User login
New KDIGO guideline encourages use of HCV-positive kidneys for HCV-negative recipients
The Kidney Disease: Improving Global Outcomes (KDIGO) Work Group has updated its guideline concerning the prevention, diagnosis, evaluation, and treatment of hepatitis C virus (HCV) infection in patients with chronic kidney disease (CKD).
Of note, KDIGO now supports transplant of HCV-positive kidneys to HCV-negative recipients.
The guidance document, authored by Ahmed Arslan Yousuf Awan, MD, of Baylor College of Medicine, Houston, and colleagues, was written in light of new evidence that has emerged since the 2018 guideline was published.
“The focused update was triggered by new data on antiviral treatment in patients with advanced stages of CKD (G4, G5, or G5D), transplant of HCV-infected kidneys into uninfected recipients, and evolution of the viewpoint on the role of kidney biopsy in managing kidney disease caused by HCV,” the guideline panelists wrote in Annals of Internal Medicine. “This update is intended to assist clinicians in the care of patients with HCV infection and CKD, including patients receiving dialysis (CKD G5D) and patients with a kidney transplant (CKD G1T-G5T).”
Anjay Rastogi, MD, PhD, professor and clinical chief of nephrology at the David Geffen School of Medicine at UCLA, said the update is both “timely and relevant,” and “will really have an impact on the organ shortage that we have for kidney transplant”
The updates are outlined below.
Expanded Access to HCV-Positive Kidneys
While the 2018 guideline recommended that HCV-positive kidneys be directed to HCV-positive recipients, the new guideline suggests that these kidneys are appropriate for all patients regardless of HCV status.
In support, the panelists cited a follow-up of THINKER-1 trial, which showed that eGFR and quality of life were not negatively affected when HCV-negative patients received an HCV-positive kidney, compared with an HCV-negative kidney. Data from 525 unmatched recipients in 16 other studies support this conclusion, the panelists noted.
Jose Debes, MD, PhD, associate professor at the University of Minnesota, Minneapolis, suggested that this is the most important update to the KDIGO guidelines.
“That [change] would be the main impact of these recommendations,” Dr. Debes said in an interview. “Several centers were already doing this, since some data [were] out there, but I think the fact that they’re making this into a guideline is quite important.”
Dr. Rastogi agreed that this recommendation is the most impactful update.
“That’s a big move,” Dr. Rastogi said in an interview. He predicted that the change will “definitely increase the donor pool, which is very, very important.”
For this new recommendation to have the greatest positive effect, however, Dr. Rastogi suggested that health care providers and treatment centers need to prepare an effective implementation strategy. He emphasized the importance of early communication with patients concerning the safety of HCV-positive kidneys, which depends on early initiation of direct-acting antiviral (DAA) therapy.
In the guideline, Dr. Awan and colleagues reported three documented cases of fibrosing cholestatic hepatitis occurred in patients who did not begin DAA therapy until 30 days after transplant.
“[Patients] should start [DAA treatment] right away,” Dr. Rastogi said, “and sometimes even before the transplant.”
This will require institutional support, he noted, as centers need to ensure that patients are covered for DAA therapy and medication is readily available.
Sofosbuvir Given the Green Light
Compared with the 2018 guideline, which recommended against sofosbuvir in patients with CKD G4 and G5, including those on dialysis, because of concerns about metabolization via the kidneys, the new guideline suggests that sofosbuvir-based DAA regimens are appropriate in patients with glomerular filtration rate (GFR) less than 30 mL/min per 1.73 m2, including those receiving dialysis.
This recommendation was based on a systematic review of 106 studies including both sofosbuvir-based and non-sofosbuvir-based DAA regimens that showed high safety and efficacy for all DAA regimen types across a broad variety of patient types.
“DAAs are highly effective and well tolerated treatments for hepatitis C in patients across all stages of CKD, including those undergoing dialysis and kidney transplant recipients, with no need for dose adjustment,” Dr. Awan and colleagues wrote.
Loosened Biopsy Requirements
Unlike the 2018 guideline, which advised kidney biopsy in HCV-positive patients with clinical evidence of glomerular disease prior to initiating DAA treatment, the new guideline suggests that HCV-infected patients with a typical presentation of immune-complex proliferative glomerulonephritis do not require confirmatory kidney biopsy.
“Because almost all patients with chronic hepatitis C (with or without glomerulonephritis) should be treated with DAAs, a kidney biopsy is unlikely to change management in most patients with hepatitis C and clinical glomerulonephritis,” the panelists wrote.
If kidney disease does not stabilize or improve with achievement of sustained virologic response, or if there is evidence of rapidly progressive glomerulonephritis, then a kidney biopsy should be considered before beginning immunosuppressive therapy, according to the guideline, which includes a flow chart to guide clinicians through this decision-making process.
Individualizing Immunosuppressive Therapy
Consistent with the old guideline, the new guideline recommends DAA treatment with concurrent immunosuppressive therapy for patients with cryoglobulinemic flare or rapidly progressive kidney failure. But in contrast, the new guideline calls for an individualized approach to immunosuppression in patients with nephrotic syndrome.
Dr. Awan and colleagues suggested that “nephrotic-range proteinuria (greater than 3.5 g/d) alone does not warrant use of immunosuppressive treatment because such patients can achieve remission of proteinuria after treatment with DAAs.” Still, if other associated complications — such as anasarca, thromboembolic disease, or severe hypoalbuminemia — are present, then immunosuppressive therapy may be warranted, with rituximab remaining the preferred first-line agent.
More Work Is Needed
Dr. Awan and colleagues concluded the guideline by highlighting areas of unmet need, and how filling these knowledge gaps could lead to additional guideline updates.
“Future studies of kidney donations from HCV-positive donors to HCV-negative recipients are needed to refine and clarify the timing of initiation and duration of DAA therapy and to assess long-term outcomes associated with this practice,” they wrote. “Also, randomized controlled trials are needed to determine which patients with HCV-associated kidney disease can be treated with DAA therapy alone versus in combination with immunosuppression and plasma exchange. KDIGO will assess the currency of its recommendations and the need to update them in the next 3 years.”
The guideline was funded by KDIGO. The investigators disclosed relationships with GSK, Gilead, Intercept, Novo Nordisk, and others. Dr. Rastogi and Dr. Debes had no conflicts of interest.
The Kidney Disease: Improving Global Outcomes (KDIGO) Work Group has updated its guideline concerning the prevention, diagnosis, evaluation, and treatment of hepatitis C virus (HCV) infection in patients with chronic kidney disease (CKD).
Of note, KDIGO now supports transplant of HCV-positive kidneys to HCV-negative recipients.
The guidance document, authored by Ahmed Arslan Yousuf Awan, MD, of Baylor College of Medicine, Houston, and colleagues, was written in light of new evidence that has emerged since the 2018 guideline was published.
“The focused update was triggered by new data on antiviral treatment in patients with advanced stages of CKD (G4, G5, or G5D), transplant of HCV-infected kidneys into uninfected recipients, and evolution of the viewpoint on the role of kidney biopsy in managing kidney disease caused by HCV,” the guideline panelists wrote in Annals of Internal Medicine. “This update is intended to assist clinicians in the care of patients with HCV infection and CKD, including patients receiving dialysis (CKD G5D) and patients with a kidney transplant (CKD G1T-G5T).”
Anjay Rastogi, MD, PhD, professor and clinical chief of nephrology at the David Geffen School of Medicine at UCLA, said the update is both “timely and relevant,” and “will really have an impact on the organ shortage that we have for kidney transplant”
The updates are outlined below.
Expanded Access to HCV-Positive Kidneys
While the 2018 guideline recommended that HCV-positive kidneys be directed to HCV-positive recipients, the new guideline suggests that these kidneys are appropriate for all patients regardless of HCV status.
In support, the panelists cited a follow-up of THINKER-1 trial, which showed that eGFR and quality of life were not negatively affected when HCV-negative patients received an HCV-positive kidney, compared with an HCV-negative kidney. Data from 525 unmatched recipients in 16 other studies support this conclusion, the panelists noted.
Jose Debes, MD, PhD, associate professor at the University of Minnesota, Minneapolis, suggested that this is the most important update to the KDIGO guidelines.
“That [change] would be the main impact of these recommendations,” Dr. Debes said in an interview. “Several centers were already doing this, since some data [were] out there, but I think the fact that they’re making this into a guideline is quite important.”
Dr. Rastogi agreed that this recommendation is the most impactful update.
“That’s a big move,” Dr. Rastogi said in an interview. He predicted that the change will “definitely increase the donor pool, which is very, very important.”
For this new recommendation to have the greatest positive effect, however, Dr. Rastogi suggested that health care providers and treatment centers need to prepare an effective implementation strategy. He emphasized the importance of early communication with patients concerning the safety of HCV-positive kidneys, which depends on early initiation of direct-acting antiviral (DAA) therapy.
In the guideline, Dr. Awan and colleagues reported three documented cases of fibrosing cholestatic hepatitis occurred in patients who did not begin DAA therapy until 30 days after transplant.
“[Patients] should start [DAA treatment] right away,” Dr. Rastogi said, “and sometimes even before the transplant.”
This will require institutional support, he noted, as centers need to ensure that patients are covered for DAA therapy and medication is readily available.
Sofosbuvir Given the Green Light
Compared with the 2018 guideline, which recommended against sofosbuvir in patients with CKD G4 and G5, including those on dialysis, because of concerns about metabolization via the kidneys, the new guideline suggests that sofosbuvir-based DAA regimens are appropriate in patients with glomerular filtration rate (GFR) less than 30 mL/min per 1.73 m2, including those receiving dialysis.
This recommendation was based on a systematic review of 106 studies including both sofosbuvir-based and non-sofosbuvir-based DAA regimens that showed high safety and efficacy for all DAA regimen types across a broad variety of patient types.
“DAAs are highly effective and well tolerated treatments for hepatitis C in patients across all stages of CKD, including those undergoing dialysis and kidney transplant recipients, with no need for dose adjustment,” Dr. Awan and colleagues wrote.
Loosened Biopsy Requirements
Unlike the 2018 guideline, which advised kidney biopsy in HCV-positive patients with clinical evidence of glomerular disease prior to initiating DAA treatment, the new guideline suggests that HCV-infected patients with a typical presentation of immune-complex proliferative glomerulonephritis do not require confirmatory kidney biopsy.
“Because almost all patients with chronic hepatitis C (with or without glomerulonephritis) should be treated with DAAs, a kidney biopsy is unlikely to change management in most patients with hepatitis C and clinical glomerulonephritis,” the panelists wrote.
If kidney disease does not stabilize or improve with achievement of sustained virologic response, or if there is evidence of rapidly progressive glomerulonephritis, then a kidney biopsy should be considered before beginning immunosuppressive therapy, according to the guideline, which includes a flow chart to guide clinicians through this decision-making process.
Individualizing Immunosuppressive Therapy
Consistent with the old guideline, the new guideline recommends DAA treatment with concurrent immunosuppressive therapy for patients with cryoglobulinemic flare or rapidly progressive kidney failure. But in contrast, the new guideline calls for an individualized approach to immunosuppression in patients with nephrotic syndrome.
Dr. Awan and colleagues suggested that “nephrotic-range proteinuria (greater than 3.5 g/d) alone does not warrant use of immunosuppressive treatment because such patients can achieve remission of proteinuria after treatment with DAAs.” Still, if other associated complications — such as anasarca, thromboembolic disease, or severe hypoalbuminemia — are present, then immunosuppressive therapy may be warranted, with rituximab remaining the preferred first-line agent.
More Work Is Needed
Dr. Awan and colleagues concluded the guideline by highlighting areas of unmet need, and how filling these knowledge gaps could lead to additional guideline updates.
“Future studies of kidney donations from HCV-positive donors to HCV-negative recipients are needed to refine and clarify the timing of initiation and duration of DAA therapy and to assess long-term outcomes associated with this practice,” they wrote. “Also, randomized controlled trials are needed to determine which patients with HCV-associated kidney disease can be treated with DAA therapy alone versus in combination with immunosuppression and plasma exchange. KDIGO will assess the currency of its recommendations and the need to update them in the next 3 years.”
The guideline was funded by KDIGO. The investigators disclosed relationships with GSK, Gilead, Intercept, Novo Nordisk, and others. Dr. Rastogi and Dr. Debes had no conflicts of interest.
The Kidney Disease: Improving Global Outcomes (KDIGO) Work Group has updated its guideline concerning the prevention, diagnosis, evaluation, and treatment of hepatitis C virus (HCV) infection in patients with chronic kidney disease (CKD).
Of note, KDIGO now supports transplant of HCV-positive kidneys to HCV-negative recipients.
The guidance document, authored by Ahmed Arslan Yousuf Awan, MD, of Baylor College of Medicine, Houston, and colleagues, was written in light of new evidence that has emerged since the 2018 guideline was published.
“The focused update was triggered by new data on antiviral treatment in patients with advanced stages of CKD (G4, G5, or G5D), transplant of HCV-infected kidneys into uninfected recipients, and evolution of the viewpoint on the role of kidney biopsy in managing kidney disease caused by HCV,” the guideline panelists wrote in Annals of Internal Medicine. “This update is intended to assist clinicians in the care of patients with HCV infection and CKD, including patients receiving dialysis (CKD G5D) and patients with a kidney transplant (CKD G1T-G5T).”
Anjay Rastogi, MD, PhD, professor and clinical chief of nephrology at the David Geffen School of Medicine at UCLA, said the update is both “timely and relevant,” and “will really have an impact on the organ shortage that we have for kidney transplant”
The updates are outlined below.
Expanded Access to HCV-Positive Kidneys
While the 2018 guideline recommended that HCV-positive kidneys be directed to HCV-positive recipients, the new guideline suggests that these kidneys are appropriate for all patients regardless of HCV status.
In support, the panelists cited a follow-up of THINKER-1 trial, which showed that eGFR and quality of life were not negatively affected when HCV-negative patients received an HCV-positive kidney, compared with an HCV-negative kidney. Data from 525 unmatched recipients in 16 other studies support this conclusion, the panelists noted.
Jose Debes, MD, PhD, associate professor at the University of Minnesota, Minneapolis, suggested that this is the most important update to the KDIGO guidelines.
“That [change] would be the main impact of these recommendations,” Dr. Debes said in an interview. “Several centers were already doing this, since some data [were] out there, but I think the fact that they’re making this into a guideline is quite important.”
Dr. Rastogi agreed that this recommendation is the most impactful update.
“That’s a big move,” Dr. Rastogi said in an interview. He predicted that the change will “definitely increase the donor pool, which is very, very important.”
For this new recommendation to have the greatest positive effect, however, Dr. Rastogi suggested that health care providers and treatment centers need to prepare an effective implementation strategy. He emphasized the importance of early communication with patients concerning the safety of HCV-positive kidneys, which depends on early initiation of direct-acting antiviral (DAA) therapy.
In the guideline, Dr. Awan and colleagues reported three documented cases of fibrosing cholestatic hepatitis occurred in patients who did not begin DAA therapy until 30 days after transplant.
“[Patients] should start [DAA treatment] right away,” Dr. Rastogi said, “and sometimes even before the transplant.”
This will require institutional support, he noted, as centers need to ensure that patients are covered for DAA therapy and medication is readily available.
Sofosbuvir Given the Green Light
Compared with the 2018 guideline, which recommended against sofosbuvir in patients with CKD G4 and G5, including those on dialysis, because of concerns about metabolization via the kidneys, the new guideline suggests that sofosbuvir-based DAA regimens are appropriate in patients with glomerular filtration rate (GFR) less than 30 mL/min per 1.73 m2, including those receiving dialysis.
This recommendation was based on a systematic review of 106 studies including both sofosbuvir-based and non-sofosbuvir-based DAA regimens that showed high safety and efficacy for all DAA regimen types across a broad variety of patient types.
“DAAs are highly effective and well tolerated treatments for hepatitis C in patients across all stages of CKD, including those undergoing dialysis and kidney transplant recipients, with no need for dose adjustment,” Dr. Awan and colleagues wrote.
Loosened Biopsy Requirements
Unlike the 2018 guideline, which advised kidney biopsy in HCV-positive patients with clinical evidence of glomerular disease prior to initiating DAA treatment, the new guideline suggests that HCV-infected patients with a typical presentation of immune-complex proliferative glomerulonephritis do not require confirmatory kidney biopsy.
“Because almost all patients with chronic hepatitis C (with or without glomerulonephritis) should be treated with DAAs, a kidney biopsy is unlikely to change management in most patients with hepatitis C and clinical glomerulonephritis,” the panelists wrote.
If kidney disease does not stabilize or improve with achievement of sustained virologic response, or if there is evidence of rapidly progressive glomerulonephritis, then a kidney biopsy should be considered before beginning immunosuppressive therapy, according to the guideline, which includes a flow chart to guide clinicians through this decision-making process.
Individualizing Immunosuppressive Therapy
Consistent with the old guideline, the new guideline recommends DAA treatment with concurrent immunosuppressive therapy for patients with cryoglobulinemic flare or rapidly progressive kidney failure. But in contrast, the new guideline calls for an individualized approach to immunosuppression in patients with nephrotic syndrome.
Dr. Awan and colleagues suggested that “nephrotic-range proteinuria (greater than 3.5 g/d) alone does not warrant use of immunosuppressive treatment because such patients can achieve remission of proteinuria after treatment with DAAs.” Still, if other associated complications — such as anasarca, thromboembolic disease, or severe hypoalbuminemia — are present, then immunosuppressive therapy may be warranted, with rituximab remaining the preferred first-line agent.
More Work Is Needed
Dr. Awan and colleagues concluded the guideline by highlighting areas of unmet need, and how filling these knowledge gaps could lead to additional guideline updates.
“Future studies of kidney donations from HCV-positive donors to HCV-negative recipients are needed to refine and clarify the timing of initiation and duration of DAA therapy and to assess long-term outcomes associated with this practice,” they wrote. “Also, randomized controlled trials are needed to determine which patients with HCV-associated kidney disease can be treated with DAA therapy alone versus in combination with immunosuppression and plasma exchange. KDIGO will assess the currency of its recommendations and the need to update them in the next 3 years.”
The guideline was funded by KDIGO. The investigators disclosed relationships with GSK, Gilead, Intercept, Novo Nordisk, and others. Dr. Rastogi and Dr. Debes had no conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
Why Are Prion Diseases on the Rise?
This transcript has been edited for clarity.
In 1986, in Britain, cattle started dying.
The condition, quickly nicknamed “mad cow disease,” was clearly infectious, but the particular pathogen was difficult to identify. By 1993, 120,000 cattle in Britain were identified as being infected. As yet, no human cases had occurred and the UK government insisted that cattle were a dead-end host for the pathogen. By the mid-1990s, however, multiple human cases, attributable to ingestion of meat and organs from infected cattle, were discovered. In humans, variant Creutzfeldt-Jakob disease (CJD) was a media sensation — a nearly uniformly fatal, untreatable condition with a rapid onset of dementia, mobility issues characterized by jerky movements, and autopsy reports finding that the brain itself had turned into a spongy mess.
The United States banned UK beef imports in 1996 and only lifted the ban in 2020.
The disease was made all the more mysterious because the pathogen involved was not a bacterium, parasite, or virus, but a protein — or a proteinaceous infectious particle, shortened to “prion.”
Prions are misfolded proteins that aggregate in cells — in this case, in nerve cells. But what makes prions different from other misfolded proteins is that the misfolded protein catalyzes the conversion of its non-misfolded counterpart into the misfolded configuration. It creates a chain reaction, leading to rapid accumulation of misfolded proteins and cell death.
And, like a time bomb, we all have prion protein inside us. In its normally folded state, the function of prion protein remains unclear — knockout mice do okay without it — but it is also highly conserved across mammalian species, so it probably does something worthwhile, perhaps protecting nerve fibers.
Far more common than humans contracting mad cow disease is the condition known as sporadic CJD, responsible for 85% of all cases of prion-induced brain disease. The cause of sporadic CJD is unknown.
But one thing is known: Cases are increasing.
I don’t want you to freak out; we are not in the midst of a CJD epidemic. But it’s been a while since I’ve seen people discussing the condition — which remains as horrible as it was in the 1990s — and a new research letter appearing in JAMA Neurology brought it back to the top of my mind.
Researchers, led by Matthew Crane at Hopkins, used the CDC’s WONDER cause-of-death database, which pulls diagnoses from death certificates. Normally, I’m not a fan of using death certificates for cause-of-death analyses, but in this case I’ll give it a pass. Assuming that the diagnosis of CJD is made, it would be really unlikely for it not to appear on a death certificate.
The main findings are seen here. 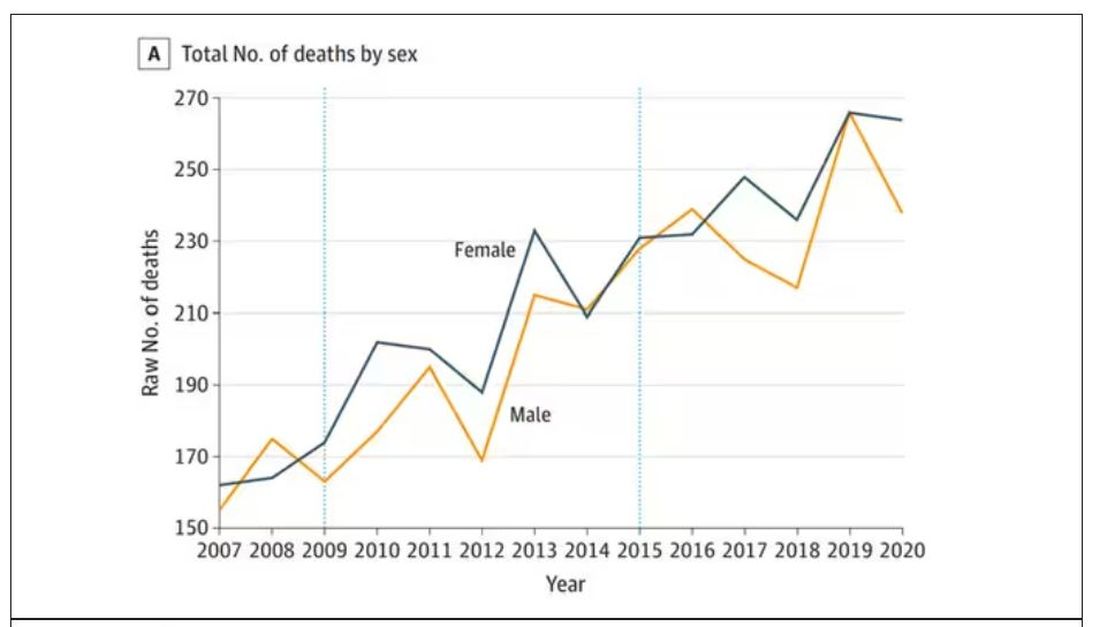
Note that we can’t tell whether these are sporadic CJD cases or variant CJD cases or even familial CJD cases; however, unless there has been a dramatic change in epidemiology, the vast majority of these will be sporadic.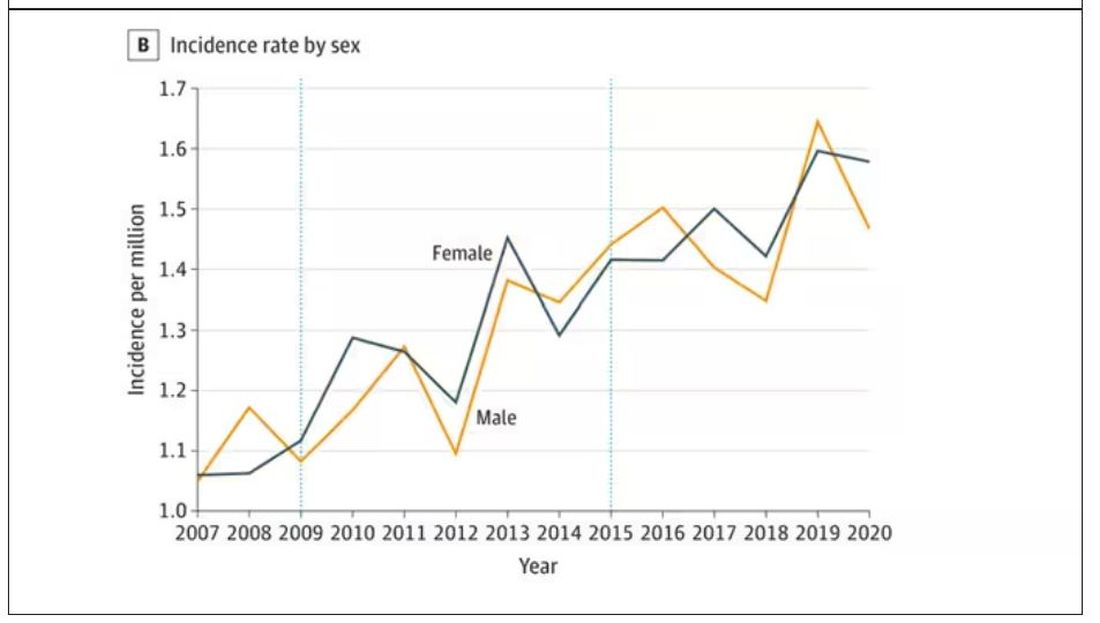
The question is, why are there more cases?
Whenever this type of question comes up with any disease, there are basically three possibilities:
First, there may be an increase in the susceptible, or at-risk, population. In this case, we know that older people are at higher risk of developing sporadic CJD, and over time, the population has aged. To be fair, the authors adjusted for this and still saw an increase, though it was attenuated.
Second, we might be better at diagnosing the condition. A lot has happened since the mid-1990s, when the diagnosis was based more or less on symptoms. The advent of more sophisticated MRI protocols as well as a new diagnostic test called “real-time quaking-induced conversion testing” may mean we are just better at detecting people with this disease.
Third (and most concerning), a new exposure has occurred. What that exposure might be, where it might come from, is anyone’s guess. It’s hard to do broad-scale epidemiology on very rare diseases.
But given these findings, it seems that a bit more surveillance for this rare but devastating condition is well merited.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and his new book, How Medicine Works and When It Doesn’t, is available now.
F. Perry Wilson, MD, MSCE, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
In 1986, in Britain, cattle started dying.
The condition, quickly nicknamed “mad cow disease,” was clearly infectious, but the particular pathogen was difficult to identify. By 1993, 120,000 cattle in Britain were identified as being infected. As yet, no human cases had occurred and the UK government insisted that cattle were a dead-end host for the pathogen. By the mid-1990s, however, multiple human cases, attributable to ingestion of meat and organs from infected cattle, were discovered. In humans, variant Creutzfeldt-Jakob disease (CJD) was a media sensation — a nearly uniformly fatal, untreatable condition with a rapid onset of dementia, mobility issues characterized by jerky movements, and autopsy reports finding that the brain itself had turned into a spongy mess.
The United States banned UK beef imports in 1996 and only lifted the ban in 2020.
The disease was made all the more mysterious because the pathogen involved was not a bacterium, parasite, or virus, but a protein — or a proteinaceous infectious particle, shortened to “prion.”
Prions are misfolded proteins that aggregate in cells — in this case, in nerve cells. But what makes prions different from other misfolded proteins is that the misfolded protein catalyzes the conversion of its non-misfolded counterpart into the misfolded configuration. It creates a chain reaction, leading to rapid accumulation of misfolded proteins and cell death.
And, like a time bomb, we all have prion protein inside us. In its normally folded state, the function of prion protein remains unclear — knockout mice do okay without it — but it is also highly conserved across mammalian species, so it probably does something worthwhile, perhaps protecting nerve fibers.
Far more common than humans contracting mad cow disease is the condition known as sporadic CJD, responsible for 85% of all cases of prion-induced brain disease. The cause of sporadic CJD is unknown.
But one thing is known: Cases are increasing.
I don’t want you to freak out; we are not in the midst of a CJD epidemic. But it’s been a while since I’ve seen people discussing the condition — which remains as horrible as it was in the 1990s — and a new research letter appearing in JAMA Neurology brought it back to the top of my mind.
Researchers, led by Matthew Crane at Hopkins, used the CDC’s WONDER cause-of-death database, which pulls diagnoses from death certificates. Normally, I’m not a fan of using death certificates for cause-of-death analyses, but in this case I’ll give it a pass. Assuming that the diagnosis of CJD is made, it would be really unlikely for it not to appear on a death certificate.
The main findings are seen here. 
Note that we can’t tell whether these are sporadic CJD cases or variant CJD cases or even familial CJD cases; however, unless there has been a dramatic change in epidemiology, the vast majority of these will be sporadic.
The question is, why are there more cases?
Whenever this type of question comes up with any disease, there are basically three possibilities:
First, there may be an increase in the susceptible, or at-risk, population. In this case, we know that older people are at higher risk of developing sporadic CJD, and over time, the population has aged. To be fair, the authors adjusted for this and still saw an increase, though it was attenuated.
Second, we might be better at diagnosing the condition. A lot has happened since the mid-1990s, when the diagnosis was based more or less on symptoms. The advent of more sophisticated MRI protocols as well as a new diagnostic test called “real-time quaking-induced conversion testing” may mean we are just better at detecting people with this disease.
Third (and most concerning), a new exposure has occurred. What that exposure might be, where it might come from, is anyone’s guess. It’s hard to do broad-scale epidemiology on very rare diseases.
But given these findings, it seems that a bit more surveillance for this rare but devastating condition is well merited.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and his new book, How Medicine Works and When It Doesn’t, is available now.
F. Perry Wilson, MD, MSCE, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
In 1986, in Britain, cattle started dying.
The condition, quickly nicknamed “mad cow disease,” was clearly infectious, but the particular pathogen was difficult to identify. By 1993, 120,000 cattle in Britain were identified as being infected. As yet, no human cases had occurred and the UK government insisted that cattle were a dead-end host for the pathogen. By the mid-1990s, however, multiple human cases, attributable to ingestion of meat and organs from infected cattle, were discovered. In humans, variant Creutzfeldt-Jakob disease (CJD) was a media sensation — a nearly uniformly fatal, untreatable condition with a rapid onset of dementia, mobility issues characterized by jerky movements, and autopsy reports finding that the brain itself had turned into a spongy mess.
The United States banned UK beef imports in 1996 and only lifted the ban in 2020.
The disease was made all the more mysterious because the pathogen involved was not a bacterium, parasite, or virus, but a protein — or a proteinaceous infectious particle, shortened to “prion.”
Prions are misfolded proteins that aggregate in cells — in this case, in nerve cells. But what makes prions different from other misfolded proteins is that the misfolded protein catalyzes the conversion of its non-misfolded counterpart into the misfolded configuration. It creates a chain reaction, leading to rapid accumulation of misfolded proteins and cell death.
And, like a time bomb, we all have prion protein inside us. In its normally folded state, the function of prion protein remains unclear — knockout mice do okay without it — but it is also highly conserved across mammalian species, so it probably does something worthwhile, perhaps protecting nerve fibers.
Far more common than humans contracting mad cow disease is the condition known as sporadic CJD, responsible for 85% of all cases of prion-induced brain disease. The cause of sporadic CJD is unknown.
But one thing is known: Cases are increasing.
I don’t want you to freak out; we are not in the midst of a CJD epidemic. But it’s been a while since I’ve seen people discussing the condition — which remains as horrible as it was in the 1990s — and a new research letter appearing in JAMA Neurology brought it back to the top of my mind.
Researchers, led by Matthew Crane at Hopkins, used the CDC’s WONDER cause-of-death database, which pulls diagnoses from death certificates. Normally, I’m not a fan of using death certificates for cause-of-death analyses, but in this case I’ll give it a pass. Assuming that the diagnosis of CJD is made, it would be really unlikely for it not to appear on a death certificate.
The main findings are seen here. 
Note that we can’t tell whether these are sporadic CJD cases or variant CJD cases or even familial CJD cases; however, unless there has been a dramatic change in epidemiology, the vast majority of these will be sporadic.
The question is, why are there more cases?
Whenever this type of question comes up with any disease, there are basically three possibilities:
First, there may be an increase in the susceptible, or at-risk, population. In this case, we know that older people are at higher risk of developing sporadic CJD, and over time, the population has aged. To be fair, the authors adjusted for this and still saw an increase, though it was attenuated.
Second, we might be better at diagnosing the condition. A lot has happened since the mid-1990s, when the diagnosis was based more or less on symptoms. The advent of more sophisticated MRI protocols as well as a new diagnostic test called “real-time quaking-induced conversion testing” may mean we are just better at detecting people with this disease.
Third (and most concerning), a new exposure has occurred. What that exposure might be, where it might come from, is anyone’s guess. It’s hard to do broad-scale epidemiology on very rare diseases.
But given these findings, it seems that a bit more surveillance for this rare but devastating condition is well merited.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and his new book, How Medicine Works and When It Doesn’t, is available now.
F. Perry Wilson, MD, MSCE, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
New COVID variant JN.1 could disrupt holiday plans
No one planning holiday gatherings or travel wants to hear this, but the rise of a new COVID-19 variant, JN.1, is concerning experts, who say it may threaten those good times.
The good news is recent research suggests the 2023-2024 COVID-19 vaccine appears to work against this newest variant. But so few people have gotten the latest vaccine — less than 16% of U.S. adults — that some experts suggest it’s time for the CDC to urge the public who haven’t it to do so now, so the antibodies can kick in before the festivities.
“A significant wave [of JN.1] has started here and could be blunted with a high booster rate and mitigation measures,” said Eric Topol, MD, professor and executive vice president of Scripps Research in La Jolla, CA, and editor-in-chief of Medscape, a sister site of this news organization.
COVID metrics, meanwhile, have started to climb again. Nearly 10,000 people were hospitalized for COVID in the U.S. for the week ending Nov. 25, the CDC said, a 10% increase over the previous week.
Who’s Who in the Family Tree
JN.1, an Omicron subvariant, was first detected in the U.S. in September and is termed “a notable descendent lineage” of Omicron subvariant BA.2.86 by the World Health Organization. When BA.2.86, also known as Pirola, was first identified in August, it appeared very different from other variants, the CDC said. That triggered concerns it might be more infectious than previous ones, even for people with immunity from vaccination and previous infections.
“JN.1 is Pirola’s kid,” said Rajendram Rajnarayanan, PhD, assistant dean of research and associate professor at the New York Institute of Technology at Arkansas State University, who maintains a COVID-19 variant database. The variant BA.2.86 and offspring are worrisome due to the mutations, he said.
How Widespread Is JN.1?
As of Nov. 27, the CDC says, BA.2.86 is projected to comprise 5%-15% of circulating variants in the U.S. “The expected public health risk of this variant, including its offshoot JN.1, is low,” the agency said.
Currently, JN.1 is reported more often in Europe, Dr. Rajnarayanan said, but some countries have better reporting data than others. “It has probably spread to every country tracking COVID,’’ he said, due to the mutations in the spike protein that make it easier for it to bind and infect.
Wastewater data suggest the variant’s rise is helping to fuel a wave, Dr. Topol said.
Vaccine Effectiveness Against JN.1, Other New Variants
The new XBB.1.5 monovalent vaccine, protects against XBB.1.5, another Omicron subvariant, but also JN.1 and other “emergent” viruses, a team of researchers reported Nov. 26 in a study on bioRxiv that has not yet been certified by peer review.
The updated vaccine, when given to uninfected people, boosted antibodies about 27-fold against XBB.1.5 and about 13- to 27-fold against JN.1 and other emergent viruses, the researchers reported.
While even primary doses of the COVID vaccine will likely help protect against the new JN.1 subvariant, “if you got the XBB.1.5 booster, it is going to be protecting you better against this new variant,” Dr. Rajnarayanan said.
2023-2024 Vaccine Uptake Low
In November, the CDC posted the first detailed estimates of who did. As of Nov. 18, less than 16% of U.S. adults had, with nearly 15% saying they planned to get it.
Coverage among children is lower, with just 6.3% of children up to date on the newest vaccine and 19% of parents saying they planned to get the 2023-2024 vaccine for their children.
Predictions, Mitigation
While some experts say a peak due to JN.1 is expected in the weeks ahead, Dr. Topol said it’s impossible to predict exactly how JN.1 will play out.
“It’s not going to be a repeat of November 2021,” when Omicron surfaced, Dr. Rajnarayanan predicted. Within 4 weeks of the World Health Organization declaring Omicron as a virus of concern, it spread around the world.
Mitigation measures can help, Dr. Rajnarayanan said. He suggested:
Get the new vaccine, and especially encourage vulnerable family and friends to do so.
If you are gathering inside for holiday festivities, improve circulation in the house, if possible.
Wear masks in airports and on planes and other public transportation.
A version of this article appeared on WebMD.com.
No one planning holiday gatherings or travel wants to hear this, but the rise of a new COVID-19 variant, JN.1, is concerning experts, who say it may threaten those good times.
The good news is recent research suggests the 2023-2024 COVID-19 vaccine appears to work against this newest variant. But so few people have gotten the latest vaccine — less than 16% of U.S. adults — that some experts suggest it’s time for the CDC to urge the public who haven’t it to do so now, so the antibodies can kick in before the festivities.
“A significant wave [of JN.1] has started here and could be blunted with a high booster rate and mitigation measures,” said Eric Topol, MD, professor and executive vice president of Scripps Research in La Jolla, CA, and editor-in-chief of Medscape, a sister site of this news organization.
COVID metrics, meanwhile, have started to climb again. Nearly 10,000 people were hospitalized for COVID in the U.S. for the week ending Nov. 25, the CDC said, a 10% increase over the previous week.
Who’s Who in the Family Tree
JN.1, an Omicron subvariant, was first detected in the U.S. in September and is termed “a notable descendent lineage” of Omicron subvariant BA.2.86 by the World Health Organization. When BA.2.86, also known as Pirola, was first identified in August, it appeared very different from other variants, the CDC said. That triggered concerns it might be more infectious than previous ones, even for people with immunity from vaccination and previous infections.
“JN.1 is Pirola’s kid,” said Rajendram Rajnarayanan, PhD, assistant dean of research and associate professor at the New York Institute of Technology at Arkansas State University, who maintains a COVID-19 variant database. The variant BA.2.86 and offspring are worrisome due to the mutations, he said.
How Widespread Is JN.1?
As of Nov. 27, the CDC says, BA.2.86 is projected to comprise 5%-15% of circulating variants in the U.S. “The expected public health risk of this variant, including its offshoot JN.1, is low,” the agency said.
Currently, JN.1 is reported more often in Europe, Dr. Rajnarayanan said, but some countries have better reporting data than others. “It has probably spread to every country tracking COVID,’’ he said, due to the mutations in the spike protein that make it easier for it to bind and infect.
Wastewater data suggest the variant’s rise is helping to fuel a wave, Dr. Topol said.
Vaccine Effectiveness Against JN.1, Other New Variants
The new XBB.1.5 monovalent vaccine, protects against XBB.1.5, another Omicron subvariant, but also JN.1 and other “emergent” viruses, a team of researchers reported Nov. 26 in a study on bioRxiv that has not yet been certified by peer review.
The updated vaccine, when given to uninfected people, boosted antibodies about 27-fold against XBB.1.5 and about 13- to 27-fold against JN.1 and other emergent viruses, the researchers reported.
While even primary doses of the COVID vaccine will likely help protect against the new JN.1 subvariant, “if you got the XBB.1.5 booster, it is going to be protecting you better against this new variant,” Dr. Rajnarayanan said.
2023-2024 Vaccine Uptake Low
In November, the CDC posted the first detailed estimates of who did. As of Nov. 18, less than 16% of U.S. adults had, with nearly 15% saying they planned to get it.
Coverage among children is lower, with just 6.3% of children up to date on the newest vaccine and 19% of parents saying they planned to get the 2023-2024 vaccine for their children.
Predictions, Mitigation
While some experts say a peak due to JN.1 is expected in the weeks ahead, Dr. Topol said it’s impossible to predict exactly how JN.1 will play out.
“It’s not going to be a repeat of November 2021,” when Omicron surfaced, Dr. Rajnarayanan predicted. Within 4 weeks of the World Health Organization declaring Omicron as a virus of concern, it spread around the world.
Mitigation measures can help, Dr. Rajnarayanan said. He suggested:
Get the new vaccine, and especially encourage vulnerable family and friends to do so.
If you are gathering inside for holiday festivities, improve circulation in the house, if possible.
Wear masks in airports and on planes and other public transportation.
A version of this article appeared on WebMD.com.
No one planning holiday gatherings or travel wants to hear this, but the rise of a new COVID-19 variant, JN.1, is concerning experts, who say it may threaten those good times.
The good news is recent research suggests the 2023-2024 COVID-19 vaccine appears to work against this newest variant. But so few people have gotten the latest vaccine — less than 16% of U.S. adults — that some experts suggest it’s time for the CDC to urge the public who haven’t it to do so now, so the antibodies can kick in before the festivities.
“A significant wave [of JN.1] has started here and could be blunted with a high booster rate and mitigation measures,” said Eric Topol, MD, professor and executive vice president of Scripps Research in La Jolla, CA, and editor-in-chief of Medscape, a sister site of this news organization.
COVID metrics, meanwhile, have started to climb again. Nearly 10,000 people were hospitalized for COVID in the U.S. for the week ending Nov. 25, the CDC said, a 10% increase over the previous week.
Who’s Who in the Family Tree
JN.1, an Omicron subvariant, was first detected in the U.S. in September and is termed “a notable descendent lineage” of Omicron subvariant BA.2.86 by the World Health Organization. When BA.2.86, also known as Pirola, was first identified in August, it appeared very different from other variants, the CDC said. That triggered concerns it might be more infectious than previous ones, even for people with immunity from vaccination and previous infections.
“JN.1 is Pirola’s kid,” said Rajendram Rajnarayanan, PhD, assistant dean of research and associate professor at the New York Institute of Technology at Arkansas State University, who maintains a COVID-19 variant database. The variant BA.2.86 and offspring are worrisome due to the mutations, he said.
How Widespread Is JN.1?
As of Nov. 27, the CDC says, BA.2.86 is projected to comprise 5%-15% of circulating variants in the U.S. “The expected public health risk of this variant, including its offshoot JN.1, is low,” the agency said.
Currently, JN.1 is reported more often in Europe, Dr. Rajnarayanan said, but some countries have better reporting data than others. “It has probably spread to every country tracking COVID,’’ he said, due to the mutations in the spike protein that make it easier for it to bind and infect.
Wastewater data suggest the variant’s rise is helping to fuel a wave, Dr. Topol said.
Vaccine Effectiveness Against JN.1, Other New Variants
The new XBB.1.5 monovalent vaccine, protects against XBB.1.5, another Omicron subvariant, but also JN.1 and other “emergent” viruses, a team of researchers reported Nov. 26 in a study on bioRxiv that has not yet been certified by peer review.
The updated vaccine, when given to uninfected people, boosted antibodies about 27-fold against XBB.1.5 and about 13- to 27-fold against JN.1 and other emergent viruses, the researchers reported.
While even primary doses of the COVID vaccine will likely help protect against the new JN.1 subvariant, “if you got the XBB.1.5 booster, it is going to be protecting you better against this new variant,” Dr. Rajnarayanan said.
2023-2024 Vaccine Uptake Low
In November, the CDC posted the first detailed estimates of who did. As of Nov. 18, less than 16% of U.S. adults had, with nearly 15% saying they planned to get it.
Coverage among children is lower, with just 6.3% of children up to date on the newest vaccine and 19% of parents saying they planned to get the 2023-2024 vaccine for their children.
Predictions, Mitigation
While some experts say a peak due to JN.1 is expected in the weeks ahead, Dr. Topol said it’s impossible to predict exactly how JN.1 will play out.
“It’s not going to be a repeat of November 2021,” when Omicron surfaced, Dr. Rajnarayanan predicted. Within 4 weeks of the World Health Organization declaring Omicron as a virus of concern, it spread around the world.
Mitigation measures can help, Dr. Rajnarayanan said. He suggested:
Get the new vaccine, and especially encourage vulnerable family and friends to do so.
If you are gathering inside for holiday festivities, improve circulation in the house, if possible.
Wear masks in airports and on planes and other public transportation.
A version of this article appeared on WebMD.com.
Some reasons to get off the fence about COVID booster
Though many people remain on the fence about getting the latest COVID vaccine booster, new research suggests a strong argument for getting the shot this winter: It sharply reduces the risk for COVID.
The risk reduction was 37% for those who received two doses. Experts say the research provides a strong argument for getting the vaccine, noting that about 10% of people infected with COVID go on to have long COVID, which can be debilitating for one quarter of those with long-lasting symptoms.
The data come from a systematic literature review and meta-analysis published in October in Antimicrobial Stewardship & Epidemiology. Researchers examined 32 studies published between December 2019 and June 2023, involving 775,931 adults. Twenty-four studies, encompassing 620,221 individuals, were included in the meta-analysis.
“The body of evidence from all these different studies converge on one single reality — that vaccines reduce the risk of long COVID, and people who keep up to date on their vaccinations also fared better than people who got it once or twice and didn’t follow up,” said Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University in St Louis.
Researchers have reported similar results for children. The National Institutes of Health RECOVER Initiative team found that vaccines are up to 42% effective in preventing long COVID in children, said Dr. Carlos Oliveira, MD, a pediatric infectious diseases specialist and Yale researcher who contributed to the study, which is in preprint.
Vaccines also protect children from multisystem inflammatory syndrome, a condition that can happen after COVID, as well as protect against other COVID-related problems, such as missed school days, Oliveira said. “Even if the vaccine doesn’t completely stop long COVID, it’s still good for kids to get vaccinated for all these other reasons.”
However, uptake for the latest boosters has been slow: the Centers for Disease Control and Prevention reported that by mid-November, less than 16% of people aged 18 years or older had received a shot. For children, the number was closer to 6%. A recent Kaiser Family Foundation survey found that booster rates for adults are similar to what it was 1 year ago.
The survey results suggest that people are no longer as worried about COVID, which is why there is less concerned about keeping up with boosters. Though the current mutation of the virus is not as debilitating as its predecessors, long COVID continues to be a problem: as of January 2023, 28% of people who had contracted the virus had experienced long-COVID symptoms. And though the mechanisms are still not fully understood, and researchers have yet to agree on a definition of long COVID, they are certain about this much: The best way to avoid it is to avoid getting infected to begin with.
The lack of a diagnostic test for long COVID and the fact that the symptoms mimic those of other diseases lead to inconsistency that can make studies hard to replicate. In the papers reviewed for the Antimicrobial Stewardship & Epidemiology study, long COVID was defined as having symptoms lasting from more than 4 weeks to more than 6 months. Alexandre Marra, MD, the lead author and a researcher at the Hospital Israelita Albert Einstein, in São Paulo, Brazil, and at the University of Iowa, said that a clear standard definition is needed to better understand the actual prevalence and evaluate vaccine effectiveness.
Al-Aly noted that there is a logical explanation for one finding in the paper: The percentage of individuals who had COVID and reported that long-COVID symptoms declined from 19% in June 2022 to 11% in January 2023.
Because a pandemic is a dynamic event, constantly producing different variants with different phenotypes, the prevalence of disease is naturally going to be affected. “People who got infected early in the pandemic may have a different long COVID profile and long COVID risk than people who got infected in the second or third year of the pandemic,” Al-Aly said.
Most of the studies reported data from before the Omicron-variant era. Only eight reported data during that era. Omicron was not as lethal as previous variants, and consequently, fewer patients developed long COVID during that time.
One of those who did is Yeng Chang, age 40 years, a family doctor who lives in Sherwood Park, Alberta, Canada. Chang developed long COVID during fall 2022 after getting the virus in June. By then, she’d been vaccinated three times, but she isn’t surprised that she got sick because each vaccine she had was developed before Omicron.
“When I had COVID I was really sick, but I was well enough to stay home,” she said. “I think if I didn’t have my immunizations, I might have been hospitalized, and I don’t know what would have happened.”
Long COVID has left Chang with brain fog, fatigue, and a lack of physical stamina that forced her to pause her medical practice. For the past year and a half, she’s spent more time as a patient than a physician.
Chang had her fifth COVID vaccination in the fall and recommends that others do the same. “The booster you got however many years ago was effective for the COVID of that time but there is a new COVID now. You can’t just say, ‘I had one and I’m fine forever.’”
A version of this article appeared on Medscape.com.
Though many people remain on the fence about getting the latest COVID vaccine booster, new research suggests a strong argument for getting the shot this winter: It sharply reduces the risk for COVID.
The risk reduction was 37% for those who received two doses. Experts say the research provides a strong argument for getting the vaccine, noting that about 10% of people infected with COVID go on to have long COVID, which can be debilitating for one quarter of those with long-lasting symptoms.
The data come from a systematic literature review and meta-analysis published in October in Antimicrobial Stewardship & Epidemiology. Researchers examined 32 studies published between December 2019 and June 2023, involving 775,931 adults. Twenty-four studies, encompassing 620,221 individuals, were included in the meta-analysis.
“The body of evidence from all these different studies converge on one single reality — that vaccines reduce the risk of long COVID, and people who keep up to date on their vaccinations also fared better than people who got it once or twice and didn’t follow up,” said Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University in St Louis.
Researchers have reported similar results for children. The National Institutes of Health RECOVER Initiative team found that vaccines are up to 42% effective in preventing long COVID in children, said Dr. Carlos Oliveira, MD, a pediatric infectious diseases specialist and Yale researcher who contributed to the study, which is in preprint.
Vaccines also protect children from multisystem inflammatory syndrome, a condition that can happen after COVID, as well as protect against other COVID-related problems, such as missed school days, Oliveira said. “Even if the vaccine doesn’t completely stop long COVID, it’s still good for kids to get vaccinated for all these other reasons.”
However, uptake for the latest boosters has been slow: the Centers for Disease Control and Prevention reported that by mid-November, less than 16% of people aged 18 years or older had received a shot. For children, the number was closer to 6%. A recent Kaiser Family Foundation survey found that booster rates for adults are similar to what it was 1 year ago.
The survey results suggest that people are no longer as worried about COVID, which is why there is less concerned about keeping up with boosters. Though the current mutation of the virus is not as debilitating as its predecessors, long COVID continues to be a problem: as of January 2023, 28% of people who had contracted the virus had experienced long-COVID symptoms. And though the mechanisms are still not fully understood, and researchers have yet to agree on a definition of long COVID, they are certain about this much: The best way to avoid it is to avoid getting infected to begin with.
The lack of a diagnostic test for long COVID and the fact that the symptoms mimic those of other diseases lead to inconsistency that can make studies hard to replicate. In the papers reviewed for the Antimicrobial Stewardship & Epidemiology study, long COVID was defined as having symptoms lasting from more than 4 weeks to more than 6 months. Alexandre Marra, MD, the lead author and a researcher at the Hospital Israelita Albert Einstein, in São Paulo, Brazil, and at the University of Iowa, said that a clear standard definition is needed to better understand the actual prevalence and evaluate vaccine effectiveness.
Al-Aly noted that there is a logical explanation for one finding in the paper: The percentage of individuals who had COVID and reported that long-COVID symptoms declined from 19% in June 2022 to 11% in January 2023.
Because a pandemic is a dynamic event, constantly producing different variants with different phenotypes, the prevalence of disease is naturally going to be affected. “People who got infected early in the pandemic may have a different long COVID profile and long COVID risk than people who got infected in the second or third year of the pandemic,” Al-Aly said.
Most of the studies reported data from before the Omicron-variant era. Only eight reported data during that era. Omicron was not as lethal as previous variants, and consequently, fewer patients developed long COVID during that time.
One of those who did is Yeng Chang, age 40 years, a family doctor who lives in Sherwood Park, Alberta, Canada. Chang developed long COVID during fall 2022 after getting the virus in June. By then, she’d been vaccinated three times, but she isn’t surprised that she got sick because each vaccine she had was developed before Omicron.
“When I had COVID I was really sick, but I was well enough to stay home,” she said. “I think if I didn’t have my immunizations, I might have been hospitalized, and I don’t know what would have happened.”
Long COVID has left Chang with brain fog, fatigue, and a lack of physical stamina that forced her to pause her medical practice. For the past year and a half, she’s spent more time as a patient than a physician.
Chang had her fifth COVID vaccination in the fall and recommends that others do the same. “The booster you got however many years ago was effective for the COVID of that time but there is a new COVID now. You can’t just say, ‘I had one and I’m fine forever.’”
A version of this article appeared on Medscape.com.
Though many people remain on the fence about getting the latest COVID vaccine booster, new research suggests a strong argument for getting the shot this winter: It sharply reduces the risk for COVID.
The risk reduction was 37% for those who received two doses. Experts say the research provides a strong argument for getting the vaccine, noting that about 10% of people infected with COVID go on to have long COVID, which can be debilitating for one quarter of those with long-lasting symptoms.
The data come from a systematic literature review and meta-analysis published in October in Antimicrobial Stewardship & Epidemiology. Researchers examined 32 studies published between December 2019 and June 2023, involving 775,931 adults. Twenty-four studies, encompassing 620,221 individuals, were included in the meta-analysis.
“The body of evidence from all these different studies converge on one single reality — that vaccines reduce the risk of long COVID, and people who keep up to date on their vaccinations also fared better than people who got it once or twice and didn’t follow up,” said Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University in St Louis.
Researchers have reported similar results for children. The National Institutes of Health RECOVER Initiative team found that vaccines are up to 42% effective in preventing long COVID in children, said Dr. Carlos Oliveira, MD, a pediatric infectious diseases specialist and Yale researcher who contributed to the study, which is in preprint.
Vaccines also protect children from multisystem inflammatory syndrome, a condition that can happen after COVID, as well as protect against other COVID-related problems, such as missed school days, Oliveira said. “Even if the vaccine doesn’t completely stop long COVID, it’s still good for kids to get vaccinated for all these other reasons.”
However, uptake for the latest boosters has been slow: the Centers for Disease Control and Prevention reported that by mid-November, less than 16% of people aged 18 years or older had received a shot. For children, the number was closer to 6%. A recent Kaiser Family Foundation survey found that booster rates for adults are similar to what it was 1 year ago.
The survey results suggest that people are no longer as worried about COVID, which is why there is less concerned about keeping up with boosters. Though the current mutation of the virus is not as debilitating as its predecessors, long COVID continues to be a problem: as of January 2023, 28% of people who had contracted the virus had experienced long-COVID symptoms. And though the mechanisms are still not fully understood, and researchers have yet to agree on a definition of long COVID, they are certain about this much: The best way to avoid it is to avoid getting infected to begin with.
The lack of a diagnostic test for long COVID and the fact that the symptoms mimic those of other diseases lead to inconsistency that can make studies hard to replicate. In the papers reviewed for the Antimicrobial Stewardship & Epidemiology study, long COVID was defined as having symptoms lasting from more than 4 weeks to more than 6 months. Alexandre Marra, MD, the lead author and a researcher at the Hospital Israelita Albert Einstein, in São Paulo, Brazil, and at the University of Iowa, said that a clear standard definition is needed to better understand the actual prevalence and evaluate vaccine effectiveness.
Al-Aly noted that there is a logical explanation for one finding in the paper: The percentage of individuals who had COVID and reported that long-COVID symptoms declined from 19% in June 2022 to 11% in January 2023.
Because a pandemic is a dynamic event, constantly producing different variants with different phenotypes, the prevalence of disease is naturally going to be affected. “People who got infected early in the pandemic may have a different long COVID profile and long COVID risk than people who got infected in the second or third year of the pandemic,” Al-Aly said.
Most of the studies reported data from before the Omicron-variant era. Only eight reported data during that era. Omicron was not as lethal as previous variants, and consequently, fewer patients developed long COVID during that time.
One of those who did is Yeng Chang, age 40 years, a family doctor who lives in Sherwood Park, Alberta, Canada. Chang developed long COVID during fall 2022 after getting the virus in June. By then, she’d been vaccinated three times, but she isn’t surprised that she got sick because each vaccine she had was developed before Omicron.
“When I had COVID I was really sick, but I was well enough to stay home,” she said. “I think if I didn’t have my immunizations, I might have been hospitalized, and I don’t know what would have happened.”
Long COVID has left Chang with brain fog, fatigue, and a lack of physical stamina that forced her to pause her medical practice. For the past year and a half, she’s spent more time as a patient than a physician.
Chang had her fifth COVID vaccination in the fall and recommends that others do the same. “The booster you got however many years ago was effective for the COVID of that time but there is a new COVID now. You can’t just say, ‘I had one and I’m fine forever.’”
A version of this article appeared on Medscape.com.
Global measles deaths increased by 43% in 2022
The number of total reported cases rose by 18% over the same period, accounting for approximately 9 million cases and 136,000 deaths globally, mostly among children. This information comes from a new report by the World Health Organization (WHO), published in partnership with the US Centers for Disease Control and Prevention (CDC).
More Measles Outbreaks
The report also notes an increase in the number of countries experiencing significant measles outbreaks. There were 37 such countries in 2022, compared with 22 the previous year. The most affected continents were Africa and Asia.
“The rise in measles outbreaks and deaths is impressive but, unfortunately, not surprising, given the decline in vaccination rates in recent years,” said John Vertefeuille, PhD, director of the CDC’s Global Immunization Division.
Vertefeuille emphasized that measles cases anywhere in the world pose a risk to “countries and communities where people are undervaccinated.” In recent years, several regions have fallen short of their immunization targets.
Vaccination Trends
In 2022, there was a slight increase in measles vaccination after a decline exacerbated by the COVID-19 pandemic and its impact on global healthcare systems. However, 33 million children did not receive at least one dose of the vaccine last year: 22 million missed the first dose, and 11 million missed the second.
For communities to be considered protected against outbreaks, immunization coverage with the full vaccine cycle should be at least 95%. The global coverage rate for the first dose was 83%, and for the second, it was 74%.
Nevertheless, immunization recovery has not reached the poorest countries, where the immunization rate stands at 66%. Brazil is among the top 10 countries where more children missed the first dose in 2022. These nations account for over half of the 22 million unadministered vaccines. According to the report, half a million children did not receive the vaccine in Brazil.
Measles in Brazil
Brazil’s results highlight setbacks in vaccination efforts. In 2016, the country was certified to have eliminated measles, but after experiencing outbreaks in 2018, the certification was lost in 2019. In 2018, Brazil confirmed 9325 cases. The situation worsened in 2019 with 20,901 diagnoses. Since then, numbers have been decreasing: 8100 in 2020, 676 in 2021, and 44 in 2022.
Last year, four Brazilian states reported confirmed virus cases: Rio de Janeiro, Pará, São Paulo, and Amapá. Ministry of Health data indicated no confirmed measles cases in Brazil as of June 15, 2023.
Vaccination in Brazil
Vaccination coverage in Brazil, which once reached 95%, has sharply declined in recent years. The rate of patients receiving the full immunization scheme was 59% in 2021.
Globally, although the COVID-19 pandemic affected measles vaccination, measures like social isolation and mask use potentially contributed to reducing measles cases. The incidence of the disease decreased in 2020 and 2021 but is now rising again.
“From 2021 to 2022, reported measles cases increased by 67% worldwide, and the number of countries experiencing large or disruptive outbreaks increased by 68%,” the report stated.
Because of these data, the WHO and the CDC urge increased efforts for vaccination, along with improvements in epidemiological surveillance systems, especially in developing nations. “Children everywhere have the right to be protected by the lifesaving measles vaccine, no matter where they live,” said Kate O’Brien, MD, director of immunization, vaccines, and biologicals at the WHO.
“Measles is called the virus of inequality for a good reason. It is the disease that will find and attack those who are not protected.”
This article was translated from the Medscape Portuguese edition.
The number of total reported cases rose by 18% over the same period, accounting for approximately 9 million cases and 136,000 deaths globally, mostly among children. This information comes from a new report by the World Health Organization (WHO), published in partnership with the US Centers for Disease Control and Prevention (CDC).
More Measles Outbreaks
The report also notes an increase in the number of countries experiencing significant measles outbreaks. There were 37 such countries in 2022, compared with 22 the previous year. The most affected continents were Africa and Asia.
“The rise in measles outbreaks and deaths is impressive but, unfortunately, not surprising, given the decline in vaccination rates in recent years,” said John Vertefeuille, PhD, director of the CDC’s Global Immunization Division.
Vertefeuille emphasized that measles cases anywhere in the world pose a risk to “countries and communities where people are undervaccinated.” In recent years, several regions have fallen short of their immunization targets.
Vaccination Trends
In 2022, there was a slight increase in measles vaccination after a decline exacerbated by the COVID-19 pandemic and its impact on global healthcare systems. However, 33 million children did not receive at least one dose of the vaccine last year: 22 million missed the first dose, and 11 million missed the second.
For communities to be considered protected against outbreaks, immunization coverage with the full vaccine cycle should be at least 95%. The global coverage rate for the first dose was 83%, and for the second, it was 74%.
Nevertheless, immunization recovery has not reached the poorest countries, where the immunization rate stands at 66%. Brazil is among the top 10 countries where more children missed the first dose in 2022. These nations account for over half of the 22 million unadministered vaccines. According to the report, half a million children did not receive the vaccine in Brazil.
Measles in Brazil
Brazil’s results highlight setbacks in vaccination efforts. In 2016, the country was certified to have eliminated measles, but after experiencing outbreaks in 2018, the certification was lost in 2019. In 2018, Brazil confirmed 9325 cases. The situation worsened in 2019 with 20,901 diagnoses. Since then, numbers have been decreasing: 8100 in 2020, 676 in 2021, and 44 in 2022.
Last year, four Brazilian states reported confirmed virus cases: Rio de Janeiro, Pará, São Paulo, and Amapá. Ministry of Health data indicated no confirmed measles cases in Brazil as of June 15, 2023.
Vaccination in Brazil
Vaccination coverage in Brazil, which once reached 95%, has sharply declined in recent years. The rate of patients receiving the full immunization scheme was 59% in 2021.
Globally, although the COVID-19 pandemic affected measles vaccination, measures like social isolation and mask use potentially contributed to reducing measles cases. The incidence of the disease decreased in 2020 and 2021 but is now rising again.
“From 2021 to 2022, reported measles cases increased by 67% worldwide, and the number of countries experiencing large or disruptive outbreaks increased by 68%,” the report stated.
Because of these data, the WHO and the CDC urge increased efforts for vaccination, along with improvements in epidemiological surveillance systems, especially in developing nations. “Children everywhere have the right to be protected by the lifesaving measles vaccine, no matter where they live,” said Kate O’Brien, MD, director of immunization, vaccines, and biologicals at the WHO.
“Measles is called the virus of inequality for a good reason. It is the disease that will find and attack those who are not protected.”
This article was translated from the Medscape Portuguese edition.
The number of total reported cases rose by 18% over the same period, accounting for approximately 9 million cases and 136,000 deaths globally, mostly among children. This information comes from a new report by the World Health Organization (WHO), published in partnership with the US Centers for Disease Control and Prevention (CDC).
More Measles Outbreaks
The report also notes an increase in the number of countries experiencing significant measles outbreaks. There were 37 such countries in 2022, compared with 22 the previous year. The most affected continents were Africa and Asia.
“The rise in measles outbreaks and deaths is impressive but, unfortunately, not surprising, given the decline in vaccination rates in recent years,” said John Vertefeuille, PhD, director of the CDC’s Global Immunization Division.
Vertefeuille emphasized that measles cases anywhere in the world pose a risk to “countries and communities where people are undervaccinated.” In recent years, several regions have fallen short of their immunization targets.
Vaccination Trends
In 2022, there was a slight increase in measles vaccination after a decline exacerbated by the COVID-19 pandemic and its impact on global healthcare systems. However, 33 million children did not receive at least one dose of the vaccine last year: 22 million missed the first dose, and 11 million missed the second.
For communities to be considered protected against outbreaks, immunization coverage with the full vaccine cycle should be at least 95%. The global coverage rate for the first dose was 83%, and for the second, it was 74%.
Nevertheless, immunization recovery has not reached the poorest countries, where the immunization rate stands at 66%. Brazil is among the top 10 countries where more children missed the first dose in 2022. These nations account for over half of the 22 million unadministered vaccines. According to the report, half a million children did not receive the vaccine in Brazil.
Measles in Brazil
Brazil’s results highlight setbacks in vaccination efforts. In 2016, the country was certified to have eliminated measles, but after experiencing outbreaks in 2018, the certification was lost in 2019. In 2018, Brazil confirmed 9325 cases. The situation worsened in 2019 with 20,901 diagnoses. Since then, numbers have been decreasing: 8100 in 2020, 676 in 2021, and 44 in 2022.
Last year, four Brazilian states reported confirmed virus cases: Rio de Janeiro, Pará, São Paulo, and Amapá. Ministry of Health data indicated no confirmed measles cases in Brazil as of June 15, 2023.
Vaccination in Brazil
Vaccination coverage in Brazil, which once reached 95%, has sharply declined in recent years. The rate of patients receiving the full immunization scheme was 59% in 2021.
Globally, although the COVID-19 pandemic affected measles vaccination, measures like social isolation and mask use potentially contributed to reducing measles cases. The incidence of the disease decreased in 2020 and 2021 but is now rising again.
“From 2021 to 2022, reported measles cases increased by 67% worldwide, and the number of countries experiencing large or disruptive outbreaks increased by 68%,” the report stated.
Because of these data, the WHO and the CDC urge increased efforts for vaccination, along with improvements in epidemiological surveillance systems, especially in developing nations. “Children everywhere have the right to be protected by the lifesaving measles vaccine, no matter where they live,” said Kate O’Brien, MD, director of immunization, vaccines, and biologicals at the WHO.
“Measles is called the virus of inequality for a good reason. It is the disease that will find and attack those who are not protected.”
This article was translated from the Medscape Portuguese edition.
COVID vaccines lower risk of serious illness in children
TOPLINE:
, according to a new study by the Centers for Disease Control and Prevention (CDC).
METHODOLOGY:
- SARS-CoV-2 infection can severely affect children who have certain chronic conditions.
- Researchers assessed the effectiveness of COVID-19 vaccines in preventing emergency ED visits and hospitalizations associated with the illness from July 2022 to September 2023.
- They drew data from the New Vaccine Surveillance Network, which conducts population-based, prospective surveillance for acute respiratory illness in children at seven pediatric medical centers.
- The period assessed was the first year vaccines were authorized for children aged 6 months to 4 years; during that period, several Omicron subvariants arose.
- Researchers used data from 7,434 infants and children; data included patients’ vaccine status and their test results for SARS-CoV-2.
TAKEAWAY:
- Of the 7,434 infants and children who had an acute respiratory illness and were hospitalized or visited the ED, 387 had COVID-19.
- Children who received two doses of a COVID-19 vaccine were 40% less likely to have a COVID-19-associated hospitalization or ED visit compared with unvaccinated youth.
- One dose of a COVID-19 vaccine reduced ED visits and hospitalizations by 31%.
IN PRACTICE:
“The findings in this report support the recommendation for COVID-19 vaccination for all children aged ≥6 months and highlight the importance of completion of a primary series for young children,” the researchers reported.
SOURCE:
The study was led by Heidi L. Moline, MD, of the CDC.
LIMITATIONS:
Because the number of children with antibodies and immunity against SARS-CoV-2 has grown, vaccine effectiveness rates in the study may no longer be as relevant. Children with preexisting chronic conditions may be more likely to be vaccinated and receive medical attention. The low rates of vaccination may have prevented researchers from conducting a more detailed analysis. The Pfizer-BioNTech vaccine requires three doses, whereas Moderna’s requires two doses; this may have skewed the estimated efficacy of the Pfizer-BioNTech vaccine.
DISCLOSURES:
The authors report a variety of potential conflicts of interest, which are detailed in the article.
A version of this article appeared on Medscape.com.
TOPLINE:
, according to a new study by the Centers for Disease Control and Prevention (CDC).
METHODOLOGY:
- SARS-CoV-2 infection can severely affect children who have certain chronic conditions.
- Researchers assessed the effectiveness of COVID-19 vaccines in preventing emergency ED visits and hospitalizations associated with the illness from July 2022 to September 2023.
- They drew data from the New Vaccine Surveillance Network, which conducts population-based, prospective surveillance for acute respiratory illness in children at seven pediatric medical centers.
- The period assessed was the first year vaccines were authorized for children aged 6 months to 4 years; during that period, several Omicron subvariants arose.
- Researchers used data from 7,434 infants and children; data included patients’ vaccine status and their test results for SARS-CoV-2.
TAKEAWAY:
- Of the 7,434 infants and children who had an acute respiratory illness and were hospitalized or visited the ED, 387 had COVID-19.
- Children who received two doses of a COVID-19 vaccine were 40% less likely to have a COVID-19-associated hospitalization or ED visit compared with unvaccinated youth.
- One dose of a COVID-19 vaccine reduced ED visits and hospitalizations by 31%.
IN PRACTICE:
“The findings in this report support the recommendation for COVID-19 vaccination for all children aged ≥6 months and highlight the importance of completion of a primary series for young children,” the researchers reported.
SOURCE:
The study was led by Heidi L. Moline, MD, of the CDC.
LIMITATIONS:
Because the number of children with antibodies and immunity against SARS-CoV-2 has grown, vaccine effectiveness rates in the study may no longer be as relevant. Children with preexisting chronic conditions may be more likely to be vaccinated and receive medical attention. The low rates of vaccination may have prevented researchers from conducting a more detailed analysis. The Pfizer-BioNTech vaccine requires three doses, whereas Moderna’s requires two doses; this may have skewed the estimated efficacy of the Pfizer-BioNTech vaccine.
DISCLOSURES:
The authors report a variety of potential conflicts of interest, which are detailed in the article.
A version of this article appeared on Medscape.com.
TOPLINE:
, according to a new study by the Centers for Disease Control and Prevention (CDC).
METHODOLOGY:
- SARS-CoV-2 infection can severely affect children who have certain chronic conditions.
- Researchers assessed the effectiveness of COVID-19 vaccines in preventing emergency ED visits and hospitalizations associated with the illness from July 2022 to September 2023.
- They drew data from the New Vaccine Surveillance Network, which conducts population-based, prospective surveillance for acute respiratory illness in children at seven pediatric medical centers.
- The period assessed was the first year vaccines were authorized for children aged 6 months to 4 years; during that period, several Omicron subvariants arose.
- Researchers used data from 7,434 infants and children; data included patients’ vaccine status and their test results for SARS-CoV-2.
TAKEAWAY:
- Of the 7,434 infants and children who had an acute respiratory illness and were hospitalized or visited the ED, 387 had COVID-19.
- Children who received two doses of a COVID-19 vaccine were 40% less likely to have a COVID-19-associated hospitalization or ED visit compared with unvaccinated youth.
- One dose of a COVID-19 vaccine reduced ED visits and hospitalizations by 31%.
IN PRACTICE:
“The findings in this report support the recommendation for COVID-19 vaccination for all children aged ≥6 months and highlight the importance of completion of a primary series for young children,” the researchers reported.
SOURCE:
The study was led by Heidi L. Moline, MD, of the CDC.
LIMITATIONS:
Because the number of children with antibodies and immunity against SARS-CoV-2 has grown, vaccine effectiveness rates in the study may no longer be as relevant. Children with preexisting chronic conditions may be more likely to be vaccinated and receive medical attention. The low rates of vaccination may have prevented researchers from conducting a more detailed analysis. The Pfizer-BioNTech vaccine requires three doses, whereas Moderna’s requires two doses; this may have skewed the estimated efficacy of the Pfizer-BioNTech vaccine.
DISCLOSURES:
The authors report a variety of potential conflicts of interest, which are detailed in the article.
A version of this article appeared on Medscape.com.
Patients exposed to HIV, hepatitis at Massachusetts hospital
The negligent administration of intravenous medications during endoscopy procedures performed between June 14, 2021, and April 19, 2023, at Salem Hospital, located about 20 miles northeast of Boston, has caused a “heightened risk of exposure to these harmful life-altering and life-threatening infections,” according to the lawsuit filed at Suffolk County Superior Court in Boston by Keches Law Group on behalf of plaintiff Melinda Cashman and others.
Although patients were notified in early November of their potential exposure, it could take months or even years to determine if infection has occurred. Attorneys for Ms. Cashman claim that the plaintiff “suffered and will continue to suffer severe emotional distress and anguish” as a result of the associated risks.
The lawyers argue that Ms. Cashman and others like her may “suffer permanent injuries,” along with “extreme anxiety and decreased quality of life.” They are seeking monetary damages to offset disruptions to relationships, increased medical bills, and any mental health therapy required.
Outreach to potentially affected patients began after the hospital was made aware, earlier this year, of an “isolated practice” that could have led to viral transmission, according to a statement from Mass General Brigham, but there is no evidence to date of any infections resulting from this incident. “We sincerely apologize to those who have been impacted and we remain committed to delivering high-quality, compassionate healthcare to our community.”
Hepatitis B and C are both treatable with antiviral mediations, and hepatitis C is curable in 95% of cases, according to the Centers for Disease Control and Prevention. HIV, although not curable, can be managed with antiretroviral therapy.
Mass General Brigham is working with the Massachusetts Department of Public Health, which will conduct an onsite investigation into quality-control practices. Affected patients can reach out to a clinician-staffed hotline with questions and receive free screening for the viruses, hospital officials report.
A version of this article appeared on Medscape.com.
The negligent administration of intravenous medications during endoscopy procedures performed between June 14, 2021, and April 19, 2023, at Salem Hospital, located about 20 miles northeast of Boston, has caused a “heightened risk of exposure to these harmful life-altering and life-threatening infections,” according to the lawsuit filed at Suffolk County Superior Court in Boston by Keches Law Group on behalf of plaintiff Melinda Cashman and others.
Although patients were notified in early November of their potential exposure, it could take months or even years to determine if infection has occurred. Attorneys for Ms. Cashman claim that the plaintiff “suffered and will continue to suffer severe emotional distress and anguish” as a result of the associated risks.
The lawyers argue that Ms. Cashman and others like her may “suffer permanent injuries,” along with “extreme anxiety and decreased quality of life.” They are seeking monetary damages to offset disruptions to relationships, increased medical bills, and any mental health therapy required.
Outreach to potentially affected patients began after the hospital was made aware, earlier this year, of an “isolated practice” that could have led to viral transmission, according to a statement from Mass General Brigham, but there is no evidence to date of any infections resulting from this incident. “We sincerely apologize to those who have been impacted and we remain committed to delivering high-quality, compassionate healthcare to our community.”
Hepatitis B and C are both treatable with antiviral mediations, and hepatitis C is curable in 95% of cases, according to the Centers for Disease Control and Prevention. HIV, although not curable, can be managed with antiretroviral therapy.
Mass General Brigham is working with the Massachusetts Department of Public Health, which will conduct an onsite investigation into quality-control practices. Affected patients can reach out to a clinician-staffed hotline with questions and receive free screening for the viruses, hospital officials report.
A version of this article appeared on Medscape.com.
The negligent administration of intravenous medications during endoscopy procedures performed between June 14, 2021, and April 19, 2023, at Salem Hospital, located about 20 miles northeast of Boston, has caused a “heightened risk of exposure to these harmful life-altering and life-threatening infections,” according to the lawsuit filed at Suffolk County Superior Court in Boston by Keches Law Group on behalf of plaintiff Melinda Cashman and others.
Although patients were notified in early November of their potential exposure, it could take months or even years to determine if infection has occurred. Attorneys for Ms. Cashman claim that the plaintiff “suffered and will continue to suffer severe emotional distress and anguish” as a result of the associated risks.
The lawyers argue that Ms. Cashman and others like her may “suffer permanent injuries,” along with “extreme anxiety and decreased quality of life.” They are seeking monetary damages to offset disruptions to relationships, increased medical bills, and any mental health therapy required.
Outreach to potentially affected patients began after the hospital was made aware, earlier this year, of an “isolated practice” that could have led to viral transmission, according to a statement from Mass General Brigham, but there is no evidence to date of any infections resulting from this incident. “We sincerely apologize to those who have been impacted and we remain committed to delivering high-quality, compassionate healthcare to our community.”
Hepatitis B and C are both treatable with antiviral mediations, and hepatitis C is curable in 95% of cases, according to the Centers for Disease Control and Prevention. HIV, although not curable, can be managed with antiretroviral therapy.
Mass General Brigham is working with the Massachusetts Department of Public Health, which will conduct an onsite investigation into quality-control practices. Affected patients can reach out to a clinician-staffed hotline with questions and receive free screening for the viruses, hospital officials report.
A version of this article appeared on Medscape.com.
COVID vaccination protects B cell–deficient patients through T-cell responses
TOPLINE:
In individuals with low B-cell counts, T cells have enhanced responses to COVID-19 vaccination and may help prevent severe disease after infection.
METHODOLOGY:
- How the immune systems of B cell–deficient patients respond to SARS-CoV-2 infection and vaccination is not fully understood.
- Researchers evaluated anti–SARS-CoV-2 T-cell responses in 33 patients treated with rituximab (RTX), 12 patients with common variable immune deficiency, and 44 controls.
- The study analyzed effector and memory CD4+ and CD8+ T-cell responses to SARS-CoV-2 after infection and vaccination.
TAKEAWAY:
- All B cell–deficient individuals (those treated with RTX or those with a diagnosis of common variable immune deficiency) had increased effector and memory T-cell responses after SARS-CoV-2 vaccination, compared with controls.
- Patients treated with RTX who were vaccinated against COVID-19 had 4.8-fold reduced odds of moderate or severe disease. (These data were not available for patients with common variable immune deficiency.)
- RTX treatment was associated with a decrease in preexisting T-cell immunity in unvaccinated patients, regardless of prior infection with SARS-CoV-2.
- This association was not found in vaccinated patients treated with RTX.
IN PRACTICE:
“[These findings] provide support for vaccination in this vulnerable population and demonstrate the potential benefit of vaccine-induced CD8+ T-cell responses on reducing disease severity from SARS-CoV-2 infection in the absence of spike protein–specific antibodies,” the authors wrote.
SOURCE:
The study was published online on November 29 in Science Translational Medicine. The first author is Reza Zonozi, MD, who conducted the research while at Massachusetts General Hospital, Boston, and is now in private practice in northern Virginia.
LIMITATIONS:
Researchers did not obtain specimens from patients with common variable immune deficiency after SARS-CoV-2 infection. Only a small subset of immunophenotyped participants had subsequent SARS-CoV-2 infection.
DISCLOSURES:
The research was supported by grants from the National Institutes of Health, the Centers for Disease Control and Prevention, the Howard Hughes Medical Institute, the Ragon Institute of Massachusetts General Hospital, Massachusetts Institute of Technology, and Harvard Medical School, the Mark and Lisa Schwartz Foundation and E. Schwartz; the Lambertus Family Foundation; and S. Edgerly and P. Edgerly. Four authors reported relationships with pharmaceutical companies including AbbVie, Bristol-Myers Squibb, Boehringer Ingelheim, Gilead Sciences, Merck, and Pfizer.
A version of this article first appeared on Medscape.com.
TOPLINE:
In individuals with low B-cell counts, T cells have enhanced responses to COVID-19 vaccination and may help prevent severe disease after infection.
METHODOLOGY:
- How the immune systems of B cell–deficient patients respond to SARS-CoV-2 infection and vaccination is not fully understood.
- Researchers evaluated anti–SARS-CoV-2 T-cell responses in 33 patients treated with rituximab (RTX), 12 patients with common variable immune deficiency, and 44 controls.
- The study analyzed effector and memory CD4+ and CD8+ T-cell responses to SARS-CoV-2 after infection and vaccination.
TAKEAWAY:
- All B cell–deficient individuals (those treated with RTX or those with a diagnosis of common variable immune deficiency) had increased effector and memory T-cell responses after SARS-CoV-2 vaccination, compared with controls.
- Patients treated with RTX who were vaccinated against COVID-19 had 4.8-fold reduced odds of moderate or severe disease. (These data were not available for patients with common variable immune deficiency.)
- RTX treatment was associated with a decrease in preexisting T-cell immunity in unvaccinated patients, regardless of prior infection with SARS-CoV-2.
- This association was not found in vaccinated patients treated with RTX.
IN PRACTICE:
“[These findings] provide support for vaccination in this vulnerable population and demonstrate the potential benefit of vaccine-induced CD8+ T-cell responses on reducing disease severity from SARS-CoV-2 infection in the absence of spike protein–specific antibodies,” the authors wrote.
SOURCE:
The study was published online on November 29 in Science Translational Medicine. The first author is Reza Zonozi, MD, who conducted the research while at Massachusetts General Hospital, Boston, and is now in private practice in northern Virginia.
LIMITATIONS:
Researchers did not obtain specimens from patients with common variable immune deficiency after SARS-CoV-2 infection. Only a small subset of immunophenotyped participants had subsequent SARS-CoV-2 infection.
DISCLOSURES:
The research was supported by grants from the National Institutes of Health, the Centers for Disease Control and Prevention, the Howard Hughes Medical Institute, the Ragon Institute of Massachusetts General Hospital, Massachusetts Institute of Technology, and Harvard Medical School, the Mark and Lisa Schwartz Foundation and E. Schwartz; the Lambertus Family Foundation; and S. Edgerly and P. Edgerly. Four authors reported relationships with pharmaceutical companies including AbbVie, Bristol-Myers Squibb, Boehringer Ingelheim, Gilead Sciences, Merck, and Pfizer.
A version of this article first appeared on Medscape.com.
TOPLINE:
In individuals with low B-cell counts, T cells have enhanced responses to COVID-19 vaccination and may help prevent severe disease after infection.
METHODOLOGY:
- How the immune systems of B cell–deficient patients respond to SARS-CoV-2 infection and vaccination is not fully understood.
- Researchers evaluated anti–SARS-CoV-2 T-cell responses in 33 patients treated with rituximab (RTX), 12 patients with common variable immune deficiency, and 44 controls.
- The study analyzed effector and memory CD4+ and CD8+ T-cell responses to SARS-CoV-2 after infection and vaccination.
TAKEAWAY:
- All B cell–deficient individuals (those treated with RTX or those with a diagnosis of common variable immune deficiency) had increased effector and memory T-cell responses after SARS-CoV-2 vaccination, compared with controls.
- Patients treated with RTX who were vaccinated against COVID-19 had 4.8-fold reduced odds of moderate or severe disease. (These data were not available for patients with common variable immune deficiency.)
- RTX treatment was associated with a decrease in preexisting T-cell immunity in unvaccinated patients, regardless of prior infection with SARS-CoV-2.
- This association was not found in vaccinated patients treated with RTX.
IN PRACTICE:
“[These findings] provide support for vaccination in this vulnerable population and demonstrate the potential benefit of vaccine-induced CD8+ T-cell responses on reducing disease severity from SARS-CoV-2 infection in the absence of spike protein–specific antibodies,” the authors wrote.
SOURCE:
The study was published online on November 29 in Science Translational Medicine. The first author is Reza Zonozi, MD, who conducted the research while at Massachusetts General Hospital, Boston, and is now in private practice in northern Virginia.
LIMITATIONS:
Researchers did not obtain specimens from patients with common variable immune deficiency after SARS-CoV-2 infection. Only a small subset of immunophenotyped participants had subsequent SARS-CoV-2 infection.
DISCLOSURES:
The research was supported by grants from the National Institutes of Health, the Centers for Disease Control and Prevention, the Howard Hughes Medical Institute, the Ragon Institute of Massachusetts General Hospital, Massachusetts Institute of Technology, and Harvard Medical School, the Mark and Lisa Schwartz Foundation and E. Schwartz; the Lambertus Family Foundation; and S. Edgerly and P. Edgerly. Four authors reported relationships with pharmaceutical companies including AbbVie, Bristol-Myers Squibb, Boehringer Ingelheim, Gilead Sciences, Merck, and Pfizer.
A version of this article first appeared on Medscape.com.
Neutrophilic Dermatosis of the Dorsal Hand: A Distinctive Variant of Sweet Syndrome
To the Editor:
Neutrophilic dermatosis of the dorsal hand (NDDH) is an uncommon reactive neutrophilic dermatosis that presents as a painful, enlarging, ulcerative nodule. It often is misdiagnosed and initially treated as an infection. Similar to other neutrophilic dermatoses, it is associated with underlying infections, inflammatory conditions, and malignancies. Neutrophilic dermatosis of the dorsal hand is considered a subset of Sweet syndrome (SS); we highlight similarities and differences between NDDH and SS, reporting the case of a 66-year-old man without systemic symptoms who developed NDDH on the right hand.
A 66-year-old man presented with a progressively enlarging, painful, ulcerative, 2-cm nodule on the right hand following mechanical trauma 2 weeks prior (Figure 1). He was afebrile with no remarkable medical history. Laboratory evaluation revealed an erythrocyte sedimentation rate (ESR) of 20 mm/h (reference range, 0-10 mm/h) and C-reactive protein (CRP) level of 3.52 mg/dL (reference range, 0-0.5 mg/dL) without leukocytosis; both were not remarkably elevated when adjusted for age.1,2 The clinical differential diagnosis was broad and included pyoderma with evolving cellulitis, neutrophilic dermatosis, atypical mycobacterial infection, subcutaneous or deep fungal infection, squamous cell carcinoma, cutaneous lymphoma, and metastasis. Due to the rapid development of the lesion, initial treatment focused on a bacterial infection, but there was no improvement on antibiotics and wound cultures were negative. The ulcerative nodule was biopsied, and histopathology demonstrated abundant neutrophilic inflammation, endothelial swelling, and leukocytoclasis without microorganisms (Figure 2). Tissue cultures for bacteria, fungi, and atypical mycobacteria were negative. A diagnosis of NDDH was made based on clinical and histologic findings. The wound improved with a 3-week course of oral prednisone.
Neutrophilic dermatosis of the dorsal hand is a subset of reactive neutrophilic dermatoses, which includes SS (acute febrile neutrophilic dermatosis) and pyoderma gangrenosum. It is described as a localized variant of SS, with similar associated underlying inflammatory, neoplastic conditions and laboratory findings.3 However, NDDH has characteristic features that differ from classic SS. Neutrophilic dermatosis of the dorsal hand typically presents as painful papules, pustules, or ulcers that progress to become larger ulcers, plaques, and nodules. The clinical appearance may more closely resemble pyoderma gangrenosum or atypical SS, with ulceration frequently present. Pathergy also may be demonstrated in NDDH, similar to our patient. The average age of presentation for NDDH is 60 years, which is older than the average age for SS or pyoderma gangrenosum.3 Similar to other neutrophilic dermatoses, NDDH responds well to oral steroids or steroid-sparing immunosuppressants such as dapsone, colchicine, azathioprine, or tetracycline antibiotics.4
The criteria for SS are well established5,6 and may be used for the diagnosis of NDDH, taking into account the localization of lesions to the dorsal aspect of the hands. The diagnostic criteria for SS include fulfillment of both major and at least 2 of 4 minor criteria. The 2 major criteria include rapid presentation of skin lesions and neutrophilic dermal infiltrate on biopsy. Minor criteria are defined as the following: (1) preceding nonspecific respiratory or gastrointestinal tract infection, inflammatory conditions, underlying malignancy, or pregnancy; (2) fever; (3) excellent response to steroids; and (4) 3 of the 4 of the following laboratory abnormalities: elevated CRP, ESR, leukocytosis, or left shift in complete blood cell count. Our patient met both major criteria and only 1 minor criterion—excellent response to systemic corticosteroids. Nofal et al7 advocated for revised diagnostic criteria for SS, with one suggestion utilizing only the 2 major criteria being necessary for diagnosis. Given that serum inflammatory markers may not be as elevated in NDDH compared to SS,3,7,8 meeting the major criteria alone may be a better way to diagnose NDDH, as in our patient.
Our patient presented with an expanding ulcerating nodule on the hand that elicited a wide list of differential diagnoses to include infections and neoplasms. Rapid development, localization to the dorsal aspect of the hand, and treatment resistance to antibiotics may help the clinician consider a diagnosis of NDDH, which should be confirmed by a biopsy. Similar to other neutrophilic dermatoses, an underlying malignancy or inflammatory condition should be sought out. Neutrophilic dermatosis of the dorsal hand responds well to systemic steroids, though recurrences may occur.
- Miller A, Green M, Robinson D. Simple rule for calculating normal erythrocyte sedimentation rate. Br Med (Clinical Res Ed). 1983;286:226.
- Wyczalkowska-Tomasik A, Czarkowska-Paczek B, Zielenkiewicz M, et al. Inflammatory markers change with age, but do not fall beyond reported normal ranges. Arch Immunol Ther Exp (Warsz). 2016;64:249-254.
- Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and Sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63.
- Gaulding J, Kohen LL. Neutrophilic dermatosis of the dorsal hands. J Am Acad Dermatol. 2017; 76(6 suppl 1):AB178.
- Sweet RD. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:349-356.
- Su WP, Liu HN. Diagnostic criteria for Sweet’s syndrome. Cutis. 1986;37:167-174.
- Nofal A, Abdelmaksoud A, Amer H, et al. Sweet’s syndrome: diagnostic criteria revisited. J Dtsch Dermatol Ges. 2017;15:1081-1088.
- Wolf R, Tüzün Y. Acral manifestations of Sweet syndrome (neutrophilic dermatosis of the hands). Clin Dermatol. 2017;35:81-84.
To the Editor:
Neutrophilic dermatosis of the dorsal hand (NDDH) is an uncommon reactive neutrophilic dermatosis that presents as a painful, enlarging, ulcerative nodule. It often is misdiagnosed and initially treated as an infection. Similar to other neutrophilic dermatoses, it is associated with underlying infections, inflammatory conditions, and malignancies. Neutrophilic dermatosis of the dorsal hand is considered a subset of Sweet syndrome (SS); we highlight similarities and differences between NDDH and SS, reporting the case of a 66-year-old man without systemic symptoms who developed NDDH on the right hand.

A 66-year-old man presented with a progressively enlarging, painful, ulcerative, 2-cm nodule on the right hand following mechanical trauma 2 weeks prior (Figure 1). He was afebrile with no remarkable medical history. Laboratory evaluation revealed an erythrocyte sedimentation rate (ESR) of 20 mm/h (reference range, 0-10 mm/h) and C-reactive protein (CRP) level of 3.52 mg/dL (reference range, 0-0.5 mg/dL) without leukocytosis; both were not remarkably elevated when adjusted for age.1,2 The clinical differential diagnosis was broad and included pyoderma with evolving cellulitis, neutrophilic dermatosis, atypical mycobacterial infection, subcutaneous or deep fungal infection, squamous cell carcinoma, cutaneous lymphoma, and metastasis. Due to the rapid development of the lesion, initial treatment focused on a bacterial infection, but there was no improvement on antibiotics and wound cultures were negative. The ulcerative nodule was biopsied, and histopathology demonstrated abundant neutrophilic inflammation, endothelial swelling, and leukocytoclasis without microorganisms (Figure 2). Tissue cultures for bacteria, fungi, and atypical mycobacteria were negative. A diagnosis of NDDH was made based on clinical and histologic findings. The wound improved with a 3-week course of oral prednisone.

Neutrophilic dermatosis of the dorsal hand is a subset of reactive neutrophilic dermatoses, which includes SS (acute febrile neutrophilic dermatosis) and pyoderma gangrenosum. It is described as a localized variant of SS, with similar associated underlying inflammatory, neoplastic conditions and laboratory findings.3 However, NDDH has characteristic features that differ from classic SS. Neutrophilic dermatosis of the dorsal hand typically presents as painful papules, pustules, or ulcers that progress to become larger ulcers, plaques, and nodules. The clinical appearance may more closely resemble pyoderma gangrenosum or atypical SS, with ulceration frequently present. Pathergy also may be demonstrated in NDDH, similar to our patient. The average age of presentation for NDDH is 60 years, which is older than the average age for SS or pyoderma gangrenosum.3 Similar to other neutrophilic dermatoses, NDDH responds well to oral steroids or steroid-sparing immunosuppressants such as dapsone, colchicine, azathioprine, or tetracycline antibiotics.4
The criteria for SS are well established5,6 and may be used for the diagnosis of NDDH, taking into account the localization of lesions to the dorsal aspect of the hands. The diagnostic criteria for SS include fulfillment of both major and at least 2 of 4 minor criteria. The 2 major criteria include rapid presentation of skin lesions and neutrophilic dermal infiltrate on biopsy. Minor criteria are defined as the following: (1) preceding nonspecific respiratory or gastrointestinal tract infection, inflammatory conditions, underlying malignancy, or pregnancy; (2) fever; (3) excellent response to steroids; and (4) 3 of the 4 of the following laboratory abnormalities: elevated CRP, ESR, leukocytosis, or left shift in complete blood cell count. Our patient met both major criteria and only 1 minor criterion—excellent response to systemic corticosteroids. Nofal et al7 advocated for revised diagnostic criteria for SS, with one suggestion utilizing only the 2 major criteria being necessary for diagnosis. Given that serum inflammatory markers may not be as elevated in NDDH compared to SS,3,7,8 meeting the major criteria alone may be a better way to diagnose NDDH, as in our patient.
Our patient presented with an expanding ulcerating nodule on the hand that elicited a wide list of differential diagnoses to include infections and neoplasms. Rapid development, localization to the dorsal aspect of the hand, and treatment resistance to antibiotics may help the clinician consider a diagnosis of NDDH, which should be confirmed by a biopsy. Similar to other neutrophilic dermatoses, an underlying malignancy or inflammatory condition should be sought out. Neutrophilic dermatosis of the dorsal hand responds well to systemic steroids, though recurrences may occur.
To the Editor:
Neutrophilic dermatosis of the dorsal hand (NDDH) is an uncommon reactive neutrophilic dermatosis that presents as a painful, enlarging, ulcerative nodule. It often is misdiagnosed and initially treated as an infection. Similar to other neutrophilic dermatoses, it is associated with underlying infections, inflammatory conditions, and malignancies. Neutrophilic dermatosis of the dorsal hand is considered a subset of Sweet syndrome (SS); we highlight similarities and differences between NDDH and SS, reporting the case of a 66-year-old man without systemic symptoms who developed NDDH on the right hand.

A 66-year-old man presented with a progressively enlarging, painful, ulcerative, 2-cm nodule on the right hand following mechanical trauma 2 weeks prior (Figure 1). He was afebrile with no remarkable medical history. Laboratory evaluation revealed an erythrocyte sedimentation rate (ESR) of 20 mm/h (reference range, 0-10 mm/h) and C-reactive protein (CRP) level of 3.52 mg/dL (reference range, 0-0.5 mg/dL) without leukocytosis; both were not remarkably elevated when adjusted for age.1,2 The clinical differential diagnosis was broad and included pyoderma with evolving cellulitis, neutrophilic dermatosis, atypical mycobacterial infection, subcutaneous or deep fungal infection, squamous cell carcinoma, cutaneous lymphoma, and metastasis. Due to the rapid development of the lesion, initial treatment focused on a bacterial infection, but there was no improvement on antibiotics and wound cultures were negative. The ulcerative nodule was biopsied, and histopathology demonstrated abundant neutrophilic inflammation, endothelial swelling, and leukocytoclasis without microorganisms (Figure 2). Tissue cultures for bacteria, fungi, and atypical mycobacteria were negative. A diagnosis of NDDH was made based on clinical and histologic findings. The wound improved with a 3-week course of oral prednisone.

Neutrophilic dermatosis of the dorsal hand is a subset of reactive neutrophilic dermatoses, which includes SS (acute febrile neutrophilic dermatosis) and pyoderma gangrenosum. It is described as a localized variant of SS, with similar associated underlying inflammatory, neoplastic conditions and laboratory findings.3 However, NDDH has characteristic features that differ from classic SS. Neutrophilic dermatosis of the dorsal hand typically presents as painful papules, pustules, or ulcers that progress to become larger ulcers, plaques, and nodules. The clinical appearance may more closely resemble pyoderma gangrenosum or atypical SS, with ulceration frequently present. Pathergy also may be demonstrated in NDDH, similar to our patient. The average age of presentation for NDDH is 60 years, which is older than the average age for SS or pyoderma gangrenosum.3 Similar to other neutrophilic dermatoses, NDDH responds well to oral steroids or steroid-sparing immunosuppressants such as dapsone, colchicine, azathioprine, or tetracycline antibiotics.4
The criteria for SS are well established5,6 and may be used for the diagnosis of NDDH, taking into account the localization of lesions to the dorsal aspect of the hands. The diagnostic criteria for SS include fulfillment of both major and at least 2 of 4 minor criteria. The 2 major criteria include rapid presentation of skin lesions and neutrophilic dermal infiltrate on biopsy. Minor criteria are defined as the following: (1) preceding nonspecific respiratory or gastrointestinal tract infection, inflammatory conditions, underlying malignancy, or pregnancy; (2) fever; (3) excellent response to steroids; and (4) 3 of the 4 of the following laboratory abnormalities: elevated CRP, ESR, leukocytosis, or left shift in complete blood cell count. Our patient met both major criteria and only 1 minor criterion—excellent response to systemic corticosteroids. Nofal et al7 advocated for revised diagnostic criteria for SS, with one suggestion utilizing only the 2 major criteria being necessary for diagnosis. Given that serum inflammatory markers may not be as elevated in NDDH compared to SS,3,7,8 meeting the major criteria alone may be a better way to diagnose NDDH, as in our patient.
Our patient presented with an expanding ulcerating nodule on the hand that elicited a wide list of differential diagnoses to include infections and neoplasms. Rapid development, localization to the dorsal aspect of the hand, and treatment resistance to antibiotics may help the clinician consider a diagnosis of NDDH, which should be confirmed by a biopsy. Similar to other neutrophilic dermatoses, an underlying malignancy or inflammatory condition should be sought out. Neutrophilic dermatosis of the dorsal hand responds well to systemic steroids, though recurrences may occur.
- Miller A, Green M, Robinson D. Simple rule for calculating normal erythrocyte sedimentation rate. Br Med (Clinical Res Ed). 1983;286:226.
- Wyczalkowska-Tomasik A, Czarkowska-Paczek B, Zielenkiewicz M, et al. Inflammatory markers change with age, but do not fall beyond reported normal ranges. Arch Immunol Ther Exp (Warsz). 2016;64:249-254.
- Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and Sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63.
- Gaulding J, Kohen LL. Neutrophilic dermatosis of the dorsal hands. J Am Acad Dermatol. 2017; 76(6 suppl 1):AB178.
- Sweet RD. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:349-356.
- Su WP, Liu HN. Diagnostic criteria for Sweet’s syndrome. Cutis. 1986;37:167-174.
- Nofal A, Abdelmaksoud A, Amer H, et al. Sweet’s syndrome: diagnostic criteria revisited. J Dtsch Dermatol Ges. 2017;15:1081-1088.
- Wolf R, Tüzün Y. Acral manifestations of Sweet syndrome (neutrophilic dermatosis of the hands). Clin Dermatol. 2017;35:81-84.
- Miller A, Green M, Robinson D. Simple rule for calculating normal erythrocyte sedimentation rate. Br Med (Clinical Res Ed). 1983;286:226.
- Wyczalkowska-Tomasik A, Czarkowska-Paczek B, Zielenkiewicz M, et al. Inflammatory markers change with age, but do not fall beyond reported normal ranges. Arch Immunol Ther Exp (Warsz). 2016;64:249-254.
- Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and Sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63.
- Gaulding J, Kohen LL. Neutrophilic dermatosis of the dorsal hands. J Am Acad Dermatol. 2017; 76(6 suppl 1):AB178.
- Sweet RD. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:349-356.
- Su WP, Liu HN. Diagnostic criteria for Sweet’s syndrome. Cutis. 1986;37:167-174.
- Nofal A, Abdelmaksoud A, Amer H, et al. Sweet’s syndrome: diagnostic criteria revisited. J Dtsch Dermatol Ges. 2017;15:1081-1088.
- Wolf R, Tüzün Y. Acral manifestations of Sweet syndrome (neutrophilic dermatosis of the hands). Clin Dermatol. 2017;35:81-84.
Practice Points
- Neutrophilic dermatosis of the dorsal hand (NDDH) is a reactive neutrophilic dermatosis that includes Sweet syndrome (SS) and pyoderma gangrenosum.
- Localization to the dorsal aspect of the hand, presence of ulcerative nodules, and older age at onset are characteristic features of NDDH.
- Meeting the major criteria alone for SS may be a more sensitive way to diagnose NDDH, as serum inflammatory markers may not be remarkably elevated in this condition.
All-oral regimen succeeds for rifampin-resistant tuberculosis
A combination oral-only therapy of bedaquiline, pretomanid, and linezolid was significantly more effective than standard care in preventing unfavorable outcomes in patients with treatment-resistant tuberculosis, based on data from more than 500 individuals.
, and data on the use of new and repurposed drug combinations are lacking, wrote Bern-Thomas Nyang’wa, MBBS, of Médecins Sans Frontières, Amsterdam, and colleagues.
In a study known as the TB-PRACTECAL trial, the researchers enrolled 552 pulmonary rifampin-resistant tuberculosis patients aged 15 years and older to examine several new and repurposed drug combinations. The participants were randomized in a 1:1:1:1 ratio to treatment with 36-80 weeks of standard care; 24-week oral bedaquiline, pretomanid, and linezolid (BPaL); BPaL plus clofazimine (BPaLC); or BPaL plus moxifloxacin (BPaLM) . This was followed by stage two of the trial, in which participants were randomized 1:1 to receive standard care or BPaLM. The current study, published in The Lancet Respiratory Medicine, reported the stage two findings; the primary outcome was a composite of unfavorable outcomes at 72 weeks including death, treatment failure, treatment discontinuation, recurrence of tuberculosis, or loss to follow-up.
The modified intent-to-treat population included 138 patients in the BPaLM group and 137 patients in the standard care group. In this population, 56 (41%) of 137 participants in the standard care group and 16 (12%) of 137 participants in the BPaLM group met criteria for the unfavorable outcome at 72 weeks; noninferiority and superiority were significantly greater in the BPaLM group (P < .0001).
Early discontinuation was the main reason patients met the unfavorable outcome criteria (89% of standard care patients and 69% of BPaLM patients); adverse events accounted for 23% of discontinuations in the standard care group and 64% of discontinuations in the BPaLM group.
However, fewer patients in the BPaLM group experienced grade 3 or higher adverse events compared with the standard care group (23% vs. 48%). The most common adverse events included hepatic disorders, cardiac disorders, and anemia.
In addition, all subgroup analyses favored BPaLM over standard care at 72 weeks including subgroups based on sex, age, disease severity, re-treatment status, and smoking status.
The findings were limited by several factors including the changes to standard of care over the course of the study, potential bias because the study was stopped for efficacy, and inclusion of loss to follow-up as part of the composite unfavorable outcome, the researchers noted.
Remaining research questions include the optimal dose of linezolid, whether use of alternative fluoroquinolones would yield similar results, and whether the results would generalize to populations including children, pregnant women, and patients with extrapulmonary tuberculosis, they added.
However, the results support BPaLM as the preferred treatment for adults and adolescents with pulmonary rifampin-resistant TB, the researchers concluded.
BPaLM poised to improve TB care
Before 2020, treatment for rifampin-resistant tuberculosis was 9-20 months in duration, toxic, and inadequately effective, and new treatment regimens are urgently needed, Mary Jo Farmer, MD, a pulmonary and critical care specialist at the University of Massachusetts Baystate Health Regional Campus, Springfield, said in an interview.
“The BPaL-based regimens perform better than the 9- to 20-month standard of care, are shorter in duration, have a lower pill burden, improve quality of life, and are cost-effective,” she said. “The BPaL regimens have the potential to improve outcomes for thousands of patients with rifampin-resistant tuberculosis.”
“The 24-week oral regimen consisting of bedaquiline, pretomanid, linezolid and moxifloxacin is noninferior to standard of care for treatment of patients with pulmonary rifampin-resistant tuberculosis, and this BPaLM regimen was added to the WHO guidance for treatment of this condition in 2022,” said Dr. Farmer, who was not involved in the study. “It remains to be seen if BPaLM will become the preferred regimen for adolescents and adults with pulmonary rifampin-resistant tuberculosis,” she said.
Dr. Farmer agreed with the study authors that the optimal dose of linezolid, optimal duration of treatment, and the role of dose reduction remain unknown, and pharmacokinetic studies are needed to identify these parameters.
The study was supported by Médecins Sans Frontières. TB Alliance donated pretomanid to the study prior to its commercialization. The researchers had no financial conflicts to disclose. Dr. Farmer had no financial conflicts to disclose, but serves on the editorial advisory board of CHEST Physician.
A combination oral-only therapy of bedaquiline, pretomanid, and linezolid was significantly more effective than standard care in preventing unfavorable outcomes in patients with treatment-resistant tuberculosis, based on data from more than 500 individuals.
, and data on the use of new and repurposed drug combinations are lacking, wrote Bern-Thomas Nyang’wa, MBBS, of Médecins Sans Frontières, Amsterdam, and colleagues.
In a study known as the TB-PRACTECAL trial, the researchers enrolled 552 pulmonary rifampin-resistant tuberculosis patients aged 15 years and older to examine several new and repurposed drug combinations. The participants were randomized in a 1:1:1:1 ratio to treatment with 36-80 weeks of standard care; 24-week oral bedaquiline, pretomanid, and linezolid (BPaL); BPaL plus clofazimine (BPaLC); or BPaL plus moxifloxacin (BPaLM) . This was followed by stage two of the trial, in which participants were randomized 1:1 to receive standard care or BPaLM. The current study, published in The Lancet Respiratory Medicine, reported the stage two findings; the primary outcome was a composite of unfavorable outcomes at 72 weeks including death, treatment failure, treatment discontinuation, recurrence of tuberculosis, or loss to follow-up.
The modified intent-to-treat population included 138 patients in the BPaLM group and 137 patients in the standard care group. In this population, 56 (41%) of 137 participants in the standard care group and 16 (12%) of 137 participants in the BPaLM group met criteria for the unfavorable outcome at 72 weeks; noninferiority and superiority were significantly greater in the BPaLM group (P < .0001).
Early discontinuation was the main reason patients met the unfavorable outcome criteria (89% of standard care patients and 69% of BPaLM patients); adverse events accounted for 23% of discontinuations in the standard care group and 64% of discontinuations in the BPaLM group.
However, fewer patients in the BPaLM group experienced grade 3 or higher adverse events compared with the standard care group (23% vs. 48%). The most common adverse events included hepatic disorders, cardiac disorders, and anemia.
In addition, all subgroup analyses favored BPaLM over standard care at 72 weeks including subgroups based on sex, age, disease severity, re-treatment status, and smoking status.
The findings were limited by several factors including the changes to standard of care over the course of the study, potential bias because the study was stopped for efficacy, and inclusion of loss to follow-up as part of the composite unfavorable outcome, the researchers noted.
Remaining research questions include the optimal dose of linezolid, whether use of alternative fluoroquinolones would yield similar results, and whether the results would generalize to populations including children, pregnant women, and patients with extrapulmonary tuberculosis, they added.
However, the results support BPaLM as the preferred treatment for adults and adolescents with pulmonary rifampin-resistant TB, the researchers concluded.
BPaLM poised to improve TB care
Before 2020, treatment for rifampin-resistant tuberculosis was 9-20 months in duration, toxic, and inadequately effective, and new treatment regimens are urgently needed, Mary Jo Farmer, MD, a pulmonary and critical care specialist at the University of Massachusetts Baystate Health Regional Campus, Springfield, said in an interview.
“The BPaL-based regimens perform better than the 9- to 20-month standard of care, are shorter in duration, have a lower pill burden, improve quality of life, and are cost-effective,” she said. “The BPaL regimens have the potential to improve outcomes for thousands of patients with rifampin-resistant tuberculosis.”
“The 24-week oral regimen consisting of bedaquiline, pretomanid, linezolid and moxifloxacin is noninferior to standard of care for treatment of patients with pulmonary rifampin-resistant tuberculosis, and this BPaLM regimen was added to the WHO guidance for treatment of this condition in 2022,” said Dr. Farmer, who was not involved in the study. “It remains to be seen if BPaLM will become the preferred regimen for adolescents and adults with pulmonary rifampin-resistant tuberculosis,” she said.
Dr. Farmer agreed with the study authors that the optimal dose of linezolid, optimal duration of treatment, and the role of dose reduction remain unknown, and pharmacokinetic studies are needed to identify these parameters.
The study was supported by Médecins Sans Frontières. TB Alliance donated pretomanid to the study prior to its commercialization. The researchers had no financial conflicts to disclose. Dr. Farmer had no financial conflicts to disclose, but serves on the editorial advisory board of CHEST Physician.
A combination oral-only therapy of bedaquiline, pretomanid, and linezolid was significantly more effective than standard care in preventing unfavorable outcomes in patients with treatment-resistant tuberculosis, based on data from more than 500 individuals.
, and data on the use of new and repurposed drug combinations are lacking, wrote Bern-Thomas Nyang’wa, MBBS, of Médecins Sans Frontières, Amsterdam, and colleagues.
In a study known as the TB-PRACTECAL trial, the researchers enrolled 552 pulmonary rifampin-resistant tuberculosis patients aged 15 years and older to examine several new and repurposed drug combinations. The participants were randomized in a 1:1:1:1 ratio to treatment with 36-80 weeks of standard care; 24-week oral bedaquiline, pretomanid, and linezolid (BPaL); BPaL plus clofazimine (BPaLC); or BPaL plus moxifloxacin (BPaLM) . This was followed by stage two of the trial, in which participants were randomized 1:1 to receive standard care or BPaLM. The current study, published in The Lancet Respiratory Medicine, reported the stage two findings; the primary outcome was a composite of unfavorable outcomes at 72 weeks including death, treatment failure, treatment discontinuation, recurrence of tuberculosis, or loss to follow-up.
The modified intent-to-treat population included 138 patients in the BPaLM group and 137 patients in the standard care group. In this population, 56 (41%) of 137 participants in the standard care group and 16 (12%) of 137 participants in the BPaLM group met criteria for the unfavorable outcome at 72 weeks; noninferiority and superiority were significantly greater in the BPaLM group (P < .0001).
Early discontinuation was the main reason patients met the unfavorable outcome criteria (89% of standard care patients and 69% of BPaLM patients); adverse events accounted for 23% of discontinuations in the standard care group and 64% of discontinuations in the BPaLM group.
However, fewer patients in the BPaLM group experienced grade 3 or higher adverse events compared with the standard care group (23% vs. 48%). The most common adverse events included hepatic disorders, cardiac disorders, and anemia.
In addition, all subgroup analyses favored BPaLM over standard care at 72 weeks including subgroups based on sex, age, disease severity, re-treatment status, and smoking status.
The findings were limited by several factors including the changes to standard of care over the course of the study, potential bias because the study was stopped for efficacy, and inclusion of loss to follow-up as part of the composite unfavorable outcome, the researchers noted.
Remaining research questions include the optimal dose of linezolid, whether use of alternative fluoroquinolones would yield similar results, and whether the results would generalize to populations including children, pregnant women, and patients with extrapulmonary tuberculosis, they added.
However, the results support BPaLM as the preferred treatment for adults and adolescents with pulmonary rifampin-resistant TB, the researchers concluded.
BPaLM poised to improve TB care
Before 2020, treatment for rifampin-resistant tuberculosis was 9-20 months in duration, toxic, and inadequately effective, and new treatment regimens are urgently needed, Mary Jo Farmer, MD, a pulmonary and critical care specialist at the University of Massachusetts Baystate Health Regional Campus, Springfield, said in an interview.
“The BPaL-based regimens perform better than the 9- to 20-month standard of care, are shorter in duration, have a lower pill burden, improve quality of life, and are cost-effective,” she said. “The BPaL regimens have the potential to improve outcomes for thousands of patients with rifampin-resistant tuberculosis.”
“The 24-week oral regimen consisting of bedaquiline, pretomanid, linezolid and moxifloxacin is noninferior to standard of care for treatment of patients with pulmonary rifampin-resistant tuberculosis, and this BPaLM regimen was added to the WHO guidance for treatment of this condition in 2022,” said Dr. Farmer, who was not involved in the study. “It remains to be seen if BPaLM will become the preferred regimen for adolescents and adults with pulmonary rifampin-resistant tuberculosis,” she said.
Dr. Farmer agreed with the study authors that the optimal dose of linezolid, optimal duration of treatment, and the role of dose reduction remain unknown, and pharmacokinetic studies are needed to identify these parameters.
The study was supported by Médecins Sans Frontières. TB Alliance donated pretomanid to the study prior to its commercialization. The researchers had no financial conflicts to disclose. Dr. Farmer had no financial conflicts to disclose, but serves on the editorial advisory board of CHEST Physician.
FROM LANCET RESPIRATORY MEDICINE
