User login
Avoid these common errors in self-defense!
This article is the second in a series of 4 derived from a symposium on malpractice risk management at the 91st Clinical Congress of the American College of Surgeons, San Francisco, Calif, in October 2005. Ms. Dobbs updated her comments in October 2006 and February 2007.
Part 1
March 2007 Informed refusal
James M. Goodman, JD
Part 2 - This issue
Common errors in self-defense
Claudia Dobbs, MA
Part 3
Patient safety as risk management tool
Thomas J. Donnelly, JD
Part 4
Responsibilities in obtaining informed consent
James M. Nelson, JD
In my work analyzing malpractice claims against physicians in 4 states, I (and my colleagues) have found that problems for defendant physicians can often be traced back to their failure to document the care and advice they provide. The 5 most common errors occur (and recur) when physicians are dealing with:
- results of tests
- informed consent
- informed refusal
- patient education
- postop follow-up discussion.
The good news is that, with some attention to detail, you may be able to avoid all these problems. Here is how.
Know the test results
When someone other than the ObGyn surgeon has ordered preoperative tests, that surgeon may say: “I didn’t order them, so I don’t need to review them or sign off on them. The primary care physician (PCP) will tell me whether the patient is ready to have the operation.”
This is a dangerous assumption. It’s your duty to make certain not only that the patient has medical clearance for the operation, but also that the chart proves it. We urge the ObGyn surgeons with whom we work to provide evidence in the chart—by initialing results—that they have reviewed preoperative tests.
If a cardiologist, pulmonologist, or other specialist has been asked to help clear a patient for surgery, the chart should include a consultation report that you review. Add your own notes or initial the report as evidence of your review. Also, document in the chart the details of any telephone communication you had with other physicians on the team.
It goes without saying that prenatal and antepartum records are vital to obstetric care. But those records can, regrettably, increase your liability if you don’t document information clearly or if you fail to monitor what you’ve entered in the record—a necessity made more of a challenge by an often cumbersome layout. Here are helpful hints for maintaining prenatal and antepartum records so that they are useful and reduce your exposure:
Use the record to alert physicians and staff to a high-risk patient. Use red ink or a highlighter or adhere bright stickers to the prenatal record—anything—to draw attention to vital data about risk factors, and thereby prevent injury.
Document sonographic findings and other diagnostic results on the prenatal form. Don’t bury this significant information in the chart—especially when findings are abnormal and must be monitored through the pregnancy. Then draw the reviewer’s eye to findings by, again, using red ink or a highlighter.
Document each prenatal visit completely—just as you would any office visit. Why limit your observations to the many check boxes on the prenatal form or to the one line provided for “Comments”—especially when the patient’s complaints are beyond what you would consider part of a “routine” prenatal examination? Instead, document any extra-routine notes on a separate sheet of paper. These notes should include her subjective complaints and your observations, assessment, and plan for care and follow-up.
Document any discussion you have about informed consent and informed refusal. Memorialize the informed consent communication in a progress note, but avoid documenting informed consent on the single line found on the prenatal form. Use a consent form to supplement your oral discussion.
Clearly document a patient’s informed refusal—whether it be of a significant diagnostic test (HIV, α-fetoprotein, amniocentesis, chorionic villus sampling), of hospital birth (in opting for home birth), and so forth. Ask her to sign a refusal document to supplement your oral discussion.
Make certain the complete prenatal record is sent to the hospital’s labor and delivery suite or operating room before the date of delivery. Some physicians periodically (eg, at the end of the second trimester or the beginning of the third trimester) submit the prenatal record in preparation for a patient’s delivery. This is an excellent practice: It provides pertinent information when a colleague is called to labor and delivery because you are off-call.
Ensure complete documentation of all vital communications and actions. Don’t take shortcuts simply because you’re using the prenatal form to document antepartum care. Document significant telephone calls with patients and consultants, referrals to specialists, missed appointments, and so on.
Don’t disregard a questionable finding
When a test reveals an incidental suspicious finding—such as a shadow on a chest radiograph or an abnormality in blood work—the surgeon is responsible for following up, even if someone else ordered the test. You cannot ignore such a finding or assume the internist or PCP will follow up.
In addition, you should notify the patient about any such problem—even if it is unrelated to the reason the patient is seeing you. If the finding is incidental to the surgery, you must tell the PCP and follow up with the patient.
We have seen situations in which diagnostic test results fall through the cracks. Cases that arise from such a lapse are, ultimately, indefensible and often involve shared responsibility or liability among the surgeon, the physician who ordered the test, and the primary care physician. Never put yourself in a position to have to say, later: “I saw that but didn’t do anything about it.”
Telephone calls to and from patients: A right way to keep records
Documenting the date, time, and content of your telephone calls with patients demonstrates competent management and provides evidence of your decision making in all aspects of patients’ care. Some guidelines:
- Always include the date, time, and content of the call.
- Document your advice to patients to come in for a follow-up appointment.
- Don’t let medical assistants offer independent medical advice. They should repeat your orders and nothing more. The notes should read, “Per Dr. Jones, advised patient to do X, Y, and Z,” and should be initialed by the staff member who spoke with the patient.
- Document follow-up calls—whether you’re on the telephone in the middle of the night or during office hours. Write in the chart what you advised the patient to do and what her response was. Don’t give any patient the ability to say, “If the doctor had told me that, I would have gone to the emergency room”—when that is precisely what you said, but you can’t prove it.
- Document missed appointments, especially postop, in the chart—not in the appointment book. Such chart notes show that the patient interrupted the treatment that you recommended. Later, if the patient claims, “My injury is a direct result of the physician’s failure to provide proper care,” you’ll be able to respond: “I can demonstrate that I asked you to return, but that you failed your appointment here, rescheduled it there, and made it almost impossible for us to provide good care.”
- Establish a mechanism for notifying patients to return after a postop no-show. Ensure that your staff returns the chart to you so that you can decide what to do next.
Be meticulous about informed consent
In documenting informed consent, your language reveals your attitude toward the process. For example, we’ve often heard physicians say “I consented the patient” instead of “the patient gave me her informed consent.”
I recommend that you document your discussion with the patient in the chart, instead of relying on a form. Typically, this is done on the history and physical, the consultation report, or the initial progress notes. Note which family members are present during the discussion: At some point, counsel may need to ask family members what they heard or what you said.
Sometimes we see informed consent discussions documented in the operative report. This is inappropriate because it represents an event after the fact. Informed consent that has been recorded after the procedure can look self-serving to a third-party observer—such as a plaintiff’s attorney. (For more advice on obtaining informed consent, see Part 4 of this series, upcoming in the June 2007 issue.)
Write it down when a patient won’t cooperate
Likewise, I recommend that you document informed refusal—whether or not the state in which you practice requires you to do so. This can dispel difficulties that may arise when a patient claims she would have consented to the surgery if you had discussed the risks properly with her. (See the March 2007 installment of this series for an in-depth look at informed refusal.)
Avoid a casual approach to patient education
Another deficiency that we encounter again and again is physicians’ failure to document their efforts to educate patients, orally and by written word. Consider that you and your peers spend a lot of time educating patients about surgical and nonsurgical options; discussing their comorbidities, smoking, and weight; and asking them to review videos, read pamphlets, and fill out lengthy questionnaires. Then, many fail to document their efforts!
Save those e-mail messages!
Physicians’ offices increasingly use e-mail to communicate with patients. Saving those e-mail messages is as important as documenting a telephone call in the chart.
Print both the patient’s e-mail and your response to it. Keep those pages in the chart to provide a running history of your management.
Notes enable defense attorneys to assert, with confidence, that, on a given date, patient and physician had a conversation or reviewed a handout, or that the nurse showed the patient a videotape. A record of this activity in writing makes it harder for the patient to claim, “The surgeon didn’t tell me any of these things.” The documentation can be as simple as a note stating: “Am CA Soc pamphlet reviewed with patient & husband; gave breast cancer booklet; nurse ran lumpectomy video.”
Keep postop notes specific
We’ve often found postoperative progress notes to be thin on detail. Typical notes are: “Wound looks good.” “Patient happy with results.” “Wound WNL” [within normal limits].
I recommend that you be specific in these notes. Describe the presence or absence of, for example, swelling, redness, adhesions, hematoma, drainage, fever, and regular urinary or bowel patterns.
When a patient claims negligent postop management, we often find sparse notes—sometimes a few words in the hospital chart or on the follow-up record. Nursing staff may identify fever, pus in the wound, and elevated laboratory values—yet the physician notes “Doing well.”
Without complete notes indicating the surgeon’s awareness of the patient’s condition, it’s impossible to convince a judge or jury that the surgeon was on top of the situation. Having inadequate notes makes the defense attorney’s job difficult.
Make the call, then write the note
Another gaping hole in documentation is poor notation of postoperative or posttreatment telephone calls during which a patient reports a significant change in her condition. Your policy may be “We never document telephone calls; we tell the patient to come in for a follow-up visit,” but what if the patient doesn’t show for the follow-up? There’s no evidence that an appointment was scheduled or that the patient failed to cancel or reschedule.
We’ve also seen situations in which the patient calls to report a problem and the medical assistant gives medical advice on the surgeon’s behalf because she (or he) has worked for the physician for, say, 15 years and “knows exactly what the surgeon would say.” We have seen that advice backfire because the assistant did not tell the surgeon what the patient said or because the staff member failed to ask how to respond to the patient’s concern.
And here’s another common scenario: The patient talks to the surgeon, who gives verbal advice but doesn’t document the discussion.
These are all dangerous areas in the use of the telephone. We advise physicians that telephone communication, including conversations after hours or when the physician is on call, must be documented. You simply cannot, ever, afford a gap in your documentation. (see “Telephone calls to and from patients: A right way to keep records,” and “Save those e-mail messages!”)
This article is the second in a series of 4 derived from a symposium on malpractice risk management at the 91st Clinical Congress of the American College of Surgeons, San Francisco, Calif, in October 2005. Ms. Dobbs updated her comments in October 2006 and February 2007.
Part 1
March 2007 Informed refusal
James M. Goodman, JD
Part 2 - This issue
Common errors in self-defense
Claudia Dobbs, MA
Part 3
Patient safety as risk management tool
Thomas J. Donnelly, JD
Part 4
Responsibilities in obtaining informed consent
James M. Nelson, JD
In my work analyzing malpractice claims against physicians in 4 states, I (and my colleagues) have found that problems for defendant physicians can often be traced back to their failure to document the care and advice they provide. The 5 most common errors occur (and recur) when physicians are dealing with:
- results of tests
- informed consent
- informed refusal
- patient education
- postop follow-up discussion.
The good news is that, with some attention to detail, you may be able to avoid all these problems. Here is how.
Know the test results
When someone other than the ObGyn surgeon has ordered preoperative tests, that surgeon may say: “I didn’t order them, so I don’t need to review them or sign off on them. The primary care physician (PCP) will tell me whether the patient is ready to have the operation.”
This is a dangerous assumption. It’s your duty to make certain not only that the patient has medical clearance for the operation, but also that the chart proves it. We urge the ObGyn surgeons with whom we work to provide evidence in the chart—by initialing results—that they have reviewed preoperative tests.
If a cardiologist, pulmonologist, or other specialist has been asked to help clear a patient for surgery, the chart should include a consultation report that you review. Add your own notes or initial the report as evidence of your review. Also, document in the chart the details of any telephone communication you had with other physicians on the team.
It goes without saying that prenatal and antepartum records are vital to obstetric care. But those records can, regrettably, increase your liability if you don’t document information clearly or if you fail to monitor what you’ve entered in the record—a necessity made more of a challenge by an often cumbersome layout. Here are helpful hints for maintaining prenatal and antepartum records so that they are useful and reduce your exposure:
Use the record to alert physicians and staff to a high-risk patient. Use red ink or a highlighter or adhere bright stickers to the prenatal record—anything—to draw attention to vital data about risk factors, and thereby prevent injury.
Document sonographic findings and other diagnostic results on the prenatal form. Don’t bury this significant information in the chart—especially when findings are abnormal and must be monitored through the pregnancy. Then draw the reviewer’s eye to findings by, again, using red ink or a highlighter.
Document each prenatal visit completely—just as you would any office visit. Why limit your observations to the many check boxes on the prenatal form or to the one line provided for “Comments”—especially when the patient’s complaints are beyond what you would consider part of a “routine” prenatal examination? Instead, document any extra-routine notes on a separate sheet of paper. These notes should include her subjective complaints and your observations, assessment, and plan for care and follow-up.
Document any discussion you have about informed consent and informed refusal. Memorialize the informed consent communication in a progress note, but avoid documenting informed consent on the single line found on the prenatal form. Use a consent form to supplement your oral discussion.
Clearly document a patient’s informed refusal—whether it be of a significant diagnostic test (HIV, α-fetoprotein, amniocentesis, chorionic villus sampling), of hospital birth (in opting for home birth), and so forth. Ask her to sign a refusal document to supplement your oral discussion.
Make certain the complete prenatal record is sent to the hospital’s labor and delivery suite or operating room before the date of delivery. Some physicians periodically (eg, at the end of the second trimester or the beginning of the third trimester) submit the prenatal record in preparation for a patient’s delivery. This is an excellent practice: It provides pertinent information when a colleague is called to labor and delivery because you are off-call.
Ensure complete documentation of all vital communications and actions. Don’t take shortcuts simply because you’re using the prenatal form to document antepartum care. Document significant telephone calls with patients and consultants, referrals to specialists, missed appointments, and so on.
Don’t disregard a questionable finding
When a test reveals an incidental suspicious finding—such as a shadow on a chest radiograph or an abnormality in blood work—the surgeon is responsible for following up, even if someone else ordered the test. You cannot ignore such a finding or assume the internist or PCP will follow up.
In addition, you should notify the patient about any such problem—even if it is unrelated to the reason the patient is seeing you. If the finding is incidental to the surgery, you must tell the PCP and follow up with the patient.
We have seen situations in which diagnostic test results fall through the cracks. Cases that arise from such a lapse are, ultimately, indefensible and often involve shared responsibility or liability among the surgeon, the physician who ordered the test, and the primary care physician. Never put yourself in a position to have to say, later: “I saw that but didn’t do anything about it.”
Telephone calls to and from patients: A right way to keep records
Documenting the date, time, and content of your telephone calls with patients demonstrates competent management and provides evidence of your decision making in all aspects of patients’ care. Some guidelines:
- Always include the date, time, and content of the call.
- Document your advice to patients to come in for a follow-up appointment.
- Don’t let medical assistants offer independent medical advice. They should repeat your orders and nothing more. The notes should read, “Per Dr. Jones, advised patient to do X, Y, and Z,” and should be initialed by the staff member who spoke with the patient.
- Document follow-up calls—whether you’re on the telephone in the middle of the night or during office hours. Write in the chart what you advised the patient to do and what her response was. Don’t give any patient the ability to say, “If the doctor had told me that, I would have gone to the emergency room”—when that is precisely what you said, but you can’t prove it.
- Document missed appointments, especially postop, in the chart—not in the appointment book. Such chart notes show that the patient interrupted the treatment that you recommended. Later, if the patient claims, “My injury is a direct result of the physician’s failure to provide proper care,” you’ll be able to respond: “I can demonstrate that I asked you to return, but that you failed your appointment here, rescheduled it there, and made it almost impossible for us to provide good care.”
- Establish a mechanism for notifying patients to return after a postop no-show. Ensure that your staff returns the chart to you so that you can decide what to do next.
Be meticulous about informed consent
In documenting informed consent, your language reveals your attitude toward the process. For example, we’ve often heard physicians say “I consented the patient” instead of “the patient gave me her informed consent.”
I recommend that you document your discussion with the patient in the chart, instead of relying on a form. Typically, this is done on the history and physical, the consultation report, or the initial progress notes. Note which family members are present during the discussion: At some point, counsel may need to ask family members what they heard or what you said.
Sometimes we see informed consent discussions documented in the operative report. This is inappropriate because it represents an event after the fact. Informed consent that has been recorded after the procedure can look self-serving to a third-party observer—such as a plaintiff’s attorney. (For more advice on obtaining informed consent, see Part 4 of this series, upcoming in the June 2007 issue.)
Write it down when a patient won’t cooperate
Likewise, I recommend that you document informed refusal—whether or not the state in which you practice requires you to do so. This can dispel difficulties that may arise when a patient claims she would have consented to the surgery if you had discussed the risks properly with her. (See the March 2007 installment of this series for an in-depth look at informed refusal.)
Avoid a casual approach to patient education
Another deficiency that we encounter again and again is physicians’ failure to document their efforts to educate patients, orally and by written word. Consider that you and your peers spend a lot of time educating patients about surgical and nonsurgical options; discussing their comorbidities, smoking, and weight; and asking them to review videos, read pamphlets, and fill out lengthy questionnaires. Then, many fail to document their efforts!
Save those e-mail messages!
Physicians’ offices increasingly use e-mail to communicate with patients. Saving those e-mail messages is as important as documenting a telephone call in the chart.
Print both the patient’s e-mail and your response to it. Keep those pages in the chart to provide a running history of your management.
Notes enable defense attorneys to assert, with confidence, that, on a given date, patient and physician had a conversation or reviewed a handout, or that the nurse showed the patient a videotape. A record of this activity in writing makes it harder for the patient to claim, “The surgeon didn’t tell me any of these things.” The documentation can be as simple as a note stating: “Am CA Soc pamphlet reviewed with patient & husband; gave breast cancer booklet; nurse ran lumpectomy video.”
Keep postop notes specific
We’ve often found postoperative progress notes to be thin on detail. Typical notes are: “Wound looks good.” “Patient happy with results.” “Wound WNL” [within normal limits].
I recommend that you be specific in these notes. Describe the presence or absence of, for example, swelling, redness, adhesions, hematoma, drainage, fever, and regular urinary or bowel patterns.
When a patient claims negligent postop management, we often find sparse notes—sometimes a few words in the hospital chart or on the follow-up record. Nursing staff may identify fever, pus in the wound, and elevated laboratory values—yet the physician notes “Doing well.”
Without complete notes indicating the surgeon’s awareness of the patient’s condition, it’s impossible to convince a judge or jury that the surgeon was on top of the situation. Having inadequate notes makes the defense attorney’s job difficult.
Make the call, then write the note
Another gaping hole in documentation is poor notation of postoperative or posttreatment telephone calls during which a patient reports a significant change in her condition. Your policy may be “We never document telephone calls; we tell the patient to come in for a follow-up visit,” but what if the patient doesn’t show for the follow-up? There’s no evidence that an appointment was scheduled or that the patient failed to cancel or reschedule.
We’ve also seen situations in which the patient calls to report a problem and the medical assistant gives medical advice on the surgeon’s behalf because she (or he) has worked for the physician for, say, 15 years and “knows exactly what the surgeon would say.” We have seen that advice backfire because the assistant did not tell the surgeon what the patient said or because the staff member failed to ask how to respond to the patient’s concern.
And here’s another common scenario: The patient talks to the surgeon, who gives verbal advice but doesn’t document the discussion.
These are all dangerous areas in the use of the telephone. We advise physicians that telephone communication, including conversations after hours or when the physician is on call, must be documented. You simply cannot, ever, afford a gap in your documentation. (see “Telephone calls to and from patients: A right way to keep records,” and “Save those e-mail messages!”)
This article is the second in a series of 4 derived from a symposium on malpractice risk management at the 91st Clinical Congress of the American College of Surgeons, San Francisco, Calif, in October 2005. Ms. Dobbs updated her comments in October 2006 and February 2007.
Part 1
March 2007 Informed refusal
James M. Goodman, JD
Part 2 - This issue
Common errors in self-defense
Claudia Dobbs, MA
Part 3
Patient safety as risk management tool
Thomas J. Donnelly, JD
Part 4
Responsibilities in obtaining informed consent
James M. Nelson, JD
In my work analyzing malpractice claims against physicians in 4 states, I (and my colleagues) have found that problems for defendant physicians can often be traced back to their failure to document the care and advice they provide. The 5 most common errors occur (and recur) when physicians are dealing with:
- results of tests
- informed consent
- informed refusal
- patient education
- postop follow-up discussion.
The good news is that, with some attention to detail, you may be able to avoid all these problems. Here is how.
Know the test results
When someone other than the ObGyn surgeon has ordered preoperative tests, that surgeon may say: “I didn’t order them, so I don’t need to review them or sign off on them. The primary care physician (PCP) will tell me whether the patient is ready to have the operation.”
This is a dangerous assumption. It’s your duty to make certain not only that the patient has medical clearance for the operation, but also that the chart proves it. We urge the ObGyn surgeons with whom we work to provide evidence in the chart—by initialing results—that they have reviewed preoperative tests.
If a cardiologist, pulmonologist, or other specialist has been asked to help clear a patient for surgery, the chart should include a consultation report that you review. Add your own notes or initial the report as evidence of your review. Also, document in the chart the details of any telephone communication you had with other physicians on the team.
It goes without saying that prenatal and antepartum records are vital to obstetric care. But those records can, regrettably, increase your liability if you don’t document information clearly or if you fail to monitor what you’ve entered in the record—a necessity made more of a challenge by an often cumbersome layout. Here are helpful hints for maintaining prenatal and antepartum records so that they are useful and reduce your exposure:
Use the record to alert physicians and staff to a high-risk patient. Use red ink or a highlighter or adhere bright stickers to the prenatal record—anything—to draw attention to vital data about risk factors, and thereby prevent injury.
Document sonographic findings and other diagnostic results on the prenatal form. Don’t bury this significant information in the chart—especially when findings are abnormal and must be monitored through the pregnancy. Then draw the reviewer’s eye to findings by, again, using red ink or a highlighter.
Document each prenatal visit completely—just as you would any office visit. Why limit your observations to the many check boxes on the prenatal form or to the one line provided for “Comments”—especially when the patient’s complaints are beyond what you would consider part of a “routine” prenatal examination? Instead, document any extra-routine notes on a separate sheet of paper. These notes should include her subjective complaints and your observations, assessment, and plan for care and follow-up.
Document any discussion you have about informed consent and informed refusal. Memorialize the informed consent communication in a progress note, but avoid documenting informed consent on the single line found on the prenatal form. Use a consent form to supplement your oral discussion.
Clearly document a patient’s informed refusal—whether it be of a significant diagnostic test (HIV, α-fetoprotein, amniocentesis, chorionic villus sampling), of hospital birth (in opting for home birth), and so forth. Ask her to sign a refusal document to supplement your oral discussion.
Make certain the complete prenatal record is sent to the hospital’s labor and delivery suite or operating room before the date of delivery. Some physicians periodically (eg, at the end of the second trimester or the beginning of the third trimester) submit the prenatal record in preparation for a patient’s delivery. This is an excellent practice: It provides pertinent information when a colleague is called to labor and delivery because you are off-call.
Ensure complete documentation of all vital communications and actions. Don’t take shortcuts simply because you’re using the prenatal form to document antepartum care. Document significant telephone calls with patients and consultants, referrals to specialists, missed appointments, and so on.
Don’t disregard a questionable finding
When a test reveals an incidental suspicious finding—such as a shadow on a chest radiograph or an abnormality in blood work—the surgeon is responsible for following up, even if someone else ordered the test. You cannot ignore such a finding or assume the internist or PCP will follow up.
In addition, you should notify the patient about any such problem—even if it is unrelated to the reason the patient is seeing you. If the finding is incidental to the surgery, you must tell the PCP and follow up with the patient.
We have seen situations in which diagnostic test results fall through the cracks. Cases that arise from such a lapse are, ultimately, indefensible and often involve shared responsibility or liability among the surgeon, the physician who ordered the test, and the primary care physician. Never put yourself in a position to have to say, later: “I saw that but didn’t do anything about it.”
Telephone calls to and from patients: A right way to keep records
Documenting the date, time, and content of your telephone calls with patients demonstrates competent management and provides evidence of your decision making in all aspects of patients’ care. Some guidelines:
- Always include the date, time, and content of the call.
- Document your advice to patients to come in for a follow-up appointment.
- Don’t let medical assistants offer independent medical advice. They should repeat your orders and nothing more. The notes should read, “Per Dr. Jones, advised patient to do X, Y, and Z,” and should be initialed by the staff member who spoke with the patient.
- Document follow-up calls—whether you’re on the telephone in the middle of the night or during office hours. Write in the chart what you advised the patient to do and what her response was. Don’t give any patient the ability to say, “If the doctor had told me that, I would have gone to the emergency room”—when that is precisely what you said, but you can’t prove it.
- Document missed appointments, especially postop, in the chart—not in the appointment book. Such chart notes show that the patient interrupted the treatment that you recommended. Later, if the patient claims, “My injury is a direct result of the physician’s failure to provide proper care,” you’ll be able to respond: “I can demonstrate that I asked you to return, but that you failed your appointment here, rescheduled it there, and made it almost impossible for us to provide good care.”
- Establish a mechanism for notifying patients to return after a postop no-show. Ensure that your staff returns the chart to you so that you can decide what to do next.
Be meticulous about informed consent
In documenting informed consent, your language reveals your attitude toward the process. For example, we’ve often heard physicians say “I consented the patient” instead of “the patient gave me her informed consent.”
I recommend that you document your discussion with the patient in the chart, instead of relying on a form. Typically, this is done on the history and physical, the consultation report, or the initial progress notes. Note which family members are present during the discussion: At some point, counsel may need to ask family members what they heard or what you said.
Sometimes we see informed consent discussions documented in the operative report. This is inappropriate because it represents an event after the fact. Informed consent that has been recorded after the procedure can look self-serving to a third-party observer—such as a plaintiff’s attorney. (For more advice on obtaining informed consent, see Part 4 of this series, upcoming in the June 2007 issue.)
Write it down when a patient won’t cooperate
Likewise, I recommend that you document informed refusal—whether or not the state in which you practice requires you to do so. This can dispel difficulties that may arise when a patient claims she would have consented to the surgery if you had discussed the risks properly with her. (See the March 2007 installment of this series for an in-depth look at informed refusal.)
Avoid a casual approach to patient education
Another deficiency that we encounter again and again is physicians’ failure to document their efforts to educate patients, orally and by written word. Consider that you and your peers spend a lot of time educating patients about surgical and nonsurgical options; discussing their comorbidities, smoking, and weight; and asking them to review videos, read pamphlets, and fill out lengthy questionnaires. Then, many fail to document their efforts!
Save those e-mail messages!
Physicians’ offices increasingly use e-mail to communicate with patients. Saving those e-mail messages is as important as documenting a telephone call in the chart.
Print both the patient’s e-mail and your response to it. Keep those pages in the chart to provide a running history of your management.
Notes enable defense attorneys to assert, with confidence, that, on a given date, patient and physician had a conversation or reviewed a handout, or that the nurse showed the patient a videotape. A record of this activity in writing makes it harder for the patient to claim, “The surgeon didn’t tell me any of these things.” The documentation can be as simple as a note stating: “Am CA Soc pamphlet reviewed with patient & husband; gave breast cancer booklet; nurse ran lumpectomy video.”
Keep postop notes specific
We’ve often found postoperative progress notes to be thin on detail. Typical notes are: “Wound looks good.” “Patient happy with results.” “Wound WNL” [within normal limits].
I recommend that you be specific in these notes. Describe the presence or absence of, for example, swelling, redness, adhesions, hematoma, drainage, fever, and regular urinary or bowel patterns.
When a patient claims negligent postop management, we often find sparse notes—sometimes a few words in the hospital chart or on the follow-up record. Nursing staff may identify fever, pus in the wound, and elevated laboratory values—yet the physician notes “Doing well.”
Without complete notes indicating the surgeon’s awareness of the patient’s condition, it’s impossible to convince a judge or jury that the surgeon was on top of the situation. Having inadequate notes makes the defense attorney’s job difficult.
Make the call, then write the note
Another gaping hole in documentation is poor notation of postoperative or posttreatment telephone calls during which a patient reports a significant change in her condition. Your policy may be “We never document telephone calls; we tell the patient to come in for a follow-up visit,” but what if the patient doesn’t show for the follow-up? There’s no evidence that an appointment was scheduled or that the patient failed to cancel or reschedule.
We’ve also seen situations in which the patient calls to report a problem and the medical assistant gives medical advice on the surgeon’s behalf because she (or he) has worked for the physician for, say, 15 years and “knows exactly what the surgeon would say.” We have seen that advice backfire because the assistant did not tell the surgeon what the patient said or because the staff member failed to ask how to respond to the patient’s concern.
And here’s another common scenario: The patient talks to the surgeon, who gives verbal advice but doesn’t document the discussion.
These are all dangerous areas in the use of the telephone. We advise physicians that telephone communication, including conversations after hours or when the physician is on call, must be documented. You simply cannot, ever, afford a gap in your documentation. (see “Telephone calls to and from patients: A right way to keep records,” and “Save those e-mail messages!”)
Dysphagia, Dizziness, and Dysarthria
Brief history: A 32-year-old female presents with dysphagia, dizziness, and dysarthria.
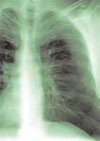
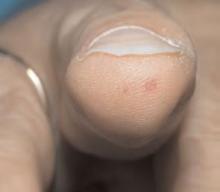
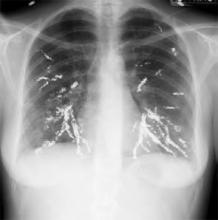
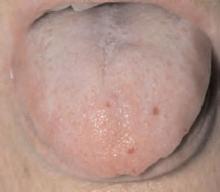
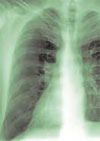
Salient findings: Chest X-ray demonstrates many embolization coils within both lungs. Photographs demonstrate superficial telangiectasias of the tongue and distal phalanx. These findings indicate the patient’s diagnosis: hereditary hemorrhagic telangiectasia (HHT), previously known as Osler-Weber-Rendu syndrome.
Patient population/natural history of disease: HHT is an autosomal dominant trait, so family members should be counseled on the implications of having a relative with the disease. HHT patients have abnormal vessels prone to bleeding and often develop arteriovenous malformations (AVMs). Diagnosis is made with 75% of the following symptoms:
- Epistaxis;
- Mucocutaneous telangiectasias;
- GI, pulmonary, or hepatic AVMs; and/or
- A first-degree relative with HHT.
Patients often present with dyspnea and hemoptysis. With pulmonary AVMs, the oxygenation and filtration functions of the lungs are bypassed, placing the patient at risk for hypoxia, polycythemia, paradoxical strokes, and brain abscesses.
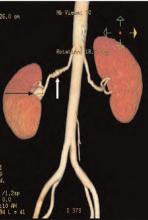
Management: AVMs can be diagnosed and treated with angiography and embolization. In this patient the coils had been placed elsewhere. The use of coils larger than 3 mm in AVMs should be treated because they are associated with significantly increased morbidity and mortality. Steel coils are covered with thrombogenic fibers that induce clotting and sealing of the AVM; blood is no longer shunted through the right-to-left shunt. Unfortunately, a long-term complication of pulmonary AVMs treated by embolization therapy is the development of new pulmonary AVMs.
The patient in this case had many metallic coils visible on chest X-ray and because she had required multiple pulmonary angiograms and embolizations over the years.
It’s important to administer an ECG to all HHT patients prior to treatment; those with a left bundle branch block must have pacing mechanisms in place or at hand because catheter placement and manipulation within the right heart can induce right heart blockage. Take care to avoid air emboli in all lines due to right-to-left shunting in these patients.
Take-Home Points:
- Untreated pulmonary AVMs are associated with paradoxical strokes, brain abscesses, and hypoxia;
- HHT is associated with pulmonary AVMs;
- Coil embolization of pulmonary AVMs has been shown to improve dyspnea and oxygen saturation while decreasing right-to-left shunt fraction in HHT patients.
- Complications of embolization therapy may include development of new pulmonary AVMs; and
- All patients should undergo ECG prior to pulmonary angiography to screen for left bundle branch block. TH
Helena Summers is a radiology resident and Erik Summers is a hospitalist at the Mayo Clinic College of Medicine, Rochester, Minn.
References
- Swanson KL, Prakash UB, Stanson AW. Pulmonary arteriovenous fistulas: Mayo Clinic experience, 1982-1997. Mayo Clin Proc. 1999 Jul;74(7):671-680.
- Cottin V, Plauchu H, Bayle JY, et al. Pulmonary arteriovenous malformations in patients with hereditary hemorrhagic telangiectasia. Am J Respir Crit Care Med. 2004 May 1;169(9):994-1000. Epub 2004 Jan 23.
Brief history: A 32-year-old female presents with dysphagia, dizziness, and dysarthria.
Salient findings: Chest X-ray demonstrates many embolization coils within both lungs. Photographs demonstrate superficial telangiectasias of the tongue and distal phalanx. These findings indicate the patient’s diagnosis: hereditary hemorrhagic telangiectasia (HHT), previously known as Osler-Weber-Rendu syndrome.
Patient population/natural history of disease: HHT is an autosomal dominant trait, so family members should be counseled on the implications of having a relative with the disease. HHT patients have abnormal vessels prone to bleeding and often develop arteriovenous malformations (AVMs). Diagnosis is made with 75% of the following symptoms:
- Epistaxis;
- Mucocutaneous telangiectasias;
- GI, pulmonary, or hepatic AVMs; and/or
- A first-degree relative with HHT.
Patients often present with dyspnea and hemoptysis. With pulmonary AVMs, the oxygenation and filtration functions of the lungs are bypassed, placing the patient at risk for hypoxia, polycythemia, paradoxical strokes, and brain abscesses.
Management: AVMs can be diagnosed and treated with angiography and embolization. In this patient the coils had been placed elsewhere. The use of coils larger than 3 mm in AVMs should be treated because they are associated with significantly increased morbidity and mortality. Steel coils are covered with thrombogenic fibers that induce clotting and sealing of the AVM; blood is no longer shunted through the right-to-left shunt. Unfortunately, a long-term complication of pulmonary AVMs treated by embolization therapy is the development of new pulmonary AVMs.
The patient in this case had many metallic coils visible on chest X-ray and because she had required multiple pulmonary angiograms and embolizations over the years.
It’s important to administer an ECG to all HHT patients prior to treatment; those with a left bundle branch block must have pacing mechanisms in place or at hand because catheter placement and manipulation within the right heart can induce right heart blockage. Take care to avoid air emboli in all lines due to right-to-left shunting in these patients.
Take-Home Points:
- Untreated pulmonary AVMs are associated with paradoxical strokes, brain abscesses, and hypoxia;
- HHT is associated with pulmonary AVMs;
- Coil embolization of pulmonary AVMs has been shown to improve dyspnea and oxygen saturation while decreasing right-to-left shunt fraction in HHT patients.
- Complications of embolization therapy may include development of new pulmonary AVMs; and
- All patients should undergo ECG prior to pulmonary angiography to screen for left bundle branch block. TH
Helena Summers is a radiology resident and Erik Summers is a hospitalist at the Mayo Clinic College of Medicine, Rochester, Minn.
References
- Swanson KL, Prakash UB, Stanson AW. Pulmonary arteriovenous fistulas: Mayo Clinic experience, 1982-1997. Mayo Clin Proc. 1999 Jul;74(7):671-680.
- Cottin V, Plauchu H, Bayle JY, et al. Pulmonary arteriovenous malformations in patients with hereditary hemorrhagic telangiectasia. Am J Respir Crit Care Med. 2004 May 1;169(9):994-1000. Epub 2004 Jan 23.
Brief history: A 32-year-old female presents with dysphagia, dizziness, and dysarthria.
Salient findings: Chest X-ray demonstrates many embolization coils within both lungs. Photographs demonstrate superficial telangiectasias of the tongue and distal phalanx. These findings indicate the patient’s diagnosis: hereditary hemorrhagic telangiectasia (HHT), previously known as Osler-Weber-Rendu syndrome.
Patient population/natural history of disease: HHT is an autosomal dominant trait, so family members should be counseled on the implications of having a relative with the disease. HHT patients have abnormal vessels prone to bleeding and often develop arteriovenous malformations (AVMs). Diagnosis is made with 75% of the following symptoms:
- Epistaxis;
- Mucocutaneous telangiectasias;
- GI, pulmonary, or hepatic AVMs; and/or
- A first-degree relative with HHT.
Patients often present with dyspnea and hemoptysis. With pulmonary AVMs, the oxygenation and filtration functions of the lungs are bypassed, placing the patient at risk for hypoxia, polycythemia, paradoxical strokes, and brain abscesses.
Management: AVMs can be diagnosed and treated with angiography and embolization. In this patient the coils had been placed elsewhere. The use of coils larger than 3 mm in AVMs should be treated because they are associated with significantly increased morbidity and mortality. Steel coils are covered with thrombogenic fibers that induce clotting and sealing of the AVM; blood is no longer shunted through the right-to-left shunt. Unfortunately, a long-term complication of pulmonary AVMs treated by embolization therapy is the development of new pulmonary AVMs.
The patient in this case had many metallic coils visible on chest X-ray and because she had required multiple pulmonary angiograms and embolizations over the years.
It’s important to administer an ECG to all HHT patients prior to treatment; those with a left bundle branch block must have pacing mechanisms in place or at hand because catheter placement and manipulation within the right heart can induce right heart blockage. Take care to avoid air emboli in all lines due to right-to-left shunting in these patients.
Take-Home Points:
- Untreated pulmonary AVMs are associated with paradoxical strokes, brain abscesses, and hypoxia;
- HHT is associated with pulmonary AVMs;
- Coil embolization of pulmonary AVMs has been shown to improve dyspnea and oxygen saturation while decreasing right-to-left shunt fraction in HHT patients.
- Complications of embolization therapy may include development of new pulmonary AVMs; and
- All patients should undergo ECG prior to pulmonary angiography to screen for left bundle branch block. TH
Helena Summers is a radiology resident and Erik Summers is a hospitalist at the Mayo Clinic College of Medicine, Rochester, Minn.
References
- Swanson KL, Prakash UB, Stanson AW. Pulmonary arteriovenous fistulas: Mayo Clinic experience, 1982-1997. Mayo Clin Proc. 1999 Jul;74(7):671-680.
- Cottin V, Plauchu H, Bayle JY, et al. Pulmonary arteriovenous malformations in patients with hereditary hemorrhagic telangiectasia. Am J Respir Crit Care Med. 2004 May 1;169(9):994-1000. Epub 2004 Jan 23.
Protect yourself! Make a plan to obtain “informed refusal”
This article is the first in a series of 4 derived from a symposium on malpractice risk management at the 91st Clinical Congress of the American College of Surgeons, San Francisco, Calif, in October 2005. Mr. Goodman updated his comments in October 2006.
Part 1
Informed refusal
James M. Goodman, JD
Part 2
Common errors in self-defense
Claudia Dobbs, MA
Part 3
Patient safety as risk management tool
Thomas J. Donnelly, JD
Part 4
Responsibilities in obtaining informed consent
James M. Nelson, JD
The concept of informed refusal is similar to that of informed consent. However, in working with physicians for 30 years, I have found that informed refusal is not nearly as well understood as informed consent.
Informed consent means a patient has the right to understand the risks of death, serious bodily injury, or other common outcomes of an operation or medical treatment. The patient also has the right to be told about the risks of refusing a particular operation, test, medication, or other medical intervention.
If a patient is reluctant or noncompliant, you may not be doing enough if you simply document that he or she refused your recommendation of treatment. You should also make a record of your efforts to explain to the patient the risks of refusing that treatment.
Informed refusal unfolds in 4 steps
Keep in mind these 4 components:
- The physician determines the patient needs a particular operation, test, medication, or other type of medical intervention
- The physician tells the patient about the needed intervention
- The patient refuses the recommended treatment for any reason: “I don’t think I need that test.” “I don’t like needles.” “I don’t care if I die.”
- The physician explains the risks of not having the treatment so the patient can make an informed decision when refusing it.
The courts first recognized the concept of informed refusal in a case in California more than 30 years ago. A woman seeing her long-time gynecologist was advised to have a Pap smear. Despite repeated recommendations, the patient declined the test. She later developed cervical cancer and sued her physician, claiming malpractice.
Because the physician and patient agreed the former had recommended the Pap smear, the trial judge threw the case out.
He determined it was the patient’s fault that she did not get the test that could have alerted her to the presence of cancer.
A higher court, however, reversed that decision because the patient had the right to make an informed decision in refusing the test—informed refusal.
The appeals judge ruled the gynecologist had a duty not only to recommend the test, but also to make sure the patient understood the consequences of her refusal. Because the evidence of informed refusal was lacking, the case was returned to the lower court.
Case: Patient refuses CT scan, dies; suit follows
The issue of informed refusal was used in a 1996 trial, about 20 years after the doctrine was first recognized in California. The emergency department (ED) physician in that case was found responsible for a patient’s death.
The patient, who had a long history of alcoholism, had fallen at home and struck his head. He was unconscious briefly before his wife took him to the ED. Although the examination did not reveal any neurologic abnormalities, the physician recommended a computed tomography (CT) scan of the head.
According to the physician, the patient and his wife refused the CT scan because they did not have health insurance. The physician failed to document his recommendation of the scan or the discussion in which the patient refused it. The patient and his wife left the ED. The next day, the patient died of multiple subdural bleeds. The widow sued for wrongful death.
Was the recommendation made?
During the trial, the physician testified that he had recommended a CT scan to the patient and his wife. The wife denied receiving such a recommendation. The trial attorneys focused considerable attention on whether the recommendation had actually been made. No one really focused on whether the physician advised the patient about the risks of refusal.
At the end of the trial, the plaintiff’s lawyer cleverly requested that the judge instruct the jury on the concept of informed refusal, knowing there had been no evidence that the doctor had advised the patient or his wife of the risk of refusing the CT scan.
The jury concluded that the physician did recommend the scan but failed to advise the patient or his wife of the risks of not having it. The trial ended in a plaintiff’s verdict of several hundred thousand dollars.
What’s happening nationally?
Four other states besides California have considered legislation regarding informed refusal: Nevada, Vermont, and Michigan have passed laws recognizing its existence, and Mississippi recognized the concept even though an informed refusal bill was defeated in the legislature.
Informed refusal is embodied in court decisions in other states. I have found no state where the doctrine has been overtly rejected.
If you want to learn more about patient safety and liability, the patient safety committee of the American College of Surgeons has published a booklet and manual, available on the organization’s Web site (www.facs.org/commerce/catsplash.html), containing essential information for surgeons and other physicians.1,2
The Joint Commission on Accreditation of Healthcare Organizations offers a 50-page book, available free online: Health Care at the Crossroads: Strategies for Improving the Medical Liability System and Preventing Patient Injury.3 It is available at www.jointcommission.org/NR/rdonlyres/167DD821-A395-48FD-87F9-6AB12BCACB0F/0/Medical_Liability.pdf.
1. Manuel BM, Nora PF, eds. Surgical Patient Safety: Essential Information for Surgeons in Today’s Environment. 05PS-0001. Chicago, Ill: American College of Surgeons; 2005.
2. Professional Liability Committee, American College of Surgeons. Nora PF, ed. Professional Liability/Risk Management: A Manual for Surgeons. 2nd ed. 04PL-0001. Chicago, Ill: American College of Surgeons; 1997.
3. Joint Commission on Accreditation of Healthcare Organizations. Health Care at the Crossroads: Strategies for Improving the Medical Liability System and Preventing Patient Injury. Oakbrook Terrace, Ill: Joint Commission on Accreditation of Healthcare Organizations; 2005.
Timing the conversation
It is not always clear when a physician should acknowledge that a patient has refused a recommended treatment. Patients are often frightened or reluctant about an operation or medical treatment. Some will want time to think about it, talk with friends or family, and, perhaps, get a second opinion.
Suppose you recommend a breast biopsy to a responsible and long-term patient. She may well want to talk to her husband or close friend about it before making a decision. You should not feel compelled to say, “Fine, but you could die of breast cancer if you don’t have this done.” The informed refusal discussion should occur when a patient makes it clear that she has rejected your recommendation.
A risk-management professional might say that the sooner you have this conversation with the patient and document it, the better. But you have to base your timing on the situation and your assessment of the patient’s reliability.
Harsh words are unnecessary
The informed refusal conversation need not take place immediately with a patient who seems reasonable and thoughtful about the process. However, obtaining informed refusal on the spot is worth the effort if you doubt the patient will return to give you a decision.
Maintaining the patient’s trust is important. Do not be so concerned about protecting yourself from a malpractice suit that you constantly make harsh comments about what could happen if patients reject medical advice. The key is for you to assess the patient’s likelihood to respond later.
This article is the first in a series of 4 derived from a symposium on malpractice risk management at the 91st Clinical Congress of the American College of Surgeons, San Francisco, Calif, in October 2005. Mr. Goodman updated his comments in October 2006.
Part 1
Informed refusal
James M. Goodman, JD
Part 2
Common errors in self-defense
Claudia Dobbs, MA
Part 3
Patient safety as risk management tool
Thomas J. Donnelly, JD
Part 4
Responsibilities in obtaining informed consent
James M. Nelson, JD
The concept of informed refusal is similar to that of informed consent. However, in working with physicians for 30 years, I have found that informed refusal is not nearly as well understood as informed consent.
Informed consent means a patient has the right to understand the risks of death, serious bodily injury, or other common outcomes of an operation or medical treatment. The patient also has the right to be told about the risks of refusing a particular operation, test, medication, or other medical intervention.
If a patient is reluctant or noncompliant, you may not be doing enough if you simply document that he or she refused your recommendation of treatment. You should also make a record of your efforts to explain to the patient the risks of refusing that treatment.
Informed refusal unfolds in 4 steps
Keep in mind these 4 components:
- The physician determines the patient needs a particular operation, test, medication, or other type of medical intervention
- The physician tells the patient about the needed intervention
- The patient refuses the recommended treatment for any reason: “I don’t think I need that test.” “I don’t like needles.” “I don’t care if I die.”
- The physician explains the risks of not having the treatment so the patient can make an informed decision when refusing it.
The courts first recognized the concept of informed refusal in a case in California more than 30 years ago. A woman seeing her long-time gynecologist was advised to have a Pap smear. Despite repeated recommendations, the patient declined the test. She later developed cervical cancer and sued her physician, claiming malpractice.
Because the physician and patient agreed the former had recommended the Pap smear, the trial judge threw the case out.
He determined it was the patient’s fault that she did not get the test that could have alerted her to the presence of cancer.
A higher court, however, reversed that decision because the patient had the right to make an informed decision in refusing the test—informed refusal.
The appeals judge ruled the gynecologist had a duty not only to recommend the test, but also to make sure the patient understood the consequences of her refusal. Because the evidence of informed refusal was lacking, the case was returned to the lower court.
Case: Patient refuses CT scan, dies; suit follows
The issue of informed refusal was used in a 1996 trial, about 20 years after the doctrine was first recognized in California. The emergency department (ED) physician in that case was found responsible for a patient’s death.
The patient, who had a long history of alcoholism, had fallen at home and struck his head. He was unconscious briefly before his wife took him to the ED. Although the examination did not reveal any neurologic abnormalities, the physician recommended a computed tomography (CT) scan of the head.
According to the physician, the patient and his wife refused the CT scan because they did not have health insurance. The physician failed to document his recommendation of the scan or the discussion in which the patient refused it. The patient and his wife left the ED. The next day, the patient died of multiple subdural bleeds. The widow sued for wrongful death.
Was the recommendation made?
During the trial, the physician testified that he had recommended a CT scan to the patient and his wife. The wife denied receiving such a recommendation. The trial attorneys focused considerable attention on whether the recommendation had actually been made. No one really focused on whether the physician advised the patient about the risks of refusal.
At the end of the trial, the plaintiff’s lawyer cleverly requested that the judge instruct the jury on the concept of informed refusal, knowing there had been no evidence that the doctor had advised the patient or his wife of the risk of refusing the CT scan.
The jury concluded that the physician did recommend the scan but failed to advise the patient or his wife of the risks of not having it. The trial ended in a plaintiff’s verdict of several hundred thousand dollars.
What’s happening nationally?
Four other states besides California have considered legislation regarding informed refusal: Nevada, Vermont, and Michigan have passed laws recognizing its existence, and Mississippi recognized the concept even though an informed refusal bill was defeated in the legislature.
Informed refusal is embodied in court decisions in other states. I have found no state where the doctrine has been overtly rejected.
If you want to learn more about patient safety and liability, the patient safety committee of the American College of Surgeons has published a booklet and manual, available on the organization’s Web site (www.facs.org/commerce/catsplash.html), containing essential information for surgeons and other physicians.1,2
The Joint Commission on Accreditation of Healthcare Organizations offers a 50-page book, available free online: Health Care at the Crossroads: Strategies for Improving the Medical Liability System and Preventing Patient Injury.3 It is available at www.jointcommission.org/NR/rdonlyres/167DD821-A395-48FD-87F9-6AB12BCACB0F/0/Medical_Liability.pdf.
1. Manuel BM, Nora PF, eds. Surgical Patient Safety: Essential Information for Surgeons in Today’s Environment. 05PS-0001. Chicago, Ill: American College of Surgeons; 2005.
2. Professional Liability Committee, American College of Surgeons. Nora PF, ed. Professional Liability/Risk Management: A Manual for Surgeons. 2nd ed. 04PL-0001. Chicago, Ill: American College of Surgeons; 1997.
3. Joint Commission on Accreditation of Healthcare Organizations. Health Care at the Crossroads: Strategies for Improving the Medical Liability System and Preventing Patient Injury. Oakbrook Terrace, Ill: Joint Commission on Accreditation of Healthcare Organizations; 2005.
Timing the conversation
It is not always clear when a physician should acknowledge that a patient has refused a recommended treatment. Patients are often frightened or reluctant about an operation or medical treatment. Some will want time to think about it, talk with friends or family, and, perhaps, get a second opinion.
Suppose you recommend a breast biopsy to a responsible and long-term patient. She may well want to talk to her husband or close friend about it before making a decision. You should not feel compelled to say, “Fine, but you could die of breast cancer if you don’t have this done.” The informed refusal discussion should occur when a patient makes it clear that she has rejected your recommendation.
A risk-management professional might say that the sooner you have this conversation with the patient and document it, the better. But you have to base your timing on the situation and your assessment of the patient’s reliability.
Harsh words are unnecessary
The informed refusal conversation need not take place immediately with a patient who seems reasonable and thoughtful about the process. However, obtaining informed refusal on the spot is worth the effort if you doubt the patient will return to give you a decision.
Maintaining the patient’s trust is important. Do not be so concerned about protecting yourself from a malpractice suit that you constantly make harsh comments about what could happen if patients reject medical advice. The key is for you to assess the patient’s likelihood to respond later.
This article is the first in a series of 4 derived from a symposium on malpractice risk management at the 91st Clinical Congress of the American College of Surgeons, San Francisco, Calif, in October 2005. Mr. Goodman updated his comments in October 2006.
Part 1
Informed refusal
James M. Goodman, JD
Part 2
Common errors in self-defense
Claudia Dobbs, MA
Part 3
Patient safety as risk management tool
Thomas J. Donnelly, JD
Part 4
Responsibilities in obtaining informed consent
James M. Nelson, JD
The concept of informed refusal is similar to that of informed consent. However, in working with physicians for 30 years, I have found that informed refusal is not nearly as well understood as informed consent.
Informed consent means a patient has the right to understand the risks of death, serious bodily injury, or other common outcomes of an operation or medical treatment. The patient also has the right to be told about the risks of refusing a particular operation, test, medication, or other medical intervention.
If a patient is reluctant or noncompliant, you may not be doing enough if you simply document that he or she refused your recommendation of treatment. You should also make a record of your efforts to explain to the patient the risks of refusing that treatment.
Informed refusal unfolds in 4 steps
Keep in mind these 4 components:
- The physician determines the patient needs a particular operation, test, medication, or other type of medical intervention
- The physician tells the patient about the needed intervention
- The patient refuses the recommended treatment for any reason: “I don’t think I need that test.” “I don’t like needles.” “I don’t care if I die.”
- The physician explains the risks of not having the treatment so the patient can make an informed decision when refusing it.
The courts first recognized the concept of informed refusal in a case in California more than 30 years ago. A woman seeing her long-time gynecologist was advised to have a Pap smear. Despite repeated recommendations, the patient declined the test. She later developed cervical cancer and sued her physician, claiming malpractice.
Because the physician and patient agreed the former had recommended the Pap smear, the trial judge threw the case out.
He determined it was the patient’s fault that she did not get the test that could have alerted her to the presence of cancer.
A higher court, however, reversed that decision because the patient had the right to make an informed decision in refusing the test—informed refusal.
The appeals judge ruled the gynecologist had a duty not only to recommend the test, but also to make sure the patient understood the consequences of her refusal. Because the evidence of informed refusal was lacking, the case was returned to the lower court.
Case: Patient refuses CT scan, dies; suit follows
The issue of informed refusal was used in a 1996 trial, about 20 years after the doctrine was first recognized in California. The emergency department (ED) physician in that case was found responsible for a patient’s death.
The patient, who had a long history of alcoholism, had fallen at home and struck his head. He was unconscious briefly before his wife took him to the ED. Although the examination did not reveal any neurologic abnormalities, the physician recommended a computed tomography (CT) scan of the head.
According to the physician, the patient and his wife refused the CT scan because they did not have health insurance. The physician failed to document his recommendation of the scan or the discussion in which the patient refused it. The patient and his wife left the ED. The next day, the patient died of multiple subdural bleeds. The widow sued for wrongful death.
Was the recommendation made?
During the trial, the physician testified that he had recommended a CT scan to the patient and his wife. The wife denied receiving such a recommendation. The trial attorneys focused considerable attention on whether the recommendation had actually been made. No one really focused on whether the physician advised the patient about the risks of refusal.
At the end of the trial, the plaintiff’s lawyer cleverly requested that the judge instruct the jury on the concept of informed refusal, knowing there had been no evidence that the doctor had advised the patient or his wife of the risk of refusing the CT scan.
The jury concluded that the physician did recommend the scan but failed to advise the patient or his wife of the risks of not having it. The trial ended in a plaintiff’s verdict of several hundred thousand dollars.
What’s happening nationally?
Four other states besides California have considered legislation regarding informed refusal: Nevada, Vermont, and Michigan have passed laws recognizing its existence, and Mississippi recognized the concept even though an informed refusal bill was defeated in the legislature.
Informed refusal is embodied in court decisions in other states. I have found no state where the doctrine has been overtly rejected.
If you want to learn more about patient safety and liability, the patient safety committee of the American College of Surgeons has published a booklet and manual, available on the organization’s Web site (www.facs.org/commerce/catsplash.html), containing essential information for surgeons and other physicians.1,2
The Joint Commission on Accreditation of Healthcare Organizations offers a 50-page book, available free online: Health Care at the Crossroads: Strategies for Improving the Medical Liability System and Preventing Patient Injury.3 It is available at www.jointcommission.org/NR/rdonlyres/167DD821-A395-48FD-87F9-6AB12BCACB0F/0/Medical_Liability.pdf.
1. Manuel BM, Nora PF, eds. Surgical Patient Safety: Essential Information for Surgeons in Today’s Environment. 05PS-0001. Chicago, Ill: American College of Surgeons; 2005.
2. Professional Liability Committee, American College of Surgeons. Nora PF, ed. Professional Liability/Risk Management: A Manual for Surgeons. 2nd ed. 04PL-0001. Chicago, Ill: American College of Surgeons; 1997.
3. Joint Commission on Accreditation of Healthcare Organizations. Health Care at the Crossroads: Strategies for Improving the Medical Liability System and Preventing Patient Injury. Oakbrook Terrace, Ill: Joint Commission on Accreditation of Healthcare Organizations; 2005.
Timing the conversation
It is not always clear when a physician should acknowledge that a patient has refused a recommended treatment. Patients are often frightened or reluctant about an operation or medical treatment. Some will want time to think about it, talk with friends or family, and, perhaps, get a second opinion.
Suppose you recommend a breast biopsy to a responsible and long-term patient. She may well want to talk to her husband or close friend about it before making a decision. You should not feel compelled to say, “Fine, but you could die of breast cancer if you don’t have this done.” The informed refusal discussion should occur when a patient makes it clear that she has rejected your recommendation.
A risk-management professional might say that the sooner you have this conversation with the patient and document it, the better. But you have to base your timing on the situation and your assessment of the patient’s reliability.
Harsh words are unnecessary
The informed refusal conversation need not take place immediately with a patient who seems reasonable and thoughtful about the process. However, obtaining informed refusal on the spot is worth the effort if you doubt the patient will return to give you a decision.
Maintaining the patient’s trust is important. Do not be so concerned about protecting yourself from a malpractice suit that you constantly make harsh comments about what could happen if patients reject medical advice. The key is for you to assess the patient’s likelihood to respond later.
In the Literature
Hospital Quality for AMI: Process Measures and Their Relationship with Short-term Mortality
Bradley EH, Herrin J, Elbel B, et al. Hospital quality for acute myocardial infarction: correlation among process measures and relationship with short-term mortality. JAMA. 2006 Jul 5;296(1):72-78.
Background
The Centers for Medicare and Medicaid Services (CMS) and the Joint Commission on Accreditation of Healthcare Organizations (JCAHO) monitor and publicly report hospital performance in the treatment of acute myocardial infarction (AMI). Core process measures are considered an indicator of quality of care, but little is known about how these measures affect outcomes (mortality). Five of the seven core measures for AMI assess medication prescription practices; the other two measures are counseling on smoking cessation and timely reperfusion therapy.
Inferences about a hospital’s quality of care for AMI are created by measuring the hospital’s success at performing these measures. No previous study had evaluated a possible correlation between performance on these measures and short-term mortality. The authors of this study used National Registry of Myocardial Infarction (NRMI) and CMS databases to determine the association between hospital performance on AMI process measures and hospital-specific, risk-standardized, 30-day mortality rates.
Methods
A cross-sectional study was performed using hospitals that reported AMI discharges to the NRMI from January 2002 through March 2003. Hospitals had to report a minimum of 10 eligible patients. Hospital performance on core measures was recorded: beta-blocker on admission, beta-blocker on dismissal, aspirin on admission, aspirin on dismissal, angiotensin-converting enzyme inhibitor (ACE) prescription on dismissal, smoking cessation counseling for smokers during admission, and time to reperfusion therapy. Risk-standardized, 30-day, all-cause mortality rates were calculated for each hospital using CMS Medicare claims for patients ages 66 and older with AMI. The primary analysis determined the association of hospital-specific, risk-standardized, 30-day mortality rates with hospital performance on the core process measures.
Results
The most successfully completed core process measure for AMI was aspirin on admission. A mean of 86.4% of participating hospitals completed this measure. The core process measure for AMI that was the least frequently documented was smoking cessation counseling; a mean of 13.9% of participating hospitals completed this measure. Notably, timely reperfusion therapy for AMI—fibrinolytic therapy within 30 minutes of arrival or percutaneous intervention within 120 minutes of arrival—was completed by only 54.5% (mean) of participating hospitals.
Each core process measure had a statistically significant but small correlation with the risk-standardized, 30-day mortality rate (explaining between 0.1% and 3.3% of variance in mortality). Of the 180 hospitals in the top quintile of risk-standardized, 30-day mortality rates, only 31% were in the top quintile of the core process measures. A composite model of all seven core process measures determined that these measures could only explain 6% of the hospital-level variation in risk-standardized, 30-day mortality rates. Secondary analyses did not differ substantially.
Conclusions
In this study, each core process measure for AMI showed a modest correlation with 30-day mortality, but accounted for only 6% of 30-day mortality. This finding highlights the fact that continued measurement of these processes is valuable, but a hospital’s short-term mortality rates for AMI cannot be reliably inferred from performance on publicly reported process measures. These measures are weighted more toward long-term outcome measures. There is a need for new research to define and study new AMI process measures that can explain more of the variance in both short- and long-term outcomes.
Clopidogrel and Aspirin versus Aspirin Alone for the Prevention of Atherothrombotic Events
Bhatt DL, Fox KA, Hacke W, et al. Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events. N Engl J Med. 2006 Apr 20;354(16):1706-1717.
Background
Atherothrombotic disorders of the circulatory system are the leading cause of death and disability in the world. Low-dose aspirin has been shown to reduce ischemic event in populations above a certain risk threshold; however, aspirin alone may be insufficient treatment to prevent ischemic events in high-risk patients. Dual antiplatelet therapy with aspirin and clopidogrel has been shown to reduce ischemic events in patients with unstable angina, non-ST segment elevation and ST segment elevation myocardial infarction, as well as in those undergoing angioplasty and stenting.
Methods
This was a prospective, multicenter, randomized, double-blind, placebo-controlled study of the efficacy and safety of aspirin plus clopidogrel in comparison with aspirin plus placebo in patients at high risk for a cardiovascular event. Patients included in the study were 45 or older and had one of the following: multiple atherothrombotic risk factors, documented coronary artery disease, documented cerebrovascular disease, or documented symptomatic peripheral vascular disease. The primary efficacy endpoint was the first occurrence of myocardial infarction (MI), stroke, or death from cardiovascular causes. The primary safety endpoint was severe bleeding.
Results
A total of 15,603 patients were enrolled in the study. Treatment was permanently discontinued by 20.4% in the clopidogrel group as compared with 18.2% in the placebo group (P<0.001). A total of 4.8% of patients in the clopidogrel group and 4.9% in the placebo group discontinued treatment because of an adverse event (P=0.67). Other than the treatment medications, concomitant medication use was similar in both groups. A median follow-up of 28 months revealed that the rates of primary efficacy events in the clopidogrel and placebo group were similar (6.8% versus 7.3%, P=0.22, respectively). The rate of primary safety events was 1.7% in the clopidogrel group and 1.3% in the placebo group, P=0.09.
Conclusions
This trial enrolled patients who either had established atherothrombotic disease or were at high risk for such disease and found that there was no significant benefit associated with the use of clopidogrel plus aspirin compared to aspirin alone in reducing myocardial infarction, stroke, or cardiovascular death. The risk of moderate or severe bleeding in symptomatic patients was higher in the clopidogrel plus aspirin group than in the aspirin plus placebo group. Overall, these findings do not support the use of dual antiplatelet therapy across this broad patient population.
D-Dimer in the Diagnosis of Pulmonary Embolism
Kearon C, Ginsberg JS, Douketis J, et al. An evaluation of D-dimer in the diagnosis of pulmonary embolism: a randomized trial. Ann Intern Med. 2006 Jun 6;144(11):812-821.
Background
The clinical usefulness of the D-dimer test in the diagnosis of pulmonary embolism (PE) has been previously studied. In patients with suspected PE, it may be safe to omit additional diagnostic testing if a patient has a negative D-dimer test; however, this approach has never been evaluated in a randomized, controlled trial.
The investigators in this trial studied two subgroups of patients with suspected PE and a negative D-dimer: patients with a low clinical probability of PE and those with a moderate or high clinical probability of PE who had a non-diagnostic ventilation perfusion scan (VQ scan) and no proximal deep vein thrombosis on venous ultrasonography. The hypothesis was that patients with a negative D-dimer who do not have further testing for PE won’t have a higher frequency of venous thromboembolism during follow-up than patients who undergo routine diagnostic testing.
Methods
Before any diagnostic testing, patients were assigned a probability score, using the Wells Criteria, to categorize the probability of PE as low or moderate to high.
Patients with low probability Wells scores: D-dimer testing was done on all patients with a low clinical probability of PE. Patients with a negative D-dimer were randomly assigned either to no additional diagnostic testing and no anticoagulation treatment or to additional diagnostic testing with an initial VQ scan. If the VQ scan was negative, then PE was excluded. If the VQ scan showed one or more segmental perfusion defects that were normally ventilated, then the scan was considered diagnostic for PE. If there were perfusion defects that did not meet the criteria for a high probability scan, then the scan was considered non-diagnostic. Patients with non-diagnostic scans underwent ultrasonography of the proximal veins of the legs. If deep vein thrombosis was present, PE was diagnosed. If ultrasonography was normal, the test was repeated after seven and 14 days. In all patients with a positive D-dimer, a VQ scan was performed.
Patients with moderate to high probability Wells scores: A VQ scan was performed on all patients with a moderate to high probability for PE. Patients with high probability scans were treated; patients with normal scans were not treated. Patients with non-diagnostic scans and normal venous ultrasonography were randomly assigned to receive either no additional testing or serial ultrasonography.
Outcomes: All patients were followed for six months for the development of venous thromboembolism after initial diagnostic testing.
Results
The study enrolled 1,126 patients. Overall, 160 patients (14.2%) had PE diagnosed at initial presentation or by venous ultrasonography. Of 952 patients who did not receive an initial diagnosis of PE, 11 (1.2%) had PE diagnosed at follow-up.
Patients with low probability Wells scores: Low clinical probability was present in 670 patients (60%). In patients with low clinical probability of PE, 373 (56%) had negative D-dimer tests and 297 (44%) had positive D-dimer tests. Of the 373 patients with low probability and negative D-dimer results, 187 were randomized to no additional testing and 186 received a VQ scan. The frequency of venous thromboembolism at six-month follow-up was similar in these two groups (-0.5% [CI, -3.0% to 1.6%]). Three patients with negative D-dimer tests were diagnosed with PE by VQ scan. Results were fairly complete (five patients without a six-month follow-up in the no additional testing group and one without a follow-up in the VQ scan group).
Twenty-four patients with low clinical probability and positive D-dimer results (n=297) were diagnosed with PE. Three patients did not complete the six-month follow-up. Of the remaining 294 patients, five patients had venous thromboembolism at six months.
Patients with moderate to high probability Wells scores: There were 456 patients (40%) had moderate or high clinical probability for PE. Each of these patients had a VQ scan. Non-diagnostic VQ scans and normal venous ultrasonography were performed on 226 patients. Of these 226 patients, 86 had a negative D-dimer and 140 had a positive D-dimer. Of the 86 patients with negative VQ scans, normal venous ultrasonography, and a negative D-dimer, 83 were randomly assigned to no additional testing or serial venous ultrasonography (42 and 41 respectively). At six months follow-up, one patient assigned to no additional testing had venous thromboembolism, and no patients in the additional testing group had venous thromboembolism.
Conclusions
The results of this trial suggest that it is safe to withhold additional diagnostic testing in patients with suspected PE, low pretest clinical probability, and a negative D-dimer test. Even in patients with moderate to high pretest clinical probability, a non-diagnostic VQ scan, and normal venous ultrasonography, only one patient with a negative D-dimer had a venous thromboembolic event at six months follow-up. The assay used for this study was an erythrocyte agglutination SimpliRED assay. Reported sensitivity is approximately 90%, and specificity is approximately 75%. To the readers, it should be noted that the authors defined a low probability Wells score as 4 rather than 1.5 or lower.
Efficacy and Safety of Inhaled Insulin Therapy in Adults with Diabetes Mellitus
Ceglia L, Lau J, Pittas AG. Meta-analysis: efficacy and safety of inhaled insulin therapy in adults with diabetes mellitus. Ann Intern Med. 2006 Nov 7;145(9):665-675.
Background
Despite its effectiveness in attaining glycemic control, there is considerable resistance to insulin use by patients and healthcare providers, primarily because of the need for subcutaneous injection. In January 2006, the U.S. Food and Drug Administration (FDA) approved the first formulation of inhaled insulin for clinical use in nonsmoking adults with type 1 or type 2 diabetes and no pulmonary disease. The authors of this paper present a systematic review to examine the efficacy, safety, and patient acceptability of inhaled insulin.
Methods
The authors conducted a search of MEDLINE to find English-language, randomized, controlled trials of inhaled insulin in nonpregnant adults with diabetes. To find unpublished studies, the authors reviewed the briefing document on Exubera powder for oral inhalation (Pfizer Inc., New York). An abstract was included if it reported original data from controlled trials in patients with type 1 or 2 diabetes and hemoglobin A1C outcomes for patients receiving inhaled insulin versus outcomes for a comparison group (subcutaneous insulin or oral hypoglycemics). Studies with less than 12 weeks duration were excluded because no comparison could be made regarding glycemic efficacy. For glycemic efficacy, the primary outcome was the treatment group difference in hemoglobin A1C from baseline. Secondary outcome was the proportion of patients with hemoglobin A1C levels less than 7%. To evaluate safety, the primary outcomes were severe hypoglycemia (glucose ≤36 mg/dL), cough, and treatment group difference in pulmonary function variables.
Results
Sixteen trials involving a total of 4,023 patients met inclusion criteria. Seven trials compared inhaled insulin with various subcutaneous insulin regimens in patients with type 1 diabetes. Nine trials compared inhaled insulin with subcutaneous insulin or oral hypoglycemic agents in patients with type 2 diabetes. Inhaled insulin was given with meals and titrated according to study-specific glucose goals. Subcutaneous insulin was titrated to the same specific goals. Doses of oral hypoglycemic agents were adjusted for glycemic targets in only two of the nine trials. The combined data from the studies demonstrated a small but statistically significant decrease in the levels of baseline hemoglobin A1C levels in favor of subcutaneous insulin (weighted mean difference 0.08%, [CI 0.03% to 0.14%]) in patients with type 1 or type 2 diabetes. The greatest advantage of subcutaneous insulin was noted in the study with the longest duration (104 weeks). There was no difference between the study groups in studies with duration of 24 weeks or less. Patients with type 1 or type 2 diabetes taking inhaled insulin were no more likely to achieve hemoglobin A1C levels less than 7% than those using subcutaneous insulin.
The combined data from studies comparing inhaled insulin to oral hypoglycemic agents in patients with type 2 diabetes showed that inhaled insulin lowered hemoglobin A1C levels more effectively (weighted mean difference -1.04%, [CI -1.59% to -0.49%]). In studies in which the oral hypoglycemic agents were titrated, inhaled insulin still lowered baseline hemoglobin A1C levels but to a lesser degree (weighted mean difference -0.20%, [CI - 0.34% to -0.07%]). Patients with type 2 diabetes taking inhaled insulin were more likely to achieve hemoglobin A1C levels less than 7% than those taking oral agents.
There was no difference in episodes of severe hypoglycemia in patients using inhaled insulin compared to those using subcutaneous insulin. A higher proportion of patients using inhaled insulin reported at least one episode of severe hypoglycemia compared to those using oral agents (risk ratio, 3.06 [CI 1.03 to 9.07]; 9.4% versus 3.5%, respectively).
With respect to pulmonary safety, all trials selected patients without histories of pulmonary problems and with at least six months of nonsmoking status. Pulmonary safety was assessed by self-reported symptoms and by pulmonary function tests. The most common pulmonary symptom associated with inhaled insulin was nonproductive cough. This symptom was reported more frequently compared to patients using subcutaneous insulin or oral agents (risk ratio, 3.52 [CI 2.23 to 5.56]; 16.9% versus 5.0%). Cough was noted early in the treatment course and diminished in frequency over time. Patients receiving inhaled insulin had a greater decrease in FEV1 (forced expiratory volume in the first second) from baseline than the comparator group (weighted mean difference, -0.031 L [CI-0.043 L to -0.020 L]). This decrease progressed slowly over the first six months but stabilized in studies of up to two years’ duration.
Only four trials reported data on overall patient satisfaction for inhaled insulin versus subcutaneous insulin. All trials reported a statistically significant increase in overall patient satisfaction with inhaled insulin over subcutaneous insulin. Patients randomly assigned to inhaled insulin were more likely to continue taking inhaled insulin than to switch back to subcutaneous insulin.
Conclusions
This meta-analysis showed that inhaled insulin is comparable to subcutaneous insulin in lowering hemoglobin A1C levels in patients with type 1 or type 2 diabetes. The proportion of patients reaching a target hemoglobin A1C of less than 7% was much lower in the studies in this meta-analysis as compared to levels in trials of intensive subcutaneous insulin therapy.
It’s more difficult to compare inhaled insulin with oral hypoglycemic agents because most studies involving oral agents used fixed dosing with different types of oral agents. There was a three-fold risk of severe hypoglycemia in patients using inhaled insulin compared to those using oral hypoglycemic agents. This is probably due to overall improved glycemic control in the inhaled insulin group. Cough was more common in the inhaled insulin groups, and there were small decreases in FEV1, but these did not progress over two years. The potential for pulmonary toxicity with long-term administration has not been evaluated and deserves further study.
Hospital Quality for AMI: Process Measures and Their Relationship with Short-term Mortality
Bradley EH, Herrin J, Elbel B, et al. Hospital quality for acute myocardial infarction: correlation among process measures and relationship with short-term mortality. JAMA. 2006 Jul 5;296(1):72-78.
Background
The Centers for Medicare and Medicaid Services (CMS) and the Joint Commission on Accreditation of Healthcare Organizations (JCAHO) monitor and publicly report hospital performance in the treatment of acute myocardial infarction (AMI). Core process measures are considered an indicator of quality of care, but little is known about how these measures affect outcomes (mortality). Five of the seven core measures for AMI assess medication prescription practices; the other two measures are counseling on smoking cessation and timely reperfusion therapy.
Inferences about a hospital’s quality of care for AMI are created by measuring the hospital’s success at performing these measures. No previous study had evaluated a possible correlation between performance on these measures and short-term mortality. The authors of this study used National Registry of Myocardial Infarction (NRMI) and CMS databases to determine the association between hospital performance on AMI process measures and hospital-specific, risk-standardized, 30-day mortality rates.
Methods
A cross-sectional study was performed using hospitals that reported AMI discharges to the NRMI from January 2002 through March 2003. Hospitals had to report a minimum of 10 eligible patients. Hospital performance on core measures was recorded: beta-blocker on admission, beta-blocker on dismissal, aspirin on admission, aspirin on dismissal, angiotensin-converting enzyme inhibitor (ACE) prescription on dismissal, smoking cessation counseling for smokers during admission, and time to reperfusion therapy. Risk-standardized, 30-day, all-cause mortality rates were calculated for each hospital using CMS Medicare claims for patients ages 66 and older with AMI. The primary analysis determined the association of hospital-specific, risk-standardized, 30-day mortality rates with hospital performance on the core process measures.
Results
The most successfully completed core process measure for AMI was aspirin on admission. A mean of 86.4% of participating hospitals completed this measure. The core process measure for AMI that was the least frequently documented was smoking cessation counseling; a mean of 13.9% of participating hospitals completed this measure. Notably, timely reperfusion therapy for AMI—fibrinolytic therapy within 30 minutes of arrival or percutaneous intervention within 120 minutes of arrival—was completed by only 54.5% (mean) of participating hospitals.
Each core process measure had a statistically significant but small correlation with the risk-standardized, 30-day mortality rate (explaining between 0.1% and 3.3% of variance in mortality). Of the 180 hospitals in the top quintile of risk-standardized, 30-day mortality rates, only 31% were in the top quintile of the core process measures. A composite model of all seven core process measures determined that these measures could only explain 6% of the hospital-level variation in risk-standardized, 30-day mortality rates. Secondary analyses did not differ substantially.
Conclusions
In this study, each core process measure for AMI showed a modest correlation with 30-day mortality, but accounted for only 6% of 30-day mortality. This finding highlights the fact that continued measurement of these processes is valuable, but a hospital’s short-term mortality rates for AMI cannot be reliably inferred from performance on publicly reported process measures. These measures are weighted more toward long-term outcome measures. There is a need for new research to define and study new AMI process measures that can explain more of the variance in both short- and long-term outcomes.
Clopidogrel and Aspirin versus Aspirin Alone for the Prevention of Atherothrombotic Events
Bhatt DL, Fox KA, Hacke W, et al. Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events. N Engl J Med. 2006 Apr 20;354(16):1706-1717.
Background
Atherothrombotic disorders of the circulatory system are the leading cause of death and disability in the world. Low-dose aspirin has been shown to reduce ischemic event in populations above a certain risk threshold; however, aspirin alone may be insufficient treatment to prevent ischemic events in high-risk patients. Dual antiplatelet therapy with aspirin and clopidogrel has been shown to reduce ischemic events in patients with unstable angina, non-ST segment elevation and ST segment elevation myocardial infarction, as well as in those undergoing angioplasty and stenting.
Methods
This was a prospective, multicenter, randomized, double-blind, placebo-controlled study of the efficacy and safety of aspirin plus clopidogrel in comparison with aspirin plus placebo in patients at high risk for a cardiovascular event. Patients included in the study were 45 or older and had one of the following: multiple atherothrombotic risk factors, documented coronary artery disease, documented cerebrovascular disease, or documented symptomatic peripheral vascular disease. The primary efficacy endpoint was the first occurrence of myocardial infarction (MI), stroke, or death from cardiovascular causes. The primary safety endpoint was severe bleeding.
Results
A total of 15,603 patients were enrolled in the study. Treatment was permanently discontinued by 20.4% in the clopidogrel group as compared with 18.2% in the placebo group (P<0.001). A total of 4.8% of patients in the clopidogrel group and 4.9% in the placebo group discontinued treatment because of an adverse event (P=0.67). Other than the treatment medications, concomitant medication use was similar in both groups. A median follow-up of 28 months revealed that the rates of primary efficacy events in the clopidogrel and placebo group were similar (6.8% versus 7.3%, P=0.22, respectively). The rate of primary safety events was 1.7% in the clopidogrel group and 1.3% in the placebo group, P=0.09.
Conclusions
This trial enrolled patients who either had established atherothrombotic disease or were at high risk for such disease and found that there was no significant benefit associated with the use of clopidogrel plus aspirin compared to aspirin alone in reducing myocardial infarction, stroke, or cardiovascular death. The risk of moderate or severe bleeding in symptomatic patients was higher in the clopidogrel plus aspirin group than in the aspirin plus placebo group. Overall, these findings do not support the use of dual antiplatelet therapy across this broad patient population.
D-Dimer in the Diagnosis of Pulmonary Embolism
Kearon C, Ginsberg JS, Douketis J, et al. An evaluation of D-dimer in the diagnosis of pulmonary embolism: a randomized trial. Ann Intern Med. 2006 Jun 6;144(11):812-821.
Background
The clinical usefulness of the D-dimer test in the diagnosis of pulmonary embolism (PE) has been previously studied. In patients with suspected PE, it may be safe to omit additional diagnostic testing if a patient has a negative D-dimer test; however, this approach has never been evaluated in a randomized, controlled trial.
The investigators in this trial studied two subgroups of patients with suspected PE and a negative D-dimer: patients with a low clinical probability of PE and those with a moderate or high clinical probability of PE who had a non-diagnostic ventilation perfusion scan (VQ scan) and no proximal deep vein thrombosis on venous ultrasonography. The hypothesis was that patients with a negative D-dimer who do not have further testing for PE won’t have a higher frequency of venous thromboembolism during follow-up than patients who undergo routine diagnostic testing.
Methods
Before any diagnostic testing, patients were assigned a probability score, using the Wells Criteria, to categorize the probability of PE as low or moderate to high.
Patients with low probability Wells scores: D-dimer testing was done on all patients with a low clinical probability of PE. Patients with a negative D-dimer were randomly assigned either to no additional diagnostic testing and no anticoagulation treatment or to additional diagnostic testing with an initial VQ scan. If the VQ scan was negative, then PE was excluded. If the VQ scan showed one or more segmental perfusion defects that were normally ventilated, then the scan was considered diagnostic for PE. If there were perfusion defects that did not meet the criteria for a high probability scan, then the scan was considered non-diagnostic. Patients with non-diagnostic scans underwent ultrasonography of the proximal veins of the legs. If deep vein thrombosis was present, PE was diagnosed. If ultrasonography was normal, the test was repeated after seven and 14 days. In all patients with a positive D-dimer, a VQ scan was performed.
Patients with moderate to high probability Wells scores: A VQ scan was performed on all patients with a moderate to high probability for PE. Patients with high probability scans were treated; patients with normal scans were not treated. Patients with non-diagnostic scans and normal venous ultrasonography were randomly assigned to receive either no additional testing or serial ultrasonography.
Outcomes: All patients were followed for six months for the development of venous thromboembolism after initial diagnostic testing.
Results
The study enrolled 1,126 patients. Overall, 160 patients (14.2%) had PE diagnosed at initial presentation or by venous ultrasonography. Of 952 patients who did not receive an initial diagnosis of PE, 11 (1.2%) had PE diagnosed at follow-up.
Patients with low probability Wells scores: Low clinical probability was present in 670 patients (60%). In patients with low clinical probability of PE, 373 (56%) had negative D-dimer tests and 297 (44%) had positive D-dimer tests. Of the 373 patients with low probability and negative D-dimer results, 187 were randomized to no additional testing and 186 received a VQ scan. The frequency of venous thromboembolism at six-month follow-up was similar in these two groups (-0.5% [CI, -3.0% to 1.6%]). Three patients with negative D-dimer tests were diagnosed with PE by VQ scan. Results were fairly complete (five patients without a six-month follow-up in the no additional testing group and one without a follow-up in the VQ scan group).
Twenty-four patients with low clinical probability and positive D-dimer results (n=297) were diagnosed with PE. Three patients did not complete the six-month follow-up. Of the remaining 294 patients, five patients had venous thromboembolism at six months.
Patients with moderate to high probability Wells scores: There were 456 patients (40%) had moderate or high clinical probability for PE. Each of these patients had a VQ scan. Non-diagnostic VQ scans and normal venous ultrasonography were performed on 226 patients. Of these 226 patients, 86 had a negative D-dimer and 140 had a positive D-dimer. Of the 86 patients with negative VQ scans, normal venous ultrasonography, and a negative D-dimer, 83 were randomly assigned to no additional testing or serial venous ultrasonography (42 and 41 respectively). At six months follow-up, one patient assigned to no additional testing had venous thromboembolism, and no patients in the additional testing group had venous thromboembolism.
Conclusions
The results of this trial suggest that it is safe to withhold additional diagnostic testing in patients with suspected PE, low pretest clinical probability, and a negative D-dimer test. Even in patients with moderate to high pretest clinical probability, a non-diagnostic VQ scan, and normal venous ultrasonography, only one patient with a negative D-dimer had a venous thromboembolic event at six months follow-up. The assay used for this study was an erythrocyte agglutination SimpliRED assay. Reported sensitivity is approximately 90%, and specificity is approximately 75%. To the readers, it should be noted that the authors defined a low probability Wells score as 4 rather than 1.5 or lower.
Efficacy and Safety of Inhaled Insulin Therapy in Adults with Diabetes Mellitus
Ceglia L, Lau J, Pittas AG. Meta-analysis: efficacy and safety of inhaled insulin therapy in adults with diabetes mellitus. Ann Intern Med. 2006 Nov 7;145(9):665-675.
Background
Despite its effectiveness in attaining glycemic control, there is considerable resistance to insulin use by patients and healthcare providers, primarily because of the need for subcutaneous injection. In January 2006, the U.S. Food and Drug Administration (FDA) approved the first formulation of inhaled insulin for clinical use in nonsmoking adults with type 1 or type 2 diabetes and no pulmonary disease. The authors of this paper present a systematic review to examine the efficacy, safety, and patient acceptability of inhaled insulin.
Methods
The authors conducted a search of MEDLINE to find English-language, randomized, controlled trials of inhaled insulin in nonpregnant adults with diabetes. To find unpublished studies, the authors reviewed the briefing document on Exubera powder for oral inhalation (Pfizer Inc., New York). An abstract was included if it reported original data from controlled trials in patients with type 1 or 2 diabetes and hemoglobin A1C outcomes for patients receiving inhaled insulin versus outcomes for a comparison group (subcutaneous insulin or oral hypoglycemics). Studies with less than 12 weeks duration were excluded because no comparison could be made regarding glycemic efficacy. For glycemic efficacy, the primary outcome was the treatment group difference in hemoglobin A1C from baseline. Secondary outcome was the proportion of patients with hemoglobin A1C levels less than 7%. To evaluate safety, the primary outcomes were severe hypoglycemia (glucose ≤36 mg/dL), cough, and treatment group difference in pulmonary function variables.
Results
Sixteen trials involving a total of 4,023 patients met inclusion criteria. Seven trials compared inhaled insulin with various subcutaneous insulin regimens in patients with type 1 diabetes. Nine trials compared inhaled insulin with subcutaneous insulin or oral hypoglycemic agents in patients with type 2 diabetes. Inhaled insulin was given with meals and titrated according to study-specific glucose goals. Subcutaneous insulin was titrated to the same specific goals. Doses of oral hypoglycemic agents were adjusted for glycemic targets in only two of the nine trials. The combined data from the studies demonstrated a small but statistically significant decrease in the levels of baseline hemoglobin A1C levels in favor of subcutaneous insulin (weighted mean difference 0.08%, [CI 0.03% to 0.14%]) in patients with type 1 or type 2 diabetes. The greatest advantage of subcutaneous insulin was noted in the study with the longest duration (104 weeks). There was no difference between the study groups in studies with duration of 24 weeks or less. Patients with type 1 or type 2 diabetes taking inhaled insulin were no more likely to achieve hemoglobin A1C levels less than 7% than those using subcutaneous insulin.
The combined data from studies comparing inhaled insulin to oral hypoglycemic agents in patients with type 2 diabetes showed that inhaled insulin lowered hemoglobin A1C levels more effectively (weighted mean difference -1.04%, [CI -1.59% to -0.49%]). In studies in which the oral hypoglycemic agents were titrated, inhaled insulin still lowered baseline hemoglobin A1C levels but to a lesser degree (weighted mean difference -0.20%, [CI - 0.34% to -0.07%]). Patients with type 2 diabetes taking inhaled insulin were more likely to achieve hemoglobin A1C levels less than 7% than those taking oral agents.
There was no difference in episodes of severe hypoglycemia in patients using inhaled insulin compared to those using subcutaneous insulin. A higher proportion of patients using inhaled insulin reported at least one episode of severe hypoglycemia compared to those using oral agents (risk ratio, 3.06 [CI 1.03 to 9.07]; 9.4% versus 3.5%, respectively).
With respect to pulmonary safety, all trials selected patients without histories of pulmonary problems and with at least six months of nonsmoking status. Pulmonary safety was assessed by self-reported symptoms and by pulmonary function tests. The most common pulmonary symptom associated with inhaled insulin was nonproductive cough. This symptom was reported more frequently compared to patients using subcutaneous insulin or oral agents (risk ratio, 3.52 [CI 2.23 to 5.56]; 16.9% versus 5.0%). Cough was noted early in the treatment course and diminished in frequency over time. Patients receiving inhaled insulin had a greater decrease in FEV1 (forced expiratory volume in the first second) from baseline than the comparator group (weighted mean difference, -0.031 L [CI-0.043 L to -0.020 L]). This decrease progressed slowly over the first six months but stabilized in studies of up to two years’ duration.
Only four trials reported data on overall patient satisfaction for inhaled insulin versus subcutaneous insulin. All trials reported a statistically significant increase in overall patient satisfaction with inhaled insulin over subcutaneous insulin. Patients randomly assigned to inhaled insulin were more likely to continue taking inhaled insulin than to switch back to subcutaneous insulin.
Conclusions
This meta-analysis showed that inhaled insulin is comparable to subcutaneous insulin in lowering hemoglobin A1C levels in patients with type 1 or type 2 diabetes. The proportion of patients reaching a target hemoglobin A1C of less than 7% was much lower in the studies in this meta-analysis as compared to levels in trials of intensive subcutaneous insulin therapy.
It’s more difficult to compare inhaled insulin with oral hypoglycemic agents because most studies involving oral agents used fixed dosing with different types of oral agents. There was a three-fold risk of severe hypoglycemia in patients using inhaled insulin compared to those using oral hypoglycemic agents. This is probably due to overall improved glycemic control in the inhaled insulin group. Cough was more common in the inhaled insulin groups, and there were small decreases in FEV1, but these did not progress over two years. The potential for pulmonary toxicity with long-term administration has not been evaluated and deserves further study.
Hospital Quality for AMI: Process Measures and Their Relationship with Short-term Mortality
Bradley EH, Herrin J, Elbel B, et al. Hospital quality for acute myocardial infarction: correlation among process measures and relationship with short-term mortality. JAMA. 2006 Jul 5;296(1):72-78.
Background
The Centers for Medicare and Medicaid Services (CMS) and the Joint Commission on Accreditation of Healthcare Organizations (JCAHO) monitor and publicly report hospital performance in the treatment of acute myocardial infarction (AMI). Core process measures are considered an indicator of quality of care, but little is known about how these measures affect outcomes (mortality). Five of the seven core measures for AMI assess medication prescription practices; the other two measures are counseling on smoking cessation and timely reperfusion therapy.
Inferences about a hospital’s quality of care for AMI are created by measuring the hospital’s success at performing these measures. No previous study had evaluated a possible correlation between performance on these measures and short-term mortality. The authors of this study used National Registry of Myocardial Infarction (NRMI) and CMS databases to determine the association between hospital performance on AMI process measures and hospital-specific, risk-standardized, 30-day mortality rates.
Methods
A cross-sectional study was performed using hospitals that reported AMI discharges to the NRMI from January 2002 through March 2003. Hospitals had to report a minimum of 10 eligible patients. Hospital performance on core measures was recorded: beta-blocker on admission, beta-blocker on dismissal, aspirin on admission, aspirin on dismissal, angiotensin-converting enzyme inhibitor (ACE) prescription on dismissal, smoking cessation counseling for smokers during admission, and time to reperfusion therapy. Risk-standardized, 30-day, all-cause mortality rates were calculated for each hospital using CMS Medicare claims for patients ages 66 and older with AMI. The primary analysis determined the association of hospital-specific, risk-standardized, 30-day mortality rates with hospital performance on the core process measures.
Results
The most successfully completed core process measure for AMI was aspirin on admission. A mean of 86.4% of participating hospitals completed this measure. The core process measure for AMI that was the least frequently documented was smoking cessation counseling; a mean of 13.9% of participating hospitals completed this measure. Notably, timely reperfusion therapy for AMI—fibrinolytic therapy within 30 minutes of arrival or percutaneous intervention within 120 minutes of arrival—was completed by only 54.5% (mean) of participating hospitals.
Each core process measure had a statistically significant but small correlation with the risk-standardized, 30-day mortality rate (explaining between 0.1% and 3.3% of variance in mortality). Of the 180 hospitals in the top quintile of risk-standardized, 30-day mortality rates, only 31% were in the top quintile of the core process measures. A composite model of all seven core process measures determined that these measures could only explain 6% of the hospital-level variation in risk-standardized, 30-day mortality rates. Secondary analyses did not differ substantially.
Conclusions
In this study, each core process measure for AMI showed a modest correlation with 30-day mortality, but accounted for only 6% of 30-day mortality. This finding highlights the fact that continued measurement of these processes is valuable, but a hospital’s short-term mortality rates for AMI cannot be reliably inferred from performance on publicly reported process measures. These measures are weighted more toward long-term outcome measures. There is a need for new research to define and study new AMI process measures that can explain more of the variance in both short- and long-term outcomes.
Clopidogrel and Aspirin versus Aspirin Alone for the Prevention of Atherothrombotic Events
Bhatt DL, Fox KA, Hacke W, et al. Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events. N Engl J Med. 2006 Apr 20;354(16):1706-1717.
Background
Atherothrombotic disorders of the circulatory system are the leading cause of death and disability in the world. Low-dose aspirin has been shown to reduce ischemic event in populations above a certain risk threshold; however, aspirin alone may be insufficient treatment to prevent ischemic events in high-risk patients. Dual antiplatelet therapy with aspirin and clopidogrel has been shown to reduce ischemic events in patients with unstable angina, non-ST segment elevation and ST segment elevation myocardial infarction, as well as in those undergoing angioplasty and stenting.
Methods
This was a prospective, multicenter, randomized, double-blind, placebo-controlled study of the efficacy and safety of aspirin plus clopidogrel in comparison with aspirin plus placebo in patients at high risk for a cardiovascular event. Patients included in the study were 45 or older and had one of the following: multiple atherothrombotic risk factors, documented coronary artery disease, documented cerebrovascular disease, or documented symptomatic peripheral vascular disease. The primary efficacy endpoint was the first occurrence of myocardial infarction (MI), stroke, or death from cardiovascular causes. The primary safety endpoint was severe bleeding.
Results
A total of 15,603 patients were enrolled in the study. Treatment was permanently discontinued by 20.4% in the clopidogrel group as compared with 18.2% in the placebo group (P<0.001). A total of 4.8% of patients in the clopidogrel group and 4.9% in the placebo group discontinued treatment because of an adverse event (P=0.67). Other than the treatment medications, concomitant medication use was similar in both groups. A median follow-up of 28 months revealed that the rates of primary efficacy events in the clopidogrel and placebo group were similar (6.8% versus 7.3%, P=0.22, respectively). The rate of primary safety events was 1.7% in the clopidogrel group and 1.3% in the placebo group, P=0.09.
Conclusions
This trial enrolled patients who either had established atherothrombotic disease or were at high risk for such disease and found that there was no significant benefit associated with the use of clopidogrel plus aspirin compared to aspirin alone in reducing myocardial infarction, stroke, or cardiovascular death. The risk of moderate or severe bleeding in symptomatic patients was higher in the clopidogrel plus aspirin group than in the aspirin plus placebo group. Overall, these findings do not support the use of dual antiplatelet therapy across this broad patient population.
D-Dimer in the Diagnosis of Pulmonary Embolism
Kearon C, Ginsberg JS, Douketis J, et al. An evaluation of D-dimer in the diagnosis of pulmonary embolism: a randomized trial. Ann Intern Med. 2006 Jun 6;144(11):812-821.
Background
The clinical usefulness of the D-dimer test in the diagnosis of pulmonary embolism (PE) has been previously studied. In patients with suspected PE, it may be safe to omit additional diagnostic testing if a patient has a negative D-dimer test; however, this approach has never been evaluated in a randomized, controlled trial.
The investigators in this trial studied two subgroups of patients with suspected PE and a negative D-dimer: patients with a low clinical probability of PE and those with a moderate or high clinical probability of PE who had a non-diagnostic ventilation perfusion scan (VQ scan) and no proximal deep vein thrombosis on venous ultrasonography. The hypothesis was that patients with a negative D-dimer who do not have further testing for PE won’t have a higher frequency of venous thromboembolism during follow-up than patients who undergo routine diagnostic testing.
Methods
Before any diagnostic testing, patients were assigned a probability score, using the Wells Criteria, to categorize the probability of PE as low or moderate to high.
Patients with low probability Wells scores: D-dimer testing was done on all patients with a low clinical probability of PE. Patients with a negative D-dimer were randomly assigned either to no additional diagnostic testing and no anticoagulation treatment or to additional diagnostic testing with an initial VQ scan. If the VQ scan was negative, then PE was excluded. If the VQ scan showed one or more segmental perfusion defects that were normally ventilated, then the scan was considered diagnostic for PE. If there were perfusion defects that did not meet the criteria for a high probability scan, then the scan was considered non-diagnostic. Patients with non-diagnostic scans underwent ultrasonography of the proximal veins of the legs. If deep vein thrombosis was present, PE was diagnosed. If ultrasonography was normal, the test was repeated after seven and 14 days. In all patients with a positive D-dimer, a VQ scan was performed.
Patients with moderate to high probability Wells scores: A VQ scan was performed on all patients with a moderate to high probability for PE. Patients with high probability scans were treated; patients with normal scans were not treated. Patients with non-diagnostic scans and normal venous ultrasonography were randomly assigned to receive either no additional testing or serial ultrasonography.
Outcomes: All patients were followed for six months for the development of venous thromboembolism after initial diagnostic testing.
Results
The study enrolled 1,126 patients. Overall, 160 patients (14.2%) had PE diagnosed at initial presentation or by venous ultrasonography. Of 952 patients who did not receive an initial diagnosis of PE, 11 (1.2%) had PE diagnosed at follow-up.
Patients with low probability Wells scores: Low clinical probability was present in 670 patients (60%). In patients with low clinical probability of PE, 373 (56%) had negative D-dimer tests and 297 (44%) had positive D-dimer tests. Of the 373 patients with low probability and negative D-dimer results, 187 were randomized to no additional testing and 186 received a VQ scan. The frequency of venous thromboembolism at six-month follow-up was similar in these two groups (-0.5% [CI, -3.0% to 1.6%]). Three patients with negative D-dimer tests were diagnosed with PE by VQ scan. Results were fairly complete (five patients without a six-month follow-up in the no additional testing group and one without a follow-up in the VQ scan group).
Twenty-four patients with low clinical probability and positive D-dimer results (n=297) were diagnosed with PE. Three patients did not complete the six-month follow-up. Of the remaining 294 patients, five patients had venous thromboembolism at six months.
Patients with moderate to high probability Wells scores: There were 456 patients (40%) had moderate or high clinical probability for PE. Each of these patients had a VQ scan. Non-diagnostic VQ scans and normal venous ultrasonography were performed on 226 patients. Of these 226 patients, 86 had a negative D-dimer and 140 had a positive D-dimer. Of the 86 patients with negative VQ scans, normal venous ultrasonography, and a negative D-dimer, 83 were randomly assigned to no additional testing or serial venous ultrasonography (42 and 41 respectively). At six months follow-up, one patient assigned to no additional testing had venous thromboembolism, and no patients in the additional testing group had venous thromboembolism.
Conclusions
The results of this trial suggest that it is safe to withhold additional diagnostic testing in patients with suspected PE, low pretest clinical probability, and a negative D-dimer test. Even in patients with moderate to high pretest clinical probability, a non-diagnostic VQ scan, and normal venous ultrasonography, only one patient with a negative D-dimer had a venous thromboembolic event at six months follow-up. The assay used for this study was an erythrocyte agglutination SimpliRED assay. Reported sensitivity is approximately 90%, and specificity is approximately 75%. To the readers, it should be noted that the authors defined a low probability Wells score as 4 rather than 1.5 or lower.
Efficacy and Safety of Inhaled Insulin Therapy in Adults with Diabetes Mellitus
Ceglia L, Lau J, Pittas AG. Meta-analysis: efficacy and safety of inhaled insulin therapy in adults with diabetes mellitus. Ann Intern Med. 2006 Nov 7;145(9):665-675.
Background
Despite its effectiveness in attaining glycemic control, there is considerable resistance to insulin use by patients and healthcare providers, primarily because of the need for subcutaneous injection. In January 2006, the U.S. Food and Drug Administration (FDA) approved the first formulation of inhaled insulin for clinical use in nonsmoking adults with type 1 or type 2 diabetes and no pulmonary disease. The authors of this paper present a systematic review to examine the efficacy, safety, and patient acceptability of inhaled insulin.
Methods
The authors conducted a search of MEDLINE to find English-language, randomized, controlled trials of inhaled insulin in nonpregnant adults with diabetes. To find unpublished studies, the authors reviewed the briefing document on Exubera powder for oral inhalation (Pfizer Inc., New York). An abstract was included if it reported original data from controlled trials in patients with type 1 or 2 diabetes and hemoglobin A1C outcomes for patients receiving inhaled insulin versus outcomes for a comparison group (subcutaneous insulin or oral hypoglycemics). Studies with less than 12 weeks duration were excluded because no comparison could be made regarding glycemic efficacy. For glycemic efficacy, the primary outcome was the treatment group difference in hemoglobin A1C from baseline. Secondary outcome was the proportion of patients with hemoglobin A1C levels less than 7%. To evaluate safety, the primary outcomes were severe hypoglycemia (glucose ≤36 mg/dL), cough, and treatment group difference in pulmonary function variables.
Results
Sixteen trials involving a total of 4,023 patients met inclusion criteria. Seven trials compared inhaled insulin with various subcutaneous insulin regimens in patients with type 1 diabetes. Nine trials compared inhaled insulin with subcutaneous insulin or oral hypoglycemic agents in patients with type 2 diabetes. Inhaled insulin was given with meals and titrated according to study-specific glucose goals. Subcutaneous insulin was titrated to the same specific goals. Doses of oral hypoglycemic agents were adjusted for glycemic targets in only two of the nine trials. The combined data from the studies demonstrated a small but statistically significant decrease in the levels of baseline hemoglobin A1C levels in favor of subcutaneous insulin (weighted mean difference 0.08%, [CI 0.03% to 0.14%]) in patients with type 1 or type 2 diabetes. The greatest advantage of subcutaneous insulin was noted in the study with the longest duration (104 weeks). There was no difference between the study groups in studies with duration of 24 weeks or less. Patients with type 1 or type 2 diabetes taking inhaled insulin were no more likely to achieve hemoglobin A1C levels less than 7% than those using subcutaneous insulin.
The combined data from studies comparing inhaled insulin to oral hypoglycemic agents in patients with type 2 diabetes showed that inhaled insulin lowered hemoglobin A1C levels more effectively (weighted mean difference -1.04%, [CI -1.59% to -0.49%]). In studies in which the oral hypoglycemic agents were titrated, inhaled insulin still lowered baseline hemoglobin A1C levels but to a lesser degree (weighted mean difference -0.20%, [CI - 0.34% to -0.07%]). Patients with type 2 diabetes taking inhaled insulin were more likely to achieve hemoglobin A1C levels less than 7% than those taking oral agents.
There was no difference in episodes of severe hypoglycemia in patients using inhaled insulin compared to those using subcutaneous insulin. A higher proportion of patients using inhaled insulin reported at least one episode of severe hypoglycemia compared to those using oral agents (risk ratio, 3.06 [CI 1.03 to 9.07]; 9.4% versus 3.5%, respectively).
With respect to pulmonary safety, all trials selected patients without histories of pulmonary problems and with at least six months of nonsmoking status. Pulmonary safety was assessed by self-reported symptoms and by pulmonary function tests. The most common pulmonary symptom associated with inhaled insulin was nonproductive cough. This symptom was reported more frequently compared to patients using subcutaneous insulin or oral agents (risk ratio, 3.52 [CI 2.23 to 5.56]; 16.9% versus 5.0%). Cough was noted early in the treatment course and diminished in frequency over time. Patients receiving inhaled insulin had a greater decrease in FEV1 (forced expiratory volume in the first second) from baseline than the comparator group (weighted mean difference, -0.031 L [CI-0.043 L to -0.020 L]). This decrease progressed slowly over the first six months but stabilized in studies of up to two years’ duration.
Only four trials reported data on overall patient satisfaction for inhaled insulin versus subcutaneous insulin. All trials reported a statistically significant increase in overall patient satisfaction with inhaled insulin over subcutaneous insulin. Patients randomly assigned to inhaled insulin were more likely to continue taking inhaled insulin than to switch back to subcutaneous insulin.
Conclusions
This meta-analysis showed that inhaled insulin is comparable to subcutaneous insulin in lowering hemoglobin A1C levels in patients with type 1 or type 2 diabetes. The proportion of patients reaching a target hemoglobin A1C of less than 7% was much lower in the studies in this meta-analysis as compared to levels in trials of intensive subcutaneous insulin therapy.
It’s more difficult to compare inhaled insulin with oral hypoglycemic agents because most studies involving oral agents used fixed dosing with different types of oral agents. There was a three-fold risk of severe hypoglycemia in patients using inhaled insulin compared to those using oral hypoglycemic agents. This is probably due to overall improved glycemic control in the inhaled insulin group. Cough was more common in the inhaled insulin groups, and there were small decreases in FEV1, but these did not progress over two years. The potential for pulmonary toxicity with long-term administration has not been evaluated and deserves further study.
From Weapon to Wonder Drug
Gas! Gas! Quick, boys! An ecstasy of fumbling, Fitting the clumsy helmets just in time; But someone still was yelling out and stumbling And flound’ring like a man in fire or lime.
—Wilfred Owen, Dulce et Decorum Est (1917)
In virtually every hospital around the world, oncology patients are treated with constantly evolving life- and limb-saving protocols. Cancer has plagued mankind for thousands of years. Records of cancer exist from as early as the ancient Egyptian era. The famous Edwin Smith Surgical Papyrus, dating to 1,600 B.C., describes a man with “bulging tumors on his breast.” The translation of the treatment for this case simply reads: “There is no treatment.” Unfortunately, the history of chemotherapy lags behind that of the disease it treats by at least 4,000 years. And ironically, the first modern day anticancer drug was spawned from a deadly World War I weapon.
Sulfur mustard, or mustard gas, has the dubious distinction of being one of the original chemical weapons. Bis-(2-chloroethyl) sulfide was first synthesized by London Institute of Physics Co-founder Frederick Guthrie in 1860. Guthrie, a profound believer in the advancement of science through experimentation rather than discussion, first documented the toxic effects of mustard gas by applying his mixture of ethylene and sulfur dichloride to his own skin, thus joining other notables in the halls of self-experimentation.
Mustard gas was mass-produced under the name LOST (an acronym fashioned from the names of its developers) for the German company Bayer AG during the first decade of the 20th century. Unfortunately, this proved to be perfect timing for the introduction of chemical warfare by the German army in 1917. The first strike was against Canadian troops. One year later the British used the same agent to destroy the “impregnable” Hindenburg Line. This culmination of the storied allied forces’ 100 Days Offensive ultimately led to the end of the First World War.
Various permutations of this compound were used sporadically by numerous world powers over the next 25 years. The malevolent use of mustard gas may be responsible for nearly 100,000 deaths and approximately five times as many wartime injuries. In fact, sulfur mustard gas—not a gas or a mustard, but rather a yellowish-brown vaporized liquid with a mustard seed-like odor—is known primarily as an incapacitant rather than as a lethal weapon. The effects of this poison may remain hidden for two to 24 hours after exposure.
Sulfur mustard is a vesicant. It exerts its effects on the body’s mucous membranes. The skin and the eyes are among the first organs to be affected. The toxicity of this agent is dose-dependent. While lower concentrations can cause symptoms as minor as skin irritation and conjunctivitis, higher titers can lead to morbid consequences such as necrotic ulcerations of the skin and blindness. At still higher concentrations, inhaled vapors can damage the mucous membrane lining of the respiratory tract, leading to hemorrhagic pulmonary edema.
Sulfur mustard also causes chronic sequelae. After exposure, surviving victims might exhibit nausea, vomiting, alopecia, and increased vulnerability to infection. These later symptoms are the result of the poison’s ability to act as an alkylating agent, cross-linking DNA and preventing the normal sequence of DNA replication. The organs primarily affected are the lining of the gastrointestinal tract and the bone marrow, due to their inherent high mitotic activity.
Despite its sinister history, mustard gas has played a key role in the development of anti-cancer chemotherapeutic agents and may justly be referred to as the egg from which medical oncology has hatched. The history of medicine contains many tales of accidental discovery, but how did a deadly gas become the first effective chemotherapeutic agent?
Fast forward 30 years to the Second World War. The interwar period resulted in numerous provisions—including the Geneva Protocol of 1929—to ban the use of chemical weapons. It was also a time during which many nations—both purposefully and accidentally—developed and stockpiled chemical agents. Thankfully, chemical weapons, including mustard gas, were not used extensively by either side during World War II. This was a period of intense—and justified—paranoia, however. General Dwight D. Eisenhower had made provisions for a stockpile of 100 tons of mustard gas on the S.S. John Harvey, which was stationed in Italy’s Bari Harbor. In December 1943, Nazi air strikes destroyed the John Harvey, among other ships, resulting in surprisingly few casualties despite the impressive amount of fire and destruction.
In the days and weeks following this catastrophe, however, survivors began to develop the familiar signs of mustard gas exposure. Lt. Col. Stewart Francis Alexander, an expert in chemical warfare, suspected exposure to the famous vapor. Autopsies of the victims reported profound lymphopenia as well as suppression of myeloid cell lines. This prompted the United States to lift the Office of Scientific Research and Development publication ban in 1946. At this time, numerous accounts were uncovered of mustard gas and its derivatives in experimental trials involving humans and cloned mice.
The first clinical trial, conducted by Louis Goodman and Alfred Gilman, involved the use of nitrogen mustard on a patient with advanced lymphosarcoma. After just four days of therapy, the patient’s tumor mass receded remarkably. Unfortunately, withdrawal of the medication due to profound neutropenia resulted in an abrupt return of the tumors. A second, shorter course resulted in a less profound reduction in tumor bulk, and a third try had no effect at all.
Interestingly, this temporary miracle with its subsequent failure illustrates two paradigms faced by medical oncologists today. Chemotherapeutic agents exert a toxic toll on the host, and tumor cells eventually develop resistance to circumvent the effects of these same medications. This is exactly why complicated protocols are used to treat patients with cancer.
Goodman and Gilman, authors of the famous medical pharmacology textbook The Pharmacological Basis of Therapeutics, used nitrogen mustard, a derivative of sulfur mustard, for their experiment. This compound, also known as mechlorethamine, is the first in-class alkylating agent, and is still used as a topical ointment in the treatment of cutaneous lymphomas. Other alkylating agents, such as cyclophosphamide, ifosfamide, and cisplatin, are used in cancer protocols worldwide.
We have not seen the last of mustard gas, which made its last acknowledged appearance in the Iran-Iraq war. Frequently, those things that are most destructive can be tamed for medical use.
The next time you have a patient receiving chemotherapy, think back to the roots of the cure. TH
References
- Pratt WB, Ruddon RW, Ensminger WD, et al. The Anticancer Drugs. 2nd ed. New York: Oxford University Press; 1994.
- Faguet GB. The War on Cancer: An Anatomy of Failure, a Blueprint for the Future. Dordrecht, Netherlands: Springer Press; 2005.
- Baguley BC, Kerr DJ, eds. Anticancer Drug Development. New York: Academic Press; 2002.
- Goodman LS, Wintrobe MM, Dameshek W, et al. Landmark article Sept. 21, 1946: Nitrogen mustard therapy. Use of methyl-bis(beta-chloroethyl)amine hydrochloride and tris(beta-chloroethyl)amine hydrochloride for Hodgkin’s disease, lymphosarcoma, leukemia and certain allied and miscellaneous disorders. JAMA. 1984;251:2255-2261.
- Kazin RA, Lowitt NR, Lowitt MH. Update in dermatology. Ann Intern Med. 2001;135:124-132.
Gas! Gas! Quick, boys! An ecstasy of fumbling, Fitting the clumsy helmets just in time; But someone still was yelling out and stumbling And flound’ring like a man in fire or lime.
—Wilfred Owen, Dulce et Decorum Est (1917)
In virtually every hospital around the world, oncology patients are treated with constantly evolving life- and limb-saving protocols. Cancer has plagued mankind for thousands of years. Records of cancer exist from as early as the ancient Egyptian era. The famous Edwin Smith Surgical Papyrus, dating to 1,600 B.C., describes a man with “bulging tumors on his breast.” The translation of the treatment for this case simply reads: “There is no treatment.” Unfortunately, the history of chemotherapy lags behind that of the disease it treats by at least 4,000 years. And ironically, the first modern day anticancer drug was spawned from a deadly World War I weapon.
Sulfur mustard, or mustard gas, has the dubious distinction of being one of the original chemical weapons. Bis-(2-chloroethyl) sulfide was first synthesized by London Institute of Physics Co-founder Frederick Guthrie in 1860. Guthrie, a profound believer in the advancement of science through experimentation rather than discussion, first documented the toxic effects of mustard gas by applying his mixture of ethylene and sulfur dichloride to his own skin, thus joining other notables in the halls of self-experimentation.
Mustard gas was mass-produced under the name LOST (an acronym fashioned from the names of its developers) for the German company Bayer AG during the first decade of the 20th century. Unfortunately, this proved to be perfect timing for the introduction of chemical warfare by the German army in 1917. The first strike was against Canadian troops. One year later the British used the same agent to destroy the “impregnable” Hindenburg Line. This culmination of the storied allied forces’ 100 Days Offensive ultimately led to the end of the First World War.
Various permutations of this compound were used sporadically by numerous world powers over the next 25 years. The malevolent use of mustard gas may be responsible for nearly 100,000 deaths and approximately five times as many wartime injuries. In fact, sulfur mustard gas—not a gas or a mustard, but rather a yellowish-brown vaporized liquid with a mustard seed-like odor—is known primarily as an incapacitant rather than as a lethal weapon. The effects of this poison may remain hidden for two to 24 hours after exposure.
Sulfur mustard is a vesicant. It exerts its effects on the body’s mucous membranes. The skin and the eyes are among the first organs to be affected. The toxicity of this agent is dose-dependent. While lower concentrations can cause symptoms as minor as skin irritation and conjunctivitis, higher titers can lead to morbid consequences such as necrotic ulcerations of the skin and blindness. At still higher concentrations, inhaled vapors can damage the mucous membrane lining of the respiratory tract, leading to hemorrhagic pulmonary edema.
Sulfur mustard also causes chronic sequelae. After exposure, surviving victims might exhibit nausea, vomiting, alopecia, and increased vulnerability to infection. These later symptoms are the result of the poison’s ability to act as an alkylating agent, cross-linking DNA and preventing the normal sequence of DNA replication. The organs primarily affected are the lining of the gastrointestinal tract and the bone marrow, due to their inherent high mitotic activity.
Despite its sinister history, mustard gas has played a key role in the development of anti-cancer chemotherapeutic agents and may justly be referred to as the egg from which medical oncology has hatched. The history of medicine contains many tales of accidental discovery, but how did a deadly gas become the first effective chemotherapeutic agent?
Fast forward 30 years to the Second World War. The interwar period resulted in numerous provisions—including the Geneva Protocol of 1929—to ban the use of chemical weapons. It was also a time during which many nations—both purposefully and accidentally—developed and stockpiled chemical agents. Thankfully, chemical weapons, including mustard gas, were not used extensively by either side during World War II. This was a period of intense—and justified—paranoia, however. General Dwight D. Eisenhower had made provisions for a stockpile of 100 tons of mustard gas on the S.S. John Harvey, which was stationed in Italy’s Bari Harbor. In December 1943, Nazi air strikes destroyed the John Harvey, among other ships, resulting in surprisingly few casualties despite the impressive amount of fire and destruction.
In the days and weeks following this catastrophe, however, survivors began to develop the familiar signs of mustard gas exposure. Lt. Col. Stewart Francis Alexander, an expert in chemical warfare, suspected exposure to the famous vapor. Autopsies of the victims reported profound lymphopenia as well as suppression of myeloid cell lines. This prompted the United States to lift the Office of Scientific Research and Development publication ban in 1946. At this time, numerous accounts were uncovered of mustard gas and its derivatives in experimental trials involving humans and cloned mice.
The first clinical trial, conducted by Louis Goodman and Alfred Gilman, involved the use of nitrogen mustard on a patient with advanced lymphosarcoma. After just four days of therapy, the patient’s tumor mass receded remarkably. Unfortunately, withdrawal of the medication due to profound neutropenia resulted in an abrupt return of the tumors. A second, shorter course resulted in a less profound reduction in tumor bulk, and a third try had no effect at all.
Interestingly, this temporary miracle with its subsequent failure illustrates two paradigms faced by medical oncologists today. Chemotherapeutic agents exert a toxic toll on the host, and tumor cells eventually develop resistance to circumvent the effects of these same medications. This is exactly why complicated protocols are used to treat patients with cancer.
Goodman and Gilman, authors of the famous medical pharmacology textbook The Pharmacological Basis of Therapeutics, used nitrogen mustard, a derivative of sulfur mustard, for their experiment. This compound, also known as mechlorethamine, is the first in-class alkylating agent, and is still used as a topical ointment in the treatment of cutaneous lymphomas. Other alkylating agents, such as cyclophosphamide, ifosfamide, and cisplatin, are used in cancer protocols worldwide.
We have not seen the last of mustard gas, which made its last acknowledged appearance in the Iran-Iraq war. Frequently, those things that are most destructive can be tamed for medical use.
The next time you have a patient receiving chemotherapy, think back to the roots of the cure. TH
References
- Pratt WB, Ruddon RW, Ensminger WD, et al. The Anticancer Drugs. 2nd ed. New York: Oxford University Press; 1994.
- Faguet GB. The War on Cancer: An Anatomy of Failure, a Blueprint for the Future. Dordrecht, Netherlands: Springer Press; 2005.
- Baguley BC, Kerr DJ, eds. Anticancer Drug Development. New York: Academic Press; 2002.
- Goodman LS, Wintrobe MM, Dameshek W, et al. Landmark article Sept. 21, 1946: Nitrogen mustard therapy. Use of methyl-bis(beta-chloroethyl)amine hydrochloride and tris(beta-chloroethyl)amine hydrochloride for Hodgkin’s disease, lymphosarcoma, leukemia and certain allied and miscellaneous disorders. JAMA. 1984;251:2255-2261.
- Kazin RA, Lowitt NR, Lowitt MH. Update in dermatology. Ann Intern Med. 2001;135:124-132.
Gas! Gas! Quick, boys! An ecstasy of fumbling, Fitting the clumsy helmets just in time; But someone still was yelling out and stumbling And flound’ring like a man in fire or lime.
—Wilfred Owen, Dulce et Decorum Est (1917)
In virtually every hospital around the world, oncology patients are treated with constantly evolving life- and limb-saving protocols. Cancer has plagued mankind for thousands of years. Records of cancer exist from as early as the ancient Egyptian era. The famous Edwin Smith Surgical Papyrus, dating to 1,600 B.C., describes a man with “bulging tumors on his breast.” The translation of the treatment for this case simply reads: “There is no treatment.” Unfortunately, the history of chemotherapy lags behind that of the disease it treats by at least 4,000 years. And ironically, the first modern day anticancer drug was spawned from a deadly World War I weapon.
Sulfur mustard, or mustard gas, has the dubious distinction of being one of the original chemical weapons. Bis-(2-chloroethyl) sulfide was first synthesized by London Institute of Physics Co-founder Frederick Guthrie in 1860. Guthrie, a profound believer in the advancement of science through experimentation rather than discussion, first documented the toxic effects of mustard gas by applying his mixture of ethylene and sulfur dichloride to his own skin, thus joining other notables in the halls of self-experimentation.
Mustard gas was mass-produced under the name LOST (an acronym fashioned from the names of its developers) for the German company Bayer AG during the first decade of the 20th century. Unfortunately, this proved to be perfect timing for the introduction of chemical warfare by the German army in 1917. The first strike was against Canadian troops. One year later the British used the same agent to destroy the “impregnable” Hindenburg Line. This culmination of the storied allied forces’ 100 Days Offensive ultimately led to the end of the First World War.
Various permutations of this compound were used sporadically by numerous world powers over the next 25 years. The malevolent use of mustard gas may be responsible for nearly 100,000 deaths and approximately five times as many wartime injuries. In fact, sulfur mustard gas—not a gas or a mustard, but rather a yellowish-brown vaporized liquid with a mustard seed-like odor—is known primarily as an incapacitant rather than as a lethal weapon. The effects of this poison may remain hidden for two to 24 hours after exposure.
Sulfur mustard is a vesicant. It exerts its effects on the body’s mucous membranes. The skin and the eyes are among the first organs to be affected. The toxicity of this agent is dose-dependent. While lower concentrations can cause symptoms as minor as skin irritation and conjunctivitis, higher titers can lead to morbid consequences such as necrotic ulcerations of the skin and blindness. At still higher concentrations, inhaled vapors can damage the mucous membrane lining of the respiratory tract, leading to hemorrhagic pulmonary edema.
Sulfur mustard also causes chronic sequelae. After exposure, surviving victims might exhibit nausea, vomiting, alopecia, and increased vulnerability to infection. These later symptoms are the result of the poison’s ability to act as an alkylating agent, cross-linking DNA and preventing the normal sequence of DNA replication. The organs primarily affected are the lining of the gastrointestinal tract and the bone marrow, due to their inherent high mitotic activity.
Despite its sinister history, mustard gas has played a key role in the development of anti-cancer chemotherapeutic agents and may justly be referred to as the egg from which medical oncology has hatched. The history of medicine contains many tales of accidental discovery, but how did a deadly gas become the first effective chemotherapeutic agent?
Fast forward 30 years to the Second World War. The interwar period resulted in numerous provisions—including the Geneva Protocol of 1929—to ban the use of chemical weapons. It was also a time during which many nations—both purposefully and accidentally—developed and stockpiled chemical agents. Thankfully, chemical weapons, including mustard gas, were not used extensively by either side during World War II. This was a period of intense—and justified—paranoia, however. General Dwight D. Eisenhower had made provisions for a stockpile of 100 tons of mustard gas on the S.S. John Harvey, which was stationed in Italy’s Bari Harbor. In December 1943, Nazi air strikes destroyed the John Harvey, among other ships, resulting in surprisingly few casualties despite the impressive amount of fire and destruction.
In the days and weeks following this catastrophe, however, survivors began to develop the familiar signs of mustard gas exposure. Lt. Col. Stewart Francis Alexander, an expert in chemical warfare, suspected exposure to the famous vapor. Autopsies of the victims reported profound lymphopenia as well as suppression of myeloid cell lines. This prompted the United States to lift the Office of Scientific Research and Development publication ban in 1946. At this time, numerous accounts were uncovered of mustard gas and its derivatives in experimental trials involving humans and cloned mice.
The first clinical trial, conducted by Louis Goodman and Alfred Gilman, involved the use of nitrogen mustard on a patient with advanced lymphosarcoma. After just four days of therapy, the patient’s tumor mass receded remarkably. Unfortunately, withdrawal of the medication due to profound neutropenia resulted in an abrupt return of the tumors. A second, shorter course resulted in a less profound reduction in tumor bulk, and a third try had no effect at all.
Interestingly, this temporary miracle with its subsequent failure illustrates two paradigms faced by medical oncologists today. Chemotherapeutic agents exert a toxic toll on the host, and tumor cells eventually develop resistance to circumvent the effects of these same medications. This is exactly why complicated protocols are used to treat patients with cancer.
Goodman and Gilman, authors of the famous medical pharmacology textbook The Pharmacological Basis of Therapeutics, used nitrogen mustard, a derivative of sulfur mustard, for their experiment. This compound, also known as mechlorethamine, is the first in-class alkylating agent, and is still used as a topical ointment in the treatment of cutaneous lymphomas. Other alkylating agents, such as cyclophosphamide, ifosfamide, and cisplatin, are used in cancer protocols worldwide.
We have not seen the last of mustard gas, which made its last acknowledged appearance in the Iran-Iraq war. Frequently, those things that are most destructive can be tamed for medical use.
The next time you have a patient receiving chemotherapy, think back to the roots of the cure. TH
References
- Pratt WB, Ruddon RW, Ensminger WD, et al. The Anticancer Drugs. 2nd ed. New York: Oxford University Press; 1994.
- Faguet GB. The War on Cancer: An Anatomy of Failure, a Blueprint for the Future. Dordrecht, Netherlands: Springer Press; 2005.
- Baguley BC, Kerr DJ, eds. Anticancer Drug Development. New York: Academic Press; 2002.
- Goodman LS, Wintrobe MM, Dameshek W, et al. Landmark article Sept. 21, 1946: Nitrogen mustard therapy. Use of methyl-bis(beta-chloroethyl)amine hydrochloride and tris(beta-chloroethyl)amine hydrochloride for Hodgkin’s disease, lymphosarcoma, leukemia and certain allied and miscellaneous disorders. JAMA. 1984;251:2255-2261.
- Kazin RA, Lowitt NR, Lowitt MH. Update in dermatology. Ann Intern Med. 2001;135:124-132.
Constructive Criticism
This is the first in a two-part series about how to provide constructive criticism to your hospitalist peers.
Part of improving your performance is learning from other hospitalists on a regular basis. You can do this through observation or discussion, and—when appropriate—by offering or receiving constructive criticism.
There are two types of physician-to-physician constructive criticism: When discussing perceived poor handling of a patient’s case, comments should take place within a formal peer review. Concerns about a physician’s non-clinical performance, such as communications problems or lack of availability, can be handled in a one-on-one conversation. Herein we’ll examine the peer review process; next month we’ll take a look at how and when to give constructive criticism to a peer informally.
Why Use Peer Review?
When a hospitalist notices a colleague’s clinical error or lack of judgment, it should be addressed in the program’s next peer review meeting, both for legal and procedural reasons.
“The key thing to understand is that ‘peer review’ offers certain protections for physicians and their colleagues,” explains Richard Rohr, MD, director, Hospitalist Service, Milford Hospital, Milford, Conn. “Ordinarily, if I discuss [another physician’s] case and render my opinion, then—in principle—if that patient were to file a lawsuit, they could subpoena me to testify about what I thought about their case. In the past, this had a chilling effect on peer review.”
Due to state laws passed years ago, peer review meetings now offer protection against subpoena. “Peer review meetings are protected,” says Dr. Rohr. “They can’t be used in court, and this makes it possible to have an organized peer review where you look at physicians’ work and provide an opinion about that work without fear of being drawn into a legal situation.”
The bottom line: “If you want to talk to another physician about their case, do so within [the peer review structure] so you’re legally protected,” says Dr. Rohr.
Focus on Improvement
When discussing a specific case or physician, remember that the reason for doing so is to improve quality of care. “Every practice should sit down, look at specific cases, and talk about possible areas of improvement,” says Dr. Rohr. “You need to take minutes of these meetings that are marked as confidential.”
The key to improvement is having an open discussion in each peer review meeting. “A good meeting is educational,” says Dr. Rohr. “The objective is to support each other and improve performance. A lot depends on the attitude that people bring to it. You have to not be afraid to say something; you must be willing to express opinions, or you’ll have a wasted meeting.”
Sometimes you may find that the problem goes beyond a single physician’s actions on a case. “If there is a problem with a case, find out whether it’s an aberration or if the problem needs to be addressed,” says Dr. Rohr. “Some things are not a physician’s fault, so much as [they are] signs that a medical system doesn’t work as effectively as it should or [that there is] a general lack of training. For example, an ER [emergency room] doctor misses a fracture. Was finding that fracture outside his competency? Does he need training reading X-rays, or can you manage to get radiologists in to check X-rays fast enough to become part of the process?”
Use a Set Structure
It’s up to the hospital medicine program director to set up a peer review process, which should be done within the structure established by the hospital. Peer review meetings “should be done on a regular basis,” advises Dr. Rohr. “How often depends on the volume of the program, but a typical group should meet monthly. You’ll probably look at three or four cases, which is a reasonable number to cover in one meeting. Look at unexpected mortalities or complications—you have a responsibility to the public to examine these.”
You might do best by bringing in an outside facilitator for the meetings. This creates an impartial atmosphere for discussions. “We bring in an external facilitator from a local teaching hospital,” says Dr. Rohr. “It’s good to have an educator lead the meeting; someone from academia will have a greater fund of knowledge and [a stronger] grasp of the medical literature, which helps bring the discussion to a more educational level. Everyone respects medical science.”
Note that the facilitator may need to be credentialed as a member of the medical staff in order for the proceedings to be protected from legal discovery.
“Peer review is difficult in smaller practices, because everyone knows everyone and they may be uncomfortable addressing problems,” explains Dr. Rohr. “Here, it’s especially helpful to have a leader from the outside who can render opinions and get everyone to chime in and render their own opinions.”
Remember that your peer review system is reportable. “As part of the hospital’s peer review structure, you’ll have to report findings from the meetings,” adds Dr. Rohr. “If someone is showing a pattern, these things have to be trended. Do they need training, or should they be dismissed?”
Giving Feedback through Peer Review
When you participate in a peer review discussion, don’t let your comments get too personal or subjective. “The most important thing is to keep it professional and make it educational to the greatest extent possible,” says Dr. Rohr. “Reference facts in the medical literature as often as possible. Point to something that’s been published to support your opinion. Base your comments on what’s known, and apply that to your analysis of the case.”
An evidence-based opinion doesn’t have to cite specific details; as long as you’re aware of major papers on the topic, you should have a grounded opinion.
Finally, as a physician participating in a peer review discussion, think before you speak. “Peer review works best when you have a basic respect for each other, as well as basic humility,” he says. TH
Jane Jerrard has written for The Hospitalist since 2005.
This is the first in a two-part series about how to provide constructive criticism to your hospitalist peers.
Part of improving your performance is learning from other hospitalists on a regular basis. You can do this through observation or discussion, and—when appropriate—by offering or receiving constructive criticism.
There are two types of physician-to-physician constructive criticism: When discussing perceived poor handling of a patient’s case, comments should take place within a formal peer review. Concerns about a physician’s non-clinical performance, such as communications problems or lack of availability, can be handled in a one-on-one conversation. Herein we’ll examine the peer review process; next month we’ll take a look at how and when to give constructive criticism to a peer informally.
Why Use Peer Review?
When a hospitalist notices a colleague’s clinical error or lack of judgment, it should be addressed in the program’s next peer review meeting, both for legal and procedural reasons.
“The key thing to understand is that ‘peer review’ offers certain protections for physicians and their colleagues,” explains Richard Rohr, MD, director, Hospitalist Service, Milford Hospital, Milford, Conn. “Ordinarily, if I discuss [another physician’s] case and render my opinion, then—in principle—if that patient were to file a lawsuit, they could subpoena me to testify about what I thought about their case. In the past, this had a chilling effect on peer review.”
Due to state laws passed years ago, peer review meetings now offer protection against subpoena. “Peer review meetings are protected,” says Dr. Rohr. “They can’t be used in court, and this makes it possible to have an organized peer review where you look at physicians’ work and provide an opinion about that work without fear of being drawn into a legal situation.”
The bottom line: “If you want to talk to another physician about their case, do so within [the peer review structure] so you’re legally protected,” says Dr. Rohr.
Focus on Improvement
When discussing a specific case or physician, remember that the reason for doing so is to improve quality of care. “Every practice should sit down, look at specific cases, and talk about possible areas of improvement,” says Dr. Rohr. “You need to take minutes of these meetings that are marked as confidential.”
The key to improvement is having an open discussion in each peer review meeting. “A good meeting is educational,” says Dr. Rohr. “The objective is to support each other and improve performance. A lot depends on the attitude that people bring to it. You have to not be afraid to say something; you must be willing to express opinions, or you’ll have a wasted meeting.”
Sometimes you may find that the problem goes beyond a single physician’s actions on a case. “If there is a problem with a case, find out whether it’s an aberration or if the problem needs to be addressed,” says Dr. Rohr. “Some things are not a physician’s fault, so much as [they are] signs that a medical system doesn’t work as effectively as it should or [that there is] a general lack of training. For example, an ER [emergency room] doctor misses a fracture. Was finding that fracture outside his competency? Does he need training reading X-rays, or can you manage to get radiologists in to check X-rays fast enough to become part of the process?”
Use a Set Structure
It’s up to the hospital medicine program director to set up a peer review process, which should be done within the structure established by the hospital. Peer review meetings “should be done on a regular basis,” advises Dr. Rohr. “How often depends on the volume of the program, but a typical group should meet monthly. You’ll probably look at three or four cases, which is a reasonable number to cover in one meeting. Look at unexpected mortalities or complications—you have a responsibility to the public to examine these.”
You might do best by bringing in an outside facilitator for the meetings. This creates an impartial atmosphere for discussions. “We bring in an external facilitator from a local teaching hospital,” says Dr. Rohr. “It’s good to have an educator lead the meeting; someone from academia will have a greater fund of knowledge and [a stronger] grasp of the medical literature, which helps bring the discussion to a more educational level. Everyone respects medical science.”
Note that the facilitator may need to be credentialed as a member of the medical staff in order for the proceedings to be protected from legal discovery.
“Peer review is difficult in smaller practices, because everyone knows everyone and they may be uncomfortable addressing problems,” explains Dr. Rohr. “Here, it’s especially helpful to have a leader from the outside who can render opinions and get everyone to chime in and render their own opinions.”
Remember that your peer review system is reportable. “As part of the hospital’s peer review structure, you’ll have to report findings from the meetings,” adds Dr. Rohr. “If someone is showing a pattern, these things have to be trended. Do they need training, or should they be dismissed?”
Giving Feedback through Peer Review
When you participate in a peer review discussion, don’t let your comments get too personal or subjective. “The most important thing is to keep it professional and make it educational to the greatest extent possible,” says Dr. Rohr. “Reference facts in the medical literature as often as possible. Point to something that’s been published to support your opinion. Base your comments on what’s known, and apply that to your analysis of the case.”
An evidence-based opinion doesn’t have to cite specific details; as long as you’re aware of major papers on the topic, you should have a grounded opinion.
Finally, as a physician participating in a peer review discussion, think before you speak. “Peer review works best when you have a basic respect for each other, as well as basic humility,” he says. TH
Jane Jerrard has written for The Hospitalist since 2005.
This is the first in a two-part series about how to provide constructive criticism to your hospitalist peers.
Part of improving your performance is learning from other hospitalists on a regular basis. You can do this through observation or discussion, and—when appropriate—by offering or receiving constructive criticism.
There are two types of physician-to-physician constructive criticism: When discussing perceived poor handling of a patient’s case, comments should take place within a formal peer review. Concerns about a physician’s non-clinical performance, such as communications problems or lack of availability, can be handled in a one-on-one conversation. Herein we’ll examine the peer review process; next month we’ll take a look at how and when to give constructive criticism to a peer informally.
Why Use Peer Review?
When a hospitalist notices a colleague’s clinical error or lack of judgment, it should be addressed in the program’s next peer review meeting, both for legal and procedural reasons.
“The key thing to understand is that ‘peer review’ offers certain protections for physicians and their colleagues,” explains Richard Rohr, MD, director, Hospitalist Service, Milford Hospital, Milford, Conn. “Ordinarily, if I discuss [another physician’s] case and render my opinion, then—in principle—if that patient were to file a lawsuit, they could subpoena me to testify about what I thought about their case. In the past, this had a chilling effect on peer review.”
Due to state laws passed years ago, peer review meetings now offer protection against subpoena. “Peer review meetings are protected,” says Dr. Rohr. “They can’t be used in court, and this makes it possible to have an organized peer review where you look at physicians’ work and provide an opinion about that work without fear of being drawn into a legal situation.”
The bottom line: “If you want to talk to another physician about their case, do so within [the peer review structure] so you’re legally protected,” says Dr. Rohr.
Focus on Improvement
When discussing a specific case or physician, remember that the reason for doing so is to improve quality of care. “Every practice should sit down, look at specific cases, and talk about possible areas of improvement,” says Dr. Rohr. “You need to take minutes of these meetings that are marked as confidential.”
The key to improvement is having an open discussion in each peer review meeting. “A good meeting is educational,” says Dr. Rohr. “The objective is to support each other and improve performance. A lot depends on the attitude that people bring to it. You have to not be afraid to say something; you must be willing to express opinions, or you’ll have a wasted meeting.”
Sometimes you may find that the problem goes beyond a single physician’s actions on a case. “If there is a problem with a case, find out whether it’s an aberration or if the problem needs to be addressed,” says Dr. Rohr. “Some things are not a physician’s fault, so much as [they are] signs that a medical system doesn’t work as effectively as it should or [that there is] a general lack of training. For example, an ER [emergency room] doctor misses a fracture. Was finding that fracture outside his competency? Does he need training reading X-rays, or can you manage to get radiologists in to check X-rays fast enough to become part of the process?”
Use a Set Structure
It’s up to the hospital medicine program director to set up a peer review process, which should be done within the structure established by the hospital. Peer review meetings “should be done on a regular basis,” advises Dr. Rohr. “How often depends on the volume of the program, but a typical group should meet monthly. You’ll probably look at three or four cases, which is a reasonable number to cover in one meeting. Look at unexpected mortalities or complications—you have a responsibility to the public to examine these.”
You might do best by bringing in an outside facilitator for the meetings. This creates an impartial atmosphere for discussions. “We bring in an external facilitator from a local teaching hospital,” says Dr. Rohr. “It’s good to have an educator lead the meeting; someone from academia will have a greater fund of knowledge and [a stronger] grasp of the medical literature, which helps bring the discussion to a more educational level. Everyone respects medical science.”
Note that the facilitator may need to be credentialed as a member of the medical staff in order for the proceedings to be protected from legal discovery.
“Peer review is difficult in smaller practices, because everyone knows everyone and they may be uncomfortable addressing problems,” explains Dr. Rohr. “Here, it’s especially helpful to have a leader from the outside who can render opinions and get everyone to chime in and render their own opinions.”
Remember that your peer review system is reportable. “As part of the hospital’s peer review structure, you’ll have to report findings from the meetings,” adds Dr. Rohr. “If someone is showing a pattern, these things have to be trended. Do they need training, or should they be dismissed?”
Giving Feedback through Peer Review
When you participate in a peer review discussion, don’t let your comments get too personal or subjective. “The most important thing is to keep it professional and make it educational to the greatest extent possible,” says Dr. Rohr. “Reference facts in the medical literature as often as possible. Point to something that’s been published to support your opinion. Base your comments on what’s known, and apply that to your analysis of the case.”
An evidence-based opinion doesn’t have to cite specific details; as long as you’re aware of major papers on the topic, you should have a grounded opinion.
Finally, as a physician participating in a peer review discussion, think before you speak. “Peer review works best when you have a basic respect for each other, as well as basic humility,” he says. TH
Jane Jerrard has written for The Hospitalist since 2005.
Fibromuscular Dysplasia
Brief history: 52-year-old female with uncontrolled hypertension.
Salient findings: The middle third of the arteries are involved with a “string of pearls” appearance of alternating webs and stenoses. This appearance is classic for fibromuscular dysplasia (FMD) (white arrow, above). The patient also has a 1.8-cm right renal artery aneurysm at the trifurcation of her first order renal artery branches (black arrow, above).
Patient population and natural history of disease: FMD is most common in young adult females, and its etiology is unknown. An association with alpha-1 antitrypsin deficiency has been reported in the literature. FMD is a leading cause of curable hypertension. Clinical manifestations of FMD include distal embolization of thrombus formed in small aneurysms, hypertension/ischemia due to obstruction by webs, and occlusion/infarct via spontaneous dissection. The natural prevalence of renal artery aneurysms is low—0.1% in all angiography patients—and its natural course is not well established. Renal artery aneurysms are most common in FMD, vasculitides, neoplasm, trauma, and Ehlers-Danlos Syndrome; they may be iatrogenic or idiopathic.
Management: Symptomatic medial fibroplasia-type FMD responds well to balloon angioplasty. Renal artery aneurysms may be managed medically or surgically, depending on risk factors. Indications for repair of renal artery aneurysms include a size of 2 cm or greater, pregnancy, expansion, renovascular hypertension, distal embolization, and rupture. Mortality from ruptured renal artery aneurysms is 10% in nonpregnant patients and 55% during pregnancy.
This patient had a good response to balloon angioplasty of the left renal artery. The right renal artery could not be angioplastied secondary to increased risk of aneurysm rupture with restoration of arterial blood flow due to increased pressure on the walls of the aneurysm. Hence, physicians surgically resected the right renal artery aneurysm and performed a bypass to the aorta.
Take Home Points
- FMD is most common in young or middle-age women;
- FMD is a type of curable hypertension, treated by renal artery angioplasty;
- FMD is diagnosed by an angiographic study—in classic cases, the involved artery has a string of pearls appearance; and
- FMD is associated with renal artery aneurysms. Consider surgical intervention in aneurysms greater than 2 cm. TH
Helena Summers is a radiology resident and Erik Summers is a hospitalist at the Mayo Clinic College of Medicine, Rochester, Minn.
Bibliography
- Kaufman JA, Lee MJ. Vascular and Interventional Radiology: The Requisites. Philadelphia: Mosby; 2004.
- Bisschops RH, Popma JJ, Meyerovitz MF. Treatment of fibromuscular dysplasia and renal artery aneurysm with use of a stent-graft. J Vasc Interv Radiol. 2001 Jun;12(6):757-760.
- Luscher TF, Lie JT, Stanson AW, et al. Arterial fibromuscular dysplasia. Mayo Clin Proc. 1987;62:931-952.
Brief history: 52-year-old female with uncontrolled hypertension.
Salient findings: The middle third of the arteries are involved with a “string of pearls” appearance of alternating webs and stenoses. This appearance is classic for fibromuscular dysplasia (FMD) (white arrow, above). The patient also has a 1.8-cm right renal artery aneurysm at the trifurcation of her first order renal artery branches (black arrow, above).
Patient population and natural history of disease: FMD is most common in young adult females, and its etiology is unknown. An association with alpha-1 antitrypsin deficiency has been reported in the literature. FMD is a leading cause of curable hypertension. Clinical manifestations of FMD include distal embolization of thrombus formed in small aneurysms, hypertension/ischemia due to obstruction by webs, and occlusion/infarct via spontaneous dissection. The natural prevalence of renal artery aneurysms is low—0.1% in all angiography patients—and its natural course is not well established. Renal artery aneurysms are most common in FMD, vasculitides, neoplasm, trauma, and Ehlers-Danlos Syndrome; they may be iatrogenic or idiopathic.
Management: Symptomatic medial fibroplasia-type FMD responds well to balloon angioplasty. Renal artery aneurysms may be managed medically or surgically, depending on risk factors. Indications for repair of renal artery aneurysms include a size of 2 cm or greater, pregnancy, expansion, renovascular hypertension, distal embolization, and rupture. Mortality from ruptured renal artery aneurysms is 10% in nonpregnant patients and 55% during pregnancy.
This patient had a good response to balloon angioplasty of the left renal artery. The right renal artery could not be angioplastied secondary to increased risk of aneurysm rupture with restoration of arterial blood flow due to increased pressure on the walls of the aneurysm. Hence, physicians surgically resected the right renal artery aneurysm and performed a bypass to the aorta.
Take Home Points
- FMD is most common in young or middle-age women;
- FMD is a type of curable hypertension, treated by renal artery angioplasty;
- FMD is diagnosed by an angiographic study—in classic cases, the involved artery has a string of pearls appearance; and
- FMD is associated with renal artery aneurysms. Consider surgical intervention in aneurysms greater than 2 cm. TH
Helena Summers is a radiology resident and Erik Summers is a hospitalist at the Mayo Clinic College of Medicine, Rochester, Minn.
Bibliography
- Kaufman JA, Lee MJ. Vascular and Interventional Radiology: The Requisites. Philadelphia: Mosby; 2004.
- Bisschops RH, Popma JJ, Meyerovitz MF. Treatment of fibromuscular dysplasia and renal artery aneurysm with use of a stent-graft. J Vasc Interv Radiol. 2001 Jun;12(6):757-760.
- Luscher TF, Lie JT, Stanson AW, et al. Arterial fibromuscular dysplasia. Mayo Clin Proc. 1987;62:931-952.
Brief history: 52-year-old female with uncontrolled hypertension.
Salient findings: The middle third of the arteries are involved with a “string of pearls” appearance of alternating webs and stenoses. This appearance is classic for fibromuscular dysplasia (FMD) (white arrow, above). The patient also has a 1.8-cm right renal artery aneurysm at the trifurcation of her first order renal artery branches (black arrow, above).
Patient population and natural history of disease: FMD is most common in young adult females, and its etiology is unknown. An association with alpha-1 antitrypsin deficiency has been reported in the literature. FMD is a leading cause of curable hypertension. Clinical manifestations of FMD include distal embolization of thrombus formed in small aneurysms, hypertension/ischemia due to obstruction by webs, and occlusion/infarct via spontaneous dissection. The natural prevalence of renal artery aneurysms is low—0.1% in all angiography patients—and its natural course is not well established. Renal artery aneurysms are most common in FMD, vasculitides, neoplasm, trauma, and Ehlers-Danlos Syndrome; they may be iatrogenic or idiopathic.
Management: Symptomatic medial fibroplasia-type FMD responds well to balloon angioplasty. Renal artery aneurysms may be managed medically or surgically, depending on risk factors. Indications for repair of renal artery aneurysms include a size of 2 cm or greater, pregnancy, expansion, renovascular hypertension, distal embolization, and rupture. Mortality from ruptured renal artery aneurysms is 10% in nonpregnant patients and 55% during pregnancy.
This patient had a good response to balloon angioplasty of the left renal artery. The right renal artery could not be angioplastied secondary to increased risk of aneurysm rupture with restoration of arterial blood flow due to increased pressure on the walls of the aneurysm. Hence, physicians surgically resected the right renal artery aneurysm and performed a bypass to the aorta.
Take Home Points
- FMD is most common in young or middle-age women;
- FMD is a type of curable hypertension, treated by renal artery angioplasty;
- FMD is diagnosed by an angiographic study—in classic cases, the involved artery has a string of pearls appearance; and
- FMD is associated with renal artery aneurysms. Consider surgical intervention in aneurysms greater than 2 cm. TH
Helena Summers is a radiology resident and Erik Summers is a hospitalist at the Mayo Clinic College of Medicine, Rochester, Minn.
Bibliography
- Kaufman JA, Lee MJ. Vascular and Interventional Radiology: The Requisites. Philadelphia: Mosby; 2004.
- Bisschops RH, Popma JJ, Meyerovitz MF. Treatment of fibromuscular dysplasia and renal artery aneurysm with use of a stent-graft. J Vasc Interv Radiol. 2001 Jun;12(6):757-760.
- Luscher TF, Lie JT, Stanson AW, et al. Arterial fibromuscular dysplasia. Mayo Clin Proc. 1987;62:931-952.
New Party in Power
Due to an overwhelming number of Democratic victories in last November’s midterm elections, the 110th Congress, which took office early this year, has new leaders and a new agenda that could bode well for healthcare legislation.
In this article, Laura Allendorf, SHM’s senior advisor for advocacy and government affairs, explains what the changes in Congress could mean for the near future of healthcare and for the legislation and issues that SHM strongly supports. Based in Washington, D.C., Allendorf is responsible for providing government relations services for SHM. She advises the organization on key legislative and regulatory healthcare issues before Congress and the Bush administration, and she works with SHM leaders and staff on policy development and advocacy strategies.
Majority Rules
The midterm elections brought about a shift in power that goes deeper than numbers of bodies on each side of the aisle. “The Democrats are now the majority in both chambers. This is significant, because they’ve been the minority since 1994, says Allendorf. “As the majority, they control the agenda now—on healthcare and other issues—and they also head the key committees.”
What can we expect to see from the Democratic Congress? “We should expect to see a more expansionist agenda” in general, according to Allendorf. “We’re going to see more activism in the area of healthcare, but whether anything gets done remains to be seen. There’s only a slim majority in the Senate, and President Bush can wield his veto pen. For example, the Democrats would like to give [the Department of] Health and Human Services the power to negotiate drug prices with pharmaceutical companies, specifically on Medicare Part D, but Bush won’t like that.”
Much depends on the issues at hand, as well as on how much bipartisan support exists for each specific bill.
Changing of the Guard
Anyone who glances at the newspaper knows that Democrat Nancy Pelosi (Calif.) is now the Speaker of the House. But Democratic leadership goes much deeper than that because the ruling party has also taken over leadership of Congressional committees. These committees shape the legislation introduced in the House and Senate.
As of press time, Congressional committee assignments had not been formally decided—at least not in the Senate—but many assignments were certain. “Typically, the highest-ranking Democrat [House or Senate] on a committee will become the new head, though Nancy Pelosi isn’t sticking to that,” explains Allendorf. “Pete Stark (D-Calif.) will likely chair the Ways and Means Committee’s Subcommittee on Health, and Charles Rangel (D-N.Y.) will head the House Ways and Means Committee. John Dingell (D-Mich.) will chair the House Energy and Commerce Committee.” (For more on committee chairs, visit http://media-newswire.com/release_1040623.html.)
For a complete list of committee members, visit SHM’s new Legislative Action Center at http://capwiz.com/hospitalmedicine/home/. See “New Advocacy Tool Available,” for more information on the Legislative Action Center, above.)
Starting Over on Key Issues
Many of the bills introduced in 2006—particularly spending bills—were not voted on by the end of the lame duck session last fall. That means that these bills must be reintroduced in the new year. Bills that recommend funding changes are frozen, so agencies continue to receive 2006 funding until the new Congress votes to change their budget.
“All bills have to be reintroduced in the 110th,” stresses Allendorf. “It will take some time—how much depends on the issue. The Democrats may want to hold hearings on legislation, or they may simply dust off legislation that was introduced last year.”
The Democrats are expected to move on many of the issues that SHM has been lobbying for. “They’ve said that they want to reform the healthcare system,” says Allendorf. “Top issues include providing coverage to the uninsured, reforming Medicare Part D, and resolving the physician payment issue.”
Allendorf believes that there will be a bipartisan effort to push through physician payment reform. “There are some 265 members of Congress who requested action on this issue this year [in 2006],” she points out. “There’s a genuine interest and desire to address physician payment reform and pay-for-performance as well. They may differ on how quickly they want to move on some of these.”
The news is not so good on the issue of gainsharing, where physicians are allowed to share the profits realized by a hospital’s cost reductions when linked to specific best practices. “Representative Nancy Johnson (R-Conn.) was a big proponent of this issue in the House, and she was not re-elected,” says Allendorf. “Stark is an opponent of gainsharing, so there may not be the same Congressional push behind it—at least in the House.”
However, the unexpected gainsharing demonstration projects approved in 2006 are underway, and Congress will hear reports on those in several years, once the projects have been analyzed.
Another issue that may not be addressed is liability. “Medical liability reform will be on the back burner,” warns Allendorf. “It’s generally not supported by the Democrats.”
In 2006, SHM supported increased funding for the Agency for Healthcare Research and Quality (AHRQ)—this was one of the major issues addressed by members during Legislative Advocacy Day during the Annual Meeting in Washington, D.C. Whether the next budget includes more money for the agency remains to be seen. “The Democrats support increased funding for NIH (National Institutes of Health), AHRQ, and other healthcare agencies,” says Allendorf. “There’s certainly political will, but where is the money going to come from?”
New Congress, New Issues
What about new issues? “Democrats have signaled that healthcare access for the uninsured will be a priority,” says Allendorf. “I think that we’ll see new legislation with a renewed emphasis on access to care.”
SHM’s Public Policy Committee will be waiting for the first legislation to be introduced regarding coverage for uninsured Americans. “This is an issue that SHM is strongly in favor of,” explains Allendorf. “SHM will look at any bills that come out on this issue and then form a policy.”
Regardless of which healthcare issues come to the forefront first, SHM’s Public Policy Committee, staff, and members are likely to be more active than ever. “I see a very busy year legislatively for SHM,” says Allendorf. TH
Jane Jerrard regularly writes “Public Policy” for The Hospitalist.
Due to an overwhelming number of Democratic victories in last November’s midterm elections, the 110th Congress, which took office early this year, has new leaders and a new agenda that could bode well for healthcare legislation.
In this article, Laura Allendorf, SHM’s senior advisor for advocacy and government affairs, explains what the changes in Congress could mean for the near future of healthcare and for the legislation and issues that SHM strongly supports. Based in Washington, D.C., Allendorf is responsible for providing government relations services for SHM. She advises the organization on key legislative and regulatory healthcare issues before Congress and the Bush administration, and she works with SHM leaders and staff on policy development and advocacy strategies.
Majority Rules
The midterm elections brought about a shift in power that goes deeper than numbers of bodies on each side of the aisle. “The Democrats are now the majority in both chambers. This is significant, because they’ve been the minority since 1994, says Allendorf. “As the majority, they control the agenda now—on healthcare and other issues—and they also head the key committees.”
What can we expect to see from the Democratic Congress? “We should expect to see a more expansionist agenda” in general, according to Allendorf. “We’re going to see more activism in the area of healthcare, but whether anything gets done remains to be seen. There’s only a slim majority in the Senate, and President Bush can wield his veto pen. For example, the Democrats would like to give [the Department of] Health and Human Services the power to negotiate drug prices with pharmaceutical companies, specifically on Medicare Part D, but Bush won’t like that.”
Much depends on the issues at hand, as well as on how much bipartisan support exists for each specific bill.
Changing of the Guard
Anyone who glances at the newspaper knows that Democrat Nancy Pelosi (Calif.) is now the Speaker of the House. But Democratic leadership goes much deeper than that because the ruling party has also taken over leadership of Congressional committees. These committees shape the legislation introduced in the House and Senate.
As of press time, Congressional committee assignments had not been formally decided—at least not in the Senate—but many assignments were certain. “Typically, the highest-ranking Democrat [House or Senate] on a committee will become the new head, though Nancy Pelosi isn’t sticking to that,” explains Allendorf. “Pete Stark (D-Calif.) will likely chair the Ways and Means Committee’s Subcommittee on Health, and Charles Rangel (D-N.Y.) will head the House Ways and Means Committee. John Dingell (D-Mich.) will chair the House Energy and Commerce Committee.” (For more on committee chairs, visit http://media-newswire.com/release_1040623.html.)
For a complete list of committee members, visit SHM’s new Legislative Action Center at http://capwiz.com/hospitalmedicine/home/. See “New Advocacy Tool Available,” for more information on the Legislative Action Center, above.)
Starting Over on Key Issues
Many of the bills introduced in 2006—particularly spending bills—were not voted on by the end of the lame duck session last fall. That means that these bills must be reintroduced in the new year. Bills that recommend funding changes are frozen, so agencies continue to receive 2006 funding until the new Congress votes to change their budget.
“All bills have to be reintroduced in the 110th,” stresses Allendorf. “It will take some time—how much depends on the issue. The Democrats may want to hold hearings on legislation, or they may simply dust off legislation that was introduced last year.”
The Democrats are expected to move on many of the issues that SHM has been lobbying for. “They’ve said that they want to reform the healthcare system,” says Allendorf. “Top issues include providing coverage to the uninsured, reforming Medicare Part D, and resolving the physician payment issue.”
Allendorf believes that there will be a bipartisan effort to push through physician payment reform. “There are some 265 members of Congress who requested action on this issue this year [in 2006],” she points out. “There’s a genuine interest and desire to address physician payment reform and pay-for-performance as well. They may differ on how quickly they want to move on some of these.”
The news is not so good on the issue of gainsharing, where physicians are allowed to share the profits realized by a hospital’s cost reductions when linked to specific best practices. “Representative Nancy Johnson (R-Conn.) was a big proponent of this issue in the House, and she was not re-elected,” says Allendorf. “Stark is an opponent of gainsharing, so there may not be the same Congressional push behind it—at least in the House.”
However, the unexpected gainsharing demonstration projects approved in 2006 are underway, and Congress will hear reports on those in several years, once the projects have been analyzed.
Another issue that may not be addressed is liability. “Medical liability reform will be on the back burner,” warns Allendorf. “It’s generally not supported by the Democrats.”
In 2006, SHM supported increased funding for the Agency for Healthcare Research and Quality (AHRQ)—this was one of the major issues addressed by members during Legislative Advocacy Day during the Annual Meeting in Washington, D.C. Whether the next budget includes more money for the agency remains to be seen. “The Democrats support increased funding for NIH (National Institutes of Health), AHRQ, and other healthcare agencies,” says Allendorf. “There’s certainly political will, but where is the money going to come from?”
New Congress, New Issues
What about new issues? “Democrats have signaled that healthcare access for the uninsured will be a priority,” says Allendorf. “I think that we’ll see new legislation with a renewed emphasis on access to care.”
SHM’s Public Policy Committee will be waiting for the first legislation to be introduced regarding coverage for uninsured Americans. “This is an issue that SHM is strongly in favor of,” explains Allendorf. “SHM will look at any bills that come out on this issue and then form a policy.”
Regardless of which healthcare issues come to the forefront first, SHM’s Public Policy Committee, staff, and members are likely to be more active than ever. “I see a very busy year legislatively for SHM,” says Allendorf. TH
Jane Jerrard regularly writes “Public Policy” for The Hospitalist.
Due to an overwhelming number of Democratic victories in last November’s midterm elections, the 110th Congress, which took office early this year, has new leaders and a new agenda that could bode well for healthcare legislation.
In this article, Laura Allendorf, SHM’s senior advisor for advocacy and government affairs, explains what the changes in Congress could mean for the near future of healthcare and for the legislation and issues that SHM strongly supports. Based in Washington, D.C., Allendorf is responsible for providing government relations services for SHM. She advises the organization on key legislative and regulatory healthcare issues before Congress and the Bush administration, and she works with SHM leaders and staff on policy development and advocacy strategies.
Majority Rules
The midterm elections brought about a shift in power that goes deeper than numbers of bodies on each side of the aisle. “The Democrats are now the majority in both chambers. This is significant, because they’ve been the minority since 1994, says Allendorf. “As the majority, they control the agenda now—on healthcare and other issues—and they also head the key committees.”
What can we expect to see from the Democratic Congress? “We should expect to see a more expansionist agenda” in general, according to Allendorf. “We’re going to see more activism in the area of healthcare, but whether anything gets done remains to be seen. There’s only a slim majority in the Senate, and President Bush can wield his veto pen. For example, the Democrats would like to give [the Department of] Health and Human Services the power to negotiate drug prices with pharmaceutical companies, specifically on Medicare Part D, but Bush won’t like that.”
Much depends on the issues at hand, as well as on how much bipartisan support exists for each specific bill.
Changing of the Guard
Anyone who glances at the newspaper knows that Democrat Nancy Pelosi (Calif.) is now the Speaker of the House. But Democratic leadership goes much deeper than that because the ruling party has also taken over leadership of Congressional committees. These committees shape the legislation introduced in the House and Senate.
As of press time, Congressional committee assignments had not been formally decided—at least not in the Senate—but many assignments were certain. “Typically, the highest-ranking Democrat [House or Senate] on a committee will become the new head, though Nancy Pelosi isn’t sticking to that,” explains Allendorf. “Pete Stark (D-Calif.) will likely chair the Ways and Means Committee’s Subcommittee on Health, and Charles Rangel (D-N.Y.) will head the House Ways and Means Committee. John Dingell (D-Mich.) will chair the House Energy and Commerce Committee.” (For more on committee chairs, visit http://media-newswire.com/release_1040623.html.)
For a complete list of committee members, visit SHM’s new Legislative Action Center at http://capwiz.com/hospitalmedicine/home/. See “New Advocacy Tool Available,” for more information on the Legislative Action Center, above.)
Starting Over on Key Issues
Many of the bills introduced in 2006—particularly spending bills—were not voted on by the end of the lame duck session last fall. That means that these bills must be reintroduced in the new year. Bills that recommend funding changes are frozen, so agencies continue to receive 2006 funding until the new Congress votes to change their budget.
“All bills have to be reintroduced in the 110th,” stresses Allendorf. “It will take some time—how much depends on the issue. The Democrats may want to hold hearings on legislation, or they may simply dust off legislation that was introduced last year.”
The Democrats are expected to move on many of the issues that SHM has been lobbying for. “They’ve said that they want to reform the healthcare system,” says Allendorf. “Top issues include providing coverage to the uninsured, reforming Medicare Part D, and resolving the physician payment issue.”
Allendorf believes that there will be a bipartisan effort to push through physician payment reform. “There are some 265 members of Congress who requested action on this issue this year [in 2006],” she points out. “There’s a genuine interest and desire to address physician payment reform and pay-for-performance as well. They may differ on how quickly they want to move on some of these.”
The news is not so good on the issue of gainsharing, where physicians are allowed to share the profits realized by a hospital’s cost reductions when linked to specific best practices. “Representative Nancy Johnson (R-Conn.) was a big proponent of this issue in the House, and she was not re-elected,” says Allendorf. “Stark is an opponent of gainsharing, so there may not be the same Congressional push behind it—at least in the House.”
However, the unexpected gainsharing demonstration projects approved in 2006 are underway, and Congress will hear reports on those in several years, once the projects have been analyzed.
Another issue that may not be addressed is liability. “Medical liability reform will be on the back burner,” warns Allendorf. “It’s generally not supported by the Democrats.”
In 2006, SHM supported increased funding for the Agency for Healthcare Research and Quality (AHRQ)—this was one of the major issues addressed by members during Legislative Advocacy Day during the Annual Meeting in Washington, D.C. Whether the next budget includes more money for the agency remains to be seen. “The Democrats support increased funding for NIH (National Institutes of Health), AHRQ, and other healthcare agencies,” says Allendorf. “There’s certainly political will, but where is the money going to come from?”
New Congress, New Issues
What about new issues? “Democrats have signaled that healthcare access for the uninsured will be a priority,” says Allendorf. “I think that we’ll see new legislation with a renewed emphasis on access to care.”
SHM’s Public Policy Committee will be waiting for the first legislation to be introduced regarding coverage for uninsured Americans. “This is an issue that SHM is strongly in favor of,” explains Allendorf. “SHM will look at any bills that come out on this issue and then form a policy.”
Regardless of which healthcare issues come to the forefront first, SHM’s Public Policy Committee, staff, and members are likely to be more active than ever. “I see a very busy year legislatively for SHM,” says Allendorf. TH
Jane Jerrard regularly writes “Public Policy” for The Hospitalist.
Hospital Medicine 2007
This year marks the 10-year anniversary of Robert Wachter’s coining of the term “hospitalist,” as well as the celebration of the decade-old SHM. The celebration culminates in a stellar annual meeting that epitomizes the growth of hospital medicine.
The SHM Annual Meeting attendance has grown from just a handful of participants to more than 1,200 expected at the 2007 Annual Meeting. More importantly, as the role of hospitalists has changed from primarily focusing on providing care to the hospitalized patient to serving as the leader of quality improvement, a key staff educator, and a facilitator of care transitions, so have their educational needs. Thus, the annual meeting has evolved in order to provide an educational experience that has relevance on a practical level.
The SHM 2007 Annual Meeting Committee (AMC), led by Course Director Chad Whelan, MD, challenged itself to develop a program that will meet the needs of a diverse audience that includes community-based and academic-based hospitalists; research-oriented and clinician-focused seasoned veteran physicians and early career hospitalists; as well as pediatric, geriatric, and family practice hospitalists. Such an effort begins with the big picture: What is happening in the environment that will impact healthcare today and in the future?
The answer is found in the role of information technology, and SHM will welcome two renowned speakers to provide current and future perspectives. David Brailer, MD, PhD, the first National Coordinator for Health Information Technology (2004-2006), will examine the forces driving health information technology and how technology affects pressures on the quality and cost of care. Jonathan Perlin, MD, PhD, chief medical officer and senior vice president for Quality for Hospital Corporation of America, will look to healthcare’s challenges and opportunities in the decade ahead, with a focus on health IT and performance and the role of hospital medicine as it relates to care improvement.
The 2007 Annual Meeting will again feature Robert Wachter, MD, as the keynote speaker in the closing plenary session. Dr. Wachter is sure to entertain as he examines “The Hospitalist Movement a Decade Later: Life as a Swiss Army Knife.”
The goal of a broad-based program will be achieved through separate breakout sessions and workshops, divided into seven tracks. Though the format is similar to other years, the 2007 program has some new twists. One clinical track focuses on “Things You Didn’t Learn in Medical School,” and a palliative care track has been added. Relevant sessions have been selected using the Core Competencies in Hospital Medicine and collaborating with SHM’s committees, which serve the interests of all of the groups.
The connection between the program and SHM’s committees will be strong. The committees will focus their efforts on topics and goals that are important to our members. The AMC solicits ideas for breakout sessions from the committees; it also profiles the output of a committee that has significance to attendees in their daily responsibilities.
Suggestions from the committees have resulted in a combination of pertinent and innovative sessions. Here are just a few to whet your appetite for the complete program:
- A workshop jointly planned by the Hospital Quality and Patient Safety (HQPS) and Education Committees, designed to obtain input and consensus on SHM-developed communication and hand-off standards;
- An Academic Track, intended to appeal to those hospitalists focused on teaching, quality improvement, research, and growing a hospital medicine program;
- A workshop, proposed by the Career Satisfaction Task Force, that addresses relevant career issues from the leaders’ and hospitalists’ perspectives;
- A Quality Track defined by the HQPS Committee that, in addition to the consensus-building workshop, features medication reconciliation and Toyota Methods sessions;
- A Pediatric Track, designed to address the needs of our fastest growing member segment, features a range of clinical and leadership topics, including electronic health records, the prevention of the transmission of infectious agents, and the utilization of dashboards to improve care;
- The work of the Benchmarks Committee will be profiled in a session that will demonstrate how to use key performance metrics to improve hospital medicine and the care of the hospitalized patient;
- The Public Policy Committee has recommended a pay-for-performance (P4P) breakout session, because P4P has been identified as an important issue in hospital medicine;
- A Palliative Care Track, proposed and developed by the Palliative Care Task Force, includes relevant topics such as pain management, the ethical and legal considerations of palliative care, and communications skills; and
- A visiting professor, Stephan Fihn, MD, MPH, will conduct poster rounds, lead a workshop, and participate in “Breakfast with Leaders in Hospital Medicine.”
The Annual Meeting Committee focused on identifying faculty members who can share their experience and expertise in an entertaining and elucidating way. We anticipate that you will enjoy presentations by experts new to the SHM Annual Meeting, as well as return engagements by past faculty.
The SHM Annual Meeting traditionally offers networking opportunities with more than 1,100 hospitalists. Special Interest Forums provide each attendee not only with a unique occasion to meet with hospitalists who share similar interests but also with the venue to express their opinions on a national level. SHM utilizes the input to change and grow the organization and hospital medicine. Planned Special Interest Forums include:
- Research;
- Community-based hospitalists;
- Pediatric hospitalists;
- Medical directors and leadership;
- Family practice hospitalists;
- Geriatric hospitalists;
- Nurse practitioners and physician assistants;
- Women in hospital medicine;
- Early career hospitalists;
- Education;
- Curriculum/fellowship;
- Public policy; and
- History of medicine.
Other networking mainstays of the Annual Meeting, including the Research, Innovations, and Clinical Vignettes Competition; Exhibits; President’s Lunch; and Town Meeting, will again be featured. Satellite symposia are planned, as well as the following pre-courses:
- Inpatient Coding and Documentation: Getting Paid What You Deserve;
- Best Practices in Managing a Hospital Medicine Program;
- Critical Care Medicine for the Hospitalist;
- Perioperative Medicine for the Hospitalist; and
- High Impact Quality Improvement: How to Ensure a Successful Project.
Additionally, a PICC Line Placement for Pediatric and Adult Hospitalists pre-course is being proposed by the Pediatrics Committee.
As you can see, 2007 Annual Meeting will present variety and choice; there is something for everyone. Don’t miss this premier educational event for hospitalists and the opportunity to be a part of the hospital medicine movement. The SHM 2007 Annual Meeting will be held at the Gaylord Texan Resort and Convention Center outside Dallas on May 24 and 25, with pre-courses held on May 23. Visit the SHM Web site at www.hospitalmedicine.org/hospitalmedicine2007 for complete program details or to register online. You may also register by calling SHM at (800) 843-3360. Come to Texas and help us celebrate 10 years of improving the quality of healthcare!
SHM: BEHIND THE SCENES
Resolutions
By Tina Budnitz, MPH
This is the time of year when many New Year’s resolutions dissolve. In some cases, people set unrealistic goals. Others lack the tools to succeed or live in an environment that makes change too difficult. In 2006 I resolved to start running each morning before work. I have the determination and the physical ability to meet this goal. Disarming the house alarm at 5 a.m., however, awakens my two toddlers. And detaching two toddlers from my legs proves to be quite a challenge. It also turns out that my tolerance for cold weather is low, while my ability to forgive a missed run is high.
Sustaining quality improvement initiatives at the hospital is lot like sustaining New Year’s resolutions. The best of intentions are often thwarted by a lack of time, resources, or energy to change the system, as well as by those within the system who resist change. For example, SHM members tell me that attempting to introduce a new discharge planning process feels a lot like trying to run through the hospital with two toddlers hanging on to your legs. SHM strives to support hospitalists in their resolutions to implement positive change in the hospital.
My role at SHM is to lead the development of programs, tool kits, and support mechanisms that will enable you, our members, to implement and sustain local quality improvement efforts.
SHM currently supports members in their efforts to improve outcomes for patients with heart failure, stroke, or diabetes; we work to prevent venous thromboembolism (VTE) and hospital-acquired blood-stream infections. We also support members in their efforts to improve the discharge process for older adults, thereby reducing readmission rates and adverse drug events while improving communications with receiving physicians.
We support members in their efforts to implement, evaluate, and sustain QI initiatives using a variety of methodologies. We offer symposia, workshops, and a full-day Annual Meeting Pre-Course on quality improvement; we also provide Leadership Academy Level I and Level II and networking opportunities for members and their mentors. We’ve designed Web-based clinical tool kits around specific disease states and special patient populations. We offer Web-based resource rooms to guide members from start to finish through a new local QI initiative. We have funded demonstration projects to pilot new approaches and tools for use in discharge planning, along with innovative research to improve care for heart failure patients. And we’ve only just begun.
Researching the Approach
For each QI area we address, an advisory board is recruited to represent the best available experts, organizations, and multidisciplinary professionals. For example, the Discharge Planning for Older Adults Advisory Board includes representatives from the American Geriatrics Society (AGS), the Institute for Healthcare Improvement (IHI), the Agency for Healthcare Research and Quality (AHRQ), the Society of General Internal Medicine (SGIM), the Case Management Society of America (CMSA), the National Quality Forum (NQF), and the Association for Health-System Pharmacists (AHSP), in addition to nationally renowned leaders in care transitions, geriatrics, nursing, patient literacy, and pharmacy.
Each Advisory Board conducts a needs assessment and a review of the literature to examine interventions and approaches. The board identifies existing “gaps” in clinical tools and guidelines. Finally, the Advisory Board determines specific, measurable targets for a hospitalist-led intervention and suggests the evidence-based approach(es) that should be most effective.
Implementing an Intervention: Where the Rubber Meets the Road
As I pointed out earlier, resolving what should be done and getting it done are two very different things. In most cases, few people debate the need for the intervention—reducing the incidence of inpatient VTEs, for example—or the validity of the proposed intervention. The challenge arises in changing the system of care so that the intervention becomes the new standard of care. Therefore, after the Advisory Board has determined the aims and intervention(s), SHM develops a “workbook,” a step-by-step field guide for hospitalists that walks them through the process of building a project team, establishing project aims and key metrics, obtaining institutional support, planning the intervention, launching the intervention, measuring impact, and sustaining system improvements.
The workbooks are posted in the SHM Resource Rooms along with other key resources, including slide sets, bedside teaching tools, patient education literature, CME modules, reviews of key literature, interactive “Ask the Expert” discussion boards, and improvement reports detailing the strategies, successes, and setbacks of other institutions. SHM currently offers resource rooms for Preventing VTE, Heart Failure, Stroke, Discharge Planning for the Elderly, Glycemic Control, and Antimicrobial Resistance. To access the resource rooms, visit our Web site at www.hospitalmedicine.org.
Training Leaders
SHM offers its members diverse training opportunities designed to accommodate the range of knowledge, expertise, and resources available at local institutions. The resource rooms and workbooks described above are made freely available to everyone. At the 2007 SHM Annual Meeting, we will offer a quality pre-course for hands-on training to lead a QI initiative for discharge planning, VTE prevention, or glycemic control.
For SHM members who desire additional support, SHM is developing “Mentored Implementation” and “On-Site Consulting” programs. The Mentored Implementation Program provides enrolled sites with all of the tools described above and an additional yearlong training program conducted via monthly conference calls with SHM mentors and/or one-day training programs. The On-Site Consulting Program surveys an institution’s resources and infrastructure. The SHM consultant team reviews the assessment with the site. Following this assessment, the consultant team visits the site to meet with hospital administrators, QI teams, and others to further assess the site and to help build internal support for the QI initiative. Following the visit, each site receives a customized report detailing recommendations and strategies to advance specific local QI initiatives.
SHM recently launched a Mentored Implementation and On-Site Consulting Program for VTE (known as the SHM VTE Collaborative). Similar programs are scheduled to launch for discharge planning this spring. In the coming years, we hope to add training programs in several other key patient safety areas.
Additionally, SHM is engaged in a strategic planning process to determine how best to attract the highest caliber medical students into hospital medicine and to mentor, train, and retain the next generation of leaders.
In summary, I hope the range of training opportunities and educational programs, the depth of SHM tool kits, and the scope of patient safety initiatives are meeting your needs to plan, implement, evaluate, and sustain positive change at your institution. If you are interested in learning more about SHM QI programs or have suggestions on how we might improve them to better meet your needs, please e-mail me at [email protected].
And, in case you were wondering, I’m making progress on my resolution to run. This year my resolution has the support of my department chairs (Jacob, five; Noah, two), with better-defined outcomes (run three times a week for more than 40 minutes each time), and a better tool kit (gloves, ear muffs). In the planning stage, I addressed my biggest barrier (leave the alarm off) and built a support team (my neighbor joins me).
If you see me at the annual meeting, please let me know how your resolutions are working to prevent DVT or to improve the discharge process, glycemic control, or heart failure care. Or better yet, submit those stories to me to share with members in our “Improvement Stories” or resource rooms Web site areas or in print via The Hospitalist or Journal of Hospital Medicine.
Budnitz is senior advisor, quality initiatives, for SHM.
This year marks the 10-year anniversary of Robert Wachter’s coining of the term “hospitalist,” as well as the celebration of the decade-old SHM. The celebration culminates in a stellar annual meeting that epitomizes the growth of hospital medicine.
The SHM Annual Meeting attendance has grown from just a handful of participants to more than 1,200 expected at the 2007 Annual Meeting. More importantly, as the role of hospitalists has changed from primarily focusing on providing care to the hospitalized patient to serving as the leader of quality improvement, a key staff educator, and a facilitator of care transitions, so have their educational needs. Thus, the annual meeting has evolved in order to provide an educational experience that has relevance on a practical level.
The SHM 2007 Annual Meeting Committee (AMC), led by Course Director Chad Whelan, MD, challenged itself to develop a program that will meet the needs of a diverse audience that includes community-based and academic-based hospitalists; research-oriented and clinician-focused seasoned veteran physicians and early career hospitalists; as well as pediatric, geriatric, and family practice hospitalists. Such an effort begins with the big picture: What is happening in the environment that will impact healthcare today and in the future?
The answer is found in the role of information technology, and SHM will welcome two renowned speakers to provide current and future perspectives. David Brailer, MD, PhD, the first National Coordinator for Health Information Technology (2004-2006), will examine the forces driving health information technology and how technology affects pressures on the quality and cost of care. Jonathan Perlin, MD, PhD, chief medical officer and senior vice president for Quality for Hospital Corporation of America, will look to healthcare’s challenges and opportunities in the decade ahead, with a focus on health IT and performance and the role of hospital medicine as it relates to care improvement.
The 2007 Annual Meeting will again feature Robert Wachter, MD, as the keynote speaker in the closing plenary session. Dr. Wachter is sure to entertain as he examines “The Hospitalist Movement a Decade Later: Life as a Swiss Army Knife.”
The goal of a broad-based program will be achieved through separate breakout sessions and workshops, divided into seven tracks. Though the format is similar to other years, the 2007 program has some new twists. One clinical track focuses on “Things You Didn’t Learn in Medical School,” and a palliative care track has been added. Relevant sessions have been selected using the Core Competencies in Hospital Medicine and collaborating with SHM’s committees, which serve the interests of all of the groups.
The connection between the program and SHM’s committees will be strong. The committees will focus their efforts on topics and goals that are important to our members. The AMC solicits ideas for breakout sessions from the committees; it also profiles the output of a committee that has significance to attendees in their daily responsibilities.
Suggestions from the committees have resulted in a combination of pertinent and innovative sessions. Here are just a few to whet your appetite for the complete program:
- A workshop jointly planned by the Hospital Quality and Patient Safety (HQPS) and Education Committees, designed to obtain input and consensus on SHM-developed communication and hand-off standards;
- An Academic Track, intended to appeal to those hospitalists focused on teaching, quality improvement, research, and growing a hospital medicine program;
- A workshop, proposed by the Career Satisfaction Task Force, that addresses relevant career issues from the leaders’ and hospitalists’ perspectives;
- A Quality Track defined by the HQPS Committee that, in addition to the consensus-building workshop, features medication reconciliation and Toyota Methods sessions;
- A Pediatric Track, designed to address the needs of our fastest growing member segment, features a range of clinical and leadership topics, including electronic health records, the prevention of the transmission of infectious agents, and the utilization of dashboards to improve care;
- The work of the Benchmarks Committee will be profiled in a session that will demonstrate how to use key performance metrics to improve hospital medicine and the care of the hospitalized patient;
- The Public Policy Committee has recommended a pay-for-performance (P4P) breakout session, because P4P has been identified as an important issue in hospital medicine;
- A Palliative Care Track, proposed and developed by the Palliative Care Task Force, includes relevant topics such as pain management, the ethical and legal considerations of palliative care, and communications skills; and
- A visiting professor, Stephan Fihn, MD, MPH, will conduct poster rounds, lead a workshop, and participate in “Breakfast with Leaders in Hospital Medicine.”
The Annual Meeting Committee focused on identifying faculty members who can share their experience and expertise in an entertaining and elucidating way. We anticipate that you will enjoy presentations by experts new to the SHM Annual Meeting, as well as return engagements by past faculty.
The SHM Annual Meeting traditionally offers networking opportunities with more than 1,100 hospitalists. Special Interest Forums provide each attendee not only with a unique occasion to meet with hospitalists who share similar interests but also with the venue to express their opinions on a national level. SHM utilizes the input to change and grow the organization and hospital medicine. Planned Special Interest Forums include:
- Research;
- Community-based hospitalists;
- Pediatric hospitalists;
- Medical directors and leadership;
- Family practice hospitalists;
- Geriatric hospitalists;
- Nurse practitioners and physician assistants;
- Women in hospital medicine;
- Early career hospitalists;
- Education;
- Curriculum/fellowship;
- Public policy; and
- History of medicine.
Other networking mainstays of the Annual Meeting, including the Research, Innovations, and Clinical Vignettes Competition; Exhibits; President’s Lunch; and Town Meeting, will again be featured. Satellite symposia are planned, as well as the following pre-courses:
- Inpatient Coding and Documentation: Getting Paid What You Deserve;
- Best Practices in Managing a Hospital Medicine Program;
- Critical Care Medicine for the Hospitalist;
- Perioperative Medicine for the Hospitalist; and
- High Impact Quality Improvement: How to Ensure a Successful Project.
Additionally, a PICC Line Placement for Pediatric and Adult Hospitalists pre-course is being proposed by the Pediatrics Committee.
As you can see, 2007 Annual Meeting will present variety and choice; there is something for everyone. Don’t miss this premier educational event for hospitalists and the opportunity to be a part of the hospital medicine movement. The SHM 2007 Annual Meeting will be held at the Gaylord Texan Resort and Convention Center outside Dallas on May 24 and 25, with pre-courses held on May 23. Visit the SHM Web site at www.hospitalmedicine.org/hospitalmedicine2007 for complete program details or to register online. You may also register by calling SHM at (800) 843-3360. Come to Texas and help us celebrate 10 years of improving the quality of healthcare!
SHM: BEHIND THE SCENES
Resolutions
By Tina Budnitz, MPH
This is the time of year when many New Year’s resolutions dissolve. In some cases, people set unrealistic goals. Others lack the tools to succeed or live in an environment that makes change too difficult. In 2006 I resolved to start running each morning before work. I have the determination and the physical ability to meet this goal. Disarming the house alarm at 5 a.m., however, awakens my two toddlers. And detaching two toddlers from my legs proves to be quite a challenge. It also turns out that my tolerance for cold weather is low, while my ability to forgive a missed run is high.
Sustaining quality improvement initiatives at the hospital is lot like sustaining New Year’s resolutions. The best of intentions are often thwarted by a lack of time, resources, or energy to change the system, as well as by those within the system who resist change. For example, SHM members tell me that attempting to introduce a new discharge planning process feels a lot like trying to run through the hospital with two toddlers hanging on to your legs. SHM strives to support hospitalists in their resolutions to implement positive change in the hospital.
My role at SHM is to lead the development of programs, tool kits, and support mechanisms that will enable you, our members, to implement and sustain local quality improvement efforts.
SHM currently supports members in their efforts to improve outcomes for patients with heart failure, stroke, or diabetes; we work to prevent venous thromboembolism (VTE) and hospital-acquired blood-stream infections. We also support members in their efforts to improve the discharge process for older adults, thereby reducing readmission rates and adverse drug events while improving communications with receiving physicians.
We support members in their efforts to implement, evaluate, and sustain QI initiatives using a variety of methodologies. We offer symposia, workshops, and a full-day Annual Meeting Pre-Course on quality improvement; we also provide Leadership Academy Level I and Level II and networking opportunities for members and their mentors. We’ve designed Web-based clinical tool kits around specific disease states and special patient populations. We offer Web-based resource rooms to guide members from start to finish through a new local QI initiative. We have funded demonstration projects to pilot new approaches and tools for use in discharge planning, along with innovative research to improve care for heart failure patients. And we’ve only just begun.
Researching the Approach
For each QI area we address, an advisory board is recruited to represent the best available experts, organizations, and multidisciplinary professionals. For example, the Discharge Planning for Older Adults Advisory Board includes representatives from the American Geriatrics Society (AGS), the Institute for Healthcare Improvement (IHI), the Agency for Healthcare Research and Quality (AHRQ), the Society of General Internal Medicine (SGIM), the Case Management Society of America (CMSA), the National Quality Forum (NQF), and the Association for Health-System Pharmacists (AHSP), in addition to nationally renowned leaders in care transitions, geriatrics, nursing, patient literacy, and pharmacy.
Each Advisory Board conducts a needs assessment and a review of the literature to examine interventions and approaches. The board identifies existing “gaps” in clinical tools and guidelines. Finally, the Advisory Board determines specific, measurable targets for a hospitalist-led intervention and suggests the evidence-based approach(es) that should be most effective.
Implementing an Intervention: Where the Rubber Meets the Road
As I pointed out earlier, resolving what should be done and getting it done are two very different things. In most cases, few people debate the need for the intervention—reducing the incidence of inpatient VTEs, for example—or the validity of the proposed intervention. The challenge arises in changing the system of care so that the intervention becomes the new standard of care. Therefore, after the Advisory Board has determined the aims and intervention(s), SHM develops a “workbook,” a step-by-step field guide for hospitalists that walks them through the process of building a project team, establishing project aims and key metrics, obtaining institutional support, planning the intervention, launching the intervention, measuring impact, and sustaining system improvements.
The workbooks are posted in the SHM Resource Rooms along with other key resources, including slide sets, bedside teaching tools, patient education literature, CME modules, reviews of key literature, interactive “Ask the Expert” discussion boards, and improvement reports detailing the strategies, successes, and setbacks of other institutions. SHM currently offers resource rooms for Preventing VTE, Heart Failure, Stroke, Discharge Planning for the Elderly, Glycemic Control, and Antimicrobial Resistance. To access the resource rooms, visit our Web site at www.hospitalmedicine.org.
Training Leaders
SHM offers its members diverse training opportunities designed to accommodate the range of knowledge, expertise, and resources available at local institutions. The resource rooms and workbooks described above are made freely available to everyone. At the 2007 SHM Annual Meeting, we will offer a quality pre-course for hands-on training to lead a QI initiative for discharge planning, VTE prevention, or glycemic control.
For SHM members who desire additional support, SHM is developing “Mentored Implementation” and “On-Site Consulting” programs. The Mentored Implementation Program provides enrolled sites with all of the tools described above and an additional yearlong training program conducted via monthly conference calls with SHM mentors and/or one-day training programs. The On-Site Consulting Program surveys an institution’s resources and infrastructure. The SHM consultant team reviews the assessment with the site. Following this assessment, the consultant team visits the site to meet with hospital administrators, QI teams, and others to further assess the site and to help build internal support for the QI initiative. Following the visit, each site receives a customized report detailing recommendations and strategies to advance specific local QI initiatives.
SHM recently launched a Mentored Implementation and On-Site Consulting Program for VTE (known as the SHM VTE Collaborative). Similar programs are scheduled to launch for discharge planning this spring. In the coming years, we hope to add training programs in several other key patient safety areas.
Additionally, SHM is engaged in a strategic planning process to determine how best to attract the highest caliber medical students into hospital medicine and to mentor, train, and retain the next generation of leaders.
In summary, I hope the range of training opportunities and educational programs, the depth of SHM tool kits, and the scope of patient safety initiatives are meeting your needs to plan, implement, evaluate, and sustain positive change at your institution. If you are interested in learning more about SHM QI programs or have suggestions on how we might improve them to better meet your needs, please e-mail me at [email protected].
And, in case you were wondering, I’m making progress on my resolution to run. This year my resolution has the support of my department chairs (Jacob, five; Noah, two), with better-defined outcomes (run three times a week for more than 40 minutes each time), and a better tool kit (gloves, ear muffs). In the planning stage, I addressed my biggest barrier (leave the alarm off) and built a support team (my neighbor joins me).
If you see me at the annual meeting, please let me know how your resolutions are working to prevent DVT or to improve the discharge process, glycemic control, or heart failure care. Or better yet, submit those stories to me to share with members in our “Improvement Stories” or resource rooms Web site areas or in print via The Hospitalist or Journal of Hospital Medicine.
Budnitz is senior advisor, quality initiatives, for SHM.
This year marks the 10-year anniversary of Robert Wachter’s coining of the term “hospitalist,” as well as the celebration of the decade-old SHM. The celebration culminates in a stellar annual meeting that epitomizes the growth of hospital medicine.
The SHM Annual Meeting attendance has grown from just a handful of participants to more than 1,200 expected at the 2007 Annual Meeting. More importantly, as the role of hospitalists has changed from primarily focusing on providing care to the hospitalized patient to serving as the leader of quality improvement, a key staff educator, and a facilitator of care transitions, so have their educational needs. Thus, the annual meeting has evolved in order to provide an educational experience that has relevance on a practical level.
The SHM 2007 Annual Meeting Committee (AMC), led by Course Director Chad Whelan, MD, challenged itself to develop a program that will meet the needs of a diverse audience that includes community-based and academic-based hospitalists; research-oriented and clinician-focused seasoned veteran physicians and early career hospitalists; as well as pediatric, geriatric, and family practice hospitalists. Such an effort begins with the big picture: What is happening in the environment that will impact healthcare today and in the future?
The answer is found in the role of information technology, and SHM will welcome two renowned speakers to provide current and future perspectives. David Brailer, MD, PhD, the first National Coordinator for Health Information Technology (2004-2006), will examine the forces driving health information technology and how technology affects pressures on the quality and cost of care. Jonathan Perlin, MD, PhD, chief medical officer and senior vice president for Quality for Hospital Corporation of America, will look to healthcare’s challenges and opportunities in the decade ahead, with a focus on health IT and performance and the role of hospital medicine as it relates to care improvement.
The 2007 Annual Meeting will again feature Robert Wachter, MD, as the keynote speaker in the closing plenary session. Dr. Wachter is sure to entertain as he examines “The Hospitalist Movement a Decade Later: Life as a Swiss Army Knife.”
The goal of a broad-based program will be achieved through separate breakout sessions and workshops, divided into seven tracks. Though the format is similar to other years, the 2007 program has some new twists. One clinical track focuses on “Things You Didn’t Learn in Medical School,” and a palliative care track has been added. Relevant sessions have been selected using the Core Competencies in Hospital Medicine and collaborating with SHM’s committees, which serve the interests of all of the groups.
The connection between the program and SHM’s committees will be strong. The committees will focus their efforts on topics and goals that are important to our members. The AMC solicits ideas for breakout sessions from the committees; it also profiles the output of a committee that has significance to attendees in their daily responsibilities.
Suggestions from the committees have resulted in a combination of pertinent and innovative sessions. Here are just a few to whet your appetite for the complete program:
- A workshop jointly planned by the Hospital Quality and Patient Safety (HQPS) and Education Committees, designed to obtain input and consensus on SHM-developed communication and hand-off standards;
- An Academic Track, intended to appeal to those hospitalists focused on teaching, quality improvement, research, and growing a hospital medicine program;
- A workshop, proposed by the Career Satisfaction Task Force, that addresses relevant career issues from the leaders’ and hospitalists’ perspectives;
- A Quality Track defined by the HQPS Committee that, in addition to the consensus-building workshop, features medication reconciliation and Toyota Methods sessions;
- A Pediatric Track, designed to address the needs of our fastest growing member segment, features a range of clinical and leadership topics, including electronic health records, the prevention of the transmission of infectious agents, and the utilization of dashboards to improve care;
- The work of the Benchmarks Committee will be profiled in a session that will demonstrate how to use key performance metrics to improve hospital medicine and the care of the hospitalized patient;
- The Public Policy Committee has recommended a pay-for-performance (P4P) breakout session, because P4P has been identified as an important issue in hospital medicine;
- A Palliative Care Track, proposed and developed by the Palliative Care Task Force, includes relevant topics such as pain management, the ethical and legal considerations of palliative care, and communications skills; and
- A visiting professor, Stephan Fihn, MD, MPH, will conduct poster rounds, lead a workshop, and participate in “Breakfast with Leaders in Hospital Medicine.”
The Annual Meeting Committee focused on identifying faculty members who can share their experience and expertise in an entertaining and elucidating way. We anticipate that you will enjoy presentations by experts new to the SHM Annual Meeting, as well as return engagements by past faculty.
The SHM Annual Meeting traditionally offers networking opportunities with more than 1,100 hospitalists. Special Interest Forums provide each attendee not only with a unique occasion to meet with hospitalists who share similar interests but also with the venue to express their opinions on a national level. SHM utilizes the input to change and grow the organization and hospital medicine. Planned Special Interest Forums include:
- Research;
- Community-based hospitalists;
- Pediatric hospitalists;
- Medical directors and leadership;
- Family practice hospitalists;
- Geriatric hospitalists;
- Nurse practitioners and physician assistants;
- Women in hospital medicine;
- Early career hospitalists;
- Education;
- Curriculum/fellowship;
- Public policy; and
- History of medicine.
Other networking mainstays of the Annual Meeting, including the Research, Innovations, and Clinical Vignettes Competition; Exhibits; President’s Lunch; and Town Meeting, will again be featured. Satellite symposia are planned, as well as the following pre-courses:
- Inpatient Coding and Documentation: Getting Paid What You Deserve;
- Best Practices in Managing a Hospital Medicine Program;
- Critical Care Medicine for the Hospitalist;
- Perioperative Medicine for the Hospitalist; and
- High Impact Quality Improvement: How to Ensure a Successful Project.
Additionally, a PICC Line Placement for Pediatric and Adult Hospitalists pre-course is being proposed by the Pediatrics Committee.
As you can see, 2007 Annual Meeting will present variety and choice; there is something for everyone. Don’t miss this premier educational event for hospitalists and the opportunity to be a part of the hospital medicine movement. The SHM 2007 Annual Meeting will be held at the Gaylord Texan Resort and Convention Center outside Dallas on May 24 and 25, with pre-courses held on May 23. Visit the SHM Web site at www.hospitalmedicine.org/hospitalmedicine2007 for complete program details or to register online. You may also register by calling SHM at (800) 843-3360. Come to Texas and help us celebrate 10 years of improving the quality of healthcare!
SHM: BEHIND THE SCENES
Resolutions
By Tina Budnitz, MPH
This is the time of year when many New Year’s resolutions dissolve. In some cases, people set unrealistic goals. Others lack the tools to succeed or live in an environment that makes change too difficult. In 2006 I resolved to start running each morning before work. I have the determination and the physical ability to meet this goal. Disarming the house alarm at 5 a.m., however, awakens my two toddlers. And detaching two toddlers from my legs proves to be quite a challenge. It also turns out that my tolerance for cold weather is low, while my ability to forgive a missed run is high.
Sustaining quality improvement initiatives at the hospital is lot like sustaining New Year’s resolutions. The best of intentions are often thwarted by a lack of time, resources, or energy to change the system, as well as by those within the system who resist change. For example, SHM members tell me that attempting to introduce a new discharge planning process feels a lot like trying to run through the hospital with two toddlers hanging on to your legs. SHM strives to support hospitalists in their resolutions to implement positive change in the hospital.
My role at SHM is to lead the development of programs, tool kits, and support mechanisms that will enable you, our members, to implement and sustain local quality improvement efforts.
SHM currently supports members in their efforts to improve outcomes for patients with heart failure, stroke, or diabetes; we work to prevent venous thromboembolism (VTE) and hospital-acquired blood-stream infections. We also support members in their efforts to improve the discharge process for older adults, thereby reducing readmission rates and adverse drug events while improving communications with receiving physicians.
We support members in their efforts to implement, evaluate, and sustain QI initiatives using a variety of methodologies. We offer symposia, workshops, and a full-day Annual Meeting Pre-Course on quality improvement; we also provide Leadership Academy Level I and Level II and networking opportunities for members and their mentors. We’ve designed Web-based clinical tool kits around specific disease states and special patient populations. We offer Web-based resource rooms to guide members from start to finish through a new local QI initiative. We have funded demonstration projects to pilot new approaches and tools for use in discharge planning, along with innovative research to improve care for heart failure patients. And we’ve only just begun.
Researching the Approach
For each QI area we address, an advisory board is recruited to represent the best available experts, organizations, and multidisciplinary professionals. For example, the Discharge Planning for Older Adults Advisory Board includes representatives from the American Geriatrics Society (AGS), the Institute for Healthcare Improvement (IHI), the Agency for Healthcare Research and Quality (AHRQ), the Society of General Internal Medicine (SGIM), the Case Management Society of America (CMSA), the National Quality Forum (NQF), and the Association for Health-System Pharmacists (AHSP), in addition to nationally renowned leaders in care transitions, geriatrics, nursing, patient literacy, and pharmacy.
Each Advisory Board conducts a needs assessment and a review of the literature to examine interventions and approaches. The board identifies existing “gaps” in clinical tools and guidelines. Finally, the Advisory Board determines specific, measurable targets for a hospitalist-led intervention and suggests the evidence-based approach(es) that should be most effective.
Implementing an Intervention: Where the Rubber Meets the Road
As I pointed out earlier, resolving what should be done and getting it done are two very different things. In most cases, few people debate the need for the intervention—reducing the incidence of inpatient VTEs, for example—or the validity of the proposed intervention. The challenge arises in changing the system of care so that the intervention becomes the new standard of care. Therefore, after the Advisory Board has determined the aims and intervention(s), SHM develops a “workbook,” a step-by-step field guide for hospitalists that walks them through the process of building a project team, establishing project aims and key metrics, obtaining institutional support, planning the intervention, launching the intervention, measuring impact, and sustaining system improvements.
The workbooks are posted in the SHM Resource Rooms along with other key resources, including slide sets, bedside teaching tools, patient education literature, CME modules, reviews of key literature, interactive “Ask the Expert” discussion boards, and improvement reports detailing the strategies, successes, and setbacks of other institutions. SHM currently offers resource rooms for Preventing VTE, Heart Failure, Stroke, Discharge Planning for the Elderly, Glycemic Control, and Antimicrobial Resistance. To access the resource rooms, visit our Web site at www.hospitalmedicine.org.
Training Leaders
SHM offers its members diverse training opportunities designed to accommodate the range of knowledge, expertise, and resources available at local institutions. The resource rooms and workbooks described above are made freely available to everyone. At the 2007 SHM Annual Meeting, we will offer a quality pre-course for hands-on training to lead a QI initiative for discharge planning, VTE prevention, or glycemic control.
For SHM members who desire additional support, SHM is developing “Mentored Implementation” and “On-Site Consulting” programs. The Mentored Implementation Program provides enrolled sites with all of the tools described above and an additional yearlong training program conducted via monthly conference calls with SHM mentors and/or one-day training programs. The On-Site Consulting Program surveys an institution’s resources and infrastructure. The SHM consultant team reviews the assessment with the site. Following this assessment, the consultant team visits the site to meet with hospital administrators, QI teams, and others to further assess the site and to help build internal support for the QI initiative. Following the visit, each site receives a customized report detailing recommendations and strategies to advance specific local QI initiatives.
SHM recently launched a Mentored Implementation and On-Site Consulting Program for VTE (known as the SHM VTE Collaborative). Similar programs are scheduled to launch for discharge planning this spring. In the coming years, we hope to add training programs in several other key patient safety areas.
Additionally, SHM is engaged in a strategic planning process to determine how best to attract the highest caliber medical students into hospital medicine and to mentor, train, and retain the next generation of leaders.
In summary, I hope the range of training opportunities and educational programs, the depth of SHM tool kits, and the scope of patient safety initiatives are meeting your needs to plan, implement, evaluate, and sustain positive change at your institution. If you are interested in learning more about SHM QI programs or have suggestions on how we might improve them to better meet your needs, please e-mail me at [email protected].
And, in case you were wondering, I’m making progress on my resolution to run. This year my resolution has the support of my department chairs (Jacob, five; Noah, two), with better-defined outcomes (run three times a week for more than 40 minutes each time), and a better tool kit (gloves, ear muffs). In the planning stage, I addressed my biggest barrier (leave the alarm off) and built a support team (my neighbor joins me).
If you see me at the annual meeting, please let me know how your resolutions are working to prevent DVT or to improve the discharge process, glycemic control, or heart failure care. Or better yet, submit those stories to me to share with members in our “Improvement Stories” or resource rooms Web site areas or in print via The Hospitalist or Journal of Hospital Medicine.
Budnitz is senior advisor, quality initiatives, for SHM.
Lessons learned from failing to follow up
As pressures upon us increase, we have begun to build teams and systems that ensure the kind of care our patients demand. We may not be able to deliver personal continuity to every patient, but a team can approximate that continuity—after it meets several key challenges. Foremost: developing systems of communication that are consistent, reliable, accurate, and accessible to any member of the team.
There is another locus in the continuum of care that is often neglected: post-event follow-up for medical, psychosocial, or legal purposes. The following case illustrates this point.
CASE A lawsuit is filed despite comprehensive care
C.S., a 34-year-old G4P3, at 35 weeks’ gestation with suspected premature rupture of membranes, was referred to the tertiary-care center where I practice. She had reported a gush of fluid, and the referring physician had observed nitrazine-positive fluid at the introitus.
On initial speculum examination at the tertiary-care center, no fluid was observed coming from the cervix, and vaginal secretions were nitrazine-negative. The cervix was long, posterior, and patulous. Ultrasonographic examination of the uterus demonstrated a normal fetus of appropriate size for the reported gestational age and a maximum pocket of amniotic fluid greater than 10 cm in depth. The fetus was active, and the nonstress test was reactive. The patient’s urine was alkaline; a specimen sent for culture was found to be negative.
Despite the reassuring clinical assessment, this mature multipara’s description of events was credible. She was offered the option of overnight observation or amniocentesis with instillation of indigo carmine. Because her husband and 3 children would have had to stay in their car overnight if she remained in the hospital, the patient chose instillation.
Amniocentesis was performed under sonographic guidance, with a return of clear fluid. The fluid was sent to the lab for fetal lung maturity testing, which was negative. Ten cubic centimeters of indigo carmine dye were instilled, and a tampon was inserted. After 2 hours of ambulation, there was no dye on the tampon, and another nonstress test was reactive. The patient was discharged.
The next day, the patient reported to her family physician complaining of severe uterine pain, fever, and a loss of fetal movement. When fetal heart activity could not be detected by Doppler ultrasound, she was again referred to the tertiary-care center. There she was noted to be in extreme pain, with a temperature of 104°F, and bulging forewaters. There was copious fluid and no fetal heart motion on ultrasonography.
Amniotomy elicited a gush of clear, blue, odorless fluid. The cervix dilated completely, and the fetal head was expelled to the chin. Examination revealed a nuchal and shoulder cord tightly wrapped and tethering descent. The vaginal wall was retracted, and the cord was visualized and divided. A stillborn male was immediately expelled, and the placenta followed rapidly. Bleeding remained within normal limits.
Although there was no explanation for the fever, the patient was treated with antibiotics during her postpartum hospital stay. She recovered quickly. Cultures from mother, baby, and placenta detected no organisms. The patient was discharged on day 4.
Ten months later, the patient filed a lawsuit alleging a failure to diagnose amnionitis at the time of the first visit.
What prompted the lawsuit?
Clearly, this patient had a tragic loss. Just as clearly, there were multiple incongruities between her clinical presentation and the outcome. The patient and the care team were both aware of these truths.
Research has demonstrated that physicians who interact in a positive manner with their patients are less likely to be sued than those who fail to communicate warmth and caring. Patients of physicians who have a history of multiple lawsuits may consider them knowledgeable and competent—but they also are likely to describe them as unavailable, abrupt, and disinterested. Patients often characterize their experiences with such physicians as negative even when the clinical outcome is good. This negativity often prevails even when the office staff is skilled in communication, education, and support.1,2
The team performed well
In the case of C.S., the physicians, by nature and by intent, were attentive to the human needs of this grieving family. Here is what we did well:
- The same residents provided care through both labor and delivery and during postpartum care
- The attending physician (me) was present through all clinical milestones
- All members of the team openly expressed their sorrow to the family
- I visited with the patient daily—providing honest answers to the family’s questions and acknowledging gaps in the medical team’s understanding of what had happened
- A follow-up plan was established to provide autopsy results to the family
So what went wrong? Why did the family—and the members of the care team—have to suffer the ordeal of litigation?
Critical lapse uncovered
Note the last bulleted item, above. This was the critical lapse: I did not call the family to relay the results of the autopsy. Why not? I knew better, after all, and prided myself on my commitment to all dimensions of the care I provided. As with most lapses in medical care, failure was multifactorial—part system design, part human failing.
At the tertiary-care institution in question, maternal transport high-risk pregnancies are managed with a group of attending obstetricians on a week-by-week rotational schedule. This provides continuity of care through the calendar week but, by its very nature, relieves the attending of the previous week from clinical responsibilities. By happenstance, the monthly rotation of residents coincided with this patient’s hospitalization. Thus, no member of her care team had continuing direct responsibility for the OB service.
To complicate matters, I left town after the delivery for a conference, with vacation tacked on afterwards. When I returned to the office 10 days later, my head was refreshed but my memory had been purged, and I failed to follow through on my promise to contact the family.
About 3 months after the delivery, I overheard a secretary trying to calm a frustrated patient on the telephone. When I heard the secretary say, “I’m sorry, ma’am, but we just don’t have the autopsy results, maybe you should call pathology,” I realized which patient it was and took over the conversation. Abjectly apologetic, I promised to get the information for her within hours. The patient was reserved but accepted my offer.
The autopsy revealed a polymorphonuclear leukocyte infiltration throughout the body, but no organisms could be identified by culture or on histologic examination. The final pathologist’s report provided no definitive explanation of the sequence of events that led to fetal death. When I explained this to the patient during a telephone call, her demeanor turned icy and she hung up. Several months later, the lawsuit was served.
1. Build a solid foundation. We all know communication is important, but many of us fail to take the extra steps necessary to standardize communication so that the entire care team is apprised of the goals for a given patient—as well as exactly how much progress has been made toward those goals. Various systems have been designed to accomplish this aim, many of them derived from the aviation industry. A small investment in time can reap big rewards. A few examples:
- “Time-out”—A pause before an invasive procedure to confirm that you have the correct patient and will be performing the appropriate procedure.
- “Snapshot”—An overview of cases within a defined time period, including identification of the team’s priorities. For example: “This morning we have 3 patients scheduled for surgery, beginning with Mrs. ‘A,’ whose hysterectomy for a large myomatous uterus will likely be time-consuming.”
- “Turn-over”—A synopsis of cases at the time they are handed over to another team member or a different team. The information provided should include outstanding tasks and tests.
- “De-brief”—Time set aside after a case to discuss what happened, what could have been handled differently, and what the next steps are. These sessions provide immediate feedback to the team and influence the care of future patients.
2. Don’t leave warmth and caring to your staff. The evidence is in: Physicians who interact in a positive manner with their patients are less likely to be sued than those who fail to communicate warmth and concern. Given the competing demands on our time, it is all too easy to rush through patient visits or other aspects of care without attending to the human component. Take a few minutes to greet each patient by name, inquire about her family and any concerns she may have about her condition, and listen attentively to her response. Then document any important details that arise during this discussion, so the rest of your team knows about them, too.
3. Offer and follow through on an evidence-based explanation of events. At times of tragedy, pay attention to the needs of grieving patients—and their families. This begins with an acknowledgment of the shock and sorrow they are experiencing and includes reassurance that the reasons for the adverse event will be explored and reported. This should not be an empty promise. It is important that the physician offers as full an explanation of an event as possible—as soon as all the facts are in—and that this explanation is voluntary, not something the patient has to ask for repeatedly.
4. Implement an effective tracking system. The case of C.S. illustrates the need for a more comprehensive tracking system. In that case, my failure to relay the need for autopsy results to other team members, and my subsequent absence from the scene, allowed a critical detail to slip through the cracks.
Because patient files tend to be forgotten once they are stored away, a tickle file or similar system is a simple way to keep track of tests and communications that have not yet been performed.
To trial
The jury deliberated for 18 minutes after a 5-day trial, and returned a verdict for the defense. As one of the physician-defendants was leaving the courtroom, he overheard the patient’s husband comforting her about the verdict. Her response resonated: “That’s OK. All I wanted was to know the real reason Bobby died.”
This was a painful way to relearn an important lesson. Although OB patients and their families file suit for any number of reasons, 20% state that one driving force is the need for information, and 24% believe a cover-up occurred.3
Further, although a defense verdict was returned, legal fees and lost time amounted to roughly $250,000 in costs—a substantial loss that a timely telephone call could have prevented.
Loss of trust can be exponential
All the warmth the care team shared with this patient and her family during her hospitalization became irrelevant after the lapse in follow-up. The team let this patient down by failing to implement a system to track her human needs as well as her acute clinical issues. One individual’s limitations of memory led to several years of anguish for a grieving family.
We have learned the importance of keeping track of Pap smear results, quad screens, mammograms, and other tests that have direct, acute impact on patient care, but many offices lack a system for tracking the fulfillment of other needs.
Such a system need not be complex. In this case, a tickler file would have sufficed—ie, a calendar or accordion file that contains individual reminders of tasks that need to be performed and the date they are required, such as the need to obtain results or to touch base about personal issues. (The reminder should also include patient contact information, to eliminate the need to look it up again.) If the results are delayed, the reminder can be refiled or reposted for the following week.
“Out of sight, out of mind”
Some of us have a photo of each patient taped to the inside front cover of her chart—along with her nickname, children’s names, life-cycle events, and key personal information. These pieces are a prompt that allows us to humanize the relationship during office visits. That approach works well for the patient, and for us: We use the chart to make notes about the need for clinical and, perhaps, personal follow-up. But there is one fatal flaw: The chart has no value once it is put back in the file rack, where we won’t see it when we need to act.
As we confront the complexity and demands of practice in the 21st century, we cannot rely on our intrinsic good character, good will, and good intentions. And we certainly cannot depend on our memory or trust that the documents that will direct us to our next step will land on our desk when we need them. We owe it to ourselves, our partners, and, most important, our patients to take the time to develop systems for the “miscellaneous” tasks that remind us when it is time to do the right thing.
The author reports no financial relationships relevant to this article.
1. Hickson GB, Clayton EW, Entman SS, et al. Obstetricians’ prior malpractice experience and patients’ satisfaction with care. JAMA. 1994;272:1583-1587.
2. Entman SS, Glass CA, Hickson GB, et al. The relationship between malpractice claims history and subsequent obstetrical care. JAMA. 1994;272:1588-1591.
3. Hickson GB, Clayton EW, Githens PB, et al. Factors that prompted families to file medical malpractice claims following perinatal injuries. JAMA. 1994;267:1359-1363.
As pressures upon us increase, we have begun to build teams and systems that ensure the kind of care our patients demand. We may not be able to deliver personal continuity to every patient, but a team can approximate that continuity—after it meets several key challenges. Foremost: developing systems of communication that are consistent, reliable, accurate, and accessible to any member of the team.
There is another locus in the continuum of care that is often neglected: post-event follow-up for medical, psychosocial, or legal purposes. The following case illustrates this point.
CASE A lawsuit is filed despite comprehensive care
C.S., a 34-year-old G4P3, at 35 weeks’ gestation with suspected premature rupture of membranes, was referred to the tertiary-care center where I practice. She had reported a gush of fluid, and the referring physician had observed nitrazine-positive fluid at the introitus.
On initial speculum examination at the tertiary-care center, no fluid was observed coming from the cervix, and vaginal secretions were nitrazine-negative. The cervix was long, posterior, and patulous. Ultrasonographic examination of the uterus demonstrated a normal fetus of appropriate size for the reported gestational age and a maximum pocket of amniotic fluid greater than 10 cm in depth. The fetus was active, and the nonstress test was reactive. The patient’s urine was alkaline; a specimen sent for culture was found to be negative.
Despite the reassuring clinical assessment, this mature multipara’s description of events was credible. She was offered the option of overnight observation or amniocentesis with instillation of indigo carmine. Because her husband and 3 children would have had to stay in their car overnight if she remained in the hospital, the patient chose instillation.
Amniocentesis was performed under sonographic guidance, with a return of clear fluid. The fluid was sent to the lab for fetal lung maturity testing, which was negative. Ten cubic centimeters of indigo carmine dye were instilled, and a tampon was inserted. After 2 hours of ambulation, there was no dye on the tampon, and another nonstress test was reactive. The patient was discharged.
The next day, the patient reported to her family physician complaining of severe uterine pain, fever, and a loss of fetal movement. When fetal heart activity could not be detected by Doppler ultrasound, she was again referred to the tertiary-care center. There she was noted to be in extreme pain, with a temperature of 104°F, and bulging forewaters. There was copious fluid and no fetal heart motion on ultrasonography.
Amniotomy elicited a gush of clear, blue, odorless fluid. The cervix dilated completely, and the fetal head was expelled to the chin. Examination revealed a nuchal and shoulder cord tightly wrapped and tethering descent. The vaginal wall was retracted, and the cord was visualized and divided. A stillborn male was immediately expelled, and the placenta followed rapidly. Bleeding remained within normal limits.
Although there was no explanation for the fever, the patient was treated with antibiotics during her postpartum hospital stay. She recovered quickly. Cultures from mother, baby, and placenta detected no organisms. The patient was discharged on day 4.
Ten months later, the patient filed a lawsuit alleging a failure to diagnose amnionitis at the time of the first visit.
What prompted the lawsuit?
Clearly, this patient had a tragic loss. Just as clearly, there were multiple incongruities between her clinical presentation and the outcome. The patient and the care team were both aware of these truths.
Research has demonstrated that physicians who interact in a positive manner with their patients are less likely to be sued than those who fail to communicate warmth and caring. Patients of physicians who have a history of multiple lawsuits may consider them knowledgeable and competent—but they also are likely to describe them as unavailable, abrupt, and disinterested. Patients often characterize their experiences with such physicians as negative even when the clinical outcome is good. This negativity often prevails even when the office staff is skilled in communication, education, and support.1,2
The team performed well
In the case of C.S., the physicians, by nature and by intent, were attentive to the human needs of this grieving family. Here is what we did well:
- The same residents provided care through both labor and delivery and during postpartum care
- The attending physician (me) was present through all clinical milestones
- All members of the team openly expressed their sorrow to the family
- I visited with the patient daily—providing honest answers to the family’s questions and acknowledging gaps in the medical team’s understanding of what had happened
- A follow-up plan was established to provide autopsy results to the family
So what went wrong? Why did the family—and the members of the care team—have to suffer the ordeal of litigation?
Critical lapse uncovered
Note the last bulleted item, above. This was the critical lapse: I did not call the family to relay the results of the autopsy. Why not? I knew better, after all, and prided myself on my commitment to all dimensions of the care I provided. As with most lapses in medical care, failure was multifactorial—part system design, part human failing.
At the tertiary-care institution in question, maternal transport high-risk pregnancies are managed with a group of attending obstetricians on a week-by-week rotational schedule. This provides continuity of care through the calendar week but, by its very nature, relieves the attending of the previous week from clinical responsibilities. By happenstance, the monthly rotation of residents coincided with this patient’s hospitalization. Thus, no member of her care team had continuing direct responsibility for the OB service.
To complicate matters, I left town after the delivery for a conference, with vacation tacked on afterwards. When I returned to the office 10 days later, my head was refreshed but my memory had been purged, and I failed to follow through on my promise to contact the family.
About 3 months after the delivery, I overheard a secretary trying to calm a frustrated patient on the telephone. When I heard the secretary say, “I’m sorry, ma’am, but we just don’t have the autopsy results, maybe you should call pathology,” I realized which patient it was and took over the conversation. Abjectly apologetic, I promised to get the information for her within hours. The patient was reserved but accepted my offer.
The autopsy revealed a polymorphonuclear leukocyte infiltration throughout the body, but no organisms could be identified by culture or on histologic examination. The final pathologist’s report provided no definitive explanation of the sequence of events that led to fetal death. When I explained this to the patient during a telephone call, her demeanor turned icy and she hung up. Several months later, the lawsuit was served.
1. Build a solid foundation. We all know communication is important, but many of us fail to take the extra steps necessary to standardize communication so that the entire care team is apprised of the goals for a given patient—as well as exactly how much progress has been made toward those goals. Various systems have been designed to accomplish this aim, many of them derived from the aviation industry. A small investment in time can reap big rewards. A few examples:
- “Time-out”—A pause before an invasive procedure to confirm that you have the correct patient and will be performing the appropriate procedure.
- “Snapshot”—An overview of cases within a defined time period, including identification of the team’s priorities. For example: “This morning we have 3 patients scheduled for surgery, beginning with Mrs. ‘A,’ whose hysterectomy for a large myomatous uterus will likely be time-consuming.”
- “Turn-over”—A synopsis of cases at the time they are handed over to another team member or a different team. The information provided should include outstanding tasks and tests.
- “De-brief”—Time set aside after a case to discuss what happened, what could have been handled differently, and what the next steps are. These sessions provide immediate feedback to the team and influence the care of future patients.
2. Don’t leave warmth and caring to your staff. The evidence is in: Physicians who interact in a positive manner with their patients are less likely to be sued than those who fail to communicate warmth and concern. Given the competing demands on our time, it is all too easy to rush through patient visits or other aspects of care without attending to the human component. Take a few minutes to greet each patient by name, inquire about her family and any concerns she may have about her condition, and listen attentively to her response. Then document any important details that arise during this discussion, so the rest of your team knows about them, too.
3. Offer and follow through on an evidence-based explanation of events. At times of tragedy, pay attention to the needs of grieving patients—and their families. This begins with an acknowledgment of the shock and sorrow they are experiencing and includes reassurance that the reasons for the adverse event will be explored and reported. This should not be an empty promise. It is important that the physician offers as full an explanation of an event as possible—as soon as all the facts are in—and that this explanation is voluntary, not something the patient has to ask for repeatedly.
4. Implement an effective tracking system. The case of C.S. illustrates the need for a more comprehensive tracking system. In that case, my failure to relay the need for autopsy results to other team members, and my subsequent absence from the scene, allowed a critical detail to slip through the cracks.
Because patient files tend to be forgotten once they are stored away, a tickle file or similar system is a simple way to keep track of tests and communications that have not yet been performed.
To trial
The jury deliberated for 18 minutes after a 5-day trial, and returned a verdict for the defense. As one of the physician-defendants was leaving the courtroom, he overheard the patient’s husband comforting her about the verdict. Her response resonated: “That’s OK. All I wanted was to know the real reason Bobby died.”
This was a painful way to relearn an important lesson. Although OB patients and their families file suit for any number of reasons, 20% state that one driving force is the need for information, and 24% believe a cover-up occurred.3
Further, although a defense verdict was returned, legal fees and lost time amounted to roughly $250,000 in costs—a substantial loss that a timely telephone call could have prevented.
Loss of trust can be exponential
All the warmth the care team shared with this patient and her family during her hospitalization became irrelevant after the lapse in follow-up. The team let this patient down by failing to implement a system to track her human needs as well as her acute clinical issues. One individual’s limitations of memory led to several years of anguish for a grieving family.
We have learned the importance of keeping track of Pap smear results, quad screens, mammograms, and other tests that have direct, acute impact on patient care, but many offices lack a system for tracking the fulfillment of other needs.
Such a system need not be complex. In this case, a tickler file would have sufficed—ie, a calendar or accordion file that contains individual reminders of tasks that need to be performed and the date they are required, such as the need to obtain results or to touch base about personal issues. (The reminder should also include patient contact information, to eliminate the need to look it up again.) If the results are delayed, the reminder can be refiled or reposted for the following week.
“Out of sight, out of mind”
Some of us have a photo of each patient taped to the inside front cover of her chart—along with her nickname, children’s names, life-cycle events, and key personal information. These pieces are a prompt that allows us to humanize the relationship during office visits. That approach works well for the patient, and for us: We use the chart to make notes about the need for clinical and, perhaps, personal follow-up. But there is one fatal flaw: The chart has no value once it is put back in the file rack, where we won’t see it when we need to act.
As we confront the complexity and demands of practice in the 21st century, we cannot rely on our intrinsic good character, good will, and good intentions. And we certainly cannot depend on our memory or trust that the documents that will direct us to our next step will land on our desk when we need them. We owe it to ourselves, our partners, and, most important, our patients to take the time to develop systems for the “miscellaneous” tasks that remind us when it is time to do the right thing.
The author reports no financial relationships relevant to this article.
As pressures upon us increase, we have begun to build teams and systems that ensure the kind of care our patients demand. We may not be able to deliver personal continuity to every patient, but a team can approximate that continuity—after it meets several key challenges. Foremost: developing systems of communication that are consistent, reliable, accurate, and accessible to any member of the team.
There is another locus in the continuum of care that is often neglected: post-event follow-up for medical, psychosocial, or legal purposes. The following case illustrates this point.
CASE A lawsuit is filed despite comprehensive care
C.S., a 34-year-old G4P3, at 35 weeks’ gestation with suspected premature rupture of membranes, was referred to the tertiary-care center where I practice. She had reported a gush of fluid, and the referring physician had observed nitrazine-positive fluid at the introitus.
On initial speculum examination at the tertiary-care center, no fluid was observed coming from the cervix, and vaginal secretions were nitrazine-negative. The cervix was long, posterior, and patulous. Ultrasonographic examination of the uterus demonstrated a normal fetus of appropriate size for the reported gestational age and a maximum pocket of amniotic fluid greater than 10 cm in depth. The fetus was active, and the nonstress test was reactive. The patient’s urine was alkaline; a specimen sent for culture was found to be negative.
Despite the reassuring clinical assessment, this mature multipara’s description of events was credible. She was offered the option of overnight observation or amniocentesis with instillation of indigo carmine. Because her husband and 3 children would have had to stay in their car overnight if she remained in the hospital, the patient chose instillation.
Amniocentesis was performed under sonographic guidance, with a return of clear fluid. The fluid was sent to the lab for fetal lung maturity testing, which was negative. Ten cubic centimeters of indigo carmine dye were instilled, and a tampon was inserted. After 2 hours of ambulation, there was no dye on the tampon, and another nonstress test was reactive. The patient was discharged.
The next day, the patient reported to her family physician complaining of severe uterine pain, fever, and a loss of fetal movement. When fetal heart activity could not be detected by Doppler ultrasound, she was again referred to the tertiary-care center. There she was noted to be in extreme pain, with a temperature of 104°F, and bulging forewaters. There was copious fluid and no fetal heart motion on ultrasonography.
Amniotomy elicited a gush of clear, blue, odorless fluid. The cervix dilated completely, and the fetal head was expelled to the chin. Examination revealed a nuchal and shoulder cord tightly wrapped and tethering descent. The vaginal wall was retracted, and the cord was visualized and divided. A stillborn male was immediately expelled, and the placenta followed rapidly. Bleeding remained within normal limits.
Although there was no explanation for the fever, the patient was treated with antibiotics during her postpartum hospital stay. She recovered quickly. Cultures from mother, baby, and placenta detected no organisms. The patient was discharged on day 4.
Ten months later, the patient filed a lawsuit alleging a failure to diagnose amnionitis at the time of the first visit.
What prompted the lawsuit?
Clearly, this patient had a tragic loss. Just as clearly, there were multiple incongruities between her clinical presentation and the outcome. The patient and the care team were both aware of these truths.
Research has demonstrated that physicians who interact in a positive manner with their patients are less likely to be sued than those who fail to communicate warmth and caring. Patients of physicians who have a history of multiple lawsuits may consider them knowledgeable and competent—but they also are likely to describe them as unavailable, abrupt, and disinterested. Patients often characterize their experiences with such physicians as negative even when the clinical outcome is good. This negativity often prevails even when the office staff is skilled in communication, education, and support.1,2
The team performed well
In the case of C.S., the physicians, by nature and by intent, were attentive to the human needs of this grieving family. Here is what we did well:
- The same residents provided care through both labor and delivery and during postpartum care
- The attending physician (me) was present through all clinical milestones
- All members of the team openly expressed their sorrow to the family
- I visited with the patient daily—providing honest answers to the family’s questions and acknowledging gaps in the medical team’s understanding of what had happened
- A follow-up plan was established to provide autopsy results to the family
So what went wrong? Why did the family—and the members of the care team—have to suffer the ordeal of litigation?
Critical lapse uncovered
Note the last bulleted item, above. This was the critical lapse: I did not call the family to relay the results of the autopsy. Why not? I knew better, after all, and prided myself on my commitment to all dimensions of the care I provided. As with most lapses in medical care, failure was multifactorial—part system design, part human failing.
At the tertiary-care institution in question, maternal transport high-risk pregnancies are managed with a group of attending obstetricians on a week-by-week rotational schedule. This provides continuity of care through the calendar week but, by its very nature, relieves the attending of the previous week from clinical responsibilities. By happenstance, the monthly rotation of residents coincided with this patient’s hospitalization. Thus, no member of her care team had continuing direct responsibility for the OB service.
To complicate matters, I left town after the delivery for a conference, with vacation tacked on afterwards. When I returned to the office 10 days later, my head was refreshed but my memory had been purged, and I failed to follow through on my promise to contact the family.
About 3 months after the delivery, I overheard a secretary trying to calm a frustrated patient on the telephone. When I heard the secretary say, “I’m sorry, ma’am, but we just don’t have the autopsy results, maybe you should call pathology,” I realized which patient it was and took over the conversation. Abjectly apologetic, I promised to get the information for her within hours. The patient was reserved but accepted my offer.
The autopsy revealed a polymorphonuclear leukocyte infiltration throughout the body, but no organisms could be identified by culture or on histologic examination. The final pathologist’s report provided no definitive explanation of the sequence of events that led to fetal death. When I explained this to the patient during a telephone call, her demeanor turned icy and she hung up. Several months later, the lawsuit was served.
1. Build a solid foundation. We all know communication is important, but many of us fail to take the extra steps necessary to standardize communication so that the entire care team is apprised of the goals for a given patient—as well as exactly how much progress has been made toward those goals. Various systems have been designed to accomplish this aim, many of them derived from the aviation industry. A small investment in time can reap big rewards. A few examples:
- “Time-out”—A pause before an invasive procedure to confirm that you have the correct patient and will be performing the appropriate procedure.
- “Snapshot”—An overview of cases within a defined time period, including identification of the team’s priorities. For example: “This morning we have 3 patients scheduled for surgery, beginning with Mrs. ‘A,’ whose hysterectomy for a large myomatous uterus will likely be time-consuming.”
- “Turn-over”—A synopsis of cases at the time they are handed over to another team member or a different team. The information provided should include outstanding tasks and tests.
- “De-brief”—Time set aside after a case to discuss what happened, what could have been handled differently, and what the next steps are. These sessions provide immediate feedback to the team and influence the care of future patients.
2. Don’t leave warmth and caring to your staff. The evidence is in: Physicians who interact in a positive manner with their patients are less likely to be sued than those who fail to communicate warmth and concern. Given the competing demands on our time, it is all too easy to rush through patient visits or other aspects of care without attending to the human component. Take a few minutes to greet each patient by name, inquire about her family and any concerns she may have about her condition, and listen attentively to her response. Then document any important details that arise during this discussion, so the rest of your team knows about them, too.
3. Offer and follow through on an evidence-based explanation of events. At times of tragedy, pay attention to the needs of grieving patients—and their families. This begins with an acknowledgment of the shock and sorrow they are experiencing and includes reassurance that the reasons for the adverse event will be explored and reported. This should not be an empty promise. It is important that the physician offers as full an explanation of an event as possible—as soon as all the facts are in—and that this explanation is voluntary, not something the patient has to ask for repeatedly.
4. Implement an effective tracking system. The case of C.S. illustrates the need for a more comprehensive tracking system. In that case, my failure to relay the need for autopsy results to other team members, and my subsequent absence from the scene, allowed a critical detail to slip through the cracks.
Because patient files tend to be forgotten once they are stored away, a tickle file or similar system is a simple way to keep track of tests and communications that have not yet been performed.
To trial
The jury deliberated for 18 minutes after a 5-day trial, and returned a verdict for the defense. As one of the physician-defendants was leaving the courtroom, he overheard the patient’s husband comforting her about the verdict. Her response resonated: “That’s OK. All I wanted was to know the real reason Bobby died.”
This was a painful way to relearn an important lesson. Although OB patients and their families file suit for any number of reasons, 20% state that one driving force is the need for information, and 24% believe a cover-up occurred.3
Further, although a defense verdict was returned, legal fees and lost time amounted to roughly $250,000 in costs—a substantial loss that a timely telephone call could have prevented.
Loss of trust can be exponential
All the warmth the care team shared with this patient and her family during her hospitalization became irrelevant after the lapse in follow-up. The team let this patient down by failing to implement a system to track her human needs as well as her acute clinical issues. One individual’s limitations of memory led to several years of anguish for a grieving family.
We have learned the importance of keeping track of Pap smear results, quad screens, mammograms, and other tests that have direct, acute impact on patient care, but many offices lack a system for tracking the fulfillment of other needs.
Such a system need not be complex. In this case, a tickler file would have sufficed—ie, a calendar or accordion file that contains individual reminders of tasks that need to be performed and the date they are required, such as the need to obtain results or to touch base about personal issues. (The reminder should also include patient contact information, to eliminate the need to look it up again.) If the results are delayed, the reminder can be refiled or reposted for the following week.
“Out of sight, out of mind”
Some of us have a photo of each patient taped to the inside front cover of her chart—along with her nickname, children’s names, life-cycle events, and key personal information. These pieces are a prompt that allows us to humanize the relationship during office visits. That approach works well for the patient, and for us: We use the chart to make notes about the need for clinical and, perhaps, personal follow-up. But there is one fatal flaw: The chart has no value once it is put back in the file rack, where we won’t see it when we need to act.
As we confront the complexity and demands of practice in the 21st century, we cannot rely on our intrinsic good character, good will, and good intentions. And we certainly cannot depend on our memory or trust that the documents that will direct us to our next step will land on our desk when we need them. We owe it to ourselves, our partners, and, most important, our patients to take the time to develop systems for the “miscellaneous” tasks that remind us when it is time to do the right thing.
The author reports no financial relationships relevant to this article.
1. Hickson GB, Clayton EW, Entman SS, et al. Obstetricians’ prior malpractice experience and patients’ satisfaction with care. JAMA. 1994;272:1583-1587.
2. Entman SS, Glass CA, Hickson GB, et al. The relationship between malpractice claims history and subsequent obstetrical care. JAMA. 1994;272:1588-1591.
3. Hickson GB, Clayton EW, Githens PB, et al. Factors that prompted families to file medical malpractice claims following perinatal injuries. JAMA. 1994;267:1359-1363.
1. Hickson GB, Clayton EW, Entman SS, et al. Obstetricians’ prior malpractice experience and patients’ satisfaction with care. JAMA. 1994;272:1583-1587.
2. Entman SS, Glass CA, Hickson GB, et al. The relationship between malpractice claims history and subsequent obstetrical care. JAMA. 1994;272:1588-1591.
3. Hickson GB, Clayton EW, Githens PB, et al. Factors that prompted families to file medical malpractice claims following perinatal injuries. JAMA. 1994;267:1359-1363.