president of the Medical Tourism Association, a nonprofit group based in West Palm Beach, Fla., that was established in 2007 to promote education, transparency, and communication in the medical tourism community. Patients use overseas hospital websites, international medical coordinators, and medical tourism companies, such as PlanetHospital.com and MedRetreat.com, to find facilities and providers, and to coordinate medical travel.
“Medical tourism is currently unregulated,” Stephano says. “One of our goals is to certify medical tourism facilitators to create the best standard of practice.”
In 2007, the American Medical Association (AMA) published medical tourism guidelines to help healthcare entities engaged in overseas medical care.8 (Download a PDF of the guidelines at www.ama-assn.org/ama1/pub/upload/mm/31/medicaltourism.pdf.) The AMA suggests that:
- Medical care outside the U.S. should be voluntary, and patients should be informed of their legal rights and recourses before agreeing to travel outside the U.S.;
- Before traveling, local follow-up care should be arranged to ensure continuity of care;
- Patients should have access to physician licensing, facility accreditation, and outcomes data for both;
- Medical record transfers should follow HIPAA guidelines; and
- Patients should be informed of the potential risks of combining surgical procedures with long flights and vacation activities.
—Kenneth Mays, senior director, marketing and business development, Bumrungrad Hospital, Bangkok, Thailand
Patient Concerns
Hospitalists are highly focused on a patient’s quality of care; the same can be said of some overseas hospitals that attract large numbers of medical tourists. “I think the service and quality of care provided at our hospital compares favorably with the very best American hospitals,” says Kenneth Mays, senior director of hospital marketing and business development at Bumrungrad Hospital in Bangkok.
That might be so, but the growth of medical tourism also raises concerns stateside. With sleek websites making it easy for U.S. patients to schedule procedure vacations from their kitchen tables, many U.S. physician and watchdog groups worry about patient safety, privacy, liability, and continuity of care. Although most international hospitals and physicians provide outcomes data, rarely do the benchmarks compare directly with U.S. hospital quality and safety data.
“Quality comparisons are difficult, even within U.S. hospitals, as hospitals use different methodologies to collect data,” says Stephano. “Patients have to rely on JCI accreditation, surgeon experience, volume, and outcomes to decide.”
Recent studies echo the Medical Tourism Association’s claim: Increased cardiac surgery volume at Apollo Hospitals—an 8,500-bed healthcare system with 50 locations throughout India—and Narayana Hospital in Bommasandra, India, has lowered costs, with similar, or even lower, mortality rates compared with the average U.S. hospital.7,9 Other challenges like getting medical records exist even within U.S. hospitals, so emerging platforms like Google Health and Microsoft Vault, where medical records can be uploaded at the touch of a button, “will benefit patients and providers,” Dr. Li says.
The Medical Tourism Association envisions U.S.-based physicians offering follow-up care to medical tourism patients. “Currently, we encourage patients to follow up with their primary-care physician,” Stephano says.
Dr. Li says malpractice is always a concern when traveling overseas; however, he also notes the legal system in the U.S. is strong enough “to handle any medical malpractice.” That said, a patient who experiences a poor medical outcome as the result of overseas treatment might seek legal remedies, but the reality is that malpractice laws are either nonexistent or not well implemented in some destination countries. That makes malpractice claims on overseas procedures a dicey proposition.
“Patients receiving overseas treatment need to realize that they are agreeing to the jurisdiction of the destination country,” Stephano says. Other risks associated with extended travel include exposure to regional infectious diseases and poor infrastructure in the destination country, which could undermine the benefits of medical travel.
Cost-saving benefits have led some U.S. insurance companies to begin integrating overseas medical coverage. For example, Blue Cross Blue Shield of South Carolina offers incentives for patients willing to obtain medical care overseas at JCI-approved hospitals. BCBS then waives deductibles and copays, and several other insurers have launched similar pilot programs.10 “We will see more of these changes,” Stephano says, “to cut costs and remain competitive.”
Immediate Impact
In 2008, U.S. healthcare spending was $2.3 trillion.11 A 2005 Institute of Medicine report suggests that 30% to 40% of current U.S. healthcare expenditure is wasted.12 U.S. lawmakers, employers, hospitals, and consumers are scrambling to find ways to reduce healthcare costs and improve efficiency. Medical tourism seems to benefit a select few Americans, only lowering U.S. healthcare spending by 1% to 2%.12
Medical tourism revenue generated in destination countries currently is limited to the private sector, but that might change soon. Government funding for healthcare initiatives in such countries as India, Brazil, and Thailand is declining. Some entrepreneurial physicians and hospitals are looking to medical tourism to fill the funding gap.
Medical tourism likely will continue to grow; so too will the legal, quality, and insurance protections for patients. Efficient resource utilization might help reduce U.S. healthcare costs, and improved distribution of destination-country resources might help improve infrastructure and access to better healthcare for their own citizens.
With their leadership skills and expertise, hospitalists can play a major role in reducing healthcare costs.
However, what actual reforms healthcare legislation brings to medical tourism remain to be seen. TH
Dr. Thakkar is a hospitalist and assistant professor in the division of hospital medicine at Johns Hopkins University School of Medicine in Baltimore.
References
- Medical tourism: Consumers in search of value. Deloitte Consulting LLP website. Available at: www.deloitte.com/dtt/cda/doc/content/us%5Fchs%5FMedicalTourismStudy(1).pdf. Accessed Sept. 13, 2010.
- Pafford B. The third wave—medical tourism in the 21st century. South Med J. 2009;102(8):810-813.
- Kher U. Outsourcing your heart. Available at: http://proquest.umi.com/pqdweb?did=1041533291&Fmt=7&clientId=5241&RQT=309&VName=PQD. Accessed Sept. 13, 2010.
- Forgione DA, Smith PC. Medical tourism and its impact on the US health care system. J Health Care Finance. 2007;34(1):27-35.
- Lancaster J. Surgeries, side trips for “medical tourists.” The Washington Post website. Available at: www.washingtonpost.com/wp-dyn/articles/A497432004Oct20.html. Accessed Sept. 13, 2010.
- Horowitz MD, Rosensweig JA, Jones CA. Medical tourism: Globaliz-ation of the healthcare marketplace. MedGenMed. 2007;9(4):33.
- Milstein A, Smith M. Will the surgical world become flat? Health Aff (Millwood). 2007;26(1):137-141.
- New AMA guidelines on medical tourism. AMA website. Available at: www.ama-assn.org/ama1/pub/upload/mm/31/medicaltourism.pdf. Accessed March 26, 2010.
- Anand G. The Henry Ford of heart surgery. Wall Street Journal website. Available at: online.wsj.com/article/SB12587589288795811.html.
- Einhorn B. Outsourcing the patients. Business Week website. Available at: www.businessweek.com/magazine/content/08_12/b40760367 77780.htm. Accessed Sept. 13, 2010.
- . Hartman M, Martin A, Nuccio O, Catlin A, et al. Health spending growth at a historic low in 2008. Health Aff (Millwood). 2010;29(1): 147-155.
- Milstein A, Smith M. America’s new refugees—seeking affordable surgery offshore. N Engl J Med. 2006;355(16):1637-1640.
David Dupray, a 60-year-old uninsured coffee shop owner from Bar Harbor, Maine, had been having left leg pain on ambulation for four years. His cardiologist recommended stent placement for left iliac artery stenosis. The estimated bill: approximately $35,000.
Unable to afford the procedure, Dupray began searching the Web for affordable medical care overseas. His physician suggested Thailand. Within days, Dupray had an appointment with a cardiologist halfway around the world—at Bumrungrad Hospital in Bangkok, Thailand. Dupray spent two days in the hotel-like hospital, had three stents placed in his leg arteries, and completed a cardiac stress test. The total bill: $18,000.
“I will never go to a hospital in the U.S.,” says Dupray, who represents a growing number of Americans searching for affordable healthcare in the global marketplace.
With rising U.S. healthcare costs and millions of Americans uninsured or underinsured, more American patients are seeking affordable, high-quality medical care abroad—known as “medical tourism.” In 2007, an estimated 750,000 Americans traveled abroad for medical care; the number is expected to increase to 6 million by the end of this year.1 On the flip side, only a little more than 400,000 nonresidents visited the U.S. in 2008 for the latest medical care.1 Globally, the medical tourism industry is estimated to grow into a $21 billion-a-year industry by 2012, with much of the growth expected from Western patients traveling overseas for affordable care.2
“As hospitalists, we have been seeing increasing numbers of patients going overseas for urgent and elective procedures, as it is a general perception the medical treatment overseas is less expensive,” says Joseph Ming Wah Li, MD, SFHM, director of hospital medicine at Beth Israel Deaconess Medical Center in Boston and SHM president-elect.
Physicians in U.S. hospitals encounter potential medical tourists all the time. Some are uninsured or underinsured. Some have insurance carriers that limit or exclude coverage for certain procedures and treatments. Even those with insurance sometimes struggle to pay deductibles, copays, and their costs after insurance has paid its part. Others are uncomfortable with the language barriers and cultural differences of U.S. hospitals and physicians.
Medical tourism also lures patients who are citizens of countries (e.g. Canada, the United Kingdom) that offer universal healthcare, Dr. Li says.3 For example, more expatriates from India and Malaysia are traveling to their native countries for medical care, as they receive affordable and quicker medical care while visiting family.
Hospitalists routinely care for patients requiring essential cardiac or orthopedic surgeries—conditions that are common in the medical tourism trade. With medical tourism growing in scope and popularity, it is essential that hospitalists are prepared to discuss with their patients the pros and cons of traveling for medical care. Hospitalists should be able to:
- Identify patients who might benefit from medical tourism;
- Know where and how to look for an accredited overseas facility; and
- Explain to patients the potential travel risks and complications, including insurance coverage and legal restrictions in destination countries.
A basic understanding of the industry and the issues can help guide your patients through medical decisions and help you care for those who have returned from a medical trip.
Big Menu, Discount Prices
Medical tourism offers a wide range of medical services performed in hospitals on nearly every continent, with a wide range of costs for certain procedures (Table 1, p. 26). Most surgeries cost 50% to 90% less than the average cost of the same surgery at a U.S. hospital. Many in the medical tourism industry say these types of savings have brought once-unaffordable surgery within the reach of most Americans, regardless of insurance status. For example, cardiac bypass surgery on average costs $144,000 in the U.S.; it costs about $8,500 in India.
The reasons the costs are so much less at overseas hospitals, as compared with U.S. costs, are many:
- Lower wages for providers;
- Less expensive medical devices and pharmaceutical products;
- Less involvement by third-party payors; and
- Lower malpractice premiums.4
For example, the annual liability insurance premium for a surgeon in India is $4,000; the average cost of a New York City surgeon’s liability insurance premium is $100,000.5
Brazil, Costa Rica, and Mexico are attractive destinations for cosmetic and dental surgeries; Singapore, Malaysia, Thailand, and India have emerged as hubs for cardiac and orthopedic surgeries (Table 1, above).6
The growth in medical tourism led the Joint Commission in 1999 to launch Joint Commission International (JCI), which ensures that offshore hospitals provide the highest-quality care to international patients (see “Ultra-Affordable Prices and No Decline in Quality of Care,” p. 28). JCI has accredited 120 overseas hospitals that meet these standards.
“Overseas hospitals are always keen in partnering with U.S. hospitals,” Dr. Li says. Collaborations, such as Johns Hopkins International Medical Center’s partnership with International Medical Clinic Singapore, and Partners Harvard Medical International’s affiliation with Wockhardt Hospitals in Mumbai, India, have helped facilitate the accreditation process and alleviate U.S. patient concerns.
Overseas hospitals not only offer greatly discounted rates than insurer-negotiated U.S. prices, but many of the international hospitals also report quality scores equal to or better than the average U.S. hospital’s.7
Before You Book a Trip …
How do patients find overseas facilities? It’s as easy as a click of the mouse.
“Our survey shows 75 percent of patients located offshore hospitals through the Internet,” says Renee-Marie Stephano, president of the Medical Tourism Association, a nonprofit group based in West Palm Beach, Fla., that was established in 2007 to promote education, transparency, and communication in the medical tourism community. Patients use overseas hospital websites, international medical coordinators, and medical tourism companies, such as PlanetHospital.com and MedRetreat.com, to find facilities and providers, and to coordinate medical travel.
“Medical tourism is currently unregulated,” Stephano says. “One of our goals is to certify medical tourism facilitators to create the best standard of practice.”
In 2007, the American Medical Association (AMA) published medical tourism guidelines to help healthcare entities engaged in overseas medical care.8 (Download a PDF of the guidelines at www.ama-assn.org/ama1/pub/upload/mm/31/medicaltourism.pdf.) The AMA suggests that:
- Medical care outside the U.S. should be voluntary, and patients should be informed of their legal rights and recourses before agreeing to travel outside the U.S.;
- Before traveling, local follow-up care should be arranged to ensure continuity of care;
- Patients should have access to physician licensing, facility accreditation, and outcomes data for both;
- Medical record transfers should follow HIPAA guidelines; and
- Patients should be informed of the potential risks of combining surgical procedures with long flights and vacation activities.
—Kenneth Mays, senior director, marketing and business development, Bumrungrad Hospital, Bangkok, Thailand
Patient Concerns
Hospitalists are highly focused on a patient’s quality of care; the same can be said of some overseas hospitals that attract large numbers of medical tourists. “I think the service and quality of care provided at our hospital compares favorably with the very best American hospitals,” says Kenneth Mays, senior director of hospital marketing and business development at Bumrungrad Hospital in Bangkok.
That might be so, but the growth of medical tourism also raises concerns stateside. With sleek websites making it easy for U.S. patients to schedule procedure vacations from their kitchen tables, many U.S. physician and watchdog groups worry about patient safety, privacy, liability, and continuity of care. Although most international hospitals and physicians provide outcomes data, rarely do the benchmarks compare directly with U.S. hospital quality and safety data.
“Quality comparisons are difficult, even within U.S. hospitals, as hospitals use different methodologies to collect data,” says Stephano. “Patients have to rely on JCI accreditation, surgeon experience, volume, and outcomes to decide.”
Recent studies echo the Medical Tourism Association’s claim: Increased cardiac surgery volume at Apollo Hospitals—an 8,500-bed healthcare system with 50 locations throughout India—and Narayana Hospital in Bommasandra, India, has lowered costs, with similar, or even lower, mortality rates compared with the average U.S. hospital.7,9 Other challenges like getting medical records exist even within U.S. hospitals, so emerging platforms like Google Health and Microsoft Vault, where medical records can be uploaded at the touch of a button, “will benefit patients and providers,” Dr. Li says.
The Medical Tourism Association envisions U.S.-based physicians offering follow-up care to medical tourism patients. “Currently, we encourage patients to follow up with their primary-care physician,” Stephano says.
Dr. Li says malpractice is always a concern when traveling overseas; however, he also notes the legal system in the U.S. is strong enough “to handle any medical malpractice.” That said, a patient who experiences a poor medical outcome as the result of overseas treatment might seek legal remedies, but the reality is that malpractice laws are either nonexistent or not well implemented in some destination countries. That makes malpractice claims on overseas procedures a dicey proposition.
“Patients receiving overseas treatment need to realize that they are agreeing to the jurisdiction of the destination country,” Stephano says. Other risks associated with extended travel include exposure to regional infectious diseases and poor infrastructure in the destination country, which could undermine the benefits of medical travel.
Cost-saving benefits have led some U.S. insurance companies to begin integrating overseas medical coverage. For example, Blue Cross Blue Shield of South Carolina offers incentives for patients willing to obtain medical care overseas at JCI-approved hospitals. BCBS then waives deductibles and copays, and several other insurers have launched similar pilot programs.10 “We will see more of these changes,” Stephano says, “to cut costs and remain competitive.”
Immediate Impact
In 2008, U.S. healthcare spending was $2.3 trillion.11 A 2005 Institute of Medicine report suggests that 30% to 40% of current U.S. healthcare expenditure is wasted.12 U.S. lawmakers, employers, hospitals, and consumers are scrambling to find ways to reduce healthcare costs and improve efficiency. Medical tourism seems to benefit a select few Americans, only lowering U.S. healthcare spending by 1% to 2%.12
Medical tourism revenue generated in destination countries currently is limited to the private sector, but that might change soon. Government funding for healthcare initiatives in such countries as India, Brazil, and Thailand is declining. Some entrepreneurial physicians and hospitals are looking to medical tourism to fill the funding gap.
Medical tourism likely will continue to grow; so too will the legal, quality, and insurance protections for patients. Efficient resource utilization might help reduce U.S. healthcare costs, and improved distribution of destination-country resources might help improve infrastructure and access to better healthcare for their own citizens.
With their leadership skills and expertise, hospitalists can play a major role in reducing healthcare costs.
However, what actual reforms healthcare legislation brings to medical tourism remain to be seen. TH
Dr. Thakkar is a hospitalist and assistant professor in the division of hospital medicine at Johns Hopkins University School of Medicine in Baltimore.
References
- Medical tourism: Consumers in search of value. Deloitte Consulting LLP website. Available at: www.deloitte.com/dtt/cda/doc/content/us%5Fchs%5FMedicalTourismStudy(1).pdf. Accessed Sept. 13, 2010.
- Pafford B. The third wave—medical tourism in the 21st century. South Med J. 2009;102(8):810-813.
- Kher U. Outsourcing your heart. Available at: http://proquest.umi.com/pqdweb?did=1041533291&Fmt=7&clientId=5241&RQT=309&VName=PQD. Accessed Sept. 13, 2010.
- Forgione DA, Smith PC. Medical tourism and its impact on the US health care system. J Health Care Finance. 2007;34(1):27-35.
- Lancaster J. Surgeries, side trips for “medical tourists.” The Washington Post website. Available at: www.washingtonpost.com/wp-dyn/articles/A497432004Oct20.html. Accessed Sept. 13, 2010.
- Horowitz MD, Rosensweig JA, Jones CA. Medical tourism: Globaliz-ation of the healthcare marketplace. MedGenMed. 2007;9(4):33.
- Milstein A, Smith M. Will the surgical world become flat? Health Aff (Millwood). 2007;26(1):137-141.
- New AMA guidelines on medical tourism. AMA website. Available at: www.ama-assn.org/ama1/pub/upload/mm/31/medicaltourism.pdf. Accessed March 26, 2010.
- Anand G. The Henry Ford of heart surgery. Wall Street Journal website. Available at: online.wsj.com/article/SB12587589288795811.html.
- Einhorn B. Outsourcing the patients. Business Week website. Available at: www.businessweek.com/magazine/content/08_12/b40760367 77780.htm. Accessed Sept. 13, 2010.
- . Hartman M, Martin A, Nuccio O, Catlin A, et al. Health spending growth at a historic low in 2008. Health Aff (Millwood). 2010;29(1): 147-155.
- Milstein A, Smith M. America’s new refugees—seeking affordable surgery offshore. N Engl J Med. 2006;355(16):1637-1640.
David Dupray, a 60-year-old uninsured coffee shop owner from Bar Harbor, Maine, had been having left leg pain on ambulation for four years. His cardiologist recommended stent placement for left iliac artery stenosis. The estimated bill: approximately $35,000.
Unable to afford the procedure, Dupray began searching the Web for affordable medical care overseas. His physician suggested Thailand. Within days, Dupray had an appointment with a cardiologist halfway around the world—at Bumrungrad Hospital in Bangkok, Thailand. Dupray spent two days in the hotel-like hospital, had three stents placed in his leg arteries, and completed a cardiac stress test. The total bill: $18,000.
“I will never go to a hospital in the U.S.,” says Dupray, who represents a growing number of Americans searching for affordable healthcare in the global marketplace.
With rising U.S. healthcare costs and millions of Americans uninsured or underinsured, more American patients are seeking affordable, high-quality medical care abroad—known as “medical tourism.” In 2007, an estimated 750,000 Americans traveled abroad for medical care; the number is expected to increase to 6 million by the end of this year.1 On the flip side, only a little more than 400,000 nonresidents visited the U.S. in 2008 for the latest medical care.1 Globally, the medical tourism industry is estimated to grow into a $21 billion-a-year industry by 2012, with much of the growth expected from Western patients traveling overseas for affordable care.2
“As hospitalists, we have been seeing increasing numbers of patients going overseas for urgent and elective procedures, as it is a general perception the medical treatment overseas is less expensive,” says Joseph Ming Wah Li, MD, SFHM, director of hospital medicine at Beth Israel Deaconess Medical Center in Boston and SHM president-elect.
Physicians in U.S. hospitals encounter potential medical tourists all the time. Some are uninsured or underinsured. Some have insurance carriers that limit or exclude coverage for certain procedures and treatments. Even those with insurance sometimes struggle to pay deductibles, copays, and their costs after insurance has paid its part. Others are uncomfortable with the language barriers and cultural differences of U.S. hospitals and physicians.
Medical tourism also lures patients who are citizens of countries (e.g. Canada, the United Kingdom) that offer universal healthcare, Dr. Li says.3 For example, more expatriates from India and Malaysia are traveling to their native countries for medical care, as they receive affordable and quicker medical care while visiting family.
Hospitalists routinely care for patients requiring essential cardiac or orthopedic surgeries—conditions that are common in the medical tourism trade. With medical tourism growing in scope and popularity, it is essential that hospitalists are prepared to discuss with their patients the pros and cons of traveling for medical care. Hospitalists should be able to:
- Identify patients who might benefit from medical tourism;
- Know where and how to look for an accredited overseas facility; and
- Explain to patients the potential travel risks and complications, including insurance coverage and legal restrictions in destination countries.
A basic understanding of the industry and the issues can help guide your patients through medical decisions and help you care for those who have returned from a medical trip.
Big Menu, Discount Prices
Medical tourism offers a wide range of medical services performed in hospitals on nearly every continent, with a wide range of costs for certain procedures (Table 1, p. 26). Most surgeries cost 50% to 90% less than the average cost of the same surgery at a U.S. hospital. Many in the medical tourism industry say these types of savings have brought once-unaffordable surgery within the reach of most Americans, regardless of insurance status. For example, cardiac bypass surgery on average costs $144,000 in the U.S.; it costs about $8,500 in India.
The reasons the costs are so much less at overseas hospitals, as compared with U.S. costs, are many:
- Lower wages for providers;
- Less expensive medical devices and pharmaceutical products;
- Less involvement by third-party payors; and
- Lower malpractice premiums.4
For example, the annual liability insurance premium for a surgeon in India is $4,000; the average cost of a New York City surgeon’s liability insurance premium is $100,000.5
Brazil, Costa Rica, and Mexico are attractive destinations for cosmetic and dental surgeries; Singapore, Malaysia, Thailand, and India have emerged as hubs for cardiac and orthopedic surgeries (Table 1, above).6
The growth in medical tourism led the Joint Commission in 1999 to launch Joint Commission International (JCI), which ensures that offshore hospitals provide the highest-quality care to international patients (see “Ultra-Affordable Prices and No Decline in Quality of Care,” p. 28). JCI has accredited 120 overseas hospitals that meet these standards.
“Overseas hospitals are always keen in partnering with U.S. hospitals,” Dr. Li says. Collaborations, such as Johns Hopkins International Medical Center’s partnership with International Medical Clinic Singapore, and Partners Harvard Medical International’s affiliation with Wockhardt Hospitals in Mumbai, India, have helped facilitate the accreditation process and alleviate U.S. patient concerns.
Overseas hospitals not only offer greatly discounted rates than insurer-negotiated U.S. prices, but many of the international hospitals also report quality scores equal to or better than the average U.S. hospital’s.7
Before You Book a Trip …
How do patients find overseas facilities? It’s as easy as a click of the mouse.
“Our survey shows 75 percent of patients located offshore hospitals through the Internet,” says Renee-Marie Stephano, president of the Medical Tourism Association, a nonprofit group based in West Palm Beach, Fla., that was established in 2007 to promote education, transparency, and communication in the medical tourism community. Patients use overseas hospital websites, international medical coordinators, and medical tourism companies, such as PlanetHospital.com and MedRetreat.com, to find facilities and providers, and to coordinate medical travel.
“Medical tourism is currently unregulated,” Stephano says. “One of our goals is to certify medical tourism facilitators to create the best standard of practice.”
In 2007, the American Medical Association (AMA) published medical tourism guidelines to help healthcare entities engaged in overseas medical care.8 (Download a PDF of the guidelines at www.ama-assn.org/ama1/pub/upload/mm/31/medicaltourism.pdf.) The AMA suggests that:
- Medical care outside the U.S. should be voluntary, and patients should be informed of their legal rights and recourses before agreeing to travel outside the U.S.;
- Before traveling, local follow-up care should be arranged to ensure continuity of care;
- Patients should have access to physician licensing, facility accreditation, and outcomes data for both;
- Medical record transfers should follow HIPAA guidelines; and
- Patients should be informed of the potential risks of combining surgical procedures with long flights and vacation activities.
—Kenneth Mays, senior director, marketing and business development, Bumrungrad Hospital, Bangkok, Thailand
Patient Concerns
Hospitalists are highly focused on a patient’s quality of care; the same can be said of some overseas hospitals that attract large numbers of medical tourists. “I think the service and quality of care provided at our hospital compares favorably with the very best American hospitals,” says Kenneth Mays, senior director of hospital marketing and business development at Bumrungrad Hospital in Bangkok.
That might be so, but the growth of medical tourism also raises concerns stateside. With sleek websites making it easy for U.S. patients to schedule procedure vacations from their kitchen tables, many U.S. physician and watchdog groups worry about patient safety, privacy, liability, and continuity of care. Although most international hospitals and physicians provide outcomes data, rarely do the benchmarks compare directly with U.S. hospital quality and safety data.
“Quality comparisons are difficult, even within U.S. hospitals, as hospitals use different methodologies to collect data,” says Stephano. “Patients have to rely on JCI accreditation, surgeon experience, volume, and outcomes to decide.”
Recent studies echo the Medical Tourism Association’s claim: Increased cardiac surgery volume at Apollo Hospitals—an 8,500-bed healthcare system with 50 locations throughout India—and Narayana Hospital in Bommasandra, India, has lowered costs, with similar, or even lower, mortality rates compared with the average U.S. hospital.7,9 Other challenges like getting medical records exist even within U.S. hospitals, so emerging platforms like Google Health and Microsoft Vault, where medical records can be uploaded at the touch of a button, “will benefit patients and providers,” Dr. Li says.
The Medical Tourism Association envisions U.S.-based physicians offering follow-up care to medical tourism patients. “Currently, we encourage patients to follow up with their primary-care physician,” Stephano says.
Dr. Li says malpractice is always a concern when traveling overseas; however, he also notes the legal system in the U.S. is strong enough “to handle any medical malpractice.” That said, a patient who experiences a poor medical outcome as the result of overseas treatment might seek legal remedies, but the reality is that malpractice laws are either nonexistent or not well implemented in some destination countries. That makes malpractice claims on overseas procedures a dicey proposition.
“Patients receiving overseas treatment need to realize that they are agreeing to the jurisdiction of the destination country,” Stephano says. Other risks associated with extended travel include exposure to regional infectious diseases and poor infrastructure in the destination country, which could undermine the benefits of medical travel.
Cost-saving benefits have led some U.S. insurance companies to begin integrating overseas medical coverage. For example, Blue Cross Blue Shield of South Carolina offers incentives for patients willing to obtain medical care overseas at JCI-approved hospitals. BCBS then waives deductibles and copays, and several other insurers have launched similar pilot programs.10 “We will see more of these changes,” Stephano says, “to cut costs and remain competitive.”
Immediate Impact
In 2008, U.S. healthcare spending was $2.3 trillion.11 A 2005 Institute of Medicine report suggests that 30% to 40% of current U.S. healthcare expenditure is wasted.12 U.S. lawmakers, employers, hospitals, and consumers are scrambling to find ways to reduce healthcare costs and improve efficiency. Medical tourism seems to benefit a select few Americans, only lowering U.S. healthcare spending by 1% to 2%.12
Medical tourism revenue generated in destination countries currently is limited to the private sector, but that might change soon. Government funding for healthcare initiatives in such countries as India, Brazil, and Thailand is declining. Some entrepreneurial physicians and hospitals are looking to medical tourism to fill the funding gap.
Medical tourism likely will continue to grow; so too will the legal, quality, and insurance protections for patients. Efficient resource utilization might help reduce U.S. healthcare costs, and improved distribution of destination-country resources might help improve infrastructure and access to better healthcare for their own citizens.
With their leadership skills and expertise, hospitalists can play a major role in reducing healthcare costs.
However, what actual reforms healthcare legislation brings to medical tourism remain to be seen. TH
Dr. Thakkar is a hospitalist and assistant professor in the division of hospital medicine at Johns Hopkins University School of Medicine in Baltimore.
References
- Medical tourism: Consumers in search of value. Deloitte Consulting LLP website. Available at: www.deloitte.com/dtt/cda/doc/content/us%5Fchs%5FMedicalTourismStudy(1).pdf. Accessed Sept. 13, 2010.
- Pafford B. The third wave—medical tourism in the 21st century. South Med J. 2009;102(8):810-813.
- Kher U. Outsourcing your heart. Available at: http://proquest.umi.com/pqdweb?did=1041533291&Fmt=7&clientId=5241&RQT=309&VName=PQD. Accessed Sept. 13, 2010.
- Forgione DA, Smith PC. Medical tourism and its impact on the US health care system. J Health Care Finance. 2007;34(1):27-35.
- Lancaster J. Surgeries, side trips for “medical tourists.” The Washington Post website. Available at: www.washingtonpost.com/wp-dyn/articles/A497432004Oct20.html. Accessed Sept. 13, 2010.
- Horowitz MD, Rosensweig JA, Jones CA. Medical tourism: Globaliz-ation of the healthcare marketplace. MedGenMed. 2007;9(4):33.
- Milstein A, Smith M. Will the surgical world become flat? Health Aff (Millwood). 2007;26(1):137-141.
- New AMA guidelines on medical tourism. AMA website. Available at: www.ama-assn.org/ama1/pub/upload/mm/31/medicaltourism.pdf. Accessed March 26, 2010.
- Anand G. The Henry Ford of heart surgery. Wall Street Journal website. Available at: online.wsj.com/article/SB12587589288795811.html.
- Einhorn B. Outsourcing the patients. Business Week website. Available at: www.businessweek.com/magazine/content/08_12/b40760367 77780.htm. Accessed Sept. 13, 2010.
- . Hartman M, Martin A, Nuccio O, Catlin A, et al. Health spending growth at a historic low in 2008. Health Aff (Millwood). 2010;29(1): 147-155.
- Milstein A, Smith M. America’s new refugees—seeking affordable surgery offshore. N Engl J Med. 2006;355(16):1637-1640.
Endangered Species?
The 1961 classic “The Ecology of Medical Care,” published in the New England Journal of Medicine, mapped out the broad features of the American healthcare landscape.1 For every 1,000 adult, the study suggested, 750 reported an illness, 250 consulted a doctor, and nine were admitted to a hospital in any given month. The subsequent arrival of Medicare and Medicaid fundamentally changed the U.S. healthcare system. And yet an updated version of the study, released in 2001, yielded surprisingly similar numbers, with 800 residents experiencing symptoms, 217 visiting a physician’s office, and eight being hospitalized in an average month.2
“It helps kind of put in perspective where the bulk of care really occurs,” says Ann O’Malley, MD, a senior researcher at the Washington, D.C.-based Center for Studying Health System Change. “It’s in outpatient provider offices, mostly primary-care provider offices.”
Dr. O’Malley and a host of other observers, however, are warning that the keystone members of this healthcare ecosystem are in serious trouble. As organizations such as SHM have likewise made clear, the accelerating shortage of general internists, family practitioners, and other PCPs has created sizable cracks in the supports of the entire healthcare infrastructure.
How big are the cracks? The number of medical school students pursuing a primary-care career has dropped by more than half since 1997, according to the American Academy of Family Physicians. And with the number of medical students entering the field unable to keep up with attrition, the remaining doctors are facing increasingly difficult working conditions. “Overloaded primary-care practices, whose doctors are aptly compared to hamsters on a treadmill, struggle to provide prompt access and high-quality care,” asserted a 2009 op-ed in the New England Journal of Medicine.3 The result: a vicious circle of decline leading to an anticipated shortfall of roughly 21,000 PCPs by 2015, according to the Association of American Medical Colleges.
Many primary-care providers had already stopped taking new patients when June’s Medicare reimbursement rate fiasco allowed the sustainable growth rate (SGR) formula’s mandated 21.2 percent rate cut to temporarily go into effect. Legislators eventually plugged the hole, but not before a new round of jitters seized the nation’s physicians, and reports proliferated throughout the summer about Medicare beneficiaries being unable to find a doctor willing to see them. The recession hasn’t helped, with more privately insured patients waiting longer to see their doctors to avoid copays, and with hospital emergency departments becoming de facto primary-care centers for those patients who have waited too long or have no other alternatives.
Uneven Challenges
Not only is there an acute shortage of primary-care physicians, Dr. O’Malley says, but there is also a distinctly uneven distribution throughout the country. For hospitalists, she says, the implications could be profound. “Hospitalists are increasingly going to be evaluated around issues such as avoiding hospital readmissions and [reducing] length of stay,” she says, “and if they want to improve both of those things, one of the keys is improving chronic care management in the outpatient setting, and improving follow-up post discharge.”
Both metrics will require the involvement of outpatient care providers, underscoring the importance of good communication and mutual respect. Despite the longstanding support of hospitalists for their primary-care counterparts, however, leaders are still being forced to address the perception that HM is somehow bad for what ails PCPs.
In a recent online article posted on the Becker’s Hospital Review website, SHM President Jeff Wiese, MD, SFHM, responded to one such criticism: that hospitalists make primary care less attractive for physicians. Hospitalists are not to blame for the decrease in interest, he asserted, but are actually complementary to the PCP role. And with millions more Americans about to be newly insured, that complementary relationship will be even more important. “It’s a tremendous waste of resources to use a primary-care provider for [a hospital visit]. We need to move into proactive mode, not reactive mode,” Dr. Wiese said. “More PCPs are going to need even more time in the clinic to handle the increased number of patients, and you lose the luxury to run back and forth between the clinic and the hospital. For those that can develop a trusting relationship with a hospitalist, you can work together to see more patients and provide more care.”
So what’s the real root of the problem? Money. According to recent surveys, PCPs earn about half the salary of dermatologists and an even smaller fraction of an average cardiologist’s pay. With medical school debt routinely reaching $200,000, Dr. O’Malley and other analysts say, many doctors simply can’t afford to go into primary care.
“It all comes down to payment, basically,” she says. “At present, our payment system for physician services and for medical procedures is quite skewed. It overcompensates for certain types of diagnostics and procedures, and it undercompensates for the more cognitive type of care that primary-care providers provide.”
The Road Ahead
Fortunately, some relief is trickling in. One measure strongly supported by SHM and included in the Affordable Care Act is a 10% Medicare reimbursement bonus for primary care delivered by qualified doctors, slated to begin next year. In June, U.S. Department of Health and Human Services Secretary Kathleen Sebelius announced a separate, $250 million initiative to boost the primary-care workforce. The money would help train PCPs by creating more residency slots, and offer new support for physician assistants, nurses, and nurse practitioners. Among the measures included in last year’s stimulus package, an expansion of the National Health Service Corps will provide more debt-relief opportunities for PCPs. And in mid-September, HHS tapped stimulus funds to award another $50.3 million for primary care training programs and loan repayment.
The Obama administration has claimed its combined actions “will support the training and development of more than 16,000 new primary-care providers over the next five years,” according to a June 16 HHS press release.
Observers say those measures alone are unlikely to be enough to stem the tide, however. “It’s definitely a step in the right direction,” Dr. O’Malley says of the Medicare bonus. “I don’t think it’s going to solve the primary-care workforce issue, because a 10% bonus, given how low primary-care physician salaries are compared to their specialist counterparts, is not going to be that much of an increase. Among the physicians that I’ve talked to and other healthcare providers, few feel that that’s sufficient enough to really encourage a lot of people to pursue primary care.”
Several other efforts now underway might help:
- Texas Tech University Health Sciences Center unveiled a new Family Medicine Accelerated Track program, which will allow primary-care medical students to complete a degree in three years. Certain students will receive a one-year scholarship, meaning that overall debt for some could be half that of the standard four-year program.
- Reid Hospital and Health Care Services in Richmond, Ind., successfully reversed a downward trend in primary-care referrals by forming its own nonprofit subsidiary corporation, Reid Physician Associates. The nonprofit will include about 50 employed outpatient providers by year’s end to complement the 233-bed hospital’s inpatient staff.
- Danville, Pa.-based Geisinger Health System has begun paying the salaries of extra nurses for both in-network and independent primary-care practices. The nurses manage patients’ chronic conditions, ensure that they are following prescribed treatments, and communicate with hospitalists and other providers about transitions of care. Although still in its early stages, the experiment suggests the nurses are helping to spot problems, prevent unnecessary hospitalizations, and save money.
The Geisinger experiment is among the first steps toward a patient-centered medical home model of care. An eventual Medicare-led expansion of such medical homes and accountable-care organizations, now in the early experimental stages, could provide even more direct support to PCPs. To be successful, though, Dr. O’Malley says the models will need to focus on paying providers fairly for the value they bring to the system. “Obviously, payment reform is what we need if we’re ever going to develop a sustainable primary-care workforce in this country,” she says. TH
Bryn Nelson is a freelance medical writer based in Seattle.
References
- White KL, Williams TF, Greenberg BG. The ecology of medical care. N Engl J Med. 1961;265:885-992.
- Green LA, Fryer GE Jr., Yawn BP, Lanier D, Dovey SM. The ecology of medical care revisited. N Engl J Med. 2001;344(26):2021-2025.
- Bodenheimer T, Grumbach K, Berenson RA. A lifeline for primary care. N Engl J Med. 2009;360(26):2693-2696.
The 1961 classic “The Ecology of Medical Care,” published in the New England Journal of Medicine, mapped out the broad features of the American healthcare landscape.1 For every 1,000 adult, the study suggested, 750 reported an illness, 250 consulted a doctor, and nine were admitted to a hospital in any given month. The subsequent arrival of Medicare and Medicaid fundamentally changed the U.S. healthcare system. And yet an updated version of the study, released in 2001, yielded surprisingly similar numbers, with 800 residents experiencing symptoms, 217 visiting a physician’s office, and eight being hospitalized in an average month.2
“It helps kind of put in perspective where the bulk of care really occurs,” says Ann O’Malley, MD, a senior researcher at the Washington, D.C.-based Center for Studying Health System Change. “It’s in outpatient provider offices, mostly primary-care provider offices.”
Dr. O’Malley and a host of other observers, however, are warning that the keystone members of this healthcare ecosystem are in serious trouble. As organizations such as SHM have likewise made clear, the accelerating shortage of general internists, family practitioners, and other PCPs has created sizable cracks in the supports of the entire healthcare infrastructure.
How big are the cracks? The number of medical school students pursuing a primary-care career has dropped by more than half since 1997, according to the American Academy of Family Physicians. And with the number of medical students entering the field unable to keep up with attrition, the remaining doctors are facing increasingly difficult working conditions. “Overloaded primary-care practices, whose doctors are aptly compared to hamsters on a treadmill, struggle to provide prompt access and high-quality care,” asserted a 2009 op-ed in the New England Journal of Medicine.3 The result: a vicious circle of decline leading to an anticipated shortfall of roughly 21,000 PCPs by 2015, according to the Association of American Medical Colleges.
Many primary-care providers had already stopped taking new patients when June’s Medicare reimbursement rate fiasco allowed the sustainable growth rate (SGR) formula’s mandated 21.2 percent rate cut to temporarily go into effect. Legislators eventually plugged the hole, but not before a new round of jitters seized the nation’s physicians, and reports proliferated throughout the summer about Medicare beneficiaries being unable to find a doctor willing to see them. The recession hasn’t helped, with more privately insured patients waiting longer to see their doctors to avoid copays, and with hospital emergency departments becoming de facto primary-care centers for those patients who have waited too long or have no other alternatives.
Uneven Challenges
Not only is there an acute shortage of primary-care physicians, Dr. O’Malley says, but there is also a distinctly uneven distribution throughout the country. For hospitalists, she says, the implications could be profound. “Hospitalists are increasingly going to be evaluated around issues such as avoiding hospital readmissions and [reducing] length of stay,” she says, “and if they want to improve both of those things, one of the keys is improving chronic care management in the outpatient setting, and improving follow-up post discharge.”
Both metrics will require the involvement of outpatient care providers, underscoring the importance of good communication and mutual respect. Despite the longstanding support of hospitalists for their primary-care counterparts, however, leaders are still being forced to address the perception that HM is somehow bad for what ails PCPs.
In a recent online article posted on the Becker’s Hospital Review website, SHM President Jeff Wiese, MD, SFHM, responded to one such criticism: that hospitalists make primary care less attractive for physicians. Hospitalists are not to blame for the decrease in interest, he asserted, but are actually complementary to the PCP role. And with millions more Americans about to be newly insured, that complementary relationship will be even more important. “It’s a tremendous waste of resources to use a primary-care provider for [a hospital visit]. We need to move into proactive mode, not reactive mode,” Dr. Wiese said. “More PCPs are going to need even more time in the clinic to handle the increased number of patients, and you lose the luxury to run back and forth between the clinic and the hospital. For those that can develop a trusting relationship with a hospitalist, you can work together to see more patients and provide more care.”
So what’s the real root of the problem? Money. According to recent surveys, PCPs earn about half the salary of dermatologists and an even smaller fraction of an average cardiologist’s pay. With medical school debt routinely reaching $200,000, Dr. O’Malley and other analysts say, many doctors simply can’t afford to go into primary care.
“It all comes down to payment, basically,” she says. “At present, our payment system for physician services and for medical procedures is quite skewed. It overcompensates for certain types of diagnostics and procedures, and it undercompensates for the more cognitive type of care that primary-care providers provide.”
The Road Ahead
Fortunately, some relief is trickling in. One measure strongly supported by SHM and included in the Affordable Care Act is a 10% Medicare reimbursement bonus for primary care delivered by qualified doctors, slated to begin next year. In June, U.S. Department of Health and Human Services Secretary Kathleen Sebelius announced a separate, $250 million initiative to boost the primary-care workforce. The money would help train PCPs by creating more residency slots, and offer new support for physician assistants, nurses, and nurse practitioners. Among the measures included in last year’s stimulus package, an expansion of the National Health Service Corps will provide more debt-relief opportunities for PCPs. And in mid-September, HHS tapped stimulus funds to award another $50.3 million for primary care training programs and loan repayment.
The Obama administration has claimed its combined actions “will support the training and development of more than 16,000 new primary-care providers over the next five years,” according to a June 16 HHS press release.
Observers say those measures alone are unlikely to be enough to stem the tide, however. “It’s definitely a step in the right direction,” Dr. O’Malley says of the Medicare bonus. “I don’t think it’s going to solve the primary-care workforce issue, because a 10% bonus, given how low primary-care physician salaries are compared to their specialist counterparts, is not going to be that much of an increase. Among the physicians that I’ve talked to and other healthcare providers, few feel that that’s sufficient enough to really encourage a lot of people to pursue primary care.”
Several other efforts now underway might help:
- Texas Tech University Health Sciences Center unveiled a new Family Medicine Accelerated Track program, which will allow primary-care medical students to complete a degree in three years. Certain students will receive a one-year scholarship, meaning that overall debt for some could be half that of the standard four-year program.
- Reid Hospital and Health Care Services in Richmond, Ind., successfully reversed a downward trend in primary-care referrals by forming its own nonprofit subsidiary corporation, Reid Physician Associates. The nonprofit will include about 50 employed outpatient providers by year’s end to complement the 233-bed hospital’s inpatient staff.
- Danville, Pa.-based Geisinger Health System has begun paying the salaries of extra nurses for both in-network and independent primary-care practices. The nurses manage patients’ chronic conditions, ensure that they are following prescribed treatments, and communicate with hospitalists and other providers about transitions of care. Although still in its early stages, the experiment suggests the nurses are helping to spot problems, prevent unnecessary hospitalizations, and save money.
The Geisinger experiment is among the first steps toward a patient-centered medical home model of care. An eventual Medicare-led expansion of such medical homes and accountable-care organizations, now in the early experimental stages, could provide even more direct support to PCPs. To be successful, though, Dr. O’Malley says the models will need to focus on paying providers fairly for the value they bring to the system. “Obviously, payment reform is what we need if we’re ever going to develop a sustainable primary-care workforce in this country,” she says. TH
Bryn Nelson is a freelance medical writer based in Seattle.
References
- White KL, Williams TF, Greenberg BG. The ecology of medical care. N Engl J Med. 1961;265:885-992.
- Green LA, Fryer GE Jr., Yawn BP, Lanier D, Dovey SM. The ecology of medical care revisited. N Engl J Med. 2001;344(26):2021-2025.
- Bodenheimer T, Grumbach K, Berenson RA. A lifeline for primary care. N Engl J Med. 2009;360(26):2693-2696.
The 1961 classic “The Ecology of Medical Care,” published in the New England Journal of Medicine, mapped out the broad features of the American healthcare landscape.1 For every 1,000 adult, the study suggested, 750 reported an illness, 250 consulted a doctor, and nine were admitted to a hospital in any given month. The subsequent arrival of Medicare and Medicaid fundamentally changed the U.S. healthcare system. And yet an updated version of the study, released in 2001, yielded surprisingly similar numbers, with 800 residents experiencing symptoms, 217 visiting a physician’s office, and eight being hospitalized in an average month.2
“It helps kind of put in perspective where the bulk of care really occurs,” says Ann O’Malley, MD, a senior researcher at the Washington, D.C.-based Center for Studying Health System Change. “It’s in outpatient provider offices, mostly primary-care provider offices.”
Dr. O’Malley and a host of other observers, however, are warning that the keystone members of this healthcare ecosystem are in serious trouble. As organizations such as SHM have likewise made clear, the accelerating shortage of general internists, family practitioners, and other PCPs has created sizable cracks in the supports of the entire healthcare infrastructure.
How big are the cracks? The number of medical school students pursuing a primary-care career has dropped by more than half since 1997, according to the American Academy of Family Physicians. And with the number of medical students entering the field unable to keep up with attrition, the remaining doctors are facing increasingly difficult working conditions. “Overloaded primary-care practices, whose doctors are aptly compared to hamsters on a treadmill, struggle to provide prompt access and high-quality care,” asserted a 2009 op-ed in the New England Journal of Medicine.3 The result: a vicious circle of decline leading to an anticipated shortfall of roughly 21,000 PCPs by 2015, according to the Association of American Medical Colleges.
Many primary-care providers had already stopped taking new patients when June’s Medicare reimbursement rate fiasco allowed the sustainable growth rate (SGR) formula’s mandated 21.2 percent rate cut to temporarily go into effect. Legislators eventually plugged the hole, but not before a new round of jitters seized the nation’s physicians, and reports proliferated throughout the summer about Medicare beneficiaries being unable to find a doctor willing to see them. The recession hasn’t helped, with more privately insured patients waiting longer to see their doctors to avoid copays, and with hospital emergency departments becoming de facto primary-care centers for those patients who have waited too long or have no other alternatives.
Uneven Challenges
Not only is there an acute shortage of primary-care physicians, Dr. O’Malley says, but there is also a distinctly uneven distribution throughout the country. For hospitalists, she says, the implications could be profound. “Hospitalists are increasingly going to be evaluated around issues such as avoiding hospital readmissions and [reducing] length of stay,” she says, “and if they want to improve both of those things, one of the keys is improving chronic care management in the outpatient setting, and improving follow-up post discharge.”
Both metrics will require the involvement of outpatient care providers, underscoring the importance of good communication and mutual respect. Despite the longstanding support of hospitalists for their primary-care counterparts, however, leaders are still being forced to address the perception that HM is somehow bad for what ails PCPs.
In a recent online article posted on the Becker’s Hospital Review website, SHM President Jeff Wiese, MD, SFHM, responded to one such criticism: that hospitalists make primary care less attractive for physicians. Hospitalists are not to blame for the decrease in interest, he asserted, but are actually complementary to the PCP role. And with millions more Americans about to be newly insured, that complementary relationship will be even more important. “It’s a tremendous waste of resources to use a primary-care provider for [a hospital visit]. We need to move into proactive mode, not reactive mode,” Dr. Wiese said. “More PCPs are going to need even more time in the clinic to handle the increased number of patients, and you lose the luxury to run back and forth between the clinic and the hospital. For those that can develop a trusting relationship with a hospitalist, you can work together to see more patients and provide more care.”
So what’s the real root of the problem? Money. According to recent surveys, PCPs earn about half the salary of dermatologists and an even smaller fraction of an average cardiologist’s pay. With medical school debt routinely reaching $200,000, Dr. O’Malley and other analysts say, many doctors simply can’t afford to go into primary care.
“It all comes down to payment, basically,” she says. “At present, our payment system for physician services and for medical procedures is quite skewed. It overcompensates for certain types of diagnostics and procedures, and it undercompensates for the more cognitive type of care that primary-care providers provide.”
The Road Ahead
Fortunately, some relief is trickling in. One measure strongly supported by SHM and included in the Affordable Care Act is a 10% Medicare reimbursement bonus for primary care delivered by qualified doctors, slated to begin next year. In June, U.S. Department of Health and Human Services Secretary Kathleen Sebelius announced a separate, $250 million initiative to boost the primary-care workforce. The money would help train PCPs by creating more residency slots, and offer new support for physician assistants, nurses, and nurse practitioners. Among the measures included in last year’s stimulus package, an expansion of the National Health Service Corps will provide more debt-relief opportunities for PCPs. And in mid-September, HHS tapped stimulus funds to award another $50.3 million for primary care training programs and loan repayment.
The Obama administration has claimed its combined actions “will support the training and development of more than 16,000 new primary-care providers over the next five years,” according to a June 16 HHS press release.
Observers say those measures alone are unlikely to be enough to stem the tide, however. “It’s definitely a step in the right direction,” Dr. O’Malley says of the Medicare bonus. “I don’t think it’s going to solve the primary-care workforce issue, because a 10% bonus, given how low primary-care physician salaries are compared to their specialist counterparts, is not going to be that much of an increase. Among the physicians that I’ve talked to and other healthcare providers, few feel that that’s sufficient enough to really encourage a lot of people to pursue primary care.”
Several other efforts now underway might help:
- Texas Tech University Health Sciences Center unveiled a new Family Medicine Accelerated Track program, which will allow primary-care medical students to complete a degree in three years. Certain students will receive a one-year scholarship, meaning that overall debt for some could be half that of the standard four-year program.
- Reid Hospital and Health Care Services in Richmond, Ind., successfully reversed a downward trend in primary-care referrals by forming its own nonprofit subsidiary corporation, Reid Physician Associates. The nonprofit will include about 50 employed outpatient providers by year’s end to complement the 233-bed hospital’s inpatient staff.
- Danville, Pa.-based Geisinger Health System has begun paying the salaries of extra nurses for both in-network and independent primary-care practices. The nurses manage patients’ chronic conditions, ensure that they are following prescribed treatments, and communicate with hospitalists and other providers about transitions of care. Although still in its early stages, the experiment suggests the nurses are helping to spot problems, prevent unnecessary hospitalizations, and save money.
The Geisinger experiment is among the first steps toward a patient-centered medical home model of care. An eventual Medicare-led expansion of such medical homes and accountable-care organizations, now in the early experimental stages, could provide even more direct support to PCPs. To be successful, though, Dr. O’Malley says the models will need to focus on paying providers fairly for the value they bring to the system. “Obviously, payment reform is what we need if we’re ever going to develop a sustainable primary-care workforce in this country,” she says. TH
Bryn Nelson is a freelance medical writer based in Seattle.
References
- White KL, Williams TF, Greenberg BG. The ecology of medical care. N Engl J Med. 1961;265:885-992.
- Green LA, Fryer GE Jr., Yawn BP, Lanier D, Dovey SM. The ecology of medical care revisited. N Engl J Med. 2001;344(26):2021-2025.
- Bodenheimer T, Grumbach K, Berenson RA. A lifeline for primary care. N Engl J Med. 2009;360(26):2693-2696.
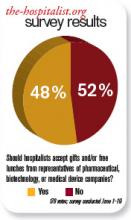
Should hospitalists accept gifts from pharmaceutical, medical device, and biotech companies?
Recent discussions on conflicts of interest in medical publications underscore the significance of the important yet fragile relationship between the pharmaceutical industry and healthcare professionals. Among these is an examination of how academic departments can maintain a relationship with the industry.1 This study suggests that if appropriate boundaries are established between industry and academia, it is possible to collaborate. However, part of the policy in this investigation included “elimination of industry-supplied meals, gifts, and favors.”2
The Institute of Medicine’s “Conflict of Interest in Medical Research, Education, and Practice” included groundbreaking recommendations.3 Among them was a call for professionals to adopt a policy that prohibits “the acceptance of items of material value from pharmaceutical, medical device, and biotechnology companies, except in specified situations.”3
Our nation has been embroiled in a healthcare debate. Questions of right versus privilege, access versus affordability, and, of course, the perpetual political overlay have monopolized most of the discourse. Some contend that healthcare reform will redefine the current relationship between pharma and physicians . . . and not a moment too soon.
Lest there be ambiguity, though, the medical profession remains a noble vocation. This notwithstanding, until 2002, physicians freely participated in golf outings, received athletic tickets, and dined at five-star restaurants. But after the pharmaceutical industry smartly adopted voluntary guidelines that restrict gifting to doctors, we are left with drug samples and, of course, the “free lunch.” Certainly, pharma can claim it has made significant contributions to furthering medical education and research. Many could argue the tangible negative effects that would follow if the funding suddenly were absent.
But let’s not kid ourselves: There is a good reason the pharmaceutical industry spends more than $12 billion per year on marketing to doctors.4 In 2006, Rep. Henry Waxman (D-Calif.) said, “It is obvious that drug companies provide these free lunches so their sales reps can get the doctor’s ear and influence the prescribing practices.”2 Most doctors would never admit any such influence. It would be, however, disingenuous for any practicing physician to say there is none.
A randomized trial conducted by Adair et al concluded the “access to drug samples in clinic influences resident prescribing decisions. This could affect resident education and increase drug costs for patients.”5 An earlier study by Chew et al concluded “the availability of drug samples led physicians to dispense and subsequently prescribe drugs that differ from their preferred drug choice. Physicians most often report using drug samples to avoid cost to the patient.”6
Sure, local culture drives some prescribing practice, but one must be mindful of the reality that the pharmaceutical industry has significant influence. Plus, free drug samples help patients in the short term. Once the samples are gone, an expensive prescription for that new drug will follow. It’s another win for the industry and another loss for the patient and the healthcare system.
Many studies have shown that gifting exerts influence, even if doctors are unwilling to admit it. But patients and doctors alike would like to state with clarity of conscience that the medication prescribed is only based on clinical evidence, not influence. TH
Dr. Pyke is a hospitalist at Geisinger Wyoming Valley Medical Systems in Mountain Top, Pa.
References
- Dubovsky SL, Kaye DL, Pristach CA, DelRegno P, Pessar L, Stiles K. Can academic departments maintain industry relationships while promoting physician professionalism? Acad Med. 2010;85(1):68-73.
- Salganik MW, Hopkins JS, Rockoff JD. Medical salesmen prescribe lunches. Catering trade feeds on rep-doctor meals. The Baltimore Sun. July 29, 2006.
- Institute of Medicine Conflict of Interest in Medical Research, Education and Practice Full Recommendations. 4-28-09.
- Wolfe SM. Why do American drug companies spend more than $12 billion a year pushing drugs? Is it education or promotion? J Gen Intern Med. 2007;11(10):637-639.
- Adair RF, Holmgren LR. Do drug samples influence resident prescribing behavior? A randomized trial. Am J Med. 2005;118(8):881-884.
- Chew LD, O’Young TS, Hazlet TK, Bradley KA, Maynard C, Lessler DS. A physician survey of the effect of drug sample availability on physicians’ behavior. J Gen Intern Med. 2000;15(7):478-483.
Recent discussions on conflicts of interest in medical publications underscore the significance of the important yet fragile relationship between the pharmaceutical industry and healthcare professionals. Among these is an examination of how academic departments can maintain a relationship with the industry.1 This study suggests that if appropriate boundaries are established between industry and academia, it is possible to collaborate. However, part of the policy in this investigation included “elimination of industry-supplied meals, gifts, and favors.”2
The Institute of Medicine’s “Conflict of Interest in Medical Research, Education, and Practice” included groundbreaking recommendations.3 Among them was a call for professionals to adopt a policy that prohibits “the acceptance of items of material value from pharmaceutical, medical device, and biotechnology companies, except in specified situations.”3
Our nation has been embroiled in a healthcare debate. Questions of right versus privilege, access versus affordability, and, of course, the perpetual political overlay have monopolized most of the discourse. Some contend that healthcare reform will redefine the current relationship between pharma and physicians . . . and not a moment too soon.
Lest there be ambiguity, though, the medical profession remains a noble vocation. This notwithstanding, until 2002, physicians freely participated in golf outings, received athletic tickets, and dined at five-star restaurants. But after the pharmaceutical industry smartly adopted voluntary guidelines that restrict gifting to doctors, we are left with drug samples and, of course, the “free lunch.” Certainly, pharma can claim it has made significant contributions to furthering medical education and research. Many could argue the tangible negative effects that would follow if the funding suddenly were absent.
But let’s not kid ourselves: There is a good reason the pharmaceutical industry spends more than $12 billion per year on marketing to doctors.4 In 2006, Rep. Henry Waxman (D-Calif.) said, “It is obvious that drug companies provide these free lunches so their sales reps can get the doctor’s ear and influence the prescribing practices.”2 Most doctors would never admit any such influence. It would be, however, disingenuous for any practicing physician to say there is none.
A randomized trial conducted by Adair et al concluded the “access to drug samples in clinic influences resident prescribing decisions. This could affect resident education and increase drug costs for patients.”5 An earlier study by Chew et al concluded “the availability of drug samples led physicians to dispense and subsequently prescribe drugs that differ from their preferred drug choice. Physicians most often report using drug samples to avoid cost to the patient.”6
Sure, local culture drives some prescribing practice, but one must be mindful of the reality that the pharmaceutical industry has significant influence. Plus, free drug samples help patients in the short term. Once the samples are gone, an expensive prescription for that new drug will follow. It’s another win for the industry and another loss for the patient and the healthcare system.
Many studies have shown that gifting exerts influence, even if doctors are unwilling to admit it. But patients and doctors alike would like to state with clarity of conscience that the medication prescribed is only based on clinical evidence, not influence. TH
Dr. Pyke is a hospitalist at Geisinger Wyoming Valley Medical Systems in Mountain Top, Pa.
References
- Dubovsky SL, Kaye DL, Pristach CA, DelRegno P, Pessar L, Stiles K. Can academic departments maintain industry relationships while promoting physician professionalism? Acad Med. 2010;85(1):68-73.
- Salganik MW, Hopkins JS, Rockoff JD. Medical salesmen prescribe lunches. Catering trade feeds on rep-doctor meals. The Baltimore Sun. July 29, 2006.
- Institute of Medicine Conflict of Interest in Medical Research, Education and Practice Full Recommendations. 4-28-09.
- Wolfe SM. Why do American drug companies spend more than $12 billion a year pushing drugs? Is it education or promotion? J Gen Intern Med. 2007;11(10):637-639.
- Adair RF, Holmgren LR. Do drug samples influence resident prescribing behavior? A randomized trial. Am J Med. 2005;118(8):881-884.
- Chew LD, O’Young TS, Hazlet TK, Bradley KA, Maynard C, Lessler DS. A physician survey of the effect of drug sample availability on physicians’ behavior. J Gen Intern Med. 2000;15(7):478-483.
Recent discussions on conflicts of interest in medical publications underscore the significance of the important yet fragile relationship between the pharmaceutical industry and healthcare professionals. Among these is an examination of how academic departments can maintain a relationship with the industry.1 This study suggests that if appropriate boundaries are established between industry and academia, it is possible to collaborate. However, part of the policy in this investigation included “elimination of industry-supplied meals, gifts, and favors.”2
The Institute of Medicine’s “Conflict of Interest in Medical Research, Education, and Practice” included groundbreaking recommendations.3 Among them was a call for professionals to adopt a policy that prohibits “the acceptance of items of material value from pharmaceutical, medical device, and biotechnology companies, except in specified situations.”3
Our nation has been embroiled in a healthcare debate. Questions of right versus privilege, access versus affordability, and, of course, the perpetual political overlay have monopolized most of the discourse. Some contend that healthcare reform will redefine the current relationship between pharma and physicians . . . and not a moment too soon.
Lest there be ambiguity, though, the medical profession remains a noble vocation. This notwithstanding, until 2002, physicians freely participated in golf outings, received athletic tickets, and dined at five-star restaurants. But after the pharmaceutical industry smartly adopted voluntary guidelines that restrict gifting to doctors, we are left with drug samples and, of course, the “free lunch.” Certainly, pharma can claim it has made significant contributions to furthering medical education and research. Many could argue the tangible negative effects that would follow if the funding suddenly were absent.
But let’s not kid ourselves: There is a good reason the pharmaceutical industry spends more than $12 billion per year on marketing to doctors.4 In 2006, Rep. Henry Waxman (D-Calif.) said, “It is obvious that drug companies provide these free lunches so their sales reps can get the doctor’s ear and influence the prescribing practices.”2 Most doctors would never admit any such influence. It would be, however, disingenuous for any practicing physician to say there is none.
A randomized trial conducted by Adair et al concluded the “access to drug samples in clinic influences resident prescribing decisions. This could affect resident education and increase drug costs for patients.”5 An earlier study by Chew et al concluded “the availability of drug samples led physicians to dispense and subsequently prescribe drugs that differ from their preferred drug choice. Physicians most often report using drug samples to avoid cost to the patient.”6
Sure, local culture drives some prescribing practice, but one must be mindful of the reality that the pharmaceutical industry has significant influence. Plus, free drug samples help patients in the short term. Once the samples are gone, an expensive prescription for that new drug will follow. It’s another win for the industry and another loss for the patient and the healthcare system.
Many studies have shown that gifting exerts influence, even if doctors are unwilling to admit it. But patients and doctors alike would like to state with clarity of conscience that the medication prescribed is only based on clinical evidence, not influence. TH
Dr. Pyke is a hospitalist at Geisinger Wyoming Valley Medical Systems in Mountain Top, Pa.
References
- Dubovsky SL, Kaye DL, Pristach CA, DelRegno P, Pessar L, Stiles K. Can academic departments maintain industry relationships while promoting physician professionalism? Acad Med. 2010;85(1):68-73.
- Salganik MW, Hopkins JS, Rockoff JD. Medical salesmen prescribe lunches. Catering trade feeds on rep-doctor meals. The Baltimore Sun. July 29, 2006.
- Institute of Medicine Conflict of Interest in Medical Research, Education and Practice Full Recommendations. 4-28-09.
- Wolfe SM. Why do American drug companies spend more than $12 billion a year pushing drugs? Is it education or promotion? J Gen Intern Med. 2007;11(10):637-639.
- Adair RF, Holmgren LR. Do drug samples influence resident prescribing behavior? A randomized trial. Am J Med. 2005;118(8):881-884.
- Chew LD, O’Young TS, Hazlet TK, Bradley KA, Maynard C, Lessler DS. A physician survey of the effect of drug sample availability on physicians’ behavior. J Gen Intern Med. 2000;15(7):478-483.
Should hospitalists accept gifts from pharmaceutical, medical device, and biotech companies?
The pharmaceutical industry is big business, and its goal is to make money. If the industry can convince physicians to prescribe its medicines, then it makes more money.
Although pharmaceutical representatives brief physicians on new medications in an effort to encourage the use of their brand-name products, they also provide substantive information on the drugs that serves an educational purpose.
In the past, pharmaceutical companies—along with the medical device and biotechnology industries—showered physicians with expensive gifts, raising ethical questions about physicians’ obligation to the drug companies. Fair enough. These excessive practices were identified and curtailed—to my knowledge—some years ago.
Watchdog groups, however, have continued to call into question every suggestion of “being in the pay” of big pharma. Everything from a plastic pen to a piece of pizza is suspect. There is considerable concern that practicing clinicians are influenced by the smallest gesture, while many large medical institutions continue to accept pharmaceutical-company-funded research grants. If big-pharma investment in research does not corrupt institutions, why is it assumed that carrying a pharmaceutical pen has such a pernicious effect on clinicians?
As a corollary to this question, does anyone really want to discontinue these important research studies just because they are funded by industry dollars?
Listening to drug representatives—even being seen in the vicinity—raises the eyebrows of purists. Do we really want physicians completely divorced from all pharmaceutical company education and communication? Do we feel there is zero benefit to hearing about new medications from the company’s viewpoint?
If physicians completely shut out the representatives, it would be expected that pharmaceutical companies would direct their efforts elsewhere—most likely, to consumers. Is that a better and healthier scenario?
Clearly, there is potential for abuse in pharmaceutical gifts to physicians. The practice should be controlled and monitored. The suspicions raised by purist groups that physicians’ prescribing habits are unalterably biased after a five-minute pharmaceutical representative detail and a chicken sandwich is hyperbole. The voice of reason is silenced in the midst of the inquisition.
In the academic setting, fear of being accused of “bought bias” has physicians clearing their pockets of tainted pens and checking their desks for corrupting paraphernalia. The positive aspects of pharma-sponsored programs and medical lectures are lost for fear of appearing to be complicit with drug companies.
The Aristotelian Golden Mean is superior to extreme positions, and I submit that the best road is the center. Listen to what the drug company representatives have to say, just like you listen to a car salesman: You can learn from both—as long as you research the data and form your own opinion. TH
Dr. Brezina is a hospitalist at Durham Regional Hospital in North Carolina.
The pharmaceutical industry is big business, and its goal is to make money. If the industry can convince physicians to prescribe its medicines, then it makes more money.
Although pharmaceutical representatives brief physicians on new medications in an effort to encourage the use of their brand-name products, they also provide substantive information on the drugs that serves an educational purpose.
In the past, pharmaceutical companies—along with the medical device and biotechnology industries—showered physicians with expensive gifts, raising ethical questions about physicians’ obligation to the drug companies. Fair enough. These excessive practices were identified and curtailed—to my knowledge—some years ago.
Watchdog groups, however, have continued to call into question every suggestion of “being in the pay” of big pharma. Everything from a plastic pen to a piece of pizza is suspect. There is considerable concern that practicing clinicians are influenced by the smallest gesture, while many large medical institutions continue to accept pharmaceutical-company-funded research grants. If big-pharma investment in research does not corrupt institutions, why is it assumed that carrying a pharmaceutical pen has such a pernicious effect on clinicians?
As a corollary to this question, does anyone really want to discontinue these important research studies just because they are funded by industry dollars?
Listening to drug representatives—even being seen in the vicinity—raises the eyebrows of purists. Do we really want physicians completely divorced from all pharmaceutical company education and communication? Do we feel there is zero benefit to hearing about new medications from the company’s viewpoint?
If physicians completely shut out the representatives, it would be expected that pharmaceutical companies would direct their efforts elsewhere—most likely, to consumers. Is that a better and healthier scenario?
Clearly, there is potential for abuse in pharmaceutical gifts to physicians. The practice should be controlled and monitored. The suspicions raised by purist groups that physicians’ prescribing habits are unalterably biased after a five-minute pharmaceutical representative detail and a chicken sandwich is hyperbole. The voice of reason is silenced in the midst of the inquisition.
In the academic setting, fear of being accused of “bought bias” has physicians clearing their pockets of tainted pens and checking their desks for corrupting paraphernalia. The positive aspects of pharma-sponsored programs and medical lectures are lost for fear of appearing to be complicit with drug companies.
The Aristotelian Golden Mean is superior to extreme positions, and I submit that the best road is the center. Listen to what the drug company representatives have to say, just like you listen to a car salesman: You can learn from both—as long as you research the data and form your own opinion. TH
Dr. Brezina is a hospitalist at Durham Regional Hospital in North Carolina.
The pharmaceutical industry is big business, and its goal is to make money. If the industry can convince physicians to prescribe its medicines, then it makes more money.
Although pharmaceutical representatives brief physicians on new medications in an effort to encourage the use of their brand-name products, they also provide substantive information on the drugs that serves an educational purpose.
In the past, pharmaceutical companies—along with the medical device and biotechnology industries—showered physicians with expensive gifts, raising ethical questions about physicians’ obligation to the drug companies. Fair enough. These excessive practices were identified and curtailed—to my knowledge—some years ago.
Watchdog groups, however, have continued to call into question every suggestion of “being in the pay” of big pharma. Everything from a plastic pen to a piece of pizza is suspect. There is considerable concern that practicing clinicians are influenced by the smallest gesture, while many large medical institutions continue to accept pharmaceutical-company-funded research grants. If big-pharma investment in research does not corrupt institutions, why is it assumed that carrying a pharmaceutical pen has such a pernicious effect on clinicians?
As a corollary to this question, does anyone really want to discontinue these important research studies just because they are funded by industry dollars?
Listening to drug representatives—even being seen in the vicinity—raises the eyebrows of purists. Do we really want physicians completely divorced from all pharmaceutical company education and communication? Do we feel there is zero benefit to hearing about new medications from the company’s viewpoint?
If physicians completely shut out the representatives, it would be expected that pharmaceutical companies would direct their efforts elsewhere—most likely, to consumers. Is that a better and healthier scenario?
Clearly, there is potential for abuse in pharmaceutical gifts to physicians. The practice should be controlled and monitored. The suspicions raised by purist groups that physicians’ prescribing habits are unalterably biased after a five-minute pharmaceutical representative detail and a chicken sandwich is hyperbole. The voice of reason is silenced in the midst of the inquisition.
In the academic setting, fear of being accused of “bought bias” has physicians clearing their pockets of tainted pens and checking their desks for corrupting paraphernalia. The positive aspects of pharma-sponsored programs and medical lectures are lost for fear of appearing to be complicit with drug companies.
The Aristotelian Golden Mean is superior to extreme positions, and I submit that the best road is the center. Listen to what the drug company representatives have to say, just like you listen to a car salesman: You can learn from both—as long as you research the data and form your own opinion. TH
Dr. Brezina is a hospitalist at Durham Regional Hospital in North Carolina.
To Err is Human
The challenges facing SHM are very different than they were 10 years ago. In the 1990s, the focus was on building a society that would represent the needs of the practicing hospitalist. Converting NAIP, with its 200 members, to SHM, with its now 10,000 members, was certainly no easy task, but the society then enjoyed some luxuries no longer afforded to an organization the size of the modern-day SHM. Early on, SHM was far from the public eye, escaping public scrutiny for each of its actions. With only a few hundred members, the society was intimate: Almost every member knew of every action before it happened. And the agenda, compared with today’s standards, was reasonably focused.
But times are different now. The organization is much larger and complex, and the challenges we now face are collectively a product of our success. SHM is squarely in the spotlight; every decision is closely monitored by the public eye. We now have a voice such that when we speak, people listen. But with greatness comes responsibility, and because we are in the spotlight, we must be especially careful in how we speak, lest the message be misunderstood. Further, with more than 10,000 members, 50 full-time staff, 44 committees, and nearly 500 physician volunteers, the organization no longer has the luxury of every action being known by every member prior to its enactment.
More challenging still is our agenda, which has grown to be a diverse and far-reaching strategy. While impressive and admirable, the size of this “footprint” creates new challenges in balancing the need to be “nimble” (i.e., being able to act quickly enough to be timely and effective) versus being “thorough” (i.e., ensuring that each action is appropriately vetted prior to execution).
I suspect that there are few practicing hospitalists who have not read To Err is Human or Crossing the Quality Chasm.1,2 Both make this essential point about quality: In complex systems, mistakes are bound to happen. And when errors do occur, each member of the team must be ready to take responsibility for the mistake, and immediately begin seeking systematic solutions to ensure that it does not happen again. SHM’s focus is to advance quality for all hospitalized patients. But an organization can only be effective if it emulates the principles that it hopes its members will individually espouse.
So let me start with this: There have been mistakes along the way.
That’s the hard truth. I believe that none of the mistakes have been intentional; rather, these missteps have been a product of an organization that has grown so fast, and whose success has gained so much public attention, that its infrastructure has struggled to keep pace with its growth. Any hospitalist who has seen his or her service size double in the span of a year or two knows of what I speak: As growth occurs, the approach to dealing with daily business has to evolve to meet new demands. If it does not, errors result.
One of the areas in which SHM’s growth has outpaced its policies and procedures regards SHM’s relationship with industry. I will say from the outset that having relationships with industry is not in and of itself a mistake. The reality is that without such relationships, in the setting of a landscape where governmental and philanthropic funding is disproportionately in deficit to the need, it would be almost impossible to advance the quality initiatives that have defined SHM’s success. SHM has, and will likely continue to have, relationships with industry. But requisite for having these relationships, especially for an organization that is a national leader, is going above and beyond the minimum standards to ensure transparency and ethics.
Two years ago, SHM began the arduous process of reviewing its partnerships and how it interacts with industry. I am pleased to announce that this has culminated in the Council of Medical Specialty Societies (CMSS; www.cmss.org) asking SHM to apply to become an affiliate member. Acceptance of SHM into CMSS is evidence of SHM’s demonstrated compliance with CMSS’s requirements, with respect to industry relationships, disclosure of conflicts of interest, and other measures of organizational transparency, all of which can be found at www.hospitalmedicine.org/industry.
But meeting the minimum standards has never been sufficient for SHM. The cost of greatness is responsibility, and as a national leader, SHM has a responsibility to ensure that its approaches to potential conflicts of interest and external relationships are above reproach.
COI Disclosure
The conflict of interest statements for each board member have long been posted on the SHM website. In an effort to go above and beyond the minimum standards, the format of the disclosure form has been revised, making it the most compete and detailed COI disclosure form of any physician organization in the country. In the coming months, SHM will make even tighter restrictions regarding disclosing potential conflicts of interest. While board members are required to report any and all financial receipts, the amended version will require board members who receive any contribution in excess of $5,000 to provide a detailed narrative as to what was required in service for the receipt of those funds. Further, to ensure collective accountability, any board member may call upon any other board member to provide a similarly detailed description of any item on his or her COI disclosure form.
Recognizing that other leaders in the organization might also have influence over important decisions, thereby being at risk for a conflict of interest, SHM is one of the first physician organizations to require public reporting of COI disclosures for all editors, course directors, and senior staff.
Next year, all committee chairs and quality-improvement (QI) project leaders will be required to submit similar COI disclosures.
But reporting potential conflicts is one thing; ensuring that those with significant conflicts are not put in a position of inescapable conflict of interest by virtue of their appointments is another. To be proactive, the executive committee has a designated meeting each year to individually review each nominee being considered for election to the board, committee chairs, editors, and course directors prior to their appointment.
The society will enforce CMSS Standard 1.4, which prohibits key society leaders (president, past-president, president-elect, CEO, editors, course directors) from having direct financial relationships with companies during his or her term of service. All people seeking such positions will be required to attest, at the time of the nomination, to cease all direct financial relationships prior to seeking office; failure to do so will negate their candidacy for the position they seek.
External Communications Regarding Industry
It is one thing to have potential conflicts disclosed on a website; it is quite another to ensure, with 100% confidence, that all recipients of all communications from SHM are aware of this website. Reminding all representatives of SHM to alert communication recipients to our potential conflicts of interest is a good start, but in quality parlance, this is tantamount to “telling people to try harder,” which is rarely an effective strategy to ensure 100% compliance. In response, SHM has designed a fail-safe systems solution to ensure that every communication alerts the recipient to SHM’s potential conflicts of interest. Beginning this year, SHM letterheads and e-mail, used for all written communications with external parties, will carry the following statement on the bottom of each page: “To Learn More About SHM’s Relationship with Industry Partners, Visit www.hospitalmedicine.org/industry.”
One of SHM’s missteps over the years has been the failure to distinguish external communications regarding pharmaceuticals/devices as being different from the organization’s other nonpharmaceutical communications. This unintentional oversight has been a product of the exponential increase in the society’s external communications during the past 10 years. But nonetheless, the distinction between these types of communications is important, especially for a society that receives industry support for its quality initiatives.
At the August board meeting in Chicago, a special ad hoc committee was appointed to develop specific policies regarding SHM’s communication strategy. This committee will bring to the board in November the following policy for approval: “Before SHM makes a specific comment, writes a letter, or posts an official statement on the SHM website about a pharmaceutical agent, a medical device, a specific disease state, or any medical IT services or products, the communication must be approved by the Executive Committee and reflected in the minutes of the Executive Committee. At the President’s discretion, the proposed communication will be brought to the entire Board for discussion and approval.”
As noted below, all agendas and decisions by the executive committee are communicated to the board, further ensuring accountability and oversight for any such decision.
Choices and Definitions
In the early years, all external relationships were initiated by SHM. Because SHM was a relative unknown on the national scene, if a relationship was to be entertained, it was based on SHM’s initiative to do so. Naturally, the smaller number of relationships, and the fact that the choice and nature of the relationship were initiated by SHM, made it easier to define the scope of such relationships. But now things are different: SHM’s agenda now encompasses a vast set of domains, and SHM is regularly on the receiving end of invitations to establish relationships with other organizations. Once again, as a leader of medical specialty organizations, SHM’s policies and procedures have to adapt to fit the needs of a larger and more diverse organization.
An intense amount of work has been devoted to evolving the mechanism by which SHM chooses and defines its relationships. An ad hoc committee from the board of directors has defined the 10 principles of SHM’s business relationships (see “10 Principles of SHM Business Relationships,” p. 42). In November, the board will adopt policies and procedures that will ensure that SHM will continue to only enter into relationships with external organizations with which it shares common interests or goals for advancing the quality and safety of patient care. SHM will continue to avoid influence from external organizations with respect to the policies, conduct, actions, and priorities of SHM.
Further, by policy, SHM will continue to reserve absolute control over all content and speakers at its educational conferences; content will continue to not be influenced by brand or product consideration during development or revisions. All potential partners will be informed from the outset that a partnership with SHM does not imply that SHM endorses the policies, values, and missions of the partner organization; any significant deviation from the values and mission of SHM will dissolve the partnership. SHM will establish from the outset that a partnership does not imply SHM’s support or endorsement of any products from a partner. As noted above, transparency of these relationships will be of paramount importance: All relationships, including the dollar amounts received as a product of those relationships, will be posted on SHM’s website.
Transparency in Decision-Making
As noted from the outset, the benefit of small organizations with limited agendas is that every member knows every decision. With limited decisions to be made, the vetting and review process is virtually assured. As organizations grow, as agendas expand exponentially, and as the pace quickens, the multiperson review of each decision becomes difficult to assure. The result is that errors start to appear—not due to intentional wrongdoing, but because the luxury of intense oversight is lost as the organization expands. For an organization to grow and still maintain oversight of its decision-making process, it is vital that the organization evolve to develop new methods of accountability and transparency.
To meet this need, SHM has enacted a change in its communication infrastructure to ensure “double-checks” for all of the important organizational decisions. An SHM leadership and staff “wiki” has been developed to promote and ensure transparency of all organizational decisions. Because it is accessible only to the SHM staff, board, and committee chairs, the wiki is invisible to the SHM membership. Nonetheless, you should know of this important innovation.
The wiki requires that all committee chairs post the results of their committee activities. This ensures that staff and committee leadership are on the same page, ensures that other committees are not duplicating work, augments collaboration across committees, and, most importantly, ensures collective accountability for each decision made.
Technology-based innovations have been enacted to improve the transparency of the executive leadership of the organization. The board of directors meets four times a year; the purpose of the board is to ensure oversight for all SHM decisions. Because the board comprises exclusively volunteer members meeting only four times a year, it is practically impossible for the board to approve every decision made by an organization as large as SHM. To ensure the necessary efficiency and effectiveness (i.e., being sufficiently “nimble” to act on important issues in between scheduled board meetings), the executive committee (EC) was established years ago. The EC, comprised of the president, the past-president, the president-elect, and the CEO, meets every two weeks via teleconference to review and approve all essential SHM decisions.
As an innovation to augment accountability and transparency, the agenda and minutes of the EC are now posted on the SHM board portal, allowing all board members to review and comment upon the decisions made by the president, the CEO, or the EC as a whole. Any board member, at any time, can request that the full board be convened to review an agenda item or decision.
And underlying all of these initiatives to improve an already exceptional organization are the tireless efforts of the SHM staff. Though there are nearly 50 staff members now, each continues to do the work of multiple people. SHM is arguably the fastest-growing organization in history, and advancing the organization to level after level has been an exceptional challenge. But regardless of the challenge, SHM leadership and staff has come through. I have no doubt that during this next chapter in SHM’s history, the result will be the same.
SHM is committed to advancing quality. Intrinsic to the “culture of quality” is the commitment to honesty, transparency, and ethics. Any permutation of the society that does not fully exemplify these standards will be ineffective in accomplishing our wished-for goal. In short, the actions of the society must model those that we wish to inspire in the day-to-day practice of our members. Although the unprecedented growth of the society is responsible for errors in the past, the importance of admitting our shortcomings is no less significant. We’ve had some missteps along the way, and while these mistakes are a product of events preceding my tenure, it does not matter. As president of the organization, I am taking responsibility for them, with a pledge to devote all time and energy, with all due speed, to finding systematic solutions that will prevent these errors from happening again.
And let me be even more honest. As we go forward, there are probably going to be more missteps; such is the nature of a growing and active organization. I cannot promise an error-free organization, but I can promise that if and when errors are made in the future, the same intensity will be applied to seek systematic solutions to ensure that we continue to evolve in becoming an organization that is emblematic of quality. Such is the promise of SHM; such is the promise of the hospitalist. TH
Dr. Wiese is president of SHM.
References
- Kohn LT, Corrigan JM, Donaldson MS, et al. To Err Is Human: Building a Safer Health System. Washington, D.C.: National Academies Press; 2000.
- Institute of Medicine Committee on Quality Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, D.C.: National Academies Press; 2001.
The challenges facing SHM are very different than they were 10 years ago. In the 1990s, the focus was on building a society that would represent the needs of the practicing hospitalist. Converting NAIP, with its 200 members, to SHM, with its now 10,000 members, was certainly no easy task, but the society then enjoyed some luxuries no longer afforded to an organization the size of the modern-day SHM. Early on, SHM was far from the public eye, escaping public scrutiny for each of its actions. With only a few hundred members, the society was intimate: Almost every member knew of every action before it happened. And the agenda, compared with today’s standards, was reasonably focused.
But times are different now. The organization is much larger and complex, and the challenges we now face are collectively a product of our success. SHM is squarely in the spotlight; every decision is closely monitored by the public eye. We now have a voice such that when we speak, people listen. But with greatness comes responsibility, and because we are in the spotlight, we must be especially careful in how we speak, lest the message be misunderstood. Further, with more than 10,000 members, 50 full-time staff, 44 committees, and nearly 500 physician volunteers, the organization no longer has the luxury of every action being known by every member prior to its enactment.
More challenging still is our agenda, which has grown to be a diverse and far-reaching strategy. While impressive and admirable, the size of this “footprint” creates new challenges in balancing the need to be “nimble” (i.e., being able to act quickly enough to be timely and effective) versus being “thorough” (i.e., ensuring that each action is appropriately vetted prior to execution).
I suspect that there are few practicing hospitalists who have not read To Err is Human or Crossing the Quality Chasm.1,2 Both make this essential point about quality: In complex systems, mistakes are bound to happen. And when errors do occur, each member of the team must be ready to take responsibility for the mistake, and immediately begin seeking systematic solutions to ensure that it does not happen again. SHM’s focus is to advance quality for all hospitalized patients. But an organization can only be effective if it emulates the principles that it hopes its members will individually espouse.
So let me start with this: There have been mistakes along the way.
That’s the hard truth. I believe that none of the mistakes have been intentional; rather, these missteps have been a product of an organization that has grown so fast, and whose success has gained so much public attention, that its infrastructure has struggled to keep pace with its growth. Any hospitalist who has seen his or her service size double in the span of a year or two knows of what I speak: As growth occurs, the approach to dealing with daily business has to evolve to meet new demands. If it does not, errors result.
One of the areas in which SHM’s growth has outpaced its policies and procedures regards SHM’s relationship with industry. I will say from the outset that having relationships with industry is not in and of itself a mistake. The reality is that without such relationships, in the setting of a landscape where governmental and philanthropic funding is disproportionately in deficit to the need, it would be almost impossible to advance the quality initiatives that have defined SHM’s success. SHM has, and will likely continue to have, relationships with industry. But requisite for having these relationships, especially for an organization that is a national leader, is going above and beyond the minimum standards to ensure transparency and ethics.
Two years ago, SHM began the arduous process of reviewing its partnerships and how it interacts with industry. I am pleased to announce that this has culminated in the Council of Medical Specialty Societies (CMSS; www.cmss.org) asking SHM to apply to become an affiliate member. Acceptance of SHM into CMSS is evidence of SHM’s demonstrated compliance with CMSS’s requirements, with respect to industry relationships, disclosure of conflicts of interest, and other measures of organizational transparency, all of which can be found at www.hospitalmedicine.org/industry.
But meeting the minimum standards has never been sufficient for SHM. The cost of greatness is responsibility, and as a national leader, SHM has a responsibility to ensure that its approaches to potential conflicts of interest and external relationships are above reproach.
COI Disclosure
The conflict of interest statements for each board member have long been posted on the SHM website. In an effort to go above and beyond the minimum standards, the format of the disclosure form has been revised, making it the most compete and detailed COI disclosure form of any physician organization in the country. In the coming months, SHM will make even tighter restrictions regarding disclosing potential conflicts of interest. While board members are required to report any and all financial receipts, the amended version will require board members who receive any contribution in excess of $5,000 to provide a detailed narrative as to what was required in service for the receipt of those funds. Further, to ensure collective accountability, any board member may call upon any other board member to provide a similarly detailed description of any item on his or her COI disclosure form.
Recognizing that other leaders in the organization might also have influence over important decisions, thereby being at risk for a conflict of interest, SHM is one of the first physician organizations to require public reporting of COI disclosures for all editors, course directors, and senior staff.
Next year, all committee chairs and quality-improvement (QI) project leaders will be required to submit similar COI disclosures.
But reporting potential conflicts is one thing; ensuring that those with significant conflicts are not put in a position of inescapable conflict of interest by virtue of their appointments is another. To be proactive, the executive committee has a designated meeting each year to individually review each nominee being considered for election to the board, committee chairs, editors, and course directors prior to their appointment.
The society will enforce CMSS Standard 1.4, which prohibits key society leaders (president, past-president, president-elect, CEO, editors, course directors) from having direct financial relationships with companies during his or her term of service. All people seeking such positions will be required to attest, at the time of the nomination, to cease all direct financial relationships prior to seeking office; failure to do so will negate their candidacy for the position they seek.
External Communications Regarding Industry
It is one thing to have potential conflicts disclosed on a website; it is quite another to ensure, with 100% confidence, that all recipients of all communications from SHM are aware of this website. Reminding all representatives of SHM to alert communication recipients to our potential conflicts of interest is a good start, but in quality parlance, this is tantamount to “telling people to try harder,” which is rarely an effective strategy to ensure 100% compliance. In response, SHM has designed a fail-safe systems solution to ensure that every communication alerts the recipient to SHM’s potential conflicts of interest. Beginning this year, SHM letterheads and e-mail, used for all written communications with external parties, will carry the following statement on the bottom of each page: “To Learn More About SHM’s Relationship with Industry Partners, Visit www.hospitalmedicine.org/industry.”
One of SHM’s missteps over the years has been the failure to distinguish external communications regarding pharmaceuticals/devices as being different from the organization’s other nonpharmaceutical communications. This unintentional oversight has been a product of the exponential increase in the society’s external communications during the past 10 years. But nonetheless, the distinction between these types of communications is important, especially for a society that receives industry support for its quality initiatives.
At the August board meeting in Chicago, a special ad hoc committee was appointed to develop specific policies regarding SHM’s communication strategy. This committee will bring to the board in November the following policy for approval: “Before SHM makes a specific comment, writes a letter, or posts an official statement on the SHM website about a pharmaceutical agent, a medical device, a specific disease state, or any medical IT services or products, the communication must be approved by the Executive Committee and reflected in the minutes of the Executive Committee. At the President’s discretion, the proposed communication will be brought to the entire Board for discussion and approval.”
As noted below, all agendas and decisions by the executive committee are communicated to the board, further ensuring accountability and oversight for any such decision.
Choices and Definitions
In the early years, all external relationships were initiated by SHM. Because SHM was a relative unknown on the national scene, if a relationship was to be entertained, it was based on SHM’s initiative to do so. Naturally, the smaller number of relationships, and the fact that the choice and nature of the relationship were initiated by SHM, made it easier to define the scope of such relationships. But now things are different: SHM’s agenda now encompasses a vast set of domains, and SHM is regularly on the receiving end of invitations to establish relationships with other organizations. Once again, as a leader of medical specialty organizations, SHM’s policies and procedures have to adapt to fit the needs of a larger and more diverse organization.
An intense amount of work has been devoted to evolving the mechanism by which SHM chooses and defines its relationships. An ad hoc committee from the board of directors has defined the 10 principles of SHM’s business relationships (see “10 Principles of SHM Business Relationships,” p. 42). In November, the board will adopt policies and procedures that will ensure that SHM will continue to only enter into relationships with external organizations with which it shares common interests or goals for advancing the quality and safety of patient care. SHM will continue to avoid influence from external organizations with respect to the policies, conduct, actions, and priorities of SHM.
Further, by policy, SHM will continue to reserve absolute control over all content and speakers at its educational conferences; content will continue to not be influenced by brand or product consideration during development or revisions. All potential partners will be informed from the outset that a partnership with SHM does not imply that SHM endorses the policies, values, and missions of the partner organization; any significant deviation from the values and mission of SHM will dissolve the partnership. SHM will establish from the outset that a partnership does not imply SHM’s support or endorsement of any products from a partner. As noted above, transparency of these relationships will be of paramount importance: All relationships, including the dollar amounts received as a product of those relationships, will be posted on SHM’s website.
Transparency in Decision-Making
As noted from the outset, the benefit of small organizations with limited agendas is that every member knows every decision. With limited decisions to be made, the vetting and review process is virtually assured. As organizations grow, as agendas expand exponentially, and as the pace quickens, the multiperson review of each decision becomes difficult to assure. The result is that errors start to appear—not due to intentional wrongdoing, but because the luxury of intense oversight is lost as the organization expands. For an organization to grow and still maintain oversight of its decision-making process, it is vital that the organization evolve to develop new methods of accountability and transparency.
To meet this need, SHM has enacted a change in its communication infrastructure to ensure “double-checks” for all of the important organizational decisions. An SHM leadership and staff “wiki” has been developed to promote and ensure transparency of all organizational decisions. Because it is accessible only to the SHM staff, board, and committee chairs, the wiki is invisible to the SHM membership. Nonetheless, you should know of this important innovation.
The wiki requires that all committee chairs post the results of their committee activities. This ensures that staff and committee leadership are on the same page, ensures that other committees are not duplicating work, augments collaboration across committees, and, most importantly, ensures collective accountability for each decision made.
Technology-based innovations have been enacted to improve the transparency of the executive leadership of the organization. The board of directors meets four times a year; the purpose of the board is to ensure oversight for all SHM decisions. Because the board comprises exclusively volunteer members meeting only four times a year, it is practically impossible for the board to approve every decision made by an organization as large as SHM. To ensure the necessary efficiency and effectiveness (i.e., being sufficiently “nimble” to act on important issues in between scheduled board meetings), the executive committee (EC) was established years ago. The EC, comprised of the president, the past-president, the president-elect, and the CEO, meets every two weeks via teleconference to review and approve all essential SHM decisions.
As an innovation to augment accountability and transparency, the agenda and minutes of the EC are now posted on the SHM board portal, allowing all board members to review and comment upon the decisions made by the president, the CEO, or the EC as a whole. Any board member, at any time, can request that the full board be convened to review an agenda item or decision.
And underlying all of these initiatives to improve an already exceptional organization are the tireless efforts of the SHM staff. Though there are nearly 50 staff members now, each continues to do the work of multiple people. SHM is arguably the fastest-growing organization in history, and advancing the organization to level after level has been an exceptional challenge. But regardless of the challenge, SHM leadership and staff has come through. I have no doubt that during this next chapter in SHM’s history, the result will be the same.
SHM is committed to advancing quality. Intrinsic to the “culture of quality” is the commitment to honesty, transparency, and ethics. Any permutation of the society that does not fully exemplify these standards will be ineffective in accomplishing our wished-for goal. In short, the actions of the society must model those that we wish to inspire in the day-to-day practice of our members. Although the unprecedented growth of the society is responsible for errors in the past, the importance of admitting our shortcomings is no less significant. We’ve had some missteps along the way, and while these mistakes are a product of events preceding my tenure, it does not matter. As president of the organization, I am taking responsibility for them, with a pledge to devote all time and energy, with all due speed, to finding systematic solutions that will prevent these errors from happening again.
And let me be even more honest. As we go forward, there are probably going to be more missteps; such is the nature of a growing and active organization. I cannot promise an error-free organization, but I can promise that if and when errors are made in the future, the same intensity will be applied to seek systematic solutions to ensure that we continue to evolve in becoming an organization that is emblematic of quality. Such is the promise of SHM; such is the promise of the hospitalist. TH
Dr. Wiese is president of SHM.
References
- Kohn LT, Corrigan JM, Donaldson MS, et al. To Err Is Human: Building a Safer Health System. Washington, D.C.: National Academies Press; 2000.
- Institute of Medicine Committee on Quality Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, D.C.: National Academies Press; 2001.
The challenges facing SHM are very different than they were 10 years ago. In the 1990s, the focus was on building a society that would represent the needs of the practicing hospitalist. Converting NAIP, with its 200 members, to SHM, with its now 10,000 members, was certainly no easy task, but the society then enjoyed some luxuries no longer afforded to an organization the size of the modern-day SHM. Early on, SHM was far from the public eye, escaping public scrutiny for each of its actions. With only a few hundred members, the society was intimate: Almost every member knew of every action before it happened. And the agenda, compared with today’s standards, was reasonably focused.
But times are different now. The organization is much larger and complex, and the challenges we now face are collectively a product of our success. SHM is squarely in the spotlight; every decision is closely monitored by the public eye. We now have a voice such that when we speak, people listen. But with greatness comes responsibility, and because we are in the spotlight, we must be especially careful in how we speak, lest the message be misunderstood. Further, with more than 10,000 members, 50 full-time staff, 44 committees, and nearly 500 physician volunteers, the organization no longer has the luxury of every action being known by every member prior to its enactment.
More challenging still is our agenda, which has grown to be a diverse and far-reaching strategy. While impressive and admirable, the size of this “footprint” creates new challenges in balancing the need to be “nimble” (i.e., being able to act quickly enough to be timely and effective) versus being “thorough” (i.e., ensuring that each action is appropriately vetted prior to execution).
I suspect that there are few practicing hospitalists who have not read To Err is Human or Crossing the Quality Chasm.1,2 Both make this essential point about quality: In complex systems, mistakes are bound to happen. And when errors do occur, each member of the team must be ready to take responsibility for the mistake, and immediately begin seeking systematic solutions to ensure that it does not happen again. SHM’s focus is to advance quality for all hospitalized patients. But an organization can only be effective if it emulates the principles that it hopes its members will individually espouse.
So let me start with this: There have been mistakes along the way.
That’s the hard truth. I believe that none of the mistakes have been intentional; rather, these missteps have been a product of an organization that has grown so fast, and whose success has gained so much public attention, that its infrastructure has struggled to keep pace with its growth. Any hospitalist who has seen his or her service size double in the span of a year or two knows of what I speak: As growth occurs, the approach to dealing with daily business has to evolve to meet new demands. If it does not, errors result.
One of the areas in which SHM’s growth has outpaced its policies and procedures regards SHM’s relationship with industry. I will say from the outset that having relationships with industry is not in and of itself a mistake. The reality is that without such relationships, in the setting of a landscape where governmental and philanthropic funding is disproportionately in deficit to the need, it would be almost impossible to advance the quality initiatives that have defined SHM’s success. SHM has, and will likely continue to have, relationships with industry. But requisite for having these relationships, especially for an organization that is a national leader, is going above and beyond the minimum standards to ensure transparency and ethics.
Two years ago, SHM began the arduous process of reviewing its partnerships and how it interacts with industry. I am pleased to announce that this has culminated in the Council of Medical Specialty Societies (CMSS; www.cmss.org) asking SHM to apply to become an affiliate member. Acceptance of SHM into CMSS is evidence of SHM’s demonstrated compliance with CMSS’s requirements, with respect to industry relationships, disclosure of conflicts of interest, and other measures of organizational transparency, all of which can be found at www.hospitalmedicine.org/industry.
But meeting the minimum standards has never been sufficient for SHM. The cost of greatness is responsibility, and as a national leader, SHM has a responsibility to ensure that its approaches to potential conflicts of interest and external relationships are above reproach.
COI Disclosure
The conflict of interest statements for each board member have long been posted on the SHM website. In an effort to go above and beyond the minimum standards, the format of the disclosure form has been revised, making it the most compete and detailed COI disclosure form of any physician organization in the country. In the coming months, SHM will make even tighter restrictions regarding disclosing potential conflicts of interest. While board members are required to report any and all financial receipts, the amended version will require board members who receive any contribution in excess of $5,000 to provide a detailed narrative as to what was required in service for the receipt of those funds. Further, to ensure collective accountability, any board member may call upon any other board member to provide a similarly detailed description of any item on his or her COI disclosure form.
Recognizing that other leaders in the organization might also have influence over important decisions, thereby being at risk for a conflict of interest, SHM is one of the first physician organizations to require public reporting of COI disclosures for all editors, course directors, and senior staff.
Next year, all committee chairs and quality-improvement (QI) project leaders will be required to submit similar COI disclosures.
But reporting potential conflicts is one thing; ensuring that those with significant conflicts are not put in a position of inescapable conflict of interest by virtue of their appointments is another. To be proactive, the executive committee has a designated meeting each year to individually review each nominee being considered for election to the board, committee chairs, editors, and course directors prior to their appointment.
The society will enforce CMSS Standard 1.4, which prohibits key society leaders (president, past-president, president-elect, CEO, editors, course directors) from having direct financial relationships with companies during his or her term of service. All people seeking such positions will be required to attest, at the time of the nomination, to cease all direct financial relationships prior to seeking office; failure to do so will negate their candidacy for the position they seek.
External Communications Regarding Industry
It is one thing to have potential conflicts disclosed on a website; it is quite another to ensure, with 100% confidence, that all recipients of all communications from SHM are aware of this website. Reminding all representatives of SHM to alert communication recipients to our potential conflicts of interest is a good start, but in quality parlance, this is tantamount to “telling people to try harder,” which is rarely an effective strategy to ensure 100% compliance. In response, SHM has designed a fail-safe systems solution to ensure that every communication alerts the recipient to SHM’s potential conflicts of interest. Beginning this year, SHM letterheads and e-mail, used for all written communications with external parties, will carry the following statement on the bottom of each page: “To Learn More About SHM’s Relationship with Industry Partners, Visit www.hospitalmedicine.org/industry.”
One of SHM’s missteps over the years has been the failure to distinguish external communications regarding pharmaceuticals/devices as being different from the organization’s other nonpharmaceutical communications. This unintentional oversight has been a product of the exponential increase in the society’s external communications during the past 10 years. But nonetheless, the distinction between these types of communications is important, especially for a society that receives industry support for its quality initiatives.
At the August board meeting in Chicago, a special ad hoc committee was appointed to develop specific policies regarding SHM’s communication strategy. This committee will bring to the board in November the following policy for approval: “Before SHM makes a specific comment, writes a letter, or posts an official statement on the SHM website about a pharmaceutical agent, a medical device, a specific disease state, or any medical IT services or products, the communication must be approved by the Executive Committee and reflected in the minutes of the Executive Committee. At the President’s discretion, the proposed communication will be brought to the entire Board for discussion and approval.”
As noted below, all agendas and decisions by the executive committee are communicated to the board, further ensuring accountability and oversight for any such decision.
Choices and Definitions
In the early years, all external relationships were initiated by SHM. Because SHM was a relative unknown on the national scene, if a relationship was to be entertained, it was based on SHM’s initiative to do so. Naturally, the smaller number of relationships, and the fact that the choice and nature of the relationship were initiated by SHM, made it easier to define the scope of such relationships. But now things are different: SHM’s agenda now encompasses a vast set of domains, and SHM is regularly on the receiving end of invitations to establish relationships with other organizations. Once again, as a leader of medical specialty organizations, SHM’s policies and procedures have to adapt to fit the needs of a larger and more diverse organization.
An intense amount of work has been devoted to evolving the mechanism by which SHM chooses and defines its relationships. An ad hoc committee from the board of directors has defined the 10 principles of SHM’s business relationships (see “10 Principles of SHM Business Relationships,” p. 42). In November, the board will adopt policies and procedures that will ensure that SHM will continue to only enter into relationships with external organizations with which it shares common interests or goals for advancing the quality and safety of patient care. SHM will continue to avoid influence from external organizations with respect to the policies, conduct, actions, and priorities of SHM.
Further, by policy, SHM will continue to reserve absolute control over all content and speakers at its educational conferences; content will continue to not be influenced by brand or product consideration during development or revisions. All potential partners will be informed from the outset that a partnership with SHM does not imply that SHM endorses the policies, values, and missions of the partner organization; any significant deviation from the values and mission of SHM will dissolve the partnership. SHM will establish from the outset that a partnership does not imply SHM’s support or endorsement of any products from a partner. As noted above, transparency of these relationships will be of paramount importance: All relationships, including the dollar amounts received as a product of those relationships, will be posted on SHM’s website.
Transparency in Decision-Making
As noted from the outset, the benefit of small organizations with limited agendas is that every member knows every decision. With limited decisions to be made, the vetting and review process is virtually assured. As organizations grow, as agendas expand exponentially, and as the pace quickens, the multiperson review of each decision becomes difficult to assure. The result is that errors start to appear—not due to intentional wrongdoing, but because the luxury of intense oversight is lost as the organization expands. For an organization to grow and still maintain oversight of its decision-making process, it is vital that the organization evolve to develop new methods of accountability and transparency.
To meet this need, SHM has enacted a change in its communication infrastructure to ensure “double-checks” for all of the important organizational decisions. An SHM leadership and staff “wiki” has been developed to promote and ensure transparency of all organizational decisions. Because it is accessible only to the SHM staff, board, and committee chairs, the wiki is invisible to the SHM membership. Nonetheless, you should know of this important innovation.
The wiki requires that all committee chairs post the results of their committee activities. This ensures that staff and committee leadership are on the same page, ensures that other committees are not duplicating work, augments collaboration across committees, and, most importantly, ensures collective accountability for each decision made.
Technology-based innovations have been enacted to improve the transparency of the executive leadership of the organization. The board of directors meets four times a year; the purpose of the board is to ensure oversight for all SHM decisions. Because the board comprises exclusively volunteer members meeting only four times a year, it is practically impossible for the board to approve every decision made by an organization as large as SHM. To ensure the necessary efficiency and effectiveness (i.e., being sufficiently “nimble” to act on important issues in between scheduled board meetings), the executive committee (EC) was established years ago. The EC, comprised of the president, the past-president, the president-elect, and the CEO, meets every two weeks via teleconference to review and approve all essential SHM decisions.
As an innovation to augment accountability and transparency, the agenda and minutes of the EC are now posted on the SHM board portal, allowing all board members to review and comment upon the decisions made by the president, the CEO, or the EC as a whole. Any board member, at any time, can request that the full board be convened to review an agenda item or decision.
And underlying all of these initiatives to improve an already exceptional organization are the tireless efforts of the SHM staff. Though there are nearly 50 staff members now, each continues to do the work of multiple people. SHM is arguably the fastest-growing organization in history, and advancing the organization to level after level has been an exceptional challenge. But regardless of the challenge, SHM leadership and staff has come through. I have no doubt that during this next chapter in SHM’s history, the result will be the same.
SHM is committed to advancing quality. Intrinsic to the “culture of quality” is the commitment to honesty, transparency, and ethics. Any permutation of the society that does not fully exemplify these standards will be ineffective in accomplishing our wished-for goal. In short, the actions of the society must model those that we wish to inspire in the day-to-day practice of our members. Although the unprecedented growth of the society is responsible for errors in the past, the importance of admitting our shortcomings is no less significant. We’ve had some missteps along the way, and while these mistakes are a product of events preceding my tenure, it does not matter. As president of the organization, I am taking responsibility for them, with a pledge to devote all time and energy, with all due speed, to finding systematic solutions that will prevent these errors from happening again.
And let me be even more honest. As we go forward, there are probably going to be more missteps; such is the nature of a growing and active organization. I cannot promise an error-free organization, but I can promise that if and when errors are made in the future, the same intensity will be applied to seek systematic solutions to ensure that we continue to evolve in becoming an organization that is emblematic of quality. Such is the promise of SHM; such is the promise of the hospitalist. TH
Dr. Wiese is president of SHM.
References
- Kohn LT, Corrigan JM, Donaldson MS, et al. To Err Is Human: Building a Safer Health System. Washington, D.C.: National Academies Press; 2000.
- Institute of Medicine Committee on Quality Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, D.C.: National Academies Press; 2001.
Playground Politics
Baseball, kick the can, Russian roulette—pick your game. Chances are good that it has worked its way into a metaphor to illustrate the infuriating, perplexing, and altogether frustrating inability of Congress to step up to the plate and pass a long-term fix to the broken sustainable growth rate (SGR) formula used to determine Medicare reimbursement rates.
On June 24, legislators avoided catastrophe by temporarily rescinding a 21.3% rate cut that went into effect June 1. The after-the-fact patch meant that some Medicare claims had to be reprocessed to recoup the full value, creating an administrative mess. The accompanying 2.2% rate increase expires Nov. 30. The reimbursement cut could reach nearly 30% next year unless Congress intervenes again.
“Obviously, there’s a lot of frustration around the issue, especially on the membership side,” says Ron Greeno, MD, FACP, SFHM, a member of SHM’s Public Policy and Leadership committees, and chief medical officer for Brentwood, Tenn.-based Cogent Healthcare. For hospitalists in many small private practices, he says, a major percentage of income comes from Medicare. “It’s a tremendous headache,” he says of the uncertainty. “It’s very hard to plan for. You’re trying to budget and you don’t know what the policy is going to be literally from week to week.”
The Blame Game
Despite the widespread sentiment among doctors that a permanent reimbursement rate fix should have been included in the healthcare reform legislation, skittishness over the price tag led legislators to drop it from the package. Based on last fall’s estimates, the total cost of a reform bill that scrapped the SGR would have ballooned by roughly $250 billion over 10 years, which would have threatened the bill’s passage.
But Congress has since been unable to pass a permanent fix as standalone legislation amid mounting concern over the national debt, and the price of inaction continues to rise. On April 30, the Congressional Budget Office (CBO) estimated that the cost of jettisoning the SGR formula and freezing rates at current levels had grown to $276 billion over 10 years.
Any serious consideration of lasting alternatives has now been pushed back to the lame-duck session, after the midterm elections. The can has been kicked down the road so many times, Dr. Greeno and others say, that most Congressional members have boot marks all over them. “So now you have a bigger problem at a more crucial time, when money is tighter than ever in a poor economy,” Dr. Greeno says. “And I just think it’s been a failure of our politicians.”
Other healthcare industry leaders have been just as critical. “Delaying the problem is not a solution,” said AMA President Cecil B. Wilson, MD, in a prepared statement after Congress passed the latest six-month reprieve in June. “It doesn’t solve the Medicare mess Congress has created with a long series of short-term Medicare patches over the last decade—including four to avert the 2010 cut alone.”
AMA-sponsored print ads have reminded legislators that delaying a fix until 2013 will again increase its cost, to $396 billion over 10 years. And the association’s June press release asserted that “Congress is playing a dangerous game of Russian roulette with seniors’ healthcare.”
Perhaps a game of “chicken” would be more apt.
Republicans have dared Democrats to spend the billions for a more lasting solution—in the absence of any cuts elsewhere in the healthcare delivery system—and be labeled as fiscally irresponsible. In turn, Democrats have dared Republicans to let the rate cut take effect and be labeled heartless as Medicare beneficiaries lose access to their healthcare providers.
Both parties blinked, resorting to almost unanimous short-term fixes that have allowed legislators to save face while putting off politically risky votes until after the November elections.
Lynne M. Allen, MN, ARNP, who works as a part-time hospitalist in hematology-oncology at 188-bed Kadlec Regional Medical Center in Richland, Wash., says she and other colleagues were initially hopeful that the Obama administration would make Congress work together to find a lasting solution. “There’s a sense of frustration because instead of that happening from our legislators, they’re playing a lot of games with the funding,” says Allen, a member of Team Hospitalist. “They’re not willing to step up to the plate, as they say, and make a decision that will allow us to go forward smoothly.”
The result, Allen says, has been a “roller-coaster ride” of uncertainty over reimbursements. Because Washington’s Tri-Cities region has a relatively high percentage of patients with private insurance, her hospital is somewhat cushioned from a precipitous drop in Medicare fees. But if CMS is ever forced to cut back on its rates, she fully expects private insurers to follow the same downward track.
Practical Concerns
Barbara Hartley, MD, a part-time hospitalist at 22-bed Benson Hospital in Benson, Ariz., says the town’s healthcare facility is somewhat protected from potential Medicare rate cuts through its official status as a Critical Access Hospital. Instead of being reimbursed through diagnosis-related group (DRG) codes, the rural hospital is repaid by Medicare for its total cost per day per patient.
The arrangement is a stable one at the moment, but not enough to dispel Dr. Hartley’s uneasy question: If the economy worsens, will Medicare be able to retain its commitment to rural hospitals? If not, the pain might be felt acutely in communities like Benson, where Dr. Hartley estimates that as much as 75% of the hospital’s in-patient business is through either Medicare or a Medicare Advantage plan.
Kirk Mathews, CEO of St. Louis-based Inpatient Management Inc. and a member of SHM’s Public Policy and Practice Management committees, says Medicare rate cuts also could significantly reduce the leverage of hospitalists during contract negotiations.
“Even if we’re employed by the hospital, but our professional fees that the hospital can recoup for our services are dramatically affected, it will affect how those future contracts go,” Mathews says. “We might be insulated temporarily by the strength of our current contract. But if the formula—however that works out—dramatically impacts the hospitalist reimbursement on the professional fee side, the hospital will feel that, and then hospitalists will eventually feel that as well.” In other words, it could strengthen the bargaining hand of the hospital at the expense of the hospitalist. “Therein lies the long-term threat,” he points out.
Independent Solution?
Some of the authority over physician payments might eventually be depoliticized via language in the reform legislation that empowers a new entity, the Independent Payment Advisory Board, to create policy on such critical monetary issues as reimbursement rates. Congress could still override the board’s policy decisions, but only if the Congressional alternative saves just as much money.
In the meantime, the money for a fix still has to come from somewhere, and no consensus has emerged. Advocates likewise refuse to coalesce around any single alternative. Some experts favor a new formula based on the Medicare economic index, which measures inflation in healthcare delivery costs. But the CBO estimates that per-beneficiary spending under such a formula would be 30% more by 2016 than under the current formula. Other proposals call for temporarily increasing rates, then reverting to annual GDP growth, plus a bit more to cover physician costs.
No matter how the crisis is resolved, experts say, doctors almost certainly will have to make do with less. “When healthcare reform is finally fully implemented, there are going to be less dollars to pay for more services. It’s inevitable,” Mathews says. “And whether it takes the form of SGR or some other form, I’m afraid physicians are going to have to get used to having less money in the pool of money that’s allocated to pay providers.”
It could be a whole new ballgame. TH
Bryn Nelson, PhD, is a freelance medical writer based in Seattle.
Baseball, kick the can, Russian roulette—pick your game. Chances are good that it has worked its way into a metaphor to illustrate the infuriating, perplexing, and altogether frustrating inability of Congress to step up to the plate and pass a long-term fix to the broken sustainable growth rate (SGR) formula used to determine Medicare reimbursement rates.
On June 24, legislators avoided catastrophe by temporarily rescinding a 21.3% rate cut that went into effect June 1. The after-the-fact patch meant that some Medicare claims had to be reprocessed to recoup the full value, creating an administrative mess. The accompanying 2.2% rate increase expires Nov. 30. The reimbursement cut could reach nearly 30% next year unless Congress intervenes again.
“Obviously, there’s a lot of frustration around the issue, especially on the membership side,” says Ron Greeno, MD, FACP, SFHM, a member of SHM’s Public Policy and Leadership committees, and chief medical officer for Brentwood, Tenn.-based Cogent Healthcare. For hospitalists in many small private practices, he says, a major percentage of income comes from Medicare. “It’s a tremendous headache,” he says of the uncertainty. “It’s very hard to plan for. You’re trying to budget and you don’t know what the policy is going to be literally from week to week.”
The Blame Game
Despite the widespread sentiment among doctors that a permanent reimbursement rate fix should have been included in the healthcare reform legislation, skittishness over the price tag led legislators to drop it from the package. Based on last fall’s estimates, the total cost of a reform bill that scrapped the SGR would have ballooned by roughly $250 billion over 10 years, which would have threatened the bill’s passage.
But Congress has since been unable to pass a permanent fix as standalone legislation amid mounting concern over the national debt, and the price of inaction continues to rise. On April 30, the Congressional Budget Office (CBO) estimated that the cost of jettisoning the SGR formula and freezing rates at current levels had grown to $276 billion over 10 years.
Any serious consideration of lasting alternatives has now been pushed back to the lame-duck session, after the midterm elections. The can has been kicked down the road so many times, Dr. Greeno and others say, that most Congressional members have boot marks all over them. “So now you have a bigger problem at a more crucial time, when money is tighter than ever in a poor economy,” Dr. Greeno says. “And I just think it’s been a failure of our politicians.”
Other healthcare industry leaders have been just as critical. “Delaying the problem is not a solution,” said AMA President Cecil B. Wilson, MD, in a prepared statement after Congress passed the latest six-month reprieve in June. “It doesn’t solve the Medicare mess Congress has created with a long series of short-term Medicare patches over the last decade—including four to avert the 2010 cut alone.”
AMA-sponsored print ads have reminded legislators that delaying a fix until 2013 will again increase its cost, to $396 billion over 10 years. And the association’s June press release asserted that “Congress is playing a dangerous game of Russian roulette with seniors’ healthcare.”
Perhaps a game of “chicken” would be more apt.
Republicans have dared Democrats to spend the billions for a more lasting solution—in the absence of any cuts elsewhere in the healthcare delivery system—and be labeled as fiscally irresponsible. In turn, Democrats have dared Republicans to let the rate cut take effect and be labeled heartless as Medicare beneficiaries lose access to their healthcare providers.
Both parties blinked, resorting to almost unanimous short-term fixes that have allowed legislators to save face while putting off politically risky votes until after the November elections.
Lynne M. Allen, MN, ARNP, who works as a part-time hospitalist in hematology-oncology at 188-bed Kadlec Regional Medical Center in Richland, Wash., says she and other colleagues were initially hopeful that the Obama administration would make Congress work together to find a lasting solution. “There’s a sense of frustration because instead of that happening from our legislators, they’re playing a lot of games with the funding,” says Allen, a member of Team Hospitalist. “They’re not willing to step up to the plate, as they say, and make a decision that will allow us to go forward smoothly.”
The result, Allen says, has been a “roller-coaster ride” of uncertainty over reimbursements. Because Washington’s Tri-Cities region has a relatively high percentage of patients with private insurance, her hospital is somewhat cushioned from a precipitous drop in Medicare fees. But if CMS is ever forced to cut back on its rates, she fully expects private insurers to follow the same downward track.
Practical Concerns
Barbara Hartley, MD, a part-time hospitalist at 22-bed Benson Hospital in Benson, Ariz., says the town’s healthcare facility is somewhat protected from potential Medicare rate cuts through its official status as a Critical Access Hospital. Instead of being reimbursed through diagnosis-related group (DRG) codes, the rural hospital is repaid by Medicare for its total cost per day per patient.
The arrangement is a stable one at the moment, but not enough to dispel Dr. Hartley’s uneasy question: If the economy worsens, will Medicare be able to retain its commitment to rural hospitals? If not, the pain might be felt acutely in communities like Benson, where Dr. Hartley estimates that as much as 75% of the hospital’s in-patient business is through either Medicare or a Medicare Advantage plan.
Kirk Mathews, CEO of St. Louis-based Inpatient Management Inc. and a member of SHM’s Public Policy and Practice Management committees, says Medicare rate cuts also could significantly reduce the leverage of hospitalists during contract negotiations.
“Even if we’re employed by the hospital, but our professional fees that the hospital can recoup for our services are dramatically affected, it will affect how those future contracts go,” Mathews says. “We might be insulated temporarily by the strength of our current contract. But if the formula—however that works out—dramatically impacts the hospitalist reimbursement on the professional fee side, the hospital will feel that, and then hospitalists will eventually feel that as well.” In other words, it could strengthen the bargaining hand of the hospital at the expense of the hospitalist. “Therein lies the long-term threat,” he points out.
Independent Solution?
Some of the authority over physician payments might eventually be depoliticized via language in the reform legislation that empowers a new entity, the Independent Payment Advisory Board, to create policy on such critical monetary issues as reimbursement rates. Congress could still override the board’s policy decisions, but only if the Congressional alternative saves just as much money.
In the meantime, the money for a fix still has to come from somewhere, and no consensus has emerged. Advocates likewise refuse to coalesce around any single alternative. Some experts favor a new formula based on the Medicare economic index, which measures inflation in healthcare delivery costs. But the CBO estimates that per-beneficiary spending under such a formula would be 30% more by 2016 than under the current formula. Other proposals call for temporarily increasing rates, then reverting to annual GDP growth, plus a bit more to cover physician costs.
No matter how the crisis is resolved, experts say, doctors almost certainly will have to make do with less. “When healthcare reform is finally fully implemented, there are going to be less dollars to pay for more services. It’s inevitable,” Mathews says. “And whether it takes the form of SGR or some other form, I’m afraid physicians are going to have to get used to having less money in the pool of money that’s allocated to pay providers.”
It could be a whole new ballgame. TH
Bryn Nelson, PhD, is a freelance medical writer based in Seattle.
Baseball, kick the can, Russian roulette—pick your game. Chances are good that it has worked its way into a metaphor to illustrate the infuriating, perplexing, and altogether frustrating inability of Congress to step up to the plate and pass a long-term fix to the broken sustainable growth rate (SGR) formula used to determine Medicare reimbursement rates.
On June 24, legislators avoided catastrophe by temporarily rescinding a 21.3% rate cut that went into effect June 1. The after-the-fact patch meant that some Medicare claims had to be reprocessed to recoup the full value, creating an administrative mess. The accompanying 2.2% rate increase expires Nov. 30. The reimbursement cut could reach nearly 30% next year unless Congress intervenes again.
“Obviously, there’s a lot of frustration around the issue, especially on the membership side,” says Ron Greeno, MD, FACP, SFHM, a member of SHM’s Public Policy and Leadership committees, and chief medical officer for Brentwood, Tenn.-based Cogent Healthcare. For hospitalists in many small private practices, he says, a major percentage of income comes from Medicare. “It’s a tremendous headache,” he says of the uncertainty. “It’s very hard to plan for. You’re trying to budget and you don’t know what the policy is going to be literally from week to week.”
The Blame Game
Despite the widespread sentiment among doctors that a permanent reimbursement rate fix should have been included in the healthcare reform legislation, skittishness over the price tag led legislators to drop it from the package. Based on last fall’s estimates, the total cost of a reform bill that scrapped the SGR would have ballooned by roughly $250 billion over 10 years, which would have threatened the bill’s passage.
But Congress has since been unable to pass a permanent fix as standalone legislation amid mounting concern over the national debt, and the price of inaction continues to rise. On April 30, the Congressional Budget Office (CBO) estimated that the cost of jettisoning the SGR formula and freezing rates at current levels had grown to $276 billion over 10 years.
Any serious consideration of lasting alternatives has now been pushed back to the lame-duck session, after the midterm elections. The can has been kicked down the road so many times, Dr. Greeno and others say, that most Congressional members have boot marks all over them. “So now you have a bigger problem at a more crucial time, when money is tighter than ever in a poor economy,” Dr. Greeno says. “And I just think it’s been a failure of our politicians.”
Other healthcare industry leaders have been just as critical. “Delaying the problem is not a solution,” said AMA President Cecil B. Wilson, MD, in a prepared statement after Congress passed the latest six-month reprieve in June. “It doesn’t solve the Medicare mess Congress has created with a long series of short-term Medicare patches over the last decade—including four to avert the 2010 cut alone.”
AMA-sponsored print ads have reminded legislators that delaying a fix until 2013 will again increase its cost, to $396 billion over 10 years. And the association’s June press release asserted that “Congress is playing a dangerous game of Russian roulette with seniors’ healthcare.”
Perhaps a game of “chicken” would be more apt.
Republicans have dared Democrats to spend the billions for a more lasting solution—in the absence of any cuts elsewhere in the healthcare delivery system—and be labeled as fiscally irresponsible. In turn, Democrats have dared Republicans to let the rate cut take effect and be labeled heartless as Medicare beneficiaries lose access to their healthcare providers.
Both parties blinked, resorting to almost unanimous short-term fixes that have allowed legislators to save face while putting off politically risky votes until after the November elections.
Lynne M. Allen, MN, ARNP, who works as a part-time hospitalist in hematology-oncology at 188-bed Kadlec Regional Medical Center in Richland, Wash., says she and other colleagues were initially hopeful that the Obama administration would make Congress work together to find a lasting solution. “There’s a sense of frustration because instead of that happening from our legislators, they’re playing a lot of games with the funding,” says Allen, a member of Team Hospitalist. “They’re not willing to step up to the plate, as they say, and make a decision that will allow us to go forward smoothly.”
The result, Allen says, has been a “roller-coaster ride” of uncertainty over reimbursements. Because Washington’s Tri-Cities region has a relatively high percentage of patients with private insurance, her hospital is somewhat cushioned from a precipitous drop in Medicare fees. But if CMS is ever forced to cut back on its rates, she fully expects private insurers to follow the same downward track.
Practical Concerns
Barbara Hartley, MD, a part-time hospitalist at 22-bed Benson Hospital in Benson, Ariz., says the town’s healthcare facility is somewhat protected from potential Medicare rate cuts through its official status as a Critical Access Hospital. Instead of being reimbursed through diagnosis-related group (DRG) codes, the rural hospital is repaid by Medicare for its total cost per day per patient.
The arrangement is a stable one at the moment, but not enough to dispel Dr. Hartley’s uneasy question: If the economy worsens, will Medicare be able to retain its commitment to rural hospitals? If not, the pain might be felt acutely in communities like Benson, where Dr. Hartley estimates that as much as 75% of the hospital’s in-patient business is through either Medicare or a Medicare Advantage plan.
Kirk Mathews, CEO of St. Louis-based Inpatient Management Inc. and a member of SHM’s Public Policy and Practice Management committees, says Medicare rate cuts also could significantly reduce the leverage of hospitalists during contract negotiations.
“Even if we’re employed by the hospital, but our professional fees that the hospital can recoup for our services are dramatically affected, it will affect how those future contracts go,” Mathews says. “We might be insulated temporarily by the strength of our current contract. But if the formula—however that works out—dramatically impacts the hospitalist reimbursement on the professional fee side, the hospital will feel that, and then hospitalists will eventually feel that as well.” In other words, it could strengthen the bargaining hand of the hospital at the expense of the hospitalist. “Therein lies the long-term threat,” he points out.
Independent Solution?
Some of the authority over physician payments might eventually be depoliticized via language in the reform legislation that empowers a new entity, the Independent Payment Advisory Board, to create policy on such critical monetary issues as reimbursement rates. Congress could still override the board’s policy decisions, but only if the Congressional alternative saves just as much money.
In the meantime, the money for a fix still has to come from somewhere, and no consensus has emerged. Advocates likewise refuse to coalesce around any single alternative. Some experts favor a new formula based on the Medicare economic index, which measures inflation in healthcare delivery costs. But the CBO estimates that per-beneficiary spending under such a formula would be 30% more by 2016 than under the current formula. Other proposals call for temporarily increasing rates, then reverting to annual GDP growth, plus a bit more to cover physician costs.
No matter how the crisis is resolved, experts say, doctors almost certainly will have to make do with less. “When healthcare reform is finally fully implemented, there are going to be less dollars to pay for more services. It’s inevitable,” Mathews says. “And whether it takes the form of SGR or some other form, I’m afraid physicians are going to have to get used to having less money in the pool of money that’s allocated to pay providers.”
It could be a whole new ballgame. TH
Bryn Nelson, PhD, is a freelance medical writer based in Seattle.
Hospital-Acquired Condition (HAC) Guidelines Produce $20M in Medicare Savings
How much has Medicare saved by not paying hospitals when patients get infections?
Hugh Black, DO
Charlotte, N.C.
Dr. Hospitalist responds: Since 2007, the Centers for Medicare and Medicaid Services (CMS) has tried to reduce the number of high-cost, hospital-acquired conditions (HACs), including infections, by encouraging providers to adhere to evidence-based guidelines. Some examples of these hospital-acquired conditions include:
- Catheter-associated urinary tract infections;
- Foreign objects retained after surgery; and
- Stage III and IV pressure ulcers.
CMS requires that acute-care hospitals, “effective with discharges occurring on or after Oct. 1, 2007, submit information on Medicare claims specifying whether diagnoses were present on admission.” Effective Oct. 1, 2008, Medicare no longer pays for charges associated with these HACs. So, if a Medicare beneficiary developed a Stage III pressure ulcer during his stay at an acute-care hospital, CMS would not pay for the incremental cost of the care associated with the “HAC.”
The U.S. government, in the May 4, 2010, edition of the Federal Register, reviewed the impact of this program. The data are based on Medicare claims data from October 2008 to June 2009. During this period of time, there were approximately 7.17 million acute-care hospital Medicare discharges.
The total net savings during this nine-month period for all HACs was $16.4 million. Three HACs (Stage III and IV pressure ulcers, DVT/PE after orthopedic procedure, and falls and trauma) accounted for more than $15.1 million in savings. Pro-rated for a 12-month period, the total net savings for all HACs would exceed $20 million.
Falls and trauma accounted for 34% of all HACs reported (11,253), followed by vascular catheter-associated infection (16%) and catheter-associated UTIs (16%). Air embolism and mediastinitis after CABG were the least recorded HACs; both were less than .01% if the total.
The goal is that, over time, with improvement in care, there would be a decrease in the number of hospital discharges where these conditions would be present. Therefore, the net savings would be expected to decline.
Medicare has considered a number of other HACs for this program, and reviewed the numbers of these conditions over the same nine-month period (see “Medicare’s Potential New Hospital-Acquired Conditions,” above). Despite some large numbers, CMS has stated it’s not proposing to add or remove HAC categories at this time. If you are interested in reviewing the entire report, visit http://edocket.access.gpo.gov/2010/pdf/ 2010-9163.pdf. TH
How much has Medicare saved by not paying hospitals when patients get infections?
Hugh Black, DO
Charlotte, N.C.
Dr. Hospitalist responds: Since 2007, the Centers for Medicare and Medicaid Services (CMS) has tried to reduce the number of high-cost, hospital-acquired conditions (HACs), including infections, by encouraging providers to adhere to evidence-based guidelines. Some examples of these hospital-acquired conditions include:
- Catheter-associated urinary tract infections;
- Foreign objects retained after surgery; and
- Stage III and IV pressure ulcers.
CMS requires that acute-care hospitals, “effective with discharges occurring on or after Oct. 1, 2007, submit information on Medicare claims specifying whether diagnoses were present on admission.” Effective Oct. 1, 2008, Medicare no longer pays for charges associated with these HACs. So, if a Medicare beneficiary developed a Stage III pressure ulcer during his stay at an acute-care hospital, CMS would not pay for the incremental cost of the care associated with the “HAC.”
The U.S. government, in the May 4, 2010, edition of the Federal Register, reviewed the impact of this program. The data are based on Medicare claims data from October 2008 to June 2009. During this period of time, there were approximately 7.17 million acute-care hospital Medicare discharges.
The total net savings during this nine-month period for all HACs was $16.4 million. Three HACs (Stage III and IV pressure ulcers, DVT/PE after orthopedic procedure, and falls and trauma) accounted for more than $15.1 million in savings. Pro-rated for a 12-month period, the total net savings for all HACs would exceed $20 million.
Falls and trauma accounted for 34% of all HACs reported (11,253), followed by vascular catheter-associated infection (16%) and catheter-associated UTIs (16%). Air embolism and mediastinitis after CABG were the least recorded HACs; both were less than .01% if the total.
The goal is that, over time, with improvement in care, there would be a decrease in the number of hospital discharges where these conditions would be present. Therefore, the net savings would be expected to decline.
Medicare has considered a number of other HACs for this program, and reviewed the numbers of these conditions over the same nine-month period (see “Medicare’s Potential New Hospital-Acquired Conditions,” above). Despite some large numbers, CMS has stated it’s not proposing to add or remove HAC categories at this time. If you are interested in reviewing the entire report, visit http://edocket.access.gpo.gov/2010/pdf/ 2010-9163.pdf. TH
How much has Medicare saved by not paying hospitals when patients get infections?
Hugh Black, DO
Charlotte, N.C.
Dr. Hospitalist responds: Since 2007, the Centers for Medicare and Medicaid Services (CMS) has tried to reduce the number of high-cost, hospital-acquired conditions (HACs), including infections, by encouraging providers to adhere to evidence-based guidelines. Some examples of these hospital-acquired conditions include:
- Catheter-associated urinary tract infections;
- Foreign objects retained after surgery; and
- Stage III and IV pressure ulcers.
CMS requires that acute-care hospitals, “effective with discharges occurring on or after Oct. 1, 2007, submit information on Medicare claims specifying whether diagnoses were present on admission.” Effective Oct. 1, 2008, Medicare no longer pays for charges associated with these HACs. So, if a Medicare beneficiary developed a Stage III pressure ulcer during his stay at an acute-care hospital, CMS would not pay for the incremental cost of the care associated with the “HAC.”
The U.S. government, in the May 4, 2010, edition of the Federal Register, reviewed the impact of this program. The data are based on Medicare claims data from October 2008 to June 2009. During this period of time, there were approximately 7.17 million acute-care hospital Medicare discharges.
The total net savings during this nine-month period for all HACs was $16.4 million. Three HACs (Stage III and IV pressure ulcers, DVT/PE after orthopedic procedure, and falls and trauma) accounted for more than $15.1 million in savings. Pro-rated for a 12-month period, the total net savings for all HACs would exceed $20 million.
Falls and trauma accounted for 34% of all HACs reported (11,253), followed by vascular catheter-associated infection (16%) and catheter-associated UTIs (16%). Air embolism and mediastinitis after CABG were the least recorded HACs; both were less than .01% if the total.
The goal is that, over time, with improvement in care, there would be a decrease in the number of hospital discharges where these conditions would be present. Therefore, the net savings would be expected to decline.
Medicare has considered a number of other HACs for this program, and reviewed the numbers of these conditions over the same nine-month period (see “Medicare’s Potential New Hospital-Acquired Conditions,” above). Despite some large numbers, CMS has stated it’s not proposing to add or remove HAC categories at this time. If you are interested in reviewing the entire report, visit http://edocket.access.gpo.gov/2010/pdf/ 2010-9163.pdf. TH
The Coming Challenges—and Opportunities—of Value-Based Purchasing
The Coming Challenges—and Opportunities—of Value-Based Purchasing
The Patient Protection and Affordable Care Act was signed into law in March, furthering the federal government’s commitment to increasing the efficiency of the U.S. healthcare system by decreasing cost and improving quality. An expansion of the “value-based purchasing” model, this law mandates that ratings and reimbursements to physicians and hospitals be increasingly tied to measured quality of care.
In this system, a physician’s quality profile will be determined by a variety of factors, including reported quality data, severity-adjusted clinical outcome measures, patient-safety indicators, and hospital-acquired conditions (HACs). Since all of these are, to a large extent, documentation issues, physicians are now forced to pay close attention to how they identify and describe diagnoses and procedures.
Medicare will, in effect, attempt to determine: “Did the clinical team correctly identify and appropriately treat all relevant patient conditions—without causing any adverse conditions—and do so safely, efficiently, and with good outcomes?”
The patient chart must “tell the story” of the episode of care in the hospital. It must accurately describe all of the patient’s conditions and demonstrate the complexity of medical decision-making and establishment of risk. Upon discharge, this risk must “match up” with the diagnoses that are being coded and the Medicare Severity Diagnostic Related Group (MS-DRGs) being assigned. Often, the information is present in the chart but is inconsistent from provider to provider, or documented in a way that is misunderstood by hospital coders.
If successful in improving our documentation skills, the reward will be a higher rating and increased reimbursement.
Medicare also is ramping up its claims denial and recovery business to help “clean up” the system. This includes the national rollout of the Recovery Audit Contactor (RAC) initiative (see “Attention to Detail,” April 2010, p. 1), as well as the new Medicare administrative contractors (MACs). Both rely on accurate documentation. The RACs will penalize hospitals and physicians financially for documentation lacking in specificity and accuracy; the MACs will deny payment for claims erroneously submitted (technical errors) or lacking documentation of medical necessity.
What makes many physicians even more uncomfortable in this new environment is that Medicare is striving to better align the priorities and financial incentives of hospitals and physicians. Alliances between the two are strongly encouraged through such programs as the Acute Care Episode (ACE) Demonstration Project (the precursor to hospital-physician bundled payments), and the establishment of accountable care organizations (ACOs).
This emerging environment presents a unique set of challenges and opportunities to HM groups. Hospitalists are historically more aligned with hospital administrations, as compared with most other specialties. Hospitalists also are asked to participate in the care of an increasingly large percentage of patients across all specialties. This could well be an opportunity to be rewarded for embracing both of these trends.
Hospitalists are uniquely positioned to function as the documentation improvement clinical team leaders, working closely with other physicians across all specialties and the administration to fulfill all documentation requirements—and to be rewarded for doing so. Though hospitalists should have a general understanding of the language and rules of documentation, a system must be in place that helps them identify and capture all the pertinent aspects of the medical record without the need to become coders themselves. To this end, a clinical documentation improvement (CDI) program is critical.
But it might not be enough.
What is needed is a clinical integration program, an enhancement of traditional CDI. This approach requires participation from ED physicians, along with clinical integration specialists, to document accurately and completely from the start. The clinical integration specialist ensures that medical necessity for inpatient admission and patient risk is being addressed and established, conditions are appropriately identified as being present on admission (POA), and all diagnoses are properly recognized and documented thoroughly and accurately. Clinical integration through collaborative documentation then continues throughout the hospitalization, with diagnostic authority and oversight from the hospitalist, all the way through discharge.
Hospitalists should welcome and champion this type of program. As documentation becomes the key to survival, a complete medical record will stand up to any and all scrutiny by Medicare or others.
As for the negatives, there are none.
Andrew H. Dombro, MD, national medical director, and Paul Weygandt, MD, JD, vice president of physician services, J.A. Thomas & Associates, Atlanta
The Coming Challenges—and Opportunities—of Value-Based Purchasing
The Patient Protection and Affordable Care Act was signed into law in March, furthering the federal government’s commitment to increasing the efficiency of the U.S. healthcare system by decreasing cost and improving quality. An expansion of the “value-based purchasing” model, this law mandates that ratings and reimbursements to physicians and hospitals be increasingly tied to measured quality of care.
In this system, a physician’s quality profile will be determined by a variety of factors, including reported quality data, severity-adjusted clinical outcome measures, patient-safety indicators, and hospital-acquired conditions (HACs). Since all of these are, to a large extent, documentation issues, physicians are now forced to pay close attention to how they identify and describe diagnoses and procedures.
Medicare will, in effect, attempt to determine: “Did the clinical team correctly identify and appropriately treat all relevant patient conditions—without causing any adverse conditions—and do so safely, efficiently, and with good outcomes?”
The patient chart must “tell the story” of the episode of care in the hospital. It must accurately describe all of the patient’s conditions and demonstrate the complexity of medical decision-making and establishment of risk. Upon discharge, this risk must “match up” with the diagnoses that are being coded and the Medicare Severity Diagnostic Related Group (MS-DRGs) being assigned. Often, the information is present in the chart but is inconsistent from provider to provider, or documented in a way that is misunderstood by hospital coders.
If successful in improving our documentation skills, the reward will be a higher rating and increased reimbursement.
Medicare also is ramping up its claims denial and recovery business to help “clean up” the system. This includes the national rollout of the Recovery Audit Contactor (RAC) initiative (see “Attention to Detail,” April 2010, p. 1), as well as the new Medicare administrative contractors (MACs). Both rely on accurate documentation. The RACs will penalize hospitals and physicians financially for documentation lacking in specificity and accuracy; the MACs will deny payment for claims erroneously submitted (technical errors) or lacking documentation of medical necessity.
What makes many physicians even more uncomfortable in this new environment is that Medicare is striving to better align the priorities and financial incentives of hospitals and physicians. Alliances between the two are strongly encouraged through such programs as the Acute Care Episode (ACE) Demonstration Project (the precursor to hospital-physician bundled payments), and the establishment of accountable care organizations (ACOs).
This emerging environment presents a unique set of challenges and opportunities to HM groups. Hospitalists are historically more aligned with hospital administrations, as compared with most other specialties. Hospitalists also are asked to participate in the care of an increasingly large percentage of patients across all specialties. This could well be an opportunity to be rewarded for embracing both of these trends.
Hospitalists are uniquely positioned to function as the documentation improvement clinical team leaders, working closely with other physicians across all specialties and the administration to fulfill all documentation requirements—and to be rewarded for doing so. Though hospitalists should have a general understanding of the language and rules of documentation, a system must be in place that helps them identify and capture all the pertinent aspects of the medical record without the need to become coders themselves. To this end, a clinical documentation improvement (CDI) program is critical.
But it might not be enough.
What is needed is a clinical integration program, an enhancement of traditional CDI. This approach requires participation from ED physicians, along with clinical integration specialists, to document accurately and completely from the start. The clinical integration specialist ensures that medical necessity for inpatient admission and patient risk is being addressed and established, conditions are appropriately identified as being present on admission (POA), and all diagnoses are properly recognized and documented thoroughly and accurately. Clinical integration through collaborative documentation then continues throughout the hospitalization, with diagnostic authority and oversight from the hospitalist, all the way through discharge.
Hospitalists should welcome and champion this type of program. As documentation becomes the key to survival, a complete medical record will stand up to any and all scrutiny by Medicare or others.
As for the negatives, there are none.
Andrew H. Dombro, MD, national medical director, and Paul Weygandt, MD, JD, vice president of physician services, J.A. Thomas & Associates, Atlanta
The Coming Challenges—and Opportunities—of Value-Based Purchasing
The Patient Protection and Affordable Care Act was signed into law in March, furthering the federal government’s commitment to increasing the efficiency of the U.S. healthcare system by decreasing cost and improving quality. An expansion of the “value-based purchasing” model, this law mandates that ratings and reimbursements to physicians and hospitals be increasingly tied to measured quality of care.
In this system, a physician’s quality profile will be determined by a variety of factors, including reported quality data, severity-adjusted clinical outcome measures, patient-safety indicators, and hospital-acquired conditions (HACs). Since all of these are, to a large extent, documentation issues, physicians are now forced to pay close attention to how they identify and describe diagnoses and procedures.
Medicare will, in effect, attempt to determine: “Did the clinical team correctly identify and appropriately treat all relevant patient conditions—without causing any adverse conditions—and do so safely, efficiently, and with good outcomes?”
The patient chart must “tell the story” of the episode of care in the hospital. It must accurately describe all of the patient’s conditions and demonstrate the complexity of medical decision-making and establishment of risk. Upon discharge, this risk must “match up” with the diagnoses that are being coded and the Medicare Severity Diagnostic Related Group (MS-DRGs) being assigned. Often, the information is present in the chart but is inconsistent from provider to provider, or documented in a way that is misunderstood by hospital coders.
If successful in improving our documentation skills, the reward will be a higher rating and increased reimbursement.
Medicare also is ramping up its claims denial and recovery business to help “clean up” the system. This includes the national rollout of the Recovery Audit Contactor (RAC) initiative (see “Attention to Detail,” April 2010, p. 1), as well as the new Medicare administrative contractors (MACs). Both rely on accurate documentation. The RACs will penalize hospitals and physicians financially for documentation lacking in specificity and accuracy; the MACs will deny payment for claims erroneously submitted (technical errors) or lacking documentation of medical necessity.
What makes many physicians even more uncomfortable in this new environment is that Medicare is striving to better align the priorities and financial incentives of hospitals and physicians. Alliances between the two are strongly encouraged through such programs as the Acute Care Episode (ACE) Demonstration Project (the precursor to hospital-physician bundled payments), and the establishment of accountable care organizations (ACOs).
This emerging environment presents a unique set of challenges and opportunities to HM groups. Hospitalists are historically more aligned with hospital administrations, as compared with most other specialties. Hospitalists also are asked to participate in the care of an increasingly large percentage of patients across all specialties. This could well be an opportunity to be rewarded for embracing both of these trends.
Hospitalists are uniquely positioned to function as the documentation improvement clinical team leaders, working closely with other physicians across all specialties and the administration to fulfill all documentation requirements—and to be rewarded for doing so. Though hospitalists should have a general understanding of the language and rules of documentation, a system must be in place that helps them identify and capture all the pertinent aspects of the medical record without the need to become coders themselves. To this end, a clinical documentation improvement (CDI) program is critical.
But it might not be enough.
What is needed is a clinical integration program, an enhancement of traditional CDI. This approach requires participation from ED physicians, along with clinical integration specialists, to document accurately and completely from the start. The clinical integration specialist ensures that medical necessity for inpatient admission and patient risk is being addressed and established, conditions are appropriately identified as being present on admission (POA), and all diagnoses are properly recognized and documented thoroughly and accurately. Clinical integration through collaborative documentation then continues throughout the hospitalization, with diagnostic authority and oversight from the hospitalist, all the way through discharge.
Hospitalists should welcome and champion this type of program. As documentation becomes the key to survival, a complete medical record will stand up to any and all scrutiny by Medicare or others.
As for the negatives, there are none.
Andrew H. Dombro, MD, national medical director, and Paul Weygandt, MD, JD, vice president of physician services, J.A. Thomas & Associates, Atlanta
Market Watch
New Drugs, Indications, Dosage Forms, and Approvals
- Hydromorphone extended-release tablets (Exalgo) have been approved by the FDA as a once-daily treatment for managing moderate to severe pain in opioid-tolerant patients needing continuous opioid analgesia for an extended period of time.1 This formulation uses the OROS osmotic delivery system to control the release rate. It is a CII controlled substance and is accompanied by a comprehensive Risk Evaluation and Mitigation Strategy (REMS) to ensure that the medication’s benefits outweigh its risks.
- IMGN910 has received orphan drug status for treating Merkel cell carcinoma, a skin cancer that usually occurs on the head or neck.2 It is in early-stage clinical trials.
- Immune globulin subcutaneous (human) 20% liquid (Hizentra) has been approved by the FDA as a once-weekly immunoglobulin replacement therapy for patients with primary immunodeficiency.3 It’s the first 20% subcutaneous immunoglobulin to receive FDA approval. This high-concentration product is stabilized with L-proline, a naturally occurring amino acid, which allows it to be stored at room temperature (up to 25°C [77°F]). Some adverse reactions include injection site bruising, pain, cysts, eczema, irritation, headache, cough, diarrhea, and fatigue.4
- Velaglucerase alfa for injection (VPRIV) has been approved by the FDA to treat adults and children with the rare genetic disorder Gaucher disease.5 Patients with Gaucher disease have a deficiency of the glucocerebrosidase enzyme. This enzyme prevents lipids from building up in the liver, spleen, bone marrow, and nervous system, which prevents them from working properly. VPRIV, a long-term replacement therapy, is approved for Type 1 Gaucher disease, the most common form, and is an alternative to imiglucerase (Cerezyme), which is in short supply. The most common reactions seen in clinical trials were allergic reactions, headache, dizziness, abdominal and back pain, nausea, fatigue/weakness, fever, and prolonged activated partial thromboplastin time.
Pipeline
- Betrixaban is a once-daily oral anticoagulant in Phase 2 clinical studies.6 Compared with warfarin in the EXPLORE-Xa study, betrixaban decreased the bleeding incidence in patients with nonvalvular atrial fibrillation or atrial flutter who had at least one stroke risk factor. The major and clinically relevant nonmajor bleeding episodes occurred less frequently in betrixaban-treated patients.
- Dabigatran etexilate is an oral anticoagulant in Phase 3 clinical trials.7 At the recent American College of Cardiology meeting in Ingelheim, Germany, dabigatran demonstrated consistent stroke prevention in patients with atrial fibrillation. It also reduced the number of strokes in patients with atrial fibrillation, compared with warfarin therapy. Additionally, dabigatran etexilate 110 mg and 150 mg twice daily was associated with a lower rate of major bleeding compared with warfarin in atrial fibrillation patients at low risk of stroke.
- Fentanyl sublingual spray (SL Spray) is in Phase 3 clinical trials to treat breakthrough pain in cancer patients. Sublingual administration of this product showed rapid, effective pain relief within five minutes.8
- Ketamine intranasal (Ereska) is a nonopioid NMDA receptor antagonist analgesic, which is undergoing Phase 3 clinical trials for managing moderate to severe acute pain.9 Studies have shown rapid, statistically significant relief of moderate to severe acute postoperative pain following dental surgery, following a variety of major orthopedic surgical procedures, and in cancer breakthrough pain.
- Lu AA21004 and Lu AA24530 are undergoing Phase 3 clinical trials for treating major depressive disorder (MDD).10 Lu AA21004 is a 5-HT3, 5-HT7 and 5-HT1B receptor antagonist, 5HT1A receptor agonist, and 5-HT transporter inhibitor. To date, it has shown a low propensity for drug-drug interactions and is extensively metabolized in the liver. Lu AA24530 has shown activity as a multimodal enhancer with reuptake inhibition at monoamine transporters, and having 5-HT3 and 5-HT2c receptor antagonist activity.
- Lurasidone is an atypical antipsychotic with high affinity and antagonist effects at the dopamine D2, serotonin 5-HT2, and serotonin 5-HT7 receptors.11 It is a partial agonist at serotonin 5HT1A receptor. The NDA was filed for this agent Dec. 30, 2009.
- Mipomersen, an apo-B synthesis inhibitor, is in Phase 3 clinical trials for treating patients with homozygous familial hypercholesterolemia (HoFM).12 This agent is proposed to reduce LDL-C by preventing the development of atherogenic lipids. In a study published in Lancet, mipomersen reduced LDL-C levels by an average of more than 100 mg/dL in HoFM patients.13
- Oxycodone/niacin (Acurox), an abuse deterrent formulation for this popular opioid, has been rejected by the FDA.14 According to the FDA and its review committee, the rejection was due to the “flushing” from the niacin, which was deemed ineffective as an abuse deterrent. In addition, the FDA said the “flushing” could be overcome by food intake or administration with over-the-counter pain relievers.
- Vilanterol/fluticasone is a combination of the inhaled corticosteroid fluticasone and the long-acting beta-agonist (LABA) vilanterol.15 It is in Phase 3 clinical trials for treating asthma. TH
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- FDA approves Exaglo (hydromorphone HCl) extended-release tablets. Drugs.com website. Available at: www.drugs.com/newdrugs/fda-approves-exaglo-hydromorphone-hcl-extended-release-2033.html?printable=1. Accessed April 27, 2010.
- ImmunoGen’s skin cancer drug gets orphan drug status. Reuters website. Available at: www.reuters.com/article/idUSSGE6270L720100308. Accessed April 27, 2010.
- CSL Behring receives FDA approval of Hizentra, first 20 percent subcutaneous immunoglobulin therapy. Drugs.com website. Available at: www.drugs.com/newdrugs/csl-behring-receives-fda-approval-hizentra-first-20-percent-subcutaneous-immunoglobulin-therapy-2037.html. Accessed April 27, 2010.
- Petrochko C. FDA okays 20% skin-injection immunodeficiency treatment. MedPage Today website. Available at: www.medpagetoday.com/tbprint.cfm?tbid=18858. Accessed April 27, 2010.
- Gansz Bobo E. FDA approves therapy to treat Gaucher disease. U.S. Food and Drug Administration website. Available at: www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm202288.htm. Accessed April 27, 2010.
- Portola Pharmaceuticals and Merck announce that Phase 2 study showed investigational factor Xa inhibitor, betrixaban, reduced incidence of bleeding compared to warfarin in patients with atrial fibrillation. Merck website. Available at: www.merck.com/newsroom/news-release-archive/research-and-development/2010_0315.html. Accessed April 27, 2010.
- Dabigatran etexilate shows greater reductions than warfarin in stroke in patients with atrial fibrillation across all stroke risk groups. Beohringer Ingelheim website. Available at: www.boehringer-ingelheim.com/news/news_releases/press_releases/2010/15_march_2010.html. Accessed April 27, 2010.
- INSYS Therapeutics, Inc. Announces Positive Phase III Efficacy Trial Results for Fentanyl Sublingual Spray. INSYS Therapeutics website. Available at: www.insysrx.com/news.htm. Accessed April 27, 2010.
- Third party reexamination of Javelin Pharmaceuticals’ Phase III trial data for Ereska (intranasal ketamine) yields statistically significant primary endpoint. Javelin website. Available at: ir.javelinpharmaceuticals.com/releasedetail.cfm?ReleaseID=444353. Accessed April 27, 2010.
- Lundbeck and Takeda finalise plans to initiate phase III pivotal clinical trials with Lu AA21004 and Lu AA24530. Takeda Pharmaceutical Company Limited website. Available at: www.takeda.com/press/article_35859.html. Accessed April 27, 2010.
- Dainippon Sumitomo Pharma America announces FDA acceptance of lurasidone new drug application for treatment of schizophrenia. PR Newswire website. Available at: www.prnewswire.com/news-releases/dainippon-sumitomo-pharma-america-announces-fda-acceptance-of-lurasidone-new-drug-application-for-treatment-of-schizophrenia-87265597.html. Accessed April 27, 2010.
- Mipomersen Phase 3 study in HoFH featured in The Lancet. Business Wire website. Available at: www.businesswire.com/portal/site/home/email/alert/?ndmViewId=news_view&newsLang=en&newsId=20100315005928. Accessed April 27, 2010.
- Raal FJ, Santos RD, Blom DJ, et al. Mipomersen, an apolipoprotein B synthesis inhibitor, for lowering of LDL cholesterol concentrations in patients with homozygous familial hypercholesterolaemia: a randomised, double-blind, placebo-controlled trial. Lancet. 2010;375(9719):998-1006.
- US FDA panel rejects King, Acura painkiller. Reuters website. Available at: www.reuters.com/assets/print?aid=USN2223552220100422. Accessed April 27, 2010.
- Dennis M. GlaxoSmithKline begins late-stage clinical programme for asthma drug Relovair. FirstWord website. Available at: www.firstwordplus.com/Fws.do?articleid=E256469FBD8F4A2F80C5DD3E844CC1E1&logRowId=356423. Accessed April 27, 2010.
New Drugs, Indications, Dosage Forms, and Approvals
- Hydromorphone extended-release tablets (Exalgo) have been approved by the FDA as a once-daily treatment for managing moderate to severe pain in opioid-tolerant patients needing continuous opioid analgesia for an extended period of time.1 This formulation uses the OROS osmotic delivery system to control the release rate. It is a CII controlled substance and is accompanied by a comprehensive Risk Evaluation and Mitigation Strategy (REMS) to ensure that the medication’s benefits outweigh its risks.
- IMGN910 has received orphan drug status for treating Merkel cell carcinoma, a skin cancer that usually occurs on the head or neck.2 It is in early-stage clinical trials.
- Immune globulin subcutaneous (human) 20% liquid (Hizentra) has been approved by the FDA as a once-weekly immunoglobulin replacement therapy for patients with primary immunodeficiency.3 It’s the first 20% subcutaneous immunoglobulin to receive FDA approval. This high-concentration product is stabilized with L-proline, a naturally occurring amino acid, which allows it to be stored at room temperature (up to 25°C [77°F]). Some adverse reactions include injection site bruising, pain, cysts, eczema, irritation, headache, cough, diarrhea, and fatigue.4
- Velaglucerase alfa for injection (VPRIV) has been approved by the FDA to treat adults and children with the rare genetic disorder Gaucher disease.5 Patients with Gaucher disease have a deficiency of the glucocerebrosidase enzyme. This enzyme prevents lipids from building up in the liver, spleen, bone marrow, and nervous system, which prevents them from working properly. VPRIV, a long-term replacement therapy, is approved for Type 1 Gaucher disease, the most common form, and is an alternative to imiglucerase (Cerezyme), which is in short supply. The most common reactions seen in clinical trials were allergic reactions, headache, dizziness, abdominal and back pain, nausea, fatigue/weakness, fever, and prolonged activated partial thromboplastin time.
Pipeline
- Betrixaban is a once-daily oral anticoagulant in Phase 2 clinical studies.6 Compared with warfarin in the EXPLORE-Xa study, betrixaban decreased the bleeding incidence in patients with nonvalvular atrial fibrillation or atrial flutter who had at least one stroke risk factor. The major and clinically relevant nonmajor bleeding episodes occurred less frequently in betrixaban-treated patients.
- Dabigatran etexilate is an oral anticoagulant in Phase 3 clinical trials.7 At the recent American College of Cardiology meeting in Ingelheim, Germany, dabigatran demonstrated consistent stroke prevention in patients with atrial fibrillation. It also reduced the number of strokes in patients with atrial fibrillation, compared with warfarin therapy. Additionally, dabigatran etexilate 110 mg and 150 mg twice daily was associated with a lower rate of major bleeding compared with warfarin in atrial fibrillation patients at low risk of stroke.
- Fentanyl sublingual spray (SL Spray) is in Phase 3 clinical trials to treat breakthrough pain in cancer patients. Sublingual administration of this product showed rapid, effective pain relief within five minutes.8
- Ketamine intranasal (Ereska) is a nonopioid NMDA receptor antagonist analgesic, which is undergoing Phase 3 clinical trials for managing moderate to severe acute pain.9 Studies have shown rapid, statistically significant relief of moderate to severe acute postoperative pain following dental surgery, following a variety of major orthopedic surgical procedures, and in cancer breakthrough pain.
- Lu AA21004 and Lu AA24530 are undergoing Phase 3 clinical trials for treating major depressive disorder (MDD).10 Lu AA21004 is a 5-HT3, 5-HT7 and 5-HT1B receptor antagonist, 5HT1A receptor agonist, and 5-HT transporter inhibitor. To date, it has shown a low propensity for drug-drug interactions and is extensively metabolized in the liver. Lu AA24530 has shown activity as a multimodal enhancer with reuptake inhibition at monoamine transporters, and having 5-HT3 and 5-HT2c receptor antagonist activity.
- Lurasidone is an atypical antipsychotic with high affinity and antagonist effects at the dopamine D2, serotonin 5-HT2, and serotonin 5-HT7 receptors.11 It is a partial agonist at serotonin 5HT1A receptor. The NDA was filed for this agent Dec. 30, 2009.
- Mipomersen, an apo-B synthesis inhibitor, is in Phase 3 clinical trials for treating patients with homozygous familial hypercholesterolemia (HoFM).12 This agent is proposed to reduce LDL-C by preventing the development of atherogenic lipids. In a study published in Lancet, mipomersen reduced LDL-C levels by an average of more than 100 mg/dL in HoFM patients.13
- Oxycodone/niacin (Acurox), an abuse deterrent formulation for this popular opioid, has been rejected by the FDA.14 According to the FDA and its review committee, the rejection was due to the “flushing” from the niacin, which was deemed ineffective as an abuse deterrent. In addition, the FDA said the “flushing” could be overcome by food intake or administration with over-the-counter pain relievers.
- Vilanterol/fluticasone is a combination of the inhaled corticosteroid fluticasone and the long-acting beta-agonist (LABA) vilanterol.15 It is in Phase 3 clinical trials for treating asthma. TH
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- FDA approves Exaglo (hydromorphone HCl) extended-release tablets. Drugs.com website. Available at: www.drugs.com/newdrugs/fda-approves-exaglo-hydromorphone-hcl-extended-release-2033.html?printable=1. Accessed April 27, 2010.
- ImmunoGen’s skin cancer drug gets orphan drug status. Reuters website. Available at: www.reuters.com/article/idUSSGE6270L720100308. Accessed April 27, 2010.
- CSL Behring receives FDA approval of Hizentra, first 20 percent subcutaneous immunoglobulin therapy. Drugs.com website. Available at: www.drugs.com/newdrugs/csl-behring-receives-fda-approval-hizentra-first-20-percent-subcutaneous-immunoglobulin-therapy-2037.html. Accessed April 27, 2010.
- Petrochko C. FDA okays 20% skin-injection immunodeficiency treatment. MedPage Today website. Available at: www.medpagetoday.com/tbprint.cfm?tbid=18858. Accessed April 27, 2010.
- Gansz Bobo E. FDA approves therapy to treat Gaucher disease. U.S. Food and Drug Administration website. Available at: www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm202288.htm. Accessed April 27, 2010.
- Portola Pharmaceuticals and Merck announce that Phase 2 study showed investigational factor Xa inhibitor, betrixaban, reduced incidence of bleeding compared to warfarin in patients with atrial fibrillation. Merck website. Available at: www.merck.com/newsroom/news-release-archive/research-and-development/2010_0315.html. Accessed April 27, 2010.
- Dabigatran etexilate shows greater reductions than warfarin in stroke in patients with atrial fibrillation across all stroke risk groups. Beohringer Ingelheim website. Available at: www.boehringer-ingelheim.com/news/news_releases/press_releases/2010/15_march_2010.html. Accessed April 27, 2010.
- INSYS Therapeutics, Inc. Announces Positive Phase III Efficacy Trial Results for Fentanyl Sublingual Spray. INSYS Therapeutics website. Available at: www.insysrx.com/news.htm. Accessed April 27, 2010.
- Third party reexamination of Javelin Pharmaceuticals’ Phase III trial data for Ereska (intranasal ketamine) yields statistically significant primary endpoint. Javelin website. Available at: ir.javelinpharmaceuticals.com/releasedetail.cfm?ReleaseID=444353. Accessed April 27, 2010.
- Lundbeck and Takeda finalise plans to initiate phase III pivotal clinical trials with Lu AA21004 and Lu AA24530. Takeda Pharmaceutical Company Limited website. Available at: www.takeda.com/press/article_35859.html. Accessed April 27, 2010.
- Dainippon Sumitomo Pharma America announces FDA acceptance of lurasidone new drug application for treatment of schizophrenia. PR Newswire website. Available at: www.prnewswire.com/news-releases/dainippon-sumitomo-pharma-america-announces-fda-acceptance-of-lurasidone-new-drug-application-for-treatment-of-schizophrenia-87265597.html. Accessed April 27, 2010.
- Mipomersen Phase 3 study in HoFH featured in The Lancet. Business Wire website. Available at: www.businesswire.com/portal/site/home/email/alert/?ndmViewId=news_view&newsLang=en&newsId=20100315005928. Accessed April 27, 2010.
- Raal FJ, Santos RD, Blom DJ, et al. Mipomersen, an apolipoprotein B synthesis inhibitor, for lowering of LDL cholesterol concentrations in patients with homozygous familial hypercholesterolaemia: a randomised, double-blind, placebo-controlled trial. Lancet. 2010;375(9719):998-1006.
- US FDA panel rejects King, Acura painkiller. Reuters website. Available at: www.reuters.com/assets/print?aid=USN2223552220100422. Accessed April 27, 2010.
- Dennis M. GlaxoSmithKline begins late-stage clinical programme for asthma drug Relovair. FirstWord website. Available at: www.firstwordplus.com/Fws.do?articleid=E256469FBD8F4A2F80C5DD3E844CC1E1&logRowId=356423. Accessed April 27, 2010.
New Drugs, Indications, Dosage Forms, and Approvals
- Hydromorphone extended-release tablets (Exalgo) have been approved by the FDA as a once-daily treatment for managing moderate to severe pain in opioid-tolerant patients needing continuous opioid analgesia for an extended period of time.1 This formulation uses the OROS osmotic delivery system to control the release rate. It is a CII controlled substance and is accompanied by a comprehensive Risk Evaluation and Mitigation Strategy (REMS) to ensure that the medication’s benefits outweigh its risks.
- IMGN910 has received orphan drug status for treating Merkel cell carcinoma, a skin cancer that usually occurs on the head or neck.2 It is in early-stage clinical trials.
- Immune globulin subcutaneous (human) 20% liquid (Hizentra) has been approved by the FDA as a once-weekly immunoglobulin replacement therapy for patients with primary immunodeficiency.3 It’s the first 20% subcutaneous immunoglobulin to receive FDA approval. This high-concentration product is stabilized with L-proline, a naturally occurring amino acid, which allows it to be stored at room temperature (up to 25°C [77°F]). Some adverse reactions include injection site bruising, pain, cysts, eczema, irritation, headache, cough, diarrhea, and fatigue.4
- Velaglucerase alfa for injection (VPRIV) has been approved by the FDA to treat adults and children with the rare genetic disorder Gaucher disease.5 Patients with Gaucher disease have a deficiency of the glucocerebrosidase enzyme. This enzyme prevents lipids from building up in the liver, spleen, bone marrow, and nervous system, which prevents them from working properly. VPRIV, a long-term replacement therapy, is approved for Type 1 Gaucher disease, the most common form, and is an alternative to imiglucerase (Cerezyme), which is in short supply. The most common reactions seen in clinical trials were allergic reactions, headache, dizziness, abdominal and back pain, nausea, fatigue/weakness, fever, and prolonged activated partial thromboplastin time.
Pipeline
- Betrixaban is a once-daily oral anticoagulant in Phase 2 clinical studies.6 Compared with warfarin in the EXPLORE-Xa study, betrixaban decreased the bleeding incidence in patients with nonvalvular atrial fibrillation or atrial flutter who had at least one stroke risk factor. The major and clinically relevant nonmajor bleeding episodes occurred less frequently in betrixaban-treated patients.
- Dabigatran etexilate is an oral anticoagulant in Phase 3 clinical trials.7 At the recent American College of Cardiology meeting in Ingelheim, Germany, dabigatran demonstrated consistent stroke prevention in patients with atrial fibrillation. It also reduced the number of strokes in patients with atrial fibrillation, compared with warfarin therapy. Additionally, dabigatran etexilate 110 mg and 150 mg twice daily was associated with a lower rate of major bleeding compared with warfarin in atrial fibrillation patients at low risk of stroke.
- Fentanyl sublingual spray (SL Spray) is in Phase 3 clinical trials to treat breakthrough pain in cancer patients. Sublingual administration of this product showed rapid, effective pain relief within five minutes.8
- Ketamine intranasal (Ereska) is a nonopioid NMDA receptor antagonist analgesic, which is undergoing Phase 3 clinical trials for managing moderate to severe acute pain.9 Studies have shown rapid, statistically significant relief of moderate to severe acute postoperative pain following dental surgery, following a variety of major orthopedic surgical procedures, and in cancer breakthrough pain.
- Lu AA21004 and Lu AA24530 are undergoing Phase 3 clinical trials for treating major depressive disorder (MDD).10 Lu AA21004 is a 5-HT3, 5-HT7 and 5-HT1B receptor antagonist, 5HT1A receptor agonist, and 5-HT transporter inhibitor. To date, it has shown a low propensity for drug-drug interactions and is extensively metabolized in the liver. Lu AA24530 has shown activity as a multimodal enhancer with reuptake inhibition at monoamine transporters, and having 5-HT3 and 5-HT2c receptor antagonist activity.
- Lurasidone is an atypical antipsychotic with high affinity and antagonist effects at the dopamine D2, serotonin 5-HT2, and serotonin 5-HT7 receptors.11 It is a partial agonist at serotonin 5HT1A receptor. The NDA was filed for this agent Dec. 30, 2009.
- Mipomersen, an apo-B synthesis inhibitor, is in Phase 3 clinical trials for treating patients with homozygous familial hypercholesterolemia (HoFM).12 This agent is proposed to reduce LDL-C by preventing the development of atherogenic lipids. In a study published in Lancet, mipomersen reduced LDL-C levels by an average of more than 100 mg/dL in HoFM patients.13
- Oxycodone/niacin (Acurox), an abuse deterrent formulation for this popular opioid, has been rejected by the FDA.14 According to the FDA and its review committee, the rejection was due to the “flushing” from the niacin, which was deemed ineffective as an abuse deterrent. In addition, the FDA said the “flushing” could be overcome by food intake or administration with over-the-counter pain relievers.
- Vilanterol/fluticasone is a combination of the inhaled corticosteroid fluticasone and the long-acting beta-agonist (LABA) vilanterol.15 It is in Phase 3 clinical trials for treating asthma. TH
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- FDA approves Exaglo (hydromorphone HCl) extended-release tablets. Drugs.com website. Available at: www.drugs.com/newdrugs/fda-approves-exaglo-hydromorphone-hcl-extended-release-2033.html?printable=1. Accessed April 27, 2010.
- ImmunoGen’s skin cancer drug gets orphan drug status. Reuters website. Available at: www.reuters.com/article/idUSSGE6270L720100308. Accessed April 27, 2010.
- CSL Behring receives FDA approval of Hizentra, first 20 percent subcutaneous immunoglobulin therapy. Drugs.com website. Available at: www.drugs.com/newdrugs/csl-behring-receives-fda-approval-hizentra-first-20-percent-subcutaneous-immunoglobulin-therapy-2037.html. Accessed April 27, 2010.
- Petrochko C. FDA okays 20% skin-injection immunodeficiency treatment. MedPage Today website. Available at: www.medpagetoday.com/tbprint.cfm?tbid=18858. Accessed April 27, 2010.
- Gansz Bobo E. FDA approves therapy to treat Gaucher disease. U.S. Food and Drug Administration website. Available at: www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm202288.htm. Accessed April 27, 2010.
- Portola Pharmaceuticals and Merck announce that Phase 2 study showed investigational factor Xa inhibitor, betrixaban, reduced incidence of bleeding compared to warfarin in patients with atrial fibrillation. Merck website. Available at: www.merck.com/newsroom/news-release-archive/research-and-development/2010_0315.html. Accessed April 27, 2010.
- Dabigatran etexilate shows greater reductions than warfarin in stroke in patients with atrial fibrillation across all stroke risk groups. Beohringer Ingelheim website. Available at: www.boehringer-ingelheim.com/news/news_releases/press_releases/2010/15_march_2010.html. Accessed April 27, 2010.
- INSYS Therapeutics, Inc. Announces Positive Phase III Efficacy Trial Results for Fentanyl Sublingual Spray. INSYS Therapeutics website. Available at: www.insysrx.com/news.htm. Accessed April 27, 2010.
- Third party reexamination of Javelin Pharmaceuticals’ Phase III trial data for Ereska (intranasal ketamine) yields statistically significant primary endpoint. Javelin website. Available at: ir.javelinpharmaceuticals.com/releasedetail.cfm?ReleaseID=444353. Accessed April 27, 2010.
- Lundbeck and Takeda finalise plans to initiate phase III pivotal clinical trials with Lu AA21004 and Lu AA24530. Takeda Pharmaceutical Company Limited website. Available at: www.takeda.com/press/article_35859.html. Accessed April 27, 2010.
- Dainippon Sumitomo Pharma America announces FDA acceptance of lurasidone new drug application for treatment of schizophrenia. PR Newswire website. Available at: www.prnewswire.com/news-releases/dainippon-sumitomo-pharma-america-announces-fda-acceptance-of-lurasidone-new-drug-application-for-treatment-of-schizophrenia-87265597.html. Accessed April 27, 2010.
- Mipomersen Phase 3 study in HoFH featured in The Lancet. Business Wire website. Available at: www.businesswire.com/portal/site/home/email/alert/?ndmViewId=news_view&newsLang=en&newsId=20100315005928. Accessed April 27, 2010.
- Raal FJ, Santos RD, Blom DJ, et al. Mipomersen, an apolipoprotein B synthesis inhibitor, for lowering of LDL cholesterol concentrations in patients with homozygous familial hypercholesterolaemia: a randomised, double-blind, placebo-controlled trial. Lancet. 2010;375(9719):998-1006.
- US FDA panel rejects King, Acura painkiller. Reuters website. Available at: www.reuters.com/assets/print?aid=USN2223552220100422. Accessed April 27, 2010.
- Dennis M. GlaxoSmithKline begins late-stage clinical programme for asthma drug Relovair. FirstWord website. Available at: www.firstwordplus.com/Fws.do?articleid=E256469FBD8F4A2F80C5DD3E844CC1E1&logRowId=356423. Accessed April 27, 2010.
Raise the ‘Red Flags’
To no one’s surprise, federal legislation doesn’t always do what its architects originally intended. A bill designed to protect consumers from identify theft can instead leave small hospitalist practices and other healthcare businesses in the lurch over whether they must meet stringent antitheft requirements intended for credit-card companies and banks. A bill designed to add millions of patients to the ranks of the insured could instead subtract millions of dollars from the reimbursements hospitals and doctors receive from private insurers.
—Jon Leibowitz, chairman, Federal Trade Commission
So What’s to Be Done?
An effort to correct one of these lingering headaches—known as the “Red Flags Rule”—is again on the table, though not everyone’s convinced it might finally be fixed seven years after it was first enacted. The rule, folded into the Fair and Accurate Credit Transactions Act of 2003, required the Federal Trade Commission (FTC) and other government agencies to come up with specific measures that “creditors” and “financial institutions” would have to design and implement to counter the growing risk of identity theft.
As intended, these measures would help businesses “identify, detect, and respond” to anything that might suggest identity theft. In other words, they could throw up red flags to warn of illegal activity.
But five years later, as the act’s Nov. 1, 2008, enforcement date was approaching, no one seemed to know exactly which businesses should be considered “creditors.” The act’s vague wording, in fact, created widespread fear that a measure designed principally for banks and credit-card companies would also apply to small accounting, legal, and healthcare practices, saddling them with cumbersome and expensive vetting protocols.
Thus began a series of requests by federal legislators that the FTC delay enforcement until the confusion could be sorted out. After three delays, including the latest pushback from June 1 through the end of this year, the commission’s patience is wearing thin. FTC Chairman Jon Leibowitz has been clear about the agency’s frustration over the extensions in lieu of a permanent resolution.
“Congress needs to fix the unintended consequences of the legislation establishing the Red Flags Rule—and to fix this problem quickly,” he said in a May 28 release. “As an agency, we’re charged with enforcing the law, and endless extensions delay enforcement.”
The not-so-subtle jab at Congressional inaction was aimed at one chamber in particular. Bill HR3763, which adds clarifying language to the rule and specifically excludes accounting, legal, and medical practices with 20 or fewer employees, sailed through the House of Representatives last October by a vote of 400-0. And then it promptly hit a giant sandbar in the form of the Senate. On May 25, Sen. John Thune (R-S.D.) and Sen. Mark Begich (D-Alaska) attempted a relaunch with their introduction of S3416, a near carbon copy of the House bill.
The measure is hardly a fait accompli, given the Senate’s recent track record, but a spokesman for Sen. Thune said the senator’s office is expecting a resolution before the FTC’s latest extension expires. Citing the commission’s decision to delay enforcement soon after the Senate bill’s introduction, he said, “We interpret that as an indication that they want to give Congress time to act, so we’re very optimistic that something will happen this year.”
Of course, the enforcement delay also might have something to do with the joint lawsuit filed May 21 by the American Medical Association, American Osteopathic Association, and the Medical Society of the District of Columbia. In their complaint, the three medical associations charged that the FTC’s application of the rule to physicians is “arbitrary, capricious, and contrary to the law.”
It’s now up to the Senate to decide whether that suit will become moot. TH
Bryn Nelson is a freelance medical writer based in Seattle.
To no one’s surprise, federal legislation doesn’t always do what its architects originally intended. A bill designed to protect consumers from identify theft can instead leave small hospitalist practices and other healthcare businesses in the lurch over whether they must meet stringent antitheft requirements intended for credit-card companies and banks. A bill designed to add millions of patients to the ranks of the insured could instead subtract millions of dollars from the reimbursements hospitals and doctors receive from private insurers.
—Jon Leibowitz, chairman, Federal Trade Commission
So What’s to Be Done?
An effort to correct one of these lingering headaches—known as the “Red Flags Rule”—is again on the table, though not everyone’s convinced it might finally be fixed seven years after it was first enacted. The rule, folded into the Fair and Accurate Credit Transactions Act of 2003, required the Federal Trade Commission (FTC) and other government agencies to come up with specific measures that “creditors” and “financial institutions” would have to design and implement to counter the growing risk of identity theft.
As intended, these measures would help businesses “identify, detect, and respond” to anything that might suggest identity theft. In other words, they could throw up red flags to warn of illegal activity.
But five years later, as the act’s Nov. 1, 2008, enforcement date was approaching, no one seemed to know exactly which businesses should be considered “creditors.” The act’s vague wording, in fact, created widespread fear that a measure designed principally for banks and credit-card companies would also apply to small accounting, legal, and healthcare practices, saddling them with cumbersome and expensive vetting protocols.
Thus began a series of requests by federal legislators that the FTC delay enforcement until the confusion could be sorted out. After three delays, including the latest pushback from June 1 through the end of this year, the commission’s patience is wearing thin. FTC Chairman Jon Leibowitz has been clear about the agency’s frustration over the extensions in lieu of a permanent resolution.
“Congress needs to fix the unintended consequences of the legislation establishing the Red Flags Rule—and to fix this problem quickly,” he said in a May 28 release. “As an agency, we’re charged with enforcing the law, and endless extensions delay enforcement.”
The not-so-subtle jab at Congressional inaction was aimed at one chamber in particular. Bill HR3763, which adds clarifying language to the rule and specifically excludes accounting, legal, and medical practices with 20 or fewer employees, sailed through the House of Representatives last October by a vote of 400-0. And then it promptly hit a giant sandbar in the form of the Senate. On May 25, Sen. John Thune (R-S.D.) and Sen. Mark Begich (D-Alaska) attempted a relaunch with their introduction of S3416, a near carbon copy of the House bill.
The measure is hardly a fait accompli, given the Senate’s recent track record, but a spokesman for Sen. Thune said the senator’s office is expecting a resolution before the FTC’s latest extension expires. Citing the commission’s decision to delay enforcement soon after the Senate bill’s introduction, he said, “We interpret that as an indication that they want to give Congress time to act, so we’re very optimistic that something will happen this year.”
Of course, the enforcement delay also might have something to do with the joint lawsuit filed May 21 by the American Medical Association, American Osteopathic Association, and the Medical Society of the District of Columbia. In their complaint, the three medical associations charged that the FTC’s application of the rule to physicians is “arbitrary, capricious, and contrary to the law.”
It’s now up to the Senate to decide whether that suit will become moot. TH
Bryn Nelson is a freelance medical writer based in Seattle.
To no one’s surprise, federal legislation doesn’t always do what its architects originally intended. A bill designed to protect consumers from identify theft can instead leave small hospitalist practices and other healthcare businesses in the lurch over whether they must meet stringent antitheft requirements intended for credit-card companies and banks. A bill designed to add millions of patients to the ranks of the insured could instead subtract millions of dollars from the reimbursements hospitals and doctors receive from private insurers.
—Jon Leibowitz, chairman, Federal Trade Commission
So What’s to Be Done?
An effort to correct one of these lingering headaches—known as the “Red Flags Rule”—is again on the table, though not everyone’s convinced it might finally be fixed seven years after it was first enacted. The rule, folded into the Fair and Accurate Credit Transactions Act of 2003, required the Federal Trade Commission (FTC) and other government agencies to come up with specific measures that “creditors” and “financial institutions” would have to design and implement to counter the growing risk of identity theft.
As intended, these measures would help businesses “identify, detect, and respond” to anything that might suggest identity theft. In other words, they could throw up red flags to warn of illegal activity.
But five years later, as the act’s Nov. 1, 2008, enforcement date was approaching, no one seemed to know exactly which businesses should be considered “creditors.” The act’s vague wording, in fact, created widespread fear that a measure designed principally for banks and credit-card companies would also apply to small accounting, legal, and healthcare practices, saddling them with cumbersome and expensive vetting protocols.
Thus began a series of requests by federal legislators that the FTC delay enforcement until the confusion could be sorted out. After three delays, including the latest pushback from June 1 through the end of this year, the commission’s patience is wearing thin. FTC Chairman Jon Leibowitz has been clear about the agency’s frustration over the extensions in lieu of a permanent resolution.
“Congress needs to fix the unintended consequences of the legislation establishing the Red Flags Rule—and to fix this problem quickly,” he said in a May 28 release. “As an agency, we’re charged with enforcing the law, and endless extensions delay enforcement.”
The not-so-subtle jab at Congressional inaction was aimed at one chamber in particular. Bill HR3763, which adds clarifying language to the rule and specifically excludes accounting, legal, and medical practices with 20 or fewer employees, sailed through the House of Representatives last October by a vote of 400-0. And then it promptly hit a giant sandbar in the form of the Senate. On May 25, Sen. John Thune (R-S.D.) and Sen. Mark Begich (D-Alaska) attempted a relaunch with their introduction of S3416, a near carbon copy of the House bill.
The measure is hardly a fait accompli, given the Senate’s recent track record, but a spokesman for Sen. Thune said the senator’s office is expecting a resolution before the FTC’s latest extension expires. Citing the commission’s decision to delay enforcement soon after the Senate bill’s introduction, he said, “We interpret that as an indication that they want to give Congress time to act, so we’re very optimistic that something will happen this year.”
Of course, the enforcement delay also might have something to do with the joint lawsuit filed May 21 by the American Medical Association, American Osteopathic Association, and the Medical Society of the District of Columbia. In their complaint, the three medical associations charged that the FTC’s application of the rule to physicians is “arbitrary, capricious, and contrary to the law.”
It’s now up to the Senate to decide whether that suit will become moot. TH
Bryn Nelson is a freelance medical writer based in Seattle.