User login
Hospitalist-Led, Post-Discharge Clinic Improves Care Transitions
Two Research, Hospital Innovations, and Clinical Vignettes (RIV) scientific posters presented at HM13 shed new light on the opportunities and challenges of hospitalist-run post-discharge clinics, which a growing number of hospitals have adopted in an attempt to smooth care transitions and prevent rehospitalizations.
The Denver VA Medical Center (VAMC) started its post-discharge clinic, located on a floor above its medicine wards, in 2003. Open two afternoons, the clinic sees up to 16 patients a week. Discharging housestaff are paged to meet their patients in the clinic as part of required afternoon activities, explains the poster’s lead author, Robert Burke, MD, a hospitalist at the VAMC and assistant professor of medicine at affiliated University of Colorado. Every patient is seen by a rotating, supervising hospitalist attending.
The clinic is able to see patients for their first post-discharge clinical encounter within five days on average, much sooner than either urgent care clinics (9.4 days) or primary care physicians (13.7 days).
However, data presented at HM13 found no reduction in readmissions for the VA clinic’s patients.1 Dr. Burke suggests that this finding reflects the challenges of connecting patients to their PCPs after the clinic visit. “Also, it’s not a full, multidisciplinary clinic—just housestaff and attendings,” he says. “The patients we see in the clinic are very ill.”
A second poster from the same team presented data from a national survey of hospitalists’ attitudes regarding post-discharge clinics.2 Three-quarters of 228 respondents believed that these clinics would reduce ED visits, but only 38% said that hospitalists should be seeing patients after discharge, and about 75% said doing so should require additional compensation for the physician.
“In my experience, I find it very valuable to see patients post-discharge as part of the larger continuum of care,” Dr. Burke says.
Larry Beresford is a freelance writer in San Francisco.
References
Two Research, Hospital Innovations, and Clinical Vignettes (RIV) scientific posters presented at HM13 shed new light on the opportunities and challenges of hospitalist-run post-discharge clinics, which a growing number of hospitals have adopted in an attempt to smooth care transitions and prevent rehospitalizations.
The Denver VA Medical Center (VAMC) started its post-discharge clinic, located on a floor above its medicine wards, in 2003. Open two afternoons, the clinic sees up to 16 patients a week. Discharging housestaff are paged to meet their patients in the clinic as part of required afternoon activities, explains the poster’s lead author, Robert Burke, MD, a hospitalist at the VAMC and assistant professor of medicine at affiliated University of Colorado. Every patient is seen by a rotating, supervising hospitalist attending.
The clinic is able to see patients for their first post-discharge clinical encounter within five days on average, much sooner than either urgent care clinics (9.4 days) or primary care physicians (13.7 days).
However, data presented at HM13 found no reduction in readmissions for the VA clinic’s patients.1 Dr. Burke suggests that this finding reflects the challenges of connecting patients to their PCPs after the clinic visit. “Also, it’s not a full, multidisciplinary clinic—just housestaff and attendings,” he says. “The patients we see in the clinic are very ill.”
A second poster from the same team presented data from a national survey of hospitalists’ attitudes regarding post-discharge clinics.2 Three-quarters of 228 respondents believed that these clinics would reduce ED visits, but only 38% said that hospitalists should be seeing patients after discharge, and about 75% said doing so should require additional compensation for the physician.
“In my experience, I find it very valuable to see patients post-discharge as part of the larger continuum of care,” Dr. Burke says.
Larry Beresford is a freelance writer in San Francisco.
References
Two Research, Hospital Innovations, and Clinical Vignettes (RIV) scientific posters presented at HM13 shed new light on the opportunities and challenges of hospitalist-run post-discharge clinics, which a growing number of hospitals have adopted in an attempt to smooth care transitions and prevent rehospitalizations.
The Denver VA Medical Center (VAMC) started its post-discharge clinic, located on a floor above its medicine wards, in 2003. Open two afternoons, the clinic sees up to 16 patients a week. Discharging housestaff are paged to meet their patients in the clinic as part of required afternoon activities, explains the poster’s lead author, Robert Burke, MD, a hospitalist at the VAMC and assistant professor of medicine at affiliated University of Colorado. Every patient is seen by a rotating, supervising hospitalist attending.
The clinic is able to see patients for their first post-discharge clinical encounter within five days on average, much sooner than either urgent care clinics (9.4 days) or primary care physicians (13.7 days).
However, data presented at HM13 found no reduction in readmissions for the VA clinic’s patients.1 Dr. Burke suggests that this finding reflects the challenges of connecting patients to their PCPs after the clinic visit. “Also, it’s not a full, multidisciplinary clinic—just housestaff and attendings,” he says. “The patients we see in the clinic are very ill.”
A second poster from the same team presented data from a national survey of hospitalists’ attitudes regarding post-discharge clinics.2 Three-quarters of 228 respondents believed that these clinics would reduce ED visits, but only 38% said that hospitalists should be seeing patients after discharge, and about 75% said doing so should require additional compensation for the physician.
“In my experience, I find it very valuable to see patients post-discharge as part of the larger continuum of care,” Dr. Burke says.
Larry Beresford is a freelance writer in San Francisco.
References
Safety WalkRounds at Children's Hospital Improve Teamwork
A recent study in The Joint Commission Journal on Quality and Patient Safety describes unit-based Patient Safety Leadership WalkRounds conducted on six pilot units at the Children’s Hospital of Philadelphia (CHOP) and how they have helped clinical leaders identify and address safety concerns.1 The WalkRound team, made up of at least one senior executive, a patient safety officer, and the manager of each unit, engages frontline staff in safety assessments and concerns.
The concept was developed by Allan Frankel, MD, director of patient safety at Partners Healthcare System in Boston, to increase awareness of safety issues by all clinicians, make safety a high priority for senior leadership, and act on the safety information collected from frontline staff.
At CHOP, the WalkRounds led to improved nurse-physician relationships, workflow, and medical safety, and uncovered previously unidentified safety concerns.
Reference
A recent study in The Joint Commission Journal on Quality and Patient Safety describes unit-based Patient Safety Leadership WalkRounds conducted on six pilot units at the Children’s Hospital of Philadelphia (CHOP) and how they have helped clinical leaders identify and address safety concerns.1 The WalkRound team, made up of at least one senior executive, a patient safety officer, and the manager of each unit, engages frontline staff in safety assessments and concerns.
The concept was developed by Allan Frankel, MD, director of patient safety at Partners Healthcare System in Boston, to increase awareness of safety issues by all clinicians, make safety a high priority for senior leadership, and act on the safety information collected from frontline staff.
At CHOP, the WalkRounds led to improved nurse-physician relationships, workflow, and medical safety, and uncovered previously unidentified safety concerns.
Reference
A recent study in The Joint Commission Journal on Quality and Patient Safety describes unit-based Patient Safety Leadership WalkRounds conducted on six pilot units at the Children’s Hospital of Philadelphia (CHOP) and how they have helped clinical leaders identify and address safety concerns.1 The WalkRound team, made up of at least one senior executive, a patient safety officer, and the manager of each unit, engages frontline staff in safety assessments and concerns.
The concept was developed by Allan Frankel, MD, director of patient safety at Partners Healthcare System in Boston, to increase awareness of safety issues by all clinicians, make safety a high priority for senior leadership, and act on the safety information collected from frontline staff.
At CHOP, the WalkRounds led to improved nurse-physician relationships, workflow, and medical safety, and uncovered previously unidentified safety concerns.
Reference
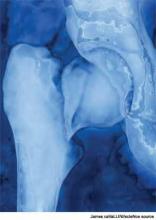
How Should Patients with Acute Hip Fractures Be Managed Perioperatively?
Case
A 91-year-old man with Alzheimer’s dementia presents with severe right hip pain after a fall at his nursing home. His family reports that he is dependent in most of his activities of daily living (ADLs) and can normally ambulate short distances with a walker. He is alert and oriented at baseline but has been more confused since his wife died a week earlier from pneumonia. His only new medication is lorazepam as needed for anxiety. On admission, the patient is diagnosed with a displaced femoral neck fracture, delirium, and healthcare-associated pneumonia, with a new oxygen requirement of 5 L/min. The orthopedic surgery service requests a medicine consult. How should this patient be managed perioperatively?
Overview
Hip fractures are a major health burden on the United States’ geriatric population. The lifetime risk of hip fracture is approximately 17% for Caucasian women and 6% for Caucasian men.1 In 2010, an estimated 258,000 people aged 65 years and older were hospitalized with hip fractures.2 This number is expected to climb to 289,000 by 2030.
In total, hip fractures directly cost the healthcare system about $18 billion per year.1
Hip fractures, like most other geriatric syndromes, are almost invariably multifactorial in etiology. They occur at the intersection of general frailty, bone fragility, and fall risk. Hip fractures too often trigger a further downward spiral in elderly patients, as deconditioning and acute complications compound chronic comorbidities and compromise any remaining physiologic reserve. Mortality after a hip fracture approaches 25% at one year.3 An excess mortality risk persists for at least 10 years.4 Of the patients who survive six months, only 50% can perform their ADLs, and only 25% can perform their instrumental ADLs as well as they could prior to their fracture.5,6
Unsurprisingly, older adults with hip fractures are five times more likely to require nursing home placement at one year.5
Hospitalists frequently encounter patients with hip fractures in the perioperative setting. Given their close collaboration with orthopedic surgeons and emphasis on transitions of care, hospitalists can play an important role in reversing the trajectory of death and disability following hip fractures. Key aspects of inpatient management are outlined below.
Hip Fracture Repair
Hip fractures can be divided into intracapsular (femoral neck) or extracapsular (intratrochanteric or subtrochanteric) fractures. Their relative frequencies are listed in Table 1.7
Surgery types. Femoral neck fractures typically are the most difficult to heal, given a limited regional blood supply.5,7 Displaced femoral neck fractures require either a hemiarthroplasty or total hip arthroplasty. Over time, hemiarthroplasties tend to cause hip pain from acetabular erosion, so they are better suited for less active, elderly patients. Nondisplaced femoral neck, intratrochanteric, and subtrochanteric fractures are usually managed with open reduction and internal fixation.
The overall goal of surgery is to return patients to their prior level of functioning. In the short term, surgery also provides pain relief and allows for early mobilization. Nonoperative management is generally reserved for patients with very high operative risk or limited life expectancies or those who are bedridden at baseline.
Timing of surgery. In general, hip fracture repair should be performed within 24-48 hours of admission in patients who are medically stable. Though early surgery may not improve functional outcomes or mortality, it has been associated with improved pain control, decreased length of stay, and fewer major complications.8 Patients with active medical conditions (e.g. pneumonia) should be medically optimized before proceeding with surgery. A 2011 study found that most of the excess in-hospital mortality associated with surgical delays beyond five days was attributable to the active medical issues rather than to the delay itself.9
Prevention of Perioperative Complications
The principles of geriatric medicine should be applied to the care of elderly patients with hip fractures. Emphasis should be placed on early recognition of treatable conditions and avoidance of iatrogenesis. Careful assessment of medical problems, social support, and functional status within an interdisciplinary framework is recommended. Such a multi-faceted approach has been shown to reduce overall complications in hip fracture patients.10 Specific complications are discussed in more detail below.
Delirium. Delirium is the most common complication after hip fracture surgery, with a prevalence of 35%-65%.7 Proper pain control, minimization of polypharmacy, removal of tethers, and frequent reorientation are among the many preventive measures that should be implemented.
Venous thromboembolism (VTE). VTE is a leading cause of morbidity and mortality for hip fracture patients.11 Without prophylaxis, about 1.8% will develop symptomatic deep venous thromboses, and 1% will develop symptomatic pulmonary emboli in the first seven to 14 days after surgery. An estimated 4.3% will develop symptomatic VTE in the first 35 days after surgery.
The American College of Chest Physicians recommends that patients undergoing hip fracture surgery receive VTE prophylaxis for a minimum of 10-14 days postoperatively.11 Extending prophylaxis out to 35 days is reasonable. Low molecular-weight heparin is preferred over low-dose unfractionated heparin, fondaparinux, warfarin, and aspirin. Patients should receive preoperative VTE prophylaxis if surgery is delayed.
Postoperative infections. Urinary tract infections (UTIs) are the most common infectious complication after hip fracture surgery.7 If not caught early, they can result in urosepsis, prosthetic joint infections, and death. After the first 48 hours of urinary catheterization, the risk of a UTI is 5%-10% per day.12
Therefore, catheters should be removed within 24-48 hours of surgery.
Acute blood loss anemia. Anemia is common in hip fracture patients. It may be present on admission or develop as a result of intraoperative blood loss, ongoing drain output, or fluid resuscitation.
The recent FOCUS trial, which helped to clarify the optimal transfusion threshold for patients after hip fracture surgery, compared a liberal versus restrictive transfusion strategy in patients with cardiovascular disease.13 Transfusing for a hemoglobin < 10 g/dL, as opposed to transfusing for symptoms or a hemoglobin < 8 g/dL, did not improve mortality, in-hospital morbidity (including myocardial infarction), or functional status at 60 days.
Pressure ulcers. Patients with hip fractures are at risk of developing decubitus ulcers. One study found the incidence of new pressure ulcers to be 16% at seven days and 36% at 32 days after initial hospitalization.14 Multicomponent interventions have been shown to successfully reduce the rate of hospital-acquired pressure ulcers.15
Medical Management of Osteoporosis
The World Heath Organization defines osteoporosis as a bone mineral density of at least 2.5 standard deviations below that of a “normal” young adult as measured on DEXA scan, or a T-score ≤ -2.5.16 However, it is important to recognize that bone strength depends not only on the quantity of bone but also on the quality. Any patient who sustains a hip fracture with minimal trauma (e.g. a fall from standing height) should be considered to have osteoporosis, regardless of T-score.
Patients with their first hip fracture are 2.5 times more likely to have a future fragility fracture.17 Hospitalists must therefore make secondary prevention a priority. Medical management focuses on maintaining bone strength, slowing further bone loss, and preventing future falls.
Evaluation. A directed history and physical examination should be completed to screen for secondary causes of osteoporosis. A basic laboratory workup is reasonable in the inpatient setting (see Table 2).17 Other tests, such as a serum and urine protein electrophoresis, can be obtained as clinically indicated.
Patients require counseling directed at lifestyle factors, including the importance of weight-bearing exercise, smoking cessation, and avoidance of excessive alcohol intake. A comprehensive falls assessment is also warranted.
Treatment. All patients with hip fractures should be discharged from the hospital on calcium and vitamin D supplementation, unless there is a specific contraindication.18 Guidelines vary by organization, but the National Osteoporosis Foundation’s recommendations are listed in Table 3.17,19,20 Dietary calcium is usually insufficient to meet the daily requirement.
Bisphosphonates are considered first-line therapy for osteoporosis.17 The HORIZON trial was a randomized, placebo-controlled study that evaluated annual zoledronic acid infusions in hip fracture patients who were intolerant of oral bisphosphonates.21 Zoledronic acid reduced the rate of new fractures by 35% after 1.9 years, with a number needed to treat (NNT) of 19. It also improved survival by 28%, for an NNT of 27. All subjects also received calcium and vitamin D supplementation.
Both hospitalists and orthopedists might worry about bisphosphonates adversely affecting bone healing in the acute setting. Subsequent analyses from the HORIZON trial suggest that bisphosphonates can be safely started as soon as two weeks after surgery.22,23
Transitions of care. Despite well-established guidelines for the treatment of osteoporosis, patients with hip fractures often are undertreated. A retrospective study of 420 acute hip fracture patients found that only 37% received calcium, 36% received vitamin D, and 31% received a bisphosphonate on discharge.24 A prospective study of 1,075 women with new osteoporotic fractures found that only 17% had started anti-osteoporosis medications at one year.25
Hospitalists should recognize and address potential barriers to appropriate medical therapy. Patient-related obstacles may include the cost of medications, concerns about side effects, and lack of a PCP.24,25 Hospitalists should document the diagnosis of osteoporosis in the medical record so subsequent providers are attuned to the issue.26 They should also clarify the ownership of osteoporosis across the continuum of care, because medicine consultants, orthopedists, primary care or rehabilitation physicians, and subspecialists may all be involved. Hospitalists can certainly take advantage of this window of opportunity by starting patients on osteoporosis treatment and ensuring smooth transitions of care on discharge.
Back to the Case
The patient was started on intravenous antibiotics for healthcare-associated pneumonia with improvement of his oxygen requirement to 3 L/min. He underwent a right hemiarthroplasty on hospital day five and tolerated the procedure well. His delirium resolved with treatment of his infection, pain control, discontinuation of lorazepam, and other conservative measures. He was given VTE prophylaxis pre- and postoperatively. His urinary catheter was discontinued on day one after surgery. He was started on calcium supplementation and vitamin D repletion after his 25-OH vitamin D level returned low at 14 ng/mL.
The patient progressed well with physical and occupational therapy and was discharged back to his skilled nursing facility, with plans to start a bisphosphonate in two weeks.
Bottom Line
Hospitalists should be familiar with the best practices for the perioperative management of hip fracture patients.
Dr. Anderson and Dr. Wolfe are hospitalists at the University of Colorado Hospital in Aurora. Dr. Anderson directs the medicine consult service, and Dr. Wolfe serves as the associate director.
References
- The Joint Commission. Improving and measuring osteoporosis treatment 2008. Oakbrook Terrace, Il; 2008. Available at: http://www.jointcommission.org/improving_and_measuring_osteoporosis_management/. Accessed September 29, 2013.
- Stevens JA, Rudd RA. The impact of decreasing U.S. hip fracture rates on future hip fracture estimates. Osteoporos Int. 2013;24(10):2725-2728.
- Braithwaite RS, Col NF, Wong JB. Estimating hip fracture morbidity, mortality, and costs. J Am Geriatr Soc. 2003;51(3):364-370. Haentjens P, Magaziner J, Colón-Emeric CS, et al.
- Meta-analysis: excess mortality after hip fracture among older women and men. Ann Intern Med. 2010;152(6):380-390.
- Hung WW, Egol KA, Zuckerman JD, Siu AL. Hip fracture management: tailoring care for the older patient. JAMA. 2012;307(20):2185-2194.
- Magaziner J, Simonsick EM, Kashner TM, Hebel JR, Kenzora JE. Predictors of functional recovery one year following hospital discharge for hip fracture: a prospective study. J Gerontol. 1990;45(3):M101-107.
- Bateman L, Vuppala S, Porada P, et al. Medical management in the acute hip fracture patient: a comprehensive review for the internist. Ochsner J. 2012;12(2):101-110.
- Orosz GM, Magaziner J, Hannan EL, et al. Association of timing of surgery for hip fracture and patient outcomes. JAMA. 2004;291(14):1738-1743.
- Vidán MT, Sánchez E, Gracia Y, Marañón E, Vaquero J, Serra JA. Causes and effects of surgical delay in patients with hip fracture: a cohort study. Ann Intern Med. 2011;155(4):226-233.
- Vidán M, Serra JA, Moreno C, Riquelme G, Ortiz J. Efficacy of a comprehensive geriatric intervention in older patients hospitalized for hip fracture: a randomized, controlled trial. J Am Geriatr Soc. 2005;53(9):1476-1482.
- Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e278S-e325S.
- Wald H, Epstein A, Kramer A. Extended use of indwelling urinary catheters in postoperative hip fracture patients. Med Care. 2005;43(10):1009-1017.
- Carson JL, Terrin ML, Noveck H, et al. Liberal or restrictive transfusion in high-risk patients after hip surgery. N Engl J Med. 2011;365(26):2453-2462.
- Baumgarten M, Margolis DJ, Orwig DL, et al. Pressure ulcers in elderly patients with hip fracture across the continuum of care. J Am Geriatr Soc. 2009;57(5):863-870.
- Sullivan N, Schoelles KM. Preventing in-facility pressure ulcers as a patient safety strategy: a systematic review. Ann Intern Med. 2013;158(5 Pt 2):410-416.
- World Health Organization. Prevention and management of osteoporosis: Report of a WHO scientific group 2000. Geneva, Switzerland; 2000. WHO technical report series; 921. Available at: http://whqlibdoc.who.int/trs/who_trs_921.pdf. Accessed July 9, 2013.
- National Osteoporosis Foundation. Clinician’s guide to prevention and treatment of osteoporosis 2013. Washington, D.C.; 2013. Available at: www.nof.org/files/nof/public/content/file/917/upload/481.pdf. Accessed September 29, 2013.
- Tang BM, Eslick GD, Nowson C, Smith C, Bensoussan A. Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet. 2007;370(9588):657-666.
- Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911-1930.
- Ross AC, Taylor CL, Yaktine AL, Del Valle HB, eds. Dietary reference intakes for calcium and vitamin D. Washington, D.C.: National Academies Press; 2011. Lyles KW, Colón-Emeric CS, Magaziner JS, et al.
- Zoledronic acid in reducing clinical fracture and mortality after hip fracture. N Engl J Med. 2007;357: nihpa40967.
- Colón-Emeric CS, Nordsletten L, Olson S, et al. Association between timing of zoledronic acid infusion and hip fracture healing. Osteoporos Int. 2011;22(8):2329-2336.
- Eriksen EF, Lyles KW, Colón-Emeric CS, et al. Antifracture efficacy and reduction of mortality in relation to timing of the first dose of zoledronic acid after hip fracture. J Bone Miner Res. 2009;24(7):1308-1313.
- Byszewski A, Lemay G, Molnar F, Azad N, McMartin SE. Closing the osteoporosis care gap in hip fracture patients: an opportunity to decrease recurrent fractures and hospital admissions. J Osteoporos. 2011;2011:404969.
- Greenspan SL, Wyman, A, Hoovan FH, et al. Predictors of treatment with osteoporosis medications after recent fragility fractures in a multinational cohort of postmenopausal women. J Am Geriatr Soc. 2012;60(3):455-461.
- Kamel HK, Hussain MS, Tariq S, Perry HM, Morley JE. Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture. Am J Med. 2000;109(4):326-328.
Case
A 91-year-old man with Alzheimer’s dementia presents with severe right hip pain after a fall at his nursing home. His family reports that he is dependent in most of his activities of daily living (ADLs) and can normally ambulate short distances with a walker. He is alert and oriented at baseline but has been more confused since his wife died a week earlier from pneumonia. His only new medication is lorazepam as needed for anxiety. On admission, the patient is diagnosed with a displaced femoral neck fracture, delirium, and healthcare-associated pneumonia, with a new oxygen requirement of 5 L/min. The orthopedic surgery service requests a medicine consult. How should this patient be managed perioperatively?
Overview
Hip fractures are a major health burden on the United States’ geriatric population. The lifetime risk of hip fracture is approximately 17% for Caucasian women and 6% for Caucasian men.1 In 2010, an estimated 258,000 people aged 65 years and older were hospitalized with hip fractures.2 This number is expected to climb to 289,000 by 2030.
In total, hip fractures directly cost the healthcare system about $18 billion per year.1
Hip fractures, like most other geriatric syndromes, are almost invariably multifactorial in etiology. They occur at the intersection of general frailty, bone fragility, and fall risk. Hip fractures too often trigger a further downward spiral in elderly patients, as deconditioning and acute complications compound chronic comorbidities and compromise any remaining physiologic reserve. Mortality after a hip fracture approaches 25% at one year.3 An excess mortality risk persists for at least 10 years.4 Of the patients who survive six months, only 50% can perform their ADLs, and only 25% can perform their instrumental ADLs as well as they could prior to their fracture.5,6
Unsurprisingly, older adults with hip fractures are five times more likely to require nursing home placement at one year.5
Hospitalists frequently encounter patients with hip fractures in the perioperative setting. Given their close collaboration with orthopedic surgeons and emphasis on transitions of care, hospitalists can play an important role in reversing the trajectory of death and disability following hip fractures. Key aspects of inpatient management are outlined below.
Hip Fracture Repair
Hip fractures can be divided into intracapsular (femoral neck) or extracapsular (intratrochanteric or subtrochanteric) fractures. Their relative frequencies are listed in Table 1.7
Surgery types. Femoral neck fractures typically are the most difficult to heal, given a limited regional blood supply.5,7 Displaced femoral neck fractures require either a hemiarthroplasty or total hip arthroplasty. Over time, hemiarthroplasties tend to cause hip pain from acetabular erosion, so they are better suited for less active, elderly patients. Nondisplaced femoral neck, intratrochanteric, and subtrochanteric fractures are usually managed with open reduction and internal fixation.
The overall goal of surgery is to return patients to their prior level of functioning. In the short term, surgery also provides pain relief and allows for early mobilization. Nonoperative management is generally reserved for patients with very high operative risk or limited life expectancies or those who are bedridden at baseline.
Timing of surgery. In general, hip fracture repair should be performed within 24-48 hours of admission in patients who are medically stable. Though early surgery may not improve functional outcomes or mortality, it has been associated with improved pain control, decreased length of stay, and fewer major complications.8 Patients with active medical conditions (e.g. pneumonia) should be medically optimized before proceeding with surgery. A 2011 study found that most of the excess in-hospital mortality associated with surgical delays beyond five days was attributable to the active medical issues rather than to the delay itself.9
Prevention of Perioperative Complications
The principles of geriatric medicine should be applied to the care of elderly patients with hip fractures. Emphasis should be placed on early recognition of treatable conditions and avoidance of iatrogenesis. Careful assessment of medical problems, social support, and functional status within an interdisciplinary framework is recommended. Such a multi-faceted approach has been shown to reduce overall complications in hip fracture patients.10 Specific complications are discussed in more detail below.
Delirium. Delirium is the most common complication after hip fracture surgery, with a prevalence of 35%-65%.7 Proper pain control, minimization of polypharmacy, removal of tethers, and frequent reorientation are among the many preventive measures that should be implemented.
Venous thromboembolism (VTE). VTE is a leading cause of morbidity and mortality for hip fracture patients.11 Without prophylaxis, about 1.8% will develop symptomatic deep venous thromboses, and 1% will develop symptomatic pulmonary emboli in the first seven to 14 days after surgery. An estimated 4.3% will develop symptomatic VTE in the first 35 days after surgery.
The American College of Chest Physicians recommends that patients undergoing hip fracture surgery receive VTE prophylaxis for a minimum of 10-14 days postoperatively.11 Extending prophylaxis out to 35 days is reasonable. Low molecular-weight heparin is preferred over low-dose unfractionated heparin, fondaparinux, warfarin, and aspirin. Patients should receive preoperative VTE prophylaxis if surgery is delayed.
Postoperative infections. Urinary tract infections (UTIs) are the most common infectious complication after hip fracture surgery.7 If not caught early, they can result in urosepsis, prosthetic joint infections, and death. After the first 48 hours of urinary catheterization, the risk of a UTI is 5%-10% per day.12
Therefore, catheters should be removed within 24-48 hours of surgery.
Acute blood loss anemia. Anemia is common in hip fracture patients. It may be present on admission or develop as a result of intraoperative blood loss, ongoing drain output, or fluid resuscitation.
The recent FOCUS trial, which helped to clarify the optimal transfusion threshold for patients after hip fracture surgery, compared a liberal versus restrictive transfusion strategy in patients with cardiovascular disease.13 Transfusing for a hemoglobin < 10 g/dL, as opposed to transfusing for symptoms or a hemoglobin < 8 g/dL, did not improve mortality, in-hospital morbidity (including myocardial infarction), or functional status at 60 days.
Pressure ulcers. Patients with hip fractures are at risk of developing decubitus ulcers. One study found the incidence of new pressure ulcers to be 16% at seven days and 36% at 32 days after initial hospitalization.14 Multicomponent interventions have been shown to successfully reduce the rate of hospital-acquired pressure ulcers.15
Medical Management of Osteoporosis
The World Heath Organization defines osteoporosis as a bone mineral density of at least 2.5 standard deviations below that of a “normal” young adult as measured on DEXA scan, or a T-score ≤ -2.5.16 However, it is important to recognize that bone strength depends not only on the quantity of bone but also on the quality. Any patient who sustains a hip fracture with minimal trauma (e.g. a fall from standing height) should be considered to have osteoporosis, regardless of T-score.
Patients with their first hip fracture are 2.5 times more likely to have a future fragility fracture.17 Hospitalists must therefore make secondary prevention a priority. Medical management focuses on maintaining bone strength, slowing further bone loss, and preventing future falls.
Evaluation. A directed history and physical examination should be completed to screen for secondary causes of osteoporosis. A basic laboratory workup is reasonable in the inpatient setting (see Table 2).17 Other tests, such as a serum and urine protein electrophoresis, can be obtained as clinically indicated.
Patients require counseling directed at lifestyle factors, including the importance of weight-bearing exercise, smoking cessation, and avoidance of excessive alcohol intake. A comprehensive falls assessment is also warranted.
Treatment. All patients with hip fractures should be discharged from the hospital on calcium and vitamin D supplementation, unless there is a specific contraindication.18 Guidelines vary by organization, but the National Osteoporosis Foundation’s recommendations are listed in Table 3.17,19,20 Dietary calcium is usually insufficient to meet the daily requirement.
Bisphosphonates are considered first-line therapy for osteoporosis.17 The HORIZON trial was a randomized, placebo-controlled study that evaluated annual zoledronic acid infusions in hip fracture patients who were intolerant of oral bisphosphonates.21 Zoledronic acid reduced the rate of new fractures by 35% after 1.9 years, with a number needed to treat (NNT) of 19. It also improved survival by 28%, for an NNT of 27. All subjects also received calcium and vitamin D supplementation.
Both hospitalists and orthopedists might worry about bisphosphonates adversely affecting bone healing in the acute setting. Subsequent analyses from the HORIZON trial suggest that bisphosphonates can be safely started as soon as two weeks after surgery.22,23
Transitions of care. Despite well-established guidelines for the treatment of osteoporosis, patients with hip fractures often are undertreated. A retrospective study of 420 acute hip fracture patients found that only 37% received calcium, 36% received vitamin D, and 31% received a bisphosphonate on discharge.24 A prospective study of 1,075 women with new osteoporotic fractures found that only 17% had started anti-osteoporosis medications at one year.25
Hospitalists should recognize and address potential barriers to appropriate medical therapy. Patient-related obstacles may include the cost of medications, concerns about side effects, and lack of a PCP.24,25 Hospitalists should document the diagnosis of osteoporosis in the medical record so subsequent providers are attuned to the issue.26 They should also clarify the ownership of osteoporosis across the continuum of care, because medicine consultants, orthopedists, primary care or rehabilitation physicians, and subspecialists may all be involved. Hospitalists can certainly take advantage of this window of opportunity by starting patients on osteoporosis treatment and ensuring smooth transitions of care on discharge.
Back to the Case
The patient was started on intravenous antibiotics for healthcare-associated pneumonia with improvement of his oxygen requirement to 3 L/min. He underwent a right hemiarthroplasty on hospital day five and tolerated the procedure well. His delirium resolved with treatment of his infection, pain control, discontinuation of lorazepam, and other conservative measures. He was given VTE prophylaxis pre- and postoperatively. His urinary catheter was discontinued on day one after surgery. He was started on calcium supplementation and vitamin D repletion after his 25-OH vitamin D level returned low at 14 ng/mL.
The patient progressed well with physical and occupational therapy and was discharged back to his skilled nursing facility, with plans to start a bisphosphonate in two weeks.
Bottom Line
Hospitalists should be familiar with the best practices for the perioperative management of hip fracture patients.
Dr. Anderson and Dr. Wolfe are hospitalists at the University of Colorado Hospital in Aurora. Dr. Anderson directs the medicine consult service, and Dr. Wolfe serves as the associate director.
References
- The Joint Commission. Improving and measuring osteoporosis treatment 2008. Oakbrook Terrace, Il; 2008. Available at: http://www.jointcommission.org/improving_and_measuring_osteoporosis_management/. Accessed September 29, 2013.
- Stevens JA, Rudd RA. The impact of decreasing U.S. hip fracture rates on future hip fracture estimates. Osteoporos Int. 2013;24(10):2725-2728.
- Braithwaite RS, Col NF, Wong JB. Estimating hip fracture morbidity, mortality, and costs. J Am Geriatr Soc. 2003;51(3):364-370. Haentjens P, Magaziner J, Colón-Emeric CS, et al.
- Meta-analysis: excess mortality after hip fracture among older women and men. Ann Intern Med. 2010;152(6):380-390.
- Hung WW, Egol KA, Zuckerman JD, Siu AL. Hip fracture management: tailoring care for the older patient. JAMA. 2012;307(20):2185-2194.
- Magaziner J, Simonsick EM, Kashner TM, Hebel JR, Kenzora JE. Predictors of functional recovery one year following hospital discharge for hip fracture: a prospective study. J Gerontol. 1990;45(3):M101-107.
- Bateman L, Vuppala S, Porada P, et al. Medical management in the acute hip fracture patient: a comprehensive review for the internist. Ochsner J. 2012;12(2):101-110.
- Orosz GM, Magaziner J, Hannan EL, et al. Association of timing of surgery for hip fracture and patient outcomes. JAMA. 2004;291(14):1738-1743.
- Vidán MT, Sánchez E, Gracia Y, Marañón E, Vaquero J, Serra JA. Causes and effects of surgical delay in patients with hip fracture: a cohort study. Ann Intern Med. 2011;155(4):226-233.
- Vidán M, Serra JA, Moreno C, Riquelme G, Ortiz J. Efficacy of a comprehensive geriatric intervention in older patients hospitalized for hip fracture: a randomized, controlled trial. J Am Geriatr Soc. 2005;53(9):1476-1482.
- Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e278S-e325S.
- Wald H, Epstein A, Kramer A. Extended use of indwelling urinary catheters in postoperative hip fracture patients. Med Care. 2005;43(10):1009-1017.
- Carson JL, Terrin ML, Noveck H, et al. Liberal or restrictive transfusion in high-risk patients after hip surgery. N Engl J Med. 2011;365(26):2453-2462.
- Baumgarten M, Margolis DJ, Orwig DL, et al. Pressure ulcers in elderly patients with hip fracture across the continuum of care. J Am Geriatr Soc. 2009;57(5):863-870.
- Sullivan N, Schoelles KM. Preventing in-facility pressure ulcers as a patient safety strategy: a systematic review. Ann Intern Med. 2013;158(5 Pt 2):410-416.
- World Health Organization. Prevention and management of osteoporosis: Report of a WHO scientific group 2000. Geneva, Switzerland; 2000. WHO technical report series; 921. Available at: http://whqlibdoc.who.int/trs/who_trs_921.pdf. Accessed July 9, 2013.
- National Osteoporosis Foundation. Clinician’s guide to prevention and treatment of osteoporosis 2013. Washington, D.C.; 2013. Available at: www.nof.org/files/nof/public/content/file/917/upload/481.pdf. Accessed September 29, 2013.
- Tang BM, Eslick GD, Nowson C, Smith C, Bensoussan A. Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet. 2007;370(9588):657-666.
- Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911-1930.
- Ross AC, Taylor CL, Yaktine AL, Del Valle HB, eds. Dietary reference intakes for calcium and vitamin D. Washington, D.C.: National Academies Press; 2011. Lyles KW, Colón-Emeric CS, Magaziner JS, et al.
- Zoledronic acid in reducing clinical fracture and mortality after hip fracture. N Engl J Med. 2007;357: nihpa40967.
- Colón-Emeric CS, Nordsletten L, Olson S, et al. Association between timing of zoledronic acid infusion and hip fracture healing. Osteoporos Int. 2011;22(8):2329-2336.
- Eriksen EF, Lyles KW, Colón-Emeric CS, et al. Antifracture efficacy and reduction of mortality in relation to timing of the first dose of zoledronic acid after hip fracture. J Bone Miner Res. 2009;24(7):1308-1313.
- Byszewski A, Lemay G, Molnar F, Azad N, McMartin SE. Closing the osteoporosis care gap in hip fracture patients: an opportunity to decrease recurrent fractures and hospital admissions. J Osteoporos. 2011;2011:404969.
- Greenspan SL, Wyman, A, Hoovan FH, et al. Predictors of treatment with osteoporosis medications after recent fragility fractures in a multinational cohort of postmenopausal women. J Am Geriatr Soc. 2012;60(3):455-461.
- Kamel HK, Hussain MS, Tariq S, Perry HM, Morley JE. Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture. Am J Med. 2000;109(4):326-328.
Case
A 91-year-old man with Alzheimer’s dementia presents with severe right hip pain after a fall at his nursing home. His family reports that he is dependent in most of his activities of daily living (ADLs) and can normally ambulate short distances with a walker. He is alert and oriented at baseline but has been more confused since his wife died a week earlier from pneumonia. His only new medication is lorazepam as needed for anxiety. On admission, the patient is diagnosed with a displaced femoral neck fracture, delirium, and healthcare-associated pneumonia, with a new oxygen requirement of 5 L/min. The orthopedic surgery service requests a medicine consult. How should this patient be managed perioperatively?
Overview
Hip fractures are a major health burden on the United States’ geriatric population. The lifetime risk of hip fracture is approximately 17% for Caucasian women and 6% for Caucasian men.1 In 2010, an estimated 258,000 people aged 65 years and older were hospitalized with hip fractures.2 This number is expected to climb to 289,000 by 2030.
In total, hip fractures directly cost the healthcare system about $18 billion per year.1
Hip fractures, like most other geriatric syndromes, are almost invariably multifactorial in etiology. They occur at the intersection of general frailty, bone fragility, and fall risk. Hip fractures too often trigger a further downward spiral in elderly patients, as deconditioning and acute complications compound chronic comorbidities and compromise any remaining physiologic reserve. Mortality after a hip fracture approaches 25% at one year.3 An excess mortality risk persists for at least 10 years.4 Of the patients who survive six months, only 50% can perform their ADLs, and only 25% can perform their instrumental ADLs as well as they could prior to their fracture.5,6
Unsurprisingly, older adults with hip fractures are five times more likely to require nursing home placement at one year.5
Hospitalists frequently encounter patients with hip fractures in the perioperative setting. Given their close collaboration with orthopedic surgeons and emphasis on transitions of care, hospitalists can play an important role in reversing the trajectory of death and disability following hip fractures. Key aspects of inpatient management are outlined below.
Hip Fracture Repair
Hip fractures can be divided into intracapsular (femoral neck) or extracapsular (intratrochanteric or subtrochanteric) fractures. Their relative frequencies are listed in Table 1.7
Surgery types. Femoral neck fractures typically are the most difficult to heal, given a limited regional blood supply.5,7 Displaced femoral neck fractures require either a hemiarthroplasty or total hip arthroplasty. Over time, hemiarthroplasties tend to cause hip pain from acetabular erosion, so they are better suited for less active, elderly patients. Nondisplaced femoral neck, intratrochanteric, and subtrochanteric fractures are usually managed with open reduction and internal fixation.
The overall goal of surgery is to return patients to their prior level of functioning. In the short term, surgery also provides pain relief and allows for early mobilization. Nonoperative management is generally reserved for patients with very high operative risk or limited life expectancies or those who are bedridden at baseline.
Timing of surgery. In general, hip fracture repair should be performed within 24-48 hours of admission in patients who are medically stable. Though early surgery may not improve functional outcomes or mortality, it has been associated with improved pain control, decreased length of stay, and fewer major complications.8 Patients with active medical conditions (e.g. pneumonia) should be medically optimized before proceeding with surgery. A 2011 study found that most of the excess in-hospital mortality associated with surgical delays beyond five days was attributable to the active medical issues rather than to the delay itself.9
Prevention of Perioperative Complications
The principles of geriatric medicine should be applied to the care of elderly patients with hip fractures. Emphasis should be placed on early recognition of treatable conditions and avoidance of iatrogenesis. Careful assessment of medical problems, social support, and functional status within an interdisciplinary framework is recommended. Such a multi-faceted approach has been shown to reduce overall complications in hip fracture patients.10 Specific complications are discussed in more detail below.
Delirium. Delirium is the most common complication after hip fracture surgery, with a prevalence of 35%-65%.7 Proper pain control, minimization of polypharmacy, removal of tethers, and frequent reorientation are among the many preventive measures that should be implemented.
Venous thromboembolism (VTE). VTE is a leading cause of morbidity and mortality for hip fracture patients.11 Without prophylaxis, about 1.8% will develop symptomatic deep venous thromboses, and 1% will develop symptomatic pulmonary emboli in the first seven to 14 days after surgery. An estimated 4.3% will develop symptomatic VTE in the first 35 days after surgery.
The American College of Chest Physicians recommends that patients undergoing hip fracture surgery receive VTE prophylaxis for a minimum of 10-14 days postoperatively.11 Extending prophylaxis out to 35 days is reasonable. Low molecular-weight heparin is preferred over low-dose unfractionated heparin, fondaparinux, warfarin, and aspirin. Patients should receive preoperative VTE prophylaxis if surgery is delayed.
Postoperative infections. Urinary tract infections (UTIs) are the most common infectious complication after hip fracture surgery.7 If not caught early, they can result in urosepsis, prosthetic joint infections, and death. After the first 48 hours of urinary catheterization, the risk of a UTI is 5%-10% per day.12
Therefore, catheters should be removed within 24-48 hours of surgery.
Acute blood loss anemia. Anemia is common in hip fracture patients. It may be present on admission or develop as a result of intraoperative blood loss, ongoing drain output, or fluid resuscitation.
The recent FOCUS trial, which helped to clarify the optimal transfusion threshold for patients after hip fracture surgery, compared a liberal versus restrictive transfusion strategy in patients with cardiovascular disease.13 Transfusing for a hemoglobin < 10 g/dL, as opposed to transfusing for symptoms or a hemoglobin < 8 g/dL, did not improve mortality, in-hospital morbidity (including myocardial infarction), or functional status at 60 days.
Pressure ulcers. Patients with hip fractures are at risk of developing decubitus ulcers. One study found the incidence of new pressure ulcers to be 16% at seven days and 36% at 32 days after initial hospitalization.14 Multicomponent interventions have been shown to successfully reduce the rate of hospital-acquired pressure ulcers.15
Medical Management of Osteoporosis
The World Heath Organization defines osteoporosis as a bone mineral density of at least 2.5 standard deviations below that of a “normal” young adult as measured on DEXA scan, or a T-score ≤ -2.5.16 However, it is important to recognize that bone strength depends not only on the quantity of bone but also on the quality. Any patient who sustains a hip fracture with minimal trauma (e.g. a fall from standing height) should be considered to have osteoporosis, regardless of T-score.
Patients with their first hip fracture are 2.5 times more likely to have a future fragility fracture.17 Hospitalists must therefore make secondary prevention a priority. Medical management focuses on maintaining bone strength, slowing further bone loss, and preventing future falls.
Evaluation. A directed history and physical examination should be completed to screen for secondary causes of osteoporosis. A basic laboratory workup is reasonable in the inpatient setting (see Table 2).17 Other tests, such as a serum and urine protein electrophoresis, can be obtained as clinically indicated.
Patients require counseling directed at lifestyle factors, including the importance of weight-bearing exercise, smoking cessation, and avoidance of excessive alcohol intake. A comprehensive falls assessment is also warranted.
Treatment. All patients with hip fractures should be discharged from the hospital on calcium and vitamin D supplementation, unless there is a specific contraindication.18 Guidelines vary by organization, but the National Osteoporosis Foundation’s recommendations are listed in Table 3.17,19,20 Dietary calcium is usually insufficient to meet the daily requirement.
Bisphosphonates are considered first-line therapy for osteoporosis.17 The HORIZON trial was a randomized, placebo-controlled study that evaluated annual zoledronic acid infusions in hip fracture patients who were intolerant of oral bisphosphonates.21 Zoledronic acid reduced the rate of new fractures by 35% after 1.9 years, with a number needed to treat (NNT) of 19. It also improved survival by 28%, for an NNT of 27. All subjects also received calcium and vitamin D supplementation.
Both hospitalists and orthopedists might worry about bisphosphonates adversely affecting bone healing in the acute setting. Subsequent analyses from the HORIZON trial suggest that bisphosphonates can be safely started as soon as two weeks after surgery.22,23
Transitions of care. Despite well-established guidelines for the treatment of osteoporosis, patients with hip fractures often are undertreated. A retrospective study of 420 acute hip fracture patients found that only 37% received calcium, 36% received vitamin D, and 31% received a bisphosphonate on discharge.24 A prospective study of 1,075 women with new osteoporotic fractures found that only 17% had started anti-osteoporosis medications at one year.25
Hospitalists should recognize and address potential barriers to appropriate medical therapy. Patient-related obstacles may include the cost of medications, concerns about side effects, and lack of a PCP.24,25 Hospitalists should document the diagnosis of osteoporosis in the medical record so subsequent providers are attuned to the issue.26 They should also clarify the ownership of osteoporosis across the continuum of care, because medicine consultants, orthopedists, primary care or rehabilitation physicians, and subspecialists may all be involved. Hospitalists can certainly take advantage of this window of opportunity by starting patients on osteoporosis treatment and ensuring smooth transitions of care on discharge.
Back to the Case
The patient was started on intravenous antibiotics for healthcare-associated pneumonia with improvement of his oxygen requirement to 3 L/min. He underwent a right hemiarthroplasty on hospital day five and tolerated the procedure well. His delirium resolved with treatment of his infection, pain control, discontinuation of lorazepam, and other conservative measures. He was given VTE prophylaxis pre- and postoperatively. His urinary catheter was discontinued on day one after surgery. He was started on calcium supplementation and vitamin D repletion after his 25-OH vitamin D level returned low at 14 ng/mL.
The patient progressed well with physical and occupational therapy and was discharged back to his skilled nursing facility, with plans to start a bisphosphonate in two weeks.
Bottom Line
Hospitalists should be familiar with the best practices for the perioperative management of hip fracture patients.
Dr. Anderson and Dr. Wolfe are hospitalists at the University of Colorado Hospital in Aurora. Dr. Anderson directs the medicine consult service, and Dr. Wolfe serves as the associate director.
References
- The Joint Commission. Improving and measuring osteoporosis treatment 2008. Oakbrook Terrace, Il; 2008. Available at: http://www.jointcommission.org/improving_and_measuring_osteoporosis_management/. Accessed September 29, 2013.
- Stevens JA, Rudd RA. The impact of decreasing U.S. hip fracture rates on future hip fracture estimates. Osteoporos Int. 2013;24(10):2725-2728.
- Braithwaite RS, Col NF, Wong JB. Estimating hip fracture morbidity, mortality, and costs. J Am Geriatr Soc. 2003;51(3):364-370. Haentjens P, Magaziner J, Colón-Emeric CS, et al.
- Meta-analysis: excess mortality after hip fracture among older women and men. Ann Intern Med. 2010;152(6):380-390.
- Hung WW, Egol KA, Zuckerman JD, Siu AL. Hip fracture management: tailoring care for the older patient. JAMA. 2012;307(20):2185-2194.
- Magaziner J, Simonsick EM, Kashner TM, Hebel JR, Kenzora JE. Predictors of functional recovery one year following hospital discharge for hip fracture: a prospective study. J Gerontol. 1990;45(3):M101-107.
- Bateman L, Vuppala S, Porada P, et al. Medical management in the acute hip fracture patient: a comprehensive review for the internist. Ochsner J. 2012;12(2):101-110.
- Orosz GM, Magaziner J, Hannan EL, et al. Association of timing of surgery for hip fracture and patient outcomes. JAMA. 2004;291(14):1738-1743.
- Vidán MT, Sánchez E, Gracia Y, Marañón E, Vaquero J, Serra JA. Causes and effects of surgical delay in patients with hip fracture: a cohort study. Ann Intern Med. 2011;155(4):226-233.
- Vidán M, Serra JA, Moreno C, Riquelme G, Ortiz J. Efficacy of a comprehensive geriatric intervention in older patients hospitalized for hip fracture: a randomized, controlled trial. J Am Geriatr Soc. 2005;53(9):1476-1482.
- Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e278S-e325S.
- Wald H, Epstein A, Kramer A. Extended use of indwelling urinary catheters in postoperative hip fracture patients. Med Care. 2005;43(10):1009-1017.
- Carson JL, Terrin ML, Noveck H, et al. Liberal or restrictive transfusion in high-risk patients after hip surgery. N Engl J Med. 2011;365(26):2453-2462.
- Baumgarten M, Margolis DJ, Orwig DL, et al. Pressure ulcers in elderly patients with hip fracture across the continuum of care. J Am Geriatr Soc. 2009;57(5):863-870.
- Sullivan N, Schoelles KM. Preventing in-facility pressure ulcers as a patient safety strategy: a systematic review. Ann Intern Med. 2013;158(5 Pt 2):410-416.
- World Health Organization. Prevention and management of osteoporosis: Report of a WHO scientific group 2000. Geneva, Switzerland; 2000. WHO technical report series; 921. Available at: http://whqlibdoc.who.int/trs/who_trs_921.pdf. Accessed July 9, 2013.
- National Osteoporosis Foundation. Clinician’s guide to prevention and treatment of osteoporosis 2013. Washington, D.C.; 2013. Available at: www.nof.org/files/nof/public/content/file/917/upload/481.pdf. Accessed September 29, 2013.
- Tang BM, Eslick GD, Nowson C, Smith C, Bensoussan A. Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet. 2007;370(9588):657-666.
- Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911-1930.
- Ross AC, Taylor CL, Yaktine AL, Del Valle HB, eds. Dietary reference intakes for calcium and vitamin D. Washington, D.C.: National Academies Press; 2011. Lyles KW, Colón-Emeric CS, Magaziner JS, et al.
- Zoledronic acid in reducing clinical fracture and mortality after hip fracture. N Engl J Med. 2007;357: nihpa40967.
- Colón-Emeric CS, Nordsletten L, Olson S, et al. Association between timing of zoledronic acid infusion and hip fracture healing. Osteoporos Int. 2011;22(8):2329-2336.
- Eriksen EF, Lyles KW, Colón-Emeric CS, et al. Antifracture efficacy and reduction of mortality in relation to timing of the first dose of zoledronic acid after hip fracture. J Bone Miner Res. 2009;24(7):1308-1313.
- Byszewski A, Lemay G, Molnar F, Azad N, McMartin SE. Closing the osteoporosis care gap in hip fracture patients: an opportunity to decrease recurrent fractures and hospital admissions. J Osteoporos. 2011;2011:404969.
- Greenspan SL, Wyman, A, Hoovan FH, et al. Predictors of treatment with osteoporosis medications after recent fragility fractures in a multinational cohort of postmenopausal women. J Am Geriatr Soc. 2012;60(3):455-461.
- Kamel HK, Hussain MS, Tariq S, Perry HM, Morley JE. Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture. Am J Med. 2000;109(4):326-328.
Blood Culture for Uncomplicated SSTI Not Helpful with Bacteriologic Diagnosis
Clinical question: What is the yield of blood cultures performed on pediatric patients admitted for uncomplicated and complicated skin and soft tissue infections (SSTIs and cSSTIs)?
Background: SSTIs are a common cause of pediatric ED visits and hospitalizations. Current Infectious Diseases Society of America (IDSA) guidelines include obtaining a blood culture for patients who show signs of systemic toxicity. Blood cultures are performed frequently in all pediatric patients hospitalized for SSTI and cSSTI. Little recent data exists about the rate of bacteremia in pediatric SSTI since the widespread emergence of community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA) and adoption of routine vaccination for Haemophilus influenzae type B (HiB) and varicella.
Study design: Single-center, retrospective case series.
Setting: University-affiliated pediatric hospital at a tertiary medical center.
Synopsis: Researchers used the hospital’s electronic medical record system to search for patients between the ages of 0 and 18 years hospitalized for SSTI/cSSTI. Initial screening of the data utilized ICD-9-CM codes for cellulitis and abscess (682.X), with subsequent review by investigators to exclude miscoded cases, immunocompromised patients, hospital-acquired infection, and incidentally noted SSTI during admissions for other problems. SSTIs were classified as being complicated in the cases of surgical or traumatic wound infection, need for surgical intervention, and infected ulcers or burns. Routine incision and drainage did not constitute surgical intervention.
Of the 580 patients remaining, 482 were classified as having SSTI, of which 455 underwent testing with blood cultures. None of the blood cultures led to pathogenic bacterial growth after 120 hours of incubation; three grew S. epidermidis. Of the 98 patients classified as having cSSTI, 80 underwent blood culture testing, of which 10 (12.5%) were positive.
Pathogens identified in positive blood cultures included MRSA (6), methicillin-sensitive S. aureus (3), and S. pneumococcus (1). Length of stay was significantly longer for patients with SSTI who underwent blood culture testing (3.24 days) compared to those who did not (2.33 days).
Bottom line: Obtaining blood cultures in a child hospitalized with uncomplicated SSTI is highly unlikely to be helpful in obtaining a bacteriologic diagnosis. Even worse, it will likely increase the length of stay for these patients.
Citation: Malone JR, Durica SR, Thompson DM, Bogie A, Naifeh M. Blood cultures in the evaluation of uncomplicated skin and soft tissue infections. Pediatrics. 2013;132:454-459.
Reviewed by Pediatric Editor Weijen Chang, MD, SFHM, FAAP, associate clinical professor of medicine and pediatrics at the University of California at San Diego School of Medicine, and a hospitalist at both UCSD Medical Center and Rady Children’s Hospital.
Clinical question: What is the yield of blood cultures performed on pediatric patients admitted for uncomplicated and complicated skin and soft tissue infections (SSTIs and cSSTIs)?
Background: SSTIs are a common cause of pediatric ED visits and hospitalizations. Current Infectious Diseases Society of America (IDSA) guidelines include obtaining a blood culture for patients who show signs of systemic toxicity. Blood cultures are performed frequently in all pediatric patients hospitalized for SSTI and cSSTI. Little recent data exists about the rate of bacteremia in pediatric SSTI since the widespread emergence of community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA) and adoption of routine vaccination for Haemophilus influenzae type B (HiB) and varicella.
Study design: Single-center, retrospective case series.
Setting: University-affiliated pediatric hospital at a tertiary medical center.
Synopsis: Researchers used the hospital’s electronic medical record system to search for patients between the ages of 0 and 18 years hospitalized for SSTI/cSSTI. Initial screening of the data utilized ICD-9-CM codes for cellulitis and abscess (682.X), with subsequent review by investigators to exclude miscoded cases, immunocompromised patients, hospital-acquired infection, and incidentally noted SSTI during admissions for other problems. SSTIs were classified as being complicated in the cases of surgical or traumatic wound infection, need for surgical intervention, and infected ulcers or burns. Routine incision and drainage did not constitute surgical intervention.
Of the 580 patients remaining, 482 were classified as having SSTI, of which 455 underwent testing with blood cultures. None of the blood cultures led to pathogenic bacterial growth after 120 hours of incubation; three grew S. epidermidis. Of the 98 patients classified as having cSSTI, 80 underwent blood culture testing, of which 10 (12.5%) were positive.
Pathogens identified in positive blood cultures included MRSA (6), methicillin-sensitive S. aureus (3), and S. pneumococcus (1). Length of stay was significantly longer for patients with SSTI who underwent blood culture testing (3.24 days) compared to those who did not (2.33 days).
Bottom line: Obtaining blood cultures in a child hospitalized with uncomplicated SSTI is highly unlikely to be helpful in obtaining a bacteriologic diagnosis. Even worse, it will likely increase the length of stay for these patients.
Citation: Malone JR, Durica SR, Thompson DM, Bogie A, Naifeh M. Blood cultures in the evaluation of uncomplicated skin and soft tissue infections. Pediatrics. 2013;132:454-459.
Reviewed by Pediatric Editor Weijen Chang, MD, SFHM, FAAP, associate clinical professor of medicine and pediatrics at the University of California at San Diego School of Medicine, and a hospitalist at both UCSD Medical Center and Rady Children’s Hospital.
Clinical question: What is the yield of blood cultures performed on pediatric patients admitted for uncomplicated and complicated skin and soft tissue infections (SSTIs and cSSTIs)?
Background: SSTIs are a common cause of pediatric ED visits and hospitalizations. Current Infectious Diseases Society of America (IDSA) guidelines include obtaining a blood culture for patients who show signs of systemic toxicity. Blood cultures are performed frequently in all pediatric patients hospitalized for SSTI and cSSTI. Little recent data exists about the rate of bacteremia in pediatric SSTI since the widespread emergence of community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA) and adoption of routine vaccination for Haemophilus influenzae type B (HiB) and varicella.
Study design: Single-center, retrospective case series.
Setting: University-affiliated pediatric hospital at a tertiary medical center.
Synopsis: Researchers used the hospital’s electronic medical record system to search for patients between the ages of 0 and 18 years hospitalized for SSTI/cSSTI. Initial screening of the data utilized ICD-9-CM codes for cellulitis and abscess (682.X), with subsequent review by investigators to exclude miscoded cases, immunocompromised patients, hospital-acquired infection, and incidentally noted SSTI during admissions for other problems. SSTIs were classified as being complicated in the cases of surgical or traumatic wound infection, need for surgical intervention, and infected ulcers or burns. Routine incision and drainage did not constitute surgical intervention.
Of the 580 patients remaining, 482 were classified as having SSTI, of which 455 underwent testing with blood cultures. None of the blood cultures led to pathogenic bacterial growth after 120 hours of incubation; three grew S. epidermidis. Of the 98 patients classified as having cSSTI, 80 underwent blood culture testing, of which 10 (12.5%) were positive.
Pathogens identified in positive blood cultures included MRSA (6), methicillin-sensitive S. aureus (3), and S. pneumococcus (1). Length of stay was significantly longer for patients with SSTI who underwent blood culture testing (3.24 days) compared to those who did not (2.33 days).
Bottom line: Obtaining blood cultures in a child hospitalized with uncomplicated SSTI is highly unlikely to be helpful in obtaining a bacteriologic diagnosis. Even worse, it will likely increase the length of stay for these patients.
Citation: Malone JR, Durica SR, Thompson DM, Bogie A, Naifeh M. Blood cultures in the evaluation of uncomplicated skin and soft tissue infections. Pediatrics. 2013;132:454-459.
Reviewed by Pediatric Editor Weijen Chang, MD, SFHM, FAAP, associate clinical professor of medicine and pediatrics at the University of California at San Diego School of Medicine, and a hospitalist at both UCSD Medical Center and Rady Children’s Hospital.
Applications Being Accepted for SHM Fellows Program
Fellows Deadline Is Jan. 10, 2014
Thousands of hospitalists across the country have earned the Fellow in Hospital Medicine (FHM) and Senior Fellow in Hospital Medicine (SFHM) designation to set themselves apart in the specialty—and among other hospital-based caregivers.
For the second year, nurse practitioners and physician assistants can also earn the FHM and SFHM designations.
Candidates for FHM and SFHM can now apply online. For more information or to apply, visit www.hospitalmedicine.org/fellows.
Fellows Deadline Is Jan. 10, 2014
Thousands of hospitalists across the country have earned the Fellow in Hospital Medicine (FHM) and Senior Fellow in Hospital Medicine (SFHM) designation to set themselves apart in the specialty—and among other hospital-based caregivers.
For the second year, nurse practitioners and physician assistants can also earn the FHM and SFHM designations.
Candidates for FHM and SFHM can now apply online. For more information or to apply, visit www.hospitalmedicine.org/fellows.
Fellows Deadline Is Jan. 10, 2014
Thousands of hospitalists across the country have earned the Fellow in Hospital Medicine (FHM) and Senior Fellow in Hospital Medicine (SFHM) designation to set themselves apart in the specialty—and among other hospital-based caregivers.
For the second year, nurse practitioners and physician assistants can also earn the FHM and SFHM designations.
Candidates for FHM and SFHM can now apply online. For more information or to apply, visit www.hospitalmedicine.org/fellows.
Submit Your HM14 Scientific Abstracts Soon
HM14: Submit Your Scientific Abstracts Soon
Abstracts are being accepted for SHM’s scientific poster and oral abstract competition, known as Research, Innovations, and Clinical Vignettes (RIV), until Sunday, Dec. 1. Visit the Academic Community page on the SHM website, or, for a full suite of resources for submitting your abstract, go directly to the abstract submission site (https://shm.confex.com/shm/HM14/cfp.cgi) to submit your abstract today.
For more on HM14, the biggest meeting in hospital medicine, visit www.hospitalmedicine2014.org.
Brendon Shank is SHM’s associate vice president of communications.
HM14: Submit Your Scientific Abstracts Soon
Abstracts are being accepted for SHM’s scientific poster and oral abstract competition, known as Research, Innovations, and Clinical Vignettes (RIV), until Sunday, Dec. 1. Visit the Academic Community page on the SHM website, or, for a full suite of resources for submitting your abstract, go directly to the abstract submission site (https://shm.confex.com/shm/HM14/cfp.cgi) to submit your abstract today.
For more on HM14, the biggest meeting in hospital medicine, visit www.hospitalmedicine2014.org.
Brendon Shank is SHM’s associate vice president of communications.
HM14: Submit Your Scientific Abstracts Soon
Abstracts are being accepted for SHM’s scientific poster and oral abstract competition, known as Research, Innovations, and Clinical Vignettes (RIV), until Sunday, Dec. 1. Visit the Academic Community page on the SHM website, or, for a full suite of resources for submitting your abstract, go directly to the abstract submission site (https://shm.confex.com/shm/HM14/cfp.cgi) to submit your abstract today.
For more on HM14, the biggest meeting in hospital medicine, visit www.hospitalmedicine2014.org.
Brendon Shank is SHM’s associate vice president of communications.
Concern about Copper's Effectiveness in Preventing Hospital-Acquired Infections
Karen Appold’s cover story, “Copper,” in the September 2013 issue, offers an exciting and encouraging development in the struggle to prevent hospital-acquired infections, but I have two concerns. As copper tarnishes, it forms a surface patina of copper hydroxide and copper carbonate. Would this patina act as a physical barrier, preventing bacteria from coming into contact with elemental copper and inhibiting the antimicrobial effect? If so, the obvious solution is to polish the surface frequently enough to prevent tarnishing.
The second concern regards the use of copper-nickel alloys. Many people are sensitive to nickel, [with reactions that] usually manifest as contact dermatitis. A study by the North American Contact Dermatitis Group (NACDG), conducted between 1992-2004 and involving 25,626 patients who were patch-tested, showed a prevalence of nickel sensitivity of 18.8% in 2004, increased from 14.5% in 1992.1
With a current U.S. population of approximately 317 million, a prevalence of 18.8% would mean nearly 60 million people with nickel sensitivity. Extrapolating from the NACDG study, the rate is probably actually higher. Medical devices made with copper-nickel alloys that contact the patient’s skin would cause contact dermatitis, and implanted devices would have the potential for more severe allergic reactions.
I simply urge foresight and caution in the use of various copper alloys for medical applications.
Rod Duraski, MD, MBA, FACP, medical director, WGH Hospital Medicine, LaGrange, Ga.
Reference
Karen Appold’s cover story, “Copper,” in the September 2013 issue, offers an exciting and encouraging development in the struggle to prevent hospital-acquired infections, but I have two concerns. As copper tarnishes, it forms a surface patina of copper hydroxide and copper carbonate. Would this patina act as a physical barrier, preventing bacteria from coming into contact with elemental copper and inhibiting the antimicrobial effect? If so, the obvious solution is to polish the surface frequently enough to prevent tarnishing.
The second concern regards the use of copper-nickel alloys. Many people are sensitive to nickel, [with reactions that] usually manifest as contact dermatitis. A study by the North American Contact Dermatitis Group (NACDG), conducted between 1992-2004 and involving 25,626 patients who were patch-tested, showed a prevalence of nickel sensitivity of 18.8% in 2004, increased from 14.5% in 1992.1
With a current U.S. population of approximately 317 million, a prevalence of 18.8% would mean nearly 60 million people with nickel sensitivity. Extrapolating from the NACDG study, the rate is probably actually higher. Medical devices made with copper-nickel alloys that contact the patient’s skin would cause contact dermatitis, and implanted devices would have the potential for more severe allergic reactions.
I simply urge foresight and caution in the use of various copper alloys for medical applications.
Rod Duraski, MD, MBA, FACP, medical director, WGH Hospital Medicine, LaGrange, Ga.
Reference
Karen Appold’s cover story, “Copper,” in the September 2013 issue, offers an exciting and encouraging development in the struggle to prevent hospital-acquired infections, but I have two concerns. As copper tarnishes, it forms a surface patina of copper hydroxide and copper carbonate. Would this patina act as a physical barrier, preventing bacteria from coming into contact with elemental copper and inhibiting the antimicrobial effect? If so, the obvious solution is to polish the surface frequently enough to prevent tarnishing.
The second concern regards the use of copper-nickel alloys. Many people are sensitive to nickel, [with reactions that] usually manifest as contact dermatitis. A study by the North American Contact Dermatitis Group (NACDG), conducted between 1992-2004 and involving 25,626 patients who were patch-tested, showed a prevalence of nickel sensitivity of 18.8% in 2004, increased from 14.5% in 1992.1
With a current U.S. population of approximately 317 million, a prevalence of 18.8% would mean nearly 60 million people with nickel sensitivity. Extrapolating from the NACDG study, the rate is probably actually higher. Medical devices made with copper-nickel alloys that contact the patient’s skin would cause contact dermatitis, and implanted devices would have the potential for more severe allergic reactions.
I simply urge foresight and caution in the use of various copper alloys for medical applications.
Rod Duraski, MD, MBA, FACP, medical director, WGH Hospital Medicine, LaGrange, Ga.
Reference
Four Recommendations to Help Hospitalists Fight Antimicrobial Resistance
Prevent infections. This might be the most obvious way to fight antibiotic-resistance—if there’s no infection, there is no need to worry about one that can’t be treated. Hospitalists can help prevent infection by quickly and effectively treating those who are infected to prevent the spread, washing hands, and promoting effective cleaning habits.
Tracking. The CDC has programs to gather information on antibiotic-resistant infections, causes of infections, and risk factors for infections. With this information, hospitalists can stay aware of the threats. They can also help by remaining vigilant about signs of new resistance and helping to get that information to the CDC.
The CDC is now working on a new module that will collect antimicrobial-susceptibility data that’s generated in hospital labs, Dr. Patel says.
“This will be compiled in a national database and then made available to state and local public health departments that could track antimicrobial resistance trends in their own state,” she says. “We hope those data will then be used to identify new trends in anti-microbial resistance and used to strategize how to prevent resistance from being transmitted locally.”
Antibiotic stewardship. The CDC says prescribing antibiotics only when necessary and tailoring treatment as narrowly as possible might be the most important step in fighting antimicrobial resistance. The CDC estimates that up to half of antibiotic use in humans is unnecessary.
The CDC is working to capture data on antibiotic use in healthcare settings, which will be used for benchmarking antibiotic use among different institutions and regions.
“I think this additional information will really help healthcare institutions measure how well antibiotics are being used in their institutions and make appropriate adjustments,” Dr. Patel says.
New drugs and diagnostic tests. New antibiotics will be needed because, while resistance can be slowed, it cannot be stopped. However, the number of New Drug Application approvals for antibiotics has fallen drastically—nearly 20 from 1980 to 1984, but fewer than five from 2005 to 2012, according to the CDC report.
Prevent infections. This might be the most obvious way to fight antibiotic-resistance—if there’s no infection, there is no need to worry about one that can’t be treated. Hospitalists can help prevent infection by quickly and effectively treating those who are infected to prevent the spread, washing hands, and promoting effective cleaning habits.
Tracking. The CDC has programs to gather information on antibiotic-resistant infections, causes of infections, and risk factors for infections. With this information, hospitalists can stay aware of the threats. They can also help by remaining vigilant about signs of new resistance and helping to get that information to the CDC.
The CDC is now working on a new module that will collect antimicrobial-susceptibility data that’s generated in hospital labs, Dr. Patel says.
“This will be compiled in a national database and then made available to state and local public health departments that could track antimicrobial resistance trends in their own state,” she says. “We hope those data will then be used to identify new trends in anti-microbial resistance and used to strategize how to prevent resistance from being transmitted locally.”
Antibiotic stewardship. The CDC says prescribing antibiotics only when necessary and tailoring treatment as narrowly as possible might be the most important step in fighting antimicrobial resistance. The CDC estimates that up to half of antibiotic use in humans is unnecessary.
The CDC is working to capture data on antibiotic use in healthcare settings, which will be used for benchmarking antibiotic use among different institutions and regions.
“I think this additional information will really help healthcare institutions measure how well antibiotics are being used in their institutions and make appropriate adjustments,” Dr. Patel says.
New drugs and diagnostic tests. New antibiotics will be needed because, while resistance can be slowed, it cannot be stopped. However, the number of New Drug Application approvals for antibiotics has fallen drastically—nearly 20 from 1980 to 1984, but fewer than five from 2005 to 2012, according to the CDC report.
Prevent infections. This might be the most obvious way to fight antibiotic-resistance—if there’s no infection, there is no need to worry about one that can’t be treated. Hospitalists can help prevent infection by quickly and effectively treating those who are infected to prevent the spread, washing hands, and promoting effective cleaning habits.
Tracking. The CDC has programs to gather information on antibiotic-resistant infections, causes of infections, and risk factors for infections. With this information, hospitalists can stay aware of the threats. They can also help by remaining vigilant about signs of new resistance and helping to get that information to the CDC.
The CDC is now working on a new module that will collect antimicrobial-susceptibility data that’s generated in hospital labs, Dr. Patel says.
“This will be compiled in a national database and then made available to state and local public health departments that could track antimicrobial resistance trends in their own state,” she says. “We hope those data will then be used to identify new trends in anti-microbial resistance and used to strategize how to prevent resistance from being transmitted locally.”
Antibiotic stewardship. The CDC says prescribing antibiotics only when necessary and tailoring treatment as narrowly as possible might be the most important step in fighting antimicrobial resistance. The CDC estimates that up to half of antibiotic use in humans is unnecessary.
The CDC is working to capture data on antibiotic use in healthcare settings, which will be used for benchmarking antibiotic use among different institutions and regions.
“I think this additional information will really help healthcare institutions measure how well antibiotics are being used in their institutions and make appropriate adjustments,” Dr. Patel says.
New drugs and diagnostic tests. New antibiotics will be needed because, while resistance can be slowed, it cannot be stopped. However, the number of New Drug Application approvals for antibiotics has fallen drastically—nearly 20 from 1980 to 1984, but fewer than five from 2005 to 2012, according to the CDC report.
Hospitalists Poised to Prevent, Combat Antibiotic-Resistant Pathogens
Describing formally for the first time the enormity of the problem of antibiotic resistance and warning of the “potentially catastrophic consequences of inaction,” the Centers for Disease Control and Prevention (CDC) announced in September that more than two million people a year are sickened by infections that are resistant to treatment with antibiotics.
Moreover, the CDC says 23,000 people die as a result.
And because those numbers are based only on the data available—and the agency assumes that many infections are not captured—the CDC says its estimate is a conservative one and the real number is probably higher.
The report is a call to action for hospitalists, who are in an almost ideal position to participate in efforts to prevent infections and control their spread once they’re discovered, says Jean Patel, PhD, deputy director of the office of antimicrobial resistance at the CDC.
“I think it’s a sobering number, and it indicates how far we have to go in combating this problem of antimicrobial resistance,” Dr. Patel says.
The medical community, she adds, cannot expect that new treatments will become available to fight all of these new infections.
“All of the drugs also are going to have some gaps in their range of activity, so there’s no drug coming that’s going to be effective against all the antimicrobial-resistant drugs that we face today,” Dr. Patel explains. “For that reason, we’re sounding the alarm that it’s important to pay attention to infection control and antibiotic stewardship practices.”
The report, “Antibiotic Resistance Threats to the United States, 2013,” creates three categories of antibiotic-resistant pathogens. In the “urgent” tier are Clostridium difficile, which the CDC estimates is responsible for 250,000 infections a year and 14,000 deaths; carbapenem-resistant Enterobacteriaceae, estimated to be responsible for 9,000 drug-resistant infections a year and 600 deaths; and drug-resistant Neisseria gonorrhoeae, at 246,000 drug-resistant infections.
These bacteria are considered an “immediate public health threat that requires urgent and aggressive action.”
There are 12 pathogens in the second category, described as “a serious concern” requiring “prompt and sustained action to ensure the problem does not grow.”
Of particular interest to hospitalists in this group, Dr. Patel says, is methicillin-resistant Staphylococcus aureus (MRSA). The CDC estimates that more than 80,000 severe MRSA infections and more than 11,000 deaths occur in the U.S. every year.
MRSA was not ranked as an “urgent” threat only because the number of infections is actually decreasing, especially in healthcare institutions, and because there are antibiotics that still work on MRSA.
“If either of those things were to change—for example, if the rate of infections were to increase, or if these isolates were to become more resistant—then we would have to think about changing this from a serious threat to an urgent threat,” Dr. Patel says.
Another infection in the serious category that should be on hospitalists’ radar is drug-resistant Streptococcus pneumoniae. A new vaccine is helping to decrease the number of these infections, but hospitalists should be vigilant about infections that could escape the vaccine and become resistant, Dr. Patel says.
The report estimates as much as $20 billion in excess healthcare costs due to antimicrobial-resistant infections, with $35 billion in lost productivity in 2008 dollars.1
Ketino Kobaidze, MD, assistant professor at the Emory University School of Medicine in Atlanta and a member of the antimicrobial stewardship and infectious disease control committees at Emory University Hospital Midtown, says the sheer numbers are sure to get people to take notice.
“Two million is lots of patients,” she says. “It’s eye-opening, really, for many doctors and patients and society.”
The silver lining, she says, is that the field is moving toward diagnostic tools that will provide quick feedback on the type of infection at work.
It may be that hospitalists have no choice but to give an antibiotic to a patient because of the risk involved in not giving one; however, providers should quickly tailor that treatment to target the specific pathogen when more information is available.
—Ketino Kobaidze, MD, assistant professor, Emory University School of Medicine, Atlanta, member, antimicrobial stewardship and infectious disease control committees, Emory University Hospital Midtown
“The most important thing, I think, for hospital medicine and medicine anywhere, is to follow up with whatever you’re ordering and notice right away what happens with these tests. If it’s positive or negative, redirect your care,” Dr. Kobaidze says. “Time is really an important issue here.
“As hospitalists, we need to be extremely cautious not to give them something they don’t need.”
Dr. Kobaidze was particularly struck by gonorrhea being listed in the “urgent” threat category.
“It was so easy to treat before,” she says. “It was nothing, piece of cake. This makes me a little bit concerned.”
Robert Orenstein, DO, an infectious disease expert at Mayo Clinic, praises the report and says hospitalists have a key role to play.
“I think this has a clear impact on hospitalists, who are the primary caregivers of many of these ill patients,” he says. “We need to educate them and build systems that target antimicrobials to the infecting agents and limit their use. Hospitalists are also the people who can help protect patients from the spread of these in the hospital by following appropriate infection prevention guidelines and educating their colleagues of the importance of this.”
He also stresses the importance of being aware of threats within your specific region.
“Many of these MDROs [multi-drug resistant organisms] have regional prevalence,” he says. “And it’s important to know which bugs are in your region so you can work with your institution and public health to tackle these.”
Tom Collins is a freelance writer in South Florida.
Reference
Describing formally for the first time the enormity of the problem of antibiotic resistance and warning of the “potentially catastrophic consequences of inaction,” the Centers for Disease Control and Prevention (CDC) announced in September that more than two million people a year are sickened by infections that are resistant to treatment with antibiotics.
Moreover, the CDC says 23,000 people die as a result.
And because those numbers are based only on the data available—and the agency assumes that many infections are not captured—the CDC says its estimate is a conservative one and the real number is probably higher.
The report is a call to action for hospitalists, who are in an almost ideal position to participate in efforts to prevent infections and control their spread once they’re discovered, says Jean Patel, PhD, deputy director of the office of antimicrobial resistance at the CDC.
“I think it’s a sobering number, and it indicates how far we have to go in combating this problem of antimicrobial resistance,” Dr. Patel says.
The medical community, she adds, cannot expect that new treatments will become available to fight all of these new infections.
“All of the drugs also are going to have some gaps in their range of activity, so there’s no drug coming that’s going to be effective against all the antimicrobial-resistant drugs that we face today,” Dr. Patel explains. “For that reason, we’re sounding the alarm that it’s important to pay attention to infection control and antibiotic stewardship practices.”
The report, “Antibiotic Resistance Threats to the United States, 2013,” creates three categories of antibiotic-resistant pathogens. In the “urgent” tier are Clostridium difficile, which the CDC estimates is responsible for 250,000 infections a year and 14,000 deaths; carbapenem-resistant Enterobacteriaceae, estimated to be responsible for 9,000 drug-resistant infections a year and 600 deaths; and drug-resistant Neisseria gonorrhoeae, at 246,000 drug-resistant infections.
These bacteria are considered an “immediate public health threat that requires urgent and aggressive action.”
There are 12 pathogens in the second category, described as “a serious concern” requiring “prompt and sustained action to ensure the problem does not grow.”
Of particular interest to hospitalists in this group, Dr. Patel says, is methicillin-resistant Staphylococcus aureus (MRSA). The CDC estimates that more than 80,000 severe MRSA infections and more than 11,000 deaths occur in the U.S. every year.
MRSA was not ranked as an “urgent” threat only because the number of infections is actually decreasing, especially in healthcare institutions, and because there are antibiotics that still work on MRSA.
“If either of those things were to change—for example, if the rate of infections were to increase, or if these isolates were to become more resistant—then we would have to think about changing this from a serious threat to an urgent threat,” Dr. Patel says.
Another infection in the serious category that should be on hospitalists’ radar is drug-resistant Streptococcus pneumoniae. A new vaccine is helping to decrease the number of these infections, but hospitalists should be vigilant about infections that could escape the vaccine and become resistant, Dr. Patel says.
The report estimates as much as $20 billion in excess healthcare costs due to antimicrobial-resistant infections, with $35 billion in lost productivity in 2008 dollars.1
Ketino Kobaidze, MD, assistant professor at the Emory University School of Medicine in Atlanta and a member of the antimicrobial stewardship and infectious disease control committees at Emory University Hospital Midtown, says the sheer numbers are sure to get people to take notice.
“Two million is lots of patients,” she says. “It’s eye-opening, really, for many doctors and patients and society.”
The silver lining, she says, is that the field is moving toward diagnostic tools that will provide quick feedback on the type of infection at work.
It may be that hospitalists have no choice but to give an antibiotic to a patient because of the risk involved in not giving one; however, providers should quickly tailor that treatment to target the specific pathogen when more information is available.
—Ketino Kobaidze, MD, assistant professor, Emory University School of Medicine, Atlanta, member, antimicrobial stewardship and infectious disease control committees, Emory University Hospital Midtown
“The most important thing, I think, for hospital medicine and medicine anywhere, is to follow up with whatever you’re ordering and notice right away what happens with these tests. If it’s positive or negative, redirect your care,” Dr. Kobaidze says. “Time is really an important issue here.
“As hospitalists, we need to be extremely cautious not to give them something they don’t need.”
Dr. Kobaidze was particularly struck by gonorrhea being listed in the “urgent” threat category.
“It was so easy to treat before,” she says. “It was nothing, piece of cake. This makes me a little bit concerned.”
Robert Orenstein, DO, an infectious disease expert at Mayo Clinic, praises the report and says hospitalists have a key role to play.
“I think this has a clear impact on hospitalists, who are the primary caregivers of many of these ill patients,” he says. “We need to educate them and build systems that target antimicrobials to the infecting agents and limit their use. Hospitalists are also the people who can help protect patients from the spread of these in the hospital by following appropriate infection prevention guidelines and educating their colleagues of the importance of this.”
He also stresses the importance of being aware of threats within your specific region.
“Many of these MDROs [multi-drug resistant organisms] have regional prevalence,” he says. “And it’s important to know which bugs are in your region so you can work with your institution and public health to tackle these.”
Tom Collins is a freelance writer in South Florida.
Reference
Describing formally for the first time the enormity of the problem of antibiotic resistance and warning of the “potentially catastrophic consequences of inaction,” the Centers for Disease Control and Prevention (CDC) announced in September that more than two million people a year are sickened by infections that are resistant to treatment with antibiotics.
Moreover, the CDC says 23,000 people die as a result.
And because those numbers are based only on the data available—and the agency assumes that many infections are not captured—the CDC says its estimate is a conservative one and the real number is probably higher.
The report is a call to action for hospitalists, who are in an almost ideal position to participate in efforts to prevent infections and control their spread once they’re discovered, says Jean Patel, PhD, deputy director of the office of antimicrobial resistance at the CDC.
“I think it’s a sobering number, and it indicates how far we have to go in combating this problem of antimicrobial resistance,” Dr. Patel says.
The medical community, she adds, cannot expect that new treatments will become available to fight all of these new infections.
“All of the drugs also are going to have some gaps in their range of activity, so there’s no drug coming that’s going to be effective against all the antimicrobial-resistant drugs that we face today,” Dr. Patel explains. “For that reason, we’re sounding the alarm that it’s important to pay attention to infection control and antibiotic stewardship practices.”
The report, “Antibiotic Resistance Threats to the United States, 2013,” creates three categories of antibiotic-resistant pathogens. In the “urgent” tier are Clostridium difficile, which the CDC estimates is responsible for 250,000 infections a year and 14,000 deaths; carbapenem-resistant Enterobacteriaceae, estimated to be responsible for 9,000 drug-resistant infections a year and 600 deaths; and drug-resistant Neisseria gonorrhoeae, at 246,000 drug-resistant infections.
These bacteria are considered an “immediate public health threat that requires urgent and aggressive action.”
There are 12 pathogens in the second category, described as “a serious concern” requiring “prompt and sustained action to ensure the problem does not grow.”
Of particular interest to hospitalists in this group, Dr. Patel says, is methicillin-resistant Staphylococcus aureus (MRSA). The CDC estimates that more than 80,000 severe MRSA infections and more than 11,000 deaths occur in the U.S. every year.
MRSA was not ranked as an “urgent” threat only because the number of infections is actually decreasing, especially in healthcare institutions, and because there are antibiotics that still work on MRSA.
“If either of those things were to change—for example, if the rate of infections were to increase, or if these isolates were to become more resistant—then we would have to think about changing this from a serious threat to an urgent threat,” Dr. Patel says.
Another infection in the serious category that should be on hospitalists’ radar is drug-resistant Streptococcus pneumoniae. A new vaccine is helping to decrease the number of these infections, but hospitalists should be vigilant about infections that could escape the vaccine and become resistant, Dr. Patel says.
The report estimates as much as $20 billion in excess healthcare costs due to antimicrobial-resistant infections, with $35 billion in lost productivity in 2008 dollars.1
Ketino Kobaidze, MD, assistant professor at the Emory University School of Medicine in Atlanta and a member of the antimicrobial stewardship and infectious disease control committees at Emory University Hospital Midtown, says the sheer numbers are sure to get people to take notice.
“Two million is lots of patients,” she says. “It’s eye-opening, really, for many doctors and patients and society.”
The silver lining, she says, is that the field is moving toward diagnostic tools that will provide quick feedback on the type of infection at work.
It may be that hospitalists have no choice but to give an antibiotic to a patient because of the risk involved in not giving one; however, providers should quickly tailor that treatment to target the specific pathogen when more information is available.
—Ketino Kobaidze, MD, assistant professor, Emory University School of Medicine, Atlanta, member, antimicrobial stewardship and infectious disease control committees, Emory University Hospital Midtown
“The most important thing, I think, for hospital medicine and medicine anywhere, is to follow up with whatever you’re ordering and notice right away what happens with these tests. If it’s positive or negative, redirect your care,” Dr. Kobaidze says. “Time is really an important issue here.
“As hospitalists, we need to be extremely cautious not to give them something they don’t need.”
Dr. Kobaidze was particularly struck by gonorrhea being listed in the “urgent” threat category.
“It was so easy to treat before,” she says. “It was nothing, piece of cake. This makes me a little bit concerned.”
Robert Orenstein, DO, an infectious disease expert at Mayo Clinic, praises the report and says hospitalists have a key role to play.
“I think this has a clear impact on hospitalists, who are the primary caregivers of many of these ill patients,” he says. “We need to educate them and build systems that target antimicrobials to the infecting agents and limit their use. Hospitalists are also the people who can help protect patients from the spread of these in the hospital by following appropriate infection prevention guidelines and educating their colleagues of the importance of this.”
He also stresses the importance of being aware of threats within your specific region.
“Many of these MDROs [multi-drug resistant organisms] have regional prevalence,” he says. “And it’s important to know which bugs are in your region so you can work with your institution and public health to tackle these.”
Tom Collins is a freelance writer in South Florida.
Reference
Hospitalists Should Take Wait-and-See Approach to Newly Approved Medications
Wait-and-See Approach Best for Newly Approved Meds
I am a new hospitalist, out of residency for two years, and feel very uncertain about using new or recently approved medications on my patients. Do you have any suggestions about how or when new medications should be used in practice?
–David Ray, MD
Dr. Hospitalist responds:
I certainly can understand your trepidation about using newly approved medications. Although our system of evaluating and approving medications for clinical use is considered the most rigorous in the world, 16 so-called novel medications were pulled from the shelves from 2000 to 2010, which equates to 6% of the total approved during that period. All in all, not a bad ratio, but the number of poor outcomes associated with a high-profile dud can be astronomical.
I think there are several major reasons why we have adverse issues with medications that have survived the rigors of the initial FDA approval process. First, many human drug trials are conducted in developing countries, where the human genome is much more homogenous and the liabilities for injuries are way less than in the U.S. Many researchers have acknowledged the significant role of pharmacogenomics, and how each physiology and pathology is unique. Couple these with the tendency to test drugs one at a time in younger cohorts—very few medications are administered in this manner in the U.S.—and one can quickly see how complex the equation becomes.
Another reason is the influence relegated to clinical trials. All clinicians should be familiar with the stages (0 to 4) and processes of how the FDA analyzes human drug trials. The FDA usually requires that two “adequate and well-controlled” trials confirm that a drug is safe and effective before it approves it for sale to the public. Once a drug completes Stage 3, an extensive statistical analysis is conducted to assure a drug’s demonstrated benefit is real and not the result of chance. But as it turns out, because the measured effects in most clinical trials are so small, chance is very hard to prove or disprove.
This was astutely demonstrated in a 2005 article published in the Journal of the American Medical Association (2005;294(2):218-228). John P. Ioannidis, MD, examined the results of 49 high-profile clinical-research studies in which 45 found that proposed intervention was effective. Of the 45 claiming effectiveness, seven (16%) were contradicted by subsequent studies, and seven others had found effects that were stronger than those of subsequent studies. Of the 26 randomly controlled trials that were followed up by larger trials, the initial finding was entirely contradicted in three cases (12%); another six cases (23%) found the benefit to be less than half of what had been initially reported.
In most instances, it wasn’t the therapy that changed but the sample size. In fact, many clinicians and biostatisticians believe many more so-called “evidence-based” practices or medicinals would be legitimately challenged if subjected to rigorous follow-up studies.
In my own personal experience as a hospitalist, I can think of two areas where the general medical community accepted initial studies only to refute them later: perioperative use of beta-blockers and inpatient glycemic control.
In light of the many high-profile medications that have been pulled from the market, I don’t like being in the first group to jump on the bandwagon. My general rule is to wait three to five years after a drug has been released before prescribing for patients. As always, there are exceptions. In instances where new medications have profound or life-altering potential (i.e. the new anticoagulants or gene-targeting meds for certain cancers) and the risks are substantiated, I’m all in!
Do you have a problem or concern that you’d like Dr. Hospitalist to address? Email your questions to [email protected].
Wait-and-See Approach Best for Newly Approved Meds
I am a new hospitalist, out of residency for two years, and feel very uncertain about using new or recently approved medications on my patients. Do you have any suggestions about how or when new medications should be used in practice?
–David Ray, MD
Dr. Hospitalist responds:
I certainly can understand your trepidation about using newly approved medications. Although our system of evaluating and approving medications for clinical use is considered the most rigorous in the world, 16 so-called novel medications were pulled from the shelves from 2000 to 2010, which equates to 6% of the total approved during that period. All in all, not a bad ratio, but the number of poor outcomes associated with a high-profile dud can be astronomical.
I think there are several major reasons why we have adverse issues with medications that have survived the rigors of the initial FDA approval process. First, many human drug trials are conducted in developing countries, where the human genome is much more homogenous and the liabilities for injuries are way less than in the U.S. Many researchers have acknowledged the significant role of pharmacogenomics, and how each physiology and pathology is unique. Couple these with the tendency to test drugs one at a time in younger cohorts—very few medications are administered in this manner in the U.S.—and one can quickly see how complex the equation becomes.
Another reason is the influence relegated to clinical trials. All clinicians should be familiar with the stages (0 to 4) and processes of how the FDA analyzes human drug trials. The FDA usually requires that two “adequate and well-controlled” trials confirm that a drug is safe and effective before it approves it for sale to the public. Once a drug completes Stage 3, an extensive statistical analysis is conducted to assure a drug’s demonstrated benefit is real and not the result of chance. But as it turns out, because the measured effects in most clinical trials are so small, chance is very hard to prove or disprove.
This was astutely demonstrated in a 2005 article published in the Journal of the American Medical Association (2005;294(2):218-228). John P. Ioannidis, MD, examined the results of 49 high-profile clinical-research studies in which 45 found that proposed intervention was effective. Of the 45 claiming effectiveness, seven (16%) were contradicted by subsequent studies, and seven others had found effects that were stronger than those of subsequent studies. Of the 26 randomly controlled trials that were followed up by larger trials, the initial finding was entirely contradicted in three cases (12%); another six cases (23%) found the benefit to be less than half of what had been initially reported.
In most instances, it wasn’t the therapy that changed but the sample size. In fact, many clinicians and biostatisticians believe many more so-called “evidence-based” practices or medicinals would be legitimately challenged if subjected to rigorous follow-up studies.
In my own personal experience as a hospitalist, I can think of two areas where the general medical community accepted initial studies only to refute them later: perioperative use of beta-blockers and inpatient glycemic control.
In light of the many high-profile medications that have been pulled from the market, I don’t like being in the first group to jump on the bandwagon. My general rule is to wait three to five years after a drug has been released before prescribing for patients. As always, there are exceptions. In instances where new medications have profound or life-altering potential (i.e. the new anticoagulants or gene-targeting meds for certain cancers) and the risks are substantiated, I’m all in!
Do you have a problem or concern that you’d like Dr. Hospitalist to address? Email your questions to [email protected].
Wait-and-See Approach Best for Newly Approved Meds
I am a new hospitalist, out of residency for two years, and feel very uncertain about using new or recently approved medications on my patients. Do you have any suggestions about how or when new medications should be used in practice?
–David Ray, MD
Dr. Hospitalist responds:
I certainly can understand your trepidation about using newly approved medications. Although our system of evaluating and approving medications for clinical use is considered the most rigorous in the world, 16 so-called novel medications were pulled from the shelves from 2000 to 2010, which equates to 6% of the total approved during that period. All in all, not a bad ratio, but the number of poor outcomes associated with a high-profile dud can be astronomical.
I think there are several major reasons why we have adverse issues with medications that have survived the rigors of the initial FDA approval process. First, many human drug trials are conducted in developing countries, where the human genome is much more homogenous and the liabilities for injuries are way less than in the U.S. Many researchers have acknowledged the significant role of pharmacogenomics, and how each physiology and pathology is unique. Couple these with the tendency to test drugs one at a time in younger cohorts—very few medications are administered in this manner in the U.S.—and one can quickly see how complex the equation becomes.
Another reason is the influence relegated to clinical trials. All clinicians should be familiar with the stages (0 to 4) and processes of how the FDA analyzes human drug trials. The FDA usually requires that two “adequate and well-controlled” trials confirm that a drug is safe and effective before it approves it for sale to the public. Once a drug completes Stage 3, an extensive statistical analysis is conducted to assure a drug’s demonstrated benefit is real and not the result of chance. But as it turns out, because the measured effects in most clinical trials are so small, chance is very hard to prove or disprove.
This was astutely demonstrated in a 2005 article published in the Journal of the American Medical Association (2005;294(2):218-228). John P. Ioannidis, MD, examined the results of 49 high-profile clinical-research studies in which 45 found that proposed intervention was effective. Of the 45 claiming effectiveness, seven (16%) were contradicted by subsequent studies, and seven others had found effects that were stronger than those of subsequent studies. Of the 26 randomly controlled trials that were followed up by larger trials, the initial finding was entirely contradicted in three cases (12%); another six cases (23%) found the benefit to be less than half of what had been initially reported.
In most instances, it wasn’t the therapy that changed but the sample size. In fact, many clinicians and biostatisticians believe many more so-called “evidence-based” practices or medicinals would be legitimately challenged if subjected to rigorous follow-up studies.
In my own personal experience as a hospitalist, I can think of two areas where the general medical community accepted initial studies only to refute them later: perioperative use of beta-blockers and inpatient glycemic control.
In light of the many high-profile medications that have been pulled from the market, I don’t like being in the first group to jump on the bandwagon. My general rule is to wait three to five years after a drug has been released before prescribing for patients. As always, there are exceptions. In instances where new medications have profound or life-altering potential (i.e. the new anticoagulants or gene-targeting meds for certain cancers) and the risks are substantiated, I’m all in!
Do you have a problem or concern that you’d like Dr. Hospitalist to address? Email your questions to [email protected].